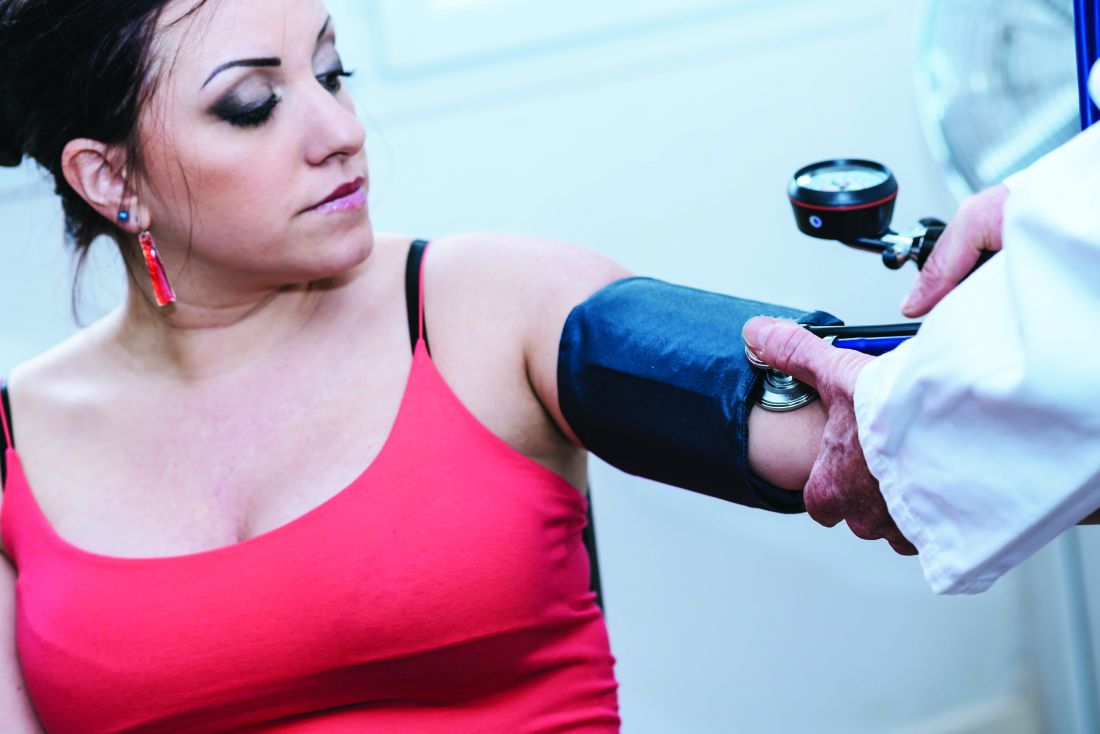
User login
No signal for CV, breast effects with bioidentical vaginal estrogen for dyspareunia
that would suggest significant systemic absorption.
The lack of sex hormone binding globulin (SHBG) changes in the subset of women who received this test bolsters support for low systemic absorption from the low-dose vaginal softgel, Lisa Larkin, MD, said at the annual meeting of the North American Menopause Society in Orlando.
These safety data show that the vaginal route for this hormone is meeting a treatment goal for many menopausal women: “One goal of vaginal estrogen is to minimize systemic absorption and potentially reduce related side effects,” Dr. Larkin said.
TX-004HR (Imvexxy) delivers bioidentical solubilized 17 beta-estradiol (E2) via a softgel vaginal insert. It is Food and Drug Administration approved in 4-mcg and 10-mcg doses for the treatment of moderate to severe dyspareunia associated with menopause.
The phase 3 clinical trial (REJOICE) of TX-004HR met the coprimary endpoints of improving vaginal physiology, lowering vaginal pH, and decreasing the severity of dyspareunia at both the 4- and 10-mcg doses, said Dr. Larkin, an internal medicine physician in private practice in Mariemont, Ohio.
Serum estradiol levels for REJOICE participants were “similar to placebo and baseline, and generally within the postmenopausal range,” she said.
The randomized, double-blind, placebo-controlled trial tested 4-, 10-, and 25-mcg doses of TX-004HR. The self-administered vaginal inserts were used once daily for 2 weeks, then twice weekly for an additional 10 weeks.
In looking at treatment emergent adverse events (TEAEs), the REJOICE investigators were particularly interested in tracking cardiovascular and breast events, Dr. Larkin said. Participants received ECGs and clinical breast exams at baseline, and at study week 12. In addition, 72 of the women had SHBG measured at baseline and at weeks 2 and 12. The trial had a high completion rate of 94% at 12 weeks. The mean age of the women was 59 years, and the mean body mass index was 26.7 kg/m2. African American women made up 12% of the study; the remainder of the women were white.
In the end, 784 menopausal women with moderate to severe dyspareunia were randomized 1:1:1:1 to placebo or to receive one of the three dose levels of TX-004HR. Overall, “no clinically significant differences in adverse events were observed between treatment and placebo groups,” Dr. Larkin said. Only headache, vaginal discharge, and vulvovaginal pruritus occurred in at least 3% of the women in any treatment arm, with no differences between those taking TX-004HR and placebo. There were no malignancies or endometrial hyperplasia among the REJOICE participants: “There was no signal of estrogenic stimulation of the endometrium,” she said.
Looking at cardiovascular-related TEAEs, the five events that occurred were judged to be mild, and mostly not related to treatment. One case of first degree atrioventricular block and one case of sinus bradycardia were reported by the same woman, who was taking the 4-mcg dose of TX-004HR. One additional woman on that dose experienced palpitations, as did one woman taking placebo. “No coronary heart disease, venous thromboembolism, or other thrombotic episodes were noted” during the REJOICE trial, Dr. Larkin said. There were no clinically significant ECG changes during the study period that were judged related to treatment. Blood pressure was mildly increased in three women, one each in the 4-mcg, 10-mcg, and placebo study arms. The elevation was considered possibly related to the study in the 4-mcg and placebo takers. Two other women in the 4-mcg group experienced mild incident hypertension, with one woman’s hypertension judged possibly related to treatment.
Blood chemistry showed incident hypercholesterolemia for one woman in the 4-mcg group and one in the placebo group, and one woman taking the 10-mcg TX-0400HR dose and two taking placebo had increases in serum triglycerides.
Seven women reported breast-related TEAEs, with five of these considered possibly or probably treatment related. One woman on the 10-mcg dose had breast tenderness; all other events were among placebo takers.
Finally, among the subset of women whose SHBG levels were tested, “no dose-related pattern was apparent, and changes with TX-004HR were comparable to changes with placebo,” said Dr. Larkin, noting that there was no suggestion of significant systemic absorption.
“These safety data, in conjunction with the improved moderate to severe dyspareunia efficacy data and minimal estradiol absorption, support a local effect of the TX-004HR vaginal insert,” she said.
The study was sponsored by TherapeuticsMD, the manufacturer of TH-004HR. Dr. Larkin disclosed that she is an advisory board member and on the speaker’s bureau for Valeant pharmaceuticals, is a consultant for TherapeuticsMD, and is an advisory board member for AMAG and Palatin Technologies.
SOURCE: Larkin L et al. NAMS 2018, Thursday concurrent session 1.
that would suggest significant systemic absorption.
The lack of sex hormone binding globulin (SHBG) changes in the subset of women who received this test bolsters support for low systemic absorption from the low-dose vaginal softgel, Lisa Larkin, MD, said at the annual meeting of the North American Menopause Society in Orlando.
These safety data show that the vaginal route for this hormone is meeting a treatment goal for many menopausal women: “One goal of vaginal estrogen is to minimize systemic absorption and potentially reduce related side effects,” Dr. Larkin said.
TX-004HR (Imvexxy) delivers bioidentical solubilized 17 beta-estradiol (E2) via a softgel vaginal insert. It is Food and Drug Administration approved in 4-mcg and 10-mcg doses for the treatment of moderate to severe dyspareunia associated with menopause.
The phase 3 clinical trial (REJOICE) of TX-004HR met the coprimary endpoints of improving vaginal physiology, lowering vaginal pH, and decreasing the severity of dyspareunia at both the 4- and 10-mcg doses, said Dr. Larkin, an internal medicine physician in private practice in Mariemont, Ohio.
Serum estradiol levels for REJOICE participants were “similar to placebo and baseline, and generally within the postmenopausal range,” she said.
The randomized, double-blind, placebo-controlled trial tested 4-, 10-, and 25-mcg doses of TX-004HR. The self-administered vaginal inserts were used once daily for 2 weeks, then twice weekly for an additional 10 weeks.
In looking at treatment emergent adverse events (TEAEs), the REJOICE investigators were particularly interested in tracking cardiovascular and breast events, Dr. Larkin said. Participants received ECGs and clinical breast exams at baseline, and at study week 12. In addition, 72 of the women had SHBG measured at baseline and at weeks 2 and 12. The trial had a high completion rate of 94% at 12 weeks. The mean age of the women was 59 years, and the mean body mass index was 26.7 kg/m2. African American women made up 12% of the study; the remainder of the women were white.
In the end, 784 menopausal women with moderate to severe dyspareunia were randomized 1:1:1:1 to placebo or to receive one of the three dose levels of TX-004HR. Overall, “no clinically significant differences in adverse events were observed between treatment and placebo groups,” Dr. Larkin said. Only headache, vaginal discharge, and vulvovaginal pruritus occurred in at least 3% of the women in any treatment arm, with no differences between those taking TX-004HR and placebo. There were no malignancies or endometrial hyperplasia among the REJOICE participants: “There was no signal of estrogenic stimulation of the endometrium,” she said.
Looking at cardiovascular-related TEAEs, the five events that occurred were judged to be mild, and mostly not related to treatment. One case of first degree atrioventricular block and one case of sinus bradycardia were reported by the same woman, who was taking the 4-mcg dose of TX-004HR. One additional woman on that dose experienced palpitations, as did one woman taking placebo. “No coronary heart disease, venous thromboembolism, or other thrombotic episodes were noted” during the REJOICE trial, Dr. Larkin said. There were no clinically significant ECG changes during the study period that were judged related to treatment. Blood pressure was mildly increased in three women, one each in the 4-mcg, 10-mcg, and placebo study arms. The elevation was considered possibly related to the study in the 4-mcg and placebo takers. Two other women in the 4-mcg group experienced mild incident hypertension, with one woman’s hypertension judged possibly related to treatment.
Blood chemistry showed incident hypercholesterolemia for one woman in the 4-mcg group and one in the placebo group, and one woman taking the 10-mcg TX-0400HR dose and two taking placebo had increases in serum triglycerides.
Seven women reported breast-related TEAEs, with five of these considered possibly or probably treatment related. One woman on the 10-mcg dose had breast tenderness; all other events were among placebo takers.
Finally, among the subset of women whose SHBG levels were tested, “no dose-related pattern was apparent, and changes with TX-004HR were comparable to changes with placebo,” said Dr. Larkin, noting that there was no suggestion of significant systemic absorption.
“These safety data, in conjunction with the improved moderate to severe dyspareunia efficacy data and minimal estradiol absorption, support a local effect of the TX-004HR vaginal insert,” she said.
The study was sponsored by TherapeuticsMD, the manufacturer of TH-004HR. Dr. Larkin disclosed that she is an advisory board member and on the speaker’s bureau for Valeant pharmaceuticals, is a consultant for TherapeuticsMD, and is an advisory board member for AMAG and Palatin Technologies.
SOURCE: Larkin L et al. NAMS 2018, Thursday concurrent session 1.
that would suggest significant systemic absorption.
The lack of sex hormone binding globulin (SHBG) changes in the subset of women who received this test bolsters support for low systemic absorption from the low-dose vaginal softgel, Lisa Larkin, MD, said at the annual meeting of the North American Menopause Society in Orlando.
These safety data show that the vaginal route for this hormone is meeting a treatment goal for many menopausal women: “One goal of vaginal estrogen is to minimize systemic absorption and potentially reduce related side effects,” Dr. Larkin said.
