User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Physicians’ bad behavior seen at work, online by colleagues: Survey
“The days of surgeons throwing retractors across the OR and screaming at nurses and medical students are hopefully gone now,” said Barron Lerner, MD, PhD, professor of medicine at New York University Langone Health and author of “The Good Doctor: A Father, a Son, and the Evolution of Medical Ethics” (Boston: Beacon Press, 2014). “We’re not going to tolerate that as an institution.”
But, Dr. Lerner said, bad behavior still happens. And according to a recent Medscape survey, it seems to be on the rise.
For the 2022 Physicians Behaving Badly Report, more than 1,500 physicians shared how often they see fellow doctors misbehaving in person or on social media, and shared some of the worse behavior they’ve seen.
Though misconduct is still relatively uncommon among doctors, and most physicians say they’re proud of the high standards and attitudes of their colleagues, respondents to the survey did say that they’re seeing more frequent incidents of other doctors acting disrespectfully toward patients and coworkers, taking too casual an approach to patient privacy, and even acting angrily or aggressively at work. While the uptick is not substantial, it’s nonetheless worrying.
“I have increased concern for my colleagues,” said Drew Ramsey, MD, an assistant clinical professor of psychiatry at Columbia University, New York. “People forget that COVID has made the physician workplace incredibly stressful. Physicians are struggling with their mental health.”
Bullying and harassment top bad behavior
When it comes to what kind of bad behavior was reported, bullying or harassing clinicians and staff was the runaway winner, with 86% of respondents saying they’d seen this type of behavior at work at some time. Making fun of or disparaging patients behind their backs was a close second, at 82%.
Dr. Ramsey thinks that these figures may reflect a deeper understanding of and sensitivity to harassment and bullying. “Five years ago, we weren’t talking about microaggression,” he said. This heightened awareness might explain the fact that doctors reported witnessing physicians mistreating other medical personnel and/or bullying or harassing patients somewhat more often than in 2021’s report.
Docs were caught using racist language by 55% of respondents, and 44% reported seeing colleagues becoming physically aggressive with patients, clinicians, or staff. Other disturbing behaviors respondents witnessed included bullying or harassing patients (45%), inebriation at work (43%), lying about credentials (34%), trying to date a patient (30%), and committing a crime, such as embezzling or stealing (27%).
Women were seen misbehaving about one-third as often as their male counterparts. This could be because women are more likely to seek help, rather than the bottle, when the stress piles up. “Some misbehavior stems from alcohol abuse, and a higher percentage of men have an alcoholism problem,” Dr. Ramsey pointed out. “Also, male physicians have historically been reluctant to seek mental health assistance.”
Speaking up
Doctors are behaving badly slightly more often, and their colleagues are slightly more willing to speak up about that behavior. In 2021, 35% of physicians said they did nothing upon witnessing inappropriate behavior. In 2022’s survey, that number fell to 29%.
Respondents largely agreed (49%) that doctors should be verbally warned when they’ve behaved badly at work, yet only 39% reported speaking to a colleague who acted inappropriately, and only 27% reported the bad behavior to an authority.
Dr. Lerner pointed out that it is very difficult for doctors to speak up, even though they know they should. There are several reasons for their reticence.
“For one thing, we all have bad days, and the reporting physician may worry that he or she could do something similar in the future,” he said. “Also, there is the liability question. A doctor might think: ‘What if I’m wrong? What if I think someone has a drinking problem and they don’t, or I can’t prove it?’ If you’re the doctor who reported the misbehavior, you’re potentially opening a can of worms. So there’s all sorts of reasons people convince themselves they don’t have to report it.” But, he added, “if you see it and don’t report it, you’re in the wrong.”
Off the job
Work isn’t the only place where doctors observe their colleagues misbehaving. About 66% of respondents had seen disparaging behavior, and 42% had heard racist language, away from the hospital or clinic, according to the survey.
Bullying and harassment weren’t limited to work, either, with 45% reporting seeing a colleague engage in this behavior off campus, and 52% reporting witnessing a colleague inebriated in public. That’s actually down from 2021 when 58% of respondents said they witnessed inebriated doctors in public.
The public sphere has broadened in recent years to include social media, and there, too, doctors sometimes behave badly. However, 47% of doctors surveyed said they saw more inappropriate behavior in person than on social media.
When doctors do act out online, they make the same mistakes other professionals make. One respondent reported seeing a fellow physician “copying and posting an interoffice memo from work and badmouthing the company and the person who wrote the memo.” Another said: “Someone got fired and stalked the supervisor and posted aggressive things.”
Not all social media transgressions were work related. One respondent reported that “a physician posted pictures of herself at a bar with multiple ER staff members, without masks during COVID restriction,” and another reported a colleague posting “unbelievable, antiscientific information expressed as valid, factual material.”
Though posting nonfactual, unscientific, and potentially unsafe information is clearly an ethics violation, Dr. Lerner said, the boundaries around posting personal peccadillos are less clear. This is a part of “digital professionalism,” he explained, adding that there is a broad range of opinions on this. “I think it’s important to discuss these things. Interestingly, while the rules for behavior at the hospital have become more strict, the culture has become less strict.”
As one respondent put it: “What exactly is bad behavior? If you’re saying physicians should be allowed to sexually assault people and use drugs, then no. Can they wear a tiny bathing suit on vacation and drink cocktails with friends? Yeah.”
A version of this article first appeared on Medscape.com.
“The days of surgeons throwing retractors across the OR and screaming at nurses and medical students are hopefully gone now,” said Barron Lerner, MD, PhD, professor of medicine at New York University Langone Health and author of “The Good Doctor: A Father, a Son, and the Evolution of Medical Ethics” (Boston: Beacon Press, 2014). “We’re not going to tolerate that as an institution.”
But, Dr. Lerner said, bad behavior still happens. And according to a recent Medscape survey, it seems to be on the rise.
For the 2022 Physicians Behaving Badly Report, more than 1,500 physicians shared how often they see fellow doctors misbehaving in person or on social media, and shared some of the worse behavior they’ve seen.
Though misconduct is still relatively uncommon among doctors, and most physicians say they’re proud of the high standards and attitudes of their colleagues, respondents to the survey did say that they’re seeing more frequent incidents of other doctors acting disrespectfully toward patients and coworkers, taking too casual an approach to patient privacy, and even acting angrily or aggressively at work. While the uptick is not substantial, it’s nonetheless worrying.
“I have increased concern for my colleagues,” said Drew Ramsey, MD, an assistant clinical professor of psychiatry at Columbia University, New York. “People forget that COVID has made the physician workplace incredibly stressful. Physicians are struggling with their mental health.”
Bullying and harassment top bad behavior
When it comes to what kind of bad behavior was reported, bullying or harassing clinicians and staff was the runaway winner, with 86% of respondents saying they’d seen this type of behavior at work at some time. Making fun of or disparaging patients behind their backs was a close second, at 82%.
Dr. Ramsey thinks that these figures may reflect a deeper understanding of and sensitivity to harassment and bullying. “Five years ago, we weren’t talking about microaggression,” he said. This heightened awareness might explain the fact that doctors reported witnessing physicians mistreating other medical personnel and/or bullying or harassing patients somewhat more often than in 2021’s report.
Docs were caught using racist language by 55% of respondents, and 44% reported seeing colleagues becoming physically aggressive with patients, clinicians, or staff. Other disturbing behaviors respondents witnessed included bullying or harassing patients (45%), inebriation at work (43%), lying about credentials (34%), trying to date a patient (30%), and committing a crime, such as embezzling or stealing (27%).
Women were seen misbehaving about one-third as often as their male counterparts. This could be because women are more likely to seek help, rather than the bottle, when the stress piles up. “Some misbehavior stems from alcohol abuse, and a higher percentage of men have an alcoholism problem,” Dr. Ramsey pointed out. “Also, male physicians have historically been reluctant to seek mental health assistance.”
Speaking up
Doctors are behaving badly slightly more often, and their colleagues are slightly more willing to speak up about that behavior. In 2021, 35% of physicians said they did nothing upon witnessing inappropriate behavior. In 2022’s survey, that number fell to 29%.
Respondents largely agreed (49%) that doctors should be verbally warned when they’ve behaved badly at work, yet only 39% reported speaking to a colleague who acted inappropriately, and only 27% reported the bad behavior to an authority.
Dr. Lerner pointed out that it is very difficult for doctors to speak up, even though they know they should. There are several reasons for their reticence.
“For one thing, we all have bad days, and the reporting physician may worry that he or she could do something similar in the future,” he said. “Also, there is the liability question. A doctor might think: ‘What if I’m wrong? What if I think someone has a drinking problem and they don’t, or I can’t prove it?’ If you’re the doctor who reported the misbehavior, you’re potentially opening a can of worms. So there’s all sorts of reasons people convince themselves they don’t have to report it.” But, he added, “if you see it and don’t report it, you’re in the wrong.”
Off the job
Work isn’t the only place where doctors observe their colleagues misbehaving. About 66% of respondents had seen disparaging behavior, and 42% had heard racist language, away from the hospital or clinic, according to the survey.
Bullying and harassment weren’t limited to work, either, with 45% reporting seeing a colleague engage in this behavior off campus, and 52% reporting witnessing a colleague inebriated in public. That’s actually down from 2021 when 58% of respondents said they witnessed inebriated doctors in public.
The public sphere has broadened in recent years to include social media, and there, too, doctors sometimes behave badly. However, 47% of doctors surveyed said they saw more inappropriate behavior in person than on social media.
When doctors do act out online, they make the same mistakes other professionals make. One respondent reported seeing a fellow physician “copying and posting an interoffice memo from work and badmouthing the company and the person who wrote the memo.” Another said: “Someone got fired and stalked the supervisor and posted aggressive things.”
Not all social media transgressions were work related. One respondent reported that “a physician posted pictures of herself at a bar with multiple ER staff members, without masks during COVID restriction,” and another reported a colleague posting “unbelievable, antiscientific information expressed as valid, factual material.”
Though posting nonfactual, unscientific, and potentially unsafe information is clearly an ethics violation, Dr. Lerner said, the boundaries around posting personal peccadillos are less clear. This is a part of “digital professionalism,” he explained, adding that there is a broad range of opinions on this. “I think it’s important to discuss these things. Interestingly, while the rules for behavior at the hospital have become more strict, the culture has become less strict.”
As one respondent put it: “What exactly is bad behavior? If you’re saying physicians should be allowed to sexually assault people and use drugs, then no. Can they wear a tiny bathing suit on vacation and drink cocktails with friends? Yeah.”
A version of this article first appeared on Medscape.com.
“The days of surgeons throwing retractors across the OR and screaming at nurses and medical students are hopefully gone now,” said Barron Lerner, MD, PhD, professor of medicine at New York University Langone Health and author of “The Good Doctor: A Father, a Son, and the Evolution of Medical Ethics” (Boston: Beacon Press, 2014). “We’re not going to tolerate that as an institution.”
But, Dr. Lerner said, bad behavior still happens. And according to a recent Medscape survey, it seems to be on the rise.
For the 2022 Physicians Behaving Badly Report, more than 1,500 physicians shared how often they see fellow doctors misbehaving in person or on social media, and shared some of the worse behavior they’ve seen.
Though misconduct is still relatively uncommon among doctors, and most physicians say they’re proud of the high standards and attitudes of their colleagues, respondents to the survey did say that they’re seeing more frequent incidents of other doctors acting disrespectfully toward patients and coworkers, taking too casual an approach to patient privacy, and even acting angrily or aggressively at work. While the uptick is not substantial, it’s nonetheless worrying.
“I have increased concern for my colleagues,” said Drew Ramsey, MD, an assistant clinical professor of psychiatry at Columbia University, New York. “People forget that COVID has made the physician workplace incredibly stressful. Physicians are struggling with their mental health.”
Bullying and harassment top bad behavior
When it comes to what kind of bad behavior was reported, bullying or harassing clinicians and staff was the runaway winner, with 86% of respondents saying they’d seen this type of behavior at work at some time. Making fun of or disparaging patients behind their backs was a close second, at 82%.
Dr. Ramsey thinks that these figures may reflect a deeper understanding of and sensitivity to harassment and bullying. “Five years ago, we weren’t talking about microaggression,” he said. This heightened awareness might explain the fact that doctors reported witnessing physicians mistreating other medical personnel and/or bullying or harassing patients somewhat more often than in 2021’s report.
Docs were caught using racist language by 55% of respondents, and 44% reported seeing colleagues becoming physically aggressive with patients, clinicians, or staff. Other disturbing behaviors respondents witnessed included bullying or harassing patients (45%), inebriation at work (43%), lying about credentials (34%), trying to date a patient (30%), and committing a crime, such as embezzling or stealing (27%).
Women were seen misbehaving about one-third as often as their male counterparts. This could be because women are more likely to seek help, rather than the bottle, when the stress piles up. “Some misbehavior stems from alcohol abuse, and a higher percentage of men have an alcoholism problem,” Dr. Ramsey pointed out. “Also, male physicians have historically been reluctant to seek mental health assistance.”
Speaking up
Doctors are behaving badly slightly more often, and their colleagues are slightly more willing to speak up about that behavior. In 2021, 35% of physicians said they did nothing upon witnessing inappropriate behavior. In 2022’s survey, that number fell to 29%.
Respondents largely agreed (49%) that doctors should be verbally warned when they’ve behaved badly at work, yet only 39% reported speaking to a colleague who acted inappropriately, and only 27% reported the bad behavior to an authority.
Dr. Lerner pointed out that it is very difficult for doctors to speak up, even though they know they should. There are several reasons for their reticence.
“For one thing, we all have bad days, and the reporting physician may worry that he or she could do something similar in the future,” he said. “Also, there is the liability question. A doctor might think: ‘What if I’m wrong? What if I think someone has a drinking problem and they don’t, or I can’t prove it?’ If you’re the doctor who reported the misbehavior, you’re potentially opening a can of worms. So there’s all sorts of reasons people convince themselves they don’t have to report it.” But, he added, “if you see it and don’t report it, you’re in the wrong.”
Off the job
Work isn’t the only place where doctors observe their colleagues misbehaving. About 66% of respondents had seen disparaging behavior, and 42% had heard racist language, away from the hospital or clinic, according to the survey.
Bullying and harassment weren’t limited to work, either, with 45% reporting seeing a colleague engage in this behavior off campus, and 52% reporting witnessing a colleague inebriated in public. That’s actually down from 2021 when 58% of respondents said they witnessed inebriated doctors in public.
The public sphere has broadened in recent years to include social media, and there, too, doctors sometimes behave badly. However, 47% of doctors surveyed said they saw more inappropriate behavior in person than on social media.
When doctors do act out online, they make the same mistakes other professionals make. One respondent reported seeing a fellow physician “copying and posting an interoffice memo from work and badmouthing the company and the person who wrote the memo.” Another said: “Someone got fired and stalked the supervisor and posted aggressive things.”
Not all social media transgressions were work related. One respondent reported that “a physician posted pictures of herself at a bar with multiple ER staff members, without masks during COVID restriction,” and another reported a colleague posting “unbelievable, antiscientific information expressed as valid, factual material.”
Though posting nonfactual, unscientific, and potentially unsafe information is clearly an ethics violation, Dr. Lerner said, the boundaries around posting personal peccadillos are less clear. This is a part of “digital professionalism,” he explained, adding that there is a broad range of opinions on this. “I think it’s important to discuss these things. Interestingly, while the rules for behavior at the hospital have become more strict, the culture has become less strict.”
As one respondent put it: “What exactly is bad behavior? If you’re saying physicians should be allowed to sexually assault people and use drugs, then no. Can they wear a tiny bathing suit on vacation and drink cocktails with friends? Yeah.”
A version of this article first appeared on Medscape.com.
