User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
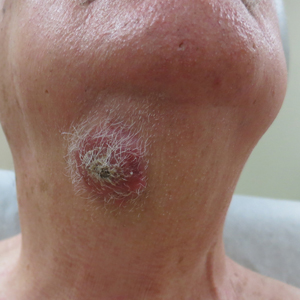
Nodule on the Neck
The Diagnosis: Primary Cutaneous Anaplastic Large Cell Lymphoma
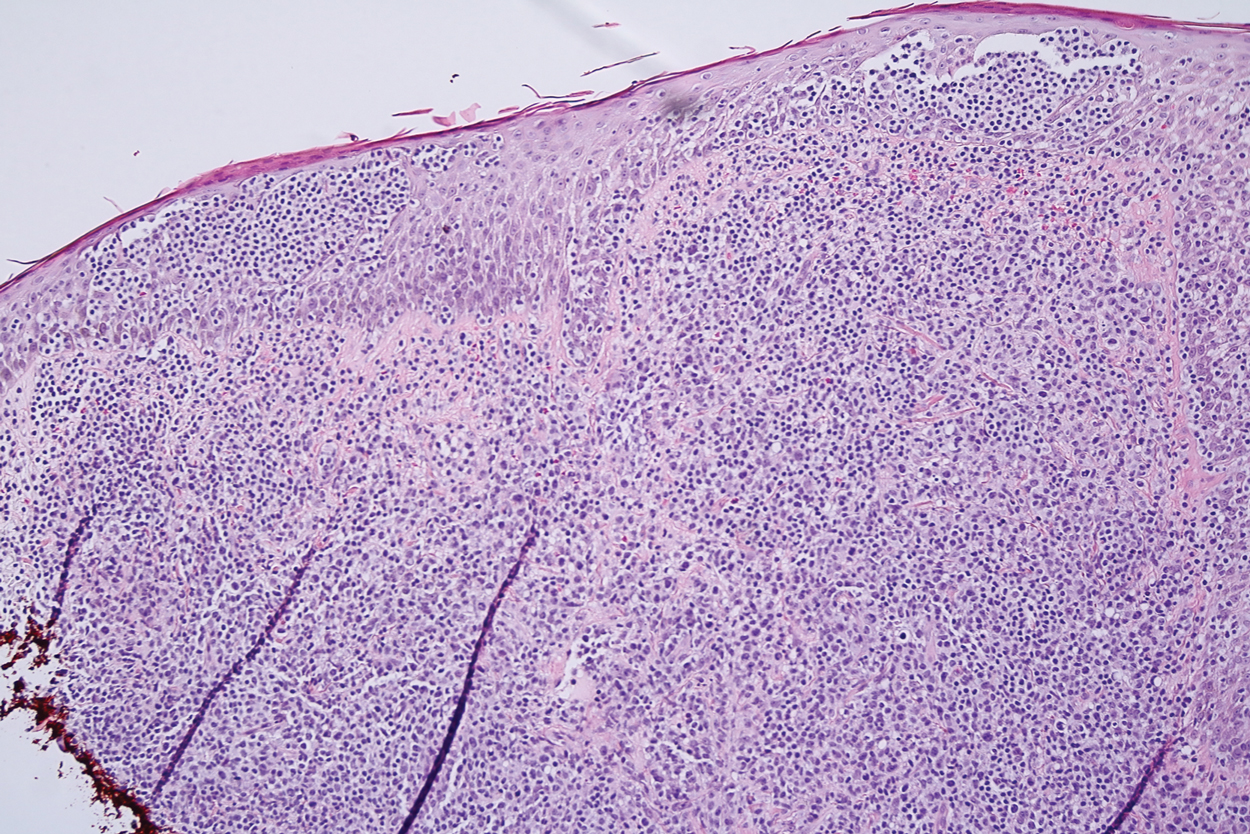
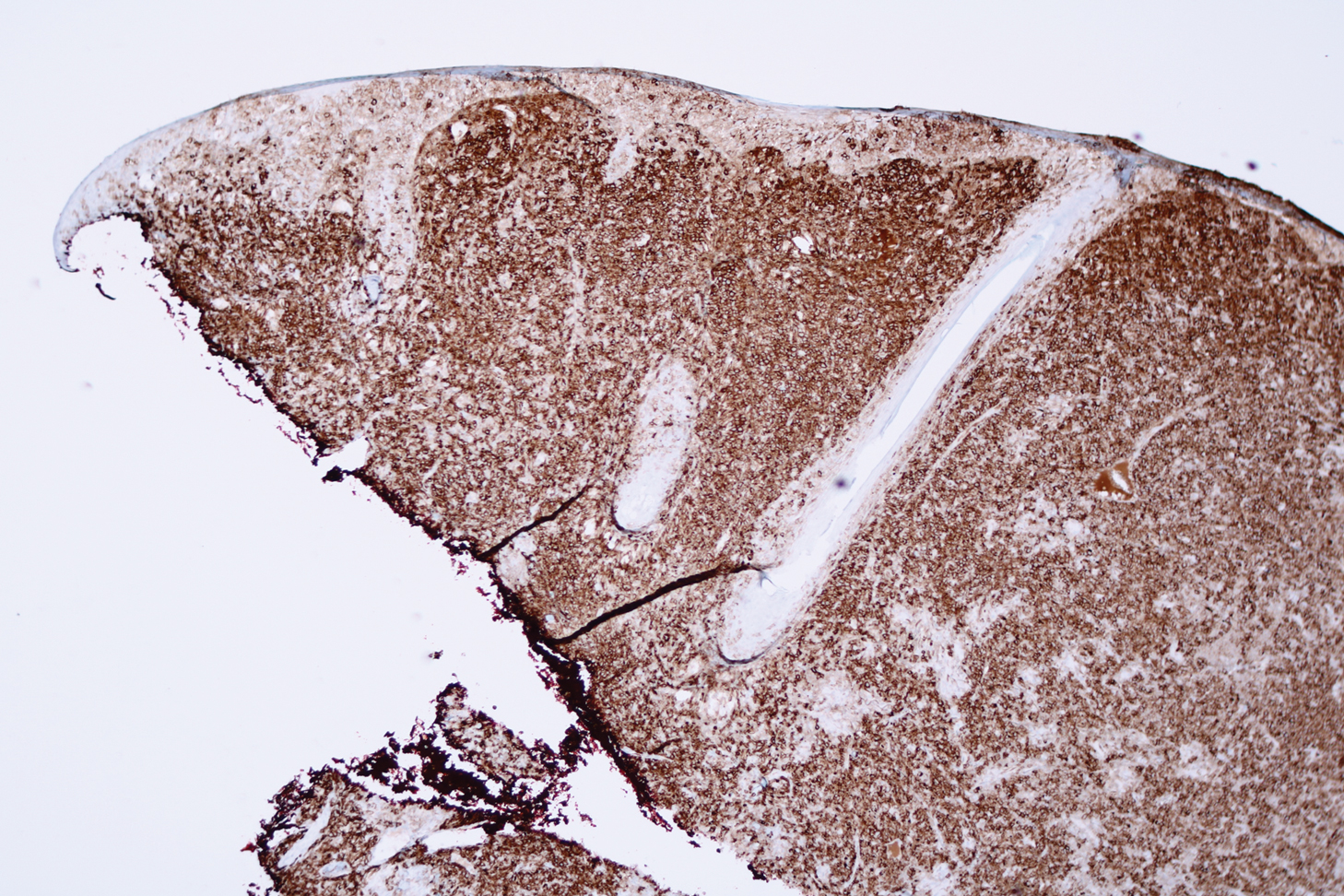
Microscopic analysis showed a dense proliferation of mononuclear cells filling and expanding the dermis with focal epidermotropism (Figure 1). Immunohistochemistry demonstrated strong and diffuse staining for CD3, CD4, and CD30 (Figure 2) and lack of staining for anaplastic lymphoma kinase (ALK). Workup to exclude systemic disease was initiated and included unremarkable computed tomography (CT) of the neck, chest, abdomen, and pelvis along with no abnormal cells on bone marrow biopsy. Complete blood cell count, basic metabolic panel, and lactate dehydrogenase were within reference range. Given the lack of evidence for systemic involvement, a diagnosis of primary cutaneous anaplastic large cell lymphoma (PC-ALCL) was made. The treatment plan for our patient with a solitary lesion was localized radiation therapy.
Primary cutaneous CD30+ lymphoproliferative disorders encompass a spectrum of conditions, with premalignant lymphomatoid papulosis (LyP) at one extreme and the malignant PC-ALCL on the other.1 The diagnosis of PC-ALCL is made by clinicopathologic correlation, and lesions typically present abruptly as solitary or grouped nodules with a tendency to ulcerate over time. Spontaneous regression has been reported, but relapse in the skin is frequent.2
A representative, typically excisional, biopsy should be performed if the clinician suspects PC-ALCL. Histologic criteria include a dense dermal infiltrate of large pleomorphic cells and the expression of CD30 in at least 75% of tumor cells.3 Primary cutaneous anaplastic large cell lymphoma typically lacks the ALK gene translocation with the nucleophosmin gene, NPM, that is common in systemic disease; however, a small subset of PC-ALCL may be ALK positive and indicate a higher chance of transformation into systemic disease.2
The extent of the lymphoma should be staged to exclude the possibility of systemic disease. This assessment includes a complete physical examination; laboratory investigation, including complete blood cell count with differential and blood chemistries; and radiography. A positron emission tomography-CT scan of the neck, chest, abdomen, and pelvis, or a whole-body integrated positron emission tomography-CT are sufficient for the radiographic examination.3
The initial choice of treatment for solitary or localized PC-ALCL is localized radiation therapy or low-dose methotrexate. Targeted therapy such as brentuximab has been shown to be effective for those with multifocal systemic involvement or refractory disease.2 Cure rates from radiation therapy alone approach 95%.3 It is important to highlight radiation therapy as the initial management plan to increase awareness and to avoid inappropriate treatment of PC-ALCL with traditional chemotherapy.
Large lesions of LyP may appear similar to PC-ALCL on histopathology, making the two entities difficult to distinguish. However, in contrast to PC-ALCL, LyP classically has a different clinical course characterized by waxing and waning crops of lesions that typically are smaller (<1 cm) than those of PC-ALCL.2 Large cell transformation of mycosis fungoides is another entity to consider, but these patients usually have a known history of mycosis fungoides.4
Keratoacanthomas, considered to be a variant of a well-differentiated squamous cell carcinoma, present as rapidly enlarging crateriform nodules with a keratotic core. They usually are found on the head and neck or sun-exposed areas of the extremities and may regress spontaneously.5 Histology will show atypical, highly differentiated squamous epithelia. Merkel cell carcinoma also has a predilection for the head and neck in older patients and may present as a rapidly growing nodule. However, histology will show an aggressive tumor with small round blue cells, and immunohistochemistry will show the characteristic paranuclear dot staining for CK20 along with staining for various neuroendocrine markers. Similarly, atypical fibroxanthoma is a low-grade sarcoma that also presents on the head and neck of elderly sun-damaged patients.5 Histology will show dermal proliferation of spindle cells that often extend up against the epidermis along with pleomorphism and atypical mitoses. Basal cell carcinoma is a common tumor that can present on the head and neck in sun-damaged patients. Nodular basal cell carcinomas can enlarge and ulcerate, but growth is seen over years rather than weeks.5 Histology characteristically will show tumor islands composed of basaloid cells with peripheral palisading and clefting between the tumor islands and the stroma.
- Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390.
- Brown RA, Fernandez-Pol S, Kim J. Primary cutaneous anaplastic large cell lymphoma. J Cutan Pathol. 2017;44:570-577.
- Kempf W, Pfaltz K, Vermeer MH, et al. EORTC, ISCL, and USCLC consensus recommendations for the treatment of primary cutaneous CD30-positive lymphoproliferative disorders: lymphomatoid papulosis and primary cutaneous anaplastic large-cell lymphoma. Blood. 2011;118:4024-4035.
- Jawed SI, Myskowski PL, Horwitz S, et al. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part II. prognosis, management, and future directions. J Am Acad Dermatol. 2014;70:223.e1-17.
- Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. Saunders Elsevier; 2015:475-489.
The Diagnosis: Primary Cutaneous Anaplastic Large Cell Lymphoma
Microscopic analysis showed a dense proliferation of mononuclear cells filling and expanding the dermis with focal epidermotropism (Figure 1). Immunohistochemistry demonstrated strong and diffuse staining for CD3, CD4, and CD30 (Figure 2) and lack of staining for anaplastic lymphoma kinase (ALK). Workup to exclude systemic disease was initiated and included unremarkable computed tomography (CT) of the neck, chest, abdomen, and pelvis along with no abnormal cells on bone marrow biopsy. Complete blood cell count, basic metabolic panel, and lactate dehydrogenase were within reference range. Given the lack of evidence for systemic involvement, a diagnosis of primary cutaneous anaplastic large cell lymphoma (PC-ALCL) was made. The treatment plan for our patient with a solitary lesion was localized radiation therapy.


Primary cutaneous CD30+ lymphoproliferative disorders encompass a spectrum of conditions, with premalignant lymphomatoid papulosis (LyP) at one extreme and the malignant PC-ALCL on the other.1 The diagnosis of PC-ALCL is made by clinicopathologic correlation, and lesions typically present abruptly as solitary or grouped nodules with a tendency to ulcerate over time. Spontaneous regression has been reported, but relapse in the skin is frequent.2
A representative, typically excisional, biopsy should be performed if the clinician suspects PC-ALCL. Histologic criteria include a dense dermal infiltrate of large pleomorphic cells and the expression of CD30 in at least 75% of tumor cells.3 Primary cutaneous anaplastic large cell lymphoma typically lacks the ALK gene translocation with the nucleophosmin gene, NPM, that is common in systemic disease; however, a small subset of PC-ALCL may be ALK positive and indicate a higher chance of transformation into systemic disease.2
The extent of the lymphoma should be staged to exclude the possibility of systemic disease. This assessment includes a complete physical examination; laboratory investigation, including complete blood cell count with differential and blood chemistries; and radiography. A positron emission tomography-CT scan of the neck, chest, abdomen, and pelvis, or a whole-body integrated positron emission tomography-CT are sufficient for the radiographic examination.3
The initial choice of treatment for solitary or localized PC-ALCL is localized radiation therapy or low-dose methotrexate. Targeted therapy such as brentuximab has been shown to be effective for those with multifocal systemic involvement or refractory disease.2 Cure rates from radiation therapy alone approach 95%.3 It is important to highlight radiation therapy as the initial management plan to increase awareness and to avoid inappropriate treatment of PC-ALCL with traditional chemotherapy.
Large lesions of LyP may appear similar to PC-ALCL on histopathology, making the two entities difficult to distinguish. However, in contrast to PC-ALCL, LyP classically has a different clinical course characterized by waxing and waning crops of lesions that typically are smaller (<1 cm) than those of PC-ALCL.2 Large cell transformation of mycosis fungoides is another entity to consider, but these patients usually have a known history of mycosis fungoides.4
Keratoacanthomas, considered to be a variant of a well-differentiated squamous cell carcinoma, present as rapidly enlarging crateriform nodules with a keratotic core. They usually are found on the head and neck or sun-exposed areas of the extremities and may regress spontaneously.5 Histology will show atypical, highly differentiated squamous epithelia. Merkel cell carcinoma also has a predilection for the head and neck in older patients and may present as a rapidly growing nodule. However, histology will show an aggressive tumor with small round blue cells, and immunohistochemistry will show the characteristic paranuclear dot staining for CK20 along with staining for various neuroendocrine markers. Similarly, atypical fibroxanthoma is a low-grade sarcoma that also presents on the head and neck of elderly sun-damaged patients.5 Histology will show dermal proliferation of spindle cells that often extend up against the epidermis along with pleomorphism and atypical mitoses. Basal cell carcinoma is a common tumor that can present on the head and neck in sun-damaged patients. Nodular basal cell carcinomas can enlarge and ulcerate, but growth is seen over years rather than weeks.5 Histology characteristically will show tumor islands composed of basaloid cells with peripheral palisading and clefting between the tumor islands and the stroma.
The Diagnosis: Primary Cutaneous Anaplastic Large Cell Lymphoma
Microscopic analysis showed a dense proliferation of mononuclear cells filling and expanding the dermis with focal epidermotropism (Figure 1). Immunohistochemistry demonstrated strong and diffuse staining for CD3, CD4, and CD30 (Figure 2) and lack of staining for anaplastic lymphoma kinase (ALK). Workup to exclude systemic disease was initiated and included unremarkable computed tomography (CT) of the neck, chest, abdomen, and pelvis along with no abnormal cells on bone marrow biopsy. Complete blood cell count, basic metabolic panel, and lactate dehydrogenase were within reference range. Given the lack of evidence for systemic involvement, a diagnosis of primary cutaneous anaplastic large cell lymphoma (PC-ALCL) was made. The treatment plan for our patient with a solitary lesion was localized radiation therapy.


Primary cutaneous CD30+ lymphoproliferative disorders encompass a spectrum of conditions, with premalignant lymphomatoid papulosis (LyP) at one extreme and the malignant PC-ALCL on the other.1 The diagnosis of PC-ALCL is made by clinicopathologic correlation, and lesions typically present abruptly as solitary or grouped nodules with a tendency to ulcerate over time. Spontaneous regression has been reported, but relapse in the skin is frequent.2
A representative, typically excisional, biopsy should be performed if the clinician suspects PC-ALCL. Histologic criteria include a dense dermal infiltrate of large pleomorphic cells and the expression of CD30 in at least 75% of tumor cells.3 Primary cutaneous anaplastic large cell lymphoma typically lacks the ALK gene translocation with the nucleophosmin gene, NPM, that is common in systemic disease; however, a small subset of PC-ALCL may be ALK positive and indicate a higher chance of transformation into systemic disease.2
The extent of the lymphoma should be staged to exclude the possibility of systemic disease. This assessment includes a complete physical examination; laboratory investigation, including complete blood cell count with differential and blood chemistries; and radiography. A positron emission tomography-CT scan of the neck, chest, abdomen, and pelvis, or a whole-body integrated positron emission tomography-CT are sufficient for the radiographic examination.3
The initial choice of treatment for solitary or localized PC-ALCL is localized radiation therapy or low-dose methotrexate. Targeted therapy such as brentuximab has been shown to be effective for those with multifocal systemic involvement or refractory disease.2 Cure rates from radiation therapy alone approach 95%.3 It is important to highlight radiation therapy as the initial management plan to increase awareness and to avoid inappropriate treatment of PC-ALCL with traditional chemotherapy.
Large lesions of LyP may appear similar to PC-ALCL on histopathology, making the two entities difficult to distinguish. However, in contrast to PC-ALCL, LyP classically has a different clinical course characterized by waxing and waning crops of lesions that typically are smaller (<1 cm) than those of PC-ALCL.2 Large cell transformation of mycosis fungoides is another entity to consider, but these patients usually have a known history of mycosis fungoides.4
Keratoacanthomas, considered to be a variant of a well-differentiated squamous cell carcinoma, present as rapidly enlarging crateriform nodules with a keratotic core. They usually are found on the head and neck or sun-exposed areas of the extremities and may regress spontaneously.5 Histology will show atypical, highly differentiated squamous epithelia. Merkel cell carcinoma also has a predilection for the head and neck in older patients and may present as a rapidly growing nodule. However, histology will show an aggressive tumor with small round blue cells, and immunohistochemistry will show the characteristic paranuclear dot staining for CK20 along with staining for various neuroendocrine markers. Similarly, atypical fibroxanthoma is a low-grade sarcoma that also presents on the head and neck of elderly sun-damaged patients.5 Histology will show dermal proliferation of spindle cells that often extend up against the epidermis along with pleomorphism and atypical mitoses. Basal cell carcinoma is a common tumor that can present on the head and neck in sun-damaged patients. Nodular basal cell carcinomas can enlarge and ulcerate, but growth is seen over years rather than weeks.5 Histology characteristically will show tumor islands composed of basaloid cells with peripheral palisading and clefting between the tumor islands and the stroma.
- Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390.
- Brown RA, Fernandez-Pol S, Kim J. Primary cutaneous anaplastic large cell lymphoma. J Cutan Pathol. 2017;44:570-577.
- Kempf W, Pfaltz K, Vermeer MH, et al. EORTC, ISCL, and USCLC consensus recommendations for the treatment of primary cutaneous CD30-positive lymphoproliferative disorders: lymphomatoid papulosis and primary cutaneous anaplastic large-cell lymphoma. Blood. 2011;118:4024-4035.
- Jawed SI, Myskowski PL, Horwitz S, et al. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part II. prognosis, management, and future directions. J Am Acad Dermatol. 2014;70:223.e1-17.
- Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. Saunders Elsevier; 2015:475-489.
- Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390.
- Brown RA, Fernandez-Pol S, Kim J. Primary cutaneous anaplastic large cell lymphoma. J Cutan Pathol. 2017;44:570-577.
- Kempf W, Pfaltz K, Vermeer MH, et al. EORTC, ISCL, and USCLC consensus recommendations for the treatment of primary cutaneous CD30-positive lymphoproliferative disorders: lymphomatoid papulosis and primary cutaneous anaplastic large-cell lymphoma. Blood. 2011;118:4024-4035.
- Jawed SI, Myskowski PL, Horwitz S, et al. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part II. prognosis, management, and future directions. J Am Acad Dermatol. 2014;70:223.e1-17.
- Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. Saunders Elsevier; 2015:475-489.
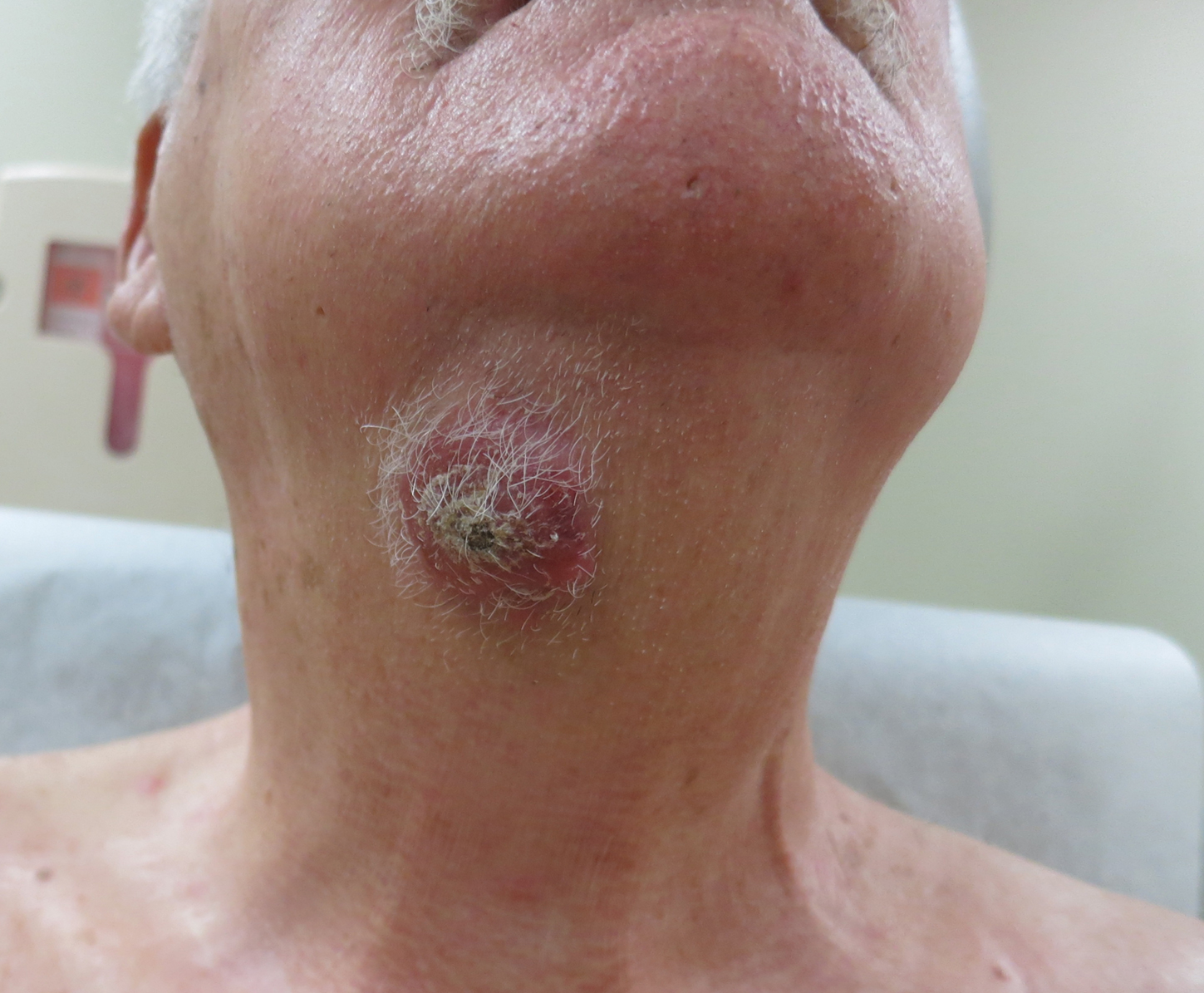
An 80-year-old man presented to our clinic with a large lesion on the right upper neck of approximately 4 weeks’ duration. He reported that it was rapidly increasing in size and had bled on several occasions. No treatments were attempted prior to the initial visit. He denied any constitutional symptoms. The patient had a history of nonmelanoma skin cancers but no other chronic medical problems. Physical examination revealed a large, 35×40-mm, erythematous nodule with central ulceration and overlying hyperkeratosis on the right upper neck. No palpable cervical, supraclavicular, or axillary lymphadenopathy was observed. An excisional biopsy of the lesion was obtained.
‘Malicious peer review’ destroyed doc’s career, he says
Cardiothoracic surgeon J. Marvin Smith III, MD, had always thrived on a busy practice schedule, often performing 20-30 surgeries a week. A practicing surgeon for more than 40 years, Dr. Smith said he had no plans to slow down anytime soon.
But Dr. Smith said his career was derailed when leaders at Methodist Healthcare System of San Antonio initiated a sudden peer review proceeding against him. The hospital system alleged certain surgeries performed by Dr. Smith had excessive mortality rates. When he proved the data inaccurate, Dr. Smith said administrators next claimed he was cognitively impaired and wasn’t safe to practice.
Dr. Smith has now been embroiled in a peer review dispute with the hospital system for more than 2 years and says the conflict has essentially forced him out of surgical practice. He believes the peer review was “malicious” and was really launched because of complaints he made about nurse staffing and other issues at the hospital.
“I think it is absolutely in bad faith and is disingenuous what they’ve told me along the way,” said Dr. Smith, 73. “It’s because I pointed out deficiencies in nursing care, and they want to get rid of me. It would be a lot easier for them if I had a contract and they could control me better. But the fact that I was independent, meant they had to resort to a malicious peer review to try and push me out.”
Dr. Smith had a peer review hearing with Methodist in March 2021, and in April, a panel found in Dr. Smith’s favor, according to Dr. Smith. The findings were sent to the hospital’s medical board for review, which issued a decision in early May.
Eric A. Pullen, an attorney for Dr. Smith, said he could not go into detail about the board’s decision for legal reasons, but that “the medical board’s decision did not completely resolve the matter, and Dr. Smith intends to exercise his procedural rights, which could include an appeal.”
Methodist Hospital Texsan and its parent company, Methodist Health System of San Antonio, did not respond to messages seeking comment about the case. Without hearing from the hospital system, its side is unknown and it is unclear if there is more to the story from Methodist’s view.
The problem is not new, but some experts, such as Lawrence Huntoon, MD, PhD, say the practice has become more common in recent years, particularly against independent doctors.
Dr. Huntoon believes there is a nationwide trend at many hospitals to get rid of independent physicians and replace them with employed doctors, he said.
However, because most sham peer reviews go on behind closed doors, there are no data to pinpoint its prevalence or measure its growth.
“Independent physicians are basically being purged from medical staffs across the United States,” said Dr. Huntoon, who is chair of the Association of American Physicians and Surgeons’ Committee to Combat Sham Peer Review. “The hospitals want more control over how physicians practice and who they refer to, and they do that by having employees.”
Anthony P. Weiss, MD, MBA, chief medical officer for Beth Israel Deaconess Medical Center said it has not been his experience that independent physicians are being targeted in such a way. Dr. Weiss responded to an inquiry sent to the American Hospital Association for this story.
“As the authority for peer review rests with the organized medical staff (i.e., physicians), and not formally with the hospital per se, the peer review lever is not typically available as a management tool for hospital administration,” said Dr. Weiss, who is a former member of the AHA’s Committee on Clinical Leadership, but who was speaking on behalf of himself.
A spokesman for the AHA said the organization stands behinds Dr. Weiss’ comments.
Peer review remains a foundational aspect of overseeing the safety and appropriateness of healthcare provided by physicians, Dr. Weiss said. Peer review likely varies from hospital to hospital, he added, although the Healthcare Quality Improvement Act provides some level of guidance as does the American Medical Association Code of Medical Ethics (section 9.4.1).
“In essence, both require that the evaluation be conducted in good faith with the intention to improve care, by physicians with adequate training and knowledge, using a process that is fair and inclusive of the physician under review,” he said. “I believe that most medical staffs abide by these ethical principles, but we have little data to confirm this supposition.”
Did hospital target doc for being vocal?
When members of Methodist’s medical staff first approached Dr. Smith with concerns about his surgery outcomes in November 2018, the physician says he was surprised, but that he was open to an assessment.
“They came to me and said they thought my numbers were bad, and I said: ‘Well my gosh, I certainly don’t want that to be the case. I need to see what numbers you are talking about,’ ” Dr. Smith recalled. “I’ve been president of the Bexar County Medical Society; I’ve been involved with standards and ethics for the Society of Thoracic Surgeons. Quality health care means a whole lot to me.”
The statistical information provided by hospital administrators indicated that Dr. Smith’s mortality rates for coronary artery surgery in 2018 were “excessive” and that his rates for aortic surgery were “unacceptable,” according to a lawsuit Dr. Smith filed against the hospital system. Dr. Smith, who is double boarded with the American Board of Surgery and the American Board of Thoracic Surgery, said his outcomes had never come into question in the past. Dr. Smith said the timing was suspicious to him, however, considering he had recently raised concerns with the hospital through letters about nursing performance, staffing, and compensation.
A peer review investigation was initiated. In the meantime, Dr. Smith agreed to intensivist consults on his postoperative patients and consults with the hospital’s “Heart Team” on all preoperative cardiac, valve, and aortic cases. A vocal critic of the Heart Team, Dr. Smith had long contended the entity provided no meaningful benefit to his patients in most cases and, rather, increased hospital stays and raised medical expenses. Despite his agreement, Dr. Smith was later asked to voluntarily stop performing surgeries at the hospital.
“I agreed, convinced that we’d get this all settled,” he said.
