User login
Long-Term Follow-Up Emphasizes HPV Vaccination Importance
This transcript has been edited for clarity.
I want to briefly discuss a critically important topic that cannot be overly emphasized. It is the relevance, the importance, the benefits, and the outcome of HPV vaccination.
The paper I’m referring to was published in Pediatrics in October 2023. It’s titled, “Ten-Year Follow-up of 9-Valent Human Papillomavirus Vaccine: Immunogenicity, Effectiveness, and Safety.”
Let me emphasize that we’re talking about a 10-year follow-up. In this particular paper and analysis, 301 boys — I emphasize boys — were included and 971 girls at 40 different sites in 13 countries, who received the 9-valent vaccine, which includes HPV 16, 18, and seven other types.
Most importantly, there was not a single case. Not one. Let me repeat this: There was not a single case of high-grade intraepithelial neoplasia, or worse, or condyloma in either males or females. There was not a single case in over 1000 individuals with a follow-up of more than 10 years.
It is difficult to overstate the magnitude of the benefit associated with HPV vaccination for our children and young adults on their risk of developing highly relevant, life-changing, potentially deadly cancers.
For those of you who are interested in this topic — which should include almost all of you, if not all of you — I encourage you to read this very important follow-up paper, again, demonstrating the simple, overwhelming magnitude of the benefit of HPV vaccination. I thank you for your attention.
Dr. Markman is a professor in the department of medical oncology and therapeutics research, City of Hope, Duarte, California, and president of medicine and science, City of Hope Atlanta, Chicago, and Phoenix. He disclosed ties with GlaxoSmithKline; AstraZeneca.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to briefly discuss a critically important topic that cannot be overly emphasized. It is the relevance, the importance, the benefits, and the outcome of HPV vaccination.
The paper I’m referring to was published in Pediatrics in October 2023. It’s titled, “Ten-Year Follow-up of 9-Valent Human Papillomavirus Vaccine: Immunogenicity, Effectiveness, and Safety.”
Let me emphasize that we’re talking about a 10-year follow-up. In this particular paper and analysis, 301 boys — I emphasize boys — were included and 971 girls at 40 different sites in 13 countries, who received the 9-valent vaccine, which includes HPV 16, 18, and seven other types.
Most importantly, there was not a single case. Not one. Let me repeat this: There was not a single case of high-grade intraepithelial neoplasia, or worse, or condyloma in either males or females. There was not a single case in over 1000 individuals with a follow-up of more than 10 years.
It is difficult to overstate the magnitude of the benefit associated with HPV vaccination for our children and young adults on their risk of developing highly relevant, life-changing, potentially deadly cancers.
For those of you who are interested in this topic — which should include almost all of you, if not all of you — I encourage you to read this very important follow-up paper, again, demonstrating the simple, overwhelming magnitude of the benefit of HPV vaccination. I thank you for your attention.
Dr. Markman is a professor in the department of medical oncology and therapeutics research, City of Hope, Duarte, California, and president of medicine and science, City of Hope Atlanta, Chicago, and Phoenix. He disclosed ties with GlaxoSmithKline; AstraZeneca.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to briefly discuss a critically important topic that cannot be overly emphasized. It is the relevance, the importance, the benefits, and the outcome of HPV vaccination.
The paper I’m referring to was published in Pediatrics in October 2023. It’s titled, “Ten-Year Follow-up of 9-Valent Human Papillomavirus Vaccine: Immunogenicity, Effectiveness, and Safety.”
Let me emphasize that we’re talking about a 10-year follow-up. In this particular paper and analysis, 301 boys — I emphasize boys — were included and 971 girls at 40 different sites in 13 countries, who received the 9-valent vaccine, which includes HPV 16, 18, and seven other types.
Most importantly, there was not a single case. Not one. Let me repeat this: There was not a single case of high-grade intraepithelial neoplasia, or worse, or condyloma in either males or females. There was not a single case in over 1000 individuals with a follow-up of more than 10 years.
It is difficult to overstate the magnitude of the benefit associated with HPV vaccination for our children and young adults on their risk of developing highly relevant, life-changing, potentially deadly cancers.
For those of you who are interested in this topic — which should include almost all of you, if not all of you — I encourage you to read this very important follow-up paper, again, demonstrating the simple, overwhelming magnitude of the benefit of HPV vaccination. I thank you for your attention.
Dr. Markman is a professor in the department of medical oncology and therapeutics research, City of Hope, Duarte, California, and president of medicine and science, City of Hope Atlanta, Chicago, and Phoenix. He disclosed ties with GlaxoSmithKline; AstraZeneca.
A version of this article appeared on Medscape.com.
Healing From Trauma
“You’ll never walk alone.” — Nettie Fowler, Carousel
A few winters ago, a young man and his fiancée were driving on the 91 freeway in southern California during a torrential downpour when their Honda Civic hydroplaned, slamming into the jersey barrier. They were both unhurt. Unsure what to do next, they made the catastrophic decision to exit the vehicle. As the man walked around the back of the car he was nearly hit by a black sedan sliding out of control trying to avoid them. When he came around the car, his fiancé was nowhere to be found. She had been struck at highway speed and lay crushed under the sedan hundreds of feet away.
I know this poor man because he was referred to me. Not as a dermatologist, but as a fellow human healing from trauma. On January 1, 2019, at about 9:30 PM, while we were home together, my beloved wife of 24 years took her own life. Even 5 years on it is difficult to believe that she isn’t proofing this paragraph like she had done for every one of my Derm News columns for years. We had been together since teenagers and had lived a joy-filled life. There isn’t any medical reason to share. But that day I joined the community of those who have carried unbearable heaviness of grief and survived. Sometimes others seek me out for help.
At first, my instinct was to guide them, to give advice, to tell them what to do and where to go. But I’ve learned that people in this dark valley don’t need a guide. They need someone to accompany them. To walk with them for a few minutes on their lonely journey. I recently read David Brooks’s new book, How to Know a Person. I’ve been a fan of his since he joined the New York Times in 2003 and have read almost everything he’s written. I sometimes even imagine how he might approach a column whenever I’m stuck (thank you, David). His The Road to Character book is in my canon of literature for self-growth. This latest book is an interesting digression from that central theme. He argues that our society is in acute need of forming better connections and that an important way we can be moral is to learn, and to practice, how to know each other. He shares an emotional experience of losing a close friend to suicide and writes a poignant explanation of what it means to accompany someone in need. It particularly resonated with me. We are doctors and are wired to find the source of a problem, like quickly rotating through the 4X, 10X, 40X on a microscope. Once identified, we spend most of our time creating and explaining treatments. I see how this makes me a great dermatologist but just an average human.
Brooks tells the story of a woman with a brain tumor who often finds herself on the ground surrounded by well-meaning people trying to help. She explains later that what she really needs in those moments is just for someone to get on the ground and lie with her. To accompany her.
Having crossed the midpoint of life, I see with the benefit of perspective how suffering has afforded me wisdom: I am more sensitive and attuned to others. It also gave me credibility: I know how it feels to walk life’s loneliest journey. I’ve also learned to make myself vulnerable for someone to share their story with me. I won’t be afraid to hear the details. I won’t judge them for weeping too little or for sobbing too much. I don’t answer whys. I won’t say what they should do next. But for a few minutes I can walk beside them as a person who cares.
I do not try to remember the hours and days after Susan’s death, but one moment stands out and makes my eyes well when I think of it. That following day my dear brother flew across the country on the next flight out. I was sitting in a psychiatry waiting room when he came down the hall with his luggage in tow. He hugged me as only a brother could, then looked me in my eyes, which were bloodshot from tears just as his were, and he said, “We’re going to be OK.” And with that he walked with me into the office.
We physicians are blessed to have so many intimate human interactions. This book reminded me that sometimes my most important job is not to be the optimized doctor, but just a good human walking alongside.
I have no conflict of interest and purchased these books.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X. Write to him at [email protected].
“You’ll never walk alone.” — Nettie Fowler, Carousel
A few winters ago, a young man and his fiancée were driving on the 91 freeway in southern California during a torrential downpour when their Honda Civic hydroplaned, slamming into the jersey barrier. They were both unhurt. Unsure what to do next, they made the catastrophic decision to exit the vehicle. As the man walked around the back of the car he was nearly hit by a black sedan sliding out of control trying to avoid them. When he came around the car, his fiancé was nowhere to be found. She had been struck at highway speed and lay crushed under the sedan hundreds of feet away.
I know this poor man because he was referred to me. Not as a dermatologist, but as a fellow human healing from trauma. On January 1, 2019, at about 9:30 PM, while we were home together, my beloved wife of 24 years took her own life. Even 5 years on it is difficult to believe that she isn’t proofing this paragraph like she had done for every one of my Derm News columns for years. We had been together since teenagers and had lived a joy-filled life. There isn’t any medical reason to share. But that day I joined the community of those who have carried unbearable heaviness of grief and survived. Sometimes others seek me out for help.
At first, my instinct was to guide them, to give advice, to tell them what to do and where to go. But I’ve learned that people in this dark valley don’t need a guide. They need someone to accompany them. To walk with them for a few minutes on their lonely journey. I recently read David Brooks’s new book, How to Know a Person. I’ve been a fan of his since he joined the New York Times in 2003 and have read almost everything he’s written. I sometimes even imagine how he might approach a column whenever I’m stuck (thank you, David). His The Road to Character book is in my canon of literature for self-growth. This latest book is an interesting digression from that central theme. He argues that our society is in acute need of forming better connections and that an important way we can be moral is to learn, and to practice, how to know each other. He shares an emotional experience of losing a close friend to suicide and writes a poignant explanation of what it means to accompany someone in need. It particularly resonated with me. We are doctors and are wired to find the source of a problem, like quickly rotating through the 4X, 10X, 40X on a microscope. Once identified, we spend most of our time creating and explaining treatments. I see how this makes me a great dermatologist but just an average human.
Brooks tells the story of a woman with a brain tumor who often finds herself on the ground surrounded by well-meaning people trying to help. She explains later that what she really needs in those moments is just for someone to get on the ground and lie with her. To accompany her.
Having crossed the midpoint of life, I see with the benefit of perspective how suffering has afforded me wisdom: I am more sensitive and attuned to others. It also gave me credibility: I know how it feels to walk life’s loneliest journey. I’ve also learned to make myself vulnerable for someone to share their story with me. I won’t be afraid to hear the details. I won’t judge them for weeping too little or for sobbing too much. I don’t answer whys. I won’t say what they should do next. But for a few minutes I can walk beside them as a person who cares.
I do not try to remember the hours and days after Susan’s death, but one moment stands out and makes my eyes well when I think of it. That following day my dear brother flew across the country on the next flight out. I was sitting in a psychiatry waiting room when he came down the hall with his luggage in tow. He hugged me as only a brother could, then looked me in my eyes, which were bloodshot from tears just as his were, and he said, “We’re going to be OK.” And with that he walked with me into the office.
We physicians are blessed to have so many intimate human interactions. This book reminded me that sometimes my most important job is not to be the optimized doctor, but just a good human walking alongside.
I have no conflict of interest and purchased these books.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X. Write to him at [email protected].
“You’ll never walk alone.” — Nettie Fowler, Carousel
A few winters ago, a young man and his fiancée were driving on the 91 freeway in southern California during a torrential downpour when their Honda Civic hydroplaned, slamming into the jersey barrier. They were both unhurt. Unsure what to do next, they made the catastrophic decision to exit the vehicle. As the man walked around the back of the car he was nearly hit by a black sedan sliding out of control trying to avoid them. When he came around the car, his fiancé was nowhere to be found. She had been struck at highway speed and lay crushed under the sedan hundreds of feet away.
I know this poor man because he was referred to me. Not as a dermatologist, but as a fellow human healing from trauma. On January 1, 2019, at about 9:30 PM, while we were home together, my beloved wife of 24 years took her own life. Even 5 years on it is difficult to believe that she isn’t proofing this paragraph like she had done for every one of my Derm News columns for years. We had been together since teenagers and had lived a joy-filled life. There isn’t any medical reason to share. But that day I joined the community of those who have carried unbearable heaviness of grief and survived. Sometimes others seek me out for help.
At first, my instinct was to guide them, to give advice, to tell them what to do and where to go. But I’ve learned that people in this dark valley don’t need a guide. They need someone to accompany them. To walk with them for a few minutes on their lonely journey. I recently read David Brooks’s new book, How to Know a Person. I’ve been a fan of his since he joined the New York Times in 2003 and have read almost everything he’s written. I sometimes even imagine how he might approach a column whenever I’m stuck (thank you, David). His The Road to Character book is in my canon of literature for self-growth. This latest book is an interesting digression from that central theme. He argues that our society is in acute need of forming better connections and that an important way we can be moral is to learn, and to practice, how to know each other. He shares an emotional experience of losing a close friend to suicide and writes a poignant explanation of what it means to accompany someone in need. It particularly resonated with me. We are doctors and are wired to find the source of a problem, like quickly rotating through the 4X, 10X, 40X on a microscope. Once identified, we spend most of our time creating and explaining treatments. I see how this makes me a great dermatologist but just an average human.
Brooks tells the story of a woman with a brain tumor who often finds herself on the ground surrounded by well-meaning people trying to help. She explains later that what she really needs in those moments is just for someone to get on the ground and lie with her. To accompany her.
Having crossed the midpoint of life, I see with the benefit of perspective how suffering has afforded me wisdom: I am more sensitive and attuned to others. It also gave me credibility: I know how it feels to walk life’s loneliest journey. I’ve also learned to make myself vulnerable for someone to share their story with me. I won’t be afraid to hear the details. I won’t judge them for weeping too little or for sobbing too much. I don’t answer whys. I won’t say what they should do next. But for a few minutes I can walk beside them as a person who cares.
I do not try to remember the hours and days after Susan’s death, but one moment stands out and makes my eyes well when I think of it. That following day my dear brother flew across the country on the next flight out. I was sitting in a psychiatry waiting room when he came down the hall with his luggage in tow. He hugged me as only a brother could, then looked me in my eyes, which were bloodshot from tears just as his were, and he said, “We’re going to be OK.” And with that he walked with me into the office.
We physicians are blessed to have so many intimate human interactions. This book reminded me that sometimes my most important job is not to be the optimized doctor, but just a good human walking alongside.
I have no conflict of interest and purchased these books.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on X. Write to him at [email protected].
Bivalent Vaccines Protect Even Children Who’ve Had COVID
This transcript has been edited for clarity.
It was only 3 years ago when we called the pathogen we now refer to as the coronavirus “nCOV-19.” It was, in many ways, more descriptive than what we have today. The little “n” there stood for “novel” — and it was really that little “n” that caused us all the trouble.
You see, coronaviruses themselves were not really new to us. Understudied, perhaps, but with four strains running around the globe at any time giving rise to the common cold, these were viruses our bodies understood.
But Instead of acting like a cold, it acted like nothing we had seen before, at least in our lifetime. The story of the pandemic is very much a bildungsroman of our immune systems — a story of how our immunity grew up.
The difference between the start of 2020 and now, when infections with the coronavirus remain common but not as deadly, can be measured in terms of immune education. Some of our immune systems were educated by infection, some by vaccination, and many by both.
When the first vaccines emerged in December 2020, the opportunity to educate our immune systems was still huge. Though, at the time, an estimated 20 million had been infected in the US and 350,000 had died, there was a large population that remained immunologically naive. I was one of them.
If 2020 into early 2021 was the era of immune education, the postvaccine period was the era of the variant. From one COVID strain to two, to five, to innumerable, our immune memory — trained on a specific version of the virus or its spike protein — became imperfect again. Not naive; these variants were not “novel” in the way COVID-19 was novel, but they were different. And different enough to cause infection.
Following the playbook of another virus that loves to come dressed up in different outfits, the flu virus, we find ourselves in the booster era — a world where yearly doses of a vaccine, ideally matched to the variants circulating when the vaccine is given, are the recommendation if not the norm.
But questions remain about the vaccination program, particularly around who should get it. And two populations with big question marks over their heads are (1) people who have already been infected and (2) kids, because their risk for bad outcomes is so much lower.
This week, we finally have some evidence that can shed light on these questions. The study under the spotlight is this one, appearing in JAMA, which tries to analyze the ability of the bivalent vaccine — that’s the second one to come out, around September 2022 — to protect kids from COVID-19.
Now, right off the bat, this was not a randomized trial. The studies that established the viability of the mRNA vaccine platform were; they happened before the vaccine was authorized. But trials of the bivalent vaccine were mostly limited to proving immune response, not protection from disease.
Nevertheless, with some good observational methods and some statistics, we can try to tease out whether bivalent vaccines in kids worked.
The study combines three prospective cohort studies. The details are in the paper, but what you need to know is that the special sauce of these studies was that the kids were tested for COVID-19 on a weekly basis, whether they had symptoms or not. This is critical because asymptomatic infections can transmit COVID-19.
Let’s do the variables of interest. First and foremost, the bivalent vaccine. Some of these kids got the bivalent vaccine, some didn’t. Other key variables include prior vaccination with the monovalent vaccine. Some had been vaccinated with the monovalent vaccine before, some hadn’t. And, of course, prior infection. Some had been infected before (based on either nasal swabs or blood tests).
Let’s focus first on the primary exposure of interest: getting that bivalent vaccine. Again, this was not randomly assigned; kids who got the bivalent vaccine were different from those who did not. In general, they lived in smaller households, they were more likely to be White, less likely to have had a prior COVID infection, and quite a bit more likely to have at least one chronic condition.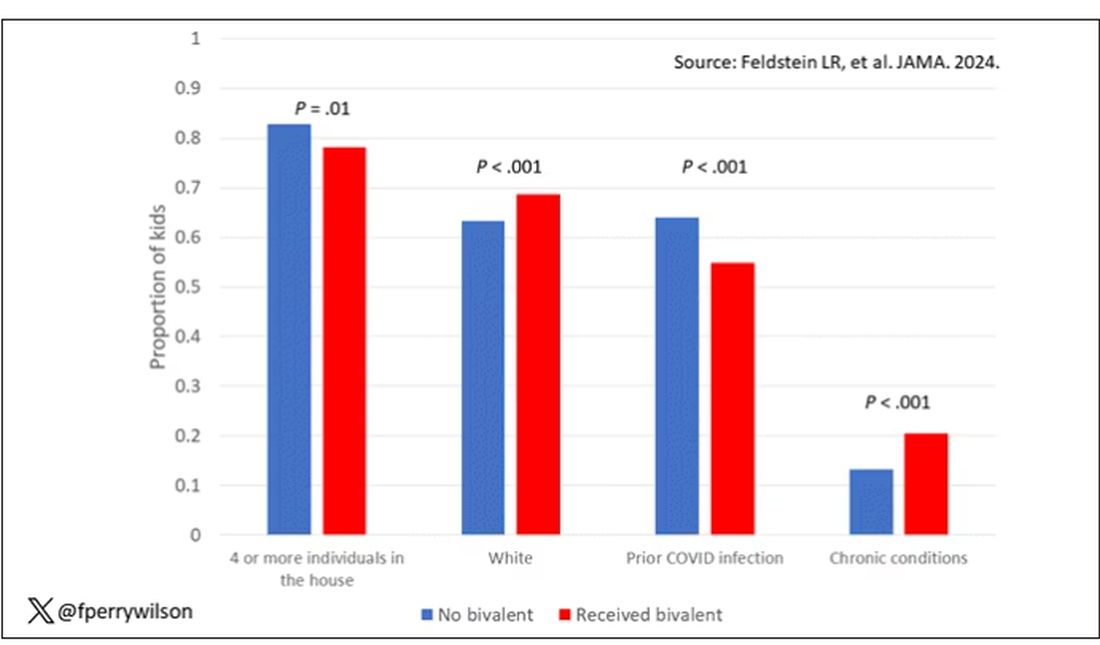
To me, this constellation of factors describes a slightly higher-risk group; it makes sense that they were more likely to get the second vaccine.
