User login
Exuberant Lymphomatoid Papulosis of the Head and Upper Trunk
To the Editor:
Lymphomatoid papulosis (LyP) is a chronic, recurring, self-healing, primary cutaneous lymphoproliferative disorder. This disease affects patients of all ages but most commonly presents in the fifth decade with a slight male predominance.1 The estimated worldwide incidence is 1.2 to 1.9 cases per 1,000,000 individuals, and the 10-year survival rate is close to 100%.1 Clinically, LyP presents as a few to more than 100 red-brown papules or nodules, some with hemorrhagic crust or central necrosis, often occurring in crops and in various stages of evolution. They most commonly are distributed on the trunk and extremities; however, the face, scalp, and oral mucosa rarely may be involved. Each lesion may last on average 3 to 8 weeks, with residual hyperpigmentation or hypopigmentation of the skin or superficial varioliform scars. The clinical characteristic of spontaneous regression is crucial for distinguishing LyP from other forms of cutaneous lymphoma.2 The disease course is variable, lasting anywhere from a few months to decades. Histopathologically, LyP consists of a frequently CD30+ lymphocytic proliferation in multiple described patterns.1 We report a case of LyP in a patient who initially presented with pink edematous papules and vesicles that progressed to crusted ulcerations, nodules, and deep necrotic eschars on the scalp, neck, and upper trunk. Multiple biopsies and T-cell gene rearrangement studies were necessary to make the diagnosis.
A 73-year-old man presented with edematous crusted papules and nodules as well as scarring with serous drainage on the scalp and upper trunk of several months’ duration. He also reported pain and pruritus. He had a medical history of B-cell CD20− chronic lymphocytic leukemia (CLL) that was treated with fludarabine, cyclophosphamide, rituximab, and intravenous immunoglobulin approximately one year prior and currently was in remission; prostate cancer treated with prostatectomy; hypertension; and type 2 diabetes mellitus. His medications included metoprolol, valsartan, and glipizide.
Histopathology revealed a hypersensitivity reaction, and the clinicopathologic correlation was believed to represent an exuberant arthropod bite reaction in the setting of CLL. The eruption responded well to oral prednisone and topical corticosteroids but recurred when the medications were withdrawn. A repeat biopsy resulted in a diagnosis of atypical eosinophil-predominant Sweet syndrome. The condition resolved.
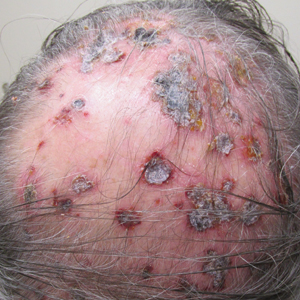
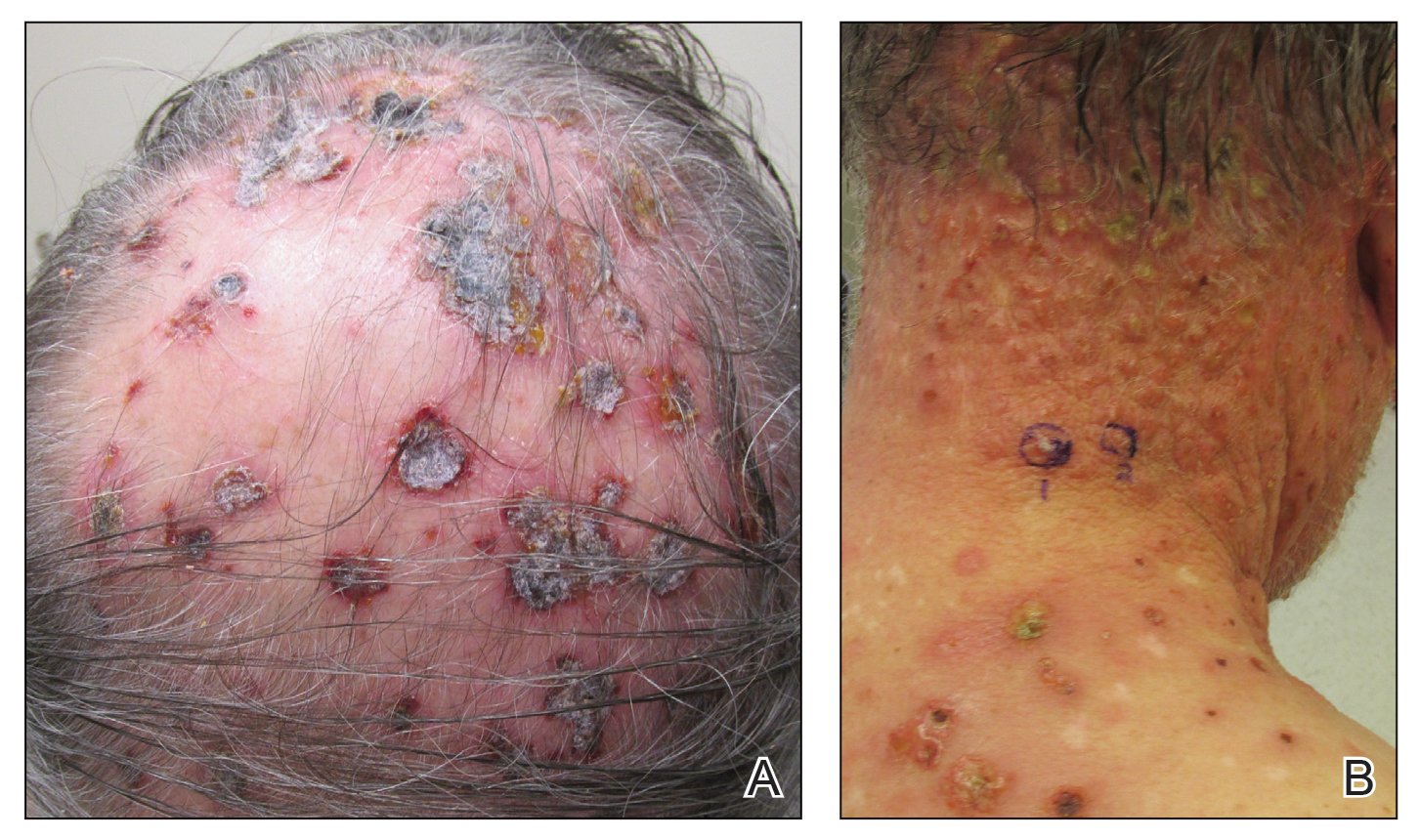
Three years later he developed multiple honey-crusted, superficial ulcers as well as serous, fluid-filled vesiculobullae on the head. A tissue culture revealed Proteus mirabilis, Staphylococcus aureus, and Enterococcus faecalis, and was negative for acid-fast bacteria and fungus. Biopsy of these lesions revealed dermal ulceration with a mixed inflammatory infiltrate and numerous eosinophils as well as a few clustered CD30+ cells; direct immunofluorescence was negative. An extensive laboratory workup including bullous pemphigoid antigens, C-reactive protein, antinuclear antibodies comprehensive profile, antineutrophil cytoplasmic antibodies, rheumatoid factor, anticyclic citrullinated peptide antibodies, serum protein electrophoresis, lactate dehydrogenase, complete blood cell count with differential, complete metabolic profile, thyroid-stimulating hormone, uric acid, C3, C4, immunoglobulin profile, angiotensin-converting enzyme level, and urinalysis was unremarkable. He improved with courses of minocycline, prednisone, and topical clobetasol, but he had periodic and progressive flares over several months with punched-out crusted ulcerations developing on the scalp (Figure 1A) and neck (Figure 1B). The oral and ocular mucosae were uninvolved, but the nasal mucosa had some involvement.
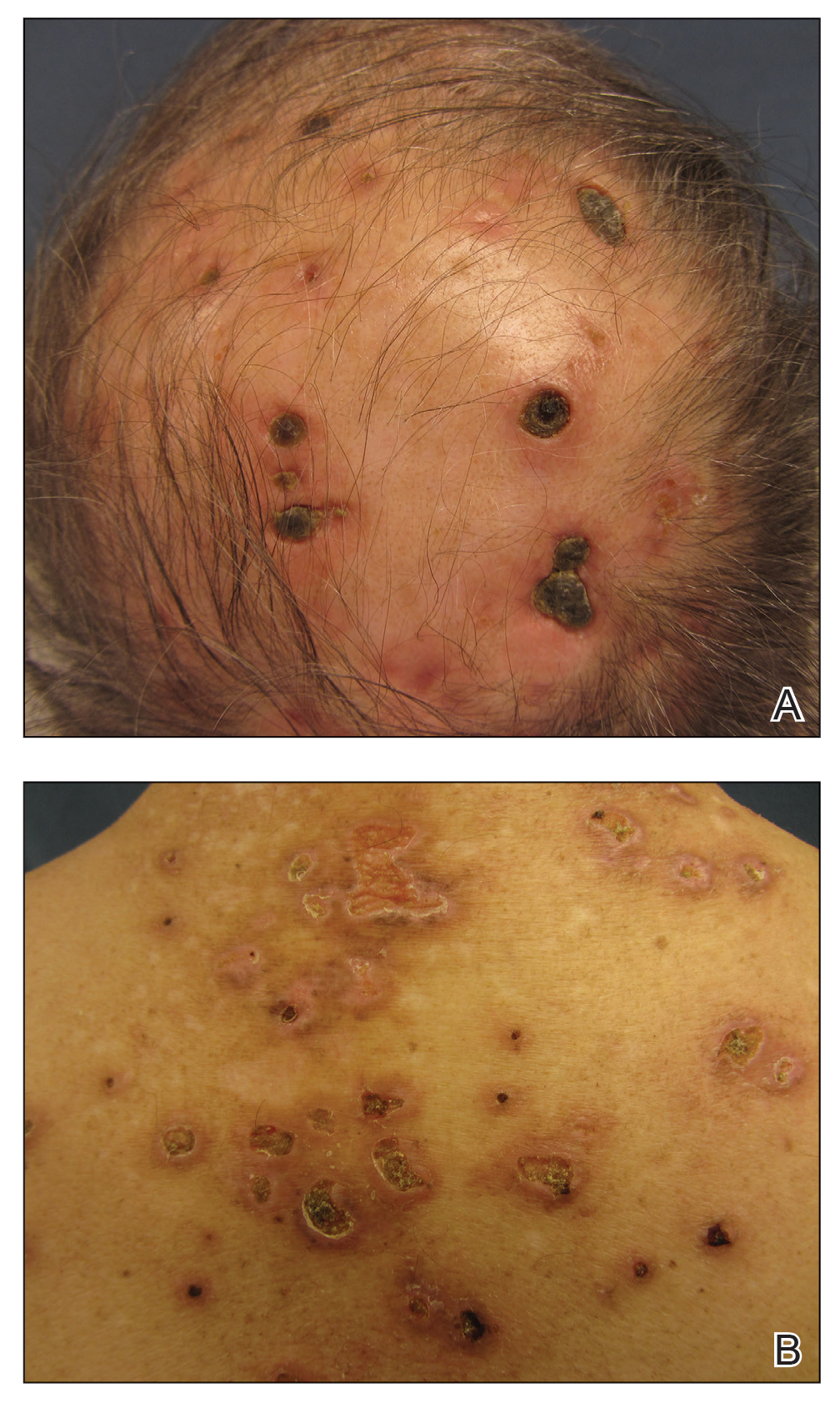
A repeat biopsy demonstrated an atypical CD30+ lymphoid infiltrate favoring LyP. T-cell clonality performed on this specimen and the prior biopsy demonstrated identical T-cell receptor β and γ clones. CD3, CD5, CD7, and CD4 immunostains highlighted the perivascular, perifollicular, and folliculotropic lymphocytic infiltrate. CD8 highlighted occasional background small T cells with only a few folliculotropic forms. A CD30 study revealed several scattered enlarged lymphocytes, and CD20 displayed a few dispersed B cells. A repeat perilesional direct immunofluorescence study was again negative. With treatment, he later formed multiple dry punched-out ulcers with dark eschars on the scalp, posterior neck, and upper back. There were multiple scars on the head, chest, and back, and no vesicles or bullae were present (Figure 2). The patient was presented at a meeting of the Philadelphia Dermatological Society and a consensus diagnosis of LyP was reached. The patient has continued to improve with oral minocycline 100 mg twice daily, topical clobetasol, and topical mupirocin.
Lymphomatoid papulosis is an indolent cutaneous lymphoma; however, it is associated with the potential development of a second hematologic malignancy, with some disagreement in the literature concerning the exact percentage.3 In some studies, lymphoma has been estimated to occur in less than 20% of cases.4,5 Wieser et al1 reported a retrospective analysis of 180 patients with LyP that revealed a secondary malignancy in 52% of patients. They also reported that the number of lesions and the symptom severity were not associated with lymphoma development.1 Similarly, Cordel et al6 reported a diagnosis of lymphoma in 41% of 106 patients. These analyses reveal that the association with lymphoma may be higher than previously thought, but referral bias may be a confounding factor in these numbers.1,5,6 Associated malignancies may occur prior to, concomitantly, or years after the diagnosis of LyP. The most frequently reported malignancies include mycosis fungoides, Hodgkin lymphoma, and primary cutaneous anaplastic large cell lymphoma.1,4
Nicolaou et al3 indicated that head involvement was more likely associated with lymphoma. Our patient had a history of CLL prior to the development of LyP, and it continues to be in remission. The incidence of CLL in patients with LyP is reported to be 0.8%.4 Our patient had an exuberant case of LyP predominantly involving the head, neck, and upper torso, which is an unusual distribution. Vesiculobullous lesions also are uncharacteristic of LyP and may have represented concomitant bullous impetigo, but bullous variants of LyP also have been reported.7 Due to the unique distribution and characteristic scarring, Brunsting-Perry cicatricial pemphigoid also was considered in the clinical differential diagnosis.
The pathogenesis of LyP associated with malignancy is not definitively known. Theories propose that progression to a malignant clonal T-cell population may come from cytogenetic events, inadequate host response, or persistent antigenic or viral stimulation.4 Studies have demonstrated overlapping T-cell receptor gene rearrangement clones in lesions in patients with both LyP and mycosis fungoides, suggesting a common origin between the diseases.8 Other theories suggest that LyP may arise from an early, reactive, polyclonal lymphoid expansion that evolves into a clonal neoplastic process.4 Interestingly, LyP is a clonal T-cell disorder, while Hodgkin lymphoma and CLL are B-cell disorders. Thus, reports of CLL occurring with LyP, as in our patient, may support the theory that LyP arises from an early stem-cell or precursor-cell defect.4
There is no cure for LyP and data regarding the potential of aggressive therapy on the prevention of secondary lymphomas is lacking. Wieser et al1 reported that treatment did not prevent the progression to lymphoma in their retrospective analysis of 180 patients. The number of lesions, frequency of outbreaks, and extent of the scarring can dictate the treatment approach for LyP. Conservative topical therapies include corticosteroids, bexarotene, and imiquimod. Mupirocin may help to prevent infection of ulcerated lesions.1,2 Low-dose methotrexate has been shown to be the most efficacious treatment in reducing the number of lesions, particularly for scarring or cosmetically sensitive areas. Oral methotrexate at a dosage of 10 mg to 25 mg weekly tapered to the lowest effective dose may suppress outbreaks of LyP lesions.1,2 Other therapies include psoralen plus UVA, UVB, interferon alfa-2a, oral bexarotene, oral acyclovir or valacyclovir, etretinate, mycophenolic acid, photodynamic therapy, oral antibiotics, excision, and radiotherapy.1,2 Systemic chemotherapy and total-skin electron beam therapy have shown efficacy in clearing the lesions; however, the disease recurs after discontinuation of therapy.2 Systemic chemotherapy is not recommended for the treatment of LyP, as risks outweigh the benefits and it does not reduce the risk for developing lymphoma.1 The prognosis generally is good, though long-term follow-up is imperative to monitor for the development of other lymphomas.
Our patient presented with LyP a few months after completing chemotherapy for his CLL. It is unknown if he developed LyP just before the time of presentation, or if he may have developed it at the same time as his CLL by a common inciting event. In the latter case, it is speculative that the LyP may have been controlled by chemotherapy for his CLL, only to become clinically apparent after discontinuation, then naturally remit for a longer period. Case reports such as ours with unusual clinical presentations, B-cell lymphoma associations, and unique timing of lymphoma onset may help to provide insight into the pathogenesis of this disease.
We highlighted an unusual case of LyP that presented clinically with crusted ulcerations as well as vesiculobullous and edematous papules that progressed into deep punched-out ulcers with eschars, nodules, and scarring on the head and upper trunk. Lymphomatoid papulosis can be difficult to diagnose histopathologically at the early stages, and multiple repeat biopsies may be necessary to confirm the diagnosis. T-cell gene rearrangement and immunohistochemistry studies are helpful along with clinical correlation to establish a diagnosis in these cases. We recommend that physicians keep LyP on the differential diagnosis for patients with similar clinical presentations and remain vigilant in monitoring for the development of secondary lymphoma.
- Wieser I, Oh C, Talpur R, et al. Lymphomatoid papulosis: treatment response and associated lymphomas in a study of 180 patients. J Am Acad Dermatol. 2016;74:59-67.
- Duvic M. CD30+ neoplasms of the skin. Curr Hematol Malig Rep. 2011;6:245-250.
- Nicolaou V, Papadavid E, Ekonomise A, et al. Association of clinicopathological characteristics with secondary neoplastic lymphoproliferative disorders in patients with lymphomatoid papulosis. Leuk Lymphoma. 2015;56:1303-1307.
- Ahn C, Orscheln C, Huang W. Lymphomatoid papulosis as a harbinger of chronic lymphocytic leukemia. Ann Hematol. 2014;93:1923-1925.
- Kunishige J, McDonald H, Alvarez G, et al. Lymphomatoid papulosis and associated lymphomas: a retrospective case series of 84 patients. Clin Exp Dermatol. 2009;34:576-5781.
- Cordelet al. Frequency and risk factors for associated lymphomas in patients with lymphomatoid papulosis. Oncologist. 2016;21:76-83.
- Sureda N, Thomas L, Bathelier E, et al. Bullous lymphomatoid papulosis. Clin Exp Dermatol. 2011;36:800-801.
- de la Garza Bravo M, Patel KP, Loghavi S, et al. Shared clonality in distinctive lesions of lymphomatoid papulosis and mycosis fungoides occurring in the same patients suggests a common origin. Hum Pathol. 2015;46:558-569.
To the Editor:
Lymphomatoid papulosis (LyP) is a chronic, recurring, self-healing, primary cutaneous lymphoproliferative disorder. This disease affects patients of all ages but most commonly presents in the fifth decade with a slight male predominance.1 The estimated worldwide incidence is 1.2 to 1.9 cases per 1,000,000 individuals, and the 10-year survival rate is close to 100%.1 Clinically, LyP presents as a few to more than 100 red-brown papules or nodules, some with hemorrhagic crust or central necrosis, often occurring in crops and in various stages of evolution. They most commonly are distributed on the trunk and extremities; however, the face, scalp, and oral mucosa rarely may be involved. Each lesion may last on average 3 to 8 weeks, with residual hyperpigmentation or hypopigmentation of the skin or superficial varioliform scars. The clinical characteristic of spontaneous regression is crucial for distinguishing LyP from other forms of cutaneous lymphoma.2 The disease course is variable, lasting anywhere from a few months to decades. Histopathologically, LyP consists of a frequently CD30+ lymphocytic proliferation in multiple described patterns.1 We report a case of LyP in a patient who initially presented with pink edematous papules and vesicles that progressed to crusted ulcerations, nodules, and deep necrotic eschars on the scalp, neck, and upper trunk. Multiple biopsies and T-cell gene rearrangement studies were necessary to make the diagnosis.
A 73-year-old man presented with edematous crusted papules and nodules as well as scarring with serous drainage on the scalp and upper trunk of several months’ duration. He also reported pain and pruritus. He had a medical history of B-cell CD20− chronic lymphocytic leukemia (CLL) that was treated with fludarabine, cyclophosphamide, rituximab, and intravenous immunoglobulin approximately one year prior and currently was in remission; prostate cancer treated with prostatectomy; hypertension; and type 2 diabetes mellitus. His medications included metoprolol, valsartan, and glipizide.
Histopathology revealed a hypersensitivity reaction, and the clinicopathologic correlation was believed to represent an exuberant arthropod bite reaction in the setting of CLL. The eruption responded well to oral prednisone and topical corticosteroids but recurred when the medications were withdrawn. A repeat biopsy resulted in a diagnosis of atypical eosinophil-predominant Sweet syndrome. The condition resolved.
Three years later he developed multiple honey-crusted, superficial ulcers as well as serous, fluid-filled vesiculobullae on the head. A tissue culture revealed Proteus mirabilis, Staphylococcus aureus, and Enterococcus faecalis, and was negative for acid-fast bacteria and fungus. Biopsy of these lesions revealed dermal ulceration with a mixed inflammatory infiltrate and numerous eosinophils as well as a few clustered CD30+ cells; direct immunofluorescence was negative. An extensive laboratory workup including bullous pemphigoid antigens, C-reactive protein, antinuclear antibodies comprehensive profile, antineutrophil cytoplasmic antibodies, rheumatoid factor, anticyclic citrullinated peptide antibodies, serum protein electrophoresis, lactate dehydrogenase, complete blood cell count with differential, complete metabolic profile, thyroid-stimulating hormone, uric acid, C3, C4, immunoglobulin profile, angiotensin-converting enzyme level, and urinalysis was unremarkable. He improved with courses of minocycline, prednisone, and topical clobetasol, but he had periodic and progressive flares over several months with punched-out crusted ulcerations developing on the scalp (Figure 1A) and neck (Figure 1B). The oral and ocular mucosae were uninvolved, but the nasal mucosa had some involvement.

