User login
Prophylactic NPWT may not improve complication rate after gynecologic surgery
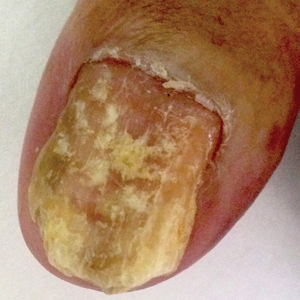
Use of prophylactic negative pressure wound therapy may not be appropriate in surgical cases where women undergo a laparotomy for presumed gynecologic malignancy, according to recent research in Obstetrics & Gynecology.
“The results of our randomized trial do not support the routine use of prophylactic negative pressure wound therapy at the time of laparotomy incision closure in women who are undergoing surgery for gynecologic malignancies or in morbidly obese women who are undergoing laparotomy for benign indications,” Mario M. Leitao Jr., MD, of Memorial Sloan Kettering Cancer Center, New York, and colleagues wrote.
Dr. Leitao and colleagues randomized 663 patients, stratified by body mass index (BMI) after skin closure, to receive negative pressure wound therapy (NPWT) or standard gauze after undergoing a laparotomy for gynecological surgery between March 2016 and August 2019. Patients in the study were aged a median 61 years with a median BMI of 26 kg/m2, but 32 patients with a BMI of 40 or higher who underwent a laparotomy for gynecologic surgery regardless of indication were also included in the study. Most women (80%-82%) were undergoing surgery to treat ovary, fallopian tube, or peritoneal cancer. The most common medical comorbidities in both groups were hypertension (34%-35%) and diabetes (8%-14%). Information on race of patients was not included in the baseline characteristics for the study.
In total, 505 patients were available for evaluation after surgery, which consisted of 254 patients in the NPWT group and 251 patients in the standard gauze group, with 495 patients (98%) having a malignant indication. The researchers examined the incidence of wound complication up to 30 days after surgery.
The results showed a similar rate of wound complications in the NPWT group (44 patients; 17.3%), compared with the group receiving standard gauze (41 patients; 16.3%), with an absolute risk difference between groups of 1% (90% confidence interval, –4.5 to 6.5%; P = .77). Nearly all patients who developed wound complications in both NPWT (92%) and standard gauze (95%) groups had the wound complication diagnosis occur after discharge from the hospital. Dr. Leitao and colleagues noted similarities between groups with regard to wound complications, with most patients having grade 1 complications, and said there were no instances of patients requiring surgery for complications. Among patients in the NPWT group, 33 patients developed skin blistering, compared with 3 patients in the standard gauze group (13% vs. 1.2%; P < .001). After an interim analysis consisting of 444 patients, the study was halted because of “low probability of showing a difference between the two groups at the end of the study.”
The analysis of patients with a BMI of 40 or higher showed 7 of 15 patients (47%) developed wound complications in the NPWT group and 6 of 17 patients (35%) in the standard gauze group (P = .51). In post hoc analyses, the researchers found a median BMI of 26 (range, 17-60) was significantly associated with not developing a wound complication, compared with a BMI of 32 (range, 17-56) (P < .001), and that 41% of patients with a BMI of at least 40 experienced wound complications, compared with 15% of patients with a BMI of less than 40 (P < .001). There was an independent association between developing a wound complication and increasing BMI, according to a multivariate analysis (adjusted odds ratio, 1.10; 95% CI, 1.06-1.14).
Applicability of results unclear for patients with higher BMI
Sarah M. Temkin, MD, a gynecologic oncologist who was not involved with the study, said in an interview that the results by Dr. Leitao and colleagues answer the question of whether patients undergoing surgery for gynecologic malignancy require NPWT, but raised questions about patient selection in the study.
“I think it’s hard to take data from this type of high-end surgical practice and apply it to the general population,” she said, who noted the median BMI of 26 for patients included in the study. A study that included only patients with a BMI of 40 or higher “would have made these results more applicable.”
The low rate of wound complications in the study could potentially be explained by patient selection, Dr. Temkin explained. She cited her own retrospective study from 2016 that showed a wound complication rate of 27.3% for patients receiving prophylactic NPWT where the BMI for the group was 41.29, compared with a complication rate of 19.7% for patients receiving standard care who had a BMI of 30.67.
“It’s hard to cross-trial compare, but that’s significantly higher than what they saw in this prospective study, and I would say that’s a difference with the patient population,” she said. “I think the question of how to reduce surgical-site infections and wound complications in the heavy patient with comorbidities is still unanswered.”
The question is important because patients with a higher BMI and medical comorbidities “still need cancer surgery and methods to reduce the morbidity of that surgery,” Dr. Temkin said. “I think this is an unmet need.”
This study was funded in part by a support grant from the National Institutes of Health/National Cancer Institute Cancer Center, and KCI/Acelity provided part of the study protocol. Nine authors reported personal and institutional relationships in the form of personal fees, grants, stock ownership, consultancies, and speaker’s bureau positions with AstraZeneca, Biom’Up, Bovie Medical, C Surgeries, CMR, ConMed, Covidien, Ethicon, GlaxoSmithKline, GRAIL, Intuitive Surgical, JNJ, Medtronic, Merck, Mylan, Olympus, Stryker/Novadaq, TransEnterix, UpToDate, and Verthermia. Dr. Temkin reported no relevant financial disclosures.
Use of prophylactic negative pressure wound therapy may not be appropriate in surgical cases where women undergo a laparotomy for presumed gynecologic malignancy, according to recent research in Obstetrics & Gynecology.
“The results of our randomized trial do not support the routine use of prophylactic negative pressure wound therapy at the time of laparotomy incision closure in women who are undergoing surgery for gynecologic malignancies or in morbidly obese women who are undergoing laparotomy for benign indications,” Mario M. Leitao Jr., MD, of Memorial Sloan Kettering Cancer Center, New York, and colleagues wrote.
Dr. Leitao and colleagues randomized 663 patients, stratified by body mass index (BMI) after skin closure, to receive negative pressure wound therapy (NPWT) or standard gauze after undergoing a laparotomy for gynecological surgery between March 2016 and August 2019. Patients in the study were aged a median 61 years with a median BMI of 26 kg/m2, but 32 patients with a BMI of 40 or higher who underwent a laparotomy for gynecologic surgery regardless of indication were also included in the study. Most women (80%-82%) were undergoing surgery to treat ovary, fallopian tube, or peritoneal cancer. The most common medical comorbidities in both groups were hypertension (34%-35%) and diabetes (8%-14%). Information on race of patients was not included in the baseline characteristics for the study.
In total, 505 patients were available for evaluation after surgery, which consisted of 254 patients in the NPWT group and 251 patients in the standard gauze group, with 495 patients (98%) having a malignant indication. The researchers examined the incidence of wound complication up to 30 days after surgery.
The results showed a similar rate of wound complications in the NPWT group (44 patients; 17.3%), compared with the group receiving standard gauze (41 patients; 16.3%), with an absolute risk difference between groups of 1% (90% confidence interval, –4.5 to 6.5%; P = .77). Nearly all patients who developed wound complications in both NPWT (92%) and standard gauze (95%) groups had the wound complication diagnosis occur after discharge from the hospital. Dr. Leitao and colleagues noted similarities between groups with regard to wound complications, with most patients having grade 1 complications, and said there were no instances of patients requiring surgery for complications. Among patients in the NPWT group, 33 patients developed skin blistering, compared with 3 patients in the standard gauze group (13% vs. 1.2%; P < .001). After an interim analysis consisting of 444 patients, the study was halted because of “low probability of showing a difference between the two groups at the end of the study.”
The analysis of patients with a BMI of 40 or higher showed 7 of 15 patients (47%) developed wound complications in the NPWT group and 6 of 17 patients (35%) in the standard gauze group (P = .51). In post hoc analyses, the researchers found a median BMI of 26 (range, 17-60) was significantly associated with not developing a wound complication, compared with a BMI of 32 (range, 17-56) (P < .001), and that 41% of patients with a BMI of at least 40 experienced wound complications, compared with 15% of patients with a BMI of less than 40 (P < .001). There was an independent association between developing a wound complication and increasing BMI, according to a multivariate analysis (adjusted odds ratio, 1.10; 95% CI, 1.06-1.14).
Applicability of results unclear for patients with higher BMI
Sarah M. Temkin, MD, a gynecologic oncologist who was not involved with the study, said in an interview that the results by Dr. Leitao and colleagues answer the question of whether patients undergoing surgery for gynecologic malignancy require NPWT, but raised questions about patient selection in the study.
“I think it’s hard to take data from this type of high-end surgical practice and apply it to the general population,” she said, who noted the median BMI of 26 for patients included in the study. A study that included only patients with a BMI of 40 or higher “would have made these results more applicable.”
The low rate of wound complications in the study could potentially be explained by patient selection, Dr. Temkin explained. She cited her own retrospective study from 2016 that showed a wound complication rate of 27.3% for patients receiving prophylactic NPWT where the BMI for the group was 41.29, compared with a complication rate of 19.7% for patients receiving standard care who had a BMI of 30.67.
“It’s hard to cross-trial compare, but that’s significantly higher than what they saw in this prospective study, and I would say that’s a difference with the patient population,” she said. “I think the question of how to reduce surgical-site infections and wound complications in the heavy patient with comorbidities is still unanswered.”
The question is important because patients with a higher BMI and medical comorbidities “still need cancer surgery and methods to reduce the morbidity of that surgery,” Dr. Temkin said. “I think this is an unmet need.”
This study was funded in part by a support grant from the National Institutes of Health/National Cancer Institute Cancer Center, and KCI/Acelity provided part of the study protocol. Nine authors reported personal and institutional relationships in the form of personal fees, grants, stock ownership, consultancies, and speaker’s bureau positions with AstraZeneca, Biom’Up, Bovie Medical, C Surgeries, CMR, ConMed, Covidien, Ethicon, GlaxoSmithKline, GRAIL, Intuitive Surgical, JNJ, Medtronic, Merck, Mylan, Olympus, Stryker/Novadaq, TransEnterix, UpToDate, and Verthermia. Dr. Temkin reported no relevant financial disclosures.
Use of prophylactic negative pressure wound therapy may not be appropriate in surgical cases where women undergo a laparotomy for presumed gynecologic malignancy, according to recent research in Obstetrics & Gynecology.
“The results of our randomized trial do not support the routine use of prophylactic negative pressure wound therapy at the time of laparotomy incision closure in women who are undergoing surgery for gynecologic malignancies or in morbidly obese women who are undergoing laparotomy for benign indications,” Mario M. Leitao Jr., MD, of Memorial Sloan Kettering Cancer Center, New York, and colleagues wrote.
Dr. Leitao and colleagues randomized 663 patients, stratified by body mass index (BMI) after skin closure, to receive negative pressure wound therapy (NPWT) or standard gauze after undergoing a laparotomy for gynecological surgery between March 2016 and August 2019. Patients in the study were aged a median 61 years with a median BMI of 26 kg/m2, but 32 patients with a BMI of 40 or higher who underwent a laparotomy for gynecologic surgery regardless of indication were also included in the study. Most women (80%-82%) were undergoing surgery to treat ovary, fallopian tube, or peritoneal cancer. The most common medical comorbidities in both groups were hypertension (34%-35%) and diabetes (8%-14%). Information on race of patients was not included in the baseline characteristics for the study.
In total, 505 patients were available for evaluation after surgery, which consisted of 254 patients in the NPWT group and 251 patients in the standard gauze group, with 495 patients (98%) having a malignant indication. The researchers examined the incidence of wound complication up to 30 days after surgery.
The results showed a similar rate of wound complications in the NPWT group (44 patients; 17.3%), compared with the group receiving standard gauze (41 patients; 16.3%), with an absolute risk difference between groups of 1% (90% confidence interval, –4.5 to 6.5%; P = .77). Nearly all patients who developed wound complications in both NPWT (92%) and standard gauze (95%) groups had the wound complication diagnosis occur after discharge from the hospital. Dr. Leitao and colleagues noted similarities between groups with regard to wound complications, with most patients having grade 1 complications, and said there were no instances of patients requiring surgery for complications. Among patients in the NPWT group, 33 patients developed skin blistering, compared with 3 patients in the standard gauze group (13% vs. 1.2%; P < .001). After an interim analysis consisting of 444 patients, the study was halted because of “low probability of showing a difference between the two groups at the end of the study.”
The analysis of patients with a BMI of 40 or higher showed 7 of 15 patients (47%) developed wound complications in the NPWT group and 6 of 17 patients (35%) in the standard gauze group (P = .51). In post hoc analyses, the researchers found a median BMI of 26 (range, 17-60) was significantly associated with not developing a wound complication, compared with a BMI of 32 (range, 17-56) (P < .001), and that 41% of patients with a BMI of at least 40 experienced wound complications, compared with 15% of patients with a BMI of less than 40 (P < .001). There was an independent association between developing a wound complication and increasing BMI, according to a multivariate analysis (adjusted odds ratio, 1.10; 95% CI, 1.06-1.14).
Applicability of results unclear for patients with higher BMI
Sarah M. Temkin, MD, a gynecologic oncologist who was not involved with the study, said in an interview that the results by Dr. Leitao and colleagues answer the question of whether patients undergoing surgery for gynecologic malignancy require NPWT, but raised questions about patient selection in the study.
“I think it’s hard to take data from this type of high-end surgical practice and apply it to the general population,” she said, who noted the median BMI of 26 for patients included in the study. A study that included only patients with a BMI of 40 or higher “would have made these results more applicable.”
The low rate of wound complications in the study could potentially be explained by patient selection, Dr. Temkin explained. She cited her own retrospective study from 2016 that showed a wound complication rate of 27.3% for patients receiving prophylactic NPWT where the BMI for the group was 41.29, compared with a complication rate of 19.7% for patients receiving standard care who had a BMI of 30.67.
“It’s hard to cross-trial compare, but that’s significantly higher than what they saw in this prospective study, and I would say that’s a difference with the patient population,” she said. “I think the question of how to reduce surgical-site infections and wound complications in the heavy patient with comorbidities is still unanswered.”
The question is important because patients with a higher BMI and medical comorbidities “still need cancer surgery and methods to reduce the morbidity of that surgery,” Dr. Temkin said. “I think this is an unmet need.”
This study was funded in part by a support grant from the National Institutes of Health/National Cancer Institute Cancer Center, and KCI/Acelity provided part of the study protocol. Nine authors reported personal and institutional relationships in the form of personal fees, grants, stock ownership, consultancies, and speaker’s bureau positions with AstraZeneca, Biom’Up, Bovie Medical, C Surgeries, CMR, ConMed, Covidien, Ethicon, GlaxoSmithKline, GRAIL, Intuitive Surgical, JNJ, Medtronic, Merck, Mylan, Olympus, Stryker/Novadaq, TransEnterix, UpToDate, and Verthermia. Dr. Temkin reported no relevant financial disclosures.
FROM OBSTETRICS & GYNECOLOGY
Should all patients be tested for SARS-CoV-2 before endoscopy?
Dear colleagues and friends,
Welcome to another edition of the Perspectives debates. The COVID-19 crisis has directly affected our endoscopy practices, and it’s raised difficult questions about how best to balance safety with continued delivery of health care services. Dr. John Inadomi and Dr. Shahnaz Sultan address the benefits and downsides of universal testing of patients before endoscopic procedures.
I hope you find this debate helpful and informative for your endoscopy unit’s policies as we navigate these uncertain times. As always, I welcome your comments and suggestions for future topics at [email protected]. Stay safe!
Charles J. Kahi, MD, MS, AGAF, is a professor of medicine at Indiana University, Indianapolis. He is an associate editor for GI & Hepatology News.
Reassurance is important to both patients and providers
The COVID-19 pandemic has been a global economic, societal, and health crisis. As health care systems shifted resources to care for the overwhelming numbers of patients infected with COVID-19 and coincident with lockdown orders issued by local and state governments, elective endoscopy came to a screeching halt. The gastrointestinal professional societies issued a joint statement advocating delays of all elective endoscopies and limiting procedures to those deemed urgent or emergent.1
Within our GI community, there was great concern raised about the degree of aerosolization and risk of virus transmission during endoscopy and colonoscopy, the type of personal protective equipment (PPE) required to minimize infection risk, and the need for negative pressure rooms or prolonged room turnover to provide adequate air exchange. Our understanding of the role of the GI tract in infection and transmission and presentation with GI symptoms, as well as the true prevalence of asymptomatic infection, was rapidly evolving. Even though our colleagues in Asia and Europe faced the COVID-19 pandemic before we did, we still had no roadmap to navigate these issues.
Public health officials emphasized that a critical step to limit the spread of infection hinged on the availability of accurate and reliable tests. However, during the initial phase of the pandemic, priority was given to patients with symptoms or exposure because of limitations of tests, sampling tools, and reagents. As testing became more available and we began to ramp up endoscopy, the American Gastroenterological Association developed a rapid review and guideline evaluating the role of testing prior to endoscopy.2 In this evidence review and guideline, the authors systematically evaluated the diagnostic accuracy of the reverse transcription polymerase chain reaction (RT-PCR) nucleic acid amplification tests (NAAT) available on the U.S. market and described a framework for how a pre-endoscopy testing strategy could help with triage and decisions around PPE use.
Specifically, they made a conditional recommendation supporting a pre-endoscopy testing strategy: “For most endoscopy centers where the prevalence of asymptomatic SARS-CoV-2 infection is intermediate (0.5%-2%), the AGA suggests implementing a pretesting strategy using information about prevalence and test performance (sensitivity/specificity) in combination with considerations about the benefits and downsides of the strategy.”2 This is a conditional recommendation based on low certainty evidence, underscoring the limitations in the evidence for diagnostic test accuracy of the currently available tests (limited sample sizes, test accuracy only for symptomatic patients, no reference standard) and limited knowledge of the true prevalence of SARS-CoV-2 infection at the population level.
