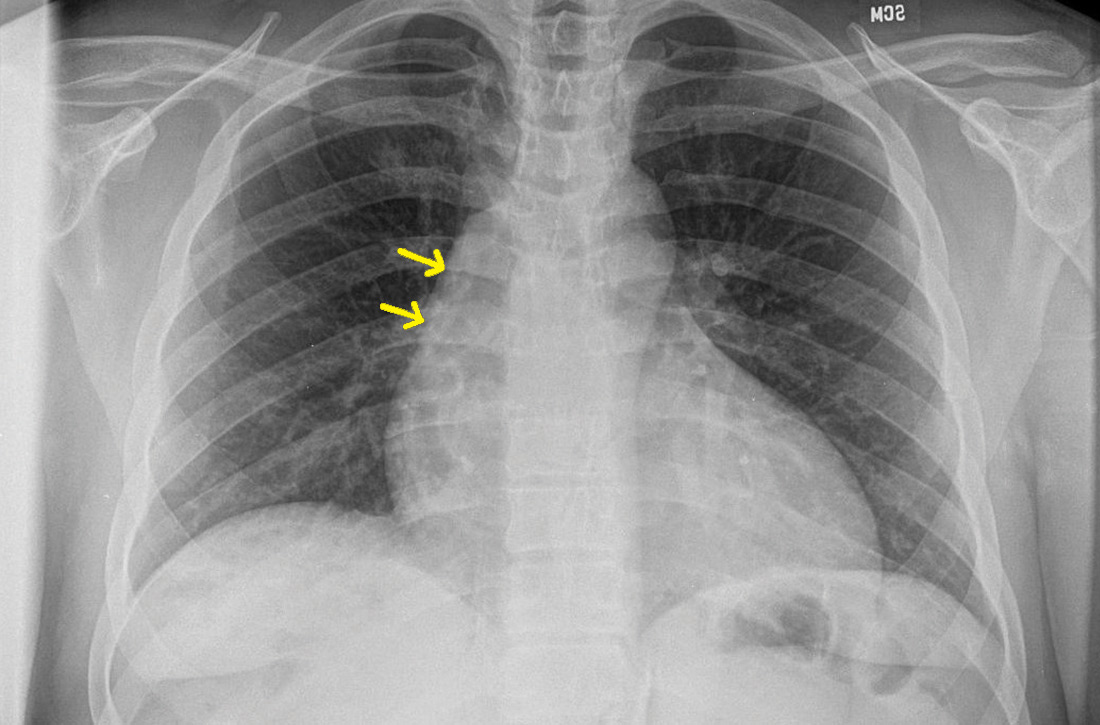
User login
Researchers examine factors associated with opioid use among migraineurs
Among patients with migraine who use prescription medications, the increasing use of prescription opioids is associated with chronic migraine, more severe disability, and anxiety and depression, according to an analysis published in the January issue of Headache . The use of prescription opioids also is associated with treatment-related variables such as poor acute treatment optimization and treatment in a pain clinic. The results indicate the continued need to educate patients and clinicians about the potential risks of opioids for migraineurs, according to the researchers.
In the Migraine in America Symptoms and Treatment (MAST) study, which the researchers analyzed for their investigation, one-third of migraineurs who use acute prescriptions reported using opioids. Among opioid users, 42% took opioids on 4 or more days per month. “These findings are like [those of] a previous report from the American Migraine Prevalence and Prevention study and more recent findings from the Observational Survey of the Epidemiology, Treatment, and Care of Migraine (OVERCOME) study,” said Richard Lipton, MD, Edwin S. Lowe professor and vice chair of neurology at Albert Einstein College of Medicine in the Bronx, New York. “High rates of opioid use are problematic because opioid use is associated with worsening of migraine over time.”
Opioids remain in widespread use for migraine, even though guidelines recommend against this treatment. Among migraineurs, opioid use is associated with more severe headache-related disability and greater use of health care resources. Opioid use also increases the risk of progressing from episodic migraine to chronic migraine.
A review of MAST data
Dr. Lipton and colleagues set out to identify the variables associated with the frequency of opioid use in people with migraine. Among the variables that they sought to examine were demographic characteristics, comorbidities, headache characteristics, medication use, and patterns of health care use. Dr. Lipton’s group hypothesized that migraine-related severity and burden would increase with increasing frequency of opioid use.
To conduct their research, the investigators examined data from the MAST study, a nationwide sample of American adults with migraine. They focused specifically on participants who reported receiving prescription acute medications. Participants eligible for this analysis reported 3 or more headache days in the previous 3 months and at least 1 monthly headache day in the previous month. In all, 15,133 participants met these criteria.
Dr. Lipton and colleagues categorized participants into four groups based on their frequency of opioid use. The groups had no opioid use, 3 or fewer monthly days of opioid use, 4 to 9 monthly days of opioid use, and 10 or more days of monthly opioid use. The last category is consistent with the International Classification of Headache Disorders-3 criteria for overuse of opioids in migraine.
At baseline, MAST participants provided information about variables such as gender, age, marital status, smoking status, education, and income. Participants also reported how many times in the previous 6 months they had visited a primary care doctor, a neurologist, a headache specialist, or a pain specialist. Dr. Lipton’s group calculated monthly headache days using the number of days during the previous 3 months affected by headache. The Migraine Disability Assessment (MIDAS) questionnaire was used to measure headache-related disability. The four-item Patient Health Questionnaire (PHQ-4) was used to screen for anxiety and depression, and the Migraine Treatment Optimization Questionnaire (mTOQ-4) evaluated participants’ treatment optimization.
Men predominated among opioid users
The investigators included 4,701 MAST participants in their analysis. The population’s mean age was 45 years, and 71.6% of participants were women. Of the entire sample, 67.5% reported no opioid use, and 32.5% reported opioid use. Of the total study population, 18.7% of patients took opioids 3 or fewer days per month, 6.5% took opioids 4 to 9 days per month, and 7.3% took opioids on 10 or more days per month.
Opioid users did not differ from nonusers on race or marital status. Men were overrepresented among all groups of opioid users, however. In addition, opioid use was more prevalent among participants with fewer than 4 years of college education (34.9%) than among participants with 4 or more years of college (30.8%). The proportion of participants with fewer than 4 years of college increased with increasing monthly opioid use. Furthermore, opioid use increased with decreasing household income. As opioid use increased, rates of employment decreased. Approximately 33% of the entire sample were obese, and the proportion of obese participants increased with increasing days per month of opioid use.
The most frequent setting during the previous 6 months for participants seeking care was primary care (49.7%). The next most frequent setting was neurology units (20.9%), pain clinics (8.3%), and headache clinics (7.7%). The prevalence of opioid use was 37.5% among participants with primary care visits, 37.3% among participants with neurologist visits, 43.0% among participants with headache clinic visits, and 53.5% with pain clinic visits.
About 15% of the population had chronic migraine. The prevalence of chronic migraine increased with increasing frequency of opioid use. About 49% of the sample had allodynia, and the prevalence of allodynia increased with increasing frequency of opioid use. Overall, disability was moderate to severe in 57.3% of participants. Participants who used opioids on 3 or fewer days per month had the lowest prevalence of moderate to severe disability (50.2%), and participants who used opioids on 10 or more days per month had the highest prevalence of moderate to severe disability (83.8%).
Approximately 21% of participants had anxiety or depression. The lowest prevalence of anxiety or depression was among participants who took opioids on 3 or fewer days per month (17.4%), and the highest prevalence was among participants who took opioids on 10 or more days per month (43.2%). About 39% of the population had very poor to poor treatment optimization. Among opioid nonusers, 35.6% had very poor to poor treatment optimization, and 59.4% of participants who used opioids on 10 or more days per month had very poor to poor treatment optimization.
Dr. Lipton and colleagues also examined the study population’s use of triptans. Overall, 51.5% of participants reported taking triptans. The prevalence of triptan use was highest among participants who did not use opioids (64.1%) and lowest among participants who used opioids on 3 or fewer days per month (20.5%). Triptan use increased as monthly days of opioid use increased.
Pain clinics and opioid prescription
“In the general population, women are more likely to receive opioids than men,” said Dr. Lipton. “This [finding] could reflect, in part, that women have more pain disorders than men and are more likely to seek medical care for pain than men.” In the current study, however, men with migraine were more likely to receive opioid prescriptions than were women with migraine. One potential explanation for this finding is that men with migraine are less likely to receive a migraine diagnosis, which might attenuate opioid prescribing, than women with migraine. “It may be that opioids are perceived to be serious drugs for serious pain, and that some physicians may be more likely to prescribe opioids to men because the disorder is taken more seriously in men than women,” said Dr. Lipton.
The observation that opioids were more likely to be prescribed for people treated in pain clinics “is consistent with my understanding of practice patterns,” he added. “Generally, neurologists strive to find effective acute treatment alternatives to opioids. The emergence of [drug classes known as] gepants and ditans provides a helpful set of alternatives to tritpans.”
Dr. Lipton and his colleagues plan further research into the treatment of migraineurs. “In a claims analysis, we showed that when people with migraine fail a triptan, they are most likely to get an opioid as their next drug,” he said. “Reasonable [clinicians] might disagree on the next step. The next step, in the absence of contraindications, could be a different oral triptan, a nonoral triptan, or a gepant or ditan. We are planning a randomized trial to probe this question.”
Why are opioids still being used?
The study’s reliance on patients’ self-report and its retrospective design are two of its weaknesses, said Alan M. Rapoport, MD, clinical professor of neurology at the University of California, Los Angeles, and editor-in-chief of Neurology Reviews. One strength, however, is that the stratified sampling methodology produced a study population that accurately reflects the demographic characteristics of the U.S. adult population, he added. Another strength is the investigators’ examination of opioid use by patient characteristics such as marital status, education, income, obesity, and smoking.
Given the harmful effects of opioids in migraine, it is hard to understand why as much as one-third of study participants using acute care medication for migraine were using opioids, said Dr. Rapoport. Using opioids for the acute treatment of migraine attacks often indicates inadequate treatment optimization, which leads to ongoing headache. As a consequence, patients may take more medication, which can increase headache frequency and lead to diagnoses of chronic migraine and medication overuse headache. Although the study found an association between the increased use of opioids and decreased household income and increased unemployment, smoking, and obesity, “it is not possible to assign causality to any of these associations, even though some would argue that decreased socioeconomic status was somehow related to more headache, disability, obesity, smoking, and unemployment,” he added.
“The paper suggests that future research should look at the risk factors for use of opioids and should determine if depression is a risk factor for or a consequence of opioid use,” said Dr. Rapoport. “Interventional studies designed to improve the acute care of migraine attacks might be able to reduce the use of opioids. I have not used opioids or butalbital-containing medication in my office for many years.”
This study was funded and sponsored by Dr. Reddy’s Laboratories group of companies, Princeton, N.J. Dr. Lipton has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as a consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Biohaven, Dr. Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva, Inc. He receives royalties from Wolff’s Headache, 8th Edition (New York: Oxford University Press, 2009) and holds stock options in eNeura Therapeutics and Biohaven.
SOURCE: Lipton RB, et al. Headache. https://doi.org/10.1111/head.14018. 2020;61(1):103-16.
Among patients with migraine who use prescription medications, the increasing use of prescription opioids is associated with chronic migraine, more severe disability, and anxiety and depression, according to an analysis published in the January issue of Headache . The use of prescription opioids also is associated with treatment-related variables such as poor acute treatment optimization and treatment in a pain clinic. The results indicate the continued need to educate patients and clinicians about the potential risks of opioids for migraineurs, according to the researchers.
In the Migraine in America Symptoms and Treatment (MAST) study, which the researchers analyzed for their investigation, one-third of migraineurs who use acute prescriptions reported using opioids. Among opioid users, 42% took opioids on 4 or more days per month. “These findings are like [those of] a previous report from the American Migraine Prevalence and Prevention study and more recent findings from the Observational Survey of the Epidemiology, Treatment, and Care of Migraine (OVERCOME) study,” said Richard Lipton, MD, Edwin S. Lowe professor and vice chair of neurology at Albert Einstein College of Medicine in the Bronx, New York. “High rates of opioid use are problematic because opioid use is associated with worsening of migraine over time.”
Opioids remain in widespread use for migraine, even though guidelines recommend against this treatment. Among migraineurs, opioid use is associated with more severe headache-related disability and greater use of health care resources. Opioid use also increases the risk of progressing from episodic migraine to chronic migraine.
A review of MAST data
Dr. Lipton and colleagues set out to identify the variables associated with the frequency of opioid use in people with migraine. Among the variables that they sought to examine were demographic characteristics, comorbidities, headache characteristics, medication use, and patterns of health care use. Dr. Lipton’s group hypothesized that migraine-related severity and burden would increase with increasing frequency of opioid use.
To conduct their research, the investigators examined data from the MAST study, a nationwide sample of American adults with migraine. They focused specifically on participants who reported receiving prescription acute medications. Participants eligible for this analysis reported 3 or more headache days in the previous 3 months and at least 1 monthly headache day in the previous month. In all, 15,133 participants met these criteria.
Dr. Lipton and colleagues categorized participants into four groups based on their frequency of opioid use. The groups had no opioid use, 3 or fewer monthly days of opioid use, 4 to 9 monthly days of opioid use, and 10 or more days of monthly opioid use. The last category is consistent with the International Classification of Headache Disorders-3 criteria for overuse of opioids in migraine.
At baseline, MAST participants provided information about variables such as gender, age, marital status, smoking status, education, and income. Participants also reported how many times in the previous 6 months they had visited a primary care doctor, a neurologist, a headache specialist, or a pain specialist. Dr. Lipton’s group calculated monthly headache days using the number of days during the previous 3 months affected by headache. The Migraine Disability Assessment (MIDAS) questionnaire was used to measure headache-related disability. The four-item Patient Health Questionnaire (PHQ-4) was used to screen for anxiety and depression, and the Migraine Treatment Optimization Questionnaire (mTOQ-4) evaluated participants’ treatment optimization.
Men predominated among opioid users
The investigators included 4,701 MAST participants in their analysis. The population’s mean age was 45 years, and 71.6% of participants were women. Of the entire sample, 67.5% reported no opioid use, and 32.5% reported opioid use. Of the total study population, 18.7% of patients took opioids 3 or fewer days per month, 6.5% took opioids 4 to 9 days per month, and 7.3% took opioids on 10 or more days per month.
Opioid users did not differ from nonusers on race or marital status. Men were overrepresented among all groups of opioid users, however. In addition, opioid use was more prevalent among participants with fewer than 4 years of college education (34.9%) than among participants with 4 or more years of college (30.8%). The proportion of participants with fewer than 4 years of college increased with increasing monthly opioid use. Furthermore, opioid use increased with decreasing household income. As opioid use increased, rates of employment decreased. Approximately 33% of the entire sample were obese, and the proportion of obese participants increased with increasing days per month of opioid use.
The most frequent setting during the previous 6 months for participants seeking care was primary care (49.7%). The next most frequent setting was neurology units (20.9%), pain clinics (8.3%), and headache clinics (7.7%). The prevalence of opioid use was 37.5% among participants with primary care visits, 37.3% among participants with neurologist visits, 43.0% among participants with headache clinic visits, and 53.5% with pain clinic visits.
About 15% of the population had chronic migraine. The prevalence of chronic migraine increased with increasing frequency of opioid use. About 49% of the sample had allodynia, and the prevalence of allodynia increased with increasing frequency of opioid use. Overall, disability was moderate to severe in 57.3% of participants. Participants who used opioids on 3 or fewer days per month had the lowest prevalence of moderate to severe disability (50.2%), and participants who used opioids on 10 or more days per month had the highest prevalence of moderate to severe disability (83.8%).
Approximately 21% of participants had anxiety or depression. The lowest prevalence of anxiety or depression was among participants who took opioids on 3 or fewer days per month (17.4%), and the highest prevalence was among participants who took opioids on 10 or more days per month (43.2%). About 39% of the population had very poor to poor treatment optimization. Among opioid nonusers, 35.6% had very poor to poor treatment optimization, and 59.4% of participants who used opioids on 10 or more days per month had very poor to poor treatment optimization.
Dr. Lipton and colleagues also examined the study population’s use of triptans. Overall, 51.5% of participants reported taking triptans. The prevalence of triptan use was highest among participants who did not use opioids (64.1%) and lowest among participants who used opioids on 3 or fewer days per month (20.5%). Triptan use increased as monthly days of opioid use increased.
Pain clinics and opioid prescription
“In the general population, women are more likely to receive opioids than men,” said Dr. Lipton. “This [finding] could reflect, in part, that women have more pain disorders than men and are more likely to seek medical care for pain than men.” In the current study, however, men with migraine were more likely to receive opioid prescriptions than were women with migraine. One potential explanation for this finding is that men with migraine are less likely to receive a migraine diagnosis, which might attenuate opioid prescribing, than women with migraine. “It may be that opioids are perceived to be serious drugs for serious pain, and that some physicians may be more likely to prescribe opioids to men because the disorder is taken more seriously in men than women,” said Dr. Lipton.
The observation that opioids were more likely to be prescribed for people treated in pain clinics “is consistent with my understanding of practice patterns,” he added. “Generally, neurologists strive to find effective acute treatment alternatives to opioids. The emergence of [drug classes known as] gepants and ditans provides a helpful set of alternatives to tritpans.”
Dr. Lipton and his colleagues plan further research into the treatment of migraineurs. “In a claims analysis, we showed that when people with migraine fail a triptan, they are most likely to get an opioid as their next drug,” he said. “Reasonable [clinicians] might disagree on the next step. The next step, in the absence of contraindications, could be a different oral triptan, a nonoral triptan, or a gepant or ditan. We are planning a randomized trial to probe this question.”
Why are opioids still being used?
The study’s reliance on patients’ self-report and its retrospective design are two of its weaknesses, said Alan M. Rapoport, MD, clinical professor of neurology at the University of California, Los Angeles, and editor-in-chief of Neurology Reviews. One strength, however, is that the stratified sampling methodology produced a study population that accurately reflects the demographic characteristics of the U.S. adult population, he added. Another strength is the investigators’ examination of opioid use by patient characteristics such as marital status, education, income, obesity, and smoking.
Given the harmful effects of opioids in migraine, it is hard to understand why as much as one-third of study participants using acute care medication for migraine were using opioids, said Dr. Rapoport. Using opioids for the acute treatment of migraine attacks often indicates inadequate treatment optimization, which leads to ongoing headache. As a consequence, patients may take more medication, which can increase headache frequency and lead to diagnoses of chronic migraine and medication overuse headache. Although the study found an association between the increased use of opioids and decreased household income and increased unemployment, smoking, and obesity, “it is not possible to assign causality to any of these associations, even though some would argue that decreased socioeconomic status was somehow related to more headache, disability, obesity, smoking, and unemployment,” he added.
“The paper suggests that future research should look at the risk factors for use of opioids and should determine if depression is a risk factor for or a consequence of opioid use,” said Dr. Rapoport. “Interventional studies designed to improve the acute care of migraine attacks might be able to reduce the use of opioids. I have not used opioids or butalbital-containing medication in my office for many years.”
This study was funded and sponsored by Dr. Reddy’s Laboratories group of companies, Princeton, N.J. Dr. Lipton has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as a consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Biohaven, Dr. Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva, Inc. He receives royalties from Wolff’s Headache, 8th Edition (New York: Oxford University Press, 2009) and holds stock options in eNeura Therapeutics and Biohaven.
SOURCE: Lipton RB, et al. Headache. https://doi.org/10.1111/head.14018. 2020;61(1):103-16.
Among patients with migraine who use prescription medications, the increasing use of prescription opioids is associated with chronic migraine, more severe disability, and anxiety and depression, according to an analysis published in the January issue of Headache . The use of prescription opioids also is associated with treatment-related variables such as poor acute treatment optimization and treatment in a pain clinic. The results indicate the continued need to educate patients and clinicians about the potential risks of opioids for migraineurs, according to the researchers.
In the Migraine in America Symptoms and Treatment (MAST) study, which the researchers analyzed for their investigation, one-third of migraineurs who use acute prescriptions reported using opioids. Among opioid users, 42% took opioids on 4 or more days per month. “These findings are like [those of] a previous report from the American Migraine Prevalence and Prevention study and more recent findings from the Observational Survey of the Epidemiology, Treatment, and Care of Migraine (OVERCOME) study,” said Richard Lipton, MD, Edwin S. Lowe professor and vice chair of neurology at Albert Einstein College of Medicine in the Bronx, New York. “High rates of opioid use are problematic because opioid use is associated with worsening of migraine over time.”
Opioids remain in widespread use for migraine, even though guidelines recommend against this treatment. Among migraineurs, opioid use is associated with more severe headache-related disability and greater use of health care resources. Opioid use also increases the risk of progressing from episodic migraine to chronic migraine.
A review of MAST data
Dr. Lipton and colleagues set out to identify the variables associated with the frequency of opioid use in people with migraine. Among the variables that they sought to examine were demographic characteristics, comorbidities, headache characteristics, medication use, and patterns of health care use. Dr. Lipton’s group hypothesized that migraine-related severity and burden would increase with increasing frequency of opioid use.
To conduct their research, the investigators examined data from the MAST study, a nationwide sample of American adults with migraine. They focused specifically on participants who reported receiving prescription acute medications. Participants eligible for this analysis reported 3 or more headache days in the previous 3 months and at least 1 monthly headache day in the previous month. In all, 15,133 participants met these criteria.
Dr. Lipton and colleagues categorized participants into four groups based on their frequency of opioid use. The groups had no opioid use, 3 or fewer monthly days of opioid use, 4 to 9 monthly days of opioid use, and 10 or more days of monthly opioid use. The last category is consistent with the International Classification of Headache Disorders-3 criteria for overuse of opioids in migraine.
At baseline, MAST participants provided information about variables such as gender, age, marital status, smoking status, education, and income. Participants also reported how many times in the previous 6 months they had visited a primary care doctor, a neurologist, a headache specialist, or a pain specialist. Dr. Lipton’s group calculated monthly headache days using the number of days during the previous 3 months affected by headache. The Migraine Disability Assessment (MIDAS) questionnaire was used to measure headache-related disability. The four-item Patient Health Questionnaire (PHQ-4) was used to screen for anxiety and depression, and the Migraine Treatment Optimization Questionnaire (mTOQ-4) evaluated participants’ treatment optimization.
Men predominated among opioid users
The investigators included 4,701 MAST participants in their analysis. The population’s mean age was 45 years, and 71.6% of participants were women. Of the entire sample, 67.5% reported no opioid use, and 32.5% reported opioid use. Of the total study population, 18.7% of patients took opioids 3 or fewer days per month, 6.5% took opioids 4 to 9 days per month, and 7.3% took opioids on 10 or more days per month.
Opioid users did not differ from nonusers on race or marital status. Men were overrepresented among all groups of opioid users, however. In addition, opioid use was more prevalent among participants with fewer than 4 years of college education (34.9%) than among participants with 4 or more years of college (30.8%). The proportion of participants with fewer than 4 years of college increased with increasing monthly opioid use. Furthermore, opioid use increased with decreasing household income. As opioid use increased, rates of employment decreased. Approximately 33% of the entire sample were obese, and the proportion of obese participants increased with increasing days per month of opioid use.
The most frequent setting during the previous 6 months for participants seeking care was primary care (49.7%). The next most frequent setting was neurology units (20.9%), pain clinics (8.3%), and headache clinics (7.7%). The prevalence of opioid use was 37.5% among participants with primary care visits, 37.3% among participants with neurologist visits, 43.0% among participants with headache clinic visits, and 53.5% with pain clinic visits.
About 15% of the population had chronic migraine. The prevalence of chronic migraine increased with increasing frequency of opioid use. About 49% of the sample had allodynia, and the prevalence of allodynia increased with increasing frequency of opioid use. Overall, disability was moderate to severe in 57.3% of participants. Participants who used opioids on 3 or fewer days per month had the lowest prevalence of moderate to severe disability (50.2%), and participants who used opioids on 10 or more days per month had the highest prevalence of moderate to severe disability (83.8%).
Approximately 21% of participants had anxiety or depression. The lowest prevalence of anxiety or depression was among participants who took opioids on 3 or fewer days per month (17.4%), and the highest prevalence was among participants who took opioids on 10 or more days per month (43.2%). About 39% of the population had very poor to poor treatment optimization. Among opioid nonusers, 35.6% had very poor to poor treatment optimization, and 59.4% of participants who used opioids on 10 or more days per month had very poor to poor treatment optimization.
Dr. Lipton and colleagues also examined the study population’s use of triptans. Overall, 51.5% of participants reported taking triptans. The prevalence of triptan use was highest among participants who did not use opioids (64.1%) and lowest among participants who used opioids on 3 or fewer days per month (20.5%). Triptan use increased as monthly days of opioid use increased.
Pain clinics and opioid prescription
“In the general population, women are more likely to receive opioids than men,” said Dr. Lipton. “This [finding] could reflect, in part, that women have more pain disorders than men and are more likely to seek medical care for pain than men.” In the current study, however, men with migraine were more likely to receive opioid prescriptions than were women with migraine. One potential explanation for this finding is that men with migraine are less likely to receive a migraine diagnosis, which might attenuate opioid prescribing, than women with migraine. “It may be that opioids are perceived to be serious drugs for serious pain, and that some physicians may be more likely to prescribe opioids to men because the disorder is taken more seriously in men than women,” said Dr. Lipton.
The observation that opioids were more likely to be prescribed for people treated in pain clinics “is consistent with my understanding of practice patterns,” he added. “Generally, neurologists strive to find effective acute treatment alternatives to opioids. The emergence of [drug classes known as] gepants and ditans provides a helpful set of alternatives to tritpans.”
Dr. Lipton and his colleagues plan further research into the treatment of migraineurs. “In a claims analysis, we showed that when people with migraine fail a triptan, they are most likely to get an opioid as their next drug,” he said. “Reasonable [clinicians] might disagree on the next step. The next step, in the absence of contraindications, could be a different oral triptan, a nonoral triptan, or a gepant or ditan. We are planning a randomized trial to probe this question.”
Why are opioids still being used?
The study’s reliance on patients’ self-report and its retrospective design are two of its weaknesses, said Alan M. Rapoport, MD, clinical professor of neurology at the University of California, Los Angeles, and editor-in-chief of Neurology Reviews. One strength, however, is that the stratified sampling methodology produced a study population that accurately reflects the demographic characteristics of the U.S. adult population, he added. Another strength is the investigators’ examination of opioid use by patient characteristics such as marital status, education, income, obesity, and smoking.
Given the harmful effects of opioids in migraine, it is hard to understand why as much as one-third of study participants using acute care medication for migraine were using opioids, said Dr. Rapoport. Using opioids for the acute treatment of migraine attacks often indicates inadequate treatment optimization, which leads to ongoing headache. As a consequence, patients may take more medication, which can increase headache frequency and lead to diagnoses of chronic migraine and medication overuse headache. Although the study found an association between the increased use of opioids and decreased household income and increased unemployment, smoking, and obesity, “it is not possible to assign causality to any of these associations, even though some would argue that decreased socioeconomic status was somehow related to more headache, disability, obesity, smoking, and unemployment,” he added.
“The paper suggests that future research should look at the risk factors for use of opioids and should determine if depression is a risk factor for or a consequence of opioid use,” said Dr. Rapoport. “Interventional studies designed to improve the acute care of migraine attacks might be able to reduce the use of opioids. I have not used opioids or butalbital-containing medication in my office for many years.”
This study was funded and sponsored by Dr. Reddy’s Laboratories group of companies, Princeton, N.J. Dr. Lipton has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as a consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Biohaven, Dr. Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva, Inc. He receives royalties from Wolff’s Headache, 8th Edition (New York: Oxford University Press, 2009) and holds stock options in eNeura Therapeutics and Biohaven.
SOURCE: Lipton RB, et al. Headache. https://doi.org/10.1111/head.14018. 2020;61(1):103-16.
FROM HEADACHE
Women increasingly turn to CBD, with or without doc’s blessing
When 42-year-old Danielle Simone Brand started having hormonal migraines, she first turned to cannabidiol (CBD) oil, eventually adding an occasional pull on a prefilled tetrahydrocannabinol (THC) vape for nighttime use. She was careful to avoid THC during work hours. A parenting and cannabis writer, Ms. Brand had more than a cursory background in cannabinoid medicine and had spent time at her local California dispensary discussing various cannabinoid components that might help alleviate her pain.
A self-professed “do-it-yourselfer,” Ms. Brand continues to use cannabinoids for her monthly headaches, forgoing any other pain medication. “There are times for conventional medicine in partnership with your doctor, but when it comes to health and wellness, women should be empowered to make decisions and self-experiment,” she said in an interview.
Ms. Brand is not alone. Significant numbers of women are replacing or supplementing prescription medications with cannabinoids, often without consulting their primary care physician, ob.gyn., or other specialist. At times, women have tried to have these conversations, only to be met with silence or worse.
Take Linda Fuller, a 58-year-old yoga instructor from Long Island who says that she uses CBD and THC for chronic sacroiliac pain after a car accident and to alleviate stress-triggered eczema flares. “I’ve had doctors turn their backs on me; I’ve had nurse practitioners walk out on me in the middle of a sentence,” she said in an interview.
Ms. Fuller said her conversion to cannabinoid medicine is relatively new; she never used cannabis recreationally before her accident but now considers it a gift. She doesn’t keep aspirin in the house and refused pain medication immediately after she injured her back.
Diana Krach, a 34-year-old writer from Maryland, says she’s encountered roadblocks about her decision to use cannabinoids for endometriosis and for pain from Crohn’s disease. When she tried to discuss her CBD use with a gastroenterologist, he interrupted her: “Whatever pot you’re smoking isn’t going to work, you’re going on biologics.”
Ms. Krach had not been smoking anything but had turned to a CBD tincture for symptom relief after prescription pain medications failed to help.
Ms. Brand, Ms. Fuller, and Ms. Krach are the tip of the iceberg when it comes to women seeking symptom relief outside the medicine cabinet. A recent survey in the Journal of Women’s Health of almost 1,000 women show that 90% (most between the ages of 35 and 44) had used cannabis and would consider using it to treat gynecologic pain. Roughly 80% said they would consider using it for procedure-related pain or other conditions. Additionally, women have reported using cannabinoids for PTSD, sleep disturbances or insomnia, anxiety, and migraine headaches.
Observational survey data have likewise shown that 80% of women with advanced or recurrent gynecologic malignancies who were prescribed cannabis reported that it was equivalent or superior to other medications for relieving pain, neuropathy, nausea, insomnia, decreased appetite, and anxiety.
In another survey, almost half (45%) of women with gynecologic malignancies who used nonprescribed cannabis for the same symptoms reported that they had reduced their use of prescription narcotics after initiating use of cannabis.
The gray zone
There has been a surge in self-reported cannabis use among pregnant women in particular. The National Survey on Drug Use and Health findings for the periods 2002-2003 and 2016-2017 highlight increases in adjusted prevalence rates from 3.4% to 7% in past-month use among pregnant women overall and from 5.7% to 12.1% during the first trimester alone.
