User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
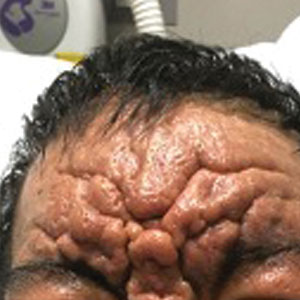
Exaggerated Facial Lines on the Forehead and Cheeks
The Diagnosis: Pachydermoperiostosis
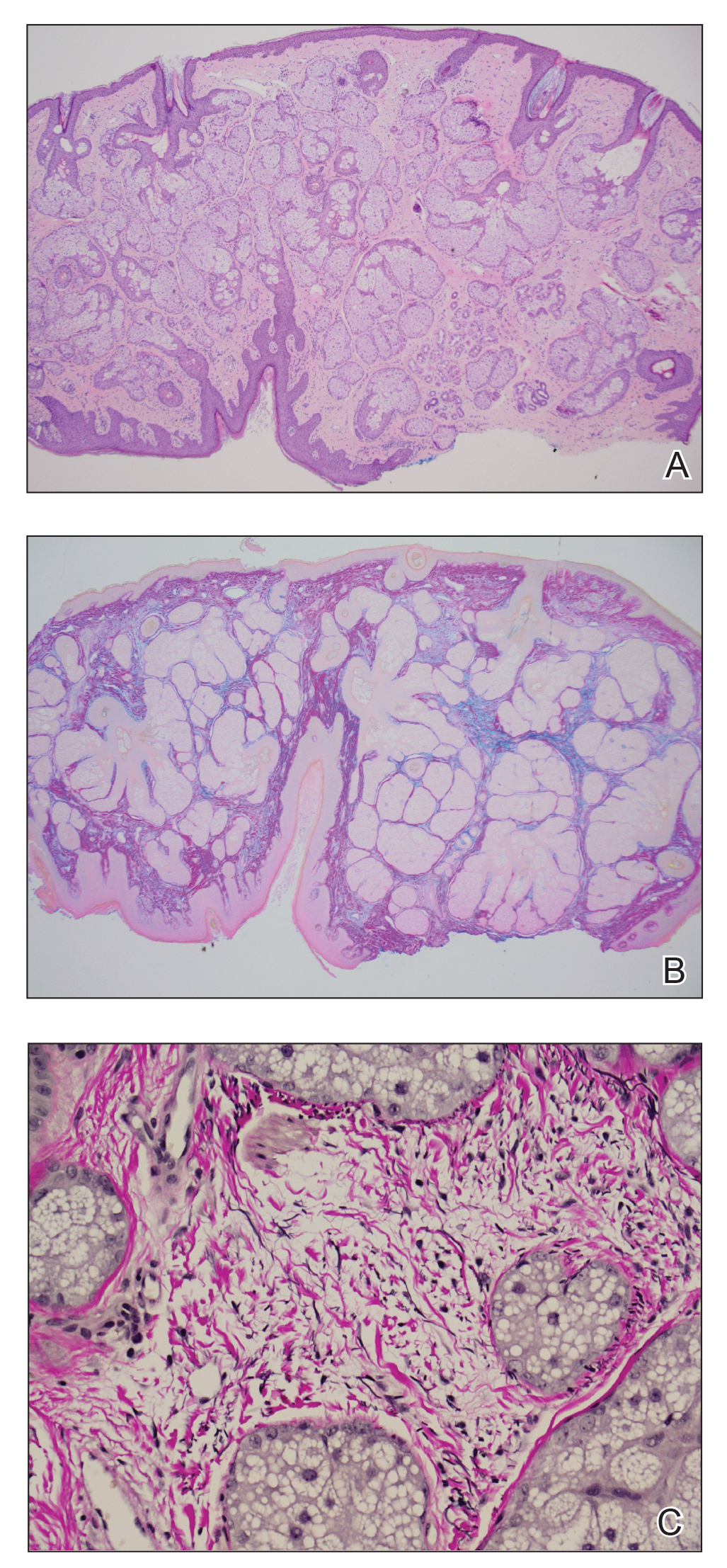
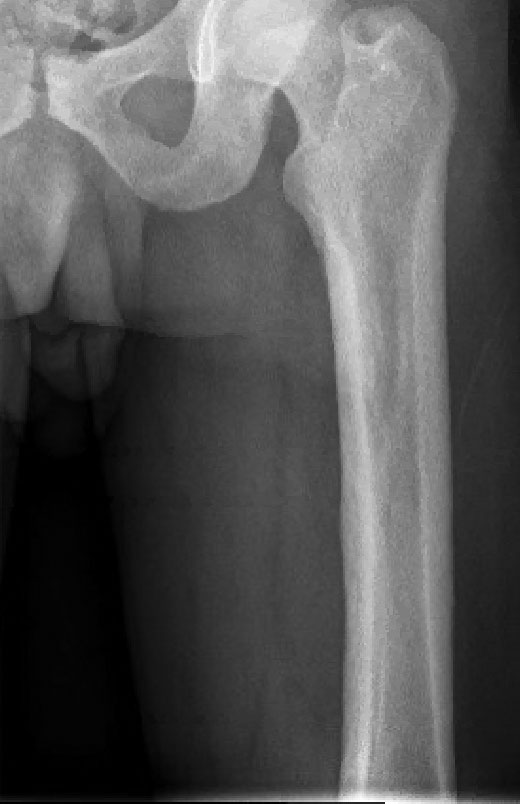
Histopathology of the forehead punch biopsy demonstrated sebaceous hyperplasia with an occupation rate of greater than 40%, increased mucin, elastic fiber degeneration, and fibrosis. Pachydermia is graded from 0 to 3 depending on the degree of these changes; our patient met criteria for grade 3 pachydermia (Figure 1). Radiography revealed diffuse cortical thickening of the long bones that was most marked in the left femur (Figure 2); however, no other findings were demonstrative of Paget disease.
Pachydermoperiostosis (PDP)(also known as Touraine-Solente-Golé syndrome or primary hypertrophic osteoarthropathy) is a rare genetic condition that affects both the dermatologic and skeletal systems. Clinical features of the disease include progressive thickening and furrowing of the skin on the scalp and face (known as pachydermia), digital clubbing, and periostosis. Other potential cutaneous features include seborrhea, acne, hyperhidrosis of the palms and soles, cutis verticis gyrata, eczema, and a burning sensation of the hands and feet. Myelofibrosis and gastrointestinal abnormalities also have been reported.1
The disease typically affects males (7:1 ratio); also, men typically display a more severe phenotype of the disease.2 It most commonly begins during puberty and follows a generally progressive course of 5 to 20 years before eventually stabilizing. Both autosomal-dominant with incomplete penetrance and recessive inheritance versions of PDP can occur. Prostaglandin E2 (PGE2) has been implicated in the pathogenesis of PDP; PGE2 is important in the inflammatory response and may evolve from disrupted protein degradation pathways.3 Sasaki et al4 additionally reported that the severity of pachydermia clinically and histologically appeared to correlate with the serum PGE2 levels in affected patients. Prostaglandin E2 causes a vasodilatory effect, perhaps explaining the clubbing observed in PDP, and also modifies the activity of osteoblasts and osteoclasts, causing the bone remodeling observed in the disease.4
In our patient, the initial differential diagnosis included PDP, as well as lepromatous leprosy, acromegaly, Paget disease of the bone, amyloidosis, scleromyxedema, and cutaneous T-cell lymphoma. However, the time course of the disease, lack of numerous symmetric thickened plaques and madarosis, and pathology argued against lepromatous leprosy. Acromegaly was ruled out due to lack of macroglossia as well as laboratory analysis within reference range including IGF-1 levels and thyroid function tests. Biopsy findings ultimately ruled out amyloidosis and cutaneous T-cell lymphoma. The bone scan revealed diffuse cortical thickening consistent with PDP, and there were no other radiologic findings suggestive of Paget disease. Pachydermoperiostosis is diagnosed using the Borochowitz criteria, which entails that 2 of the following 4 fulfillment criteria must be met: familial transmission, pachydermia, digital clubbing, and/or bony involvement with evidence of radiologic alterations or pain. Our patient met all 4 criteria. The clinical manifestations of PDP are variable with respect to skin and bone changes. The various clinical expressions include the complete form (ie, pachydermia, cutis verticis gyrata, periostosis), the incomplete form (ie, absence of cutis verticis gyrata), and forme fruste (ie, pachydermia with minimal or absent periostosis).5
Management for PDP involves surgical correction for cosmesis as well as for functional concerns if present. Symptoms secondary to periostosis should be managed with symptomatic treatment such as nonsteroidal antiinflammatory drugs. Patients managed with etoricoxib, a COX-2–selective nonsteroidal anti-inflammatory drug, have had normalized inflammatory markers that resulted in the lessening of forehead skin folds. Oral aescin has been shown to relieve joint pain due to its antiedematous effect.6 Our patient received treatment with nonsteroidal anti-inflammatory drugs for symptomatic management of the associated joint pain but unfortunately was lost to follow-up.
- Castori M, Sinibaldi L, Mingarelli R, et al. Pachydermoperiostosis: an update. Clin Genet. 2005;68:477-486.
- Reginato AJ, Shipachasse V, Guerrero R. Familial idiopathic hypertrophic osteoarthropathy and cranial suture defects in children. Skel Radiol. 1982;8:105-109.
- Coggins KG, Coffman TM, Koller BH. The Hippocratic finger points the blame at PGE2. Nat Genet. 2008;40:691-692.
- Sasaki T, Niizeki H, Shimizu A, et al. Identification of mutations in the prostaglandin transporter gene SLCO2A1 and its phenotype-genotype correlation in Japanese patients with pachydermoperiostosis. J Dermatol Sci. 2012;68:36-44.
- Bhaskaranand K, Shetty RR, Bhat AK. Pachydermoperiostosis: three case reports. J Orthop Surg (Hong Kong). 2001;9:61-66.
- Zhang H, Yang B. Successful treatment of pachydermoperiostosis patients with etoricoxib, aescin, and arthroscopic synovectomy: two case reports. Medicine (Baltimore). 2017;96:E8865.
The Diagnosis: Pachydermoperiostosis
Histopathology of the forehead punch biopsy demonstrated sebaceous hyperplasia with an occupation rate of greater than 40%, increased mucin, elastic fiber degeneration, and fibrosis. Pachydermia is graded from 0 to 3 depending on the degree of these changes; our patient met criteria for grade 3 pachydermia (Figure 1). Radiography revealed diffuse cortical thickening of the long bones that was most marked in the left femur (Figure 2); however, no other findings were demonstrative of Paget disease.

Pachydermoperiostosis (PDP)(also known as Touraine-Solente-Golé syndrome or primary hypertrophic osteoarthropathy) is a rare genetic condition that affects both the dermatologic and skeletal systems. Clinical features of the disease include progressive thickening and furrowing of the skin on the scalp and face (known as pachydermia), digital clubbing, and periostosis. Other potential cutaneous features include seborrhea, acne, hyperhidrosis of the palms and soles, cutis verticis gyrata, eczema, and a burning sensation of the hands and feet. Myelofibrosis and gastrointestinal abnormalities also have been reported.1

The disease typically affects males (7:1 ratio); also, men typically display a more severe phenotype of the disease.2 It most commonly begins during puberty and follows a generally progressive course of 5 to 20 years before eventually stabilizing. Both autosomal-dominant with incomplete penetrance and recessive inheritance versions of PDP can occur. Prostaglandin E2 (PGE2) has been implicated in the pathogenesis of PDP; PGE2 is important in the inflammatory response and may evolve from disrupted protein degradation pathways.3 Sasaki et al4 additionally reported that the severity of pachydermia clinically and histologically appeared to correlate with the serum PGE2 levels in affected patients. Prostaglandin E2 causes a vasodilatory effect, perhaps explaining the clubbing observed in PDP, and also modifies the activity of osteoblasts and osteoclasts, causing the bone remodeling observed in the disease.4
In our patient, the initial differential diagnosis included PDP, as well as lepromatous leprosy, acromegaly, Paget disease of the bone, amyloidosis, scleromyxedema, and cutaneous T-cell lymphoma. However, the time course of the disease, lack of numerous symmetric thickened plaques and madarosis, and pathology argued against lepromatous leprosy. Acromegaly was ruled out due to lack of macroglossia as well as laboratory analysis within reference range including IGF-1 levels and thyroid function tests. Biopsy findings ultimately ruled out amyloidosis and cutaneous T-cell lymphoma. The bone scan revealed diffuse cortical thickening consistent with PDP, and there were no other radiologic findings suggestive of Paget disease. Pachydermoperiostosis is diagnosed using the Borochowitz criteria, which entails that 2 of the following 4 fulfillment criteria must be met: familial transmission, pachydermia, digital clubbing, and/or bony involvement with evidence of radiologic alterations or pain. Our patient met all 4 criteria. The clinical manifestations of PDP are variable with respect to skin and bone changes. The various clinical expressions include the complete form (ie, pachydermia, cutis verticis gyrata, periostosis), the incomplete form (ie, absence of cutis verticis gyrata), and forme fruste (ie, pachydermia with minimal or absent periostosis).5
Management for PDP involves surgical correction for cosmesis as well as for functional concerns if present. Symptoms secondary to periostosis should be managed with symptomatic treatment such as nonsteroidal antiinflammatory drugs. Patients managed with etoricoxib, a COX-2–selective nonsteroidal anti-inflammatory drug, have had normalized inflammatory markers that resulted in the lessening of forehead skin folds. Oral aescin has been shown to relieve joint pain due to its antiedematous effect.6 Our patient received treatment with nonsteroidal anti-inflammatory drugs for symptomatic management of the associated joint pain but unfortunately was lost to follow-up.
The Diagnosis: Pachydermoperiostosis
Histopathology of the forehead punch biopsy demonstrated sebaceous hyperplasia with an occupation rate of greater than 40%, increased mucin, elastic fiber degeneration, and fibrosis. Pachydermia is graded from 0 to 3 depending on the degree of these changes; our patient met criteria for grade 3 pachydermia (Figure 1). Radiography revealed diffuse cortical thickening of the long bones that was most marked in the left femur (Figure 2); however, no other findings were demonstrative of Paget disease.

Pachydermoperiostosis (PDP)(also known as Touraine-Solente-Golé syndrome or primary hypertrophic osteoarthropathy) is a rare genetic condition that affects both the dermatologic and skeletal systems. Clinical features of the disease include progressive thickening and furrowing of the skin on the scalp and face (known as pachydermia), digital clubbing, and periostosis. Other potential cutaneous features include seborrhea, acne, hyperhidrosis of the palms and soles, cutis verticis gyrata, eczema, and a burning sensation of the hands and feet. Myelofibrosis and gastrointestinal abnormalities also have been reported.1

The disease typically affects males (7:1 ratio); also, men typically display a more severe phenotype of the disease.2 It most commonly begins during puberty and follows a generally progressive course of 5 to 20 years before eventually stabilizing. Both autosomal-dominant with incomplete penetrance and recessive inheritance versions of PDP can occur. Prostaglandin E2 (PGE2) has been implicated in the pathogenesis of PDP; PGE2 is important in the inflammatory response and may evolve from disrupted protein degradation pathways.3 Sasaki et al4 additionally reported that the severity of pachydermia clinically and histologically appeared to correlate with the serum PGE2 levels in affected patients. Prostaglandin E2 causes a vasodilatory effect, perhaps explaining the clubbing observed in PDP, and also modifies the activity of osteoblasts and osteoclasts, causing the bone remodeling observed in the disease.4
In our patient, the initial differential diagnosis included PDP, as well as lepromatous leprosy, acromegaly, Paget disease of the bone, amyloidosis, scleromyxedema, and cutaneous T-cell lymphoma. However, the time course of the disease, lack of numerous symmetric thickened plaques and madarosis, and pathology argued against lepromatous leprosy. Acromegaly was ruled out due to lack of macroglossia as well as laboratory analysis within reference range including IGF-1 levels and thyroid function tests. Biopsy findings ultimately ruled out amyloidosis and cutaneous T-cell lymphoma. The bone scan revealed diffuse cortical thickening consistent with PDP, and there were no other radiologic findings suggestive of Paget disease. Pachydermoperiostosis is diagnosed using the Borochowitz criteria, which entails that 2 of the following 4 fulfillment criteria must be met: familial transmission, pachydermia, digital clubbing, and/or bony involvement with evidence of radiologic alterations or pain. Our patient met all 4 criteria. The clinical manifestations of PDP are variable with respect to skin and bone changes. The various clinical expressions include the complete form (ie, pachydermia, cutis verticis gyrata, periostosis), the incomplete form (ie, absence of cutis verticis gyrata), and forme fruste (ie, pachydermia with minimal or absent periostosis).5
Management for PDP involves surgical correction for cosmesis as well as for functional concerns if present. Symptoms secondary to periostosis should be managed with symptomatic treatment such as nonsteroidal antiinflammatory drugs. Patients managed with etoricoxib, a COX-2–selective nonsteroidal anti-inflammatory drug, have had normalized inflammatory markers that resulted in the lessening of forehead skin folds. Oral aescin has been shown to relieve joint pain due to its antiedematous effect.6 Our patient received treatment with nonsteroidal anti-inflammatory drugs for symptomatic management of the associated joint pain but unfortunately was lost to follow-up.