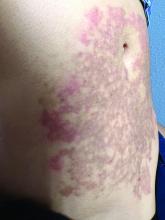
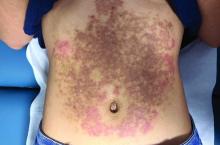
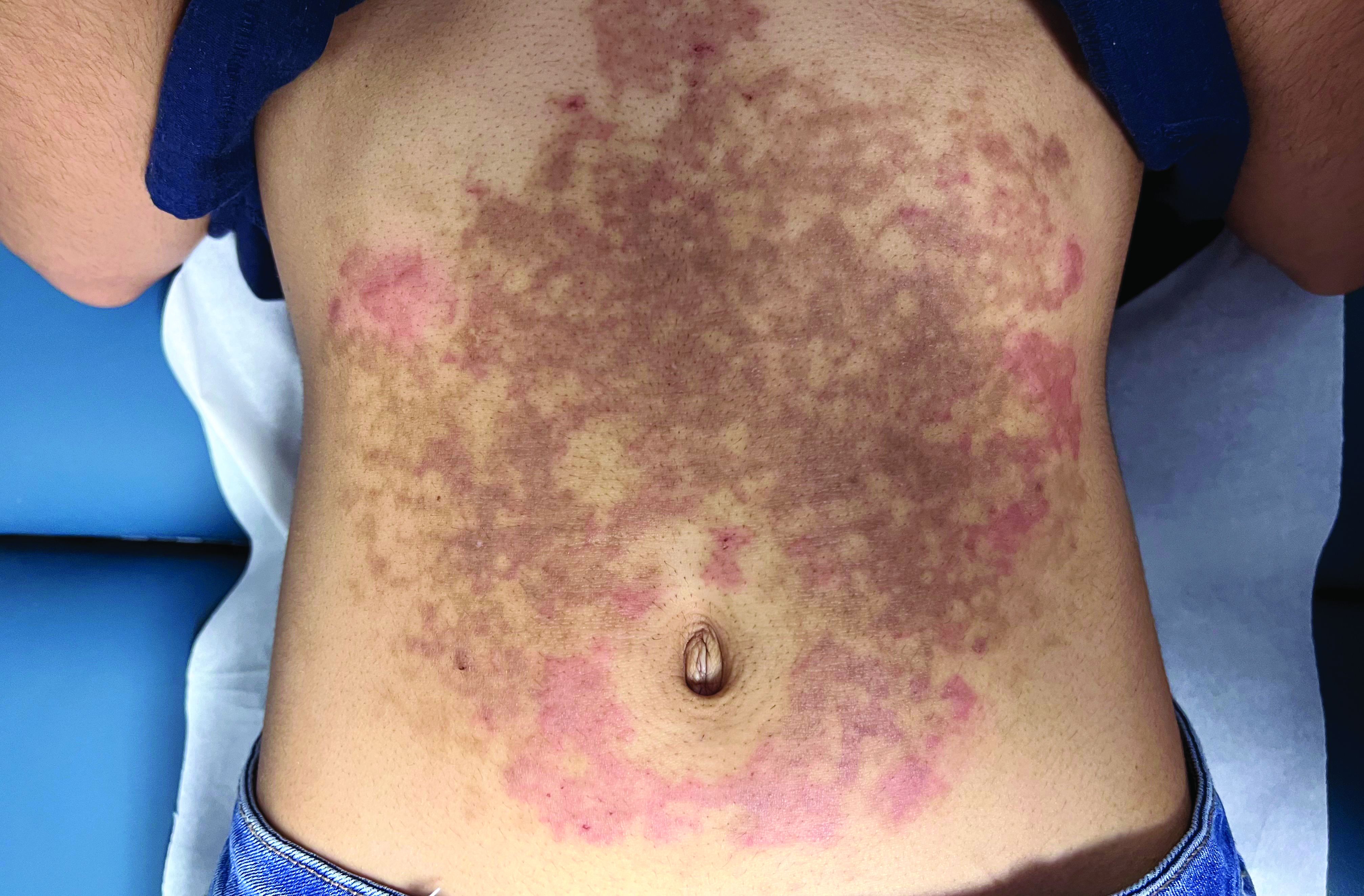
A White female presented with pruritic, reticulated, erythematous plaques on the abdomen
It is characterized by pruritic, erythematous papules, papulovesicles, and vesicles that appear in a reticular pattern, most commonly on the trunk. The lesions are typically followed by postinflammatory hyperpigmentation (PIH).
Although PP has been described in people of all races, ages, and sexes, it is predominantly observed in Japan, often in female young adults. Triggers may include a ketogenic diet, diabetes mellitus, and pregnancy. Friction and contact allergic reactions to chrome or nickel have been proposed as exogenous trigger factors. Individual cases of Sjögren’s syndrome, Helicobacter pylori infections, and adult Still syndrome have also been associated with recurrent eruptions.
The diagnosis of PP is made both clinically and by biopsy. The histological features vary according to the stage of the disease. In early-stage disease, superficial and perivascular infiltration of neutrophils are prominent. Later stages are characterized by spongiosis and necrotic keratinocytes.
The first-line therapy for prurigo pigmentosa is oral minocycline. However, for some patients, doxycycline, macrolide antibiotics, or dapsone may be indicated. Adding carbohydrates to a keto diet may be helpful. In this patient, a punch biopsy was performed, which revealed an interface dermatitis with eosinophils and neutrophils, consistent with prurigo pigmentosa. The cause of her PP remains idiopathic. She was treated with 100 mg doxycycline twice a day, which resulted in a resolution of active lesions. The patient did have postinflammatory hyperpigmentation.
This case and photo were submitted by Brooke Resh Sateesh, MD, of San Diego Family Dermatology, San Diego, California, and Mina Zulal, University Medical Center Hamburg-Eppendorf (UKE), Hamburg, Germany. Dr. Bilu Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Beutler et al. Am J Clin Dermatol. 2015 Dec;16(6):533-43.
2. Kim et al. J Dermatol. 2012 Nov;39(11):891-7.
3. Mufti et al. JAAD Int. 2021 Apr 10;3:79-87.
It is characterized by pruritic, erythematous papules, papulovesicles, and vesicles that appear in a reticular pattern, most commonly on the trunk. The lesions are typically followed by postinflammatory hyperpigmentation (PIH).
Although PP has been described in people of all races, ages, and sexes, it is predominantly observed in Japan, often in female young adults. Triggers may include a ketogenic diet, diabetes mellitus, and pregnancy. Friction and contact allergic reactions to chrome or nickel have been proposed as exogenous trigger factors. Individual cases of Sjögren’s syndrome, Helicobacter pylori infections, and adult Still syndrome have also been associated with recurrent eruptions.
The diagnosis of PP is made both clinically and by biopsy. The histological features vary according to the stage of the disease. In early-stage disease, superficial and perivascular infiltration of neutrophils are prominent. Later stages are characterized by spongiosis and necrotic keratinocytes.
The first-line therapy for prurigo pigmentosa is oral minocycline. However, for some patients, doxycycline, macrolide antibiotics, or dapsone may be indicated. Adding carbohydrates to a keto diet may be helpful. In this patient, a punch biopsy was performed, which revealed an interface dermatitis with eosinophils and neutrophils, consistent with prurigo pigmentosa. The cause of her PP remains idiopathic. She was treated with 100 mg doxycycline twice a day, which resulted in a resolution of active lesions. The patient did have postinflammatory hyperpigmentation.
This case and photo were submitted by Brooke Resh Sateesh, MD, of San Diego Family Dermatology, San Diego, California, and Mina Zulal, University Medical Center Hamburg-Eppendorf (UKE), Hamburg, Germany. Dr. Bilu Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Beutler et al. Am J Clin Dermatol. 2015 Dec;16(6):533-43.
2. Kim et al. J Dermatol. 2012 Nov;39(11):891-7.
3. Mufti et al. JAAD Int. 2021 Apr 10;3:79-87.
It is characterized by pruritic, erythematous papules, papulovesicles, and vesicles that appear in a reticular pattern, most commonly on the trunk. The lesions are typically followed by postinflammatory hyperpigmentation (PIH).
Although PP has been described in people of all races, ages, and sexes, it is predominantly observed in Japan, often in female young adults. Triggers may include a ketogenic diet, diabetes mellitus, and pregnancy. Friction and contact allergic reactions to chrome or nickel have been proposed as exogenous trigger factors. Individual cases of Sjögren’s syndrome, Helicobacter pylori infections, and adult Still syndrome have also been associated with recurrent eruptions.
The diagnosis of PP is made both clinically and by biopsy. The histological features vary according to the stage of the disease. In early-stage disease, superficial and perivascular infiltration of neutrophils are prominent. Later stages are characterized by spongiosis and necrotic keratinocytes.
The first-line therapy for prurigo pigmentosa is oral minocycline. However, for some patients, doxycycline, macrolide antibiotics, or dapsone may be indicated. Adding carbohydrates to a keto diet may be helpful. In this patient, a punch biopsy was performed, which revealed an interface dermatitis with eosinophils and neutrophils, consistent with prurigo pigmentosa. The cause of her PP remains idiopathic. She was treated with 100 mg doxycycline twice a day, which resulted in a resolution of active lesions. The patient did have postinflammatory hyperpigmentation.
This case and photo were submitted by Brooke Resh Sateesh, MD, of San Diego Family Dermatology, San Diego, California, and Mina Zulal, University Medical Center Hamburg-Eppendorf (UKE), Hamburg, Germany. Dr. Bilu Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Beutler et al. Am J Clin Dermatol. 2015 Dec;16(6):533-43.
2. Kim et al. J Dermatol. 2012 Nov;39(11):891-7.
3. Mufti et al. JAAD Int. 2021 Apr 10;3:79-87.
Primary care now offering physicians the 26.7-hour day
Taking ‘not enough hours in the day’ to new heights
It’s no secret that there’s a big doctor shortage in the United States. Going through medical school is long, expensive, and stressful, and it’s not like those long, stressful hours stop once you finally do get that degree. There is, however, an excellent reason to take that dive into doctorhood: You’ll gain mastery over time itself.
A study from the University of Chicago, Johns Hopkins University, and Imperial College London has revealed the truth. By using data pulled from the National Health and Nutrition Examination Survey, the researchers found that primary care physicians who see an average number of patients and follow all the current national guidelines for preventive care, chronic disease care, and acute care – plus administrative tasks – must work 26.7 hours a day. That works out to 14.1 hours of preventive care, 7.2 hours of chronic disease care, 2.2 hours of acute care, and 3.2 hours of documentation and inbox management.
Astute readers may note that this is a bit more than the traditional 8-hour workday. It is, in fact, more hours than there actually are in a day. As it turns out, Doctor Strange is more of a documentary than …
Hang on, we’re receiving word that doctors are not in fact wizards who can bend time and space to their will, nor are they sitting on a stash of Time-Turners they saved from the Ministry of Magic before Voldemort destroyed them all. They are, according to the study, overworked and overburdened with too many things and too little time. This is why outcomes haven’t improved despite technological advances and why burnout is so common. We’d be burned out too, having to work temporally impossible hours.
The study authors suggested a team-based approach to medicine that would spread the workload out to nurses, physician assistants, dietitians, etc., estimating that about two-thirds of what a primary care physician does can be handled by someone else. A team-based approach would reduce the physician’s required hours down to 9.3 hours a day, which is at least physically possible. It’s either that or we make the day longer, which sounds like the plot of an episode of Futurama. Swap overwork for global warming and a longer day for a longer year and it is actually the plot of an episode of Futurama.
After a hard day of thinking, brains need their rest
Do you ever feel like you have no more capacity to think or make any more decisions after a long day at work? Do you need a few extra cups of coffee to even make it through the day, even though you’re mostly just sitting around talking and typing? Have we got the research for you: Mental exhaustion is an actual thing. Imagine that double whammy of having a job that’s physically and mentally demanding.
A recent study in Current Biology explained why we feel so exhausted after doing something mentally demanding for several hours. Over that time, glutamate builds up in synapses of the prefrontal cortex, which affects our decision making and leads to cognitive lethargy. Your brain eventually becomes more interested in tasks that are less mentally fatiguing, and that’s probably why you’re reading this LOTME right now instead of getting back to work.
“Our findings show that cognitive work results in a true functional alteration – accumulation of noxious substances – so fatigue would indeed be a signal that makes us stop working but for a different purpose: to preserve the integrity of brain functioning,” senior author Mathias Pessiglione of Pitié-Salpêtrière University, Paris, said in a written statement.
The group of researchers conducted studies by using magnetic resonance spectroscopy to look at two groups of people over the course of a workday: One group had mentally tasking jobs and one didn’t. Those who had to think harder for their jobs had more signs of fatigue, such as reduced pupil dilation and glutamate in synapses of the prefrontal cortex. They also looked for more rewards that required less thinking.
For those whose mentally exhausting jobs probably won’t get better or change, the researchers suggest getting as much rest as possible. Those who don’t have that option will have to continue drinking those 7 cups of coffee a day. ... and reading LOTME.
Hmm, might be a new tagline for us in there somewhere. LOTME: Tired brains love us? When you’re too tired to think, think of LOTME? You can’t spell mental exhaustion without L-O-T-M-E?
Testosterone shows its warm and fuzzy side
Stereotypically, men are loud, knuckle-dragging Neanderthals. The hair coming out of our faces is kind of a dead giveaway, right? We grunt, we scratch, we start wars, we watch sports on TV. But why? It’s the testosterone. Everyone knows that. Testosterone makes men aggressive … or does it?
Since this sort of research generally isn’t done with actual men, investigators at Emory University used Mongolian gerbils. The advantage being that males exhibit cuddling behavior after females become pregnant and they don’t watch a lot of sports on TV. They introduced a male and female gerbil, who then formed a pair bond and the female became pregnant. When the male started displaying cuddling behaviors, the researchers injected him with testosterone, expecting to see his antisocial side.
“Instead, we were surprised that a male gerbil became even more cuddly and prosocial with his partner. He became like ‘super partner,’ ” lead author Aubrey Kelly, PhD, said in a written statement from the university.
For the next experiment, the female was removed and another male was introduced to a male who had already received a testosterone injection. That male was surprisingly unaggressive toward the intruder, at least initially. Then he received a second injection of testosterone. “It was like they suddenly woke up and realized they weren’t supposed to be friendly in that context,” Dr. Kelly said.
The testosterone seemed to influence the activity of oxytocin, the so-called “love hormone,” the investigators suggested. “It’s surprising because normally we think of testosterone as increasing sexual behaviors and aggression. But we’ve shown that it can have more nuanced effects, depending on the social context.”
The researchers were not as surprised when their use of the phrase “super partner” led to a bidding war between DC and Marvel. Then came the contact from the Department of Defense, wondering about weaponized testosterone: Would it be possible for some sort of bomb to turn Vlad “the Impaler” Putin into Vlad “the Cuddler” Putin?
Are instruments spreading the sounds of COVID?
COVID restrictions are practically a thing of the past now. With more people laxed on being in close proximity to each other and the CDC not even recommending social distancing anymore, live concerts and events are back in full swing. But with new variants on the rise and people being a little more cautious, should we be worried about musical instruments spreading COVID?
Yes and no.
A study published in Physics of Fluids looked at wind instruments specifically and how much aerosol is produced and dispersed when playing them. For the study, the investigators measured fog particles with a laser and aerosol concentration with a particle counter to see how fast these particles decay in the air from the distance of the instrument.
Musicians in an orchestra typically would sit close together to produce the best sound, but with COVID that became an issue, senior author Paulo Arratia of the University of Pennsylvania, Philadelphia, noted in a separate written statement. By looking at the distance traveled by the particles coming from a single instrument and how quickly they decayed, they could determine if sitting in close proximity is an actual threat.
Well, the threat was no greater than talking to someone face to face. Particle exit speeds were lower than for a cough or a sneeze, and the maximum decay length was 2 meters from the instrument’s opening.
But that’s just one instrument: What kind of impact does a whole orchestra have on a space? The researchers are looking into that too, but for now they suggest that musicians continue to stay 6 feet away from each other.
So, yeah, there is a threat, but it’s probably safer for you to see that orchestra than have someone sneeze on you.
Music to our ears.
Taking ‘not enough hours in the day’ to new heights
It’s no secret that there’s a big doctor shortage in the United States. Going through medical school is long, expensive, and stressful, and it’s not like those long, stressful hours stop once you finally do get that degree. There is, however, an excellent reason to take that dive into doctorhood: You’ll gain mastery over time itself.
A study from the University of Chicago, Johns Hopkins University, and Imperial College London has revealed the truth. By using data pulled from the National Health and Nutrition Examination Survey, the researchers found that primary care physicians who see an average number of patients and follow all the current national guidelines for preventive care, chronic disease care, and acute care – plus administrative tasks – must work 26.7 hours a day. That works out to 14.1 hours of preventive care, 7.2 hours of chronic disease care, 2.2 hours of acute care, and 3.2 hours of documentation and inbox management.
Astute readers may note that this is a bit more than the traditional 8-hour workday. It is, in fact, more hours than there actually are in a day. As it turns out, Doctor Strange is more of a documentary than …
Hang on, we’re receiving word that doctors are not in fact wizards who can bend time and space to their will, nor are they sitting on a stash of Time-Turners they saved from the Ministry of Magic before Voldemort destroyed them all. They are, according to the study, overworked and overburdened with too many things and too little time. This is why outcomes haven’t improved despite technological advances and why burnout is so common. We’d be burned out too, having to work temporally impossible hours.
The study authors suggested a team-based approach to medicine that would spread the workload out to nurses, physician assistants, dietitians, etc., estimating that about two-thirds of what a primary care physician does can be handled by someone else. A team-based approach would reduce the physician’s required hours down to 9.3 hours a day, which is at least physically possible. It’s either that or we make the day longer, which sounds like the plot of an episode of Futurama. Swap overwork for global warming and a longer day for a longer year and it is actually the plot of an episode of Futurama.
