User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Psoriasiform Dermatitis Associated With the Moderna COVID-19 Messenger RNA Vaccine
To the Editor:
The Moderna COVID-19 messenger RNA (mRNA) vaccine was authorized for use on December 18, 2020, with the second dose beginning on January 15, 2021.1-3 Some individuals who received the Moderna vaccine experienced an intense rash known as “COVID arm,” a harmless but bothersome adverse effect that typically appears within a week and is a localized and transient immunogenic response.4 COVID arm differs from most vaccine adverse effects. The rash emerges not immediately but 5 to 9 days after the initial dose—on average, 1 week later. Apart from being itchy, the rash does not appear to be harmful and is not a reason to hesitate getting vaccinated.
Dermatologists and allergists have been studying this adverse effect, which has been formally termed delayed cutaneous hypersensitivity. Of potential clinical consequence is that the efficacy of the mRNA COVID-19 vaccine may be harmed if postvaccination dermal reactions necessitate systemic corticosteroid therapy. Because this vaccine stimulates an immune response as viral RNA integrates in cells secondary to production of the spike protein of the virus, the skin may be affected secondarily and manifestations of any underlying disease may be aggravated.5 We report a patient who developed a psoriasiform dermatitis after the first dose of the Moderna vaccine.
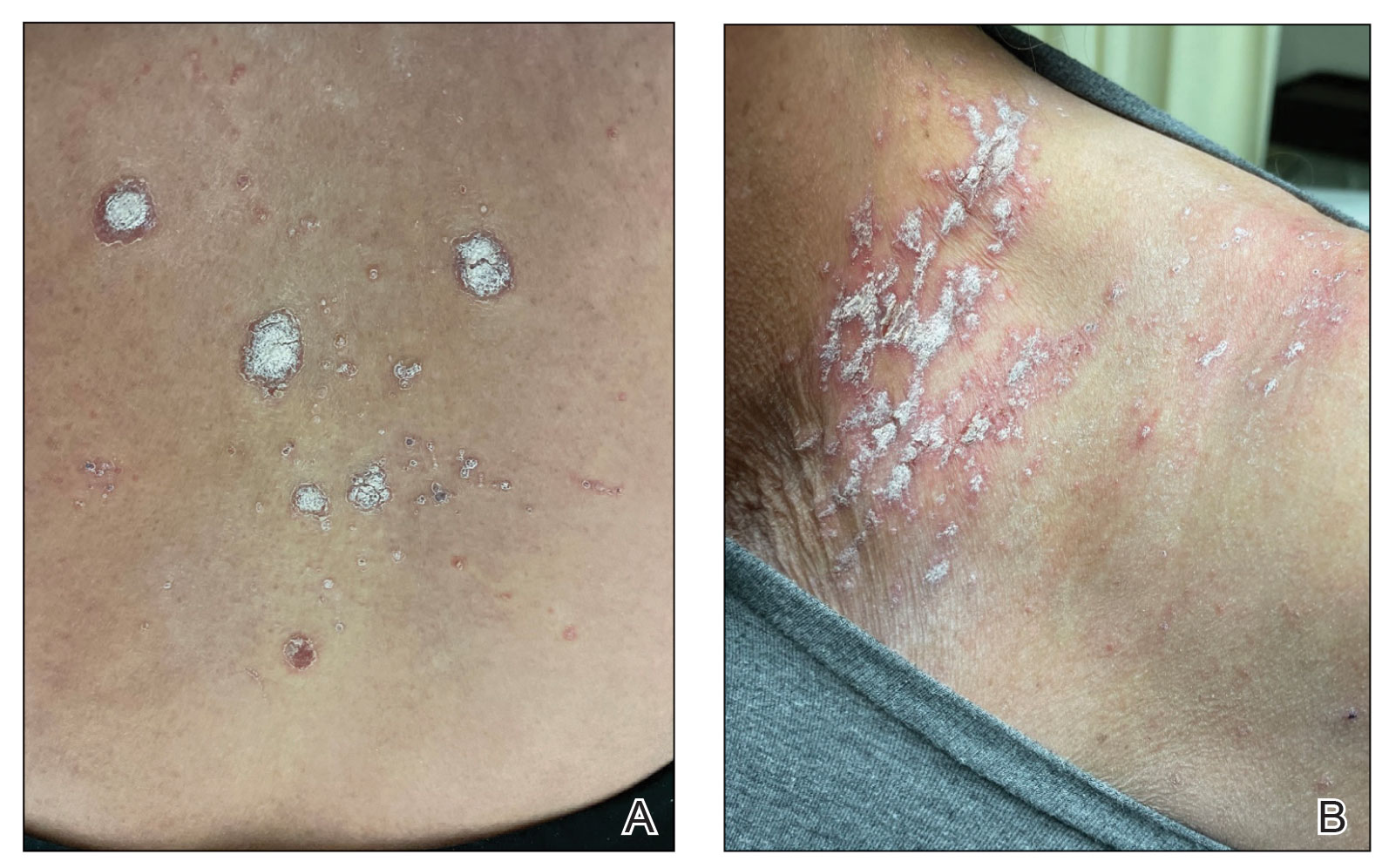
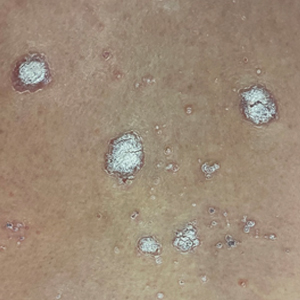
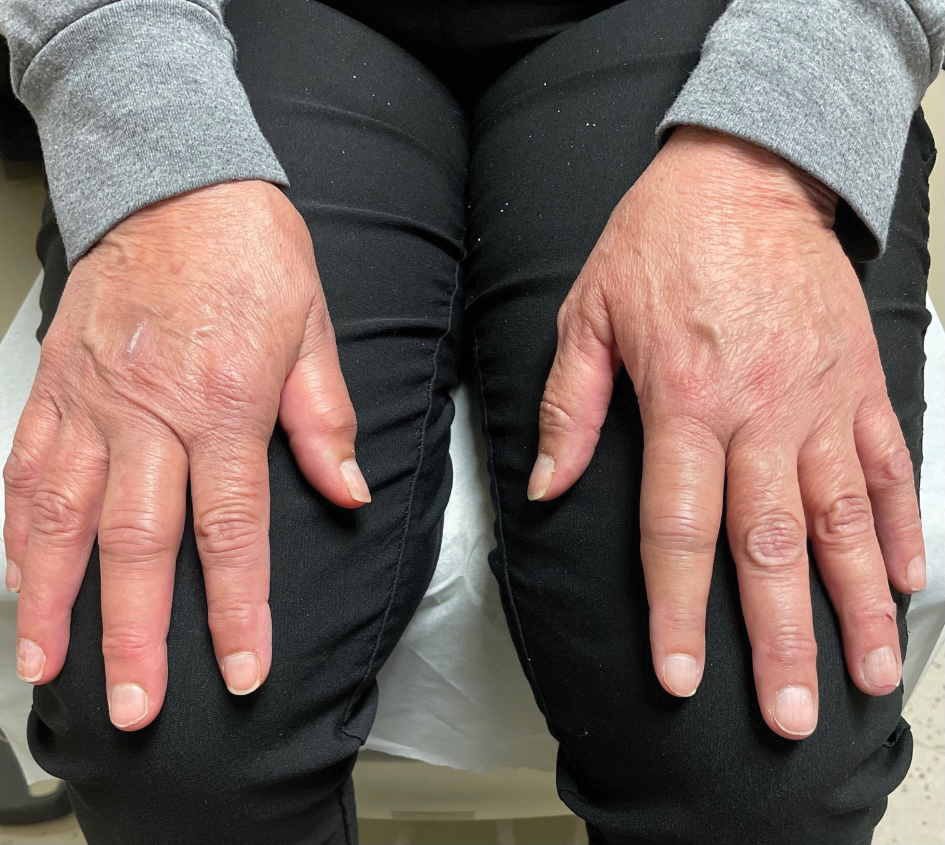
A 65-year-old woman presented to her primary care physician because of the severity of psoriasiform dermatitis that developed 5 days after she received the first dose of the Moderna COVID-19 mRNA vaccine. The patient had a medical history of Sjögren syndrome. Her medication history was negative, and her family history was negative for autoimmune disease. Physical examination by primary care revealed an erythematous scaly rash with plaques and papules on the neck and back (Figure 1). The patient presented again to primary care 2 days later with swollen, painful, discolored digits (Figure 2) and a stiff, sore neck.
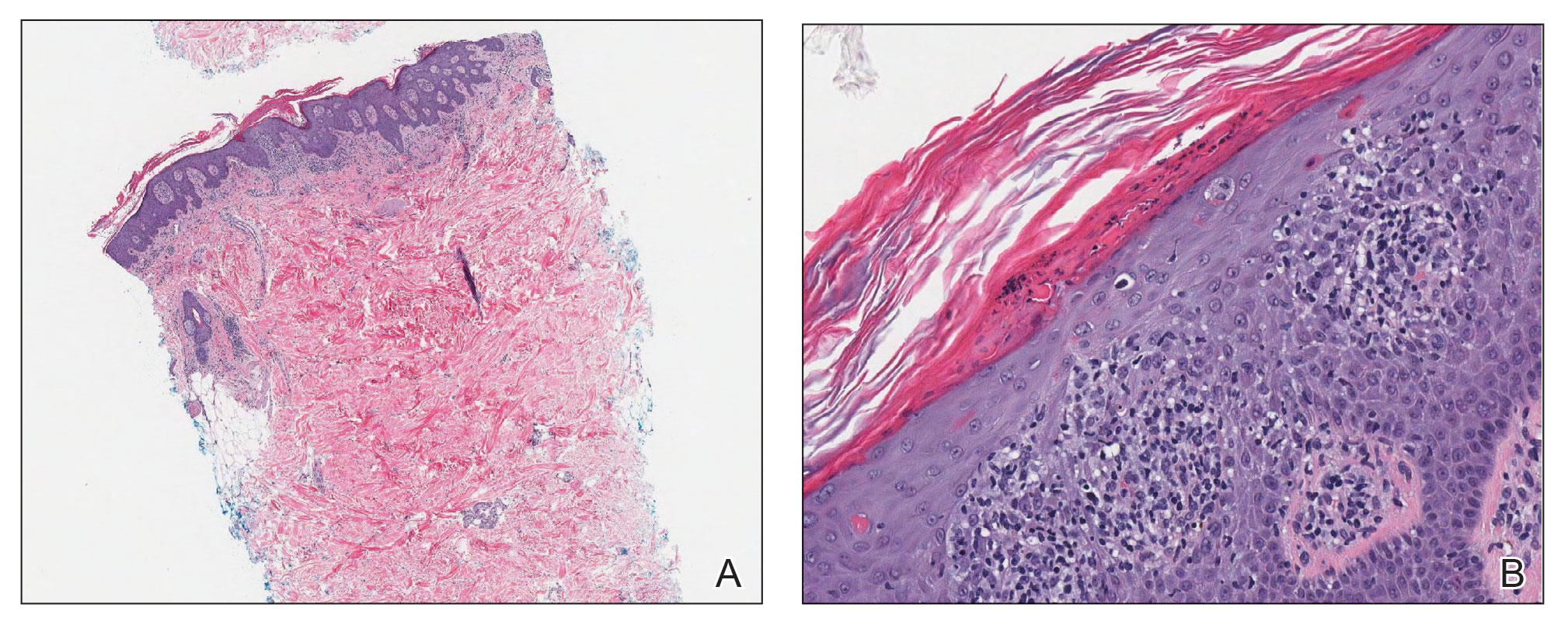
Laboratory results were positive for anti–Sjögren syndrome–related antigens A and B. A complete blood cell count; comprehensive metabolic panel; erythrocyte sedimentation rate; and assays of rheumatoid factor, C-reactive protein, and anti–cyclic citrullinated peptide were within reference range. A biopsy of a lesion on the back showed psoriasiform dermatitis with confluent parakeratosis and scattered necrotic keratinocytes. There was superficial perivascular inflammation with rare eosinophils (Figure 3).
The patient was treated with a course of systemic corticosteroids. The rash resolved in 1 week. She did not receive the second dose due to the rash.
