User login
75-year-old man • recent history of hand-foot-mouth disease • discolored fingernails and toenails lifting from the proximal end • Dx?
THE CASE
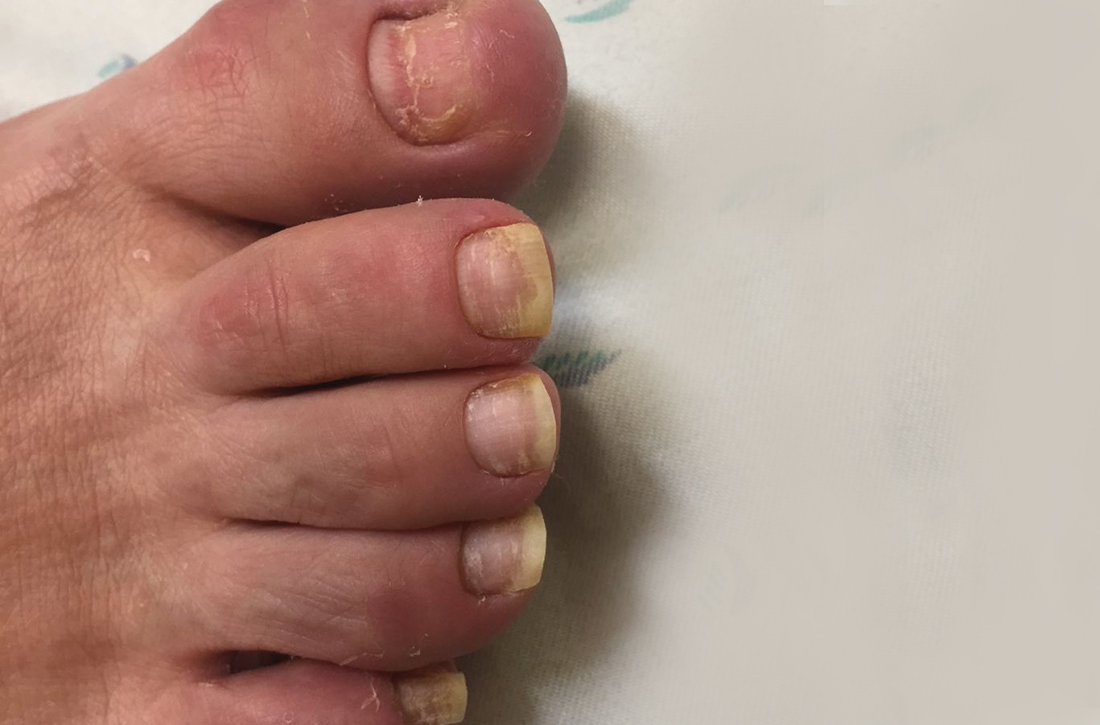
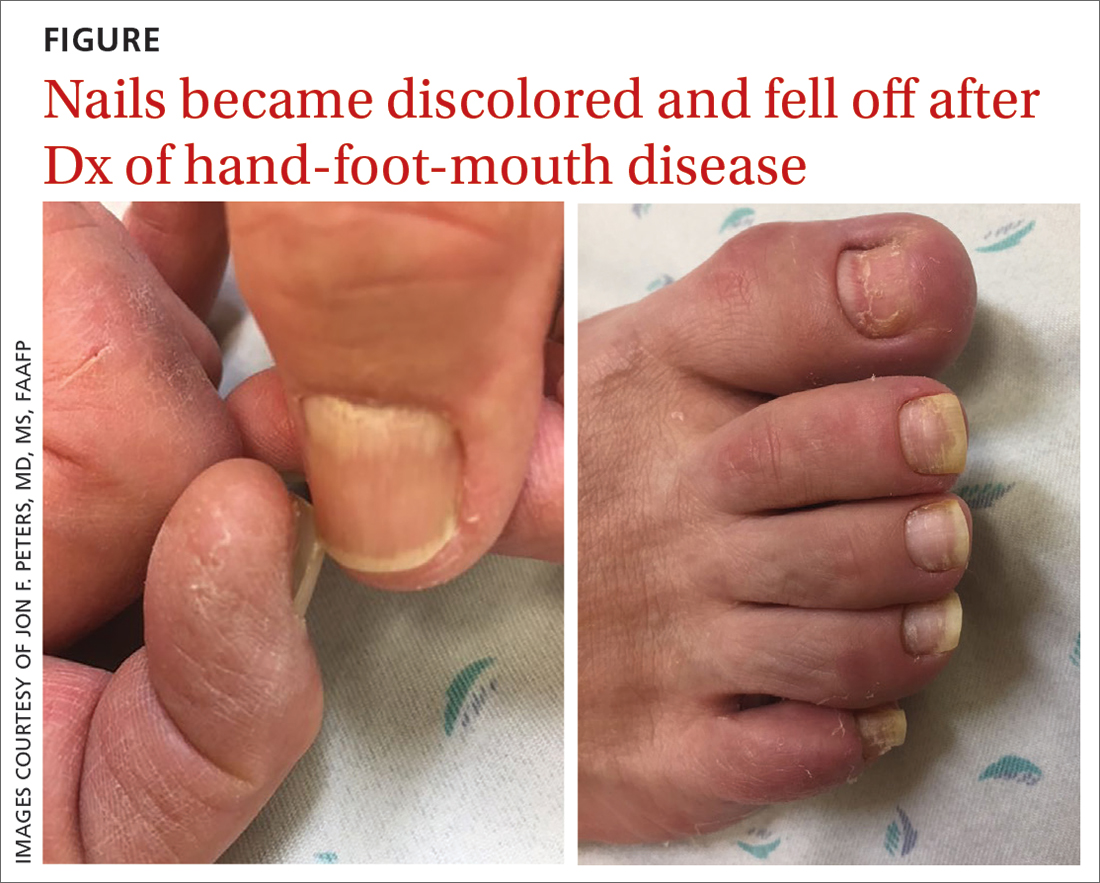
A 75-year-old man sought care from his primary care physician because his “fingernails and toenails [were] all falling off.” He did not feel ill and had no other complaints. His vital signs were unremarkable. He had no history of malignancies, chronic skin conditions, or systemic diseases. His fingernails and toenails were discolored and lifting from the proximal end of his nail beds (FIGURE). One of his great toenails had already fallen off, 1 thumb nail was minimally attached with the cuticle, and the rest of his nails were loose and in the process of separating from their nail beds. There was no nail pitting, rash, or joint swelling and tenderness.
The patient reported that while on vacation in Hawaii 3 weeks earlier, he had sought care at an urgent care clinic for a painless rash on his hands and the soles of his feet. At that time, he did not feel ill or have mouth ulcers, penile discharge, or arthralgia. There had been no recent changes to his prescription medications, which included finasteride, terazosin, omeprazole, and an albuterol inhaler. He denied taking over-the-counter medications or supplements.
The physical exam at the urgent care had revealed multiple blotchy, dark, 0.5- to 1-cm nonpruritic lesions that were desquamating. No oral lesions were seen. He had been given a diagnosis of hand-foot-mouth disease (HFMD) and reassured that it would resolve on its own in about 10 days.
THE DIAGNOSIS
Several possible diagnoses for nail disorders came to mind with this patient, including onychomycosis, onychoschizia, onycholysis, and onychomadesis.
Onychomycosis is a chronic fungal infection of the nail that affects toenails more often than fingernails.1 The most common form is distal subungual onychomycosis, which begins distally and slowly migrates proximally through the nail matrix.1 Often onychomycosis affects only a few nails unless the patient is elderly or has comorbid conditions, and the nails rarely separate from the nail bed.
Onychoschizia involves lamellar splitting and peeling of the dorsal surface of the nail plate.2 Usually white discolorations appear on the distal edges of the nail.3 It is more common in women than in men and is often caused by nail dehydration from repeated excessive immersion in water with detergents or recurrent application of nail polish.2 However, the nails do not separate from the nail bed, and usually only the fingernails are involved.
Onycholysis is a nail attachment disorder in which the nail plate distally separates from the nail bed. Areas of separation will appear white or yellow. There are many etiologies for onycholysis, including trauma, psoriasis, fungal infection, and contact irritant reactions.3 It also can be caused by medications and thyroid disease.3,4
Continue to: Onychomadesis
Onychomadesis, sometimes considered a severe form of Beau’s line,5,6 is defined by the spontaneous separation of the nail plate from the nail matrix. Although the nail will initially remain attached, proximal shedding will eventually occur.7 When several nails are involved, a systemic source—such as an acute infection, autoimmune disease, medication, malignancy (eg, cutaneous T-cell lymphoma), Kawasaki disease, skin disorders (eg, pemphigus vulgaris or keratosis punctata et planters), or chemotherapy—may be the cause.6-8 If only a few nails are involved, it may be associated with trauma, and in rare cases, onychomadesis can be idiopathic.5,7
In this case, all signs pointed to onychomadesis. All of the patient’s nails were affected (discolored and lifting), his nail loss involved spontaneous proximal separation of the nail plate from the nail matrix, and he had a recent previous infection: HFMD.
DISCUSSION
Onychomadesis is a rare nail-shedding disorder thought to be caused by the temporary arrest of the nail matrix.8 It is a potential late complication of infection, such as HFMD,9 and was first reported in children in Chicago in 2000.10 Since then, onychomadesis has been noted in children in many countries.8 Reports of onychomadesis following HFMD in adults are rare, but it may be underreported because HFMD is more common in children and symptoms are usually minor in adults.11
Molecular studies have associated onychomadesis with coxsackievirus (CV)A6 and CVA10.4 Other serotypes associated with onychomadesis include CVB1, CVB2, CVA5, CVA16, and enteroviruses 71 and 9.4 Most known outbreaks seem to be caused by CVA6.4
No treatment is needed for onychomadesis; physicians can reassure patients that normal nail growth will begin within 1 to 4 months. Because onychomadesis is rare, it does not have its own billing code, so one can use code L60.8 for “Other nail disorders.”12
Our patient was seen in the primary care clinic 3 months after his initial visit. At that time, his nails were no longer discolored and no other abnormalities were present. All of the nails on his fingers and toes were firmly attached and growing normally.
THE TAKEAWAY
The sudden asymptomatic loss of multiple fingernails and toenails—especially with proximal nail shedding—is a rare disorder known as onychomadesis. It can be caused by various etiologies and can be a late complication of HFMD or other viral infections. Onychomadesis should be considered when evaluating older patients, particularly when all of their nails are involved after a viral infection.
CORRESPONDENCE
Jon F. Peters, MD, MS, FAAFP, 14486 SE Lyon Court, Happy Valley, OR 97086; [email protected]
1. Rodgers P, Bassler M. Treating onychomycosis. Am Fam Physician. 2001;63:663-672, 677-678.
2. Sparavigna A, Tenconi B, La Penna L. Efficacy and tolerability of a biomineral formulation for treatment of onychoschizia: a randomized trial. Clin Cosmet Investig Dermatol. 2019:12:355-362. doi: 10.2147/CCID.S187305
3. Singal A, Arora R. Nail as a window of systemic diseases. Indian Dermatol Online J. 2015;6:67-74. doi: 10.4103/2229-5178.153002
4. Cleveland Clinic. Onycholysis. Accessed March 1, 2023. https://my.clevelandclinic.org/health/diseases/22903-onycholysis
5. Chiu H-H, Liu M-T, Chung W-H, et al. The mechanism of onychomadesis (nail shedding) and Beau’s lines following hand-foot-mouth disease. Viruses. 2019;11:522. doi: 10.3390/v11060522
6. Suchonwanit P, Nitayavardhana S. Idiopathic sporadic onychomadesis of toenails. Case Rep Dermatol Med. 2016;2016:6451327. doi: 10.1155/2016/6451327
7. Hardin J, Haber RM. Onychomadesis: literature review. Br J Dermatol. 2015;172:592-596. doi: 10.1111/bjd.13339
8. Li D, Yang W, Xing X, et al. Onychomadesis and potential association with HFMD outbreak in a kindergarten in Hubei providence, China, 2017. BMC Infect Dis. 2019:19:995. doi: 10.1186/s12879-019-4560-8
9. Chiu HH, Wu CS, Lan CE. Onychomadesis: a late complication of hand, foot, and mouth disease. J Emerg Med. 2017;52:243-245. doi: 10.1016/j.jemermed.2016.01.034
10. Clementz GC, Mancini AJ. Nail matrix arrest following hand-foot-mouth disease: a report of five children. Pediatr Dermatol. 2000;17:7-11. doi: 10.1046/j.1525-1470.2000.01702.x
11. Scarfi F, Arunachalam M, Galeone M, et al. An uncommon onychomadesis in adults. Int J Derm. 2014;53:1392-1394. doi: 10.1111/j.1365-4632.2012.05774.x
12. ICD10Data.com. 2023 ICD-10-CM codes. Accessed February 15, 2023. www.icd10data.com/ICD10CM/codes
THE CASE
A 75-year-old man sought care from his primary care physician because his “fingernails and toenails [were] all falling off.” He did not feel ill and had no other complaints. His vital signs were unremarkable. He had no history of malignancies, chronic skin conditions, or systemic diseases. His fingernails and toenails were discolored and lifting from the proximal end of his nail beds (FIGURE). One of his great toenails had already fallen off, 1 thumb nail was minimally attached with the cuticle, and the rest of his nails were loose and in the process of separating from their nail beds. There was no nail pitting, rash, or joint swelling and tenderness.

The patient reported that while on vacation in Hawaii 3 weeks earlier, he had sought care at an urgent care clinic for a painless rash on his hands and the soles of his feet. At that time, he did not feel ill or have mouth ulcers, penile discharge, or arthralgia. There had been no recent changes to his prescription medications, which included finasteride, terazosin, omeprazole, and an albuterol inhaler. He denied taking over-the-counter medications or supplements.
The physical exam at the urgent care had revealed multiple blotchy, dark, 0.5- to 1-cm nonpruritic lesions that were desquamating. No oral lesions were seen. He had been given a diagnosis of hand-foot-mouth disease (HFMD) and reassured that it would resolve on its own in about 10 days.
THE DIAGNOSIS
Several possible diagnoses for nail disorders came to mind with this patient, including onychomycosis, onychoschizia, onycholysis, and onychomadesis.
Onychomycosis is a chronic fungal infection of the nail that affects toenails more often than fingernails.1 The most common form is distal subungual onychomycosis, which begins distally and slowly migrates proximally through the nail matrix.1 Often onychomycosis affects only a few nails unless the patient is elderly or has comorbid conditions, and the nails rarely separate from the nail bed.
Onychoschizia involves lamellar splitting and peeling of the dorsal surface of the nail plate.2 Usually white discolorations appear on the distal edges of the nail.3 It is more common in women than in men and is often caused by nail dehydration from repeated excessive immersion in water with detergents or recurrent application of nail polish.2 However, the nails do not separate from the nail bed, and usually only the fingernails are involved.
Onycholysis is a nail attachment disorder in which the nail plate distally separates from the nail bed. Areas of separation will appear white or yellow. There are many etiologies for onycholysis, including trauma, psoriasis, fungal infection, and contact irritant reactions.3 It also can be caused by medications and thyroid disease.3,4
Continue to: Onychomadesis
Onychomadesis, sometimes considered a severe form of Beau’s line,5,6 is defined by the spontaneous separation of the nail plate from the nail matrix. Although the nail will initially remain attached, proximal shedding will eventually occur.7 When several nails are involved, a systemic source—such as an acute infection, autoimmune disease, medication, malignancy (eg, cutaneous T-cell lymphoma), Kawasaki disease, skin disorders (eg, pemphigus vulgaris or keratosis punctata et planters), or chemotherapy—may be the cause.6-8 If only a few nails are involved, it may be associated with trauma, and in rare cases, onychomadesis can be idiopathic.5,7
In this case, all signs pointed to onychomadesis. All of the patient’s nails were affected (discolored and lifting), his nail loss involved spontaneous proximal separation of the nail plate from the nail matrix, and he had a recent previous infection: HFMD.
DISCUSSION
Onychomadesis is a rare nail-shedding disorder thought to be caused by the temporary arrest of the nail matrix.8 It is a potential late complication of infection, such as HFMD,9 and was first reported in children in Chicago in 2000.10 Since then, onychomadesis has been noted in children in many countries.8 Reports of onychomadesis following HFMD in adults are rare, but it may be underreported because HFMD is more common in children and symptoms are usually minor in adults.11
Molecular studies have associated onychomadesis with coxsackievirus (CV)A6 and CVA10.4 Other serotypes associated with onychomadesis include CVB1, CVB2, CVA5, CVA16, and enteroviruses 71 and 9.4 Most known outbreaks seem to be caused by CVA6.4
No treatment is needed for onychomadesis; physicians can reassure patients that normal nail growth will begin within 1 to 4 months. Because onychomadesis is rare, it does not have its own billing code, so one can use code L60.8 for “Other nail disorders.”12
Our patient was seen in the primary care clinic 3 months after his initial visit. At that time, his nails were no longer discolored and no other abnormalities were present. All of the nails on his fingers and toes were firmly attached and growing normally.
THE TAKEAWAY
The sudden asymptomatic loss of multiple fingernails and toenails—especially with proximal nail shedding—is a rare disorder known as onychomadesis. It can be caused by various etiologies and can be a late complication of HFMD or other viral infections. Onychomadesis should be considered when evaluating older patients, particularly when all of their nails are involved after a viral infection.
CORRESPONDENCE
Jon F. Peters, MD, MS, FAAFP, 14486 SE Lyon Court, Happy Valley, OR 97086; [email protected]
THE CASE
A 75-year-old man sought care from his primary care physician because his “fingernails and toenails [were] all falling off.” He did not feel ill and had no other complaints. His vital signs were unremarkable. He had no history of malignancies, chronic skin conditions, or systemic diseases. His fingernails and toenails were discolored and lifting from the proximal end of his nail beds (FIGURE). One of his great toenails had already fallen off, 1 thumb nail was minimally attached with the cuticle, and the rest of his nails were loose and in the process of separating from their nail beds. There was no nail pitting, rash, or joint swelling and tenderness.

The patient reported that while on vacation in Hawaii 3 weeks earlier, he had sought care at an urgent care clinic for a painless rash on his hands and the soles of his feet. At that time, he did not feel ill or have mouth ulcers, penile discharge, or arthralgia. There had been no recent changes to his prescription medications, which included finasteride, terazosin, omeprazole, and an albuterol inhaler. He denied taking over-the-counter medications or supplements.
The physical exam at the urgent care had revealed multiple blotchy, dark, 0.5- to 1-cm nonpruritic lesions that were desquamating. No oral lesions were seen. He had been given a diagnosis of hand-foot-mouth disease (HFMD) and reassured that it would resolve on its own in about 10 days.
THE DIAGNOSIS
Several possible diagnoses for nail disorders came to mind with this patient, including onychomycosis, onychoschizia, onycholysis, and onychomadesis.
Onychomycosis is a chronic fungal infection of the nail that affects toenails more often than fingernails.1 The most common form is distal subungual onychomycosis, which begins distally and slowly migrates proximally through the nail matrix.1 Often onychomycosis affects only a few nails unless the patient is elderly or has comorbid conditions, and the nails rarely separate from the nail bed.
Onychoschizia involves lamellar splitting and peeling of the dorsal surface of the nail plate.2 Usually white discolorations appear on the distal edges of the nail.3 It is more common in women than in men and is often caused by nail dehydration from repeated excessive immersion in water with detergents or recurrent application of nail polish.2 However, the nails do not separate from the nail bed, and usually only the fingernails are involved.
Onycholysis is a nail attachment disorder in which the nail plate distally separates from the nail bed. Areas of separation will appear white or yellow. There are many etiologies for onycholysis, including trauma, psoriasis, fungal infection, and contact irritant reactions.3 It also can be caused by medications and thyroid disease.3,4
Continue to: Onychomadesis
Onychomadesis, sometimes considered a severe form of Beau’s line,5,6 is defined by the spontaneous separation of the nail plate from the nail matrix. Although the nail will initially remain attached, proximal shedding will eventually occur.7 When several nails are involved, a systemic source—such as an acute infection, autoimmune disease, medication, malignancy (eg, cutaneous T-cell lymphoma), Kawasaki disease, skin disorders (eg, pemphigus vulgaris or keratosis punctata et planters), or chemotherapy—may be the cause.6-8 If only a few nails are involved, it may be associated with trauma, and in rare cases, onychomadesis can be idiopathic.5,7
In this case, all signs pointed to onychomadesis. All of the patient’s nails were affected (discolored and lifting), his nail loss involved spontaneous proximal separation of the nail plate from the nail matrix, and he had a recent previous infection: HFMD.
DISCUSSION
Onychomadesis is a rare nail-shedding disorder thought to be caused by the temporary arrest of the nail matrix.8 It is a potential late complication of infection, such as HFMD,9 and was first reported in children in Chicago in 2000.10 Since then, onychomadesis has been noted in children in many countries.8 Reports of onychomadesis following HFMD in adults are rare, but it may be underreported because HFMD is more common in children and symptoms are usually minor in adults.11
Molecular studies have associated onychomadesis with coxsackievirus (CV)A6 and CVA10.4 Other serotypes associated with onychomadesis include CVB1, CVB2, CVA5, CVA16, and enteroviruses 71 and 9.4 Most known outbreaks seem to be caused by CVA6.4
No treatment is needed for onychomadesis; physicians can reassure patients that normal nail growth will begin within 1 to 4 months. Because onychomadesis is rare, it does not have its own billing code, so one can use code L60.8 for “Other nail disorders.”12
Our patient was seen in the primary care clinic 3 months after his initial visit. At that time, his nails were no longer discolored and no other abnormalities were present. All of the nails on his fingers and toes were firmly attached and growing normally.
THE TAKEAWAY
The sudden asymptomatic loss of multiple fingernails and toenails—especially with proximal nail shedding—is a rare disorder known as onychomadesis. It can be caused by various etiologies and can be a late complication of HFMD or other viral infections. Onychomadesis should be considered when evaluating older patients, particularly when all of their nails are involved after a viral infection.