A repeat biopsy demonstrated an atypical CD30+ lymphoid infiltrate favoring LyP. T-cell clonality performed on this specimen and the prior biopsy demonstrated identical T-cell receptor β and γ clones. CD3, CD5, CD7, and CD4 immunostains highlighted the perivascular, perifollicular, and folliculotropic lymphocytic infiltrate. CD8 highlighted occasional background small T cells with only a few folliculotropic forms. A CD30 study revealed several scattered enlarged lymphocytes, and CD20 displayed a few dispersed B cells. A repeat perilesional direct immunofluorescence study was again negative. With treatment, he later formed multiple dry punched-out ulcers with dark eschars on the scalp, posterior neck, and upper back. There were multiple scars on the head, chest, and back, and no vesicles or bullae were present (Figure 2). The patient was presented at a meeting of the Philadelphia Dermatological Society and a consensus diagnosis of LyP was reached. The patient has continued to improve with oral minocycline 100 mg twice daily, topical clobetasol, and topical mupirocin.

Lymphomatoid papulosis is an indolent cutaneous lymphoma; however, it is associated with the potential development of a second hematologic malignancy, with some disagreement in the literature concerning the exact percentage.3 In some studies, lymphoma has been estimated to occur in less than 20% of cases.4,5 Wieser et al1 reported a retrospective analysis of 180 patients with LyP that revealed a secondary malignancy in 52% of patients. They also reported that the number of lesions and the symptom severity were not associated with lymphoma development.1 Similarly, Cordel et al6 reported a diagnosis of lymphoma in 41% of 106 patients. These analyses reveal that the association with lymphoma may be higher than previously thought, but referral bias may be a confounding factor in these numbers.1,5,6 Associated malignancies may occur prior to, concomitantly, or years after the diagnosis of LyP. The most frequently reported malignancies include mycosis fungoides, Hodgkin lymphoma, and primary cutaneous anaplastic large cell lymphoma.1,4
Nicolaou et al3 indicated that head involvement was more likely associated with lymphoma. Our patient had a history of CLL prior to the development of LyP, and it continues to be in remission. The incidence of CLL in patients with LyP is reported to be 0.8%.4 Our patient had an exuberant case of LyP predominantly involving the head, neck, and upper torso, which is an unusual distribution. Vesiculobullous lesions also are uncharacteristic of LyP and may have represented concomitant bullous impetigo, but bullous variants of LyP also have been reported.7 Due to the unique distribution and characteristic scarring, Brunsting-Perry cicatricial pemphigoid also was considered in the clinical differential diagnosis.
The pathogenesis of LyP associated with malignancy is not definitively known. Theories propose that progression to a malignant clonal T-cell population may come from cytogenetic events, inadequate host response, or persistent antigenic or viral stimulation.4 Studies have demonstrated overlapping T-cell receptor gene rearrangement clones in lesions in patients with both LyP and mycosis fungoides, suggesting a common origin between the diseases.8 Other theories suggest that LyP may arise from an early, reactive, polyclonal lymphoid expansion that evolves into a clonal neoplastic process.4 Interestingly, LyP is a clonal T-cell disorder, while Hodgkin lymphoma and CLL are B-cell disorders. Thus, reports of CLL occurring with LyP, as in our patient, may support the theory that LyP arises from an early stem-cell or precursor-cell defect.4
There is no cure for LyP and data regarding the potential of aggressive therapy on the prevention of secondary lymphomas is lacking. Wieser et al1 reported that treatment did not prevent the progression to lymphoma in their retrospective analysis of 180 patients. The number of lesions, frequency of outbreaks, and extent of the scarring can dictate the treatment approach for LyP. Conservative topical therapies include corticosteroids, bexarotene, and imiquimod. Mupirocin may help to prevent infection of ulcerated lesions.1,2 Low-dose methotrexate has been shown to be the most efficacious treatment in reducing the number of lesions, particularly for scarring or cosmetically sensitive areas. Oral methotrexate at a dosage of 10 mg to 25 mg weekly tapered to the lowest effective dose may suppress outbreaks of LyP lesions.1,2 Other therapies include psoralen plus UVA, UVB, interferon alfa-2a, oral bexarotene, oral acyclovir or valacyclovir, etretinate, mycophenolic acid, photodynamic therapy, oral antibiotics, excision, and radiotherapy.1,2 Systemic chemotherapy and total-skin electron beam therapy have shown efficacy in clearing the lesions; however, the disease recurs after discontinuation of therapy.2 Systemic chemotherapy is not recommended for the treatment of LyP, as risks outweigh the benefits and it does not reduce the risk for developing lymphoma.1 The prognosis generally is good, though long-term follow-up is imperative to monitor for the development of other lymphomas.
Our patient presented with LyP a few months after completing chemotherapy for his CLL. It is unknown if he developed LyP just before the time of presentation, or if he may have developed it at the same time as his CLL by a common inciting event. In the latter case, it is speculative that the LyP may have been controlled by chemotherapy for his CLL, only to become clinically apparent after discontinuation, then naturally remit for a longer period. Case reports such as ours with unusual clinical presentations, B-cell lymphoma associations, and unique timing of lymphoma onset may help to provide insight into the pathogenesis of this disease.
We highlighted an unusual case of LyP that presented clinically with crusted ulcerations as well as vesiculobullous and edematous papules that progressed into deep punched-out ulcers with eschars, nodules, and scarring on the head and upper trunk. Lymphomatoid papulosis can be difficult to diagnose histopathologically at the early stages, and multiple repeat biopsies may be necessary to confirm the diagnosis. T-cell gene rearrangement and immunohistochemistry studies are helpful along with clinical correlation to establish a diagnosis in these cases. We recommend that physicians keep LyP on the differential diagnosis for patients with similar clinical presentations and remain vigilant in monitoring for the development of secondary lymphoma.
To the Editor:
Lymphomatoid papulosis (LyP) is a chronic, recurring, self-healing, primary cutaneous lymphoproliferative disorder. This disease affects patients of all ages but most commonly presents in the fifth decade with a slight male predominance.1 The estimated worldwide incidence is 1.2 to 1.9 cases per 1,000,000 individuals, and the 10-year survival rate is close to 100%.1 Clinically, LyP presents as a few to more than 100 red-brown papules or nodules, some with hemorrhagic crust or central necrosis, often occurring in crops and in various stages of evolution. They most commonly are distributed on the trunk and extremities; however, the face, scalp, and oral mucosa rarely may be involved. Each lesion may last on average 3 to 8 weeks, with residual hyperpigmentation or hypopigmentation of the skin or superficial varioliform scars. The clinical characteristic of spontaneous regression is crucial for distinguishing LyP from other forms of cutaneous lymphoma.2 The disease course is variable, lasting anywhere from a few months to decades. Histopathologically, LyP consists of a frequently CD30+ lymphocytic proliferation in multiple described patterns.1 We report a case of LyP in a patient who initially presented with pink edematous papules and vesicles that progressed to crusted ulcerations, nodules, and deep necrotic eschars on the scalp, neck, and upper trunk. Multiple biopsies and T-cell gene rearrangement studies were necessary to make the diagnosis.
A 73-year-old man presented with edematous crusted papules and nodules as well as scarring with serous drainage on the scalp and upper trunk of several months’ duration. He also reported pain and pruritus. He had a medical history of B-cell CD20− chronic lymphocytic leukemia (CLL) that was treated with fludarabine, cyclophosphamide, rituximab, and intravenous immunoglobulin approximately one year prior and currently was in remission; prostate cancer treated with prostatectomy; hypertension; and type 2 diabetes mellitus. His medications included metoprolol, valsartan, and glipizide.
Histopathology revealed a hypersensitivity reaction, and the clinicopathologic correlation was believed to represent an exuberant arthropod bite reaction in the setting of CLL. The eruption responded well to oral prednisone and topical corticosteroids but recurred when the medications were withdrawn. A repeat biopsy resulted in a diagnosis of atypical eosinophil-predominant Sweet syndrome. The condition resolved.
Three years later he developed multiple honey-crusted, superficial ulcers as well as serous, fluid-filled vesiculobullae on the head. A tissue culture revealed Proteus mirabilis, Staphylococcus aureus, and Enterococcus faecalis, and was negative for acid-fast bacteria and fungus. Biopsy of these lesions revealed dermal ulceration with a mixed inflammatory infiltrate and numerous eosinophils as well as a few clustered CD30+ cells; direct immunofluorescence was negative. An extensive laboratory workup including bullous pemphigoid antigens, C-reactive protein, antinuclear antibodies comprehensive profile, antineutrophil cytoplasmic antibodies, rheumatoid factor, anticyclic citrullinated peptide antibodies, serum protein electrophoresis, lactate dehydrogenase, complete blood cell count with differential, complete metabolic profile, thyroid-stimulating hormone, uric acid, C3, C4, immunoglobulin profile, angiotensin-converting enzyme level, and urinalysis was unremarkable. He improved with courses of minocycline, prednisone, and topical clobetasol, but he had periodic and progressive flares over several months with punched-out crusted ulcerations developing on the scalp (Figure 1A) and neck (Figure 1B). The oral and ocular mucosae were uninvolved, but the nasal mucosa had some involvement.

A repeat biopsy demonstrated an atypical CD30+ lymphoid infiltrate favoring LyP. T-cell clonality performed on this specimen and the prior biopsy demonstrated identical T-cell receptor β and γ clones. CD3, CD5, CD7, and CD4 immunostains highlighted the perivascular, perifollicular, and folliculotropic lymphocytic infiltrate. CD8 highlighted occasional background small T cells with only a few folliculotropic forms. A CD30 study revealed several scattered enlarged lymphocytes, and CD20 displayed a few dispersed B cells. A repeat perilesional direct immunofluorescence study was again negative. With treatment, he later formed multiple dry punched-out ulcers with dark eschars on the scalp, posterior neck, and upper back. There were multiple scars on the head, chest, and back, and no vesicles or bullae were present (Figure 2). The patient was presented at a meeting of the Philadelphia Dermatological Society and a consensus diagnosis of LyP was reached. The patient has continued to improve with oral minocycline 100 mg twice daily, topical clobetasol, and topical mupirocin.

Lymphomatoid papulosis is an indolent cutaneous lymphoma; however, it is associated with the potential development of a second hematologic malignancy, with some disagreement in the literature concerning the exact percentage.3 In some studies, lymphoma has been estimated to occur in less than 20% of cases.4,5 Wieser et al1 reported a retrospective analysis of 180 patients with LyP that revealed a secondary malignancy in 52% of patients. They also reported that the number of lesions and the symptom severity were not associated with lymphoma development.1 Similarly, Cordel et al6 reported a diagnosis of lymphoma in 41% of 106 patients. These analyses reveal that the association with lymphoma may be higher than previously thought, but referral bias may be a confounding factor in these numbers.1,5,6 Associated malignancies may occur prior to, concomitantly, or years after the diagnosis of LyP. The most frequently reported malignancies include mycosis fungoides, Hodgkin lymphoma, and primary cutaneous anaplastic large cell lymphoma.1,4
Nicolaou et al3 indicated that head involvement was more likely associated with lymphoma. Our patient had a history of CLL prior to the development of LyP, and it continues to be in remission. The incidence of CLL in patients with LyP is reported to be 0.8%.4 Our patient had an exuberant case of LyP predominantly involving the head, neck, and upper torso, which is an unusual distribution. Vesiculobullous lesions also are uncharacteristic of LyP and may have represented concomitant bullous impetigo, but bullous variants of LyP also have been reported.7 Due to the unique distribution and characteristic scarring, Brunsting-Perry cicatricial pemphigoid also was considered in the clinical differential diagnosis.
The pathogenesis of LyP associated with malignancy is not definitively known. Theories propose that progression to a malignant clonal T-cell population may come from cytogenetic events, inadequate host response, or persistent antigenic or viral stimulation.4 Studies have demonstrated overlapping T-cell receptor gene rearrangement clones in lesions in patients with both LyP and mycosis fungoides, suggesting a common origin between the diseases.8 Other theories suggest that LyP may arise from an early, reactive, polyclonal lymphoid expansion that evolves into a clonal neoplastic process.4 Interestingly, LyP is a clonal T-cell disorder, while Hodgkin lymphoma and CLL are B-cell disorders. Thus, reports of CLL occurring with LyP, as in our patient, may support the theory that LyP arises from an early stem-cell or precursor-cell defect.4
There is no cure for LyP and data regarding the potential of aggressive therapy on the prevention of secondary lymphomas is lacking. Wieser et al1 reported that treatment did not prevent the progression to lymphoma in their retrospective analysis of 180 patients. The number of lesions, frequency of outbreaks, and extent of the scarring can dictate the treatment approach for LyP. Conservative topical therapies include corticosteroids, bexarotene, and imiquimod. Mupirocin may help to prevent infection of ulcerated lesions.1,2 Low-dose methotrexate has been shown to be the most efficacious treatment in reducing the number of lesions, particularly for scarring or cosmetically sensitive areas. Oral methotrexate at a dosage of 10 mg to 25 mg weekly tapered to the lowest effective dose may suppress outbreaks of LyP lesions.1,2 Other therapies include psoralen plus UVA, UVB, interferon alfa-2a, oral bexarotene, oral acyclovir or valacyclovir, etretinate, mycophenolic acid, photodynamic therapy, oral antibiotics, excision, and radiotherapy.1,2 Systemic chemotherapy and total-skin electron beam therapy have shown efficacy in clearing the lesions; however, the disease recurs after discontinuation of therapy.2 Systemic chemotherapy is not recommended for the treatment of LyP, as risks outweigh the benefits and it does not reduce the risk for developing lymphoma.1 The prognosis generally is good, though long-term follow-up is imperative to monitor for the development of other lymphomas.
Our patient presented with LyP a few months after completing chemotherapy for his CLL. It is unknown if he developed LyP just before the time of presentation, or if he may have developed it at the same time as his CLL by a common inciting event. In the latter case, it is speculative that the LyP may have been controlled by chemotherapy for his CLL, only to become clinically apparent after discontinuation, then naturally remit for a longer period. Case reports such as ours with unusual clinical presentations, B-cell lymphoma associations, and unique timing of lymphoma onset may help to provide insight into the pathogenesis of this disease.
We highlighted an unusual case of LyP that presented clinically with crusted ulcerations as well as vesiculobullous and edematous papules that progressed into deep punched-out ulcers with eschars, nodules, and scarring on the head and upper trunk. Lymphomatoid papulosis can be difficult to diagnose histopathologically at the early stages, and multiple repeat biopsies may be necessary to confirm the diagnosis. T-cell gene rearrangement and immunohistochemistry studies are helpful along with clinical correlation to establish a diagnosis in these cases. We recommend that physicians keep LyP on the differential diagnosis for patients with similar clinical presentations and remain vigilant in monitoring for the development of secondary lymphoma.
- Wieser I, Oh C, Talpur R, et al. Lymphomatoid papulosis: treatment response and associated lymphomas in a study of 180 patients. J Am Acad Dermatol. 2016;74:59-67.
- Duvic M. CD30+ neoplasms of the skin. Curr Hematol Malig Rep. 2011;6:245-250.
- Nicolaou V, Papadavid E, Ekonomise A, et al. Association of clinicopathological characteristics with secondary neoplastic lymphoproliferative disorders in patients with lymphomatoid papulosis. Leuk Lymphoma. 2015;56:1303-1307.
- Ahn C, Orscheln C, Huang W. Lymphomatoid papulosis as a harbinger of chronic lymphocytic leukemia. Ann Hematol. 2014;93:1923-1925.
- Kunishige J, McDonald H, Alvarez G, et al. Lymphomatoid papulosis and associated lymphomas: a retrospective case series of 84 patients. Clin Exp Dermatol. 2009;34:576-5781.
- Cordelet al. Frequency and risk factors for associated lymphomas in patients with lymphomatoid papulosis. Oncologist. 2016;21:76-83.
- Sureda N, Thomas L, Bathelier E, et al. Bullous lymphomatoid papulosis. Clin Exp Dermatol. 2011;36:800-801.
- de la Garza Bravo M, Patel KP, Loghavi S, et al. Shared clonality in distinctive lesions of lymphomatoid papulosis and mycosis fungoides occurring in the same patients suggests a common origin. Hum Pathol. 2015;46:558-569.
- Wieser I, Oh C, Talpur R, et al. Lymphomatoid papulosis: treatment response and associated lymphomas in a study of 180 patients. J Am Acad Dermatol. 2016;74:59-67.
- Duvic M. CD30+ neoplasms of the skin. Curr Hematol Malig Rep. 2011;6:245-250.
- Nicolaou V, Papadavid E, Ekonomise A, et al. Association of clinicopathological characteristics with secondary neoplastic lymphoproliferative disorders in patients with lymphomatoid papulosis. Leuk Lymphoma. 2015;56:1303-1307.
- Ahn C, Orscheln C, Huang W. Lymphomatoid papulosis as a harbinger of chronic lymphocytic leukemia. Ann Hematol. 2014;93:1923-1925.
- Kunishige J, McDonald H, Alvarez G, et al. Lymphomatoid papulosis and associated lymphomas: a retrospective case series of 84 patients. Clin Exp Dermatol. 2009;34:576-5781.
- Cordelet al. Frequency and risk factors for associated lymphomas in patients with lymphomatoid papulosis. Oncologist. 2016;21:76-83.
- Sureda N, Thomas L, Bathelier E, et al. Bullous lymphomatoid papulosis. Clin Exp Dermatol. 2011;36:800-801.
- de la Garza Bravo M, Patel KP, Loghavi S, et al. Shared clonality in distinctive lesions of lymphomatoid papulosis and mycosis fungoides occurring in the same patients suggests a common origin. Hum Pathol. 2015;46:558-569.
Practice Points
- Lymphomatoid papulosis (LyP) is a chronic, recurring, self-healing, primary cutaneous lymphoproliferative disorder characterized by red-brown papules or nodules, some with hemorrhagic crust or central necrosis, often occurring in crops and in various stages of evolution.
- Histopathologically, LyP consists of a frequently CD30Mathematical Pi LT Std+ lymphocytic proliferation in multiple described patterns.
- Lymphomatoid papulosis is an indolent cutaneous lymphoma; however, it is associated with the potential development of a second hematologic malignancy.
Combined imaging methods found to enhance detection of squamous cell carcinoma
and distinguishing SCC in-situ and actinic keratosis (AK) from invasive SCC, results from a small prospective study demonstrated.
“A solitary scaly papule or plaque could represent an inflammatory or neoplastic process, and when neoplastic, it could be benign, premalignant, malignant in situ, or invasive malignant,” lead study author Abdullah Aleisa, MD, said in an interview during the annual conference of the American Society for Laser Medicine and Surgery. Noninvasive imaging devices, such as reflectance confocal microscopy (RCM) and optical coherence tomography (OCT), “have been used to help in the diagnosis of those clinically suspicious lesions, however each device has its own limitation.”
RCM images are horizontal sections of the skin with high cellular resolution but limited to 250 mcm of depth in skin, he said, while OCT images are vertical sections of the skin with low cellular resolution, but image up to 1,000-2,000 mcm of depth in skin.
“Combined RCM-OCT enables high cellular resolution and deep tissue evaluation,” said Dr. Aleisa, a micrographic surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York. “The value of combined RCM-OCT has been shown in the detection and depth assessment of basal cell carcinoma, but it has never been studied in SCC. Our objective is to combine RCM and OCT simultaneously to detect SCC and assess the depth of invasion.”
Between September and December 2020, Dr. Aleisa and colleagues prospectively imaged 36 lesions suspicious of SCC, SCC in situ, or AK between September 2020 and December 2020. The mean age of the cohort was 68 years and 63% were male. Using a prototype device from Andover, Mass.–based Caliber I.D., the investigators performed handheld RCM-OCT imaging at the center of clinically suspected lesions before biopsy and to previously diagnosed lesions before Mohs micrographic surgery (to check for residual tumor) and correlated RCM-OCT findings with histopathology results. A total of 36 lesions were treated.
Dr. Aleisa reported that most common RCM-OCT feature for invasive SCC was presence of vertical blood vessels (in 89% of lesions), while for SCC in situ/AK, it was acanthosis and parakeratosis without vertical blood vessels (in 84% of lesions). For the detection of invasive SCC, RCM-OCT had a sensitivity of 82%, a specificity of 92%, a negative predictive value of 92%, and a positive predictive value of 82%. For the detection of SCC in situ/AK, RCM-OCT had a sensitivity of 86%, a specificity of 100%, a negative predictive value of 92%, and a positive predictive value of 100%. The OCT depth measurement correlated well with histopathology with a concordance correlation coefficient of r2 = 0.9.
“Using RCM’s high-resolution pictures allowed us to easily spot the vertical ‘buttonhole’ vessels associated with SCC,” Dr. Aleisa said. “However, given the depth limitation of RCM, the distinction between SCC in situ and invasive SCC could not be accomplished using RCM alone. Therefore, having simultaneous OCT live feedback to the RCM images in the combined RCM-OCT device enabled us to assess the depth of those vertical ‘buttonholes’ and distinguish between SCC in situ and invasive SCC.”
He acknowledged certain limitations of the approach, including that it requires approximately 20 minutes per imaging session, there is a steep learning curve for interpreting images, and certain anatomical sites are challenging to image, especially the nose, periocular area, and lip.
The study won a “best of session” emerging technologies abstract award from the ASLMS.
Milind Rajadhyaksha, PhD, of Memorial Sloan Kettering Cancer Center helped to develop the prototype device. Dr. Aleisa reported having no relevant financial disclosures.
and distinguishing SCC in-situ and actinic keratosis (AK) from invasive SCC, results from a small prospective study demonstrated.
“A solitary scaly papule or plaque could represent an inflammatory or neoplastic process, and when neoplastic, it could be benign, premalignant, malignant in situ, or invasive malignant,” lead study author Abdullah Aleisa, MD, said in an interview during the annual conference of the American Society for Laser Medicine and Surgery. Noninvasive imaging devices, such as reflectance confocal microscopy (RCM) and optical coherence tomography (OCT), “have been used to help in the diagnosis of those clinically suspicious lesions, however each device has its own limitation.”
RCM images are horizontal sections of the skin with high cellular resolution but limited to 250 mcm of depth in skin, he said, while OCT images are vertical sections of the skin with low cellular resolution, but image up to 1,000-2,000 mcm of depth in skin.
“Combined RCM-OCT enables high cellular resolution and deep tissue evaluation,” said Dr. Aleisa, a micrographic surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York. “The value of combined RCM-OCT has been shown in the detection and depth assessment of basal cell carcinoma, but it has never been studied in SCC. Our objective is to combine RCM and OCT simultaneously to detect SCC and assess the depth of invasion.”
