User login
Flu susceptibility driven by birth year
Differences in susceptibility to an influenza A virus (IAV) strain may be traceable to the first lifetime influenza infection, according to a new statistical model, which could have implications for epidemiology and future flu vaccines.
In the Nov. 11 issue of Science, researchers described infection models of the H5N1 and H7N9 strains of influenza A. The former occurs more commonly in younger people, and the latter in older individuals, but the reasons for those associations have puzzled scientists.
The researchers, led by James Lloyd-Smith, PhD, of the department of ecology and evolutionary biology at the University of California, Los Angeles, looked at susceptibility to IAV strains by birth year, and found that this was the best predictor of vulnerability. For example, an analysis of H5N1 cases in Egypt, where had many H5N1 cases spread over the past decade, showed that individuals born in the same year had the same average risk of severe H5N1 infection, even after they had aged by 10 years. That suggests that it is the birth year, not advancing age, which influences susceptibility (Science. 2016 Nov 11;354[6313]:721-5. doi:10.1126/science.aag1322).
The researchers suggest that the immune system “imprints” on the hemagglutinin (HA) subtype during an individual’s first infection, which confers protection against severe disease caused by other, related viruses, though it may not reduce infection rates overall.
The year 1968 may have marked an important inflection point. That year marked a shift in the identify of circulating viruses, from group 1 HA (which includes H5N1) to group 2 HA (which includes H7N9). Individuals born before 1968 were likely first infected with a group 1 virus, while those born later were most likely initially exposed to a group 2 virus. If the imprint theory is correct, younger people would have imprinted on group 2 viruses similar to H7N9, which would explain their greater vulnerability to group 1 viruses like H5N1.
“Imprinting was the dominant explanatory factor for observed incidence and mortality patterns for both H5N1 and H7N9. It was the only tested factor included in all plausible models for both viruses,” the researchers wrote.
According to the model, imprinting explains 75% of protection against severe infection and 80% of the protection against mortality for H5N1 and H7N9.
That information adds a previously unrecognized layer to influenza epidemiology, which should be accounted for in public health measures. “The methods shown here can provide rolling estimates of which age groups would be at highest risk for severe disease should particular novel HA subtypes emerge,” the researchers wrote.
The researchers received funding from the National Institutes of Health, the National Science Foundation, the David and Lucile Packard Foundation, and the Department of Homeland Security. They reported having no financial disclosures.
A growing body of epidemiological evidence points to the prolonged effects of cross-immunity, including competition between strains during seasonal and pandemic outbreaks, reduced risk of pandemic infection in those with previous seasonal exposure, and – as reported by Gostic et al. – lifelong protection against viruses of different subtypes but in the same hemagglutinin (HA) homology group. Basic science efforts are now needed to fully validate the HA imprinting hypothesis. More broadly, further experimental and theoretical work should map the relationship between early childhood exposure to influenza and immune protection and the implications of lifelong immunity for vaccination strategies and pandemic risk.
Cécile Viboud, PhD, is the acting director of the division of international epidemiology and population studies at the Fogarty International Center at the National Institutes of Health. Suzanne L. Epstein, PhD, is the associate director for research at the office of tissues and advanced therapies at the Food and Drug Administration Center for Biologics Evaluation and Research. They had no relevant financial disclosures and made these remarks in an editorial that accompanied the published study (Science. 2016 Nov 11;354[6313]:706-7. doi:10.1126/science.aak9816).
A growing body of epidemiological evidence points to the prolonged effects of cross-immunity, including competition between strains during seasonal and pandemic outbreaks, reduced risk of pandemic infection in those with previous seasonal exposure, and – as reported by Gostic et al. – lifelong protection against viruses of different subtypes but in the same hemagglutinin (HA) homology group. Basic science efforts are now needed to fully validate the HA imprinting hypothesis. More broadly, further experimental and theoretical work should map the relationship between early childhood exposure to influenza and immune protection and the implications of lifelong immunity for vaccination strategies and pandemic risk.
Cécile Viboud, PhD, is the acting director of the division of international epidemiology and population studies at the Fogarty International Center at the National Institutes of Health. Suzanne L. Epstein, PhD, is the associate director for research at the office of tissues and advanced therapies at the Food and Drug Administration Center for Biologics Evaluation and Research. They had no relevant financial disclosures and made these remarks in an editorial that accompanied the published study (Science. 2016 Nov 11;354[6313]:706-7. doi:10.1126/science.aak9816).
A growing body of epidemiological evidence points to the prolonged effects of cross-immunity, including competition between strains during seasonal and pandemic outbreaks, reduced risk of pandemic infection in those with previous seasonal exposure, and – as reported by Gostic et al. – lifelong protection against viruses of different subtypes but in the same hemagglutinin (HA) homology group. Basic science efforts are now needed to fully validate the HA imprinting hypothesis. More broadly, further experimental and theoretical work should map the relationship between early childhood exposure to influenza and immune protection and the implications of lifelong immunity for vaccination strategies and pandemic risk.
Cécile Viboud, PhD, is the acting director of the division of international epidemiology and population studies at the Fogarty International Center at the National Institutes of Health. Suzanne L. Epstein, PhD, is the associate director for research at the office of tissues and advanced therapies at the Food and Drug Administration Center for Biologics Evaluation and Research. They had no relevant financial disclosures and made these remarks in an editorial that accompanied the published study (Science. 2016 Nov 11;354[6313]:706-7. doi:10.1126/science.aak9816).
Differences in susceptibility to an influenza A virus (IAV) strain may be traceable to the first lifetime influenza infection, according to a new statistical model, which could have implications for epidemiology and future flu vaccines.
In the Nov. 11 issue of Science, researchers described infection models of the H5N1 and H7N9 strains of influenza A. The former occurs more commonly in younger people, and the latter in older individuals, but the reasons for those associations have puzzled scientists.
The researchers, led by James Lloyd-Smith, PhD, of the department of ecology and evolutionary biology at the University of California, Los Angeles, looked at susceptibility to IAV strains by birth year, and found that this was the best predictor of vulnerability. For example, an analysis of H5N1 cases in Egypt, where had many H5N1 cases spread over the past decade, showed that individuals born in the same year had the same average risk of severe H5N1 infection, even after they had aged by 10 years. That suggests that it is the birth year, not advancing age, which influences susceptibility (Science. 2016 Nov 11;354[6313]:721-5. doi:10.1126/science.aag1322).
The researchers suggest that the immune system “imprints” on the hemagglutinin (HA) subtype during an individual’s first infection, which confers protection against severe disease caused by other, related viruses, though it may not reduce infection rates overall.
The year 1968 may have marked an important inflection point. That year marked a shift in the identify of circulating viruses, from group 1 HA (which includes H5N1) to group 2 HA (which includes H7N9). Individuals born before 1968 were likely first infected with a group 1 virus, while those born later were most likely initially exposed to a group 2 virus. If the imprint theory is correct, younger people would have imprinted on group 2 viruses similar to H7N9, which would explain their greater vulnerability to group 1 viruses like H5N1.
“Imprinting was the dominant explanatory factor for observed incidence and mortality patterns for both H5N1 and H7N9. It was the only tested factor included in all plausible models for both viruses,” the researchers wrote.
According to the model, imprinting explains 75% of protection against severe infection and 80% of the protection against mortality for H5N1 and H7N9.
That information adds a previously unrecognized layer to influenza epidemiology, which should be accounted for in public health measures. “The methods shown here can provide rolling estimates of which age groups would be at highest risk for severe disease should particular novel HA subtypes emerge,” the researchers wrote.
The researchers received funding from the National Institutes of Health, the National Science Foundation, the David and Lucile Packard Foundation, and the Department of Homeland Security. They reported having no financial disclosures.
Differences in susceptibility to an influenza A virus (IAV) strain may be traceable to the first lifetime influenza infection, according to a new statistical model, which could have implications for epidemiology and future flu vaccines.
In the Nov. 11 issue of Science, researchers described infection models of the H5N1 and H7N9 strains of influenza A. The former occurs more commonly in younger people, and the latter in older individuals, but the reasons for those associations have puzzled scientists.
The researchers, led by James Lloyd-Smith, PhD, of the department of ecology and evolutionary biology at the University of California, Los Angeles, looked at susceptibility to IAV strains by birth year, and found that this was the best predictor of vulnerability. For example, an analysis of H5N1 cases in Egypt, where had many H5N1 cases spread over the past decade, showed that individuals born in the same year had the same average risk of severe H5N1 infection, even after they had aged by 10 years. That suggests that it is the birth year, not advancing age, which influences susceptibility (Science. 2016 Nov 11;354[6313]:721-5. doi:10.1126/science.aag1322).
The researchers suggest that the immune system “imprints” on the hemagglutinin (HA) subtype during an individual’s first infection, which confers protection against severe disease caused by other, related viruses, though it may not reduce infection rates overall.
The year 1968 may have marked an important inflection point. That year marked a shift in the identify of circulating viruses, from group 1 HA (which includes H5N1) to group 2 HA (which includes H7N9). Individuals born before 1968 were likely first infected with a group 1 virus, while those born later were most likely initially exposed to a group 2 virus. If the imprint theory is correct, younger people would have imprinted on group 2 viruses similar to H7N9, which would explain their greater vulnerability to group 1 viruses like H5N1.
“Imprinting was the dominant explanatory factor for observed incidence and mortality patterns for both H5N1 and H7N9. It was the only tested factor included in all plausible models for both viruses,” the researchers wrote.
According to the model, imprinting explains 75% of protection against severe infection and 80% of the protection against mortality for H5N1 and H7N9.
That information adds a previously unrecognized layer to influenza epidemiology, which should be accounted for in public health measures. “The methods shown here can provide rolling estimates of which age groups would be at highest risk for severe disease should particular novel HA subtypes emerge,” the researchers wrote.
The researchers received funding from the National Institutes of Health, the National Science Foundation, the David and Lucile Packard Foundation, and the Department of Homeland Security. They reported having no financial disclosures.
Key clinical point:
Major finding: Early exposure to virus subtype explains 75% of protection against severe disease in later life.
Data source: Statistical model of retrospective data.
Disclosures: The researchers received funding from the National Institutes of Health, the National Science Foundation, the David and Lucile Packard Foundation, and the Department of Homeland Security. They reported having no financial disclosures.
Cooling, occlusion, and antihistamines are among the options that optimize ALA-PDT results
Treatment of a broad area, occluding extremities, and the use of antihistamines are among the measures that can help optimize the results of treating actinic keratoses (AKs) with topical photodynamic therapy (PDT) using aminolevulinic acid (ALA), according to Dr. Brian Berman.
For many patients with AKs, ALA-PDT can be an effective and well-tolerated option, Dr. Berman said in a presentation at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
ALA-PDT is a two-step process, Dr. Berman explained. The first step involves the application of ALA with presumed selective cellular uptake, followed by conversion to protoporphyrin IX (PpIX). Next, the light activation of PpIX causes cell death via high-energy oxygen molecules.
Dr. Berman’s recommendations for optimizing ALA-PDT to treat AKs include a shorter ALA incubation time; treatment of a broad area, not just the baseline visible AKs; occlusion of ALA for AKs on the arms and legs; increased skin temperature during ALA incubation; and moderate cooling during light exposure. To reduce pain, he recommended a very short ALA incubation time with a longer time of light exposure.
He referred to a 2004 study of 18 patients, which found that AK reductions were not significantly different at 1 month post treatment with ALA incubation times of 1, 2, or 3 hours (Arch Dermatol. 2004 Jan;140[1]:33-40). As for occlusion, a 2012 study found that AK clearance was significantly greater for extremities that were occluded during incubation in patients undergoing blue light ALA-PDT, compared with areas that were not occluded (J Drugs Dermatol. 2012 Dec;11[12]:1483-9).
Skin cooling can be useful in reducing patients’ pain, said Dr. Berman of the department of dermatology and cutaneous surgery, University of Miami. Data from a retrospective study showed that cooling pain relief, with an air-cooling device during treatment, resulted in lower PpIX photobleaching in AK lesions, compared with no cooling (J Photochem Photobiol B. 2011 Apr 4;103[1]:1-7). But cooling was associated with decreased efficacy of the PDT treatment in terms of complete AK response (68% for the cooling device group vs. 82% for controls without cooling), he said.
Increasing skin temperature has been shown to reduce AK lesions significantly, compared with no heat, Dr. Berman pointed out. “PpIX synthesis is temperature dependent,” he said. The median difference in AK lesion counts was significantly greater on a heated extremity side than a control side in an unpublished study, he noted.
Finally, when it comes to facial AKs, less may be more in terms of treatment time. In a split face study, a “painless PDT” protocol of 15 minutes of ALA incubation with 1 hour of blue light yielded a 52% reduction in AKs at 8 weeks post treatment, vs. a 44% reduction with a standard treatment of 75 minutes of ALA incubation with 16 minutes, 45 seconds of blue light, Dr. Berman said.
Dr. Berman disclosed relationships with multiple companies including Ferndale, LEO, Halscion, Sensus, Exeltis, Dermira, Celumigen, Sun, DUSA, Biofrontera, and Berg. He holds stock in Halscion, Dermira, Celumigen, and Berg.
SDEF and this news organization are owned by the same parent company.
Treatment of a broad area, occluding extremities, and the use of antihistamines are among the measures that can help optimize the results of treating actinic keratoses (AKs) with topical photodynamic therapy (PDT) using aminolevulinic acid (ALA), according to Dr. Brian Berman.
For many patients with AKs, ALA-PDT can be an effective and well-tolerated option, Dr. Berman said in a presentation at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
ALA-PDT is a two-step process, Dr. Berman explained. The first step involves the application of ALA with presumed selective cellular uptake, followed by conversion to protoporphyrin IX (PpIX). Next, the light activation of PpIX causes cell death via high-energy oxygen molecules.
Dr. Berman’s recommendations for optimizing ALA-PDT to treat AKs include a shorter ALA incubation time; treatment of a broad area, not just the baseline visible AKs; occlusion of ALA for AKs on the arms and legs; increased skin temperature during ALA incubation; and moderate cooling during light exposure. To reduce pain, he recommended a very short ALA incubation time with a longer time of light exposure.
He referred to a 2004 study of 18 patients, which found that AK reductions were not significantly different at 1 month post treatment with ALA incubation times of 1, 2, or 3 hours (Arch Dermatol. 2004 Jan;140[1]:33-40). As for occlusion, a 2012 study found that AK clearance was significantly greater for extremities that were occluded during incubation in patients undergoing blue light ALA-PDT, compared with areas that were not occluded (J Drugs Dermatol. 2012 Dec;11[12]:1483-9).
Skin cooling can be useful in reducing patients’ pain, said Dr. Berman of the department of dermatology and cutaneous surgery, University of Miami. Data from a retrospective study showed that cooling pain relief, with an air-cooling device during treatment, resulted in lower PpIX photobleaching in AK lesions, compared with no cooling (J Photochem Photobiol B. 2011 Apr 4;103[1]:1-7). But cooling was associated with decreased efficacy of the PDT treatment in terms of complete AK response (68% for the cooling device group vs. 82% for controls without cooling), he said.
Increasing skin temperature has been shown to reduce AK lesions significantly, compared with no heat, Dr. Berman pointed out. “PpIX synthesis is temperature dependent,” he said. The median difference in AK lesion counts was significantly greater on a heated extremity side than a control side in an unpublished study, he noted.
Finally, when it comes to facial AKs, less may be more in terms of treatment time. In a split face study, a “painless PDT” protocol of 15 minutes of ALA incubation with 1 hour of blue light yielded a 52% reduction in AKs at 8 weeks post treatment, vs. a 44% reduction with a standard treatment of 75 minutes of ALA incubation with 16 minutes, 45 seconds of blue light, Dr. Berman said.
Dr. Berman disclosed relationships with multiple companies including Ferndale, LEO, Halscion, Sensus, Exeltis, Dermira, Celumigen, Sun, DUSA, Biofrontera, and Berg. He holds stock in Halscion, Dermira, Celumigen, and Berg.
SDEF and this news organization are owned by the same parent company.
Treatment of a broad area, occluding extremities, and the use of antihistamines are among the measures that can help optimize the results of treating actinic keratoses (AKs) with topical photodynamic therapy (PDT) using aminolevulinic acid (ALA), according to Dr. Brian Berman.
For many patients with AKs, ALA-PDT can be an effective and well-tolerated option, Dr. Berman said in a presentation at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
ALA-PDT is a two-step process, Dr. Berman explained. The first step involves the application of ALA with presumed selective cellular uptake, followed by conversion to protoporphyrin IX (PpIX). Next, the light activation of PpIX causes cell death via high-energy oxygen molecules.
Dr. Berman’s recommendations for optimizing ALA-PDT to treat AKs include a shorter ALA incubation time; treatment of a broad area, not just the baseline visible AKs; occlusion of ALA for AKs on the arms and legs; increased skin temperature during ALA incubation; and moderate cooling during light exposure. To reduce pain, he recommended a very short ALA incubation time with a longer time of light exposure.
He referred to a 2004 study of 18 patients, which found that AK reductions were not significantly different at 1 month post treatment with ALA incubation times of 1, 2, or 3 hours (Arch Dermatol. 2004 Jan;140[1]:33-40). As for occlusion, a 2012 study found that AK clearance was significantly greater for extremities that were occluded during incubation in patients undergoing blue light ALA-PDT, compared with areas that were not occluded (J Drugs Dermatol. 2012 Dec;11[12]:1483-9).
Skin cooling can be useful in reducing patients’ pain, said Dr. Berman of the department of dermatology and cutaneous surgery, University of Miami. Data from a retrospective study showed that cooling pain relief, with an air-cooling device during treatment, resulted in lower PpIX photobleaching in AK lesions, compared with no cooling (J Photochem Photobiol B. 2011 Apr 4;103[1]:1-7). But cooling was associated with decreased efficacy of the PDT treatment in terms of complete AK response (68% for the cooling device group vs. 82% for controls without cooling), he said.
Increasing skin temperature has been shown to reduce AK lesions significantly, compared with no heat, Dr. Berman pointed out. “PpIX synthesis is temperature dependent,” he said. The median difference in AK lesion counts was significantly greater on a heated extremity side than a control side in an unpublished study, he noted.
Finally, when it comes to facial AKs, less may be more in terms of treatment time. In a split face study, a “painless PDT” protocol of 15 minutes of ALA incubation with 1 hour of blue light yielded a 52% reduction in AKs at 8 weeks post treatment, vs. a 44% reduction with a standard treatment of 75 minutes of ALA incubation with 16 minutes, 45 seconds of blue light, Dr. Berman said.
Dr. Berman disclosed relationships with multiple companies including Ferndale, LEO, Halscion, Sensus, Exeltis, Dermira, Celumigen, Sun, DUSA, Biofrontera, and Berg. He holds stock in Halscion, Dermira, Celumigen, and Berg.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
OA progresses equally with new focal partial- or full-thickness cartilage damage
A small, partial-thickness focal cartilage defect in the tibiofemoral joint compartment has the same impact on osteoarthritis disease progression – defined as new cartilage damage – as does a full-thickness lesion, according to an analysis of data from the Multicenter Osteoarthritis Study.
Ali Guermazi, MD, PhD, of Boston University, and his associates used MRI to show that both full- and partial-thickness small focal defects (less than 1 cm) in the tibiofemoral compartment contributed significantly to the risk of incident cartilage damage 30 months later in other tibiofemoral compartment subregions (adjusted odds ratio, 1.62; 95% confidence interval, 1.06-2.47, for partial-thickness defects and aOR, 1.92; 95% CI, 1.00-3.66, for full-thickness defects) when compared with subregions with no baseline cartilage damage, defined as a Kellgren-Lawrence grade of 0-1.
The results indicate that “partial-thickness and full-thickness defects are similarly relevant in regard to cartilage damage development in knee osteoarthritis,” the investigators wrote.
They studied 374 compartments (359 knees), of which 140 knees (39%) had radiographic osteoarthritis defined as a Kellgren-Lawrence grade of 2 or higher).
“It is potentially important to detect focal cartilage defects early as there are various options for repair of focal cartilage defects ... [and it could be possible to use MRI] to screen persons with early-stage osteoarthritis or those at high risk of osteoarthritis and initiate treatment for focal cartilage defects,” they wrote.
Read the full study in Arthritis & Rheumatology (2016 Oct 27. doi: 10.1002/art.39970).
A small, partial-thickness focal cartilage defect in the tibiofemoral joint compartment has the same impact on osteoarthritis disease progression – defined as new cartilage damage – as does a full-thickness lesion, according to an analysis of data from the Multicenter Osteoarthritis Study.
Ali Guermazi, MD, PhD, of Boston University, and his associates used MRI to show that both full- and partial-thickness small focal defects (less than 1 cm) in the tibiofemoral compartment contributed significantly to the risk of incident cartilage damage 30 months later in other tibiofemoral compartment subregions (adjusted odds ratio, 1.62; 95% confidence interval, 1.06-2.47, for partial-thickness defects and aOR, 1.92; 95% CI, 1.00-3.66, for full-thickness defects) when compared with subregions with no baseline cartilage damage, defined as a Kellgren-Lawrence grade of 0-1.
The results indicate that “partial-thickness and full-thickness defects are similarly relevant in regard to cartilage damage development in knee osteoarthritis,” the investigators wrote.
They studied 374 compartments (359 knees), of which 140 knees (39%) had radiographic osteoarthritis defined as a Kellgren-Lawrence grade of 2 or higher).
“It is potentially important to detect focal cartilage defects early as there are various options for repair of focal cartilage defects ... [and it could be possible to use MRI] to screen persons with early-stage osteoarthritis or those at high risk of osteoarthritis and initiate treatment for focal cartilage defects,” they wrote.
Read the full study in Arthritis & Rheumatology (2016 Oct 27. doi: 10.1002/art.39970).
A small, partial-thickness focal cartilage defect in the tibiofemoral joint compartment has the same impact on osteoarthritis disease progression – defined as new cartilage damage – as does a full-thickness lesion, according to an analysis of data from the Multicenter Osteoarthritis Study.
Ali Guermazi, MD, PhD, of Boston University, and his associates used MRI to show that both full- and partial-thickness small focal defects (less than 1 cm) in the tibiofemoral compartment contributed significantly to the risk of incident cartilage damage 30 months later in other tibiofemoral compartment subregions (adjusted odds ratio, 1.62; 95% confidence interval, 1.06-2.47, for partial-thickness defects and aOR, 1.92; 95% CI, 1.00-3.66, for full-thickness defects) when compared with subregions with no baseline cartilage damage, defined as a Kellgren-Lawrence grade of 0-1.
The results indicate that “partial-thickness and full-thickness defects are similarly relevant in regard to cartilage damage development in knee osteoarthritis,” the investigators wrote.
They studied 374 compartments (359 knees), of which 140 knees (39%) had radiographic osteoarthritis defined as a Kellgren-Lawrence grade of 2 or higher).
“It is potentially important to detect focal cartilage defects early as there are various options for repair of focal cartilage defects ... [and it could be possible to use MRI] to screen persons with early-stage osteoarthritis or those at high risk of osteoarthritis and initiate treatment for focal cartilage defects,” they wrote.
Read the full study in Arthritis & Rheumatology (2016 Oct 27. doi: 10.1002/art.39970).
FROM ARTHRITIS & RHEUMATOLOGY
Dystrophic epidermolysis bullosa: 104 mutations found in single gene
according to a report published online in the Journal of Investigative Dermatology.
Identifying which specific mutations in the COL7A1 gene a given patient is carrying should allow clinicians to render a more accurate prognosis and may even dictate management strategies to counteract manifestations of the disease. Dystrophic epidermolysis bullosa (EB) can have an extremely variable progression that depends, in part, on the underlying molecular defects in many different genes expressed in the dermal-epidermal junction, according to study investigators Hassan Vahidnezhad, MD, of the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia, and his associates.
The study cohort included many consanguineous marriages, which are customary in Iran as in many parts of the Middle East, North Africa, and sub-Saharan Africa. In addition, many migrant communities from these regions now reside in Western countries and continue to practice these marriage customs, most commonly marriages between first cousins. “It is estimated that globally at least 20% of the human population live in communities with a preference for consanguineous marriage,” the researchers wrote (J Investig Dermatol. 2016. doi: 10.1016/j.jid.2016.10.023).
Dystrophic epidermolysis bullosa is characterized by blistering of the sublamina densa that leads to erosions; chronic ulcers; and extensive scarring, especially at sites of trauma on the hands and feet. Blistering can also develop in corneal and gastrointestinal epithelium, which adversely affects vision and feeding, and patients often develop aggressive squamous cell carcinoma with its attendant premature mortality.
Dystrophic EB has long been associated with mutations in COL7A1 as well as other genes. COL7A1 encodes type VII collagen, “a major protein component of the anchoring fibrils, which play a critical role in securing the attachment of the dermal-epidermal basement membrane to the underlying dermis.” Many patients with dystrophic EB show marked reductions in, or a total absence of, anchoring fibrils.
The investigators explored COL7A1 mutations in a multiethnic database comprising 238 patients in 152 extended families from different parts of the country. (The approximately 80 million people in Iran include several ethnic groups with distinct ancestries, languages, cultures, and geographic areas of residence.) A total of 139 of these families were consanguineous.
A total of 104 distinct COL7A1 mutations were detected in 149 of the 152 families, for a detection rate of 98%. Many mutations had been reported previously, but 56 had never been reported before. Approximately 90% of the families in the study cohort showed homozygous recessive mutations, which appears to reflect the consanguinity of this population, Dr. Vahidnezhad and his associates said.
They concluded that “the overwhelming majority” of patients with dystrophic EB carry COL7A1 genes harboring mutations.
This study was supported by DEBRA International and the Sidney Kimmel Cancer Center at Thomas Jefferson University. Dr. Vahidnezhad and his associates reported having no relevant financial disclosures.
according to a report published online in the Journal of Investigative Dermatology.
Identifying which specific mutations in the COL7A1 gene a given patient is carrying should allow clinicians to render a more accurate prognosis and may even dictate management strategies to counteract manifestations of the disease. Dystrophic epidermolysis bullosa (EB) can have an extremely variable progression that depends, in part, on the underlying molecular defects in many different genes expressed in the dermal-epidermal junction, according to study investigators Hassan Vahidnezhad, MD, of the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia, and his associates.
The study cohort included many consanguineous marriages, which are customary in Iran as in many parts of the Middle East, North Africa, and sub-Saharan Africa. In addition, many migrant communities from these regions now reside in Western countries and continue to practice these marriage customs, most commonly marriages between first cousins. “It is estimated that globally at least 20% of the human population live in communities with a preference for consanguineous marriage,” the researchers wrote (J Investig Dermatol. 2016. doi: 10.1016/j.jid.2016.10.023).
Dystrophic epidermolysis bullosa is characterized by blistering of the sublamina densa that leads to erosions; chronic ulcers; and extensive scarring, especially at sites of trauma on the hands and feet. Blistering can also develop in corneal and gastrointestinal epithelium, which adversely affects vision and feeding, and patients often develop aggressive squamous cell carcinoma with its attendant premature mortality.
Dystrophic EB has long been associated with mutations in COL7A1 as well as other genes. COL7A1 encodes type VII collagen, “a major protein component of the anchoring fibrils, which play a critical role in securing the attachment of the dermal-epidermal basement membrane to the underlying dermis.” Many patients with dystrophic EB show marked reductions in, or a total absence of, anchoring fibrils.
The investigators explored COL7A1 mutations in a multiethnic database comprising 238 patients in 152 extended families from different parts of the country. (The approximately 80 million people in Iran include several ethnic groups with distinct ancestries, languages, cultures, and geographic areas of residence.) A total of 139 of these families were consanguineous.
A total of 104 distinct COL7A1 mutations were detected in 149 of the 152 families, for a detection rate of 98%. Many mutations had been reported previously, but 56 had never been reported before. Approximately 90% of the families in the study cohort showed homozygous recessive mutations, which appears to reflect the consanguinity of this population, Dr. Vahidnezhad and his associates said.
They concluded that “the overwhelming majority” of patients with dystrophic EB carry COL7A1 genes harboring mutations.
This study was supported by DEBRA International and the Sidney Kimmel Cancer Center at Thomas Jefferson University. Dr. Vahidnezhad and his associates reported having no relevant financial disclosures.
according to a report published online in the Journal of Investigative Dermatology.
Identifying which specific mutations in the COL7A1 gene a given patient is carrying should allow clinicians to render a more accurate prognosis and may even dictate management strategies to counteract manifestations of the disease. Dystrophic epidermolysis bullosa (EB) can have an extremely variable progression that depends, in part, on the underlying molecular defects in many different genes expressed in the dermal-epidermal junction, according to study investigators Hassan Vahidnezhad, MD, of the department of dermatology and cutaneous biology, Thomas Jefferson University, Philadelphia, and his associates.
The study cohort included many consanguineous marriages, which are customary in Iran as in many parts of the Middle East, North Africa, and sub-Saharan Africa. In addition, many migrant communities from these regions now reside in Western countries and continue to practice these marriage customs, most commonly marriages between first cousins. “It is estimated that globally at least 20% of the human population live in communities with a preference for consanguineous marriage,” the researchers wrote (J Investig Dermatol. 2016. doi: 10.1016/j.jid.2016.10.023).
Dystrophic epidermolysis bullosa is characterized by blistering of the sublamina densa that leads to erosions; chronic ulcers; and extensive scarring, especially at sites of trauma on the hands and feet. Blistering can also develop in corneal and gastrointestinal epithelium, which adversely affects vision and feeding, and patients often develop aggressive squamous cell carcinoma with its attendant premature mortality.
Dystrophic EB has long been associated with mutations in COL7A1 as well as other genes. COL7A1 encodes type VII collagen, “a major protein component of the anchoring fibrils, which play a critical role in securing the attachment of the dermal-epidermal basement membrane to the underlying dermis.” Many patients with dystrophic EB show marked reductions in, or a total absence of, anchoring fibrils.
The investigators explored COL7A1 mutations in a multiethnic database comprising 238 patients in 152 extended families from different parts of the country. (The approximately 80 million people in Iran include several ethnic groups with distinct ancestries, languages, cultures, and geographic areas of residence.) A total of 139 of these families were consanguineous.
A total of 104 distinct COL7A1 mutations were detected in 149 of the 152 families, for a detection rate of 98%. Many mutations had been reported previously, but 56 had never been reported before. Approximately 90% of the families in the study cohort showed homozygous recessive mutations, which appears to reflect the consanguinity of this population, Dr. Vahidnezhad and his associates said.
They concluded that “the overwhelming majority” of patients with dystrophic EB carry COL7A1 genes harboring mutations.
This study was supported by DEBRA International and the Sidney Kimmel Cancer Center at Thomas Jefferson University. Dr. Vahidnezhad and his associates reported having no relevant financial disclosures.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Key clinical point: 104 distinct mutations in a single gene, COL7A1, were discovered in a multiethnic cohort of 152 Iranian families with dystrophic epidermolysis bullosa.
Major finding: 104 distinct COL7A1 mutations were detected in 149 of the 152 families in the database, for a detection rate of 98%.
Data source: Analyses of mutations in the COL741 gene in 152 extended Iranian families with a high frequency of dystrophic epidermolysis bullosa.
Disclosures: This study was supported by DEBRA International and the Sidney Kimmel Cancer Center at Thomas Jefferson University. Dr. Vahidnezhad and his associates reported having no relevant financial disclosures.
The voice of the patient in a doctor-centric world
The American health care system has been plagued by poor medical outcomes and an inefficient cost structure for many years.1,2 The primary loser in this game is the patient, the player who bears the brunt of health care expenses, either directly or through taxes, and whose voice has been largely ignored until recently. As with many other service industries, health care delivery represents a highly complex process that is difficult to define or measure. Innovative procedures and state-of-the-art medical therapies have provided effective options that extend or improve life, but at increasing cost.
Further complicating the existing cost-vs.-benefit debate is a relatively new polemic regarding the degree to which patient satisfaction should be a measure of health care quality. How satisfied will a patient be with his or her health care experience? This depends, in large part, on the medical outcome.3
Many public and private organizations use patient satisfaction surveys to measure the performance of health care delivery systems. In many cases, patient satisfaction data are being used to determine insurance payouts, physician compensation, and institutional rankings.5 It may seem logical to adopt pay-for-performance strategies based on patient satisfaction surveys, but there is a fundamental flaw: Survey data are not always accurate.
It is not entirely clear what constitutes a positive or negative health care experience. Much of this depends on the expectations of the consumer.6 Those with low expectations may be delighted with mediocre performance. Those with inflated expectations may be disappointed even when provided excellent customer service.
Surveys are not durable. That is, when performed under distinct environmental conditions or at different times, surveys may not produce the same results when repeated by the same respondent.7 Surveys are easily manipulated by simple changes in wording or punctuation. Some specific encounters may be rated as “unsatisfactory” because of external factors, circumstances beyond the control of the health care provider.
Many health care providers feel that surveys are poor indicators of individual performance. Some critics highlight a paucity of data. A limited number of returned surveys, relative to the total number of encounters, may yield results that are not statistically significant. Increasing the amount of data decreases the risk that a sample set taken from the studied population is the result of sampling error alone. Nonetheless, sampling error is never completely eliminated, and it is not entirely clear to what degree statistical significance should be used to substantiate satisfaction, a subjective measure.
Surveys often provide data in a very small range, making ranking of facilities or providers difficult. For example, national polling services utilize surveys with thousands of respondents and the margin of error often exceeds plus or minus 3%. Data sets with a smaller number of responses have margins of error that are even greater. Even plus or minus 3% is a sizable deviation when considering that health care survey results often are compared and ranked based on a distribution of scores in a narrow response range. In the author’s experience, a 6% difference in survey scores can represent the difference between a ranking of “excellent” and “poor.”
Many patients are disenfranchised by survey methodology. In the most extreme example, deceased or severely disabled patients are unable to provide feedback. Patients transferred to other facilities and those who are lost to follow-up will be missed also. Many patient surveys may not be successfully retrieved from the homeless, the illiterate, minors, or those without phone or e-mail access. Because surveys are voluntarily submitted, the results may skew opinion toward a select group of outspoken customers who may not be representative of the general population.