CORRESPONDENCE
Jon F. Peters, MD, MS, FAAFP, 14486 SE Lyon Court, Happy Valley, OR 97086; [email protected]
1. Rodgers P, Bassler M. Treating onychomycosis. Am Fam Physician. 2001;63:663-672, 677-678.
2. Sparavigna A, Tenconi B, La Penna L. Efficacy and tolerability of a biomineral formulation for treatment of onychoschizia: a randomized trial. Clin Cosmet Investig Dermatol. 2019:12:355-362. doi: 10.2147/CCID.S187305
3. Singal A, Arora R. Nail as a window of systemic diseases. Indian Dermatol Online J. 2015;6:67-74. doi: 10.4103/2229-5178.153002
4. Cleveland Clinic. Onycholysis. Accessed March 1, 2023. https://my.clevelandclinic.org/health/diseases/22903-onycholysis
5. Chiu H-H, Liu M-T, Chung W-H, et al. The mechanism of onychomadesis (nail shedding) and Beau’s lines following hand-foot-mouth disease. Viruses. 2019;11:522. doi: 10.3390/v11060522
6. Suchonwanit P, Nitayavardhana S. Idiopathic sporadic onychomadesis of toenails. Case Rep Dermatol Med. 2016;2016:6451327. doi: 10.1155/2016/6451327
7. Hardin J, Haber RM. Onychomadesis: literature review. Br J Dermatol. 2015;172:592-596. doi: 10.1111/bjd.13339
8. Li D, Yang W, Xing X, et al. Onychomadesis and potential association with HFMD outbreak in a kindergarten in Hubei providence, China, 2017. BMC Infect Dis. 2019:19:995. doi: 10.1186/s12879-019-4560-8
9. Chiu HH, Wu CS, Lan CE. Onychomadesis: a late complication of hand, foot, and mouth disease. J Emerg Med. 2017;52:243-245. doi: 10.1016/j.jemermed.2016.01.034
10. Clementz GC, Mancini AJ. Nail matrix arrest following hand-foot-mouth disease: a report of five children. Pediatr Dermatol. 2000;17:7-11. doi: 10.1046/j.1525-1470.2000.01702.x
11. Scarfi F, Arunachalam M, Galeone M, et al. An uncommon onychomadesis in adults. Int J Derm. 2014;53:1392-1394. doi: 10.1111/j.1365-4632.2012.05774.x
12. ICD10Data.com. 2023 ICD-10-CM codes. Accessed February 15, 2023. www.icd10data.com/ICD10CM/codes
1. Rodgers P, Bassler M. Treating onychomycosis. Am Fam Physician. 2001;63:663-672, 677-678.
2. Sparavigna A, Tenconi B, La Penna L. Efficacy and tolerability of a biomineral formulation for treatment of onychoschizia: a randomized trial. Clin Cosmet Investig Dermatol. 2019:12:355-362. doi: 10.2147/CCID.S187305
3. Singal A, Arora R. Nail as a window of systemic diseases. Indian Dermatol Online J. 2015;6:67-74. doi: 10.4103/2229-5178.153002
4. Cleveland Clinic. Onycholysis. Accessed March 1, 2023. https://my.clevelandclinic.org/health/diseases/22903-onycholysis
5. Chiu H-H, Liu M-T, Chung W-H, et al. The mechanism of onychomadesis (nail shedding) and Beau’s lines following hand-foot-mouth disease. Viruses. 2019;11:522. doi: 10.3390/v11060522
6. Suchonwanit P, Nitayavardhana S. Idiopathic sporadic onychomadesis of toenails. Case Rep Dermatol Med. 2016;2016:6451327. doi: 10.1155/2016/6451327
7. Hardin J, Haber RM. Onychomadesis: literature review. Br J Dermatol. 2015;172:592-596. doi: 10.1111/bjd.13339
8. Li D, Yang W, Xing X, et al. Onychomadesis and potential association with HFMD outbreak in a kindergarten in Hubei providence, China, 2017. BMC Infect Dis. 2019:19:995. doi: 10.1186/s12879-019-4560-8
9. Chiu HH, Wu CS, Lan CE. Onychomadesis: a late complication of hand, foot, and mouth disease. J Emerg Med. 2017;52:243-245. doi: 10.1016/j.jemermed.2016.01.034
10. Clementz GC, Mancini AJ. Nail matrix arrest following hand-foot-mouth disease: a report of five children. Pediatr Dermatol. 2000;17:7-11. doi: 10.1046/j.1525-1470.2000.01702.x
11. Scarfi F, Arunachalam M, Galeone M, et al. An uncommon onychomadesis in adults. Int J Derm. 2014;53:1392-1394. doi: 10.1111/j.1365-4632.2012.05774.x
12. ICD10Data.com. 2023 ICD-10-CM codes. Accessed February 15, 2023. www.icd10data.com/ICD10CM/codes
► Recent history of hand-foot-mouth disease
► Discolored fingernails and toenails lifting from the proximal end
The earlier baricitinib for severe alopecia areata is started, the better
NEW ORLEANS – In the nearly 1 year .
“The journey to JAK inhibition in alopecia areata has been incredible,” Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, said at the annual meeting of the American Academy of Dermatology. “JAK inhibitors are here to stay, and I think baricitinib offers an amazing opportunity for the right patients.”
The efficacy and safety of baricitinib (Olumiant) for AA was studied in two randomized, double-blind, placebo-controlled trials (BRAVE-AA1 and BRAVE-AA2) with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool (SALT) for more than 6 months. Patients in these trials received either a placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved a SALT score of 20 or less, or at least 80% scalp hair coverage at week 36. The researchers found that 36%-39% of individuals in the 4-mg arm achieved a SALT score of less than 20, compared with 19%-23% of individuals in the 2 mg arm. Similar outcomes were observed for eyebrow and eyelash hair loss.
Most adverse events observed in BRAVE-AA1 and BRAVE-AA2 were in the mild to moderate range, and the actual number of adverse events leading to permanent discontinuation was extremely low. The most common adverse events were upper respiratory tract infections, headache, nasopharyngitis, acne, urinary tract infections, and an increase in blood creatine kinase.
Baricitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other potent immunosuppressants, Dr. Chovatiya said. Required lab evaluations include baseline testing for tuberculosis and viral hepatitis; CBC, hepatic function, and renal function at baseline and then as clinically indicated; and lipids after 12 weeks of therapy, then as clinically indicated. The recommended starting dose of baricitinib is 2 mg per day, which can be increased to 4 mg per day if the response is not adequate. “However, for patients with nearly complete or complete scalp hair loss, with or without substantial eyelash or eyebrow hair loss, 4 mg once daily is recommended,” he said. “Once an adequate response is achieved, it’s recommended to reduce from 4 to 2 mg daily.”
52-week, 76-week data
According to pooled data from BRAVE-AA1 and BRAVE-AA2 published online March 1, 2023, efficacy continues to increase out to 52 weeks. Specifically, by week 52, 39% of individuals in the 4 mg arm achieved a SALT score of 20 or less, compared with 22.6% of individuals in the 2 mg arm. “You see similar linear growth in the eyebrow and eyelash response loss as well,” Dr. Chovatiya said.
In other findings, patients in the 4 mg treatment arm who achieved a SALT score of 20 or less at week 52 were eligible for randomized down titration, provided that they had stayed on the same dose of baricitinib from initial randomization. According to data from baricitinib manufacturer Eli Lilly, 77.5% of patients who stepped down to the 2 mg dose from the 4 mg dose at week 52 achieved a SALT score of 20 or less at week 76, Dr. Chovatiya said. “If I can keep someone on 4 mg that’s great, but it looks like you can go to a lower dose and do a pretty good job,” he said.
Patients in the baricitinib arms who achieved a SALT score of 20 or less at week 52 were eligible for randomized withdrawal, provided that they had stayed on the same dose of the drug from initial randomization. According to Dr. Chovatiya, 89.4% of individuals who remained on the 4 mg dose to week 76 maintained a SALT score of 20 or less, compared with 33.3% of those who switched from the 4 mg to placebo. “The takeaway here is that clinically, longitudinal treatment looks to be required in this time period” for continued efficacy, he said. “However, what this looks like in the real world remains to be seen.”
A recently published integrated analysis of safety data from BRAVE-AA1 and BRAVE-AA2 reported that no deaths occurred and of the few reported serious infections, nearly half were COVID-19. There was a single case of multidermatomal herpes zoster and no cases of tuberculosis. One patient with risk factors for MI had an MI during a placebo-controlled period, and one study participant with a history of COVID-19 infection developed a pulmonary embolism at day 638. There was one case each of chronic lymphocytic leukemia, B-cell lymphoma, breast cancer, and appendicitis.
Baseline severity and treatment response
“Does treatment response vary with baseline disease status?” Dr. Chovatiya asked. “Yes. People with very severe hair loss [defined as a SALT score of 95 or higher] tended to do worse, while the rest of the study population did even better – an almost twofold difference. This means that you want to treat as early as you possibly can. It’s interesting to note that you don’t see this difference as much in the case of eyebrows and eyelashes. This makes sense, though. Eyebrows and eyelashes probably behave differently in terms of growth than the scalp does.”
Certain baseline characteristics of patients in BRAVE-AA1 and BRAVE-AA2 portended better outcomes. Women tended to fare better than men, but individuals who had longer histories of AA did not respond well. “People who had a shorter duration of their current episode of AA also did better than people who had a longer current episode, so we want to think about treating as soon as we possibly can,” Dr. Chovatiya said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including Eli Lilly.
NEW ORLEANS – In the nearly 1 year .
“The journey to JAK inhibition in alopecia areata has been incredible,” Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, said at the annual meeting of the American Academy of Dermatology. “JAK inhibitors are here to stay, and I think baricitinib offers an amazing opportunity for the right patients.”
The efficacy and safety of baricitinib (Olumiant) for AA was studied in two randomized, double-blind, placebo-controlled trials (BRAVE-AA1 and BRAVE-AA2) with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool (SALT) for more than 6 months. Patients in these trials received either a placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved a SALT score of 20 or less, or at least 80% scalp hair coverage at week 36. The researchers found that 36%-39% of individuals in the 4-mg arm achieved a SALT score of less than 20, compared with 19%-23% of individuals in the 2 mg arm. Similar outcomes were observed for eyebrow and eyelash hair loss.
Most adverse events observed in BRAVE-AA1 and BRAVE-AA2 were in the mild to moderate range, and the actual number of adverse events leading to permanent discontinuation was extremely low. The most common adverse events were upper respiratory tract infections, headache, nasopharyngitis, acne, urinary tract infections, and an increase in blood creatine kinase.
Baricitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other potent immunosuppressants, Dr. Chovatiya said. Required lab evaluations include baseline testing for tuberculosis and viral hepatitis; CBC, hepatic function, and renal function at baseline and then as clinically indicated; and lipids after 12 weeks of therapy, then as clinically indicated. The recommended starting dose of baricitinib is 2 mg per day, which can be increased to 4 mg per day if the response is not adequate. “However, for patients with nearly complete or complete scalp hair loss, with or without substantial eyelash or eyebrow hair loss, 4 mg once daily is recommended,” he said. “Once an adequate response is achieved, it’s recommended to reduce from 4 to 2 mg daily.”
52-week, 76-week data
According to pooled data from BRAVE-AA1 and BRAVE-AA2 published online March 1, 2023, efficacy continues to increase out to 52 weeks. Specifically, by week 52, 39% of individuals in the 4 mg arm achieved a SALT score of 20 or less, compared with 22.6% of individuals in the 2 mg arm. “You see similar linear growth in the eyebrow and eyelash response loss as well,” Dr. Chovatiya said.
In other findings, patients in the 4 mg treatment arm who achieved a SALT score of 20 or less at week 52 were eligible for randomized down titration, provided that they had stayed on the same dose of baricitinib from initial randomization. According to data from baricitinib manufacturer Eli Lilly, 77.5% of patients who stepped down to the 2 mg dose from the 4 mg dose at week 52 achieved a SALT score of 20 or less at week 76, Dr. Chovatiya said. “If I can keep someone on 4 mg that’s great, but it looks like you can go to a lower dose and do a pretty good job,” he said.
Patients in the baricitinib arms who achieved a SALT score of 20 or less at week 52 were eligible for randomized withdrawal, provided that they had stayed on the same dose of the drug from initial randomization. According to Dr. Chovatiya, 89.4% of individuals who remained on the 4 mg dose to week 76 maintained a SALT score of 20 or less, compared with 33.3% of those who switched from the 4 mg to placebo. “The takeaway here is that clinically, longitudinal treatment looks to be required in this time period” for continued efficacy, he said. “However, what this looks like in the real world remains to be seen.”
A recently published integrated analysis of safety data from BRAVE-AA1 and BRAVE-AA2 reported that no deaths occurred and of the few reported serious infections, nearly half were COVID-19. There was a single case of multidermatomal herpes zoster and no cases of tuberculosis. One patient with risk factors for MI had an MI during a placebo-controlled period, and one study participant with a history of COVID-19 infection developed a pulmonary embolism at day 638. There was one case each of chronic lymphocytic leukemia, B-cell lymphoma, breast cancer, and appendicitis.
Baseline severity and treatment response
“Does treatment response vary with baseline disease status?” Dr. Chovatiya asked. “Yes. People with very severe hair loss [defined as a SALT score of 95 or higher] tended to do worse, while the rest of the study population did even better – an almost twofold difference. This means that you want to treat as early as you possibly can. It’s interesting to note that you don’t see this difference as much in the case of eyebrows and eyelashes. This makes sense, though. Eyebrows and eyelashes probably behave differently in terms of growth than the scalp does.”
Certain baseline characteristics of patients in BRAVE-AA1 and BRAVE-AA2 portended better outcomes. Women tended to fare better than men, but individuals who had longer histories of AA did not respond well. “People who had a shorter duration of their current episode of AA also did better than people who had a longer current episode, so we want to think about treating as soon as we possibly can,” Dr. Chovatiya said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including Eli Lilly.
NEW ORLEANS – In the nearly 1 year .
“The journey to JAK inhibition in alopecia areata has been incredible,” Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, said at the annual meeting of the American Academy of Dermatology. “JAK inhibitors are here to stay, and I think baricitinib offers an amazing opportunity for the right patients.”
The efficacy and safety of baricitinib (Olumiant) for AA was studied in two randomized, double-blind, placebo-controlled trials (BRAVE-AA1 and BRAVE-AA2) with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool (SALT) for more than 6 months. Patients in these trials received either a placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved a SALT score of 20 or less, or at least 80% scalp hair coverage at week 36. The researchers found that 36%-39% of individuals in the 4-mg arm achieved a SALT score of less than 20, compared with 19%-23% of individuals in the 2 mg arm. Similar outcomes were observed for eyebrow and eyelash hair loss.
Most adverse events observed in BRAVE-AA1 and BRAVE-AA2 were in the mild to moderate range, and the actual number of adverse events leading to permanent discontinuation was extremely low. The most common adverse events were upper respiratory tract infections, headache, nasopharyngitis, acne, urinary tract infections, and an increase in blood creatine kinase.
Baricitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other potent immunosuppressants, Dr. Chovatiya said. Required lab evaluations include baseline testing for tuberculosis and viral hepatitis; CBC, hepatic function, and renal function at baseline and then as clinically indicated; and lipids after 12 weeks of therapy, then as clinically indicated. The recommended starting dose of baricitinib is 2 mg per day, which can be increased to 4 mg per day if the response is not adequate. “However, for patients with nearly complete or complete scalp hair loss, with or without substantial eyelash or eyebrow hair loss, 4 mg once daily is recommended,” he said. “Once an adequate response is achieved, it’s recommended to reduce from 4 to 2 mg daily.”
52-week, 76-week data
According to pooled data from BRAVE-AA1 and BRAVE-AA2 published online March 1, 2023, efficacy continues to increase out to 52 weeks. Specifically, by week 52, 39% of individuals in the 4 mg arm achieved a SALT score of 20 or less, compared with 22.6% of individuals in the 2 mg arm. “You see similar linear growth in the eyebrow and eyelash response loss as well,” Dr. Chovatiya said.
In other findings, patients in the 4 mg treatment arm who achieved a SALT score of 20 or less at week 52 were eligible for randomized down titration, provided that they had stayed on the same dose of baricitinib from initial randomization. According to data from baricitinib manufacturer Eli Lilly, 77.5% of patients who stepped down to the 2 mg dose from the 4 mg dose at week 52 achieved a SALT score of 20 or less at week 76, Dr. Chovatiya said. “If I can keep someone on 4 mg that’s great, but it looks like you can go to a lower dose and do a pretty good job,” he said.
Patients in the baricitinib arms who achieved a SALT score of 20 or less at week 52 were eligible for randomized withdrawal, provided that they had stayed on the same dose of the drug from initial randomization. According to Dr. Chovatiya, 89.4% of individuals who remained on the 4 mg dose to week 76 maintained a SALT score of 20 or less, compared with 33.3% of those who switched from the 4 mg to placebo. “The takeaway here is that clinically, longitudinal treatment looks to be required in this time period” for continued efficacy, he said. “However, what this looks like in the real world remains to be seen.”
A recently published integrated analysis of safety data from BRAVE-AA1 and BRAVE-AA2 reported that no deaths occurred and of the few reported serious infections, nearly half were COVID-19. There was a single case of multidermatomal herpes zoster and no cases of tuberculosis. One patient with risk factors for MI had an MI during a placebo-controlled period, and one study participant with a history of COVID-19 infection developed a pulmonary embolism at day 638. There was one case each of chronic lymphocytic leukemia, B-cell lymphoma, breast cancer, and appendicitis.
Baseline severity and treatment response
“Does treatment response vary with baseline disease status?” Dr. Chovatiya asked. “Yes. People with very severe hair loss [defined as a SALT score of 95 or higher] tended to do worse, while the rest of the study population did even better – an almost twofold difference. This means that you want to treat as early as you possibly can. It’s interesting to note that you don’t see this difference as much in the case of eyebrows and eyelashes. This makes sense, though. Eyebrows and eyelashes probably behave differently in terms of growth than the scalp does.”
Certain baseline characteristics of patients in BRAVE-AA1 and BRAVE-AA2 portended better outcomes. Women tended to fare better than men, but individuals who had longer histories of AA did not respond well. “People who had a shorter duration of their current episode of AA also did better than people who had a longer current episode, so we want to think about treating as soon as we possibly can,” Dr. Chovatiya said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including Eli Lilly.
AT AAD 2023
Melasma
THE COMPARISON
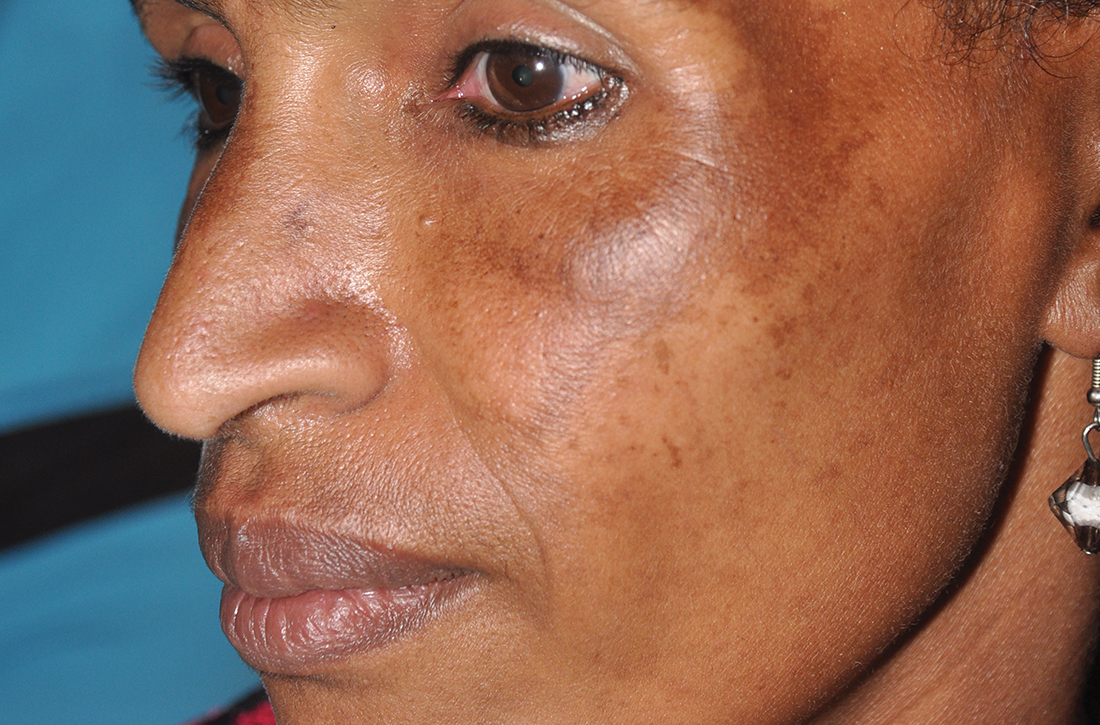
A Melasma on the face of a Hispanic woman, with hyperpigmentation on the cheeks, bridge of the nose, and upper lip.
B Melasma on the face of a Malaysian woman, with hyperpigmentation on the upper cheeks and bridge of the nose.
C Melasma on the face of an African woman, with hyperpigmentation on the upper cheeks and lateral to the eyes.
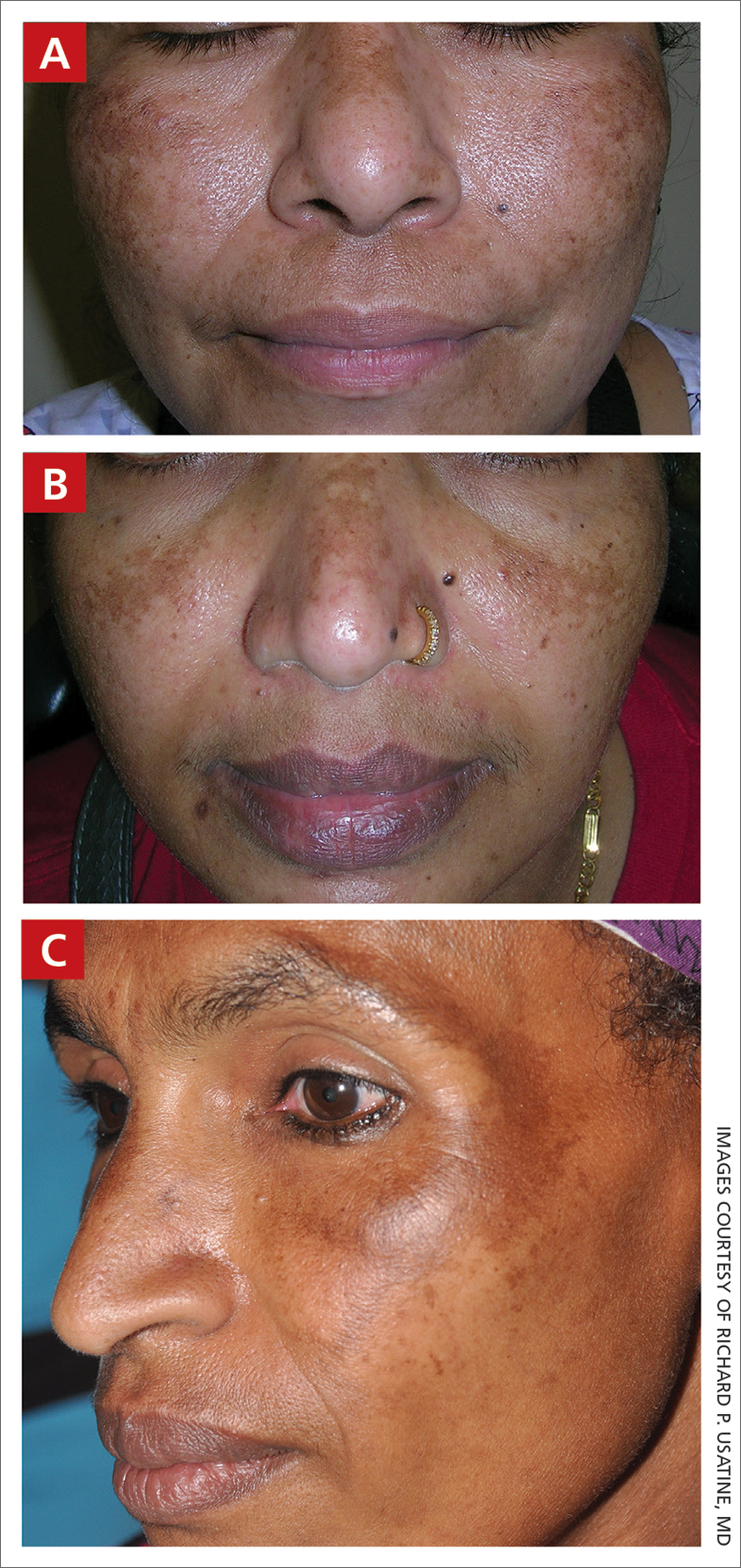
Melasma (also known as chloasma) is a pigmentary disorder that causes chronic symmetric hyperpigmentation on the face. In patients with darker skin tones, centrofacial areas are affected.1 Increased deposition of melanin distributed in the dermis leads to dermal melanosis. Newer research suggests that mast cell and keratinocyte interactions, altered gene regulation, neovascularization, and disruptions in the basement membrane cause melasma.2 Patients present with epidermal or dermal melasma or a combination of both (mixed melasma).3 Wood lamp examination is helpful to distinguish between epidermal and dermal melasma. Dermal and mixed melasma can be difficult to treat and require multimodal treatments.
