User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID-19 vaccination does not increase risk of flare in patients with lupus
COVID-19 vaccinations appear to be well tolerated in patients with systemic lupus erythematosus (SLE) and come with a low risk of flare, according to the results of a global, web-based survey.
“Disseminating these reassuring data might prove crucial to increasing vaccine coverage in patients with SLE,” wrote lead author Renaud Felten, MD, of Strasbourg (France) University Hospital. Their results were published as a comment in Lancet Rheumatology.
To assess vaccine tolerability among lupus patients, the cross-sectional Tolerance and Consequences of Vaccination Against COVID-19 in Lupus Patients (VACOLUP) study analyzed a 43-question survey of 696 participants with a self-reported, medically confirmed diagnosis of SLE from 30 countries between March 22, 2021, and May 17, 2021. The cohort was 96% women, and their median age was 42 (interquartile range, 34-51). Nearly 36% of respondents were from Italy, 27% were from Chile, 13% were from France, and just under 9% were Americans. All participants received at least one dose of COVID-19 vaccine, and 49% received a second dose. The most common vaccines were Pfizer-BioNTech (57%), Sinovac (22%), AstraZeneca (10%), and Moderna (8%).
Only 21 participants (3%) reported a medically confirmed SLE flare after a median of 3 days (IQR, 0-29) post COVID vaccination, with most experiencing musculoskeletal symptoms (90%) and fatigue (86%). Of the 21 cases, 15 reported a subsequent change in SLE treatment and 4 were admitted to the hospital. A previous flare that occurred within a year before vaccination was associated with an increased risk of flare post vaccination (relative risk, 5.52; 95% confidence interval, 2.17-14.03; P < .0001).
Side effects – including swelling, soreness, fever, chills, fatigue, joint and muscle pain, nausea, and headache – were reported in 45% of participants (n = 316) after their first dose and in 53% of the 343 participants who received a second dose. There was no notable difference in the likelihood of side effects across gender and age or in patients who received mRNA vaccines, compared with vaccines with other modes of action. Patients who reported side effects after the first dose were more likely to also report them after the second, compared with those who reported none (109 [81%] of 135 vs. 72 [35%] of 205; RR, 2.30; 95% CI, 1.88-2.82; P < .0001).
In the majority of cases (2,232 of 2,683), the side effects were of minor or moderate intensity and did not affect the participants’ ability to perform daily tasks. The study found no significant association between side effects and a SLE flare and SLE medications or previous SLE disease manifestations.
When asked to comment on the study, Amit Saxena, MD, of the Lupus Center at New York University Langone Health, said: “What we are seeing is pretty mild to moderate in terms of follow-up side effects or lupus-related activity. Several studies have shown this amongst our autoimmune rheumatology cohort, as well as what I’ve seen clinically in my own patients. More than anything else, numbers are the most important, and this is a large study.”
He acknowledged the benefits of going directly to patients to gauge their responses and reactions, giving them the opportunity to share concerns that physicians may not think about.
“As rheumatologists, we tend to focus on certain things that might not necessarily be what the patients themselves focus on,” he said. “I think the fact that this questionnaire dealt with a lot of what people complain about – fatigue, sore arm, things that we know are part of getting the vaccine – they aren’t necessarily things we capture with tools that screen for lupus flares, for example.”
More than anything, Dr. Saxena commended the study’s timeliness. “Patients are constantly asking us about the vaccine, and there’s so much misinformation,” he said. “People say, ‘Because I have lupus, I was told not to get vaccinated.’ I don’t know where they get that information from; we are telling everyone to get it, especially our lupus patients.”
The authors recognized their study’s main limitation as the self-reported and subjective nature of the survey, which they attempted to mitigate by asking for medically confirmed flares only. They noted, however, that the short median time between vaccination and flare onset could be caused by patients confusing expected side effects for something more serious, meaning the 3% figure “could be an overestimation of the actual flare rate.”
“Vaccination is recommended for patients with rheumatic and musculoskeletal diseases according to the American College of Rheumatology,” they added, “irrespective of disease activity and severity.”
Several authors reported potential conflicts of interest, including receiving consultancy fees and grants from Pfizer, GlaxoSmithKline, AbbVie, and Janssen, all unrelated to the study.
COVID-19 vaccinations appear to be well tolerated in patients with systemic lupus erythematosus (SLE) and come with a low risk of flare, according to the results of a global, web-based survey.
“Disseminating these reassuring data might prove crucial to increasing vaccine coverage in patients with SLE,” wrote lead author Renaud Felten, MD, of Strasbourg (France) University Hospital. Their results were published as a comment in Lancet Rheumatology.
To assess vaccine tolerability among lupus patients, the cross-sectional Tolerance and Consequences of Vaccination Against COVID-19 in Lupus Patients (VACOLUP) study analyzed a 43-question survey of 696 participants with a self-reported, medically confirmed diagnosis of SLE from 30 countries between March 22, 2021, and May 17, 2021. The cohort was 96% women, and their median age was 42 (interquartile range, 34-51). Nearly 36% of respondents were from Italy, 27% were from Chile, 13% were from France, and just under 9% were Americans. All participants received at least one dose of COVID-19 vaccine, and 49% received a second dose. The most common vaccines were Pfizer-BioNTech (57%), Sinovac (22%), AstraZeneca (10%), and Moderna (8%).
Only 21 participants (3%) reported a medically confirmed SLE flare after a median of 3 days (IQR, 0-29) post COVID vaccination, with most experiencing musculoskeletal symptoms (90%) and fatigue (86%). Of the 21 cases, 15 reported a subsequent change in SLE treatment and 4 were admitted to the hospital. A previous flare that occurred within a year before vaccination was associated with an increased risk of flare post vaccination (relative risk, 5.52; 95% confidence interval, 2.17-14.03; P < .0001).
Side effects – including swelling, soreness, fever, chills, fatigue, joint and muscle pain, nausea, and headache – were reported in 45% of participants (n = 316) after their first dose and in 53% of the 343 participants who received a second dose. There was no notable difference in the likelihood of side effects across gender and age or in patients who received mRNA vaccines, compared with vaccines with other modes of action. Patients who reported side effects after the first dose were more likely to also report them after the second, compared with those who reported none (109 [81%] of 135 vs. 72 [35%] of 205; RR, 2.30; 95% CI, 1.88-2.82; P < .0001).
In the majority of cases (2,232 of 2,683), the side effects were of minor or moderate intensity and did not affect the participants’ ability to perform daily tasks. The study found no significant association between side effects and a SLE flare and SLE medications or previous SLE disease manifestations.
When asked to comment on the study, Amit Saxena, MD, of the Lupus Center at New York University Langone Health, said: “What we are seeing is pretty mild to moderate in terms of follow-up side effects or lupus-related activity. Several studies have shown this amongst our autoimmune rheumatology cohort, as well as what I’ve seen clinically in my own patients. More than anything else, numbers are the most important, and this is a large study.”
He acknowledged the benefits of going directly to patients to gauge their responses and reactions, giving them the opportunity to share concerns that physicians may not think about.
“As rheumatologists, we tend to focus on certain things that might not necessarily be what the patients themselves focus on,” he said. “I think the fact that this questionnaire dealt with a lot of what people complain about – fatigue, sore arm, things that we know are part of getting the vaccine – they aren’t necessarily things we capture with tools that screen for lupus flares, for example.”
More than anything, Dr. Saxena commended the study’s timeliness. “Patients are constantly asking us about the vaccine, and there’s so much misinformation,” he said. “People say, ‘Because I have lupus, I was told not to get vaccinated.’ I don’t know where they get that information from; we are telling everyone to get it, especially our lupus patients.”
The authors recognized their study’s main limitation as the self-reported and subjective nature of the survey, which they attempted to mitigate by asking for medically confirmed flares only. They noted, however, that the short median time between vaccination and flare onset could be caused by patients confusing expected side effects for something more serious, meaning the 3% figure “could be an overestimation of the actual flare rate.”
“Vaccination is recommended for patients with rheumatic and musculoskeletal diseases according to the American College of Rheumatology,” they added, “irrespective of disease activity and severity.”
Several authors reported potential conflicts of interest, including receiving consultancy fees and grants from Pfizer, GlaxoSmithKline, AbbVie, and Janssen, all unrelated to the study.
COVID-19 vaccinations appear to be well tolerated in patients with systemic lupus erythematosus (SLE) and come with a low risk of flare, according to the results of a global, web-based survey.
“Disseminating these reassuring data might prove crucial to increasing vaccine coverage in patients with SLE,” wrote lead author Renaud Felten, MD, of Strasbourg (France) University Hospital. Their results were published as a comment in Lancet Rheumatology.
To assess vaccine tolerability among lupus patients, the cross-sectional Tolerance and Consequences of Vaccination Against COVID-19 in Lupus Patients (VACOLUP) study analyzed a 43-question survey of 696 participants with a self-reported, medically confirmed diagnosis of SLE from 30 countries between March 22, 2021, and May 17, 2021. The cohort was 96% women, and their median age was 42 (interquartile range, 34-51). Nearly 36% of respondents were from Italy, 27% were from Chile, 13% were from France, and just under 9% were Americans. All participants received at least one dose of COVID-19 vaccine, and 49% received a second dose. The most common vaccines were Pfizer-BioNTech (57%), Sinovac (22%), AstraZeneca (10%), and Moderna (8%).
Only 21 participants (3%) reported a medically confirmed SLE flare after a median of 3 days (IQR, 0-29) post COVID vaccination, with most experiencing musculoskeletal symptoms (90%) and fatigue (86%). Of the 21 cases, 15 reported a subsequent change in SLE treatment and 4 were admitted to the hospital. A previous flare that occurred within a year before vaccination was associated with an increased risk of flare post vaccination (relative risk, 5.52; 95% confidence interval, 2.17-14.03; P < .0001).
Side effects – including swelling, soreness, fever, chills, fatigue, joint and muscle pain, nausea, and headache – were reported in 45% of participants (n = 316) after their first dose and in 53% of the 343 participants who received a second dose. There was no notable difference in the likelihood of side effects across gender and age or in patients who received mRNA vaccines, compared with vaccines with other modes of action. Patients who reported side effects after the first dose were more likely to also report them after the second, compared with those who reported none (109 [81%] of 135 vs. 72 [35%] of 205; RR, 2.30; 95% CI, 1.88-2.82; P < .0001).
In the majority of cases (2,232 of 2,683), the side effects were of minor or moderate intensity and did not affect the participants’ ability to perform daily tasks. The study found no significant association between side effects and a SLE flare and SLE medications or previous SLE disease manifestations.
When asked to comment on the study, Amit Saxena, MD, of the Lupus Center at New York University Langone Health, said: “What we are seeing is pretty mild to moderate in terms of follow-up side effects or lupus-related activity. Several studies have shown this amongst our autoimmune rheumatology cohort, as well as what I’ve seen clinically in my own patients. More than anything else, numbers are the most important, and this is a large study.”
He acknowledged the benefits of going directly to patients to gauge their responses and reactions, giving them the opportunity to share concerns that physicians may not think about.
“As rheumatologists, we tend to focus on certain things that might not necessarily be what the patients themselves focus on,” he said. “I think the fact that this questionnaire dealt with a lot of what people complain about – fatigue, sore arm, things that we know are part of getting the vaccine – they aren’t necessarily things we capture with tools that screen for lupus flares, for example.”
More than anything, Dr. Saxena commended the study’s timeliness. “Patients are constantly asking us about the vaccine, and there’s so much misinformation,” he said. “People say, ‘Because I have lupus, I was told not to get vaccinated.’ I don’t know where they get that information from; we are telling everyone to get it, especially our lupus patients.”
The authors recognized their study’s main limitation as the self-reported and subjective nature of the survey, which they attempted to mitigate by asking for medically confirmed flares only. They noted, however, that the short median time between vaccination and flare onset could be caused by patients confusing expected side effects for something more serious, meaning the 3% figure “could be an overestimation of the actual flare rate.”
“Vaccination is recommended for patients with rheumatic and musculoskeletal diseases according to the American College of Rheumatology,” they added, “irrespective of disease activity and severity.”
Several authors reported potential conflicts of interest, including receiving consultancy fees and grants from Pfizer, GlaxoSmithKline, AbbVie, and Janssen, all unrelated to the study.
FROM THE LANCET RHEUMATOLOGY
Genetic testing for neurofibromatosis 1: An imperfect science
According to Peter Kannu, MB, ChB, DCH, PhD, a definitive diagnosis of NF1 can be made in most children using National Institutes of Health criteria published in 1988, which include the presence of two of the following:
- Six or more café au lait macules over 5 mm in diameter in prepubertal individuals and over 15 mm in greatest diameter in postpubertal individuals
- Two or more neurofibromas of any type or one plexiform neurofibroma
- Freckling in the axillary or inguinal regions
- Two or more Lisch nodules
- Optic glioma
- A distinctive osseous lesion such as sphenoid dysplasia or thinning of long bone cortex, with or without pseudarthrosis
- Having a first-degree relative with NF1
For example, in the case of an 8-year-old child who presents with multiple café au lait macules, axillary and inguinal freckling, Lisch nodules, and an optic glioma, “the diagnosis is secure and genetic testing is not going to change clinical management or surveillance,” Dr. Kannu, a clinical geneticist at the University of Alberta, Edmonton, said during the annual meeting of the Society for Pediatric Dermatology. “The only reason for genetic testing in this situation is so that we know the mutation in order to inform reproductive risk counseling in the future.”
However, while a diagnosis of NF1 may be suspected in a 6- to 12-month-old presenting with only café au lait macules, “the diagnosis is not secure because the clinical criteria cannot be met. In this situation, a genetic test can speed up the diagnosis,” he added. “Or, if the test is negative, it can decrease your suspicion for NF1 and you wouldn’t refer the child on to an NF1 screening clinic for intensive surveillance.”
Dr. Kannu based his remarks largely on his 5 years working at the multidisciplinary Genodermatoses Clinic at the Hospital for Sick Children, Toronto. Founded in 2015, the clinic is a “one-stop shop” designed to reduce the wait time for diagnosis and management and the number of hospital visits. The team – composed of a dermatologist, medical geneticist, genetic counselor, residents, and fellows – meets to review the charts of each patient before the appointment, and decides on a preliminary management plan. All children are then seen by one of the trainees in the clinic who devises a differential diagnosis that is presented to staff physicians, at which point genetic testing is decided on. A genetics counselor handles follow-up for those who do have genetic testing.
In 2018, Dr. Kannu and colleagues conducted an informal review of 300 patients who had been seen in the clinic. The mean age at referral was about 6 years, 51% were female, and the top three referral sources were pediatricians (51%), dermatologists (18%), and family physicians (18%). Of the 300 children, 84 (28%) were confirmed to have a diagnosis of NF1. Two patients were diagnosed with NF2 and 5% of the total cohort was diagnosed with mosaic NF1 (MNF1), “which is higher than what you would expect based on the incidence of MNF1 in the literature,” he said.
He separates genetic tests for NF1 into one of two categories: Conventional testing, which is offered by most labs in North America; and comprehensive testing, which is offered by the medical genomics lab at the University of Alabama at Birmingham. Conventional testing focuses on the exons, “the protein coding regions of the gene where most of the mutations lie,” he said. “The test also sequences about 20 base pairs or so of the intron exon boundary and may pick up some intronic mutations. But this test will not detect anything that’s hidden deep in the intronic region.”
Comprehensive testing, meanwhile, checks for mutations in both introns and exons.
Dr. Kannu and colleagues published a case of a paraspinal ganglioneuroma in the proband of a large family with mild cutaneous manifestations of NF1, carrying a deep NF1 intronic mutation. “The clinicians were suspicious that this was NF1, rightly so. The diagnosis was only confirmed after we sent samples to the University of Alabama lab where the deep intronic mutation was found,” he said.
The other situation where conventional genetic testing may be negative is in the case of MNF1, where there “are mutations in some cells but not all cells,” Dr. Kannu explained. “It may only be present in the melanocytes of the skin but not present in the lymphocytes in the blood. Mosaicism is characterized by the regional distribution of pigmentary or other NF1 associated findings. Mosaicism may be detected in the blood if it’s more than 20%. Anything less than that is not detected with conventional genetic testing using DNA from blood and requires extracting DNA from a punch biopsy sample of a café au lait macule.”
The differential diagnosis of café au lait macules includes several conditions associated mutations in the RAS pathway. “Neurofibromin is a key signal of molecules which regulates the activation of RAS,” Dr. Kannu said. “A close binding partner of NF1 is SPRED 1. We know that mutations in this gene cause Legius syndrome, a condition which presents with multiple café au lait macules.”
Two key receptors in the RAS pathway include EGFR and KITL, he continued. Mutations in the EGFR receptor cause a rare condition known as neonatal skin and bowel disease, while mutations in the KITL receptor cause familial progressive hyperpigmentation with or without hypopigmentation. “Looking into the pathway and focusing downstream of RAS, we have genes such as RAF and CBL, which are mutated in Noonan syndrome,” he said. “Further along in the pathway you have mutations in PTEN, which cause Cowden syndrome, and mutations in TSC1 and TSC2, which cause tuberous sclerosis. Mutations in any of these genes can also present with café au lait macules.”