Between September and December 2020, Dr. Aleisa and colleagues prospectively imaged 36 lesions suspicious of SCC, SCC in situ, or AK between September 2020 and December 2020. The mean age of the cohort was 68 years and 63% were male. Using a prototype device from Andover, Mass.–based Caliber I.D., the investigators performed handheld RCM-OCT imaging at the center of clinically suspected lesions before biopsy and to previously diagnosed lesions before Mohs micrographic surgery (to check for residual tumor) and correlated RCM-OCT findings with histopathology results. A total of 36 lesions were treated.
Dr. Aleisa reported that most common RCM-OCT feature for invasive SCC was presence of vertical blood vessels (in 89% of lesions), while for SCC in situ/AK, it was acanthosis and parakeratosis without vertical blood vessels (in 84% of lesions). For the detection of invasive SCC, RCM-OCT had a sensitivity of 82%, a specificity of 92%, a negative predictive value of 92%, and a positive predictive value of 82%. For the detection of SCC in situ/AK, RCM-OCT had a sensitivity of 86%, a specificity of 100%, a negative predictive value of 92%, and a positive predictive value of 100%. The OCT depth measurement correlated well with histopathology with a concordance correlation coefficient of r2 = 0.9.
“Using RCM’s high-resolution pictures allowed us to easily spot the vertical ‘buttonhole’ vessels associated with SCC,” Dr. Aleisa said. “However, given the depth limitation of RCM, the distinction between SCC in situ and invasive SCC could not be accomplished using RCM alone. Therefore, having simultaneous OCT live feedback to the RCM images in the combined RCM-OCT device enabled us to assess the depth of those vertical ‘buttonholes’ and distinguish between SCC in situ and invasive SCC.”
He acknowledged certain limitations of the approach, including that it requires approximately 20 minutes per imaging session, there is a steep learning curve for interpreting images, and certain anatomical sites are challenging to image, especially the nose, periocular area, and lip.
The study won a “best of session” emerging technologies abstract award from the ASLMS.
Milind Rajadhyaksha, PhD, of Memorial Sloan Kettering Cancer Center helped to develop the prototype device. Dr. Aleisa reported having no relevant financial disclosures.
and distinguishing SCC in-situ and actinic keratosis (AK) from invasive SCC, results from a small prospective study demonstrated.
“A solitary scaly papule or plaque could represent an inflammatory or neoplastic process, and when neoplastic, it could be benign, premalignant, malignant in situ, or invasive malignant,” lead study author Abdullah Aleisa, MD, said in an interview during the annual conference of the American Society for Laser Medicine and Surgery. Noninvasive imaging devices, such as reflectance confocal microscopy (RCM) and optical coherence tomography (OCT), “have been used to help in the diagnosis of those clinically suspicious lesions, however each device has its own limitation.”
RCM images are horizontal sections of the skin with high cellular resolution but limited to 250 mcm of depth in skin, he said, while OCT images are vertical sections of the skin with low cellular resolution, but image up to 1,000-2,000 mcm of depth in skin.
“Combined RCM-OCT enables high cellular resolution and deep tissue evaluation,” said Dr. Aleisa, a micrographic surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York. “The value of combined RCM-OCT has been shown in the detection and depth assessment of basal cell carcinoma, but it has never been studied in SCC. Our objective is to combine RCM and OCT simultaneously to detect SCC and assess the depth of invasion.”
Between September and December 2020, Dr. Aleisa and colleagues prospectively imaged 36 lesions suspicious of SCC, SCC in situ, or AK between September 2020 and December 2020. The mean age of the cohort was 68 years and 63% were male. Using a prototype device from Andover, Mass.–based Caliber I.D., the investigators performed handheld RCM-OCT imaging at the center of clinically suspected lesions before biopsy and to previously diagnosed lesions before Mohs micrographic surgery (to check for residual tumor) and correlated RCM-OCT findings with histopathology results. A total of 36 lesions were treated.
Dr. Aleisa reported that most common RCM-OCT feature for invasive SCC was presence of vertical blood vessels (in 89% of lesions), while for SCC in situ/AK, it was acanthosis and parakeratosis without vertical blood vessels (in 84% of lesions). For the detection of invasive SCC, RCM-OCT had a sensitivity of 82%, a specificity of 92%, a negative predictive value of 92%, and a positive predictive value of 82%. For the detection of SCC in situ/AK, RCM-OCT had a sensitivity of 86%, a specificity of 100%, a negative predictive value of 92%, and a positive predictive value of 100%. The OCT depth measurement correlated well with histopathology with a concordance correlation coefficient of r2 = 0.9.
“Using RCM’s high-resolution pictures allowed us to easily spot the vertical ‘buttonhole’ vessels associated with SCC,” Dr. Aleisa said. “However, given the depth limitation of RCM, the distinction between SCC in situ and invasive SCC could not be accomplished using RCM alone. Therefore, having simultaneous OCT live feedback to the RCM images in the combined RCM-OCT device enabled us to assess the depth of those vertical ‘buttonholes’ and distinguish between SCC in situ and invasive SCC.”
He acknowledged certain limitations of the approach, including that it requires approximately 20 minutes per imaging session, there is a steep learning curve for interpreting images, and certain anatomical sites are challenging to image, especially the nose, periocular area, and lip.
The study won a “best of session” emerging technologies abstract award from the ASLMS.
Milind Rajadhyaksha, PhD, of Memorial Sloan Kettering Cancer Center helped to develop the prototype device. Dr. Aleisa reported having no relevant financial disclosures.
FROM ASLMS 2021
Telemedicine is popular among Mohs surgeons – for now
A majority of
A variety of factors combine to make it “very difficult for surgeons to make long-term plans for implementing telemedicine in their practices,” said Mario Maruthur, MD, who presented the findings at the annual meeting of the American College of Mohs Surgery. “Telemedicine likely has a role in Mohs practices, particularly with postop follow-up visits. However, postpandemic reimbursement and regulatory issues need to be formally laid out before Mohs surgeons are able to incorporate it into their permanent work flow.”
Dr. Maruthur, a Mohs surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York, and colleagues sent a survey to ACMS members in September and October 2020. “We saw first-hand in our surgical practice that telemedicine quickly became an important tool when the pandemic surged in the spring of 2020,” he said. Considering that surgical practices are highly dependent on in-person visits, the impetus for this study was to assess to what degree Mohs practices from across the spectrum, including academic and private practices, embraced telemedicine during the pandemic, and “what these surgical practices used telemedicine for, how it was received by their patients, which telemedicine platforms were most often utilized, and lastly, what are their plans if any for incorporating telemedicine into their surgical practices after the pandemic subsides.”
The researchers received responses from 115 surgeons representing all regions of the country (40% Northeast, 21% South, 21% Midwest, and 18% West). Half practiced in urban areas (37%) and large cities (13%), and 40% were in an academic setting versus 36% in a single-specialty private practice.
More than 70% of the respondents said their case load fell by at least 75% during the initial surge of the pandemic; 80% turned to telemedicine, compared with just 23% who relied on the technology prior to the pandemic. The most commonly used telemedicine technologies were FaceTime, Zoom, Doximity, and Epic.
Mohs surgeons reported most commonly using telemedicine for postsurgery management (77% of the total 115 responses). “Telemedicine is a great fit for this category of visits as they allow the surgeon to view the surgical site and answer any questions they patient may have,” Dr. Maruthur said. “If the surgeon does suspect a postop infection or other concern based on a patient’s signs or symptoms, they can easily schedule the patient for an in-person assessment. We suspect that postop follow-up visits may be the best candidate for long-term use of telemedicine in Mohs surgery practices.”
Surgeons also reported using telemedicine for “spot checks” (61%) and surgical consultations (59%).
However, Dr. Maruther noted that preoperative assessments and spot checks can be difficult to perform using telemedicine. “The quality of the video image is not always great, patients can have a difficult time pointing the camera at the right spot and at the right distance. Even appreciating the actual size of the lesion are all difficult over a video encounter. And there is a lot of information gleaned from in-person physical examination, such as whether the lesion is fixed to a deeper structure and whether there are any nearby scars or other suspicious lesions.”
Nearly three-quarters of the surgeons using the technology said most or all patients were receptive to telemedicine.
However, the surgeons reported multiple barriers to the use of telemedicine: Limitations when compared with physical exams (88%), fitting it into the work flow (58%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
In an interview, Sumaira Z. Aasi, MD, director of Mohs and dermatologic surgery, Stanford University, agreed that there are many obstacles to routine use of telemedicine by Mohs surgeons. “As surgeons, we rely on the physical and tactile exam to get a sense of the size and extent of the cancer and characteristics such as the laxity of the surrounding tissue whether the tumor is fixed,” she said. “It is very difficult to access this on a telemedicine visit.”
In addition, she said, “many of our patients are in the elderly population, and some may not be comfortable using this technology. Also, it’s not a work flow that we are comfortable or familiar with. And I think that the technology has to improve to allow for better resolution of images as we ‘examine’ patients through a telemedicine visit.”
She added that “another con is there is a reliance on having the patient point out lesions of concern. Many cancers are picked by a careful in-person examination by a qualified physician/dermatologist/Mohs surgeon when the lesion is quite small or subtle and not even noticed by the patient themselves. This approach invariably leads to earlier biopsies and earlier treatments that can prevent morbidity and save health care money.”
On the other hand, she said, telemedicine “may save patients some time and money in terms of the effort and cost of transportation to come in for simpler postoperative medical visits that are often short in their very nature, such as postop check-ups.”
Most of the surgeons surveyed (69%) said telemedicine probably or definitely deserves a place in the practice Mohs surgery, but only 50% said they’d like to or would definitely pursue giving telemedicine a role in their practices once the pandemic is over.
“At the start of the pandemic, many regulations in areas such as HIPAA were eased, and reimbursements were increased, which allowed telemedicine to be quickly adopted,” Dr. Maruther said. “The government and payers have yet to decide which regulations and reimbursements will be in place after the pandemic. That makes it very difficult for surgeons to make long-term plans for implementing telemedicine in their practices.”
Dr. Aasi predicted that telemedicine will become more appealing to patients and physicians as it its technology and usability improves. More familiarity with its use will also be helpful, she said, and surgeons will be more receptive as it’s incorporated into efficient daily work flow.
The study was funded in part by the National Institutes of Health.
A majority of
A variety of factors combine to make it “very difficult for surgeons to make long-term plans for implementing telemedicine in their practices,” said Mario Maruthur, MD, who presented the findings at the annual meeting of the American College of Mohs Surgery. “Telemedicine likely has a role in Mohs practices, particularly with postop follow-up visits. However, postpandemic reimbursement and regulatory issues need to be formally laid out before Mohs surgeons are able to incorporate it into their permanent work flow.”
Dr. Maruthur, a Mohs surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York, and colleagues sent a survey to ACMS members in September and October 2020. “We saw first-hand in our surgical practice that telemedicine quickly became an important tool when the pandemic surged in the spring of 2020,” he said. Considering that surgical practices are highly dependent on in-person visits, the impetus for this study was to assess to what degree Mohs practices from across the spectrum, including academic and private practices, embraced telemedicine during the pandemic, and “what these surgical practices used telemedicine for, how it was received by their patients, which telemedicine platforms were most often utilized, and lastly, what are their plans if any for incorporating telemedicine into their surgical practices after the pandemic subsides.”
The researchers received responses from 115 surgeons representing all regions of the country (40% Northeast, 21% South, 21% Midwest, and 18% West). Half practiced in urban areas (37%) and large cities (13%), and 40% were in an academic setting versus 36% in a single-specialty private practice.
More than 70% of the respondents said their case load fell by at least 75% during the initial surge of the pandemic; 80% turned to telemedicine, compared with just 23% who relied on the technology prior to the pandemic. The most commonly used telemedicine technologies were FaceTime, Zoom, Doximity, and Epic.
Mohs surgeons reported most commonly using telemedicine for postsurgery management (77% of the total 115 responses). “Telemedicine is a great fit for this category of visits as they allow the surgeon to view the surgical site and answer any questions they patient may have,” Dr. Maruthur said. “If the surgeon does suspect a postop infection or other concern based on a patient’s signs or symptoms, they can easily schedule the patient for an in-person assessment. We suspect that postop follow-up visits may be the best candidate for long-term use of telemedicine in Mohs surgery practices.”
Surgeons also reported using telemedicine for “spot checks” (61%) and surgical consultations (59%).
However, Dr. Maruther noted that preoperative assessments and spot checks can be difficult to perform using telemedicine. “The quality of the video image is not always great, patients can have a difficult time pointing the camera at the right spot and at the right distance. Even appreciating the actual size of the lesion are all difficult over a video encounter. And there is a lot of information gleaned from in-person physical examination, such as whether the lesion is fixed to a deeper structure and whether there are any nearby scars or other suspicious lesions.”
Nearly three-quarters of the surgeons using the technology said most or all patients were receptive to telemedicine.
However, the surgeons reported multiple barriers to the use of telemedicine: Limitations when compared with physical exams (88%), fitting it into the work flow (58%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
In an interview, Sumaira Z. Aasi, MD, director of Mohs and dermatologic surgery, Stanford University, agreed that there are many obstacles to routine use of telemedicine by Mohs surgeons. “As surgeons, we rely on the physical and tactile exam to get a sense of the size and extent of the cancer and characteristics such as the laxity of the surrounding tissue whether the tumor is fixed,” she said. “It is very difficult to access this on a telemedicine visit.”
In addition, she said, “many of our patients are in the elderly population, and some may not be comfortable using this technology. Also, it’s not a work flow that we are comfortable or familiar with. And I think that the technology has to improve to allow for better resolution of images as we ‘examine’ patients through a telemedicine visit.”
She added that “another con is there is a reliance on having the patient point out lesions of concern. Many cancers are picked by a careful in-person examination by a qualified physician/dermatologist/Mohs surgeon when the lesion is quite small or subtle and not even noticed by the patient themselves. This approach invariably leads to earlier biopsies and earlier treatments that can prevent morbidity and save health care money.”
On the other hand, she said, telemedicine “may save patients some time and money in terms of the effort and cost of transportation to come in for simpler postoperative medical visits that are often short in their very nature, such as postop check-ups.”
Most of the surgeons surveyed (69%) said telemedicine probably or definitely deserves a place in the practice Mohs surgery, but only 50% said they’d like to or would definitely pursue giving telemedicine a role in their practices once the pandemic is over.
“At the start of the pandemic, many regulations in areas such as HIPAA were eased, and reimbursements were increased, which allowed telemedicine to be quickly adopted,” Dr. Maruther said. “The government and payers have yet to decide which regulations and reimbursements will be in place after the pandemic. That makes it very difficult for surgeons to make long-term plans for implementing telemedicine in their practices.”
Dr. Aasi predicted that telemedicine will become more appealing to patients and physicians as it its technology and usability improves. More familiarity with its use will also be helpful, she said, and surgeons will be more receptive as it’s incorporated into efficient daily work flow.
The study was funded in part by the National Institutes of Health.
A majority of
A variety of factors combine to make it “very difficult for surgeons to make long-term plans for implementing telemedicine in their practices,” said Mario Maruthur, MD, who presented the findings at the annual meeting of the American College of Mohs Surgery. “Telemedicine likely has a role in Mohs practices, particularly with postop follow-up visits. However, postpandemic reimbursement and regulatory issues need to be formally laid out before Mohs surgeons are able to incorporate it into their permanent work flow.”
Dr. Maruthur, a Mohs surgery and dermatologic oncology fellow at Memorial Sloan Kettering Cancer Center, New York, and colleagues sent a survey to ACMS members in September and October 2020. “We saw first-hand in our surgical practice that telemedicine quickly became an important tool when the pandemic surged in the spring of 2020,” he said. Considering that surgical practices are highly dependent on in-person visits, the impetus for this study was to assess to what degree Mohs practices from across the spectrum, including academic and private practices, embraced telemedicine during the pandemic, and “what these surgical practices used telemedicine for, how it was received by their patients, which telemedicine platforms were most often utilized, and lastly, what are their plans if any for incorporating telemedicine into their surgical practices after the pandemic subsides.”
The researchers received responses from 115 surgeons representing all regions of the country (40% Northeast, 21% South, 21% Midwest, and 18% West). Half practiced in urban areas (37%) and large cities (13%), and 40% were in an academic setting versus 36% in a single-specialty private practice.
More than 70% of the respondents said their case load fell by at least 75% during the initial surge of the pandemic; 80% turned to telemedicine, compared with just 23% who relied on the technology prior to the pandemic. The most commonly used telemedicine technologies were FaceTime, Zoom, Doximity, and Epic.
Mohs surgeons reported most commonly using telemedicine for postsurgery management (77% of the total 115 responses). “Telemedicine is a great fit for this category of visits as they allow the surgeon to view the surgical site and answer any questions they patient may have,” Dr. Maruthur said. “If the surgeon does suspect a postop infection or other concern based on a patient’s signs or symptoms, they can easily schedule the patient for an in-person assessment. We suspect that postop follow-up visits may be the best candidate for long-term use of telemedicine in Mohs surgery practices.”
Surgeons also reported using telemedicine for “spot checks” (61%) and surgical consultations (59%).
However, Dr. Maruther noted that preoperative assessments and spot checks can be difficult to perform using telemedicine. “The quality of the video image is not always great, patients can have a difficult time pointing the camera at the right spot and at the right distance. Even appreciating the actual size of the lesion are all difficult over a video encounter. And there is a lot of information gleaned from in-person physical examination, such as whether the lesion is fixed to a deeper structure and whether there are any nearby scars or other suspicious lesions.”
Nearly three-quarters of the surgeons using the technology said most or all patients were receptive to telemedicine.
However, the surgeons reported multiple barriers to the use of telemedicine: Limitations when compared with physical exams (88%), fitting it into the work flow (58%), patient response and training (57%), reimbursement concerns (50%), implementation of the technology (37%), regulations such as HIPAA (24%), training of staff (17%), and licensing (8%).
In an interview, Sumaira Z. Aasi, MD, director of Mohs and dermatologic surgery, Stanford University, agreed that there are many obstacles to routine use of telemedicine by Mohs surgeons. “As surgeons, we rely on the physical and tactile exam to get a sense of the size and extent of the cancer and characteristics such as the laxity of the surrounding tissue whether the tumor is fixed,” she said. “It is very difficult to access this on a telemedicine visit.”
In addition, she said, “many of our patients are in the elderly population, and some may not be comfortable using this technology. Also, it’s not a work flow that we are comfortable or familiar with. And I think that the technology has to improve to allow for better resolution of images as we ‘examine’ patients through a telemedicine visit.”
She added that “another con is there is a reliance on having the patient point out lesions of concern. Many cancers are picked by a careful in-person examination by a qualified physician/dermatologist/Mohs surgeon when the lesion is quite small or subtle and not even noticed by the patient themselves. This approach invariably leads to earlier biopsies and earlier treatments that can prevent morbidity and save health care money.”
On the other hand, she said, telemedicine “may save patients some time and money in terms of the effort and cost of transportation to come in for simpler postoperative medical visits that are often short in their very nature, such as postop check-ups.”
Most of the surgeons surveyed (69%) said telemedicine probably or definitely deserves a place in the practice Mohs surgery, but only 50% said they’d like to or would definitely pursue giving telemedicine a role in their practices once the pandemic is over.
“At the start of the pandemic, many regulations in areas such as HIPAA were eased, and reimbursements were increased, which allowed telemedicine to be quickly adopted,” Dr. Maruther said. “The government and payers have yet to decide which regulations and reimbursements will be in place after the pandemic. That makes it very difficult for surgeons to make long-term plans for implementing telemedicine in their practices.”
Dr. Aasi predicted that telemedicine will become more appealing to patients and physicians as it its technology and usability improves. More familiarity with its use will also be helpful, she said, and surgeons will be more receptive as it’s incorporated into efficient daily work flow.
The study was funded in part by the National Institutes of Health.
FROM THE ACMS ANNUAL MEETING
Updates in skin cancer from AAD VMX 2021
Anthony Rossi, MD, Assistant Attending Physician at Memorial Sloan Kettering Cancer Center, shares several updates in non-melanoma skin cancers emerging from the American Academy of Dermatology’s AAD VMX 2021 meeting.
With PD-1 inhibitors emerging as an immunotherapeutic option in this area, Dr. Rossi discusses a literature review that was conducted to compare the efficacy and safety of pembrolizumab, nivolumab, and cemiplimab in patients with advanced basal cell carcinoma or cutaneous squamous cell carcinoma.
Another review analyzed statin use as a predictor of more aggressive tumor characteristics in squamous cell carcinoma. Dr. Rossi shares that while there were no significant findings when examining the cohort as a whole, the male study population did see an elevated correlation between statin history and high-risk tumors.
Lastly, Dr. Rossi highlights an analysis of tumor burden in patients taking sonidegib 200 mg once daily as part of the 42-month BOLT study. Sonidegib demonstrated durable tumor response and substantial reduction in tumor burden, and safety and tolerability were consistent with earlier data.
--
Anthony Rossi, MD, Assistant Professor, Department of Dermatology, Weill Cornell Medical College; Assistant Attending Physician, Dermatology Service, Memorial Sloan Kettering Cancer Center, New York, NY
Anthony Rossi, MD, has disclosed the following relevant financial relationships:
Serve(d) as a board member, director, officer, partner, employee, advisor, consultant, or trustee for: DAR Inc; American Society for Dermatologic Surgery Association.
Received research grant from: Regeneron; Biofrontera; Memorial Sloan Kettering Society; Skin Cancer Foundation.
Have a 5% or greater equity interest in: DAR Inc.
Received income in an amount equal to or greater than $250 from: Allergan; Regeneron; Evolus; Cutera; Biofrontera; Iam; DynaMed; Canfield; Merz.
Anthony Rossi, MD, Assistant Attending Physician at Memorial Sloan Kettering Cancer Center, shares several updates in non-melanoma skin cancers emerging from the American Academy of Dermatology’s AAD VMX 2021 meeting.