After a hard day of thinking, brains need their rest
Do you ever feel like you have no more capacity to think or make any more decisions after a long day at work? Do you need a few extra cups of coffee to even make it through the day, even though you’re mostly just sitting around talking and typing? Have we got the research for you: Mental exhaustion is an actual thing. Imagine that double whammy of having a job that’s physically and mentally demanding.
A recent study in Current Biology explained why we feel so exhausted after doing something mentally demanding for several hours. Over that time, glutamate builds up in synapses of the prefrontal cortex, which affects our decision making and leads to cognitive lethargy. Your brain eventually becomes more interested in tasks that are less mentally fatiguing, and that’s probably why you’re reading this LOTME right now instead of getting back to work.
“Our findings show that cognitive work results in a true functional alteration – accumulation of noxious substances – so fatigue would indeed be a signal that makes us stop working but for a different purpose: to preserve the integrity of brain functioning,” senior author Mathias Pessiglione of Pitié-Salpêtrière University, Paris, said in a written statement.
The group of researchers conducted studies by using magnetic resonance spectroscopy to look at two groups of people over the course of a workday: One group had mentally tasking jobs and one didn’t. Those who had to think harder for their jobs had more signs of fatigue, such as reduced pupil dilation and glutamate in synapses of the prefrontal cortex. They also looked for more rewards that required less thinking.
For those whose mentally exhausting jobs probably won’t get better or change, the researchers suggest getting as much rest as possible. Those who don’t have that option will have to continue drinking those 7 cups of coffee a day. ... and reading LOTME.
Hmm, might be a new tagline for us in there somewhere. LOTME: Tired brains love us? When you’re too tired to think, think of LOTME? You can’t spell mental exhaustion without L-O-T-M-E?
Testosterone shows its warm and fuzzy side
Stereotypically, men are loud, knuckle-dragging Neanderthals. The hair coming out of our faces is kind of a dead giveaway, right? We grunt, we scratch, we start wars, we watch sports on TV. But why? It’s the testosterone. Everyone knows that. Testosterone makes men aggressive … or does it?
Since this sort of research generally isn’t done with actual men, investigators at Emory University used Mongolian gerbils. The advantage being that males exhibit cuddling behavior after females become pregnant and they don’t watch a lot of sports on TV. They introduced a male and female gerbil, who then formed a pair bond and the female became pregnant. When the male started displaying cuddling behaviors, the researchers injected him with testosterone, expecting to see his antisocial side.
“Instead, we were surprised that a male gerbil became even more cuddly and prosocial with his partner. He became like ‘super partner,’ ” lead author Aubrey Kelly, PhD, said in a written statement from the university.
For the next experiment, the female was removed and another male was introduced to a male who had already received a testosterone injection. That male was surprisingly unaggressive toward the intruder, at least initially. Then he received a second injection of testosterone. “It was like they suddenly woke up and realized they weren’t supposed to be friendly in that context,” Dr. Kelly said.
The testosterone seemed to influence the activity of oxytocin, the so-called “love hormone,” the investigators suggested. “It’s surprising because normally we think of testosterone as increasing sexual behaviors and aggression. But we’ve shown that it can have more nuanced effects, depending on the social context.”
The researchers were not as surprised when their use of the phrase “super partner” led to a bidding war between DC and Marvel. Then came the contact from the Department of Defense, wondering about weaponized testosterone: Would it be possible for some sort of bomb to turn Vlad “the Impaler” Putin into Vlad “the Cuddler” Putin?
Are instruments spreading the sounds of COVID?
COVID restrictions are practically a thing of the past now. With more people laxed on being in close proximity to each other and the CDC not even recommending social distancing anymore, live concerts and events are back in full swing. But with new variants on the rise and people being a little more cautious, should we be worried about musical instruments spreading COVID?
Yes and no.
A study published in Physics of Fluids looked at wind instruments specifically and how much aerosol is produced and dispersed when playing them. For the study, the investigators measured fog particles with a laser and aerosol concentration with a particle counter to see how fast these particles decay in the air from the distance of the instrument.
Musicians in an orchestra typically would sit close together to produce the best sound, but with COVID that became an issue, senior author Paulo Arratia of the University of Pennsylvania, Philadelphia, noted in a separate written statement. By looking at the distance traveled by the particles coming from a single instrument and how quickly they decayed, they could determine if sitting in close proximity is an actual threat.
Well, the threat was no greater than talking to someone face to face. Particle exit speeds were lower than for a cough or a sneeze, and the maximum decay length was 2 meters from the instrument’s opening.
But that’s just one instrument: What kind of impact does a whole orchestra have on a space? The researchers are looking into that too, but for now they suggest that musicians continue to stay 6 feet away from each other.
So, yeah, there is a threat, but it’s probably safer for you to see that orchestra than have someone sneeze on you.
Music to our ears.
Taking ‘not enough hours in the day’ to new heights
It’s no secret that there’s a big doctor shortage in the United States. Going through medical school is long, expensive, and stressful, and it’s not like those long, stressful hours stop once you finally do get that degree. There is, however, an excellent reason to take that dive into doctorhood: You’ll gain mastery over time itself.
A study from the University of Chicago, Johns Hopkins University, and Imperial College London has revealed the truth. By using data pulled from the National Health and Nutrition Examination Survey, the researchers found that primary care physicians who see an average number of patients and follow all the current national guidelines for preventive care, chronic disease care, and acute care – plus administrative tasks – must work 26.7 hours a day. That works out to 14.1 hours of preventive care, 7.2 hours of chronic disease care, 2.2 hours of acute care, and 3.2 hours of documentation and inbox management.
Astute readers may note that this is a bit more than the traditional 8-hour workday. It is, in fact, more hours than there actually are in a day. As it turns out, Doctor Strange is more of a documentary than …
Hang on, we’re receiving word that doctors are not in fact wizards who can bend time and space to their will, nor are they sitting on a stash of Time-Turners they saved from the Ministry of Magic before Voldemort destroyed them all. They are, according to the study, overworked and overburdened with too many things and too little time. This is why outcomes haven’t improved despite technological advances and why burnout is so common. We’d be burned out too, having to work temporally impossible hours.
The study authors suggested a team-based approach to medicine that would spread the workload out to nurses, physician assistants, dietitians, etc., estimating that about two-thirds of what a primary care physician does can be handled by someone else. A team-based approach would reduce the physician’s required hours down to 9.3 hours a day, which is at least physically possible. It’s either that or we make the day longer, which sounds like the plot of an episode of Futurama. Swap overwork for global warming and a longer day for a longer year and it is actually the plot of an episode of Futurama.
After a hard day of thinking, brains need their rest
Do you ever feel like you have no more capacity to think or make any more decisions after a long day at work? Do you need a few extra cups of coffee to even make it through the day, even though you’re mostly just sitting around talking and typing? Have we got the research for you: Mental exhaustion is an actual thing. Imagine that double whammy of having a job that’s physically and mentally demanding.
A recent study in Current Biology explained why we feel so exhausted after doing something mentally demanding for several hours. Over that time, glutamate builds up in synapses of the prefrontal cortex, which affects our decision making and leads to cognitive lethargy. Your brain eventually becomes more interested in tasks that are less mentally fatiguing, and that’s probably why you’re reading this LOTME right now instead of getting back to work.
“Our findings show that cognitive work results in a true functional alteration – accumulation of noxious substances – so fatigue would indeed be a signal that makes us stop working but for a different purpose: to preserve the integrity of brain functioning,” senior author Mathias Pessiglione of Pitié-Salpêtrière University, Paris, said in a written statement.
The group of researchers conducted studies by using magnetic resonance spectroscopy to look at two groups of people over the course of a workday: One group had mentally tasking jobs and one didn’t. Those who had to think harder for their jobs had more signs of fatigue, such as reduced pupil dilation and glutamate in synapses of the prefrontal cortex. They also looked for more rewards that required less thinking.
For those whose mentally exhausting jobs probably won’t get better or change, the researchers suggest getting as much rest as possible. Those who don’t have that option will have to continue drinking those 7 cups of coffee a day. ... and reading LOTME.
Hmm, might be a new tagline for us in there somewhere. LOTME: Tired brains love us? When you’re too tired to think, think of LOTME? You can’t spell mental exhaustion without L-O-T-M-E?
Testosterone shows its warm and fuzzy side
Stereotypically, men are loud, knuckle-dragging Neanderthals. The hair coming out of our faces is kind of a dead giveaway, right? We grunt, we scratch, we start wars, we watch sports on TV. But why? It’s the testosterone. Everyone knows that. Testosterone makes men aggressive … or does it?
Since this sort of research generally isn’t done with actual men, investigators at Emory University used Mongolian gerbils. The advantage being that males exhibit cuddling behavior after females become pregnant and they don’t watch a lot of sports on TV. They introduced a male and female gerbil, who then formed a pair bond and the female became pregnant. When the male started displaying cuddling behaviors, the researchers injected him with testosterone, expecting to see his antisocial side.
“Instead, we were surprised that a male gerbil became even more cuddly and prosocial with his partner. He became like ‘super partner,’ ” lead author Aubrey Kelly, PhD, said in a written statement from the university.
For the next experiment, the female was removed and another male was introduced to a male who had already received a testosterone injection. That male was surprisingly unaggressive toward the intruder, at least initially. Then he received a second injection of testosterone. “It was like they suddenly woke up and realized they weren’t supposed to be friendly in that context,” Dr. Kelly said.
The testosterone seemed to influence the activity of oxytocin, the so-called “love hormone,” the investigators suggested. “It’s surprising because normally we think of testosterone as increasing sexual behaviors and aggression. But we’ve shown that it can have more nuanced effects, depending on the social context.”
The researchers were not as surprised when their use of the phrase “super partner” led to a bidding war between DC and Marvel. Then came the contact from the Department of Defense, wondering about weaponized testosterone: Would it be possible for some sort of bomb to turn Vlad “the Impaler” Putin into Vlad “the Cuddler” Putin?
Are instruments spreading the sounds of COVID?
COVID restrictions are practically a thing of the past now. With more people laxed on being in close proximity to each other and the CDC not even recommending social distancing anymore, live concerts and events are back in full swing. But with new variants on the rise and people being a little more cautious, should we be worried about musical instruments spreading COVID?
Yes and no.
A study published in Physics of Fluids looked at wind instruments specifically and how much aerosol is produced and dispersed when playing them. For the study, the investigators measured fog particles with a laser and aerosol concentration with a particle counter to see how fast these particles decay in the air from the distance of the instrument.
Musicians in an orchestra typically would sit close together to produce the best sound, but with COVID that became an issue, senior author Paulo Arratia of the University of Pennsylvania, Philadelphia, noted in a separate written statement. By looking at the distance traveled by the particles coming from a single instrument and how quickly they decayed, they could determine if sitting in close proximity is an actual threat.
Well, the threat was no greater than talking to someone face to face. Particle exit speeds were lower than for a cough or a sneeze, and the maximum decay length was 2 meters from the instrument’s opening.
But that’s just one instrument: What kind of impact does a whole orchestra have on a space? The researchers are looking into that too, but for now they suggest that musicians continue to stay 6 feet away from each other.
So, yeah, there is a threat, but it’s probably safer for you to see that orchestra than have someone sneeze on you.
Music to our ears.
FDA approves adalimumab-bwwd biosimilar (Hadlima) in high-concentration form
The U.S. Food and Drug Administration today approved a citrate-free, high-concentration formulation of adalimumab-bwwd (Hadlima), the manufacturer, Samsung Bioepis, and its commercialization partner Organon said in an announcement.
Hadlima is a biosimilar of the tumor necrosis factor inhibitor reference product adalimumab (Humira).
Hadlima was first approved in July 2019 in a citrated, 50-mg/mL formulation. The new citrate-free, 100-mg/mL version will be available in prefilled syringe and autoinjector options.
The 100-mg/mL formulation is indicated for the same seven conditions as its 50-mg/mL counterpart: rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, adult and pediatric Crohn’s disease, and ulcerative colitis.
The approval was based on clinical data from a randomized, single-blind, two-arm, parallel group, single-dose study that compared the pharmacokinetics, safety, tolerability, and immunogenicity of the 100-mg/mL and 50-mg/mL formulations of Hadlima in healthy volunteers.
Both low- and high-concentration formulations of Humira are currently marketed in the United States. Organon said that it expects to market Hadlima in the United States on or after July 1, 2023, in accordance with a licensing agreement with AbbVie.
The prescribing information for Hadlima includes specific warnings and areas of concern. The drug should not be administered to individuals who are known to be hypersensitive to adalimumab. The drug may lower the ability of the immune system to fight infections and may increase risk of infections, including serious infections leading to hospitalization or death, such as tuberculosis, bacterial sepsis, invasive fungal infections (such as histoplasmosis), and infections attributable to other opportunistic pathogens.
A test for latent TB infection should be given before administration, and treatment of TB should begin before administration of Hadlima.
Patients taking Hadlima should not take a live vaccine.
The most common adverse effects (incidence > 10%) include infections (for example, upper respiratory infections, sinusitis), injection site reactions, headache, and rash.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration today approved a citrate-free, high-concentration formulation of adalimumab-bwwd (Hadlima), the manufacturer, Samsung Bioepis, and its commercialization partner Organon said in an announcement.
Hadlima is a biosimilar of the tumor necrosis factor inhibitor reference product adalimumab (Humira).
Hadlima was first approved in July 2019 in a citrated, 50-mg/mL formulation. The new citrate-free, 100-mg/mL version will be available in prefilled syringe and autoinjector options.
The 100-mg/mL formulation is indicated for the same seven conditions as its 50-mg/mL counterpart: rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, adult and pediatric Crohn’s disease, and ulcerative colitis.
The approval was based on clinical data from a randomized, single-blind, two-arm, parallel group, single-dose study that compared the pharmacokinetics, safety, tolerability, and immunogenicity of the 100-mg/mL and 50-mg/mL formulations of Hadlima in healthy volunteers.
Both low- and high-concentration formulations of Humira are currently marketed in the United States. Organon said that it expects to market Hadlima in the United States on or after July 1, 2023, in accordance with a licensing agreement with AbbVie.
The prescribing information for Hadlima includes specific warnings and areas of concern. The drug should not be administered to individuals who are known to be hypersensitive to adalimumab. The drug may lower the ability of the immune system to fight infections and may increase risk of infections, including serious infections leading to hospitalization or death, such as tuberculosis, bacterial sepsis, invasive fungal infections (such as histoplasmosis), and infections attributable to other opportunistic pathogens.
A test for latent TB infection should be given before administration, and treatment of TB should begin before administration of Hadlima.
Patients taking Hadlima should not take a live vaccine.
The most common adverse effects (incidence > 10%) include infections (for example, upper respiratory infections, sinusitis), injection site reactions, headache, and rash.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration today approved a citrate-free, high-concentration formulation of adalimumab-bwwd (Hadlima), the manufacturer, Samsung Bioepis, and its commercialization partner Organon said in an announcement.
Hadlima is a biosimilar of the tumor necrosis factor inhibitor reference product adalimumab (Humira).
Hadlima was first approved in July 2019 in a citrated, 50-mg/mL formulation. The new citrate-free, 100-mg/mL version will be available in prefilled syringe and autoinjector options.
The 100-mg/mL formulation is indicated for the same seven conditions as its 50-mg/mL counterpart: rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, adult and pediatric Crohn’s disease, and ulcerative colitis.
The approval was based on clinical data from a randomized, single-blind, two-arm, parallel group, single-dose study that compared the pharmacokinetics, safety, tolerability, and immunogenicity of the 100-mg/mL and 50-mg/mL formulations of Hadlima in healthy volunteers.
Both low- and high-concentration formulations of Humira are currently marketed in the United States. Organon said that it expects to market Hadlima in the United States on or after July 1, 2023, in accordance with a licensing agreement with AbbVie.
The prescribing information for Hadlima includes specific warnings and areas of concern. The drug should not be administered to individuals who are known to be hypersensitive to adalimumab. The drug may lower the ability of the immune system to fight infections and may increase risk of infections, including serious infections leading to hospitalization or death, such as tuberculosis, bacterial sepsis, invasive fungal infections (such as histoplasmosis), and infections attributable to other opportunistic pathogens.
A test for latent TB infection should be given before administration, and treatment of TB should begin before administration of Hadlima.
Patients taking Hadlima should not take a live vaccine.
The most common adverse effects (incidence > 10%) include infections (for example, upper respiratory infections, sinusitis), injection site reactions, headache, and rash.
A version of this article first appeared on Medscape.com.