Given those factors, what were the rates of COVID infection? After nearly a year of follow-up, around 15% of the kids who hadn’t received the bivalent vaccine got infected compared with 5% of the vaccinated kids. Symptomatic infections represented roughly half of all infections in both groups.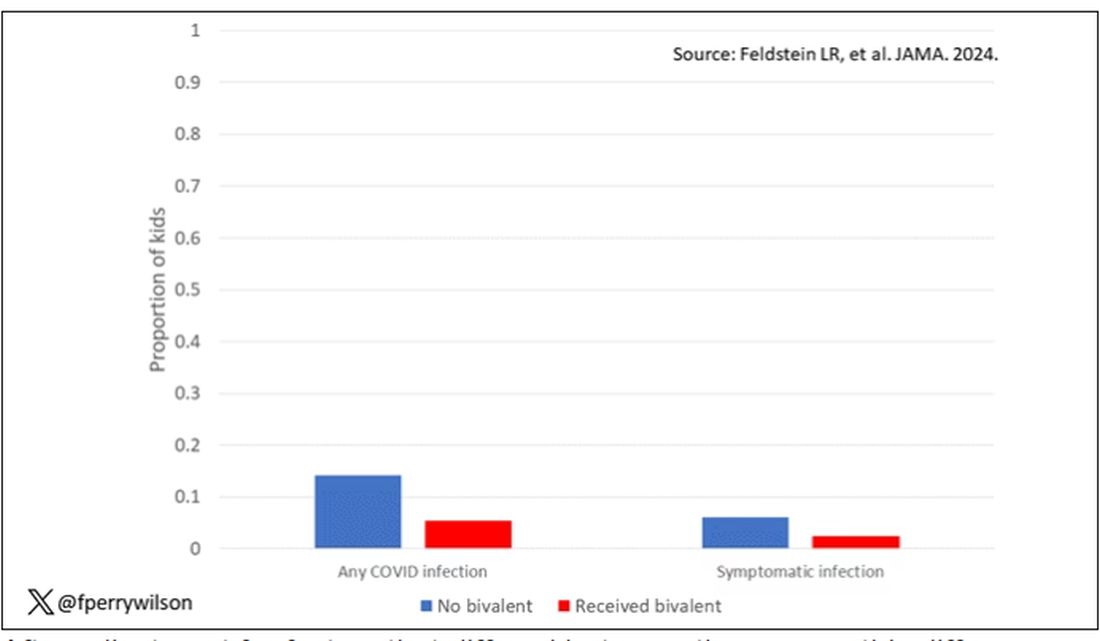
After adjustment for factors that differed between the groups, this difference translated into a vaccine efficacy of about 50% in this population. That’s our first data point. Yes, the bivalent vaccine worked. Not amazingly, of course. But it worked.
What about the kids who had had a prior COVID infection? Somewhat surprisingly, the vaccine was just as effective in this population, despite the fact that their immune systems already had some knowledge of COVID. Ten percent of unvaccinated kids got infected, even though they had been infected before. Just 2.5% of kids who received the bivalent vaccine got infected, suggesting some synergy between prior infection and vaccination.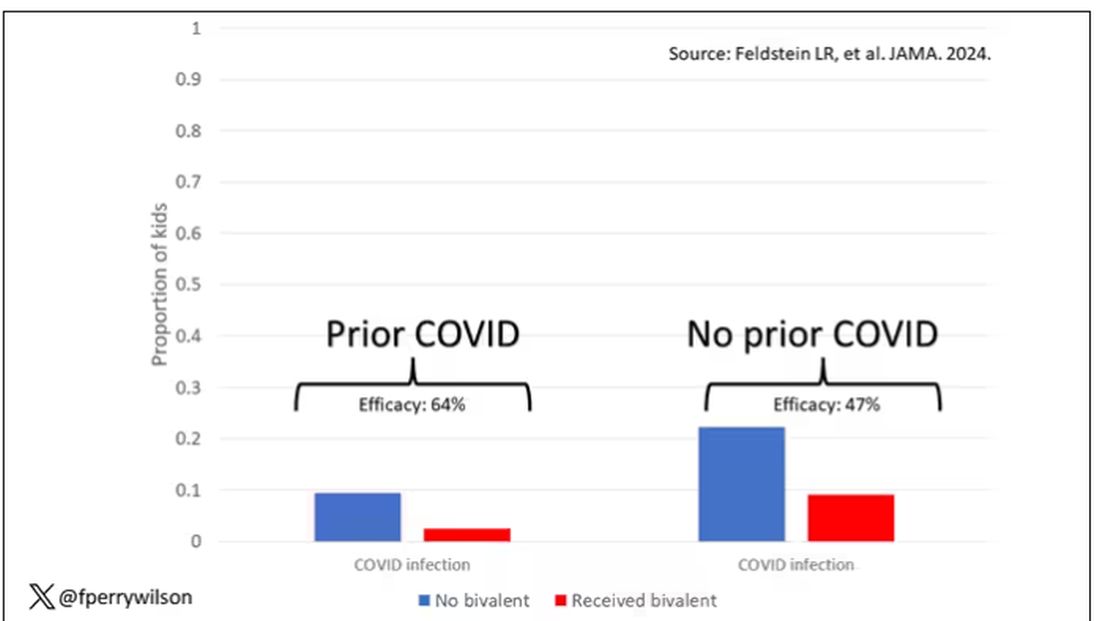
These data suggest that the bivalent vaccine did reduce the risk for COVID infection in kids. All good. But the piece still missing is how severe these infections were. It doesn’t appear that any of the 426 infections documented in this study resulted in hospitalization or death, fortunately. And no data are presented on the incidence of multisystem inflammatory syndrome of children, though given the rarity, I’d be surprised if any of these kids have this either.
So where are we? Well, it seems that the narrative out there that says “the vaccines don’t work” or “the vaccines don’t work if you’ve already been infected” is probably not true. They do work. This study and others in adults show that. If they work to reduce infections, as this study shows, they will also work to reduce deaths. It’s just that death is fortunately so rare in children that the number needed to vaccinate to prevent one death is very large. In that situation, the decision to vaccinate comes down to the risks associated with vaccination. So far, those risk seem very minimal.
Perhaps falling into a flu-like yearly vaccination schedule is not simply the result of old habits dying hard. Maybe it’s actually not a bad idea.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It was only 3 years ago when we called the pathogen we now refer to as the coronavirus “nCOV-19.” It was, in many ways, more descriptive than what we have today. The little “n” there stood for “novel” — and it was really that little “n” that caused us all the trouble.
You see, coronaviruses themselves were not really new to us. Understudied, perhaps, but with four strains running around the globe at any time giving rise to the common cold, these were viruses our bodies understood.
But Instead of acting like a cold, it acted like nothing we had seen before, at least in our lifetime. The story of the pandemic is very much a bildungsroman of our immune systems — a story of how our immunity grew up.
The difference between the start of 2020 and now, when infections with the coronavirus remain common but not as deadly, can be measured in terms of immune education. Some of our immune systems were educated by infection, some by vaccination, and many by both.
When the first vaccines emerged in December 2020, the opportunity to educate our immune systems was still huge. Though, at the time, an estimated 20 million had been infected in the US and 350,000 had died, there was a large population that remained immunologically naive. I was one of them.
If 2020 into early 2021 was the era of immune education, the postvaccine period was the era of the variant. From one COVID strain to two, to five, to innumerable, our immune memory — trained on a specific version of the virus or its spike protein — became imperfect again. Not naive; these variants were not “novel” in the way COVID-19 was novel, but they were different. And different enough to cause infection.
Following the playbook of another virus that loves to come dressed up in different outfits, the flu virus, we find ourselves in the booster era — a world where yearly doses of a vaccine, ideally matched to the variants circulating when the vaccine is given, are the recommendation if not the norm.
But questions remain about the vaccination program, particularly around who should get it. And two populations with big question marks over their heads are (1) people who have already been infected and (2) kids, because their risk for bad outcomes is so much lower.
This week, we finally have some evidence that can shed light on these questions. The study under the spotlight is this one, appearing in JAMA, which tries to analyze the ability of the bivalent vaccine — that’s the second one to come out, around September 2022 — to protect kids from COVID-19.
Now, right off the bat, this was not a randomized trial. The studies that established the viability of the mRNA vaccine platform were; they happened before the vaccine was authorized. But trials of the bivalent vaccine were mostly limited to proving immune response, not protection from disease.
Nevertheless, with some good observational methods and some statistics, we can try to tease out whether bivalent vaccines in kids worked.
The study combines three prospective cohort studies. The details are in the paper, but what you need to know is that the special sauce of these studies was that the kids were tested for COVID-19 on a weekly basis, whether they had symptoms or not. This is critical because asymptomatic infections can transmit COVID-19.
Let’s do the variables of interest. First and foremost, the bivalent vaccine. Some of these kids got the bivalent vaccine, some didn’t. Other key variables include prior vaccination with the monovalent vaccine. Some had been vaccinated with the monovalent vaccine before, some hadn’t. And, of course, prior infection. Some had been infected before (based on either nasal swabs or blood tests).
Let’s focus first on the primary exposure of interest: getting that bivalent vaccine. Again, this was not randomly assigned; kids who got the bivalent vaccine were different from those who did not. In general, they lived in smaller households, they were more likely to be White, less likely to have had a prior COVID infection, and quite a bit more likely to have at least one chronic condition.
To me, this constellation of factors describes a slightly higher-risk group; it makes sense that they were more likely to get the second vaccine.
Given those factors, what were the rates of COVID infection? After nearly a year of follow-up, around 15% of the kids who hadn’t received the bivalent vaccine got infected compared with 5% of the vaccinated kids. Symptomatic infections represented roughly half of all infections in both groups.
After adjustment for factors that differed between the groups, this difference translated into a vaccine efficacy of about 50% in this population. That’s our first data point. Yes, the bivalent vaccine worked. Not amazingly, of course. But it worked.
What about the kids who had had a prior COVID infection? Somewhat surprisingly, the vaccine was just as effective in this population, despite the fact that their immune systems already had some knowledge of COVID. Ten percent of unvaccinated kids got infected, even though they had been infected before. Just 2.5% of kids who received the bivalent vaccine got infected, suggesting some synergy between prior infection and vaccination.
These data suggest that the bivalent vaccine did reduce the risk for COVID infection in kids. All good. But the piece still missing is how severe these infections were. It doesn’t appear that any of the 426 infections documented in this study resulted in hospitalization or death, fortunately. And no data are presented on the incidence of multisystem inflammatory syndrome of children, though given the rarity, I’d be surprised if any of these kids have this either.
So where are we? Well, it seems that the narrative out there that says “the vaccines don’t work” or “the vaccines don’t work if you’ve already been infected” is probably not true. They do work. This study and others in adults show that. If they work to reduce infections, as this study shows, they will also work to reduce deaths. It’s just that death is fortunately so rare in children that the number needed to vaccinate to prevent one death is very large. In that situation, the decision to vaccinate comes down to the risks associated with vaccination. So far, those risk seem very minimal.
Perhaps falling into a flu-like yearly vaccination schedule is not simply the result of old habits dying hard. Maybe it’s actually not a bad idea.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It was only 3 years ago when we called the pathogen we now refer to as the coronavirus “nCOV-19.” It was, in many ways, more descriptive than what we have today. The little “n” there stood for “novel” — and it was really that little “n” that caused us all the trouble.
You see, coronaviruses themselves were not really new to us. Understudied, perhaps, but with four strains running around the globe at any time giving rise to the common cold, these were viruses our bodies understood.
But Instead of acting like a cold, it acted like nothing we had seen before, at least in our lifetime. The story of the pandemic is very much a bildungsroman of our immune systems — a story of how our immunity grew up.
The difference between the start of 2020 and now, when infections with the coronavirus remain common but not as deadly, can be measured in terms of immune education. Some of our immune systems were educated by infection, some by vaccination, and many by both.
When the first vaccines emerged in December 2020, the opportunity to educate our immune systems was still huge. Though, at the time, an estimated 20 million had been infected in the US and 350,000 had died, there was a large population that remained immunologically naive. I was one of them.
If 2020 into early 2021 was the era of immune education, the postvaccine period was the era of the variant. From one COVID strain to two, to five, to innumerable, our immune memory — trained on a specific version of the virus or its spike protein — became imperfect again. Not naive; these variants were not “novel” in the way COVID-19 was novel, but they were different. And different enough to cause infection.
Following the playbook of another virus that loves to come dressed up in different outfits, the flu virus, we find ourselves in the booster era — a world where yearly doses of a vaccine, ideally matched to the variants circulating when the vaccine is given, are the recommendation if not the norm.
But questions remain about the vaccination program, particularly around who should get it. And two populations with big question marks over their heads are (1) people who have already been infected and (2) kids, because their risk for bad outcomes is so much lower.
This week, we finally have some evidence that can shed light on these questions. The study under the spotlight is this one, appearing in JAMA, which tries to analyze the ability of the bivalent vaccine — that’s the second one to come out, around September 2022 — to protect kids from COVID-19.
Now, right off the bat, this was not a randomized trial. The studies that established the viability of the mRNA vaccine platform were; they happened before the vaccine was authorized. But trials of the bivalent vaccine were mostly limited to proving immune response, not protection from disease.
Nevertheless, with some good observational methods and some statistics, we can try to tease out whether bivalent vaccines in kids worked.
The study combines three prospective cohort studies. The details are in the paper, but what you need to know is that the special sauce of these studies was that the kids were tested for COVID-19 on a weekly basis, whether they had symptoms or not. This is critical because asymptomatic infections can transmit COVID-19.
Let’s do the variables of interest. First and foremost, the bivalent vaccine. Some of these kids got the bivalent vaccine, some didn’t. Other key variables include prior vaccination with the monovalent vaccine. Some had been vaccinated with the monovalent vaccine before, some hadn’t. And, of course, prior infection. Some had been infected before (based on either nasal swabs or blood tests).
Let’s focus first on the primary exposure of interest: getting that bivalent vaccine. Again, this was not randomly assigned; kids who got the bivalent vaccine were different from those who did not. In general, they lived in smaller households, they were more likely to be White, less likely to have had a prior COVID infection, and quite a bit more likely to have at least one chronic condition.
To me, this constellation of factors describes a slightly higher-risk group; it makes sense that they were more likely to get the second vaccine.
Given those factors, what were the rates of COVID infection? After nearly a year of follow-up, around 15% of the kids who hadn’t received the bivalent vaccine got infected compared with 5% of the vaccinated kids. Symptomatic infections represented roughly half of all infections in both groups.
After adjustment for factors that differed between the groups, this difference translated into a vaccine efficacy of about 50% in this population. That’s our first data point. Yes, the bivalent vaccine worked. Not amazingly, of course. But it worked.
What about the kids who had had a prior COVID infection? Somewhat surprisingly, the vaccine was just as effective in this population, despite the fact that their immune systems already had some knowledge of COVID. Ten percent of unvaccinated kids got infected, even though they had been infected before. Just 2.5% of kids who received the bivalent vaccine got infected, suggesting some synergy between prior infection and vaccination.
These data suggest that the bivalent vaccine did reduce the risk for COVID infection in kids. All good. But the piece still missing is how severe these infections were. It doesn’t appear that any of the 426 infections documented in this study resulted in hospitalization or death, fortunately. And no data are presented on the incidence of multisystem inflammatory syndrome of children, though given the rarity, I’d be surprised if any of these kids have this either.
So where are we? Well, it seems that the narrative out there that says “the vaccines don’t work” or “the vaccines don’t work if you’ve already been infected” is probably not true. They do work. This study and others in adults show that. If they work to reduce infections, as this study shows, they will also work to reduce deaths. It’s just that death is fortunately so rare in children that the number needed to vaccinate to prevent one death is very large. In that situation, the decision to vaccinate comes down to the risks associated with vaccination. So far, those risk seem very minimal.
Perhaps falling into a flu-like yearly vaccination schedule is not simply the result of old habits dying hard. Maybe it’s actually not a bad idea.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Hospital Marriage Proposals: The Good, the Bad, the Helipad
Picture your marriage proposal fantasy. Do you see a beautiful beach at sunset? The place where you first met your partner? Maybe a dream vacation — Paris, anyone? And perhaps most popular of all ... the ER?
Why not? For some couples who share medical careers, the hospital is home, and they turn the moment into something just as romantic as any Eiffel Tower backdrop. (And admittedly, sometimes they don’t.)
We spoke with three couples whose medical-themed proposals ended in the word “yes!”
Heaven on the Helipad
When emergency medicine physician Anna Darby, MD, heard a trauma patient was arriving and urgently needed to be intubated, she raced up to the rooftop helipad. As soon as the elevator doors opened, she was met with quite a different scene than expected. There were rose petals ... lots and lots of rose petals.
With her best friends and colleagues lining a red carpet, the roof had been turned into a scene from The Bachelor. Each person gave her a rose. A friend even touched up her makeup and handed over her favorite hoop earrings, transforming her from busy doctor to soon-to-be fiancée. Her boyfriend, cardiologist Merije Chukumerije, MD, stood waiting. You can guess what happened next.
Dr. Chukumerije later wrote in an Instagram post, “We met at this hospital. So, it was only right that I bring her to its highest place as we’ve reached the peak of our union.” The couple actually met in the hospital cafeteria “like all the clichés,” Dr. Darby jokes. For them, the helipad experience was just as Insta-worthy as any braggable, grandiose proposal at a fancy restaurant or on a mountaintop.