TX-004HR (Imvexxy) delivers bioidentical solubilized 17 beta-estradiol (E2) via a softgel vaginal insert. It is Food and Drug Administration approved in 4-mcg and 10-mcg doses for the treatment of moderate to severe dyspareunia associated with menopause.
The phase 3 clinical trial (REJOICE) of TX-004HR met the coprimary endpoints of improving vaginal physiology, lowering vaginal pH, and decreasing the severity of dyspareunia at both the 4- and 10-mcg doses, said Dr. Larkin, an internal medicine physician in private practice in Mariemont, Ohio.
Serum estradiol levels for REJOICE participants were “similar to placebo and baseline, and generally within the postmenopausal range,” she said.
The randomized, double-blind, placebo-controlled trial tested 4-, 10-, and 25-mcg doses of TX-004HR. The self-administered vaginal inserts were used once daily for 2 weeks, then twice weekly for an additional 10 weeks.
In looking at treatment emergent adverse events (TEAEs), the REJOICE investigators were particularly interested in tracking cardiovascular and breast events, Dr. Larkin said. Participants received ECGs and clinical breast exams at baseline, and at study week 12. In addition, 72 of the women had SHBG measured at baseline and at weeks 2 and 12. The trial had a high completion rate of 94% at 12 weeks. The mean age of the women was 59 years, and the mean body mass index was 26.7 kg/m2. African American women made up 12% of the study; the remainder of the women were white.
In the end, 784 menopausal women with moderate to severe dyspareunia were randomized 1:1:1:1 to placebo or to receive one of the three dose levels of TX-004HR. Overall, “no clinically significant differences in adverse events were observed between treatment and placebo groups,” Dr. Larkin said. Only headache, vaginal discharge, and vulvovaginal pruritus occurred in at least 3% of the women in any treatment arm, with no differences between those taking TX-004HR and placebo. There were no malignancies or endometrial hyperplasia among the REJOICE participants: “There was no signal of estrogenic stimulation of the endometrium,” she said.
Looking at cardiovascular-related TEAEs, the five events that occurred were judged to be mild, and mostly not related to treatment. One case of first degree atrioventricular block and one case of sinus bradycardia were reported by the same woman, who was taking the 4-mcg dose of TX-004HR. One additional woman on that dose experienced palpitations, as did one woman taking placebo. “No coronary heart disease, venous thromboembolism, or other thrombotic episodes were noted” during the REJOICE trial, Dr. Larkin said. There were no clinically significant ECG changes during the study period that were judged related to treatment. Blood pressure was mildly increased in three women, one each in the 4-mcg, 10-mcg, and placebo study arms. The elevation was considered possibly related to the study in the 4-mcg and placebo takers. Two other women in the 4-mcg group experienced mild incident hypertension, with one woman’s hypertension judged possibly related to treatment.
Blood chemistry showed incident hypercholesterolemia for one woman in the 4-mcg group and one in the placebo group, and one woman taking the 10-mcg TX-0400HR dose and two taking placebo had increases in serum triglycerides.
Seven women reported breast-related TEAEs, with five of these considered possibly or probably treatment related. One woman on the 10-mcg dose had breast tenderness; all other events were among placebo takers.
Finally, among the subset of women whose SHBG levels were tested, “no dose-related pattern was apparent, and changes with TX-004HR were comparable to changes with placebo,” said Dr. Larkin, noting that there was no suggestion of significant systemic absorption.
“These safety data, in conjunction with the improved moderate to severe dyspareunia efficacy data and minimal estradiol absorption, support a local effect of the TX-004HR vaginal insert,” she said.
The study was sponsored by TherapeuticsMD, the manufacturer of TH-004HR. Dr. Larkin disclosed that she is an advisory board member and on the speaker’s bureau for Valeant pharmaceuticals, is a consultant for TherapeuticsMD, and is an advisory board member for AMAG and Palatin Technologies.
SOURCE: Larkin L et al. NAMS 2018, Thursday concurrent session 1.
FROM NAMS 2018
Key clinical point: Safety data from clinical trials of a bioidentical vaginal estrogen for dyspareunia in menopausal women showed no signs of CV or breast risks.
Major finding: There were no cardiovascular events or thrombotic episodes among menopausal women with dyspareunia treated with TX-004HR.
Study details: Randomized, double-blind, placebo-controlled trial of 784 menopausal women with moderate to severe dyspareunia.
Disclosures: The study was sponsored by TherapeuticsMD, the manufacturer of TH-004HR. Dr. Larkin reported financial relationships with several pharmaceutical companies, including TherapeuticsMD.
Source: Larkin L et al. NAMS 2018, Thursday concurrent session 1.
With more mindfulness, menopausal symptoms wane
An observational Furthermore, mindfulness had the greatest positive effect on menopausal symptoms for those women with the highest self-reported stress levels.
“In this cross-sectional study, mindfulness was associated with lower menopausal symptom burden. In women with higher stress, the magnitude of association between mindfulness and menopausal symptoms appeared more robust,” said Richa Sood, MD, speaking at the annual meeting of the North American Menopause Society.
Menopausal symptoms can exist alongside many other midlife issues because women often are trying to keep many balls in the air: This age group may be facing aging parents, a household with teenagers, and work-related pressures, she noted.
Thus, menopausal symptoms can be amplified by stressors. New mood problems – or worsening of preexisting ones – can interfere with work productivity and negatively affect relationships. Life satisfaction can take a steep dive during midlife for some women, said Dr. Sood of the Mayo Clinic, Rochester, Minn.
Could mindfulness be effective for stress management in this complex landscape of physiological and lifespan changes? “Mindfulness is paying attention,” said Dr. Sood. Practitioners of mindfulness focus on purposeful attention, staying in the present moment, and avoiding judgment.
Mindfulness may work as a stress-management tool for a variety of reasons, said Dr. Sood. The technique can help retrain people with tendencies for emotional reactivity in stressful situations; additionally, the focus on the present and on observation, rather than judgment, may help avoid maladaptive rumination.
To see how mindfulness in everyday life could affect the burden of menopausal symptoms, Dr. Sood and her collaborators designed an observational, cross-sectional study of 1,744 women aged 40-64 years.
The investigators used three scales in their assessments. The first, the Menopause Rating Scale (MRS), is an 11-item scale ranging from 0 to 44 that assesses psychological, somatovegetative, and urogenital domains. The second, the Perceived Stress Scale 4 (PSS-4), is a four-item scale that is a global measure of stress over the last 4 weeks, with tallied scores in the 0-16 range. Finally, the Mindful Attention Awareness Scale (MAAS) is a 15-item scale that captures how frequently respondents are in a mindful state during their daily life, with higher scores meaning more mindfulness.
The 1,744 women were seen in a women’s health clinic over the period of one year. Participants were a mean 53.4 years old (standard deviation, 6.1 years). Almost all were white (93%), most were married (82.7%), and most also had at least a 4-year college degree (64.6%) and were employed (65.3%).
The investigators mapped each scale against each of the others, which yielded three plots. In the first, higher MAAS scores were correlated with lower MRS scores (correlation, –0.49; P less than .001), which means that more time in a mindful state was associated with less severe menopausal symptoms.
In the second plot, lower MRS scores were associated with lower PSS-4 scores (correlation, 0.55; P less than .001). The last plot mapped PSS-4 scores against MAAS scores, showing that higher MAAS scores were correlated with lower PSS-4 scores, which means that more time in a mindful state was also associated with less perceived stress (correlation, –0.53; P less than .001).
Most of these associations remained statistically significant after multivariable linear regression analysis and breaking out the subscales of the MRS. Only the association between higher MAAS scores and the somatovegetative domain of the PSS-4 lost significance (P = 0.44).
Next, Dr. Sood and her collaborators probed how higher perceived stress, as measured by higher PSS-4 scores, altered the effects that mindful activity had on menopausal symptoms (as measured by the MRS).
The effect of mindfulness became stronger in the milieu of higher perceived stress. At a relatively low PSS-4 value of 4, the MRS score dropped 1.53 points for each one-point increase in the MAAS total score. However, with a PSS-4 score of 12, the decrease in MRS was 2.27 points for each one-point increase in MAAS, and with the maximum perceived stress score of 16, the MRS fell 2.64 points for each one-point increase in the MAAS score.
These findings are set against the backdrop of previous literature linking mindfulness to positive health behaviors and outcomes, she said, noting that work looking specifically at mindfulness-based stress reduction in peri- and postmenopausal women showed a halving of symptoms and improved quality of life.
Dr. Sood said that the present study was observational only, noting that it looked only at time spent in a mindful state in an untrained cohort of women in midlife. “Trait, or dispositional, mindfulness appears to be protective against stress and symptoms in midlife women,” she commented. “More mindful women may be choosing to shift their attention to more pleasant aspects of life, rather than their symptoms.”
“If you allow me to speculate a bit,” Dr. Sood continued, “the underpinnings of psychological symptoms rest in threat-focused attention and emotional reactivity – so the mindfulness approach appears to fit very well to impact such a change.”
Mindfulness, she added, “might be a tool to impact the emotional component of the overall experience, thereby decreasing the total suffering.” However, she noted that what’s needed are studies with a more heterogeneous population, as well as ones designed to tease out causality. Still, “the current study adds to the wealth of data supporting the role of mindfulness in various settings for impacting positive change in health and behaviors.”
Dr. Sood reported that she has ownership stake in the Global Center for Resiliency and Well-Being.
SOURCE: Sood R et al. NAMS 2018, Top-Scoring Abstract Session.
An observational Furthermore, mindfulness had the greatest positive effect on menopausal symptoms for those women with the highest self-reported stress levels.
“In this cross-sectional study, mindfulness was associated with lower menopausal symptom burden. In women with higher stress, the magnitude of association between mindfulness and menopausal symptoms appeared more robust,” said Richa Sood, MD, speaking at the annual meeting of the North American Menopause Society.
Menopausal symptoms can exist alongside many other midlife issues because women often are trying to keep many balls in the air: This age group may be facing aging parents, a household with teenagers, and work-related pressures, she noted.
Thus, menopausal symptoms can be amplified by stressors. New mood problems – or worsening of preexisting ones – can interfere with work productivity and negatively affect relationships. Life satisfaction can take a steep dive during midlife for some women, said Dr. Sood of the Mayo Clinic, Rochester, Minn.
Could mindfulness be effective for stress management in this complex landscape of physiological and lifespan changes? “Mindfulness is paying attention,” said Dr. Sood. Practitioners of mindfulness focus on purposeful attention, staying in the present moment, and avoiding judgment.
Mindfulness may work as a stress-management tool for a variety of reasons, said Dr. Sood. The technique can help retrain people with tendencies for emotional reactivity in stressful situations; additionally, the focus on the present and on observation, rather than judgment, may help avoid maladaptive rumination.
To see how mindfulness in everyday life could affect the burden of menopausal symptoms, Dr. Sood and her collaborators designed an observational, cross-sectional study of 1,744 women aged 40-64 years.
The investigators used three scales in their assessments. The first, the Menopause Rating Scale (MRS), is an 11-item scale ranging from 0 to 44 that assesses psychological, somatovegetative, and urogenital domains. The second, the Perceived Stress Scale 4 (PSS-4), is a four-item scale that is a global measure of stress over the last 4 weeks, with tallied scores in the 0-16 range. Finally, the Mindful Attention Awareness Scale (MAAS) is a 15-item scale that captures how frequently respondents are in a mindful state during their daily life, with higher scores meaning more mindfulness.
The 1,744 women were seen in a women’s health clinic over the period of one year. Participants were a mean 53.4 years old (standard deviation, 6.1 years). Almost all were white (93%), most were married (82.7%), and most also had at least a 4-year college degree (64.6%) and were employed (65.3%).
The investigators mapped each scale against each of the others, which yielded three plots. In the first, higher MAAS scores were correlated with lower MRS scores (correlation, –0.49; P less than .001), which means that more time in a mindful state was associated with less severe menopausal symptoms.
In the second plot, lower MRS scores were associated with lower PSS-4 scores (correlation, 0.55; P less than .001). The last plot mapped PSS-4 scores against MAAS scores, showing that higher MAAS scores were correlated with lower PSS-4 scores, which means that more time in a mindful state was also associated with less perceived stress (correlation, –0.53; P less than .001).