With PD-1 inhibitors emerging as an immunotherapeutic option in this area, Dr. Rossi discusses a literature review that was conducted to compare the efficacy and safety of pembrolizumab, nivolumab, and cemiplimab in patients with advanced basal cell carcinoma or cutaneous squamous cell carcinoma.
Another review analyzed statin use as a predictor of more aggressive tumor characteristics in squamous cell carcinoma. Dr. Rossi shares that while there were no significant findings when examining the cohort as a whole, the male study population did see an elevated correlation between statin history and high-risk tumors.
Lastly, Dr. Rossi highlights an analysis of tumor burden in patients taking sonidegib 200 mg once daily as part of the 42-month BOLT study. Sonidegib demonstrated durable tumor response and substantial reduction in tumor burden, and safety and tolerability were consistent with earlier data.
--
Anthony Rossi, MD, Assistant Professor, Department of Dermatology, Weill Cornell Medical College; Assistant Attending Physician, Dermatology Service, Memorial Sloan Kettering Cancer Center, New York, NY
Anthony Rossi, MD, has disclosed the following relevant financial relationships:
Serve(d) as a board member, director, officer, partner, employee, advisor, consultant, or trustee for: DAR Inc; American Society for Dermatologic Surgery Association.
Received research grant from: Regeneron; Biofrontera; Memorial Sloan Kettering Society; Skin Cancer Foundation.
Have a 5% or greater equity interest in: DAR Inc.
Received income in an amount equal to or greater than $250 from: Allergan; Regeneron; Evolus; Cutera; Biofrontera; Iam; DynaMed; Canfield; Merz.
Anthony Rossi, MD, Assistant Attending Physician at Memorial Sloan Kettering Cancer Center, shares several updates in non-melanoma skin cancers emerging from the American Academy of Dermatology’s AAD VMX 2021 meeting.
With PD-1 inhibitors emerging as an immunotherapeutic option in this area, Dr. Rossi discusses a literature review that was conducted to compare the efficacy and safety of pembrolizumab, nivolumab, and cemiplimab in patients with advanced basal cell carcinoma or cutaneous squamous cell carcinoma.
Another review analyzed statin use as a predictor of more aggressive tumor characteristics in squamous cell carcinoma. Dr. Rossi shares that while there were no significant findings when examining the cohort as a whole, the male study population did see an elevated correlation between statin history and high-risk tumors.
Lastly, Dr. Rossi highlights an analysis of tumor burden in patients taking sonidegib 200 mg once daily as part of the 42-month BOLT study. Sonidegib demonstrated durable tumor response and substantial reduction in tumor burden, and safety and tolerability were consistent with earlier data.
--
Anthony Rossi, MD, Assistant Professor, Department of Dermatology, Weill Cornell Medical College; Assistant Attending Physician, Dermatology Service, Memorial Sloan Kettering Cancer Center, New York, NY
Anthony Rossi, MD, has disclosed the following relevant financial relationships:
Serve(d) as a board member, director, officer, partner, employee, advisor, consultant, or trustee for: DAR Inc; American Society for Dermatologic Surgery Association.
Received research grant from: Regeneron; Biofrontera; Memorial Sloan Kettering Society; Skin Cancer Foundation.
Have a 5% or greater equity interest in: DAR Inc.
Received income in an amount equal to or greater than $250 from: Allergan; Regeneron; Evolus; Cutera; Biofrontera; Iam; DynaMed; Canfield; Merz.

New guideline provides recommendations on reconstruction after skin cancer resection
You’ve successfully resected a skin cancer lesion, leaving clear margins. Now what?
That’s
The guideline – a joint effort of the American Society of Plastic Surgeons, American Society for Dermatologic Surgery, American Academy of Dermatology, American Academy of Facial Plastic and Reconstructive Surgery, American Academy of Otolaryngology – Head and Neck Surgery Foundation, American College of Mohs Surgery, American Society for Mohs Surgery, and American Society of Ophthalmic Plastic and Reconstructive Surgery – was published online in the Journal of the American Academy of Dermatology.
From the outset, the panel members realized that to keep the guideline manageable they had to limit recommendations to the practice of reconstruction defined as “cutaneous closure that requires a flap, graft, or tissue rearrangement.”
Other wound closure methods, such as secondary intention healing; simple closures; and complex closures that do not involve flaps, grafts, muscle, or bone, were not covered in the recommendations.
As with similar guidelines, the developers selected seven clinical questions to be addressed, and attempted to find consensus through literature searches, appraisal of the evidence, grading of recommendations, peer review, and public comment.
“We had a very heterogeneous set of things that we were trying to comment on, so we had to keep things somewhat generic,” lead author Andrew Chen, MD, chief of the division of plastic surgery, at the University of Connecticut Health Center, Farmington, said in an interview.
“Skin cancer and reconstruction affect different body areas and areas of different sizes. When we were creating the guidelines, we had to tailor the questions we could ask based on things that would make sense to answer, because obviously we couldn’t ask a question such as: ‘What’s better, a skin graft or a flap?’ Well, there are some things you can’t put a skin graft on – it won’t last, so we couldn’t ask that kind of question,” Dr. Chen said.
Curtis Cetrulo, MD, a plastic and reconstructive surgeon at Massachusetts General Hospital, Boston, who was not involved in the guideline process, said in an interview that the broad recommendations are in keeping with his practice and experience. He also acknowledged, however, the difficulty in creating a guideline that covers the complexity and heterogeneity of reconstructive surgery.
“These are generally good recommendations, but they’re recommendations only, with generally weak levels of evidence. What we really need are clinical trials that can give us definitive answers to some of these questions,” he said.
Recommendations
The seven key recommendations, based on the clinical questions raised, are summarized below:
- Delayed (asynchronous) reconstruction is acceptable. Although the quality of the evidence is low and the recommendations are listed as an option, the guideline authors said that depending on the situation, reconstruction can be performed either immediately after resection or delayed by days, weeks, “or even months.”
- Systemic antibiotics should not be routinely prescribed in the interim between resection and reconstruction in adults. Here too, the evidence is low and the recommendation strength is weak, but in “the absence of data showing convincing benefits, systemic antibiotic therapy does not appear necessary or desirable in most cases when there is an interval between cancer resection and reconstruction,” the work group wrote.
- Clinicians may administer perioperative systemic antibiotics in a facility-based setting for adults undergoing reconstruction (3a), but antibiotics should not be routinely prescribed in an office-based setting (3b). The rationale for these recommendations, supported by a moderate level of evidence, is that the risk of surgical-site infection is generally higher in facilities, compared with an office-based setting. Patients who undergo reconstruction in hospitals or surgical centers are more likely to have complex reconstructions or have risks that may make them suitable candidates for antibiotics, but patients in office-based setting may often be spared from the additional costs, side effects, and possible drug interactions from antibiotic use. “There is no evidence in either setting that long-term antibiotic prophylaxis provides infection risk reduction, compared with short-term prophylaxis,” the guideline working group wrote.
- Continue anticoagulant, antithrombotic, and antiplatelet medications for adult patients undergoing reconstruction after skin cancer resection in the office-based setting (4a), and in the facility-based setting should coordinate with the physician managing anticoagulation before modifying the medication prior to surgery (4b). Evidence quality and recommendation strength are both moderate.
- The guideline authors recommend against routine prescription of narcotics as first-line treatment for pain in adults undergoing skin reconstruction (5a), favoring instead acetaminophen and NSAIDs as first-line therapy (5b). Evidence quality and recommendation strength are both moderate.
- In the absence of standardized protocols for the management of pain medications, oral antibiotics, and/or anticoagulants in the perioperative period, clinicians should discuss possible approaches with adult patients. “Educating patients about their perioperative treatment through discussion of treatment strategies may help alleviate anxiety, improve communication, increase patient satisfaction, and maximize patient compliance with the postoperative orders,” the guideline authors wrote.
- The authors suggest that adult patients may be offered follow-up assessments to discuss functional and cosmetic outcomes. “The return of the patient for follow-up visits is an excellent opportunity to better understand and measure these outcomes, improve patient-physician communication, and foster quality improvement. Postoperative follow-up can lead to increased communication between the patient and physician, thereby empowering patients to comment on satisfaction and other important outcomes measures,” they wrote.
What’s next
The guideline developers acknowledged that data are limited regarding reconstructive surgery following skin cancer resection, and that higher-quality studies would help to improve future guidelines. Dr. Chen said that greater use of prospective surgical databases and more systematic collection of patient-reported outcomes could inform further efforts.
The guideline development process was supported by the various groups represented. Dr. Chen and Dr. Cetrulo reported no relevant disclosures.
You’ve successfully resected a skin cancer lesion, leaving clear margins. Now what?
That’s
The guideline – a joint effort of the American Society of Plastic Surgeons, American Society for Dermatologic Surgery, American Academy of Dermatology, American Academy of Facial Plastic and Reconstructive Surgery, American Academy of Otolaryngology – Head and Neck Surgery Foundation, American College of Mohs Surgery, American Society for Mohs Surgery, and American Society of Ophthalmic Plastic and Reconstructive Surgery – was published online in the Journal of the American Academy of Dermatology.
From the outset, the panel members realized that to keep the guideline manageable they had to limit recommendations to the practice of reconstruction defined as “cutaneous closure that requires a flap, graft, or tissue rearrangement.”
Other wound closure methods, such as secondary intention healing; simple closures; and complex closures that do not involve flaps, grafts, muscle, or bone, were not covered in the recommendations.
As with similar guidelines, the developers selected seven clinical questions to be addressed, and attempted to find consensus through literature searches, appraisal of the evidence, grading of recommendations, peer review, and public comment.
“We had a very heterogeneous set of things that we were trying to comment on, so we had to keep things somewhat generic,” lead author Andrew Chen, MD, chief of the division of plastic surgery, at the University of Connecticut Health Center, Farmington, said in an interview.
“Skin cancer and reconstruction affect different body areas and areas of different sizes. When we were creating the guidelines, we had to tailor the questions we could ask based on things that would make sense to answer, because obviously we couldn’t ask a question such as: ‘What’s better, a skin graft or a flap?’ Well, there are some things you can’t put a skin graft on – it won’t last, so we couldn’t ask that kind of question,” Dr. Chen said.
Curtis Cetrulo, MD, a plastic and reconstructive surgeon at Massachusetts General Hospital, Boston, who was not involved in the guideline process, said in an interview that the broad recommendations are in keeping with his practice and experience. He also acknowledged, however, the difficulty in creating a guideline that covers the complexity and heterogeneity of reconstructive surgery.
“These are generally good recommendations, but they’re recommendations only, with generally weak levels of evidence. What we really need are clinical trials that can give us definitive answers to some of these questions,” he said.
Recommendations
The seven key recommendations, based on the clinical questions raised, are summarized below:
- Delayed (asynchronous) reconstruction is acceptable. Although the quality of the evidence is low and the recommendations are listed as an option, the guideline authors said that depending on the situation, reconstruction can be performed either immediately after resection or delayed by days, weeks, “or even months.”
- Systemic antibiotics should not be routinely prescribed in the interim between resection and reconstruction in adults. Here too, the evidence is low and the recommendation strength is weak, but in “the absence of data showing convincing benefits, systemic antibiotic therapy does not appear necessary or desirable in most cases when there is an interval between cancer resection and reconstruction,” the work group wrote.
- Clinicians may administer perioperative systemic antibiotics in a facility-based setting for adults undergoing reconstruction (3a), but antibiotics should not be routinely prescribed in an office-based setting (3b). The rationale for these recommendations, supported by a moderate level of evidence, is that the risk of surgical-site infection is generally higher in facilities, compared with an office-based setting. Patients who undergo reconstruction in hospitals or surgical centers are more likely to have complex reconstructions or have risks that may make them suitable candidates for antibiotics, but patients in office-based setting may often be spared from the additional costs, side effects, and possible drug interactions from antibiotic use. “There is no evidence in either setting that long-term antibiotic prophylaxis provides infection risk reduction, compared with short-term prophylaxis,” the guideline working group wrote.
- Continue anticoagulant, antithrombotic, and antiplatelet medications for adult patients undergoing reconstruction after skin cancer resection in the office-based setting (4a), and in the facility-based setting should coordinate with the physician managing anticoagulation before modifying the medication prior to surgery (4b). Evidence quality and recommendation strength are both moderate.
- The guideline authors recommend against routine prescription of narcotics as first-line treatment for pain in adults undergoing skin reconstruction (5a), favoring instead acetaminophen and NSAIDs as first-line therapy (5b). Evidence quality and recommendation strength are both moderate.
- In the absence of standardized protocols for the management of pain medications, oral antibiotics, and/or anticoagulants in the perioperative period, clinicians should discuss possible approaches with adult patients. “Educating patients about their perioperative treatment through discussion of treatment strategies may help alleviate anxiety, improve communication, increase patient satisfaction, and maximize patient compliance with the postoperative orders,” the guideline authors wrote.
- The authors suggest that adult patients may be offered follow-up assessments to discuss functional and cosmetic outcomes. “The return of the patient for follow-up visits is an excellent opportunity to better understand and measure these outcomes, improve patient-physician communication, and foster quality improvement. Postoperative follow-up can lead to increased communication between the patient and physician, thereby empowering patients to comment on satisfaction and other important outcomes measures,” they wrote.
What’s next
The guideline developers acknowledged that data are limited regarding reconstructive surgery following skin cancer resection, and that higher-quality studies would help to improve future guidelines. Dr. Chen said that greater use of prospective surgical databases and more systematic collection of patient-reported outcomes could inform further efforts.
The guideline development process was supported by the various groups represented. Dr. Chen and Dr. Cetrulo reported no relevant disclosures.
You’ve successfully resected a skin cancer lesion, leaving clear margins. Now what?
That’s
The guideline – a joint effort of the American Society of Plastic Surgeons, American Society for Dermatologic Surgery, American Academy of Dermatology, American Academy of Facial Plastic and Reconstructive Surgery, American Academy of Otolaryngology – Head and Neck Surgery Foundation, American College of Mohs Surgery, American Society for Mohs Surgery, and American Society of Ophthalmic Plastic and Reconstructive Surgery – was published online in the Journal of the American Academy of Dermatology.
From the outset, the panel members realized that to keep the guideline manageable they had to limit recommendations to the practice of reconstruction defined as “cutaneous closure that requires a flap, graft, or tissue rearrangement.”
Other wound closure methods, such as secondary intention healing; simple closures; and complex closures that do not involve flaps, grafts, muscle, or bone, were not covered in the recommendations.
As with similar guidelines, the developers selected seven clinical questions to be addressed, and attempted to find consensus through literature searches, appraisal of the evidence, grading of recommendations, peer review, and public comment.
“We had a very heterogeneous set of things that we were trying to comment on, so we had to keep things somewhat generic,” lead author Andrew Chen, MD, chief of the division of plastic surgery, at the University of Connecticut Health Center, Farmington, said in an interview.
“Skin cancer and reconstruction affect different body areas and areas of different sizes. When we were creating the guidelines, we had to tailor the questions we could ask based on things that would make sense to answer, because obviously we couldn’t ask a question such as: ‘What’s better, a skin graft or a flap?’ Well, there are some things you can’t put a skin graft on – it won’t last, so we couldn’t ask that kind of question,” Dr. Chen said.
Curtis Cetrulo, MD, a plastic and reconstructive surgeon at Massachusetts General Hospital, Boston, who was not involved in the guideline process, said in an interview that the broad recommendations are in keeping with his practice and experience. He also acknowledged, however, the difficulty in creating a guideline that covers the complexity and heterogeneity of reconstructive surgery.
“These are generally good recommendations, but they’re recommendations only, with generally weak levels of evidence. What we really need are clinical trials that can give us definitive answers to some of these questions,” he said.
Recommendations
The seven key recommendations, based on the clinical questions raised, are summarized below:
- Delayed (asynchronous) reconstruction is acceptable. Although the quality of the evidence is low and the recommendations are listed as an option, the guideline authors said that depending on the situation, reconstruction can be performed either immediately after resection or delayed by days, weeks, “or even months.”
- Systemic antibiotics should not be routinely prescribed in the interim between resection and reconstruction in adults. Here too, the evidence is low and the recommendation strength is weak, but in “the absence of data showing convincing benefits, systemic antibiotic therapy does not appear necessary or desirable in most cases when there is an interval between cancer resection and reconstruction,” the work group wrote.
- Clinicians may administer perioperative systemic antibiotics in a facility-based setting for adults undergoing reconstruction (3a), but antibiotics should not be routinely prescribed in an office-based setting (3b). The rationale for these recommendations, supported by a moderate level of evidence, is that the risk of surgical-site infection is generally higher in facilities, compared with an office-based setting. Patients who undergo reconstruction in hospitals or surgical centers are more likely to have complex reconstructions or have risks that may make them suitable candidates for antibiotics, but patients in office-based setting may often be spared from the additional costs, side effects, and possible drug interactions from antibiotic use. “There is no evidence in either setting that long-term antibiotic prophylaxis provides infection risk reduction, compared with short-term prophylaxis,” the guideline working group wrote.
- Continue anticoagulant, antithrombotic, and antiplatelet medications for adult patients undergoing reconstruction after skin cancer resection in the office-based setting (4a), and in the facility-based setting should coordinate with the physician managing anticoagulation before modifying the medication prior to surgery (4b). Evidence quality and recommendation strength are both moderate.
- The guideline authors recommend against routine prescription of narcotics as first-line treatment for pain in adults undergoing skin reconstruction (5a), favoring instead acetaminophen and NSAIDs as first-line therapy (5b). Evidence quality and recommendation strength are both moderate.
- In the absence of standardized protocols for the management of pain medications, oral antibiotics, and/or anticoagulants in the perioperative period, clinicians should discuss possible approaches with adult patients. “Educating patients about their perioperative treatment through discussion of treatment strategies may help alleviate anxiety, improve communication, increase patient satisfaction, and maximize patient compliance with the postoperative orders,” the guideline authors wrote.
- The authors suggest that adult patients may be offered follow-up assessments to discuss functional and cosmetic outcomes. “The return of the patient for follow-up visits is an excellent opportunity to better understand and measure these outcomes, improve patient-physician communication, and foster quality improvement. Postoperative follow-up can lead to increased communication between the patient and physician, thereby empowering patients to comment on satisfaction and other important outcomes measures,” they wrote.
What’s next
The guideline developers acknowledged that data are limited regarding reconstructive surgery following skin cancer resection, and that higher-quality studies would help to improve future guidelines. Dr. Chen said that greater use of prospective surgical databases and more systematic collection of patient-reported outcomes could inform further efforts.
The guideline development process was supported by the various groups represented. Dr. Chen and Dr. Cetrulo reported no relevant disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Mohs Micrographic Surgery During the COVID-19 Pandemic: Considering the Patient Perspective
Guidelines on Skin Cancer Surgeries During the COVID-19 Pandemic
At the start of the COVID-19 pandemic, the Centers for Disease Control and Prevention issued recommendations to decrease the spread of SARS-CoV-2 and optimize the use of personal protective equipment (PPE) for frontline workers.1 In the field of dermatologic surgery, the American College of Mohs Surgery, the National Comprehensive Cancer Network, the American Society for Dermatologic Surgery, and the American Academy of Dermatology made recommendations to postpone nonessential and nonurgent procedures.2-4 The initial guidelines of the American College of Mohs Surgery advised cancellation of all elective surgeries and deferred treatment of most cases of basal cell carcinoma for as long as 3 months; low-risk squamous cell carcinoma (SCC) and melanoma in situ treatment was deferred for as long as 2 or 3 months.3 Additional recommendations were made to reserve inpatient visits for suspicious lesions and high-risk cancers, postpone other nonessential and nonurgent appointments, and utilize telemedicine whenever possible.5
These recommendations led to great uncertainty and stress for patients and providers. Although numerous important variables, such as patient risk factors, severity of disease, availability of PPE and staff, and patient-to-provider transmission were considered when creating these guidelines, the patient’s experience likely was not a contributing factor.
COVID-19 Transmission During Mohs Surgery
There have been concerns that surgeons performing Mohs micrographic surgery (MMS) might be at an increased risk for COVID-19, given their close contact with high-risk sites (ie, nose, mouth) and cautery-generated aerosols; most of the estimated transmission risk associated with MMS has been based on head and neck surgery experience and publications.6-8 Tee and colleagues9 recently published their institution’s MMS COVID-19 preventive measures, which, to their knowledge, have prevented all intraoperative transmission of SARS-CoV-2, even in disease-positive patients. Currently, evidence is lacking to support a high risk for SARS-CoV-2 transmission during MMS when proper PPE and personal hygiene measures as well as strict infection control protocols—presurgical COVID-19 testing in high-risk cases, COVID-19 screening optimization, visitor restrictions, and appropriate disinfection between patients—are in place.
The Impact of Postponing Treatment on Patients
Although studies have focused on the effects of the COVID-19 pandemic on physicians practicing MMS,10 little is known about the effects of delays in skin cancer treatment on patients. A survey conducted in the United Kingdom investigating the patient’s perspective found that patients expressed worry and concern about the possibility that their MMS would be postponed and greatly appreciated continuation of treatment during the pandemic.11
Other medical specialties have reported their patient experiences during the pandemic. In a study examining patient perception of postponed surgical treatment of pelvic floor disorders due to COVID-19, nearly half of survey respondents were unhappy with the delay in receiving care. Furthermore, patients who reported being unhappy were more likely to report feelings of isolation and anxiety because their surgery was postponed.12 In another study involving patients with lung cancer, 9.1% (N=15) of patients postponed their treatment during the COVID-19 pandemic because of pandemic-related anxiety.13
With the goal of improving care at our institution, we conducted a brief institutional review board–approved survey to evaluate how postponing MMS treatment due to the COVID-19 pandemic affected patients. All MMS patients undergoing surgery in June 2020 and July 2020 (N=99) were asked to complete our voluntary and anonymous 23-question survey in person during their procedure. We obtained 88 responses (response rate, 89%). Twenty percent of surveyed patients (n=18) reported that their MMS had been postponed; 78% of those whose MMS was postponed (n=14) indicated some level of anxiety during the waiting period. It was unclear which patients had their treatment postponed based on national guidelines and which ones elected to postpone surgery.