The authors of the recommendations emphasized that preprocedure testing could help decrease the risk of transmission by triage – that is, delaying the procedure of patients with positive tests who could infect other patients and health care workers. In addition, for patients with negative tests, surgical masks can be considered during endoscopy to allow preservation of N95/N99 masks that are a limited resource in many settings.
Varying strategies for reopening endoscopy have been adopted by endoscopy centers and health systems. According to one survey, most of responding U.S. endoscopy centers (87%) had developed a formal COVID-19 mitigation protocol, with only 52% of the responding centers testing all patients prior to endoscopy, which highlights the large variation of policies in clinical practice.3 In making the case for a strategy of pretesting of all patients prior to endoscopy, it’s important to emphasize that the benefits of testing outweigh any downsides and that, for health care professionals and patients alike, providing reassurance about the safety of endoscopy for everyone is an important aspect to resuming endoscopy operations.
Concerns regarding acquiring COVID-19 infection in the workplace and infecting family members was and remains a source of significant stress for endoscopy unit personnel across the U.S. Recognizing these issues, many institutions initiated a program for preprocedure COVID-19 testing for all patients undergoing endoscopy. An online survey completed by 47 endoscopy unit personnel found that, after implementation of pre-endoscopy testing, fewer personnel reported anxiety regarding contracting infection (58.1% before vs. 44.7% after; P < .001), there was less concern about infecting family members (88.4% before vs. 68.4% after; P < .05), and fewer providers reported self-isolation practices (living in a room separate from family) (21.3% before vs. 10.8% after; P < .05).4
Moreover, implementation of a pre-endoscopy testing strategy could decrease patient anxiety and decrease patient reluctance to complete endoscopy. With recognition of the long-term consequences of delaying endoscopic evaluation, especially for colorectal cancer screening and surveillance, improving attendance may lessen the impact on colorectal cancer–related morbidity and mortality and other GI-related conditions.5
A pre-endoscopy testing strategy can inform PPE decisions so that N95s and power air-purifying respirators (PAPRs) are reserved for high-risk procedures (such as EGDs in COVID-19-positive patients) with use of surgical masks or extended/reused N95s for patients who test negative. In addition, preprocedure testing can improve endoscopic efficiency eliminating the need to wait for the necessary amount of air exchange between procedures in test-negative patients. In the last 6 months, the testing landscape has changed significantly with the availability of numerous platforms that allow for more efficient processing of tests and the capability of testing saliva instead of nasopharyngeal or nasal swabs. In addition to the original RT-PCR NAAT, more rapid PCR tests, and antigen tests are available. Testing is no longer a scarce resource and for this reason, we should continue our practice of testing all patients prior to endoscopy. Further studies of the false-negative/false-positive rate of various test modalities and test-timing will be important.
John M. Inadomi, MD, AGAF, is with the department of medicine at the University of Utah, Salt Lake City. He has no conflicts to declare.
References
1. Gastroenterology Professional Society Guidance on Endoscopic Procedures during the COVID-19 Pandemic. American College of Gastroenterology. 2020 Apr 1.
2. Sultan S et al. Gastroenterology. 2020 Nov;159(5):1935-48.e5.
3. Moraveji S et al. Gastroenterology. 2020 Oct 1;159(4):1568-70.e5.
4. Podboy A et al. Gastroenterology. 2020 Oct 1;1586-8.e4.
5. Dekker E et al. Gastroenterology. 2020 Dec;159(6):1998-2003.
Barriers to care should be avoided
In the AGA Institute Rapid Review and Recommendations on the Role of Pre-Procedure SARS-CoV-2 Testing and Endoscopy, the authors made a conditional recommendation for implementation of a preprocedure testing strategy, specifically for endoscopy centers where the prevalence of asymptomatic SARS-CoV-2 infection is intermediate (0.5%-2%).1 The authors stated that, in settings where testing is feasible and there is less perceived burden on patients and when the benefits outweigh the downsides (such as when false positives do not significantly outnumber the true positives), an endoscopy center should implement a preprocedure testing strategy. In addition to the prevalence of SARS-CoV-2 infection, which influences both the positive and negative predictive value of testing, the authors took into account the downstream consequences of test results, the pros and cons of a testing strategy, and the availability of PPE and tests.
The authors further clarify that, in areas with either low or high prevalence of asymptomatic cases, a pre-endoscopy testing strategy may not be informative. The group made a conditional recommendation against testing in these settings, highlighting concerns around the false positive rates in low-prevalence areas and potentially limited availability of tests and false negative rates in high-prevalence areas.
Early in the pandemic, because of the limited availability of tests, symptomatic or high-risk patients were given priority for SARS-CoV-2 testing. Since that time, over 180 different commercially available SARS-CoV-2 assays have become available, and many health systems have developed their own laboratory-developed tests.2 However, despite the aggressive efforts to ramp up test capacity, many endoscopy centers still struggle to obtain timely and reliable results. In light of continued challenges with testing, many endoscopy centers chose to not implement a pretesting strategy and instead proceed with N95 respirators or PAPRs for all procedures. Endoscopy centers that implemented a no-testing strategy emphasized the importance of ramping up productivity as quickly as possible and simultaneously reducing any barriers for patients.
They also highlighted reports that demonstrated a low yield of positive tests and that triaging could be performed equally well with the use of a symptom checklist or fever check. In one study from New York, among 623 asymptomatic patients tested before endoscopy during May and June, which was the height of the first surge, only 6 patients tested positive (overall percentage of SARS-CoV-2–positive tests in asymptomatic patients, 0.96%).3 Similarly, low rates of positive tests were reported in a center in Miami: Among a total of 396 PCR swabs in preparation for endoscopy, one patient had a positive PCR result (positive test rate, 0.25%).4
What are the implications for patients, providers, and endoscopy centers if a preprocedure testing strategy is not adopted? For individual patients, there is a potential for heightened concern about risk of infection from health care staff or other patients, but this may be offset by the decreased burden of testing. For health care professionals, it means that all procedures need to be performed with universal precautions and appropriate PPE. From an endoscopy operations perspective, there remain unanswered questions regarding room turnover and the necessity for negative pressure rooms. Symptom screening, which is locally required for all health visits, may help identify symptomatic patients, and thus, procedures for these individuals could be canceled and rescheduled or moved to negative pressure rooms (based on urgency).
Questions remain about the need to perform all procedures in negative pressure rooms or with prolonged room turnover to allow for a requisite number of air exchanges. There is general agreement among gastroenterologists that upper endoscopy is an aerosol-generating procedure. The passage of the scope near the pharynx (where the virus has a propensity for colonization and infection) may lead to aerosolization of infective particles and confer an increased risk of infection. However, it is unclear whether colonoscopy actually confers an increased risk. This would require that an asymptomatic patient would be infected with the SARS-CoV-2 virus and have viable viral particles in the colon that are released during the passage of flatus in high enough quantity to allow for aerosolization and transmission of infection. Conceptually, while there are potential risks associated with both endoscopy and colonoscopy, it is reassuring that we are not seeing published reports (or anecdotal evidence) suggesting high rates of COVID-19 infection among endoscopy staff or at endoscopy centers. Requiring prolonged room turnover between every procedure would negatively impact endoscopy efficiency and recovery of endoscopy centers.
Indeed, the aftermath of the COVID-19 pandemic will be far reaching. While telemedicine has helped mitigate some of the collateral damage, the disruption of cancer screening and surveillance programs may lead to high cancer-related morbidity and mortality. In one study evaluating the impact of COVID-19 on the U.S. cancer population, authors analyzed 6,227,474 Medicare Fee for Service claims (representing 5%-7% of the Medicare population) and found a substantial decrease in cancer screening and cancer care (therapy and surgeries).5 Screening for colon cancer was reduced by 75% in April.
Eliminating any potential barriers to care should be the highest priority. A requirement for patients to undergo preprocedure testing may contribute to increased anxiety and added costs and may further delay care. From a patient perspective, finding a testing facility, obtaining the test within 48-72 hours, self-isolating until the day of endoscopy, and dealing with the uncertainty of the test result may serve as additional barriers for completion of endoscopy. Moreover, the differential availability of testing may further exacerbate health inequities.
In the absence of pre-endoscopy testing for all patients, routine screening for symptoms, following COVID-19 infection precautions for all cases with strict adherence to physical distancing, and use of N95 (or PAPRs) during endoscopy may minimize viral transmission among patients and staff while maximizing patient adherence to endoscopy, ensuring resumption of endoscopic services, and ultimately mitigating some of the devastating impact of COVID-19 on population health.
Shahnaz Sultan, MD, MHSc, AGAF, FACG, is with the division of gastroenterology at the University of Minnesota in Minneapolis and the Center for Care Delivery and Outcomes Research at Minneapolis Veterans Affairs Healthcare System. She has no conflicts to declare.
References
1. Sultan S et al. Gastroenterology. 2020 Nov;159(5):1935-48.e5.
2. Food and Drug Administration. Emergency Use Authorization: Coronavirus Disease 2019 (COVID-19) EUA Information. 3. Dollinger MT et al. Gastroenterology. 2020;159:1962-4.
4. Forde JJ et al. Gastroenterology. 2020;159:1538-40.
5. Patt D et al. JCO Clin Cancer Inform. 2020 Nov;4:1059-71.
Dear colleagues and friends,
Welcome to another edition of the Perspectives debates. The COVID-19 crisis has directly affected our endoscopy practices, and it’s raised difficult questions about how best to balance safety with continued delivery of health care services. Dr. John Inadomi and Dr. Shahnaz Sultan address the benefits and downsides of universal testing of patients before endoscopic procedures.
I hope you find this debate helpful and informative for your endoscopy unit’s policies as we navigate these uncertain times. As always, I welcome your comments and suggestions for future topics at [email protected]. Stay safe!
Charles J. Kahi, MD, MS, AGAF, is a professor of medicine at Indiana University, Indianapolis. He is an associate editor for GI & Hepatology News.
Reassurance is important to both patients and providers
The COVID-19 pandemic has been a global economic, societal, and health crisis. As health care systems shifted resources to care for the overwhelming numbers of patients infected with COVID-19 and coincident with lockdown orders issued by local and state governments, elective endoscopy came to a screeching halt. The gastrointestinal professional societies issued a joint statement advocating delays of all elective endoscopies and limiting procedures to those deemed urgent or emergent.1
Within our GI community, there was great concern raised about the degree of aerosolization and risk of virus transmission during endoscopy and colonoscopy, the type of personal protective equipment (PPE) required to minimize infection risk, and the need for negative pressure rooms or prolonged room turnover to provide adequate air exchange. Our understanding of the role of the GI tract in infection and transmission and presentation with GI symptoms, as well as the true prevalence of asymptomatic infection, was rapidly evolving. Even though our colleagues in Asia and Europe faced the COVID-19 pandemic before we did, we still had no roadmap to navigate these issues.
Public health officials emphasized that a critical step to limit the spread of infection hinged on the availability of accurate and reliable tests. However, during the initial phase of the pandemic, priority was given to patients with symptoms or exposure because of limitations of tests, sampling tools, and reagents. As testing became more available and we began to ramp up endoscopy, the American Gastroenterological Association developed a rapid review and guideline evaluating the role of testing prior to endoscopy.2 In this evidence review and guideline, the authors systematically evaluated the diagnostic accuracy of the reverse transcription polymerase chain reaction (RT-PCR) nucleic acid amplification tests (NAAT) available on the U.S. market and described a framework for how a pre-endoscopy testing strategy could help with triage and decisions around PPE use.
Specifically, they made a conditional recommendation supporting a pre-endoscopy testing strategy: “For most endoscopy centers where the prevalence of asymptomatic SARS-CoV-2 infection is intermediate (0.5%-2%), the AGA suggests implementing a pretesting strategy using information about prevalence and test performance (sensitivity/specificity) in combination with considerations about the benefits and downsides of the strategy.”2 This is a conditional recommendation based on low certainty evidence, underscoring the limitations in the evidence for diagnostic test accuracy of the currently available tests (limited sample sizes, test accuracy only for symptomatic patients, no reference standard) and limited knowledge of the true prevalence of SARS-CoV-2 infection at the population level.
The authors of the recommendations emphasized that preprocedure testing could help decrease the risk of transmission by triage – that is, delaying the procedure of patients with positive tests who could infect other patients and health care workers. In addition, for patients with negative tests, surgical masks can be considered during endoscopy to allow preservation of N95/N99 masks that are a limited resource in many settings.
Varying strategies for reopening endoscopy have been adopted by endoscopy centers and health systems. According to one survey, most of responding U.S. endoscopy centers (87%) had developed a formal COVID-19 mitigation protocol, with only 52% of the responding centers testing all patients prior to endoscopy, which highlights the large variation of policies in clinical practice.3 In making the case for a strategy of pretesting of all patients prior to endoscopy, it’s important to emphasize that the benefits of testing outweigh any downsides and that, for health care professionals and patients alike, providing reassurance about the safety of endoscopy for everyone is an important aspect to resuming endoscopy operations.
Concerns regarding acquiring COVID-19 infection in the workplace and infecting family members was and remains a source of significant stress for endoscopy unit personnel across the U.S. Recognizing these issues, many institutions initiated a program for preprocedure COVID-19 testing for all patients undergoing endoscopy. An online survey completed by 47 endoscopy unit personnel found that, after implementation of pre-endoscopy testing, fewer personnel reported anxiety regarding contracting infection (58.1% before vs. 44.7% after; P < .001), there was less concern about infecting family members (88.4% before vs. 68.4% after; P < .05), and fewer providers reported self-isolation practices (living in a room separate from family) (21.3% before vs. 10.8% after; P < .05).4
Moreover, implementation of a pre-endoscopy testing strategy could decrease patient anxiety and decrease patient reluctance to complete endoscopy. With recognition of the long-term consequences of delaying endoscopic evaluation, especially for colorectal cancer screening and surveillance, improving attendance may lessen the impact on colorectal cancer–related morbidity and mortality and other GI-related conditions.5
A pre-endoscopy testing strategy can inform PPE decisions so that N95s and power air-purifying respirators (PAPRs) are reserved for high-risk procedures (such as EGDs in COVID-19-positive patients) with use of surgical masks or extended/reused N95s for patients who test negative. In addition, preprocedure testing can improve endoscopic efficiency eliminating the need to wait for the necessary amount of air exchange between procedures in test-negative patients. In the last 6 months, the testing landscape has changed significantly with the availability of numerous platforms that allow for more efficient processing of tests and the capability of testing saliva instead of nasopharyngeal or nasal swabs. In addition to the original RT-PCR NAAT, more rapid PCR tests, and antigen tests are available. Testing is no longer a scarce resource and for this reason, we should continue our practice of testing all patients prior to endoscopy. Further studies of the false-negative/false-positive rate of various test modalities and test-timing will be important.
John M. Inadomi, MD, AGAF, is with the department of medicine at the University of Utah, Salt Lake City. He has no conflicts to declare.
References
1. Gastroenterology Professional Society Guidance on Endoscopic Procedures during the COVID-19 Pandemic. American College of Gastroenterology. 2020 Apr 1.
2. Sultan S et al. Gastroenterology. 2020 Nov;159(5):1935-48.e5.
3. Moraveji S et al. Gastroenterology. 2020 Oct 1;159(4):1568-70.e5.
4. Podboy A et al. Gastroenterology. 2020 Oct 1;1586-8.e4.
5. Dekker E et al. Gastroenterology. 2020 Dec;159(6):1998-2003.
Barriers to care should be avoided
In the AGA Institute Rapid Review and Recommendations on the Role of Pre-Procedure SARS-CoV-2 Testing and Endoscopy, the authors made a conditional recommendation for implementation of a preprocedure testing strategy, specifically for endoscopy centers where the prevalence of asymptomatic SARS-CoV-2 infection is intermediate (0.5%-2%).1 The authors stated that, in settings where testing is feasible and there is less perceived burden on patients and when the benefits outweigh the downsides (such as when false positives do not significantly outnumber the true positives), an endoscopy center should implement a preprocedure testing strategy. In addition to the prevalence of SARS-CoV-2 infection, which influences both the positive and negative predictive value of testing, the authors took into account the downstream consequences of test results, the pros and cons of a testing strategy, and the availability of PPE and tests.
The authors further clarify that, in areas with either low or high prevalence of asymptomatic cases, a pre-endoscopy testing strategy may not be informative. The group made a conditional recommendation against testing in these settings, highlighting concerns around the false positive rates in low-prevalence areas and potentially limited availability of tests and false negative rates in high-prevalence areas.
Early in the pandemic, because of the limited availability of tests, symptomatic or high-risk patients were given priority for SARS-CoV-2 testing. Since that time, over 180 different commercially available SARS-CoV-2 assays have become available, and many health systems have developed their own laboratory-developed tests.2 However, despite the aggressive efforts to ramp up test capacity, many endoscopy centers still struggle to obtain timely and reliable results. In light of continued challenges with testing, many endoscopy centers chose to not implement a pretesting strategy and instead proceed with N95 respirators or PAPRs for all procedures. Endoscopy centers that implemented a no-testing strategy emphasized the importance of ramping up productivity as quickly as possible and simultaneously reducing any barriers for patients.
They also highlighted reports that demonstrated a low yield of positive tests and that triaging could be performed equally well with the use of a symptom checklist or fever check. In one study from New York, among 623 asymptomatic patients tested before endoscopy during May and June, which was the height of the first surge, only 6 patients tested positive (overall percentage of SARS-CoV-2–positive tests in asymptomatic patients, 0.96%).3 Similarly, low rates of positive tests were reported in a center in Miami: Among a total of 396 PCR swabs in preparation for endoscopy, one patient had a positive PCR result (positive test rate, 0.25%).4
What are the implications for patients, providers, and endoscopy centers if a preprocedure testing strategy is not adopted? For individual patients, there is a potential for heightened concern about risk of infection from health care staff or other patients, but this may be offset by the decreased burden of testing. For health care professionals, it means that all procedures need to be performed with universal precautions and appropriate PPE. From an endoscopy operations perspective, there remain unanswered questions regarding room turnover and the necessity for negative pressure rooms. Symptom screening, which is locally required for all health visits, may help identify symptomatic patients, and thus, procedures for these individuals could be canceled and rescheduled or moved to negative pressure rooms (based on urgency).