Most of these associations remained statistically significant after multivariable linear regression analysis and breaking out the subscales of the MRS. Only the association between higher MAAS scores and the somatovegetative domain of the PSS-4 lost significance (P = 0.44).
Next, Dr. Sood and her collaborators probed how higher perceived stress, as measured by higher PSS-4 scores, altered the effects that mindful activity had on menopausal symptoms (as measured by the MRS).
The effect of mindfulness became stronger in the milieu of higher perceived stress. At a relatively low PSS-4 value of 4, the MRS score dropped 1.53 points for each one-point increase in the MAAS total score. However, with a PSS-4 score of 12, the decrease in MRS was 2.27 points for each one-point increase in MAAS, and with the maximum perceived stress score of 16, the MRS fell 2.64 points for each one-point increase in the MAAS score.
These findings are set against the backdrop of previous literature linking mindfulness to positive health behaviors and outcomes, she said, noting that work looking specifically at mindfulness-based stress reduction in peri- and postmenopausal women showed a halving of symptoms and improved quality of life.
Dr. Sood said that the present study was observational only, noting that it looked only at time spent in a mindful state in an untrained cohort of women in midlife. “Trait, or dispositional, mindfulness appears to be protective against stress and symptoms in midlife women,” she commented. “More mindful women may be choosing to shift their attention to more pleasant aspects of life, rather than their symptoms.”
“If you allow me to speculate a bit,” Dr. Sood continued, “the underpinnings of psychological symptoms rest in threat-focused attention and emotional reactivity – so the mindfulness approach appears to fit very well to impact such a change.”
Mindfulness, she added, “might be a tool to impact the emotional component of the overall experience, thereby decreasing the total suffering.” However, she noted that what’s needed are studies with a more heterogeneous population, as well as ones designed to tease out causality. Still, “the current study adds to the wealth of data supporting the role of mindfulness in various settings for impacting positive change in health and behaviors.”
Dr. Sood reported that she has ownership stake in the Global Center for Resiliency and Well-Being.
SOURCE: Sood R et al. NAMS 2018, Top-Scoring Abstract Session.
An observational Furthermore, mindfulness had the greatest positive effect on menopausal symptoms for those women with the highest self-reported stress levels.
“In this cross-sectional study, mindfulness was associated with lower menopausal symptom burden. In women with higher stress, the magnitude of association between mindfulness and menopausal symptoms appeared more robust,” said Richa Sood, MD, speaking at the annual meeting of the North American Menopause Society.
Menopausal symptoms can exist alongside many other midlife issues because women often are trying to keep many balls in the air: This age group may be facing aging parents, a household with teenagers, and work-related pressures, she noted.
Thus, menopausal symptoms can be amplified by stressors. New mood problems – or worsening of preexisting ones – can interfere with work productivity and negatively affect relationships. Life satisfaction can take a steep dive during midlife for some women, said Dr. Sood of the Mayo Clinic, Rochester, Minn.
Could mindfulness be effective for stress management in this complex landscape of physiological and lifespan changes? “Mindfulness is paying attention,” said Dr. Sood. Practitioners of mindfulness focus on purposeful attention, staying in the present moment, and avoiding judgment.
Mindfulness may work as a stress-management tool for a variety of reasons, said Dr. Sood. The technique can help retrain people with tendencies for emotional reactivity in stressful situations; additionally, the focus on the present and on observation, rather than judgment, may help avoid maladaptive rumination.
To see how mindfulness in everyday life could affect the burden of menopausal symptoms, Dr. Sood and her collaborators designed an observational, cross-sectional study of 1,744 women aged 40-64 years.
The investigators used three scales in their assessments. The first, the Menopause Rating Scale (MRS), is an 11-item scale ranging from 0 to 44 that assesses psychological, somatovegetative, and urogenital domains. The second, the Perceived Stress Scale 4 (PSS-4), is a four-item scale that is a global measure of stress over the last 4 weeks, with tallied scores in the 0-16 range. Finally, the Mindful Attention Awareness Scale (MAAS) is a 15-item scale that captures how frequently respondents are in a mindful state during their daily life, with higher scores meaning more mindfulness.
The 1,744 women were seen in a women’s health clinic over the period of one year. Participants were a mean 53.4 years old (standard deviation, 6.1 years). Almost all were white (93%), most were married (82.7%), and most also had at least a 4-year college degree (64.6%) and were employed (65.3%).
The investigators mapped each scale against each of the others, which yielded three plots. In the first, higher MAAS scores were correlated with lower MRS scores (correlation, –0.49; P less than .001), which means that more time in a mindful state was associated with less severe menopausal symptoms.
In the second plot, lower MRS scores were associated with lower PSS-4 scores (correlation, 0.55; P less than .001). The last plot mapped PSS-4 scores against MAAS scores, showing that higher MAAS scores were correlated with lower PSS-4 scores, which means that more time in a mindful state was also associated with less perceived stress (correlation, –0.53; P less than .001).
Most of these associations remained statistically significant after multivariable linear regression analysis and breaking out the subscales of the MRS. Only the association between higher MAAS scores and the somatovegetative domain of the PSS-4 lost significance (P = 0.44).
Next, Dr. Sood and her collaborators probed how higher perceived stress, as measured by higher PSS-4 scores, altered the effects that mindful activity had on menopausal symptoms (as measured by the MRS).
The effect of mindfulness became stronger in the milieu of higher perceived stress. At a relatively low PSS-4 value of 4, the MRS score dropped 1.53 points for each one-point increase in the MAAS total score. However, with a PSS-4 score of 12, the decrease in MRS was 2.27 points for each one-point increase in MAAS, and with the maximum perceived stress score of 16, the MRS fell 2.64 points for each one-point increase in the MAAS score.
These findings are set against the backdrop of previous literature linking mindfulness to positive health behaviors and outcomes, she said, noting that work looking specifically at mindfulness-based stress reduction in peri- and postmenopausal women showed a halving of symptoms and improved quality of life.
Dr. Sood said that the present study was observational only, noting that it looked only at time spent in a mindful state in an untrained cohort of women in midlife. “Trait, or dispositional, mindfulness appears to be protective against stress and symptoms in midlife women,” she commented. “More mindful women may be choosing to shift their attention to more pleasant aspects of life, rather than their symptoms.”
“If you allow me to speculate a bit,” Dr. Sood continued, “the underpinnings of psychological symptoms rest in threat-focused attention and emotional reactivity – so the mindfulness approach appears to fit very well to impact such a change.”
Mindfulness, she added, “might be a tool to impact the emotional component of the overall experience, thereby decreasing the total suffering.” However, she noted that what’s needed are studies with a more heterogeneous population, as well as ones designed to tease out causality. Still, “the current study adds to the wealth of data supporting the role of mindfulness in various settings for impacting positive change in health and behaviors.”
Dr. Sood reported that she has ownership stake in the Global Center for Resiliency and Well-Being.
SOURCE: Sood R et al. NAMS 2018, Top-Scoring Abstract Session.
FROM NAMS 2018
Key clinical point: Time spent in a mindful state was associated with fewer menopause symptoms.
Major finding: With maximum stress, each 1-point increase in mindfulness was associated with a 2.64-point drop in menopausal symptoms.
Study details: A cross-sectional, single-center study of 1,744 women aged 40-64 years.
Disclosures: Dr. Sood reported that she has ownership stake in the Global Center for Resiliency and Well-Being.
Source: Sood R et al. NAMS 2018, Top-Scoring Abstract Session.
Delayed pushing during labor does not benefit mother or baby, study finds
according to the results of a randomized trial.
“The optimal technique for managing maternal pushing during the second stage of labor is unknown,” wrote Alison G. Cahill, MD, of Washington University, St. Louis, and her coauthors, who published their study in JAMA on Oct. 9. “The two most common approaches to the second stage of labor management are to either initiate pushing with uterine contractions once complete cervical dilation occurs (immediate pushing) or to allow for spontaneous descent (delayed pushing),” they noted. “Both approaches are commonly used, and neither is considered the gold standard.”
They addressed this question in the multicenter trial of nulliparous women (mean age, 26.5 years) who, during May 2014 to December 2017, were at or past 37 weeks’ gestation and had received neuraxial analgesia. The primary outcome was the rate of spontaneous vaginal delivery; secondary outcomes included maternal and neonatal morbidity outcomes. When they reached complete cervical dilation, women were randomized to immediate pushing or delayed pushing, in which they were instructed to wait 60 minutes.
The study was terminated after the data and safety monitoring board conducted a planned interim analysis; the analysis found futility in the delayed pushing group and raised concerns about increased morbidity in that group.
Among the 1,031 women in the immediate pushing group, the rate of spontaneous vaginal delivery was 85.9%; the rate was 86.5% among the 1,041 women in the delayed pushing group (P = .67).
The mean duration of the second stage of labor was significantly shorter in the immediate pushing group (102.4 minutes), compared with that seen in the delayed pushing group (134.2 minutes; P less than .001). The mean duration of active pushing was significantly longer in the immediate pushing group (83.7 minutes), compared with that seen in the delayed pushing group (74.5 minutes; P less than .001).
In terms of secondary outcomes, rates of postpartum hemorrhage were lower in the immediate pushing group (2.3%), compared with the rate among those in the delayed pushing group (4%; P = .03). During the second stage of labor, chorioamnionitis was significantly more common among women in the delayed pushing group (9.1%), compared with rate among women in the immediate pushing group (6.7%; P = .005). There was no significant difference between the two groups in the rates of a composite neonatal morbidity outcome (which included birth injury, respiratory distress, and neonatal acidemia), which was 7.3% in the immediate pushing group and 9.9% in the delayed pushing group. There were no neonatal deaths.
Among prespecified exploratory outcomes, the rates of neonatal acidemia and suspected neonatal sepsis were significantly higher in the delayed pushing group, whereas the rate of third-degree perineal lacerations was significantly higher in the immediate pushing group.
“The finding of no effect on spontaneous vaginal delivery for pushing timing during the second stage of labor and the evidence suggesting increased maternal and neonatal complications in the delayed pushing group support the view that women immediately pushing after complete cervical dilation may be preferred because women without neuraxial analgesia reflexively push immediately,” the authors pointed out. Their results, they concluded, “may help inform decisions about the preferred timing of second stage pushing efforts when considered with other maternal and neonatal outcomes.”
They noted that the trial had several limitations, including the unblinded design, which raised the possibility of bias that may have influenced the management of patients or diagnoses.
The authors had no disclosures. The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and by funding from the department of obstetrics and gynecology at Washington University, St. Louis.
SOURCE: Cahill AG et al. JAMA. 2018;320(14):1444-54.
Of the almost 4 million births in the United States each year, 68% are vaginal deliveries, which illustrates how important it is to have additional evidence to guide the management of labor, Jeffrey D. Sperling, MD, and Dana R. Gossett, MD, wrote in an accompanying editorial. They noted that, despite a lack of blinding and other limitations, as well as the fact there is no one risk-free solution regarding labor management or route of delivery, the results of this study have improved the understanding “of the risks and benefits of different strategies of labor management.”
The study, they added, “presents the only contemporary level 1 evidence available on the topic of whether the timing of pushing in the second stage of labor can have an effect on outcomes.”
The data “contribute to a growing body of literature on how to optimally manage labor in an evidenced-based fashion and improve perinatal outcomes without compromising maternal satisfaction,” they wrote. They recommended that future research should include exploring the effects and outcomes of delaying pushing among multiparous women, women with a previous cesarean delivery, and women who have not received epidural analgesia.
Dr. Sperling and Dr. Gossett are with the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco. Dr. Gossett reported serving as a consultant to Bayer Pharmaceuticals. No other disclosures were reported (JAMA. 2018;320[14]:1439-40).
Of the almost 4 million births in the United States each year, 68% are vaginal deliveries, which illustrates how important it is to have additional evidence to guide the management of labor, Jeffrey D. Sperling, MD, and Dana R. Gossett, MD, wrote in an accompanying editorial. They noted that, despite a lack of blinding and other limitations, as well as the fact there is no one risk-free solution regarding labor management or route of delivery, the results of this study have improved the understanding “of the risks and benefits of different strategies of labor management.”
