User login
Mutations on LRRK2 gene modify risks for both Crohn’s and Parkinson’s
Crohn’s and Parkinson’s diseases seem to share a common genetic risk pathway, mediated by mutations in a gene that modify disease risk in both directions.
A multidisciplinary collaboration of researchers have discovered that the Crohn’s risk variant N2081D in the leucine-rich repeat kinase 2 (LRRK2) gene lies close to variant G2019S, the major genetic risk factor for both sporadic and familial Parkinson’s. However, protective mutations also occur in the LRRK2 gene, and Ken Y. Hui, MD, of Yale University, New Haven, Conn., and his associates identified two that, when combined, significantly decrease the risk of Crohn’s.
The team first discovered the association in an exome sequencing study of 50 Crohn’s patients of Ashkenazi Jewish descent. This identified more than 4,000 potentially high-yield mutations, which they then examined in a pure Ashkenazi cohort of 1,477 Crohn’s patients and 2,614 healthy controls.
In this phase, the researchers found that N2081D in LRRK2 was associated with a 73% increased odds of Crohn’s. The LRRK2 N551K variant was associated with protection against Crohn’s, reducing odds for the disease by 35% (odds ratio, 0.65), as was R1398H, which conferred a 29% reduction in the likelihood of developing the disease (OR, 0.71). It has been known that these two mutations can combine to provide a protective genetic signature, and the team investigated this compound interaction as well.
After repeated analyses in large, unique case-control cohorts and tissue samples, they determined that the N2081D allele increased the odds of Crohn’s by 70% in the Ashkenazi subjects and by 60% in non-Ashkenazi subjects. The allele was associated with a 10% increased likelihood of Parkinson’s among the Ashkenazi cohort and a 30% increased likelihood among the non-Ashkenazi cohort.
The researchers also examined the two protective alleles, N551K and R1398H, in these further analyses. In the Ashkenazi cohort, N551K decreased the likelihood of Crohn’s by 33%, and it decreased the likelihood of Parkinson’s by 23% in non-Ashkenazi subjects (OR, 0.67 and 0.77, respectively).
In the Ashkenazi cohort, R1398H reduced the odds of Crohn’s by 29% and the odds of Parkinson’s by 16% (OR, 0.71 and 0.84, respectively). In the non-Ashkenazi cohort, the risk reduction was not significant.
Finally, they examined the effect of the mutations on macrophages isolated from four Crohn’s patients who carried the N2081D allele, as well as five patients with the two protective mutations N551K and R1398H and four patients with no mutation. When the macrophages were put in nutrient starvation, those from the N2081D carriers exhibited impaired resting acetylation of alpha-tubulin and poor response to cellular stress. Decreased alpha-tubulin acetylation is associated with poor protein transport through the cytoskeleton. “In contrast, the highest basal acetylation of alpha-tubulin was detected in macrophages of noncarriers and carriers of the protective N551K + R1398H mutations,” the investigators wrote.
“Our study strongly implicates the contribution of LRRK2 in Crohn’s disease risk,” they concluded. “The LRRK2 N2081D risk allele and the N551K/R1398H protective alleles, as well as numerous other variants within the LRRK2 locus, revealed shared genetic effects between Crohn’s and Parkinson’s risk, providing a potential biological basis for clinical co-occurrence. Our findings suggest that LRRK2 may be a useful target for developing drugs to treat Crohn’s disease.”
The research was supported by a variety of grants from the National Institutes of Health, the National Science Foundation, and various other foundations. Five of the authors reported consulting for various pharmaceutical companies or companies selling genetic information products. None of the other authors had relevant disclosures.
SOURCE: Hui K et al. Sci Transl Med. 2018. doi: 10.1126/scitranslmed.aai7795
Crohn’s and Parkinson’s diseases seem to share a common genetic risk pathway, mediated by mutations in a gene that modify disease risk in both directions.
A multidisciplinary collaboration of researchers have discovered that the Crohn’s risk variant N2081D in the leucine-rich repeat kinase 2 (LRRK2) gene lies close to variant G2019S, the major genetic risk factor for both sporadic and familial Parkinson’s. However, protective mutations also occur in the LRRK2 gene, and Ken Y. Hui, MD, of Yale University, New Haven, Conn., and his associates identified two that, when combined, significantly decrease the risk of Crohn’s.
The team first discovered the association in an exome sequencing study of 50 Crohn’s patients of Ashkenazi Jewish descent. This identified more than 4,000 potentially high-yield mutations, which they then examined in a pure Ashkenazi cohort of 1,477 Crohn’s patients and 2,614 healthy controls.
In this phase, the researchers found that N2081D in LRRK2 was associated with a 73% increased odds of Crohn’s. The LRRK2 N551K variant was associated with protection against Crohn’s, reducing odds for the disease by 35% (odds ratio, 0.65), as was R1398H, which conferred a 29% reduction in the likelihood of developing the disease (OR, 0.71). It has been known that these two mutations can combine to provide a protective genetic signature, and the team investigated this compound interaction as well.
After repeated analyses in large, unique case-control cohorts and tissue samples, they determined that the N2081D allele increased the odds of Crohn’s by 70% in the Ashkenazi subjects and by 60% in non-Ashkenazi subjects. The allele was associated with a 10% increased likelihood of Parkinson’s among the Ashkenazi cohort and a 30% increased likelihood among the non-Ashkenazi cohort.
The researchers also examined the two protective alleles, N551K and R1398H, in these further analyses. In the Ashkenazi cohort, N551K decreased the likelihood of Crohn’s by 33%, and it decreased the likelihood of Parkinson’s by 23% in non-Ashkenazi subjects (OR, 0.67 and 0.77, respectively).
In the Ashkenazi cohort, R1398H reduced the odds of Crohn’s by 29% and the odds of Parkinson’s by 16% (OR, 0.71 and 0.84, respectively). In the non-Ashkenazi cohort, the risk reduction was not significant.
Finally, they examined the effect of the mutations on macrophages isolated from four Crohn’s patients who carried the N2081D allele, as well as five patients with the two protective mutations N551K and R1398H and four patients with no mutation. When the macrophages were put in nutrient starvation, those from the N2081D carriers exhibited impaired resting acetylation of alpha-tubulin and poor response to cellular stress. Decreased alpha-tubulin acetylation is associated with poor protein transport through the cytoskeleton. “In contrast, the highest basal acetylation of alpha-tubulin was detected in macrophages of noncarriers and carriers of the protective N551K + R1398H mutations,” the investigators wrote.
“Our study strongly implicates the contribution of LRRK2 in Crohn’s disease risk,” they concluded. “The LRRK2 N2081D risk allele and the N551K/R1398H protective alleles, as well as numerous other variants within the LRRK2 locus, revealed shared genetic effects between Crohn’s and Parkinson’s risk, providing a potential biological basis for clinical co-occurrence. Our findings suggest that LRRK2 may be a useful target for developing drugs to treat Crohn’s disease.”
The research was supported by a variety of grants from the National Institutes of Health, the National Science Foundation, and various other foundations. Five of the authors reported consulting for various pharmaceutical companies or companies selling genetic information products. None of the other authors had relevant disclosures.
SOURCE: Hui K et al. Sci Transl Med. 2018. doi: 10.1126/scitranslmed.aai7795
Crohn’s and Parkinson’s diseases seem to share a common genetic risk pathway, mediated by mutations in a gene that modify disease risk in both directions.
A multidisciplinary collaboration of researchers have discovered that the Crohn’s risk variant N2081D in the leucine-rich repeat kinase 2 (LRRK2) gene lies close to variant G2019S, the major genetic risk factor for both sporadic and familial Parkinson’s. However, protective mutations also occur in the LRRK2 gene, and Ken Y. Hui, MD, of Yale University, New Haven, Conn., and his associates identified two that, when combined, significantly decrease the risk of Crohn’s.
The team first discovered the association in an exome sequencing study of 50 Crohn’s patients of Ashkenazi Jewish descent. This identified more than 4,000 potentially high-yield mutations, which they then examined in a pure Ashkenazi cohort of 1,477 Crohn’s patients and 2,614 healthy controls.
In this phase, the researchers found that N2081D in LRRK2 was associated with a 73% increased odds of Crohn’s. The LRRK2 N551K variant was associated with protection against Crohn’s, reducing odds for the disease by 35% (odds ratio, 0.65), as was R1398H, which conferred a 29% reduction in the likelihood of developing the disease (OR, 0.71). It has been known that these two mutations can combine to provide a protective genetic signature, and the team investigated this compound interaction as well.
After repeated analyses in large, unique case-control cohorts and tissue samples, they determined that the N2081D allele increased the odds of Crohn’s by 70% in the Ashkenazi subjects and by 60% in non-Ashkenazi subjects. The allele was associated with a 10% increased likelihood of Parkinson’s among the Ashkenazi cohort and a 30% increased likelihood among the non-Ashkenazi cohort.
The researchers also examined the two protective alleles, N551K and R1398H, in these further analyses. In the Ashkenazi cohort, N551K decreased the likelihood of Crohn’s by 33%, and it decreased the likelihood of Parkinson’s by 23% in non-Ashkenazi subjects (OR, 0.67 and 0.77, respectively).
In the Ashkenazi cohort, R1398H reduced the odds of Crohn’s by 29% and the odds of Parkinson’s by 16% (OR, 0.71 and 0.84, respectively). In the non-Ashkenazi cohort, the risk reduction was not significant.
Finally, they examined the effect of the mutations on macrophages isolated from four Crohn’s patients who carried the N2081D allele, as well as five patients with the two protective mutations N551K and R1398H and four patients with no mutation. When the macrophages were put in nutrient starvation, those from the N2081D carriers exhibited impaired resting acetylation of alpha-tubulin and poor response to cellular stress. Decreased alpha-tubulin acetylation is associated with poor protein transport through the cytoskeleton. “In contrast, the highest basal acetylation of alpha-tubulin was detected in macrophages of noncarriers and carriers of the protective N551K + R1398H mutations,” the investigators wrote.
“Our study strongly implicates the contribution of LRRK2 in Crohn’s disease risk,” they concluded. “The LRRK2 N2081D risk allele and the N551K/R1398H protective alleles, as well as numerous other variants within the LRRK2 locus, revealed shared genetic effects between Crohn’s and Parkinson’s risk, providing a potential biological basis for clinical co-occurrence. Our findings suggest that LRRK2 may be a useful target for developing drugs to treat Crohn’s disease.”
The research was supported by a variety of grants from the National Institutes of Health, the National Science Foundation, and various other foundations. Five of the authors reported consulting for various pharmaceutical companies or companies selling genetic information products. None of the other authors had relevant disclosures.
SOURCE: Hui K et al. Sci Transl Med. 2018. doi: 10.1126/scitranslmed.aai7795
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point:
Major finding: Variant N2081D in LRRK2 was associated with a 73% increased risk of Crohn’s.
Study details: The study comprised data from 30,269 patients and controls.
Disclosures: The research was supported by a variety of grants from the National Institutes of Health, the National Science Foundation, and various other foundations. Five of the authors reported consulting for various pharmaceutical companies or companies selling genetic information products. None of the other authors had relevant disclosures.
Source: Hui K et al. Sci Transl Med. 2018. doi: 10.1126/scitranslmed.aai7795
Debunking Acne Myths: Do Patients Need to Worry About Acne After Adolescence?
Myth: Acne only occurs in teenagers
Acne typically is associated with teenagers and puberty, and many adult patients may not be aware that acne can persist beyond adolescence or even develop for the first time in adulthood. As the prevalence of adults with acne increases, it is important to educate this population about factors associated with postadolescent acne development and let them know that effective treatments are available.
There are 2 types of adult acne: persistent acne, which refers to adolescent acne that continues beyond 25 years of age, and late-onset acne, which develops for the first time after 25 years of age. Adult acne generally is mild to moderate in severity and may be refractory to treatment. Unlike adolescent acne, which is more prominent in adolescent boys and manifests as the more severe forms of the disease, adult acne primarily affects women and is more inflammatory in nature, making these patients more susceptible to scarring. In one study, acne prevalence among 1055 adult participants (age range, 20–60 years) was estimated at 61.5%; however, only 36.8% were aware of their condition and only 25% sought treatment. The most commonly affected area was the malar region, which differs from acne seen in teenagers. In addition to the cheeks, adult acne generally is more prominent on the lower chin, jawline, and neck, and lesions more commonly present as closed comedones.
Fluctuating hormone levels are a common cause of adult acne, particularly in women during menses or pregnancy, menopause, or perimenopause; women also may experience breakouts after starting or discontinuing birth control pills. Acne flare-ups in adults also have been linked to chronic stress, family history, hair and skin care products, medication side effects, undiagnosed medical conditions, steroid use, increased calorie intake, whole and fat-reduced milk consumption, and tobacco smoking. Adult acne also has been found to be associated with other dermatologic conditions including hirsutism, alopecia, and seborrhea.
Early diagnosis and treatment of adult acne is crucial to ensure good cosmetic outcomes and minimize disease burden. When treating adult acne, particularly in women, dermatologists should consider a variety of factors that set this condition apart from adolescent acne, including the predisposition of older skin to irritation, possible slow response to treatment, a high likelihood of good adherence to treatment, and the psychosocial impact of acne in the adult population. In adult women, it also is important to consider whether patients are of childbearing age when selecting a treatment. Patients also should be encouraged to read the labels on their personal care products to ensure they are noncomedogenic and will not clog pores.
Adult acne. American Academy of Dermatology website. https://www.aad.org/public/diseases/acne-and-rosacea/adult-acne. Accessed January 9, 2018.
Dréno B, Layton A, Zouboulis CC, et al. Adult female acne: a new paradigm [published online January 10, 2013]. J Eur Acad Dermatol Venereol. 2013;27:1063-1070.
Khunger N, Kumar C. A clinic-epidemiological study of adult acne: is it different from adolescent acne? Indian J Dermatol Venereol Leprol. 2012;78:335-341.
Semedo D, Ladeiro F, Ruivo M, et al. Adult acne: prevalence and portrayal in primary healthcare patients, in the Great Porto Area, Portugal [published online September 30, 2016]. Acta Med Port. 2016;29:507-513.
Myth: Acne only occurs in teenagers
Acne typically is associated with teenagers and puberty, and many adult patients may not be aware that acne can persist beyond adolescence or even develop for the first time in adulthood. As the prevalence of adults with acne increases, it is important to educate this population about factors associated with postadolescent acne development and let them know that effective treatments are available.
There are 2 types of adult acne: persistent acne, which refers to adolescent acne that continues beyond 25 years of age, and late-onset acne, which develops for the first time after 25 years of age. Adult acne generally is mild to moderate in severity and may be refractory to treatment. Unlike adolescent acne, which is more prominent in adolescent boys and manifests as the more severe forms of the disease, adult acne primarily affects women and is more inflammatory in nature, making these patients more susceptible to scarring. In one study, acne prevalence among 1055 adult participants (age range, 20–60 years) was estimated at 61.5%; however, only 36.8% were aware of their condition and only 25% sought treatment. The most commonly affected area was the malar region, which differs from acne seen in teenagers. In addition to the cheeks, adult acne generally is more prominent on the lower chin, jawline, and neck, and lesions more commonly present as closed comedones.