During a question-and-answer session Dr. Kannu was asked to comment about revised diagnostic criteria for NF1 based on an international consensus recommendation, such as changes in the eye that require a formal opthalmologic examination, which were recently published.
“We are understanding more about the phenotype,” he said. “If you fulfill diagnostic criteria for NF1, the main reasons for doing genetic testing are, one, if the family wants to know that information, and two, it informs our reproductive risk counseling. Genotype-phenotype correlations do exist in NF1 but they’re not very robust, so that information is not clinically useful.”
Dr. Kannu disclosed that he has been an advisory board member for Ipsen, Novartis, and Alexion. He has also been a primary investigator for QED and Clementia.
According to Peter Kannu, MB, ChB, DCH, PhD, a definitive diagnosis of NF1 can be made in most children using National Institutes of Health criteria published in 1988, which include the presence of two of the following:
- Six or more café au lait macules over 5 mm in diameter in prepubertal individuals and over 15 mm in greatest diameter in postpubertal individuals
- Two or more neurofibromas of any type or one plexiform neurofibroma
- Freckling in the axillary or inguinal regions
- Two or more Lisch nodules
- Optic glioma
- A distinctive osseous lesion such as sphenoid dysplasia or thinning of long bone cortex, with or without pseudarthrosis
- Having a first-degree relative with NF1
For example, in the case of an 8-year-old child who presents with multiple café au lait macules, axillary and inguinal freckling, Lisch nodules, and an optic glioma, “the diagnosis is secure and genetic testing is not going to change clinical management or surveillance,” Dr. Kannu, a clinical geneticist at the University of Alberta, Edmonton, said during the annual meeting of the Society for Pediatric Dermatology. “The only reason for genetic testing in this situation is so that we know the mutation in order to inform reproductive risk counseling in the future.”
However, while a diagnosis of NF1 may be suspected in a 6- to 12-month-old presenting with only café au lait macules, “the diagnosis is not secure because the clinical criteria cannot be met. In this situation, a genetic test can speed up the diagnosis,” he added. “Or, if the test is negative, it can decrease your suspicion for NF1 and you wouldn’t refer the child on to an NF1 screening clinic for intensive surveillance.”
Dr. Kannu based his remarks largely on his 5 years working at the multidisciplinary Genodermatoses Clinic at the Hospital for Sick Children, Toronto. Founded in 2015, the clinic is a “one-stop shop” designed to reduce the wait time for diagnosis and management and the number of hospital visits. The team – composed of a dermatologist, medical geneticist, genetic counselor, residents, and fellows – meets to review the charts of each patient before the appointment, and decides on a preliminary management plan. All children are then seen by one of the trainees in the clinic who devises a differential diagnosis that is presented to staff physicians, at which point genetic testing is decided on. A genetics counselor handles follow-up for those who do have genetic testing.
In 2018, Dr. Kannu and colleagues conducted an informal review of 300 patients who had been seen in the clinic. The mean age at referral was about 6 years, 51% were female, and the top three referral sources were pediatricians (51%), dermatologists (18%), and family physicians (18%). Of the 300 children, 84 (28%) were confirmed to have a diagnosis of NF1. Two patients were diagnosed with NF2 and 5% of the total cohort was diagnosed with mosaic NF1 (MNF1), “which is higher than what you would expect based on the incidence of MNF1 in the literature,” he said.
He separates genetic tests for NF1 into one of two categories: Conventional testing, which is offered by most labs in North America; and comprehensive testing, which is offered by the medical genomics lab at the University of Alabama at Birmingham. Conventional testing focuses on the exons, “the protein coding regions of the gene where most of the mutations lie,” he said. “The test also sequences about 20 base pairs or so of the intron exon boundary and may pick up some intronic mutations. But this test will not detect anything that’s hidden deep in the intronic region.”
Comprehensive testing, meanwhile, checks for mutations in both introns and exons.
Dr. Kannu and colleagues published a case of a paraspinal ganglioneuroma in the proband of a large family with mild cutaneous manifestations of NF1, carrying a deep NF1 intronic mutation. “The clinicians were suspicious that this was NF1, rightly so. The diagnosis was only confirmed after we sent samples to the University of Alabama lab where the deep intronic mutation was found,” he said.
The other situation where conventional genetic testing may be negative is in the case of MNF1, where there “are mutations in some cells but not all cells,” Dr. Kannu explained. “It may only be present in the melanocytes of the skin but not present in the lymphocytes in the blood. Mosaicism is characterized by the regional distribution of pigmentary or other NF1 associated findings. Mosaicism may be detected in the blood if it’s more than 20%. Anything less than that is not detected with conventional genetic testing using DNA from blood and requires extracting DNA from a punch biopsy sample of a café au lait macule.”
The differential diagnosis of café au lait macules includes several conditions associated mutations in the RAS pathway. “Neurofibromin is a key signal of molecules which regulates the activation of RAS,” Dr. Kannu said. “A close binding partner of NF1 is SPRED 1. We know that mutations in this gene cause Legius syndrome, a condition which presents with multiple café au lait macules.”
Two key receptors in the RAS pathway include EGFR and KITL, he continued. Mutations in the EGFR receptor cause a rare condition known as neonatal skin and bowel disease, while mutations in the KITL receptor cause familial progressive hyperpigmentation with or without hypopigmentation. “Looking into the pathway and focusing downstream of RAS, we have genes such as RAF and CBL, which are mutated in Noonan syndrome,” he said. “Further along in the pathway you have mutations in PTEN, which cause Cowden syndrome, and mutations in TSC1 and TSC2, which cause tuberous sclerosis. Mutations in any of these genes can also present with café au lait macules.”
During a question-and-answer session Dr. Kannu was asked to comment about revised diagnostic criteria for NF1 based on an international consensus recommendation, such as changes in the eye that require a formal opthalmologic examination, which were recently published.
“We are understanding more about the phenotype,” he said. “If you fulfill diagnostic criteria for NF1, the main reasons for doing genetic testing are, one, if the family wants to know that information, and two, it informs our reproductive risk counseling. Genotype-phenotype correlations do exist in NF1 but they’re not very robust, so that information is not clinically useful.”
Dr. Kannu disclosed that he has been an advisory board member for Ipsen, Novartis, and Alexion. He has also been a primary investigator for QED and Clementia.
According to Peter Kannu, MB, ChB, DCH, PhD, a definitive diagnosis of NF1 can be made in most children using National Institutes of Health criteria published in 1988, which include the presence of two of the following:
- Six or more café au lait macules over 5 mm in diameter in prepubertal individuals and over 15 mm in greatest diameter in postpubertal individuals
- Two or more neurofibromas of any type or one plexiform neurofibroma
- Freckling in the axillary or inguinal regions
- Two or more Lisch nodules
- Optic glioma
- A distinctive osseous lesion such as sphenoid dysplasia or thinning of long bone cortex, with or without pseudarthrosis
- Having a first-degree relative with NF1
For example, in the case of an 8-year-old child who presents with multiple café au lait macules, axillary and inguinal freckling, Lisch nodules, and an optic glioma, “the diagnosis is secure and genetic testing is not going to change clinical management or surveillance,” Dr. Kannu, a clinical geneticist at the University of Alberta, Edmonton, said during the annual meeting of the Society for Pediatric Dermatology. “The only reason for genetic testing in this situation is so that we know the mutation in order to inform reproductive risk counseling in the future.”
However, while a diagnosis of NF1 may be suspected in a 6- to 12-month-old presenting with only café au lait macules, “the diagnosis is not secure because the clinical criteria cannot be met. In this situation, a genetic test can speed up the diagnosis,” he added. “Or, if the test is negative, it can decrease your suspicion for NF1 and you wouldn’t refer the child on to an NF1 screening clinic for intensive surveillance.”
Dr. Kannu based his remarks largely on his 5 years working at the multidisciplinary Genodermatoses Clinic at the Hospital for Sick Children, Toronto. Founded in 2015, the clinic is a “one-stop shop” designed to reduce the wait time for diagnosis and management and the number of hospital visits. The team – composed of a dermatologist, medical geneticist, genetic counselor, residents, and fellows – meets to review the charts of each patient before the appointment, and decides on a preliminary management plan. All children are then seen by one of the trainees in the clinic who devises a differential diagnosis that is presented to staff physicians, at which point genetic testing is decided on. A genetics counselor handles follow-up for those who do have genetic testing.
In 2018, Dr. Kannu and colleagues conducted an informal review of 300 patients who had been seen in the clinic. The mean age at referral was about 6 years, 51% were female, and the top three referral sources were pediatricians (51%), dermatologists (18%), and family physicians (18%). Of the 300 children, 84 (28%) were confirmed to have a diagnosis of NF1. Two patients were diagnosed with NF2 and 5% of the total cohort was diagnosed with mosaic NF1 (MNF1), “which is higher than what you would expect based on the incidence of MNF1 in the literature,” he said.
He separates genetic tests for NF1 into one of two categories: Conventional testing, which is offered by most labs in North America; and comprehensive testing, which is offered by the medical genomics lab at the University of Alabama at Birmingham. Conventional testing focuses on the exons, “the protein coding regions of the gene where most of the mutations lie,” he said. “The test also sequences about 20 base pairs or so of the intron exon boundary and may pick up some intronic mutations. But this test will not detect anything that’s hidden deep in the intronic region.”
Comprehensive testing, meanwhile, checks for mutations in both introns and exons.
Dr. Kannu and colleagues published a case of a paraspinal ganglioneuroma in the proband of a large family with mild cutaneous manifestations of NF1, carrying a deep NF1 intronic mutation. “The clinicians were suspicious that this was NF1, rightly so. The diagnosis was only confirmed after we sent samples to the University of Alabama lab where the deep intronic mutation was found,” he said.
The other situation where conventional genetic testing may be negative is in the case of MNF1, where there “are mutations in some cells but not all cells,” Dr. Kannu explained. “It may only be present in the melanocytes of the skin but not present in the lymphocytes in the blood. Mosaicism is characterized by the regional distribution of pigmentary or other NF1 associated findings. Mosaicism may be detected in the blood if it’s more than 20%. Anything less than that is not detected with conventional genetic testing using DNA from blood and requires extracting DNA from a punch biopsy sample of a café au lait macule.”
The differential diagnosis of café au lait macules includes several conditions associated mutations in the RAS pathway. “Neurofibromin is a key signal of molecules which regulates the activation of RAS,” Dr. Kannu said. “A close binding partner of NF1 is SPRED 1. We know that mutations in this gene cause Legius syndrome, a condition which presents with multiple café au lait macules.”
Two key receptors in the RAS pathway include EGFR and KITL, he continued. Mutations in the EGFR receptor cause a rare condition known as neonatal skin and bowel disease, while mutations in the KITL receptor cause familial progressive hyperpigmentation with or without hypopigmentation. “Looking into the pathway and focusing downstream of RAS, we have genes such as RAF and CBL, which are mutated in Noonan syndrome,” he said. “Further along in the pathway you have mutations in PTEN, which cause Cowden syndrome, and mutations in TSC1 and TSC2, which cause tuberous sclerosis. Mutations in any of these genes can also present with café au lait macules.”
During a question-and-answer session Dr. Kannu was asked to comment about revised diagnostic criteria for NF1 based on an international consensus recommendation, such as changes in the eye that require a formal opthalmologic examination, which were recently published.
“We are understanding more about the phenotype,” he said. “If you fulfill diagnostic criteria for NF1, the main reasons for doing genetic testing are, one, if the family wants to know that information, and two, it informs our reproductive risk counseling. Genotype-phenotype correlations do exist in NF1 but they’re not very robust, so that information is not clinically useful.”
Dr. Kannu disclosed that he has been an advisory board member for Ipsen, Novartis, and Alexion. He has also been a primary investigator for QED and Clementia.
FROM SPD 2021
Mayo, Cleveland Clinics top latest U.S. News & World Report hospital rankings
This year’s expanded report debuts new ratings for seven “important procedures and conditions to help patients, in consultation with their doctors, narrow down their choice of hospital based on the specific type of care they need,” Ben Harder, managing editor and chief of health analysis, said in a news release.
With new ratings for myocardial infarction, stroke, hip fracture, and back surgery (spinal fusion), the report now ranks 17 procedures and conditions.
Also new to the 2021 report, which marks the 32nd edition, is a look at racial disparities in health care and the inclusion of health equity measures alongside the hospital rankings.
The new measures examine whether the patients each hospital has treated reflect the racial and ethnic diversity of the surrounding community, among other aspects of health equity.
“At roughly four out of five hospitals, we found that the community’s minority residents were underrepresented among patients receiving services such as joint replacement, cancer surgery and common heart procedures,” Mr. Harder said.
“Against this backdrop, however, we found important exceptions – hospitals that provide care to a disproportionate share of their community’s minority residents. These metrics are just a beginning; we aim to expand on our measurement of health equity in the future,” Mr. Harder added.
Mayo and Cleveland Clinic remain tops
Following the Mayo Clinic, the Cleveland Clinic once again takes the No. 2 spot in the magazine’s latest annual honor roll of best hospitals, which highlights hospitals that deliver exceptional treatment across multiple areas of care.
UCLA Medical Center, Los Angeles, holds the No. 3 spot in 2021. In 2020, UCLA Medical Center and New York–Presbyterian Hospital–Columbia and Cornell, New York, sat in a tie at No. 4.
In 2021, Johns Hopkins Hospital, Baltimore, which held the No. 3 spot in 2020, drops to No. 4, while Massachusetts General Hospital in Boston takes the No. 5 spot, up from No. 6 in 2020.
Rounding out the top 10 (in order) are Cedars-Sinai Medical Center, Los Angeles; New York–Presbyterian Hospital–Columbia and Cornell, New York; NYU Langone Hospitals, New York; UCSF Medical Center, San Francisco; and Northwestern Memorial Hospital, Chicago.
2021-2022 Best Hospitals honor roll
1. Mayo Clinic, Rochester, Minn.
2. Cleveland Clinic, Cleveland
3. UCLA Medical Center, Los Angeles
4. Johns Hopkins Hospital, Baltimore
5. Massachusetts General Hospital, Boston
6. Cedars-Sinai Medical Center, San Francisco
7. New York–Presbyterian Hospital–Columbia and Cornell, New York
8. NYU Langone Hospitals, New York
9. UCSF Medical Center, San Francisco
10. Northwestern Memorial Hospital, Chicago
11. University of Michigan Hospitals–Michigan Medicine, Ann Arbor.
12. Stanford Health Care–Stanford Hospital, Palo Alto, Calif.
13. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
14. Brigham and Women’s Hospital, Boston
15. Mayo Clinic–Phoenix, Phoenix
16. Houston Methodist Hospital, Houston
17. (tie) Barnes-Jewish Hospital, St. Louis
17. (tie) Mount Sinai Hospital, New York Rush University Medical Center, Chicago
19. Rush University Medical Center, Chicago
20. Vanderbilt University Medical Center, Nashville, Tenn.
For the 2021-2022 rankings and ratings, the magazine compared more than 4,750 hospitals nationwide in 15 specialties and 17 procedures and conditions.
At least 2,039 hospitals received a high performance rating in at least one of the services rated; 11 hospitals received high performance in all 17. A total of 175 hospitals were nationally ranked in at least one specialty
For specialty rankings, the University of Texas MD Anderson Cancer Center continues to hold the No. 1 spot in cancer care, the Hospital for Special Surgery continues to be No. 1 in orthopedics, and the Cleveland Clinic continues to be No. 1 in cardiology and heart surgery.
Top five for cancer
1. University of Texas MD Anderson Cancer Center, Houston
2. Memorial Sloan Kettering Cancer Center, New York
3. Mayo Clinic, Rochester, Minn.
4. Dana-Farber/Brigham & Women’s Cancer Center, Boston
5. Cleveland Clinic, Cleveland
Top five for cardiology and heart surgery
1. Cleveland Clinic, Cleveland
2. Mayo Clinic, Rochester, Minn.
3. Cedars-Sinai Medical Center, Los Angeles
4. New York–Presbyterian Hospital–Columbia and Cornell, New York
5. NYU Langone Hospitals, New York
Top five for orthopedics
1. Hospital for Special Surgery, New York
2. Mayo Clinic, Rochester, Minn.
3. Cedars-Sinai Medical Center, Los Angeles
4. NYU Langone Orthopedic Hospital, New York
5. UCLA Medical Center, Los Angeles
The magazine noted that data for the 2021-2022 Best Hospitals rankings and ratings were not affected by the COVID-19 pandemic, which began after the end of the data collection period.
The methodologies used in determining the rankings are based largely on objective measures, such as risk-adjusted survival, discharge-to-home rates, volume, and quality of nursing, among other care-related indicators.
The full report is available online.