- Castori M, Sinibaldi L, Mingarelli R, et al. Pachydermoperiostosis: an update. Clin Genet. 2005;68:477-486.
- Reginato AJ, Shipachasse V, Guerrero R. Familial idiopathic hypertrophic osteoarthropathy and cranial suture defects in children. Skel Radiol. 1982;8:105-109.
- Coggins KG, Coffman TM, Koller BH. The Hippocratic finger points the blame at PGE2. Nat Genet. 2008;40:691-692.
- Sasaki T, Niizeki H, Shimizu A, et al. Identification of mutations in the prostaglandin transporter gene SLCO2A1 and its phenotype-genotype correlation in Japanese patients with pachydermoperiostosis. J Dermatol Sci. 2012;68:36-44.
- Bhaskaranand K, Shetty RR, Bhat AK. Pachydermoperiostosis: three case reports. J Orthop Surg (Hong Kong). 2001;9:61-66.
- Zhang H, Yang B. Successful treatment of pachydermoperiostosis patients with etoricoxib, aescin, and arthroscopic synovectomy: two case reports. Medicine (Baltimore). 2017;96:E8865.
- Castori M, Sinibaldi L, Mingarelli R, et al. Pachydermoperiostosis: an update. Clin Genet. 2005;68:477-486.
- Reginato AJ, Shipachasse V, Guerrero R. Familial idiopathic hypertrophic osteoarthropathy and cranial suture defects in children. Skel Radiol. 1982;8:105-109.
- Coggins KG, Coffman TM, Koller BH. The Hippocratic finger points the blame at PGE2. Nat Genet. 2008;40:691-692.
- Sasaki T, Niizeki H, Shimizu A, et al. Identification of mutations in the prostaglandin transporter gene SLCO2A1 and its phenotype-genotype correlation in Japanese patients with pachydermoperiostosis. J Dermatol Sci. 2012;68:36-44.
- Bhaskaranand K, Shetty RR, Bhat AK. Pachydermoperiostosis: three case reports. J Orthop Surg (Hong Kong). 2001;9:61-66.
- Zhang H, Yang B. Successful treatment of pachydermoperiostosis patients with etoricoxib, aescin, and arthroscopic synovectomy: two case reports. Medicine (Baltimore). 2017;96:E8865.
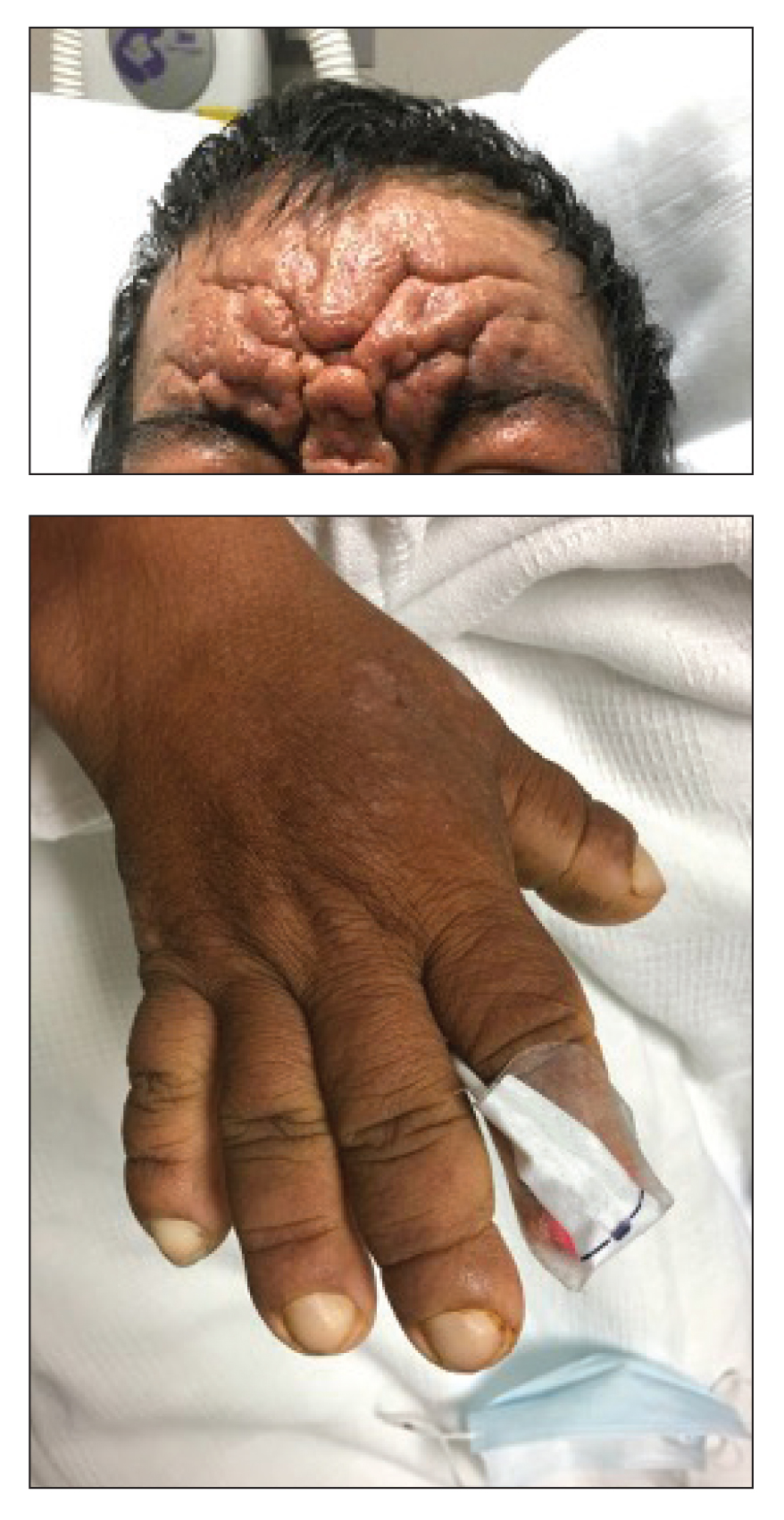
A 36-year-old man presented to the emergency department with an olecranon fracture after falling from a tree. The patient had a medical history of type 2 diabetes mellitus and a surgical history of facial cosmetic surgery. He underwent internal fixation with orthopedic surgery for the olecranon fracture, and dermatology subsequently was consulted due to diffuse skin changes on the face. He reported that these dermatologic changes began around 17 years of age and had progressed to the current presentation. He denied itching, burning, pain, or contact with armadillos. A family history revealed the patient’s brother also had a similar appearance. Physical examination revealed exaggerated facial lines on the forehead (top) and cheeks. Digital clubbing and skin thickening were noted on the hands (bottom) and feet; examination of the back revealed multiple hypopigmented patches. Observation of the scalp showed multiple symmetric ridges and grooves with sparse overlying hair consistent with cutis verticis gyrata. A punch biopsy of the forehead was obtained as well as bone radiography taken previously by the primary team.
Dig like an archaeologist
You can observe a lot by watching. – Yogi Berra
He was a fit man in his 40s. Thick legs. Maybe he was a long-distance walker? The bones of his right arm were more developed than his left – a right-handed thrower. His lower left fibula was fractured from a severely rolled ankle. He carried a walking stick that was glossy in the middle from where he gripped it with his left hand, dragging his bad left foot along. Dental cavities tell the story of his diet: honey, carobs, dates. Carbon 14 dating confirms that he lived during the Chalcolithic period, approximately 6,000 years ago. He was likely a shepherd in the Judean Desert.
Isn’t it amazing how much we can know about another human even across such an enormous chasm of time? If you’d asked me when I was 11 what I wanted to be, I’d have said archaeologist. .
A 64-year-old woman with a 4-cm red, brown shiny plaque on her right calf. She burned it on her boyfriend’s Harley Davidson nearly 40 years ago. She wonders where he is now.
A 58-year-old man with a 3-inch scar on his right wrist. He fell off his 6-year-old’s skimboard. ORIF.
A 40-year-old woman with bilateral mastectomy scars.
A 66-year-old with a lichenified nodule on his left forearm. When I shaved it off, a quarter inch spicule of glass came out. It was from a car accident in his first car, a Chevy Impala. He saved the piece of glass as a souvenir.
A fit 50-year-old with extensive scars on his feet and ankles. “Yeah, I went ‘whistling-in’ on a training jump,” he said. He was a retired Navy Seal and raconteur with quite a tale about the day his parachute malfunctioned. Some well placed live oak trees is why he’s around for his skin screening.
A classic, rope-like open-heart scar on the chest of a thin, young, healthy, flaxen-haired woman. Dissected aorta.
A 30-something woman dressed in a pants suit with razor-thin parallel scars on her volar forearms and proximal thighs. She asks if any laser could remove them.
A rotund, hard-living, bearded man with chest and upper-arm tattoos of flames and nudie girls now mixed with the striking face of an old woman and three little kids: His mom and grandkids. He shows me where the fourth grandkid will go and gives me a bear hug to thank me for the care when he leaves.
Attending to these details shifts us from autopilot to present. It keeps us involved, holding our attention even if it’s the 20th skin screening or diabetic foot exam of the day. And what a gift to share in the intimate details of another’s life.
Like examining the minute details of an ancient bone, dig for the history with curiosity, pity, humility. The perfect moment for asking might be when you stand with your #15 blade ready to introduce a new scar and become part of this human’s story forever.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
You can observe a lot by watching. – Yogi Berra
He was a fit man in his 40s. Thick legs. Maybe he was a long-distance walker? The bones of his right arm were more developed than his left – a right-handed thrower. His lower left fibula was fractured from a severely rolled ankle. He carried a walking stick that was glossy in the middle from where he gripped it with his left hand, dragging his bad left foot along. Dental cavities tell the story of his diet: honey, carobs, dates. Carbon 14 dating confirms that he lived during the Chalcolithic period, approximately 6,000 years ago. He was likely a shepherd in the Judean Desert.
Isn’t it amazing how much we can know about another human even across such an enormous chasm of time? If you’d asked me when I was 11 what I wanted to be, I’d have said archaeologist. .
A 64-year-old woman with a 4-cm red, brown shiny plaque on her right calf. She burned it on her boyfriend’s Harley Davidson nearly 40 years ago. She wonders where he is now.
A 58-year-old man with a 3-inch scar on his right wrist. He fell off his 6-year-old’s skimboard. ORIF.
A 40-year-old woman with bilateral mastectomy scars.
A 66-year-old with a lichenified nodule on his left forearm. When I shaved it off, a quarter inch spicule of glass came out. It was from a car accident in his first car, a Chevy Impala. He saved the piece of glass as a souvenir.
A fit 50-year-old with extensive scars on his feet and ankles. “Yeah, I went ‘whistling-in’ on a training jump,” he said. He was a retired Navy Seal and raconteur with quite a tale about the day his parachute malfunctioned. Some well placed live oak trees is why he’s around for his skin screening.
A classic, rope-like open-heart scar on the chest of a thin, young, healthy, flaxen-haired woman. Dissected aorta.
A 30-something woman dressed in a pants suit with razor-thin parallel scars on her volar forearms and proximal thighs. She asks if any laser could remove them.
A rotund, hard-living, bearded man with chest and upper-arm tattoos of flames and nudie girls now mixed with the striking face of an old woman and three little kids: His mom and grandkids. He shows me where the fourth grandkid will go and gives me a bear hug to thank me for the care when he leaves.
Attending to these details shifts us from autopilot to present. It keeps us involved, holding our attention even if it’s the 20th skin screening or diabetic foot exam of the day. And what a gift to share in the intimate details of another’s life.
Like examining the minute details of an ancient bone, dig for the history with curiosity, pity, humility. The perfect moment for asking might be when you stand with your #15 blade ready to introduce a new scar and become part of this human’s story forever.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
You can observe a lot by watching. – Yogi Berra
He was a fit man in his 40s. Thick legs. Maybe he was a long-distance walker? The bones of his right arm were more developed than his left – a right-handed thrower. His lower left fibula was fractured from a severely rolled ankle. He carried a walking stick that was glossy in the middle from where he gripped it with his left hand, dragging his bad left foot along. Dental cavities tell the story of his diet: honey, carobs, dates. Carbon 14 dating confirms that he lived during the Chalcolithic period, approximately 6,000 years ago. He was likely a shepherd in the Judean Desert.
Isn’t it amazing how much we can know about another human even across such an enormous chasm of time? If you’d asked me when I was 11 what I wanted to be, I’d have said archaeologist. .
A 64-year-old woman with a 4-cm red, brown shiny plaque on her right calf. She burned it on her boyfriend’s Harley Davidson nearly 40 years ago. She wonders where he is now.
A 58-year-old man with a 3-inch scar on his right wrist. He fell off his 6-year-old’s skimboard. ORIF.
A 40-year-old woman with bilateral mastectomy scars.
A 66-year-old with a lichenified nodule on his left forearm. When I shaved it off, a quarter inch spicule of glass came out. It was from a car accident in his first car, a Chevy Impala. He saved the piece of glass as a souvenir.
A fit 50-year-old with extensive scars on his feet and ankles. “Yeah, I went ‘whistling-in’ on a training jump,” he said. He was a retired Navy Seal and raconteur with quite a tale about the day his parachute malfunctioned. Some well placed live oak trees is why he’s around for his skin screening.
A classic, rope-like open-heart scar on the chest of a thin, young, healthy, flaxen-haired woman. Dissected aorta.
A 30-something woman dressed in a pants suit with razor-thin parallel scars on her volar forearms and proximal thighs. She asks if any laser could remove them.
A rotund, hard-living, bearded man with chest and upper-arm tattoos of flames and nudie girls now mixed with the striking face of an old woman and three little kids: His mom and grandkids. He shows me where the fourth grandkid will go and gives me a bear hug to thank me for the care when he leaves.
Attending to these details shifts us from autopilot to present. It keeps us involved, holding our attention even if it’s the 20th skin screening or diabetic foot exam of the day. And what a gift to share in the intimate details of another’s life.
Like examining the minute details of an ancient bone, dig for the history with curiosity, pity, humility. The perfect moment for asking might be when you stand with your #15 blade ready to introduce a new scar and become part of this human’s story forever.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
Does PREDICT accurately estimate breast cancer survival?
The PREDICT score does not seem to be particularly accurate when it comes to estimating overall survival (OS) in patients with HER2-positive early breast cancer who are treated with modern chemotherapy and anti-HER2 targeted therapies. This is the conclusion of an international study published in the journal npj Breast Cancer. The work was supervised by Matteo Lambertini, MD, PhD, an oncologist at the IRCCS San Martino Polyclinic Hospital in Genoa, Italy.