Sun Protection Factor Testing: A Call for an In Vitro Method
The sun protection factor (SPF) value indicates to consumers the level of protection that a given sunscreen formulation provides against erythemally effective UV radiation (UVR). 1 In vivo SPF testing, the gold standard for determining SPF, yields highly variable results and can harm human test participants. 2 In vitro SPF testing methodologies have been under development for years but none have (yet) replaced the in vivo test required by national and international regulatory agencies.
Recent European studies have shown strong data to support a highly standardized in vitro method,1 now under development by the International Organization for Standardization (ISO)—potentially to serve as a new SPF determination standard.1,3 Academia and industry should follow this example and actively take steps to develop and validate a suitable replacement for in vivo SPF testing.
In Vivo SPF Testing
The in vivo SPF test involves comparing doses of UVR necessary to induce erythema in human participants with and without sunscreen applied.2 Although this method has long been the standard for SPF determination, it is associated with the following major disadvantages:
- Cost: The in vivo test is expensive.
- Variability: Results of the test are subject to high interlaboratory variability due to the inherent subjectivity of identifying erythema, the variable skin types of human participants, and other laboratory-dependent factors.2 A study found that the average coefficient of variation for SPF values obtained from 3 or 4 laboratories to be 20%—with values exceeding 50% in some cases. With that level of variability, the same sunscreen may be labeled SPF 30, SPF 50, or SPF 50+, thereby posing a health risk to consumers who rely on the accuracy of such claims. In fact, Miksa et al2 concluded that “the largest obstacle to a reliable SPF assessment for consumer health is the in vivo SPF test itself.”
- Ethical concerns: Human participants are intentionally exposed to harmful UVR until sunburn is achieved. For that reason, there have been calls to abandon the practice of in vivo testing.1
Alternatives to In Vivo SPF Testing
There has been international interest in developing in silico and in vitro alternatives to the in vivo SPF test. These options are attractive because they are relatively inexpensive; avoid exposing human participants to harmful UVR; and have the potential to be more accurate and more reproducible than in vivo tests.
In Vitro Protocols—Many such in vitro tests exist; all generally involve applying a layer of sunscreen to an artificial substrate, exposing it to UVR from a solar simulator, and measuring the UVR transmittance through the product and film by spectrophotometry.1 Prior shortcomings of this method have included suboptimal reproducibility, lack of data on substrate and product properties, and lack of demonstrated equivalency to in vivo SPF testing.4
In Silico Protocols—These tests use data on the UV spectra of sunscreen filters, physical characteristics of sunscreen films on skin, and the unique photoinstability of filters to calculate expected UVR transmittance and SPF of sunscreens based on their ingredients.5 Reports have shown high correlation with in vivo values. Results are not subject to random error; reproducibility is theoretically perfect.5
Regulatory Agencies and In Vitro Testing
In the United States, sunscreens are regulated as over-the-counter drugs. In vivo testing is the only US Food and Drug Administration (FDA)–approved method for determining SPF for labeling purposes.1 In a 2007 Proposed Rule and a 2011 Final Rule, the FDA stated that in vitro SPF tests were an inadequate alternative to in vivo tests because of their shortcomings.4,6
Acknowledging the potential benefits of in vitro testing, the FDA wrote that it would consider in vitro alternatives if equivalency to the in vivo test could be proved.6 The agency has not published an official stance on in vitro SPF testing since those statements in 2007 and 2011. Of note, the FDA deems in vitro testing sufficient for making claims of broad-spectrum coverage.4
In contrast to the regulatory scenario in the United States, Europe regulates sunscreens as cosmetics, and the European Union (EU) has banned animal testing of cosmetics,7 which poses a problem for the development of new sunscreens. It is not surprising, therefore, that in 2006 the European Commission (the executive arm of the EU) published a mandate that in vitro SPF testing methods be actively developed due to ethical concerns associated with in vivo methods.8 In 2017, the International Organization for Standardization released specific validation criteria for proposed in vitro tests to facilitate the eventual approval of such methods.1
Progress of In Vitro Methods
In recent years, advances in in vitro SPF testing methods have addressed shortcomings noted previously by the FDA, which has led to notably improved reproducibility of results and correlation with in vivo values, in large part due to strict standardization of protocols,1 such as tight temperature control of samples, a multisubstrate approach, robotic product application to ensure even distribution, and pre-irradiation of sunscreen samples.
With these improvements, a 2018 study demonstrated an in vitro SPF testing methodology that exceeded published ISO validation criteria for emulsion-type products.1 This method was found to have low interlaboratory variability and high correlation with in vivo SPF values (Pearson r=0.88). Importantly, the authors noted that the consistency and reliability of in vitro SPF testing requires broad institution of a single unified method.1
The method described in the 2018 study1 has been accepted by the ISO Technical Committee and is undergoing further development3
Final Thoughts and Future Steps
Recent data confirm the potential viability of in vitro testing as a primary method of determining SPF values.1 Although ISO has moved forward with development of this method, the FDA has been quiet on in vitro SPF testing since 2011.4 The agency has, however, acknowledged the disadvantages of in vivo broad-spectrum testing, including exposure of human participants to harmful UVR and poor interlaboratory reproducibility.6
Given the technical developments and substantial potential benefits of in vitro testing, we believe that it is time for the FDA to revisit this matter. We propose that the FDA take 2 steps toward in vitro testing. First, publish specific validation criteria that would be deemed necessary for approval of such a test, similar to what ISO published in 2017. Second, thoroughly assess new data supporting the viability of available in vitro testing to determine if the FDA’s stated position that in vitro testing is inadequate remains true.
Although these 2 steps will be important to the process, adoption of an in vitro standard will require more than statements from the FDA. Additional funding should be allocated to researchers who are studying in vitro methodologies, and companies that profit from the multibillion-dollar sunscreen industry should be encouraged to invest in the development of more accurate and more ethical alternatives to in vivo SPF testing.
In vitro SPF testing is inexpensive, avoids the moral quandary of intentionally sunburning human participants, and is more reliable than in vivo testing. It is time for the FDA to facilitate the efforts of academia and industry in taking concrete steps toward approval of an in vitro alternative to in vivo SPF testing.
- Pissavini M, Tricaud C, Wiener G, et al. Validation of an in vitro sun protection factor (SPF) method in blinded ring-testing. Int J Cosmet Sci. 2018;40:263-268. doi:10.1111/ics.12459
- Miksa S, Lutz D, Guy C, et al. Sunscreen sun protection factor claim based on in vivo interlaboratory variability. Int J Cosmet Sci. 2016;38:541-549. doi:10.1111/ics.12333
- ISO/CD 23675: Cosmetics—sun protection test methods—in vitro determination of sun protection factor. International Organization for Standardization (ISO). July 25, 2020. Accessed May 17, 2022. https://www.iso.org/standard/76616.html
- US Food and Drug Administration. Labeling and effectiveness testing; sunscreen drug products for over-the-counter human use. Fed Regist. 2011;76(117):35620-35665. Accessed August 9, 2022. https://www.govinfo.gov/content/pkg/FR-2011-06-17/pdf/2011-14766.pdf
- Herzog B, Osterwalder U. Simulation of sunscreen performance. Pure Appl Chem. 2015;87:937-951. doi:10.1515/pac-2015-0401
- US Food and Drug Administration. Sunscreen drug products for over-the-counter human use; proposed amendment of final monograph. Fed Regist. 2007;72(165):49070-49122. Published August 27, 2007. Accessed August 9, 2022. https://www.govinfo.gov/content/pkg/FR-2007-08-27/pdf/07-4131.pdf
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products. November 30, 2009. Accessed August 10, 2022. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02009R1223-20190813
- European Commission Recommendation 2006/647/EC. Published September 22, 2006. Accessed August 10, 2022. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32006H0647
The sun protection factor (SPF) value indicates to consumers the level of protection that a given sunscreen formulation provides against erythemally effective UV radiation (UVR). 1 In vivo SPF testing, the gold standard for determining SPF, yields highly variable results and can harm human test participants. 2 In vitro SPF testing methodologies have been under development for years but none have (yet) replaced the in vivo test required by national and international regulatory agencies.
Recent European studies have shown strong data to support a highly standardized in vitro method,1 now under development by the International Organization for Standardization (ISO)—potentially to serve as a new SPF determination standard.1,3 Academia and industry should follow this example and actively take steps to develop and validate a suitable replacement for in vivo SPF testing.
In Vivo SPF Testing
The in vivo SPF test involves comparing doses of UVR necessary to induce erythema in human participants with and without sunscreen applied.2 Although this method has long been the standard for SPF determination, it is associated with the following major disadvantages:
- Cost: The in vivo test is expensive.
- Variability: Results of the test are subject to high interlaboratory variability due to the inherent subjectivity of identifying erythema, the variable skin types of human participants, and other laboratory-dependent factors.2 A study found that the average coefficient of variation for SPF values obtained from 3 or 4 laboratories to be 20%—with values exceeding 50% in some cases. With that level of variability, the same sunscreen may be labeled SPF 30, SPF 50, or SPF 50+, thereby posing a health risk to consumers who rely on the accuracy of such claims. In fact, Miksa et al2 concluded that “the largest obstacle to a reliable SPF assessment for consumer health is the in vivo SPF test itself.”
- Ethical concerns: Human participants are intentionally exposed to harmful UVR until sunburn is achieved. For that reason, there have been calls to abandon the practice of in vivo testing.1
Alternatives to In Vivo SPF Testing
There has been international interest in developing in silico and in vitro alternatives to the in vivo SPF test. These options are attractive because they are relatively inexpensive; avoid exposing human participants to harmful UVR; and have the potential to be more accurate and more reproducible than in vivo tests.
In Vitro Protocols—Many such in vitro tests exist; all generally involve applying a layer of sunscreen to an artificial substrate, exposing it to UVR from a solar simulator, and measuring the UVR transmittance through the product and film by spectrophotometry.1 Prior shortcomings of this method have included suboptimal reproducibility, lack of data on substrate and product properties, and lack of demonstrated equivalency to in vivo SPF testing.4
In Silico Protocols—These tests use data on the UV spectra of sunscreen filters, physical characteristics of sunscreen films on skin, and the unique photoinstability of filters to calculate expected UVR transmittance and SPF of sunscreens based on their ingredients.5 Reports have shown high correlation with in vivo values. Results are not subject to random error; reproducibility is theoretically perfect.5
Regulatory Agencies and In Vitro Testing
In the United States, sunscreens are regulated as over-the-counter drugs. In vivo testing is the only US Food and Drug Administration (FDA)–approved method for determining SPF for labeling purposes.1 In a 2007 Proposed Rule and a 2011 Final Rule, the FDA stated that in vitro SPF tests were an inadequate alternative to in vivo tests because of their shortcomings.4,6
Acknowledging the potential benefits of in vitro testing, the FDA wrote that it would consider in vitro alternatives if equivalency to the in vivo test could be proved.6 The agency has not published an official stance on in vitro SPF testing since those statements in 2007 and 2011. Of note, the FDA deems in vitro testing sufficient for making claims of broad-spectrum coverage.4
In contrast to the regulatory scenario in the United States, Europe regulates sunscreens as cosmetics, and the European Union (EU) has banned animal testing of cosmetics,7 which poses a problem for the development of new sunscreens. It is not surprising, therefore, that in 2006 the European Commission (the executive arm of the EU) published a mandate that in vitro SPF testing methods be actively developed due to ethical concerns associated with in vivo methods.8 In 2017, the International Organization for Standardization released specific validation criteria for proposed in vitro tests to facilitate the eventual approval of such methods.1
Progress of In Vitro Methods
In recent years, advances in in vitro SPF testing methods have addressed shortcomings noted previously by the FDA, which has led to notably improved reproducibility of results and correlation with in vivo values, in large part due to strict standardization of protocols,1 such as tight temperature control of samples, a multisubstrate approach, robotic product application to ensure even distribution, and pre-irradiation of sunscreen samples.
With these improvements, a 2018 study demonstrated an in vitro SPF testing methodology that exceeded published ISO validation criteria for emulsion-type products.1 This method was found to have low interlaboratory variability and high correlation with in vivo SPF values (Pearson r=0.88). Importantly, the authors noted that the consistency and reliability of in vitro SPF testing requires broad institution of a single unified method.1
The method described in the 2018 study1 has been accepted by the ISO Technical Committee and is undergoing further development3
Final Thoughts and Future Steps
Recent data confirm the potential viability of in vitro testing as a primary method of determining SPF values.1 Although ISO has moved forward with development of this method, the FDA has been quiet on in vitro SPF testing since 2011.4 The agency has, however, acknowledged the disadvantages of in vivo broad-spectrum testing, including exposure of human participants to harmful UVR and poor interlaboratory reproducibility.6
Given the technical developments and substantial potential benefits of in vitro testing, we believe that it is time for the FDA to revisit this matter. We propose that the FDA take 2 steps toward in vitro testing. First, publish specific validation criteria that would be deemed necessary for approval of such a test, similar to what ISO published in 2017. Second, thoroughly assess new data supporting the viability of available in vitro testing to determine if the FDA’s stated position that in vitro testing is inadequate remains true.
Although these 2 steps will be important to the process, adoption of an in vitro standard will require more than statements from the FDA. Additional funding should be allocated to researchers who are studying in vitro methodologies, and companies that profit from the multibillion-dollar sunscreen industry should be encouraged to invest in the development of more accurate and more ethical alternatives to in vivo SPF testing.
In vitro SPF testing is inexpensive, avoids the moral quandary of intentionally sunburning human participants, and is more reliable than in vivo testing. It is time for the FDA to facilitate the efforts of academia and industry in taking concrete steps toward approval of an in vitro alternative to in vivo SPF testing.
The sun protection factor (SPF) value indicates to consumers the level of protection that a given sunscreen formulation provides against erythemally effective UV radiation (UVR). 1 In vivo SPF testing, the gold standard for determining SPF, yields highly variable results and can harm human test participants. 2 In vitro SPF testing methodologies have been under development for years but none have (yet) replaced the in vivo test required by national and international regulatory agencies.
Recent European studies have shown strong data to support a highly standardized in vitro method,1 now under development by the International Organization for Standardization (ISO)—potentially to serve as a new SPF determination standard.1,3 Academia and industry should follow this example and actively take steps to develop and validate a suitable replacement for in vivo SPF testing.
In Vivo SPF Testing
The in vivo SPF test involves comparing doses of UVR necessary to induce erythema in human participants with and without sunscreen applied.2 Although this method has long been the standard for SPF determination, it is associated with the following major disadvantages:
- Cost: The in vivo test is expensive.
- Variability: Results of the test are subject to high interlaboratory variability due to the inherent subjectivity of identifying erythema, the variable skin types of human participants, and other laboratory-dependent factors.2 A study found that the average coefficient of variation for SPF values obtained from 3 or 4 laboratories to be 20%—with values exceeding 50% in some cases. With that level of variability, the same sunscreen may be labeled SPF 30, SPF 50, or SPF 50+, thereby posing a health risk to consumers who rely on the accuracy of such claims. In fact, Miksa et al2 concluded that “the largest obstacle to a reliable SPF assessment for consumer health is the in vivo SPF test itself.”
- Ethical concerns: Human participants are intentionally exposed to harmful UVR until sunburn is achieved. For that reason, there have been calls to abandon the practice of in vivo testing.1
Alternatives to In Vivo SPF Testing
There has been international interest in developing in silico and in vitro alternatives to the in vivo SPF test. These options are attractive because they are relatively inexpensive; avoid exposing human participants to harmful UVR; and have the potential to be more accurate and more reproducible than in vivo tests.
In Vitro Protocols—Many such in vitro tests exist; all generally involve applying a layer of sunscreen to an artificial substrate, exposing it to UVR from a solar simulator, and measuring the UVR transmittance through the product and film by spectrophotometry.1 Prior shortcomings of this method have included suboptimal reproducibility, lack of data on substrate and product properties, and lack of demonstrated equivalency to in vivo SPF testing.4
In Silico Protocols—These tests use data on the UV spectra of sunscreen filters, physical characteristics of sunscreen films on skin, and the unique photoinstability of filters to calculate expected UVR transmittance and SPF of sunscreens based on their ingredients.5 Reports have shown high correlation with in vivo values. Results are not subject to random error; reproducibility is theoretically perfect.5
Regulatory Agencies and In Vitro Testing
In the United States, sunscreens are regulated as over-the-counter drugs. In vivo testing is the only US Food and Drug Administration (FDA)–approved method for determining SPF for labeling purposes.1 In a 2007 Proposed Rule and a 2011 Final Rule, the FDA stated that in vitro SPF tests were an inadequate alternative to in vivo tests because of their shortcomings.4,6
Acknowledging the potential benefits of in vitro testing, the FDA wrote that it would consider in vitro alternatives if equivalency to the in vivo test could be proved.6 The agency has not published an official stance on in vitro SPF testing since those statements in 2007 and 2011. Of note, the FDA deems in vitro testing sufficient for making claims of broad-spectrum coverage.4
In contrast to the regulatory scenario in the United States, Europe regulates sunscreens as cosmetics, and the European Union (EU) has banned animal testing of cosmetics,7 which poses a problem for the development of new sunscreens. It is not surprising, therefore, that in 2006 the European Commission (the executive arm of the EU) published a mandate that in vitro SPF testing methods be actively developed due to ethical concerns associated with in vivo methods.8 In 2017, the International Organization for Standardization released specific validation criteria for proposed in vitro tests to facilitate the eventual approval of such methods.1
Progress of In Vitro Methods
In recent years, advances in in vitro SPF testing methods have addressed shortcomings noted previously by the FDA, which has led to notably improved reproducibility of results and correlation with in vivo values, in large part due to strict standardization of protocols,1 such as tight temperature control of samples, a multisubstrate approach, robotic product application to ensure even distribution, and pre-irradiation of sunscreen samples.