Two mRNA COVID-19 vaccines—Pfizer BioNTech and Moderna—have been granted emergency use authorization by the US Food and Drug Administration.6 The safety profile of the mRNA-1273 vaccine for the median 2-month follow-up showed no safety concerns.3 Minor localized adverse effects (eg, pain, redness, swelling) have been observed more frequently with the vaccines than with placebo. Systemic symptoms, such as fever, fatigue, headache, and muscle and joint pain, also were seen somewhat more often with the vaccines than with placebo; most such effects occurred 24 to 48 hours after vaccination.3,6,7 The frequency of unsolicited adverse events and serious adverse events reported during the 28-day period after vaccination generally was similar among participants in the vaccine and placebo groups.3
There are 2 types of reactions to COVID-19 vaccination: immediate and delayed. Immediate reactions usually are due to anaphylaxis, requiring prompt recognition and treatment with epinephrine to stop rapid progression of life-threatening symptoms. Delayed reactions include localized reactions, such as urticaria and benign exanthema; serum sickness and serum sickness–like reactions; fever; and rare skin, organ, and neurologic sequelae.1,6-8
Cutaneous manifestations, present in 16% to 50% of patients with Sjögren syndrome, are considered one of the most common extraglandular presentations of the syndrome. They are classified as nonvascular (eg, xerosis, angular cheilitis, eyelid dermatitis, annular erythema) and vascular (eg, Raynaud phenomenon, vasculitis).9-11 Our patient did not have any of those findings. She had not taken any medications before the rash appeared, thereby ruling out a drug reaction.
The differential for our patient included post–urinary tract infection immune-reactive arthritis and rash, which is not typical with Escherichia coli infection but is described with infection with Chlamydia species and Salmonella species. Moreover, post–urinary tract infection immune-reactive arthritis and rash appear mostly on the palms and soles. Systemic lupus erythematosus–like rashes have a different histology and appear on sun-exposed areas; our patient’s rash was found mainly on unexposed areas.12
Because our patient received the Moderna vaccine 5 days before the rash appeared and later developed swelling of the digits with morning stiffness, a delayed serum sickness–like reaction secondary to COVID-19 vaccination was possible.3,6
COVID-19 mRNA vaccines developed by Pfizer-BioNTech and Moderna incorporate a lipid-based nanoparticle carrier system that prevents rapid enzymatic degradation of mRNA and facilitates in vivo delivery of mRNA. This lipid-based nanoparticle carrier system is further stabilized by a polyethylene glycol 2000 lipid conjugate that provides a hydrophilic layer, thus prolonging half-life. The presence of lipid polyethylene glycol 2000 in mRNA vaccines has led to concern that this component could be implicated in anaphylaxis.6
COVID-19 antigens can give rise to varying clinical manifestations that are directly related to viral tissue damage or are indirectly induced by the antiviral immune response.13,14 Hyperactivation of the immune system to eradicate COVID-19 may trigger autoimmunity; several immune-mediated disorders have been described in individuals infected with SARS-CoV-2. Dermal manifestations include cutaneous rash and vasculitis.13-16 Crucial immunologic steps occur during SARS-CoV-2 infection that may link autoimmunity to COVID-19.13,14 In preliminary published data on the efficacy of the Moderna vaccine on 45 trial enrollees, 3 did not receive the second dose of vaccination, including 1 who developed urticaria on both legs 5 days after the first dose.1
Introduction of viral RNA can induce autoimmunity that can be explained by various phenomena, including epitope spreading, molecular mimicry, cryptic antigen, and bystander activation. Remarkably, more than one-third of immunogenic proteins in SARS-CoV-2 have potentially problematic homology to proteins that are key to the human adaptive immune system.5
Moreover, SARS-CoV-2 seems to induce organ injury through alternative mechanisms beyond direct viral infection, including immunologic injury. In some situations, hyperactivation of the immune response to SARS-CoV-2 RNA can result in autoimmune disease. COVID-19 has been associated with immune-mediated systemic or organ-selective manifestations, some of which fulfill the diagnostic or classification criteria of specific autoimmune diseases. It is unclear whether those medical disorders are the result of transitory postinfectious epiphenomena.5
A few studies have shown that patients with rheumatic disease have an incidence and prevalence of COVID-19 that is similar to the general population. A similar pattern has been detected in COVID-19 morbidity and mortality rates, even among patients with an autoimmune disease, such as rheumatoid arthritis and Sjögren syndrome.5,17 Furthermore, exacerbation of preexisting rheumatic symptoms may be due to hyperactivation of antiviral pathways in a person with an autoimmune disease.17-19 The findings in our patient suggested a direct role for the vaccine in skin manifestations, rather than for reactivation or development of new systemic autoimmune processes, such as systemic lupus erythematosus.
Exacerbation of psoriasis following COVID-19 vaccination has been described20; however, the case patient did not have a history of psoriasis. The mechanism(s) of such exacerbation remain unclear; COVID-19 vaccine–induced helper T cells (TH17) may play a role.21 Other skin manifestations encountered following COVID-19 vaccination include lichen planus, leukocytoclastic vasculitic rash, erythema multiforme–like rash, and pityriasis rosea–like rash.22-25 The immune mechanisms of these manifestations remain unclear.
The clinical presentation of delayed vaccination reactions can be attributed to the timing of symptoms and, in this case, the immune-mediated background of a psoriasiform reaction. Although adverse reactions to the SARS-CoV-2 mRNA vaccine are rare, more individuals should be studied after vaccination to confirm and better understand this phenomenon.
- Jackson LA, Anderson EJ, Rouphael NG, et al; . An mRNA vaccine against SARS-CoV-2—preliminary report. N Engl J Med. 2020;383:1920-1931. doi:10.1056/NEJMoa2022483
- Anderson EJ, Rouphael NG, Widge AT, et al; . Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N Engl J Med. 2020;383:2427-2438. doi:10.1056/NEJMoa2028436
- Baden LR, El Sahly HM, Essink B, et al; COVE Study Group. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021;384:403-416. doi:10.1056/NEJMoa2035389
- Weise E. ‘COVID arm’ rash seen after Moderna vaccine annoying but harmless, doctors say. USA Today. January 27, 2021. Accessed September 4, 2022. https://www.usatoday.com/story/news/health/2021/01/27/covid-arm-moderna-vaccine-rash-harmless-side-effect-doctors-say/4277725001/
- Talotta R, Robertson E. Autoimmunity as the comet tail of COVID-19 pandemic. World J Clin Cases. 2020;8:3621-3644. doi:10.12998/wjcc.v8.i17.3621
- Castells MC, Phillips EJ. Maintaining safety with SARS-CoV-2 vaccines. N Engl J Med. 2021;384:643-649. doi:10.1056/NEJMra2035343
- Polack FP, Thomas SJ, Kitchin N, et al; . Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603-2615. doi:10.1056/NEJMoa2034577
- Dooling K, McClung N, Chamberland M, et al. The Advisory Committee on Immunization Practices’ interim recommendation for allocating initial supplies of COVID-19 vaccine—United States, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1857-1859. doi:10.15585/mmwr.mm6949e1
- Roguedas AM, Misery L, Sassolas B, et al. Cutaneous manifestations of primary Sjögren’s syndrome are underestimated. Clin Exp Rheumatol. 2004;22:632-636.
- Katayama I. Dry skin manifestations in Sjögren syndrome and atopic dermatitis related to aberrant sudomotor function in inflammatory allergic skin diseases. Allergol Int. 2018;67:448-454. doi:10.1016/j.alit.2018.07.001
- Generali E, Costanzo A, Mainetti C, et al. Cutaneous and mucosal manifestations of Sjögren’s syndrome. Clin Rev Allergy Immunol. 2017;53:357-370. doi:10.1007/s12016-017-8639-y
- Chanprapaph K, Tankunakorn J, Suchonwanit P, et al. Dermatologic manifestations, histologic features and disease progression among cutaneous lupus erythematosus subtypes: a prospective observational study in Asians. Dermatol Ther (Heidelb). 2021;11:131-147. doi:10.1007/s13555-020-00471-y
- Ortega-Quijano D, Jimenez-Cauhe J, Selda-Enriquez G, et al. Algorithm for the classification of COVID-19 rashes. J Am Acad Dermatol. 2020;83:e103-e104. doi:10.1016/j.jaad.2020.05.034
- Rahimi H, Tehranchinia Z. A comprehensive review of cutaneous manifestations associated with COVID-19. Biomed Res Int. 2020;2020:1236520. doi:10.1155/2020/1236520
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98:75-81. doi:10.1016/j.jdermsci.2020.04.011
- Landa N, Mendieta-Eckert M, Fonda-Pascual P, et al. Chilblain-like lesions on feet and hands during the COVID-19 pandemic. Int J Dermatol. 2020;59:739-743. doi:10.1111/ijd.14937
- Dellavance A, Coelho Andrade LE. Immunologic derangement preceding clinical autoimmunity. Lupus. 2014;23:1305-1308. doi:10.1177/0961203314531346
- Parodi A, Gasparini G, Cozzani E. Could antiphospholipid antibodies contribute to coagulopathy in COVID-19? J Am Acad Dermatol. 2020;83:e249. doi:10.1016/j.jaad.2020.06.003
- Zhou Y, Han T, Chen J, et al. Clinical and autoimmune characteristics of severe and critical cases of COVID-19. Clin Transl Sci. 2020;13:1077-1086. doi:10.1111/cts.12805
- Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010. doi:10.3389/fmed.2021.812010
- Rouai M, Slimane MB, Sassi W, et al. Pustular rash triggered by Pfizer-BioNTech COVID-19 vaccination: a case report. Dermatol Ther. 2022:e15465. doi:10.1111/dth.15465
- Altun E, Kuzucular E. Leukocytoclastic vasculitis after COVID-19 vaccination. Dermatol Ther. 2022;35:e15279. doi:10.1111/dth.15279
- Buckley JE, Landis LN, Rapini RP. Pityriasis rosea-like rash after mRNA COVID-19 vaccination: a case report and review of the literature. JAAD Int. 2022;7:164-168. doi:10.1016/j.jdin.2022.01.009
- Gökçek GE, Öksüm Solak E, Çölgeçen E. Pityriasis rosea like eruption: a dermatological manifestation of Coronavac-COVID-19 vaccine. Dermatol Ther. 2022;35:e15256. doi:10.1111/dth.15256
- Kim MJ, Kim JW, Kim MS, et al. Generalized erythema multiforme-like skin rash following the first dose of COVID-19 vaccine (Pfizer-BioNTech). J Eur Acad Dermatol Venereol. 2022;36:e98-e100. doi:10.1111/jdv.17757
To the Editor:
The Moderna COVID-19 messenger RNA (mRNA) vaccine was authorized for use on December 18, 2020, with the second dose beginning on January 15, 2021.1-3 Some individuals who received the Moderna vaccine experienced an intense rash known as “COVID arm,” a harmless but bothersome adverse effect that typically appears within a week and is a localized and transient immunogenic response.4 COVID arm differs from most vaccine adverse effects. The rash emerges not immediately but 5 to 9 days after the initial dose—on average, 1 week later. Apart from being itchy, the rash does not appear to be harmful and is not a reason to hesitate getting vaccinated.
Dermatologists and allergists have been studying this adverse effect, which has been formally termed delayed cutaneous hypersensitivity. Of potential clinical consequence is that the efficacy of the mRNA COVID-19 vaccine may be harmed if postvaccination dermal reactions necessitate systemic corticosteroid therapy. Because this vaccine stimulates an immune response as viral RNA integrates in cells secondary to production of the spike protein of the virus, the skin may be affected secondarily and manifestations of any underlying disease may be aggravated.5 We report a patient who developed a psoriasiform dermatitis after the first dose of the Moderna vaccine.

A 65-year-old woman presented to her primary care physician because of the severity of psoriasiform dermatitis that developed 5 days after she received the first dose of the Moderna COVID-19 mRNA vaccine. The patient had a medical history of Sjögren syndrome. Her medication history was negative, and her family history was negative for autoimmune disease. Physical examination by primary care revealed an erythematous scaly rash with plaques and papules on the neck and back (Figure 1). The patient presented again to primary care 2 days later with swollen, painful, discolored digits (Figure 2) and a stiff, sore neck.

