User login
Perinatal HIV nearly eradicated in U.S.
, with less than 1 baby for every 100,000 live births having the virus, a new study released by researchers at the Centers for Disease Control and Prevention finds.
The report marks significant progress on the U.S. government’s goal to eradicate perinatal HIV, an immune-weakening and potentially deadly virus that is passed from mother to baby during pregnancy. Just 32 children in the country were diagnosed in 2019, compared with twice as many in 2010, according to the CDC.
Mothers who are HIV positive can prevent transmission of the infection by receiving antiretroviral therapy, according to Monica Gandhi, MD, MPH, a professor of medicine at University of California, San Francisco’s division of HIV, infectious disease and global medicine.
Dr. Gandhi said she could recall only one case of perinatal HIV in the San Francisco area over the last decade.
“This country has been really aggressive about counseling women who are pregnant and getting mothers in care,” Dr. Gandhi said.
The treatment method was discovered more than 30 years ago. Prior to the therapy and ensuing awareness campaigns to prevent transmission, mothers with HIV would typically pass the virus to their child in utero, during delivery, or while breastfeeding.
“There should be zero children born with HIV, given that we’ve had these drugs for so long,” Dr. Ghandi said.
Disparities persist
But challenges remain in some communities, where babies born to Black mothers are disproportionately affected by the disease, the new study found. “Racial and ethnic differences in perinatal HIV diagnoses persisted through the 10-year period,” the report’s authors concluded. “The highest rates of perinatal HIV diagnoses were seen among infants born to Black women.”
Although rates of perinatal HIV declined for babies born to Black mothers over the decade-long study, the diagnosis rate was above the goal of elimination at 3.1 for every 100,000 live births, according to the data.
Meanwhile, transmission rates hovered around 1%-2% for Latinx and Hispanic women and mothers who identified as “other races,” including Native American.
Despite the availability of medication, expectant mothers may face several hurdles to getting the daily treatment they need to prevent transmission to their fetus, according to Jennifer Jao, MD, MPH, a physician of infectious diseases at Lurie Children’s Hospital of Chicago.
They might have trouble securing health insurance or finding transportation to doctor’s appointments, or face other problems like lacking secure housing or food – all factors that prevent them from prioritizing the care.
“All of those things play into the mix,” Dr. Jao said. “We see over and over again that closing the gap means you’ve got to reach the women who are pregnant and who don’t have resources.”
Progress in ‘danger’
Experts said they’re not sure what the impact of the COVID-19 pandemic, accompanied by a recent uptick in sexually transmitted diseases, will be on rates of perinatal HIV. Some women were unable to access prenatal health care during the pandemic because they couldn’t access public transportation or childcare, the U.S. Government Accountability Office said in 2022.
Globally, a decline in rates of HIV and AIDS rates has slowed, prompting the World Health Organization to warn last year that progress on the disease is in danger. Researchers only included HIV rates in the United States through 2019, so the data are outdated, Dr. Gandhi noted.
“All of this put together means we don’t know where we are with perinatal transmission over the last 3 years,” she said.
In an accompanying editorial, coauthors Nahida Chakhtoura, MD, MsGH, and Bill Kapogiannis, MD, both with the National Institutes of Health, urge health care professionals to take an active role in eliminating these racial and ethnic disparities in an effort to – as the title of their editorial proclaims – achieve a “road to zero perinatal HIV transmission” in the United States.
“The more proactive we are in identifying and promptly addressing systematic deficiencies that exacerbate health inequities in cutting-edge research innovations and optimal clinical service provision,” they write, “the less reactive we will need to be when new transmissible infections appear at our doorstep.”
A version of this article first appeared on Medscape.com.
, with less than 1 baby for every 100,000 live births having the virus, a new study released by researchers at the Centers for Disease Control and Prevention finds.
The report marks significant progress on the U.S. government’s goal to eradicate perinatal HIV, an immune-weakening and potentially deadly virus that is passed from mother to baby during pregnancy. Just 32 children in the country were diagnosed in 2019, compared with twice as many in 2010, according to the CDC.
Mothers who are HIV positive can prevent transmission of the infection by receiving antiretroviral therapy, according to Monica Gandhi, MD, MPH, a professor of medicine at University of California, San Francisco’s division of HIV, infectious disease and global medicine.
Dr. Gandhi said she could recall only one case of perinatal HIV in the San Francisco area over the last decade.
“This country has been really aggressive about counseling women who are pregnant and getting mothers in care,” Dr. Gandhi said.
The treatment method was discovered more than 30 years ago. Prior to the therapy and ensuing awareness campaigns to prevent transmission, mothers with HIV would typically pass the virus to their child in utero, during delivery, or while breastfeeding.
“There should be zero children born with HIV, given that we’ve had these drugs for so long,” Dr. Ghandi said.
Disparities persist
But challenges remain in some communities, where babies born to Black mothers are disproportionately affected by the disease, the new study found. “Racial and ethnic differences in perinatal HIV diagnoses persisted through the 10-year period,” the report’s authors concluded. “The highest rates of perinatal HIV diagnoses were seen among infants born to Black women.”
Although rates of perinatal HIV declined for babies born to Black mothers over the decade-long study, the diagnosis rate was above the goal of elimination at 3.1 for every 100,000 live births, according to the data.
Meanwhile, transmission rates hovered around 1%-2% for Latinx and Hispanic women and mothers who identified as “other races,” including Native American.
Despite the availability of medication, expectant mothers may face several hurdles to getting the daily treatment they need to prevent transmission to their fetus, according to Jennifer Jao, MD, MPH, a physician of infectious diseases at Lurie Children’s Hospital of Chicago.
They might have trouble securing health insurance or finding transportation to doctor’s appointments, or face other problems like lacking secure housing or food – all factors that prevent them from prioritizing the care.
“All of those things play into the mix,” Dr. Jao said. “We see over and over again that closing the gap means you’ve got to reach the women who are pregnant and who don’t have resources.”
Progress in ‘danger’
Experts said they’re not sure what the impact of the COVID-19 pandemic, accompanied by a recent uptick in sexually transmitted diseases, will be on rates of perinatal HIV. Some women were unable to access prenatal health care during the pandemic because they couldn’t access public transportation or childcare, the U.S. Government Accountability Office said in 2022.
Globally, a decline in rates of HIV and AIDS rates has slowed, prompting the World Health Organization to warn last year that progress on the disease is in danger. Researchers only included HIV rates in the United States through 2019, so the data are outdated, Dr. Gandhi noted.
“All of this put together means we don’t know where we are with perinatal transmission over the last 3 years,” she said.
In an accompanying editorial, coauthors Nahida Chakhtoura, MD, MsGH, and Bill Kapogiannis, MD, both with the National Institutes of Health, urge health care professionals to take an active role in eliminating these racial and ethnic disparities in an effort to – as the title of their editorial proclaims – achieve a “road to zero perinatal HIV transmission” in the United States.
“The more proactive we are in identifying and promptly addressing systematic deficiencies that exacerbate health inequities in cutting-edge research innovations and optimal clinical service provision,” they write, “the less reactive we will need to be when new transmissible infections appear at our doorstep.”
A version of this article first appeared on Medscape.com.
, with less than 1 baby for every 100,000 live births having the virus, a new study released by researchers at the Centers for Disease Control and Prevention finds.
The report marks significant progress on the U.S. government’s goal to eradicate perinatal HIV, an immune-weakening and potentially deadly virus that is passed from mother to baby during pregnancy. Just 32 children in the country were diagnosed in 2019, compared with twice as many in 2010, according to the CDC.
Mothers who are HIV positive can prevent transmission of the infection by receiving antiretroviral therapy, according to Monica Gandhi, MD, MPH, a professor of medicine at University of California, San Francisco’s division of HIV, infectious disease and global medicine.
Dr. Gandhi said she could recall only one case of perinatal HIV in the San Francisco area over the last decade.
“This country has been really aggressive about counseling women who are pregnant and getting mothers in care,” Dr. Gandhi said.
The treatment method was discovered more than 30 years ago. Prior to the therapy and ensuing awareness campaigns to prevent transmission, mothers with HIV would typically pass the virus to their child in utero, during delivery, or while breastfeeding.
“There should be zero children born with HIV, given that we’ve had these drugs for so long,” Dr. Ghandi said.
Disparities persist
But challenges remain in some communities, where babies born to Black mothers are disproportionately affected by the disease, the new study found. “Racial and ethnic differences in perinatal HIV diagnoses persisted through the 10-year period,” the report’s authors concluded. “The highest rates of perinatal HIV diagnoses were seen among infants born to Black women.”
Although rates of perinatal HIV declined for babies born to Black mothers over the decade-long study, the diagnosis rate was above the goal of elimination at 3.1 for every 100,000 live births, according to the data.
Meanwhile, transmission rates hovered around 1%-2% for Latinx and Hispanic women and mothers who identified as “other races,” including Native American.
Despite the availability of medication, expectant mothers may face several hurdles to getting the daily treatment they need to prevent transmission to their fetus, according to Jennifer Jao, MD, MPH, a physician of infectious diseases at Lurie Children’s Hospital of Chicago.
They might have trouble securing health insurance or finding transportation to doctor’s appointments, or face other problems like lacking secure housing or food – all factors that prevent them from prioritizing the care.
“All of those things play into the mix,” Dr. Jao said. “We see over and over again that closing the gap means you’ve got to reach the women who are pregnant and who don’t have resources.”
Progress in ‘danger’
Experts said they’re not sure what the impact of the COVID-19 pandemic, accompanied by a recent uptick in sexually transmitted diseases, will be on rates of perinatal HIV. Some women were unable to access prenatal health care during the pandemic because they couldn’t access public transportation or childcare, the U.S. Government Accountability Office said in 2022.
Globally, a decline in rates of HIV and AIDS rates has slowed, prompting the World Health Organization to warn last year that progress on the disease is in danger. Researchers only included HIV rates in the United States through 2019, so the data are outdated, Dr. Gandhi noted.
“All of this put together means we don’t know where we are with perinatal transmission over the last 3 years,” she said.
In an accompanying editorial, coauthors Nahida Chakhtoura, MD, MsGH, and Bill Kapogiannis, MD, both with the National Institutes of Health, urge health care professionals to take an active role in eliminating these racial and ethnic disparities in an effort to – as the title of their editorial proclaims – achieve a “road to zero perinatal HIV transmission” in the United States.
“The more proactive we are in identifying and promptly addressing systematic deficiencies that exacerbate health inequities in cutting-edge research innovations and optimal clinical service provision,” they write, “the less reactive we will need to be when new transmissible infections appear at our doorstep.”
A version of this article first appeared on Medscape.com.
New variant jumps to second place on COVID list
Officially labeled XBB.1.16, Arcturus is a subvariant of Omicron that was first seen in India and has been on the World Health Organization’s watchlist since the end of March. The CDC’s most recent update now lists Arcturus as causing 7% of U.S. coronavirus cases, landing it in second place behind its long-predominant Omicron cousin XBB.1.5, which causes 78% of cases.
Arcturus is more transmissible but not more dangerous than recent chart-topping strains, experts say.
“It is causing increasing case counts in certain parts of the world, including India. We’re not seeing high rates of XBB.1.16 yet in the United States, but it may become more prominent in coming weeks,” Mayo Clinic viral disease expert Matthew Binnicker, PhD, told The Seattle Times.
Arcturus has been causing a new symptom in children, Indian medical providers have reported.
“One new feature of cases caused by this variant is that it seems to be causing conjunctivitis, or red and itchy eyes, in young patients,” Dr. Binnicker said. “This is not something that we’ve seen with prior strains of the virus.”
More than 11,000 people in the United States remained hospitalized with COVID at the end of last week, and 1,327 people died of the virus last week, CDC data show. To date, 6.9 million people worldwide have died from COVID, the WHO says. Of those deaths, more than 1.1 million occurred in the U.S.
A version of this article originally appeared on WebMD.com.
Officially labeled XBB.1.16, Arcturus is a subvariant of Omicron that was first seen in India and has been on the World Health Organization’s watchlist since the end of March. The CDC’s most recent update now lists Arcturus as causing 7% of U.S. coronavirus cases, landing it in second place behind its long-predominant Omicron cousin XBB.1.5, which causes 78% of cases.
Arcturus is more transmissible but not more dangerous than recent chart-topping strains, experts say.
“It is causing increasing case counts in certain parts of the world, including India. We’re not seeing high rates of XBB.1.16 yet in the United States, but it may become more prominent in coming weeks,” Mayo Clinic viral disease expert Matthew Binnicker, PhD, told The Seattle Times.
Arcturus has been causing a new symptom in children, Indian medical providers have reported.
“One new feature of cases caused by this variant is that it seems to be causing conjunctivitis, or red and itchy eyes, in young patients,” Dr. Binnicker said. “This is not something that we’ve seen with prior strains of the virus.”
More than 11,000 people in the United States remained hospitalized with COVID at the end of last week, and 1,327 people died of the virus last week, CDC data show. To date, 6.9 million people worldwide have died from COVID, the WHO says. Of those deaths, more than 1.1 million occurred in the U.S.
A version of this article originally appeared on WebMD.com.
Officially labeled XBB.1.16, Arcturus is a subvariant of Omicron that was first seen in India and has been on the World Health Organization’s watchlist since the end of March. The CDC’s most recent update now lists Arcturus as causing 7% of U.S. coronavirus cases, landing it in second place behind its long-predominant Omicron cousin XBB.1.5, which causes 78% of cases.
Arcturus is more transmissible but not more dangerous than recent chart-topping strains, experts say.
“It is causing increasing case counts in certain parts of the world, including India. We’re not seeing high rates of XBB.1.16 yet in the United States, but it may become more prominent in coming weeks,” Mayo Clinic viral disease expert Matthew Binnicker, PhD, told The Seattle Times.
Arcturus has been causing a new symptom in children, Indian medical providers have reported.
“One new feature of cases caused by this variant is that it seems to be causing conjunctivitis, or red and itchy eyes, in young patients,” Dr. Binnicker said. “This is not something that we’ve seen with prior strains of the virus.”
More than 11,000 people in the United States remained hospitalized with COVID at the end of last week, and 1,327 people died of the virus last week, CDC data show. To date, 6.9 million people worldwide have died from COVID, the WHO says. Of those deaths, more than 1.1 million occurred in the U.S.
A version of this article originally appeared on WebMD.com.
U.S. syphilis cases reach 70-year high
Cases of the sexually transmitted disease syphilis soared in 2021 to the highest total in more than 70 years, a new report says.
Earlier in 2023, the Centers for Disease Control and Prevention issued preliminary projections that syphilis rates had made a startling jump from 2020 to 2021. But now that health officials have finalized all of the 2021 data, the increase is worse than what was announced back in March.
In just a 1-year period, from 2020 to 2021, cases increased by 32%, to 176,713, according to newly finalized data from the CDC. That is the highest total number of syphilis cases the U.S. has seen since 1950.
The total number of STD cases in the U.S. in 2021 was 2.5 million, including 1.6 million cases of chlamydia, which was up 4% over the year prior.
A CDC official labeled the situation an epidemic.
“The reasons for the ongoing increases are multifaceted – and so are the solutions,” said Leandro Mena, MD, MPH, director of the CDC’s STD prevention division, in a statement. “It will take many of us working together to effectively use new and existing tools to increase access to quality sexual health care services for more people and to encourage ongoing innovation and prioritization of STI prevention and treatment in this country.”
Syphilis causes sores and rashes and, left untreated over a long period of time, can cause severe problems in organs, the brain, and the nervous system. Untreated congenital syphilis can lead to stillbirth. The treatment for syphilis is antibiotics.
The CDC called a 32% increase from 2020 to 2021 of congenital syphilis cases “alarming,” reporting that it resulted in 220 stillbirths and infant deaths in 2021.
The rise in STDs during the pandemic has been attributed to decreased attention and resources devoted to sexual health. Opioid use is also considered a contributing factor.
A version of this article first appeared on WebMD.com.
Cases of the sexually transmitted disease syphilis soared in 2021 to the highest total in more than 70 years, a new report says.
Earlier in 2023, the Centers for Disease Control and Prevention issued preliminary projections that syphilis rates had made a startling jump from 2020 to 2021. But now that health officials have finalized all of the 2021 data, the increase is worse than what was announced back in March.
In just a 1-year period, from 2020 to 2021, cases increased by 32%, to 176,713, according to newly finalized data from the CDC. That is the highest total number of syphilis cases the U.S. has seen since 1950.
The total number of STD cases in the U.S. in 2021 was 2.5 million, including 1.6 million cases of chlamydia, which was up 4% over the year prior.
A CDC official labeled the situation an epidemic.
“The reasons for the ongoing increases are multifaceted – and so are the solutions,” said Leandro Mena, MD, MPH, director of the CDC’s STD prevention division, in a statement. “It will take many of us working together to effectively use new and existing tools to increase access to quality sexual health care services for more people and to encourage ongoing innovation and prioritization of STI prevention and treatment in this country.”
Syphilis causes sores and rashes and, left untreated over a long period of time, can cause severe problems in organs, the brain, and the nervous system. Untreated congenital syphilis can lead to stillbirth. The treatment for syphilis is antibiotics.
The CDC called a 32% increase from 2020 to 2021 of congenital syphilis cases “alarming,” reporting that it resulted in 220 stillbirths and infant deaths in 2021.
The rise in STDs during the pandemic has been attributed to decreased attention and resources devoted to sexual health. Opioid use is also considered a contributing factor.
A version of this article first appeared on WebMD.com.
Cases of the sexually transmitted disease syphilis soared in 2021 to the highest total in more than 70 years, a new report says.
Earlier in 2023, the Centers for Disease Control and Prevention issued preliminary projections that syphilis rates had made a startling jump from 2020 to 2021. But now that health officials have finalized all of the 2021 data, the increase is worse than what was announced back in March.
In just a 1-year period, from 2020 to 2021, cases increased by 32%, to 176,713, according to newly finalized data from the CDC. That is the highest total number of syphilis cases the U.S. has seen since 1950.
The total number of STD cases in the U.S. in 2021 was 2.5 million, including 1.6 million cases of chlamydia, which was up 4% over the year prior.
A CDC official labeled the situation an epidemic.
“The reasons for the ongoing increases are multifaceted – and so are the solutions,” said Leandro Mena, MD, MPH, director of the CDC’s STD prevention division, in a statement. “It will take many of us working together to effectively use new and existing tools to increase access to quality sexual health care services for more people and to encourage ongoing innovation and prioritization of STI prevention and treatment in this country.”