A version of this article first appeared on Medscape.com.
This year’s expanded report debuts new ratings for seven “important procedures and conditions to help patients, in consultation with their doctors, narrow down their choice of hospital based on the specific type of care they need,” Ben Harder, managing editor and chief of health analysis, said in a news release.
With new ratings for myocardial infarction, stroke, hip fracture, and back surgery (spinal fusion), the report now ranks 17 procedures and conditions.
Also new to the 2021 report, which marks the 32nd edition, is a look at racial disparities in health care and the inclusion of health equity measures alongside the hospital rankings.
The new measures examine whether the patients each hospital has treated reflect the racial and ethnic diversity of the surrounding community, among other aspects of health equity.
“At roughly four out of five hospitals, we found that the community’s minority residents were underrepresented among patients receiving services such as joint replacement, cancer surgery and common heart procedures,” Mr. Harder said.
“Against this backdrop, however, we found important exceptions – hospitals that provide care to a disproportionate share of their community’s minority residents. These metrics are just a beginning; we aim to expand on our measurement of health equity in the future,” Mr. Harder added.
Mayo and Cleveland Clinic remain tops
Following the Mayo Clinic, the Cleveland Clinic once again takes the No. 2 spot in the magazine’s latest annual honor roll of best hospitals, which highlights hospitals that deliver exceptional treatment across multiple areas of care.
UCLA Medical Center, Los Angeles, holds the No. 3 spot in 2021. In 2020, UCLA Medical Center and New York–Presbyterian Hospital–Columbia and Cornell, New York, sat in a tie at No. 4.
In 2021, Johns Hopkins Hospital, Baltimore, which held the No. 3 spot in 2020, drops to No. 4, while Massachusetts General Hospital in Boston takes the No. 5 spot, up from No. 6 in 2020.
Rounding out the top 10 (in order) are Cedars-Sinai Medical Center, Los Angeles; New York–Presbyterian Hospital–Columbia and Cornell, New York; NYU Langone Hospitals, New York; UCSF Medical Center, San Francisco; and Northwestern Memorial Hospital, Chicago.
2021-2022 Best Hospitals honor roll
1. Mayo Clinic, Rochester, Minn.
2. Cleveland Clinic, Cleveland
3. UCLA Medical Center, Los Angeles
4. Johns Hopkins Hospital, Baltimore
5. Massachusetts General Hospital, Boston
6. Cedars-Sinai Medical Center, San Francisco
7. New York–Presbyterian Hospital–Columbia and Cornell, New York
8. NYU Langone Hospitals, New York
9. UCSF Medical Center, San Francisco
10. Northwestern Memorial Hospital, Chicago
11. University of Michigan Hospitals–Michigan Medicine, Ann Arbor.
12. Stanford Health Care–Stanford Hospital, Palo Alto, Calif.
13. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
14. Brigham and Women’s Hospital, Boston
15. Mayo Clinic–Phoenix, Phoenix
16. Houston Methodist Hospital, Houston
17. (tie) Barnes-Jewish Hospital, St. Louis
17. (tie) Mount Sinai Hospital, New York Rush University Medical Center, Chicago
19. Rush University Medical Center, Chicago
20. Vanderbilt University Medical Center, Nashville, Tenn.
For the 2021-2022 rankings and ratings, the magazine compared more than 4,750 hospitals nationwide in 15 specialties and 17 procedures and conditions.
At least 2,039 hospitals received a high performance rating in at least one of the services rated; 11 hospitals received high performance in all 17. A total of 175 hospitals were nationally ranked in at least one specialty
For specialty rankings, the University of Texas MD Anderson Cancer Center continues to hold the No. 1 spot in cancer care, the Hospital for Special Surgery continues to be No. 1 in orthopedics, and the Cleveland Clinic continues to be No. 1 in cardiology and heart surgery.
Top five for cancer
1. University of Texas MD Anderson Cancer Center, Houston
2. Memorial Sloan Kettering Cancer Center, New York
3. Mayo Clinic, Rochester, Minn.
4. Dana-Farber/Brigham & Women’s Cancer Center, Boston
5. Cleveland Clinic, Cleveland
Top five for cardiology and heart surgery
1. Cleveland Clinic, Cleveland
2. Mayo Clinic, Rochester, Minn.
3. Cedars-Sinai Medical Center, Los Angeles
4. New York–Presbyterian Hospital–Columbia and Cornell, New York
5. NYU Langone Hospitals, New York
Top five for orthopedics
1. Hospital for Special Surgery, New York
2. Mayo Clinic, Rochester, Minn.
3. Cedars-Sinai Medical Center, Los Angeles
4. NYU Langone Orthopedic Hospital, New York
5. UCLA Medical Center, Los Angeles
The magazine noted that data for the 2021-2022 Best Hospitals rankings and ratings were not affected by the COVID-19 pandemic, which began after the end of the data collection period.
The methodologies used in determining the rankings are based largely on objective measures, such as risk-adjusted survival, discharge-to-home rates, volume, and quality of nursing, among other care-related indicators.
The full report is available online.
A version of this article first appeared on Medscape.com.
This year’s expanded report debuts new ratings for seven “important procedures and conditions to help patients, in consultation with their doctors, narrow down their choice of hospital based on the specific type of care they need,” Ben Harder, managing editor and chief of health analysis, said in a news release.
With new ratings for myocardial infarction, stroke, hip fracture, and back surgery (spinal fusion), the report now ranks 17 procedures and conditions.
Also new to the 2021 report, which marks the 32nd edition, is a look at racial disparities in health care and the inclusion of health equity measures alongside the hospital rankings.
The new measures examine whether the patients each hospital has treated reflect the racial and ethnic diversity of the surrounding community, among other aspects of health equity.
“At roughly four out of five hospitals, we found that the community’s minority residents were underrepresented among patients receiving services such as joint replacement, cancer surgery and common heart procedures,” Mr. Harder said.
“Against this backdrop, however, we found important exceptions – hospitals that provide care to a disproportionate share of their community’s minority residents. These metrics are just a beginning; we aim to expand on our measurement of health equity in the future,” Mr. Harder added.
Mayo and Cleveland Clinic remain tops
Following the Mayo Clinic, the Cleveland Clinic once again takes the No. 2 spot in the magazine’s latest annual honor roll of best hospitals, which highlights hospitals that deliver exceptional treatment across multiple areas of care.
UCLA Medical Center, Los Angeles, holds the No. 3 spot in 2021. In 2020, UCLA Medical Center and New York–Presbyterian Hospital–Columbia and Cornell, New York, sat in a tie at No. 4.
In 2021, Johns Hopkins Hospital, Baltimore, which held the No. 3 spot in 2020, drops to No. 4, while Massachusetts General Hospital in Boston takes the No. 5 spot, up from No. 6 in 2020.
Rounding out the top 10 (in order) are Cedars-Sinai Medical Center, Los Angeles; New York–Presbyterian Hospital–Columbia and Cornell, New York; NYU Langone Hospitals, New York; UCSF Medical Center, San Francisco; and Northwestern Memorial Hospital, Chicago.
2021-2022 Best Hospitals honor roll
1. Mayo Clinic, Rochester, Minn.
2. Cleveland Clinic, Cleveland
3. UCLA Medical Center, Los Angeles
4. Johns Hopkins Hospital, Baltimore
5. Massachusetts General Hospital, Boston
6. Cedars-Sinai Medical Center, San Francisco
7. New York–Presbyterian Hospital–Columbia and Cornell, New York
8. NYU Langone Hospitals, New York
9. UCSF Medical Center, San Francisco
10. Northwestern Memorial Hospital, Chicago
11. University of Michigan Hospitals–Michigan Medicine, Ann Arbor.
12. Stanford Health Care–Stanford Hospital, Palo Alto, Calif.
13. Hospitals of the University of Pennsylvania–Penn Presbyterian, Philadelphia
14. Brigham and Women’s Hospital, Boston
15. Mayo Clinic–Phoenix, Phoenix
16. Houston Methodist Hospital, Houston
17. (tie) Barnes-Jewish Hospital, St. Louis
17. (tie) Mount Sinai Hospital, New York Rush University Medical Center, Chicago
19. Rush University Medical Center, Chicago
20. Vanderbilt University Medical Center, Nashville, Tenn.
For the 2021-2022 rankings and ratings, the magazine compared more than 4,750 hospitals nationwide in 15 specialties and 17 procedures and conditions.
At least 2,039 hospitals received a high performance rating in at least one of the services rated; 11 hospitals received high performance in all 17. A total of 175 hospitals were nationally ranked in at least one specialty
For specialty rankings, the University of Texas MD Anderson Cancer Center continues to hold the No. 1 spot in cancer care, the Hospital for Special Surgery continues to be No. 1 in orthopedics, and the Cleveland Clinic continues to be No. 1 in cardiology and heart surgery.
Top five for cancer
1. University of Texas MD Anderson Cancer Center, Houston
2. Memorial Sloan Kettering Cancer Center, New York
3. Mayo Clinic, Rochester, Minn.
4. Dana-Farber/Brigham & Women’s Cancer Center, Boston
5. Cleveland Clinic, Cleveland
Top five for cardiology and heart surgery
1. Cleveland Clinic, Cleveland
2. Mayo Clinic, Rochester, Minn.
3. Cedars-Sinai Medical Center, Los Angeles
4. New York–Presbyterian Hospital–Columbia and Cornell, New York
5. NYU Langone Hospitals, New York
Top five for orthopedics
1. Hospital for Special Surgery, New York
2. Mayo Clinic, Rochester, Minn.
3. Cedars-Sinai Medical Center, Los Angeles
4. NYU Langone Orthopedic Hospital, New York
5. UCLA Medical Center, Los Angeles
The magazine noted that data for the 2021-2022 Best Hospitals rankings and ratings were not affected by the COVID-19 pandemic, which began after the end of the data collection period.
The methodologies used in determining the rankings are based largely on objective measures, such as risk-adjusted survival, discharge-to-home rates, volume, and quality of nursing, among other care-related indicators.
The full report is available online.
A version of this article first appeared on Medscape.com.
AMA, 55 other groups urge health care vax mandate
As COVID-19 cases, hospitalizations, and deaths mount again across the country, the American Medical Association (AMA), the American Nursing Association, and 54 other
This injunction, issued July 26, covers everyone in healthcare, Emanuel Ezekiel, MD, PhD, chair of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and the organizer of the joint statement, said in an interview.
That includes not only hospitals, but also physician offices, ambulatory surgery centers, home care agencies, skilled nursing facilities, pharmacies, laboratories, and imaging centers, he said.
The exhortation to get vaccinated also extends to federal and state healthcare facilities, including those of the military health system — TRICARE and the Department of Veterans Affairs — which instituted a mandate the same day.
The American Hospital Association (AHA) and other hospital groups recently said they supported hospitals and health systems that required their personnel to get vaccinated. Several dozen healthcare organizations have already done so, including some of the nation’s largest health systems.
A substantial fraction of U.S. healthcare workers have not yet gotten vaccinated, although how many are unvaccinated is unclear. An analysis by WebMD and Medscape Medical News estimated that 25% of hospital workers who had contact with patients were unvaccinated at the end of May.
More than 38% of nursing workers were not fully vaccinated by July 11, according to an analysis of Centers for Medicare & Medicaid Services data by LeadingAge, which was cited by the Washington Post. And more than 40% of nursing home employees have not been fully vaccinated, according to the Centers for Disease Control and Prevention.
The joint statement did not give any indication of how many employees of physician practices have failed to get COVID shots. However, a recent AMA survey shows that 96% of physicians have been fully vaccinated.
Ethical commitment
The main reason for vaccine mandates, according to the healthcare associations’ statement, is “the ethical commitment to put patients as well as residents of long-term care facilities first and take all steps necessary to ensure their health and well-being.”
In addition, the statement noted, vaccination can protect healthcare workers and their families from getting COVID-19.
The statement also pointed out that many healthcare and long-term care organizations already require vaccinations for influenza, hepatitis B, and pertussis.
Workers who have certain medical conditions should be exempt from the vaccination mandates, the statement added.
While recognizing the “historical mistrust of health care institutions” among some healthcare workers, the statement said, “We must continue to address workers’ concerns, engage with marginalized populations, and work with trusted messengers to improve vaccine acceptance.”
There has been some skepticism about the legality of requiring healthcare workers to get vaccinated as a condition of employment, partly because the U.S. Food and Drug Administration has not yet fully authorized any of the COVID-19 vaccines.
But in June, a federal judge turned down a legal challenge to Houston Methodist’s vaccination mandate.
“It is critical that all people in the health care workforce get vaccinated against COVID-19 for the safety of our patients and our colleagues. With more than 300 million doses administered in the United States and nearly 4 billion doses administered worldwide, we know the vaccines are safe and highly effective at preventing severe illness and death from COVID-19.
“Increased vaccinations among health care personnel will not only reduce the spread of COVID-19 but also reduce the harmful toll this virus is taking within the health care workforce and those we are striving to serve,” Susan Bailey, MD, immediate past president of the AMA, said in a news release.
A version of this article first appeared on Medscape.com.
As COVID-19 cases, hospitalizations, and deaths mount again across the country, the American Medical Association (AMA), the American Nursing Association, and 54 other
This injunction, issued July 26, covers everyone in healthcare, Emanuel Ezekiel, MD, PhD, chair of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and the organizer of the joint statement, said in an interview.
That includes not only hospitals, but also physician offices, ambulatory surgery centers, home care agencies, skilled nursing facilities, pharmacies, laboratories, and imaging centers, he said.
The exhortation to get vaccinated also extends to federal and state healthcare facilities, including those of the military health system — TRICARE and the Department of Veterans Affairs — which instituted a mandate the same day.
The American Hospital Association (AHA) and other hospital groups recently said they supported hospitals and health systems that required their personnel to get vaccinated. Several dozen healthcare organizations have already done so, including some of the nation’s largest health systems.
A substantial fraction of U.S. healthcare workers have not yet gotten vaccinated, although how many are unvaccinated is unclear. An analysis by WebMD and Medscape Medical News estimated that 25% of hospital workers who had contact with patients were unvaccinated at the end of May.
More than 38% of nursing workers were not fully vaccinated by July 11, according to an analysis of Centers for Medicare & Medicaid Services data by LeadingAge, which was cited by the Washington Post. And more than 40% of nursing home employees have not been fully vaccinated, according to the Centers for Disease Control and Prevention.
The joint statement did not give any indication of how many employees of physician practices have failed to get COVID shots. However, a recent AMA survey shows that 96% of physicians have been fully vaccinated.
Ethical commitment
The main reason for vaccine mandates, according to the healthcare associations’ statement, is “the ethical commitment to put patients as well as residents of long-term care facilities first and take all steps necessary to ensure their health and well-being.”
In addition, the statement noted, vaccination can protect healthcare workers and their families from getting COVID-19.
The statement also pointed out that many healthcare and long-term care organizations already require vaccinations for influenza, hepatitis B, and pertussis.
Workers who have certain medical conditions should be exempt from the vaccination mandates, the statement added.
While recognizing the “historical mistrust of health care institutions” among some healthcare workers, the statement said, “We must continue to address workers’ concerns, engage with marginalized populations, and work with trusted messengers to improve vaccine acceptance.”
There has been some skepticism about the legality of requiring healthcare workers to get vaccinated as a condition of employment, partly because the U.S. Food and Drug Administration has not yet fully authorized any of the COVID-19 vaccines.
But in June, a federal judge turned down a legal challenge to Houston Methodist’s vaccination mandate.
“It is critical that all people in the health care workforce get vaccinated against COVID-19 for the safety of our patients and our colleagues. With more than 300 million doses administered in the United States and nearly 4 billion doses administered worldwide, we know the vaccines are safe and highly effective at preventing severe illness and death from COVID-19.
“Increased vaccinations among health care personnel will not only reduce the spread of COVID-19 but also reduce the harmful toll this virus is taking within the health care workforce and those we are striving to serve,” Susan Bailey, MD, immediate past president of the AMA, said in a news release.
A version of this article first appeared on Medscape.com.
As COVID-19 cases, hospitalizations, and deaths mount again across the country, the American Medical Association (AMA), the American Nursing Association, and 54 other
This injunction, issued July 26, covers everyone in healthcare, Emanuel Ezekiel, MD, PhD, chair of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and the organizer of the joint statement, said in an interview.
That includes not only hospitals, but also physician offices, ambulatory surgery centers, home care agencies, skilled nursing facilities, pharmacies, laboratories, and imaging centers, he said.
The exhortation to get vaccinated also extends to federal and state healthcare facilities, including those of the military health system — TRICARE and the Department of Veterans Affairs — which instituted a mandate the same day.
The American Hospital Association (AHA) and other hospital groups recently said they supported hospitals and health systems that required their personnel to get vaccinated. Several dozen healthcare organizations have already done so, including some of the nation’s largest health systems.