Laboratory results were positive for anti–Sjögren syndrome–related antigens A and B. A complete blood cell count; comprehensive metabolic panel; erythrocyte sedimentation rate; and assays of rheumatoid factor, C-reactive protein, and anti–cyclic citrullinated peptide were within reference range. A biopsy of a lesion on the back showed psoriasiform dermatitis with confluent parakeratosis and scattered necrotic keratinocytes. There was superficial perivascular inflammation with rare eosinophils (Figure 3).

The patient was treated with a course of systemic corticosteroids. The rash resolved in 1 week. She did not receive the second dose due to the rash.
Two mRNA COVID-19 vaccines—Pfizer BioNTech and Moderna—have been granted emergency use authorization by the US Food and Drug Administration.6 The safety profile of the mRNA-1273 vaccine for the median 2-month follow-up showed no safety concerns.3 Minor localized adverse effects (eg, pain, redness, swelling) have been observed more frequently with the vaccines than with placebo. Systemic symptoms, such as fever, fatigue, headache, and muscle and joint pain, also were seen somewhat more often with the vaccines than with placebo; most such effects occurred 24 to 48 hours after vaccination.3,6,7 The frequency of unsolicited adverse events and serious adverse events reported during the 28-day period after vaccination generally was similar among participants in the vaccine and placebo groups.3
There are 2 types of reactions to COVID-19 vaccination: immediate and delayed. Immediate reactions usually are due to anaphylaxis, requiring prompt recognition and treatment with epinephrine to stop rapid progression of life-threatening symptoms. Delayed reactions include localized reactions, such as urticaria and benign exanthema; serum sickness and serum sickness–like reactions; fever; and rare skin, organ, and neurologic sequelae.1,6-8
Cutaneous manifestations, present in 16% to 50% of patients with Sjögren syndrome, are considered one of the most common extraglandular presentations of the syndrome. They are classified as nonvascular (eg, xerosis, angular cheilitis, eyelid dermatitis, annular erythema) and vascular (eg, Raynaud phenomenon, vasculitis).9-11 Our patient did not have any of those findings. She had not taken any medications before the rash appeared, thereby ruling out a drug reaction.
The differential for our patient included post–urinary tract infection immune-reactive arthritis and rash, which is not typical with Escherichia coli infection but is described with infection with Chlamydia species and Salmonella species. Moreover, post–urinary tract infection immune-reactive arthritis and rash appear mostly on the palms and soles. Systemic lupus erythematosus–like rashes have a different histology and appear on sun-exposed areas; our patient’s rash was found mainly on unexposed areas.12
Because our patient received the Moderna vaccine 5 days before the rash appeared and later developed swelling of the digits with morning stiffness, a delayed serum sickness–like reaction secondary to COVID-19 vaccination was possible.3,6
COVID-19 mRNA vaccines developed by Pfizer-BioNTech and Moderna incorporate a lipid-based nanoparticle carrier system that prevents rapid enzymatic degradation of mRNA and facilitates in vivo delivery of mRNA. This lipid-based nanoparticle carrier system is further stabilized by a polyethylene glycol 2000 lipid conjugate that provides a hydrophilic layer, thus prolonging half-life. The presence of lipid polyethylene glycol 2000 in mRNA vaccines has led to concern that this component could be implicated in anaphylaxis.6
COVID-19 antigens can give rise to varying clinical manifestations that are directly related to viral tissue damage or are indirectly induced by the antiviral immune response.13,14 Hyperactivation of the immune system to eradicate COVID-19 may trigger autoimmunity; several immune-mediated disorders have been described in individuals infected with SARS-CoV-2. Dermal manifestations include cutaneous rash and vasculitis.13-16 Crucial immunologic steps occur during SARS-CoV-2 infection that may link autoimmunity to COVID-19.13,14 In preliminary published data on the efficacy of the Moderna vaccine on 45 trial enrollees, 3 did not receive the second dose of vaccination, including 1 who developed urticaria on both legs 5 days after the first dose.1
Introduction of viral RNA can induce autoimmunity that can be explained by various phenomena, including epitope spreading, molecular mimicry, cryptic antigen, and bystander activation. Remarkably, more than one-third of immunogenic proteins in SARS-CoV-2 have potentially problematic homology to proteins that are key to the human adaptive immune system.5
Moreover, SARS-CoV-2 seems to induce organ injury through alternative mechanisms beyond direct viral infection, including immunologic injury. In some situations, hyperactivation of the immune response to SARS-CoV-2 RNA can result in autoimmune disease. COVID-19 has been associated with immune-mediated systemic or organ-selective manifestations, some of which fulfill the diagnostic or classification criteria of specific autoimmune diseases. It is unclear whether those medical disorders are the result of transitory postinfectious epiphenomena.5
A few studies have shown that patients with rheumatic disease have an incidence and prevalence of COVID-19 that is similar to the general population. A similar pattern has been detected in COVID-19 morbidity and mortality rates, even among patients with an autoimmune disease, such as rheumatoid arthritis and Sjögren syndrome.5,17 Furthermore, exacerbation of preexisting rheumatic symptoms may be due to hyperactivation of antiviral pathways in a person with an autoimmune disease.17-19 The findings in our patient suggested a direct role for the vaccine in skin manifestations, rather than for reactivation or development of new systemic autoimmune processes, such as systemic lupus erythematosus.
Exacerbation of psoriasis following COVID-19 vaccination has been described20; however, the case patient did not have a history of psoriasis. The mechanism(s) of such exacerbation remain unclear; COVID-19 vaccine–induced helper T cells (TH17) may play a role.21 Other skin manifestations encountered following COVID-19 vaccination include lichen planus, leukocytoclastic vasculitic rash, erythema multiforme–like rash, and pityriasis rosea–like rash.22-25 The immune mechanisms of these manifestations remain unclear.
The clinical presentation of delayed vaccination reactions can be attributed to the timing of symptoms and, in this case, the immune-mediated background of a psoriasiform reaction. Although adverse reactions to the SARS-CoV-2 mRNA vaccine are rare, more individuals should be studied after vaccination to confirm and better understand this phenomenon.
To the Editor:
The Moderna COVID-19 messenger RNA (mRNA) vaccine was authorized for use on December 18, 2020, with the second dose beginning on January 15, 2021.1-3 Some individuals who received the Moderna vaccine experienced an intense rash known as “COVID arm,” a harmless but bothersome adverse effect that typically appears within a week and is a localized and transient immunogenic response.4 COVID arm differs from most vaccine adverse effects. The rash emerges not immediately but 5 to 9 days after the initial dose—on average, 1 week later. Apart from being itchy, the rash does not appear to be harmful and is not a reason to hesitate getting vaccinated.
Dermatologists and allergists have been studying this adverse effect, which has been formally termed delayed cutaneous hypersensitivity. Of potential clinical consequence is that the efficacy of the mRNA COVID-19 vaccine may be harmed if postvaccination dermal reactions necessitate systemic corticosteroid therapy. Because this vaccine stimulates an immune response as viral RNA integrates in cells secondary to production of the spike protein of the virus, the skin may be affected secondarily and manifestations of any underlying disease may be aggravated.5 We report a patient who developed a psoriasiform dermatitis after the first dose of the Moderna vaccine.

A 65-year-old woman presented to her primary care physician because of the severity of psoriasiform dermatitis that developed 5 days after she received the first dose of the Moderna COVID-19 mRNA vaccine. The patient had a medical history of Sjögren syndrome. Her medication history was negative, and her family history was negative for autoimmune disease. Physical examination by primary care revealed an erythematous scaly rash with plaques and papules on the neck and back (Figure 1). The patient presented again to primary care 2 days later with swollen, painful, discolored digits (Figure 2) and a stiff, sore neck.

Laboratory results were positive for anti–Sjögren syndrome–related antigens A and B. A complete blood cell count; comprehensive metabolic panel; erythrocyte sedimentation rate; and assays of rheumatoid factor, C-reactive protein, and anti–cyclic citrullinated peptide were within reference range. A biopsy of a lesion on the back showed psoriasiform dermatitis with confluent parakeratosis and scattered necrotic keratinocytes. There was superficial perivascular inflammation with rare eosinophils (Figure 3).

The patient was treated with a course of systemic corticosteroids. The rash resolved in 1 week. She did not receive the second dose due to the rash.
Two mRNA COVID-19 vaccines—Pfizer BioNTech and Moderna—have been granted emergency use authorization by the US Food and Drug Administration.6 The safety profile of the mRNA-1273 vaccine for the median 2-month follow-up showed no safety concerns.3 Minor localized adverse effects (eg, pain, redness, swelling) have been observed more frequently with the vaccines than with placebo. Systemic symptoms, such as fever, fatigue, headache, and muscle and joint pain, also were seen somewhat more often with the vaccines than with placebo; most such effects occurred 24 to 48 hours after vaccination.3,6,7 The frequency of unsolicited adverse events and serious adverse events reported during the 28-day period after vaccination generally was similar among participants in the vaccine and placebo groups.3
There are 2 types of reactions to COVID-19 vaccination: immediate and delayed. Immediate reactions usually are due to anaphylaxis, requiring prompt recognition and treatment with epinephrine to stop rapid progression of life-threatening symptoms. Delayed reactions include localized reactions, such as urticaria and benign exanthema; serum sickness and serum sickness–like reactions; fever; and rare skin, organ, and neurologic sequelae.1,6-8
Cutaneous manifestations, present in 16% to 50% of patients with Sjögren syndrome, are considered one of the most common extraglandular presentations of the syndrome. They are classified as nonvascular (eg, xerosis, angular cheilitis, eyelid dermatitis, annular erythema) and vascular (eg, Raynaud phenomenon, vasculitis).9-11 Our patient did not have any of those findings. She had not taken any medications before the rash appeared, thereby ruling out a drug reaction.
The differential for our patient included post–urinary tract infection immune-reactive arthritis and rash, which is not typical with Escherichia coli infection but is described with infection with Chlamydia species and Salmonella species. Moreover, post–urinary tract infection immune-reactive arthritis and rash appear mostly on the palms and soles. Systemic lupus erythematosus–like rashes have a different histology and appear on sun-exposed areas; our patient’s rash was found mainly on unexposed areas.12
Because our patient received the Moderna vaccine 5 days before the rash appeared and later developed swelling of the digits with morning stiffness, a delayed serum sickness–like reaction secondary to COVID-19 vaccination was possible.3,6
COVID-19 mRNA vaccines developed by Pfizer-BioNTech and Moderna incorporate a lipid-based nanoparticle carrier system that prevents rapid enzymatic degradation of mRNA and facilitates in vivo delivery of mRNA. This lipid-based nanoparticle carrier system is further stabilized by a polyethylene glycol 2000 lipid conjugate that provides a hydrophilic layer, thus prolonging half-life. The presence of lipid polyethylene glycol 2000 in mRNA vaccines has led to concern that this component could be implicated in anaphylaxis.6
COVID-19 antigens can give rise to varying clinical manifestations that are directly related to viral tissue damage or are indirectly induced by the antiviral immune response.13,14 Hyperactivation of the immune system to eradicate COVID-19 may trigger autoimmunity; several immune-mediated disorders have been described in individuals infected with SARS-CoV-2. Dermal manifestations include cutaneous rash and vasculitis.13-16 Crucial immunologic steps occur during SARS-CoV-2 infection that may link autoimmunity to COVID-19.13,14 In preliminary published data on the efficacy of the Moderna vaccine on 45 trial enrollees, 3 did not receive the second dose of vaccination, including 1 who developed urticaria on both legs 5 days after the first dose.1
Introduction of viral RNA can induce autoimmunity that can be explained by various phenomena, including epitope spreading, molecular mimicry, cryptic antigen, and bystander activation. Remarkably, more than one-third of immunogenic proteins in SARS-CoV-2 have potentially problematic homology to proteins that are key to the human adaptive immune system.5
Moreover, SARS-CoV-2 seems to induce organ injury through alternative mechanisms beyond direct viral infection, including immunologic injury. In some situations, hyperactivation of the immune response to SARS-CoV-2 RNA can result in autoimmune disease. COVID-19 has been associated with immune-mediated systemic or organ-selective manifestations, some of which fulfill the diagnostic or classification criteria of specific autoimmune diseases. It is unclear whether those medical disorders are the result of transitory postinfectious epiphenomena.5
A few studies have shown that patients with rheumatic disease have an incidence and prevalence of COVID-19 that is similar to the general population. A similar pattern has been detected in COVID-19 morbidity and mortality rates, even among patients with an autoimmune disease, such as rheumatoid arthritis and Sjögren syndrome.5,17 Furthermore, exacerbation of preexisting rheumatic symptoms may be due to hyperactivation of antiviral pathways in a person with an autoimmune disease.17-19 The findings in our patient suggested a direct role for the vaccine in skin manifestations, rather than for reactivation or development of new systemic autoimmune processes, such as systemic lupus erythematosus.