The study, they added, “presents the only contemporary level 1 evidence available on the topic of whether the timing of pushing in the second stage of labor can have an effect on outcomes.”
The data “contribute to a growing body of literature on how to optimally manage labor in an evidenced-based fashion and improve perinatal outcomes without compromising maternal satisfaction,” they wrote. They recommended that future research should include exploring the effects and outcomes of delaying pushing among multiparous women, women with a previous cesarean delivery, and women who have not received epidural analgesia.
Dr. Sperling and Dr. Gossett are with the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco. Dr. Gossett reported serving as a consultant to Bayer Pharmaceuticals. No other disclosures were reported (JAMA. 2018;320[14]:1439-40).
Of the almost 4 million births in the United States each year, 68% are vaginal deliveries, which illustrates how important it is to have additional evidence to guide the management of labor, Jeffrey D. Sperling, MD, and Dana R. Gossett, MD, wrote in an accompanying editorial. They noted that, despite a lack of blinding and other limitations, as well as the fact there is no one risk-free solution regarding labor management or route of delivery, the results of this study have improved the understanding “of the risks and benefits of different strategies of labor management.”
The study, they added, “presents the only contemporary level 1 evidence available on the topic of whether the timing of pushing in the second stage of labor can have an effect on outcomes.”
The data “contribute to a growing body of literature on how to optimally manage labor in an evidenced-based fashion and improve perinatal outcomes without compromising maternal satisfaction,” they wrote. They recommended that future research should include exploring the effects and outcomes of delaying pushing among multiparous women, women with a previous cesarean delivery, and women who have not received epidural analgesia.
Dr. Sperling and Dr. Gossett are with the department of obstetrics, gynecology, and reproductive sciences at the University of California, San Francisco. Dr. Gossett reported serving as a consultant to Bayer Pharmaceuticals. No other disclosures were reported (JAMA. 2018;320[14]:1439-40).
according to the results of a randomized trial.
“The optimal technique for managing maternal pushing during the second stage of labor is unknown,” wrote Alison G. Cahill, MD, of Washington University, St. Louis, and her coauthors, who published their study in JAMA on Oct. 9. “The two most common approaches to the second stage of labor management are to either initiate pushing with uterine contractions once complete cervical dilation occurs (immediate pushing) or to allow for spontaneous descent (delayed pushing),” they noted. “Both approaches are commonly used, and neither is considered the gold standard.”
They addressed this question in the multicenter trial of nulliparous women (mean age, 26.5 years) who, during May 2014 to December 2017, were at or past 37 weeks’ gestation and had received neuraxial analgesia. The primary outcome was the rate of spontaneous vaginal delivery; secondary outcomes included maternal and neonatal morbidity outcomes. When they reached complete cervical dilation, women were randomized to immediate pushing or delayed pushing, in which they were instructed to wait 60 minutes.
The study was terminated after the data and safety monitoring board conducted a planned interim analysis; the analysis found futility in the delayed pushing group and raised concerns about increased morbidity in that group.
Among the 1,031 women in the immediate pushing group, the rate of spontaneous vaginal delivery was 85.9%; the rate was 86.5% among the 1,041 women in the delayed pushing group (P = .67).
The mean duration of the second stage of labor was significantly shorter in the immediate pushing group (102.4 minutes), compared with that seen in the delayed pushing group (134.2 minutes; P less than .001). The mean duration of active pushing was significantly longer in the immediate pushing group (83.7 minutes), compared with that seen in the delayed pushing group (74.5 minutes; P less than .001).
In terms of secondary outcomes, rates of postpartum hemorrhage were lower in the immediate pushing group (2.3%), compared with the rate among those in the delayed pushing group (4%; P = .03). During the second stage of labor, chorioamnionitis was significantly more common among women in the delayed pushing group (9.1%), compared with rate among women in the immediate pushing group (6.7%; P = .005). There was no significant difference between the two groups in the rates of a composite neonatal morbidity outcome (which included birth injury, respiratory distress, and neonatal acidemia), which was 7.3% in the immediate pushing group and 9.9% in the delayed pushing group. There were no neonatal deaths.
Among prespecified exploratory outcomes, the rates of neonatal acidemia and suspected neonatal sepsis were significantly higher in the delayed pushing group, whereas the rate of third-degree perineal lacerations was significantly higher in the immediate pushing group.
“The finding of no effect on spontaneous vaginal delivery for pushing timing during the second stage of labor and the evidence suggesting increased maternal and neonatal complications in the delayed pushing group support the view that women immediately pushing after complete cervical dilation may be preferred because women without neuraxial analgesia reflexively push immediately,” the authors pointed out. Their results, they concluded, “may help inform decisions about the preferred timing of second stage pushing efforts when considered with other maternal and neonatal outcomes.”
They noted that the trial had several limitations, including the unblinded design, which raised the possibility of bias that may have influenced the management of patients or diagnoses.
The authors had no disclosures. The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and by funding from the department of obstetrics and gynecology at Washington University, St. Louis.
SOURCE: Cahill AG et al. JAMA. 2018;320(14):1444-54.
according to the results of a randomized trial.
“The optimal technique for managing maternal pushing during the second stage of labor is unknown,” wrote Alison G. Cahill, MD, of Washington University, St. Louis, and her coauthors, who published their study in JAMA on Oct. 9. “The two most common approaches to the second stage of labor management are to either initiate pushing with uterine contractions once complete cervical dilation occurs (immediate pushing) or to allow for spontaneous descent (delayed pushing),” they noted. “Both approaches are commonly used, and neither is considered the gold standard.”
They addressed this question in the multicenter trial of nulliparous women (mean age, 26.5 years) who, during May 2014 to December 2017, were at or past 37 weeks’ gestation and had received neuraxial analgesia. The primary outcome was the rate of spontaneous vaginal delivery; secondary outcomes included maternal and neonatal morbidity outcomes. When they reached complete cervical dilation, women were randomized to immediate pushing or delayed pushing, in which they were instructed to wait 60 minutes.
The study was terminated after the data and safety monitoring board conducted a planned interim analysis; the analysis found futility in the delayed pushing group and raised concerns about increased morbidity in that group.
Among the 1,031 women in the immediate pushing group, the rate of spontaneous vaginal delivery was 85.9%; the rate was 86.5% among the 1,041 women in the delayed pushing group (P = .67).
The mean duration of the second stage of labor was significantly shorter in the immediate pushing group (102.4 minutes), compared with that seen in the delayed pushing group (134.2 minutes; P less than .001). The mean duration of active pushing was significantly longer in the immediate pushing group (83.7 minutes), compared with that seen in the delayed pushing group (74.5 minutes; P less than .001).
In terms of secondary outcomes, rates of postpartum hemorrhage were lower in the immediate pushing group (2.3%), compared with the rate among those in the delayed pushing group (4%; P = .03). During the second stage of labor, chorioamnionitis was significantly more common among women in the delayed pushing group (9.1%), compared with rate among women in the immediate pushing group (6.7%; P = .005). There was no significant difference between the two groups in the rates of a composite neonatal morbidity outcome (which included birth injury, respiratory distress, and neonatal acidemia), which was 7.3% in the immediate pushing group and 9.9% in the delayed pushing group. There were no neonatal deaths.
Among prespecified exploratory outcomes, the rates of neonatal acidemia and suspected neonatal sepsis were significantly higher in the delayed pushing group, whereas the rate of third-degree perineal lacerations was significantly higher in the immediate pushing group.
“The finding of no effect on spontaneous vaginal delivery for pushing timing during the second stage of labor and the evidence suggesting increased maternal and neonatal complications in the delayed pushing group support the view that women immediately pushing after complete cervical dilation may be preferred because women without neuraxial analgesia reflexively push immediately,” the authors pointed out. Their results, they concluded, “may help inform decisions about the preferred timing of second stage pushing efforts when considered with other maternal and neonatal outcomes.”
They noted that the trial had several limitations, including the unblinded design, which raised the possibility of bias that may have influenced the management of patients or diagnoses.
The authors had no disclosures. The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and by funding from the department of obstetrics and gynecology at Washington University, St. Louis.
SOURCE: Cahill AG et al. JAMA. 2018;320(14):1444-54.
FROM JAMA
Key clinical point: Delayed pushing during the second stage of labor may not reduce the risk of cesarean delivery and may lengthen the duration of labor.
Major finding: The rate of spontaneous vaginal delivery did not differ between groups (85.9% in the immediate pushing group versus 86.5% in the delayed pushing group).
Study details: A multicenter trial involving 2,414 nulliparous women receiving neuraxial analgesia, who were randomized to immediate pushing or delayed pushing during the second stage of labor.
Disclosures: The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and by funding from the department of obstetrics and gynecology at Washington University, St. Louis.
Source: Cahill AG et al. JAMA. 2018;320(14):1444-54.
Most dermatologic drugs safe for breastfeeding mothers
CHICAGO – A common reason why a women stops breastfeeding is the use of medication her doctor has claimed is unsafe during lactation. But most drugs have little or no effect on an infant’s well-being or milk supply, explained Jenny Eileen Murase, MD, of Palo Alto (Calif.) Foundation Medical Group.
“The bottom line I want you to take away from this [session] is that ,” Dr. Murase told attendees at the American Academy of Dermatology summer meeting. “I really want everyone in this room to understand that most of the time, you should not be recommending that a woman is pumping and dumping her milk or stopping breastfeeding because she’s on an agent.”
Dr. Murase, also affiliated with the University of California, San Francisco, provided an overview of drug safety during lactation for major categories of medications that dermatologists prescribe. She recommended that physicians get a copy of Medications and Mother’s Milk by Thomas Hale, PhD, which she considers the best reference for looking up specific drugs. It categorizes drugs as L1 (safest) to L5 (contraindicated), and L2 as “safer,” L3 as “moderately safe,” and L4 as “possibly hazardous.”
Steroids
Contrary to what many believe, prednisone is not contraindicated in breastfeeding, Dr. Murase said. Instead of advising patients to “pump and dump their milk,” she said, “the only recommendation you need to make is that they wait 4 hours after taking the medicine to breastfeed.” For example, a mother can take prednisone before bed and then wake 4 hours later to nurse. Higher doses, such as more than 40 mg daily over long periods, may have the potential to affect growth and development, but more typical doses don’t pose the same risk.
Topical steroids (except for those that are class 1) also are safe to apply directly to the nipple in breastfeeding women, she noted.
Biologics and immunosuppressants
One of the few medications that are contraindicated are topical pimecrolimus and tacrolimus if applied directly to the nipple, since “oral consumption in the infant could be significant,” Dr. Murase said.
Biologics, on the other hand, are not a concern during lactation. “They have low oral bioavailability because of their large molecular size,” and are broken down in the stomach “in a proteolytic environment,” Dr. Murase explained. The CRADLE study, for example, examined the concentration of certolizumab (Cimzia) in mothers’ mature breast milk and found the highest concentration to be just 0.077 mcg/mL, resulting in an average daily infant dose of less than 0.01 mg/kg per day.
Antihistamines and cosmetic topicals
The major antihistamines – brompheniramine, chlorpheniramine, diphenhydramine, hydroxyzine, cetirizine, fexofenadine and loratadine – are likewise safe as L1-L3 drugs. It is preferable to prescribe nonsedating antihistamines, opting for loratadine as the first-line choice. But dermatologists should be reassured that no data support concerns about milk supply reduction from antihistamines, Dr. Murase said.
It’s best to avoid cosmetic topical products, but hydroquinone (L3), topical minoxidil (L2), and botulinum toxin A (L3) do not pose significant risk to the infant. Neither do the anesthetics lidocaine (L2) and epinephrine (L1) for breastfeeding women who need surgery.
Antibiotics
The vast majority of antibiotics are safe for women to use while breastfeeding, but a few notable exceptions exist, including erythromycin.
“People associate erythromycin as safe in lactation because it’s safe in pregnancy, but that’s not the case,” Dr. Murase pointed out. Erythromycin has been linked to pyloric stenosis in newborns and therefore should be avoided in the early months of breastfeeding. In older infants, however, erythromycin becomes an L1 medication.”