Fluctuating hormone levels are a common cause of adult acne, particularly in women during menses or pregnancy, menopause, or perimenopause; women also may experience breakouts after starting or discontinuing birth control pills. Acne flare-ups in adults also have been linked to chronic stress, family history, hair and skin care products, medication side effects, undiagnosed medical conditions, steroid use, increased calorie intake, whole and fat-reduced milk consumption, and tobacco smoking. Adult acne also has been found to be associated with other dermatologic conditions including hirsutism, alopecia, and seborrhea.
Early diagnosis and treatment of adult acne is crucial to ensure good cosmetic outcomes and minimize disease burden. When treating adult acne, particularly in women, dermatologists should consider a variety of factors that set this condition apart from adolescent acne, including the predisposition of older skin to irritation, possible slow response to treatment, a high likelihood of good adherence to treatment, and the psychosocial impact of acne in the adult population. In adult women, it also is important to consider whether patients are of childbearing age when selecting a treatment. Patients also should be encouraged to read the labels on their personal care products to ensure they are noncomedogenic and will not clog pores.
Myth: Acne only occurs in teenagers
Acne typically is associated with teenagers and puberty, and many adult patients may not be aware that acne can persist beyond adolescence or even develop for the first time in adulthood. As the prevalence of adults with acne increases, it is important to educate this population about factors associated with postadolescent acne development and let them know that effective treatments are available.
There are 2 types of adult acne: persistent acne, which refers to adolescent acne that continues beyond 25 years of age, and late-onset acne, which develops for the first time after 25 years of age. Adult acne generally is mild to moderate in severity and may be refractory to treatment. Unlike adolescent acne, which is more prominent in adolescent boys and manifests as the more severe forms of the disease, adult acne primarily affects women and is more inflammatory in nature, making these patients more susceptible to scarring. In one study, acne prevalence among 1055 adult participants (age range, 20–60 years) was estimated at 61.5%; however, only 36.8% were aware of their condition and only 25% sought treatment. The most commonly affected area was the malar region, which differs from acne seen in teenagers. In addition to the cheeks, adult acne generally is more prominent on the lower chin, jawline, and neck, and lesions more commonly present as closed comedones.
Fluctuating hormone levels are a common cause of adult acne, particularly in women during menses or pregnancy, menopause, or perimenopause; women also may experience breakouts after starting or discontinuing birth control pills. Acne flare-ups in adults also have been linked to chronic stress, family history, hair and skin care products, medication side effects, undiagnosed medical conditions, steroid use, increased calorie intake, whole and fat-reduced milk consumption, and tobacco smoking. Adult acne also has been found to be associated with other dermatologic conditions including hirsutism, alopecia, and seborrhea.
Early diagnosis and treatment of adult acne is crucial to ensure good cosmetic outcomes and minimize disease burden. When treating adult acne, particularly in women, dermatologists should consider a variety of factors that set this condition apart from adolescent acne, including the predisposition of older skin to irritation, possible slow response to treatment, a high likelihood of good adherence to treatment, and the psychosocial impact of acne in the adult population. In adult women, it also is important to consider whether patients are of childbearing age when selecting a treatment. Patients also should be encouraged to read the labels on their personal care products to ensure they are noncomedogenic and will not clog pores.
Adult acne. American Academy of Dermatology website. https://www.aad.org/public/diseases/acne-and-rosacea/adult-acne. Accessed January 9, 2018.
Dréno B, Layton A, Zouboulis CC, et al. Adult female acne: a new paradigm [published online January 10, 2013]. J Eur Acad Dermatol Venereol. 2013;27:1063-1070.
Khunger N, Kumar C. A clinic-epidemiological study of adult acne: is it different from adolescent acne? Indian J Dermatol Venereol Leprol. 2012;78:335-341.
Semedo D, Ladeiro F, Ruivo M, et al. Adult acne: prevalence and portrayal in primary healthcare patients, in the Great Porto Area, Portugal [published online September 30, 2016]. Acta Med Port. 2016;29:507-513.
Adult acne. American Academy of Dermatology website. https://www.aad.org/public/diseases/acne-and-rosacea/adult-acne. Accessed January 9, 2018.
Dréno B, Layton A, Zouboulis CC, et al. Adult female acne: a new paradigm [published online January 10, 2013]. J Eur Acad Dermatol Venereol. 2013;27:1063-1070.
Khunger N, Kumar C. A clinic-epidemiological study of adult acne: is it different from adolescent acne? Indian J Dermatol Venereol Leprol. 2012;78:335-341.
Semedo D, Ladeiro F, Ruivo M, et al. Adult acne: prevalence and portrayal in primary healthcare patients, in the Great Porto Area, Portugal [published online September 30, 2016]. Acta Med Port. 2016;29:507-513.
CMV colitis mortality rates similar in both immunocompetent and immunocompromised patients
Cytomegalovirus (CMV) colitis has mortality rates similar in immunocompetent patients to that in immunocompromised patients, according to Puo-Hsien Le, MD, and associates.
In a retrospective study, the investigators analyzed data from 42 immunocompetent patients and 27 patients who were immunocompromised because of HIV infection, solid organ or bone marrow transplantation, immunosuppressive drug use, chemotherapeutic agent use within 6 months, or other reasons. In-hospital mortality was 26.2% in immunocompetent patients and 25.9% in immunocompromised patients.
While diarrhea and melena were the first presenting symptom in a similar number of patients, immunocompetent patients were more likely to present with melena while immunocompromised patients were more likely to present with diarrhea. The number of days until diagnosis was the only independent predictor of in-hospital mortality according to the analysis, with patients who were diagnosed within 9 days of admittance having a significantly higher survival rate.
“Contrary to data published up to this point, we found that CMV colitis was not rare and that it could be fatal in immunocompetent hosts, especially those patients with advanced age, specific comorbidities associated with immune dysfunction, critical illness, or IBD. ... Being alert to the different presentations can greatly help make an accurate early diagnosis and materially improve survival for CMV colitis patients,” the investigators concluded.
Find the full study in Therapeutics and Clinical Risk Management (doi: 10.2147/TCRM.S151180).
Cytomegalovirus (CMV) colitis has mortality rates similar in immunocompetent patients to that in immunocompromised patients, according to Puo-Hsien Le, MD, and associates.
In a retrospective study, the investigators analyzed data from 42 immunocompetent patients and 27 patients who were immunocompromised because of HIV infection, solid organ or bone marrow transplantation, immunosuppressive drug use, chemotherapeutic agent use within 6 months, or other reasons. In-hospital mortality was 26.2% in immunocompetent patients and 25.9% in immunocompromised patients.
While diarrhea and melena were the first presenting symptom in a similar number of patients, immunocompetent patients were more likely to present with melena while immunocompromised patients were more likely to present with diarrhea. The number of days until diagnosis was the only independent predictor of in-hospital mortality according to the analysis, with patients who were diagnosed within 9 days of admittance having a significantly higher survival rate.
“Contrary to data published up to this point, we found that CMV colitis was not rare and that it could be fatal in immunocompetent hosts, especially those patients with advanced age, specific comorbidities associated with immune dysfunction, critical illness, or IBD. ... Being alert to the different presentations can greatly help make an accurate early diagnosis and materially improve survival for CMV colitis patients,” the investigators concluded.
Find the full study in Therapeutics and Clinical Risk Management (doi: 10.2147/TCRM.S151180).
Cytomegalovirus (CMV) colitis has mortality rates similar in immunocompetent patients to that in immunocompromised patients, according to Puo-Hsien Le, MD, and associates.
In a retrospective study, the investigators analyzed data from 42 immunocompetent patients and 27 patients who were immunocompromised because of HIV infection, solid organ or bone marrow transplantation, immunosuppressive drug use, chemotherapeutic agent use within 6 months, or other reasons. In-hospital mortality was 26.2% in immunocompetent patients and 25.9% in immunocompromised patients.
While diarrhea and melena were the first presenting symptom in a similar number of patients, immunocompetent patients were more likely to present with melena while immunocompromised patients were more likely to present with diarrhea. The number of days until diagnosis was the only independent predictor of in-hospital mortality according to the analysis, with patients who were diagnosed within 9 days of admittance having a significantly higher survival rate.
“Contrary to data published up to this point, we found that CMV colitis was not rare and that it could be fatal in immunocompetent hosts, especially those patients with advanced age, specific comorbidities associated with immune dysfunction, critical illness, or IBD. ... Being alert to the different presentations can greatly help make an accurate early diagnosis and materially improve survival for CMV colitis patients,” the investigators concluded.
Find the full study in Therapeutics and Clinical Risk Management (doi: 10.2147/TCRM.S151180).
FROM THERAPEUTICS AND CLINICAL RISK MANAGEMENT
Optimal rate of flow for high-flow nasal cannula in young children
Clinical question
Is there an optimal rate of flow for high-flow nasal cannula in respiratory distress?
Background
High-flow nasal cannula (HFNC) has been increasingly used to treat children with moderate to severe bronchiolitis, both in intensive care unit (ICU) settings and on inpatient wards. Studies have shown it may allow children with bronchiolitis to avoid ICU admission and intubation. In preterm infants it has been shown to decrease work of breathing. No prior studies, however, have examined optimizing the rate of flow for individual patients, and considerable heterogeneity exists in choosing initial HFNC flow rates.
Reliably measuring effort of breathing has proved challenging. Placing a manometer in the esophagus allows measurement of the pressure-rate product (PRP), a previously validated measure of effort of breathing computed by multiplying the difference between maximum and minimum esophageal pressures by the respiratory rate.1 An increasing PRP indicates increasing effort of breathing. The authors chose systems from Fisher & Paykel and Vapotherm for their testing.
Study design
Single-center prospective observational trial.
Setting
24-bed pediatric intensive care unit in a 347-bed urban free-standing children’s hospital.
Synopsis
A single center recruited patients aged 37 weeks corrected gestational age to 3 years who were admitted to the ICU with respiratory distress. Fifty-four patients met inclusion criteria and 21 were enrolled and completed the study. Prior data suggested a sample size of 20 would be sufficient to identify a clinically significant effect size. Median age was 6 months.
Thirteen patients had bronchiolitis, three had pneumonia, and five had other respiratory illnesses. Each patient received HFNC delivered by both systems in sequence with flow rates of 0.5, 1, 1.5, and 2 L/kg per minute to a maximum of 30 L/min. Following the trials, patients remained on HFNC as per usual care with twice-daily PRP measurements until weaned off HFNC.
A dose-dependent relationship existed between flow and change in PRP, with the greatest reduction in PRP at 2 L/kg per minute flow (P less than .001) and a slightly smaller but similar reduction in PRP at 1.5 L/kg per minute. When stratifying the subjects by weight, this effect was not statistically significant for patients heavier than 8 kg (P = .38), with all significant changes being in patients less than 8 kg (P less than .001) with a median drop in PRP of 25%. Further examining these younger and lighter patients, the greatest reduction in PRP was in the lightest patients (less than 5 kg).
Given the similarity in drop in PRP at 1.5 L/kg per minute and 2 L/kg per minute, the authors suggest this flow rate yields a plateau effect and minimal further improvement would be seen with increasing flow rates. A rate of 2 L/kg per minute was chosen as a maximum a priori as it was judged the highest level of HFNC patients could tolerate without worsening agitation or air leak. There was no difference seen between the two HFNC systems in the study. The authors did not report the fraction of inspired oxygen settings used, the size of HFNC cannulas, or how PRP changed over several days as HFNC was weaned.
Bottom line
The optimal HFNC rate to decrease effort of breathing for children less than 3 years old is between 1.5 and 2 L/kg/min with the greatest improvement expected in children under 5 kg.
Citation
Weiler T et al. The Relationship between High Flow Nasal Cannula Flow Rate and Effort of Breathing in Children. The Journal of Pediatrics. October 2017. doi: 10.1016/j.jpeds.2017.06.006.
Reference
1. Argent AC, Newth CJL, Klein M. The mechanics of breathing in children with acute severe croup. Intensive Care Med. 2008;34(2):324-32. doi: 10.1007/s00134-007-0910-x.
Dr. Stubblefield is a pediatric hospitalist at Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del., and clinical assistant professor of pediatrics at Jefferson Medical College in Philadelphia.
Clinical question
Is there an optimal rate of flow for high-flow nasal cannula in respiratory distress?
Background
High-flow nasal cannula (HFNC) has been increasingly used to treat children with moderate to severe bronchiolitis, both in intensive care unit (ICU) settings and on inpatient wards. Studies have shown it may allow children with bronchiolitis to avoid ICU admission and intubation. In preterm infants it has been shown to decrease work of breathing. No prior studies, however, have examined optimizing the rate of flow for individual patients, and considerable heterogeneity exists in choosing initial HFNC flow rates.
Reliably measuring effort of breathing has proved challenging. Placing a manometer in the esophagus allows measurement of the pressure-rate product (PRP), a previously validated measure of effort of breathing computed by multiplying the difference between maximum and minimum esophageal pressures by the respiratory rate.1 An increasing PRP indicates increasing effort of breathing. The authors chose systems from Fisher & Paykel and Vapotherm for their testing.
Study design
Single-center prospective observational trial.
Setting
24-bed pediatric intensive care unit in a 347-bed urban free-standing children’s hospital.
Synopsis
A single center recruited patients aged 37 weeks corrected gestational age to 3 years who were admitted to the ICU with respiratory distress. Fifty-four patients met inclusion criteria and 21 were enrolled and completed the study. Prior data suggested a sample size of 20 would be sufficient to identify a clinically significant effect size. Median age was 6 months.
Thirteen patients had bronchiolitis, three had pneumonia, and five had other respiratory illnesses. Each patient received HFNC delivered by both systems in sequence with flow rates of 0.5, 1, 1.5, and 2 L/kg per minute to a maximum of 30 L/min. Following the trials, patients remained on HFNC as per usual care with twice-daily PRP measurements until weaned off HFNC.
A dose-dependent relationship existed between flow and change in PRP, with the greatest reduction in PRP at 2 L/kg per minute flow (P less than .001) and a slightly smaller but similar reduction in PRP at 1.5 L/kg per minute. When stratifying the subjects by weight, this effect was not statistically significant for patients heavier than 8 kg (P = .38), with all significant changes being in patients less than 8 kg (P less than .001) with a median drop in PRP of 25%. Further examining these younger and lighter patients, the greatest reduction in PRP was in the lightest patients (less than 5 kg).