Exacerbation of psoriasis following COVID-19 vaccination has been described20; however, the case patient did not have a history of psoriasis. The mechanism(s) of such exacerbation remain unclear; COVID-19 vaccine–induced helper T cells (TH17) may play a role.21 Other skin manifestations encountered following COVID-19 vaccination include lichen planus, leukocytoclastic vasculitic rash, erythema multiforme–like rash, and pityriasis rosea–like rash.22-25 The immune mechanisms of these manifestations remain unclear.
The clinical presentation of delayed vaccination reactions can be attributed to the timing of symptoms and, in this case, the immune-mediated background of a psoriasiform reaction. Although adverse reactions to the SARS-CoV-2 mRNA vaccine are rare, more individuals should be studied after vaccination to confirm and better understand this phenomenon.
- Jackson LA, Anderson EJ, Rouphael NG, et al; . An mRNA vaccine against SARS-CoV-2—preliminary report. N Engl J Med. 2020;383:1920-1931. doi:10.1056/NEJMoa2022483
- Anderson EJ, Rouphael NG, Widge AT, et al; . Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N Engl J Med. 2020;383:2427-2438. doi:10.1056/NEJMoa2028436
- Baden LR, El Sahly HM, Essink B, et al; COVE Study Group. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021;384:403-416. doi:10.1056/NEJMoa2035389
- Weise E. ‘COVID arm’ rash seen after Moderna vaccine annoying but harmless, doctors say. USA Today. January 27, 2021. Accessed September 4, 2022. https://www.usatoday.com/story/news/health/2021/01/27/covid-arm-moderna-vaccine-rash-harmless-side-effect-doctors-say/4277725001/
- Talotta R, Robertson E. Autoimmunity as the comet tail of COVID-19 pandemic. World J Clin Cases. 2020;8:3621-3644. doi:10.12998/wjcc.v8.i17.3621
- Castells MC, Phillips EJ. Maintaining safety with SARS-CoV-2 vaccines. N Engl J Med. 2021;384:643-649. doi:10.1056/NEJMra2035343
- Polack FP, Thomas SJ, Kitchin N, et al; . Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603-2615. doi:10.1056/NEJMoa2034577
- Dooling K, McClung N, Chamberland M, et al. The Advisory Committee on Immunization Practices’ interim recommendation for allocating initial supplies of COVID-19 vaccine—United States, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1857-1859. doi:10.15585/mmwr.mm6949e1
- Roguedas AM, Misery L, Sassolas B, et al. Cutaneous manifestations of primary Sjögren’s syndrome are underestimated. Clin Exp Rheumatol. 2004;22:632-636.
- Katayama I. Dry skin manifestations in Sjögren syndrome and atopic dermatitis related to aberrant sudomotor function in inflammatory allergic skin diseases. Allergol Int. 2018;67:448-454. doi:10.1016/j.alit.2018.07.001
- Generali E, Costanzo A, Mainetti C, et al. Cutaneous and mucosal manifestations of Sjögren’s syndrome. Clin Rev Allergy Immunol. 2017;53:357-370. doi:10.1007/s12016-017-8639-y
- Chanprapaph K, Tankunakorn J, Suchonwanit P, et al. Dermatologic manifestations, histologic features and disease progression among cutaneous lupus erythematosus subtypes: a prospective observational study in Asians. Dermatol Ther (Heidelb). 2021;11:131-147. doi:10.1007/s13555-020-00471-y
- Ortega-Quijano D, Jimenez-Cauhe J, Selda-Enriquez G, et al. Algorithm for the classification of COVID-19 rashes. J Am Acad Dermatol. 2020;83:e103-e104. doi:10.1016/j.jaad.2020.05.034
- Rahimi H, Tehranchinia Z. A comprehensive review of cutaneous manifestations associated with COVID-19. Biomed Res Int. 2020;2020:1236520. doi:10.1155/2020/1236520
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98:75-81. doi:10.1016/j.jdermsci.2020.04.011
- Landa N, Mendieta-Eckert M, Fonda-Pascual P, et al. Chilblain-like lesions on feet and hands during the COVID-19 pandemic. Int J Dermatol. 2020;59:739-743. doi:10.1111/ijd.14937
- Dellavance A, Coelho Andrade LE. Immunologic derangement preceding clinical autoimmunity. Lupus. 2014;23:1305-1308. doi:10.1177/0961203314531346
- Parodi A, Gasparini G, Cozzani E. Could antiphospholipid antibodies contribute to coagulopathy in COVID-19? J Am Acad Dermatol. 2020;83:e249. doi:10.1016/j.jaad.2020.06.003
- Zhou Y, Han T, Chen J, et al. Clinical and autoimmune characteristics of severe and critical cases of COVID-19. Clin Transl Sci. 2020;13:1077-1086. doi:10.1111/cts.12805
- Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010. doi:10.3389/fmed.2021.812010
- Rouai M, Slimane MB, Sassi W, et al. Pustular rash triggered by Pfizer-BioNTech COVID-19 vaccination: a case report. Dermatol Ther. 2022:e15465. doi:10.1111/dth.15465
- Altun E, Kuzucular E. Leukocytoclastic vasculitis after COVID-19 vaccination. Dermatol Ther. 2022;35:e15279. doi:10.1111/dth.15279
- Buckley JE, Landis LN, Rapini RP. Pityriasis rosea-like rash after mRNA COVID-19 vaccination: a case report and review of the literature. JAAD Int. 2022;7:164-168. doi:10.1016/j.jdin.2022.01.009
- Gökçek GE, Öksüm Solak E, Çölgeçen E. Pityriasis rosea like eruption: a dermatological manifestation of Coronavac-COVID-19 vaccine. Dermatol Ther. 2022;35:e15256. doi:10.1111/dth.15256
- Kim MJ, Kim JW, Kim MS, et al. Generalized erythema multiforme-like skin rash following the first dose of COVID-19 vaccine (Pfizer-BioNTech). J Eur Acad Dermatol Venereol. 2022;36:e98-e100. doi:10.1111/jdv.17757
- Jackson LA, Anderson EJ, Rouphael NG, et al; . An mRNA vaccine against SARS-CoV-2—preliminary report. N Engl J Med. 2020;383:1920-1931. doi:10.1056/NEJMoa2022483
- Anderson EJ, Rouphael NG, Widge AT, et al; . Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N Engl J Med. 2020;383:2427-2438. doi:10.1056/NEJMoa2028436
- Baden LR, El Sahly HM, Essink B, et al; COVE Study Group. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021;384:403-416. doi:10.1056/NEJMoa2035389
- Weise E. ‘COVID arm’ rash seen after Moderna vaccine annoying but harmless, doctors say. USA Today. January 27, 2021. Accessed September 4, 2022. https://www.usatoday.com/story/news/health/2021/01/27/covid-arm-moderna-vaccine-rash-harmless-side-effect-doctors-say/4277725001/
- Talotta R, Robertson E. Autoimmunity as the comet tail of COVID-19 pandemic. World J Clin Cases. 2020;8:3621-3644. doi:10.12998/wjcc.v8.i17.3621
- Castells MC, Phillips EJ. Maintaining safety with SARS-CoV-2 vaccines. N Engl J Med. 2021;384:643-649. doi:10.1056/NEJMra2035343
- Polack FP, Thomas SJ, Kitchin N, et al; . Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603-2615. doi:10.1056/NEJMoa2034577
- Dooling K, McClung N, Chamberland M, et al. The Advisory Committee on Immunization Practices’ interim recommendation for allocating initial supplies of COVID-19 vaccine—United States, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1857-1859. doi:10.15585/mmwr.mm6949e1
- Roguedas AM, Misery L, Sassolas B, et al. Cutaneous manifestations of primary Sjögren’s syndrome are underestimated. Clin Exp Rheumatol. 2004;22:632-636.
- Katayama I. Dry skin manifestations in Sjögren syndrome and atopic dermatitis related to aberrant sudomotor function in inflammatory allergic skin diseases. Allergol Int. 2018;67:448-454. doi:10.1016/j.alit.2018.07.001
- Generali E, Costanzo A, Mainetti C, et al. Cutaneous and mucosal manifestations of Sjögren’s syndrome. Clin Rev Allergy Immunol. 2017;53:357-370. doi:10.1007/s12016-017-8639-y
- Chanprapaph K, Tankunakorn J, Suchonwanit P, et al. Dermatologic manifestations, histologic features and disease progression among cutaneous lupus erythematosus subtypes: a prospective observational study in Asians. Dermatol Ther (Heidelb). 2021;11:131-147. doi:10.1007/s13555-020-00471-y
- Ortega-Quijano D, Jimenez-Cauhe J, Selda-Enriquez G, et al. Algorithm for the classification of COVID-19 rashes. J Am Acad Dermatol. 2020;83:e103-e104. doi:10.1016/j.jaad.2020.05.034
- Rahimi H, Tehranchinia Z. A comprehensive review of cutaneous manifestations associated with COVID-19. Biomed Res Int. 2020;2020:1236520. doi:10.1155/2020/1236520
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98:75-81. doi:10.1016/j.jdermsci.2020.04.011
- Landa N, Mendieta-Eckert M, Fonda-Pascual P, et al. Chilblain-like lesions on feet and hands during the COVID-19 pandemic. Int J Dermatol. 2020;59:739-743. doi:10.1111/ijd.14937
- Dellavance A, Coelho Andrade LE. Immunologic derangement preceding clinical autoimmunity. Lupus. 2014;23:1305-1308. doi:10.1177/0961203314531346
- Parodi A, Gasparini G, Cozzani E. Could antiphospholipid antibodies contribute to coagulopathy in COVID-19? J Am Acad Dermatol. 2020;83:e249. doi:10.1016/j.jaad.2020.06.003
- Zhou Y, Han T, Chen J, et al. Clinical and autoimmune characteristics of severe and critical cases of COVID-19. Clin Transl Sci. 2020;13:1077-1086. doi:10.1111/cts.12805
- Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010. doi:10.3389/fmed.2021.812010
- Rouai M, Slimane MB, Sassi W, et al. Pustular rash triggered by Pfizer-BioNTech COVID-19 vaccination: a case report. Dermatol Ther. 2022:e15465. doi:10.1111/dth.15465
- Altun E, Kuzucular E. Leukocytoclastic vasculitis after COVID-19 vaccination. Dermatol Ther. 2022;35:e15279. doi:10.1111/dth.15279
- Buckley JE, Landis LN, Rapini RP. Pityriasis rosea-like rash after mRNA COVID-19 vaccination: a case report and review of the literature. JAAD Int. 2022;7:164-168. doi:10.1016/j.jdin.2022.01.009
- Gökçek GE, Öksüm Solak E, Çölgeçen E. Pityriasis rosea like eruption: a dermatological manifestation of Coronavac-COVID-19 vaccine. Dermatol Ther. 2022;35:e15256. doi:10.1111/dth.15256
- Kim MJ, Kim JW, Kim MS, et al. Generalized erythema multiforme-like skin rash following the first dose of COVID-19 vaccine (Pfizer-BioNTech). J Eur Acad Dermatol Venereol. 2022;36:e98-e100. doi:10.1111/jdv.17757
PRACTICE POINTS
- The differential diagnosis for a new-onset psoriasiform rash in an elderly patient should include a vaccine-related rash.
- A rash following vaccination that necessitates systemic corticosteroid therapy can decrease vaccine efficacy.
Microtox and Mesotox
The terms when they mention one of these terms.
Let’s settle the nomenclature confusion. In this column, I define and outline suggested terminology based on studies and my 15 years of experience using neuromodulators. If any readers or colleagues disagree, please write to me and we can discuss the alternatives in a subsequent article; if you agree, please also write to me so we can collaboratively correct the discrepancies in the literature accordingly.