Tips for Health Care Providers
Patient-provider communication highlighting specific skin cancer risk and the risk vs benefit of postponing treatment might reduce anxiety and stress during the waiting period.14 A study found that COVID-19 posed a bigger threat than most noninvasive skin cancers; therefore, the authors of that study concluded that treatment for most skin cancers could be safely postponed.15 Specifically, those authors recommended prioritizing treatment for Merkel cell carcinoma, invasive SCC, and melanoma with positive margins or macroscopic residual disease. They proposed that all other skin cancers, including basal cell carcinoma, SCC in situ, and melanoma with negative margins and no macroscopic residual disease, could be safely delayed for as long as 3 months.15
For patients with multiple risk factors for COVID-19–related morbidity or mortality, delaying skin cancer treatment likely has less risk than contracting the virus.15 This information should be communicated with patients. Investigation of specific patient concerns is warranted, and case-by-case evaluation of patients’ risk factors and skin cancer risk should be considered.
Based on the current, though limited, literature, delaying medical treatment can have a negative impact on the patient experience. Furthermore, proper precautions have been shown to limit intraoperative transmission of SARS-CoV-2 during MMS, but research is lacking. Practitioners should utilize shared decision-making and evaluate a given patient’s risk factors and concerns when deciding whether to postpone treatment. We encourage other institutions to evaluate the effects that delaying MMS has had on their patients, as further studies would improve understanding of patients’ experiences during a pandemic and potentially influence future dermatology guidelines.
- Center for Disease Control and Prevention. COVID-19. Accessed April 20, 2021. https://www.cdc.gov/coronavirus/2019-ncov/index.html
- American College of Mohs Surgery. Mohs surgery ambulatory protocol during COVID pandemic (version 6-3-20). June 4, 2020. Accessed April 20, 2021. http://staging.mohscollege.org/UserFiles/AM20/Member%20Alert/MohsSurgeryAmbulatoryProtocolDuringCOVIDPandemicFinal.pdf
- COVID-19 resources. National Comprehensive Cancer Network website. Accessed April 20, 2021. https://www.nccn.org/covid-19
- Narla S, Alam M, Ozog DM, et al. American Society of Dermatologic Surgery Association (ASDSA) and American Society for Laser Medicine & Surgery (ASLMS) guidance for cosmetic dermatology practices during COVID-19. Updated January 11, 2021. Accessed April 10, 2021. https://www.asds.net/Portals/0/PDF/asdsa/asdsa-aslms-cosmetic-reopening-guidance.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic.J Am Acad Dermatol. 2020;83:295-296. doi:10.1016/j.jaad.2020.05.002
- Yuan JT, Jiang SIB. Urgent safety considerations for dermatologic surgeons in the COVID-19 pandemic. Dermatol Online J. 2020;26:1. Accessed April 20, 2021. http://escholarship.org/uc/item/2qr3w771
- Otolaryngologists may contract COVID-19 during surgery. ENTtoday. March 20, 2020. Accessed April 20, 2021. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/
- Howard BE. High-risk aerosol-generating procedures in COVID-19: respiratory protective equipment considerations. Otolaryngol Head Neck Surg. 2020;163:98-103. doi:10.1177/0194599820927335
- Tee MW, Stewart C, Aliessa S, et al. Dermatological surgery during the COVID-19 pandemic: experience of a large academic center. J Am Acad Dermatol. 2021;84:1094-1096. doi:10.1016/j.jaad.2020.12.003
- Hooper J, Feng H. The impact of COVID-19 on micrographic surgery and dermatologic oncology fellows. Dermatol Surg. 2020;46:1762-1763. doi:10.1097/DSS.0000000000002766
- Nicholson P, Ali FR, Patalay R, et al. Patient perceptions of Mohs micrographic surgery during the COVID-19 pandemic and lessons for the next outbreak. Clin Exp Dermatol. 2021;46:179-180. doi:10.1111/ced.14423
- Mou T, Brown O, Gillingham A, et al. Patients’ perceptions on surgical care suspension for pelvic floor disorders during the COVID-19 pandemic. Female Pelvic Med Reconstr Surg. 2020;26:477-482. doi:10.1097/SPV.0000000000000918
- Fujita K, Ito T, Saito Z, et al. Impact of COVID-19 pandemic on lung cancer treatment scheduling. Thorac Cancer. 2020;11:2983-2986. doi:10.1111/1759-7714.13615
- Nikumb VB, Banerjee A, Kaur G, et al. Impact of doctor-patient communication on preoperative anxiety: study at industrial township, Pimpri, Pune. Ind Psychiatry J. 2009;18:19-21. doi:10.4103/0972-6748.57852
- Baumann BC, MacArthur KM, Brewer JD, et al. Management of primary skin cancer during a pandemic: multidisciplinary recommendations. Cancer. 2020;126:3900-3906. doi:10.1002/cncr.32969
Guidelines on Skin Cancer Surgeries During the COVID-19 Pandemic
At the start of the COVID-19 pandemic, the Centers for Disease Control and Prevention issued recommendations to decrease the spread of SARS-CoV-2 and optimize the use of personal protective equipment (PPE) for frontline workers.1 In the field of dermatologic surgery, the American College of Mohs Surgery, the National Comprehensive Cancer Network, the American Society for Dermatologic Surgery, and the American Academy of Dermatology made recommendations to postpone nonessential and nonurgent procedures.2-4 The initial guidelines of the American College of Mohs Surgery advised cancellation of all elective surgeries and deferred treatment of most cases of basal cell carcinoma for as long as 3 months; low-risk squamous cell carcinoma (SCC) and melanoma in situ treatment was deferred for as long as 2 or 3 months.3 Additional recommendations were made to reserve inpatient visits for suspicious lesions and high-risk cancers, postpone other nonessential and nonurgent appointments, and utilize telemedicine whenever possible.5
These recommendations led to great uncertainty and stress for patients and providers. Although numerous important variables, such as patient risk factors, severity of disease, availability of PPE and staff, and patient-to-provider transmission were considered when creating these guidelines, the patient’s experience likely was not a contributing factor.
COVID-19 Transmission During Mohs Surgery
There have been concerns that surgeons performing Mohs micrographic surgery (MMS) might be at an increased risk for COVID-19, given their close contact with high-risk sites (ie, nose, mouth) and cautery-generated aerosols; most of the estimated transmission risk associated with MMS has been based on head and neck surgery experience and publications.6-8 Tee and colleagues9 recently published their institution’s MMS COVID-19 preventive measures, which, to their knowledge, have prevented all intraoperative transmission of SARS-CoV-2, even in disease-positive patients. Currently, evidence is lacking to support a high risk for SARS-CoV-2 transmission during MMS when proper PPE and personal hygiene measures as well as strict infection control protocols—presurgical COVID-19 testing in high-risk cases, COVID-19 screening optimization, visitor restrictions, and appropriate disinfection between patients—are in place.
The Impact of Postponing Treatment on Patients
Although studies have focused on the effects of the COVID-19 pandemic on physicians practicing MMS,10 little is known about the effects of delays in skin cancer treatment on patients. A survey conducted in the United Kingdom investigating the patient’s perspective found that patients expressed worry and concern about the possibility that their MMS would be postponed and greatly appreciated continuation of treatment during the pandemic.11
Other medical specialties have reported their patient experiences during the pandemic. In a study examining patient perception of postponed surgical treatment of pelvic floor disorders due to COVID-19, nearly half of survey respondents were unhappy with the delay in receiving care. Furthermore, patients who reported being unhappy were more likely to report feelings of isolation and anxiety because their surgery was postponed.12 In another study involving patients with lung cancer, 9.1% (N=15) of patients postponed their treatment during the COVID-19 pandemic because of pandemic-related anxiety.13
With the goal of improving care at our institution, we conducted a brief institutional review board–approved survey to evaluate how postponing MMS treatment due to the COVID-19 pandemic affected patients. All MMS patients undergoing surgery in June 2020 and July 2020 (N=99) were asked to complete our voluntary and anonymous 23-question survey in person during their procedure. We obtained 88 responses (response rate, 89%). Twenty percent of surveyed patients (n=18) reported that their MMS had been postponed; 78% of those whose MMS was postponed (n=14) indicated some level of anxiety during the waiting period. It was unclear which patients had their treatment postponed based on national guidelines and which ones elected to postpone surgery.
Tips for Health Care Providers
Patient-provider communication highlighting specific skin cancer risk and the risk vs benefit of postponing treatment might reduce anxiety and stress during the waiting period.14 A study found that COVID-19 posed a bigger threat than most noninvasive skin cancers; therefore, the authors of that study concluded that treatment for most skin cancers could be safely postponed.15 Specifically, those authors recommended prioritizing treatment for Merkel cell carcinoma, invasive SCC, and melanoma with positive margins or macroscopic residual disease. They proposed that all other skin cancers, including basal cell carcinoma, SCC in situ, and melanoma with negative margins and no macroscopic residual disease, could be safely delayed for as long as 3 months.15
For patients with multiple risk factors for COVID-19–related morbidity or mortality, delaying skin cancer treatment likely has less risk than contracting the virus.15 This information should be communicated with patients. Investigation of specific patient concerns is warranted, and case-by-case evaluation of patients’ risk factors and skin cancer risk should be considered.
Based on the current, though limited, literature, delaying medical treatment can have a negative impact on the patient experience. Furthermore, proper precautions have been shown to limit intraoperative transmission of SARS-CoV-2 during MMS, but research is lacking. Practitioners should utilize shared decision-making and evaluate a given patient’s risk factors and concerns when deciding whether to postpone treatment. We encourage other institutions to evaluate the effects that delaying MMS has had on their patients, as further studies would improve understanding of patients’ experiences during a pandemic and potentially influence future dermatology guidelines.
Guidelines on Skin Cancer Surgeries During the COVID-19 Pandemic
At the start of the COVID-19 pandemic, the Centers for Disease Control and Prevention issued recommendations to decrease the spread of SARS-CoV-2 and optimize the use of personal protective equipment (PPE) for frontline workers.1 In the field of dermatologic surgery, the American College of Mohs Surgery, the National Comprehensive Cancer Network, the American Society for Dermatologic Surgery, and the American Academy of Dermatology made recommendations to postpone nonessential and nonurgent procedures.2-4 The initial guidelines of the American College of Mohs Surgery advised cancellation of all elective surgeries and deferred treatment of most cases of basal cell carcinoma for as long as 3 months; low-risk squamous cell carcinoma (SCC) and melanoma in situ treatment was deferred for as long as 2 or 3 months.3 Additional recommendations were made to reserve inpatient visits for suspicious lesions and high-risk cancers, postpone other nonessential and nonurgent appointments, and utilize telemedicine whenever possible.5
These recommendations led to great uncertainty and stress for patients and providers. Although numerous important variables, such as patient risk factors, severity of disease, availability of PPE and staff, and patient-to-provider transmission were considered when creating these guidelines, the patient’s experience likely was not a contributing factor.
COVID-19 Transmission During Mohs Surgery
There have been concerns that surgeons performing Mohs micrographic surgery (MMS) might be at an increased risk for COVID-19, given their close contact with high-risk sites (ie, nose, mouth) and cautery-generated aerosols; most of the estimated transmission risk associated with MMS has been based on head and neck surgery experience and publications.6-8 Tee and colleagues9 recently published their institution’s MMS COVID-19 preventive measures, which, to their knowledge, have prevented all intraoperative transmission of SARS-CoV-2, even in disease-positive patients. Currently, evidence is lacking to support a high risk for SARS-CoV-2 transmission during MMS when proper PPE and personal hygiene measures as well as strict infection control protocols—presurgical COVID-19 testing in high-risk cases, COVID-19 screening optimization, visitor restrictions, and appropriate disinfection between patients—are in place.
The Impact of Postponing Treatment on Patients
Although studies have focused on the effects of the COVID-19 pandemic on physicians practicing MMS,10 little is known about the effects of delays in skin cancer treatment on patients. A survey conducted in the United Kingdom investigating the patient’s perspective found that patients expressed worry and concern about the possibility that their MMS would be postponed and greatly appreciated continuation of treatment during the pandemic.11
Other medical specialties have reported their patient experiences during the pandemic. In a study examining patient perception of postponed surgical treatment of pelvic floor disorders due to COVID-19, nearly half of survey respondents were unhappy with the delay in receiving care. Furthermore, patients who reported being unhappy were more likely to report feelings of isolation and anxiety because their surgery was postponed.12 In another study involving patients with lung cancer, 9.1% (N=15) of patients postponed their treatment during the COVID-19 pandemic because of pandemic-related anxiety.13
With the goal of improving care at our institution, we conducted a brief institutional review board–approved survey to evaluate how postponing MMS treatment due to the COVID-19 pandemic affected patients. All MMS patients undergoing surgery in June 2020 and July 2020 (N=99) were asked to complete our voluntary and anonymous 23-question survey in person during their procedure. We obtained 88 responses (response rate, 89%). Twenty percent of surveyed patients (n=18) reported that their MMS had been postponed; 78% of those whose MMS was postponed (n=14) indicated some level of anxiety during the waiting period. It was unclear which patients had their treatment postponed based on national guidelines and which ones elected to postpone surgery.
Tips for Health Care Providers
Patient-provider communication highlighting specific skin cancer risk and the risk vs benefit of postponing treatment might reduce anxiety and stress during the waiting period.14 A study found that COVID-19 posed a bigger threat than most noninvasive skin cancers; therefore, the authors of that study concluded that treatment for most skin cancers could be safely postponed.15 Specifically, those authors recommended prioritizing treatment for Merkel cell carcinoma, invasive SCC, and melanoma with positive margins or macroscopic residual disease. They proposed that all other skin cancers, including basal cell carcinoma, SCC in situ, and melanoma with negative margins and no macroscopic residual disease, could be safely delayed for as long as 3 months.15
For patients with multiple risk factors for COVID-19–related morbidity or mortality, delaying skin cancer treatment likely has less risk than contracting the virus.15 This information should be communicated with patients. Investigation of specific patient concerns is warranted, and case-by-case evaluation of patients’ risk factors and skin cancer risk should be considered.
Based on the current, though limited, literature, delaying medical treatment can have a negative impact on the patient experience. Furthermore, proper precautions have been shown to limit intraoperative transmission of SARS-CoV-2 during MMS, but research is lacking. Practitioners should utilize shared decision-making and evaluate a given patient’s risk factors and concerns when deciding whether to postpone treatment. We encourage other institutions to evaluate the effects that delaying MMS has had on their patients, as further studies would improve understanding of patients’ experiences during a pandemic and potentially influence future dermatology guidelines.
- Center for Disease Control and Prevention. COVID-19. Accessed April 20, 2021. https://www.cdc.gov/coronavirus/2019-ncov/index.html
- American College of Mohs Surgery. Mohs surgery ambulatory protocol during COVID pandemic (version 6-3-20). June 4, 2020. Accessed April 20, 2021. http://staging.mohscollege.org/UserFiles/AM20/Member%20Alert/MohsSurgeryAmbulatoryProtocolDuringCOVIDPandemicFinal.pdf
- COVID-19 resources. National Comprehensive Cancer Network website. Accessed April 20, 2021. https://www.nccn.org/covid-19
- Narla S, Alam M, Ozog DM, et al. American Society of Dermatologic Surgery Association (ASDSA) and American Society for Laser Medicine & Surgery (ASLMS) guidance for cosmetic dermatology practices during COVID-19. Updated January 11, 2021. Accessed April 10, 2021. https://www.asds.net/Portals/0/PDF/asdsa/asdsa-aslms-cosmetic-reopening-guidance.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic.J Am Acad Dermatol. 2020;83:295-296. doi:10.1016/j.jaad.2020.05.002
- Yuan JT, Jiang SIB. Urgent safety considerations for dermatologic surgeons in the COVID-19 pandemic. Dermatol Online J. 2020;26:1. Accessed April 20, 2021. http://escholarship.org/uc/item/2qr3w771
- Otolaryngologists may contract COVID-19 during surgery. ENTtoday. March 20, 2020. Accessed April 20, 2021. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/
- Howard BE. High-risk aerosol-generating procedures in COVID-19: respiratory protective equipment considerations. Otolaryngol Head Neck Surg. 2020;163:98-103. doi:10.1177/0194599820927335
- Tee MW, Stewart C, Aliessa S, et al. Dermatological surgery during the COVID-19 pandemic: experience of a large academic center. J Am Acad Dermatol. 2021;84:1094-1096. doi:10.1016/j.jaad.2020.12.003
- Hooper J, Feng H. The impact of COVID-19 on micrographic surgery and dermatologic oncology fellows. Dermatol Surg. 2020;46:1762-1763. doi:10.1097/DSS.0000000000002766
- Nicholson P, Ali FR, Patalay R, et al. Patient perceptions of Mohs micrographic surgery during the COVID-19 pandemic and lessons for the next outbreak. Clin Exp Dermatol. 2021;46:179-180. doi:10.1111/ced.14423
- Mou T, Brown O, Gillingham A, et al. Patients’ perceptions on surgical care suspension for pelvic floor disorders during the COVID-19 pandemic. Female Pelvic Med Reconstr Surg. 2020;26:477-482. doi:10.1097/SPV.0000000000000918
- Fujita K, Ito T, Saito Z, et al. Impact of COVID-19 pandemic on lung cancer treatment scheduling. Thorac Cancer. 2020;11:2983-2986. doi:10.1111/1759-7714.13615
- Nikumb VB, Banerjee A, Kaur G, et al. Impact of doctor-patient communication on preoperative anxiety: study at industrial township, Pimpri, Pune. Ind Psychiatry J. 2009;18:19-21. doi:10.4103/0972-6748.57852
- Baumann BC, MacArthur KM, Brewer JD, et al. Management of primary skin cancer during a pandemic: multidisciplinary recommendations. Cancer. 2020;126:3900-3906. doi:10.1002/cncr.32969
- Center for Disease Control and Prevention. COVID-19. Accessed April 20, 2021. https://www.cdc.gov/coronavirus/2019-ncov/index.html
- American College of Mohs Surgery. Mohs surgery ambulatory protocol during COVID pandemic (version 6-3-20). June 4, 2020. Accessed April 20, 2021. http://staging.mohscollege.org/UserFiles/AM20/Member%20Alert/MohsSurgeryAmbulatoryProtocolDuringCOVIDPandemicFinal.pdf
- COVID-19 resources. National Comprehensive Cancer Network website. Accessed April 20, 2021. https://www.nccn.org/covid-19
- Narla S, Alam M, Ozog DM, et al. American Society of Dermatologic Surgery Association (ASDSA) and American Society for Laser Medicine & Surgery (ASLMS) guidance for cosmetic dermatology practices during COVID-19. Updated January 11, 2021. Accessed April 10, 2021. https://www.asds.net/Portals/0/PDF/asdsa/asdsa-aslms-cosmetic-reopening-guidance.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic.J Am Acad Dermatol. 2020;83:295-296. doi:10.1016/j.jaad.2020.05.002
- Yuan JT, Jiang SIB. Urgent safety considerations for dermatologic surgeons in the COVID-19 pandemic. Dermatol Online J. 2020;26:1. Accessed April 20, 2021. http://escholarship.org/uc/item/2qr3w771
- Otolaryngologists may contract COVID-19 during surgery. ENTtoday. March 20, 2020. Accessed April 20, 2021. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/
- Howard BE. High-risk aerosol-generating procedures in COVID-19: respiratory protective equipment considerations. Otolaryngol Head Neck Surg. 2020;163:98-103. doi:10.1177/0194599820927335
- Tee MW, Stewart C, Aliessa S, et al. Dermatological surgery during the COVID-19 pandemic: experience of a large academic center. J Am Acad Dermatol. 2021;84:1094-1096. doi:10.1016/j.jaad.2020.12.003
- Hooper J, Feng H. The impact of COVID-19 on micrographic surgery and dermatologic oncology fellows. Dermatol Surg. 2020;46:1762-1763. doi:10.1097/DSS.0000000000002766
- Nicholson P, Ali FR, Patalay R, et al. Patient perceptions of Mohs micrographic surgery during the COVID-19 pandemic and lessons for the next outbreak. Clin Exp Dermatol. 2021;46:179-180. doi:10.1111/ced.14423
- Mou T, Brown O, Gillingham A, et al. Patients’ perceptions on surgical care suspension for pelvic floor disorders during the COVID-19 pandemic. Female Pelvic Med Reconstr Surg. 2020;26:477-482. doi:10.1097/SPV.0000000000000918
- Fujita K, Ito T, Saito Z, et al. Impact of COVID-19 pandemic on lung cancer treatment scheduling. Thorac Cancer. 2020;11:2983-2986. doi:10.1111/1759-7714.13615
- Nikumb VB, Banerjee A, Kaur G, et al. Impact of doctor-patient communication on preoperative anxiety: study at industrial township, Pimpri, Pune. Ind Psychiatry J. 2009;18:19-21. doi:10.4103/0972-6748.57852
- Baumann BC, MacArthur KM, Brewer JD, et al. Management of primary skin cancer during a pandemic: multidisciplinary recommendations. Cancer. 2020;126:3900-3906. doi:10.1002/cncr.32969
Practice Points
- There is little evidence that supports a high risk for SARS-CoV-2 transmission during Mohs micrographic surgery when proper personal protective equipment and strict infection control protocols are in place.
- The effects of treatment delays due to COVID-19 on the patient experience have not been well studied, but the limited literature suggests a negative association.
- Shared decision-making and evaluation of individual patient risk factors and concerns should be considered when deciding whether to postpone skin cancer treatment.
Novel hedgehog inhibitor strategies improve BCC outcomes
MD, a Mohs surgeon and chair of the department of dermatology at the Cleveland Clinic.
She and her colleagues have noticed an accelerated and durable response to hedgehog inhibitors after debulking and are studying cell signaling before and after debulking to better understand the issue.
Dr. Vidimos shared a remarkable case to illustrate the point during a clinical pearls talk at the annual meeting of the American College of Mohs Surgery.
An 82-year-old woman presented with a crusted, hemorrhagic, nodular basal cell carcinoma (BCC) that had overgrown over nearly her entire nose and left lower eyelid. A recurrence of a previous BCC, the tumor had been growing for a decade and had invaded her nasal bones but not the periorbital tissue.
