User login
Why dermatologists should support artificial intelligence efforts
“AI is meant to be an enhancement strategy, a support tool to improve our diagnostic abilities,” Dr. Patel, a Mohs surgeon who is director of cutaneous oncology at the George Washington University Cancer Center, Washington, said during the ODAC Dermatology, Aesthetic & Surgical Conference. “Dermatologists should embrace AI and drive how it is utilized – be the captain of the plane (technology) and the passenger (patient). If we’re not in the forefront of the plane, we’re not to be able to dictate which way we are going with this.”
In 2019, a group of German researchers found that AI can improve accuracy and efficiency of specialists in classifying skin cancer based on dermoscopic images. “I really do believe this is going to be the future,” said Dr. Patel, who was not involved with the study. “Current research involves using supervised learning on known outcomes to determine inputs to predict them. In dermatology, think of identifying melanoma from clinical or dermoscopic images or predicting metastasis risk from digitized pathology slides.”
However, there are currently no universal guidelines on how large an AI dataset needs to be to yield accurate results. In the dermatology literature, most AI datasets range between 600 and 14,000 examples, Dr. Patel said, with a large study-specific variation in performance. “Misleading results can result from unanticipated training errors,” he said.
“The AI network may learn its intended task or an unrelated situational cue. For example, you can use great images to predict melanoma, but you may have an unintended poor outcome related to images that have, say, a ruler inside of them clustered within the melanoma diagnoses.” And unbeknown to the system’s developer, “the algorithm picks up that the ruler is predictive of an image being a melanoma and not the pigmented lesion itself.” In other words, the algorithm is only as good as the dataset being used, he said. “This is the key element, to ask what the dataset is that’s training the tool that you may one day use.”
Convolutional neural network
In 2017, a seminal study published in Nature showed that for classification of melanoma and epidermal lesions, a type of AI used in image processing known as a convolutional neural network (CNN) was on par with dermatologists and outperformed the average. For epidermal lesions, the network was one standard deviation higher above the average for dermatologists, while for melanocytic lesions, the network was just below one standard deviation above the average of the dermatologists. A CNN “clearly can perform well because it works on a different level than how our brains work,” Dr. Patel said.
In a separate study, a CNN trained to recognize melanoma in dermoscopic images was compared to 58 international dermatologists with varying levels of dermoscopy experience; 29% were “beginners,” with less than 2 years of experience; 19% were “skilled,” with 2-5 years of experience; and 52% were “experts,” with at least 5 years of experience. The analysis consisted of two experiments: In level I, dermatologists classified lesions based on dermoscopy only. In level II, dermatologists were provided dermoscopy, clinical images, and additional clinical information, while the CNN was trained on images only. The researchers found that most dermatologists were outperformed by the CNN. “Physicians of all different levels of training and experience may benefit from assistance by a CNN’s image classification,” they concluded.
Gene expression profiling
Another aspect of AI is gene expression profiling (GEP), which Dr. Patel defined as the evaluation of frequency and intensity of genetic activity at once to create a global picture of cellular function. “It’s AI that uses machine learning to evaluate genetic expression to assess lesion behavior,” he explained.
One GEP test on the market is the Pigmented Lesion Assay (PLA) from DermTech, a noninvasive test that looks at the expression of two genes to predict if a lesion is malignant or not. “Based on their validation set, they have shown some impressive numbers,” with sensitivities above 90%, and published registry data that have shown higher sensitivities “and even specificities above 90%,” he said.
“On the surface, it looks like this would be a useful test,” Dr. Patel said. A study published in 2021 looked at the evidence of applying real-world evidence with this test to see if results held up. Based on the authors’ analysis, he noted, “you would need a sensitivity and specificity of 95% to yield a positivity rate of 9.5% for the PLA test, which is what has been reported in real-world use. So, there’s a disconnect somewhere and we are not quite there yet.” That may be a result of the dataset itself not being as uniform between the validation and the training datasets, he continued. Also, the expression of certain genes is different “if you don’t have a clean input variable” of what the test is being used for, he added.
“If you’re not mirroring the dataset, you’re not going to get clean data,” he said. “So, if you’re using this on younger patients or for sun-damaged lesional skin or nonmelanocytic lesions around sun-damaged areas, there are variable expressions that may not be accurately captured by that algorithm. This might help explain the real-world variation that we’re seeing.”
Another GEP test in use is the 31-Gene Expression Profile Test for Melanoma, which evaluates gene expressions in melanoma tumors and what the behavior of that tumor may be. The test has been available for more than a decade “and there is a lot of speculation about its use,” Dr. Patel said. “A recent paper attempted to come up with an algorithm of how to use this, but there’s a lot of concern about the endpoints of what changes in management might result from this test. That is what we need to be thinking about. There’s a lot of back and forth about this.”
In 2020, authors of a consensus statement on prognostic GEP in cutaneous melanoma concluded that before GEP testing is routinely used, the clinical benefit in the management of patients with melanoma should be established through further clinical investigation. Dr. Patel recommended the accompanying editorial on GEP in melanoma, written by Hensin Tsao, MD, PhD, and Warren H. Chan, MS, in JAMA Dermatology.
In Dr. Patel’s opinion, T1a melanomas (0.8 mm, nonulcerated) do not need routine GEP, but the GEP test may be useful in cases that are in the “gray zone,” such as those with T1b or some borderline T2a melanomas (> 0.8 mm, < 1.2mm, nonulcerated, but with high mitosis, etc.); patients with unique coexisting conditions such as pregnancy, and patients who may not tolerate sentinel lymph node biopsy (SLNB) or adjuvant therapy.
Echoing sentiments expressed in the JAMA Dermatology editorial, he advised dermatologists to “remember your training and know the data. GEP predicting survival is not the same as SLNB positive rate. GEP should not replace standard guidelines in T2a and higher melanomas. Nodal sampling remains part of all major guidelines and determines adjuvant therapy.”
He cited the characterization of GEP in the editorial as “a powerful technology” that heralds the age of personalized medicine, but it is not ready for ubiquitous use. Prospective studies and time will lead to highly accurate tools.”
Dr. Patel disclosed that he is chief medical officer for Lazarus AI.
“AI is meant to be an enhancement strategy, a support tool to improve our diagnostic abilities,” Dr. Patel, a Mohs surgeon who is director of cutaneous oncology at the George Washington University Cancer Center, Washington, said during the ODAC Dermatology, Aesthetic & Surgical Conference. “Dermatologists should embrace AI and drive how it is utilized – be the captain of the plane (technology) and the passenger (patient). If we’re not in the forefront of the plane, we’re not to be able to dictate which way we are going with this.”
In 2019, a group of German researchers found that AI can improve accuracy and efficiency of specialists in classifying skin cancer based on dermoscopic images. “I really do believe this is going to be the future,” said Dr. Patel, who was not involved with the study. “Current research involves using supervised learning on known outcomes to determine inputs to predict them. In dermatology, think of identifying melanoma from clinical or dermoscopic images or predicting metastasis risk from digitized pathology slides.”
However, there are currently no universal guidelines on how large an AI dataset needs to be to yield accurate results. In the dermatology literature, most AI datasets range between 600 and 14,000 examples, Dr. Patel said, with a large study-specific variation in performance. “Misleading results can result from unanticipated training errors,” he said.
“The AI network may learn its intended task or an unrelated situational cue. For example, you can use great images to predict melanoma, but you may have an unintended poor outcome related to images that have, say, a ruler inside of them clustered within the melanoma diagnoses.” And unbeknown to the system’s developer, “the algorithm picks up that the ruler is predictive of an image being a melanoma and not the pigmented lesion itself.” In other words, the algorithm is only as good as the dataset being used, he said. “This is the key element, to ask what the dataset is that’s training the tool that you may one day use.”
Convolutional neural network
In 2017, a seminal study published in Nature showed that for classification of melanoma and epidermal lesions, a type of AI used in image processing known as a convolutional neural network (CNN) was on par with dermatologists and outperformed the average. For epidermal lesions, the network was one standard deviation higher above the average for dermatologists, while for melanocytic lesions, the network was just below one standard deviation above the average of the dermatologists. A CNN “clearly can perform well because it works on a different level than how our brains work,” Dr. Patel said.
In a separate study, a CNN trained to recognize melanoma in dermoscopic images was compared to 58 international dermatologists with varying levels of dermoscopy experience; 29% were “beginners,” with less than 2 years of experience; 19% were “skilled,” with 2-5 years of experience; and 52% were “experts,” with at least 5 years of experience. The analysis consisted of two experiments: In level I, dermatologists classified lesions based on dermoscopy only. In level II, dermatologists were provided dermoscopy, clinical images, and additional clinical information, while the CNN was trained on images only. The researchers found that most dermatologists were outperformed by the CNN. “Physicians of all different levels of training and experience may benefit from assistance by a CNN’s image classification,” they concluded.
Gene expression profiling
Another aspect of AI is gene expression profiling (GEP), which Dr. Patel defined as the evaluation of frequency and intensity of genetic activity at once to create a global picture of cellular function. “It’s AI that uses machine learning to evaluate genetic expression to assess lesion behavior,” he explained.
One GEP test on the market is the Pigmented Lesion Assay (PLA) from DermTech, a noninvasive test that looks at the expression of two genes to predict if a lesion is malignant or not. “Based on their validation set, they have shown some impressive numbers,” with sensitivities above 90%, and published registry data that have shown higher sensitivities “and even specificities above 90%,” he said.
“On the surface, it looks like this would be a useful test,” Dr. Patel said. A study published in 2021 looked at the evidence of applying real-world evidence with this test to see if results held up. Based on the authors’ analysis, he noted, “you would need a sensitivity and specificity of 95% to yield a positivity rate of 9.5% for the PLA test, which is what has been reported in real-world use. So, there’s a disconnect somewhere and we are not quite there yet.” That may be a result of the dataset itself not being as uniform between the validation and the training datasets, he continued. Also, the expression of certain genes is different “if you don’t have a clean input variable” of what the test is being used for, he added.
“If you’re not mirroring the dataset, you’re not going to get clean data,” he said. “So, if you’re using this on younger patients or for sun-damaged lesional skin or nonmelanocytic lesions around sun-damaged areas, there are variable expressions that may not be accurately captured by that algorithm. This might help explain the real-world variation that we’re seeing.”
Another GEP test in use is the 31-Gene Expression Profile Test for Melanoma, which evaluates gene expressions in melanoma tumors and what the behavior of that tumor may be. The test has been available for more than a decade “and there is a lot of speculation about its use,” Dr. Patel said. “A recent paper attempted to come up with an algorithm of how to use this, but there’s a lot of concern about the endpoints of what changes in management might result from this test. That is what we need to be thinking about. There’s a lot of back and forth about this.”
In 2020, authors of a consensus statement on prognostic GEP in cutaneous melanoma concluded that before GEP testing is routinely used, the clinical benefit in the management of patients with melanoma should be established through further clinical investigation. Dr. Patel recommended the accompanying editorial on GEP in melanoma, written by Hensin Tsao, MD, PhD, and Warren H. Chan, MS, in JAMA Dermatology.
In Dr. Patel’s opinion, T1a melanomas (0.8 mm, nonulcerated) do not need routine GEP, but the GEP test may be useful in cases that are in the “gray zone,” such as those with T1b or some borderline T2a melanomas (> 0.8 mm, < 1.2mm, nonulcerated, but with high mitosis, etc.); patients with unique coexisting conditions such as pregnancy, and patients who may not tolerate sentinel lymph node biopsy (SLNB) or adjuvant therapy.
Echoing sentiments expressed in the JAMA Dermatology editorial, he advised dermatologists to “remember your training and know the data. GEP predicting survival is not the same as SLNB positive rate. GEP should not replace standard guidelines in T2a and higher melanomas. Nodal sampling remains part of all major guidelines and determines adjuvant therapy.”
He cited the characterization of GEP in the editorial as “a powerful technology” that heralds the age of personalized medicine, but it is not ready for ubiquitous use. Prospective studies and time will lead to highly accurate tools.”
Dr. Patel disclosed that he is chief medical officer for Lazarus AI.
“AI is meant to be an enhancement strategy, a support tool to improve our diagnostic abilities,” Dr. Patel, a Mohs surgeon who is director of cutaneous oncology at the George Washington University Cancer Center, Washington, said during the ODAC Dermatology, Aesthetic & Surgical Conference. “Dermatologists should embrace AI and drive how it is utilized – be the captain of the plane (technology) and the passenger (patient). If we’re not in the forefront of the plane, we’re not to be able to dictate which way we are going with this.”
In 2019, a group of German researchers found that AI can improve accuracy and efficiency of specialists in classifying skin cancer based on dermoscopic images. “I really do believe this is going to be the future,” said Dr. Patel, who was not involved with the study. “Current research involves using supervised learning on known outcomes to determine inputs to predict them. In dermatology, think of identifying melanoma from clinical or dermoscopic images or predicting metastasis risk from digitized pathology slides.”
However, there are currently no universal guidelines on how large an AI dataset needs to be to yield accurate results. In the dermatology literature, most AI datasets range between 600 and 14,000 examples, Dr. Patel said, with a large study-specific variation in performance. “Misleading results can result from unanticipated training errors,” he said.
“The AI network may learn its intended task or an unrelated situational cue. For example, you can use great images to predict melanoma, but you may have an unintended poor outcome related to images that have, say, a ruler inside of them clustered within the melanoma diagnoses.” And unbeknown to the system’s developer, “the algorithm picks up that the ruler is predictive of an image being a melanoma and not the pigmented lesion itself.” In other words, the algorithm is only as good as the dataset being used, he said. “This is the key element, to ask what the dataset is that’s training the tool that you may one day use.”
Convolutional neural network
In 2017, a seminal study published in Nature showed that for classification of melanoma and epidermal lesions, a type of AI used in image processing known as a convolutional neural network (CNN) was on par with dermatologists and outperformed the average. For epidermal lesions, the network was one standard deviation higher above the average for dermatologists, while for melanocytic lesions, the network was just below one standard deviation above the average of the dermatologists. A CNN “clearly can perform well because it works on a different level than how our brains work,” Dr. Patel said.
In a separate study, a CNN trained to recognize melanoma in dermoscopic images was compared to 58 international dermatologists with varying levels of dermoscopy experience; 29% were “beginners,” with less than 2 years of experience; 19% were “skilled,” with 2-5 years of experience; and 52% were “experts,” with at least 5 years of experience. The analysis consisted of two experiments: In level I, dermatologists classified lesions based on dermoscopy only. In level II, dermatologists were provided dermoscopy, clinical images, and additional clinical information, while the CNN was trained on images only. The researchers found that most dermatologists were outperformed by the CNN. “Physicians of all different levels of training and experience may benefit from assistance by a CNN’s image classification,” they concluded.
Gene expression profiling
Another aspect of AI is gene expression profiling (GEP), which Dr. Patel defined as the evaluation of frequency and intensity of genetic activity at once to create a global picture of cellular function. “It’s AI that uses machine learning to evaluate genetic expression to assess lesion behavior,” he explained.
One GEP test on the market is the Pigmented Lesion Assay (PLA) from DermTech, a noninvasive test that looks at the expression of two genes to predict if a lesion is malignant or not. “Based on their validation set, they have shown some impressive numbers,” with sensitivities above 90%, and published registry data that have shown higher sensitivities “and even specificities above 90%,” he said.
“On the surface, it looks like this would be a useful test,” Dr. Patel said. A study published in 2021 looked at the evidence of applying real-world evidence with this test to see if results held up. Based on the authors’ analysis, he noted, “you would need a sensitivity and specificity of 95% to yield a positivity rate of 9.5% for the PLA test, which is what has been reported in real-world use. So, there’s a disconnect somewhere and we are not quite there yet.” That may be a result of the dataset itself not being as uniform between the validation and the training datasets, he continued. Also, the expression of certain genes is different “if you don’t have a clean input variable” of what the test is being used for, he added.
“If you’re not mirroring the dataset, you’re not going to get clean data,” he said. “So, if you’re using this on younger patients or for sun-damaged lesional skin or nonmelanocytic lesions around sun-damaged areas, there are variable expressions that may not be accurately captured by that algorithm. This might help explain the real-world variation that we’re seeing.”
Another GEP test in use is the 31-Gene Expression Profile Test for Melanoma, which evaluates gene expressions in melanoma tumors and what the behavior of that tumor may be. The test has been available for more than a decade “and there is a lot of speculation about its use,” Dr. Patel said. “A recent paper attempted to come up with an algorithm of how to use this, but there’s a lot of concern about the endpoints of what changes in management might result from this test. That is what we need to be thinking about. There’s a lot of back and forth about this.”
In 2020, authors of a consensus statement on prognostic GEP in cutaneous melanoma concluded that before GEP testing is routinely used, the clinical benefit in the management of patients with melanoma should be established through further clinical investigation. Dr. Patel recommended the accompanying editorial on GEP in melanoma, written by Hensin Tsao, MD, PhD, and Warren H. Chan, MS, in JAMA Dermatology.
In Dr. Patel’s opinion, T1a melanomas (0.8 mm, nonulcerated) do not need routine GEP, but the GEP test may be useful in cases that are in the “gray zone,” such as those with T1b or some borderline T2a melanomas (> 0.8 mm, < 1.2mm, nonulcerated, but with high mitosis, etc.); patients with unique coexisting conditions such as pregnancy, and patients who may not tolerate sentinel lymph node biopsy (SLNB) or adjuvant therapy.
Echoing sentiments expressed in the JAMA Dermatology editorial, he advised dermatologists to “remember your training and know the data. GEP predicting survival is not the same as SLNB positive rate. GEP should not replace standard guidelines in T2a and higher melanomas. Nodal sampling remains part of all major guidelines and determines adjuvant therapy.”
He cited the characterization of GEP in the editorial as “a powerful technology” that heralds the age of personalized medicine, but it is not ready for ubiquitous use. Prospective studies and time will lead to highly accurate tools.”
Dr. Patel disclosed that he is chief medical officer for Lazarus AI.
FROM ODAC 2022
Pencil-core Granuloma Forming 62 Years After Initial Injury
To the Editor:
Trauma from a pencil tip can sometimes result in a fragment of lead being left embedded within the skin. Pencil lead is composed of 66% graphite carbon, 26% aluminum silicate, and 8% paraffin.1,2 While the toxicity of these individual elements is low, paraffin can cause nonallergic foreign-body reactions, aluminum silicate can induce epithelioid granulomatous reactions, and graphite has been reported to cause chronic granulomatous reactions in the lungs of those who work with graphite.2 Penetrating trauma with a pencil can result in the formation of a cutaneous granulomatous reaction that can sometimes occur years to decades after the initial injury.3,4 Several cases of pencil-core granulomas have been published, with lag times between the initial trauma and lesion growth as long as 58 years.1-10 The pencil-core granuloma may simulate malignant melanoma, as it presents clinically as a growing, darkly pigmented lesion, thus prompting biopsy. We present a case of a pencil-core granuloma that began to grow 62 years after the initial trauma.
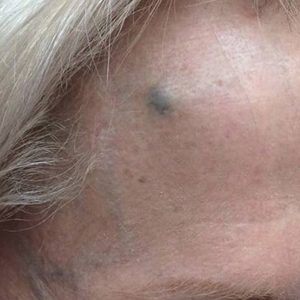
A 72-year-old woman was referred to our clinic for evaluation of a dark nodule on the forehead. The lesion had been present since the age of 10 years, reportedly from an accidental stabbing with a pencil. The lesion had been flat, stable, and asymptomatic since the trauma occurred; however, the patient reported that approximately 9 months prior to presentation, it had started growing and became painful. Physical examination revealed a 1.0-cm, round, bluish-black nodule on the right superior forehead (Figure 1A). No satellite lesions or local lymphadenopathy were noted on general examination.