A substantial fraction of U.S. healthcare workers have not yet gotten vaccinated, although how many are unvaccinated is unclear. An analysis by WebMD and Medscape Medical News estimated that 25% of hospital workers who had contact with patients were unvaccinated at the end of May.
More than 38% of nursing workers were not fully vaccinated by July 11, according to an analysis of Centers for Medicare & Medicaid Services data by LeadingAge, which was cited by the Washington Post. And more than 40% of nursing home employees have not been fully vaccinated, according to the Centers for Disease Control and Prevention.
The joint statement did not give any indication of how many employees of physician practices have failed to get COVID shots. However, a recent AMA survey shows that 96% of physicians have been fully vaccinated.
Ethical commitment
The main reason for vaccine mandates, according to the healthcare associations’ statement, is “the ethical commitment to put patients as well as residents of long-term care facilities first and take all steps necessary to ensure their health and well-being.”
In addition, the statement noted, vaccination can protect healthcare workers and their families from getting COVID-19.
The statement also pointed out that many healthcare and long-term care organizations already require vaccinations for influenza, hepatitis B, and pertussis.
Workers who have certain medical conditions should be exempt from the vaccination mandates, the statement added.
While recognizing the “historical mistrust of health care institutions” among some healthcare workers, the statement said, “We must continue to address workers’ concerns, engage with marginalized populations, and work with trusted messengers to improve vaccine acceptance.”
There has been some skepticism about the legality of requiring healthcare workers to get vaccinated as a condition of employment, partly because the U.S. Food and Drug Administration has not yet fully authorized any of the COVID-19 vaccines.
But in June, a federal judge turned down a legal challenge to Houston Methodist’s vaccination mandate.
“It is critical that all people in the health care workforce get vaccinated against COVID-19 for the safety of our patients and our colleagues. With more than 300 million doses administered in the United States and nearly 4 billion doses administered worldwide, we know the vaccines are safe and highly effective at preventing severe illness and death from COVID-19.
“Increased vaccinations among health care personnel will not only reduce the spread of COVID-19 but also reduce the harmful toll this virus is taking within the health care workforce and those we are striving to serve,” Susan Bailey, MD, immediate past president of the AMA, said in a news release.
A version of this article first appeared on Medscape.com.
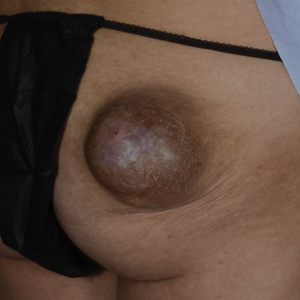
Exophytic Tumor on the Buttock
The Diagnosis: Hidradenocarcinoma
An excisional biopsy revealed a neoplasm in the dermis with focal invasion into the adjacent soft tissue (Figure 1). The tumor consisted of sheets of cells with cytoplasmic vacuoles and ductal differentiation (Figure 2), as well as cells with mild atypia, mild pleomorphism, rare mitotic figures, and abundant pale cytoplasm. Immunohistochemical staining was positive for cytokeratin (CK) 5, CK7, CK20, CK AE1/AE3, and p63 (Figure 3). The culmination of features including the large tumor size, immunohistochemical staining pattern, and mild pleomorphism with focal invasion into the soft tissue supported the diagnosis of hidradenocarcinoma.

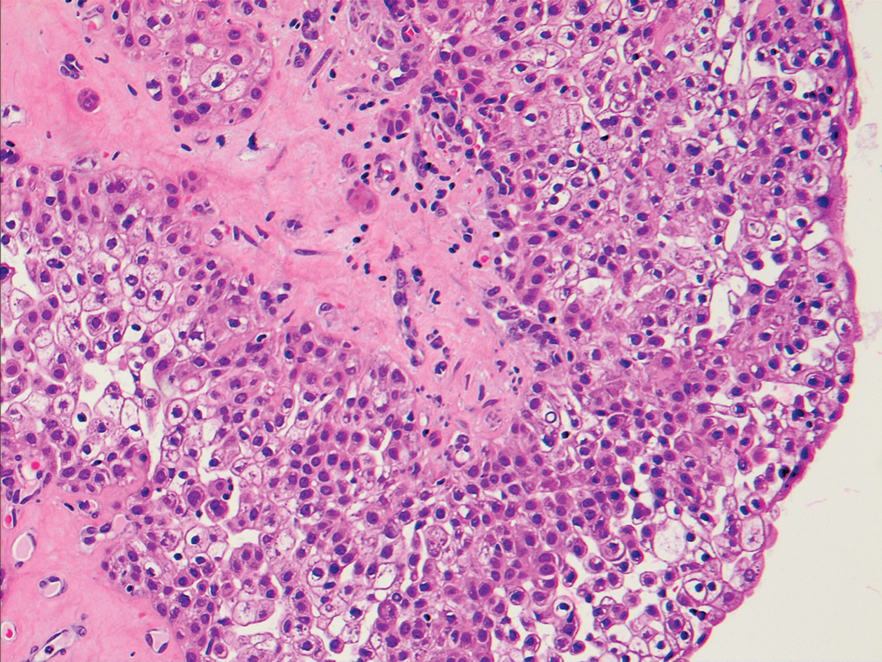
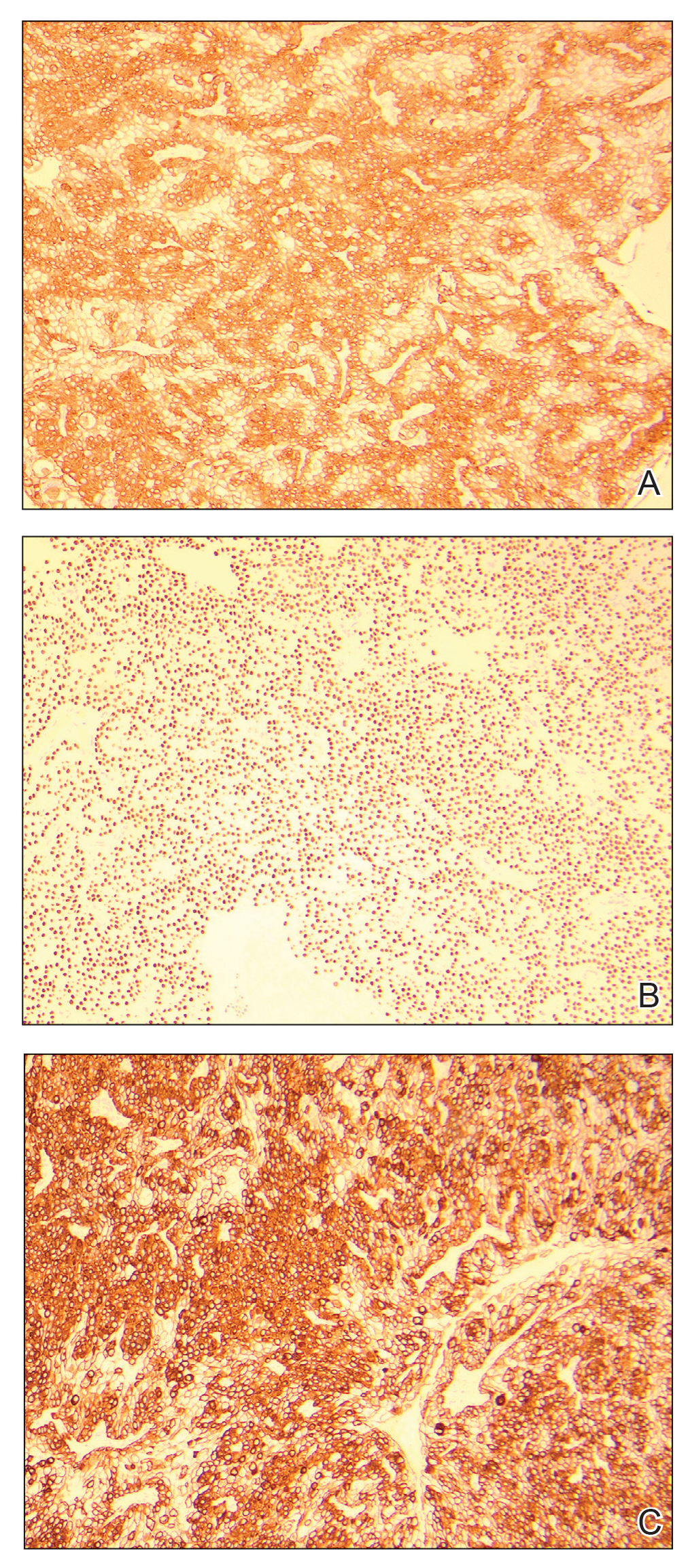
Hidradenocarcinoma is an exceedingly rare malignant tumor of eccrine and/or apocrine origin.1 It accounts for less than 0.001% of all tumors and 1 of 13,000 skin biopsies.2 It usually arises in the head and neck region and most commonly affects older adults aged 50 to 70 years.3 The size of hidradenocarcinomas can vary; however, they typically are large, often growing to be greater than 5 cm in diameter.2 It tends to be an aggressive tumor that generally spreads to regional lymph nodes and distant viscera.4 Although it most commonly arises de novo, it may occasionally derive from a benign hidradenoma.1 The diagnosis of hidradenocarcinoma is made based on the tumor’s morphologic and pathologic characteristics. Histologically, it is characterized by an infiltrative and invasive proliferation of lobules made of large clear cells with atypical mitotic figures and nuclear pleomorphism as well as immunohistochemical features displaying various positive markers, such as carcinoembryonic antigen, epithelial membrane antigen, S-100 protein, and CKs AE1/AE3 and 5/6.2 Invasion of the adjacent soft tissue can be present and helps to confirm the diagnosis.
The differential diagnosis for hidradenocarcinoma primarily is the benign hidradenoma, which is similar both clinically and histologically with a few important differences. Hidradenocarcinomas often are larger and ulcerated. Histologically, they usually are more pleomorphic with the presence of mitotic figures in clear cells and tend to invade locally into the surrounding soft tissue. Other similar lesions such as spiradenoma, Merkel cell carcinoma, lymphangioma, cutaneous Crohn disease, tumors metastatic to the skin, and metastatic clear cell carcinomas originating from other organs also are included in the differential diagnosis.2
Spiradenomas are dermal tumors originating from the sweat glands. They typically present as bluish, painful, solitary nodules on the ventral surfaces of the upper body, though multiple nodules also are reported.5 Spiradenomas manifest as a central constellation of pale large cells surrounded by small, dark, basaloid cells containing hyperchromatic nuclei. The microscopic appearance of the blue basaloid cells contrasts with the clear cells seen in hidradenoma.5
Merkel cell carcinoma is a cutaneous neuroendocrine tumor affecting elderly or immunosuppressed individuals. It arises in sun-exposed areas and often is associated with Merkel cell polyomavirus infection. The histologic features display small and round cells that stain positive for CK8, CK18, CK19, and CK20 but stain negative for CK7, a marker that often is positive in hidradenocarcinoma.6
Lymphangioma, particularly cavernous lymphangioma, may resemble the gross appearance of hidradenoma/ hidradenocarcinoma. It usually presents as irregular clear blue papules and nodules in the skin and subcutaneous tissue.7 The key histopathologic finding in this tumor is the endothelium-lined channels that stain positive for D2-40, a lymphatic endothelium marker.7,8
Cutaneous Crohn disease is classified as noncaseating granulomatous skin lesions that are noncontinuous with the gastrointestinal tract.9 Clinical presentations in addition to skin edema include erythematous plaques, ulcerations, and erosions. Histopathology reveals sterile noncaseating granulomas made of Langerhans giant cells, epithelioid histocytes, and plasma cells.9
Metastatic clear cell carcinomas, such as renal cell carcinoma, can be differentiated by a history of primary carcinoma, demonstration of histologic vascular stroma, and other features related to metastatic clear cell carcinoma.2
There are no well-established therapeutic guidelines for hidradenocarcinoma. Wide local excision with margins greater than 2 cm is the preferred initial treatment and often is performed in conjunction with sentinel lymph node biopsy. External beam radiotherapy and adjunctive chemotherapy have been used for tumors that could not be surgically cleared. However, the efficacy of these treatments has not been well established.2 Targeted therapies recently have emerged as an alternative treatment choice for hidradenocarcinoma due to the utilization of immunohistochemical and genomic testing. The discovery of specific gene mutations or the expression of hormonal receptors in this tumor have paved the way for targeting HER2-expressing hidradenocarcinomas with trastuzumab and those expressing estrogen receptor with the estrogen receptor inhibitor tamoxifen.1 Epidermal growth factor receptor inhibitors and PI3K/Akt/mTOR (phosphatidylinositol-3-kinase/AKT/mammalian target of rapamycin) pathway inhibitors also have been used to target various signal transduction pathways.2
Wide excision with 2.5-cm margins was performed on our patient, and a positron emission tomography– computed tomography scan revealed no metastatic disease. She declined sentinel lymph node biopsy and additional treatment. Due to the risk for recurrence, she was monitored closely with skin examinations and positron emission tomography–computed tomography every 3 months for the first year and every 6 months thereafter. Thus far, she has had no evidence of local or regional recurrence.
- Miller DH, Peterson JL, Buskirk SJ, et al. Management of metastatic apocrine hidradenocarcinoma with chemotherapy and radiation. Rare Tumors. 2015;7:6082.
- Soni A, Bansal N, Kaushal V, et al. Current management approach to hidradenocarcinoma: a comprehensive review of the literature. Ecancermedicalscience. 2015;9:517.
- Jinnah AH, Emory CL, Mai NH, et al. Hidradenocarcinoma presenting as soft tissue mass: case report with cytomorphologic description, histologic correlation, and differential diagnosis. Diagn Cytopathol. 2016;44:438-441.
- Khan BM, Mansha MA, Ali N, et al. Hidradenocarcinoma: five years of local and systemic control of a rare sweat gland neoplasm with nodal metastasis. Cureus. 2018;10:E2884.
- Miceli A, Ferrer-Bruker SJ. Spiradenoma. StatPearls. StatPearls Publishing; 2019.
- Banks PD, Sandhu S, Gyorki DE, et al. Recent insights and advances in the management of Merkel cell carcinoma. J Oncol Pract. 2016; 12:637-646.
- Flanagan BP, Helwig EB. Cutaneous lymphangioma. Arch Dermatol. 1977;113:24-30.
- Kalof AN, Cooper K. D2-40 immunohistochemistry—so far! Adv Anat Pathol. 2009;16:62-64.
- Schneider SL, Foster K, Patel D, et al. Cutaneous manifestations of metastatic Crohn’s disease. Pediatr Dermatol. 2018;35:566-574.
The Diagnosis: Hidradenocarcinoma
An excisional biopsy revealed a neoplasm in the dermis with focal invasion into the adjacent soft tissue (Figure 1). The tumor consisted of sheets of cells with cytoplasmic vacuoles and ductal differentiation (Figure 2), as well as cells with mild atypia, mild pleomorphism, rare mitotic figures, and abundant pale cytoplasm. Immunohistochemical staining was positive for cytokeratin (CK) 5, CK7, CK20, CK AE1/AE3, and p63 (Figure 3). The culmination of features including the large tumor size, immunohistochemical staining pattern, and mild pleomorphism with focal invasion into the soft tissue supported the diagnosis of hidradenocarcinoma.



Hidradenocarcinoma is an exceedingly rare malignant tumor of eccrine and/or apocrine origin.1 It accounts for less than 0.001% of all tumors and 1 of 13,000 skin biopsies.2 It usually arises in the head and neck region and most commonly affects older adults aged 50 to 70 years.3 The size of hidradenocarcinomas can vary; however, they typically are large, often growing to be greater than 5 cm in diameter.2 It tends to be an aggressive tumor that generally spreads to regional lymph nodes and distant viscera.4 Although it most commonly arises de novo, it may occasionally derive from a benign hidradenoma.1 The diagnosis of hidradenocarcinoma is made based on the tumor’s morphologic and pathologic characteristics. Histologically, it is characterized by an infiltrative and invasive proliferation of lobules made of large clear cells with atypical mitotic figures and nuclear pleomorphism as well as immunohistochemical features displaying various positive markers, such as carcinoembryonic antigen, epithelial membrane antigen, S-100 protein, and CKs AE1/AE3 and 5/6.2 Invasion of the adjacent soft tissue can be present and helps to confirm the diagnosis.