“Seeing that scene was totally not what I expected,” Dr. Darby says. “I can’t even describe it. It’s like the second biggest hormonal shift, [second only to] having a baby.” She and Dr. Chukumerije now have two babies of their own, aged 2 months and 2 years old.
Good Morning, Doctor
It was February 2021, the height of the pandemic, and Raaga Vemula, MD, now in her palliative care/hospice care fellowship, was “selected” for a local news interview on COVID-19. Except the interview was really with Good Morning America. And the topic was really a proposal.
Dr. Vemula met Steven Bean, MD, now doing a sleep medicine fellowship, in 2015. “I first saw her, and thought she was one of the prettiest women I’d ever seen. ... We ended up being in the same study group,” he says. “Let’s be honest, I applied to every med school she applied to.”
Six years later, Dr. Bean connected with GMA through The Knot, a wedding planning website and registry. The made-up interview request for Dr. Vemula came from the residency program director, who was in on the surprise. Dr. Vemula’s family also knew what was up when she called with the “news.”
The live broadcast took place at the hospital. Dr. Bean had an earpiece for the producers to give him directions. But “I was so nervous, I walked out immediately,” he says. He ended up standing behind Dr. Vemula. The mistake worked well for viewers though, building anticipation while she answered a COVID-19 question. “We got everybody excited,” says Dr. Bean. “So, when they said ’Raaga, turn around’ it worked out perfectly. She was confused as hell.”
Luckily, Dr. Vemula loves a good surprise. “He knows me very well,” she says.
For her, the proposal was even more meaningful given their background together. “Medicine means so much to both of us and was such a big part of our lives,” she says. “That’s what shaped us to do this. ... I think in our hearts it was meant to be this way.”
Who Says Masks Don’t Work?
Masks conjure up feelings for anyone living through the pandemic, especially medical personnel. But for Rhett Franklin and wife Lauren Gray, they will always symbolize of one of the biggest days of their lives.
Mr. Franklin worked in registration, often following Ms. Gray, an emergency room nurse, around with a wheeled computer station to gather patient information (what’s known as a “creeper,” which isn’t as creepy as it sounds). Eventually, she offered to grab a coffee with him, and when he suggested another coffee, she said it was time for him to buy her a drink.
Mr. Franklin, now a manager of business operations for nursing administration, originally planned to propose to Ms. Gray on a trip to England. But the pandemic prevented their vacation with its potential castle backdrop.
Mr. Franklin often picked up shifts making masks for frontline workers, and an alternate proposal idea started brewing. He schemed to have two very special masks made. “Mine was a black tuxedo that said, ‘Will you marry me?’ and hers resembled a white dress that said, ‘I said yes!’ ” Mr. Franklin says.
But a text almost ruined the surprise. When Mr. Franklin messaged family members about his proposal plan the day before, one relative responded in a group chat that included Ms. Gray. This was when the busy ER came to the rescue — no time to read texts. Family members also started calling Ms. Gray on the hospital’s phoneline as a distraction. Unfazed, Mr. Franklin simply moved up the proposal to that night.
At their favorite dog beach, as the sunset gleamed on the water, Mr. Franklin pulled his mask out and took a knee. He can’t recall what he said behind that mask. “It was kind of one of those blackout moments.” But Ms. Gray remembers for him — “You said ‘Let’s do this.’ ”
Warning Label
Everyone has different tastes. Some healthcare professionals have taken the medical theme further than these couples — maybe too far. A few have even faked life-threatening emergencies, showing up in the ER on a gurney with a made-up peanut allergy reaction or a severe injury and then pulling out a ring.
But who’s to judge? For some, thinking your partner is “dying” and then learning you’ve been tricked might not conjure up the warmest feelings. For others, apparently, it’s a virtual bouquet of roses.
A Few Proposal Pointers
If you’re planning to pop the question, this group says, “go for the medical setting!” But according to them, there are other must-haves to get that “yes” and the lifetime of wedded bliss, of course:
- Make it a hospital-wide morale-booster. “Everyone loves surprises,” Dr. Bean maintains. So, why not bring your colleagues in on the conspiracy? “Involving coworkers will strengthen relationships with their work family by leaving lasting memories for everyone,” he says. “In a busy medical setting, it’s usually unexpected, so it makes it extra special.”
- Have a backup plan. As healthcare professionals, you know that schedules get in the way of everything. So, practice that flexibility you will need as a marriage skill. When Mr. Franklin’s first two engagement locations fell though, he says, it was important to adapt and not panic when things went awry.
- Seize the moment. Think you can’t get engaged during residency? “Planning a proposal during intern year of residency is totally manageable,” Dr. Vemula says. “That way as residency progresses and you have more time, there is more time to focus on the wedding planning.” But she cautions that, “wedding planning during the intern year would be quite difficult.”
A version of this article appeared on Medscape.com.
Picture your marriage proposal fantasy. Do you see a beautiful beach at sunset? The place where you first met your partner? Maybe a dream vacation — Paris, anyone? And perhaps most popular of all ... the ER?
Why not? For some couples who share medical careers, the hospital is home, and they turn the moment into something just as romantic as any Eiffel Tower backdrop. (And admittedly, sometimes they don’t.)
We spoke with three couples whose medical-themed proposals ended in the word “yes!”
Heaven on the Helipad
When emergency medicine physician Anna Darby, MD, heard a trauma patient was arriving and urgently needed to be intubated, she raced up to the rooftop helipad. As soon as the elevator doors opened, she was met with quite a different scene than expected. There were rose petals ... lots and lots of rose petals.
With her best friends and colleagues lining a red carpet, the roof had been turned into a scene from The Bachelor. Each person gave her a rose. A friend even touched up her makeup and handed over her favorite hoop earrings, transforming her from busy doctor to soon-to-be fiancée. Her boyfriend, cardiologist Merije Chukumerije, MD, stood waiting. You can guess what happened next.
Dr. Chukumerije later wrote in an Instagram post, “We met at this hospital. So, it was only right that I bring her to its highest place as we’ve reached the peak of our union.” The couple actually met in the hospital cafeteria “like all the clichés,” Dr. Darby jokes. For them, the helipad experience was just as Insta-worthy as any braggable, grandiose proposal at a fancy restaurant or on a mountaintop.
“Seeing that scene was totally not what I expected,” Dr. Darby says. “I can’t even describe it. It’s like the second biggest hormonal shift, [second only to] having a baby.” She and Dr. Chukumerije now have two babies of their own, aged 2 months and 2 years old.
Good Morning, Doctor
It was February 2021, the height of the pandemic, and Raaga Vemula, MD, now in her palliative care/hospice care fellowship, was “selected” for a local news interview on COVID-19. Except the interview was really with Good Morning America. And the topic was really a proposal.
Dr. Vemula met Steven Bean, MD, now doing a sleep medicine fellowship, in 2015. “I first saw her, and thought she was one of the prettiest women I’d ever seen. ... We ended up being in the same study group,” he says. “Let’s be honest, I applied to every med school she applied to.”
Six years later, Dr. Bean connected with GMA through The Knot, a wedding planning website and registry. The made-up interview request for Dr. Vemula came from the residency program director, who was in on the surprise. Dr. Vemula’s family also knew what was up when she called with the “news.”
The live broadcast took place at the hospital. Dr. Bean had an earpiece for the producers to give him directions. But “I was so nervous, I walked out immediately,” he says. He ended up standing behind Dr. Vemula. The mistake worked well for viewers though, building anticipation while she answered a COVID-19 question. “We got everybody excited,” says Dr. Bean. “So, when they said ’Raaga, turn around’ it worked out perfectly. She was confused as hell.”
Luckily, Dr. Vemula loves a good surprise. “He knows me very well,” she says.
For her, the proposal was even more meaningful given their background together. “Medicine means so much to both of us and was such a big part of our lives,” she says. “That’s what shaped us to do this. ... I think in our hearts it was meant to be this way.”
Who Says Masks Don’t Work?
Masks conjure up feelings for anyone living through the pandemic, especially medical personnel. But for Rhett Franklin and wife Lauren Gray, they will always symbolize of one of the biggest days of their lives.
Mr. Franklin worked in registration, often following Ms. Gray, an emergency room nurse, around with a wheeled computer station to gather patient information (what’s known as a “creeper,” which isn’t as creepy as it sounds). Eventually, she offered to grab a coffee with him, and when he suggested another coffee, she said it was time for him to buy her a drink.
Mr. Franklin, now a manager of business operations for nursing administration, originally planned to propose to Ms. Gray on a trip to England. But the pandemic prevented their vacation with its potential castle backdrop.
Mr. Franklin often picked up shifts making masks for frontline workers, and an alternate proposal idea started brewing. He schemed to have two very special masks made. “Mine was a black tuxedo that said, ‘Will you marry me?’ and hers resembled a white dress that said, ‘I said yes!’ ” Mr. Franklin says.
But a text almost ruined the surprise. When Mr. Franklin messaged family members about his proposal plan the day before, one relative responded in a group chat that included Ms. Gray. This was when the busy ER came to the rescue — no time to read texts. Family members also started calling Ms. Gray on the hospital’s phoneline as a distraction. Unfazed, Mr. Franklin simply moved up the proposal to that night.
At their favorite dog beach, as the sunset gleamed on the water, Mr. Franklin pulled his mask out and took a knee. He can’t recall what he said behind that mask. “It was kind of one of those blackout moments.” But Ms. Gray remembers for him — “You said ‘Let’s do this.’ ”
Warning Label
Everyone has different tastes. Some healthcare professionals have taken the medical theme further than these couples — maybe too far. A few have even faked life-threatening emergencies, showing up in the ER on a gurney with a made-up peanut allergy reaction or a severe injury and then pulling out a ring.
But who’s to judge? For some, thinking your partner is “dying” and then learning you’ve been tricked might not conjure up the warmest feelings. For others, apparently, it’s a virtual bouquet of roses.
A Few Proposal Pointers
If you’re planning to pop the question, this group says, “go for the medical setting!” But according to them, there are other must-haves to get that “yes” and the lifetime of wedded bliss, of course:
- Make it a hospital-wide morale-booster. “Everyone loves surprises,” Dr. Bean maintains. So, why not bring your colleagues in on the conspiracy? “Involving coworkers will strengthen relationships with their work family by leaving lasting memories for everyone,” he says. “In a busy medical setting, it’s usually unexpected, so it makes it extra special.”
- Have a backup plan. As healthcare professionals, you know that schedules get in the way of everything. So, practice that flexibility you will need as a marriage skill. When Mr. Franklin’s first two engagement locations fell though, he says, it was important to adapt and not panic when things went awry.
- Seize the moment. Think you can’t get engaged during residency? “Planning a proposal during intern year of residency is totally manageable,” Dr. Vemula says. “That way as residency progresses and you have more time, there is more time to focus on the wedding planning.” But she cautions that, “wedding planning during the intern year would be quite difficult.”
A version of this article appeared on Medscape.com.
Picture your marriage proposal fantasy. Do you see a beautiful beach at sunset? The place where you first met your partner? Maybe a dream vacation — Paris, anyone? And perhaps most popular of all ... the ER?
Why not? For some couples who share medical careers, the hospital is home, and they turn the moment into something just as romantic as any Eiffel Tower backdrop. (And admittedly, sometimes they don’t.)
We spoke with three couples whose medical-themed proposals ended in the word “yes!”
Heaven on the Helipad
When emergency medicine physician Anna Darby, MD, heard a trauma patient was arriving and urgently needed to be intubated, she raced up to the rooftop helipad. As soon as the elevator doors opened, she was met with quite a different scene than expected. There were rose petals ... lots and lots of rose petals.
With her best friends and colleagues lining a red carpet, the roof had been turned into a scene from The Bachelor. Each person gave her a rose. A friend even touched up her makeup and handed over her favorite hoop earrings, transforming her from busy doctor to soon-to-be fiancée. Her boyfriend, cardiologist Merije Chukumerije, MD, stood waiting. You can guess what happened next.
Dr. Chukumerije later wrote in an Instagram post, “We met at this hospital. So, it was only right that I bring her to its highest place as we’ve reached the peak of our union.” The couple actually met in the hospital cafeteria “like all the clichés,” Dr. Darby jokes. For them, the helipad experience was just as Insta-worthy as any braggable, grandiose proposal at a fancy restaurant or on a mountaintop.
“Seeing that scene was totally not what I expected,” Dr. Darby says. “I can’t even describe it. It’s like the second biggest hormonal shift, [second only to] having a baby.” She and Dr. Chukumerije now have two babies of their own, aged 2 months and 2 years old.
Good Morning, Doctor
It was February 2021, the height of the pandemic, and Raaga Vemula, MD, now in her palliative care/hospice care fellowship, was “selected” for a local news interview on COVID-19. Except the interview was really with Good Morning America. And the topic was really a proposal.
Dr. Vemula met Steven Bean, MD, now doing a sleep medicine fellowship, in 2015. “I first saw her, and thought she was one of the prettiest women I’d ever seen. ... We ended up being in the same study group,” he says. “Let’s be honest, I applied to every med school she applied to.”
Six years later, Dr. Bean connected with GMA through The Knot, a wedding planning website and registry. The made-up interview request for Dr. Vemula came from the residency program director, who was in on the surprise. Dr. Vemula’s family also knew what was up when she called with the “news.”
The live broadcast took place at the hospital. Dr. Bean had an earpiece for the producers to give him directions. But “I was so nervous, I walked out immediately,” he says. He ended up standing behind Dr. Vemula. The mistake worked well for viewers though, building anticipation while she answered a COVID-19 question. “We got everybody excited,” says Dr. Bean. “So, when they said ’Raaga, turn around’ it worked out perfectly. She was confused as hell.”
Luckily, Dr. Vemula loves a good surprise. “He knows me very well,” she says.
For her, the proposal was even more meaningful given their background together. “Medicine means so much to both of us and was such a big part of our lives,” she says. “That’s what shaped us to do this. ... I think in our hearts it was meant to be this way.”
Who Says Masks Don’t Work?
Masks conjure up feelings for anyone living through the pandemic, especially medical personnel. But for Rhett Franklin and wife Lauren Gray, they will always symbolize of one of the biggest days of their lives.
Mr. Franklin worked in registration, often following Ms. Gray, an emergency room nurse, around with a wheeled computer station to gather patient information (what’s known as a “creeper,” which isn’t as creepy as it sounds). Eventually, she offered to grab a coffee with him, and when he suggested another coffee, she said it was time for him to buy her a drink.
Mr. Franklin, now a manager of business operations for nursing administration, originally planned to propose to Ms. Gray on a trip to England. But the pandemic prevented their vacation with its potential castle backdrop.
Mr. Franklin often picked up shifts making masks for frontline workers, and an alternate proposal idea started brewing. He schemed to have two very special masks made. “Mine was a black tuxedo that said, ‘Will you marry me?’ and hers resembled a white dress that said, ‘I said yes!’ ” Mr. Franklin says.
But a text almost ruined the surprise. When Mr. Franklin messaged family members about his proposal plan the day before, one relative responded in a group chat that included Ms. Gray. This was when the busy ER came to the rescue — no time to read texts. Family members also started calling Ms. Gray on the hospital’s phoneline as a distraction. Unfazed, Mr. Franklin simply moved up the proposal to that night.
At their favorite dog beach, as the sunset gleamed on the water, Mr. Franklin pulled his mask out and took a knee. He can’t recall what he said behind that mask. “It was kind of one of those blackout moments.” But Ms. Gray remembers for him — “You said ‘Let’s do this.’ ”
Warning Label
Everyone has different tastes. Some healthcare professionals have taken the medical theme further than these couples — maybe too far. A few have even faked life-threatening emergencies, showing up in the ER on a gurney with a made-up peanut allergy reaction or a severe injury and then pulling out a ring.
But who’s to judge? For some, thinking your partner is “dying” and then learning you’ve been tricked might not conjure up the warmest feelings. For others, apparently, it’s a virtual bouquet of roses.
A Few Proposal Pointers
If you’re planning to pop the question, this group says, “go for the medical setting!” But according to them, there are other must-haves to get that “yes” and the lifetime of wedded bliss, of course:
- Make it a hospital-wide morale-booster. “Everyone loves surprises,” Dr. Bean maintains. So, why not bring your colleagues in on the conspiracy? “Involving coworkers will strengthen relationships with their work family by leaving lasting memories for everyone,” he says. “In a busy medical setting, it’s usually unexpected, so it makes it extra special.”
- Have a backup plan. As healthcare professionals, you know that schedules get in the way of everything. So, practice that flexibility you will need as a marriage skill. When Mr. Franklin’s first two engagement locations fell though, he says, it was important to adapt and not panic when things went awry.
- Seize the moment. Think you can’t get engaged during residency? “Planning a proposal during intern year of residency is totally manageable,” Dr. Vemula says. “That way as residency progresses and you have more time, there is more time to focus on the wedding planning.” But she cautions that, “wedding planning during the intern year would be quite difficult.”
A version of this article appeared on Medscape.com.
An Ethical Analysis of Treatment of an Active-Duty Service Member With Limited Follow-up
For active-duty service members, dermatologic conditions are among the most common presenting concerns, comprising 15% to 75% of wartime outpatient visits.1 In general, there are unique considerations when caring for active-duty service members, including meeting designated active-duty retention and hierarchical standards.2 We present a hypothetical case: An active-duty military patient presents to a new dermatologist for cosmetic enhancement of facial skin dyspigmentation. The patient will be leaving soon for deployment and will not be able to follow up for 9 months. How should the dermatologist treat a patient who cannot follow up for so long?
The therapeutic modalities offered can be impacted by forthcoming deployments3 that may result in delayed time to administer repeat treatments or follow-up. The patient may have high expectations for a single appointment for a condition that requires prolonged treatment courses. Because there often is no reliable mechanism for patients to obtain refills during deployment, any medications prescribed would need to be provided in advance for the entire deployment duration, which often is 6 to 9 months. Additionally, treatment monitoring or modifications are severely limited, especially in the context of treatment nonresponse or adverse reactions. Considering the unique limitations of this patient population, both military and civilian physicians are faced with a need to maximize beneficence and autonomy while balancing nonmaleficence and justice.
One possible option is to decline to treat until the patient can follow up after returning from deployment. However, denying a request for an active treatable indication for which the patient desires treatment compromises patient autonomy and beneficence. Further, treatment should be provided to patients equitably to maintain justice. Although there may be a role for discussing active monitoring with nonintervention with the patient, denying treatment can negatively impact their physical and mental health and may be harmful. However, the patient should know and fully understand the risks and benefits of nonintervention with limited follow-up, including suboptimal outcomes or adverse events.
