User login
Nab-paclitaxel marginally superior to dacarbazine for metastatic melanoma
In chemotherapy-naive patients with metastatic melanoma, progression-free survival (PFS) was significantly longer with nab-paclitaxel, compared with dacarbazine, according to results from a phase III trial.
Median PFS for the nab-paclitaxel arm vs. the dacarbazine arm was 4.8 vs. 2.5 months, respectively (hazard ratio, 0.792; 95% confidence interval, 0.631-0.992; P = .044). Overall survival was similar in the two arms: 12.6 months for nab-paclitaxel vs. 10.5 months for dacarbazine (P = .27). The nab-paclitaxel group had an increased, but not significant, overall response rate (15% vs. 11%) and a significantly improved disease control rate (P = .004). The disease control rate reflects the number of complete responses plus partial responses plus stable disease for 16 or more weeks.
Nab-paclitaxel benefited patients regardless of BRAF mutation status, noted Dr. Evan Hersh, professor of medicine at the University of Arizona, Tucson, and colleagues.
“Additionally, in a post hoc analysis of this trial, nab-paclitaxel was shown to benefit a subgroup of patients with low or absent TILs [tumor-infiltrating lymphocytes], a poor prognostic factor in melanoma,” they wrote (Ann Oncol 2015 Sep 26. doi: 10.1093/annonc/mdv324).
The most common treatment-related adverse events of grade 3 or greater were neuropathy (25% vs. 0%), neutropenia (20% vs. 10%), and leukopenia (12% vs. 7%) for the nab-paclitaxel and dacarbazine arms, respectively. The median onset of grade 3 or greater peripheral neuropathy was 101 days after treatment began.
Between April 2009 and June 2011, the multicenter, phase III, randomized, controlled trial enrolled 529 adults with stage IV malignant melanoma who had no prior cytotoxic therapy.
A majority of participants from both treatment arms underwent post-study therapy, including newer agents such as BRAF inhibitors and ipilimumab. This may account for the diminished treatment affect of nab-paclitaxel on overall survival, compared with the more significant benefit in PFS observed early in the study.
A biomarker analysis found no correlation between tumor expression of the albumin-binding protein SPARC and PFS with nab-paclitaxel treatment. SPARC was hypothesized to enrich nab-paclitaxel in the tumor microenvironment thereby increasing its efficacy.
The study by Hersh et al. was conceived prior to the success of immune checkpoint inhibitors and targeted therapies for melanoma. The treatment landscape has changed rapidly, and the current role of chemotherapy, and nab-paclitaxel specifically, is uncertain.
In the last 5 years, four classes of therapies have significantly improved overall survival over dacarbazine: ipilimumab (HR, 0.72; P less than .001), vemurafenib (HR, 0.37; P less than .001), trametinib (HR, 0.54; P = .01), and nivolumab (HR, 0.42; P less than .001).
For the large majority of patients, chemotherapy would not be considered for the first line of therapy. Patients typically considered for chemotherapy would have failed or would have been intolerant to ipilimumab and an anti-PD1 agent, as well as BRAF/MEK inhibitors if the tumors were BRAF-mutation positive. In these heavily pretreated patients, however, toxicities associated with chemotherapy may outweigh benefits.
In patients with a history of autoimmune disease, some have suggested checkpoint inhibitors may exacerbate these conditions. However, recent retrospective studies suggest that ipilimumab is safe for patients with autoimmune disorders such as inflammatory bowel disease and rheumatoid arthritis.
The current study did not include quality of life data, and given the lack of improvement in overall survival, the superiority of nab-paclitaxel over dacarbazine has not been shown definitively.
Chemotherapy may have a role in combination with targeted or immunotherapeutic agents. Checkpoint inhibitors may be more active in patients with baseline tumor-infiltrating lymphocytes (TILs) and tumor PDL1 expression. Chemotherapy may induce TILs in tumors where the cells are low or absent, and nab-paclitaxel may be a good candidate to explore such combinations.
It remains to be determined if chemotherapy offers greater benefit than the many other strategies under consideration. This brings to the forefront the urgent need for preclinical models to enable comparisons of combination strategies.
Dr. Matteo Carlino is a clinical senior lecturer at the Sydney Medical School, University of Sydney and a medical oncologist at Westmead Hospital and Blacktown Hospital, Australia. Dr. Georgina Long is an associate professor and medical oncologist at Melanoma Institute Australia, University of Sydney. These remarks were part of an editorial accompanying the report (Ann Oncol. 2015 Sep 15. doi: 10.1093/annonc/mdv361). Dr. Carlino and Dr. Long reported having no disclosures.
The study by Hersh et al. was conceived prior to the success of immune checkpoint inhibitors and targeted therapies for melanoma. The treatment landscape has changed rapidly, and the current role of chemotherapy, and nab-paclitaxel specifically, is uncertain.
In the last 5 years, four classes of therapies have significantly improved overall survival over dacarbazine: ipilimumab (HR, 0.72; P less than .001), vemurafenib (HR, 0.37; P less than .001), trametinib (HR, 0.54; P = .01), and nivolumab (HR, 0.42; P less than .001).
For the large majority of patients, chemotherapy would not be considered for the first line of therapy. Patients typically considered for chemotherapy would have failed or would have been intolerant to ipilimumab and an anti-PD1 agent, as well as BRAF/MEK inhibitors if the tumors were BRAF-mutation positive. In these heavily pretreated patients, however, toxicities associated with chemotherapy may outweigh benefits.
In patients with a history of autoimmune disease, some have suggested checkpoint inhibitors may exacerbate these conditions. However, recent retrospective studies suggest that ipilimumab is safe for patients with autoimmune disorders such as inflammatory bowel disease and rheumatoid arthritis.
The current study did not include quality of life data, and given the lack of improvement in overall survival, the superiority of nab-paclitaxel over dacarbazine has not been shown definitively.
Chemotherapy may have a role in combination with targeted or immunotherapeutic agents. Checkpoint inhibitors may be more active in patients with baseline tumor-infiltrating lymphocytes (TILs) and tumor PDL1 expression. Chemotherapy may induce TILs in tumors where the cells are low or absent, and nab-paclitaxel may be a good candidate to explore such combinations.
It remains to be determined if chemotherapy offers greater benefit than the many other strategies under consideration. This brings to the forefront the urgent need for preclinical models to enable comparisons of combination strategies.
Dr. Matteo Carlino is a clinical senior lecturer at the Sydney Medical School, University of Sydney and a medical oncologist at Westmead Hospital and Blacktown Hospital, Australia. Dr. Georgina Long is an associate professor and medical oncologist at Melanoma Institute Australia, University of Sydney. These remarks were part of an editorial accompanying the report (Ann Oncol. 2015 Sep 15. doi: 10.1093/annonc/mdv361). Dr. Carlino and Dr. Long reported having no disclosures.
The study by Hersh et al. was conceived prior to the success of immune checkpoint inhibitors and targeted therapies for melanoma. The treatment landscape has changed rapidly, and the current role of chemotherapy, and nab-paclitaxel specifically, is uncertain.
In the last 5 years, four classes of therapies have significantly improved overall survival over dacarbazine: ipilimumab (HR, 0.72; P less than .001), vemurafenib (HR, 0.37; P less than .001), trametinib (HR, 0.54; P = .01), and nivolumab (HR, 0.42; P less than .001).
For the large majority of patients, chemotherapy would not be considered for the first line of therapy. Patients typically considered for chemotherapy would have failed or would have been intolerant to ipilimumab and an anti-PD1 agent, as well as BRAF/MEK inhibitors if the tumors were BRAF-mutation positive. In these heavily pretreated patients, however, toxicities associated with chemotherapy may outweigh benefits.
In patients with a history of autoimmune disease, some have suggested checkpoint inhibitors may exacerbate these conditions. However, recent retrospective studies suggest that ipilimumab is safe for patients with autoimmune disorders such as inflammatory bowel disease and rheumatoid arthritis.
The current study did not include quality of life data, and given the lack of improvement in overall survival, the superiority of nab-paclitaxel over dacarbazine has not been shown definitively.
Chemotherapy may have a role in combination with targeted or immunotherapeutic agents. Checkpoint inhibitors may be more active in patients with baseline tumor-infiltrating lymphocytes (TILs) and tumor PDL1 expression. Chemotherapy may induce TILs in tumors where the cells are low or absent, and nab-paclitaxel may be a good candidate to explore such combinations.
It remains to be determined if chemotherapy offers greater benefit than the many other strategies under consideration. This brings to the forefront the urgent need for preclinical models to enable comparisons of combination strategies.