As the authors explain, “PREDICT is a publicly available online tool that helps to predict the individual prognosis of patients with early breast cancer and to show the impact of adjuvant treatments administered after breast cancer surgery.” The tool uses traditional clinical-pathological factors. The authors also point out that the original version of this tool was validated in several datasets of patients with breast cancer. In 2011, it was updated to include HER2 status.
The investigators noted that, although the use of PREDICT is recommended to aid decision-making in the adjuvant setting, its prognostic role in patients with HER2-positive early breast cancer who are treated with modern chemotherapy and anti-HER2 therapies – even trastuzumab-based ones – remains unclear.
Therefore, they decided to analyze PREDICT’s prognostic performance using data extracted from the ALTTO trial, the largest adjuvant study ever conducted in the field of HER2-positive early breast cancer. That trial “represented a unique opportunity to investigate the reliability and prognostic performance of PREDICT in women with HER2-positive disease,” according to the investigators. They went on to specify that ALTTO evaluated adjuvant lapatinib plus trastuzumab vs. trastuzumab alone in 8,381 patients – 2,794 of whom were included in their own analysis.
What the analysis found was that, overall, PREDICT underestimated 5-year OS by 6.7%. The observed 5-year OS was 94.7%, and the predicted 5-year OS was 88.0%.
The highest absolute differences were observed for patients with hormone receptor–negative disease, nodal involvement, and large tumor size (13.0%, 15.8%, and 15.3%, respectively),” they wrote. Furthermore, they reported that “the suboptimal performance of this prognostic tool was observed irrespective of type of anti-HER2 treatment, type of chemotherapy regimen, age of the patients at the time of diagnosis, central hormone receptor status, pathological nodal status, and pathological tumor size.”
To potentially explain the reasons for the underestimation of patients’ OS, the authors questioned whether the population used to validate PREDICT accurately mirrored the real-world population of patients with HER2-positive disease treated in the modern era with effective chemotherapy and anti-HER2 targeted therapies. “Moreover, the current standard of care for early breast cancer is even superior to the treatment received by many patients in the ALTTO study. … As such, the discordance between OS estimated by PREDICT and the current real-world OS is expected to be even higher. Therefore,” the researchers concluded, “our results suggest that the current version of PREDICT should be used with caution for prognostication in HER2-positive early breast cancer patients treated in the modern era with effective chemotherapy and anti-HER2 targeted therapies.”
A version of this article first appeared on Medscape.com. This article was translated from Univadis Italy.
The PREDICT score does not seem to be particularly accurate when it comes to estimating overall survival (OS) in patients with HER2-positive early breast cancer who are treated with modern chemotherapy and anti-HER2 targeted therapies. This is the conclusion of an international study published in the journal npj Breast Cancer. The work was supervised by Matteo Lambertini, MD, PhD, an oncologist at the IRCCS San Martino Polyclinic Hospital in Genoa, Italy.
As the authors explain, “PREDICT is a publicly available online tool that helps to predict the individual prognosis of patients with early breast cancer and to show the impact of adjuvant treatments administered after breast cancer surgery.” The tool uses traditional clinical-pathological factors. The authors also point out that the original version of this tool was validated in several datasets of patients with breast cancer. In 2011, it was updated to include HER2 status.
The investigators noted that, although the use of PREDICT is recommended to aid decision-making in the adjuvant setting, its prognostic role in patients with HER2-positive early breast cancer who are treated with modern chemotherapy and anti-HER2 therapies – even trastuzumab-based ones – remains unclear.
Therefore, they decided to analyze PREDICT’s prognostic performance using data extracted from the ALTTO trial, the largest adjuvant study ever conducted in the field of HER2-positive early breast cancer. That trial “represented a unique opportunity to investigate the reliability and prognostic performance of PREDICT in women with HER2-positive disease,” according to the investigators. They went on to specify that ALTTO evaluated adjuvant lapatinib plus trastuzumab vs. trastuzumab alone in 8,381 patients – 2,794 of whom were included in their own analysis.
What the analysis found was that, overall, PREDICT underestimated 5-year OS by 6.7%. The observed 5-year OS was 94.7%, and the predicted 5-year OS was 88.0%.
The highest absolute differences were observed for patients with hormone receptor–negative disease, nodal involvement, and large tumor size (13.0%, 15.8%, and 15.3%, respectively),” they wrote. Furthermore, they reported that “the suboptimal performance of this prognostic tool was observed irrespective of type of anti-HER2 treatment, type of chemotherapy regimen, age of the patients at the time of diagnosis, central hormone receptor status, pathological nodal status, and pathological tumor size.”
To potentially explain the reasons for the underestimation of patients’ OS, the authors questioned whether the population used to validate PREDICT accurately mirrored the real-world population of patients with HER2-positive disease treated in the modern era with effective chemotherapy and anti-HER2 targeted therapies. “Moreover, the current standard of care for early breast cancer is even superior to the treatment received by many patients in the ALTTO study. … As such, the discordance between OS estimated by PREDICT and the current real-world OS is expected to be even higher. Therefore,” the researchers concluded, “our results suggest that the current version of PREDICT should be used with caution for prognostication in HER2-positive early breast cancer patients treated in the modern era with effective chemotherapy and anti-HER2 targeted therapies.”
A version of this article first appeared on Medscape.com. This article was translated from Univadis Italy.
The PREDICT score does not seem to be particularly accurate when it comes to estimating overall survival (OS) in patients with HER2-positive early breast cancer who are treated with modern chemotherapy and anti-HER2 targeted therapies. This is the conclusion of an international study published in the journal npj Breast Cancer. The work was supervised by Matteo Lambertini, MD, PhD, an oncologist at the IRCCS San Martino Polyclinic Hospital in Genoa, Italy.
As the authors explain, “PREDICT is a publicly available online tool that helps to predict the individual prognosis of patients with early breast cancer and to show the impact of adjuvant treatments administered after breast cancer surgery.” The tool uses traditional clinical-pathological factors. The authors also point out that the original version of this tool was validated in several datasets of patients with breast cancer. In 2011, it was updated to include HER2 status.
The investigators noted that, although the use of PREDICT is recommended to aid decision-making in the adjuvant setting, its prognostic role in patients with HER2-positive early breast cancer who are treated with modern chemotherapy and anti-HER2 therapies – even trastuzumab-based ones – remains unclear.
Therefore, they decided to analyze PREDICT’s prognostic performance using data extracted from the ALTTO trial, the largest adjuvant study ever conducted in the field of HER2-positive early breast cancer. That trial “represented a unique opportunity to investigate the reliability and prognostic performance of PREDICT in women with HER2-positive disease,” according to the investigators. They went on to specify that ALTTO evaluated adjuvant lapatinib plus trastuzumab vs. trastuzumab alone in 8,381 patients – 2,794 of whom were included in their own analysis.
What the analysis found was that, overall, PREDICT underestimated 5-year OS by 6.7%. The observed 5-year OS was 94.7%, and the predicted 5-year OS was 88.0%.
The highest absolute differences were observed for patients with hormone receptor–negative disease, nodal involvement, and large tumor size (13.0%, 15.8%, and 15.3%, respectively),” they wrote. Furthermore, they reported that “the suboptimal performance of this prognostic tool was observed irrespective of type of anti-HER2 treatment, type of chemotherapy regimen, age of the patients at the time of diagnosis, central hormone receptor status, pathological nodal status, and pathological tumor size.”
To potentially explain the reasons for the underestimation of patients’ OS, the authors questioned whether the population used to validate PREDICT accurately mirrored the real-world population of patients with HER2-positive disease treated in the modern era with effective chemotherapy and anti-HER2 targeted therapies. “Moreover, the current standard of care for early breast cancer is even superior to the treatment received by many patients in the ALTTO study. … As such, the discordance between OS estimated by PREDICT and the current real-world OS is expected to be even higher. Therefore,” the researchers concluded, “our results suggest that the current version of PREDICT should be used with caution for prognostication in HER2-positive early breast cancer patients treated in the modern era with effective chemotherapy and anti-HER2 targeted therapies.”
A version of this article first appeared on Medscape.com. This article was translated from Univadis Italy.
FROM NPJ BREAST CANCER
Review cautions against influencer-promoted hair-growth remedies
One day in 2020, Ronda S. Farah, MD, was spending some downtime from her dermatology practice scrolling through social media. When she opened TikTok, she came across something that piqued her interest: A popular content creator was promoting the supplement biotin as a way to grow hair. Dr. Farah was immediately alarmed, because not only was the evidence that biotin increases hair growth shoddy, but the FDA had also warned that biotin supplements may interfere with lab tests for troponin.
Dr. Farah was moved to action and made a brief TikTok stating that use of biotin does not result in hair growth for most patients, which quickly shot up to over half a million views. She was flooded with messages from influencers and people desperate for an answer to their hair growth questions.
From that point on, Dr. Farah was immersed in the world of hairfluencers, the social media personalities who promote hair care trends, which formed the basis of a review, published in the Journal of Cosmetic Dermatology that she conducted with her colleagues at the University of Minnesota, Minneapolis. .
They reviewed five treatments that represent some of the most frequently discussed hair-growth trends on social media: rosemary, onion juice, rice water, castor oil, and aloe vera. For each, they evaluated recommendations on how the treatments were applied, possible harmful effects to the user, claims that weren’t totally based on scientific evidence, and the theoretical mechanism of action. “Overall,” they concluded, “there is little to no literature supporting these social media trends for hair growth.”
Of the five, rosemary, applied to the scalp or hair, has perhaps the most significant research behind it, according to Dr. Farah and coauthors. Methods of applying rosemary described on social media included use of prepackaged oil, boiling fresh rosemary leaves, adding leaves to oils and spraying it on or massaging it on the scalp, applying it in the hair, or using it as a rinse. Dr. Farah noted that the literature supporting the use of rosemary for hair growth does not represent the most robust science; the studies had small sample sizes and used nonstandardized methods of measuring hair growth.
“It didn’t really meet rigorous, strong study methods that a board-certified dermatologist with their expertise would consider a really solid study,” she said.
For the remaining methods, there was little research to support their use for hair growth. A few, the authors pointed out, can cause scalp burns (aloe vera), damage to hair follicles (rice water), contact dermatitis (aloe vera, onion juice), and, in the case of castor oil, hair felting..
Dr. Farah thinks social media can be a great tool to reach patients, but that people should be wary of what kind of information they’re consuming “and need to be aware of who their hairfluencer is,” she said. And, as she and her coauthors wrote: “We call on dermatologists, as hair and scalp disease experts, to serve as authorities on ‘hairfluencer’ trends and appropriately counsel patients.”
The study was independently supported. Dr. Farah reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One day in 2020, Ronda S. Farah, MD, was spending some downtime from her dermatology practice scrolling through social media. When she opened TikTok, she came across something that piqued her interest: A popular content creator was promoting the supplement biotin as a way to grow hair. Dr. Farah was immediately alarmed, because not only was the evidence that biotin increases hair growth shoddy, but the FDA had also warned that biotin supplements may interfere with lab tests for troponin.
Dr. Farah was moved to action and made a brief TikTok stating that use of biotin does not result in hair growth for most patients, which quickly shot up to over half a million views. She was flooded with messages from influencers and people desperate for an answer to their hair growth questions.
From that point on, Dr. Farah was immersed in the world of hairfluencers, the social media personalities who promote hair care trends, which formed the basis of a review, published in the Journal of Cosmetic Dermatology that she conducted with her colleagues at the University of Minnesota, Minneapolis. .
They reviewed five treatments that represent some of the most frequently discussed hair-growth trends on social media: rosemary, onion juice, rice water, castor oil, and aloe vera. For each, they evaluated recommendations on how the treatments were applied, possible harmful effects to the user, claims that weren’t totally based on scientific evidence, and the theoretical mechanism of action. “Overall,” they concluded, “there is little to no literature supporting these social media trends for hair growth.”
Of the five, rosemary, applied to the scalp or hair, has perhaps the most significant research behind it, according to Dr. Farah and coauthors. Methods of applying rosemary described on social media included use of prepackaged oil, boiling fresh rosemary leaves, adding leaves to oils and spraying it on or massaging it on the scalp, applying it in the hair, or using it as a rinse. Dr. Farah noted that the literature supporting the use of rosemary for hair growth does not represent the most robust science; the studies had small sample sizes and used nonstandardized methods of measuring hair growth.
“It didn’t really meet rigorous, strong study methods that a board-certified dermatologist with their expertise would consider a really solid study,” she said.
For the remaining methods, there was little research to support their use for hair growth. A few, the authors pointed out, can cause scalp burns (aloe vera), damage to hair follicles (rice water), contact dermatitis (aloe vera, onion juice), and, in the case of castor oil, hair felting..
Dr. Farah thinks social media can be a great tool to reach patients, but that people should be wary of what kind of information they’re consuming “and need to be aware of who their hairfluencer is,” she said. And, as she and her coauthors wrote: “We call on dermatologists, as hair and scalp disease experts, to serve as authorities on ‘hairfluencer’ trends and appropriately counsel patients.”
The study was independently supported. Dr. Farah reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One day in 2020, Ronda S. Farah, MD, was spending some downtime from her dermatology practice scrolling through social media. When she opened TikTok, she came across something that piqued her interest: A popular content creator was promoting the supplement biotin as a way to grow hair. Dr. Farah was immediately alarmed, because not only was the evidence that biotin increases hair growth shoddy, but the FDA had also warned that biotin supplements may interfere with lab tests for troponin.
Dr. Farah was moved to action and made a brief TikTok stating that use of biotin does not result in hair growth for most patients, which quickly shot up to over half a million views. She was flooded with messages from influencers and people desperate for an answer to their hair growth questions.
From that point on, Dr. Farah was immersed in the world of hairfluencers, the social media personalities who promote hair care trends, which formed the basis of a review, published in the Journal of Cosmetic Dermatology that she conducted with her colleagues at the University of Minnesota, Minneapolis. .
They reviewed five treatments that represent some of the most frequently discussed hair-growth trends on social media: rosemary, onion juice, rice water, castor oil, and aloe vera. For each, they evaluated recommendations on how the treatments were applied, possible harmful effects to the user, claims that weren’t totally based on scientific evidence, and the theoretical mechanism of action. “Overall,” they concluded, “there is little to no literature supporting these social media trends for hair growth.”
Of the five, rosemary, applied to the scalp or hair, has perhaps the most significant research behind it, according to Dr. Farah and coauthors. Methods of applying rosemary described on social media included use of prepackaged oil, boiling fresh rosemary leaves, adding leaves to oils and spraying it on or massaging it on the scalp, applying it in the hair, or using it as a rinse. Dr. Farah noted that the literature supporting the use of rosemary for hair growth does not represent the most robust science; the studies had small sample sizes and used nonstandardized methods of measuring hair growth.
“It didn’t really meet rigorous, strong study methods that a board-certified dermatologist with their expertise would consider a really solid study,” she said.
For the remaining methods, there was little research to support their use for hair growth. A few, the authors pointed out, can cause scalp burns (aloe vera), damage to hair follicles (rice water), contact dermatitis (aloe vera, onion juice), and, in the case of castor oil, hair felting..
Dr. Farah thinks social media can be a great tool to reach patients, but that people should be wary of what kind of information they’re consuming “and need to be aware of who their hairfluencer is,” she said. And, as she and her coauthors wrote: “We call on dermatologists, as hair and scalp disease experts, to serve as authorities on ‘hairfluencer’ trends and appropriately counsel patients.”
The study was independently supported. Dr. Farah reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF COSMETIC DERMATOLOGY
No-shows
Of all the headaches inherent in a private medical practice, few are more frustrating than patients who make appointments and then fail to keep them.
– almost double the average for all medical offices.
The problem has become so pervasive that many physicians are now charging a fee for missed appointments. I have never been a fan of such fees for a variety of reasons, starting with the anger and bad will that they engender; but also, in my experience, they seldom accomplish their intended goal of changing the behavior.
That’s because fees imply some sort of conscious decision made by a patient to miss an appointment, but studies show that this is rarely the case. Some patients cite transportation issues or childcare obligations. One Canadian study found that nearly a quarter of patients who missed an appointment felt too sick to keep it. Another reason is lack of insurance coverage. Studies have shown that the no-show rate is far higher when the patient is paying out-of-pocket for the visit.