Another possibility for the management of this case may be conducting a one-time laser or light-based therapy or a one-time superficial- to medium-depth chemical peel before the patient leaves on deployment. Often, a series of laser- or light-based treatments is required to maximize outcomes for dyspigmentation. Without follow-up and with possible deployment to an environment with high UV exposure, the patient may experience disease exacerbation or other adverse effects. Treatment of those adverse effects may be delayed, as further intervention is not possible during deployment. Lower initial laser settings may be safer but may not be highly effective initially. More rigorous treatment upon return from deployment may be considered. Similar to laser therapies, chemical peels usually require several treatments for optimal outcomes. Without follow-up and with potential deployment to remote environments, there is a risk for adverse events that outweighs the minimal benefit of a single treatment. Therefore, either intervention may violate the principle of nonmaleficence.
A more reasonable approach may be initiating topical therapy and following up via telemedicine evaluation. Topical therapy often is the least-invasive approach and carries a reduced risk for adverse effects. Triple-combination therapy with topical retinoids, hydroquinone, and topical steroids is a common first-line approach.4 Because this approach is patient dependent, therapy can be more easily modulated or halted in the context of undesired results. Additionally, if internet connectivity is available, an asynchronous telemedicine approach could be utilized during deployment to monitor and advise changes as necessary, provided the regulatory framework allows for it.5
Although there is no uniformly correct approach in a scenario of limited patient follow-up, the last solution may be most ethically favorable: to begin therapy with milder and safer therapies (topical) and defer higher-intensity regimens until the patient returns from deployment. This allows some treatment initiation to preserve justice, beneficence, and patient autonomy. Associated virtual follow-up via telemedicine also allows avoidance of nonmaleficence in this context.
- Hwang J, Kakimoto C. Teledermatology in the US military: a historic foundation for current and future applications. Cutis. 2018;101:335;337;345.
- Dodd JG, Grant-Kels JM. Ethical concerns in caring for active duty service members who may be seeking dermatologic care outside the military soon. Int J Womens Dermatol. 2020;6:445-447. doi:10.1016/j.ijwd.2020.07.001
- Burke KR, Larrymore DC, Cho S. Treatment consideration for US military members with skin disease. Cutis. 2019;103:329-332.
- Desai SR. Hyperpigmentation therapy: a review. J Clin Aesthet Dermatol. 2014;7:13-17.
- Hwang JS, Lappan CM, Sperling LC, et al. Utilization of telemedicine in the U.S. military in a deployed setting. Mil Med. 2014;179:1347-1353. doi:10.7205/MILMED-D-14-00115
For active-duty service members, dermatologic conditions are among the most common presenting concerns, comprising 15% to 75% of wartime outpatient visits.1 In general, there are unique considerations when caring for active-duty service members, including meeting designated active-duty retention and hierarchical standards.2 We present a hypothetical case: An active-duty military patient presents to a new dermatologist for cosmetic enhancement of facial skin dyspigmentation. The patient will be leaving soon for deployment and will not be able to follow up for 9 months. How should the dermatologist treat a patient who cannot follow up for so long?
The therapeutic modalities offered can be impacted by forthcoming deployments3 that may result in delayed time to administer repeat treatments or follow-up. The patient may have high expectations for a single appointment for a condition that requires prolonged treatment courses. Because there often is no reliable mechanism for patients to obtain refills during deployment, any medications prescribed would need to be provided in advance for the entire deployment duration, which often is 6 to 9 months. Additionally, treatment monitoring or modifications are severely limited, especially in the context of treatment nonresponse or adverse reactions. Considering the unique limitations of this patient population, both military and civilian physicians are faced with a need to maximize beneficence and autonomy while balancing nonmaleficence and justice.
One possible option is to decline to treat until the patient can follow up after returning from deployment. However, denying a request for an active treatable indication for which the patient desires treatment compromises patient autonomy and beneficence. Further, treatment should be provided to patients equitably to maintain justice. Although there may be a role for discussing active monitoring with nonintervention with the patient, denying treatment can negatively impact their physical and mental health and may be harmful. However, the patient should know and fully understand the risks and benefits of nonintervention with limited follow-up, including suboptimal outcomes or adverse events.
Another possibility for the management of this case may be conducting a one-time laser or light-based therapy or a one-time superficial- to medium-depth chemical peel before the patient leaves on deployment. Often, a series of laser- or light-based treatments is required to maximize outcomes for dyspigmentation. Without follow-up and with possible deployment to an environment with high UV exposure, the patient may experience disease exacerbation or other adverse effects. Treatment of those adverse effects may be delayed, as further intervention is not possible during deployment. Lower initial laser settings may be safer but may not be highly effective initially. More rigorous treatment upon return from deployment may be considered. Similar to laser therapies, chemical peels usually require several treatments for optimal outcomes. Without follow-up and with potential deployment to remote environments, there is a risk for adverse events that outweighs the minimal benefit of a single treatment. Therefore, either intervention may violate the principle of nonmaleficence.
A more reasonable approach may be initiating topical therapy and following up via telemedicine evaluation. Topical therapy often is the least-invasive approach and carries a reduced risk for adverse effects. Triple-combination therapy with topical retinoids, hydroquinone, and topical steroids is a common first-line approach.4 Because this approach is patient dependent, therapy can be more easily modulated or halted in the context of undesired results. Additionally, if internet connectivity is available, an asynchronous telemedicine approach could be utilized during deployment to monitor and advise changes as necessary, provided the regulatory framework allows for it.5
Although there is no uniformly correct approach in a scenario of limited patient follow-up, the last solution may be most ethically favorable: to begin therapy with milder and safer therapies (topical) and defer higher-intensity regimens until the patient returns from deployment. This allows some treatment initiation to preserve justice, beneficence, and patient autonomy. Associated virtual follow-up via telemedicine also allows avoidance of nonmaleficence in this context.
For active-duty service members, dermatologic conditions are among the most common presenting concerns, comprising 15% to 75% of wartime outpatient visits.1 In general, there are unique considerations when caring for active-duty service members, including meeting designated active-duty retention and hierarchical standards.2 We present a hypothetical case: An active-duty military patient presents to a new dermatologist for cosmetic enhancement of facial skin dyspigmentation. The patient will be leaving soon for deployment and will not be able to follow up for 9 months. How should the dermatologist treat a patient who cannot follow up for so long?
The therapeutic modalities offered can be impacted by forthcoming deployments3 that may result in delayed time to administer repeat treatments or follow-up. The patient may have high expectations for a single appointment for a condition that requires prolonged treatment courses. Because there often is no reliable mechanism for patients to obtain refills during deployment, any medications prescribed would need to be provided in advance for the entire deployment duration, which often is 6 to 9 months. Additionally, treatment monitoring or modifications are severely limited, especially in the context of treatment nonresponse or adverse reactions. Considering the unique limitations of this patient population, both military and civilian physicians are faced with a need to maximize beneficence and autonomy while balancing nonmaleficence and justice.
One possible option is to decline to treat until the patient can follow up after returning from deployment. However, denying a request for an active treatable indication for which the patient desires treatment compromises patient autonomy and beneficence. Further, treatment should be provided to patients equitably to maintain justice. Although there may be a role for discussing active monitoring with nonintervention with the patient, denying treatment can negatively impact their physical and mental health and may be harmful. However, the patient should know and fully understand the risks and benefits of nonintervention with limited follow-up, including suboptimal outcomes or adverse events.
Another possibility for the management of this case may be conducting a one-time laser or light-based therapy or a one-time superficial- to medium-depth chemical peel before the patient leaves on deployment. Often, a series of laser- or light-based treatments is required to maximize outcomes for dyspigmentation. Without follow-up and with possible deployment to an environment with high UV exposure, the patient may experience disease exacerbation or other adverse effects. Treatment of those adverse effects may be delayed, as further intervention is not possible during deployment. Lower initial laser settings may be safer but may not be highly effective initially. More rigorous treatment upon return from deployment may be considered. Similar to laser therapies, chemical peels usually require several treatments for optimal outcomes. Without follow-up and with potential deployment to remote environments, there is a risk for adverse events that outweighs the minimal benefit of a single treatment. Therefore, either intervention may violate the principle of nonmaleficence.
A more reasonable approach may be initiating topical therapy and following up via telemedicine evaluation. Topical therapy often is the least-invasive approach and carries a reduced risk for adverse effects. Triple-combination therapy with topical retinoids, hydroquinone, and topical steroids is a common first-line approach.4 Because this approach is patient dependent, therapy can be more easily modulated or halted in the context of undesired results. Additionally, if internet connectivity is available, an asynchronous telemedicine approach could be utilized during deployment to monitor and advise changes as necessary, provided the regulatory framework allows for it.5
Although there is no uniformly correct approach in a scenario of limited patient follow-up, the last solution may be most ethically favorable: to begin therapy with milder and safer therapies (topical) and defer higher-intensity regimens until the patient returns from deployment. This allows some treatment initiation to preserve justice, beneficence, and patient autonomy. Associated virtual follow-up via telemedicine also allows avoidance of nonmaleficence in this context.
- Hwang J, Kakimoto C. Teledermatology in the US military: a historic foundation for current and future applications. Cutis. 2018;101:335;337;345.
- Dodd JG, Grant-Kels JM. Ethical concerns in caring for active duty service members who may be seeking dermatologic care outside the military soon. Int J Womens Dermatol. 2020;6:445-447. doi:10.1016/j.ijwd.2020.07.001
- Burke KR, Larrymore DC, Cho S. Treatment consideration for US military members with skin disease. Cutis. 2019;103:329-332.
- Desai SR. Hyperpigmentation therapy: a review. J Clin Aesthet Dermatol. 2014;7:13-17.
- Hwang JS, Lappan CM, Sperling LC, et al. Utilization of telemedicine in the U.S. military in a deployed setting. Mil Med. 2014;179:1347-1353. doi:10.7205/MILMED-D-14-00115
- Hwang J, Kakimoto C. Teledermatology in the US military: a historic foundation for current and future applications. Cutis. 2018;101:335;337;345.
- Dodd JG, Grant-Kels JM. Ethical concerns in caring for active duty service members who may be seeking dermatologic care outside the military soon. Int J Womens Dermatol. 2020;6:445-447. doi:10.1016/j.ijwd.2020.07.001
- Burke KR, Larrymore DC, Cho S. Treatment consideration for US military members with skin disease. Cutis. 2019;103:329-332.
- Desai SR. Hyperpigmentation therapy: a review. J Clin Aesthet Dermatol. 2014;7:13-17.
- Hwang JS, Lappan CM, Sperling LC, et al. Utilization of telemedicine in the U.S. military in a deployed setting. Mil Med. 2014;179:1347-1353. doi:10.7205/MILMED-D-14-00115
PRACTICE POINTS
- Dermatologic conditions are among the most common concerns reported by active-duty service members.
- The unique considerations of deployments are important for dermatologists to consider in the treatment of skin disease.
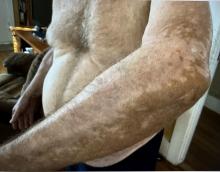
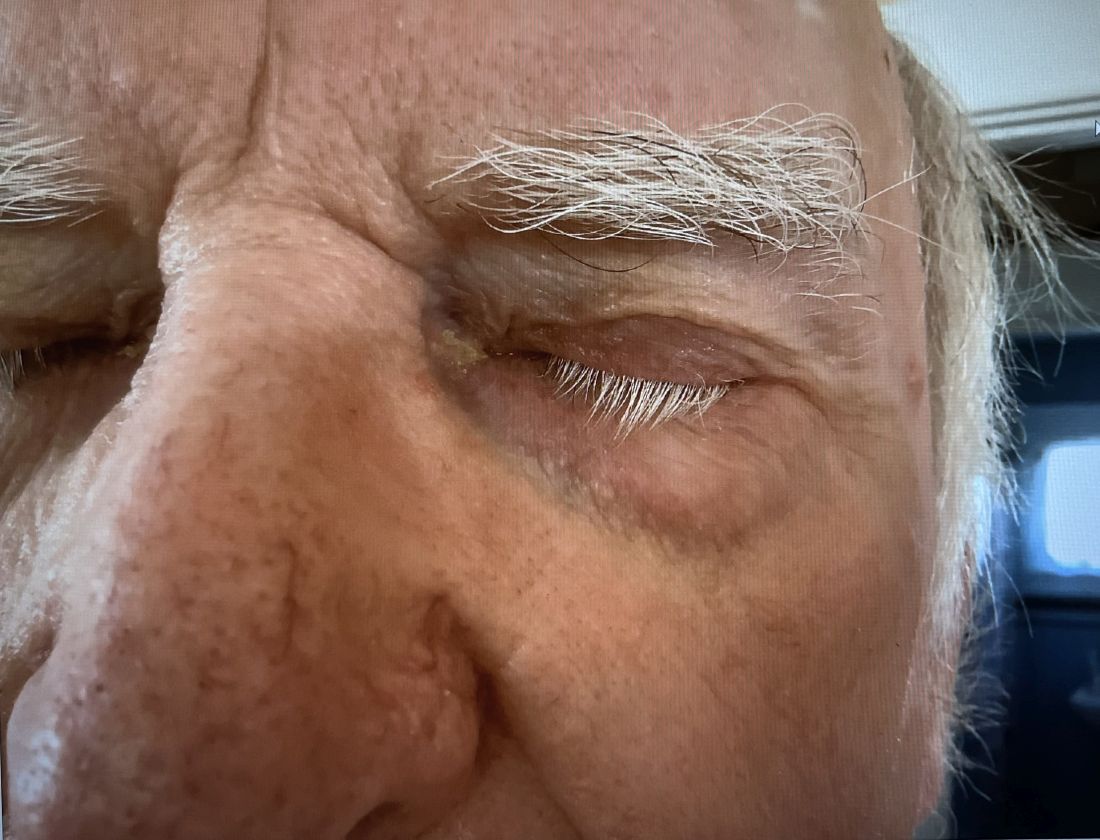
A 74-year-old White male presented with a 1-year history of depigmented patches on the hands, arms, and face, as well as white eyelashes and eyebrows
This patient showed no evidence of recurrence in the scar where the melanoma was excised, and had no enlarged lymph nodes on palpation. His complete blood count and liver function tests were normal. A positron emission tomography (PET) scan was ordered by Dr. Nasser that revealed hypermetabolic right paratracheal, right hilar, and subcarinal lymph nodes, highly suspicious for malignant lymph nodes. The patient was referred to oncology for metastatic melanoma treatment and has been doing well on ipilimumab and nivolumab.
Vitiligo is an autoimmune condition characterized by the progressive destruction of melanocytes resulting in hypopigmentation or depigmentation of the skin. Vitiligo has been associated with cutaneous melanoma. Melanoma-associated leukoderma occurs in a portion of patients with melanoma and is correlated with a favorable prognosis. Additionally, leukoderma has been described as a side effect of melanoma treatment itself. However, cases such as this one have also been reported of vitiligo-like depigmentation presenting prior to the diagnosis of metastatic melanoma.
Melanoma, like vitiligo, is considered highly immunogenic, and cytotoxic T lymphocytes (CTLs) can recognize antigens in melanoma. Furthermore, studies have shown a vitiligo-like halo around melanoma tumors, likely caused by T-cell recruitment, and this may lead to tumor destruction, but rarely total clearance. It seems that the CTL infiltrate in both diseases is similar, but regulatory T cells are decreased in vitiligo, whereas they are present in melanomas and may contribute to the immunosuppressive tumor microenvironment found at the margin of these lesions.
Leukoderma is also associated with melanoma immunotherapy which may be described as drug-induced leukoderma. Additionally, the frequency of recognition of melanoma cells by CTLs leading to hypopigmentation appears to be higher in those with metastatic disease. High immune infiltrate with CTLs and interferon-gamma (IFN-gamma) expression by type 1 T helper cells is associated with favorable prognosis. Immunotherapy with checkpoint inhibitors has shown promise in treatment augmentation for melanoma, but not all patients fully respond to therapy. Nonetheless, development of leukoderma with these treatments has been significantly associated with good therapeutic response. Depigmentation of hair and retinal epithelium has also been reported. However, drug-induced leukoderma and vitiligo seem to have clinical and biological differences, including family history of disease and serum chemokine levels. Vaccines are in production to aid in the treatment of melanoma, but researchers must first identify the appropriate antigen(s) to include.
Conversely, vitiligo-like depigmentation has been reported as a harbinger of metastatic melanoma. Patients with previous excision of primary melanoma have presented months or years later with depigmentation and, upon further evaluation, have been diagnosed with metastatic melanoma. The prevalence of depigmentation in melanoma patients is about 3%-6%, and is estimated to be 7-10 times more common in those with melanoma than in the general population. In most cases, hypopigmentation follows the diagnosis of melanoma, with an average of 4.8 years after the initial diagnosis and 1-2 years after lymph node or distant metastases. It is unclear whether hypopigmentation occurs before or after the growth of metastatic lesions, but this clinical finding in a patient with previous melanoma may serve as an important clue to conduct further investigation for metastasis.
This case and the photos were submitted by Lucas Shapiro, BS, of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Florida, and Natalie Y. Nasser, MD, Kaiser Permanente Riverside Medical Center; Riverside, California. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cerci FB et al. Cutis. 2017 Jun;99(6):E1-E2. PMID: 28686764.
Cho EA et al. Ann Dermatol. 2009 May;21(2):178-181.
Failla CM et al. Int J Mol Sci. 2019 Nov 15;20(22):5731.
This patient showed no evidence of recurrence in the scar where the melanoma was excised, and had no enlarged lymph nodes on palpation. His complete blood count and liver function tests were normal. A positron emission tomography (PET) scan was ordered by Dr. Nasser that revealed hypermetabolic right paratracheal, right hilar, and subcarinal lymph nodes, highly suspicious for malignant lymph nodes. The patient was referred to oncology for metastatic melanoma treatment and has been doing well on ipilimumab and nivolumab.
Vitiligo is an autoimmune condition characterized by the progressive destruction of melanocytes resulting in hypopigmentation or depigmentation of the skin. Vitiligo has been associated with cutaneous melanoma. Melanoma-associated leukoderma occurs in a portion of patients with melanoma and is correlated with a favorable prognosis. Additionally, leukoderma has been described as a side effect of melanoma treatment itself. However, cases such as this one have also been reported of vitiligo-like depigmentation presenting prior to the diagnosis of metastatic melanoma.
Melanoma, like vitiligo, is considered highly immunogenic, and cytotoxic T lymphocytes (CTLs) can recognize antigens in melanoma. Furthermore, studies have shown a vitiligo-like halo around melanoma tumors, likely caused by T-cell recruitment, and this may lead to tumor destruction, but rarely total clearance. It seems that the CTL infiltrate in both diseases is similar, but regulatory T cells are decreased in vitiligo, whereas they are present in melanomas and may contribute to the immunosuppressive tumor microenvironment found at the margin of these lesions.