Another report issued by the hospital in 2019 also indicated elevated mortality rates associated with some of Smith’s surgeries, although the document differed from the first report, according to the lawsuit. Dr. Smith says he was ignored when he pointed out problems with the data, including a lack of appropriate risk stratification in the report, departure from Society of Thoracic Surgeons data rules, and improper inclusion of his cases in the denominator of the ratio when a comparison was made of his outcomes with those hospitalwide. A subsequent report from Methodist in March 2019 indicated Dr. Smith’s surgery outcomes were “within the expected parameters of performance,” according to court documents.
The surgery accusations were dropped, but the peer review proceeding against Dr. Smith wasn’t over. The hospital next requested that Dr. Smith undergo a competency evaluation.
“When they realized the data was bad, they then changed their argument in the peer review proceeding and essentially started to argue that Dr. Smith had some sort of cognitive disability that prevented him from continuing to practice,” said Mr. Pullen. “The way I look at it, when the initial basis for the peer review was proven false, the hospital found something else and some other reason to try to keep Dr. Smith from practicing.”
Thus began a lengthy disagreement about which entity would conduct the evaluation, who would pay, and the type of acceptable assessment. An evaluation by the hospital’s preferred organization resulted in a finding of mild cognitive impairment, Dr. Smith said. He hired his own experts who conducted separate evaluations, finding no impairment and no basis for the former evaluation’s conclusion.
“Literally, the determinant as to whether I was normal or below normal on their test was one point, which was associated with a finding that I didn’t draw a clock correctly,” Dr. Smith claimed. “The reviewer said my minute hand was a little too short and docked me a point. It was purely subjective. To me, the gold standard of whether you are learned in thoracic surgery is the American Board of Thoracic Surgery’s test. The board’s test shows my cognitive ability is entirely in keeping with my practice. That contrasts with the one point off I got for drawing a clock wrong in somebody’s estimation.”
Conflict leads to legal case
In September 2020, Dr. Smith filed a lawsuit against Methodist Healthcare System of San Antonio, alleging business disparagement by Methodist for allegedly publishing false and disparaging information about Dr. Smith and tortious interference with business relations. The latter claim stems from Methodist refusing to provide documents to other hospitals about the status of Dr. Smith’s privileges at Methodist, Mr. Pullen said.
Because Methodist refused to confirm his status, the renewal process for Baptist Health System could not be completed and Dr. Smith lost his privileges at Baptist Health System facilities, according to the lawsuit.
Notably, Dr. Smith’s legal challenge also asks the court to take a stance against alleged amendments by Methodist to its Unified Medical Staff Bylaws. The hospital allegedly proposed changes that would prevent physicians from seeking legal action against the hospital for malicious peer review, according to Dr. Smith’s lawsuit.
The amendments would make the peer review process itself the “sole and exclusive remedy with respect to any action or recommendation taken at the hospital affecting medical staff appointment and/or clinical privileges,” according to an excerpt of the proposed amendments included in Dr. Smith’s lawsuit. In addition, the changes would hold practitioners liable for lost revenues if the doctor initiates “any type of legal action challenging credentialing, privileging, or other medical peer review or professional review activity,” according to the lawsuit.
Dr. Smith’s lawsuit seeks a declaration that the proposed amendments to the bylaws are “void as against public policy,” and a declaration that the proposed amendments to the bylaws cannot take away physicians’ statutory right to bring litigation against Methodist for malicious peer review.
“The proposed amendments have a tendency to and will injure the public good,” Dr. Smith argued in the lawsuit. “The proposed amendments allow Methodist to act with malice and in bad faith in conducting peer review proceedings and face no legal repercussions.”
Regardless of the final outcome of the peer review proceeding, Mr. Pullen said the harm Dr. Smith has already endured cannot be reversed.
“Even if comes out in his favor, the damage is already done,” he said. “It will not remedy the damage Dr. Smith has incurred.”
Fighting sham peer review is difficult
Battling a malicious peer review has long been an uphill battle for physicians, according to Dr. Huntoon. That’s because the Health Care Quality Improvement Act (HCQIA), a federal law passed in 1986, provides near absolute immunity to hospitals and peer reviewers in legal disputes.
The HCQIA was created by Congress to extend immunity to good-faith peer review of doctors and to increase overall participation in peer review by removing fear of litigation. However, the act has also enabled abuse of peer review by shielding bad-faith reviewers from accountability, said Dr. Huntoon.
“The Health Care Quality Improvement Act presumes that what the hospital did was warranted and reasonable and shifts the burden to the physician to prove his innocence by a preponderance of evidence,” he said. “That’s an entirely foreign concept to most people who think a person should be considered innocent until proven guilty. Here, it’s the exact opposite.”
The HCQIA has been challenged numerous times over the years and tested at the appellate level, but continues to survive and remain settled law, added Richard B. Willner, DPM, founder and director of the Center for Peer Review Justice, which assists and counsels physicians about sham peer review.
In 2011, former Rep. Joe Heck, DO, (R-Nev.) introduced a bill that would have amended the HCQIA to prohibit a professional review entity from submitting a report to the National Practitioner Data Bank (NPDB) while the doctor was still under investigation and before the doctor was afforded adequate notice and a hearing. Although the measure had 16 cosponsors and plenty of support from the physician community, it failed.
In addition to a heavy legal burden, physicians who experience malicious peer reviews also face ramifications from being reported to the NPDB. Peer review organizations are required to report certain negative actions or findings to the NPDB.
“A databank entry is a scarlet letter on your forehead,” Dr. Willner said. “The rules at a lot of institutions are not to take anyone who has been databanked, rightfully or wrongfully. And what is the evidence necessary to databank you? None. There’s no evidence needed to databank somebody.”
Despite the bleak landscape, experts say progress has been made on a case-by-case basis by physicians who have succeeded in fighting back against questionable peer reviews in recent years.
In January 2020, Indiana ob.gyn. Rebecca Denman, MD, prevailed in her defamation lawsuit against St Vincent Carmel Hospital and St Vincent Carmel Medical Group, winning $4.75 million in damages. Dr. Denman alleged administrators failed to conduct a proper peer review investigation after a false allegation by a nurse that she was under the influence while on the job.
Indianapolis attorney Kathleen A. DeLaney, who represented Dr. Denman, said hospital leaders misled Dr. Denman into believing a peer review had occurred when no formal peer review hearing or proceeding took place.
“The CMO of the medical group claimed that he performed a peer review ‘screening,’ but he never informed the other members of the peer review executive committee of the matter until after he had placed Dr. Denman on administrative leave,” Ms. DeLaney said. “He also neglected to tell the peer review executive committee that the substance abuse policy had not been followed, or that Dr. Denman had not been tested for alcohol use – due to the 12-hour delay in report.”
Dr. Denman was ultimately required to undergo an alcohol abuse evaluation, enter a treatment program, and sign a 5-year monitoring contract with the Indiana State Medical Association as a condition of her employment, according to the lawsuit. She claimed repercussions from the false allegation resulted in lost compensation, out-of-pocket expenses, emotional distress, and damage to her professional reputation.
She sued the hospital in July 2018, alleging fraud, defamation, tortious interference with an employment relationship, and negligent misrepresentation. After a 4-day trial, jurors found in her favor, awarding Dr. Denman $2 million for her defamation claims, $2 million for her claims of fraud and constructive fraud, $500,000 for her claim of tortious interference with an employment relationship, and $250,000 for her claim of negligent misrepresentation.
A hospital spokesperson said Ascension St Vincent is pursuing an appeal, and that it looks “forward to the opportunity to bring this matter before the Indiana Court of Appeals in June.”
In another case, South Dakota surgeon Linda Miller, MD, was awarded $1.1 million in 2017 after a federal jury found Huron Regional Medical Center breached her contract and violated her due process rights. Dr. Miller became the subject of a peer review at Huron Regional Medical Center when the hospital began analyzing some of her surgery outcomes.
Ken Barker, an attorney for Dr. Miller, said he feels it became evident at trial that the campaign to force Dr. Miller to either resign or lose her privileges was led by the lay board of directors of the hospital and upper-level administration at the hospital.
“They began the process by ordering an unprecedented 90-day review of her medical charts, looking for errors in the medical care she provided patients,” he said. “They could find nothing, so they did a second 90-day review, waiting for a patient’s ‘bad outcome.’ As any general surgeon will say, a ‘bad outcome’ is inevitable. And so it was. Upon that occurrence, they had a medical review committee review the patient’s chart and use it as an excuse to force her to reduce her privileges. Unbeknown to Dr. Miller, an external review had been conducted on another patient’s chart, in which the external review found her care above the standards and, in some measure, ‘exemplary.’ ”
Dr. Miller was eventually pressured to resign, according to her claim. Because of reports made to the NPDB by the medical center, including a patient complication that was allegedly falsified by the hospital, Dr. Miller said she was unable to find work as a general surgeon and went to work as a wound care doctor. At trial, jurors awarded Dr. Miller $586,617 in lost wages, $343,640 for lost future earning capacity, and $250,000 for mental anguish. (The mental anguish award was subsequently struck by a district court.)
Attorneys for Huron Regional Medical Center argued the jury improperly awarded damages and requested a new trial, which was denied by an appeals court.
In the end, the evidence came to light and the jury’s verdict spoke loudly that the hospital had taken unfair advantage of Dr. Miller, Mr. Barker said. But he emphasized that such cases often end differently.
“There are a handful of cases in which physicians like Dr. Miller have challenged the system and won,” he said. “In most cases, however, it is a ‘David vs. Goliath’ scenario where the giant prevails.”
What to do if faced with malicious peer review
An important step when doctors encounter a peer review that they believe is malicious is to consult with an experienced attorney as early as possible, Dr. Huntoon said. “Not all attorneys who set themselves out to be health law attorneys necessarily have knowledge and expertise in sham peer review. And before such a thing happens, I always encourage physicians to read their medical staff bylaws. That’s where everything is set forth, [such as] the corrective action section that tells how peer review is to take place.”
Mr. Barker added that documentation is also key in the event of a potential malicious peer review.
“When a physician senses [the] administration has targeted them, they should start documenting their conversations and actions very carefully, and if possible, recruit another ‘observer’ who can provide a third-party perspective, if necessary,” Mr. Barker said.
Dr. Huntoon recently wrote an article with advice about preparedness and defense of sham peer reviews. The guidance includes that physicians educate themselves about the tactics used by some hospitals to conduct sham peer reviews and the factors that place doctors more at risk. Factors that may raise a doctor’s danger of being targeted include being in solo practice or a small group, being new on staff, or being an older physician approaching retirement as some bad-actor hospitals may view older physicians as being less likely to fight back, said Dr. Huntoon.
Doctors should also keep detailed records and a timeline in the event of a malicious peer review and insist that an independent court reporter record all peer review hearings, even if that means the physician has to pay for the reporter him or herself, according to the guidance. An independent record is invaluable should the physician ultimately issue a future legal challenge against the hospital.
Mr. Willner encourages physicians to call the Center for Peer Review Justice hotline at (504) 621-1670 or visit the website for help with peer review and NPDB issues.
As for Dr. Smith, his days are much quieter and slower today, compared with the active practice he was accustomed to for more than half his life. He misses the fast pace, the patients, and the work that always brought him great joy.
“I hope to get back to doing surgeries eventually,” he said. “I graduated medical school in 1972. Practicing surgery has been my whole life and my career. They have taken my identity and my livelihood away from me based on false numbers and false premises. I want it back.”
A version of this article first appeared on Medscape.com.
Cardiothoracic surgeon J. Marvin Smith III, MD, had always thrived on a busy practice schedule, often performing 20-30 surgeries a week. A practicing surgeon for more than 40 years, Dr. Smith said he had no plans to slow down anytime soon.
But Dr. Smith said his career was derailed when leaders at Methodist Healthcare System of San Antonio initiated a sudden peer review proceeding against him. The hospital system alleged certain surgeries performed by Dr. Smith had excessive mortality rates. When he proved the data inaccurate, Dr. Smith said administrators next claimed he was cognitively impaired and wasn’t safe to practice.
Dr. Smith has now been embroiled in a peer review dispute with the hospital system for more than 2 years and says the conflict has essentially forced him out of surgical practice. He believes the peer review was “malicious” and was really launched because of complaints he made about nurse staffing and other issues at the hospital.
“I think it is absolutely in bad faith and is disingenuous what they’ve told me along the way,” said Dr. Smith, 73. “It’s because I pointed out deficiencies in nursing care, and they want to get rid of me. It would be a lot easier for them if I had a contract and they could control me better. But the fact that I was independent, meant they had to resort to a malicious peer review to try and push me out.”
Dr. Smith had a peer review hearing with Methodist in March 2021, and in April, a panel found in Dr. Smith’s favor, according to Dr. Smith. The findings were sent to the hospital’s medical board for review, which issued a decision in early May.
Eric A. Pullen, an attorney for Dr. Smith, said he could not go into detail about the board’s decision for legal reasons, but that “the medical board’s decision did not completely resolve the matter, and Dr. Smith intends to exercise his procedural rights, which could include an appeal.”
Methodist Hospital Texsan and its parent company, Methodist Health System of San Antonio, did not respond to messages seeking comment about the case. Without hearing from the hospital system, its side is unknown and it is unclear if there is more to the story from Methodist’s view.
The problem is not new, but some experts, such as Lawrence Huntoon, MD, PhD, say the practice has become more common in recent years, particularly against independent doctors.
Dr. Huntoon believes there is a nationwide trend at many hospitals to get rid of independent physicians and replace them with employed doctors, he said.
However, because most sham peer reviews go on behind closed doors, there are no data to pinpoint its prevalence or measure its growth.
“Independent physicians are basically being purged from medical staffs across the United States,” said Dr. Huntoon, who is chair of the Association of American Physicians and Surgeons’ Committee to Combat Sham Peer Review. “The hospitals want more control over how physicians practice and who they refer to, and they do that by having employees.”
Anthony P. Weiss, MD, MBA, chief medical officer for Beth Israel Deaconess Medical Center said it has not been his experience that independent physicians are being targeted in such a way. Dr. Weiss responded to an inquiry sent to the American Hospital Association for this story.
“As the authority for peer review rests with the organized medical staff (i.e., physicians), and not formally with the hospital per se, the peer review lever is not typically available as a management tool for hospital administration,” said Dr. Weiss, who is a former member of the AHA’s Committee on Clinical Leadership, but who was speaking on behalf of himself.
A spokesman for the AHA said the organization stands behinds Dr. Weiss’ comments.
Peer review remains a foundational aspect of overseeing the safety and appropriateness of healthcare provided by physicians, Dr. Weiss said. Peer review likely varies from hospital to hospital, he added, although the Healthcare Quality Improvement Act provides some level of guidance as does the American Medical Association Code of Medical Ethics (section 9.4.1).
“In essence, both require that the evaluation be conducted in good faith with the intention to improve care, by physicians with adequate training and knowledge, using a process that is fair and inclusive of the physician under review,” he said. “I believe that most medical staffs abide by these ethical principles, but we have little data to confirm this supposition.”
Did hospital target doc for being vocal?
When members of Methodist’s medical staff first approached Dr. Smith with concerns about his surgery outcomes in November 2018, the physician says he was surprised, but that he was open to an assessment.
“They came to me and said they thought my numbers were bad, and I said: ‘Well my gosh, I certainly don’t want that to be the case. I need to see what numbers you are talking about,’ ” Dr. Smith recalled. “I’ve been president of the Bexar County Medical Society; I’ve been involved with standards and ethics for the Society of Thoracic Surgeons. Quality health care means a whole lot to me.”
The statistical information provided by hospital administrators indicated that Dr. Smith’s mortality rates for coronary artery surgery in 2018 were “excessive” and that his rates for aortic surgery were “unacceptable,” according to a lawsuit Dr. Smith filed against the hospital system. Dr. Smith, who is double boarded with the American Board of Surgery and the American Board of Thoracic Surgery, said his outcomes had never come into question in the past. Dr. Smith said the timing was suspicious to him, however, considering he had recently raised concerns with the hospital through letters about nursing performance, staffing, and compensation.
A peer review investigation was initiated. In the meantime, Dr. Smith agreed to intensivist consults on his postoperative patients and consults with the hospital’s “Heart Team” on all preoperative cardiac, valve, and aortic cases. A vocal critic of the Heart Team, Dr. Smith had long contended the entity provided no meaningful benefit to his patients in most cases and, rather, increased hospital stays and raised medical expenses. Despite his agreement, Dr. Smith was later asked to voluntarily stop performing surgeries at the hospital.
“I agreed, convinced that we’d get this all settled,” he said.
Another report issued by the hospital in 2019 also indicated elevated mortality rates associated with some of Smith’s surgeries, although the document differed from the first report, according to the lawsuit. Dr. Smith says he was ignored when he pointed out problems with the data, including a lack of appropriate risk stratification in the report, departure from Society of Thoracic Surgeons data rules, and improper inclusion of his cases in the denominator of the ratio when a comparison was made of his outcomes with those hospitalwide. A subsequent report from Methodist in March 2019 indicated Dr. Smith’s surgery outcomes were “within the expected parameters of performance,” according to court documents.
The surgery accusations were dropped, but the peer review proceeding against Dr. Smith wasn’t over. The hospital next requested that Dr. Smith undergo a competency evaluation.
“When they realized the data was bad, they then changed their argument in the peer review proceeding and essentially started to argue that Dr. Smith had some sort of cognitive disability that prevented him from continuing to practice,” said Mr. Pullen. “The way I look at it, when the initial basis for the peer review was proven false, the hospital found something else and some other reason to try to keep Dr. Smith from practicing.”
Thus began a lengthy disagreement about which entity would conduct the evaluation, who would pay, and the type of acceptable assessment. An evaluation by the hospital’s preferred organization resulted in a finding of mild cognitive impairment, Dr. Smith said. He hired his own experts who conducted separate evaluations, finding no impairment and no basis for the former evaluation’s conclusion.
“Literally, the determinant as to whether I was normal or below normal on their test was one point, which was associated with a finding that I didn’t draw a clock correctly,” Dr. Smith claimed. “The reviewer said my minute hand was a little too short and docked me a point. It was purely subjective. To me, the gold standard of whether you are learned in thoracic surgery is the American Board of Thoracic Surgery’s test. The board’s test shows my cognitive ability is entirely in keeping with my practice. That contrasts with the one point off I got for drawing a clock wrong in somebody’s estimation.”
Conflict leads to legal case
In September 2020, Dr. Smith filed a lawsuit against Methodist Healthcare System of San Antonio, alleging business disparagement by Methodist for allegedly publishing false and disparaging information about Dr. Smith and tortious interference with business relations. The latter claim stems from Methodist refusing to provide documents to other hospitals about the status of Dr. Smith’s privileges at Methodist, Mr. Pullen said.
Because Methodist refused to confirm his status, the renewal process for Baptist Health System could not be completed and Dr. Smith lost his privileges at Baptist Health System facilities, according to the lawsuit.
Notably, Dr. Smith’s legal challenge also asks the court to take a stance against alleged amendments by Methodist to its Unified Medical Staff Bylaws. The hospital allegedly proposed changes that would prevent physicians from seeking legal action against the hospital for malicious peer review, according to Dr. Smith’s lawsuit.
The amendments would make the peer review process itself the “sole and exclusive remedy with respect to any action or recommendation taken at the hospital affecting medical staff appointment and/or clinical privileges,” according to an excerpt of the proposed amendments included in Dr. Smith’s lawsuit. In addition, the changes would hold practitioners liable for lost revenues if the doctor initiates “any type of legal action challenging credentialing, privileging, or other medical peer review or professional review activity,” according to the lawsuit.
Dr. Smith’s lawsuit seeks a declaration that the proposed amendments to the bylaws are “void as against public policy,” and a declaration that the proposed amendments to the bylaws cannot take away physicians’ statutory right to bring litigation against Methodist for malicious peer review.
“The proposed amendments have a tendency to and will injure the public good,” Dr. Smith argued in the lawsuit. “The proposed amendments allow Methodist to act with malice and in bad faith in conducting peer review proceedings and face no legal repercussions.”
Regardless of the final outcome of the peer review proceeding, Mr. Pullen said the harm Dr. Smith has already endured cannot be reversed.
“Even if comes out in his favor, the damage is already done,” he said. “It will not remedy the damage Dr. Smith has incurred.”
Fighting sham peer review is difficult
Battling a malicious peer review has long been an uphill battle for physicians, according to Dr. Huntoon. That’s because the Health Care Quality Improvement Act (HCQIA), a federal law passed in 1986, provides near absolute immunity to hospitals and peer reviewers in legal disputes.
The HCQIA was created by Congress to extend immunity to good-faith peer review of doctors and to increase overall participation in peer review by removing fear of litigation. However, the act has also enabled abuse of peer review by shielding bad-faith reviewers from accountability, said Dr. Huntoon.
“The Health Care Quality Improvement Act presumes that what the hospital did was warranted and reasonable and shifts the burden to the physician to prove his innocence by a preponderance of evidence,” he said. “That’s an entirely foreign concept to most people who think a person should be considered innocent until proven guilty. Here, it’s the exact opposite.”
The HCQIA has been challenged numerous times over the years and tested at the appellate level, but continues to survive and remain settled law, added Richard B. Willner, DPM, founder and director of the Center for Peer Review Justice, which assists and counsels physicians about sham peer review.
In 2011, former Rep. Joe Heck, DO, (R-Nev.) introduced a bill that would have amended the HCQIA to prohibit a professional review entity from submitting a report to the National Practitioner Data Bank (NPDB) while the doctor was still under investigation and before the doctor was afforded adequate notice and a hearing. Although the measure had 16 cosponsors and plenty of support from the physician community, it failed.
In addition to a heavy legal burden, physicians who experience malicious peer reviews also face ramifications from being reported to the NPDB. Peer review organizations are required to report certain negative actions or findings to the NPDB.
“A databank entry is a scarlet letter on your forehead,” Dr. Willner said. “The rules at a lot of institutions are not to take anyone who has been databanked, rightfully or wrongfully. And what is the evidence necessary to databank you? None. There’s no evidence needed to databank somebody.”
Despite the bleak landscape, experts say progress has been made on a case-by-case basis by physicians who have succeeded in fighting back against questionable peer reviews in recent years.
In January 2020, Indiana ob.gyn. Rebecca Denman, MD, prevailed in her defamation lawsuit against St Vincent Carmel Hospital and St Vincent Carmel Medical Group, winning $4.75 million in damages. Dr. Denman alleged administrators failed to conduct a proper peer review investigation after a false allegation by a nurse that she was under the influence while on the job.
Indianapolis attorney Kathleen A. DeLaney, who represented Dr. Denman, said hospital leaders misled Dr. Denman into believing a peer review had occurred when no formal peer review hearing or proceeding took place.
“The CMO of the medical group claimed that he performed a peer review ‘screening,’ but he never informed the other members of the peer review executive committee of the matter until after he had placed Dr. Denman on administrative leave,” Ms. DeLaney said. “He also neglected to tell the peer review executive committee that the substance abuse policy had not been followed, or that Dr. Denman had not been tested for alcohol use – due to the 12-hour delay in report.”
Dr. Denman was ultimately required to undergo an alcohol abuse evaluation, enter a treatment program, and sign a 5-year monitoring contract with the Indiana State Medical Association as a condition of her employment, according to the lawsuit. She claimed repercussions from the false allegation resulted in lost compensation, out-of-pocket expenses, emotional distress, and damage to her professional reputation.
She sued the hospital in July 2018, alleging fraud, defamation, tortious interference with an employment relationship, and negligent misrepresentation. After a 4-day trial, jurors found in her favor, awarding Dr. Denman $2 million for her defamation claims, $2 million for her claims of fraud and constructive fraud, $500,000 for her claim of tortious interference with an employment relationship, and $250,000 for her claim of negligent misrepresentation.
A hospital spokesperson said Ascension St Vincent is pursuing an appeal, and that it looks “forward to the opportunity to bring this matter before the Indiana Court of Appeals in June.”
In another case, South Dakota surgeon Linda Miller, MD, was awarded $1.1 million in 2017 after a federal jury found Huron Regional Medical Center breached her contract and violated her due process rights. Dr. Miller became the subject of a peer review at Huron Regional Medical Center when the hospital began analyzing some of her surgery outcomes.
Ken Barker, an attorney for Dr. Miller, said he feels it became evident at trial that the campaign to force Dr. Miller to either resign or lose her privileges was led by the lay board of directors of the hospital and upper-level administration at the hospital.
“They began the process by ordering an unprecedented 90-day review of her medical charts, looking for errors in the medical care she provided patients,” he said. “They could find nothing, so they did a second 90-day review, waiting for a patient’s ‘bad outcome.’ As any general surgeon will say, a ‘bad outcome’ is inevitable. And so it was. Upon that occurrence, they had a medical review committee review the patient’s chart and use it as an excuse to force her to reduce her privileges. Unbeknown to Dr. Miller, an external review had been conducted on another patient’s chart, in which the external review found her care above the standards and, in some measure, ‘exemplary.’ ”
Dr. Miller was eventually pressured to resign, according to her claim. Because of reports made to the NPDB by the medical center, including a patient complication that was allegedly falsified by the hospital, Dr. Miller said she was unable to find work as a general surgeon and went to work as a wound care doctor. At trial, jurors awarded Dr. Miller $586,617 in lost wages, $343,640 for lost future earning capacity, and $250,000 for mental anguish. (The mental anguish award was subsequently struck by a district court.)
Attorneys for Huron Regional Medical Center argued the jury improperly awarded damages and requested a new trial, which was denied by an appeals court.
In the end, the evidence came to light and the jury’s verdict spoke loudly that the hospital had taken unfair advantage of Dr. Miller, Mr. Barker said. But he emphasized that such cases often end differently.
“There are a handful of cases in which physicians like Dr. Miller have challenged the system and won,” he said. “In most cases, however, it is a ‘David vs. Goliath’ scenario where the giant prevails.”
What to do if faced with malicious peer review
An important step when doctors encounter a peer review that they believe is malicious is to consult with an experienced attorney as early as possible, Dr. Huntoon said. “Not all attorneys who set themselves out to be health law attorneys necessarily have knowledge and expertise in sham peer review. And before such a thing happens, I always encourage physicians to read their medical staff bylaws. That’s where everything is set forth, [such as] the corrective action section that tells how peer review is to take place.”
Mr. Barker added that documentation is also key in the event of a potential malicious peer review.
“When a physician senses [the] administration has targeted them, they should start documenting their conversations and actions very carefully, and if possible, recruit another ‘observer’ who can provide a third-party perspective, if necessary,” Mr. Barker said.
Dr. Huntoon recently wrote an article with advice about preparedness and defense of sham peer reviews. The guidance includes that physicians educate themselves about the tactics used by some hospitals to conduct sham peer reviews and the factors that place doctors more at risk. Factors that may raise a doctor’s danger of being targeted include being in solo practice or a small group, being new on staff, or being an older physician approaching retirement as some bad-actor hospitals may view older physicians as being less likely to fight back, said Dr. Huntoon.
Doctors should also keep detailed records and a timeline in the event of a malicious peer review and insist that an independent court reporter record all peer review hearings, even if that means the physician has to pay for the reporter him or herself, according to the guidance. An independent record is invaluable should the physician ultimately issue a future legal challenge against the hospital.
Mr. Willner encourages physicians to call the Center for Peer Review Justice hotline at (504) 621-1670 or visit the website for help with peer review and NPDB issues.
As for Dr. Smith, his days are much quieter and slower today, compared with the active practice he was accustomed to for more than half his life. He misses the fast pace, the patients, and the work that always brought him great joy.
“I hope to get back to doing surgeries eventually,” he said. “I graduated medical school in 1972. Practicing surgery has been my whole life and my career. They have taken my identity and my livelihood away from me based on false numbers and false premises. I want it back.”
A version of this article first appeared on Medscape.com.
Cardiothoracic surgeon J. Marvin Smith III, MD, had always thrived on a busy practice schedule, often performing 20-30 surgeries a week. A practicing surgeon for more than 40 years, Dr. Smith said he had no plans to slow down anytime soon.
But Dr. Smith said his career was derailed when leaders at Methodist Healthcare System of San Antonio initiated a sudden peer review proceeding against him. The hospital system alleged certain surgeries performed by Dr. Smith had excessive mortality rates. When he proved the data inaccurate, Dr. Smith said administrators next claimed he was cognitively impaired and wasn’t safe to practice.
Dr. Smith has now been embroiled in a peer review dispute with the hospital system for more than 2 years and says the conflict has essentially forced him out of surgical practice. He believes the peer review was “malicious” and was really launched because of complaints he made about nurse staffing and other issues at the hospital.
“I think it is absolutely in bad faith and is disingenuous what they’ve told me along the way,” said Dr. Smith, 73. “It’s because I pointed out deficiencies in nursing care, and they want to get rid of me. It would be a lot easier for them if I had a contract and they could control me better. But the fact that I was independent, meant they had to resort to a malicious peer review to try and push me out.”
Dr. Smith had a peer review hearing with Methodist in March 2021, and in April, a panel found in Dr. Smith’s favor, according to Dr. Smith. The findings were sent to the hospital’s medical board for review, which issued a decision in early May.
Eric A. Pullen, an attorney for Dr. Smith, said he could not go into detail about the board’s decision for legal reasons, but that “the medical board’s decision did not completely resolve the matter, and Dr. Smith intends to exercise his procedural rights, which could include an appeal.”
Methodist Hospital Texsan and its parent company, Methodist Health System of San Antonio, did not respond to messages seeking comment about the case. Without hearing from the hospital system, its side is unknown and it is unclear if there is more to the story from Methodist’s view.
The problem is not new, but some experts, such as Lawrence Huntoon, MD, PhD, say the practice has become more common in recent years, particularly against independent doctors.
Dr. Huntoon believes there is a nationwide trend at many hospitals to get rid of independent physicians and replace them with employed doctors, he said.
However, because most sham peer reviews go on behind closed doors, there are no data to pinpoint its prevalence or measure its growth.
“Independent physicians are basically being purged from medical staffs across the United States,” said Dr. Huntoon, who is chair of the Association of American Physicians and Surgeons’ Committee to Combat Sham Peer Review. “The hospitals want more control over how physicians practice and who they refer to, and they do that by having employees.”
Anthony P. Weiss, MD, MBA, chief medical officer for Beth Israel Deaconess Medical Center said it has not been his experience that independent physicians are being targeted in such a way. Dr. Weiss responded to an inquiry sent to the American Hospital Association for this story.
“As the authority for peer review rests with the organized medical staff (i.e., physicians), and not formally with the hospital per se, the peer review lever is not typically available as a management tool for hospital administration,” said Dr. Weiss, who is a former member of the AHA’s Committee on Clinical Leadership, but who was speaking on behalf of himself.
A spokesman for the AHA said the organization stands behinds Dr. Weiss’ comments.
Peer review remains a foundational aspect of overseeing the safety and appropriateness of healthcare provided by physicians, Dr. Weiss said. Peer review likely varies from hospital to hospital, he added, although the Healthcare Quality Improvement Act provides some level of guidance as does the American Medical Association Code of Medical Ethics (section 9.4.1).
“In essence, both require that the evaluation be conducted in good faith with the intention to improve care, by physicians with adequate training and knowledge, using a process that is fair and inclusive of the physician under review,” he said. “I believe that most medical staffs abide by these ethical principles, but we have little data to confirm this supposition.”
Did hospital target doc for being vocal?