Patients who don’t show up for appointments tend to be younger and poorer, and live farther away from the office than those who attend consistently. Some patients may be unaware that they need to cancel, while others report that they don’t feel obliged to keep appointments because they feel disrespected by the system. One person posted on a medical forum, “Everyone’s time is valuable. When the doctor makes me wait, there are consequences too. Why are there two standards in the situation?”
The most common reason for missed appointments, however, according to multiple studies, is that patients simply forget that they have one. One reason for that is a lag between appointment and visit. Many dermatologists are booked well in advance; by the time the appointment arrives, some patients’ complaints will have resolved spontaneously, while other patients will have found another office willing to see them sooner.
Another big reason is the absence of a strong physician-patient relationship. Perhaps the patient sees a different doctor or physician assistant at each visit and doesn’t feel a particular bond with any of them. Some patients may perceive a lack of concern on the part of the physician. And others may suffer from poor communication; for example, patients frequently become frustrated that a chronic condition has not resolved, when it has not been clearly explained to them that such problems cannot be expected to resolve rapidly or completely.
Whatever the reasons, no-shows are an economic and medicolegal liability. It is worth the considerable effort it often takes to minimize them.
Research suggests that no-show rates can be reduced by providing more same-day or next-day appointments. One large-scale analysis of national data found that same-day appointments accounted for just 2% of no-shows, while appointments booked 15 days or more in advance accounted for nearly a third of them. Canadian studies have likewise found the risk of no-shows increases the further in advance clinics book patients.
Deal with simple forgetfulness by calling your patients the day before to remind them of their appointments. Reasonably priced phone software is available from a variety of vendors to automate this process. Or hire a teenager to do it after school each day.
Whenever possible, use cellphone numbers for reminder calls. Patients often aren’t home during the day, and many don’t listen to their messages when they come in. And patients who have moved will often have a new home phone number, but their cellphone number will be the same.
Decrease the wait for new appointments. Keep some slots open each week for new patients, who will often “shop around” for a faster appointment while they’re waiting for an appointment they already have elsewhere.
But above all, seek to maximize the strength of your physician-patient relationships. Try not to shuttle patients between different physicians or PAs, and make it clear that you are genuinely concerned about their health. Impress upon them the crucial role they play in their own care, which includes keeping all their appointments.
In our office, significant no-shows (for example, a patient with a melanoma who misses a follow-up visit) receive a phone call and a certified letter, and their records go into a special file for close follow-up by the nursing staff.
If you choose to go the missed-appointment-fee route, be sure to post notices in your office and on your website clearly delineating your policy. Emphasize that it is not a service fee, and cannot be billed to insurance.
All missed appointments should be documented in the patient’s record; it’s important clinical and medicolegal information. And habitual no-shows should be dismissed from your practice. You cannot afford them.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Of all the headaches inherent in a private medical practice, few are more frustrating than patients who make appointments and then fail to keep them.
– almost double the average for all medical offices.
The problem has become so pervasive that many physicians are now charging a fee for missed appointments. I have never been a fan of such fees for a variety of reasons, starting with the anger and bad will that they engender; but also, in my experience, they seldom accomplish their intended goal of changing the behavior.
That’s because fees imply some sort of conscious decision made by a patient to miss an appointment, but studies show that this is rarely the case. Some patients cite transportation issues or childcare obligations. One Canadian study found that nearly a quarter of patients who missed an appointment felt too sick to keep it. Another reason is lack of insurance coverage. Studies have shown that the no-show rate is far higher when the patient is paying out-of-pocket for the visit.
Patients who don’t show up for appointments tend to be younger and poorer, and live farther away from the office than those who attend consistently. Some patients may be unaware that they need to cancel, while others report that they don’t feel obliged to keep appointments because they feel disrespected by the system. One person posted on a medical forum, “Everyone’s time is valuable. When the doctor makes me wait, there are consequences too. Why are there two standards in the situation?”
The most common reason for missed appointments, however, according to multiple studies, is that patients simply forget that they have one. One reason for that is a lag between appointment and visit. Many dermatologists are booked well in advance; by the time the appointment arrives, some patients’ complaints will have resolved spontaneously, while other patients will have found another office willing to see them sooner.
Another big reason is the absence of a strong physician-patient relationship. Perhaps the patient sees a different doctor or physician assistant at each visit and doesn’t feel a particular bond with any of them. Some patients may perceive a lack of concern on the part of the physician. And others may suffer from poor communication; for example, patients frequently become frustrated that a chronic condition has not resolved, when it has not been clearly explained to them that such problems cannot be expected to resolve rapidly or completely.
Whatever the reasons, no-shows are an economic and medicolegal liability. It is worth the considerable effort it often takes to minimize them.
Research suggests that no-show rates can be reduced by providing more same-day or next-day appointments. One large-scale analysis of national data found that same-day appointments accounted for just 2% of no-shows, while appointments booked 15 days or more in advance accounted for nearly a third of them. Canadian studies have likewise found the risk of no-shows increases the further in advance clinics book patients.
Deal with simple forgetfulness by calling your patients the day before to remind them of their appointments. Reasonably priced phone software is available from a variety of vendors to automate this process. Or hire a teenager to do it after school each day.
Whenever possible, use cellphone numbers for reminder calls. Patients often aren’t home during the day, and many don’t listen to their messages when they come in. And patients who have moved will often have a new home phone number, but their cellphone number will be the same.
Decrease the wait for new appointments. Keep some slots open each week for new patients, who will often “shop around” for a faster appointment while they’re waiting for an appointment they already have elsewhere.
But above all, seek to maximize the strength of your physician-patient relationships. Try not to shuttle patients between different physicians or PAs, and make it clear that you are genuinely concerned about their health. Impress upon them the crucial role they play in their own care, which includes keeping all their appointments.
In our office, significant no-shows (for example, a patient with a melanoma who misses a follow-up visit) receive a phone call and a certified letter, and their records go into a special file for close follow-up by the nursing staff.
If you choose to go the missed-appointment-fee route, be sure to post notices in your office and on your website clearly delineating your policy. Emphasize that it is not a service fee, and cannot be billed to insurance.
All missed appointments should be documented in the patient’s record; it’s important clinical and medicolegal information. And habitual no-shows should be dismissed from your practice. You cannot afford them.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Of all the headaches inherent in a private medical practice, few are more frustrating than patients who make appointments and then fail to keep them.
– almost double the average for all medical offices.
The problem has become so pervasive that many physicians are now charging a fee for missed appointments. I have never been a fan of such fees for a variety of reasons, starting with the anger and bad will that they engender; but also, in my experience, they seldom accomplish their intended goal of changing the behavior.
That’s because fees imply some sort of conscious decision made by a patient to miss an appointment, but studies show that this is rarely the case. Some patients cite transportation issues or childcare obligations. One Canadian study found that nearly a quarter of patients who missed an appointment felt too sick to keep it. Another reason is lack of insurance coverage. Studies have shown that the no-show rate is far higher when the patient is paying out-of-pocket for the visit.
Patients who don’t show up for appointments tend to be younger and poorer, and live farther away from the office than those who attend consistently. Some patients may be unaware that they need to cancel, while others report that they don’t feel obliged to keep appointments because they feel disrespected by the system. One person posted on a medical forum, “Everyone’s time is valuable. When the doctor makes me wait, there are consequences too. Why are there two standards in the situation?”
The most common reason for missed appointments, however, according to multiple studies, is that patients simply forget that they have one. One reason for that is a lag between appointment and visit. Many dermatologists are booked well in advance; by the time the appointment arrives, some patients’ complaints will have resolved spontaneously, while other patients will have found another office willing to see them sooner.
Another big reason is the absence of a strong physician-patient relationship. Perhaps the patient sees a different doctor or physician assistant at each visit and doesn’t feel a particular bond with any of them. Some patients may perceive a lack of concern on the part of the physician. And others may suffer from poor communication; for example, patients frequently become frustrated that a chronic condition has not resolved, when it has not been clearly explained to them that such problems cannot be expected to resolve rapidly or completely.
Whatever the reasons, no-shows are an economic and medicolegal liability. It is worth the considerable effort it often takes to minimize them.
Research suggests that no-show rates can be reduced by providing more same-day or next-day appointments. One large-scale analysis of national data found that same-day appointments accounted for just 2% of no-shows, while appointments booked 15 days or more in advance accounted for nearly a third of them. Canadian studies have likewise found the risk of no-shows increases the further in advance clinics book patients.
Deal with simple forgetfulness by calling your patients the day before to remind them of their appointments. Reasonably priced phone software is available from a variety of vendors to automate this process. Or hire a teenager to do it after school each day.
Whenever possible, use cellphone numbers for reminder calls. Patients often aren’t home during the day, and many don’t listen to their messages when they come in. And patients who have moved will often have a new home phone number, but their cellphone number will be the same.
Decrease the wait for new appointments. Keep some slots open each week for new patients, who will often “shop around” for a faster appointment while they’re waiting for an appointment they already have elsewhere.
But above all, seek to maximize the strength of your physician-patient relationships. Try not to shuttle patients between different physicians or PAs, and make it clear that you are genuinely concerned about their health. Impress upon them the crucial role they play in their own care, which includes keeping all their appointments.
In our office, significant no-shows (for example, a patient with a melanoma who misses a follow-up visit) receive a phone call and a certified letter, and their records go into a special file for close follow-up by the nursing staff.
If you choose to go the missed-appointment-fee route, be sure to post notices in your office and on your website clearly delineating your policy. Emphasize that it is not a service fee, and cannot be billed to insurance.
All missed appointments should be documented in the patient’s record; it’s important clinical and medicolegal information. And habitual no-shows should be dismissed from your practice. You cannot afford them.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Dermatologists skeptical of calamine lotion TikTok trend
Though this may seem to work as a base layer for some people, dermatologists have concerns about this trend, particularly the risk of dryness.
As of Aug. 15, the #calaminelotion tag had more than 20.9 million views on TikTok, with hundreds of videos hailing the cream for its opaque pink tint and matte effect when used under foundation.
Calamine lotion has been used to treat itchy rashes, insect bites, and pain from chickenpox and poison ivy for years. It’s sold over the counter and is a common first-line treatment for skin discomfort that has been used for hundreds of years, says Doris Day, MD, a dermatologist who practices in New York City. It is also on the World Health Organization’s list of essential drugs, she points out in an interview.
“This is something that has been around for a long time. It’s recognized as a drug that has importance. So every now and then, I guess somebody comes across it” and says it’s a “new panacea” for something, “but it’s really not. It’s just an old-time simple product.”
Calamine lotion is made of ferric oxide and zinc oxide, which gives it its antiseptic and anti-itch properties, in addition to its characteristic pink color. Zinc oxide is also commonly used in mineral sunscreens, Dr. Day points out.
Although these ingredients are exceedingly safe with temporary, localized use, high concentrations and chronic use of calamine lotion can be irritating to the skin, says Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington.
At these high concentrations, calamine lotion can be drying, which may cause skin clumping and can be abrasive, says Dr. Sodha. She also cautions that the astringent properties of the zinc and the high pH may disrupt proteins on the skin, which breaks down the skin’s natural defenses. Using calamine lotion all over the face daily can “potentially damage your skin barrier to a point where you’re going to have to do a lot of extra work ... to bring it back,” says Dr. Sodha.
Dr. Day also worries about this trend resulting in dry skin among followers. Even in situations where using calamine lotion is appropriate, like treating poison ivy, its drying effects can sometimes irritate the skin.
And dry skin can be more than an aesthetic issue: It can lead to breaks in the skin, which can result in infections and scarring, she points out. Although this may not occur in someone with extremely oily skin, most people don’t have extremely oily skin, says Dr. Day, so this will be ineffective at best, and at worst, damaging.
If someone is looking for a good makeup base layer, Dr. Sodha recommends something that’s noncomedogenic and nonsensitizing, like silicon-based primers. “The great thing about these products is that they are noncomedogenic, so they won’t clog your pores. They’re synthetic, so they’re not going to cause some sort of allergy,” she says.
In general, both dermatologists warn their patients to be wary of the TikTok trends they see online, and they cautioned about possible effects with long term use of calamine lotion on the face, even if it appears to work with one-time use. “Consumers have to think about this like they do with any sort of product that they come across, just thinking about the long-term effects of something like this and how it works for their own skin,” says Dr. Sodha.
A version of this article first appeared on Medscape.com.
Though this may seem to work as a base layer for some people, dermatologists have concerns about this trend, particularly the risk of dryness.
As of Aug. 15, the #calaminelotion tag had more than 20.9 million views on TikTok, with hundreds of videos hailing the cream for its opaque pink tint and matte effect when used under foundation.
Calamine lotion has been used to treat itchy rashes, insect bites, and pain from chickenpox and poison ivy for years. It’s sold over the counter and is a common first-line treatment for skin discomfort that has been used for hundreds of years, says Doris Day, MD, a dermatologist who practices in New York City. It is also on the World Health Organization’s list of essential drugs, she points out in an interview.
“This is something that has been around for a long time. It’s recognized as a drug that has importance. So every now and then, I guess somebody comes across it” and says it’s a “new panacea” for something, “but it’s really not. It’s just an old-time simple product.”
Calamine lotion is made of ferric oxide and zinc oxide, which gives it its antiseptic and anti-itch properties, in addition to its characteristic pink color. Zinc oxide is also commonly used in mineral sunscreens, Dr. Day points out.
Although these ingredients are exceedingly safe with temporary, localized use, high concentrations and chronic use of calamine lotion can be irritating to the skin, says Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington.
At these high concentrations, calamine lotion can be drying, which may cause skin clumping and can be abrasive, says Dr. Sodha. She also cautions that the astringent properties of the zinc and the high pH may disrupt proteins on the skin, which breaks down the skin’s natural defenses. Using calamine lotion all over the face daily can “potentially damage your skin barrier to a point where you’re going to have to do a lot of extra work ... to bring it back,” says Dr. Sodha.
Dr. Day also worries about this trend resulting in dry skin among followers. Even in situations where using calamine lotion is appropriate, like treating poison ivy, its drying effects can sometimes irritate the skin.
And dry skin can be more than an aesthetic issue: It can lead to breaks in the skin, which can result in infections and scarring, she points out. Although this may not occur in someone with extremely oily skin, most people don’t have extremely oily skin, says Dr. Day, so this will be ineffective at best, and at worst, damaging.
If someone is looking for a good makeup base layer, Dr. Sodha recommends something that’s noncomedogenic and nonsensitizing, like silicon-based primers. “The great thing about these products is that they are noncomedogenic, so they won’t clog your pores. They’re synthetic, so they’re not going to cause some sort of allergy,” she says.
In general, both dermatologists warn their patients to be wary of the TikTok trends they see online, and they cautioned about possible effects with long term use of calamine lotion on the face, even if it appears to work with one-time use. “Consumers have to think about this like they do with any sort of product that they come across, just thinking about the long-term effects of something like this and how it works for their own skin,” says Dr. Sodha.
A version of this article first appeared on Medscape.com.
Though this may seem to work as a base layer for some people, dermatologists have concerns about this trend, particularly the risk of dryness.
As of Aug. 15, the #calaminelotion tag had more than 20.9 million views on TikTok, with hundreds of videos hailing the cream for its opaque pink tint and matte effect when used under foundation.
Calamine lotion has been used to treat itchy rashes, insect bites, and pain from chickenpox and poison ivy for years. It’s sold over the counter and is a common first-line treatment for skin discomfort that has been used for hundreds of years, says Doris Day, MD, a dermatologist who practices in New York City. It is also on the World Health Organization’s list of essential drugs, she points out in an interview.
“This is something that has been around for a long time. It’s recognized as a drug that has importance. So every now and then, I guess somebody comes across it” and says it’s a “new panacea” for something, “but it’s really not. It’s just an old-time simple product.”
Calamine lotion is made of ferric oxide and zinc oxide, which gives it its antiseptic and anti-itch properties, in addition to its characteristic pink color. Zinc oxide is also commonly used in mineral sunscreens, Dr. Day points out.