Leukoderma is also associated with melanoma immunotherapy which may be described as drug-induced leukoderma. Additionally, the frequency of recognition of melanoma cells by CTLs leading to hypopigmentation appears to be higher in those with metastatic disease. High immune infiltrate with CTLs and interferon-gamma (IFN-gamma) expression by type 1 T helper cells is associated with favorable prognosis. Immunotherapy with checkpoint inhibitors has shown promise in treatment augmentation for melanoma, but not all patients fully respond to therapy. Nonetheless, development of leukoderma with these treatments has been significantly associated with good therapeutic response. Depigmentation of hair and retinal epithelium has also been reported. However, drug-induced leukoderma and vitiligo seem to have clinical and biological differences, including family history of disease and serum chemokine levels. Vaccines are in production to aid in the treatment of melanoma, but researchers must first identify the appropriate antigen(s) to include.
Conversely, vitiligo-like depigmentation has been reported as a harbinger of metastatic melanoma. Patients with previous excision of primary melanoma have presented months or years later with depigmentation and, upon further evaluation, have been diagnosed with metastatic melanoma. The prevalence of depigmentation in melanoma patients is about 3%-6%, and is estimated to be 7-10 times more common in those with melanoma than in the general population. In most cases, hypopigmentation follows the diagnosis of melanoma, with an average of 4.8 years after the initial diagnosis and 1-2 years after lymph node or distant metastases. It is unclear whether hypopigmentation occurs before or after the growth of metastatic lesions, but this clinical finding in a patient with previous melanoma may serve as an important clue to conduct further investigation for metastasis.
This case and the photos were submitted by Lucas Shapiro, BS, of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Florida, and Natalie Y. Nasser, MD, Kaiser Permanente Riverside Medical Center; Riverside, California. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cerci FB et al. Cutis. 2017 Jun;99(6):E1-E2. PMID: 28686764.
Cho EA et al. Ann Dermatol. 2009 May;21(2):178-181.
Failla CM et al. Int J Mol Sci. 2019 Nov 15;20(22):5731.
This patient showed no evidence of recurrence in the scar where the melanoma was excised, and had no enlarged lymph nodes on palpation. His complete blood count and liver function tests were normal. A positron emission tomography (PET) scan was ordered by Dr. Nasser that revealed hypermetabolic right paratracheal, right hilar, and subcarinal lymph nodes, highly suspicious for malignant lymph nodes. The patient was referred to oncology for metastatic melanoma treatment and has been doing well on ipilimumab and nivolumab.
Vitiligo is an autoimmune condition characterized by the progressive destruction of melanocytes resulting in hypopigmentation or depigmentation of the skin. Vitiligo has been associated with cutaneous melanoma. Melanoma-associated leukoderma occurs in a portion of patients with melanoma and is correlated with a favorable prognosis. Additionally, leukoderma has been described as a side effect of melanoma treatment itself. However, cases such as this one have also been reported of vitiligo-like depigmentation presenting prior to the diagnosis of metastatic melanoma.
Melanoma, like vitiligo, is considered highly immunogenic, and cytotoxic T lymphocytes (CTLs) can recognize antigens in melanoma. Furthermore, studies have shown a vitiligo-like halo around melanoma tumors, likely caused by T-cell recruitment, and this may lead to tumor destruction, but rarely total clearance. It seems that the CTL infiltrate in both diseases is similar, but regulatory T cells are decreased in vitiligo, whereas they are present in melanomas and may contribute to the immunosuppressive tumor microenvironment found at the margin of these lesions.
Leukoderma is also associated with melanoma immunotherapy which may be described as drug-induced leukoderma. Additionally, the frequency of recognition of melanoma cells by CTLs leading to hypopigmentation appears to be higher in those with metastatic disease. High immune infiltrate with CTLs and interferon-gamma (IFN-gamma) expression by type 1 T helper cells is associated with favorable prognosis. Immunotherapy with checkpoint inhibitors has shown promise in treatment augmentation for melanoma, but not all patients fully respond to therapy. Nonetheless, development of leukoderma with these treatments has been significantly associated with good therapeutic response. Depigmentation of hair and retinal epithelium has also been reported. However, drug-induced leukoderma and vitiligo seem to have clinical and biological differences, including family history of disease and serum chemokine levels. Vaccines are in production to aid in the treatment of melanoma, but researchers must first identify the appropriate antigen(s) to include.
Conversely, vitiligo-like depigmentation has been reported as a harbinger of metastatic melanoma. Patients with previous excision of primary melanoma have presented months or years later with depigmentation and, upon further evaluation, have been diagnosed with metastatic melanoma. The prevalence of depigmentation in melanoma patients is about 3%-6%, and is estimated to be 7-10 times more common in those with melanoma than in the general population. In most cases, hypopigmentation follows the diagnosis of melanoma, with an average of 4.8 years after the initial diagnosis and 1-2 years after lymph node or distant metastases. It is unclear whether hypopigmentation occurs before or after the growth of metastatic lesions, but this clinical finding in a patient with previous melanoma may serve as an important clue to conduct further investigation for metastasis.
This case and the photos were submitted by Lucas Shapiro, BS, of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Florida, and Natalie Y. Nasser, MD, Kaiser Permanente Riverside Medical Center; Riverside, California. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cerci FB et al. Cutis. 2017 Jun;99(6):E1-E2. PMID: 28686764.
Cho EA et al. Ann Dermatol. 2009 May;21(2):178-181.
Failla CM et al. Int J Mol Sci. 2019 Nov 15;20(22):5731.
When Babies ‘Stop Breathing,’ Who Needs Admission and a Workup?
Many infants have experienced an episode of apnea, defined as a pause in respiration of 20 seconds or more. Most episodes remain unexplained, and no underlying cause can be found. Historically, these were referred to as “near-miss SIDS,” episodes, but that label suggested that all of these events would have ended in death had someone not intervened. New descriptive terminology was needed.
In the mid-1980s, the term “apparent life-threatening event” (ALTE) was adopted. But that term, too, was an overstatement, because although scary for parents, these brief apnea episodes were not, in most cases, truly life-threatening.
In 2013, authors of a systematic review coined the term “brief resolved unexplained event” (BRUE). This review also addressed the history and physical exam features associated with risk for a subsequent episode. It was felt that hospitalization and testing might be warranted if certain infants could be identified as high risk for recurrence.
What Is Considered a BRUE?
In the current working definition of BRUE, the child must be < 1 year old. The episode must be a sudden, brief, and resolved, with one or more of these characteristics:
- Cyanosis or pallor (but not turning red)
- A change in breathing (absent, decreased, or irregular)
- A change in tone (hypertonia or hypotonia)
- A change in responsiveness.
Furthermore, to qualify as a BRUE, no explanation can be found for the event based on the history and physical examination but before any laboratory testing is done. The definition also excludes children with known potential explanatory diagnoses (such as gastroesophageal reflux or bronchiolitis) and those who are otherwise symptomatically ill at the time of the event.
Decision to Admit and Recurrence Risk
An apnea event in an otherwise healthy infant, regardless of what it’s called, puts providers and parents in a difficult position. Should the infant be hospitalized for further monitoring and potentially more invasive testing to determine the cause of the episode? And what are the chances that the episode will be repeated?
A clinical practice guideline (CPG) for BRUE, widely adopted in 2016, resulted in significant reductions in healthcare utilization. The CPG attempted to identify low-risk infants who could safely be discharged from the emergency department. Although the CPG improved outcomes, experts acknowledged that an underlying problem was not likely to be identified even among infants deemed high risk, and these infants would be hospitalized unnecessarily.
Available data were simply insufficient to support this decision. So, with the goal of identifying factors that could help predict recurrent BRUE risk, a 15-hospital collaborative study was undertaken, followed by the development and validation of a clinical decision rule for predicting the risk for a serious underlying diagnosis or event recurrence among infants presenting with BRUE.
Here’s what we learned from more than 3000 cases of BRUE.
First, it turns out that it’s not easy to determine whether an infant is at low or high risk for recurrence of BRUE. Initially, 91.5% of patients enrolled in the study would have been labeled high risk.
Furthermore, a BRUE recurred in 14.3% of the cohort, and 4.8% of high-risk infants were found to have a serious undiagnosed condition. Seizures, airway anomalies, and gastroesophageal reflux were the top three causes of BRUE, but the spectrum of underlying pathology was quite considerable.
The problem was that 4.6% of the entire cohort were found to have a serious underlying condition, nearly identical to the proportion of high-risk infants with these conditions. This prompted the question of whether simply labeling infants “high risk” was really appropriate any longer.
Revised BRUE Management
Although it hasn’t been possible to group infants neatly in low and high-risk categories, the data from that large cohort led to the development of the BRUE 2.0 criteria, which enabled more focused risk assessment of an infant who experienced a BRUE. With an app on MDCalc, these criteria allow providers to ascertain, and show families, a visual representation of their infant’s individualized risk for a subsequent BRUE and of having a serious underlying condition.
The cohort study also identified red flags from the history or physical exam of infants who experienced a BRUE: weight loss, failure to thrive, or a history of feeding problems. Exam findings such as a bulging fontanelle, forceful or bilious emesis, and evidence of gastrointestinal (GI) bleeding suggest a medical diagnosis rather than a BRUE. If GI-related causes are high on the differential, a feeding evaluation can be helpful. A feeding evaluation can be done in the outpatient setting and does not require hospitalization.
For suspicion of an underlying neurological condition (such as seizures), experts recommend obtaining a short EEG, which is highly sensitive for detecting infantile spasms and encephalopathy. They recommend reserving MRI for infants with abnormalities on EEG or physical exam. Metabolic or genetic testing should be done only if the infant looks ill, because most patients with genetic or inborn errors of metabolism will continue to have symptoms as they become older.
The approach to BRUE has moved into the realm of shared decision-making with families. The likelihood of identifying a serious diagnosis is low for most of these children. And unfortunately, no single test can diagnose the full spectrum of potential explanatory diagnoses. For example, data from 2023 demonstrate that only 1.1% of lab tests following a BRUE contributed to a diagnosis, and most of the time that was a positive viral test. Similarly, imaging was helpful in only 1.5% of cases. So, explaining the evidence and deciding along with parents what is reasonable to do (or not do) is the current state of affairs.
My Take
As I reflect back on two and a half decades of caring for these patients, I believe that recent data have helped us a great deal. We do less testing and admit fewer infants to the hospital than we did 20 years ago, and that’s a good thing. Nevertheless, looking for a few red flags, having a high index of suspicion when the clinical exam is abnormal, and engaging in shared decision-making with families can help make the caring for these challenging patients more bearable and lead to better outcomes for all involved.
Dr. Basco is Professor, Department of Pediatrics, Medical University of South Carolina (MUSC); Director, Division of General Pediatrics, Department of Pediatrics, MUSC Children’s Hospital, Charleston, South Carolina. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many infants have experienced an episode of apnea, defined as a pause in respiration of 20 seconds or more. Most episodes remain unexplained, and no underlying cause can be found. Historically, these were referred to as “near-miss SIDS,” episodes, but that label suggested that all of these events would have ended in death had someone not intervened. New descriptive terminology was needed.
In the mid-1980s, the term “apparent life-threatening event” (ALTE) was adopted. But that term, too, was an overstatement, because although scary for parents, these brief apnea episodes were not, in most cases, truly life-threatening.
In 2013, authors of a systematic review coined the term “brief resolved unexplained event” (BRUE). This review also addressed the history and physical exam features associated with risk for a subsequent episode. It was felt that hospitalization and testing might be warranted if certain infants could be identified as high risk for recurrence.
What Is Considered a BRUE?
In the current working definition of BRUE, the child must be < 1 year old. The episode must be a sudden, brief, and resolved, with one or more of these characteristics:
- Cyanosis or pallor (but not turning red)
- A change in breathing (absent, decreased, or irregular)
- A change in tone (hypertonia or hypotonia)
- A change in responsiveness.
Furthermore, to qualify as a BRUE, no explanation can be found for the event based on the history and physical examination but before any laboratory testing is done. The definition also excludes children with known potential explanatory diagnoses (such as gastroesophageal reflux or bronchiolitis) and those who are otherwise symptomatically ill at the time of the event.
Decision to Admit and Recurrence Risk
An apnea event in an otherwise healthy infant, regardless of what it’s called, puts providers and parents in a difficult position. Should the infant be hospitalized for further monitoring and potentially more invasive testing to determine the cause of the episode? And what are the chances that the episode will be repeated?
A clinical practice guideline (CPG) for BRUE, widely adopted in 2016, resulted in significant reductions in healthcare utilization. The CPG attempted to identify low-risk infants who could safely be discharged from the emergency department. Although the CPG improved outcomes, experts acknowledged that an underlying problem was not likely to be identified even among infants deemed high risk, and these infants would be hospitalized unnecessarily.
Available data were simply insufficient to support this decision. So, with the goal of identifying factors that could help predict recurrent BRUE risk, a 15-hospital collaborative study was undertaken, followed by the development and validation of a clinical decision rule for predicting the risk for a serious underlying diagnosis or event recurrence among infants presenting with BRUE.
Here’s what we learned from more than 3000 cases of BRUE.
First, it turns out that it’s not easy to determine whether an infant is at low or high risk for recurrence of BRUE. Initially, 91.5% of patients enrolled in the study would have been labeled high risk.
Furthermore, a BRUE recurred in 14.3% of the cohort, and 4.8% of high-risk infants were found to have a serious undiagnosed condition. Seizures, airway anomalies, and gastroesophageal reflux were the top three causes of BRUE, but the spectrum of underlying pathology was quite considerable.
The problem was that 4.6% of the entire cohort were found to have a serious underlying condition, nearly identical to the proportion of high-risk infants with these conditions. This prompted the question of whether simply labeling infants “high risk” was really appropriate any longer.
Revised BRUE Management
Although it hasn’t been possible to group infants neatly in low and high-risk categories, the data from that large cohort led to the development of the BRUE 2.0 criteria, which enabled more focused risk assessment of an infant who experienced a BRUE. With an app on MDCalc, these criteria allow providers to ascertain, and show families, a visual representation of their infant’s individualized risk for a subsequent BRUE and of having a serious underlying condition.
The cohort study also identified red flags from the history or physical exam of infants who experienced a BRUE: weight loss, failure to thrive, or a history of feeding problems. Exam findings such as a bulging fontanelle, forceful or bilious emesis, and evidence of gastrointestinal (GI) bleeding suggest a medical diagnosis rather than a BRUE. If GI-related causes are high on the differential, a feeding evaluation can be helpful. A feeding evaluation can be done in the outpatient setting and does not require hospitalization.
For suspicion of an underlying neurological condition (such as seizures), experts recommend obtaining a short EEG, which is highly sensitive for detecting infantile spasms and encephalopathy. They recommend reserving MRI for infants with abnormalities on EEG or physical exam. Metabolic or genetic testing should be done only if the infant looks ill, because most patients with genetic or inborn errors of metabolism will continue to have symptoms as they become older.
The approach to BRUE has moved into the realm of shared decision-making with families. The likelihood of identifying a serious diagnosis is low for most of these children. And unfortunately, no single test can diagnose the full spectrum of potential explanatory diagnoses. For example, data from 2023 demonstrate that only 1.1% of lab tests following a BRUE contributed to a diagnosis, and most of the time that was a positive viral test. Similarly, imaging was helpful in only 1.5% of cases. So, explaining the evidence and deciding along with parents what is reasonable to do (or not do) is the current state of affairs.
My Take
As I reflect back on two and a half decades of caring for these patients, I believe that recent data have helped us a great deal. We do less testing and admit fewer infants to the hospital than we did 20 years ago, and that’s a good thing. Nevertheless, looking for a few red flags, having a high index of suspicion when the clinical exam is abnormal, and engaging in shared decision-making with families can help make the caring for these challenging patients more bearable and lead to better outcomes for all involved.
Dr. Basco is Professor, Department of Pediatrics, Medical University of South Carolina (MUSC); Director, Division of General Pediatrics, Department of Pediatrics, MUSC Children’s Hospital, Charleston, South Carolina. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many infants have experienced an episode of apnea, defined as a pause in respiration of 20 seconds or more. Most episodes remain unexplained, and no underlying cause can be found. Historically, these were referred to as “near-miss SIDS,” episodes, but that label suggested that all of these events would have ended in death had someone not intervened. New descriptive terminology was needed.
In the mid-1980s, the term “apparent life-threatening event” (ALTE) was adopted. But that term, too, was an overstatement, because although scary for parents, these brief apnea episodes were not, in most cases, truly life-threatening.
In 2013, authors of a systematic review coined the term “brief resolved unexplained event” (BRUE). This review also addressed the history and physical exam features associated with risk for a subsequent episode. It was felt that hospitalization and testing might be warranted if certain infants could be identified as high risk for recurrence.
What Is Considered a BRUE?
In the current working definition of BRUE, the child must be < 1 year old. The episode must be a sudden, brief, and resolved, with one or more of these characteristics:
- Cyanosis or pallor (but not turning red)
- A change in breathing (absent, decreased, or irregular)
- A change in tone (hypertonia or hypotonia)
- A change in responsiveness.
Furthermore, to qualify as a BRUE, no explanation can be found for the event based on the history and physical examination but before any laboratory testing is done. The definition also excludes children with known potential explanatory diagnoses (such as gastroesophageal reflux or bronchiolitis) and those who are otherwise symptomatically ill at the time of the event.
Decision to Admit and Recurrence Risk
An apnea event in an otherwise healthy infant, regardless of what it’s called, puts providers and parents in a difficult position. Should the infant be hospitalized for further monitoring and potentially more invasive testing to determine the cause of the episode? And what are the chances that the episode will be repeated?
A clinical practice guideline (CPG) for BRUE, widely adopted in 2016, resulted in significant reductions in healthcare utilization. The CPG attempted to identify low-risk infants who could safely be discharged from the emergency department. Although the CPG improved outcomes, experts acknowledged that an underlying problem was not likely to be identified even among infants deemed high risk, and these infants would be hospitalized unnecessarily.
Available data were simply insufficient to support this decision. So, with the goal of identifying factors that could help predict recurrent BRUE risk, a 15-hospital collaborative study was undertaken, followed by the development and validation of a clinical decision rule for predicting the risk for a serious underlying diagnosis or event recurrence among infants presenting with BRUE.
Here’s what we learned from more than 3000 cases of BRUE.
First, it turns out that it’s not easy to determine whether an infant is at low or high risk for recurrence of BRUE. Initially, 91.5% of patients enrolled in the study would have been labeled high risk.
Furthermore, a BRUE recurred in 14.3% of the cohort, and 4.8% of high-risk infants were found to have a serious undiagnosed condition. Seizures, airway anomalies, and gastroesophageal reflux were the top three causes of BRUE, but the spectrum of underlying pathology was quite considerable.