Tetracyclines fall into a borderline category. “Tetracyclines would be fine for a complicated infection,” but should not be used for more than 3 weeks, at which point they are regarded as L4, Dr. Murase said. “So long-term use of the tetracyclines should be avoided.”
Aside from these, topical antibiotics are considered safe. Women taking other oral antibiotics should be monitored for gastrointestinal symptoms or allergic responses.
Antifungals
As for antifungals, topicals are safe, and nystatin and clotrimazole are the best first-line options (both L1). Oral antifungals are similarly fine, with griseofulvin, fluconazole, ketoconazole, itraconazole, and terbinafine all classified as L2 and amphotericin B as L3.
If antifungals or antibiotics are being prescribed for a breast fungal infection or for mastitis, Dr. Murase underscored the importance of not stopping breastfeeding.
“The most important thing is that they continue to actually breastfeed on the affected breast that has the staph infection,” she said. She then reiterated that physicians should “reassure new mothers that the majority of oral and topical medications are safe.”
Dr. Murase disclosed serving on the advisory boards of Dermira, UCB, and Genzyme/Sanofi, and she has consulted for Ferndale and UpToDate.
CHICAGO – A common reason why a women stops breastfeeding is the use of medication her doctor has claimed is unsafe during lactation. But most drugs have little or no effect on an infant’s well-being or milk supply, explained Jenny Eileen Murase, MD, of Palo Alto (Calif.) Foundation Medical Group.
“The bottom line I want you to take away from this [session] is that ,” Dr. Murase told attendees at the American Academy of Dermatology summer meeting. “I really want everyone in this room to understand that most of the time, you should not be recommending that a woman is pumping and dumping her milk or stopping breastfeeding because she’s on an agent.”
Dr. Murase, also affiliated with the University of California, San Francisco, provided an overview of drug safety during lactation for major categories of medications that dermatologists prescribe. She recommended that physicians get a copy of Medications and Mother’s Milk by Thomas Hale, PhD, which she considers the best reference for looking up specific drugs. It categorizes drugs as L1 (safest) to L5 (contraindicated), and L2 as “safer,” L3 as “moderately safe,” and L4 as “possibly hazardous.”
Steroids
Contrary to what many believe, prednisone is not contraindicated in breastfeeding, Dr. Murase said. Instead of advising patients to “pump and dump their milk,” she said, “the only recommendation you need to make is that they wait 4 hours after taking the medicine to breastfeed.” For example, a mother can take prednisone before bed and then wake 4 hours later to nurse. Higher doses, such as more than 40 mg daily over long periods, may have the potential to affect growth and development, but more typical doses don’t pose the same risk.
Topical steroids (except for those that are class 1) also are safe to apply directly to the nipple in breastfeeding women, she noted.
Biologics and immunosuppressants
One of the few medications that are contraindicated are topical pimecrolimus and tacrolimus if applied directly to the nipple, since “oral consumption in the infant could be significant,” Dr. Murase said.
Biologics, on the other hand, are not a concern during lactation. “They have low oral bioavailability because of their large molecular size,” and are broken down in the stomach “in a proteolytic environment,” Dr. Murase explained. The CRADLE study, for example, examined the concentration of certolizumab (Cimzia) in mothers’ mature breast milk and found the highest concentration to be just 0.077 mcg/mL, resulting in an average daily infant dose of less than 0.01 mg/kg per day.
Antihistamines and cosmetic topicals
The major antihistamines – brompheniramine, chlorpheniramine, diphenhydramine, hydroxyzine, cetirizine, fexofenadine and loratadine – are likewise safe as L1-L3 drugs. It is preferable to prescribe nonsedating antihistamines, opting for loratadine as the first-line choice. But dermatologists should be reassured that no data support concerns about milk supply reduction from antihistamines, Dr. Murase said.
It’s best to avoid cosmetic topical products, but hydroquinone (L3), topical minoxidil (L2), and botulinum toxin A (L3) do not pose significant risk to the infant. Neither do the anesthetics lidocaine (L2) and epinephrine (L1) for breastfeeding women who need surgery.
Antibiotics
The vast majority of antibiotics are safe for women to use while breastfeeding, but a few notable exceptions exist, including erythromycin.
“People associate erythromycin as safe in lactation because it’s safe in pregnancy, but that’s not the case,” Dr. Murase pointed out. Erythromycin has been linked to pyloric stenosis in newborns and therefore should be avoided in the early months of breastfeeding. In older infants, however, erythromycin becomes an L1 medication.”
Tetracyclines fall into a borderline category. “Tetracyclines would be fine for a complicated infection,” but should not be used for more than 3 weeks, at which point they are regarded as L4, Dr. Murase said. “So long-term use of the tetracyclines should be avoided.”
Aside from these, topical antibiotics are considered safe. Women taking other oral antibiotics should be monitored for gastrointestinal symptoms or allergic responses.
Antifungals
As for antifungals, topicals are safe, and nystatin and clotrimazole are the best first-line options (both L1). Oral antifungals are similarly fine, with griseofulvin, fluconazole, ketoconazole, itraconazole, and terbinafine all classified as L2 and amphotericin B as L3.
If antifungals or antibiotics are being prescribed for a breast fungal infection or for mastitis, Dr. Murase underscored the importance of not stopping breastfeeding.
“The most important thing is that they continue to actually breastfeed on the affected breast that has the staph infection,” she said. She then reiterated that physicians should “reassure new mothers that the majority of oral and topical medications are safe.”
Dr. Murase disclosed serving on the advisory boards of Dermira, UCB, and Genzyme/Sanofi, and she has consulted for Ferndale and UpToDate.
CHICAGO – A common reason why a women stops breastfeeding is the use of medication her doctor has claimed is unsafe during lactation. But most drugs have little or no effect on an infant’s well-being or milk supply, explained Jenny Eileen Murase, MD, of Palo Alto (Calif.) Foundation Medical Group.
“The bottom line I want you to take away from this [session] is that ,” Dr. Murase told attendees at the American Academy of Dermatology summer meeting. “I really want everyone in this room to understand that most of the time, you should not be recommending that a woman is pumping and dumping her milk or stopping breastfeeding because she’s on an agent.”
Dr. Murase, also affiliated with the University of California, San Francisco, provided an overview of drug safety during lactation for major categories of medications that dermatologists prescribe. She recommended that physicians get a copy of Medications and Mother’s Milk by Thomas Hale, PhD, which she considers the best reference for looking up specific drugs. It categorizes drugs as L1 (safest) to L5 (contraindicated), and L2 as “safer,” L3 as “moderately safe,” and L4 as “possibly hazardous.”
Steroids
Contrary to what many believe, prednisone is not contraindicated in breastfeeding, Dr. Murase said. Instead of advising patients to “pump and dump their milk,” she said, “the only recommendation you need to make is that they wait 4 hours after taking the medicine to breastfeed.” For example, a mother can take prednisone before bed and then wake 4 hours later to nurse. Higher doses, such as more than 40 mg daily over long periods, may have the potential to affect growth and development, but more typical doses don’t pose the same risk.
Topical steroids (except for those that are class 1) also are safe to apply directly to the nipple in breastfeeding women, she noted.
Biologics and immunosuppressants
One of the few medications that are contraindicated are topical pimecrolimus and tacrolimus if applied directly to the nipple, since “oral consumption in the infant could be significant,” Dr. Murase said.
Biologics, on the other hand, are not a concern during lactation. “They have low oral bioavailability because of their large molecular size,” and are broken down in the stomach “in a proteolytic environment,” Dr. Murase explained. The CRADLE study, for example, examined the concentration of certolizumab (Cimzia) in mothers’ mature breast milk and found the highest concentration to be just 0.077 mcg/mL, resulting in an average daily infant dose of less than 0.01 mg/kg per day.
Antihistamines and cosmetic topicals
The major antihistamines – brompheniramine, chlorpheniramine, diphenhydramine, hydroxyzine, cetirizine, fexofenadine and loratadine – are likewise safe as L1-L3 drugs. It is preferable to prescribe nonsedating antihistamines, opting for loratadine as the first-line choice. But dermatologists should be reassured that no data support concerns about milk supply reduction from antihistamines, Dr. Murase said.
It’s best to avoid cosmetic topical products, but hydroquinone (L3), topical minoxidil (L2), and botulinum toxin A (L3) do not pose significant risk to the infant. Neither do the anesthetics lidocaine (L2) and epinephrine (L1) for breastfeeding women who need surgery.
Antibiotics
The vast majority of antibiotics are safe for women to use while breastfeeding, but a few notable exceptions exist, including erythromycin.
“People associate erythromycin as safe in lactation because it’s safe in pregnancy, but that’s not the case,” Dr. Murase pointed out. Erythromycin has been linked to pyloric stenosis in newborns and therefore should be avoided in the early months of breastfeeding. In older infants, however, erythromycin becomes an L1 medication.”
Tetracyclines fall into a borderline category. “Tetracyclines would be fine for a complicated infection,” but should not be used for more than 3 weeks, at which point they are regarded as L4, Dr. Murase said. “So long-term use of the tetracyclines should be avoided.”
Aside from these, topical antibiotics are considered safe. Women taking other oral antibiotics should be monitored for gastrointestinal symptoms or allergic responses.
Antifungals
As for antifungals, topicals are safe, and nystatin and clotrimazole are the best first-line options (both L1). Oral antifungals are similarly fine, with griseofulvin, fluconazole, ketoconazole, itraconazole, and terbinafine all classified as L2 and amphotericin B as L3.
If antifungals or antibiotics are being prescribed for a breast fungal infection or for mastitis, Dr. Murase underscored the importance of not stopping breastfeeding.
“The most important thing is that they continue to actually breastfeed on the affected breast that has the staph infection,” she said. She then reiterated that physicians should “reassure new mothers that the majority of oral and topical medications are safe.”
Dr. Murase disclosed serving on the advisory boards of Dermira, UCB, and Genzyme/Sanofi, and she has consulted for Ferndale and UpToDate.
EXPERT ANALYSIS FROM SUMMER AAD 2018
FDA expands approval of 9-valent HPV vaccine
The , men and women aged 27-45 years, the Food and Drug Administration announced on Oct. 5.
The vaccine (Gardasil 9) was previously approved for those aged 9-26 years.
The approval “represents an important opportunity to help prevent HPV-related diseases and cancers in a broader age range,” Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research, said in the FDA statement announcing the approval.
“The Centers for Disease Control and Prevention has stated that HPV vaccination prior to becoming infected with the HPV types covered by the vaccine has the potential to prevent more than 90 percent of these cancers, or 31,200 cases every year, from ever developing,” he added.
Gardasil 9, approved in 2014, covers the four HPV types included in the original Gardasil vaccine approved in 2006, plus five additional HPV types.
The approval is based on the results of a study and follow-up of about 3,200 women aged 27-45 years, followed for an average of 3.5 years, which found that the vaccine was 88% percent effective “in the prevention of a combined endpoint of persistent infection, genital warts, vulvar and vaginal precancerous lesions, cervical precancerous lesions, and cervical cancer related to HPV types covered by the vaccine,” according to the FDA. The vaccine’s effectiveness in men in this age group is “inferred” from these results and from data on Gardasil in men aged 16-26 years, as well as “immunogenicity data from a clinical trial in which 150 men, 27 through 45 years of age, received a 3-dose regimen of Gardasil over 6 months,” the FDA statement noted.
Based on safety data in about 13,000 men and women, injection-site pain, swelling, redness, and headaches are the most common adverse reactions associated with Gardasil 9, the statement said. Gardasil 9 is manufactured by Merck.
The , men and women aged 27-45 years, the Food and Drug Administration announced on Oct. 5.
The vaccine (Gardasil 9) was previously approved for those aged 9-26 years.
The approval “represents an important opportunity to help prevent HPV-related diseases and cancers in a broader age range,” Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research, said in the FDA statement announcing the approval.
“The Centers for Disease Control and Prevention has stated that HPV vaccination prior to becoming infected with the HPV types covered by the vaccine has the potential to prevent more than 90 percent of these cancers, or 31,200 cases every year, from ever developing,” he added.