With these improvements, a 2018 study demonstrated an in vitro SPF testing methodology that exceeded published ISO validation criteria for emulsion-type products.1 This method was found to have low interlaboratory variability and high correlation with in vivo SPF values (Pearson r=0.88). Importantly, the authors noted that the consistency and reliability of in vitro SPF testing requires broad institution of a single unified method.1
The method described in the 2018 study1 has been accepted by the ISO Technical Committee and is undergoing further development3
Final Thoughts and Future Steps
Recent data confirm the potential viability of in vitro testing as a primary method of determining SPF values.1 Although ISO has moved forward with development of this method, the FDA has been quiet on in vitro SPF testing since 2011.4 The agency has, however, acknowledged the disadvantages of in vivo broad-spectrum testing, including exposure of human participants to harmful UVR and poor interlaboratory reproducibility.6
Given the technical developments and substantial potential benefits of in vitro testing, we believe that it is time for the FDA to revisit this matter. We propose that the FDA take 2 steps toward in vitro testing. First, publish specific validation criteria that would be deemed necessary for approval of such a test, similar to what ISO published in 2017. Second, thoroughly assess new data supporting the viability of available in vitro testing to determine if the FDA’s stated position that in vitro testing is inadequate remains true.
Although these 2 steps will be important to the process, adoption of an in vitro standard will require more than statements from the FDA. Additional funding should be allocated to researchers who are studying in vitro methodologies, and companies that profit from the multibillion-dollar sunscreen industry should be encouraged to invest in the development of more accurate and more ethical alternatives to in vivo SPF testing.
In vitro SPF testing is inexpensive, avoids the moral quandary of intentionally sunburning human participants, and is more reliable than in vivo testing. It is time for the FDA to facilitate the efforts of academia and industry in taking concrete steps toward approval of an in vitro alternative to in vivo SPF testing.
- Pissavini M, Tricaud C, Wiener G, et al. Validation of an in vitro sun protection factor (SPF) method in blinded ring-testing. Int J Cosmet Sci. 2018;40:263-268. doi:10.1111/ics.12459
- Miksa S, Lutz D, Guy C, et al. Sunscreen sun protection factor claim based on in vivo interlaboratory variability. Int J Cosmet Sci. 2016;38:541-549. doi:10.1111/ics.12333
- ISO/CD 23675: Cosmetics—sun protection test methods—in vitro determination of sun protection factor. International Organization for Standardization (ISO). July 25, 2020. Accessed May 17, 2022. https://www.iso.org/standard/76616.html
- US Food and Drug Administration. Labeling and effectiveness testing; sunscreen drug products for over-the-counter human use. Fed Regist. 2011;76(117):35620-35665. Accessed August 9, 2022. https://www.govinfo.gov/content/pkg/FR-2011-06-17/pdf/2011-14766.pdf
- Herzog B, Osterwalder U. Simulation of sunscreen performance. Pure Appl Chem. 2015;87:937-951. doi:10.1515/pac-2015-0401
- US Food and Drug Administration. Sunscreen drug products for over-the-counter human use; proposed amendment of final monograph. Fed Regist. 2007;72(165):49070-49122. Published August 27, 2007. Accessed August 9, 2022. https://www.govinfo.gov/content/pkg/FR-2007-08-27/pdf/07-4131.pdf
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products. November 30, 2009. Accessed August 10, 2022. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02009R1223-20190813
- European Commission Recommendation 2006/647/EC. Published September 22, 2006. Accessed August 10, 2022. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32006H0647
- Pissavini M, Tricaud C, Wiener G, et al. Validation of an in vitro sun protection factor (SPF) method in blinded ring-testing. Int J Cosmet Sci. 2018;40:263-268. doi:10.1111/ics.12459
- Miksa S, Lutz D, Guy C, et al. Sunscreen sun protection factor claim based on in vivo interlaboratory variability. Int J Cosmet Sci. 2016;38:541-549. doi:10.1111/ics.12333
- ISO/CD 23675: Cosmetics—sun protection test methods—in vitro determination of sun protection factor. International Organization for Standardization (ISO). July 25, 2020. Accessed May 17, 2022. https://www.iso.org/standard/76616.html
- US Food and Drug Administration. Labeling and effectiveness testing; sunscreen drug products for over-the-counter human use. Fed Regist. 2011;76(117):35620-35665. Accessed August 9, 2022. https://www.govinfo.gov/content/pkg/FR-2011-06-17/pdf/2011-14766.pdf
- Herzog B, Osterwalder U. Simulation of sunscreen performance. Pure Appl Chem. 2015;87:937-951. doi:10.1515/pac-2015-0401
- US Food and Drug Administration. Sunscreen drug products for over-the-counter human use; proposed amendment of final monograph. Fed Regist. 2007;72(165):49070-49122. Published August 27, 2007. Accessed August 9, 2022. https://www.govinfo.gov/content/pkg/FR-2007-08-27/pdf/07-4131.pdf
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products. November 30, 2009. Accessed August 10, 2022. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02009R1223-20190813
- European Commission Recommendation 2006/647/EC. Published September 22, 2006. Accessed August 10, 2022. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32006H0647
Practice Points
- The methodology for determining sun protection factor (SPF) that currently is accepted by the US Food and Drug Administration is an expensive and imprecise in vivo test that exposes human participants to harmful UV radiation.
- In vitro tests for determining SPF may be viable alternatives to the current in vivo gold standard.
- Researchers and the sunscreen industry should actively develop these in vitro methodologies to adopt a more accurate and less harmful test for SPF.
Intralesional Human Papillomavirus Vaccine Therapy for Recalcitrant Plantar Wart Triggers Gout Flare
To the Editor:
There is increasing evidence supporting the use of the human papillomavirus (HPV) vaccine in the treatment of recalcitrant common warts.1 We describe a potential complication associated with HPV vaccine treatment of warts that would be of interest to dermatologists.
A 70-year-old woman presented with a plantar wart measuring 6 mm in diameter at the base of the right hallux of 5 years’ duration. Prior failed therapies for wart removal included multiple paring treatments, cryotherapy, and topical salicylic acid 40% to 60%. The patient had no notable comorbidities; no history of gout; and no known risk factors for gout, such as hypertension, renal insufficiency, diuretic use, obesity, family history, or trauma.
Prior reports cited effective treatment of recalcitrant warts with recombinant HPV vaccines, both intralesionally1 and intramuscularly.2,3 With this knowledge in mind, we administered an intralesional injection with 0.1-mL recombinant HPV 9-valent vaccine to the patient’s plantar wart. Gradual erythema and swelling of the right first metatarsophalangeal joint developed over the next 7 days. Synovial fluid analysis demonstrated negatively birefringent crystals. The patient commenced treatment with colchicine and indomethacin and improved over the next 5 days. The wart resolved 3 months later and required no further treatment.
Prophylactic quadrivalent HPV vaccines have shown efficacy in treating HPV-associated precancerous and cancerous lesions.4 Case reports have suggested that HPV vaccines may be an effective treatment option for recalcitrant warts,1-3,5 especially in cases that do not respond to traditional treatment. It is possible that the mechanism of wart treatment involves overlap in the antigenic epitopes of the HPV types targeted by the vaccine vs the HPV types responsible for causing warts.2 Papillomaviruslike particles, based on the L1 capsid protein, can induce a specific CD8+ activation signal, leading to a vaccine-induced cytotoxic T-cell response that targets the wart cells with HPV-like antigens.6 The HPV vaccine contains aluminium, which has been shown to activate NLRP3 inflammasome,5 which may trigger gout by increasing monosodium urate crystal deposition via IL-1β production.7 This may lead to an increased risk for gout flares, an adverse effect of the HPV vaccine. This finding is supported by other studies of aluminium-containing vaccines that show an association with gout.6 It is noted that these vaccines are mostly delivered intramuscularly or subcutaneously in some cases.
We reported a case of gout triggered by intralesional HPV vaccine treatment of warts. It is unclear whether the gout was induced by the vaccine itself or whether it was due to trauma caused by the intralesional injection near the joint space. Based on our findings, we recommend that patients receiving intralesional injections for wart treatment be advised of this potential adverse effect, especially if they have risk factors for gout or have a history of gout.
- Nofal A, Marei A, Ibrahim AM et al. Intralesional versus intramuscular bivalent human papillomavirus vaccine in the treatment of recalcitrant common warts. J Am Acad Dermatol. 2020;82:94-100.
- Venugopal SS, Murrell DF. Recalcitrant cutaneous warts treated with recombinant quadrivalent human papillomavirus vaccine (types 6, 11, 16, and 18) in a developmentally delayed, 31-year-old white man. Arch Dermatol. 2010;146:475-477.
- Daniel BS, Murrell DF. Complete resolution of chronic multiple verruca vulgaris treated with quadrivalent human papillomavirus vaccine. JAMA Dermatol. 2013;149:370-372.
- Kenter GG, Welters MJ, Valentijn AR, et al. Vaccination against HPV-16 oncoproteins for vulvar intraepithelial neoplasia. N Engl J Med. 2009;361:1838-1847.
- Eisenbarth SC, Colegio OR, O’Connor W, et al. Crucial role for the NALP3 inflammasome in the immunostimulatory properties of aluminium adjuvants. Nature. 2008;453:1122-1166.
- Bellone S, El-Sahwi K, Cocco E, et al. Human papillomavirus type 16 (HPV-16) virus-like particle L1-specific CD8+ cytotoxic T lymphocytes (CTLs) are equally effective as E7-specific CD8+ CTLs in killing autologous HPV-16-positive tumor cells in cervical cancer patients: implications for L1 dendritic cell-based therapeutic vaccines. J Virol. 2009;83:6779-6789.
- Yokose C, McCormick N, Chen C, et al. Risk of gout flares after vaccination: a prospective case cross-over study. Ann Rheum Dis. 2019;78:1601-1604.
To the Editor:
There is increasing evidence supporting the use of the human papillomavirus (HPV) vaccine in the treatment of recalcitrant common warts.1 We describe a potential complication associated with HPV vaccine treatment of warts that would be of interest to dermatologists.
A 70-year-old woman presented with a plantar wart measuring 6 mm in diameter at the base of the right hallux of 5 years’ duration. Prior failed therapies for wart removal included multiple paring treatments, cryotherapy, and topical salicylic acid 40% to 60%. The patient had no notable comorbidities; no history of gout; and no known risk factors for gout, such as hypertension, renal insufficiency, diuretic use, obesity, family history, or trauma.
Prior reports cited effective treatment of recalcitrant warts with recombinant HPV vaccines, both intralesionally1 and intramuscularly.2,3 With this knowledge in mind, we administered an intralesional injection with 0.1-mL recombinant HPV 9-valent vaccine to the patient’s plantar wart. Gradual erythema and swelling of the right first metatarsophalangeal joint developed over the next 7 days. Synovial fluid analysis demonstrated negatively birefringent crystals. The patient commenced treatment with colchicine and indomethacin and improved over the next 5 days. The wart resolved 3 months later and required no further treatment.
Prophylactic quadrivalent HPV vaccines have shown efficacy in treating HPV-associated precancerous and cancerous lesions.4 Case reports have suggested that HPV vaccines may be an effective treatment option for recalcitrant warts,1-3,5 especially in cases that do not respond to traditional treatment. It is possible that the mechanism of wart treatment involves overlap in the antigenic epitopes of the HPV types targeted by the vaccine vs the HPV types responsible for causing warts.2 Papillomaviruslike particles, based on the L1 capsid protein, can induce a specific CD8+ activation signal, leading to a vaccine-induced cytotoxic T-cell response that targets the wart cells with HPV-like antigens.6 The HPV vaccine contains aluminium, which has been shown to activate NLRP3 inflammasome,5 which may trigger gout by increasing monosodium urate crystal deposition via IL-1β production.7 This may lead to an increased risk for gout flares, an adverse effect of the HPV vaccine. This finding is supported by other studies of aluminium-containing vaccines that show an association with gout.6 It is noted that these vaccines are mostly delivered intramuscularly or subcutaneously in some cases.
We reported a case of gout triggered by intralesional HPV vaccine treatment of warts. It is unclear whether the gout was induced by the vaccine itself or whether it was due to trauma caused by the intralesional injection near the joint space. Based on our findings, we recommend that patients receiving intralesional injections for wart treatment be advised of this potential adverse effect, especially if they have risk factors for gout or have a history of gout.
To the Editor:
There is increasing evidence supporting the use of the human papillomavirus (HPV) vaccine in the treatment of recalcitrant common warts.1 We describe a potential complication associated with HPV vaccine treatment of warts that would be of interest to dermatologists.
A 70-year-old woman presented with a plantar wart measuring 6 mm in diameter at the base of the right hallux of 5 years’ duration. Prior failed therapies for wart removal included multiple paring treatments, cryotherapy, and topical salicylic acid 40% to 60%. The patient had no notable comorbidities; no history of gout; and no known risk factors for gout, such as hypertension, renal insufficiency, diuretic use, obesity, family history, or trauma.
Prior reports cited effective treatment of recalcitrant warts with recombinant HPV vaccines, both intralesionally1 and intramuscularly.2,3 With this knowledge in mind, we administered an intralesional injection with 0.1-mL recombinant HPV 9-valent vaccine to the patient’s plantar wart. Gradual erythema and swelling of the right first metatarsophalangeal joint developed over the next 7 days. Synovial fluid analysis demonstrated negatively birefringent crystals. The patient commenced treatment with colchicine and indomethacin and improved over the next 5 days. The wart resolved 3 months later and required no further treatment.
Prophylactic quadrivalent HPV vaccines have shown efficacy in treating HPV-associated precancerous and cancerous lesions.4 Case reports have suggested that HPV vaccines may be an effective treatment option for recalcitrant warts,1-3,5 especially in cases that do not respond to traditional treatment. It is possible that the mechanism of wart treatment involves overlap in the antigenic epitopes of the HPV types targeted by the vaccine vs the HPV types responsible for causing warts.2 Papillomaviruslike particles, based on the L1 capsid protein, can induce a specific CD8+ activation signal, leading to a vaccine-induced cytotoxic T-cell response that targets the wart cells with HPV-like antigens.6 The HPV vaccine contains aluminium, which has been shown to activate NLRP3 inflammasome,5 which may trigger gout by increasing monosodium urate crystal deposition via IL-1β production.7 This may lead to an increased risk for gout flares, an adverse effect of the HPV vaccine. This finding is supported by other studies of aluminium-containing vaccines that show an association with gout.6 It is noted that these vaccines are mostly delivered intramuscularly or subcutaneously in some cases.
We reported a case of gout triggered by intralesional HPV vaccine treatment of warts. It is unclear whether the gout was induced by the vaccine itself or whether it was due to trauma caused by the intralesional injection near the joint space. Based on our findings, we recommend that patients receiving intralesional injections for wart treatment be advised of this potential adverse effect, especially if they have risk factors for gout or have a history of gout.
- Nofal A, Marei A, Ibrahim AM et al. Intralesional versus intramuscular bivalent human papillomavirus vaccine in the treatment of recalcitrant common warts. J Am Acad Dermatol. 2020;82:94-100.
- Venugopal SS, Murrell DF. Recalcitrant cutaneous warts treated with recombinant quadrivalent human papillomavirus vaccine (types 6, 11, 16, and 18) in a developmentally delayed, 31-year-old white man. Arch Dermatol. 2010;146:475-477.
- Daniel BS, Murrell DF. Complete resolution of chronic multiple verruca vulgaris treated with quadrivalent human papillomavirus vaccine. JAMA Dermatol. 2013;149:370-372.
- Kenter GG, Welters MJ, Valentijn AR, et al. Vaccination against HPV-16 oncoproteins for vulvar intraepithelial neoplasia. N Engl J Med. 2009;361:1838-1847.
- Eisenbarth SC, Colegio OR, O’Connor W, et al. Crucial role for the NALP3 inflammasome in the immunostimulatory properties of aluminium adjuvants. Nature. 2008;453:1122-1166.