Dr. Matteo Carlino is a clinical senior lecturer at the Sydney Medical School, University of Sydney and a medical oncologist at Westmead Hospital and Blacktown Hospital, Australia. Dr. Georgina Long is an associate professor and medical oncologist at Melanoma Institute Australia, University of Sydney. These remarks were part of an editorial accompanying the report (Ann Oncol. 2015 Sep 15. doi: 10.1093/annonc/mdv361). Dr. Carlino and Dr. Long reported having no disclosures.
In chemotherapy-naive patients with metastatic melanoma, progression-free survival (PFS) was significantly longer with nab-paclitaxel, compared with dacarbazine, according to results from a phase III trial.
Median PFS for the nab-paclitaxel arm vs. the dacarbazine arm was 4.8 vs. 2.5 months, respectively (hazard ratio, 0.792; 95% confidence interval, 0.631-0.992; P = .044). Overall survival was similar in the two arms: 12.6 months for nab-paclitaxel vs. 10.5 months for dacarbazine (P = .27). The nab-paclitaxel group had an increased, but not significant, overall response rate (15% vs. 11%) and a significantly improved disease control rate (P = .004). The disease control rate reflects the number of complete responses plus partial responses plus stable disease for 16 or more weeks.
Nab-paclitaxel benefited patients regardless of BRAF mutation status, noted Dr. Evan Hersh, professor of medicine at the University of Arizona, Tucson, and colleagues.
“Additionally, in a post hoc analysis of this trial, nab-paclitaxel was shown to benefit a subgroup of patients with low or absent TILs [tumor-infiltrating lymphocytes], a poor prognostic factor in melanoma,” they wrote (Ann Oncol 2015 Sep 26. doi: 10.1093/annonc/mdv324).
The most common treatment-related adverse events of grade 3 or greater were neuropathy (25% vs. 0%), neutropenia (20% vs. 10%), and leukopenia (12% vs. 7%) for the nab-paclitaxel and dacarbazine arms, respectively. The median onset of grade 3 or greater peripheral neuropathy was 101 days after treatment began.
Between April 2009 and June 2011, the multicenter, phase III, randomized, controlled trial enrolled 529 adults with stage IV malignant melanoma who had no prior cytotoxic therapy.
A majority of participants from both treatment arms underwent post-study therapy, including newer agents such as BRAF inhibitors and ipilimumab. This may account for the diminished treatment affect of nab-paclitaxel on overall survival, compared with the more significant benefit in PFS observed early in the study.
A biomarker analysis found no correlation between tumor expression of the albumin-binding protein SPARC and PFS with nab-paclitaxel treatment. SPARC was hypothesized to enrich nab-paclitaxel in the tumor microenvironment thereby increasing its efficacy.
In chemotherapy-naive patients with metastatic melanoma, progression-free survival (PFS) was significantly longer with nab-paclitaxel, compared with dacarbazine, according to results from a phase III trial.
Median PFS for the nab-paclitaxel arm vs. the dacarbazine arm was 4.8 vs. 2.5 months, respectively (hazard ratio, 0.792; 95% confidence interval, 0.631-0.992; P = .044). Overall survival was similar in the two arms: 12.6 months for nab-paclitaxel vs. 10.5 months for dacarbazine (P = .27). The nab-paclitaxel group had an increased, but not significant, overall response rate (15% vs. 11%) and a significantly improved disease control rate (P = .004). The disease control rate reflects the number of complete responses plus partial responses plus stable disease for 16 or more weeks.
Nab-paclitaxel benefited patients regardless of BRAF mutation status, noted Dr. Evan Hersh, professor of medicine at the University of Arizona, Tucson, and colleagues.
“Additionally, in a post hoc analysis of this trial, nab-paclitaxel was shown to benefit a subgroup of patients with low or absent TILs [tumor-infiltrating lymphocytes], a poor prognostic factor in melanoma,” they wrote (Ann Oncol 2015 Sep 26. doi: 10.1093/annonc/mdv324).
The most common treatment-related adverse events of grade 3 or greater were neuropathy (25% vs. 0%), neutropenia (20% vs. 10%), and leukopenia (12% vs. 7%) for the nab-paclitaxel and dacarbazine arms, respectively. The median onset of grade 3 or greater peripheral neuropathy was 101 days after treatment began.
Between April 2009 and June 2011, the multicenter, phase III, randomized, controlled trial enrolled 529 adults with stage IV malignant melanoma who had no prior cytotoxic therapy.
A majority of participants from both treatment arms underwent post-study therapy, including newer agents such as BRAF inhibitors and ipilimumab. This may account for the diminished treatment affect of nab-paclitaxel on overall survival, compared with the more significant benefit in PFS observed early in the study.
A biomarker analysis found no correlation between tumor expression of the albumin-binding protein SPARC and PFS with nab-paclitaxel treatment. SPARC was hypothesized to enrich nab-paclitaxel in the tumor microenvironment thereby increasing its efficacy.
FROM ANNALS OF ONCOLOGY
Key clinical point: Nab-pacilitaxel had significantly longer PFS than dacarbazine in chemotherapy-naive patients with metastatic melanoma.
Major finding: Median PFS in the nab-paclitaxel vs. dacarbazine arms was 4.8 vs. 2.5 months, respectively (HR, 0.792; 95% CI, 0.631-0.992; P = .044).
Data source: The phase III, randomized, controlled trial enrolled 529 patients between April 2009 and June 2011.
Disclosures: Dr. Hersh reported research funding from Celgene. His coauthors reported financial ties to several industry sources.
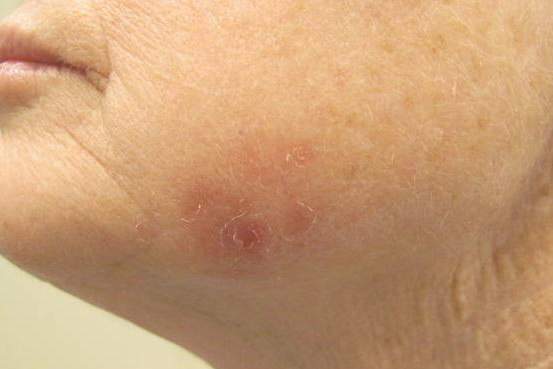
Erythematous Scaly Patch on the Jawline
The Diagnosis: Amelanotic Melanoma In Situ
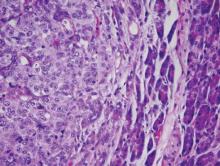
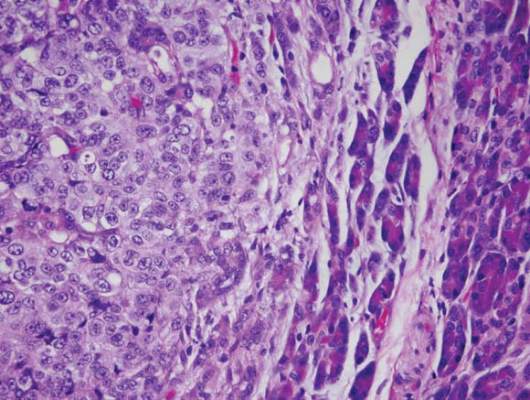
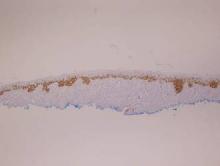
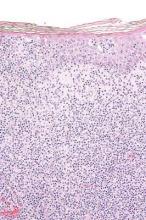
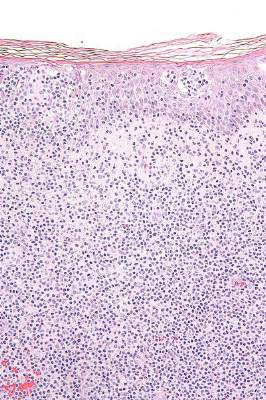
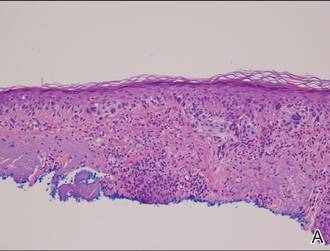
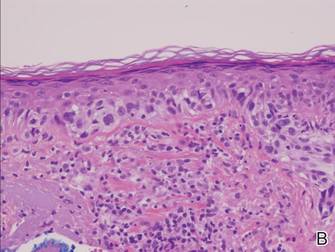
Histopathology revealed a broad asymmetric melanocytic proliferation at the dermoepidermal junction, consisting both of singly dispersed cells as well as randomly positioned nests (Figure 1). The single cells demonstrated junctional confluence and extension along adnexal structures highlighted by melan-A stain (Figure 2). The melanocytes were markedly atypical with enlarged and hyperchromatic nuclei containing multiple nucleoli. No dermal involvement was seen. There was papillary dermal fibrosis and an active host lymphocytic response. Based on these findings, a diagnosis of amelanotic melanoma in situ was made.