Questions remain about the need to perform all procedures in negative pressure rooms or with prolonged room turnover to allow for a requisite number of air exchanges. There is general agreement among gastroenterologists that upper endoscopy is an aerosol-generating procedure. The passage of the scope near the pharynx (where the virus has a propensity for colonization and infection) may lead to aerosolization of infective particles and confer an increased risk of infection. However, it is unclear whether colonoscopy actually confers an increased risk. This would require that an asymptomatic patient would be infected with the SARS-CoV-2 virus and have viable viral particles in the colon that are released during the passage of flatus in high enough quantity to allow for aerosolization and transmission of infection. Conceptually, while there are potential risks associated with both endoscopy and colonoscopy, it is reassuring that we are not seeing published reports (or anecdotal evidence) suggesting high rates of COVID-19 infection among endoscopy staff or at endoscopy centers. Requiring prolonged room turnover between every procedure would negatively impact endoscopy efficiency and recovery of endoscopy centers.
Indeed, the aftermath of the COVID-19 pandemic will be far reaching. While telemedicine has helped mitigate some of the collateral damage, the disruption of cancer screening and surveillance programs may lead to high cancer-related morbidity and mortality. In one study evaluating the impact of COVID-19 on the U.S. cancer population, authors analyzed 6,227,474 Medicare Fee for Service claims (representing 5%-7% of the Medicare population) and found a substantial decrease in cancer screening and cancer care (therapy and surgeries).5 Screening for colon cancer was reduced by 75% in April.
Eliminating any potential barriers to care should be the highest priority. A requirement for patients to undergo preprocedure testing may contribute to increased anxiety and added costs and may further delay care. From a patient perspective, finding a testing facility, obtaining the test within 48-72 hours, self-isolating until the day of endoscopy, and dealing with the uncertainty of the test result may serve as additional barriers for completion of endoscopy. Moreover, the differential availability of testing may further exacerbate health inequities.
In the absence of pre-endoscopy testing for all patients, routine screening for symptoms, following COVID-19 infection precautions for all cases with strict adherence to physical distancing, and use of N95 (or PAPRs) during endoscopy may minimize viral transmission among patients and staff while maximizing patient adherence to endoscopy, ensuring resumption of endoscopic services, and ultimately mitigating some of the devastating impact of COVID-19 on population health.
Shahnaz Sultan, MD, MHSc, AGAF, FACG, is with the division of gastroenterology at the University of Minnesota in Minneapolis and the Center for Care Delivery and Outcomes Research at Minneapolis Veterans Affairs Healthcare System. She has no conflicts to declare.
References
1. Sultan S et al. Gastroenterology. 2020 Nov;159(5):1935-48.e5.
2. Food and Drug Administration. Emergency Use Authorization: Coronavirus Disease 2019 (COVID-19) EUA Information. 3. Dollinger MT et al. Gastroenterology. 2020;159:1962-4.
4. Forde JJ et al. Gastroenterology. 2020;159:1538-40.
5. Patt D et al. JCO Clin Cancer Inform. 2020 Nov;4:1059-71.
Dear colleagues and friends,
Welcome to another edition of the Perspectives debates. The COVID-19 crisis has directly affected our endoscopy practices, and it’s raised difficult questions about how best to balance safety with continued delivery of health care services. Dr. John Inadomi and Dr. Shahnaz Sultan address the benefits and downsides of universal testing of patients before endoscopic procedures.
I hope you find this debate helpful and informative for your endoscopy unit’s policies as we navigate these uncertain times. As always, I welcome your comments and suggestions for future topics at [email protected]. Stay safe!
Charles J. Kahi, MD, MS, AGAF, is a professor of medicine at Indiana University, Indianapolis. He is an associate editor for GI & Hepatology News.
Reassurance is important to both patients and providers
The COVID-19 pandemic has been a global economic, societal, and health crisis. As health care systems shifted resources to care for the overwhelming numbers of patients infected with COVID-19 and coincident with lockdown orders issued by local and state governments, elective endoscopy came to a screeching halt. The gastrointestinal professional societies issued a joint statement advocating delays of all elective endoscopies and limiting procedures to those deemed urgent or emergent.1
Within our GI community, there was great concern raised about the degree of aerosolization and risk of virus transmission during endoscopy and colonoscopy, the type of personal protective equipment (PPE) required to minimize infection risk, and the need for negative pressure rooms or prolonged room turnover to provide adequate air exchange. Our understanding of the role of the GI tract in infection and transmission and presentation with GI symptoms, as well as the true prevalence of asymptomatic infection, was rapidly evolving. Even though our colleagues in Asia and Europe faced the COVID-19 pandemic before we did, we still had no roadmap to navigate these issues.
Public health officials emphasized that a critical step to limit the spread of infection hinged on the availability of accurate and reliable tests. However, during the initial phase of the pandemic, priority was given to patients with symptoms or exposure because of limitations of tests, sampling tools, and reagents. As testing became more available and we began to ramp up endoscopy, the American Gastroenterological Association developed a rapid review and guideline evaluating the role of testing prior to endoscopy.2 In this evidence review and guideline, the authors systematically evaluated the diagnostic accuracy of the reverse transcription polymerase chain reaction (RT-PCR) nucleic acid amplification tests (NAAT) available on the U.S. market and described a framework for how a pre-endoscopy testing strategy could help with triage and decisions around PPE use.
Specifically, they made a conditional recommendation supporting a pre-endoscopy testing strategy: “For most endoscopy centers where the prevalence of asymptomatic SARS-CoV-2 infection is intermediate (0.5%-2%), the AGA suggests implementing a pretesting strategy using information about prevalence and test performance (sensitivity/specificity) in combination with considerations about the benefits and downsides of the strategy.”2 This is a conditional recommendation based on low certainty evidence, underscoring the limitations in the evidence for diagnostic test accuracy of the currently available tests (limited sample sizes, test accuracy only for symptomatic patients, no reference standard) and limited knowledge of the true prevalence of SARS-CoV-2 infection at the population level.
The authors of the recommendations emphasized that preprocedure testing could help decrease the risk of transmission by triage – that is, delaying the procedure of patients with positive tests who could infect other patients and health care workers. In addition, for patients with negative tests, surgical masks can be considered during endoscopy to allow preservation of N95/N99 masks that are a limited resource in many settings.
Varying strategies for reopening endoscopy have been adopted by endoscopy centers and health systems. According to one survey, most of responding U.S. endoscopy centers (87%) had developed a formal COVID-19 mitigation protocol, with only 52% of the responding centers testing all patients prior to endoscopy, which highlights the large variation of policies in clinical practice.3 In making the case for a strategy of pretesting of all patients prior to endoscopy, it’s important to emphasize that the benefits of testing outweigh any downsides and that, for health care professionals and patients alike, providing reassurance about the safety of endoscopy for everyone is an important aspect to resuming endoscopy operations.
Concerns regarding acquiring COVID-19 infection in the workplace and infecting family members was and remains a source of significant stress for endoscopy unit personnel across the U.S. Recognizing these issues, many institutions initiated a program for preprocedure COVID-19 testing for all patients undergoing endoscopy. An online survey completed by 47 endoscopy unit personnel found that, after implementation of pre-endoscopy testing, fewer personnel reported anxiety regarding contracting infection (58.1% before vs. 44.7% after; P < .001), there was less concern about infecting family members (88.4% before vs. 68.4% after; P < .05), and fewer providers reported self-isolation practices (living in a room separate from family) (21.3% before vs. 10.8% after; P < .05).4
Moreover, implementation of a pre-endoscopy testing strategy could decrease patient anxiety and decrease patient reluctance to complete endoscopy. With recognition of the long-term consequences of delaying endoscopic evaluation, especially for colorectal cancer screening and surveillance, improving attendance may lessen the impact on colorectal cancer–related morbidity and mortality and other GI-related conditions.5
A pre-endoscopy testing strategy can inform PPE decisions so that N95s and power air-purifying respirators (PAPRs) are reserved for high-risk procedures (such as EGDs in COVID-19-positive patients) with use of surgical masks or extended/reused N95s for patients who test negative. In addition, preprocedure testing can improve endoscopic efficiency eliminating the need to wait for the necessary amount of air exchange between procedures in test-negative patients. In the last 6 months, the testing landscape has changed significantly with the availability of numerous platforms that allow for more efficient processing of tests and the capability of testing saliva instead of nasopharyngeal or nasal swabs. In addition to the original RT-PCR NAAT, more rapid PCR tests, and antigen tests are available. Testing is no longer a scarce resource and for this reason, we should continue our practice of testing all patients prior to endoscopy. Further studies of the false-negative/false-positive rate of various test modalities and test-timing will be important.
John M. Inadomi, MD, AGAF, is with the department of medicine at the University of Utah, Salt Lake City. He has no conflicts to declare.
References
1. Gastroenterology Professional Society Guidance on Endoscopic Procedures during the COVID-19 Pandemic. American College of Gastroenterology. 2020 Apr 1.
2. Sultan S et al. Gastroenterology. 2020 Nov;159(5):1935-48.e5.
3. Moraveji S et al. Gastroenterology. 2020 Oct 1;159(4):1568-70.e5.
4. Podboy A et al. Gastroenterology. 2020 Oct 1;1586-8.e4.
5. Dekker E et al. Gastroenterology. 2020 Dec;159(6):1998-2003.
Barriers to care should be avoided
In the AGA Institute Rapid Review and Recommendations on the Role of Pre-Procedure SARS-CoV-2 Testing and Endoscopy, the authors made a conditional recommendation for implementation of a preprocedure testing strategy, specifically for endoscopy centers where the prevalence of asymptomatic SARS-CoV-2 infection is intermediate (0.5%-2%).1 The authors stated that, in settings where testing is feasible and there is less perceived burden on patients and when the benefits outweigh the downsides (such as when false positives do not significantly outnumber the true positives), an endoscopy center should implement a preprocedure testing strategy. In addition to the prevalence of SARS-CoV-2 infection, which influences both the positive and negative predictive value of testing, the authors took into account the downstream consequences of test results, the pros and cons of a testing strategy, and the availability of PPE and tests.
The authors further clarify that, in areas with either low or high prevalence of asymptomatic cases, a pre-endoscopy testing strategy may not be informative. The group made a conditional recommendation against testing in these settings, highlighting concerns around the false positive rates in low-prevalence areas and potentially limited availability of tests and false negative rates in high-prevalence areas.
Early in the pandemic, because of the limited availability of tests, symptomatic or high-risk patients were given priority for SARS-CoV-2 testing. Since that time, over 180 different commercially available SARS-CoV-2 assays have become available, and many health systems have developed their own laboratory-developed tests.2 However, despite the aggressive efforts to ramp up test capacity, many endoscopy centers still struggle to obtain timely and reliable results. In light of continued challenges with testing, many endoscopy centers chose to not implement a pretesting strategy and instead proceed with N95 respirators or PAPRs for all procedures. Endoscopy centers that implemented a no-testing strategy emphasized the importance of ramping up productivity as quickly as possible and simultaneously reducing any barriers for patients.
They also highlighted reports that demonstrated a low yield of positive tests and that triaging could be performed equally well with the use of a symptom checklist or fever check. In one study from New York, among 623 asymptomatic patients tested before endoscopy during May and June, which was the height of the first surge, only 6 patients tested positive (overall percentage of SARS-CoV-2–positive tests in asymptomatic patients, 0.96%).3 Similarly, low rates of positive tests were reported in a center in Miami: Among a total of 396 PCR swabs in preparation for endoscopy, one patient had a positive PCR result (positive test rate, 0.25%).4
What are the implications for patients, providers, and endoscopy centers if a preprocedure testing strategy is not adopted? For individual patients, there is a potential for heightened concern about risk of infection from health care staff or other patients, but this may be offset by the decreased burden of testing. For health care professionals, it means that all procedures need to be performed with universal precautions and appropriate PPE. From an endoscopy operations perspective, there remain unanswered questions regarding room turnover and the necessity for negative pressure rooms. Symptom screening, which is locally required for all health visits, may help identify symptomatic patients, and thus, procedures for these individuals could be canceled and rescheduled or moved to negative pressure rooms (based on urgency).
Questions remain about the need to perform all procedures in negative pressure rooms or with prolonged room turnover to allow for a requisite number of air exchanges. There is general agreement among gastroenterologists that upper endoscopy is an aerosol-generating procedure. The passage of the scope near the pharynx (where the virus has a propensity for colonization and infection) may lead to aerosolization of infective particles and confer an increased risk of infection. However, it is unclear whether colonoscopy actually confers an increased risk. This would require that an asymptomatic patient would be infected with the SARS-CoV-2 virus and have viable viral particles in the colon that are released during the passage of flatus in high enough quantity to allow for aerosolization and transmission of infection. Conceptually, while there are potential risks associated with both endoscopy and colonoscopy, it is reassuring that we are not seeing published reports (or anecdotal evidence) suggesting high rates of COVID-19 infection among endoscopy staff or at endoscopy centers. Requiring prolonged room turnover between every procedure would negatively impact endoscopy efficiency and recovery of endoscopy centers.
Indeed, the aftermath of the COVID-19 pandemic will be far reaching. While telemedicine has helped mitigate some of the collateral damage, the disruption of cancer screening and surveillance programs may lead to high cancer-related morbidity and mortality. In one study evaluating the impact of COVID-19 on the U.S. cancer population, authors analyzed 6,227,474 Medicare Fee for Service claims (representing 5%-7% of the Medicare population) and found a substantial decrease in cancer screening and cancer care (therapy and surgeries).5 Screening for colon cancer was reduced by 75% in April.
Eliminating any potential barriers to care should be the highest priority. A requirement for patients to undergo preprocedure testing may contribute to increased anxiety and added costs and may further delay care. From a patient perspective, finding a testing facility, obtaining the test within 48-72 hours, self-isolating until the day of endoscopy, and dealing with the uncertainty of the test result may serve as additional barriers for completion of endoscopy. Moreover, the differential availability of testing may further exacerbate health inequities.
In the absence of pre-endoscopy testing for all patients, routine screening for symptoms, following COVID-19 infection precautions for all cases with strict adherence to physical distancing, and use of N95 (or PAPRs) during endoscopy may minimize viral transmission among patients and staff while maximizing patient adherence to endoscopy, ensuring resumption of endoscopic services, and ultimately mitigating some of the devastating impact of COVID-19 on population health.
Shahnaz Sultan, MD, MHSc, AGAF, FACG, is with the division of gastroenterology at the University of Minnesota in Minneapolis and the Center for Care Delivery and Outcomes Research at Minneapolis Veterans Affairs Healthcare System. She has no conflicts to declare.
References
1. Sultan S et al. Gastroenterology. 2020 Nov;159(5):1935-48.e5.
2. Food and Drug Administration. Emergency Use Authorization: Coronavirus Disease 2019 (COVID-19) EUA Information. 3. Dollinger MT et al. Gastroenterology. 2020;159:1962-4.
4. Forde JJ et al. Gastroenterology. 2020;159:1538-40.
5. Patt D et al. JCO Clin Cancer Inform. 2020 Nov;4:1059-71.
Strep A and tic worsening: Final word?
Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one-third of whom also had attention deficit hyperactivity disorder and one-third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online in Neurology.
‘Intense debate’
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) – a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary (Alta.), in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD, and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals.
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit.
- Weekly diaries: Parents were asked to indicate any worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report, by phone or email, any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of greater than or equal to 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into four categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposure.
Unlikely trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs. 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or cornerstone?
Commenting on the study for this news organization, Margo Thienemann, MD, clinical professor of psychiatry, Stanford (Calif.) University, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Dr. Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham (England) Medical School and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Mo., suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick – a cornerstone? – in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Dr. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary (Alta.), the Michael P. Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Dr. Cavanna, Dr. Coffman, and Dr. Thienemann have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one-third of whom also had attention deficit hyperactivity disorder and one-third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online in Neurology.
‘Intense debate’
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) – a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary (Alta.), in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD, and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals.
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit.
- Weekly diaries: Parents were asked to indicate any worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report, by phone or email, any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of greater than or equal to 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into four categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposure.
Unlikely trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs. 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or cornerstone?
Commenting on the study for this news organization, Margo Thienemann, MD, clinical professor of psychiatry, Stanford (Calif.) University, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Dr. Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham (England) Medical School and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Mo., suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick – a cornerstone? – in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Dr. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary (Alta.), the Michael P. Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Dr. Cavanna, Dr. Coffman, and Dr. Thienemann have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one-third of whom also had attention deficit hyperactivity disorder and one-third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online in Neurology.
‘Intense debate’
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) – a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary (Alta.), in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD, and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals.
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit.
- Weekly diaries: Parents were asked to indicate any worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report, by phone or email, any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of greater than or equal to 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into four categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposure.
Unlikely trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs. 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or cornerstone?
Commenting on the study for this news organization, Margo Thienemann, MD, clinical professor of psychiatry, Stanford (Calif.) University, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Dr. Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham (England) Medical School and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Mo., suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick – a cornerstone? – in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Dr. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary (Alta.), the Michael P. Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Dr. Cavanna, Dr. Coffman, and Dr. Thienemann have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SHM Converge: New format, fresh content
While we all long for a traditional in-person meeting “like the good old days”, there are some significant advantages to a virtual meeting like Converge.
The most significant advantage is the ability to review more content than ever before, as we offer a combination of live and recorded “on-demand” sessions. This allows for incredible flexibility in garnering “top-shelf” content from hospital medicine experts around the country, without having to choose from competing sessions. We are especially looking forward to new sessions this year focused on COVID-19; diversity, equity, and inclusion; and resilience.