Epidemiology
Melasma commonly affects women ages 20 to 40 years,4 with a female to male ratio of 9:1.5 Potential triggers of melasma include hormones (eg, pregnancy, oral contraceptives, hormone replacement therapy) and exposure to UV light.2,5 Melasma occurs in patients of all racial and ethnic backgrounds; however, the prevalence is higher in patients with darker skin tones.2
Key clinical features in people with darker skin tones
Melasma commonly manifests as symmetrically distributed, reticulated (lacy), dark brown to grayish brown patches on the cheeks, nose, forehead, upper lip, and chin in patients with darker skin tones.5 The pigment can be tan brown in patients with lighter skin tones. Given that postinflammatory hyperpigmentation and other pigmentary disorders can cause a similar appearance, a biopsy sometimes is needed to confirm the diagnosis, but melasma is diagnosed via physical examination in most patients. Melasma can be misdiagnosed as postinflammatory hyperpigmentation, solar lentigines, exogenous ochronosis, and Hori nevus.5
Worth noting
Prevention
- Daily sunscreen use is critical to prevent worsening of melasma. Sunscreen may not appear cosmetically elegant on darker skin tones, which creates a barrier to its use.6 Protection from both sunlight and visible light is necessary. Visible light, including light from light bulbs and device-emitted blue light, can worsen melasma. Iron oxides in tinted sunscreen offer protection from visible light.
- Physicians can recommend sunscreens that are more transparent or tinted for a better cosmetic match.
- Severe flares of melasma can occur with sun exposure despite good control with medications and laser modalities.
Treatment
- First-line therapies include topical hydroquinone 2% to 4%, tretinoin, azelaic acid, kojic acid, or ascorbic acid (vitamin C). A popular topical compound is a steroid, tretinoin, and hydroquinone.1,5 Over-the-counter hydroquinone has been removed from the market due to safety concerns; however, it is still first line in the treatment of melasma. If hydroquinone is prescribed, treatment intervals of 6 to 8 weeks followed by a hydroquinone-free period is advised to reduce the risk for exogenous ochronosis (a paradoxical darkening of the skin).
- Chemical peels are second-line treatments that are effective for melasma. Improvement in epidermal melasma has been shown with chemical peels containing Jessner solution, salicylic acid, or a-hydroxy acid. Patients with dermal and mixed melasma have seen improvement with trichloroacetic acid 25% to 35% with or without Jessner solution.1
- Cysteamine is a topical treatment created from the degradation of coenzyme A. It disrupts the synthesis of melanin to create a more even skin tone. It may be recommended in combination with sunscreen as a first-line or secondline topical therapy.
- Oral tranexamic acid is a third-line treatment that is an analogue for lysine. It decreases prostaglandin production, which leads to a lower number of tyrosine precursors available for the creation of melanin. Tranexamic acid has been shown to lighten the appearance of melasma.7 The most common and dangerous adverse effect of tranexamic acid is blood clots, and this treatment should be avoided in those on combination (estrogen and progestin) contraceptives or those with a personal or family history of clotting disorders.8
- Fourth-line treatments such as lasers (performed by dermatologists) can destroy the deposition of pigment while avoiding destruction of epidermal keratinocytes.1,9,10 They also are commonly employed in refractive melasma. The most common lasers are nonablative fractionated lasers and low-fluence Q-switched lasers. The Q-switched Nd:YAG and picosecond lasers are safe for treating melasma in darker skin tones. Ablative fractionated lasers such as CO2 lasers and erbium:YAG lasers also have been used in the treatment of melasma; however, there is still an extremely high risk for postinflammatory dyspigmentation 1 to 2 months after the procedure.10
- Although there is still a risk for rebound hyperpigmentation after laser treatment, use of topical hydroquinone pretreatment may help decrease postoperative hyperpigmentation.1,5 Patients who are treated with the incorrect laser or overtreated may develop postinflammatory hyperpigmentation, rebound hyperpigmentation, or hypopigmentation.
Health disparity highlight
Melasma, most common in patients with skin of color, is a common chronic pigmentation disorder that is cosmetically and psychologically burdensome,11 leading to decreased quality of life, emotional functioning, and self-esteem.12 Clinicians should counsel patients and work closely on long-term management. The treatment options for melasma are considered cosmetic and may be cost prohibitive for many to cover out of pocket. Topical treatments have been found to be the most cost-effective.13 Some compounding pharmacies and drug discount programs provide more affordable treatment pricing; however, some patients are still unable to afford these options.
1. Cunha PR, Kroumpouzos G. Melasma and vitiligo: novel and experimental therapies. J Clin Exp Derm Res. 2016;7:2. doi:10.4172/2155-9554.1000e106
2. Rajanala S, Maymone MBC, Vashi NA. Melasma pathogenesis: a review of the latest research, pathological findings, and investigational therapies. Dermatol Online J. 2019;25:13030/qt47b7r28c.
3. Grimes PE, Yamada N, Bhawan J. Light microscopic, immunohistochemical, and ultrastructural alterations in patients with melasma. Am J Dermatopathol. 2005;27:96-101.
4. Achar A, Rathi SK. Melasma: a clinico-epidemiological study of 312 cases. Indian J Dermatol. 2011;56:380-382.
5. Ogbechie-Godec OA, Elbuluk N. Melasma: an up-to-date comprehensive review. Dermatol Ther. 2017;7:305-318.
6. Morquette AJ, Waples ER, Heath CR. The importance of cosmetically elegant sunscreen in skin of color populations. J Cosmet Dermatol. 2022;21:1337-1338.
7. Taraz M, Nikham S, Ehsani AH. Tranexamic acid in treatment of melasma: a comprehensive review of clinical studies. Dermatol Ther. 2017;30(3). doi:10.1111/dth.12465
8. Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
9. Castanedo-Cazares JP, Hernandez-Blanco D, Carlos-Ortega B, et al. Near-visible light and UV photoprotection in the treatment of melasma: a double-blind randomized trial. Photodermatol Photoimmunol Photomed. 2014;30:35-42.
10. Trivedi MK, Yang FC, Cho BK. A review of laser and light therapy in melasma. Int J Womens Dermatol. 2017;3:11-20.
11. Dodmani PN, Deshmukh AR. Assessment of quality of life of melasma patients as per melasma quality of life scale (MELASQoL). Pigment Int. 2020;7:75-79.
12. Balkrishnan R, McMichael A, Camacho FT, et al. Development and validation of a health‐related quality of life instrument for women with melasma. Br J Dermatol. 2003;149:572-577.
13. Alikhan A, Daly M, Wu J, et al. Cost-effectiveness of a hydroquinone/tretinoin/fluocinolone acetonide cream combination in treating melasma in the United States. J Dermatolog Treat. 2010;21:276-281.
THE COMPARISON
A Melasma on the face of a Hispanic woman, with hyperpigmentation on the cheeks, bridge of the nose, and upper lip.
B Melasma on the face of a Malaysian woman, with hyperpigmentation on the upper cheeks and bridge of the nose.
C Melasma on the face of an African woman, with hyperpigmentation on the upper cheeks and lateral to the eyes.

Melasma (also known as chloasma) is a pigmentary disorder that causes chronic symmetric hyperpigmentation on the face. In patients with darker skin tones, centrofacial areas are affected.1 Increased deposition of melanin distributed in the dermis leads to dermal melanosis. Newer research suggests that mast cell and keratinocyte interactions, altered gene regulation, neovascularization, and disruptions in the basement membrane cause melasma.2 Patients present with epidermal or dermal melasma or a combination of both (mixed melasma).3 Wood lamp examination is helpful to distinguish between epidermal and dermal melasma. Dermal and mixed melasma can be difficult to treat and require multimodal treatments.
Epidemiology
Melasma commonly affects women ages 20 to 40 years,4 with a female to male ratio of 9:1.5 Potential triggers of melasma include hormones (eg, pregnancy, oral contraceptives, hormone replacement therapy) and exposure to UV light.2,5 Melasma occurs in patients of all racial and ethnic backgrounds; however, the prevalence is higher in patients with darker skin tones.2
Key clinical features in people with darker skin tones
Melasma commonly manifests as symmetrically distributed, reticulated (lacy), dark brown to grayish brown patches on the cheeks, nose, forehead, upper lip, and chin in patients with darker skin tones.5 The pigment can be tan brown in patients with lighter skin tones. Given that postinflammatory hyperpigmentation and other pigmentary disorders can cause a similar appearance, a biopsy sometimes is needed to confirm the diagnosis, but melasma is diagnosed via physical examination in most patients. Melasma can be misdiagnosed as postinflammatory hyperpigmentation, solar lentigines, exogenous ochronosis, and Hori nevus.5
Worth noting
Prevention
- Daily sunscreen use is critical to prevent worsening of melasma. Sunscreen may not appear cosmetically elegant on darker skin tones, which creates a barrier to its use.6 Protection from both sunlight and visible light is necessary. Visible light, including light from light bulbs and device-emitted blue light, can worsen melasma. Iron oxides in tinted sunscreen offer protection from visible light.
- Physicians can recommend sunscreens that are more transparent or tinted for a better cosmetic match.
- Severe flares of melasma can occur with sun exposure despite good control with medications and laser modalities.
Treatment
- First-line therapies include topical hydroquinone 2% to 4%, tretinoin, azelaic acid, kojic acid, or ascorbic acid (vitamin C). A popular topical compound is a steroid, tretinoin, and hydroquinone.1,5 Over-the-counter hydroquinone has been removed from the market due to safety concerns; however, it is still first line in the treatment of melasma. If hydroquinone is prescribed, treatment intervals of 6 to 8 weeks followed by a hydroquinone-free period is advised to reduce the risk for exogenous ochronosis (a paradoxical darkening of the skin).
- Chemical peels are second-line treatments that are effective for melasma. Improvement in epidermal melasma has been shown with chemical peels containing Jessner solution, salicylic acid, or a-hydroxy acid. Patients with dermal and mixed melasma have seen improvement with trichloroacetic acid 25% to 35% with or without Jessner solution.1
- Cysteamine is a topical treatment created from the degradation of coenzyme A. It disrupts the synthesis of melanin to create a more even skin tone. It may be recommended in combination with sunscreen as a first-line or secondline topical therapy.
- Oral tranexamic acid is a third-line treatment that is an analogue for lysine. It decreases prostaglandin production, which leads to a lower number of tyrosine precursors available for the creation of melanin. Tranexamic acid has been shown to lighten the appearance of melasma.7 The most common and dangerous adverse effect of tranexamic acid is blood clots, and this treatment should be avoided in those on combination (estrogen and progestin) contraceptives or those with a personal or family history of clotting disorders.8
- Fourth-line treatments such as lasers (performed by dermatologists) can destroy the deposition of pigment while avoiding destruction of epidermal keratinocytes.1,9,10 They also are commonly employed in refractive melasma. The most common lasers are nonablative fractionated lasers and low-fluence Q-switched lasers. The Q-switched Nd:YAG and picosecond lasers are safe for treating melasma in darker skin tones. Ablative fractionated lasers such as CO2 lasers and erbium:YAG lasers also have been used in the treatment of melasma; however, there is still an extremely high risk for postinflammatory dyspigmentation 1 to 2 months after the procedure.10
- Although there is still a risk for rebound hyperpigmentation after laser treatment, use of topical hydroquinone pretreatment may help decrease postoperative hyperpigmentation.1,5 Patients who are treated with the incorrect laser or overtreated may develop postinflammatory hyperpigmentation, rebound hyperpigmentation, or hypopigmentation.
Health disparity highlight
Melasma, most common in patients with skin of color, is a common chronic pigmentation disorder that is cosmetically and psychologically burdensome,11 leading to decreased quality of life, emotional functioning, and self-esteem.12 Clinicians should counsel patients and work closely on long-term management. The treatment options for melasma are considered cosmetic and may be cost prohibitive for many to cover out of pocket. Topical treatments have been found to be the most cost-effective.13 Some compounding pharmacies and drug discount programs provide more affordable treatment pricing; however, some patients are still unable to afford these options.
THE COMPARISON
A Melasma on the face of a Hispanic woman, with hyperpigmentation on the cheeks, bridge of the nose, and upper lip.
B Melasma on the face of a Malaysian woman, with hyperpigmentation on the upper cheeks and bridge of the nose.
C Melasma on the face of an African woman, with hyperpigmentation on the upper cheeks and lateral to the eyes.

Melasma (also known as chloasma) is a pigmentary disorder that causes chronic symmetric hyperpigmentation on the face. In patients with darker skin tones, centrofacial areas are affected.1 Increased deposition of melanin distributed in the dermis leads to dermal melanosis. Newer research suggests that mast cell and keratinocyte interactions, altered gene regulation, neovascularization, and disruptions in the basement membrane cause melasma.2 Patients present with epidermal or dermal melasma or a combination of both (mixed melasma).3 Wood lamp examination is helpful to distinguish between epidermal and dermal melasma. Dermal and mixed melasma can be difficult to treat and require multimodal treatments.
Epidemiology
Melasma commonly affects women ages 20 to 40 years,4 with a female to male ratio of 9:1.5 Potential triggers of melasma include hormones (eg, pregnancy, oral contraceptives, hormone replacement therapy) and exposure to UV light.2,5 Melasma occurs in patients of all racial and ethnic backgrounds; however, the prevalence is higher in patients with darker skin tones.2
Key clinical features in people with darker skin tones
Melasma commonly manifests as symmetrically distributed, reticulated (lacy), dark brown to grayish brown patches on the cheeks, nose, forehead, upper lip, and chin in patients with darker skin tones.5 The pigment can be tan brown in patients with lighter skin tones. Given that postinflammatory hyperpigmentation and other pigmentary disorders can cause a similar appearance, a biopsy sometimes is needed to confirm the diagnosis, but melasma is diagnosed via physical examination in most patients. Melasma can be misdiagnosed as postinflammatory hyperpigmentation, solar lentigines, exogenous ochronosis, and Hori nevus.5
Worth noting
Prevention
- Daily sunscreen use is critical to prevent worsening of melasma. Sunscreen may not appear cosmetically elegant on darker skin tones, which creates a barrier to its use.6 Protection from both sunlight and visible light is necessary. Visible light, including light from light bulbs and device-emitted blue light, can worsen melasma. Iron oxides in tinted sunscreen offer protection from visible light.
- Physicians can recommend sunscreens that are more transparent or tinted for a better cosmetic match.
- Severe flares of melasma can occur with sun exposure despite good control with medications and laser modalities.
Treatment
- First-line therapies include topical hydroquinone 2% to 4%, tretinoin, azelaic acid, kojic acid, or ascorbic acid (vitamin C). A popular topical compound is a steroid, tretinoin, and hydroquinone.1,5 Over-the-counter hydroquinone has been removed from the market due to safety concerns; however, it is still first line in the treatment of melasma. If hydroquinone is prescribed, treatment intervals of 6 to 8 weeks followed by a hydroquinone-free period is advised to reduce the risk for exogenous ochronosis (a paradoxical darkening of the skin).
- Chemical peels are second-line treatments that are effective for melasma. Improvement in epidermal melasma has been shown with chemical peels containing Jessner solution, salicylic acid, or a-hydroxy acid. Patients with dermal and mixed melasma have seen improvement with trichloroacetic acid 25% to 35% with or without Jessner solution.1
- Cysteamine is a topical treatment created from the degradation of coenzyme A. It disrupts the synthesis of melanin to create a more even skin tone. It may be recommended in combination with sunscreen as a first-line or secondline topical therapy.
- Oral tranexamic acid is a third-line treatment that is an analogue for lysine. It decreases prostaglandin production, which leads to a lower number of tyrosine precursors available for the creation of melanin. Tranexamic acid has been shown to lighten the appearance of melasma.7 The most common and dangerous adverse effect of tranexamic acid is blood clots, and this treatment should be avoided in those on combination (estrogen and progestin) contraceptives or those with a personal or family history of clotting disorders.8
- Fourth-line treatments such as lasers (performed by dermatologists) can destroy the deposition of pigment while avoiding destruction of epidermal keratinocytes.1,9,10 They also are commonly employed in refractive melasma. The most common lasers are nonablative fractionated lasers and low-fluence Q-switched lasers. The Q-switched Nd:YAG and picosecond lasers are safe for treating melasma in darker skin tones. Ablative fractionated lasers such as CO2 lasers and erbium:YAG lasers also have been used in the treatment of melasma; however, there is still an extremely high risk for postinflammatory dyspigmentation 1 to 2 months after the procedure.10
- Although there is still a risk for rebound hyperpigmentation after laser treatment, use of topical hydroquinone pretreatment may help decrease postoperative hyperpigmentation.1,5 Patients who are treated with the incorrect laser or overtreated may develop postinflammatory hyperpigmentation, rebound hyperpigmentation, or hypopigmentation.
Health disparity highlight
Melasma, most common in patients with skin of color, is a common chronic pigmentation disorder that is cosmetically and psychologically burdensome,11 leading to decreased quality of life, emotional functioning, and self-esteem.12 Clinicians should counsel patients and work closely on long-term management. The treatment options for melasma are considered cosmetic and may be cost prohibitive for many to cover out of pocket. Topical treatments have been found to be the most cost-effective.13 Some compounding pharmacies and drug discount programs provide more affordable treatment pricing; however, some patients are still unable to afford these options.
1. Cunha PR, Kroumpouzos G. Melasma and vitiligo: novel and experimental therapies. J Clin Exp Derm Res. 2016;7:2. doi:10.4172/2155-9554.1000e106
2. Rajanala S, Maymone MBC, Vashi NA. Melasma pathogenesis: a review of the latest research, pathological findings, and investigational therapies. Dermatol Online J. 2019;25:13030/qt47b7r28c.
3. Grimes PE, Yamada N, Bhawan J. Light microscopic, immunohistochemical, and ultrastructural alterations in patients with melasma. Am J Dermatopathol. 2005;27:96-101.
4. Achar A, Rathi SK. Melasma: a clinico-epidemiological study of 312 cases. Indian J Dermatol. 2011;56:380-382.
5. Ogbechie-Godec OA, Elbuluk N. Melasma: an up-to-date comprehensive review. Dermatol Ther. 2017;7:305-318.
6. Morquette AJ, Waples ER, Heath CR. The importance of cosmetically elegant sunscreen in skin of color populations. J Cosmet Dermatol. 2022;21:1337-1338.
7. Taraz M, Nikham S, Ehsani AH. Tranexamic acid in treatment of melasma: a comprehensive review of clinical studies. Dermatol Ther. 2017;30(3). doi:10.1111/dth.12465
8. Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
9. Castanedo-Cazares JP, Hernandez-Blanco D, Carlos-Ortega B, et al. Near-visible light and UV photoprotection in the treatment of melasma: a double-blind randomized trial. Photodermatol Photoimmunol Photomed. 2014;30:35-42.
10. Trivedi MK, Yang FC, Cho BK. A review of laser and light therapy in melasma. Int J Womens Dermatol. 2017;3:11-20.
11. Dodmani PN, Deshmukh AR. Assessment of quality of life of melasma patients as per melasma quality of life scale (MELASQoL). Pigment Int. 2020;7:75-79.
12. Balkrishnan R, McMichael A, Camacho FT, et al. Development and validation of a health‐related quality of life instrument for women with melasma. Br J Dermatol. 2003;149:572-577.
13. Alikhan A, Daly M, Wu J, et al. Cost-effectiveness of a hydroquinone/tretinoin/fluocinolone acetonide cream combination in treating melasma in the United States. J Dermatolog Treat. 2010;21:276-281.
1. Cunha PR, Kroumpouzos G. Melasma and vitiligo: novel and experimental therapies. J Clin Exp Derm Res. 2016;7:2. doi:10.4172/2155-9554.1000e106
2. Rajanala S, Maymone MBC, Vashi NA. Melasma pathogenesis: a review of the latest research, pathological findings, and investigational therapies. Dermatol Online J. 2019;25:13030/qt47b7r28c.
3. Grimes PE, Yamada N, Bhawan J. Light microscopic, immunohistochemical, and ultrastructural alterations in patients with melasma. Am J Dermatopathol. 2005;27:96-101.
4. Achar A, Rathi SK. Melasma: a clinico-epidemiological study of 312 cases. Indian J Dermatol. 2011;56:380-382.
5. Ogbechie-Godec OA, Elbuluk N. Melasma: an up-to-date comprehensive review. Dermatol Ther. 2017;7:305-318.
6. Morquette AJ, Waples ER, Heath CR. The importance of cosmetically elegant sunscreen in skin of color populations. J Cosmet Dermatol. 2022;21:1337-1338.
7. Taraz M, Nikham S, Ehsani AH. Tranexamic acid in treatment of melasma: a comprehensive review of clinical studies. Dermatol Ther. 2017;30(3). doi:10.1111/dth.12465
8. Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
9. Castanedo-Cazares JP, Hernandez-Blanco D, Carlos-Ortega B, et al. Near-visible light and UV photoprotection in the treatment of melasma: a double-blind randomized trial. Photodermatol Photoimmunol Photomed. 2014;30:35-42.
10. Trivedi MK, Yang FC, Cho BK. A review of laser and light therapy in melasma. Int J Womens Dermatol. 2017;3:11-20.
11. Dodmani PN, Deshmukh AR. Assessment of quality of life of melasma patients as per melasma quality of life scale (MELASQoL). Pigment Int. 2020;7:75-79.
12. Balkrishnan R, McMichael A, Camacho FT, et al. Development and validation of a health‐related quality of life instrument for women with melasma. Br J Dermatol. 2003;149:572-577.
13. Alikhan A, Daly M, Wu J, et al. Cost-effectiveness of a hydroquinone/tretinoin/fluocinolone acetonide cream combination in treating melasma in the United States. J Dermatolog Treat. 2010;21:276-281.
Study highlights potential skin cancer risk of UV nail polish dryers
Results of a study recently published in Nature Communications suggests that According to two experts, these findings raise concerns regarding the safety of frequent use of these nail dryers.
In the study, human and mouse cells were exposed to radiation from UV nail dryers. Exposing human and mice skin cells to UVA light for 20 minutes resulted in the death of 20%-30% of cells; three consecutive 20-minute sessions resulted in the death of 65%-70% of cells. Additionally, surviving cells suffered oxidative damage to their DNA and mitochondria, with mutational patterns similar to those seen in skin cancer, study investigator Maria Zhivagui, PhD, of the University of California, San Diego, and associates reported.
“This study showed that irradiation of human and mouse cell lines using UV nail polish dryers resulted in DNA damage and genome mutations,” Shari Lipner, MD, PhD, director of the nail division at New York–Presbyterian Hospital/Weill Cornell Medicine, New York, said in an interview. The study “ties together exposure to UV light from nail polish dryers and genetic mutations that are associated with skin cancers,” added Dr. Lipner, who was not involved with the study.