When members of Methodist’s medical staff first approached Dr. Smith with concerns about his surgery outcomes in November 2018, the physician says he was surprised, but that he was open to an assessment.
“They came to me and said they thought my numbers were bad, and I said: ‘Well my gosh, I certainly don’t want that to be the case. I need to see what numbers you are talking about,’ ” Dr. Smith recalled. “I’ve been president of the Bexar County Medical Society; I’ve been involved with standards and ethics for the Society of Thoracic Surgeons. Quality health care means a whole lot to me.”
The statistical information provided by hospital administrators indicated that Dr. Smith’s mortality rates for coronary artery surgery in 2018 were “excessive” and that his rates for aortic surgery were “unacceptable,” according to a lawsuit Dr. Smith filed against the hospital system. Dr. Smith, who is double boarded with the American Board of Surgery and the American Board of Thoracic Surgery, said his outcomes had never come into question in the past. Dr. Smith said the timing was suspicious to him, however, considering he had recently raised concerns with the hospital through letters about nursing performance, staffing, and compensation.
A peer review investigation was initiated. In the meantime, Dr. Smith agreed to intensivist consults on his postoperative patients and consults with the hospital’s “Heart Team” on all preoperative cardiac, valve, and aortic cases. A vocal critic of the Heart Team, Dr. Smith had long contended the entity provided no meaningful benefit to his patients in most cases and, rather, increased hospital stays and raised medical expenses. Despite his agreement, Dr. Smith was later asked to voluntarily stop performing surgeries at the hospital.
“I agreed, convinced that we’d get this all settled,” he said.
Another report issued by the hospital in 2019 also indicated elevated mortality rates associated with some of Smith’s surgeries, although the document differed from the first report, according to the lawsuit. Dr. Smith says he was ignored when he pointed out problems with the data, including a lack of appropriate risk stratification in the report, departure from Society of Thoracic Surgeons data rules, and improper inclusion of his cases in the denominator of the ratio when a comparison was made of his outcomes with those hospitalwide. A subsequent report from Methodist in March 2019 indicated Dr. Smith’s surgery outcomes were “within the expected parameters of performance,” according to court documents.
The surgery accusations were dropped, but the peer review proceeding against Dr. Smith wasn’t over. The hospital next requested that Dr. Smith undergo a competency evaluation.
“When they realized the data was bad, they then changed their argument in the peer review proceeding and essentially started to argue that Dr. Smith had some sort of cognitive disability that prevented him from continuing to practice,” said Mr. Pullen. “The way I look at it, when the initial basis for the peer review was proven false, the hospital found something else and some other reason to try to keep Dr. Smith from practicing.”
Thus began a lengthy disagreement about which entity would conduct the evaluation, who would pay, and the type of acceptable assessment. An evaluation by the hospital’s preferred organization resulted in a finding of mild cognitive impairment, Dr. Smith said. He hired his own experts who conducted separate evaluations, finding no impairment and no basis for the former evaluation’s conclusion.
“Literally, the determinant as to whether I was normal or below normal on their test was one point, which was associated with a finding that I didn’t draw a clock correctly,” Dr. Smith claimed. “The reviewer said my minute hand was a little too short and docked me a point. It was purely subjective. To me, the gold standard of whether you are learned in thoracic surgery is the American Board of Thoracic Surgery’s test. The board’s test shows my cognitive ability is entirely in keeping with my practice. That contrasts with the one point off I got for drawing a clock wrong in somebody’s estimation.”
Conflict leads to legal case
In September 2020, Dr. Smith filed a lawsuit against Methodist Healthcare System of San Antonio, alleging business disparagement by Methodist for allegedly publishing false and disparaging information about Dr. Smith and tortious interference with business relations. The latter claim stems from Methodist refusing to provide documents to other hospitals about the status of Dr. Smith’s privileges at Methodist, Mr. Pullen said.
Because Methodist refused to confirm his status, the renewal process for Baptist Health System could not be completed and Dr. Smith lost his privileges at Baptist Health System facilities, according to the lawsuit.
Notably, Dr. Smith’s legal challenge also asks the court to take a stance against alleged amendments by Methodist to its Unified Medical Staff Bylaws. The hospital allegedly proposed changes that would prevent physicians from seeking legal action against the hospital for malicious peer review, according to Dr. Smith’s lawsuit.
The amendments would make the peer review process itself the “sole and exclusive remedy with respect to any action or recommendation taken at the hospital affecting medical staff appointment and/or clinical privileges,” according to an excerpt of the proposed amendments included in Dr. Smith’s lawsuit. In addition, the changes would hold practitioners liable for lost revenues if the doctor initiates “any type of legal action challenging credentialing, privileging, or other medical peer review or professional review activity,” according to the lawsuit.
Dr. Smith’s lawsuit seeks a declaration that the proposed amendments to the bylaws are “void as against public policy,” and a declaration that the proposed amendments to the bylaws cannot take away physicians’ statutory right to bring litigation against Methodist for malicious peer review.
“The proposed amendments have a tendency to and will injure the public good,” Dr. Smith argued in the lawsuit. “The proposed amendments allow Methodist to act with malice and in bad faith in conducting peer review proceedings and face no legal repercussions.”
Regardless of the final outcome of the peer review proceeding, Mr. Pullen said the harm Dr. Smith has already endured cannot be reversed.
“Even if comes out in his favor, the damage is already done,” he said. “It will not remedy the damage Dr. Smith has incurred.”
Fighting sham peer review is difficult
Battling a malicious peer review has long been an uphill battle for physicians, according to Dr. Huntoon. That’s because the Health Care Quality Improvement Act (HCQIA), a federal law passed in 1986, provides near absolute immunity to hospitals and peer reviewers in legal disputes.
The HCQIA was created by Congress to extend immunity to good-faith peer review of doctors and to increase overall participation in peer review by removing fear of litigation. However, the act has also enabled abuse of peer review by shielding bad-faith reviewers from accountability, said Dr. Huntoon.
“The Health Care Quality Improvement Act presumes that what the hospital did was warranted and reasonable and shifts the burden to the physician to prove his innocence by a preponderance of evidence,” he said. “That’s an entirely foreign concept to most people who think a person should be considered innocent until proven guilty. Here, it’s the exact opposite.”
The HCQIA has been challenged numerous times over the years and tested at the appellate level, but continues to survive and remain settled law, added Richard B. Willner, DPM, founder and director of the Center for Peer Review Justice, which assists and counsels physicians about sham peer review.
In 2011, former Rep. Joe Heck, DO, (R-Nev.) introduced a bill that would have amended the HCQIA to prohibit a professional review entity from submitting a report to the National Practitioner Data Bank (NPDB) while the doctor was still under investigation and before the doctor was afforded adequate notice and a hearing. Although the measure had 16 cosponsors and plenty of support from the physician community, it failed.
In addition to a heavy legal burden, physicians who experience malicious peer reviews also face ramifications from being reported to the NPDB. Peer review organizations are required to report certain negative actions or findings to the NPDB.
“A databank entry is a scarlet letter on your forehead,” Dr. Willner said. “The rules at a lot of institutions are not to take anyone who has been databanked, rightfully or wrongfully. And what is the evidence necessary to databank you? None. There’s no evidence needed to databank somebody.”
Despite the bleak landscape, experts say progress has been made on a case-by-case basis by physicians who have succeeded in fighting back against questionable peer reviews in recent years.
In January 2020, Indiana ob.gyn. Rebecca Denman, MD, prevailed in her defamation lawsuit against St Vincent Carmel Hospital and St Vincent Carmel Medical Group, winning $4.75 million in damages. Dr. Denman alleged administrators failed to conduct a proper peer review investigation after a false allegation by a nurse that she was under the influence while on the job.
Indianapolis attorney Kathleen A. DeLaney, who represented Dr. Denman, said hospital leaders misled Dr. Denman into believing a peer review had occurred when no formal peer review hearing or proceeding took place.
“The CMO of the medical group claimed that he performed a peer review ‘screening,’ but he never informed the other members of the peer review executive committee of the matter until after he had placed Dr. Denman on administrative leave,” Ms. DeLaney said. “He also neglected to tell the peer review executive committee that the substance abuse policy had not been followed, or that Dr. Denman had not been tested for alcohol use – due to the 12-hour delay in report.”
Dr. Denman was ultimately required to undergo an alcohol abuse evaluation, enter a treatment program, and sign a 5-year monitoring contract with the Indiana State Medical Association as a condition of her employment, according to the lawsuit. She claimed repercussions from the false allegation resulted in lost compensation, out-of-pocket expenses, emotional distress, and damage to her professional reputation.
She sued the hospital in July 2018, alleging fraud, defamation, tortious interference with an employment relationship, and negligent misrepresentation. After a 4-day trial, jurors found in her favor, awarding Dr. Denman $2 million for her defamation claims, $2 million for her claims of fraud and constructive fraud, $500,000 for her claim of tortious interference with an employment relationship, and $250,000 for her claim of negligent misrepresentation.
A hospital spokesperson said Ascension St Vincent is pursuing an appeal, and that it looks “forward to the opportunity to bring this matter before the Indiana Court of Appeals in June.”
In another case, South Dakota surgeon Linda Miller, MD, was awarded $1.1 million in 2017 after a federal jury found Huron Regional Medical Center breached her contract and violated her due process rights. Dr. Miller became the subject of a peer review at Huron Regional Medical Center when the hospital began analyzing some of her surgery outcomes.
Ken Barker, an attorney for Dr. Miller, said he feels it became evident at trial that the campaign to force Dr. Miller to either resign or lose her privileges was led by the lay board of directors of the hospital and upper-level administration at the hospital.
“They began the process by ordering an unprecedented 90-day review of her medical charts, looking for errors in the medical care she provided patients,” he said. “They could find nothing, so they did a second 90-day review, waiting for a patient’s ‘bad outcome.’ As any general surgeon will say, a ‘bad outcome’ is inevitable. And so it was. Upon that occurrence, they had a medical review committee review the patient’s chart and use it as an excuse to force her to reduce her privileges. Unbeknown to Dr. Miller, an external review had been conducted on another patient’s chart, in which the external review found her care above the standards and, in some measure, ‘exemplary.’ ”
Dr. Miller was eventually pressured to resign, according to her claim. Because of reports made to the NPDB by the medical center, including a patient complication that was allegedly falsified by the hospital, Dr. Miller said she was unable to find work as a general surgeon and went to work as a wound care doctor. At trial, jurors awarded Dr. Miller $586,617 in lost wages, $343,640 for lost future earning capacity, and $250,000 for mental anguish. (The mental anguish award was subsequently struck by a district court.)
Attorneys for Huron Regional Medical Center argued the jury improperly awarded damages and requested a new trial, which was denied by an appeals court.
In the end, the evidence came to light and the jury’s verdict spoke loudly that the hospital had taken unfair advantage of Dr. Miller, Mr. Barker said. But he emphasized that such cases often end differently.
“There are a handful of cases in which physicians like Dr. Miller have challenged the system and won,” he said. “In most cases, however, it is a ‘David vs. Goliath’ scenario where the giant prevails.”
What to do if faced with malicious peer review
An important step when doctors encounter a peer review that they believe is malicious is to consult with an experienced attorney as early as possible, Dr. Huntoon said. “Not all attorneys who set themselves out to be health law attorneys necessarily have knowledge and expertise in sham peer review. And before such a thing happens, I always encourage physicians to read their medical staff bylaws. That’s where everything is set forth, [such as] the corrective action section that tells how peer review is to take place.”
Mr. Barker added that documentation is also key in the event of a potential malicious peer review.
“When a physician senses [the] administration has targeted them, they should start documenting their conversations and actions very carefully, and if possible, recruit another ‘observer’ who can provide a third-party perspective, if necessary,” Mr. Barker said.
Dr. Huntoon recently wrote an article with advice about preparedness and defense of sham peer reviews. The guidance includes that physicians educate themselves about the tactics used by some hospitals to conduct sham peer reviews and the factors that place doctors more at risk. Factors that may raise a doctor’s danger of being targeted include being in solo practice or a small group, being new on staff, or being an older physician approaching retirement as some bad-actor hospitals may view older physicians as being less likely to fight back, said Dr. Huntoon.
Doctors should also keep detailed records and a timeline in the event of a malicious peer review and insist that an independent court reporter record all peer review hearings, even if that means the physician has to pay for the reporter him or herself, according to the guidance. An independent record is invaluable should the physician ultimately issue a future legal challenge against the hospital.
Mr. Willner encourages physicians to call the Center for Peer Review Justice hotline at (504) 621-1670 or visit the website for help with peer review and NPDB issues.
As for Dr. Smith, his days are much quieter and slower today, compared with the active practice he was accustomed to for more than half his life. He misses the fast pace, the patients, and the work that always brought him great joy.
“I hope to get back to doing surgeries eventually,” he said. “I graduated medical school in 1972. Practicing surgery has been my whole life and my career. They have taken my identity and my livelihood away from me based on false numbers and false premises. I want it back.”
A version of this article first appeared on Medscape.com.
Mohs Micrographic Surgery During the COVID-19 Pandemic: Considering the Patient Perspective
Guidelines on Skin Cancer Surgeries During the COVID-19 Pandemic
At the start of the COVID-19 pandemic, the Centers for Disease Control and Prevention issued recommendations to decrease the spread of SARS-CoV-2 and optimize the use of personal protective equipment (PPE) for frontline workers.1 In the field of dermatologic surgery, the American College of Mohs Surgery, the National Comprehensive Cancer Network, the American Society for Dermatologic Surgery, and the American Academy of Dermatology made recommendations to postpone nonessential and nonurgent procedures.2-4 The initial guidelines of the American College of Mohs Surgery advised cancellation of all elective surgeries and deferred treatment of most cases of basal cell carcinoma for as long as 3 months; low-risk squamous cell carcinoma (SCC) and melanoma in situ treatment was deferred for as long as 2 or 3 months.3 Additional recommendations were made to reserve inpatient visits for suspicious lesions and high-risk cancers, postpone other nonessential and nonurgent appointments, and utilize telemedicine whenever possible.5
These recommendations led to great uncertainty and stress for patients and providers. Although numerous important variables, such as patient risk factors, severity of disease, availability of PPE and staff, and patient-to-provider transmission were considered when creating these guidelines, the patient’s experience likely was not a contributing factor.
COVID-19 Transmission During Mohs Surgery
There have been concerns that surgeons performing Mohs micrographic surgery (MMS) might be at an increased risk for COVID-19, given their close contact with high-risk sites (ie, nose, mouth) and cautery-generated aerosols; most of the estimated transmission risk associated with MMS has been based on head and neck surgery experience and publications.6-8 Tee and colleagues9 recently published their institution’s MMS COVID-19 preventive measures, which, to their knowledge, have prevented all intraoperative transmission of SARS-CoV-2, even in disease-positive patients. Currently, evidence is lacking to support a high risk for SARS-CoV-2 transmission during MMS when proper PPE and personal hygiene measures as well as strict infection control protocols—presurgical COVID-19 testing in high-risk cases, COVID-19 screening optimization, visitor restrictions, and appropriate disinfection between patients—are in place.
The Impact of Postponing Treatment on Patients
Although studies have focused on the effects of the COVID-19 pandemic on physicians practicing MMS,10 little is known about the effects of delays in skin cancer treatment on patients. A survey conducted in the United Kingdom investigating the patient’s perspective found that patients expressed worry and concern about the possibility that their MMS would be postponed and greatly appreciated continuation of treatment during the pandemic.11
Other medical specialties have reported their patient experiences during the pandemic. In a study examining patient perception of postponed surgical treatment of pelvic floor disorders due to COVID-19, nearly half of survey respondents were unhappy with the delay in receiving care. Furthermore, patients who reported being unhappy were more likely to report feelings of isolation and anxiety because their surgery was postponed.12 In another study involving patients with lung cancer, 9.1% (N=15) of patients postponed their treatment during the COVID-19 pandemic because of pandemic-related anxiety.13
With the goal of improving care at our institution, we conducted a brief institutional review board–approved survey to evaluate how postponing MMS treatment due to the COVID-19 pandemic affected patients. All MMS patients undergoing surgery in June 2020 and July 2020 (N=99) were asked to complete our voluntary and anonymous 23-question survey in person during their procedure. We obtained 88 responses (response rate, 89%). Twenty percent of surveyed patients (n=18) reported that their MMS had been postponed; 78% of those whose MMS was postponed (n=14) indicated some level of anxiety during the waiting period. It was unclear which patients had their treatment postponed based on national guidelines and which ones elected to postpone surgery.
Tips for Health Care Providers
Patient-provider communication highlighting specific skin cancer risk and the risk vs benefit of postponing treatment might reduce anxiety and stress during the waiting period.14 A study found that COVID-19 posed a bigger threat than most noninvasive skin cancers; therefore, the authors of that study concluded that treatment for most skin cancers could be safely postponed.15 Specifically, those authors recommended prioritizing treatment for Merkel cell carcinoma, invasive SCC, and melanoma with positive margins or macroscopic residual disease. They proposed that all other skin cancers, including basal cell carcinoma, SCC in situ, and melanoma with negative margins and no macroscopic residual disease, could be safely delayed for as long as 3 months.15
For patients with multiple risk factors for COVID-19–related morbidity or mortality, delaying skin cancer treatment likely has less risk than contracting the virus.15 This information should be communicated with patients. Investigation of specific patient concerns is warranted, and case-by-case evaluation of patients’ risk factors and skin cancer risk should be considered.
Based on the current, though limited, literature, delaying medical treatment can have a negative impact on the patient experience. Furthermore, proper precautions have been shown to limit intraoperative transmission of SARS-CoV-2 during MMS, but research is lacking. Practitioners should utilize shared decision-making and evaluate a given patient’s risk factors and concerns when deciding whether to postpone treatment. We encourage other institutions to evaluate the effects that delaying MMS has had on their patients, as further studies would improve understanding of patients’ experiences during a pandemic and potentially influence future dermatology guidelines.
- Center for Disease Control and Prevention. COVID-19. Accessed April 20, 2021. https://www.cdc.gov/coronavirus/2019-ncov/index.html
- American College of Mohs Surgery. Mohs surgery ambulatory protocol during COVID pandemic (version 6-3-20). June 4, 2020. Accessed April 20, 2021. http://staging.mohscollege.org/UserFiles/AM20/Member%20Alert/MohsSurgeryAmbulatoryProtocolDuringCOVIDPandemicFinal.pdf
- COVID-19 resources. National Comprehensive Cancer Network website. Accessed April 20, 2021. https://www.nccn.org/covid-19
- Narla S, Alam M, Ozog DM, et al. American Society of Dermatologic Surgery Association (ASDSA) and American Society for Laser Medicine & Surgery (ASLMS) guidance for cosmetic dermatology practices during COVID-19. Updated January 11, 2021. Accessed April 10, 2021. https://www.asds.net/Portals/0/PDF/asdsa/asdsa-aslms-cosmetic-reopening-guidance.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic.J Am Acad Dermatol. 2020;83:295-296. doi:10.1016/j.jaad.2020.05.002
- Yuan JT, Jiang SIB. Urgent safety considerations for dermatologic surgeons in the COVID-19 pandemic. Dermatol Online J. 2020;26:1. Accessed April 20, 2021. http://escholarship.org/uc/item/2qr3w771
- Otolaryngologists may contract COVID-19 during surgery. ENTtoday. March 20, 2020. Accessed April 20, 2021. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/
- Howard BE. High-risk aerosol-generating procedures in COVID-19: respiratory protective equipment considerations. Otolaryngol Head Neck Surg. 2020;163:98-103. doi:10.1177/0194599820927335
- Tee MW, Stewart C, Aliessa S, et al. Dermatological surgery during the COVID-19 pandemic: experience of a large academic center. J Am Acad Dermatol. 2021;84:1094-1096. doi:10.1016/j.jaad.2020.12.003
- Hooper J, Feng H. The impact of COVID-19 on micrographic surgery and dermatologic oncology fellows. Dermatol Surg. 2020;46:1762-1763. doi:10.1097/DSS.0000000000002766
- Nicholson P, Ali FR, Patalay R, et al. Patient perceptions of Mohs micrographic surgery during the COVID-19 pandemic and lessons for the next outbreak. Clin Exp Dermatol. 2021;46:179-180. doi:10.1111/ced.14423
- Mou T, Brown O, Gillingham A, et al. Patients’ perceptions on surgical care suspension for pelvic floor disorders during the COVID-19 pandemic. Female Pelvic Med Reconstr Surg. 2020;26:477-482. doi:10.1097/SPV.0000000000000918
- Fujita K, Ito T, Saito Z, et al. Impact of COVID-19 pandemic on lung cancer treatment scheduling. Thorac Cancer. 2020;11:2983-2986. doi:10.1111/1759-7714.13615
- Nikumb VB, Banerjee A, Kaur G, et al. Impact of doctor-patient communication on preoperative anxiety: study at industrial township, Pimpri, Pune. Ind Psychiatry J. 2009;18:19-21. doi:10.4103/0972-6748.57852
- Baumann BC, MacArthur KM, Brewer JD, et al. Management of primary skin cancer during a pandemic: multidisciplinary recommendations. Cancer. 2020;126:3900-3906. doi:10.1002/cncr.32969
Guidelines on Skin Cancer Surgeries During the COVID-19 Pandemic
At the start of the COVID-19 pandemic, the Centers for Disease Control and Prevention issued recommendations to decrease the spread of SARS-CoV-2 and optimize the use of personal protective equipment (PPE) for frontline workers.1 In the field of dermatologic surgery, the American College of Mohs Surgery, the National Comprehensive Cancer Network, the American Society for Dermatologic Surgery, and the American Academy of Dermatology made recommendations to postpone nonessential and nonurgent procedures.2-4 The initial guidelines of the American College of Mohs Surgery advised cancellation of all elective surgeries and deferred treatment of most cases of basal cell carcinoma for as long as 3 months; low-risk squamous cell carcinoma (SCC) and melanoma in situ treatment was deferred for as long as 2 or 3 months.3 Additional recommendations were made to reserve inpatient visits for suspicious lesions and high-risk cancers, postpone other nonessential and nonurgent appointments, and utilize telemedicine whenever possible.5
These recommendations led to great uncertainty and stress for patients and providers. Although numerous important variables, such as patient risk factors, severity of disease, availability of PPE and staff, and patient-to-provider transmission were considered when creating these guidelines, the patient’s experience likely was not a contributing factor.
COVID-19 Transmission During Mohs Surgery
There have been concerns that surgeons performing Mohs micrographic surgery (MMS) might be at an increased risk for COVID-19, given their close contact with high-risk sites (ie, nose, mouth) and cautery-generated aerosols; most of the estimated transmission risk associated with MMS has been based on head and neck surgery experience and publications.6-8 Tee and colleagues9 recently published their institution’s MMS COVID-19 preventive measures, which, to their knowledge, have prevented all intraoperative transmission of SARS-CoV-2, even in disease-positive patients. Currently, evidence is lacking to support a high risk for SARS-CoV-2 transmission during MMS when proper PPE and personal hygiene measures as well as strict infection control protocols—presurgical COVID-19 testing in high-risk cases, COVID-19 screening optimization, visitor restrictions, and appropriate disinfection between patients—are in place.
The Impact of Postponing Treatment on Patients
Although studies have focused on the effects of the COVID-19 pandemic on physicians practicing MMS,10 little is known about the effects of delays in skin cancer treatment on patients. A survey conducted in the United Kingdom investigating the patient’s perspective found that patients expressed worry and concern about the possibility that their MMS would be postponed and greatly appreciated continuation of treatment during the pandemic.11
Other medical specialties have reported their patient experiences during the pandemic. In a study examining patient perception of postponed surgical treatment of pelvic floor disorders due to COVID-19, nearly half of survey respondents were unhappy with the delay in receiving care. Furthermore, patients who reported being unhappy were more likely to report feelings of isolation and anxiety because their surgery was postponed.12 In another study involving patients with lung cancer, 9.1% (N=15) of patients postponed their treatment during the COVID-19 pandemic because of pandemic-related anxiety.13
With the goal of improving care at our institution, we conducted a brief institutional review board–approved survey to evaluate how postponing MMS treatment due to the COVID-19 pandemic affected patients. All MMS patients undergoing surgery in June 2020 and July 2020 (N=99) were asked to complete our voluntary and anonymous 23-question survey in person during their procedure. We obtained 88 responses (response rate, 89%). Twenty percent of surveyed patients (n=18) reported that their MMS had been postponed; 78% of those whose MMS was postponed (n=14) indicated some level of anxiety during the waiting period. It was unclear which patients had their treatment postponed based on national guidelines and which ones elected to postpone surgery.
Tips for Health Care Providers
Patient-provider communication highlighting specific skin cancer risk and the risk vs benefit of postponing treatment might reduce anxiety and stress during the waiting period.14 A study found that COVID-19 posed a bigger threat than most noninvasive skin cancers; therefore, the authors of that study concluded that treatment for most skin cancers could be safely postponed.15 Specifically, those authors recommended prioritizing treatment for Merkel cell carcinoma, invasive SCC, and melanoma with positive margins or macroscopic residual disease. They proposed that all other skin cancers, including basal cell carcinoma, SCC in situ, and melanoma with negative margins and no macroscopic residual disease, could be safely delayed for as long as 3 months.15
For patients with multiple risk factors for COVID-19–related morbidity or mortality, delaying skin cancer treatment likely has less risk than contracting the virus.15 This information should be communicated with patients. Investigation of specific patient concerns is warranted, and case-by-case evaluation of patients’ risk factors and skin cancer risk should be considered.
Based on the current, though limited, literature, delaying medical treatment can have a negative impact on the patient experience. Furthermore, proper precautions have been shown to limit intraoperative transmission of SARS-CoV-2 during MMS, but research is lacking. Practitioners should utilize shared decision-making and evaluate a given patient’s risk factors and concerns when deciding whether to postpone treatment. We encourage other institutions to evaluate the effects that delaying MMS has had on their patients, as further studies would improve understanding of patients’ experiences during a pandemic and potentially influence future dermatology guidelines.
Guidelines on Skin Cancer Surgeries During the COVID-19 Pandemic
At the start of the COVID-19 pandemic, the Centers for Disease Control and Prevention issued recommendations to decrease the spread of SARS-CoV-2 and optimize the use of personal protective equipment (PPE) for frontline workers.1 In the field of dermatologic surgery, the American College of Mohs Surgery, the National Comprehensive Cancer Network, the American Society for Dermatologic Surgery, and the American Academy of Dermatology made recommendations to postpone nonessential and nonurgent procedures.2-4 The initial guidelines of the American College of Mohs Surgery advised cancellation of all elective surgeries and deferred treatment of most cases of basal cell carcinoma for as long as 3 months; low-risk squamous cell carcinoma (SCC) and melanoma in situ treatment was deferred for as long as 2 or 3 months.3 Additional recommendations were made to reserve inpatient visits for suspicious lesions and high-risk cancers, postpone other nonessential and nonurgent appointments, and utilize telemedicine whenever possible.5
These recommendations led to great uncertainty and stress for patients and providers. Although numerous important variables, such as patient risk factors, severity of disease, availability of PPE and staff, and patient-to-provider transmission were considered when creating these guidelines, the patient’s experience likely was not a contributing factor.
COVID-19 Transmission During Mohs Surgery
There have been concerns that surgeons performing Mohs micrographic surgery (MMS) might be at an increased risk for COVID-19, given their close contact with high-risk sites (ie, nose, mouth) and cautery-generated aerosols; most of the estimated transmission risk associated with MMS has been based on head and neck surgery experience and publications.6-8 Tee and colleagues9 recently published their institution’s MMS COVID-19 preventive measures, which, to their knowledge, have prevented all intraoperative transmission of SARS-CoV-2, even in disease-positive patients. Currently, evidence is lacking to support a high risk for SARS-CoV-2 transmission during MMS when proper PPE and personal hygiene measures as well as strict infection control protocols—presurgical COVID-19 testing in high-risk cases, COVID-19 screening optimization, visitor restrictions, and appropriate disinfection between patients—are in place.
The Impact of Postponing Treatment on Patients
Although studies have focused on the effects of the COVID-19 pandemic on physicians practicing MMS,10 little is known about the effects of delays in skin cancer treatment on patients. A survey conducted in the United Kingdom investigating the patient’s perspective found that patients expressed worry and concern about the possibility that their MMS would be postponed and greatly appreciated continuation of treatment during the pandemic.11
Other medical specialties have reported their patient experiences during the pandemic. In a study examining patient perception of postponed surgical treatment of pelvic floor disorders due to COVID-19, nearly half of survey respondents were unhappy with the delay in receiving care. Furthermore, patients who reported being unhappy were more likely to report feelings of isolation and anxiety because their surgery was postponed.12 In another study involving patients with lung cancer, 9.1% (N=15) of patients postponed their treatment during the COVID-19 pandemic because of pandemic-related anxiety.13
With the goal of improving care at our institution, we conducted a brief institutional review board–approved survey to evaluate how postponing MMS treatment due to the COVID-19 pandemic affected patients. All MMS patients undergoing surgery in June 2020 and July 2020 (N=99) were asked to complete our voluntary and anonymous 23-question survey in person during their procedure. We obtained 88 responses (response rate, 89%). Twenty percent of surveyed patients (n=18) reported that their MMS had been postponed; 78% of those whose MMS was postponed (n=14) indicated some level of anxiety during the waiting period. It was unclear which patients had their treatment postponed based on national guidelines and which ones elected to postpone surgery.
Tips for Health Care Providers
Patient-provider communication highlighting specific skin cancer risk and the risk vs benefit of postponing treatment might reduce anxiety and stress during the waiting period.14 A study found that COVID-19 posed a bigger threat than most noninvasive skin cancers; therefore, the authors of that study concluded that treatment for most skin cancers could be safely postponed.15 Specifically, those authors recommended prioritizing treatment for Merkel cell carcinoma, invasive SCC, and melanoma with positive margins or macroscopic residual disease. They proposed that all other skin cancers, including basal cell carcinoma, SCC in situ, and melanoma with negative margins and no macroscopic residual disease, could be safely delayed for as long as 3 months.15
For patients with multiple risk factors for COVID-19–related morbidity or mortality, delaying skin cancer treatment likely has less risk than contracting the virus.15 This information should be communicated with patients. Investigation of specific patient concerns is warranted, and case-by-case evaluation of patients’ risk factors and skin cancer risk should be considered.
Based on the current, though limited, literature, delaying medical treatment can have a negative impact on the patient experience. Furthermore, proper precautions have been shown to limit intraoperative transmission of SARS-CoV-2 during MMS, but research is lacking. Practitioners should utilize shared decision-making and evaluate a given patient’s risk factors and concerns when deciding whether to postpone treatment. We encourage other institutions to evaluate the effects that delaying MMS has had on their patients, as further studies would improve understanding of patients’ experiences during a pandemic and potentially influence future dermatology guidelines.