An outside surgeon suggested a full rhinectomy and removal of the lower eyelid, but the woman refused.
Dr. Vidimos decided to treat her with vismodegib, but prior to doing so, she debulked the tumor to help with the pain and bleeding. She did not curette the portion of tumor extending through the ala into the nasal vestibule. “I let the vismodegib take care of that,” she said.
After 9 months, the tumor was virtually gone, with no recurrence after 3 years. Surgical debulking prior to hedgehog inhibition “reduces the tumor burden and may increase the efficacy and shorten the course of therapy,” Dr. Vidimos said.
The hedgehog inhibitors vismodegib (Erivedge) and sonidegib Odomzo are both approved for treating locally advanced BCC, with a complete response of 31% of locally advanced disease with vismodegib, according to one report.
But monotherapy is limited by intolerable side effects, most commonly muscle spasms, alopecia, and dysgeusia. To minimize the impact, Dr. Vidimos generally puts patients on treatment with Monday through Friday dosing and gives them the weekends off, a schedule she and her colleagues have reported works as well as daily dosing.
Still, many patients discontinue the drugs because of the side effects. Hedgehog inhibitors are also expensive and responses aren’t always durable. To increase efficacy and shorten the course of therapy, “we need alternative treatment strategies,” Dr. Vidimos said.
Up-front tumor debulking is one such strategy. Altered cell signaling pathways associated with tissue remodeling might improve response, and debulking may reduce the genetic heterogeneity of tumor cells, rendering remaining cells less resistant to hedgehog inhibition, she explained.
“It is exciting to see how tumor debulking may reduce tumor burden and heterogeneity, and thus lead to a durable response in extensive tumors,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington, who heard the presentation. “More investigation is needed to reproduce these results, but this approach may lead to improved outcomes with targeted therapies,” he said in an interview.
Combination therapy with other agents is another option, and there also seems to be a synergistic effect with radiation, with hedgehog inhibitors increasing cellular response to radiation therapy, Dr. Vidimos said.
Hedgehog inhibitors can also be used to shrink tumors before surgery. One small series found a 27% decrease in the area of the tumor after 3 to 6 months of preoperative vismodegib.
Dr. Vidimos shared another case to illustrate the point.
A 64-year-old woman fainted and presented to the ED with a hemoglobin of 3.2 mg/dL because of chronic blood loss from an ulcerated BCC on her upper back. The lesion measured 25 cm by 9 cm, and was 3.5 cm deep with no bone involvement. The woman was addicted to opioids by the time she presented.
She was started on vismodegib; the ulcer shrunk considerably after 6 months, and the woman underwent a resection. Only one small focus of BCC was found across 78 specimens submitted to Dr. Vidimos for Mohs reading.
Resection was followed by a muscle flap repair and radiation. At 5 and a half years, there is no evidence of disease; the only sign that the lesion had been there was a scar running along the woman’s upper spine.
The approach “was very successful for a very aggressive and worrisome tumor,” Dr. Vidimos said.
Dr. Vidimos did not have any relevant disclosures. Dr. Patel had no relevant disclosures.
MD, a Mohs surgeon and chair of the department of dermatology at the Cleveland Clinic.
She and her colleagues have noticed an accelerated and durable response to hedgehog inhibitors after debulking and are studying cell signaling before and after debulking to better understand the issue.
Dr. Vidimos shared a remarkable case to illustrate the point during a clinical pearls talk at the annual meeting of the American College of Mohs Surgery.
An 82-year-old woman presented with a crusted, hemorrhagic, nodular basal cell carcinoma (BCC) that had overgrown over nearly her entire nose and left lower eyelid. A recurrence of a previous BCC, the tumor had been growing for a decade and had invaded her nasal bones but not the periorbital tissue.
An outside surgeon suggested a full rhinectomy and removal of the lower eyelid, but the woman refused.
Dr. Vidimos decided to treat her with vismodegib, but prior to doing so, she debulked the tumor to help with the pain and bleeding. She did not curette the portion of tumor extending through the ala into the nasal vestibule. “I let the vismodegib take care of that,” she said.
After 9 months, the tumor was virtually gone, with no recurrence after 3 years. Surgical debulking prior to hedgehog inhibition “reduces the tumor burden and may increase the efficacy and shorten the course of therapy,” Dr. Vidimos said.
The hedgehog inhibitors vismodegib (Erivedge) and sonidegib Odomzo are both approved for treating locally advanced BCC, with a complete response of 31% of locally advanced disease with vismodegib, according to one report.
But monotherapy is limited by intolerable side effects, most commonly muscle spasms, alopecia, and dysgeusia. To minimize the impact, Dr. Vidimos generally puts patients on treatment with Monday through Friday dosing and gives them the weekends off, a schedule she and her colleagues have reported works as well as daily dosing.
Still, many patients discontinue the drugs because of the side effects. Hedgehog inhibitors are also expensive and responses aren’t always durable. To increase efficacy and shorten the course of therapy, “we need alternative treatment strategies,” Dr. Vidimos said.
Up-front tumor debulking is one such strategy. Altered cell signaling pathways associated with tissue remodeling might improve response, and debulking may reduce the genetic heterogeneity of tumor cells, rendering remaining cells less resistant to hedgehog inhibition, she explained.
“It is exciting to see how tumor debulking may reduce tumor burden and heterogeneity, and thus lead to a durable response in extensive tumors,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington, who heard the presentation. “More investigation is needed to reproduce these results, but this approach may lead to improved outcomes with targeted therapies,” he said in an interview.
Combination therapy with other agents is another option, and there also seems to be a synergistic effect with radiation, with hedgehog inhibitors increasing cellular response to radiation therapy, Dr. Vidimos said.
Hedgehog inhibitors can also be used to shrink tumors before surgery. One small series found a 27% decrease in the area of the tumor after 3 to 6 months of preoperative vismodegib.
Dr. Vidimos shared another case to illustrate the point.
A 64-year-old woman fainted and presented to the ED with a hemoglobin of 3.2 mg/dL because of chronic blood loss from an ulcerated BCC on her upper back. The lesion measured 25 cm by 9 cm, and was 3.5 cm deep with no bone involvement. The woman was addicted to opioids by the time she presented.
She was started on vismodegib; the ulcer shrunk considerably after 6 months, and the woman underwent a resection. Only one small focus of BCC was found across 78 specimens submitted to Dr. Vidimos for Mohs reading.
Resection was followed by a muscle flap repair and radiation. At 5 and a half years, there is no evidence of disease; the only sign that the lesion had been there was a scar running along the woman’s upper spine.
The approach “was very successful for a very aggressive and worrisome tumor,” Dr. Vidimos said.
Dr. Vidimos did not have any relevant disclosures. Dr. Patel had no relevant disclosures.
MD, a Mohs surgeon and chair of the department of dermatology at the Cleveland Clinic.
She and her colleagues have noticed an accelerated and durable response to hedgehog inhibitors after debulking and are studying cell signaling before and after debulking to better understand the issue.
Dr. Vidimos shared a remarkable case to illustrate the point during a clinical pearls talk at the annual meeting of the American College of Mohs Surgery.
An 82-year-old woman presented with a crusted, hemorrhagic, nodular basal cell carcinoma (BCC) that had overgrown over nearly her entire nose and left lower eyelid. A recurrence of a previous BCC, the tumor had been growing for a decade and had invaded her nasal bones but not the periorbital tissue.
An outside surgeon suggested a full rhinectomy and removal of the lower eyelid, but the woman refused.
Dr. Vidimos decided to treat her with vismodegib, but prior to doing so, she debulked the tumor to help with the pain and bleeding. She did not curette the portion of tumor extending through the ala into the nasal vestibule. “I let the vismodegib take care of that,” she said.
After 9 months, the tumor was virtually gone, with no recurrence after 3 years. Surgical debulking prior to hedgehog inhibition “reduces the tumor burden and may increase the efficacy and shorten the course of therapy,” Dr. Vidimos said.
The hedgehog inhibitors vismodegib (Erivedge) and sonidegib Odomzo are both approved for treating locally advanced BCC, with a complete response of 31% of locally advanced disease with vismodegib, according to one report.
But monotherapy is limited by intolerable side effects, most commonly muscle spasms, alopecia, and dysgeusia. To minimize the impact, Dr. Vidimos generally puts patients on treatment with Monday through Friday dosing and gives them the weekends off, a schedule she and her colleagues have reported works as well as daily dosing.
Still, many patients discontinue the drugs because of the side effects. Hedgehog inhibitors are also expensive and responses aren’t always durable. To increase efficacy and shorten the course of therapy, “we need alternative treatment strategies,” Dr. Vidimos said.
Up-front tumor debulking is one such strategy. Altered cell signaling pathways associated with tissue remodeling might improve response, and debulking may reduce the genetic heterogeneity of tumor cells, rendering remaining cells less resistant to hedgehog inhibition, she explained.
“It is exciting to see how tumor debulking may reduce tumor burden and heterogeneity, and thus lead to a durable response in extensive tumors,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington, who heard the presentation. “More investigation is needed to reproduce these results, but this approach may lead to improved outcomes with targeted therapies,” he said in an interview.
Combination therapy with other agents is another option, and there also seems to be a synergistic effect with radiation, with hedgehog inhibitors increasing cellular response to radiation therapy, Dr. Vidimos said.
Hedgehog inhibitors can also be used to shrink tumors before surgery. One small series found a 27% decrease in the area of the tumor after 3 to 6 months of preoperative vismodegib.
Dr. Vidimos shared another case to illustrate the point.
A 64-year-old woman fainted and presented to the ED with a hemoglobin of 3.2 mg/dL because of chronic blood loss from an ulcerated BCC on her upper back. The lesion measured 25 cm by 9 cm, and was 3.5 cm deep with no bone involvement. The woman was addicted to opioids by the time she presented.
She was started on vismodegib; the ulcer shrunk considerably after 6 months, and the woman underwent a resection. Only one small focus of BCC was found across 78 specimens submitted to Dr. Vidimos for Mohs reading.
Resection was followed by a muscle flap repair and radiation. At 5 and a half years, there is no evidence of disease; the only sign that the lesion had been there was a scar running along the woman’s upper spine.
The approach “was very successful for a very aggressive and worrisome tumor,” Dr. Vidimos said.
Dr. Vidimos did not have any relevant disclosures. Dr. Patel had no relevant disclosures.
FROM THE ACMS ANNUAL MEETING
Mycosis Fungoides in Black Patients: Time for a Better Look
Recent advances in the immunopathogenesis and therapy of cutaneous T-cell lymphoma (CTCL) have shown great promise for the care of patients with mycosis fungoides (MF) and Sézary syndrome (SS).1-3 Research into the tumor microenvironment, microbiome, and molecular genetics may yield further information as we strive to develop MF/SS therapy from the bench to the bedside.3 Although progress has been made on multiple fronts in MF, some important—particularly epidemiologic and clinical—questions remain unanswered.
Racial disparities are well known to exist in CTCLs, particularly MF and SS.4-7 The incidence of MF and SS in the United States is higher in African American/Black patients than in White patients4; in addition, MF has an earlier age at onset in Black patients compared with White patients.4,5 Gender disparities also exist, with relatively more Black females than males affected with MF4-6; in particular, early-onset MF (ie, <40 years of age) is more common in Black females than Black males.6,7 According to Surveillance, Epidemiology, and End Results (SEER) data4 and the US National Cancer Database,5 African American/Black patients with MF have worse outcomes compared with other races (shorter overall survival and higher mortality) and also exhibit higher stages of disease at presentation (stage IIb or higher).5 Black race also was found to be a predictor of poor overall survival after accounting for disease characteristics, socioeconomicfactors, and types of treatment. The factors responsible for these racial disparities remain unclear.
A fortuitous collision of interests and technology may have helped to shed light on some of the reasons for these racial disparities in MF. Nearly 2 decades ago, high-quality, whole-body digital cutaneous photography was implemented by the Dermatology Service at Memorial Sloan Kettering Cancer Center Dermatology Service (New York, New York).8 Although the standardized 20-pose positioning images initially were used for the follow-up evaluation of patients with multiple nevi and melanomas, we incorporated the same photography technique into our multidisciplinary Cutaneous Lymphoma Clinic at Memorial Sloan Kettering Cancer Center. The multiplicity and clinical heterogeneity of MF lesions is well known, as is the fact that individual MF lesions may develop, respond to therapy, or change independently of other lesions in a given patient. We regularly reviewed these digital images with patients during their visits to assess treatment responses, discussed the need for changes in therapy in the face of progressive disease, and provided encouragement and positive reinforcement for those who improved with time-consuming regimens (eg, phototherapy).
Ultimately, as we became more familiar with looking at images in skin of color, we recognized different clinical features among our Black patients. In the literature, hypopigmented MF is a variant that typically is characterized by CD8+-predominant T cells and is seen more frequently in dark-skinned patients.9 In contrast, hyperpigmented MF has been considered a relatively rare presentation of MF.10 However, using only clinical and demographic information, we were able to identify 2 very different prognostic groups: those with hypopigmented lesions and those with only hyperpigmented and/or erythematous skin lesions.11 In our retrospective review of 157 African American/Black MF patients at our institution—122 with early-stage and 35 with late-stage MF—45% of patients had hypopigmented lesions vs 52% with hyperpigmented and/or erythematous lesions but no hypopigmentation. Those with hypopigmentation had superior outcomes, with better overall survival (P=.002) and progression-free survival (P=.014). In addition, more than 80% of patients who progressed or died from disease had hyperpigmented and/or erythematous lesions without hypopigmentation.11
Sometimes we have to go backward to go forward. Going from the bedside to the bench in our Black MF/SS patients—initially through the clinical recognition of prognostically different lesions, and then through clinicopathologic correlation with immunophenotyping and molecular studies—should provide important clues. Further investigation of Black patients who share similar pigmentary phenotypes of MF also may shed light on the pathogenetic mechanisms responsible for these prognostically significant skin findings. Through these efforts, we hope to identify higher-risk patients, which ultimately will lead to earlier intervention, more effective therapeutic regimens, and improved outcomes.
- Durgin JS, Weiner DM, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: pathways and targets for immune restoration and tumor eradication. J Am Acad Dermatol. 2021;84:587-595.
- Weiner DM, Durgin JS, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: current and future approaches. J Am Acad Dermatol. 2021;84:597-604.
- Quaglino P, Fava P, Pileri A, et al. Phenotypical markers, molecular mutations, and immune microenvironment as targets for new treatments in patients with mycosis fungoides and/or Sézary syndrome. J Invest Dermatol. 2021;141:484-495.
- Nath SK, Yu JB, Wilson LD. Poorer prognosis of African-American patients with mycosis fungoides: an analysis of the SEER dataset, 1988 to 2008. Clin Lymphoma Myeloma Leuk. 2014;14:419-423.
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.
- Balagula Y, Dusza SW, Zampella J, et al. Early-onset mycosis fungoides among African American women: a single-institution study. J Am Acad Dermatol. 2014;71:597-598.
- Virmani P, Levin L, Myskowski PL, et al. Clinical outcome and prognosis of young patients with mycosis fungoides. Pediatr Dermatol. 2017;34:547-553.
- Halpern AC, Marghoob AA, Bialoglow TW, et al. Standardized positioning of patients (poses) for whole body cutaneous photography. J Am Acad Dermatol. 2003;49:593-598.
- Rodney IJ, Kindred C, Angra K, et al. Hypopigmented mycosis fungoides: a retrospective clinicohistopathologic study. J Eur Acad Dermatol Venereol. 2017;31:808-814.
- Kondo M, Igawa K, Munetsugu T, et al. Increasing numbers of mast cells in skin lesions of hyperpigmented mycosis fungoides with large-cell transformation. Ann Dermatol. 2016;28:115-116.
- Geller S, Lebowitz E, Pulitzer MP, et al. Outcomes and prognostic factors in African American and Black patients with mycosis fungoides/Sézary syndrome: retrospective analysis of 157 patients from a referral cancer center. J Am Acad Dermatol. 2020;83:430-439.
Recent advances in the immunopathogenesis and therapy of cutaneous T-cell lymphoma (CTCL) have shown great promise for the care of patients with mycosis fungoides (MF) and Sézary syndrome (SS).1-3 Research into the tumor microenvironment, microbiome, and molecular genetics may yield further information as we strive to develop MF/SS therapy from the bench to the bedside.3 Although progress has been made on multiple fronts in MF, some important—particularly epidemiologic and clinical—questions remain unanswered.
Racial disparities are well known to exist in CTCLs, particularly MF and SS.4-7 The incidence of MF and SS in the United States is higher in African American/Black patients than in White patients4; in addition, MF has an earlier age at onset in Black patients compared with White patients.4,5 Gender disparities also exist, with relatively more Black females than males affected with MF4-6; in particular, early-onset MF (ie, <40 years of age) is more common in Black females than Black males.6,7 According to Surveillance, Epidemiology, and End Results (SEER) data4 and the US National Cancer Database,5 African American/Black patients with MF have worse outcomes compared with other races (shorter overall survival and higher mortality) and also exhibit higher stages of disease at presentation (stage IIb or higher).5 Black race also was found to be a predictor of poor overall survival after accounting for disease characteristics, socioeconomicfactors, and types of treatment. The factors responsible for these racial disparities remain unclear.
A fortuitous collision of interests and technology may have helped to shed light on some of the reasons for these racial disparities in MF. Nearly 2 decades ago, high-quality, whole-body digital cutaneous photography was implemented by the Dermatology Service at Memorial Sloan Kettering Cancer Center Dermatology Service (New York, New York).8 Although the standardized 20-pose positioning images initially were used for the follow-up evaluation of patients with multiple nevi and melanomas, we incorporated the same photography technique into our multidisciplinary Cutaneous Lymphoma Clinic at Memorial Sloan Kettering Cancer Center. The multiplicity and clinical heterogeneity of MF lesions is well known, as is the fact that individual MF lesions may develop, respond to therapy, or change independently of other lesions in a given patient. We regularly reviewed these digital images with patients during their visits to assess treatment responses, discussed the need for changes in therapy in the face of progressive disease, and provided encouragement and positive reinforcement for those who improved with time-consuming regimens (eg, phototherapy).
Ultimately, as we became more familiar with looking at images in skin of color, we recognized different clinical features among our Black patients. In the literature, hypopigmented MF is a variant that typically is characterized by CD8+-predominant T cells and is seen more frequently in dark-skinned patients.9 In contrast, hyperpigmented MF has been considered a relatively rare presentation of MF.10 However, using only clinical and demographic information, we were able to identify 2 very different prognostic groups: those with hypopigmented lesions and those with only hyperpigmented and/or erythematous skin lesions.11 In our retrospective review of 157 African American/Black MF patients at our institution—122 with early-stage and 35 with late-stage MF—45% of patients had hypopigmented lesions vs 52% with hyperpigmented and/or erythematous lesions but no hypopigmentation. Those with hypopigmentation had superior outcomes, with better overall survival (P=.002) and progression-free survival (P=.014). In addition, more than 80% of patients who progressed or died from disease had hyperpigmented and/or erythematous lesions without hypopigmentation.11
Sometimes we have to go backward to go forward. Going from the bedside to the bench in our Black MF/SS patients—initially through the clinical recognition of prognostically different lesions, and then through clinicopathologic correlation with immunophenotyping and molecular studies—should provide important clues. Further investigation of Black patients who share similar pigmentary phenotypes of MF also may shed light on the pathogenetic mechanisms responsible for these prognostically significant skin findings. Through these efforts, we hope to identify higher-risk patients, which ultimately will lead to earlier intervention, more effective therapeutic regimens, and improved outcomes.
Recent advances in the immunopathogenesis and therapy of cutaneous T-cell lymphoma (CTCL) have shown great promise for the care of patients with mycosis fungoides (MF) and Sézary syndrome (SS).1-3 Research into the tumor microenvironment, microbiome, and molecular genetics may yield further information as we strive to develop MF/SS therapy from the bench to the bedside.3 Although progress has been made on multiple fronts in MF, some important—particularly epidemiologic and clinical—questions remain unanswered.
Racial disparities are well known to exist in CTCLs, particularly MF and SS.4-7 The incidence of MF and SS in the United States is higher in African American/Black patients than in White patients4; in addition, MF has an earlier age at onset in Black patients compared with White patients.4,5 Gender disparities also exist, with relatively more Black females than males affected with MF4-6; in particular, early-onset MF (ie, <40 years of age) is more common in Black females than Black males.6,7 According to Surveillance, Epidemiology, and End Results (SEER) data4 and the US National Cancer Database,5 African American/Black patients with MF have worse outcomes compared with other races (shorter overall survival and higher mortality) and also exhibit higher stages of disease at presentation (stage IIb or higher).5 Black race also was found to be a predictor of poor overall survival after accounting for disease characteristics, socioeconomicfactors, and types of treatment. The factors responsible for these racial disparities remain unclear.
A fortuitous collision of interests and technology may have helped to shed light on some of the reasons for these racial disparities in MF. Nearly 2 decades ago, high-quality, whole-body digital cutaneous photography was implemented by the Dermatology Service at Memorial Sloan Kettering Cancer Center Dermatology Service (New York, New York).8 Although the standardized 20-pose positioning images initially were used for the follow-up evaluation of patients with multiple nevi and melanomas, we incorporated the same photography technique into our multidisciplinary Cutaneous Lymphoma Clinic at Memorial Sloan Kettering Cancer Center. The multiplicity and clinical heterogeneity of MF lesions is well known, as is the fact that individual MF lesions may develop, respond to therapy, or change independently of other lesions in a given patient. We regularly reviewed these digital images with patients during their visits to assess treatment responses, discussed the need for changes in therapy in the face of progressive disease, and provided encouragement and positive reinforcement for those who improved with time-consuming regimens (eg, phototherapy).