UV nail lamps are commonly used to dry and harden gel nail polish formulas. Often referred to as “mini tanning beds,” these devices emit UVA radiation, classified as a Group 1 Carcinogen by the International Agency for Research on Cancer.
“Both UVA and UVB are main drivers of both melanoma and keratinocyte carcinomas (basal cell carcinoma and squamous cell carcinoma),” said Anthony Rossi, MD, a dermatologic surgeon at Memorial Sloan Kettering Cancer Center, New York, who was also not a study investigator. UV irradiance “produces DNA mutations that are specific to forming types of skin cancer,” he said in an interview.
UVA wavelengths commonly used in nail dryers can penetrate all layers of the epidermis, the top layer of the skin, potentially affecting stem cells in the skin, according to the study.
Dr. Lipner noted that “there have been several case reports of patients with histories of gel manicures using UV nail polish dryers who later developed squamous cell carcinomas on the dorsal hands, fingers, and nails, and articles describing high UV emissions from nail polish dryers, but the direct connection between UV dryers and skin cancer development was tenuous.” The first of its kind, the new study investigated the impact of UV nail drying devices at a cellular level.
The results of this study, in combination with previous case reports suggesting the development of skin cancers following UVA dryer use, raise concern regarding the safety of these commonly used devices. The study, the authors wrote, “does not provide direct evidence for an increased cancer risk in human beings,” but their findings and “prior evidence strongly suggest that radiation emitted by UV nail polish dryers may cause cancers of the hand and that UV nail polish dryers, similar to tanning beds, may increase the risk of early onset skin cancer.”
Dr. Rossi said that, “while this study shows that the UV exposure does affect human cells and causes mutations, the study was not done in vivo in human beings, so further studies are needed to know at what dose and frequency gel manicures would be needed to cause detrimental effects.” However, for people who regularly receive gel manicures involving UV nail dryers, both Dr. Lipner and Dr. Rossi recommend applying a broad-spectrum sunscreen to protect the dorsal hands, fingertips, and skin surrounding the nails, or wearing UV-protective gloves.
The study was supported by an Alfred B. Sloan Research Fellowship to one of the authors and grants from the National Institutes of Health to two authors. One author reported being a compensated consultant and having an equity interest in io9. Dr. Lipner and Dr. Rossi reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a study recently published in Nature Communications suggests that According to two experts, these findings raise concerns regarding the safety of frequent use of these nail dryers.
In the study, human and mouse cells were exposed to radiation from UV nail dryers. Exposing human and mice skin cells to UVA light for 20 minutes resulted in the death of 20%-30% of cells; three consecutive 20-minute sessions resulted in the death of 65%-70% of cells. Additionally, surviving cells suffered oxidative damage to their DNA and mitochondria, with mutational patterns similar to those seen in skin cancer, study investigator Maria Zhivagui, PhD, of the University of California, San Diego, and associates reported.
“This study showed that irradiation of human and mouse cell lines using UV nail polish dryers resulted in DNA damage and genome mutations,” Shari Lipner, MD, PhD, director of the nail division at New York–Presbyterian Hospital/Weill Cornell Medicine, New York, said in an interview. The study “ties together exposure to UV light from nail polish dryers and genetic mutations that are associated with skin cancers,” added Dr. Lipner, who was not involved with the study.
UV nail lamps are commonly used to dry and harden gel nail polish formulas. Often referred to as “mini tanning beds,” these devices emit UVA radiation, classified as a Group 1 Carcinogen by the International Agency for Research on Cancer.
“Both UVA and UVB are main drivers of both melanoma and keratinocyte carcinomas (basal cell carcinoma and squamous cell carcinoma),” said Anthony Rossi, MD, a dermatologic surgeon at Memorial Sloan Kettering Cancer Center, New York, who was also not a study investigator. UV irradiance “produces DNA mutations that are specific to forming types of skin cancer,” he said in an interview.
UVA wavelengths commonly used in nail dryers can penetrate all layers of the epidermis, the top layer of the skin, potentially affecting stem cells in the skin, according to the study.
Dr. Lipner noted that “there have been several case reports of patients with histories of gel manicures using UV nail polish dryers who later developed squamous cell carcinomas on the dorsal hands, fingers, and nails, and articles describing high UV emissions from nail polish dryers, but the direct connection between UV dryers and skin cancer development was tenuous.” The first of its kind, the new study investigated the impact of UV nail drying devices at a cellular level.
The results of this study, in combination with previous case reports suggesting the development of skin cancers following UVA dryer use, raise concern regarding the safety of these commonly used devices. The study, the authors wrote, “does not provide direct evidence for an increased cancer risk in human beings,” but their findings and “prior evidence strongly suggest that radiation emitted by UV nail polish dryers may cause cancers of the hand and that UV nail polish dryers, similar to tanning beds, may increase the risk of early onset skin cancer.”
Dr. Rossi said that, “while this study shows that the UV exposure does affect human cells and causes mutations, the study was not done in vivo in human beings, so further studies are needed to know at what dose and frequency gel manicures would be needed to cause detrimental effects.” However, for people who regularly receive gel manicures involving UV nail dryers, both Dr. Lipner and Dr. Rossi recommend applying a broad-spectrum sunscreen to protect the dorsal hands, fingertips, and skin surrounding the nails, or wearing UV-protective gloves.
The study was supported by an Alfred B. Sloan Research Fellowship to one of the authors and grants from the National Institutes of Health to two authors. One author reported being a compensated consultant and having an equity interest in io9. Dr. Lipner and Dr. Rossi reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a study recently published in Nature Communications suggests that According to two experts, these findings raise concerns regarding the safety of frequent use of these nail dryers.
In the study, human and mouse cells were exposed to radiation from UV nail dryers. Exposing human and mice skin cells to UVA light for 20 minutes resulted in the death of 20%-30% of cells; three consecutive 20-minute sessions resulted in the death of 65%-70% of cells. Additionally, surviving cells suffered oxidative damage to their DNA and mitochondria, with mutational patterns similar to those seen in skin cancer, study investigator Maria Zhivagui, PhD, of the University of California, San Diego, and associates reported.
“This study showed that irradiation of human and mouse cell lines using UV nail polish dryers resulted in DNA damage and genome mutations,” Shari Lipner, MD, PhD, director of the nail division at New York–Presbyterian Hospital/Weill Cornell Medicine, New York, said in an interview. The study “ties together exposure to UV light from nail polish dryers and genetic mutations that are associated with skin cancers,” added Dr. Lipner, who was not involved with the study.
UV nail lamps are commonly used to dry and harden gel nail polish formulas. Often referred to as “mini tanning beds,” these devices emit UVA radiation, classified as a Group 1 Carcinogen by the International Agency for Research on Cancer.
“Both UVA and UVB are main drivers of both melanoma and keratinocyte carcinomas (basal cell carcinoma and squamous cell carcinoma),” said Anthony Rossi, MD, a dermatologic surgeon at Memorial Sloan Kettering Cancer Center, New York, who was also not a study investigator. UV irradiance “produces DNA mutations that are specific to forming types of skin cancer,” he said in an interview.
UVA wavelengths commonly used in nail dryers can penetrate all layers of the epidermis, the top layer of the skin, potentially affecting stem cells in the skin, according to the study.
Dr. Lipner noted that “there have been several case reports of patients with histories of gel manicures using UV nail polish dryers who later developed squamous cell carcinomas on the dorsal hands, fingers, and nails, and articles describing high UV emissions from nail polish dryers, but the direct connection between UV dryers and skin cancer development was tenuous.” The first of its kind, the new study investigated the impact of UV nail drying devices at a cellular level.
The results of this study, in combination with previous case reports suggesting the development of skin cancers following UVA dryer use, raise concern regarding the safety of these commonly used devices. The study, the authors wrote, “does not provide direct evidence for an increased cancer risk in human beings,” but their findings and “prior evidence strongly suggest that radiation emitted by UV nail polish dryers may cause cancers of the hand and that UV nail polish dryers, similar to tanning beds, may increase the risk of early onset skin cancer.”
Dr. Rossi said that, “while this study shows that the UV exposure does affect human cells and causes mutations, the study was not done in vivo in human beings, so further studies are needed to know at what dose and frequency gel manicures would be needed to cause detrimental effects.” However, for people who regularly receive gel manicures involving UV nail dryers, both Dr. Lipner and Dr. Rossi recommend applying a broad-spectrum sunscreen to protect the dorsal hands, fingertips, and skin surrounding the nails, or wearing UV-protective gloves.
The study was supported by an Alfred B. Sloan Research Fellowship to one of the authors and grants from the National Institutes of Health to two authors. One author reported being a compensated consultant and having an equity interest in io9. Dr. Lipner and Dr. Rossi reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE COMMUNICATIONS
What are the clinical implications of recent skin dysbiosis discoveries?
NEW ORLEANS – .
“There’s still a lot for us to learn,” Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said at the annual meeting of the American Academy of Dermatology. “Multiple factors contribute to the variability in the skin microbiota, including age, sex, environment, immune system, host genotype, lifestyle, and pathobiology. The question becomes, when do these factors or impacts on the microbiota become clinically significant?”
According to Dr. Friedman, there are 10 times more bacteria cells than human cells in the human body, “but it’s not a fight to the finish; it’s not us versus them,” he said. “Together, we are a super organism.” There are also more than 500 species of bacteria on human skin excluding viruses and fungi, and each person carries up to 5 pounds of bacteria, which is akin to finding a new organ in the body.
“What’s so unique is that we each have our own bacterial fingerprint,” he said. “Whoever is sitting next to you? Their microbiota makeup is different than yours.”
Beyond genetics and environment, activities that can contribute to alterations in skin flora or skin dysbiosis include topical application of steroids, antibiotics, retinoids, harsh soaps, chemical and physical exfoliants, and resurfacing techniques. “With anything we apply or do to the skin, we are literally changing the home of many microorganisms, for good or bad,” he said.
In the realm of atopic dermatitis (AD), Staphylococcus aureus has been implicated as an offender in the pathophysiology of the disease. “It’s not about one single species of Staphylococcus, though,” said Dr. Friedman, who also is director of translational research at George Washington University. “We’re finding out that, depending on the severity of disease, Staph. epidermis may be part of the problem as opposed to it just being about Staph. aureus. Furthermore, and more importantly, these changes in the microbiota, specifically a decrease in microbial diversity, has been shown to precede a disease flare, highlighting the central role of maintaining microbial diversity and by definition, supporting the living barrier in our management of AD.”
With this in mind, researchers in one study used high-throughput sequencing to evaluate the microbial communities associated with affected and unaffected skin of 49 patients with AD before and after emollient treatment. Following 84 days of emollient application, clinical symptoms of AD improved in 72% of the study population and Stenotrophomonas species were significantly more abundant among responders.
Prebiotics, probiotics
“Our treatments certainly can positively impact the microbiota, as we have seen even recently with some of our new targeted therapies, but we can also directly provide support,” he continued. Prebiotics, which he defined as supplements or foods that contain a nondigestible ingredient that selectively stimulates the growth and/or activity of indigenous bacteria, can be found in many over-the-counter moisturizers.
For example, colloidal oatmeal has been found to support the growth of S. epidermidis and enhance the production of lactic acid. “We really don’t know much about what these induced changes mean from a clinical perspective; that has yet to be elucidated,” Dr. Friedman said.
In light of the recent attention to the early application of moisturizers in infants at high risk of developing AD in an effort to prevent or limit AD, “maybe part of this has to do with applying something that’s nurturing an evolving microbiota,” Dr. Friedman noted. “It’s something to think about.”
Yet another area of study involves the use of probiotics, which Dr. Friedman defined as supplements or foods that contain viable microorganisms that alter the microflora of the host. In a first-of-its-kind trial, researchers evaluated the safety and efficacy of self-administered topical Roseomonas mucosa in 10 adults and 5 children with AD. No adverse events or treatment complications were observed, and the topical R. mucosa was associated with significant decreases in measures of disease severity, topical steroid requirement, and S. aureus burden
In a more recent randomized trial of 11 patients with AD, Richard L. Gallo, MD, PhD, chair of dermatology, University of California, San Diego, and colleagues found that application of a personalized topical cream formulated from coagulase-negative Staphylococcus with antimicrobial activity against S. aureus reduced colonization of S. aureus and improved disease severity.
And in another randomized, controlled trial, Italian researchers enrolled 80 adults with mild to severe AD to receive a placebo or a supplement that was a mixture of lactobacilli for 56 days. They found that adults in the treatment arm showed an improvement in skin smoothness, skin moisturization, self-perception, and a decrease in the SCORing Atopic Dermatitis (SCORAD) index as well as in levels of inflammatory markers associated with AD.
Dr. Friedman also discussed postbiotics, nonviable bacterial products or metabolic byproducts from probiotic microorganisms that have biologic activity in the host. In one trial, French researchers enrolled 75 people with AD who ranged in age from 6 to 70 years to receive a cream containing a 5% lysate of the nonpathogenic bacteria Vitreoscilla filiformis, or a vehicle cream for 30 days. They found that compared with the vehicle, V. filiformis lysate significantly decreased SCORAD levels and pruritus; active cream was shown to significantly decrease loss of sleep from day 0 to day 29.
Dr. Friedman characterized these novel approaches to AD as “an exciting area, one we need to pay attention to. But what I really want to know is, aside from these purposefully made and marketed products that have pre- and postprobiotics, is there a difference with some of the products we use already? My assumption is that there is, but we need to see that data.”
Dr. Friedman disclosed that he is a consultant and/or advisory board member for Medscape/SanovaWorks, Oakstone Institute, L’Oréal, La Roche Posay, Galderma, Aveeno, Ortho Dermatologic, Microcures, Pfizer, Novartis, Lilly, Hoth Therapeutics, Zylo Therapeutics, BMS, Vial, Janssen, Novocure, Dermavant, Regeneron/Sanofi, and Incyte. He has also received grants from Pfizer, the Dermatology Foundation, Lilly, Janssen, Incyte, and Galderma.
NEW ORLEANS – .
“There’s still a lot for us to learn,” Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said at the annual meeting of the American Academy of Dermatology. “Multiple factors contribute to the variability in the skin microbiota, including age, sex, environment, immune system, host genotype, lifestyle, and pathobiology. The question becomes, when do these factors or impacts on the microbiota become clinically significant?”
According to Dr. Friedman, there are 10 times more bacteria cells than human cells in the human body, “but it’s not a fight to the finish; it’s not us versus them,” he said. “Together, we are a super organism.” There are also more than 500 species of bacteria on human skin excluding viruses and fungi, and each person carries up to 5 pounds of bacteria, which is akin to finding a new organ in the body.
“What’s so unique is that we each have our own bacterial fingerprint,” he said. “Whoever is sitting next to you? Their microbiota makeup is different than yours.”
Beyond genetics and environment, activities that can contribute to alterations in skin flora or skin dysbiosis include topical application of steroids, antibiotics, retinoids, harsh soaps, chemical and physical exfoliants, and resurfacing techniques. “With anything we apply or do to the skin, we are literally changing the home of many microorganisms, for good or bad,” he said.
In the realm of atopic dermatitis (AD), Staphylococcus aureus has been implicated as an offender in the pathophysiology of the disease. “It’s not about one single species of Staphylococcus, though,” said Dr. Friedman, who also is director of translational research at George Washington University. “We’re finding out that, depending on the severity of disease, Staph. epidermis may be part of the problem as opposed to it just being about Staph. aureus. Furthermore, and more importantly, these changes in the microbiota, specifically a decrease in microbial diversity, has been shown to precede a disease flare, highlighting the central role of maintaining microbial diversity and by definition, supporting the living barrier in our management of AD.”
With this in mind, researchers in one study used high-throughput sequencing to evaluate the microbial communities associated with affected and unaffected skin of 49 patients with AD before and after emollient treatment. Following 84 days of emollient application, clinical symptoms of AD improved in 72% of the study population and Stenotrophomonas species were significantly more abundant among responders.
Prebiotics, probiotics
“Our treatments certainly can positively impact the microbiota, as we have seen even recently with some of our new targeted therapies, but we can also directly provide support,” he continued. Prebiotics, which he defined as supplements or foods that contain a nondigestible ingredient that selectively stimulates the growth and/or activity of indigenous bacteria, can be found in many over-the-counter moisturizers.
For example, colloidal oatmeal has been found to support the growth of S. epidermidis and enhance the production of lactic acid. “We really don’t know much about what these induced changes mean from a clinical perspective; that has yet to be elucidated,” Dr. Friedman said.
In light of the recent attention to the early application of moisturizers in infants at high risk of developing AD in an effort to prevent or limit AD, “maybe part of this has to do with applying something that’s nurturing an evolving microbiota,” Dr. Friedman noted. “It’s something to think about.”
Yet another area of study involves the use of probiotics, which Dr. Friedman defined as supplements or foods that contain viable microorganisms that alter the microflora of the host. In a first-of-its-kind trial, researchers evaluated the safety and efficacy of self-administered topical Roseomonas mucosa in 10 adults and 5 children with AD. No adverse events or treatment complications were observed, and the topical R. mucosa was associated with significant decreases in measures of disease severity, topical steroid requirement, and S. aureus burden
In a more recent randomized trial of 11 patients with AD, Richard L. Gallo, MD, PhD, chair of dermatology, University of California, San Diego, and colleagues found that application of a personalized topical cream formulated from coagulase-negative Staphylococcus with antimicrobial activity against S. aureus reduced colonization of S. aureus and improved disease severity.
And in another randomized, controlled trial, Italian researchers enrolled 80 adults with mild to severe AD to receive a placebo or a supplement that was a mixture of lactobacilli for 56 days. They found that adults in the treatment arm showed an improvement in skin smoothness, skin moisturization, self-perception, and a decrease in the SCORing Atopic Dermatitis (SCORAD) index as well as in levels of inflammatory markers associated with AD.
Dr. Friedman also discussed postbiotics, nonviable bacterial products or metabolic byproducts from probiotic microorganisms that have biologic activity in the host. In one trial, French researchers enrolled 75 people with AD who ranged in age from 6 to 70 years to receive a cream containing a 5% lysate of the nonpathogenic bacteria Vitreoscilla filiformis, or a vehicle cream for 30 days. They found that compared with the vehicle, V. filiformis lysate significantly decreased SCORAD levels and pruritus; active cream was shown to significantly decrease loss of sleep from day 0 to day 29.
Dr. Friedman characterized these novel approaches to AD as “an exciting area, one we need to pay attention to. But what I really want to know is, aside from these purposefully made and marketed products that have pre- and postprobiotics, is there a difference with some of the products we use already? My assumption is that there is, but we need to see that data.”
Dr. Friedman disclosed that he is a consultant and/or advisory board member for Medscape/SanovaWorks, Oakstone Institute, L’Oréal, La Roche Posay, Galderma, Aveeno, Ortho Dermatologic, Microcures, Pfizer, Novartis, Lilly, Hoth Therapeutics, Zylo Therapeutics, BMS, Vial, Janssen, Novocure, Dermavant, Regeneron/Sanofi, and Incyte. He has also received grants from Pfizer, the Dermatology Foundation, Lilly, Janssen, Incyte, and Galderma.
NEW ORLEANS – .
“There’s still a lot for us to learn,” Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said at the annual meeting of the American Academy of Dermatology. “Multiple factors contribute to the variability in the skin microbiota, including age, sex, environment, immune system, host genotype, lifestyle, and pathobiology. The question becomes, when do these factors or impacts on the microbiota become clinically significant?”
According to Dr. Friedman, there are 10 times more bacteria cells than human cells in the human body, “but it’s not a fight to the finish; it’s not us versus them,” he said. “Together, we are a super organism.” There are also more than 500 species of bacteria on human skin excluding viruses and fungi, and each person carries up to 5 pounds of bacteria, which is akin to finding a new organ in the body.
“What’s so unique is that we each have our own bacterial fingerprint,” he said. “Whoever is sitting next to you? Their microbiota makeup is different than yours.”
Beyond genetics and environment, activities that can contribute to alterations in skin flora or skin dysbiosis include topical application of steroids, antibiotics, retinoids, harsh soaps, chemical and physical exfoliants, and resurfacing techniques. “With anything we apply or do to the skin, we are literally changing the home of many microorganisms, for good or bad,” he said.
In the realm of atopic dermatitis (AD), Staphylococcus aureus has been implicated as an offender in the pathophysiology of the disease. “It’s not about one single species of Staphylococcus, though,” said Dr. Friedman, who also is director of translational research at George Washington University. “We’re finding out that, depending on the severity of disease, Staph. epidermis may be part of the problem as opposed to it just being about Staph. aureus. Furthermore, and more importantly, these changes in the microbiota, specifically a decrease in microbial diversity, has been shown to precede a disease flare, highlighting the central role of maintaining microbial diversity and by definition, supporting the living barrier in our management of AD.”
With this in mind, researchers in one study used high-throughput sequencing to evaluate the microbial communities associated with affected and unaffected skin of 49 patients with AD before and after emollient treatment. Following 84 days of emollient application, clinical symptoms of AD improved in 72% of the study population and Stenotrophomonas species were significantly more abundant among responders.
Prebiotics, probiotics
“Our treatments certainly can positively impact the microbiota, as we have seen even recently with some of our new targeted therapies, but we can also directly provide support,” he continued. Prebiotics, which he defined as supplements or foods that contain a nondigestible ingredient that selectively stimulates the growth and/or activity of indigenous bacteria, can be found in many over-the-counter moisturizers.
For example, colloidal oatmeal has been found to support the growth of S. epidermidis and enhance the production of lactic acid. “We really don’t know much about what these induced changes mean from a clinical perspective; that has yet to be elucidated,” Dr. Friedman said.
In light of the recent attention to the early application of moisturizers in infants at high risk of developing AD in an effort to prevent or limit AD, “maybe part of this has to do with applying something that’s nurturing an evolving microbiota,” Dr. Friedman noted. “It’s something to think about.”
Yet another area of study involves the use of probiotics, which Dr. Friedman defined as supplements or foods that contain viable microorganisms that alter the microflora of the host. In a first-of-its-kind trial, researchers evaluated the safety and efficacy of self-administered topical Roseomonas mucosa in 10 adults and 5 children with AD. No adverse events or treatment complications were observed, and the topical R. mucosa was associated with significant decreases in measures of disease severity, topical steroid requirement, and S. aureus burden
In a more recent randomized trial of 11 patients with AD, Richard L. Gallo, MD, PhD, chair of dermatology, University of California, San Diego, and colleagues found that application of a personalized topical cream formulated from coagulase-negative Staphylococcus with antimicrobial activity against S. aureus reduced colonization of S. aureus and improved disease severity.
And in another randomized, controlled trial, Italian researchers enrolled 80 adults with mild to severe AD to receive a placebo or a supplement that was a mixture of lactobacilli for 56 days. They found that adults in the treatment arm showed an improvement in skin smoothness, skin moisturization, self-perception, and a decrease in the SCORing Atopic Dermatitis (SCORAD) index as well as in levels of inflammatory markers associated with AD.
Dr. Friedman also discussed postbiotics, nonviable bacterial products or metabolic byproducts from probiotic microorganisms that have biologic activity in the host. In one trial, French researchers enrolled 75 people with AD who ranged in age from 6 to 70 years to receive a cream containing a 5% lysate of the nonpathogenic bacteria Vitreoscilla filiformis, or a vehicle cream for 30 days. They found that compared with the vehicle, V. filiformis lysate significantly decreased SCORAD levels and pruritus; active cream was shown to significantly decrease loss of sleep from day 0 to day 29.