Although these ingredients are exceedingly safe with temporary, localized use, high concentrations and chronic use of calamine lotion can be irritating to the skin, says Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington.
At these high concentrations, calamine lotion can be drying, which may cause skin clumping and can be abrasive, says Dr. Sodha. She also cautions that the astringent properties of the zinc and the high pH may disrupt proteins on the skin, which breaks down the skin’s natural defenses. Using calamine lotion all over the face daily can “potentially damage your skin barrier to a point where you’re going to have to do a lot of extra work ... to bring it back,” says Dr. Sodha.
Dr. Day also worries about this trend resulting in dry skin among followers. Even in situations where using calamine lotion is appropriate, like treating poison ivy, its drying effects can sometimes irritate the skin.
And dry skin can be more than an aesthetic issue: It can lead to breaks in the skin, which can result in infections and scarring, she points out. Although this may not occur in someone with extremely oily skin, most people don’t have extremely oily skin, says Dr. Day, so this will be ineffective at best, and at worst, damaging.
If someone is looking for a good makeup base layer, Dr. Sodha recommends something that’s noncomedogenic and nonsensitizing, like silicon-based primers. “The great thing about these products is that they are noncomedogenic, so they won’t clog your pores. They’re synthetic, so they’re not going to cause some sort of allergy,” she says.
In general, both dermatologists warn their patients to be wary of the TikTok trends they see online, and they cautioned about possible effects with long term use of calamine lotion on the face, even if it appears to work with one-time use. “Consumers have to think about this like they do with any sort of product that they come across, just thinking about the long-term effects of something like this and how it works for their own skin,” says Dr. Sodha.
A version of this article first appeared on Medscape.com.
Patients who won’t pay: What’s your recourse?
Owing to the pandemic, job loss, and the possible loss of health insurance, patients have had more difficulty managing copays, coinsurance, and deductibles, not to mention other out-of-pocket health care charges.
“Many of our patients have lost their jobs or have had their hours cut back, and as a result, they are struggling to make ends meet,” said Ahmad Chaudhry, MD, a cardiothoracic surgeon in Lexington, Ky. “However, we cannot continue to provide care if our patients do not pay their bills.”
This news organization asked physicians what they do when their patients don’t pay. About 43% said that they continue to treat them and develop a payment plan; 13% send their bill to collections; 12% continue their care and write off their balance, and 25% choose other actions. Only 8% of physicians drop patients if they don’t pay.
Because you need to pay your own bills, what can you do about nonpaying patients?
Start with price transparency
In the past, patients never knew what their lab work or a chest EKG would cost because it wasn’t listed anywhere, and it was usually more than expected. Because of new legislation concerning health care price transparency, hospitals, health plans, and insurers must pony up with the actual fees, making them transparent to patients. Physician practices should follow suit and keep prices transparent too. Patients are more likely to pay their bills when prepared for the expense.
Patients with insurance often don’t know what they’ll be paying for their visit or their tests because they don’t know how much insurance will cover and what will be left for them to pay. Also, they may not know if they’ve met their deductible yet so they’re unsure whether insurance will even kick in. And patients without insurance still need to know what their costs will be upfront.
According to 10 insights from the Primary Care Consumer Choice Survey, 74% of health care consumers were willing to pay a $50 out-of-pocket charge to know the cost of their primary care visit.
Provide payment plans
Many patients have always needed payment plans. It’s one thing to post a sign at check-in telling patients that all monies are due at the time of service, but it’s another reality for a patient who can’t fork over the $250 charge they just unexpectedly spent in your office.
Discover Financial Services recently ran a survey, with results presented in the press release Americans are Delaying Non-Emergency Medical Care in Higher Numbers than Last Year, and found that many Americans with medical debt are delaying nonemergency medical care. For example, they put off seeing a specialist (52%), seeing a doctor for sickness (41%), and undergoing treatment plans recommended by their doctor (31%).
Turning an account over to collections should be a last resort. In addition, agencies typically charge 30%-40% of the total collected off the top.
Though collecting that amount is better than nothing, using a collection agency may have unexpected consequences. For instance, you’re trusting the agency you hire to collect to represent you and act on your practice’s behalf. If they’re rude or their tactics are harsh in the eyes of the patient or their relatives, it’s your reputation that is on the line.
Rather than use a collection agency, you could collect the payments yourself. When a patient fails to pay within about 3 months, begin mailing statements from the office, followed by firm but generous phone calls trying to collect. Industry estimates put the average cost of sending an invoice, including staff labor, printing, and postage, at about $35 per mailer. Some practices combat the added costs by offering a 20% prompt-pay discount. Offering payment plans is another option that helps garner eventual payment. Plus, practices should direct patients to third-party lenders such as CareCredit for larger bills.
On occasion, some small practices may allow a swap, such as allowing a patient to provide a service such as plumbing, electrical, or painting in exchange for working off the bill. Though it’s not ideal when it comes to finances, you may find it can work in a pinch for a cash-strapped patient. Make sure to keep records of what bills the patient’s work goes toward.
It often helps to incentivize your billing staff to follow up regularly, with various suggestions and tactics, to get patients to pay their bills. The incentive amount you offer will probably be less than if you had to use a collection agency.
Have a payment policy
Because your practice’s primary job is caring for patients’ physical and emotional needs, payment collection without coming off as insensitive can be tricky. “We understand these are difficult times for everyone, and we are doing our best to work with our patients,” said Dr. Chaudhry. Having a written payment policy can help build the bridge. A policy lets patients know what they can expect and can help prevent surprises over what occurs in the event of nonpayment. Your written policy should include:
- When payment is due.
- How the practice handles copays and deductibles.
- What forms of payment are accepted.
- Your policy regarding nonpayment.
Why patients don’t pay
A 2021 Healthcare Consumer Experience Study from Cedar found that medical bills are a source of anxiety and frustration for most patients, affecting their financial experience. More than half of the respondents said that paying a medical bill is stressful. Complicating matters, many health care practices rely on outdated payment systems, which may not provide patients with a clear view of what they owe and how to pay it.
The study found that 53% of respondents find understanding their plan’s coverage and benefits stressful, and 37% of patients won’t pay their bill if they can’t understand it.
People may think the patient is trying to get out of paying, which, of course, is sometimes true, but most of the time they want to pay, concluded the study. Most patients need a better explanation, communication, and accurate accounting of their out-of-pocket costs.
What can doctors do?
If you’re a physician who regularly sees patients who have problems paying their bills, you can take a few steps to minimize the financial impact on your practice:
- Bill the patient’s insurance directly to ensure you receive at least partial payment.
- Keep adequate records of services in case you need to pursue legal action.
- “Be understanding and flexible when it comes to payment arrangements, as this can often be the difference between getting paid and not getting paid at all,” said Dr. Chaudhry.
Distance yourself
When discussing payment policies, physicians should try to distance themselves from the actual collection process as much as possible. Well-meaning physicians often tell patients things like they can “figure something out “ financially or “work them in” during a scheduling conflict, but that often undermines the authority and credibility of the practice’s office staff. Plus, it teaches patients they can get their way if they work on the doctor’s soft spot – something you don’t want to encourage.
By following some of these measures, you can help ensure that your practice continues to thrive despite the challenges posed by nonpaying patients.
A version of this article first appeared on Medscape.com.
Owing to the pandemic, job loss, and the possible loss of health insurance, patients have had more difficulty managing copays, coinsurance, and deductibles, not to mention other out-of-pocket health care charges.
“Many of our patients have lost their jobs or have had their hours cut back, and as a result, they are struggling to make ends meet,” said Ahmad Chaudhry, MD, a cardiothoracic surgeon in Lexington, Ky. “However, we cannot continue to provide care if our patients do not pay their bills.”
This news organization asked physicians what they do when their patients don’t pay. About 43% said that they continue to treat them and develop a payment plan; 13% send their bill to collections; 12% continue their care and write off their balance, and 25% choose other actions. Only 8% of physicians drop patients if they don’t pay.
Because you need to pay your own bills, what can you do about nonpaying patients?
Start with price transparency
In the past, patients never knew what their lab work or a chest EKG would cost because it wasn’t listed anywhere, and it was usually more than expected. Because of new legislation concerning health care price transparency, hospitals, health plans, and insurers must pony up with the actual fees, making them transparent to patients. Physician practices should follow suit and keep prices transparent too. Patients are more likely to pay their bills when prepared for the expense.
Patients with insurance often don’t know what they’ll be paying for their visit or their tests because they don’t know how much insurance will cover and what will be left for them to pay. Also, they may not know if they’ve met their deductible yet so they’re unsure whether insurance will even kick in. And patients without insurance still need to know what their costs will be upfront.
According to 10 insights from the Primary Care Consumer Choice Survey, 74% of health care consumers were willing to pay a $50 out-of-pocket charge to know the cost of their primary care visit.
Provide payment plans
Many patients have always needed payment plans. It’s one thing to post a sign at check-in telling patients that all monies are due at the time of service, but it’s another reality for a patient who can’t fork over the $250 charge they just unexpectedly spent in your office.
Discover Financial Services recently ran a survey, with results presented in the press release Americans are Delaying Non-Emergency Medical Care in Higher Numbers than Last Year, and found that many Americans with medical debt are delaying nonemergency medical care. For example, they put off seeing a specialist (52%), seeing a doctor for sickness (41%), and undergoing treatment plans recommended by their doctor (31%).
Turning an account over to collections should be a last resort. In addition, agencies typically charge 30%-40% of the total collected off the top.
Though collecting that amount is better than nothing, using a collection agency may have unexpected consequences. For instance, you’re trusting the agency you hire to collect to represent you and act on your practice’s behalf. If they’re rude or their tactics are harsh in the eyes of the patient or their relatives, it’s your reputation that is on the line.
Rather than use a collection agency, you could collect the payments yourself. When a patient fails to pay within about 3 months, begin mailing statements from the office, followed by firm but generous phone calls trying to collect. Industry estimates put the average cost of sending an invoice, including staff labor, printing, and postage, at about $35 per mailer. Some practices combat the added costs by offering a 20% prompt-pay discount. Offering payment plans is another option that helps garner eventual payment. Plus, practices should direct patients to third-party lenders such as CareCredit for larger bills.
On occasion, some small practices may allow a swap, such as allowing a patient to provide a service such as plumbing, electrical, or painting in exchange for working off the bill. Though it’s not ideal when it comes to finances, you may find it can work in a pinch for a cash-strapped patient. Make sure to keep records of what bills the patient’s work goes toward.
It often helps to incentivize your billing staff to follow up regularly, with various suggestions and tactics, to get patients to pay their bills. The incentive amount you offer will probably be less than if you had to use a collection agency.
Have a payment policy
Because your practice’s primary job is caring for patients’ physical and emotional needs, payment collection without coming off as insensitive can be tricky. “We understand these are difficult times for everyone, and we are doing our best to work with our patients,” said Dr. Chaudhry. Having a written payment policy can help build the bridge. A policy lets patients know what they can expect and can help prevent surprises over what occurs in the event of nonpayment. Your written policy should include:
- When payment is due.
- How the practice handles copays and deductibles.
- What forms of payment are accepted.
- Your policy regarding nonpayment.
Why patients don’t pay
A 2021 Healthcare Consumer Experience Study from Cedar found that medical bills are a source of anxiety and frustration for most patients, affecting their financial experience. More than half of the respondents said that paying a medical bill is stressful. Complicating matters, many health care practices rely on outdated payment systems, which may not provide patients with a clear view of what they owe and how to pay it.
The study found that 53% of respondents find understanding their plan’s coverage and benefits stressful, and 37% of patients won’t pay their bill if they can’t understand it.
People may think the patient is trying to get out of paying, which, of course, is sometimes true, but most of the time they want to pay, concluded the study. Most patients need a better explanation, communication, and accurate accounting of their out-of-pocket costs.
What can doctors do?
If you’re a physician who regularly sees patients who have problems paying their bills, you can take a few steps to minimize the financial impact on your practice:
- Bill the patient’s insurance directly to ensure you receive at least partial payment.
- Keep adequate records of services in case you need to pursue legal action.
- “Be understanding and flexible when it comes to payment arrangements, as this can often be the difference between getting paid and not getting paid at all,” said Dr. Chaudhry.
Distance yourself
When discussing payment policies, physicians should try to distance themselves from the actual collection process as much as possible. Well-meaning physicians often tell patients things like they can “figure something out “ financially or “work them in” during a scheduling conflict, but that often undermines the authority and credibility of the practice’s office staff. Plus, it teaches patients they can get their way if they work on the doctor’s soft spot – something you don’t want to encourage.
By following some of these measures, you can help ensure that your practice continues to thrive despite the challenges posed by nonpaying patients.
A version of this article first appeared on Medscape.com.
Owing to the pandemic, job loss, and the possible loss of health insurance, patients have had more difficulty managing copays, coinsurance, and deductibles, not to mention other out-of-pocket health care charges.
“Many of our patients have lost their jobs or have had their hours cut back, and as a result, they are struggling to make ends meet,” said Ahmad Chaudhry, MD, a cardiothoracic surgeon in Lexington, Ky. “However, we cannot continue to provide care if our patients do not pay their bills.”
This news organization asked physicians what they do when their patients don’t pay. About 43% said that they continue to treat them and develop a payment plan; 13% send their bill to collections; 12% continue their care and write off their balance, and 25% choose other actions. Only 8% of physicians drop patients if they don’t pay.
Because you need to pay your own bills, what can you do about nonpaying patients?
Start with price transparency
In the past, patients never knew what their lab work or a chest EKG would cost because it wasn’t listed anywhere, and it was usually more than expected. Because of new legislation concerning health care price transparency, hospitals, health plans, and insurers must pony up with the actual fees, making them transparent to patients. Physician practices should follow suit and keep prices transparent too. Patients are more likely to pay their bills when prepared for the expense.
Patients with insurance often don’t know what they’ll be paying for their visit or their tests because they don’t know how much insurance will cover and what will be left for them to pay. Also, they may not know if they’ve met their deductible yet so they’re unsure whether insurance will even kick in. And patients without insurance still need to know what their costs will be upfront.
According to 10 insights from the Primary Care Consumer Choice Survey, 74% of health care consumers were willing to pay a $50 out-of-pocket charge to know the cost of their primary care visit.
Provide payment plans
Many patients have always needed payment plans. It’s one thing to post a sign at check-in telling patients that all monies are due at the time of service, but it’s another reality for a patient who can’t fork over the $250 charge they just unexpectedly spent in your office.
Discover Financial Services recently ran a survey, with results presented in the press release Americans are Delaying Non-Emergency Medical Care in Higher Numbers than Last Year, and found that many Americans with medical debt are delaying nonemergency medical care. For example, they put off seeing a specialist (52%), seeing a doctor for sickness (41%), and undergoing treatment plans recommended by their doctor (31%).
Turning an account over to collections should be a last resort. In addition, agencies typically charge 30%-40% of the total collected off the top.
Though collecting that amount is better than nothing, using a collection agency may have unexpected consequences. For instance, you’re trusting the agency you hire to collect to represent you and act on your practice’s behalf. If they’re rude or their tactics are harsh in the eyes of the patient or their relatives, it’s your reputation that is on the line.
Rather than use a collection agency, you could collect the payments yourself. When a patient fails to pay within about 3 months, begin mailing statements from the office, followed by firm but generous phone calls trying to collect. Industry estimates put the average cost of sending an invoice, including staff labor, printing, and postage, at about $35 per mailer. Some practices combat the added costs by offering a 20% prompt-pay discount. Offering payment plans is another option that helps garner eventual payment. Plus, practices should direct patients to third-party lenders such as CareCredit for larger bills.
On occasion, some small practices may allow a swap, such as allowing a patient to provide a service such as plumbing, electrical, or painting in exchange for working off the bill. Though it’s not ideal when it comes to finances, you may find it can work in a pinch for a cash-strapped patient. Make sure to keep records of what bills the patient’s work goes toward.