Ultimately, as we became more familiar with looking at images in skin of color, we recognized different clinical features among our Black patients. In the literature, hypopigmented MF is a variant that typically is characterized by CD8+-predominant T cells and is seen more frequently in dark-skinned patients.9 In contrast, hyperpigmented MF has been considered a relatively rare presentation of MF.10 However, using only clinical and demographic information, we were able to identify 2 very different prognostic groups: those with hypopigmented lesions and those with only hyperpigmented and/or erythematous skin lesions.11 In our retrospective review of 157 African American/Black MF patients at our institution—122 with early-stage and 35 with late-stage MF—45% of patients had hypopigmented lesions vs 52% with hyperpigmented and/or erythematous lesions but no hypopigmentation. Those with hypopigmentation had superior outcomes, with better overall survival (P=.002) and progression-free survival (P=.014). In addition, more than 80% of patients who progressed or died from disease had hyperpigmented and/or erythematous lesions without hypopigmentation.11
Sometimes we have to go backward to go forward. Going from the bedside to the bench in our Black MF/SS patients—initially through the clinical recognition of prognostically different lesions, and then through clinicopathologic correlation with immunophenotyping and molecular studies—should provide important clues. Further investigation of Black patients who share similar pigmentary phenotypes of MF also may shed light on the pathogenetic mechanisms responsible for these prognostically significant skin findings. Through these efforts, we hope to identify higher-risk patients, which ultimately will lead to earlier intervention, more effective therapeutic regimens, and improved outcomes.
- Durgin JS, Weiner DM, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: pathways and targets for immune restoration and tumor eradication. J Am Acad Dermatol. 2021;84:587-595.
- Weiner DM, Durgin JS, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: current and future approaches. J Am Acad Dermatol. 2021;84:597-604.
- Quaglino P, Fava P, Pileri A, et al. Phenotypical markers, molecular mutations, and immune microenvironment as targets for new treatments in patients with mycosis fungoides and/or Sézary syndrome. J Invest Dermatol. 2021;141:484-495.
- Nath SK, Yu JB, Wilson LD. Poorer prognosis of African-American patients with mycosis fungoides: an analysis of the SEER dataset, 1988 to 2008. Clin Lymphoma Myeloma Leuk. 2014;14:419-423.
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.
- Balagula Y, Dusza SW, Zampella J, et al. Early-onset mycosis fungoides among African American women: a single-institution study. J Am Acad Dermatol. 2014;71:597-598.
- Virmani P, Levin L, Myskowski PL, et al. Clinical outcome and prognosis of young patients with mycosis fungoides. Pediatr Dermatol. 2017;34:547-553.
- Halpern AC, Marghoob AA, Bialoglow TW, et al. Standardized positioning of patients (poses) for whole body cutaneous photography. J Am Acad Dermatol. 2003;49:593-598.
- Rodney IJ, Kindred C, Angra K, et al. Hypopigmented mycosis fungoides: a retrospective clinicohistopathologic study. J Eur Acad Dermatol Venereol. 2017;31:808-814.
- Kondo M, Igawa K, Munetsugu T, et al. Increasing numbers of mast cells in skin lesions of hyperpigmented mycosis fungoides with large-cell transformation. Ann Dermatol. 2016;28:115-116.
- Geller S, Lebowitz E, Pulitzer MP, et al. Outcomes and prognostic factors in African American and Black patients with mycosis fungoides/Sézary syndrome: retrospective analysis of 157 patients from a referral cancer center. J Am Acad Dermatol. 2020;83:430-439.
- Durgin JS, Weiner DM, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: pathways and targets for immune restoration and tumor eradication. J Am Acad Dermatol. 2021;84:587-595.
- Weiner DM, Durgin JS, Wysocka M, et al. The immunopathogenesis and immunotherapy of cutaneous T cell lymphoma: current and future approaches. J Am Acad Dermatol. 2021;84:597-604.
- Quaglino P, Fava P, Pileri A, et al. Phenotypical markers, molecular mutations, and immune microenvironment as targets for new treatments in patients with mycosis fungoides and/or Sézary syndrome. J Invest Dermatol. 2021;141:484-495.
- Nath SK, Yu JB, Wilson LD. Poorer prognosis of African-American patients with mycosis fungoides: an analysis of the SEER dataset, 1988 to 2008. Clin Lymphoma Myeloma Leuk. 2014;14:419-423.
- Su C, Nguyen KA, Bai HX, et al. Racial disparity in mycosis fungoides: an analysis of 4495 cases from the US National Cancer Database. J Am Acad Dermatol. 2017;77:497-502.
- Balagula Y, Dusza SW, Zampella J, et al. Early-onset mycosis fungoides among African American women: a single-institution study. J Am Acad Dermatol. 2014;71:597-598.
- Virmani P, Levin L, Myskowski PL, et al. Clinical outcome and prognosis of young patients with mycosis fungoides. Pediatr Dermatol. 2017;34:547-553.
- Halpern AC, Marghoob AA, Bialoglow TW, et al. Standardized positioning of patients (poses) for whole body cutaneous photography. J Am Acad Dermatol. 2003;49:593-598.
- Rodney IJ, Kindred C, Angra K, et al. Hypopigmented mycosis fungoides: a retrospective clinicohistopathologic study. J Eur Acad Dermatol Venereol. 2017;31:808-814.
- Kondo M, Igawa K, Munetsugu T, et al. Increasing numbers of mast cells in skin lesions of hyperpigmented mycosis fungoides with large-cell transformation. Ann Dermatol. 2016;28:115-116.
- Geller S, Lebowitz E, Pulitzer MP, et al. Outcomes and prognostic factors in African American and Black patients with mycosis fungoides/Sézary syndrome: retrospective analysis of 157 patients from a referral cancer center. J Am Acad Dermatol. 2020;83:430-439.
Reconstruction Technique for Defects of the Cutaneous and Mucosal Lip: V-to-flying-Y Closure
Practice Gap
Reconstruction of a lip defect poses challenges to the dermatologic surgeon. The lip is a free margin, where excess tension can cause noticeable distortion in facial aesthetics. Distortion of that free margin might not only disrupt the appearance of the lip but affect function by impairing oral competency and mobility; therefore, when choosing a method of reconstruction, the surgeon must take free margin distortion into account. Misalignment of the vermilion border upon reconstruction will cause a poor aesthetic result in the absence of free margin distortion. When a surgical defect involves more than one cosmetic subunit of the lip, great care must be taken to repair each subunit individually to achieve the best cosmetic and functional results.
The suitability of traditional approaches to reconstruction of a defect that crosses the vermilion border—healing by secondary intention, primary linear repair, full-thickness wedge repair, partial-thickness wedge repair, and combined cutaneous and mucosal advancement1—depends on the depth of the lesion.
Clinical Presentation
A 66-year-old woman with a 4×6-mm, invasive, well-differentiated squamous cell carcinoma of the left lower lip was referred for Mohs micrographic surgery. Removal of the tumor required 2 stages to obtain clear margins, leaving a 1.0×1.2-cm defect that crossed the vermilion border (Figure, A). How would you repair this defect?
Selecting a Technique to Close the Surgical Defect
For this patient, we had several options to consider in approaching closure, including several that we rejected. Because the defect crossed cosmetic subunit boundaries, healing by secondary intention was avoided, as it would cause contraction, obliterate the vermilion border, and result in poor functional and cosmetic results. We decided against primary closure, even with careful attention to reapproximation of the vermilion border, because the width of the defect would have required a large Burow triangle that extended into the oral cavity. For defects less than one-third the width of the lip, full-thickness wedge repair can yield excellent cosmetic results but, in this case, would decrease the oral aperture and was deemed too extensive a procedure for a relatively shallow defect.2
Instead, we chose to perform repair with V to Y advancement of skin below the cutaneous defect, up to the location of the absent vermilion border, combined with small, horizontal, linear closure of the mucosal portion of the defect. This approach is a variation of a repair described by Jin et al,3 who described using 2 opposing V-Y advancement flaps to repair defects of the lip. This repair has provided excellent cosmetic results for a small series of our patients, preserving the oral aperture and maintaining the important aesthetic location of the vermilion border. In addition, the technique makes it unnecessary for the patient to undergo a much larger repair, such as a full- or partial-thickness wedge when the initial defect is relatively shallow.
Closure Technique
It is essential to properly outline the vermilion border of the lip before initiating the repair, ideally before any infiltration of local anesthesia if the surgeon anticipates that tumor extirpation might cross the vermilion border.
Repair
Closing then proceeds as follows:
• The cutaneous portion of the defect is drawn out in standard V to Y fashion, carrying the incision through the dermis and into subcutaneous tissue. The pedicle of the flap is maintained at the base of the island, serving as the blood supply to the flap.
• The periphery of the flap and surrounding tissue is undermined to facilitate movement superiorly into the cutaneous portion of the defect.
• A single buried vertical mattress suture can be placed at the advancing border of the island, holding it in place at the anticipated location of the vermilion border. The secondary defect created by the advancing V is closed to help push the island into place and prevent downward tension on the free margin of the lip.
• The remaining defect of the vermilion lip is closed by removing Burow triangles at the horizontal edges on each side of the remaining defect (Figure, B). The triangles are removed completely within the mucosal lip, with the inferior edge of the triangle placed at the vermilion border.
• The defect is closed in a primary linear horizontal fashion, using buried vertical mattress sutures and cutaneous approximation.
The final appearance of the sutured defect yields a small lateral extension at the superior edge of the V to Y closure, giving the appearance of wings on the Y, prompting us to term the closure V-to-flying-Y (Figure, C).
Although the limited portion of the mucosal lip that is closed in this fashion might appear thinner than the remaining lip, it generally yields a cosmetically acceptable result in the properly selected patient. Our experience also has shown an improvement in this difference in the months following repair. A full mucosal advancement flap might result in a more uniform appearance of the lower lip, but it is a larger and more difficult procedure for the patient to endure. Additionally, a full mucosal advancement flap risks uniformly creating a much thinner lip.
Postoperative Course
Sutures were removed 1 week postoperatively. Proper location of the vermilion border, without distortion of the free margin, was demonstrated. At 11-month follow-up, excellent cosmetic and functional results were noted (Figure, D).
Practice Implications
This repair (1) demonstrates an elegant method of closing a relatively shallow defect that crosses the vermilion border and (2) allows the surgeon to address each cosmetic subunit individually. We have found that this repair provides excellent cosmetic and functional results, with little morbidity.
The lip is a common site of nonmelanoma squamous cell carcinoma. Poorly planned closing after excision of the tumor risks notable impairment of function or cosmetic distortion. When a defect of the lip crosses cosmetic subunits, it is helpful to repair each subunit individually. V-to-flying-Y closure is an effective method to close defects that cross the vermilion border, resulting in well-preserved cosmetic appearance and function.
- Ishii LE, Byrne PJ. Lip reconstruction. Facial Plast Surg Clin North Am. 2009;17:445-453. doi:10.1016/j.fsc.2009.05.007
- Sebben JE. Wedge resection of the lip: minimizing problems. J Dermatol Surg Oncol. 1985;11:60-64. doi:10.1111/j.1524-4725.1985.tb02892.x
- Jin X, Teng L, Zhang C, et al. Reconstruction of partial-thickness vermilion defects with a mucosal V-Y advancement flap based on the orbicularis oris muscle. J Plast Reconstr Aesthet Surg. 2011;64:472-476. doi:10.1016/j.bjps.2010.07.017
Practice Gap
Reconstruction of a lip defect poses challenges to the dermatologic surgeon. The lip is a free margin, where excess tension can cause noticeable distortion in facial aesthetics. Distortion of that free margin might not only disrupt the appearance of the lip but affect function by impairing oral competency and mobility; therefore, when choosing a method of reconstruction, the surgeon must take free margin distortion into account. Misalignment of the vermilion border upon reconstruction will cause a poor aesthetic result in the absence of free margin distortion. When a surgical defect involves more than one cosmetic subunit of the lip, great care must be taken to repair each subunit individually to achieve the best cosmetic and functional results.
The suitability of traditional approaches to reconstruction of a defect that crosses the vermilion border—healing by secondary intention, primary linear repair, full-thickness wedge repair, partial-thickness wedge repair, and combined cutaneous and mucosal advancement1—depends on the depth of the lesion.
Clinical Presentation
A 66-year-old woman with a 4×6-mm, invasive, well-differentiated squamous cell carcinoma of the left lower lip was referred for Mohs micrographic surgery. Removal of the tumor required 2 stages to obtain clear margins, leaving a 1.0×1.2-cm defect that crossed the vermilion border (Figure, A). How would you repair this defect?

Selecting a Technique to Close the Surgical Defect
For this patient, we had several options to consider in approaching closure, including several that we rejected. Because the defect crossed cosmetic subunit boundaries, healing by secondary intention was avoided, as it would cause contraction, obliterate the vermilion border, and result in poor functional and cosmetic results. We decided against primary closure, even with careful attention to reapproximation of the vermilion border, because the width of the defect would have required a large Burow triangle that extended into the oral cavity. For defects less than one-third the width of the lip, full-thickness wedge repair can yield excellent cosmetic results but, in this case, would decrease the oral aperture and was deemed too extensive a procedure for a relatively shallow defect.2
Instead, we chose to perform repair with V to Y advancement of skin below the cutaneous defect, up to the location of the absent vermilion border, combined with small, horizontal, linear closure of the mucosal portion of the defect. This approach is a variation of a repair described by Jin et al,3 who described using 2 opposing V-Y advancement flaps to repair defects of the lip. This repair has provided excellent cosmetic results for a small series of our patients, preserving the oral aperture and maintaining the important aesthetic location of the vermilion border. In addition, the technique makes it unnecessary for the patient to undergo a much larger repair, such as a full- or partial-thickness wedge when the initial defect is relatively shallow.
Closure Technique
It is essential to properly outline the vermilion border of the lip before initiating the repair, ideally before any infiltration of local anesthesia if the surgeon anticipates that tumor extirpation might cross the vermilion border.
Repair
Closing then proceeds as follows:
• The cutaneous portion of the defect is drawn out in standard V to Y fashion, carrying the incision through the dermis and into subcutaneous tissue. The pedicle of the flap is maintained at the base of the island, serving as the blood supply to the flap.
• The periphery of the flap and surrounding tissue is undermined to facilitate movement superiorly into the cutaneous portion of the defect.
• A single buried vertical mattress suture can be placed at the advancing border of the island, holding it in place at the anticipated location of the vermilion border. The secondary defect created by the advancing V is closed to help push the island into place and prevent downward tension on the free margin of the lip.
• The remaining defect of the vermilion lip is closed by removing Burow triangles at the horizontal edges on each side of the remaining defect (Figure, B). The triangles are removed completely within the mucosal lip, with the inferior edge of the triangle placed at the vermilion border.
• The defect is closed in a primary linear horizontal fashion, using buried vertical mattress sutures and cutaneous approximation.
The final appearance of the sutured defect yields a small lateral extension at the superior edge of the V to Y closure, giving the appearance of wings on the Y, prompting us to term the closure V-to-flying-Y (Figure, C).
Although the limited portion of the mucosal lip that is closed in this fashion might appear thinner than the remaining lip, it generally yields a cosmetically acceptable result in the properly selected patient. Our experience also has shown an improvement in this difference in the months following repair. A full mucosal advancement flap might result in a more uniform appearance of the lower lip, but it is a larger and more difficult procedure for the patient to endure. Additionally, a full mucosal advancement flap risks uniformly creating a much thinner lip.
Postoperative Course
Sutures were removed 1 week postoperatively. Proper location of the vermilion border, without distortion of the free margin, was demonstrated. At 11-month follow-up, excellent cosmetic and functional results were noted (Figure, D).
Practice Implications
This repair (1) demonstrates an elegant method of closing a relatively shallow defect that crosses the vermilion border and (2) allows the surgeon to address each cosmetic subunit individually. We have found that this repair provides excellent cosmetic and functional results, with little morbidity.
The lip is a common site of nonmelanoma squamous cell carcinoma. Poorly planned closing after excision of the tumor risks notable impairment of function or cosmetic distortion. When a defect of the lip crosses cosmetic subunits, it is helpful to repair each subunit individually. V-to-flying-Y closure is an effective method to close defects that cross the vermilion border, resulting in well-preserved cosmetic appearance and function.
Practice Gap
Reconstruction of a lip defect poses challenges to the dermatologic surgeon. The lip is a free margin, where excess tension can cause noticeable distortion in facial aesthetics. Distortion of that free margin might not only disrupt the appearance of the lip but affect function by impairing oral competency and mobility; therefore, when choosing a method of reconstruction, the surgeon must take free margin distortion into account. Misalignment of the vermilion border upon reconstruction will cause a poor aesthetic result in the absence of free margin distortion. When a surgical defect involves more than one cosmetic subunit of the lip, great care must be taken to repair each subunit individually to achieve the best cosmetic and functional results.
The suitability of traditional approaches to reconstruction of a defect that crosses the vermilion border—healing by secondary intention, primary linear repair, full-thickness wedge repair, partial-thickness wedge repair, and combined cutaneous and mucosal advancement1—depends on the depth of the lesion.
Clinical Presentation
A 66-year-old woman with a 4×6-mm, invasive, well-differentiated squamous cell carcinoma of the left lower lip was referred for Mohs micrographic surgery. Removal of the tumor required 2 stages to obtain clear margins, leaving a 1.0×1.2-cm defect that crossed the vermilion border (Figure, A). How would you repair this defect?

Selecting a Technique to Close the Surgical Defect
For this patient, we had several options to consider in approaching closure, including several that we rejected. Because the defect crossed cosmetic subunit boundaries, healing by secondary intention was avoided, as it would cause contraction, obliterate the vermilion border, and result in poor functional and cosmetic results. We decided against primary closure, even with careful attention to reapproximation of the vermilion border, because the width of the defect would have required a large Burow triangle that extended into the oral cavity. For defects less than one-third the width of the lip, full-thickness wedge repair can yield excellent cosmetic results but, in this case, would decrease the oral aperture and was deemed too extensive a procedure for a relatively shallow defect.2
Instead, we chose to perform repair with V to Y advancement of skin below the cutaneous defect, up to the location of the absent vermilion border, combined with small, horizontal, linear closure of the mucosal portion of the defect. This approach is a variation of a repair described by Jin et al,3 who described using 2 opposing V-Y advancement flaps to repair defects of the lip. This repair has provided excellent cosmetic results for a small series of our patients, preserving the oral aperture and maintaining the important aesthetic location of the vermilion border. In addition, the technique makes it unnecessary for the patient to undergo a much larger repair, such as a full- or partial-thickness wedge when the initial defect is relatively shallow.
Closure Technique
It is essential to properly outline the vermilion border of the lip before initiating the repair, ideally before any infiltration of local anesthesia if the surgeon anticipates that tumor extirpation might cross the vermilion border.
Repair
Closing then proceeds as follows:
• The cutaneous portion of the defect is drawn out in standard V to Y fashion, carrying the incision through the dermis and into subcutaneous tissue. The pedicle of the flap is maintained at the base of the island, serving as the blood supply to the flap.
• The periphery of the flap and surrounding tissue is undermined to facilitate movement superiorly into the cutaneous portion of the defect.
• A single buried vertical mattress suture can be placed at the advancing border of the island, holding it in place at the anticipated location of the vermilion border. The secondary defect created by the advancing V is closed to help push the island into place and prevent downward tension on the free margin of the lip.
• The remaining defect of the vermilion lip is closed by removing Burow triangles at the horizontal edges on each side of the remaining defect (Figure, B). The triangles are removed completely within the mucosal lip, with the inferior edge of the triangle placed at the vermilion border.
• The defect is closed in a primary linear horizontal fashion, using buried vertical mattress sutures and cutaneous approximation.
The final appearance of the sutured defect yields a small lateral extension at the superior edge of the V to Y closure, giving the appearance of wings on the Y, prompting us to term the closure V-to-flying-Y (Figure, C).
Although the limited portion of the mucosal lip that is closed in this fashion might appear thinner than the remaining lip, it generally yields a cosmetically acceptable result in the properly selected patient. Our experience also has shown an improvement in this difference in the months following repair. A full mucosal advancement flap might result in a more uniform appearance of the lower lip, but it is a larger and more difficult procedure for the patient to endure. Additionally, a full mucosal advancement flap risks uniformly creating a much thinner lip.
Postoperative Course
Sutures were removed 1 week postoperatively. Proper location of the vermilion border, without distortion of the free margin, was demonstrated. At 11-month follow-up, excellent cosmetic and functional results were noted (Figure, D).
Practice Implications
This repair (1) demonstrates an elegant method of closing a relatively shallow defect that crosses the vermilion border and (2) allows the surgeon to address each cosmetic subunit individually. We have found that this repair provides excellent cosmetic and functional results, with little morbidity.
The lip is a common site of nonmelanoma squamous cell carcinoma. Poorly planned closing after excision of the tumor risks notable impairment of function or cosmetic distortion. When a defect of the lip crosses cosmetic subunits, it is helpful to repair each subunit individually. V-to-flying-Y closure is an effective method to close defects that cross the vermilion border, resulting in well-preserved cosmetic appearance and function.
- Ishii LE, Byrne PJ. Lip reconstruction. Facial Plast Surg Clin North Am. 2009;17:445-453. doi:10.1016/j.fsc.2009.05.007
- Sebben JE. Wedge resection of the lip: minimizing problems. J Dermatol Surg Oncol. 1985;11:60-64. doi:10.1111/j.1524-4725.1985.tb02892.x
- Jin X, Teng L, Zhang C, et al. Reconstruction of partial-thickness vermilion defects with a mucosal V-Y advancement flap based on the orbicularis oris muscle. J Plast Reconstr Aesthet Surg. 2011;64:472-476. doi:10.1016/j.bjps.2010.07.017
- Ishii LE, Byrne PJ. Lip reconstruction. Facial Plast Surg Clin North Am. 2009;17:445-453. doi:10.1016/j.fsc.2009.05.007
- Sebben JE. Wedge resection of the lip: minimizing problems. J Dermatol Surg Oncol. 1985;11:60-64. doi:10.1111/j.1524-4725.1985.tb02892.x
- Jin X, Teng L, Zhang C, et al. Reconstruction of partial-thickness vermilion defects with a mucosal V-Y advancement flap based on the orbicularis oris muscle. J Plast Reconstr Aesthet Surg. 2011;64:472-476. doi:10.1016/j.bjps.2010.07.017
Tanning Attitudes and Behaviors in Adolescents and Young Adults
Intentional tanning—through sun exposure and tanning beds—is an easily avoidable contributor to skin cancer development and an important area for public education. Since the advent of social media, a correlation between social media use and increased indoor tanning behaviors has been reported.1 In 2010, 11.3% of US adults aged 18 to 29 years reported using a tanning bed in the last 12 months.2 The American Academy of Dermatology first published their “Position Statement on Indoor Tanning” in 1998, endorsing a ban on the sale of indoor tanning equipment for nonmedical purposes.3
Although there has been no outright ban on indoor tanning, regulations have been put in place in many states—including Texas, where (as of 2013) a person younger than 18 years must have written consent from their parent(s) to use a tanning bed. Despite efforts of organizations including the American Academy of Dermatology and the government to educate the public on skin cancer prevention and sun safety, the skin cancer rate has been steadily increasing over the last 20 years.