Gardasil 9, approved in 2014, covers the four HPV types included in the original Gardasil vaccine approved in 2006, plus five additional HPV types.
The approval is based on the results of a study and follow-up of about 3,200 women aged 27-45 years, followed for an average of 3.5 years, which found that the vaccine was 88% percent effective “in the prevention of a combined endpoint of persistent infection, genital warts, vulvar and vaginal precancerous lesions, cervical precancerous lesions, and cervical cancer related to HPV types covered by the vaccine,” according to the FDA. The vaccine’s effectiveness in men in this age group is “inferred” from these results and from data on Gardasil in men aged 16-26 years, as well as “immunogenicity data from a clinical trial in which 150 men, 27 through 45 years of age, received a 3-dose regimen of Gardasil over 6 months,” the FDA statement noted.
Based on safety data in about 13,000 men and women, injection-site pain, swelling, redness, and headaches are the most common adverse reactions associated with Gardasil 9, the statement said. Gardasil 9 is manufactured by Merck.
The , men and women aged 27-45 years, the Food and Drug Administration announced on Oct. 5.
The vaccine (Gardasil 9) was previously approved for those aged 9-26 years.
The approval “represents an important opportunity to help prevent HPV-related diseases and cancers in a broader age range,” Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research, said in the FDA statement announcing the approval.
“The Centers for Disease Control and Prevention has stated that HPV vaccination prior to becoming infected with the HPV types covered by the vaccine has the potential to prevent more than 90 percent of these cancers, or 31,200 cases every year, from ever developing,” he added.
Gardasil 9, approved in 2014, covers the four HPV types included in the original Gardasil vaccine approved in 2006, plus five additional HPV types.
The approval is based on the results of a study and follow-up of about 3,200 women aged 27-45 years, followed for an average of 3.5 years, which found that the vaccine was 88% percent effective “in the prevention of a combined endpoint of persistent infection, genital warts, vulvar and vaginal precancerous lesions, cervical precancerous lesions, and cervical cancer related to HPV types covered by the vaccine,” according to the FDA. The vaccine’s effectiveness in men in this age group is “inferred” from these results and from data on Gardasil in men aged 16-26 years, as well as “immunogenicity data from a clinical trial in which 150 men, 27 through 45 years of age, received a 3-dose regimen of Gardasil over 6 months,” the FDA statement noted.
Based on safety data in about 13,000 men and women, injection-site pain, swelling, redness, and headaches are the most common adverse reactions associated with Gardasil 9, the statement said. Gardasil 9 is manufactured by Merck.
For dyspareunia, intravaginal prasterone may work best soon after menopause
Neither age nor previous hormone therapy had statistically significant associations with the effect of intravaginal prasterone on dyspareunia severity, according to a new subgroup analysis of clinical trial data. In a trend that did not reach statistical significance, though, said David F. Archer, MD.
“This was an unexpected finding,” he said in an interview.
In a subgroup analysis of data from two clinical trials of intravaginal prasterone (Intrarosa), Dr. Archer and his colleagues sought to investigate whether age, time since menopause, or any previous use of hormone replacement therapy influenced prasterone’s efficacy in treating dyspareunia.
Dr. Archer and his collaborators pooled data from two prospective, randomized, double-blind, placebo-controlled trials (NCT02013544 and NCT01256684) of intravaginal prasterone dosed at 0.50%, 6.5 mg once daily for 12 weeks; he presented the subgroup analyses at the annual meeting of the North American Menopause Society in San Diego.
For each subgroup, Dr. Archer, a professor of obstetrics and gynecology at Eastern Virginia Medical School, Norfolk, and his coinvestigators compared the mean differences in dyspareunia severity score of women who received prasterone and those who received placebo.
All subgroup analyses used the endpoint of improvement in moderate to severe dyspareunia or whether dyspareunia was the most bothersome symptoms for the women participating in the study. The investigators began by looking at the subgroup of 460 women who were 56 years and older at baseline and compared them with the 180 younger participants.
The 283 older participants who received prasterone saw a decrease of 0.36 points in a dyspareunia severity score versus a 0.44 point decrease for the 123 women aged 55 and younger who received prasterone, a nonsignificant difference between subgroups. The decrease compared with placebo-takers was significant in both cases, however (P = .0003 and P =.0031, respectively).
Looking at time since menopause, Dr. Archer and his collaborators divided participants into 33 individuals who were 1 or 2 years post menopause, 86 women who were 3-5 years post menopause, and 521 women who had experienced menopause at least 6 years before study baseline.
In this analysis, 22 of the earliest postmenopause women received prasterone, seeing a 1.59 point drop in dyspareunia severity. For the 59 women in the prasterone study arms who were 3-5 year past menopause, the decrease from baseline was 0.59 points. Finally, among the 325 women who received prasterone and experienced menopause 6 or more years ago, the decrease was 0.27 points.
Although there was a numeric difference in the change in dyspareunia score severity among these groups, the differences were not statistically significant, said Dr. Archer. Again, though, those who took prasterone had a significant reduction in dyspareunia severity scores when compared with those taking placebo (P less than .0001, P = .0136, and P = .0024, respectively).
In the prasterone study arms, 184 had previously used hormone therapy, and 222 had not. After 12 weeks of intravaginal prasterone, there was no statistically significant difference between the two subgroups, with a decreases in dyspareunia severity scores of 0.45 and 0.32, respectively. The decreases in severity scores when compared with those among women who took placebo were again statistically significant for both subgroups, however (P = .0002 and P = .0057, respectively).
Prasterone is a steroid that is also known as dehydroepiandrosterone (DHEA) and is an endogenous hormone that is a precursor for estrogens and androgens. Prasterone’s mechanism of action to reduce vulvar and vaginal atrophy is not completely understood, according to the Food and Drug Administration.
“The nonstatistically significant smaller effect on dyspareunia observed when treatment is initiated after a longer period after menopause suggests that a longer treatment period could be needed to achieve optimal benefit and that treatment of dyspareunia should be initiated as early as possible after menopause,” said Dr. Archer.
Dr. Archer reported grant support from and consulting relationships with several pharmaceutical companies, including Endoceutics, the producer of Intrarosa intravaginal prasterone.
Neither age nor previous hormone therapy had statistically significant associations with the effect of intravaginal prasterone on dyspareunia severity, according to a new subgroup analysis of clinical trial data. In a trend that did not reach statistical significance, though, said David F. Archer, MD.
“This was an unexpected finding,” he said in an interview.
In a subgroup analysis of data from two clinical trials of intravaginal prasterone (Intrarosa), Dr. Archer and his colleagues sought to investigate whether age, time since menopause, or any previous use of hormone replacement therapy influenced prasterone’s efficacy in treating dyspareunia.
Dr. Archer and his collaborators pooled data from two prospective, randomized, double-blind, placebo-controlled trials (NCT02013544 and NCT01256684) of intravaginal prasterone dosed at 0.50%, 6.5 mg once daily for 12 weeks; he presented the subgroup analyses at the annual meeting of the North American Menopause Society in San Diego.
For each subgroup, Dr. Archer, a professor of obstetrics and gynecology at Eastern Virginia Medical School, Norfolk, and his coinvestigators compared the mean differences in dyspareunia severity score of women who received prasterone and those who received placebo.
All subgroup analyses used the endpoint of improvement in moderate to severe dyspareunia or whether dyspareunia was the most bothersome symptoms for the women participating in the study. The investigators began by looking at the subgroup of 460 women who were 56 years and older at baseline and compared them with the 180 younger participants.
The 283 older participants who received prasterone saw a decrease of 0.36 points in a dyspareunia severity score versus a 0.44 point decrease for the 123 women aged 55 and younger who received prasterone, a nonsignificant difference between subgroups. The decrease compared with placebo-takers was significant in both cases, however (P = .0003 and P =.0031, respectively).
Looking at time since menopause, Dr. Archer and his collaborators divided participants into 33 individuals who were 1 or 2 years post menopause, 86 women who were 3-5 years post menopause, and 521 women who had experienced menopause at least 6 years before study baseline.
In this analysis, 22 of the earliest postmenopause women received prasterone, seeing a 1.59 point drop in dyspareunia severity. For the 59 women in the prasterone study arms who were 3-5 year past menopause, the decrease from baseline was 0.59 points. Finally, among the 325 women who received prasterone and experienced menopause 6 or more years ago, the decrease was 0.27 points.
Although there was a numeric difference in the change in dyspareunia score severity among these groups, the differences were not statistically significant, said Dr. Archer. Again, though, those who took prasterone had a significant reduction in dyspareunia severity scores when compared with those taking placebo (P less than .0001, P = .0136, and P = .0024, respectively).
In the prasterone study arms, 184 had previously used hormone therapy, and 222 had not. After 12 weeks of intravaginal prasterone, there was no statistically significant difference between the two subgroups, with a decreases in dyspareunia severity scores of 0.45 and 0.32, respectively. The decreases in severity scores when compared with those among women who took placebo were again statistically significant for both subgroups, however (P = .0002 and P = .0057, respectively).
Prasterone is a steroid that is also known as dehydroepiandrosterone (DHEA) and is an endogenous hormone that is a precursor for estrogens and androgens. Prasterone’s mechanism of action to reduce vulvar and vaginal atrophy is not completely understood, according to the Food and Drug Administration.
“The nonstatistically significant smaller effect on dyspareunia observed when treatment is initiated after a longer period after menopause suggests that a longer treatment period could be needed to achieve optimal benefit and that treatment of dyspareunia should be initiated as early as possible after menopause,” said Dr. Archer.
Dr. Archer reported grant support from and consulting relationships with several pharmaceutical companies, including Endoceutics, the producer of Intrarosa intravaginal prasterone.
Neither age nor previous hormone therapy had statistically significant associations with the effect of intravaginal prasterone on dyspareunia severity, according to a new subgroup analysis of clinical trial data. In a trend that did not reach statistical significance, though, said David F. Archer, MD.
“This was an unexpected finding,” he said in an interview.
In a subgroup analysis of data from two clinical trials of intravaginal prasterone (Intrarosa), Dr. Archer and his colleagues sought to investigate whether age, time since menopause, or any previous use of hormone replacement therapy influenced prasterone’s efficacy in treating dyspareunia.
Dr. Archer and his collaborators pooled data from two prospective, randomized, double-blind, placebo-controlled trials (NCT02013544 and NCT01256684) of intravaginal prasterone dosed at 0.50%, 6.5 mg once daily for 12 weeks; he presented the subgroup analyses at the annual meeting of the North American Menopause Society in San Diego.
For each subgroup, Dr. Archer, a professor of obstetrics and gynecology at Eastern Virginia Medical School, Norfolk, and his coinvestigators compared the mean differences in dyspareunia severity score of women who received prasterone and those who received placebo.
All subgroup analyses used the endpoint of improvement in moderate to severe dyspareunia or whether dyspareunia was the most bothersome symptoms for the women participating in the study. The investigators began by looking at the subgroup of 460 women who were 56 years and older at baseline and compared them with the 180 younger participants.
The 283 older participants who received prasterone saw a decrease of 0.36 points in a dyspareunia severity score versus a 0.44 point decrease for the 123 women aged 55 and younger who received prasterone, a nonsignificant difference between subgroups. The decrease compared with placebo-takers was significant in both cases, however (P = .0003 and P =.0031, respectively).
Looking at time since menopause, Dr. Archer and his collaborators divided participants into 33 individuals who were 1 or 2 years post menopause, 86 women who were 3-5 years post menopause, and 521 women who had experienced menopause at least 6 years before study baseline.
In this analysis, 22 of the earliest postmenopause women received prasterone, seeing a 1.59 point drop in dyspareunia severity. For the 59 women in the prasterone study arms who were 3-5 year past menopause, the decrease from baseline was 0.59 points. Finally, among the 325 women who received prasterone and experienced menopause 6 or more years ago, the decrease was 0.27 points.