Syphilis causes sores and rashes and, left untreated over a long period of time, can cause severe problems in organs, the brain, and the nervous system. Untreated congenital syphilis can lead to stillbirth. The treatment for syphilis is antibiotics.
The CDC called a 32% increase from 2020 to 2021 of congenital syphilis cases “alarming,” reporting that it resulted in 220 stillbirths and infant deaths in 2021.
The rise in STDs during the pandemic has been attributed to decreased attention and resources devoted to sexual health. Opioid use is also considered a contributing factor.
A version of this article first appeared on WebMD.com.
Cancer, heart disease vaccines may be ready by 2030, Moderna says
The announcement is yet another sign of what many are calling “the golden age” of vaccine development, which is largely credited to the pandemic’s use of mRNA technology to create COVID-19 vaccines.
“I think what we have learned in recent months is that if you ever thought that mRNA was just for infectious diseases, or just for COVID, the evidence now is that that’s absolutely not the case,” Moderna Chief Medical Officer Paul Burton, MD, PhD, told The Guardian. “It can be applied to all sorts of disease areas; we are in cancer, infectious disease, cardiovascular disease, autoimmune diseases, rare disease. We have studies in all of those areas, and they have all shown tremendous promise.”
The U.S. Food and Drug Administration recently designated two new Moderna vaccines as breakthrough therapies: a shot that prevents respiratory syncytial virus (RSV) in older people and a shot that helps prevent the recurrence of melanoma. The FDA’s breakthrough designation is given when a new treatment’s early trial results are substantially better than an existing therapy.
The mRNA vaccine technology that made headlines for its role in COVID-19 vaccines works by teaching the body how to make a specific protein to help the immune system prevent or target a certain disease.
Dr. Burton anticipates that mRNA technology will result in breakthroughs such as a cancer vaccine that can be personalized based on the features of a specific tumor.
“I think we will have mRNA-based therapies for rare diseases that were previously undruggable, and I think that 10 years from now, we will be approaching a world where you truly can identify the genetic cause of a disease and, with relative simplicity, go and edit that out and repair it using mRNA-based technology,” he said.
The Moderna executive made the statements before its annual update on its vaccine pipeline projects, which the company calls “Vaccines Day.” The Massachusetts-based drugmaker said it has given someone the first dose of a “next-generation” COVID-19 vaccine in a phase III trial, has made progress on a Lyme disease shot, and is developing a vaccine for the highly contagious norovirus.
In all, Moderna expects “six major vaccine product launches in the next few years,” the company said in a statement, adding that it expects the COVID-19 booster market alone to be valued at $15 billion.
A version of this article first appeared on WebMD.com.
The announcement is yet another sign of what many are calling “the golden age” of vaccine development, which is largely credited to the pandemic’s use of mRNA technology to create COVID-19 vaccines.
“I think what we have learned in recent months is that if you ever thought that mRNA was just for infectious diseases, or just for COVID, the evidence now is that that’s absolutely not the case,” Moderna Chief Medical Officer Paul Burton, MD, PhD, told The Guardian. “It can be applied to all sorts of disease areas; we are in cancer, infectious disease, cardiovascular disease, autoimmune diseases, rare disease. We have studies in all of those areas, and they have all shown tremendous promise.”
The U.S. Food and Drug Administration recently designated two new Moderna vaccines as breakthrough therapies: a shot that prevents respiratory syncytial virus (RSV) in older people and a shot that helps prevent the recurrence of melanoma. The FDA’s breakthrough designation is given when a new treatment’s early trial results are substantially better than an existing therapy.
The mRNA vaccine technology that made headlines for its role in COVID-19 vaccines works by teaching the body how to make a specific protein to help the immune system prevent or target a certain disease.
Dr. Burton anticipates that mRNA technology will result in breakthroughs such as a cancer vaccine that can be personalized based on the features of a specific tumor.
“I think we will have mRNA-based therapies for rare diseases that were previously undruggable, and I think that 10 years from now, we will be approaching a world where you truly can identify the genetic cause of a disease and, with relative simplicity, go and edit that out and repair it using mRNA-based technology,” he said.
The Moderna executive made the statements before its annual update on its vaccine pipeline projects, which the company calls “Vaccines Day.” The Massachusetts-based drugmaker said it has given someone the first dose of a “next-generation” COVID-19 vaccine in a phase III trial, has made progress on a Lyme disease shot, and is developing a vaccine for the highly contagious norovirus.
In all, Moderna expects “six major vaccine product launches in the next few years,” the company said in a statement, adding that it expects the COVID-19 booster market alone to be valued at $15 billion.
A version of this article first appeared on WebMD.com.
The announcement is yet another sign of what many are calling “the golden age” of vaccine development, which is largely credited to the pandemic’s use of mRNA technology to create COVID-19 vaccines.
“I think what we have learned in recent months is that if you ever thought that mRNA was just for infectious diseases, or just for COVID, the evidence now is that that’s absolutely not the case,” Moderna Chief Medical Officer Paul Burton, MD, PhD, told The Guardian. “It can be applied to all sorts of disease areas; we are in cancer, infectious disease, cardiovascular disease, autoimmune diseases, rare disease. We have studies in all of those areas, and they have all shown tremendous promise.”
The U.S. Food and Drug Administration recently designated two new Moderna vaccines as breakthrough therapies: a shot that prevents respiratory syncytial virus (RSV) in older people and a shot that helps prevent the recurrence of melanoma. The FDA’s breakthrough designation is given when a new treatment’s early trial results are substantially better than an existing therapy.
The mRNA vaccine technology that made headlines for its role in COVID-19 vaccines works by teaching the body how to make a specific protein to help the immune system prevent or target a certain disease.
Dr. Burton anticipates that mRNA technology will result in breakthroughs such as a cancer vaccine that can be personalized based on the features of a specific tumor.
“I think we will have mRNA-based therapies for rare diseases that were previously undruggable, and I think that 10 years from now, we will be approaching a world where you truly can identify the genetic cause of a disease and, with relative simplicity, go and edit that out and repair it using mRNA-based technology,” he said.
The Moderna executive made the statements before its annual update on its vaccine pipeline projects, which the company calls “Vaccines Day.” The Massachusetts-based drugmaker said it has given someone the first dose of a “next-generation” COVID-19 vaccine in a phase III trial, has made progress on a Lyme disease shot, and is developing a vaccine for the highly contagious norovirus.
In all, Moderna expects “six major vaccine product launches in the next few years,” the company said in a statement, adding that it expects the COVID-19 booster market alone to be valued at $15 billion.
A version of this article first appeared on WebMD.com.
New COVID variant on WHO’s radar causing itchy eyes in children
A new COVID-19 variant that recently landed on the World Health Organization’s radar may cause previously unseen symptoms in children, according to a new report.
While the variant, called “Arcturus,” hasn’t yet made the Centers for Disease Control and Prevention’s watchlist, , according to The Times of India.
The new itchy eye symptom is in addition to a high fever and cough, Vipin M. Vashishtha, MD, said on Twitter, noting that pediatric COVID cases have picked up there for the first time in 6 months.
The country has also seen a rise in adenovirus cases among children with similar symptoms. COVID and adenovirus cannot be distinguished without testing, and many parents don’t want to have their children tested because the swabs are uncomfortable, The Times of India reported. One doctor told the newspaper that among every 10 children with COVID-like symptoms, 2 or 3 of them had tested positive on a COVID test taken at home.
Health officials in India are doing mock drills to check how prepared the country’s hospitals are as India sees cases rise, the BBC reported. India struggled during a COVID-19 surge in 2021, at which time sickened people were seen lying on sidewalks outside overflowing hospitals, and reports surfaced of a black market for private citizens to buy oxygen.
Arcturus (formally, Omicron subvariant XBB.1.16) made news recently as it landed on the WHO’s radar after surfacing in India. A WHO official called it “one to watch.” The Times of India reported that 234 new cases of XBB.1.16 were included in the country’s latest 5,676 new infections, meaning the subvariant accounts for 4% of new COVID cases.
A version of this article originally appeared on WebMD.com.
A new COVID-19 variant that recently landed on the World Health Organization’s radar may cause previously unseen symptoms in children, according to a new report.
While the variant, called “Arcturus,” hasn’t yet made the Centers for Disease Control and Prevention’s watchlist, , according to The Times of India.
The new itchy eye symptom is in addition to a high fever and cough, Vipin M. Vashishtha, MD, said on Twitter, noting that pediatric COVID cases have picked up there for the first time in 6 months.
The country has also seen a rise in adenovirus cases among children with similar symptoms. COVID and adenovirus cannot be distinguished without testing, and many parents don’t want to have their children tested because the swabs are uncomfortable, The Times of India reported. One doctor told the newspaper that among every 10 children with COVID-like symptoms, 2 or 3 of them had tested positive on a COVID test taken at home.
Health officials in India are doing mock drills to check how prepared the country’s hospitals are as India sees cases rise, the BBC reported. India struggled during a COVID-19 surge in 2021, at which time sickened people were seen lying on sidewalks outside overflowing hospitals, and reports surfaced of a black market for private citizens to buy oxygen.
Arcturus (formally, Omicron subvariant XBB.1.16) made news recently as it landed on the WHO’s radar after surfacing in India. A WHO official called it “one to watch.” The Times of India reported that 234 new cases of XBB.1.16 were included in the country’s latest 5,676 new infections, meaning the subvariant accounts for 4% of new COVID cases.
A version of this article originally appeared on WebMD.com.
A new COVID-19 variant that recently landed on the World Health Organization’s radar may cause previously unseen symptoms in children, according to a new report.
While the variant, called “Arcturus,” hasn’t yet made the Centers for Disease Control and Prevention’s watchlist, , according to The Times of India.
The new itchy eye symptom is in addition to a high fever and cough, Vipin M. Vashishtha, MD, said on Twitter, noting that pediatric COVID cases have picked up there for the first time in 6 months.
The country has also seen a rise in adenovirus cases among children with similar symptoms. COVID and adenovirus cannot be distinguished without testing, and many parents don’t want to have their children tested because the swabs are uncomfortable, The Times of India reported. One doctor told the newspaper that among every 10 children with COVID-like symptoms, 2 or 3 of them had tested positive on a COVID test taken at home.
Health officials in India are doing mock drills to check how prepared the country’s hospitals are as India sees cases rise, the BBC reported. India struggled during a COVID-19 surge in 2021, at which time sickened people were seen lying on sidewalks outside overflowing hospitals, and reports surfaced of a black market for private citizens to buy oxygen.
Arcturus (formally, Omicron subvariant XBB.1.16) made news recently as it landed on the WHO’s radar after surfacing in India. A WHO official called it “one to watch.” The Times of India reported that 234 new cases of XBB.1.16 were included in the country’s latest 5,676 new infections, meaning the subvariant accounts for 4% of new COVID cases.
A version of this article originally appeared on WebMD.com.
75-year-old man • recent history of hand-foot-mouth disease • discolored fingernails and toenails lifting from the proximal end • Dx?
THE CASE
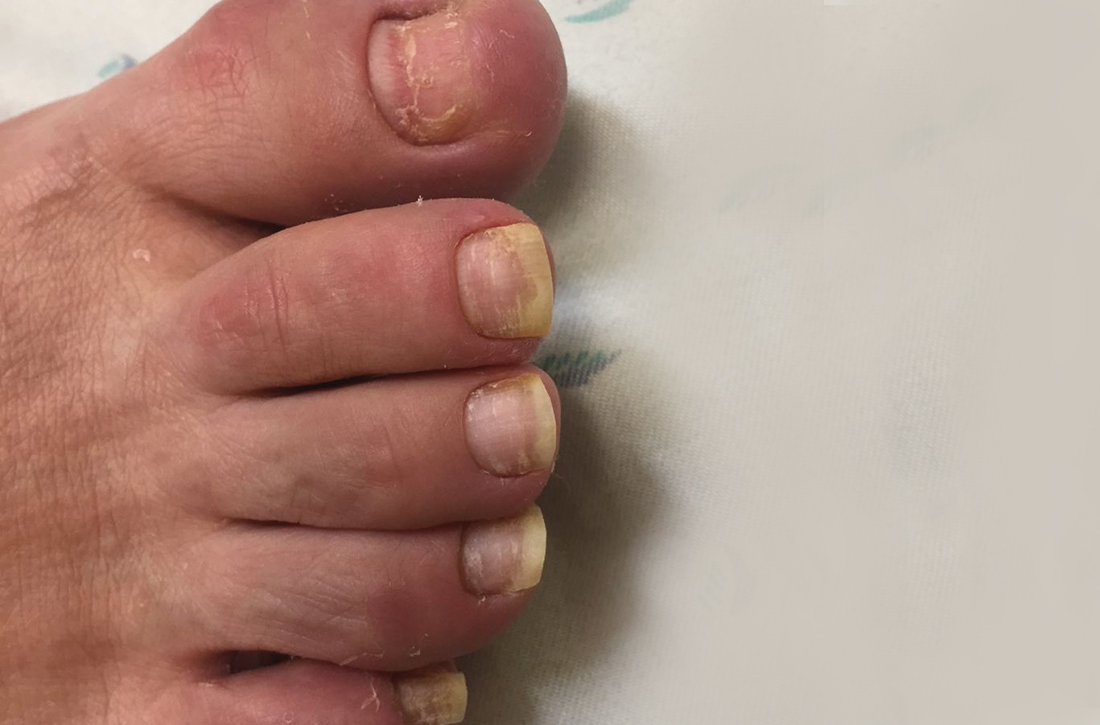
A 75-year-old man sought care from his primary care physician because his “fingernails and toenails [were] all falling off.” He did not feel ill and had no other complaints. His vital signs were unremarkable. He had no history of malignancies, chronic skin conditions, or systemic diseases. His fingernails and toenails were discolored and lifting from the proximal end of his nail beds (FIGURE). One of his great toenails had already fallen off, 1 thumb nail was minimally attached with the cuticle, and the rest of his nails were loose and in the process of separating from their nail beds. There was no nail pitting, rash, or joint swelling and tenderness.
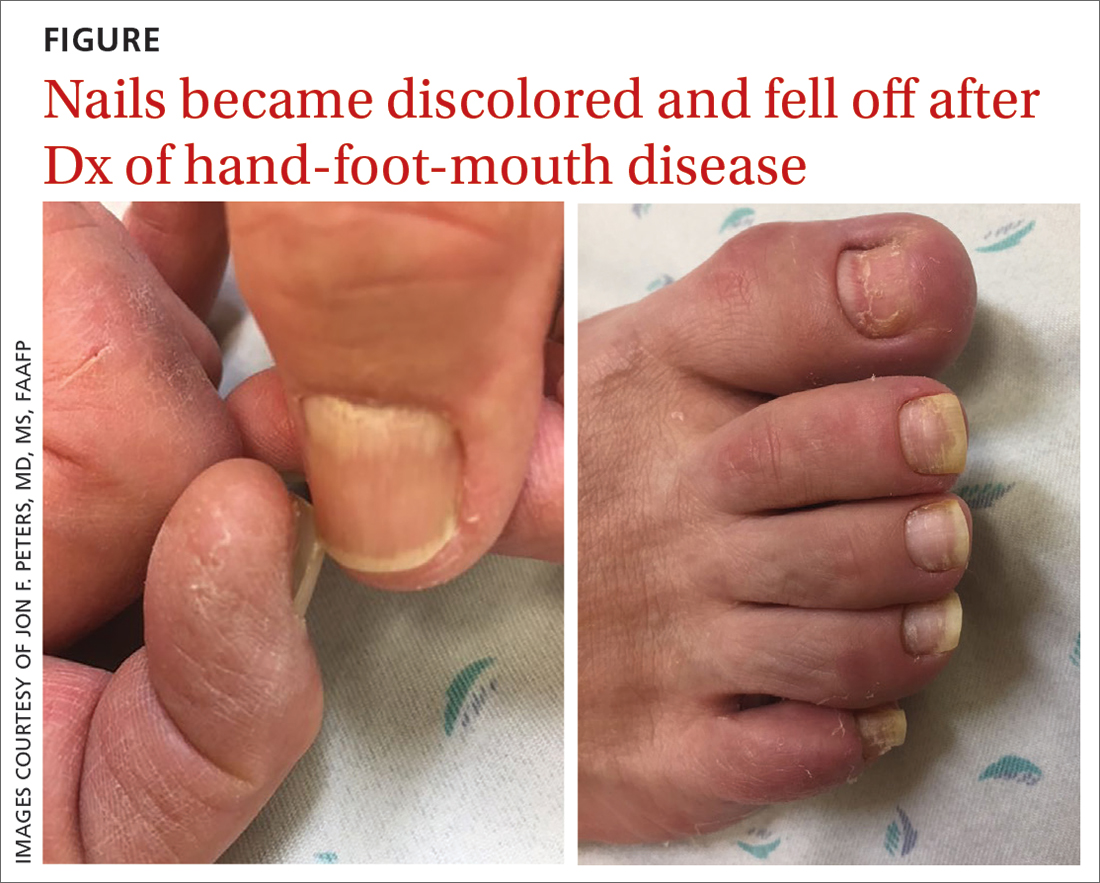
The patient reported that while on vacation in Hawaii 3 weeks earlier, he had sought care at an urgent care clinic for a painless rash on his hands and the soles of his feet. At that time, he did not feel ill or have mouth ulcers, penile discharge, or arthralgia. There had been no recent changes to his prescription medications, which included finasteride, terazosin, omeprazole, and an albuterol inhaler. He denied taking over-the-counter medications or supplements.
The physical exam at the urgent care had revealed multiple blotchy, dark, 0.5- to 1-cm nonpruritic lesions that were desquamating. No oral lesions were seen. He had been given a diagnosis of hand-foot-mouth disease (HFMD) and reassured that it would resolve on its own in about 10 days.
THE DIAGNOSIS
Several possible diagnoses for nail disorders came to mind with this patient, including onychomycosis, onychoschizia, onycholysis, and onychomadesis.
Onychomycosis is a chronic fungal infection of the nail that affects toenails more often than fingernails.1 The most common form is distal subungual onychomycosis, which begins distally and slowly migrates proximally through the nail matrix.1 Often onychomycosis affects only a few nails unless the patient is elderly or has comorbid conditions, and the nails rarely separate from the nail bed.
Onychoschizia involves lamellar splitting and peeling of the dorsal surface of the nail plate.2 Usually white discolorations appear on the distal edges of the nail.3 It is more common in women than in men and is often caused by nail dehydration from repeated excessive immersion in water with detergents or recurrent application of nail polish.2 However, the nails do not separate from the nail bed, and usually only the fingernails are involved.