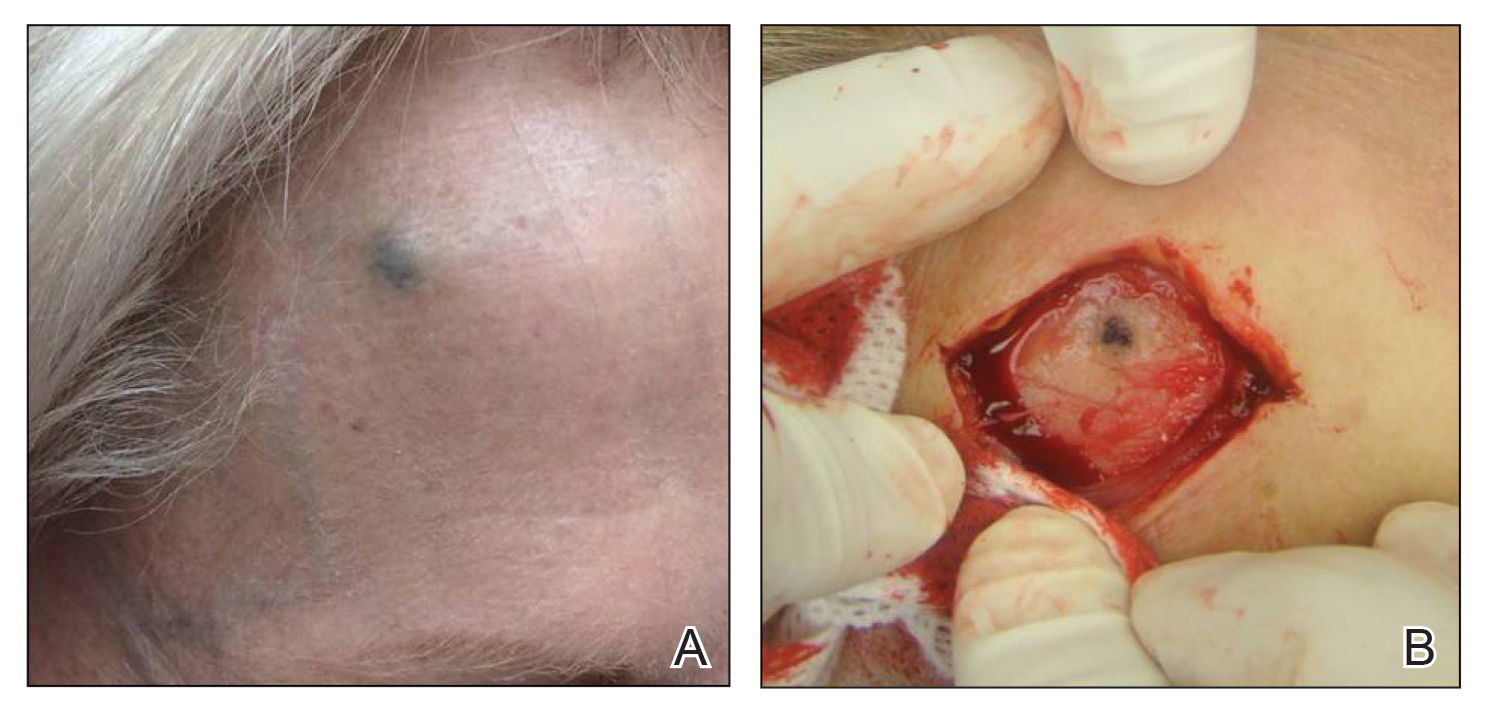
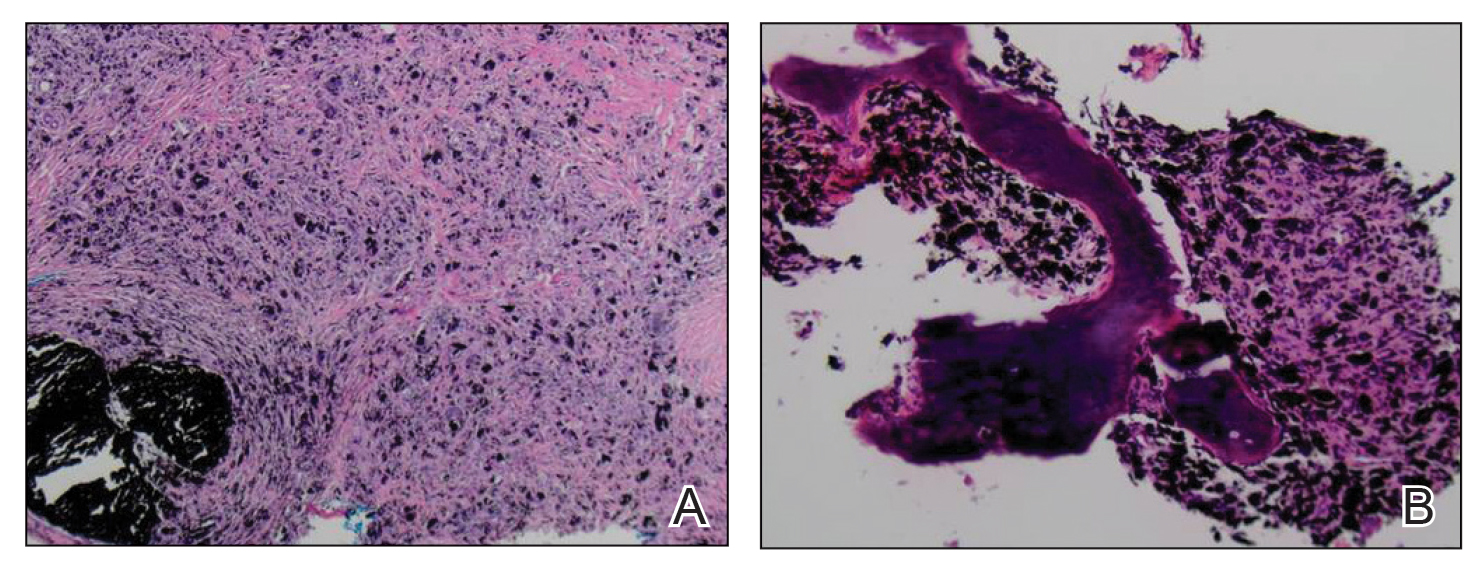
An elliptical excision of the lesion with 1-cm margins revealed a bluish-black mass extending through the dermis, through the frontalis muscle, and into the periosteum and frontal bone (Figure 1B). A No. 15 blade was then used to remove the remaining pigment from the outer table of the frontal bone. Histopathologic findings demonstrated a sarcoidal granulomatous dermatitis associated with abundant, nonpolarizable, black, granular pigment consistent with carbon tattoo. This foreign material was readily identifiable in large extracellular deposits and also within histiocytes, including numerous multinucleated giant cells (Figure 2). Immunostaining for MART-1 and SOX-10 antigens failed to demonstrate a melanocytic proliferation. These findings were consistent with a sarcoidal foreign-body granulomatous reaction to carbon tattoo following traumatic graphite implantation.
Granulomatous reactions to carbon tattoo may be sarcoidal (foreign-body granulomatous dermatitis), palisading, or rarely tuberculoid (caseating). Sarcoidal granulomatous tattoo reactions may occur in patients with sarcoidosis due to koebnerization, and histology alone is not discriminatory; however, in our patient, the absence of underlying sarcoidosis or clinical or histologic findings of sarcoidosis outside of the site of the pencil-core granuloma excluded that possibility.11 Pencil-core granulomas are characterized by a delayed foreign-body reaction to retained fragments of lead often years following a penetrating trauma with a pencil. Previous reports have described various lag times from injury to lesion growth of up to 58 years.1-10 Our patient claimed to have noticed the lesion growing and becoming painful only after a 62-year lag time following the initial trauma. To our knowledge, this is the longest lag time between the initial pencil injury and induction of the foreign-body reaction reported in the literature. Clinically, the lesion appeared and behaved very similar to a melanoma, prompting further treatment and evaluation.
It has been suggested that the lag period between the initial trauma and the rapid growth of the lesion may correspond to the amount of time required for the breakdown of the pencil lead to a critical size followed by the dispersal of those particles within the interstitium, where they can induce a granulomatous reaction.1,2,9 One case described a patient who reported that the growth and clinical change of the pencil-core granuloma only started when the patient accidentally hit the area where the trauma had occurred 31 years prior.1 This additional trauma may have caused further mechanical breakdown of the lead to set off the tissue reaction. In our case, the patient did not recall any additional trauma to the head prior to the onset of growth of the nodule on the forehead.
Our case indicates that carbon tattoo may be a possible sequela of a penetrating injury from a pencil with retained pencil lead fragments; however, many of these carbon tattoos may remain stable throughout the remainder of the patient’s life.
- Gormley RH, Kovach SJ III, Zhang PJ. Role for trauma in inducing pencil “lead” granuloma in the skin. J Am Acad Dermatol. 2010;62:1074-1075.
- Terasawa N, Kishimoto S, Kibe Y, et al. Graphite foreign body granuloma. Br J Dermatol. 1999;141:774-776.
- Fukunaga Y, Hashimoto I, Nakanishi H, et al. Pencil-core granuloma of the face: report of two rare cases. J Plast Reconstr Aesthet Surg. 2011;64:1235-1237.
- Aswani VH, Kim SL. Fifty-three years after a pencil puncture wound. Case Rep Dermatol. 2015;7:303-305.
- Taylor B, Frumkin A, Pitha JV. Delayed reaction to “lead” pencil simulating melanoma. Cutis. 1988;42:199-201.
- Granick MS, Erickson ER, Solomon MP. Pencil-core granuloma. Plast Reconstr Surg. 1992;89:136-138.
- Andreano J. Stump the experts. foreign body granuloma. J Dermatol Surg Oncol. 1992;18:277, 343.
- Yoshitatsu S, Takagi T. A case of giant pencil-core granuloma. J Dermatol. 2000;27:329-332.
- Hatano Y, Asada Y, Komada S, et al. A case of pencil core granuloma with an unusual temporal profile. Dermatology. 2000;201:151-153.
- Seitz IA, Silva BA, Schechter LS. Unusual sequela from a pencil stab wound reveals a retained graphite foreign body. Pediatr Emerg Care. 2014;30:568-570.
- Motaparthi K. Tattoo ink. In: Cockerell CJ, Hall BJ, eds. Nonneoplastic Dermatopathology. 2nd ed. Amirsys; 2016: 270.
To the Editor:
Trauma from a pencil tip can sometimes result in a fragment of lead being left embedded within the skin. Pencil lead is composed of 66% graphite carbon, 26% aluminum silicate, and 8% paraffin.1,2 While the toxicity of these individual elements is low, paraffin can cause nonallergic foreign-body reactions, aluminum silicate can induce epithelioid granulomatous reactions, and graphite has been reported to cause chronic granulomatous reactions in the lungs of those who work with graphite.2 Penetrating trauma with a pencil can result in the formation of a cutaneous granulomatous reaction that can sometimes occur years to decades after the initial injury.3,4 Several cases of pencil-core granulomas have been published, with lag times between the initial trauma and lesion growth as long as 58 years.1-10 The pencil-core granuloma may simulate malignant melanoma, as it presents clinically as a growing, darkly pigmented lesion, thus prompting biopsy. We present a case of a pencil-core granuloma that began to grow 62 years after the initial trauma.
A 72-year-old woman was referred to our clinic for evaluation of a dark nodule on the forehead. The lesion had been present since the age of 10 years, reportedly from an accidental stabbing with a pencil. The lesion had been flat, stable, and asymptomatic since the trauma occurred; however, the patient reported that approximately 9 months prior to presentation, it had started growing and became painful. Physical examination revealed a 1.0-cm, round, bluish-black nodule on the right superior forehead (Figure 1A). No satellite lesions or local lymphadenopathy were noted on general examination.

An elliptical excision of the lesion with 1-cm margins revealed a bluish-black mass extending through the dermis, through the frontalis muscle, and into the periosteum and frontal bone (Figure 1B). A No. 15 blade was then used to remove the remaining pigment from the outer table of the frontal bone. Histopathologic findings demonstrated a sarcoidal granulomatous dermatitis associated with abundant, nonpolarizable, black, granular pigment consistent with carbon tattoo. This foreign material was readily identifiable in large extracellular deposits and also within histiocytes, including numerous multinucleated giant cells (Figure 2). Immunostaining for MART-1 and SOX-10 antigens failed to demonstrate a melanocytic proliferation. These findings were consistent with a sarcoidal foreign-body granulomatous reaction to carbon tattoo following traumatic graphite implantation.

Granulomatous reactions to carbon tattoo may be sarcoidal (foreign-body granulomatous dermatitis), palisading, or rarely tuberculoid (caseating). Sarcoidal granulomatous tattoo reactions may occur in patients with sarcoidosis due to koebnerization, and histology alone is not discriminatory; however, in our patient, the absence of underlying sarcoidosis or clinical or histologic findings of sarcoidosis outside of the site of the pencil-core granuloma excluded that possibility.11 Pencil-core granulomas are characterized by a delayed foreign-body reaction to retained fragments of lead often years following a penetrating trauma with a pencil. Previous reports have described various lag times from injury to lesion growth of up to 58 years.1-10 Our patient claimed to have noticed the lesion growing and becoming painful only after a 62-year lag time following the initial trauma. To our knowledge, this is the longest lag time between the initial pencil injury and induction of the foreign-body reaction reported in the literature. Clinically, the lesion appeared and behaved very similar to a melanoma, prompting further treatment and evaluation.
It has been suggested that the lag period between the initial trauma and the rapid growth of the lesion may correspond to the amount of time required for the breakdown of the pencil lead to a critical size followed by the dispersal of those particles within the interstitium, where they can induce a granulomatous reaction.1,2,9 One case described a patient who reported that the growth and clinical change of the pencil-core granuloma only started when the patient accidentally hit the area where the trauma had occurred 31 years prior.1 This additional trauma may have caused further mechanical breakdown of the lead to set off the tissue reaction. In our case, the patient did not recall any additional trauma to the head prior to the onset of growth of the nodule on the forehead.
Our case indicates that carbon tattoo may be a possible sequela of a penetrating injury from a pencil with retained pencil lead fragments; however, many of these carbon tattoos may remain stable throughout the remainder of the patient’s life.
To the Editor:
Trauma from a pencil tip can sometimes result in a fragment of lead being left embedded within the skin. Pencil lead is composed of 66% graphite carbon, 26% aluminum silicate, and 8% paraffin.1,2 While the toxicity of these individual elements is low, paraffin can cause nonallergic foreign-body reactions, aluminum silicate can induce epithelioid granulomatous reactions, and graphite has been reported to cause chronic granulomatous reactions in the lungs of those who work with graphite.2 Penetrating trauma with a pencil can result in the formation of a cutaneous granulomatous reaction that can sometimes occur years to decades after the initial injury.3,4 Several cases of pencil-core granulomas have been published, with lag times between the initial trauma and lesion growth as long as 58 years.1-10 The pencil-core granuloma may simulate malignant melanoma, as it presents clinically as a growing, darkly pigmented lesion, thus prompting biopsy. We present a case of a pencil-core granuloma that began to grow 62 years after the initial trauma.
A 72-year-old woman was referred to our clinic for evaluation of a dark nodule on the forehead. The lesion had been present since the age of 10 years, reportedly from an accidental stabbing with a pencil. The lesion had been flat, stable, and asymptomatic since the trauma occurred; however, the patient reported that approximately 9 months prior to presentation, it had started growing and became painful. Physical examination revealed a 1.0-cm, round, bluish-black nodule on the right superior forehead (Figure 1A). No satellite lesions or local lymphadenopathy were noted on general examination.

An elliptical excision of the lesion with 1-cm margins revealed a bluish-black mass extending through the dermis, through the frontalis muscle, and into the periosteum and frontal bone (Figure 1B). A No. 15 blade was then used to remove the remaining pigment from the outer table of the frontal bone. Histopathologic findings demonstrated a sarcoidal granulomatous dermatitis associated with abundant, nonpolarizable, black, granular pigment consistent with carbon tattoo. This foreign material was readily identifiable in large extracellular deposits and also within histiocytes, including numerous multinucleated giant cells (Figure 2). Immunostaining for MART-1 and SOX-10 antigens failed to demonstrate a melanocytic proliferation. These findings were consistent with a sarcoidal foreign-body granulomatous reaction to carbon tattoo following traumatic graphite implantation.

Granulomatous reactions to carbon tattoo may be sarcoidal (foreign-body granulomatous dermatitis), palisading, or rarely tuberculoid (caseating). Sarcoidal granulomatous tattoo reactions may occur in patients with sarcoidosis due to koebnerization, and histology alone is not discriminatory; however, in our patient, the absence of underlying sarcoidosis or clinical or histologic findings of sarcoidosis outside of the site of the pencil-core granuloma excluded that possibility.11 Pencil-core granulomas are characterized by a delayed foreign-body reaction to retained fragments of lead often years following a penetrating trauma with a pencil. Previous reports have described various lag times from injury to lesion growth of up to 58 years.1-10 Our patient claimed to have noticed the lesion growing and becoming painful only after a 62-year lag time following the initial trauma. To our knowledge, this is the longest lag time between the initial pencil injury and induction of the foreign-body reaction reported in the literature. Clinically, the lesion appeared and behaved very similar to a melanoma, prompting further treatment and evaluation.
It has been suggested that the lag period between the initial trauma and the rapid growth of the lesion may correspond to the amount of time required for the breakdown of the pencil lead to a critical size followed by the dispersal of those particles within the interstitium, where they can induce a granulomatous reaction.1,2,9 One case described a patient who reported that the growth and clinical change of the pencil-core granuloma only started when the patient accidentally hit the area where the trauma had occurred 31 years prior.1 This additional trauma may have caused further mechanical breakdown of the lead to set off the tissue reaction. In our case, the patient did not recall any additional trauma to the head prior to the onset of growth of the nodule on the forehead.
Our case indicates that carbon tattoo may be a possible sequela of a penetrating injury from a pencil with retained pencil lead fragments; however, many of these carbon tattoos may remain stable throughout the remainder of the patient’s life.
- Gormley RH, Kovach SJ III, Zhang PJ. Role for trauma in inducing pencil “lead” granuloma in the skin. J Am Acad Dermatol. 2010;62:1074-1075.
- Terasawa N, Kishimoto S, Kibe Y, et al. Graphite foreign body granuloma. Br J Dermatol. 1999;141:774-776.
- Fukunaga Y, Hashimoto I, Nakanishi H, et al. Pencil-core granuloma of the face: report of two rare cases. J Plast Reconstr Aesthet Surg. 2011;64:1235-1237.
- Aswani VH, Kim SL. Fifty-three years after a pencil puncture wound. Case Rep Dermatol. 2015;7:303-305.
- Taylor B, Frumkin A, Pitha JV. Delayed reaction to “lead” pencil simulating melanoma. Cutis. 1988;42:199-201.
- Granick MS, Erickson ER, Solomon MP. Pencil-core granuloma. Plast Reconstr Surg. 1992;89:136-138.
- Andreano J. Stump the experts. foreign body granuloma. J Dermatol Surg Oncol. 1992;18:277, 343.
- Yoshitatsu S, Takagi T. A case of giant pencil-core granuloma. J Dermatol. 2000;27:329-332.
- Hatano Y, Asada Y, Komada S, et al. A case of pencil core granuloma with an unusual temporal profile. Dermatology. 2000;201:151-153.
- Seitz IA, Silva BA, Schechter LS. Unusual sequela from a pencil stab wound reveals a retained graphite foreign body. Pediatr Emerg Care. 2014;30:568-570.
- Motaparthi K. Tattoo ink. In: Cockerell CJ, Hall BJ, eds. Nonneoplastic Dermatopathology. 2nd ed. Amirsys; 2016: 270.
- Gormley RH, Kovach SJ III, Zhang PJ. Role for trauma in inducing pencil “lead” granuloma in the skin. J Am Acad Dermatol. 2010;62:1074-1075.
- Terasawa N, Kishimoto S, Kibe Y, et al. Graphite foreign body granuloma. Br J Dermatol. 1999;141:774-776.
- Fukunaga Y, Hashimoto I, Nakanishi H, et al. Pencil-core granuloma of the face: report of two rare cases. J Plast Reconstr Aesthet Surg. 2011;64:1235-1237.
- Aswani VH, Kim SL. Fifty-three years after a pencil puncture wound. Case Rep Dermatol. 2015;7:303-305.
- Taylor B, Frumkin A, Pitha JV. Delayed reaction to “lead” pencil simulating melanoma. Cutis. 1988;42:199-201.
- Granick MS, Erickson ER, Solomon MP. Pencil-core granuloma. Plast Reconstr Surg. 1992;89:136-138.
- Andreano J. Stump the experts. foreign body granuloma. J Dermatol Surg Oncol. 1992;18:277, 343.
- Yoshitatsu S, Takagi T. A case of giant pencil-core granuloma. J Dermatol. 2000;27:329-332.
- Hatano Y, Asada Y, Komada S, et al. A case of pencil core granuloma with an unusual temporal profile. Dermatology. 2000;201:151-153.
- Seitz IA, Silva BA, Schechter LS. Unusual sequela from a pencil stab wound reveals a retained graphite foreign body. Pediatr Emerg Care. 2014;30:568-570.
- Motaparthi K. Tattoo ink. In: Cockerell CJ, Hall BJ, eds. Nonneoplastic Dermatopathology. 2nd ed. Amirsys; 2016: 270.
Practice Points
- Pencil-core granulomas can arise even decades after the lead is embedded in the skin.
- It is important to biopsy to confirm the diagnosis, as pencil-core granulomas can very closely mimic melanomas.
“I didn’t want to meet you.” Dispelling myths about palliative care
The names of health care professionals and patients cited within the dialogue text have been changed to protect their privacy.
but over the years I have come to realize that she was right – most people, including many within health care, don’t have a good appreciation of what palliative care is or how it can help patients and health care teams.
A recent national survey about cancer-related health information found that of more than 1,000 surveyed Americans, less than 30% professed any knowledge of palliative care. Of those who had some knowledge of palliative care, around 30% believed palliative care was synonymous with hospice.1 Another 15% believed that a patient would have to give up cancer-directed treatments to receive palliative care.1
It’s not giving up
This persistent belief that palliative care is equivalent to hospice, or is tantamount to “giving up,” is one of the most commonly held myths I encounter in everyday practice.
I knock on the exam door and walk in.
A small, trim woman in her late 50s is sitting in a chair, arms folded across her chest, face drawn in.
“Hi,” I start. “I’m Sarah, the palliative care nurse practitioner who works in this clinic. I work closely with Dr. Smith.”
Dr. Smith is the patient’s oncologist.
“I really didn’t want to meet you,” she says in a quiet voice, her eyes large with concern.
I don’t take it personally. Few patients really want to be in the position of needing to meet the palliative care team.
“I looked up palliative care on Google and saw the word hospice.”
“Yeah,” I say. “I hear that a lot. Well, I can reassure you that this isn’t hospice.
In this clinic, our focus is on your cancer symptoms, your treatment side effects, and your quality of life.”
She looks visibly relieved. “Quality of life,” she echoes. “I need more of that.”
“OK,” I say. “So, tell me what you’re struggling with the most right now.”
That’s how many palliative care visits start. I actually prefer if patients haven’t heard of palliative care because it allows me to frame it for them, rather than having to start by addressing a myth or a prior negative experience. Even when patients haven’t had a negative experience with palliative care per se, typically, if they’ve interacted with palliative care in the past, it’s usually because someone they loved died in a hospital setting and it is the memory of that terrible loss that becomes synonymous with their recollection of palliative care.
Many patients I meet have never seen another outpatient palliative care practitioner – and this makes sense – we are still too few and far between. Most established palliative care teams are hospital based and many patients seen in the community do not have easy access to palliative care teams where they receive oncologic care.2 As an embedded practitioner, I see patients in the same exam rooms and infusion centers where they receive their cancer therapies, so I’m effectively woven into the fabric of their oncology experience. Just being there in the cancer center allows me to be in the right place at the right time for the right patients and their care teams.
More than pain management
Another myth I tend to dispel a lot is that palliative care is just a euphemism for “pain management.” I have seen this less lately, but still occasionally in the chart I’ll see documented in a note, “patient is seeing palliative/pain management,” when a patient is seeing me or one of my colleagues. Unfortunately, when providers have limited or outdated views of what palliative care is or the value it brings to patient-centered cancer care, referrals to palliative care tend to be delayed.3
“I really think Ms. Lopez could benefit from seeing palliative care,” an oncology nurse practitioner says to an oncologist.