Given the similarity in drop in PRP at 1.5 L/kg per minute and 2 L/kg per minute, the authors suggest this flow rate yields a plateau effect and minimal further improvement would be seen with increasing flow rates. A rate of 2 L/kg per minute was chosen as a maximum a priori as it was judged the highest level of HFNC patients could tolerate without worsening agitation or air leak. There was no difference seen between the two HFNC systems in the study. The authors did not report the fraction of inspired oxygen settings used, the size of HFNC cannulas, or how PRP changed over several days as HFNC was weaned.
Bottom line
The optimal HFNC rate to decrease effort of breathing for children less than 3 years old is between 1.5 and 2 L/kg/min with the greatest improvement expected in children under 5 kg.
Citation
Weiler T et al. The Relationship between High Flow Nasal Cannula Flow Rate and Effort of Breathing in Children. The Journal of Pediatrics. October 2017. doi: 10.1016/j.jpeds.2017.06.006.
Reference
1. Argent AC, Newth CJL, Klein M. The mechanics of breathing in children with acute severe croup. Intensive Care Med. 2008;34(2):324-32. doi: 10.1007/s00134-007-0910-x.
Dr. Stubblefield is a pediatric hospitalist at Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del., and clinical assistant professor of pediatrics at Jefferson Medical College in Philadelphia.
Clinical question
Is there an optimal rate of flow for high-flow nasal cannula in respiratory distress?
Background
High-flow nasal cannula (HFNC) has been increasingly used to treat children with moderate to severe bronchiolitis, both in intensive care unit (ICU) settings and on inpatient wards. Studies have shown it may allow children with bronchiolitis to avoid ICU admission and intubation. In preterm infants it has been shown to decrease work of breathing. No prior studies, however, have examined optimizing the rate of flow for individual patients, and considerable heterogeneity exists in choosing initial HFNC flow rates.
Reliably measuring effort of breathing has proved challenging. Placing a manometer in the esophagus allows measurement of the pressure-rate product (PRP), a previously validated measure of effort of breathing computed by multiplying the difference between maximum and minimum esophageal pressures by the respiratory rate.1 An increasing PRP indicates increasing effort of breathing. The authors chose systems from Fisher & Paykel and Vapotherm for their testing.
Study design
Single-center prospective observational trial.
Setting
24-bed pediatric intensive care unit in a 347-bed urban free-standing children’s hospital.
Synopsis
A single center recruited patients aged 37 weeks corrected gestational age to 3 years who were admitted to the ICU with respiratory distress. Fifty-four patients met inclusion criteria and 21 were enrolled and completed the study. Prior data suggested a sample size of 20 would be sufficient to identify a clinically significant effect size. Median age was 6 months.
Thirteen patients had bronchiolitis, three had pneumonia, and five had other respiratory illnesses. Each patient received HFNC delivered by both systems in sequence with flow rates of 0.5, 1, 1.5, and 2 L/kg per minute to a maximum of 30 L/min. Following the trials, patients remained on HFNC as per usual care with twice-daily PRP measurements until weaned off HFNC.
A dose-dependent relationship existed between flow and change in PRP, with the greatest reduction in PRP at 2 L/kg per minute flow (P less than .001) and a slightly smaller but similar reduction in PRP at 1.5 L/kg per minute. When stratifying the subjects by weight, this effect was not statistically significant for patients heavier than 8 kg (P = .38), with all significant changes being in patients less than 8 kg (P less than .001) with a median drop in PRP of 25%. Further examining these younger and lighter patients, the greatest reduction in PRP was in the lightest patients (less than 5 kg).
Given the similarity in drop in PRP at 1.5 L/kg per minute and 2 L/kg per minute, the authors suggest this flow rate yields a plateau effect and minimal further improvement would be seen with increasing flow rates. A rate of 2 L/kg per minute was chosen as a maximum a priori as it was judged the highest level of HFNC patients could tolerate without worsening agitation or air leak. There was no difference seen between the two HFNC systems in the study. The authors did not report the fraction of inspired oxygen settings used, the size of HFNC cannulas, or how PRP changed over several days as HFNC was weaned.
Bottom line
The optimal HFNC rate to decrease effort of breathing for children less than 3 years old is between 1.5 and 2 L/kg/min with the greatest improvement expected in children under 5 kg.
Citation
Weiler T et al. The Relationship between High Flow Nasal Cannula Flow Rate and Effort of Breathing in Children. The Journal of Pediatrics. October 2017. doi: 10.1016/j.jpeds.2017.06.006.
Reference
1. Argent AC, Newth CJL, Klein M. The mechanics of breathing in children with acute severe croup. Intensive Care Med. 2008;34(2):324-32. doi: 10.1007/s00134-007-0910-x.
Dr. Stubblefield is a pediatric hospitalist at Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del., and clinical assistant professor of pediatrics at Jefferson Medical College in Philadelphia.
Best of 2017: Top News Articles
VA Central California Improves Access to Clinical Trials
The Hematology and Oncology Department at the VA Central California Health Care System (VACCHCS) in Fresno, California, is developing multiple programs to increase access to clinical trials for veterans. The department secured funding for an investigator-initiated supportive care clinical trial, which looks at the feasibility of using olanzapine to replace dexamethasone in elderly patients with diabetes who are undergoing moderate-intensity chemotherapy for prevention of nausea and vomiting. The department received a grant from the Southwest Oncology Group (SWOG) HOPE Foundation to help its clinical trials research team become members of SWOG. Currently, VACCHCS Fresno has 4 active industry-sponsored clinical trials to offer to veterans with myelodysplastic syndrome/acute myeloid leukemia, lung, prostate, and bladder cancers. They are in the process of getting another 4 clinical trials in the next 6 months.
“Our primary responsibility is to provide timely and excellent care to our veterans,” said Sachdev P. Thomas, MD, Chief of Hematology/Oncology at Fresno VACCHCS. “Wait time for new patients in our Hematology/Oncology clinic is less than 7 business days. We also have a walk-in clinic. Before we initiated our Clinical Trial program, we ensured that this metric for timely care was in place.”
Through the Association of VA Hematology/Oncology (AVAHO), Fresno VACCHCS oncologists and a team at the Richmond VAMC in, Virginia are currently working with the National Institutes of Health, to create a VA-specific cancer clinical trials’ filter at the website Clinicaltrials.gov. This VA-specific filter will enable, any VA physician, nationwide to easily search for available clinical trials for any cancer type, throughout the VHA.
The Hematology and Oncology Department at the VA Central California Health Care System (VACCHCS) in Fresno, California, is developing multiple programs to increase access to clinical trials for veterans. The department secured funding for an investigator-initiated supportive care clinical trial, which looks at the feasibility of using olanzapine to replace dexamethasone in elderly patients with diabetes who are undergoing moderate-intensity chemotherapy for prevention of nausea and vomiting. The department received a grant from the Southwest Oncology Group (SWOG) HOPE Foundation to help its clinical trials research team become members of SWOG. Currently, VACCHCS Fresno has 4 active industry-sponsored clinical trials to offer to veterans with myelodysplastic syndrome/acute myeloid leukemia, lung, prostate, and bladder cancers. They are in the process of getting another 4 clinical trials in the next 6 months.
“Our primary responsibility is to provide timely and excellent care to our veterans,” said Sachdev P. Thomas, MD, Chief of Hematology/Oncology at Fresno VACCHCS. “Wait time for new patients in our Hematology/Oncology clinic is less than 7 business days. We also have a walk-in clinic. Before we initiated our Clinical Trial program, we ensured that this metric for timely care was in place.”
Through the Association of VA Hematology/Oncology (AVAHO), Fresno VACCHCS oncologists and a team at the Richmond VAMC in, Virginia are currently working with the National Institutes of Health, to create a VA-specific cancer clinical trials’ filter at the website Clinicaltrials.gov. This VA-specific filter will enable, any VA physician, nationwide to easily search for available clinical trials for any cancer type, throughout the VHA.
The Hematology and Oncology Department at the VA Central California Health Care System (VACCHCS) in Fresno, California, is developing multiple programs to increase access to clinical trials for veterans. The department secured funding for an investigator-initiated supportive care clinical trial, which looks at the feasibility of using olanzapine to replace dexamethasone in elderly patients with diabetes who are undergoing moderate-intensity chemotherapy for prevention of nausea and vomiting. The department received a grant from the Southwest Oncology Group (SWOG) HOPE Foundation to help its clinical trials research team become members of SWOG. Currently, VACCHCS Fresno has 4 active industry-sponsored clinical trials to offer to veterans with myelodysplastic syndrome/acute myeloid leukemia, lung, prostate, and bladder cancers. They are in the process of getting another 4 clinical trials in the next 6 months.
“Our primary responsibility is to provide timely and excellent care to our veterans,” said Sachdev P. Thomas, MD, Chief of Hematology/Oncology at Fresno VACCHCS. “Wait time for new patients in our Hematology/Oncology clinic is less than 7 business days. We also have a walk-in clinic. Before we initiated our Clinical Trial program, we ensured that this metric for timely care was in place.”
Through the Association of VA Hematology/Oncology (AVAHO), Fresno VACCHCS oncologists and a team at the Richmond VAMC in, Virginia are currently working with the National Institutes of Health, to create a VA-specific cancer clinical trials’ filter at the website Clinicaltrials.gov. This VA-specific filter will enable, any VA physician, nationwide to easily search for available clinical trials for any cancer type, throughout the VHA.
Is the VA Ready for New Veterans?
Beginning in less than 90 days, new veterans will become eligible for VA mental health care as soon as they leave military service. More than 250,000 service members transition out of the military each year. On Tuesday, the President signed an executive order that gives the DoD, Department of Homeland Security, and VA just 60 days to develop a plan for handling the influx of new patients into the system.
“As service members transition to veteran status, they face higher risk of suicide and mental health difficulties,” said VA Secretary David Shulkin, MD. “During this critical phase, many transitioning service members may not qualify for enrollment in health care. The focus of this executive order is to coordinate federal assets to close that gap.”
How the VA will meet the increased demand remains to be seen. As an executive order, the mandate does not come with additional funds. And in October testimony, Secretary Shulkin admitted that the VA already was having difficulty hiring and training enough mental health care providers to meet existing demand.
The executive order is focused on the 60% of new veterans who are not immediately eligible for VA health care benefits. According to Secretary Shulkin, in the first year postseparation, often before VA eligibility is determined, these new veterans are especially at risk for suicide. “That 12-month period after you leave service is the highest risk for suicide,” Secretary Shulkin noted. Veterans during that period face almost one and a half to 2 times the risk of suicide in that first 12 months when they leave the service.
The VA outlined 3 approaches that would be implemented to meet the mental health needs for transitioning veterans. Peer counseling will play a particularly important role in the new services. First, the VA will expand its peer community outreach and group session offers and the VA whole health patient-centered care initiative. Currently, the VA Whole Health program is in 18 facilities, but it will be expanded to all facilities. The program focuses on wellness and establishing individual health goals.
In addition, the DoD “Be There Peer Support Call and Outreach Center” services will be expanded. The program will begin to offer peer support for veterans in the year following separation from the uniformed service.
Most consequently, the executive order will expand the DoD Military OneSource, which currently provides free mental health care counseling to active-duty service members and their families. The program will be expanded to include recently transitioned veterans as well. And according to a report in Military Times, Military OneSource care also may be available to the families of new veterans.
Beginning in less than 90 days, new veterans will become eligible for VA mental health care as soon as they leave military service. More than 250,000 service members transition out of the military each year. On Tuesday, the President signed an executive order that gives the DoD, Department of Homeland Security, and VA just 60 days to develop a plan for handling the influx of new patients into the system.
“As service members transition to veteran status, they face higher risk of suicide and mental health difficulties,” said VA Secretary David Shulkin, MD. “During this critical phase, many transitioning service members may not qualify for enrollment in health care. The focus of this executive order is to coordinate federal assets to close that gap.”
How the VA will meet the increased demand remains to be seen. As an executive order, the mandate does not come with additional funds. And in October testimony, Secretary Shulkin admitted that the VA already was having difficulty hiring and training enough mental health care providers to meet existing demand.
The executive order is focused on the 60% of new veterans who are not immediately eligible for VA health care benefits. According to Secretary Shulkin, in the first year postseparation, often before VA eligibility is determined, these new veterans are especially at risk for suicide. “That 12-month period after you leave service is the highest risk for suicide,” Secretary Shulkin noted. Veterans during that period face almost one and a half to 2 times the risk of suicide in that first 12 months when they leave the service.
The VA outlined 3 approaches that would be implemented to meet the mental health needs for transitioning veterans. Peer counseling will play a particularly important role in the new services. First, the VA will expand its peer community outreach and group session offers and the VA whole health patient-centered care initiative. Currently, the VA Whole Health program is in 18 facilities, but it will be expanded to all facilities. The program focuses on wellness and establishing individual health goals.
In addition, the DoD “Be There Peer Support Call and Outreach Center” services will be expanded. The program will begin to offer peer support for veterans in the year following separation from the uniformed service.
Most consequently, the executive order will expand the DoD Military OneSource, which currently provides free mental health care counseling to active-duty service members and their families. The program will be expanded to include recently transitioned veterans as well. And according to a report in Military Times, Military OneSource care also may be available to the families of new veterans.
Beginning in less than 90 days, new veterans will become eligible for VA mental health care as soon as they leave military service. More than 250,000 service members transition out of the military each year. On Tuesday, the President signed an executive order that gives the DoD, Department of Homeland Security, and VA just 60 days to develop a plan for handling the influx of new patients into the system.
“As service members transition to veteran status, they face higher risk of suicide and mental health difficulties,” said VA Secretary David Shulkin, MD. “During this critical phase, many transitioning service members may not qualify for enrollment in health care. The focus of this executive order is to coordinate federal assets to close that gap.”
How the VA will meet the increased demand remains to be seen. As an executive order, the mandate does not come with additional funds. And in October testimony, Secretary Shulkin admitted that the VA already was having difficulty hiring and training enough mental health care providers to meet existing demand.
The executive order is focused on the 60% of new veterans who are not immediately eligible for VA health care benefits. According to Secretary Shulkin, in the first year postseparation, often before VA eligibility is determined, these new veterans are especially at risk for suicide. “That 12-month period after you leave service is the highest risk for suicide,” Secretary Shulkin noted. Veterans during that period face almost one and a half to 2 times the risk of suicide in that first 12 months when they leave the service.
The VA outlined 3 approaches that would be implemented to meet the mental health needs for transitioning veterans. Peer counseling will play a particularly important role in the new services. First, the VA will expand its peer community outreach and group session offers and the VA whole health patient-centered care initiative. Currently, the VA Whole Health program is in 18 facilities, but it will be expanded to all facilities. The program focuses on wellness and establishing individual health goals.
In addition, the DoD “Be There Peer Support Call and Outreach Center” services will be expanded. The program will begin to offer peer support for veterans in the year following separation from the uniformed service.