The problem was that 4.6% of the entire cohort were found to have a serious underlying condition, nearly identical to the proportion of high-risk infants with these conditions. This prompted the question of whether simply labeling infants “high risk” was really appropriate any longer.
Revised BRUE Management
Although it hasn’t been possible to group infants neatly in low and high-risk categories, the data from that large cohort led to the development of the BRUE 2.0 criteria, which enabled more focused risk assessment of an infant who experienced a BRUE. With an app on MDCalc, these criteria allow providers to ascertain, and show families, a visual representation of their infant’s individualized risk for a subsequent BRUE and of having a serious underlying condition.
The cohort study also identified red flags from the history or physical exam of infants who experienced a BRUE: weight loss, failure to thrive, or a history of feeding problems. Exam findings such as a bulging fontanelle, forceful or bilious emesis, and evidence of gastrointestinal (GI) bleeding suggest a medical diagnosis rather than a BRUE. If GI-related causes are high on the differential, a feeding evaluation can be helpful. A feeding evaluation can be done in the outpatient setting and does not require hospitalization.
For suspicion of an underlying neurological condition (such as seizures), experts recommend obtaining a short EEG, which is highly sensitive for detecting infantile spasms and encephalopathy. They recommend reserving MRI for infants with abnormalities on EEG or physical exam. Metabolic or genetic testing should be done only if the infant looks ill, because most patients with genetic or inborn errors of metabolism will continue to have symptoms as they become older.
The approach to BRUE has moved into the realm of shared decision-making with families. The likelihood of identifying a serious diagnosis is low for most of these children. And unfortunately, no single test can diagnose the full spectrum of potential explanatory diagnoses. For example, data from 2023 demonstrate that only 1.1% of lab tests following a BRUE contributed to a diagnosis, and most of the time that was a positive viral test. Similarly, imaging was helpful in only 1.5% of cases. So, explaining the evidence and deciding along with parents what is reasonable to do (or not do) is the current state of affairs.
My Take
As I reflect back on two and a half decades of caring for these patients, I believe that recent data have helped us a great deal. We do less testing and admit fewer infants to the hospital than we did 20 years ago, and that’s a good thing. Nevertheless, looking for a few red flags, having a high index of suspicion when the clinical exam is abnormal, and engaging in shared decision-making with families can help make the caring for these challenging patients more bearable and lead to better outcomes for all involved.
Dr. Basco is Professor, Department of Pediatrics, Medical University of South Carolina (MUSC); Director, Division of General Pediatrics, Department of Pediatrics, MUSC Children’s Hospital, Charleston, South Carolina. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New Guidelines: Brain Death Is Equal to Heart Death, Says Ethicist
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the New York University Grossman School of Medicine in New York City.
I think we had a breakthrough on a very controversial subject over the past month. Over and over again, debates have been breaking out, cases have been going to court, and fights have been coming to ethics committees about brain death. How do we know what brain death is, how do we diagnose it, and what rights do families have with respect to the diagnosis?
The American Academy of Neurology decided to form a task force, and they just issued guidelines on the definition, tests to use it, and the rights of families. They did a wonderful job, in my view. They›ve achieved clarity.
First, they tried to handle both adults and children. Children are, if you will, more difficult — and that’s been known — to test for brain death. Their brains are smaller. You get more interference and false signals coming from muscle or nerve activity that might be going on elsewhere in their bodies.
The guidelines say we’re going to try to see whether a person can breathe without support. If it’s an adult, one test over a 24-hour period would be sufficient. If you had them off the ventilator and they can’t breathe and show no signs of being able to do that, that’s a very fundamental test for brain death. For children, you’re going to have to do it twice. The guidelines are saying to be cautious.
Second, they say it’s very important to know the cause of the suspected brain death condition. If someone has a massive head injury, that’s different from a situation in which someone overdoses from drugs or drowns. Those conditions can be a little deceptive. In the case of drowning, sometimes the brain has protective mechanisms to protect circulation to the brain naturally for a little bit of time. I’m talking about minutes, not hours.
You want to be careful to make sure that you know the cause of the massive brain injury or insult that makes someone believe that the patient is brain-dead, whether it’s a stroke, an embolism, a bleed, a gunshot wound, or trauma to the head. Those factors really drive the certainty with which brain death should be pronounced. I think that’s very, very important.
They also said that brain death means the permanent loss of brain function. You may get a few cells still firing or you may be in a situation, because the life support is still there, where the body looks pink and perhaps might appear to still be alive to someone. When you know that the damage to the brain is so severe that there’s nothing that can be done to bring back the support of heart function, breathing, and most likely any ability to sense or feel anything, that is death.
I believe it’s very important, when talking to families, to say there are two ways that we pronounce people dead, and they’re equal: One is to say their heart has stopped, their breathing has stopped, and there’s nothing we can do to resuscitate them, which is cardiac death. The other is to say their brain has permanently ceased to function in any kind of integrated way. That means no heartbeat, no breathing, and no mental sensations. That is death.
In approaching families, it is critical that doctors and nurses don’t say, “Your relative is brain-dead.” That gives the family a sense that maybe they’re only “partially dead” or maybe there’s one key organ that has stopped working but maybe you can bring it back. Death is death. The law recognizes both cardiac death and brain death as death.
When you approach a family, if you believe that death has occurred, you say, “I’m very sorry. With regret, I have to tell you, your loved one is dead.” If they ask how you know, you can say, “We’ve determined it through brain death or through cardiac death.” You don’t give them a sense that people could be kind of dead, sort of dead, or nearly dead. Those states are comas or permanent vegetative states; they’re not the same as death.
What if the family says, “I don’t want you to do any testing. I don’t want to find out whether my relative is dead”? The American Academy of Neurology looked at this carefully and said that any test for death can be done without the permission or consent of the family. They said that because doctors need to know what steps to take to treat someone.
If a person is dead, then treatment is going to stop. It may not stop immediately. There may be issues about organ donation. There may be issues about gathering the family to come to the bedside to say goodbye, because many people think that’s more humane than saying goodbye at the morgue or in another setting.
This is all well and good, but patients cannot protect against bad news when it comes to death. We don’t want to be doing things to the dead that cost money or are futile because of death and using resources that might go to others.
We’ve got much more clarity than we have ever had with respect to the issue of brain death and how it works in any hospital. We have certain tests, including being off the ventilator and some other tests, that the guidelines supply. We know we have to be more careful with children. We want to know the etiology of the cause of the brain trauma, the devastating brain injury, to be sure that this is something that really is permanent cessation of integrated brain function.
We know that if you believe the person has died, you don’t need the consent of the family in order to do a brain-death test. You have to do it because there is no point in continuing treatment in expensive ICU settings and denying resources to others who might want to use those resources. The family can’t hold the medical team hostage.
We do know that when we approach someone with the determination, whatever it is, we should lead by saying that the person has died and then explain how that was determined, whether it be by cardiac death pronouncement — where you tried to resuscitate and the heart’s not beating — or brain-death analysis.
I’m Art Caplan at the Division of Medical Ethics at the NYU Grossman School of Medicine. Thanks for watching.
Dr. Caplan has disclosed the following relevant financial relationships: Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position); serves as a contributing author and adviser for this news organization.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the New York University Grossman School of Medicine in New York City.
I think we had a breakthrough on a very controversial subject over the past month. Over and over again, debates have been breaking out, cases have been going to court, and fights have been coming to ethics committees about brain death. How do we know what brain death is, how do we diagnose it, and what rights do families have with respect to the diagnosis?
The American Academy of Neurology decided to form a task force, and they just issued guidelines on the definition, tests to use it, and the rights of families. They did a wonderful job, in my view. They›ve achieved clarity.
First, they tried to handle both adults and children. Children are, if you will, more difficult — and that’s been known — to test for brain death. Their brains are smaller. You get more interference and false signals coming from muscle or nerve activity that might be going on elsewhere in their bodies.
The guidelines say we’re going to try to see whether a person can breathe without support. If it’s an adult, one test over a 24-hour period would be sufficient. If you had them off the ventilator and they can’t breathe and show no signs of being able to do that, that’s a very fundamental test for brain death. For children, you’re going to have to do it twice. The guidelines are saying to be cautious.
Second, they say it’s very important to know the cause of the suspected brain death condition. If someone has a massive head injury, that’s different from a situation in which someone overdoses from drugs or drowns. Those conditions can be a little deceptive. In the case of drowning, sometimes the brain has protective mechanisms to protect circulation to the brain naturally for a little bit of time. I’m talking about minutes, not hours.
You want to be careful to make sure that you know the cause of the massive brain injury or insult that makes someone believe that the patient is brain-dead, whether it’s a stroke, an embolism, a bleed, a gunshot wound, or trauma to the head. Those factors really drive the certainty with which brain death should be pronounced. I think that’s very, very important.
They also said that brain death means the permanent loss of brain function. You may get a few cells still firing or you may be in a situation, because the life support is still there, where the body looks pink and perhaps might appear to still be alive to someone. When you know that the damage to the brain is so severe that there’s nothing that can be done to bring back the support of heart function, breathing, and most likely any ability to sense or feel anything, that is death.
I believe it’s very important, when talking to families, to say there are two ways that we pronounce people dead, and they’re equal: One is to say their heart has stopped, their breathing has stopped, and there’s nothing we can do to resuscitate them, which is cardiac death. The other is to say their brain has permanently ceased to function in any kind of integrated way. That means no heartbeat, no breathing, and no mental sensations. That is death.
In approaching families, it is critical that doctors and nurses don’t say, “Your relative is brain-dead.” That gives the family a sense that maybe they’re only “partially dead” or maybe there’s one key organ that has stopped working but maybe you can bring it back. Death is death. The law recognizes both cardiac death and brain death as death.
When you approach a family, if you believe that death has occurred, you say, “I’m very sorry. With regret, I have to tell you, your loved one is dead.” If they ask how you know, you can say, “We’ve determined it through brain death or through cardiac death.” You don’t give them a sense that people could be kind of dead, sort of dead, or nearly dead. Those states are comas or permanent vegetative states; they’re not the same as death.
What if the family says, “I don’t want you to do any testing. I don’t want to find out whether my relative is dead”? The American Academy of Neurology looked at this carefully and said that any test for death can be done without the permission or consent of the family. They said that because doctors need to know what steps to take to treat someone.
If a person is dead, then treatment is going to stop. It may not stop immediately. There may be issues about organ donation. There may be issues about gathering the family to come to the bedside to say goodbye, because many people think that’s more humane than saying goodbye at the morgue or in another setting.
This is all well and good, but patients cannot protect against bad news when it comes to death. We don’t want to be doing things to the dead that cost money or are futile because of death and using resources that might go to others.
We’ve got much more clarity than we have ever had with respect to the issue of brain death and how it works in any hospital. We have certain tests, including being off the ventilator and some other tests, that the guidelines supply. We know we have to be more careful with children. We want to know the etiology of the cause of the brain trauma, the devastating brain injury, to be sure that this is something that really is permanent cessation of integrated brain function.
We know that if you believe the person has died, you don’t need the consent of the family in order to do a brain-death test. You have to do it because there is no point in continuing treatment in expensive ICU settings and denying resources to others who might want to use those resources. The family can’t hold the medical team hostage.
We do know that when we approach someone with the determination, whatever it is, we should lead by saying that the person has died and then explain how that was determined, whether it be by cardiac death pronouncement — where you tried to resuscitate and the heart’s not beating — or brain-death analysis.
I’m Art Caplan at the Division of Medical Ethics at the NYU Grossman School of Medicine. Thanks for watching.
Dr. Caplan has disclosed the following relevant financial relationships: Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position); serves as a contributing author and adviser for this news organization.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the New York University Grossman School of Medicine in New York City.
I think we had a breakthrough on a very controversial subject over the past month. Over and over again, debates have been breaking out, cases have been going to court, and fights have been coming to ethics committees about brain death. How do we know what brain death is, how do we diagnose it, and what rights do families have with respect to the diagnosis?
The American Academy of Neurology decided to form a task force, and they just issued guidelines on the definition, tests to use it, and the rights of families. They did a wonderful job, in my view. They›ve achieved clarity.
First, they tried to handle both adults and children. Children are, if you will, more difficult — and that’s been known — to test for brain death. Their brains are smaller. You get more interference and false signals coming from muscle or nerve activity that might be going on elsewhere in their bodies.
The guidelines say we’re going to try to see whether a person can breathe without support. If it’s an adult, one test over a 24-hour period would be sufficient. If you had them off the ventilator and they can’t breathe and show no signs of being able to do that, that’s a very fundamental test for brain death. For children, you’re going to have to do it twice. The guidelines are saying to be cautious.
Second, they say it’s very important to know the cause of the suspected brain death condition. If someone has a massive head injury, that’s different from a situation in which someone overdoses from drugs or drowns. Those conditions can be a little deceptive. In the case of drowning, sometimes the brain has protective mechanisms to protect circulation to the brain naturally for a little bit of time. I’m talking about minutes, not hours.
You want to be careful to make sure that you know the cause of the massive brain injury or insult that makes someone believe that the patient is brain-dead, whether it’s a stroke, an embolism, a bleed, a gunshot wound, or trauma to the head. Those factors really drive the certainty with which brain death should be pronounced. I think that’s very, very important.
They also said that brain death means the permanent loss of brain function. You may get a few cells still firing or you may be in a situation, because the life support is still there, where the body looks pink and perhaps might appear to still be alive to someone. When you know that the damage to the brain is so severe that there’s nothing that can be done to bring back the support of heart function, breathing, and most likely any ability to sense or feel anything, that is death.
I believe it’s very important, when talking to families, to say there are two ways that we pronounce people dead, and they’re equal: One is to say their heart has stopped, their breathing has stopped, and there’s nothing we can do to resuscitate them, which is cardiac death. The other is to say their brain has permanently ceased to function in any kind of integrated way. That means no heartbeat, no breathing, and no mental sensations. That is death.
In approaching families, it is critical that doctors and nurses don’t say, “Your relative is brain-dead.” That gives the family a sense that maybe they’re only “partially dead” or maybe there’s one key organ that has stopped working but maybe you can bring it back. Death is death. The law recognizes both cardiac death and brain death as death.
When you approach a family, if you believe that death has occurred, you say, “I’m very sorry. With regret, I have to tell you, your loved one is dead.” If they ask how you know, you can say, “We’ve determined it through brain death or through cardiac death.” You don’t give them a sense that people could be kind of dead, sort of dead, or nearly dead. Those states are comas or permanent vegetative states; they’re not the same as death.
What if the family says, “I don’t want you to do any testing. I don’t want to find out whether my relative is dead”? The American Academy of Neurology looked at this carefully and said that any test for death can be done without the permission or consent of the family. They said that because doctors need to know what steps to take to treat someone.
If a person is dead, then treatment is going to stop. It may not stop immediately. There may be issues about organ donation. There may be issues about gathering the family to come to the bedside to say goodbye, because many people think that’s more humane than saying goodbye at the morgue or in another setting.
This is all well and good, but patients cannot protect against bad news when it comes to death. We don’t want to be doing things to the dead that cost money or are futile because of death and using resources that might go to others.
We’ve got much more clarity than we have ever had with respect to the issue of brain death and how it works in any hospital. We have certain tests, including being off the ventilator and some other tests, that the guidelines supply. We know we have to be more careful with children. We want to know the etiology of the cause of the brain trauma, the devastating brain injury, to be sure that this is something that really is permanent cessation of integrated brain function.
We know that if you believe the person has died, you don’t need the consent of the family in order to do a brain-death test. You have to do it because there is no point in continuing treatment in expensive ICU settings and denying resources to others who might want to use those resources. The family can’t hold the medical team hostage.
We do know that when we approach someone with the determination, whatever it is, we should lead by saying that the person has died and then explain how that was determined, whether it be by cardiac death pronouncement — where you tried to resuscitate and the heart’s not beating — or brain-death analysis.
I’m Art Caplan at the Division of Medical Ethics at the NYU Grossman School of Medicine. Thanks for watching.
Dr. Caplan has disclosed the following relevant financial relationships: Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position); serves as a contributing author and adviser for this news organization.
A version of this article appeared on Medscape.com.
Long COVID: Another Great Pretender
Has COVID replaced syphilis as the great imitator? At the turn of the 20th century, Sir William Osler was quoted as saying, “He who knows syphilis knows medicine.”
If you have any doubts about the “imitator” analogy, simply use any broad search engine or AI portal and enter “what is the differential diagnosis of primary syphilis, or secondary syphilis, or tertiary syphilis?” A plethora of diseases across many body systems appears.
Another Great Pretender?
Did you or do you now have long COVID? How do you know? Do you even know what long COVID is? How would you diagnosis it?
When asked about long COVID, this is what large language model source bard.google.com had to say:
Long COVID, also known as post-COVID conditions or PASC, is a complex condition that can occur in people who have had COVID-19, even if their initial illness was mild or even asymptomatic. It’s characterized by a wide range of symptoms that can last for weeks, months, or even years after the initial infection.
Symptoms of long COVID can include: fatigue, shortness of breath, brain fog (trouble thinking or concentrating), loss of smell or taste, sleep problems, muscle pain, joint pain, headache, dizziness, depression or anxiety, heart palpitations, gastrointestinal problems.
Duh!!! It reads much like a continuation of the symptoms of acute COVID. Bard continued:
The exact cause of long COVID is still unknown, but there are several theories. One possibility is that the virus itself can damage organs and tissues, leading to ongoing symptoms. Another possibility is that the immune system’s response to the virus can continue to cause inflammation even after the virus is gone.
Human intelligence source Wikipedia says this:
Long COVID or long-haul COVID is a group of health problems persisting or developing after an initial COVID-19 infection. Symptoms can last weeks, months or years and are often debilitating. The World Health Organization defines long COVID as starting three months after infection, but other definitions put the start of long COVID at four weeks.
Highly varied, including post-exertional malaise (symptoms made worse with effort), fatigue, muscle pain, shortness of breath, chest pain, and cognitive dysfunction (brain fog).
Acute COVID to Long COVID
The World Health Organization estimates that 36 million people in the European region have developed long COVID in the first 3 years of the pandemic. That›s a lot.
We all know that the common signs and symptoms of acute COVID-19 include fever or chills, a dry cough and shortness of breath, feeling very tired, muscle or body aches, headache, loss of taste or smell, sore throat, congestion, runny nose, nausea, vomiting, and diarrhea. Except for the taste and smell findings, every one of these symptoms or signs could indicate a different virus infection or even some type of allergy. My point is the nonspecificity in this list.
Uncommon signs and symptoms of acute COVID include a flat skin rash covered with small bumps, discolored swollen areas on the fingers and toes (COVID toes), and hives. The skin of hands, wrists, or ankles also can be affected. Blisters, itchiness, rough skin, or pus can be seen.