“The more that you talk to pregnant women, the more that you realize that a lot are using cannabinoids for something that is basically medicinal, for sleep, for anxiety, or for nausea,” Katrina Mark, MD, an ob.gyn. and associate professor of medicine at the University of Maryland, College Park, said in an interview. “I’m not saying it’s fine to use drugs in pregnancy, but it is a grayer conversation than a lot of colleagues want to believe. Telling women to quit seems foolish since the alternative is to be anxious, don’t sleep, don’t eat, or use a medication that also has risks to it.”
One observational study shows that pregnant women themselves are conflicted. Although the majority believe that cannabis is “natural” and “safe,” compared with prescription drugs, they aren’t entirely in the dark about potential risks. They often express frustration with practitioners’ responses when these topics are broached during office visits. An observational survey among women and practitioners published in 2020 highlights that only half of doctors openly discouraged perinatal cannabis use and that others opted out of the discussion entirely.
This is the experience of many of the women that this news organization spoke with. Ms. Krach pointed out that “there’s a big deficit in listening; the doctor is supposed to be working for our behalf, especially when it comes to reproductive health.”
Dr. Mark believed that a lot of the conversation has been clouded by the illegality of the substance but that cannabinoids deserve as much of a fair chance for discussion and consideration as other medicines, which also carry risks in pregnancy. “There’s literally no evidence that it will work in pregnancy [for these symptoms], but there’s no evidence that it doesn’t, either,” she said in an interview. “When I have this conversation with colleagues who do not share my views, I try to encourage them to look at the actual risks versus the benefits versus the alternatives.”
The ‘entourage effect’
Data supporting cannabinoids have been mostly laboratory based, case based, or observational. However, several well-designed (albeit small) trials have demonstrated efficacy for chronic pain conditions, including neuropathic and headache pain, as well as in Crohn’s disease. Most investigators have concluded that dosage is important and that there is a synergistic interaction between compounds (known as the “entourage effect”) that relates to cannabinoid efficacy or lack thereof, as well as possible adverse effects.
In addition to legality issues, the entourage effect is one of the most important factors related to the medical use of cannabinoids. “There are literally thousands of cultivars of cannabis, each with their own phytocannabinoid and terpenic profiles that may produce distinct therapeutic effects, [so] it is misguided to speak of cannabis in monolithic terms. It is like making broad claims about soup,” wrote coauthor Samoon Ahmad, MD, in Medical Marijuana: A Clinical Handbook.
Additionally, the role that reproductive hormones play is not entirely understood. Reproductive-aged women appear to be more susceptible to a “telescoping” (gender-related progression to dependence) effect in comparison with men. Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, said in an interview. She explained that research has shown that factors such as the degree of exposure, frequency of use, and menses confound this susceptibility.
It’s the data
Frustration over cannabinoid therapeutics abound, especially when it comes to data, legal issues, and lack of training. “The feedback that I hear from providers is that there isn’t enough information; we just don’t know enough about it,” Dr. Mark said, “but there is information that we do have, and ignoring it is not beneficial.”
Dr. Cooper concurred. Although she readily acknowledges that data from randomized, placebo-controlled trials are mostly lacking, she says, “There are signals in the literature providing evidence for the utility of cannabis and cannabinoids for pain and some other effects.”
Other practitioners said in an interview that some patients admit to using cannabinoids but that they lack the ample information to guide these patients. By and large, many women equate “natural” with “safe,” and some will experiment on their own to see what works.
Those experiments are not without risk, which is why “it’s just as important for physicians to talk to their patients about cannabis use as it is for patients to be forthcoming about that use,” said Dr. Cooper. “It could have implications on their overall health as well as interactions with other drugs that they’re using.”
That balance from a clinical perspective on cannabis is crucial, wrote coauthor Kenneth Hill, MD, in Medical Marijuana: A Clinical Handbook. “Without it,” he wrote, “the window of opportunity for a patient to accept treatment that she needs may not be open very long.”
A version of this article first appeared on Medscape.com.
When 42-year-old Danielle Simone Brand started having hormonal migraines, she first turned to cannabidiol (CBD) oil, eventually adding an occasional pull on a prefilled tetrahydrocannabinol (THC) vape for nighttime use. She was careful to avoid THC during work hours. A parenting and cannabis writer, Ms. Brand had more than a cursory background in cannabinoid medicine and had spent time at her local California dispensary discussing various cannabinoid components that might help alleviate her pain.
A self-professed “do-it-yourselfer,” Ms. Brand continues to use cannabinoids for her monthly headaches, forgoing any other pain medication. “There are times for conventional medicine in partnership with your doctor, but when it comes to health and wellness, women should be empowered to make decisions and self-experiment,” she said in an interview.
Ms. Brand is not alone. Significant numbers of women are replacing or supplementing prescription medications with cannabinoids, often without consulting their primary care physician, ob.gyn., or other specialist. At times, women have tried to have these conversations, only to be met with silence or worse.
Take Linda Fuller, a 58-year-old yoga instructor from Long Island who says that she uses CBD and THC for chronic sacroiliac pain after a car accident and to alleviate stress-triggered eczema flares. “I’ve had doctors turn their backs on me; I’ve had nurse practitioners walk out on me in the middle of a sentence,” she said in an interview.
Ms. Fuller said her conversion to cannabinoid medicine is relatively new; she never used cannabis recreationally before her accident but now considers it a gift. She doesn’t keep aspirin in the house and refused pain medication immediately after she injured her back.
Diana Krach, a 34-year-old writer from Maryland, says she’s encountered roadblocks about her decision to use cannabinoids for endometriosis and for pain from Crohn’s disease. When she tried to discuss her CBD use with a gastroenterologist, he interrupted her: “Whatever pot you’re smoking isn’t going to work, you’re going on biologics.”
Ms. Krach had not been smoking anything but had turned to a CBD tincture for symptom relief after prescription pain medications failed to help.
Ms. Brand, Ms. Fuller, and Ms. Krach are the tip of the iceberg when it comes to women seeking symptom relief outside the medicine cabinet. A recent survey in the Journal of Women’s Health of almost 1,000 women show that 90% (most between the ages of 35 and 44) had used cannabis and would consider using it to treat gynecologic pain. Roughly 80% said they would consider using it for procedure-related pain or other conditions. Additionally, women have reported using cannabinoids for PTSD, sleep disturbances or insomnia, anxiety, and migraine headaches.
Observational survey data have likewise shown that 80% of women with advanced or recurrent gynecologic malignancies who were prescribed cannabis reported that it was equivalent or superior to other medications for relieving pain, neuropathy, nausea, insomnia, decreased appetite, and anxiety.
In another survey, almost half (45%) of women with gynecologic malignancies who used nonprescribed cannabis for the same symptoms reported that they had reduced their use of prescription narcotics after initiating use of cannabis.
The gray zone
There has been a surge in self-reported cannabis use among pregnant women in particular. The National Survey on Drug Use and Health findings for the periods 2002-2003 and 2016-2017 highlight increases in adjusted prevalence rates from 3.4% to 7% in past-month use among pregnant women overall and from 5.7% to 12.1% during the first trimester alone.
“The more that you talk to pregnant women, the more that you realize that a lot are using cannabinoids for something that is basically medicinal, for sleep, for anxiety, or for nausea,” Katrina Mark, MD, an ob.gyn. and associate professor of medicine at the University of Maryland, College Park, said in an interview. “I’m not saying it’s fine to use drugs in pregnancy, but it is a grayer conversation than a lot of colleagues want to believe. Telling women to quit seems foolish since the alternative is to be anxious, don’t sleep, don’t eat, or use a medication that also has risks to it.”
One observational study shows that pregnant women themselves are conflicted. Although the majority believe that cannabis is “natural” and “safe,” compared with prescription drugs, they aren’t entirely in the dark about potential risks. They often express frustration with practitioners’ responses when these topics are broached during office visits. An observational survey among women and practitioners published in 2020 highlights that only half of doctors openly discouraged perinatal cannabis use and that others opted out of the discussion entirely.
This is the experience of many of the women that this news organization spoke with. Ms. Krach pointed out that “there’s a big deficit in listening; the doctor is supposed to be working for our behalf, especially when it comes to reproductive health.”
Dr. Mark believed that a lot of the conversation has been clouded by the illegality of the substance but that cannabinoids deserve as much of a fair chance for discussion and consideration as other medicines, which also carry risks in pregnancy. “There’s literally no evidence that it will work in pregnancy [for these symptoms], but there’s no evidence that it doesn’t, either,” she said in an interview. “When I have this conversation with colleagues who do not share my views, I try to encourage them to look at the actual risks versus the benefits versus the alternatives.”
The ‘entourage effect’
Data supporting cannabinoids have been mostly laboratory based, case based, or observational. However, several well-designed (albeit small) trials have demonstrated efficacy for chronic pain conditions, including neuropathic and headache pain, as well as in Crohn’s disease. Most investigators have concluded that dosage is important and that there is a synergistic interaction between compounds (known as the “entourage effect”) that relates to cannabinoid efficacy or lack thereof, as well as possible adverse effects.
In addition to legality issues, the entourage effect is one of the most important factors related to the medical use of cannabinoids. “There are literally thousands of cultivars of cannabis, each with their own phytocannabinoid and terpenic profiles that may produce distinct therapeutic effects, [so] it is misguided to speak of cannabis in monolithic terms. It is like making broad claims about soup,” wrote coauthor Samoon Ahmad, MD, in Medical Marijuana: A Clinical Handbook.
Additionally, the role that reproductive hormones play is not entirely understood. Reproductive-aged women appear to be more susceptible to a “telescoping” (gender-related progression to dependence) effect in comparison with men. Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, said in an interview. She explained that research has shown that factors such as the degree of exposure, frequency of use, and menses confound this susceptibility.
It’s the data
Frustration over cannabinoid therapeutics abound, especially when it comes to data, legal issues, and lack of training. “The feedback that I hear from providers is that there isn’t enough information; we just don’t know enough about it,” Dr. Mark said, “but there is information that we do have, and ignoring it is not beneficial.”
Dr. Cooper concurred. Although she readily acknowledges that data from randomized, placebo-controlled trials are mostly lacking, she says, “There are signals in the literature providing evidence for the utility of cannabis and cannabinoids for pain and some other effects.”
Other practitioners said in an interview that some patients admit to using cannabinoids but that they lack the ample information to guide these patients. By and large, many women equate “natural” with “safe,” and some will experiment on their own to see what works.
Those experiments are not without risk, which is why “it’s just as important for physicians to talk to their patients about cannabis use as it is for patients to be forthcoming about that use,” said Dr. Cooper. “It could have implications on their overall health as well as interactions with other drugs that they’re using.”
That balance from a clinical perspective on cannabis is crucial, wrote coauthor Kenneth Hill, MD, in Medical Marijuana: A Clinical Handbook. “Without it,” he wrote, “the window of opportunity for a patient to accept treatment that she needs may not be open very long.”
A version of this article first appeared on Medscape.com.
When 42-year-old Danielle Simone Brand started having hormonal migraines, she first turned to cannabidiol (CBD) oil, eventually adding an occasional pull on a prefilled tetrahydrocannabinol (THC) vape for nighttime use. She was careful to avoid THC during work hours. A parenting and cannabis writer, Ms. Brand had more than a cursory background in cannabinoid medicine and had spent time at her local California dispensary discussing various cannabinoid components that might help alleviate her pain.
A self-professed “do-it-yourselfer,” Ms. Brand continues to use cannabinoids for her monthly headaches, forgoing any other pain medication. “There are times for conventional medicine in partnership with your doctor, but when it comes to health and wellness, women should be empowered to make decisions and self-experiment,” she said in an interview.
Ms. Brand is not alone. Significant numbers of women are replacing or supplementing prescription medications with cannabinoids, often without consulting their primary care physician, ob.gyn., or other specialist. At times, women have tried to have these conversations, only to be met with silence or worse.
Take Linda Fuller, a 58-year-old yoga instructor from Long Island who says that she uses CBD and THC for chronic sacroiliac pain after a car accident and to alleviate stress-triggered eczema flares. “I’ve had doctors turn their backs on me; I’ve had nurse practitioners walk out on me in the middle of a sentence,” she said in an interview.
Ms. Fuller said her conversion to cannabinoid medicine is relatively new; she never used cannabis recreationally before her accident but now considers it a gift. She doesn’t keep aspirin in the house and refused pain medication immediately after she injured her back.
Diana Krach, a 34-year-old writer from Maryland, says she’s encountered roadblocks about her decision to use cannabinoids for endometriosis and for pain from Crohn’s disease. When she tried to discuss her CBD use with a gastroenterologist, he interrupted her: “Whatever pot you’re smoking isn’t going to work, you’re going on biologics.”
Ms. Krach had not been smoking anything but had turned to a CBD tincture for symptom relief after prescription pain medications failed to help.
Ms. Brand, Ms. Fuller, and Ms. Krach are the tip of the iceberg when it comes to women seeking symptom relief outside the medicine cabinet. A recent survey in the Journal of Women’s Health of almost 1,000 women show that 90% (most between the ages of 35 and 44) had used cannabis and would consider using it to treat gynecologic pain. Roughly 80% said they would consider using it for procedure-related pain or other conditions. Additionally, women have reported using cannabinoids for PTSD, sleep disturbances or insomnia, anxiety, and migraine headaches.
Observational survey data have likewise shown that 80% of women with advanced or recurrent gynecologic malignancies who were prescribed cannabis reported that it was equivalent or superior to other medications for relieving pain, neuropathy, nausea, insomnia, decreased appetite, and anxiety.
In another survey, almost half (45%) of women with gynecologic malignancies who used nonprescribed cannabis for the same symptoms reported that they had reduced their use of prescription narcotics after initiating use of cannabis.
The gray zone
There has been a surge in self-reported cannabis use among pregnant women in particular. The National Survey on Drug Use and Health findings for the periods 2002-2003 and 2016-2017 highlight increases in adjusted prevalence rates from 3.4% to 7% in past-month use among pregnant women overall and from 5.7% to 12.1% during the first trimester alone.
“The more that you talk to pregnant women, the more that you realize that a lot are using cannabinoids for something that is basically medicinal, for sleep, for anxiety, or for nausea,” Katrina Mark, MD, an ob.gyn. and associate professor of medicine at the University of Maryland, College Park, said in an interview. “I’m not saying it’s fine to use drugs in pregnancy, but it is a grayer conversation than a lot of colleagues want to believe. Telling women to quit seems foolish since the alternative is to be anxious, don’t sleep, don’t eat, or use a medication that also has risks to it.”
One observational study shows that pregnant women themselves are conflicted. Although the majority believe that cannabis is “natural” and “safe,” compared with prescription drugs, they aren’t entirely in the dark about potential risks. They often express frustration with practitioners’ responses when these topics are broached during office visits. An observational survey among women and practitioners published in 2020 highlights that only half of doctors openly discouraged perinatal cannabis use and that others opted out of the discussion entirely.
This is the experience of many of the women that this news organization spoke with. Ms. Krach pointed out that “there’s a big deficit in listening; the doctor is supposed to be working for our behalf, especially when it comes to reproductive health.”
Dr. Mark believed that a lot of the conversation has been clouded by the illegality of the substance but that cannabinoids deserve as much of a fair chance for discussion and consideration as other medicines, which also carry risks in pregnancy. “There’s literally no evidence that it will work in pregnancy [for these symptoms], but there’s no evidence that it doesn’t, either,” she said in an interview. “When I have this conversation with colleagues who do not share my views, I try to encourage them to look at the actual risks versus the benefits versus the alternatives.”
The ‘entourage effect’
Data supporting cannabinoids have been mostly laboratory based, case based, or observational. However, several well-designed (albeit small) trials have demonstrated efficacy for chronic pain conditions, including neuropathic and headache pain, as well as in Crohn’s disease. Most investigators have concluded that dosage is important and that there is a synergistic interaction between compounds (known as the “entourage effect”) that relates to cannabinoid efficacy or lack thereof, as well as possible adverse effects.
In addition to legality issues, the entourage effect is one of the most important factors related to the medical use of cannabinoids. “There are literally thousands of cultivars of cannabis, each with their own phytocannabinoid and terpenic profiles that may produce distinct therapeutic effects, [so] it is misguided to speak of cannabis in monolithic terms. It is like making broad claims about soup,” wrote coauthor Samoon Ahmad, MD, in Medical Marijuana: A Clinical Handbook.
Additionally, the role that reproductive hormones play is not entirely understood. Reproductive-aged women appear to be more susceptible to a “telescoping” (gender-related progression to dependence) effect in comparison with men. Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, said in an interview. She explained that research has shown that factors such as the degree of exposure, frequency of use, and menses confound this susceptibility.
It’s the data
Frustration over cannabinoid therapeutics abound, especially when it comes to data, legal issues, and lack of training. “The feedback that I hear from providers is that there isn’t enough information; we just don’t know enough about it,” Dr. Mark said, “but there is information that we do have, and ignoring it is not beneficial.”
Dr. Cooper concurred. Although she readily acknowledges that data from randomized, placebo-controlled trials are mostly lacking, she says, “There are signals in the literature providing evidence for the utility of cannabis and cannabinoids for pain and some other effects.”
Other practitioners said in an interview that some patients admit to using cannabinoids but that they lack the ample information to guide these patients. By and large, many women equate “natural” with “safe,” and some will experiment on their own to see what works.
Those experiments are not without risk, which is why “it’s just as important for physicians to talk to their patients about cannabis use as it is for patients to be forthcoming about that use,” said Dr. Cooper. “It could have implications on their overall health as well as interactions with other drugs that they’re using.”
That balance from a clinical perspective on cannabis is crucial, wrote coauthor Kenneth Hill, MD, in Medical Marijuana: A Clinical Handbook. “Without it,” he wrote, “the window of opportunity for a patient to accept treatment that she needs may not be open very long.”
A version of this article first appeared on Medscape.com.
Minimizing Opioids After Joint Operation: Protocol to Decrease Postoperative Opioid Use After Primary Total Knee Arthroplasty
For decades, opioids have been a mainstay in the management of pain after total joint arthroplasty. In the past 10 years, however, opioid prescribing has come under increased scrutiny due to a rise in rates of opioid abuse, pill diversion, and opioid-related deaths.1,2 Opioids are associated with adverse effects, including nausea, vomiting, constipation, apathy, and respiratory depression, all of which influence arthroplasty outcomes and affect the patient experience. Although primary care groups account for nearly half of prescriptions written, orthopedic surgeons have the third highest per capita rate of opioid prescribing of all medical specialties.3,4 This puts orthopedic surgeons, particularly those who perform routine procedures, in an opportune but challenging position to confront this problem through novel pain management strategies.
Approximately 1 million total knee arthroplasties (TKAs) are performed in the US every year, and the US Department of Veterans Affairs (VA) health system performs about 10,000 hip and knee joint replacements.5,6 There is no standardization of opioid prescribing in the postoperative period following these procedures, and studies have reported a wide variation in prescribing habits even within a single institution for a specific surgery.7 Patients who undergo TKA are at particularly high risk of long-term opioid use if they are on continuous opioids at the time of surgery; this is problematic in a VA patient population in which at least 16% of patients are prescribed opioids in a given year.8 Furthermore, veterans are twice as likely as nonveterans to die of an accidental overdose.9 Despite these risks, opioids remain a cornerstone of postoperative pain management both within and outside of the VA.10
In 2018, to limit unnecessary prescribing of opioid pain medication, the total joint service at the VA Portland Health Care System (VAPHCS) in Oregon implemented the Minimizing Opioids after Joint Operation (MOJO) postoperative pain protocol. The goal of the protocol was to reduce opioid use following TKA. The objectives were to provide safe, appropriate analgesia while allowing early mobilization and discharge without a concomitant increase in readmissions or emergency department (ED) visits. The purpose of this retrospective chart review was to compare the efficacy of the MOJO protocol with our historical experience and report our preliminary results.
Methods
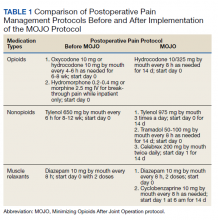
Institutional review board approval was obtained to retrospectively review the medical records of patients who had undergone TKA surgery during 2018 at VAPHCS. The MOJO protocol was composed of several simultaneous changes. The centerpiece of the new protocol was a drastic decrease in routine prescription of postoperative opioids (Table 1). Other changes included instructing patients to reduce the use of preoperative opioid pain medication 6 weeks before surgery with a goal of no opioid consumption, perform daily sets of preoperative exercises, and attend a preoperative consultation/education session with a nurse coordinator to emphasize early recovery and discharge. In patients with chronic use of opioid pain medication (particularly those for whom the medication had been prescribed for other sources of pain, such as lumbar back pain), the goal was daily opioid use of ≤ 30 morphine equivalent doses (MEDs). During the inpatient stay, we stopped prescribing prophylactic pain medication prior to physical therapy (PT).
We encouraged preoperative optimization of muscle strength by giving instructions for 4 to 8 weeks of daily exercises (Appendix). We introduced perioperative adductor canal blocks (at the discretion of the anesthesia team) and transitioned to surgery without a tourniquet. Patients in both groups received intraoperative antibiotics and IV tranexamic acid (TXA); the MOJO group also received topical TXA.
Further patient care optimization included providing patients with a team-based approach, which consisted of nurse coordinators, physician assistants and nurse practitioners, residents, and the attending surgeon. Our team reviews the planned pain management protocol, perioperative expectations, criteria for discharge, and anticipated surgical outcomes with the patient during their preoperative visits. On postoperative day 1, these members round as a team to encourage patients in their immediate postoperative recovery and rehabilitation. During rounds, the team assesses whether the patient meets the criteria for discharge, adjusting the pain management protocol if necessary.
Changes in surgical technique included arthrotomy with electrocautery, minimizing traumatic dissection or resection of the synovial tissue, and intra-articular injection of a cocktail of ropivacaine 5 mg/mL 40 mL, epinephrine 1:1,000 0.5 mL, and methylprednisolone sodium 40 mg diluted with normal saline to a total volume of 120 mL.
The new routine was gradually implemented beginning January 2017 and fully implemented by July 2018. This study compared the first 20 consecutive patients undergoing primary TKA after July 2018 to the last 20 consecutive patients undergoing primary TKA prior to January 2017. Exclusion criteria included bilateral TKA, death before 90 days, and revision as the indication for surgery. The senior attending surgeon performed all surgeries using a standard midline approach. The majority of surgeries were performed using a cemented Vanguard total knee system (Zimmer Biomet); 4 patients in the historical group had a NexGen knee system, cementless monoblock tibial components (Zimmer Biomet); and 1 patient had a Logic knee system (Exactech). Surgical selection criteria for patients did not differ between groups.
Electronic health records were reviewed and data were abstracted. The data included demographic information (age, gender, body mass index [BMI], diagnosis, and procedure), surgical factors (American Society of Anesthesiologists score, Risk Assessment and Predictive Tool score, operative time, tourniquet time, estimated blood loss), hospital factors (length of stay [LOS], discharge location), postoperative pain scores (measured on postoperative day 1 and on day of discharge), and postdischarge events (90-day complications, telephone calls reporting pain, reoperations, returns to the ED, 90-day readmissions).
The primary outcome was the mean postoperative daily MED during the inpatient stay. Secondary outcomes included pain on postoperative day 1, pain at the time of discharge, LOS, hospital readmissions, and ED visits within 90 days of surgery. Because different opioid pain medications were used by patients postoperatively, all opioids were converted to MED prior to the final analysis. Collected patient data were de-identified prior to analysis.
Power analysis was conducted to determine whether the study had sufficient population size to reject the null hypothesis for the primary outcome measure. Because practitioners controlled postoperative opioid use, a Cohen’s d of 1.0 was used so that a very large effect size was needed to reach clinical significance. Statistical significance was set to 0.05, and patient groups were set at 20 patients each. This yielded an appropriate power of 0.87. Population characteristics were compared between groups using t tests and χ2 tests as appropriate. To analyze the primary outcome, comparisons were made between the 2 cohorts using 2-tailed t tests. Secondary outcomes were compared between groups using t tests or χ2 tests. All statistics were performed using R version 3.5.2. Power analysis was conducted using the package pwr.11 Statistical significance was set at
Results
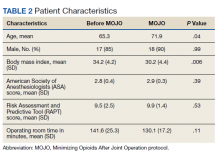
Forty patients met the inclusion criteria, evenly divided between those undergoing TKA before and after instituting the MOJO protocol (Table 2). A single patient in the MOJO group died and was excluded. A patient who underwent bilateral TKA also was excluded. Both groups reflected the male predominance of the VA patient population. MOJO patients tended to have lower BMIs (34 vs 30, P < .01). All patients indicated for surgery with preoperative opioid use were able to titrate down to their preoperative goal as verified by prescriptions filled at VA pharmacies. Twelve of the patients in the MOJO group received adductor canal blocks.
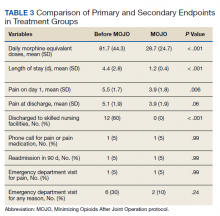
Results of t tests and χ2 tests comparing primary and secondary endpoints are listed in Table 3. Differences between the daily MEDs given in the historical and MOJO groups are shown. There were significant differences between the pre-MOJO and MOJO groups with regard to daily inpatient MEDs (82 mg vs 29 mg, P < .01) and total inpatient MEDs (306 mg vs 32 mg, P < .01). There was less self-reported pain on postoperative day 1 in the MOJO group (5.5 vs 3.9, P < .01), decreased LOS (4.4 days vs 1.2 days, P < .01), a trend toward fewer total ED visits (6 vs 2, P = .24), and fewer discharges to skilled nursing facilities (12 vs 0, P < .01). There were no blood transfusions in either group.
There were no readmissions due to uncontrolled pain. There was 1 readmission for shortness of breath in the MOJO group. The patient was discharged home the following day after ruling out thromboembolic and cardiovascular events. One patient from the control group was readmitted after missing a step on a staircase and falling. The patient sustained a quadriceps tendon rupture and underwent primary suture repair.
Discussion
Our results demonstrate that a multimodal approach to significantly reduce postoperative opioid use in patients with TKA is possible without increasing readmissions or ED visits for pain control. The patients in the MOJO group had a faster recovery, earlier discharge, and less use of postoperative opioid medication. Our approach to postoperative pain management was divided into 2 main categories: patient optimization and surgical optimization.
Patient Selection
Besides the standard evaluation and optimization of patients’ medical conditions, identifying and optimizing at-risk patients before surgery was a critical component of our protocol. Managing postoperative pain in patients with prior opioid use is an intractable challenge in orthopedic surgery. Patients with a history of chronic pain and preoperative use of opioid medications remain at higher risk of postoperative chronic pain and persistent use of opioid medication despite no obvious surgical complications.8 In a sample of > 6,000 veterans who underwent TKA at VA hospitals in 2014, 57% of the patients with daily use of opioids in the 90 days before surgery remained on opioids 1 year after surgery (vs 2 % in patients not on long-term opioids).8 This relationship between pre- and postoperative opioid use also was dose dependent.12
Furthermore, those with high preoperative use may experience worse outcomes relative to the opioid naive population as measured by arthritis-specific pain indices.13 In a well-powered retrospective study of patients who underwent elective orthopedic procedures, preoperative opioid abuse or dependence (determined by the International Classification of Diseases, Ninth Revision diagnosis) increased inpatient mortality, aggregate morbidity, surgical site infection, myocardial infarction, and LOS.14 Preoperative opioid use also has been associated with increased risk of ED visits, readmission, infection, stiffness, and aseptic revision.15 In patients with TKA in the VA specifically, preoperative opioid use (> 3 months in the prior year) was associated with increased revision rates that were even higher than those for patients with diabetes mellitus.16
Patient Education
Based on this evidence, we instruct patients to reduce their preoperative opioid dosing to zero (for patients with joint pain) or < 30 MED (for patients using opioids for other reasons). Although preoperative reduction of opioid use has been shown to improve outcomes after TKA, pain subspecialty recommendations for patients with chronic opioid use recommend considering adjunctive therapies, including transcutaneous electrical nerve stimulation, cognitive behavioral therapy, gabapentin, or ketamine.17,18 Through patient education our team has been successful in decreasing preoperative opioid use without adding other drugs or modalities.
Patient Optimization
Preoperative patient optimization included 4 to 8 weeks of daily sets of physical activity instructions (prehab) to improve the musculoskeletal function. These instructions are given to patients 4 to 8 weeks before surgery and aim to improve the patient’s balance, mobility, and functional ability (Appendix). Meta-analysis has shown that patients who undergo preoperative PT have a small but statistically significant decrease in postoperative pain at 4 weeks, though this does not persist beyond that period.19
We did note a lower BMI in patients in the MOJO group. Though this has the potential to be a confounder, a study of BMI in > 4,000 patients who underwent joint replacement surgery has shown that BMI is not associated with differences in postoperative pain.20
Surgeon and Surgical-Related Variables
Patients in the MOJO group had increased use of adductor canal blocks. A 2017 meta-analysis of 12,530 patients comparing analgesic modalities found that peripheral nerve blocks targeting multiple nerves (eg, femoral/sciatic) decreased pain at rest, decreased opioid consumption, and improved range of motion postoperatively.21 Also, these were found to be superior to single nerve blocks, periarticular infiltration, and epidural blocks.21 However, major nerve and epidural blocks affecting the lower extremity may increase the risk of falls and prolong LOS.22,23 The preferred peripheral block at VAPHCS is a single shot ultrasound-guided adductor canal block before the induction of general or spinal anesthesia. A randomized controlled trial has demonstrated superiority of this block to the femoral nerve block with regard to postoperative quadriceps strength, conferring the theoretical advantage of decreased fall risk and ability to participate in immediate PT.24 Although we are unable to confirm an association between anesthetic modalities and opioid burden, our clinical impression is that blocks were effective at reducing immediate postoperative pain. However, among MOJO patients there were no differences in patients with and without blocks for either pain (4.2 vs 3.8, P = .69) or opioid consumption (28.8 vs 33.0, P = .72) after surgery, though our study was not powered to detect a difference in this restricted subgroup.