Dr. Friedman characterized these novel approaches to AD as “an exciting area, one we need to pay attention to. But what I really want to know is, aside from these purposefully made and marketed products that have pre- and postprobiotics, is there a difference with some of the products we use already? My assumption is that there is, but we need to see that data.”
Dr. Friedman disclosed that he is a consultant and/or advisory board member for Medscape/SanovaWorks, Oakstone Institute, L’Oréal, La Roche Posay, Galderma, Aveeno, Ortho Dermatologic, Microcures, Pfizer, Novartis, Lilly, Hoth Therapeutics, Zylo Therapeutics, BMS, Vial, Janssen, Novocure, Dermavant, Regeneron/Sanofi, and Incyte. He has also received grants from Pfizer, the Dermatology Foundation, Lilly, Janssen, Incyte, and Galderma.
AT AAD 2023
Painful axillary lesions
The patient’s recurrent indurated nodules under her arms and other intertriginous areas, often draining pus, are consistent with a diagnosis of hidradenitis suppurativa (HS).
HS is a chronic inflammatory and suppurative skin condition that primarily involves the sweat glands.1 The most commonly affected sites are intertriginous areas that include the axillae, groin, and perianal and inframammary regions.2 Prevalence of this disorder ranges from 0.05% to 4.1% of the population with an onset from puberty to adulthood, usually at around 40 years of age.3 Its incidence is twice as high in women as men and is more common in Black individuals.4,5
While its pathogenesis is not fully understood, it’s believed that excess proliferation of keratinocytes contributes to occlusion, leading to plugging of hair follicle ducts. Hormones, smoking, and obesity may contribute to and exacerbate HS. Intertriginous areas are prone to friction, leading to inflammation and further clogging.
The inflammation evolves into a chronic foreign body-type granulomatous inflammation with the potential for rupture, tunneling, and draining sinuses, which, although malodorous, are sterile, separating HS from an infected abscess.5 The result is thick, dense, scarred tissue.
The diagnosis is clinical in nature, with the history and physical exam distinguishing it from other skin disorders. In addition to the recurring physical pain, there is the emotional distress and self-consciousness about the drainage, odor, and scarring. This particular patient said that she avoided wearing sleeveless shirts due to the lesions’ appearance.
Treatment is multifactorial. Smoking and obesity are contributory factors, so smoking cessation and weight loss are recommended. For very mild HS, topical clindamycin 1% twice daily may suffice, but usually, due to the amount of inflammation, oral antibiotics are the initial therapy. (The use of antibiotics is for their anti-inflammatory component, as the nodules and unruptured tracts are sterile.)
Doxycycline 100 mg twice daily is the usual starting systemic antibiotic. In more severe or resistant cases, a combination of clindamycin and rifampin 300 mg each twice daily is used. (Worth noting: Rifampin interacts with oral contraceptives and many of these patients are women of reproductive age.) Treatment length is usually long (10 to 12 weeks) and recurrence is common.3
Spironolactone 100 mg daily and metformin 1000 mg extended release daily, which reduces insulin resistance, may be helpful. Intralesional injections of 10 mg/mL of triamcinolone in sterile saline can relieve the painful inflamed tracts. Referral for biologic agents, including infliximab, may be needed in severe cases that do not respond to other measures. Although invasive, wide debridement of the diseased tissue can reduce the disease burden.6
This particular patient said that she’d stopped smoking 3 years earlier and would work on losing weight. She was prescribed topical clindamycin 1% lotion twice daily along with oral clindamycin and rifampin dosed as above for 3 months. She declined metformin and intralesional injections. At a follow-up appointment 3 weeks later, she was pleased with the decrease in inflammation and had only 1 remaining tender area of fluctuance. She again declined injections and planned to continue on her oral and topical antibiotics.
Photo courtesy of Daniel Stulberg, MD, FAAFP. Text courtesy of Derissa F. Raynold, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Wolkenstein P, Loundou A, Barrau K, et al. Quality of life impairment in hidradenitis suppurativa: a study of 61 cases. J Am Acad Dermatol. 2007;56:621-623. doi: 10.1016/j.jaad.2006.08.061
2. Storer MA, Danesh MJ, Sandhu ME, et al. An assessment of the relative impact of hidradenitis suppurativa, psoriasis, and obesity on quality of life. Int J Womens Dermatol. 2018;4:198-202. doi: 10.1016/j.ijwd.2018.08.009
3. Saunte DML, Jemec GBE. Hidradenitis suppurativa: advances in diagnosis and treatment. JAMA. 2017;318:2019-2032. doi: 10.1001/jama.2017.16691
4. Matusiak L, Bieniek A, Szepietowski JC. Psychophysical aspects of hidradenitis suppurativa. Acta Derm Venereol. 2010;90:264-268. doi: 10.2340/00015555-0866
5. Esmann S, Jemec GB. Psychosocial impact of hidradenitis suppurativa: a qualitative study. Acta Derm Venereol. 2011;91:328-332. doi: 10.2340/00015555-1082
6. Caposiena Caro RD, Cannizzaro MV, Botti E, et al. Clindamycin versus clindamycin plus rifampicin in hidradenitis suppurativa treatment: clinical and ultrasound observations. J Am Acad Dermatol. 2019;80:1314-1321. doi: 10.1016/j.jaad.2018.11.035

The patient’s recurrent indurated nodules under her arms and other intertriginous areas, often draining pus, are consistent with a diagnosis of hidradenitis suppurativa (HS).
HS is a chronic inflammatory and suppurative skin condition that primarily involves the sweat glands.1 The most commonly affected sites are intertriginous areas that include the axillae, groin, and perianal and inframammary regions.2 Prevalence of this disorder ranges from 0.05% to 4.1% of the population with an onset from puberty to adulthood, usually at around 40 years of age.3 Its incidence is twice as high in women as men and is more common in Black individuals.4,5
While its pathogenesis is not fully understood, it’s believed that excess proliferation of keratinocytes contributes to occlusion, leading to plugging of hair follicle ducts. Hormones, smoking, and obesity may contribute to and exacerbate HS. Intertriginous areas are prone to friction, leading to inflammation and further clogging.
The inflammation evolves into a chronic foreign body-type granulomatous inflammation with the potential for rupture, tunneling, and draining sinuses, which, although malodorous, are sterile, separating HS from an infected abscess.5 The result is thick, dense, scarred tissue.
The diagnosis is clinical in nature, with the history and physical exam distinguishing it from other skin disorders. In addition to the recurring physical pain, there is the emotional distress and self-consciousness about the drainage, odor, and scarring. This particular patient said that she avoided wearing sleeveless shirts due to the lesions’ appearance.
Treatment is multifactorial. Smoking and obesity are contributory factors, so smoking cessation and weight loss are recommended. For very mild HS, topical clindamycin 1% twice daily may suffice, but usually, due to the amount of inflammation, oral antibiotics are the initial therapy. (The use of antibiotics is for their anti-inflammatory component, as the nodules and unruptured tracts are sterile.)
Doxycycline 100 mg twice daily is the usual starting systemic antibiotic. In more severe or resistant cases, a combination of clindamycin and rifampin 300 mg each twice daily is used. (Worth noting: Rifampin interacts with oral contraceptives and many of these patients are women of reproductive age.) Treatment length is usually long (10 to 12 weeks) and recurrence is common.3
Spironolactone 100 mg daily and metformin 1000 mg extended release daily, which reduces insulin resistance, may be helpful. Intralesional injections of 10 mg/mL of triamcinolone in sterile saline can relieve the painful inflamed tracts. Referral for biologic agents, including infliximab, may be needed in severe cases that do not respond to other measures. Although invasive, wide debridement of the diseased tissue can reduce the disease burden.6
This particular patient said that she’d stopped smoking 3 years earlier and would work on losing weight. She was prescribed topical clindamycin 1% lotion twice daily along with oral clindamycin and rifampin dosed as above for 3 months. She declined metformin and intralesional injections. At a follow-up appointment 3 weeks later, she was pleased with the decrease in inflammation and had only 1 remaining tender area of fluctuance. She again declined injections and planned to continue on her oral and topical antibiotics.
Photo courtesy of Daniel Stulberg, MD, FAAFP. Text courtesy of Derissa F. Raynold, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

The patient’s recurrent indurated nodules under her arms and other intertriginous areas, often draining pus, are consistent with a diagnosis of hidradenitis suppurativa (HS).
HS is a chronic inflammatory and suppurative skin condition that primarily involves the sweat glands.1 The most commonly affected sites are intertriginous areas that include the axillae, groin, and perianal and inframammary regions.2 Prevalence of this disorder ranges from 0.05% to 4.1% of the population with an onset from puberty to adulthood, usually at around 40 years of age.3 Its incidence is twice as high in women as men and is more common in Black individuals.4,5
While its pathogenesis is not fully understood, it’s believed that excess proliferation of keratinocytes contributes to occlusion, leading to plugging of hair follicle ducts. Hormones, smoking, and obesity may contribute to and exacerbate HS. Intertriginous areas are prone to friction, leading to inflammation and further clogging.
The inflammation evolves into a chronic foreign body-type granulomatous inflammation with the potential for rupture, tunneling, and draining sinuses, which, although malodorous, are sterile, separating HS from an infected abscess.5 The result is thick, dense, scarred tissue.
The diagnosis is clinical in nature, with the history and physical exam distinguishing it from other skin disorders. In addition to the recurring physical pain, there is the emotional distress and self-consciousness about the drainage, odor, and scarring. This particular patient said that she avoided wearing sleeveless shirts due to the lesions’ appearance.
Treatment is multifactorial. Smoking and obesity are contributory factors, so smoking cessation and weight loss are recommended. For very mild HS, topical clindamycin 1% twice daily may suffice, but usually, due to the amount of inflammation, oral antibiotics are the initial therapy. (The use of antibiotics is for their anti-inflammatory component, as the nodules and unruptured tracts are sterile.)
Doxycycline 100 mg twice daily is the usual starting systemic antibiotic. In more severe or resistant cases, a combination of clindamycin and rifampin 300 mg each twice daily is used. (Worth noting: Rifampin interacts with oral contraceptives and many of these patients are women of reproductive age.) Treatment length is usually long (10 to 12 weeks) and recurrence is common.3
Spironolactone 100 mg daily and metformin 1000 mg extended release daily, which reduces insulin resistance, may be helpful. Intralesional injections of 10 mg/mL of triamcinolone in sterile saline can relieve the painful inflamed tracts. Referral for biologic agents, including infliximab, may be needed in severe cases that do not respond to other measures. Although invasive, wide debridement of the diseased tissue can reduce the disease burden.6
This particular patient said that she’d stopped smoking 3 years earlier and would work on losing weight. She was prescribed topical clindamycin 1% lotion twice daily along with oral clindamycin and rifampin dosed as above for 3 months. She declined metformin and intralesional injections. At a follow-up appointment 3 weeks later, she was pleased with the decrease in inflammation and had only 1 remaining tender area of fluctuance. She again declined injections and planned to continue on her oral and topical antibiotics.
Photo courtesy of Daniel Stulberg, MD, FAAFP. Text courtesy of Derissa F. Raynold, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Wolkenstein P, Loundou A, Barrau K, et al. Quality of life impairment in hidradenitis suppurativa: a study of 61 cases. J Am Acad Dermatol. 2007;56:621-623. doi: 10.1016/j.jaad.2006.08.061
2. Storer MA, Danesh MJ, Sandhu ME, et al. An assessment of the relative impact of hidradenitis suppurativa, psoriasis, and obesity on quality of life. Int J Womens Dermatol. 2018;4:198-202. doi: 10.1016/j.ijwd.2018.08.009
3. Saunte DML, Jemec GBE. Hidradenitis suppurativa: advances in diagnosis and treatment. JAMA. 2017;318:2019-2032. doi: 10.1001/jama.2017.16691
4. Matusiak L, Bieniek A, Szepietowski JC. Psychophysical aspects of hidradenitis suppurativa. Acta Derm Venereol. 2010;90:264-268. doi: 10.2340/00015555-0866
5. Esmann S, Jemec GB. Psychosocial impact of hidradenitis suppurativa: a qualitative study. Acta Derm Venereol. 2011;91:328-332. doi: 10.2340/00015555-1082
6. Caposiena Caro RD, Cannizzaro MV, Botti E, et al. Clindamycin versus clindamycin plus rifampicin in hidradenitis suppurativa treatment: clinical and ultrasound observations. J Am Acad Dermatol. 2019;80:1314-1321. doi: 10.1016/j.jaad.2018.11.035
1. Wolkenstein P, Loundou A, Barrau K, et al. Quality of life impairment in hidradenitis suppurativa: a study of 61 cases. J Am Acad Dermatol. 2007;56:621-623. doi: 10.1016/j.jaad.2006.08.061
2. Storer MA, Danesh MJ, Sandhu ME, et al. An assessment of the relative impact of hidradenitis suppurativa, psoriasis, and obesity on quality of life. Int J Womens Dermatol. 2018;4:198-202. doi: 10.1016/j.ijwd.2018.08.009
3. Saunte DML, Jemec GBE. Hidradenitis suppurativa: advances in diagnosis and treatment. JAMA. 2017;318:2019-2032. doi: 10.1001/jama.2017.16691
4. Matusiak L, Bieniek A, Szepietowski JC. Psychophysical aspects of hidradenitis suppurativa. Acta Derm Venereol. 2010;90:264-268. doi: 10.2340/00015555-0866
5. Esmann S, Jemec GB. Psychosocial impact of hidradenitis suppurativa: a qualitative study. Acta Derm Venereol. 2011;91:328-332. doi: 10.2340/00015555-1082
6. Caposiena Caro RD, Cannizzaro MV, Botti E, et al. Clindamycin versus clindamycin plus rifampicin in hidradenitis suppurativa treatment: clinical and ultrasound observations. J Am Acad Dermatol. 2019;80:1314-1321. doi: 10.1016/j.jaad.2018.11.035
Penile rash
Biopsy revealed a lichenoid infiltrate in the upper dermis with thinning of the epidermis and a predominance of plasma cells. This finding, along with a red-orange, glossy lesion on the glans penis in an uncircumcised man is a classic presentation of Zoon balanitis.
Zoon balanitis is a chronic, idiopathic disorder that affects uncircumcised men who are middle-aged and older.1 Although the exact pathogenesis is unknown, it is hypothesized to be the result of chronic irritation due to poor hygiene/urine retention with prepuce dysfunction. The classic clinical presentation is an asymptomatic, well-defined, orange-to-red glazed patch with symmetric small red “cayenne pepper” spots contained within the glans penis and/or prepuce. Often there are symmetric “kissing lesions” where a second lesion of similar morphology is apparent on the prepuce where it meets the glans penis. While this disorder is typically asymptomatic, it can be associated with itching and/or burning.
There are several other inflammatory disorders in the differential for Zoon balanitis (erosive lichen planus, lichen sclerosus, psoriasis), but the most important consideration is penile intraepithelial carcinoma, specifically erythroplasia of Queyrat. Erythroplasia of Queyrat can be difficult to distinguish clinically and often requires a biopsy. Zoon balanitis will show a plasma cell predominant lichenoid infiltrate, whereas erythroplasia of Queyrat will show squamous cell carcinoma in situ.
It is important to note that Zoon balanitis is a clinicopathologic diagnosis and that zoonoid inflammation on biopsy is not pathognomonic, as this can also be seen in other inflammatory and neoplastic conditions. It is, therefore, advisable to follow up with patients with Zoon balanitis to ensure that the lesion is resolving and/or not getting worse.
Improved hygiene measures are the mainstay of treatment; circumcision is also effective.2 Both can help keep the glans clean and dry. In those with symptomatic disease, low-strength topical steroids including 1% hydrocortisone ointment or cream twice daily or topical calcineurin inhibitors (pimecrolimus 1% or tacrolimus 0.1%) twice daily can be used for symptom management.
Because this patient’s disease was asymptomatic, treatment was deferred. He was counseled to draw back the foreskin when urinating and to do the same while showering so that he could wash, then dry, the glans before returning the foreskin to its normal position. The patient is being monitored clinically.
Photo courtesy of Drew Mitchell, MD. Text courtesy of Drew Mitchell, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Mallon E, Hawkins D, Dinneen M, et al. Circumcision and genital dermatoses. Arch Dermatol. 2000;136:350-354. doi: 10.1001/archderm.136.3.350
2. Kumar B, Sharma R, Rajagopalan M, et al. Plasma cell balanitis: clinical and histopathological features—response to circumcision. Genitourin Med. 1995;71:32-34. doi: 10.1136/sti.71.1.32

Biopsy revealed a lichenoid infiltrate in the upper dermis with thinning of the epidermis and a predominance of plasma cells. This finding, along with a red-orange, glossy lesion on the glans penis in an uncircumcised man is a classic presentation of Zoon balanitis.
Zoon balanitis is a chronic, idiopathic disorder that affects uncircumcised men who are middle-aged and older.1 Although the exact pathogenesis is unknown, it is hypothesized to be the result of chronic irritation due to poor hygiene/urine retention with prepuce dysfunction. The classic clinical presentation is an asymptomatic, well-defined, orange-to-red glazed patch with symmetric small red “cayenne pepper” spots contained within the glans penis and/or prepuce. Often there are symmetric “kissing lesions” where a second lesion of similar morphology is apparent on the prepuce where it meets the glans penis. While this disorder is typically asymptomatic, it can be associated with itching and/or burning.
There are several other inflammatory disorders in the differential for Zoon balanitis (erosive lichen planus, lichen sclerosus, psoriasis), but the most important consideration is penile intraepithelial carcinoma, specifically erythroplasia of Queyrat. Erythroplasia of Queyrat can be difficult to distinguish clinically and often requires a biopsy. Zoon balanitis will show a plasma cell predominant lichenoid infiltrate, whereas erythroplasia of Queyrat will show squamous cell carcinoma in situ.
It is important to note that Zoon balanitis is a clinicopathologic diagnosis and that zoonoid inflammation on biopsy is not pathognomonic, as this can also be seen in other inflammatory and neoplastic conditions. It is, therefore, advisable to follow up with patients with Zoon balanitis to ensure that the lesion is resolving and/or not getting worse.
Improved hygiene measures are the mainstay of treatment; circumcision is also effective.2 Both can help keep the glans clean and dry. In those with symptomatic disease, low-strength topical steroids including 1% hydrocortisone ointment or cream twice daily or topical calcineurin inhibitors (pimecrolimus 1% or tacrolimus 0.1%) twice daily can be used for symptom management.
Because this patient’s disease was asymptomatic, treatment was deferred. He was counseled to draw back the foreskin when urinating and to do the same while showering so that he could wash, then dry, the glans before returning the foreskin to its normal position. The patient is being monitored clinically.
Photo courtesy of Drew Mitchell, MD. Text courtesy of Drew Mitchell, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

Biopsy revealed a lichenoid infiltrate in the upper dermis with thinning of the epidermis and a predominance of plasma cells. This finding, along with a red-orange, glossy lesion on the glans penis in an uncircumcised man is a classic presentation of Zoon balanitis.
Zoon balanitis is a chronic, idiopathic disorder that affects uncircumcised men who are middle-aged and older.1 Although the exact pathogenesis is unknown, it is hypothesized to be the result of chronic irritation due to poor hygiene/urine retention with prepuce dysfunction. The classic clinical presentation is an asymptomatic, well-defined, orange-to-red glazed patch with symmetric small red “cayenne pepper” spots contained within the glans penis and/or prepuce. Often there are symmetric “kissing lesions” where a second lesion of similar morphology is apparent on the prepuce where it meets the glans penis. While this disorder is typically asymptomatic, it can be associated with itching and/or burning.
There are several other inflammatory disorders in the differential for Zoon balanitis (erosive lichen planus, lichen sclerosus, psoriasis), but the most important consideration is penile intraepithelial carcinoma, specifically erythroplasia of Queyrat. Erythroplasia of Queyrat can be difficult to distinguish clinically and often requires a biopsy. Zoon balanitis will show a plasma cell predominant lichenoid infiltrate, whereas erythroplasia of Queyrat will show squamous cell carcinoma in situ.
It is important to note that Zoon balanitis is a clinicopathologic diagnosis and that zoonoid inflammation on biopsy is not pathognomonic, as this can also be seen in other inflammatory and neoplastic conditions. It is, therefore, advisable to follow up with patients with Zoon balanitis to ensure that the lesion is resolving and/or not getting worse.
Improved hygiene measures are the mainstay of treatment; circumcision is also effective.2 Both can help keep the glans clean and dry. In those with symptomatic disease, low-strength topical steroids including 1% hydrocortisone ointment or cream twice daily or topical calcineurin inhibitors (pimecrolimus 1% or tacrolimus 0.1%) twice daily can be used for symptom management.
Because this patient’s disease was asymptomatic, treatment was deferred. He was counseled to draw back the foreskin when urinating and to do the same while showering so that he could wash, then dry, the glans before returning the foreskin to its normal position. The patient is being monitored clinically.
Photo courtesy of Drew Mitchell, MD. Text courtesy of Drew Mitchell, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Mallon E, Hawkins D, Dinneen M, et al. Circumcision and genital dermatoses. Arch Dermatol. 2000;136:350-354. doi: 10.1001/archderm.136.3.350
2. Kumar B, Sharma R, Rajagopalan M, et al. Plasma cell balanitis: clinical and histopathological features—response to circumcision. Genitourin Med. 1995;71:32-34. doi: 10.1136/sti.71.1.32
1. Mallon E, Hawkins D, Dinneen M, et al. Circumcision and genital dermatoses. Arch Dermatol. 2000;136:350-354. doi: 10.1001/archderm.136.3.350
2. Kumar B, Sharma R, Rajagopalan M, et al. Plasma cell balanitis: clinical and histopathological features—response to circumcision. Genitourin Med. 1995;71:32-34. doi: 10.1136/sti.71.1.32
Analysis identifies gaps in CV risk screening of patients with psoriasis
Just , according to an analysis of 10 years of national survey data.
From 2007 to 2016, national screening rates for four CV risk factors at 14.8 million psoriasis-related visits to dermatology providers were 11% (body-mass index), 7.4% (blood pressure), 2.9% (cholesterol), and 1.7% (glucose). Data from the National Ambulatory Medical Care Survey showed that at least one of the four factors was screened at 16% of dermatology visits, said William B. Song, BS, of the department of dermatology, University of Pennsylvania, Philadelphia, and associates.
The main focus of their study, however, was regional differences. “CV risk factor screening by dermatology providers for patients with psoriasis is low across all regions of the United States and lowest in the South, the region that experiences the highest CVD burden in the United States,” they wrote in a letter to the editor.
Compared with the South, the adjusted odds of any CV screening were 0.98 in the West, 1.25 in the Northeast, and 1.92 in the Midwest. Blood pressure screening was significantly higher in all three regions, compared with the South, while BMI screening was actually lower in the West (0.74), the investigators reported. Odds ratios were not available for cholesterol and glucose screening because of sample size limitations.
The regional variation in screening rates “is not explained by patient demographics or disease severity,” they noted, adding that 2.8 million visits with BP screening would have been added over the 10-year study period “if providers in the South screened patients with psoriasis for high blood pressure at the same rate as providers in the Northeast.”