Onycholysis is a nail attachment disorder in which the nail plate distally separates from the nail bed. Areas of separation will appear white or yellow. There are many etiologies for onycholysis, including trauma, psoriasis, fungal infection, and contact irritant reactions.3 It also can be caused by medications and thyroid disease.3,4
Continue to: Onychomadesis
Onychomadesis, sometimes considered a severe form of Beau’s line,5,6 is defined by the spontaneous separation of the nail plate from the nail matrix. Although the nail will initially remain attached, proximal shedding will eventually occur.7 When several nails are involved, a systemic source—such as an acute infection, autoimmune disease, medication, malignancy (eg, cutaneous T-cell lymphoma), Kawasaki disease, skin disorders (eg, pemphigus vulgaris or keratosis punctata et planters), or chemotherapy—may be the cause.6-8 If only a few nails are involved, it may be associated with trauma, and in rare cases, onychomadesis can be idiopathic.5,7
In this case, all signs pointed to onychomadesis. All of the patient’s nails were affected (discolored and lifting), his nail loss involved spontaneous proximal separation of the nail plate from the nail matrix, and he had a recent previous infection: HFMD.
DISCUSSION
Onychomadesis is a rare nail-shedding disorder thought to be caused by the temporary arrest of the nail matrix.8 It is a potential late complication of infection, such as HFMD,9 and was first reported in children in Chicago in 2000.10 Since then, onychomadesis has been noted in children in many countries.8 Reports of onychomadesis following HFMD in adults are rare, but it may be underreported because HFMD is more common in children and symptoms are usually minor in adults.11
Molecular studies have associated onychomadesis with coxsackievirus (CV)A6 and CVA10.4 Other serotypes associated with onychomadesis include CVB1, CVB2, CVA5, CVA16, and enteroviruses 71 and 9.4 Most known outbreaks seem to be caused by CVA6.4
No treatment is needed for onychomadesis; physicians can reassure patients that normal nail growth will begin within 1 to 4 months. Because onychomadesis is rare, it does not have its own billing code, so one can use code L60.8 for “Other nail disorders.”12
Our patient was seen in the primary care clinic 3 months after his initial visit. At that time, his nails were no longer discolored and no other abnormalities were present. All of the nails on his fingers and toes were firmly attached and growing normally.
THE TAKEAWAY
The sudden asymptomatic loss of multiple fingernails and toenails—especially with proximal nail shedding—is a rare disorder known as onychomadesis. It can be caused by various etiologies and can be a late complication of HFMD or other viral infections. Onychomadesis should be considered when evaluating older patients, particularly when all of their nails are involved after a viral infection.
CORRESPONDENCE
Jon F. Peters, MD, MS, FAAFP, 14486 SE Lyon Court, Happy Valley, OR 97086; [email protected]
1. Rodgers P, Bassler M. Treating onychomycosis. Am Fam Physician. 2001;63:663-672, 677-678.
2. Sparavigna A, Tenconi B, La Penna L. Efficacy and tolerability of a biomineral formulation for treatment of onychoschizia: a randomized trial. Clin Cosmet Investig Dermatol. 2019:12:355-362. doi: 10.2147/CCID.S187305
3. Singal A, Arora R. Nail as a window of systemic diseases. Indian Dermatol Online J. 2015;6:67-74. doi: 10.4103/2229-5178.153002
4. Cleveland Clinic. Onycholysis. Accessed March 1, 2023. https://my.clevelandclinic.org/health/diseases/22903-onycholysis
5. Chiu H-H, Liu M-T, Chung W-H, et al. The mechanism of onychomadesis (nail shedding) and Beau’s lines following hand-foot-mouth disease. Viruses. 2019;11:522. doi: 10.3390/v11060522
6. Suchonwanit P, Nitayavardhana S. Idiopathic sporadic onychomadesis of toenails. Case Rep Dermatol Med. 2016;2016:6451327. doi: 10.1155/2016/6451327
7. Hardin J, Haber RM. Onychomadesis: literature review. Br J Dermatol. 2015;172:592-596. doi: 10.1111/bjd.13339
8. Li D, Yang W, Xing X, et al. Onychomadesis and potential association with HFMD outbreak in a kindergarten in Hubei providence, China, 2017. BMC Infect Dis. 2019:19:995. doi: 10.1186/s12879-019-4560-8
9. Chiu HH, Wu CS, Lan CE. Onychomadesis: a late complication of hand, foot, and mouth disease. J Emerg Med. 2017;52:243-245. doi: 10.1016/j.jemermed.2016.01.034
10. Clementz GC, Mancini AJ. Nail matrix arrest following hand-foot-mouth disease: a report of five children. Pediatr Dermatol. 2000;17:7-11. doi: 10.1046/j.1525-1470.2000.01702.x
11. Scarfi F, Arunachalam M, Galeone M, et al. An uncommon onychomadesis in adults. Int J Derm. 2014;53:1392-1394. doi: 10.1111/j.1365-4632.2012.05774.x
12. ICD10Data.com. 2023 ICD-10-CM codes. Accessed February 15, 2023. www.icd10data.com/ICD10CM/codes
THE CASE
A 75-year-old man sought care from his primary care physician because his “fingernails and toenails [were] all falling off.” He did not feel ill and had no other complaints. His vital signs were unremarkable. He had no history of malignancies, chronic skin conditions, or systemic diseases. His fingernails and toenails were discolored and lifting from the proximal end of his nail beds (FIGURE). One of his great toenails had already fallen off, 1 thumb nail was minimally attached with the cuticle, and the rest of his nails were loose and in the process of separating from their nail beds. There was no nail pitting, rash, or joint swelling and tenderness.

The patient reported that while on vacation in Hawaii 3 weeks earlier, he had sought care at an urgent care clinic for a painless rash on his hands and the soles of his feet. At that time, he did not feel ill or have mouth ulcers, penile discharge, or arthralgia. There had been no recent changes to his prescription medications, which included finasteride, terazosin, omeprazole, and an albuterol inhaler. He denied taking over-the-counter medications or supplements.
The physical exam at the urgent care had revealed multiple blotchy, dark, 0.5- to 1-cm nonpruritic lesions that were desquamating. No oral lesions were seen. He had been given a diagnosis of hand-foot-mouth disease (HFMD) and reassured that it would resolve on its own in about 10 days.
THE DIAGNOSIS
Several possible diagnoses for nail disorders came to mind with this patient, including onychomycosis, onychoschizia, onycholysis, and onychomadesis.
Onychomycosis is a chronic fungal infection of the nail that affects toenails more often than fingernails.1 The most common form is distal subungual onychomycosis, which begins distally and slowly migrates proximally through the nail matrix.1 Often onychomycosis affects only a few nails unless the patient is elderly or has comorbid conditions, and the nails rarely separate from the nail bed.
Onychoschizia involves lamellar splitting and peeling of the dorsal surface of the nail plate.2 Usually white discolorations appear on the distal edges of the nail.3 It is more common in women than in men and is often caused by nail dehydration from repeated excessive immersion in water with detergents or recurrent application of nail polish.2 However, the nails do not separate from the nail bed, and usually only the fingernails are involved.
Onycholysis is a nail attachment disorder in which the nail plate distally separates from the nail bed. Areas of separation will appear white or yellow. There are many etiologies for onycholysis, including trauma, psoriasis, fungal infection, and contact irritant reactions.3 It also can be caused by medications and thyroid disease.3,4
Continue to: Onychomadesis
Onychomadesis, sometimes considered a severe form of Beau’s line,5,6 is defined by the spontaneous separation of the nail plate from the nail matrix. Although the nail will initially remain attached, proximal shedding will eventually occur.7 When several nails are involved, a systemic source—such as an acute infection, autoimmune disease, medication, malignancy (eg, cutaneous T-cell lymphoma), Kawasaki disease, skin disorders (eg, pemphigus vulgaris or keratosis punctata et planters), or chemotherapy—may be the cause.6-8 If only a few nails are involved, it may be associated with trauma, and in rare cases, onychomadesis can be idiopathic.5,7
In this case, all signs pointed to onychomadesis. All of the patient’s nails were affected (discolored and lifting), his nail loss involved spontaneous proximal separation of the nail plate from the nail matrix, and he had a recent previous infection: HFMD.
DISCUSSION
Onychomadesis is a rare nail-shedding disorder thought to be caused by the temporary arrest of the nail matrix.8 It is a potential late complication of infection, such as HFMD,9 and was first reported in children in Chicago in 2000.10 Since then, onychomadesis has been noted in children in many countries.8 Reports of onychomadesis following HFMD in adults are rare, but it may be underreported because HFMD is more common in children and symptoms are usually minor in adults.11
Molecular studies have associated onychomadesis with coxsackievirus (CV)A6 and CVA10.4 Other serotypes associated with onychomadesis include CVB1, CVB2, CVA5, CVA16, and enteroviruses 71 and 9.4 Most known outbreaks seem to be caused by CVA6.4
No treatment is needed for onychomadesis; physicians can reassure patients that normal nail growth will begin within 1 to 4 months. Because onychomadesis is rare, it does not have its own billing code, so one can use code L60.8 for “Other nail disorders.”12
Our patient was seen in the primary care clinic 3 months after his initial visit. At that time, his nails were no longer discolored and no other abnormalities were present. All of the nails on his fingers and toes were firmly attached and growing normally.
THE TAKEAWAY
The sudden asymptomatic loss of multiple fingernails and toenails—especially with proximal nail shedding—is a rare disorder known as onychomadesis. It can be caused by various etiologies and can be a late complication of HFMD or other viral infections. Onychomadesis should be considered when evaluating older patients, particularly when all of their nails are involved after a viral infection.
CORRESPONDENCE
Jon F. Peters, MD, MS, FAAFP, 14486 SE Lyon Court, Happy Valley, OR 97086; [email protected]
THE CASE
A 75-year-old man sought care from his primary care physician because his “fingernails and toenails [were] all falling off.” He did not feel ill and had no other complaints. His vital signs were unremarkable. He had no history of malignancies, chronic skin conditions, or systemic diseases. His fingernails and toenails were discolored and lifting from the proximal end of his nail beds (FIGURE). One of his great toenails had already fallen off, 1 thumb nail was minimally attached with the cuticle, and the rest of his nails were loose and in the process of separating from their nail beds. There was no nail pitting, rash, or joint swelling and tenderness.

The patient reported that while on vacation in Hawaii 3 weeks earlier, he had sought care at an urgent care clinic for a painless rash on his hands and the soles of his feet. At that time, he did not feel ill or have mouth ulcers, penile discharge, or arthralgia. There had been no recent changes to his prescription medications, which included finasteride, terazosin, omeprazole, and an albuterol inhaler. He denied taking over-the-counter medications or supplements.
The physical exam at the urgent care had revealed multiple blotchy, dark, 0.5- to 1-cm nonpruritic lesions that were desquamating. No oral lesions were seen. He had been given a diagnosis of hand-foot-mouth disease (HFMD) and reassured that it would resolve on its own in about 10 days.
THE DIAGNOSIS
Several possible diagnoses for nail disorders came to mind with this patient, including onychomycosis, onychoschizia, onycholysis, and onychomadesis.
Onychomycosis is a chronic fungal infection of the nail that affects toenails more often than fingernails.1 The most common form is distal subungual onychomycosis, which begins distally and slowly migrates proximally through the nail matrix.1 Often onychomycosis affects only a few nails unless the patient is elderly or has comorbid conditions, and the nails rarely separate from the nail bed.
Onychoschizia involves lamellar splitting and peeling of the dorsal surface of the nail plate.2 Usually white discolorations appear on the distal edges of the nail.3 It is more common in women than in men and is often caused by nail dehydration from repeated excessive immersion in water with detergents or recurrent application of nail polish.2 However, the nails do not separate from the nail bed, and usually only the fingernails are involved.
Onycholysis is a nail attachment disorder in which the nail plate distally separates from the nail bed. Areas of separation will appear white or yellow. There are many etiologies for onycholysis, including trauma, psoriasis, fungal infection, and contact irritant reactions.3 It also can be caused by medications and thyroid disease.3,4
Continue to: Onychomadesis
Onychomadesis, sometimes considered a severe form of Beau’s line,5,6 is defined by the spontaneous separation of the nail plate from the nail matrix. Although the nail will initially remain attached, proximal shedding will eventually occur.7 When several nails are involved, a systemic source—such as an acute infection, autoimmune disease, medication, malignancy (eg, cutaneous T-cell lymphoma), Kawasaki disease, skin disorders (eg, pemphigus vulgaris or keratosis punctata et planters), or chemotherapy—may be the cause.6-8 If only a few nails are involved, it may be associated with trauma, and in rare cases, onychomadesis can be idiopathic.5,7
In this case, all signs pointed to onychomadesis. All of the patient’s nails were affected (discolored and lifting), his nail loss involved spontaneous proximal separation of the nail plate from the nail matrix, and he had a recent previous infection: HFMD.
DISCUSSION
Onychomadesis is a rare nail-shedding disorder thought to be caused by the temporary arrest of the nail matrix.8 It is a potential late complication of infection, such as HFMD,9 and was first reported in children in Chicago in 2000.10 Since then, onychomadesis has been noted in children in many countries.8 Reports of onychomadesis following HFMD in adults are rare, but it may be underreported because HFMD is more common in children and symptoms are usually minor in adults.11
Molecular studies have associated onychomadesis with coxsackievirus (CV)A6 and CVA10.4 Other serotypes associated with onychomadesis include CVB1, CVB2, CVA5, CVA16, and enteroviruses 71 and 9.4 Most known outbreaks seem to be caused by CVA6.4
No treatment is needed for onychomadesis; physicians can reassure patients that normal nail growth will begin within 1 to 4 months. Because onychomadesis is rare, it does not have its own billing code, so one can use code L60.8 for “Other nail disorders.”12
Our patient was seen in the primary care clinic 3 months after his initial visit. At that time, his nails were no longer discolored and no other abnormalities were present. All of the nails on his fingers and toes were firmly attached and growing normally.
THE TAKEAWAY
The sudden asymptomatic loss of multiple fingernails and toenails—especially with proximal nail shedding—is a rare disorder known as onychomadesis. It can be caused by various etiologies and can be a late complication of HFMD or other viral infections. Onychomadesis should be considered when evaluating older patients, particularly when all of their nails are involved after a viral infection.
CORRESPONDENCE
Jon F. Peters, MD, MS, FAAFP, 14486 SE Lyon Court, Happy Valley, OR 97086; [email protected]
1. Rodgers P, Bassler M. Treating onychomycosis. Am Fam Physician. 2001;63:663-672, 677-678.
2. Sparavigna A, Tenconi B, La Penna L. Efficacy and tolerability of a biomineral formulation for treatment of onychoschizia: a randomized trial. Clin Cosmet Investig Dermatol. 2019:12:355-362. doi: 10.2147/CCID.S187305
3. Singal A, Arora R. Nail as a window of systemic diseases. Indian Dermatol Online J. 2015;6:67-74. doi: 10.4103/2229-5178.153002
4. Cleveland Clinic. Onycholysis. Accessed March 1, 2023. https://my.clevelandclinic.org/health/diseases/22903-onycholysis
5. Chiu H-H, Liu M-T, Chung W-H, et al. The mechanism of onychomadesis (nail shedding) and Beau’s lines following hand-foot-mouth disease. Viruses. 2019;11:522. doi: 10.3390/v11060522
6. Suchonwanit P, Nitayavardhana S. Idiopathic sporadic onychomadesis of toenails. Case Rep Dermatol Med. 2016;2016:6451327. doi: 10.1155/2016/6451327
7. Hardin J, Haber RM. Onychomadesis: literature review. Br J Dermatol. 2015;172:592-596. doi: 10.1111/bjd.13339
8. Li D, Yang W, Xing X, et al. Onychomadesis and potential association with HFMD outbreak in a kindergarten in Hubei providence, China, 2017. BMC Infect Dis. 2019:19:995. doi: 10.1186/s12879-019-4560-8
9. Chiu HH, Wu CS, Lan CE. Onychomadesis: a late complication of hand, foot, and mouth disease. J Emerg Med. 2017;52:243-245. doi: 10.1016/j.jemermed.2016.01.034
10. Clementz GC, Mancini AJ. Nail matrix arrest following hand-foot-mouth disease: a report of five children. Pediatr Dermatol. 2000;17:7-11. doi: 10.1046/j.1525-1470.2000.01702.x
11. Scarfi F, Arunachalam M, Galeone M, et al. An uncommon onychomadesis in adults. Int J Derm. 2014;53:1392-1394. doi: 10.1111/j.1365-4632.2012.05774.x
12. ICD10Data.com. 2023 ICD-10-CM codes. Accessed February 15, 2023. www.icd10data.com/ICD10CM/codes
1. Rodgers P, Bassler M. Treating onychomycosis. Am Fam Physician. 2001;63:663-672, 677-678.
2. Sparavigna A, Tenconi B, La Penna L. Efficacy and tolerability of a biomineral formulation for treatment of onychoschizia: a randomized trial. Clin Cosmet Investig Dermatol. 2019:12:355-362. doi: 10.2147/CCID.S187305
3. Singal A, Arora R. Nail as a window of systemic diseases. Indian Dermatol Online J. 2015;6:67-74. doi: 10.4103/2229-5178.153002
4. Cleveland Clinic. Onycholysis. Accessed March 1, 2023. https://my.clevelandclinic.org/health/diseases/22903-onycholysis
5. Chiu H-H, Liu M-T, Chung W-H, et al. The mechanism of onychomadesis (nail shedding) and Beau’s lines following hand-foot-mouth disease. Viruses. 2019;11:522. doi: 10.3390/v11060522
6. Suchonwanit P, Nitayavardhana S. Idiopathic sporadic onychomadesis of toenails. Case Rep Dermatol Med. 2016;2016:6451327. doi: 10.1155/2016/6451327
7. Hardin J, Haber RM. Onychomadesis: literature review. Br J Dermatol. 2015;172:592-596. doi: 10.1111/bjd.13339
8. Li D, Yang W, Xing X, et al. Onychomadesis and potential association with HFMD outbreak in a kindergarten in Hubei providence, China, 2017. BMC Infect Dis. 2019:19:995. doi: 10.1186/s12879-019-4560-8
9. Chiu HH, Wu CS, Lan CE. Onychomadesis: a late complication of hand, foot, and mouth disease. J Emerg Med. 2017;52:243-245. doi: 10.1016/j.jemermed.2016.01.034
10. Clementz GC, Mancini AJ. Nail matrix arrest following hand-foot-mouth disease: a report of five children. Pediatr Dermatol. 2000;17:7-11. doi: 10.1046/j.1525-1470.2000.01702.x
11. Scarfi F, Arunachalam M, Galeone M, et al. An uncommon onychomadesis in adults. Int J Derm. 2014;53:1392-1394. doi: 10.1111/j.1365-4632.2012.05774.x
12. ICD10Data.com. 2023 ICD-10-CM codes. Accessed February 15, 2023. www.icd10data.com/ICD10CM/codes
► Recent history of hand-foot-mouth disease
► Discolored fingernails and toenails lifting from the proximal end
Racial disparities not found in chronic hepatitis B treatment initiation
Researchers studying differences in treatment initiation for chronic hepatitis B (CHB) among a large, multiracial cohort in North America did not find evidence of disparities by race or socioeconomic status.