The Converge conference will still be offering networking sessions throughout – even in the virtual conference environment. We consider networking a vital and endearing part of the value equation for SHM members. For example, we now can participate in several Special Interest Forums, since many of us have several niche interests and want to take advantage of more than one of these networking opportunities. We also carefully preserved the signature “Update in Hospital Medicine” session, as well as the scientific abstract poster reception and the Best of Research and Innovation sessions. These are long-term favorites at the annual conference and lend themselves well to virtual transformation. Some of the workshops and special sessions have exclusive audience engagement and are not offered on demand, so signing up early for these sessions is highly recommended.
SHM remains the professional home for hospitalists, and we rely on the annual conference to keep us all informed on current and forward-thinking clinical practice, practice management, leadership, academics, research, and other topics. This is one of many examples of how SHM has been able to pivot to meet the needs of hospitalists throughout the pandemic. Not only have we successfully converted “traditional” meetings into virtual meetings, but we have been able to curate and deliver content faster and more seamlessly than ever before.
Whether via The Hospitalist, the Journal of Hospital Medicine, the SHM website, or our other educational platforms, SHM has remained committed to being the single “source of truth” for all things hospital medicine. Within the tumultuous political landscape of the past year, the SHM advocacy team has been more active and engaged than ever, in advocating for a myriad of hospitalist-related legislative changes. These are just a few of the ways SHM continues to add value to hospitalist members every day.
Although we will certainly miss seeing each other in person, we are confident that the SHM team will meet and exceed expectations on content delivery and will take advantage of the virtual format to improve content access. We look forward to “seeing” you at SHM Converge this year and hope you take advantage of the enhanced delivery and access to an array of amazing content!
Dr. Scheurer is president of the Society of Hospital Medicine. She is a hospitalist and chief quality officer, MUSC Health System, Medical University of South Carolina, Charleston.
While we all long for a traditional in-person meeting “like the good old days”, there are some significant advantages to a virtual meeting like Converge.
The most significant advantage is the ability to review more content than ever before, as we offer a combination of live and recorded “on-demand” sessions. This allows for incredible flexibility in garnering “top-shelf” content from hospital medicine experts around the country, without having to choose from competing sessions. We are especially looking forward to new sessions this year focused on COVID-19; diversity, equity, and inclusion; and resilience.
The Converge conference will still be offering networking sessions throughout – even in the virtual conference environment. We consider networking a vital and endearing part of the value equation for SHM members. For example, we now can participate in several Special Interest Forums, since many of us have several niche interests and want to take advantage of more than one of these networking opportunities. We also carefully preserved the signature “Update in Hospital Medicine” session, as well as the scientific abstract poster reception and the Best of Research and Innovation sessions. These are long-term favorites at the annual conference and lend themselves well to virtual transformation. Some of the workshops and special sessions have exclusive audience engagement and are not offered on demand, so signing up early for these sessions is highly recommended.
SHM remains the professional home for hospitalists, and we rely on the annual conference to keep us all informed on current and forward-thinking clinical practice, practice management, leadership, academics, research, and other topics. This is one of many examples of how SHM has been able to pivot to meet the needs of hospitalists throughout the pandemic. Not only have we successfully converted “traditional” meetings into virtual meetings, but we have been able to curate and deliver content faster and more seamlessly than ever before.
Whether via The Hospitalist, the Journal of Hospital Medicine, the SHM website, or our other educational platforms, SHM has remained committed to being the single “source of truth” for all things hospital medicine. Within the tumultuous political landscape of the past year, the SHM advocacy team has been more active and engaged than ever, in advocating for a myriad of hospitalist-related legislative changes. These are just a few of the ways SHM continues to add value to hospitalist members every day.
Although we will certainly miss seeing each other in person, we are confident that the SHM team will meet and exceed expectations on content delivery and will take advantage of the virtual format to improve content access. We look forward to “seeing” you at SHM Converge this year and hope you take advantage of the enhanced delivery and access to an array of amazing content!
Dr. Scheurer is president of the Society of Hospital Medicine. She is a hospitalist and chief quality officer, MUSC Health System, Medical University of South Carolina, Charleston.
While we all long for a traditional in-person meeting “like the good old days”, there are some significant advantages to a virtual meeting like Converge.
The most significant advantage is the ability to review more content than ever before, as we offer a combination of live and recorded “on-demand” sessions. This allows for incredible flexibility in garnering “top-shelf” content from hospital medicine experts around the country, without having to choose from competing sessions. We are especially looking forward to new sessions this year focused on COVID-19; diversity, equity, and inclusion; and resilience.
The Converge conference will still be offering networking sessions throughout – even in the virtual conference environment. We consider networking a vital and endearing part of the value equation for SHM members. For example, we now can participate in several Special Interest Forums, since many of us have several niche interests and want to take advantage of more than one of these networking opportunities. We also carefully preserved the signature “Update in Hospital Medicine” session, as well as the scientific abstract poster reception and the Best of Research and Innovation sessions. These are long-term favorites at the annual conference and lend themselves well to virtual transformation. Some of the workshops and special sessions have exclusive audience engagement and are not offered on demand, so signing up early for these sessions is highly recommended.
SHM remains the professional home for hospitalists, and we rely on the annual conference to keep us all informed on current and forward-thinking clinical practice, practice management, leadership, academics, research, and other topics. This is one of many examples of how SHM has been able to pivot to meet the needs of hospitalists throughout the pandemic. Not only have we successfully converted “traditional” meetings into virtual meetings, but we have been able to curate and deliver content faster and more seamlessly than ever before.
Whether via The Hospitalist, the Journal of Hospital Medicine, the SHM website, or our other educational platforms, SHM has remained committed to being the single “source of truth” for all things hospital medicine. Within the tumultuous political landscape of the past year, the SHM advocacy team has been more active and engaged than ever, in advocating for a myriad of hospitalist-related legislative changes. These are just a few of the ways SHM continues to add value to hospitalist members every day.
Although we will certainly miss seeing each other in person, we are confident that the SHM team will meet and exceed expectations on content delivery and will take advantage of the virtual format to improve content access. We look forward to “seeing” you at SHM Converge this year and hope you take advantage of the enhanced delivery and access to an array of amazing content!
Dr. Scheurer is president of the Society of Hospital Medicine. She is a hospitalist and chief quality officer, MUSC Health System, Medical University of South Carolina, Charleston.
Crusted Scabies Presenting as White Superficial Onychomycosislike Lesions
To the Editor:
We report the case of an 83-year-old male nursing home resident with a history of end-stage renal disease who presented with multiple small white islands on the surface of the nail plate, similar to those seen in white superficial onychomycosis (Figure 1). Minimal subungual hyperkeratosis of the fingernails also was observed. Three digits were affected with no toenail involvement. Wet mount examination with potassium hydroxide 20% showed a mite (Figure 2A) and multiple eggs (Figure 2B). Treatment consisted of oral ivermectin 3 mg immediately and permethrin solution 5% applied under occlusion to each of the affected nails for 5 consecutive nights, which resulted in complete clearance of the lesion on the nail plate after 2 weeks.


Crusted scabies was first described as Norwegian scabies in 1848 by Danielsen and Boeck,1 and the name was later changed to crusted scabies in 1976 by Parish and Lumholt2 because there was no inherent connection between Norway and Norwegian scabies. It is a skin infestation of Sarcoptes scabiei var hominis and more commonly is seen in immunocompromised individuals such as the elderly and malnourished patients as well as those with diabetes mellitus and alcoholism.3,4 Patients typically present with widespread hyperkeratosis, mostly involving the palms and soles. Subungual hyperkeratosis and nail dystrophy also can be seen when nail involvement is present, and the scalp rarely is involved.5 Unlike common scabies, skin burrows and pruritus may be minimal or absent, thus making the diagnosis of crusted scabies more difficult than normal scabies.6 Diagnosis of crusted scabies is confirmed by direct microscopy, which demonstrates mites, eggs, or feces. Strict isolation of the patient is necessary, as the disease is very contagious. Treatment with oral ivermectin (1–3 doses of 3 mg at 14-day intervals) in combination with topical permethrin is effective.7
We present a case of crusted scabies with nail involvement that presented with white superficial onychomycosislike lesions. The patient’s nails were successfully treated with a combination of oral ivermectin and topical permethrin occlusion of the nails. In cases with subungual hyperkeratosis, nonsurgical nail avulsion with 40% urea cream or ointment has been used to improve the penetration of permethrin. Partial nail avulsion may be necessary if subungual hyperkeratosis or nail dystrophy becomes extreme.8
- Danielsen DG, Boeck W. Treatment of Leprosy or Greek Elephantiasis. JB Balliere; 1848.
- Parish L, Lumholt G. Crusted scabies: alias Norwegian scabies. Int J Dermatol. 1976;15:747-748.
- Centers for Disease Control and Prevention. Parasites: scabies. Updated November 2, 2010. Accessed January 17, 2021. https://www.cdc.gov/parasites/scabies/
- Roberts LJ, Huffam SE, Walton SF, et al. Crusted scabies: clinical and immunological findings in seventy-eight patient and a review of the literature. J Infect. 2005;50:375-381.
- Dourmisher AL, Serafimova DK, Dourmisher LA, et al. Crusted scabies of the scalp in dermatomyositis patients: three cases treated with oral ivermectin. Int J Dermatol. 1998;37:231-234.
- Barnes L, McCallister RE, Lucky AW. Crusted (Norwegian) scabies: occurrence in a child undergoing a bone marrow transplant. Arch Dermatol. 1987;123:95-97.
- Huffam SE, Currie BJ. Ivermectin for Sarcoptes scabiei hyperinfestation. Int J Infect Dis. 1998;2:152-154.
- De Paoli R, Mark SV. Crusted (Norwegian) scabies: treatment of nail involvement. J Am Acad Dermatol. 1987;17:136-138.
To the Editor:
We report the case of an 83-year-old male nursing home resident with a history of end-stage renal disease who presented with multiple small white islands on the surface of the nail plate, similar to those seen in white superficial onychomycosis (Figure 1). Minimal subungual hyperkeratosis of the fingernails also was observed. Three digits were affected with no toenail involvement. Wet mount examination with potassium hydroxide 20% showed a mite (Figure 2A) and multiple eggs (Figure 2B). Treatment consisted of oral ivermectin 3 mg immediately and permethrin solution 5% applied under occlusion to each of the affected nails for 5 consecutive nights, which resulted in complete clearance of the lesion on the nail plate after 2 weeks.


Crusted scabies was first described as Norwegian scabies in 1848 by Danielsen and Boeck,1 and the name was later changed to crusted scabies in 1976 by Parish and Lumholt2 because there was no inherent connection between Norway and Norwegian scabies. It is a skin infestation of Sarcoptes scabiei var hominis and more commonly is seen in immunocompromised individuals such as the elderly and malnourished patients as well as those with diabetes mellitus and alcoholism.3,4 Patients typically present with widespread hyperkeratosis, mostly involving the palms and soles. Subungual hyperkeratosis and nail dystrophy also can be seen when nail involvement is present, and the scalp rarely is involved.5 Unlike common scabies, skin burrows and pruritus may be minimal or absent, thus making the diagnosis of crusted scabies more difficult than normal scabies.6 Diagnosis of crusted scabies is confirmed by direct microscopy, which demonstrates mites, eggs, or feces. Strict isolation of the patient is necessary, as the disease is very contagious. Treatment with oral ivermectin (1–3 doses of 3 mg at 14-day intervals) in combination with topical permethrin is effective.7
We present a case of crusted scabies with nail involvement that presented with white superficial onychomycosislike lesions. The patient’s nails were successfully treated with a combination of oral ivermectin and topical permethrin occlusion of the nails. In cases with subungual hyperkeratosis, nonsurgical nail avulsion with 40% urea cream or ointment has been used to improve the penetration of permethrin. Partial nail avulsion may be necessary if subungual hyperkeratosis or nail dystrophy becomes extreme.8
To the Editor:
We report the case of an 83-year-old male nursing home resident with a history of end-stage renal disease who presented with multiple small white islands on the surface of the nail plate, similar to those seen in white superficial onychomycosis (Figure 1). Minimal subungual hyperkeratosis of the fingernails also was observed. Three digits were affected with no toenail involvement. Wet mount examination with potassium hydroxide 20% showed a mite (Figure 2A) and multiple eggs (Figure 2B). Treatment consisted of oral ivermectin 3 mg immediately and permethrin solution 5% applied under occlusion to each of the affected nails for 5 consecutive nights, which resulted in complete clearance of the lesion on the nail plate after 2 weeks.


Crusted scabies was first described as Norwegian scabies in 1848 by Danielsen and Boeck,1 and the name was later changed to crusted scabies in 1976 by Parish and Lumholt2 because there was no inherent connection between Norway and Norwegian scabies. It is a skin infestation of Sarcoptes scabiei var hominis and more commonly is seen in immunocompromised individuals such as the elderly and malnourished patients as well as those with diabetes mellitus and alcoholism.3,4 Patients typically present with widespread hyperkeratosis, mostly involving the palms and soles. Subungual hyperkeratosis and nail dystrophy also can be seen when nail involvement is present, and the scalp rarely is involved.5 Unlike common scabies, skin burrows and pruritus may be minimal or absent, thus making the diagnosis of crusted scabies more difficult than normal scabies.6 Diagnosis of crusted scabies is confirmed by direct microscopy, which demonstrates mites, eggs, or feces. Strict isolation of the patient is necessary, as the disease is very contagious. Treatment with oral ivermectin (1–3 doses of 3 mg at 14-day intervals) in combination with topical permethrin is effective.7
We present a case of crusted scabies with nail involvement that presented with white superficial onychomycosislike lesions. The patient’s nails were successfully treated with a combination of oral ivermectin and topical permethrin occlusion of the nails. In cases with subungual hyperkeratosis, nonsurgical nail avulsion with 40% urea cream or ointment has been used to improve the penetration of permethrin. Partial nail avulsion may be necessary if subungual hyperkeratosis or nail dystrophy becomes extreme.8
- Danielsen DG, Boeck W. Treatment of Leprosy or Greek Elephantiasis. JB Balliere; 1848.
- Parish L, Lumholt G. Crusted scabies: alias Norwegian scabies. Int J Dermatol. 1976;15:747-748.
- Centers for Disease Control and Prevention. Parasites: scabies. Updated November 2, 2010. Accessed January 17, 2021. https://www.cdc.gov/parasites/scabies/
- Roberts LJ, Huffam SE, Walton SF, et al. Crusted scabies: clinical and immunological findings in seventy-eight patient and a review of the literature. J Infect. 2005;50:375-381.
- Dourmisher AL, Serafimova DK, Dourmisher LA, et al. Crusted scabies of the scalp in dermatomyositis patients: three cases treated with oral ivermectin. Int J Dermatol. 1998;37:231-234.
- Barnes L, McCallister RE, Lucky AW. Crusted (Norwegian) scabies: occurrence in a child undergoing a bone marrow transplant. Arch Dermatol. 1987;123:95-97.
- Huffam SE, Currie BJ. Ivermectin for Sarcoptes scabiei hyperinfestation. Int J Infect Dis. 1998;2:152-154.
- De Paoli R, Mark SV. Crusted (Norwegian) scabies: treatment of nail involvement. J Am Acad Dermatol. 1987;17:136-138.
- Danielsen DG, Boeck W. Treatment of Leprosy or Greek Elephantiasis. JB Balliere; 1848.
- Parish L, Lumholt G. Crusted scabies: alias Norwegian scabies. Int J Dermatol. 1976;15:747-748.
- Centers for Disease Control and Prevention. Parasites: scabies. Updated November 2, 2010. Accessed January 17, 2021. https://www.cdc.gov/parasites/scabies/
- Roberts LJ, Huffam SE, Walton SF, et al. Crusted scabies: clinical and immunological findings in seventy-eight patient and a review of the literature. J Infect. 2005;50:375-381.
- Dourmisher AL, Serafimova DK, Dourmisher LA, et al. Crusted scabies of the scalp in dermatomyositis patients: three cases treated with oral ivermectin. Int J Dermatol. 1998;37:231-234.
- Barnes L, McCallister RE, Lucky AW. Crusted (Norwegian) scabies: occurrence in a child undergoing a bone marrow transplant. Arch Dermatol. 1987;123:95-97.
- Huffam SE, Currie BJ. Ivermectin for Sarcoptes scabiei hyperinfestation. Int J Infect Dis. 1998;2:152-154.
- De Paoli R, Mark SV. Crusted (Norwegian) scabies: treatment of nail involvement. J Am Acad Dermatol. 1987;17:136-138.
Practice Points
- Crusted scabies is asymptomatic; therefore, any white lesion at the surface of the nail should be scraped and examined with potassium hydroxide.
- Immunosuppressed patients are at risk for infection.
Recurrent Painful Nodules Following Synthol Injection to Enhance Bicep Volume
To the Editor:
A 28-year-old man presented to the dermatology clinic with red, tender, swollen nodules on the left arm of 5 days’ duration, which had been a recurrent issue involving both arms. He also experienced intermittent fatigue and mild myalgia but denied associated fevers or chills. Oral clindamycin prescribed by a local emergency department provided some improvement. Upon further questioning, the patient admitted to injecting an unknown substance into the muscles 10 years prior for the purpose of enhancing their volume and appearance. Physical examination revealed large bilateral biceps with firm, mobile, nontender, subcutaneous nodules and mild erythema on the inner aspects of the arms. An incisional biopsy of a left arm nodule was performed with tissue culture (Figure 1). Microscopic evaluation revealed mild dermal sclerosis with edema and sclerosis of fat septae (Figure 2A). The fat lobules contained granulomas with surrounding lymphocytes and clear holes noted within the histiocytic giant cells, indicating a likely foreign substance (Figure 2B). Immunohistochemical staining of the histiocytes with CD68 highlighted the clear vacuoles (Figure 3). Polarization examination, Alcian blue, periodic acid–Schiff, and acid-fast bacilli staining were negative. Bacterial, fungal, and mycobacterial tissue cultures and staining also were negative. The histologic findings of septal and lobular panniculitis with sclerosis and granulomatous inflammation in the clinical setting were consistent with a foreign body reaction secondary to synthol injection.