The differential diagnosis for hidradenocarcinoma primarily is the benign hidradenoma, which is similar both clinically and histologically with a few important differences. Hidradenocarcinomas often are larger and ulcerated. Histologically, they usually are more pleomorphic with the presence of mitotic figures in clear cells and tend to invade locally into the surrounding soft tissue. Other similar lesions such as spiradenoma, Merkel cell carcinoma, lymphangioma, cutaneous Crohn disease, tumors metastatic to the skin, and metastatic clear cell carcinomas originating from other organs also are included in the differential diagnosis.2
Spiradenomas are dermal tumors originating from the sweat glands. They typically present as bluish, painful, solitary nodules on the ventral surfaces of the upper body, though multiple nodules also are reported.5 Spiradenomas manifest as a central constellation of pale large cells surrounded by small, dark, basaloid cells containing hyperchromatic nuclei. The microscopic appearance of the blue basaloid cells contrasts with the clear cells seen in hidradenoma.5
Merkel cell carcinoma is a cutaneous neuroendocrine tumor affecting elderly or immunosuppressed individuals. It arises in sun-exposed areas and often is associated with Merkel cell polyomavirus infection. The histologic features display small and round cells that stain positive for CK8, CK18, CK19, and CK20 but stain negative for CK7, a marker that often is positive in hidradenocarcinoma.6
Lymphangioma, particularly cavernous lymphangioma, may resemble the gross appearance of hidradenoma/ hidradenocarcinoma. It usually presents as irregular clear blue papules and nodules in the skin and subcutaneous tissue.7 The key histopathologic finding in this tumor is the endothelium-lined channels that stain positive for D2-40, a lymphatic endothelium marker.7,8
Cutaneous Crohn disease is classified as noncaseating granulomatous skin lesions that are noncontinuous with the gastrointestinal tract.9 Clinical presentations in addition to skin edema include erythematous plaques, ulcerations, and erosions. Histopathology reveals sterile noncaseating granulomas made of Langerhans giant cells, epithelioid histocytes, and plasma cells.9
Metastatic clear cell carcinomas, such as renal cell carcinoma, can be differentiated by a history of primary carcinoma, demonstration of histologic vascular stroma, and other features related to metastatic clear cell carcinoma.2
There are no well-established therapeutic guidelines for hidradenocarcinoma. Wide local excision with margins greater than 2 cm is the preferred initial treatment and often is performed in conjunction with sentinel lymph node biopsy. External beam radiotherapy and adjunctive chemotherapy have been used for tumors that could not be surgically cleared. However, the efficacy of these treatments has not been well established.2 Targeted therapies recently have emerged as an alternative treatment choice for hidradenocarcinoma due to the utilization of immunohistochemical and genomic testing. The discovery of specific gene mutations or the expression of hormonal receptors in this tumor have paved the way for targeting HER2-expressing hidradenocarcinomas with trastuzumab and those expressing estrogen receptor with the estrogen receptor inhibitor tamoxifen.1 Epidermal growth factor receptor inhibitors and PI3K/Akt/mTOR (phosphatidylinositol-3-kinase/AKT/mammalian target of rapamycin) pathway inhibitors also have been used to target various signal transduction pathways.2
Wide excision with 2.5-cm margins was performed on our patient, and a positron emission tomography– computed tomography scan revealed no metastatic disease. She declined sentinel lymph node biopsy and additional treatment. Due to the risk for recurrence, she was monitored closely with skin examinations and positron emission tomography–computed tomography every 3 months for the first year and every 6 months thereafter. Thus far, she has had no evidence of local or regional recurrence.
The Diagnosis: Hidradenocarcinoma
An excisional biopsy revealed a neoplasm in the dermis with focal invasion into the adjacent soft tissue (Figure 1). The tumor consisted of sheets of cells with cytoplasmic vacuoles and ductal differentiation (Figure 2), as well as cells with mild atypia, mild pleomorphism, rare mitotic figures, and abundant pale cytoplasm. Immunohistochemical staining was positive for cytokeratin (CK) 5, CK7, CK20, CK AE1/AE3, and p63 (Figure 3). The culmination of features including the large tumor size, immunohistochemical staining pattern, and mild pleomorphism with focal invasion into the soft tissue supported the diagnosis of hidradenocarcinoma.



Hidradenocarcinoma is an exceedingly rare malignant tumor of eccrine and/or apocrine origin.1 It accounts for less than 0.001% of all tumors and 1 of 13,000 skin biopsies.2 It usually arises in the head and neck region and most commonly affects older adults aged 50 to 70 years.3 The size of hidradenocarcinomas can vary; however, they typically are large, often growing to be greater than 5 cm in diameter.2 It tends to be an aggressive tumor that generally spreads to regional lymph nodes and distant viscera.4 Although it most commonly arises de novo, it may occasionally derive from a benign hidradenoma.1 The diagnosis of hidradenocarcinoma is made based on the tumor’s morphologic and pathologic characteristics. Histologically, it is characterized by an infiltrative and invasive proliferation of lobules made of large clear cells with atypical mitotic figures and nuclear pleomorphism as well as immunohistochemical features displaying various positive markers, such as carcinoembryonic antigen, epithelial membrane antigen, S-100 protein, and CKs AE1/AE3 and 5/6.2 Invasion of the adjacent soft tissue can be present and helps to confirm the diagnosis.
The differential diagnosis for hidradenocarcinoma primarily is the benign hidradenoma, which is similar both clinically and histologically with a few important differences. Hidradenocarcinomas often are larger and ulcerated. Histologically, they usually are more pleomorphic with the presence of mitotic figures in clear cells and tend to invade locally into the surrounding soft tissue. Other similar lesions such as spiradenoma, Merkel cell carcinoma, lymphangioma, cutaneous Crohn disease, tumors metastatic to the skin, and metastatic clear cell carcinomas originating from other organs also are included in the differential diagnosis.2
Spiradenomas are dermal tumors originating from the sweat glands. They typically present as bluish, painful, solitary nodules on the ventral surfaces of the upper body, though multiple nodules also are reported.5 Spiradenomas manifest as a central constellation of pale large cells surrounded by small, dark, basaloid cells containing hyperchromatic nuclei. The microscopic appearance of the blue basaloid cells contrasts with the clear cells seen in hidradenoma.5
Merkel cell carcinoma is a cutaneous neuroendocrine tumor affecting elderly or immunosuppressed individuals. It arises in sun-exposed areas and often is associated with Merkel cell polyomavirus infection. The histologic features display small and round cells that stain positive for CK8, CK18, CK19, and CK20 but stain negative for CK7, a marker that often is positive in hidradenocarcinoma.6
Lymphangioma, particularly cavernous lymphangioma, may resemble the gross appearance of hidradenoma/ hidradenocarcinoma. It usually presents as irregular clear blue papules and nodules in the skin and subcutaneous tissue.7 The key histopathologic finding in this tumor is the endothelium-lined channels that stain positive for D2-40, a lymphatic endothelium marker.7,8
Cutaneous Crohn disease is classified as noncaseating granulomatous skin lesions that are noncontinuous with the gastrointestinal tract.9 Clinical presentations in addition to skin edema include erythematous plaques, ulcerations, and erosions. Histopathology reveals sterile noncaseating granulomas made of Langerhans giant cells, epithelioid histocytes, and plasma cells.9
Metastatic clear cell carcinomas, such as renal cell carcinoma, can be differentiated by a history of primary carcinoma, demonstration of histologic vascular stroma, and other features related to metastatic clear cell carcinoma.2
There are no well-established therapeutic guidelines for hidradenocarcinoma. Wide local excision with margins greater than 2 cm is the preferred initial treatment and often is performed in conjunction with sentinel lymph node biopsy. External beam radiotherapy and adjunctive chemotherapy have been used for tumors that could not be surgically cleared. However, the efficacy of these treatments has not been well established.2 Targeted therapies recently have emerged as an alternative treatment choice for hidradenocarcinoma due to the utilization of immunohistochemical and genomic testing. The discovery of specific gene mutations or the expression of hormonal receptors in this tumor have paved the way for targeting HER2-expressing hidradenocarcinomas with trastuzumab and those expressing estrogen receptor with the estrogen receptor inhibitor tamoxifen.1 Epidermal growth factor receptor inhibitors and PI3K/Akt/mTOR (phosphatidylinositol-3-kinase/AKT/mammalian target of rapamycin) pathway inhibitors also have been used to target various signal transduction pathways.2
Wide excision with 2.5-cm margins was performed on our patient, and a positron emission tomography– computed tomography scan revealed no metastatic disease. She declined sentinel lymph node biopsy and additional treatment. Due to the risk for recurrence, she was monitored closely with skin examinations and positron emission tomography–computed tomography every 3 months for the first year and every 6 months thereafter. Thus far, she has had no evidence of local or regional recurrence.
- Miller DH, Peterson JL, Buskirk SJ, et al. Management of metastatic apocrine hidradenocarcinoma with chemotherapy and radiation. Rare Tumors. 2015;7:6082.
- Soni A, Bansal N, Kaushal V, et al. Current management approach to hidradenocarcinoma: a comprehensive review of the literature. Ecancermedicalscience. 2015;9:517.
- Jinnah AH, Emory CL, Mai NH, et al. Hidradenocarcinoma presenting as soft tissue mass: case report with cytomorphologic description, histologic correlation, and differential diagnosis. Diagn Cytopathol. 2016;44:438-441.
- Khan BM, Mansha MA, Ali N, et al. Hidradenocarcinoma: five years of local and systemic control of a rare sweat gland neoplasm with nodal metastasis. Cureus. 2018;10:E2884.
- Miceli A, Ferrer-Bruker SJ. Spiradenoma. StatPearls. StatPearls Publishing; 2019.
- Banks PD, Sandhu S, Gyorki DE, et al. Recent insights and advances in the management of Merkel cell carcinoma. J Oncol Pract. 2016; 12:637-646.
- Flanagan BP, Helwig EB. Cutaneous lymphangioma. Arch Dermatol. 1977;113:24-30.
- Kalof AN, Cooper K. D2-40 immunohistochemistry—so far! Adv Anat Pathol. 2009;16:62-64.
- Schneider SL, Foster K, Patel D, et al. Cutaneous manifestations of metastatic Crohn’s disease. Pediatr Dermatol. 2018;35:566-574.
- Miller DH, Peterson JL, Buskirk SJ, et al. Management of metastatic apocrine hidradenocarcinoma with chemotherapy and radiation. Rare Tumors. 2015;7:6082.
- Soni A, Bansal N, Kaushal V, et al. Current management approach to hidradenocarcinoma: a comprehensive review of the literature. Ecancermedicalscience. 2015;9:517.
- Jinnah AH, Emory CL, Mai NH, et al. Hidradenocarcinoma presenting as soft tissue mass: case report with cytomorphologic description, histologic correlation, and differential diagnosis. Diagn Cytopathol. 2016;44:438-441.
- Khan BM, Mansha MA, Ali N, et al. Hidradenocarcinoma: five years of local and systemic control of a rare sweat gland neoplasm with nodal metastasis. Cureus. 2018;10:E2884.
- Miceli A, Ferrer-Bruker SJ. Spiradenoma. StatPearls. StatPearls Publishing; 2019.
- Banks PD, Sandhu S, Gyorki DE, et al. Recent insights and advances in the management of Merkel cell carcinoma. J Oncol Pract. 2016; 12:637-646.
- Flanagan BP, Helwig EB. Cutaneous lymphangioma. Arch Dermatol. 1977;113:24-30.
- Kalof AN, Cooper K. D2-40 immunohistochemistry—so far! Adv Anat Pathol. 2009;16:62-64.
- Schneider SL, Foster K, Patel D, et al. Cutaneous manifestations of metastatic Crohn’s disease. Pediatr Dermatol. 2018;35:566-574.
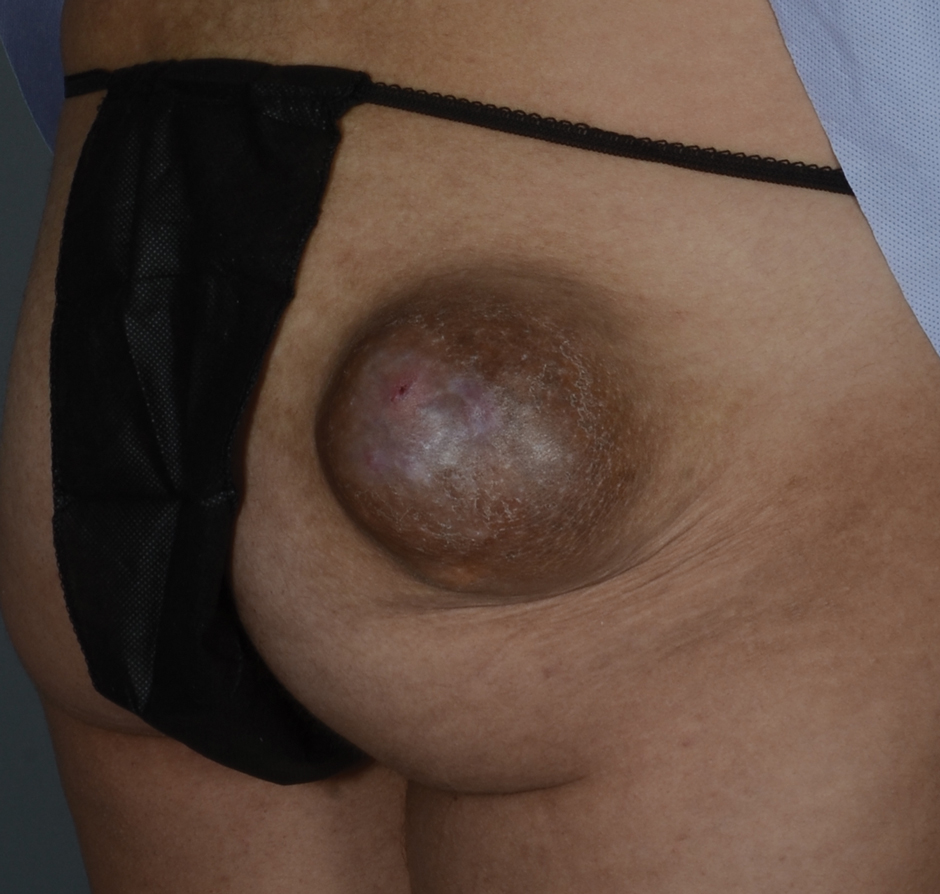
A 20-year-old woman with no notable medical history presented to the dermatology clinic with an enlarging mass on the right buttock that had been growing over the course of several years. The mass progressed from a small, mildly tender nodule to a 10×10-cm, hyperpigmented, exophytic tumor. There were no other abnormal findings on physical examination, and the patient denied any systemic symptoms.
A clarion call for regulating PBMs: Health care groups, states push back on legal challenges
Mark Nelson, PharmD, recalls the anguish when a major pharmacy benefit manager (PBM) moved all veteran patients with prostate cancer at his facility from an effective medication to a pricier alternative therapy. “All of these patients were stable on their therapy and were extremely distraught about their medications being changed,” said Dr. Nelson, CEO of Northwest Medical Specialties in Washington State. While there was no clinical reason to change the medication, “our oncologists had no choice other than to comply,” he said.
It’s unclear why a PBM would switch to a more expensive medication that has no additional clinical benefit, he continued. “Why upset so many veterans? For what reason? We were not given a reason despite our very vocal protest.”
Angus B. Worthing, MD, sees these scenarios unfold every day in his rheumatology practice in the Washington, D.C., area. “In my clinic with 25 doctors, we have three full-time people that only handle PBMs,” he said in an interview. He and others in the medical community, as well as many states, have been pushing back on what they see as efforts by PBMs to raise drug prices and collect the profits at the expense of patients.
PCMA’s challenges against PBM law
The Pharmaceutical Care Management Association (PCMA), a trade group that represents PBMs, has sued at least a half dozen states on their ability to regulate PBMs. However, a landmark case in late 2020 (Pharmaceutical Care Management Association v. Rutledge) set a new precedent. Reversing a lower appeals court decision, the Supreme Court unanimously ruled in favor of allowing states to put in place fair regulation of these entities.
Dr. Worthing and others hope that the medical community and states can leverage this ruling in another lawsuit PCMA brought against North Dakota (PCMA v. Wehbi). PCMA filed this lawsuit in 2017, which challenges two statutes on PBM regulation. The group has issued similar legal challenges in Maine, the District of Columbia, Iowa, Oklahoma, and Arkansas with the Rutledge case.
“PBMs have become massive profit centers while (ironically) increasing patients’ out-of-pocket costs, interfering with doctor-patient relationships, and impairing patient access to appropriate treatment,” according to an amicus brief filed by The Alliance for Transparent & Affordable Prescriptions (ATAP), the Community Oncology Alliance (COA), and American Pharmacies, supporting North Dakota in the Wehbi case.
This is to ensure the case represents the voices of physicians, patients, nurses, and other stakeholders, and underscores PBM abuses, said Dr. Worthing, vice president of ATAP. He also serves as the American College of Rheumatology’s representative on ATAP’s Executive Committee.
PCMA did not respond to requests for comment. Its CEO and president, J.C. Scott, emphasizes that PBMs have a long track record of reducing drug costs for patients and plan sponsors. In 2021, PCMA released 21 policy solutions, a set of industry principles and a three-part policy platform, all with an aim to bring down costs and increase access to pharmaceutical care, according to the organization.
PCMA estimates that the strategies in its platform (updating Medicare Part D, accelerating value-based care, and eliminating anticompetitive ‘pay for delay’ agreements) would save the federal government a maximum of $398.7 billion over 10 years.