It often helps to incentivize your billing staff to follow up regularly, with various suggestions and tactics, to get patients to pay their bills. The incentive amount you offer will probably be less than if you had to use a collection agency.
Have a payment policy
Because your practice’s primary job is caring for patients’ physical and emotional needs, payment collection without coming off as insensitive can be tricky. “We understand these are difficult times for everyone, and we are doing our best to work with our patients,” said Dr. Chaudhry. Having a written payment policy can help build the bridge. A policy lets patients know what they can expect and can help prevent surprises over what occurs in the event of nonpayment. Your written policy should include:
- When payment is due.
- How the practice handles copays and deductibles.
- What forms of payment are accepted.
- Your policy regarding nonpayment.
Why patients don’t pay
A 2021 Healthcare Consumer Experience Study from Cedar found that medical bills are a source of anxiety and frustration for most patients, affecting their financial experience. More than half of the respondents said that paying a medical bill is stressful. Complicating matters, many health care practices rely on outdated payment systems, which may not provide patients with a clear view of what they owe and how to pay it.
The study found that 53% of respondents find understanding their plan’s coverage and benefits stressful, and 37% of patients won’t pay their bill if they can’t understand it.
People may think the patient is trying to get out of paying, which, of course, is sometimes true, but most of the time they want to pay, concluded the study. Most patients need a better explanation, communication, and accurate accounting of their out-of-pocket costs.
What can doctors do?
If you’re a physician who regularly sees patients who have problems paying their bills, you can take a few steps to minimize the financial impact on your practice:
- Bill the patient’s insurance directly to ensure you receive at least partial payment.
- Keep adequate records of services in case you need to pursue legal action.
- “Be understanding and flexible when it comes to payment arrangements, as this can often be the difference between getting paid and not getting paid at all,” said Dr. Chaudhry.
Distance yourself
When discussing payment policies, physicians should try to distance themselves from the actual collection process as much as possible. Well-meaning physicians often tell patients things like they can “figure something out “ financially or “work them in” during a scheduling conflict, but that often undermines the authority and credibility of the practice’s office staff. Plus, it teaches patients they can get their way if they work on the doctor’s soft spot – something you don’t want to encourage.
By following some of these measures, you can help ensure that your practice continues to thrive despite the challenges posed by nonpaying patients.
A version of this article first appeared on Medscape.com.
A 9-year-old girl was evaluated for a week-long history of rash on the feet
A complete body examination failed to reveal any other lesions suggestive of a fungal infection. A blood count and urinalysis were within normal limits. She had no lymphadenopathy or hepatosplenomegaly. She was diagnosed with cutaneous larva migrans (CLM) given the clinical appearance of the lesions and the recent travel history.
CLM is a zoonotic infection caused by several hookworms such as Ancylostoma braziliense, Ancylostoma caninum, and Uncinaria stenocephala, as well as human hookworms such as Ancylostoma duodenale and Necator americanus. The hookworms can be present in contaminated soils and sandy beaches on the coastal regions of South America, the Caribbean, the Southeastern United States, Southeast Asia, and Africa.1-5
It is a common disease in the tourist population visiting tropical countries because of exposure to the hookworms in the soil without use of proper foot protection.
The clinical features are of an erythematous linear serpiginous plaque that is pruritic and can progress from millimeters to centimeters in size within a few days to weeks. Vesicles and multiple tracks can also be seen. The most common locations are the feet, buttocks, and thighs.
The larvae in the soil come from eggs excreted in the feces of infected cats and dogs. The infection is caused by direct contact of the larvae with the stratum corneum of the skin creating a burrow and an inflammatory response that will cause erythema, edema, track formation, and pruritus.
Diagnosis is made clinically. Rarely, a skin biopsy is warranted. The differential diagnosis includes tinea pedis, granuloma annulare, larva currens, contact dermatitis, and herpes zoster.
Tinea pedis is a fungal infection of the skin of the feet, commonly localized on the web spaces. The risk factors are a hot and humid environment, prolonged wear of occlusive footwear, excess sweating, and prolonged exposure to water.6 Diagnosis is confirmed by microscopic evaluation of skin scrapings with potassium hydroxide or a fungal culture. The infection is treated with topical antifungal creams and, in severe cases, systemic antifungals. Granuloma annulare is a benign chronic skin condition that presents with annular-shaped lesions. Its etiology is unknown. The lesions may be asymptomatic or mildly pruritic. Localized granuloma annulare typically presents as reddish-brown papules or plaques on the fingers, hands, elbows, dorsal feet, or ankles. The feature distinguishing granuloma annulare from other annular lesions is its absence of scale.
Allergic contact dermatitis is caused by skin exposure to an allergen and a secondary inflammatory response to this material on the skin causing inflammation, vesiculation, and pruritus. Lesions are treated with topical corticosteroids and avoidance of the allergen.
Herpes zoster is caused by a viral infection of the latent varicella-zoster virus. Its reactivation causes the presence of vesicles with an erythematous base that have a dermatomal distribution. The lesions are usually tender. Treatment is recommended to be started within 72 hours of the eruption with antivirals such as acyclovir or valacyclovir.
Cutaneous larva currens is caused by the cutaneous infection with Strongyloides stercoralis. In comparison with CLM, the lesions progress faster, at up to a centimeter within hours.
CLM is usually self-limited. If the patient has multiple lesions or more severe disease, oral albendazole or ivermectin can be prescribed. Other treatments, though not preferred, include freezing and topical thiabendazole solutions.
As our patient had several lesions, oral ivermectin was chosen as treatment and the lesions cleared within a week. Also, she was recommended to always wear shoes when walking on the beach.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Dr. Valderrama is a pediatric dermatologist at Fundación Cardioinfantil, Bogota, Colombia.
References
1. Feldmeier H and Schuster A. Eur J Clin Microbiol Infect Dis. 2012 Jun;31(6):915-8.
2. Jacobson CC and Abel EA. J Am Acad Dermatol. 2007 Jun;56(6):1026-43.
3. Kincaid L et al. Travel Med Infect Dis. 2015 Sep-Oct;13(5):382-7.
4. Gill N et al. Adv Skin Wound Care. 2020 Jul;33(7):356-9.
5. Rodenas-Herranz T et al. Dermatol Ther. 2020 May;33(3):e13316.
6. Pramod K et al. In: StatPearls [Internet]. Treasure Island (Fla): StatPearls Publishing; 2022 Jan.
A complete body examination failed to reveal any other lesions suggestive of a fungal infection. A blood count and urinalysis were within normal limits. She had no lymphadenopathy or hepatosplenomegaly. She was diagnosed with cutaneous larva migrans (CLM) given the clinical appearance of the lesions and the recent travel history.
CLM is a zoonotic infection caused by several hookworms such as Ancylostoma braziliense, Ancylostoma caninum, and Uncinaria stenocephala, as well as human hookworms such as Ancylostoma duodenale and Necator americanus. The hookworms can be present in contaminated soils and sandy beaches on the coastal regions of South America, the Caribbean, the Southeastern United States, Southeast Asia, and Africa.1-5
It is a common disease in the tourist population visiting tropical countries because of exposure to the hookworms in the soil without use of proper foot protection.
The clinical features are of an erythematous linear serpiginous plaque that is pruritic and can progress from millimeters to centimeters in size within a few days to weeks. Vesicles and multiple tracks can also be seen. The most common locations are the feet, buttocks, and thighs.
The larvae in the soil come from eggs excreted in the feces of infected cats and dogs. The infection is caused by direct contact of the larvae with the stratum corneum of the skin creating a burrow and an inflammatory response that will cause erythema, edema, track formation, and pruritus.
Diagnosis is made clinically. Rarely, a skin biopsy is warranted. The differential diagnosis includes tinea pedis, granuloma annulare, larva currens, contact dermatitis, and herpes zoster.
Tinea pedis is a fungal infection of the skin of the feet, commonly localized on the web spaces. The risk factors are a hot and humid environment, prolonged wear of occlusive footwear, excess sweating, and prolonged exposure to water.6 Diagnosis is confirmed by microscopic evaluation of skin scrapings with potassium hydroxide or a fungal culture. The infection is treated with topical antifungal creams and, in severe cases, systemic antifungals. Granuloma annulare is a benign chronic skin condition that presents with annular-shaped lesions. Its etiology is unknown. The lesions may be asymptomatic or mildly pruritic. Localized granuloma annulare typically presents as reddish-brown papules or plaques on the fingers, hands, elbows, dorsal feet, or ankles. The feature distinguishing granuloma annulare from other annular lesions is its absence of scale.
Allergic contact dermatitis is caused by skin exposure to an allergen and a secondary inflammatory response to this material on the skin causing inflammation, vesiculation, and pruritus. Lesions are treated with topical corticosteroids and avoidance of the allergen.
Herpes zoster is caused by a viral infection of the latent varicella-zoster virus. Its reactivation causes the presence of vesicles with an erythematous base that have a dermatomal distribution. The lesions are usually tender. Treatment is recommended to be started within 72 hours of the eruption with antivirals such as acyclovir or valacyclovir.
Cutaneous larva currens is caused by the cutaneous infection with Strongyloides stercoralis. In comparison with CLM, the lesions progress faster, at up to a centimeter within hours.
CLM is usually self-limited. If the patient has multiple lesions or more severe disease, oral albendazole or ivermectin can be prescribed. Other treatments, though not preferred, include freezing and topical thiabendazole solutions.
As our patient had several lesions, oral ivermectin was chosen as treatment and the lesions cleared within a week. Also, she was recommended to always wear shoes when walking on the beach.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Dr. Valderrama is a pediatric dermatologist at Fundación Cardioinfantil, Bogota, Colombia.
References
1. Feldmeier H and Schuster A. Eur J Clin Microbiol Infect Dis. 2012 Jun;31(6):915-8.
2. Jacobson CC and Abel EA. J Am Acad Dermatol. 2007 Jun;56(6):1026-43.
3. Kincaid L et al. Travel Med Infect Dis. 2015 Sep-Oct;13(5):382-7.
4. Gill N et al. Adv Skin Wound Care. 2020 Jul;33(7):356-9.
5. Rodenas-Herranz T et al. Dermatol Ther. 2020 May;33(3):e13316.
6. Pramod K et al. In: StatPearls [Internet]. Treasure Island (Fla): StatPearls Publishing; 2022 Jan.
A complete body examination failed to reveal any other lesions suggestive of a fungal infection. A blood count and urinalysis were within normal limits. She had no lymphadenopathy or hepatosplenomegaly. She was diagnosed with cutaneous larva migrans (CLM) given the clinical appearance of the lesions and the recent travel history.
CLM is a zoonotic infection caused by several hookworms such as Ancylostoma braziliense, Ancylostoma caninum, and Uncinaria stenocephala, as well as human hookworms such as Ancylostoma duodenale and Necator americanus. The hookworms can be present in contaminated soils and sandy beaches on the coastal regions of South America, the Caribbean, the Southeastern United States, Southeast Asia, and Africa.1-5
It is a common disease in the tourist population visiting tropical countries because of exposure to the hookworms in the soil without use of proper foot protection.
The clinical features are of an erythematous linear serpiginous plaque that is pruritic and can progress from millimeters to centimeters in size within a few days to weeks. Vesicles and multiple tracks can also be seen. The most common locations are the feet, buttocks, and thighs.
The larvae in the soil come from eggs excreted in the feces of infected cats and dogs. The infection is caused by direct contact of the larvae with the stratum corneum of the skin creating a burrow and an inflammatory response that will cause erythema, edema, track formation, and pruritus.
Diagnosis is made clinically. Rarely, a skin biopsy is warranted. The differential diagnosis includes tinea pedis, granuloma annulare, larva currens, contact dermatitis, and herpes zoster.
Tinea pedis is a fungal infection of the skin of the feet, commonly localized on the web spaces. The risk factors are a hot and humid environment, prolonged wear of occlusive footwear, excess sweating, and prolonged exposure to water.6 Diagnosis is confirmed by microscopic evaluation of skin scrapings with potassium hydroxide or a fungal culture. The infection is treated with topical antifungal creams and, in severe cases, systemic antifungals. Granuloma annulare is a benign chronic skin condition that presents with annular-shaped lesions. Its etiology is unknown. The lesions may be asymptomatic or mildly pruritic. Localized granuloma annulare typically presents as reddish-brown papules or plaques on the fingers, hands, elbows, dorsal feet, or ankles. The feature distinguishing granuloma annulare from other annular lesions is its absence of scale.
Allergic contact dermatitis is caused by skin exposure to an allergen and a secondary inflammatory response to this material on the skin causing inflammation, vesiculation, and pruritus. Lesions are treated with topical corticosteroids and avoidance of the allergen.
Herpes zoster is caused by a viral infection of the latent varicella-zoster virus. Its reactivation causes the presence of vesicles with an erythematous base that have a dermatomal distribution. The lesions are usually tender. Treatment is recommended to be started within 72 hours of the eruption with antivirals such as acyclovir or valacyclovir.
Cutaneous larva currens is caused by the cutaneous infection with Strongyloides stercoralis. In comparison with CLM, the lesions progress faster, at up to a centimeter within hours.
CLM is usually self-limited. If the patient has multiple lesions or more severe disease, oral albendazole or ivermectin can be prescribed. Other treatments, though not preferred, include freezing and topical thiabendazole solutions.
As our patient had several lesions, oral ivermectin was chosen as treatment and the lesions cleared within a week. Also, she was recommended to always wear shoes when walking on the beach.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Dr. Valderrama is a pediatric dermatologist at Fundación Cardioinfantil, Bogota, Colombia.
References
1. Feldmeier H and Schuster A. Eur J Clin Microbiol Infect Dis. 2012 Jun;31(6):915-8.
2. Jacobson CC and Abel EA. J Am Acad Dermatol. 2007 Jun;56(6):1026-43.
3. Kincaid L et al. Travel Med Infect Dis. 2015 Sep-Oct;13(5):382-7.
4. Gill N et al. Adv Skin Wound Care. 2020 Jul;33(7):356-9.
5. Rodenas-Herranz T et al. Dermatol Ther. 2020 May;33(3):e13316.
6. Pramod K et al. In: StatPearls [Internet]. Treasure Island (Fla): StatPearls Publishing; 2022 Jan.
Her mother reported recent travel to a beachside city in Colombia. A review of systems was negative. She was not taking any other medications or vitamin supplements. There were no pets at home and no other affected family members. Physical exam was notable for an erythematous curvilinear plaque on the feet and a small vesicle.
Biosimilar-to-biosimilar switches deemed safe and effective, systematic review reveals
Switching from one biosimilar medication to another is safe and effective, a new systematic review indicates, even though this clinical practice is not governed by current health authority regulations or guidance.
“No reduction in effectiveness or increase in adverse events was detected in biosimilar-to-biosimilar switching studies conducted to date,” the review’s authors noted in their study, published online in BioDrugs.
“The possibility of multiple switches between biosimilars of the same reference biologic is already a reality, and these types of switches are expected to become more common in the future. ... Although it is not covered by current health authority regulations or guidance,” added the authors, led by Hillel P. Cohen, PhD, executive director of scientific affairs at Sandoz, a division of Novartis.
The researchers searched electronic databases through December 2021 and found 23 observational studies that met their search criteria, of which 13 were published in peer-reviewed journals; the remainder appeared in abstract form. The studies totaled 3,657 patients. The researchers did not identify any randomized clinical trials.
“The studies were heterogeneous in size, design, and endpoints, providing data on safety, effectiveness, immunogenicity, pharmacokinetics, patient retention, patient and physician perceptions, and drug-use patterns,” the authors wrote.
The authors found that the majority of studies evaluated switches between biosimilars of infliximab, but they also identified switches between biosimilars of adalimumab, etanercept, and rituximab.
“Some health care providers are hesitant to switch patients from one biosimilar to another biosimilar because of a perceived lack of clinical data on such switches,” Dr. Cohen said in an interview.
The review’s findings – that there were no clinically relevant differences when switching patients from one biosimilar to another – are consistent with the science, Dr. Cohen said. “Physicians should have confidence that the data demonstrate that safety and effectiveness are not impacted if patients switch from one biosimilar to another biosimilar of the same reference biologic,” he said.