The term mesotherapy, originating from the Greek “mesos” referring to the early embryonic mesoderm, was identified in the 1950’s by Dr. Michel Pistor, a French physician who administered drugs intradermally. The term was defined as a minimally invasive technique by which drugs or bioactive substances are given in small quantities through dermal micropunctures. Drugs administered intradermally diffuse very slowly and therefore, stay in the tissue longer than those administered intramuscularly.
Thus, Mesotox is defined not by the concentration of the neuromodulator or location, but by the depth of injection in the superficial dermis. It can be delivered through individual injections or through a microneedling pen.
Microtox refers to the dilution of the neuromodulator at concentrations below the proposed dilution guidelines of the manufacturer: Less than 2.5 U per 0.1 mL for onabotulinumtoxinA (OBA), incobotulinumtoxinA (IBA), and prabotulinumtoxinA (PBA); and less than 10 U per 0.1 mL for abobotulinumtoxinA (ABO), This method allows for the injection of superficial cutaneous muscles softening the dynamic rhytids without complete paralysis.
Mesotox is widely used off label for facial lifting, reduction in skin laxity or crepiness, flushing of rosacea, acne, hyperhidrosis of the face, keloids, seborrhea, neck rejuvenation, contouring of the mandibular border, and scalp oiliness. Based on a review of articles using this technique, dilution methods were less than 2.5 U per 1 mL (OBA, IBA) and less than 10 U per 0.1 mL (ABO) depth of injection was the superficial to mid-dermis with injection points 0.5 cm to 1 cm apart.
In a study by Atwa and colleagues, 25 patients with mild facial skin laxity received intradermal Botox-A on one side and saline on the other. This split face study showed a highly significant difference with facial lifting on the treated side. Mesotox injection points vary based on the clinical indication and area being treated.
The treatment of dynamic muscles using standard neuromodulator dosing protocols include the treatment of the glabella, crow’s feet, forehead lines, masseter hypertrophy, bunny lines, gummy smile, perioral lines, mentalis hypertonia, platysmal bands, and marionette lines.
However, hyperdilute neuromodulators or Microtox can effectively be used alone or in combination with standard dosing for the following off-label uses. Used in combination with standard dosing of the forehead lines, I use Microtox in the lateral brow to soften the frontalis muscle without dropping the brow in patients with a low-set brow or lid laxity. I also use it for the jelly roll of the eyes and to open the aperture of the eyes. Along the nose, Microtox can also be used to treat a sagging nasal tip, decrease the width of the ala, and treat overactive facial muscles adjacent to the nose resulting in an overactive nasolabial fold.
Similarly, Microtox can be used to treat lateral smile lines and downward extensions of the crow’s feet. In all of the aforementioned treatment areas, I recommend approximately 0.5-1 U of toxin in each area divided at 1-cm intervals.Mesotox and Microtox are both highly effective strategies to treat the aging face. However, the nomenclature is not interchangeable. I propose that the term Mesotox be used only to articulate or define the superficial injection of a neuromodulator for the improvement of the skin that does not involve the injection into or paralysis of a cutaneous muscle (“tox” being used generically for all neuromodulators). I also propose that the term Microtox should be used to define the dilution of a neuromodulator beyond the manufacturer-recommended dilution protocols – used for the paralysis of a cutaneous muscle. In addition, I recommend that the terms MicroBotox and MesoBotox no longer be used. These procedures all have risks, and adverse events associated with Microtox and Mesotox are similar to those of any neuromodulator injection at FDA-recommended maximum doses, and dilution and storage protocols and proper injection techniques need to be followed. Expertise and training is crucial and treatment by a board-certified dermatologist or plastic surgeon is imperative.
Dr. Talakoub and Naissan O. Wesley, MD, are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to her at [email protected]. Dr. Talakoub had no relevant disclosures.
References
Awaida CJ et al. Plast Reconstr Surg. 2018 Sep;142(3):640-9.
Calvani F et al. Plast Surg (Oakv). 2019 May;27(2):156-61.
Iranmanesh B et al. J Cosmet Dermatol. 2022 Oct;21(10):4160-70.
Kandhari R et al. J Cutan Aesthet Surg. 2022 Apr-Jun;15(2):101-7.
Lewandowski M et al. Molecules. 2022 May 13;27(10):3143.
Mammucari M et al. Eur Rev Med Pharmacol Sci. 2011 Jun;15(6):682-94.
Park KY et al. Ann Dermatol. 2018 Dec;30(6):688-93.
Pistor M. Chir Dent Fr. 1976;46:59-60.
Rho NK, Gil YC. Toxins (Basel). 2021 Nov 19;13(11):817.
Wu WTL. Plast Reconstr Surg. 2015 Nov;136(5 Suppl):92S-100S.
Zhang H et al. Clin Cosmet Investig Dermatol. 2021 Apr 30;14:407-17.
The terms when they mention one of these terms.
Let’s settle the nomenclature confusion. In this column, I define and outline suggested terminology based on studies and my 15 years of experience using neuromodulators. If any readers or colleagues disagree, please write to me and we can discuss the alternatives in a subsequent article; if you agree, please also write to me so we can collaboratively correct the discrepancies in the literature accordingly.
The term mesotherapy, originating from the Greek “mesos” referring to the early embryonic mesoderm, was identified in the 1950’s by Dr. Michel Pistor, a French physician who administered drugs intradermally. The term was defined as a minimally invasive technique by which drugs or bioactive substances are given in small quantities through dermal micropunctures. Drugs administered intradermally diffuse very slowly and therefore, stay in the tissue longer than those administered intramuscularly.
Thus, Mesotox is defined not by the concentration of the neuromodulator or location, but by the depth of injection in the superficial dermis. It can be delivered through individual injections or through a microneedling pen.
Microtox refers to the dilution of the neuromodulator at concentrations below the proposed dilution guidelines of the manufacturer: Less than 2.5 U per 0.1 mL for onabotulinumtoxinA (OBA), incobotulinumtoxinA (IBA), and prabotulinumtoxinA (PBA); and less than 10 U per 0.1 mL for abobotulinumtoxinA (ABO), This method allows for the injection of superficial cutaneous muscles softening the dynamic rhytids without complete paralysis.
Mesotox is widely used off label for facial lifting, reduction in skin laxity or crepiness, flushing of rosacea, acne, hyperhidrosis of the face, keloids, seborrhea, neck rejuvenation, contouring of the mandibular border, and scalp oiliness. Based on a review of articles using this technique, dilution methods were less than 2.5 U per 1 mL (OBA, IBA) and less than 10 U per 0.1 mL (ABO) depth of injection was the superficial to mid-dermis with injection points 0.5 cm to 1 cm apart.
In a study by Atwa and colleagues, 25 patients with mild facial skin laxity received intradermal Botox-A on one side and saline on the other. This split face study showed a highly significant difference with facial lifting on the treated side. Mesotox injection points vary based on the clinical indication and area being treated.
The treatment of dynamic muscles using standard neuromodulator dosing protocols include the treatment of the glabella, crow’s feet, forehead lines, masseter hypertrophy, bunny lines, gummy smile, perioral lines, mentalis hypertonia, platysmal bands, and marionette lines.
However, hyperdilute neuromodulators or Microtox can effectively be used alone or in combination with standard dosing for the following off-label uses. Used in combination with standard dosing of the forehead lines, I use Microtox in the lateral brow to soften the frontalis muscle without dropping the brow in patients with a low-set brow or lid laxity. I also use it for the jelly roll of the eyes and to open the aperture of the eyes. Along the nose, Microtox can also be used to treat a sagging nasal tip, decrease the width of the ala, and treat overactive facial muscles adjacent to the nose resulting in an overactive nasolabial fold.
Similarly, Microtox can be used to treat lateral smile lines and downward extensions of the crow’s feet. In all of the aforementioned treatment areas, I recommend approximately 0.5-1 U of toxin in each area divided at 1-cm intervals.Mesotox and Microtox are both highly effective strategies to treat the aging face. However, the nomenclature is not interchangeable. I propose that the term Mesotox be used only to articulate or define the superficial injection of a neuromodulator for the improvement of the skin that does not involve the injection into or paralysis of a cutaneous muscle (“tox” being used generically for all neuromodulators). I also propose that the term Microtox should be used to define the dilution of a neuromodulator beyond the manufacturer-recommended dilution protocols – used for the paralysis of a cutaneous muscle. In addition, I recommend that the terms MicroBotox and MesoBotox no longer be used. These procedures all have risks, and adverse events associated with Microtox and Mesotox are similar to those of any neuromodulator injection at FDA-recommended maximum doses, and dilution and storage protocols and proper injection techniques need to be followed. Expertise and training is crucial and treatment by a board-certified dermatologist or plastic surgeon is imperative.
Dr. Talakoub and Naissan O. Wesley, MD, are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to her at [email protected]. Dr. Talakoub had no relevant disclosures.
References
Awaida CJ et al. Plast Reconstr Surg. 2018 Sep;142(3):640-9.
Calvani F et al. Plast Surg (Oakv). 2019 May;27(2):156-61.
Iranmanesh B et al. J Cosmet Dermatol. 2022 Oct;21(10):4160-70.
Kandhari R et al. J Cutan Aesthet Surg. 2022 Apr-Jun;15(2):101-7.
Lewandowski M et al. Molecules. 2022 May 13;27(10):3143.
Mammucari M et al. Eur Rev Med Pharmacol Sci. 2011 Jun;15(6):682-94.
Park KY et al. Ann Dermatol. 2018 Dec;30(6):688-93.
Pistor M. Chir Dent Fr. 1976;46:59-60.
Rho NK, Gil YC. Toxins (Basel). 2021 Nov 19;13(11):817.
Wu WTL. Plast Reconstr Surg. 2015 Nov;136(5 Suppl):92S-100S.
Zhang H et al. Clin Cosmet Investig Dermatol. 2021 Apr 30;14:407-17.
The terms when they mention one of these terms.
Let’s settle the nomenclature confusion. In this column, I define and outline suggested terminology based on studies and my 15 years of experience using neuromodulators. If any readers or colleagues disagree, please write to me and we can discuss the alternatives in a subsequent article; if you agree, please also write to me so we can collaboratively correct the discrepancies in the literature accordingly.
The term mesotherapy, originating from the Greek “mesos” referring to the early embryonic mesoderm, was identified in the 1950’s by Dr. Michel Pistor, a French physician who administered drugs intradermally. The term was defined as a minimally invasive technique by which drugs or bioactive substances are given in small quantities through dermal micropunctures. Drugs administered intradermally diffuse very slowly and therefore, stay in the tissue longer than those administered intramuscularly.
Thus, Mesotox is defined not by the concentration of the neuromodulator or location, but by the depth of injection in the superficial dermis. It can be delivered through individual injections or through a microneedling pen.
Microtox refers to the dilution of the neuromodulator at concentrations below the proposed dilution guidelines of the manufacturer: Less than 2.5 U per 0.1 mL for onabotulinumtoxinA (OBA), incobotulinumtoxinA (IBA), and prabotulinumtoxinA (PBA); and less than 10 U per 0.1 mL for abobotulinumtoxinA (ABO), This method allows for the injection of superficial cutaneous muscles softening the dynamic rhytids without complete paralysis.
Mesotox is widely used off label for facial lifting, reduction in skin laxity or crepiness, flushing of rosacea, acne, hyperhidrosis of the face, keloids, seborrhea, neck rejuvenation, contouring of the mandibular border, and scalp oiliness. Based on a review of articles using this technique, dilution methods were less than 2.5 U per 1 mL (OBA, IBA) and less than 10 U per 0.1 mL (ABO) depth of injection was the superficial to mid-dermis with injection points 0.5 cm to 1 cm apart.
In a study by Atwa and colleagues, 25 patients with mild facial skin laxity received intradermal Botox-A on one side and saline on the other. This split face study showed a highly significant difference with facial lifting on the treated side. Mesotox injection points vary based on the clinical indication and area being treated.
The treatment of dynamic muscles using standard neuromodulator dosing protocols include the treatment of the glabella, crow’s feet, forehead lines, masseter hypertrophy, bunny lines, gummy smile, perioral lines, mentalis hypertonia, platysmal bands, and marionette lines.