|
Figure 1. Histopathology revealed confluence of atypical melanocytes at the dermoepidermal junction and pagetoid scatter of melanocytes to the spinous layer (A)(H&E, original magnification ×4). Higher-power magnification highlighted the atypia of the individual melanocytes (B)(H&E, original magnification ×10). |
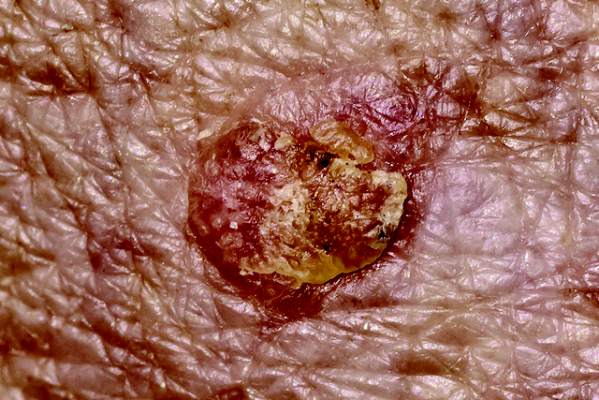
Subsequent scouting punch biopsies at the superior, anterior, and posterior aspects of the lesion were performed (Figure 3). All 3 revealed a similar nested and single cell proliferation at the dermoepidermal junction, confirming residual amelanotic melanoma in situ. The patient was referred to the otolaryngology department and underwent wide local excision with 5-mm margins and reconstructive repair.
Amelanotic melanoma comprises 2% to 8% of cutaneous melanomas. It is more common in fair-skinned elderly women with an average age of diagnosis of 61.8 years. Because features typically associated with melanoma such as asymmetry, border irregularity, and color variegation often are absent, amelanotic melanoma represents a notable diagnostic challenge for clinicians. Lesions can present nonspecifically as erythematous macules, papules, patches, or plaques and can have associated pruritus and scale.1,2
Clinical misdiagnoses for amelanotic melanoma include Bowen disease, basal cell carcinoma, actinic keratosis, lichenoid keratosis, intradermal nevus, dermatofibroma, inflamed seborrheic keratosis, nummular dermatitis, pyogenic granuloma, and granuloma annulare.1-6 There have been few case reports of amelanotic melanoma in situ, with most being the lentigo maligna variant that were initially clinically diagnosed as superficial basal cell carcinoma, Bowen disease, or dermatitis.7,8 In one case report, an amelanotic lentigo maligna was incidentally discovered after performing a mapping shave biopsy on what was normal-appearing skin.9
Dermoscopic evidence of vascular structures in lesions, including the presence of dotted vessels, milky red areas, and/or serpentine (linear irregular) vessels, may be the only clues to suggest amelanotic melanoma before biopsy. However, these findings are nonspecific and can be seen in other benign and malignant skin conditions.2
Complete surgical excision is the standard treatment of amelanotic melanoma in situ given its potential for invasion. However, the lack of pigment can make margins difficult to define. Because of its ability to detect disease beyond visual margins, Mohs micrographic surgery may have better cure rates than conventional excision.4 Prognosis for amelanotic melanoma is the same as other melanomas of equal thickness and location, though delay in diagnosis can adversely affect outcomes. Furthermore, amelanotic melanoma in situ can rapidly progress to invasive melanoma.3,5 Thus it is important to maintain clinical suspicion for amelanotic melanoma in fair-skinned elderly women presenting with a persistent or recurring erythematous scaly lesion on sun-exposed skin.
- Rahbari H, Nabai H, Mehregan AH, et al. Amelanotic lentigo maligna melanoma: a diagnostic conundrum— presentation of four new cases. Cancer. 1996;77:2052-2057.
- Jaimes N, Braun RP, Thomas L, et al. Clinical and dermoscopic characteristics of amelanotic melanomas that are not of the nodular subtype. J Eur Acad Dermatol Venereol. 2012;26:591-596.
- Koch SE, Lange JR. Amelanotic melanoma: the great masquerader. J Am Acad Dermatol. 2000;42:731-734.
- Conrad N, Jackson B, Goldberg L. Amelanotic lentigo maligna melanoma: a unique case presentation. Dermatol Surg. 1999;25:408-411.
- Cliff S, Otter M, Holden CA. Amelanotic lentigo maligna melanoma of the face: a case report and review of the literature. Clin Exp Dermatol. 1997;22:177-179.
- Dalton SR, Fillman EP, Altman CE, et al. Atypical junctional melanocytic proliferations in benign lichenoid keratosis. Hum Pathol. 2003;34:706-709.
- Paver K, Stewart M, Kossard S, et al. Amelanotic lentigo maligna. Australas J Dermatol. 1981;22:106-108.
- Lewis JE. Lentigo maligna presenting as an eczematous lesion. Cutis. 1987;40:357-359.
- Perera E, Mellick N, Teng P, et al. A clinically invisible melanoma. Australas J Dermatol. 2014;55:e58-e59.
The Diagnosis: Amelanotic Melanoma In Situ
Histopathology revealed a broad asymmetric melanocytic proliferation at the dermoepidermal junction, consisting both of singly dispersed cells as well as randomly positioned nests (Figure 1). The single cells demonstrated junctional confluence and extension along adnexal structures highlighted by melan-A stain (Figure 2). The melanocytes were markedly atypical with enlarged and hyperchromatic nuclei containing multiple nucleoli. No dermal involvement was seen. There was papillary dermal fibrosis and an active host lymphocytic response. Based on these findings, a diagnosis of amelanotic melanoma in situ was made.


|
Figure 1. Histopathology revealed confluence of atypical melanocytes at the dermoepidermal junction and pagetoid scatter of melanocytes to the spinous layer (A)(H&E, original magnification ×4). Higher-power magnification highlighted the atypia of the individual melanocytes (B)(H&E, original magnification ×10). |
Subsequent scouting punch biopsies at the superior, anterior, and posterior aspects of the lesion were performed (Figure 3). All 3 revealed a similar nested and single cell proliferation at the dermoepidermal junction, confirming residual amelanotic melanoma in situ. The patient was referred to the otolaryngology department and underwent wide local excision with 5-mm margins and reconstructive repair.
Amelanotic melanoma comprises 2% to 8% of cutaneous melanomas. It is more common in fair-skinned elderly women with an average age of diagnosis of 61.8 years. Because features typically associated with melanoma such as asymmetry, border irregularity, and color variegation often are absent, amelanotic melanoma represents a notable diagnostic challenge for clinicians. Lesions can present nonspecifically as erythematous macules, papules, patches, or plaques and can have associated pruritus and scale.1,2
Clinical misdiagnoses for amelanotic melanoma include Bowen disease, basal cell carcinoma, actinic keratosis, lichenoid keratosis, intradermal nevus, dermatofibroma, inflamed seborrheic keratosis, nummular dermatitis, pyogenic granuloma, and granuloma annulare.1-6 There have been few case reports of amelanotic melanoma in situ, with most being the lentigo maligna variant that were initially clinically diagnosed as superficial basal cell carcinoma, Bowen disease, or dermatitis.7,8 In one case report, an amelanotic lentigo maligna was incidentally discovered after performing a mapping shave biopsy on what was normal-appearing skin.9
Dermoscopic evidence of vascular structures in lesions, including the presence of dotted vessels, milky red areas, and/or serpentine (linear irregular) vessels, may be the only clues to suggest amelanotic melanoma before biopsy. However, these findings are nonspecific and can be seen in other benign and malignant skin conditions.2
Complete surgical excision is the standard treatment of amelanotic melanoma in situ given its potential for invasion. However, the lack of pigment can make margins difficult to define. Because of its ability to detect disease beyond visual margins, Mohs micrographic surgery may have better cure rates than conventional excision.4 Prognosis for amelanotic melanoma is the same as other melanomas of equal thickness and location, though delay in diagnosis can adversely affect outcomes. Furthermore, amelanotic melanoma in situ can rapidly progress to invasive melanoma.3,5 Thus it is important to maintain clinical suspicion for amelanotic melanoma in fair-skinned elderly women presenting with a persistent or recurring erythematous scaly lesion on sun-exposed skin.