- Bellone S, El-Sahwi K, Cocco E, et al. Human papillomavirus type 16 (HPV-16) virus-like particle L1-specific CD8+ cytotoxic T lymphocytes (CTLs) are equally effective as E7-specific CD8+ CTLs in killing autologous HPV-16-positive tumor cells in cervical cancer patients: implications for L1 dendritic cell-based therapeutic vaccines. J Virol. 2009;83:6779-6789.
- Yokose C, McCormick N, Chen C, et al. Risk of gout flares after vaccination: a prospective case cross-over study. Ann Rheum Dis. 2019;78:1601-1604.
- Nofal A, Marei A, Ibrahim AM et al. Intralesional versus intramuscular bivalent human papillomavirus vaccine in the treatment of recalcitrant common warts. J Am Acad Dermatol. 2020;82:94-100.
- Venugopal SS, Murrell DF. Recalcitrant cutaneous warts treated with recombinant quadrivalent human papillomavirus vaccine (types 6, 11, 16, and 18) in a developmentally delayed, 31-year-old white man. Arch Dermatol. 2010;146:475-477.
- Daniel BS, Murrell DF. Complete resolution of chronic multiple verruca vulgaris treated with quadrivalent human papillomavirus vaccine. JAMA Dermatol. 2013;149:370-372.
- Kenter GG, Welters MJ, Valentijn AR, et al. Vaccination against HPV-16 oncoproteins for vulvar intraepithelial neoplasia. N Engl J Med. 2009;361:1838-1847.
- Eisenbarth SC, Colegio OR, O’Connor W, et al. Crucial role for the NALP3 inflammasome in the immunostimulatory properties of aluminium adjuvants. Nature. 2008;453:1122-1166.
- Bellone S, El-Sahwi K, Cocco E, et al. Human papillomavirus type 16 (HPV-16) virus-like particle L1-specific CD8+ cytotoxic T lymphocytes (CTLs) are equally effective as E7-specific CD8+ CTLs in killing autologous HPV-16-positive tumor cells in cervical cancer patients: implications for L1 dendritic cell-based therapeutic vaccines. J Virol. 2009;83:6779-6789.
- Yokose C, McCormick N, Chen C, et al. Risk of gout flares after vaccination: a prospective case cross-over study. Ann Rheum Dis. 2019;78:1601-1604.
Practice Points
- Human papillomavirus (HPV) vaccines are increasingly used for recalcitrant warts.
- We describe an unreported adverse effect of gout flare following HPV vaccine treatment of plantar wart.
Asking patients about their gender identity: ‘Normalize’ the discussion and other recommendations
PORTLAND, ORE. –
“From the patient perspective, there’s nothing more awkward than having an awkward provider asking awkward questions,” Howa Yeung, MD, MSc, assistant professor of dermatology at Emory University, Atlanta, said at the annual meeting of the Pacific Dermatologic Association.
In 2014, Sean Cahill, PhD, and Harvey Makadon, MD, published an article recommending the inclusion of sexual orientation and gender identity questions in electronic medical records, a practice that Dr. Yeung characterized as “the most patient-centered way to collect sexual orientation and gender identity information. The most important thing is to ask routinely on an intake form where they fill it out themselves. All electronic medical records have the capacity to do so.”
On the other hand, when asking new patients about their sexual orientation and gender identity in person, it’s important to normalize the discussion and ask in an inclusive way, said Dr. Yeung, who was the lead author on published recommendations on dermatologic care for LGBTQ persons published in the Journal of the American Academy of Dermatology. “For example, I always say, ‘I’m Howa Yeung. I use him pronouns,’ ” he said. “ ‘How should I address you?’ Then they will tell you. Allow people to lead the way.”
Other suggested tips in the JAAD article include to avoid using terms such as “sir” or “miss” until the patient’s gender identify is ascertained. Instead, use gender-neutral terms such as “they” or “the patient” when referring to new patients. Do not use the pronoun “it.” If a patient’s name does not match a name in the medical record, ask, “What is the name on your insurance/records?” and avoid assuming gender(s) of a patient’s partner or parents. Instead, consider asking, “Who did you bring with you today?” “Are you in a relationship?” “What are the names of your parents?”
Normalizing questions about the patient’s sexual history is also key. “I tell patients that I routinely ask about sexual history for patients with similar skin issues because it helps me provide the best care for them,” Dr. Yeung said. “I also discuss confidentiality and documentation.”
Dr. Yeung reported having no relevant disclosures.
PORTLAND, ORE. –
“From the patient perspective, there’s nothing more awkward than having an awkward provider asking awkward questions,” Howa Yeung, MD, MSc, assistant professor of dermatology at Emory University, Atlanta, said at the annual meeting of the Pacific Dermatologic Association.
In 2014, Sean Cahill, PhD, and Harvey Makadon, MD, published an article recommending the inclusion of sexual orientation and gender identity questions in electronic medical records, a practice that Dr. Yeung characterized as “the most patient-centered way to collect sexual orientation and gender identity information. The most important thing is to ask routinely on an intake form where they fill it out themselves. All electronic medical records have the capacity to do so.”
On the other hand, when asking new patients about their sexual orientation and gender identity in person, it’s important to normalize the discussion and ask in an inclusive way, said Dr. Yeung, who was the lead author on published recommendations on dermatologic care for LGBTQ persons published in the Journal of the American Academy of Dermatology. “For example, I always say, ‘I’m Howa Yeung. I use him pronouns,’ ” he said. “ ‘How should I address you?’ Then they will tell you. Allow people to lead the way.”
Other suggested tips in the JAAD article include to avoid using terms such as “sir” or “miss” until the patient’s gender identify is ascertained. Instead, use gender-neutral terms such as “they” or “the patient” when referring to new patients. Do not use the pronoun “it.” If a patient’s name does not match a name in the medical record, ask, “What is the name on your insurance/records?” and avoid assuming gender(s) of a patient’s partner or parents. Instead, consider asking, “Who did you bring with you today?” “Are you in a relationship?” “What are the names of your parents?”
Normalizing questions about the patient’s sexual history is also key. “I tell patients that I routinely ask about sexual history for patients with similar skin issues because it helps me provide the best care for them,” Dr. Yeung said. “I also discuss confidentiality and documentation.”
Dr. Yeung reported having no relevant disclosures.
PORTLAND, ORE. –
“From the patient perspective, there’s nothing more awkward than having an awkward provider asking awkward questions,” Howa Yeung, MD, MSc, assistant professor of dermatology at Emory University, Atlanta, said at the annual meeting of the Pacific Dermatologic Association.
In 2014, Sean Cahill, PhD, and Harvey Makadon, MD, published an article recommending the inclusion of sexual orientation and gender identity questions in electronic medical records, a practice that Dr. Yeung characterized as “the most patient-centered way to collect sexual orientation and gender identity information. The most important thing is to ask routinely on an intake form where they fill it out themselves. All electronic medical records have the capacity to do so.”
On the other hand, when asking new patients about their sexual orientation and gender identity in person, it’s important to normalize the discussion and ask in an inclusive way, said Dr. Yeung, who was the lead author on published recommendations on dermatologic care for LGBTQ persons published in the Journal of the American Academy of Dermatology. “For example, I always say, ‘I’m Howa Yeung. I use him pronouns,’ ” he said. “ ‘How should I address you?’ Then they will tell you. Allow people to lead the way.”
Other suggested tips in the JAAD article include to avoid using terms such as “sir” or “miss” until the patient’s gender identify is ascertained. Instead, use gender-neutral terms such as “they” or “the patient” when referring to new patients. Do not use the pronoun “it.” If a patient’s name does not match a name in the medical record, ask, “What is the name on your insurance/records?” and avoid assuming gender(s) of a patient’s partner or parents. Instead, consider asking, “Who did you bring with you today?” “Are you in a relationship?” “What are the names of your parents?”
Normalizing questions about the patient’s sexual history is also key. “I tell patients that I routinely ask about sexual history for patients with similar skin issues because it helps me provide the best care for them,” Dr. Yeung said. “I also discuss confidentiality and documentation.”
Dr. Yeung reported having no relevant disclosures.
AT PDA 2022
Doctors using fake positive reviews to boost business
Five years ago, Kay Dean relied upon Yelp! and Google reviews in her search for a doctor in her area. After finding a physician with fairly high reviews, Ms. Dean was shocked when her personal experience was significantly worse than patients on the review platforms.
Following her experience, Ms. Dean, a former federal government investigator, became skeptical and used her skills to investigate the practice on all review platforms. She uncovered that the practice had a review from an individual who was involved in a review trading group on Facebook, where organizations openly barter their services in exchange for positive reviews fraud.
“I discovered that the online review world was just saturated with fake reviews, much more so than I think most people are aware ... and law enforcement regulators aren’t doing anything to address the problem,” said Ms. Dean. “In this online space, it’s the Wild West; cheating is rewarded.”
Ms. Dean decided to take matters into her own hands. She created a YouTube channel called Fake Review Watch, where she exposes real businesses and their attempts to dupe potential consumers with fake positive reviews.
For example, one video analyzes an orthopedic surgeon in Manhattan with an abundance of five-star reviews. Through her detailed analysis, Ms. Dean created a spreadsheet of the 26 alleged patients of the orthopedic surgeon that had submitted glowing reviews. She looked into other businesses that the individuals had left reviews for and found a significant amount of overlap.
According to the video, 19 of the doctor’s reviewers had left high reviews for the same moving company in Las Vegas, and 18 of them reviewed the same locksmith in Texas. Overall, eight of the patients reviewed the same mover, locksmith, and hotel in New Zealand.
A matter of trust
Ms. Dean expressed the gravity of this phenomenon, especially in health care, as patients often head online first when searching for care options. Based on a survey by Software Advice, about 84% of patients use online reviews to assess a physician, and 77% use review sites as the first step in finding a doctor.
Patient trust has continued to diminish in recent years, particularly following the pandemic. In a 2021 global ranking of trust levels towards health care by country, the U.S. health care system ranked 19th, far below those of several developing countries.
Owing to the rise of fake patient reviews and their inscrutable nature, Ms. Dean advises staying away from online review platforms. Instead, she suggests sticking to the old-fashioned method of getting recommendations from friends and relatives, not virtual people.
Ms. Dean explained a few indicators that she looks for when trying to identify a fake review.
“The business has all five-star reviews, negative reviews are followed by five-star reviews, or the business has an abnormal number of positive reviews in a short period of time,” she noted. “Some businesses try to bury legitimate negative reviews by obtaining more recent, fake, positive ones. The recent reviews will contradict the specific criticisms in the negative review.”
She warned that consumers should not give credibility to reviews simply because the reviewer is dubbed “Elite” or a Google Local Guide, because she has seen plenty of these individuals posting fake reviews.
Unfortunately, review platforms haven’t been doing much self-policing. Google and Healthgrades have a series of policies against fake engagement, impersonation, misinformation, and misrepresentation, according to their websites. However, the only consequence of these violations is review removal.
Both Yelp! and Google say they have automated software that distinguishes real versus fake reviews. When Yelp! uncovers users engaging in compensation review activity, it removes their reviews, closes their account, and blocks those users from creating future Yelp! accounts.
Physicians’ basis
Moreover,
“I think there’s an erosion of business ethics because cheating is rewarded. You can’t compete in an environment where your competition is allowed to accumulate numerous fake reviews while you’re still trying to fill chairs in your business,” said Ms. Dean. “Your competition is then getting the business because the tech companies are allowing this fraud.”
Family physician and practice owner Mike Woo-Ming, MD, MPH, provides career coaching for physicians, including maintaining a good reputation – in-person and online. He has seen physicians bumping up their own five-star reviews personally as well as posting negative reviews for their competition.
“I’ve seen where they’re going to lose business, as many practices were affected through COVID,” he said. “Business owners can become desperate and may decide to start posting or buying reviews because they know people will choose certain services these days based upon reviews.”
Dr. Woo-Ming expressed his frustration with fellow physicians who give in to purchasing fake reviews, because the patients have no idea whether reviews are genuine or not.
To encourage genuine positive reviews, Dr. Woo-Ming’s practice uses a third-party app system that sends patients a follow-up email or text asking about their experience with a link to review sites.
“Honest reviews are a reflection of what I can do to improve my business. At the end of the day, if you’re truly providing great service and you’re helping people by providing great medical care, those are going to win out,” he said. “I would rather, as a responsible practice owner, improve the experience and outcome for the patient.”
A version of this article first appeared on Medscape.com.
Five years ago, Kay Dean relied upon Yelp! and Google reviews in her search for a doctor in her area. After finding a physician with fairly high reviews, Ms. Dean was shocked when her personal experience was significantly worse than patients on the review platforms.
Following her experience, Ms. Dean, a former federal government investigator, became skeptical and used her skills to investigate the practice on all review platforms. She uncovered that the practice had a review from an individual who was involved in a review trading group on Facebook, where organizations openly barter their services in exchange for positive reviews fraud.
“I discovered that the online review world was just saturated with fake reviews, much more so than I think most people are aware ... and law enforcement regulators aren’t doing anything to address the problem,” said Ms. Dean. “In this online space, it’s the Wild West; cheating is rewarded.”
Ms. Dean decided to take matters into her own hands. She created a YouTube channel called Fake Review Watch, where she exposes real businesses and their attempts to dupe potential consumers with fake positive reviews.
For example, one video analyzes an orthopedic surgeon in Manhattan with an abundance of five-star reviews. Through her detailed analysis, Ms. Dean created a spreadsheet of the 26 alleged patients of the orthopedic surgeon that had submitted glowing reviews. She looked into other businesses that the individuals had left reviews for and found a significant amount of overlap.
According to the video, 19 of the doctor’s reviewers had left high reviews for the same moving company in Las Vegas, and 18 of them reviewed the same locksmith in Texas. Overall, eight of the patients reviewed the same mover, locksmith, and hotel in New Zealand.
A matter of trust
Ms. Dean expressed the gravity of this phenomenon, especially in health care, as patients often head online first when searching for care options. Based on a survey by Software Advice, about 84% of patients use online reviews to assess a physician, and 77% use review sites as the first step in finding a doctor.
Patient trust has continued to diminish in recent years, particularly following the pandemic. In a 2021 global ranking of trust levels towards health care by country, the U.S. health care system ranked 19th, far below those of several developing countries.
Owing to the rise of fake patient reviews and their inscrutable nature, Ms. Dean advises staying away from online review platforms. Instead, she suggests sticking to the old-fashioned method of getting recommendations from friends and relatives, not virtual people.
Ms. Dean explained a few indicators that she looks for when trying to identify a fake review.
“The business has all five-star reviews, negative reviews are followed by five-star reviews, or the business has an abnormal number of positive reviews in a short period of time,” she noted. “Some businesses try to bury legitimate negative reviews by obtaining more recent, fake, positive ones. The recent reviews will contradict the specific criticisms in the negative review.”
She warned that consumers should not give credibility to reviews simply because the reviewer is dubbed “Elite” or a Google Local Guide, because she has seen plenty of these individuals posting fake reviews.
Unfortunately, review platforms haven’t been doing much self-policing. Google and Healthgrades have a series of policies against fake engagement, impersonation, misinformation, and misrepresentation, according to their websites. However, the only consequence of these violations is review removal.
Both Yelp! and Google say they have automated software that distinguishes real versus fake reviews. When Yelp! uncovers users engaging in compensation review activity, it removes their reviews, closes their account, and blocks those users from creating future Yelp! accounts.
Physicians’ basis
Moreover,
“I think there’s an erosion of business ethics because cheating is rewarded. You can’t compete in an environment where your competition is allowed to accumulate numerous fake reviews while you’re still trying to fill chairs in your business,” said Ms. Dean. “Your competition is then getting the business because the tech companies are allowing this fraud.”
Family physician and practice owner Mike Woo-Ming, MD, MPH, provides career coaching for physicians, including maintaining a good reputation – in-person and online. He has seen physicians bumping up their own five-star reviews personally as well as posting negative reviews for their competition.
“I’ve seen where they’re going to lose business, as many practices were affected through COVID,” he said. “Business owners can become desperate and may decide to start posting or buying reviews because they know people will choose certain services these days based upon reviews.”
Dr. Woo-Ming expressed his frustration with fellow physicians who give in to purchasing fake reviews, because the patients have no idea whether reviews are genuine or not.
To encourage genuine positive reviews, Dr. Woo-Ming’s practice uses a third-party app system that sends patients a follow-up email or text asking about their experience with a link to review sites.
“Honest reviews are a reflection of what I can do to improve my business. At the end of the day, if you’re truly providing great service and you’re helping people by providing great medical care, those are going to win out,” he said. “I would rather, as a responsible practice owner, improve the experience and outcome for the patient.”