Although there was a numeric difference in the change in dyspareunia score severity among these groups, the differences were not statistically significant, said Dr. Archer. Again, though, those who took prasterone had a significant reduction in dyspareunia severity scores when compared with those taking placebo (P less than .0001, P = .0136, and P = .0024, respectively).
In the prasterone study arms, 184 had previously used hormone therapy, and 222 had not. After 12 weeks of intravaginal prasterone, there was no statistically significant difference between the two subgroups, with a decreases in dyspareunia severity scores of 0.45 and 0.32, respectively. The decreases in severity scores when compared with those among women who took placebo were again statistically significant for both subgroups, however (P = .0002 and P = .0057, respectively).
Prasterone is a steroid that is also known as dehydroepiandrosterone (DHEA) and is an endogenous hormone that is a precursor for estrogens and androgens. Prasterone’s mechanism of action to reduce vulvar and vaginal atrophy is not completely understood, according to the Food and Drug Administration.
“The nonstatistically significant smaller effect on dyspareunia observed when treatment is initiated after a longer period after menopause suggests that a longer treatment period could be needed to achieve optimal benefit and that treatment of dyspareunia should be initiated as early as possible after menopause,” said Dr. Archer.
Dr. Archer reported grant support from and consulting relationships with several pharmaceutical companies, including Endoceutics, the producer of Intrarosa intravaginal prasterone.
FROM NAMS 2018
Key clinical point: Dyspareunia improvement was numerically, but not statistically, better soon after menopause.
Major finding: Dyspareunia scores dropped 1.59 points for those within 2 years of menopause, and 0.27 points for those 6 or more years post menopause.
Study details: Subgroup analysis of 640 postmenopausal women taking part in two clinical trials.
Disclosures: Dr. Archer reported receiving support from several pharmaceutical companies, including Endoceutics, the manufacturer of Intrarosa intravaginal prasterone.
Encourage influenza vaccination in pregnant women
They are at greater risk for more severe illness, and influenza can lead to adverse outcomes in infants. The good news is that recent studies have shown that flu vaccines are safe and effective in pregnant women.
The bad news is that many women are hesitant to be vaccinated out of concerns over safety, in a trend that reflects broader societal worries over vaccination, said Dr. Chu, of the University of Washington, Seattle. In a video interview at an annual scientific meeting on infectious diseases, Dr. Chu advised steps to ensure that pregnant women are aware of the safety and efficacy of flu vaccines, and the benefits to the infant who acquires immunity through the mother. It’s also a good idea to have vaccine on hand to be able to offer it immediately during an office visit.
They are at greater risk for more severe illness, and influenza can lead to adverse outcomes in infants. The good news is that recent studies have shown that flu vaccines are safe and effective in pregnant women.
The bad news is that many women are hesitant to be vaccinated out of concerns over safety, in a trend that reflects broader societal worries over vaccination, said Dr. Chu, of the University of Washington, Seattle. In a video interview at an annual scientific meeting on infectious diseases, Dr. Chu advised steps to ensure that pregnant women are aware of the safety and efficacy of flu vaccines, and the benefits to the infant who acquires immunity through the mother. It’s also a good idea to have vaccine on hand to be able to offer it immediately during an office visit.
They are at greater risk for more severe illness, and influenza can lead to adverse outcomes in infants. The good news is that recent studies have shown that flu vaccines are safe and effective in pregnant women.
The bad news is that many women are hesitant to be vaccinated out of concerns over safety, in a trend that reflects broader societal worries over vaccination, said Dr. Chu, of the University of Washington, Seattle. In a video interview at an annual scientific meeting on infectious diseases, Dr. Chu advised steps to ensure that pregnant women are aware of the safety and efficacy of flu vaccines, and the benefits to the infant who acquires immunity through the mother. It’s also a good idea to have vaccine on hand to be able to offer it immediately during an office visit.
REPORTING FROM ID WEEK 2018
Sexual assault and harassment linked to hypertension, depression, and anxiety
Sexual harassment and assault may have significant health impacts on women in midlife, including greater risk of hypertension, poor sleep, depression, and anxiety, research suggests.
In the Oct. 3 online edition of JAMA Internal Medicine, a study of 304 women aged 40-60 years showed that 19% reported a history of workplace sexual harassment, 22% reported a history of sexual assault, and 10% reported both. The report was presented simultaneously at the North American Menopause Society annual meeting in San Diego.
The researchers found that those with a history of sexual assault had an almost threefold higher odds of clinically elevated depressive symptoms (OR, 2.86, P = .003), and more than twofold greater odds of anxiety and poor sleep (OR, 2.26, P = .006 and OR, 2.15, P = .007 respectively).
Women who reported experiencing sexual harassment in the workplace – and who were not taking antihypertensive medication – were more than twice as likely to have stage 1 or 2 hypertension, compared with women who had not experienced sexual harassment (OR, 2.36, P = .03). They also had 89% higher odds of poor sleep consistent with clinical insomnia (P = .03).
These associations all persisted even after adjustment for demographic and biomedical factors such as age, ethnicity, body mass index, snoring, and the use of antihypertensive, antidepressant, and anti-anxiety medications.
“Given the high prevalence of sexual harassment and assault, addressing these prevalent and potent social exposures may be critical to promoting health and preventing disease in women,” wrote Rebecca C. Thurston, PhD, of the department of psychiatry at the University of Pittsburgh, and her coauthors.
They noted that the 1-in-5 rate of sexual harassment or assault seen in the study was actually lower than that seen in national samples, which may be have been because of the exclusion of women who smoked, had undergone hysterectomies, or were using common antidepressants or cardiovascular medications.
“Few characteristics distinguished between women who had been sexually harassed and those who had been sexually assaulted, with the exception that women who were sexually harassed were more highly educated yet more financially strained,” they wrote. “Notably, women who are younger or are in more precarious employment situations are more likely to be harassed, and financially stressed women can lack the financial security to leave abusive work situations.”
The study was supported by the National Institutes of Health, National Heart Lung and Blood Institute, and the University of Pittsburgh Clinical and Translational Science Institute. Dr. Thurston declared consultancies for MAS Innovations, Procter & Gamble, and Pfizer, but no other conflicts of interest were declared.
SOURCE: Thurston R et al. JAMA Intern Med. 2018, Oct 3. doi: 10.1001/jamainternmed.2018.4886.
Sexual harassment and assault may have significant health impacts on women in midlife, including greater risk of hypertension, poor sleep, depression, and anxiety, research suggests.
In the Oct. 3 online edition of JAMA Internal Medicine, a study of 304 women aged 40-60 years showed that 19% reported a history of workplace sexual harassment, 22% reported a history of sexual assault, and 10% reported both. The report was presented simultaneously at the North American Menopause Society annual meeting in San Diego.
The researchers found that those with a history of sexual assault had an almost threefold higher odds of clinically elevated depressive symptoms (OR, 2.86, P = .003), and more than twofold greater odds of anxiety and poor sleep (OR, 2.26, P = .006 and OR, 2.15, P = .007 respectively).
Women who reported experiencing sexual harassment in the workplace – and who were not taking antihypertensive medication – were more than twice as likely to have stage 1 or 2 hypertension, compared with women who had not experienced sexual harassment (OR, 2.36, P = .03). They also had 89% higher odds of poor sleep consistent with clinical insomnia (P = .03).
These associations all persisted even after adjustment for demographic and biomedical factors such as age, ethnicity, body mass index, snoring, and the use of antihypertensive, antidepressant, and anti-anxiety medications.
“Given the high prevalence of sexual harassment and assault, addressing these prevalent and potent social exposures may be critical to promoting health and preventing disease in women,” wrote Rebecca C. Thurston, PhD, of the department of psychiatry at the University of Pittsburgh, and her coauthors.
They noted that the 1-in-5 rate of sexual harassment or assault seen in the study was actually lower than that seen in national samples, which may be have been because of the exclusion of women who smoked, had undergone hysterectomies, or were using common antidepressants or cardiovascular medications.
“Few characteristics distinguished between women who had been sexually harassed and those who had been sexually assaulted, with the exception that women who were sexually harassed were more highly educated yet more financially strained,” they wrote. “Notably, women who are younger or are in more precarious employment situations are more likely to be harassed, and financially stressed women can lack the financial security to leave abusive work situations.”
The study was supported by the National Institutes of Health, National Heart Lung and Blood Institute, and the University of Pittsburgh Clinical and Translational Science Institute. Dr. Thurston declared consultancies for MAS Innovations, Procter & Gamble, and Pfizer, but no other conflicts of interest were declared.
SOURCE: Thurston R et al. JAMA Intern Med. 2018, Oct 3. doi: 10.1001/jamainternmed.2018.4886.
Sexual harassment and assault may have significant health impacts on women in midlife, including greater risk of hypertension, poor sleep, depression, and anxiety, research suggests.
In the Oct. 3 online edition of JAMA Internal Medicine, a study of 304 women aged 40-60 years showed that 19% reported a history of workplace sexual harassment, 22% reported a history of sexual assault, and 10% reported both. The report was presented simultaneously at the North American Menopause Society annual meeting in San Diego.
The researchers found that those with a history of sexual assault had an almost threefold higher odds of clinically elevated depressive symptoms (OR, 2.86, P = .003), and more than twofold greater odds of anxiety and poor sleep (OR, 2.26, P = .006 and OR, 2.15, P = .007 respectively).
Women who reported experiencing sexual harassment in the workplace – and who were not taking antihypertensive medication – were more than twice as likely to have stage 1 or 2 hypertension, compared with women who had not experienced sexual harassment (OR, 2.36, P = .03). They also had 89% higher odds of poor sleep consistent with clinical insomnia (P = .03).
These associations all persisted even after adjustment for demographic and biomedical factors such as age, ethnicity, body mass index, snoring, and the use of antihypertensive, antidepressant, and anti-anxiety medications.
“Given the high prevalence of sexual harassment and assault, addressing these prevalent and potent social exposures may be critical to promoting health and preventing disease in women,” wrote Rebecca C. Thurston, PhD, of the department of psychiatry at the University of Pittsburgh, and her coauthors.
They noted that the 1-in-5 rate of sexual harassment or assault seen in the study was actually lower than that seen in national samples, which may be have been because of the exclusion of women who smoked, had undergone hysterectomies, or were using common antidepressants or cardiovascular medications.
“Few characteristics distinguished between women who had been sexually harassed and those who had been sexually assaulted, with the exception that women who were sexually harassed were more highly educated yet more financially strained,” they wrote. “Notably, women who are younger or are in more precarious employment situations are more likely to be harassed, and financially stressed women can lack the financial security to leave abusive work situations.”
The study was supported by the National Institutes of Health, National Heart Lung and Blood Institute, and the University of Pittsburgh Clinical and Translational Science Institute. Dr. Thurston declared consultancies for MAS Innovations, Procter & Gamble, and Pfizer, but no other conflicts of interest were declared.
SOURCE: Thurston R et al. JAMA Intern Med. 2018, Oct 3. doi: 10.1001/jamainternmed.2018.4886.
FROM JAMA INTERNAL MEDICINE
Key clinical point:
Major finding: Women who have experienced sexual assault showed nearly threefold higher odds of depressive symptoms.
Study details: Study of 304 women aged 40-60 years.
Disclosures: The study was supported by the National Institutes of Health, National Heart Lung and Blood Institute, and the University of Pittsburgh Clinical and Translational Science Institute. Dr. Thurston declared consultancies for MAS Innovations, Procter & Gamble, and Pfizer, but no other conflicts of interest were declared.
Source: Thurston R et al. JAMA Intern Med. 2018 Oct 3. doi: 10.1001/jamainternmed.2018.4886.
Anti-TNF agents preferred for severe psoriasis in pregnancy
CHICAGO – according to Kenneth B. Gordon, MD, professor and chair of dermatology at Medical College of Wisconsin in Milwaukee.
“We always had this concept that psoriasis gets better during pregnancy, that you might have 20% or 30% of patients who might have a little bit of a flare or maintain, but most keep on getting better,” Dr. Gordon told attendees at the American Academy of Dermatology summer meeting.
But the majority doesn’t mean everyone. He shared the case of one pregnant woman who came to him with severe psoriasis, covering the whole of her inner thigh, to underscore that severe cases do happen in pregnancy.