The Diagnosis: Amelanotic Melanoma In Situ
Histopathology revealed a broad asymmetric melanocytic proliferation at the dermoepidermal junction, consisting both of singly dispersed cells as well as randomly positioned nests (Figure 1). The single cells demonstrated junctional confluence and extension along adnexal structures highlighted by melan-A stain (Figure 2). The melanocytes were markedly atypical with enlarged and hyperchromatic nuclei containing multiple nucleoli. No dermal involvement was seen. There was papillary dermal fibrosis and an active host lymphocytic response. Based on these findings, a diagnosis of amelanotic melanoma in situ was made.


|
Figure 1. Histopathology revealed confluence of atypical melanocytes at the dermoepidermal junction and pagetoid scatter of melanocytes to the spinous layer (A)(H&E, original magnification ×4). Higher-power magnification highlighted the atypia of the individual melanocytes (B)(H&E, original magnification ×10). |
Subsequent scouting punch biopsies at the superior, anterior, and posterior aspects of the lesion were performed (Figure 3). All 3 revealed a similar nested and single cell proliferation at the dermoepidermal junction, confirming residual amelanotic melanoma in situ. The patient was referred to the otolaryngology department and underwent wide local excision with 5-mm margins and reconstructive repair.
Amelanotic melanoma comprises 2% to 8% of cutaneous melanomas. It is more common in fair-skinned elderly women with an average age of diagnosis of 61.8 years. Because features typically associated with melanoma such as asymmetry, border irregularity, and color variegation often are absent, amelanotic melanoma represents a notable diagnostic challenge for clinicians. Lesions can present nonspecifically as erythematous macules, papules, patches, or plaques and can have associated pruritus and scale.1,2
Clinical misdiagnoses for amelanotic melanoma include Bowen disease, basal cell carcinoma, actinic keratosis, lichenoid keratosis, intradermal nevus, dermatofibroma, inflamed seborrheic keratosis, nummular dermatitis, pyogenic granuloma, and granuloma annulare.1-6 There have been few case reports of amelanotic melanoma in situ, with most being the lentigo maligna variant that were initially clinically diagnosed as superficial basal cell carcinoma, Bowen disease, or dermatitis.7,8 In one case report, an amelanotic lentigo maligna was incidentally discovered after performing a mapping shave biopsy on what was normal-appearing skin.9
Dermoscopic evidence of vascular structures in lesions, including the presence of dotted vessels, milky red areas, and/or serpentine (linear irregular) vessels, may be the only clues to suggest amelanotic melanoma before biopsy. However, these findings are nonspecific and can be seen in other benign and malignant skin conditions.2
Complete surgical excision is the standard treatment of amelanotic melanoma in situ given its potential for invasion. However, the lack of pigment can make margins difficult to define. Because of its ability to detect disease beyond visual margins, Mohs micrographic surgery may have better cure rates than conventional excision.4 Prognosis for amelanotic melanoma is the same as other melanomas of equal thickness and location, though delay in diagnosis can adversely affect outcomes. Furthermore, amelanotic melanoma in situ can rapidly progress to invasive melanoma.3,5 Thus it is important to maintain clinical suspicion for amelanotic melanoma in fair-skinned elderly women presenting with a persistent or recurring erythematous scaly lesion on sun-exposed skin.
- Rahbari H, Nabai H, Mehregan AH, et al. Amelanotic lentigo maligna melanoma: a diagnostic conundrum— presentation of four new cases. Cancer. 1996;77:2052-2057.
- Jaimes N, Braun RP, Thomas L, et al. Clinical and dermoscopic characteristics of amelanotic melanomas that are not of the nodular subtype. J Eur Acad Dermatol Venereol. 2012;26:591-596.
- Koch SE, Lange JR. Amelanotic melanoma: the great masquerader. J Am Acad Dermatol. 2000;42:731-734.
- Conrad N, Jackson B, Goldberg L. Amelanotic lentigo maligna melanoma: a unique case presentation. Dermatol Surg. 1999;25:408-411.
- Cliff S, Otter M, Holden CA. Amelanotic lentigo maligna melanoma of the face: a case report and review of the literature. Clin Exp Dermatol. 1997;22:177-179.
- Dalton SR, Fillman EP, Altman CE, et al. Atypical junctional melanocytic proliferations in benign lichenoid keratosis. Hum Pathol. 2003;34:706-709.
- Paver K, Stewart M, Kossard S, et al. Amelanotic lentigo maligna. Australas J Dermatol. 1981;22:106-108.
- Lewis JE. Lentigo maligna presenting as an eczematous lesion. Cutis. 1987;40:357-359.
- Perera E, Mellick N, Teng P, et al. A clinically invisible melanoma. Australas J Dermatol. 2014;55:e58-e59.
- Rahbari H, Nabai H, Mehregan AH, et al. Amelanotic lentigo maligna melanoma: a diagnostic conundrum— presentation of four new cases. Cancer. 1996;77:2052-2057.
- Jaimes N, Braun RP, Thomas L, et al. Clinical and dermoscopic characteristics of amelanotic melanomas that are not of the nodular subtype. J Eur Acad Dermatol Venereol. 2012;26:591-596.
- Koch SE, Lange JR. Amelanotic melanoma: the great masquerader. J Am Acad Dermatol. 2000;42:731-734.
- Conrad N, Jackson B, Goldberg L. Amelanotic lentigo maligna melanoma: a unique case presentation. Dermatol Surg. 1999;25:408-411.
- Cliff S, Otter M, Holden CA. Amelanotic lentigo maligna melanoma of the face: a case report and review of the literature. Clin Exp Dermatol. 1997;22:177-179.
- Dalton SR, Fillman EP, Altman CE, et al. Atypical junctional melanocytic proliferations in benign lichenoid keratosis. Hum Pathol. 2003;34:706-709.
- Paver K, Stewart M, Kossard S, et al. Amelanotic lentigo maligna. Australas J Dermatol. 1981;22:106-108.
- Lewis JE. Lentigo maligna presenting as an eczematous lesion. Cutis. 1987;40:357-359.
- Perera E, Mellick N, Teng P, et al. A clinically invisible melanoma. Australas J Dermatol. 2014;55:e58-e59.

Atypia of the individual melanocytes.
Melan-A stain highlighted the density and confluence of melanocytes within the epidermis.
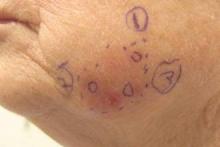
Three scouting punch biopsies were performed along the periphery of the lesion.
A 70-year-old white woman with a history of basal cell carcinoma presented with a 2.7×1.9-cm ill-defined, erythematous, scaly patch along the left side of the jawline. Ten months prior to presentation, the lesion appeared as a grayish macule that was clinically diagnosed as a pigmented actinic keratosis and was treated with cryotherapy with resolution noted at 6-month follow-up. Differential diagnosis of the current lesion included actinic keratosis, lichenoid keratosis, and superficial basal cell carcinoma. A shave biopsy was performed.
Children with multiple congenital melanocytic nevi should have CNS MRI
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
Children with multiple congenital melanocytic nevi should have CNS MRI
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
For children born with multiple congenital melanocytic nevi, a single MRI of the central nervous system within the first year of life was an effective way to screen for future outcomes, according to Dr. Regula Waelchli of Great Ormond Street Hospital for Children, London, and her associates.
Of the 271 children screened, 21% had an abnormal CNS MRI. An abnormal MRI increased the odds of negative future outcomes significantly. The most likely outcome was requirement for neurosurgery with an odds ratio of 71, followed by seizure with an OR of 13.4, and neurodevelopmental problems with an OR of 3.
The most common abnormality was intraparenchymal melanosis, present in most of the children with abnormal MRIs. However, if the only abnormality present was intraparenchymal melanosis, as it was for more than half of the abnormal group, no routine follow-up MRI was necessary and neurosurgery wasn’t required.
In patients with normal MRIs, seizures were temporary and/or easy to control with a single medication, and neurodevelopmental issues were mild, the investigators found.
While only those patients with an abnormality other than intraparenchymal melanosis require repeat MRIs, “any change in neurological status at any age should always trigger a repeat MRI, independent of the initial MRI findings,” Dr. Waelchli and her associates concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.13898).
FDA approves nivolumab-ipilimumab combination for melanoma
Nivolumab, a programmed death receptor-1 (PD-1)–blocking antibody, has been approved for use in combination with ipilimumab for treating people with BRAF V600 wild-type, unresectable or metastatic melanoma.