.
That gap suggests that treatment guidelines need to be simplified and that efforts to increase hepatitis B virus (HBV) awareness and train more clinicians are needed to achieve the World Health Organization’s goal of eliminating HBV by 2030, the researchers write.
The Hepatitis B Research Network study was published online in JAMA Network Open.
The prevalence of CHB in the United States is estimated at 2.4 million. It disproportionately affects persons of Asian or African descent, the investigators note. Their study examined whether treatment initiation and outcomes differ between African American and Black, Asian, and White participants, as well as between African American and Black participants born in North America and East or West Africa.
The research involved 1,550 adult patients: 1,157 Asian American, 193 African American or Black (39 born in the United States, 90 in East Africa, 53 in West Africa, and 11 elsewhere), 157 White, and 43 who identified as being of “other races.” All had CHB but were not receiving antiviral treatment at enrollment.
Participants came from 20 centers in the United States and one in Canada. They underwent clinical and laboratory assessments and could receive anti-HBV treatment after they enrolled. Enrollment was from Jan. 14, 2011, to Jan. 28, 2018. Participants were followed at 12 and 24 weeks and every 24 weeks thereafter in the longitudinal cohort study by Mandana Khalili, MD, division of gastroenterology and hepatology, University of California, San Francisco, and colleagues.
Information on patients’ country of birth, duration of U.S. or Canadian residency, educational level, employment, insurance, prior antiviral treatment, family history of HBV or hepatocellular carcinoma (HCC), and mode of transmission were collected by research coordinators.
Treatment initiation
During the study period, slightly fewer than one-third (32.5%) of the participants initiated treatment. The incidences were 4.8 per 100 person-years in African American or Black participants, 9.9 per 100 person-years in Asian participants, 6.6 per 100 person-years in White participants, and 7.9 per 100 person-years in those of other races (P < .001).
A lower percentage of African American and Black participants (14%) met the American Association for the Study of Liver Diseases treatment criteria, compared with Asian (22%) and White (27%) participants (P = .01).
When the researchers compared cumulative probability of initiating treatment by race for those who met criteria for treatment, they found no significant differences by race.
At 72 weeks, initiation probability was 0.45 for African American and Black patients and 0.51 for Asian and White patients (P = .68). Similarly, among African American and Black participants who met treatment criteria, there were no significant differences in cumulative probability of treatment by region of birth.
The cumulative percentage of treatment initiation for those who met guideline-based criteria was 62%.
“Among participants with a treatment indication, treatment rates did not differ significantly by race, despite marked differences in educational level, income, and type of health care insurance across the racial groups,” the researchers write. “Moreover, race was not an independent estimator of treatment initiation when adjusting for known factors associated with a higher risk of adverse clinical outcomes, namely, HBV DNA, disease severity, sex, and age.”
Adverse liver outcomes (hepatic decompensation, HCC, liver transplant, and death) were rare and did not vary significantly by race, the researchers write.
One study limitation is that participants were linked to specialty liver clinics, so the findings may not be generalizable to patients who receive care in other settings, the authors note.
The results are “reassuring,” said senior author Anna S. Lok, MD, division of gastroenterology and hepatology at University of Michigan in Ann Arbor. However, she noted, study participants had already overcome barriers to receiving care at major academic centers.
“Once you get into the big academic liver centers, then maybe everything is equal, but in the real world, a lot of people don’t ever get to the big liver centers,” she said. The question becomes: “Are we serving only a portion of the patient population?”
Many factors drive the decision to undergo treatment, including the doctor’s opinion as to need and the patient’s desire to receive treatment, she said.
The study participants who were more likely to get treated were those with higher-level disease who had a stronger indication for treatment, Dr. Lok said.
Finding the disparities
Centers for Disease Control and Prevention statistics show that Black people are 3.9 times more likely to have CHB and 2.5 times more likely to die from it than White people, notes H. Nina Kim, MD, with the department of medicine, University of Washington, Seattle, in an accompanying invited commentary.
“The fact that we have not observed racial disparities in treatment initiation does not mean none exist; it means we have to look harder to find them,” she writes.
“We need to examine whether our guidelines for HBV treatment are so complex that it becomes the purview of specialists, thereby restricting access and deepening inequities,” Dr. Kim adds. “We should look closely at retention in care, the step that precedes treatment, and stratify this outcome by race and ethnicity.”
Primary care physicians in some regions might find it difficult to manage patients who have hepatitis B because they see so few of them, Dr. Lok noted.
Dr. Khalili has received grants and consulting fees from Gilead Sciences Inc and grants from Intercept Pharmaceuticals outside the submitted work. Dr. Lok has received grants from Target and consultant fees from Abbott, Ambys, Arbutus, Chroma, Clear B, Enanta, Enochian, GNI, GlaxoSmithKline, Eli Lilly, and Virion outside the submitted work. Coauthors have received grants, consulting fees, or personal fees from Bayer, Boston Scientific, Exact Sciences, Fujifilm Medical Sciences, Gilead Sciences, Glycotest, Redhill Biopharma, Target RWE, MedEd Design, Pontifax, Global Life, the Lynx Group, AstraZeneca, Eisai, Novartis Venture Fund, Grail, QED Therapeutics, Genentech, Hepion Pharmaceuticals, Roche, Abbott, AbbVie, and Pfizer. Dr. Kim has received grants from Gilead Sciences (paid to her institution) outside the submitted work.
A version of this article first appeared on Medscape.com.
Researchers studying differences in treatment initiation for chronic hepatitis B (CHB) among a large, multiracial cohort in North America did not find evidence of disparities by race or socioeconomic status.
.
That gap suggests that treatment guidelines need to be simplified and that efforts to increase hepatitis B virus (HBV) awareness and train more clinicians are needed to achieve the World Health Organization’s goal of eliminating HBV by 2030, the researchers write.
The Hepatitis B Research Network study was published online in JAMA Network Open.
The prevalence of CHB in the United States is estimated at 2.4 million. It disproportionately affects persons of Asian or African descent, the investigators note. Their study examined whether treatment initiation and outcomes differ between African American and Black, Asian, and White participants, as well as between African American and Black participants born in North America and East or West Africa.
The research involved 1,550 adult patients: 1,157 Asian American, 193 African American or Black (39 born in the United States, 90 in East Africa, 53 in West Africa, and 11 elsewhere), 157 White, and 43 who identified as being of “other races.” All had CHB but were not receiving antiviral treatment at enrollment.
Participants came from 20 centers in the United States and one in Canada. They underwent clinical and laboratory assessments and could receive anti-HBV treatment after they enrolled. Enrollment was from Jan. 14, 2011, to Jan. 28, 2018. Participants were followed at 12 and 24 weeks and every 24 weeks thereafter in the longitudinal cohort study by Mandana Khalili, MD, division of gastroenterology and hepatology, University of California, San Francisco, and colleagues.
Information on patients’ country of birth, duration of U.S. or Canadian residency, educational level, employment, insurance, prior antiviral treatment, family history of HBV or hepatocellular carcinoma (HCC), and mode of transmission were collected by research coordinators.
Treatment initiation
During the study period, slightly fewer than one-third (32.5%) of the participants initiated treatment. The incidences were 4.8 per 100 person-years in African American or Black participants, 9.9 per 100 person-years in Asian participants, 6.6 per 100 person-years in White participants, and 7.9 per 100 person-years in those of other races (P < .001).
A lower percentage of African American and Black participants (14%) met the American Association for the Study of Liver Diseases treatment criteria, compared with Asian (22%) and White (27%) participants (P = .01).
When the researchers compared cumulative probability of initiating treatment by race for those who met criteria for treatment, they found no significant differences by race.
At 72 weeks, initiation probability was 0.45 for African American and Black patients and 0.51 for Asian and White patients (P = .68). Similarly, among African American and Black participants who met treatment criteria, there were no significant differences in cumulative probability of treatment by region of birth.
The cumulative percentage of treatment initiation for those who met guideline-based criteria was 62%.
“Among participants with a treatment indication, treatment rates did not differ significantly by race, despite marked differences in educational level, income, and type of health care insurance across the racial groups,” the researchers write. “Moreover, race was not an independent estimator of treatment initiation when adjusting for known factors associated with a higher risk of adverse clinical outcomes, namely, HBV DNA, disease severity, sex, and age.”
Adverse liver outcomes (hepatic decompensation, HCC, liver transplant, and death) were rare and did not vary significantly by race, the researchers write.
One study limitation is that participants were linked to specialty liver clinics, so the findings may not be generalizable to patients who receive care in other settings, the authors note.
The results are “reassuring,” said senior author Anna S. Lok, MD, division of gastroenterology and hepatology at University of Michigan in Ann Arbor. However, she noted, study participants had already overcome barriers to receiving care at major academic centers.
“Once you get into the big academic liver centers, then maybe everything is equal, but in the real world, a lot of people don’t ever get to the big liver centers,” she said. The question becomes: “Are we serving only a portion of the patient population?”
Many factors drive the decision to undergo treatment, including the doctor’s opinion as to need and the patient’s desire to receive treatment, she said.
The study participants who were more likely to get treated were those with higher-level disease who had a stronger indication for treatment, Dr. Lok said.
Finding the disparities
Centers for Disease Control and Prevention statistics show that Black people are 3.9 times more likely to have CHB and 2.5 times more likely to die from it than White people, notes H. Nina Kim, MD, with the department of medicine, University of Washington, Seattle, in an accompanying invited commentary.
“The fact that we have not observed racial disparities in treatment initiation does not mean none exist; it means we have to look harder to find them,” she writes.
“We need to examine whether our guidelines for HBV treatment are so complex that it becomes the purview of specialists, thereby restricting access and deepening inequities,” Dr. Kim adds. “We should look closely at retention in care, the step that precedes treatment, and stratify this outcome by race and ethnicity.”
Primary care physicians in some regions might find it difficult to manage patients who have hepatitis B because they see so few of them, Dr. Lok noted.
Dr. Khalili has received grants and consulting fees from Gilead Sciences Inc and grants from Intercept Pharmaceuticals outside the submitted work. Dr. Lok has received grants from Target and consultant fees from Abbott, Ambys, Arbutus, Chroma, Clear B, Enanta, Enochian, GNI, GlaxoSmithKline, Eli Lilly, and Virion outside the submitted work. Coauthors have received grants, consulting fees, or personal fees from Bayer, Boston Scientific, Exact Sciences, Fujifilm Medical Sciences, Gilead Sciences, Glycotest, Redhill Biopharma, Target RWE, MedEd Design, Pontifax, Global Life, the Lynx Group, AstraZeneca, Eisai, Novartis Venture Fund, Grail, QED Therapeutics, Genentech, Hepion Pharmaceuticals, Roche, Abbott, AbbVie, and Pfizer. Dr. Kim has received grants from Gilead Sciences (paid to her institution) outside the submitted work.
A version of this article first appeared on Medscape.com.
Researchers studying differences in treatment initiation for chronic hepatitis B (CHB) among a large, multiracial cohort in North America did not find evidence of disparities by race or socioeconomic status.
.
That gap suggests that treatment guidelines need to be simplified and that efforts to increase hepatitis B virus (HBV) awareness and train more clinicians are needed to achieve the World Health Organization’s goal of eliminating HBV by 2030, the researchers write.
The Hepatitis B Research Network study was published online in JAMA Network Open.
The prevalence of CHB in the United States is estimated at 2.4 million. It disproportionately affects persons of Asian or African descent, the investigators note. Their study examined whether treatment initiation and outcomes differ between African American and Black, Asian, and White participants, as well as between African American and Black participants born in North America and East or West Africa.
The research involved 1,550 adult patients: 1,157 Asian American, 193 African American or Black (39 born in the United States, 90 in East Africa, 53 in West Africa, and 11 elsewhere), 157 White, and 43 who identified as being of “other races.” All had CHB but were not receiving antiviral treatment at enrollment.
Participants came from 20 centers in the United States and one in Canada. They underwent clinical and laboratory assessments and could receive anti-HBV treatment after they enrolled. Enrollment was from Jan. 14, 2011, to Jan. 28, 2018. Participants were followed at 12 and 24 weeks and every 24 weeks thereafter in the longitudinal cohort study by Mandana Khalili, MD, division of gastroenterology and hepatology, University of California, San Francisco, and colleagues.
Information on patients’ country of birth, duration of U.S. or Canadian residency, educational level, employment, insurance, prior antiviral treatment, family history of HBV or hepatocellular carcinoma (HCC), and mode of transmission were collected by research coordinators.
Treatment initiation
During the study period, slightly fewer than one-third (32.5%) of the participants initiated treatment. The incidences were 4.8 per 100 person-years in African American or Black participants, 9.9 per 100 person-years in Asian participants, 6.6 per 100 person-years in White participants, and 7.9 per 100 person-years in those of other races (P < .001).
A lower percentage of African American and Black participants (14%) met the American Association for the Study of Liver Diseases treatment criteria, compared with Asian (22%) and White (27%) participants (P = .01).
When the researchers compared cumulative probability of initiating treatment by race for those who met criteria for treatment, they found no significant differences by race.
At 72 weeks, initiation probability was 0.45 for African American and Black patients and 0.51 for Asian and White patients (P = .68). Similarly, among African American and Black participants who met treatment criteria, there were no significant differences in cumulative probability of treatment by region of birth.
The cumulative percentage of treatment initiation for those who met guideline-based criteria was 62%.
“Among participants with a treatment indication, treatment rates did not differ significantly by race, despite marked differences in educational level, income, and type of health care insurance across the racial groups,” the researchers write. “Moreover, race was not an independent estimator of treatment initiation when adjusting for known factors associated with a higher risk of adverse clinical outcomes, namely, HBV DNA, disease severity, sex, and age.”
Adverse liver outcomes (hepatic decompensation, HCC, liver transplant, and death) were rare and did not vary significantly by race, the researchers write.
One study limitation is that participants were linked to specialty liver clinics, so the findings may not be generalizable to patients who receive care in other settings, the authors note.
The results are “reassuring,” said senior author Anna S. Lok, MD, division of gastroenterology and hepatology at University of Michigan in Ann Arbor. However, she noted, study participants had already overcome barriers to receiving care at major academic centers.
“Once you get into the big academic liver centers, then maybe everything is equal, but in the real world, a lot of people don’t ever get to the big liver centers,” she said. The question becomes: “Are we serving only a portion of the patient population?”
Many factors drive the decision to undergo treatment, including the doctor’s opinion as to need and the patient’s desire to receive treatment, she said.
The study participants who were more likely to get treated were those with higher-level disease who had a stronger indication for treatment, Dr. Lok said.
Finding the disparities
Centers for Disease Control and Prevention statistics show that Black people are 3.9 times more likely to have CHB and 2.5 times more likely to die from it than White people, notes H. Nina Kim, MD, with the department of medicine, University of Washington, Seattle, in an accompanying invited commentary.
“The fact that we have not observed racial disparities in treatment initiation does not mean none exist; it means we have to look harder to find them,” she writes.
“We need to examine whether our guidelines for HBV treatment are so complex that it becomes the purview of specialists, thereby restricting access and deepening inequities,” Dr. Kim adds. “We should look closely at retention in care, the step that precedes treatment, and stratify this outcome by race and ethnicity.”
Primary care physicians in some regions might find it difficult to manage patients who have hepatitis B because they see so few of them, Dr. Lok noted.
Dr. Khalili has received grants and consulting fees from Gilead Sciences Inc and grants from Intercept Pharmaceuticals outside the submitted work. Dr. Lok has received grants from Target and consultant fees from Abbott, Ambys, Arbutus, Chroma, Clear B, Enanta, Enochian, GNI, GlaxoSmithKline, Eli Lilly, and Virion outside the submitted work. Coauthors have received grants, consulting fees, or personal fees from Bayer, Boston Scientific, Exact Sciences, Fujifilm Medical Sciences, Gilead Sciences, Glycotest, Redhill Biopharma, Target RWE, MedEd Design, Pontifax, Global Life, the Lynx Group, AstraZeneca, Eisai, Novartis Venture Fund, Grail, QED Therapeutics, Genentech, Hepion Pharmaceuticals, Roche, Abbott, AbbVie, and Pfizer. Dr. Kim has received grants from Gilead Sciences (paid to her institution) outside the submitted work.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
High-Grade Staphylococcus lugdunensis Bacteremia in a Patient on Home Hemodialysis
Staphylococcus lugdunensis (S lugdunensis) is a species of coagulase-negative Staphylococcus (CoNS) and a constituent of human skin flora. Unlike other strains of CoNS, however, S lugdunensis has gained notoriety for virulence that resembles Staphylococcus aureus (S aureus). S lugdunensis is now recognized as an important nosocomial pathogen and cause of prosthetic device infections, including vascular catheter infections. We present a case of persistent S lugdunensis bacteremia occurring in a patient on hemodialysis (HD) without any implanted prosthetic materials.
Case Presentation
A 60-year-old man with a history of uncontrolled type 2 diabetes mellitus (T2DM) and end-stage renal disease on home HD via arteriovenous fistula (AVF) presented to the emergency department (ED) for evaluation of subacute progressive low back pain. His symptoms began abruptly 2 weeks prior to presentation without any identifiable trigger or trauma. His pain localized to the lower thoracic spine, radiating anteriorly into his abdomen. He reported tactile fever for several days before presentation but no chills, night sweats, paresthesia, weakness, or bowel/bladder incontinence. He had no recent surgeries, implanted hardware, or invasive procedures involving the spine. HD was performed 5 times a week at home with a family member cannulating his AVF via buttonhole technique. He initially sought evaluation in a community hospital several days prior, where he underwent magnetic resonance imaging (MRI) of the thoracic spine. He was discharged from the community ED with oral opioids prior to the MRI results. He presented to West Los Angeles Veterans Affairs Medical Center (WLAVAMC) ED when MRI results came back indicating abnormalities and he reported recalcitrant pain.