I’m standing nearby, about to see another patient in one of the exam rooms in our clinic.
“But I don’t think she’s ready. And besides, she doesn’t have any pain,” he says.
He turns to me quizzically. “What do you think?”
“Tell me about the patient,” I ask, taking a few steps in their direction.
“Well, she’s a 64-year-old woman with metastatic cancer.
She has a really poor appetite and is losing some weight.
Seems a bit down, kind of pessimistic about things.
Her scan showed some new growth, so guess I’m not surprised by that.”
“I might be able to help her with the appetite and the mood changes.
I can at least talk with her and see where she’s at,” I offer.
“Alright,” he says. “We’ll put the palliative referral in.”
He hesitates. “But are you sure you want to see her?
She doesn’t have any pain.” He sounds skeptical.
“Yeah, I mean, it sounds like she has symptoms that are bothering her, so I’d be happy to see her. She sounds completely appropriate for palliative care.”
I hear this assumption a lot – that palliative care is somehow equivalent to pain management and that unless a patient’s pain is severe, it’s not worth referring the patient to palliative care. Don’t get me wrong – we do a lot of pain management, but at its heart, palliative care is an interdisciplinary specialty focused on improving or maintaining quality of life for people with serious illness. Because the goal is so broad, care can take many shapes.4
In addition to pain, palliative care clinicians commonly treat nausea, shortness of breath, constipation or diarrhea, poor appetite, fatigue, anxiety, depression, and insomnia.
Palliative care is more than medical or nursing care
A related misconception about palliative care held by many lay people and health care workers alike is that palliative care is primarily medical or nursing care focused mostly on alleviating physical symptoms such as pain or nausea. This couldn’t be further from the truth.
We’ve been talking for a while.
Ms. Lopez tells me about her struggles to maintain her weight while undergoing chemotherapy. She has low-grade nausea that is impacting her ability and desire to eat more and didn’t think that her weight loss was severe enough to warrant taking medication.
We talk about how she may be able to use antinausea medication sparingly to alleviate nausea while also limiting side effects from the medications—which was a big concern for her.
I ask her what else is bothering her.
She tells me that she has always been a strong Catholic and even when life has gotten tough, her faith was never shaken – until now.
She is struggling to understand why she ended up with metastatic cancer at such a relatively young age—why would God do this to her?
She had plans for retirement that have since evaporated in the face of a foreshortened life.
Why did this happen to her of all people? She was completely healthy until her diagnosis.
Her face is wet with tears.
We talk a little about how a diagnosis like this can change so much of a person’s life and identity. I try to validate her experience. She’s clearly suffering from a sense that her life is not what she expected, and she is struggling to integrate how her future looks at this point.
I ask her what conversations with her priest have been like.
At this point you may be wondering where this conversation is going. Why are we talking about Ms. Lopez’s religion? Palliative care is best delivered through high functioning interdisciplinary teams that can include other supportive people in a patient’s life. We work in concert to try to bring comfort to a patient and their family.4 That support network can include nurses, physicians, social workers, and chaplains. In this case, Ms. Lopez had not yet reached out to her priest. She hasn’t had the time or energy to contact her priest given her symptoms.
“Can I contact your priest for you?
Maybe he can visit or call and chat with you?”
She nods and wipes tears away.
“That would be really nice,” she says. “I’d love it if he could pray with me.”
A few hours after the visit, I call Ms. Lopez’s priest.
I ask him to reach out to her and about her request for prayer.
He says he’s been thinking about her and that her presence has been missed at weekly Mass. He thanks me for the call and says he’ll call her tomorrow.
I say my own small prayer for Ms. Lopez and head home, the day’s work completed.
Sarah D'Ambruoso was born and raised in Maine. She completed her undergraduate and graduate nursing education at New York University and UCLA, respectively, and currently works as a palliative care nurse practitioner in an oncology clinic in Los Angeles.
References
1. Cheng BT et al. Patterns of palliative care beliefs among adults in the U.S.: Analysis of a National Cancer Database. J Pain Symptom Manage. 2019 Aug 10. doi: 10.1016/j.jpainsymman.2019.07.030.
2. Finlay E et al. Filling the gap: Creating an outpatient palliative care program in your institution. Am Soc Clin Oncol Educ Book. 2018 May 23. doi: 10.1200/EDBK_200775.
3. Von Roenn JH et al. Barriers and approaches to the successful integration of palliative care and oncology practice. J Natl Compr Canc Netw. 2013 Mar. doi: 10.6004/jnccn.2013.0209.
4. Ferrell BR et al. Integration of palliative care into standard oncology care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2016 Oct 31. doi: 10.1200/JCO.2016.70.1474.
The names of health care professionals and patients cited within the dialogue text have been changed to protect their privacy.
but over the years I have come to realize that she was right – most people, including many within health care, don’t have a good appreciation of what palliative care is or how it can help patients and health care teams.
A recent national survey about cancer-related health information found that of more than 1,000 surveyed Americans, less than 30% professed any knowledge of palliative care. Of those who had some knowledge of palliative care, around 30% believed palliative care was synonymous with hospice.1 Another 15% believed that a patient would have to give up cancer-directed treatments to receive palliative care.1
It’s not giving up
This persistent belief that palliative care is equivalent to hospice, or is tantamount to “giving up,” is one of the most commonly held myths I encounter in everyday practice.
I knock on the exam door and walk in.
A small, trim woman in her late 50s is sitting in a chair, arms folded across her chest, face drawn in.
“Hi,” I start. “I’m Sarah, the palliative care nurse practitioner who works in this clinic. I work closely with Dr. Smith.”
Dr. Smith is the patient’s oncologist.
“I really didn’t want to meet you,” she says in a quiet voice, her eyes large with concern.
I don’t take it personally. Few patients really want to be in the position of needing to meet the palliative care team.
“I looked up palliative care on Google and saw the word hospice.”
“Yeah,” I say. “I hear that a lot. Well, I can reassure you that this isn’t hospice.
In this clinic, our focus is on your cancer symptoms, your treatment side effects, and your quality of life.”
She looks visibly relieved. “Quality of life,” she echoes. “I need more of that.”
“OK,” I say. “So, tell me what you’re struggling with the most right now.”
That’s how many palliative care visits start. I actually prefer if patients haven’t heard of palliative care because it allows me to frame it for them, rather than having to start by addressing a myth or a prior negative experience. Even when patients haven’t had a negative experience with palliative care per se, typically, if they’ve interacted with palliative care in the past, it’s usually because someone they loved died in a hospital setting and it is the memory of that terrible loss that becomes synonymous with their recollection of palliative care.
Many patients I meet have never seen another outpatient palliative care practitioner – and this makes sense – we are still too few and far between. Most established palliative care teams are hospital based and many patients seen in the community do not have easy access to palliative care teams where they receive oncologic care.2 As an embedded practitioner, I see patients in the same exam rooms and infusion centers where they receive their cancer therapies, so I’m effectively woven into the fabric of their oncology experience. Just being there in the cancer center allows me to be in the right place at the right time for the right patients and their care teams.
More than pain management
Another myth I tend to dispel a lot is that palliative care is just a euphemism for “pain management.” I have seen this less lately, but still occasionally in the chart I’ll see documented in a note, “patient is seeing palliative/pain management,” when a patient is seeing me or one of my colleagues. Unfortunately, when providers have limited or outdated views of what palliative care is or the value it brings to patient-centered cancer care, referrals to palliative care tend to be delayed.3
“I really think Ms. Lopez could benefit from seeing palliative care,” an oncology nurse practitioner says to an oncologist.
I’m standing nearby, about to see another patient in one of the exam rooms in our clinic.
“But I don’t think she’s ready. And besides, she doesn’t have any pain,” he says.
He turns to me quizzically. “What do you think?”
“Tell me about the patient,” I ask, taking a few steps in their direction.
“Well, she’s a 64-year-old woman with metastatic cancer.
She has a really poor appetite and is losing some weight.
Seems a bit down, kind of pessimistic about things.
Her scan showed some new growth, so guess I’m not surprised by that.”
“I might be able to help her with the appetite and the mood changes.
I can at least talk with her and see where she’s at,” I offer.
“Alright,” he says. “We’ll put the palliative referral in.”
He hesitates. “But are you sure you want to see her?
She doesn’t have any pain.” He sounds skeptical.
“Yeah, I mean, it sounds like she has symptoms that are bothering her, so I’d be happy to see her. She sounds completely appropriate for palliative care.”
I hear this assumption a lot – that palliative care is somehow equivalent to pain management and that unless a patient’s pain is severe, it’s not worth referring the patient to palliative care. Don’t get me wrong – we do a lot of pain management, but at its heart, palliative care is an interdisciplinary specialty focused on improving or maintaining quality of life for people with serious illness. Because the goal is so broad, care can take many shapes.4
In addition to pain, palliative care clinicians commonly treat nausea, shortness of breath, constipation or diarrhea, poor appetite, fatigue, anxiety, depression, and insomnia.
Palliative care is more than medical or nursing care
A related misconception about palliative care held by many lay people and health care workers alike is that palliative care is primarily medical or nursing care focused mostly on alleviating physical symptoms such as pain or nausea. This couldn’t be further from the truth.
We’ve been talking for a while.
Ms. Lopez tells me about her struggles to maintain her weight while undergoing chemotherapy. She has low-grade nausea that is impacting her ability and desire to eat more and didn’t think that her weight loss was severe enough to warrant taking medication.
We talk about how she may be able to use antinausea medication sparingly to alleviate nausea while also limiting side effects from the medications—which was a big concern for her.
I ask her what else is bothering her.
She tells me that she has always been a strong Catholic and even when life has gotten tough, her faith was never shaken – until now.
She is struggling to understand why she ended up with metastatic cancer at such a relatively young age—why would God do this to her?
She had plans for retirement that have since evaporated in the face of a foreshortened life.
Why did this happen to her of all people? She was completely healthy until her diagnosis.
Her face is wet with tears.
We talk a little about how a diagnosis like this can change so much of a person’s life and identity. I try to validate her experience. She’s clearly suffering from a sense that her life is not what she expected, and she is struggling to integrate how her future looks at this point.
I ask her what conversations with her priest have been like.
At this point you may be wondering where this conversation is going. Why are we talking about Ms. Lopez’s religion? Palliative care is best delivered through high functioning interdisciplinary teams that can include other supportive people in a patient’s life. We work in concert to try to bring comfort to a patient and their family.4 That support network can include nurses, physicians, social workers, and chaplains. In this case, Ms. Lopez had not yet reached out to her priest. She hasn’t had the time or energy to contact her priest given her symptoms.
“Can I contact your priest for you?
Maybe he can visit or call and chat with you?”
She nods and wipes tears away.
“That would be really nice,” she says. “I’d love it if he could pray with me.”
A few hours after the visit, I call Ms. Lopez’s priest.
I ask him to reach out to her and about her request for prayer.
He says he’s been thinking about her and that her presence has been missed at weekly Mass. He thanks me for the call and says he’ll call her tomorrow.
I say my own small prayer for Ms. Lopez and head home, the day’s work completed.
Sarah D'Ambruoso was born and raised in Maine. She completed her undergraduate and graduate nursing education at New York University and UCLA, respectively, and currently works as a palliative care nurse practitioner in an oncology clinic in Los Angeles.
References
1. Cheng BT et al. Patterns of palliative care beliefs among adults in the U.S.: Analysis of a National Cancer Database. J Pain Symptom Manage. 2019 Aug 10. doi: 10.1016/j.jpainsymman.2019.07.030.
2. Finlay E et al. Filling the gap: Creating an outpatient palliative care program in your institution. Am Soc Clin Oncol Educ Book. 2018 May 23. doi: 10.1200/EDBK_200775.
3. Von Roenn JH et al. Barriers and approaches to the successful integration of palliative care and oncology practice. J Natl Compr Canc Netw. 2013 Mar. doi: 10.6004/jnccn.2013.0209.
4. Ferrell BR et al. Integration of palliative care into standard oncology care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2016 Oct 31. doi: 10.1200/JCO.2016.70.1474.
The names of health care professionals and patients cited within the dialogue text have been changed to protect their privacy.
but over the years I have come to realize that she was right – most people, including many within health care, don’t have a good appreciation of what palliative care is or how it can help patients and health care teams.
A recent national survey about cancer-related health information found that of more than 1,000 surveyed Americans, less than 30% professed any knowledge of palliative care. Of those who had some knowledge of palliative care, around 30% believed palliative care was synonymous with hospice.1 Another 15% believed that a patient would have to give up cancer-directed treatments to receive palliative care.1
It’s not giving up
This persistent belief that palliative care is equivalent to hospice, or is tantamount to “giving up,” is one of the most commonly held myths I encounter in everyday practice.
I knock on the exam door and walk in.
A small, trim woman in her late 50s is sitting in a chair, arms folded across her chest, face drawn in.
“Hi,” I start. “I’m Sarah, the palliative care nurse practitioner who works in this clinic. I work closely with Dr. Smith.”
Dr. Smith is the patient’s oncologist.
“I really didn’t want to meet you,” she says in a quiet voice, her eyes large with concern.
I don’t take it personally. Few patients really want to be in the position of needing to meet the palliative care team.
“I looked up palliative care on Google and saw the word hospice.”
“Yeah,” I say. “I hear that a lot. Well, I can reassure you that this isn’t hospice.
In this clinic, our focus is on your cancer symptoms, your treatment side effects, and your quality of life.”
She looks visibly relieved. “Quality of life,” she echoes. “I need more of that.”
“OK,” I say. “So, tell me what you’re struggling with the most right now.”
That’s how many palliative care visits start. I actually prefer if patients haven’t heard of palliative care because it allows me to frame it for them, rather than having to start by addressing a myth or a prior negative experience. Even when patients haven’t had a negative experience with palliative care per se, typically, if they’ve interacted with palliative care in the past, it’s usually because someone they loved died in a hospital setting and it is the memory of that terrible loss that becomes synonymous with their recollection of palliative care.
Many patients I meet have never seen another outpatient palliative care practitioner – and this makes sense – we are still too few and far between. Most established palliative care teams are hospital based and many patients seen in the community do not have easy access to palliative care teams where they receive oncologic care.2 As an embedded practitioner, I see patients in the same exam rooms and infusion centers where they receive their cancer therapies, so I’m effectively woven into the fabric of their oncology experience. Just being there in the cancer center allows me to be in the right place at the right time for the right patients and their care teams.
More than pain management
Another myth I tend to dispel a lot is that palliative care is just a euphemism for “pain management.” I have seen this less lately, but still occasionally in the chart I’ll see documented in a note, “patient is seeing palliative/pain management,” when a patient is seeing me or one of my colleagues. Unfortunately, when providers have limited or outdated views of what palliative care is or the value it brings to patient-centered cancer care, referrals to palliative care tend to be delayed.3
“I really think Ms. Lopez could benefit from seeing palliative care,” an oncology nurse practitioner says to an oncologist.
I’m standing nearby, about to see another patient in one of the exam rooms in our clinic.
“But I don’t think she’s ready. And besides, she doesn’t have any pain,” he says.
He turns to me quizzically. “What do you think?”
“Tell me about the patient,” I ask, taking a few steps in their direction.
“Well, she’s a 64-year-old woman with metastatic cancer.
She has a really poor appetite and is losing some weight.
Seems a bit down, kind of pessimistic about things.
Her scan showed some new growth, so guess I’m not surprised by that.”
“I might be able to help her with the appetite and the mood changes.
I can at least talk with her and see where she’s at,” I offer.
“Alright,” he says. “We’ll put the palliative referral in.”
He hesitates. “But are you sure you want to see her?
She doesn’t have any pain.” He sounds skeptical.
“Yeah, I mean, it sounds like she has symptoms that are bothering her, so I’d be happy to see her. She sounds completely appropriate for palliative care.”
I hear this assumption a lot – that palliative care is somehow equivalent to pain management and that unless a patient’s pain is severe, it’s not worth referring the patient to palliative care. Don’t get me wrong – we do a lot of pain management, but at its heart, palliative care is an interdisciplinary specialty focused on improving or maintaining quality of life for people with serious illness. Because the goal is so broad, care can take many shapes.4
In addition to pain, palliative care clinicians commonly treat nausea, shortness of breath, constipation or diarrhea, poor appetite, fatigue, anxiety, depression, and insomnia.
Palliative care is more than medical or nursing care
A related misconception about palliative care held by many lay people and health care workers alike is that palliative care is primarily medical or nursing care focused mostly on alleviating physical symptoms such as pain or nausea. This couldn’t be further from the truth.
We’ve been talking for a while.
Ms. Lopez tells me about her struggles to maintain her weight while undergoing chemotherapy. She has low-grade nausea that is impacting her ability and desire to eat more and didn’t think that her weight loss was severe enough to warrant taking medication.
We talk about how she may be able to use antinausea medication sparingly to alleviate nausea while also limiting side effects from the medications—which was a big concern for her.
I ask her what else is bothering her.
She tells me that she has always been a strong Catholic and even when life has gotten tough, her faith was never shaken – until now.
She is struggling to understand why she ended up with metastatic cancer at such a relatively young age—why would God do this to her?
She had plans for retirement that have since evaporated in the face of a foreshortened life.
Why did this happen to her of all people? She was completely healthy until her diagnosis.
Her face is wet with tears.
We talk a little about how a diagnosis like this can change so much of a person’s life and identity. I try to validate her experience. She’s clearly suffering from a sense that her life is not what she expected, and she is struggling to integrate how her future looks at this point.
I ask her what conversations with her priest have been like.
At this point you may be wondering where this conversation is going. Why are we talking about Ms. Lopez’s religion? Palliative care is best delivered through high functioning interdisciplinary teams that can include other supportive people in a patient’s life. We work in concert to try to bring comfort to a patient and their family.4 That support network can include nurses, physicians, social workers, and chaplains. In this case, Ms. Lopez had not yet reached out to her priest. She hasn’t had the time or energy to contact her priest given her symptoms.
“Can I contact your priest for you?
Maybe he can visit or call and chat with you?”
She nods and wipes tears away.
“That would be really nice,” she says. “I’d love it if he could pray with me.”
A few hours after the visit, I call Ms. Lopez’s priest.
I ask him to reach out to her and about her request for prayer.
He says he’s been thinking about her and that her presence has been missed at weekly Mass. He thanks me for the call and says he’ll call her tomorrow.
I say my own small prayer for Ms. Lopez and head home, the day’s work completed.
Sarah D'Ambruoso was born and raised in Maine. She completed her undergraduate and graduate nursing education at New York University and UCLA, respectively, and currently works as a palliative care nurse practitioner in an oncology clinic in Los Angeles.
References
1. Cheng BT et al. Patterns of palliative care beliefs among adults in the U.S.: Analysis of a National Cancer Database. J Pain Symptom Manage. 2019 Aug 10. doi: 10.1016/j.jpainsymman.2019.07.030.
2. Finlay E et al. Filling the gap: Creating an outpatient palliative care program in your institution. Am Soc Clin Oncol Educ Book. 2018 May 23. doi: 10.1200/EDBK_200775.
3. Von Roenn JH et al. Barriers and approaches to the successful integration of palliative care and oncology practice. J Natl Compr Canc Netw. 2013 Mar. doi: 10.6004/jnccn.2013.0209.
4. Ferrell BR et al. Integration of palliative care into standard oncology care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2016 Oct 31. doi: 10.1200/JCO.2016.70.1474.
Medicare NCDs hinder access to cancer biomarker testing for minorities
of data from patients with advanced non–small cell lung cancer (aNSCLC), metastatic colorectal cancer, metastatic breast cancer, or advanced melanoma. The finding was reported in JAMA Network Open.
Biomarker testing has become an essential tool in cancer care over the last decade. In 2011, for example, less than 1% of patients with aNSCLC, metastatic colorectal cancer, metastatic breast cancer, and advanced melanoma underwent NGS testing, but by 2019, 40% of patients with these cancers received the testing.
“Next-generation sequencing testing has become increasingly important because it enables identification of multiple biomarkers simultaneously and efficiently while minimizing the number of biopsies required,” wrote the authors, led by William B. Wong, PharmD, of Genentech.
It has been unknown whether for Medicare beneficiaries and the overall population, if the NCD affected health equity issues, the authors wrote. While increased use of appropriate targeted therapies facilitated by NGS testing is associated with improved survival rates in patients with advanced or metastatic cancer, variability in health care coverage policies has posed a significant barrier to obtaining NGS testing for cancer patients, specifically through policy coverage limitations. It has remained unclear if the NCD has influenced NGS testing coverage in insurance types (for example, Medicaid) encompassing a larger population of minority racial and ethnic groups often experiencing poorer care and outcomes.
The retrospective cohort analysis compared EHR data from 280 U.S. cancer clinics in the (800 sites of care) pre- versus post-NCD period for patients with aNSCLC, metastatic colorectal cancer, metastatic breast cancer, or advanced melanoma (January 2011–March 2020). Nearly 70% of all patients in the study were Medicare recipients who needed NCD approval to cover the cost of testing.
Among 92,687 patients (mean age, 66.6 years; 55.7% women), compared with Medicare beneficiaries, changes in pre- to post-NCD NGS testing trends were similar in commercially insured patients (odds ratio, 1.03; 95% CI, 0.98-1.08; P = .25). Pre- to post-NCD NGS testing trends increased at a slower rate among patients in assistance programs (OR, 0.93; 95% CI, 0.87-0.99; P = .03), compared with Medicare beneficiaries. The rate of increase for patients receiving Medicaid was not significantly different statistically compared with those receiving Medicare (OR, 0.92; 95% CI, 0.84-1.01; P = .07). Also, the NCD was not associated with racial and ethnic groups within Medicare beneficiaries alone or across all insurance types.
Compared with non-Hispanic White individuals, increases in average NGS use from the pre-NCD to post-NCD period were 14% lower (OR, 0.86; 95% CI, 0.74-0.99; P = .04) among African American and 23% lower (OR, 0.77; 95% CI, 0.62-0.96; P = .02) among Hispanic/Latino individuals; increases were similar, however, among Asian individuals and other races and ethnicities.