The use of patient satisfaction surveys, especially when they are linked to employee compensation, may create a system of survey-based value. This is similar to the problem of defensive medicine, where providers perform medicine in a way that reduces legal risk. Aware that patients will be asked to fill out satisfaction surveys, associates may perform in a way that increases patient satisfaction scores at the expense of patient outcomes or the bottom line. Some institutions may inappropriately “cherry-pick” the easy-to-treat patient and transfer medically complex cases elsewhere.
It is not clear how to best measure the quality of a health care experience. With the broad range of patient encounter types and the inherent complexity of collaboration among providers, it is difficult to determine to what degree satisfaction can be attributed to individual providers or specific environmental factors. Patients do not typically interact with a specific provider, but are treated by a service delivery system, which often encompasses multiple players and multiple physical locations. Moreover, it is not always clear when the patient encounter begins and ends.
Despite the criticism of patient satisfaction survey methodology, the patient must ultimately define the value of the health care service offering. This “voice of the customer” approach is a diversion from the antiquated practitioner-centric model. Traditionally, patient appointment times and locations are decided by the availability and convenience of the provider. Many consumers have compensated for this inefficiency by accessing local emergency departments for nonurgent ambulatory care. Nonetheless,EDs often suffer from long waits and higher costs. Facilities designed for urgent, but nonemergent, care have attempted to address convenience issues but these facilities sacrifice continuity and specialization of care. In a truly patient-centric health care model, patients would be provided the care that they need, when and where they need it.
The patient satisfaction survey remains a primary tool for linking patient-centered value to health care reform. Ranking the results among market competitors can provide an incentive for improvement. Health care professionals are competitive by nature and the extrinsic motivation of quality rankings can be beneficial if well controlled. Employers should use caution when using survey data for performance measurement because survey data are subject to a variety of sources of bias or error. Patient survey data should be used to drive improvement, not to punish. Further research on patient survey methodology is needed to elucidate improved methods of bringing the voice of the patient to the forefront of health care reform.
References
1. N Engl J Med. 2003 Aug 21;349(8):768-75.
2. Centers for Medicare & Medicaid Services. Medicare hospital quality chartbook: Performance report on outcome measures. September 2014.
3. N Engl J Med. 2008 Oct 30;359(18):1921-31.
4. “Better Customer Insight – in Real Time,” by Emma K. Macdonald, Hugh N. Wilson, and Umut Konuş (Harvard Business Review, September 2012).
5. “The Dangers of Linking Pay to Customer Feedback,” by Rob Markey, (Harvard Business Review, Sept. 8, 2011).
6. “Health Care’s Service Fanatics,” by James I. Merlino and Ananth Raman, (Harvard Business Review, May 2013).
7. Trochim, WMK. Research Methods Knowledge Base.
Dr. Davis is a pediatric gastroenterologists at University of Florida Health, Gainesville. He has no financial relationships relevant to this article to disclose.
The American health care system has been plagued by poor medical outcomes and an inefficient cost structure for many years.1,2 The primary loser in this game is the patient, the player who bears the brunt of health care expenses, either directly or through taxes, and whose voice has been largely ignored until recently. As with many other service industries, health care delivery represents a highly complex process that is difficult to define or measure. Innovative procedures and state-of-the-art medical therapies have provided effective options that extend or improve life, but at increasing cost.
Further complicating the existing cost-vs.-benefit debate is a relatively new polemic regarding the degree to which patient satisfaction should be a measure of health care quality. How satisfied will a patient be with his or her health care experience? This depends, in large part, on the medical outcome.3
Many public and private organizations use patient satisfaction surveys to measure the performance of health care delivery systems. In many cases, patient satisfaction data are being used to determine insurance payouts, physician compensation, and institutional rankings.5 It may seem logical to adopt pay-for-performance strategies based on patient satisfaction surveys, but there is a fundamental flaw: Survey data are not always accurate.
It is not entirely clear what constitutes a positive or negative health care experience. Much of this depends on the expectations of the consumer.6 Those with low expectations may be delighted with mediocre performance. Those with inflated expectations may be disappointed even when provided excellent customer service.
Surveys are not durable. That is, when performed under distinct environmental conditions or at different times, surveys may not produce the same results when repeated by the same respondent.7 Surveys are easily manipulated by simple changes in wording or punctuation. Some specific encounters may be rated as “unsatisfactory” because of external factors, circumstances beyond the control of the health care provider.
Many health care providers feel that surveys are poor indicators of individual performance. Some critics highlight a paucity of data. A limited number of returned surveys, relative to the total number of encounters, may yield results that are not statistically significant. Increasing the amount of data decreases the risk that a sample set taken from the studied population is the result of sampling error alone. Nonetheless, sampling error is never completely eliminated, and it is not entirely clear to what degree statistical significance should be used to substantiate satisfaction, a subjective measure.
Surveys often provide data in a very small range, making ranking of facilities or providers difficult. For example, national polling services utilize surveys with thousands of respondents and the margin of error often exceeds plus or minus 3%. Data sets with a smaller number of responses have margins of error that are even greater. Even plus or minus 3% is a sizable deviation when considering that health care survey results often are compared and ranked based on a distribution of scores in a narrow response range. In the author’s experience, a 6% difference in survey scores can represent the difference between a ranking of “excellent” and “poor.”
Many patients are disenfranchised by survey methodology. In the most extreme example, deceased or severely disabled patients are unable to provide feedback. Patients transferred to other facilities and those who are lost to follow-up will be missed also. Many patient surveys may not be successfully retrieved from the homeless, the illiterate, minors, or those without phone or e-mail access. Because surveys are voluntarily submitted, the results may skew opinion toward a select group of outspoken customers who may not be representative of the general population.
The use of patient satisfaction surveys, especially when they are linked to employee compensation, may create a system of survey-based value. This is similar to the problem of defensive medicine, where providers perform medicine in a way that reduces legal risk. Aware that patients will be asked to fill out satisfaction surveys, associates may perform in a way that increases patient satisfaction scores at the expense of patient outcomes or the bottom line. Some institutions may inappropriately “cherry-pick” the easy-to-treat patient and transfer medically complex cases elsewhere.
It is not clear how to best measure the quality of a health care experience. With the broad range of patient encounter types and the inherent complexity of collaboration among providers, it is difficult to determine to what degree satisfaction can be attributed to individual providers or specific environmental factors. Patients do not typically interact with a specific provider, but are treated by a service delivery system, which often encompasses multiple players and multiple physical locations. Moreover, it is not always clear when the patient encounter begins and ends.
Despite the criticism of patient satisfaction survey methodology, the patient must ultimately define the value of the health care service offering. This “voice of the customer” approach is a diversion from the antiquated practitioner-centric model. Traditionally, patient appointment times and locations are decided by the availability and convenience of the provider. Many consumers have compensated for this inefficiency by accessing local emergency departments for nonurgent ambulatory care. Nonetheless,EDs often suffer from long waits and higher costs. Facilities designed for urgent, but nonemergent, care have attempted to address convenience issues but these facilities sacrifice continuity and specialization of care. In a truly patient-centric health care model, patients would be provided the care that they need, when and where they need it.
The patient satisfaction survey remains a primary tool for linking patient-centered value to health care reform. Ranking the results among market competitors can provide an incentive for improvement. Health care professionals are competitive by nature and the extrinsic motivation of quality rankings can be beneficial if well controlled. Employers should use caution when using survey data for performance measurement because survey data are subject to a variety of sources of bias or error. Patient survey data should be used to drive improvement, not to punish. Further research on patient survey methodology is needed to elucidate improved methods of bringing the voice of the patient to the forefront of health care reform.
References
1. N Engl J Med. 2003 Aug 21;349(8):768-75.
2. Centers for Medicare & Medicaid Services. Medicare hospital quality chartbook: Performance report on outcome measures. September 2014.
3. N Engl J Med. 2008 Oct 30;359(18):1921-31.
4. “Better Customer Insight – in Real Time,” by Emma K. Macdonald, Hugh N. Wilson, and Umut Konuş (Harvard Business Review, September 2012).
5. “The Dangers of Linking Pay to Customer Feedback,” by Rob Markey, (Harvard Business Review, Sept. 8, 2011).
6. “Health Care’s Service Fanatics,” by James I. Merlino and Ananth Raman, (Harvard Business Review, May 2013).
7. Trochim, WMK. Research Methods Knowledge Base.
Dr. Davis is a pediatric gastroenterologists at University of Florida Health, Gainesville. He has no financial relationships relevant to this article to disclose.
The American health care system has been plagued by poor medical outcomes and an inefficient cost structure for many years.1,2 The primary loser in this game is the patient, the player who bears the brunt of health care expenses, either directly or through taxes, and whose voice has been largely ignored until recently. As with many other service industries, health care delivery represents a highly complex process that is difficult to define or measure. Innovative procedures and state-of-the-art medical therapies have provided effective options that extend or improve life, but at increasing cost.
Further complicating the existing cost-vs.-benefit debate is a relatively new polemic regarding the degree to which patient satisfaction should be a measure of health care quality. How satisfied will a patient be with his or her health care experience? This depends, in large part, on the medical outcome.3
Many public and private organizations use patient satisfaction surveys to measure the performance of health care delivery systems. In many cases, patient satisfaction data are being used to determine insurance payouts, physician compensation, and institutional rankings.5 It may seem logical to adopt pay-for-performance strategies based on patient satisfaction surveys, but there is a fundamental flaw: Survey data are not always accurate.
It is not entirely clear what constitutes a positive or negative health care experience. Much of this depends on the expectations of the consumer.6 Those with low expectations may be delighted with mediocre performance. Those with inflated expectations may be disappointed even when provided excellent customer service.
Surveys are not durable. That is, when performed under distinct environmental conditions or at different times, surveys may not produce the same results when repeated by the same respondent.7 Surveys are easily manipulated by simple changes in wording or punctuation. Some specific encounters may be rated as “unsatisfactory” because of external factors, circumstances beyond the control of the health care provider.
Many health care providers feel that surveys are poor indicators of individual performance. Some critics highlight a paucity of data. A limited number of returned surveys, relative to the total number of encounters, may yield results that are not statistically significant. Increasing the amount of data decreases the risk that a sample set taken from the studied population is the result of sampling error alone. Nonetheless, sampling error is never completely eliminated, and it is not entirely clear to what degree statistical significance should be used to substantiate satisfaction, a subjective measure.
Surveys often provide data in a very small range, making ranking of facilities or providers difficult. For example, national polling services utilize surveys with thousands of respondents and the margin of error often exceeds plus or minus 3%. Data sets with a smaller number of responses have margins of error that are even greater. Even plus or minus 3% is a sizable deviation when considering that health care survey results often are compared and ranked based on a distribution of scores in a narrow response range. In the author’s experience, a 6% difference in survey scores can represent the difference between a ranking of “excellent” and “poor.”
Many patients are disenfranchised by survey methodology. In the most extreme example, deceased or severely disabled patients are unable to provide feedback. Patients transferred to other facilities and those who are lost to follow-up will be missed also. Many patient surveys may not be successfully retrieved from the homeless, the illiterate, minors, or those without phone or e-mail access. Because surveys are voluntarily submitted, the results may skew opinion toward a select group of outspoken customers who may not be representative of the general population.
The use of patient satisfaction surveys, especially when they are linked to employee compensation, may create a system of survey-based value. This is similar to the problem of defensive medicine, where providers perform medicine in a way that reduces legal risk. Aware that patients will be asked to fill out satisfaction surveys, associates may perform in a way that increases patient satisfaction scores at the expense of patient outcomes or the bottom line. Some institutions may inappropriately “cherry-pick” the easy-to-treat patient and transfer medically complex cases elsewhere.
It is not clear how to best measure the quality of a health care experience. With the broad range of patient encounter types and the inherent complexity of collaboration among providers, it is difficult to determine to what degree satisfaction can be attributed to individual providers or specific environmental factors. Patients do not typically interact with a specific provider, but are treated by a service delivery system, which often encompasses multiple players and multiple physical locations. Moreover, it is not always clear when the patient encounter begins and ends.
Despite the criticism of patient satisfaction survey methodology, the patient must ultimately define the value of the health care service offering. This “voice of the customer” approach is a diversion from the antiquated practitioner-centric model. Traditionally, patient appointment times and locations are decided by the availability and convenience of the provider. Many consumers have compensated for this inefficiency by accessing local emergency departments for nonurgent ambulatory care. Nonetheless,EDs often suffer from long waits and higher costs. Facilities designed for urgent, but nonemergent, care have attempted to address convenience issues but these facilities sacrifice continuity and specialization of care. In a truly patient-centric health care model, patients would be provided the care that they need, when and where they need it.
The patient satisfaction survey remains a primary tool for linking patient-centered value to health care reform. Ranking the results among market competitors can provide an incentive for improvement. Health care professionals are competitive by nature and the extrinsic motivation of quality rankings can be beneficial if well controlled. Employers should use caution when using survey data for performance measurement because survey data are subject to a variety of sources of bias or error. Patient survey data should be used to drive improvement, not to punish. Further research on patient survey methodology is needed to elucidate improved methods of bringing the voice of the patient to the forefront of health care reform.
References
1. N Engl J Med. 2003 Aug 21;349(8):768-75.
2. Centers for Medicare & Medicaid Services. Medicare hospital quality chartbook: Performance report on outcome measures. September 2014.
3. N Engl J Med. 2008 Oct 30;359(18):1921-31.
4. “Better Customer Insight – in Real Time,” by Emma K. Macdonald, Hugh N. Wilson, and Umut Konuş (Harvard Business Review, September 2012).
5. “The Dangers of Linking Pay to Customer Feedback,” by Rob Markey, (Harvard Business Review, Sept. 8, 2011).
6. “Health Care’s Service Fanatics,” by James I. Merlino and Ananth Raman, (Harvard Business Review, May 2013).
7. Trochim, WMK. Research Methods Knowledge Base.
Dr. Davis is a pediatric gastroenterologists at University of Florida Health, Gainesville. He has no financial relationships relevant to this article to disclose.
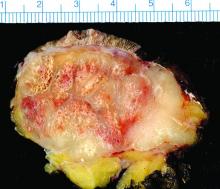
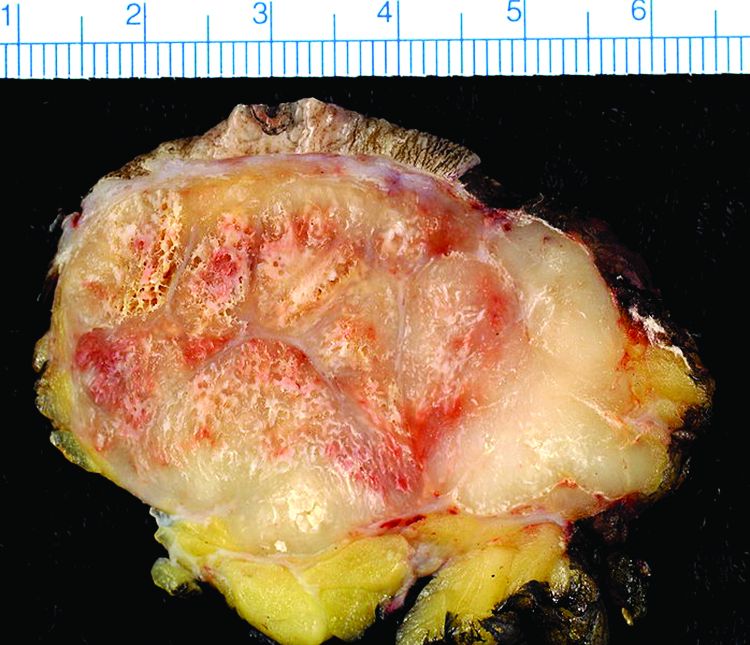
Study finds 19% of Merkel cell carcinomas are virus negative
Nineteen percent of Merkel cell carcinomas are not driven by the Merkel cell polyomavirus and are substantially more aggressive than those that are virus positive, according to a report published online in the Journal of Investigative Dermatology.
This and other findings from a retrospective analysis of samples from 282 Merkel cell carcinomas in a Seattle repository “suggest that it may be clinically indicated to determine tumor viral status at the time of diagnosis, as the results may affect prognosis as well as optimal clinical management,” wrote Ata Moshiri, MD, who was with the University of Washington, Seattle, at the time of the study, and his associates.
Given that virus-negative Merkel cell carcinomas carry a markedly higher risk of recurrence, progression, and patient mortality, “clinicians may consider larger initial surgical margins, larger radiotherapy fields, and the use of regional nodal therapy even in the absence of documented nodal metastasis. Closer clinical follow-up and more frequent radiologic surveillance may be justified for patients with virus-negative tumors because ... serologic monitoring is not feasible for this patient population,” the investigators noted.
The incidence of Merkel cell carcinoma, a rare and aggressive neuroendocrine skin cancer with an overall disease-related mortality of 40%, has quadrupled during the last 20 years. This is likely because of the increasing prevalence of risk factors for the cancer, including advanced age, increased cumulative exposure to ultraviolet light, and systemic immune suppression.
Data concerning the presence of Merkel cell polyomavirus in these cancers are conflicting, with estimates of virus positivity ranging from 20% all the way to 100% in some studies. Part of the reason for this wide range of estimates is that there is no accepted preferred method for measuring the viral status of these tumors. Moreover, the prognostic significance of that viral status is also debated. Thus, most Merkel cell cancers are not routinely analyzed for the presence of Merkel cell polyomavirus.
To pin down the prevalence of virus positivity and establish whether it impacts clinical outcomes, Dr. Moshiri and his associates analyzed 282 Merkel cell specimens collected since 1980 and stored in a Seattle repository, along with clinical data. They tested each specimen using an immunohistochemical assay to detect one antibody (CM2B4), a different immunohistochemical assay to detect another antibody (Ab3), and a quantitative PCR assay for polyomavirus DNA. To be considered virus positive, each specimen had to show the presence of the virus on at least two of these tests.
By these criteria, 53 tumors (18.8%) were found to be virus negative and 229 (81.2%) to be virus positive.
Virus-negative tumors tended to be smaller than virus-positive tumors at presentation. Despite their smaller size, virus-negative tumors tended to be more advanced at presentation: 66.7% had nodal or distant metastases, compared with 48.3% of virus-positive tumors.
A total of 66.7% of virus-negative carcinomas progressed, compared with only 43.6% of virus-positive carcinomas. The median time to progression was 1.2 years for virus-negative cancers, but was not reached for virus-positive cancers. In a univariate analysis, virus-negative tumors had a nearly twofold higher risk of progression. In a multivariate analysis that adjusted for differences in disease stage at presentation, the HR fell slightly to 1.55.
Cancer-specific mortality was 45.3% for virus-negative tumors, compared with 26.3% for virus-positive tumors. Median time to death from Merkel cell carcinoma was 3.7 years for virus-negative tumors but was not reached for virus-positive tumors. In a univariate analysis, virus-negative tumors carried a nearly twofold higher risk of death from Merkel cell carcinoma. In a multivariate analysis that adjusted for differences in disease stage at presentation, the HR fell somewhat to 1.50.
Median overall survival was 3.3 years for patients with virus-negative tumors, compared with 4.6 years for patients with virus-positive tumors.
These findings indicate that a more advanced cancer stage at diagnosis accounts for some but not all of the poorer clinical outcomes seen with virus-negative tumors, the investigators said.
This study could not assess why virus-negative Merkel cell carcinomas are more aggressive and lethal than virus-positive ones, but previous studies have proposed some plausible biological mechanisms. Virus-negative tumors carry a greater number of chromosomal aberrations, a greater burden of nucleotide mutations, and a greater number of mutations in known oncogenic pathways. They also may be more immunogenic “due to their constitutive expression of oncoproteins that may serve as targets for cytotoxic tumor-infiltrating lymphocytes,” Dr. Moshiri and his associates said.
They added that in this study, the immunohistochemical assay for CM2B4 antibodies was the test that most accurately identified tumors that had worse outcomes. “We believe that the CM2B4 antibody test may be well-suited for routine clinical use” because of its sensitivity and specificity in this application, its commercial availability, “and the ease with which it could be included in the work flow of clinical laboratories accustomed to immunohistochemistry.”
Dr. Moshiri is currently at the University of Pennsylvania, Philadelphia.
The National Institutes of Health, the Colin Johnston Fund, and the Janet Canning Fund supported the study. Dr. Moshiri reported having no relevant financial disclosures; one of his associates reported that her institute received research funding from Valeant and Pfizer unrelated to this work.
Nineteen percent of Merkel cell carcinomas are not driven by the Merkel cell polyomavirus and are substantially more aggressive than those that are virus positive, according to a report published online in the Journal of Investigative Dermatology.
This and other findings from a retrospective analysis of samples from 282 Merkel cell carcinomas in a Seattle repository “suggest that it may be clinically indicated to determine tumor viral status at the time of diagnosis, as the results may affect prognosis as well as optimal clinical management,” wrote Ata Moshiri, MD, who was with the University of Washington, Seattle, at the time of the study, and his associates.
Given that virus-negative Merkel cell carcinomas carry a markedly higher risk of recurrence, progression, and patient mortality, “clinicians may consider larger initial surgical margins, larger radiotherapy fields, and the use of regional nodal therapy even in the absence of documented nodal metastasis. Closer clinical follow-up and more frequent radiologic surveillance may be justified for patients with virus-negative tumors because ... serologic monitoring is not feasible for this patient population,” the investigators noted.
The incidence of Merkel cell carcinoma, a rare and aggressive neuroendocrine skin cancer with an overall disease-related mortality of 40%, has quadrupled during the last 20 years. This is likely because of the increasing prevalence of risk factors for the cancer, including advanced age, increased cumulative exposure to ultraviolet light, and systemic immune suppression.
Data concerning the presence of Merkel cell polyomavirus in these cancers are conflicting, with estimates of virus positivity ranging from 20% all the way to 100% in some studies. Part of the reason for this wide range of estimates is that there is no accepted preferred method for measuring the viral status of these tumors. Moreover, the prognostic significance of that viral status is also debated. Thus, most Merkel cell cancers are not routinely analyzed for the presence of Merkel cell polyomavirus.
To pin down the prevalence of virus positivity and establish whether it impacts clinical outcomes, Dr. Moshiri and his associates analyzed 282 Merkel cell specimens collected since 1980 and stored in a Seattle repository, along with clinical data. They tested each specimen using an immunohistochemical assay to detect one antibody (CM2B4), a different immunohistochemical assay to detect another antibody (Ab3), and a quantitative PCR assay for polyomavirus DNA. To be considered virus positive, each specimen had to show the presence of the virus on at least two of these tests.
By these criteria, 53 tumors (18.8%) were found to be virus negative and 229 (81.2%) to be virus positive.
Virus-negative tumors tended to be smaller than virus-positive tumors at presentation. Despite their smaller size, virus-negative tumors tended to be more advanced at presentation: 66.7% had nodal or distant metastases, compared with 48.3% of virus-positive tumors.
A total of 66.7% of virus-negative carcinomas progressed, compared with only 43.6% of virus-positive carcinomas. The median time to progression was 1.2 years for virus-negative cancers, but was not reached for virus-positive cancers. In a univariate analysis, virus-negative tumors had a nearly twofold higher risk of progression. In a multivariate analysis that adjusted for differences in disease stage at presentation, the HR fell slightly to 1.55.
Cancer-specific mortality was 45.3% for virus-negative tumors, compared with 26.3% for virus-positive tumors. Median time to death from Merkel cell carcinoma was 3.7 years for virus-negative tumors but was not reached for virus-positive tumors. In a univariate analysis, virus-negative tumors carried a nearly twofold higher risk of death from Merkel cell carcinoma. In a multivariate analysis that adjusted for differences in disease stage at presentation, the HR fell somewhat to 1.50.
Median overall survival was 3.3 years for patients with virus-negative tumors, compared with 4.6 years for patients with virus-positive tumors.
These findings indicate that a more advanced cancer stage at diagnosis accounts for some but not all of the poorer clinical outcomes seen with virus-negative tumors, the investigators said.
This study could not assess why virus-negative Merkel cell carcinomas are more aggressive and lethal than virus-positive ones, but previous studies have proposed some plausible biological mechanisms. Virus-negative tumors carry a greater number of chromosomal aberrations, a greater burden of nucleotide mutations, and a greater number of mutations in known oncogenic pathways. They also may be more immunogenic “due to their constitutive expression of oncoproteins that may serve as targets for cytotoxic tumor-infiltrating lymphocytes,” Dr. Moshiri and his associates said.
They added that in this study, the immunohistochemical assay for CM2B4 antibodies was the test that most accurately identified tumors that had worse outcomes. “We believe that the CM2B4 antibody test may be well-suited for routine clinical use” because of its sensitivity and specificity in this application, its commercial availability, “and the ease with which it could be included in the work flow of clinical laboratories accustomed to immunohistochemistry.”
Dr. Moshiri is currently at the University of Pennsylvania, Philadelphia.
The National Institutes of Health, the Colin Johnston Fund, and the Janet Canning Fund supported the study. Dr. Moshiri reported having no relevant financial disclosures; one of his associates reported that her institute received research funding from Valeant and Pfizer unrelated to this work.
Nineteen percent of Merkel cell carcinomas are not driven by the Merkel cell polyomavirus and are substantially more aggressive than those that are virus positive, according to a report published online in the Journal of Investigative Dermatology.
This and other findings from a retrospective analysis of samples from 282 Merkel cell carcinomas in a Seattle repository “suggest that it may be clinically indicated to determine tumor viral status at the time of diagnosis, as the results may affect prognosis as well as optimal clinical management,” wrote Ata Moshiri, MD, who was with the University of Washington, Seattle, at the time of the study, and his associates.
Given that virus-negative Merkel cell carcinomas carry a markedly higher risk of recurrence, progression, and patient mortality, “clinicians may consider larger initial surgical margins, larger radiotherapy fields, and the use of regional nodal therapy even in the absence of documented nodal metastasis. Closer clinical follow-up and more frequent radiologic surveillance may be justified for patients with virus-negative tumors because ... serologic monitoring is not feasible for this patient population,” the investigators noted.
The incidence of Merkel cell carcinoma, a rare and aggressive neuroendocrine skin cancer with an overall disease-related mortality of 40%, has quadrupled during the last 20 years. This is likely because of the increasing prevalence of risk factors for the cancer, including advanced age, increased cumulative exposure to ultraviolet light, and systemic immune suppression.
Data concerning the presence of Merkel cell polyomavirus in these cancers are conflicting, with estimates of virus positivity ranging from 20% all the way to 100% in some studies. Part of the reason for this wide range of estimates is that there is no accepted preferred method for measuring the viral status of these tumors. Moreover, the prognostic significance of that viral status is also debated. Thus, most Merkel cell cancers are not routinely analyzed for the presence of Merkel cell polyomavirus.
To pin down the prevalence of virus positivity and establish whether it impacts clinical outcomes, Dr. Moshiri and his associates analyzed 282 Merkel cell specimens collected since 1980 and stored in a Seattle repository, along with clinical data. They tested each specimen using an immunohistochemical assay to detect one antibody (CM2B4), a different immunohistochemical assay to detect another antibody (Ab3), and a quantitative PCR assay for polyomavirus DNA. To be considered virus positive, each specimen had to show the presence of the virus on at least two of these tests.
By these criteria, 53 tumors (18.8%) were found to be virus negative and 229 (81.2%) to be virus positive.
Virus-negative tumors tended to be smaller than virus-positive tumors at presentation. Despite their smaller size, virus-negative tumors tended to be more advanced at presentation: 66.7% had nodal or distant metastases, compared with 48.3% of virus-positive tumors.
A total of 66.7% of virus-negative carcinomas progressed, compared with only 43.6% of virus-positive carcinomas. The median time to progression was 1.2 years for virus-negative cancers, but was not reached for virus-positive cancers. In a univariate analysis, virus-negative tumors had a nearly twofold higher risk of progression. In a multivariate analysis that adjusted for differences in disease stage at presentation, the HR fell slightly to 1.55.
Cancer-specific mortality was 45.3% for virus-negative tumors, compared with 26.3% for virus-positive tumors. Median time to death from Merkel cell carcinoma was 3.7 years for virus-negative tumors but was not reached for virus-positive tumors. In a univariate analysis, virus-negative tumors carried a nearly twofold higher risk of death from Merkel cell carcinoma. In a multivariate analysis that adjusted for differences in disease stage at presentation, the HR fell somewhat to 1.50.
Median overall survival was 3.3 years for patients with virus-negative tumors, compared with 4.6 years for patients with virus-positive tumors.
These findings indicate that a more advanced cancer stage at diagnosis accounts for some but not all of the poorer clinical outcomes seen with virus-negative tumors, the investigators said.
This study could not assess why virus-negative Merkel cell carcinomas are more aggressive and lethal than virus-positive ones, but previous studies have proposed some plausible biological mechanisms. Virus-negative tumors carry a greater number of chromosomal aberrations, a greater burden of nucleotide mutations, and a greater number of mutations in known oncogenic pathways. They also may be more immunogenic “due to their constitutive expression of oncoproteins that may serve as targets for cytotoxic tumor-infiltrating lymphocytes,” Dr. Moshiri and his associates said.
They added that in this study, the immunohistochemical assay for CM2B4 antibodies was the test that most accurately identified tumors that had worse outcomes. “We believe that the CM2B4 antibody test may be well-suited for routine clinical use” because of its sensitivity and specificity in this application, its commercial availability, “and the ease with which it could be included in the work flow of clinical laboratories accustomed to immunohistochemistry.”
Dr. Moshiri is currently at the University of Pennsylvania, Philadelphia.
The National Institutes of Health, the Colin Johnston Fund, and the Janet Canning Fund supported the study. Dr. Moshiri reported having no relevant financial disclosures; one of his associates reported that her institute received research funding from Valeant and Pfizer unrelated to this work.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Key clinical point: Nineteen percent of Merkel cell carcinomas are not driven by the Merkel cell polyomavirus and are substantially more aggressive than those that are virus positive.
Major finding: The 53 virus-negative tumors carried a cancer-specific mortality of 45.3%, while the 229 virus-positive tumors carried a cancer-specific mortality of 26.2%.
Data source: A retrospective molecular analysis of samples from 282 Merkel cell carcinomas in a Seattle repository for the presence of Merkel cell polyomavirus.
Disclosures: The National Institutes of Health, the Colin Johnston Fund, and the Janet Canning Fund supported the study. Dr. Moshiri reported having no relevant financial disclosures; one of his associates reported that her institute received research funding from Valeant and Pfizer unrelated to this work.
Predicting 30-Day Readmissions
Rates of 30-day readmissions, which are both common and difficult to predict, are of major concern to hospitalists.
“Unfortunately, interventions developed to date have not been universally successful in preventing hospital readmissions for various medical conditions and patient types,” according to a recent article in the Journal of Hospital Medicine. “One potential explanation for this is the inability to reliably predict which patients are at risk for readmission to better target preventative interventions.”
This fact led the authors to perform a study to determine whether the occurrence of automated clinical deterioration alerts (CDAs) could predict 30-day hospital readmission. The 36 variables in the CDA algorithm included age, radiologic agents, and temperature. The retrospective study assessed 3,015 patients admitted to eight general medicine units for all-cause 30-day readmission. Of these, 1,141 patients triggered a CDA, and they were significantly more likely to have a 30-day readmission compared to those who did not trigger a CDA (23.6% versus 15.9%).
The researchers concluded that readily identifiable clinical variables can be identified that predict 30-day readmission.
“It may be important to include these variables in existing prediction tools if pay for performance and across-institution comparisons are to be ‘fair’ to institutions that care for more seriously ill patients,” they write. “The development of an accurate real-time early warning system has the potential to identify patients at risk for various adverse outcomes including clinical deterioration, hospital death and post-discharge readmission. By identifying patients at greatest risk for readmission, valuable healthcare resources can be better targeted to such populations.”
Reference
- Micek ST, Samant M, Bailey T, et al. Real-time automated clinical deterioration alerts predict thirty-day hospital readmission [published online ahead of print June 3, 2016]. J Hosp Med. doi:10.1002/jhm.2617.
Quick Byte
The Cost of Vaccine Avoidance
Many Americans avoid their recommended vaccines: For example, the Centers for Disease Control and Prevention (CDC) reports that only 42% of U.S. adults age 18 or older received the flu vaccine during the 2015–2016 flu season. A study recently released online by Health Affairs calculated the annual cost of the diseases associated with 10 vaccines the CDC recommends for adults. In 2015, that economic burden was $8.95 billion. A full 80% of that—$7.1 billion—was attributed to unvaccinated people.
Reference
- The cost of US adult vaccine avoidance: $8.95 billion in 2015. Health Affairs website. Accessed October 17, 2016
Rates of 30-day readmissions, which are both common and difficult to predict, are of major concern to hospitalists.
“Unfortunately, interventions developed to date have not been universally successful in preventing hospital readmissions for various medical conditions and patient types,” according to a recent article in the Journal of Hospital Medicine. “One potential explanation for this is the inability to reliably predict which patients are at risk for readmission to better target preventative interventions.”
This fact led the authors to perform a study to determine whether the occurrence of automated clinical deterioration alerts (CDAs) could predict 30-day hospital readmission. The 36 variables in the CDA algorithm included age, radiologic agents, and temperature. The retrospective study assessed 3,015 patients admitted to eight general medicine units for all-cause 30-day readmission. Of these, 1,141 patients triggered a CDA, and they were significantly more likely to have a 30-day readmission compared to those who did not trigger a CDA (23.6% versus 15.9%).
The researchers concluded that readily identifiable clinical variables can be identified that predict 30-day readmission.
“It may be important to include these variables in existing prediction tools if pay for performance and across-institution comparisons are to be ‘fair’ to institutions that care for more seriously ill patients,” they write. “The development of an accurate real-time early warning system has the potential to identify patients at risk for various adverse outcomes including clinical deterioration, hospital death and post-discharge readmission. By identifying patients at greatest risk for readmission, valuable healthcare resources can be better targeted to such populations.”
Reference
- Micek ST, Samant M, Bailey T, et al. Real-time automated clinical deterioration alerts predict thirty-day hospital readmission [published online ahead of print June 3, 2016]. J Hosp Med. doi:10.1002/jhm.2617.