However, hyperdilute neuromodulators or Microtox can effectively be used alone or in combination with standard dosing for the following off-label uses. Used in combination with standard dosing of the forehead lines, I use Microtox in the lateral brow to soften the frontalis muscle without dropping the brow in patients with a low-set brow or lid laxity. I also use it for the jelly roll of the eyes and to open the aperture of the eyes. Along the nose, Microtox can also be used to treat a sagging nasal tip, decrease the width of the ala, and treat overactive facial muscles adjacent to the nose resulting in an overactive nasolabial fold.
Similarly, Microtox can be used to treat lateral smile lines and downward extensions of the crow’s feet. In all of the aforementioned treatment areas, I recommend approximately 0.5-1 U of toxin in each area divided at 1-cm intervals.Mesotox and Microtox are both highly effective strategies to treat the aging face. However, the nomenclature is not interchangeable. I propose that the term Mesotox be used only to articulate or define the superficial injection of a neuromodulator for the improvement of the skin that does not involve the injection into or paralysis of a cutaneous muscle (“tox” being used generically for all neuromodulators). I also propose that the term Microtox should be used to define the dilution of a neuromodulator beyond the manufacturer-recommended dilution protocols – used for the paralysis of a cutaneous muscle. In addition, I recommend that the terms MicroBotox and MesoBotox no longer be used. These procedures all have risks, and adverse events associated with Microtox and Mesotox are similar to those of any neuromodulator injection at FDA-recommended maximum doses, and dilution and storage protocols and proper injection techniques need to be followed. Expertise and training is crucial and treatment by a board-certified dermatologist or plastic surgeon is imperative.
Dr. Talakoub and Naissan O. Wesley, MD, are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to her at [email protected]. Dr. Talakoub had no relevant disclosures.
References
Awaida CJ et al. Plast Reconstr Surg. 2018 Sep;142(3):640-9.
Calvani F et al. Plast Surg (Oakv). 2019 May;27(2):156-61.
Iranmanesh B et al. J Cosmet Dermatol. 2022 Oct;21(10):4160-70.
Kandhari R et al. J Cutan Aesthet Surg. 2022 Apr-Jun;15(2):101-7.
Lewandowski M et al. Molecules. 2022 May 13;27(10):3143.
Mammucari M et al. Eur Rev Med Pharmacol Sci. 2011 Jun;15(6):682-94.
Park KY et al. Ann Dermatol. 2018 Dec;30(6):688-93.
Pistor M. Chir Dent Fr. 1976;46:59-60.
Rho NK, Gil YC. Toxins (Basel). 2021 Nov 19;13(11):817.
Wu WTL. Plast Reconstr Surg. 2015 Nov;136(5 Suppl):92S-100S.
Zhang H et al. Clin Cosmet Investig Dermatol. 2021 Apr 30;14:407-17.
Ulmus davidiana root extract
Ulmus davidiana, commonly known as yugeunpi, has a long history of use in Korea in treating burns, eczema, frostbite, difficulties in urination, inflammation, and psoriasis,1 and has also been used in China for some of these indications, including skin inflammation.2,3 Currently, there are several areas in which the bioactivity of U. davidiana are under investigation, with numerous potential applications in dermatology. This column focuses briefly on the evidence supporting the traditional uses of the plant and potential new applications.
Anti-inflammatory activity
Eom and colleagues studied the potential of a polysaccharide extract from the root bark of U. davidiana to serve as a suitable cosmetic ingredient for conferring moisturizing, anti-inflammatory, and photoprotective activity. In this 2006 investigation, the composition of the polysaccharide extract was found to be primarily rhamnose, galactose, and glucose. The root extract exhibited a similar humectant moisturizing effect as hyaluronic acid, the researchers reported. The U. davidiana root extract was also found to dose-dependently suppress prostaglandin E2. The inhibition of the release of interleukin-6 and IL-8 was also reported to be significant. The use of the U. davidiana extract also stimulated the recovery of human fibroblasts (two times that of positive control) exposed to UVA irradiation. The researchers suggested that their overall results point to the viability of U. davidiana root extract as a cosmetic agent ingredient to protect skin from UV exposure and the inflammation that follows.2
In 2013, Choi and colleagues found that a methanol extract of the stem and root barks of U. davidiana revealed anti-inflammatory properties, with activity attributed to two trihydroxy acids [then-new trihydroxy fatty acid, 9,12,13-trihydroxyoctadeca-10(Z),15(Z)-dienoic acid, and pinellic acid], both of which blocked prostaglandin D₂ production.4
That same year, Lyu and colleagues studied the antiallergic and anti-inflammatory effects of U. davidiana using a 1-fluoro-2,4-dinitrofluorobenzene (DNFB)–induced contact dermatitis mouse model. They found that treatment at a dose of 10 mg/mL successfully prevented skin lesions caused by consistent DNFB application. Further, the researchers observed that topically applied U. davidiana suppressed spongiosis and reduced total serum immunoglobulin and IgG2a levels. Overall, they concluded that the botanical treatment improved contact dermatitis in mice.1
In 2019, So and colleagues studied the chemical components of U. davidiana root bark (isolating a chromane derivative and 22 known substances) and reported data supporting the traditional use of the root bark for gastroenteric and inflammatory indications.3
Bakuchiol [(1E,3S)-3-ethenyl-3,7-dimethyl-1,6-octadien-1-yl]phenol, a prenylated phenolic monoterpene found in the seeds and leaves of various plants, including U. davidiana, is used for its anti-inflammatory properties in traditional Korean medicine.5 Choi and colleagues determined that bakuchiol exhibited robust anti-inflammatory activity in a study of U. davidiana constituents, at least partially accounting for the anti-inflammatory functions of the plant.5
Antifungal activity
In 2021, Alishir and colleagues conducted a phytochemical analysis of the root bark extract of U. davidiana, resulting in the isolation of 10 substances including the novel coumarin glycoside derivative ulmusakidian. Some of the compounds exhibited antifungal activity against Cryptococcus neoformans, though none demonstrated antifungal activity against Candida albicans.6
Wound dressing
Park and colleagues demonstrated in 2020 that superabsorbing hydrogel wound dressings composed of U. davidiana root bark powders, which exhibit gelling activity, performed effectively in speeding up wound closure and cutaneous regeneration in skin-wound mice models. These dressings also displayed thermal stability and superior mechanical properties to pullulan-only gel films. The researchers concluded that gel films composed of U. davidiana have potential to surpass the effectiveness of current products.7
Anti–hair loss activity
Early in 2022, Kwon and colleagues investigated the anti–hair loss mechanism of U. davidiana and determined that supercritical extraction-residues of U. davidiana significantly hinder the secretion of transforming growth factor–beta but dose dependently salvage insulinlike growth factor 1, and substantially decrease dihydrotestosterone synthesis. They concluded that these U. davidiana supercritical fluid extract residues have the potential to halt the loss of human hair.8
Photoprotective potential
Late in 2020, Her and colleagues reported on their development and analysis of a new distillate derived from a fermented mixture of nine anti-inflammatory herbs including U. davidiana. The investigators assessed the effects of the topically applied distillate on UVB-induced skin damage in Institute of Cancer Research mice, finding significant improvements in the dorsal skin photodamage. Application of the distillate also ameliorated collagen production impairment and diminished proinflammatory cytokine levels of tumor necrosis factor (TNF)–alpha and IL-1B. The researchers concluded that this anti-inflammatory herbal distillate, which includes U. davidiana, displays the potential to serve as a photoprotective agent.9
Antiaging activity
In 2011, Yang and colleagues set out to identify constituent substances of the root bark of U. davidiana that have the capacity to suppress cellular senescence in human fibroblasts and human umbilical vein endothelial cells. They isolated 22 compounds, of which epifriedelanol, ssioriside, and catechin-7-O-beta-D-glucopyranoside impeded adriamycin-induced cellular senescence in human dermal fibroblasts and friedelin, epifriedelanol, and catechin-7-O-beta-apiofuranoside in the umbilical vein endothelial cells. Epifriedelanol was the most potent of the substances, leading the researchers to conclude that this U. davidiana component can diminish cellular senescence in human primary cells and has the potential as an oral and/or topical antiaging agent.10
Also that year, in a study on the protective effects of U. davidiana on UVB-irradiated hairless mice, the authors claimed that an ethanol extract of U. davidiana significantly suppressed wrinkle development in mice chronically exposed to UVB.11 This study showed that U. davidiana extract exerts antioxidant activity as evidenced by a decrease in MMP-1 activity. It also demonstrated antielastase activity. The treated mice showed a decrease in wrinkles as compared with water-treated mice.11 Although this is just one study in mice, it may demonstrate a protective effect on elastic fibers on skin exposed to UVB light.
Late in 2020, Lee and colleagues reported on their study of the possible antiaging effects on the skin of (-)-phenolic compounds isolated from the root bark of U. davidiana. The function of collagenase MMP-1 was found to be inhibited by the isolate (-)-catechin, which also halted collagen degradation caused by TNF-alpha in normal human dermal fibroblasts. Further, the investigators demonstrated that the U. davidiana isolate (-)-catechin reduced the expression of proinflammatory cytokines such as IL-1B and IL-6. They concluded that the U. davidiana isolate exhibits the potential to combat intrinsic as well as extrinsic cutaneous aging.12
These findings are particularly intriguing. There is much overlap between intrinsic and extrinsic aging. If U. davidiana can keep collagen intact and inhibit cellular senescence, it may serve as an early intervention toward slowing or preventing skin aging.
Summary
Of greatest interest now, perhaps, is its potential to impede cellular senescence. Senescent cells release a multitude of inflammatory and other factors that hasten intrinsic aging. Blocking cellular senescence is an important approach to the prevention and treatment of skin aging.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in the office and as an ecommerce solution. Write to her at [email protected].
References
1. Lyu J et al. J Pharmacopuncture. 2013 Jun;16(2):41-5.
2. Eom SY et al. J Cosmet Sci. 2006 Sep-Oct;57(5):355-67.
3. So HM et al. Bioorg Chem. 2019 Oct;91:103145.
4. Choi HG et al. Phytother Res. 2013 Sep;27(9):1376-80.
5. Choi SY et al. J Med Food. 2010 Aug;13(4):1019-23.
6. Alishir A et al. Bioorg Med Chem Lett. 2021 Mar 15;36:127828.
7. Park TH et al. Saudi Pharm J. 2020 Jul;28(7):791-802.
8. Kwon YE et al. Molecules. 2022 Feb 19;27(4):1419.
9. Her Y et al. Molecules. 2020 Dec 29;26(1):124.
10. Yang HH et al. Planta Med. 2011 Mar;77(5):441-9.
11. Kim YO et al. Korean Journal of Medicinal Crop Science. 2011;19(6):508-13.
12. Lee S et al. Antioxidants (Basel). 2020 Oct 13;9(10):981.
Ulmus davidiana, commonly known as yugeunpi, has a long history of use in Korea in treating burns, eczema, frostbite, difficulties in urination, inflammation, and psoriasis,1 and has also been used in China for some of these indications, including skin inflammation.2,3 Currently, there are several areas in which the bioactivity of U. davidiana are under investigation, with numerous potential applications in dermatology. This column focuses briefly on the evidence supporting the traditional uses of the plant and potential new applications.