The authors observed that the post-NCD trend of increasing NGS testing seen in Medicare beneficiaries was similarly observed in those with commercial insurance. Testing rate differences, however, widened or were maintained after versus before the NCD in PAP (personal assistance program) and Medicaid beneficiaries relative to Medicare beneficiaries, suggesting that access to NGS testing did not improve equally across insurance types. Since Medicare coverage is determined at the state level, the authors urged research examining individual state coverage policies to further elucidate factors slowing uptake among Medicaid beneficiaries. “Additional efforts beyond coverage policies,” the authors concluded, “are needed to ensure equitable access to the benefits of precision medicine.”
The study was supported by Genentech.
of data from patients with advanced non–small cell lung cancer (aNSCLC), metastatic colorectal cancer, metastatic breast cancer, or advanced melanoma. The finding was reported in JAMA Network Open.
Biomarker testing has become an essential tool in cancer care over the last decade. In 2011, for example, less than 1% of patients with aNSCLC, metastatic colorectal cancer, metastatic breast cancer, and advanced melanoma underwent NGS testing, but by 2019, 40% of patients with these cancers received the testing.
“Next-generation sequencing testing has become increasingly important because it enables identification of multiple biomarkers simultaneously and efficiently while minimizing the number of biopsies required,” wrote the authors, led by William B. Wong, PharmD, of Genentech.
It has been unknown whether for Medicare beneficiaries and the overall population, if the NCD affected health equity issues, the authors wrote. While increased use of appropriate targeted therapies facilitated by NGS testing is associated with improved survival rates in patients with advanced or metastatic cancer, variability in health care coverage policies has posed a significant barrier to obtaining NGS testing for cancer patients, specifically through policy coverage limitations. It has remained unclear if the NCD has influenced NGS testing coverage in insurance types (for example, Medicaid) encompassing a larger population of minority racial and ethnic groups often experiencing poorer care and outcomes.
The retrospective cohort analysis compared EHR data from 280 U.S. cancer clinics in the (800 sites of care) pre- versus post-NCD period for patients with aNSCLC, metastatic colorectal cancer, metastatic breast cancer, or advanced melanoma (January 2011–March 2020). Nearly 70% of all patients in the study were Medicare recipients who needed NCD approval to cover the cost of testing.
Among 92,687 patients (mean age, 66.6 years; 55.7% women), compared with Medicare beneficiaries, changes in pre- to post-NCD NGS testing trends were similar in commercially insured patients (odds ratio, 1.03; 95% CI, 0.98-1.08; P = .25). Pre- to post-NCD NGS testing trends increased at a slower rate among patients in assistance programs (OR, 0.93; 95% CI, 0.87-0.99; P = .03), compared with Medicare beneficiaries. The rate of increase for patients receiving Medicaid was not significantly different statistically compared with those receiving Medicare (OR, 0.92; 95% CI, 0.84-1.01; P = .07). Also, the NCD was not associated with racial and ethnic groups within Medicare beneficiaries alone or across all insurance types.
Compared with non-Hispanic White individuals, increases in average NGS use from the pre-NCD to post-NCD period were 14% lower (OR, 0.86; 95% CI, 0.74-0.99; P = .04) among African American and 23% lower (OR, 0.77; 95% CI, 0.62-0.96; P = .02) among Hispanic/Latino individuals; increases were similar, however, among Asian individuals and other races and ethnicities.
The authors observed that the post-NCD trend of increasing NGS testing seen in Medicare beneficiaries was similarly observed in those with commercial insurance. Testing rate differences, however, widened or were maintained after versus before the NCD in PAP (personal assistance program) and Medicaid beneficiaries relative to Medicare beneficiaries, suggesting that access to NGS testing did not improve equally across insurance types. Since Medicare coverage is determined at the state level, the authors urged research examining individual state coverage policies to further elucidate factors slowing uptake among Medicaid beneficiaries. “Additional efforts beyond coverage policies,” the authors concluded, “are needed to ensure equitable access to the benefits of precision medicine.”
The study was supported by Genentech.
of data from patients with advanced non–small cell lung cancer (aNSCLC), metastatic colorectal cancer, metastatic breast cancer, or advanced melanoma. The finding was reported in JAMA Network Open.
Biomarker testing has become an essential tool in cancer care over the last decade. In 2011, for example, less than 1% of patients with aNSCLC, metastatic colorectal cancer, metastatic breast cancer, and advanced melanoma underwent NGS testing, but by 2019, 40% of patients with these cancers received the testing.
“Next-generation sequencing testing has become increasingly important because it enables identification of multiple biomarkers simultaneously and efficiently while minimizing the number of biopsies required,” wrote the authors, led by William B. Wong, PharmD, of Genentech.
It has been unknown whether for Medicare beneficiaries and the overall population, if the NCD affected health equity issues, the authors wrote. While increased use of appropriate targeted therapies facilitated by NGS testing is associated with improved survival rates in patients with advanced or metastatic cancer, variability in health care coverage policies has posed a significant barrier to obtaining NGS testing for cancer patients, specifically through policy coverage limitations. It has remained unclear if the NCD has influenced NGS testing coverage in insurance types (for example, Medicaid) encompassing a larger population of minority racial and ethnic groups often experiencing poorer care and outcomes.
The retrospective cohort analysis compared EHR data from 280 U.S. cancer clinics in the (800 sites of care) pre- versus post-NCD period for patients with aNSCLC, metastatic colorectal cancer, metastatic breast cancer, or advanced melanoma (January 2011–March 2020). Nearly 70% of all patients in the study were Medicare recipients who needed NCD approval to cover the cost of testing.
Among 92,687 patients (mean age, 66.6 years; 55.7% women), compared with Medicare beneficiaries, changes in pre- to post-NCD NGS testing trends were similar in commercially insured patients (odds ratio, 1.03; 95% CI, 0.98-1.08; P = .25). Pre- to post-NCD NGS testing trends increased at a slower rate among patients in assistance programs (OR, 0.93; 95% CI, 0.87-0.99; P = .03), compared with Medicare beneficiaries. The rate of increase for patients receiving Medicaid was not significantly different statistically compared with those receiving Medicare (OR, 0.92; 95% CI, 0.84-1.01; P = .07). Also, the NCD was not associated with racial and ethnic groups within Medicare beneficiaries alone or across all insurance types.
Compared with non-Hispanic White individuals, increases in average NGS use from the pre-NCD to post-NCD period were 14% lower (OR, 0.86; 95% CI, 0.74-0.99; P = .04) among African American and 23% lower (OR, 0.77; 95% CI, 0.62-0.96; P = .02) among Hispanic/Latino individuals; increases were similar, however, among Asian individuals and other races and ethnicities.
The authors observed that the post-NCD trend of increasing NGS testing seen in Medicare beneficiaries was similarly observed in those with commercial insurance. Testing rate differences, however, widened or were maintained after versus before the NCD in PAP (personal assistance program) and Medicaid beneficiaries relative to Medicare beneficiaries, suggesting that access to NGS testing did not improve equally across insurance types. Since Medicare coverage is determined at the state level, the authors urged research examining individual state coverage policies to further elucidate factors slowing uptake among Medicaid beneficiaries. “Additional efforts beyond coverage policies,” the authors concluded, “are needed to ensure equitable access to the benefits of precision medicine.”
The study was supported by Genentech.
FROM JAMA NETWORK OPEN
Pioneering test predicts return of malignant melanoma
Their research, published in the British Journal of Dermatology, describes how early-stage melanomas at risk of spreading secrete transforming growth factor beta2 (TGF-beta2), which causes the reduction, or down-regulation, of the proteins AMBRA1 and loricrin, both of which are found in the skin overlaying the tumor. TGF-beta2 also causes the loss of claudin-1, which in turn leads to loss of skin integrity, facilitating ulceration.
Senior author Penny Lovat, PhD, professor of cellular dermatology and oncology at Newcastle University, and chief scientific officer at AMLo Biosciences, explained: “AMBRA1, loricrin, and claudin-1 are all proteins key to maintaining the integrity of the upper layer of the skin,” and that the loss of these proteins causes gaps to develop, allowing the tumor to spread and ulcerate – a process associated with high-risk tumors. Dr. Lovat likened the process to that of “mortar and bricks holding together a wall”, with the loss of these proteins being “like the mortar crumbling away in the wall.”
According to Cancer Research UK, there are over 16,000 new cases of melanoma skin cancer each year in the United Kingdom, with over 2,000 deaths annually. After being surgically removed, primary tumors are histologically staged, with even low-risk cases being followed up for a number of years, a process that can be time-consuming for patients and costly for the NHS.
Some reassurance for those with melanoma
The creators of the new test say that it is these low-risk patients that the test is able to identify, offering a degree of reassurance to those diagnosed with the disease, and potentially reducing the number of hospital clinic visits they require.
Dr. Lovat commented: “Our test offers a personalized prognosis as it more accurately predicts if your skin cancer is unlikely to spread.”
She added that the test will aid clinicians to identify genuinely low-risk patients diagnosed with an early-stage melanoma, reducing the number of follow-up appointments for those identified as low risk. It, therefore, offers the opportunity to save the NHS time and money.
Excellent news for those with skin cancer
Phil Brady, chief operating officer of the British Skin Foundation, echoed Dr. Lovat’s comments, saying: “The test can alleviate stress and anxiety for patients caused by this potentially deadly skin cancer, whilst increasing efficiency and reducing costs to the NHS.”
Nick Levell, MD, consultant dermatologist & British Skin Foundation spokesperson, who has not been involved in the research, commented how the arrival of the test was “excellent news,” adding that “people at low risk can be reassured and will not have to attend hospital so often for check-ups”.
The development of the new test AMBLor has been led by Dr. Lovat, in association with the university spin-out company AMLo Biosciences, and is accredited by the National Accreditation Body for the United Kingdom. The test involves tissue sections from the standard biopsy being sent in the post to the lab for analysis and costs £293 plus VAT. Currently available through a private referral service, the Newcastle team have applied for the test to be made available on the NHS.
A version of this article first appeared on Medscape UK.
Their research, published in the British Journal of Dermatology, describes how early-stage melanomas at risk of spreading secrete transforming growth factor beta2 (TGF-beta2), which causes the reduction, or down-regulation, of the proteins AMBRA1 and loricrin, both of which are found in the skin overlaying the tumor. TGF-beta2 also causes the loss of claudin-1, which in turn leads to loss of skin integrity, facilitating ulceration.
Senior author Penny Lovat, PhD, professor of cellular dermatology and oncology at Newcastle University, and chief scientific officer at AMLo Biosciences, explained: “AMBRA1, loricrin, and claudin-1 are all proteins key to maintaining the integrity of the upper layer of the skin,” and that the loss of these proteins causes gaps to develop, allowing the tumor to spread and ulcerate – a process associated with high-risk tumors. Dr. Lovat likened the process to that of “mortar and bricks holding together a wall”, with the loss of these proteins being “like the mortar crumbling away in the wall.”
According to Cancer Research UK, there are over 16,000 new cases of melanoma skin cancer each year in the United Kingdom, with over 2,000 deaths annually. After being surgically removed, primary tumors are histologically staged, with even low-risk cases being followed up for a number of years, a process that can be time-consuming for patients and costly for the NHS.
Some reassurance for those with melanoma
The creators of the new test say that it is these low-risk patients that the test is able to identify, offering a degree of reassurance to those diagnosed with the disease, and potentially reducing the number of hospital clinic visits they require.
Dr. Lovat commented: “Our test offers a personalized prognosis as it more accurately predicts if your skin cancer is unlikely to spread.”
She added that the test will aid clinicians to identify genuinely low-risk patients diagnosed with an early-stage melanoma, reducing the number of follow-up appointments for those identified as low risk. It, therefore, offers the opportunity to save the NHS time and money.
Excellent news for those with skin cancer
Phil Brady, chief operating officer of the British Skin Foundation, echoed Dr. Lovat’s comments, saying: “The test can alleviate stress and anxiety for patients caused by this potentially deadly skin cancer, whilst increasing efficiency and reducing costs to the NHS.”
Nick Levell, MD, consultant dermatologist & British Skin Foundation spokesperson, who has not been involved in the research, commented how the arrival of the test was “excellent news,” adding that “people at low risk can be reassured and will not have to attend hospital so often for check-ups”.
The development of the new test AMBLor has been led by Dr. Lovat, in association with the university spin-out company AMLo Biosciences, and is accredited by the National Accreditation Body for the United Kingdom. The test involves tissue sections from the standard biopsy being sent in the post to the lab for analysis and costs £293 plus VAT. Currently available through a private referral service, the Newcastle team have applied for the test to be made available on the NHS.
A version of this article first appeared on Medscape UK.
Their research, published in the British Journal of Dermatology, describes how early-stage melanomas at risk of spreading secrete transforming growth factor beta2 (TGF-beta2), which causes the reduction, or down-regulation, of the proteins AMBRA1 and loricrin, both of which are found in the skin overlaying the tumor. TGF-beta2 also causes the loss of claudin-1, which in turn leads to loss of skin integrity, facilitating ulceration.
Senior author Penny Lovat, PhD, professor of cellular dermatology and oncology at Newcastle University, and chief scientific officer at AMLo Biosciences, explained: “AMBRA1, loricrin, and claudin-1 are all proteins key to maintaining the integrity of the upper layer of the skin,” and that the loss of these proteins causes gaps to develop, allowing the tumor to spread and ulcerate – a process associated with high-risk tumors. Dr. Lovat likened the process to that of “mortar and bricks holding together a wall”, with the loss of these proteins being “like the mortar crumbling away in the wall.”
According to Cancer Research UK, there are over 16,000 new cases of melanoma skin cancer each year in the United Kingdom, with over 2,000 deaths annually. After being surgically removed, primary tumors are histologically staged, with even low-risk cases being followed up for a number of years, a process that can be time-consuming for patients and costly for the NHS.
Some reassurance for those with melanoma
The creators of the new test say that it is these low-risk patients that the test is able to identify, offering a degree of reassurance to those diagnosed with the disease, and potentially reducing the number of hospital clinic visits they require.
Dr. Lovat commented: “Our test offers a personalized prognosis as it more accurately predicts if your skin cancer is unlikely to spread.”
She added that the test will aid clinicians to identify genuinely low-risk patients diagnosed with an early-stage melanoma, reducing the number of follow-up appointments for those identified as low risk. It, therefore, offers the opportunity to save the NHS time and money.
Excellent news for those with skin cancer
Phil Brady, chief operating officer of the British Skin Foundation, echoed Dr. Lovat’s comments, saying: “The test can alleviate stress and anxiety for patients caused by this potentially deadly skin cancer, whilst increasing efficiency and reducing costs to the NHS.”
Nick Levell, MD, consultant dermatologist & British Skin Foundation spokesperson, who has not been involved in the research, commented how the arrival of the test was “excellent news,” adding that “people at low risk can be reassured and will not have to attend hospital so often for check-ups”.
The development of the new test AMBLor has been led by Dr. Lovat, in association with the university spin-out company AMLo Biosciences, and is accredited by the National Accreditation Body for the United Kingdom. The test involves tissue sections from the standard biopsy being sent in the post to the lab for analysis and costs £293 plus VAT. Currently available through a private referral service, the Newcastle team have applied for the test to be made available on the NHS.
A version of this article first appeared on Medscape UK.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
U.S. cancer deaths continue to fall, especially lung cancer
There has been an overall decline of 32% in cancer deaths as of 2019, or approximately 3.5 million cancer deaths averted, the report noted.
“This success is largely because of reductions in smoking that resulted in downstream declines in lung and other smoking-related cancers,” lead author Rebecca L. Siegel of the ACS, and colleagues, noted in the latest edition of the society’s annual report on cancer rates and trends.
The paper was published online Jan. 12 in CA: A Cancer Journal for Clinicians.
In particular, there has been a fall in both the incidence of and mortality from lung cancer, largely due to successful efforts to get people to quit smoking, but also from earlier diagnosis at a stage when the disease is far more amenable to treatment, noted the authors.
For example, the incidence of lung cancer declined by almost 3% per year in men between the years 2009 and 2018 and by 1% a year in women. Currently, the historically large gender gap in lung cancer incidence is disappearing such that in 2018, lung cancer rates were 24% higher in men than they were in women, and rates in women were actually higher in some younger age groups than they were in men.
Moreover, 28% of lung cancers detected in 2018 were found at a localized stage of disease compared with 17% in 2004.
Patients diagnosed with lung cancer are also living longer, with almost one-third of lung cancer patients still alive 3 years after their diagnosis compared with 21% a decade ago.
However, lung cancer is still the biggest contributor to cancer-related mortality overall, at a death toll of 350 per day – more than breast, prostate, and pancreatic cancer combined, the authors wrote.
This is 2.5 times higher than the death rate from colorectal cancer (CRC), the second leading cause of cancer death in the United States, they added.
Nevertheless, the decrease in lung cancer mortality accelerated from 3.1% per year between 2010 and 2014 to 5.4% per year during 2015 to 2019 in men and from 1.8% to 4.3% in women. “Overall, the lung cancer death rate has dropped by 56% from 1990 to 2019 in men and by 32% from 2002 to 2019 in women,” Ms. Siegel and colleagues emphasized.
Overall, the ACS projects there will be over 1.9 million new cancer cases and over 600,000 cancer deaths across the United States in 2022.
Patterns are changing
With prostate cancer now accounting for some 27% of all cancer diagnoses in men, recent trends in the incidence of prostate cancer are somewhat worrisome, the authors wrote. While the incidence for local-stage disease remained stable from 2014 through to 2018, the incidence of advanced-stage disease has increased by 6% a year since 2011. “Consequently, the proportion of distant-stage diagnoses has more than doubled,” the authors noted, “from a low of 3.9% in 2007 to 8.2% in 2018.”
The incidence of breast cancer among women has been slowly increasing by 0.5% per year since about the mid-2000s. This increase is due at least in part to declines in fertility and increases in body weight among women, the authors suggested. Declines in breast cancer mortality have slowed in recent years, dropping from 1% per year from 2013 to 2019 from 2%-3% per year seen during the 1990s and the early 2000s.
As for CRC, incidence patterns are similar by sex but differ by age. For example, incidence rates of CRC declined by about 2% per year between 2014 and 2018 in individuals 50 years and older, but they increased by 1.5% per year in adults under the age of 50. Overall, however, mortality from CRC decreased by about 2% per year between 2010 and 2019, although this trend again masks increasing mortality from CRC among younger adults, where death rates rose by 1.2% per year from 2005 through 2019 in patients under the age of 50.
The third leading cause of death in men and women combined is pancreatic cancer. Here again, mortality rates slowly increased in men between 2000 and 2013 but have remained relatively stable in women.
Between 2010 and 2019, cancers of the tongue, tonsils, and oropharynx caused by human papilloma virus (HPV) increased by about 2% per year in men and by 1% per year in women.
Death from cervical cancer – despite its being one of the most preventable cancers overall – is still the second leading cause of cancer death in women between 20 and 39 years of age. “Most of these women have never been screened so this is low-hanging fruit easily addressed by increasing access to screening and [HPV] vaccination among underserved women,” Ms. Siegel said in a statement.
On the other hand, mortality from liver cancer – having increased rapidly over the past number of decades – appears to have stabilized in more recent years.
Survival at 5 years
For all cancers combined, survival at 5 years between the mid-1970s and 2011 through 2017 increased from 50% to 68% for White patients and by 39% to 63% for Black patients. “For all stages combined, survival is highest for prostate cancer (98%), melanoma of the skin (93%) and female breast cancer (90%),” the authors pointed out.
In contrast, survival at 5 years is lowest, at 11% for pancreatic cancer, 20% for cancers of the liver and esophagus, and 22% for lung cancer.
Indeed, for most of the common cancers, cancer survival has improved since the mid-1970s with the exception or uterine and cervical cancer, the latter because there have been few advancements in treatment.
Even among the more rare blood and lymphoid malignancies, improvements in treatment strategies, including the use of targeted therapies, have resulted in major survival gains from around 20% in the mid-1970s for chronic myeloid leukemia (CML) patients to over 70% for CML patients diagnosed between 2011 and 2017.
Similarly, the discovery and use of immunotherapy has doubled 5-year survival rates to 30% for patients with metastatic melanoma from 15% in 2004. On the other hand, racial disparities in survival odds continue to persist. For every cancer type except for cancer of the pancreas and kidney, survival rates were lower for Black patients than for White patients, the researchers pointed out.
“Black individuals also have lower stage-specific survival for most cancer types,” the report authors noted. Indeed, after adjustment for sex, age, and stage at diagnosis, the risk of death is 33% higher in Black patients than White patients and 51% higher in American Indian/Alaska Natives compared to White patients.
That said, the overall incidence of cancer is still highest among White individuals, in part because of high rates of breast cancer in White women, which may in part reflect overdiagnosis of breast cancer in this patient population, as the authors suggested.
“However, Black women have the highest cancer mortality rates – 12% higher than White women,” they observed. Even more striking, Black women have a 4% lower incidence of breast cancer than White women but a 41% higher mortality risk from it.
As for pediatric and adolescent cancers, incidence rates may be increasing slightly among both age groups, but dramatic reductions in death by 71% among children and by 61% among adolescents from the mid-70s until now continue as a singular success story in the treatment of cancer overall.
All the authors are employed by the ACS.
A version of this article first appeared on Medscape.com.
There has been an overall decline of 32% in cancer deaths as of 2019, or approximately 3.5 million cancer deaths averted, the report noted.
“This success is largely because of reductions in smoking that resulted in downstream declines in lung and other smoking-related cancers,” lead author Rebecca L. Siegel of the ACS, and colleagues, noted in the latest edition of the society’s annual report on cancer rates and trends.
The paper was published online Jan. 12 in CA: A Cancer Journal for Clinicians.
In particular, there has been a fall in both the incidence of and mortality from lung cancer, largely due to successful efforts to get people to quit smoking, but also from earlier diagnosis at a stage when the disease is far more amenable to treatment, noted the authors.
For example, the incidence of lung cancer declined by almost 3% per year in men between the years 2009 and 2018 and by 1% a year in women. Currently, the historically large gender gap in lung cancer incidence is disappearing such that in 2018, lung cancer rates were 24% higher in men than they were in women, and rates in women were actually higher in some younger age groups than they were in men.