Quick Byte
The Cost of Vaccine Avoidance
Many Americans avoid their recommended vaccines: For example, the Centers for Disease Control and Prevention (CDC) reports that only 42% of U.S. adults age 18 or older received the flu vaccine during the 2015–2016 flu season. A study recently released online by Health Affairs calculated the annual cost of the diseases associated with 10 vaccines the CDC recommends for adults. In 2015, that economic burden was $8.95 billion. A full 80% of that—$7.1 billion—was attributed to unvaccinated people.
Reference
- The cost of US adult vaccine avoidance: $8.95 billion in 2015. Health Affairs website. Accessed October 17, 2016
Rates of 30-day readmissions, which are both common and difficult to predict, are of major concern to hospitalists.
“Unfortunately, interventions developed to date have not been universally successful in preventing hospital readmissions for various medical conditions and patient types,” according to a recent article in the Journal of Hospital Medicine. “One potential explanation for this is the inability to reliably predict which patients are at risk for readmission to better target preventative interventions.”
This fact led the authors to perform a study to determine whether the occurrence of automated clinical deterioration alerts (CDAs) could predict 30-day hospital readmission. The 36 variables in the CDA algorithm included age, radiologic agents, and temperature. The retrospective study assessed 3,015 patients admitted to eight general medicine units for all-cause 30-day readmission. Of these, 1,141 patients triggered a CDA, and they were significantly more likely to have a 30-day readmission compared to those who did not trigger a CDA (23.6% versus 15.9%).
The researchers concluded that readily identifiable clinical variables can be identified that predict 30-day readmission.
“It may be important to include these variables in existing prediction tools if pay for performance and across-institution comparisons are to be ‘fair’ to institutions that care for more seriously ill patients,” they write. “The development of an accurate real-time early warning system has the potential to identify patients at risk for various adverse outcomes including clinical deterioration, hospital death and post-discharge readmission. By identifying patients at greatest risk for readmission, valuable healthcare resources can be better targeted to such populations.”
Reference
- Micek ST, Samant M, Bailey T, et al. Real-time automated clinical deterioration alerts predict thirty-day hospital readmission [published online ahead of print June 3, 2016]. J Hosp Med. doi:10.1002/jhm.2617.
Quick Byte
The Cost of Vaccine Avoidance
Many Americans avoid their recommended vaccines: For example, the Centers for Disease Control and Prevention (CDC) reports that only 42% of U.S. adults age 18 or older received the flu vaccine during the 2015–2016 flu season. A study recently released online by Health Affairs calculated the annual cost of the diseases associated with 10 vaccines the CDC recommends for adults. In 2015, that economic burden was $8.95 billion. A full 80% of that—$7.1 billion—was attributed to unvaccinated people.
Reference
- The cost of US adult vaccine avoidance: $8.95 billion in 2015. Health Affairs website. Accessed October 17, 2016
Ramping Up Telehealth’s Possibilities
Twenty percent of Americans live in areas where there are shortages of physicians, according to a policy brief in Health Affairs. Some analysts believe the answer to this problem is telehealth, which they say could also save the healthcare industry some $4.28 billion annually.
While the Affordable Care Act signaled a move toward telehealth development at the federal level (through Medicare), states still largely govern coverage of telehealth services by Medicaid or private insurers.
“Currently there is no uniform legal approach to telehealth, and this continues to be a major challenge in its provision. In particular, concerns about reimbursements, for both private insurers and public programs such as Medicaid, continue to limit the implementation and use of telehealth services,” according to the brief.
Now, Congress is considering the Medicare Telehealth Parity Act, intended to modernize the way Medicare reimburses telehealth services and to expand locations and coverage. To enjoy the benefits of telehealth services, states are likely to move toward full-parity laws for the services, the brief notes.
“Without parity, there are limited incentives for the development of telehealth or for providers to move toward telehealth services,” according to the brief. “If there are no incentives to use telehealth, then providers will continue to focus on in-person care, which will keep healthcare costs high, continue to create access issues, and possibly provide lesser standards of care for chronic disease patients who benefit from remote monitoring.”
Reference
1. Yang T. Telehealth parity laws. Health Affairs Website. Accessed October 17, 2016.
Twenty percent of Americans live in areas where there are shortages of physicians, according to a policy brief in Health Affairs. Some analysts believe the answer to this problem is telehealth, which they say could also save the healthcare industry some $4.28 billion annually.
While the Affordable Care Act signaled a move toward telehealth development at the federal level (through Medicare), states still largely govern coverage of telehealth services by Medicaid or private insurers.
“Currently there is no uniform legal approach to telehealth, and this continues to be a major challenge in its provision. In particular, concerns about reimbursements, for both private insurers and public programs such as Medicaid, continue to limit the implementation and use of telehealth services,” according to the brief.
Now, Congress is considering the Medicare Telehealth Parity Act, intended to modernize the way Medicare reimburses telehealth services and to expand locations and coverage. To enjoy the benefits of telehealth services, states are likely to move toward full-parity laws for the services, the brief notes.
“Without parity, there are limited incentives for the development of telehealth or for providers to move toward telehealth services,” according to the brief. “If there are no incentives to use telehealth, then providers will continue to focus on in-person care, which will keep healthcare costs high, continue to create access issues, and possibly provide lesser standards of care for chronic disease patients who benefit from remote monitoring.”
Reference
1. Yang T. Telehealth parity laws. Health Affairs Website. Accessed October 17, 2016.
Twenty percent of Americans live in areas where there are shortages of physicians, according to a policy brief in Health Affairs. Some analysts believe the answer to this problem is telehealth, which they say could also save the healthcare industry some $4.28 billion annually.
While the Affordable Care Act signaled a move toward telehealth development at the federal level (through Medicare), states still largely govern coverage of telehealth services by Medicaid or private insurers.
“Currently there is no uniform legal approach to telehealth, and this continues to be a major challenge in its provision. In particular, concerns about reimbursements, for both private insurers and public programs such as Medicaid, continue to limit the implementation and use of telehealth services,” according to the brief.
Now, Congress is considering the Medicare Telehealth Parity Act, intended to modernize the way Medicare reimburses telehealth services and to expand locations and coverage. To enjoy the benefits of telehealth services, states are likely to move toward full-parity laws for the services, the brief notes.
“Without parity, there are limited incentives for the development of telehealth or for providers to move toward telehealth services,” according to the brief. “If there are no incentives to use telehealth, then providers will continue to focus on in-person care, which will keep healthcare costs high, continue to create access issues, and possibly provide lesser standards of care for chronic disease patients who benefit from remote monitoring.”
Reference
1. Yang T. Telehealth parity laws. Health Affairs Website. Accessed October 17, 2016.
Secondary reference panel expands access for CML monitoring
A cell-based BCR-ABL1 secondary reference panel traceable to the World Health Organization BCR-ABL1 International Genetic Reference Panel – with an additional MR4.5 level – provides easier access to International Scale calibration and can act as a tool for assay optimization, validation, and quality assurance for molecular monitoring in chronic myeloid leukemia patients.
Such monitoring is important to disease management, especially for decision making with respect to treatment cessation. The new secondary reference panel would allow laboratories to circumvent the oft-used and time-consuming sample exchange process with reference laboratories for International Scale (IS) calibration, Nicholas C. P. Cross, PhD, of the University of Southampton (England) and colleagues wrote in an article published in Leukemia (2016 Jun 3;30:1844-52).
“The development of BCR-ABL1 tyrosine kinase inhibitors ... has enabled progressively deeper molecular responses in CML patients undergoing tyrosine kinase inhibitor therapy,” the authors wrote, noting that deeper molecular responses are “important milestones for patients considering treatment cessation,” and that “other landmarks on the IS also represent different treatment decision thresholds and prognostic outcomes.”
For this reason, regular molecular monitoring using real-time reverse-transcription quantitative PCR (RqPCR) is recommended for optimal disease management, they said.
“As treatment decisions are directly impacted by test results, accuracy and precision of BCR-ABL1 assays across the entire measurement range is crucial for patient management, especially in patients with deep molecular responses when considering possible treatment cessation,” they wrote.
Access to the material for the WHO BCR-ABL1 reference panel (MR1-MR4), which was developed in 2010 as a primary standard for BCR-ABL1 assay IS calibration, is limited, particularly for smaller laboratories, the authors said.
The secondary reference panel they developed, however, is “traceable to and faithfully replicates the WHO panel in both raw materials (lyopholized.K562 and HL-60 cell mixes) and manufacturing process, with the addition of a MR4.5 level.” The secondary panel was calibrated to IS using digital PCR against ABL1, BCR, and GUSB as reference genes, and was successfully evaluated by 45 different BCR-ABL1 assays at 44 different clinical laboratories in a multinational evaluation study.
The MR4.5 level was added to allow for more accurate IS calibration “as CML patients reaching this deep molecular response are increasingly being considered for treatment cessation,” the authors noted.
“Quality-control assessments indicated that the secondary panel had minimal residual moisture, excellent vial-to-vial homogeneity and greater than 2.5 years of real-time stability,” they said.
Further, the panel was successfully processed by all of the laboratories, indicating that it is compatible with many different BCR-ABL1 test configurations, they added.
Of note, the number of assays that achieved good precision and sensitivity exceeded the number that achieved good IS accuracy, suggesting an unmet need for “a simple and broadly available calibration mechanism, such as this secondary panel, to ensure IS accuracy is maintained in laboratories over time,” they wrote, concluding that such a panel “can provide easier access to IS calibration, as well as act as a tool for assay optimization, validation and quality assurance.”
In a letter to the editor in regard to the findings by Cross et al., Maria Sol Ruiz of Instituto Alexander Fleming in Buenos Aires, and colleagues wrote about their own development and validation of “secondary reference materials calibrated to the IS through the WHO primary standards in order to facilitate standardization of molecular monitoring in Latin America,” (Leukemia. 2016 Aug 19. doi: 10.1038/leu.2016.197).
In their study, the letter’s authors demonstrated that secondary reference biological calibrators anchored to the WHO primary standards can decrease inter-laboratory variability.
“Our results, together with those recently reported by Cross et al., substantiate the objective initially set during the establishment of the WHO primary standards, that is, to facilitate worldwide diffusion of the IS. For the first time in Latin America, this study provides a platform on which to assess the performance of distinct clinical BCR-ABL1 tests and confirm the utility of secondary reference materials to further improve IS accuracy and inter-laboratory precision,” they wrote, noting that their efforts will continue through provision of secondary reference material to centers involved in their project, as well as to potential new participants.
“Moreover, due to its higher precision and absolute quantification capability, we are evaluating the possibility of including digital PCR as the calibration method for the future,” they said.
The study discussed in the letter to the editor was supported by a grant from Novartis to one of the authors. Novartis also paid speaking fees to some of the authors. The study by Dr. Cross et al. was also funded by Novartis and some authors are employed by Novartis. The remaining authors, including Dr. Cross, reported having no disclosures.
Harmonizing BCR-ABL1 real time quantitative PCR (RqPCR) is extremely important for precisely interpreting therapeutic response in CML patients and for being able to compare results from various laboratories.
Levels of BCR-ABL1 RNA transcripts are expected to progressively decline with successful response to tyrosine kinase inhibitor therapy. A rise in those levels indicates a loss of response to therapy and typically prompts dose modification or a change in therapy.
Initially, only a qualitative test was available and it measured only the presence or absence of the transcript. The International Standard (IS) allowed the development of a quantitative test. The test is identical to that used in the International Randomized Study of Interferons and STI571 (IRIS), which has a standard baseline of 100% of BCR-ABL1 and major molecular response is defined as a 3 log reduction relative to standard baseline or 0.1% of BCR-ABL1 IS.
However, access became limited – especially for smaller laboratories – to the material for the WHO BCR-ABL1 reference panel (MR1-MR4), a primary standard for BCR-ABL1 assay IS calibration. Calibrated, accredited, primary reference reagents for BCR-ABL1 RqPCR were often too expensive for emergent economies. In response, some laboratories developed conversion factors – a laborious process – and other alternative methods.
By using locally produced secondary cellular calibrators anchored to the WHO primary standards, the elegant work of Cross et al. and Ruiz et al. provided standardization. With this type of initiative, more regional laboratories can assess the performance of their tests and improve their accuracy. Indeed, it is of utmost importance that the exchange of reference standards and quality control samples becomes a common practice in such regions to maximize the reliability of this test. The authors of these studies have started down that path, but there is still a long way to go to achieve higher sensitivity that would permit the detection of even deeper responses and the introduction of digitalized PCR testing.
Maria de Lourdes Chauffaille, MD, and Daniella Kerbauy, MD, are with Fleury Medicina Diagnostica, Sao Paulo, Brazil. Both reported having no disclosures.
Harmonizing BCR-ABL1 real time quantitative PCR (RqPCR) is extremely important for precisely interpreting therapeutic response in CML patients and for being able to compare results from various laboratories.
Levels of BCR-ABL1 RNA transcripts are expected to progressively decline with successful response to tyrosine kinase inhibitor therapy. A rise in those levels indicates a loss of response to therapy and typically prompts dose modification or a change in therapy.
Initially, only a qualitative test was available and it measured only the presence or absence of the transcript. The International Standard (IS) allowed the development of a quantitative test. The test is identical to that used in the International Randomized Study of Interferons and STI571 (IRIS), which has a standard baseline of 100% of BCR-ABL1 and major molecular response is defined as a 3 log reduction relative to standard baseline or 0.1% of BCR-ABL1 IS.
However, access became limited – especially for smaller laboratories – to the material for the WHO BCR-ABL1 reference panel (MR1-MR4), a primary standard for BCR-ABL1 assay IS calibration. Calibrated, accredited, primary reference reagents for BCR-ABL1 RqPCR were often too expensive for emergent economies. In response, some laboratories developed conversion factors – a laborious process – and other alternative methods.
By using locally produced secondary cellular calibrators anchored to the WHO primary standards, the elegant work of Cross et al. and Ruiz et al. provided standardization. With this type of initiative, more regional laboratories can assess the performance of their tests and improve their accuracy. Indeed, it is of utmost importance that the exchange of reference standards and quality control samples becomes a common practice in such regions to maximize the reliability of this test. The authors of these studies have started down that path, but there is still a long way to go to achieve higher sensitivity that would permit the detection of even deeper responses and the introduction of digitalized PCR testing.
Maria de Lourdes Chauffaille, MD, and Daniella Kerbauy, MD, are with Fleury Medicina Diagnostica, Sao Paulo, Brazil. Both reported having no disclosures.
Harmonizing BCR-ABL1 real time quantitative PCR (RqPCR) is extremely important for precisely interpreting therapeutic response in CML patients and for being able to compare results from various laboratories.
Levels of BCR-ABL1 RNA transcripts are expected to progressively decline with successful response to tyrosine kinase inhibitor therapy. A rise in those levels indicates a loss of response to therapy and typically prompts dose modification or a change in therapy.
Initially, only a qualitative test was available and it measured only the presence or absence of the transcript. The International Standard (IS) allowed the development of a quantitative test. The test is identical to that used in the International Randomized Study of Interferons and STI571 (IRIS), which has a standard baseline of 100% of BCR-ABL1 and major molecular response is defined as a 3 log reduction relative to standard baseline or 0.1% of BCR-ABL1 IS.
However, access became limited – especially for smaller laboratories – to the material for the WHO BCR-ABL1 reference panel (MR1-MR4), a primary standard for BCR-ABL1 assay IS calibration. Calibrated, accredited, primary reference reagents for BCR-ABL1 RqPCR were often too expensive for emergent economies. In response, some laboratories developed conversion factors – a laborious process – and other alternative methods.
By using locally produced secondary cellular calibrators anchored to the WHO primary standards, the elegant work of Cross et al. and Ruiz et al. provided standardization. With this type of initiative, more regional laboratories can assess the performance of their tests and improve their accuracy. Indeed, it is of utmost importance that the exchange of reference standards and quality control samples becomes a common practice in such regions to maximize the reliability of this test. The authors of these studies have started down that path, but there is still a long way to go to achieve higher sensitivity that would permit the detection of even deeper responses and the introduction of digitalized PCR testing.
Maria de Lourdes Chauffaille, MD, and Daniella Kerbauy, MD, are with Fleury Medicina Diagnostica, Sao Paulo, Brazil. Both reported having no disclosures.
A cell-based BCR-ABL1 secondary reference panel traceable to the World Health Organization BCR-ABL1 International Genetic Reference Panel – with an additional MR4.5 level – provides easier access to International Scale calibration and can act as a tool for assay optimization, validation, and quality assurance for molecular monitoring in chronic myeloid leukemia patients.
Such monitoring is important to disease management, especially for decision making with respect to treatment cessation. The new secondary reference panel would allow laboratories to circumvent the oft-used and time-consuming sample exchange process with reference laboratories for International Scale (IS) calibration, Nicholas C. P. Cross, PhD, of the University of Southampton (England) and colleagues wrote in an article published in Leukemia (2016 Jun 3;30:1844-52).
“The development of BCR-ABL1 tyrosine kinase inhibitors ... has enabled progressively deeper molecular responses in CML patients undergoing tyrosine kinase inhibitor therapy,” the authors wrote, noting that deeper molecular responses are “important milestones for patients considering treatment cessation,” and that “other landmarks on the IS also represent different treatment decision thresholds and prognostic outcomes.”
For this reason, regular molecular monitoring using real-time reverse-transcription quantitative PCR (RqPCR) is recommended for optimal disease management, they said.
“As treatment decisions are directly impacted by test results, accuracy and precision of BCR-ABL1 assays across the entire measurement range is crucial for patient management, especially in patients with deep molecular responses when considering possible treatment cessation,” they wrote.
Access to the material for the WHO BCR-ABL1 reference panel (MR1-MR4), which was developed in 2010 as a primary standard for BCR-ABL1 assay IS calibration, is limited, particularly for smaller laboratories, the authors said.
The secondary reference panel they developed, however, is “traceable to and faithfully replicates the WHO panel in both raw materials (lyopholized.K562 and HL-60 cell mixes) and manufacturing process, with the addition of a MR4.5 level.” The secondary panel was calibrated to IS using digital PCR against ABL1, BCR, and GUSB as reference genes, and was successfully evaluated by 45 different BCR-ABL1 assays at 44 different clinical laboratories in a multinational evaluation study.
The MR4.5 level was added to allow for more accurate IS calibration “as CML patients reaching this deep molecular response are increasingly being considered for treatment cessation,” the authors noted.
“Quality-control assessments indicated that the secondary panel had minimal residual moisture, excellent vial-to-vial homogeneity and greater than 2.5 years of real-time stability,” they said.
Further, the panel was successfully processed by all of the laboratories, indicating that it is compatible with many different BCR-ABL1 test configurations, they added.
Of note, the number of assays that achieved good precision and sensitivity exceeded the number that achieved good IS accuracy, suggesting an unmet need for “a simple and broadly available calibration mechanism, such as this secondary panel, to ensure IS accuracy is maintained in laboratories over time,” they wrote, concluding that such a panel “can provide easier access to IS calibration, as well as act as a tool for assay optimization, validation and quality assurance.”
In a letter to the editor in regard to the findings by Cross et al., Maria Sol Ruiz of Instituto Alexander Fleming in Buenos Aires, and colleagues wrote about their own development and validation of “secondary reference materials calibrated to the IS through the WHO primary standards in order to facilitate standardization of molecular monitoring in Latin America,” (Leukemia. 2016 Aug 19. doi: 10.1038/leu.2016.197).
In their study, the letter’s authors demonstrated that secondary reference biological calibrators anchored to the WHO primary standards can decrease inter-laboratory variability.
“Our results, together with those recently reported by Cross et al., substantiate the objective initially set during the establishment of the WHO primary standards, that is, to facilitate worldwide diffusion of the IS. For the first time in Latin America, this study provides a platform on which to assess the performance of distinct clinical BCR-ABL1 tests and confirm the utility of secondary reference materials to further improve IS accuracy and inter-laboratory precision,” they wrote, noting that their efforts will continue through provision of secondary reference material to centers involved in their project, as well as to potential new participants.
“Moreover, due to its higher precision and absolute quantification capability, we are evaluating the possibility of including digital PCR as the calibration method for the future,” they said.
The study discussed in the letter to the editor was supported by a grant from Novartis to one of the authors. Novartis also paid speaking fees to some of the authors. The study by Dr. Cross et al. was also funded by Novartis and some authors are employed by Novartis. The remaining authors, including Dr. Cross, reported having no disclosures.
A cell-based BCR-ABL1 secondary reference panel traceable to the World Health Organization BCR-ABL1 International Genetic Reference Panel – with an additional MR4.5 level – provides easier access to International Scale calibration and can act as a tool for assay optimization, validation, and quality assurance for molecular monitoring in chronic myeloid leukemia patients.
Such monitoring is important to disease management, especially for decision making with respect to treatment cessation. The new secondary reference panel would allow laboratories to circumvent the oft-used and time-consuming sample exchange process with reference laboratories for International Scale (IS) calibration, Nicholas C. P. Cross, PhD, of the University of Southampton (England) and colleagues wrote in an article published in Leukemia (2016 Jun 3;30:1844-52).
“The development of BCR-ABL1 tyrosine kinase inhibitors ... has enabled progressively deeper molecular responses in CML patients undergoing tyrosine kinase inhibitor therapy,” the authors wrote, noting that deeper molecular responses are “important milestones for patients considering treatment cessation,” and that “other landmarks on the IS also represent different treatment decision thresholds and prognostic outcomes.”
For this reason, regular molecular monitoring using real-time reverse-transcription quantitative PCR (RqPCR) is recommended for optimal disease management, they said.
“As treatment decisions are directly impacted by test results, accuracy and precision of BCR-ABL1 assays across the entire measurement range is crucial for patient management, especially in patients with deep molecular responses when considering possible treatment cessation,” they wrote.
Access to the material for the WHO BCR-ABL1 reference panel (MR1-MR4), which was developed in 2010 as a primary standard for BCR-ABL1 assay IS calibration, is limited, particularly for smaller laboratories, the authors said.
The secondary reference panel they developed, however, is “traceable to and faithfully replicates the WHO panel in both raw materials (lyopholized.K562 and HL-60 cell mixes) and manufacturing process, with the addition of a MR4.5 level.” The secondary panel was calibrated to IS using digital PCR against ABL1, BCR, and GUSB as reference genes, and was successfully evaluated by 45 different BCR-ABL1 assays at 44 different clinical laboratories in a multinational evaluation study.
The MR4.5 level was added to allow for more accurate IS calibration “as CML patients reaching this deep molecular response are increasingly being considered for treatment cessation,” the authors noted.
“Quality-control assessments indicated that the secondary panel had minimal residual moisture, excellent vial-to-vial homogeneity and greater than 2.5 years of real-time stability,” they said.
Further, the panel was successfully processed by all of the laboratories, indicating that it is compatible with many different BCR-ABL1 test configurations, they added.
Of note, the number of assays that achieved good precision and sensitivity exceeded the number that achieved good IS accuracy, suggesting an unmet need for “a simple and broadly available calibration mechanism, such as this secondary panel, to ensure IS accuracy is maintained in laboratories over time,” they wrote, concluding that such a panel “can provide easier access to IS calibration, as well as act as a tool for assay optimization, validation and quality assurance.”
In a letter to the editor in regard to the findings by Cross et al., Maria Sol Ruiz of Instituto Alexander Fleming in Buenos Aires, and colleagues wrote about their own development and validation of “secondary reference materials calibrated to the IS through the WHO primary standards in order to facilitate standardization of molecular monitoring in Latin America,” (Leukemia. 2016 Aug 19. doi: 10.1038/leu.2016.197).
In their study, the letter’s authors demonstrated that secondary reference biological calibrators anchored to the WHO primary standards can decrease inter-laboratory variability.
“Our results, together with those recently reported by Cross et al., substantiate the objective initially set during the establishment of the WHO primary standards, that is, to facilitate worldwide diffusion of the IS. For the first time in Latin America, this study provides a platform on which to assess the performance of distinct clinical BCR-ABL1 tests and confirm the utility of secondary reference materials to further improve IS accuracy and inter-laboratory precision,” they wrote, noting that their efforts will continue through provision of secondary reference material to centers involved in their project, as well as to potential new participants.
“Moreover, due to its higher precision and absolute quantification capability, we are evaluating the possibility of including digital PCR as the calibration method for the future,” they said.
The study discussed in the letter to the editor was supported by a grant from Novartis to one of the authors. Novartis also paid speaking fees to some of the authors. The study by Dr. Cross et al. was also funded by Novartis and some authors are employed by Novartis. The remaining authors, including Dr. Cross, reported having no disclosures.
FROM LEUKEMIA
Key clinical point:
Major finding: The secondary panel was calibrated to IS using digital PCR against ABL1, BCR, and GUSB as reference genes, and was successfully evaluated by 45 different BCR-ABL1 assays at 44 different clinical laboratories.
Data source: A multicenter evaluation study of a secondary reference panel for BCR-ABL1 quantification on the IS.
Disclosures: The study discussed in the letter to the editor was supported by a grant from Novartis to one of the authors. Novartis also paid speaking fees to some of the authors. The study by Dr. Cross et al. was also funded by Novartis and some authors are employed by Novartis. The remaining authors, including Dr. Cross, reported having no disclosures.
Preservation of the Anterior Cruciate Ligament: A Treatment Algorithm Based on Tear Location and Tissue Quality
Injury of the anterior cruciate ligament (ACL) is very common with over 200,000 annual injuries in the United Status.1,2 There is a general consensus that these injuries should not be treated conservatively in patients that are younger, or who wish to remain active.3,4 Reconstructive surgery is currently the preferred treatment in these patients, and anatomic single-bundle reconstruction with autografts is considered the gold standard.5,6
Reconstruction of the ACL is, however, not a perfect treatment. Following single-bundle autograft reconstruction, revision rates of 3% to 8%,6-9 contralateral injury rates of 3% to 8%,10,11 and infection rates of 0.5% to 3%7,12,13 have been reported. Furthermore, due to the invasive nature of graft harvesting and the surgical procedure, 10% to 25% of the patients are not satisfied following ACL reconstruction.14,15 This can often be explained by common complaints, such as anterior knee pain (13%-43%), kneeling pain (12%-54%), quadriceps muscle atrophy (20%-30%),16,17 and loss of range of motion (ROM) (12%-23%).7,9,18,19 Furthermore, as a result of the invasive nature of reconstructive surgery, revisions can be difficult due to complications, such as tunnel widening, tunnel malpositioning, and preexisting hardware.20-22 This can lead to inferior outcomes and higher rates (13%) of revision surgery compared to primary reconstruction.23-26 Finally, reconstructive surgery does not restore native kinematics of the ACL,27-29 which may partially explain why reconstructive surgery has not been shown to prevent osteoarthritis.28-31
Over the past decades, there has been an increasing interest in the preservation of the ACL in an attempt to ameliorate these issues.32-37 Ligament preservation focuses on preserving the native tissues and biology, while minimizing the surgical morbidity to the patients.
Some authors have recently reported on arthroscopic primary repair of proximal ACL tears in which the ligament is reattached onto the femoral wall using modern-day suture anchor technology.32,38 Others have augmented this repair technique with an internal brace39,40 or with a synthetic device.33,41 When performing primary repair, it is believed that proprioception is maintained,42-44 while experimental studies have suggested that primary repair also restores the native kinematics,45 and may prevent osteoarthritis.46 Furthermore, primary repair is a conservative approach in that no grafts need to be harvested, no tunnels need to be drilled, and revision surgery, if necessary, is more analogous to primary reconstructions.32In patients with partial tears, some surgeons have advocated preserving the anteromedial (AM) or posterolateral (PL) bundle and performing selective single-bundle augmentation.34,35 In addition, several authors have used remnant tensioning36,47 or remnant preservation37,48 in combination with reconstructive surgery in order to benefit from the biological characteristics of the remnant. These techniques lead to better proprioceptive function,44,49,50 vascularization and ligamentization of the graft,50-52 provide an optical guide for anatomic tunnel placement,53 and decrease the incidence of tunnel widening.54,55The feasibility and applicability of these surgical techniques mainly depends on the tear type and tissue quality of the torn ligament. In this article we (I) discuss the history of ACL preservation, (II) discuss how modern advances alter the risk-benefit ratio for ACL preservation, and (III) propose a treatment algorithm for ACL injuries that is based on tear location and tissue quality.
History of ACL Preservation
The history of the surgical treatment of ACL injuries started in 1895 when Robson56 treated a 41-year-old male who tore both cruciate ligaments from the femoral wall. Performing primary repair with catgut ligatures, both cruciate ligaments were preserved and the patients had resolution of pain symptoms and full function at 6-year follow-up. Over the following decades, Palmer57,58 and O’Donoghue59,60 further popularized open primary repair for the treatment of ACL injuries, and this technique was the most commonly performed treatment in the 1970s and early 1980s.61-65 The initial short-term results of primary repair were excellent,61,62 but Feagin and Curl66 were the first to note that the results deteriorated at mid-term follow-up. Despite improvements in the surgical technique of repairing the ACL, such as the usage of nonabsorbable sutures and directly tying the sutures over bone,63,67 the results remained disappointing at longer-term follow-up.68-70
In response to these disappointing results, surgeons sought to improve the surgical treatment by either augmenting the primary repair with a semitendinosus, a patella tendon graft or an augmentation device,71-74 or by performing primary reconstruction.75-77 At the end of the 1980s and early 1990s, several randomized and prospective clinical trials were performed in order to compare the outcomes of these techniques.74,78-82 Many studies showed that results of augmented repair were more reliable when compared to primary repair, which led to the abandonment of primary repair in favor of augmented repair, and eventually primary reconstruction.65
The Important Role of Tear Location in Ligament Preservation
When taking a closer look at the outcomes of primary repair and augmented repair, it seems that the results of these preservation techniques were not as disappointing as was suggested. This can be explained, in large part, by the fact that the important roles of tear location and tissue quality were not widely recognized.
Sherman and colleagues70 reported in 1991 their mid-term results of open primary repair. Similar to others, they noted a deterioration of their results at mid-term follow-up. However, they uniquely performed an extensive subgroup analysis in order to find an explanation for this. In their study, considered a landmark paper on primary repair,65,70 they concluded that, “poor tissue quality is typical for midsubstance tears and that a repair of these injuries will predictably fail while type I tears (proximal), with better tissue quality, show a definite trend towards better results.”70 With these findings, they confirmed the findings of others that had recognized a trend of better outcomes with proximal tears.64,67,83-85
A majority of the historical studies that were published before 1991 had not considered the role of tear location and tissue quality on outcomes of open primary repair. This was also true for the aforementioned randomized studies that compared primary repair with augmented repair and primary reconstruction. Because these studies randomized patients and did not take tear location into account, it can be expected that patients with midsubstance tears were included in the cohorts of primary repair and the outcomes of these studies were therefore confounded.74,78-82 If these studies would have been aware of the role that tear location plays on primary repair outcomes, different outcomes may have been found and different conclusions on the optimal treatment for different tear types may have been drawn.86
Open Primary ACL Repair Outcomes Stratified by Tear Location
When reviewing the literature of open primary repair outcomes stratified by tear location, it is noted that multiple studies reported excellent outcomes following primary repair of proximal ACL tears.73,83,84,87-90 Weaver and colleagues64 were among the first to stratify their results by tear location, and they found that more patients with proximal tears (52 of 66; 79%) were satisfied after the procedure when compared to patients with midsubstance tears (3 of 13; 23%) at 3.5-year follow-up. They concluded that, “selection can be made with some predictability of the type of injury to the ligament as to which patients will do better.”64 Kühne and colleagues89 reported the outcomes of 75 patients with proximal tears treated with open primary repair and noted no failures, negative pivot shift in 88% of patients, stable or nearly stable Lachman test in 87% of patients, and 89% return to sports rate at 4-year follow-up. Raunest and colleagues91 reported a negative pivot shift and negative anterior drawer test in 84%, return to sports in 71%, and satisfaction in 75% of 51 patients that underwent open primary repair of proximal tears at 3.5-year follow-up.
Interestingly, and in contrast to the findings of Feagin and Curl,66 no deterioration of the outcomes at mid-term follow-up was noted in patients with proximal tears. Genelin and colleagues88 reported their results of 42 patients with proximal tears treated with open primary repair at 5- to 7-year follow-up. They found a negative pivot shift in 81%, stable or nearly stable Lachman test in 81%, and patient satisfaction in 86% of patients. Similarly, Bräm and colleagues87 found good results at mid-term follow-up with a good-excellent Lysholm score in 79%, return to a similar level of sports in 76%, stable or nearly stable Lachman test in 91%, and anterior drawer test in 94% of patients, along with an 88% satisfaction rate and 7% failure rate in patients who underwent open primary repair of proximal tears.
On the contrary, when the outcomes of studies that performed open primary repair in mainly, or only, patients with midsubstance tears are reviewed, significantly inferior results are found. Frank and colleagues92 reported outcomes in 42 patients with midsubstance tears at 4-year follow-up. They reported that 56% had a stable or nearly stable anterior drawer test, 78% had a positive pivot shift, and that only 61% were satisfied with the procedure. Odensten and colleagues78 reported outcomes of open primary repair in a subgroup of 22 patients with midsubstance tears at 1.5-year follow-up, and noted a 14% failure rate.
When reviewing the mid-term results in patients with midsubstance tears, it seems that there was more deterioration in outcomes.69,70 Firstly, the aforementioned study by Sherman and colleagues70 showed poor results in the patients with (type IV) midsubstance tears at mid-term follow-up. Furthermore, Kaplan and colleagues69 reported the mid-term outcomes of 70 patients, of which 56 patients had midsubstance tears. After having reported good outcomes at short-term follow-up,63,67 they noted that 42% of patients had >3 mm anteroposterior stability when compared to the contralateral leg, only a 62% return to sport rate, and a 17% failure rate. They concluded that, “Although … primary repair of the anterior cruciate may work in some patients, it is an unpredictable operative procedure.”