Most consequently, the executive order will expand the DoD Military OneSource, which currently provides free mental health care counseling to active-duty service members and their families. The program will be expanded to include recently transitioned veterans as well. And according to a report in Military Times, Military OneSource care also may be available to the families of new veterans.
Alopecia tied to nearly fivefold increase in fibroids in African American women
based on data from more than 400,000 women.
In a study published in JAMA Dermatology, researchers reviewed data from 487,104 black women seen at a single center between Aug. 1, 2013, and Aug. 1, 2017. Overall, 14% of women with central centrifugal cicatricial alopecia (CCCA) also had a history of uterine fibroids, compared with 3% percent of black women without CCCA.
“Alopecia is more than just a cosmetic problem. … It could signal an increased risk of developing other conditions,” corresponding author Crystal Aguh, MD, of Johns Hopkins University in Baltimore said in an interview. “To our knowledge, this is the first time that an association has been noted between these two conditions. We believe that the fact that both are related to excess scarring and fibrous tissue deposition may reflect similarities in how both [conditions] develop, but this is still unknown.”
Overall, 62 of 447 women who met criteria for CCCA also had fibroids, representing a nearly fivefold increase in fibroid risk for women with CCCA.
“I was definitely surprised by the findings,” said Dr. Aguh. “I thought it would be interesting to look at any possible correlation between the two diseases, but did not expect to see such a large difference between black women with and without this form of hair loss,” she noted.
As fibroids are often asymptomatic, “physicians should screen their patients with CCCA for symptoms of fibroids such as painful menstrual cycles, heavy bleeding, unexplained anemia, or difficulty conceiving,” said Dr. Aguh. “In those patients who may not know they have fibroids, early recognition that allows for treatment will be especially beneficial.”
The findings were limited by the retrospective nature of the study. “I believe that larger studies are warranted to help us fully understand how these two conditions are connected,” Dr. Aguh said.
Lead author Yemisi Dina of Meharry Medical College, Nashville, Tenn., is supported in part by a grant from the National Institutes of Health. The other researchers had no financial conflicts to disclose.
SOURCE: Dina Y et al. JAMA Dermatol. 2017 Dec 27. doi: 10.1001/jamadermatol.2017.5163
based on data from more than 400,000 women.
In a study published in JAMA Dermatology, researchers reviewed data from 487,104 black women seen at a single center between Aug. 1, 2013, and Aug. 1, 2017. Overall, 14% of women with central centrifugal cicatricial alopecia (CCCA) also had a history of uterine fibroids, compared with 3% percent of black women without CCCA.
“Alopecia is more than just a cosmetic problem. … It could signal an increased risk of developing other conditions,” corresponding author Crystal Aguh, MD, of Johns Hopkins University in Baltimore said in an interview. “To our knowledge, this is the first time that an association has been noted between these two conditions. We believe that the fact that both are related to excess scarring and fibrous tissue deposition may reflect similarities in how both [conditions] develop, but this is still unknown.”
Overall, 62 of 447 women who met criteria for CCCA also had fibroids, representing a nearly fivefold increase in fibroid risk for women with CCCA.
“I was definitely surprised by the findings,” said Dr. Aguh. “I thought it would be interesting to look at any possible correlation between the two diseases, but did not expect to see such a large difference between black women with and without this form of hair loss,” she noted.
As fibroids are often asymptomatic, “physicians should screen their patients with CCCA for symptoms of fibroids such as painful menstrual cycles, heavy bleeding, unexplained anemia, or difficulty conceiving,” said Dr. Aguh. “In those patients who may not know they have fibroids, early recognition that allows for treatment will be especially beneficial.”
The findings were limited by the retrospective nature of the study. “I believe that larger studies are warranted to help us fully understand how these two conditions are connected,” Dr. Aguh said.
Lead author Yemisi Dina of Meharry Medical College, Nashville, Tenn., is supported in part by a grant from the National Institutes of Health. The other researchers had no financial conflicts to disclose.
SOURCE: Dina Y et al. JAMA Dermatol. 2017 Dec 27. doi: 10.1001/jamadermatol.2017.5163
based on data from more than 400,000 women.
In a study published in JAMA Dermatology, researchers reviewed data from 487,104 black women seen at a single center between Aug. 1, 2013, and Aug. 1, 2017. Overall, 14% of women with central centrifugal cicatricial alopecia (CCCA) also had a history of uterine fibroids, compared with 3% percent of black women without CCCA.
“Alopecia is more than just a cosmetic problem. … It could signal an increased risk of developing other conditions,” corresponding author Crystal Aguh, MD, of Johns Hopkins University in Baltimore said in an interview. “To our knowledge, this is the first time that an association has been noted between these two conditions. We believe that the fact that both are related to excess scarring and fibrous tissue deposition may reflect similarities in how both [conditions] develop, but this is still unknown.”
Overall, 62 of 447 women who met criteria for CCCA also had fibroids, representing a nearly fivefold increase in fibroid risk for women with CCCA.
“I was definitely surprised by the findings,” said Dr. Aguh. “I thought it would be interesting to look at any possible correlation between the two diseases, but did not expect to see such a large difference between black women with and without this form of hair loss,” she noted.
As fibroids are often asymptomatic, “physicians should screen their patients with CCCA for symptoms of fibroids such as painful menstrual cycles, heavy bleeding, unexplained anemia, or difficulty conceiving,” said Dr. Aguh. “In those patients who may not know they have fibroids, early recognition that allows for treatment will be especially beneficial.”
The findings were limited by the retrospective nature of the study. “I believe that larger studies are warranted to help us fully understand how these two conditions are connected,” Dr. Aguh said.
Lead author Yemisi Dina of Meharry Medical College, Nashville, Tenn., is supported in part by a grant from the National Institutes of Health. The other researchers had no financial conflicts to disclose.
SOURCE: Dina Y et al. JAMA Dermatol. 2017 Dec 27. doi: 10.1001/jamadermatol.2017.5163
FROM JAMA DERMATOLOGY
Key clinical point: Dermatologists should screen patients with central centrifugal cicatricial alopecia for potential fibroids.
Major finding: Women with CCCA were nearly five times more likely to have fibroids, compared with controls.
Data source: The data come from a review of 487,104 black women seen at a single center between Aug. 1, 2013, and Aug. 1, 2017.
Disclosures: Lead author Yemisi Dina of Meharry Medical College, Nashville, Tenn., is supported in part by a grant from the National Institutes of Health. The other researchers had no financial conflicts to disclose.
Source: Dina Y et al. JAMA Dermatol. 2017 Dec 27. doi: 10.1001/jamadermatol.2017.5163.
Risk of Diabetes Climbs Among Veterans
More veterans are developing diabetes, say researchers from East Tennessee University in Johnson City, Tennessee. The rise is linked to a similar climb in obesity rates.
The researchers analyzed data from 5 cycles of the National Health and Nutrition Examination Survey (NHANES). The survey sample sizes ranged from 472 to 685.
Diabetes prevalence rose from 15.5% in 2005-2006 to 20.5% in 2013-2014, and rose significantly among men, from 16.5% in 2005-2006 to 22% in 2013-2014. Diabetes was most prevalent among veterans who were aged > 65 years, had more than 12 years of education, and had an income below the 100% federal poverty level. Those same subgroups had the highest prevalence of obesity except for the age subgroup. Obesity was more prevalent among veterans aged 45 to 64 years. Hispanic veterans had the highest prevalence of both obesity and diabetes.
The researchers note that some factors limited the accuracy of the estimated prevalence of diabetes among U.S. veterans when using VA databases. One is that in fiscal year 2014, < 30% of the total veteran population sought VA health care, and > 70% sought care outside the VA system even though some were enrolled.
More veterans are developing diabetes, say researchers from East Tennessee University in Johnson City, Tennessee. The rise is linked to a similar climb in obesity rates.
The researchers analyzed data from 5 cycles of the National Health and Nutrition Examination Survey (NHANES). The survey sample sizes ranged from 472 to 685.
Diabetes prevalence rose from 15.5% in 2005-2006 to 20.5% in 2013-2014, and rose significantly among men, from 16.5% in 2005-2006 to 22% in 2013-2014. Diabetes was most prevalent among veterans who were aged > 65 years, had more than 12 years of education, and had an income below the 100% federal poverty level. Those same subgroups had the highest prevalence of obesity except for the age subgroup. Obesity was more prevalent among veterans aged 45 to 64 years. Hispanic veterans had the highest prevalence of both obesity and diabetes.
The researchers note that some factors limited the accuracy of the estimated prevalence of diabetes among U.S. veterans when using VA databases. One is that in fiscal year 2014, < 30% of the total veteran population sought VA health care, and > 70% sought care outside the VA system even though some were enrolled.
More veterans are developing diabetes, say researchers from East Tennessee University in Johnson City, Tennessee. The rise is linked to a similar climb in obesity rates.
The researchers analyzed data from 5 cycles of the National Health and Nutrition Examination Survey (NHANES). The survey sample sizes ranged from 472 to 685.
Diabetes prevalence rose from 15.5% in 2005-2006 to 20.5% in 2013-2014, and rose significantly among men, from 16.5% in 2005-2006 to 22% in 2013-2014. Diabetes was most prevalent among veterans who were aged > 65 years, had more than 12 years of education, and had an income below the 100% federal poverty level. Those same subgroups had the highest prevalence of obesity except for the age subgroup. Obesity was more prevalent among veterans aged 45 to 64 years. Hispanic veterans had the highest prevalence of both obesity and diabetes.
The researchers note that some factors limited the accuracy of the estimated prevalence of diabetes among U.S. veterans when using VA databases. One is that in fiscal year 2014, < 30% of the total veteran population sought VA health care, and > 70% sought care outside the VA system even though some were enrolled.
Clinical presentation, diagnosis, and management of typical and atypical bronchopulmonary carcinoid
Carcinoid lung tumors represent the most indolent form of a spectrum of bronchopulmonary neuroendocrine tumors (NETs) that includes small-cell carcinoma as its most malignant member, as well as several other forms of intermediately aggressive tumors, such as atypical carcinoid.1 Carcinoids represent 1.2% of all primary lung malignancies. Their incidence in the United States has increased rapidly over the last 30 years and is currently about 6% a year. Lung carcinoids are more prevalent in whites compared with blacks, and in Asians compared with non-Asians. They are less common in Hispanics compared with non-Hispanics.1 Typical carcinoids represent 80%-90% of all lung carcinoids and occur more frequently in the fifth and sixth decades of life. They can, however, occur at any age, and are the most common lung tumor in childhood.1
Etiology and risk factors
Unlike carcinoma of the lung, no external environmental toxin or other stimulus has been identified as a causative agent for the development of pulmonary carcinoid tumors. It is not clear if there is an association between bronchial NETs and smoking.1 Nearly all bronchial NETs are sporadic; however, they can rarely occur in the setting of multiple endocrine neoplasia type 1.1
Presentation
About 60% of the patients with bronchial carcinoids are symptomatic at presentation. The most common clinical findings are those associated with bronchial obstruction, such as persistent cough, hemoptysis, and recurrent or obstructive pneumonitis. Wheezing, chest pain, and dyspnea also may be noted.2 Various endocrine or neuroendocrine syndromes can be initial clinical manifestations of either typical or atypical pulmonary carcinoid tumors, but that is not common Cushing syndrome (ectopic production and secretion of adrenocorticotropic hormone [ACTH]) may occur in about 2% of lung carcinoid.3 In cases of malignancy, the presence of metastatic disease can produce weight loss, weakness, and a general feeling of ill health.
Diagnostic work-up
Biochemical test
There is no biochemical study that can be used as a screening test to determine the presence of a carcinoid tumor or to diagnose a known pulmonary mass as a carcinoid tumor. Neuroendocrine cells produce biologically active amines and peptides that can be detected in serum and urine. Although the syndromes associated with lung carcinoids are seen in about 1%-2% of the patients, assays of specific hormones or other circulating neuroendocrine substances, such as ACTH, melanocyte-stimulating hormone, or growth hormone may establish the existence of a clinically suspected syndrome.
Chest radiography
An abnormal finding on chest radiography is present in about 75% of patients with a pulmonary carcinoid tumor.1 Findings include either the presence of the tumor mass itself or indirect evidence of its presence observed as parenchymal changes associated with bronchial obstruction from the mass.
Computed-tomography imaging
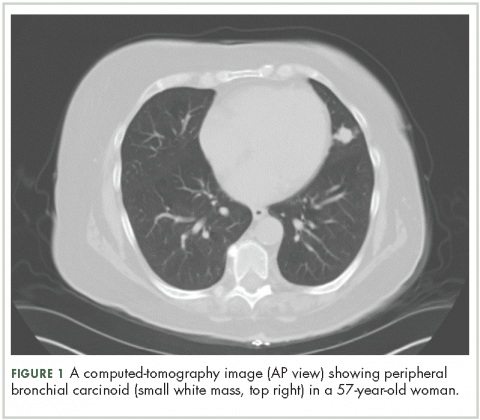
High-resolution computed-tomography (CT) imaging is the one of the best types of CT examination for evaluation of a pulmonary carcinoid tumor.4 A CT scan provides excellent resolution of tumor extent, location, and the presence or absence of mediastinal adenopathy. It also aids in morphologic characterization of peripheral (Figure 1) and especially centrally located carcinoids, which may be purely intraluminal (polypoid configuration), exclusively extra luminal, or more frequently, a mixture of intraluminal and extraluminal components.
CT scans may also be helpful for differentiating tumor from postobstructive atelectasis or bronchial obstruction-related mucoid impaction. Intravenous contrast in CT imaging can be useful in differentiating malignant from benign lesions. Because carcinoid tumors are highly vascular, they show greater enhancement on contrast CT than do benign lesions. The sensitivity of CT for detecting metastatic hilar or mediastinal nodes is high, but specificity is as low as 45%.4
Typical carcinoid is rarely metastatic so most patients do not need CT or MRI imaging to evaluate for liver involvement. Liver imaging is appropriate in patients with evidence of mediastinal involvement, relatively high mitotic rate, or clinical evidence of the carcinoid syndrome.8 To evaluate for metastatic spread to the liver, multiphase contrast-enhanced liver CT scans should be performed with arterial and portal-venous phases because carcinoid liver metastases are often hypervascular and appear isodense relative to the liver parenchyma after contrast administration.4 An MRI is often preferred the modality to evaluate for metastatic spread to the liver because of its higher sensitivity.5
Positron-emission tomography
Although carcinoid tumors of the lung are highly vascular, they do not show increased metabolic activity on positron-emission tomography (PET) and would be incorrectly designated as benign lesions on the basis of findings from a PET scan. Fludeoxyglucose F-18 PET has shown utility as a radiologic marker for atypical carcinoids, particularly for those with a higher proliferation index with Ki-67 index of 10%-20%.6
Radionucleotide studies
Somatostatin receptors (SSRs) are present in many tumors of neuroendocrine origin, including carcinoid tumors. These receptors interact with each other and undergo dimerization and internalization. SSTR subtypes (SSTRs) overexpressed in NETs are related to the type, origin, and grade of differentiation of tumor. The overexpression of an SSTR is a characteristic feature of bronchial NETs, which can be used to localize the primary tumor and its metastases by imaging with the radiolabeled SST analogues. Radionucleotide imaging modalities commonly used include single-photon–emission tomography and positron-emission tomography.