Moreover, 28% of lung cancers detected in 2018 were found at a localized stage of disease compared with 17% in 2004.
Patients diagnosed with lung cancer are also living longer, with almost one-third of lung cancer patients still alive 3 years after their diagnosis compared with 21% a decade ago.
However, lung cancer is still the biggest contributor to cancer-related mortality overall, at a death toll of 350 per day – more than breast, prostate, and pancreatic cancer combined, the authors wrote.
This is 2.5 times higher than the death rate from colorectal cancer (CRC), the second leading cause of cancer death in the United States, they added.
Nevertheless, the decrease in lung cancer mortality accelerated from 3.1% per year between 2010 and 2014 to 5.4% per year during 2015 to 2019 in men and from 1.8% to 4.3% in women. “Overall, the lung cancer death rate has dropped by 56% from 1990 to 2019 in men and by 32% from 2002 to 2019 in women,” Ms. Siegel and colleagues emphasized.
Overall, the ACS projects there will be over 1.9 million new cancer cases and over 600,000 cancer deaths across the United States in 2022.
Patterns are changing
With prostate cancer now accounting for some 27% of all cancer diagnoses in men, recent trends in the incidence of prostate cancer are somewhat worrisome, the authors wrote. While the incidence for local-stage disease remained stable from 2014 through to 2018, the incidence of advanced-stage disease has increased by 6% a year since 2011. “Consequently, the proportion of distant-stage diagnoses has more than doubled,” the authors noted, “from a low of 3.9% in 2007 to 8.2% in 2018.”
The incidence of breast cancer among women has been slowly increasing by 0.5% per year since about the mid-2000s. This increase is due at least in part to declines in fertility and increases in body weight among women, the authors suggested. Declines in breast cancer mortality have slowed in recent years, dropping from 1% per year from 2013 to 2019 from 2%-3% per year seen during the 1990s and the early 2000s.
As for CRC, incidence patterns are similar by sex but differ by age. For example, incidence rates of CRC declined by about 2% per year between 2014 and 2018 in individuals 50 years and older, but they increased by 1.5% per year in adults under the age of 50. Overall, however, mortality from CRC decreased by about 2% per year between 2010 and 2019, although this trend again masks increasing mortality from CRC among younger adults, where death rates rose by 1.2% per year from 2005 through 2019 in patients under the age of 50.
The third leading cause of death in men and women combined is pancreatic cancer. Here again, mortality rates slowly increased in men between 2000 and 2013 but have remained relatively stable in women.
Between 2010 and 2019, cancers of the tongue, tonsils, and oropharynx caused by human papilloma virus (HPV) increased by about 2% per year in men and by 1% per year in women.
Death from cervical cancer – despite its being one of the most preventable cancers overall – is still the second leading cause of cancer death in women between 20 and 39 years of age. “Most of these women have never been screened so this is low-hanging fruit easily addressed by increasing access to screening and [HPV] vaccination among underserved women,” Ms. Siegel said in a statement.
On the other hand, mortality from liver cancer – having increased rapidly over the past number of decades – appears to have stabilized in more recent years.
Survival at 5 years
For all cancers combined, survival at 5 years between the mid-1970s and 2011 through 2017 increased from 50% to 68% for White patients and by 39% to 63% for Black patients. “For all stages combined, survival is highest for prostate cancer (98%), melanoma of the skin (93%) and female breast cancer (90%),” the authors pointed out.
In contrast, survival at 5 years is lowest, at 11% for pancreatic cancer, 20% for cancers of the liver and esophagus, and 22% for lung cancer.
Indeed, for most of the common cancers, cancer survival has improved since the mid-1970s with the exception or uterine and cervical cancer, the latter because there have been few advancements in treatment.
Even among the more rare blood and lymphoid malignancies, improvements in treatment strategies, including the use of targeted therapies, have resulted in major survival gains from around 20% in the mid-1970s for chronic myeloid leukemia (CML) patients to over 70% for CML patients diagnosed between 2011 and 2017.
Similarly, the discovery and use of immunotherapy has doubled 5-year survival rates to 30% for patients with metastatic melanoma from 15% in 2004. On the other hand, racial disparities in survival odds continue to persist. For every cancer type except for cancer of the pancreas and kidney, survival rates were lower for Black patients than for White patients, the researchers pointed out.
“Black individuals also have lower stage-specific survival for most cancer types,” the report authors noted. Indeed, after adjustment for sex, age, and stage at diagnosis, the risk of death is 33% higher in Black patients than White patients and 51% higher in American Indian/Alaska Natives compared to White patients.
That said, the overall incidence of cancer is still highest among White individuals, in part because of high rates of breast cancer in White women, which may in part reflect overdiagnosis of breast cancer in this patient population, as the authors suggested.
“However, Black women have the highest cancer mortality rates – 12% higher than White women,” they observed. Even more striking, Black women have a 4% lower incidence of breast cancer than White women but a 41% higher mortality risk from it.
As for pediatric and adolescent cancers, incidence rates may be increasing slightly among both age groups, but dramatic reductions in death by 71% among children and by 61% among adolescents from the mid-70s until now continue as a singular success story in the treatment of cancer overall.
All the authors are employed by the ACS.
A version of this article first appeared on Medscape.com.
There has been an overall decline of 32% in cancer deaths as of 2019, or approximately 3.5 million cancer deaths averted, the report noted.
“This success is largely because of reductions in smoking that resulted in downstream declines in lung and other smoking-related cancers,” lead author Rebecca L. Siegel of the ACS, and colleagues, noted in the latest edition of the society’s annual report on cancer rates and trends.
The paper was published online Jan. 12 in CA: A Cancer Journal for Clinicians.
In particular, there has been a fall in both the incidence of and mortality from lung cancer, largely due to successful efforts to get people to quit smoking, but also from earlier diagnosis at a stage when the disease is far more amenable to treatment, noted the authors.
For example, the incidence of lung cancer declined by almost 3% per year in men between the years 2009 and 2018 and by 1% a year in women. Currently, the historically large gender gap in lung cancer incidence is disappearing such that in 2018, lung cancer rates were 24% higher in men than they were in women, and rates in women were actually higher in some younger age groups than they were in men.
Moreover, 28% of lung cancers detected in 2018 were found at a localized stage of disease compared with 17% in 2004.
Patients diagnosed with lung cancer are also living longer, with almost one-third of lung cancer patients still alive 3 years after their diagnosis compared with 21% a decade ago.
However, lung cancer is still the biggest contributor to cancer-related mortality overall, at a death toll of 350 per day – more than breast, prostate, and pancreatic cancer combined, the authors wrote.
This is 2.5 times higher than the death rate from colorectal cancer (CRC), the second leading cause of cancer death in the United States, they added.
Nevertheless, the decrease in lung cancer mortality accelerated from 3.1% per year between 2010 and 2014 to 5.4% per year during 2015 to 2019 in men and from 1.8% to 4.3% in women. “Overall, the lung cancer death rate has dropped by 56% from 1990 to 2019 in men and by 32% from 2002 to 2019 in women,” Ms. Siegel and colleagues emphasized.
Overall, the ACS projects there will be over 1.9 million new cancer cases and over 600,000 cancer deaths across the United States in 2022.
Patterns are changing
With prostate cancer now accounting for some 27% of all cancer diagnoses in men, recent trends in the incidence of prostate cancer are somewhat worrisome, the authors wrote. While the incidence for local-stage disease remained stable from 2014 through to 2018, the incidence of advanced-stage disease has increased by 6% a year since 2011. “Consequently, the proportion of distant-stage diagnoses has more than doubled,” the authors noted, “from a low of 3.9% in 2007 to 8.2% in 2018.”
The incidence of breast cancer among women has been slowly increasing by 0.5% per year since about the mid-2000s. This increase is due at least in part to declines in fertility and increases in body weight among women, the authors suggested. Declines in breast cancer mortality have slowed in recent years, dropping from 1% per year from 2013 to 2019 from 2%-3% per year seen during the 1990s and the early 2000s.
As for CRC, incidence patterns are similar by sex but differ by age. For example, incidence rates of CRC declined by about 2% per year between 2014 and 2018 in individuals 50 years and older, but they increased by 1.5% per year in adults under the age of 50. Overall, however, mortality from CRC decreased by about 2% per year between 2010 and 2019, although this trend again masks increasing mortality from CRC among younger adults, where death rates rose by 1.2% per year from 2005 through 2019 in patients under the age of 50.
The third leading cause of death in men and women combined is pancreatic cancer. Here again, mortality rates slowly increased in men between 2000 and 2013 but have remained relatively stable in women.
Between 2010 and 2019, cancers of the tongue, tonsils, and oropharynx caused by human papilloma virus (HPV) increased by about 2% per year in men and by 1% per year in women.
Death from cervical cancer – despite its being one of the most preventable cancers overall – is still the second leading cause of cancer death in women between 20 and 39 years of age. “Most of these women have never been screened so this is low-hanging fruit easily addressed by increasing access to screening and [HPV] vaccination among underserved women,” Ms. Siegel said in a statement.
On the other hand, mortality from liver cancer – having increased rapidly over the past number of decades – appears to have stabilized in more recent years.
Survival at 5 years
For all cancers combined, survival at 5 years between the mid-1970s and 2011 through 2017 increased from 50% to 68% for White patients and by 39% to 63% for Black patients. “For all stages combined, survival is highest for prostate cancer (98%), melanoma of the skin (93%) and female breast cancer (90%),” the authors pointed out.
In contrast, survival at 5 years is lowest, at 11% for pancreatic cancer, 20% for cancers of the liver and esophagus, and 22% for lung cancer.
Indeed, for most of the common cancers, cancer survival has improved since the mid-1970s with the exception or uterine and cervical cancer, the latter because there have been few advancements in treatment.
Even among the more rare blood and lymphoid malignancies, improvements in treatment strategies, including the use of targeted therapies, have resulted in major survival gains from around 20% in the mid-1970s for chronic myeloid leukemia (CML) patients to over 70% for CML patients diagnosed between 2011 and 2017.
Similarly, the discovery and use of immunotherapy has doubled 5-year survival rates to 30% for patients with metastatic melanoma from 15% in 2004. On the other hand, racial disparities in survival odds continue to persist. For every cancer type except for cancer of the pancreas and kidney, survival rates were lower for Black patients than for White patients, the researchers pointed out.
“Black individuals also have lower stage-specific survival for most cancer types,” the report authors noted. Indeed, after adjustment for sex, age, and stage at diagnosis, the risk of death is 33% higher in Black patients than White patients and 51% higher in American Indian/Alaska Natives compared to White patients.
That said, the overall incidence of cancer is still highest among White individuals, in part because of high rates of breast cancer in White women, which may in part reflect overdiagnosis of breast cancer in this patient population, as the authors suggested.
“However, Black women have the highest cancer mortality rates – 12% higher than White women,” they observed. Even more striking, Black women have a 4% lower incidence of breast cancer than White women but a 41% higher mortality risk from it.
As for pediatric and adolescent cancers, incidence rates may be increasing slightly among both age groups, but dramatic reductions in death by 71% among children and by 61% among adolescents from the mid-70s until now continue as a singular success story in the treatment of cancer overall.
All the authors are employed by the ACS.
A version of this article first appeared on Medscape.com.
FROM CA: A CANCER JOURNAL FOR CLINICIANS
Soon-to-be medical student awarded $10K after spotting melanoma
A soon-to-be medical student and former oncology ward volunteer has received a $10,000 scholarship for her education recently after tipping off a Vancouver Canucks staff member about a cancerous mole on the back of his neck during a National Hockey League game in Seattle this past October.
Sitting immediately behind the visiting team’s bench, Nadia Popovici wrote a large-font message on her cell phone and tapped the protective glass to get the attention of Brian Hamilton, assistant equipment manager for the Canucks.
“The mole on the back of your neck is possibly cancerous. Please go see a doctor!” read the message.
Mr. Hamilton acted on the tip and was eventually diagnosed with a malignant stage II melanoma, according to a report in the Seattle Times.
As noted in a Medscape Q&A, “ABCDE” is the acronym that indicates the visible, physical characteristics suggestive of melanoma. ABCDE stands for asymmetry, irregular border, color variations (especially red, white, and blue tones in a brown or black lesion), diameter greater than 6 mm, and elevated surface. The lesions may itch, bleed, ulcerate, or develop satellites.
The Canucks returned to Seattle recently for another game against the Seattle Kraken, and the visiting team posted a note on social media from Mr. Hamilton seeking the identity of the good Samaritan.
“... the message you showed me on your cell phone will forever be etched into my brain and has made a true life-changing difference for me and my family,” wrote Mr. Hamilton.
Within hours, Ms. Popovici’s mother, whose family has season tickets to the Seattle team’s games, responded to the message.
Ms. Popovici and Mr. Hamilton met up again at the Jan. 1 game, where Ms. Popovici was rewarded with a $10,000 medical school scholarship in a surprise announcement, shared on Twitter and liked more than 42,000 times.
“She didn’t take me out of a burning car like the big stories, but she took me out of a slow fire. And the words out of the doctor’s mouth were, if I ignored that for 4-5 years, I wouldn’t be here,” Mr. Hamilton said at a news conference on Jan. 1.
Ms. Popovici says she has been accepted to several medical schools and will start school in the fall, according to a press release from the National Hockey League.
More money for medical school may be on the way for Ms. Popovici after a GoFundMe page was started. With a goal of $25,000, the fund had received just over $2,500 as of Jan. 4.
“The teams made a kind gesture of giving her 10K, but I think we can do better!” Josh Doxey, a sales manager from Lehi, Utah, wrote on the page he created for Ms. Popovici.
Mr. Doxey told this news organization, “I started the GoFundMe thinking it would be a nice gesture especially for someone going into health care after 2 crazy years of COVID ... I have gotten in touch with her and her mother, and have been chatting with both. They both seem incredibly kind, grateful, and humble.”
A version of this article first appeared on Medscape.com.
A soon-to-be medical student and former oncology ward volunteer has received a $10,000 scholarship for her education recently after tipping off a Vancouver Canucks staff member about a cancerous mole on the back of his neck during a National Hockey League game in Seattle this past October.
Sitting immediately behind the visiting team’s bench, Nadia Popovici wrote a large-font message on her cell phone and tapped the protective glass to get the attention of Brian Hamilton, assistant equipment manager for the Canucks.
“The mole on the back of your neck is possibly cancerous. Please go see a doctor!” read the message.
Mr. Hamilton acted on the tip and was eventually diagnosed with a malignant stage II melanoma, according to a report in the Seattle Times.
As noted in a Medscape Q&A, “ABCDE” is the acronym that indicates the visible, physical characteristics suggestive of melanoma. ABCDE stands for asymmetry, irregular border, color variations (especially red, white, and blue tones in a brown or black lesion), diameter greater than 6 mm, and elevated surface. The lesions may itch, bleed, ulcerate, or develop satellites.
The Canucks returned to Seattle recently for another game against the Seattle Kraken, and the visiting team posted a note on social media from Mr. Hamilton seeking the identity of the good Samaritan.
“... the message you showed me on your cell phone will forever be etched into my brain and has made a true life-changing difference for me and my family,” wrote Mr. Hamilton.
Within hours, Ms. Popovici’s mother, whose family has season tickets to the Seattle team’s games, responded to the message.
Ms. Popovici and Mr. Hamilton met up again at the Jan. 1 game, where Ms. Popovici was rewarded with a $10,000 medical school scholarship in a surprise announcement, shared on Twitter and liked more than 42,000 times.
“She didn’t take me out of a burning car like the big stories, but she took me out of a slow fire. And the words out of the doctor’s mouth were, if I ignored that for 4-5 years, I wouldn’t be here,” Mr. Hamilton said at a news conference on Jan. 1.
Ms. Popovici says she has been accepted to several medical schools and will start school in the fall, according to a press release from the National Hockey League.
More money for medical school may be on the way for Ms. Popovici after a GoFundMe page was started. With a goal of $25,000, the fund had received just over $2,500 as of Jan. 4.
“The teams made a kind gesture of giving her 10K, but I think we can do better!” Josh Doxey, a sales manager from Lehi, Utah, wrote on the page he created for Ms. Popovici.
Mr. Doxey told this news organization, “I started the GoFundMe thinking it would be a nice gesture especially for someone going into health care after 2 crazy years of COVID ... I have gotten in touch with her and her mother, and have been chatting with both. They both seem incredibly kind, grateful, and humble.”
A version of this article first appeared on Medscape.com.
A soon-to-be medical student and former oncology ward volunteer has received a $10,000 scholarship for her education recently after tipping off a Vancouver Canucks staff member about a cancerous mole on the back of his neck during a National Hockey League game in Seattle this past October.
Sitting immediately behind the visiting team’s bench, Nadia Popovici wrote a large-font message on her cell phone and tapped the protective glass to get the attention of Brian Hamilton, assistant equipment manager for the Canucks.
“The mole on the back of your neck is possibly cancerous. Please go see a doctor!” read the message.
Mr. Hamilton acted on the tip and was eventually diagnosed with a malignant stage II melanoma, according to a report in the Seattle Times.
As noted in a Medscape Q&A, “ABCDE” is the acronym that indicates the visible, physical characteristics suggestive of melanoma. ABCDE stands for asymmetry, irregular border, color variations (especially red, white, and blue tones in a brown or black lesion), diameter greater than 6 mm, and elevated surface. The lesions may itch, bleed, ulcerate, or develop satellites.
The Canucks returned to Seattle recently for another game against the Seattle Kraken, and the visiting team posted a note on social media from Mr. Hamilton seeking the identity of the good Samaritan.
“... the message you showed me on your cell phone will forever be etched into my brain and has made a true life-changing difference for me and my family,” wrote Mr. Hamilton.
Within hours, Ms. Popovici’s mother, whose family has season tickets to the Seattle team’s games, responded to the message.
Ms. Popovici and Mr. Hamilton met up again at the Jan. 1 game, where Ms. Popovici was rewarded with a $10,000 medical school scholarship in a surprise announcement, shared on Twitter and liked more than 42,000 times.
“She didn’t take me out of a burning car like the big stories, but she took me out of a slow fire. And the words out of the doctor’s mouth were, if I ignored that for 4-5 years, I wouldn’t be here,” Mr. Hamilton said at a news conference on Jan. 1.
Ms. Popovici says she has been accepted to several medical schools and will start school in the fall, according to a press release from the National Hockey League.
More money for medical school may be on the way for Ms. Popovici after a GoFundMe page was started. With a goal of $25,000, the fund had received just over $2,500 as of Jan. 4.
“The teams made a kind gesture of giving her 10K, but I think we can do better!” Josh Doxey, a sales manager from Lehi, Utah, wrote on the page he created for Ms. Popovici.
Mr. Doxey told this news organization, “I started the GoFundMe thinking it would be a nice gesture especially for someone going into health care after 2 crazy years of COVID ... I have gotten in touch with her and her mother, and have been chatting with both. They both seem incredibly kind, grateful, and humble.”
A version of this article first appeared on Medscape.com.
High-fiber diet may improve melanoma immunotherapy response, outcomes
a new study shows.
Investigators found that the patients who reported consuming at least 20 g of dietary fiber daily had significantly better progression-free survival (PFS) than those who reported consuming lower amounts of dietary fiber. However, patients who took a probiotic supplement in the past month had slightly shorter PFS, but the results were not statistically significant.
And after adjusting for clinical factors, each 5-g increase in daily dietary fiber intake corresponded to a 30% lower risk of disease progression, according to the analysis, published online Dec. 23, 2021, in Science.
“Our study sheds light on the potential effects of a patient’s diet and supplement use when starting treatment with immune checkpoint blockade,” co–lead study author Jennifer Wargo, MD, professor of genomic medicine and surgical oncology at University of Texas MD Anderson Cancer Center, Houston, said in a press release. “These results provide further support for clinical trials to modulate the microbiome with the goal of improving cancer outcomes using dietary and other strategies.”
Previous research has suggested that the microbiome can influence patients’ response to immunotherapy. One recent analysis, for instance, found that fecal microbiota transplant can improve response to immunotherapy in advanced melanoma. And a small 2019 analysis from Dr. Dr. Wargo and colleagues hinted that a high-fiber diet may enhance patients’ ability to respond to immunotherapy in advanced melanoma, while probiotics appear to dampen that response.
Still, the role diet and probiotic supplements play in treatment response remains poorly understood.
In the current study, Dr. Wargo and colleagues assessed fecal microbiota profiles and dietary habits, including fiber intake and probiotic use, in 158 patients with advanced melanoma who received immune checkpoint blockade inhibitors.
In the cohort, 31% (49 of 158) of late-stage melanoma patients reported taking a commercially available probiotic in the past month. When assessing whether probiotic use influenced patient outcomes, the investigators observed a shorter but not statistically significant difference in PFS in those who took a probiotic (median, 17 months) versus those who did not (23 months).
Higher dietary fiber, however, was associated with significantly improved PFS in a subset of 128 patients. The team divided patients into a higher-fiber intake group (those consuming at least 20 g/day) and a low-fiber group (those consuming less than 20 g).
The 37 patients reporting higher fiber intake demonstrated improved PFS, compared with those in the low-intake group (median PFS not reached vs. 13 months), plus a 30% lower risk of disease progression or death for each additional 5 g consumed each day.
“The observed protective effect of dietary fiber intake in relation to PFS and response remained consistent among the subset of patients treated with anti–PD-1 monotherapy, with the exclusion of patients reporting recent antibiotic use,” the authors noted.
When assessing fiber and probiotic intake together, the researchers found that immunotherapy response rate was higher (82%) in the 22 patients who reported sufficient dietary fiber intake with no probiotic use versus 59% in 101 patients who reported either insufficient fiber intake or probiotic use.
Overall, the research suggests that “consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy,” co–lead author Giorgio Trinchieri, MD, chief of the Laboratory of Integrative Cancer Immunology in the National Cancer Institute’s Center for Cancer Research, Bethesda, Md., said in a press statement. “The data also suggest that it’s probably better for people with cancer receiving immunotherapy not to use commercially available probiotics.”