These studies showed that the outcomes of open primary repair were significantly better in patients with proximal ACL tears and sufficient tissue quality when compared to midsubstance tears. This suggests that open primary ACL repair may have been prematurely abandoned as a treatment option for patients with proximal tears.
Augmented ACL Repair
There were several reasons why augmented repair became the preferred treatment in the early and mid 1990s. First of all, the results of augmented repair were more consistent compared to primary repair in the aforementioned randomized and prospective studies,74,78-82 which is not surprising given the fact that the role of tear location was not widely recognized at the time. Secondly, in the 1970s and early 1980s, patients were treated postoperatively in a cast for 6 weeks, which led to problems, such as loss of ROM, pain, and decreased function.93,94 At the end of the1980s and 1990s, the focus shifted from prolonged joint immobilization towards early postoperative ROM.95-97 Since many authors believed that primary repair of the ACL was not strong enough to tolerate early mobilization, an augmentation was added to the technique in order to fortify the repair and enable early ROM.98
Interestingly, augmented repair, which is essentially a combination of primary ACL repair and ACL reconstruction, was mainly performed in the 1990s and many surgeons did recognize the role of tear location in this treatment at this point.73,98-103 In these years, the treatment algorithm consisted of augmented ACL repair in patients with proximal tears in the acute setting and ACL reconstruction in patients with midsubstance or chronic tears. Several different augmentation techniques were used to reinforce the primary repair in these years including autograft tissues (semitendinosus tendon,102-104 patellar tendon,100 or iliotibial band [ITB]105) synthetic materials (polydioxanone [PDS],101,102,106 carbon fibre,74 and polyester [Trevira]97), augmentation devices (Kennedy Ligament Augmentation Device [LAD]98-100) and extra-articular augmentations.73
When reviewing the outcomes of augmented repair of the ACL, good to excellent results can be found in studies that used this technique in patients with proximal tears.73,98-106 Kdolsky and colleagues98 were in one of the first groups that reported their results of augmented repair in only patients with proximal tears. In 1993, they reported their mid-term outcomes (5 to 8 years) in 66 patients who underwent primary repair and augmentation with the Kennedy LAD and found that 97% of patients had stable knees (<3 mm on KT-1000 examination), 98% had a negative pivot shift, and 76% returned to previous level of sports. However, often-reported problems with the augmentation devices were found in this study with rupture of the device (12%) and decreased ROM (14%).98 In 1995, Grøntvedt and Engebretsen100 compared augmentation with the Kennedy LAD to patellar tendon augmentation in a randomized study of patients with acute proximal tears. They noted that 50% of the patients in the Kennedy LAD group had a positive pivot shift compared to 23% in the patellar tendon group. Furthermore, they found KT-1000 leg differences of <3 mm in 92% of the patellar tendon group and 54% of the Kennedy LAD group. Because the authors found significant differences between both groups at 1- and 2-year follow-up, they stopped the clinical trial.
Several authors in the following years reported good results of augmented repair using autograft tissues. Natri and colleagues105 reported the outcomes of 72 patients treated with primary repair of proximal tears augmented with the ITB at 3.5-year follow-up. They found 89% negative pivot shift rate, 93% stable or nearly stable Lachman test, 99% stable or nearly stable anterior drawer test, 79% satisfaction rate, and 91% return to previous level of sports rate. Krueger-Franke and colleagues104 reported the outcomes of primary repair of proximal tears with augmentation using the semitendinosus tendon. In a retrospective study of 76 patients, they noted that 96% of patients had a negative pivot shift, 75% of patients had stable or nearly stable Lachman test, 93% were satisfied with the procedure, a mean Lysholm score of 92, a Tegner score that only decreased from 7.2 to 7.1, and KT-1000 testing with 78% <4 mm leg difference with the contralateral leg. The authors concluded that patients with femoral ruptures could be treated with augmented repair when performed in the acute setting. As this study was published in 1998, they stated that magnetic resonance imaging and arthroscopy could be helpful in identifying the tear location.
Final Abandonment of ACL Preservation
Reviewing these outcomes raises the question as to why these techniques were ultimately abandoned in the treatment algorithm of proximal ACL injuries, especially given the aforementioned advantages of ACL preservation. One of the possible answers can be found in a landmark study on ACL reconstruction and rehabilitation published by Shelbourne and colleagues107 in 1991. At that time, arthrofibrosis and knee stiffness were frequently reported problems following ACL surgery, which could partially be explained by the standard conservative rehabilitation using postoperative joint immobilization.67,70,80,88
Shelbourne and colleagues107 aimed to assess the cause of arthrofibrosis and knee stiffness, and divided the patients into groups by number of days between injury and surgery (<7, 7 to 21 days, and >21 days between injury and surgery). Furthermore, patients within these groups underwent either a conventional or accelerated rehabilitation program. The authors not only found that patients undergoing accelerated rehabilitation had less arthrofibrosis, but they also noted that less arthrofibrosis was seen when surgery was delayed. These findings, however, contrasted with the general perception that the ACL should be repaired in the first 3 weeks postinjury to ensure optimal tissue quality with an augmented approach. As a result, the treatment of ACL injuries shifted towards ACL reconstruction after these findings. Krueger-Franke and colleagues104 commented on the trend after the study of Shelbourne and colleagues:107 “Less consideration has been given to the importance of the proprioceptive receptors in the tibial remnants of the torn ACL and the value of their preservation as part of a primary reconstruction.”
In addition to the trend away from an augmented repair approach due to the novel understanding of the importance of early mobilization, some discussion should focus on the technical limitations of arthroscopy at that time. While arthroscopy had been around for several decades, fluid management and arthroscopic instrumentation was slow to develop. All of the repair and augmentation techniques previously discussed had been performed via an open arthrotomy. Arthroscopic technologies of the time were not refined enough to enable surgeons to perform such complex, intra-articular techniques that would enable suturing of the ligament remnant. In this regard, arthroscopic ACL reconstruction was a much simpler technique to accomplish, and this also likely contributed to the final abandonment of the ligament preservation approach.
Role for ACL Preservation with Modern Advances
As stated in the introduction, there has been a recent resurgence of interest in preservation of the native ligament.32-37 With the passage of time, many technologic advances have been made, which has allowed surgeons to reconsider the concept of ligament preservation.
First of all, appropriate patient selection was not applied historically, as the critical factors of tear location and tissue quality were not recognized in the era of open primary repair. In modern days, however, advances such as MRI have been developed, which can give the surgeon an idea of the status, and tear type of the ACL pre-operatively.108 This may help the orthopaedic surgeon to plan the surgery and make an assessment as to whether ACL preservation is possible. Secondly, in the historic literature the postoperative regimen consisted of casting for 5 or 6 weeks,67,70,80,88 while the focus later shifted towards early ROM.95-97Modern day ACL rehabilitation focuses on immediate ROM to avoid the complications stiffness, pain and decreased function that plagued the outcomes when immobilization was used.93,94 Thirdly, historically small tunnels were drilled with primary repair and sutures had to be tied over bone,57,67 whereas currently suture anchors are available that prevent the need for tunnel drilling and enable direct suture tensioning.32,38 Finally, and most importantly, in the historic literature patients were treated with an invasive arthrotomy technique, while modern day arthroscopic techniques readily enable the surgeon to effectively suture the remnant arthroscopically. Interestingly, in 2005, in their 20-year follow-up of primary repair surgeries, Strand and colleagues109 stated, “if the same results could be accomplished by a smaller, arthroscopic procedure, primary repair might reduce the number of patients needing later reconstructions with small ‘costs’ in the way of risk and inconvenience for the patients. We therefore believe that further research and development of methods for closed (arthroscopic) repair are justified.”
Altered Risk-Benefit Ratio
Historically, the treatments of open primary repair and open ACL reconstruction were both invasive surgeries with an arthrotomy, drilling of bone tunnels, and postoperative joint immobilization for 4 to 6 weeks. However, with the modern-day advances, the risk-benefit ratio of both treatments has changed, as Strand and colleagues109 had already suggested. Although ACL reconstruction can be performed arthroscopically, it remains an invasive procedure, in which tunnels are drilled, patellar tendons or hamstring tendons are harvested, and complications, such as knee pain and quadriceps atrophy, are common. The surgery of primary ACL repair, however, has benefited significantly from the modern developments.32,38 Primary ACL repair can now be performed arthroscopically, and by using suture anchors no tunnels need to be drilled and the remnant can be tensioned directly. An additional benefit of the use of suture anchors is that revision surgery of a failed primary repair is analogous to primary reconstruction, whereas revision surgery of a failed ACL reconstruction can be problematic due to tunnel widening, tunnel malpositioning, and preexisting hardware.20-22
Reviewing the differences between arthroscopic primary ACL repair and ACL reconstruction, it becomes clear that primary repair has benefited significantly from the modern advances and that the risk-benefit ratio for primary repair has been altered. This means that patients with proximal tears can be treated with a relatively straight forward, minimally invasive surgery, which has been shown to be effective in 85% to 90% of patients.32,38
Treatment Algorithm Based on Tear Location
Since 2008, in the practice of the senior author (GSD), the surgical treatment algorithm for ACL injuries is completely based on the tear location and tissue quality of the ligament.110,111 To describe the different tear types, we use the modified Sherman classification in which we extended his classification towards the tibial side whereas Sherman and colleagues70 only described the femoral side of the tears (Figures A-F, Table).
Type I Tears: Primary Repair
Type I tears are soft tissue avulsion type tears that can be easily treated with arthroscopic primary repair.107 The length of the distal remnant has to be at least 90% and the tissue quality has to be good to excellent in order to approximate the remnant towards the femoral wall (Table).112 The incidence of type I tears was 26% in the study of Sherman and colleagues,70 although recent studies showed a lower incidence (6% to 10%) in a larger population.32,38 Certainly, individual practices will see different percentages of type I tears based upon the mix of injury mechanisms they see most frequently. Over the last 2 years, with the recognition of the importance of tear type and tissue quality, there has been a renewed interest in arthroscopic primary ACL repair.32,38
DiFelice and colleagues32 were the first to arthroscopically perform primary repair of the ACL in proximal tears using suture anchors. They reported the outcomes of the first 11 consecutive patients that underwent primary repair in a previously described technique.113 At mean 3.5-year follow-up, they noted only 1 failure (9%) due to re-injury; mean Lysholm score of 93.2; mean modified Cincinnati score of 91.5; pre- and postoperative Tegner score of 7.3 and 6.9, respectively; SANE score of 91.8; and subjective International Knee Documentation Committee (IKDC) score of 86.4. Of the patients with an intact repair, 9 patients had an objective IKDC rating A and 1 patient had B and all patients had KT-1000 leg differences of <3 mm with the contralateral side (three patients were not available for KT-1000 testing). The authors concluded that arthroscopic primary ACL repair could achieve short-term clinical success in a selected group of patients with proximal avulsion tears and excellent tissue quality. They further noted that mid-term outcomes are necessary given that the results of open primary repair deteriorated at longer-term follow-up in the historical literature. Recently, the senior author (GSD) has added an Internal Brace (Arthrex) to the primary repair with the goal of protecting the ligament in the first weeks to further promote healing of the ligament.39,40,114
More recently, Achtnich and colleagues38 compared the treatment of arthroscopic primary ACL repair with primary ACL reconstruction in 41 patients with type I tears at 2.3-years follow-up. Twenty-one patients consented for primary repair while 20 patients declined this procedure and underwent primary reconstruction. They noted no significant differences in Lachman test, pivot shift test, objective IKDC score, and KT-1000 scores. Although not significant, the clinical failure rate in the primary repair group (15%) was higher than the reconstruction group (0%). Interestingly, despite the higher failure rate in the repair group, the authors concluded that primary ACL repair is recommended in a carefully selected group of patients with type I tears and excellent tissue quality, which can likely be explained by the differences in the risk-benefit ratio between both procedures.
Over the last decade, the research group led by Murray46,115,116 has performed experimental research on primary repair with a biological scaffold and reported many interesting findings that could be extrapolated to primary ACL repair. First of all, they compared bioenhanced primary repair with bioenhanced primary reconstruction in 64 Yucutan pigs and noted that there was significantly less macroscopic cartilage damage in the primary repair group at 1-year follow-up.46 They concluded that bioenhanced ACL repair may provide a new, less invasive treatment option that reduces cartilage damage following joint injury. This may suggest that primary repair may have a lower incidence of osteoarthritis when compared to ACL reconstruction, which is interesting as osteoarthritis is very common after ACL reconstruction. Further research in this area is certainly warranted.
In another study they compared bioenhanced primary repair in juvenile, adolescent and mature Yucutan pigs and noted that functional healing depended on the level of skeletal maturity with immature animals having a more productive healing response.116 This indicates that primary repair might be a good treatment option in skeletally immature patients, especially since reconstruction increases the risk of premature closure of the epiphysis117,118 and delaying treatment increases the risk of meniscus injury.119 Interestingly, a recent meta-analysis showed indeed that the risk of epiphysis closure was lower in primary repair when compared to ACL reconstruction and the rupture rate was also lower.118 Primary repair may be a good treatment option in children as the procedure has all the attributes that should be applicable to children: it is minimally morbid, tissue sparing, and it is a conservative approach that does not burn any surgical bridges for future reconstructive surgery if necessary.
Finally, the research group of Murray115 assessed the effect of surgical delay of primary repair following injury in Yucutan pigs and noted that better biomechanical outcomes were noted after delaying surgery for 2 weeks when compared to 6 weeks. This suggests that primary repair should preferably be performed in the acute setting, which has also been shown in historical studies since the ligament in the acute setting has optimal tissue quality and the ligament is less likely to be retracted or reabsorbed.59,60,115
One Bundle Type I Tears: Single Bundle Augmented Repair
In some cases, the tear locations of the AM and PL bundle are not at the same location and Zantop and colleagues120 reported in an arthroscopic study that this could be as frequent as in 30% of all complete tears. In some of these tears, one of the bundles can be avulsed of the femoral wall (type I tear) while the other bundle is not directly repairable (non-type I tear). In these cases, the senior author (GSD) will repair the type I tear bundle, whereas a hamstring augmentation is placed at the location of the other bundle. When reviewing the literature, a combination of primary repair of one bundle and reconstruction of the second bundle has not been described before. However, over the last decade several surgeons have performed augmentation of one bundle in the setting of partial tears.34,35,121-124
Buda and colleagues34 were the first to perform selective AM or PL bundle reconstruction in the setting of partial tears.34 At 5-year follow-up, they reported no reruptures and only 1 patient with an IKDC C-score, although reoperation was necessary in 4 out of 47 patients (9%). Following this publication, many others reported on selective bundle reconstruction.35,121-124 However, with partial tears, the knee is often stable and a selective augmentation technique is utilized to prevent complete rupture of the ligament. The application of this technique is essentially different from reconstruction for complete ACL tears in which the knee is unstable, there is a giving way sensation and patients have problems participating in sports.
Type II Tears: Augmented Repair
Type II tears often have good or excellent tissue quality and can be pulled up towards the femoral footprint, but are too short to be firmly attached. Sherman and colleagues70 reported that approximately 22% patients had a type II tear, which corresponds to a tear located in the proximal part of the ligament. With this technique, multiple suture passes are used to stitch the remnant and, in addition, a smaller hamstring autograft or allograft is passed through the middle of the tibial remnant. A suture button is used proximally for the graft, and the tensioning repair sutures through the remnant are also passed through the suture button. The suture button is passed through the femoral tunnel and flipped so that the graft is proximally fixed. Then, the repair sutures of the remnant are tensioned, and the ligament is pulled towards the femoral wall as a sleeve around the graft. When the ligament is approximated to the femoral wall, the sutures are tied over the suture button. The graft is then tensioned distally to complete the augmented repair.
In the recent literature, the technique of augmentation of a primary repair using autograft tissue has not been reported. However, augmented repair using an internal brace39,40 or augmentation devices33,41 have been recently performed. MacKay and colleagues39 reported good outcomes of arthroscopic primary repair of proximal tears using an internal brace. Eggli and colleagues33 reported the results of the first 10 patients treated with ACL preservation using primary repair of the ligament with the addition of a dynamic screw-spring mechanism. The authors reported good preliminary results with one failure (10%) and good objective and subjective outcomes. In a next study, they reported the outcomes of 278 patients and although they reported good clinical outcomes and a revision rate of 4%, the reoperation rate for removal of the screw-spring mechanism was high (24%).41 This is not surprising when reviewing the historical literature in which high complication rates of the augmentation devices were reported.99,100 We were unable to identify any other studies reporting surgical techniques of augmenting primary repair in the literature.
Type III Tears: Reconstruction With Remnant Tensioning
In patients with type III tears, the ligament cannot be approximated to the wall and reconstruction is necessary in order to restore knee stability. However, in these cases the ligament has sufficient length (25%-75%) and can be tensioned along or around the graft. Preservation of the ligament remnant has several (theoretical) advantages, such as better proprioceptive function,42,49,50 vascularization and ligamentization of the graft,50-52 an optical guide for anatomic tunnel placement,53 and a decreased incidence of tunnel widening.54,55 Furthermore, tensioning of the remnant is thought to lower the risk of cyclops lesions when compared to remnant preservation.125 Although the difference between augmented repair and remnant tensioning seems small, the purpose of surgery is different. With augmented repair, the ligament can be approximated close to the femoral wall and the goal of surgery is to use the healing capacity that the ACL has in the proximal part of the ligament,126 while with remnant tensioning the goal is only to benefit from some of the aforementioned advantages. Ahn and colleagues36 were the first to perform this technique and stated, “Our concept is that the remnant tissue has only an additive effect.” Furthermore, with augmented repair multiple sutures are passed through the AM and PL bundle in order to sufficiently approximate the ligament to the femoral wall, while with the remnant tensioning technique generally one or a few sutures or lasso loop are passed through the proximal part to tension the ligament, prevent sagging of the remnant, and decrease the risk of cyclops lesions.127,128
Several authors have recently performed remnant tensioning during ACL.36,47,125-127 Ahn and colleagues47 reported excellent objective and subjective outcomes following this procedure and found that with re-arthroscopy nearly all patients had fair synovialization of the graft. Others have reported similarly good outcomes of these techniques.125,129,130 However, studies comparing this treatment with normal ACL reconstruction and assessing outcomes, failure rates and proprioception are lacking.
Type IV Tears: Reconstruction With Remnant Preservation
Finally, in some patients the ligament is torn distally or the tissue quality is not optimal. In these patients, the remnant can be debrided to the part of good tissue quality in order to preserve the biology and minimize the risk for cyclops lesions. A standard reconstruction needs to be performed to restore the instability, but by preserving the remnant, advantages, such as proprioception,44,49,50 graft vascularization,50-52 an optical guide for tibial tunnel placement,53 and a decreased incidence of tunnel widening54,55 can be expected.
Lee and colleagues37 presented the tibial remnant technique in which standard reconstruction was performed, and the tibial tunnel was drilled through the center of the remnant. In a later study, they compared remnant preservation with a remnant of <20% of the total ACL length with >20% of the length and found that proprioception was better with more remnant volume.48 Similarly, Muneta and colleagues131 assessed the role of remnant length and found that remnant length is positively correlated with better stability measured on KT-1000 anteroposterior stability.
Several studies compared ACL reconstruction with remnant preservation vs conventional ACL reconstruction.52,54,129 Takazawa and colleagues52 performed a retrospective study of 183 patients and found that patients in the remnant preservation group had significantly better KT-2000 stability, while they also reported a significantly lower graft rupture rate in this group (1.1% vs 7.1%) at 2-year follow-up. Hong and colleagues129 performed a randomized clinical trial of 80 patients and did not find these differences, although there was a trend towards higher Lysholm scores in the remnant preservation group. Finally, Zhang and colleagues54 performed a randomized clinical trial and found a lower incidence and amount of tibial tunnel widening in the preserving-remnant group when compared to the removing-remnant group. These studies show that there is likely a role for remnant preservation.
Type V Tears: Primary Repair
In some patients, the ligament is torn in the distal 10% of the ligament, which can occur as a distal avulsion tear or as a distal bony avulsion fracture.132 Bony avulsion fractures are most commonly seen in children whereas true distal soft tissue avulsion tears are very rare.132
Treatments of these tear types include antegrade screw fixation, pullout sutures or the use of suture anchors in case of bony avulsion fractures and pullout sutures with tying over a bony bridge or ligament button in case of soft tissue avulsions. Leeberg and colleagues132 recently performed a systematic review of all studies reporting on treatment of distal avulsion fractures.They noted that most treatments were currently performed arthroscopically and that outcomes were generally good. Another recent biomechanical study compared antegrade screw fixation with suture anchor fixation and pullout suture fixation.133 The authors noted that suture anchor fixation has slightly less displacement of the bony fragment when compared to screw fixation and pull-out sutures, and that the strength to failure was higher in the suture anchor fixation when compared to the pullout suture fixation. The outcomes of this study suggest that screw fixation and suture anchor fixation might be superior to pullout suture fixation, which might be interesting as with pullout suture fixation the ligament cannot be directly tensioned to the tibial footprint, which can lead to anteroposterior laxity.132 Clinical studies are necessary to assess the preferred treatment in these tear types but it seems that screw fixation is preferred in large bony avulsion fractures, while suture anchor fixation or pullout suture fixation can be used for soft tissue avulsion tears.
Complex Tears or Poor Tissue Quality: Reconstruction
If the tear is complex, multiple tears are present, or the tissue quality is poor, then preservation of the ligament is not possible, and in these cases a standard reconstruction should be performed.
Conclusion
When reviewing the literature of ACL preservation, it becomes clear that the evolution of surgical treatment of ACL injuries was biased. Preservation of the native ligament has many advantages, such as better proprioception, graft vascularization, an optical guide for tibial tunnel placement, and a decreased incidence of tunnel widening that can be expected. Furthermore, arthroscopic primary ACL repair is minimally invasive and does not burn any bridges for future reconstructions, if necessary. This is in addition to the other (theoretical) advantages of primary repair, such as restoration of native kinematics and a decreased risk of osteoarthritis. Modern advances have significantly changed the risk-benefit ratio that should make us reconsider ACL preservation approaches. Certainly, further research in this area is warranted. In this article we have presented a treatment algorithm for ACL preservation, which is based on tear location and remnant tissue quality.
Am J Orthop. 2016;45(7):E393-E405. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Mall NA, Chalmers PN, Moric M, et al. Incidence and trends of anterior cruciate ligament reconstruction in the United States. Am J Sports Med. 2014;42(10):2363-2370.
2. Sanders TL, Maradit Kremers H, Bryan AJ, et al. Incidence of anterior cruciate ligament tears and reconstruction: a 21-year population-based study. Am J Sports Med. 2016;44(6):1502-1507.
3. Ciccotti MG, Lombardo SJ, Nonweiler B, Pink M. Non-operative treatment of ruptures of the anterior cruciate ligament in middle-aged patients. Results after long-term follow-up. J Bone Joint Surg Am. 1994;76(9):1315-1321.
4. Sanders TL, Pareek A, Kremers HM, et al. Long-term follow-up of isolated ACL tears treated without ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2016 May 24. [Epub ahead of print]
5. Irarrázaval S, Kurosaka M, Cohen M, Fu FH. Anterior cruciate ligament reconstruction. J ISAKOS. 2016;1(1):38-52.
6. Gabler CM, Jacobs CA, Howard JS, Mattacola CG, Johnson DL. Comparison of graft failure rate between autografts placed via an anatomic anterior cruciate ligament reconstruction technique: a systematic review, meta-analysis, and meta-regression. Am J Sports Med. 2016;44(4):1069-1079.
7. Li S, Chen Y, Lin Z, Cui W, Zhao J, Su W. A systematic review of randomized controlled clinical trials comparing hamstring autografts versus bone-patellar tendon-bone autografts for the reconstruction of the anterior cruciate ligament. Arch Orthop Trauma Surg. 2012;132(9):1287-1297.
8. Rahr-Wagner L, Thillemann TM, Pedersen AB, Lind M. Comparison of hamstring tendon and patellar tendon grafts in anterior cruciate ligament reconstruction in a nationwide population-based cohort study: results from the danish registry of knee ligament reconstruction. Am J Sports Med. 2014;42(2):278-284.
9. Xie X, Liu X, Chen Z, Yu Y, Peng S, Li Q. A meta-analysis of bone-patellar tendon-bone autograft versus four-strand hamstring tendon autograft for anterior cruciate ligament reconstruction. Knee. 2015;22(2):100-110.
10. Andernord D, Desai N, Björnsson H, Gillén S, Karlsson J, Samuelsson K. Predictors of contralateral anterior cruciate ligament reconstruction: a cohort study of 9061 patients with 5-year follow-up. Am J Sports Med. 2015;43(2):295-302.
11. Maletis GB, Inacio MC, Funahashi TT. Risk factors associated with revision and contralateral anterior cruciate ligament reconstructions in the Kaiser Permanente ACLR registry. Am J Sports Med. 2015;43(3):641-647.
12. Kim SJ, Postigo R, Koo S, Kim JH. Infection after arthroscopic anterior cruciate ligament reconstruction. Orthopedics. 2014;37(7):477-484.
13. Makhni EC, Steinhaus ME, Mehran N, Schulz BS, Ahmad CS. Functional outcome and graft retention in patients with septic arthritis after anterior cruciate ligament reconstruction: a systematic review. Arthroscopy. 2015;31(7):1392-1401.
14. Kocher MS, Steadman JR, Briggs K, Zurakowski D, Sterett WI, Hawkins RJ. Determinants of patient satisfaction with outcome after anterior cruciate ligament reconstruction. J Bone Joint Surg Am. 2002;84-A(9):1560-1572.
15. Ardern CL, Österberg A, Sonesson S, Gauffin H, Webster KE, Kvist J. Satisfaction with knee function after primary anterior cruciate ligament reconstruction is associated with self-efficacy, quality of life, and returning to the preinjury physical activity. Arthroscopy. 2016;32(8):1631-1638.e3.
16. Grant JA, Mohtadi NG, Maitland ME, Zernicke RF. Comparison of home versus physical therapy-supervised rehabilitation programs after anterior cruciate ligament reconstruction: a randomized clinical trial. Am J Sports Med. 2005;33(9):1288-1297.
17. Lindström M, Strandberg S, Wredmark T, Fell änder-Tsai L, Henriksson M. Functional and muscle morphometric effects of ACL reconstruction. A prospective CT study with 1 year follow-up. Scand J Med Sci Sports. 2013;23(4):431-442.
18. Biau DJ, Tournoux C, Katsahian S, Schranz PJ, Nizard RS. Bone-patellar tendon-bone autografts versus hamstring autografts for reconstruction of anterior cruciate ligament: meta-analysis. BMJ. 2006;332(7548):995-1001.
19. Spindler KP, Kuhn JE, Freedman KB, Matthews CE, Dittus RS, Harrell FE Jr. Anterior cruciate ligament reconstruction autograft choice: bone-tendon-bone versus hamstring: does it really matter? A systematic review. Am J Sports Med. 2004;32(8):1986-1995.
20. Aga C, Wilson KJ, Johansen S, Dornan G, La Prade RF, Engebretsen L. Tunnel widening in single- versus double-bundle anterior cruciate ligament reconstructed knees. Knee Surg Sports Traumatol Arthrosc. 2016 Jun 21. [Epub ahead of print]
21. Maak TG, Voos JE, Wickiewicz TL, Warren RF. Tunnel widening in revision anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2010;18(11):695-706.
22. Cheatham SA, Johnson DL. Anticipating problems unique to revision ACL surgery. Sports Med Arthrosc. 2013;21(2):129-134.
23. Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39(1):199-217.
24. Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531-536.
25. Andriolo L, Filardo G, Kon E, et al. Revision anterior cruciate ligament reconstruction: clinical outcome and evidence for return to sport. Knee Surg Sports Traumatol Arthrosc. 2015;23(10):2825-2845.
26. Grassi A, Ardern CL, Marcheggiani Muccioli GM, Neri MP, Marcacci M, Zaffagnini S. Does revision ACL reconstruction measure up to primary surgery? A meta-analysis comparing patient-reported and clinician-reported outcomes, and radiographic results. Br J Sports Med. 2016;50(12):716-724.
27. Ristanis S, Stergiou N, Patras K, Vasiliadis HS, Giakas G, Georgoulis AD. Excessive tibial rotation during high-demand activities is not restored by anterior cruciate ligament reconstruction. Arthroscopy. 2005;21(11):1323-1329.
28. Andriacchi TP, Mündermann A, Smith RL, Alexander EJ, Dyrby CO, Koo S. A framework for the in vivo pathomechanics of osteoarthritis at the knee. Ann Biomed Eng. 2004;32(3):447-457.
29. Imhauser C, Mauro C, Choi D, et al. Abnormal tibiofemoral contact stress and its association with altered kinematics after center-center anterior cruciate ligament reconstruction: an in vitro study. Am J Sports Med. 2013;41(4):815-825.
30. Ajuied A, Wong F, Smith C, et al. Anterior cruciate ligament injury and radiologic progression of knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2014;42(9):2242-2252.
31. Chalmers PN, Mall NA, Moric M, et al. Does ACL reconstruction alter natural history?: A systematic literature review of long-term outcomes. J Bone Joint Surg Am. 2014;96(4):292-300.
32. DiFelice GS, Villegas C, Taylor SA. Anterior cruciate ligament preservation: early results of a novel arthroscopic technique for suture anchor primary anterior cruciate ligament repair. Arthroscopy. 2015;31(11):2162-2171.
33. Eggli S, Kohlhof H, Zumstein M, et al. Dynamic intraligamentary stabilization: novel technique for preserving the ruptured ACL. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):1215-1221.
34. Buda R, Ferruzzi A, Vannini F, Zambelli L, Di Caprio F. Augmentation technique with semitendinosus and gracilis tendons in chronic partial lesions of the ACL: clinical and arthrometric analysis. Knee Surg Sports Traumatol Arthrosc. 2006;14(11):1101-1107.
35. Ochi M, Adachi N, Uchio Y, et al. A minimum 2-year follow-up after selective anteromedial or posterolateral bundle anterior cruciate ligament reconstruction. Arthroscopy. 2009;25(2):117-122.
36. Ahn JH, Lee YS, Ha HC. Anterior cruciate ligament reconstruction with preservation of remnant bundle using hamstring autograft: technical note. Arch Orthop Trauma Surg. 2009;129(8):1011-1015.
37. Lee BI, Min KD, Choi HS, Kim JB, Kim ST. Arthroscopic anterior cruciate ligament reconstruction with the tibial-remnant preserving technique using a hamstring graft. Arthroscopy. 2006;22(3):340.e1-e7.
38. Achtnich A, Herbst E, Forkel P, et al. Acute proximal anterior cruciate ligament tears: outcomes after arthroscopic suture anchor repair versus anatomic single-bundle reconstruction. Arthroscopy. 2016 Jun 17. [Epub ahead of print]
39. MacKay G, Anthony IC, Jenkins PJ, Blyth M. Anterior cruciate ligament repair revisited. Preliminary results of primary repair with internal brace ligament augmentation: a case series. Orthop Muscul Syst. 2015;4:188.
40. Mackay GM, Blyth MJ, Anthony I, Hopper GP, Ribbans WJ. A review of ligament augmentation with the InternalBrace™: the surgical principle is described for the lateral ankle ligament and ACL repair in particular, and a comprehensive review of other surgical applications and techniques is presented. Surg Technol Int. 2015;26:239-255.
41. Henle P, Röder C, Perler G, Heitkemper S, Eggli S. Dynamic intraligamentary stabilization (DIS) for treatment of acute anterior cruciate ligament ruptures: case series experience of the first three years. BMC Musculoskelet Disord. 2015;16:27.
42. Adachi N, Ochi M, Uchio Y, Iwasa J, Ryoke K, Kuriwaka M. Mechanoreceptors in the anterior cruciate ligament contribute to the joint position sense. Acta Orthop Scand. 2002;73(3):330-334.
43. Gao F, Zhou J, He C, et al. A morphologic and quantitative study of mechanoreceptors in the remnant stump of the human anterior cruciate ligament. Arthroscopy. 2016;32(2):273-280.
44. Georgoulis AD, Pappa L, Moebius U, et al. The presence of proprioceptive mechanoreceptors in the remnants of the ruptured ACL as a possible source of re-innervation of the ACL autograft. Knee Surg Sports Traumatol Arthrosc. 2001;9(6):364-368.
45. Fleming BC, Carey JL, Spindler KP, Murray MM. Can suture repair of ACL transection restore normal anteroposterior laxity of the knee? An ex vivo study. J Orthop Res. 2008;26(11):1500-1505.
46. Murray MM, Fleming BC. Use of a bioactive scaffold to stimulate anterior cruciate ligament healing also minimizes posttraumatic osteoarthritis after surgery. Am J Sports Med. 2013;41(8):1762-1770.
47. Ahn JH, Wang JH, Lee YS, Kim JG, Kang JH, Koh KH. Anterior cruciate ligament reconstruction using remnant preservation and a femoral tensioning technique: clinical and magnetic resonance imaging results. Arthroscopy. 2011;27(8):1079-1089.
48. Lee BI, Kwon SW, Kim JB, Choi HS, Min KD. Comparison of clinical results according to amount of preserved remnant in arthroscopic anterior cruciate ligament reconstruction using quadrupled hamstring graft. Arthroscopy. 2008;24(5):560-568.
49. Lee BI, Min KD, Choi HS, et al. Immunohistochemical study of mechanoreceptors in the tibial remnant of the ruptured anterior cruciate ligament in human knees. Knee Surg Sports Traumatol Arthrosc. 2009;17(9):1095-1101.
50. Takahashi T, Kondo E, Yasuda K, et al. Effects of remnant tissue preservation on the tendon graft in anterior cruciate ligament reconstruction: a biomechanical and histological study. Am J Sports Med. 2016;44(7):1708-1716.
51. Dong S, Xie G, Zhang Y, Shen P, Huangfu X, Zhao J. Ligamentization of autogenous hamstring grafts after anterior cruciate ligament reconstruction: midterm versus long-term results. Am J Sports Med. 2015;43(8):1908-1917.
52. Takazawa Y, Ikeda H, Kawasaki T, et al. ACL reconstruction preserving the ACL remnant achieves good clinical outcomes and can reduce subsequent graft rupture. Orthop J Sports Med. 2013;1(4):2325967113505076.