Severe confusion (delirium) might be the main or only symptom of COVID-19 in older people. This COVID-19 symptom is linked with a high risk for poor outcomes, including death. Pink eye (conjunctivitis) can be a COVID-19 symptom. Other eye problems linked to COVID-19 are light sensitivity, sore eyes, and itchy eyes. Acute myocarditis, tinnitus, vertigo, and hearing loss have been reported. And 1-4 weeks after the onset of COVID-19 infection, a patient may experience de novo reactive synovitis and arthritis of any joints.
So, take your pick. Myriad symptoms, signs, diseases, diagnoses, and organ systems — still present, recurring, just appearing, apparently de novo, or after asymptomatic infection. We have so much still to learn.
What big-time symptoms, signs, and major diseases are not on any of these lists? Obviously, cancer, atherosclerotic cardiovascular diseases, obesity, bone diseases, and competitive infections. But be patient; the lingering effects of direct tissue invasion by the virus as well as a wide range of immunologic reactions may just be getting started. Mitochondrial damage, especially in muscles, is increasingly a pathophysiologic suspect.
Human diseases can be physical or mental; and in COVID, that twain not only meet but mix and mingle freely, and may even merge into psychosoma. Don’t ever forget that. Consider “fatigue.” Who among us, COVID or NOVID, does not experience that from time to time?
Or consider brain fog as a common reported symptom of COVID. What on earth is that actually? How can a person know they have brain fog, or whether they had it and are over it?
We need one or more lab or other diagnostic tests that can objectively confirm the diagnosis of long COVID.
Useful Progress?
A recent research paper in Science reported intriguing chemical findings that seemed to point a finger at some form of complement dysregulation as a potential disease marker for long COVID. Unfortunately, some critics have pointed out that this entire study may be invalid or irrelevant because the New York cohort was recruited in 2020, before vaccines were available. The Zurich cohort was recruited up until April 2021, so some may have been vaccinated.
Then this news organization came along in early January 2024 with an article about COVID causing not only more than a million American deaths but also more than 5000 deaths from long COVID. We physicians don’t really know what long COVID even is, but we have to sign death certificates blaming thousands of deaths on it anyway? And rolling back the clock to 2020: Are patients dying from COVID or with COVID, according to death certificates?Now, armed with the knowledge that “documented serious post–COVID-19 conditions include cardiovascular, pulmonary, neurological, renal, endocrine, hematological, and gastrointestinal complications, as well as death,” CDC has published clear and fairly concise instructions on how to address post-acute COVID sequelae on death certificates.
In late January, this news organization painted a hopeful picture by naming four phenotypes of long COVID, suggesting that such divisions might further our understanding, including prognosis, and even therapy for this condition. Among the clinical phenotypes of (1) chronic fatigue–like syndrome, headache, and memory loss; (2) respiratory syndrome (which includes cough and difficulty breathing); (3) chronic pain; and (4) neurosensorial syndrome (which causes an altered sense of taste and smell), overlap is clearly possible but isn›t addressed.
I see these recent developments as needed and useful progress, but we are still left with…not much. So, when you tell me that you do or do not have long COVID, I will say to you, “How do you know?”
I also say: She/he/they who know COVID know medicine.
A version of this article first appeared on Medscape.com.
Has COVID replaced syphilis as the great imitator? At the turn of the 20th century, Sir William Osler was quoted as saying, “He who knows syphilis knows medicine.”
If you have any doubts about the “imitator” analogy, simply use any broad search engine or AI portal and enter “what is the differential diagnosis of primary syphilis, or secondary syphilis, or tertiary syphilis?” A plethora of diseases across many body systems appears.
Another Great Pretender?
Did you or do you now have long COVID? How do you know? Do you even know what long COVID is? How would you diagnosis it?
When asked about long COVID, this is what large language model source bard.google.com had to say:
Long COVID, also known as post-COVID conditions or PASC, is a complex condition that can occur in people who have had COVID-19, even if their initial illness was mild or even asymptomatic. It’s characterized by a wide range of symptoms that can last for weeks, months, or even years after the initial infection.
Symptoms of long COVID can include: fatigue, shortness of breath, brain fog (trouble thinking or concentrating), loss of smell or taste, sleep problems, muscle pain, joint pain, headache, dizziness, depression or anxiety, heart palpitations, gastrointestinal problems.
Duh!!! It reads much like a continuation of the symptoms of acute COVID. Bard continued:
The exact cause of long COVID is still unknown, but there are several theories. One possibility is that the virus itself can damage organs and tissues, leading to ongoing symptoms. Another possibility is that the immune system’s response to the virus can continue to cause inflammation even after the virus is gone.
Human intelligence source Wikipedia says this:
Long COVID or long-haul COVID is a group of health problems persisting or developing after an initial COVID-19 infection. Symptoms can last weeks, months or years and are often debilitating. The World Health Organization defines long COVID as starting three months after infection, but other definitions put the start of long COVID at four weeks.
Highly varied, including post-exertional malaise (symptoms made worse with effort), fatigue, muscle pain, shortness of breath, chest pain, and cognitive dysfunction (brain fog).
Acute COVID to Long COVID
The World Health Organization estimates that 36 million people in the European region have developed long COVID in the first 3 years of the pandemic. That›s a lot.
We all know that the common signs and symptoms of acute COVID-19 include fever or chills, a dry cough and shortness of breath, feeling very tired, muscle or body aches, headache, loss of taste or smell, sore throat, congestion, runny nose, nausea, vomiting, and diarrhea. Except for the taste and smell findings, every one of these symptoms or signs could indicate a different virus infection or even some type of allergy. My point is the nonspecificity in this list.
Uncommon signs and symptoms of acute COVID include a flat skin rash covered with small bumps, discolored swollen areas on the fingers and toes (COVID toes), and hives. The skin of hands, wrists, or ankles also can be affected. Blisters, itchiness, rough skin, or pus can be seen.
Severe confusion (delirium) might be the main or only symptom of COVID-19 in older people. This COVID-19 symptom is linked with a high risk for poor outcomes, including death. Pink eye (conjunctivitis) can be a COVID-19 symptom. Other eye problems linked to COVID-19 are light sensitivity, sore eyes, and itchy eyes. Acute myocarditis, tinnitus, vertigo, and hearing loss have been reported. And 1-4 weeks after the onset of COVID-19 infection, a patient may experience de novo reactive synovitis and arthritis of any joints.
So, take your pick. Myriad symptoms, signs, diseases, diagnoses, and organ systems — still present, recurring, just appearing, apparently de novo, or after asymptomatic infection. We have so much still to learn.
What big-time symptoms, signs, and major diseases are not on any of these lists? Obviously, cancer, atherosclerotic cardiovascular diseases, obesity, bone diseases, and competitive infections. But be patient; the lingering effects of direct tissue invasion by the virus as well as a wide range of immunologic reactions may just be getting started. Mitochondrial damage, especially in muscles, is increasingly a pathophysiologic suspect.
Human diseases can be physical or mental; and in COVID, that twain not only meet but mix and mingle freely, and may even merge into psychosoma. Don’t ever forget that. Consider “fatigue.” Who among us, COVID or NOVID, does not experience that from time to time?
Or consider brain fog as a common reported symptom of COVID. What on earth is that actually? How can a person know they have brain fog, or whether they had it and are over it?
We need one or more lab or other diagnostic tests that can objectively confirm the diagnosis of long COVID.
Useful Progress?
A recent research paper in Science reported intriguing chemical findings that seemed to point a finger at some form of complement dysregulation as a potential disease marker for long COVID. Unfortunately, some critics have pointed out that this entire study may be invalid or irrelevant because the New York cohort was recruited in 2020, before vaccines were available. The Zurich cohort was recruited up until April 2021, so some may have been vaccinated.
Then this news organization came along in early January 2024 with an article about COVID causing not only more than a million American deaths but also more than 5000 deaths from long COVID. We physicians don’t really know what long COVID even is, but we have to sign death certificates blaming thousands of deaths on it anyway? And rolling back the clock to 2020: Are patients dying from COVID or with COVID, according to death certificates?Now, armed with the knowledge that “documented serious post–COVID-19 conditions include cardiovascular, pulmonary, neurological, renal, endocrine, hematological, and gastrointestinal complications, as well as death,” CDC has published clear and fairly concise instructions on how to address post-acute COVID sequelae on death certificates.
In late January, this news organization painted a hopeful picture by naming four phenotypes of long COVID, suggesting that such divisions might further our understanding, including prognosis, and even therapy for this condition. Among the clinical phenotypes of (1) chronic fatigue–like syndrome, headache, and memory loss; (2) respiratory syndrome (which includes cough and difficulty breathing); (3) chronic pain; and (4) neurosensorial syndrome (which causes an altered sense of taste and smell), overlap is clearly possible but isn›t addressed.
I see these recent developments as needed and useful progress, but we are still left with…not much. So, when you tell me that you do or do not have long COVID, I will say to you, “How do you know?”
I also say: She/he/they who know COVID know medicine.
A version of this article first appeared on Medscape.com.
Has COVID replaced syphilis as the great imitator? At the turn of the 20th century, Sir William Osler was quoted as saying, “He who knows syphilis knows medicine.”
If you have any doubts about the “imitator” analogy, simply use any broad search engine or AI portal and enter “what is the differential diagnosis of primary syphilis, or secondary syphilis, or tertiary syphilis?” A plethora of diseases across many body systems appears.
Another Great Pretender?
Did you or do you now have long COVID? How do you know? Do you even know what long COVID is? How would you diagnosis it?
When asked about long COVID, this is what large language model source bard.google.com had to say:
Long COVID, also known as post-COVID conditions or PASC, is a complex condition that can occur in people who have had COVID-19, even if their initial illness was mild or even asymptomatic. It’s characterized by a wide range of symptoms that can last for weeks, months, or even years after the initial infection.
Symptoms of long COVID can include: fatigue, shortness of breath, brain fog (trouble thinking or concentrating), loss of smell or taste, sleep problems, muscle pain, joint pain, headache, dizziness, depression or anxiety, heart palpitations, gastrointestinal problems.
Duh!!! It reads much like a continuation of the symptoms of acute COVID. Bard continued:
The exact cause of long COVID is still unknown, but there are several theories. One possibility is that the virus itself can damage organs and tissues, leading to ongoing symptoms. Another possibility is that the immune system’s response to the virus can continue to cause inflammation even after the virus is gone.
Human intelligence source Wikipedia says this:
Long COVID or long-haul COVID is a group of health problems persisting or developing after an initial COVID-19 infection. Symptoms can last weeks, months or years and are often debilitating. The World Health Organization defines long COVID as starting three months after infection, but other definitions put the start of long COVID at four weeks.
Highly varied, including post-exertional malaise (symptoms made worse with effort), fatigue, muscle pain, shortness of breath, chest pain, and cognitive dysfunction (brain fog).
Acute COVID to Long COVID
The World Health Organization estimates that 36 million people in the European region have developed long COVID in the first 3 years of the pandemic. That›s a lot.
We all know that the common signs and symptoms of acute COVID-19 include fever or chills, a dry cough and shortness of breath, feeling very tired, muscle or body aches, headache, loss of taste or smell, sore throat, congestion, runny nose, nausea, vomiting, and diarrhea. Except for the taste and smell findings, every one of these symptoms or signs could indicate a different virus infection or even some type of allergy. My point is the nonspecificity in this list.
Uncommon signs and symptoms of acute COVID include a flat skin rash covered with small bumps, discolored swollen areas on the fingers and toes (COVID toes), and hives. The skin of hands, wrists, or ankles also can be affected. Blisters, itchiness, rough skin, or pus can be seen.
Severe confusion (delirium) might be the main or only symptom of COVID-19 in older people. This COVID-19 symptom is linked with a high risk for poor outcomes, including death. Pink eye (conjunctivitis) can be a COVID-19 symptom. Other eye problems linked to COVID-19 are light sensitivity, sore eyes, and itchy eyes. Acute myocarditis, tinnitus, vertigo, and hearing loss have been reported. And 1-4 weeks after the onset of COVID-19 infection, a patient may experience de novo reactive synovitis and arthritis of any joints.
So, take your pick. Myriad symptoms, signs, diseases, diagnoses, and organ systems — still present, recurring, just appearing, apparently de novo, or after asymptomatic infection. We have so much still to learn.
What big-time symptoms, signs, and major diseases are not on any of these lists? Obviously, cancer, atherosclerotic cardiovascular diseases, obesity, bone diseases, and competitive infections. But be patient; the lingering effects of direct tissue invasion by the virus as well as a wide range of immunologic reactions may just be getting started. Mitochondrial damage, especially in muscles, is increasingly a pathophysiologic suspect.
Human diseases can be physical or mental; and in COVID, that twain not only meet but mix and mingle freely, and may even merge into psychosoma. Don’t ever forget that. Consider “fatigue.” Who among us, COVID or NOVID, does not experience that from time to time?
Or consider brain fog as a common reported symptom of COVID. What on earth is that actually? How can a person know they have brain fog, or whether they had it and are over it?
We need one or more lab or other diagnostic tests that can objectively confirm the diagnosis of long COVID.
Useful Progress?
A recent research paper in Science reported intriguing chemical findings that seemed to point a finger at some form of complement dysregulation as a potential disease marker for long COVID. Unfortunately, some critics have pointed out that this entire study may be invalid or irrelevant because the New York cohort was recruited in 2020, before vaccines were available. The Zurich cohort was recruited up until April 2021, so some may have been vaccinated.
Then this news organization came along in early January 2024 with an article about COVID causing not only more than a million American deaths but also more than 5000 deaths from long COVID. We physicians don’t really know what long COVID even is, but we have to sign death certificates blaming thousands of deaths on it anyway? And rolling back the clock to 2020: Are patients dying from COVID or with COVID, according to death certificates?Now, armed with the knowledge that “documented serious post–COVID-19 conditions include cardiovascular, pulmonary, neurological, renal, endocrine, hematological, and gastrointestinal complications, as well as death,” CDC has published clear and fairly concise instructions on how to address post-acute COVID sequelae on death certificates.
In late January, this news organization painted a hopeful picture by naming four phenotypes of long COVID, suggesting that such divisions might further our understanding, including prognosis, and even therapy for this condition. Among the clinical phenotypes of (1) chronic fatigue–like syndrome, headache, and memory loss; (2) respiratory syndrome (which includes cough and difficulty breathing); (3) chronic pain; and (4) neurosensorial syndrome (which causes an altered sense of taste and smell), overlap is clearly possible but isn›t addressed.
I see these recent developments as needed and useful progress, but we are still left with…not much. So, when you tell me that you do or do not have long COVID, I will say to you, “How do you know?”
I also say: She/he/they who know COVID know medicine.
A version of this article first appeared on Medscape.com.
How to Prescribe Physical Activity in Patients With Obesity
Exercise should no longer be a mere “complement” or a standard recommendation within healthy lifestyle guidelines, say experts. Recent evidence confirms its physiological importance and endorses its beneficial and therapeutic effects on overall health, particularly in the case of obesity and its comorbidities. These findings emphasized the reasons to include exercise prescription in addressing this condition. This conclusion emerged from discussions among experts in Physical Activity and Sports Sciences during the XIX Congress of the Spanish Society for Obesity, where the role of physical exercise as a therapeutic strategy was analyzed from various perspectives.
Javier Butragueño, PhD, coordinator of the Exercise Working Group at the Spanish Society of Obesity, emphasized the need to “reposition” the role of exercise and the message conveyed to the population. “We must move beyond the typical recommendation to ‘just walk’ and rethink this message. When working with patients with obesity, you realize that, for example, the guideline of 10,000 steps per day makes little sense for those who weigh 140 kg, have been sedentary for a long time, and have not reached 2000 daily steps. Clinically, it becomes evident that current recommendations may not align with the needs of these patients,” he said.
Precision Focus
Dr. Butragueño highlighted the necessity of shifting the central focus from weight-related variables alone. While weight is crucial, evidence suggests that it should be evaluated along with other strategies, such as nutrition and pharmacology.
“The approach must change to view exercise as a metabolism regulator,” said Dr. Butragueño. “For specialists, this means educating the population about the need to stay active for overall health. This is a disruptive message because the prevailing idea, almost obsessive, associates exercise primarily with weight loss, a completely incorrect approach that can even be detrimental in some cases.”
Dr. Butragueño emphasized the supportive role of physical exercise in interventions for these patients. “Data show that it is both an enhancer and a co-adjuvant in strategies that also include psychology and endocrinology. It should be part of the approach to obesity but individualized and phenotyped to give physical activity the necessary dimension in each specific case.”
As an example of this adaptability in therapeutic strategy, Dr. Butragueño referred to addressing binge eating disorder. “In this case, specialists must acknowledge that sports are a third-line option, always behind the psychologist, who plays a primary role. Exercise is used to enhance the emotions triggered through its practice, considering that many of these patients maintain a very negative relationship with their bodies.”
Spanish ‘Prescription Guide’
During his presentation, Dr. Butragueño introduced the positioning document from the Exercise Group of the Spanish Society of Obesity, which is aimed at designing physical activity programs for patients with obesity. He emphasized its importance as a much-needed effort at proposing intervention strategies to guide health professionals and establish a reference framework for collaboration across different approaches to obesity.
Among the noteworthy aspects of the guidelines outlined in this document, Dr. Butragueño highlighted the assessment and classification of physical activity into four levels based on each patient’s physical condition. “This aspect should be studied by the scientific community because ‘humanizing’ exercise prescription by understanding individuals’ needs beyond their BMI is crucial.”
He also discussed the strategy outlined in the document that he said is crucial for implementing an exercise program. “Essentially, it involves two guidelines: First, engage in physical activity for at least 30-60 minutes in what we call zone 2. This includes activities like walking, cycling, or rowing, where one can speak easily with another person or sing without getting out of breath. This is a fundamental part of addressing obesity, as it improves mitochondrial biogenesis, the correct utilization of fatty acids, which is a significant concern in the pathophysiology of obesity and other diseases like cancer.”
The second strategy involves strength training alone or combined with aerobic-cardiovascular exercise. “Studies show that just 20 minutes of strength training 1 day a week for 10 consecutive weeks significantly improves strength levels in sedentary individuals.”
Dr. Butragueño emphasized that to date, there is no doubt that the most effective approach is to combine strength exercises with cardiorespiratory exercises. “This is not only to address obesity but also because, beyond weight impact, this training has proven additional benefits, such as increased oxygenation and improved cognitive capacity.”
Finally, regarding the challenges this shift in focus poses for exercise specialists, Dr. Butragueño pointed out, “Synergies in obesity treatment require sports experts to receive training in other disciplines, elevating our knowledge level and communication with the medical community to emphasize that we are indeed talking about exercise physiology applied to a condition like obesity.”