Patients who frequently had reported postoperative thigh pain prompted us to make changes in our surgical technique, performing TKA without use of a tourniquet. Tourniquet use has been associated with an increased risk of thigh pain after TKA by multiple authors.25,26 Postoperative thigh pain also is pressure dependent.27 In addition, its use may be associated with a slightly increased risk of thromboembolic events and delayed functional recovery.28,29
Because postoperative hemarthrosis is associated with more pain and reduced joint recovery function, we used topical TXA to reduce postoperative surgical site and joint hematoma. TXA (either oral, IV, or topical) during TKA is used to control postoperative bleeding primarily and decrease the need for transfusion without concomitant increase in thromboembolic events.30,31 Topical TXA may be more effective than IV, particularly in the immediate postoperative period.32 Although pain typically is not an endpoint in studies of TXA, a prospective study of 48 patients showed evidence that its use may be associated with decreased postoperative pain in the first 24 hours after surgery (though not after).33 Finally, the use of intra-articular injection has evolved in our clinical practice, but literature is lacking with regard to its efficacy; more studies are needed to determine its effect relative to no injection. We have not seen any benefits to using
Limitations
This is a nonrandomized retrospective single-institution study. Our study population is composed of mostly males with military experience and is not necessarily a representative sample of the general population eligible for joint arthroplasty. Our primary endpoint (reduction of opioid use postoperatively) also was a cornerstone of our intervention. To account for this, we set a very large effect size in our power analysis and evaluated multiple secondary endpoints to determine whether postoperative pain remained well controlled and complications/readmission minimized with our interventions. Because our intervention was multimodal, our study cannot make conclusions about the effect of a particular component of our treatment strategy. We did not measure or compare functional outcomes between both groups, which offers an opportunity for further research.
These limitations are balanced by several strengths. Our cohort was well controlled with respect to the dose and type of drug used. There is staff dedicated to postoperative telephone follow-up after discharge, and veterans are apt to seek care within the VA health care system, which improves case finding for complications and ED visits. No patients were lost to follow-up. Moreover, our drastic reduction in opioid use is promising enough to warrant reporting, while the broader orthopedic literature explores the relative impact of each variable.
Conclusions
The MOJO protocol has been effective for reducing postoperative opioid use after TKA without compromising effective pain management. The drastic reduction in the postoperative use of opioid pain medications and LOS have contributed to a cultural shift within our department, comprehensive team approach, multimodal pain management, and preoperative patient optimization. Further investigations are required to assess the impact of each intervention on observed outcomes. However, the framework and routines are applicable to other institutions and surgical specialties.
Acknowledgments
The authors recognize Derek Bond, MD, for his help in creating the MOJO acronym.
1. Hedegaard H, Miniño AM, Warner M. Drug overdose deaths in the United States, 1999-2017. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics Data Brief No. 329. Published November 2018. Accessed January 12, 2021. https://www.cdc.gov/nchs/data/databriefs/db329-h.pdf
2. Hedegaard H, Warner M, Miniño AM. Drug overdose deaths in the United States, 1999-2016. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics NCHS data brief No. 294. Published December 2017. Accessed January 12, 2021. https://www.cdc.gov/nchs/data/databriefs/db294.pdf
3. Levy B, Paulozzi L, Mack KA, Jones CM. Trends in opioid analgesic–prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413. doi:10.1016/j.amepre.2015.02.020
4. Guy GP, Zhang K. Opioid prescribing by specialty and volume in the U.S. Am J Prev Med. 2018;55(5):e153-155. doi:10.1016/j.amepre.2018.06.008
5. Kremers HM, Larson DR, Crowson CS, et al. Prevalence of total hip and knee replacement in the United States. J Bone Joint Surgery Am. 2015;17:1386-1397. doi:10.2106/JBJS.N.01141
6. Giori NJ, Amanatullah DF, Gupta S, Bowe T, Harris AHS. Risk reduction compared with access to care: quantifying the trade-off of enforcing a body mass index eligibility criterion for joint replacement. J Bone Joint Surg Am. 2018; 4(100):539-545. doi:10.2106/JBJS.17.00120
7. Sabatino MJ, Kunkel ST, Ramkumar DB, Keeney BJ, Jevsevar DS. Excess opioid medication and variation in prescribing patterns following common orthopaedic procedures. J Bone Joint Surg Am. 2018;100(3):180-188. doi:10.2106/JBJS.17.00672
8. Hadlandsmyth K, Vander Weg MW, McCoy KD, Mosher HJ, Vaughan-Sarrazin MS, Lund BC. Risk for prolonged opioid use following total knee arthroplasty in veterans. J Arthroplasty. 2018;33(1):119-123. doi:10.1016/j.arth.2017.08.022
9. Bohnert ASB, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305(13):1315-1321. doi:10.1001/jama.2011.370
10. Hall MJ, Schwartzman A, Zhang J, Liu X. Ambulatory surgery data from hospitals and ambulatory surgery centers: United States, 2010. Natl Health Stat Report. 2017(102):1-15.
11. Champely S. pwr: basic functions for power analysis. R package version 1.2-2; 2018. Accessed January 13, 2021. https://rdrr.io/cran/pwr/
12. Goesling J, Moser SE, Zaidi B, et al. Trends and predictors of opioid use after total knee and total hip arthroplasty. Pain. 2016;157(6):1259-1265. doi:10.1097/j.pain.0000000000000516
13. Smith SR, Bido J, Collins JE, Yang H, Katz JN, Losina E. Impact of preoperative opioid use on total knee arthroplasty outcomes. J Bone Joint Surg Am. 2017;99(10):803-808. doi:10.2106/JBJS.16.01200
14. Menendez ME, Ring D, Bateman BT. Preoperative opioid misuse is associated with increased morbidity and mortality after elective orthopaedic surgery. Clin Orthop Relat Res. 2015;473(7):2402-412. doi:10.1007/s11999-015-4173-5
15. Cancienne JM, Patel KJ, Browne JA, Werner BC. Narcotic use and total knee arthroplasty. J Arthroplasty. 2018;33(1):113-118. doi:10.1016/j.arth.2017.08.006
16. Ben-Ari A, Chansky H, Rozet I. Preoperative opioid use is associated with early revision after total knee arthroplasty: a study of male patients treated in the Veterans Affairs System. J Bone Joint Surg Am. 2017;99(1):1-9. doi:10.2106/JBJS.16.00167
17. Nguyen L-CL, Sing DC, Bozic KJ. Preoperative reduction of opioid use before total joint arthroplasty. J Arthroplasty. 2016;31(suppl 9):282-287. doi:10.1016/j.arth.2016.01.068
18. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-157. doi:10.1016/j.jpain.2015.12.008
19. Wang L, Lee M, Zhang Z, Moodie J, Cheng D, Martin J. Does preoperative rehabilitation for patients planning to undergo joint replacement surgery improve outcomes? A systematic review and meta-analysis of randomised controlled trials. BMJ Open. 2016;6(2):e009857. doi:10.1136/bmjopen-2015-009857
20. Li W, Ayers DC, Lewis CG, Bowen TR, Allison JJ, Franklin PD. Functional gain and pain relief after total joint replacement according to obesity status. J Bone Joint Surg. 2017;99(14):1183-1189. doi:10.2106/JBJS.16.00960
21. Terkawi AS, Mavridis D, Sessler DI, et al. Pain management modalities after total knee arthroplasty: a network meta-analysis of 170 randomized controlled trials. Anesthesiology. 2017;126(5):923-937. doi:10.1097/ALN.0000000000001607
22. Ilfeld BM, Duke KB, Donohue MC. The association between lower extremity continuous peripheral nerve blocks and patient falls after knee and hip arthroplasty. Anesth Analg. 2010;111(6):1552-1554. doi:10.1213/ANE.0b013e3181fb9507
23. Elkassabany NM, Antosh S, Ahmed M, et al. The risk of falls after total knee arthroplasty with the use of a femoral nerve block versus an adductor canal block. Anest Analg. 2016;122(5):1696-1703. doi:10.1213/ane.0000000000001237
24. Wang D, Yang Y, Li Q, et al. Adductor canal block versus femoral nerve block for total knee arthroplasty: a meta-analysis of randomized controlled trials. Sci Rep. 2017;7:40721. doi:10.1038/srep40721
25. Liu D, Graham D, Gillies K, Gillies RM. Effects of tourniquet use on quadriceps function and pain in total knee arthroplasty. Knee Surg Relat Res. 2014;26(4):207-213. doi:10.5792/ksrr.2014.26.4.207
26. Abdel-Salam A, Eyres KS. Effects of tourniquet during total knee arthroplasty. A prospective randomised study. J Bone Joint Surg Br. 1995;77(2):250-253.
27. Worland RL, Arredondo J, Angles F, Lopez-Jimenez F, Jessup DE. Thigh pain following tourniquet application in simultaneous bilateral total knee replacement arthroplasty. J Arthroplasty. 1997;12(8):848-852. doi:10.1016/s0883-5403(97)90153-4
28. Tai T-W, Lin C-J, Jou I-M, Chang C-W, Lai K-A, Yang C-Y. Tourniquet use in total knee arthroplasty: a meta-analysis. Knee Surg Sports Traumatol, Arthrosc. 2011;19(7):1121-1130. doi:10.1007/s00167-010-1342-7
29. Jiang F-Z, Zhong H-M, Hong Y-C, Zhao G-F. Use of a tourniquet in total knee arthroplasty: a systematic review and meta-analysis of randomized controlled trials. J Orthop Sci. 2015;20(21):110-123. doi:10.1007/s00776-014-0664-6
30. Alshryda S, Sarda P, Sukeik M, Nargol A, Blenkinsopp J, Mason JM. Tranexamic acid in total knee replacement: a systematic review and meta-analysis. J Bone Joint Surg Br. 2011;93(12):1577-1585. doi:10.1302/0301-620X.93B12.26989
31. Panteli M, Papakostidis C, Dahabreh Z, Giannoudis PV. Topical tranexamic acid in total knee replacement: a systematic review and meta-analysis. Knee. 2013;20(5):300-309. doi:10.1016/j.knee.2013.05.014
32. Wang J, Wang Q, Zhang X, Wang Q. Intra-articular application is more effective than intravenous application of tranexamic acid in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2017;32(11):3385-3389. doi:10.1016/j.arth.2017.06.024
33. Guerreiro JPF, Badaro BS, Balbino JRM, Danieli MV, Queiroz AO, Cataneo DC. Application of tranexamic acid in total knee arthroplasty – prospective randomized trial. J Open Orthop J. 2017;11:1049-1057. doi:10.2174/1874325001711011049
For decades, opioids have been a mainstay in the management of pain after total joint arthroplasty. In the past 10 years, however, opioid prescribing has come under increased scrutiny due to a rise in rates of opioid abuse, pill diversion, and opioid-related deaths.1,2 Opioids are associated with adverse effects, including nausea, vomiting, constipation, apathy, and respiratory depression, all of which influence arthroplasty outcomes and affect the patient experience. Although primary care groups account for nearly half of prescriptions written, orthopedic surgeons have the third highest per capita rate of opioid prescribing of all medical specialties.3,4 This puts orthopedic surgeons, particularly those who perform routine procedures, in an opportune but challenging position to confront this problem through novel pain management strategies.
Approximately 1 million total knee arthroplasties (TKAs) are performed in the US every year, and the US Department of Veterans Affairs (VA) health system performs about 10,000 hip and knee joint replacements.5,6 There is no standardization of opioid prescribing in the postoperative period following these procedures, and studies have reported a wide variation in prescribing habits even within a single institution for a specific surgery.7 Patients who undergo TKA are at particularly high risk of long-term opioid use if they are on continuous opioids at the time of surgery; this is problematic in a VA patient population in which at least 16% of patients are prescribed opioids in a given year.8 Furthermore, veterans are twice as likely as nonveterans to die of an accidental overdose.9 Despite these risks, opioids remain a cornerstone of postoperative pain management both within and outside of the VA.10
In 2018, to limit unnecessary prescribing of opioid pain medication, the total joint service at the VA Portland Health Care System (VAPHCS) in Oregon implemented the Minimizing Opioids after Joint Operation (MOJO) postoperative pain protocol. The goal of the protocol was to reduce opioid use following TKA. The objectives were to provide safe, appropriate analgesia while allowing early mobilization and discharge without a concomitant increase in readmissions or emergency department (ED) visits. The purpose of this retrospective chart review was to compare the efficacy of the MOJO protocol with our historical experience and report our preliminary results.
Methods
Institutional review board approval was obtained to retrospectively review the medical records of patients who had undergone TKA surgery during 2018 at VAPHCS. The MOJO protocol was composed of several simultaneous changes. The centerpiece of the new protocol was a drastic decrease in routine prescription of postoperative opioids (Table 1). Other changes included instructing patients to reduce the use of preoperative opioid pain medication 6 weeks before surgery with a goal of no opioid consumption, perform daily sets of preoperative exercises, and attend a preoperative consultation/education session with a nurse coordinator to emphasize early recovery and discharge. In patients with chronic use of opioid pain medication (particularly those for whom the medication had been prescribed for other sources of pain, such as lumbar back pain), the goal was daily opioid use of ≤ 30 morphine equivalent doses (MEDs). During the inpatient stay, we stopped prescribing prophylactic pain medication prior to physical therapy (PT).
We encouraged preoperative optimization of muscle strength by giving instructions for 4 to 8 weeks of daily exercises (Appendix). We introduced perioperative adductor canal blocks (at the discretion of the anesthesia team) and transitioned to surgery without a tourniquet. Patients in both groups received intraoperative antibiotics and IV tranexamic acid (TXA); the MOJO group also received topical TXA.
Further patient care optimization included providing patients with a team-based approach, which consisted of nurse coordinators, physician assistants and nurse practitioners, residents, and the attending surgeon. Our team reviews the planned pain management protocol, perioperative expectations, criteria for discharge, and anticipated surgical outcomes with the patient during their preoperative visits. On postoperative day 1, these members round as a team to encourage patients in their immediate postoperative recovery and rehabilitation. During rounds, the team assesses whether the patient meets the criteria for discharge, adjusting the pain management protocol if necessary.
Changes in surgical technique included arthrotomy with electrocautery, minimizing traumatic dissection or resection of the synovial tissue, and intra-articular injection of a cocktail of ropivacaine 5 mg/mL 40 mL, epinephrine 1:1,000 0.5 mL, and methylprednisolone sodium 40 mg diluted with normal saline to a total volume of 120 mL.
The new routine was gradually implemented beginning January 2017 and fully implemented by July 2018. This study compared the first 20 consecutive patients undergoing primary TKA after July 2018 to the last 20 consecutive patients undergoing primary TKA prior to January 2017. Exclusion criteria included bilateral TKA, death before 90 days, and revision as the indication for surgery. The senior attending surgeon performed all surgeries using a standard midline approach. The majority of surgeries were performed using a cemented Vanguard total knee system (Zimmer Biomet); 4 patients in the historical group had a NexGen knee system, cementless monoblock tibial components (Zimmer Biomet); and 1 patient had a Logic knee system (Exactech). Surgical selection criteria for patients did not differ between groups.
Electronic health records were reviewed and data were abstracted. The data included demographic information (age, gender, body mass index [BMI], diagnosis, and procedure), surgical factors (American Society of Anesthesiologists score, Risk Assessment and Predictive Tool score, operative time, tourniquet time, estimated blood loss), hospital factors (length of stay [LOS], discharge location), postoperative pain scores (measured on postoperative day 1 and on day of discharge), and postdischarge events (90-day complications, telephone calls reporting pain, reoperations, returns to the ED, 90-day readmissions).
The primary outcome was the mean postoperative daily MED during the inpatient stay. Secondary outcomes included pain on postoperative day 1, pain at the time of discharge, LOS, hospital readmissions, and ED visits within 90 days of surgery. Because different opioid pain medications were used by patients postoperatively, all opioids were converted to MED prior to the final analysis. Collected patient data were de-identified prior to analysis.
Power analysis was conducted to determine whether the study had sufficient population size to reject the null hypothesis for the primary outcome measure. Because practitioners controlled postoperative opioid use, a Cohen’s d of 1.0 was used so that a very large effect size was needed to reach clinical significance. Statistical significance was set to 0.05, and patient groups were set at 20 patients each. This yielded an appropriate power of 0.87. Population characteristics were compared between groups using t tests and χ2 tests as appropriate. To analyze the primary outcome, comparisons were made between the 2 cohorts using 2-tailed t tests. Secondary outcomes were compared between groups using t tests or χ2 tests. All statistics were performed using R version 3.5.2. Power analysis was conducted using the package pwr.11 Statistical significance was set at
Results
Forty patients met the inclusion criteria, evenly divided between those undergoing TKA before and after instituting the MOJO protocol (Table 2). A single patient in the MOJO group died and was excluded. A patient who underwent bilateral TKA also was excluded. Both groups reflected the male predominance of the VA patient population. MOJO patients tended to have lower BMIs (34 vs 30, P < .01). All patients indicated for surgery with preoperative opioid use were able to titrate down to their preoperative goal as verified by prescriptions filled at VA pharmacies. Twelve of the patients in the MOJO group received adductor canal blocks.
Results of t tests and χ2 tests comparing primary and secondary endpoints are listed in Table 3. Differences between the daily MEDs given in the historical and MOJO groups are shown. There were significant differences between the pre-MOJO and MOJO groups with regard to daily inpatient MEDs (82 mg vs 29 mg, P < .01) and total inpatient MEDs (306 mg vs 32 mg, P < .01). There was less self-reported pain on postoperative day 1 in the MOJO group (5.5 vs 3.9, P < .01), decreased LOS (4.4 days vs 1.2 days, P < .01), a trend toward fewer total ED visits (6 vs 2, P = .24), and fewer discharges to skilled nursing facilities (12 vs 0, P < .01). There were no blood transfusions in either group.
There were no readmissions due to uncontrolled pain. There was 1 readmission for shortness of breath in the MOJO group. The patient was discharged home the following day after ruling out thromboembolic and cardiovascular events. One patient from the control group was readmitted after missing a step on a staircase and falling. The patient sustained a quadriceps tendon rupture and underwent primary suture repair.
Discussion
Our results demonstrate that a multimodal approach to significantly reduce postoperative opioid use in patients with TKA is possible without increasing readmissions or ED visits for pain control. The patients in the MOJO group had a faster recovery, earlier discharge, and less use of postoperative opioid medication. Our approach to postoperative pain management was divided into 2 main categories: patient optimization and surgical optimization.
Patient Selection
Besides the standard evaluation and optimization of patients’ medical conditions, identifying and optimizing at-risk patients before surgery was a critical component of our protocol. Managing postoperative pain in patients with prior opioid use is an intractable challenge in orthopedic surgery. Patients with a history of chronic pain and preoperative use of opioid medications remain at higher risk of postoperative chronic pain and persistent use of opioid medication despite no obvious surgical complications.8 In a sample of > 6,000 veterans who underwent TKA at VA hospitals in 2014, 57% of the patients with daily use of opioids in the 90 days before surgery remained on opioids 1 year after surgery (vs 2 % in patients not on long-term opioids).8 This relationship between pre- and postoperative opioid use also was dose dependent.12
Furthermore, those with high preoperative use may experience worse outcomes relative to the opioid naive population as measured by arthritis-specific pain indices.13 In a well-powered retrospective study of patients who underwent elective orthopedic procedures, preoperative opioid abuse or dependence (determined by the International Classification of Diseases, Ninth Revision diagnosis) increased inpatient mortality, aggregate morbidity, surgical site infection, myocardial infarction, and LOS.14 Preoperative opioid use also has been associated with increased risk of ED visits, readmission, infection, stiffness, and aseptic revision.15 In patients with TKA in the VA specifically, preoperative opioid use (> 3 months in the prior year) was associated with increased revision rates that were even higher than those for patients with diabetes mellitus.16
Patient Education
Based on this evidence, we instruct patients to reduce their preoperative opioid dosing to zero (for patients with joint pain) or < 30 MED (for patients using opioids for other reasons). Although preoperative reduction of opioid use has been shown to improve outcomes after TKA, pain subspecialty recommendations for patients with chronic opioid use recommend considering adjunctive therapies, including transcutaneous electrical nerve stimulation, cognitive behavioral therapy, gabapentin, or ketamine.17,18 Through patient education our team has been successful in decreasing preoperative opioid use without adding other drugs or modalities.
Patient Optimization
Preoperative patient optimization included 4 to 8 weeks of daily sets of physical activity instructions (prehab) to improve the musculoskeletal function. These instructions are given to patients 4 to 8 weeks before surgery and aim to improve the patient’s balance, mobility, and functional ability (Appendix). Meta-analysis has shown that patients who undergo preoperative PT have a small but statistically significant decrease in postoperative pain at 4 weeks, though this does not persist beyond that period.19
We did note a lower BMI in patients in the MOJO group. Though this has the potential to be a confounder, a study of BMI in > 4,000 patients who underwent joint replacement surgery has shown that BMI is not associated with differences in postoperative pain.20
Surgeon and Surgical-Related Variables
Patients in the MOJO group had increased use of adductor canal blocks. A 2017 meta-analysis of 12,530 patients comparing analgesic modalities found that peripheral nerve blocks targeting multiple nerves (eg, femoral/sciatic) decreased pain at rest, decreased opioid consumption, and improved range of motion postoperatively.21 Also, these were found to be superior to single nerve blocks, periarticular infiltration, and epidural blocks.21 However, major nerve and epidural blocks affecting the lower extremity may increase the risk of falls and prolong LOS.22,23 The preferred peripheral block at VAPHCS is a single shot ultrasound-guided adductor canal block before the induction of general or spinal anesthesia. A randomized controlled trial has demonstrated superiority of this block to the femoral nerve block with regard to postoperative quadriceps strength, conferring the theoretical advantage of decreased fall risk and ability to participate in immediate PT.24 Although we are unable to confirm an association between anesthetic modalities and opioid burden, our clinical impression is that blocks were effective at reducing immediate postoperative pain. However, among MOJO patients there were no differences in patients with and without blocks for either pain (4.2 vs 3.8, P = .69) or opioid consumption (28.8 vs 33.0, P = .72) after surgery, though our study was not powered to detect a difference in this restricted subgroup.
Patients who frequently had reported postoperative thigh pain prompted us to make changes in our surgical technique, performing TKA without use of a tourniquet. Tourniquet use has been associated with an increased risk of thigh pain after TKA by multiple authors.25,26 Postoperative thigh pain also is pressure dependent.27 In addition, its use may be associated with a slightly increased risk of thromboembolic events and delayed functional recovery.28,29
Because postoperative hemarthrosis is associated with more pain and reduced joint recovery function, we used topical TXA to reduce postoperative surgical site and joint hematoma. TXA (either oral, IV, or topical) during TKA is used to control postoperative bleeding primarily and decrease the need for transfusion without concomitant increase in thromboembolic events.30,31 Topical TXA may be more effective than IV, particularly in the immediate postoperative period.32 Although pain typically is not an endpoint in studies of TXA, a prospective study of 48 patients showed evidence that its use may be associated with decreased postoperative pain in the first 24 hours after surgery (though not after).33 Finally, the use of intra-articular injection has evolved in our clinical practice, but literature is lacking with regard to its efficacy; more studies are needed to determine its effect relative to no injection. We have not seen any benefits to using
Limitations
This is a nonrandomized retrospective single-institution study. Our study population is composed of mostly males with military experience and is not necessarily a representative sample of the general population eligible for joint arthroplasty. Our primary endpoint (reduction of opioid use postoperatively) also was a cornerstone of our intervention. To account for this, we set a very large effect size in our power analysis and evaluated multiple secondary endpoints to determine whether postoperative pain remained well controlled and complications/readmission minimized with our interventions. Because our intervention was multimodal, our study cannot make conclusions about the effect of a particular component of our treatment strategy. We did not measure or compare functional outcomes between both groups, which offers an opportunity for further research.
These limitations are balanced by several strengths. Our cohort was well controlled with respect to the dose and type of drug used. There is staff dedicated to postoperative telephone follow-up after discharge, and veterans are apt to seek care within the VA health care system, which improves case finding for complications and ED visits. No patients were lost to follow-up. Moreover, our drastic reduction in opioid use is promising enough to warrant reporting, while the broader orthopedic literature explores the relative impact of each variable.
Conclusions
The MOJO protocol has been effective for reducing postoperative opioid use after TKA without compromising effective pain management. The drastic reduction in the postoperative use of opioid pain medications and LOS have contributed to a cultural shift within our department, comprehensive team approach, multimodal pain management, and preoperative patient optimization. Further investigations are required to assess the impact of each intervention on observed outcomes. However, the framework and routines are applicable to other institutions and surgical specialties.
Acknowledgments
The authors recognize Derek Bond, MD, for his help in creating the MOJO acronym.
For decades, opioids have been a mainstay in the management of pain after total joint arthroplasty. In the past 10 years, however, opioid prescribing has come under increased scrutiny due to a rise in rates of opioid abuse, pill diversion, and opioid-related deaths.1,2 Opioids are associated with adverse effects, including nausea, vomiting, constipation, apathy, and respiratory depression, all of which influence arthroplasty outcomes and affect the patient experience. Although primary care groups account for nearly half of prescriptions written, orthopedic surgeons have the third highest per capita rate of opioid prescribing of all medical specialties.3,4 This puts orthopedic surgeons, particularly those who perform routine procedures, in an opportune but challenging position to confront this problem through novel pain management strategies.
Approximately 1 million total knee arthroplasties (TKAs) are performed in the US every year, and the US Department of Veterans Affairs (VA) health system performs about 10,000 hip and knee joint replacements.5,6 There is no standardization of opioid prescribing in the postoperative period following these procedures, and studies have reported a wide variation in prescribing habits even within a single institution for a specific surgery.7 Patients who undergo TKA are at particularly high risk of long-term opioid use if they are on continuous opioids at the time of surgery; this is problematic in a VA patient population in which at least 16% of patients are prescribed opioids in a given year.8 Furthermore, veterans are twice as likely as nonveterans to die of an accidental overdose.9 Despite these risks, opioids remain a cornerstone of postoperative pain management both within and outside of the VA.10
In 2018, to limit unnecessary prescribing of opioid pain medication, the total joint service at the VA Portland Health Care System (VAPHCS) in Oregon implemented the Minimizing Opioids after Joint Operation (MOJO) postoperative pain protocol. The goal of the protocol was to reduce opioid use following TKA. The objectives were to provide safe, appropriate analgesia while allowing early mobilization and discharge without a concomitant increase in readmissions or emergency department (ED) visits. The purpose of this retrospective chart review was to compare the efficacy of the MOJO protocol with our historical experience and report our preliminary results.
Methods
Institutional review board approval was obtained to retrospectively review the medical records of patients who had undergone TKA surgery during 2018 at VAPHCS. The MOJO protocol was composed of several simultaneous changes. The centerpiece of the new protocol was a drastic decrease in routine prescription of postoperative opioids (Table 1). Other changes included instructing patients to reduce the use of preoperative opioid pain medication 6 weeks before surgery with a goal of no opioid consumption, perform daily sets of preoperative exercises, and attend a preoperative consultation/education session with a nurse coordinator to emphasize early recovery and discharge. In patients with chronic use of opioid pain medication (particularly those for whom the medication had been prescribed for other sources of pain, such as lumbar back pain), the goal was daily opioid use of ≤ 30 morphine equivalent doses (MEDs). During the inpatient stay, we stopped prescribing prophylactic pain medication prior to physical therapy (PT).
We encouraged preoperative optimization of muscle strength by giving instructions for 4 to 8 weeks of daily exercises (Appendix). We introduced perioperative adductor canal blocks (at the discretion of the anesthesia team) and transitioned to surgery without a tourniquet. Patients in both groups received intraoperative antibiotics and IV tranexamic acid (TXA); the MOJO group also received topical TXA.
Further patient care optimization included providing patients with a team-based approach, which consisted of nurse coordinators, physician assistants and nurse practitioners, residents, and the attending surgeon. Our team reviews the planned pain management protocol, perioperative expectations, criteria for discharge, and anticipated surgical outcomes with the patient during their preoperative visits. On postoperative day 1, these members round as a team to encourage patients in their immediate postoperative recovery and rehabilitation. During rounds, the team assesses whether the patient meets the criteria for discharge, adjusting the pain management protocol if necessary.
Changes in surgical technique included arthrotomy with electrocautery, minimizing traumatic dissection or resection of the synovial tissue, and intra-articular injection of a cocktail of ropivacaine 5 mg/mL 40 mL, epinephrine 1:1,000 0.5 mL, and methylprednisolone sodium 40 mg diluted with normal saline to a total volume of 120 mL.
The new routine was gradually implemented beginning January 2017 and fully implemented by July 2018. This study compared the first 20 consecutive patients undergoing primary TKA after July 2018 to the last 20 consecutive patients undergoing primary TKA prior to January 2017. Exclusion criteria included bilateral TKA, death before 90 days, and revision as the indication for surgery. The senior attending surgeon performed all surgeries using a standard midline approach. The majority of surgeries were performed using a cemented Vanguard total knee system (Zimmer Biomet); 4 patients in the historical group had a NexGen knee system, cementless monoblock tibial components (Zimmer Biomet); and 1 patient had a Logic knee system (Exactech). Surgical selection criteria for patients did not differ between groups.
Electronic health records were reviewed and data were abstracted. The data included demographic information (age, gender, body mass index [BMI], diagnosis, and procedure), surgical factors (American Society of Anesthesiologists score, Risk Assessment and Predictive Tool score, operative time, tourniquet time, estimated blood loss), hospital factors (length of stay [LOS], discharge location), postoperative pain scores (measured on postoperative day 1 and on day of discharge), and postdischarge events (90-day complications, telephone calls reporting pain, reoperations, returns to the ED, 90-day readmissions).
The primary outcome was the mean postoperative daily MED during the inpatient stay. Secondary outcomes included pain on postoperative day 1, pain at the time of discharge, LOS, hospital readmissions, and ED visits within 90 days of surgery. Because different opioid pain medications were used by patients postoperatively, all opioids were converted to MED prior to the final analysis. Collected patient data were de-identified prior to analysis.
Power analysis was conducted to determine whether the study had sufficient population size to reject the null hypothesis for the primary outcome measure. Because practitioners controlled postoperative opioid use, a Cohen’s d of 1.0 was used so that a very large effect size was needed to reach clinical significance. Statistical significance was set to 0.05, and patient groups were set at 20 patients each. This yielded an appropriate power of 0.87. Population characteristics were compared between groups using t tests and χ2 tests as appropriate. To analyze the primary outcome, comparisons were made between the 2 cohorts using 2-tailed t tests. Secondary outcomes were compared between groups using t tests or χ2 tests. All statistics were performed using R version 3.5.2. Power analysis was conducted using the package pwr.11 Statistical significance was set at
Results
Forty patients met the inclusion criteria, evenly divided between those undergoing TKA before and after instituting the MOJO protocol (Table 2). A single patient in the MOJO group died and was excluded. A patient who underwent bilateral TKA also was excluded. Both groups reflected the male predominance of the VA patient population. MOJO patients tended to have lower BMIs (34 vs 30, P < .01). All patients indicated for surgery with preoperative opioid use were able to titrate down to their preoperative goal as verified by prescriptions filled at VA pharmacies. Twelve of the patients in the MOJO group received adductor canal blocks.
Results of t tests and χ2 tests comparing primary and secondary endpoints are listed in Table 3. Differences between the daily MEDs given in the historical and MOJO groups are shown. There were significant differences between the pre-MOJO and MOJO groups with regard to daily inpatient MEDs (82 mg vs 29 mg, P < .01) and total inpatient MEDs (306 mg vs 32 mg, P < .01). There was less self-reported pain on postoperative day 1 in the MOJO group (5.5 vs 3.9, P < .01), decreased LOS (4.4 days vs 1.2 days, P < .01), a trend toward fewer total ED visits (6 vs 2, P = .24), and fewer discharges to skilled nursing facilities (12 vs 0, P < .01). There were no blood transfusions in either group.