53. Shimodaira H, Tensho K, Akaoka Y, Takanashi S, Kato H, Saito N. Remnant-preserving tibial tunnel positioning using anatomic landmarks in double-bundle anterior cruciate ligament reconstruction. Arthroscopy. 2016;32(9):1822-1830.
54. Zhang Q, Zhang S, Cao X, Liu L, Liu Y, Li R. The effect of remnant preservation on tibial tunnel enlargement in ACL reconstruction with hamstring autograft: a prospective randomized controlled trial. Knee Surg Sports Traumatol Arthrosc. 2014;22(1):166-173.
55. Tie K, Chen L, Hu D, Wang H. The difference in clinical outcome of single-bundle anterior cruciate ligament reconstructions with and without remnant preservation: A meta-analysis. Knee. 2016;23(4):566-574.
56. Robson AW. VI. Ruptured crucial ligaments and their repair by operation. Ann Surg. 1903;37(5):716-718.
57. Palmer I. On the injuries to the ligaments of the knee joint. Acta Orthop Scand. 1938;53.
58. Palmer I. On the injuries to the ligaments of the knee joint: a clinical study. 1938. Clin Orthop Relat Res. 2007;454:17-22.
59 O’Donoghue DH. An analysis of end results of surgical treatment of major injuries to the ligaments of the knee. J Bone Joint Surg Am. 1955;37-A(1):1-13.
60. O’Donoghue DH. Surgical treatment of fresh injuries to the major ligaments of the knee. J Bone Joint Surg Am. 1950;32 A(4):721-738.
61. Feagin JA, Abbott HG, Rokous JR. The isolated tear of the anterior cruciate ligament. J Bone Joint Surg Am. 1972;54-A:1340-1341.
62. England RL. Repair of the ligaments about the knee. Orthop Clin North Am. 1976;7(1):195-204.
63. Marshall JL, Warren RF, Wickiewicz TL, Reider B. The anterior cruciate ligament: a technique of repair and reconstruction. Clin Orthop Relat Res. 1979;(143):97-106.
64. Weaver JK, Derkash RS, Freeman JR, Kirk RE, Oden RR, Matyas J. Primary knee ligament repair--revisited. Clin Orthop Relat Res. 1985;(199):185-191.
65. Nogalski MP, Bach BR Jr. A review of early anterior cruciate ligament surgical repair or reconstruction. Results and caveats. Orthop Rev. 1993;22(11):1213-1223.
66. Feagin JA Jr, Curl WW. Isolated tear of the anterior cruciate ligament: 5-year follow-up study. Am J Sports Med. 1976;4(3):95-100.
67. Marshall JL, Warren RF, Wickiewicz TL. Primary surgical treatment of anterior cruciate ligament lesions. Am J Sports Med. 1982;10(2):103-107.
68. Straub T, Hunter RE. Acute anterior cruciate ligament repair. Clin Orthop Relat Res. 1988;227:238-250.
69. Kaplan N, Wickiewicz TL, Warren RF. Primary surgical treatment of anterior cruciate ligament ruptures. A long-term follow-up study. Am J Sports Med. 1990;18(4):354-358.
70. Sherman MF, Lieber L, Bonamo JR, Podesta L, Reiter I. The long-term followup of primary anterior cruciate ligament repair. Defining a rationale for augmentation. Am J Sports Med. 1991;19(3):243-255.
71. Paar O. Use of semitendinosus tendon to strengthen a freshly repaired anterior cruciate ligament. Chirurg. 1985;56(11):728-734.
72. Aglietti P, Buzzi R, Pisaneschi A, Salvi M. Comparison between suture and augmentation with the semitendinosus tendon in the repair of acute lesions of the anterior cruciate ligament. Ital J Orthop Traumatol. 1986;8(4):217-231.
73. Higgins RW, Steadman JR. Anterior cruciate ligament repairs in world class skiers. Am J Sports Med. 1987;15(5):439-447.
74. Harilainen A, Myllynen P. Treatment of fresh tears of the anterior cruciate ligament. A comparison of primary suture and augmentation with carbon fibre. Injury. 1987;18(6):396-400.
75. Jones KG. Results of use of the central one-third of the patellar ligament to compensate for anterior cruciate ligament deficiency. Clin Orthop Relat Res. 1980;(147):39-44.
76. Puddu G. Method for reconstruction of the anterior cruciate ligament using the semitendinosus tendon. Am J Sports Med. 1980;8(6):402-404.
77. Hefti F, Gächter A, Jenny H, Morscher E. Replacement of the anterior cruciate ligament. a comparative study of four different methods of reconstruction. Arch Orthop Trauma Surg. 1982;100(2):83-94.
78. Odensten M, Hamberg P, Nordin M, Lysholm J, Gillquist J. Surgical or conservative treatment of the acutely torn anterior cruciate ligament. A randomized study with short-term follow-up observations. Clin Orthop Relat Res. 1985;(198):87-93.
79. Andersson C, Odensten M, Good L, Gillquist J. Surgical or non-surgical treatment of acute rupture of the anterior cruciate ligament. A randomized study with long-term follow-up. J Bone Joint Surg Am. 1989;71(7):965-974.
80. Engebretsen L, Benum P, Fasting O, Mølster A, Strand T. A prospective, randomized study of three surgical techniques for treatment of acute ruptures of the anterior cruciate ligament. Am J Sports Med. 1990;18(6):585-590.
81. Jonsson T, Peterson L, Renström P. Anterior cruciate ligament repair with and without augmentation. A prospective 7-year study of 51 patients. Acta Orthop Scand. 1990;61(6):562-566.
82. Andersson C, Odensten M, Gillquist J. Knee function after surgical or nonsurgical treatment of acute rupture of the anterior cruciate ligament: a randomized study with a long-term follow-up period. Clin Orthop Relat Res. 1991;(264):255-263.
83. Heim U, Bachmann B, Infanger K. Reinsertion of the anterior cruciate ligament or primary ligamentous plasty? Helv Chir Acta. 1982;48(5):703-708.
84. Strand T, Engesaeter LB, Mølster AO, et al. Knee function following suture of fresh tear of the anterior cruciate ligament. Acta Orthop Scand. 1984;55(2):181-184.
85. Marcacci M, Spinelli M, Chiellini F, Buccolieri V. Notes on 53 cases of immediate suture of acute lesions of the anterior cruciate ligament. Ital J Orthop Traumatol. 1985;7(2):69-79.
86. van der List JP, DiFelice GS. Primary repair of the anterior cruciate ligament: a paradigm shift. Surgeon. 2016 Oct 6. [Epub ahead of print]
87. Bräm J, Plaschy S, Lütolf M, Leutenegger A. [The primary cruciate ligament suture--is the method outdated? Results in follow-up of 58 patients]. Z Unfallchir Versicherungsmed. 1994;87(2):91-109.
88. Genelin F, Trost A, Primavesi C, Knoll P. Late results following proximal reinsertion of isolated ruptured ACL ligaments. Knee Surg Sports Traumatol Arthrosc. 1993;1(1):17-19.
89. Kühne JH, Theermann R, Neumann R, Sagasser J. [Acute uncomplicated anterior knee instability. 2-5 year follow-up of surgical treatment]. Unfallchirurg. 1991;94(2):81-87.
90. Simonet WT, Sim FH. Repair and reconstruction of rotatory instability of the knee. Am J Sports Med. 1984;12(2):89-97.
91. Raunest J, Derra E, Ohmann C. [Clinical results of Palmer’s primary cruciate ligament insertion without augmentation]. Unfallchirurgie. 1991;17(3):166-174.
92. Frank C, Beaver P, Rademaker F, Becker K, Schachar N, Edwards G. A computerized study of knee-ligament injuries: repair versus removal of the torn anterior cruciate ligament. Can J Surg. 1982;25(4):454-458.
93. Enneking WF, Horowitz M. The intra-articular effects of immobilization on the human knee. J Bone Joint Surg Am. 1972;54(5):973-985.
94. Millett PJ, Wickiewicz TL, Warren RF. Motion loss after ligament injuries to the knee. Part I: causes. Am J Sports Med. 2001;29(5):664-675.
95. Bilko TE, Paulos LE, Feagin JA Jr, Lambert KL, Cunningham HR. Current trends in repair and rehabilitation of complete (acute) anterior cruciate ligament injuries. Analysis of 1984 questionnaire completed by ACL Study Group. Am J Sports Med. 1986;14(2):143-147.
96. Paulos L, Noyes FR, Grood E, Butler DL. Knee rehabilitation after anterior cruciate ligament reconstruction and repair. J Orthop Sports Phys Ther. 1991;13(2):60-70.
97. Paessler HH, Deneke J, Dahners LE. Augmented repair and early mobilization of acute anterior cruciate ligament injuries. Am J Sports Med. 1992;20(6):667-674.
98.
99. Kdolsky RK, Gibbons DF, Kwasny O, Schabus R, Plenk H Jr. Braided polypropylene augmentation device in reconstructive surgery of the anterior cruciate ligament: long-term clinical performance of 594 patients and short-term arthroscopic results, failure analysis by scanning electron microscopy, and synovial histomorphology. J Orthop Res. 1997;15(1):1-10.
100. Grøntvedt T, Engebretsen L. Comparison between two techniques for surgical repair of the acutely torn anterior cruciate ligament. A prospective, randomized follow-up study of 48 patients. Scand J Med Sci Sports. 1995;5(6):358-363.
101. Hehl G, Strecker W, Richter M, Kiefer H, Wissmeyer T. Clinical experience with PDS II augmentation for operative treatment of acute proximal ACL ruptures--2-year follow-up. Knee Surg Sports Traumatol Arthrosc. 1999;7(2):102-106.
102. Schenk S, Landsiedl F, Enenkel M. Arthroscopic single-stranded semitendinosus tendon- versus PDS-augmentation of reinserted acute femoral anterior cruciate ligament tears: 7 year follow-up study. Knee Surg Sports Traumatol Arthrosc. 2006;14(4):318-324.
103. Zysk SP, Refior HJ. Operative or conservative treatment of the acutely torn anterior cruciate ligament in middle-aged patients. A follow-up study of 133 patients between the ages of 40 and 59 years. Arch Orthop Trauma Surg. 2000;120(1-2):59-64.
104. Krueger-Franke M, Siebert CH, Schupp A. Refixation of femoral anterior cruciate ligament tears combined with a semitendinosus tendon augmentation. Technique and results. Arch Orthop Trauma Surg. 1998;117(1-2):68-72.
105. Natri A, Järvinen M, Kannus P. Primary repair plus intra-articular iliotibial band augmentation in the treatment of an acute anterior cruciate ligament rupture. A follow-up study of 70 patients. Arch Orthop Trauma Surg. 1996;115(1):22-27.
106. Träger D, Pohle K, Tschirner W. Anterior cruciate ligament suture in comparison with plasty. A 5-year follow-up study. Arch Orthop Trauma Surg. 1995;114(5):278-280.
107. Shelbourne KD, Wilckens JH, Mollabashy A, DeCarlo M. Arthrofibrosis in acute anterior cruciate ligament reconstruction. The effect of timing of reconstruction and rehabilitation. Am J Sports Med. 1991;19(4):332-336.
108. Volokhina YV, Syed HM, Pham PH, Blackburn AK. Two helpful MRI signs for evaluation of posterolateral bundle tears of the anterior cruciate ligament: a pilot study. Orthop J Sports Med. 2015;3(8):2325967115597641.
109. Strand T, Mølster A, Hordvik M, Krukhaug Y. Long-term follow-up after primary repair of the anterior cruciate ligament: clinical and radiological evaluation 15-23 years postoperatively. Arch Orthop Trauma Surg. 2005;125(4):217-221.
110. van der List JP, DiFelice GS. Successful arthroscopic primary repair of a chronic anterior cruciate ligament tear 11 years following injury. HSS J. 2016. In press.
111. van der List JP, DiFelice GS. The role of ligament repair in anterior cruciate ligament surgery. In: Mascarenhas R, Bhatia S, Lowe WR, eds. Ligamentous Injuries of the Knee. 1st ed. Houston: Nova Science Publishers; 2016:199-220.
112. van der List JP, DiFelice GS. Gap formation following primary anterior cruciate ligament repair: a biomechanical study. Knee. 2016. In press.
113. DiFelice GS, van der List JP. Arthroscopic primary repair of proximal anterior cruciate ligament tears. Arthrosc Tech. 2016. In press.
114. Smith JO, Yasen SK, Palmer HC, Lord BR, Britton EM, Wilson AJ. Paediatric ACL repair reinforced with temporary internal bracing. Knee Surg Sports Traumatol Arthrosc. 2016;24(6):1845-1851.
115. Magarian EM, Fleming BC, Harrison SL, Mastrangelo AN, Badger GJ, Murray MM. Delay of 2 or 6 weeks adversely affects the functional outcome of augmented primary repair of the porcine anterior cruciate ligament. Am J Sports Med. 2010;38(12):2528-2534.
116. Murray MM, Magarian EM, Harrison SL, Mastrangelo AN, Zurakowski D, Fleming BC. The effect of skeletal maturity on functional healing of the anterior cruciate ligament. J Bone Joint Surg Am. 2010;92(11):2039-2049.
117. Werner BC, Yang S, Looney AM, Gwathmey FW Jr. Trends in pediatric and adolescent anterior cruciate ligament iInjury and reconstruction. J Pediatr Orthop. 2016;36(5):447-452.
118. Frosch KH, Stengel D, Brodhun T, et al. Outcomes and risks of operative treatment of rupture of the anterior cruciate ligament in children and adolescents. Arthroscopy. 2010;26(11):1539-1550.
119. Ramski DE, Kanj WW, Franklin CC, Baldwin KD, Ganley TJ. Anterior cruciate ligament tears in children and adolescents: a meta-analysis of nonoperative versus operative treatment. Am J Sports Med. 2014;42(11):2769-2776.
120. Zantop T, Brucker PU, Vidal A, Zelle BA, Fu FH. Intraarticular rupture pattern of the ACL. Clin Orthop Relat Res. 2007;454:48-53.
121. Yoon KH, Bae DK, Cho SM, Park SY, Lee JH. Standard anterior cruciate ligament reconstruction versus isolated single-bundle augmentation with hamstring autograft. Arthroscopy. 2009;25(11):1265-1274.
122 Demirağ B, Ermutlu C, Aydemir F, Durak K. A comparison of clinical outcome of augmentation and standard reconstruction techniques for partial anterior cruciate ligament tears. Eklem Hastalik Cerrahisi. 2012;23(3):140-144.
123. Sonnery-Cottet B, Zayni R, Conteduca J, et al. Posterolateral bundle reconstruction with anteromedial bundle remnant preservation in ACL tears: clinical and MRI evaluation of 39 patients with 24-month follow-up. Orthop J Sports Med. 2013;1(3):2325967113501624.
124. Sabat D, Kumar V. Partial tears of anterior cruciate ligament: results of single bundle augmentation. Indian J Orthop. 2015;49(2):129-135.
125. Jung YB, Jung HJ, Siti HT, et al. Comparison of anterior cruciate ligament reconstruction with preservation only versus remnant tensioning technique. Arthroscopy. 2011;27(9):1252-1258.
126. Nguyen DT, Ramwadhdoebe TH, van der Hart CP, Blankevoort L, Tak PP, van Dijk CN. Intrinsic healing response of the human anterior cruciate ligament: an histological study of reattached ACL remnants. J Orthop Res. 2014;32(2):296-301.
127. Boutsiadis A, Karampalis C, Tzavelas A, Vraggalas V, Christodoulou P, Bisbinas I. Anterior cruciate ligament remnant-preserving reconstruction using a “lasso-loop” knot configuration. Arthrosc Tech. 2015;4(6):e741-e746.
128. Noh JH, Yoon KH, Song SJ, Roh YH. Re-tensioning technique to cover the graft with remnant in anterior cruciate ligament reconstruction. Arthrosc Tech. 2014;3(6):e679-e682.
129. Hong L, Li X, Zhang H, et al. Anterior cruciate ligament reconstruction with remnant preservation: a prospective, randomized controlled study. Am J Sports Med. 2012;40(12):2747-2755.
130. Noh JH, Kyung HS, Roh YH, Kang TS. Remnant-preserving and re-tensioning technique to cover the graft in anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2015 Nov 12. [Epub ahead of print]
131. Muneta T, Koga H, Ju YJ, Horie M, Nakamura T, Sekiya I. Remnant volume of anterior cruciate ligament correlates preoperative patients’ status and postoperative outcome. Knee Surg Sports Traumatol Arthrosc. 2013;21(4):906-913.
132. Leeberg V, Lekdorf J, Wong C, Sonne-Holm S. Tibial eminentia avulsion fracture in children - a systematic review of the current literature. Dan Med J. 2014;61(3):A4792.
133. In Y, Kwak DS, Moon CW, Han SH, Choi NY. Biomechanical comparison of three techniques for fixation of tibial avulsion fractures of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 2012;20(8):1470-1478.
Injury of the anterior cruciate ligament (ACL) is very common with over 200,000 annual injuries in the United Status.1,2 There is a general consensus that these injuries should not be treated conservatively in patients that are younger, or who wish to remain active.3,4 Reconstructive surgery is currently the preferred treatment in these patients, and anatomic single-bundle reconstruction with autografts is considered the gold standard.5,6
Reconstruction of the ACL is, however, not a perfect treatment. Following single-bundle autograft reconstruction, revision rates of 3% to 8%,6-9 contralateral injury rates of 3% to 8%,10,11 and infection rates of 0.5% to 3%7,12,13 have been reported. Furthermore, due to the invasive nature of graft harvesting and the surgical procedure, 10% to 25% of the patients are not satisfied following ACL reconstruction.14,15 This can often be explained by common complaints, such as anterior knee pain (13%-43%), kneeling pain (12%-54%), quadriceps muscle atrophy (20%-30%),16,17 and loss of range of motion (ROM) (12%-23%).7,9,18,19 Furthermore, as a result of the invasive nature of reconstructive surgery, revisions can be difficult due to complications, such as tunnel widening, tunnel malpositioning, and preexisting hardware.20-22 This can lead to inferior outcomes and higher rates (13%) of revision surgery compared to primary reconstruction.23-26 Finally, reconstructive surgery does not restore native kinematics of the ACL,27-29 which may partially explain why reconstructive surgery has not been shown to prevent osteoarthritis.28-31
Over the past decades, there has been an increasing interest in the preservation of the ACL in an attempt to ameliorate these issues.32-37 Ligament preservation focuses on preserving the native tissues and biology, while minimizing the surgical morbidity to the patients.
Some authors have recently reported on arthroscopic primary repair of proximal ACL tears in which the ligament is reattached onto the femoral wall using modern-day suture anchor technology.32,38 Others have augmented this repair technique with an internal brace39,40 or with a synthetic device.33,41 When performing primary repair, it is believed that proprioception is maintained,42-44 while experimental studies have suggested that primary repair also restores the native kinematics,45 and may prevent osteoarthritis.46 Furthermore, primary repair is a conservative approach in that no grafts need to be harvested, no tunnels need to be drilled, and revision surgery, if necessary, is more analogous to primary reconstructions.32In patients with partial tears, some surgeons have advocated preserving the anteromedial (AM) or posterolateral (PL) bundle and performing selective single-bundle augmentation.34,35 In addition, several authors have used remnant tensioning36,47 or remnant preservation37,48 in combination with reconstructive surgery in order to benefit from the biological characteristics of the remnant. These techniques lead to better proprioceptive function,44,49,50 vascularization and ligamentization of the graft,50-52 provide an optical guide for anatomic tunnel placement,53 and decrease the incidence of tunnel widening.54,55The feasibility and applicability of these surgical techniques mainly depends on the tear type and tissue quality of the torn ligament. In this article we (I) discuss the history of ACL preservation, (II) discuss how modern advances alter the risk-benefit ratio for ACL preservation, and (III) propose a treatment algorithm for ACL injuries that is based on tear location and tissue quality.
History of ACL Preservation
The history of the surgical treatment of ACL injuries started in 1895 when Robson56 treated a 41-year-old male who tore both cruciate ligaments from the femoral wall. Performing primary repair with catgut ligatures, both cruciate ligaments were preserved and the patients had resolution of pain symptoms and full function at 6-year follow-up. Over the following decades, Palmer57,58 and O’Donoghue59,60 further popularized open primary repair for the treatment of ACL injuries, and this technique was the most commonly performed treatment in the 1970s and early 1980s.61-65 The initial short-term results of primary repair were excellent,61,62 but Feagin and Curl66 were the first to note that the results deteriorated at mid-term follow-up. Despite improvements in the surgical technique of repairing the ACL, such as the usage of nonabsorbable sutures and directly tying the sutures over bone,63,67 the results remained disappointing at longer-term follow-up.68-70
In response to these disappointing results, surgeons sought to improve the surgical treatment by either augmenting the primary repair with a semitendinosus, a patella tendon graft or an augmentation device,71-74 or by performing primary reconstruction.75-77 At the end of the 1980s and early 1990s, several randomized and prospective clinical trials were performed in order to compare the outcomes of these techniques.74,78-82 Many studies showed that results of augmented repair were more reliable when compared to primary repair, which led to the abandonment of primary repair in favor of augmented repair, and eventually primary reconstruction.65
The Important Role of Tear Location in Ligament Preservation
When taking a closer look at the outcomes of primary repair and augmented repair, it seems that the results of these preservation techniques were not as disappointing as was suggested. This can be explained, in large part, by the fact that the important roles of tear location and tissue quality were not widely recognized.
Sherman and colleagues70 reported in 1991 their mid-term results of open primary repair. Similar to others, they noted a deterioration of their results at mid-term follow-up. However, they uniquely performed an extensive subgroup analysis in order to find an explanation for this. In their study, considered a landmark paper on primary repair,65,70 they concluded that, “poor tissue quality is typical for midsubstance tears and that a repair of these injuries will predictably fail while type I tears (proximal), with better tissue quality, show a definite trend towards better results.”70 With these findings, they confirmed the findings of others that had recognized a trend of better outcomes with proximal tears.64,67,83-85
A majority of the historical studies that were published before 1991 had not considered the role of tear location and tissue quality on outcomes of open primary repair. This was also true for the aforementioned randomized studies that compared primary repair with augmented repair and primary reconstruction. Because these studies randomized patients and did not take tear location into account, it can be expected that patients with midsubstance tears were included in the cohorts of primary repair and the outcomes of these studies were therefore confounded.74,78-82 If these studies would have been aware of the role that tear location plays on primary repair outcomes, different outcomes may have been found and different conclusions on the optimal treatment for different tear types may have been drawn.86
Open Primary ACL Repair Outcomes Stratified by Tear Location
When reviewing the literature of open primary repair outcomes stratified by tear location, it is noted that multiple studies reported excellent outcomes following primary repair of proximal ACL tears.73,83,84,87-90 Weaver and colleagues64 were among the first to stratify their results by tear location, and they found that more patients with proximal tears (52 of 66; 79%) were satisfied after the procedure when compared to patients with midsubstance tears (3 of 13; 23%) at 3.5-year follow-up. They concluded that, “selection can be made with some predictability of the type of injury to the ligament as to which patients will do better.”64 Kühne and colleagues89 reported the outcomes of 75 patients with proximal tears treated with open primary repair and noted no failures, negative pivot shift in 88% of patients, stable or nearly stable Lachman test in 87% of patients, and 89% return to sports rate at 4-year follow-up. Raunest and colleagues91 reported a negative pivot shift and negative anterior drawer test in 84%, return to sports in 71%, and satisfaction in 75% of 51 patients that underwent open primary repair of proximal tears at 3.5-year follow-up.
Interestingly, and in contrast to the findings of Feagin and Curl,66 no deterioration of the outcomes at mid-term follow-up was noted in patients with proximal tears. Genelin and colleagues88 reported their results of 42 patients with proximal tears treated with open primary repair at 5- to 7-year follow-up. They found a negative pivot shift in 81%, stable or nearly stable Lachman test in 81%, and patient satisfaction in 86% of patients. Similarly, Bräm and colleagues87 found good results at mid-term follow-up with a good-excellent Lysholm score in 79%, return to a similar level of sports in 76%, stable or nearly stable Lachman test in 91%, and anterior drawer test in 94% of patients, along with an 88% satisfaction rate and 7% failure rate in patients who underwent open primary repair of proximal tears.
On the contrary, when the outcomes of studies that performed open primary repair in mainly, or only, patients with midsubstance tears are reviewed, significantly inferior results are found. Frank and colleagues92 reported outcomes in 42 patients with midsubstance tears at 4-year follow-up. They reported that 56% had a stable or nearly stable anterior drawer test, 78% had a positive pivot shift, and that only 61% were satisfied with the procedure. Odensten and colleagues78 reported outcomes of open primary repair in a subgroup of 22 patients with midsubstance tears at 1.5-year follow-up, and noted a 14% failure rate.
When reviewing the mid-term results in patients with midsubstance tears, it seems that there was more deterioration in outcomes.69,70 Firstly, the aforementioned study by Sherman and colleagues70 showed poor results in the patients with (type IV) midsubstance tears at mid-term follow-up. Furthermore, Kaplan and colleagues69 reported the mid-term outcomes of 70 patients, of which 56 patients had midsubstance tears. After having reported good outcomes at short-term follow-up,63,67 they noted that 42% of patients had >3 mm anteroposterior stability when compared to the contralateral leg, only a 62% return to sport rate, and a 17% failure rate. They concluded that, “Although … primary repair of the anterior cruciate may work in some patients, it is an unpredictable operative procedure.”
These studies showed that the outcomes of open primary repair were significantly better in patients with proximal ACL tears and sufficient tissue quality when compared to midsubstance tears. This suggests that open primary ACL repair may have been prematurely abandoned as a treatment option for patients with proximal tears.
Augmented ACL Repair
There were several reasons why augmented repair became the preferred treatment in the early and mid 1990s. First of all, the results of augmented repair were more consistent compared to primary repair in the aforementioned randomized and prospective studies,74,78-82 which is not surprising given the fact that the role of tear location was not widely recognized at the time. Secondly, in the 1970s and early 1980s, patients were treated postoperatively in a cast for 6 weeks, which led to problems, such as loss of ROM, pain, and decreased function.93,94 At the end of the1980s and 1990s, the focus shifted from prolonged joint immobilization towards early postoperative ROM.95-97 Since many authors believed that primary repair of the ACL was not strong enough to tolerate early mobilization, an augmentation was added to the technique in order to fortify the repair and enable early ROM.98
Interestingly, augmented repair, which is essentially a combination of primary ACL repair and ACL reconstruction, was mainly performed in the 1990s and many surgeons did recognize the role of tear location in this treatment at this point.73,98-103 In these years, the treatment algorithm consisted of augmented ACL repair in patients with proximal tears in the acute setting and ACL reconstruction in patients with midsubstance or chronic tears. Several different augmentation techniques were used to reinforce the primary repair in these years including autograft tissues (semitendinosus tendon,102-104 patellar tendon,100 or iliotibial band [ITB]105) synthetic materials (polydioxanone [PDS],101,102,106 carbon fibre,74 and polyester [Trevira]97), augmentation devices (Kennedy Ligament Augmentation Device [LAD]98-100) and extra-articular augmentations.73
When reviewing the outcomes of augmented repair of the ACL, good to excellent results can be found in studies that used this technique in patients with proximal tears.73,98-106 Kdolsky and colleagues98 were in one of the first groups that reported their results of augmented repair in only patients with proximal tears. In 1993, they reported their mid-term outcomes (5 to 8 years) in 66 patients who underwent primary repair and augmentation with the Kennedy LAD and found that 97% of patients had stable knees (<3 mm on KT-1000 examination), 98% had a negative pivot shift, and 76% returned to previous level of sports. However, often-reported problems with the augmentation devices were found in this study with rupture of the device (12%) and decreased ROM (14%).98 In 1995, Grøntvedt and Engebretsen100 compared augmentation with the Kennedy LAD to patellar tendon augmentation in a randomized study of patients with acute proximal tears. They noted that 50% of the patients in the Kennedy LAD group had a positive pivot shift compared to 23% in the patellar tendon group. Furthermore, they found KT-1000 leg differences of <3 mm in 92% of the patellar tendon group and 54% of the Kennedy LAD group. Because the authors found significant differences between both groups at 1- and 2-year follow-up, they stopped the clinical trial.
Several authors in the following years reported good results of augmented repair using autograft tissues. Natri and colleagues105 reported the outcomes of 72 patients treated with primary repair of proximal tears augmented with the ITB at 3.5-year follow-up. They found 89% negative pivot shift rate, 93% stable or nearly stable Lachman test, 99% stable or nearly stable anterior drawer test, 79% satisfaction rate, and 91% return to previous level of sports rate. Krueger-Franke and colleagues104 reported the outcomes of primary repair of proximal tears with augmentation using the semitendinosus tendon. In a retrospective study of 76 patients, they noted that 96% of patients had a negative pivot shift, 75% of patients had stable or nearly stable Lachman test, 93% were satisfied with the procedure, a mean Lysholm score of 92, a Tegner score that only decreased from 7.2 to 7.1, and KT-1000 testing with 78% <4 mm leg difference with the contralateral leg. The authors concluded that patients with femoral ruptures could be treated with augmented repair when performed in the acute setting. As this study was published in 1998, they stated that magnetic resonance imaging and arthroscopy could be helpful in identifying the tear location.
Final Abandonment of ACL Preservation
Reviewing these outcomes raises the question as to why these techniques were ultimately abandoned in the treatment algorithm of proximal ACL injuries, especially given the aforementioned advantages of ACL preservation. One of the possible answers can be found in a landmark study on ACL reconstruction and rehabilitation published by Shelbourne and colleagues107 in 1991. At that time, arthrofibrosis and knee stiffness were frequently reported problems following ACL surgery, which could partially be explained by the standard conservative rehabilitation using postoperative joint immobilization.67,70,80,88
Shelbourne and colleagues107 aimed to assess the cause of arthrofibrosis and knee stiffness, and divided the patients into groups by number of days between injury and surgery (<7, 7 to 21 days, and >21 days between injury and surgery). Furthermore, patients within these groups underwent either a conventional or accelerated rehabilitation program. The authors not only found that patients undergoing accelerated rehabilitation had less arthrofibrosis, but they also noted that less arthrofibrosis was seen when surgery was delayed. These findings, however, contrasted with the general perception that the ACL should be repaired in the first 3 weeks postinjury to ensure optimal tissue quality with an augmented approach. As a result, the treatment of ACL injuries shifted towards ACL reconstruction after these findings. Krueger-Franke and colleagues104 commented on the trend after the study of Shelbourne and colleagues:107 “Less consideration has been given to the importance of the proprioceptive receptors in the tibial remnants of the torn ACL and the value of their preservation as part of a primary reconstruction.”
In addition to the trend away from an augmented repair approach due to the novel understanding of the importance of early mobilization, some discussion should focus on the technical limitations of arthroscopy at that time. While arthroscopy had been around for several decades, fluid management and arthroscopic instrumentation was slow to develop. All of the repair and augmentation techniques previously discussed had been performed via an open arthrotomy. Arthroscopic technologies of the time were not refined enough to enable surgeons to perform such complex, intra-articular techniques that would enable suturing of the ligament remnant. In this regard, arthroscopic ACL reconstruction was a much simpler technique to accomplish, and this also likely contributed to the final abandonment of the ligament preservation approach.
Role for ACL Preservation with Modern Advances
As stated in the introduction, there has been a recent resurgence of interest in preservation of the native ligament.32-37 With the passage of time, many technologic advances have been made, which has allowed surgeons to reconsider the concept of ligament preservation.
First of all, appropriate patient selection was not applied historically, as the critical factors of tear location and tissue quality were not recognized in the era of open primary repair. In modern days, however, advances such as MRI have been developed, which can give the surgeon an idea of the status, and tear type of the ACL pre-operatively.108 This may help the orthopaedic surgeon to plan the surgery and make an assessment as to whether ACL preservation is possible. Secondly, in the historic literature the postoperative regimen consisted of casting for 5 or 6 weeks,67,70,80,88 while the focus later shifted towards early ROM.95-97Modern day ACL rehabilitation focuses on immediate ROM to avoid the complications stiffness, pain and decreased function that plagued the outcomes when immobilization was used.93,94 Thirdly, historically small tunnels were drilled with primary repair and sutures had to be tied over bone,57,67 whereas currently suture anchors are available that prevent the need for tunnel drilling and enable direct suture tensioning.32,38 Finally, and most importantly, in the historic literature patients were treated with an invasive arthrotomy technique, while modern day arthroscopic techniques readily enable the surgeon to effectively suture the remnant arthroscopically. Interestingly, in 2005, in their 20-year follow-up of primary repair surgeries, Strand and colleagues109 stated, “if the same results could be accomplished by a smaller, arthroscopic procedure, primary repair might reduce the number of patients needing later reconstructions with small ‘costs’ in the way of risk and inconvenience for the patients. We therefore believe that further research and development of methods for closed (arthroscopic) repair are justified.”
Altered Risk-Benefit Ratio
Historically, the treatments of open primary repair and open ACL reconstruction were both invasive surgeries with an arthrotomy, drilling of bone tunnels, and postoperative joint immobilization for 4 to 6 weeks. However, with the modern-day advances, the risk-benefit ratio of both treatments has changed, as Strand and colleagues109 had already suggested. Although ACL reconstruction can be performed arthroscopically, it remains an invasive procedure, in which tunnels are drilled, patellar tendons or hamstring tendons are harvested, and complications, such as knee pain and quadriceps atrophy, are common. The surgery of primary ACL repair, however, has benefited significantly from the modern developments.32,38 Primary ACL repair can now be performed arthroscopically, and by using suture anchors no tunnels need to be drilled and the remnant can be tensioned directly. An additional benefit of the use of suture anchors is that revision surgery of a failed primary repair is analogous to primary reconstruction, whereas revision surgery of a failed ACL reconstruction can be problematic due to tunnel widening, tunnel malpositioning, and preexisting hardware.20-22
Reviewing the differences between arthroscopic primary ACL repair and ACL reconstruction, it becomes clear that primary repair has benefited significantly from the modern advances and that the risk-benefit ratio for primary repair has been altered. This means that patients with proximal tears can be treated with a relatively straight forward, minimally invasive surgery, which has been shown to be effective in 85% to 90% of patients.32,38
Treatment Algorithm Based on Tear Location
Since 2008, in the practice of the senior author (GSD), the surgical treatment algorithm for ACL injuries is completely based on the tear location and tissue quality of the ligament.110,111 To describe the different tear types, we use the modified Sherman classification in which we extended his classification towards the tibial side whereas Sherman and colleagues70 only described the femoral side of the tears (Figures A-F, Table).