“In addition, as scientists, we must challenge ourselves to disseminate information at the societal level, surpassing the typical and outdated message of ‘eat less and move more,’ which we know is incorrect. This simplistic formula doesn’t help many patients resolve their issues like fatty liver, diabetes, and other metabolic disorders,” he concluded.
Active Breaks
Other topics debated during the congress included the importance of making exercise prescription a de facto reality in clinical practice and the challenge of achieving therapeutic compliance.
According to experts, one of the well-positioned trends in this regard is the concept of “active breaks” or “exercise snacks.” These breaks involve engaging in short-duration, moderate- to high-intensity activities throughout the day or working hours.
César Bustos, a board member of the Spanish Society of Obesity, mentioned that several studies have demonstrated that simple activities like climbing three flights of stairs or engaging in 1-minute training sessions can increase the metabolic equivalent of cardiovascular capacity and cardiorespiratory fitness. This approach could help reduce cardiovascular disease risk and all-cause mortality by 13%-15%.
“Cardiorespiratory fitness is the ability to engage in physical activity. It has been proven to be a more powerful predictor of mortality risk than traditional risk factors such as hypertension, smoking, obesity, hyperlipidemia, and type 2 diabetes,” said Mr. Bustos.
The expert added that these findings on the benefits of exercise snacks are particularly relevant in the current context, where lack of time is the primary obstacle cited by individuals with obesity for not engaging in regular physical activity. In addition, exercise prescription is considered the primary preventive measure for obesity and its associated diseases.
“Exercise is an essential complement to various treatments and strategies aimed at managing obesity and maintaining long-term weight reductions. However, patient compliance with recommended measures to stay active remains low. This deficiency can be overcome with the adoption of exercise snacks or small doses of exercise, which have become the most effective tool for achieving this goal,” he emphasized.
Also, in line with other experts, Mr. Bustos emphasized the importance of combined strength and cardiovascular training within the same session. “Undoubtedly, this is the most effective modality, as recent meta-analyses reflect. There is also a second effective modality for improving cardiometabolic parameters in patients with obesity: Hybrid training, including games, skipping ropes, and various devices.”
Exerkines and Poly Pills
Antonio García-Hermoso, PhD, a specialist in physical activity and sports at Navarrabiomed, University Hospital of Navarra in Pamplona, Spain, provided an update on the latest evidence regarding exerkines, which are molecules released during exercise. Research into these molecules attempts to analyze and understand the complex network of interactions between various exercise response systems.
Dr. García-Hermoso said that in the case of obesity and type 2 diabetes, research focuses on how exercise can affect patients’ exerkine levels and how these molecules can affect cardiometabolic control.
“The results demonstrate that these molecules are associated with multiple benefits, including improved insulin sensitivity and glucose homeostasis,” said Dr. García-Hermoso. “Concerning obesity, regular exercise has been shown to reduce interleukin-6 levels, positively affecting inflammation in these patients, also being associated with increased lipolysis and fatty acid utilization.”
Dr. García-Hermoso considered that studying exerkines supports the importance of individualized exercise prescription, like prescription of diet or medications.
He emphasized the importance of intensity, “which is even more crucial than the type of physical activity. Intense exercise activates physiological mechanisms, such as increased blood lactate levels, favoring the inhibition of ghrelin signaling associated with appetite. Therefore, higher exercise intensity leads to more lactate and greater inhibition of post-training hunger.”
“It is essential to understand that exercise is a poly pill with many advantages, and one of them is that even in small amounts, if intensity is increased, health benefits increase considerably,” Dr. García-Hermoso concluded.
Dr. Butragueño, Mr. Bustos, and Dr. García-Hermoso declared no relevant economic conflicts of interest.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
Exercise should no longer be a mere “complement” or a standard recommendation within healthy lifestyle guidelines, say experts. Recent evidence confirms its physiological importance and endorses its beneficial and therapeutic effects on overall health, particularly in the case of obesity and its comorbidities. These findings emphasized the reasons to include exercise prescription in addressing this condition. This conclusion emerged from discussions among experts in Physical Activity and Sports Sciences during the XIX Congress of the Spanish Society for Obesity, where the role of physical exercise as a therapeutic strategy was analyzed from various perspectives.
Javier Butragueño, PhD, coordinator of the Exercise Working Group at the Spanish Society of Obesity, emphasized the need to “reposition” the role of exercise and the message conveyed to the population. “We must move beyond the typical recommendation to ‘just walk’ and rethink this message. When working with patients with obesity, you realize that, for example, the guideline of 10,000 steps per day makes little sense for those who weigh 140 kg, have been sedentary for a long time, and have not reached 2000 daily steps. Clinically, it becomes evident that current recommendations may not align with the needs of these patients,” he said.
Precision Focus
Dr. Butragueño highlighted the necessity of shifting the central focus from weight-related variables alone. While weight is crucial, evidence suggests that it should be evaluated along with other strategies, such as nutrition and pharmacology.
“The approach must change to view exercise as a metabolism regulator,” said Dr. Butragueño. “For specialists, this means educating the population about the need to stay active for overall health. This is a disruptive message because the prevailing idea, almost obsessive, associates exercise primarily with weight loss, a completely incorrect approach that can even be detrimental in some cases.”
Dr. Butragueño emphasized the supportive role of physical exercise in interventions for these patients. “Data show that it is both an enhancer and a co-adjuvant in strategies that also include psychology and endocrinology. It should be part of the approach to obesity but individualized and phenotyped to give physical activity the necessary dimension in each specific case.”
As an example of this adaptability in therapeutic strategy, Dr. Butragueño referred to addressing binge eating disorder. “In this case, specialists must acknowledge that sports are a third-line option, always behind the psychologist, who plays a primary role. Exercise is used to enhance the emotions triggered through its practice, considering that many of these patients maintain a very negative relationship with their bodies.”
Spanish ‘Prescription Guide’
During his presentation, Dr. Butragueño introduced the positioning document from the Exercise Group of the Spanish Society of Obesity, which is aimed at designing physical activity programs for patients with obesity. He emphasized its importance as a much-needed effort at proposing intervention strategies to guide health professionals and establish a reference framework for collaboration across different approaches to obesity.
Among the noteworthy aspects of the guidelines outlined in this document, Dr. Butragueño highlighted the assessment and classification of physical activity into four levels based on each patient’s physical condition. “This aspect should be studied by the scientific community because ‘humanizing’ exercise prescription by understanding individuals’ needs beyond their BMI is crucial.”
He also discussed the strategy outlined in the document that he said is crucial for implementing an exercise program. “Essentially, it involves two guidelines: First, engage in physical activity for at least 30-60 minutes in what we call zone 2. This includes activities like walking, cycling, or rowing, where one can speak easily with another person or sing without getting out of breath. This is a fundamental part of addressing obesity, as it improves mitochondrial biogenesis, the correct utilization of fatty acids, which is a significant concern in the pathophysiology of obesity and other diseases like cancer.”
The second strategy involves strength training alone or combined with aerobic-cardiovascular exercise. “Studies show that just 20 minutes of strength training 1 day a week for 10 consecutive weeks significantly improves strength levels in sedentary individuals.”
Dr. Butragueño emphasized that to date, there is no doubt that the most effective approach is to combine strength exercises with cardiorespiratory exercises. “This is not only to address obesity but also because, beyond weight impact, this training has proven additional benefits, such as increased oxygenation and improved cognitive capacity.”
Finally, regarding the challenges this shift in focus poses for exercise specialists, Dr. Butragueño pointed out, “Synergies in obesity treatment require sports experts to receive training in other disciplines, elevating our knowledge level and communication with the medical community to emphasize that we are indeed talking about exercise physiology applied to a condition like obesity.”
“In addition, as scientists, we must challenge ourselves to disseminate information at the societal level, surpassing the typical and outdated message of ‘eat less and move more,’ which we know is incorrect. This simplistic formula doesn’t help many patients resolve their issues like fatty liver, diabetes, and other metabolic disorders,” he concluded.
Active Breaks
Other topics debated during the congress included the importance of making exercise prescription a de facto reality in clinical practice and the challenge of achieving therapeutic compliance.
According to experts, one of the well-positioned trends in this regard is the concept of “active breaks” or “exercise snacks.” These breaks involve engaging in short-duration, moderate- to high-intensity activities throughout the day or working hours.
César Bustos, a board member of the Spanish Society of Obesity, mentioned that several studies have demonstrated that simple activities like climbing three flights of stairs or engaging in 1-minute training sessions can increase the metabolic equivalent of cardiovascular capacity and cardiorespiratory fitness. This approach could help reduce cardiovascular disease risk and all-cause mortality by 13%-15%.
“Cardiorespiratory fitness is the ability to engage in physical activity. It has been proven to be a more powerful predictor of mortality risk than traditional risk factors such as hypertension, smoking, obesity, hyperlipidemia, and type 2 diabetes,” said Mr. Bustos.
The expert added that these findings on the benefits of exercise snacks are particularly relevant in the current context, where lack of time is the primary obstacle cited by individuals with obesity for not engaging in regular physical activity. In addition, exercise prescription is considered the primary preventive measure for obesity and its associated diseases.
“Exercise is an essential complement to various treatments and strategies aimed at managing obesity and maintaining long-term weight reductions. However, patient compliance with recommended measures to stay active remains low. This deficiency can be overcome with the adoption of exercise snacks or small doses of exercise, which have become the most effective tool for achieving this goal,” he emphasized.
Also, in line with other experts, Mr. Bustos emphasized the importance of combined strength and cardiovascular training within the same session. “Undoubtedly, this is the most effective modality, as recent meta-analyses reflect. There is also a second effective modality for improving cardiometabolic parameters in patients with obesity: Hybrid training, including games, skipping ropes, and various devices.”
Exerkines and Poly Pills
Antonio García-Hermoso, PhD, a specialist in physical activity and sports at Navarrabiomed, University Hospital of Navarra in Pamplona, Spain, provided an update on the latest evidence regarding exerkines, which are molecules released during exercise. Research into these molecules attempts to analyze and understand the complex network of interactions between various exercise response systems.
Dr. García-Hermoso said that in the case of obesity and type 2 diabetes, research focuses on how exercise can affect patients’ exerkine levels and how these molecules can affect cardiometabolic control.
“The results demonstrate that these molecules are associated with multiple benefits, including improved insulin sensitivity and glucose homeostasis,” said Dr. García-Hermoso. “Concerning obesity, regular exercise has been shown to reduce interleukin-6 levels, positively affecting inflammation in these patients, also being associated with increased lipolysis and fatty acid utilization.”
Dr. García-Hermoso considered that studying exerkines supports the importance of individualized exercise prescription, like prescription of diet or medications.
He emphasized the importance of intensity, “which is even more crucial than the type of physical activity. Intense exercise activates physiological mechanisms, such as increased blood lactate levels, favoring the inhibition of ghrelin signaling associated with appetite. Therefore, higher exercise intensity leads to more lactate and greater inhibition of post-training hunger.”
“It is essential to understand that exercise is a poly pill with many advantages, and one of them is that even in small amounts, if intensity is increased, health benefits increase considerably,” Dr. García-Hermoso concluded.
Dr. Butragueño, Mr. Bustos, and Dr. García-Hermoso declared no relevant economic conflicts of interest.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
Exercise should no longer be a mere “complement” or a standard recommendation within healthy lifestyle guidelines, say experts. Recent evidence confirms its physiological importance and endorses its beneficial and therapeutic effects on overall health, particularly in the case of obesity and its comorbidities. These findings emphasized the reasons to include exercise prescription in addressing this condition. This conclusion emerged from discussions among experts in Physical Activity and Sports Sciences during the XIX Congress of the Spanish Society for Obesity, where the role of physical exercise as a therapeutic strategy was analyzed from various perspectives.
Javier Butragueño, PhD, coordinator of the Exercise Working Group at the Spanish Society of Obesity, emphasized the need to “reposition” the role of exercise and the message conveyed to the population. “We must move beyond the typical recommendation to ‘just walk’ and rethink this message. When working with patients with obesity, you realize that, for example, the guideline of 10,000 steps per day makes little sense for those who weigh 140 kg, have been sedentary for a long time, and have not reached 2000 daily steps. Clinically, it becomes evident that current recommendations may not align with the needs of these patients,” he said.
Precision Focus
Dr. Butragueño highlighted the necessity of shifting the central focus from weight-related variables alone. While weight is crucial, evidence suggests that it should be evaluated along with other strategies, such as nutrition and pharmacology.
“The approach must change to view exercise as a metabolism regulator,” said Dr. Butragueño. “For specialists, this means educating the population about the need to stay active for overall health. This is a disruptive message because the prevailing idea, almost obsessive, associates exercise primarily with weight loss, a completely incorrect approach that can even be detrimental in some cases.”
Dr. Butragueño emphasized the supportive role of physical exercise in interventions for these patients. “Data show that it is both an enhancer and a co-adjuvant in strategies that also include psychology and endocrinology. It should be part of the approach to obesity but individualized and phenotyped to give physical activity the necessary dimension in each specific case.”
As an example of this adaptability in therapeutic strategy, Dr. Butragueño referred to addressing binge eating disorder. “In this case, specialists must acknowledge that sports are a third-line option, always behind the psychologist, who plays a primary role. Exercise is used to enhance the emotions triggered through its practice, considering that many of these patients maintain a very negative relationship with their bodies.”
Spanish ‘Prescription Guide’
During his presentation, Dr. Butragueño introduced the positioning document from the Exercise Group of the Spanish Society of Obesity, which is aimed at designing physical activity programs for patients with obesity. He emphasized its importance as a much-needed effort at proposing intervention strategies to guide health professionals and establish a reference framework for collaboration across different approaches to obesity.
Among the noteworthy aspects of the guidelines outlined in this document, Dr. Butragueño highlighted the assessment and classification of physical activity into four levels based on each patient’s physical condition. “This aspect should be studied by the scientific community because ‘humanizing’ exercise prescription by understanding individuals’ needs beyond their BMI is crucial.”
He also discussed the strategy outlined in the document that he said is crucial for implementing an exercise program. “Essentially, it involves two guidelines: First, engage in physical activity for at least 30-60 minutes in what we call zone 2. This includes activities like walking, cycling, or rowing, where one can speak easily with another person or sing without getting out of breath. This is a fundamental part of addressing obesity, as it improves mitochondrial biogenesis, the correct utilization of fatty acids, which is a significant concern in the pathophysiology of obesity and other diseases like cancer.”
The second strategy involves strength training alone or combined with aerobic-cardiovascular exercise. “Studies show that just 20 minutes of strength training 1 day a week for 10 consecutive weeks significantly improves strength levels in sedentary individuals.”
Dr. Butragueño emphasized that to date, there is no doubt that the most effective approach is to combine strength exercises with cardiorespiratory exercises. “This is not only to address obesity but also because, beyond weight impact, this training has proven additional benefits, such as increased oxygenation and improved cognitive capacity.”
Finally, regarding the challenges this shift in focus poses for exercise specialists, Dr. Butragueño pointed out, “Synergies in obesity treatment require sports experts to receive training in other disciplines, elevating our knowledge level and communication with the medical community to emphasize that we are indeed talking about exercise physiology applied to a condition like obesity.”
“In addition, as scientists, we must challenge ourselves to disseminate information at the societal level, surpassing the typical and outdated message of ‘eat less and move more,’ which we know is incorrect. This simplistic formula doesn’t help many patients resolve their issues like fatty liver, diabetes, and other metabolic disorders,” he concluded.
Active Breaks
Other topics debated during the congress included the importance of making exercise prescription a de facto reality in clinical practice and the challenge of achieving therapeutic compliance.
According to experts, one of the well-positioned trends in this regard is the concept of “active breaks” or “exercise snacks.” These breaks involve engaging in short-duration, moderate- to high-intensity activities throughout the day or working hours.
César Bustos, a board member of the Spanish Society of Obesity, mentioned that several studies have demonstrated that simple activities like climbing three flights of stairs or engaging in 1-minute training sessions can increase the metabolic equivalent of cardiovascular capacity and cardiorespiratory fitness. This approach could help reduce cardiovascular disease risk and all-cause mortality by 13%-15%.
“Cardiorespiratory fitness is the ability to engage in physical activity. It has been proven to be a more powerful predictor of mortality risk than traditional risk factors such as hypertension, smoking, obesity, hyperlipidemia, and type 2 diabetes,” said Mr. Bustos.
The expert added that these findings on the benefits of exercise snacks are particularly relevant in the current context, where lack of time is the primary obstacle cited by individuals with obesity for not engaging in regular physical activity. In addition, exercise prescription is considered the primary preventive measure for obesity and its associated diseases.
“Exercise is an essential complement to various treatments and strategies aimed at managing obesity and maintaining long-term weight reductions. However, patient compliance with recommended measures to stay active remains low. This deficiency can be overcome with the adoption of exercise snacks or small doses of exercise, which have become the most effective tool for achieving this goal,” he emphasized.
Also, in line with other experts, Mr. Bustos emphasized the importance of combined strength and cardiovascular training within the same session. “Undoubtedly, this is the most effective modality, as recent meta-analyses reflect. There is also a second effective modality for improving cardiometabolic parameters in patients with obesity: Hybrid training, including games, skipping ropes, and various devices.”
Exerkines and Poly Pills
Antonio García-Hermoso, PhD, a specialist in physical activity and sports at Navarrabiomed, University Hospital of Navarra in Pamplona, Spain, provided an update on the latest evidence regarding exerkines, which are molecules released during exercise. Research into these molecules attempts to analyze and understand the complex network of interactions between various exercise response systems.
Dr. García-Hermoso said that in the case of obesity and type 2 diabetes, research focuses on how exercise can affect patients’ exerkine levels and how these molecules can affect cardiometabolic control.
“The results demonstrate that these molecules are associated with multiple benefits, including improved insulin sensitivity and glucose homeostasis,” said Dr. García-Hermoso. “Concerning obesity, regular exercise has been shown to reduce interleukin-6 levels, positively affecting inflammation in these patients, also being associated with increased lipolysis and fatty acid utilization.”
Dr. García-Hermoso considered that studying exerkines supports the importance of individualized exercise prescription, like prescription of diet or medications.
He emphasized the importance of intensity, “which is even more crucial than the type of physical activity. Intense exercise activates physiological mechanisms, such as increased blood lactate levels, favoring the inhibition of ghrelin signaling associated with appetite. Therefore, higher exercise intensity leads to more lactate and greater inhibition of post-training hunger.”
“It is essential to understand that exercise is a poly pill with many advantages, and one of them is that even in small amounts, if intensity is increased, health benefits increase considerably,” Dr. García-Hermoso concluded.
Dr. Butragueño, Mr. Bustos, and Dr. García-Hermoso declared no relevant economic conflicts of interest.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.