There were no readmissions due to uncontrolled pain. There was 1 readmission for shortness of breath in the MOJO group. The patient was discharged home the following day after ruling out thromboembolic and cardiovascular events. One patient from the control group was readmitted after missing a step on a staircase and falling. The patient sustained a quadriceps tendon rupture and underwent primary suture repair.
Discussion
Our results demonstrate that a multimodal approach to significantly reduce postoperative opioid use in patients with TKA is possible without increasing readmissions or ED visits for pain control. The patients in the MOJO group had a faster recovery, earlier discharge, and less use of postoperative opioid medication. Our approach to postoperative pain management was divided into 2 main categories: patient optimization and surgical optimization.
Patient Selection
Besides the standard evaluation and optimization of patients’ medical conditions, identifying and optimizing at-risk patients before surgery was a critical component of our protocol. Managing postoperative pain in patients with prior opioid use is an intractable challenge in orthopedic surgery. Patients with a history of chronic pain and preoperative use of opioid medications remain at higher risk of postoperative chronic pain and persistent use of opioid medication despite no obvious surgical complications.8 In a sample of > 6,000 veterans who underwent TKA at VA hospitals in 2014, 57% of the patients with daily use of opioids in the 90 days before surgery remained on opioids 1 year after surgery (vs 2 % in patients not on long-term opioids).8 This relationship between pre- and postoperative opioid use also was dose dependent.12
Furthermore, those with high preoperative use may experience worse outcomes relative to the opioid naive population as measured by arthritis-specific pain indices.13 In a well-powered retrospective study of patients who underwent elective orthopedic procedures, preoperative opioid abuse or dependence (determined by the International Classification of Diseases, Ninth Revision diagnosis) increased inpatient mortality, aggregate morbidity, surgical site infection, myocardial infarction, and LOS.14 Preoperative opioid use also has been associated with increased risk of ED visits, readmission, infection, stiffness, and aseptic revision.15 In patients with TKA in the VA specifically, preoperative opioid use (> 3 months in the prior year) was associated with increased revision rates that were even higher than those for patients with diabetes mellitus.16
Patient Education
Based on this evidence, we instruct patients to reduce their preoperative opioid dosing to zero (for patients with joint pain) or < 30 MED (for patients using opioids for other reasons). Although preoperative reduction of opioid use has been shown to improve outcomes after TKA, pain subspecialty recommendations for patients with chronic opioid use recommend considering adjunctive therapies, including transcutaneous electrical nerve stimulation, cognitive behavioral therapy, gabapentin, or ketamine.17,18 Through patient education our team has been successful in decreasing preoperative opioid use without adding other drugs or modalities.
Patient Optimization
Preoperative patient optimization included 4 to 8 weeks of daily sets of physical activity instructions (prehab) to improve the musculoskeletal function. These instructions are given to patients 4 to 8 weeks before surgery and aim to improve the patient’s balance, mobility, and functional ability (Appendix). Meta-analysis has shown that patients who undergo preoperative PT have a small but statistically significant decrease in postoperative pain at 4 weeks, though this does not persist beyond that period.19
We did note a lower BMI in patients in the MOJO group. Though this has the potential to be a confounder, a study of BMI in > 4,000 patients who underwent joint replacement surgery has shown that BMI is not associated with differences in postoperative pain.20
Surgeon and Surgical-Related Variables
Patients in the MOJO group had increased use of adductor canal blocks. A 2017 meta-analysis of 12,530 patients comparing analgesic modalities found that peripheral nerve blocks targeting multiple nerves (eg, femoral/sciatic) decreased pain at rest, decreased opioid consumption, and improved range of motion postoperatively.21 Also, these were found to be superior to single nerve blocks, periarticular infiltration, and epidural blocks.21 However, major nerve and epidural blocks affecting the lower extremity may increase the risk of falls and prolong LOS.22,23 The preferred peripheral block at VAPHCS is a single shot ultrasound-guided adductor canal block before the induction of general or spinal anesthesia. A randomized controlled trial has demonstrated superiority of this block to the femoral nerve block with regard to postoperative quadriceps strength, conferring the theoretical advantage of decreased fall risk and ability to participate in immediate PT.24 Although we are unable to confirm an association between anesthetic modalities and opioid burden, our clinical impression is that blocks were effective at reducing immediate postoperative pain. However, among MOJO patients there were no differences in patients with and without blocks for either pain (4.2 vs 3.8, P = .69) or opioid consumption (28.8 vs 33.0, P = .72) after surgery, though our study was not powered to detect a difference in this restricted subgroup.
Patients who frequently had reported postoperative thigh pain prompted us to make changes in our surgical technique, performing TKA without use of a tourniquet. Tourniquet use has been associated with an increased risk of thigh pain after TKA by multiple authors.25,26 Postoperative thigh pain also is pressure dependent.27 In addition, its use may be associated with a slightly increased risk of thromboembolic events and delayed functional recovery.28,29
Because postoperative hemarthrosis is associated with more pain and reduced joint recovery function, we used topical TXA to reduce postoperative surgical site and joint hematoma. TXA (either oral, IV, or topical) during TKA is used to control postoperative bleeding primarily and decrease the need for transfusion without concomitant increase in thromboembolic events.30,31 Topical TXA may be more effective than IV, particularly in the immediate postoperative period.32 Although pain typically is not an endpoint in studies of TXA, a prospective study of 48 patients showed evidence that its use may be associated with decreased postoperative pain in the first 24 hours after surgery (though not after).33 Finally, the use of intra-articular injection has evolved in our clinical practice, but literature is lacking with regard to its efficacy; more studies are needed to determine its effect relative to no injection. We have not seen any benefits to using
Limitations
This is a nonrandomized retrospective single-institution study. Our study population is composed of mostly males with military experience and is not necessarily a representative sample of the general population eligible for joint arthroplasty. Our primary endpoint (reduction of opioid use postoperatively) also was a cornerstone of our intervention. To account for this, we set a very large effect size in our power analysis and evaluated multiple secondary endpoints to determine whether postoperative pain remained well controlled and complications/readmission minimized with our interventions. Because our intervention was multimodal, our study cannot make conclusions about the effect of a particular component of our treatment strategy. We did not measure or compare functional outcomes between both groups, which offers an opportunity for further research.
These limitations are balanced by several strengths. Our cohort was well controlled with respect to the dose and type of drug used. There is staff dedicated to postoperative telephone follow-up after discharge, and veterans are apt to seek care within the VA health care system, which improves case finding for complications and ED visits. No patients were lost to follow-up. Moreover, our drastic reduction in opioid use is promising enough to warrant reporting, while the broader orthopedic literature explores the relative impact of each variable.
Conclusions
The MOJO protocol has been effective for reducing postoperative opioid use after TKA without compromising effective pain management. The drastic reduction in the postoperative use of opioid pain medications and LOS have contributed to a cultural shift within our department, comprehensive team approach, multimodal pain management, and preoperative patient optimization. Further investigations are required to assess the impact of each intervention on observed outcomes. However, the framework and routines are applicable to other institutions and surgical specialties.
Acknowledgments
The authors recognize Derek Bond, MD, for his help in creating the MOJO acronym.
1. Hedegaard H, Miniño AM, Warner M. Drug overdose deaths in the United States, 1999-2017. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics Data Brief No. 329. Published November 2018. Accessed January 12, 2021. https://www.cdc.gov/nchs/data/databriefs/db329-h.pdf
2. Hedegaard H, Warner M, Miniño AM. Drug overdose deaths in the United States, 1999-2016. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics NCHS data brief No. 294. Published December 2017. Accessed January 12, 2021. https://www.cdc.gov/nchs/data/databriefs/db294.pdf
3. Levy B, Paulozzi L, Mack KA, Jones CM. Trends in opioid analgesic–prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413. doi:10.1016/j.amepre.2015.02.020
4. Guy GP, Zhang K. Opioid prescribing by specialty and volume in the U.S. Am J Prev Med. 2018;55(5):e153-155. doi:10.1016/j.amepre.2018.06.008
5. Kremers HM, Larson DR, Crowson CS, et al. Prevalence of total hip and knee replacement in the United States. J Bone Joint Surgery Am. 2015;17:1386-1397. doi:10.2106/JBJS.N.01141
6. Giori NJ, Amanatullah DF, Gupta S, Bowe T, Harris AHS. Risk reduction compared with access to care: quantifying the trade-off of enforcing a body mass index eligibility criterion for joint replacement. J Bone Joint Surg Am. 2018; 4(100):539-545. doi:10.2106/JBJS.17.00120
7. Sabatino MJ, Kunkel ST, Ramkumar DB, Keeney BJ, Jevsevar DS. Excess opioid medication and variation in prescribing patterns following common orthopaedic procedures. J Bone Joint Surg Am. 2018;100(3):180-188. doi:10.2106/JBJS.17.00672
8. Hadlandsmyth K, Vander Weg MW, McCoy KD, Mosher HJ, Vaughan-Sarrazin MS, Lund BC. Risk for prolonged opioid use following total knee arthroplasty in veterans. J Arthroplasty. 2018;33(1):119-123. doi:10.1016/j.arth.2017.08.022
9. Bohnert ASB, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305(13):1315-1321. doi:10.1001/jama.2011.370
10. Hall MJ, Schwartzman A, Zhang J, Liu X. Ambulatory surgery data from hospitals and ambulatory surgery centers: United States, 2010. Natl Health Stat Report. 2017(102):1-15.
11. Champely S. pwr: basic functions for power analysis. R package version 1.2-2; 2018. Accessed January 13, 2021. https://rdrr.io/cran/pwr/
12. Goesling J, Moser SE, Zaidi B, et al. Trends and predictors of opioid use after total knee and total hip arthroplasty. Pain. 2016;157(6):1259-1265. doi:10.1097/j.pain.0000000000000516
13. Smith SR, Bido J, Collins JE, Yang H, Katz JN, Losina E. Impact of preoperative opioid use on total knee arthroplasty outcomes. J Bone Joint Surg Am. 2017;99(10):803-808. doi:10.2106/JBJS.16.01200
14. Menendez ME, Ring D, Bateman BT. Preoperative opioid misuse is associated with increased morbidity and mortality after elective orthopaedic surgery. Clin Orthop Relat Res. 2015;473(7):2402-412. doi:10.1007/s11999-015-4173-5
15. Cancienne JM, Patel KJ, Browne JA, Werner BC. Narcotic use and total knee arthroplasty. J Arthroplasty. 2018;33(1):113-118. doi:10.1016/j.arth.2017.08.006
16. Ben-Ari A, Chansky H, Rozet I. Preoperative opioid use is associated with early revision after total knee arthroplasty: a study of male patients treated in the Veterans Affairs System. J Bone Joint Surg Am. 2017;99(1):1-9. doi:10.2106/JBJS.16.00167
17. Nguyen L-CL, Sing DC, Bozic KJ. Preoperative reduction of opioid use before total joint arthroplasty. J Arthroplasty. 2016;31(suppl 9):282-287. doi:10.1016/j.arth.2016.01.068
18. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-157. doi:10.1016/j.jpain.2015.12.008
19. Wang L, Lee M, Zhang Z, Moodie J, Cheng D, Martin J. Does preoperative rehabilitation for patients planning to undergo joint replacement surgery improve outcomes? A systematic review and meta-analysis of randomised controlled trials. BMJ Open. 2016;6(2):e009857. doi:10.1136/bmjopen-2015-009857
20. Li W, Ayers DC, Lewis CG, Bowen TR, Allison JJ, Franklin PD. Functional gain and pain relief after total joint replacement according to obesity status. J Bone Joint Surg. 2017;99(14):1183-1189. doi:10.2106/JBJS.16.00960
21. Terkawi AS, Mavridis D, Sessler DI, et al. Pain management modalities after total knee arthroplasty: a network meta-analysis of 170 randomized controlled trials. Anesthesiology. 2017;126(5):923-937. doi:10.1097/ALN.0000000000001607
22. Ilfeld BM, Duke KB, Donohue MC. The association between lower extremity continuous peripheral nerve blocks and patient falls after knee and hip arthroplasty. Anesth Analg. 2010;111(6):1552-1554. doi:10.1213/ANE.0b013e3181fb9507
23. Elkassabany NM, Antosh S, Ahmed M, et al. The risk of falls after total knee arthroplasty with the use of a femoral nerve block versus an adductor canal block. Anest Analg. 2016;122(5):1696-1703. doi:10.1213/ane.0000000000001237
24. Wang D, Yang Y, Li Q, et al. Adductor canal block versus femoral nerve block for total knee arthroplasty: a meta-analysis of randomized controlled trials. Sci Rep. 2017;7:40721. doi:10.1038/srep40721
25. Liu D, Graham D, Gillies K, Gillies RM. Effects of tourniquet use on quadriceps function and pain in total knee arthroplasty. Knee Surg Relat Res. 2014;26(4):207-213. doi:10.5792/ksrr.2014.26.4.207
26. Abdel-Salam A, Eyres KS. Effects of tourniquet during total knee arthroplasty. A prospective randomised study. J Bone Joint Surg Br. 1995;77(2):250-253.
27. Worland RL, Arredondo J, Angles F, Lopez-Jimenez F, Jessup DE. Thigh pain following tourniquet application in simultaneous bilateral total knee replacement arthroplasty. J Arthroplasty. 1997;12(8):848-852. doi:10.1016/s0883-5403(97)90153-4
28. Tai T-W, Lin C-J, Jou I-M, Chang C-W, Lai K-A, Yang C-Y. Tourniquet use in total knee arthroplasty: a meta-analysis. Knee Surg Sports Traumatol, Arthrosc. 2011;19(7):1121-1130. doi:10.1007/s00167-010-1342-7
29. Jiang F-Z, Zhong H-M, Hong Y-C, Zhao G-F. Use of a tourniquet in total knee arthroplasty: a systematic review and meta-analysis of randomized controlled trials. J Orthop Sci. 2015;20(21):110-123. doi:10.1007/s00776-014-0664-6
30. Alshryda S, Sarda P, Sukeik M, Nargol A, Blenkinsopp J, Mason JM. Tranexamic acid in total knee replacement: a systematic review and meta-analysis. J Bone Joint Surg Br. 2011;93(12):1577-1585. doi:10.1302/0301-620X.93B12.26989
31. Panteli M, Papakostidis C, Dahabreh Z, Giannoudis PV. Topical tranexamic acid in total knee replacement: a systematic review and meta-analysis. Knee. 2013;20(5):300-309. doi:10.1016/j.knee.2013.05.014
32. Wang J, Wang Q, Zhang X, Wang Q. Intra-articular application is more effective than intravenous application of tranexamic acid in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2017;32(11):3385-3389. doi:10.1016/j.arth.2017.06.024
33. Guerreiro JPF, Badaro BS, Balbino JRM, Danieli MV, Queiroz AO, Cataneo DC. Application of tranexamic acid in total knee arthroplasty – prospective randomized trial. J Open Orthop J. 2017;11:1049-1057. doi:10.2174/1874325001711011049
1. Hedegaard H, Miniño AM, Warner M. Drug overdose deaths in the United States, 1999-2017. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics Data Brief No. 329. Published November 2018. Accessed January 12, 2021. https://www.cdc.gov/nchs/data/databriefs/db329-h.pdf
2. Hedegaard H, Warner M, Miniño AM. Drug overdose deaths in the United States, 1999-2016. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics NCHS data brief No. 294. Published December 2017. Accessed January 12, 2021. https://www.cdc.gov/nchs/data/databriefs/db294.pdf
3. Levy B, Paulozzi L, Mack KA, Jones CM. Trends in opioid analgesic–prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413. doi:10.1016/j.amepre.2015.02.020
4. Guy GP, Zhang K. Opioid prescribing by specialty and volume in the U.S. Am J Prev Med. 2018;55(5):e153-155. doi:10.1016/j.amepre.2018.06.008
5. Kremers HM, Larson DR, Crowson CS, et al. Prevalence of total hip and knee replacement in the United States. J Bone Joint Surgery Am. 2015;17:1386-1397. doi:10.2106/JBJS.N.01141
6. Giori NJ, Amanatullah DF, Gupta S, Bowe T, Harris AHS. Risk reduction compared with access to care: quantifying the trade-off of enforcing a body mass index eligibility criterion for joint replacement. J Bone Joint Surg Am. 2018; 4(100):539-545. doi:10.2106/JBJS.17.00120
7. Sabatino MJ, Kunkel ST, Ramkumar DB, Keeney BJ, Jevsevar DS. Excess opioid medication and variation in prescribing patterns following common orthopaedic procedures. J Bone Joint Surg Am. 2018;100(3):180-188. doi:10.2106/JBJS.17.00672
8. Hadlandsmyth K, Vander Weg MW, McCoy KD, Mosher HJ, Vaughan-Sarrazin MS, Lund BC. Risk for prolonged opioid use following total knee arthroplasty in veterans. J Arthroplasty. 2018;33(1):119-123. doi:10.1016/j.arth.2017.08.022
9. Bohnert ASB, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305(13):1315-1321. doi:10.1001/jama.2011.370
10. Hall MJ, Schwartzman A, Zhang J, Liu X. Ambulatory surgery data from hospitals and ambulatory surgery centers: United States, 2010. Natl Health Stat Report. 2017(102):1-15.
11. Champely S. pwr: basic functions for power analysis. R package version 1.2-2; 2018. Accessed January 13, 2021. https://rdrr.io/cran/pwr/
12. Goesling J, Moser SE, Zaidi B, et al. Trends and predictors of opioid use after total knee and total hip arthroplasty. Pain. 2016;157(6):1259-1265. doi:10.1097/j.pain.0000000000000516
13. Smith SR, Bido J, Collins JE, Yang H, Katz JN, Losina E. Impact of preoperative opioid use on total knee arthroplasty outcomes. J Bone Joint Surg Am. 2017;99(10):803-808. doi:10.2106/JBJS.16.01200
14. Menendez ME, Ring D, Bateman BT. Preoperative opioid misuse is associated with increased morbidity and mortality after elective orthopaedic surgery. Clin Orthop Relat Res. 2015;473(7):2402-412. doi:10.1007/s11999-015-4173-5
15. Cancienne JM, Patel KJ, Browne JA, Werner BC. Narcotic use and total knee arthroplasty. J Arthroplasty. 2018;33(1):113-118. doi:10.1016/j.arth.2017.08.006
16. Ben-Ari A, Chansky H, Rozet I. Preoperative opioid use is associated with early revision after total knee arthroplasty: a study of male patients treated in the Veterans Affairs System. J Bone Joint Surg Am. 2017;99(1):1-9. doi:10.2106/JBJS.16.00167
17. Nguyen L-CL, Sing DC, Bozic KJ. Preoperative reduction of opioid use before total joint arthroplasty. J Arthroplasty. 2016;31(suppl 9):282-287. doi:10.1016/j.arth.2016.01.068
18. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-157. doi:10.1016/j.jpain.2015.12.008
19. Wang L, Lee M, Zhang Z, Moodie J, Cheng D, Martin J. Does preoperative rehabilitation for patients planning to undergo joint replacement surgery improve outcomes? A systematic review and meta-analysis of randomised controlled trials. BMJ Open. 2016;6(2):e009857. doi:10.1136/bmjopen-2015-009857
20. Li W, Ayers DC, Lewis CG, Bowen TR, Allison JJ, Franklin PD. Functional gain and pain relief after total joint replacement according to obesity status. J Bone Joint Surg. 2017;99(14):1183-1189. doi:10.2106/JBJS.16.00960
21. Terkawi AS, Mavridis D, Sessler DI, et al. Pain management modalities after total knee arthroplasty: a network meta-analysis of 170 randomized controlled trials. Anesthesiology. 2017;126(5):923-937. doi:10.1097/ALN.0000000000001607
22. Ilfeld BM, Duke KB, Donohue MC. The association between lower extremity continuous peripheral nerve blocks and patient falls after knee and hip arthroplasty. Anesth Analg. 2010;111(6):1552-1554. doi:10.1213/ANE.0b013e3181fb9507
23. Elkassabany NM, Antosh S, Ahmed M, et al. The risk of falls after total knee arthroplasty with the use of a femoral nerve block versus an adductor canal block. Anest Analg. 2016;122(5):1696-1703. doi:10.1213/ane.0000000000001237
24. Wang D, Yang Y, Li Q, et al. Adductor canal block versus femoral nerve block for total knee arthroplasty: a meta-analysis of randomized controlled trials. Sci Rep. 2017;7:40721. doi:10.1038/srep40721
25. Liu D, Graham D, Gillies K, Gillies RM. Effects of tourniquet use on quadriceps function and pain in total knee arthroplasty. Knee Surg Relat Res. 2014;26(4):207-213. doi:10.5792/ksrr.2014.26.4.207
26. Abdel-Salam A, Eyres KS. Effects of tourniquet during total knee arthroplasty. A prospective randomised study. J Bone Joint Surg Br. 1995;77(2):250-253.
27. Worland RL, Arredondo J, Angles F, Lopez-Jimenez F, Jessup DE. Thigh pain following tourniquet application in simultaneous bilateral total knee replacement arthroplasty. J Arthroplasty. 1997;12(8):848-852. doi:10.1016/s0883-5403(97)90153-4
28. Tai T-W, Lin C-J, Jou I-M, Chang C-W, Lai K-A, Yang C-Y. Tourniquet use in total knee arthroplasty: a meta-analysis. Knee Surg Sports Traumatol, Arthrosc. 2011;19(7):1121-1130. doi:10.1007/s00167-010-1342-7
29. Jiang F-Z, Zhong H-M, Hong Y-C, Zhao G-F. Use of a tourniquet in total knee arthroplasty: a systematic review and meta-analysis of randomized controlled trials. J Orthop Sci. 2015;20(21):110-123. doi:10.1007/s00776-014-0664-6
30. Alshryda S, Sarda P, Sukeik M, Nargol A, Blenkinsopp J, Mason JM. Tranexamic acid in total knee replacement: a systematic review and meta-analysis. J Bone Joint Surg Br. 2011;93(12):1577-1585. doi:10.1302/0301-620X.93B12.26989
31. Panteli M, Papakostidis C, Dahabreh Z, Giannoudis PV. Topical tranexamic acid in total knee replacement: a systematic review and meta-analysis. Knee. 2013;20(5):300-309. doi:10.1016/j.knee.2013.05.014
32. Wang J, Wang Q, Zhang X, Wang Q. Intra-articular application is more effective than intravenous application of tranexamic acid in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2017;32(11):3385-3389. doi:10.1016/j.arth.2017.06.024
33. Guerreiro JPF, Badaro BS, Balbino JRM, Danieli MV, Queiroz AO, Cataneo DC. Application of tranexamic acid in total knee arthroplasty – prospective randomized trial. J Open Orthop J. 2017;11:1049-1057. doi:10.2174/1874325001711011049
Opioid-related deaths lower in counties with active cannabis dispensaries
Areas with active cannabis dispensaries have seen a decrease in opioid-related mortalities, recent research has shown.
“Our findings suggest that higher storefront cannabis dispensary counts are associated with reduced opioid related mortality rates at the county level,” wrote Greta Hsu, PhD, professor of management, University of California, Davis, and Balázs Kovács, PhD, associate professor of organizational behavior, Yale University, New Haven, Conn. “, which include the highly potent synthetic opioid fentanyl and its analogs.”
In the study, published in BMJ, the researchers evaluated the prevalence of medical and recreational cannabis dispensaries in 812 U.S. counties within 23 states with some degree of cannabis legalization between 2014 and 2018. Overall, dispensaries located in counties in eight U.S. states and the District of Columbia that sold cannabis recreationally and an additional 15 states that contained medical cannabis dispensaries were included.
Dr. Hsu and Dr. Kovács performed their analysis by examining dispensaries that were operating storefronts by the end of 2017 at the county level using panel-regression methods, combining data obtained from the consumer-facing website Weedmaps.com, Centers for Disease Control and Prevention U.S. mortality data, and data from the U.S. Census Bureau.
To measure opioid-related mortality, the researchers measured ICD-10 codes specific to natural opioid analgesics and semisynthetic opioids, methadone, heroin, nonmethadone synthetic opioid analgesics, and fentanyl-related deaths.
The analysis showed a negative association between the number of cannabis dispensaries at the county level and overall opioid-related mortality rates (95% confidence interval, −0.23 to −0.11), with an increase from one to two dispensaries in a county resulting in a 17% decrease in opioid-related mortality rates and an increase from two to three dispensaries resulting in another decrease in opioid-related mortality of 8.5%.
When evaluating mortality by specific opioid type, the researchers found a negative association between the number of dispensaries and synthetic nonmethadone opioids, with an increase from one to two dispensaries resulting in a 21% decrease in mortality attributable to synthetic nonmethadone opioids (95% CI, −0.27 to −0.14; P = .002). There were also negative associations between the number of dispensaries and prescription opioid-related mortality rates (95% CI, −0.13 to −0.03) and heroin-related mortality rates (95% CI, −0.13 to −0.02). The negative association was similar in comparisons between synthetic nonmethadone opioid-related mortality and the number of dispensaries for medical cannabis (95% CI, −0.21 to −0.09; P = .002) and recreational cannabis (95% CI, −0.17 to −0.04; P = .01).
Evidence of a negative association between legalization of medical or recreational cannabis and opioid-related mortality has been mixed in the literature, with some studies also showing a “spurious or nonsignificant” association, according to Dr. Hsu and Dr. Kovács.
While previous studies have looked at the legalization of cannabis for medical or recreational use, legalization on its own is an “incomplete picture,” they said, which might offer one explanation for these mixed findings. Some states that legalize medical cannabis, for example, might not allow dispensaries to legally sell cannabis, and there may be a delay of 1-2 years between the time a state legalizes cannabis for recreational use and when dispensaries are open and available to the public.
“These results were obtained after controlling for county level population characteristics, yearly effects, whether recreational dispensaries were legal or not in the focal county’s state, and opioid-related state policies,” the authors wrote.
Results ‘may be even stronger’ than reported
Christopher G. Fichtner, MD, clinical professor of psychiatry and neuroscience at the University of California, Riverside, said in an interview that the evidence for using cannabis as an opioid substitution for pain management has not been balanced, but noted “the bulk of it suggests that there is some harm reduction benefit by having liberalized access to cannabis.”
One strength of the study by Dr. Hsu and Dr. Kovács was how they were able to examine implementation of legalization of medical or recreational cannabis, rather than simply a change in the law, he said.
“By looking at dispensary count, it’s actually looking at a better measure of on-the-ground implementation than just change in policy,” Dr. Fichtner explained. “You’re looking at what was actually accomplished in terms of making cannabis legally available.”
The choice to evaluate storefront dispensaries only and not include delivery services in their data, “probably makes it a relatively conservative estimate. I think that would be a strength, that their findings may be even stronger than what it is they’re reporting,” Dr. Fichtner said.
“I do think, if anything, the paper is relatively tentative about advancing its conclusions, which I think is a weakness in a lot of these studies,” he added. In 2017, the National Academy of Sciences released a report that found evidence cannabis or cannabinoids can significantly reduce pain symptoms. In that report, “one of their strongest conclusions is that there’s conclusive or substantial evidence that cannabis or cannabinoids are effective management of chronic pain,” Dr. Fichtner said.
He said that digging deeper into what kinds of pain cannabis can treat is one area for future research. “Certainly, it seems that it’s unlikely that cannabis is going to be good for every kind of pain,” he said. “What kinds of pain is it better for than others? Is it some benefit for many kinds of pain, or only a few types of pain?”
The authors reported no relevant financial disclosures. Dr. Fichtner is the author of a book on cannabis policy in the United States, but reported no other financial disclosures.
Areas with active cannabis dispensaries have seen a decrease in opioid-related mortalities, recent research has shown.
“Our findings suggest that higher storefront cannabis dispensary counts are associated with reduced opioid related mortality rates at the county level,” wrote Greta Hsu, PhD, professor of management, University of California, Davis, and Balázs Kovács, PhD, associate professor of organizational behavior, Yale University, New Haven, Conn. “, which include the highly potent synthetic opioid fentanyl and its analogs.”
In the study, published in BMJ, the researchers evaluated the prevalence of medical and recreational cannabis dispensaries in 812 U.S. counties within 23 states with some degree of cannabis legalization between 2014 and 2018. Overall, dispensaries located in counties in eight U.S. states and the District of Columbia that sold cannabis recreationally and an additional 15 states that contained medical cannabis dispensaries were included.
Dr. Hsu and Dr. Kovács performed their analysis by examining dispensaries that were operating storefronts by the end of 2017 at the county level using panel-regression methods, combining data obtained from the consumer-facing website Weedmaps.com, Centers for Disease Control and Prevention U.S. mortality data, and data from the U.S. Census Bureau.
To measure opioid-related mortality, the researchers measured ICD-10 codes specific to natural opioid analgesics and semisynthetic opioids, methadone, heroin, nonmethadone synthetic opioid analgesics, and fentanyl-related deaths.
The analysis showed a negative association between the number of cannabis dispensaries at the county level and overall opioid-related mortality rates (95% confidence interval, −0.23 to −0.11), with an increase from one to two dispensaries in a county resulting in a 17% decrease in opioid-related mortality rates and an increase from two to three dispensaries resulting in another decrease in opioid-related mortality of 8.5%.
When evaluating mortality by specific opioid type, the researchers found a negative association between the number of dispensaries and synthetic nonmethadone opioids, with an increase from one to two dispensaries resulting in a 21% decrease in mortality attributable to synthetic nonmethadone opioids (95% CI, −0.27 to −0.14; P = .002). There were also negative associations between the number of dispensaries and prescription opioid-related mortality rates (95% CI, −0.13 to −0.03) and heroin-related mortality rates (95% CI, −0.13 to −0.02). The negative association was similar in comparisons between synthetic nonmethadone opioid-related mortality and the number of dispensaries for medical cannabis (95% CI, −0.21 to −0.09; P = .002) and recreational cannabis (95% CI, −0.17 to −0.04; P = .01).
Evidence of a negative association between legalization of medical or recreational cannabis and opioid-related mortality has been mixed in the literature, with some studies also showing a “spurious or nonsignificant” association, according to Dr. Hsu and Dr. Kovács.
While previous studies have looked at the legalization of cannabis for medical or recreational use, legalization on its own is an “incomplete picture,” they said, which might offer one explanation for these mixed findings. Some states that legalize medical cannabis, for example, might not allow dispensaries to legally sell cannabis, and there may be a delay of 1-2 years between the time a state legalizes cannabis for recreational use and when dispensaries are open and available to the public.
“These results were obtained after controlling for county level population characteristics, yearly effects, whether recreational dispensaries were legal or not in the focal county’s state, and opioid-related state policies,” the authors wrote.