Approval was based on demonstration of an increase in the objective response rate, prolonged response durations, and improvement in progression-free survival (PFS) in an international, multicenter, double-blind, randomized, two-arm, active-controlled trial in patients who were previously untreated for unresectable or metastatic, BRAF V600 wild-type melanoma, according to a Sept. 30 statement issued by the Food and Drug Administration.
Nivolumab, marketed as Opdivo Injection by Bristol-Myers Squibb, was approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab therapy and, if BRAF V600 mutation positive, a BRAF inhibitor. Ipilimumab, approved in 2011 and marketed as Yervoy by BMS, is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4)–blocking antibody indicated for treating unresectable or metastatic melanoma.
“Combined nivolumab (anti–PD-1) and ipilimumab (anti–CTLA-4) mediated inhibition results in enhanced T-cell function that is greater than the effects of either antibody alone, and results in improved antitumor responses in metastatic melanoma,” the updated nivolumab prescribing information states.
This is the first FDA approval of a cancer treatment regimen that includes two immuno-oncology agents, according to the company’s statement announcing the approval.
The phase II CheckMate 069 study enrolled 142 patients, including 109 patients with BRAF V600 wild-type melanoma, randomized in a 2:1 ratio to receive the combination or ipilimumab plus placebo; their median age was 66 years; 84% had an ECOG performance score of 0, and 15% had a score of 1. The overall response rate was 60% among those on the combination therapy vs. 11% among those on ipilimumab alone – a 49% improvement (P less than .001), according to the FDA.
Nine of the 43 patients with an objective response (ranging from 3 to 7 months) in the combination therapy group “have progressed after response, died, or received subsequent therapy,” the FDA said. Among the 34 patients who continued to have a response at the last analysis, 14 had responses that had lasted from at least 6 months to less than 9 months. The remaining 20 patients had responses that had lasted at least 9 months.
The estimated median PFS was 8.9 months in the combination group vs. 4.7 months in the ipilimumab-only arm, a 60% reduced risk (P less than .002)
Serious adverse reactions, adverse reactions resulting in permanent discontinuation or a delayed dose, and grade 3 or 4 adverse reaction were higher among those treated with the combination. Colitis, diarrhea, pyrexia, and pneumonitis were the most common serious adverse reactions among those on the combination.
The recommended dose and schedule is as follows, according to the FDA: 1 mg/kg of nivolumab administered as an IV infusion over 60 minutes, followed by ipilimumab on the same day, every 3 weeks for four doses. “The recommended subsequent dose of nivolumab, as a single agent, is 3 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.”
Both indications for nivolumab were approved as accelerated approvals, based on the “tumor response rate and durability of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in the confirmatory trials,” according to the prescribing information.
Serious adverse events associated with this therapy should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch.
Nivolumab, a programmed death receptor-1 (PD-1)–blocking antibody, has been approved for use in combination with ipilimumab for treating people with BRAF V600 wild-type, unresectable or metastatic melanoma.
Approval was based on demonstration of an increase in the objective response rate, prolonged response durations, and improvement in progression-free survival (PFS) in an international, multicenter, double-blind, randomized, two-arm, active-controlled trial in patients who were previously untreated for unresectable or metastatic, BRAF V600 wild-type melanoma, according to a Sept. 30 statement issued by the Food and Drug Administration.
Nivolumab, marketed as Opdivo Injection by Bristol-Myers Squibb, was approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab therapy and, if BRAF V600 mutation positive, a BRAF inhibitor. Ipilimumab, approved in 2011 and marketed as Yervoy by BMS, is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4)–blocking antibody indicated for treating unresectable or metastatic melanoma.
“Combined nivolumab (anti–PD-1) and ipilimumab (anti–CTLA-4) mediated inhibition results in enhanced T-cell function that is greater than the effects of either antibody alone, and results in improved antitumor responses in metastatic melanoma,” the updated nivolumab prescribing information states.
This is the first FDA approval of a cancer treatment regimen that includes two immuno-oncology agents, according to the company’s statement announcing the approval.
The phase II CheckMate 069 study enrolled 142 patients, including 109 patients with BRAF V600 wild-type melanoma, randomized in a 2:1 ratio to receive the combination or ipilimumab plus placebo; their median age was 66 years; 84% had an ECOG performance score of 0, and 15% had a score of 1. The overall response rate was 60% among those on the combination therapy vs. 11% among those on ipilimumab alone – a 49% improvement (P less than .001), according to the FDA.
Nine of the 43 patients with an objective response (ranging from 3 to 7 months) in the combination therapy group “have progressed after response, died, or received subsequent therapy,” the FDA said. Among the 34 patients who continued to have a response at the last analysis, 14 had responses that had lasted from at least 6 months to less than 9 months. The remaining 20 patients had responses that had lasted at least 9 months.
The estimated median PFS was 8.9 months in the combination group vs. 4.7 months in the ipilimumab-only arm, a 60% reduced risk (P less than .002)
Serious adverse reactions, adverse reactions resulting in permanent discontinuation or a delayed dose, and grade 3 or 4 adverse reaction were higher among those treated with the combination. Colitis, diarrhea, pyrexia, and pneumonitis were the most common serious adverse reactions among those on the combination.
The recommended dose and schedule is as follows, according to the FDA: 1 mg/kg of nivolumab administered as an IV infusion over 60 minutes, followed by ipilimumab on the same day, every 3 weeks for four doses. “The recommended subsequent dose of nivolumab, as a single agent, is 3 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.”
Both indications for nivolumab were approved as accelerated approvals, based on the “tumor response rate and durability of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in the confirmatory trials,” according to the prescribing information.
Serious adverse events associated with this therapy should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch.
Nivolumab, a programmed death receptor-1 (PD-1)–blocking antibody, has been approved for use in combination with ipilimumab for treating people with BRAF V600 wild-type, unresectable or metastatic melanoma.
Approval was based on demonstration of an increase in the objective response rate, prolonged response durations, and improvement in progression-free survival (PFS) in an international, multicenter, double-blind, randomized, two-arm, active-controlled trial in patients who were previously untreated for unresectable or metastatic, BRAF V600 wild-type melanoma, according to a Sept. 30 statement issued by the Food and Drug Administration.
Nivolumab, marketed as Opdivo Injection by Bristol-Myers Squibb, was approved in 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab therapy and, if BRAF V600 mutation positive, a BRAF inhibitor. Ipilimumab, approved in 2011 and marketed as Yervoy by BMS, is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4)–blocking antibody indicated for treating unresectable or metastatic melanoma.
“Combined nivolumab (anti–PD-1) and ipilimumab (anti–CTLA-4) mediated inhibition results in enhanced T-cell function that is greater than the effects of either antibody alone, and results in improved antitumor responses in metastatic melanoma,” the updated nivolumab prescribing information states.
This is the first FDA approval of a cancer treatment regimen that includes two immuno-oncology agents, according to the company’s statement announcing the approval.
The phase II CheckMate 069 study enrolled 142 patients, including 109 patients with BRAF V600 wild-type melanoma, randomized in a 2:1 ratio to receive the combination or ipilimumab plus placebo; their median age was 66 years; 84% had an ECOG performance score of 0, and 15% had a score of 1. The overall response rate was 60% among those on the combination therapy vs. 11% among those on ipilimumab alone – a 49% improvement (P less than .001), according to the FDA.
Nine of the 43 patients with an objective response (ranging from 3 to 7 months) in the combination therapy group “have progressed after response, died, or received subsequent therapy,” the FDA said. Among the 34 patients who continued to have a response at the last analysis, 14 had responses that had lasted from at least 6 months to less than 9 months. The remaining 20 patients had responses that had lasted at least 9 months.
The estimated median PFS was 8.9 months in the combination group vs. 4.7 months in the ipilimumab-only arm, a 60% reduced risk (P less than .002)
Serious adverse reactions, adverse reactions resulting in permanent discontinuation or a delayed dose, and grade 3 or 4 adverse reaction were higher among those treated with the combination. Colitis, diarrhea, pyrexia, and pneumonitis were the most common serious adverse reactions among those on the combination.
The recommended dose and schedule is as follows, according to the FDA: 1 mg/kg of nivolumab administered as an IV infusion over 60 minutes, followed by ipilimumab on the same day, every 3 weeks for four doses. “The recommended subsequent dose of nivolumab, as a single agent, is 3 mg/kg as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity.”
Both indications for nivolumab were approved as accelerated approvals, based on the “tumor response rate and durability of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in the confirmatory trials,” according to the prescribing information.
Serious adverse events associated with this therapy should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/medwatch.