The investigators also explored whether dietary fiber intake enhanced treatment response in preclinical mouse models of melanoma. In this instance, mice receiving a fiber-rich diet showed delayed tumor growth after anti–PD-1 treatment, compared with mice given a low-fiber diet or probiotics.
According to the authors, “our preclinical models support the hypothesis that dietary fiber and probiotics modulate the microbiome and that antitumor immunity is impaired in mice receiving a low-fiber diet and in those receiving probiotics – with suppression of intratumoral [interferon-gamma] T-cell responses in both cases.”
Dietary fiber may exert beneficial effect by increasing specific types of bacteria in the gut, such as Ruminococcaceae, which “produce high levels of certain short-chain fatty acids that have an antitumor effect,” Dr. Trinchieri explained.
However, “the impact of dietary fiber and probiotics on the gut microbiota is only part of the bigger picture,” Dr. Trinchieri said in a press release. “Many factors can affect the ability of a patient with melanoma to respond to immunotherapy” but, according to this analysis, “the microbiota seems to be one of the dominant factors.”
While Jeffrey S. Weber, MD, PhD, applauded the “innovative and interesting” research, he believes the patient population is too small to confirm that a high-fiber diet does indeed contribute to improved immunotherapy response and PFS in patients with advanced melanoma.
Additional data are needed to clarify these findings. “I will believe it if I could see it replicated in a larger study,” Dr. Weber, professor and deputy director of the Laura and Isaac Perlmutter Cancer Center, New York University, said in an interview.
Dr. Wargo noted that a randomized clinical trial exploring how diets with varying fiber content affect the microbiome and immune response is currently enrolling patients with stage III and IV melanoma receiving immunotherapy.
This study was supported by the Melanoma Moon Shot, among others. Dr. Wargo is a collaborator on a U.S. patent application that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome. Dr. Weber reported relationships with Bristol-Myers Squibb, GlaxoSmithKline, Genentech BioOncology, Merck, Novartis, EMD Serono, Celldex, CytomX, Nektar, Roche, Altor, Daiichi Sankyo, and Eli Lilly, and is named on patents filed for biomarkers for ipilimumab and nivolumab.
A version of this article first appeared on Medscape.com.
a new study shows.
Investigators found that the patients who reported consuming at least 20 g of dietary fiber daily had significantly better progression-free survival (PFS) than those who reported consuming lower amounts of dietary fiber. However, patients who took a probiotic supplement in the past month had slightly shorter PFS, but the results were not statistically significant.
And after adjusting for clinical factors, each 5-g increase in daily dietary fiber intake corresponded to a 30% lower risk of disease progression, according to the analysis, published online Dec. 23, 2021, in Science.
“Our study sheds light on the potential effects of a patient’s diet and supplement use when starting treatment with immune checkpoint blockade,” co–lead study author Jennifer Wargo, MD, professor of genomic medicine and surgical oncology at University of Texas MD Anderson Cancer Center, Houston, said in a press release. “These results provide further support for clinical trials to modulate the microbiome with the goal of improving cancer outcomes using dietary and other strategies.”
Previous research has suggested that the microbiome can influence patients’ response to immunotherapy. One recent analysis, for instance, found that fecal microbiota transplant can improve response to immunotherapy in advanced melanoma. And a small 2019 analysis from Dr. Dr. Wargo and colleagues hinted that a high-fiber diet may enhance patients’ ability to respond to immunotherapy in advanced melanoma, while probiotics appear to dampen that response.
Still, the role diet and probiotic supplements play in treatment response remains poorly understood.
In the current study, Dr. Wargo and colleagues assessed fecal microbiota profiles and dietary habits, including fiber intake and probiotic use, in 158 patients with advanced melanoma who received immune checkpoint blockade inhibitors.
In the cohort, 31% (49 of 158) of late-stage melanoma patients reported taking a commercially available probiotic in the past month. When assessing whether probiotic use influenced patient outcomes, the investigators observed a shorter but not statistically significant difference in PFS in those who took a probiotic (median, 17 months) versus those who did not (23 months).
Higher dietary fiber, however, was associated with significantly improved PFS in a subset of 128 patients. The team divided patients into a higher-fiber intake group (those consuming at least 20 g/day) and a low-fiber group (those consuming less than 20 g).
The 37 patients reporting higher fiber intake demonstrated improved PFS, compared with those in the low-intake group (median PFS not reached vs. 13 months), plus a 30% lower risk of disease progression or death for each additional 5 g consumed each day.
“The observed protective effect of dietary fiber intake in relation to PFS and response remained consistent among the subset of patients treated with anti–PD-1 monotherapy, with the exclusion of patients reporting recent antibiotic use,” the authors noted.
When assessing fiber and probiotic intake together, the researchers found that immunotherapy response rate was higher (82%) in the 22 patients who reported sufficient dietary fiber intake with no probiotic use versus 59% in 101 patients who reported either insufficient fiber intake or probiotic use.
Overall, the research suggests that “consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy,” co–lead author Giorgio Trinchieri, MD, chief of the Laboratory of Integrative Cancer Immunology in the National Cancer Institute’s Center for Cancer Research, Bethesda, Md., said in a press statement. “The data also suggest that it’s probably better for people with cancer receiving immunotherapy not to use commercially available probiotics.”
The investigators also explored whether dietary fiber intake enhanced treatment response in preclinical mouse models of melanoma. In this instance, mice receiving a fiber-rich diet showed delayed tumor growth after anti–PD-1 treatment, compared with mice given a low-fiber diet or probiotics.
According to the authors, “our preclinical models support the hypothesis that dietary fiber and probiotics modulate the microbiome and that antitumor immunity is impaired in mice receiving a low-fiber diet and in those receiving probiotics – with suppression of intratumoral [interferon-gamma] T-cell responses in both cases.”
Dietary fiber may exert beneficial effect by increasing specific types of bacteria in the gut, such as Ruminococcaceae, which “produce high levels of certain short-chain fatty acids that have an antitumor effect,” Dr. Trinchieri explained.
However, “the impact of dietary fiber and probiotics on the gut microbiota is only part of the bigger picture,” Dr. Trinchieri said in a press release. “Many factors can affect the ability of a patient with melanoma to respond to immunotherapy” but, according to this analysis, “the microbiota seems to be one of the dominant factors.”
While Jeffrey S. Weber, MD, PhD, applauded the “innovative and interesting” research, he believes the patient population is too small to confirm that a high-fiber diet does indeed contribute to improved immunotherapy response and PFS in patients with advanced melanoma.
Additional data are needed to clarify these findings. “I will believe it if I could see it replicated in a larger study,” Dr. Weber, professor and deputy director of the Laura and Isaac Perlmutter Cancer Center, New York University, said in an interview.
Dr. Wargo noted that a randomized clinical trial exploring how diets with varying fiber content affect the microbiome and immune response is currently enrolling patients with stage III and IV melanoma receiving immunotherapy.
This study was supported by the Melanoma Moon Shot, among others. Dr. Wargo is a collaborator on a U.S. patent application that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome. Dr. Weber reported relationships with Bristol-Myers Squibb, GlaxoSmithKline, Genentech BioOncology, Merck, Novartis, EMD Serono, Celldex, CytomX, Nektar, Roche, Altor, Daiichi Sankyo, and Eli Lilly, and is named on patents filed for biomarkers for ipilimumab and nivolumab.
A version of this article first appeared on Medscape.com.
a new study shows.
Investigators found that the patients who reported consuming at least 20 g of dietary fiber daily had significantly better progression-free survival (PFS) than those who reported consuming lower amounts of dietary fiber. However, patients who took a probiotic supplement in the past month had slightly shorter PFS, but the results were not statistically significant.
And after adjusting for clinical factors, each 5-g increase in daily dietary fiber intake corresponded to a 30% lower risk of disease progression, according to the analysis, published online Dec. 23, 2021, in Science.
“Our study sheds light on the potential effects of a patient’s diet and supplement use when starting treatment with immune checkpoint blockade,” co–lead study author Jennifer Wargo, MD, professor of genomic medicine and surgical oncology at University of Texas MD Anderson Cancer Center, Houston, said in a press release. “These results provide further support for clinical trials to modulate the microbiome with the goal of improving cancer outcomes using dietary and other strategies.”
Previous research has suggested that the microbiome can influence patients’ response to immunotherapy. One recent analysis, for instance, found that fecal microbiota transplant can improve response to immunotherapy in advanced melanoma. And a small 2019 analysis from Dr. Dr. Wargo and colleagues hinted that a high-fiber diet may enhance patients’ ability to respond to immunotherapy in advanced melanoma, while probiotics appear to dampen that response.
Still, the role diet and probiotic supplements play in treatment response remains poorly understood.
In the current study, Dr. Wargo and colleagues assessed fecal microbiota profiles and dietary habits, including fiber intake and probiotic use, in 158 patients with advanced melanoma who received immune checkpoint blockade inhibitors.
In the cohort, 31% (49 of 158) of late-stage melanoma patients reported taking a commercially available probiotic in the past month. When assessing whether probiotic use influenced patient outcomes, the investigators observed a shorter but not statistically significant difference in PFS in those who took a probiotic (median, 17 months) versus those who did not (23 months).
Higher dietary fiber, however, was associated with significantly improved PFS in a subset of 128 patients. The team divided patients into a higher-fiber intake group (those consuming at least 20 g/day) and a low-fiber group (those consuming less than 20 g).
The 37 patients reporting higher fiber intake demonstrated improved PFS, compared with those in the low-intake group (median PFS not reached vs. 13 months), plus a 30% lower risk of disease progression or death for each additional 5 g consumed each day.
“The observed protective effect of dietary fiber intake in relation to PFS and response remained consistent among the subset of patients treated with anti–PD-1 monotherapy, with the exclusion of patients reporting recent antibiotic use,” the authors noted.
When assessing fiber and probiotic intake together, the researchers found that immunotherapy response rate was higher (82%) in the 22 patients who reported sufficient dietary fiber intake with no probiotic use versus 59% in 101 patients who reported either insufficient fiber intake or probiotic use.
Overall, the research suggests that “consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy,” co–lead author Giorgio Trinchieri, MD, chief of the Laboratory of Integrative Cancer Immunology in the National Cancer Institute’s Center for Cancer Research, Bethesda, Md., said in a press statement. “The data also suggest that it’s probably better for people with cancer receiving immunotherapy not to use commercially available probiotics.”
The investigators also explored whether dietary fiber intake enhanced treatment response in preclinical mouse models of melanoma. In this instance, mice receiving a fiber-rich diet showed delayed tumor growth after anti–PD-1 treatment, compared with mice given a low-fiber diet or probiotics.
According to the authors, “our preclinical models support the hypothesis that dietary fiber and probiotics modulate the microbiome and that antitumor immunity is impaired in mice receiving a low-fiber diet and in those receiving probiotics – with suppression of intratumoral [interferon-gamma] T-cell responses in both cases.”
Dietary fiber may exert beneficial effect by increasing specific types of bacteria in the gut, such as Ruminococcaceae, which “produce high levels of certain short-chain fatty acids that have an antitumor effect,” Dr. Trinchieri explained.
However, “the impact of dietary fiber and probiotics on the gut microbiota is only part of the bigger picture,” Dr. Trinchieri said in a press release. “Many factors can affect the ability of a patient with melanoma to respond to immunotherapy” but, according to this analysis, “the microbiota seems to be one of the dominant factors.”
While Jeffrey S. Weber, MD, PhD, applauded the “innovative and interesting” research, he believes the patient population is too small to confirm that a high-fiber diet does indeed contribute to improved immunotherapy response and PFS in patients with advanced melanoma.
Additional data are needed to clarify these findings. “I will believe it if I could see it replicated in a larger study,” Dr. Weber, professor and deputy director of the Laura and Isaac Perlmutter Cancer Center, New York University, said in an interview.
Dr. Wargo noted that a randomized clinical trial exploring how diets with varying fiber content affect the microbiome and immune response is currently enrolling patients with stage III and IV melanoma receiving immunotherapy.
This study was supported by the Melanoma Moon Shot, among others. Dr. Wargo is a collaborator on a U.S. patent application that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome. Dr. Weber reported relationships with Bristol-Myers Squibb, GlaxoSmithKline, Genentech BioOncology, Merck, Novartis, EMD Serono, Celldex, CytomX, Nektar, Roche, Altor, Daiichi Sankyo, and Eli Lilly, and is named on patents filed for biomarkers for ipilimumab and nivolumab.
A version of this article first appeared on Medscape.com.
FROM NATURE
Skin imaging working group releases first guidelines for AI algorithms used in dermatology
The
The guidelines, published in JAMA Dermatology on Dec. 1, 2021, contain a broad range of recommendations stakeholders should consider when developing and assessing image-based AI algorithms in dermatology. The recommendations are divided into categories of data, technique, technical assessment, and application. ISIC is “an academia and industry partnership designed to facilitate the application of digital skin imaging to help reduce melanoma mortality,” and is organized into different working groups, including the AI working group, according to its website.
“Our goal with these guidelines was to create higher-quality reporting of dataset and algorithm characteristics for dermatology AI,” first author Roxana Daneshjou, MD, PhD, clinical scholar in dermatology, in the department of dermatology at Stanford (Calif.) University, said in an interview. “We hope these guidelines also aid regulatory bodies around the world when they are assessing algorithms to be used in dermatology.”
Recommendations for data
The authors recommended that datasets used by AI algorithms have image descriptions and details on image artifacts. “For photography, these include the type of camera used; whether images were taken under standardized or varying conditions; whether they were taken by professional photographers, laymen, or health care professionals; and image quality,” they wrote. They also recommended that developers include in an image description the type of lighting used and whether the photo contains pen markings, hair, tattoos, injuries, surgical effects, or other “physical perturbations.”
Exchangeable image file format data obtained from the camera, and preprocessing procedures like color normalization and “postprocessing” of images, such as filtering, should also be disclosed. In addition, developers should disclose and justify inclusion of images that have been created by an algorithm within a dataset. Any public images used in the datasets should have references, and privately used images should be made public where possible, the authors said.
The ISIC working group guidelines also provided recommendations for patient-level metadata. Each image should include a patient’s geographical location and medical center they visited as well as their age, sex and gender, ethnicity and/or race, and skin tone. Dr. Daneshjou said this was one area where she and her colleagues found a lack of transparency in AI datasets in algorithms in a recent review. “We found that many AI papers provided sparse details about the images used to train and test their algorithms,” Dr. Daneshjou explained. “For example, only 7 out of 70 papers had any information about the skin tones in the images used for developing and/or testing AI algorithms. Understanding the diversity of images used to train and test algorithms is important because algorithms that are developed on images of predominantly white skin likely won’t work as well on Black and brown skin.”
The guideline authors also asked algorithm developers to describe the limitations of not including patient-level metadata information when it is incomplete or unavailable. In addition, “we ask that algorithm developers comment on potential biases of their algorithms,” Dr. Daneshjou said. “For example, an algorithm based only on telemedicine images may not capture the full range of diseases seen within an in-person clinic.”
When describing their AI algorithm, developers should detail their reasoning for the dataset size and partitions, inclusion and exclusion criteria for images, and use of any external samples for test sets. “Authors should consider any differences between the image characteristics used for algorithm development and those that might be encountered in the real world,” the guidelines stated.
Recommendations for technique
How the images in a dataset are labeled is a unique challenge in developing AI algorithms for dermatology, the authors noted. Developers should use histopathological diagnosis in their labeling, but this can sometimes result in label noise.
“Many of the AI algorithms in dermatology use supervised learning, which requires labeled examples to help the algorithm ‘learn’ features for discriminating between lesions. We found that some papers use consensus labeling – dermatologists providing a label – to label skin cancers; however, the standard for diagnosing skin cancer is using histopathology from a biopsy,” she said. “Dermatologists can biopsy seven to eight suspected melanomas before discovering a true melanoma, so dermatologist labeling of skin cancers is prone to label noise.”
ISIC’s guidelines stated a gold standard of labeling for dermatologic images is one area that still needs future research, but currently, “diagnoses, labels and diagnostic groups used in data repositories as well as public ontologies” such as ICD-11, AnatomyMapper, and SNOMED-CT should be included in dermatologic image datasets.
AI developers should also provide a detailed description of their algorithm, which includes methods, work flows, mathematical formulas as well as the generalizability of the algorithm across more than one dataset.
Recommendations for technical assessment
“Another important recommendation is that algorithm developers should provide a way for algorithms to be publicly evaluable by researchers,” Dr. Daneshjou said. “Many dermatology AI algorithms do not share either their data or their algorithm. Algorithm sharing is important for assessing reproducibility and robustness.”
Google’s recently announced AI-powered dermatology assistant tool, for example, “has made claims about its accuracy and ability to diagnose skin disease at a dermatologist level, but there is no way for researchers to independently test these claims,” she said. Other options like Model Dermatology, developed by Seung Seog Han, MD, PhD, of the Dermatology Clinic in Seoul, South Korea, and colleagues, offer an application programming interface “that allows researchers to test the algorithm,” Dr. Daneshjou said. “This kind of openness is key for assessing algorithm robustness.”
Developers should also note in their algorithm explanations how performance markers and benchmarks would translate to proposed clinical application. “In this context,” the use case – the context in which the AI application is being used – “should be clearly described – who are the intended users and under what clinical scenario are they using the algorithm,” the authors wrote.
Recommendations for application
The guidelines note that use case for the model should also be described by the AI developers. “Our checklist includes delineating use cases for algorithms and describing what use cases may be within the scope of the algorithm versus which use cases are out of scope,” Dr. Daneshjou said. “For example, an algorithm developed to provide decision support to dermatologists, with a human in the loop, may not be accurate enough to release directly to consumers.”
As the goal of AI algorithms in dermatology is eventual implementation for clinicians and patients, the authors asked developers to consider shortcomings and potential harms of the algorithm during implementation. “Ethical considerations and impact on vulnerable populations should also be considered and discussed,” they wrote. An algorithm “suggesting aesthetic medical treatments may have negative effects given the biased nature of beauty standards,” and “an algorithm that diagnoses basal cell carcinomas but lacks any pigmented basal cell carcinomas, which are more often seen in skin of color, will not perform equitably across populations.”
Prior to implementing an AI algorithm, the ISIC working group recommended developers perform prospective clinical trials for validation. Checklists and guidelines like SPIRIT-AI and CONSORT-AI “provide guidance on how to design clinical trials to test AI algorithms,” Dr. Daneshjou said.
After implementation, “I believe we need additional research in how we monitor algorithms after they are deployed clinically, Dr. Daneshjou said. “Currently there are no [Food and Drug Administration]–approved AI algorithms in dermatology; however, there are several applications that have CE mark in Europe, and there are no mechanisms for postmarket surveillance there.
'Timely' recommendations
Commenting on the ISIC working group guidelines, Justin M. Ko, MD, MBA, director and chief of medical dermatology for Stanford Health Care, who was not involved with the work, said that the recommendations are timely and provide “a framework for a ‘common language’ around AI datasets specifically tailored to dermatology.” Dr. Ko, chair of the American Academy of Dermatology’s Ad Hoc Task Force on Augmented Intelligence, noted the work by Dr. Daneshjou and colleagues “is consistent with and builds further details” on the position statement released by the AAD AI task force in 2019.
“As machine-learning capabilities and commercial efforts continue to mature, it becomes increasingly important that we are able to ‘look under the hood,’ and evaluate all the critical factors that influence development of these capabilities,” he said in an interview. “A standard set of reporting guidelines not only allows for transparency in evaluating data and performance of models and algorithms, but also forces the consideration of issues of equity, fairness, mitigation of bias, and clinically meaningful outcomes.”
One concern is the impact of AI algorithms on societal or health systems, he noted, which is brought up in the guidelines. “The last thing we would want is the development of robust AI systems that exacerbate access challenges, or generate patient anxiety/worry, or drive low-value utilization, or adds to care team burden, or create a technological barrier to care, or increases inequity in dermatologic care,” he said.
In developing AI algorithms for dermatology, a “major practical issue” is how performance on paper will translate to real-world use, Dr. Ko explained, and the ISIC guidelines “provide a critical step in empowering clinicians, practices, and our field to shape the advent of the AI and augmented intelligence tools and systems to promote and enhance meaningful clinical outcomes, and augment the core patient-clinician relationship and ensure they are grounded in principles of fairness, equity and transparency.”
This research was funded by awards and grants to individual authors from the Charina Fund, a Google Research Award, Melanoma Research Alliance, National Health and Medical Research Council, National Institutes of Health/National Cancer Institute, National Science Foundation, and the Department of Veterans Affairs. The authors disclosed relationships with governmental entities, pharmaceutical companies, technology startups, medical publishers, charitable trusts, consulting firms, dermatology training companies, providers of medical devices, manufacturers of dermatologic products, and other organizations related to the paper in the form of supplied equipment, having founded a company; receiving grants, patents, or personal fees; holding shares; and medical reporting. Dr. Ko reported that he serves as a clinical advisor for Skin Analytics, and has an ongoing research collaboration with Google.
The
The guidelines, published in JAMA Dermatology on Dec. 1, 2021, contain a broad range of recommendations stakeholders should consider when developing and assessing image-based AI algorithms in dermatology. The recommendations are divided into categories of data, technique, technical assessment, and application. ISIC is “an academia and industry partnership designed to facilitate the application of digital skin imaging to help reduce melanoma mortality,” and is organized into different working groups, including the AI working group, according to its website.
“Our goal with these guidelines was to create higher-quality reporting of dataset and algorithm characteristics for dermatology AI,” first author Roxana Daneshjou, MD, PhD, clinical scholar in dermatology, in the department of dermatology at Stanford (Calif.) University, said in an interview. “We hope these guidelines also aid regulatory bodies around the world when they are assessing algorithms to be used in dermatology.”
Recommendations for data
The authors recommended that datasets used by AI algorithms have image descriptions and details on image artifacts. “For photography, these include the type of camera used; whether images were taken under standardized or varying conditions; whether they were taken by professional photographers, laymen, or health care professionals; and image quality,” they wrote. They also recommended that developers include in an image description the type of lighting used and whether the photo contains pen markings, hair, tattoos, injuries, surgical effects, or other “physical perturbations.”