On arrival at WLAVAMC, the patient was afebrile with a heart rate of 107 bpm and blood pressure of 152/97 mm Hg. The remainder of his vital signs were normal. The physical examination revealed midline tenderness on palpation of the distal thoracic and proximal lumbar spine. Muscle strength was 4 of 5 in the bilateral hip flexors, though this was limited by pain. The remainder of his neurologic examination was nonfocal. The cardiac examination was unremarkable with no murmurs auscultated. His left upper extremity AVF had an audible bruit and palpable thrill. The skin examination was notable for acanthosis nigricans but no areas of skin erythema or induration and no obvious stigmata of infective endocarditis.
The initial laboratory workup was remarkable for a white blood cell (WBC) count of 10.0 × 103/µL with left shift, blood urea nitrogen level of 59 mg/dL, and creatinine level of 9.3 mg/dL. The patient’s erythrocyte sedimentation rate (ESR) was 45 mm/h (reference range, ≤ 20 mm/h) and C-reactive protein level was > 8.0 mg/L (reference range, ≤ 0.74 mg/L). Two months prior the hemoglobin A1c had been recorded at 9.9%.
Given his intractable low back pain and elevated inflammatory markers, the patient underwent an MRI of the thoracic and lumbar spine with contrast while in the ED. This MRI revealed abnormal marrow edema in the T11-T12 vertebrae with abnormal fluid signal in the T11-T12 disc space. Subjacent paravertebral edema also was noted. There was no well-defined fluid collection or abnormal signal in the spinal cord. Taken together, these findings were concerning for T11-T12 discitis with osteomyelitis.
Two sets of blood cultures were obtained, and empiric IV vancomycin and ceftriaxone were started. Interventional radiology was consulted for consideration of vertebral biopsy but deferred while awaiting blood culture data. Neurosurgery also was consulted and recommended nonoperative management given his nonfocal neurologic examination and imaging without evidence of abscess. Both sets of blood cultures collected on admission later grew methicillin-sensitive S lugdunensis, a species of CoNS. A transthoracic and later transesophageal echocardiogram did not show any valvular vegetations. The patient’s antibiotic regimen was narrowed to IV oxacillin based on susceptibility data. It was later discovered that both blood cultures obtained during his outside ED encounter were also growing S lugdunensis.
The patient’s S lugdunensis bacteremia persisted for the first 8 days of his admission despite appropriate dosing of oxacillin. During this time, the patient remained afebrile with stable vital signs and a normal WBC count. Positron emission tomography was obtained to evaluate for potential sources of his persistent bacteremia. Aside from tracer uptake in the T11-T12 vertebral bodies and intervertebral disc space, no other areas showed suspicious uptake. Neurosurgery reevaluated the patient and again recommended nonoperative management. Blood cultures cleared and based on recommendations from an infectious disease specialist, the patient was transitioned to IV cefazolin dosed 3 times weekly after HD, which was transitioned to an outpatient dialysis center. The patient continued taking cefazolin for 6 weeks with subsequent improvement in back pain and normalization of inflammatory markers at outpatient follow-up.
Discussion
CoNS are a major contributor to human skin flora, a common contaminant of blood cultures, and an important cause of nosocomial bloodstream infections.1,2 These species have a predilection for forming biofilms, making CoNS a major cause of prosthetic device infections.3S lugdunensis is a CoNS species that was first described in 1988.4 In addition to foreign body–related infections, S lugdunensis has been implicated in bone/joint infections, native valve endocarditis, toxic shock syndrome, and brain abscesses.5-8 Infections due to S lugdunensis are notorious for their aggressive and fulminant courses. With its increased virulence that is atypical of other CoNS, S lugdunensis has understandably been likened more to S aureus.
Prior cases have been reported of S lugdunensis bacteremia in patients using HD. However, the suspected source of bacteremia in these cases has generally been central venous catheters.9-12
Notably, our patient’s AVF was accessed using the buttonhole technique for his home HD sessions, which involves cannulating the same site along the fistula until an epithelialized track has formed from scar tissue. At later HD sessions, duller needles can then be used to cannulate this same track. In contrast, the rope-ladder technique involves cannulating a different site along the fistula until the entire length of the fistula has been used. Patients report higher levels of satisfaction with the buttonhole technique, citing decreased pain, decreased oozing, and the perception of easier cannulation by HD nurses.14 However, the buttonhole technique also appears to confer a higher risk of vascular access-related bloodstream infection when compared with the rope-ladder technique.13,15,16
The buttonhole technique is hypothesized to increase infection risk due to the repeated use of the same site for needle entry. Skin flora, including CoNS, may colonize the scab that forms after dialysis access. If proper sterilization techniques are not rigorously followed, the bacteria colonizing the scab and adjacent skin may be introduced into a patient’s bloodstream during needle puncture. Loss of skin integrity due to frequent cannulation of the same site may also contribute to this increased infection risk. It is relevant to recall that our patient received HD 5 times weekly using the buttonhole technique. The use of the buttonhole technique, frequency of his HD sessions, unclear sterilization methods, and immune dysfunction related to his uncontrolled T2DM and renal disease all likely contributed to our patient’s bacteremia.
Using topical mupirocin for prophylaxis at the intended buttonhole puncture site has shown promising results in decreasing rates of S aureus bacteremia.17 It is unclear whether this intervention also would be effective against S lugdunensis. Increasing rates of mupirocin resistance have been reported among S lugdunensis isolates in dialysis settings, but further research in this area is warranted.18
There are no established treatment guidelines for S lugdunensis infections. In vitro studies suggest that S lugdunensis is susceptible to a wide variety of antibiotics. The mecA gene is a major determinant of methicillin resistance that is commonly observed among CoNS but is uncommonly seen with S lugdunensis.5 In a study by Tan and colleagues of 106 S lugdunensis isolates, they found that only 5 (4.7%) were mecA positive.19
Vancomycin is generally reasonable for empiric antibiotic coverage of staphylococci while speciation is pending. However, if S lugdunensis is isolated, its favorable susceptibility pattern typically allows for de-escalation to an antistaphylococcal β-lactam, such as oxacillin or nafcillin. In cases of bloodstream infections caused by methicillin-sensitive S aureus, treatment with a β-lactam has demonstrated superiority over vancomycin due to the lower rates of treatment failure and mortality with β-lactams.20,21 It is unknown whether β-lactams is superior for treating bacteremia with methicillin-sensitive S lugdunensis.
Our patient’s isolate of S lugdunensis was pansensitive to all antibiotics tested, including penicillin. These susceptibility data were used to guide the de-escalation of his empiric vancomycin and ceftriaxone to oxacillin on hospital day 1.
Due to their virulence, bloodstream infections caused by S aureus and S lugdunensis often require more than timely antimicrobial treatment to ensure eradication. Consultation with an infectious disease specialist to manage patients with S aureus bacteremia has been proven to reduce mortality.25 A similar mortality benefit is seen when infectious disease specialists are consulted for S lugdunensis bacteremia.26 This mortality benefit is likely explained by S lugdunensis’ propensity to cause aggressive, metastatic infections. In such cases, infectious disease consultants may recommend additional imaging (eg, transthoracic echocardiogram) to evaluate for occult sources of infection, advocate for appropriate source control, and guide the selection of an appropriate antibiotic course to ensure resolution of the bacteremia.
Conclusions
S lugdunensis is an increasingly recognized cause of nosocomial bloodstream infections. Given the commonalities in virulence that S lugdunensis shares with S aureus, treatment of bacteremia caused by either species should follow similar management principles: prompt initiation of IV antistaphylococcal therapy, a thorough evaluation for the source(s) of bacteremia as well as metastatic complications, and consultation with an infectious disease specialist. This case report also highlights the importance of considering a patient’s AVF as a potential source for infection even in the absence of localized signs of infection. The buttonhole method of AVF cannulation was thought to be a major contributor to the development and persistence of our patient’s bacteremia. This risk should be discussed with patients using a shared decision-making approach when developing a dialysis treatment plan.
1. Huebner J, Goldmann DA. Coagulase-negative staphylococci: role as pathogens. Annu Rev Med. 1999;50(1):223-236. doi:10.1146/annurev.med.50.1.223
2. Beekmann SE, Diekema DJ, Doern GV. Determining the clinical significance of coagulase-negative staphylococci isolated from blood cultures. Infect Control Hosp Epidemiol. 2005;26(6):559-566. doi:10.1086/502584
3. Arrecubieta C, Toba FA, von Bayern M, et al. SdrF, a Staphylococcus epidermidis surface protein, contributes to the initiation of ventricular assist device driveline–related infections. PLoS Pathog. 2009;5(5):e1000411. doi.10.1371/journal.ppat.1000411
4. Freney J, Brun Y, Bes M, et al. Staphylococcus lugdunensis sp. nov. and Staphylococcus schleiferi sp. nov., two species from human clinical specimens. Int J Syst Bacteriol. 1988;38(2):168-172. doi:10.1099/00207713-38-2-168
5. Frank KL, del Pozo JL, Patel R. From clinical microbiology to infection pathogenesis: how daring to be different works for Staphylococcus lugdunensis. Clin Microbiol Rev. 2008;21(1):111-133. doi:10.1128/CMR.00036-07
6. Anguera I, Del Río A, Miró JM; Hospital Clinic Endocarditis Study Group. Staphylococcus lugdunensis infective endocarditis: description of 10 cases and analysis of native valve, prosthetic valve, and pacemaker lead endocarditis clinical profiles. Heart. 2005;91(2):e10. doi:10.1136/hrt.2004.040659
7. Pareja J, Gupta K, Koziel H. The toxic shock syndrome and Staphylococcus lugdunensis bacteremia. Ann Intern Med. 1998;128(7):603-604. doi:10.7326/0003-4819-128-7-199804010-00029
8. Woznowski M, Quack I, Bölke E, et al. Fulminant Staphylococcus lugdunensis septicaemia following a pelvic varicella-zoster virus infection in an immune-deficient patient: a case report. Eur J Med Res. 201;15(9):410-414. doi:10.1186/2047-783x-15-9-410
9. Mallappallil M, Salifu M, Woredekal Y, et al. Staphylococcus lugdunensis bacteremia in hemodialysis patients. Int J Microbiol Res. 2012;4(2):178-181. doi:10.9735/0975-5276.4.2.178-181
10. Shuttleworth R, Colby W. Staphylococcus lugdunensis endocarditis. J Clin Microbiol. 1992;30(8):5. doi:10.1128/jcm.30.8.1948-1952.1992
11. Conner RC, Byrnes TJ, Clough LA, Myers JP. Staphylococcus lugdunensis tricuspid valve endocarditis associated with home hemodialysis therapy: report of a case and review of the literature. Infect Dis Clin Pract. 2012;20(3):182-183. doi:1097/IPC.0b013e318245d4f1
12. Kamaraju S, Nelson K, Williams D, Ayenew W, Modi K. Staphylococcus lugdunensis pulmonary valve endocarditis in a patient on chronic hemodialysis. Am J Nephrol. 1999;19(5):605-608. doi:1097/IPC.0b013e318245d4f1
13. Lok C, Sontrop J, Faratro R, Chan C, Zimmerman DL. Frequent hemodialysis fistula infectious complications. Nephron Extra. 2014;4(3):159-167. doi:10.1159/000366477
14. Hashmi A, Cheema MQ, Moss AH. Hemodialysis patients’ experience with and attitudes toward the buttonhole technique for arteriovenous fistula cannulation. Clin Nephrol. 2010;74(5):346-350. doi:10.5414/cnp74346
15. Lyman M, Nguyen DB, Shugart A, Gruhler H, Lines C, Patel PR. Risk of vascular access infection associated with buttonhole cannulation of fistulas: data from the National Healthcare Safety Network. Am J Kidney Dis. 2020;76(1):82-89. doi:10.1053/j.ajkd.2019.11.006
16. MacRae JM, Ahmed SB, Atkar R, Hemmelgarn BR. A randomized trial comparing buttonhole with rope ladder needling in conventional hemodialysis patients. Clin J Am Soc Nephrol. 2012;7(10):1632-1638. doi:10.2215/CJN.02730312
17. Nesrallah GE, Cuerden M, Wong JHS, Pierratos A. Staphylococcus aureus bacteremia and buttonhole cannulation: long-term safety and efficacy of mupirocin prophylaxis. Clin J Am Soc Nephrol. 2010;5(6):1047-1053. doi:10.2215/CJN.00280110
18. Ho PL, Liu MCJ, Chow KH, et al. Emergence of ileS2 -carrying, multidrug-resistant plasmids in Staphylococcus lugdunensis. Antimicrob Agents Chemother. 2016;60(10):6411-6414. doi:10.1128/AAC.00948-16
19. Tan TY, Ng SY, He J. Microbiological characteristics, presumptive identification, and antibiotic susceptibilities of Staphylococcus lugdunensis. J Clin Microbiol. 2008;46(7):2393-2395. doi:10.1128/JCM.00740-08
20. Chang FY, Peacock JE, Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore). 2003;82(5):333-339. doi:10.1097/01.md.0000091184.93122.09
21. Shurland S, Zhan M, Bradham DD, Roghmann MC. Comparison of mortality risk associated with bacteremia due to methicillin-resistant and methicillin-susceptible Staphylococcus aureus. Infect Control Hosp Epidemiol. 2007;28(3):273-279. doi:10.1086/512627
22. Levine DP, Fromm BS, Reddy BR. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann Intern Med. 1991;115(9):674. doi:10.7326/0003-4819-115-9-674
23. Fowler VG, Karchmer AW, Tally FP, et al; S. aureus Endocarditis and Bacteremia Study Group. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355(7):653-665 . doi:10.1056/NEJMoa053783
24. Duhon B, Dallas S, Velasquez ST, Hand E. Staphylococcus lugdunensis bacteremia and endocarditis treated with cefazolin and rifampin. Am J Health Syst Pharm. 2015;72(13):1114-1118. doi:10.2146/ajhp140498
25. Lahey T, Shah R, Gittzus J, Schwartzman J, Kirkland K. Infectious diseases consultation lowers mortality from Staphylococcus aureus bacteremia. Medicine (Baltimore). 2009;88(5):263-267. doi:10.1097/MD.0b013e3181b8fccb
26. Forsblom E, Högnäs E, Syrjänen J, Järvinen A. Infectious diseases specialist consultation in Staphylococcus lugdunensis bacteremia. PLoS ONE. 2021;16(10):e0258511. doi:10.1371/journal.pone.0258511
Staphylococcus lugdunensis (S lugdunensis) is a species of coagulase-negative Staphylococcus (CoNS) and a constituent of human skin flora. Unlike other strains of CoNS, however, S lugdunensis has gained notoriety for virulence that resembles Staphylococcus aureus (S aureus). S lugdunensis is now recognized as an important nosocomial pathogen and cause of prosthetic device infections, including vascular catheter infections. We present a case of persistent S lugdunensis bacteremia occurring in a patient on hemodialysis (HD) without any implanted prosthetic materials.
Case Presentation
A 60-year-old man with a history of uncontrolled type 2 diabetes mellitus (T2DM) and end-stage renal disease on home HD via arteriovenous fistula (AVF) presented to the emergency department (ED) for evaluation of subacute progressive low back pain. His symptoms began abruptly 2 weeks prior to presentation without any identifiable trigger or trauma. His pain localized to the lower thoracic spine, radiating anteriorly into his abdomen. He reported tactile fever for several days before presentation but no chills, night sweats, paresthesia, weakness, or bowel/bladder incontinence. He had no recent surgeries, implanted hardware, or invasive procedures involving the spine. HD was performed 5 times a week at home with a family member cannulating his AVF via buttonhole technique. He initially sought evaluation in a community hospital several days prior, where he underwent magnetic resonance imaging (MRI) of the thoracic spine. He was discharged from the community ED with oral opioids prior to the MRI results. He presented to West Los Angeles Veterans Affairs Medical Center (WLAVAMC) ED when MRI results came back indicating abnormalities and he reported recalcitrant pain.
On arrival at WLAVAMC, the patient was afebrile with a heart rate of 107 bpm and blood pressure of 152/97 mm Hg. The remainder of his vital signs were normal. The physical examination revealed midline tenderness on palpation of the distal thoracic and proximal lumbar spine. Muscle strength was 4 of 5 in the bilateral hip flexors, though this was limited by pain. The remainder of his neurologic examination was nonfocal. The cardiac examination was unremarkable with no murmurs auscultated. His left upper extremity AVF had an audible bruit and palpable thrill. The skin examination was notable for acanthosis nigricans but no areas of skin erythema or induration and no obvious stigmata of infective endocarditis.
The initial laboratory workup was remarkable for a white blood cell (WBC) count of 10.0 × 103/µL with left shift, blood urea nitrogen level of 59 mg/dL, and creatinine level of 9.3 mg/dL. The patient’s erythrocyte sedimentation rate (ESR) was 45 mm/h (reference range, ≤ 20 mm/h) and C-reactive protein level was > 8.0 mg/L (reference range, ≤ 0.74 mg/L). Two months prior the hemoglobin A1c had been recorded at 9.9%.
Given his intractable low back pain and elevated inflammatory markers, the patient underwent an MRI of the thoracic and lumbar spine with contrast while in the ED. This MRI revealed abnormal marrow edema in the T11-T12 vertebrae with abnormal fluid signal in the T11-T12 disc space. Subjacent paravertebral edema also was noted. There was no well-defined fluid collection or abnormal signal in the spinal cord. Taken together, these findings were concerning for T11-T12 discitis with osteomyelitis.
Two sets of blood cultures were obtained, and empiric IV vancomycin and ceftriaxone were started. Interventional radiology was consulted for consideration of vertebral biopsy but deferred while awaiting blood culture data. Neurosurgery also was consulted and recommended nonoperative management given his nonfocal neurologic examination and imaging without evidence of abscess. Both sets of blood cultures collected on admission later grew methicillin-sensitive S lugdunensis, a species of CoNS. A transthoracic and later transesophageal echocardiogram did not show any valvular vegetations. The patient’s antibiotic regimen was narrowed to IV oxacillin based on susceptibility data. It was later discovered that both blood cultures obtained during his outside ED encounter were also growing S lugdunensis.