Results ‘may be even stronger’ than reported
Christopher G. Fichtner, MD, clinical professor of psychiatry and neuroscience at the University of California, Riverside, said in an interview that the evidence for using cannabis as an opioid substitution for pain management has not been balanced, but noted “the bulk of it suggests that there is some harm reduction benefit by having liberalized access to cannabis.”
One strength of the study by Dr. Hsu and Dr. Kovács was how they were able to examine implementation of legalization of medical or recreational cannabis, rather than simply a change in the law, he said.
“By looking at dispensary count, it’s actually looking at a better measure of on-the-ground implementation than just change in policy,” Dr. Fichtner explained. “You’re looking at what was actually accomplished in terms of making cannabis legally available.”
The choice to evaluate storefront dispensaries only and not include delivery services in their data, “probably makes it a relatively conservative estimate. I think that would be a strength, that their findings may be even stronger than what it is they’re reporting,” Dr. Fichtner said.
“I do think, if anything, the paper is relatively tentative about advancing its conclusions, which I think is a weakness in a lot of these studies,” he added. In 2017, the National Academy of Sciences released a report that found evidence cannabis or cannabinoids can significantly reduce pain symptoms. In that report, “one of their strongest conclusions is that there’s conclusive or substantial evidence that cannabis or cannabinoids are effective management of chronic pain,” Dr. Fichtner said.
He said that digging deeper into what kinds of pain cannabis can treat is one area for future research. “Certainly, it seems that it’s unlikely that cannabis is going to be good for every kind of pain,” he said. “What kinds of pain is it better for than others? Is it some benefit for many kinds of pain, or only a few types of pain?”
The authors reported no relevant financial disclosures. Dr. Fichtner is the author of a book on cannabis policy in the United States, but reported no other financial disclosures.
Areas with active cannabis dispensaries have seen a decrease in opioid-related mortalities, recent research has shown.
“Our findings suggest that higher storefront cannabis dispensary counts are associated with reduced opioid related mortality rates at the county level,” wrote Greta Hsu, PhD, professor of management, University of California, Davis, and Balázs Kovács, PhD, associate professor of organizational behavior, Yale University, New Haven, Conn. “, which include the highly potent synthetic opioid fentanyl and its analogs.”
In the study, published in BMJ, the researchers evaluated the prevalence of medical and recreational cannabis dispensaries in 812 U.S. counties within 23 states with some degree of cannabis legalization between 2014 and 2018. Overall, dispensaries located in counties in eight U.S. states and the District of Columbia that sold cannabis recreationally and an additional 15 states that contained medical cannabis dispensaries were included.
Dr. Hsu and Dr. Kovács performed their analysis by examining dispensaries that were operating storefronts by the end of 2017 at the county level using panel-regression methods, combining data obtained from the consumer-facing website Weedmaps.com, Centers for Disease Control and Prevention U.S. mortality data, and data from the U.S. Census Bureau.
To measure opioid-related mortality, the researchers measured ICD-10 codes specific to natural opioid analgesics and semisynthetic opioids, methadone, heroin, nonmethadone synthetic opioid analgesics, and fentanyl-related deaths.
The analysis showed a negative association between the number of cannabis dispensaries at the county level and overall opioid-related mortality rates (95% confidence interval, −0.23 to −0.11), with an increase from one to two dispensaries in a county resulting in a 17% decrease in opioid-related mortality rates and an increase from two to three dispensaries resulting in another decrease in opioid-related mortality of 8.5%.
When evaluating mortality by specific opioid type, the researchers found a negative association between the number of dispensaries and synthetic nonmethadone opioids, with an increase from one to two dispensaries resulting in a 21% decrease in mortality attributable to synthetic nonmethadone opioids (95% CI, −0.27 to −0.14; P = .002). There were also negative associations between the number of dispensaries and prescription opioid-related mortality rates (95% CI, −0.13 to −0.03) and heroin-related mortality rates (95% CI, −0.13 to −0.02). The negative association was similar in comparisons between synthetic nonmethadone opioid-related mortality and the number of dispensaries for medical cannabis (95% CI, −0.21 to −0.09; P = .002) and recreational cannabis (95% CI, −0.17 to −0.04; P = .01).
Evidence of a negative association between legalization of medical or recreational cannabis and opioid-related mortality has been mixed in the literature, with some studies also showing a “spurious or nonsignificant” association, according to Dr. Hsu and Dr. Kovács.
While previous studies have looked at the legalization of cannabis for medical or recreational use, legalization on its own is an “incomplete picture,” they said, which might offer one explanation for these mixed findings. Some states that legalize medical cannabis, for example, might not allow dispensaries to legally sell cannabis, and there may be a delay of 1-2 years between the time a state legalizes cannabis for recreational use and when dispensaries are open and available to the public.
“These results were obtained after controlling for county level population characteristics, yearly effects, whether recreational dispensaries were legal or not in the focal county’s state, and opioid-related state policies,” the authors wrote.
Results ‘may be even stronger’ than reported
Christopher G. Fichtner, MD, clinical professor of psychiatry and neuroscience at the University of California, Riverside, said in an interview that the evidence for using cannabis as an opioid substitution for pain management has not been balanced, but noted “the bulk of it suggests that there is some harm reduction benefit by having liberalized access to cannabis.”
One strength of the study by Dr. Hsu and Dr. Kovács was how they were able to examine implementation of legalization of medical or recreational cannabis, rather than simply a change in the law, he said.
“By looking at dispensary count, it’s actually looking at a better measure of on-the-ground implementation than just change in policy,” Dr. Fichtner explained. “You’re looking at what was actually accomplished in terms of making cannabis legally available.”
The choice to evaluate storefront dispensaries only and not include delivery services in their data, “probably makes it a relatively conservative estimate. I think that would be a strength, that their findings may be even stronger than what it is they’re reporting,” Dr. Fichtner said.
“I do think, if anything, the paper is relatively tentative about advancing its conclusions, which I think is a weakness in a lot of these studies,” he added. In 2017, the National Academy of Sciences released a report that found evidence cannabis or cannabinoids can significantly reduce pain symptoms. In that report, “one of their strongest conclusions is that there’s conclusive or substantial evidence that cannabis or cannabinoids are effective management of chronic pain,” Dr. Fichtner said.
He said that digging deeper into what kinds of pain cannabis can treat is one area for future research. “Certainly, it seems that it’s unlikely that cannabis is going to be good for every kind of pain,” he said. “What kinds of pain is it better for than others? Is it some benefit for many kinds of pain, or only a few types of pain?”
The authors reported no relevant financial disclosures. Dr. Fichtner is the author of a book on cannabis policy in the United States, but reported no other financial disclosures.
FROM BMJ
No pain, if you’ve got game
ILLUSTRATIVE CASE
An 8-year-old girl with congenital heart disease (status: post repair) arrives at your clinic for a routine appointment. Since the age of 12 months, she has experienced significant anxiety during medical visits, especially with blood draws and injections. She enjoys playing video games on her new tablet computer. Her parents want to know what you can do to reduce her anxiety and pain during today’s scheduled blood draw. Should you recommend that she continue playing video games during the venipuncture?
Adequately managing pain while performing venipuncture in children can improve the quality of the experience, reduce children’s fear of going to the doctor, and increase efficiency in medical practice.2 Since pharmacologic pain-control methods may have adverse effects, distraction techniques—engaging the child in another activity during a procedure—are commonly used instead to help reduce a child’s pain. These techniques can be active or passive.
Studies have demonstrated that both active and passive distraction techniques reduce children’s pain during medical procedures, including venipuncture. Passive techniques, such as nurse coaching3 and watching cartoons,4 have been found to reduce distress and pain. Active distraction techniques, such as playing video games while undergoing a painful procedure (eg, dressing a wound), have been shown to be more effective than passive techniques.5,6
A Cochrane review and meta-analysis of distraction and hypnosis for needle-related pain and distress in children demonstrated reduced pain, but the quality of evidence was low and the review recommended improved methodological rigor and trial reporting.7 Another systematic review and analysis showed strong support for distraction for reducing pain; however, the quality of evidence was low and the researchers cited problems with characteristics of the distraction interventions, child age, and risk of bias in the studies.8
There has been a lack of RCTs comparing the effectiveness and superiority of active vs passive distraction techniques. The first high-quality RCT to directly compare 3 of the most common distraction techniques to a control group was recently conducted in a large training and research hospital in Turkey.1
STUDY SUMMARY
Pain and anxiety levels were lowest in actively distracted children
The RCT included 180 children ages 6 to 10 years randomly assigned to 1 of 3 intervention groups or a control group.1 Phlebotomy was performed while children watched a cartoon, played a video game, were distracted by parental interaction, or had no distraction (control group).
Investigators independently measured pain and anxiety in the patient and perceived pain and anxiety according to both a family member and a health care worker (medical observer). Researchers used the previously validated Children’s Fear Scale and the Wong-Baker Pain Scale.9,10 The Children’s Fear Scale was used to assess anxiety in children on a scale of 0 (picture of a calm face) to 4 (picture of the most fearful face). The Wong-Baker Pain Scale was used to assess pain on a scale of 0 (no hurt: happy face) to 10 (hurts worst: saddest face).
Continue to: Results
Results. The pain and anxiety scores were significantly lower in all of the intervention groups compared with the control group (P < .05). The video game (active distraction) group had the lowest levels of both pain and anxiety. The self-reported Children’s Fear Scale scores of children in the video game group were 0.27, compared with 0.76 in the cartoon group, 1.24 in the parental distraction group, and 2.22 in the control group. The anxiety scores recorded by the family member and the medical observer showed similar significant differences.
The Wong-Baker Pain Scale scores showed similar differences in self-reported pain for the video game group (1.42) compared with the cartoon group (3.02), the parental distraction group (2.89), and the control group (5.11). Pain scores reported by the family member and the medical observer (respectively) also reflected benefit from any type of distraction, with active game-playing as the most effective type of distraction (video game: 1.69 and 1.96; cartoon: 3.07 and 3.20; parental distraction: 3.56 and 4.22; and control: 5.29 and 6.13).
In addition, the intraclass correlation coefficient was 0.67 to 0.924 (P < .01), suggesting that the reports from the child, parent, and medical observer about the child’s pain and anxiety were highly correlated.
WHAT'S NEW
All distraction techniques provide benefit, but there’s a clear winner
In this RCT of children undergoing phlebotomy, both active and passive distraction techniques were superior to no distraction in terms of perceived pain and anxiety by the child, a health care provider, or a parent. The active-distraction group played a video game, while the passive-distraction groups watched a cartoon or interacted with a parent. Active distraction was superior to passive distraction.
CAVEATS
Procedure time was short; intervention not blinded
One potential weakness of this study is that it was not a double-blinded trial. Blinding was not possible for much of the study as the patient, parent, and medical observer were fully aware of the intervention or lack thereof. However, the parent and medical observer were blinded to each other’s assessments of the child’s pain and anxiety.
Continue to: Furthermore, the study...
Furthermore, the study was conducted at a single institution in Turkey. There could be cultural differences in reporting of pain and anxiety compared to Western cultures.
Finally, the average duration of the procedure in this study was 3 minutes, with a range of 1 to 5 minutes. It is unclear if the findings can be extrapolated to more time-consuming procedures.
CHALLENGES TO IMPLEMENTATION
Technology is not available to all
The use of tablet computers may seem increasingly ubiquitous, but not all families have access to these devices. Another challenge is that phlebotomy/clinic personnel must learn to work around the device.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Inan G, Inal S. The impact of 3 different distraction techniques on the pain and anxiety levels of children during venipuncture: a clinical trial. Clin J Pain. 2019;35:140-147.
2. Fein JA, Zempsky WT, Cravero JP, Committee on Pediatric Emergency Medicine and Section on Anesthesiology and Pain Medicine; American Academy of Pediatrics. Relief of pain and anxiety in pediatric patients in emergency medical systems. Pediatrics. 2012;130:e1391-e1405.
3. Cohen LL, Blount RL, Panopoulos G. Nurse coaching and cartoon distraction: an effective and practical intervention to reduce child, parent, and nurse distress during immunizations. J Pediatr Psychol. 1997;22:355-370.
4. Downey VA, Zun LS. The impact of watching cartoons for distraction during painful procedures in the emergency department. Pediatr Emerg. 2012;28:1033-1035.
5. Hussein H. Effect of active and passive distraction on decreasing pain associated with painful medical procedures among school aged children. World J Nurs Sci. 2015;1:13-23.
6. Nilsson S, Enskär K, Hallqvist C, et al. Active and passive distraction in children undergoing wound dressing. J Pediatr Nurs. 2013;28:158-166.
7. Birnie KA, Noel M, Chambers CT, et al. Psychological interventions for needle-related procedural pain and distress in children and adolescents. Cochrane Database Syst Rev. 2018;10:CD005179.
8. Birnie KA, Noel M, Parker JA, et al. Systematic review and meta-analysis of distraction and hypnosis for needle-related pain and distress in children and adolescents. J Pediatr Psychol. 2014;39:783-808.
9. McMurtry CM, Noel M, Chambers CT, et al. Children’s fear during procedural pain: preliminary investigation of the Children’s Fear Scale. Health Psychol. 2011;30:780-788.
10. Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatric Nurs. 1988;14:9-17.
ILLUSTRATIVE CASE
An 8-year-old girl with congenital heart disease (status: post repair) arrives at your clinic for a routine appointment. Since the age of 12 months, she has experienced significant anxiety during medical visits, especially with blood draws and injections. She enjoys playing video games on her new tablet computer. Her parents want to know what you can do to reduce her anxiety and pain during today’s scheduled blood draw. Should you recommend that she continue playing video games during the venipuncture?
Adequately managing pain while performing venipuncture in children can improve the quality of the experience, reduce children’s fear of going to the doctor, and increase efficiency in medical practice.2 Since pharmacologic pain-control methods may have adverse effects, distraction techniques—engaging the child in another activity during a procedure—are commonly used instead to help reduce a child’s pain. These techniques can be active or passive.
Studies have demonstrated that both active and passive distraction techniques reduce children’s pain during medical procedures, including venipuncture. Passive techniques, such as nurse coaching3 and watching cartoons,4 have been found to reduce distress and pain. Active distraction techniques, such as playing video games while undergoing a painful procedure (eg, dressing a wound), have been shown to be more effective than passive techniques.5,6
A Cochrane review and meta-analysis of distraction and hypnosis for needle-related pain and distress in children demonstrated reduced pain, but the quality of evidence was low and the review recommended improved methodological rigor and trial reporting.7 Another systematic review and analysis showed strong support for distraction for reducing pain; however, the quality of evidence was low and the researchers cited problems with characteristics of the distraction interventions, child age, and risk of bias in the studies.8
There has been a lack of RCTs comparing the effectiveness and superiority of active vs passive distraction techniques. The first high-quality RCT to directly compare 3 of the most common distraction techniques to a control group was recently conducted in a large training and research hospital in Turkey.1
STUDY SUMMARY
Pain and anxiety levels were lowest in actively distracted children
The RCT included 180 children ages 6 to 10 years randomly assigned to 1 of 3 intervention groups or a control group.1 Phlebotomy was performed while children watched a cartoon, played a video game, were distracted by parental interaction, or had no distraction (control group).
Investigators independently measured pain and anxiety in the patient and perceived pain and anxiety according to both a family member and a health care worker (medical observer). Researchers used the previously validated Children’s Fear Scale and the Wong-Baker Pain Scale.9,10 The Children’s Fear Scale was used to assess anxiety in children on a scale of 0 (picture of a calm face) to 4 (picture of the most fearful face). The Wong-Baker Pain Scale was used to assess pain on a scale of 0 (no hurt: happy face) to 10 (hurts worst: saddest face).
Continue to: Results
Results. The pain and anxiety scores were significantly lower in all of the intervention groups compared with the control group (P < .05). The video game (active distraction) group had the lowest levels of both pain and anxiety. The self-reported Children’s Fear Scale scores of children in the video game group were 0.27, compared with 0.76 in the cartoon group, 1.24 in the parental distraction group, and 2.22 in the control group. The anxiety scores recorded by the family member and the medical observer showed similar significant differences.
The Wong-Baker Pain Scale scores showed similar differences in self-reported pain for the video game group (1.42) compared with the cartoon group (3.02), the parental distraction group (2.89), and the control group (5.11). Pain scores reported by the family member and the medical observer (respectively) also reflected benefit from any type of distraction, with active game-playing as the most effective type of distraction (video game: 1.69 and 1.96; cartoon: 3.07 and 3.20; parental distraction: 3.56 and 4.22; and control: 5.29 and 6.13).
In addition, the intraclass correlation coefficient was 0.67 to 0.924 (P < .01), suggesting that the reports from the child, parent, and medical observer about the child’s pain and anxiety were highly correlated.
WHAT'S NEW
All distraction techniques provide benefit, but there’s a clear winner
In this RCT of children undergoing phlebotomy, both active and passive distraction techniques were superior to no distraction in terms of perceived pain and anxiety by the child, a health care provider, or a parent. The active-distraction group played a video game, while the passive-distraction groups watched a cartoon or interacted with a parent. Active distraction was superior to passive distraction.
CAVEATS
Procedure time was short; intervention not blinded
One potential weakness of this study is that it was not a double-blinded trial. Blinding was not possible for much of the study as the patient, parent, and medical observer were fully aware of the intervention or lack thereof. However, the parent and medical observer were blinded to each other’s assessments of the child’s pain and anxiety.
Continue to: Furthermore, the study...
Furthermore, the study was conducted at a single institution in Turkey. There could be cultural differences in reporting of pain and anxiety compared to Western cultures.
Finally, the average duration of the procedure in this study was 3 minutes, with a range of 1 to 5 minutes. It is unclear if the findings can be extrapolated to more time-consuming procedures.
CHALLENGES TO IMPLEMENTATION
Technology is not available to all
The use of tablet computers may seem increasingly ubiquitous, but not all families have access to these devices. Another challenge is that phlebotomy/clinic personnel must learn to work around the device.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
An 8-year-old girl with congenital heart disease (status: post repair) arrives at your clinic for a routine appointment. Since the age of 12 months, she has experienced significant anxiety during medical visits, especially with blood draws and injections. She enjoys playing video games on her new tablet computer. Her parents want to know what you can do to reduce her anxiety and pain during today’s scheduled blood draw. Should you recommend that she continue playing video games during the venipuncture?
Adequately managing pain while performing venipuncture in children can improve the quality of the experience, reduce children’s fear of going to the doctor, and increase efficiency in medical practice.2 Since pharmacologic pain-control methods may have adverse effects, distraction techniques—engaging the child in another activity during a procedure—are commonly used instead to help reduce a child’s pain. These techniques can be active or passive.
Studies have demonstrated that both active and passive distraction techniques reduce children’s pain during medical procedures, including venipuncture. Passive techniques, such as nurse coaching3 and watching cartoons,4 have been found to reduce distress and pain. Active distraction techniques, such as playing video games while undergoing a painful procedure (eg, dressing a wound), have been shown to be more effective than passive techniques.5,6
A Cochrane review and meta-analysis of distraction and hypnosis for needle-related pain and distress in children demonstrated reduced pain, but the quality of evidence was low and the review recommended improved methodological rigor and trial reporting.7 Another systematic review and analysis showed strong support for distraction for reducing pain; however, the quality of evidence was low and the researchers cited problems with characteristics of the distraction interventions, child age, and risk of bias in the studies.8
There has been a lack of RCTs comparing the effectiveness and superiority of active vs passive distraction techniques. The first high-quality RCT to directly compare 3 of the most common distraction techniques to a control group was recently conducted in a large training and research hospital in Turkey.1
STUDY SUMMARY
Pain and anxiety levels were lowest in actively distracted children
The RCT included 180 children ages 6 to 10 years randomly assigned to 1 of 3 intervention groups or a control group.1 Phlebotomy was performed while children watched a cartoon, played a video game, were distracted by parental interaction, or had no distraction (control group).
Investigators independently measured pain and anxiety in the patient and perceived pain and anxiety according to both a family member and a health care worker (medical observer). Researchers used the previously validated Children’s Fear Scale and the Wong-Baker Pain Scale.9,10 The Children’s Fear Scale was used to assess anxiety in children on a scale of 0 (picture of a calm face) to 4 (picture of the most fearful face). The Wong-Baker Pain Scale was used to assess pain on a scale of 0 (no hurt: happy face) to 10 (hurts worst: saddest face).
Continue to: Results
Results. The pain and anxiety scores were significantly lower in all of the intervention groups compared with the control group (P < .05). The video game (active distraction) group had the lowest levels of both pain and anxiety. The self-reported Children’s Fear Scale scores of children in the video game group were 0.27, compared with 0.76 in the cartoon group, 1.24 in the parental distraction group, and 2.22 in the control group. The anxiety scores recorded by the family member and the medical observer showed similar significant differences.
The Wong-Baker Pain Scale scores showed similar differences in self-reported pain for the video game group (1.42) compared with the cartoon group (3.02), the parental distraction group (2.89), and the control group (5.11). Pain scores reported by the family member and the medical observer (respectively) also reflected benefit from any type of distraction, with active game-playing as the most effective type of distraction (video game: 1.69 and 1.96; cartoon: 3.07 and 3.20; parental distraction: 3.56 and 4.22; and control: 5.29 and 6.13).
In addition, the intraclass correlation coefficient was 0.67 to 0.924 (P < .01), suggesting that the reports from the child, parent, and medical observer about the child’s pain and anxiety were highly correlated.
WHAT'S NEW
All distraction techniques provide benefit, but there’s a clear winner
In this RCT of children undergoing phlebotomy, both active and passive distraction techniques were superior to no distraction in terms of perceived pain and anxiety by the child, a health care provider, or a parent. The active-distraction group played a video game, while the passive-distraction groups watched a cartoon or interacted with a parent. Active distraction was superior to passive distraction.
CAVEATS
Procedure time was short; intervention not blinded
One potential weakness of this study is that it was not a double-blinded trial. Blinding was not possible for much of the study as the patient, parent, and medical observer were fully aware of the intervention or lack thereof. However, the parent and medical observer were blinded to each other’s assessments of the child’s pain and anxiety.
Continue to: Furthermore, the study...
Furthermore, the study was conducted at a single institution in Turkey. There could be cultural differences in reporting of pain and anxiety compared to Western cultures.
Finally, the average duration of the procedure in this study was 3 minutes, with a range of 1 to 5 minutes. It is unclear if the findings can be extrapolated to more time-consuming procedures.
CHALLENGES TO IMPLEMENTATION
Technology is not available to all
The use of tablet computers may seem increasingly ubiquitous, but not all families have access to these devices. Another challenge is that phlebotomy/clinic personnel must learn to work around the device.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Inan G, Inal S. The impact of 3 different distraction techniques on the pain and anxiety levels of children during venipuncture: a clinical trial. Clin J Pain. 2019;35:140-147.
2. Fein JA, Zempsky WT, Cravero JP, Committee on Pediatric Emergency Medicine and Section on Anesthesiology and Pain Medicine; American Academy of Pediatrics. Relief of pain and anxiety in pediatric patients in emergency medical systems. Pediatrics. 2012;130:e1391-e1405.
3. Cohen LL, Blount RL, Panopoulos G. Nurse coaching and cartoon distraction: an effective and practical intervention to reduce child, parent, and nurse distress during immunizations. J Pediatr Psychol. 1997;22:355-370.
4. Downey VA, Zun LS. The impact of watching cartoons for distraction during painful procedures in the emergency department. Pediatr Emerg. 2012;28:1033-1035.
5. Hussein H. Effect of active and passive distraction on decreasing pain associated with painful medical procedures among school aged children. World J Nurs Sci. 2015;1:13-23.
6. Nilsson S, Enskär K, Hallqvist C, et al. Active and passive distraction in children undergoing wound dressing. J Pediatr Nurs. 2013;28:158-166.
7. Birnie KA, Noel M, Chambers CT, et al. Psychological interventions for needle-related procedural pain and distress in children and adolescents. Cochrane Database Syst Rev. 2018;10:CD005179.
8. Birnie KA, Noel M, Parker JA, et al. Systematic review and meta-analysis of distraction and hypnosis for needle-related pain and distress in children and adolescents. J Pediatr Psychol. 2014;39:783-808.
9. McMurtry CM, Noel M, Chambers CT, et al. Children’s fear during procedural pain: preliminary investigation of the Children’s Fear Scale. Health Psychol. 2011;30:780-788.
10. Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatric Nurs. 1988;14:9-17.
1. Inan G, Inal S. The impact of 3 different distraction techniques on the pain and anxiety levels of children during venipuncture: a clinical trial. Clin J Pain. 2019;35:140-147.
2. Fein JA, Zempsky WT, Cravero JP, Committee on Pediatric Emergency Medicine and Section on Anesthesiology and Pain Medicine; American Academy of Pediatrics. Relief of pain and anxiety in pediatric patients in emergency medical systems. Pediatrics. 2012;130:e1391-e1405.
3. Cohen LL, Blount RL, Panopoulos G. Nurse coaching and cartoon distraction: an effective and practical intervention to reduce child, parent, and nurse distress during immunizations. J Pediatr Psychol. 1997;22:355-370.
4. Downey VA, Zun LS. The impact of watching cartoons for distraction during painful procedures in the emergency department. Pediatr Emerg. 2012;28:1033-1035.
5. Hussein H. Effect of active and passive distraction on decreasing pain associated with painful medical procedures among school aged children. World J Nurs Sci. 2015;1:13-23.
6. Nilsson S, Enskär K, Hallqvist C, et al. Active and passive distraction in children undergoing wound dressing. J Pediatr Nurs. 2013;28:158-166.
7. Birnie KA, Noel M, Chambers CT, et al. Psychological interventions for needle-related procedural pain and distress in children and adolescents. Cochrane Database Syst Rev. 2018;10:CD005179.
8. Birnie KA, Noel M, Parker JA, et al. Systematic review and meta-analysis of distraction and hypnosis for needle-related pain and distress in children and adolescents. J Pediatr Psychol. 2014;39:783-808.
9. McMurtry CM, Noel M, Chambers CT, et al. Children’s fear during procedural pain: preliminary investigation of the Children’s Fear Scale. Health Psychol. 2011;30:780-788.
10. Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatric Nurs. 1988;14:9-17.
PRACTICE CHANGER
Employ active distraction, such as playing a video game, rather than passive distraction (eg, watching a video) to reduce pain and anxiety during pediatric venipuncture.
STRENGTH OF RECOMMENDATION
B: Based on a single, high-quality, randomized controlled trial (RCT). 1
Inan G, Inal S. The impact of 3 different distraction techniques on the pain and anxiety levels of children during venipuncture: a clinical trial. Clin J Pain. 2019;35:140-147.
Tough pain relief choices in the COVID-19 pandemic
More people with fever and body aches are turning to NSAIDs to ease symptoms, but the drugs have come under new scrutiny as investigators work to determine whether they are a safe way to relieve the pain of COVID-19 vaccination or symptoms of the disease.
Early on in the pandemic, French health officials warned that NSAIDs, such as ibuprofen, could worsen coronavirus disease, and they recommended switching to acetaminophen instead.
The National Health Service in the United Kingdom followed with a similar recommendation for acetaminophen.
But the European Medicines Agency took a different approach, reporting “no scientific evidence” that NSAIDs could worsen COVID-19. The U.S. Food and Drug Administration also opted not to take a stance.
The debate prompted discussion on social media, with various reactions from around the world. It also inspired Craig Wilen, MD, PhD, from Yale University, New Haven, Conn., and associates to examine the effect of NSAIDs on COVID-19 infection and immune response. Their findings were published online Jan.20 in the Journal of Virology.
“It really bothered me that non–evidence-based decisions were driving the conversation,” Dr. Wilen said. “Millions of people are taking NSAIDs every day and clinical decisions about their care shouldn’t be made on a hypothesis.”
One theory is that NSAIDs alter susceptibility to infection by modifying ACE2. The drugs might also change the cell entry receptor for SARS-CoV-2, alter virus replication, or even modify the immune response.
British researchers, also questioning the safety of NSAIDs in patients with COVID-19, delved into National Health Service records to study two large groups of patients, some of whom were taking the pain relievers.
“We were watching the controversy and the lack of evidence and wanted to contribute,” lead investigator Angel Wong, PhD, from the London School of Hygiene and Tropical Medicine, said in an interview.
And with nearly 11 million NSAID prescriptions dispensed in primary care in England alone in the past 12 months, the inconsistency was concerning.
The team compared COVID-19–related deaths in two groups: one group of more than 700,000 people taking NSAIDs, including patients with rheumatoid arthritis and osteoarthritis; and another of almost 3.5 million people not on the medication.
NSAIDs work by inhibiting cyclooxygenase-1 and COX-2 enzymes in the body, which are crucial for the generation of prostaglandins. These lipid molecules play a role in inflammation and are blocked by NSAIDs.
The investigators found no evidence of a harmful effect of NSAIDs on COVID-19-related deaths; their results were published online Jan. 21 in the Annals of the Rheumatic Diseases.
The results, they pointed out, are in line with a Danish study that also showed no evidence of a higher risk for severe COVID-19 outcomes with NSAID use.
“It’s reassuring,” Dr. Wong said, “that patients can safely continue treatment.”
More new evidence
Dr. Wilen’s team found that SARS-CoV-2 infection stimulated COX-2 expression in human and mice cells. However, suppression of COX-2 by two commonly used NSAIDs, ibuprofen and meloxicam, had no effect on ACE2 expression, viral entry, or viral replication.
In their mouse model of SARS-CoV-2 infection, the investigators saw that NSAIDs impaired the production of proinflammatory cytokines and neutralizing antibodies. The findings suggest that NSAIDs influence COVID-19 outcomes by dampening the inflammatory response and production of protective antibodies, rather than modifying susceptibility to infection or viral replication.
Understanding the effect of NSAIDs on cytokine production is critical, Dr. Wilen pointed out, because they might be protective early in COVID-19 but pathologic at later stages.
Timing is crucial in the case of other immunomodulatory drugs. For example, dexamethasone lowers mortality in COVID-19 patients on respiratory support but is potentially harmful for those with milder disease.
There still is a lot to learn, Dr. Wilen acknowledged. “We may be seeing something similar going on with NSAIDs, where the timing of treatment is important.”
A version of this article first appeared on Medscape.com.
More people with fever and body aches are turning to NSAIDs to ease symptoms, but the drugs have come under new scrutiny as investigators work to determine whether they are a safe way to relieve the pain of COVID-19 vaccination or symptoms of the disease.
Early on in the pandemic, French health officials warned that NSAIDs, such as ibuprofen, could worsen coronavirus disease, and they recommended switching to acetaminophen instead.
The National Health Service in the United Kingdom followed with a similar recommendation for acetaminophen.
But the European Medicines Agency took a different approach, reporting “no scientific evidence” that NSAIDs could worsen COVID-19. The U.S. Food and Drug Administration also opted not to take a stance.