A version of this article first appeared on Medscape.com.
Five years ago, Kay Dean relied upon Yelp! and Google reviews in her search for a doctor in her area. After finding a physician with fairly high reviews, Ms. Dean was shocked when her personal experience was significantly worse than patients on the review platforms.
Following her experience, Ms. Dean, a former federal government investigator, became skeptical and used her skills to investigate the practice on all review platforms. She uncovered that the practice had a review from an individual who was involved in a review trading group on Facebook, where organizations openly barter their services in exchange for positive reviews fraud.
“I discovered that the online review world was just saturated with fake reviews, much more so than I think most people are aware ... and law enforcement regulators aren’t doing anything to address the problem,” said Ms. Dean. “In this online space, it’s the Wild West; cheating is rewarded.”
Ms. Dean decided to take matters into her own hands. She created a YouTube channel called Fake Review Watch, where she exposes real businesses and their attempts to dupe potential consumers with fake positive reviews.
For example, one video analyzes an orthopedic surgeon in Manhattan with an abundance of five-star reviews. Through her detailed analysis, Ms. Dean created a spreadsheet of the 26 alleged patients of the orthopedic surgeon that had submitted glowing reviews. She looked into other businesses that the individuals had left reviews for and found a significant amount of overlap.
According to the video, 19 of the doctor’s reviewers had left high reviews for the same moving company in Las Vegas, and 18 of them reviewed the same locksmith in Texas. Overall, eight of the patients reviewed the same mover, locksmith, and hotel in New Zealand.
A matter of trust
Ms. Dean expressed the gravity of this phenomenon, especially in health care, as patients often head online first when searching for care options. Based on a survey by Software Advice, about 84% of patients use online reviews to assess a physician, and 77% use review sites as the first step in finding a doctor.
Patient trust has continued to diminish in recent years, particularly following the pandemic. In a 2021 global ranking of trust levels towards health care by country, the U.S. health care system ranked 19th, far below those of several developing countries.
Owing to the rise of fake patient reviews and their inscrutable nature, Ms. Dean advises staying away from online review platforms. Instead, she suggests sticking to the old-fashioned method of getting recommendations from friends and relatives, not virtual people.
Ms. Dean explained a few indicators that she looks for when trying to identify a fake review.
“The business has all five-star reviews, negative reviews are followed by five-star reviews, or the business has an abnormal number of positive reviews in a short period of time,” she noted. “Some businesses try to bury legitimate negative reviews by obtaining more recent, fake, positive ones. The recent reviews will contradict the specific criticisms in the negative review.”
She warned that consumers should not give credibility to reviews simply because the reviewer is dubbed “Elite” or a Google Local Guide, because she has seen plenty of these individuals posting fake reviews.
Unfortunately, review platforms haven’t been doing much self-policing. Google and Healthgrades have a series of policies against fake engagement, impersonation, misinformation, and misrepresentation, according to their websites. However, the only consequence of these violations is review removal.
Both Yelp! and Google say they have automated software that distinguishes real versus fake reviews. When Yelp! uncovers users engaging in compensation review activity, it removes their reviews, closes their account, and blocks those users from creating future Yelp! accounts.
Physicians’ basis
Moreover,
“I think there’s an erosion of business ethics because cheating is rewarded. You can’t compete in an environment where your competition is allowed to accumulate numerous fake reviews while you’re still trying to fill chairs in your business,” said Ms. Dean. “Your competition is then getting the business because the tech companies are allowing this fraud.”
Family physician and practice owner Mike Woo-Ming, MD, MPH, provides career coaching for physicians, including maintaining a good reputation – in-person and online. He has seen physicians bumping up their own five-star reviews personally as well as posting negative reviews for their competition.
“I’ve seen where they’re going to lose business, as many practices were affected through COVID,” he said. “Business owners can become desperate and may decide to start posting or buying reviews because they know people will choose certain services these days based upon reviews.”
Dr. Woo-Ming expressed his frustration with fellow physicians who give in to purchasing fake reviews, because the patients have no idea whether reviews are genuine or not.
To encourage genuine positive reviews, Dr. Woo-Ming’s practice uses a third-party app system that sends patients a follow-up email or text asking about their experience with a link to review sites.
“Honest reviews are a reflection of what I can do to improve my business. At the end of the day, if you’re truly providing great service and you’re helping people by providing great medical care, those are going to win out,” he said. “I would rather, as a responsible practice owner, improve the experience and outcome for the patient.”
A version of this article first appeared on Medscape.com.
New international dermatology registry tracks monkeypox cases
The American Academy of Dermatology and the International League of Dermatological Societies (ILDS) have created a new registry that now accepts reports from health care providers worldwide about monkeypox cases and monkeypox vaccine reactions.
Patient data such as names and dates of birth will not be collected.
“As with our joint COVID-19 registry, we will be doing real-time data analysis during the outbreak,” dermatologist Esther Freeman, MD, PhD, director of MGH Global Health Dermatology at Massachusetts General Hospital, Boston, and a member of the AAD’s monkeypox task force, said in an interview. “We will to try to feed information back to our front line in terms of clinical characteristics of cases, morphology, and any unexpected findings.”
According to Dr. Freeman, the principal investigator for the COVID-19 registry, this registry has allowed the quick gathering of information about dermatologic findings of COVID-19 from over 53 countries. “We have published over 15 papers, and we share data with outside investigators wishing to do their own analysis of registry-related data,” she said. “Our most-cited paper on COVID vaccine skin reactions has been cited almost 500 times since 2021. It has been used to educate the public on vaccine side effects and to combat vaccine hesitancy.”
The monkeypox registry “doesn’t belong to any one group or person,” Dr. Freeman said. “The idea with rapid data analysis is to be able to give back to the dermatologic community what is hard for us to see with any single case: Patterns and new findings that can be helpful to share with dermatologists and other physicians worldwide, all working together to stop an outbreak.”
The American Academy of Dermatology and the International League of Dermatological Societies (ILDS) have created a new registry that now accepts reports from health care providers worldwide about monkeypox cases and monkeypox vaccine reactions.
Patient data such as names and dates of birth will not be collected.
“As with our joint COVID-19 registry, we will be doing real-time data analysis during the outbreak,” dermatologist Esther Freeman, MD, PhD, director of MGH Global Health Dermatology at Massachusetts General Hospital, Boston, and a member of the AAD’s monkeypox task force, said in an interview. “We will to try to feed information back to our front line in terms of clinical characteristics of cases, morphology, and any unexpected findings.”
According to Dr. Freeman, the principal investigator for the COVID-19 registry, this registry has allowed the quick gathering of information about dermatologic findings of COVID-19 from over 53 countries. “We have published over 15 papers, and we share data with outside investigators wishing to do their own analysis of registry-related data,” she said. “Our most-cited paper on COVID vaccine skin reactions has been cited almost 500 times since 2021. It has been used to educate the public on vaccine side effects and to combat vaccine hesitancy.”
The monkeypox registry “doesn’t belong to any one group or person,” Dr. Freeman said. “The idea with rapid data analysis is to be able to give back to the dermatologic community what is hard for us to see with any single case: Patterns and new findings that can be helpful to share with dermatologists and other physicians worldwide, all working together to stop an outbreak.”
The American Academy of Dermatology and the International League of Dermatological Societies (ILDS) have created a new registry that now accepts reports from health care providers worldwide about monkeypox cases and monkeypox vaccine reactions.
Patient data such as names and dates of birth will not be collected.
“As with our joint COVID-19 registry, we will be doing real-time data analysis during the outbreak,” dermatologist Esther Freeman, MD, PhD, director of MGH Global Health Dermatology at Massachusetts General Hospital, Boston, and a member of the AAD’s monkeypox task force, said in an interview. “We will to try to feed information back to our front line in terms of clinical characteristics of cases, morphology, and any unexpected findings.”
According to Dr. Freeman, the principal investigator for the COVID-19 registry, this registry has allowed the quick gathering of information about dermatologic findings of COVID-19 from over 53 countries. “We have published over 15 papers, and we share data with outside investigators wishing to do their own analysis of registry-related data,” she said. “Our most-cited paper on COVID vaccine skin reactions has been cited almost 500 times since 2021. It has been used to educate the public on vaccine side effects and to combat vaccine hesitancy.”
The monkeypox registry “doesn’t belong to any one group or person,” Dr. Freeman said. “The idea with rapid data analysis is to be able to give back to the dermatologic community what is hard for us to see with any single case: Patterns and new findings that can be helpful to share with dermatologists and other physicians worldwide, all working together to stop an outbreak.”
Dermatology and monkeypox: What you need to know
.
Diagnosing cases “can be hard and folks should keep a very open mind and consider monkeypox virus,” said Misha Rosenbach, MD, a University of Pennsylvania dermatologist and member of the American Academy of Dermatology’s ad hoc task force to develop monkeypox content.
Although it’s named after a primate, it turns out that monkeypox is quite the copycat. As dermatologists have learned, its lesions can look like those caused by a long list of other diseases including herpes, varicella, and syphilis. In small numbers, they can even appear to be insect bites.
To make things more complicated, a patient can have one or two lesions – or dozens. They often cluster in the anogenital area, likely reflecting transmission via sexual intercourse, unlike previous outbreaks in which lesions appeared all over the body. “We have to let go of some of our conceptions about what monkeypox might look like,” said dermatologist Esther Freeman, MD, PhD, associate professor of dermatology, Harvard University, Boston, and a member of the AAD task force.
To make things even more complicated, “the spectrum of illness that we are seeing has ranged from limited, subtle lesions to dramatic, widespread, ulcerative/necrotic lesions,” said Dr. Rosenbach, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
But monkeypox has unique traits that can set it apart and pave the way toward a diagnosis, dermatologists say. And important patient data can help dermatologists gauge the likelihood of a case: Almost 99% of cases with data available have been in men, and among men with available information, 94% reported male-to-male sexual or close intimate contact during the 3 weeks before developing symptoms, according to a CDC report tracking cases from May through late July. So far, cases in women and children are extremely rare, although there have been some reported in the United States.
Are dermatologists likely to see monkeypox in the clinic? It’s unclear so far. Of four dermatologists interviewed for this article, only one has seen patients with monkeypox in person. But others say they’ve been sought for consultations. “I have been asked by infectious disease colleagues for advice remotely but have not seen it,” said dermatologist Howa Yeung, MD, MSc, assistant professor of dermatology, Emory University, Atlanta. “Most of the time, they’re catching all the symptomatic cases before any need for dermatology in-person referrals.”
Still, the rapid rate of growth of the outbreak – up from 3,487 in the United States on July 25 to 12,689 as of Aug.16 – suggests that more dermatologists will see cases, and consultations may become more common too.
Know your lesions
Lesions are the telltale signs of symptomatic monkeypox. According to a recent New England Journal of Medicine study of 528 monkeypox cases from 16 nations, diagnosed between April 27 and June 24, 2022, 95% had skin lesions (58% were vesiculopustular), most commonly in the anogenital area (73%), and on the trunk/arms/or legs (55%) and face (25%), and the palms/soles (10%).
However, “the current monkeypox outbreak often presents differently from the multiple classic vesiculopustules on the skin we see in textbooks,” Dr. Yeung said. “Sometimes people can present with throat pain or rectal pain, with isolated pharyngitis or proctitis. Sometimes there are so few lesions on the skin that it can be easily confused with a bug bite, folliculitis, herpes, dyshidrotic eczema, or other skin problems. This is where dermatologists will get consulted to clarify the diagnosis while the monkeypox PCR test is pending.”
Dr. Rosenbach, who has provided consultation services to other physicians about cases, said the lesions often appear to be vesicles or pustules, “but if you go to ‘pop’ it – e.g., for testing – it’s firm and without fluid. This is likely due to pox virus inclusion, similar to other diseases such as molluscum,” caused by another pox virus, he said. Molluscum lesions are “characteristically umbilicated, with a dimple in the center, and monkeypox lesions seem to be showing a roughly similar morphology with many bowl- or caldera-shaped lesions that are donut-like in appearance,” he added.
Over time, Dr. Rosenbach said, “lesions tend to evolve slowly from smaller flesh-colored or vaguely white firm papules to broader more umbilicated/donut-shaped lesions which may erode, ulcerate, develop a crust or scab, and then heal. The amount of scarring is not yet clear, but we anticipate it to be significant, especially in patients with more widespread or severe disease.”
Jon Peebles, MD, a dermatologist at Kaiser Permanente in Largo, Md., who has treated a few in-person monkeypox cases, said the lesions can be “exquisitely painful,” although he’s also seen patients with asymptomatic lesions. “Lesions are showing a predilection for the anogenital skin, though they can occur anywhere and not uncommonly involve the oral mucosa,” said Dr. Peebles, also a member of the AAD monkeypox task force.
Dr. Yeung said it’s important to ask patients about their sexual orientation, gender identity, and sexual behaviors. “That is the only way to know who your patients are and the only way to understand who else may be at risks and can benefit from contact tracing and additional prevention measures, such as vaccination for asymptomatic sex partners.” (The Jynneos smallpox vaccine is Food and Drug Administration–approved to prevent monkeypox, although its efficacy is not entirely clear, and there’s controversy over expanding its limited availability by administering the vaccine intradermally.)
It’s also important to keep in mind that sexually transmitted infections (STIs) are common in gay and bisexual men. “Just because the patient is diagnosed with gonorrhea or syphilis does not mean the patient cannot also have monkeypox,” Dr. Rosenbach said. Indeed, the NEJM study reported that of 377 patients screened, 29% had an STI other than HIV, mostly syphilis (9%) and gonorrhea (8%). Of all 528 patients in the study (all male or transgender/nonbinary), 41% were HIV-positive, and the median number of sex partners in the last 3 months was 5 (range, 3-15).
Testing is crucial to rule monkeypox in – or out
While monkeypox lesions can be confused for other diseases, Dr. Rosenbach said that a diagnosis can be confirmed through various tests. Varicella zoster virus (VZV) and herpes simplex virus (HSV) have distinct findings on Tzanck smears (nuclear molding, multinucleated cells), and have widely available fairly rapid tests (PCR, or in some places, DFA). “Staph and bacterial folliculitis can usually be cultured quickly,” he said. “If you have someone with no risk factors/exposure, and you test for VZV, HSV, folliculitis, and it’s negative – you should know within 24 hours in most places – then you can broaden your differential diagnosis and consider alternate explanations, including monkeypox.”
Quest Diagnostics and Labcorp, two of the largest commercial labs in the United States, are now offering monkeypox tests. Labcorp says its test has a 2- to 3-day turnaround time.
As for treatment, some physicians are prescribing off-label use of tecovirimat (also known as TPOXX or ST-246), a smallpox antiviral treatment. The CDC offers guidelines about its use. “It seems to work very fast, with patients improving in 24-72 hours,” Dr. Rosenbach said. However, “it is still very challenging to give and get. There’s a cumbersome system to prescribe it, and it needs to be shipped from the national stockpile. Dermatologists should be working with their state health department, infection control, and infectious disease doctors.”
It’s likely that dermatologists are not comfortable with the process to access the drug, he said, “but if we do not act quickly to control the current outbreak, we will all – unfortunately – need to learn to be comfortable prescribing it.”
In regard to pain control, an over-the-counter painkiller approach may be appropriate depending on comorbidities, Dr. Rosenbach said. “Some patients with very severe disease, such as perianal involvement and proctitis, have such severe pain they need to be hospitalized. This is less common.”
Recommendations pending on scarring prevention
There’s limited high-quality evidence about the prevention of scarring in diseases like monkeypox, Dr. Rosenbach noted. “Any recommendations are usually based on very small, limited, uncontrolled studies. In the case of monkeypox, truly we are off the edge of the map.”
He advises cleaning lesions with gentle soap and water – keeping in mind that contaminated towels may spread disease – and potentially using a topical ointment-based dressing such as a Vaseline/nonstick dressing or Vaseline-impregnated gauze. If there’s concern about superinfection, as can occur with staph infections, topical antibiotics such as mupirocin 2% ointment may be appropriate, he said.
“Some folks like to try silica gel sheets to prevent scarring,” Dr. Rosenbach said. “There’s not a lot of evidence to support that, but they’re unlikely to be harmful. I would personally consider them, but it really depends on the extent of disease, anatomic sites involved, and access to care.”
Emory University’s Dr. Yeung also suggested using silicone gel or sheets to optimize the scar appearance once the lesions have crusted over. “People have used lasers, microneedling, etc., to improve smallpox scar appearance,” he added, “and I’m sure dermatologists will be the ones to study what works best for treating monkeypox scars.”
As for the big picture, Dr. Yeung said that dermatologists are critical in the fight to control monkeypox: “We can help our colleagues and patients manage symptoms and wound care, advocate for vaccination and treatment, treat long-term scarring sequelae, and destigmatize LGBTQ health care.”