- Center for Disease Control and Prevention. COVID-19. Accessed April 20, 2021. https://www.cdc.gov/coronavirus/2019-ncov/index.html
- American College of Mohs Surgery. Mohs surgery ambulatory protocol during COVID pandemic (version 6-3-20). June 4, 2020. Accessed April 20, 2021. http://staging.mohscollege.org/UserFiles/AM20/Member%20Alert/MohsSurgeryAmbulatoryProtocolDuringCOVIDPandemicFinal.pdf
- COVID-19 resources. National Comprehensive Cancer Network website. Accessed April 20, 2021. https://www.nccn.org/covid-19
- Narla S, Alam M, Ozog DM, et al. American Society of Dermatologic Surgery Association (ASDSA) and American Society for Laser Medicine & Surgery (ASLMS) guidance for cosmetic dermatology practices during COVID-19. Updated January 11, 2021. Accessed April 10, 2021. https://www.asds.net/Portals/0/PDF/asdsa/asdsa-aslms-cosmetic-reopening-guidance.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic.J Am Acad Dermatol. 2020;83:295-296. doi:10.1016/j.jaad.2020.05.002
- Yuan JT, Jiang SIB. Urgent safety considerations for dermatologic surgeons in the COVID-19 pandemic. Dermatol Online J. 2020;26:1. Accessed April 20, 2021. http://escholarship.org/uc/item/2qr3w771
- Otolaryngologists may contract COVID-19 during surgery. ENTtoday. March 20, 2020. Accessed April 20, 2021. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/
- Howard BE. High-risk aerosol-generating procedures in COVID-19: respiratory protective equipment considerations. Otolaryngol Head Neck Surg. 2020;163:98-103. doi:10.1177/0194599820927335
- Tee MW, Stewart C, Aliessa S, et al. Dermatological surgery during the COVID-19 pandemic: experience of a large academic center. J Am Acad Dermatol. 2021;84:1094-1096. doi:10.1016/j.jaad.2020.12.003
- Hooper J, Feng H. The impact of COVID-19 on micrographic surgery and dermatologic oncology fellows. Dermatol Surg. 2020;46:1762-1763. doi:10.1097/DSS.0000000000002766
- Nicholson P, Ali FR, Patalay R, et al. Patient perceptions of Mohs micrographic surgery during the COVID-19 pandemic and lessons for the next outbreak. Clin Exp Dermatol. 2021;46:179-180. doi:10.1111/ced.14423
- Mou T, Brown O, Gillingham A, et al. Patients’ perceptions on surgical care suspension for pelvic floor disorders during the COVID-19 pandemic. Female Pelvic Med Reconstr Surg. 2020;26:477-482. doi:10.1097/SPV.0000000000000918
- Fujita K, Ito T, Saito Z, et al. Impact of COVID-19 pandemic on lung cancer treatment scheduling. Thorac Cancer. 2020;11:2983-2986. doi:10.1111/1759-7714.13615
- Nikumb VB, Banerjee A, Kaur G, et al. Impact of doctor-patient communication on preoperative anxiety: study at industrial township, Pimpri, Pune. Ind Psychiatry J. 2009;18:19-21. doi:10.4103/0972-6748.57852
- Baumann BC, MacArthur KM, Brewer JD, et al. Management of primary skin cancer during a pandemic: multidisciplinary recommendations. Cancer. 2020;126:3900-3906. doi:10.1002/cncr.32969
- Center for Disease Control and Prevention. COVID-19. Accessed April 20, 2021. https://www.cdc.gov/coronavirus/2019-ncov/index.html
- American College of Mohs Surgery. Mohs surgery ambulatory protocol during COVID pandemic (version 6-3-20). June 4, 2020. Accessed April 20, 2021. http://staging.mohscollege.org/UserFiles/AM20/Member%20Alert/MohsSurgeryAmbulatoryProtocolDuringCOVIDPandemicFinal.pdf
- COVID-19 resources. National Comprehensive Cancer Network website. Accessed April 20, 2021. https://www.nccn.org/covid-19
- Narla S, Alam M, Ozog DM, et al. American Society of Dermatologic Surgery Association (ASDSA) and American Society for Laser Medicine & Surgery (ASLMS) guidance for cosmetic dermatology practices during COVID-19. Updated January 11, 2021. Accessed April 10, 2021. https://www.asds.net/Portals/0/PDF/asdsa/asdsa-aslms-cosmetic-reopening-guidance.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic.J Am Acad Dermatol. 2020;83:295-296. doi:10.1016/j.jaad.2020.05.002
- Yuan JT, Jiang SIB. Urgent safety considerations for dermatologic surgeons in the COVID-19 pandemic. Dermatol Online J. 2020;26:1. Accessed April 20, 2021. http://escholarship.org/uc/item/2qr3w771
- Otolaryngologists may contract COVID-19 during surgery. ENTtoday. March 20, 2020. Accessed April 20, 2021. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/
- Howard BE. High-risk aerosol-generating procedures in COVID-19: respiratory protective equipment considerations. Otolaryngol Head Neck Surg. 2020;163:98-103. doi:10.1177/0194599820927335
- Tee MW, Stewart C, Aliessa S, et al. Dermatological surgery during the COVID-19 pandemic: experience of a large academic center. J Am Acad Dermatol. 2021;84:1094-1096. doi:10.1016/j.jaad.2020.12.003
- Hooper J, Feng H. The impact of COVID-19 on micrographic surgery and dermatologic oncology fellows. Dermatol Surg. 2020;46:1762-1763. doi:10.1097/DSS.0000000000002766
- Nicholson P, Ali FR, Patalay R, et al. Patient perceptions of Mohs micrographic surgery during the COVID-19 pandemic and lessons for the next outbreak. Clin Exp Dermatol. 2021;46:179-180. doi:10.1111/ced.14423
- Mou T, Brown O, Gillingham A, et al. Patients’ perceptions on surgical care suspension for pelvic floor disorders during the COVID-19 pandemic. Female Pelvic Med Reconstr Surg. 2020;26:477-482. doi:10.1097/SPV.0000000000000918
- Fujita K, Ito T, Saito Z, et al. Impact of COVID-19 pandemic on lung cancer treatment scheduling. Thorac Cancer. 2020;11:2983-2986. doi:10.1111/1759-7714.13615
- Nikumb VB, Banerjee A, Kaur G, et al. Impact of doctor-patient communication on preoperative anxiety: study at industrial township, Pimpri, Pune. Ind Psychiatry J. 2009;18:19-21. doi:10.4103/0972-6748.57852
- Baumann BC, MacArthur KM, Brewer JD, et al. Management of primary skin cancer during a pandemic: multidisciplinary recommendations. Cancer. 2020;126:3900-3906. doi:10.1002/cncr.32969
Practice Points
- There is little evidence that supports a high risk for SARS-CoV-2 transmission during Mohs micrographic surgery when proper personal protective equipment and strict infection control protocols are in place.
- The effects of treatment delays due to COVID-19 on the patient experience have not been well studied, but the limited literature suggests a negative association.
- Shared decision-making and evaluation of individual patient risk factors and concerns should be considered when deciding whether to postpone skin cancer treatment.
Possible obesity effect detected in cancer death rates
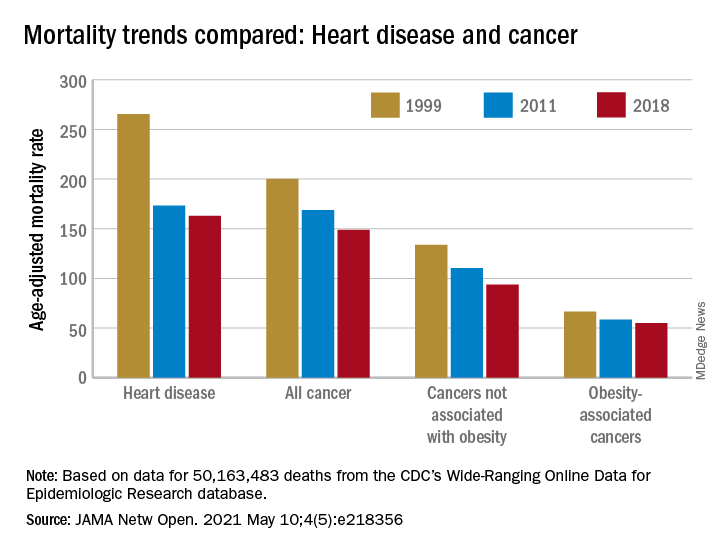
“By integrating 20 years of cancer mortality data, we demonstrated that trends in obesity-associated cancer mortality showed signs of recent deceleration, consistent with recent findings for heart disease mortality,” Christy L. Avery, PhD, and associates wrote in JAMA Network Open.
Improvements in mortality related to heart disease slowed after 2011, a phenomenon that has been associated with rising obesity rates. The age-adjusted mortality rate (AAMR) declined at an average of 3.8 deaths per 100,000 persons from 1999 to 2011 but only 0.7 deaths per 100,000 from 2011 to 2018, based on data from the Centers for Disease Control and Prevention’s Wide-Ranging Online Data for Epidemiologic Research (WONDER).
To understand trends in cancer mortality and their possible connection with obesity, data for 1999-2018 from the WONDER database were divided into obesity-associated and non–obesity-associated categories and compared with heart disease mortality, they explained. The database included more than 50 million deaths that matched inclusion criteria.
The analysis showed there was difference between obesity-associated and non–obesity-associated cancers that was obscured when all cancer deaths were considered together. The average annual change in AAMR for obesity-associated cancers slowed from –1.19 deaths per 100,000 in 1999-2011 to –0.83 in 2011-2018, Dr. Avery and associates reported.
For non–obesity-associated cancers, the annual change in AAMR increased from –1.62 per 100,000 for 1999-2011 to –2.29 for 2011-2018, following the trend for all cancers: –1.48 per 100,000 during 1999-2011 and –1.77 in 2011-2018, they said.
“The largest mortality decreases were observed for melanoma of the skin and lung cancer, two cancers not associated with obesity. For obesity-associated cancers, stable or increasing mortality rates have been observed for liver and pancreatic cancer among both men and women as well as for uterine cancer among women,” the investigators wrote.
Demographically, however, the slowing improvement in mortality for obesity-associated cancers did not follow the trend for heart disease. The deceleration for cancer was more pronounced for women and for non-Hispanic Whites and not seen at all in non-Hispanic Asian/Pacific Islander individuals. “For heart disease, evidence of a deceleration was consistent across sex, race, and ethnicity,” they said.
There are “longstanding disparities in obesity” among various populations in the United States, and the recent trend of obesity occurring earlier in life may be having an effect. “Whether the findings of decelerating mortality rates potentially signal a changing profile of cancer and heart disease mortality as the consequences of the obesity epidemic are realized remains to be seen,” they concluded.
The investigators reported receiving grants from the National Institutes of Health during the conduct of the study, but no other disclosures were reported.

“By integrating 20 years of cancer mortality data, we demonstrated that trends in obesity-associated cancer mortality showed signs of recent deceleration, consistent with recent findings for heart disease mortality,” Christy L. Avery, PhD, and associates wrote in JAMA Network Open.
Improvements in mortality related to heart disease slowed after 2011, a phenomenon that has been associated with rising obesity rates. The age-adjusted mortality rate (AAMR) declined at an average of 3.8 deaths per 100,000 persons from 1999 to 2011 but only 0.7 deaths per 100,000 from 2011 to 2018, based on data from the Centers for Disease Control and Prevention’s Wide-Ranging Online Data for Epidemiologic Research (WONDER).
To understand trends in cancer mortality and their possible connection with obesity, data for 1999-2018 from the WONDER database were divided into obesity-associated and non–obesity-associated categories and compared with heart disease mortality, they explained. The database included more than 50 million deaths that matched inclusion criteria.
The analysis showed there was difference between obesity-associated and non–obesity-associated cancers that was obscured when all cancer deaths were considered together. The average annual change in AAMR for obesity-associated cancers slowed from –1.19 deaths per 100,000 in 1999-2011 to –0.83 in 2011-2018, Dr. Avery and associates reported.
For non–obesity-associated cancers, the annual change in AAMR increased from –1.62 per 100,000 for 1999-2011 to –2.29 for 2011-2018, following the trend for all cancers: –1.48 per 100,000 during 1999-2011 and –1.77 in 2011-2018, they said.
“The largest mortality decreases were observed for melanoma of the skin and lung cancer, two cancers not associated with obesity. For obesity-associated cancers, stable or increasing mortality rates have been observed for liver and pancreatic cancer among both men and women as well as for uterine cancer among women,” the investigators wrote.
Demographically, however, the slowing improvement in mortality for obesity-associated cancers did not follow the trend for heart disease. The deceleration for cancer was more pronounced for women and for non-Hispanic Whites and not seen at all in non-Hispanic Asian/Pacific Islander individuals. “For heart disease, evidence of a deceleration was consistent across sex, race, and ethnicity,” they said.
There are “longstanding disparities in obesity” among various populations in the United States, and the recent trend of obesity occurring earlier in life may be having an effect. “Whether the findings of decelerating mortality rates potentially signal a changing profile of cancer and heart disease mortality as the consequences of the obesity epidemic are realized remains to be seen,” they concluded.
The investigators reported receiving grants from the National Institutes of Health during the conduct of the study, but no other disclosures were reported.

“By integrating 20 years of cancer mortality data, we demonstrated that trends in obesity-associated cancer mortality showed signs of recent deceleration, consistent with recent findings for heart disease mortality,” Christy L. Avery, PhD, and associates wrote in JAMA Network Open.
Improvements in mortality related to heart disease slowed after 2011, a phenomenon that has been associated with rising obesity rates. The age-adjusted mortality rate (AAMR) declined at an average of 3.8 deaths per 100,000 persons from 1999 to 2011 but only 0.7 deaths per 100,000 from 2011 to 2018, based on data from the Centers for Disease Control and Prevention’s Wide-Ranging Online Data for Epidemiologic Research (WONDER).
To understand trends in cancer mortality and their possible connection with obesity, data for 1999-2018 from the WONDER database were divided into obesity-associated and non–obesity-associated categories and compared with heart disease mortality, they explained. The database included more than 50 million deaths that matched inclusion criteria.
The analysis showed there was difference between obesity-associated and non–obesity-associated cancers that was obscured when all cancer deaths were considered together. The average annual change in AAMR for obesity-associated cancers slowed from –1.19 deaths per 100,000 in 1999-2011 to –0.83 in 2011-2018, Dr. Avery and associates reported.
For non–obesity-associated cancers, the annual change in AAMR increased from –1.62 per 100,000 for 1999-2011 to –2.29 for 2011-2018, following the trend for all cancers: –1.48 per 100,000 during 1999-2011 and –1.77 in 2011-2018, they said.
“The largest mortality decreases were observed for melanoma of the skin and lung cancer, two cancers not associated with obesity. For obesity-associated cancers, stable or increasing mortality rates have been observed for liver and pancreatic cancer among both men and women as well as for uterine cancer among women,” the investigators wrote.
Demographically, however, the slowing improvement in mortality for obesity-associated cancers did not follow the trend for heart disease. The deceleration for cancer was more pronounced for women and for non-Hispanic Whites and not seen at all in non-Hispanic Asian/Pacific Islander individuals. “For heart disease, evidence of a deceleration was consistent across sex, race, and ethnicity,” they said.
There are “longstanding disparities in obesity” among various populations in the United States, and the recent trend of obesity occurring earlier in life may be having an effect. “Whether the findings of decelerating mortality rates potentially signal a changing profile of cancer and heart disease mortality as the consequences of the obesity epidemic are realized remains to be seen,” they concluded.
The investigators reported receiving grants from the National Institutes of Health during the conduct of the study, but no other disclosures were reported.
FROM JAMA NETWORK OPEN
Novel hedgehog inhibitor strategies improve BCC outcomes
MD, a Mohs surgeon and chair of the department of dermatology at the Cleveland Clinic.
She and her colleagues have noticed an accelerated and durable response to hedgehog inhibitors after debulking and are studying cell signaling before and after debulking to better understand the issue.
Dr. Vidimos shared a remarkable case to illustrate the point during a clinical pearls talk at the annual meeting of the American College of Mohs Surgery.
An 82-year-old woman presented with a crusted, hemorrhagic, nodular basal cell carcinoma (BCC) that had overgrown over nearly her entire nose and left lower eyelid. A recurrence of a previous BCC, the tumor had been growing for a decade and had invaded her nasal bones but not the periorbital tissue.
An outside surgeon suggested a full rhinectomy and removal of the lower eyelid, but the woman refused.
Dr. Vidimos decided to treat her with vismodegib, but prior to doing so, she debulked the tumor to help with the pain and bleeding. She did not curette the portion of tumor extending through the ala into the nasal vestibule. “I let the vismodegib take care of that,” she said.
After 9 months, the tumor was virtually gone, with no recurrence after 3 years. Surgical debulking prior to hedgehog inhibition “reduces the tumor burden and may increase the efficacy and shorten the course of therapy,” Dr. Vidimos said.
The hedgehog inhibitors vismodegib (Erivedge) and sonidegib Odomzo are both approved for treating locally advanced BCC, with a complete response of 31% of locally advanced disease with vismodegib, according to one report.
But monotherapy is limited by intolerable side effects, most commonly muscle spasms, alopecia, and dysgeusia. To minimize the impact, Dr. Vidimos generally puts patients on treatment with Monday through Friday dosing and gives them the weekends off, a schedule she and her colleagues have reported works as well as daily dosing.
Still, many patients discontinue the drugs because of the side effects. Hedgehog inhibitors are also expensive and responses aren’t always durable. To increase efficacy and shorten the course of therapy, “we need alternative treatment strategies,” Dr. Vidimos said.
Up-front tumor debulking is one such strategy. Altered cell signaling pathways associated with tissue remodeling might improve response, and debulking may reduce the genetic heterogeneity of tumor cells, rendering remaining cells less resistant to hedgehog inhibition, she explained.
“It is exciting to see how tumor debulking may reduce tumor burden and heterogeneity, and thus lead to a durable response in extensive tumors,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington, who heard the presentation. “More investigation is needed to reproduce these results, but this approach may lead to improved outcomes with targeted therapies,” he said in an interview.
Combination therapy with other agents is another option, and there also seems to be a synergistic effect with radiation, with hedgehog inhibitors increasing cellular response to radiation therapy, Dr. Vidimos said.
Hedgehog inhibitors can also be used to shrink tumors before surgery. One small series found a 27% decrease in the area of the tumor after 3 to 6 months of preoperative vismodegib.
Dr. Vidimos shared another case to illustrate the point.
A 64-year-old woman fainted and presented to the ED with a hemoglobin of 3.2 mg/dL because of chronic blood loss from an ulcerated BCC on her upper back. The lesion measured 25 cm by 9 cm, and was 3.5 cm deep with no bone involvement. The woman was addicted to opioids by the time she presented.
She was started on vismodegib; the ulcer shrunk considerably after 6 months, and the woman underwent a resection. Only one small focus of BCC was found across 78 specimens submitted to Dr. Vidimos for Mohs reading.
Resection was followed by a muscle flap repair and radiation. At 5 and a half years, there is no evidence of disease; the only sign that the lesion had been there was a scar running along the woman’s upper spine.
The approach “was very successful for a very aggressive and worrisome tumor,” Dr. Vidimos said.
Dr. Vidimos did not have any relevant disclosures. Dr. Patel had no relevant disclosures.
MD, a Mohs surgeon and chair of the department of dermatology at the Cleveland Clinic.
She and her colleagues have noticed an accelerated and durable response to hedgehog inhibitors after debulking and are studying cell signaling before and after debulking to better understand the issue.
Dr. Vidimos shared a remarkable case to illustrate the point during a clinical pearls talk at the annual meeting of the American College of Mohs Surgery.
An 82-year-old woman presented with a crusted, hemorrhagic, nodular basal cell carcinoma (BCC) that had overgrown over nearly her entire nose and left lower eyelid. A recurrence of a previous BCC, the tumor had been growing for a decade and had invaded her nasal bones but not the periorbital tissue.
An outside surgeon suggested a full rhinectomy and removal of the lower eyelid, but the woman refused.
Dr. Vidimos decided to treat her with vismodegib, but prior to doing so, she debulked the tumor to help with the pain and bleeding. She did not curette the portion of tumor extending through the ala into the nasal vestibule. “I let the vismodegib take care of that,” she said.
After 9 months, the tumor was virtually gone, with no recurrence after 3 years. Surgical debulking prior to hedgehog inhibition “reduces the tumor burden and may increase the efficacy and shorten the course of therapy,” Dr. Vidimos said.
The hedgehog inhibitors vismodegib (Erivedge) and sonidegib Odomzo are both approved for treating locally advanced BCC, with a complete response of 31% of locally advanced disease with vismodegib, according to one report.
But monotherapy is limited by intolerable side effects, most commonly muscle spasms, alopecia, and dysgeusia. To minimize the impact, Dr. Vidimos generally puts patients on treatment with Monday through Friday dosing and gives them the weekends off, a schedule she and her colleagues have reported works as well as daily dosing.
Still, many patients discontinue the drugs because of the side effects. Hedgehog inhibitors are also expensive and responses aren’t always durable. To increase efficacy and shorten the course of therapy, “we need alternative treatment strategies,” Dr. Vidimos said.
Up-front tumor debulking is one such strategy. Altered cell signaling pathways associated with tissue remodeling might improve response, and debulking may reduce the genetic heterogeneity of tumor cells, rendering remaining cells less resistant to hedgehog inhibition, she explained.
“It is exciting to see how tumor debulking may reduce tumor burden and heterogeneity, and thus lead to a durable response in extensive tumors,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington, who heard the presentation. “More investigation is needed to reproduce these results, but this approach may lead to improved outcomes with targeted therapies,” he said in an interview.
Combination therapy with other agents is another option, and there also seems to be a synergistic effect with radiation, with hedgehog inhibitors increasing cellular response to radiation therapy, Dr. Vidimos said.
Hedgehog inhibitors can also be used to shrink tumors before surgery. One small series found a 27% decrease in the area of the tumor after 3 to 6 months of preoperative vismodegib.
Dr. Vidimos shared another case to illustrate the point.
A 64-year-old woman fainted and presented to the ED with a hemoglobin of 3.2 mg/dL because of chronic blood loss from an ulcerated BCC on her upper back. The lesion measured 25 cm by 9 cm, and was 3.5 cm deep with no bone involvement. The woman was addicted to opioids by the time she presented.
She was started on vismodegib; the ulcer shrunk considerably after 6 months, and the woman underwent a resection. Only one small focus of BCC was found across 78 specimens submitted to Dr. Vidimos for Mohs reading.
Resection was followed by a muscle flap repair and radiation. At 5 and a half years, there is no evidence of disease; the only sign that the lesion had been there was a scar running along the woman’s upper spine.
The approach “was very successful for a very aggressive and worrisome tumor,” Dr. Vidimos said.
Dr. Vidimos did not have any relevant disclosures. Dr. Patel had no relevant disclosures.
MD, a Mohs surgeon and chair of the department of dermatology at the Cleveland Clinic.
She and her colleagues have noticed an accelerated and durable response to hedgehog inhibitors after debulking and are studying cell signaling before and after debulking to better understand the issue.
Dr. Vidimos shared a remarkable case to illustrate the point during a clinical pearls talk at the annual meeting of the American College of Mohs Surgery.
An 82-year-old woman presented with a crusted, hemorrhagic, nodular basal cell carcinoma (BCC) that had overgrown over nearly her entire nose and left lower eyelid. A recurrence of a previous BCC, the tumor had been growing for a decade and had invaded her nasal bones but not the periorbital tissue.
An outside surgeon suggested a full rhinectomy and removal of the lower eyelid, but the woman refused.
Dr. Vidimos decided to treat her with vismodegib, but prior to doing so, she debulked the tumor to help with the pain and bleeding. She did not curette the portion of tumor extending through the ala into the nasal vestibule. “I let the vismodegib take care of that,” she said.
After 9 months, the tumor was virtually gone, with no recurrence after 3 years. Surgical debulking prior to hedgehog inhibition “reduces the tumor burden and may increase the efficacy and shorten the course of therapy,” Dr. Vidimos said.
The hedgehog inhibitors vismodegib (Erivedge) and sonidegib Odomzo are both approved for treating locally advanced BCC, with a complete response of 31% of locally advanced disease with vismodegib, according to one report.
But monotherapy is limited by intolerable side effects, most commonly muscle spasms, alopecia, and dysgeusia. To minimize the impact, Dr. Vidimos generally puts patients on treatment with Monday through Friday dosing and gives them the weekends off, a schedule she and her colleagues have reported works as well as daily dosing.
Still, many patients discontinue the drugs because of the side effects. Hedgehog inhibitors are also expensive and responses aren’t always durable. To increase efficacy and shorten the course of therapy, “we need alternative treatment strategies,” Dr. Vidimos said.
Up-front tumor debulking is one such strategy. Altered cell signaling pathways associated with tissue remodeling might improve response, and debulking may reduce the genetic heterogeneity of tumor cells, rendering remaining cells less resistant to hedgehog inhibition, she explained.
“It is exciting to see how tumor debulking may reduce tumor burden and heterogeneity, and thus lead to a durable response in extensive tumors,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington, who heard the presentation. “More investigation is needed to reproduce these results, but this approach may lead to improved outcomes with targeted therapies,” he said in an interview.
Combination therapy with other agents is another option, and there also seems to be a synergistic effect with radiation, with hedgehog inhibitors increasing cellular response to radiation therapy, Dr. Vidimos said.
Hedgehog inhibitors can also be used to shrink tumors before surgery. One small series found a 27% decrease in the area of the tumor after 3 to 6 months of preoperative vismodegib.
Dr. Vidimos shared another case to illustrate the point.
A 64-year-old woman fainted and presented to the ED with a hemoglobin of 3.2 mg/dL because of chronic blood loss from an ulcerated BCC on her upper back. The lesion measured 25 cm by 9 cm, and was 3.5 cm deep with no bone involvement. The woman was addicted to opioids by the time she presented.
She was started on vismodegib; the ulcer shrunk considerably after 6 months, and the woman underwent a resection. Only one small focus of BCC was found across 78 specimens submitted to Dr. Vidimos for Mohs reading.
Resection was followed by a muscle flap repair and radiation. At 5 and a half years, there is no evidence of disease; the only sign that the lesion had been there was a scar running along the woman’s upper spine.
The approach “was very successful for a very aggressive and worrisome tumor,” Dr. Vidimos said.
Dr. Vidimos did not have any relevant disclosures. Dr. Patel had no relevant disclosures.
FROM THE ACMS ANNUAL MEETING
Dr. Topol talks: COVID-19 variants are innocent until proven guilty
Editor in Chief of this news organization Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and professor of molecular medicine, has been closely following COVID-19 data since the pandemic began. He spoke with writer Miriam E. Tucker about the latest on SARS-CoV-2 variants and their impact on vaccine efficacy. The conversation serves as a follow-up to his April 13, 2021, New York Times opinion piece, in which he advised readers that “all variants are innocent until proven guilty.”
You have expressed overall confidence in the efficacy of the vaccines thus far despite the emergence of variants, with some caveats. How do you see the current situation?
The Centers for Disease Control and Prevention has designated five “variants of concern,” but only three of them are real concerns – B.1.1.7, first detected in the United Kingdom; P.1, in Brazil and Japan; and B.1.351, in South Africa. Yet, all three are susceptible to our current vaccines.
The U.K. B.1.1.7 is the worst variant of all because it’s hypertransmissible, so I call it a “superspreader strain.” It also causes more severe illness independent of the spread, so it’s a double whammy. It’s clear that it also causes more deaths. The only arguable point is whether it’s 30% or 50% more deaths, but regardless, it’s more lethal and more transmissible.
The B.1.1.7 is going to be the dominant strain worldwide. It could develop new mutations within it that could come back to haunt us. We must keep watch.
But for now, it’s fully responsive to all the vaccines, which is great because if we didn’t have them, we wouldn’t have gotten through this U.S. pandemic like we have, and neither would Israel and the United Kingdom and other countries that have been able to get out of the crisis. We met the enemy and put it in check.
As for the South Africa variant of concern, B.1.351, we just got some encouraging news showing that it›s very responsive to the Pfizer/BioNTech mRNA vaccine in large numbers of people. The study was conducted in Qatar following that country’s mass immunization campaign in which a total of 385,853 people had received at least one vaccine dose and 265,410 had completed the two doses as of March 31, 2021.
At 2 weeks past the second dose, the vaccine was 75% effective at preventing any documented infection with the B.1.351 variant and 89.5% effective against B.1.1.7. The vaccine’s effectiveness against severe, critical, or fatal COVID-19 was greater than 97.4% for all circulating strains in Qatar, where B.1.1.7 and B.1.351 are most prominent.
We also know that B.1.351 is very responsive to the Johnson & Johnson vaccine and the Novavax [vaccine in development] to a lesser degree. It is the most immune-evading variant we’ve seen thus far, with the highest likelihood of providing some vaccine resistance, yet not enough to interfere with vaccination campaigns. So that’s great news.
The caveats here are that you definitely need two doses of the mRNA vaccines to combat the B.1.351 variant. Also, the AstraZeneca vaccine failed to prevent it in South Africa. However, that study was hard to judge because it was underpowered for number of people with mild infections. So, it didn’t look as if it had any efficacy, but maybe it would if tested in a real trial.
The P.1 (Brazil) variant is the second-highest concern after B.1.1.7 because it’s the only one in the United States that’s still headed up. It seems to be competing a bit with B.1.1.7 here. We know it was associated with the crisis in Brazil, in Chile, and some other South American countries. It has some immune escape, but not as bad as B.1.351. It also appears to have somewhat greater transmissibility but not as much as B.1.1.7.
With P.1, we just don’t know enough yet. It was difficult to assess in Brazil because they were in the midst of a catastrophe – like India is now – and you don’t know how much of it is dragged by the catastrophe vs driving it.
We have to respond to P.1 carefully. There are some good data that it does respond to the Chinese vaccine Sinovac and the AstraZeneca vaccine, and it appears to respond to the others as well, based on serum studies. So it doesn’t look like vaccines will be the worry with this variant. Rather, it could be competing with B.1.1.7 and could lead to breakthrough infections in vaccinated people or reinfections in unvaccinated people who had COVID-19. We need several more weeks to sort it out.
Although the B.1.427 and B.1.429 variants initially seen in California remain on the CDC’s concern list, I’m not worried about them.
You mentioned the current COVID-19 crisis in India, where a new variant has been described as a “double mutant,” but on Twitter you called it a “scariant.” Why?
First of all, the B.1.617 variant isn’t a double mutant. It has 15 mutations. It’s a stupid term, focusing on two mutations which largely have been put aside as to concern. One of them is the L452R, which is the same as one of the California variants, and that hasn’t proved to be particularly serious or concerning. The other is the 484Q, and it’s not clear whether that has any function.
The B.1.617 is not the driver of the catastrophe in India. It may be contributing a small amount, but it has been overhyped as the double mutant that’s causing it all. Adding to that are what I call “scariant” headlines here in the United States when a few cases of that variant have been seen.
I coined the term scariant in early February because it was a pretty clear trend. People don’t know what variants are. They know a little bit about mutations but not variants, and they’re scared. A few variants are concerning, but we keep learning more and more things to decrease the concern. That’s why I wrote the New York Times op-ed, to try to provide some reassurance, since there’s such paranoia.
Do you think booster vaccinations will be necessary? If so, will those be of the original vaccines or new ones that incorporate the variants?