The patient’s S lugdunensis bacteremia persisted for the first 8 days of his admission despite appropriate dosing of oxacillin. During this time, the patient remained afebrile with stable vital signs and a normal WBC count. Positron emission tomography was obtained to evaluate for potential sources of his persistent bacteremia. Aside from tracer uptake in the T11-T12 vertebral bodies and intervertebral disc space, no other areas showed suspicious uptake. Neurosurgery reevaluated the patient and again recommended nonoperative management. Blood cultures cleared and based on recommendations from an infectious disease specialist, the patient was transitioned to IV cefazolin dosed 3 times weekly after HD, which was transitioned to an outpatient dialysis center. The patient continued taking cefazolin for 6 weeks with subsequent improvement in back pain and normalization of inflammatory markers at outpatient follow-up.
Discussion
CoNS are a major contributor to human skin flora, a common contaminant of blood cultures, and an important cause of nosocomial bloodstream infections.1,2 These species have a predilection for forming biofilms, making CoNS a major cause of prosthetic device infections.3S lugdunensis is a CoNS species that was first described in 1988.4 In addition to foreign body–related infections, S lugdunensis has been implicated in bone/joint infections, native valve endocarditis, toxic shock syndrome, and brain abscesses.5-8 Infections due to S lugdunensis are notorious for their aggressive and fulminant courses. With its increased virulence that is atypical of other CoNS, S lugdunensis has understandably been likened more to S aureus.
Prior cases have been reported of S lugdunensis bacteremia in patients using HD. However, the suspected source of bacteremia in these cases has generally been central venous catheters.9-12
Notably, our patient’s AVF was accessed using the buttonhole technique for his home HD sessions, which involves cannulating the same site along the fistula until an epithelialized track has formed from scar tissue. At later HD sessions, duller needles can then be used to cannulate this same track. In contrast, the rope-ladder technique involves cannulating a different site along the fistula until the entire length of the fistula has been used. Patients report higher levels of satisfaction with the buttonhole technique, citing decreased pain, decreased oozing, and the perception of easier cannulation by HD nurses.14 However, the buttonhole technique also appears to confer a higher risk of vascular access-related bloodstream infection when compared with the rope-ladder technique.13,15,16
The buttonhole technique is hypothesized to increase infection risk due to the repeated use of the same site for needle entry. Skin flora, including CoNS, may colonize the scab that forms after dialysis access. If proper sterilization techniques are not rigorously followed, the bacteria colonizing the scab and adjacent skin may be introduced into a patient’s bloodstream during needle puncture. Loss of skin integrity due to frequent cannulation of the same site may also contribute to this increased infection risk. It is relevant to recall that our patient received HD 5 times weekly using the buttonhole technique. The use of the buttonhole technique, frequency of his HD sessions, unclear sterilization methods, and immune dysfunction related to his uncontrolled T2DM and renal disease all likely contributed to our patient’s bacteremia.
Using topical mupirocin for prophylaxis at the intended buttonhole puncture site has shown promising results in decreasing rates of S aureus bacteremia.17 It is unclear whether this intervention also would be effective against S lugdunensis. Increasing rates of mupirocin resistance have been reported among S lugdunensis isolates in dialysis settings, but further research in this area is warranted.18
There are no established treatment guidelines for S lugdunensis infections. In vitro studies suggest that S lugdunensis is susceptible to a wide variety of antibiotics. The mecA gene is a major determinant of methicillin resistance that is commonly observed among CoNS but is uncommonly seen with S lugdunensis.5 In a study by Tan and colleagues of 106 S lugdunensis isolates, they found that only 5 (4.7%) were mecA positive.19
Vancomycin is generally reasonable for empiric antibiotic coverage of staphylococci while speciation is pending. However, if S lugdunensis is isolated, its favorable susceptibility pattern typically allows for de-escalation to an antistaphylococcal β-lactam, such as oxacillin or nafcillin. In cases of bloodstream infections caused by methicillin-sensitive S aureus, treatment with a β-lactam has demonstrated superiority over vancomycin due to the lower rates of treatment failure and mortality with β-lactams.20,21 It is unknown whether β-lactams is superior for treating bacteremia with methicillin-sensitive S lugdunensis.
Our patient’s isolate of S lugdunensis was pansensitive to all antibiotics tested, including penicillin. These susceptibility data were used to guide the de-escalation of his empiric vancomycin and ceftriaxone to oxacillin on hospital day 1.
Due to their virulence, bloodstream infections caused by S aureus and S lugdunensis often require more than timely antimicrobial treatment to ensure eradication. Consultation with an infectious disease specialist to manage patients with S aureus bacteremia has been proven to reduce mortality.25 A similar mortality benefit is seen when infectious disease specialists are consulted for S lugdunensis bacteremia.26 This mortality benefit is likely explained by S lugdunensis’ propensity to cause aggressive, metastatic infections. In such cases, infectious disease consultants may recommend additional imaging (eg, transthoracic echocardiogram) to evaluate for occult sources of infection, advocate for appropriate source control, and guide the selection of an appropriate antibiotic course to ensure resolution of the bacteremia.
Conclusions
S lugdunensis is an increasingly recognized cause of nosocomial bloodstream infections. Given the commonalities in virulence that S lugdunensis shares with S aureus, treatment of bacteremia caused by either species should follow similar management principles: prompt initiation of IV antistaphylococcal therapy, a thorough evaluation for the source(s) of bacteremia as well as metastatic complications, and consultation with an infectious disease specialist. This case report also highlights the importance of considering a patient’s AVF as a potential source for infection even in the absence of localized signs of infection. The buttonhole method of AVF cannulation was thought to be a major contributor to the development and persistence of our patient’s bacteremia. This risk should be discussed with patients using a shared decision-making approach when developing a dialysis treatment plan.
Staphylococcus lugdunensis (S lugdunensis) is a species of coagulase-negative Staphylococcus (CoNS) and a constituent of human skin flora. Unlike other strains of CoNS, however, S lugdunensis has gained notoriety for virulence that resembles Staphylococcus aureus (S aureus). S lugdunensis is now recognized as an important nosocomial pathogen and cause of prosthetic device infections, including vascular catheter infections. We present a case of persistent S lugdunensis bacteremia occurring in a patient on hemodialysis (HD) without any implanted prosthetic materials.
Case Presentation
A 60-year-old man with a history of uncontrolled type 2 diabetes mellitus (T2DM) and end-stage renal disease on home HD via arteriovenous fistula (AVF) presented to the emergency department (ED) for evaluation of subacute progressive low back pain. His symptoms began abruptly 2 weeks prior to presentation without any identifiable trigger or trauma. His pain localized to the lower thoracic spine, radiating anteriorly into his abdomen. He reported tactile fever for several days before presentation but no chills, night sweats, paresthesia, weakness, or bowel/bladder incontinence. He had no recent surgeries, implanted hardware, or invasive procedures involving the spine. HD was performed 5 times a week at home with a family member cannulating his AVF via buttonhole technique. He initially sought evaluation in a community hospital several days prior, where he underwent magnetic resonance imaging (MRI) of the thoracic spine. He was discharged from the community ED with oral opioids prior to the MRI results. He presented to West Los Angeles Veterans Affairs Medical Center (WLAVAMC) ED when MRI results came back indicating abnormalities and he reported recalcitrant pain.
On arrival at WLAVAMC, the patient was afebrile with a heart rate of 107 bpm and blood pressure of 152/97 mm Hg. The remainder of his vital signs were normal. The physical examination revealed midline tenderness on palpation of the distal thoracic and proximal lumbar spine. Muscle strength was 4 of 5 in the bilateral hip flexors, though this was limited by pain. The remainder of his neurologic examination was nonfocal. The cardiac examination was unremarkable with no murmurs auscultated. His left upper extremity AVF had an audible bruit and palpable thrill. The skin examination was notable for acanthosis nigricans but no areas of skin erythema or induration and no obvious stigmata of infective endocarditis.
The initial laboratory workup was remarkable for a white blood cell (WBC) count of 10.0 × 103/µL with left shift, blood urea nitrogen level of 59 mg/dL, and creatinine level of 9.3 mg/dL. The patient’s erythrocyte sedimentation rate (ESR) was 45 mm/h (reference range, ≤ 20 mm/h) and C-reactive protein level was > 8.0 mg/L (reference range, ≤ 0.74 mg/L). Two months prior the hemoglobin A1c had been recorded at 9.9%.
Given his intractable low back pain and elevated inflammatory markers, the patient underwent an MRI of the thoracic and lumbar spine with contrast while in the ED. This MRI revealed abnormal marrow edema in the T11-T12 vertebrae with abnormal fluid signal in the T11-T12 disc space. Subjacent paravertebral edema also was noted. There was no well-defined fluid collection or abnormal signal in the spinal cord. Taken together, these findings were concerning for T11-T12 discitis with osteomyelitis.
Two sets of blood cultures were obtained, and empiric IV vancomycin and ceftriaxone were started. Interventional radiology was consulted for consideration of vertebral biopsy but deferred while awaiting blood culture data. Neurosurgery also was consulted and recommended nonoperative management given his nonfocal neurologic examination and imaging without evidence of abscess. Both sets of blood cultures collected on admission later grew methicillin-sensitive S lugdunensis, a species of CoNS. A transthoracic and later transesophageal echocardiogram did not show any valvular vegetations. The patient’s antibiotic regimen was narrowed to IV oxacillin based on susceptibility data. It was later discovered that both blood cultures obtained during his outside ED encounter were also growing S lugdunensis.
The patient’s S lugdunensis bacteremia persisted for the first 8 days of his admission despite appropriate dosing of oxacillin. During this time, the patient remained afebrile with stable vital signs and a normal WBC count. Positron emission tomography was obtained to evaluate for potential sources of his persistent bacteremia. Aside from tracer uptake in the T11-T12 vertebral bodies and intervertebral disc space, no other areas showed suspicious uptake. Neurosurgery reevaluated the patient and again recommended nonoperative management. Blood cultures cleared and based on recommendations from an infectious disease specialist, the patient was transitioned to IV cefazolin dosed 3 times weekly after HD, which was transitioned to an outpatient dialysis center. The patient continued taking cefazolin for 6 weeks with subsequent improvement in back pain and normalization of inflammatory markers at outpatient follow-up.
Discussion
CoNS are a major contributor to human skin flora, a common contaminant of blood cultures, and an important cause of nosocomial bloodstream infections.1,2 These species have a predilection for forming biofilms, making CoNS a major cause of prosthetic device infections.3S lugdunensis is a CoNS species that was first described in 1988.4 In addition to foreign body–related infections, S lugdunensis has been implicated in bone/joint infections, native valve endocarditis, toxic shock syndrome, and brain abscesses.5-8 Infections due to S lugdunensis are notorious for their aggressive and fulminant courses. With its increased virulence that is atypical of other CoNS, S lugdunensis has understandably been likened more to S aureus.
Prior cases have been reported of S lugdunensis bacteremia in patients using HD. However, the suspected source of bacteremia in these cases has generally been central venous catheters.9-12
Notably, our patient’s AVF was accessed using the buttonhole technique for his home HD sessions, which involves cannulating the same site along the fistula until an epithelialized track has formed from scar tissue. At later HD sessions, duller needles can then be used to cannulate this same track. In contrast, the rope-ladder technique involves cannulating a different site along the fistula until the entire length of the fistula has been used. Patients report higher levels of satisfaction with the buttonhole technique, citing decreased pain, decreased oozing, and the perception of easier cannulation by HD nurses.14 However, the buttonhole technique also appears to confer a higher risk of vascular access-related bloodstream infection when compared with the rope-ladder technique.13,15,16
The buttonhole technique is hypothesized to increase infection risk due to the repeated use of the same site for needle entry. Skin flora, including CoNS, may colonize the scab that forms after dialysis access. If proper sterilization techniques are not rigorously followed, the bacteria colonizing the scab and adjacent skin may be introduced into a patient’s bloodstream during needle puncture. Loss of skin integrity due to frequent cannulation of the same site may also contribute to this increased infection risk. It is relevant to recall that our patient received HD 5 times weekly using the buttonhole technique. The use of the buttonhole technique, frequency of his HD sessions, unclear sterilization methods, and immune dysfunction related to his uncontrolled T2DM and renal disease all likely contributed to our patient’s bacteremia.
Using topical mupirocin for prophylaxis at the intended buttonhole puncture site has shown promising results in decreasing rates of S aureus bacteremia.17 It is unclear whether this intervention also would be effective against S lugdunensis. Increasing rates of mupirocin resistance have been reported among S lugdunensis isolates in dialysis settings, but further research in this area is warranted.18
There are no established treatment guidelines for S lugdunensis infections. In vitro studies suggest that S lugdunensis is susceptible to a wide variety of antibiotics. The mecA gene is a major determinant of methicillin resistance that is commonly observed among CoNS but is uncommonly seen with S lugdunensis.5 In a study by Tan and colleagues of 106 S lugdunensis isolates, they found that only 5 (4.7%) were mecA positive.19
Vancomycin is generally reasonable for empiric antibiotic coverage of staphylococci while speciation is pending. However, if S lugdunensis is isolated, its favorable susceptibility pattern typically allows for de-escalation to an antistaphylococcal β-lactam, such as oxacillin or nafcillin. In cases of bloodstream infections caused by methicillin-sensitive S aureus, treatment with a β-lactam has demonstrated superiority over vancomycin due to the lower rates of treatment failure and mortality with β-lactams.20,21 It is unknown whether β-lactams is superior for treating bacteremia with methicillin-sensitive S lugdunensis.
Our patient’s isolate of S lugdunensis was pansensitive to all antibiotics tested, including penicillin. These susceptibility data were used to guide the de-escalation of his empiric vancomycin and ceftriaxone to oxacillin on hospital day 1.
Due to their virulence, bloodstream infections caused by S aureus and S lugdunensis often require more than timely antimicrobial treatment to ensure eradication. Consultation with an infectious disease specialist to manage patients with S aureus bacteremia has been proven to reduce mortality.25 A similar mortality benefit is seen when infectious disease specialists are consulted for S lugdunensis bacteremia.26 This mortality benefit is likely explained by S lugdunensis’ propensity to cause aggressive, metastatic infections. In such cases, infectious disease consultants may recommend additional imaging (eg, transthoracic echocardiogram) to evaluate for occult sources of infection, advocate for appropriate source control, and guide the selection of an appropriate antibiotic course to ensure resolution of the bacteremia.
Conclusions
S lugdunensis is an increasingly recognized cause of nosocomial bloodstream infections. Given the commonalities in virulence that S lugdunensis shares with S aureus, treatment of bacteremia caused by either species should follow similar management principles: prompt initiation of IV antistaphylococcal therapy, a thorough evaluation for the source(s) of bacteremia as well as metastatic complications, and consultation with an infectious disease specialist. This case report also highlights the importance of considering a patient’s AVF as a potential source for infection even in the absence of localized signs of infection. The buttonhole method of AVF cannulation was thought to be a major contributor to the development and persistence of our patient’s bacteremia. This risk should be discussed with patients using a shared decision-making approach when developing a dialysis treatment plan.