The dermatologists interviewed for this article report no disclosures.
.
Diagnosing cases “can be hard and folks should keep a very open mind and consider monkeypox virus,” said Misha Rosenbach, MD, a University of Pennsylvania dermatologist and member of the American Academy of Dermatology’s ad hoc task force to develop monkeypox content.
Although it’s named after a primate, it turns out that monkeypox is quite the copycat. As dermatologists have learned, its lesions can look like those caused by a long list of other diseases including herpes, varicella, and syphilis. In small numbers, they can even appear to be insect bites.
To make things more complicated, a patient can have one or two lesions – or dozens. They often cluster in the anogenital area, likely reflecting transmission via sexual intercourse, unlike previous outbreaks in which lesions appeared all over the body. “We have to let go of some of our conceptions about what monkeypox might look like,” said dermatologist Esther Freeman, MD, PhD, associate professor of dermatology, Harvard University, Boston, and a member of the AAD task force.
To make things even more complicated, “the spectrum of illness that we are seeing has ranged from limited, subtle lesions to dramatic, widespread, ulcerative/necrotic lesions,” said Dr. Rosenbach, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
But monkeypox has unique traits that can set it apart and pave the way toward a diagnosis, dermatologists say. And important patient data can help dermatologists gauge the likelihood of a case: Almost 99% of cases with data available have been in men, and among men with available information, 94% reported male-to-male sexual or close intimate contact during the 3 weeks before developing symptoms, according to a CDC report tracking cases from May through late July. So far, cases in women and children are extremely rare, although there have been some reported in the United States.
Are dermatologists likely to see monkeypox in the clinic? It’s unclear so far. Of four dermatologists interviewed for this article, only one has seen patients with monkeypox in person. But others say they’ve been sought for consultations. “I have been asked by infectious disease colleagues for advice remotely but have not seen it,” said dermatologist Howa Yeung, MD, MSc, assistant professor of dermatology, Emory University, Atlanta. “Most of the time, they’re catching all the symptomatic cases before any need for dermatology in-person referrals.”
Still, the rapid rate of growth of the outbreak – up from 3,487 in the United States on July 25 to 12,689 as of Aug.16 – suggests that more dermatologists will see cases, and consultations may become more common too.
Know your lesions
Lesions are the telltale signs of symptomatic monkeypox. According to a recent New England Journal of Medicine study of 528 monkeypox cases from 16 nations, diagnosed between April 27 and June 24, 2022, 95% had skin lesions (58% were vesiculopustular), most commonly in the anogenital area (73%), and on the trunk/arms/or legs (55%) and face (25%), and the palms/soles (10%).
However, “the current monkeypox outbreak often presents differently from the multiple classic vesiculopustules on the skin we see in textbooks,” Dr. Yeung said. “Sometimes people can present with throat pain or rectal pain, with isolated pharyngitis or proctitis. Sometimes there are so few lesions on the skin that it can be easily confused with a bug bite, folliculitis, herpes, dyshidrotic eczema, or other skin problems. This is where dermatologists will get consulted to clarify the diagnosis while the monkeypox PCR test is pending.”
Dr. Rosenbach, who has provided consultation services to other physicians about cases, said the lesions often appear to be vesicles or pustules, “but if you go to ‘pop’ it – e.g., for testing – it’s firm and without fluid. This is likely due to pox virus inclusion, similar to other diseases such as molluscum,” caused by another pox virus, he said. Molluscum lesions are “characteristically umbilicated, with a dimple in the center, and monkeypox lesions seem to be showing a roughly similar morphology with many bowl- or caldera-shaped lesions that are donut-like in appearance,” he added.
Over time, Dr. Rosenbach said, “lesions tend to evolve slowly from smaller flesh-colored or vaguely white firm papules to broader more umbilicated/donut-shaped lesions which may erode, ulcerate, develop a crust or scab, and then heal. The amount of scarring is not yet clear, but we anticipate it to be significant, especially in patients with more widespread or severe disease.”
Jon Peebles, MD, a dermatologist at Kaiser Permanente in Largo, Md., who has treated a few in-person monkeypox cases, said the lesions can be “exquisitely painful,” although he’s also seen patients with asymptomatic lesions. “Lesions are showing a predilection for the anogenital skin, though they can occur anywhere and not uncommonly involve the oral mucosa,” said Dr. Peebles, also a member of the AAD monkeypox task force.
Dr. Yeung said it’s important to ask patients about their sexual orientation, gender identity, and sexual behaviors. “That is the only way to know who your patients are and the only way to understand who else may be at risks and can benefit from contact tracing and additional prevention measures, such as vaccination for asymptomatic sex partners.” (The Jynneos smallpox vaccine is Food and Drug Administration–approved to prevent monkeypox, although its efficacy is not entirely clear, and there’s controversy over expanding its limited availability by administering the vaccine intradermally.)
It’s also important to keep in mind that sexually transmitted infections (STIs) are common in gay and bisexual men. “Just because the patient is diagnosed with gonorrhea or syphilis does not mean the patient cannot also have monkeypox,” Dr. Rosenbach said. Indeed, the NEJM study reported that of 377 patients screened, 29% had an STI other than HIV, mostly syphilis (9%) and gonorrhea (8%). Of all 528 patients in the study (all male or transgender/nonbinary), 41% were HIV-positive, and the median number of sex partners in the last 3 months was 5 (range, 3-15).
Testing is crucial to rule monkeypox in – or out
While monkeypox lesions can be confused for other diseases, Dr. Rosenbach said that a diagnosis can be confirmed through various tests. Varicella zoster virus (VZV) and herpes simplex virus (HSV) have distinct findings on Tzanck smears (nuclear molding, multinucleated cells), and have widely available fairly rapid tests (PCR, or in some places, DFA). “Staph and bacterial folliculitis can usually be cultured quickly,” he said. “If you have someone with no risk factors/exposure, and you test for VZV, HSV, folliculitis, and it’s negative – you should know within 24 hours in most places – then you can broaden your differential diagnosis and consider alternate explanations, including monkeypox.”
Quest Diagnostics and Labcorp, two of the largest commercial labs in the United States, are now offering monkeypox tests. Labcorp says its test has a 2- to 3-day turnaround time.
As for treatment, some physicians are prescribing off-label use of tecovirimat (also known as TPOXX or ST-246), a smallpox antiviral treatment. The CDC offers guidelines about its use. “It seems to work very fast, with patients improving in 24-72 hours,” Dr. Rosenbach said. However, “it is still very challenging to give and get. There’s a cumbersome system to prescribe it, and it needs to be shipped from the national stockpile. Dermatologists should be working with their state health department, infection control, and infectious disease doctors.”
It’s likely that dermatologists are not comfortable with the process to access the drug, he said, “but if we do not act quickly to control the current outbreak, we will all – unfortunately – need to learn to be comfortable prescribing it.”
In regard to pain control, an over-the-counter painkiller approach may be appropriate depending on comorbidities, Dr. Rosenbach said. “Some patients with very severe disease, such as perianal involvement and proctitis, have such severe pain they need to be hospitalized. This is less common.”
Recommendations pending on scarring prevention
There’s limited high-quality evidence about the prevention of scarring in diseases like monkeypox, Dr. Rosenbach noted. “Any recommendations are usually based on very small, limited, uncontrolled studies. In the case of monkeypox, truly we are off the edge of the map.”
He advises cleaning lesions with gentle soap and water – keeping in mind that contaminated towels may spread disease – and potentially using a topical ointment-based dressing such as a Vaseline/nonstick dressing or Vaseline-impregnated gauze. If there’s concern about superinfection, as can occur with staph infections, topical antibiotics such as mupirocin 2% ointment may be appropriate, he said.
“Some folks like to try silica gel sheets to prevent scarring,” Dr. Rosenbach said. “There’s not a lot of evidence to support that, but they’re unlikely to be harmful. I would personally consider them, but it really depends on the extent of disease, anatomic sites involved, and access to care.”
Emory University’s Dr. Yeung also suggested using silicone gel or sheets to optimize the scar appearance once the lesions have crusted over. “People have used lasers, microneedling, etc., to improve smallpox scar appearance,” he added, “and I’m sure dermatologists will be the ones to study what works best for treating monkeypox scars.”
As for the big picture, Dr. Yeung said that dermatologists are critical in the fight to control monkeypox: “We can help our colleagues and patients manage symptoms and wound care, advocate for vaccination and treatment, treat long-term scarring sequelae, and destigmatize LGBTQ health care.”
The dermatologists interviewed for this article report no disclosures.
.
Diagnosing cases “can be hard and folks should keep a very open mind and consider monkeypox virus,” said Misha Rosenbach, MD, a University of Pennsylvania dermatologist and member of the American Academy of Dermatology’s ad hoc task force to develop monkeypox content.
Although it’s named after a primate, it turns out that monkeypox is quite the copycat. As dermatologists have learned, its lesions can look like those caused by a long list of other diseases including herpes, varicella, and syphilis. In small numbers, they can even appear to be insect bites.
To make things more complicated, a patient can have one or two lesions – or dozens. They often cluster in the anogenital area, likely reflecting transmission via sexual intercourse, unlike previous outbreaks in which lesions appeared all over the body. “We have to let go of some of our conceptions about what monkeypox might look like,” said dermatologist Esther Freeman, MD, PhD, associate professor of dermatology, Harvard University, Boston, and a member of the AAD task force.
To make things even more complicated, “the spectrum of illness that we are seeing has ranged from limited, subtle lesions to dramatic, widespread, ulcerative/necrotic lesions,” said Dr. Rosenbach, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
But monkeypox has unique traits that can set it apart and pave the way toward a diagnosis, dermatologists say. And important patient data can help dermatologists gauge the likelihood of a case: Almost 99% of cases with data available have been in men, and among men with available information, 94% reported male-to-male sexual or close intimate contact during the 3 weeks before developing symptoms, according to a CDC report tracking cases from May through late July. So far, cases in women and children are extremely rare, although there have been some reported in the United States.
Are dermatologists likely to see monkeypox in the clinic? It’s unclear so far. Of four dermatologists interviewed for this article, only one has seen patients with monkeypox in person. But others say they’ve been sought for consultations. “I have been asked by infectious disease colleagues for advice remotely but have not seen it,” said dermatologist Howa Yeung, MD, MSc, assistant professor of dermatology, Emory University, Atlanta. “Most of the time, they’re catching all the symptomatic cases before any need for dermatology in-person referrals.”
Still, the rapid rate of growth of the outbreak – up from 3,487 in the United States on July 25 to 12,689 as of Aug.16 – suggests that more dermatologists will see cases, and consultations may become more common too.
Know your lesions
Lesions are the telltale signs of symptomatic monkeypox. According to a recent New England Journal of Medicine study of 528 monkeypox cases from 16 nations, diagnosed between April 27 and June 24, 2022, 95% had skin lesions (58% were vesiculopustular), most commonly in the anogenital area (73%), and on the trunk/arms/or legs (55%) and face (25%), and the palms/soles (10%).
However, “the current monkeypox outbreak often presents differently from the multiple classic vesiculopustules on the skin we see in textbooks,” Dr. Yeung said. “Sometimes people can present with throat pain or rectal pain, with isolated pharyngitis or proctitis. Sometimes there are so few lesions on the skin that it can be easily confused with a bug bite, folliculitis, herpes, dyshidrotic eczema, or other skin problems. This is where dermatologists will get consulted to clarify the diagnosis while the monkeypox PCR test is pending.”
Dr. Rosenbach, who has provided consultation services to other physicians about cases, said the lesions often appear to be vesicles or pustules, “but if you go to ‘pop’ it – e.g., for testing – it’s firm and without fluid. This is likely due to pox virus inclusion, similar to other diseases such as molluscum,” caused by another pox virus, he said. Molluscum lesions are “characteristically umbilicated, with a dimple in the center, and monkeypox lesions seem to be showing a roughly similar morphology with many bowl- or caldera-shaped lesions that are donut-like in appearance,” he added.
Over time, Dr. Rosenbach said, “lesions tend to evolve slowly from smaller flesh-colored or vaguely white firm papules to broader more umbilicated/donut-shaped lesions which may erode, ulcerate, develop a crust or scab, and then heal. The amount of scarring is not yet clear, but we anticipate it to be significant, especially in patients with more widespread or severe disease.”
Jon Peebles, MD, a dermatologist at Kaiser Permanente in Largo, Md., who has treated a few in-person monkeypox cases, said the lesions can be “exquisitely painful,” although he’s also seen patients with asymptomatic lesions. “Lesions are showing a predilection for the anogenital skin, though they can occur anywhere and not uncommonly involve the oral mucosa,” said Dr. Peebles, also a member of the AAD monkeypox task force.
Dr. Yeung said it’s important to ask patients about their sexual orientation, gender identity, and sexual behaviors. “That is the only way to know who your patients are and the only way to understand who else may be at risks and can benefit from contact tracing and additional prevention measures, such as vaccination for asymptomatic sex partners.” (The Jynneos smallpox vaccine is Food and Drug Administration–approved to prevent monkeypox, although its efficacy is not entirely clear, and there’s controversy over expanding its limited availability by administering the vaccine intradermally.)
It’s also important to keep in mind that sexually transmitted infections (STIs) are common in gay and bisexual men. “Just because the patient is diagnosed with gonorrhea or syphilis does not mean the patient cannot also have monkeypox,” Dr. Rosenbach said. Indeed, the NEJM study reported that of 377 patients screened, 29% had an STI other than HIV, mostly syphilis (9%) and gonorrhea (8%). Of all 528 patients in the study (all male or transgender/nonbinary), 41% were HIV-positive, and the median number of sex partners in the last 3 months was 5 (range, 3-15).
Testing is crucial to rule monkeypox in – or out
While monkeypox lesions can be confused for other diseases, Dr. Rosenbach said that a diagnosis can be confirmed through various tests. Varicella zoster virus (VZV) and herpes simplex virus (HSV) have distinct findings on Tzanck smears (nuclear molding, multinucleated cells), and have widely available fairly rapid tests (PCR, or in some places, DFA). “Staph and bacterial folliculitis can usually be cultured quickly,” he said. “If you have someone with no risk factors/exposure, and you test for VZV, HSV, folliculitis, and it’s negative – you should know within 24 hours in most places – then you can broaden your differential diagnosis and consider alternate explanations, including monkeypox.”
Quest Diagnostics and Labcorp, two of the largest commercial labs in the United States, are now offering monkeypox tests. Labcorp says its test has a 2- to 3-day turnaround time.
As for treatment, some physicians are prescribing off-label use of tecovirimat (also known as TPOXX or ST-246), a smallpox antiviral treatment. The CDC offers guidelines about its use. “It seems to work very fast, with patients improving in 24-72 hours,” Dr. Rosenbach said. However, “it is still very challenging to give and get. There’s a cumbersome system to prescribe it, and it needs to be shipped from the national stockpile. Dermatologists should be working with their state health department, infection control, and infectious disease doctors.”
It’s likely that dermatologists are not comfortable with the process to access the drug, he said, “but if we do not act quickly to control the current outbreak, we will all – unfortunately – need to learn to be comfortable prescribing it.”
In regard to pain control, an over-the-counter painkiller approach may be appropriate depending on comorbidities, Dr. Rosenbach said. “Some patients with very severe disease, such as perianal involvement and proctitis, have such severe pain they need to be hospitalized. This is less common.”
Recommendations pending on scarring prevention
There’s limited high-quality evidence about the prevention of scarring in diseases like monkeypox, Dr. Rosenbach noted. “Any recommendations are usually based on very small, limited, uncontrolled studies. In the case of monkeypox, truly we are off the edge of the map.”
He advises cleaning lesions with gentle soap and water – keeping in mind that contaminated towels may spread disease – and potentially using a topical ointment-based dressing such as a Vaseline/nonstick dressing or Vaseline-impregnated gauze. If there’s concern about superinfection, as can occur with staph infections, topical antibiotics such as mupirocin 2% ointment may be appropriate, he said.
“Some folks like to try silica gel sheets to prevent scarring,” Dr. Rosenbach said. “There’s not a lot of evidence to support that, but they’re unlikely to be harmful. I would personally consider them, but it really depends on the extent of disease, anatomic sites involved, and access to care.”
Emory University’s Dr. Yeung also suggested using silicone gel or sheets to optimize the scar appearance once the lesions have crusted over. “People have used lasers, microneedling, etc., to improve smallpox scar appearance,” he added, “and I’m sure dermatologists will be the ones to study what works best for treating monkeypox scars.”
As for the big picture, Dr. Yeung said that dermatologists are critical in the fight to control monkeypox: “We can help our colleagues and patients manage symptoms and wound care, advocate for vaccination and treatment, treat long-term scarring sequelae, and destigmatize LGBTQ health care.”
The dermatologists interviewed for this article report no disclosures.