As we go forward, there’s still potential for new variants that we haven’t seen yet that combine the worst of all features – transmissibility and immune evasion – especially since we have a world where COVID-19 is unchecked. So, we’re not out of it yet, but at least for the moment, we have vaccines that are capable of protecting against all variants.
In most people, the immune response against SARS-CoV-2 is very durable and strong and may well last for years. With the most closely related SARS-CoV-1, people still had immune responses up to 18 years later. However, some people will have less robust vaccine responses, including the elderly and the immunocompromised. If they don’t have great responses to the vaccine to start with, over time they’re likely to become more vulnerable, especially if they’re exposed to the variants with some degree of immune evasion.
I think we need to study these individuals post vaccination. A lot of people fit into those categories, including seniors, people being treated for cancer or autoimmune conditions, or post organ transplant. We could set up a prospective study to see whether they develop symptomatic COVID-19 and if so, from what – the original strain, B.1.1.7, or the newer variants.
That’s where I think booster shots may be needed. They may not be necessary across the board, but perhaps just in these special subgroups.
All of the current vaccines can be tweaked to include new variants, but the need for that is uncertain as of now. Moderna is working on a so-called bivalent vaccine that includes the original SARS-CoV-2 strain plus the B.1.351 variant, but it isn’t clear that that’s going to be necessary.
Currently, at least 200 COVID-19 vaccines are in development. There will be vaccines you can inhale, room temperature mRNA vaccines, and potentially even oral vaccines.
In the near future, Novavax is close, and there will likely be a two-dose Johnson & Johnson version that has the same potency as the mRNA vaccines. There are a lot of moving parts here.
There may be a step down in efficacy from mRNA to the others, though, and that shouldn’t be discounted. All of the available vaccines so far protect very well against severe disease and death, but some are less effective against mild to moderate infections, which may then lead to long COVID. We don’t yet know whether those who get mild infection post vaccination can still get long COVID.
What do you think it will take to achieve herd immunity?
I prefer the term “containment.” It’s quantitative. If you get to an infection rate of less than 1 in 100,000 people, as they’ve done in Israel, with 0.8 per 100,000, then you have the virus in check, and there will be very little spread when it’s at that controlled rate, with no outbreaks. The United States is currently at about 15 per 100,000. California is at 4. That still has to get lower.
It will be a challenge to get to President Biden’s goal of having 70% of U.S. adults given at least one dose by July 4. We’re now at about 57%. To get that next 13% of adults is going to take an all-out effort: mobile units, going to homes, making it ultraconvenient, education for people with safety concerns, incentivization, and days off.
We also need to get employers, universities, and health systems to get to the mandatory level. We haven’t done that yet. Some universities have mandated it for students, faculty, and staff. We need it in more health care systems. Right now, we only have a couple. We mandate flu shots, and flu is nothing, compared with COVID-19. And the COVID-19 vaccine is far more efficacious – flu shots are 40% efficacious, while these are 95%. COVID-19 is a tenfold more lethal and serious disease, and much more spreadable.
People are using the lack of full licensure by the Food and Drug Administration – as opposed to emergency use authorization – as an excuse not to get vaccinated. A biologics license application takes time to approve. Meanwhile, we have hundreds of millions of doses that have been well tolerated and incredibly effective.
Another aspect to consider regarding containment is that about 110 million Americans have already had COVID-19, even though only about 30 million cases have been confirmed. Most of these people have immune protection, although it’s not as good as if they have one vaccine dose. But they have enough protection to be part of the story here of the wall against COVID-19 and will help us get through this.
That’s a silver lining of having an unchecked epidemic for the entire year of 2020. The good part is that’s helping to get us to achieve an incredible level of containment when we haven’t even been close. Right now, we’re as good as the country has been in the pandemic, but we still have a long gap to get down to that 1 per 100,000. That’s what we should be working toward, and we can get there.
A version of this article first appeared on Medscape.com.
Editor in Chief of this news organization Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and professor of molecular medicine, has been closely following COVID-19 data since the pandemic began. He spoke with writer Miriam E. Tucker about the latest on SARS-CoV-2 variants and their impact on vaccine efficacy. The conversation serves as a follow-up to his April 13, 2021, New York Times opinion piece, in which he advised readers that “all variants are innocent until proven guilty.”
You have expressed overall confidence in the efficacy of the vaccines thus far despite the emergence of variants, with some caveats. How do you see the current situation?
The Centers for Disease Control and Prevention has designated five “variants of concern,” but only three of them are real concerns – B.1.1.7, first detected in the United Kingdom; P.1, in Brazil and Japan; and B.1.351, in South Africa. Yet, all three are susceptible to our current vaccines.
The U.K. B.1.1.7 is the worst variant of all because it’s hypertransmissible, so I call it a “superspreader strain.” It also causes more severe illness independent of the spread, so it’s a double whammy. It’s clear that it also causes more deaths. The only arguable point is whether it’s 30% or 50% more deaths, but regardless, it’s more lethal and more transmissible.
The B.1.1.7 is going to be the dominant strain worldwide. It could develop new mutations within it that could come back to haunt us. We must keep watch.
But for now, it’s fully responsive to all the vaccines, which is great because if we didn’t have them, we wouldn’t have gotten through this U.S. pandemic like we have, and neither would Israel and the United Kingdom and other countries that have been able to get out of the crisis. We met the enemy and put it in check.
As for the South Africa variant of concern, B.1.351, we just got some encouraging news showing that it›s very responsive to the Pfizer/BioNTech mRNA vaccine in large numbers of people. The study was conducted in Qatar following that country’s mass immunization campaign in which a total of 385,853 people had received at least one vaccine dose and 265,410 had completed the two doses as of March 31, 2021.
At 2 weeks past the second dose, the vaccine was 75% effective at preventing any documented infection with the B.1.351 variant and 89.5% effective against B.1.1.7. The vaccine’s effectiveness against severe, critical, or fatal COVID-19 was greater than 97.4% for all circulating strains in Qatar, where B.1.1.7 and B.1.351 are most prominent.
We also know that B.1.351 is very responsive to the Johnson & Johnson vaccine and the Novavax [vaccine in development] to a lesser degree. It is the most immune-evading variant we’ve seen thus far, with the highest likelihood of providing some vaccine resistance, yet not enough to interfere with vaccination campaigns. So that’s great news.
The caveats here are that you definitely need two doses of the mRNA vaccines to combat the B.1.351 variant. Also, the AstraZeneca vaccine failed to prevent it in South Africa. However, that study was hard to judge because it was underpowered for number of people with mild infections. So, it didn’t look as if it had any efficacy, but maybe it would if tested in a real trial.
The P.1 (Brazil) variant is the second-highest concern after B.1.1.7 because it’s the only one in the United States that’s still headed up. It seems to be competing a bit with B.1.1.7 here. We know it was associated with the crisis in Brazil, in Chile, and some other South American countries. It has some immune escape, but not as bad as B.1.351. It also appears to have somewhat greater transmissibility but not as much as B.1.1.7.
With P.1, we just don’t know enough yet. It was difficult to assess in Brazil because they were in the midst of a catastrophe – like India is now – and you don’t know how much of it is dragged by the catastrophe vs driving it.
We have to respond to P.1 carefully. There are some good data that it does respond to the Chinese vaccine Sinovac and the AstraZeneca vaccine, and it appears to respond to the others as well, based on serum studies. So it doesn’t look like vaccines will be the worry with this variant. Rather, it could be competing with B.1.1.7 and could lead to breakthrough infections in vaccinated people or reinfections in unvaccinated people who had COVID-19. We need several more weeks to sort it out.
Although the B.1.427 and B.1.429 variants initially seen in California remain on the CDC’s concern list, I’m not worried about them.
You mentioned the current COVID-19 crisis in India, where a new variant has been described as a “double mutant,” but on Twitter you called it a “scariant.” Why?
First of all, the B.1.617 variant isn’t a double mutant. It has 15 mutations. It’s a stupid term, focusing on two mutations which largely have been put aside as to concern. One of them is the L452R, which is the same as one of the California variants, and that hasn’t proved to be particularly serious or concerning. The other is the 484Q, and it’s not clear whether that has any function.
The B.1.617 is not the driver of the catastrophe in India. It may be contributing a small amount, but it has been overhyped as the double mutant that’s causing it all. Adding to that are what I call “scariant” headlines here in the United States when a few cases of that variant have been seen.
I coined the term scariant in early February because it was a pretty clear trend. People don’t know what variants are. They know a little bit about mutations but not variants, and they’re scared. A few variants are concerning, but we keep learning more and more things to decrease the concern. That’s why I wrote the New York Times op-ed, to try to provide some reassurance, since there’s such paranoia.
Do you think booster vaccinations will be necessary? If so, will those be of the original vaccines or new ones that incorporate the variants?
As we go forward, there’s still potential for new variants that we haven’t seen yet that combine the worst of all features – transmissibility and immune evasion – especially since we have a world where COVID-19 is unchecked. So, we’re not out of it yet, but at least for the moment, we have vaccines that are capable of protecting against all variants.
In most people, the immune response against SARS-CoV-2 is very durable and strong and may well last for years. With the most closely related SARS-CoV-1, people still had immune responses up to 18 years later. However, some people will have less robust vaccine responses, including the elderly and the immunocompromised. If they don’t have great responses to the vaccine to start with, over time they’re likely to become more vulnerable, especially if they’re exposed to the variants with some degree of immune evasion.
I think we need to study these individuals post vaccination. A lot of people fit into those categories, including seniors, people being treated for cancer or autoimmune conditions, or post organ transplant. We could set up a prospective study to see whether they develop symptomatic COVID-19 and if so, from what – the original strain, B.1.1.7, or the newer variants.
That’s where I think booster shots may be needed. They may not be necessary across the board, but perhaps just in these special subgroups.
All of the current vaccines can be tweaked to include new variants, but the need for that is uncertain as of now. Moderna is working on a so-called bivalent vaccine that includes the original SARS-CoV-2 strain plus the B.1.351 variant, but it isn’t clear that that’s going to be necessary.
Currently, at least 200 COVID-19 vaccines are in development. There will be vaccines you can inhale, room temperature mRNA vaccines, and potentially even oral vaccines.
In the near future, Novavax is close, and there will likely be a two-dose Johnson & Johnson version that has the same potency as the mRNA vaccines. There are a lot of moving parts here.
There may be a step down in efficacy from mRNA to the others, though, and that shouldn’t be discounted. All of the available vaccines so far protect very well against severe disease and death, but some are less effective against mild to moderate infections, which may then lead to long COVID. We don’t yet know whether those who get mild infection post vaccination can still get long COVID.
What do you think it will take to achieve herd immunity?
I prefer the term “containment.” It’s quantitative. If you get to an infection rate of less than 1 in 100,000 people, as they’ve done in Israel, with 0.8 per 100,000, then you have the virus in check, and there will be very little spread when it’s at that controlled rate, with no outbreaks. The United States is currently at about 15 per 100,000. California is at 4. That still has to get lower.
It will be a challenge to get to President Biden’s goal of having 70% of U.S. adults given at least one dose by July 4. We’re now at about 57%. To get that next 13% of adults is going to take an all-out effort: mobile units, going to homes, making it ultraconvenient, education for people with safety concerns, incentivization, and days off.
We also need to get employers, universities, and health systems to get to the mandatory level. We haven’t done that yet. Some universities have mandated it for students, faculty, and staff. We need it in more health care systems. Right now, we only have a couple. We mandate flu shots, and flu is nothing, compared with COVID-19. And the COVID-19 vaccine is far more efficacious – flu shots are 40% efficacious, while these are 95%. COVID-19 is a tenfold more lethal and serious disease, and much more spreadable.
People are using the lack of full licensure by the Food and Drug Administration – as opposed to emergency use authorization – as an excuse not to get vaccinated. A biologics license application takes time to approve. Meanwhile, we have hundreds of millions of doses that have been well tolerated and incredibly effective.
Another aspect to consider regarding containment is that about 110 million Americans have already had COVID-19, even though only about 30 million cases have been confirmed. Most of these people have immune protection, although it’s not as good as if they have one vaccine dose. But they have enough protection to be part of the story here of the wall against COVID-19 and will help us get through this.
That’s a silver lining of having an unchecked epidemic for the entire year of 2020. The good part is that’s helping to get us to achieve an incredible level of containment when we haven’t even been close. Right now, we’re as good as the country has been in the pandemic, but we still have a long gap to get down to that 1 per 100,000. That’s what we should be working toward, and we can get there.
A version of this article first appeared on Medscape.com.
Editor in Chief of this news organization Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and professor of molecular medicine, has been closely following COVID-19 data since the pandemic began. He spoke with writer Miriam E. Tucker about the latest on SARS-CoV-2 variants and their impact on vaccine efficacy. The conversation serves as a follow-up to his April 13, 2021, New York Times opinion piece, in which he advised readers that “all variants are innocent until proven guilty.”
You have expressed overall confidence in the efficacy of the vaccines thus far despite the emergence of variants, with some caveats. How do you see the current situation?
The Centers for Disease Control and Prevention has designated five “variants of concern,” but only three of them are real concerns – B.1.1.7, first detected in the United Kingdom; P.1, in Brazil and Japan; and B.1.351, in South Africa. Yet, all three are susceptible to our current vaccines.
The U.K. B.1.1.7 is the worst variant of all because it’s hypertransmissible, so I call it a “superspreader strain.” It also causes more severe illness independent of the spread, so it’s a double whammy. It’s clear that it also causes more deaths. The only arguable point is whether it’s 30% or 50% more deaths, but regardless, it’s more lethal and more transmissible.
The B.1.1.7 is going to be the dominant strain worldwide. It could develop new mutations within it that could come back to haunt us. We must keep watch.
But for now, it’s fully responsive to all the vaccines, which is great because if we didn’t have them, we wouldn’t have gotten through this U.S. pandemic like we have, and neither would Israel and the United Kingdom and other countries that have been able to get out of the crisis. We met the enemy and put it in check.
As for the South Africa variant of concern, B.1.351, we just got some encouraging news showing that it›s very responsive to the Pfizer/BioNTech mRNA vaccine in large numbers of people. The study was conducted in Qatar following that country’s mass immunization campaign in which a total of 385,853 people had received at least one vaccine dose and 265,410 had completed the two doses as of March 31, 2021.
At 2 weeks past the second dose, the vaccine was 75% effective at preventing any documented infection with the B.1.351 variant and 89.5% effective against B.1.1.7. The vaccine’s effectiveness against severe, critical, or fatal COVID-19 was greater than 97.4% for all circulating strains in Qatar, where B.1.1.7 and B.1.351 are most prominent.
We also know that B.1.351 is very responsive to the Johnson & Johnson vaccine and the Novavax [vaccine in development] to a lesser degree. It is the most immune-evading variant we’ve seen thus far, with the highest likelihood of providing some vaccine resistance, yet not enough to interfere with vaccination campaigns. So that’s great news.
The caveats here are that you definitely need two doses of the mRNA vaccines to combat the B.1.351 variant. Also, the AstraZeneca vaccine failed to prevent it in South Africa. However, that study was hard to judge because it was underpowered for number of people with mild infections. So, it didn’t look as if it had any efficacy, but maybe it would if tested in a real trial.
The P.1 (Brazil) variant is the second-highest concern after B.1.1.7 because it’s the only one in the United States that’s still headed up. It seems to be competing a bit with B.1.1.7 here. We know it was associated with the crisis in Brazil, in Chile, and some other South American countries. It has some immune escape, but not as bad as B.1.351. It also appears to have somewhat greater transmissibility but not as much as B.1.1.7.
With P.1, we just don’t know enough yet. It was difficult to assess in Brazil because they were in the midst of a catastrophe – like India is now – and you don’t know how much of it is dragged by the catastrophe vs driving it.
We have to respond to P.1 carefully. There are some good data that it does respond to the Chinese vaccine Sinovac and the AstraZeneca vaccine, and it appears to respond to the others as well, based on serum studies. So it doesn’t look like vaccines will be the worry with this variant. Rather, it could be competing with B.1.1.7 and could lead to breakthrough infections in vaccinated people or reinfections in unvaccinated people who had COVID-19. We need several more weeks to sort it out.
Although the B.1.427 and B.1.429 variants initially seen in California remain on the CDC’s concern list, I’m not worried about them.
You mentioned the current COVID-19 crisis in India, where a new variant has been described as a “double mutant,” but on Twitter you called it a “scariant.” Why?
First of all, the B.1.617 variant isn’t a double mutant. It has 15 mutations. It’s a stupid term, focusing on two mutations which largely have been put aside as to concern. One of them is the L452R, which is the same as one of the California variants, and that hasn’t proved to be particularly serious or concerning. The other is the 484Q, and it’s not clear whether that has any function.
The B.1.617 is not the driver of the catastrophe in India. It may be contributing a small amount, but it has been overhyped as the double mutant that’s causing it all. Adding to that are what I call “scariant” headlines here in the United States when a few cases of that variant have been seen.
I coined the term scariant in early February because it was a pretty clear trend. People don’t know what variants are. They know a little bit about mutations but not variants, and they’re scared. A few variants are concerning, but we keep learning more and more things to decrease the concern. That’s why I wrote the New York Times op-ed, to try to provide some reassurance, since there’s such paranoia.
Do you think booster vaccinations will be necessary? If so, will those be of the original vaccines or new ones that incorporate the variants?
As we go forward, there’s still potential for new variants that we haven’t seen yet that combine the worst of all features – transmissibility and immune evasion – especially since we have a world where COVID-19 is unchecked. So, we’re not out of it yet, but at least for the moment, we have vaccines that are capable of protecting against all variants.
In most people, the immune response against SARS-CoV-2 is very durable and strong and may well last for years. With the most closely related SARS-CoV-1, people still had immune responses up to 18 years later. However, some people will have less robust vaccine responses, including the elderly and the immunocompromised. If they don’t have great responses to the vaccine to start with, over time they’re likely to become more vulnerable, especially if they’re exposed to the variants with some degree of immune evasion.
I think we need to study these individuals post vaccination. A lot of people fit into those categories, including seniors, people being treated for cancer or autoimmune conditions, or post organ transplant. We could set up a prospective study to see whether they develop symptomatic COVID-19 and if so, from what – the original strain, B.1.1.7, or the newer variants.
That’s where I think booster shots may be needed. They may not be necessary across the board, but perhaps just in these special subgroups.
All of the current vaccines can be tweaked to include new variants, but the need for that is uncertain as of now. Moderna is working on a so-called bivalent vaccine that includes the original SARS-CoV-2 strain plus the B.1.351 variant, but it isn’t clear that that’s going to be necessary.
Currently, at least 200 COVID-19 vaccines are in development. There will be vaccines you can inhale, room temperature mRNA vaccines, and potentially even oral vaccines.
In the near future, Novavax is close, and there will likely be a two-dose Johnson & Johnson version that has the same potency as the mRNA vaccines. There are a lot of moving parts here.
There may be a step down in efficacy from mRNA to the others, though, and that shouldn’t be discounted. All of the available vaccines so far protect very well against severe disease and death, but some are less effective against mild to moderate infections, which may then lead to long COVID. We don’t yet know whether those who get mild infection post vaccination can still get long COVID.
What do you think it will take to achieve herd immunity?
I prefer the term “containment.” It’s quantitative. If you get to an infection rate of less than 1 in 100,000 people, as they’ve done in Israel, with 0.8 per 100,000, then you have the virus in check, and there will be very little spread when it’s at that controlled rate, with no outbreaks. The United States is currently at about 15 per 100,000. California is at 4. That still has to get lower.
It will be a challenge to get to President Biden’s goal of having 70% of U.S. adults given at least one dose by July 4. We’re now at about 57%. To get that next 13% of adults is going to take an all-out effort: mobile units, going to homes, making it ultraconvenient, education for people with safety concerns, incentivization, and days off.
We also need to get employers, universities, and health systems to get to the mandatory level. We haven’t done that yet. Some universities have mandated it for students, faculty, and staff. We need it in more health care systems. Right now, we only have a couple. We mandate flu shots, and flu is nothing, compared with COVID-19. And the COVID-19 vaccine is far more efficacious – flu shots are 40% efficacious, while these are 95%. COVID-19 is a tenfold more lethal and serious disease, and much more spreadable.
People are using the lack of full licensure by the Food and Drug Administration – as opposed to emergency use authorization – as an excuse not to get vaccinated. A biologics license application takes time to approve. Meanwhile, we have hundreds of millions of doses that have been well tolerated and incredibly effective.
Another aspect to consider regarding containment is that about 110 million Americans have already had COVID-19, even though only about 30 million cases have been confirmed. Most of these people have immune protection, although it’s not as good as if they have one vaccine dose. But they have enough protection to be part of the story here of the wall against COVID-19 and will help us get through this.
That’s a silver lining of having an unchecked epidemic for the entire year of 2020. The good part is that’s helping to get us to achieve an incredible level of containment when we haven’t even been close. Right now, we’re as good as the country has been in the pandemic, but we still have a long gap to get down to that 1 per 100,000. That’s what we should be working toward, and we can get there.
A version of this article first appeared on Medscape.com.
Trial: Fecal transplantation safe but ineffective in PsA
The first clinical trial of fecal microbiota transplantation in patients with psoriatic arthritis has found the procedure to be as safe as a sham procedure, but it didn’t show any effectiveness in decreasing PsA symptoms over 6 months, a team of researchers in Denmark reported in Annals of the Rheumatic Diseases (2021 Apr 29. 10.1136/annrheumdis-2020-219511).
Nonetheless, the investigators said the trial indicates fecal microbiota transplantation (FMT) is worthy of further study.
“Overall, we think that the results are very interesting and that the feasibility and safety aspects as well as the clinical results of the trial may encourage more research into the potential of FMT in the treatment of inflammatory arthritis and may help guide the direction of future trials within the field,” lead author Maja S. Kragsnaes, MD, PhD, and principal investigator Torkell Ellingsen, MD, PhD, of Odense (Denmark) University Hospital said together in an interview.
“The most important findings from this trial is that FMT appears to be safe in patients with PsA and that the patients find the treatment acceptable, and it supports future research into the therapeutic potential of FMT in PsA,” they said.
The study evaluated 6-month outcomes of 31 patients randomized to the FMT and sham groups. FMT patients were three times more likely to experience treatment failure – defined by the need for treatment intensification – with failure rates of 60% versus 20% in the sham group.
As a secondary endpoint, the study used 6-month change in the Health Assessment Questionnaire Disability Index (HAQ-DI) and 20% improvement in American College of Rheumatology criteria (ACR20). The sham group demonstrated a greater decrease in HAQ-DI, indicating better physical function (–0.30 vs. –0.07; P = .031). The proportion of ACR20 responders was similar between both groups: 47% for the FMT patients (7 of 15) and 53% for sham (8 of 15).
The study included adults aged 18-75 years with active peripheral disease, defined as three or more swollen joints, who’d been taking at least15 mg methotrexate a week for at least 3 months before enrolling in the study, with a washout period of 12 weeks (26 weeks for those on biologic agents). Four healthy donors provided the stool transplants.
In the study, Dr. Kragsnaes and Dr. Ellingsen acknowledged that FMT has been shown to be safe for Clostridioides difficile infection or inflammatory bowel disease when “thoroughly screened stool” is used. “Hence,” they wrote, “our findings add to the growing body of evidence suggesting a gut-joint axis in the pathogenesis of PsA.”
Factors that may influence the effectiveness of FMT in PsA merit further investigation, Dr. Kragsnaes and Dr. Ellingsen said. “From FMT trials in patients with active ulcerative colitis, higher dose and repeated administration appear to be effective and safe in inducing remission,” they said in their joint statement, pointing to research from China.
“Moreover,” they added, “successes of FMT in inflammatory bowel disease appear to have been driven by ‘superdonors’ characterized by the presence or absence of specific bacteria species.”
They said will continue to investigate the effectiveness of FMT in immune-mediated diseases, including how to characterize superdonors.
“We will conduct new randomized trials using different FMT strategies – by changing the type of administration form, dose, and treatment frequency – to explore whether microbial dysbiosis or specific bacteria are common or decisive mediators of disease activity in inflammatory diseases and whether this proposed relation can be modified without exacerbating the disease,” Dr. Kragsnaes and Dr. Ellingsen said.
Dr. Kragsnaes and Dr. Ellingsen had no relevant financial relationships to disclose.
The first clinical trial of fecal microbiota transplantation in patients with psoriatic arthritis has found the procedure to be as safe as a sham procedure, but it didn’t show any effectiveness in decreasing PsA symptoms over 6 months, a team of researchers in Denmark reported in Annals of the Rheumatic Diseases (2021 Apr 29. 10.1136/annrheumdis-2020-219511).
Nonetheless, the investigators said the trial indicates fecal microbiota transplantation (FMT) is worthy of further study.
“Overall, we think that the results are very interesting and that the feasibility and safety aspects as well as the clinical results of the trial may encourage more research into the potential of FMT in the treatment of inflammatory arthritis and may help guide the direction of future trials within the field,” lead author Maja S. Kragsnaes, MD, PhD, and principal investigator Torkell Ellingsen, MD, PhD, of Odense (Denmark) University Hospital said together in an interview.
“The most important findings from this trial is that FMT appears to be safe in patients with PsA and that the patients find the treatment acceptable, and it supports future research into the therapeutic potential of FMT in PsA,” they said.
The study evaluated 6-month outcomes of 31 patients randomized to the FMT and sham groups. FMT patients were three times more likely to experience treatment failure – defined by the need for treatment intensification – with failure rates of 60% versus 20% in the sham group.
As a secondary endpoint, the study used 6-month change in the Health Assessment Questionnaire Disability Index (HAQ-DI) and 20% improvement in American College of Rheumatology criteria (ACR20). The sham group demonstrated a greater decrease in HAQ-DI, indicating better physical function (–0.30 vs. –0.07; P = .031). The proportion of ACR20 responders was similar between both groups: 47% for the FMT patients (7 of 15) and 53% for sham (8 of 15).
The study included adults aged 18-75 years with active peripheral disease, defined as three or more swollen joints, who’d been taking at least15 mg methotrexate a week for at least 3 months before enrolling in the study, with a washout period of 12 weeks (26 weeks for those on biologic agents). Four healthy donors provided the stool transplants.
In the study, Dr. Kragsnaes and Dr. Ellingsen acknowledged that FMT has been shown to be safe for Clostridioides difficile infection or inflammatory bowel disease when “thoroughly screened stool” is used. “Hence,” they wrote, “our findings add to the growing body of evidence suggesting a gut-joint axis in the pathogenesis of PsA.”
Factors that may influence the effectiveness of FMT in PsA merit further investigation, Dr. Kragsnaes and Dr. Ellingsen said. “From FMT trials in patients with active ulcerative colitis, higher dose and repeated administration appear to be effective and safe in inducing remission,” they said in their joint statement, pointing to research from China.
“Moreover,” they added, “successes of FMT in inflammatory bowel disease appear to have been driven by ‘superdonors’ characterized by the presence or absence of specific bacteria species.”
They said will continue to investigate the effectiveness of FMT in immune-mediated diseases, including how to characterize superdonors.
“We will conduct new randomized trials using different FMT strategies – by changing the type of administration form, dose, and treatment frequency – to explore whether microbial dysbiosis or specific bacteria are common or decisive mediators of disease activity in inflammatory diseases and whether this proposed relation can be modified without exacerbating the disease,” Dr. Kragsnaes and Dr. Ellingsen said.
Dr. Kragsnaes and Dr. Ellingsen had no relevant financial relationships to disclose.
The first clinical trial of fecal microbiota transplantation in patients with psoriatic arthritis has found the procedure to be as safe as a sham procedure, but it didn’t show any effectiveness in decreasing PsA symptoms over 6 months, a team of researchers in Denmark reported in Annals of the Rheumatic Diseases (2021 Apr 29. 10.1136/annrheumdis-2020-219511).
Nonetheless, the investigators said the trial indicates fecal microbiota transplantation (FMT) is worthy of further study.
“Overall, we think that the results are very interesting and that the feasibility and safety aspects as well as the clinical results of the trial may encourage more research into the potential of FMT in the treatment of inflammatory arthritis and may help guide the direction of future trials within the field,” lead author Maja S. Kragsnaes, MD, PhD, and principal investigator Torkell Ellingsen, MD, PhD, of Odense (Denmark) University Hospital said together in an interview.
“The most important findings from this trial is that FMT appears to be safe in patients with PsA and that the patients find the treatment acceptable, and it supports future research into the therapeutic potential of FMT in PsA,” they said.
The study evaluated 6-month outcomes of 31 patients randomized to the FMT and sham groups. FMT patients were three times more likely to experience treatment failure – defined by the need for treatment intensification – with failure rates of 60% versus 20% in the sham group.
As a secondary endpoint, the study used 6-month change in the Health Assessment Questionnaire Disability Index (HAQ-DI) and 20% improvement in American College of Rheumatology criteria (ACR20). The sham group demonstrated a greater decrease in HAQ-DI, indicating better physical function (–0.30 vs. –0.07; P = .031). The proportion of ACR20 responders was similar between both groups: 47% for the FMT patients (7 of 15) and 53% for sham (8 of 15).
The study included adults aged 18-75 years with active peripheral disease, defined as three or more swollen joints, who’d been taking at least15 mg methotrexate a week for at least 3 months before enrolling in the study, with a washout period of 12 weeks (26 weeks for those on biologic agents). Four healthy donors provided the stool transplants.
In the study, Dr. Kragsnaes and Dr. Ellingsen acknowledged that FMT has been shown to be safe for Clostridioides difficile infection or inflammatory bowel disease when “thoroughly screened stool” is used. “Hence,” they wrote, “our findings add to the growing body of evidence suggesting a gut-joint axis in the pathogenesis of PsA.”
Factors that may influence the effectiveness of FMT in PsA merit further investigation, Dr. Kragsnaes and Dr. Ellingsen said. “From FMT trials in patients with active ulcerative colitis, higher dose and repeated administration appear to be effective and safe in inducing remission,” they said in their joint statement, pointing to research from China.
“Moreover,” they added, “successes of FMT in inflammatory bowel disease appear to have been driven by ‘superdonors’ characterized by the presence or absence of specific bacteria species.”
They said will continue to investigate the effectiveness of FMT in immune-mediated diseases, including how to characterize superdonors.
“We will conduct new randomized trials using different FMT strategies – by changing the type of administration form, dose, and treatment frequency – to explore whether microbial dysbiosis or specific bacteria are common or decisive mediators of disease activity in inflammatory diseases and whether this proposed relation can be modified without exacerbating the disease,” Dr. Kragsnaes and Dr. Ellingsen said.
Dr. Kragsnaes and Dr. Ellingsen had no relevant financial relationships to disclose.
FROM ANNALS OF THE RHEUMATIC DISEASES
Genital skin exams in girls: Conduct with care, look for signs of abuse
at the American Academy of Dermatology Virtual Meeting Experience.
“One in four adult women report being childhood victims of sexual abuse, which is just a staggering number. This is an opportunity for us to identify these patients early and give them the terminology to be able to report what is happening to them,” said pediatric dermatologist Kalyani Marathe, MD, MPH, director of the division of dermatology at Cincinnati Children’s Hospital. “We also have the chance to give them a sense of agency over their bodies.”