With regard to SSR scintigraphy testing, PET using Ga–DOTATATE/TOC is preferable to Octreoscan if it is available, because offers better spatial resolution and has a shorter scanning time. It has sensitivity of 97% and specificity of 92% and hence is preferable over Octreoscan in highly aggressive, atypical bronchial NETs. It also provides an estimate of receptor density and evidence of the functionality of receptors, which helps with selection of suitable treatments that act on these receptors.7
Tumor markers
Serum levels of chromogranin A in bronchial NETs are expressed at a lower rate than are other sites of carcinoid tumors, so its measurement is of limited utility in following disease activity in bronchial NETs.4,8
Bronchoscopy
About 75% of pulmonary carcinoids are visible on bronchoscopy. The bronchoscopic appearance may be characteristic but it is preferable that brushings or biopsy be performed to confirm the diagnosis. For central tumors endobronchial; and for peripheral tumors, CT-guided percutaneous biopsy is the accepted diagnostic approach. Cytologic study of bronchial brushings is more sensitive than sputum cytology, but the diagnostic yield of brushing is low overall (about 40%) and hence fine-needle biopsy is preferred. 8
A negative finding on biopsy should not produce a false sense of confidence. If a suspicion of malignancy exists despite a negative finding on transthoracic biopsy, surgical excision of the nodule and pathologic analysis should be undertaken.
Histological findings
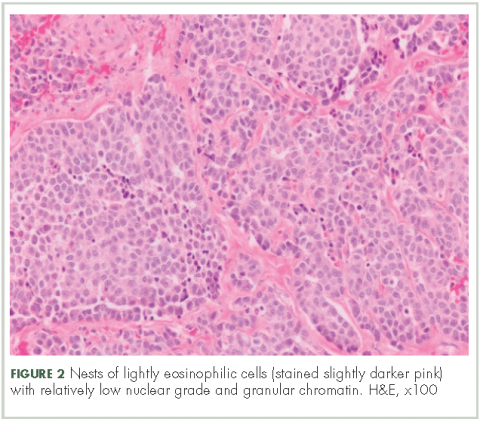
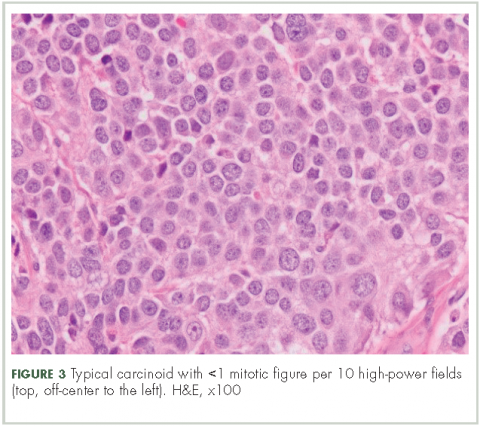
In typical carcinoid tumors, cells tend to group in nests, cords, or broad sheets. Arrangement is orderly, with groups of cells separated by highly vascular septa of connective tissue.9Individual cell features include small and polygonal cells with finely granular eosinophilic cytoplasm (Figure 2). Nuclei are small and round. Mitoses are infrequent (Figure 3).
On electron microscopy, well-formed desmosomes and abundant neurosecretory granules are seen. Many pulmonary carcinoid tumors stain positive for a variety of neuroendocrine markers. Electron microscopy is of historical interest but is not used for tissue diagnosis for bronchial carcinoid patients.
Typical vs atypical tumors
In all, 10% of the carcinoid tumors are atypical in nature. They are generally larger than typical carcinoids and are located in the periphery of the lung in about 50% of cases. They have more aggressive behavior and tend to metastasize more commonly.2 Neither location nor size are distinguishing features. The distinction is based on histology and includes one or all of the following features:8,9
n Increased mitotic activity in a tumor with an identifiable carcinoid cellular arrangement with 2-10 mitotic figures per high-power field.9
n Pleomorphism and irregular nuclei with hyperchromatism and prominent nucleoli.
n Areas of increased cellularity with loss of the regular, organized architecture observed in typical carcinoid.
n Areas of necrosis within the tumor.
Ki-67 cell proliferation labeling index can be used to distinguish between high-grade lung NETs (>40%) and carcinoids (<20%), particularly in crushed biopsy specimens in which carcinoids may be mistaken for small-cell lung cancers. However, given overlap in the distribution of Ki-67 labeling index between typical carcinoids (≤5%) and atypical carcinoids (≤20%), Ki-67 expression does not reliably distinguish between well-differentiated lung carcinoids. The utility of Ki-67 to differentiate between typical and atypical carcinoids has yet to be established, and it is not presently recommended.9 Hence, the number of mitotic figures per high-power field of viable tumor area and presence or absence of necrosis continue to be the salient features distinguishing typical and atypical bronchial NETs.
Staging10
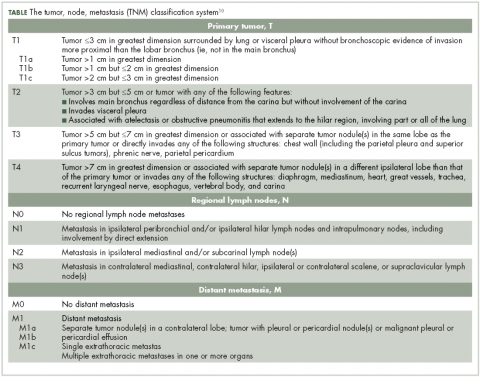
Lung NETs are staged using the same tumor, node, metastasis (TNM) classification from the American Joint Committee on Cancer (AJCC) that is used for bronchogenic lung carcinomas (Table).
Typical bronchial NETs most commonly present as stage I tumors, whereas more than one-half of atypical tumors are stage II (bronchopulmonary nodal involvement) or III (mediastinal nodal involvement) at presentation.
Treatment
Localized or nonmetastatic and rescetable disease
Surgical treatment. As with other non–small-cell lung cancers (NSCLCs), surgical resection is the treatment of choice for early-stage carcinoid. The long-term prognosis is typically excellent, with a 10-year disease-free survival of 77%-94%.11, 12 The extent of resection is determined by the tumor size, histology, and location. For NSCLC, the standard surgical approach is the minimal anatomic resection (lobectomy, sleeve lobectomy, bilobectomy, or pneumonectomy) needed to get microscopically negative margins, with an associated mediastinal and hilar lymph node dissection for staging.13
Given the indolent nature of typical carcinoids, there has been extensive research to evaluate whether a sublobar resection is oncologically appropriate for these tumors. Although there are no comprehensive randomized studies comparing sublobar resection with lobectomy for typical carcinoids, findings from numerous database reviews and single-center studies suggest that sublobar resections are noninferior.14-17 Due to the higher nodal metastatic rate and the overall poorer prognosis associated with atypical carcinoids, formal anatomic resection is still recommended with atypical histology.18
An adaptive approach must be taken for patients who undergo wedge resection of pulmonary lesions without a known diagnosis. If intraoperative frozen section is consistent with carcinoid and the margins are negative, mediastinal lymph node dissection should be performed. If the patient is node negative, then completion lobectomy is not required. In node-positive patients with adequate pulmonary reserve, lobectomy should be performed regardless of histology. If atypical features are found during pathologic evaluation, then interval completion lobectomy may be patients with adequate pulmonary reserve.19,20
As with other pulmonary malignancies, clinical or radiographic suspicion of mediastinal lymph node involvement requires invasive staging before pulmonary resection is considered. If the patient is proven to have mediastinal metastatic disease, then multimodality treatment should be considered.20
Adjuvant therapy. Postoperative adjuvant therapy for most resected bronchial NETs, even in the setting of positive lymph nodes, is generally not recommended.7 In clinical practice, adjuvant platinum-based chemotherapy with or without radiation therapy (RT) is a reasonable option for patients with histologically aggressive-appearing or poorly differentiated stage III atypical bronchial NETs, although there is only limited evidence to support this. RT is a reasonable option for atypical bronchial NETs if gross residual disease remains after surgery, although it has not been proven that this improves outcomes.7
Nonmetastatic and unresectable disease
For inoperable patients and for those with surgically unresectable but nonmetastatic disease, options for local control of tumor growth include RT with or without concurrent chemotherapy and palliative endobronchial resection of obstructing tumor.21
Metastatic and unresectable disease
Everolimus. In February 2016, everolimus was approved by the US Food and Drug Administration (FDA) as first-line therapy for progressive, well-differentiated, nonfunctional NETs of lung origin that are unresectable, locally advanced, or metastatic. The aApproval was based on the RADIANT-4 trial, in which median progression-free survival was 11 months in the 205 patients allocated to receive everolimus (10 mg/day) and 3.9 months in the 97 patients who received placebo. Everolimus was associated with a 52% reduction in the estimated risk of progression or death.22
Somastatin analogues (SSA). There is lack of comprehensive data on the role of SSA compared with everolimus in lung carcinoid. The National Comprehensive Cancer Network (NCCN) guidelines on NETs and SCLCs recommend the consideration of octreotide or lanreotide as first-line therapies for select patients with symptoms of carcinoid syndrome or octreotide-positive scans.21 Guidelines from the European Neuroendocrine Tumor Society (ENETS)19 also recommend the use of SSAs as a first-line option in patients with: lung carcinoids exhibiting hormone-related symptoms or slowly progressive typical or atypical carcinoid with a low proliferative index (preferably Ki-67 <10%), provided there is a strongly positive SSTR status.
In cases in which metastatic lung NETs are associated with the carcinoid syndrome, initiation of long-acting SSA therapy in combination with everolimus is recommended.
Cytotoxic chemotherapy. According to the NCCN guidelines, cisplatin-etoposide or other cytotoxic regimens (eg, those that are temozolomide based) are recommended for advanced typical and atypical carcinoids, with cisplatin-etoposide being the preferred first-line systemic regimen in stage IV atypical carcinoid.22 ENETS guidelines stipulate that systemic chemotherapy is generally restricted to atypical carcinoid after failure of first-line therapies and only under certain conditions (Ki-67 >15%, rapidly progressive disease, and SSTR-negative disease).19 Based on a summary of NCCN and ENET guidelines:
n For patients with highly aggressive atypical bronchial NETs, a combination of platinum- and etoposide-based regimens such as those used for small-cell lung cancer has shown better response rate and overall survival data.
n For patients with typical or atypical bronchial NETs, temozolomide can be used as monotherapy or combination with capecitabine, although there are no findings from large randomized controlled trials to support this. Capecitabine-temozolomide has recently shown moderate activity in a small, single-institution study of patients with advanced lung carcinoids (N = 19), with 11 of 17 assessable patients (65%) demonstrating stable disease or partial response.23
n The following regimens can also be used for advanced disease after failure of somastatin analogues, although there are limited data for objective responses:24,25fluorouracil plus dacarbazine; epirubicin, capecitabine plus oxaliplatin; and capecitabine plus liposomal doxorubicin.
Participation in a clinical trial should be encouraged for patients with progressive bronchial NETs during any line of therapy. For patients who have a limited, potentially resectable liver-isolated metastatic NET, surgical resection should be pursued. For more extensive unresectable liver-dominant metastatic disease, treatment options include embolization, radiofrequency ablation, and cryoablation.20,22
Posttreatment surveillance
Posttreatment surveillance after resection of node-positive typical bronchial NETs and for all atypical tumors.26 Patients with lymph-node–negative typical bronchial NETs are very unlikely to benefit from postoperative surveillance because of the very low risk of recurrence. CT imaging (including the thorax and abdomen) every 6 months for 2 years, followed by annual scans for a total of 5-10 years are a reasonable surveillance schedule.
Prognosis 18,27
Typical bronchial NETs have an excellent prognosis after surgical resection. Reported 5-year survival rates are 87%-100%; the corresponding rates at 10 years are 82%-87%. Features associated with negative prognostic significance include lymph-node involvement and incomplete resection.
Atypical bronchial NETs have a worse prognosis than do typical tumors. Five-year survival rates range widely, from 30%-95%; the corresponding rates at 10 years are 35%-56%. Atypical tumors have a greater tendency to metastasize (16%-23%) and recur locally (3%-25%). Distant metastases to the liver or bone are more common than local recurrence. Adverse influence of nodal metastases on prognosis is more profound than for typical tumors. Survival rates by stage for patients who underwent surgical resection (including typical and atypical carcinoid27) are: stage I, 93%; stage II, 85%; stage III, 75%; and stage IV, 57%.
1. Hauso O, Gustafsson BI, Kidd M, et al. Neuroendocrine tumor epidemiology: contrasting Norway and North America. Cancer. 2008;113(10):2655-2664.
2. Fink G, Krelbaum T, Yellin A, et al. Pulmonary carcinoid: presentation, diagnosis, and outcome in 142 cases in Israel and review of 640 cases from the literature. Chest. 2001;119(6):1647-1651.
3. Limper AH, Carpenter PC, Scheithauer B, Staats BA. The Cushing syndrome induced by bronchial carcinoid tumors. Ann Intern Med. 1992;117(3):209-214.
4. Meisinger QC, Klein JS, Butnor KJ, Gentchos G, Leavitt BJ. CT features of peripheral pulmonary carcinoid tumors. AJR Am J Roentgenol. 2011;197(5):1073-1080.
5. Guckel C, Schnabel K, Deimling M, Steinbrich W. Solitary pulmonary nodules: MR evaluation of enhancement patterns with contrast-enhanced dynamic snapshot gradient-echo imaging. Radiology. 1996;200(3):681-686.
6. Jindal T, Kumar A, Venkitaraman B, et al. Evaluation of the role of [18F]FDG-PET/CT and [68Ga]DOTATOC-PET/CT in differentiating typical and atypical pulmonary carcinoids. Cancer Imaging. 2011;11:70-75.
7. Caplin ME, Baudin E, Ferolla P, et al. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604-1620.
8. Travis WD. Pathology and diagnosis of neuroendocrine tumors: lung neuroendocrine. Thorac Surg Clin. 2014;24(3):257-266.
9. Warren WH, Memoli VA, Gould VE. Immunohistochemical and ultrastructural analysis of bronchopulmonary neuroendocrine neoplasms. II. Well-differentiated neuroendocrine carcinomas. Ultrastruct Pathol. 1984;7(2-3):185-199.
10. Goldstraw P, Chansky K, Crowley J, et al. The IASLC Lung Cancer Staging Project: Proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J Thorac Oncol. 2016;11(1):39-51.