Type I Tears: Primary Repair
Type I tears are soft tissue avulsion type tears that can be easily treated with arthroscopic primary repair.107 The length of the distal remnant has to be at least 90% and the tissue quality has to be good to excellent in order to approximate the remnant towards the femoral wall (Table).112 The incidence of type I tears was 26% in the study of Sherman and colleagues,70 although recent studies showed a lower incidence (6% to 10%) in a larger population.32,38 Certainly, individual practices will see different percentages of type I tears based upon the mix of injury mechanisms they see most frequently. Over the last 2 years, with the recognition of the importance of tear type and tissue quality, there has been a renewed interest in arthroscopic primary ACL repair.32,38
DiFelice and colleagues32 were the first to arthroscopically perform primary repair of the ACL in proximal tears using suture anchors. They reported the outcomes of the first 11 consecutive patients that underwent primary repair in a previously described technique.113 At mean 3.5-year follow-up, they noted only 1 failure (9%) due to re-injury; mean Lysholm score of 93.2; mean modified Cincinnati score of 91.5; pre- and postoperative Tegner score of 7.3 and 6.9, respectively; SANE score of 91.8; and subjective International Knee Documentation Committee (IKDC) score of 86.4. Of the patients with an intact repair, 9 patients had an objective IKDC rating A and 1 patient had B and all patients had KT-1000 leg differences of <3 mm with the contralateral side (three patients were not available for KT-1000 testing). The authors concluded that arthroscopic primary ACL repair could achieve short-term clinical success in a selected group of patients with proximal avulsion tears and excellent tissue quality. They further noted that mid-term outcomes are necessary given that the results of open primary repair deteriorated at longer-term follow-up in the historical literature. Recently, the senior author (GSD) has added an Internal Brace (Arthrex) to the primary repair with the goal of protecting the ligament in the first weeks to further promote healing of the ligament.39,40,114
More recently, Achtnich and colleagues38 compared the treatment of arthroscopic primary ACL repair with primary ACL reconstruction in 41 patients with type I tears at 2.3-years follow-up. Twenty-one patients consented for primary repair while 20 patients declined this procedure and underwent primary reconstruction. They noted no significant differences in Lachman test, pivot shift test, objective IKDC score, and KT-1000 scores. Although not significant, the clinical failure rate in the primary repair group (15%) was higher than the reconstruction group (0%). Interestingly, despite the higher failure rate in the repair group, the authors concluded that primary ACL repair is recommended in a carefully selected group of patients with type I tears and excellent tissue quality, which can likely be explained by the differences in the risk-benefit ratio between both procedures.
Over the last decade, the research group led by Murray46,115,116 has performed experimental research on primary repair with a biological scaffold and reported many interesting findings that could be extrapolated to primary ACL repair. First of all, they compared bioenhanced primary repair with bioenhanced primary reconstruction in 64 Yucutan pigs and noted that there was significantly less macroscopic cartilage damage in the primary repair group at 1-year follow-up.46 They concluded that bioenhanced ACL repair may provide a new, less invasive treatment option that reduces cartilage damage following joint injury. This may suggest that primary repair may have a lower incidence of osteoarthritis when compared to ACL reconstruction, which is interesting as osteoarthritis is very common after ACL reconstruction. Further research in this area is certainly warranted.
In another study they compared bioenhanced primary repair in juvenile, adolescent and mature Yucutan pigs and noted that functional healing depended on the level of skeletal maturity with immature animals having a more productive healing response.116 This indicates that primary repair might be a good treatment option in skeletally immature patients, especially since reconstruction increases the risk of premature closure of the epiphysis117,118 and delaying treatment increases the risk of meniscus injury.119 Interestingly, a recent meta-analysis showed indeed that the risk of epiphysis closure was lower in primary repair when compared to ACL reconstruction and the rupture rate was also lower.118 Primary repair may be a good treatment option in children as the procedure has all the attributes that should be applicable to children: it is minimally morbid, tissue sparing, and it is a conservative approach that does not burn any surgical bridges for future reconstructive surgery if necessary.
Finally, the research group of Murray115 assessed the effect of surgical delay of primary repair following injury in Yucutan pigs and noted that better biomechanical outcomes were noted after delaying surgery for 2 weeks when compared to 6 weeks. This suggests that primary repair should preferably be performed in the acute setting, which has also been shown in historical studies since the ligament in the acute setting has optimal tissue quality and the ligament is less likely to be retracted or reabsorbed.59,60,115
One Bundle Type I Tears: Single Bundle Augmented Repair
In some cases, the tear locations of the AM and PL bundle are not at the same location and Zantop and colleagues120 reported in an arthroscopic study that this could be as frequent as in 30% of all complete tears. In some of these tears, one of the bundles can be avulsed of the femoral wall (type I tear) while the other bundle is not directly repairable (non-type I tear). In these cases, the senior author (GSD) will repair the type I tear bundle, whereas a hamstring augmentation is placed at the location of the other bundle. When reviewing the literature, a combination of primary repair of one bundle and reconstruction of the second bundle has not been described before. However, over the last decade several surgeons have performed augmentation of one bundle in the setting of partial tears.34,35,121-124
Buda and colleagues34 were the first to perform selective AM or PL bundle reconstruction in the setting of partial tears.34 At 5-year follow-up, they reported no reruptures and only 1 patient with an IKDC C-score, although reoperation was necessary in 4 out of 47 patients (9%). Following this publication, many others reported on selective bundle reconstruction.35,121-124 However, with partial tears, the knee is often stable and a selective augmentation technique is utilized to prevent complete rupture of the ligament. The application of this technique is essentially different from reconstruction for complete ACL tears in which the knee is unstable, there is a giving way sensation and patients have problems participating in sports.
Type II Tears: Augmented Repair
Type II tears often have good or excellent tissue quality and can be pulled up towards the femoral footprint, but are too short to be firmly attached. Sherman and colleagues70 reported that approximately 22% patients had a type II tear, which corresponds to a tear located in the proximal part of the ligament. With this technique, multiple suture passes are used to stitch the remnant and, in addition, a smaller hamstring autograft or allograft is passed through the middle of the tibial remnant. A suture button is used proximally for the graft, and the tensioning repair sutures through the remnant are also passed through the suture button. The suture button is passed through the femoral tunnel and flipped so that the graft is proximally fixed. Then, the repair sutures of the remnant are tensioned, and the ligament is pulled towards the femoral wall as a sleeve around the graft. When the ligament is approximated to the femoral wall, the sutures are tied over the suture button. The graft is then tensioned distally to complete the augmented repair.
In the recent literature, the technique of augmentation of a primary repair using autograft tissue has not been reported. However, augmented repair using an internal brace39,40 or augmentation devices33,41 have been recently performed. MacKay and colleagues39 reported good outcomes of arthroscopic primary repair of proximal tears using an internal brace. Eggli and colleagues33 reported the results of the first 10 patients treated with ACL preservation using primary repair of the ligament with the addition of a dynamic screw-spring mechanism. The authors reported good preliminary results with one failure (10%) and good objective and subjective outcomes. In a next study, they reported the outcomes of 278 patients and although they reported good clinical outcomes and a revision rate of 4%, the reoperation rate for removal of the screw-spring mechanism was high (24%).41 This is not surprising when reviewing the historical literature in which high complication rates of the augmentation devices were reported.99,100 We were unable to identify any other studies reporting surgical techniques of augmenting primary repair in the literature.
Type III Tears: Reconstruction With Remnant Tensioning
In patients with type III tears, the ligament cannot be approximated to the wall and reconstruction is necessary in order to restore knee stability. However, in these cases the ligament has sufficient length (25%-75%) and can be tensioned along or around the graft. Preservation of the ligament remnant has several (theoretical) advantages, such as better proprioceptive function,42,49,50 vascularization and ligamentization of the graft,50-52 an optical guide for anatomic tunnel placement,53 and a decreased incidence of tunnel widening.54,55 Furthermore, tensioning of the remnant is thought to lower the risk of cyclops lesions when compared to remnant preservation.125 Although the difference between augmented repair and remnant tensioning seems small, the purpose of surgery is different. With augmented repair, the ligament can be approximated close to the femoral wall and the goal of surgery is to use the healing capacity that the ACL has in the proximal part of the ligament,126 while with remnant tensioning the goal is only to benefit from some of the aforementioned advantages. Ahn and colleagues36 were the first to perform this technique and stated, “Our concept is that the remnant tissue has only an additive effect.” Furthermore, with augmented repair multiple sutures are passed through the AM and PL bundle in order to sufficiently approximate the ligament to the femoral wall, while with the remnant tensioning technique generally one or a few sutures or lasso loop are passed through the proximal part to tension the ligament, prevent sagging of the remnant, and decrease the risk of cyclops lesions.127,128
Several authors have recently performed remnant tensioning during ACL.36,47,125-127 Ahn and colleagues47 reported excellent objective and subjective outcomes following this procedure and found that with re-arthroscopy nearly all patients had fair synovialization of the graft. Others have reported similarly good outcomes of these techniques.125,129,130 However, studies comparing this treatment with normal ACL reconstruction and assessing outcomes, failure rates and proprioception are lacking.
Type IV Tears: Reconstruction With Remnant Preservation
Finally, in some patients the ligament is torn distally or the tissue quality is not optimal. In these patients, the remnant can be debrided to the part of good tissue quality in order to preserve the biology and minimize the risk for cyclops lesions. A standard reconstruction needs to be performed to restore the instability, but by preserving the remnant, advantages, such as proprioception,44,49,50 graft vascularization,50-52 an optical guide for tibial tunnel placement,53 and a decreased incidence of tunnel widening54,55 can be expected.
Lee and colleagues37 presented the tibial remnant technique in which standard reconstruction was performed, and the tibial tunnel was drilled through the center of the remnant. In a later study, they compared remnant preservation with a remnant of <20% of the total ACL length with >20% of the length and found that proprioception was better with more remnant volume.48 Similarly, Muneta and colleagues131 assessed the role of remnant length and found that remnant length is positively correlated with better stability measured on KT-1000 anteroposterior stability.
Several studies compared ACL reconstruction with remnant preservation vs conventional ACL reconstruction.52,54,129 Takazawa and colleagues52 performed a retrospective study of 183 patients and found that patients in the remnant preservation group had significantly better KT-2000 stability, while they also reported a significantly lower graft rupture rate in this group (1.1% vs 7.1%) at 2-year follow-up. Hong and colleagues129 performed a randomized clinical trial of 80 patients and did not find these differences, although there was a trend towards higher Lysholm scores in the remnant preservation group. Finally, Zhang and colleagues54 performed a randomized clinical trial and found a lower incidence and amount of tibial tunnel widening in the preserving-remnant group when compared to the removing-remnant group. These studies show that there is likely a role for remnant preservation.
Type V Tears: Primary Repair
In some patients, the ligament is torn in the distal 10% of the ligament, which can occur as a distal avulsion tear or as a distal bony avulsion fracture.132 Bony avulsion fractures are most commonly seen in children whereas true distal soft tissue avulsion tears are very rare.132
Treatments of these tear types include antegrade screw fixation, pullout sutures or the use of suture anchors in case of bony avulsion fractures and pullout sutures with tying over a bony bridge or ligament button in case of soft tissue avulsions. Leeberg and colleagues132 recently performed a systematic review of all studies reporting on treatment of distal avulsion fractures.They noted that most treatments were currently performed arthroscopically and that outcomes were generally good. Another recent biomechanical study compared antegrade screw fixation with suture anchor fixation and pullout suture fixation.133 The authors noted that suture anchor fixation has slightly less displacement of the bony fragment when compared to screw fixation and pull-out sutures, and that the strength to failure was higher in the suture anchor fixation when compared to the pullout suture fixation. The outcomes of this study suggest that screw fixation and suture anchor fixation might be superior to pullout suture fixation, which might be interesting as with pullout suture fixation the ligament cannot be directly tensioned to the tibial footprint, which can lead to anteroposterior laxity.132 Clinical studies are necessary to assess the preferred treatment in these tear types but it seems that screw fixation is preferred in large bony avulsion fractures, while suture anchor fixation or pullout suture fixation can be used for soft tissue avulsion tears.
Complex Tears or Poor Tissue Quality: Reconstruction
If the tear is complex, multiple tears are present, or the tissue quality is poor, then preservation of the ligament is not possible, and in these cases a standard reconstruction should be performed.
Conclusion
When reviewing the literature of ACL preservation, it becomes clear that the evolution of surgical treatment of ACL injuries was biased. Preservation of the native ligament has many advantages, such as better proprioception, graft vascularization, an optical guide for tibial tunnel placement, and a decreased incidence of tunnel widening that can be expected. Furthermore, arthroscopic primary ACL repair is minimally invasive and does not burn any bridges for future reconstructions, if necessary. This is in addition to the other (theoretical) advantages of primary repair, such as restoration of native kinematics and a decreased risk of osteoarthritis. Modern advances have significantly changed the risk-benefit ratio that should make us reconsider ACL preservation approaches. Certainly, further research in this area is warranted. In this article we have presented a treatment algorithm for ACL preservation, which is based on tear location and remnant tissue quality.
Am J Orthop. 2016;45(7):E393-E405. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
Injury of the anterior cruciate ligament (ACL) is very common with over 200,000 annual injuries in the United Status.1,2 There is a general consensus that these injuries should not be treated conservatively in patients that are younger, or who wish to remain active.3,4 Reconstructive surgery is currently the preferred treatment in these patients, and anatomic single-bundle reconstruction with autografts is considered the gold standard.5,6
Reconstruction of the ACL is, however, not a perfect treatment. Following single-bundle autograft reconstruction, revision rates of 3% to 8%,6-9 contralateral injury rates of 3% to 8%,10,11 and infection rates of 0.5% to 3%7,12,13 have been reported. Furthermore, due to the invasive nature of graft harvesting and the surgical procedure, 10% to 25% of the patients are not satisfied following ACL reconstruction.14,15 This can often be explained by common complaints, such as anterior knee pain (13%-43%), kneeling pain (12%-54%), quadriceps muscle atrophy (20%-30%),16,17 and loss of range of motion (ROM) (12%-23%).7,9,18,19 Furthermore, as a result of the invasive nature of reconstructive surgery, revisions can be difficult due to complications, such as tunnel widening, tunnel malpositioning, and preexisting hardware.20-22 This can lead to inferior outcomes and higher rates (13%) of revision surgery compared to primary reconstruction.23-26 Finally, reconstructive surgery does not restore native kinematics of the ACL,27-29 which may partially explain why reconstructive surgery has not been shown to prevent osteoarthritis.28-31
Over the past decades, there has been an increasing interest in the preservation of the ACL in an attempt to ameliorate these issues.32-37 Ligament preservation focuses on preserving the native tissues and biology, while minimizing the surgical morbidity to the patients.
Some authors have recently reported on arthroscopic primary repair of proximal ACL tears in which the ligament is reattached onto the femoral wall using modern-day suture anchor technology.32,38 Others have augmented this repair technique with an internal brace39,40 or with a synthetic device.33,41 When performing primary repair, it is believed that proprioception is maintained,42-44 while experimental studies have suggested that primary repair also restores the native kinematics,45 and may prevent osteoarthritis.46 Furthermore, primary repair is a conservative approach in that no grafts need to be harvested, no tunnels need to be drilled, and revision surgery, if necessary, is more analogous to primary reconstructions.32In patients with partial tears, some surgeons have advocated preserving the anteromedial (AM) or posterolateral (PL) bundle and performing selective single-bundle augmentation.34,35 In addition, several authors have used remnant tensioning36,47 or remnant preservation37,48 in combination with reconstructive surgery in order to benefit from the biological characteristics of the remnant. These techniques lead to better proprioceptive function,44,49,50 vascularization and ligamentization of the graft,50-52 provide an optical guide for anatomic tunnel placement,53 and decrease the incidence of tunnel widening.54,55The feasibility and applicability of these surgical techniques mainly depends on the tear type and tissue quality of the torn ligament. In this article we (I) discuss the history of ACL preservation, (II) discuss how modern advances alter the risk-benefit ratio for ACL preservation, and (III) propose a treatment algorithm for ACL injuries that is based on tear location and tissue quality.
History of ACL Preservation
The history of the surgical treatment of ACL injuries started in 1895 when Robson56 treated a 41-year-old male who tore both cruciate ligaments from the femoral wall. Performing primary repair with catgut ligatures, both cruciate ligaments were preserved and the patients had resolution of pain symptoms and full function at 6-year follow-up. Over the following decades, Palmer57,58 and O’Donoghue59,60 further popularized open primary repair for the treatment of ACL injuries, and this technique was the most commonly performed treatment in the 1970s and early 1980s.61-65 The initial short-term results of primary repair were excellent,61,62 but Feagin and Curl66 were the first to note that the results deteriorated at mid-term follow-up. Despite improvements in the surgical technique of repairing the ACL, such as the usage of nonabsorbable sutures and directly tying the sutures over bone,63,67 the results remained disappointing at longer-term follow-up.68-70
In response to these disappointing results, surgeons sought to improve the surgical treatment by either augmenting the primary repair with a semitendinosus, a patella tendon graft or an augmentation device,71-74 or by performing primary reconstruction.75-77 At the end of the 1980s and early 1990s, several randomized and prospective clinical trials were performed in order to compare the outcomes of these techniques.74,78-82 Many studies showed that results of augmented repair were more reliable when compared to primary repair, which led to the abandonment of primary repair in favor of augmented repair, and eventually primary reconstruction.65
The Important Role of Tear Location in Ligament Preservation
When taking a closer look at the outcomes of primary repair and augmented repair, it seems that the results of these preservation techniques were not as disappointing as was suggested. This can be explained, in large part, by the fact that the important roles of tear location and tissue quality were not widely recognized.
Sherman and colleagues70 reported in 1991 their mid-term results of open primary repair. Similar to others, they noted a deterioration of their results at mid-term follow-up. However, they uniquely performed an extensive subgroup analysis in order to find an explanation for this. In their study, considered a landmark paper on primary repair,65,70 they concluded that, “poor tissue quality is typical for midsubstance tears and that a repair of these injuries will predictably fail while type I tears (proximal), with better tissue quality, show a definite trend towards better results.”70 With these findings, they confirmed the findings of others that had recognized a trend of better outcomes with proximal tears.64,67,83-85
A majority of the historical studies that were published before 1991 had not considered the role of tear location and tissue quality on outcomes of open primary repair. This was also true for the aforementioned randomized studies that compared primary repair with augmented repair and primary reconstruction. Because these studies randomized patients and did not take tear location into account, it can be expected that patients with midsubstance tears were included in the cohorts of primary repair and the outcomes of these studies were therefore confounded.74,78-82 If these studies would have been aware of the role that tear location plays on primary repair outcomes, different outcomes may have been found and different conclusions on the optimal treatment for different tear types may have been drawn.86
Open Primary ACL Repair Outcomes Stratified by Tear Location
When reviewing the literature of open primary repair outcomes stratified by tear location, it is noted that multiple studies reported excellent outcomes following primary repair of proximal ACL tears.73,83,84,87-90 Weaver and colleagues64 were among the first to stratify their results by tear location, and they found that more patients with proximal tears (52 of 66; 79%) were satisfied after the procedure when compared to patients with midsubstance tears (3 of 13; 23%) at 3.5-year follow-up. They concluded that, “selection can be made with some predictability of the type of injury to the ligament as to which patients will do better.”64 Kühne and colleagues89 reported the outcomes of 75 patients with proximal tears treated with open primary repair and noted no failures, negative pivot shift in 88% of patients, stable or nearly stable Lachman test in 87% of patients, and 89% return to sports rate at 4-year follow-up. Raunest and colleagues91 reported a negative pivot shift and negative anterior drawer test in 84%, return to sports in 71%, and satisfaction in 75% of 51 patients that underwent open primary repair of proximal tears at 3.5-year follow-up.
Interestingly, and in contrast to the findings of Feagin and Curl,66 no deterioration of the outcomes at mid-term follow-up was noted in patients with proximal tears. Genelin and colleagues88 reported their results of 42 patients with proximal tears treated with open primary repair at 5- to 7-year follow-up. They found a negative pivot shift in 81%, stable or nearly stable Lachman test in 81%, and patient satisfaction in 86% of patients. Similarly, Bräm and colleagues87 found good results at mid-term follow-up with a good-excellent Lysholm score in 79%, return to a similar level of sports in 76%, stable or nearly stable Lachman test in 91%, and anterior drawer test in 94% of patients, along with an 88% satisfaction rate and 7% failure rate in patients who underwent open primary repair of proximal tears.
On the contrary, when the outcomes of studies that performed open primary repair in mainly, or only, patients with midsubstance tears are reviewed, significantly inferior results are found. Frank and colleagues92 reported outcomes in 42 patients with midsubstance tears at 4-year follow-up. They reported that 56% had a stable or nearly stable anterior drawer test, 78% had a positive pivot shift, and that only 61% were satisfied with the procedure. Odensten and colleagues78 reported outcomes of open primary repair in a subgroup of 22 patients with midsubstance tears at 1.5-year follow-up, and noted a 14% failure rate.
When reviewing the mid-term results in patients with midsubstance tears, it seems that there was more deterioration in outcomes.69,70 Firstly, the aforementioned study by Sherman and colleagues70 showed poor results in the patients with (type IV) midsubstance tears at mid-term follow-up. Furthermore, Kaplan and colleagues69 reported the mid-term outcomes of 70 patients, of which 56 patients had midsubstance tears. After having reported good outcomes at short-term follow-up,63,67 they noted that 42% of patients had >3 mm anteroposterior stability when compared to the contralateral leg, only a 62% return to sport rate, and a 17% failure rate. They concluded that, “Although … primary repair of the anterior cruciate may work in some patients, it is an unpredictable operative procedure.”
These studies showed that the outcomes of open primary repair were significantly better in patients with proximal ACL tears and sufficient tissue quality when compared to midsubstance tears. This suggests that open primary ACL repair may have been prematurely abandoned as a treatment option for patients with proximal tears.
Augmented ACL Repair
There were several reasons why augmented repair became the preferred treatment in the early and mid 1990s. First of all, the results of augmented repair were more consistent compared to primary repair in the aforementioned randomized and prospective studies,74,78-82 which is not surprising given the fact that the role of tear location was not widely recognized at the time. Secondly, in the 1970s and early 1980s, patients were treated postoperatively in a cast for 6 weeks, which led to problems, such as loss of ROM, pain, and decreased function.93,94 At the end of the1980s and 1990s, the focus shifted from prolonged joint immobilization towards early postoperative ROM.95-97 Since many authors believed that primary repair of the ACL was not strong enough to tolerate early mobilization, an augmentation was added to the technique in order to fortify the repair and enable early ROM.98
Interestingly, augmented repair, which is essentially a combination of primary ACL repair and ACL reconstruction, was mainly performed in the 1990s and many surgeons did recognize the role of tear location in this treatment at this point.73,98-103 In these years, the treatment algorithm consisted of augmented ACL repair in patients with proximal tears in the acute setting and ACL reconstruction in patients with midsubstance or chronic tears. Several different augmentation techniques were used to reinforce the primary repair in these years including autograft tissues (semitendinosus tendon,102-104 patellar tendon,100 or iliotibial band [ITB]105) synthetic materials (polydioxanone [PDS],101,102,106 carbon fibre,74 and polyester [Trevira]97), augmentation devices (Kennedy Ligament Augmentation Device [LAD]98-100) and extra-articular augmentations.73
When reviewing the outcomes of augmented repair of the ACL, good to excellent results can be found in studies that used this technique in patients with proximal tears.73,98-106 Kdolsky and colleagues98 were in one of the first groups that reported their results of augmented repair in only patients with proximal tears. In 1993, they reported their mid-term outcomes (5 to 8 years) in 66 patients who underwent primary repair and augmentation with the Kennedy LAD and found that 97% of patients had stable knees (<3 mm on KT-1000 examination), 98% had a negative pivot shift, and 76% returned to previous level of sports. However, often-reported problems with the augmentation devices were found in this study with rupture of the device (12%) and decreased ROM (14%).98 In 1995, Grøntvedt and Engebretsen100 compared augmentation with the Kennedy LAD to patellar tendon augmentation in a randomized study of patients with acute proximal tears. They noted that 50% of the patients in the Kennedy LAD group had a positive pivot shift compared to 23% in the patellar tendon group. Furthermore, they found KT-1000 leg differences of <3 mm in 92% of the patellar tendon group and 54% of the Kennedy LAD group. Because the authors found significant differences between both groups at 1- and 2-year follow-up, they stopped the clinical trial.
Several authors in the following years reported good results of augmented repair using autograft tissues. Natri and colleagues105 reported the outcomes of 72 patients treated with primary repair of proximal tears augmented with the ITB at 3.5-year follow-up. They found 89% negative pivot shift rate, 93% stable or nearly stable Lachman test, 99% stable or nearly stable anterior drawer test, 79% satisfaction rate, and 91% return to previous level of sports rate. Krueger-Franke and colleagues104 reported the outcomes of primary repair of proximal tears with augmentation using the semitendinosus tendon. In a retrospective study of 76 patients, they noted that 96% of patients had a negative pivot shift, 75% of patients had stable or nearly stable Lachman test, 93% were satisfied with the procedure, a mean Lysholm score of 92, a Tegner score that only decreased from 7.2 to 7.1, and KT-1000 testing with 78% <4 mm leg difference with the contralateral leg. The authors concluded that patients with femoral ruptures could be treated with augmented repair when performed in the acute setting. As this study was published in 1998, they stated that magnetic resonance imaging and arthroscopy could be helpful in identifying the tear location.
Final Abandonment of ACL Preservation
Reviewing these outcomes raises the question as to why these techniques were ultimately abandoned in the treatment algorithm of proximal ACL injuries, especially given the aforementioned advantages of ACL preservation. One of the possible answers can be found in a landmark study on ACL reconstruction and rehabilitation published by Shelbourne and colleagues107 in 1991. At that time, arthrofibrosis and knee stiffness were frequently reported problems following ACL surgery, which could partially be explained by the standard conservative rehabilitation using postoperative joint immobilization.67,70,80,88
Shelbourne and colleagues107 aimed to assess the cause of arthrofibrosis and knee stiffness, and divided the patients into groups by number of days between injury and surgery (<7, 7 to 21 days, and >21 days between injury and surgery). Furthermore, patients within these groups underwent either a conventional or accelerated rehabilitation program. The authors not only found that patients undergoing accelerated rehabilitation had less arthrofibrosis, but they also noted that less arthrofibrosis was seen when surgery was delayed. These findings, however, contrasted with the general perception that the ACL should be repaired in the first 3 weeks postinjury to ensure optimal tissue quality with an augmented approach. As a result, the treatment of ACL injuries shifted towards ACL reconstruction after these findings. Krueger-Franke and colleagues104 commented on the trend after the study of Shelbourne and colleagues:107 “Less consideration has been given to the importance of the proprioceptive receptors in the tibial remnants of the torn ACL and the value of their preservation as part of a primary reconstruction.”
In addition to the trend away from an augmented repair approach due to the novel understanding of the importance of early mobilization, some discussion should focus on the technical limitations of arthroscopy at that time. While arthroscopy had been around for several decades, fluid management and arthroscopic instrumentation was slow to develop. All of the repair and augmentation techniques previously discussed had been performed via an open arthrotomy. Arthroscopic technologies of the time were not refined enough to enable surgeons to perform such complex, intra-articular techniques that would enable suturing of the ligament remnant. In this regard, arthroscopic ACL reconstruction was a much simpler technique to accomplish, and this also likely contributed to the final abandonment of the ligament preservation approach.
Role for ACL Preservation with Modern Advances
As stated in the introduction, there has been a recent resurgence of interest in preservation of the native ligament.32-37 With the passage of time, many technologic advances have been made, which has allowed surgeons to reconsider the concept of ligament preservation.
First of all, appropriate patient selection was not applied historically, as the critical factors of tear location and tissue quality were not recognized in the era of open primary repair. In modern days, however, advances such as MRI have been developed, which can give the surgeon an idea of the status, and tear type of the ACL pre-operatively.108 This may help the orthopaedic surgeon to plan the surgery and make an assessment as to whether ACL preservation is possible. Secondly, in the historic literature the postoperative regimen consisted of casting for 5 or 6 weeks,67,70,80,88 while the focus later shifted towards early ROM.95-97Modern day ACL rehabilitation focuses on immediate ROM to avoid the complications stiffness, pain and decreased function that plagued the outcomes when immobilization was used.93,94 Thirdly, historically small tunnels were drilled with primary repair and sutures had to be tied over bone,57,67 whereas currently suture anchors are available that prevent the need for tunnel drilling and enable direct suture tensioning.32,38 Finally, and most importantly, in the historic literature patients were treated with an invasive arthrotomy technique, while modern day arthroscopic techniques readily enable the surgeon to effectively suture the remnant arthroscopically. Interestingly, in 2005, in their 20-year follow-up of primary repair surgeries, Strand and colleagues109 stated, “if the same results could be accomplished by a smaller, arthroscopic procedure, primary repair might reduce the number of patients needing later reconstructions with small ‘costs’ in the way of risk and inconvenience for the patients. We therefore believe that further research and development of methods for closed (arthroscopic) repair are justified.”
Altered Risk-Benefit Ratio
Historically, the treatments of open primary repair and open ACL reconstruction were both invasive surgeries with an arthrotomy, drilling of bone tunnels, and postoperative joint immobilization for 4 to 6 weeks. However, with the modern-day advances, the risk-benefit ratio of both treatments has changed, as Strand and colleagues109 had already suggested. Although ACL reconstruction can be performed arthroscopically, it remains an invasive procedure, in which tunnels are drilled, patellar tendons or hamstring tendons are harvested, and complications, such as knee pain and quadriceps atrophy, are common. The surgery of primary ACL repair, however, has benefited significantly from the modern developments.32,38 Primary ACL repair can now be performed arthroscopically, and by using suture anchors no tunnels need to be drilled and the remnant can be tensioned directly. An additional benefit of the use of suture anchors is that revision surgery of a failed primary repair is analogous to primary reconstruction, whereas revision surgery of a failed ACL reconstruction can be problematic due to tunnel widening, tunnel malpositioning, and preexisting hardware.20-22
Reviewing the differences between arthroscopic primary ACL repair and ACL reconstruction, it becomes clear that primary repair has benefited significantly from the modern advances and that the risk-benefit ratio for primary repair has been altered. This means that patients with proximal tears can be treated with a relatively straight forward, minimally invasive surgery, which has been shown to be effective in 85% to 90% of patients.32,38
Treatment Algorithm Based on Tear Location
Since 2008, in the practice of the senior author (GSD), the surgical treatment algorithm for ACL injuries is completely based on the tear location and tissue quality of the ligament.110,111 To describe the different tear types, we use the modified Sherman classification in which we extended his classification towards the tibial side whereas Sherman and colleagues70 only described the femoral side of the tears (Figures A-F, Table).
Type I Tears: Primary Repair
Type I tears are soft tissue avulsion type tears that can be easily treated with arthroscopic primary repair.107 The length of the distal remnant has to be at least 90% and the tissue quality has to be good to excellent in order to approximate the remnant towards the femoral wall (Table).112 The incidence of type I tears was 26% in the study of Sherman and colleagues,70 although recent studies showed a lower incidence (6% to 10%) in a larger population.32,38 Certainly, individual practices will see different percentages of type I tears based upon the mix of injury mechanisms they see most frequently. Over the last 2 years, with the recognition of the importance of tear type and tissue quality, there has been a renewed interest in arthroscopic primary ACL repair.32,38
DiFelice and colleagues32 were the first to arthroscopically perform primary repair of the ACL in proximal tears using suture anchors. They reported the outcomes of the first 11 consecutive patients that underwent primary repair in a previously described technique.113 At mean 3.5-year follow-up, they noted only 1 failure (9%) due to re-injury; mean Lysholm score of 93.2; mean modified Cincinnati score of 91.5; pre- and postoperative Tegner score of 7.3 and 6.9, respectively; SANE score of 91.8; and subjective International Knee Documentation Committee (IKDC) score of 86.4. Of the patients with an intact repair, 9 patients had an objective IKDC rating A and 1 patient had B and all patients had KT-1000 leg differences of <3 mm with the contralateral side (three patients were not available for KT-1000 testing). The authors concluded that arthroscopic primary ACL repair could achieve short-term clinical success in a selected group of patients with proximal avulsion tears and excellent tissue quality. They further noted that mid-term outcomes are necessary given that the results of open primary repair deteriorated at longer-term follow-up in the historical literature. Recently, the senior author (GSD) has added an Internal Brace (Arthrex) to the primary repair with the goal of protecting the ligament in the first weeks to further promote healing of the ligament.39,40,114
More recently, Achtnich and colleagues38 compared the treatment of arthroscopic primary ACL repair with primary ACL reconstruction in 41 patients with type I tears at 2.3-years follow-up. Twenty-one patients consented for primary repair while 20 patients declined this procedure and underwent primary reconstruction. They noted no significant differences in Lachman test, pivot shift test, objective IKDC score, and KT-1000 scores. Although not significant, the clinical failure rate in the primary repair group (15%) was higher than the reconstruction group (0%). Interestingly, despite the higher failure rate in the repair group, the authors concluded that primary ACL repair is recommended in a carefully selected group of patients with type I tears and excellent tissue quality, which can likely be explained by the differences in the risk-benefit ratio between both procedures.