Dr. Marathe offered the following recommendations when performing a genital skin exam:
- Make sure a “chaperone” is present. “Chaperones are a must when you’re examining children and teens,” she said. “Ask whom they prefer. For prepubertal children, you’re going to usually use the parent who’s there with them. If the parent is their father, they might ask him to step behind the curtain, in which case you can bring over your nurse or medical assistant.” Teens may ask either parent to step out of the room, she said. In that case, a nurse, medical assistant, resident, or trainee can fill in. “If you have male residents or trainees with you and the patient really does not want to be examined by a male, honor their request. Do not force them.”
- Explain why the exam is being performed. Make sure the patient understands why she is being seen, Dr. Marathe advised. For example, say something like “your pediatrician told us that you have an itchy area” or “your mom told us that there’s some loss of color in that area, that you’re having a problem there.” She added that it’s helpful to explain the type of doctor you are, with a comment such as the following: “We’re examining you because we’re doctors who specialize in skin. ... We want to help you feel better and make sure that your skin heals and is healthy.”
- Ask both the child and the parent for permission to perform the exam. While this may seem trivial, “it’s very, very important in setting the right tone for the encounter,” she said. “If the child says yes, we turn to the mom and say: ‘Mom, is it okay for us to do this exam today?’ You can see visible relief on the part of the parent, and as the parent relaxes, the child relaxes. Just saying those few things really makes the encounter so much smoother.” However, “if they say no, you have to honor the response. ... You say: ‘Okay, we’re not going to do the exam today,” and see the patient in a few weeks. If it’s urgent, an exam under anesthesia may be an option, she added.
- Talk to the child about the terms they use for private parts. It can be helpful to ask: “Do you have any terms for your private area?” According to Dr. Marathe, “this is a good chance to educate them on the terms vulva and vagina since they may be using other terminology. Making sure that they have the correct terms will actually help patients identify and report abuse earlier.” Dr. Marathe recalled that a colleague had a patient who’d been calling her private area “pound cake” and had been “reporting to her teacher that someone had been touching her ‘pound cake.’ Her teacher did not know what she meant by that, and this led to a great delay in her childhood abuse being reported.”
- Talk about what will happen during the exam. “I like to show them any instruments that we’re going to be using,” Dr. Marathe said. “If we’re using a flashlight, for example, I like to show them a picture [of a flashlight] or show them that flashlight. If we’re using a camera to do digital photography, show them that. If we’re going to be using a Q-tip or a swab to demonstrate anything or to take a culture, I like to show them that beforehand to make sure that they know what we’re doing.” In regard to photography, “make sure the parent and child know where the photos are going to go, who’s going to see them, what are they going to be used for. If they’re going to be used for educational purposes, make sure they have given explicit permission for that and they know they’ll be deidentified.”
- Make it clear that the exam won’t be painful. It’s important to put both the patient and the parent at ease on this front, Dr. Marathe said. “A lot of parents are concerned that we’re going to do a speculum exam in their prepubertal child. So make sure that it’s clarified ahead of time that we’re not going to be doing a speculum exam.”
Commenting on this topic, Tor Shwayder, MD, a pediatric dermatologist at Henry Ford Health System, Detroit, urged colleagues to take action if they feel suspicious about a possible sign of child abuse, even if they’re far from certain that anything is wrong. “Don’t ignore those feelings in the back of the brain,” he said in an interview.
Most states have child-abuse hotlines for medical professionals, and major hospitals will have child-abuse teams, Dr. Shwayder said. He urged dermatologists to take advantage of these resources when appropriate. “The professionals on the other side of the 800 number or at the hospital will help you. You don’t have to decide immediately whether this is child abuse. You just need to have a suspicion.”
Dr. Marathe and Dr. Shwayder report no disclosures.
at the American Academy of Dermatology Virtual Meeting Experience.
“One in four adult women report being childhood victims of sexual abuse, which is just a staggering number. This is an opportunity for us to identify these patients early and give them the terminology to be able to report what is happening to them,” said pediatric dermatologist Kalyani Marathe, MD, MPH, director of the division of dermatology at Cincinnati Children’s Hospital. “We also have the chance to give them a sense of agency over their bodies.”
Dr. Marathe offered the following recommendations when performing a genital skin exam:
- Make sure a “chaperone” is present. “Chaperones are a must when you’re examining children and teens,” she said. “Ask whom they prefer. For prepubertal children, you’re going to usually use the parent who’s there with them. If the parent is their father, they might ask him to step behind the curtain, in which case you can bring over your nurse or medical assistant.” Teens may ask either parent to step out of the room, she said. In that case, a nurse, medical assistant, resident, or trainee can fill in. “If you have male residents or trainees with you and the patient really does not want to be examined by a male, honor their request. Do not force them.”
- Explain why the exam is being performed. Make sure the patient understands why she is being seen, Dr. Marathe advised. For example, say something like “your pediatrician told us that you have an itchy area” or “your mom told us that there’s some loss of color in that area, that you’re having a problem there.” She added that it’s helpful to explain the type of doctor you are, with a comment such as the following: “We’re examining you because we’re doctors who specialize in skin. ... We want to help you feel better and make sure that your skin heals and is healthy.”
- Ask both the child and the parent for permission to perform the exam. While this may seem trivial, “it’s very, very important in setting the right tone for the encounter,” she said. “If the child says yes, we turn to the mom and say: ‘Mom, is it okay for us to do this exam today?’ You can see visible relief on the part of the parent, and as the parent relaxes, the child relaxes. Just saying those few things really makes the encounter so much smoother.” However, “if they say no, you have to honor the response. ... You say: ‘Okay, we’re not going to do the exam today,” and see the patient in a few weeks. If it’s urgent, an exam under anesthesia may be an option, she added.
- Talk to the child about the terms they use for private parts. It can be helpful to ask: “Do you have any terms for your private area?” According to Dr. Marathe, “this is a good chance to educate them on the terms vulva and vagina since they may be using other terminology. Making sure that they have the correct terms will actually help patients identify and report abuse earlier.” Dr. Marathe recalled that a colleague had a patient who’d been calling her private area “pound cake” and had been “reporting to her teacher that someone had been touching her ‘pound cake.’ Her teacher did not know what she meant by that, and this led to a great delay in her childhood abuse being reported.”
- Talk about what will happen during the exam. “I like to show them any instruments that we’re going to be using,” Dr. Marathe said. “If we’re using a flashlight, for example, I like to show them a picture [of a flashlight] or show them that flashlight. If we’re using a camera to do digital photography, show them that. If we’re going to be using a Q-tip or a swab to demonstrate anything or to take a culture, I like to show them that beforehand to make sure that they know what we’re doing.” In regard to photography, “make sure the parent and child know where the photos are going to go, who’s going to see them, what are they going to be used for. If they’re going to be used for educational purposes, make sure they have given explicit permission for that and they know they’ll be deidentified.”
- Make it clear that the exam won’t be painful. It’s important to put both the patient and the parent at ease on this front, Dr. Marathe said. “A lot of parents are concerned that we’re going to do a speculum exam in their prepubertal child. So make sure that it’s clarified ahead of time that we’re not going to be doing a speculum exam.”
Commenting on this topic, Tor Shwayder, MD, a pediatric dermatologist at Henry Ford Health System, Detroit, urged colleagues to take action if they feel suspicious about a possible sign of child abuse, even if they’re far from certain that anything is wrong. “Don’t ignore those feelings in the back of the brain,” he said in an interview.
Most states have child-abuse hotlines for medical professionals, and major hospitals will have child-abuse teams, Dr. Shwayder said. He urged dermatologists to take advantage of these resources when appropriate. “The professionals on the other side of the 800 number or at the hospital will help you. You don’t have to decide immediately whether this is child abuse. You just need to have a suspicion.”
Dr. Marathe and Dr. Shwayder report no disclosures.
at the American Academy of Dermatology Virtual Meeting Experience.
“One in four adult women report being childhood victims of sexual abuse, which is just a staggering number. This is an opportunity for us to identify these patients early and give them the terminology to be able to report what is happening to them,” said pediatric dermatologist Kalyani Marathe, MD, MPH, director of the division of dermatology at Cincinnati Children’s Hospital. “We also have the chance to give them a sense of agency over their bodies.”
Dr. Marathe offered the following recommendations when performing a genital skin exam:
- Make sure a “chaperone” is present. “Chaperones are a must when you’re examining children and teens,” she said. “Ask whom they prefer. For prepubertal children, you’re going to usually use the parent who’s there with them. If the parent is their father, they might ask him to step behind the curtain, in which case you can bring over your nurse or medical assistant.” Teens may ask either parent to step out of the room, she said. In that case, a nurse, medical assistant, resident, or trainee can fill in. “If you have male residents or trainees with you and the patient really does not want to be examined by a male, honor their request. Do not force them.”
- Explain why the exam is being performed. Make sure the patient understands why she is being seen, Dr. Marathe advised. For example, say something like “your pediatrician told us that you have an itchy area” or “your mom told us that there’s some loss of color in that area, that you’re having a problem there.” She added that it’s helpful to explain the type of doctor you are, with a comment such as the following: “We’re examining you because we’re doctors who specialize in skin. ... We want to help you feel better and make sure that your skin heals and is healthy.”
- Ask both the child and the parent for permission to perform the exam. While this may seem trivial, “it’s very, very important in setting the right tone for the encounter,” she said. “If the child says yes, we turn to the mom and say: ‘Mom, is it okay for us to do this exam today?’ You can see visible relief on the part of the parent, and as the parent relaxes, the child relaxes. Just saying those few things really makes the encounter so much smoother.” However, “if they say no, you have to honor the response. ... You say: ‘Okay, we’re not going to do the exam today,” and see the patient in a few weeks. If it’s urgent, an exam under anesthesia may be an option, she added.
- Talk to the child about the terms they use for private parts. It can be helpful to ask: “Do you have any terms for your private area?” According to Dr. Marathe, “this is a good chance to educate them on the terms vulva and vagina since they may be using other terminology. Making sure that they have the correct terms will actually help patients identify and report abuse earlier.” Dr. Marathe recalled that a colleague had a patient who’d been calling her private area “pound cake” and had been “reporting to her teacher that someone had been touching her ‘pound cake.’ Her teacher did not know what she meant by that, and this led to a great delay in her childhood abuse being reported.”
- Talk about what will happen during the exam. “I like to show them any instruments that we’re going to be using,” Dr. Marathe said. “If we’re using a flashlight, for example, I like to show them a picture [of a flashlight] or show them that flashlight. If we’re using a camera to do digital photography, show them that. If we’re going to be using a Q-tip or a swab to demonstrate anything or to take a culture, I like to show them that beforehand to make sure that they know what we’re doing.” In regard to photography, “make sure the parent and child know where the photos are going to go, who’s going to see them, what are they going to be used for. If they’re going to be used for educational purposes, make sure they have given explicit permission for that and they know they’ll be deidentified.”
- Make it clear that the exam won’t be painful. It’s important to put both the patient and the parent at ease on this front, Dr. Marathe said. “A lot of parents are concerned that we’re going to do a speculum exam in their prepubertal child. So make sure that it’s clarified ahead of time that we’re not going to be doing a speculum exam.”
Commenting on this topic, Tor Shwayder, MD, a pediatric dermatologist at Henry Ford Health System, Detroit, urged colleagues to take action if they feel suspicious about a possible sign of child abuse, even if they’re far from certain that anything is wrong. “Don’t ignore those feelings in the back of the brain,” he said in an interview.
Most states have child-abuse hotlines for medical professionals, and major hospitals will have child-abuse teams, Dr. Shwayder said. He urged dermatologists to take advantage of these resources when appropriate. “The professionals on the other side of the 800 number or at the hospital will help you. You don’t have to decide immediately whether this is child abuse. You just need to have a suspicion.”
Dr. Marathe and Dr. Shwayder report no disclosures.
FROM AAD VMX 2021
Clinical Use of a Diagnostic Gene Expression Signature for Melanocytic Neoplasms
According to National Institutes of Health estimates, more than 90,000 new cases of melanoma were diagnosed in 2018.1 Overall 5-year survival for patients with melanoma exceeds 90%, but individual survival estimates are highly dependent on stage at diagnosis, and survival decreases markedly with metastasis. Therefore, early and accurate diagnosis is critical.
Diagnosis of melanocytic neoplasms usually is performed by dermatopathologists through microscopic examination of stained tissue biopsy sections, a technically simple and effective method that enables a definitive diagnosis of benign nevus or malignant melanoma to be made in most cases. However, approximately 15% of all biopsied melanocytic lesions will exhibit some degree of histopathologic ambiguity,2-4 meaning that some of their microscopic features will be characteristic of a benign nevus while others will suggest the possibility of malignant melanoma. Diagnostic interpretations often vary in these cases, even among experts, and a definitive diagnosis of benign or malignant may be difficult to achieve by microscopy alone.2-4 Because of the marked reduction in survival once a melanoma has metastasized, these diagnostically ambiguous lesions often are treated as possible malignant melanomas with complete surgical excision (or re-excision). However, some experts suggest that many histopathologically ambiguous melanocytic neoplasms are, in fact, benign,5 a notion supported by epidemiologic evidence.6,7 Therefore, excision of many ambiguous melanocytic neoplasms might be avoided if definitive diagnosis could be achieved.
A gene expression signature was developed and validated for use as an adjunct to traditional methods of differentiating malignant melanocytic neoplasms from their benign counterparts.8-11 This test quantifies the RNA transcripts produced by 14 genes known to be overexpressed in malignant melanomas by comparison to benign nevi. These values are then combined algorithmically with measurements of 9 reference genes to produce an objective numerical score that is classified as benign, malignant, or indeterminate. When used by board-certified dermatopathologists and dermatologists confronting ambiguous melanocytic lesions, the test produces substantial increases in definitive diagnoses and prompts changes in treatment recommendations.12,13 However, the long-term consequences of foregoing surgical excision of melanocytic neoplasms that are diagnostically ambiguous but classified as benign by this test have not yet been formally assessed. In the current study, prospectively tested patients whose ambiguous melanocytic neoplasms were classified as benign by the gene expression signature were followed for up to 4.5 years to evaluate the long-term safety of treatment decisions aligned with benign test results.
Methods
Study Population
As part of a prior study,12 US-based dermatopathologists submitted tissue sections from biopsied melanocytic neoplasms determined to be diagnostically ambiguous by histopathology for analysis with the gene expression signature (Myriad Genetics, Inc). Diagnostically ambiguous lesions were those lesions that were described as ambiguous, uncertain, equivocal, indeterminate, or other synonymous terms by the submitting dermatopathologist and therefore lacked a confident diagnosis of benign or malignant prior to testing. Patients initially were tested between May 2014 and August 2014, with samples submitted through a prospective clinical experience study designed to assess the impact of the test on diagnosis and treatment decisions. This study was performed under an institutional review board waiver of consent (Quorum #33403/1).
Patients were eligible for inclusion in the current study if their biopsy specimens (1) had an uncertain preliminary diagnosis according to the submitting dermatopathologist (pretest diagnosis of indeterminate); (2) received a negative (benign) score from the gene expression test; (3) were treated as benign by the dermatologist(s) involved in follow-up care; and (4) were submitted by a single site (St. Joseph Medical Center, Houston, Texas). Although a single dermatopathology site was used for this study, multiple dermatologists were involved in the final treatment of these patients. Patients with benign scores who received additional intervention were excluded, as they may have a lower rate of adverse events (ie, metastasis) than those who did not receive intervention and would therefore skew the analysis population. A total of 25 patients from the prior study met these inclusion criteria. The previously collected12 pretest and posttest de-identified data were compiled from the commercial laboratory databases, and the patients were followed from the time of testing via medical record review performed by the dermatology providers at participating sites. Clinical follow-up data were collected using study-specific case report forms (CRFs) that captured the following: (1) the dates and results of clinical follow-up visits; (2) the type(s) of treatment and interventions (if any) performed at those visits; (3) the specific indication for any intervention performed; (4) any evidence of persistent, locally recurrent, and/or distant melanocytic neoplasia (whether definitively attributable to the tested lesion or not); and (5) death from any cause. The CRF assigned interventions to 1 of 5 categories: excision, excision with sentinel lymph node biopsy, referral to dermatologic or other surgeon, examination only (without surgical intervention), and other. Selection of other required a free-text description of the treatment and indications. Pertinent information not otherwise captured by the CRF also was recordable as free text.
Gene Expression Testing
Gene expression testing was carried out at the time of specimen submission in the prior study12 as described previously.14 Briefly, formalin-fixed, paraffin-embedded, unstained tissue sections and/or tissue blocks were submitted for testing along with a single hematoxylin and eosin–stained slide used to identify and designate the representative portion(s) of the lesion to be tested. These areas were macrodissected from unstained tissue sections and pooled for RNA extraction. Expression of 14 biomarker genes and 9 reference genes was measured via
Statistical Analysis
Demographic and other baseline characteristics of the patient population were summarized. Follow-up time was calculated as the interval between the date a patient’s gene expression test result was first issued to the provider and the date of the patient’s last recorded visit during the study period. All patient dermatology office visits within the designated follow-up period were documented, with a nonstandard number of visits and follow-up time across all study patients. Statistical analyses were conducted using SAS software (SAS Institute Inc), R software version 3.5.0 (R Foundation for Statistical Computing), and IBM SPSS Statistics software (IBM SPSS Statistics for Windows, Version 25).
Results
Patient Sample
A total of 25 ambiguous melanocytic neoplasms from 25 patients met the study inclusion criteria of a benign gene expression result with subsequent treatment as a benign neoplasm during follow-up. The patient sample statistics are summarized in Table 1. Most patients were younger than 65 years, with an average age at the time of biopsy of 48.4 years. All 25 neoplasms produced negative (benign) gene expression signature scores, all were diagnosed as benign nevi posttest by the submitting dermatopathologist, and all patients were initially treated in accordance with the benign diagnosis by the dermatologist(s) involved in clinical follow-up care. Prior to testing with the gene expression signature, most of these histopathologically indeterminate lesions received differential diagnoses, the most common of which were dysplastic nevus (84%), melanoma arising from a nevus (72%), and superficial spreading melanoma (64%; eTable). After testing with the gene expression signature and receiving a benign score, most lesions received a single differential diagnosis of dysplastic nevus (88%).


Follow-up and Survival
Clinical follow-up time ranged from 0.6 to 53.3 months, with a mean duration (SD) of 38.5 (16.6) months, and patients attended an average of 4 postbiopsy dermatology appointments (mean [SD], 4.6 [3.6]). According to the participating dermatology care providers, none of the 25 patients developed any indication during follow-up that the diagnosis of benign nevus was inaccurate. No patient had evidence of locally recurrent or metastatic melanoma, and none died during the study period.
Treatment/Interventions
The treatment recorded in the CRF was examination only for 21 of 25 patients, excision for 3, and other for 1 (Table 2). Because the explanation for the selection of other in this case described an excision performed at the same anatomic location as the biopsy, this treatment also was considered an excision for purposes of the study analyses. The 3 excisions all occurred at the first postbiopsy dermatology encounter. Across all follow-up visits, no additional surgical interventions occurred (Table 2).
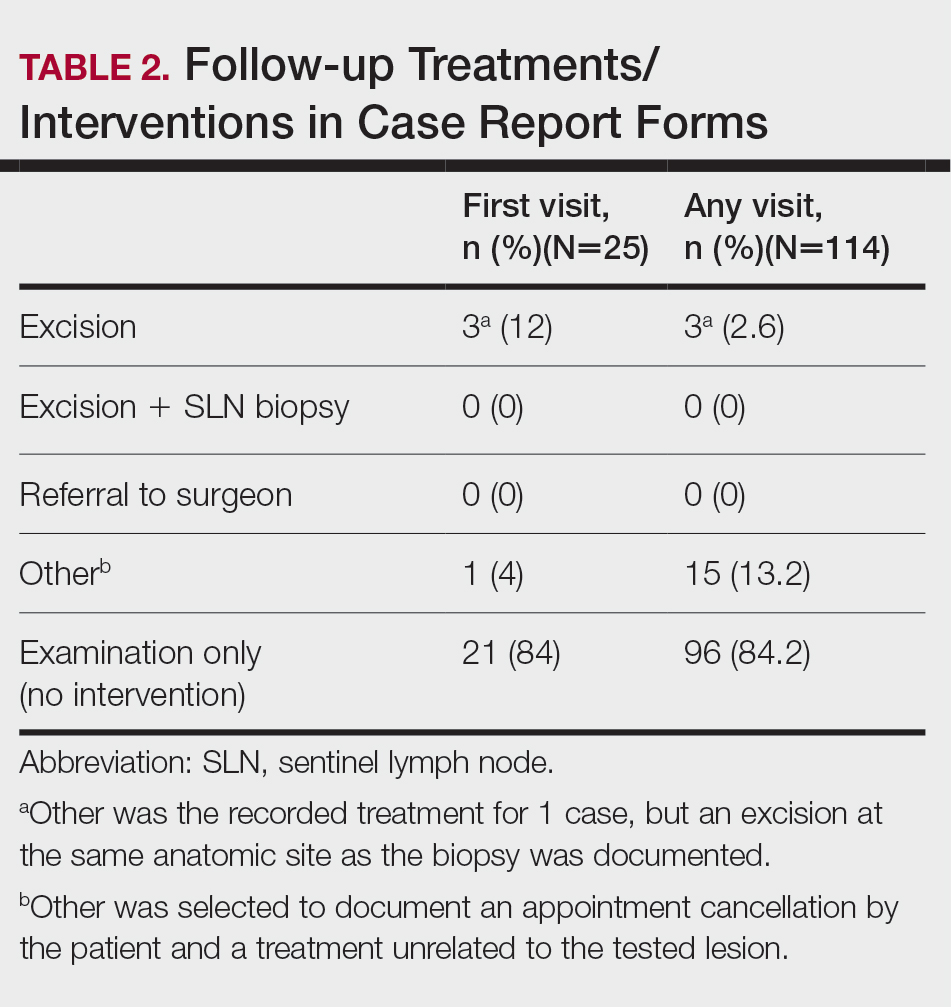
The first excision (case 1) involved a 67-year-old woman with a lesion on the mid pubic region described clinically as an atypical nevus that generated a pretest histopathologic differential diagnosis including dysplastic nevus, superficial spreading melanoma, and melanoma arising within a nevus (Table 3; Figure, A and B). The gene expression test result was benign (score, −5.4), and the final pathology report diagnosis was nevus with junctional dysplasia, moderate. Surgical excision was performed at the patient’s first return visit, 505 days after initial diagnosis, with moderately dysplastic nevus as the recorded indication for removal. No repigmentation or other evidence of local recurrence or progression was detected, and the treating dermatologist indicated no suspicion that the original diagnosis of benign nevus was incorrect during the 23-month follow-up period.
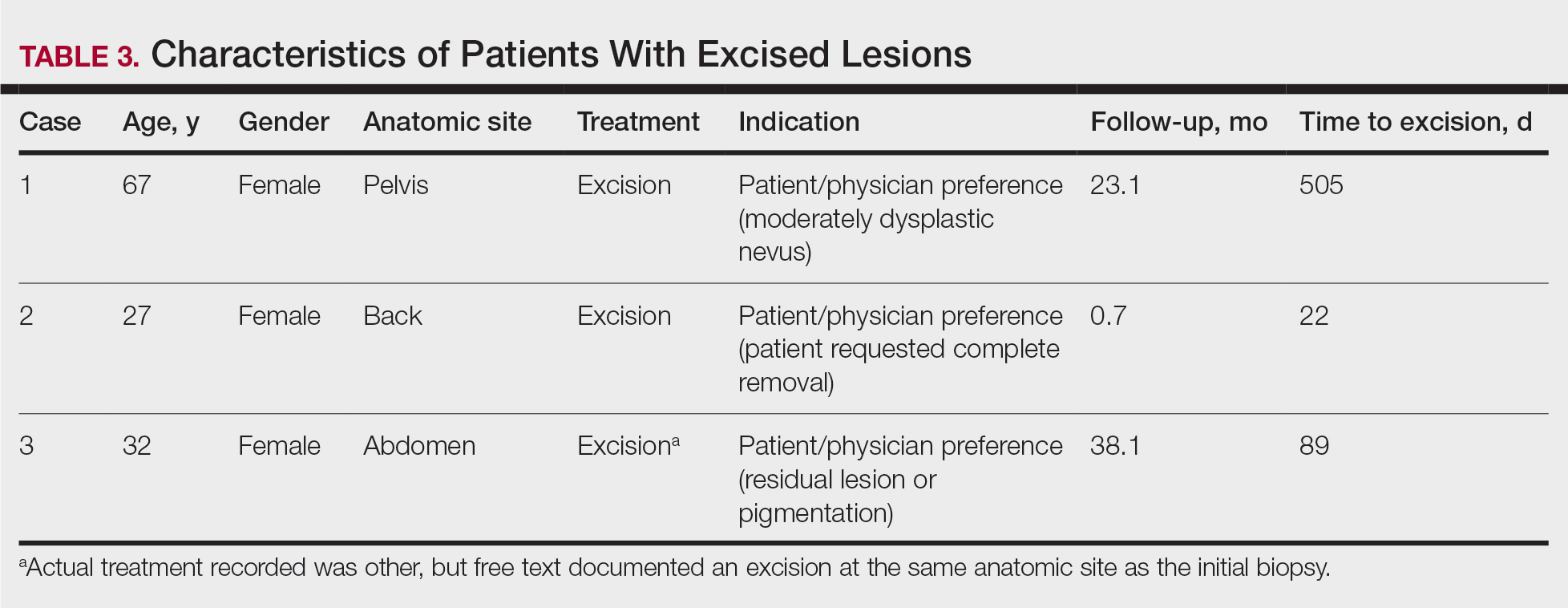
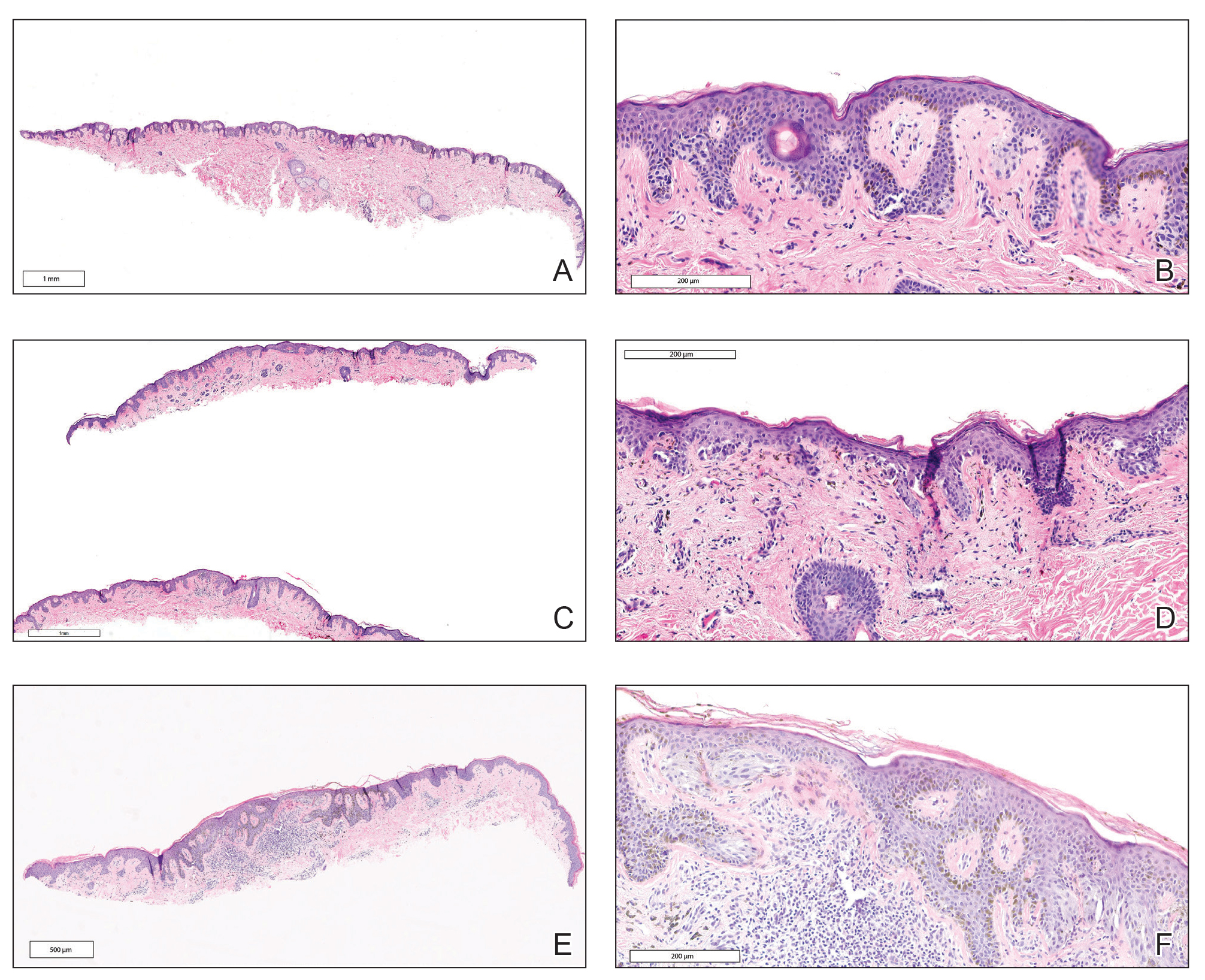
The second excision (case 2) involved a 27-year-old woman with a pigmented neoplasm on the mid upper back (Figure, C and D) biopsied to rule out dysplastic nevus that resulted in a pretest histopathologic differential diagnosis of dysplastic nevus vs superficial spreading melanoma or melanoma arising within a nevus. The gene expression test result classified the lesion as benign (score, −2.9), and the final pathology diagnosis was nevus, compound, with moderate dysplasia. Despite the benign diagnosis, residual neoplasm (or pigmentation) at the biopsy site prompted the patient to request excision at her first postbiopsy visit, 22 days after testing (Table 3). The CRF completed by the dermatologist reported no indication that the benign diagnosis was inaccurate, but the patient was subsequently lost to follow-up.
The third excision (case 3) involved a 32-year-old woman with a pigmented lesion on the abdomen (Table 3; Figure, E and F). The clinical description was irregular-appearing black papule, nevus with atypia, and the histopathologic differential diagnosis again included dysplastic nevus, superficial spreading melanoma, and melanoma arising within a preexisting nevus. The gene expression signature result was benign (score, −7.2), and the final diagnosis issued within the accompanying pathology report was nevus with moderate junctional dysplasia. Despite the benign diagnosis, excision was performed 89 days after test result availability, with apparent residual pigmentation as the specified indication. As with the other 2 cases, the treating dermatologist confirmed that neither clinical features nor follow-up events suggested malignancy.