Anti-inflammatory activity
Eom and colleagues studied the potential of a polysaccharide extract from the root bark of U. davidiana to serve as a suitable cosmetic ingredient for conferring moisturizing, anti-inflammatory, and photoprotective activity. In this 2006 investigation, the composition of the polysaccharide extract was found to be primarily rhamnose, galactose, and glucose. The root extract exhibited a similar humectant moisturizing effect as hyaluronic acid, the researchers reported. The U. davidiana root extract was also found to dose-dependently suppress prostaglandin E2. The inhibition of the release of interleukin-6 and IL-8 was also reported to be significant. The use of the U. davidiana extract also stimulated the recovery of human fibroblasts (two times that of positive control) exposed to UVA irradiation. The researchers suggested that their overall results point to the viability of U. davidiana root extract as a cosmetic agent ingredient to protect skin from UV exposure and the inflammation that follows.2
In 2013, Choi and colleagues found that a methanol extract of the stem and root barks of U. davidiana revealed anti-inflammatory properties, with activity attributed to two trihydroxy acids [then-new trihydroxy fatty acid, 9,12,13-trihydroxyoctadeca-10(Z),15(Z)-dienoic acid, and pinellic acid], both of which blocked prostaglandin D₂ production.4
That same year, Lyu and colleagues studied the antiallergic and anti-inflammatory effects of U. davidiana using a 1-fluoro-2,4-dinitrofluorobenzene (DNFB)–induced contact dermatitis mouse model. They found that treatment at a dose of 10 mg/mL successfully prevented skin lesions caused by consistent DNFB application. Further, the researchers observed that topically applied U. davidiana suppressed spongiosis and reduced total serum immunoglobulin and IgG2a levels. Overall, they concluded that the botanical treatment improved contact dermatitis in mice.1
In 2019, So and colleagues studied the chemical components of U. davidiana root bark (isolating a chromane derivative and 22 known substances) and reported data supporting the traditional use of the root bark for gastroenteric and inflammatory indications.3
Bakuchiol [(1E,3S)-3-ethenyl-3,7-dimethyl-1,6-octadien-1-yl]phenol, a prenylated phenolic monoterpene found in the seeds and leaves of various plants, including U. davidiana, is used for its anti-inflammatory properties in traditional Korean medicine.5 Choi and colleagues determined that bakuchiol exhibited robust anti-inflammatory activity in a study of U. davidiana constituents, at least partially accounting for the anti-inflammatory functions of the plant.5
Antifungal activity
In 2021, Alishir and colleagues conducted a phytochemical analysis of the root bark extract of U. davidiana, resulting in the isolation of 10 substances including the novel coumarin glycoside derivative ulmusakidian. Some of the compounds exhibited antifungal activity against Cryptococcus neoformans, though none demonstrated antifungal activity against Candida albicans.6
Wound dressing
Park and colleagues demonstrated in 2020 that superabsorbing hydrogel wound dressings composed of U. davidiana root bark powders, which exhibit gelling activity, performed effectively in speeding up wound closure and cutaneous regeneration in skin-wound mice models. These dressings also displayed thermal stability and superior mechanical properties to pullulan-only gel films. The researchers concluded that gel films composed of U. davidiana have potential to surpass the effectiveness of current products.7
Anti–hair loss activity
Early in 2022, Kwon and colleagues investigated the anti–hair loss mechanism of U. davidiana and determined that supercritical extraction-residues of U. davidiana significantly hinder the secretion of transforming growth factor–beta but dose dependently salvage insulinlike growth factor 1, and substantially decrease dihydrotestosterone synthesis. They concluded that these U. davidiana supercritical fluid extract residues have the potential to halt the loss of human hair.8
Photoprotective potential
Late in 2020, Her and colleagues reported on their development and analysis of a new distillate derived from a fermented mixture of nine anti-inflammatory herbs including U. davidiana. The investigators assessed the effects of the topically applied distillate on UVB-induced skin damage in Institute of Cancer Research mice, finding significant improvements in the dorsal skin photodamage. Application of the distillate also ameliorated collagen production impairment and diminished proinflammatory cytokine levels of tumor necrosis factor (TNF)–alpha and IL-1B. The researchers concluded that this anti-inflammatory herbal distillate, which includes U. davidiana, displays the potential to serve as a photoprotective agent.9
Antiaging activity
In 2011, Yang and colleagues set out to identify constituent substances of the root bark of U. davidiana that have the capacity to suppress cellular senescence in human fibroblasts and human umbilical vein endothelial cells. They isolated 22 compounds, of which epifriedelanol, ssioriside, and catechin-7-O-beta-D-glucopyranoside impeded adriamycin-induced cellular senescence in human dermal fibroblasts and friedelin, epifriedelanol, and catechin-7-O-beta-apiofuranoside in the umbilical vein endothelial cells. Epifriedelanol was the most potent of the substances, leading the researchers to conclude that this U. davidiana component can diminish cellular senescence in human primary cells and has the potential as an oral and/or topical antiaging agent.10
Also that year, in a study on the protective effects of U. davidiana on UVB-irradiated hairless mice, the authors claimed that an ethanol extract of U. davidiana significantly suppressed wrinkle development in mice chronically exposed to UVB.11 This study showed that U. davidiana extract exerts antioxidant activity as evidenced by a decrease in MMP-1 activity. It also demonstrated antielastase activity. The treated mice showed a decrease in wrinkles as compared with water-treated mice.11 Although this is just one study in mice, it may demonstrate a protective effect on elastic fibers on skin exposed to UVB light.
Late in 2020, Lee and colleagues reported on their study of the possible antiaging effects on the skin of (-)-phenolic compounds isolated from the root bark of U. davidiana. The function of collagenase MMP-1 was found to be inhibited by the isolate (-)-catechin, which also halted collagen degradation caused by TNF-alpha in normal human dermal fibroblasts. Further, the investigators demonstrated that the U. davidiana isolate (-)-catechin reduced the expression of proinflammatory cytokines such as IL-1B and IL-6. They concluded that the U. davidiana isolate exhibits the potential to combat intrinsic as well as extrinsic cutaneous aging.12
These findings are particularly intriguing. There is much overlap between intrinsic and extrinsic aging. If U. davidiana can keep collagen intact and inhibit cellular senescence, it may serve as an early intervention toward slowing or preventing skin aging.
Summary
Of greatest interest now, perhaps, is its potential to impede cellular senescence. Senescent cells release a multitude of inflammatory and other factors that hasten intrinsic aging. Blocking cellular senescence is an important approach to the prevention and treatment of skin aging.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in the office and as an ecommerce solution. Write to her at [email protected].
References
1. Lyu J et al. J Pharmacopuncture. 2013 Jun;16(2):41-5.
2. Eom SY et al. J Cosmet Sci. 2006 Sep-Oct;57(5):355-67.
3. So HM et al. Bioorg Chem. 2019 Oct;91:103145.
4. Choi HG et al. Phytother Res. 2013 Sep;27(9):1376-80.
5. Choi SY et al. J Med Food. 2010 Aug;13(4):1019-23.
6. Alishir A et al. Bioorg Med Chem Lett. 2021 Mar 15;36:127828.
7. Park TH et al. Saudi Pharm J. 2020 Jul;28(7):791-802.
8. Kwon YE et al. Molecules. 2022 Feb 19;27(4):1419.
9. Her Y et al. Molecules. 2020 Dec 29;26(1):124.
10. Yang HH et al. Planta Med. 2011 Mar;77(5):441-9.
11. Kim YO et al. Korean Journal of Medicinal Crop Science. 2011;19(6):508-13.
12. Lee S et al. Antioxidants (Basel). 2020 Oct 13;9(10):981.
Ulmus davidiana, commonly known as yugeunpi, has a long history of use in Korea in treating burns, eczema, frostbite, difficulties in urination, inflammation, and psoriasis,1 and has also been used in China for some of these indications, including skin inflammation.2,3 Currently, there are several areas in which the bioactivity of U. davidiana are under investigation, with numerous potential applications in dermatology. This column focuses briefly on the evidence supporting the traditional uses of the plant and potential new applications.
Anti-inflammatory activity
Eom and colleagues studied the potential of a polysaccharide extract from the root bark of U. davidiana to serve as a suitable cosmetic ingredient for conferring moisturizing, anti-inflammatory, and photoprotective activity. In this 2006 investigation, the composition of the polysaccharide extract was found to be primarily rhamnose, galactose, and glucose. The root extract exhibited a similar humectant moisturizing effect as hyaluronic acid, the researchers reported. The U. davidiana root extract was also found to dose-dependently suppress prostaglandin E2. The inhibition of the release of interleukin-6 and IL-8 was also reported to be significant. The use of the U. davidiana extract also stimulated the recovery of human fibroblasts (two times that of positive control) exposed to UVA irradiation. The researchers suggested that their overall results point to the viability of U. davidiana root extract as a cosmetic agent ingredient to protect skin from UV exposure and the inflammation that follows.2
In 2013, Choi and colleagues found that a methanol extract of the stem and root barks of U. davidiana revealed anti-inflammatory properties, with activity attributed to two trihydroxy acids [then-new trihydroxy fatty acid, 9,12,13-trihydroxyoctadeca-10(Z),15(Z)-dienoic acid, and pinellic acid], both of which blocked prostaglandin D₂ production.4
That same year, Lyu and colleagues studied the antiallergic and anti-inflammatory effects of U. davidiana using a 1-fluoro-2,4-dinitrofluorobenzene (DNFB)–induced contact dermatitis mouse model. They found that treatment at a dose of 10 mg/mL successfully prevented skin lesions caused by consistent DNFB application. Further, the researchers observed that topically applied U. davidiana suppressed spongiosis and reduced total serum immunoglobulin and IgG2a levels. Overall, they concluded that the botanical treatment improved contact dermatitis in mice.1
In 2019, So and colleagues studied the chemical components of U. davidiana root bark (isolating a chromane derivative and 22 known substances) and reported data supporting the traditional use of the root bark for gastroenteric and inflammatory indications.3
Bakuchiol [(1E,3S)-3-ethenyl-3,7-dimethyl-1,6-octadien-1-yl]phenol, a prenylated phenolic monoterpene found in the seeds and leaves of various plants, including U. davidiana, is used for its anti-inflammatory properties in traditional Korean medicine.5 Choi and colleagues determined that bakuchiol exhibited robust anti-inflammatory activity in a study of U. davidiana constituents, at least partially accounting for the anti-inflammatory functions of the plant.5
Antifungal activity
In 2021, Alishir and colleagues conducted a phytochemical analysis of the root bark extract of U. davidiana, resulting in the isolation of 10 substances including the novel coumarin glycoside derivative ulmusakidian. Some of the compounds exhibited antifungal activity against Cryptococcus neoformans, though none demonstrated antifungal activity against Candida albicans.6
Wound dressing
Park and colleagues demonstrated in 2020 that superabsorbing hydrogel wound dressings composed of U. davidiana root bark powders, which exhibit gelling activity, performed effectively in speeding up wound closure and cutaneous regeneration in skin-wound mice models. These dressings also displayed thermal stability and superior mechanical properties to pullulan-only gel films. The researchers concluded that gel films composed of U. davidiana have potential to surpass the effectiveness of current products.7
Anti–hair loss activity
Early in 2022, Kwon and colleagues investigated the anti–hair loss mechanism of U. davidiana and determined that supercritical extraction-residues of U. davidiana significantly hinder the secretion of transforming growth factor–beta but dose dependently salvage insulinlike growth factor 1, and substantially decrease dihydrotestosterone synthesis. They concluded that these U. davidiana supercritical fluid extract residues have the potential to halt the loss of human hair.8
Photoprotective potential
Late in 2020, Her and colleagues reported on their development and analysis of a new distillate derived from a fermented mixture of nine anti-inflammatory herbs including U. davidiana. The investigators assessed the effects of the topically applied distillate on UVB-induced skin damage in Institute of Cancer Research mice, finding significant improvements in the dorsal skin photodamage. Application of the distillate also ameliorated collagen production impairment and diminished proinflammatory cytokine levels of tumor necrosis factor (TNF)–alpha and IL-1B. The researchers concluded that this anti-inflammatory herbal distillate, which includes U. davidiana, displays the potential to serve as a photoprotective agent.9
Antiaging activity
In 2011, Yang and colleagues set out to identify constituent substances of the root bark of U. davidiana that have the capacity to suppress cellular senescence in human fibroblasts and human umbilical vein endothelial cells. They isolated 22 compounds, of which epifriedelanol, ssioriside, and catechin-7-O-beta-D-glucopyranoside impeded adriamycin-induced cellular senescence in human dermal fibroblasts and friedelin, epifriedelanol, and catechin-7-O-beta-apiofuranoside in the umbilical vein endothelial cells. Epifriedelanol was the most potent of the substances, leading the researchers to conclude that this U. davidiana component can diminish cellular senescence in human primary cells and has the potential as an oral and/or topical antiaging agent.10
Also that year, in a study on the protective effects of U. davidiana on UVB-irradiated hairless mice, the authors claimed that an ethanol extract of U. davidiana significantly suppressed wrinkle development in mice chronically exposed to UVB.11 This study showed that U. davidiana extract exerts antioxidant activity as evidenced by a decrease in MMP-1 activity. It also demonstrated antielastase activity. The treated mice showed a decrease in wrinkles as compared with water-treated mice.11 Although this is just one study in mice, it may demonstrate a protective effect on elastic fibers on skin exposed to UVB light.