Guidelines published in 2019 by the American Academy of Dermatology and the National Psoriasis Foundation – which were cowritten by Joel M. Gelfand, MD, senior author of the current study – noted that dermatologists “play an important role in evidence-based screening of CV risk factors in patients with psoriasis,” the investigators wrote. But the regional variations suggest “that some regions experience barriers to appropriate screening or challenges in adhering to guidelines for managing psoriasis and CV risk.”
While the lack of data from after 2016 is one of the study limitations, they added, “continued efforts to develop effective interventions to improve CV screening and care for people with psoriasis in all regions of the U.S. are needed to more effectively address the burden of CV disease experienced by people with psoriasis.”
The study was partly funded by the National Psoriasis Foundation. Three of the seven investigators disclosed earnings from private companies in the form of consultant fees, research support, and honoraria. Dr. Gelfand is a deputy editor for the Journal of Investigative Dermatology.
Just , according to an analysis of 10 years of national survey data.
From 2007 to 2016, national screening rates for four CV risk factors at 14.8 million psoriasis-related visits to dermatology providers were 11% (body-mass index), 7.4% (blood pressure), 2.9% (cholesterol), and 1.7% (glucose). Data from the National Ambulatory Medical Care Survey showed that at least one of the four factors was screened at 16% of dermatology visits, said William B. Song, BS, of the department of dermatology, University of Pennsylvania, Philadelphia, and associates.
The main focus of their study, however, was regional differences. “CV risk factor screening by dermatology providers for patients with psoriasis is low across all regions of the United States and lowest in the South, the region that experiences the highest CVD burden in the United States,” they wrote in a letter to the editor.
Compared with the South, the adjusted odds of any CV screening were 0.98 in the West, 1.25 in the Northeast, and 1.92 in the Midwest. Blood pressure screening was significantly higher in all three regions, compared with the South, while BMI screening was actually lower in the West (0.74), the investigators reported. Odds ratios were not available for cholesterol and glucose screening because of sample size limitations.
The regional variation in screening rates “is not explained by patient demographics or disease severity,” they noted, adding that 2.8 million visits with BP screening would have been added over the 10-year study period “if providers in the South screened patients with psoriasis for high blood pressure at the same rate as providers in the Northeast.”
Guidelines published in 2019 by the American Academy of Dermatology and the National Psoriasis Foundation – which were cowritten by Joel M. Gelfand, MD, senior author of the current study – noted that dermatologists “play an important role in evidence-based screening of CV risk factors in patients with psoriasis,” the investigators wrote. But the regional variations suggest “that some regions experience barriers to appropriate screening or challenges in adhering to guidelines for managing psoriasis and CV risk.”
While the lack of data from after 2016 is one of the study limitations, they added, “continued efforts to develop effective interventions to improve CV screening and care for people with psoriasis in all regions of the U.S. are needed to more effectively address the burden of CV disease experienced by people with psoriasis.”
The study was partly funded by the National Psoriasis Foundation. Three of the seven investigators disclosed earnings from private companies in the form of consultant fees, research support, and honoraria. Dr. Gelfand is a deputy editor for the Journal of Investigative Dermatology.
Just , according to an analysis of 10 years of national survey data.
From 2007 to 2016, national screening rates for four CV risk factors at 14.8 million psoriasis-related visits to dermatology providers were 11% (body-mass index), 7.4% (blood pressure), 2.9% (cholesterol), and 1.7% (glucose). Data from the National Ambulatory Medical Care Survey showed that at least one of the four factors was screened at 16% of dermatology visits, said William B. Song, BS, of the department of dermatology, University of Pennsylvania, Philadelphia, and associates.
The main focus of their study, however, was regional differences. “CV risk factor screening by dermatology providers for patients with psoriasis is low across all regions of the United States and lowest in the South, the region that experiences the highest CVD burden in the United States,” they wrote in a letter to the editor.
Compared with the South, the adjusted odds of any CV screening were 0.98 in the West, 1.25 in the Northeast, and 1.92 in the Midwest. Blood pressure screening was significantly higher in all three regions, compared with the South, while BMI screening was actually lower in the West (0.74), the investigators reported. Odds ratios were not available for cholesterol and glucose screening because of sample size limitations.
The regional variation in screening rates “is not explained by patient demographics or disease severity,” they noted, adding that 2.8 million visits with BP screening would have been added over the 10-year study period “if providers in the South screened patients with psoriasis for high blood pressure at the same rate as providers in the Northeast.”
Guidelines published in 2019 by the American Academy of Dermatology and the National Psoriasis Foundation – which were cowritten by Joel M. Gelfand, MD, senior author of the current study – noted that dermatologists “play an important role in evidence-based screening of CV risk factors in patients with psoriasis,” the investigators wrote. But the regional variations suggest “that some regions experience barriers to appropriate screening or challenges in adhering to guidelines for managing psoriasis and CV risk.”
While the lack of data from after 2016 is one of the study limitations, they added, “continued efforts to develop effective interventions to improve CV screening and care for people with psoriasis in all regions of the U.S. are needed to more effectively address the burden of CV disease experienced by people with psoriasis.”
The study was partly funded by the National Psoriasis Foundation. Three of the seven investigators disclosed earnings from private companies in the form of consultant fees, research support, and honoraria. Dr. Gelfand is a deputy editor for the Journal of Investigative Dermatology.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Frustration over iPLEDGE evident at FDA meeting
During 2 days of after the chaotic rollout of the new REMS platform at the end of 2021.
On March 29, at the end of the FDA’s joint meeting of two advisory committees that addressed ways to improve the iPLEDGE program, most panelists voted to change the 19-day lockout period for patients who can become pregnant, and the requirement that every month, providers must document counseling of those who cannot get pregnant and are taking the drug for acne.
However, there was no consensus on whether there should be a lockout at all or for how long, and what an appropriate interval for counseling those who cannot get pregnant would be, if not monthly. Those voting on the questions repeatedly cited a lack of data to make well-informed decisions.
The meeting of the two panels, the FDA’s Drug Safety and Risk Management Advisory Committee and the Dermatologic and Ophthalmic Drugs Advisory Committee, was held March 28-29, to discuss proposed changes to iPLEDGE requirements, to minimize the program’s burden on patients, prescribers, and pharmacies – while maintaining safe use of the highly teratogenic drug.
Lockout based on outdated reasoning
John S. Barbieri, MD, a dermatologist and epidemiologist, and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital in Boston, speaking as deputy chair of the American Academy of Dermatology Association (AADA) iPLEDGE work group, described the burden of getting the drug to patients. He was not on the panel, but spoke during the open public hearing.
“Compared to other acne medications, the time it takes to successfully go from prescribed (isotretinoin) to when the patient actually has it in their hands is 5- to 10-fold higher,” he said.
Among the barriers is the 19-day lockout period for people who can get pregnant and miss the 7-day window for picking up their prescriptions. They must then wait 19 days to get a pregnancy test to clear them for receiving the medication.
Gregory Wedin, PharmD, pharmacovigilance and risk management director of Upsher-Smith Laboratories, who spoke on behalf of the Isotretinoin Products Manufacturer Group (IPMG), which manages iPLEDGE, said, “The rationale for the 19-day wait is to ensure the next confirmatory pregnancy test is completed after the most fertile period of the menstrual cycle is passed.”
Many don’t have a monthly cycle
But Dr. Barbieri said that reasoning is outdated.
“The current program’s focus on the menstrual cycle is really an antiquated approach,” he said. “Many patients do not have a monthly cycle due to medical conditions like polycystic ovarian syndrome, or due to [certain kinds of] contraception.”
He added, “By removing this 19-day lockout and, really, the archaic timing around the menstrual cycle in general in this program, we can simplify the program, improve it, and better align it with the real-world biology of our patients.” He added that patients are often missing the 7-day window for picking up their prescriptions through no fault of their own. Speakers at the hearing also mentioned insurance hassles and ordering delays.
Communication with IPMG
Ilona Frieden, MD, professor of dermatology and pediatrics at the University of California, San Francisco, and outgoing chair of the AADA iPLEDGE work group, cited difficulty in working with IPMG on modifications as another barrier. She also spoke during the open public hearing.
“Despite many, many attempts to work with the IPMG, we are not aware of any organizational structure or key leaders to communicate with. Instead we have been given repeatedly a generic email address for trying to establish a working relationship and we believe this may explain the inaction of the IPMG since our proposals 4 years ago in 2019.”
Among those proposals, she said, were allowing telemedicine visits as part of the iPLEDGE REMS program and reducing counseling attestation to every 6 months instead of monthly for those who cannot become pregnant.
She pointed to the chaotic rollout of modifications to the iPLEDGE program on a new website at the end of 2021.
In 2021, she said, “despite 6 months of notification, no prescriber input was solicited before revamping the website. This lack of transparency and accountability has been a major hurdle in improving iPLEDGE.”
Dr. Barbieri called the rollout “a debacle” that could have been mitigated with communication with IPMG. “We warned about every issue that happened and talked about ways to mitigate it and were largely ignored,” he said.
“By including dermatologists and key stakeholders in these discussions, as we move forward with changes to improve this program, we can make sure that it’s patient-centered.”
IPMG did not address the specific complaints about the working relationship with the AADA workgroup at the meeting.
Monthly attestation for counseling patients who cannot get pregnant
Dr. Barbieri said the monthly requirement to counsel patients who cannot get pregnant and document that counseling unfairly burdens clinicians and patients. “We’re essentially asking patients to come in monthly just to tell them not to share their drugs [or] donate blood,” he said.
Ken Katz, MD, MSc, a dermatologist at Kaiser Permanente in San Francisco, was among the panel members voting not to continue the 19-day lockout.
“I think this places an unduly high burden physically and psychologically on our patients. It seems arbitrary,” he said. “Likely we will miss some pregnancies; we are missing some already. But the burden is not matched by the benefit.”
IPMG representative Dr. Wedin, said, “while we cannot support eliminating or extending the confirmation interval to a year, the [iPLEDGE] sponsors are agreeable [to] a 120-day confirmation interval.”
He said that while an extension to 120 days would reduce burden on prescribers, it comes with the risk in reducing oversight by a certified iPLEDGE prescriber and potentially increasing the risk for drug sharing.
“A patient may be more likely to share their drug with another person the further along with therapy they get as their condition improves,” Dr. Wedin said.
Home pregnancy testing
The advisory groups were also tasked with discussing whether home pregnancy tests, allowed during the COVID-19 public health emergency, should continue to be allowed. Most committee members and those in the public hearing who spoke on the issue agreed that home tests should continue in an effort to increase access and decrease burden.
During the pandemic, iPLEDGE rules have been relaxed from having a pregnancy test done only at a Clinical Laboratory Improvement Amendments–certified laboratory.
Lindsey Crist, PharmD, a risk management analyst at the FDA, who presented the FDA review committee’s analysis, said that the FDA’s review committee recommends ending the allowance of home tests, citing insufficient data on use and the discovery of instances of falsification of pregnancy tests.
“One study at an academic medical center reviewed the medical records of 89 patients who used home pregnancy tests while taking isotretinoin during the public health emergency. It found that 15.7% submitted falsified pregnancy test results,” Dr. Crist said.
Dr. Crist added, however, that the review committee recommends allowing the tests to be done in a provider’s office as an alternative.
Workaround to avoid falsification
Advisory committee member Brian P. Green, DO, associate professor of dermatology at Penn State University, Hershey, Pa., spoke in support of home pregnancy tests.
“What we have people do for telemedicine is take the stick, write their name, write the date on it, and send a picture of that the same day as their visit,” he said. “That way we have the pregnancy test the same day. Allowing this to continue to happen at home is important. Bringing people in is burdensome and costly.”
Emmy Graber, MD, a dermatologist who practices in Boston, and a director of the American Acne and Rosacea Society (AARS), relayed an example of the burden for a patient using isotretinoin who lives 1.5 hours away from the dermatology office. She is able to meet the requirements of iPLEDGE only through telehealth.
“Home pregnancy tests are highly sensitive, equal to the ones done in CLIA-certified labs, and highly accurate when interpreted by a dermatology provider,” said Dr. Graber, who spoke on behalf of the AARS during the open public hearing.
“Notably, CLIA [Clinical Laboratory Improvement Amendments] certification is not required by other REMS programs” for teratogenic drugs, she added.
Dr. Graber said it’s important to note that in the time the pandemic exceptions have been made for isotretinoin patients, “there has been no reported spike in pregnancy in the past three years.
“We do have some data to show that it is not imposing additional harms,” she said.
Suggestions for improvement
At the end of the hearing, advisory committee members were asked to propose improvements to the iPLEDGE REMS program.
Dr. Green advocated for the addition of an iPLEDGE mobile app.
“Most people go to their phones rather than their computers, particularly teenagers and younger people,” he noted.
Advisory committee member Megha M. Tollefson, MD, professor of dermatology and pediatric and adolescent medicine at Mayo Clinic in Rochester, Minn., echoed the need for an iPLEDGE app.
The young patients getting isotretinoin “don’t respond to email, they don’t necessarily go onto web pages. If we’re going to be as effective as possible, it’s going to have to be through an app-based system.”
Dr. Tollefson said she would like to see patient counseling standardized through the app. “I think there’s a lot of variability in what counseling is given when it’s left to the individual prescriber or practice,” she said.
Exceptions for long-acting contraceptives?
Advisory committee member Abbey B. Berenson, MD, PhD, professor of obstetrics and gynecology at University of Texas Medical Branch in Galveston, said that patients taking long-acting reversible contraceptives (LARCs) may need to be considered differently when deciding the intervals for attestation or whether to have a lockout period.
“LARC methods’ rate of failure is extremely low,” she said. “While it is true, as it has been pointed out, that all methods can fail, when they’re over 99% effective, I think that we can treat those methods differently than we treat methods such as birth control pills or abstinence that fail far more often. That is one way we could minimize burden on the providers and the patients.”
She also suggested using members of the health care team other than physicians to complete counseling, such as a nurse or pharmacist.
Prescriptions for emergency contraception
Advisory committee member Sascha Dublin, MD, PhD, senior scientific investigator for Kaiser Permanente Washington Health Research Institute in Seattle, said most patients taking the drug who can get pregnant should get a prescription for emergency contraception at the time of the first isotretinoin prescription.
“They don’t have to buy it, but to make it available at the very beginning sets the expectation that it would be good to have in your medicine cabinet, particularly if the [contraception] choice is abstinence or birth control pills.”
Dr. Dublin also called for better transparency surrounding the role of IPMG.
She said IPMG should be expected to collect data in a way that allows examination of health disparities, including by race and ethnicity and insurance status. Dr. Dublin added that she was concerned about the poor communication between dermatological societies and IPMG.
“The FDA should really require that IPMG hold periodic, regularly scheduled stakeholder forums,” she said. “There has to be a mechanism in place for IPMG to listen to those concerns in real time and respond.”
The advisory committees’ recommendations to the FDA are nonbinding, but the FDA generally follows the recommendations of advisory panels.
During 2 days of after the chaotic rollout of the new REMS platform at the end of 2021.
On March 29, at the end of the FDA’s joint meeting of two advisory committees that addressed ways to improve the iPLEDGE program, most panelists voted to change the 19-day lockout period for patients who can become pregnant, and the requirement that every month, providers must document counseling of those who cannot get pregnant and are taking the drug for acne.
However, there was no consensus on whether there should be a lockout at all or for how long, and what an appropriate interval for counseling those who cannot get pregnant would be, if not monthly. Those voting on the questions repeatedly cited a lack of data to make well-informed decisions.
The meeting of the two panels, the FDA’s Drug Safety and Risk Management Advisory Committee and the Dermatologic and Ophthalmic Drugs Advisory Committee, was held March 28-29, to discuss proposed changes to iPLEDGE requirements, to minimize the program’s burden on patients, prescribers, and pharmacies – while maintaining safe use of the highly teratogenic drug.
Lockout based on outdated reasoning
John S. Barbieri, MD, a dermatologist and epidemiologist, and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital in Boston, speaking as deputy chair of the American Academy of Dermatology Association (AADA) iPLEDGE work group, described the burden of getting the drug to patients. He was not on the panel, but spoke during the open public hearing.
“Compared to other acne medications, the time it takes to successfully go from prescribed (isotretinoin) to when the patient actually has it in their hands is 5- to 10-fold higher,” he said.
Among the barriers is the 19-day lockout period for people who can get pregnant and miss the 7-day window for picking up their prescriptions. They must then wait 19 days to get a pregnancy test to clear them for receiving the medication.
Gregory Wedin, PharmD, pharmacovigilance and risk management director of Upsher-Smith Laboratories, who spoke on behalf of the Isotretinoin Products Manufacturer Group (IPMG), which manages iPLEDGE, said, “The rationale for the 19-day wait is to ensure the next confirmatory pregnancy test is completed after the most fertile period of the menstrual cycle is passed.”
Many don’t have a monthly cycle
But Dr. Barbieri said that reasoning is outdated.
“The current program’s focus on the menstrual cycle is really an antiquated approach,” he said. “Many patients do not have a monthly cycle due to medical conditions like polycystic ovarian syndrome, or due to [certain kinds of] contraception.”
He added, “By removing this 19-day lockout and, really, the archaic timing around the menstrual cycle in general in this program, we can simplify the program, improve it, and better align it with the real-world biology of our patients.” He added that patients are often missing the 7-day window for picking up their prescriptions through no fault of their own. Speakers at the hearing also mentioned insurance hassles and ordering delays.
Communication with IPMG
Ilona Frieden, MD, professor of dermatology and pediatrics at the University of California, San Francisco, and outgoing chair of the AADA iPLEDGE work group, cited difficulty in working with IPMG on modifications as another barrier. She also spoke during the open public hearing.
“Despite many, many attempts to work with the IPMG, we are not aware of any organizational structure or key leaders to communicate with. Instead we have been given repeatedly a generic email address for trying to establish a working relationship and we believe this may explain the inaction of the IPMG since our proposals 4 years ago in 2019.”
Among those proposals, she said, were allowing telemedicine visits as part of the iPLEDGE REMS program and reducing counseling attestation to every 6 months instead of monthly for those who cannot become pregnant.
She pointed to the chaotic rollout of modifications to the iPLEDGE program on a new website at the end of 2021.
In 2021, she said, “despite 6 months of notification, no prescriber input was solicited before revamping the website. This lack of transparency and accountability has been a major hurdle in improving iPLEDGE.”
Dr. Barbieri called the rollout “a debacle” that could have been mitigated with communication with IPMG. “We warned about every issue that happened and talked about ways to mitigate it and were largely ignored,” he said.
“By including dermatologists and key stakeholders in these discussions, as we move forward with changes to improve this program, we can make sure that it’s patient-centered.”
IPMG did not address the specific complaints about the working relationship with the AADA workgroup at the meeting.
Monthly attestation for counseling patients who cannot get pregnant
Dr. Barbieri said the monthly requirement to counsel patients who cannot get pregnant and document that counseling unfairly burdens clinicians and patients. “We’re essentially asking patients to come in monthly just to tell them not to share their drugs [or] donate blood,” he said.
Ken Katz, MD, MSc, a dermatologist at Kaiser Permanente in San Francisco, was among the panel members voting not to continue the 19-day lockout.
“I think this places an unduly high burden physically and psychologically on our patients. It seems arbitrary,” he said. “Likely we will miss some pregnancies; we are missing some already. But the burden is not matched by the benefit.”
IPMG representative Dr. Wedin, said, “while we cannot support eliminating or extending the confirmation interval to a year, the [iPLEDGE] sponsors are agreeable [to] a 120-day confirmation interval.”
He said that while an extension to 120 days would reduce burden on prescribers, it comes with the risk in reducing oversight by a certified iPLEDGE prescriber and potentially increasing the risk for drug sharing.
“A patient may be more likely to share their drug with another person the further along with therapy they get as their condition improves,” Dr. Wedin said.
Home pregnancy testing
The advisory groups were also tasked with discussing whether home pregnancy tests, allowed during the COVID-19 public health emergency, should continue to be allowed. Most committee members and those in the public hearing who spoke on the issue agreed that home tests should continue in an effort to increase access and decrease burden.
During the pandemic, iPLEDGE rules have been relaxed from having a pregnancy test done only at a Clinical Laboratory Improvement Amendments–certified laboratory.
Lindsey Crist, PharmD, a risk management analyst at the FDA, who presented the FDA review committee’s analysis, said that the FDA’s review committee recommends ending the allowance of home tests, citing insufficient data on use and the discovery of instances of falsification of pregnancy tests.
“One study at an academic medical center reviewed the medical records of 89 patients who used home pregnancy tests while taking isotretinoin during the public health emergency. It found that 15.7% submitted falsified pregnancy test results,” Dr. Crist said.
Dr. Crist added, however, that the review committee recommends allowing the tests to be done in a provider’s office as an alternative.
Workaround to avoid falsification
Advisory committee member Brian P. Green, DO, associate professor of dermatology at Penn State University, Hershey, Pa., spoke in support of home pregnancy tests.
“What we have people do for telemedicine is take the stick, write their name, write the date on it, and send a picture of that the same day as their visit,” he said. “That way we have the pregnancy test the same day. Allowing this to continue to happen at home is important. Bringing people in is burdensome and costly.”
Emmy Graber, MD, a dermatologist who practices in Boston, and a director of the American Acne and Rosacea Society (AARS), relayed an example of the burden for a patient using isotretinoin who lives 1.5 hours away from the dermatology office. She is able to meet the requirements of iPLEDGE only through telehealth.
“Home pregnancy tests are highly sensitive, equal to the ones done in CLIA-certified labs, and highly accurate when interpreted by a dermatology provider,” said Dr. Graber, who spoke on behalf of the AARS during the open public hearing.
“Notably, CLIA [Clinical Laboratory Improvement Amendments] certification is not required by other REMS programs” for teratogenic drugs, she added.
Dr. Graber said it’s important to note that in the time the pandemic exceptions have been made for isotretinoin patients, “there has been no reported spike in pregnancy in the past three years.
“We do have some data to show that it is not imposing additional harms,” she said.
Suggestions for improvement
At the end of the hearing, advisory committee members were asked to propose improvements to the iPLEDGE REMS program.
Dr. Green advocated for the addition of an iPLEDGE mobile app.
“Most people go to their phones rather than their computers, particularly teenagers and younger people,” he noted.
Advisory committee member Megha M. Tollefson, MD, professor of dermatology and pediatric and adolescent medicine at Mayo Clinic in Rochester, Minn., echoed the need for an iPLEDGE app.
The young patients getting isotretinoin “don’t respond to email, they don’t necessarily go onto web pages. If we’re going to be as effective as possible, it’s going to have to be through an app-based system.”
Dr. Tollefson said she would like to see patient counseling standardized through the app. “I think there’s a lot of variability in what counseling is given when it’s left to the individual prescriber or practice,” she said.
Exceptions for long-acting contraceptives?
Advisory committee member Abbey B. Berenson, MD, PhD, professor of obstetrics and gynecology at University of Texas Medical Branch in Galveston, said that patients taking long-acting reversible contraceptives (LARCs) may need to be considered differently when deciding the intervals for attestation or whether to have a lockout period.
“LARC methods’ rate of failure is extremely low,” she said. “While it is true, as it has been pointed out, that all methods can fail, when they’re over 99% effective, I think that we can treat those methods differently than we treat methods such as birth control pills or abstinence that fail far more often. That is one way we could minimize burden on the providers and the patients.”
She also suggested using members of the health care team other than physicians to complete counseling, such as a nurse or pharmacist.