11. McCaughan BC, Martini N, Bains MS. Bronchial carcinoids. Review of 124 cases. J Thorac Cardiovasc Surg. 1985;89(1):8-17.
12. Hurt R, Bates M. Carcinoid tumours of the bronchus: a 33-year experience. Thorax. 1984;39(8):617-623.
13. Ettinger DS, Wood DE, Akerley W, et al. Non-small cell lung cancer, version 1.2015. J Natl Compr Cancer Netw. 2014;12(12):1738-1761.
14. Ferguson MK, Landreneau RJ, Hazelrigg SR, et al. Long-term outcome after resection for bronchial carcinoid tumors. Eur J Cardiothorac Surg. 2000;18(2):156-161.
15. Lucchi M, Melfi F, Ribechini A, et al. Sleeve and wedge parenchyma-sparing bronchial resections in low-grade neoplasms of the bronchial airway. J Thorac Cardiovasc Surg. 2007;134(2):373-377.
16. Yendamuri S, Gold D, Jayaprakash V, Dexter E, Nwogu C, Demmy T. Is sublobar resection sufficient for carcinoid tumors? Ann Thorac Surg. 2011;92(5):1774-1778; discussion 8-9.
17. Fox M, Van Berkel V, Bousamra M II, Sloan S, Martin RC II. Surgical management of pulmonary carcinoid tumors: sublobar resection versus lobectomy. Am J Surg. 2013;205(2):200-208.
18. Cardillo G, Sera F, Di Martino M, et al. Bronchial carcinoid tumors: nodal status and long-term survival after resection. Ann Thorac Surg. 2004;77(5):1781-1785.
19. Oberg K, Hellman P, Ferolla P, Papotti M; ESMO Guidelines Working Group. Neuroendocrine bronchial and thymic tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(suppl 7:vii120-3).
20. Filosso PL, Ferolla P, Guerrera F, et al. Multidisciplinary management of advanced lung neuroendocrine tumors. J Thorac Dis. 2015;7(Suppl 2):S163-171.
21. Kulke MH, Shah MH, Benson AB III, et al. Neuroendocrine tumors, version 1.2015. J Natl Compr Cancer Netw. 2015;13(1):78-108.
22. Yao JC, Fazio N, Singh S, et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): a randomised, placebo-controlled, phase 3 study. Lancet. 2016;387(10022):968-977.
23. Ramirez RA, Beyer DT, Chauhan A, Boudreaux JP, Wang YZ, Woltering EA. The role of capecitabine/temozolomide in metastatic neuroendocrine tumors. Oncologist. 2016;21(6):671-675.
24. Bajetta E, Rimassa L, Carnaghi C, et al. 5-Fluorouracil, dacarbazine, and epirubicin in the treatment of patients with neuroendocrine tumors. Cancer. 1998;83(2):372-378.
25. Masi G, Fornaro L, Cupini S, et al. Refractory neuroendocrine tumor-response to liposomal doxorubicin and capecitabine. Nat Rev Clin Oncol. 2009;6(11):670-674.
26. Lou F, Sarkaria I, Pietanza C, et al. Recurrence of pulmonary carcinoid tumors after resection: implications for postoperative surveillance. Ann Thorac Surg. 2013;96(4):1156-1162.
27. Beasley MB, Thunnissen FB, Brambilla E, et al. Pulmonary atypical carcinoid: predictors of survival in 106 cases. Human Pathol. 2000;31(10):1255-1265.
Carcinoid lung tumors represent the most indolent form of a spectrum of bronchopulmonary neuroendocrine tumors (NETs) that includes small-cell carcinoma as its most malignant member, as well as several other forms of intermediately aggressive tumors, such as atypical carcinoid.1 Carcinoids represent 1.2% of all primary lung malignancies. Their incidence in the United States has increased rapidly over the last 30 years and is currently about 6% a year. Lung carcinoids are more prevalent in whites compared with blacks, and in Asians compared with non-Asians. They are less common in Hispanics compared with non-Hispanics.1 Typical carcinoids represent 80%-90% of all lung carcinoids and occur more frequently in the fifth and sixth decades of life. They can, however, occur at any age, and are the most common lung tumor in childhood.1
Etiology and risk factors
Unlike carcinoma of the lung, no external environmental toxin or other stimulus has been identified as a causative agent for the development of pulmonary carcinoid tumors. It is not clear if there is an association between bronchial NETs and smoking.1 Nearly all bronchial NETs are sporadic; however, they can rarely occur in the setting of multiple endocrine neoplasia type 1.1
Presentation
About 60% of the patients with bronchial carcinoids are symptomatic at presentation. The most common clinical findings are those associated with bronchial obstruction, such as persistent cough, hemoptysis, and recurrent or obstructive pneumonitis. Wheezing, chest pain, and dyspnea also may be noted.2 Various endocrine or neuroendocrine syndromes can be initial clinical manifestations of either typical or atypical pulmonary carcinoid tumors, but that is not common Cushing syndrome (ectopic production and secretion of adrenocorticotropic hormone [ACTH]) may occur in about 2% of lung carcinoid.3 In cases of malignancy, the presence of metastatic disease can produce weight loss, weakness, and a general feeling of ill health.
Diagnostic work-up
Biochemical test
There is no biochemical study that can be used as a screening test to determine the presence of a carcinoid tumor or to diagnose a known pulmonary mass as a carcinoid tumor. Neuroendocrine cells produce biologically active amines and peptides that can be detected in serum and urine. Although the syndromes associated with lung carcinoids are seen in about 1%-2% of the patients, assays of specific hormones or other circulating neuroendocrine substances, such as ACTH, melanocyte-stimulating hormone, or growth hormone may establish the existence of a clinically suspected syndrome.
Chest radiography
An abnormal finding on chest radiography is present in about 75% of patients with a pulmonary carcinoid tumor.1 Findings include either the presence of the tumor mass itself or indirect evidence of its presence observed as parenchymal changes associated with bronchial obstruction from the mass.
Computed-tomography imaging
High-resolution computed-tomography (CT) imaging is the one of the best types of CT examination for evaluation of a pulmonary carcinoid tumor.4 A CT scan provides excellent resolution of tumor extent, location, and the presence or absence of mediastinal adenopathy. It also aids in morphologic characterization of peripheral (Figure 1) and especially centrally located carcinoids, which may be purely intraluminal (polypoid configuration), exclusively extra luminal, or more frequently, a mixture of intraluminal and extraluminal components.
CT scans may also be helpful for differentiating tumor from postobstructive atelectasis or bronchial obstruction-related mucoid impaction. Intravenous contrast in CT imaging can be useful in differentiating malignant from benign lesions. Because carcinoid tumors are highly vascular, they show greater enhancement on contrast CT than do benign lesions. The sensitivity of CT for detecting metastatic hilar or mediastinal nodes is high, but specificity is as low as 45%.4
Typical carcinoid is rarely metastatic so most patients do not need CT or MRI imaging to evaluate for liver involvement. Liver imaging is appropriate in patients with evidence of mediastinal involvement, relatively high mitotic rate, or clinical evidence of the carcinoid syndrome.8 To evaluate for metastatic spread to the liver, multiphase contrast-enhanced liver CT scans should be performed with arterial and portal-venous phases because carcinoid liver metastases are often hypervascular and appear isodense relative to the liver parenchyma after contrast administration.4 An MRI is often preferred the modality to evaluate for metastatic spread to the liver because of its higher sensitivity.5
Positron-emission tomography
Although carcinoid tumors of the lung are highly vascular, they do not show increased metabolic activity on positron-emission tomography (PET) and would be incorrectly designated as benign lesions on the basis of findings from a PET scan. Fludeoxyglucose F-18 PET has shown utility as a radiologic marker for atypical carcinoids, particularly for those with a higher proliferation index with Ki-67 index of 10%-20%.6
Radionucleotide studies
Somatostatin receptors (SSRs) are present in many tumors of neuroendocrine origin, including carcinoid tumors. These receptors interact with each other and undergo dimerization and internalization. SSTR subtypes (SSTRs) overexpressed in NETs are related to the type, origin, and grade of differentiation of tumor. The overexpression of an SSTR is a characteristic feature of bronchial NETs, which can be used to localize the primary tumor and its metastases by imaging with the radiolabeled SST analogues. Radionucleotide imaging modalities commonly used include single-photon–emission tomography and positron-emission tomography.
With regard to SSR scintigraphy testing, PET using Ga–DOTATATE/TOC is preferable to Octreoscan if it is available, because offers better spatial resolution and has a shorter scanning time. It has sensitivity of 97% and specificity of 92% and hence is preferable over Octreoscan in highly aggressive, atypical bronchial NETs. It also provides an estimate of receptor density and evidence of the functionality of receptors, which helps with selection of suitable treatments that act on these receptors.7
Tumor markers
Serum levels of chromogranin A in bronchial NETs are expressed at a lower rate than are other sites of carcinoid tumors, so its measurement is of limited utility in following disease activity in bronchial NETs.4,8
Bronchoscopy
About 75% of pulmonary carcinoids are visible on bronchoscopy. The bronchoscopic appearance may be characteristic but it is preferable that brushings or biopsy be performed to confirm the diagnosis. For central tumors endobronchial; and for peripheral tumors, CT-guided percutaneous biopsy is the accepted diagnostic approach. Cytologic study of bronchial brushings is more sensitive than sputum cytology, but the diagnostic yield of brushing is low overall (about 40%) and hence fine-needle biopsy is preferred. 8
A negative finding on biopsy should not produce a false sense of confidence. If a suspicion of malignancy exists despite a negative finding on transthoracic biopsy, surgical excision of the nodule and pathologic analysis should be undertaken.
Histological findings
In typical carcinoid tumors, cells tend to group in nests, cords, or broad sheets. Arrangement is orderly, with groups of cells separated by highly vascular septa of connective tissue.9Individual cell features include small and polygonal cells with finely granular eosinophilic cytoplasm (Figure 2). Nuclei are small and round. Mitoses are infrequent (Figure 3).
On electron microscopy, well-formed desmosomes and abundant neurosecretory granules are seen. Many pulmonary carcinoid tumors stain positive for a variety of neuroendocrine markers. Electron microscopy is of historical interest but is not used for tissue diagnosis for bronchial carcinoid patients.
Typical vs atypical tumors
In all, 10% of the carcinoid tumors are atypical in nature. They are generally larger than typical carcinoids and are located in the periphery of the lung in about 50% of cases. They have more aggressive behavior and tend to metastasize more commonly.2 Neither location nor size are distinguishing features. The distinction is based on histology and includes one or all of the following features:8,9
n Increased mitotic activity in a tumor with an identifiable carcinoid cellular arrangement with 2-10 mitotic figures per high-power field.9
n Pleomorphism and irregular nuclei with hyperchromatism and prominent nucleoli.
n Areas of increased cellularity with loss of the regular, organized architecture observed in typical carcinoid.
n Areas of necrosis within the tumor.
Ki-67 cell proliferation labeling index can be used to distinguish between high-grade lung NETs (>40%) and carcinoids (<20%), particularly in crushed biopsy specimens in which carcinoids may be mistaken for small-cell lung cancers. However, given overlap in the distribution of Ki-67 labeling index between typical carcinoids (≤5%) and atypical carcinoids (≤20%), Ki-67 expression does not reliably distinguish between well-differentiated lung carcinoids. The utility of Ki-67 to differentiate between typical and atypical carcinoids has yet to be established, and it is not presently recommended.9 Hence, the number of mitotic figures per high-power field of viable tumor area and presence or absence of necrosis continue to be the salient features distinguishing typical and atypical bronchial NETs.
Staging10
Lung NETs are staged using the same tumor, node, metastasis (TNM) classification from the American Joint Committee on Cancer (AJCC) that is used for bronchogenic lung carcinomas (Table).
Typical bronchial NETs most commonly present as stage I tumors, whereas more than one-half of atypical tumors are stage II (bronchopulmonary nodal involvement) or III (mediastinal nodal involvement) at presentation.
Treatment
Localized or nonmetastatic and rescetable disease
Surgical treatment. As with other non–small-cell lung cancers (NSCLCs), surgical resection is the treatment of choice for early-stage carcinoid. The long-term prognosis is typically excellent, with a 10-year disease-free survival of 77%-94%.11, 12 The extent of resection is determined by the tumor size, histology, and location. For NSCLC, the standard surgical approach is the minimal anatomic resection (lobectomy, sleeve lobectomy, bilobectomy, or pneumonectomy) needed to get microscopically negative margins, with an associated mediastinal and hilar lymph node dissection for staging.13
Given the indolent nature of typical carcinoids, there has been extensive research to evaluate whether a sublobar resection is oncologically appropriate for these tumors. Although there are no comprehensive randomized studies comparing sublobar resection with lobectomy for typical carcinoids, findings from numerous database reviews and single-center studies suggest that sublobar resections are noninferior.14-17 Due to the higher nodal metastatic rate and the overall poorer prognosis associated with atypical carcinoids, formal anatomic resection is still recommended with atypical histology.18
An adaptive approach must be taken for patients who undergo wedge resection of pulmonary lesions without a known diagnosis. If intraoperative frozen section is consistent with carcinoid and the margins are negative, mediastinal lymph node dissection should be performed. If the patient is node negative, then completion lobectomy is not required. In node-positive patients with adequate pulmonary reserve, lobectomy should be performed regardless of histology. If atypical features are found during pathologic evaluation, then interval completion lobectomy may be patients with adequate pulmonary reserve.19,20
As with other pulmonary malignancies, clinical or radiographic suspicion of mediastinal lymph node involvement requires invasive staging before pulmonary resection is considered. If the patient is proven to have mediastinal metastatic disease, then multimodality treatment should be considered.20
Adjuvant therapy. Postoperative adjuvant therapy for most resected bronchial NETs, even in the setting of positive lymph nodes, is generally not recommended.7 In clinical practice, adjuvant platinum-based chemotherapy with or without radiation therapy (RT) is a reasonable option for patients with histologically aggressive-appearing or poorly differentiated stage III atypical bronchial NETs, although there is only limited evidence to support this. RT is a reasonable option for atypical bronchial NETs if gross residual disease remains after surgery, although it has not been proven that this improves outcomes.7
Nonmetastatic and unresectable disease
For inoperable patients and for those with surgically unresectable but nonmetastatic disease, options for local control of tumor growth include RT with or without concurrent chemotherapy and palliative endobronchial resection of obstructing tumor.21
Metastatic and unresectable disease
Everolimus. In February 2016, everolimus was approved by the US Food and Drug Administration (FDA) as first-line therapy for progressive, well-differentiated, nonfunctional NETs of lung origin that are unresectable, locally advanced, or metastatic. The aApproval was based on the RADIANT-4 trial, in which median progression-free survival was 11 months in the 205 patients allocated to receive everolimus (10 mg/day) and 3.9 months in the 97 patients who received placebo. Everolimus was associated with a 52% reduction in the estimated risk of progression or death.22
Somastatin analogues (SSA). There is lack of comprehensive data on the role of SSA compared with everolimus in lung carcinoid. The National Comprehensive Cancer Network (NCCN) guidelines on NETs and SCLCs recommend the consideration of octreotide or lanreotide as first-line therapies for select patients with symptoms of carcinoid syndrome or octreotide-positive scans.21 Guidelines from the European Neuroendocrine Tumor Society (ENETS)19 also recommend the use of SSAs as a first-line option in patients with: lung carcinoids exhibiting hormone-related symptoms or slowly progressive typical or atypical carcinoid with a low proliferative index (preferably Ki-67 <10%), provided there is a strongly positive SSTR status.