Exchangeable image file format data obtained from the camera, and preprocessing procedures like color normalization and “postprocessing” of images, such as filtering, should also be disclosed. In addition, developers should disclose and justify inclusion of images that have been created by an algorithm within a dataset. Any public images used in the datasets should have references, and privately used images should be made public where possible, the authors said.
The ISIC working group guidelines also provided recommendations for patient-level metadata. Each image should include a patient’s geographical location and medical center they visited as well as their age, sex and gender, ethnicity and/or race, and skin tone. Dr. Daneshjou said this was one area where she and her colleagues found a lack of transparency in AI datasets in algorithms in a recent review. “We found that many AI papers provided sparse details about the images used to train and test their algorithms,” Dr. Daneshjou explained. “For example, only 7 out of 70 papers had any information about the skin tones in the images used for developing and/or testing AI algorithms. Understanding the diversity of images used to train and test algorithms is important because algorithms that are developed on images of predominantly white skin likely won’t work as well on Black and brown skin.”
The guideline authors also asked algorithm developers to describe the limitations of not including patient-level metadata information when it is incomplete or unavailable. In addition, “we ask that algorithm developers comment on potential biases of their algorithms,” Dr. Daneshjou said. “For example, an algorithm based only on telemedicine images may not capture the full range of diseases seen within an in-person clinic.”
When describing their AI algorithm, developers should detail their reasoning for the dataset size and partitions, inclusion and exclusion criteria for images, and use of any external samples for test sets. “Authors should consider any differences between the image characteristics used for algorithm development and those that might be encountered in the real world,” the guidelines stated.
Recommendations for technique
How the images in a dataset are labeled is a unique challenge in developing AI algorithms for dermatology, the authors noted. Developers should use histopathological diagnosis in their labeling, but this can sometimes result in label noise.
“Many of the AI algorithms in dermatology use supervised learning, which requires labeled examples to help the algorithm ‘learn’ features for discriminating between lesions. We found that some papers use consensus labeling – dermatologists providing a label – to label skin cancers; however, the standard for diagnosing skin cancer is using histopathology from a biopsy,” she said. “Dermatologists can biopsy seven to eight suspected melanomas before discovering a true melanoma, so dermatologist labeling of skin cancers is prone to label noise.”
ISIC’s guidelines stated a gold standard of labeling for dermatologic images is one area that still needs future research, but currently, “diagnoses, labels and diagnostic groups used in data repositories as well as public ontologies” such as ICD-11, AnatomyMapper, and SNOMED-CT should be included in dermatologic image datasets.
AI developers should also provide a detailed description of their algorithm, which includes methods, work flows, mathematical formulas as well as the generalizability of the algorithm across more than one dataset.
Recommendations for technical assessment
“Another important recommendation is that algorithm developers should provide a way for algorithms to be publicly evaluable by researchers,” Dr. Daneshjou said. “Many dermatology AI algorithms do not share either their data or their algorithm. Algorithm sharing is important for assessing reproducibility and robustness.”
Google’s recently announced AI-powered dermatology assistant tool, for example, “has made claims about its accuracy and ability to diagnose skin disease at a dermatologist level, but there is no way for researchers to independently test these claims,” she said. Other options like Model Dermatology, developed by Seung Seog Han, MD, PhD, of the Dermatology Clinic in Seoul, South Korea, and colleagues, offer an application programming interface “that allows researchers to test the algorithm,” Dr. Daneshjou said. “This kind of openness is key for assessing algorithm robustness.”
Developers should also note in their algorithm explanations how performance markers and benchmarks would translate to proposed clinical application. “In this context,” the use case – the context in which the AI application is being used – “should be clearly described – who are the intended users and under what clinical scenario are they using the algorithm,” the authors wrote.
Recommendations for application
The guidelines note that use case for the model should also be described by the AI developers. “Our checklist includes delineating use cases for algorithms and describing what use cases may be within the scope of the algorithm versus which use cases are out of scope,” Dr. Daneshjou said. “For example, an algorithm developed to provide decision support to dermatologists, with a human in the loop, may not be accurate enough to release directly to consumers.”
As the goal of AI algorithms in dermatology is eventual implementation for clinicians and patients, the authors asked developers to consider shortcomings and potential harms of the algorithm during implementation. “Ethical considerations and impact on vulnerable populations should also be considered and discussed,” they wrote. An algorithm “suggesting aesthetic medical treatments may have negative effects given the biased nature of beauty standards,” and “an algorithm that diagnoses basal cell carcinomas but lacks any pigmented basal cell carcinomas, which are more often seen in skin of color, will not perform equitably across populations.”
Prior to implementing an AI algorithm, the ISIC working group recommended developers perform prospective clinical trials for validation. Checklists and guidelines like SPIRIT-AI and CONSORT-AI “provide guidance on how to design clinical trials to test AI algorithms,” Dr. Daneshjou said.
After implementation, “I believe we need additional research in how we monitor algorithms after they are deployed clinically, Dr. Daneshjou said. “Currently there are no [Food and Drug Administration]–approved AI algorithms in dermatology; however, there are several applications that have CE mark in Europe, and there are no mechanisms for postmarket surveillance there.
'Timely' recommendations
Commenting on the ISIC working group guidelines, Justin M. Ko, MD, MBA, director and chief of medical dermatology for Stanford Health Care, who was not involved with the work, said that the recommendations are timely and provide “a framework for a ‘common language’ around AI datasets specifically tailored to dermatology.” Dr. Ko, chair of the American Academy of Dermatology’s Ad Hoc Task Force on Augmented Intelligence, noted the work by Dr. Daneshjou and colleagues “is consistent with and builds further details” on the position statement released by the AAD AI task force in 2019.
“As machine-learning capabilities and commercial efforts continue to mature, it becomes increasingly important that we are able to ‘look under the hood,’ and evaluate all the critical factors that influence development of these capabilities,” he said in an interview. “A standard set of reporting guidelines not only allows for transparency in evaluating data and performance of models and algorithms, but also forces the consideration of issues of equity, fairness, mitigation of bias, and clinically meaningful outcomes.”
One concern is the impact of AI algorithms on societal or health systems, he noted, which is brought up in the guidelines. “The last thing we would want is the development of robust AI systems that exacerbate access challenges, or generate patient anxiety/worry, or drive low-value utilization, or adds to care team burden, or create a technological barrier to care, or increases inequity in dermatologic care,” he said.
In developing AI algorithms for dermatology, a “major practical issue” is how performance on paper will translate to real-world use, Dr. Ko explained, and the ISIC guidelines “provide a critical step in empowering clinicians, practices, and our field to shape the advent of the AI and augmented intelligence tools and systems to promote and enhance meaningful clinical outcomes, and augment the core patient-clinician relationship and ensure they are grounded in principles of fairness, equity and transparency.”
This research was funded by awards and grants to individual authors from the Charina Fund, a Google Research Award, Melanoma Research Alliance, National Health and Medical Research Council, National Institutes of Health/National Cancer Institute, National Science Foundation, and the Department of Veterans Affairs. The authors disclosed relationships with governmental entities, pharmaceutical companies, technology startups, medical publishers, charitable trusts, consulting firms, dermatology training companies, providers of medical devices, manufacturers of dermatologic products, and other organizations related to the paper in the form of supplied equipment, having founded a company; receiving grants, patents, or personal fees; holding shares; and medical reporting. Dr. Ko reported that he serves as a clinical advisor for Skin Analytics, and has an ongoing research collaboration with Google.
The
The guidelines, published in JAMA Dermatology on Dec. 1, 2021, contain a broad range of recommendations stakeholders should consider when developing and assessing image-based AI algorithms in dermatology. The recommendations are divided into categories of data, technique, technical assessment, and application. ISIC is “an academia and industry partnership designed to facilitate the application of digital skin imaging to help reduce melanoma mortality,” and is organized into different working groups, including the AI working group, according to its website.
“Our goal with these guidelines was to create higher-quality reporting of dataset and algorithm characteristics for dermatology AI,” first author Roxana Daneshjou, MD, PhD, clinical scholar in dermatology, in the department of dermatology at Stanford (Calif.) University, said in an interview. “We hope these guidelines also aid regulatory bodies around the world when they are assessing algorithms to be used in dermatology.”
Recommendations for data
The authors recommended that datasets used by AI algorithms have image descriptions and details on image artifacts. “For photography, these include the type of camera used; whether images were taken under standardized or varying conditions; whether they were taken by professional photographers, laymen, or health care professionals; and image quality,” they wrote. They also recommended that developers include in an image description the type of lighting used and whether the photo contains pen markings, hair, tattoos, injuries, surgical effects, or other “physical perturbations.”
Exchangeable image file format data obtained from the camera, and preprocessing procedures like color normalization and “postprocessing” of images, such as filtering, should also be disclosed. In addition, developers should disclose and justify inclusion of images that have been created by an algorithm within a dataset. Any public images used in the datasets should have references, and privately used images should be made public where possible, the authors said.
The ISIC working group guidelines also provided recommendations for patient-level metadata. Each image should include a patient’s geographical location and medical center they visited as well as their age, sex and gender, ethnicity and/or race, and skin tone. Dr. Daneshjou said this was one area where she and her colleagues found a lack of transparency in AI datasets in algorithms in a recent review. “We found that many AI papers provided sparse details about the images used to train and test their algorithms,” Dr. Daneshjou explained. “For example, only 7 out of 70 papers had any information about the skin tones in the images used for developing and/or testing AI algorithms. Understanding the diversity of images used to train and test algorithms is important because algorithms that are developed on images of predominantly white skin likely won’t work as well on Black and brown skin.”
The guideline authors also asked algorithm developers to describe the limitations of not including patient-level metadata information when it is incomplete or unavailable. In addition, “we ask that algorithm developers comment on potential biases of their algorithms,” Dr. Daneshjou said. “For example, an algorithm based only on telemedicine images may not capture the full range of diseases seen within an in-person clinic.”
When describing their AI algorithm, developers should detail their reasoning for the dataset size and partitions, inclusion and exclusion criteria for images, and use of any external samples for test sets. “Authors should consider any differences between the image characteristics used for algorithm development and those that might be encountered in the real world,” the guidelines stated.
Recommendations for technique
How the images in a dataset are labeled is a unique challenge in developing AI algorithms for dermatology, the authors noted. Developers should use histopathological diagnosis in their labeling, but this can sometimes result in label noise.
“Many of the AI algorithms in dermatology use supervised learning, which requires labeled examples to help the algorithm ‘learn’ features for discriminating between lesions. We found that some papers use consensus labeling – dermatologists providing a label – to label skin cancers; however, the standard for diagnosing skin cancer is using histopathology from a biopsy,” she said. “Dermatologists can biopsy seven to eight suspected melanomas before discovering a true melanoma, so dermatologist labeling of skin cancers is prone to label noise.”
ISIC’s guidelines stated a gold standard of labeling for dermatologic images is one area that still needs future research, but currently, “diagnoses, labels and diagnostic groups used in data repositories as well as public ontologies” such as ICD-11, AnatomyMapper, and SNOMED-CT should be included in dermatologic image datasets.
AI developers should also provide a detailed description of their algorithm, which includes methods, work flows, mathematical formulas as well as the generalizability of the algorithm across more than one dataset.
Recommendations for technical assessment
“Another important recommendation is that algorithm developers should provide a way for algorithms to be publicly evaluable by researchers,” Dr. Daneshjou said. “Many dermatology AI algorithms do not share either their data or their algorithm. Algorithm sharing is important for assessing reproducibility and robustness.”
Google’s recently announced AI-powered dermatology assistant tool, for example, “has made claims about its accuracy and ability to diagnose skin disease at a dermatologist level, but there is no way for researchers to independently test these claims,” she said. Other options like Model Dermatology, developed by Seung Seog Han, MD, PhD, of the Dermatology Clinic in Seoul, South Korea, and colleagues, offer an application programming interface “that allows researchers to test the algorithm,” Dr. Daneshjou said. “This kind of openness is key for assessing algorithm robustness.”
Developers should also note in their algorithm explanations how performance markers and benchmarks would translate to proposed clinical application. “In this context,” the use case – the context in which the AI application is being used – “should be clearly described – who are the intended users and under what clinical scenario are they using the algorithm,” the authors wrote.
Recommendations for application
The guidelines note that use case for the model should also be described by the AI developers. “Our checklist includes delineating use cases for algorithms and describing what use cases may be within the scope of the algorithm versus which use cases are out of scope,” Dr. Daneshjou said. “For example, an algorithm developed to provide decision support to dermatologists, with a human in the loop, may not be accurate enough to release directly to consumers.”
As the goal of AI algorithms in dermatology is eventual implementation for clinicians and patients, the authors asked developers to consider shortcomings and potential harms of the algorithm during implementation. “Ethical considerations and impact on vulnerable populations should also be considered and discussed,” they wrote. An algorithm “suggesting aesthetic medical treatments may have negative effects given the biased nature of beauty standards,” and “an algorithm that diagnoses basal cell carcinomas but lacks any pigmented basal cell carcinomas, which are more often seen in skin of color, will not perform equitably across populations.”
Prior to implementing an AI algorithm, the ISIC working group recommended developers perform prospective clinical trials for validation. Checklists and guidelines like SPIRIT-AI and CONSORT-AI “provide guidance on how to design clinical trials to test AI algorithms,” Dr. Daneshjou said.
After implementation, “I believe we need additional research in how we monitor algorithms after they are deployed clinically, Dr. Daneshjou said. “Currently there are no [Food and Drug Administration]–approved AI algorithms in dermatology; however, there are several applications that have CE mark in Europe, and there are no mechanisms for postmarket surveillance there.
'Timely' recommendations
Commenting on the ISIC working group guidelines, Justin M. Ko, MD, MBA, director and chief of medical dermatology for Stanford Health Care, who was not involved with the work, said that the recommendations are timely and provide “a framework for a ‘common language’ around AI datasets specifically tailored to dermatology.” Dr. Ko, chair of the American Academy of Dermatology’s Ad Hoc Task Force on Augmented Intelligence, noted the work by Dr. Daneshjou and colleagues “is consistent with and builds further details” on the position statement released by the AAD AI task force in 2019.
“As machine-learning capabilities and commercial efforts continue to mature, it becomes increasingly important that we are able to ‘look under the hood,’ and evaluate all the critical factors that influence development of these capabilities,” he said in an interview. “A standard set of reporting guidelines not only allows for transparency in evaluating data and performance of models and algorithms, but also forces the consideration of issues of equity, fairness, mitigation of bias, and clinically meaningful outcomes.”
One concern is the impact of AI algorithms on societal or health systems, he noted, which is brought up in the guidelines. “The last thing we would want is the development of robust AI systems that exacerbate access challenges, or generate patient anxiety/worry, or drive low-value utilization, or adds to care team burden, or create a technological barrier to care, or increases inequity in dermatologic care,” he said.
In developing AI algorithms for dermatology, a “major practical issue” is how performance on paper will translate to real-world use, Dr. Ko explained, and the ISIC guidelines “provide a critical step in empowering clinicians, practices, and our field to shape the advent of the AI and augmented intelligence tools and systems to promote and enhance meaningful clinical outcomes, and augment the core patient-clinician relationship and ensure they are grounded in principles of fairness, equity and transparency.”
This research was funded by awards and grants to individual authors from the Charina Fund, a Google Research Award, Melanoma Research Alliance, National Health and Medical Research Council, National Institutes of Health/National Cancer Institute, National Science Foundation, and the Department of Veterans Affairs. The authors disclosed relationships with governmental entities, pharmaceutical companies, technology startups, medical publishers, charitable trusts, consulting firms, dermatology training companies, providers of medical devices, manufacturers of dermatologic products, and other organizations related to the paper in the form of supplied equipment, having founded a company; receiving grants, patents, or personal fees; holding shares; and medical reporting. Dr. Ko reported that he serves as a clinical advisor for Skin Analytics, and has an ongoing research collaboration with Google.
FROM JAMA DERMATOLOGY
Metastatic uveal melanoma: New drugs in pipeline, but prognoses remain grim
No one’s quite sure what causes uveal melanoma (UM). Unlike skin cancers, UM doesn’t seem to have any link to exposure to ultraviolet rays, although it’s most likely to strike people who are vulnerable to sun damage, like Caucasians and people with lighter eyes and lighter skin (but not lighter hair), and an inability to tan. Up to half of those affected by the disease will recover after treatment. In the other half, the cancer spreads from the eye – typically to the liver – and patient prognoses remain extremely poor despite extensive efforts to develop effective treatments.
“The median survival is probably about 2 years, and there are a number of papers out there that talk about life expectancy as short as 6 months,” said Marlana Orloff, MD, an associate professor of medical oncology at Thomas Jefferson University Hospital, Philadelphia.
But there is hope on the horizon, even if it’s not as near as patients would prefer. “Just over the last 5-10 years, we’ve gained a lot more knowledge about this disease as we try to understand how distinctly different it is, how mutations drive it, and how we can approach it using immunotherapy,” Dr. Orloff said. “I hope we’ll come up with better options for prolonging survival.”
Tracking uveal melanoma’s dangerous course
All melanomas, including UM, strike the melanocytes (cells) that provide pigment. According to a 2017 report1 in the journal Eye, “uveal melanoma is the most common primary intraocular tumor in adults with a mean age-adjusted incidence of 5.1 cases per million per year. Tumors are located either in the iris (4%), ciliary body (6%), or choroid (90%) . … As in many other cancer indications, both early detection and early treatment could be critical for a positive long-term survival outcome in uveal melanoma.”
The median age of diagnosis is 59-62 years, the report says, although non-Whites seem to develop the disease earlier.
The vast majority of patients receive treatment by plaque brachytherapy via radioactive seeds. “It’s like brachytherapy of the prostate,” said medical oncologist Rino S. Seedor, MD, of Thomas Jefferson University Hospital. “If the eye tumor is too big or invasive, they’ll cut out the eye.”
As many as 50% of patients will develop metastasis, sometimes within 2-3 years in those who have large tumors and high genetic risk, said ophthalmologist and radiation oncologist Miguel Materin, MD, of Duke University Eye Center, Durham, N.C. “There’s probably micrometastasis early in the development of the tumor,” he said. “The metastasis might develop before we or the patient knows there’s a tumor.”
Some physicians question the value of prognostic testing in patients who don’t yet show signs of metastasis, Dr. Materin said, because the findings can be grim.
Unlike his more cautious colleagues, Dr. Materin prefers to pursue testing, he said. Most patients agree to it. “It’s up to them to decide if they want to know if they have a bad prognosis,” he said, and the findings can be helpful to physicians because they provide useful genetic information about tumors.
Monitoring for liver metastasis is key
UM metastases are most likely to strike the liver, and prognoses are especially poor when they do. According to a 2019 analysis of 175 patients with metastatic UM in the Netherlands, “the presence of liver metastases is negatively associated with survival (hazard ratio = 2.09; 95% confidence interval, 1.07-4.08). … In 154 (88%) patients, the liver was affected, and only 3 patients were reported to have brain metastases.”2
As a result, physicians recommend close monitoring in patients with UM. Thomas Jefferson University’s Dr. Orloff uses tumor stages and genetic risk profiles to guide surveillance. “Very large tumors and/or monosomy 3 and 8q amplification or a Class 2 gene signature would suggest a higher-risk tumor,” she said. “For these patients we recommend MRI of the abdomen every 3 months for 2 years, CT of the chest every 6 months for 2 years, labs every 3 months for 2 years, then MRI every 6 months until year 5 with chest imaging yearly, then at 5 years everything yearly. For lower- or intermediate-risk patients we recommend MRI of the abdomen every 6 months for 5 years, chest imaging yearly, labs every 6 months, then at 5 years everything yearly.”
In the United States, patients with metastatic disease are typically sent to referral centers at institutions such as Duke, Yale (New Haven, Conn.), and Thomas Jefferson universities.
Metastasis treatments offer limited relief
There are no FDA-approved treatments for metastatic MU, and the treatments that physicians do use don’t seem to have much of an effect on life span. A 2019 study examined 73 patients with MU metastasis to the liver who were treated from 2004 to 2011 and 2012 to 2016. Among both cohorts, those who had no treatment lived nearly as long (median of 15 months) as those treated with local therapy (median of 18.7 months). Median survival for the entire population was 15 months (95% CI: 11–18 months). There was no statistically significant difference between the periods.3
However, there are signs that a move away from first-line chemotherapy in recent decades has led to longer life spans. Dr. Seedor led a 2018 study4 that compared two cohorts of MU patients with liver metastasis at her university: 98 patients from 1971 to 1993 (81% received systemic chemotherapy as their initial therapy) and 574 from 2000 to 2017 (they received various liver-directed initial treatments such as chemoembolization, drug-eluting beads, immunoembolization, and radioembolization).
The patients in the second group lived longer after treatment of initial UM than the first group (5.1 years vs. 3.3 years, P < .001) and after the development of liver metastasis (16.4 months vs. 4.8 months, P < .001). A 2020 follow-up study reported similar findings and noted that a “combination of liver-directed and newly developed systemic treatments may further improve the survival of these patients.”5
At Thomas Jefferson Medical Center, liver-directed therapy includes radioembolization, chemomobilization, and microwave ablation, Dr. Seedor said. “Which one we choose is based on how big the tumors are.”
Treatments in development could make advances
Physicians are working on several fronts to develop new treatments. A 2021 review of clinical trials found numerous trials regarding checkpoint inhibition, one devoted to a vaccine, and several involving checkpoint inhibitors. The review author notes that “the low mutational burden and poor immunogenicity of UM tumors may underlie poor responses and resistance to [immune checkpoint inhibitors] alone.”6
Earlier this year, grant-funded researchers reported encouraging news on the G protein inhibitor front. Their study found that FR900359, a selective inhibitor of the Gq/11/14 subfamily of heterotrimeric G proteins, could hold promise for “treating UM and potentially other diseases caused by constitutively active Gq/11.”7
In another 2021 study, this one with no reported funding, researchers explored the tumor microenvironment of UM and reported that their findings “provided a robust gene-based prognostic signature for predicting prognosis of UM patients and proposed a potential targeted therapy for preventing UM metastasis.”8
Experimental drug may add months of life
Physicians often recommend that patients take part in clinical trials. Earlier this year, researchers reported that a drug called tebentafusp – a bispecific fusion protein – slightly boosted metastatic UM survival in an open-label, phase 3 clinical trial when used as a first-line treatment. Patients were randomly assigned to tebentafusp, 1 of 2 immunotherapy drugs (ipilimumab or pembrolizumab), or the chemotherapy drug dacarbazine. Those who took tebentafusp vs. the other options lived longer with an estimated 1-year overall rate of 73.2% (95% CI: 66.3-78.9) vs. 57.5% (95% CI: 47.0-66.6), respectively. Fewer than 4% of those on tebentafusp needed to stop it because of adverse effects, and no treatment-related deaths occurred.9
Dr. Orloff is one of the coauthors of this study.