“These are real situations, and when you talk about maternal health, this woman is uncomfortable, she can’t sleep, and she’s having huge stressors that are not only going to impact her and her pregnancy but also that impact her child,” Dr. Gordon said.
Dr. Gordon clarified that he is not referring to patients with limited psoriasis or those who respond to topicals or phototherapy. But because methotrexate or acitretin are “hands-off during pregnancy,” he said, the only systemic therapy available for serious cases besides biologics is cyclosporine, which has its own risks. “We know that [cyclosporine] is associated with preterm labor and preterm birth and significant low birth weight, so even in the best scenario, when we have someone with persistent severe psoriasis in pregnancy, our best agent has a lot of downsides.”
Too few data exist on anti–interleukin (IL)-17 or anti-IL-23 therapies to draw conclusions about their use, he said, and but gastroenterology and rheumatology have a fair amount of evidence on anti–tumor necrosis factor (TNF) therapies during pregnancy because it’s usually too risky to stop treating conditions such as Crohn’s with these drugs. Still, Dr. Gordon cautioned, much of the data on biologics in pregnancy are conflicting.
The question of what medications to use, and in whom, centers on balancing risks to the fetus from the medication versus risks from the condition.
“There are impacts on the fetus of having severe psoriasis, and it varies with severity of disease,” Dr. Gordon said. For example, data suggest an increased likelihood of low birth weight in children born to mothers with severe psoriasis, and that risk may extend to preterm birth as well, although “we don’t know exactly the magnitude of that effect.”
Meanwhile, the consensus from the literature throughout dermatology, rheumatology, and gastroenterology is that anti-TNF agents do not cause birth defects or affect risk of preterm birth or low birth weight.
“The bigger question is what’s the impact on the immune system of the child,” Dr. Gordon said. Data from a small Scandinavian study suggested no increased risk of allergies, infections, or similar immunologic outcomes, but evidence remains limited.
Research has shown that infants’ exposure to anti-TNF medications persists for 3-6 months after delivery, and the American Academy of Pediatrics recommends delaying immunization in children exposed to anti-TNF agents in pregnancy. But actual evidence on immunization outcomes shows no reduced immunogenicity in such children.
“Clearly there is persistence of drug in the child, but in fact you have normal responses to immunization,” Dr. Gordon said. “The pediatricians’ argument is not based on data of what actually happens in immunization; it’s based on the fact that the drug is there.”
So what’s the bottom line?
The National Psoriasis Foundation recommends moisturizers and topical corticosteroids as first-line therapy in pregnant women with psoriasis, followed by phototherapy for second-line treatment.
But some patients will need systemic therapy during pregnancy, although it’s “best not to introduce more medications than needed in pregnancy,” Dr. Gordon said. For women with a significant flare-up or very persistent volatile disease, NPF first recommends cyclosporine, but Dr. Gordon disagrees and would go with anti-TNF agents before cyclosporine.
Data show that certolizumab is not actively transported across the placenta therefore reducing fetal exposure, so Dr. Gordon would specifically use certolizumab first, all other things being equal.
“But if the patient has been on another anti-TNF that’s been working, I don’t really have an issue with staying with it,” he added.
Existing evidence so far shows no impact in terms of genetic abnormalities, birth weight, premature birth, or even infant immunizations from anti-TNF agents. But beyond those, “there is simply not enough information on pregnancy with other forms of biologic therapy to draw conclusions.” Dr. Gordon said.
Dr. Gordon disclosed that he has received grant support and/or honoraria from Abbvie, Amgen, Almirall, and Boehringer Ingelheim.
CHICAGO – according to Kenneth B. Gordon, MD, professor and chair of dermatology at Medical College of Wisconsin in Milwaukee.
“We always had this concept that psoriasis gets better during pregnancy, that you might have 20% or 30% of patients who might have a little bit of a flare or maintain, but most keep on getting better,” Dr. Gordon told attendees at the American Academy of Dermatology summer meeting.
But the majority doesn’t mean everyone. He shared the case of one pregnant woman who came to him with severe psoriasis, covering the whole of her inner thigh, to underscore that severe cases do happen in pregnancy.
“These are real situations, and when you talk about maternal health, this woman is uncomfortable, she can’t sleep, and she’s having huge stressors that are not only going to impact her and her pregnancy but also that impact her child,” Dr. Gordon said.
Dr. Gordon clarified that he is not referring to patients with limited psoriasis or those who respond to topicals or phototherapy. But because methotrexate or acitretin are “hands-off during pregnancy,” he said, the only systemic therapy available for serious cases besides biologics is cyclosporine, which has its own risks. “We know that [cyclosporine] is associated with preterm labor and preterm birth and significant low birth weight, so even in the best scenario, when we have someone with persistent severe psoriasis in pregnancy, our best agent has a lot of downsides.”
Too few data exist on anti–interleukin (IL)-17 or anti-IL-23 therapies to draw conclusions about their use, he said, and but gastroenterology and rheumatology have a fair amount of evidence on anti–tumor necrosis factor (TNF) therapies during pregnancy because it’s usually too risky to stop treating conditions such as Crohn’s with these drugs. Still, Dr. Gordon cautioned, much of the data on biologics in pregnancy are conflicting.
The question of what medications to use, and in whom, centers on balancing risks to the fetus from the medication versus risks from the condition.
“There are impacts on the fetus of having severe psoriasis, and it varies with severity of disease,” Dr. Gordon said. For example, data suggest an increased likelihood of low birth weight in children born to mothers with severe psoriasis, and that risk may extend to preterm birth as well, although “we don’t know exactly the magnitude of that effect.”
Meanwhile, the consensus from the literature throughout dermatology, rheumatology, and gastroenterology is that anti-TNF agents do not cause birth defects or affect risk of preterm birth or low birth weight.
“The bigger question is what’s the impact on the immune system of the child,” Dr. Gordon said. Data from a small Scandinavian study suggested no increased risk of allergies, infections, or similar immunologic outcomes, but evidence remains limited.
Research has shown that infants’ exposure to anti-TNF medications persists for 3-6 months after delivery, and the American Academy of Pediatrics recommends delaying immunization in children exposed to anti-TNF agents in pregnancy. But actual evidence on immunization outcomes shows no reduced immunogenicity in such children.
“Clearly there is persistence of drug in the child, but in fact you have normal responses to immunization,” Dr. Gordon said. “The pediatricians’ argument is not based on data of what actually happens in immunization; it’s based on the fact that the drug is there.”
So what’s the bottom line?
The National Psoriasis Foundation recommends moisturizers and topical corticosteroids as first-line therapy in pregnant women with psoriasis, followed by phototherapy for second-line treatment.
But some patients will need systemic therapy during pregnancy, although it’s “best not to introduce more medications than needed in pregnancy,” Dr. Gordon said. For women with a significant flare-up or very persistent volatile disease, NPF first recommends cyclosporine, but Dr. Gordon disagrees and would go with anti-TNF agents before cyclosporine.
Data show that certolizumab is not actively transported across the placenta therefore reducing fetal exposure, so Dr. Gordon would specifically use certolizumab first, all other things being equal.
“But if the patient has been on another anti-TNF that’s been working, I don’t really have an issue with staying with it,” he added.
Existing evidence so far shows no impact in terms of genetic abnormalities, birth weight, premature birth, or even infant immunizations from anti-TNF agents. But beyond those, “there is simply not enough information on pregnancy with other forms of biologic therapy to draw conclusions.” Dr. Gordon said.
Dr. Gordon disclosed that he has received grant support and/or honoraria from Abbvie, Amgen, Almirall, and Boehringer Ingelheim.
CHICAGO – according to Kenneth B. Gordon, MD, professor and chair of dermatology at Medical College of Wisconsin in Milwaukee.
“We always had this concept that psoriasis gets better during pregnancy, that you might have 20% or 30% of patients who might have a little bit of a flare or maintain, but most keep on getting better,” Dr. Gordon told attendees at the American Academy of Dermatology summer meeting.
But the majority doesn’t mean everyone. He shared the case of one pregnant woman who came to him with severe psoriasis, covering the whole of her inner thigh, to underscore that severe cases do happen in pregnancy.
“These are real situations, and when you talk about maternal health, this woman is uncomfortable, she can’t sleep, and she’s having huge stressors that are not only going to impact her and her pregnancy but also that impact her child,” Dr. Gordon said.
Dr. Gordon clarified that he is not referring to patients with limited psoriasis or those who respond to topicals or phototherapy. But because methotrexate or acitretin are “hands-off during pregnancy,” he said, the only systemic therapy available for serious cases besides biologics is cyclosporine, which has its own risks. “We know that [cyclosporine] is associated with preterm labor and preterm birth and significant low birth weight, so even in the best scenario, when we have someone with persistent severe psoriasis in pregnancy, our best agent has a lot of downsides.”
Too few data exist on anti–interleukin (IL)-17 or anti-IL-23 therapies to draw conclusions about their use, he said, and but gastroenterology and rheumatology have a fair amount of evidence on anti–tumor necrosis factor (TNF) therapies during pregnancy because it’s usually too risky to stop treating conditions such as Crohn’s with these drugs. Still, Dr. Gordon cautioned, much of the data on biologics in pregnancy are conflicting.
The question of what medications to use, and in whom, centers on balancing risks to the fetus from the medication versus risks from the condition.
“There are impacts on the fetus of having severe psoriasis, and it varies with severity of disease,” Dr. Gordon said. For example, data suggest an increased likelihood of low birth weight in children born to mothers with severe psoriasis, and that risk may extend to preterm birth as well, although “we don’t know exactly the magnitude of that effect.”
Meanwhile, the consensus from the literature throughout dermatology, rheumatology, and gastroenterology is that anti-TNF agents do not cause birth defects or affect risk of preterm birth or low birth weight.
“The bigger question is what’s the impact on the immune system of the child,” Dr. Gordon said. Data from a small Scandinavian study suggested no increased risk of allergies, infections, or similar immunologic outcomes, but evidence remains limited.
Research has shown that infants’ exposure to anti-TNF medications persists for 3-6 months after delivery, and the American Academy of Pediatrics recommends delaying immunization in children exposed to anti-TNF agents in pregnancy. But actual evidence on immunization outcomes shows no reduced immunogenicity in such children.
“Clearly there is persistence of drug in the child, but in fact you have normal responses to immunization,” Dr. Gordon said. “The pediatricians’ argument is not based on data of what actually happens in immunization; it’s based on the fact that the drug is there.”
So what’s the bottom line?
The National Psoriasis Foundation recommends moisturizers and topical corticosteroids as first-line therapy in pregnant women with psoriasis, followed by phototherapy for second-line treatment.
But some patients will need systemic therapy during pregnancy, although it’s “best not to introduce more medications than needed in pregnancy,” Dr. Gordon said. For women with a significant flare-up or very persistent volatile disease, NPF first recommends cyclosporine, but Dr. Gordon disagrees and would go with anti-TNF agents before cyclosporine.
Data show that certolizumab is not actively transported across the placenta therefore reducing fetal exposure, so Dr. Gordon would specifically use certolizumab first, all other things being equal.
“But if the patient has been on another anti-TNF that’s been working, I don’t really have an issue with staying with it,” he added.
Existing evidence so far shows no impact in terms of genetic abnormalities, birth weight, premature birth, or even infant immunizations from anti-TNF agents. But beyond those, “there is simply not enough information on pregnancy with other forms of biologic therapy to draw conclusions.” Dr. Gordon said.
Dr. Gordon disclosed that he has received grant support and/or honoraria from Abbvie, Amgen, Almirall, and Boehringer Ingelheim.
EXPERT ANALYSIS FROM SUMMER AAD 2018
Cervical cancer: Who should you screen?
Resource
US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686. https://jamanetwork.com/journals/jama/fullarticle/2697704. Accessed September 14, 2018.
Resource
US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686. https://jamanetwork.com/journals/jama/fullarticle/2697704. Accessed September 14, 2018.
Resource
US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686. https://jamanetwork.com/journals/jama/fullarticle/2697704. Accessed September 14, 2018.