In cases in which metastatic lung NETs are associated with the carcinoid syndrome, initiation of long-acting SSA therapy in combination with everolimus is recommended.
Cytotoxic chemotherapy. According to the NCCN guidelines, cisplatin-etoposide or other cytotoxic regimens (eg, those that are temozolomide based) are recommended for advanced typical and atypical carcinoids, with cisplatin-etoposide being the preferred first-line systemic regimen in stage IV atypical carcinoid.22 ENETS guidelines stipulate that systemic chemotherapy is generally restricted to atypical carcinoid after failure of first-line therapies and only under certain conditions (Ki-67 >15%, rapidly progressive disease, and SSTR-negative disease).19 Based on a summary of NCCN and ENET guidelines:
n For patients with highly aggressive atypical bronchial NETs, a combination of platinum- and etoposide-based regimens such as those used for small-cell lung cancer has shown better response rate and overall survival data.
n For patients with typical or atypical bronchial NETs, temozolomide can be used as monotherapy or combination with capecitabine, although there are no findings from large randomized controlled trials to support this. Capecitabine-temozolomide has recently shown moderate activity in a small, single-institution study of patients with advanced lung carcinoids (N = 19), with 11 of 17 assessable patients (65%) demonstrating stable disease or partial response.23
n The following regimens can also be used for advanced disease after failure of somastatin analogues, although there are limited data for objective responses:24,25fluorouracil plus dacarbazine; epirubicin, capecitabine plus oxaliplatin; and capecitabine plus liposomal doxorubicin.
Participation in a clinical trial should be encouraged for patients with progressive bronchial NETs during any line of therapy. For patients who have a limited, potentially resectable liver-isolated metastatic NET, surgical resection should be pursued. For more extensive unresectable liver-dominant metastatic disease, treatment options include embolization, radiofrequency ablation, and cryoablation.20,22
Posttreatment surveillance
Posttreatment surveillance after resection of node-positive typical bronchial NETs and for all atypical tumors.26 Patients with lymph-node–negative typical bronchial NETs are very unlikely to benefit from postoperative surveillance because of the very low risk of recurrence. CT imaging (including the thorax and abdomen) every 6 months for 2 years, followed by annual scans for a total of 5-10 years are a reasonable surveillance schedule.
Prognosis 18,27
Typical bronchial NETs have an excellent prognosis after surgical resection. Reported 5-year survival rates are 87%-100%; the corresponding rates at 10 years are 82%-87%. Features associated with negative prognostic significance include lymph-node involvement and incomplete resection.
Atypical bronchial NETs have a worse prognosis than do typical tumors. Five-year survival rates range widely, from 30%-95%; the corresponding rates at 10 years are 35%-56%. Atypical tumors have a greater tendency to metastasize (16%-23%) and recur locally (3%-25%). Distant metastases to the liver or bone are more common than local recurrence. Adverse influence of nodal metastases on prognosis is more profound than for typical tumors. Survival rates by stage for patients who underwent surgical resection (including typical and atypical carcinoid27) are: stage I, 93%; stage II, 85%; stage III, 75%; and stage IV, 57%.
Carcinoid lung tumors represent the most indolent form of a spectrum of bronchopulmonary neuroendocrine tumors (NETs) that includes small-cell carcinoma as its most malignant member, as well as several other forms of intermediately aggressive tumors, such as atypical carcinoid.1 Carcinoids represent 1.2% of all primary lung malignancies. Their incidence in the United States has increased rapidly over the last 30 years and is currently about 6% a year. Lung carcinoids are more prevalent in whites compared with blacks, and in Asians compared with non-Asians. They are less common in Hispanics compared with non-Hispanics.1 Typical carcinoids represent 80%-90% of all lung carcinoids and occur more frequently in the fifth and sixth decades of life. They can, however, occur at any age, and are the most common lung tumor in childhood.1
Etiology and risk factors
Unlike carcinoma of the lung, no external environmental toxin or other stimulus has been identified as a causative agent for the development of pulmonary carcinoid tumors. It is not clear if there is an association between bronchial NETs and smoking.1 Nearly all bronchial NETs are sporadic; however, they can rarely occur in the setting of multiple endocrine neoplasia type 1.1
Presentation
About 60% of the patients with bronchial carcinoids are symptomatic at presentation. The most common clinical findings are those associated with bronchial obstruction, such as persistent cough, hemoptysis, and recurrent or obstructive pneumonitis. Wheezing, chest pain, and dyspnea also may be noted.2 Various endocrine or neuroendocrine syndromes can be initial clinical manifestations of either typical or atypical pulmonary carcinoid tumors, but that is not common Cushing syndrome (ectopic production and secretion of adrenocorticotropic hormone [ACTH]) may occur in about 2% of lung carcinoid.3 In cases of malignancy, the presence of metastatic disease can produce weight loss, weakness, and a general feeling of ill health.
Diagnostic work-up
Biochemical test
There is no biochemical study that can be used as a screening test to determine the presence of a carcinoid tumor or to diagnose a known pulmonary mass as a carcinoid tumor. Neuroendocrine cells produce biologically active amines and peptides that can be detected in serum and urine. Although the syndromes associated with lung carcinoids are seen in about 1%-2% of the patients, assays of specific hormones or other circulating neuroendocrine substances, such as ACTH, melanocyte-stimulating hormone, or growth hormone may establish the existence of a clinically suspected syndrome.
Chest radiography
An abnormal finding on chest radiography is present in about 75% of patients with a pulmonary carcinoid tumor.1 Findings include either the presence of the tumor mass itself or indirect evidence of its presence observed as parenchymal changes associated with bronchial obstruction from the mass.
Computed-tomography imaging
High-resolution computed-tomography (CT) imaging is the one of the best types of CT examination for evaluation of a pulmonary carcinoid tumor.4 A CT scan provides excellent resolution of tumor extent, location, and the presence or absence of mediastinal adenopathy. It also aids in morphologic characterization of peripheral (Figure 1) and especially centrally located carcinoids, which may be purely intraluminal (polypoid configuration), exclusively extra luminal, or more frequently, a mixture of intraluminal and extraluminal components.
CT scans may also be helpful for differentiating tumor from postobstructive atelectasis or bronchial obstruction-related mucoid impaction. Intravenous contrast in CT imaging can be useful in differentiating malignant from benign lesions. Because carcinoid tumors are highly vascular, they show greater enhancement on contrast CT than do benign lesions. The sensitivity of CT for detecting metastatic hilar or mediastinal nodes is high, but specificity is as low as 45%.4
Typical carcinoid is rarely metastatic so most patients do not need CT or MRI imaging to evaluate for liver involvement. Liver imaging is appropriate in patients with evidence of mediastinal involvement, relatively high mitotic rate, or clinical evidence of the carcinoid syndrome.8 To evaluate for metastatic spread to the liver, multiphase contrast-enhanced liver CT scans should be performed with arterial and portal-venous phases because carcinoid liver metastases are often hypervascular and appear isodense relative to the liver parenchyma after contrast administration.4 An MRI is often preferred the modality to evaluate for metastatic spread to the liver because of its higher sensitivity.5
Positron-emission tomography
Although carcinoid tumors of the lung are highly vascular, they do not show increased metabolic activity on positron-emission tomography (PET) and would be incorrectly designated as benign lesions on the basis of findings from a PET scan. Fludeoxyglucose F-18 PET has shown utility as a radiologic marker for atypical carcinoids, particularly for those with a higher proliferation index with Ki-67 index of 10%-20%.6
Radionucleotide studies
Somatostatin receptors (SSRs) are present in many tumors of neuroendocrine origin, including carcinoid tumors. These receptors interact with each other and undergo dimerization and internalization. SSTR subtypes (SSTRs) overexpressed in NETs are related to the type, origin, and grade of differentiation of tumor. The overexpression of an SSTR is a characteristic feature of bronchial NETs, which can be used to localize the primary tumor and its metastases by imaging with the radiolabeled SST analogues. Radionucleotide imaging modalities commonly used include single-photon–emission tomography and positron-emission tomography.
With regard to SSR scintigraphy testing, PET using Ga–DOTATATE/TOC is preferable to Octreoscan if it is available, because offers better spatial resolution and has a shorter scanning time. It has sensitivity of 97% and specificity of 92% and hence is preferable over Octreoscan in highly aggressive, atypical bronchial NETs. It also provides an estimate of receptor density and evidence of the functionality of receptors, which helps with selection of suitable treatments that act on these receptors.7
Tumor markers
Serum levels of chromogranin A in bronchial NETs are expressed at a lower rate than are other sites of carcinoid tumors, so its measurement is of limited utility in following disease activity in bronchial NETs.4,8
Bronchoscopy
About 75% of pulmonary carcinoids are visible on bronchoscopy. The bronchoscopic appearance may be characteristic but it is preferable that brushings or biopsy be performed to confirm the diagnosis. For central tumors endobronchial; and for peripheral tumors, CT-guided percutaneous biopsy is the accepted diagnostic approach. Cytologic study of bronchial brushings is more sensitive than sputum cytology, but the diagnostic yield of brushing is low overall (about 40%) and hence fine-needle biopsy is preferred. 8
A negative finding on biopsy should not produce a false sense of confidence. If a suspicion of malignancy exists despite a negative finding on transthoracic biopsy, surgical excision of the nodule and pathologic analysis should be undertaken.
Histological findings
In typical carcinoid tumors, cells tend to group in nests, cords, or broad sheets. Arrangement is orderly, with groups of cells separated by highly vascular septa of connective tissue.9Individual cell features include small and polygonal cells with finely granular eosinophilic cytoplasm (Figure 2). Nuclei are small and round. Mitoses are infrequent (Figure 3).
On electron microscopy, well-formed desmosomes and abundant neurosecretory granules are seen. Many pulmonary carcinoid tumors stain positive for a variety of neuroendocrine markers. Electron microscopy is of historical interest but is not used for tissue diagnosis for bronchial carcinoid patients.
Typical vs atypical tumors
In all, 10% of the carcinoid tumors are atypical in nature. They are generally larger than typical carcinoids and are located in the periphery of the lung in about 50% of cases. They have more aggressive behavior and tend to metastasize more commonly.2 Neither location nor size are distinguishing features. The distinction is based on histology and includes one or all of the following features:8,9
n Increased mitotic activity in a tumor with an identifiable carcinoid cellular arrangement with 2-10 mitotic figures per high-power field.9
n Pleomorphism and irregular nuclei with hyperchromatism and prominent nucleoli.
n Areas of increased cellularity with loss of the regular, organized architecture observed in typical carcinoid.
n Areas of necrosis within the tumor.
Ki-67 cell proliferation labeling index can be used to distinguish between high-grade lung NETs (>40%) and carcinoids (<20%), particularly in crushed biopsy specimens in which carcinoids may be mistaken for small-cell lung cancers. However, given overlap in the distribution of Ki-67 labeling index between typical carcinoids (≤5%) and atypical carcinoids (≤20%), Ki-67 expression does not reliably distinguish between well-differentiated lung carcinoids. The utility of Ki-67 to differentiate between typical and atypical carcinoids has yet to be established, and it is not presently recommended.9 Hence, the number of mitotic figures per high-power field of viable tumor area and presence or absence of necrosis continue to be the salient features distinguishing typical and atypical bronchial NETs.
Staging10
Lung NETs are staged using the same tumor, node, metastasis (TNM) classification from the American Joint Committee on Cancer (AJCC) that is used for bronchogenic lung carcinomas (Table).
Typical bronchial NETs most commonly present as stage I tumors, whereas more than one-half of atypical tumors are stage II (bronchopulmonary nodal involvement) or III (mediastinal nodal involvement) at presentation.
Treatment
Localized or nonmetastatic and rescetable disease
Surgical treatment. As with other non–small-cell lung cancers (NSCLCs), surgical resection is the treatment of choice for early-stage carcinoid. The long-term prognosis is typically excellent, with a 10-year disease-free survival of 77%-94%.11, 12 The extent of resection is determined by the tumor size, histology, and location. For NSCLC, the standard surgical approach is the minimal anatomic resection (lobectomy, sleeve lobectomy, bilobectomy, or pneumonectomy) needed to get microscopically negative margins, with an associated mediastinal and hilar lymph node dissection for staging.13
Given the indolent nature of typical carcinoids, there has been extensive research to evaluate whether a sublobar resection is oncologically appropriate for these tumors. Although there are no comprehensive randomized studies comparing sublobar resection with lobectomy for typical carcinoids, findings from numerous database reviews and single-center studies suggest that sublobar resections are noninferior.14-17 Due to the higher nodal metastatic rate and the overall poorer prognosis associated with atypical carcinoids, formal anatomic resection is still recommended with atypical histology.18
An adaptive approach must be taken for patients who undergo wedge resection of pulmonary lesions without a known diagnosis. If intraoperative frozen section is consistent with carcinoid and the margins are negative, mediastinal lymph node dissection should be performed. If the patient is node negative, then completion lobectomy is not required. In node-positive patients with adequate pulmonary reserve, lobectomy should be performed regardless of histology. If atypical features are found during pathologic evaluation, then interval completion lobectomy may be patients with adequate pulmonary reserve.19,20
As with other pulmonary malignancies, clinical or radiographic suspicion of mediastinal lymph node involvement requires invasive staging before pulmonary resection is considered. If the patient is proven to have mediastinal metastatic disease, then multimodality treatment should be considered.20
Adjuvant therapy. Postoperative adjuvant therapy for most resected bronchial NETs, even in the setting of positive lymph nodes, is generally not recommended.7 In clinical practice, adjuvant platinum-based chemotherapy with or without radiation therapy (RT) is a reasonable option for patients with histologically aggressive-appearing or poorly differentiated stage III atypical bronchial NETs, although there is only limited evidence to support this. RT is a reasonable option for atypical bronchial NETs if gross residual disease remains after surgery, although it has not been proven that this improves outcomes.7
Nonmetastatic and unresectable disease
For inoperable patients and for those with surgically unresectable but nonmetastatic disease, options for local control of tumor growth include RT with or without concurrent chemotherapy and palliative endobronchial resection of obstructing tumor.21
Metastatic and unresectable disease
Everolimus. In February 2016, everolimus was approved by the US Food and Drug Administration (FDA) as first-line therapy for progressive, well-differentiated, nonfunctional NETs of lung origin that are unresectable, locally advanced, or metastatic. The aApproval was based on the RADIANT-4 trial, in which median progression-free survival was 11 months in the 205 patients allocated to receive everolimus (10 mg/day) and 3.9 months in the 97 patients who received placebo. Everolimus was associated with a 52% reduction in the estimated risk of progression or death.22
Somastatin analogues (SSA). There is lack of comprehensive data on the role of SSA compared with everolimus in lung carcinoid. The National Comprehensive Cancer Network (NCCN) guidelines on NETs and SCLCs recommend the consideration of octreotide or lanreotide as first-line therapies for select patients with symptoms of carcinoid syndrome or octreotide-positive scans.21 Guidelines from the European Neuroendocrine Tumor Society (ENETS)19 also recommend the use of SSAs as a first-line option in patients with: lung carcinoids exhibiting hormone-related symptoms or slowly progressive typical or atypical carcinoid with a low proliferative index (preferably Ki-67 <10%), provided there is a strongly positive SSTR status.