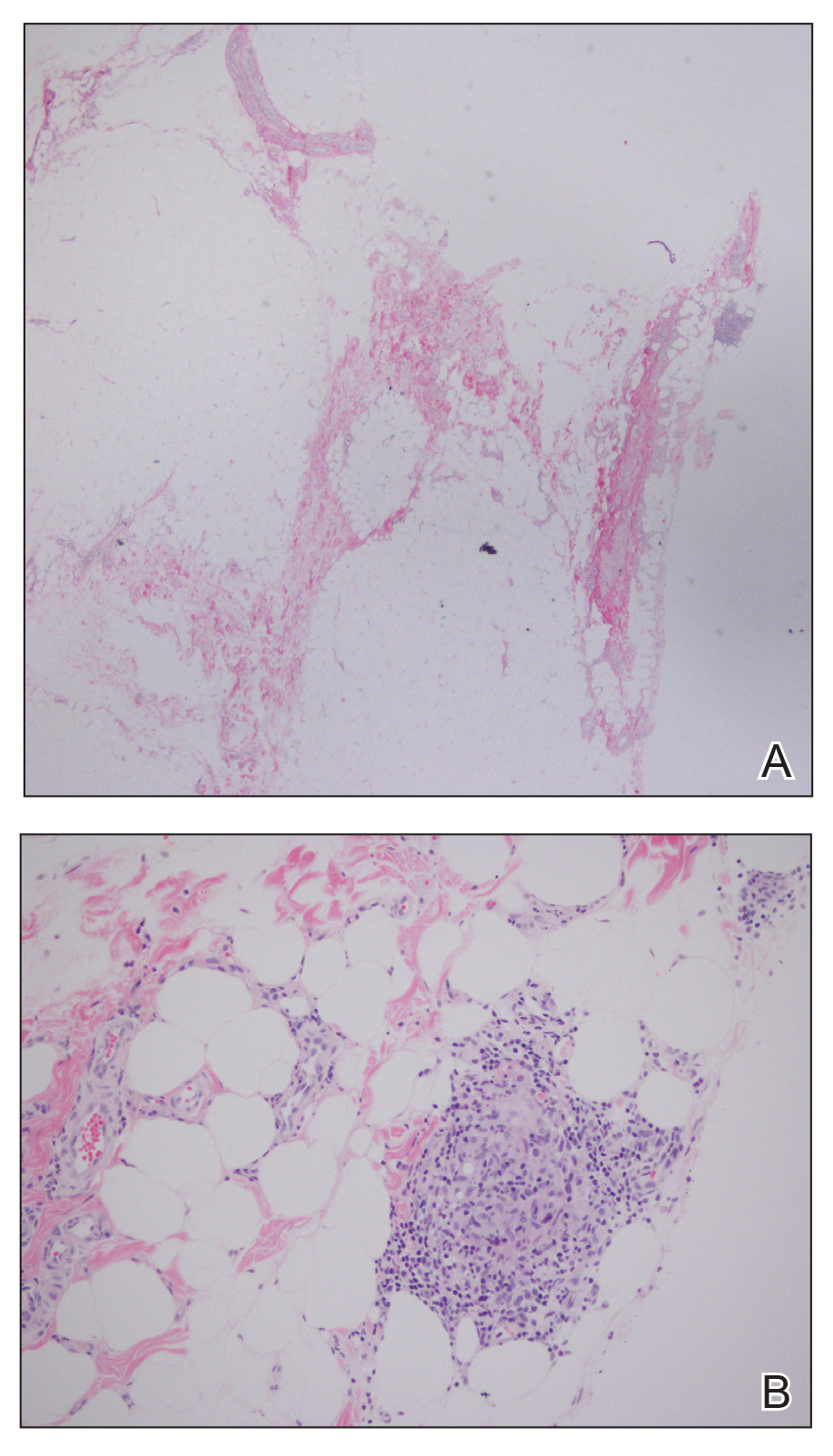
The willingness of athletes in competitive sports to undergo procedures or utilize substances for a competitive advantage despite both immediate and long-term consequences is well documented.1,2 In bodybuilding, use of anabolic steroids and intramuscular oil injections has been documented.3 The use of site enhancements in the form of “fillers” such as petroleum jelly and paraffin have been used for more than 100 years.4 The use of oil for volumetric site enhancement began in the 1960s in Italy with formebolone and evolved to the use of synthol in the 1990s.5 Synthol is a substance composed of 85% oil in the form of medium-chain triglycerides, 7.5% alcohol, and 7.5% lidocaine.6 The presumed mechanism of action of injected oils consists of an initial inflammatory response followed by fibrosis and chronic macrophagocytosis, ultimately leading to expanded volume in the subcutaneous tissue.7 These procedures are purely aesthetic with no increase in muscle strength or performance.
There are few cases in the literature of side effects from intramuscular synthol injections. In one report, a 29-year-old man presented with painful muscle fibrosis requiring open surgical excision of massively fibrotic bicep tissue.8 Another report documented a 45-year-old man who presented with spontaneous ulcerations on the biceps that initially were treated with antibiotics and compression therapy but eventually required surgical intervention and skin grafting.9 Complications have been more frequently reported from injections of other oils such as paraffin and sesame.10,11 Given the similar underlying mechanisms of action, injected oils share the local side effects of inflammation, infection, chronic wounds, and ulceration,9,10 as well as a systemic risk for embolization leading to pulmonary emboli, myocardial infarction, and stroke.6 Although no standard of care exists for the management of complications arising from intramuscular oil injections, treatments that have been employed include antibiotics, corticosteroids, wound care, and compression therapy; definitive treatment typically is surgical excision.6,8,9,11,12 Psychiatric evaluation also should be considered to evaluate for the possibility of body dysmorphic disorder and other associated psychiatric conditions.11
Pressure for a particular aesthetic appearance, both within and outside the world of competitive sports, has driven individuals to various methods of muscular enhancement. Volumetric site enhancements have become increasingly popular, in part due to the perceived lack of systemic side effects, such as those associated with anabolic steroids.8 However, most users are unaware of the notable short-term and long-term risks associated with intramuscular oil injections. Synthol is widely available on the Internet and easily can be purchased and injected by anyone.13 Medical providers should be aware of the possibility of aesthetic site enhancement use in their patients and be able to recognize and intervene in these cases to prevent chronic damage to muscle tissue and accompanying complications. Despite extensive commercialization of these products, few reports in the medical literature exist detailing the side effects of intramuscular oil injections, which may be contributing to the trivialization of these procedures by the general public.12
- Baron DA, Martin DM, Abol Magd S. Doping in sports and its spread to at-risk populations: an international review. World Psychiatry. 2007;6:118-123.
- Holt RIG, Erotokritou-Mulligan I, Sönksen PH. The history of doping and growth hormone abuse in sport. Growth Horm IGF Res. 2009;19:320-326.
- Figueiredo VC, Pedroso da Silva PR. Cosmetic doping—when anabolic-androgenic steroids are not enough. Subst Use Misuse. 2014;49:1163-1167.
- Glicenstein J. The first “fillers,” vaseline and paraffin. from miracle to disaster [in French]. Ann Chir Plast Esthet. 2007;52:157-161.
- Evans NA. Gym and tonic: a profile of 100 male steroid users. Br J Sports Med. 1997;31:54-58.
- Pupka A, Sikora J, Mauricz J, et al. The usage of synthol in the body building [in Polish]. Polim Med. 2009;39:63-65.
- Di Benedetto G, Pierangeli M, Scalise A, et al. Paraffin oil injection in the body: an obsolete and destructive procedure. Ann Plast Surg. 2002;49:391-396.
- Ghandourah S, Hofer MJ, Kiessling A, et al. Painful muscle fibrosis following synthol injections in a bodybuilder: a case report. J Med Case Rep. 2012;6:248.
- Ikander P, Nielsen AM, Sørensen JA. Injection of synthol in a bodybuilder can cause chronic wounds and ulceration [in Danish]. Ugeskr Laeger. 2015;177:V12140642.
- Henriksen TF, Løvenwald JB, Matzen SH. Paraffin oil injection in bodybuilders calls for preventive action [in Danish]. Ugeskr Laeger. 2010;172:219-220.
- Darsow U, Bruckbauer H, Worret WI, et al. Subcutaneous oleomas induced by self-injection of sesame seed oil for muscle augmentation. J Am Acad Dermatol. 2000;42(2, pt 1):292-294.
- Banke IJ, Prodinger PM, Waldt S, et al. Irreversible muscle damage in bodybuilding due to long-term intramuscular oil injection. Int J Sports Med. 2012;33:829-834.
- Hall M, Grogan S, Gough B. Bodybuilders’ accounts of synthol use: the construction of lay expertise online. J Health Psychol. 2016;21:1939-1948.
To the Editor:
A 28-year-old man presented to the dermatology clinic with red, tender, swollen nodules on the left arm of 5 days’ duration, which had been a recurrent issue involving both arms. He also experienced intermittent fatigue and mild myalgia but denied associated fevers or chills. Oral clindamycin prescribed by a local emergency department provided some improvement. Upon further questioning, the patient admitted to injecting an unknown substance into the muscles 10 years prior for the purpose of enhancing their volume and appearance. Physical examination revealed large bilateral biceps with firm, mobile, nontender, subcutaneous nodules and mild erythema on the inner aspects of the arms. An incisional biopsy of a left arm nodule was performed with tissue culture (Figure 1). Microscopic evaluation revealed mild dermal sclerosis with edema and sclerosis of fat septae (Figure 2A). The fat lobules contained granulomas with surrounding lymphocytes and clear holes noted within the histiocytic giant cells, indicating a likely foreign substance (Figure 2B). Immunohistochemical staining of the histiocytes with CD68 highlighted the clear vacuoles (Figure 3). Polarization examination, Alcian blue, periodic acid–Schiff, and acid-fast bacilli staining were negative. Bacterial, fungal, and mycobacterial tissue cultures and staining also were negative. The histologic findings of septal and lobular panniculitis with sclerosis and granulomatous inflammation in the clinical setting were consistent with a foreign body reaction secondary to synthol injection.



The willingness of athletes in competitive sports to undergo procedures or utilize substances for a competitive advantage despite both immediate and long-term consequences is well documented.1,2 In bodybuilding, use of anabolic steroids and intramuscular oil injections has been documented.3 The use of site enhancements in the form of “fillers” such as petroleum jelly and paraffin have been used for more than 100 years.4 The use of oil for volumetric site enhancement began in the 1960s in Italy with formebolone and evolved to the use of synthol in the 1990s.5 Synthol is a substance composed of 85% oil in the form of medium-chain triglycerides, 7.5% alcohol, and 7.5% lidocaine.6 The presumed mechanism of action of injected oils consists of an initial inflammatory response followed by fibrosis and chronic macrophagocytosis, ultimately leading to expanded volume in the subcutaneous tissue.7 These procedures are purely aesthetic with no increase in muscle strength or performance.
There are few cases in the literature of side effects from intramuscular synthol injections. In one report, a 29-year-old man presented with painful muscle fibrosis requiring open surgical excision of massively fibrotic bicep tissue.8 Another report documented a 45-year-old man who presented with spontaneous ulcerations on the biceps that initially were treated with antibiotics and compression therapy but eventually required surgical intervention and skin grafting.9 Complications have been more frequently reported from injections of other oils such as paraffin and sesame.10,11 Given the similar underlying mechanisms of action, injected oils share the local side effects of inflammation, infection, chronic wounds, and ulceration,9,10 as well as a systemic risk for embolization leading to pulmonary emboli, myocardial infarction, and stroke.6 Although no standard of care exists for the management of complications arising from intramuscular oil injections, treatments that have been employed include antibiotics, corticosteroids, wound care, and compression therapy; definitive treatment typically is surgical excision.6,8,9,11,12 Psychiatric evaluation also should be considered to evaluate for the possibility of body dysmorphic disorder and other associated psychiatric conditions.11
Pressure for a particular aesthetic appearance, both within and outside the world of competitive sports, has driven individuals to various methods of muscular enhancement. Volumetric site enhancements have become increasingly popular, in part due to the perceived lack of systemic side effects, such as those associated with anabolic steroids.8 However, most users are unaware of the notable short-term and long-term risks associated with intramuscular oil injections. Synthol is widely available on the Internet and easily can be purchased and injected by anyone.13 Medical providers should be aware of the possibility of aesthetic site enhancement use in their patients and be able to recognize and intervene in these cases to prevent chronic damage to muscle tissue and accompanying complications. Despite extensive commercialization of these products, few reports in the medical literature exist detailing the side effects of intramuscular oil injections, which may be contributing to the trivialization of these procedures by the general public.12
To the Editor:
A 28-year-old man presented to the dermatology clinic with red, tender, swollen nodules on the left arm of 5 days’ duration, which had been a recurrent issue involving both arms. He also experienced intermittent fatigue and mild myalgia but denied associated fevers or chills. Oral clindamycin prescribed by a local emergency department provided some improvement. Upon further questioning, the patient admitted to injecting an unknown substance into the muscles 10 years prior for the purpose of enhancing their volume and appearance. Physical examination revealed large bilateral biceps with firm, mobile, nontender, subcutaneous nodules and mild erythema on the inner aspects of the arms. An incisional biopsy of a left arm nodule was performed with tissue culture (Figure 1). Microscopic evaluation revealed mild dermal sclerosis with edema and sclerosis of fat septae (Figure 2A). The fat lobules contained granulomas with surrounding lymphocytes and clear holes noted within the histiocytic giant cells, indicating a likely foreign substance (Figure 2B). Immunohistochemical staining of the histiocytes with CD68 highlighted the clear vacuoles (Figure 3). Polarization examination, Alcian blue, periodic acid–Schiff, and acid-fast bacilli staining were negative. Bacterial, fungal, and mycobacterial tissue cultures and staining also were negative. The histologic findings of septal and lobular panniculitis with sclerosis and granulomatous inflammation in the clinical setting were consistent with a foreign body reaction secondary to synthol injection.



The willingness of athletes in competitive sports to undergo procedures or utilize substances for a competitive advantage despite both immediate and long-term consequences is well documented.1,2 In bodybuilding, use of anabolic steroids and intramuscular oil injections has been documented.3 The use of site enhancements in the form of “fillers” such as petroleum jelly and paraffin have been used for more than 100 years.4 The use of oil for volumetric site enhancement began in the 1960s in Italy with formebolone and evolved to the use of synthol in the 1990s.5 Synthol is a substance composed of 85% oil in the form of medium-chain triglycerides, 7.5% alcohol, and 7.5% lidocaine.6 The presumed mechanism of action of injected oils consists of an initial inflammatory response followed by fibrosis and chronic macrophagocytosis, ultimately leading to expanded volume in the subcutaneous tissue.7 These procedures are purely aesthetic with no increase in muscle strength or performance.
There are few cases in the literature of side effects from intramuscular synthol injections. In one report, a 29-year-old man presented with painful muscle fibrosis requiring open surgical excision of massively fibrotic bicep tissue.8 Another report documented a 45-year-old man who presented with spontaneous ulcerations on the biceps that initially were treated with antibiotics and compression therapy but eventually required surgical intervention and skin grafting.9 Complications have been more frequently reported from injections of other oils such as paraffin and sesame.10,11 Given the similar underlying mechanisms of action, injected oils share the local side effects of inflammation, infection, chronic wounds, and ulceration,9,10 as well as a systemic risk for embolization leading to pulmonary emboli, myocardial infarction, and stroke.6 Although no standard of care exists for the management of complications arising from intramuscular oil injections, treatments that have been employed include antibiotics, corticosteroids, wound care, and compression therapy; definitive treatment typically is surgical excision.6,8,9,11,12 Psychiatric evaluation also should be considered to evaluate for the possibility of body dysmorphic disorder and other associated psychiatric conditions.11
Pressure for a particular aesthetic appearance, both within and outside the world of competitive sports, has driven individuals to various methods of muscular enhancement. Volumetric site enhancements have become increasingly popular, in part due to the perceived lack of systemic side effects, such as those associated with anabolic steroids.8 However, most users are unaware of the notable short-term and long-term risks associated with intramuscular oil injections. Synthol is widely available on the Internet and easily can be purchased and injected by anyone.13 Medical providers should be aware of the possibility of aesthetic site enhancement use in their patients and be able to recognize and intervene in these cases to prevent chronic damage to muscle tissue and accompanying complications. Despite extensive commercialization of these products, few reports in the medical literature exist detailing the side effects of intramuscular oil injections, which may be contributing to the trivialization of these procedures by the general public.12
- Baron DA, Martin DM, Abol Magd S. Doping in sports and its spread to at-risk populations: an international review. World Psychiatry. 2007;6:118-123.
- Holt RIG, Erotokritou-Mulligan I, Sönksen PH. The history of doping and growth hormone abuse in sport. Growth Horm IGF Res. 2009;19:320-326.
- Figueiredo VC, Pedroso da Silva PR. Cosmetic doping—when anabolic-androgenic steroids are not enough. Subst Use Misuse. 2014;49:1163-1167.
- Glicenstein J. The first “fillers,” vaseline and paraffin. from miracle to disaster [in French]. Ann Chir Plast Esthet. 2007;52:157-161.
- Evans NA. Gym and tonic: a profile of 100 male steroid users. Br J Sports Med. 1997;31:54-58.
- Pupka A, Sikora J, Mauricz J, et al. The usage of synthol in the body building [in Polish]. Polim Med. 2009;39:63-65.
- Di Benedetto G, Pierangeli M, Scalise A, et al. Paraffin oil injection in the body: an obsolete and destructive procedure. Ann Plast Surg. 2002;49:391-396.
- Ghandourah S, Hofer MJ, Kiessling A, et al. Painful muscle fibrosis following synthol injections in a bodybuilder: a case report. J Med Case Rep. 2012;6:248.