Prescriptions for emergency contraception
Advisory committee member Sascha Dublin, MD, PhD, senior scientific investigator for Kaiser Permanente Washington Health Research Institute in Seattle, said most patients taking the drug who can get pregnant should get a prescription for emergency contraception at the time of the first isotretinoin prescription.
“They don’t have to buy it, but to make it available at the very beginning sets the expectation that it would be good to have in your medicine cabinet, particularly if the [contraception] choice is abstinence or birth control pills.”
Dr. Dublin also called for better transparency surrounding the role of IPMG.
She said IPMG should be expected to collect data in a way that allows examination of health disparities, including by race and ethnicity and insurance status. Dr. Dublin added that she was concerned about the poor communication between dermatological societies and IPMG.
“The FDA should really require that IPMG hold periodic, regularly scheduled stakeholder forums,” she said. “There has to be a mechanism in place for IPMG to listen to those concerns in real time and respond.”
The advisory committees’ recommendations to the FDA are nonbinding, but the FDA generally follows the recommendations of advisory panels.
During 2 days of after the chaotic rollout of the new REMS platform at the end of 2021.
On March 29, at the end of the FDA’s joint meeting of two advisory committees that addressed ways to improve the iPLEDGE program, most panelists voted to change the 19-day lockout period for patients who can become pregnant, and the requirement that every month, providers must document counseling of those who cannot get pregnant and are taking the drug for acne.
However, there was no consensus on whether there should be a lockout at all or for how long, and what an appropriate interval for counseling those who cannot get pregnant would be, if not monthly. Those voting on the questions repeatedly cited a lack of data to make well-informed decisions.
The meeting of the two panels, the FDA’s Drug Safety and Risk Management Advisory Committee and the Dermatologic and Ophthalmic Drugs Advisory Committee, was held March 28-29, to discuss proposed changes to iPLEDGE requirements, to minimize the program’s burden on patients, prescribers, and pharmacies – while maintaining safe use of the highly teratogenic drug.
Lockout based on outdated reasoning
John S. Barbieri, MD, a dermatologist and epidemiologist, and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital in Boston, speaking as deputy chair of the American Academy of Dermatology Association (AADA) iPLEDGE work group, described the burden of getting the drug to patients. He was not on the panel, but spoke during the open public hearing.
“Compared to other acne medications, the time it takes to successfully go from prescribed (isotretinoin) to when the patient actually has it in their hands is 5- to 10-fold higher,” he said.
Among the barriers is the 19-day lockout period for people who can get pregnant and miss the 7-day window for picking up their prescriptions. They must then wait 19 days to get a pregnancy test to clear them for receiving the medication.
Gregory Wedin, PharmD, pharmacovigilance and risk management director of Upsher-Smith Laboratories, who spoke on behalf of the Isotretinoin Products Manufacturer Group (IPMG), which manages iPLEDGE, said, “The rationale for the 19-day wait is to ensure the next confirmatory pregnancy test is completed after the most fertile period of the menstrual cycle is passed.”
Many don’t have a monthly cycle
But Dr. Barbieri said that reasoning is outdated.
“The current program’s focus on the menstrual cycle is really an antiquated approach,” he said. “Many patients do not have a monthly cycle due to medical conditions like polycystic ovarian syndrome, or due to [certain kinds of] contraception.”
He added, “By removing this 19-day lockout and, really, the archaic timing around the menstrual cycle in general in this program, we can simplify the program, improve it, and better align it with the real-world biology of our patients.” He added that patients are often missing the 7-day window for picking up their prescriptions through no fault of their own. Speakers at the hearing also mentioned insurance hassles and ordering delays.
Communication with IPMG
Ilona Frieden, MD, professor of dermatology and pediatrics at the University of California, San Francisco, and outgoing chair of the AADA iPLEDGE work group, cited difficulty in working with IPMG on modifications as another barrier. She also spoke during the open public hearing.
“Despite many, many attempts to work with the IPMG, we are not aware of any organizational structure or key leaders to communicate with. Instead we have been given repeatedly a generic email address for trying to establish a working relationship and we believe this may explain the inaction of the IPMG since our proposals 4 years ago in 2019.”
Among those proposals, she said, were allowing telemedicine visits as part of the iPLEDGE REMS program and reducing counseling attestation to every 6 months instead of monthly for those who cannot become pregnant.
She pointed to the chaotic rollout of modifications to the iPLEDGE program on a new website at the end of 2021.
In 2021, she said, “despite 6 months of notification, no prescriber input was solicited before revamping the website. This lack of transparency and accountability has been a major hurdle in improving iPLEDGE.”
Dr. Barbieri called the rollout “a debacle” that could have been mitigated with communication with IPMG. “We warned about every issue that happened and talked about ways to mitigate it and were largely ignored,” he said.
“By including dermatologists and key stakeholders in these discussions, as we move forward with changes to improve this program, we can make sure that it’s patient-centered.”
IPMG did not address the specific complaints about the working relationship with the AADA workgroup at the meeting.
Monthly attestation for counseling patients who cannot get pregnant
Dr. Barbieri said the monthly requirement to counsel patients who cannot get pregnant and document that counseling unfairly burdens clinicians and patients. “We’re essentially asking patients to come in monthly just to tell them not to share their drugs [or] donate blood,” he said.
Ken Katz, MD, MSc, a dermatologist at Kaiser Permanente in San Francisco, was among the panel members voting not to continue the 19-day lockout.
“I think this places an unduly high burden physically and psychologically on our patients. It seems arbitrary,” he said. “Likely we will miss some pregnancies; we are missing some already. But the burden is not matched by the benefit.”
IPMG representative Dr. Wedin, said, “while we cannot support eliminating or extending the confirmation interval to a year, the [iPLEDGE] sponsors are agreeable [to] a 120-day confirmation interval.”
He said that while an extension to 120 days would reduce burden on prescribers, it comes with the risk in reducing oversight by a certified iPLEDGE prescriber and potentially increasing the risk for drug sharing.
“A patient may be more likely to share their drug with another person the further along with therapy they get as their condition improves,” Dr. Wedin said.
Home pregnancy testing
The advisory groups were also tasked with discussing whether home pregnancy tests, allowed during the COVID-19 public health emergency, should continue to be allowed. Most committee members and those in the public hearing who spoke on the issue agreed that home tests should continue in an effort to increase access and decrease burden.
During the pandemic, iPLEDGE rules have been relaxed from having a pregnancy test done only at a Clinical Laboratory Improvement Amendments–certified laboratory.
Lindsey Crist, PharmD, a risk management analyst at the FDA, who presented the FDA review committee’s analysis, said that the FDA’s review committee recommends ending the allowance of home tests, citing insufficient data on use and the discovery of instances of falsification of pregnancy tests.
“One study at an academic medical center reviewed the medical records of 89 patients who used home pregnancy tests while taking isotretinoin during the public health emergency. It found that 15.7% submitted falsified pregnancy test results,” Dr. Crist said.
Dr. Crist added, however, that the review committee recommends allowing the tests to be done in a provider’s office as an alternative.
Workaround to avoid falsification
Advisory committee member Brian P. Green, DO, associate professor of dermatology at Penn State University, Hershey, Pa., spoke in support of home pregnancy tests.
“What we have people do for telemedicine is take the stick, write their name, write the date on it, and send a picture of that the same day as their visit,” he said. “That way we have the pregnancy test the same day. Allowing this to continue to happen at home is important. Bringing people in is burdensome and costly.”
Emmy Graber, MD, a dermatologist who practices in Boston, and a director of the American Acne and Rosacea Society (AARS), relayed an example of the burden for a patient using isotretinoin who lives 1.5 hours away from the dermatology office. She is able to meet the requirements of iPLEDGE only through telehealth.
“Home pregnancy tests are highly sensitive, equal to the ones done in CLIA-certified labs, and highly accurate when interpreted by a dermatology provider,” said Dr. Graber, who spoke on behalf of the AARS during the open public hearing.
“Notably, CLIA [Clinical Laboratory Improvement Amendments] certification is not required by other REMS programs” for teratogenic drugs, she added.
Dr. Graber said it’s important to note that in the time the pandemic exceptions have been made for isotretinoin patients, “there has been no reported spike in pregnancy in the past three years.
“We do have some data to show that it is not imposing additional harms,” she said.
Suggestions for improvement
At the end of the hearing, advisory committee members were asked to propose improvements to the iPLEDGE REMS program.
Dr. Green advocated for the addition of an iPLEDGE mobile app.
“Most people go to their phones rather than their computers, particularly teenagers and younger people,” he noted.
Advisory committee member Megha M. Tollefson, MD, professor of dermatology and pediatric and adolescent medicine at Mayo Clinic in Rochester, Minn., echoed the need for an iPLEDGE app.
The young patients getting isotretinoin “don’t respond to email, they don’t necessarily go onto web pages. If we’re going to be as effective as possible, it’s going to have to be through an app-based system.”
Dr. Tollefson said she would like to see patient counseling standardized through the app. “I think there’s a lot of variability in what counseling is given when it’s left to the individual prescriber or practice,” she said.
Exceptions for long-acting contraceptives?
Advisory committee member Abbey B. Berenson, MD, PhD, professor of obstetrics and gynecology at University of Texas Medical Branch in Galveston, said that patients taking long-acting reversible contraceptives (LARCs) may need to be considered differently when deciding the intervals for attestation or whether to have a lockout period.
“LARC methods’ rate of failure is extremely low,” she said. “While it is true, as it has been pointed out, that all methods can fail, when they’re over 99% effective, I think that we can treat those methods differently than we treat methods such as birth control pills or abstinence that fail far more often. That is one way we could minimize burden on the providers and the patients.”
She also suggested using members of the health care team other than physicians to complete counseling, such as a nurse or pharmacist.
Prescriptions for emergency contraception
Advisory committee member Sascha Dublin, MD, PhD, senior scientific investigator for Kaiser Permanente Washington Health Research Institute in Seattle, said most patients taking the drug who can get pregnant should get a prescription for emergency contraception at the time of the first isotretinoin prescription.
“They don’t have to buy it, but to make it available at the very beginning sets the expectation that it would be good to have in your medicine cabinet, particularly if the [contraception] choice is abstinence or birth control pills.”
Dr. Dublin also called for better transparency surrounding the role of IPMG.
She said IPMG should be expected to collect data in a way that allows examination of health disparities, including by race and ethnicity and insurance status. Dr. Dublin added that she was concerned about the poor communication between dermatological societies and IPMG.
“The FDA should really require that IPMG hold periodic, regularly scheduled stakeholder forums,” she said. “There has to be a mechanism in place for IPMG to listen to those concerns in real time and respond.”
The advisory committees’ recommendations to the FDA are nonbinding, but the FDA generally follows the recommendations of advisory panels.
Biosimilars and patients: Discussions should address safety, cost, and anxiety about change
Rheumatologist Marcus Snow, MD, is comfortable with prescribing biosimilars as a first-line, first-time biologic, and discussing them with patients.
“If a biosimilar is on the market, it has gone through rigorous study proving its effectiveness and equivalence to a bio-originator,” said Dr. Snow, a rheumatologist with the University of Nebraska Medical Center, Omaha, and chair of the American College of Rheumatology’s Committee on Rheumatologic Care.
The formulary makes a big difference in the conversation about options, he said. “The formularies dictate what we can prescribe. It may not be appropriate, but it is reality. The cost of biologics for a patient without insurance coverage makes it impossible to afford.”
He will often tell patients that he’ll fight any changes or formulary restrictions he does not agree with. “However, when I see patients in follow-up, even if there is no known change on the horizon, I may bring up biosimilars when we have a moment to chat about them to familiarize them with what may happen in the future.”
The need for patient education on biosimilars presents a barrier to realizing their potential to save money and expand choice, noted Cardinal Health in its 2023 biosimilars report. Of 103 rheumatologists who responded to a Cardinal Health survey, 85% agreed that patient education was important. But those conversations can take an uncomfortable turn if the patient pushes back against taking a biosimilar owing to cost or safety concerns.
It’s not uncommon for a patient to express some anxiety about biosimilars, especially if they’re doing well on a current treatment plan. Most patients do not want any changes that may lead to worsening disease control, Dr. Snow said.
Patients and physicians alike often don’t understand the mechanics of biosimilars. “There’s a lot of misinformation about this,” said Sameer Awsare, MD, an associate executive director for The Permanente Medical Group in Campbell, Calif. Patients should know that a biosimilar will be as clinically efficacious as the medicine they’ve been on, with the same safety profiles, said Dr. Awsare, who works with Kaiser Permanente’s pharmacy partners on biosimilars.
Insurance often drives the conversation
The global anti-inflammatory biologics market is anticipated to reach $150 billion by 2027, according to a recent CVS report. As of March 2023, the Food and Drug Administration had approved 40 biosimilars to 11 different reference products. There are 28 on the U.S. market and 100 more in development. Projected to save more than $180 billion over the next 5 years, they are anticipated to expand choice and drive competition.
Rheumatologists, dermatologists, and gastroenterologists are frequent prescribers, although their choices for immune-mediated inflammatory diseases are limited to tumor necrosis factor inhibitors (infliximab [Remicade] originator and adalimumab [Humira] originator) and anti-CD20 agents, such as rituximab (Rituxan) originator.
Benefit design or formulary usually dictates what medicine a patient receives. “Because of significantly higher out-of-pocket cost or formulary positioning, patients may end up with a generic or a biosimilar instead of a brand-name medicine or branded biologic,” said Robert Popovian, PharmD, MS, chief science policy officer of the Global Healthy Living Foundation.
Insurers rarely offer both Remicade and biosimilar infliximab, allowing the doctor to choose, said Miguel Regueiro, MD, chair of the Cleveland Clinic’s Digestive Disease & Surgery Institute, who prescribes infliximab biosimilars. Most often, the payer will choose the lower-cost biosimilar. “I am fine with the biosimilar, either as a new start or a switch from the reference product.”
However, the patient might feel differently. They can form an attachment to the reference medication if it has prevented severe illness. “They do not want to change, as they feel they are going on a ‘new’ medication that will not work as well,” Dr. Regueiro said.
This is where the education comes in: to reassure patients that a biosimilar will work just as well as the reference product. “For patients who have done well for years on a biologic, more time needs to be spent reassuring them and answering questions,” compared with a patient just starting on a biosimilar, he advised.
But not all physicians are quick to prescribe biosimilars.
Especially with psoriasis, which has so many strong options for reference drugs, a switch may be hard to justify, said dermatologist Stephanie K. Fabbro, MD, assistant professor at Northeast Ohio Medical University, Rootstown. “If I have a preference, I would rather switch a patient to a drug from a different class without a biosimilar option to reduce the possibility of pushback.”
Dr. Fabbro, part of the core faculty in the Riverside Methodist Hospital Dermatology Residency Program in Columbus, will share data from clinical trials and postmarket surveillance with patients to support her decision.
Conversations about cost
Patients may also push back if they don’t save money when switching to a biosimilar. “This dilemma raises the question of who is profiting when a biosimilar is dispensed,” Dr. Popovian said. Insurers and pharmacy benefit managers (PBMs) that take additional concessions from biopharmaceutical manufacturers in the form of rebates and fees will often pocket this money as profit instead of passing savings back to the patient to help reduce their out-of-pocket requirement, he added.
If an originator biologic and a biosimilar are available, “as a pharmacist, I will choose the medicine that will incur the lowest out-of-pocket cost for the patient,” Dr. Popovian said.
Discussing cost – and who dictates which biosimilar is on the formulary – is an important conversation to have with patients, said Vivek Kaul, MD, Segal-Watson Professor of Medicine at the University of Rochester (N.Y.) Medical Center.
Providing equivalent clinical efficacy while saving costs is the economic reality of biosimilars, Dr. Kaul said. Third-party payers regularly evaluate how to provide the same quality of care while saving money. Physicians and patients alike “must be mindful that as time goes on, if the science on biosimilars stays robust, if the adoption is more widespread and the cost-saving proposition turns out to be true, more formularies will be attracted to replacing the reference product with the biosimilar counterpart.”
Providers and patients can weigh the options if a formulary suddenly switches to a biosimilar, Dr. Kaul continued. “You can accept the novel product on the formulary or may have to face out-of-pocket expenses as a patient.” If providers and patients have concerns about the biosimilar, they can always appeal if there’s solid scientific evidence that supports reverting back to the reference product.
“If you think the biosimilar is equally efficacious, comes at a lower cost, and is right for the patient, then the providers should tell the patient that,” he added.
Some studies have questioned whether the biosimilars will save money, compared with the reference drug, Dr. Fabbro noted. Medicare, for example, may pay only for a certain percentage of an approved biosimilar, saddling the patient with a monthly copay costing thousands of dollars. “It is unclear whether biosimilar manufacturers will have the same level of patient support programs as the reference drug companies.”
For that reason, physicians should also inform patients about the robust patient assistance and copay assistance programs many reference drug manufacturers offer, she said.
Biosimilars 101: Familiarizing patients
Safety and ease of use are other common concerns about biosimilars. Patients may ask if the application is different, or why it’s advantageous to switch to a biosimilar, Dr. Awsare said.
Sometimes the syringe or injector for a biosimilar might look different from that of the originator drug, he said.
Anecdotally, Dr. Fabbro has heard stories of patients having injection reactions that they did not experience with the reference drug or having a disease flare-up after starting a biosimilar.
As is the case with reference products, in their conversations with patients, clinicians should address the adverse event profile of biosimilars, offering data points from published studies and clinical guidelines that support the use of these products. “There should be an emphasis on patient education around efficacy and any side effects, and how the profile of the reference product compares with a proposed biosimilar,” Dr. Kaul suggested.
When Dr. Snow discusses biosimilars and generics, “I make sure to share this in an understandable way based on the patient’s scientific background, or lack thereof,” he said. If there is enough time, he also discusses how European- and U.S.-sourced biologics are slightly different.
Pharmacists should tell patients to expect the same clinical outcomes from a biosimilar, Dr. Popovian said. However, if they have any reduction in efficacy or potential safety concerns, they should communicate with their physician or pharmacist immediately.
In Dr. Regueiro’s practice, a pharmacist specializing in inflammatory bowel disease often has a one-on-one meeting with patients to educate and answer questions. “Additionally, we provide them the Crohn’s and Colitis Foundation web link on biosimilars,” said Dr. Regueiro.
A village approach to education
When biosimilars first came out, there were no formal education materials, Dr. Awsare said. Kaiser Permanente decided to create its own educational materials, not just for patients but also to help educate its primary care doctors; the rheumatologists, dermatologists, and gastroenterologists using the biosimilars; the nurses infusing patients; and the pharmacists preparing the biosimilars.
The health system also has a different approach to choosing medication. Instead of having an insurance company or PBM decide what’s in the formulary, clinicians work with the pharmacists at Kaiser to look at clinical evidence and decide which biosimilar to use. Most of its plans also provide lower copays to patients when they use the biosimilar.
This was the approach for Humira biosimilars, Dr. Awsare said. Eight will be on the market in 2023. “Our rheumatologists, dermatologists, and gastroenterologists looked at the data from Europe, looked at some real-world evidence, and then said: ‘We think this one’s going to be the best one for our patients.’ ”
Having clinicians choose the biosimilar instead of a health plan makes it a lot easier to have conversations with patients, he said. “Once we’ve moved that market share to that particular biosimilar, we give our physicians the time to have those discussions.”
Clinical pharmacists also provide educational support, offering guidance on issues such as side effects, as patients transition to the biosimilar. “We like to use the word ‘transition’ because it’s essentially the same biologic. So, you’re not actually switching,” Dr. Awsare said.
No consensus on interchangeability
Whether the conversation on interchangeability will affect patient conversations with physicians depends on who you ask.
If a biosimilar has an interchangeability designation, it means that the pharmacist can substitute it without the intervention of the clinician who prescribed the reference product. It does not relate to the quality, safety, or effectiveness of biosimilars or interchangeable biosimilar products, Dr. Popovian said.
The United States is the only country that has this designation. Even though it’s not identical to the originator drug, a biosimilar has the same clinical efficacy and safety profile. “So clinically, interchangeability is meaningless,” Dr. Awsare said.
In its report on biosimilars in the autoimmune category, CVS acknowledged that interchangeability was important but would not be a significant factor in driving adoption of biosimilars. However, in a Cardinal Health survey of 72 gastroenterologists, 38% cited the interchangeability of biosimilars as a top concern for adalimumab biosimilars, along with transitioning patients from Humira to a biosimilar (44%).
“Patient education regarding biosimilar safety, efficacy, and interchangeability appears paramount to the acceptance of these products, particularly for patients who are switched from a reference product,” Dr. Kaul noted in the Cardinal Health report.
Wherever supported by data, Dr. Kaul recommends incorporating biosimilar use and interchangeability into best practice guidelines going forward. “That will go a long way in disseminating the latest information on this topic and position this paradigm for increased adoption among providers.”
Some physicians like Dr. Snow aren’t that concerned with interchangeability. This hasn’t affected conversations with patients, he said. Multiple studies demonstrating the lack of antibody formation with multiple switches from different biosimilar drugs has eased his concern about multiple switches causing problems.
“Initially, there was a gap in demonstrating the long-term effect of multiple switches on antibody production and drug effectiveness. That gap has started to close as more data from Europe’s experience with biosimilars becomes available,” Dr. Snow said.
Resources for physicians, patients
The federal government has taken steps to advance biosimilars education and adoption. In 2021, President Biden signed the Advancing Education on Biosimilars Act into law, which directs the FDA to develop or improve continuing education programs that address prescribing of biosimilars and biological products.
The FDA provides educational materials on its website, including a comprehensive curriculum toolkit. The Accreditation Council for Medical Affairs has also created an online 40-hour curriculum for health care professionals called the Board-Certified Biologics and Biosimilars Specialist Program.
Dr. Fabbro recommended patients use the FDA page Biosimilar Basics for Patients to educate themselves on biosimilars. The Global Healthy Living Foundation’s podcast, Breaking Down Biosimilars, is another free resource for patients.
“While much has changed, the continued need for multistakeholder education, awareness, and dedicated research remains even more important as we expand into newer therapeutic areas and classes,” wrote the authors of the Cardinal Health report.
Help patients understand biologics and biosimilars by using AGA resources for providers and patients available at gastro.org/biosimilars.
Dr. Regueiro is on advisory boards and consults for AbbVie, Janssen, UCB, Takeda, Pfizer, Bristol-Myers Squibb, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, TARGET PharmaSolutions, Trellis, and Boehringer Ingelheim. Dr. Fabbro is a principal investigator for Castle Biosciences, on the speakers bureau for Valchlor, and on the advisory boards of Janssen and Bristol-Myers Squibb. Dr. Popovian, Dr. Snow, Dr. Awsare, and Dr. Kaul had no disclosures.
A version of this article originally appeared on Medscape.com.
Rheumatologist Marcus Snow, MD, is comfortable with prescribing biosimilars as a first-line, first-time biologic, and discussing them with patients.
“If a biosimilar is on the market, it has gone through rigorous study proving its effectiveness and equivalence to a bio-originator,” said Dr. Snow, a rheumatologist with the University of Nebraska Medical Center, Omaha, and chair of the American College of Rheumatology’s Committee on Rheumatologic Care.
The formulary makes a big difference in the conversation about options, he said. “The formularies dictate what we can prescribe. It may not be appropriate, but it is reality. The cost of biologics for a patient without insurance coverage makes it impossible to afford.”
He will often tell patients that he’ll fight any changes or formulary restrictions he does not agree with. “However, when I see patients in follow-up, even if there is no known change on the horizon, I may bring up biosimilars when we have a moment to chat about them to familiarize them with what may happen in the future.”