The National Cancer Institute provided more details about the industry-funded research and noted that median overall survival for patients who received the drug was 21.7 months vs. 16 months for the control group.
Not every patient is eligible for this treatment, however. A coauthor told the American Association for Cancer Research that “the major limitation of tebentafusp is that it can only be used in patients who have a specific HLA [human leukocyte antigen] type.” Patients must be HLA-A*0201-positive.10
In August 2021, the FDA granted priority review for tebentafusp.11 And in September 2021, a company called TriSalus announced the first patient enrollment in a “clinical study evaluating the administration of SD-101, an investigational toll-like receptor 9 (TLR9) agonist in adults with metastatic uveal melanoma.”12
According to the company, the research “is designed to evaluate the intravascular administration of SD-101 into uveal melanoma liver metastasis lesions in combination with checkpoint inhibitors using the novel Pressure-Enabled Drug Delivery (PEDD) approach.” This strategy is “designed to overcome the inherent intratumoral pressure of solid tumors,” the company said.
Dr. Materin serves on a scientific advisory board for Castle Biosciences. Dr. Orloff is a consultant for Immunocore, which funded the tebentafusp study, and serves on a scientific advisory board for TriSalus. Dr. Seedor reports no disclosures.
References
1.Kaliki S and Shields C. Eye. 2017 Feb;31:241-57.
2.Jochems A et al. Cancers. 2019 July;11(7):1007.
3.Xu LT et al. Ocul Oncol Pathol. 2019;5:323-32.
4.Seedor RS et al. J Clin Oncol. 2018 May;36(15_suppl):9592.
5.Seedor RS et al. Cancers (Basel). 2020 Jan 1;12(1):117.
6.Orloff M. Ocul Oncol Pathol. 2021 July;7:168-76.
7.Onken MD et al. J Biol Chem. 2021;296:100403.
8.Lei S and Zhang Y. Int Immunopharmacol. 2021 July;96:107816.
9.Piperno-Neumann S et al. Proceedings of the 112th Annual Meeting of the American Association for Cancer Research; 2021 April 10-15. Philadelphia (Pa.): AACR; 2021. Abstract nr 5133.
10.National Cancer Institute: https://www.cancer.gov/news-events/cancer-currents-blog/2021/tebentafusp-uveal-melanoma-improves-survival
11.Immunocore press release: https://ir.immunocore.com/news-releases/news-release-details/immunocore-announces-us-food-and-drug-administration-and
12.Trisalus announcement: https://finance.yahoo.com/news/trisalus-life-sciences-announces-first-130000215.html?guccounter=1
No one’s quite sure what causes uveal melanoma (UM). Unlike skin cancers, UM doesn’t seem to have any link to exposure to ultraviolet rays, although it’s most likely to strike people who are vulnerable to sun damage, like Caucasians and people with lighter eyes and lighter skin (but not lighter hair), and an inability to tan. Up to half of those affected by the disease will recover after treatment. In the other half, the cancer spreads from the eye – typically to the liver – and patient prognoses remain extremely poor despite extensive efforts to develop effective treatments.
“The median survival is probably about 2 years, and there are a number of papers out there that talk about life expectancy as short as 6 months,” said Marlana Orloff, MD, an associate professor of medical oncology at Thomas Jefferson University Hospital, Philadelphia.
But there is hope on the horizon, even if it’s not as near as patients would prefer. “Just over the last 5-10 years, we’ve gained a lot more knowledge about this disease as we try to understand how distinctly different it is, how mutations drive it, and how we can approach it using immunotherapy,” Dr. Orloff said. “I hope we’ll come up with better options for prolonging survival.”
Tracking uveal melanoma’s dangerous course
All melanomas, including UM, strike the melanocytes (cells) that provide pigment. According to a 2017 report1 in the journal Eye, “uveal melanoma is the most common primary intraocular tumor in adults with a mean age-adjusted incidence of 5.1 cases per million per year. Tumors are located either in the iris (4%), ciliary body (6%), or choroid (90%) . … As in many other cancer indications, both early detection and early treatment could be critical for a positive long-term survival outcome in uveal melanoma.”
The median age of diagnosis is 59-62 years, the report says, although non-Whites seem to develop the disease earlier.
The vast majority of patients receive treatment by plaque brachytherapy via radioactive seeds. “It’s like brachytherapy of the prostate,” said medical oncologist Rino S. Seedor, MD, of Thomas Jefferson University Hospital. “If the eye tumor is too big or invasive, they’ll cut out the eye.”
As many as 50% of patients will develop metastasis, sometimes within 2-3 years in those who have large tumors and high genetic risk, said ophthalmologist and radiation oncologist Miguel Materin, MD, of Duke University Eye Center, Durham, N.C. “There’s probably micrometastasis early in the development of the tumor,” he said. “The metastasis might develop before we or the patient knows there’s a tumor.”
Some physicians question the value of prognostic testing in patients who don’t yet show signs of metastasis, Dr. Materin said, because the findings can be grim.
Unlike his more cautious colleagues, Dr. Materin prefers to pursue testing, he said. Most patients agree to it. “It’s up to them to decide if they want to know if they have a bad prognosis,” he said, and the findings can be helpful to physicians because they provide useful genetic information about tumors.
Monitoring for liver metastasis is key
UM metastases are most likely to strike the liver, and prognoses are especially poor when they do. According to a 2019 analysis of 175 patients with metastatic UM in the Netherlands, “the presence of liver metastases is negatively associated with survival (hazard ratio = 2.09; 95% confidence interval, 1.07-4.08). … In 154 (88%) patients, the liver was affected, and only 3 patients were reported to have brain metastases.”2
As a result, physicians recommend close monitoring in patients with UM. Thomas Jefferson University’s Dr. Orloff uses tumor stages and genetic risk profiles to guide surveillance. “Very large tumors and/or monosomy 3 and 8q amplification or a Class 2 gene signature would suggest a higher-risk tumor,” she said. “For these patients we recommend MRI of the abdomen every 3 months for 2 years, CT of the chest every 6 months for 2 years, labs every 3 months for 2 years, then MRI every 6 months until year 5 with chest imaging yearly, then at 5 years everything yearly. For lower- or intermediate-risk patients we recommend MRI of the abdomen every 6 months for 5 years, chest imaging yearly, labs every 6 months, then at 5 years everything yearly.”
In the United States, patients with metastatic disease are typically sent to referral centers at institutions such as Duke, Yale (New Haven, Conn.), and Thomas Jefferson universities.
Metastasis treatments offer limited relief
There are no FDA-approved treatments for metastatic MU, and the treatments that physicians do use don’t seem to have much of an effect on life span. A 2019 study examined 73 patients with MU metastasis to the liver who were treated from 2004 to 2011 and 2012 to 2016. Among both cohorts, those who had no treatment lived nearly as long (median of 15 months) as those treated with local therapy (median of 18.7 months). Median survival for the entire population was 15 months (95% CI: 11–18 months). There was no statistically significant difference between the periods.3
However, there are signs that a move away from first-line chemotherapy in recent decades has led to longer life spans. Dr. Seedor led a 2018 study4 that compared two cohorts of MU patients with liver metastasis at her university: 98 patients from 1971 to 1993 (81% received systemic chemotherapy as their initial therapy) and 574 from 2000 to 2017 (they received various liver-directed initial treatments such as chemoembolization, drug-eluting beads, immunoembolization, and radioembolization).
The patients in the second group lived longer after treatment of initial UM than the first group (5.1 years vs. 3.3 years, P < .001) and after the development of liver metastasis (16.4 months vs. 4.8 months, P < .001). A 2020 follow-up study reported similar findings and noted that a “combination of liver-directed and newly developed systemic treatments may further improve the survival of these patients.”5
At Thomas Jefferson Medical Center, liver-directed therapy includes radioembolization, chemomobilization, and microwave ablation, Dr. Seedor said. “Which one we choose is based on how big the tumors are.”
Treatments in development could make advances
Physicians are working on several fronts to develop new treatments. A 2021 review of clinical trials found numerous trials regarding checkpoint inhibition, one devoted to a vaccine, and several involving checkpoint inhibitors. The review author notes that “the low mutational burden and poor immunogenicity of UM tumors may underlie poor responses and resistance to [immune checkpoint inhibitors] alone.”6
Earlier this year, grant-funded researchers reported encouraging news on the G protein inhibitor front. Their study found that FR900359, a selective inhibitor of the Gq/11/14 subfamily of heterotrimeric G proteins, could hold promise for “treating UM and potentially other diseases caused by constitutively active Gq/11.”7
In another 2021 study, this one with no reported funding, researchers explored the tumor microenvironment of UM and reported that their findings “provided a robust gene-based prognostic signature for predicting prognosis of UM patients and proposed a potential targeted therapy for preventing UM metastasis.”8
Experimental drug may add months of life
Physicians often recommend that patients take part in clinical trials. Earlier this year, researchers reported that a drug called tebentafusp – a bispecific fusion protein – slightly boosted metastatic UM survival in an open-label, phase 3 clinical trial when used as a first-line treatment. Patients were randomly assigned to tebentafusp, 1 of 2 immunotherapy drugs (ipilimumab or pembrolizumab), or the chemotherapy drug dacarbazine. Those who took tebentafusp vs. the other options lived longer with an estimated 1-year overall rate of 73.2% (95% CI: 66.3-78.9) vs. 57.5% (95% CI: 47.0-66.6), respectively. Fewer than 4% of those on tebentafusp needed to stop it because of adverse effects, and no treatment-related deaths occurred.9
Dr. Orloff is one of the coauthors of this study.
The National Cancer Institute provided more details about the industry-funded research and noted that median overall survival for patients who received the drug was 21.7 months vs. 16 months for the control group.
Not every patient is eligible for this treatment, however. A coauthor told the American Association for Cancer Research that “the major limitation of tebentafusp is that it can only be used in patients who have a specific HLA [human leukocyte antigen] type.” Patients must be HLA-A*0201-positive.10
In August 2021, the FDA granted priority review for tebentafusp.11 And in September 2021, a company called TriSalus announced the first patient enrollment in a “clinical study evaluating the administration of SD-101, an investigational toll-like receptor 9 (TLR9) agonist in adults with metastatic uveal melanoma.”12
According to the company, the research “is designed to evaluate the intravascular administration of SD-101 into uveal melanoma liver metastasis lesions in combination with checkpoint inhibitors using the novel Pressure-Enabled Drug Delivery (PEDD) approach.” This strategy is “designed to overcome the inherent intratumoral pressure of solid tumors,” the company said.
Dr. Materin serves on a scientific advisory board for Castle Biosciences. Dr. Orloff is a consultant for Immunocore, which funded the tebentafusp study, and serves on a scientific advisory board for TriSalus. Dr. Seedor reports no disclosures.
References
1.Kaliki S and Shields C. Eye. 2017 Feb;31:241-57.
2.Jochems A et al. Cancers. 2019 July;11(7):1007.
3.Xu LT et al. Ocul Oncol Pathol. 2019;5:323-32.
4.Seedor RS et al. J Clin Oncol. 2018 May;36(15_suppl):9592.
5.Seedor RS et al. Cancers (Basel). 2020 Jan 1;12(1):117.
6.Orloff M. Ocul Oncol Pathol. 2021 July;7:168-76.
7.Onken MD et al. J Biol Chem. 2021;296:100403.
8.Lei S and Zhang Y. Int Immunopharmacol. 2021 July;96:107816.
9.Piperno-Neumann S et al. Proceedings of the 112th Annual Meeting of the American Association for Cancer Research; 2021 April 10-15. Philadelphia (Pa.): AACR; 2021. Abstract nr 5133.
10.National Cancer Institute: https://www.cancer.gov/news-events/cancer-currents-blog/2021/tebentafusp-uveal-melanoma-improves-survival
11.Immunocore press release: https://ir.immunocore.com/news-releases/news-release-details/immunocore-announces-us-food-and-drug-administration-and
12.Trisalus announcement: https://finance.yahoo.com/news/trisalus-life-sciences-announces-first-130000215.html?guccounter=1
No one’s quite sure what causes uveal melanoma (UM). Unlike skin cancers, UM doesn’t seem to have any link to exposure to ultraviolet rays, although it’s most likely to strike people who are vulnerable to sun damage, like Caucasians and people with lighter eyes and lighter skin (but not lighter hair), and an inability to tan. Up to half of those affected by the disease will recover after treatment. In the other half, the cancer spreads from the eye – typically to the liver – and patient prognoses remain extremely poor despite extensive efforts to develop effective treatments.
“The median survival is probably about 2 years, and there are a number of papers out there that talk about life expectancy as short as 6 months,” said Marlana Orloff, MD, an associate professor of medical oncology at Thomas Jefferson University Hospital, Philadelphia.
But there is hope on the horizon, even if it’s not as near as patients would prefer. “Just over the last 5-10 years, we’ve gained a lot more knowledge about this disease as we try to understand how distinctly different it is, how mutations drive it, and how we can approach it using immunotherapy,” Dr. Orloff said. “I hope we’ll come up with better options for prolonging survival.”
Tracking uveal melanoma’s dangerous course
All melanomas, including UM, strike the melanocytes (cells) that provide pigment. According to a 2017 report1 in the journal Eye, “uveal melanoma is the most common primary intraocular tumor in adults with a mean age-adjusted incidence of 5.1 cases per million per year. Tumors are located either in the iris (4%), ciliary body (6%), or choroid (90%) . … As in many other cancer indications, both early detection and early treatment could be critical for a positive long-term survival outcome in uveal melanoma.”
The median age of diagnosis is 59-62 years, the report says, although non-Whites seem to develop the disease earlier.
The vast majority of patients receive treatment by plaque brachytherapy via radioactive seeds. “It’s like brachytherapy of the prostate,” said medical oncologist Rino S. Seedor, MD, of Thomas Jefferson University Hospital. “If the eye tumor is too big or invasive, they’ll cut out the eye.”
As many as 50% of patients will develop metastasis, sometimes within 2-3 years in those who have large tumors and high genetic risk, said ophthalmologist and radiation oncologist Miguel Materin, MD, of Duke University Eye Center, Durham, N.C. “There’s probably micrometastasis early in the development of the tumor,” he said. “The metastasis might develop before we or the patient knows there’s a tumor.”
Some physicians question the value of prognostic testing in patients who don’t yet show signs of metastasis, Dr. Materin said, because the findings can be grim.
Unlike his more cautious colleagues, Dr. Materin prefers to pursue testing, he said. Most patients agree to it. “It’s up to them to decide if they want to know if they have a bad prognosis,” he said, and the findings can be helpful to physicians because they provide useful genetic information about tumors.
Monitoring for liver metastasis is key
UM metastases are most likely to strike the liver, and prognoses are especially poor when they do. According to a 2019 analysis of 175 patients with metastatic UM in the Netherlands, “the presence of liver metastases is negatively associated with survival (hazard ratio = 2.09; 95% confidence interval, 1.07-4.08). … In 154 (88%) patients, the liver was affected, and only 3 patients were reported to have brain metastases.”2
As a result, physicians recommend close monitoring in patients with UM. Thomas Jefferson University’s Dr. Orloff uses tumor stages and genetic risk profiles to guide surveillance. “Very large tumors and/or monosomy 3 and 8q amplification or a Class 2 gene signature would suggest a higher-risk tumor,” she said. “For these patients we recommend MRI of the abdomen every 3 months for 2 years, CT of the chest every 6 months for 2 years, labs every 3 months for 2 years, then MRI every 6 months until year 5 with chest imaging yearly, then at 5 years everything yearly. For lower- or intermediate-risk patients we recommend MRI of the abdomen every 6 months for 5 years, chest imaging yearly, labs every 6 months, then at 5 years everything yearly.”
In the United States, patients with metastatic disease are typically sent to referral centers at institutions such as Duke, Yale (New Haven, Conn.), and Thomas Jefferson universities.
Metastasis treatments offer limited relief
There are no FDA-approved treatments for metastatic MU, and the treatments that physicians do use don’t seem to have much of an effect on life span. A 2019 study examined 73 patients with MU metastasis to the liver who were treated from 2004 to 2011 and 2012 to 2016. Among both cohorts, those who had no treatment lived nearly as long (median of 15 months) as those treated with local therapy (median of 18.7 months). Median survival for the entire population was 15 months (95% CI: 11–18 months). There was no statistically significant difference between the periods.3
However, there are signs that a move away from first-line chemotherapy in recent decades has led to longer life spans. Dr. Seedor led a 2018 study4 that compared two cohorts of MU patients with liver metastasis at her university: 98 patients from 1971 to 1993 (81% received systemic chemotherapy as their initial therapy) and 574 from 2000 to 2017 (they received various liver-directed initial treatments such as chemoembolization, drug-eluting beads, immunoembolization, and radioembolization).
The patients in the second group lived longer after treatment of initial UM than the first group (5.1 years vs. 3.3 years, P < .001) and after the development of liver metastasis (16.4 months vs. 4.8 months, P < .001). A 2020 follow-up study reported similar findings and noted that a “combination of liver-directed and newly developed systemic treatments may further improve the survival of these patients.”5
At Thomas Jefferson Medical Center, liver-directed therapy includes radioembolization, chemomobilization, and microwave ablation, Dr. Seedor said. “Which one we choose is based on how big the tumors are.”
Treatments in development could make advances
Physicians are working on several fronts to develop new treatments. A 2021 review of clinical trials found numerous trials regarding checkpoint inhibition, one devoted to a vaccine, and several involving checkpoint inhibitors. The review author notes that “the low mutational burden and poor immunogenicity of UM tumors may underlie poor responses and resistance to [immune checkpoint inhibitors] alone.”6
Earlier this year, grant-funded researchers reported encouraging news on the G protein inhibitor front. Their study found that FR900359, a selective inhibitor of the Gq/11/14 subfamily of heterotrimeric G proteins, could hold promise for “treating UM and potentially other diseases caused by constitutively active Gq/11.”7
In another 2021 study, this one with no reported funding, researchers explored the tumor microenvironment of UM and reported that their findings “provided a robust gene-based prognostic signature for predicting prognosis of UM patients and proposed a potential targeted therapy for preventing UM metastasis.”8
Experimental drug may add months of life
Physicians often recommend that patients take part in clinical trials. Earlier this year, researchers reported that a drug called tebentafusp – a bispecific fusion protein – slightly boosted metastatic UM survival in an open-label, phase 3 clinical trial when used as a first-line treatment. Patients were randomly assigned to tebentafusp, 1 of 2 immunotherapy drugs (ipilimumab or pembrolizumab), or the chemotherapy drug dacarbazine. Those who took tebentafusp vs. the other options lived longer with an estimated 1-year overall rate of 73.2% (95% CI: 66.3-78.9) vs. 57.5% (95% CI: 47.0-66.6), respectively. Fewer than 4% of those on tebentafusp needed to stop it because of adverse effects, and no treatment-related deaths occurred.9
Dr. Orloff is one of the coauthors of this study.
The National Cancer Institute provided more details about the industry-funded research and noted that median overall survival for patients who received the drug was 21.7 months vs. 16 months for the control group.
Not every patient is eligible for this treatment, however. A coauthor told the American Association for Cancer Research that “the major limitation of tebentafusp is that it can only be used in patients who have a specific HLA [human leukocyte antigen] type.” Patients must be HLA-A*0201-positive.10
In August 2021, the FDA granted priority review for tebentafusp.11 And in September 2021, a company called TriSalus announced the first patient enrollment in a “clinical study evaluating the administration of SD-101, an investigational toll-like receptor 9 (TLR9) agonist in adults with metastatic uveal melanoma.”12
According to the company, the research “is designed to evaluate the intravascular administration of SD-101 into uveal melanoma liver metastasis lesions in combination with checkpoint inhibitors using the novel Pressure-Enabled Drug Delivery (PEDD) approach.” This strategy is “designed to overcome the inherent intratumoral pressure of solid tumors,” the company said.
Dr. Materin serves on a scientific advisory board for Castle Biosciences. Dr. Orloff is a consultant for Immunocore, which funded the tebentafusp study, and serves on a scientific advisory board for TriSalus. Dr. Seedor reports no disclosures.
References
1.Kaliki S and Shields C. Eye. 2017 Feb;31:241-57.
2.Jochems A et al. Cancers. 2019 July;11(7):1007.
3.Xu LT et al. Ocul Oncol Pathol. 2019;5:323-32.
4.Seedor RS et al. J Clin Oncol. 2018 May;36(15_suppl):9592.
5.Seedor RS et al. Cancers (Basel). 2020 Jan 1;12(1):117.
6.Orloff M. Ocul Oncol Pathol. 2021 July;7:168-76.
7.Onken MD et al. J Biol Chem. 2021;296:100403.
8.Lei S and Zhang Y. Int Immunopharmacol. 2021 July;96:107816.
9.Piperno-Neumann S et al. Proceedings of the 112th Annual Meeting of the American Association for Cancer Research; 2021 April 10-15. Philadelphia (Pa.): AACR; 2021. Abstract nr 5133.
10.National Cancer Institute: https://www.cancer.gov/news-events/cancer-currents-blog/2021/tebentafusp-uveal-melanoma-improves-survival
11.Immunocore press release: https://ir.immunocore.com/news-releases/news-release-details/immunocore-announces-us-food-and-drug-administration-and
12.Trisalus announcement: https://finance.yahoo.com/news/trisalus-life-sciences-announces-first-130000215.html?guccounter=1