According to Wendy Hemmen, senior director with Texas Oncology in Dallas, PBMs do their own unique calculations to arrive at their cost reductions. “Essentially in a PBM, they use things that make their story. Numbers reported to plan sponsors and to the public are not audited and are usually in terms of percentages or a per member per month. Data points are moved around, dropped, or reclassified to make the story that the PBM needs to tell,” Ms. Hemmen said.
Amicus briefs dispute ERISA connection
North Dakota legislation prohibits PBMs from charging copays to patients that exceed the cost of a drug. It also prohibits gag clause provisions that restrict what pharmacists may discuss with patients. PBMs may charge fees based on performance metrics, but they must use nationally recognized metrics. Fees must be disclosed at the point of sale.
In its legal challenges, PCMA has asserted that state laws violate the preemption clause in the Employee Retirement Income Security Act (ERISA). “Federal preemption allows employers flexibility to administer innovative benefit plans in an environment of increasing health care costs. The court’s decision in Rutledge v. PCMA will either uphold or threaten these federal protections,” PCMA asserted in a statement issued in March 2020.
ATAP’s amicus brief, and another one filed by 34 attorneys general that supports the North Dakota statute to regulate PBMs, counter that this isn’t the case.
“First, PBM regulation (in its common and standard form) does not reference ERISA itself. These laws leave all plans on equal footing; they do not single out ERISA plans for preferred or disfavored coverage, and they do not change the playing field for ERISA plans alone ... Second, PBM regulation does not have any prohibited connection with ERISA plans,” noted authors in the ATAP brief.
PCMA has also included Medicare preemption in its arguments against PBM regulation. This is meritless, wrote the state attorneys general. “Medicare preempts state laws only if a Medicare ‘standard’ particularly addresses the subject of state regulation. Because the challenged North Dakota laws do not dictate plan benefits or conflict with a Medicare standard, they are not preempted.”
The auctioning of medications
PBMs in theory could use their market power to drive down costs by extracting discounts from drug makers and pharmacies. In reality, they retain any price concessions and discounts for themselves, ATAP’s brief continued.
A system that PBMs have put into place, called step therapy, is essentially an auction for the preferred spot that will be authorized and covered, Dr. Worthing explained.
PBMs create formularies through this auction. The highest rebate to the PBM earns the top spot in the auction and becomes the preferred drug. “That highest bid gets paid for by passing the cost along to patients and insurance plans, and PBMs pocket the profits. This provides an incentive for pharmaceutical manufacturers to raise prices,” he said.
Dr. Worthing has seen these practices trickle down and affect his patients. “Frequently, the medication I prescribe based on what’s best for the patient based on their disease activity, values, and medical history is often not covered because a different drug or portfolio of drugs has earned the top spot in step therapy. This is an extremely frustrating and cumbersome process that not only delays access to treatments but also provides an incentive for higher drug prices,” he said.
There are other ways in which PBMs get in the way of care, said Ms. Hemmen, whose facility serves complex-care oncology patients.
“PBMs force scripts out of higher-quality pharmacies that preserve unfragmented care. They incentivize plan sponsors to put programs into place that take away patient choice, fragment care, and drive scripts to their own owned pharmacies,” she said.
Rutledge case sets precedent
In the Rutledge case, PCMA had challenged an Arkansas law that forbid PBMs from paying local pharmacies at a lower rate than what the pharmacies were reimbursed to fill prescriptions. Although the 8th Circuit Court of Appeals agreed with PCMA, the Supreme Court ruled in favor of Arkansas in late 2020.
The appeals court also backed PCMA in PCMA v. Wehbi. However, the Supreme Court vacated this decision and remanded it back to the appeals court, asking for a reconsideration in wake of the outcome in Rutledge v. PCMA.
PCMA has argued that Rutledge was a narrow decision, limited to state laws that regulate PBM reimbursements, and that Rutledge has no bearing on North Dakota law.
While it’s unfortunate that PCMA is trying to delay implementation of sensible regulations, “a lot of us are happy that this issue is coming to light,” Dr. Worthing said. “As a rheumatologist and health policy advocate, exposing drug middlemen is the most important bipartisan issue in the country today because it gets at the core of making sure that sick people get access to the medications they need and reducing the budget of insurance carriers, hospitals, and the federal budget.”
The ATAP brief noted that 28 state attorneys general have filed suit against PBMs, “securing settlements compelling PBMs to correct deceptive trade practices.”
Many people at the state and local level were waiting for the Supreme Court to decide on Rutledge before enacting legislation and sensible regulations, and now they can go ahead and do it, said Dr. Worthing. “I expect to see this across the country as states look at budgets, and as patients bring personal stories to light. We look forward to states passing these kinds of laws to regulate PBMs.”
The ACR doesn’t anticipate a ruling in the Wehbi case until the spring of 2022.
Recent laws passed around PBMs and the pharmacy benefit are a good first step in holding PBMs accountable for quality of care and honoring patient choice, Ms. Hemmen said. The laws also begin to address the fiscal manipulations PBMs use to gain advantage and direct scripts to their own coffers, she added. However, this may not have enough teeth. “These state laws are coming from a provider perspective, and they don’t anticipate what PBMs will do in response. The PBMs are going to work around it.”
Mark Nelson, PharmD, recalls the anguish when a major pharmacy benefit manager (PBM) moved all veteran patients with prostate cancer at his facility from an effective medication to a pricier alternative therapy. “All of these patients were stable on their therapy and were extremely distraught about their medications being changed,” said Dr. Nelson, CEO of Northwest Medical Specialties in Washington State. While there was no clinical reason to change the medication, “our oncologists had no choice other than to comply,” he said.
It’s unclear why a PBM would switch to a more expensive medication that has no additional clinical benefit, he continued. “Why upset so many veterans? For what reason? We were not given a reason despite our very vocal protest.”
Angus B. Worthing, MD, sees these scenarios unfold every day in his rheumatology practice in the Washington, D.C., area. “In my clinic with 25 doctors, we have three full-time people that only handle PBMs,” he said in an interview. He and others in the medical community, as well as many states, have been pushing back on what they see as efforts by PBMs to raise drug prices and collect the profits at the expense of patients.
PCMA’s challenges against PBM law
The Pharmaceutical Care Management Association (PCMA), a trade group that represents PBMs, has sued at least a half dozen states on their ability to regulate PBMs. However, a landmark case in late 2020 (Pharmaceutical Care Management Association v. Rutledge) set a new precedent. Reversing a lower appeals court decision, the Supreme Court unanimously ruled in favor of allowing states to put in place fair regulation of these entities.
Dr. Worthing and others hope that the medical community and states can leverage this ruling in another lawsuit PCMA brought against North Dakota (PCMA v. Wehbi). PCMA filed this lawsuit in 2017, which challenges two statutes on PBM regulation. The group has issued similar legal challenges in Maine, the District of Columbia, Iowa, Oklahoma, and Arkansas with the Rutledge case.
“PBMs have become massive profit centers while (ironically) increasing patients’ out-of-pocket costs, interfering with doctor-patient relationships, and impairing patient access to appropriate treatment,” according to an amicus brief filed by The Alliance for Transparent & Affordable Prescriptions (ATAP), the Community Oncology Alliance (COA), and American Pharmacies, supporting North Dakota in the Wehbi case.
This is to ensure the case represents the voices of physicians, patients, nurses, and other stakeholders, and underscores PBM abuses, said Dr. Worthing, vice president of ATAP. He also serves as the American College of Rheumatology’s representative on ATAP’s Executive Committee.
PCMA did not respond to requests for comment. Its CEO and president, J.C. Scott, emphasizes that PBMs have a long track record of reducing drug costs for patients and plan sponsors. In 2021, PCMA released 21 policy solutions, a set of industry principles and a three-part policy platform, all with an aim to bring down costs and increase access to pharmaceutical care, according to the organization.
PCMA estimates that the strategies in its platform (updating Medicare Part D, accelerating value-based care, and eliminating anticompetitive ‘pay for delay’ agreements) would save the federal government a maximum of $398.7 billion over 10 years.
According to Wendy Hemmen, senior director with Texas Oncology in Dallas, PBMs do their own unique calculations to arrive at their cost reductions. “Essentially in a PBM, they use things that make their story. Numbers reported to plan sponsors and to the public are not audited and are usually in terms of percentages or a per member per month. Data points are moved around, dropped, or reclassified to make the story that the PBM needs to tell,” Ms. Hemmen said.
Amicus briefs dispute ERISA connection
North Dakota legislation prohibits PBMs from charging copays to patients that exceed the cost of a drug. It also prohibits gag clause provisions that restrict what pharmacists may discuss with patients. PBMs may charge fees based on performance metrics, but they must use nationally recognized metrics. Fees must be disclosed at the point of sale.
In its legal challenges, PCMA has asserted that state laws violate the preemption clause in the Employee Retirement Income Security Act (ERISA). “Federal preemption allows employers flexibility to administer innovative benefit plans in an environment of increasing health care costs. The court’s decision in Rutledge v. PCMA will either uphold or threaten these federal protections,” PCMA asserted in a statement issued in March 2020.
ATAP’s amicus brief, and another one filed by 34 attorneys general that supports the North Dakota statute to regulate PBMs, counter that this isn’t the case.
“First, PBM regulation (in its common and standard form) does not reference ERISA itself. These laws leave all plans on equal footing; they do not single out ERISA plans for preferred or disfavored coverage, and they do not change the playing field for ERISA plans alone ... Second, PBM regulation does not have any prohibited connection with ERISA plans,” noted authors in the ATAP brief.
PCMA has also included Medicare preemption in its arguments against PBM regulation. This is meritless, wrote the state attorneys general. “Medicare preempts state laws only if a Medicare ‘standard’ particularly addresses the subject of state regulation. Because the challenged North Dakota laws do not dictate plan benefits or conflict with a Medicare standard, they are not preempted.”
The auctioning of medications
PBMs in theory could use their market power to drive down costs by extracting discounts from drug makers and pharmacies. In reality, they retain any price concessions and discounts for themselves, ATAP’s brief continued.
A system that PBMs have put into place, called step therapy, is essentially an auction for the preferred spot that will be authorized and covered, Dr. Worthing explained.
PBMs create formularies through this auction. The highest rebate to the PBM earns the top spot in the auction and becomes the preferred drug. “That highest bid gets paid for by passing the cost along to patients and insurance plans, and PBMs pocket the profits. This provides an incentive for pharmaceutical manufacturers to raise prices,” he said.
Dr. Worthing has seen these practices trickle down and affect his patients. “Frequently, the medication I prescribe based on what’s best for the patient based on their disease activity, values, and medical history is often not covered because a different drug or portfolio of drugs has earned the top spot in step therapy. This is an extremely frustrating and cumbersome process that not only delays access to treatments but also provides an incentive for higher drug prices,” he said.
There are other ways in which PBMs get in the way of care, said Ms. Hemmen, whose facility serves complex-care oncology patients.
“PBMs force scripts out of higher-quality pharmacies that preserve unfragmented care. They incentivize plan sponsors to put programs into place that take away patient choice, fragment care, and drive scripts to their own owned pharmacies,” she said.
Rutledge case sets precedent
In the Rutledge case, PCMA had challenged an Arkansas law that forbid PBMs from paying local pharmacies at a lower rate than what the pharmacies were reimbursed to fill prescriptions. Although the 8th Circuit Court of Appeals agreed with PCMA, the Supreme Court ruled in favor of Arkansas in late 2020.
The appeals court also backed PCMA in PCMA v. Wehbi. However, the Supreme Court vacated this decision and remanded it back to the appeals court, asking for a reconsideration in wake of the outcome in Rutledge v. PCMA.
PCMA has argued that Rutledge was a narrow decision, limited to state laws that regulate PBM reimbursements, and that Rutledge has no bearing on North Dakota law.
While it’s unfortunate that PCMA is trying to delay implementation of sensible regulations, “a lot of us are happy that this issue is coming to light,” Dr. Worthing said. “As a rheumatologist and health policy advocate, exposing drug middlemen is the most important bipartisan issue in the country today because it gets at the core of making sure that sick people get access to the medications they need and reducing the budget of insurance carriers, hospitals, and the federal budget.”
The ATAP brief noted that 28 state attorneys general have filed suit against PBMs, “securing settlements compelling PBMs to correct deceptive trade practices.”
Many people at the state and local level were waiting for the Supreme Court to decide on Rutledge before enacting legislation and sensible regulations, and now they can go ahead and do it, said Dr. Worthing. “I expect to see this across the country as states look at budgets, and as patients bring personal stories to light. We look forward to states passing these kinds of laws to regulate PBMs.”
The ACR doesn’t anticipate a ruling in the Wehbi case until the spring of 2022.
Recent laws passed around PBMs and the pharmacy benefit are a good first step in holding PBMs accountable for quality of care and honoring patient choice, Ms. Hemmen said. The laws also begin to address the fiscal manipulations PBMs use to gain advantage and direct scripts to their own coffers, she added. However, this may not have enough teeth. “These state laws are coming from a provider perspective, and they don’t anticipate what PBMs will do in response. The PBMs are going to work around it.”
Mark Nelson, PharmD, recalls the anguish when a major pharmacy benefit manager (PBM) moved all veteran patients with prostate cancer at his facility from an effective medication to a pricier alternative therapy. “All of these patients were stable on their therapy and were extremely distraught about their medications being changed,” said Dr. Nelson, CEO of Northwest Medical Specialties in Washington State. While there was no clinical reason to change the medication, “our oncologists had no choice other than to comply,” he said.
It’s unclear why a PBM would switch to a more expensive medication that has no additional clinical benefit, he continued. “Why upset so many veterans? For what reason? We were not given a reason despite our very vocal protest.”
Angus B. Worthing, MD, sees these scenarios unfold every day in his rheumatology practice in the Washington, D.C., area. “In my clinic with 25 doctors, we have three full-time people that only handle PBMs,” he said in an interview. He and others in the medical community, as well as many states, have been pushing back on what they see as efforts by PBMs to raise drug prices and collect the profits at the expense of patients.
PCMA’s challenges against PBM law
The Pharmaceutical Care Management Association (PCMA), a trade group that represents PBMs, has sued at least a half dozen states on their ability to regulate PBMs. However, a landmark case in late 2020 (Pharmaceutical Care Management Association v. Rutledge) set a new precedent. Reversing a lower appeals court decision, the Supreme Court unanimously ruled in favor of allowing states to put in place fair regulation of these entities.
Dr. Worthing and others hope that the medical community and states can leverage this ruling in another lawsuit PCMA brought against North Dakota (PCMA v. Wehbi). PCMA filed this lawsuit in 2017, which challenges two statutes on PBM regulation. The group has issued similar legal challenges in Maine, the District of Columbia, Iowa, Oklahoma, and Arkansas with the Rutledge case.
“PBMs have become massive profit centers while (ironically) increasing patients’ out-of-pocket costs, interfering with doctor-patient relationships, and impairing patient access to appropriate treatment,” according to an amicus brief filed by The Alliance for Transparent & Affordable Prescriptions (ATAP), the Community Oncology Alliance (COA), and American Pharmacies, supporting North Dakota in the Wehbi case.
This is to ensure the case represents the voices of physicians, patients, nurses, and other stakeholders, and underscores PBM abuses, said Dr. Worthing, vice president of ATAP. He also serves as the American College of Rheumatology’s representative on ATAP’s Executive Committee.
PCMA did not respond to requests for comment. Its CEO and president, J.C. Scott, emphasizes that PBMs have a long track record of reducing drug costs for patients and plan sponsors. In 2021, PCMA released 21 policy solutions, a set of industry principles and a three-part policy platform, all with an aim to bring down costs and increase access to pharmaceutical care, according to the organization.
PCMA estimates that the strategies in its platform (updating Medicare Part D, accelerating value-based care, and eliminating anticompetitive ‘pay for delay’ agreements) would save the federal government a maximum of $398.7 billion over 10 years.
According to Wendy Hemmen, senior director with Texas Oncology in Dallas, PBMs do their own unique calculations to arrive at their cost reductions. “Essentially in a PBM, they use things that make their story. Numbers reported to plan sponsors and to the public are not audited and are usually in terms of percentages or a per member per month. Data points are moved around, dropped, or reclassified to make the story that the PBM needs to tell,” Ms. Hemmen said.
Amicus briefs dispute ERISA connection
North Dakota legislation prohibits PBMs from charging copays to patients that exceed the cost of a drug. It also prohibits gag clause provisions that restrict what pharmacists may discuss with patients. PBMs may charge fees based on performance metrics, but they must use nationally recognized metrics. Fees must be disclosed at the point of sale.
In its legal challenges, PCMA has asserted that state laws violate the preemption clause in the Employee Retirement Income Security Act (ERISA). “Federal preemption allows employers flexibility to administer innovative benefit plans in an environment of increasing health care costs. The court’s decision in Rutledge v. PCMA will either uphold or threaten these federal protections,” PCMA asserted in a statement issued in March 2020.