Topical fluorouracil shows long-term benefit for actinic keratoses
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
FROM JAMA DERMATOLOGY
Key clinical point:One course of topical fluorouracil cream, 5%, decreased the need for localized treatment and the number of actinic keratoses long term.
Major finding: Participants whose AKs were treated with a course of fluorouracil cream, 5%, had significantly fewer AKs and required fewer treatments, compared with the control group, for over 2 years.
Data source: The randomized, double-blind study compared the effect of a course of topical fluorouracil cream with a vehicle cream on the number of AKs and other measures, over a mean follow-up of 2.6 years, in 932 patients treated at 12 VA dermatology clinics.
Disclosures: The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others had no disclosures.
Topical resiquimod effective for early-stage cutaneous T-cell lymphoma
Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL), in some cases inducing regression in both treated and untreated lesions, according to researchers.
The mean number of prior unsuccessful therapies among the patients was 6, yet the majority of patients (11 of 12) experienced significant improvement, and 2 patients had complete clinical responses with no evidence of disease after treatment. One patient, despite a 15-year history of disease and 11 unsuccessful treatments, experienced a complete resolution of both treated and untreated skin lesions.
The open-label, phase I trial evaluated 12 patients with early-stage CTCL. Patients experienced minor adverse effects (all grade 1), which were primarily skin irritation. The trial evaluated 0.03% and 0.06% resiquimod, with complete and more rapid responses occurring at the higher dose. Both doses were equally well tolerated.
“These studies support further trials of this medication in early-stage, skin-limited CTCL and suggest resiquimod might also be useful as an adjuvant therapy in the treatment of more advanced CTCL,” wrote Dr. Alain Rook of the Department of Dermatology and the Center for Clinical Biostatistics and Epidemiology, Perelman School of Medicine, Philadelphia, and colleagues.
Arising from T cells that traffic to the skin, CTCLs are non-Hodgkin lymphomas whose only potential cure is stem cell transplantation. Studies suggest that host antitumor immunity plays an important role in the disease, and in this study, high responders showed recruitment and expansion of benign T-cell clones and activation of T cells and natural killer cells in the skin.
In the absence of cell-surface markers to distinguish malignant from benign T cells in the lesion, the team used high throughput screening of the T-cell receptor–beta gene to quantify malignant cells and monitor response to therapy. Of the 10 patients with identified malignant cells, biopsied lesions showed that most had reduction of malignant T-cell clones and 3 had complete eradication. The results may not reflect responses in nonbiopsied lesions.
Resiquimod recruits T cells and other immune cells to the skin, causing inflammation that the researchers observed persisted after complete or nearly complete malignant T-cell eradication. Study results suggested that activation of CD4+ cells and expansion of tumor-specific T cells is critical for effectiveness of resiquimod.
Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL), in some cases inducing regression in both treated and untreated lesions, according to researchers.
The mean number of prior unsuccessful therapies among the patients was 6, yet the majority of patients (11 of 12) experienced significant improvement, and 2 patients had complete clinical responses with no evidence of disease after treatment. One patient, despite a 15-year history of disease and 11 unsuccessful treatments, experienced a complete resolution of both treated and untreated skin lesions.
The open-label, phase I trial evaluated 12 patients with early-stage CTCL. Patients experienced minor adverse effects (all grade 1), which were primarily skin irritation. The trial evaluated 0.03% and 0.06% resiquimod, with complete and more rapid responses occurring at the higher dose. Both doses were equally well tolerated.
“These studies support further trials of this medication in early-stage, skin-limited CTCL and suggest resiquimod might also be useful as an adjuvant therapy in the treatment of more advanced CTCL,” wrote Dr. Alain Rook of the Department of Dermatology and the Center for Clinical Biostatistics and Epidemiology, Perelman School of Medicine, Philadelphia, and colleagues.
Arising from T cells that traffic to the skin, CTCLs are non-Hodgkin lymphomas whose only potential cure is stem cell transplantation. Studies suggest that host antitumor immunity plays an important role in the disease, and in this study, high responders showed recruitment and expansion of benign T-cell clones and activation of T cells and natural killer cells in the skin.
In the absence of cell-surface markers to distinguish malignant from benign T cells in the lesion, the team used high throughput screening of the T-cell receptor–beta gene to quantify malignant cells and monitor response to therapy. Of the 10 patients with identified malignant cells, biopsied lesions showed that most had reduction of malignant T-cell clones and 3 had complete eradication. The results may not reflect responses in nonbiopsied lesions.
Resiquimod recruits T cells and other immune cells to the skin, causing inflammation that the researchers observed persisted after complete or nearly complete malignant T-cell eradication. Study results suggested that activation of CD4+ cells and expansion of tumor-specific T cells is critical for effectiveness of resiquimod.
Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL), in some cases inducing regression in both treated and untreated lesions, according to researchers.
The mean number of prior unsuccessful therapies among the patients was 6, yet the majority of patients (11 of 12) experienced significant improvement, and 2 patients had complete clinical responses with no evidence of disease after treatment. One patient, despite a 15-year history of disease and 11 unsuccessful treatments, experienced a complete resolution of both treated and untreated skin lesions.
The open-label, phase I trial evaluated 12 patients with early-stage CTCL. Patients experienced minor adverse effects (all grade 1), which were primarily skin irritation. The trial evaluated 0.03% and 0.06% resiquimod, with complete and more rapid responses occurring at the higher dose. Both doses were equally well tolerated.
“These studies support further trials of this medication in early-stage, skin-limited CTCL and suggest resiquimod might also be useful as an adjuvant therapy in the treatment of more advanced CTCL,” wrote Dr. Alain Rook of the Department of Dermatology and the Center for Clinical Biostatistics and Epidemiology, Perelman School of Medicine, Philadelphia, and colleagues.
Arising from T cells that traffic to the skin, CTCLs are non-Hodgkin lymphomas whose only potential cure is stem cell transplantation. Studies suggest that host antitumor immunity plays an important role in the disease, and in this study, high responders showed recruitment and expansion of benign T-cell clones and activation of T cells and natural killer cells in the skin.
In the absence of cell-surface markers to distinguish malignant from benign T cells in the lesion, the team used high throughput screening of the T-cell receptor–beta gene to quantify malignant cells and monitor response to therapy. Of the 10 patients with identified malignant cells, biopsied lesions showed that most had reduction of malignant T-cell clones and 3 had complete eradication. The results may not reflect responses in nonbiopsied lesions.
Resiquimod recruits T cells and other immune cells to the skin, causing inflammation that the researchers observed persisted after complete or nearly complete malignant T-cell eradication. Study results suggested that activation of CD4+ cells and expansion of tumor-specific T cells is critical for effectiveness of resiquimod.
FROM BLOOD
Key clinical point: Topical resiquimod was effective and well tolerated in patients with early-stage cutaneous T-cell lymphoma (CTCL).
Major finding: In total, 11 of 12 patients had significant improvement: 2 had resolution of all evidence of disease, and 9 experienced improvement greater than or equal to 50%.
Data source: The open-label, phase I trial evaluated 12 patients with early stage CTCL.
Disclosures: Dr. Rook and one coauthor have patents pending on HTS in cutaneous lymphoma. His coauthor is employed by Adaptive Biotechnologies.
Natalizumab-Associated Melanoma: A Report of 139 Cases From the Southern Network on Adverse Reactions (SONAR)
Background: Natalizumab is an effective immunosuppressive therapy for multiple sclerosis (MS) that received its initial FDA approval in 2004. Its most notable toxicity is progressive multifocal leukoencephalopathy (PML), an opportunistic infection that is the focus of an FDA-mandated registry, TOUCH. The Southern Network on Adverse Reactions identified a fatal case of natalizumab-associated urethral melanoma and undertook an extensive evaluation of all cases of natalizumab-associated melanoma included in the FDA’s Adverse Event Reporting System (FAERS) (between 2005 and 2014).
Methods: Patient characteristics were determined from the FAERS reports and put into a comprehensive dataset. The quality of these reports was assessed based on a 15-point scale of various clinical, demographic, and pharmacy components. Cases were identified as being reported through the TOUCH system, using TOUCH information but reporting outside the system, not using TOUCH at all in the U.S. or not U.S. Quality scores and their components within the 4 Touch groups were statistically compared across all attributes using the mathematical programming-based statistical methodology univariate optimal discriminant analysis.