Over the last decade, the research group led by Murray46,115,116 has performed experimental research on primary repair with a biological scaffold and reported many interesting findings that could be extrapolated to primary ACL repair. First of all, they compared bioenhanced primary repair with bioenhanced primary reconstruction in 64 Yucutan pigs and noted that there was significantly less macroscopic cartilage damage in the primary repair group at 1-year follow-up.46 They concluded that bioenhanced ACL repair may provide a new, less invasive treatment option that reduces cartilage damage following joint injury. This may suggest that primary repair may have a lower incidence of osteoarthritis when compared to ACL reconstruction, which is interesting as osteoarthritis is very common after ACL reconstruction. Further research in this area is certainly warranted.
In another study they compared bioenhanced primary repair in juvenile, adolescent and mature Yucutan pigs and noted that functional healing depended on the level of skeletal maturity with immature animals having a more productive healing response.116 This indicates that primary repair might be a good treatment option in skeletally immature patients, especially since reconstruction increases the risk of premature closure of the epiphysis117,118 and delaying treatment increases the risk of meniscus injury.119 Interestingly, a recent meta-analysis showed indeed that the risk of epiphysis closure was lower in primary repair when compared to ACL reconstruction and the rupture rate was also lower.118 Primary repair may be a good treatment option in children as the procedure has all the attributes that should be applicable to children: it is minimally morbid, tissue sparing, and it is a conservative approach that does not burn any surgical bridges for future reconstructive surgery if necessary.
Finally, the research group of Murray115 assessed the effect of surgical delay of primary repair following injury in Yucutan pigs and noted that better biomechanical outcomes were noted after delaying surgery for 2 weeks when compared to 6 weeks. This suggests that primary repair should preferably be performed in the acute setting, which has also been shown in historical studies since the ligament in the acute setting has optimal tissue quality and the ligament is less likely to be retracted or reabsorbed.59,60,115
One Bundle Type I Tears: Single Bundle Augmented Repair
In some cases, the tear locations of the AM and PL bundle are not at the same location and Zantop and colleagues120 reported in an arthroscopic study that this could be as frequent as in 30% of all complete tears. In some of these tears, one of the bundles can be avulsed of the femoral wall (type I tear) while the other bundle is not directly repairable (non-type I tear). In these cases, the senior author (GSD) will repair the type I tear bundle, whereas a hamstring augmentation is placed at the location of the other bundle. When reviewing the literature, a combination of primary repair of one bundle and reconstruction of the second bundle has not been described before. However, over the last decade several surgeons have performed augmentation of one bundle in the setting of partial tears.34,35,121-124
Buda and colleagues34 were the first to perform selective AM or PL bundle reconstruction in the setting of partial tears.34 At 5-year follow-up, they reported no reruptures and only 1 patient with an IKDC C-score, although reoperation was necessary in 4 out of 47 patients (9%). Following this publication, many others reported on selective bundle reconstruction.35,121-124 However, with partial tears, the knee is often stable and a selective augmentation technique is utilized to prevent complete rupture of the ligament. The application of this technique is essentially different from reconstruction for complete ACL tears in which the knee is unstable, there is a giving way sensation and patients have problems participating in sports.
Type II Tears: Augmented Repair
Type II tears often have good or excellent tissue quality and can be pulled up towards the femoral footprint, but are too short to be firmly attached. Sherman and colleagues70 reported that approximately 22% patients had a type II tear, which corresponds to a tear located in the proximal part of the ligament. With this technique, multiple suture passes are used to stitch the remnant and, in addition, a smaller hamstring autograft or allograft is passed through the middle of the tibial remnant. A suture button is used proximally for the graft, and the tensioning repair sutures through the remnant are also passed through the suture button. The suture button is passed through the femoral tunnel and flipped so that the graft is proximally fixed. Then, the repair sutures of the remnant are tensioned, and the ligament is pulled towards the femoral wall as a sleeve around the graft. When the ligament is approximated to the femoral wall, the sutures are tied over the suture button. The graft is then tensioned distally to complete the augmented repair.
In the recent literature, the technique of augmentation of a primary repair using autograft tissue has not been reported. However, augmented repair using an internal brace39,40 or augmentation devices33,41 have been recently performed. MacKay and colleagues39 reported good outcomes of arthroscopic primary repair of proximal tears using an internal brace. Eggli and colleagues33 reported the results of the first 10 patients treated with ACL preservation using primary repair of the ligament with the addition of a dynamic screw-spring mechanism. The authors reported good preliminary results with one failure (10%) and good objective and subjective outcomes. In a next study, they reported the outcomes of 278 patients and although they reported good clinical outcomes and a revision rate of 4%, the reoperation rate for removal of the screw-spring mechanism was high (24%).41 This is not surprising when reviewing the historical literature in which high complication rates of the augmentation devices were reported.99,100 We were unable to identify any other studies reporting surgical techniques of augmenting primary repair in the literature.
Type III Tears: Reconstruction With Remnant Tensioning
In patients with type III tears, the ligament cannot be approximated to the wall and reconstruction is necessary in order to restore knee stability. However, in these cases the ligament has sufficient length (25%-75%) and can be tensioned along or around the graft. Preservation of the ligament remnant has several (theoretical) advantages, such as better proprioceptive function,42,49,50 vascularization and ligamentization of the graft,50-52 an optical guide for anatomic tunnel placement,53 and a decreased incidence of tunnel widening.54,55 Furthermore, tensioning of the remnant is thought to lower the risk of cyclops lesions when compared to remnant preservation.125 Although the difference between augmented repair and remnant tensioning seems small, the purpose of surgery is different. With augmented repair, the ligament can be approximated close to the femoral wall and the goal of surgery is to use the healing capacity that the ACL has in the proximal part of the ligament,126 while with remnant tensioning the goal is only to benefit from some of the aforementioned advantages. Ahn and colleagues36 were the first to perform this technique and stated, “Our concept is that the remnant tissue has only an additive effect.” Furthermore, with augmented repair multiple sutures are passed through the AM and PL bundle in order to sufficiently approximate the ligament to the femoral wall, while with the remnant tensioning technique generally one or a few sutures or lasso loop are passed through the proximal part to tension the ligament, prevent sagging of the remnant, and decrease the risk of cyclops lesions.127,128
Several authors have recently performed remnant tensioning during ACL.36,47,125-127 Ahn and colleagues47 reported excellent objective and subjective outcomes following this procedure and found that with re-arthroscopy nearly all patients had fair synovialization of the graft. Others have reported similarly good outcomes of these techniques.125,129,130 However, studies comparing this treatment with normal ACL reconstruction and assessing outcomes, failure rates and proprioception are lacking.
Type IV Tears: Reconstruction With Remnant Preservation
Finally, in some patients the ligament is torn distally or the tissue quality is not optimal. In these patients, the remnant can be debrided to the part of good tissue quality in order to preserve the biology and minimize the risk for cyclops lesions. A standard reconstruction needs to be performed to restore the instability, but by preserving the remnant, advantages, such as proprioception,44,49,50 graft vascularization,50-52 an optical guide for tibial tunnel placement,53 and a decreased incidence of tunnel widening54,55 can be expected.
Lee and colleagues37 presented the tibial remnant technique in which standard reconstruction was performed, and the tibial tunnel was drilled through the center of the remnant. In a later study, they compared remnant preservation with a remnant of <20% of the total ACL length with >20% of the length and found that proprioception was better with more remnant volume.48 Similarly, Muneta and colleagues131 assessed the role of remnant length and found that remnant length is positively correlated with better stability measured on KT-1000 anteroposterior stability.
Several studies compared ACL reconstruction with remnant preservation vs conventional ACL reconstruction.52,54,129 Takazawa and colleagues52 performed a retrospective study of 183 patients and found that patients in the remnant preservation group had significantly better KT-2000 stability, while they also reported a significantly lower graft rupture rate in this group (1.1% vs 7.1%) at 2-year follow-up. Hong and colleagues129 performed a randomized clinical trial of 80 patients and did not find these differences, although there was a trend towards higher Lysholm scores in the remnant preservation group. Finally, Zhang and colleagues54 performed a randomized clinical trial and found a lower incidence and amount of tibial tunnel widening in the preserving-remnant group when compared to the removing-remnant group. These studies show that there is likely a role for remnant preservation.
Type V Tears: Primary Repair
In some patients, the ligament is torn in the distal 10% of the ligament, which can occur as a distal avulsion tear or as a distal bony avulsion fracture.132 Bony avulsion fractures are most commonly seen in children whereas true distal soft tissue avulsion tears are very rare.132
Treatments of these tear types include antegrade screw fixation, pullout sutures or the use of suture anchors in case of bony avulsion fractures and pullout sutures with tying over a bony bridge or ligament button in case of soft tissue avulsions. Leeberg and colleagues132 recently performed a systematic review of all studies reporting on treatment of distal avulsion fractures.They noted that most treatments were currently performed arthroscopically and that outcomes were generally good. Another recent biomechanical study compared antegrade screw fixation with suture anchor fixation and pullout suture fixation.133 The authors noted that suture anchor fixation has slightly less displacement of the bony fragment when compared to screw fixation and pull-out sutures, and that the strength to failure was higher in the suture anchor fixation when compared to the pullout suture fixation. The outcomes of this study suggest that screw fixation and suture anchor fixation might be superior to pullout suture fixation, which might be interesting as with pullout suture fixation the ligament cannot be directly tensioned to the tibial footprint, which can lead to anteroposterior laxity.132 Clinical studies are necessary to assess the preferred treatment in these tear types but it seems that screw fixation is preferred in large bony avulsion fractures, while suture anchor fixation or pullout suture fixation can be used for soft tissue avulsion tears.
Complex Tears or Poor Tissue Quality: Reconstruction
If the tear is complex, multiple tears are present, or the tissue quality is poor, then preservation of the ligament is not possible, and in these cases a standard reconstruction should be performed.
Conclusion
When reviewing the literature of ACL preservation, it becomes clear that the evolution of surgical treatment of ACL injuries was biased. Preservation of the native ligament has many advantages, such as better proprioception, graft vascularization, an optical guide for tibial tunnel placement, and a decreased incidence of tunnel widening that can be expected. Furthermore, arthroscopic primary ACL repair is minimally invasive and does not burn any bridges for future reconstructions, if necessary. This is in addition to the other (theoretical) advantages of primary repair, such as restoration of native kinematics and a decreased risk of osteoarthritis. Modern advances have significantly changed the risk-benefit ratio that should make us reconsider ACL preservation approaches. Certainly, further research in this area is warranted. In this article we have presented a treatment algorithm for ACL preservation, which is based on tear location and remnant tissue quality.
Am J Orthop. 2016;45(7):E393-E405. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Mall NA, Chalmers PN, Moric M, et al. Incidence and trends of anterior cruciate ligament reconstruction in the United States. Am J Sports Med. 2014;42(10):2363-2370.
2. Sanders TL, Maradit Kremers H, Bryan AJ, et al. Incidence of anterior cruciate ligament tears and reconstruction: a 21-year population-based study. Am J Sports Med. 2016;44(6):1502-1507.
3. Ciccotti MG, Lombardo SJ, Nonweiler B, Pink M. Non-operative treatment of ruptures of the anterior cruciate ligament in middle-aged patients. Results after long-term follow-up. J Bone Joint Surg Am. 1994;76(9):1315-1321.
4. Sanders TL, Pareek A, Kremers HM, et al. Long-term follow-up of isolated ACL tears treated without ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2016 May 24. [Epub ahead of print]
5. Irarrázaval S, Kurosaka M, Cohen M, Fu FH. Anterior cruciate ligament reconstruction. J ISAKOS. 2016;1(1):38-52.
6. Gabler CM, Jacobs CA, Howard JS, Mattacola CG, Johnson DL. Comparison of graft failure rate between autografts placed via an anatomic anterior cruciate ligament reconstruction technique: a systematic review, meta-analysis, and meta-regression. Am J Sports Med. 2016;44(4):1069-1079.
7. Li S, Chen Y, Lin Z, Cui W, Zhao J, Su W. A systematic review of randomized controlled clinical trials comparing hamstring autografts versus bone-patellar tendon-bone autografts for the reconstruction of the anterior cruciate ligament. Arch Orthop Trauma Surg. 2012;132(9):1287-1297.
8. Rahr-Wagner L, Thillemann TM, Pedersen AB, Lind M. Comparison of hamstring tendon and patellar tendon grafts in anterior cruciate ligament reconstruction in a nationwide population-based cohort study: results from the danish registry of knee ligament reconstruction. Am J Sports Med. 2014;42(2):278-284.
9. Xie X, Liu X, Chen Z, Yu Y, Peng S, Li Q. A meta-analysis of bone-patellar tendon-bone autograft versus four-strand hamstring tendon autograft for anterior cruciate ligament reconstruction. Knee. 2015;22(2):100-110.
10. Andernord D, Desai N, Björnsson H, Gillén S, Karlsson J, Samuelsson K. Predictors of contralateral anterior cruciate ligament reconstruction: a cohort study of 9061 patients with 5-year follow-up. Am J Sports Med. 2015;43(2):295-302.
11. Maletis GB, Inacio MC, Funahashi TT. Risk factors associated with revision and contralateral anterior cruciate ligament reconstructions in the Kaiser Permanente ACLR registry. Am J Sports Med. 2015;43(3):641-647.
12. Kim SJ, Postigo R, Koo S, Kim JH. Infection after arthroscopic anterior cruciate ligament reconstruction. Orthopedics. 2014;37(7):477-484.
13. Makhni EC, Steinhaus ME, Mehran N, Schulz BS, Ahmad CS. Functional outcome and graft retention in patients with septic arthritis after anterior cruciate ligament reconstruction: a systematic review. Arthroscopy. 2015;31(7):1392-1401.
14. Kocher MS, Steadman JR, Briggs K, Zurakowski D, Sterett WI, Hawkins RJ. Determinants of patient satisfaction with outcome after anterior cruciate ligament reconstruction. J Bone Joint Surg Am. 2002;84-A(9):1560-1572.
15. Ardern CL, Österberg A, Sonesson S, Gauffin H, Webster KE, Kvist J. Satisfaction with knee function after primary anterior cruciate ligament reconstruction is associated with self-efficacy, quality of life, and returning to the preinjury physical activity. Arthroscopy. 2016;32(8):1631-1638.e3.
16. Grant JA, Mohtadi NG, Maitland ME, Zernicke RF. Comparison of home versus physical therapy-supervised rehabilitation programs after anterior cruciate ligament reconstruction: a randomized clinical trial. Am J Sports Med. 2005;33(9):1288-1297.
17. Lindström M, Strandberg S, Wredmark T, Fell änder-Tsai L, Henriksson M. Functional and muscle morphometric effects of ACL reconstruction. A prospective CT study with 1 year follow-up. Scand J Med Sci Sports. 2013;23(4):431-442.
18. Biau DJ, Tournoux C, Katsahian S, Schranz PJ, Nizard RS. Bone-patellar tendon-bone autografts versus hamstring autografts for reconstruction of anterior cruciate ligament: meta-analysis. BMJ. 2006;332(7548):995-1001.
19. Spindler KP, Kuhn JE, Freedman KB, Matthews CE, Dittus RS, Harrell FE Jr. Anterior cruciate ligament reconstruction autograft choice: bone-tendon-bone versus hamstring: does it really matter? A systematic review. Am J Sports Med. 2004;32(8):1986-1995.
20. Aga C, Wilson KJ, Johansen S, Dornan G, La Prade RF, Engebretsen L. Tunnel widening in single- versus double-bundle anterior cruciate ligament reconstructed knees. Knee Surg Sports Traumatol Arthrosc. 2016 Jun 21. [Epub ahead of print]
21. Maak TG, Voos JE, Wickiewicz TL, Warren RF. Tunnel widening in revision anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2010;18(11):695-706.
22. Cheatham SA, Johnson DL. Anticipating problems unique to revision ACL surgery. Sports Med Arthrosc. 2013;21(2):129-134.
23. Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39(1):199-217.
24. Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531-536.
25. Andriolo L, Filardo G, Kon E, et al. Revision anterior cruciate ligament reconstruction: clinical outcome and evidence for return to sport. Knee Surg Sports Traumatol Arthrosc. 2015;23(10):2825-2845.
26. Grassi A, Ardern CL, Marcheggiani Muccioli GM, Neri MP, Marcacci M, Zaffagnini S. Does revision ACL reconstruction measure up to primary surgery? A meta-analysis comparing patient-reported and clinician-reported outcomes, and radiographic results. Br J Sports Med. 2016;50(12):716-724.
27. Ristanis S, Stergiou N, Patras K, Vasiliadis HS, Giakas G, Georgoulis AD. Excessive tibial rotation during high-demand activities is not restored by anterior cruciate ligament reconstruction. Arthroscopy. 2005;21(11):1323-1329.
28. Andriacchi TP, Mündermann A, Smith RL, Alexander EJ, Dyrby CO, Koo S. A framework for the in vivo pathomechanics of osteoarthritis at the knee. Ann Biomed Eng. 2004;32(3):447-457.
29. Imhauser C, Mauro C, Choi D, et al. Abnormal tibiofemoral contact stress and its association with altered kinematics after center-center anterior cruciate ligament reconstruction: an in vitro study. Am J Sports Med. 2013;41(4):815-825.
30. Ajuied A, Wong F, Smith C, et al. Anterior cruciate ligament injury and radiologic progression of knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2014;42(9):2242-2252.
31. Chalmers PN, Mall NA, Moric M, et al. Does ACL reconstruction alter natural history?: A systematic literature review of long-term outcomes. J Bone Joint Surg Am. 2014;96(4):292-300.
32. DiFelice GS, Villegas C, Taylor SA. Anterior cruciate ligament preservation: early results of a novel arthroscopic technique for suture anchor primary anterior cruciate ligament repair. Arthroscopy. 2015;31(11):2162-2171.
33. Eggli S, Kohlhof H, Zumstein M, et al. Dynamic intraligamentary stabilization: novel technique for preserving the ruptured ACL. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):1215-1221.
34. Buda R, Ferruzzi A, Vannini F, Zambelli L, Di Caprio F. Augmentation technique with semitendinosus and gracilis tendons in chronic partial lesions of the ACL: clinical and arthrometric analysis. Knee Surg Sports Traumatol Arthrosc. 2006;14(11):1101-1107.
35. Ochi M, Adachi N, Uchio Y, et al. A minimum 2-year follow-up after selective anteromedial or posterolateral bundle anterior cruciate ligament reconstruction. Arthroscopy. 2009;25(2):117-122.
36. Ahn JH, Lee YS, Ha HC. Anterior cruciate ligament reconstruction with preservation of remnant bundle using hamstring autograft: technical note. Arch Orthop Trauma Surg. 2009;129(8):1011-1015.
37. Lee BI, Min KD, Choi HS, Kim JB, Kim ST. Arthroscopic anterior cruciate ligament reconstruction with the tibial-remnant preserving technique using a hamstring graft. Arthroscopy. 2006;22(3):340.e1-e7.
38. Achtnich A, Herbst E, Forkel P, et al. Acute proximal anterior cruciate ligament tears: outcomes after arthroscopic suture anchor repair versus anatomic single-bundle reconstruction. Arthroscopy. 2016 Jun 17. [Epub ahead of print]
39. MacKay G, Anthony IC, Jenkins PJ, Blyth M. Anterior cruciate ligament repair revisited. Preliminary results of primary repair with internal brace ligament augmentation: a case series. Orthop Muscul Syst. 2015;4:188.
40. Mackay GM, Blyth MJ, Anthony I, Hopper GP, Ribbans WJ. A review of ligament augmentation with the InternalBrace™: the surgical principle is described for the lateral ankle ligament and ACL repair in particular, and a comprehensive review of other surgical applications and techniques is presented. Surg Technol Int. 2015;26:239-255.
41. Henle P, Röder C, Perler G, Heitkemper S, Eggli S. Dynamic intraligamentary stabilization (DIS) for treatment of acute anterior cruciate ligament ruptures: case series experience of the first three years. BMC Musculoskelet Disord. 2015;16:27.
42. Adachi N, Ochi M, Uchio Y, Iwasa J, Ryoke K, Kuriwaka M. Mechanoreceptors in the anterior cruciate ligament contribute to the joint position sense. Acta Orthop Scand. 2002;73(3):330-334.
43. Gao F, Zhou J, He C, et al. A morphologic and quantitative study of mechanoreceptors in the remnant stump of the human anterior cruciate ligament. Arthroscopy. 2016;32(2):273-280.
44. Georgoulis AD, Pappa L, Moebius U, et al. The presence of proprioceptive mechanoreceptors in the remnants of the ruptured ACL as a possible source of re-innervation of the ACL autograft. Knee Surg Sports Traumatol Arthrosc. 2001;9(6):364-368.
45. Fleming BC, Carey JL, Spindler KP, Murray MM. Can suture repair of ACL transection restore normal anteroposterior laxity of the knee? An ex vivo study. J Orthop Res. 2008;26(11):1500-1505.
46. Murray MM, Fleming BC. Use of a bioactive scaffold to stimulate anterior cruciate ligament healing also minimizes posttraumatic osteoarthritis after surgery. Am J Sports Med. 2013;41(8):1762-1770.
47. Ahn JH, Wang JH, Lee YS, Kim JG, Kang JH, Koh KH. Anterior cruciate ligament reconstruction using remnant preservation and a femoral tensioning technique: clinical and magnetic resonance imaging results. Arthroscopy. 2011;27(8):1079-1089.
48. Lee BI, Kwon SW, Kim JB, Choi HS, Min KD. Comparison of clinical results according to amount of preserved remnant in arthroscopic anterior cruciate ligament reconstruction using quadrupled hamstring graft. Arthroscopy. 2008;24(5):560-568.
49. Lee BI, Min KD, Choi HS, et al. Immunohistochemical study of mechanoreceptors in the tibial remnant of the ruptured anterior cruciate ligament in human knees. Knee Surg Sports Traumatol Arthrosc. 2009;17(9):1095-1101.
50. Takahashi T, Kondo E, Yasuda K, et al. Effects of remnant tissue preservation on the tendon graft in anterior cruciate ligament reconstruction: a biomechanical and histological study. Am J Sports Med. 2016;44(7):1708-1716.
51. Dong S, Xie G, Zhang Y, Shen P, Huangfu X, Zhao J. Ligamentization of autogenous hamstring grafts after anterior cruciate ligament reconstruction: midterm versus long-term results. Am J Sports Med. 2015;43(8):1908-1917.
52. Takazawa Y, Ikeda H, Kawasaki T, et al. ACL reconstruction preserving the ACL remnant achieves good clinical outcomes and can reduce subsequent graft rupture. Orthop J Sports Med. 2013;1(4):2325967113505076.
53. Shimodaira H, Tensho K, Akaoka Y, Takanashi S, Kato H, Saito N. Remnant-preserving tibial tunnel positioning using anatomic landmarks in double-bundle anterior cruciate ligament reconstruction. Arthroscopy. 2016;32(9):1822-1830.
54. Zhang Q, Zhang S, Cao X, Liu L, Liu Y, Li R. The effect of remnant preservation on tibial tunnel enlargement in ACL reconstruction with hamstring autograft: a prospective randomized controlled trial. Knee Surg Sports Traumatol Arthrosc. 2014;22(1):166-173.
55. Tie K, Chen L, Hu D, Wang H. The difference in clinical outcome of single-bundle anterior cruciate ligament reconstructions with and without remnant preservation: A meta-analysis. Knee. 2016;23(4):566-574.
56. Robson AW. VI. Ruptured crucial ligaments and their repair by operation. Ann Surg. 1903;37(5):716-718.
57. Palmer I. On the injuries to the ligaments of the knee joint. Acta Orthop Scand. 1938;53.
58. Palmer I. On the injuries to the ligaments of the knee joint: a clinical study. 1938. Clin Orthop Relat Res. 2007;454:17-22.
59 O’Donoghue DH. An analysis of end results of surgical treatment of major injuries to the ligaments of the knee. J Bone Joint Surg Am. 1955;37-A(1):1-13.
60. O’Donoghue DH. Surgical treatment of fresh injuries to the major ligaments of the knee. J Bone Joint Surg Am. 1950;32 A(4):721-738.
61. Feagin JA, Abbott HG, Rokous JR. The isolated tear of the anterior cruciate ligament. J Bone Joint Surg Am. 1972;54-A:1340-1341.
62. England RL. Repair of the ligaments about the knee. Orthop Clin North Am. 1976;7(1):195-204.
63. Marshall JL, Warren RF, Wickiewicz TL, Reider B. The anterior cruciate ligament: a technique of repair and reconstruction. Clin Orthop Relat Res. 1979;(143):97-106.
64. Weaver JK, Derkash RS, Freeman JR, Kirk RE, Oden RR, Matyas J. Primary knee ligament repair--revisited. Clin Orthop Relat Res. 1985;(199):185-191.
65. Nogalski MP, Bach BR Jr. A review of early anterior cruciate ligament surgical repair or reconstruction. Results and caveats. Orthop Rev. 1993;22(11):1213-1223.
66. Feagin JA Jr, Curl WW. Isolated tear of the anterior cruciate ligament: 5-year follow-up study. Am J Sports Med. 1976;4(3):95-100.
67. Marshall JL, Warren RF, Wickiewicz TL. Primary surgical treatment of anterior cruciate ligament lesions. Am J Sports Med. 1982;10(2):103-107.
68. Straub T, Hunter RE. Acute anterior cruciate ligament repair. Clin Orthop Relat Res. 1988;227:238-250.
69. Kaplan N, Wickiewicz TL, Warren RF. Primary surgical treatment of anterior cruciate ligament ruptures. A long-term follow-up study. Am J Sports Med. 1990;18(4):354-358.
70. Sherman MF, Lieber L, Bonamo JR, Podesta L, Reiter I. The long-term followup of primary anterior cruciate ligament repair. Defining a rationale for augmentation. Am J Sports Med. 1991;19(3):243-255.
71. Paar O. Use of semitendinosus tendon to strengthen a freshly repaired anterior cruciate ligament. Chirurg. 1985;56(11):728-734.
72. Aglietti P, Buzzi R, Pisaneschi A, Salvi M. Comparison between suture and augmentation with the semitendinosus tendon in the repair of acute lesions of the anterior cruciate ligament. Ital J Orthop Traumatol. 1986;8(4):217-231.
73. Higgins RW, Steadman JR. Anterior cruciate ligament repairs in world class skiers. Am J Sports Med. 1987;15(5):439-447.
74. Harilainen A, Myllynen P. Treatment of fresh tears of the anterior cruciate ligament. A comparison of primary suture and augmentation with carbon fibre. Injury. 1987;18(6):396-400.
75. Jones KG. Results of use of the central one-third of the patellar ligament to compensate for anterior cruciate ligament deficiency. Clin Orthop Relat Res. 1980;(147):39-44.
76. Puddu G. Method for reconstruction of the anterior cruciate ligament using the semitendinosus tendon. Am J Sports Med. 1980;8(6):402-404.
77. Hefti F, Gächter A, Jenny H, Morscher E. Replacement of the anterior cruciate ligament. a comparative study of four different methods of reconstruction. Arch Orthop Trauma Surg. 1982;100(2):83-94.
78. Odensten M, Hamberg P, Nordin M, Lysholm J, Gillquist J. Surgical or conservative treatment of the acutely torn anterior cruciate ligament. A randomized study with short-term follow-up observations. Clin Orthop Relat Res. 1985;(198):87-93.
79. Andersson C, Odensten M, Good L, Gillquist J. Surgical or non-surgical treatment of acute rupture of the anterior cruciate ligament. A randomized study with long-term follow-up. J Bone Joint Surg Am. 1989;71(7):965-974.
80. Engebretsen L, Benum P, Fasting O, Mølster A, Strand T. A prospective, randomized study of three surgical techniques for treatment of acute ruptures of the anterior cruciate ligament. Am J Sports Med. 1990;18(6):585-590.
81. Jonsson T, Peterson L, Renström P. Anterior cruciate ligament repair with and without augmentation. A prospective 7-year study of 51 patients. Acta Orthop Scand. 1990;61(6):562-566.
82. Andersson C, Odensten M, Gillquist J. Knee function after surgical or nonsurgical treatment of acute rupture of the anterior cruciate ligament: a randomized study with a long-term follow-up period. Clin Orthop Relat Res. 1991;(264):255-263.
83. Heim U, Bachmann B, Infanger K. Reinsertion of the anterior cruciate ligament or primary ligamentous plasty? Helv Chir Acta. 1982;48(5):703-708.
84. Strand T, Engesaeter LB, Mølster AO, et al. Knee function following suture of fresh tear of the anterior cruciate ligament. Acta Orthop Scand. 1984;55(2):181-184.
85. Marcacci M, Spinelli M, Chiellini F, Buccolieri V. Notes on 53 cases of immediate suture of acute lesions of the anterior cruciate ligament. Ital J Orthop Traumatol. 1985;7(2):69-79.
86. van der List JP, DiFelice GS. Primary repair of the anterior cruciate ligament: a paradigm shift. Surgeon. 2016 Oct 6. [Epub ahead of print]
87. Bräm J, Plaschy S, Lütolf M, Leutenegger A. [The primary cruciate ligament suture--is the method outdated? Results in follow-up of 58 patients]. Z Unfallchir Versicherungsmed. 1994;87(2):91-109.
88. Genelin F, Trost A, Primavesi C, Knoll P. Late results following proximal reinsertion of isolated ruptured ACL ligaments. Knee Surg Sports Traumatol Arthrosc. 1993;1(1):17-19.
89. Kühne JH, Theermann R, Neumann R, Sagasser J. [Acute uncomplicated anterior knee instability. 2-5 year follow-up of surgical treatment]. Unfallchirurg. 1991;94(2):81-87.
90. Simonet WT, Sim FH. Repair and reconstruction of rotatory instability of the knee. Am J Sports Med. 1984;12(2):89-97.
91. Raunest J, Derra E, Ohmann C. [Clinical results of Palmer’s primary cruciate ligament insertion without augmentation]. Unfallchirurgie. 1991;17(3):166-174.
92. Frank C, Beaver P, Rademaker F, Becker K, Schachar N, Edwards G. A computerized study of knee-ligament injuries: repair versus removal of the torn anterior cruciate ligament. Can J Surg. 1982;25(4):454-458.
93. Enneking WF, Horowitz M. The intra-articular effects of immobilization on the human knee. J Bone Joint Surg Am. 1972;54(5):973-985.
94. Millett PJ, Wickiewicz TL, Warren RF. Motion loss after ligament injuries to the knee. Part I: causes. Am J Sports Med. 2001;29(5):664-675.
95. Bilko TE, Paulos LE, Feagin JA Jr, Lambert KL, Cunningham HR. Current trends in repair and rehabilitation of complete (acute) anterior cruciate ligament injuries. Analysis of 1984 questionnaire completed by ACL Study Group. Am J Sports Med. 1986;14(2):143-147.
96. Paulos L, Noyes FR, Grood E, Butler DL. Knee rehabilitation after anterior cruciate ligament reconstruction and repair. J Orthop Sports Phys Ther. 1991;13(2):60-70.
97. Paessler HH, Deneke J, Dahners LE. Augmented repair and early mobilization of acute anterior cruciate ligament injuries. Am J Sports Med. 1992;20(6):667-674.
98.
99. Kdolsky RK, Gibbons DF, Kwasny O, Schabus R, Plenk H Jr. Braided polypropylene augmentation device in reconstructive surgery of the anterior cruciate ligament: long-term clinical performance of 594 patients and short-term arthroscopic results, failure analysis by scanning electron microscopy, and synovial histomorphology. J Orthop Res. 1997;15(1):1-10.
100. Grøntvedt T, Engebretsen L. Comparison between two techniques for surgical repair of the acutely torn anterior cruciate ligament. A prospective, randomized follow-up study of 48 patients. Scand J Med Sci Sports. 1995;5(6):358-363.
101. Hehl G, Strecker W, Richter M, Kiefer H, Wissmeyer T. Clinical experience with PDS II augmentation for operative treatment of acute proximal ACL ruptures--2-year follow-up. Knee Surg Sports Traumatol Arthrosc. 1999;7(2):102-106.
102. Schenk S, Landsiedl F, Enenkel M. Arthroscopic single-stranded semitendinosus tendon- versus PDS-augmentation of reinserted acute femoral anterior cruciate ligament tears: 7 year follow-up study. Knee Surg Sports Traumatol Arthrosc. 2006;14(4):318-324.
103. Zysk SP, Refior HJ. Operative or conservative treatment of the acutely torn anterior cruciate ligament in middle-aged patients. A follow-up study of 133 patients between the ages of 40 and 59 years. Arch Orthop Trauma Surg. 2000;120(1-2):59-64.
104. Krueger-Franke M, Siebert CH, Schupp A. Refixation of femoral anterior cruciate ligament tears combined with a semitendinosus tendon augmentation. Technique and results. Arch Orthop Trauma Surg. 1998;117(1-2):68-72.
105. Natri A, Järvinen M, Kannus P. Primary repair plus intra-articular iliotibial band augmentation in the treatment of an acute anterior cruciate ligament rupture. A follow-up study of 70 patients. Arch Orthop Trauma Surg. 1996;115(1):22-27.
106. Träger D, Pohle K, Tschirner W. Anterior cruciate ligament suture in comparison with plasty. A 5-year follow-up study. Arch Orthop Trauma Surg. 1995;114(5):278-280.
107. Shelbourne KD, Wilckens JH, Mollabashy A, DeCarlo M. Arthrofibrosis in acute anterior cruciate ligament reconstruction. The effect of timing of reconstruction and rehabilitation. Am J Sports Med. 1991;19(4):332-336.
108. Volokhina YV, Syed HM, Pham PH, Blackburn AK. Two helpful MRI signs for evaluation of posterolateral bundle tears of the anterior cruciate ligament: a pilot study. Orthop J Sports Med. 2015;3(8):2325967115597641.
109. Strand T, Mølster A, Hordvik M, Krukhaug Y. Long-term follow-up after primary repair of the anterior cruciate ligament: clinical and radiological evaluation 15-23 years postoperatively. Arch Orthop Trauma Surg. 2005;125(4):217-221.
110. van der List JP, DiFelice GS. Successful arthroscopic primary repair of a chronic anterior cruciate ligament tear 11 years following injury. HSS J. 2016. In press.