1. Huebner J, Goldmann DA. Coagulase-negative staphylococci: role as pathogens. Annu Rev Med. 1999;50(1):223-236. doi:10.1146/annurev.med.50.1.223
2. Beekmann SE, Diekema DJ, Doern GV. Determining the clinical significance of coagulase-negative staphylococci isolated from blood cultures. Infect Control Hosp Epidemiol. 2005;26(6):559-566. doi:10.1086/502584
3. Arrecubieta C, Toba FA, von Bayern M, et al. SdrF, a Staphylococcus epidermidis surface protein, contributes to the initiation of ventricular assist device driveline–related infections. PLoS Pathog. 2009;5(5):e1000411. doi.10.1371/journal.ppat.1000411
4. Freney J, Brun Y, Bes M, et al. Staphylococcus lugdunensis sp. nov. and Staphylococcus schleiferi sp. nov., two species from human clinical specimens. Int J Syst Bacteriol. 1988;38(2):168-172. doi:10.1099/00207713-38-2-168
5. Frank KL, del Pozo JL, Patel R. From clinical microbiology to infection pathogenesis: how daring to be different works for Staphylococcus lugdunensis. Clin Microbiol Rev. 2008;21(1):111-133. doi:10.1128/CMR.00036-07
6. Anguera I, Del Río A, Miró JM; Hospital Clinic Endocarditis Study Group. Staphylococcus lugdunensis infective endocarditis: description of 10 cases and analysis of native valve, prosthetic valve, and pacemaker lead endocarditis clinical profiles. Heart. 2005;91(2):e10. doi:10.1136/hrt.2004.040659
7. Pareja J, Gupta K, Koziel H. The toxic shock syndrome and Staphylococcus lugdunensis bacteremia. Ann Intern Med. 1998;128(7):603-604. doi:10.7326/0003-4819-128-7-199804010-00029
8. Woznowski M, Quack I, Bölke E, et al. Fulminant Staphylococcus lugdunensis septicaemia following a pelvic varicella-zoster virus infection in an immune-deficient patient: a case report. Eur J Med Res. 201;15(9):410-414. doi:10.1186/2047-783x-15-9-410
9. Mallappallil M, Salifu M, Woredekal Y, et al. Staphylococcus lugdunensis bacteremia in hemodialysis patients. Int J Microbiol Res. 2012;4(2):178-181. doi:10.9735/0975-5276.4.2.178-181
10. Shuttleworth R, Colby W. Staphylococcus lugdunensis endocarditis. J Clin Microbiol. 1992;30(8):5. doi:10.1128/jcm.30.8.1948-1952.1992
11. Conner RC, Byrnes TJ, Clough LA, Myers JP. Staphylococcus lugdunensis tricuspid valve endocarditis associated with home hemodialysis therapy: report of a case and review of the literature. Infect Dis Clin Pract. 2012;20(3):182-183. doi:1097/IPC.0b013e318245d4f1
12. Kamaraju S, Nelson K, Williams D, Ayenew W, Modi K. Staphylococcus lugdunensis pulmonary valve endocarditis in a patient on chronic hemodialysis. Am J Nephrol. 1999;19(5):605-608. doi:1097/IPC.0b013e318245d4f1
13. Lok C, Sontrop J, Faratro R, Chan C, Zimmerman DL. Frequent hemodialysis fistula infectious complications. Nephron Extra. 2014;4(3):159-167. doi:10.1159/000366477
14. Hashmi A, Cheema MQ, Moss AH. Hemodialysis patients’ experience with and attitudes toward the buttonhole technique for arteriovenous fistula cannulation. Clin Nephrol. 2010;74(5):346-350. doi:10.5414/cnp74346
15. Lyman M, Nguyen DB, Shugart A, Gruhler H, Lines C, Patel PR. Risk of vascular access infection associated with buttonhole cannulation of fistulas: data from the National Healthcare Safety Network. Am J Kidney Dis. 2020;76(1):82-89. doi:10.1053/j.ajkd.2019.11.006
16. MacRae JM, Ahmed SB, Atkar R, Hemmelgarn BR. A randomized trial comparing buttonhole with rope ladder needling in conventional hemodialysis patients. Clin J Am Soc Nephrol. 2012;7(10):1632-1638. doi:10.2215/CJN.02730312
17. Nesrallah GE, Cuerden M, Wong JHS, Pierratos A. Staphylococcus aureus bacteremia and buttonhole cannulation: long-term safety and efficacy of mupirocin prophylaxis. Clin J Am Soc Nephrol. 2010;5(6):1047-1053. doi:10.2215/CJN.00280110
18. Ho PL, Liu MCJ, Chow KH, et al. Emergence of ileS2 -carrying, multidrug-resistant plasmids in Staphylococcus lugdunensis. Antimicrob Agents Chemother. 2016;60(10):6411-6414. doi:10.1128/AAC.00948-16
19. Tan TY, Ng SY, He J. Microbiological characteristics, presumptive identification, and antibiotic susceptibilities of Staphylococcus lugdunensis. J Clin Microbiol. 2008;46(7):2393-2395. doi:10.1128/JCM.00740-08
20. Chang FY, Peacock JE, Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore). 2003;82(5):333-339. doi:10.1097/01.md.0000091184.93122.09
21. Shurland S, Zhan M, Bradham DD, Roghmann MC. Comparison of mortality risk associated with bacteremia due to methicillin-resistant and methicillin-susceptible Staphylococcus aureus. Infect Control Hosp Epidemiol. 2007;28(3):273-279. doi:10.1086/512627
22. Levine DP, Fromm BS, Reddy BR. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann Intern Med. 1991;115(9):674. doi:10.7326/0003-4819-115-9-674
23. Fowler VG, Karchmer AW, Tally FP, et al; S. aureus Endocarditis and Bacteremia Study Group. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355(7):653-665 . doi:10.1056/NEJMoa053783
24. Duhon B, Dallas S, Velasquez ST, Hand E. Staphylococcus lugdunensis bacteremia and endocarditis treated with cefazolin and rifampin. Am J Health Syst Pharm. 2015;72(13):1114-1118. doi:10.2146/ajhp140498
25. Lahey T, Shah R, Gittzus J, Schwartzman J, Kirkland K. Infectious diseases consultation lowers mortality from Staphylococcus aureus bacteremia. Medicine (Baltimore). 2009;88(5):263-267. doi:10.1097/MD.0b013e3181b8fccb
26. Forsblom E, Högnäs E, Syrjänen J, Järvinen A. Infectious diseases specialist consultation in Staphylococcus lugdunensis bacteremia. PLoS ONE. 2021;16(10):e0258511. doi:10.1371/journal.pone.0258511
1. Huebner J, Goldmann DA. Coagulase-negative staphylococci: role as pathogens. Annu Rev Med. 1999;50(1):223-236. doi:10.1146/annurev.med.50.1.223
2. Beekmann SE, Diekema DJ, Doern GV. Determining the clinical significance of coagulase-negative staphylococci isolated from blood cultures. Infect Control Hosp Epidemiol. 2005;26(6):559-566. doi:10.1086/502584
3. Arrecubieta C, Toba FA, von Bayern M, et al. SdrF, a Staphylococcus epidermidis surface protein, contributes to the initiation of ventricular assist device driveline–related infections. PLoS Pathog. 2009;5(5):e1000411. doi.10.1371/journal.ppat.1000411
4. Freney J, Brun Y, Bes M, et al. Staphylococcus lugdunensis sp. nov. and Staphylococcus schleiferi sp. nov., two species from human clinical specimens. Int J Syst Bacteriol. 1988;38(2):168-172. doi:10.1099/00207713-38-2-168
5. Frank KL, del Pozo JL, Patel R. From clinical microbiology to infection pathogenesis: how daring to be different works for Staphylococcus lugdunensis. Clin Microbiol Rev. 2008;21(1):111-133. doi:10.1128/CMR.00036-07
6. Anguera I, Del Río A, Miró JM; Hospital Clinic Endocarditis Study Group. Staphylococcus lugdunensis infective endocarditis: description of 10 cases and analysis of native valve, prosthetic valve, and pacemaker lead endocarditis clinical profiles. Heart. 2005;91(2):e10. doi:10.1136/hrt.2004.040659
7. Pareja J, Gupta K, Koziel H. The toxic shock syndrome and Staphylococcus lugdunensis bacteremia. Ann Intern Med. 1998;128(7):603-604. doi:10.7326/0003-4819-128-7-199804010-00029
8. Woznowski M, Quack I, Bölke E, et al. Fulminant Staphylococcus lugdunensis septicaemia following a pelvic varicella-zoster virus infection in an immune-deficient patient: a case report. Eur J Med Res. 201;15(9):410-414. doi:10.1186/2047-783x-15-9-410
9. Mallappallil M, Salifu M, Woredekal Y, et al. Staphylococcus lugdunensis bacteremia in hemodialysis patients. Int J Microbiol Res. 2012;4(2):178-181. doi:10.9735/0975-5276.4.2.178-181
10. Shuttleworth R, Colby W. Staphylococcus lugdunensis endocarditis. J Clin Microbiol. 1992;30(8):5. doi:10.1128/jcm.30.8.1948-1952.1992
11. Conner RC, Byrnes TJ, Clough LA, Myers JP. Staphylococcus lugdunensis tricuspid valve endocarditis associated with home hemodialysis therapy: report of a case and review of the literature. Infect Dis Clin Pract. 2012;20(3):182-183. doi:1097/IPC.0b013e318245d4f1
12. Kamaraju S, Nelson K, Williams D, Ayenew W, Modi K. Staphylococcus lugdunensis pulmonary valve endocarditis in a patient on chronic hemodialysis. Am J Nephrol. 1999;19(5):605-608. doi:1097/IPC.0b013e318245d4f1
13. Lok C, Sontrop J, Faratro R, Chan C, Zimmerman DL. Frequent hemodialysis fistula infectious complications. Nephron Extra. 2014;4(3):159-167. doi:10.1159/000366477
14. Hashmi A, Cheema MQ, Moss AH. Hemodialysis patients’ experience with and attitudes toward the buttonhole technique for arteriovenous fistula cannulation. Clin Nephrol. 2010;74(5):346-350. doi:10.5414/cnp74346
15. Lyman M, Nguyen DB, Shugart A, Gruhler H, Lines C, Patel PR. Risk of vascular access infection associated with buttonhole cannulation of fistulas: data from the National Healthcare Safety Network. Am J Kidney Dis. 2020;76(1):82-89. doi:10.1053/j.ajkd.2019.11.006
16. MacRae JM, Ahmed SB, Atkar R, Hemmelgarn BR. A randomized trial comparing buttonhole with rope ladder needling in conventional hemodialysis patients. Clin J Am Soc Nephrol. 2012;7(10):1632-1638. doi:10.2215/CJN.02730312
17. Nesrallah GE, Cuerden M, Wong JHS, Pierratos A. Staphylococcus aureus bacteremia and buttonhole cannulation: long-term safety and efficacy of mupirocin prophylaxis. Clin J Am Soc Nephrol. 2010;5(6):1047-1053. doi:10.2215/CJN.00280110
18. Ho PL, Liu MCJ, Chow KH, et al. Emergence of ileS2 -carrying, multidrug-resistant plasmids in Staphylococcus lugdunensis. Antimicrob Agents Chemother. 2016;60(10):6411-6414. doi:10.1128/AAC.00948-16
19. Tan TY, Ng SY, He J. Microbiological characteristics, presumptive identification, and antibiotic susceptibilities of Staphylococcus lugdunensis. J Clin Microbiol. 2008;46(7):2393-2395. doi:10.1128/JCM.00740-08
20. Chang FY, Peacock JE, Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore). 2003;82(5):333-339. doi:10.1097/01.md.0000091184.93122.09
21. Shurland S, Zhan M, Bradham DD, Roghmann MC. Comparison of mortality risk associated with bacteremia due to methicillin-resistant and methicillin-susceptible Staphylococcus aureus. Infect Control Hosp Epidemiol. 2007;28(3):273-279. doi:10.1086/512627
22. Levine DP, Fromm BS, Reddy BR. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann Intern Med. 1991;115(9):674. doi:10.7326/0003-4819-115-9-674
23. Fowler VG, Karchmer AW, Tally FP, et al; S. aureus Endocarditis and Bacteremia Study Group. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355(7):653-665 . doi:10.1056/NEJMoa053783
24. Duhon B, Dallas S, Velasquez ST, Hand E. Staphylococcus lugdunensis bacteremia and endocarditis treated with cefazolin and rifampin. Am J Health Syst Pharm. 2015;72(13):1114-1118. doi:10.2146/ajhp140498
25. Lahey T, Shah R, Gittzus J, Schwartzman J, Kirkland K. Infectious diseases consultation lowers mortality from Staphylococcus aureus bacteremia. Medicine (Baltimore). 2009;88(5):263-267. doi:10.1097/MD.0b013e3181b8fccb
26. Forsblom E, Högnäs E, Syrjänen J, Järvinen A. Infectious diseases specialist consultation in Staphylococcus lugdunensis bacteremia. PLoS ONE. 2021;16(10):e0258511. doi:10.1371/journal.pone.0258511
Antimicrobial resistance requires a manifold response
BUENOS AIRES – Antimicrobial resistance (AMR) has become a global concern. And while one issue to be addressed is the deficit in research and development for new antibiotics, efforts to tackle this public health threat also should be directed toward promoting more rational prescription practices and strengthening the ability to identify the microorganisms responsible for infections, according to the World Health Organization. This was the conclusion reached at the fourth meeting of the WHO AMR Surveillance and Quality Assessment Collaborating Centres Network, which was held in Buenos Aires.
“We have to provide assistance to countries to ensure that the drugs are being used responsibly. We can come up with new antibiotics, but the issue at hand is not simply one of innovation: If nothing is done to correct inappropriate prescription practices and to overcome the lack of diagnostic laboratories at the country level, we’re going to miss out on those drugs as soon as they become available,” Kitty van Weezenbeek, MD, PhD, MPH, director of the AMR Surveillance, Prevention, and Control (AMR/SPC) Department at the WHO’s headquarters in Geneva, told this news organization.
Dr. van Weezenbeek pointed out that although there are currently no shortages of antimicrobials, the development and launch of new drugs that fight multidrug-resistant infections – infections for which there are few therapeutic options – has proceeded slowly. “It takes 10 to 15 years to develop a new antibiotic,” she said, adding that “the majority of pharmaceutical companies that had been engaged in the development of antimicrobials have filed for bankruptcy.”
In 2019, more people died – 1.2 million – from AMR than from malaria, tuberculosis, and HIV combined. Why are there so few market incentives when there is such a great need for those drugs? “One reason is that the pharmaceutical industry makes more money with long-term treatments, such as those for cancer and respiratory diseases. The other problem is that people everywhere are told not to use antibiotics,” said Dr. van Weezenbeek.
“A course of antibiotics lasts a few days, especially because we’re promoting rational use. Therefore, the trend is for the total amount of antimicrobials being used to be lower. So, it’s not as profitable,” added Carmem Lucia Pessoa-Silva, MD, PhD, head of the Surveillance, Evidence, and Laboratory Strengthening Unit of the WHO’s AMR/SPC Department.
On that note, Dr. van Weezenbeek mentioned that member countries are working with pharmaceutical companies and universities to address this problem. The WHO, for its part, has responded by implementing a global mechanism with a public health approach to create a “healthy” and equitable market for these medicines.
AMR is one of the top 10 global threats to human health. But it also has an impact on animal production, agricultural production, and the environment. Strategies to tackle AMR based on the One Health approach should involve all actors, social sectors, and citizens, according to Eva Jané Llopis, PhD, the representative of the Pan American Health Organization/WHO in Argentina.
At the root of the AMR problem is the widespread use of these drugs as growth promoters in animal production – for which several countries have enacted regulations – as well as “misunderstandings” between patients and physicians when there is not sufficient, timely access to laboratory diagnostics, especially in low- and middle-income countries.
“People think that if they’re given broad-spectrum antibiotics, they’re being prescribed the best antibiotics; and doctors, because there are no laboratory services, prescribe broad-spectrum antibiotics because they want to help patients. But that ends up causing more resistance to drugs, and thus, those antibiotics aren’t good for the patients,” said Dr. van Weezenbeek.
The WHO Global AMR and Use Surveillance System (GLASS) was launched in 2015. Its 2022 report, which marked the end of the system’s early implementation period, noted that the reported AMR rates are often lower in countries, territories, and areas with better testing coverage for most pathogen-drug-infection site combinations. However, as Dr. Pessoa-Silva acknowledged, monitoring “has not yet generated representative data,” because in many cases, countries either do not have surveillance systems or have only recently started implementing them.
Even so, the indicators that are available paint an increasingly worrisome picture. “For example, in many countries, resistance rates to first-line antibiotics were around 10%-20% with respect to Escherichia coli urinary tract infections and bloodstream bacteriologically confirmed infections. So, the risk of treatment failure is very high,” explained Dr. Pessoa-Silva.
The latest estimates indicate that every 2 or 3 minutes, somewhere in the world, a child dies from AMR. And the situation is particularly “dramatic” in neonatal intensive care units, where outbreaks of multidrug-resistant infections have a mortality rate of 50%, said Pilar Ramón-Pardo, MD, PhD, lead of the Special Program on AMR at the Pan American Health Organization, the WHO Regional Office for the Americas.
AMR rates also got worse during the pandemic because of the inappropriate prescription of massive amounts of antibiotics to hospitalized patients – something that was not in compliance with guidelines or protocols. Silvia Bertagnolio, MD, is an infectious disease specialist and the head of the Control and Response Strategies Unit in the WHO’s AMR Division. She spoke about the global clinical platform data pertaining to more than 1,500,000 patients who were hospitalized for COVID-19. Since 2020, 85% received antimicrobial treatment, despite the fact that only 5% had a concomitant infection at admission. “It’s easier to give antibiotics than to make a proper diagnosis,” said Dr. Bertagnolio.
This article was translated from Medscape’s Spanish edition and a version appeared on Medscape.com.
BUENOS AIRES – Antimicrobial resistance (AMR) has become a global concern. And while one issue to be addressed is the deficit in research and development for new antibiotics, efforts to tackle this public health threat also should be directed toward promoting more rational prescription practices and strengthening the ability to identify the microorganisms responsible for infections, according to the World Health Organization. This was the conclusion reached at the fourth meeting of the WHO AMR Surveillance and Quality Assessment Collaborating Centres Network, which was held in Buenos Aires.
“We have to provide assistance to countries to ensure that the drugs are being used responsibly. We can come up with new antibiotics, but the issue at hand is not simply one of innovation: If nothing is done to correct inappropriate prescription practices and to overcome the lack of diagnostic laboratories at the country level, we’re going to miss out on those drugs as soon as they become available,” Kitty van Weezenbeek, MD, PhD, MPH, director of the AMR Surveillance, Prevention, and Control (AMR/SPC) Department at the WHO’s headquarters in Geneva, told this news organization.
Dr. van Weezenbeek pointed out that although there are currently no shortages of antimicrobials, the development and launch of new drugs that fight multidrug-resistant infections – infections for which there are few therapeutic options – has proceeded slowly. “It takes 10 to 15 years to develop a new antibiotic,” she said, adding that “the majority of pharmaceutical companies that had been engaged in the development of antimicrobials have filed for bankruptcy.”
In 2019, more people died – 1.2 million – from AMR than from malaria, tuberculosis, and HIV combined. Why are there so few market incentives when there is such a great need for those drugs? “One reason is that the pharmaceutical industry makes more money with long-term treatments, such as those for cancer and respiratory diseases. The other problem is that people everywhere are told not to use antibiotics,” said Dr. van Weezenbeek.
“A course of antibiotics lasts a few days, especially because we’re promoting rational use. Therefore, the trend is for the total amount of antimicrobials being used to be lower. So, it’s not as profitable,” added Carmem Lucia Pessoa-Silva, MD, PhD, head of the Surveillance, Evidence, and Laboratory Strengthening Unit of the WHO’s AMR/SPC Department.
On that note, Dr. van Weezenbeek mentioned that member countries are working with pharmaceutical companies and universities to address this problem. The WHO, for its part, has responded by implementing a global mechanism with a public health approach to create a “healthy” and equitable market for these medicines.
AMR is one of the top 10 global threats to human health. But it also has an impact on animal production, agricultural production, and the environment. Strategies to tackle AMR based on the One Health approach should involve all actors, social sectors, and citizens, according to Eva Jané Llopis, PhD, the representative of the Pan American Health Organization/WHO in Argentina.
At the root of the AMR problem is the widespread use of these drugs as growth promoters in animal production – for which several countries have enacted regulations – as well as “misunderstandings” between patients and physicians when there is not sufficient, timely access to laboratory diagnostics, especially in low- and middle-income countries.
“People think that if they’re given broad-spectrum antibiotics, they’re being prescribed the best antibiotics; and doctors, because there are no laboratory services, prescribe broad-spectrum antibiotics because they want to help patients. But that ends up causing more resistance to drugs, and thus, those antibiotics aren’t good for the patients,” said Dr. van Weezenbeek.
The WHO Global AMR and Use Surveillance System (GLASS) was launched in 2015. Its 2022 report, which marked the end of the system’s early implementation period, noted that the reported AMR rates are often lower in countries, territories, and areas with better testing coverage for most pathogen-drug-infection site combinations. However, as Dr. Pessoa-Silva acknowledged, monitoring “has not yet generated representative data,” because in many cases, countries either do not have surveillance systems or have only recently started implementing them.
Even so, the indicators that are available paint an increasingly worrisome picture. “For example, in many countries, resistance rates to first-line antibiotics were around 10%-20% with respect to Escherichia coli urinary tract infections and bloodstream bacteriologically confirmed infections. So, the risk of treatment failure is very high,” explained Dr. Pessoa-Silva.
The latest estimates indicate that every 2 or 3 minutes, somewhere in the world, a child dies from AMR. And the situation is particularly “dramatic” in neonatal intensive care units, where outbreaks of multidrug-resistant infections have a mortality rate of 50%, said Pilar Ramón-Pardo, MD, PhD, lead of the Special Program on AMR at the Pan American Health Organization, the WHO Regional Office for the Americas.