Currently, the published data include biosimilars to only four reference biologics. “However, I anticipate additional biosimilar-to-biosimilar switching data will become available in the future,” Dr. Cohen said. “In fact, several new studies have been published in recent months, after the cut-off date for inclusion in our systematic review.”
Switching common in rheumatology, dermatology, and gastroenterology
Biosimilar-to-biosimilar switching was observed most commonly in rheumatology practice, but also was seen in the specialties of dermatology and gastroenterology.
Jeffrey Weinberg, MD, clinical professor of dermatology, Icahn School of Medicine at Mount Sinai, New York City, said in an interview that the study is among the best to date showing that switching biosimilars does not compromise efficacy or safety.
“I would hypothesize that the interchangeability would apply to psoriasis patients,” Dr. Weinberg said. However, “over the next few years, we will have an increasing number of biosimilars for an increasing number of different molecules. We will need to be vigilant to observe if similar behavior is observed with the biosimilars yet to come.”
Keith Choate, MD, PhD, professor of dermatology, pathology, and genetics, and associate dean for physician-scientist development at Yale University, New Haven, Conn., said that biosimilars have comparable efficacy to the branded medication they replace. “If response is lost to an individual agent, we would not typically then switch to a biosimilar, but would favor another class of therapy or a distinct therapeutic which targets the same pathway.”
When physicians prescribe a biosimilar for rheumatoid arthritis or psoriatic arthritis, in 9 out 10 people, “it’s going to work as well, and it’s not going to cause any more side effects,” said Stanford Shoor, MD, clinical professor of medicine and rheumatology, Stanford (Calif.) University.
The systematic review, even within its limitations, reinforces confidence in the antitumor necrosis factor biosimilars, said Jean-Frederic Colombel, MD, codirector of the Feinstein Inflammatory Bowel Disease Clinical Center at Mount Sinai, New York, and professor of medicine, division of gastroenterology, Icahn School of Medicine at Mount Sinai.
“Still, studies with longer follow-up are needed,” Dr. Colombel said, adding that the remaining questions relate to the efficacy and safety of switching multiple times, which will likely occur in the near future. There will be a “need to provide information to the patient regarding what originator or biosimilar(s) he has been exposed to during the course of his disease.”
Switching will increasingly become the norm, said Miguel Regueiro, MD, chair of the Digestive Disease & Surgery Institute, Cleveland Clinic. In his clinical practice, he has the most experience with Crohn’s disease and ulcerative colitis, and biosimilar-to-biosimilar infliximab switches. “Unless there are data that emerge, I have no concerns with this.”
He added that it’s an “interesting study that affirms my findings in clinical practice – that one can switch from a biosimilar to biosimilar (of the same reference product).”
The review’s results also make sense from an economic standpoint, said Rajat Bhatt, MD, owner of Prime Rheumatology in Richmond, Tex., and an adjunct faculty member at Caribbean Medical University, Willemstad, Curaçao. “Switching to biosimilars will result in cost savings for the health care system.” Patients on certain insurances also will save by switching to a biosimilar with a lower copay.
However, the review is limited by a relatively small number of studies that have provided primary data on this topic, and most of these were switching from infliximab to a biosimilar for inflammatory bowel disease, said Alfred Kim, MD, PhD, an adult rheumatologist at Barnes-Jewish Hospital, St. Louis, and assistant professor of medicine at Washington University in St. Louis.
As with any meta-analysis evaluating a small number of studies, “broad applicability to all conditions and reference/biosimilar pair can only be assumed. Also, many of the studies used for this meta-analysis are observational, which can introduce a variety of biases that can be difficult to adjust for,” Dr. Kim said. “Nevertheless, these analyses are an important first step in validating the [Food and Drug Administration’s] approach to evaluating biosimilars, as the clinical outcomes are consistent between different biosimilars.”
This systematic review is not enough to prove that all patients will do fine when switching from one biosimilar to another, said Florence Aslinia, MD, a gastroenterologist at the University of Kansas Health System in Kansas City. It’s possible that some patients may not do as well, she said, noting that, in one study of patients with inflammatory bowel disease, 10% of patients on a biosimilar infliximab needed to switch back to the originator infliximab (Remicade, Janssen) because of side effects attributed to the biosimilar. The same thing may or may not happen with biosimilar-to-biosimilar switching, and it requires further study.
The authors did not receive any funding for writing this review. Dr. Cohen is an employee of Sandoz, a division of Novartis. He may own stock in Novartis. Two coauthors are also employees of Sandoz. The other three coauthors reported having financial relationships with numerous pharmaceutical companies, including Sandoz and/or Novartis. Dr. Colombel reported financial relationships with many pharmaceutical companies, including Novartis and other manufacturers of biosimilars. Dr. Regueiro reports financial relationships with numerous pharmaceutical companies, including some manufacturers of biosimilars. Dr. Weinberg reported financial relationships with Celgene, AbbVie, Eli Lilly, and Novartis. Kim reports financial relationships with GlaxoSmithKline, Pfizer, and AstraZeneca. Dr. Aslinia, Dr. Shoor, Dr. Choate, and Dr. Bhatt reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Switching from one biosimilar medication to another is safe and effective, a new systematic review indicates, even though this clinical practice is not governed by current health authority regulations or guidance.
“No reduction in effectiveness or increase in adverse events was detected in biosimilar-to-biosimilar switching studies conducted to date,” the review’s authors noted in their study, published online in BioDrugs.
“The possibility of multiple switches between biosimilars of the same reference biologic is already a reality, and these types of switches are expected to become more common in the future. ... Although it is not covered by current health authority regulations or guidance,” added the authors, led by Hillel P. Cohen, PhD, executive director of scientific affairs at Sandoz, a division of Novartis.
The researchers searched electronic databases through December 2021 and found 23 observational studies that met their search criteria, of which 13 were published in peer-reviewed journals; the remainder appeared in abstract form. The studies totaled 3,657 patients. The researchers did not identify any randomized clinical trials.
“The studies were heterogeneous in size, design, and endpoints, providing data on safety, effectiveness, immunogenicity, pharmacokinetics, patient retention, patient and physician perceptions, and drug-use patterns,” the authors wrote.
The authors found that the majority of studies evaluated switches between biosimilars of infliximab, but they also identified switches between biosimilars of adalimumab, etanercept, and rituximab.
“Some health care providers are hesitant to switch patients from one biosimilar to another biosimilar because of a perceived lack of clinical data on such switches,” Dr. Cohen said in an interview.
The review’s findings – that there were no clinically relevant differences when switching patients from one biosimilar to another – are consistent with the science, Dr. Cohen said. “Physicians should have confidence that the data demonstrate that safety and effectiveness are not impacted if patients switch from one biosimilar to another biosimilar of the same reference biologic,” he said.
Currently, the published data include biosimilars to only four reference biologics. “However, I anticipate additional biosimilar-to-biosimilar switching data will become available in the future,” Dr. Cohen said. “In fact, several new studies have been published in recent months, after the cut-off date for inclusion in our systematic review.”
Switching common in rheumatology, dermatology, and gastroenterology
Biosimilar-to-biosimilar switching was observed most commonly in rheumatology practice, but also was seen in the specialties of dermatology and gastroenterology.
Jeffrey Weinberg, MD, clinical professor of dermatology, Icahn School of Medicine at Mount Sinai, New York City, said in an interview that the study is among the best to date showing that switching biosimilars does not compromise efficacy or safety.
“I would hypothesize that the interchangeability would apply to psoriasis patients,” Dr. Weinberg said. However, “over the next few years, we will have an increasing number of biosimilars for an increasing number of different molecules. We will need to be vigilant to observe if similar behavior is observed with the biosimilars yet to come.”
Keith Choate, MD, PhD, professor of dermatology, pathology, and genetics, and associate dean for physician-scientist development at Yale University, New Haven, Conn., said that biosimilars have comparable efficacy to the branded medication they replace. “If response is lost to an individual agent, we would not typically then switch to a biosimilar, but would favor another class of therapy or a distinct therapeutic which targets the same pathway.”
When physicians prescribe a biosimilar for rheumatoid arthritis or psoriatic arthritis, in 9 out 10 people, “it’s going to work as well, and it’s not going to cause any more side effects,” said Stanford Shoor, MD, clinical professor of medicine and rheumatology, Stanford (Calif.) University.
The systematic review, even within its limitations, reinforces confidence in the antitumor necrosis factor biosimilars, said Jean-Frederic Colombel, MD, codirector of the Feinstein Inflammatory Bowel Disease Clinical Center at Mount Sinai, New York, and professor of medicine, division of gastroenterology, Icahn School of Medicine at Mount Sinai.
“Still, studies with longer follow-up are needed,” Dr. Colombel said, adding that the remaining questions relate to the efficacy and safety of switching multiple times, which will likely occur in the near future. There will be a “need to provide information to the patient regarding what originator or biosimilar(s) he has been exposed to during the course of his disease.”
Switching will increasingly become the norm, said Miguel Regueiro, MD, chair of the Digestive Disease & Surgery Institute, Cleveland Clinic. In his clinical practice, he has the most experience with Crohn’s disease and ulcerative colitis, and biosimilar-to-biosimilar infliximab switches. “Unless there are data that emerge, I have no concerns with this.”
He added that it’s an “interesting study that affirms my findings in clinical practice – that one can switch from a biosimilar to biosimilar (of the same reference product).”
The review’s results also make sense from an economic standpoint, said Rajat Bhatt, MD, owner of Prime Rheumatology in Richmond, Tex., and an adjunct faculty member at Caribbean Medical University, Willemstad, Curaçao. “Switching to biosimilars will result in cost savings for the health care system.” Patients on certain insurances also will save by switching to a biosimilar with a lower copay.
However, the review is limited by a relatively small number of studies that have provided primary data on this topic, and most of these were switching from infliximab to a biosimilar for inflammatory bowel disease, said Alfred Kim, MD, PhD, an adult rheumatologist at Barnes-Jewish Hospital, St. Louis, and assistant professor of medicine at Washington University in St. Louis.
As with any meta-analysis evaluating a small number of studies, “broad applicability to all conditions and reference/biosimilar pair can only be assumed. Also, many of the studies used for this meta-analysis are observational, which can introduce a variety of biases that can be difficult to adjust for,” Dr. Kim said. “Nevertheless, these analyses are an important first step in validating the [Food and Drug Administration’s] approach to evaluating biosimilars, as the clinical outcomes are consistent between different biosimilars.”
This systematic review is not enough to prove that all patients will do fine when switching from one biosimilar to another, said Florence Aslinia, MD, a gastroenterologist at the University of Kansas Health System in Kansas City. It’s possible that some patients may not do as well, she said, noting that, in one study of patients with inflammatory bowel disease, 10% of patients on a biosimilar infliximab needed to switch back to the originator infliximab (Remicade, Janssen) because of side effects attributed to the biosimilar. The same thing may or may not happen with biosimilar-to-biosimilar switching, and it requires further study.
The authors did not receive any funding for writing this review. Dr. Cohen is an employee of Sandoz, a division of Novartis. He may own stock in Novartis. Two coauthors are also employees of Sandoz. The other three coauthors reported having financial relationships with numerous pharmaceutical companies, including Sandoz and/or Novartis. Dr. Colombel reported financial relationships with many pharmaceutical companies, including Novartis and other manufacturers of biosimilars. Dr. Regueiro reports financial relationships with numerous pharmaceutical companies, including some manufacturers of biosimilars. Dr. Weinberg reported financial relationships with Celgene, AbbVie, Eli Lilly, and Novartis. Kim reports financial relationships with GlaxoSmithKline, Pfizer, and AstraZeneca. Dr. Aslinia, Dr. Shoor, Dr. Choate, and Dr. Bhatt reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Switching from one biosimilar medication to another is safe and effective, a new systematic review indicates, even though this clinical practice is not governed by current health authority regulations or guidance.
“No reduction in effectiveness or increase in adverse events was detected in biosimilar-to-biosimilar switching studies conducted to date,” the review’s authors noted in their study, published online in BioDrugs.
“The possibility of multiple switches between biosimilars of the same reference biologic is already a reality, and these types of switches are expected to become more common in the future. ... Although it is not covered by current health authority regulations or guidance,” added the authors, led by Hillel P. Cohen, PhD, executive director of scientific affairs at Sandoz, a division of Novartis.
The researchers searched electronic databases through December 2021 and found 23 observational studies that met their search criteria, of which 13 were published in peer-reviewed journals; the remainder appeared in abstract form. The studies totaled 3,657 patients. The researchers did not identify any randomized clinical trials.
“The studies were heterogeneous in size, design, and endpoints, providing data on safety, effectiveness, immunogenicity, pharmacokinetics, patient retention, patient and physician perceptions, and drug-use patterns,” the authors wrote.
The authors found that the majority of studies evaluated switches between biosimilars of infliximab, but they also identified switches between biosimilars of adalimumab, etanercept, and rituximab.
“Some health care providers are hesitant to switch patients from one biosimilar to another biosimilar because of a perceived lack of clinical data on such switches,” Dr. Cohen said in an interview.
The review’s findings – that there were no clinically relevant differences when switching patients from one biosimilar to another – are consistent with the science, Dr. Cohen said. “Physicians should have confidence that the data demonstrate that safety and effectiveness are not impacted if patients switch from one biosimilar to another biosimilar of the same reference biologic,” he said.
Currently, the published data include biosimilars to only four reference biologics. “However, I anticipate additional biosimilar-to-biosimilar switching data will become available in the future,” Dr. Cohen said. “In fact, several new studies have been published in recent months, after the cut-off date for inclusion in our systematic review.”
Switching common in rheumatology, dermatology, and gastroenterology
Biosimilar-to-biosimilar switching was observed most commonly in rheumatology practice, but also was seen in the specialties of dermatology and gastroenterology.
Jeffrey Weinberg, MD, clinical professor of dermatology, Icahn School of Medicine at Mount Sinai, New York City, said in an interview that the study is among the best to date showing that switching biosimilars does not compromise efficacy or safety.
“I would hypothesize that the interchangeability would apply to psoriasis patients,” Dr. Weinberg said. However, “over the next few years, we will have an increasing number of biosimilars for an increasing number of different molecules. We will need to be vigilant to observe if similar behavior is observed with the biosimilars yet to come.”
Keith Choate, MD, PhD, professor of dermatology, pathology, and genetics, and associate dean for physician-scientist development at Yale University, New Haven, Conn., said that biosimilars have comparable efficacy to the branded medication they replace. “If response is lost to an individual agent, we would not typically then switch to a biosimilar, but would favor another class of therapy or a distinct therapeutic which targets the same pathway.”
When physicians prescribe a biosimilar for rheumatoid arthritis or psoriatic arthritis, in 9 out 10 people, “it’s going to work as well, and it’s not going to cause any more side effects,” said Stanford Shoor, MD, clinical professor of medicine and rheumatology, Stanford (Calif.) University.
The systematic review, even within its limitations, reinforces confidence in the antitumor necrosis factor biosimilars, said Jean-Frederic Colombel, MD, codirector of the Feinstein Inflammatory Bowel Disease Clinical Center at Mount Sinai, New York, and professor of medicine, division of gastroenterology, Icahn School of Medicine at Mount Sinai.
“Still, studies with longer follow-up are needed,” Dr. Colombel said, adding that the remaining questions relate to the efficacy and safety of switching multiple times, which will likely occur in the near future. There will be a “need to provide information to the patient regarding what originator or biosimilar(s) he has been exposed to during the course of his disease.”
Switching will increasingly become the norm, said Miguel Regueiro, MD, chair of the Digestive Disease & Surgery Institute, Cleveland Clinic. In his clinical practice, he has the most experience with Crohn’s disease and ulcerative colitis, and biosimilar-to-biosimilar infliximab switches. “Unless there are data that emerge, I have no concerns with this.”
He added that it’s an “interesting study that affirms my findings in clinical practice – that one can switch from a biosimilar to biosimilar (of the same reference product).”