Comment
This study followed a cohort of 25 patients with histopathologically ambiguous melanocytic neoplasms that were classified as benign by a diagnostic gene expression test with the intent of determining the outcomes of patients whose treatment aligned with their benign test result. All patients initially were managed according to their test result. During an average posttest clinical follow-up time of more than 3 years (38.5 months), the 25 biopsied lesions, most of which received a differential diagnosis of dysplastic nevus, were regarded as benign nevi by their dermatologists, and the vast majority (88%) received no further surgical intervention. Three patients underwent subsequent excision of the biopsied lesion, with patient or physician preference as the indication in each instance. None of the 25 patients developed evidence of local recurrence, metastasis, or other findings that prompted doubt of the benign diagnosis. The absence of adverse events during clinical follow-up, particularly given that most lesions were not subjected to further intervention, supports use of the gene expression test as a safe and effective adjunct to the diagnosis and treatment of ambiguous melanocytic neoplasms by dermatologists and dermatopathologists.
Ambiguous melanocytic neoplasms evaluated without the aid of molecular adjuncts often result in equivocal or less-than-definitive diagnoses, and further surgical intervention is commonly undertaken to mitigate against the possibility of a missed melanoma.13 In this study, treatment that was aligned with the benign test result allowed most patients to avoid further surgical intervention, which suggests that adjunctive use of the gene signature can contribute to reductions in the physical and economic burdens imposed by unnecessary surgical interventions.15,16 Moreover, any means of increasing accurate and definitive diagnoses may produce an immediate impact on health outcomes by reducing the anxiety that uncertainty often provokes in patients and health care providers alike.
Study Limitations
This study must be interpreted within the context of its limitations. Obtaining meaningful patient outcome data is a common challenge in health care research due to the requisite length of follow-up and sometimes the lack of definitive evidence of adverse events. This is particularly difficult for melanocytic neoplasms because of an apparent inclination for patients with benign diagnoses to abandon follow-up and an increasing tendency for even minimal diagnostic uncertainty to prompt complete excision. Additionally, the only definitive clinical outcome for melanocytic neoplasms is distant metastasis, which (fortunately for patients) is relatively rare. Not surprisingly, studies documenting clinical outcomes of patients with ambiguous melanocytic neoplasms tested prospectively with diagnostic adjuncts are scarce, and this study’s sample size and clinical follow-up compare favorably with the few that exist.17,18 Although most melanomas declare themselves through recurrence or metastasis within several years of initial biopsy,1,19 some are clinically dormant for as long as 10 years after initial detection.20,21 This may be particularly true for the small or early-stage lesions that now comprise the majority of biopsied neoplasms, and such events would go undetected by this study and many others. It also must be recognized that uneventful follow-up, regardless of duration, cannot prove that a biopsied melanocytic neoplasm was benign. Although only 5 patients had a follow-up time of less than 2 years (the time frame in which most recurrence or metastasis will occur), it cannot be definitively proven that a minimum of 2 years recurrence- or metastasis-free survival indicates a benign lesion. Many early-stage malignant melanomas are eradicated by complete excision or even by the initial biopsy if margins are uninvolved.
Because these limitations are intrinsic to melanocytic neoplasms and current management strategies, they pertain to all investigations seeking insights into biological potential through clinical outcomes. Similarly, all current diagnostic tools and procedures have the potential for sampling error, including histopathology. The rarity of adverse outcomes (recurrence and metastasis) in patients with benign test results within this cohort indicates that false-negative results are uncommon, which is further evidenced by a similar rarity of adverse events in prior studies of the gene expression signature.8-10,22 A particular strength of this study is that most of the ambiguous melanocytic neoplasms followed did not undergo excision after the initial biopsy, an increasingly uncommon situation that may increase their likelihood to be informative.
It must be emphasized that the gene expression test, similar to other diagnostic adjuncts, is neither a replacement for histopathologic interpretation nor a substitute for judgment. As with all tests, it can produce false-positive and false-negative results. Therefore, it should always be interpreted within the constellation of the many other data points that must be considered when making a distinction between benign nevus and malignant melanoma, including but not limited to patient age, family and personal history of melanoma, anatomic location, clinical features, and histopathologic findings. As is the case for many diseases, careful consideration of all relevant input is necessary to minimize the risk of misdiagnosis that might occur should any single data point prove inaccurate, including the results of adjunctive molecular tests.
Conclusion
Ancillary methods are emerging as useful tools for the diagnostic evaluation of melanocytic neoplasms that cannot be assigned definitive diagnoses using traditional techniques alone. This study suggests that patients with ambiguous melanocytic neoplasms may benefit from diagnoses and treatment decisions aligned with the results of a gene expression test, and that for those with a benign result, simple observation may be a safe alternative to surgical excision. This expands upon prior observations of the test’s influence on diagnoses and treatment decisions and supports its role as part of dermatopathologists’ and dermatologists’ decision-making process for histopathologically ambiguous melanocytic lesions.
- Noone AM, Howlander N, Krapcho M, et al, eds. SEER Cancer Statistics Review, 1975-2015. National Cancer Institute website. Updated September 10, 2018. Accessed April 21, 2021. https://seer.cancer.gov/archive/csr/1975_2015/
- Shoo BA, Sagebiel RW, Kashani-Sabet M. Discordance in the histopathologic diagnosis of melanoma at a melanoma referral center. J Am Acad Dermatol. 2010;62:751-756.
- Veenhuizen KC, De Wit PE, Mooi WJ, et al. Quality assessment by expert opinion in melanoma pathology: experience of the pathology panel of the Dutch Melanoma Working Party. J Pathol. 1997;182:266-272.
- Elmore JG, Barnhill RL, Elder DE, et al. Pathologists’ diagnosis of invasive melanoma and melanocytic proliferations: observer accuracy and reproducibility study. BMJ. 2017;357:j2813. doi:10.1136/bmj.j2813
- Glusac EJ. The melanoma ‘epidemic’, a dermatopathologist’s perspective. J Cutan Pathol. 2011;38:264-267.
- Welch HG, Woloshin S, Schwartz LM. Skin biopsy rates and incidence of melanoma: population based ecological study. BMJ. 2005;331:481.
- Swerlick RA, Chen S. The melanoma epidemic. Is increased surveillance the solution or the problem? Arch Dermatol. 1996;132:881-884.
- Ko JS, Matharoo-Ball B, Billings SD, et al. Diagnostic distinction of malignant melanoma and benign nevi by a gene expression signature and correlation to clinical outcomes. Cancer Epidemiol Biomarkers Prev. 2017;26:1107-1113.
- Clarke LE, Flake DD 2nd, Busam K, et al. An independent validation of a gene expression signature to differentiate malignant melanoma from benign melanocytic nevi. Cancer. 2017;123:617-628.
- Clarke LE, Warf BM, Flake DD 2nd, et al. Clinical validation of a gene expression signature that differentiates benign nevi from malignant melanoma. J Cutan Pathol. 2015;42:244-252.
- Minca EC, Al-Rohil RN, Wang M, et al. Comparison between melanoma gene expression score and fluorescence in situ hybridization for the classification of melanocytic lesions. Mod Pathol. 2016;29:832-843.
- Cockerell CJ, Tschen J, Evans B, et al. The influence of a gene expression signature on the diagnosis and recommended treatment of melanocytic tumors by dermatopathologists. Medicine (Baltimore). 2016;95:e4887. doi:10.1097/MD.0000000000004887
- Cockerell C, Tschen J, Billings SD, et al. The influence of a gene-expression signature on the treatment of diagnostically challenging melanocytic lesions. Per Med. 2017;14:123-130.
- Warf MB, Flake DD 2nd, Adams D, et al. Analytical validation of a melanoma diagnostic gene signature using formalin-fixed paraffin-embedded melanocytic lesions. Biomark Med. 2015;9:407-416.
- Guy GP Jr, Ekwueme DU, Tangka FK, et al. Melanoma treatment costs: a systematic review of the literature, 1990-2011. Am J Prev Med. 2012;43:537-545.
- Guy GP Jr, Machlin SR, Ekwueme DU, et al. Prevalence and costs of skin cancer treatment in the U.S., 2002-2006 and 2007-2011. Am J Prev Med. 2015;48:183-187.
- Egnatios GL, Ferringer TC. Clinical follow-up of atypical spitzoid tumors analyzed by fluorescence in situ hybridization. Am J Dermatopathol. 2016;38:289-296.
- Fischer AS, High WA. The difficulty in interpreting gene expression profiling in BAP-negative melanocytic tumors. J Cutan Pathol. 2018;45:659-666. doi:10.1111/cup.13277
- Vollmer RT. The dynamics of death in melanoma. J Cutan Pathol. 2012;39:1075-1082.
- Osella-Abate S, Ribero S, Sanlorenzo M, et al. Risk factors related to late metastases in 1,372 melanoma patients disease free more than 10 years. Int J Cancer. 2015;136:2453-2457.
- Faries MB, Steen S, Ye X, et al. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg. 2013;217:27-34.
- Ko JS, Clarke LE, Minca EC, et al. Correlation of melanoma gene expression score with clinical outcomes on a series of melanocytic lesions. Hum Pathol. 2019;86:213-221.
According to National Institutes of Health estimates, more than 90,000 new cases of melanoma were diagnosed in 2018.1 Overall 5-year survival for patients with melanoma exceeds 90%, but individual survival estimates are highly dependent on stage at diagnosis, and survival decreases markedly with metastasis. Therefore, early and accurate diagnosis is critical.
Diagnosis of melanocytic neoplasms usually is performed by dermatopathologists through microscopic examination of stained tissue biopsy sections, a technically simple and effective method that enables a definitive diagnosis of benign nevus or malignant melanoma to be made in most cases. However, approximately 15% of all biopsied melanocytic lesions will exhibit some degree of histopathologic ambiguity,2-4 meaning that some of their microscopic features will be characteristic of a benign nevus while others will suggest the possibility of malignant melanoma. Diagnostic interpretations often vary in these cases, even among experts, and a definitive diagnosis of benign or malignant may be difficult to achieve by microscopy alone.2-4 Because of the marked reduction in survival once a melanoma has metastasized, these diagnostically ambiguous lesions often are treated as possible malignant melanomas with complete surgical excision (or re-excision). However, some experts suggest that many histopathologically ambiguous melanocytic neoplasms are, in fact, benign,5 a notion supported by epidemiologic evidence.6,7 Therefore, excision of many ambiguous melanocytic neoplasms might be avoided if definitive diagnosis could be achieved.
A gene expression signature was developed and validated for use as an adjunct to traditional methods of differentiating malignant melanocytic neoplasms from their benign counterparts.8-11 This test quantifies the RNA transcripts produced by 14 genes known to be overexpressed in malignant melanomas by comparison to benign nevi. These values are then combined algorithmically with measurements of 9 reference genes to produce an objective numerical score that is classified as benign, malignant, or indeterminate. When used by board-certified dermatopathologists and dermatologists confronting ambiguous melanocytic lesions, the test produces substantial increases in definitive diagnoses and prompts changes in treatment recommendations.12,13 However, the long-term consequences of foregoing surgical excision of melanocytic neoplasms that are diagnostically ambiguous but classified as benign by this test have not yet been formally assessed. In the current study, prospectively tested patients whose ambiguous melanocytic neoplasms were classified as benign by the gene expression signature were followed for up to 4.5 years to evaluate the long-term safety of treatment decisions aligned with benign test results.
Methods
Study Population
As part of a prior study,12 US-based dermatopathologists submitted tissue sections from biopsied melanocytic neoplasms determined to be diagnostically ambiguous by histopathology for analysis with the gene expression signature (Myriad Genetics, Inc). Diagnostically ambiguous lesions were those lesions that were described as ambiguous, uncertain, equivocal, indeterminate, or other synonymous terms by the submitting dermatopathologist and therefore lacked a confident diagnosis of benign or malignant prior to testing. Patients initially were tested between May 2014 and August 2014, with samples submitted through a prospective clinical experience study designed to assess the impact of the test on diagnosis and treatment decisions. This study was performed under an institutional review board waiver of consent (Quorum #33403/1).
Patients were eligible for inclusion in the current study if their biopsy specimens (1) had an uncertain preliminary diagnosis according to the submitting dermatopathologist (pretest diagnosis of indeterminate); (2) received a negative (benign) score from the gene expression test; (3) were treated as benign by the dermatologist(s) involved in follow-up care; and (4) were submitted by a single site (St. Joseph Medical Center, Houston, Texas). Although a single dermatopathology site was used for this study, multiple dermatologists were involved in the final treatment of these patients. Patients with benign scores who received additional intervention were excluded, as they may have a lower rate of adverse events (ie, metastasis) than those who did not receive intervention and would therefore skew the analysis population. A total of 25 patients from the prior study met these inclusion criteria. The previously collected12 pretest and posttest de-identified data were compiled from the commercial laboratory databases, and the patients were followed from the time of testing via medical record review performed by the dermatology providers at participating sites. Clinical follow-up data were collected using study-specific case report forms (CRFs) that captured the following: (1) the dates and results of clinical follow-up visits; (2) the type(s) of treatment and interventions (if any) performed at those visits; (3) the specific indication for any intervention performed; (4) any evidence of persistent, locally recurrent, and/or distant melanocytic neoplasia (whether definitively attributable to the tested lesion or not); and (5) death from any cause. The CRF assigned interventions to 1 of 5 categories: excision, excision with sentinel lymph node biopsy, referral to dermatologic or other surgeon, examination only (without surgical intervention), and other. Selection of other required a free-text description of the treatment and indications. Pertinent information not otherwise captured by the CRF also was recordable as free text.
Gene Expression Testing
Gene expression testing was carried out at the time of specimen submission in the prior study12 as described previously.14 Briefly, formalin-fixed, paraffin-embedded, unstained tissue sections and/or tissue blocks were submitted for testing along with a single hematoxylin and eosin–stained slide used to identify and designate the representative portion(s) of the lesion to be tested. These areas were macrodissected from unstained tissue sections and pooled for RNA extraction. Expression of 14 biomarker genes and 9 reference genes was measured via
Statistical Analysis
Demographic and other baseline characteristics of the patient population were summarized. Follow-up time was calculated as the interval between the date a patient’s gene expression test result was first issued to the provider and the date of the patient’s last recorded visit during the study period. All patient dermatology office visits within the designated follow-up period were documented, with a nonstandard number of visits and follow-up time across all study patients. Statistical analyses were conducted using SAS software (SAS Institute Inc), R software version 3.5.0 (R Foundation for Statistical Computing), and IBM SPSS Statistics software (IBM SPSS Statistics for Windows, Version 25).
Results
Patient Sample
A total of 25 ambiguous melanocytic neoplasms from 25 patients met the study inclusion criteria of a benign gene expression result with subsequent treatment as a benign neoplasm during follow-up. The patient sample statistics are summarized in Table 1. Most patients were younger than 65 years, with an average age at the time of biopsy of 48.4 years. All 25 neoplasms produced negative (benign) gene expression signature scores, all were diagnosed as benign nevi posttest by the submitting dermatopathologist, and all patients were initially treated in accordance with the benign diagnosis by the dermatologist(s) involved in clinical follow-up care. Prior to testing with the gene expression signature, most of these histopathologically indeterminate lesions received differential diagnoses, the most common of which were dysplastic nevus (84%), melanoma arising from a nevus (72%), and superficial spreading melanoma (64%; eTable). After testing with the gene expression signature and receiving a benign score, most lesions received a single differential diagnosis of dysplastic nevus (88%).


Follow-up and Survival
Clinical follow-up time ranged from 0.6 to 53.3 months, with a mean duration (SD) of 38.5 (16.6) months, and patients attended an average of 4 postbiopsy dermatology appointments (mean [SD], 4.6 [3.6]). According to the participating dermatology care providers, none of the 25 patients developed any indication during follow-up that the diagnosis of benign nevus was inaccurate. No patient had evidence of locally recurrent or metastatic melanoma, and none died during the study period.
Treatment/Interventions
The treatment recorded in the CRF was examination only for 21 of 25 patients, excision for 3, and other for 1 (Table 2). Because the explanation for the selection of other in this case described an excision performed at the same anatomic location as the biopsy, this treatment also was considered an excision for purposes of the study analyses. The 3 excisions all occurred at the first postbiopsy dermatology encounter. Across all follow-up visits, no additional surgical interventions occurred (Table 2).

The first excision (case 1) involved a 67-year-old woman with a lesion on the mid pubic region described clinically as an atypical nevus that generated a pretest histopathologic differential diagnosis including dysplastic nevus, superficial spreading melanoma, and melanoma arising within a nevus (Table 3; Figure, A and B). The gene expression test result was benign (score, −5.4), and the final pathology report diagnosis was nevus with junctional dysplasia, moderate. Surgical excision was performed at the patient’s first return visit, 505 days after initial diagnosis, with moderately dysplastic nevus as the recorded indication for removal. No repigmentation or other evidence of local recurrence or progression was detected, and the treating dermatologist indicated no suspicion that the original diagnosis of benign nevus was incorrect during the 23-month follow-up period.


The second excision (case 2) involved a 27-year-old woman with a pigmented neoplasm on the mid upper back (Figure, C and D) biopsied to rule out dysplastic nevus that resulted in a pretest histopathologic differential diagnosis of dysplastic nevus vs superficial spreading melanoma or melanoma arising within a nevus. The gene expression test result classified the lesion as benign (score, −2.9), and the final pathology diagnosis was nevus, compound, with moderate dysplasia. Despite the benign diagnosis, residual neoplasm (or pigmentation) at the biopsy site prompted the patient to request excision at her first postbiopsy visit, 22 days after testing (Table 3). The CRF completed by the dermatologist reported no indication that the benign diagnosis was inaccurate, but the patient was subsequently lost to follow-up.
The third excision (case 3) involved a 32-year-old woman with a pigmented lesion on the abdomen (Table 3; Figure, E and F). The clinical description was irregular-appearing black papule, nevus with atypia, and the histopathologic differential diagnosis again included dysplastic nevus, superficial spreading melanoma, and melanoma arising within a preexisting nevus. The gene expression signature result was benign (score, −7.2), and the final diagnosis issued within the accompanying pathology report was nevus with moderate junctional dysplasia. Despite the benign diagnosis, excision was performed 89 days after test result availability, with apparent residual pigmentation as the specified indication. As with the other 2 cases, the treating dermatologist confirmed that neither clinical features nor follow-up events suggested malignancy.
Comment
This study followed a cohort of 25 patients with histopathologically ambiguous melanocytic neoplasms that were classified as benign by a diagnostic gene expression test with the intent of determining the outcomes of patients whose treatment aligned with their benign test result. All patients initially were managed according to their test result. During an average posttest clinical follow-up time of more than 3 years (38.5 months), the 25 biopsied lesions, most of which received a differential diagnosis of dysplastic nevus, were regarded as benign nevi by their dermatologists, and the vast majority (88%) received no further surgical intervention. Three patients underwent subsequent excision of the biopsied lesion, with patient or physician preference as the indication in each instance. None of the 25 patients developed evidence of local recurrence, metastasis, or other findings that prompted doubt of the benign diagnosis. The absence of adverse events during clinical follow-up, particularly given that most lesions were not subjected to further intervention, supports use of the gene expression test as a safe and effective adjunct to the diagnosis and treatment of ambiguous melanocytic neoplasms by dermatologists and dermatopathologists.
Ambiguous melanocytic neoplasms evaluated without the aid of molecular adjuncts often result in equivocal or less-than-definitive diagnoses, and further surgical intervention is commonly undertaken to mitigate against the possibility of a missed melanoma.13 In this study, treatment that was aligned with the benign test result allowed most patients to avoid further surgical intervention, which suggests that adjunctive use of the gene signature can contribute to reductions in the physical and economic burdens imposed by unnecessary surgical interventions.15,16 Moreover, any means of increasing accurate and definitive diagnoses may produce an immediate impact on health outcomes by reducing the anxiety that uncertainty often provokes in patients and health care providers alike.
Study Limitations
This study must be interpreted within the context of its limitations. Obtaining meaningful patient outcome data is a common challenge in health care research due to the requisite length of follow-up and sometimes the lack of definitive evidence of adverse events. This is particularly difficult for melanocytic neoplasms because of an apparent inclination for patients with benign diagnoses to abandon follow-up and an increasing tendency for even minimal diagnostic uncertainty to prompt complete excision. Additionally, the only definitive clinical outcome for melanocytic neoplasms is distant metastasis, which (fortunately for patients) is relatively rare. Not surprisingly, studies documenting clinical outcomes of patients with ambiguous melanocytic neoplasms tested prospectively with diagnostic adjuncts are scarce, and this study’s sample size and clinical follow-up compare favorably with the few that exist.17,18 Although most melanomas declare themselves through recurrence or metastasis within several years of initial biopsy,1,19 some are clinically dormant for as long as 10 years after initial detection.20,21 This may be particularly true for the small or early-stage lesions that now comprise the majority of biopsied neoplasms, and such events would go undetected by this study and many others. It also must be recognized that uneventful follow-up, regardless of duration, cannot prove that a biopsied melanocytic neoplasm was benign. Although only 5 patients had a follow-up time of less than 2 years (the time frame in which most recurrence or metastasis will occur), it cannot be definitively proven that a minimum of 2 years recurrence- or metastasis-free survival indicates a benign lesion. Many early-stage malignant melanomas are eradicated by complete excision or even by the initial biopsy if margins are uninvolved.
Because these limitations are intrinsic to melanocytic neoplasms and current management strategies, they pertain to all investigations seeking insights into biological potential through clinical outcomes. Similarly, all current diagnostic tools and procedures have the potential for sampling error, including histopathology. The rarity of adverse outcomes (recurrence and metastasis) in patients with benign test results within this cohort indicates that false-negative results are uncommon, which is further evidenced by a similar rarity of adverse events in prior studies of the gene expression signature.8-10,22 A particular strength of this study is that most of the ambiguous melanocytic neoplasms followed did not undergo excision after the initial biopsy, an increasingly uncommon situation that may increase their likelihood to be informative.
It must be emphasized that the gene expression test, similar to other diagnostic adjuncts, is neither a replacement for histopathologic interpretation nor a substitute for judgment. As with all tests, it can produce false-positive and false-negative results. Therefore, it should always be interpreted within the constellation of the many other data points that must be considered when making a distinction between benign nevus and malignant melanoma, including but not limited to patient age, family and personal history of melanoma, anatomic location, clinical features, and histopathologic findings. As is the case for many diseases, careful consideration of all relevant input is necessary to minimize the risk of misdiagnosis that might occur should any single data point prove inaccurate, including the results of adjunctive molecular tests.
Conclusion
Ancillary methods are emerging as useful tools for the diagnostic evaluation of melanocytic neoplasms that cannot be assigned definitive diagnoses using traditional techniques alone. This study suggests that patients with ambiguous melanocytic neoplasms may benefit from diagnoses and treatment decisions aligned with the results of a gene expression test, and that for those with a benign result, simple observation may be a safe alternative to surgical excision. This expands upon prior observations of the test’s influence on diagnoses and treatment decisions and supports its role as part of dermatopathologists’ and dermatologists’ decision-making process for histopathologically ambiguous melanocytic lesions.
According to National Institutes of Health estimates, more than 90,000 new cases of melanoma were diagnosed in 2018.1 Overall 5-year survival for patients with melanoma exceeds 90%, but individual survival estimates are highly dependent on stage at diagnosis, and survival decreases markedly with metastasis. Therefore, early and accurate diagnosis is critical.
Diagnosis of melanocytic neoplasms usually is performed by dermatopathologists through microscopic examination of stained tissue biopsy sections, a technically simple and effective method that enables a definitive diagnosis of benign nevus or malignant melanoma to be made in most cases. However, approximately 15% of all biopsied melanocytic lesions will exhibit some degree of histopathologic ambiguity,2-4 meaning that some of their microscopic features will be characteristic of a benign nevus while others will suggest the possibility of malignant melanoma. Diagnostic interpretations often vary in these cases, even among experts, and a definitive diagnosis of benign or malignant may be difficult to achieve by microscopy alone.2-4 Because of the marked reduction in survival once a melanoma has metastasized, these diagnostically ambiguous lesions often are treated as possible malignant melanomas with complete surgical excision (or re-excision). However, some experts suggest that many histopathologically ambiguous melanocytic neoplasms are, in fact, benign,5 a notion supported by epidemiologic evidence.6,7 Therefore, excision of many ambiguous melanocytic neoplasms might be avoided if definitive diagnosis could be achieved.
A gene expression signature was developed and validated for use as an adjunct to traditional methods of differentiating malignant melanocytic neoplasms from their benign counterparts.8-11 This test quantifies the RNA transcripts produced by 14 genes known to be overexpressed in malignant melanomas by comparison to benign nevi. These values are then combined algorithmically with measurements of 9 reference genes to produce an objective numerical score that is classified as benign, malignant, or indeterminate. When used by board-certified dermatopathologists and dermatologists confronting ambiguous melanocytic lesions, the test produces substantial increases in definitive diagnoses and prompts changes in treatment recommendations.12,13 However, the long-term consequences of foregoing surgical excision of melanocytic neoplasms that are diagnostically ambiguous but classified as benign by this test have not yet been formally assessed. In the current study, prospectively tested patients whose ambiguous melanocytic neoplasms were classified as benign by the gene expression signature were followed for up to 4.5 years to evaluate the long-term safety of treatment decisions aligned with benign test results.
Methods
Study Population
As part of a prior study,12 US-based dermatopathologists submitted tissue sections from biopsied melanocytic neoplasms determined to be diagnostically ambiguous by histopathology for analysis with the gene expression signature (Myriad Genetics, Inc). Diagnostically ambiguous lesions were those lesions that were described as ambiguous, uncertain, equivocal, indeterminate, or other synonymous terms by the submitting dermatopathologist and therefore lacked a confident diagnosis of benign or malignant prior to testing. Patients initially were tested between May 2014 and August 2014, with samples submitted through a prospective clinical experience study designed to assess the impact of the test on diagnosis and treatment decisions. This study was performed under an institutional review board waiver of consent (Quorum #33403/1).
Patients were eligible for inclusion in the current study if their biopsy specimens (1) had an uncertain preliminary diagnosis according to the submitting dermatopathologist (pretest diagnosis of indeterminate); (2) received a negative (benign) score from the gene expression test; (3) were treated as benign by the dermatologist(s) involved in follow-up care; and (4) were submitted by a single site (St. Joseph Medical Center, Houston, Texas). Although a single dermatopathology site was used for this study, multiple dermatologists were involved in the final treatment of these patients. Patients with benign scores who received additional intervention were excluded, as they may have a lower rate of adverse events (ie, metastasis) than those who did not receive intervention and would therefore skew the analysis population. A total of 25 patients from the prior study met these inclusion criteria. The previously collected12 pretest and posttest de-identified data were compiled from the commercial laboratory databases, and the patients were followed from the time of testing via medical record review performed by the dermatology providers at participating sites. Clinical follow-up data were collected using study-specific case report forms (CRFs) that captured the following: (1) the dates and results of clinical follow-up visits; (2) the type(s) of treatment and interventions (if any) performed at those visits; (3) the specific indication for any intervention performed; (4) any evidence of persistent, locally recurrent, and/or distant melanocytic neoplasia (whether definitively attributable to the tested lesion or not); and (5) death from any cause. The CRF assigned interventions to 1 of 5 categories: excision, excision with sentinel lymph node biopsy, referral to dermatologic or other surgeon, examination only (without surgical intervention), and other. Selection of other required a free-text description of the treatment and indications. Pertinent information not otherwise captured by the CRF also was recordable as free text.
Gene Expression Testing
Gene expression testing was carried out at the time of specimen submission in the prior study12 as described previously.14 Briefly, formalin-fixed, paraffin-embedded, unstained tissue sections and/or tissue blocks were submitted for testing along with a single hematoxylin and eosin–stained slide used to identify and designate the representative portion(s) of the lesion to be tested. These areas were macrodissected from unstained tissue sections and pooled for RNA extraction. Expression of 14 biomarker genes and 9 reference genes was measured via
Statistical Analysis
Demographic and other baseline characteristics of the patient population were summarized. Follow-up time was calculated as the interval between the date a patient’s gene expression test result was first issued to the provider and the date of the patient’s last recorded visit during the study period. All patient dermatology office visits within the designated follow-up period were documented, with a nonstandard number of visits and follow-up time across all study patients. Statistical analyses were conducted using SAS software (SAS Institute Inc), R software version 3.5.0 (R Foundation for Statistical Computing), and IBM SPSS Statistics software (IBM SPSS Statistics for Windows, Version 25).
Results
Patient Sample
A total of 25 ambiguous melanocytic neoplasms from 25 patients met the study inclusion criteria of a benign gene expression result with subsequent treatment as a benign neoplasm during follow-up. The patient sample statistics are summarized in Table 1. Most patients were younger than 65 years, with an average age at the time of biopsy of 48.4 years. All 25 neoplasms produced negative (benign) gene expression signature scores, all were diagnosed as benign nevi posttest by the submitting dermatopathologist, and all patients were initially treated in accordance with the benign diagnosis by the dermatologist(s) involved in clinical follow-up care. Prior to testing with the gene expression signature, most of these histopathologically indeterminate lesions received differential diagnoses, the most common of which were dysplastic nevus (84%), melanoma arising from a nevus (72%), and superficial spreading melanoma (64%; eTable). After testing with the gene expression signature and receiving a benign score, most lesions received a single differential diagnosis of dysplastic nevus (88%).


Follow-up and Survival
Clinical follow-up time ranged from 0.6 to 53.3 months, with a mean duration (SD) of 38.5 (16.6) months, and patients attended an average of 4 postbiopsy dermatology appointments (mean [SD], 4.6 [3.6]). According to the participating dermatology care providers, none of the 25 patients developed any indication during follow-up that the diagnosis of benign nevus was inaccurate. No patient had evidence of locally recurrent or metastatic melanoma, and none died during the study period.
Treatment/Interventions
The treatment recorded in the CRF was examination only for 21 of 25 patients, excision for 3, and other for 1 (Table 2). Because the explanation for the selection of other in this case described an excision performed at the same anatomic location as the biopsy, this treatment also was considered an excision for purposes of the study analyses. The 3 excisions all occurred at the first postbiopsy dermatology encounter. Across all follow-up visits, no additional surgical interventions occurred (Table 2).

The first excision (case 1) involved a 67-year-old woman with a lesion on the mid pubic region described clinically as an atypical nevus that generated a pretest histopathologic differential diagnosis including dysplastic nevus, superficial spreading melanoma, and melanoma arising within a nevus (Table 3; Figure, A and B). The gene expression test result was benign (score, −5.4), and the final pathology report diagnosis was nevus with junctional dysplasia, moderate. Surgical excision was performed at the patient’s first return visit, 505 days after initial diagnosis, with moderately dysplastic nevus as the recorded indication for removal. No repigmentation or other evidence of local recurrence or progression was detected, and the treating dermatologist indicated no suspicion that the original diagnosis of benign nevus was incorrect during the 23-month follow-up period.


The second excision (case 2) involved a 27-year-old woman with a pigmented neoplasm on the mid upper back (Figure, C and D) biopsied to rule out dysplastic nevus that resulted in a pretest histopathologic differential diagnosis of dysplastic nevus vs superficial spreading melanoma or melanoma arising within a nevus. The gene expression test result classified the lesion as benign (score, −2.9), and the final pathology diagnosis was nevus, compound, with moderate dysplasia. Despite the benign diagnosis, residual neoplasm (or pigmentation) at the biopsy site prompted the patient to request excision at her first postbiopsy visit, 22 days after testing (Table 3). The CRF completed by the dermatologist reported no indication that the benign diagnosis was inaccurate, but the patient was subsequently lost to follow-up.