AMR rates also got worse during the pandemic because of the inappropriate prescription of massive amounts of antibiotics to hospitalized patients – something that was not in compliance with guidelines or protocols. Silvia Bertagnolio, MD, is an infectious disease specialist and the head of the Control and Response Strategies Unit in the WHO’s AMR Division. She spoke about the global clinical platform data pertaining to more than 1,500,000 patients who were hospitalized for COVID-19. Since 2020, 85% received antimicrobial treatment, despite the fact that only 5% had a concomitant infection at admission. “It’s easier to give antibiotics than to make a proper diagnosis,” said Dr. Bertagnolio.
This article was translated from Medscape’s Spanish edition and a version appeared on Medscape.com.
BUENOS AIRES – Antimicrobial resistance (AMR) has become a global concern. And while one issue to be addressed is the deficit in research and development for new antibiotics, efforts to tackle this public health threat also should be directed toward promoting more rational prescription practices and strengthening the ability to identify the microorganisms responsible for infections, according to the World Health Organization. This was the conclusion reached at the fourth meeting of the WHO AMR Surveillance and Quality Assessment Collaborating Centres Network, which was held in Buenos Aires.
“We have to provide assistance to countries to ensure that the drugs are being used responsibly. We can come up with new antibiotics, but the issue at hand is not simply one of innovation: If nothing is done to correct inappropriate prescription practices and to overcome the lack of diagnostic laboratories at the country level, we’re going to miss out on those drugs as soon as they become available,” Kitty van Weezenbeek, MD, PhD, MPH, director of the AMR Surveillance, Prevention, and Control (AMR/SPC) Department at the WHO’s headquarters in Geneva, told this news organization.
Dr. van Weezenbeek pointed out that although there are currently no shortages of antimicrobials, the development and launch of new drugs that fight multidrug-resistant infections – infections for which there are few therapeutic options – has proceeded slowly. “It takes 10 to 15 years to develop a new antibiotic,” she said, adding that “the majority of pharmaceutical companies that had been engaged in the development of antimicrobials have filed for bankruptcy.”
In 2019, more people died – 1.2 million – from AMR than from malaria, tuberculosis, and HIV combined. Why are there so few market incentives when there is such a great need for those drugs? “One reason is that the pharmaceutical industry makes more money with long-term treatments, such as those for cancer and respiratory diseases. The other problem is that people everywhere are told not to use antibiotics,” said Dr. van Weezenbeek.
“A course of antibiotics lasts a few days, especially because we’re promoting rational use. Therefore, the trend is for the total amount of antimicrobials being used to be lower. So, it’s not as profitable,” added Carmem Lucia Pessoa-Silva, MD, PhD, head of the Surveillance, Evidence, and Laboratory Strengthening Unit of the WHO’s AMR/SPC Department.
On that note, Dr. van Weezenbeek mentioned that member countries are working with pharmaceutical companies and universities to address this problem. The WHO, for its part, has responded by implementing a global mechanism with a public health approach to create a “healthy” and equitable market for these medicines.
AMR is one of the top 10 global threats to human health. But it also has an impact on animal production, agricultural production, and the environment. Strategies to tackle AMR based on the One Health approach should involve all actors, social sectors, and citizens, according to Eva Jané Llopis, PhD, the representative of the Pan American Health Organization/WHO in Argentina.
At the root of the AMR problem is the widespread use of these drugs as growth promoters in animal production – for which several countries have enacted regulations – as well as “misunderstandings” between patients and physicians when there is not sufficient, timely access to laboratory diagnostics, especially in low- and middle-income countries.
“People think that if they’re given broad-spectrum antibiotics, they’re being prescribed the best antibiotics; and doctors, because there are no laboratory services, prescribe broad-spectrum antibiotics because they want to help patients. But that ends up causing more resistance to drugs, and thus, those antibiotics aren’t good for the patients,” said Dr. van Weezenbeek.
The WHO Global AMR and Use Surveillance System (GLASS) was launched in 2015. Its 2022 report, which marked the end of the system’s early implementation period, noted that the reported AMR rates are often lower in countries, territories, and areas with better testing coverage for most pathogen-drug-infection site combinations. However, as Dr. Pessoa-Silva acknowledged, monitoring “has not yet generated representative data,” because in many cases, countries either do not have surveillance systems or have only recently started implementing them.
Even so, the indicators that are available paint an increasingly worrisome picture. “For example, in many countries, resistance rates to first-line antibiotics were around 10%-20% with respect to Escherichia coli urinary tract infections and bloodstream bacteriologically confirmed infections. So, the risk of treatment failure is very high,” explained Dr. Pessoa-Silva.
The latest estimates indicate that every 2 or 3 minutes, somewhere in the world, a child dies from AMR. And the situation is particularly “dramatic” in neonatal intensive care units, where outbreaks of multidrug-resistant infections have a mortality rate of 50%, said Pilar Ramón-Pardo, MD, PhD, lead of the Special Program on AMR at the Pan American Health Organization, the WHO Regional Office for the Americas.
AMR rates also got worse during the pandemic because of the inappropriate prescription of massive amounts of antibiotics to hospitalized patients – something that was not in compliance with guidelines or protocols. Silvia Bertagnolio, MD, is an infectious disease specialist and the head of the Control and Response Strategies Unit in the WHO’s AMR Division. She spoke about the global clinical platform data pertaining to more than 1,500,000 patients who were hospitalized for COVID-19. Since 2020, 85% received antimicrobial treatment, despite the fact that only 5% had a concomitant infection at admission. “It’s easier to give antibiotics than to make a proper diagnosis,” said Dr. Bertagnolio.
This article was translated from Medscape’s Spanish edition and a version appeared on Medscape.com.
Lack of food for thought: Starve a bacterium, feed an infection
A whole new, tiny level of hangry
Ever been so hungry that everything just got on your nerves? Maybe you feel a little snappy right now? Like you’ll just lash out unless you get something to eat? Been there. And so have bacteria.
New research shows that some bacteria go into a full-on Hulk smash if they’re not getting the nutrients they need by releasing toxins into the body. Sounds like a bacterial temper tantrum.
Even though two cells may be genetically identical, they don’t always behave the same in a bacterial community. Some do their job and stay in line, but some evil twins rage out and make people sick by releasing toxins into the environment, Adam Rosenthal, PhD, of the University of North Carolina and his colleagues discovered.
To figure out why some cells were all business as usual while others were not, the investigators looked at Clostridium perfringens, a bacterium found in the intestines of humans and other vertebrates. When the C. perfringens cells were fed a little acetate to munch on, the hangry cells calmed down faster than a kid with a bag of fruit snacks, reducing toxin levels. Some cells even disappeared, falling in line with their model-citizen counterparts.
So what does this really mean? More research, duh. Now that we know nutrients play a role in toxicity, it may open the door to finding a way to fight against antibiotic resistance in humans and reduce antibiotic use in the food industry.
So think to yourself. Are you bothered for no reason? Getting a little testy with your friends and coworkers? Maybe you just haven’t eaten in a while. You’re literally not alone. Even a single-cell organism can behave based on its hunger levels.
Now go have a snack. Your bacteria are getting restless.
The very hangry iguana?
Imagine yourself on a warm, sunny tropical beach. You are enjoying a piece of cake as you take in the slow beat of the waves lapping against the shore. Life is as good as it could be.
Then you feel a presence nearby. Hostility. Hunger. A set of feral, covetous eyes in the nearby jungle. A reptilian beast stalks you, and its all-encompassing sweet tooth desires your cake.
Wait, hold on, what?
As an unfortunate 3-year-old on vacation in Costa Rica found out, there’s at least one iguana in the world out there with a taste for sugar (better than a taste for blood, we suppose).
While out on the beach, the lizard darted out of nowhere, bit the girl on the back of the hand, and stole her cake. Still not the worst party guest ever. The child was taken to a local clinic, where the wound was cleaned and a 5-day antibiotic treatment (lizards carry salmonella) was provided. Things seemed fine, and the girl returned home without incident.
But of course, that’s not the end of the story. Five months later, the girl’s parents noticed a red bump at the wound site. Over the next 3 months, the surrounding skin grew red and painful. A trip to the hospital in California revealed that she had a ganglion cyst and a discharge of pus. Turns out our cake-obsessed lizard friend did give the little girl a gift: the first known human case of Mycobacterium marinum infection following an iguana bite on record.
M. marinum, which causes a disease similar to tuberculosis, typically infects fish but can infect humans if skin wounds are exposed to contaminated water. It’s also resistant to most antibiotics, which is why the first round didn’t clear up the infection. A second round of more-potent antibiotics seems to be working well.
So, to sum up, this poor child got bitten by a lizard, had her cake stolen, and contracted a rare illness in exchange. For a 3-year-old, that’s gotta be in the top-10 worst days ever. Unless, of course, we’re actually living in the Marvel universe (sorry, multiverse at this point). Then we’re totally going to see the emergence of the new superhero Iguana Girl in 15 years or so. Keep your eyes open.
No allergies? Let them give up cake
Allergy season is already here – starting earlier every year, it seems – and many people are not happy about it. So unhappy, actually, that there’s a list of things they would be willing to give up for a year to get rid of their of allergies, according to a survey conducted by OnePoll on behalf of Flonase.
Nearly 40% of 2,000 respondents with allergies would go a year without eating cake or chocolate or playing video games in exchange for allergy-free status, the survey results show. Almost as many would forgo coffee (38%) or pizza (37%) for a year, while 36% would stay off social media and 31% would take a pay cut or give up their smartphones, the Independent reported.
More than half of the allergic Americans – 54%, to be exact – who were polled this past winter – Feb. 24 to March 1, to be exact – consider allergy symptoms to be the most frustrating part of the spring. Annoying things that were less frustrating to the group included mosquitoes (41%), filing tax returns (38%), and daylight savings time (37%).
The Trump arraignment circus, of course, occurred too late to make the list, as did the big “We’re going back to the office! No wait, we’re closing the office forever!” email extravaganza and emotional roller coaster. That second one, however, did not get nearly as much media coverage.
A whole new, tiny level of hangry
Ever been so hungry that everything just got on your nerves? Maybe you feel a little snappy right now? Like you’ll just lash out unless you get something to eat? Been there. And so have bacteria.
New research shows that some bacteria go into a full-on Hulk smash if they’re not getting the nutrients they need by releasing toxins into the body. Sounds like a bacterial temper tantrum.
Even though two cells may be genetically identical, they don’t always behave the same in a bacterial community. Some do their job and stay in line, but some evil twins rage out and make people sick by releasing toxins into the environment, Adam Rosenthal, PhD, of the University of North Carolina and his colleagues discovered.
To figure out why some cells were all business as usual while others were not, the investigators looked at Clostridium perfringens, a bacterium found in the intestines of humans and other vertebrates. When the C. perfringens cells were fed a little acetate to munch on, the hangry cells calmed down faster than a kid with a bag of fruit snacks, reducing toxin levels. Some cells even disappeared, falling in line with their model-citizen counterparts.
So what does this really mean? More research, duh. Now that we know nutrients play a role in toxicity, it may open the door to finding a way to fight against antibiotic resistance in humans and reduce antibiotic use in the food industry.
So think to yourself. Are you bothered for no reason? Getting a little testy with your friends and coworkers? Maybe you just haven’t eaten in a while. You’re literally not alone. Even a single-cell organism can behave based on its hunger levels.
Now go have a snack. Your bacteria are getting restless.
The very hangry iguana?
Imagine yourself on a warm, sunny tropical beach. You are enjoying a piece of cake as you take in the slow beat of the waves lapping against the shore. Life is as good as it could be.
Then you feel a presence nearby. Hostility. Hunger. A set of feral, covetous eyes in the nearby jungle. A reptilian beast stalks you, and its all-encompassing sweet tooth desires your cake.
Wait, hold on, what?
As an unfortunate 3-year-old on vacation in Costa Rica found out, there’s at least one iguana in the world out there with a taste for sugar (better than a taste for blood, we suppose).
While out on the beach, the lizard darted out of nowhere, bit the girl on the back of the hand, and stole her cake. Still not the worst party guest ever. The child was taken to a local clinic, where the wound was cleaned and a 5-day antibiotic treatment (lizards carry salmonella) was provided. Things seemed fine, and the girl returned home without incident.
But of course, that’s not the end of the story. Five months later, the girl’s parents noticed a red bump at the wound site. Over the next 3 months, the surrounding skin grew red and painful. A trip to the hospital in California revealed that she had a ganglion cyst and a discharge of pus. Turns out our cake-obsessed lizard friend did give the little girl a gift: the first known human case of Mycobacterium marinum infection following an iguana bite on record.
M. marinum, which causes a disease similar to tuberculosis, typically infects fish but can infect humans if skin wounds are exposed to contaminated water. It’s also resistant to most antibiotics, which is why the first round didn’t clear up the infection. A second round of more-potent antibiotics seems to be working well.
So, to sum up, this poor child got bitten by a lizard, had her cake stolen, and contracted a rare illness in exchange. For a 3-year-old, that’s gotta be in the top-10 worst days ever. Unless, of course, we’re actually living in the Marvel universe (sorry, multiverse at this point). Then we’re totally going to see the emergence of the new superhero Iguana Girl in 15 years or so. Keep your eyes open.
No allergies? Let them give up cake
Allergy season is already here – starting earlier every year, it seems – and many people are not happy about it. So unhappy, actually, that there’s a list of things they would be willing to give up for a year to get rid of their of allergies, according to a survey conducted by OnePoll on behalf of Flonase.
Nearly 40% of 2,000 respondents with allergies would go a year without eating cake or chocolate or playing video games in exchange for allergy-free status, the survey results show. Almost as many would forgo coffee (38%) or pizza (37%) for a year, while 36% would stay off social media and 31% would take a pay cut or give up their smartphones, the Independent reported.
More than half of the allergic Americans – 54%, to be exact – who were polled this past winter – Feb. 24 to March 1, to be exact – consider allergy symptoms to be the most frustrating part of the spring. Annoying things that were less frustrating to the group included mosquitoes (41%), filing tax returns (38%), and daylight savings time (37%).
The Trump arraignment circus, of course, occurred too late to make the list, as did the big “We’re going back to the office! No wait, we’re closing the office forever!” email extravaganza and emotional roller coaster. That second one, however, did not get nearly as much media coverage.
A whole new, tiny level of hangry
Ever been so hungry that everything just got on your nerves? Maybe you feel a little snappy right now? Like you’ll just lash out unless you get something to eat? Been there. And so have bacteria.
New research shows that some bacteria go into a full-on Hulk smash if they’re not getting the nutrients they need by releasing toxins into the body. Sounds like a bacterial temper tantrum.
Even though two cells may be genetically identical, they don’t always behave the same in a bacterial community. Some do their job and stay in line, but some evil twins rage out and make people sick by releasing toxins into the environment, Adam Rosenthal, PhD, of the University of North Carolina and his colleagues discovered.
To figure out why some cells were all business as usual while others were not, the investigators looked at Clostridium perfringens, a bacterium found in the intestines of humans and other vertebrates. When the C. perfringens cells were fed a little acetate to munch on, the hangry cells calmed down faster than a kid with a bag of fruit snacks, reducing toxin levels. Some cells even disappeared, falling in line with their model-citizen counterparts.
So what does this really mean? More research, duh. Now that we know nutrients play a role in toxicity, it may open the door to finding a way to fight against antibiotic resistance in humans and reduce antibiotic use in the food industry.
So think to yourself. Are you bothered for no reason? Getting a little testy with your friends and coworkers? Maybe you just haven’t eaten in a while. You’re literally not alone. Even a single-cell organism can behave based on its hunger levels.
Now go have a snack. Your bacteria are getting restless.
The very hangry iguana?
Imagine yourself on a warm, sunny tropical beach. You are enjoying a piece of cake as you take in the slow beat of the waves lapping against the shore. Life is as good as it could be.
Then you feel a presence nearby. Hostility. Hunger. A set of feral, covetous eyes in the nearby jungle. A reptilian beast stalks you, and its all-encompassing sweet tooth desires your cake.
Wait, hold on, what?
As an unfortunate 3-year-old on vacation in Costa Rica found out, there’s at least one iguana in the world out there with a taste for sugar (better than a taste for blood, we suppose).
While out on the beach, the lizard darted out of nowhere, bit the girl on the back of the hand, and stole her cake. Still not the worst party guest ever. The child was taken to a local clinic, where the wound was cleaned and a 5-day antibiotic treatment (lizards carry salmonella) was provided. Things seemed fine, and the girl returned home without incident.
But of course, that’s not the end of the story. Five months later, the girl’s parents noticed a red bump at the wound site. Over the next 3 months, the surrounding skin grew red and painful. A trip to the hospital in California revealed that she had a ganglion cyst and a discharge of pus. Turns out our cake-obsessed lizard friend did give the little girl a gift: the first known human case of Mycobacterium marinum infection following an iguana bite on record.
M. marinum, which causes a disease similar to tuberculosis, typically infects fish but can infect humans if skin wounds are exposed to contaminated water. It’s also resistant to most antibiotics, which is why the first round didn’t clear up the infection. A second round of more-potent antibiotics seems to be working well.
So, to sum up, this poor child got bitten by a lizard, had her cake stolen, and contracted a rare illness in exchange. For a 3-year-old, that’s gotta be in the top-10 worst days ever. Unless, of course, we’re actually living in the Marvel universe (sorry, multiverse at this point). Then we’re totally going to see the emergence of the new superhero Iguana Girl in 15 years or so. Keep your eyes open.
No allergies? Let them give up cake
Allergy season is already here – starting earlier every year, it seems – and many people are not happy about it. So unhappy, actually, that there’s a list of things they would be willing to give up for a year to get rid of their of allergies, according to a survey conducted by OnePoll on behalf of Flonase.
Nearly 40% of 2,000 respondents with allergies would go a year without eating cake or chocolate or playing video games in exchange for allergy-free status, the survey results show. Almost as many would forgo coffee (38%) or pizza (37%) for a year, while 36% would stay off social media and 31% would take a pay cut or give up their smartphones, the Independent reported.
More than half of the allergic Americans – 54%, to be exact – who were polled this past winter – Feb. 24 to March 1, to be exact – consider allergy symptoms to be the most frustrating part of the spring. Annoying things that were less frustrating to the group included mosquitoes (41%), filing tax returns (38%), and daylight savings time (37%).
The Trump arraignment circus, of course, occurred too late to make the list, as did the big “We’re going back to the office! No wait, we’re closing the office forever!” email extravaganza and emotional roller coaster. That second one, however, did not get nearly as much media coverage.