111. van der List JP, DiFelice GS. The role of ligament repair in anterior cruciate ligament surgery. In: Mascarenhas R, Bhatia S, Lowe WR, eds. Ligamentous Injuries of the Knee. 1st ed. Houston: Nova Science Publishers; 2016:199-220.
112. van der List JP, DiFelice GS. Gap formation following primary anterior cruciate ligament repair: a biomechanical study. Knee. 2016. In press.
113. DiFelice GS, van der List JP. Arthroscopic primary repair of proximal anterior cruciate ligament tears. Arthrosc Tech. 2016. In press.
114. Smith JO, Yasen SK, Palmer HC, Lord BR, Britton EM, Wilson AJ. Paediatric ACL repair reinforced with temporary internal bracing. Knee Surg Sports Traumatol Arthrosc. 2016;24(6):1845-1851.
115. Magarian EM, Fleming BC, Harrison SL, Mastrangelo AN, Badger GJ, Murray MM. Delay of 2 or 6 weeks adversely affects the functional outcome of augmented primary repair of the porcine anterior cruciate ligament. Am J Sports Med. 2010;38(12):2528-2534.
116. Murray MM, Magarian EM, Harrison SL, Mastrangelo AN, Zurakowski D, Fleming BC. The effect of skeletal maturity on functional healing of the anterior cruciate ligament. J Bone Joint Surg Am. 2010;92(11):2039-2049.
117. Werner BC, Yang S, Looney AM, Gwathmey FW Jr. Trends in pediatric and adolescent anterior cruciate ligament iInjury and reconstruction. J Pediatr Orthop. 2016;36(5):447-452.
118. Frosch KH, Stengel D, Brodhun T, et al. Outcomes and risks of operative treatment of rupture of the anterior cruciate ligament in children and adolescents. Arthroscopy. 2010;26(11):1539-1550.
119. Ramski DE, Kanj WW, Franklin CC, Baldwin KD, Ganley TJ. Anterior cruciate ligament tears in children and adolescents: a meta-analysis of nonoperative versus operative treatment. Am J Sports Med. 2014;42(11):2769-2776.
120. Zantop T, Brucker PU, Vidal A, Zelle BA, Fu FH. Intraarticular rupture pattern of the ACL. Clin Orthop Relat Res. 2007;454:48-53.
121. Yoon KH, Bae DK, Cho SM, Park SY, Lee JH. Standard anterior cruciate ligament reconstruction versus isolated single-bundle augmentation with hamstring autograft. Arthroscopy. 2009;25(11):1265-1274.
122 Demirağ B, Ermutlu C, Aydemir F, Durak K. A comparison of clinical outcome of augmentation and standard reconstruction techniques for partial anterior cruciate ligament tears. Eklem Hastalik Cerrahisi. 2012;23(3):140-144.
123. Sonnery-Cottet B, Zayni R, Conteduca J, et al. Posterolateral bundle reconstruction with anteromedial bundle remnant preservation in ACL tears: clinical and MRI evaluation of 39 patients with 24-month follow-up. Orthop J Sports Med. 2013;1(3):2325967113501624.
124. Sabat D, Kumar V. Partial tears of anterior cruciate ligament: results of single bundle augmentation. Indian J Orthop. 2015;49(2):129-135.
125. Jung YB, Jung HJ, Siti HT, et al. Comparison of anterior cruciate ligament reconstruction with preservation only versus remnant tensioning technique. Arthroscopy. 2011;27(9):1252-1258.
126. Nguyen DT, Ramwadhdoebe TH, van der Hart CP, Blankevoort L, Tak PP, van Dijk CN. Intrinsic healing response of the human anterior cruciate ligament: an histological study of reattached ACL remnants. J Orthop Res. 2014;32(2):296-301.
127. Boutsiadis A, Karampalis C, Tzavelas A, Vraggalas V, Christodoulou P, Bisbinas I. Anterior cruciate ligament remnant-preserving reconstruction using a “lasso-loop” knot configuration. Arthrosc Tech. 2015;4(6):e741-e746.
128. Noh JH, Yoon KH, Song SJ, Roh YH. Re-tensioning technique to cover the graft with remnant in anterior cruciate ligament reconstruction. Arthrosc Tech. 2014;3(6):e679-e682.
129. Hong L, Li X, Zhang H, et al. Anterior cruciate ligament reconstruction with remnant preservation: a prospective, randomized controlled study. Am J Sports Med. 2012;40(12):2747-2755.
130. Noh JH, Kyung HS, Roh YH, Kang TS. Remnant-preserving and re-tensioning technique to cover the graft in anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2015 Nov 12. [Epub ahead of print]
131. Muneta T, Koga H, Ju YJ, Horie M, Nakamura T, Sekiya I. Remnant volume of anterior cruciate ligament correlates preoperative patients’ status and postoperative outcome. Knee Surg Sports Traumatol Arthrosc. 2013;21(4):906-913.
132. Leeberg V, Lekdorf J, Wong C, Sonne-Holm S. Tibial eminentia avulsion fracture in children - a systematic review of the current literature. Dan Med J. 2014;61(3):A4792.
133. In Y, Kwak DS, Moon CW, Han SH, Choi NY. Biomechanical comparison of three techniques for fixation of tibial avulsion fractures of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 2012;20(8):1470-1478.
1. Mall NA, Chalmers PN, Moric M, et al. Incidence and trends of anterior cruciate ligament reconstruction in the United States. Am J Sports Med. 2014;42(10):2363-2370.
2. Sanders TL, Maradit Kremers H, Bryan AJ, et al. Incidence of anterior cruciate ligament tears and reconstruction: a 21-year population-based study. Am J Sports Med. 2016;44(6):1502-1507.
3. Ciccotti MG, Lombardo SJ, Nonweiler B, Pink M. Non-operative treatment of ruptures of the anterior cruciate ligament in middle-aged patients. Results after long-term follow-up. J Bone Joint Surg Am. 1994;76(9):1315-1321.
4. Sanders TL, Pareek A, Kremers HM, et al. Long-term follow-up of isolated ACL tears treated without ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2016 May 24. [Epub ahead of print]
5. Irarrázaval S, Kurosaka M, Cohen M, Fu FH. Anterior cruciate ligament reconstruction. J ISAKOS. 2016;1(1):38-52.
6. Gabler CM, Jacobs CA, Howard JS, Mattacola CG, Johnson DL. Comparison of graft failure rate between autografts placed via an anatomic anterior cruciate ligament reconstruction technique: a systematic review, meta-analysis, and meta-regression. Am J Sports Med. 2016;44(4):1069-1079.
7. Li S, Chen Y, Lin Z, Cui W, Zhao J, Su W. A systematic review of randomized controlled clinical trials comparing hamstring autografts versus bone-patellar tendon-bone autografts for the reconstruction of the anterior cruciate ligament. Arch Orthop Trauma Surg. 2012;132(9):1287-1297.
8. Rahr-Wagner L, Thillemann TM, Pedersen AB, Lind M. Comparison of hamstring tendon and patellar tendon grafts in anterior cruciate ligament reconstruction in a nationwide population-based cohort study: results from the danish registry of knee ligament reconstruction. Am J Sports Med. 2014;42(2):278-284.
9. Xie X, Liu X, Chen Z, Yu Y, Peng S, Li Q. A meta-analysis of bone-patellar tendon-bone autograft versus four-strand hamstring tendon autograft for anterior cruciate ligament reconstruction. Knee. 2015;22(2):100-110.
10. Andernord D, Desai N, Björnsson H, Gillén S, Karlsson J, Samuelsson K. Predictors of contralateral anterior cruciate ligament reconstruction: a cohort study of 9061 patients with 5-year follow-up. Am J Sports Med. 2015;43(2):295-302.
11. Maletis GB, Inacio MC, Funahashi TT. Risk factors associated with revision and contralateral anterior cruciate ligament reconstructions in the Kaiser Permanente ACLR registry. Am J Sports Med. 2015;43(3):641-647.
12. Kim SJ, Postigo R, Koo S, Kim JH. Infection after arthroscopic anterior cruciate ligament reconstruction. Orthopedics. 2014;37(7):477-484.
13. Makhni EC, Steinhaus ME, Mehran N, Schulz BS, Ahmad CS. Functional outcome and graft retention in patients with septic arthritis after anterior cruciate ligament reconstruction: a systematic review. Arthroscopy. 2015;31(7):1392-1401.
14. Kocher MS, Steadman JR, Briggs K, Zurakowski D, Sterett WI, Hawkins RJ. Determinants of patient satisfaction with outcome after anterior cruciate ligament reconstruction. J Bone Joint Surg Am. 2002;84-A(9):1560-1572.
15. Ardern CL, Österberg A, Sonesson S, Gauffin H, Webster KE, Kvist J. Satisfaction with knee function after primary anterior cruciate ligament reconstruction is associated with self-efficacy, quality of life, and returning to the preinjury physical activity. Arthroscopy. 2016;32(8):1631-1638.e3.
16. Grant JA, Mohtadi NG, Maitland ME, Zernicke RF. Comparison of home versus physical therapy-supervised rehabilitation programs after anterior cruciate ligament reconstruction: a randomized clinical trial. Am J Sports Med. 2005;33(9):1288-1297.
17. Lindström M, Strandberg S, Wredmark T, Fell änder-Tsai L, Henriksson M. Functional and muscle morphometric effects of ACL reconstruction. A prospective CT study with 1 year follow-up. Scand J Med Sci Sports. 2013;23(4):431-442.
18. Biau DJ, Tournoux C, Katsahian S, Schranz PJ, Nizard RS. Bone-patellar tendon-bone autografts versus hamstring autografts for reconstruction of anterior cruciate ligament: meta-analysis. BMJ. 2006;332(7548):995-1001.
19. Spindler KP, Kuhn JE, Freedman KB, Matthews CE, Dittus RS, Harrell FE Jr. Anterior cruciate ligament reconstruction autograft choice: bone-tendon-bone versus hamstring: does it really matter? A systematic review. Am J Sports Med. 2004;32(8):1986-1995.
20. Aga C, Wilson KJ, Johansen S, Dornan G, La Prade RF, Engebretsen L. Tunnel widening in single- versus double-bundle anterior cruciate ligament reconstructed knees. Knee Surg Sports Traumatol Arthrosc. 2016 Jun 21. [Epub ahead of print]
21. Maak TG, Voos JE, Wickiewicz TL, Warren RF. Tunnel widening in revision anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2010;18(11):695-706.
22. Cheatham SA, Johnson DL. Anticipating problems unique to revision ACL surgery. Sports Med Arthrosc. 2013;21(2):129-134.
23. Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39(1):199-217.
24. Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531-536.
25. Andriolo L, Filardo G, Kon E, et al. Revision anterior cruciate ligament reconstruction: clinical outcome and evidence for return to sport. Knee Surg Sports Traumatol Arthrosc. 2015;23(10):2825-2845.
26. Grassi A, Ardern CL, Marcheggiani Muccioli GM, Neri MP, Marcacci M, Zaffagnini S. Does revision ACL reconstruction measure up to primary surgery? A meta-analysis comparing patient-reported and clinician-reported outcomes, and radiographic results. Br J Sports Med. 2016;50(12):716-724.
27. Ristanis S, Stergiou N, Patras K, Vasiliadis HS, Giakas G, Georgoulis AD. Excessive tibial rotation during high-demand activities is not restored by anterior cruciate ligament reconstruction. Arthroscopy. 2005;21(11):1323-1329.
28. Andriacchi TP, Mündermann A, Smith RL, Alexander EJ, Dyrby CO, Koo S. A framework for the in vivo pathomechanics of osteoarthritis at the knee. Ann Biomed Eng. 2004;32(3):447-457.
29. Imhauser C, Mauro C, Choi D, et al. Abnormal tibiofemoral contact stress and its association with altered kinematics after center-center anterior cruciate ligament reconstruction: an in vitro study. Am J Sports Med. 2013;41(4):815-825.
30. Ajuied A, Wong F, Smith C, et al. Anterior cruciate ligament injury and radiologic progression of knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2014;42(9):2242-2252.
31. Chalmers PN, Mall NA, Moric M, et al. Does ACL reconstruction alter natural history?: A systematic literature review of long-term outcomes. J Bone Joint Surg Am. 2014;96(4):292-300.
32. DiFelice GS, Villegas C, Taylor SA. Anterior cruciate ligament preservation: early results of a novel arthroscopic technique for suture anchor primary anterior cruciate ligament repair. Arthroscopy. 2015;31(11):2162-2171.
33. Eggli S, Kohlhof H, Zumstein M, et al. Dynamic intraligamentary stabilization: novel technique for preserving the ruptured ACL. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):1215-1221.
34. Buda R, Ferruzzi A, Vannini F, Zambelli L, Di Caprio F. Augmentation technique with semitendinosus and gracilis tendons in chronic partial lesions of the ACL: clinical and arthrometric analysis. Knee Surg Sports Traumatol Arthrosc. 2006;14(11):1101-1107.
35. Ochi M, Adachi N, Uchio Y, et al. A minimum 2-year follow-up after selective anteromedial or posterolateral bundle anterior cruciate ligament reconstruction. Arthroscopy. 2009;25(2):117-122.
36. Ahn JH, Lee YS, Ha HC. Anterior cruciate ligament reconstruction with preservation of remnant bundle using hamstring autograft: technical note. Arch Orthop Trauma Surg. 2009;129(8):1011-1015.
37. Lee BI, Min KD, Choi HS, Kim JB, Kim ST. Arthroscopic anterior cruciate ligament reconstruction with the tibial-remnant preserving technique using a hamstring graft. Arthroscopy. 2006;22(3):340.e1-e7.
38. Achtnich A, Herbst E, Forkel P, et al. Acute proximal anterior cruciate ligament tears: outcomes after arthroscopic suture anchor repair versus anatomic single-bundle reconstruction. Arthroscopy. 2016 Jun 17. [Epub ahead of print]
39. MacKay G, Anthony IC, Jenkins PJ, Blyth M. Anterior cruciate ligament repair revisited. Preliminary results of primary repair with internal brace ligament augmentation: a case series. Orthop Muscul Syst. 2015;4:188.
40. Mackay GM, Blyth MJ, Anthony I, Hopper GP, Ribbans WJ. A review of ligament augmentation with the InternalBrace™: the surgical principle is described for the lateral ankle ligament and ACL repair in particular, and a comprehensive review of other surgical applications and techniques is presented. Surg Technol Int. 2015;26:239-255.
41. Henle P, Röder C, Perler G, Heitkemper S, Eggli S. Dynamic intraligamentary stabilization (DIS) for treatment of acute anterior cruciate ligament ruptures: case series experience of the first three years. BMC Musculoskelet Disord. 2015;16:27.
42. Adachi N, Ochi M, Uchio Y, Iwasa J, Ryoke K, Kuriwaka M. Mechanoreceptors in the anterior cruciate ligament contribute to the joint position sense. Acta Orthop Scand. 2002;73(3):330-334.
43. Gao F, Zhou J, He C, et al. A morphologic and quantitative study of mechanoreceptors in the remnant stump of the human anterior cruciate ligament. Arthroscopy. 2016;32(2):273-280.
44. Georgoulis AD, Pappa L, Moebius U, et al. The presence of proprioceptive mechanoreceptors in the remnants of the ruptured ACL as a possible source of re-innervation of the ACL autograft. Knee Surg Sports Traumatol Arthrosc. 2001;9(6):364-368.
45. Fleming BC, Carey JL, Spindler KP, Murray MM. Can suture repair of ACL transection restore normal anteroposterior laxity of the knee? An ex vivo study. J Orthop Res. 2008;26(11):1500-1505.
46. Murray MM, Fleming BC. Use of a bioactive scaffold to stimulate anterior cruciate ligament healing also minimizes posttraumatic osteoarthritis after surgery. Am J Sports Med. 2013;41(8):1762-1770.
47. Ahn JH, Wang JH, Lee YS, Kim JG, Kang JH, Koh KH. Anterior cruciate ligament reconstruction using remnant preservation and a femoral tensioning technique: clinical and magnetic resonance imaging results. Arthroscopy. 2011;27(8):1079-1089.
48. Lee BI, Kwon SW, Kim JB, Choi HS, Min KD. Comparison of clinical results according to amount of preserved remnant in arthroscopic anterior cruciate ligament reconstruction using quadrupled hamstring graft. Arthroscopy. 2008;24(5):560-568.
49. Lee BI, Min KD, Choi HS, et al. Immunohistochemical study of mechanoreceptors in the tibial remnant of the ruptured anterior cruciate ligament in human knees. Knee Surg Sports Traumatol Arthrosc. 2009;17(9):1095-1101.
50. Takahashi T, Kondo E, Yasuda K, et al. Effects of remnant tissue preservation on the tendon graft in anterior cruciate ligament reconstruction: a biomechanical and histological study. Am J Sports Med. 2016;44(7):1708-1716.
51. Dong S, Xie G, Zhang Y, Shen P, Huangfu X, Zhao J. Ligamentization of autogenous hamstring grafts after anterior cruciate ligament reconstruction: midterm versus long-term results. Am J Sports Med. 2015;43(8):1908-1917.
52. Takazawa Y, Ikeda H, Kawasaki T, et al. ACL reconstruction preserving the ACL remnant achieves good clinical outcomes and can reduce subsequent graft rupture. Orthop J Sports Med. 2013;1(4):2325967113505076.
53. Shimodaira H, Tensho K, Akaoka Y, Takanashi S, Kato H, Saito N. Remnant-preserving tibial tunnel positioning using anatomic landmarks in double-bundle anterior cruciate ligament reconstruction. Arthroscopy. 2016;32(9):1822-1830.
54. Zhang Q, Zhang S, Cao X, Liu L, Liu Y, Li R. The effect of remnant preservation on tibial tunnel enlargement in ACL reconstruction with hamstring autograft: a prospective randomized controlled trial. Knee Surg Sports Traumatol Arthrosc. 2014;22(1):166-173.
55. Tie K, Chen L, Hu D, Wang H. The difference in clinical outcome of single-bundle anterior cruciate ligament reconstructions with and without remnant preservation: A meta-analysis. Knee. 2016;23(4):566-574.
56. Robson AW. VI. Ruptured crucial ligaments and their repair by operation. Ann Surg. 1903;37(5):716-718.
57. Palmer I. On the injuries to the ligaments of the knee joint. Acta Orthop Scand. 1938;53.
58. Palmer I. On the injuries to the ligaments of the knee joint: a clinical study. 1938. Clin Orthop Relat Res. 2007;454:17-22.
59 O’Donoghue DH. An analysis of end results of surgical treatment of major injuries to the ligaments of the knee. J Bone Joint Surg Am. 1955;37-A(1):1-13.
60. O’Donoghue DH. Surgical treatment of fresh injuries to the major ligaments of the knee. J Bone Joint Surg Am. 1950;32 A(4):721-738.
61. Feagin JA, Abbott HG, Rokous JR. The isolated tear of the anterior cruciate ligament. J Bone Joint Surg Am. 1972;54-A:1340-1341.
62. England RL. Repair of the ligaments about the knee. Orthop Clin North Am. 1976;7(1):195-204.
63. Marshall JL, Warren RF, Wickiewicz TL, Reider B. The anterior cruciate ligament: a technique of repair and reconstruction. Clin Orthop Relat Res. 1979;(143):97-106.
64. Weaver JK, Derkash RS, Freeman JR, Kirk RE, Oden RR, Matyas J. Primary knee ligament repair--revisited. Clin Orthop Relat Res. 1985;(199):185-191.
65. Nogalski MP, Bach BR Jr. A review of early anterior cruciate ligament surgical repair or reconstruction. Results and caveats. Orthop Rev. 1993;22(11):1213-1223.
66. Feagin JA Jr, Curl WW. Isolated tear of the anterior cruciate ligament: 5-year follow-up study. Am J Sports Med. 1976;4(3):95-100.
67. Marshall JL, Warren RF, Wickiewicz TL. Primary surgical treatment of anterior cruciate ligament lesions. Am J Sports Med. 1982;10(2):103-107.
68. Straub T, Hunter RE. Acute anterior cruciate ligament repair. Clin Orthop Relat Res. 1988;227:238-250.
69. Kaplan N, Wickiewicz TL, Warren RF. Primary surgical treatment of anterior cruciate ligament ruptures. A long-term follow-up study. Am J Sports Med. 1990;18(4):354-358.
70. Sherman MF, Lieber L, Bonamo JR, Podesta L, Reiter I. The long-term followup of primary anterior cruciate ligament repair. Defining a rationale for augmentation. Am J Sports Med. 1991;19(3):243-255.
71. Paar O. Use of semitendinosus tendon to strengthen a freshly repaired anterior cruciate ligament. Chirurg. 1985;56(11):728-734.
72. Aglietti P, Buzzi R, Pisaneschi A, Salvi M. Comparison between suture and augmentation with the semitendinosus tendon in the repair of acute lesions of the anterior cruciate ligament. Ital J Orthop Traumatol. 1986;8(4):217-231.
73. Higgins RW, Steadman JR. Anterior cruciate ligament repairs in world class skiers. Am J Sports Med. 1987;15(5):439-447.
74. Harilainen A, Myllynen P. Treatment of fresh tears of the anterior cruciate ligament. A comparison of primary suture and augmentation with carbon fibre. Injury. 1987;18(6):396-400.
75. Jones KG. Results of use of the central one-third of the patellar ligament to compensate for anterior cruciate ligament deficiency. Clin Orthop Relat Res. 1980;(147):39-44.
76. Puddu G. Method for reconstruction of the anterior cruciate ligament using the semitendinosus tendon. Am J Sports Med. 1980;8(6):402-404.
77. Hefti F, Gächter A, Jenny H, Morscher E. Replacement of the anterior cruciate ligament. a comparative study of four different methods of reconstruction. Arch Orthop Trauma Surg. 1982;100(2):83-94.
78. Odensten M, Hamberg P, Nordin M, Lysholm J, Gillquist J. Surgical or conservative treatment of the acutely torn anterior cruciate ligament. A randomized study with short-term follow-up observations. Clin Orthop Relat Res. 1985;(198):87-93.
79. Andersson C, Odensten M, Good L, Gillquist J. Surgical or non-surgical treatment of acute rupture of the anterior cruciate ligament. A randomized study with long-term follow-up. J Bone Joint Surg Am. 1989;71(7):965-974.
80. Engebretsen L, Benum P, Fasting O, Mølster A, Strand T. A prospective, randomized study of three surgical techniques for treatment of acute ruptures of the anterior cruciate ligament. Am J Sports Med. 1990;18(6):585-590.
81. Jonsson T, Peterson L, Renström P. Anterior cruciate ligament repair with and without augmentation. A prospective 7-year study of 51 patients. Acta Orthop Scand. 1990;61(6):562-566.
82. Andersson C, Odensten M, Gillquist J. Knee function after surgical or nonsurgical treatment of acute rupture of the anterior cruciate ligament: a randomized study with a long-term follow-up period. Clin Orthop Relat Res. 1991;(264):255-263.
83. Heim U, Bachmann B, Infanger K. Reinsertion of the anterior cruciate ligament or primary ligamentous plasty? Helv Chir Acta. 1982;48(5):703-708.
84. Strand T, Engesaeter LB, Mølster AO, et al. Knee function following suture of fresh tear of the anterior cruciate ligament. Acta Orthop Scand. 1984;55(2):181-184.
85. Marcacci M, Spinelli M, Chiellini F, Buccolieri V. Notes on 53 cases of immediate suture of acute lesions of the anterior cruciate ligament. Ital J Orthop Traumatol. 1985;7(2):69-79.
86. van der List JP, DiFelice GS. Primary repair of the anterior cruciate ligament: a paradigm shift. Surgeon. 2016 Oct 6. [Epub ahead of print]
87. Bräm J, Plaschy S, Lütolf M, Leutenegger A. [The primary cruciate ligament suture--is the method outdated? Results in follow-up of 58 patients]. Z Unfallchir Versicherungsmed. 1994;87(2):91-109.
88. Genelin F, Trost A, Primavesi C, Knoll P. Late results following proximal reinsertion of isolated ruptured ACL ligaments. Knee Surg Sports Traumatol Arthrosc. 1993;1(1):17-19.
89. Kühne JH, Theermann R, Neumann R, Sagasser J. [Acute uncomplicated anterior knee instability. 2-5 year follow-up of surgical treatment]. Unfallchirurg. 1991;94(2):81-87.
90. Simonet WT, Sim FH. Repair and reconstruction of rotatory instability of the knee. Am J Sports Med. 1984;12(2):89-97.
91. Raunest J, Derra E, Ohmann C. [Clinical results of Palmer’s primary cruciate ligament insertion without augmentation]. Unfallchirurgie. 1991;17(3):166-174.
92. Frank C, Beaver P, Rademaker F, Becker K, Schachar N, Edwards G. A computerized study of knee-ligament injuries: repair versus removal of the torn anterior cruciate ligament. Can J Surg. 1982;25(4):454-458.
93. Enneking WF, Horowitz M. The intra-articular effects of immobilization on the human knee. J Bone Joint Surg Am. 1972;54(5):973-985.
94. Millett PJ, Wickiewicz TL, Warren RF. Motion loss after ligament injuries to the knee. Part I: causes. Am J Sports Med. 2001;29(5):664-675.
95. Bilko TE, Paulos LE, Feagin JA Jr, Lambert KL, Cunningham HR. Current trends in repair and rehabilitation of complete (acute) anterior cruciate ligament injuries. Analysis of 1984 questionnaire completed by ACL Study Group. Am J Sports Med. 1986;14(2):143-147.
96. Paulos L, Noyes FR, Grood E, Butler DL. Knee rehabilitation after anterior cruciate ligament reconstruction and repair. J Orthop Sports Phys Ther. 1991;13(2):60-70.
97. Paessler HH, Deneke J, Dahners LE. Augmented repair and early mobilization of acute anterior cruciate ligament injuries. Am J Sports Med. 1992;20(6):667-674.
98.
99. Kdolsky RK, Gibbons DF, Kwasny O, Schabus R, Plenk H Jr. Braided polypropylene augmentation device in reconstructive surgery of the anterior cruciate ligament: long-term clinical performance of 594 patients and short-term arthroscopic results, failure analysis by scanning electron microscopy, and synovial histomorphology. J Orthop Res. 1997;15(1):1-10.
100. Grøntvedt T, Engebretsen L. Comparison between two techniques for surgical repair of the acutely torn anterior cruciate ligament. A prospective, randomized follow-up study of 48 patients. Scand J Med Sci Sports. 1995;5(6):358-363.
101. Hehl G, Strecker W, Richter M, Kiefer H, Wissmeyer T. Clinical experience with PDS II augmentation for operative treatment of acute proximal ACL ruptures--2-year follow-up. Knee Surg Sports Traumatol Arthrosc. 1999;7(2):102-106.
102. Schenk S, Landsiedl F, Enenkel M. Arthroscopic single-stranded semitendinosus tendon- versus PDS-augmentation of reinserted acute femoral anterior cruciate ligament tears: 7 year follow-up study. Knee Surg Sports Traumatol Arthrosc. 2006;14(4):318-324.
103. Zysk SP, Refior HJ. Operative or conservative treatment of the acutely torn anterior cruciate ligament in middle-aged patients. A follow-up study of 133 patients between the ages of 40 and 59 years. Arch Orthop Trauma Surg. 2000;120(1-2):59-64.
104. Krueger-Franke M, Siebert CH, Schupp A. Refixation of femoral anterior cruciate ligament tears combined with a semitendinosus tendon augmentation. Technique and results. Arch Orthop Trauma Surg. 1998;117(1-2):68-72.
105. Natri A, Järvinen M, Kannus P. Primary repair plus intra-articular iliotibial band augmentation in the treatment of an acute anterior cruciate ligament rupture. A follow-up study of 70 patients. Arch Orthop Trauma Surg. 1996;115(1):22-27.
106. Träger D, Pohle K, Tschirner W. Anterior cruciate ligament suture in comparison with plasty. A 5-year follow-up study. Arch Orthop Trauma Surg. 1995;114(5):278-280.
107. Shelbourne KD, Wilckens JH, Mollabashy A, DeCarlo M. Arthrofibrosis in acute anterior cruciate ligament reconstruction. The effect of timing of reconstruction and rehabilitation. Am J Sports Med. 1991;19(4):332-336.
108. Volokhina YV, Syed HM, Pham PH, Blackburn AK. Two helpful MRI signs for evaluation of posterolateral bundle tears of the anterior cruciate ligament: a pilot study. Orthop J Sports Med. 2015;3(8):2325967115597641.
109. Strand T, Mølster A, Hordvik M, Krukhaug Y. Long-term follow-up after primary repair of the anterior cruciate ligament: clinical and radiological evaluation 15-23 years postoperatively. Arch Orthop Trauma Surg. 2005;125(4):217-221.
110. van der List JP, DiFelice GS. Successful arthroscopic primary repair of a chronic anterior cruciate ligament tear 11 years following injury. HSS J. 2016. In press.
111. van der List JP, DiFelice GS. The role of ligament repair in anterior cruciate ligament surgery. In: Mascarenhas R, Bhatia S, Lowe WR, eds. Ligamentous Injuries of the Knee. 1st ed. Houston: Nova Science Publishers; 2016:199-220.
112. van der List JP, DiFelice GS. Gap formation following primary anterior cruciate ligament repair: a biomechanical study. Knee. 2016. In press.
113. DiFelice GS, van der List JP. Arthroscopic primary repair of proximal anterior cruciate ligament tears. Arthrosc Tech. 2016. In press.
114. Smith JO, Yasen SK, Palmer HC, Lord BR, Britton EM, Wilson AJ. Paediatric ACL repair reinforced with temporary internal bracing. Knee Surg Sports Traumatol Arthrosc. 2016;24(6):1845-1851.
115. Magarian EM, Fleming BC, Harrison SL, Mastrangelo AN, Badger GJ, Murray MM. Delay of 2 or 6 weeks adversely affects the functional outcome of augmented primary repair of the porcine anterior cruciate ligament. Am J Sports Med. 2010;38(12):2528-2534.
116. Murray MM, Magarian EM, Harrison SL, Mastrangelo AN, Zurakowski D, Fleming BC. The effect of skeletal maturity on functional healing of the anterior cruciate ligament. J Bone Joint Surg Am. 2010;92(11):2039-2049.
117. Werner BC, Yang S, Looney AM, Gwathmey FW Jr. Trends in pediatric and adolescent anterior cruciate ligament iInjury and reconstruction. J Pediatr Orthop. 2016;36(5):447-452.
118. Frosch KH, Stengel D, Brodhun T, et al. Outcomes and risks of operative treatment of rupture of the anterior cruciate ligament in children and adolescents. Arthroscopy. 2010;26(11):1539-1550.
119. Ramski DE, Kanj WW, Franklin CC, Baldwin KD, Ganley TJ. Anterior cruciate ligament tears in children and adolescents: a meta-analysis of nonoperative versus operative treatment. Am J Sports Med. 2014;42(11):2769-2776.
120. Zantop T, Brucker PU, Vidal A, Zelle BA, Fu FH. Intraarticular rupture pattern of the ACL. Clin Orthop Relat Res. 2007;454:48-53.
121. Yoon KH, Bae DK, Cho SM, Park SY, Lee JH. Standard anterior cruciate ligament reconstruction versus isolated single-bundle augmentation with hamstring autograft. Arthroscopy. 2009;25(11):1265-1274.
122 Demirağ B, Ermutlu C, Aydemir F, Durak K. A comparison of clinical outcome of augmentation and standard reconstruction techniques for partial anterior cruciate ligament tears. Eklem Hastalik Cerrahisi. 2012;23(3):140-144.
123. Sonnery-Cottet B, Zayni R, Conteduca J, et al. Posterolateral bundle reconstruction with anteromedial bundle remnant preservation in ACL tears: clinical and MRI evaluation of 39 patients with 24-month follow-up. Orthop J Sports Med. 2013;1(3):2325967113501624.
124. Sabat D, Kumar V. Partial tears of anterior cruciate ligament: results of single bundle augmentation. Indian J Orthop. 2015;49(2):129-135.
125. Jung YB, Jung HJ, Siti HT, et al. Comparison of anterior cruciate ligament reconstruction with preservation only versus remnant tensioning technique. Arthroscopy. 2011;27(9):1252-1258.
126. Nguyen DT, Ramwadhdoebe TH, van der Hart CP, Blankevoort L, Tak PP, van Dijk CN. Intrinsic healing response of the human anterior cruciate ligament: an histological study of reattached ACL remnants. J Orthop Res. 2014;32(2):296-301.
127. Boutsiadis A, Karampalis C, Tzavelas A, Vraggalas V, Christodoulou P, Bisbinas I. Anterior cruciate ligament remnant-preserving reconstruction using a “lasso-loop” knot configuration. Arthrosc Tech. 2015;4(6):e741-e746.
128. Noh JH, Yoon KH, Song SJ, Roh YH. Re-tensioning technique to cover the graft with remnant in anterior cruciate ligament reconstruction. Arthrosc Tech. 2014;3(6):e679-e682.
129. Hong L, Li X, Zhang H, et al. Anterior cruciate ligament reconstruction with remnant preservation: a prospective, randomized controlled study. Am J Sports Med. 2012;40(12):2747-2755.
130. Noh JH, Kyung HS, Roh YH, Kang TS. Remnant-preserving and re-tensioning technique to cover the graft in anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2015 Nov 12. [Epub ahead of print]
131. Muneta T, Koga H, Ju YJ, Horie M, Nakamura T, Sekiya I. Remnant volume of anterior cruciate ligament correlates preoperative patients’ status and postoperative outcome. Knee Surg Sports Traumatol Arthrosc. 2013;21(4):906-913.
132. Leeberg V, Lekdorf J, Wong C, Sonne-Holm S. Tibial eminentia avulsion fracture in children - a systematic review of the current literature. Dan Med J. 2014;61(3):A4792.
133. In Y, Kwak DS, Moon CW, Han SH, Choi NY. Biomechanical comparison of three techniques for fixation of tibial avulsion fractures of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 2012;20(8):1470-1478.