ATAP’s amicus brief, and another one filed by 34 attorneys general that supports the North Dakota statute to regulate PBMs, counter that this isn’t the case.
“First, PBM regulation (in its common and standard form) does not reference ERISA itself. These laws leave all plans on equal footing; they do not single out ERISA plans for preferred or disfavored coverage, and they do not change the playing field for ERISA plans alone ... Second, PBM regulation does not have any prohibited connection with ERISA plans,” noted authors in the ATAP brief.
PCMA has also included Medicare preemption in its arguments against PBM regulation. This is meritless, wrote the state attorneys general. “Medicare preempts state laws only if a Medicare ‘standard’ particularly addresses the subject of state regulation. Because the challenged North Dakota laws do not dictate plan benefits or conflict with a Medicare standard, they are not preempted.”
The auctioning of medications
PBMs in theory could use their market power to drive down costs by extracting discounts from drug makers and pharmacies. In reality, they retain any price concessions and discounts for themselves, ATAP’s brief continued.
A system that PBMs have put into place, called step therapy, is essentially an auction for the preferred spot that will be authorized and covered, Dr. Worthing explained.
PBMs create formularies through this auction. The highest rebate to the PBM earns the top spot in the auction and becomes the preferred drug. “That highest bid gets paid for by passing the cost along to patients and insurance plans, and PBMs pocket the profits. This provides an incentive for pharmaceutical manufacturers to raise prices,” he said.
Dr. Worthing has seen these practices trickle down and affect his patients. “Frequently, the medication I prescribe based on what’s best for the patient based on their disease activity, values, and medical history is often not covered because a different drug or portfolio of drugs has earned the top spot in step therapy. This is an extremely frustrating and cumbersome process that not only delays access to treatments but also provides an incentive for higher drug prices,” he said.
There are other ways in which PBMs get in the way of care, said Ms. Hemmen, whose facility serves complex-care oncology patients.
“PBMs force scripts out of higher-quality pharmacies that preserve unfragmented care. They incentivize plan sponsors to put programs into place that take away patient choice, fragment care, and drive scripts to their own owned pharmacies,” she said.
Rutledge case sets precedent
In the Rutledge case, PCMA had challenged an Arkansas law that forbid PBMs from paying local pharmacies at a lower rate than what the pharmacies were reimbursed to fill prescriptions. Although the 8th Circuit Court of Appeals agreed with PCMA, the Supreme Court ruled in favor of Arkansas in late 2020.
The appeals court also backed PCMA in PCMA v. Wehbi. However, the Supreme Court vacated this decision and remanded it back to the appeals court, asking for a reconsideration in wake of the outcome in Rutledge v. PCMA.
PCMA has argued that Rutledge was a narrow decision, limited to state laws that regulate PBM reimbursements, and that Rutledge has no bearing on North Dakota law.
While it’s unfortunate that PCMA is trying to delay implementation of sensible regulations, “a lot of us are happy that this issue is coming to light,” Dr. Worthing said. “As a rheumatologist and health policy advocate, exposing drug middlemen is the most important bipartisan issue in the country today because it gets at the core of making sure that sick people get access to the medications they need and reducing the budget of insurance carriers, hospitals, and the federal budget.”
The ATAP brief noted that 28 state attorneys general have filed suit against PBMs, “securing settlements compelling PBMs to correct deceptive trade practices.”
Many people at the state and local level were waiting for the Supreme Court to decide on Rutledge before enacting legislation and sensible regulations, and now they can go ahead and do it, said Dr. Worthing. “I expect to see this across the country as states look at budgets, and as patients bring personal stories to light. We look forward to states passing these kinds of laws to regulate PBMs.”
The ACR doesn’t anticipate a ruling in the Wehbi case until the spring of 2022.
Recent laws passed around PBMs and the pharmacy benefit are a good first step in holding PBMs accountable for quality of care and honoring patient choice, Ms. Hemmen said. The laws also begin to address the fiscal manipulations PBMs use to gain advantage and direct scripts to their own coffers, she added. However, this may not have enough teeth. “These state laws are coming from a provider perspective, and they don’t anticipate what PBMs will do in response. The PBMs are going to work around it.”
No link between childhood vaccinations and allergies or asthma
A meta-analysis by Australian researchers found no link between childhood vaccinations and an increase in allergies and asthma. In fact, children who received the BCG vaccine actually had a lesser incidence of eczema than other children, but there was no difference shown in any of the allergies or asthma.
The researchers, in a report published in the journal Allergy, write, “We found no evidence that childhood vaccination with commonly administered vaccines was associated with increased risk of later allergic disease.”
“Allergies have increased worldwide in the last 50 years, and in developed countries, earlier,” said study author Caroline J. Lodge, PhD, principal research fellow at the University of Melbourne, in an interview. “In developing countries, it is still a crisis.” No one knows why, she said. That was the reason for the recent study.
Allergic diseases such as allergic rhinitis (hay fever) and food allergies have a serious influence on quality of life, and the incidence is growing. According to the Global Asthma Network, there are 334 million people living with asthma. Between 2%-10% of adults have atopic eczema, and more than a 250,000 people have food allergies. This coincides temporally with an increase in mass vaccination of children.
Unlike the controversy surrounding vaccinations and autism, which has long been debunked as baseless, a hygiene hypothesis postulates that when children acquire immunity from many diseases, they become vulnerable to allergic reactions. Thanks to vaccinations, children in the developed world now are routinely immune to dozens of diseases.
That immunity leads to suppression of a major antibody response, increasing sensitivity to allergens and allergic disease. Suspicion of a link with childhood vaccinations has been used by opponents of vaccines in lobbying campaigns jeopardizing the sustainability of vaccine programs. In recent days, for example, the state of Tennessee has halted a program to encourage vaccination for COVID-19 as well as all other vaccinations, the result of pressure on the state by anti-vaccination lobbying.
But the Melbourne researchers reported that the meta-analysis of 42 published research studies doesn’t support the vaccine–allergy hypothesis. Using PubMed and EMBASE records between January 1946 and January 2018, researchers selected studies to be included in the analysis, looking for allergic outcomes in children given BCG or vaccines for measles or pertussis. Thirty-five publications reported cohort studies, and seven were based on randomized controlled trials.
The Australian study is not the only one showing the same lack of linkage between vaccination and allergy. The International Study of Asthma and Allergies in Childhood (ISAAC) found no association between mass vaccination and atopic disease. A 1998 Swedish study of 669 children found no differences in the incidence of allergic diseases between those who received pertussis vaccine and those who did not.
“The bottom line is that vaccines prevent infectious diseases,” said Matthew B. Laurens, associate professor of pediatrics at the University of Maryland, Baltimore, in an interview. Dr. Laurens was not part of the Australian study.
“Large-scale epidemiological studies do not support the theory that vaccines are associated with an increased risk of allergy or asthma,” he stressed. “Parents should not be deterred from vaccinating their children because of fears that this would increase risks of allergy and/or asthma.”
Dr. Lodge and Dr. Laurens have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A meta-analysis by Australian researchers found no link between childhood vaccinations and an increase in allergies and asthma. In fact, children who received the BCG vaccine actually had a lesser incidence of eczema than other children, but there was no difference shown in any of the allergies or asthma.
The researchers, in a report published in the journal Allergy, write, “We found no evidence that childhood vaccination with commonly administered vaccines was associated with increased risk of later allergic disease.”
“Allergies have increased worldwide in the last 50 years, and in developed countries, earlier,” said study author Caroline J. Lodge, PhD, principal research fellow at the University of Melbourne, in an interview. “In developing countries, it is still a crisis.” No one knows why, she said. That was the reason for the recent study.
Allergic diseases such as allergic rhinitis (hay fever) and food allergies have a serious influence on quality of life, and the incidence is growing. According to the Global Asthma Network, there are 334 million people living with asthma. Between 2%-10% of adults have atopic eczema, and more than a 250,000 people have food allergies. This coincides temporally with an increase in mass vaccination of children.
Unlike the controversy surrounding vaccinations and autism, which has long been debunked as baseless, a hygiene hypothesis postulates that when children acquire immunity from many diseases, they become vulnerable to allergic reactions. Thanks to vaccinations, children in the developed world now are routinely immune to dozens of diseases.
That immunity leads to suppression of a major antibody response, increasing sensitivity to allergens and allergic disease. Suspicion of a link with childhood vaccinations has been used by opponents of vaccines in lobbying campaigns jeopardizing the sustainability of vaccine programs. In recent days, for example, the state of Tennessee has halted a program to encourage vaccination for COVID-19 as well as all other vaccinations, the result of pressure on the state by anti-vaccination lobbying.
But the Melbourne researchers reported that the meta-analysis of 42 published research studies doesn’t support the vaccine–allergy hypothesis. Using PubMed and EMBASE records between January 1946 and January 2018, researchers selected studies to be included in the analysis, looking for allergic outcomes in children given BCG or vaccines for measles or pertussis. Thirty-five publications reported cohort studies, and seven were based on randomized controlled trials.
The Australian study is not the only one showing the same lack of linkage between vaccination and allergy. The International Study of Asthma and Allergies in Childhood (ISAAC) found no association between mass vaccination and atopic disease. A 1998 Swedish study of 669 children found no differences in the incidence of allergic diseases between those who received pertussis vaccine and those who did not.
“The bottom line is that vaccines prevent infectious diseases,” said Matthew B. Laurens, associate professor of pediatrics at the University of Maryland, Baltimore, in an interview. Dr. Laurens was not part of the Australian study.
“Large-scale epidemiological studies do not support the theory that vaccines are associated with an increased risk of allergy or asthma,” he stressed. “Parents should not be deterred from vaccinating their children because of fears that this would increase risks of allergy and/or asthma.”
Dr. Lodge and Dr. Laurens have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A meta-analysis by Australian researchers found no link between childhood vaccinations and an increase in allergies and asthma. In fact, children who received the BCG vaccine actually had a lesser incidence of eczema than other children, but there was no difference shown in any of the allergies or asthma.
The researchers, in a report published in the journal Allergy, write, “We found no evidence that childhood vaccination with commonly administered vaccines was associated with increased risk of later allergic disease.”
“Allergies have increased worldwide in the last 50 years, and in developed countries, earlier,” said study author Caroline J. Lodge, PhD, principal research fellow at the University of Melbourne, in an interview. “In developing countries, it is still a crisis.” No one knows why, she said. That was the reason for the recent study.
Allergic diseases such as allergic rhinitis (hay fever) and food allergies have a serious influence on quality of life, and the incidence is growing. According to the Global Asthma Network, there are 334 million people living with asthma. Between 2%-10% of adults have atopic eczema, and more than a 250,000 people have food allergies. This coincides temporally with an increase in mass vaccination of children.
Unlike the controversy surrounding vaccinations and autism, which has long been debunked as baseless, a hygiene hypothesis postulates that when children acquire immunity from many diseases, they become vulnerable to allergic reactions. Thanks to vaccinations, children in the developed world now are routinely immune to dozens of diseases.
That immunity leads to suppression of a major antibody response, increasing sensitivity to allergens and allergic disease. Suspicion of a link with childhood vaccinations has been used by opponents of vaccines in lobbying campaigns jeopardizing the sustainability of vaccine programs. In recent days, for example, the state of Tennessee has halted a program to encourage vaccination for COVID-19 as well as all other vaccinations, the result of pressure on the state by anti-vaccination lobbying.
But the Melbourne researchers reported that the meta-analysis of 42 published research studies doesn’t support the vaccine–allergy hypothesis. Using PubMed and EMBASE records between January 1946 and January 2018, researchers selected studies to be included in the analysis, looking for allergic outcomes in children given BCG or vaccines for measles or pertussis. Thirty-five publications reported cohort studies, and seven were based on randomized controlled trials.
The Australian study is not the only one showing the same lack of linkage between vaccination and allergy. The International Study of Asthma and Allergies in Childhood (ISAAC) found no association between mass vaccination and atopic disease. A 1998 Swedish study of 669 children found no differences in the incidence of allergic diseases between those who received pertussis vaccine and those who did not.
“The bottom line is that vaccines prevent infectious diseases,” said Matthew B. Laurens, associate professor of pediatrics at the University of Maryland, Baltimore, in an interview. Dr. Laurens was not part of the Australian study.
“Large-scale epidemiological studies do not support the theory that vaccines are associated with an increased risk of allergy or asthma,” he stressed. “Parents should not be deterred from vaccinating their children because of fears that this would increase risks of allergy and/or asthma.”
Dr. Lodge and Dr. Laurens have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The first signs of elusive dysautonomia may appear on the skin
During the annual meeting of the Society for Pediatric Dermatology, Adelaide A. Hebert, MD, defined dysautonomia as an umbrella term describing conditions that result in a malfunction of the autonomic nervous system. “This encompasses both the sympathetic and the parasympathetic components of the nervous system,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “Clinical findings may be neurometabolic, developmental, and/or degenerative,” representing a “whole constellation of issues” that physicians may encounter in practice, she noted. Of particular interest is postural orthostatic tachycardia syndrome (POTS), which affects between 1 million and 3 million people in the United States. Typical symptoms include lightheadedness, fainting, and a rapid increase in heartbeat after standing up from a seated position. Other conditions associated with dysautonomia include neurocardiogenic syncope and multiple system atrophy.
Dysautonomia can impact the brain, heart, mouth, blood vessels, eyes, immune cells, and bladder, as well as the skin. Patient presentations vary with symptoms that can range from mild to debilitating. The average time from symptom onset to diagnosis of dysautonomia is 7 years. “It is very difficult to put together these mysterious symptoms that patients have unless one really thinks about dysautonomia as a possible diagnosis,” Dr. Hebert said.
One of the common symptoms that she has seen in her clinical practice is joint hypermobility. “There is a known association between dysautonomia and hypermobile-type Ehlers-Danlos syndrome (EDS), and these patients often have hyperhidrosis,” she said. “So, keep in mind that you could see hypermobility, especially in those with EDS, with associated hyperhidrosis and dysautonomia.” Two key references that she recommends to clinicians when evaluating patients with possible dysautonomia are a study on postural tachycardia in hypermobile EDS, and an article on cardiovascular autonomic dysfunction in hypermobile EDS.
The Beighton Scoring System, which measures joint mobility on a 9-point scale, involves assessment of the joint mobility of the knuckle of both pinky fingers, the base of both thumbs, the elbows, knees, and spine. An instructional video on how to perform a joint hypermobility assessment is available on the Ehler-Danlos Society website.
Literature review
In March 2021, Dr. Hebert and colleagues from other medical specialties published a summary of the literature on cutaneous manifestations in dysautonomia, with an emphasis on syndromes of orthostatic intolerance. “We had neurology, cardiology, along with dermatology involved in contributing the findings they had seen in the UTHealth McGovern Dysautonomia Center of Excellence as there was a dearth of literature that taught us about the cutaneous manifestations of orthostatic intolerance syndromes,” Dr. Hebert said.
One study included in the review showed that 23 out of 26 patients with POTS had at least one of the following cutaneous manifestations: flushing, Raynaud’s phenomenon, evanescent hyperemia, livedo reticularis, erythromelalgia, and hypo- or hyperhidrosis. “If you see a patient with any of these findings, you want to think about the possibility of dysautonomia,” she said, adding that urticaria can also be a finding.
To screen for dysautonomia, she advised, “ask patients if they have difficulty sitting or standing upright, if they have indigestion or other gastric symptoms, abnormal blood vessel functioning such as low or high blood pressure, increased or decreased sweating, changes in urinary frequency or urinary incontinence, or challenges with vision.”
If the patient answers yes to two or more of these questions, she said, consider a referral to neurology and/or cardiology or a center of excellence for further evaluation with tilt-table testing and other screening tools. She also recommended a review published in 2015 that describes the dermatological manifestations of postural tachycardia syndrome and includes illustrated cases.
One of Dr. Hebert’s future dermatology residents assembled a composite of data from the Dysautonomia Center of Excellence, and in the study, found that, compared with males, females with dysautonomia suffer more from excessive sweating, paleness of the face, pale extremities, swelling, cyanosis, cold intolerance, flushing, and hot flashes.
Dr. Hebert disclosed that she has been a consultant to and an adviser for several pharmaceutical companies.
During the annual meeting of the Society for Pediatric Dermatology, Adelaide A. Hebert, MD, defined dysautonomia as an umbrella term describing conditions that result in a malfunction of the autonomic nervous system. “This encompasses both the sympathetic and the parasympathetic components of the nervous system,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “Clinical findings may be neurometabolic, developmental, and/or degenerative,” representing a “whole constellation of issues” that physicians may encounter in practice, she noted. Of particular interest is postural orthostatic tachycardia syndrome (POTS), which affects between 1 million and 3 million people in the United States. Typical symptoms include lightheadedness, fainting, and a rapid increase in heartbeat after standing up from a seated position. Other conditions associated with dysautonomia include neurocardiogenic syncope and multiple system atrophy.