The third excision (case 3) involved a 32-year-old woman with a pigmented lesion on the abdomen (Table 3; Figure, E and F). The clinical description was irregular-appearing black papule, nevus with atypia, and the histopathologic differential diagnosis again included dysplastic nevus, superficial spreading melanoma, and melanoma arising within a preexisting nevus. The gene expression signature result was benign (score, −7.2), and the final diagnosis issued within the accompanying pathology report was nevus with moderate junctional dysplasia. Despite the benign diagnosis, excision was performed 89 days after test result availability, with apparent residual pigmentation as the specified indication. As with the other 2 cases, the treating dermatologist confirmed that neither clinical features nor follow-up events suggested malignancy.
Comment
This study followed a cohort of 25 patients with histopathologically ambiguous melanocytic neoplasms that were classified as benign by a diagnostic gene expression test with the intent of determining the outcomes of patients whose treatment aligned with their benign test result. All patients initially were managed according to their test result. During an average posttest clinical follow-up time of more than 3 years (38.5 months), the 25 biopsied lesions, most of which received a differential diagnosis of dysplastic nevus, were regarded as benign nevi by their dermatologists, and the vast majority (88%) received no further surgical intervention. Three patients underwent subsequent excision of the biopsied lesion, with patient or physician preference as the indication in each instance. None of the 25 patients developed evidence of local recurrence, metastasis, or other findings that prompted doubt of the benign diagnosis. The absence of adverse events during clinical follow-up, particularly given that most lesions were not subjected to further intervention, supports use of the gene expression test as a safe and effective adjunct to the diagnosis and treatment of ambiguous melanocytic neoplasms by dermatologists and dermatopathologists.
Ambiguous melanocytic neoplasms evaluated without the aid of molecular adjuncts often result in equivocal or less-than-definitive diagnoses, and further surgical intervention is commonly undertaken to mitigate against the possibility of a missed melanoma.13 In this study, treatment that was aligned with the benign test result allowed most patients to avoid further surgical intervention, which suggests that adjunctive use of the gene signature can contribute to reductions in the physical and economic burdens imposed by unnecessary surgical interventions.15,16 Moreover, any means of increasing accurate and definitive diagnoses may produce an immediate impact on health outcomes by reducing the anxiety that uncertainty often provokes in patients and health care providers alike.
Study Limitations
This study must be interpreted within the context of its limitations. Obtaining meaningful patient outcome data is a common challenge in health care research due to the requisite length of follow-up and sometimes the lack of definitive evidence of adverse events. This is particularly difficult for melanocytic neoplasms because of an apparent inclination for patients with benign diagnoses to abandon follow-up and an increasing tendency for even minimal diagnostic uncertainty to prompt complete excision. Additionally, the only definitive clinical outcome for melanocytic neoplasms is distant metastasis, which (fortunately for patients) is relatively rare. Not surprisingly, studies documenting clinical outcomes of patients with ambiguous melanocytic neoplasms tested prospectively with diagnostic adjuncts are scarce, and this study’s sample size and clinical follow-up compare favorably with the few that exist.17,18 Although most melanomas declare themselves through recurrence or metastasis within several years of initial biopsy,1,19 some are clinically dormant for as long as 10 years after initial detection.20,21 This may be particularly true for the small or early-stage lesions that now comprise the majority of biopsied neoplasms, and such events would go undetected by this study and many others. It also must be recognized that uneventful follow-up, regardless of duration, cannot prove that a biopsied melanocytic neoplasm was benign. Although only 5 patients had a follow-up time of less than 2 years (the time frame in which most recurrence or metastasis will occur), it cannot be definitively proven that a minimum of 2 years recurrence- or metastasis-free survival indicates a benign lesion. Many early-stage malignant melanomas are eradicated by complete excision or even by the initial biopsy if margins are uninvolved.
Because these limitations are intrinsic to melanocytic neoplasms and current management strategies, they pertain to all investigations seeking insights into biological potential through clinical outcomes. Similarly, all current diagnostic tools and procedures have the potential for sampling error, including histopathology. The rarity of adverse outcomes (recurrence and metastasis) in patients with benign test results within this cohort indicates that false-negative results are uncommon, which is further evidenced by a similar rarity of adverse events in prior studies of the gene expression signature.8-10,22 A particular strength of this study is that most of the ambiguous melanocytic neoplasms followed did not undergo excision after the initial biopsy, an increasingly uncommon situation that may increase their likelihood to be informative.
It must be emphasized that the gene expression test, similar to other diagnostic adjuncts, is neither a replacement for histopathologic interpretation nor a substitute for judgment. As with all tests, it can produce false-positive and false-negative results. Therefore, it should always be interpreted within the constellation of the many other data points that must be considered when making a distinction between benign nevus and malignant melanoma, including but not limited to patient age, family and personal history of melanoma, anatomic location, clinical features, and histopathologic findings. As is the case for many diseases, careful consideration of all relevant input is necessary to minimize the risk of misdiagnosis that might occur should any single data point prove inaccurate, including the results of adjunctive molecular tests.
Conclusion
Ancillary methods are emerging as useful tools for the diagnostic evaluation of melanocytic neoplasms that cannot be assigned definitive diagnoses using traditional techniques alone. This study suggests that patients with ambiguous melanocytic neoplasms may benefit from diagnoses and treatment decisions aligned with the results of a gene expression test, and that for those with a benign result, simple observation may be a safe alternative to surgical excision. This expands upon prior observations of the test’s influence on diagnoses and treatment decisions and supports its role as part of dermatopathologists’ and dermatologists’ decision-making process for histopathologically ambiguous melanocytic lesions.
- Noone AM, Howlander N, Krapcho M, et al, eds. SEER Cancer Statistics Review, 1975-2015. National Cancer Institute website. Updated September 10, 2018. Accessed April 21, 2021. https://seer.cancer.gov/archive/csr/1975_2015/
- Shoo BA, Sagebiel RW, Kashani-Sabet M. Discordance in the histopathologic diagnosis of melanoma at a melanoma referral center. J Am Acad Dermatol. 2010;62:751-756.
- Veenhuizen KC, De Wit PE, Mooi WJ, et al. Quality assessment by expert opinion in melanoma pathology: experience of the pathology panel of the Dutch Melanoma Working Party. J Pathol. 1997;182:266-272.
- Elmore JG, Barnhill RL, Elder DE, et al. Pathologists’ diagnosis of invasive melanoma and melanocytic proliferations: observer accuracy and reproducibility study. BMJ. 2017;357:j2813. doi:10.1136/bmj.j2813
- Glusac EJ. The melanoma ‘epidemic’, a dermatopathologist’s perspective. J Cutan Pathol. 2011;38:264-267.
- Welch HG, Woloshin S, Schwartz LM. Skin biopsy rates and incidence of melanoma: population based ecological study. BMJ. 2005;331:481.
- Swerlick RA, Chen S. The melanoma epidemic. Is increased surveillance the solution or the problem? Arch Dermatol. 1996;132:881-884.
- Ko JS, Matharoo-Ball B, Billings SD, et al. Diagnostic distinction of malignant melanoma and benign nevi by a gene expression signature and correlation to clinical outcomes. Cancer Epidemiol Biomarkers Prev. 2017;26:1107-1113.
- Clarke LE, Flake DD 2nd, Busam K, et al. An independent validation of a gene expression signature to differentiate malignant melanoma from benign melanocytic nevi. Cancer. 2017;123:617-628.
- Clarke LE, Warf BM, Flake DD 2nd, et al. Clinical validation of a gene expression signature that differentiates benign nevi from malignant melanoma. J Cutan Pathol. 2015;42:244-252.
- Minca EC, Al-Rohil RN, Wang M, et al. Comparison between melanoma gene expression score and fluorescence in situ hybridization for the classification of melanocytic lesions. Mod Pathol. 2016;29:832-843.
- Cockerell CJ, Tschen J, Evans B, et al. The influence of a gene expression signature on the diagnosis and recommended treatment of melanocytic tumors by dermatopathologists. Medicine (Baltimore). 2016;95:e4887. doi:10.1097/MD.0000000000004887
- Cockerell C, Tschen J, Billings SD, et al. The influence of a gene-expression signature on the treatment of diagnostically challenging melanocytic lesions. Per Med. 2017;14:123-130.
- Warf MB, Flake DD 2nd, Adams D, et al. Analytical validation of a melanoma diagnostic gene signature using formalin-fixed paraffin-embedded melanocytic lesions. Biomark Med. 2015;9:407-416.
- Guy GP Jr, Ekwueme DU, Tangka FK, et al. Melanoma treatment costs: a systematic review of the literature, 1990-2011. Am J Prev Med. 2012;43:537-545.
- Guy GP Jr, Machlin SR, Ekwueme DU, et al. Prevalence and costs of skin cancer treatment in the U.S., 2002-2006 and 2007-2011. Am J Prev Med. 2015;48:183-187.
- Egnatios GL, Ferringer TC. Clinical follow-up of atypical spitzoid tumors analyzed by fluorescence in situ hybridization. Am J Dermatopathol. 2016;38:289-296.
- Fischer AS, High WA. The difficulty in interpreting gene expression profiling in BAP-negative melanocytic tumors. J Cutan Pathol. 2018;45:659-666. doi:10.1111/cup.13277
- Vollmer RT. The dynamics of death in melanoma. J Cutan Pathol. 2012;39:1075-1082.
- Osella-Abate S, Ribero S, Sanlorenzo M, et al. Risk factors related to late metastases in 1,372 melanoma patients disease free more than 10 years. Int J Cancer. 2015;136:2453-2457.
- Faries MB, Steen S, Ye X, et al. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg. 2013;217:27-34.
- Ko JS, Clarke LE, Minca EC, et al. Correlation of melanoma gene expression score with clinical outcomes on a series of melanocytic lesions. Hum Pathol. 2019;86:213-221.
- Noone AM, Howlander N, Krapcho M, et al, eds. SEER Cancer Statistics Review, 1975-2015. National Cancer Institute website. Updated September 10, 2018. Accessed April 21, 2021. https://seer.cancer.gov/archive/csr/1975_2015/
- Shoo BA, Sagebiel RW, Kashani-Sabet M. Discordance in the histopathologic diagnosis of melanoma at a melanoma referral center. J Am Acad Dermatol. 2010;62:751-756.
- Veenhuizen KC, De Wit PE, Mooi WJ, et al. Quality assessment by expert opinion in melanoma pathology: experience of the pathology panel of the Dutch Melanoma Working Party. J Pathol. 1997;182:266-272.
- Elmore JG, Barnhill RL, Elder DE, et al. Pathologists’ diagnosis of invasive melanoma and melanocytic proliferations: observer accuracy and reproducibility study. BMJ. 2017;357:j2813. doi:10.1136/bmj.j2813
- Glusac EJ. The melanoma ‘epidemic’, a dermatopathologist’s perspective. J Cutan Pathol. 2011;38:264-267.
- Welch HG, Woloshin S, Schwartz LM. Skin biopsy rates and incidence of melanoma: population based ecological study. BMJ. 2005;331:481.
- Swerlick RA, Chen S. The melanoma epidemic. Is increased surveillance the solution or the problem? Arch Dermatol. 1996;132:881-884.
- Ko JS, Matharoo-Ball B, Billings SD, et al. Diagnostic distinction of malignant melanoma and benign nevi by a gene expression signature and correlation to clinical outcomes. Cancer Epidemiol Biomarkers Prev. 2017;26:1107-1113.
- Clarke LE, Flake DD 2nd, Busam K, et al. An independent validation of a gene expression signature to differentiate malignant melanoma from benign melanocytic nevi. Cancer. 2017;123:617-628.
- Clarke LE, Warf BM, Flake DD 2nd, et al. Clinical validation of a gene expression signature that differentiates benign nevi from malignant melanoma. J Cutan Pathol. 2015;42:244-252.
- Minca EC, Al-Rohil RN, Wang M, et al. Comparison between melanoma gene expression score and fluorescence in situ hybridization for the classification of melanocytic lesions. Mod Pathol. 2016;29:832-843.
- Cockerell CJ, Tschen J, Evans B, et al. The influence of a gene expression signature on the diagnosis and recommended treatment of melanocytic tumors by dermatopathologists. Medicine (Baltimore). 2016;95:e4887. doi:10.1097/MD.0000000000004887
- Cockerell C, Tschen J, Billings SD, et al. The influence of a gene-expression signature on the treatment of diagnostically challenging melanocytic lesions. Per Med. 2017;14:123-130.
- Warf MB, Flake DD 2nd, Adams D, et al. Analytical validation of a melanoma diagnostic gene signature using formalin-fixed paraffin-embedded melanocytic lesions. Biomark Med. 2015;9:407-416.
- Guy GP Jr, Ekwueme DU, Tangka FK, et al. Melanoma treatment costs: a systematic review of the literature, 1990-2011. Am J Prev Med. 2012;43:537-545.
- Guy GP Jr, Machlin SR, Ekwueme DU, et al. Prevalence and costs of skin cancer treatment in the U.S., 2002-2006 and 2007-2011. Am J Prev Med. 2015;48:183-187.
- Egnatios GL, Ferringer TC. Clinical follow-up of atypical spitzoid tumors analyzed by fluorescence in situ hybridization. Am J Dermatopathol. 2016;38:289-296.
- Fischer AS, High WA. The difficulty in interpreting gene expression profiling in BAP-negative melanocytic tumors. J Cutan Pathol. 2018;45:659-666. doi:10.1111/cup.13277
- Vollmer RT. The dynamics of death in melanoma. J Cutan Pathol. 2012;39:1075-1082.
- Osella-Abate S, Ribero S, Sanlorenzo M, et al. Risk factors related to late metastases in 1,372 melanoma patients disease free more than 10 years. Int J Cancer. 2015;136:2453-2457.
- Faries MB, Steen S, Ye X, et al. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg. 2013;217:27-34.
- Ko JS, Clarke LE, Minca EC, et al. Correlation of melanoma gene expression score with clinical outcomes on a series of melanocytic lesions. Hum Pathol. 2019;86:213-221.
Practice Point
- Implementation of a gene expression signature in the diagnosis of histopathologically ambiguous lesions can safely increase diagnostic accuracy and optimize treatment.
Mycosis Fungoides in Black Patients: Time for a Better Look
Recent advances in the immunopathogenesis and therapy of cutaneous T-cell lymphoma (CTCL) have shown great promise for the care of patients with mycosis fungoides (MF) and Sézary syndrome (SS).1-3 Research into the tumor microenvironment, microbiome, and molecular genetics may yield further information as we strive to develop MF/SS therapy from the bench to the bedside.3 Although progress has been made on multiple fronts in MF, some important—particularly epidemiologic and clinical—questions remain unanswered.
Racial disparities are well known to exist in CTCLs, particularly MF and SS.4-7 The incidence of MF and SS in the United States is higher in African American/Black patients than in White patients4; in addition, MF has an earlier age at onset in Black patients compared with White patients.4,5 Gender disparities also exist, with relatively more Black females than males affected with MF4-6; in particular, early-onset MF (ie, <40 years of age) is more common in Black females than Black males.6,7 According to Surveillance, Epidemiology, and End Results (SEER) data4 and the US National Cancer Database,5 African American/Black patients with MF have worse outcomes compared with other races (shorter overall survival and higher mortality) and also exhibit higher stages of disease at presentation (stage IIb or higher).5 Black race also was found to be a predictor of poor overall survival after accounting for disease characteristics, socioeconomicfactors, and types of treatment. The factors responsible for these racial disparities remain unclear.
A fortuitous collision of interests and technology may have helped to shed light on some of the reasons for these racial disparities in MF. Nearly 2 decades ago, high-quality, whole-body digital cutaneous photography was implemented by the Dermatology Service at Memorial Sloan Kettering Cancer Center Dermatology Service (New York, New York).8 Although the standardized 20-pose positioning images initially were used for the follow-up evaluation of patients with multiple nevi and melanomas, we incorporated the same photography technique into our multidisciplinary Cutaneous Lymphoma Clinic at Memorial Sloan Kettering Cancer Center. The multiplicity and clinical heterogeneity of MF lesions is well known, as is the fact that individual MF lesions may develop, respond to therapy, or change independently of other lesions in a given patient. We regularly reviewed these digital images with patients during their visits to assess treatment responses, discussed the need for changes in therapy in the face of progressive disease, and provided encouragement and positive reinforcement for those who improved with time-consuming regimens (eg, phototherapy).
Ultimately, as we became more familiar with looking at images in skin of color, we recognized different clinical features among our Black patients. In the literature, hypopigmented MF is a variant that typically is characterized by CD8+-predominant T cells and is seen more frequently in dark-skinned patients.9 In contrast, hyperpigmented MF has been considered a relatively rare presentation of MF.10 However, using only clinical and demographic information, we were able to identify 2 very different prognostic groups: those with hypopigmented lesions and those with only hyperpigmented and/or erythematous skin lesions.11 In our retrospective review of 157 African American/Black MF patients at our institution—122 with early-stage and 35 with late-stage MF—45% of patients had hypopigmented lesions vs 52% with hyperpigmented and/or erythematous lesions but no hypopigmentation. Those with hypopigmentation had superior outcomes, with better overall survival (P=.002) and progression-free survival (P=.014). In addition, more than 80% of patients who progressed or died from disease had hyperpigmented and/or erythematous lesions without hypopigmentation.11
Sometimes we have to go backward to go forward. Going from the bedside to the bench in our Black MF/SS patients—initially through the clinical recognition of prognostically different lesions, and then through clinicopathologic correlation with immunophenotyping and molecular studies—should provide important clues. Further investigation of Black patients who share similar pigmentary phenotypes of MF also may shed light on the pathogenetic mechanisms responsible for these prognostically significant skin findings. Through these efforts, we hope to identify higher-risk patients, which ultimately will lead to earlier intervention, more effective therapeutic regimens, and improved outcomes.
- Durgin JS, Weiner DM, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: pathways and targets for immune restoration and tumor eradication. J Am Acad Dermatol. 2021;84:587-595.
- Weiner DM, Durgin JS, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: current and future approaches. J Am Acad Dermatol. 2021;84:597-604.
- Quaglino P, Fava P, Pileri A, et al. Phenotypical markers, molecular mutations, and immune microenvironment as targets for new treatments in patients with mycosis fungoides and/or Sézary syndrome. J Invest Dermatol. 2021;141:484-495.
- Nath SK, Yu JB, Wilson LD. Poorer prognosis of African-American patients with mycosis fungoides: an analysis of the SEER dataset, 1988 to 2008. Clin Lymphoma Myeloma Leuk. 2014;14:419-423.
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.
- Balagula Y, Dusza SW, Zampella J, et al. Early-onset mycosis fungoides among African American women: a single-institution study. J Am Acad Dermatol. 2014;71:597-598.
- Virmani P, Levin L, Myskowski PL, et al. Clinical outcome and prognosis of young patients with mycosis fungoides. Pediatr Dermatol. 2017;34:547-553.
- Halpern AC, Marghoob AA, Bialoglow TW, et al. Standardized positioning of patients (poses) for whole body cutaneous photography. J Am Acad Dermatol. 2003;49:593-598.
- Rodney IJ, Kindred C, Angra K, et al. Hypopigmented mycosis fungoides: a retrospective clinicohistopathologic study. J Eur Acad Dermatol Venereol. 2017;31:808-814.
- Kondo M, Igawa K, Munetsugu T, et al. Increasing numbers of mast cells in skin lesions of hyperpigmented mycosis fungoides with large-cell transformation. Ann Dermatol. 2016;28:115-116.
- Geller S, Lebowitz E, Pulitzer MP, et al. Outcomes and prognostic factors in African American and Black patients with mycosis fungoides/Sézary syndrome: retrospective analysis of 157 patients from a referral cancer center. J Am Acad Dermatol. 2020;83:430-439.
Recent advances in the immunopathogenesis and therapy of cutaneous T-cell lymphoma (CTCL) have shown great promise for the care of patients with mycosis fungoides (MF) and Sézary syndrome (SS).1-3 Research into the tumor microenvironment, microbiome, and molecular genetics may yield further information as we strive to develop MF/SS therapy from the bench to the bedside.3 Although progress has been made on multiple fronts in MF, some important—particularly epidemiologic and clinical—questions remain unanswered.
Racial disparities are well known to exist in CTCLs, particularly MF and SS.4-7 The incidence of MF and SS in the United States is higher in African American/Black patients than in White patients4; in addition, MF has an earlier age at onset in Black patients compared with White patients.4,5 Gender disparities also exist, with relatively more Black females than males affected with MF4-6; in particular, early-onset MF (ie, <40 years of age) is more common in Black females than Black males.6,7 According to Surveillance, Epidemiology, and End Results (SEER) data4 and the US National Cancer Database,5 African American/Black patients with MF have worse outcomes compared with other races (shorter overall survival and higher mortality) and also exhibit higher stages of disease at presentation (stage IIb or higher).5 Black race also was found to be a predictor of poor overall survival after accounting for disease characteristics, socioeconomicfactors, and types of treatment. The factors responsible for these racial disparities remain unclear.
A fortuitous collision of interests and technology may have helped to shed light on some of the reasons for these racial disparities in MF. Nearly 2 decades ago, high-quality, whole-body digital cutaneous photography was implemented by the Dermatology Service at Memorial Sloan Kettering Cancer Center Dermatology Service (New York, New York).8 Although the standardized 20-pose positioning images initially were used for the follow-up evaluation of patients with multiple nevi and melanomas, we incorporated the same photography technique into our multidisciplinary Cutaneous Lymphoma Clinic at Memorial Sloan Kettering Cancer Center. The multiplicity and clinical heterogeneity of MF lesions is well known, as is the fact that individual MF lesions may develop, respond to therapy, or change independently of other lesions in a given patient. We regularly reviewed these digital images with patients during their visits to assess treatment responses, discussed the need for changes in therapy in the face of progressive disease, and provided encouragement and positive reinforcement for those who improved with time-consuming regimens (eg, phototherapy).
Ultimately, as we became more familiar with looking at images in skin of color, we recognized different clinical features among our Black patients. In the literature, hypopigmented MF is a variant that typically is characterized by CD8+-predominant T cells and is seen more frequently in dark-skinned patients.9 In contrast, hyperpigmented MF has been considered a relatively rare presentation of MF.10 However, using only clinical and demographic information, we were able to identify 2 very different prognostic groups: those with hypopigmented lesions and those with only hyperpigmented and/or erythematous skin lesions.11 In our retrospective review of 157 African American/Black MF patients at our institution—122 with early-stage and 35 with late-stage MF—45% of patients had hypopigmented lesions vs 52% with hyperpigmented and/or erythematous lesions but no hypopigmentation. Those with hypopigmentation had superior outcomes, with better overall survival (P=.002) and progression-free survival (P=.014). In addition, more than 80% of patients who progressed or died from disease had hyperpigmented and/or erythematous lesions without hypopigmentation.11
Sometimes we have to go backward to go forward. Going from the bedside to the bench in our Black MF/SS patients—initially through the clinical recognition of prognostically different lesions, and then through clinicopathologic correlation with immunophenotyping and molecular studies—should provide important clues. Further investigation of Black patients who share similar pigmentary phenotypes of MF also may shed light on the pathogenetic mechanisms responsible for these prognostically significant skin findings. Through these efforts, we hope to identify higher-risk patients, which ultimately will lead to earlier intervention, more effective therapeutic regimens, and improved outcomes.
Recent advances in the immunopathogenesis and therapy of cutaneous T-cell lymphoma (CTCL) have shown great promise for the care of patients with mycosis fungoides (MF) and Sézary syndrome (SS).1-3 Research into the tumor microenvironment, microbiome, and molecular genetics may yield further information as we strive to develop MF/SS therapy from the bench to the bedside.3 Although progress has been made on multiple fronts in MF, some important—particularly epidemiologic and clinical—questions remain unanswered.
Racial disparities are well known to exist in CTCLs, particularly MF and SS.4-7 The incidence of MF and SS in the United States is higher in African American/Black patients than in White patients4; in addition, MF has an earlier age at onset in Black patients compared with White patients.4,5 Gender disparities also exist, with relatively more Black females than males affected with MF4-6; in particular, early-onset MF (ie, <40 years of age) is more common in Black females than Black males.6,7 According to Surveillance, Epidemiology, and End Results (SEER) data4 and the US National Cancer Database,5 African American/Black patients with MF have worse outcomes compared with other races (shorter overall survival and higher mortality) and also exhibit higher stages of disease at presentation (stage IIb or higher).5 Black race also was found to be a predictor of poor overall survival after accounting for disease characteristics, socioeconomicfactors, and types of treatment. The factors responsible for these racial disparities remain unclear.
A fortuitous collision of interests and technology may have helped to shed light on some of the reasons for these racial disparities in MF. Nearly 2 decades ago, high-quality, whole-body digital cutaneous photography was implemented by the Dermatology Service at Memorial Sloan Kettering Cancer Center Dermatology Service (New York, New York).8 Although the standardized 20-pose positioning images initially were used for the follow-up evaluation of patients with multiple nevi and melanomas, we incorporated the same photography technique into our multidisciplinary Cutaneous Lymphoma Clinic at Memorial Sloan Kettering Cancer Center. The multiplicity and clinical heterogeneity of MF lesions is well known, as is the fact that individual MF lesions may develop, respond to therapy, or change independently of other lesions in a given patient. We regularly reviewed these digital images with patients during their visits to assess treatment responses, discussed the need for changes in therapy in the face of progressive disease, and provided encouragement and positive reinforcement for those who improved with time-consuming regimens (eg, phototherapy).
Ultimately, as we became more familiar with looking at images in skin of color, we recognized different clinical features among our Black patients. In the literature, hypopigmented MF is a variant that typically is characterized by CD8+-predominant T cells and is seen more frequently in dark-skinned patients.9 In contrast, hyperpigmented MF has been considered a relatively rare presentation of MF.10 However, using only clinical and demographic information, we were able to identify 2 very different prognostic groups: those with hypopigmented lesions and those with only hyperpigmented and/or erythematous skin lesions.11 In our retrospective review of 157 African American/Black MF patients at our institution—122 with early-stage and 35 with late-stage MF—45% of patients had hypopigmented lesions vs 52% with hyperpigmented and/or erythematous lesions but no hypopigmentation. Those with hypopigmentation had superior outcomes, with better overall survival (P=.002) and progression-free survival (P=.014). In addition, more than 80% of patients who progressed or died from disease had hyperpigmented and/or erythematous lesions without hypopigmentation.11
Sometimes we have to go backward to go forward. Going from the bedside to the bench in our Black MF/SS patients—initially through the clinical recognition of prognostically different lesions, and then through clinicopathologic correlation with immunophenotyping and molecular studies—should provide important clues. Further investigation of Black patients who share similar pigmentary phenotypes of MF also may shed light on the pathogenetic mechanisms responsible for these prognostically significant skin findings. Through these efforts, we hope to identify higher-risk patients, which ultimately will lead to earlier intervention, more effective therapeutic regimens, and improved outcomes.
- Durgin JS, Weiner DM, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: pathways and targets for immune restoration and tumor eradication. J Am Acad Dermatol. 2021;84:587-595.
- Weiner DM, Durgin JS, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: current and future approaches. J Am Acad Dermatol. 2021;84:597-604.
- Quaglino P, Fava P, Pileri A, et al. Phenotypical markers, molecular mutations, and immune microenvironment as targets for new treatments in patients with mycosis fungoides and/or Sézary syndrome. J Invest Dermatol. 2021;141:484-495.
- Nath SK, Yu JB, Wilson LD. Poorer prognosis of African-American patients with mycosis fungoides: an analysis of the SEER dataset, 1988 to 2008. Clin Lymphoma Myeloma Leuk. 2014;14:419-423.
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.
- Balagula Y, Dusza SW, Zampella J, et al. Early-onset mycosis fungoides among African American women: a single-institution study. J Am Acad Dermatol. 2014;71:597-598.
- Virmani P, Levin L, Myskowski PL, et al. Clinical outcome and prognosis of young patients with mycosis fungoides. Pediatr Dermatol. 2017;34:547-553.
- Halpern AC, Marghoob AA, Bialoglow TW, et al. Standardized positioning of patients (poses) for whole body cutaneous photography. J Am Acad Dermatol. 2003;49:593-598.
- Rodney IJ, Kindred C, Angra K, et al. Hypopigmented mycosis fungoides: a retrospective clinicohistopathologic study. J Eur Acad Dermatol Venereol. 2017;31:808-814.
- Kondo M, Igawa K, Munetsugu T, et al. Increasing numbers of mast cells in skin lesions of hyperpigmented mycosis fungoides with large-cell transformation. Ann Dermatol. 2016;28:115-116.
- Geller S, Lebowitz E, Pulitzer MP, et al. Outcomes and prognostic factors in African American and Black patients with mycosis fungoides/Sézary syndrome: retrospective analysis of 157 patients from a referral cancer center. J Am Acad Dermatol. 2020;83:430-439.
- Durgin JS, Weiner DM, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: pathways and targets for immune restoration and tumor eradication. J Am Acad Dermatol. 2021;84:587-595.
- Weiner DM, Durgin JS, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: current and future approaches. J Am Acad Dermatol. 2021;84:597-604.
- Quaglino P, Fava P, Pileri A, et al. Phenotypical markers, molecular mutations, and immune microenvironment as targets for new treatments in patients with mycosis fungoides and/or Sézary syndrome. J Invest Dermatol. 2021;141:484-495.
- Nath SK, Yu JB, Wilson LD. Poorer prognosis of African-American patients with mycosis fungoides: an analysis of the SEER dataset, 1988 to 2008. Clin Lymphoma Myeloma Leuk. 2014;14:419-423.
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.
- Balagula Y, Dusza SW, Zampella J, et al. Early-onset mycosis fungoides among African American women: a single-institution study. J Am Acad Dermatol. 2014;71:597-598.
- Virmani P, Levin L, Myskowski PL, et al. Clinical outcome and prognosis of young patients with mycosis fungoides. Pediatr Dermatol. 2017;34:547-553.
- Halpern AC, Marghoob AA, Bialoglow TW, et al. Standardized positioning of patients (poses) for whole body cutaneous photography. J Am Acad Dermatol. 2003;49:593-598.
- Rodney IJ, Kindred C, Angra K, et al. Hypopigmented mycosis fungoides: a retrospective clinicohistopathologic study. J Eur Acad Dermatol Venereol. 2017;31:808-814.
- Kondo M, Igawa K, Munetsugu T, et al. Increasing numbers of mast cells in skin lesions of hyperpigmented mycosis fungoides with large-cell transformation. Ann Dermatol. 2016;28:115-116.
- Geller S, Lebowitz E, Pulitzer MP, et al. Outcomes and prognostic factors in African American and Black patients with mycosis fungoides/Sézary syndrome: retrospective analysis of 157 patients from a referral cancer center. J Am Acad Dermatol. 2020;83:430-439.