- Ikander P, Nielsen AM, Sørensen JA. Injection of synthol in a bodybuilder can cause chronic wounds and ulceration [in Danish]. Ugeskr Laeger. 2015;177:V12140642.
- Henriksen TF, Løvenwald JB, Matzen SH. Paraffin oil injection in bodybuilders calls for preventive action [in Danish]. Ugeskr Laeger. 2010;172:219-220.
- Darsow U, Bruckbauer H, Worret WI, et al. Subcutaneous oleomas induced by self-injection of sesame seed oil for muscle augmentation. J Am Acad Dermatol. 2000;42(2, pt 1):292-294.
- Banke IJ, Prodinger PM, Waldt S, et al. Irreversible muscle damage in bodybuilding due to long-term intramuscular oil injection. Int J Sports Med. 2012;33:829-834.
- Hall M, Grogan S, Gough B. Bodybuilders’ accounts of synthol use: the construction of lay expertise online. J Health Psychol. 2016;21:1939-1948.
- Baron DA, Martin DM, Abol Magd S. Doping in sports and its spread to at-risk populations: an international review. World Psychiatry. 2007;6:118-123.
- Holt RIG, Erotokritou-Mulligan I, Sönksen PH. The history of doping and growth hormone abuse in sport. Growth Horm IGF Res. 2009;19:320-326.
- Figueiredo VC, Pedroso da Silva PR. Cosmetic doping—when anabolic-androgenic steroids are not enough. Subst Use Misuse. 2014;49:1163-1167.
- Glicenstein J. The first “fillers,” vaseline and paraffin. from miracle to disaster [in French]. Ann Chir Plast Esthet. 2007;52:157-161.
- Evans NA. Gym and tonic: a profile of 100 male steroid users. Br J Sports Med. 1997;31:54-58.
- Pupka A, Sikora J, Mauricz J, et al. The usage of synthol in the body building [in Polish]. Polim Med. 2009;39:63-65.
- Di Benedetto G, Pierangeli M, Scalise A, et al. Paraffin oil injection in the body: an obsolete and destructive procedure. Ann Plast Surg. 2002;49:391-396.
- Ghandourah S, Hofer MJ, Kiessling A, et al. Painful muscle fibrosis following synthol injections in a bodybuilder: a case report. J Med Case Rep. 2012;6:248.
- Ikander P, Nielsen AM, Sørensen JA. Injection of synthol in a bodybuilder can cause chronic wounds and ulceration [in Danish]. Ugeskr Laeger. 2015;177:V12140642.
- Henriksen TF, Løvenwald JB, Matzen SH. Paraffin oil injection in bodybuilders calls for preventive action [in Danish]. Ugeskr Laeger. 2010;172:219-220.
- Darsow U, Bruckbauer H, Worret WI, et al. Subcutaneous oleomas induced by self-injection of sesame seed oil for muscle augmentation. J Am Acad Dermatol. 2000;42(2, pt 1):292-294.
- Banke IJ, Prodinger PM, Waldt S, et al. Irreversible muscle damage in bodybuilding due to long-term intramuscular oil injection. Int J Sports Med. 2012;33:829-834.
- Hall M, Grogan S, Gough B. Bodybuilders’ accounts of synthol use: the construction of lay expertise online. J Health Psychol. 2016;21:1939-1948.
Practice Points
- The use of injectable volumetric site enhancers in the form of oils to improve the aesthetic appearance of muscles has been prevalent for decades despite potentially serious adverse reactions.
- Complications from these procedures are underrecognized in the medical setting, perhaps owing to the trivialization of these procedures by the general public.
7 key changes: The 2021 child and adolescent immunization schedules
Each February, the Centers for Disease Control and Prevention, along with multiple professional organizations, releases an updated Recommended Child and Adolescent Immunization Schedule.
Recent years have seen fewer changes in the vaccine schedule, mostly with adjustments based on products coming on or off the market, and sometimes with slight changes in recommendations. This year is no different, with mostly minor changes in store. As most practitioners know, having quick access to the tables that accompany the recommendations is always handy. Table 1 contains the typical, recommended immunization schedule. Table 2 contains the catch-up provisions, and Table 3 provides guidance on vaccines for special circumstances and for children with specific medical conditions.
2021 childhood and adolescent immunization schedule
One update is a recommendation that patients with egg allergies who had symptoms more extensive than hives should receive the influenza vaccine in a medical setting where severe allergic reactions or anaphylaxis can be recognized and treated, with the exclusion of two specific preparations, Flublok and Flucelvax.
In regard to the live attenuated influenza vaccine (LAIV), there are several points of reinforcement. First, the nomenclature has generally been changed to “LAIV4” throughout the document because only quadrivalent preparations are available. There are specific recommendations that patients should not receive LAIV4 if they recently took antiviral medication for influenza, with “lockout” periods lasting from 2 days to 17 days, depending on the antiviral preparation used. In addition, there is an emphasis on not using LAIV4 for children younger than 2 years.
Two updates to the meningococcal group B vaccine are worth reviewing. The first is that children aged 10 years or older with complement deficiency, complement inhibitor use, or asplenia should receive a meningitis B booster dose beginning 1 year after completion of the primary series, with boosters thereafter every 2 or 3 years as long as that patient remains at greater risk. Another recommendation for patients 10 years or older is that, even if they have received a primary series of meningitis B vaccines, they should receive a booster dose in the setting of an outbreak if it has been 1 year or more since completion of their primary series.
Recommendations have generally been relaxed for tetanus prophylaxis in older children, indicating that individuals requiring tetanus prophylaxis or their 10-year tetanus booster after receipt of at least one Tdap vaccine can receive either tetanus-diphtheria toxoid or Tdap.
COVID-19 vaccines
Although childhood vaccination against COVID-19 is still currently limited to adolescents involved in clinical trials, pediatricians surely are getting peppered with questions from parents about whether they should be vaccinated and what to make of the recent reports about allergic reactions. Fortunately, there are several resources for pediatricians. First, two reports point out that true anaphylactic reactions to COVID-19 vaccines appear quite rare. The reported data on Pfizer-developed mRNA vaccine demonstrated an anaphylaxis rate of approximately 2 cases per 1 million doses administered. Among the 21 recipients who experienced anaphylaxis (out of over 11 million total doses administered), fully one third had a history of anaphylaxis episodes. The report also reviews vaccine reactions that were reported but were not classified as anaphylaxis, pointing out that when reporting vaccine reactions, we should be very careful in the nomenclature we use.
Reporting on the Moderna mRNA vaccine showed anaphylaxis rates of about 2.5 per 1 million doses, with 50% of the recipients who experienced true anaphylaxis having a history of anaphylaxis. Most of those who experienced anaphylaxis (90% in the Moderna group and 86% in the Pfizer group) exhibited symptoms of anaphylaxis within 30 minutes of receiving the vaccine. The take-home point, and the current CDC recommendation, is that many individuals, even those with a history of anaphylaxis, can still receive COVID-19 vaccines. The rates of observed anaphylaxis after COVID vaccination are far below population rates of a history of allergy or severe allergic reactions. When coupled with an estimated mortality rate of 0.5%-1% for SARS-CoV-2 disease, that CDC recommends that we encourage people, even those with severe allergies, to get vaccinated.
One clear caveat is that individuals with a history of severe anaphylaxis, and even those concerned about allergies, should be observed for a longer period after vaccination (at least 30 minutes) than the 15 minutes recommended for the general population. In addition, individuals with a specific anaphylactic reaction or severe allergic reaction to any injectable vaccine should confer with an immunologist before considering vaccination.
Another useful resource is a column published by the American Medical Association that walks through some talking points for providers when discussing whether a patient should receive COVID-19 vaccination. Advice is offered on answering patient questions about which preparation to get, what side effects to watch for, and how to report an adverse reaction. Providers are reminded to urge patients to complete whichever series they begin (get that second dose!), and that they currently should not have to pay for a vaccine. FAQ resource pages are available for patients and health care providers.
More vaccine news: HPV and influenza
Meanwhile, published vaccine reports provide evidence from the field to demonstrate the benefits of vaccination. A study published in the New England Journal of Medicine reported on the effectiveness of human papillomavirus (HPV) vaccine in a Swedish cohort. The report evaluated females aged between 10 and 30 years beginning in 2006 and followed them through 2017, comparing rates of invasive cervical cancer among the group who received one or more HPV vaccine doses with the group who receive none. Even without adjustment, the raw rate of invasive cervical cancer in the vaccinated group was half of that in the unvaccinated group. After full adjustment, some populations experienced incident rate ratios that were greater than 80% reduced. The largest reduction, and therefore the biggest benefit, was among those who received the HPV vaccine before age 17.
A report from the United States looking at the 2018-2019 influenza season demonstrated a vaccine effectiveness rate against hospitalization of 41% and 51% against any ED visit related to influenza. The authors note that there was considerable drift in the influenza A type that appeared late in the influenza season, reducing the overall effectiveness, but that the vaccine was still largely effective.
William T. Basco Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Each February, the Centers for Disease Control and Prevention, along with multiple professional organizations, releases an updated Recommended Child and Adolescent Immunization Schedule.
Recent years have seen fewer changes in the vaccine schedule, mostly with adjustments based on products coming on or off the market, and sometimes with slight changes in recommendations. This year is no different, with mostly minor changes in store. As most practitioners know, having quick access to the tables that accompany the recommendations is always handy. Table 1 contains the typical, recommended immunization schedule. Table 2 contains the catch-up provisions, and Table 3 provides guidance on vaccines for special circumstances and for children with specific medical conditions.
2021 childhood and adolescent immunization schedule
One update is a recommendation that patients with egg allergies who had symptoms more extensive than hives should receive the influenza vaccine in a medical setting where severe allergic reactions or anaphylaxis can be recognized and treated, with the exclusion of two specific preparations, Flublok and Flucelvax.
In regard to the live attenuated influenza vaccine (LAIV), there are several points of reinforcement. First, the nomenclature has generally been changed to “LAIV4” throughout the document because only quadrivalent preparations are available. There are specific recommendations that patients should not receive LAIV4 if they recently took antiviral medication for influenza, with “lockout” periods lasting from 2 days to 17 days, depending on the antiviral preparation used. In addition, there is an emphasis on not using LAIV4 for children younger than 2 years.
Two updates to the meningococcal group B vaccine are worth reviewing. The first is that children aged 10 years or older with complement deficiency, complement inhibitor use, or asplenia should receive a meningitis B booster dose beginning 1 year after completion of the primary series, with boosters thereafter every 2 or 3 years as long as that patient remains at greater risk. Another recommendation for patients 10 years or older is that, even if they have received a primary series of meningitis B vaccines, they should receive a booster dose in the setting of an outbreak if it has been 1 year or more since completion of their primary series.
Recommendations have generally been relaxed for tetanus prophylaxis in older children, indicating that individuals requiring tetanus prophylaxis or their 10-year tetanus booster after receipt of at least one Tdap vaccine can receive either tetanus-diphtheria toxoid or Tdap.
COVID-19 vaccines
Although childhood vaccination against COVID-19 is still currently limited to adolescents involved in clinical trials, pediatricians surely are getting peppered with questions from parents about whether they should be vaccinated and what to make of the recent reports about allergic reactions. Fortunately, there are several resources for pediatricians. First, two reports point out that true anaphylactic reactions to COVID-19 vaccines appear quite rare. The reported data on Pfizer-developed mRNA vaccine demonstrated an anaphylaxis rate of approximately 2 cases per 1 million doses administered. Among the 21 recipients who experienced anaphylaxis (out of over 11 million total doses administered), fully one third had a history of anaphylaxis episodes. The report also reviews vaccine reactions that were reported but were not classified as anaphylaxis, pointing out that when reporting vaccine reactions, we should be very careful in the nomenclature we use.
Reporting on the Moderna mRNA vaccine showed anaphylaxis rates of about 2.5 per 1 million doses, with 50% of the recipients who experienced true anaphylaxis having a history of anaphylaxis. Most of those who experienced anaphylaxis (90% in the Moderna group and 86% in the Pfizer group) exhibited symptoms of anaphylaxis within 30 minutes of receiving the vaccine. The take-home point, and the current CDC recommendation, is that many individuals, even those with a history of anaphylaxis, can still receive COVID-19 vaccines. The rates of observed anaphylaxis after COVID vaccination are far below population rates of a history of allergy or severe allergic reactions. When coupled with an estimated mortality rate of 0.5%-1% for SARS-CoV-2 disease, that CDC recommends that we encourage people, even those with severe allergies, to get vaccinated.
One clear caveat is that individuals with a history of severe anaphylaxis, and even those concerned about allergies, should be observed for a longer period after vaccination (at least 30 minutes) than the 15 minutes recommended for the general population. In addition, individuals with a specific anaphylactic reaction or severe allergic reaction to any injectable vaccine should confer with an immunologist before considering vaccination.
Another useful resource is a column published by the American Medical Association that walks through some talking points for providers when discussing whether a patient should receive COVID-19 vaccination. Advice is offered on answering patient questions about which preparation to get, what side effects to watch for, and how to report an adverse reaction. Providers are reminded to urge patients to complete whichever series they begin (get that second dose!), and that they currently should not have to pay for a vaccine. FAQ resource pages are available for patients and health care providers.
More vaccine news: HPV and influenza
Meanwhile, published vaccine reports provide evidence from the field to demonstrate the benefits of vaccination. A study published in the New England Journal of Medicine reported on the effectiveness of human papillomavirus (HPV) vaccine in a Swedish cohort. The report evaluated females aged between 10 and 30 years beginning in 2006 and followed them through 2017, comparing rates of invasive cervical cancer among the group who received one or more HPV vaccine doses with the group who receive none. Even without adjustment, the raw rate of invasive cervical cancer in the vaccinated group was half of that in the unvaccinated group. After full adjustment, some populations experienced incident rate ratios that were greater than 80% reduced. The largest reduction, and therefore the biggest benefit, was among those who received the HPV vaccine before age 17.
A report from the United States looking at the 2018-2019 influenza season demonstrated a vaccine effectiveness rate against hospitalization of 41% and 51% against any ED visit related to influenza. The authors note that there was considerable drift in the influenza A type that appeared late in the influenza season, reducing the overall effectiveness, but that the vaccine was still largely effective.
William T. Basco Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Each February, the Centers for Disease Control and Prevention, along with multiple professional organizations, releases an updated Recommended Child and Adolescent Immunization Schedule.
Recent years have seen fewer changes in the vaccine schedule, mostly with adjustments based on products coming on or off the market, and sometimes with slight changes in recommendations. This year is no different, with mostly minor changes in store. As most practitioners know, having quick access to the tables that accompany the recommendations is always handy. Table 1 contains the typical, recommended immunization schedule. Table 2 contains the catch-up provisions, and Table 3 provides guidance on vaccines for special circumstances and for children with specific medical conditions.
2021 childhood and adolescent immunization schedule
One update is a recommendation that patients with egg allergies who had symptoms more extensive than hives should receive the influenza vaccine in a medical setting where severe allergic reactions or anaphylaxis can be recognized and treated, with the exclusion of two specific preparations, Flublok and Flucelvax.
In regard to the live attenuated influenza vaccine (LAIV), there are several points of reinforcement. First, the nomenclature has generally been changed to “LAIV4” throughout the document because only quadrivalent preparations are available. There are specific recommendations that patients should not receive LAIV4 if they recently took antiviral medication for influenza, with “lockout” periods lasting from 2 days to 17 days, depending on the antiviral preparation used. In addition, there is an emphasis on not using LAIV4 for children younger than 2 years.
Two updates to the meningococcal group B vaccine are worth reviewing. The first is that children aged 10 years or older with complement deficiency, complement inhibitor use, or asplenia should receive a meningitis B booster dose beginning 1 year after completion of the primary series, with boosters thereafter every 2 or 3 years as long as that patient remains at greater risk. Another recommendation for patients 10 years or older is that, even if they have received a primary series of meningitis B vaccines, they should receive a booster dose in the setting of an outbreak if it has been 1 year or more since completion of their primary series.
Recommendations have generally been relaxed for tetanus prophylaxis in older children, indicating that individuals requiring tetanus prophylaxis or their 10-year tetanus booster after receipt of at least one Tdap vaccine can receive either tetanus-diphtheria toxoid or Tdap.
COVID-19 vaccines
Although childhood vaccination against COVID-19 is still currently limited to adolescents involved in clinical trials, pediatricians surely are getting peppered with questions from parents about whether they should be vaccinated and what to make of the recent reports about allergic reactions. Fortunately, there are several resources for pediatricians. First, two reports point out that true anaphylactic reactions to COVID-19 vaccines appear quite rare. The reported data on Pfizer-developed mRNA vaccine demonstrated an anaphylaxis rate of approximately 2 cases per 1 million doses administered. Among the 21 recipients who experienced anaphylaxis (out of over 11 million total doses administered), fully one third had a history of anaphylaxis episodes. The report also reviews vaccine reactions that were reported but were not classified as anaphylaxis, pointing out that when reporting vaccine reactions, we should be very careful in the nomenclature we use.
Reporting on the Moderna mRNA vaccine showed anaphylaxis rates of about 2.5 per 1 million doses, with 50% of the recipients who experienced true anaphylaxis having a history of anaphylaxis. Most of those who experienced anaphylaxis (90% in the Moderna group and 86% in the Pfizer group) exhibited symptoms of anaphylaxis within 30 minutes of receiving the vaccine. The take-home point, and the current CDC recommendation, is that many individuals, even those with a history of anaphylaxis, can still receive COVID-19 vaccines. The rates of observed anaphylaxis after COVID vaccination are far below population rates of a history of allergy or severe allergic reactions. When coupled with an estimated mortality rate of 0.5%-1% for SARS-CoV-2 disease, that CDC recommends that we encourage people, even those with severe allergies, to get vaccinated.
One clear caveat is that individuals with a history of severe anaphylaxis, and even those concerned about allergies, should be observed for a longer period after vaccination (at least 30 minutes) than the 15 minutes recommended for the general population. In addition, individuals with a specific anaphylactic reaction or severe allergic reaction to any injectable vaccine should confer with an immunologist before considering vaccination.