The need for patient education on biosimilars presents a barrier to realizing their potential to save money and expand choice, noted Cardinal Health in its 2023 biosimilars report. Of 103 rheumatologists who responded to a Cardinal Health survey, 85% agreed that patient education was important. But those conversations can take an uncomfortable turn if the patient pushes back against taking a biosimilar owing to cost or safety concerns.
It’s not uncommon for a patient to express some anxiety about biosimilars, especially if they’re doing well on a current treatment plan. Most patients do not want any changes that may lead to worsening disease control, Dr. Snow said.
Patients and physicians alike often don’t understand the mechanics of biosimilars. “There’s a lot of misinformation about this,” said Sameer Awsare, MD, an associate executive director for The Permanente Medical Group in Campbell, Calif. Patients should know that a biosimilar will be as clinically efficacious as the medicine they’ve been on, with the same safety profiles, said Dr. Awsare, who works with Kaiser Permanente’s pharmacy partners on biosimilars.
Insurance often drives the conversation
The global anti-inflammatory biologics market is anticipated to reach $150 billion by 2027, according to a recent CVS report. As of March 2023, the Food and Drug Administration had approved 40 biosimilars to 11 different reference products. There are 28 on the U.S. market and 100 more in development. Projected to save more than $180 billion over the next 5 years, they are anticipated to expand choice and drive competition.
Rheumatologists, dermatologists, and gastroenterologists are frequent prescribers, although their choices for immune-mediated inflammatory diseases are limited to tumor necrosis factor inhibitors (infliximab [Remicade] originator and adalimumab [Humira] originator) and anti-CD20 agents, such as rituximab (Rituxan) originator.
Benefit design or formulary usually dictates what medicine a patient receives. “Because of significantly higher out-of-pocket cost or formulary positioning, patients may end up with a generic or a biosimilar instead of a brand-name medicine or branded biologic,” said Robert Popovian, PharmD, MS, chief science policy officer of the Global Healthy Living Foundation.
Insurers rarely offer both Remicade and biosimilar infliximab, allowing the doctor to choose, said Miguel Regueiro, MD, chair of the Cleveland Clinic’s Digestive Disease & Surgery Institute, who prescribes infliximab biosimilars. Most often, the payer will choose the lower-cost biosimilar. “I am fine with the biosimilar, either as a new start or a switch from the reference product.”
However, the patient might feel differently. They can form an attachment to the reference medication if it has prevented severe illness. “They do not want to change, as they feel they are going on a ‘new’ medication that will not work as well,” Dr. Regueiro said.
This is where the education comes in: to reassure patients that a biosimilar will work just as well as the reference product. “For patients who have done well for years on a biologic, more time needs to be spent reassuring them and answering questions,” compared with a patient just starting on a biosimilar, he advised.
But not all physicians are quick to prescribe biosimilars.
Especially with psoriasis, which has so many strong options for reference drugs, a switch may be hard to justify, said dermatologist Stephanie K. Fabbro, MD, assistant professor at Northeast Ohio Medical University, Rootstown. “If I have a preference, I would rather switch a patient to a drug from a different class without a biosimilar option to reduce the possibility of pushback.”
Dr. Fabbro, part of the core faculty in the Riverside Methodist Hospital Dermatology Residency Program in Columbus, will share data from clinical trials and postmarket surveillance with patients to support her decision.
Conversations about cost
Patients may also push back if they don’t save money when switching to a biosimilar. “This dilemma raises the question of who is profiting when a biosimilar is dispensed,” Dr. Popovian said. Insurers and pharmacy benefit managers (PBMs) that take additional concessions from biopharmaceutical manufacturers in the form of rebates and fees will often pocket this money as profit instead of passing savings back to the patient to help reduce their out-of-pocket requirement, he added.
If an originator biologic and a biosimilar are available, “as a pharmacist, I will choose the medicine that will incur the lowest out-of-pocket cost for the patient,” Dr. Popovian said.
Discussing cost – and who dictates which biosimilar is on the formulary – is an important conversation to have with patients, said Vivek Kaul, MD, Segal-Watson Professor of Medicine at the University of Rochester (N.Y.) Medical Center.
Providing equivalent clinical efficacy while saving costs is the economic reality of biosimilars, Dr. Kaul said. Third-party payers regularly evaluate how to provide the same quality of care while saving money. Physicians and patients alike “must be mindful that as time goes on, if the science on biosimilars stays robust, if the adoption is more widespread and the cost-saving proposition turns out to be true, more formularies will be attracted to replacing the reference product with the biosimilar counterpart.”
Providers and patients can weigh the options if a formulary suddenly switches to a biosimilar, Dr. Kaul continued. “You can accept the novel product on the formulary or may have to face out-of-pocket expenses as a patient.” If providers and patients have concerns about the biosimilar, they can always appeal if there’s solid scientific evidence that supports reverting back to the reference product.
“If you think the biosimilar is equally efficacious, comes at a lower cost, and is right for the patient, then the providers should tell the patient that,” he added.
Some studies have questioned whether the biosimilars will save money, compared with the reference drug, Dr. Fabbro noted. Medicare, for example, may pay only for a certain percentage of an approved biosimilar, saddling the patient with a monthly copay costing thousands of dollars. “It is unclear whether biosimilar manufacturers will have the same level of patient support programs as the reference drug companies.”
For that reason, physicians should also inform patients about the robust patient assistance and copay assistance programs many reference drug manufacturers offer, she said.
Biosimilars 101: Familiarizing patients
Safety and ease of use are other common concerns about biosimilars. Patients may ask if the application is different, or why it’s advantageous to switch to a biosimilar, Dr. Awsare said.
Sometimes the syringe or injector for a biosimilar might look different from that of the originator drug, he said.
Anecdotally, Dr. Fabbro has heard stories of patients having injection reactions that they did not experience with the reference drug or having a disease flare-up after starting a biosimilar.
As is the case with reference products, in their conversations with patients, clinicians should address the adverse event profile of biosimilars, offering data points from published studies and clinical guidelines that support the use of these products. “There should be an emphasis on patient education around efficacy and any side effects, and how the profile of the reference product compares with a proposed biosimilar,” Dr. Kaul suggested.
When Dr. Snow discusses biosimilars and generics, “I make sure to share this in an understandable way based on the patient’s scientific background, or lack thereof,” he said. If there is enough time, he also discusses how European- and U.S.-sourced biologics are slightly different.
Pharmacists should tell patients to expect the same clinical outcomes from a biosimilar, Dr. Popovian said. However, if they have any reduction in efficacy or potential safety concerns, they should communicate with their physician or pharmacist immediately.
In Dr. Regueiro’s practice, a pharmacist specializing in inflammatory bowel disease often has a one-on-one meeting with patients to educate and answer questions. “Additionally, we provide them the Crohn’s and Colitis Foundation web link on biosimilars,” said Dr. Regueiro.
A village approach to education
When biosimilars first came out, there were no formal education materials, Dr. Awsare said. Kaiser Permanente decided to create its own educational materials, not just for patients but also to help educate its primary care doctors; the rheumatologists, dermatologists, and gastroenterologists using the biosimilars; the nurses infusing patients; and the pharmacists preparing the biosimilars.
The health system also has a different approach to choosing medication. Instead of having an insurance company or PBM decide what’s in the formulary, clinicians work with the pharmacists at Kaiser to look at clinical evidence and decide which biosimilar to use. Most of its plans also provide lower copays to patients when they use the biosimilar.
This was the approach for Humira biosimilars, Dr. Awsare said. Eight will be on the market in 2023. “Our rheumatologists, dermatologists, and gastroenterologists looked at the data from Europe, looked at some real-world evidence, and then said: ‘We think this one’s going to be the best one for our patients.’ ”
Having clinicians choose the biosimilar instead of a health plan makes it a lot easier to have conversations with patients, he said. “Once we’ve moved that market share to that particular biosimilar, we give our physicians the time to have those discussions.”
Clinical pharmacists also provide educational support, offering guidance on issues such as side effects, as patients transition to the biosimilar. “We like to use the word ‘transition’ because it’s essentially the same biologic. So, you’re not actually switching,” Dr. Awsare said.
No consensus on interchangeability
Whether the conversation on interchangeability will affect patient conversations with physicians depends on who you ask.
If a biosimilar has an interchangeability designation, it means that the pharmacist can substitute it without the intervention of the clinician who prescribed the reference product. It does not relate to the quality, safety, or effectiveness of biosimilars or interchangeable biosimilar products, Dr. Popovian said.
The United States is the only country that has this designation. Even though it’s not identical to the originator drug, a biosimilar has the same clinical efficacy and safety profile. “So clinically, interchangeability is meaningless,” Dr. Awsare said.
In its report on biosimilars in the autoimmune category, CVS acknowledged that interchangeability was important but would not be a significant factor in driving adoption of biosimilars. However, in a Cardinal Health survey of 72 gastroenterologists, 38% cited the interchangeability of biosimilars as a top concern for adalimumab biosimilars, along with transitioning patients from Humira to a biosimilar (44%).
“Patient education regarding biosimilar safety, efficacy, and interchangeability appears paramount to the acceptance of these products, particularly for patients who are switched from a reference product,” Dr. Kaul noted in the Cardinal Health report.
Wherever supported by data, Dr. Kaul recommends incorporating biosimilar use and interchangeability into best practice guidelines going forward. “That will go a long way in disseminating the latest information on this topic and position this paradigm for increased adoption among providers.”
Some physicians like Dr. Snow aren’t that concerned with interchangeability. This hasn’t affected conversations with patients, he said. Multiple studies demonstrating the lack of antibody formation with multiple switches from different biosimilar drugs has eased his concern about multiple switches causing problems.
“Initially, there was a gap in demonstrating the long-term effect of multiple switches on antibody production and drug effectiveness. That gap has started to close as more data from Europe’s experience with biosimilars becomes available,” Dr. Snow said.
Resources for physicians, patients
The federal government has taken steps to advance biosimilars education and adoption. In 2021, President Biden signed the Advancing Education on Biosimilars Act into law, which directs the FDA to develop or improve continuing education programs that address prescribing of biosimilars and biological products.
The FDA provides educational materials on its website, including a comprehensive curriculum toolkit. The Accreditation Council for Medical Affairs has also created an online 40-hour curriculum for health care professionals called the Board-Certified Biologics and Biosimilars Specialist Program.
Dr. Fabbro recommended patients use the FDA page Biosimilar Basics for Patients to educate themselves on biosimilars. The Global Healthy Living Foundation’s podcast, Breaking Down Biosimilars, is another free resource for patients.
“While much has changed, the continued need for multistakeholder education, awareness, and dedicated research remains even more important as we expand into newer therapeutic areas and classes,” wrote the authors of the Cardinal Health report.
Help patients understand biologics and biosimilars by using AGA resources for providers and patients available at gastro.org/biosimilars.
Dr. Regueiro is on advisory boards and consults for AbbVie, Janssen, UCB, Takeda, Pfizer, Bristol-Myers Squibb, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, TARGET PharmaSolutions, Trellis, and Boehringer Ingelheim. Dr. Fabbro is a principal investigator for Castle Biosciences, on the speakers bureau for Valchlor, and on the advisory boards of Janssen and Bristol-Myers Squibb. Dr. Popovian, Dr. Snow, Dr. Awsare, and Dr. Kaul had no disclosures.
A version of this article originally appeared on Medscape.com.
Rheumatologist Marcus Snow, MD, is comfortable with prescribing biosimilars as a first-line, first-time biologic, and discussing them with patients.
“If a biosimilar is on the market, it has gone through rigorous study proving its effectiveness and equivalence to a bio-originator,” said Dr. Snow, a rheumatologist with the University of Nebraska Medical Center, Omaha, and chair of the American College of Rheumatology’s Committee on Rheumatologic Care.
The formulary makes a big difference in the conversation about options, he said. “The formularies dictate what we can prescribe. It may not be appropriate, but it is reality. The cost of biologics for a patient without insurance coverage makes it impossible to afford.”
He will often tell patients that he’ll fight any changes or formulary restrictions he does not agree with. “However, when I see patients in follow-up, even if there is no known change on the horizon, I may bring up biosimilars when we have a moment to chat about them to familiarize them with what may happen in the future.”
The need for patient education on biosimilars presents a barrier to realizing their potential to save money and expand choice, noted Cardinal Health in its 2023 biosimilars report. Of 103 rheumatologists who responded to a Cardinal Health survey, 85% agreed that patient education was important. But those conversations can take an uncomfortable turn if the patient pushes back against taking a biosimilar owing to cost or safety concerns.
It’s not uncommon for a patient to express some anxiety about biosimilars, especially if they’re doing well on a current treatment plan. Most patients do not want any changes that may lead to worsening disease control, Dr. Snow said.
Patients and physicians alike often don’t understand the mechanics of biosimilars. “There’s a lot of misinformation about this,” said Sameer Awsare, MD, an associate executive director for The Permanente Medical Group in Campbell, Calif. Patients should know that a biosimilar will be as clinically efficacious as the medicine they’ve been on, with the same safety profiles, said Dr. Awsare, who works with Kaiser Permanente’s pharmacy partners on biosimilars.
Insurance often drives the conversation
The global anti-inflammatory biologics market is anticipated to reach $150 billion by 2027, according to a recent CVS report. As of March 2023, the Food and Drug Administration had approved 40 biosimilars to 11 different reference products. There are 28 on the U.S. market and 100 more in development. Projected to save more than $180 billion over the next 5 years, they are anticipated to expand choice and drive competition.
Rheumatologists, dermatologists, and gastroenterologists are frequent prescribers, although their choices for immune-mediated inflammatory diseases are limited to tumor necrosis factor inhibitors (infliximab [Remicade] originator and adalimumab [Humira] originator) and anti-CD20 agents, such as rituximab (Rituxan) originator.
Benefit design or formulary usually dictates what medicine a patient receives. “Because of significantly higher out-of-pocket cost or formulary positioning, patients may end up with a generic or a biosimilar instead of a brand-name medicine or branded biologic,” said Robert Popovian, PharmD, MS, chief science policy officer of the Global Healthy Living Foundation.
Insurers rarely offer both Remicade and biosimilar infliximab, allowing the doctor to choose, said Miguel Regueiro, MD, chair of the Cleveland Clinic’s Digestive Disease & Surgery Institute, who prescribes infliximab biosimilars. Most often, the payer will choose the lower-cost biosimilar. “I am fine with the biosimilar, either as a new start or a switch from the reference product.”
However, the patient might feel differently. They can form an attachment to the reference medication if it has prevented severe illness. “They do not want to change, as they feel they are going on a ‘new’ medication that will not work as well,” Dr. Regueiro said.
This is where the education comes in: to reassure patients that a biosimilar will work just as well as the reference product. “For patients who have done well for years on a biologic, more time needs to be spent reassuring them and answering questions,” compared with a patient just starting on a biosimilar, he advised.
But not all physicians are quick to prescribe biosimilars.
Especially with psoriasis, which has so many strong options for reference drugs, a switch may be hard to justify, said dermatologist Stephanie K. Fabbro, MD, assistant professor at Northeast Ohio Medical University, Rootstown. “If I have a preference, I would rather switch a patient to a drug from a different class without a biosimilar option to reduce the possibility of pushback.”
Dr. Fabbro, part of the core faculty in the Riverside Methodist Hospital Dermatology Residency Program in Columbus, will share data from clinical trials and postmarket surveillance with patients to support her decision.
Conversations about cost
Patients may also push back if they don’t save money when switching to a biosimilar. “This dilemma raises the question of who is profiting when a biosimilar is dispensed,” Dr. Popovian said. Insurers and pharmacy benefit managers (PBMs) that take additional concessions from biopharmaceutical manufacturers in the form of rebates and fees will often pocket this money as profit instead of passing savings back to the patient to help reduce their out-of-pocket requirement, he added.
If an originator biologic and a biosimilar are available, “as a pharmacist, I will choose the medicine that will incur the lowest out-of-pocket cost for the patient,” Dr. Popovian said.
Discussing cost – and who dictates which biosimilar is on the formulary – is an important conversation to have with patients, said Vivek Kaul, MD, Segal-Watson Professor of Medicine at the University of Rochester (N.Y.) Medical Center.
Providing equivalent clinical efficacy while saving costs is the economic reality of biosimilars, Dr. Kaul said. Third-party payers regularly evaluate how to provide the same quality of care while saving money. Physicians and patients alike “must be mindful that as time goes on, if the science on biosimilars stays robust, if the adoption is more widespread and the cost-saving proposition turns out to be true, more formularies will be attracted to replacing the reference product with the biosimilar counterpart.”
Providers and patients can weigh the options if a formulary suddenly switches to a biosimilar, Dr. Kaul continued. “You can accept the novel product on the formulary or may have to face out-of-pocket expenses as a patient.” If providers and patients have concerns about the biosimilar, they can always appeal if there’s solid scientific evidence that supports reverting back to the reference product.
“If you think the biosimilar is equally efficacious, comes at a lower cost, and is right for the patient, then the providers should tell the patient that,” he added.
Some studies have questioned whether the biosimilars will save money, compared with the reference drug, Dr. Fabbro noted. Medicare, for example, may pay only for a certain percentage of an approved biosimilar, saddling the patient with a monthly copay costing thousands of dollars. “It is unclear whether biosimilar manufacturers will have the same level of patient support programs as the reference drug companies.”
For that reason, physicians should also inform patients about the robust patient assistance and copay assistance programs many reference drug manufacturers offer, she said.
Biosimilars 101: Familiarizing patients
Safety and ease of use are other common concerns about biosimilars. Patients may ask if the application is different, or why it’s advantageous to switch to a biosimilar, Dr. Awsare said.
Sometimes the syringe or injector for a biosimilar might look different from that of the originator drug, he said.
Anecdotally, Dr. Fabbro has heard stories of patients having injection reactions that they did not experience with the reference drug or having a disease flare-up after starting a biosimilar.
As is the case with reference products, in their conversations with patients, clinicians should address the adverse event profile of biosimilars, offering data points from published studies and clinical guidelines that support the use of these products. “There should be an emphasis on patient education around efficacy and any side effects, and how the profile of the reference product compares with a proposed biosimilar,” Dr. Kaul suggested.
When Dr. Snow discusses biosimilars and generics, “I make sure to share this in an understandable way based on the patient’s scientific background, or lack thereof,” he said. If there is enough time, he also discusses how European- and U.S.-sourced biologics are slightly different.
Pharmacists should tell patients to expect the same clinical outcomes from a biosimilar, Dr. Popovian said. However, if they have any reduction in efficacy or potential safety concerns, they should communicate with their physician or pharmacist immediately.
In Dr. Regueiro’s practice, a pharmacist specializing in inflammatory bowel disease often has a one-on-one meeting with patients to educate and answer questions. “Additionally, we provide them the Crohn’s and Colitis Foundation web link on biosimilars,” said Dr. Regueiro.
A village approach to education
When biosimilars first came out, there were no formal education materials, Dr. Awsare said. Kaiser Permanente decided to create its own educational materials, not just for patients but also to help educate its primary care doctors; the rheumatologists, dermatologists, and gastroenterologists using the biosimilars; the nurses infusing patients; and the pharmacists preparing the biosimilars.
The health system also has a different approach to choosing medication. Instead of having an insurance company or PBM decide what’s in the formulary, clinicians work with the pharmacists at Kaiser to look at clinical evidence and decide which biosimilar to use. Most of its plans also provide lower copays to patients when they use the biosimilar.
This was the approach for Humira biosimilars, Dr. Awsare said. Eight will be on the market in 2023. “Our rheumatologists, dermatologists, and gastroenterologists looked at the data from Europe, looked at some real-world evidence, and then said: ‘We think this one’s going to be the best one for our patients.’ ”
Having clinicians choose the biosimilar instead of a health plan makes it a lot easier to have conversations with patients, he said. “Once we’ve moved that market share to that particular biosimilar, we give our physicians the time to have those discussions.”
Clinical pharmacists also provide educational support, offering guidance on issues such as side effects, as patients transition to the biosimilar. “We like to use the word ‘transition’ because it’s essentially the same biologic. So, you’re not actually switching,” Dr. Awsare said.
No consensus on interchangeability
Whether the conversation on interchangeability will affect patient conversations with physicians depends on who you ask.
If a biosimilar has an interchangeability designation, it means that the pharmacist can substitute it without the intervention of the clinician who prescribed the reference product. It does not relate to the quality, safety, or effectiveness of biosimilars or interchangeable biosimilar products, Dr. Popovian said.
The United States is the only country that has this designation. Even though it’s not identical to the originator drug, a biosimilar has the same clinical efficacy and safety profile. “So clinically, interchangeability is meaningless,” Dr. Awsare said.
In its report on biosimilars in the autoimmune category, CVS acknowledged that interchangeability was important but would not be a significant factor in driving adoption of biosimilars. However, in a Cardinal Health survey of 72 gastroenterologists, 38% cited the interchangeability of biosimilars as a top concern for adalimumab biosimilars, along with transitioning patients from Humira to a biosimilar (44%).
“Patient education regarding biosimilar safety, efficacy, and interchangeability appears paramount to the acceptance of these products, particularly for patients who are switched from a reference product,” Dr. Kaul noted in the Cardinal Health report.
Wherever supported by data, Dr. Kaul recommends incorporating biosimilar use and interchangeability into best practice guidelines going forward. “That will go a long way in disseminating the latest information on this topic and position this paradigm for increased adoption among providers.”
Some physicians like Dr. Snow aren’t that concerned with interchangeability. This hasn’t affected conversations with patients, he said. Multiple studies demonstrating the lack of antibody formation with multiple switches from different biosimilar drugs has eased his concern about multiple switches causing problems.
“Initially, there was a gap in demonstrating the long-term effect of multiple switches on antibody production and drug effectiveness. That gap has started to close as more data from Europe’s experience with biosimilars becomes available,” Dr. Snow said.
Resources for physicians, patients
The federal government has taken steps to advance biosimilars education and adoption. In 2021, President Biden signed the Advancing Education on Biosimilars Act into law, which directs the FDA to develop or improve continuing education programs that address prescribing of biosimilars and biological products.
The FDA provides educational materials on its website, including a comprehensive curriculum toolkit. The Accreditation Council for Medical Affairs has also created an online 40-hour curriculum for health care professionals called the Board-Certified Biologics and Biosimilars Specialist Program.
Dr. Fabbro recommended patients use the FDA page Biosimilar Basics for Patients to educate themselves on biosimilars. The Global Healthy Living Foundation’s podcast, Breaking Down Biosimilars, is another free resource for patients.
“While much has changed, the continued need for multistakeholder education, awareness, and dedicated research remains even more important as we expand into newer therapeutic areas and classes,” wrote the authors of the Cardinal Health report.
Help patients understand biologics and biosimilars by using AGA resources for providers and patients available at gastro.org/biosimilars.
Dr. Regueiro is on advisory boards and consults for AbbVie, Janssen, UCB, Takeda, Pfizer, Bristol-Myers Squibb, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, TARGET PharmaSolutions, Trellis, and Boehringer Ingelheim. Dr. Fabbro is a principal investigator for Castle Biosciences, on the speakers bureau for Valchlor, and on the advisory boards of Janssen and Bristol-Myers Squibb. Dr. Popovian, Dr. Snow, Dr. Awsare, and Dr. Kaul had no disclosures.
A version of this article originally appeared on Medscape.com.