The debate prompted discussion on social media, with various reactions from around the world. It also inspired Craig Wilen, MD, PhD, from Yale University, New Haven, Conn., and associates to examine the effect of NSAIDs on COVID-19 infection and immune response. Their findings were published online Jan.20 in the Journal of Virology.
“It really bothered me that non–evidence-based decisions were driving the conversation,” Dr. Wilen said. “Millions of people are taking NSAIDs every day and clinical decisions about their care shouldn’t be made on a hypothesis.”
One theory is that NSAIDs alter susceptibility to infection by modifying ACE2. The drugs might also change the cell entry receptor for SARS-CoV-2, alter virus replication, or even modify the immune response.
British researchers, also questioning the safety of NSAIDs in patients with COVID-19, delved into National Health Service records to study two large groups of patients, some of whom were taking the pain relievers.
“We were watching the controversy and the lack of evidence and wanted to contribute,” lead investigator Angel Wong, PhD, from the London School of Hygiene and Tropical Medicine, said in an interview.
And with nearly 11 million NSAID prescriptions dispensed in primary care in England alone in the past 12 months, the inconsistency was concerning.
The team compared COVID-19–related deaths in two groups: one group of more than 700,000 people taking NSAIDs, including patients with rheumatoid arthritis and osteoarthritis; and another of almost 3.5 million people not on the medication.
NSAIDs work by inhibiting cyclooxygenase-1 and COX-2 enzymes in the body, which are crucial for the generation of prostaglandins. These lipid molecules play a role in inflammation and are blocked by NSAIDs.
The investigators found no evidence of a harmful effect of NSAIDs on COVID-19-related deaths; their results were published online Jan. 21 in the Annals of the Rheumatic Diseases.
The results, they pointed out, are in line with a Danish study that also showed no evidence of a higher risk for severe COVID-19 outcomes with NSAID use.
“It’s reassuring,” Dr. Wong said, “that patients can safely continue treatment.”
More new evidence
Dr. Wilen’s team found that SARS-CoV-2 infection stimulated COX-2 expression in human and mice cells. However, suppression of COX-2 by two commonly used NSAIDs, ibuprofen and meloxicam, had no effect on ACE2 expression, viral entry, or viral replication.
In their mouse model of SARS-CoV-2 infection, the investigators saw that NSAIDs impaired the production of proinflammatory cytokines and neutralizing antibodies. The findings suggest that NSAIDs influence COVID-19 outcomes by dampening the inflammatory response and production of protective antibodies, rather than modifying susceptibility to infection or viral replication.
Understanding the effect of NSAIDs on cytokine production is critical, Dr. Wilen pointed out, because they might be protective early in COVID-19 but pathologic at later stages.
Timing is crucial in the case of other immunomodulatory drugs. For example, dexamethasone lowers mortality in COVID-19 patients on respiratory support but is potentially harmful for those with milder disease.
There still is a lot to learn, Dr. Wilen acknowledged. “We may be seeing something similar going on with NSAIDs, where the timing of treatment is important.”
A version of this article first appeared on Medscape.com.
More people with fever and body aches are turning to NSAIDs to ease symptoms, but the drugs have come under new scrutiny as investigators work to determine whether they are a safe way to relieve the pain of COVID-19 vaccination or symptoms of the disease.
Early on in the pandemic, French health officials warned that NSAIDs, such as ibuprofen, could worsen coronavirus disease, and they recommended switching to acetaminophen instead.
The National Health Service in the United Kingdom followed with a similar recommendation for acetaminophen.
But the European Medicines Agency took a different approach, reporting “no scientific evidence” that NSAIDs could worsen COVID-19. The U.S. Food and Drug Administration also opted not to take a stance.
The debate prompted discussion on social media, with various reactions from around the world. It also inspired Craig Wilen, MD, PhD, from Yale University, New Haven, Conn., and associates to examine the effect of NSAIDs on COVID-19 infection and immune response. Their findings were published online Jan.20 in the Journal of Virology.
“It really bothered me that non–evidence-based decisions were driving the conversation,” Dr. Wilen said. “Millions of people are taking NSAIDs every day and clinical decisions about their care shouldn’t be made on a hypothesis.”
One theory is that NSAIDs alter susceptibility to infection by modifying ACE2. The drugs might also change the cell entry receptor for SARS-CoV-2, alter virus replication, or even modify the immune response.
British researchers, also questioning the safety of NSAIDs in patients with COVID-19, delved into National Health Service records to study two large groups of patients, some of whom were taking the pain relievers.
“We were watching the controversy and the lack of evidence and wanted to contribute,” lead investigator Angel Wong, PhD, from the London School of Hygiene and Tropical Medicine, said in an interview.
And with nearly 11 million NSAID prescriptions dispensed in primary care in England alone in the past 12 months, the inconsistency was concerning.
The team compared COVID-19–related deaths in two groups: one group of more than 700,000 people taking NSAIDs, including patients with rheumatoid arthritis and osteoarthritis; and another of almost 3.5 million people not on the medication.
NSAIDs work by inhibiting cyclooxygenase-1 and COX-2 enzymes in the body, which are crucial for the generation of prostaglandins. These lipid molecules play a role in inflammation and are blocked by NSAIDs.
The investigators found no evidence of a harmful effect of NSAIDs on COVID-19-related deaths; their results were published online Jan. 21 in the Annals of the Rheumatic Diseases.
The results, they pointed out, are in line with a Danish study that also showed no evidence of a higher risk for severe COVID-19 outcomes with NSAID use.
“It’s reassuring,” Dr. Wong said, “that patients can safely continue treatment.”
More new evidence
Dr. Wilen’s team found that SARS-CoV-2 infection stimulated COX-2 expression in human and mice cells. However, suppression of COX-2 by two commonly used NSAIDs, ibuprofen and meloxicam, had no effect on ACE2 expression, viral entry, or viral replication.
In their mouse model of SARS-CoV-2 infection, the investigators saw that NSAIDs impaired the production of proinflammatory cytokines and neutralizing antibodies. The findings suggest that NSAIDs influence COVID-19 outcomes by dampening the inflammatory response and production of protective antibodies, rather than modifying susceptibility to infection or viral replication.
Understanding the effect of NSAIDs on cytokine production is critical, Dr. Wilen pointed out, because they might be protective early in COVID-19 but pathologic at later stages.
Timing is crucial in the case of other immunomodulatory drugs. For example, dexamethasone lowers mortality in COVID-19 patients on respiratory support but is potentially harmful for those with milder disease.
There still is a lot to learn, Dr. Wilen acknowledged. “We may be seeing something similar going on with NSAIDs, where the timing of treatment is important.”
A version of this article first appeared on Medscape.com.
Pelvic pain
A 34-year-old woman with no significant past medical history presented as a new patient to our family medicine clinic with 2 weeks of intermittent lower abdominal and pelvic pain. She was sexually active with 1 partner and denied abnormal vaginal discharge or bleeding. She mentioned she’d had an intrauterine contraceptive device (IUD) placed a few weeks ago. The patient was afebrile, and her pelvic examination was unremarkable.
Physical examination showed mild tenderness to palpation over the lower abdomen without rebound tenderness or guarding. A complete metabolic panel revealed no significant abnormalities, and her human chorionic gonadotropin levels were normal.
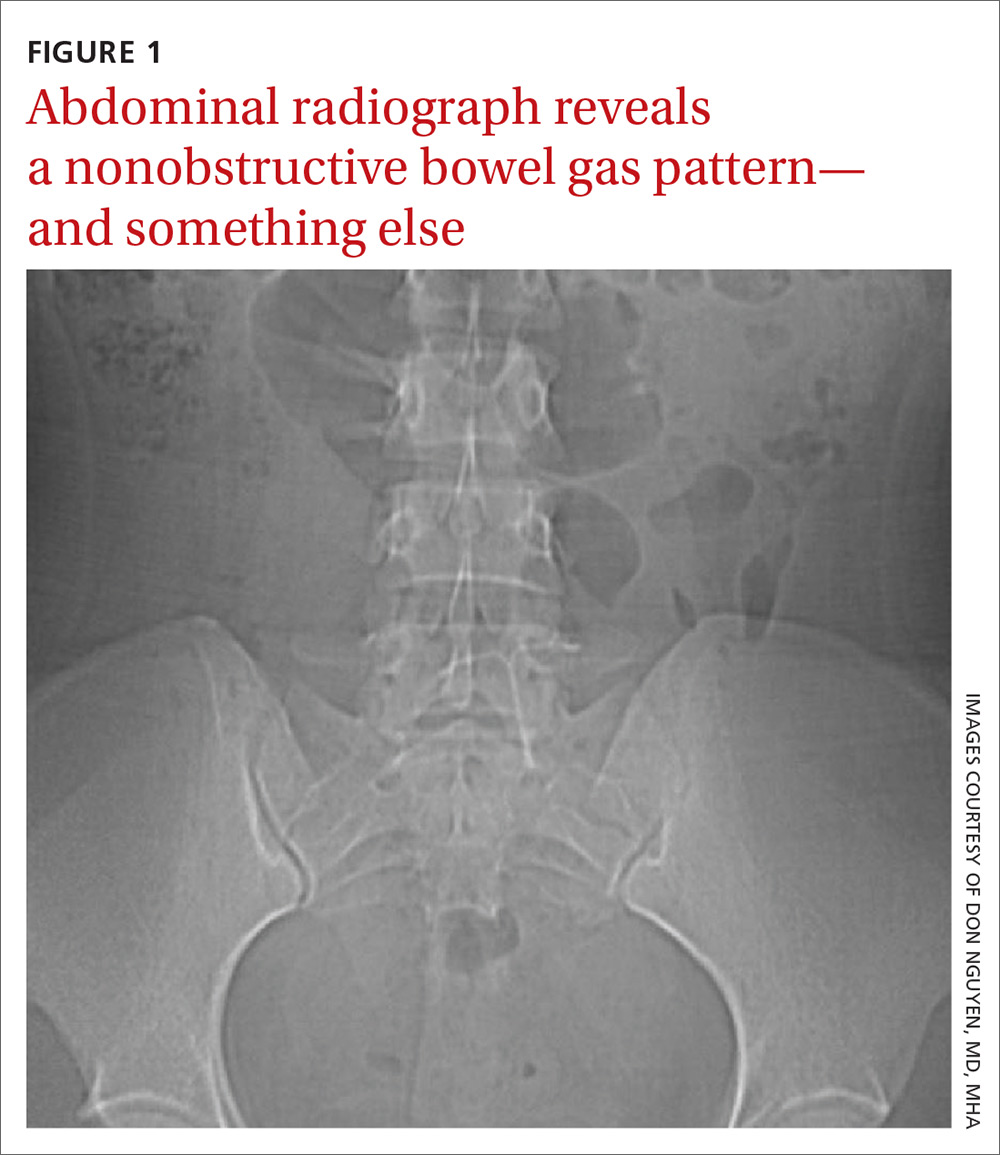
Findings from the physical exam and her clinical history prompted the need for imaging. An abdominal radiograph (FIGURE 1) and noncontrast computed tomography (FIGURES 2A and 2B) were subsequently ordered.
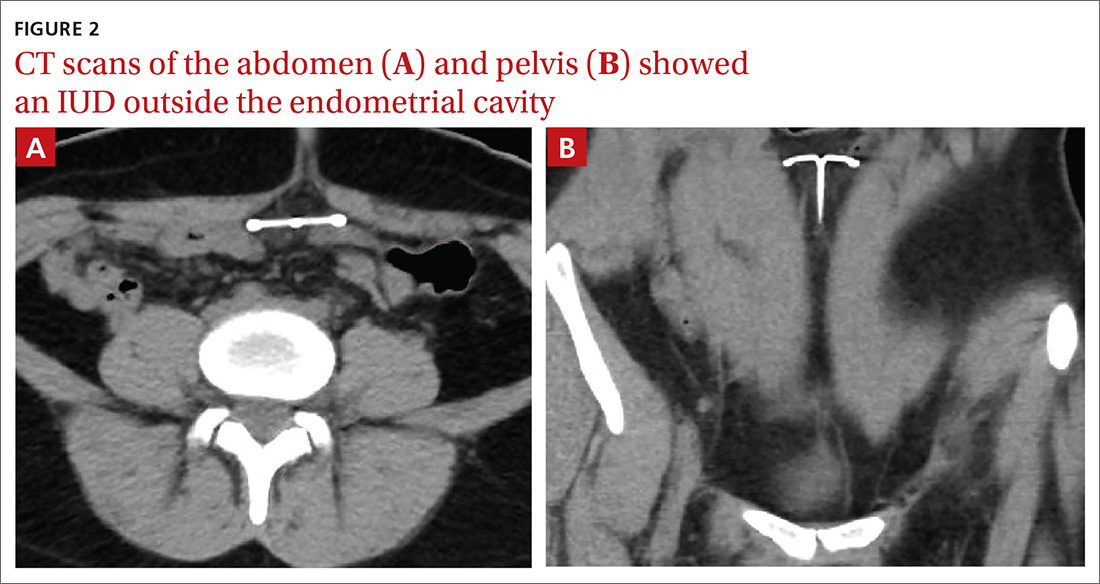
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Intra-abdominal IUD migration
The abdominal radiograph revealed a nonobstructive bowel gas pattern with an IUD overlaying the central lower abdomen and pelvis at the L5-S1 level (FIGURE 1). Computed tomography (CT) of her abdomen and pelvis showed that the IUD was outside the endometrial cavity (FIGURES 2A and 2B). There was no evidence of pneumoperitoneum or bowel perforation. Based on the work-up and imaging, the patient’s pain was due to intra-abdominal IUD malpositioning.
Diagnostic criteria for IUD malpositioning include device migration into 1 of several locations, such as the lower uterine segment or cervix. IUD malpositioning can involve the rotation or protrusion of the device into or through the myometrium. On imaging, a well-positioned IUD should have a straight stem contained within the endometrial cavity, with the arms of the IUD extending laterally at the uterine fundus.
For our patient, an abdominal radiograph showed that her IUD was superiorly displaced outside the expected region of the endometrial cavity. CT helped to confirm this.
Complications with IUDs are few
Using an IUD is an increasingly popular method of contraception because it is effective and generally well tolerated, with minimal adverse effects or complications. In a multicenter retrospective chart review of 2138 patients who had IUDs, Aoun et al found that serious complications included pelvic inflammatory disease (2%), IUD expulsion (6%), and pregnancy (1%).1 In a retrospective cohort study examining complications among 90,489 women with IUDs, Berenson et al found ectopic pregnancy and uterine perforation affected < 1%.2
A less serious complication is IUD malpositioning. Although it does seem to occur more often than other, more serious complications, the exact incidence is unknown. In a retrospective case-control study, Braaten et al reported the rate for IUD malpositioning was 10.4% among 182 women.3 Malpositioned IUDs may be more likely to occur in those with suspected adenomyosis.3 In a study by de Kroon et al, the estimated prevalence rate for an abnormal IUD position ranged from 4% to 7.7% among 195 patients.4
Continue to: The clinical presentation of IUD migration
The clinical presentation of IUD migration
Identification of a malpositioned IUD is needed to avoid the possible increased risk for uterine perforation, IUD expulsion, or pregnancy.5
IUDs that have perforated the uterus float freely in the pelvis or abdomen and can result in injury to adjacent structures as well as peritonitis, fistulas, and hemorrhage.5-7 In addition, adhesion formation over the IUD can lead to intestinal obstruction, infertility, and chronic pain.6
Common symptoms of IUD malpositioning include abdominal or pelvic pain and abnormal bleeding, although many patients may be asymptomatic.8 In a retrospective study of 167 patients with IUDs who underwent pelvic ultrasound, 28 patients were found to have an IUD in an abnormal position.8 Rates of bleeding and pain were higher in patients with malpositioned IUDs (35.7% and 39.3%, respectively) than in those with a normally positioned IUD (15.1% and 19.4%, respectively).8
The differential Dx includes endometriosis and fibroids
IUD malpositioning can be distinguished from other diagnoses that cause pelvic pain and have similar presentations—including endometriosis, ectopic pregnancy, and fibroids—through imaging study findings, clinical history, and presentation.
Other conditions that may need to be ruled out include pelvic inflammatory disease, acute appendicitis, and ovarian cysts.9 A thorough history and physical examination can help rule out these conditions by organ system, and laboratory and imaging studies can help to confirm the diagnosis.
Continue to: Which imaging tool to use, and when
Which imaging tool to use, and when
Assessment of intrauterine contraception placement requires evaluation of the uterine cavity; gynecologic examination alone is not sufficient to fully evaluate for IUD position. Certain imaging studies are particularly helpful for revealing possible IUD migration.
Ultrasound—a widely available, radiation-free modality—is the first-line imaging tool for evaluation of an IUD’s position.10 In addition, ultrasound can provide effective evaluation of other pelvic structures, which is helpful in identifying or eliminating other causes of pain or abnormal bleeding.
Conventional radiography. If the IUD is not visualized on ultrasound, the American College of Obstetricians and Gynecologists (ACOG) recommends radiography to determine if the IUD has been expelled or has migrated to an extra-uterine position.6
CT may be best suited for the evaluation of more severe complications of IUD malpositioning, including visceral perforation, abscess formation, or bowel obstruction. CT should be considered if the patient’s clinical presentation is suspicious for a more serious intra-abdominal pathology.
Management depends on the IUD’s position
For patients whose IUD has an uncertain position or nonvisualized intravaginal strings, ACOG’s first-line recommendations include ruling out pregnancy, using an alternative method for contraception, and ordering pelvic ultrasonography.6 ACOG recommendations for the management of IUD malpositioning depend on the device’s location and the patient’s symptomatology.
Continue to: Management of low-lying IUDs
Management of low-lying IUDs is complex. An IUD that is malpositioned in the cervix is considered partially expelled and should be completely removed.6 For asymptomatic patients with an IUD located in the lower uterine segment and above the internal cervical os, there should be strong consideration given to leaving the IUD in place because removal is associated with higher rates of pregnancy given the low rates of initiation of effective contraception following removal.6
IUD malpositioning in the peritoneal cavity requires surgical intervention. Although ACOG’s first-line recommendation is laparoscopic intervention, laparotomy can be considered if laparoscopy does not result in the removal of the IUD or the patient has more severe complications (sepsis or bowel perforation).6 At the time of IUD removal, the clinician should also discuss and/or prescribe interim contraception.
Treatment for our patient included uncomplicated laparoscopic surgical removal of the intra-abdominal IUD. The patient’s symptoms went away following the procedure, and she was subsequently switched to an oral contraceptive.
1. Aoun J, Dines VA, Stovall DW, et al. Effects of age, parity, and device type on complications and discontinuation of intrauterine devices. Obstet Gynecol. 2014;123:585-592.
2. Berenson AB, Tan A, Hirth JM, et al. Complications and continuation of intrauterine device use among commercially insured teenagers. Obstet Gynecol. 2013;121:951-958.
3. Braaten KP, Benson CB, Maurer R, et al. Malpositioned intrauterine contraceptive devices: risk factors, outcomes, and future pregnancies. Obstet Gynecol. 2011;118:1014-1020.
4. de Kroon CD, van Houwelingen JC, Trimbos JB, et al. The value of transvaginal ultrasound to monitor the position of an intrauterine device after insertion. A technology assessment study. Hum Reprod. 2003;18:2323-2327.
5. Thonneau P, Almont T, de La Rochebrochard E, et al. Risk factors for IUD failure: results of a large multicentre case-control study. Hum Reprod. 2006;21:2612-2616.
6. ACOG Committee on Gynecologic Practice. Committee Opinion No 672: clinical challenges of long-acting reversible contraceptive methods. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2016;128:e69-e77.
7. Heinemann K, Reed S, Moehner S, et al. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. Contraception. 2015;91:274-279.
8. Benacerraf BR, Shipp TD, Bromley B. Three-dimensional ultrasound detection of abnormally located intrauterine contraceptive devices which are a source of pelvic pain and abnormal bleeding. Ultrasound Obstet Gynecol. 2009;34:110-115.
9. Bhavasr AK, Felner EJ, Shorma T. Common questions about the evaluation of acute pelvic pain. Am Fam Physician. 2016;93:41-48.
10. Peri N, Graham D, Levine D. Imaging of intrauterine contraceptive devices. J Ultrasound Med. 2007;26:1389-1401.
A 34-year-old woman with no significant past medical history presented as a new patient to our family medicine clinic with 2 weeks of intermittent lower abdominal and pelvic pain. She was sexually active with 1 partner and denied abnormal vaginal discharge or bleeding. She mentioned she’d had an intrauterine contraceptive device (IUD) placed a few weeks ago. The patient was afebrile, and her pelvic examination was unremarkable.
Physical examination showed mild tenderness to palpation over the lower abdomen without rebound tenderness or guarding. A complete metabolic panel revealed no significant abnormalities, and her human chorionic gonadotropin levels were normal.

Findings from the physical exam and her clinical history prompted the need for imaging. An abdominal radiograph (FIGURE 1) and noncontrast computed tomography (FIGURES 2A and 2B) were subsequently ordered.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Intra-abdominal IUD migration
The abdominal radiograph revealed a nonobstructive bowel gas pattern with an IUD overlaying the central lower abdomen and pelvis at the L5-S1 level (FIGURE 1). Computed tomography (CT) of her abdomen and pelvis showed that the IUD was outside the endometrial cavity (FIGURES 2A and 2B). There was no evidence of pneumoperitoneum or bowel perforation. Based on the work-up and imaging, the patient’s pain was due to intra-abdominal IUD malpositioning.
Diagnostic criteria for IUD malpositioning include device migration into 1 of several locations, such as the lower uterine segment or cervix. IUD malpositioning can involve the rotation or protrusion of the device into or through the myometrium. On imaging, a well-positioned IUD should have a straight stem contained within the endometrial cavity, with the arms of the IUD extending laterally at the uterine fundus.
For our patient, an abdominal radiograph showed that her IUD was superiorly displaced outside the expected region of the endometrial cavity. CT helped to confirm this.
Complications with IUDs are few
Using an IUD is an increasingly popular method of contraception because it is effective and generally well tolerated, with minimal adverse effects or complications. In a multicenter retrospective chart review of 2138 patients who had IUDs, Aoun et al found that serious complications included pelvic inflammatory disease (2%), IUD expulsion (6%), and pregnancy (1%).1 In a retrospective cohort study examining complications among 90,489 women with IUDs, Berenson et al found ectopic pregnancy and uterine perforation affected < 1%.2
A less serious complication is IUD malpositioning. Although it does seem to occur more often than other, more serious complications, the exact incidence is unknown. In a retrospective case-control study, Braaten et al reported the rate for IUD malpositioning was 10.4% among 182 women.3 Malpositioned IUDs may be more likely to occur in those with suspected adenomyosis.3 In a study by de Kroon et al, the estimated prevalence rate for an abnormal IUD position ranged from 4% to 7.7% among 195 patients.4
Continue to: The clinical presentation of IUD migration
The clinical presentation of IUD migration
Identification of a malpositioned IUD is needed to avoid the possible increased risk for uterine perforation, IUD expulsion, or pregnancy.5
IUDs that have perforated the uterus float freely in the pelvis or abdomen and can result in injury to adjacent structures as well as peritonitis, fistulas, and hemorrhage.5-7 In addition, adhesion formation over the IUD can lead to intestinal obstruction, infertility, and chronic pain.6
Common symptoms of IUD malpositioning include abdominal or pelvic pain and abnormal bleeding, although many patients may be asymptomatic.8 In a retrospective study of 167 patients with IUDs who underwent pelvic ultrasound, 28 patients were found to have an IUD in an abnormal position.8 Rates of bleeding and pain were higher in patients with malpositioned IUDs (35.7% and 39.3%, respectively) than in those with a normally positioned IUD (15.1% and 19.4%, respectively).8
The differential Dx includes endometriosis and fibroids
IUD malpositioning can be distinguished from other diagnoses that cause pelvic pain and have similar presentations—including endometriosis, ectopic pregnancy, and fibroids—through imaging study findings, clinical history, and presentation.
Other conditions that may need to be ruled out include pelvic inflammatory disease, acute appendicitis, and ovarian cysts.9 A thorough history and physical examination can help rule out these conditions by organ system, and laboratory and imaging studies can help to confirm the diagnosis.
Continue to: Which imaging tool to use, and when
Which imaging tool to use, and when
Assessment of intrauterine contraception placement requires evaluation of the uterine cavity; gynecologic examination alone is not sufficient to fully evaluate for IUD position. Certain imaging studies are particularly helpful for revealing possible IUD migration.
Ultrasound—a widely available, radiation-free modality—is the first-line imaging tool for evaluation of an IUD’s position.10 In addition, ultrasound can provide effective evaluation of other pelvic structures, which is helpful in identifying or eliminating other causes of pain or abnormal bleeding.
Conventional radiography. If the IUD is not visualized on ultrasound, the American College of Obstetricians and Gynecologists (ACOG) recommends radiography to determine if the IUD has been expelled or has migrated to an extra-uterine position.6
CT may be best suited for the evaluation of more severe complications of IUD malpositioning, including visceral perforation, abscess formation, or bowel obstruction. CT should be considered if the patient’s clinical presentation is suspicious for a more serious intra-abdominal pathology.
Management depends on the IUD’s position
For patients whose IUD has an uncertain position or nonvisualized intravaginal strings, ACOG’s first-line recommendations include ruling out pregnancy, using an alternative method for contraception, and ordering pelvic ultrasonography.6 ACOG recommendations for the management of IUD malpositioning depend on the device’s location and the patient’s symptomatology.
Continue to: Management of low-lying IUDs
Management of low-lying IUDs is complex. An IUD that is malpositioned in the cervix is considered partially expelled and should be completely removed.6 For asymptomatic patients with an IUD located in the lower uterine segment and above the internal cervical os, there should be strong consideration given to leaving the IUD in place because removal is associated with higher rates of pregnancy given the low rates of initiation of effective contraception following removal.6
IUD malpositioning in the peritoneal cavity requires surgical intervention. Although ACOG’s first-line recommendation is laparoscopic intervention, laparotomy can be considered if laparoscopy does not result in the removal of the IUD or the patient has more severe complications (sepsis or bowel perforation).6 At the time of IUD removal, the clinician should also discuss and/or prescribe interim contraception.
Treatment for our patient included uncomplicated laparoscopic surgical removal of the intra-abdominal IUD. The patient’s symptoms went away following the procedure, and she was subsequently switched to an oral contraceptive.
A 34-year-old woman with no significant past medical history presented as a new patient to our family medicine clinic with 2 weeks of intermittent lower abdominal and pelvic pain. She was sexually active with 1 partner and denied abnormal vaginal discharge or bleeding. She mentioned she’d had an intrauterine contraceptive device (IUD) placed a few weeks ago. The patient was afebrile, and her pelvic examination was unremarkable.
Physical examination showed mild tenderness to palpation over the lower abdomen without rebound tenderness or guarding. A complete metabolic panel revealed no significant abnormalities, and her human chorionic gonadotropin levels were normal.

Findings from the physical exam and her clinical history prompted the need for imaging. An abdominal radiograph (FIGURE 1) and noncontrast computed tomography (FIGURES 2A and 2B) were subsequently ordered.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Intra-abdominal IUD migration
The abdominal radiograph revealed a nonobstructive bowel gas pattern with an IUD overlaying the central lower abdomen and pelvis at the L5-S1 level (FIGURE 1). Computed tomography (CT) of her abdomen and pelvis showed that the IUD was outside the endometrial cavity (FIGURES 2A and 2B). There was no evidence of pneumoperitoneum or bowel perforation. Based on the work-up and imaging, the patient’s pain was due to intra-abdominal IUD malpositioning.
Diagnostic criteria for IUD malpositioning include device migration into 1 of several locations, such as the lower uterine segment or cervix. IUD malpositioning can involve the rotation or protrusion of the device into or through the myometrium. On imaging, a well-positioned IUD should have a straight stem contained within the endometrial cavity, with the arms of the IUD extending laterally at the uterine fundus.
For our patient, an abdominal radiograph showed that her IUD was superiorly displaced outside the expected region of the endometrial cavity. CT helped to confirm this.
Complications with IUDs are few
Using an IUD is an increasingly popular method of contraception because it is effective and generally well tolerated, with minimal adverse effects or complications. In a multicenter retrospective chart review of 2138 patients who had IUDs, Aoun et al found that serious complications included pelvic inflammatory disease (2%), IUD expulsion (6%), and pregnancy (1%).1 In a retrospective cohort study examining complications among 90,489 women with IUDs, Berenson et al found ectopic pregnancy and uterine perforation affected < 1%.2
A less serious complication is IUD malpositioning. Although it does seem to occur more often than other, more serious complications, the exact incidence is unknown. In a retrospective case-control study, Braaten et al reported the rate for IUD malpositioning was 10.4% among 182 women.3 Malpositioned IUDs may be more likely to occur in those with suspected adenomyosis.3 In a study by de Kroon et al, the estimated prevalence rate for an abnormal IUD position ranged from 4% to 7.7% among 195 patients.4
Continue to: The clinical presentation of IUD migration
The clinical presentation of IUD migration
Identification of a malpositioned IUD is needed to avoid the possible increased risk for uterine perforation, IUD expulsion, or pregnancy.5
IUDs that have perforated the uterus float freely in the pelvis or abdomen and can result in injury to adjacent structures as well as peritonitis, fistulas, and hemorrhage.5-7 In addition, adhesion formation over the IUD can lead to intestinal obstruction, infertility, and chronic pain.6
Common symptoms of IUD malpositioning include abdominal or pelvic pain and abnormal bleeding, although many patients may be asymptomatic.8 In a retrospective study of 167 patients with IUDs who underwent pelvic ultrasound, 28 patients were found to have an IUD in an abnormal position.8 Rates of bleeding and pain were higher in patients with malpositioned IUDs (35.7% and 39.3%, respectively) than in those with a normally positioned IUD (15.1% and 19.4%, respectively).8
The differential Dx includes endometriosis and fibroids
IUD malpositioning can be distinguished from other diagnoses that cause pelvic pain and have similar presentations—including endometriosis, ectopic pregnancy, and fibroids—through imaging study findings, clinical history, and presentation.
Other conditions that may need to be ruled out include pelvic inflammatory disease, acute appendicitis, and ovarian cysts.9 A thorough history and physical examination can help rule out these conditions by organ system, and laboratory and imaging studies can help to confirm the diagnosis.
Continue to: Which imaging tool to use, and when
Which imaging tool to use, and when
Assessment of intrauterine contraception placement requires evaluation of the uterine cavity; gynecologic examination alone is not sufficient to fully evaluate for IUD position. Certain imaging studies are particularly helpful for revealing possible IUD migration.
Ultrasound—a widely available, radiation-free modality—is the first-line imaging tool for evaluation of an IUD’s position.10 In addition, ultrasound can provide effective evaluation of other pelvic structures, which is helpful in identifying or eliminating other causes of pain or abnormal bleeding.