Another useful resource is a column published by the American Medical Association that walks through some talking points for providers when discussing whether a patient should receive COVID-19 vaccination. Advice is offered on answering patient questions about which preparation to get, what side effects to watch for, and how to report an adverse reaction. Providers are reminded to urge patients to complete whichever series they begin (get that second dose!), and that they currently should not have to pay for a vaccine. FAQ resource pages are available for patients and health care providers.
More vaccine news: HPV and influenza
Meanwhile, published vaccine reports provide evidence from the field to demonstrate the benefits of vaccination. A study published in the New England Journal of Medicine reported on the effectiveness of human papillomavirus (HPV) vaccine in a Swedish cohort. The report evaluated females aged between 10 and 30 years beginning in 2006 and followed them through 2017, comparing rates of invasive cervical cancer among the group who received one or more HPV vaccine doses with the group who receive none. Even without adjustment, the raw rate of invasive cervical cancer in the vaccinated group was half of that in the unvaccinated group. After full adjustment, some populations experienced incident rate ratios that were greater than 80% reduced. The largest reduction, and therefore the biggest benefit, was among those who received the HPV vaccine before age 17.
A report from the United States looking at the 2018-2019 influenza season demonstrated a vaccine effectiveness rate against hospitalization of 41% and 51% against any ED visit related to influenza. The authors note that there was considerable drift in the influenza A type that appeared late in the influenza season, reducing the overall effectiveness, but that the vaccine was still largely effective.
William T. Basco Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Psychiatrists’ happiness, well-being hit hard by COVID-19
Events of the past year have taken a huge toll on the happiness, wellness, and lifestyles of many, but especially those in the health care field, including psychiatrists.
The newly released Medscape Psychiatrist Lifestyle, Happiness & Burnout Report 2021 reveals how psychiatrists are coping with burnout and trying to maintain personal wellness, and how they view their workplaces and their futures amid the ongoing COVID-19 pandemic.
Before the pandemic hit in March 2020, 84% of psychiatrists who responded to the survey reported being happy outside of work, similar to the percentage (82%) of physicians overall.
But as the pandemic has worn on, feelings have shifted, and there are clear signs of strain on those in the health care field. Now, just over half (55%) of psychiatrists say they are happy outside of work, similar to the percentage (58%) of physicians overall.
Perhaps not surprising given the specific challenges of COVID-19, infectious disease physicians, pulmonologists, rheumatologists, and intensivists currently rank lowest in happiness outside of work.
Anxiety, depression, burnout
With the ongoing COVID-19 pandemic, more than three quarters (77%) of psychiatrists surveyed report experiencing some degree of anxiety about their future, the same percentage as for physicians overall.
This year, more psychiatrists reported being either burned out or burned out and depressed (41% vs. 35% last year). About two-thirds of psychiatrists said burnout has had at least a moderate impact on their lives; 5% consider the impact so severe that they are thinking of leaving medicine altogether.
The majority of burned-out psychiatrists (63%) said they felt that way even before the pandemic began; for about one-third (37%), burnout set in after the pandemic began.
in the workplace (39%) and spending too many hours at work (37%).
Psychiatrists’ top tactic to cope with burnout is talking with family or friends (53%), followed by isolating themselves from others (51%), sleeping (45%), and exercising (43%); 42% said they eat junk food to cope; 35% play music; and 25% drink alcohol.
Most psychiatrists (63%) suffering burnout and/or depression don’t plan on seeking professional help. About one-third are currently seeking help or plan to do so, the highest proportion among all specialties.
Considering their symptoms not severe enough (57%) and feeling that they could deal with the problem on their own (41%) are the top reasons for not seeking professional help; 36% said they were too busy to get help, and 17% said they didn’t want to risk disclosing a problem.
Fifteen percent of psychiatrists who are burned out, depressed, or both have contemplated suicide, and 2% have attempted suicide.
Striving for work-life balance
Work-life balance is the most pressing workplace issue for 45% of psychiatrists, and 44% would sacrifice some of their salary for better work-life balance. These figures are about the same for physicians overall.
Forty-seven percent of psychiatrists take 3-4 weeks of vacation each year; 16% take 5 or more weeks. In this there was no change from last year’s report.
About one-third (35%) of psychiatrists generally make time to focus on their own well-being, the same proportion as physicians overall.
About two-thirds (68%) of psychiatrists exercise two or more times per week. Half of psychiatrists said they are currently trying to lose weight; about one-quarter are trying to maintain their current weight.
About one-quarter (26%) of psychiatrists said they do not drink alcohol at all; 17% have five or more drinks per week.
Most psychiatrists are currently in a committed relationship, with 81% either married or living with a partner. Among psychiatrists who are married or living with a partner, 43% are with someone who also works in medicine. About 81% of psychiatrists say their marriages are very good or good. These percentages are similar to those of physicians overall (85%).
Most psychiatrists (58%) spend up to 10 hours per week online for personal reasons; 82% spend this amount of time online each week for work.
It’s likely that the amount of time spent online for work will increase, given the pandemic-fueled surge in telemedicine. Yet even when their personal and professional Internet use are combined, psychiatrists, on average, spend far less time online than the nearly 7 hours per day of the average Internet user, according to recent data.
Findings from the latest happiness, wellness, and lifestyle survey are based on 12,339 Medscape member physicians practicing in the United States who completed an online survey conducted between Aug. 30 and Nov. 5, 2020.
A version of this article first appeared on Medscape.com.
Events of the past year have taken a huge toll on the happiness, wellness, and lifestyles of many, but especially those in the health care field, including psychiatrists.
The newly released Medscape Psychiatrist Lifestyle, Happiness & Burnout Report 2021 reveals how psychiatrists are coping with burnout and trying to maintain personal wellness, and how they view their workplaces and their futures amid the ongoing COVID-19 pandemic.
Before the pandemic hit in March 2020, 84% of psychiatrists who responded to the survey reported being happy outside of work, similar to the percentage (82%) of physicians overall.
But as the pandemic has worn on, feelings have shifted, and there are clear signs of strain on those in the health care field. Now, just over half (55%) of psychiatrists say they are happy outside of work, similar to the percentage (58%) of physicians overall.
Perhaps not surprising given the specific challenges of COVID-19, infectious disease physicians, pulmonologists, rheumatologists, and intensivists currently rank lowest in happiness outside of work.
Anxiety, depression, burnout
With the ongoing COVID-19 pandemic, more than three quarters (77%) of psychiatrists surveyed report experiencing some degree of anxiety about their future, the same percentage as for physicians overall.
This year, more psychiatrists reported being either burned out or burned out and depressed (41% vs. 35% last year). About two-thirds of psychiatrists said burnout has had at least a moderate impact on their lives; 5% consider the impact so severe that they are thinking of leaving medicine altogether.
The majority of burned-out psychiatrists (63%) said they felt that way even before the pandemic began; for about one-third (37%), burnout set in after the pandemic began.
in the workplace (39%) and spending too many hours at work (37%).
Psychiatrists’ top tactic to cope with burnout is talking with family or friends (53%), followed by isolating themselves from others (51%), sleeping (45%), and exercising (43%); 42% said they eat junk food to cope; 35% play music; and 25% drink alcohol.
Most psychiatrists (63%) suffering burnout and/or depression don’t plan on seeking professional help. About one-third are currently seeking help or plan to do so, the highest proportion among all specialties.
Considering their symptoms not severe enough (57%) and feeling that they could deal with the problem on their own (41%) are the top reasons for not seeking professional help; 36% said they were too busy to get help, and 17% said they didn’t want to risk disclosing a problem.
Fifteen percent of psychiatrists who are burned out, depressed, or both have contemplated suicide, and 2% have attempted suicide.
Striving for work-life balance
Work-life balance is the most pressing workplace issue for 45% of psychiatrists, and 44% would sacrifice some of their salary for better work-life balance. These figures are about the same for physicians overall.
Forty-seven percent of psychiatrists take 3-4 weeks of vacation each year; 16% take 5 or more weeks. In this there was no change from last year’s report.
About one-third (35%) of psychiatrists generally make time to focus on their own well-being, the same proportion as physicians overall.
About two-thirds (68%) of psychiatrists exercise two or more times per week. Half of psychiatrists said they are currently trying to lose weight; about one-quarter are trying to maintain their current weight.
About one-quarter (26%) of psychiatrists said they do not drink alcohol at all; 17% have five or more drinks per week.
Most psychiatrists are currently in a committed relationship, with 81% either married or living with a partner. Among psychiatrists who are married or living with a partner, 43% are with someone who also works in medicine. About 81% of psychiatrists say their marriages are very good or good. These percentages are similar to those of physicians overall (85%).
Most psychiatrists (58%) spend up to 10 hours per week online for personal reasons; 82% spend this amount of time online each week for work.
It’s likely that the amount of time spent online for work will increase, given the pandemic-fueled surge in telemedicine. Yet even when their personal and professional Internet use are combined, psychiatrists, on average, spend far less time online than the nearly 7 hours per day of the average Internet user, according to recent data.
Findings from the latest happiness, wellness, and lifestyle survey are based on 12,339 Medscape member physicians practicing in the United States who completed an online survey conducted between Aug. 30 and Nov. 5, 2020.
A version of this article first appeared on Medscape.com.
Events of the past year have taken a huge toll on the happiness, wellness, and lifestyles of many, but especially those in the health care field, including psychiatrists.
The newly released Medscape Psychiatrist Lifestyle, Happiness & Burnout Report 2021 reveals how psychiatrists are coping with burnout and trying to maintain personal wellness, and how they view their workplaces and their futures amid the ongoing COVID-19 pandemic.
Before the pandemic hit in March 2020, 84% of psychiatrists who responded to the survey reported being happy outside of work, similar to the percentage (82%) of physicians overall.
But as the pandemic has worn on, feelings have shifted, and there are clear signs of strain on those in the health care field. Now, just over half (55%) of psychiatrists say they are happy outside of work, similar to the percentage (58%) of physicians overall.
Perhaps not surprising given the specific challenges of COVID-19, infectious disease physicians, pulmonologists, rheumatologists, and intensivists currently rank lowest in happiness outside of work.
Anxiety, depression, burnout
With the ongoing COVID-19 pandemic, more than three quarters (77%) of psychiatrists surveyed report experiencing some degree of anxiety about their future, the same percentage as for physicians overall.
This year, more psychiatrists reported being either burned out or burned out and depressed (41% vs. 35% last year). About two-thirds of psychiatrists said burnout has had at least a moderate impact on their lives; 5% consider the impact so severe that they are thinking of leaving medicine altogether.
The majority of burned-out psychiatrists (63%) said they felt that way even before the pandemic began; for about one-third (37%), burnout set in after the pandemic began.
in the workplace (39%) and spending too many hours at work (37%).
Psychiatrists’ top tactic to cope with burnout is talking with family or friends (53%), followed by isolating themselves from others (51%), sleeping (45%), and exercising (43%); 42% said they eat junk food to cope; 35% play music; and 25% drink alcohol.
Most psychiatrists (63%) suffering burnout and/or depression don’t plan on seeking professional help. About one-third are currently seeking help or plan to do so, the highest proportion among all specialties.
Considering their symptoms not severe enough (57%) and feeling that they could deal with the problem on their own (41%) are the top reasons for not seeking professional help; 36% said they were too busy to get help, and 17% said they didn’t want to risk disclosing a problem.
Fifteen percent of psychiatrists who are burned out, depressed, or both have contemplated suicide, and 2% have attempted suicide.
Striving for work-life balance
Work-life balance is the most pressing workplace issue for 45% of psychiatrists, and 44% would sacrifice some of their salary for better work-life balance. These figures are about the same for physicians overall.
Forty-seven percent of psychiatrists take 3-4 weeks of vacation each year; 16% take 5 or more weeks. In this there was no change from last year’s report.
About one-third (35%) of psychiatrists generally make time to focus on their own well-being, the same proportion as physicians overall.
About two-thirds (68%) of psychiatrists exercise two or more times per week. Half of psychiatrists said they are currently trying to lose weight; about one-quarter are trying to maintain their current weight.
About one-quarter (26%) of psychiatrists said they do not drink alcohol at all; 17% have five or more drinks per week.
Most psychiatrists are currently in a committed relationship, with 81% either married or living with a partner. Among psychiatrists who are married or living with a partner, 43% are with someone who also works in medicine. About 81% of psychiatrists say their marriages are very good or good. These percentages are similar to those of physicians overall (85%).
Most psychiatrists (58%) spend up to 10 hours per week online for personal reasons; 82% spend this amount of time online each week for work.
It’s likely that the amount of time spent online for work will increase, given the pandemic-fueled surge in telemedicine. Yet even when their personal and professional Internet use are combined, psychiatrists, on average, spend far less time online than the nearly 7 hours per day of the average Internet user, according to recent data.
Findings from the latest happiness, wellness, and lifestyle survey are based on 12,339 Medscape member physicians practicing in the United States who completed an online survey conducted between Aug. 30 and Nov. 5, 2020.
A version of this article first appeared on Medscape.com.
Endometriosis-associated ovarian cancer
Endometriosis, which affects 1 in 10 women, is one of the most common conditions that gynecologists treat. It is known to cause pain, pelvic adhesive disease, endometriotic cyst formation, and infertility. However, even more sinister, it also increases a woman’s risk for the development of epithelial ovarian cancer (known as endometriosis-associated ovarian cancer or EAOC). A woman with endometriosis has a two- to threefold increased risk of developing epithelial ovarian cancer, compared with nonaffected women.1 This risk appears to be concentrated in the premenopausal age group, particularly the fifth decade of life. After menopause their risk of developing cancer returns to a baseline level.
EAOC classically presents as clear cell or endometrioid adenocarcinomas, rather than high-grade serous carcinomas. However, low-grade serous carcinomas are also frequently observed in this cohort.2,3 Unlike high-grade serous carcinoma, EAOC is more likely to be diagnosed at an early stage, with the majority at stage I or II, and prognosis is better. After matching for age and stage with cases of high-grade serous carcinoma, there is improved disease-free and overall survival observed among cases of EAOC of clear cell and endometrioid histologic cell types.4 The phenomenon of dual primaries (synchronous endometrial and ovarian cancer) occurs more frequently in EAOC than it does in patients with nonendometriosis-related high-grade serous cancer (25% vs. 4%).
The genomics of these endometriosis-associated cancers are quite distinct. Similar to benign endometriosis implants, EAOC is associated with genomic mutations in ARID1A, PIK3CA, and PTEN, as well as progesterone resistance.1,2 Multiple studies have shown that the adjacent eutopic endometrium carries similar gene mutations as those found in both benign endometriotic implants and EAOC.2 This may explain the higher incidence (twofold) of endometrial cancer in patients with endometriosis as well as the increased incidence of dual ovarian and endometrial cancer primaries.
Just as there are multiple theories regarding the mechanism of benign endometriosis, we have theories rather than conclusions regarding the origins of EAOC. One such theory is that it develops from malignant transformation in an existing endometriotic cyst.5 Endometriotic cysts provide an iron-rich environment which promotes reactive oxygen species that promote carcinogenesis by inducing gene mutations and epigenetic alterations. However, if prolonged exposure to oxidative stress within endometriotic cysts were to be the cause for EAOC, we would expect to see a progressively increasing incidence of ovarian cancer over time in patients with expectantly managed cysts. However, in cases of expectant management, an initial, early, increased risk for cancer within the first 5 years is followed by a subsequent decreasing incidence over time.6 This early incidence spike suggests that some endometriotic cysts may have been misclassified as benign, then rapidly declare themselves as malignant during the observation period rather than a transformation into malignancy from a benign endometrioma over time.
An alternative, and favored, theory for the origins of EAOC are that endometrial cells with carcinogenic genomic alterations reflux through the fallopian tubes during menstruation and settle onto the ovarian epithelium which itself is damaged from recent ovulation thus providing an environment that is highly suitable for oncogenesis.2 Genomic analyses of both the eutopic endometrium and malignant cells in patients with EAOC have shown that both tissues contain the same genomic alterations.1 Given that menstruation, including retrograde menstruation, ends after menopause, this mechanism supports the observation that EAOC is predominantly a malignancy of premenopausal women. Additionally, salpingectomy and hysterectomy confers a protective effect on the development of EAOC, theoretically by preventing the retrograde transfer of these mutant progenitor endometrial cells. Furthermore, the factors that increase the number of menstrual cycles (such as an early age of menarche and delayed or nonchildbearing states) increases the risk for EAOC and factors that inhibit menstruation, such as oral contraceptive pill use, appear to decrease its risk.
EAOC most commonly arises in the ovary, and not in the deep endometriosis implants of adjacent pelvic structures (such as the anterior and posterior cul de sac and pelvic peritoneum). It is suggested that the ovary itself provides a uniquely favorable environment for carcinogenesis. As stated above, it is hypothesized that refluxed endometrial cells, carrying important progenitor mutations, may become trapped in the tissues of traumatized ovarian epithelium, ripe with inflammatory changes, post ovulation.2 This microenvironment may promote the development of malignancy.
Given these theories and their supporting evidence, how can we attempt to reduce the incidence of this cancer for our patients with endometriosis? Despite their increased risk for ovarian and endometrial cancers, current recommendations do not support routine cancer screening in women with endometriosis.7 However, risk-mitigation strategies can still be pursued. Hormonal contraceptives to decrease ovulation and menstrual cycling are protective against ovarian cancer and are also helpful in mitigating the symptoms of endometriosis. While removal of endometriotic cysts may not, in and of itself, be a strategy to prevent EAOC, it is still generally recommended because these cysts are commonly a source of pain and infertility. While they do not appear to undergo malignant transformation, it can be difficult to definitively rule out an early ovarian cancer in these complex ovarian cysts, particularly as they are often associated with tumor marker abnormalities such as elevations in CA 125. Therefore, if surgical excision of an endometriotic cyst is not performed, it should be closely followed for at least 5 years to ensure it is a benign structure. If surgery is pursued and ovarian preservation is desired, removal of the fallopian tubes and uterus can help mitigate the risk for EAOC.8
Endometriosis is a morbid condition for many young women. In addition to causing pain and infertility it increases a woman’s risk for ovarian and endometrial cancer, particularly ovarian clear cell, endometrioid, and low-grade serous cancers and synchronous endometrial and ovarian cancers. Endometriotic cysts should be removed or closely monitored, and clinicians should discuss treatment options that minimize frequency of ovulation and menstruation events as a preventative strategy.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill.