Results: The mean patient age at the time of diagnosis of melanoma was 46 years (SD 11). Seventeen patients were diagnosed with cutaneous melanoma developing in nonsun-exposed areas. We found that cases reported through the TOUCH registry were of lower quality (mean score 7.7) compared with others that reported outside of the U.S. (mean score 8.5, P < .008). Those cases reported through the TOUCH system were less likely to report the site of melanoma (P < .019) and have overall lower clinical scores (P < .04) compared with reports using TOUCH information but reporting outside of the system.
Conclusions: Our findings suggest that in the U.S., the TOUCH Registry should be expanded to require clinicians to report details of natalizumab-associated melanoma, an opportunistic illness that frequently develops in immunocompromised persons. Also, the FDA-approved product label for natalizumab should be revised to include information on occurrences of melanoma among natalizumab-treated MS patients, particularly those who have cutaneous nevi prior to natalizumab initiation. Natalizumab-treated MS patients and their physicians should be vigilant for changes in nevi appearances and development of new cutaneous lesions (particularly in nonsun-exposed cutaneous areas).
Background: Natalizumab is an effective immunosuppressive therapy for multiple sclerosis (MS) that received its initial FDA approval in 2004. Its most notable toxicity is progressive multifocal leukoencephalopathy (PML), an opportunistic infection that is the focus of an FDA-mandated registry, TOUCH. The Southern Network on Adverse Reactions identified a fatal case of natalizumab-associated urethral melanoma and undertook an extensive evaluation of all cases of natalizumab-associated melanoma included in the FDA’s Adverse Event Reporting System (FAERS) (between 2005 and 2014).
Methods: Patient characteristics were determined from the FAERS reports and put into a comprehensive dataset. The quality of these reports was assessed based on a 15-point scale of various clinical, demographic, and pharmacy components. Cases were identified as being reported through the TOUCH system, using TOUCH information but reporting outside the system, not using TOUCH at all in the U.S. or not U.S. Quality scores and their components within the 4 Touch groups were statistically compared across all attributes using the mathematical programming-based statistical methodology univariate optimal discriminant analysis.
Results: The mean patient age at the time of diagnosis of melanoma was 46 years (SD 11). Seventeen patients were diagnosed with cutaneous melanoma developing in nonsun-exposed areas. We found that cases reported through the TOUCH registry were of lower quality (mean score 7.7) compared with others that reported outside of the U.S. (mean score 8.5, P < .008). Those cases reported through the TOUCH system were less likely to report the site of melanoma (P < .019) and have overall lower clinical scores (P < .04) compared with reports using TOUCH information but reporting outside of the system.
Conclusions: Our findings suggest that in the U.S., the TOUCH Registry should be expanded to require clinicians to report details of natalizumab-associated melanoma, an opportunistic illness that frequently develops in immunocompromised persons. Also, the FDA-approved product label for natalizumab should be revised to include information on occurrences of melanoma among natalizumab-treated MS patients, particularly those who have cutaneous nevi prior to natalizumab initiation. Natalizumab-treated MS patients and their physicians should be vigilant for changes in nevi appearances and development of new cutaneous lesions (particularly in nonsun-exposed cutaneous areas).
Background: Natalizumab is an effective immunosuppressive therapy for multiple sclerosis (MS) that received its initial FDA approval in 2004. Its most notable toxicity is progressive multifocal leukoencephalopathy (PML), an opportunistic infection that is the focus of an FDA-mandated registry, TOUCH. The Southern Network on Adverse Reactions identified a fatal case of natalizumab-associated urethral melanoma and undertook an extensive evaluation of all cases of natalizumab-associated melanoma included in the FDA’s Adverse Event Reporting System (FAERS) (between 2005 and 2014).
Methods: Patient characteristics were determined from the FAERS reports and put into a comprehensive dataset. The quality of these reports was assessed based on a 15-point scale of various clinical, demographic, and pharmacy components. Cases were identified as being reported through the TOUCH system, using TOUCH information but reporting outside the system, not using TOUCH at all in the U.S. or not U.S. Quality scores and their components within the 4 Touch groups were statistically compared across all attributes using the mathematical programming-based statistical methodology univariate optimal discriminant analysis.
Results: The mean patient age at the time of diagnosis of melanoma was 46 years (SD 11). Seventeen patients were diagnosed with cutaneous melanoma developing in nonsun-exposed areas. We found that cases reported through the TOUCH registry were of lower quality (mean score 7.7) compared with others that reported outside of the U.S. (mean score 8.5, P < .008). Those cases reported through the TOUCH system were less likely to report the site of melanoma (P < .019) and have overall lower clinical scores (P < .04) compared with reports using TOUCH information but reporting outside of the system.
Conclusions: Our findings suggest that in the U.S., the TOUCH Registry should be expanded to require clinicians to report details of natalizumab-associated melanoma, an opportunistic illness that frequently develops in immunocompromised persons. Also, the FDA-approved product label for natalizumab should be revised to include information on occurrences of melanoma among natalizumab-treated MS patients, particularly those who have cutaneous nevi prior to natalizumab initiation. Natalizumab-treated MS patients and their physicians should be vigilant for changes in nevi appearances and development of new cutaneous lesions (particularly in nonsun-exposed cutaneous areas).
New cancer cases to reach nearly 2.4 million in 2035 – AACR
There will be nearly 2.4 million new cases of cancer in the U.S. by 2035, according to an estimate from the American Association for Cancer Research.
In its Cancer Progress Report 2015, AACR notes that new cases in the current year are estimated at nearly 1.7 million.
“Given that cancer is primarily a disease of aging and that the portion of the U.S. population age 65 and older is expected to double in size by 2060, it is anticipated that the number of new cancer cases diagnosed each year in the United States will increase dramatically,” the report states.
“Also contributing to the projected increase are the continual use of cigarettes by 18% of U.S. adults, and high rates of obesity and physical inactivity, both of which are linked to an increased risk for several types of cancer.”
The spike in new cases comes as death rates are trending downward. From 2002 to 2011, overall cancer death rates declined per year by 1.8% for U.S. men, 1.4% for U.S. women, and 2.1% for 0- to 19-year-olds, though the overall number of cancer deaths is expected to rise. Nearly 600,000 people living in the United States are expected to die from cancer in 2015.
The new diagnoses are expected to drive up the cost of cancer.
“In the United States alone, it is estimated that the direct medical costs of cancer care will rise to $156 billion in 2020, from nearly $125 billion in 2010,” AACR says in the report. “When these costs are compared to the total NCI budget for fiscal year 2015, which is just $5 billion, it is clear that research that spurs lifesaving progress against cancer is a wise national investment.”
To that end, AACR is calling on Congress and the White House to boost funding for both the National Institutes of Health and the National Cancer Institute by at least 7%, which would result in funding levels in fiscal year 2020 for NIH and NCI of $42.5 billion and $7 billion, respectively. AACR is also calling for a similar 7% increase the Food and Drug Administration’s budget to help drive private biomedical research.
There will be nearly 2.4 million new cases of cancer in the U.S. by 2035, according to an estimate from the American Association for Cancer Research.
In its Cancer Progress Report 2015, AACR notes that new cases in the current year are estimated at nearly 1.7 million.
“Given that cancer is primarily a disease of aging and that the portion of the U.S. population age 65 and older is expected to double in size by 2060, it is anticipated that the number of new cancer cases diagnosed each year in the United States will increase dramatically,” the report states.
“Also contributing to the projected increase are the continual use of cigarettes by 18% of U.S. adults, and high rates of obesity and physical inactivity, both of which are linked to an increased risk for several types of cancer.”
The spike in new cases comes as death rates are trending downward. From 2002 to 2011, overall cancer death rates declined per year by 1.8% for U.S. men, 1.4% for U.S. women, and 2.1% for 0- to 19-year-olds, though the overall number of cancer deaths is expected to rise. Nearly 600,000 people living in the United States are expected to die from cancer in 2015.
The new diagnoses are expected to drive up the cost of cancer.
“In the United States alone, it is estimated that the direct medical costs of cancer care will rise to $156 billion in 2020, from nearly $125 billion in 2010,” AACR says in the report. “When these costs are compared to the total NCI budget for fiscal year 2015, which is just $5 billion, it is clear that research that spurs lifesaving progress against cancer is a wise national investment.”
To that end, AACR is calling on Congress and the White House to boost funding for both the National Institutes of Health and the National Cancer Institute by at least 7%, which would result in funding levels in fiscal year 2020 for NIH and NCI of $42.5 billion and $7 billion, respectively. AACR is also calling for a similar 7% increase the Food and Drug Administration’s budget to help drive private biomedical research.
There will be nearly 2.4 million new cases of cancer in the U.S. by 2035, according to an estimate from the American Association for Cancer Research.