In cases in which metastatic lung NETs are associated with the carcinoid syndrome, initiation of long-acting SSA therapy in combination with everolimus is recommended.
Cytotoxic chemotherapy. According to the NCCN guidelines, cisplatin-etoposide or other cytotoxic regimens (eg, those that are temozolomide based) are recommended for advanced typical and atypical carcinoids, with cisplatin-etoposide being the preferred first-line systemic regimen in stage IV atypical carcinoid.22 ENETS guidelines stipulate that systemic chemotherapy is generally restricted to atypical carcinoid after failure of first-line therapies and only under certain conditions (Ki-67 >15%, rapidly progressive disease, and SSTR-negative disease).19 Based on a summary of NCCN and ENET guidelines:
n For patients with highly aggressive atypical bronchial NETs, a combination of platinum- and etoposide-based regimens such as those used for small-cell lung cancer has shown better response rate and overall survival data.
n For patients with typical or atypical bronchial NETs, temozolomide can be used as monotherapy or combination with capecitabine, although there are no findings from large randomized controlled trials to support this. Capecitabine-temozolomide has recently shown moderate activity in a small, single-institution study of patients with advanced lung carcinoids (N = 19), with 11 of 17 assessable patients (65%) demonstrating stable disease or partial response.23
n The following regimens can also be used for advanced disease after failure of somastatin analogues, although there are limited data for objective responses:24,25fluorouracil plus dacarbazine; epirubicin, capecitabine plus oxaliplatin; and capecitabine plus liposomal doxorubicin.
Participation in a clinical trial should be encouraged for patients with progressive bronchial NETs during any line of therapy. For patients who have a limited, potentially resectable liver-isolated metastatic NET, surgical resection should be pursued. For more extensive unresectable liver-dominant metastatic disease, treatment options include embolization, radiofrequency ablation, and cryoablation.20,22
Posttreatment surveillance
Posttreatment surveillance after resection of node-positive typical bronchial NETs and for all atypical tumors.26 Patients with lymph-node–negative typical bronchial NETs are very unlikely to benefit from postoperative surveillance because of the very low risk of recurrence. CT imaging (including the thorax and abdomen) every 6 months for 2 years, followed by annual scans for a total of 5-10 years are a reasonable surveillance schedule.
Prognosis 18,27
Typical bronchial NETs have an excellent prognosis after surgical resection. Reported 5-year survival rates are 87%-100%; the corresponding rates at 10 years are 82%-87%. Features associated with negative prognostic significance include lymph-node involvement and incomplete resection.
Atypical bronchial NETs have a worse prognosis than do typical tumors. Five-year survival rates range widely, from 30%-95%; the corresponding rates at 10 years are 35%-56%. Atypical tumors have a greater tendency to metastasize (16%-23%) and recur locally (3%-25%). Distant metastases to the liver or bone are more common than local recurrence. Adverse influence of nodal metastases on prognosis is more profound than for typical tumors. Survival rates by stage for patients who underwent surgical resection (including typical and atypical carcinoid27) are: stage I, 93%; stage II, 85%; stage III, 75%; and stage IV, 57%.
1. Hauso O, Gustafsson BI, Kidd M, et al. Neuroendocrine tumor epidemiology: contrasting Norway and North America. Cancer. 2008;113(10):2655-2664.
2. Fink G, Krelbaum T, Yellin A, et al. Pulmonary carcinoid: presentation, diagnosis, and outcome in 142 cases in Israel and review of 640 cases from the literature. Chest. 2001;119(6):1647-1651.
3. Limper AH, Carpenter PC, Scheithauer B, Staats BA. The Cushing syndrome induced by bronchial carcinoid tumors. Ann Intern Med. 1992;117(3):209-214.
4. Meisinger QC, Klein JS, Butnor KJ, Gentchos G, Leavitt BJ. CT features of peripheral pulmonary carcinoid tumors. AJR Am J Roentgenol. 2011;197(5):1073-1080.
5. Guckel C, Schnabel K, Deimling M, Steinbrich W. Solitary pulmonary nodules: MR evaluation of enhancement patterns with contrast-enhanced dynamic snapshot gradient-echo imaging. Radiology. 1996;200(3):681-686.
6. Jindal T, Kumar A, Venkitaraman B, et al. Evaluation of the role of [18F]FDG-PET/CT and [68Ga]DOTATOC-PET/CT in differentiating typical and atypical pulmonary carcinoids. Cancer Imaging. 2011;11:70-75.
7. Caplin ME, Baudin E, Ferolla P, et al. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604-1620.
8. Travis WD. Pathology and diagnosis of neuroendocrine tumors: lung neuroendocrine. Thorac Surg Clin. 2014;24(3):257-266.
9. Warren WH, Memoli VA, Gould VE. Immunohistochemical and ultrastructural analysis of bronchopulmonary neuroendocrine neoplasms. II. Well-differentiated neuroendocrine carcinomas. Ultrastruct Pathol. 1984;7(2-3):185-199.
10. Goldstraw P, Chansky K, Crowley J, et al. The IASLC Lung Cancer Staging Project: Proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J Thorac Oncol. 2016;11(1):39-51.
11. McCaughan BC, Martini N, Bains MS. Bronchial carcinoids. Review of 124 cases. J Thorac Cardiovasc Surg. 1985;89(1):8-17.
12. Hurt R, Bates M. Carcinoid tumours of the bronchus: a 33-year experience. Thorax. 1984;39(8):617-623.
13. Ettinger DS, Wood DE, Akerley W, et al. Non-small cell lung cancer, version 1.2015. J Natl Compr Cancer Netw. 2014;12(12):1738-1761.
14. Ferguson MK, Landreneau RJ, Hazelrigg SR, et al. Long-term outcome after resection for bronchial carcinoid tumors. Eur J Cardiothorac Surg. 2000;18(2):156-161.
15. Lucchi M, Melfi F, Ribechini A, et al. Sleeve and wedge parenchyma-sparing bronchial resections in low-grade neoplasms of the bronchial airway. J Thorac Cardiovasc Surg. 2007;134(2):373-377.
16. Yendamuri S, Gold D, Jayaprakash V, Dexter E, Nwogu C, Demmy T. Is sublobar resection sufficient for carcinoid tumors? Ann Thorac Surg. 2011;92(5):1774-1778; discussion 8-9.
17. Fox M, Van Berkel V, Bousamra M II, Sloan S, Martin RC II. Surgical management of pulmonary carcinoid tumors: sublobar resection versus lobectomy. Am J Surg. 2013;205(2):200-208.
18. Cardillo G, Sera F, Di Martino M, et al. Bronchial carcinoid tumors: nodal status and long-term survival after resection. Ann Thorac Surg. 2004;77(5):1781-1785.
19. Oberg K, Hellman P, Ferolla P, Papotti M; ESMO Guidelines Working Group. Neuroendocrine bronchial and thymic tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(suppl 7:vii120-3).
20. Filosso PL, Ferolla P, Guerrera F, et al. Multidisciplinary management of advanced lung neuroendocrine tumors. J Thorac Dis. 2015;7(Suppl 2):S163-171.
21. Kulke MH, Shah MH, Benson AB III, et al. Neuroendocrine tumors, version 1.2015. J Natl Compr Cancer Netw. 2015;13(1):78-108.
22. Yao JC, Fazio N, Singh S, et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): a randomised, placebo-controlled, phase 3 study. Lancet. 2016;387(10022):968-977.
23. Ramirez RA, Beyer DT, Chauhan A, Boudreaux JP, Wang YZ, Woltering EA. The role of capecitabine/temozolomide in metastatic neuroendocrine tumors. Oncologist. 2016;21(6):671-675.
24. Bajetta E, Rimassa L, Carnaghi C, et al. 5-Fluorouracil, dacarbazine, and epirubicin in the treatment of patients with neuroendocrine tumors. Cancer. 1998;83(2):372-378.
25. Masi G, Fornaro L, Cupini S, et al. Refractory neuroendocrine tumor-response to liposomal doxorubicin and capecitabine. Nat Rev Clin Oncol. 2009;6(11):670-674.
26. Lou F, Sarkaria I, Pietanza C, et al. Recurrence of pulmonary carcinoid tumors after resection: implications for postoperative surveillance. Ann Thorac Surg. 2013;96(4):1156-1162.
27. Beasley MB, Thunnissen FB, Brambilla E, et al. Pulmonary atypical carcinoid: predictors of survival in 106 cases. Human Pathol. 2000;31(10):1255-1265.
1. Hauso O, Gustafsson BI, Kidd M, et al. Neuroendocrine tumor epidemiology: contrasting Norway and North America. Cancer. 2008;113(10):2655-2664.
2. Fink G, Krelbaum T, Yellin A, et al. Pulmonary carcinoid: presentation, diagnosis, and outcome in 142 cases in Israel and review of 640 cases from the literature. Chest. 2001;119(6):1647-1651.
3. Limper AH, Carpenter PC, Scheithauer B, Staats BA. The Cushing syndrome induced by bronchial carcinoid tumors. Ann Intern Med. 1992;117(3):209-214.
4. Meisinger QC, Klein JS, Butnor KJ, Gentchos G, Leavitt BJ. CT features of peripheral pulmonary carcinoid tumors. AJR Am J Roentgenol. 2011;197(5):1073-1080.
5. Guckel C, Schnabel K, Deimling M, Steinbrich W. Solitary pulmonary nodules: MR evaluation of enhancement patterns with contrast-enhanced dynamic snapshot gradient-echo imaging. Radiology. 1996;200(3):681-686.
6. Jindal T, Kumar A, Venkitaraman B, et al. Evaluation of the role of [18F]FDG-PET/CT and [68Ga]DOTATOC-PET/CT in differentiating typical and atypical pulmonary carcinoids. Cancer Imaging. 2011;11:70-75.
7. Caplin ME, Baudin E, Ferolla P, et al. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604-1620.
8. Travis WD. Pathology and diagnosis of neuroendocrine tumors: lung neuroendocrine. Thorac Surg Clin. 2014;24(3):257-266.
9. Warren WH, Memoli VA, Gould VE. Immunohistochemical and ultrastructural analysis of bronchopulmonary neuroendocrine neoplasms. II. Well-differentiated neuroendocrine carcinomas. Ultrastruct Pathol. 1984;7(2-3):185-199.
10. Goldstraw P, Chansky K, Crowley J, et al. The IASLC Lung Cancer Staging Project: Proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J Thorac Oncol. 2016;11(1):39-51.
11. McCaughan BC, Martini N, Bains MS. Bronchial carcinoids. Review of 124 cases. J Thorac Cardiovasc Surg. 1985;89(1):8-17.
12. Hurt R, Bates M. Carcinoid tumours of the bronchus: a 33-year experience. Thorax. 1984;39(8):617-623.
13. Ettinger DS, Wood DE, Akerley W, et al. Non-small cell lung cancer, version 1.2015. J Natl Compr Cancer Netw. 2014;12(12):1738-1761.
14. Ferguson MK, Landreneau RJ, Hazelrigg SR, et al. Long-term outcome after resection for bronchial carcinoid tumors. Eur J Cardiothorac Surg. 2000;18(2):156-161.
15. Lucchi M, Melfi F, Ribechini A, et al. Sleeve and wedge parenchyma-sparing bronchial resections in low-grade neoplasms of the bronchial airway. J Thorac Cardiovasc Surg. 2007;134(2):373-377.
16. Yendamuri S, Gold D, Jayaprakash V, Dexter E, Nwogu C, Demmy T. Is sublobar resection sufficient for carcinoid tumors? Ann Thorac Surg. 2011;92(5):1774-1778; discussion 8-9.
17. Fox M, Van Berkel V, Bousamra M II, Sloan S, Martin RC II. Surgical management of pulmonary carcinoid tumors: sublobar resection versus lobectomy. Am J Surg. 2013;205(2):200-208.
18. Cardillo G, Sera F, Di Martino M, et al. Bronchial carcinoid tumors: nodal status and long-term survival after resection. Ann Thorac Surg. 2004;77(5):1781-1785.
19. Oberg K, Hellman P, Ferolla P, Papotti M; ESMO Guidelines Working Group. Neuroendocrine bronchial and thymic tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(suppl 7:vii120-3).
20. Filosso PL, Ferolla P, Guerrera F, et al. Multidisciplinary management of advanced lung neuroendocrine tumors. J Thorac Dis. 2015;7(Suppl 2):S163-171.
21. Kulke MH, Shah MH, Benson AB III, et al. Neuroendocrine tumors, version 1.2015. J Natl Compr Cancer Netw. 2015;13(1):78-108.
22. Yao JC, Fazio N, Singh S, et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): a randomised, placebo-controlled, phase 3 study. Lancet. 2016;387(10022):968-977.
23. Ramirez RA, Beyer DT, Chauhan A, Boudreaux JP, Wang YZ, Woltering EA. The role of capecitabine/temozolomide in metastatic neuroendocrine tumors. Oncologist. 2016;21(6):671-675.
24. Bajetta E, Rimassa L, Carnaghi C, et al. 5-Fluorouracil, dacarbazine, and epirubicin in the treatment of patients with neuroendocrine tumors. Cancer. 1998;83(2):372-378.
25. Masi G, Fornaro L, Cupini S, et al. Refractory neuroendocrine tumor-response to liposomal doxorubicin and capecitabine. Nat Rev Clin Oncol. 2009;6(11):670-674.
26. Lou F, Sarkaria I, Pietanza C, et al. Recurrence of pulmonary carcinoid tumors after resection: implications for postoperative surveillance. Ann Thorac Surg. 2013;96(4):1156-1162.
27. Beasley MB, Thunnissen FB, Brambilla E, et al. Pulmonary atypical carcinoid: predictors of survival in 106 cases. Human Pathol. 2000;31(10):1255-1265.