References
1. Endocrinology. 2019;160(3):626-38.
2. Cancers. 2020;12(6):1676.
3. Lancet Oncol. 2012;13:385-94.
4. Gynecol Oncol. 2014;132(3):760-6.
5. Redox Rep. 2016;21:119-26.
6. Int. J Clin Oncol. 2020;25:51-8.
7. Hum Reprod. 2013;28:1552-68.
8. J Natl Cancer Inst. 2019;111:1097-103.
Endometriosis, which affects 1 in 10 women, is one of the most common conditions that gynecologists treat. It is known to cause pain, pelvic adhesive disease, endometriotic cyst formation, and infertility. However, even more sinister, it also increases a woman’s risk for the development of epithelial ovarian cancer (known as endometriosis-associated ovarian cancer or EAOC). A woman with endometriosis has a two- to threefold increased risk of developing epithelial ovarian cancer, compared with nonaffected women.1 This risk appears to be concentrated in the premenopausal age group, particularly the fifth decade of life. After menopause their risk of developing cancer returns to a baseline level.
EAOC classically presents as clear cell or endometrioid adenocarcinomas, rather than high-grade serous carcinomas. However, low-grade serous carcinomas are also frequently observed in this cohort.2,3 Unlike high-grade serous carcinoma, EAOC is more likely to be diagnosed at an early stage, with the majority at stage I or II, and prognosis is better. After matching for age and stage with cases of high-grade serous carcinoma, there is improved disease-free and overall survival observed among cases of EAOC of clear cell and endometrioid histologic cell types.4 The phenomenon of dual primaries (synchronous endometrial and ovarian cancer) occurs more frequently in EAOC than it does in patients with nonendometriosis-related high-grade serous cancer (25% vs. 4%).
The genomics of these endometriosis-associated cancers are quite distinct. Similar to benign endometriosis implants, EAOC is associated with genomic mutations in ARID1A, PIK3CA, and PTEN, as well as progesterone resistance.1,2 Multiple studies have shown that the adjacent eutopic endometrium carries similar gene mutations as those found in both benign endometriotic implants and EAOC.2 This may explain the higher incidence (twofold) of endometrial cancer in patients with endometriosis as well as the increased incidence of dual ovarian and endometrial cancer primaries.
Just as there are multiple theories regarding the mechanism of benign endometriosis, we have theories rather than conclusions regarding the origins of EAOC. One such theory is that it develops from malignant transformation in an existing endometriotic cyst.5 Endometriotic cysts provide an iron-rich environment which promotes reactive oxygen species that promote carcinogenesis by inducing gene mutations and epigenetic alterations. However, if prolonged exposure to oxidative stress within endometriotic cysts were to be the cause for EAOC, we would expect to see a progressively increasing incidence of ovarian cancer over time in patients with expectantly managed cysts. However, in cases of expectant management, an initial, early, increased risk for cancer within the first 5 years is followed by a subsequent decreasing incidence over time.6 This early incidence spike suggests that some endometriotic cysts may have been misclassified as benign, then rapidly declare themselves as malignant during the observation period rather than a transformation into malignancy from a benign endometrioma over time.
An alternative, and favored, theory for the origins of EAOC are that endometrial cells with carcinogenic genomic alterations reflux through the fallopian tubes during menstruation and settle onto the ovarian epithelium which itself is damaged from recent ovulation thus providing an environment that is highly suitable for oncogenesis.2 Genomic analyses of both the eutopic endometrium and malignant cells in patients with EAOC have shown that both tissues contain the same genomic alterations.1 Given that menstruation, including retrograde menstruation, ends after menopause, this mechanism supports the observation that EAOC is predominantly a malignancy of premenopausal women. Additionally, salpingectomy and hysterectomy confers a protective effect on the development of EAOC, theoretically by preventing the retrograde transfer of these mutant progenitor endometrial cells. Furthermore, the factors that increase the number of menstrual cycles (such as an early age of menarche and delayed or nonchildbearing states) increases the risk for EAOC and factors that inhibit menstruation, such as oral contraceptive pill use, appear to decrease its risk.
EAOC most commonly arises in the ovary, and not in the deep endometriosis implants of adjacent pelvic structures (such as the anterior and posterior cul de sac and pelvic peritoneum). It is suggested that the ovary itself provides a uniquely favorable environment for carcinogenesis. As stated above, it is hypothesized that refluxed endometrial cells, carrying important progenitor mutations, may become trapped in the tissues of traumatized ovarian epithelium, ripe with inflammatory changes, post ovulation.2 This microenvironment may promote the development of malignancy.
Given these theories and their supporting evidence, how can we attempt to reduce the incidence of this cancer for our patients with endometriosis? Despite their increased risk for ovarian and endometrial cancers, current recommendations do not support routine cancer screening in women with endometriosis.7 However, risk-mitigation strategies can still be pursued. Hormonal contraceptives to decrease ovulation and menstrual cycling are protective against ovarian cancer and are also helpful in mitigating the symptoms of endometriosis. While removal of endometriotic cysts may not, in and of itself, be a strategy to prevent EAOC, it is still generally recommended because these cysts are commonly a source of pain and infertility. While they do not appear to undergo malignant transformation, it can be difficult to definitively rule out an early ovarian cancer in these complex ovarian cysts, particularly as they are often associated with tumor marker abnormalities such as elevations in CA 125. Therefore, if surgical excision of an endometriotic cyst is not performed, it should be closely followed for at least 5 years to ensure it is a benign structure. If surgery is pursued and ovarian preservation is desired, removal of the fallopian tubes and uterus can help mitigate the risk for EAOC.8
Endometriosis is a morbid condition for many young women. In addition to causing pain and infertility it increases a woman’s risk for ovarian and endometrial cancer, particularly ovarian clear cell, endometrioid, and low-grade serous cancers and synchronous endometrial and ovarian cancers. Endometriotic cysts should be removed or closely monitored, and clinicians should discuss treatment options that minimize frequency of ovulation and menstruation events as a preventative strategy.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill.
References
1. Endocrinology. 2019;160(3):626-38.
2. Cancers. 2020;12(6):1676.
3. Lancet Oncol. 2012;13:385-94.
4. Gynecol Oncol. 2014;132(3):760-6.
5. Redox Rep. 2016;21:119-26.
6. Int. J Clin Oncol. 2020;25:51-8.
7. Hum Reprod. 2013;28:1552-68.
8. J Natl Cancer Inst. 2019;111:1097-103.
Endometriosis, which affects 1 in 10 women, is one of the most common conditions that gynecologists treat. It is known to cause pain, pelvic adhesive disease, endometriotic cyst formation, and infertility. However, even more sinister, it also increases a woman’s risk for the development of epithelial ovarian cancer (known as endometriosis-associated ovarian cancer or EAOC). A woman with endometriosis has a two- to threefold increased risk of developing epithelial ovarian cancer, compared with nonaffected women.1 This risk appears to be concentrated in the premenopausal age group, particularly the fifth decade of life. After menopause their risk of developing cancer returns to a baseline level.
EAOC classically presents as clear cell or endometrioid adenocarcinomas, rather than high-grade serous carcinomas. However, low-grade serous carcinomas are also frequently observed in this cohort.2,3 Unlike high-grade serous carcinoma, EAOC is more likely to be diagnosed at an early stage, with the majority at stage I or II, and prognosis is better. After matching for age and stage with cases of high-grade serous carcinoma, there is improved disease-free and overall survival observed among cases of EAOC of clear cell and endometrioid histologic cell types.4 The phenomenon of dual primaries (synchronous endometrial and ovarian cancer) occurs more frequently in EAOC than it does in patients with nonendometriosis-related high-grade serous cancer (25% vs. 4%).
The genomics of these endometriosis-associated cancers are quite distinct. Similar to benign endometriosis implants, EAOC is associated with genomic mutations in ARID1A, PIK3CA, and PTEN, as well as progesterone resistance.1,2 Multiple studies have shown that the adjacent eutopic endometrium carries similar gene mutations as those found in both benign endometriotic implants and EAOC.2 This may explain the higher incidence (twofold) of endometrial cancer in patients with endometriosis as well as the increased incidence of dual ovarian and endometrial cancer primaries.
Just as there are multiple theories regarding the mechanism of benign endometriosis, we have theories rather than conclusions regarding the origins of EAOC. One such theory is that it develops from malignant transformation in an existing endometriotic cyst.5 Endometriotic cysts provide an iron-rich environment which promotes reactive oxygen species that promote carcinogenesis by inducing gene mutations and epigenetic alterations. However, if prolonged exposure to oxidative stress within endometriotic cysts were to be the cause for EAOC, we would expect to see a progressively increasing incidence of ovarian cancer over time in patients with expectantly managed cysts. However, in cases of expectant management, an initial, early, increased risk for cancer within the first 5 years is followed by a subsequent decreasing incidence over time.6 This early incidence spike suggests that some endometriotic cysts may have been misclassified as benign, then rapidly declare themselves as malignant during the observation period rather than a transformation into malignancy from a benign endometrioma over time.
An alternative, and favored, theory for the origins of EAOC are that endometrial cells with carcinogenic genomic alterations reflux through the fallopian tubes during menstruation and settle onto the ovarian epithelium which itself is damaged from recent ovulation thus providing an environment that is highly suitable for oncogenesis.2 Genomic analyses of both the eutopic endometrium and malignant cells in patients with EAOC have shown that both tissues contain the same genomic alterations.1 Given that menstruation, including retrograde menstruation, ends after menopause, this mechanism supports the observation that EAOC is predominantly a malignancy of premenopausal women. Additionally, salpingectomy and hysterectomy confers a protective effect on the development of EAOC, theoretically by preventing the retrograde transfer of these mutant progenitor endometrial cells. Furthermore, the factors that increase the number of menstrual cycles (such as an early age of menarche and delayed or nonchildbearing states) increases the risk for EAOC and factors that inhibit menstruation, such as oral contraceptive pill use, appear to decrease its risk.
EAOC most commonly arises in the ovary, and not in the deep endometriosis implants of adjacent pelvic structures (such as the anterior and posterior cul de sac and pelvic peritoneum). It is suggested that the ovary itself provides a uniquely favorable environment for carcinogenesis. As stated above, it is hypothesized that refluxed endometrial cells, carrying important progenitor mutations, may become trapped in the tissues of traumatized ovarian epithelium, ripe with inflammatory changes, post ovulation.2 This microenvironment may promote the development of malignancy.
Given these theories and their supporting evidence, how can we attempt to reduce the incidence of this cancer for our patients with endometriosis? Despite their increased risk for ovarian and endometrial cancers, current recommendations do not support routine cancer screening in women with endometriosis.7 However, risk-mitigation strategies can still be pursued. Hormonal contraceptives to decrease ovulation and menstrual cycling are protective against ovarian cancer and are also helpful in mitigating the symptoms of endometriosis. While removal of endometriotic cysts may not, in and of itself, be a strategy to prevent EAOC, it is still generally recommended because these cysts are commonly a source of pain and infertility. While they do not appear to undergo malignant transformation, it can be difficult to definitively rule out an early ovarian cancer in these complex ovarian cysts, particularly as they are often associated with tumor marker abnormalities such as elevations in CA 125. Therefore, if surgical excision of an endometriotic cyst is not performed, it should be closely followed for at least 5 years to ensure it is a benign structure. If surgery is pursued and ovarian preservation is desired, removal of the fallopian tubes and uterus can help mitigate the risk for EAOC.8
Endometriosis is a morbid condition for many young women. In addition to causing pain and infertility it increases a woman’s risk for ovarian and endometrial cancer, particularly ovarian clear cell, endometrioid, and low-grade serous cancers and synchronous endometrial and ovarian cancers. Endometriotic cysts should be removed or closely monitored, and clinicians should discuss treatment options that minimize frequency of ovulation and menstruation events as a preventative strategy.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill.
References
1. Endocrinology. 2019;160(3):626-38.
2. Cancers. 2020;12(6):1676.
3. Lancet Oncol. 2012;13:385-94.
4. Gynecol Oncol. 2014;132(3):760-6.
5. Redox Rep. 2016;21:119-26.
6. Int. J Clin Oncol. 2020;25:51-8.
7. Hum Reprod. 2013;28:1552-68.
8. J Natl Cancer Inst. 2019;111:1097-103.
New cases of child COVID-19 drop for fifth straight week
The fifth consecutive week with a decline has the number of new COVID-19 cases in children at its lowest level since late October, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
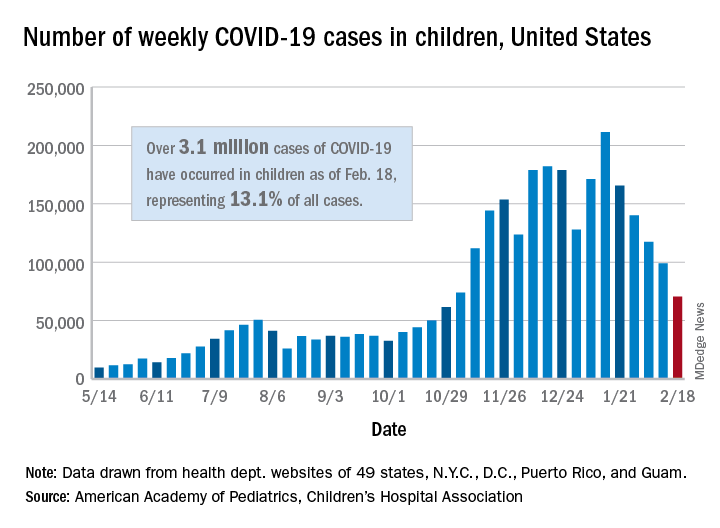
, when 61,000 cases were reported, the AAP and CHA said in their weekly COVID-19 report.
The cumulative number of COVID-19 cases in children is now just over 3.1 million, which represents 13.1% of cases among all ages in the United States, based on data gathered from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
More children in California (439,000) have been infected than in any other state, while Illinois (176,000), Florida (145,000), Tennessee (137,000), Arizona (127,000), Ohio (121,000), and Pennsylvania (111,000) are the only other states with more than 100,000 cases, the AAP/CHA report shows.
Proportionally, the children of Wyoming have been hardest hit: Pediatric cases represent 19.4% of all cases in the state. The other four states with proportions of 18% or more are Alaska, Vermont, South Carolina, and Tennessee. Cumulative rates, however, tell a somewhat different story, as North Dakota leads with just over 8,500 cases per 100,000 children, followed by Tennessee (7,700 per 100,000) and Rhode Island (7,000 per 100,000), the AAP and CHA said.
Deaths in children, which had not been following the trend of fewer new cases over the last few weeks, dropped below double digits for the first time in a month. The six deaths that occurred during the week of Feb. 12-18 bring the total to 247 since the start of the pandemic in the 43 states, along with New York City and Guam, that are reporting such data, according to the report.
The fifth consecutive week with a decline has the number of new COVID-19 cases in children at its lowest level since late October, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

, when 61,000 cases were reported, the AAP and CHA said in their weekly COVID-19 report.
The cumulative number of COVID-19 cases in children is now just over 3.1 million, which represents 13.1% of cases among all ages in the United States, based on data gathered from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
More children in California (439,000) have been infected than in any other state, while Illinois (176,000), Florida (145,000), Tennessee (137,000), Arizona (127,000), Ohio (121,000), and Pennsylvania (111,000) are the only other states with more than 100,000 cases, the AAP/CHA report shows.
Proportionally, the children of Wyoming have been hardest hit: Pediatric cases represent 19.4% of all cases in the state. The other four states with proportions of 18% or more are Alaska, Vermont, South Carolina, and Tennessee. Cumulative rates, however, tell a somewhat different story, as North Dakota leads with just over 8,500 cases per 100,000 children, followed by Tennessee (7,700 per 100,000) and Rhode Island (7,000 per 100,000), the AAP and CHA said.
Deaths in children, which had not been following the trend of fewer new cases over the last few weeks, dropped below double digits for the first time in a month. The six deaths that occurred during the week of Feb. 12-18 bring the total to 247 since the start of the pandemic in the 43 states, along with New York City and Guam, that are reporting such data, according to the report.
The fifth consecutive week with a decline has the number of new COVID-19 cases in children at its lowest level since late October, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

, when 61,000 cases were reported, the AAP and CHA said in their weekly COVID-19 report.
The cumulative number of COVID-19 cases in children is now just over 3.1 million, which represents 13.1% of cases among all ages in the United States, based on data gathered from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
More children in California (439,000) have been infected than in any other state, while Illinois (176,000), Florida (145,000), Tennessee (137,000), Arizona (127,000), Ohio (121,000), and Pennsylvania (111,000) are the only other states with more than 100,000 cases, the AAP/CHA report shows.
Proportionally, the children of Wyoming have been hardest hit: Pediatric cases represent 19.4% of all cases in the state. The other four states with proportions of 18% or more are Alaska, Vermont, South Carolina, and Tennessee. Cumulative rates, however, tell a somewhat different story, as North Dakota leads with just over 8,500 cases per 100,000 children, followed by Tennessee (7,700 per 100,000) and Rhode Island (7,000 per 100,000), the AAP and CHA said.
Deaths in children, which had not been following the trend of fewer new cases over the last few weeks, dropped below double digits for the first time in a month. The six deaths that occurred during the week of Feb. 12-18 bring the total to 247 since the start of the pandemic in the 43 states, along with New York City and Guam, that are reporting such data, according to the report.