In its Cancer Progress Report 2015, AACR notes that new cases in the current year are estimated at nearly 1.7 million.
“Given that cancer is primarily a disease of aging and that the portion of the U.S. population age 65 and older is expected to double in size by 2060, it is anticipated that the number of new cancer cases diagnosed each year in the United States will increase dramatically,” the report states.
“Also contributing to the projected increase are the continual use of cigarettes by 18% of U.S. adults, and high rates of obesity and physical inactivity, both of which are linked to an increased risk for several types of cancer.”
The spike in new cases comes as death rates are trending downward. From 2002 to 2011, overall cancer death rates declined per year by 1.8% for U.S. men, 1.4% for U.S. women, and 2.1% for 0- to 19-year-olds, though the overall number of cancer deaths is expected to rise. Nearly 600,000 people living in the United States are expected to die from cancer in 2015.
The new diagnoses are expected to drive up the cost of cancer.
“In the United States alone, it is estimated that the direct medical costs of cancer care will rise to $156 billion in 2020, from nearly $125 billion in 2010,” AACR says in the report. “When these costs are compared to the total NCI budget for fiscal year 2015, which is just $5 billion, it is clear that research that spurs lifesaving progress against cancer is a wise national investment.”
To that end, AACR is calling on Congress and the White House to boost funding for both the National Institutes of Health and the National Cancer Institute by at least 7%, which would result in funding levels in fiscal year 2020 for NIH and NCI of $42.5 billion and $7 billion, respectively. AACR is also calling for a similar 7% increase the Food and Drug Administration’s budget to help drive private biomedical research.
FROM AACR CANCER PROGRESS REPORT 2015
Acne and Melanoma: What to Do With the Reported Connection?
Dermatologists have become accustomed to reading about the associations of dermatologic disease with extracutaneous comorbidities (psoriasis certainly takes the lead). One may see the headline “Study finds increased risk for melanoma in female acne patients” and say “Sure, why not?” However, before we all jump on the association bandwagon, let’s better appreciate this finding.
A study published online January 8 in Cancer by Zhang et al followed 99,128 female nurses in the Nurses’ Health Study II cohort for 20 years. This cohort has been utilized for numerous prospective studies over the year. Even after adjusting for known risk factors, investigators discovered that women with a history of severe cystic teenage acne had a hazard ratio of 1.44 for melanoma. The authors replicated the association with an independent melanoma case-control study of 930 cases and 1026 controls, finding an odds ratio of 1.27. They also found that individuals with teenage acne were more likely to have nevi (52.7% vs 50.1% in the cohort study; 55.2% vs 45.1% in the control study).
These data points ultimately led the team to conclude that acne may serve as an independent risk factor for melanoma, attributing androgens in female acne as a possible and plausible explanation due to their known effect on telomere elongation; melanocytes with longer telomere lengths have more opportunity to develop mutations, which could lead to malignant transformation, as the extended length ultimately delays initiation of cellular senescence. The longer these cells are “awake,” more moles can form, which means more room for trouble.
What’s the issue?
The size of this cohort certainly gives credibility to the data and statistics presented. Although the study is powered very well by the numbers, it is a unique cohort because all participants were nurses, narrowing down the demographics to some degree given general patterns, behaviors, and backgrounds when it comes to this group, an issue that has been previously raised with using this cohort. That said, more research is certainly warranted to elucidate the proposed mechanism and further clarify the association.
From a purely clinical standpoint, this paper is powerful ammo that can be used in our war against skin cancer. This very large cohort probably does not follow the American Academy of Dermatology guidelines for sun protection, skin cancer prevention, and surveillance. It could be a nice tidbit for patients at the end of your spiel on acne and then work in the photoprotection discussion, something we haven’t been the best at according to a recent study published in JAMA Dermatology (JAMA Dermatol. 2014;150:51-55)! Would it be such a bad thing if this paper helped us encourage all women with moderate to severe acne to undertake more effective sun-safe behaviors and to visit their dermatologist every year for total-body skin examinations?
Dermatologists have become accustomed to reading about the associations of dermatologic disease with extracutaneous comorbidities (psoriasis certainly takes the lead). One may see the headline “Study finds increased risk for melanoma in female acne patients” and say “Sure, why not?” However, before we all jump on the association bandwagon, let’s better appreciate this finding.
A study published online January 8 in Cancer by Zhang et al followed 99,128 female nurses in the Nurses’ Health Study II cohort for 20 years. This cohort has been utilized for numerous prospective studies over the year. Even after adjusting for known risk factors, investigators discovered that women with a history of severe cystic teenage acne had a hazard ratio of 1.44 for melanoma. The authors replicated the association with an independent melanoma case-control study of 930 cases and 1026 controls, finding an odds ratio of 1.27. They also found that individuals with teenage acne were more likely to have nevi (52.7% vs 50.1% in the cohort study; 55.2% vs 45.1% in the control study).
These data points ultimately led the team to conclude that acne may serve as an independent risk factor for melanoma, attributing androgens in female acne as a possible and plausible explanation due to their known effect on telomere elongation; melanocytes with longer telomere lengths have more opportunity to develop mutations, which could lead to malignant transformation, as the extended length ultimately delays initiation of cellular senescence. The longer these cells are “awake,” more moles can form, which means more room for trouble.
What’s the issue?
The size of this cohort certainly gives credibility to the data and statistics presented. Although the study is powered very well by the numbers, it is a unique cohort because all participants were nurses, narrowing down the demographics to some degree given general patterns, behaviors, and backgrounds when it comes to this group, an issue that has been previously raised with using this cohort. That said, more research is certainly warranted to elucidate the proposed mechanism and further clarify the association.
From a purely clinical standpoint, this paper is powerful ammo that can be used in our war against skin cancer. This very large cohort probably does not follow the American Academy of Dermatology guidelines for sun protection, skin cancer prevention, and surveillance. It could be a nice tidbit for patients at the end of your spiel on acne and then work in the photoprotection discussion, something we haven’t been the best at according to a recent study published in JAMA Dermatology (JAMA Dermatol. 2014;150:51-55)! Would it be such a bad thing if this paper helped us encourage all women with moderate to severe acne to undertake more effective sun-safe behaviors and to visit their dermatologist every year for total-body skin examinations?
Dermatologists have become accustomed to reading about the associations of dermatologic disease with extracutaneous comorbidities (psoriasis certainly takes the lead). One may see the headline “Study finds increased risk for melanoma in female acne patients” and say “Sure, why not?” However, before we all jump on the association bandwagon, let’s better appreciate this finding.
A study published online January 8 in Cancer by Zhang et al followed 99,128 female nurses in the Nurses’ Health Study II cohort for 20 years. This cohort has been utilized for numerous prospective studies over the year. Even after adjusting for known risk factors, investigators discovered that women with a history of severe cystic teenage acne had a hazard ratio of 1.44 for melanoma. The authors replicated the association with an independent melanoma case-control study of 930 cases and 1026 controls, finding an odds ratio of 1.27. They also found that individuals with teenage acne were more likely to have nevi (52.7% vs 50.1% in the cohort study; 55.2% vs 45.1% in the control study).
These data points ultimately led the team to conclude that acne may serve as an independent risk factor for melanoma, attributing androgens in female acne as a possible and plausible explanation due to their known effect on telomere elongation; melanocytes with longer telomere lengths have more opportunity to develop mutations, which could lead to malignant transformation, as the extended length ultimately delays initiation of cellular senescence. The longer these cells are “awake,” more moles can form, which means more room for trouble.
What’s the issue?
The size of this cohort certainly gives credibility to the data and statistics presented. Although the study is powered very well by the numbers, it is a unique cohort because all participants were nurses, narrowing down the demographics to some degree given general patterns, behaviors, and backgrounds when it comes to this group, an issue that has been previously raised with using this cohort. That said, more research is certainly warranted to elucidate the proposed mechanism and further clarify the association.
From a purely clinical standpoint, this paper is powerful ammo that can be used in our war against skin cancer. This very large cohort probably does not follow the American Academy of Dermatology guidelines for sun protection, skin cancer prevention, and surveillance. It could be a nice tidbit for patients at the end of your spiel on acne and then work in the photoprotection discussion, something we haven’t been the best at according to a recent study published in JAMA Dermatology (JAMA Dermatol. 2014;150:51-55)! Would it be such a bad thing if this paper helped us encourage all women with moderate to severe acne to undertake more effective sun-safe behaviors and to visit their dermatologist every year for total-body skin examinations?