Conventional radiography. If the IUD is not visualized on ultrasound, the American College of Obstetricians and Gynecologists (ACOG) recommends radiography to determine if the IUD has been expelled or has migrated to an extra-uterine position.6
CT may be best suited for the evaluation of more severe complications of IUD malpositioning, including visceral perforation, abscess formation, or bowel obstruction. CT should be considered if the patient’s clinical presentation is suspicious for a more serious intra-abdominal pathology.
Management depends on the IUD’s position
For patients whose IUD has an uncertain position or nonvisualized intravaginal strings, ACOG’s first-line recommendations include ruling out pregnancy, using an alternative method for contraception, and ordering pelvic ultrasonography.6 ACOG recommendations for the management of IUD malpositioning depend on the device’s location and the patient’s symptomatology.
Continue to: Management of low-lying IUDs
Management of low-lying IUDs is complex. An IUD that is malpositioned in the cervix is considered partially expelled and should be completely removed.6 For asymptomatic patients with an IUD located in the lower uterine segment and above the internal cervical os, there should be strong consideration given to leaving the IUD in place because removal is associated with higher rates of pregnancy given the low rates of initiation of effective contraception following removal.6
IUD malpositioning in the peritoneal cavity requires surgical intervention. Although ACOG’s first-line recommendation is laparoscopic intervention, laparotomy can be considered if laparoscopy does not result in the removal of the IUD or the patient has more severe complications (sepsis or bowel perforation).6 At the time of IUD removal, the clinician should also discuss and/or prescribe interim contraception.
Treatment for our patient included uncomplicated laparoscopic surgical removal of the intra-abdominal IUD. The patient’s symptoms went away following the procedure, and she was subsequently switched to an oral contraceptive.
1. Aoun J, Dines VA, Stovall DW, et al. Effects of age, parity, and device type on complications and discontinuation of intrauterine devices. Obstet Gynecol. 2014;123:585-592.
2. Berenson AB, Tan A, Hirth JM, et al. Complications and continuation of intrauterine device use among commercially insured teenagers. Obstet Gynecol. 2013;121:951-958.
3. Braaten KP, Benson CB, Maurer R, et al. Malpositioned intrauterine contraceptive devices: risk factors, outcomes, and future pregnancies. Obstet Gynecol. 2011;118:1014-1020.
4. de Kroon CD, van Houwelingen JC, Trimbos JB, et al. The value of transvaginal ultrasound to monitor the position of an intrauterine device after insertion. A technology assessment study. Hum Reprod. 2003;18:2323-2327.
5. Thonneau P, Almont T, de La Rochebrochard E, et al. Risk factors for IUD failure: results of a large multicentre case-control study. Hum Reprod. 2006;21:2612-2616.
6. ACOG Committee on Gynecologic Practice. Committee Opinion No 672: clinical challenges of long-acting reversible contraceptive methods. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2016;128:e69-e77.
7. Heinemann K, Reed S, Moehner S, et al. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. Contraception. 2015;91:274-279.
8. Benacerraf BR, Shipp TD, Bromley B. Three-dimensional ultrasound detection of abnormally located intrauterine contraceptive devices which are a source of pelvic pain and abnormal bleeding. Ultrasound Obstet Gynecol. 2009;34:110-115.
9. Bhavasr AK, Felner EJ, Shorma T. Common questions about the evaluation of acute pelvic pain. Am Fam Physician. 2016;93:41-48.
10. Peri N, Graham D, Levine D. Imaging of intrauterine contraceptive devices. J Ultrasound Med. 2007;26:1389-1401.
1. Aoun J, Dines VA, Stovall DW, et al. Effects of age, parity, and device type on complications and discontinuation of intrauterine devices. Obstet Gynecol. 2014;123:585-592.
2. Berenson AB, Tan A, Hirth JM, et al. Complications and continuation of intrauterine device use among commercially insured teenagers. Obstet Gynecol. 2013;121:951-958.
3. Braaten KP, Benson CB, Maurer R, et al. Malpositioned intrauterine contraceptive devices: risk factors, outcomes, and future pregnancies. Obstet Gynecol. 2011;118:1014-1020.
4. de Kroon CD, van Houwelingen JC, Trimbos JB, et al. The value of transvaginal ultrasound to monitor the position of an intrauterine device after insertion. A technology assessment study. Hum Reprod. 2003;18:2323-2327.
5. Thonneau P, Almont T, de La Rochebrochard E, et al. Risk factors for IUD failure: results of a large multicentre case-control study. Hum Reprod. 2006;21:2612-2616.
6. ACOG Committee on Gynecologic Practice. Committee Opinion No 672: clinical challenges of long-acting reversible contraceptive methods. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2016;128:e69-e77.
7. Heinemann K, Reed S, Moehner S, et al. Risk of uterine perforation with levonorgestrel-releasing and copper intrauterine devices in the European Active Surveillance Study on Intrauterine Devices. Contraception. 2015;91:274-279.
8. Benacerraf BR, Shipp TD, Bromley B. Three-dimensional ultrasound detection of abnormally located intrauterine contraceptive devices which are a source of pelvic pain and abnormal bleeding. Ultrasound Obstet Gynecol. 2009;34:110-115.
9. Bhavasr AK, Felner EJ, Shorma T. Common questions about the evaluation of acute pelvic pain. Am Fam Physician. 2016;93:41-48.
10. Peri N, Graham D, Levine D. Imaging of intrauterine contraceptive devices. J Ultrasound Med. 2007;26:1389-1401.
60-year-old man • chronic cough • history of GERD & dyslipidemia • throat tickle • Dx?
THE CASE
A 60-year-old man with a past medical history of gastroesophageal reflux disease (GERD) and dyslipidemia presented to his family physician for evaluation of chronic cough. Five years prior, the patient had developed a high fever and respiratory symptoms, including a cough, and was believed to have had severe otitis media. He was treated with multiple courses of antibiotics and corticosteroids for persistent otitis media. Although the condition eventually resolved, his cough continued.
The persistent cough prompted the patient to consult a succession of specialists. First, he saw a gastroenterologist; following an esophagogastroduodenoscopy, he was prescribed pantoprazole. Despite the proton-pump inhibitor (PPI) therapy, the cough remained. Next, he had multiple visits with an otolaryngologist but that yielded no specific diagnosis for the cough. He also saw an allergist-immunologist, who identified a ragweed allergy, gave him a diagnosis of cough-variant asthma, and prescribed antihistamines and mometasone furoate and formoterol fumarate dihydrate. Neither was helpful.
After 5 years of frustration, the patient complained to his family physician that he still had a cough and “a tickle” in his throat that was worsened by speaking and drinking cold beverages. He denied fever, shortness of breath, nausea, vomiting, or any other associated symptoms.
THE DIAGNOSIS
The failed treatment attempts with antihistamines, corticosteroids, bronchodilators, and PPI therapy excluded multiple etiologies for the cough. The throat discomfort and feeling of a “tickle” prompted us to consider a nerve-related disorder on the differential. The diagnosis of laryngeal sensory neuropathy (LSN) was considered.
DISCUSSION
LSN is a relatively uncommon cause of chronic refractory cough that can also manifest with throat discomfort, dysphagia, and dysphonia.1 It is thought to result from some type of insult to the recurrent laryngeal nerve or superior laryngeal nerve via viral infections, metabolic changes, or mechanical trauma, leading to a change in the firing threshold.2 The hypothesis of nerve damage is supported by the increased incidence of LSN in patients with goiters and those with type 2 diabetes.3,4 When there is a decrease in the laryngeal sensory threshold, dysfunctional laryngeal behavior results, leading to symptoms such as persistent cough and throat clearing.
Diagnosis. LSN is often diagnosed clinically, after GERD, allergies, asthma, angiotensin-converting enzyme inhibitor intake, and psychogenic disorders have been ruled out.1 Our patient had a prior diagnosis or investigation of nearly all of these conditions. Other clues pointing to an LSN diagnosis include a cough lasting 8 weeks or more, recurrent sensory disturbances (such as a tickle) of instantaneous onset before each cough episode, triggers that can include talking or a change in air temperature, daily coughing episodes numbering in the 10s to 100s, and a nonproductive cough.5,6
Beyond clinical clues, laryngeal electromyography, which evaluates the neuromuscular system in the larynx by recording action potentials generated in the laryngeal muscles during contraction, can be used for diagnosis.4 Videostroboscopy, which allows for an enlarged and slow motion view of the vocal cords, can also be used.
Continue to: Treatment
Treatment. To both confirm the diagnosis and treat the patient in a rapid, practical fashion, a trial of a neuromodulating agent such as pregabalin or gabapentin can be employed.6-9 A study identifying 28 LSN patients found symptomatic relief in 68% of patients taking gabapentin 100 to 900 mg/d.2 In another study, 12 LSN patients given pregabalin found relief after a 1-month regimen.1 Another study of 12 patients showed amitriptyline hydrochloride and gabapentin provided a positive response in 2 months, and the addition of reflux precautions and acid-suppression therapy was helpful.9 Finally, a group of 32 patients trialed on 3 different medications (amitriptyline, desipramine, and gabapentin) found similar efficacy among the 3.6
Another option. Aside from medications, botulinum toxin type A has been shown in a case series to directly decrease laryngeal hypertonicity and possibly reduce neurogenic inflammation and neuropeptide-mediated cough.10 Another study found that 18 patients with neurogenic cough who received superior laryngeal nerve blocks had cough severity index scores decrease from an average of 26.8 pretreatment to 14.6 posttreatment (P < .0001).11
Our patient agreed to a trial of gabapentin 300 mg once a day, with titration up to a maximum of 900 mg tid. When the patient returned to the clinic 4 months later, he reported that when he reached 300 mg bid, the cough completely resolved.
THE TAKEAWAY
A persistent cough with minimal identifiable triggers is a huge disruption to a patient’s life; having to visit multiple specialists before receiving a diagnosis compounds that. In our patient’s case, the process took 5 years, which underscores how important it is that LSN be considered in the differential diagnosis. Since this is generally a diagnosis of exclusion, it is important to take a careful history of a patient with a chronic cough. If LSN seems likely, trialing a patient on neuromodulating medication is the next best step, with dose titration if necessary.
Selena R. Pasadyn, 675 West 130th Street, Hinckley, OH, 44233; [email protected]
1. Halum SL, Sycamore DL, McRae BR. A new treatment option for laryngeal sensory neuropathy. Laryngoscope. 2009;119:1844-1847.
2. Lee B, Woo P. Chronic cough as a sign of laryngeal sensory neuropathy: diagnosis and treatment. Ann Otol Rhinol Laryngol. 2005;114:253-257.
3. Hamdan AL, Jabour J, Azar ST. Goiter and laryngeal sensory neuropathy. Int J Otolaryngol. 2013;2013:765265.
4. Hamdan AL, Dowli A, Barazi R, et al. Laryngeal sensory neuropathy in patients with diabetes mellitus. J Laryngol Otol. 2014;128:725-729.
5. Bastian RW, Vaidya AM, Delsupehe KG. Sensory neuropathic cough: a common and treatable cause of chronic cough. Otolaryngol Head Neck Surg. 2006;135:17-21.
6. Bastian ZJ, Bastian RW. The use of neuralgia medications to treat sensory neuropathic cough: our experience in a retrospective cohort of thirty-two patients. PeerJ. 2015;3:e816.
7. Van de Kerkhove C, Goeminne PC, Van Bleyenbergh P, et al. A cohort description and analysis of the effect of gabapentin on idiopathic cough. Cough. 2012;8:9.
8. Mishriki YY. Laryngeal neuropathy as a cause of chronic intractable cough. Am J Med. 2007;120:e5.
9. Norris BK, Schweinfurth JM. Management of recurrent laryngeal sensory neuropathic symptoms. Ann Otol Rhinol Laryngol. 2010;119:188-191.
10. Chu MW, Lieser JD, Sinacori JT. Use of botulinum toxin type a for chronic cough: a neuropathic model. Arch Otolaryngol Head Neck Surg. 2010;136:447.
11. Simpson CB, Tibbetts KM, Loochtan MJ, et al. Treatment of chronic neurogenic cough with in-office superior laryngeal nerve block. Laryngoscope. 2018;128:1898-1903.
THE CASE
A 60-year-old man with a past medical history of gastroesophageal reflux disease (GERD) and dyslipidemia presented to his family physician for evaluation of chronic cough. Five years prior, the patient had developed a high fever and respiratory symptoms, including a cough, and was believed to have had severe otitis media. He was treated with multiple courses of antibiotics and corticosteroids for persistent otitis media. Although the condition eventually resolved, his cough continued.
The persistent cough prompted the patient to consult a succession of specialists. First, he saw a gastroenterologist; following an esophagogastroduodenoscopy, he was prescribed pantoprazole. Despite the proton-pump inhibitor (PPI) therapy, the cough remained. Next, he had multiple visits with an otolaryngologist but that yielded no specific diagnosis for the cough. He also saw an allergist-immunologist, who identified a ragweed allergy, gave him a diagnosis of cough-variant asthma, and prescribed antihistamines and mometasone furoate and formoterol fumarate dihydrate. Neither was helpful.
After 5 years of frustration, the patient complained to his family physician that he still had a cough and “a tickle” in his throat that was worsened by speaking and drinking cold beverages. He denied fever, shortness of breath, nausea, vomiting, or any other associated symptoms.
THE DIAGNOSIS
The failed treatment attempts with antihistamines, corticosteroids, bronchodilators, and PPI therapy excluded multiple etiologies for the cough. The throat discomfort and feeling of a “tickle” prompted us to consider a nerve-related disorder on the differential. The diagnosis of laryngeal sensory neuropathy (LSN) was considered.
DISCUSSION
LSN is a relatively uncommon cause of chronic refractory cough that can also manifest with throat discomfort, dysphagia, and dysphonia.1 It is thought to result from some type of insult to the recurrent laryngeal nerve or superior laryngeal nerve via viral infections, metabolic changes, or mechanical trauma, leading to a change in the firing threshold.2 The hypothesis of nerve damage is supported by the increased incidence of LSN in patients with goiters and those with type 2 diabetes.3,4 When there is a decrease in the laryngeal sensory threshold, dysfunctional laryngeal behavior results, leading to symptoms such as persistent cough and throat clearing.
Diagnosis. LSN is often diagnosed clinically, after GERD, allergies, asthma, angiotensin-converting enzyme inhibitor intake, and psychogenic disorders have been ruled out.1 Our patient had a prior diagnosis or investigation of nearly all of these conditions. Other clues pointing to an LSN diagnosis include a cough lasting 8 weeks or more, recurrent sensory disturbances (such as a tickle) of instantaneous onset before each cough episode, triggers that can include talking or a change in air temperature, daily coughing episodes numbering in the 10s to 100s, and a nonproductive cough.5,6
Beyond clinical clues, laryngeal electromyography, which evaluates the neuromuscular system in the larynx by recording action potentials generated in the laryngeal muscles during contraction, can be used for diagnosis.4 Videostroboscopy, which allows for an enlarged and slow motion view of the vocal cords, can also be used.
Continue to: Treatment
Treatment. To both confirm the diagnosis and treat the patient in a rapid, practical fashion, a trial of a neuromodulating agent such as pregabalin or gabapentin can be employed.6-9 A study identifying 28 LSN patients found symptomatic relief in 68% of patients taking gabapentin 100 to 900 mg/d.2 In another study, 12 LSN patients given pregabalin found relief after a 1-month regimen.1 Another study of 12 patients showed amitriptyline hydrochloride and gabapentin provided a positive response in 2 months, and the addition of reflux precautions and acid-suppression therapy was helpful.9 Finally, a group of 32 patients trialed on 3 different medications (amitriptyline, desipramine, and gabapentin) found similar efficacy among the 3.6
Another option. Aside from medications, botulinum toxin type A has been shown in a case series to directly decrease laryngeal hypertonicity and possibly reduce neurogenic inflammation and neuropeptide-mediated cough.10 Another study found that 18 patients with neurogenic cough who received superior laryngeal nerve blocks had cough severity index scores decrease from an average of 26.8 pretreatment to 14.6 posttreatment (P < .0001).11
Our patient agreed to a trial of gabapentin 300 mg once a day, with titration up to a maximum of 900 mg tid. When the patient returned to the clinic 4 months later, he reported that when he reached 300 mg bid, the cough completely resolved.
THE TAKEAWAY
A persistent cough with minimal identifiable triggers is a huge disruption to a patient’s life; having to visit multiple specialists before receiving a diagnosis compounds that. In our patient’s case, the process took 5 years, which underscores how important it is that LSN be considered in the differential diagnosis. Since this is generally a diagnosis of exclusion, it is important to take a careful history of a patient with a chronic cough. If LSN seems likely, trialing a patient on neuromodulating medication is the next best step, with dose titration if necessary.
Selena R. Pasadyn, 675 West 130th Street, Hinckley, OH, 44233; [email protected]
THE CASE
A 60-year-old man with a past medical history of gastroesophageal reflux disease (GERD) and dyslipidemia presented to his family physician for evaluation of chronic cough. Five years prior, the patient had developed a high fever and respiratory symptoms, including a cough, and was believed to have had severe otitis media. He was treated with multiple courses of antibiotics and corticosteroids for persistent otitis media. Although the condition eventually resolved, his cough continued.
The persistent cough prompted the patient to consult a succession of specialists. First, he saw a gastroenterologist; following an esophagogastroduodenoscopy, he was prescribed pantoprazole. Despite the proton-pump inhibitor (PPI) therapy, the cough remained. Next, he had multiple visits with an otolaryngologist but that yielded no specific diagnosis for the cough. He also saw an allergist-immunologist, who identified a ragweed allergy, gave him a diagnosis of cough-variant asthma, and prescribed antihistamines and mometasone furoate and formoterol fumarate dihydrate. Neither was helpful.
After 5 years of frustration, the patient complained to his family physician that he still had a cough and “a tickle” in his throat that was worsened by speaking and drinking cold beverages. He denied fever, shortness of breath, nausea, vomiting, or any other associated symptoms.
THE DIAGNOSIS
The failed treatment attempts with antihistamines, corticosteroids, bronchodilators, and PPI therapy excluded multiple etiologies for the cough. The throat discomfort and feeling of a “tickle” prompted us to consider a nerve-related disorder on the differential. The diagnosis of laryngeal sensory neuropathy (LSN) was considered.
DISCUSSION
LSN is a relatively uncommon cause of chronic refractory cough that can also manifest with throat discomfort, dysphagia, and dysphonia.1 It is thought to result from some type of insult to the recurrent laryngeal nerve or superior laryngeal nerve via viral infections, metabolic changes, or mechanical trauma, leading to a change in the firing threshold.2 The hypothesis of nerve damage is supported by the increased incidence of LSN in patients with goiters and those with type 2 diabetes.3,4 When there is a decrease in the laryngeal sensory threshold, dysfunctional laryngeal behavior results, leading to symptoms such as persistent cough and throat clearing.
Diagnosis. LSN is often diagnosed clinically, after GERD, allergies, asthma, angiotensin-converting enzyme inhibitor intake, and psychogenic disorders have been ruled out.1 Our patient had a prior diagnosis or investigation of nearly all of these conditions. Other clues pointing to an LSN diagnosis include a cough lasting 8 weeks or more, recurrent sensory disturbances (such as a tickle) of instantaneous onset before each cough episode, triggers that can include talking or a change in air temperature, daily coughing episodes numbering in the 10s to 100s, and a nonproductive cough.5,6
Beyond clinical clues, laryngeal electromyography, which evaluates the neuromuscular system in the larynx by recording action potentials generated in the laryngeal muscles during contraction, can be used for diagnosis.4 Videostroboscopy, which allows for an enlarged and slow motion view of the vocal cords, can also be used.
Continue to: Treatment
Treatment. To both confirm the diagnosis and treat the patient in a rapid, practical fashion, a trial of a neuromodulating agent such as pregabalin or gabapentin can be employed.6-9 A study identifying 28 LSN patients found symptomatic relief in 68% of patients taking gabapentin 100 to 900 mg/d.2 In another study, 12 LSN patients given pregabalin found relief after a 1-month regimen.1 Another study of 12 patients showed amitriptyline hydrochloride and gabapentin provided a positive response in 2 months, and the addition of reflux precautions and acid-suppression therapy was helpful.9 Finally, a group of 32 patients trialed on 3 different medications (amitriptyline, desipramine, and gabapentin) found similar efficacy among the 3.6
Another option. Aside from medications, botulinum toxin type A has been shown in a case series to directly decrease laryngeal hypertonicity and possibly reduce neurogenic inflammation and neuropeptide-mediated cough.10 Another study found that 18 patients with neurogenic cough who received superior laryngeal nerve blocks had cough severity index scores decrease from an average of 26.8 pretreatment to 14.6 posttreatment (P < .0001).11
Our patient agreed to a trial of gabapentin 300 mg once a day, with titration up to a maximum of 900 mg tid. When the patient returned to the clinic 4 months later, he reported that when he reached 300 mg bid, the cough completely resolved.
THE TAKEAWAY
A persistent cough with minimal identifiable triggers is a huge disruption to a patient’s life; having to visit multiple specialists before receiving a diagnosis compounds that. In our patient’s case, the process took 5 years, which underscores how important it is that LSN be considered in the differential diagnosis. Since this is generally a diagnosis of exclusion, it is important to take a careful history of a patient with a chronic cough. If LSN seems likely, trialing a patient on neuromodulating medication is the next best step, with dose titration if necessary.
Selena R. Pasadyn, 675 West 130th Street, Hinckley, OH, 44233; [email protected]
1. Halum SL, Sycamore DL, McRae BR. A new treatment option for laryngeal sensory neuropathy. Laryngoscope. 2009;119:1844-1847.
2. Lee B, Woo P. Chronic cough as a sign of laryngeal sensory neuropathy: diagnosis and treatment. Ann Otol Rhinol Laryngol. 2005;114:253-257.
3. Hamdan AL, Jabour J, Azar ST. Goiter and laryngeal sensory neuropathy. Int J Otolaryngol. 2013;2013:765265.
4. Hamdan AL, Dowli A, Barazi R, et al. Laryngeal sensory neuropathy in patients with diabetes mellitus. J Laryngol Otol. 2014;128:725-729.
5. Bastian RW, Vaidya AM, Delsupehe KG. Sensory neuropathic cough: a common and treatable cause of chronic cough. Otolaryngol Head Neck Surg. 2006;135:17-21.
6. Bastian ZJ, Bastian RW. The use of neuralgia medications to treat sensory neuropathic cough: our experience in a retrospective cohort of thirty-two patients. PeerJ. 2015;3:e816.
7. Van de Kerkhove C, Goeminne PC, Van Bleyenbergh P, et al. A cohort description and analysis of the effect of gabapentin on idiopathic cough. Cough. 2012;8:9.
8. Mishriki YY. Laryngeal neuropathy as a cause of chronic intractable cough. Am J Med. 2007;120:e5.
9. Norris BK, Schweinfurth JM. Management of recurrent laryngeal sensory neuropathic symptoms. Ann Otol Rhinol Laryngol. 2010;119:188-191.
10. Chu MW, Lieser JD, Sinacori JT. Use of botulinum toxin type a for chronic cough: a neuropathic model. Arch Otolaryngol Head Neck Surg. 2010;136:447.
11. Simpson CB, Tibbetts KM, Loochtan MJ, et al. Treatment of chronic neurogenic cough with in-office superior laryngeal nerve block. Laryngoscope. 2018;128:1898-1903.
1. Halum SL, Sycamore DL, McRae BR. A new treatment option for laryngeal sensory neuropathy. Laryngoscope. 2009;119:1844-1847.
2. Lee B, Woo P. Chronic cough as a sign of laryngeal sensory neuropathy: diagnosis and treatment. Ann Otol Rhinol Laryngol. 2005;114:253-257.
3. Hamdan AL, Jabour J, Azar ST. Goiter and laryngeal sensory neuropathy. Int J Otolaryngol. 2013;2013:765265.
4. Hamdan AL, Dowli A, Barazi R, et al. Laryngeal sensory neuropathy in patients with diabetes mellitus. J Laryngol Otol. 2014;128:725-729.
5. Bastian RW, Vaidya AM, Delsupehe KG. Sensory neuropathic cough: a common and treatable cause of chronic cough. Otolaryngol Head Neck Surg. 2006;135:17-21.
6. Bastian ZJ, Bastian RW. The use of neuralgia medications to treat sensory neuropathic cough: our experience in a retrospective cohort of thirty-two patients. PeerJ. 2015;3:e816.
7. Van de Kerkhove C, Goeminne PC, Van Bleyenbergh P, et al. A cohort description and analysis of the effect of gabapentin on idiopathic cough. Cough. 2012;8:9.
8. Mishriki YY. Laryngeal neuropathy as a cause of chronic intractable cough. Am J Med. 2007;120:e5.
9. Norris BK, Schweinfurth JM. Management of recurrent laryngeal sensory neuropathic symptoms. Ann Otol Rhinol Laryngol. 2010;119:188-191.
10. Chu MW, Lieser JD, Sinacori JT. Use of botulinum toxin type a for chronic cough: a neuropathic model. Arch Otolaryngol Head Neck Surg. 2010;136:447.
11. Simpson CB, Tibbetts KM, Loochtan MJ, et al. Treatment of chronic neurogenic cough with in-office superior laryngeal nerve block. Laryngoscope. 2018;128:1898-1903.
20-year-old man • sudden-onset chest pain • worsening pain with cough and exertion • Dx?
THE CASE
A 20-year-old man presented to our clinic with a 3-day history of nonradiating chest pain located at the center of his chest. Past medical history included idiopathic neonatal giant-cell hepatitis and subsequent liver transplant at 1 month of age; he had been followed by the transplant team without rejection or infection and was in otherwise good health prior to the chest pain.
On the day of symptom onset, he was walking inside his house and fell to his knees with a chest pain described as “a punch” to the center of the chest that lasted for a few seconds. He was able to continue his daily activities without limitation despite a constant, squeezing, centrally located chest pain. The pain worsened with cough and exertion.
A few hours later, he went to an urgent care center for evaluation. There, he reported, his chest radiograph and electrocardiogram (EKG) results were normal and he was given a diagnosis of musculoskeletal chest pain. Over the next 3 days, his chest pain persisted but did not worsen. He was taking 500 mg of naproxen every 8 hours with no improvement. No other acute or chronic medications were being taken. He had no significant family history. A review of systems was otherwise negative.
On physical exam, his vital statistics included a height of 6’4”; weight, 261 lb; body mass index, 31.8; temperature, 98.7 °F; blood pressure, 134/77 mm Hg; heart rate, 92 beats/min; respiratory rate, 18 breaths/min; and oxygen saturation, 96%. Throughout the exam, he demonstrated no acute distress, appeared well, and was talkative; however, he reported having a “constant, squeezing” chest pain that did not worsen with palpation of the chest. The rest of his physical exam was unremarkable.
Although he reported that his EKG and chest radiograph were normal 3 days prior, repeat chest radiograph and EKG were ordered due to his unexplained, active chest pain and the lack of immediate access to the prior results.
THE DIAGNOSIS
The chest radiograph (FIGURE 1A) showed a “mildly ectatic ascending thoracic aorta” that had increased since a chest radiograph from 6 years prior (FIGURE 1B) and “was concerning for an aneurysm.” Computed tomography (CT) angiography (FIGURE 2) then confirmed a 7-cm aneurysm of the ascending aorta, with findings suggestive of a retrograde ascending aortic dissection.
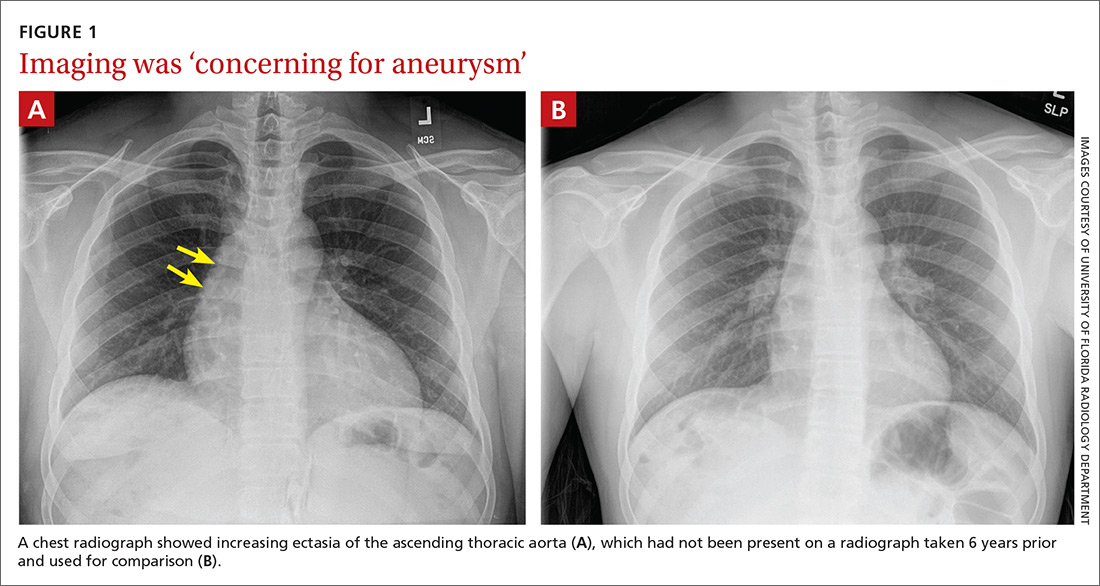
DISCUSSION
The average age of a patient with acute aortic dissection (AAD) is 63 years; only 7% occur in people younger than 40.1 AAD is often accompanied by a predisposing risk factor such as a connective tissue disease, bicuspid aortic valve, longstanding hypertension, trauma, or larger aortic dimensions.2,3 Younger patients are more likely to have predisposing risk factors of Marfan syndrome, prior aortic surgery, or a bicuspid aortic valve.3
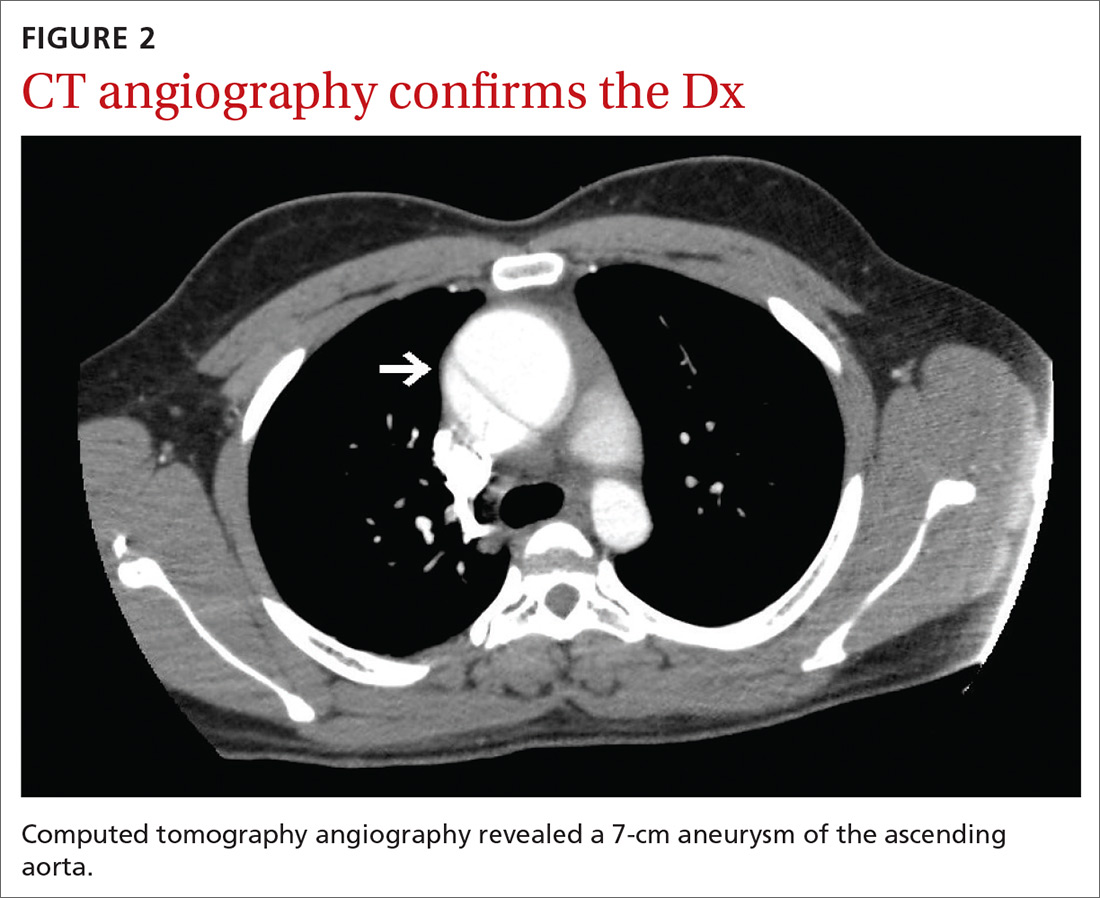
Continue to: A literature review did not reveal...