Dysautonomia can impact the brain, heart, mouth, blood vessels, eyes, immune cells, and bladder, as well as the skin. Patient presentations vary with symptoms that can range from mild to debilitating. The average time from symptom onset to diagnosis of dysautonomia is 7 years. “It is very difficult to put together these mysterious symptoms that patients have unless one really thinks about dysautonomia as a possible diagnosis,” Dr. Hebert said.
One of the common symptoms that she has seen in her clinical practice is joint hypermobility. “There is a known association between dysautonomia and hypermobile-type Ehlers-Danlos syndrome (EDS), and these patients often have hyperhidrosis,” she said. “So, keep in mind that you could see hypermobility, especially in those with EDS, with associated hyperhidrosis and dysautonomia.” Two key references that she recommends to clinicians when evaluating patients with possible dysautonomia are a study on postural tachycardia in hypermobile EDS, and an article on cardiovascular autonomic dysfunction in hypermobile EDS.
The Beighton Scoring System, which measures joint mobility on a 9-point scale, involves assessment of the joint mobility of the knuckle of both pinky fingers, the base of both thumbs, the elbows, knees, and spine. An instructional video on how to perform a joint hypermobility assessment is available on the Ehler-Danlos Society website.
Literature review
In March 2021, Dr. Hebert and colleagues from other medical specialties published a summary of the literature on cutaneous manifestations in dysautonomia, with an emphasis on syndromes of orthostatic intolerance. “We had neurology, cardiology, along with dermatology involved in contributing the findings they had seen in the UTHealth McGovern Dysautonomia Center of Excellence as there was a dearth of literature that taught us about the cutaneous manifestations of orthostatic intolerance syndromes,” Dr. Hebert said.
One study included in the review showed that 23 out of 26 patients with POTS had at least one of the following cutaneous manifestations: flushing, Raynaud’s phenomenon, evanescent hyperemia, livedo reticularis, erythromelalgia, and hypo- or hyperhidrosis. “If you see a patient with any of these findings, you want to think about the possibility of dysautonomia,” she said, adding that urticaria can also be a finding.
To screen for dysautonomia, she advised, “ask patients if they have difficulty sitting or standing upright, if they have indigestion or other gastric symptoms, abnormal blood vessel functioning such as low or high blood pressure, increased or decreased sweating, changes in urinary frequency or urinary incontinence, or challenges with vision.”
If the patient answers yes to two or more of these questions, she said, consider a referral to neurology and/or cardiology or a center of excellence for further evaluation with tilt-table testing and other screening tools. She also recommended a review published in 2015 that describes the dermatological manifestations of postural tachycardia syndrome and includes illustrated cases.
One of Dr. Hebert’s future dermatology residents assembled a composite of data from the Dysautonomia Center of Excellence, and in the study, found that, compared with males, females with dysautonomia suffer more from excessive sweating, paleness of the face, pale extremities, swelling, cyanosis, cold intolerance, flushing, and hot flashes.
Dr. Hebert disclosed that she has been a consultant to and an adviser for several pharmaceutical companies.
During the annual meeting of the Society for Pediatric Dermatology, Adelaide A. Hebert, MD, defined dysautonomia as an umbrella term describing conditions that result in a malfunction of the autonomic nervous system. “This encompasses both the sympathetic and the parasympathetic components of the nervous system,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “Clinical findings may be neurometabolic, developmental, and/or degenerative,” representing a “whole constellation of issues” that physicians may encounter in practice, she noted. Of particular interest is postural orthostatic tachycardia syndrome (POTS), which affects between 1 million and 3 million people in the United States. Typical symptoms include lightheadedness, fainting, and a rapid increase in heartbeat after standing up from a seated position. Other conditions associated with dysautonomia include neurocardiogenic syncope and multiple system atrophy.
Dysautonomia can impact the brain, heart, mouth, blood vessels, eyes, immune cells, and bladder, as well as the skin. Patient presentations vary with symptoms that can range from mild to debilitating. The average time from symptom onset to diagnosis of dysautonomia is 7 years. “It is very difficult to put together these mysterious symptoms that patients have unless one really thinks about dysautonomia as a possible diagnosis,” Dr. Hebert said.
One of the common symptoms that she has seen in her clinical practice is joint hypermobility. “There is a known association between dysautonomia and hypermobile-type Ehlers-Danlos syndrome (EDS), and these patients often have hyperhidrosis,” she said. “So, keep in mind that you could see hypermobility, especially in those with EDS, with associated hyperhidrosis and dysautonomia.” Two key references that she recommends to clinicians when evaluating patients with possible dysautonomia are a study on postural tachycardia in hypermobile EDS, and an article on cardiovascular autonomic dysfunction in hypermobile EDS.
The Beighton Scoring System, which measures joint mobility on a 9-point scale, involves assessment of the joint mobility of the knuckle of both pinky fingers, the base of both thumbs, the elbows, knees, and spine. An instructional video on how to perform a joint hypermobility assessment is available on the Ehler-Danlos Society website.
Literature review
In March 2021, Dr. Hebert and colleagues from other medical specialties published a summary of the literature on cutaneous manifestations in dysautonomia, with an emphasis on syndromes of orthostatic intolerance. “We had neurology, cardiology, along with dermatology involved in contributing the findings they had seen in the UTHealth McGovern Dysautonomia Center of Excellence as there was a dearth of literature that taught us about the cutaneous manifestations of orthostatic intolerance syndromes,” Dr. Hebert said.
One study included in the review showed that 23 out of 26 patients with POTS had at least one of the following cutaneous manifestations: flushing, Raynaud’s phenomenon, evanescent hyperemia, livedo reticularis, erythromelalgia, and hypo- or hyperhidrosis. “If you see a patient with any of these findings, you want to think about the possibility of dysautonomia,” she said, adding that urticaria can also be a finding.
To screen for dysautonomia, she advised, “ask patients if they have difficulty sitting or standing upright, if they have indigestion or other gastric symptoms, abnormal blood vessel functioning such as low or high blood pressure, increased or decreased sweating, changes in urinary frequency or urinary incontinence, or challenges with vision.”
If the patient answers yes to two or more of these questions, she said, consider a referral to neurology and/or cardiology or a center of excellence for further evaluation with tilt-table testing and other screening tools. She also recommended a review published in 2015 that describes the dermatological manifestations of postural tachycardia syndrome and includes illustrated cases.
One of Dr. Hebert’s future dermatology residents assembled a composite of data from the Dysautonomia Center of Excellence, and in the study, found that, compared with males, females with dysautonomia suffer more from excessive sweating, paleness of the face, pale extremities, swelling, cyanosis, cold intolerance, flushing, and hot flashes.
Dr. Hebert disclosed that she has been a consultant to and an adviser for several pharmaceutical companies.
FROM SPD 2021
Face mask–related injuries rose dramatically in 2020

How dramatic? The number of mask-related injuries treated in U.S. emergency departments averaged about 200 per year from 2016 to 2019. In 2020, that figure soared to 4,976 – an increase of almost 2,400%, Gerald McGwin Jr., PhD, and associates said in a research letter published in the Journal to the American Academy of Dermatology.
“Prior to the COVID-19 pandemic the use of respiratory protection equipment was largely limited to healthcare and industrial settings. As [face mask] use by the general population increased, so too have reports of dermatologic reactions,” said Dr. McGwin and associates of the department of epidemiology at the University of Alabama at Birmingham.
Dermatitis was the most common mask-related injury treated last year, affecting 28.3% of those presenting to EDs, followed by lacerations at 10.1%. Injuries were more common in women than men, but while and black patients “were equally represented,” they noted, based on data from the National Electronic Injury Surveillance System, which includes about 100 hospitals and EDs.
Most injuries were caused by rashes/allergic reactions (38%) from prolonged use, poorly fitting masks (19%), and obscured vision (14%). “There was a small (5%) but meaningful number of injuries, all among children, attributable to consuming pieces of a mask or inserting dismantled pieces of a mask into body orifices,” the investigators said.
Guidance from the Centers for Disease Control and Prevention is available “to aid in the choice and proper fit of face masks,” they wrote, and “increased awareness of these resources [could] minimize the future occurrence of mask-related injuries.”
There was no funding source for the study, and the investigators did not declare any conflicts of interest.

How dramatic? The number of mask-related injuries treated in U.S. emergency departments averaged about 200 per year from 2016 to 2019. In 2020, that figure soared to 4,976 – an increase of almost 2,400%, Gerald McGwin Jr., PhD, and associates said in a research letter published in the Journal to the American Academy of Dermatology.
“Prior to the COVID-19 pandemic the use of respiratory protection equipment was largely limited to healthcare and industrial settings. As [face mask] use by the general population increased, so too have reports of dermatologic reactions,” said Dr. McGwin and associates of the department of epidemiology at the University of Alabama at Birmingham.
Dermatitis was the most common mask-related injury treated last year, affecting 28.3% of those presenting to EDs, followed by lacerations at 10.1%. Injuries were more common in women than men, but while and black patients “were equally represented,” they noted, based on data from the National Electronic Injury Surveillance System, which includes about 100 hospitals and EDs.
Most injuries were caused by rashes/allergic reactions (38%) from prolonged use, poorly fitting masks (19%), and obscured vision (14%). “There was a small (5%) but meaningful number of injuries, all among children, attributable to consuming pieces of a mask or inserting dismantled pieces of a mask into body orifices,” the investigators said.
Guidance from the Centers for Disease Control and Prevention is available “to aid in the choice and proper fit of face masks,” they wrote, and “increased awareness of these resources [could] minimize the future occurrence of mask-related injuries.”
There was no funding source for the study, and the investigators did not declare any conflicts of interest.

How dramatic? The number of mask-related injuries treated in U.S. emergency departments averaged about 200 per year from 2016 to 2019. In 2020, that figure soared to 4,976 – an increase of almost 2,400%, Gerald McGwin Jr., PhD, and associates said in a research letter published in the Journal to the American Academy of Dermatology.
“Prior to the COVID-19 pandemic the use of respiratory protection equipment was largely limited to healthcare and industrial settings. As [face mask] use by the general population increased, so too have reports of dermatologic reactions,” said Dr. McGwin and associates of the department of epidemiology at the University of Alabama at Birmingham.
Dermatitis was the most common mask-related injury treated last year, affecting 28.3% of those presenting to EDs, followed by lacerations at 10.1%. Injuries were more common in women than men, but while and black patients “were equally represented,” they noted, based on data from the National Electronic Injury Surveillance System, which includes about 100 hospitals and EDs.
Most injuries were caused by rashes/allergic reactions (38%) from prolonged use, poorly fitting masks (19%), and obscured vision (14%). “There was a small (5%) but meaningful number of injuries, all among children, attributable to consuming pieces of a mask or inserting dismantled pieces of a mask into body orifices,” the investigators said.
Guidance from the Centers for Disease Control and Prevention is available “to aid in the choice and proper fit of face masks,” they wrote, and “increased awareness of these resources [could] minimize the future occurrence of mask-related injuries.”
There was no funding source for the study, and the investigators did not declare any conflicts of interest.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Dyspigmentation common in SOC patients with bullous pemphigoid
Patients of skin of color (SOC) with bullous pemphigoid presented significantly more often with dyspigmentation than did White patients in a retrospective observational study of patients diagnosed with BP at New York University Langone Health and Bellevue Hospital, also in New York.
“Dyspigmentation in the skin-of-color patient population is important to recognize not only for an objective evaluation of the disease process, but also from a quality of life perspective ... to ensure there is timely diagnosis and initiation of treatment in the skin-of-color population,” said medical student Payal Shah, BS, of New York University, in presenting the findings at the annual Skin of Color Society symposium.
Ms. Shah and coresearchers identified 94 cases of BP through retrospective view of electronic health records – 59 in White patients and 35 in SOC patients. The physical examination features most commonly found at initial presentation were bullae or vesicles in both White patients (64.4% ) and SOC patients (80%). Erosions or ulcers were also commonly found in both groups (42.4% of White patients and 60% of SOC patients).
Erythema was more commonly found in White patients at initial presentation: 35.6% vs. 14.3% of SOC patients (P = .032). Dyspigmentation, defined as areas of hyper- or hypopigmentation, was more commonly found in SOC patients: 54.3% versus 10.2% in White patients (P < .001). The difference in erythema of inflammatory bullae in BP may stem from the fact that erythema is more difficult to discern in patients with darker skin types, Ms. Shah said.
SOC patients also were significantly younger at the time of initial presentation; their mean age was 63 years, compared with 77 years in the White population (P < .001).
The time to diagnosis, defined as the time from initial symptoms to dermatologic diagnosis, was greater for the SOC population –7.6 months vs. 6.2 months for white patients –though the difference was not statistically significant, they said in the abstract .
Dyspigmentation has been shown to be among the top dermatologic concerns of Black patients and has important quality of life implications. “Early diagnosis to prevent difficult-to-treat dyspigmentation is therefore of utmost importance,” they said in the abstract.
Prior research has demonstrated that non-White populations are at greater risk for hospitalization secondary to BP and have a greater risk of disease mortality, Ms. Shah noted in her presentation.
Patients of skin of color (SOC) with bullous pemphigoid presented significantly more often with dyspigmentation than did White patients in a retrospective observational study of patients diagnosed with BP at New York University Langone Health and Bellevue Hospital, also in New York.
“Dyspigmentation in the skin-of-color patient population is important to recognize not only for an objective evaluation of the disease process, but also from a quality of life perspective ... to ensure there is timely diagnosis and initiation of treatment in the skin-of-color population,” said medical student Payal Shah, BS, of New York University, in presenting the findings at the annual Skin of Color Society symposium.
Ms. Shah and coresearchers identified 94 cases of BP through retrospective view of electronic health records – 59 in White patients and 35 in SOC patients. The physical examination features most commonly found at initial presentation were bullae or vesicles in both White patients (64.4% ) and SOC patients (80%). Erosions or ulcers were also commonly found in both groups (42.4% of White patients and 60% of SOC patients).
Erythema was more commonly found in White patients at initial presentation: 35.6% vs. 14.3% of SOC patients (P = .032). Dyspigmentation, defined as areas of hyper- or hypopigmentation, was more commonly found in SOC patients: 54.3% versus 10.2% in White patients (P < .001). The difference in erythema of inflammatory bullae in BP may stem from the fact that erythema is more difficult to discern in patients with darker skin types, Ms. Shah said.
SOC patients also were significantly younger at the time of initial presentation; their mean age was 63 years, compared with 77 years in the White population (P < .001).
The time to diagnosis, defined as the time from initial symptoms to dermatologic diagnosis, was greater for the SOC population –7.6 months vs. 6.2 months for white patients –though the difference was not statistically significant, they said in the abstract .
Dyspigmentation has been shown to be among the top dermatologic concerns of Black patients and has important quality of life implications. “Early diagnosis to prevent difficult-to-treat dyspigmentation is therefore of utmost importance,” they said in the abstract.
Prior research has demonstrated that non-White populations are at greater risk for hospitalization secondary to BP and have a greater risk of disease mortality, Ms. Shah noted in her presentation.
Patients of skin of color (SOC) with bullous pemphigoid presented significantly more often with dyspigmentation than did White patients in a retrospective observational study of patients diagnosed with BP at New York University Langone Health and Bellevue Hospital, also in New York.
“Dyspigmentation in the skin-of-color patient population is important to recognize not only for an objective evaluation of the disease process, but also from a quality of life perspective ... to ensure there is timely diagnosis and initiation of treatment in the skin-of-color population,” said medical student Payal Shah, BS, of New York University, in presenting the findings at the annual Skin of Color Society symposium.
Ms. Shah and coresearchers identified 94 cases of BP through retrospective view of electronic health records – 59 in White patients and 35 in SOC patients. The physical examination features most commonly found at initial presentation were bullae or vesicles in both White patients (64.4% ) and SOC patients (80%). Erosions or ulcers were also commonly found in both groups (42.4% of White patients and 60% of SOC patients).
Erythema was more commonly found in White patients at initial presentation: 35.6% vs. 14.3% of SOC patients (P = .032). Dyspigmentation, defined as areas of hyper- or hypopigmentation, was more commonly found in SOC patients: 54.3% versus 10.2% in White patients (P < .001). The difference in erythema of inflammatory bullae in BP may stem from the fact that erythema is more difficult to discern in patients with darker skin types, Ms. Shah said.
SOC patients also were significantly younger at the time of initial presentation; their mean age was 63 years, compared with 77 years in the White population (P < .001).
The time to diagnosis, defined as the time from initial symptoms to dermatologic diagnosis, was greater for the SOC population –7.6 months vs. 6.2 months for white patients –though the difference was not statistically significant, they said in the abstract .
Dyspigmentation has been shown to be among the top dermatologic concerns of Black patients and has important quality of life implications. “Early diagnosis to prevent difficult-to-treat dyspigmentation is therefore of utmost importance,” they said in the abstract.
Prior research has demonstrated that non-White populations are at greater risk for hospitalization secondary to BP and have a greater risk of disease mortality, Ms. Shah noted in her presentation.
FROM SOC 2021