The review’s results also make sense from an economic standpoint, said Rajat Bhatt, MD, owner of Prime Rheumatology in Richmond, Tex., and an adjunct faculty member at Caribbean Medical University, Willemstad, Curaçao. “Switching to biosimilars will result in cost savings for the health care system.” Patients on certain insurances also will save by switching to a biosimilar with a lower copay.
However, the review is limited by a relatively small number of studies that have provided primary data on this topic, and most of these were switching from infliximab to a biosimilar for inflammatory bowel disease, said Alfred Kim, MD, PhD, an adult rheumatologist at Barnes-Jewish Hospital, St. Louis, and assistant professor of medicine at Washington University in St. Louis.
As with any meta-analysis evaluating a small number of studies, “broad applicability to all conditions and reference/biosimilar pair can only be assumed. Also, many of the studies used for this meta-analysis are observational, which can introduce a variety of biases that can be difficult to adjust for,” Dr. Kim said. “Nevertheless, these analyses are an important first step in validating the [Food and Drug Administration’s] approach to evaluating biosimilars, as the clinical outcomes are consistent between different biosimilars.”
This systematic review is not enough to prove that all patients will do fine when switching from one biosimilar to another, said Florence Aslinia, MD, a gastroenterologist at the University of Kansas Health System in Kansas City. It’s possible that some patients may not do as well, she said, noting that, in one study of patients with inflammatory bowel disease, 10% of patients on a biosimilar infliximab needed to switch back to the originator infliximab (Remicade, Janssen) because of side effects attributed to the biosimilar. The same thing may or may not happen with biosimilar-to-biosimilar switching, and it requires further study.
The authors did not receive any funding for writing this review. Dr. Cohen is an employee of Sandoz, a division of Novartis. He may own stock in Novartis. Two coauthors are also employees of Sandoz. The other three coauthors reported having financial relationships with numerous pharmaceutical companies, including Sandoz and/or Novartis. Dr. Colombel reported financial relationships with many pharmaceutical companies, including Novartis and other manufacturers of biosimilars. Dr. Regueiro reports financial relationships with numerous pharmaceutical companies, including some manufacturers of biosimilars. Dr. Weinberg reported financial relationships with Celgene, AbbVie, Eli Lilly, and Novartis. Kim reports financial relationships with GlaxoSmithKline, Pfizer, and AstraZeneca. Dr. Aslinia, Dr. Shoor, Dr. Choate, and Dr. Bhatt reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM BIODRUGS
Sexual dysfunction, hair loss linked with long COVID
according to findings of a large study.
Anuradhaa Subramanian, PhD, with the Institute of Applied Health Research at the University of Birmingham (England), led the research published online in Nature Medicine.
The team analyzed 486,149 electronic health records from adult patients with confirmed COVID in the United Kingdom, compared with 1.9 million people with no history of COVID, from January 2020 to April 2021. Researchers matched both groups closely in terms of demographic, social, and clinical traits.
New symptoms
The team identified 62 symptoms, including the well-known indicators of long COVID, such as fatigue, loss of sense of smell, shortness of breath, and brain fog, but also hair loss, sexual dysfunction, chest pain, fever, loss of control of bowel movements, and limb swelling.
“These differences in symptoms reported between the infected and uninfected groups remained even after we accounted for age, sex, ethnic group, socioeconomic status, body mass index, smoking status, the presence of more than 80 health conditions, and past reporting of the same symptom,” Dr. Subramanian and coresearcher Shamil Haroon, PhD, wrote in a summary of their research in The Conversation.
They pointed out that only 20 of the symptoms they found are included in the World Health Organization’s clinical case definition for long COVID.
They also found that people more likely to have persistent symptoms 3 months after COVID infection were also more likely to be young, female, smokers, to belong to certain minority ethnic groups, and to have lower socioeconomic status. They were also more likely to be obese and have a wide range of health conditions.
Dr. Haroon, an associate clinical professor at the University of Birmingham, said that one reason it appeared that younger people were more likely to get symptoms of long COVID may be that older adults with COVID were more likely to be hospitalized and weren’t included in this study.
“Since we only considered nonhospitalized adults, the older adults we included in our study may have been relatively healthier and thus had a lower symptom burden,” he said.
Dr. Subramania noted that older patients were more likely to report lasting COVID-related symptoms in the study, but when researchers accounted for a wide range of other conditions that patients had before infection (which generally more commonly happen in older adults), they found younger age as a risk factor for long-term COVID-related symptoms.
In the study period, most patients were unvaccinated, and results came before the widespread Delta and Omicron variants.
More than half (56.6%) of the patients infected with the virus that causes COVID had been diagnosed in 2020, and 43.4% in 2021. Less than 5% (4.5%) of the patients infected with the virus and 4.7% of the patients with no recorded evidence of a COVID infection had received at least a single dose of a COVID vaccine before the study started.
Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, said more studies need to be done to see whether results would be different with vaccination status and evolving variants.
But he noted that this study has several strengths: “The hair loss, libido loss, and ejaculation difficulty are all new symptoms,” and the study – large and carefully controlled – shows these issues were among those more likely to occur.
A loss of sense of smell – which is not a new observation – was still the most likely risk shown in the study, followed by hair loss, sneezing, ejaculation difficulty, and reduced sex drive; followed by shortness of breath, fatigue, chest pain associated with breathing difficulties, hoarseness, and fever.
Three main clusters of symptoms
Given the wide range of symptoms, long COVID likely represents a group of conditions, the authors wrote.
They found three main clusters. The largest, with roughly 80% of people with long COVID in the study, faced a broad spectrum of symptoms, ranging from fatigue to headache and pain. The second-largest group, (15%) mostly had symptoms having to do with mental health and thinking skills, including depression, anxiety, brain fog, and insomnia. The smallest group (5%) had mainly respiratory symptoms such as shortness of breath, coughing, and wheezing.
Putting symptoms in clusters will be important to start understanding what leads to long COVID, said Farha Ikramuddin, MD, a rehabilitation specialist at the University of Minnesota, Minneapolis.
She added that, while the symptoms listed in this paper are new in published research, she has certainly been seeing them over time in her long COVID clinic. (The researchers also used only coded health care data, so they were limited in what symptoms they could discover, she notes.)
Dr. Ikramuddin said a strength of the paper is its large size, but she also cautioned that it’s difficult to determine whether members of the comparison group truly had no COVID infection when the information is taken from their medical records. Often, people test at home or assume they have COVID and don’t test; therefore the information wouldn’t be recorded.
Evaluating nonhospitalized patients is also important, she said, as much of the research on long COVID has come from hospitalized patients, so little has been known about the symptoms of those with milder infections.
“Patients who have been hospitalized and have long COVID look very different from the patients who were not hospitalized,” Dr. Ikramuddin said.
One clear message from the paper, she said, is that listening and asking extensive questions about symptoms are important with patients who have had COVID.
“Counseling has also become very important for our patients in the pandemic,” she said.
It will also be important to do studies on returning to work for patients with long COVID to see how many are able to return and at what capacity, Dr. Ikramuddin said.
A version of this article first appeared on WebMD.com.
according to findings of a large study.
Anuradhaa Subramanian, PhD, with the Institute of Applied Health Research at the University of Birmingham (England), led the research published online in Nature Medicine.
The team analyzed 486,149 electronic health records from adult patients with confirmed COVID in the United Kingdom, compared with 1.9 million people with no history of COVID, from January 2020 to April 2021. Researchers matched both groups closely in terms of demographic, social, and clinical traits.
New symptoms
The team identified 62 symptoms, including the well-known indicators of long COVID, such as fatigue, loss of sense of smell, shortness of breath, and brain fog, but also hair loss, sexual dysfunction, chest pain, fever, loss of control of bowel movements, and limb swelling.
“These differences in symptoms reported between the infected and uninfected groups remained even after we accounted for age, sex, ethnic group, socioeconomic status, body mass index, smoking status, the presence of more than 80 health conditions, and past reporting of the same symptom,” Dr. Subramanian and coresearcher Shamil Haroon, PhD, wrote in a summary of their research in The Conversation.
They pointed out that only 20 of the symptoms they found are included in the World Health Organization’s clinical case definition for long COVID.
They also found that people more likely to have persistent symptoms 3 months after COVID infection were also more likely to be young, female, smokers, to belong to certain minority ethnic groups, and to have lower socioeconomic status. They were also more likely to be obese and have a wide range of health conditions.
Dr. Haroon, an associate clinical professor at the University of Birmingham, said that one reason it appeared that younger people were more likely to get symptoms of long COVID may be that older adults with COVID were more likely to be hospitalized and weren’t included in this study.
“Since we only considered nonhospitalized adults, the older adults we included in our study may have been relatively healthier and thus had a lower symptom burden,” he said.
Dr. Subramania noted that older patients were more likely to report lasting COVID-related symptoms in the study, but when researchers accounted for a wide range of other conditions that patients had before infection (which generally more commonly happen in older adults), they found younger age as a risk factor for long-term COVID-related symptoms.
In the study period, most patients were unvaccinated, and results came before the widespread Delta and Omicron variants.
More than half (56.6%) of the patients infected with the virus that causes COVID had been diagnosed in 2020, and 43.4% in 2021. Less than 5% (4.5%) of the patients infected with the virus and 4.7% of the patients with no recorded evidence of a COVID infection had received at least a single dose of a COVID vaccine before the study started.
Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, said more studies need to be done to see whether results would be different with vaccination status and evolving variants.
But he noted that this study has several strengths: “The hair loss, libido loss, and ejaculation difficulty are all new symptoms,” and the study – large and carefully controlled – shows these issues were among those more likely to occur.
A loss of sense of smell – which is not a new observation – was still the most likely risk shown in the study, followed by hair loss, sneezing, ejaculation difficulty, and reduced sex drive; followed by shortness of breath, fatigue, chest pain associated with breathing difficulties, hoarseness, and fever.
Three main clusters of symptoms
Given the wide range of symptoms, long COVID likely represents a group of conditions, the authors wrote.
They found three main clusters. The largest, with roughly 80% of people with long COVID in the study, faced a broad spectrum of symptoms, ranging from fatigue to headache and pain. The second-largest group, (15%) mostly had symptoms having to do with mental health and thinking skills, including depression, anxiety, brain fog, and insomnia. The smallest group (5%) had mainly respiratory symptoms such as shortness of breath, coughing, and wheezing.
Putting symptoms in clusters will be important to start understanding what leads to long COVID, said Farha Ikramuddin, MD, a rehabilitation specialist at the University of Minnesota, Minneapolis.
She added that, while the symptoms listed in this paper are new in published research, she has certainly been seeing them over time in her long COVID clinic. (The researchers also used only coded health care data, so they were limited in what symptoms they could discover, she notes.)
Dr. Ikramuddin said a strength of the paper is its large size, but she also cautioned that it’s difficult to determine whether members of the comparison group truly had no COVID infection when the information is taken from their medical records. Often, people test at home or assume they have COVID and don’t test; therefore the information wouldn’t be recorded.
Evaluating nonhospitalized patients is also important, she said, as much of the research on long COVID has come from hospitalized patients, so little has been known about the symptoms of those with milder infections.
“Patients who have been hospitalized and have long COVID look very different from the patients who were not hospitalized,” Dr. Ikramuddin said.
One clear message from the paper, she said, is that listening and asking extensive questions about symptoms are important with patients who have had COVID.
“Counseling has also become very important for our patients in the pandemic,” she said.
It will also be important to do studies on returning to work for patients with long COVID to see how many are able to return and at what capacity, Dr. Ikramuddin said.
A version of this article first appeared on WebMD.com.
according to findings of a large study.
Anuradhaa Subramanian, PhD, with the Institute of Applied Health Research at the University of Birmingham (England), led the research published online in Nature Medicine.
The team analyzed 486,149 electronic health records from adult patients with confirmed COVID in the United Kingdom, compared with 1.9 million people with no history of COVID, from January 2020 to April 2021. Researchers matched both groups closely in terms of demographic, social, and clinical traits.
New symptoms
The team identified 62 symptoms, including the well-known indicators of long COVID, such as fatigue, loss of sense of smell, shortness of breath, and brain fog, but also hair loss, sexual dysfunction, chest pain, fever, loss of control of bowel movements, and limb swelling.
“These differences in symptoms reported between the infected and uninfected groups remained even after we accounted for age, sex, ethnic group, socioeconomic status, body mass index, smoking status, the presence of more than 80 health conditions, and past reporting of the same symptom,” Dr. Subramanian and coresearcher Shamil Haroon, PhD, wrote in a summary of their research in The Conversation.
They pointed out that only 20 of the symptoms they found are included in the World Health Organization’s clinical case definition for long COVID.
They also found that people more likely to have persistent symptoms 3 months after COVID infection were also more likely to be young, female, smokers, to belong to certain minority ethnic groups, and to have lower socioeconomic status. They were also more likely to be obese and have a wide range of health conditions.
Dr. Haroon, an associate clinical professor at the University of Birmingham, said that one reason it appeared that younger people were more likely to get symptoms of long COVID may be that older adults with COVID were more likely to be hospitalized and weren’t included in this study.
“Since we only considered nonhospitalized adults, the older adults we included in our study may have been relatively healthier and thus had a lower symptom burden,” he said.
Dr. Subramania noted that older patients were more likely to report lasting COVID-related symptoms in the study, but when researchers accounted for a wide range of other conditions that patients had before infection (which generally more commonly happen in older adults), they found younger age as a risk factor for long-term COVID-related symptoms.
In the study period, most patients were unvaccinated, and results came before the widespread Delta and Omicron variants.
More than half (56.6%) of the patients infected with the virus that causes COVID had been diagnosed in 2020, and 43.4% in 2021. Less than 5% (4.5%) of the patients infected with the virus and 4.7% of the patients with no recorded evidence of a COVID infection had received at least a single dose of a COVID vaccine before the study started.
Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, said more studies need to be done to see whether results would be different with vaccination status and evolving variants.
But he noted that this study has several strengths: “The hair loss, libido loss, and ejaculation difficulty are all new symptoms,” and the study – large and carefully controlled – shows these issues were among those more likely to occur.
A loss of sense of smell – which is not a new observation – was still the most likely risk shown in the study, followed by hair loss, sneezing, ejaculation difficulty, and reduced sex drive; followed by shortness of breath, fatigue, chest pain associated with breathing difficulties, hoarseness, and fever.
Three main clusters of symptoms
Given the wide range of symptoms, long COVID likely represents a group of conditions, the authors wrote.
They found three main clusters. The largest, with roughly 80% of people with long COVID in the study, faced a broad spectrum of symptoms, ranging from fatigue to headache and pain. The second-largest group, (15%) mostly had symptoms having to do with mental health and thinking skills, including depression, anxiety, brain fog, and insomnia. The smallest group (5%) had mainly respiratory symptoms such as shortness of breath, coughing, and wheezing.
Putting symptoms in clusters will be important to start understanding what leads to long COVID, said Farha Ikramuddin, MD, a rehabilitation specialist at the University of Minnesota, Minneapolis.
She added that, while the symptoms listed in this paper are new in published research, she has certainly been seeing them over time in her long COVID clinic. (The researchers also used only coded health care data, so they were limited in what symptoms they could discover, she notes.)
Dr. Ikramuddin said a strength of the paper is its large size, but she also cautioned that it’s difficult to determine whether members of the comparison group truly had no COVID infection when the information is taken from their medical records. Often, people test at home or assume they have COVID and don’t test; therefore the information wouldn’t be recorded.
Evaluating nonhospitalized patients is also important, she said, as much of the research on long COVID has come from hospitalized patients, so little has been known about the symptoms of those with milder infections.
“Patients who have been hospitalized and have long COVID look very different from the patients who were not hospitalized,” Dr. Ikramuddin said.
One clear message from the paper, she said, is that listening and asking extensive questions about symptoms are important with patients who have had COVID.
“Counseling has also become very important for our patients in the pandemic,” she said.
It will also be important to do studies on returning to work for patients with long COVID to see how many are able to return and at what capacity, Dr. Ikramuddin said.
A version of this article first appeared on WebMD.com.
FROM NATURE MEDICINE