There is a constant campaign among dermatologists to educate their patients on how to reduce or avoid the risk for skin cancer, including the use of sunscreen and avoidance of tanning. Adolescents and young adults are an especially important demographic to reach and educate because increased UV light exposure during these years leads to a greatly increased risk for skin cancer later in life.4 Data on the overall prevalence of tanning and the demographics of participation in tanning activities are important to capture and can be used to efficiently target higher-risk populations.
In this study, we aimed to investigate the attitudes and behaviors of adolescents and young adults regarding sun protection and tanning. We also aimed to determine which avenues, including social media, would be most effective at educating about skin cancer awareness and sun protection to the higher-risk younger population.
Materials and Methods
We developed an institutional review board–approved protocol for the prospective collection of data from registered patients at the dermatology clinic of the Mays Cancer Center at the University of Texas Health at San Antonio. A paper survey containing 15 rating-scale questions was administered to 60 patients aged 13 to 27 years. Surveys were administered during intake, prior to the patients’ visit with a dermatologist; all visits were of a functional (not cosmetic) nature. Data collection spanned June to August 2018. Survey results were entered into Research Electronic Data Capture (REDCap) software for qualitative analysis.
Results
Sixty patients responded to the survey. The mean age of respondents was 19.5 years. No surveys were excluded from the data set. Table 1 provides baseline characteristics of respondents. Some respondents left questions unanswered, resulting in questions with fewer than 60 responses.

Among respondents to the survey, 70% (42/60) reported it is very important to protect their skin from sun exposure, and 30% (18/60) reported it is somewhat important. Regarding sunscreen use, 70% (42/60) indicated they use sunscreen only before outdoor activities, 12% (7/60) use sunscreen daily, and 17% (10/60) never use sunscreen. Of those who use sunscreen, 52% (28/54) do so to prevent skin damage and aging and 44% (24/54) to prevent skin cancer. Twenty-three percent (13/56) of respondents reported finding tanned skin attractive; 26% (14/55) reported wanting to be tan. Looking at race, 28% (10/36) of Whites, 25% (5/20) of Spanish/Hispanic/Latinos, and 22% (2/9) of Asians found tanned skin attractive; no Black respondents found tanned skin attractive.
Regarding tanning, 12% (7/57) reported using a tanning bed in their lifetime and 4% (2/57) in the last year; 34% (19/56) reported deliberately tanning outdoors; and 9% (5/56) reported using sunless or spray-on tanning. Dermatologists (75% [42/56]), primary care physicians (69.6% [39/56]), and parents (46.4% [26/56]) were perceived as more effective sources of skin care education; among media modalities, television (33.9% [19/56]), Instagram (30.4% [17/60]), and YouTube (23.2% [13/60]) were perceived as more effective sources of skin care education (Table 2).
Comment
Perceptions of Tanning
Almost one-quarter of respondents found tanned skin attractive, which might reflect a shift from prior generations. Compared to the 11% of respondents in the 2010 survey,2 only 3.5% (2/57) of our respondents reported using a tanning bed in the last year, which could reflect the results of recent Texas legislation restricting the use of tanning beds by adolescents.
An alarming number of respondents reported going outdoors with the intention of tanning; although it appears that indoor tanning education has been successful, this finding shows that there is still a need for sun protection education because outdoor tanning is not a suitable alternative. A small number of respondents reported getting a sunless or spray-on tan, which is a risk-free alternative to indoor tanning.
Despite all respondents stating that protecting skin from the sun is important, most respondents surveyed do not use sunscreen daily. More respondents use sunscreen to prevent damage and aging than to prevent skin cancer. Young people might be more alarmed by the threat of early aging and losing their “youthful appearance” than by the possibility of developing skin cancer in the distant future. This discrepancy might indicate a lack of knowledge and be an important focus for future education efforts.
Perceptions of Trustworthiness of Education Sources
Our findings show dermatologists and primary care physicians are important educators on skin protection. Primary care physicians should remain vigilant to recognize at-risk patients who would benefit from skin protection education, especially those who do not see a dermatologist. Education of young people focusing on their concern over maintaining a youthful appearance instead of the possibility of developing skin cancer in the future might be more effective.
Although education provided by a physician is effective, using media—particularly social media—might be more efficient. Television, Instagram, and YouTube were listed by respondents as the 3 most preferred media outlets for skin health education, which shows important areas of focus for future advertising. Facebook was listed at a surprisingly low level, possibly showing the change in use of certain social media websites among this age group. According to the Pew Research Center, the most widely used social media apps among young adults aged 18 to 29 years are YouTube (91%), Facebook (63%), Instagram (67%), and Snapchat (62%). More than half of the same demographic visit Facebook (74%), Instagram (63%), Snapchat (61%), and YouTube (51%) daily.5 Although respondents to our survey were not specifically asked about the frequency of their use of social media and our data set includes patients younger than 18 years, we know that social media use has been increasing over the last decade among adolescents.1 Therefore, we assume that more than one-half of respondents to our survey use their reported social media platforms daily.
Social media is an underused medium for skin cancer prevention education and can reach those who do not regularly see a dermatologist. Unlike printed pamphlets and posters, advertisements through social media can use metrics such as age, race, gender, and interests to target high-risk individuals.
Study Limitations
This was a single-site study of currently enrolled dermatology patients who might be more aware of skin protection than the general population because they are being treated by a dermatologist. Survey questions regarding demographics, required by our institution, could not effectively differentiate Hispanic and White patients. Respondents could have been subject to the Hawthorne effect—awareness that their behavior is being observed—when responding to the survey because it was administered in the office prior to being seen by a dermatologist.
- Falzone AE, Brindis CD, Chren M-M, et al. Teens, tweets, and tanning beds: rethinking the use of social media for skin cancer prevention. Am J Prev Med. 2017;53(3 suppl 1):S86-S94.
- Centers for Disease Control and Prevention. Use of indoor tanning devices by adults—United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61:323-326.
- American Academy of Dermatology. Position statement on indoor tanning. Amended November 14, 2009. Accessed January 10, 2021. https://server.aad.org/Forms/Policies/Uploads/PS/PS-Indoor%20Tanning%2011-16-09.pdf?
- American Academy of Dermatology. Indoor tanning. Accessed January 10, 2020. https://www.aad.org/media/stats-indoor-tanning
- Perrin A, Anderson M. Share of U.S. adults using social media, including Facebook, is mostly unchanged since 2018. Pew Research Center; April 10, 2019. Accessed April 16, 2021. https://www.pewresearch.org/fact-tank/2019/04/10/share-of-u-s-adults-using-social-media-including-facebook-is-mostly-unchanged-since-2018/
Intentional tanning—through sun exposure and tanning beds—is an easily avoidable contributor to skin cancer development and an important area for public education. Since the advent of social media, a correlation between social media use and increased indoor tanning behaviors has been reported.1 In 2010, 11.3% of US adults aged 18 to 29 years reported using a tanning bed in the last 12 months.2 The American Academy of Dermatology first published their “Position Statement on Indoor Tanning” in 1998, endorsing a ban on the sale of indoor tanning equipment for nonmedical purposes.3
Although there has been no outright ban on indoor tanning, regulations have been put in place in many states—including Texas, where (as of 2013) a person younger than 18 years must have written consent from their parent(s) to use a tanning bed. Despite efforts of organizations including the American Academy of Dermatology and the government to educate the public on skin cancer prevention and sun safety, the skin cancer rate has been steadily increasing over the last 20 years.
There is a constant campaign among dermatologists to educate their patients on how to reduce or avoid the risk for skin cancer, including the use of sunscreen and avoidance of tanning. Adolescents and young adults are an especially important demographic to reach and educate because increased UV light exposure during these years leads to a greatly increased risk for skin cancer later in life.4 Data on the overall prevalence of tanning and the demographics of participation in tanning activities are important to capture and can be used to efficiently target higher-risk populations.
In this study, we aimed to investigate the attitudes and behaviors of adolescents and young adults regarding sun protection and tanning. We also aimed to determine which avenues, including social media, would be most effective at educating about skin cancer awareness and sun protection to the higher-risk younger population.
Materials and Methods
We developed an institutional review board–approved protocol for the prospective collection of data from registered patients at the dermatology clinic of the Mays Cancer Center at the University of Texas Health at San Antonio. A paper survey containing 15 rating-scale questions was administered to 60 patients aged 13 to 27 years. Surveys were administered during intake, prior to the patients’ visit with a dermatologist; all visits were of a functional (not cosmetic) nature. Data collection spanned June to August 2018. Survey results were entered into Research Electronic Data Capture (REDCap) software for qualitative analysis.
Results
Sixty patients responded to the survey. The mean age of respondents was 19.5 years. No surveys were excluded from the data set. Table 1 provides baseline characteristics of respondents. Some respondents left questions unanswered, resulting in questions with fewer than 60 responses.

Among respondents to the survey, 70% (42/60) reported it is very important to protect their skin from sun exposure, and 30% (18/60) reported it is somewhat important. Regarding sunscreen use, 70% (42/60) indicated they use sunscreen only before outdoor activities, 12% (7/60) use sunscreen daily, and 17% (10/60) never use sunscreen. Of those who use sunscreen, 52% (28/54) do so to prevent skin damage and aging and 44% (24/54) to prevent skin cancer. Twenty-three percent (13/56) of respondents reported finding tanned skin attractive; 26% (14/55) reported wanting to be tan. Looking at race, 28% (10/36) of Whites, 25% (5/20) of Spanish/Hispanic/Latinos, and 22% (2/9) of Asians found tanned skin attractive; no Black respondents found tanned skin attractive.
Regarding tanning, 12% (7/57) reported using a tanning bed in their lifetime and 4% (2/57) in the last year; 34% (19/56) reported deliberately tanning outdoors; and 9% (5/56) reported using sunless or spray-on tanning. Dermatologists (75% [42/56]), primary care physicians (69.6% [39/56]), and parents (46.4% [26/56]) were perceived as more effective sources of skin care education; among media modalities, television (33.9% [19/56]), Instagram (30.4% [17/60]), and YouTube (23.2% [13/60]) were perceived as more effective sources of skin care education (Table 2).

Comment
Perceptions of Tanning
Almost one-quarter of respondents found tanned skin attractive, which might reflect a shift from prior generations. Compared to the 11% of respondents in the 2010 survey,2 only 3.5% (2/57) of our respondents reported using a tanning bed in the last year, which could reflect the results of recent Texas legislation restricting the use of tanning beds by adolescents.
An alarming number of respondents reported going outdoors with the intention of tanning; although it appears that indoor tanning education has been successful, this finding shows that there is still a need for sun protection education because outdoor tanning is not a suitable alternative. A small number of respondents reported getting a sunless or spray-on tan, which is a risk-free alternative to indoor tanning.
Despite all respondents stating that protecting skin from the sun is important, most respondents surveyed do not use sunscreen daily. More respondents use sunscreen to prevent damage and aging than to prevent skin cancer. Young people might be more alarmed by the threat of early aging and losing their “youthful appearance” than by the possibility of developing skin cancer in the distant future. This discrepancy might indicate a lack of knowledge and be an important focus for future education efforts.
Perceptions of Trustworthiness of Education Sources
Our findings show dermatologists and primary care physicians are important educators on skin protection. Primary care physicians should remain vigilant to recognize at-risk patients who would benefit from skin protection education, especially those who do not see a dermatologist. Education of young people focusing on their concern over maintaining a youthful appearance instead of the possibility of developing skin cancer in the future might be more effective.
Although education provided by a physician is effective, using media—particularly social media—might be more efficient. Television, Instagram, and YouTube were listed by respondents as the 3 most preferred media outlets for skin health education, which shows important areas of focus for future advertising. Facebook was listed at a surprisingly low level, possibly showing the change in use of certain social media websites among this age group. According to the Pew Research Center, the most widely used social media apps among young adults aged 18 to 29 years are YouTube (91%), Facebook (63%), Instagram (67%), and Snapchat (62%). More than half of the same demographic visit Facebook (74%), Instagram (63%), Snapchat (61%), and YouTube (51%) daily.5 Although respondents to our survey were not specifically asked about the frequency of their use of social media and our data set includes patients younger than 18 years, we know that social media use has been increasing over the last decade among adolescents.1 Therefore, we assume that more than one-half of respondents to our survey use their reported social media platforms daily.
Social media is an underused medium for skin cancer prevention education and can reach those who do not regularly see a dermatologist. Unlike printed pamphlets and posters, advertisements through social media can use metrics such as age, race, gender, and interests to target high-risk individuals.
Study Limitations
This was a single-site study of currently enrolled dermatology patients who might be more aware of skin protection than the general population because they are being treated by a dermatologist. Survey questions regarding demographics, required by our institution, could not effectively differentiate Hispanic and White patients. Respondents could have been subject to the Hawthorne effect—awareness that their behavior is being observed—when responding to the survey because it was administered in the office prior to being seen by a dermatologist.
Intentional tanning—through sun exposure and tanning beds—is an easily avoidable contributor to skin cancer development and an important area for public education. Since the advent of social media, a correlation between social media use and increased indoor tanning behaviors has been reported.1 In 2010, 11.3% of US adults aged 18 to 29 years reported using a tanning bed in the last 12 months.2 The American Academy of Dermatology first published their “Position Statement on Indoor Tanning” in 1998, endorsing a ban on the sale of indoor tanning equipment for nonmedical purposes.3
Although there has been no outright ban on indoor tanning, regulations have been put in place in many states—including Texas, where (as of 2013) a person younger than 18 years must have written consent from their parent(s) to use a tanning bed. Despite efforts of organizations including the American Academy of Dermatology and the government to educate the public on skin cancer prevention and sun safety, the skin cancer rate has been steadily increasing over the last 20 years.
There is a constant campaign among dermatologists to educate their patients on how to reduce or avoid the risk for skin cancer, including the use of sunscreen and avoidance of tanning. Adolescents and young adults are an especially important demographic to reach and educate because increased UV light exposure during these years leads to a greatly increased risk for skin cancer later in life.4 Data on the overall prevalence of tanning and the demographics of participation in tanning activities are important to capture and can be used to efficiently target higher-risk populations.
In this study, we aimed to investigate the attitudes and behaviors of adolescents and young adults regarding sun protection and tanning. We also aimed to determine which avenues, including social media, would be most effective at educating about skin cancer awareness and sun protection to the higher-risk younger population.
Materials and Methods
We developed an institutional review board–approved protocol for the prospective collection of data from registered patients at the dermatology clinic of the Mays Cancer Center at the University of Texas Health at San Antonio. A paper survey containing 15 rating-scale questions was administered to 60 patients aged 13 to 27 years. Surveys were administered during intake, prior to the patients’ visit with a dermatologist; all visits were of a functional (not cosmetic) nature. Data collection spanned June to August 2018. Survey results were entered into Research Electronic Data Capture (REDCap) software for qualitative analysis.
Results
Sixty patients responded to the survey. The mean age of respondents was 19.5 years. No surveys were excluded from the data set. Table 1 provides baseline characteristics of respondents. Some respondents left questions unanswered, resulting in questions with fewer than 60 responses.

Among respondents to the survey, 70% (42/60) reported it is very important to protect their skin from sun exposure, and 30% (18/60) reported it is somewhat important. Regarding sunscreen use, 70% (42/60) indicated they use sunscreen only before outdoor activities, 12% (7/60) use sunscreen daily, and 17% (10/60) never use sunscreen. Of those who use sunscreen, 52% (28/54) do so to prevent skin damage and aging and 44% (24/54) to prevent skin cancer. Twenty-three percent (13/56) of respondents reported finding tanned skin attractive; 26% (14/55) reported wanting to be tan. Looking at race, 28% (10/36) of Whites, 25% (5/20) of Spanish/Hispanic/Latinos, and 22% (2/9) of Asians found tanned skin attractive; no Black respondents found tanned skin attractive.
Regarding tanning, 12% (7/57) reported using a tanning bed in their lifetime and 4% (2/57) in the last year; 34% (19/56) reported deliberately tanning outdoors; and 9% (5/56) reported using sunless or spray-on tanning. Dermatologists (75% [42/56]), primary care physicians (69.6% [39/56]), and parents (46.4% [26/56]) were perceived as more effective sources of skin care education; among media modalities, television (33.9% [19/56]), Instagram (30.4% [17/60]), and YouTube (23.2% [13/60]) were perceived as more effective sources of skin care education (Table 2).

Comment
Perceptions of Tanning
Almost one-quarter of respondents found tanned skin attractive, which might reflect a shift from prior generations. Compared to the 11% of respondents in the 2010 survey,2 only 3.5% (2/57) of our respondents reported using a tanning bed in the last year, which could reflect the results of recent Texas legislation restricting the use of tanning beds by adolescents.
An alarming number of respondents reported going outdoors with the intention of tanning; although it appears that indoor tanning education has been successful, this finding shows that there is still a need for sun protection education because outdoor tanning is not a suitable alternative. A small number of respondents reported getting a sunless or spray-on tan, which is a risk-free alternative to indoor tanning.
Despite all respondents stating that protecting skin from the sun is important, most respondents surveyed do not use sunscreen daily. More respondents use sunscreen to prevent damage and aging than to prevent skin cancer. Young people might be more alarmed by the threat of early aging and losing their “youthful appearance” than by the possibility of developing skin cancer in the distant future. This discrepancy might indicate a lack of knowledge and be an important focus for future education efforts.
Perceptions of Trustworthiness of Education Sources
Our findings show dermatologists and primary care physicians are important educators on skin protection. Primary care physicians should remain vigilant to recognize at-risk patients who would benefit from skin protection education, especially those who do not see a dermatologist. Education of young people focusing on their concern over maintaining a youthful appearance instead of the possibility of developing skin cancer in the future might be more effective.
Although education provided by a physician is effective, using media—particularly social media—might be more efficient. Television, Instagram, and YouTube were listed by respondents as the 3 most preferred media outlets for skin health education, which shows important areas of focus for future advertising. Facebook was listed at a surprisingly low level, possibly showing the change in use of certain social media websites among this age group. According to the Pew Research Center, the most widely used social media apps among young adults aged 18 to 29 years are YouTube (91%), Facebook (63%), Instagram (67%), and Snapchat (62%). More than half of the same demographic visit Facebook (74%), Instagram (63%), Snapchat (61%), and YouTube (51%) daily.5 Although respondents to our survey were not specifically asked about the frequency of their use of social media and our data set includes patients younger than 18 years, we know that social media use has been increasing over the last decade among adolescents.1 Therefore, we assume that more than one-half of respondents to our survey use their reported social media platforms daily.
Social media is an underused medium for skin cancer prevention education and can reach those who do not regularly see a dermatologist. Unlike printed pamphlets and posters, advertisements through social media can use metrics such as age, race, gender, and interests to target high-risk individuals.
Study Limitations
This was a single-site study of currently enrolled dermatology patients who might be more aware of skin protection than the general population because they are being treated by a dermatologist. Survey questions regarding demographics, required by our institution, could not effectively differentiate Hispanic and White patients. Respondents could have been subject to the Hawthorne effect—awareness that their behavior is being observed—when responding to the survey because it was administered in the office prior to being seen by a dermatologist.
- Falzone AE, Brindis CD, Chren M-M, et al. Teens, tweets, and tanning beds: rethinking the use of social media for skin cancer prevention. Am J Prev Med. 2017;53(3 suppl 1):S86-S94.
- Centers for Disease Control and Prevention. Use of indoor tanning devices by adults—United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61:323-326.
- American Academy of Dermatology. Position statement on indoor tanning. Amended November 14, 2009. Accessed January 10, 2021. https://server.aad.org/Forms/Policies/Uploads/PS/PS-Indoor%20Tanning%2011-16-09.pdf?
- American Academy of Dermatology. Indoor tanning. Accessed January 10, 2020. https://www.aad.org/media/stats-indoor-tanning
- Perrin A, Anderson M. Share of U.S. adults using social media, including Facebook, is mostly unchanged since 2018. Pew Research Center; April 10, 2019. Accessed April 16, 2021. https://www.pewresearch.org/fact-tank/2019/04/10/share-of-u-s-adults-using-social-media-including-facebook-is-mostly-unchanged-since-2018/
- Falzone AE, Brindis CD, Chren M-M, et al. Teens, tweets, and tanning beds: rethinking the use of social media for skin cancer prevention. Am J Prev Med. 2017;53(3 suppl 1):S86-S94.
- Centers for Disease Control and Prevention. Use of indoor tanning devices by adults—United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61:323-326.
- American Academy of Dermatology. Position statement on indoor tanning. Amended November 14, 2009. Accessed January 10, 2021. https://server.aad.org/Forms/Policies/Uploads/PS/PS-Indoor%20Tanning%2011-16-09.pdf?
- American Academy of Dermatology. Indoor tanning. Accessed January 10, 2020. https://www.aad.org/media/stats-indoor-tanning
- Perrin A, Anderson M. Share of U.S. adults using social media, including Facebook, is mostly unchanged since 2018. Pew Research Center; April 10, 2019. Accessed April 16, 2021. https://www.pewresearch.org/fact-tank/2019/04/10/share-of-u-s-adults-using-social-media-including-facebook-is-mostly-unchanged-since-2018/
PRACTICE POINTS
- Dermatologists are the preferred educators of skin care for adolescents and young adults.
- Social media is an underused medium for skin cancer prevention education and can reach those who do not regularly see a dermatologist.
- Education of young people focusing on their concerns about maintaining a youthful appearance instead of the possibility of developing skin cancer in the future might be more effective.