A literature review did not reveal any known correlation between the patient’s history of giant-cell hepatitis or antirejection therapy with thoracic aortic dissection. Furthermore, liver transplant is not known to be a specific risk factor for AAD in pediatric patients or outside the immediate postoperative period. Therefore, there were no known predisposing risk factors for AAD in our patient.
The most common clinical feature of AAD is chest pain, which occurs in 75% of patients.1 Other clinical symptoms include hypertension and diaphoresis.2,4 However, classic clinical findings are not always displayed, making the diagnosis difficult.2,4 The classical description of “tearing pain” is seen in only 51% of patients, and 5% to 15% of patients present without any pain.1
Commonly missed or misdiagnosed. The diagnosis of AAD has been missed during the initial exam in 38% of patients.4 As seen in our case, symptoms may be initially diagnosed as musculoskeletal chest pain. Based on symptoms, AAD can be incorrectly diagnosed as an acute myocardial infarction or vascular embolization.2,4
Every hour after symptom onset, the mortality rate of untreated AAD increases 1% to 2%,with no difference based on age.3,4 Different reports have shown mortality rates between 7% and 30%.4
Effective imaging is crucial to the diagnosis and treatment of AAD, given the occurrence of atypical presentation, missed diagnosis, and high mortality rate.4 A chest radiograph will show a widened mediastinum, but the preferred diagnostic tests are a CT or transthoracic echocardiogram.2,4 Once the diagnosis of AAD is confirmed, an aortic angiogram is the preferred test to determine the extent of the dissection prior to surgical treatment.2
Continue to: Classification dictates treatment
Classification dictates treatment. AAD is classified based on where the dissection of the aorta occurs. If the dissection involves the ascending aorta, it is classified as a type A AAD and should immediately be treated with emergent surgery in order to prevent complications including myocardial infarction, cardiac tamponade, and aortic rupture.2,4,5 If the dissection is limited to the descending aorta, it is classified as a type B AAD and can be medically managed by controlling pain and lowering blood pressure; if symptoms persist, surgical management may be required.2 After hospital discharge, AAD patients are followed closely with medical therapy, serial imaging, and reoperation if necessary.4
Our patient underwent emergent surgery for aortic root/ascending aortic replacement with a mechanical valve. He tolerated the procedure well. Surgical tissue pathology of the aortic segment showed a wall of elastic vessel with medial degeneration and dissection, and the tissue pathology of the aorta leaflets showed valvular tissue with myxoid degeneration.
THE TAKEAWAY
It is critical to keep AAD in the differential diagnosis of a patient presenting with acute onset of chest pain, as AAD often has an atypical presentation and can easily be misdiagnosed. Effective imaging is crucial to diagnosis, and immediate treatment is essential to patient survival.
CORRESPONDENCE
Rachel A. Reedy, PA, University of Florida, Department of General Pediatrics, 7046 SW Archer Road, Gainesville, FL 32608; [email protected]
1. Pineault J, Ouimet D, Pichette V, Vallée M. A case of aortic dissection in a young adult: a refresher of the literature of this “great masquerader.” Int J Gen Med. 2011;4:889-893.
2. Agabegi SS, Agabegi ElD, Ring AC. Diseases of the cardiovascular system. In: Jackson A, ed. Step-up to Medicine. 3rd ed. Lippincott Williams & Wilkins; 2012:54-55.
3. Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol. 2004;43:665-669.
4. Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD). Eur J Vasc Endovasc Surg. 2009;37:149-159.
5. Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: Report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg. 2010;140:784-789.
THE CASE
A 20-year-old man presented to our clinic with a 3-day history of nonradiating chest pain located at the center of his chest. Past medical history included idiopathic neonatal giant-cell hepatitis and subsequent liver transplant at 1 month of age; he had been followed by the transplant team without rejection or infection and was in otherwise good health prior to the chest pain.
On the day of symptom onset, he was walking inside his house and fell to his knees with a chest pain described as “a punch” to the center of the chest that lasted for a few seconds. He was able to continue his daily activities without limitation despite a constant, squeezing, centrally located chest pain. The pain worsened with cough and exertion.
A few hours later, he went to an urgent care center for evaluation. There, he reported, his chest radiograph and electrocardiogram (EKG) results were normal and he was given a diagnosis of musculoskeletal chest pain. Over the next 3 days, his chest pain persisted but did not worsen. He was taking 500 mg of naproxen every 8 hours with no improvement. No other acute or chronic medications were being taken. He had no significant family history. A review of systems was otherwise negative.
On physical exam, his vital statistics included a height of 6’4”; weight, 261 lb; body mass index, 31.8; temperature, 98.7 °F; blood pressure, 134/77 mm Hg; heart rate, 92 beats/min; respiratory rate, 18 breaths/min; and oxygen saturation, 96%. Throughout the exam, he demonstrated no acute distress, appeared well, and was talkative; however, he reported having a “constant, squeezing” chest pain that did not worsen with palpation of the chest. The rest of his physical exam was unremarkable.
Although he reported that his EKG and chest radiograph were normal 3 days prior, repeat chest radiograph and EKG were ordered due to his unexplained, active chest pain and the lack of immediate access to the prior results.
THE DIAGNOSIS
The chest radiograph (FIGURE 1A) showed a “mildly ectatic ascending thoracic aorta” that had increased since a chest radiograph from 6 years prior (FIGURE 1B) and “was concerning for an aneurysm.” Computed tomography (CT) angiography (FIGURE 2) then confirmed a 7-cm aneurysm of the ascending aorta, with findings suggestive of a retrograde ascending aortic dissection.

DISCUSSION
The average age of a patient with acute aortic dissection (AAD) is 63 years; only 7% occur in people younger than 40.1 AAD is often accompanied by a predisposing risk factor such as a connective tissue disease, bicuspid aortic valve, longstanding hypertension, trauma, or larger aortic dimensions.2,3 Younger patients are more likely to have predisposing risk factors of Marfan syndrome, prior aortic surgery, or a bicuspid aortic valve.3

Continue to: A literature review did not reveal...
A literature review did not reveal any known correlation between the patient’s history of giant-cell hepatitis or antirejection therapy with thoracic aortic dissection. Furthermore, liver transplant is not known to be a specific risk factor for AAD in pediatric patients or outside the immediate postoperative period. Therefore, there were no known predisposing risk factors for AAD in our patient.
The most common clinical feature of AAD is chest pain, which occurs in 75% of patients.1 Other clinical symptoms include hypertension and diaphoresis.2,4 However, classic clinical findings are not always displayed, making the diagnosis difficult.2,4 The classical description of “tearing pain” is seen in only 51% of patients, and 5% to 15% of patients present without any pain.1
Commonly missed or misdiagnosed. The diagnosis of AAD has been missed during the initial exam in 38% of patients.4 As seen in our case, symptoms may be initially diagnosed as musculoskeletal chest pain. Based on symptoms, AAD can be incorrectly diagnosed as an acute myocardial infarction or vascular embolization.2,4
Every hour after symptom onset, the mortality rate of untreated AAD increases 1% to 2%,with no difference based on age.3,4 Different reports have shown mortality rates between 7% and 30%.4
Effective imaging is crucial to the diagnosis and treatment of AAD, given the occurrence of atypical presentation, missed diagnosis, and high mortality rate.4 A chest radiograph will show a widened mediastinum, but the preferred diagnostic tests are a CT or transthoracic echocardiogram.2,4 Once the diagnosis of AAD is confirmed, an aortic angiogram is the preferred test to determine the extent of the dissection prior to surgical treatment.2
Continue to: Classification dictates treatment
Classification dictates treatment. AAD is classified based on where the dissection of the aorta occurs. If the dissection involves the ascending aorta, it is classified as a type A AAD and should immediately be treated with emergent surgery in order to prevent complications including myocardial infarction, cardiac tamponade, and aortic rupture.2,4,5 If the dissection is limited to the descending aorta, it is classified as a type B AAD and can be medically managed by controlling pain and lowering blood pressure; if symptoms persist, surgical management may be required.2 After hospital discharge, AAD patients are followed closely with medical therapy, serial imaging, and reoperation if necessary.4
Our patient underwent emergent surgery for aortic root/ascending aortic replacement with a mechanical valve. He tolerated the procedure well. Surgical tissue pathology of the aortic segment showed a wall of elastic vessel with medial degeneration and dissection, and the tissue pathology of the aorta leaflets showed valvular tissue with myxoid degeneration.
THE TAKEAWAY
It is critical to keep AAD in the differential diagnosis of a patient presenting with acute onset of chest pain, as AAD often has an atypical presentation and can easily be misdiagnosed. Effective imaging is crucial to diagnosis, and immediate treatment is essential to patient survival.
CORRESPONDENCE
Rachel A. Reedy, PA, University of Florida, Department of General Pediatrics, 7046 SW Archer Road, Gainesville, FL 32608; [email protected]
THE CASE
A 20-year-old man presented to our clinic with a 3-day history of nonradiating chest pain located at the center of his chest. Past medical history included idiopathic neonatal giant-cell hepatitis and subsequent liver transplant at 1 month of age; he had been followed by the transplant team without rejection or infection and was in otherwise good health prior to the chest pain.
On the day of symptom onset, he was walking inside his house and fell to his knees with a chest pain described as “a punch” to the center of the chest that lasted for a few seconds. He was able to continue his daily activities without limitation despite a constant, squeezing, centrally located chest pain. The pain worsened with cough and exertion.
A few hours later, he went to an urgent care center for evaluation. There, he reported, his chest radiograph and electrocardiogram (EKG) results were normal and he was given a diagnosis of musculoskeletal chest pain. Over the next 3 days, his chest pain persisted but did not worsen. He was taking 500 mg of naproxen every 8 hours with no improvement. No other acute or chronic medications were being taken. He had no significant family history. A review of systems was otherwise negative.
On physical exam, his vital statistics included a height of 6’4”; weight, 261 lb; body mass index, 31.8; temperature, 98.7 °F; blood pressure, 134/77 mm Hg; heart rate, 92 beats/min; respiratory rate, 18 breaths/min; and oxygen saturation, 96%. Throughout the exam, he demonstrated no acute distress, appeared well, and was talkative; however, he reported having a “constant, squeezing” chest pain that did not worsen with palpation of the chest. The rest of his physical exam was unremarkable.
Although he reported that his EKG and chest radiograph were normal 3 days prior, repeat chest radiograph and EKG were ordered due to his unexplained, active chest pain and the lack of immediate access to the prior results.
THE DIAGNOSIS
The chest radiograph (FIGURE 1A) showed a “mildly ectatic ascending thoracic aorta” that had increased since a chest radiograph from 6 years prior (FIGURE 1B) and “was concerning for an aneurysm.” Computed tomography (CT) angiography (FIGURE 2) then confirmed a 7-cm aneurysm of the ascending aorta, with findings suggestive of a retrograde ascending aortic dissection.

DISCUSSION
The average age of a patient with acute aortic dissection (AAD) is 63 years; only 7% occur in people younger than 40.1 AAD is often accompanied by a predisposing risk factor such as a connective tissue disease, bicuspid aortic valve, longstanding hypertension, trauma, or larger aortic dimensions.2,3 Younger patients are more likely to have predisposing risk factors of Marfan syndrome, prior aortic surgery, or a bicuspid aortic valve.3

Continue to: A literature review did not reveal...
A literature review did not reveal any known correlation between the patient’s history of giant-cell hepatitis or antirejection therapy with thoracic aortic dissection. Furthermore, liver transplant is not known to be a specific risk factor for AAD in pediatric patients or outside the immediate postoperative period. Therefore, there were no known predisposing risk factors for AAD in our patient.
The most common clinical feature of AAD is chest pain, which occurs in 75% of patients.1 Other clinical symptoms include hypertension and diaphoresis.2,4 However, classic clinical findings are not always displayed, making the diagnosis difficult.2,4 The classical description of “tearing pain” is seen in only 51% of patients, and 5% to 15% of patients present without any pain.1
Commonly missed or misdiagnosed. The diagnosis of AAD has been missed during the initial exam in 38% of patients.4 As seen in our case, symptoms may be initially diagnosed as musculoskeletal chest pain. Based on symptoms, AAD can be incorrectly diagnosed as an acute myocardial infarction or vascular embolization.2,4
Every hour after symptom onset, the mortality rate of untreated AAD increases 1% to 2%,with no difference based on age.3,4 Different reports have shown mortality rates between 7% and 30%.4
Effective imaging is crucial to the diagnosis and treatment of AAD, given the occurrence of atypical presentation, missed diagnosis, and high mortality rate.4 A chest radiograph will show a widened mediastinum, but the preferred diagnostic tests are a CT or transthoracic echocardiogram.2,4 Once the diagnosis of AAD is confirmed, an aortic angiogram is the preferred test to determine the extent of the dissection prior to surgical treatment.2
Continue to: Classification dictates treatment
Classification dictates treatment. AAD is classified based on where the dissection of the aorta occurs. If the dissection involves the ascending aorta, it is classified as a type A AAD and should immediately be treated with emergent surgery in order to prevent complications including myocardial infarction, cardiac tamponade, and aortic rupture.2,4,5 If the dissection is limited to the descending aorta, it is classified as a type B AAD and can be medically managed by controlling pain and lowering blood pressure; if symptoms persist, surgical management may be required.2 After hospital discharge, AAD patients are followed closely with medical therapy, serial imaging, and reoperation if necessary.4
Our patient underwent emergent surgery for aortic root/ascending aortic replacement with a mechanical valve. He tolerated the procedure well. Surgical tissue pathology of the aortic segment showed a wall of elastic vessel with medial degeneration and dissection, and the tissue pathology of the aorta leaflets showed valvular tissue with myxoid degeneration.
THE TAKEAWAY
It is critical to keep AAD in the differential diagnosis of a patient presenting with acute onset of chest pain, as AAD often has an atypical presentation and can easily be misdiagnosed. Effective imaging is crucial to diagnosis, and immediate treatment is essential to patient survival.
CORRESPONDENCE
Rachel A. Reedy, PA, University of Florida, Department of General Pediatrics, 7046 SW Archer Road, Gainesville, FL 32608; [email protected]
1. Pineault J, Ouimet D, Pichette V, Vallée M. A case of aortic dissection in a young adult: a refresher of the literature of this “great masquerader.” Int J Gen Med. 2011;4:889-893.
2. Agabegi SS, Agabegi ElD, Ring AC. Diseases of the cardiovascular system. In: Jackson A, ed. Step-up to Medicine. 3rd ed. Lippincott Williams & Wilkins; 2012:54-55.
3. Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol. 2004;43:665-669.
4. Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD). Eur J Vasc Endovasc Surg. 2009;37:149-159.
5. Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: Report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg. 2010;140:784-789.
1. Pineault J, Ouimet D, Pichette V, Vallée M. A case of aortic dissection in a young adult: a refresher of the literature of this “great masquerader.” Int J Gen Med. 2011;4:889-893.
2. Agabegi SS, Agabegi ElD, Ring AC. Diseases of the cardiovascular system. In: Jackson A, ed. Step-up to Medicine. 3rd ed. Lippincott Williams & Wilkins; 2012:54-55.
3. Januzzi JL, Isselbacher EM, Fattori R, et al. Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J Am Coll Cardiol. 2004;43:665-669.
4. Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD). Eur J Vasc Endovasc Surg. 2009;37:149-159.
5. Trimarchi S, Eagle KA, Nienaber CA, et al. Role of age in acute type A aortic dissection outcome: Report from the International Registry of Acute Aortic Dissection (IRAD). J Thorac Cardiovasc Surg. 2010;140:784-789.
PCPs play a small part in low-value care spending
according to a brief report published online Jan. 18 in Annals of Internal Medicine.
However, one expert said there are better ways to curb low-value care than focusing on which specialties are guilty of the practice.
Analyzing a 20% random sample of Medicare Part B claims, Aaron Baum, PhD, with the Icahn School of Medicine at Mount Sinai, New York, and colleagues found that the services primary care physicians performed or ordered made up on average 8.3% of the low-value care their patients received (interquartile range, 3.9%-15.1%; 95th percentile, 35.6%) and their referrals made up 15.4% (IQR, 6.3%-26.4%; 95th percentile, 44.6%).
By specialty, cardiology had the worst record with 27% of all spending on low-value services ($1.8 billion) attributed to that specialty. Yet, of the 25 highest-spending specialties in the report, 12 of them were associated with 1% or less than 1% each of all low-value spending, indicating the waste was widely distributed.
Dr. Baum said in an interview that though there are some PCPs guilty of high spending on low-value services, overall, most primary care physicians’ low-value services add up to only 0.3% of Part B spending. He noted that Part B spending is about one-third of all Medicare spending.
Primary care is often thought to be at the core of care management and spending and PCPs are often seen as the gatekeepers, but this analysis suggests that efforts to make big differences in curtailing low-value spending might be more effective elsewhere.
“There’s only so much spending you can reduce by changing primary care physicians’ services that they directly perform,” Dr. Baum said.
Low-value care is costly, can be harmful
Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, said in an interview that the report adds confirmation to previous research that has consistently shown low-value care is “extremely common, very costly, and provided by primary care providers and specialists alike.” He noted that it can also be harmful.
“The math is simple,” he said. “If we want to improve coverage and lower patient costs for essential services like visits, diagnostic tests, and drugs, we have to reduce spending on those services that do not make Americans any healthier.”
The study ranked 31 clinical services judged to be low value by physician societies, Medicare and clinical guidelines, and their use among beneficiaries enrolled between 2007 and 2014. Here’s how the top six low-value services compare.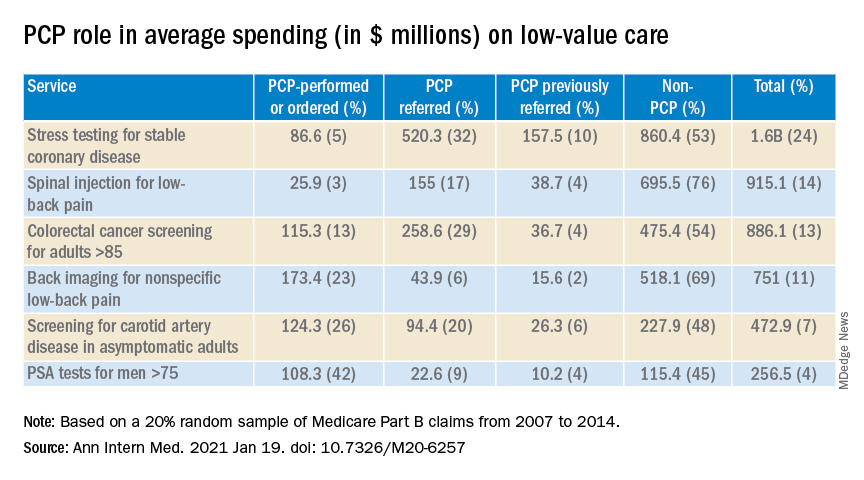
Dr. Fendrick said a weakness of the paper is the years of the data (2007-2014). Some of the criteria around low-value care have changed since then. The age that a prostate-specific antigen test becomes low-value is now 70 years, for instance, instead of 75. He added that some of the figures attributed to non-PCP providers appear out of date.
Dr. Fendrick said, “I understand that there are Medicare patients who end up at a gastroenterologist or surgeon’s office to get colorectal cancer screening, but it would be very hard for me to believe that half of stress tests and over half of colon cancer screening over [age] 85 [years] and half of PSA for people over 75 did not have some type of referring clinicians involved. I certainly don’t think that would be the case in 2020-2021.”
Dr. Baum said those years were the latest years available for the data points needed for this analysis, but he and his colleagues were working to update the data for future publication.
Dr. Fendrick said not much has changed in recent years in terms of waste on low-value care, even with campaigns such as Choosing Wisely dedicated to identifying low-value services or procedures in each specialty.
“I believe there’s not a particular group of clinicians one way or the other who are actually doing any better now than they were 7 years ago,” he said. He would rather focus less on which specialties are associated with the most low-value care and more on the underlying policies that encourage low-value care.
“If you’re going to get paid for doing a stress test and get paid nothing or significantly less if you don’t, the incentives are in the wrong direction,” he said.
Dr. Fendrick said the pandemic era provides an opportunity to eliminate low-value care because use of those services has dropped drastically as resources have been diverted to COVID-19 patients and many services have been delayed or canceled.
He said he has been pushing an approach that providers should be paid more after the pandemic “to do the things we want them to do.”
As an example, he said, instead of paying $886 million on colonoscopies for people over the age of 85, “why don’t we put a policy in place that would make it better for patients by lowering cost sharing and better for providers by paying them more to do the service on the people who need it as opposed to the people who don’t?”
The research was funded by the American Board of Family Medicine Foundation. Dr. Baum and a coauthor reported receiving personal fees from American Board of Family Medicine Foundation during the conduct of the study. Another coauthor reported receiving personal fees from Collective Health, HealthRight 360, PLOS Medicine, and the New England Journal of Medicine, outside the submitted work. Dr. Fendrick disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a brief report published online Jan. 18 in Annals of Internal Medicine.
However, one expert said there are better ways to curb low-value care than focusing on which specialties are guilty of the practice.
Analyzing a 20% random sample of Medicare Part B claims, Aaron Baum, PhD, with the Icahn School of Medicine at Mount Sinai, New York, and colleagues found that the services primary care physicians performed or ordered made up on average 8.3% of the low-value care their patients received (interquartile range, 3.9%-15.1%; 95th percentile, 35.6%) and their referrals made up 15.4% (IQR, 6.3%-26.4%; 95th percentile, 44.6%).
By specialty, cardiology had the worst record with 27% of all spending on low-value services ($1.8 billion) attributed to that specialty. Yet, of the 25 highest-spending specialties in the report, 12 of them were associated with 1% or less than 1% each of all low-value spending, indicating the waste was widely distributed.
Dr. Baum said in an interview that though there are some PCPs guilty of high spending on low-value services, overall, most primary care physicians’ low-value services add up to only 0.3% of Part B spending. He noted that Part B spending is about one-third of all Medicare spending.
Primary care is often thought to be at the core of care management and spending and PCPs are often seen as the gatekeepers, but this analysis suggests that efforts to make big differences in curtailing low-value spending might be more effective elsewhere.
“There’s only so much spending you can reduce by changing primary care physicians’ services that they directly perform,” Dr. Baum said.
Low-value care is costly, can be harmful
Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, said in an interview that the report adds confirmation to previous research that has consistently shown low-value care is “extremely common, very costly, and provided by primary care providers and specialists alike.” He noted that it can also be harmful.
“The math is simple,” he said. “If we want to improve coverage and lower patient costs for essential services like visits, diagnostic tests, and drugs, we have to reduce spending on those services that do not make Americans any healthier.”
The study ranked 31 clinical services judged to be low value by physician societies, Medicare and clinical guidelines, and their use among beneficiaries enrolled between 2007 and 2014. Here’s how the top six low-value services compare.
Dr. Fendrick said a weakness of the paper is the years of the data (2007-2014). Some of the criteria around low-value care have changed since then. The age that a prostate-specific antigen test becomes low-value is now 70 years, for instance, instead of 75. He added that some of the figures attributed to non-PCP providers appear out of date.
Dr. Fendrick said, “I understand that there are Medicare patients who end up at a gastroenterologist or surgeon’s office to get colorectal cancer screening, but it would be very hard for me to believe that half of stress tests and over half of colon cancer screening over [age] 85 [years] and half of PSA for people over 75 did not have some type of referring clinicians involved. I certainly don’t think that would be the case in 2020-2021.”
Dr. Baum said those years were the latest years available for the data points needed for this analysis, but he and his colleagues were working to update the data for future publication.
Dr. Fendrick said not much has changed in recent years in terms of waste on low-value care, even with campaigns such as Choosing Wisely dedicated to identifying low-value services or procedures in each specialty.
“I believe there’s not a particular group of clinicians one way or the other who are actually doing any better now than they were 7 years ago,” he said. He would rather focus less on which specialties are associated with the most low-value care and more on the underlying policies that encourage low-value care.
“If you’re going to get paid for doing a stress test and get paid nothing or significantly less if you don’t, the incentives are in the wrong direction,” he said.
Dr. Fendrick said the pandemic era provides an opportunity to eliminate low-value care because use of those services has dropped drastically as resources have been diverted to COVID-19 patients and many services have been delayed or canceled.
He said he has been pushing an approach that providers should be paid more after the pandemic “to do the things we want them to do.”
As an example, he said, instead of paying $886 million on colonoscopies for people over the age of 85, “why don’t we put a policy in place that would make it better for patients by lowering cost sharing and better for providers by paying them more to do the service on the people who need it as opposed to the people who don’t?”
The research was funded by the American Board of Family Medicine Foundation. Dr. Baum and a coauthor reported receiving personal fees from American Board of Family Medicine Foundation during the conduct of the study. Another coauthor reported receiving personal fees from Collective Health, HealthRight 360, PLOS Medicine, and the New England Journal of Medicine, outside the submitted work. Dr. Fendrick disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a brief report published online Jan. 18 in Annals of Internal Medicine.
However, one expert said there are better ways to curb low-value care than focusing on which specialties are guilty of the practice.
Analyzing a 20% random sample of Medicare Part B claims, Aaron Baum, PhD, with the Icahn School of Medicine at Mount Sinai, New York, and colleagues found that the services primary care physicians performed or ordered made up on average 8.3% of the low-value care their patients received (interquartile range, 3.9%-15.1%; 95th percentile, 35.6%) and their referrals made up 15.4% (IQR, 6.3%-26.4%; 95th percentile, 44.6%).
By specialty, cardiology had the worst record with 27% of all spending on low-value services ($1.8 billion) attributed to that specialty. Yet, of the 25 highest-spending specialties in the report, 12 of them were associated with 1% or less than 1% each of all low-value spending, indicating the waste was widely distributed.
Dr. Baum said in an interview that though there are some PCPs guilty of high spending on low-value services, overall, most primary care physicians’ low-value services add up to only 0.3% of Part B spending. He noted that Part B spending is about one-third of all Medicare spending.
Primary care is often thought to be at the core of care management and spending and PCPs are often seen as the gatekeepers, but this analysis suggests that efforts to make big differences in curtailing low-value spending might be more effective elsewhere.
“There’s only so much spending you can reduce by changing primary care physicians’ services that they directly perform,” Dr. Baum said.
Low-value care is costly, can be harmful
Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, said in an interview that the report adds confirmation to previous research that has consistently shown low-value care is “extremely common, very costly, and provided by primary care providers and specialists alike.” He noted that it can also be harmful.
“The math is simple,” he said. “If we want to improve coverage and lower patient costs for essential services like visits, diagnostic tests, and drugs, we have to reduce spending on those services that do not make Americans any healthier.”
The study ranked 31 clinical services judged to be low value by physician societies, Medicare and clinical guidelines, and their use among beneficiaries enrolled between 2007 and 2014. Here’s how the top six low-value services compare.
Dr. Fendrick said a weakness of the paper is the years of the data (2007-2014). Some of the criteria around low-value care have changed since then. The age that a prostate-specific antigen test becomes low-value is now 70 years, for instance, instead of 75. He added that some of the figures attributed to non-PCP providers appear out of date.
Dr. Fendrick said, “I understand that there are Medicare patients who end up at a gastroenterologist or surgeon’s office to get colorectal cancer screening, but it would be very hard for me to believe that half of stress tests and over half of colon cancer screening over [age] 85 [years] and half of PSA for people over 75 did not have some type of referring clinicians involved. I certainly don’t think that would be the case in 2020-2021.”
Dr. Baum said those years were the latest years available for the data points needed for this analysis, but he and his colleagues were working to update the data for future publication.
Dr. Fendrick said not much has changed in recent years in terms of waste on low-value care, even with campaigns such as Choosing Wisely dedicated to identifying low-value services or procedures in each specialty.
“I believe there’s not a particular group of clinicians one way or the other who are actually doing any better now than they were 7 years ago,” he said. He would rather focus less on which specialties are associated with the most low-value care and more on the underlying policies that encourage low-value care.
“If you’re going to get paid for doing a stress test and get paid nothing or significantly less if you don’t, the incentives are in the wrong direction,” he said.
Dr. Fendrick said the pandemic era provides an opportunity to eliminate low-value care because use of those services has dropped drastically as resources have been diverted to COVID-19 patients and many services have been delayed or canceled.
He said he has been pushing an approach that providers should be paid more after the pandemic “to do the things we want them to do.”
As an example, he said, instead of paying $886 million on colonoscopies for people over the age of 85, “why don’t we put a policy in place that would make it better for patients by lowering cost sharing and better for providers by paying them more to do the service on the people who need it as opposed to the people who don’t?”
The research was funded by the American Board of Family Medicine Foundation. Dr. Baum and a coauthor reported receiving personal fees from American Board of Family Medicine Foundation during the conduct of the study. Another coauthor reported receiving personal fees from Collective Health, HealthRight 360, PLOS Medicine, and the New England Journal of Medicine, outside the submitted work. Dr. Fendrick disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.