Late in 2020, Lee and colleagues reported on their study of the possible antiaging effects on the skin of (-)-phenolic compounds isolated from the root bark of U. davidiana. The function of collagenase MMP-1 was found to be inhibited by the isolate (-)-catechin, which also halted collagen degradation caused by TNF-alpha in normal human dermal fibroblasts. Further, the investigators demonstrated that the U. davidiana isolate (-)-catechin reduced the expression of proinflammatory cytokines such as IL-1B and IL-6. They concluded that the U. davidiana isolate exhibits the potential to combat intrinsic as well as extrinsic cutaneous aging.12
These findings are particularly intriguing. There is much overlap between intrinsic and extrinsic aging. If U. davidiana can keep collagen intact and inhibit cellular senescence, it may serve as an early intervention toward slowing or preventing skin aging.
Summary
Of greatest interest now, perhaps, is its potential to impede cellular senescence. Senescent cells release a multitude of inflammatory and other factors that hasten intrinsic aging. Blocking cellular senescence is an important approach to the prevention and treatment of skin aging.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in the office and as an ecommerce solution. Write to her at [email protected].
References
1. Lyu J et al. J Pharmacopuncture. 2013 Jun;16(2):41-5.
2. Eom SY et al. J Cosmet Sci. 2006 Sep-Oct;57(5):355-67.
3. So HM et al. Bioorg Chem. 2019 Oct;91:103145.
4. Choi HG et al. Phytother Res. 2013 Sep;27(9):1376-80.
5. Choi SY et al. J Med Food. 2010 Aug;13(4):1019-23.
6. Alishir A et al. Bioorg Med Chem Lett. 2021 Mar 15;36:127828.
7. Park TH et al. Saudi Pharm J. 2020 Jul;28(7):791-802.
8. Kwon YE et al. Molecules. 2022 Feb 19;27(4):1419.
9. Her Y et al. Molecules. 2020 Dec 29;26(1):124.
10. Yang HH et al. Planta Med. 2011 Mar;77(5):441-9.
11. Kim YO et al. Korean Journal of Medicinal Crop Science. 2011;19(6):508-13.
12. Lee S et al. Antioxidants (Basel). 2020 Oct 13;9(10):981.
Combination therapy shows mixed results for scleroderma-related lung disease
PHILADELPHIA – Combining the immunomodulatory agent mycophenolate with the antifibrotic pirfenidone led to more rapid improvement and showed a trend to be more effective than mycophenolate mofetil alone for treating the signs and symptoms of scleroderma-related interstitial lung disease, but the combination therapy came with an increase in side effects, according to results from the Scleroderma Lung Study III.
Dinesh Khanna, MBBS, MSc, of the University of Michigan, Ann Arbor, presented the results at the annual meeting of the American College of Rheumatology. He noted some problems with the study – namely its small size, enrolling only 51 patients, about one-third of its original goal. But he also said it showed a potential signal for efficacy and that the study itself could serve as a “template” for future studies of combination mycophenolate mofetil (MMF) plus pirfenidone therapy for scleroderma-related interstitial lung disease (SSc-ILD).
“The pirfenidone patients had quite a bit more GI side effects and photosensitivity, and those are known side effects,” Dr. Khanna said in an interview. “So the combination therapy had more side effects but trends to higher efficacy.”
The design of SLS-III, a phase 2 clinical trial, was a challenge, Dr. Khanna explained. The goal was to enroll 150 SSc-ILD patients who hadn’t had any previous treatment for their disease. Finding those patients proved difficult. “In fact, if you look at the recent history, 70% of the patients with early diffuse scleroderma are on MMF,” he said in his presentation. Compounding low study enrollment was the intervening COVID-19 pandemic, he added.
Testing a faster-acting combination
Nonetheless, the trial managed to enroll 27 patients in the combination therapy group and 24 in the MMF-plus-placebo group and compared their outcomes over 18 months. Study dosing was 1,500 mg MMF twice daily and pirfenidone 801 mg three times daily, titrated to the tolerable dose.
Despite the study’s being underpowered, Dr. Khanna said, it still reported some notable outcomes that merit further investigation. “I think what was intriguing in the study was the long-term benefit in the patient-reported outcomes and the structural changes,” he said in the interview.
Among those notable outcomes was a clinically significant change in forced vital capacity (FVC) percentage for the combination vs. the placebo groups: 2.24% vs. 2.09%. He also noted that the combination group saw a somewhat more robust improvement in FVC at six months: 2.59% (± 0.98%) vs. 0.92% (± 1.1%) in the placebo group.
The combination group showed greater improvements in high-resolution computed tomography-evaluated lung involvement and lung fibrosis and patient-reported outcomes, including a statistically significant 3.67-point greater improvement in PROMIS-29 physical function score (4.42 vs. 0.75).
The patients on combination therapy had higher rates of serious adverse events (SAEs), and seven discontinued one or both study drugs early, all in the combined arm. Four combination therapy patients had six SAEs, compared to two placebo patients with three SAEs. In the combination group, SAEs included chest pain, herpes zoster ophthalmicus, nodular basal cell cancer, marginal zone B cell lymphoma, renal crisis, and dyspnea. SAEs in the placebo group were colitis, COVID-19 and hypoxic respiratory failure.
Study design challenges
Nonetheless, Dr. Khanna said the SLS-III data are consistent with the SLS-II findings, with mean improvements in FVC of 2.24% and 2.1%, respectively.
“The next study may be able to replicate what we tried to do, keeping in mind that there are really no MMF-naive patients who are walking around,” Dr. Khanna said. “So the challenge is about the feasibility of recruiting within a trial vs. trying to show a statistical difference between the drug and placebo.”
This study could serve as a foundation for future studies of MMF in patients with SSc-ILD, Robert Spiera, MD, of the Hospital for Special Surgery in New York, said in an interview. “There are lessons to be learned both from the study but also from prior studies looking at MMF use in the background in patients treated with other drugs in clinical trials,” he said.
Dr. Spiera noted that the study had other challenges besides the difficulty in recruiting patients who hadn’t been on MMF therapy. “A great challenge is that the benefit with regard to the impact on the lungs from MMF seems most prominent in the first 6 months to a year to even 2 years that somebody is on the drug,” he said.
The other challenge with this study is that a large proportion of patients had limited systemic disease and relatively lower levels of skin disease compared with other studies of patients on MMF, Dr. Spiera said.
“The optimal treatment of scleroderma-associated lung disease remains a very important and not-adequately met need,” he said. “Particularly, we’re looking for drugs that are tolerable in a patient population that are very prone to GI side effects in general. This study and others have taught us a lot about trial design, and I think more globally this will allow us to move this field forward.”
Dr. Khanna disclosed relationships with Actelion, Boehringer Ingelheim, Bristol-Myers Squibb, CSL Behring, Horizon Therapeutics USA, Janssen Global Services, Prometheus Biosciences, Mitsubishi Tanabe Pharma Corp., Genentech/Roche, Theraly, and Pfizer. Genentech provided funding for the study and pirfenidone and placebo drugs at no cost.
Dr. Spiera disclosed relationships with GlaxoSmithKline, Boehringer-Ingelheim, Corbus Pharmaceutical, InflaRx, AbbVie/Abbott, Sanofi, Novartis, Chemocentryx, Roche and Vera.
PHILADELPHIA – Combining the immunomodulatory agent mycophenolate with the antifibrotic pirfenidone led to more rapid improvement and showed a trend to be more effective than mycophenolate mofetil alone for treating the signs and symptoms of scleroderma-related interstitial lung disease, but the combination therapy came with an increase in side effects, according to results from the Scleroderma Lung Study III.
Dinesh Khanna, MBBS, MSc, of the University of Michigan, Ann Arbor, presented the results at the annual meeting of the American College of Rheumatology. He noted some problems with the study – namely its small size, enrolling only 51 patients, about one-third of its original goal. But he also said it showed a potential signal for efficacy and that the study itself could serve as a “template” for future studies of combination mycophenolate mofetil (MMF) plus pirfenidone therapy for scleroderma-related interstitial lung disease (SSc-ILD).
“The pirfenidone patients had quite a bit more GI side effects and photosensitivity, and those are known side effects,” Dr. Khanna said in an interview. “So the combination therapy had more side effects but trends to higher efficacy.”
The design of SLS-III, a phase 2 clinical trial, was a challenge, Dr. Khanna explained. The goal was to enroll 150 SSc-ILD patients who hadn’t had any previous treatment for their disease. Finding those patients proved difficult. “In fact, if you look at the recent history, 70% of the patients with early diffuse scleroderma are on MMF,” he said in his presentation. Compounding low study enrollment was the intervening COVID-19 pandemic, he added.
Testing a faster-acting combination
Nonetheless, the trial managed to enroll 27 patients in the combination therapy group and 24 in the MMF-plus-placebo group and compared their outcomes over 18 months. Study dosing was 1,500 mg MMF twice daily and pirfenidone 801 mg three times daily, titrated to the tolerable dose.
Despite the study’s being underpowered, Dr. Khanna said, it still reported some notable outcomes that merit further investigation. “I think what was intriguing in the study was the long-term benefit in the patient-reported outcomes and the structural changes,” he said in the interview.
Among those notable outcomes was a clinically significant change in forced vital capacity (FVC) percentage for the combination vs. the placebo groups: 2.24% vs. 2.09%. He also noted that the combination group saw a somewhat more robust improvement in FVC at six months: 2.59% (± 0.98%) vs. 0.92% (± 1.1%) in the placebo group.
The combination group showed greater improvements in high-resolution computed tomography-evaluated lung involvement and lung fibrosis and patient-reported outcomes, including a statistically significant 3.67-point greater improvement in PROMIS-29 physical function score (4.42 vs. 0.75).
The patients on combination therapy had higher rates of serious adverse events (SAEs), and seven discontinued one or both study drugs early, all in the combined arm. Four combination therapy patients had six SAEs, compared to two placebo patients with three SAEs. In the combination group, SAEs included chest pain, herpes zoster ophthalmicus, nodular basal cell cancer, marginal zone B cell lymphoma, renal crisis, and dyspnea. SAEs in the placebo group were colitis, COVID-19 and hypoxic respiratory failure.
Study design challenges
Nonetheless, Dr. Khanna said the SLS-III data are consistent with the SLS-II findings, with mean improvements in FVC of 2.24% and 2.1%, respectively.
“The next study may be able to replicate what we tried to do, keeping in mind that there are really no MMF-naive patients who are walking around,” Dr. Khanna said. “So the challenge is about the feasibility of recruiting within a trial vs. trying to show a statistical difference between the drug and placebo.”
This study could serve as a foundation for future studies of MMF in patients with SSc-ILD, Robert Spiera, MD, of the Hospital for Special Surgery in New York, said in an interview. “There are lessons to be learned both from the study but also from prior studies looking at MMF use in the background in patients treated with other drugs in clinical trials,” he said.
Dr. Spiera noted that the study had other challenges besides the difficulty in recruiting patients who hadn’t been on MMF therapy. “A great challenge is that the benefit with regard to the impact on the lungs from MMF seems most prominent in the first 6 months to a year to even 2 years that somebody is on the drug,” he said.
The other challenge with this study is that a large proportion of patients had limited systemic disease and relatively lower levels of skin disease compared with other studies of patients on MMF, Dr. Spiera said.
“The optimal treatment of scleroderma-associated lung disease remains a very important and not-adequately met need,” he said. “Particularly, we’re looking for drugs that are tolerable in a patient population that are very prone to GI side effects in general. This study and others have taught us a lot about trial design, and I think more globally this will allow us to move this field forward.”
Dr. Khanna disclosed relationships with Actelion, Boehringer Ingelheim, Bristol-Myers Squibb, CSL Behring, Horizon Therapeutics USA, Janssen Global Services, Prometheus Biosciences, Mitsubishi Tanabe Pharma Corp., Genentech/Roche, Theraly, and Pfizer. Genentech provided funding for the study and pirfenidone and placebo drugs at no cost.
Dr. Spiera disclosed relationships with GlaxoSmithKline, Boehringer-Ingelheim, Corbus Pharmaceutical, InflaRx, AbbVie/Abbott, Sanofi, Novartis, Chemocentryx, Roche and Vera.
PHILADELPHIA – Combining the immunomodulatory agent mycophenolate with the antifibrotic pirfenidone led to more rapid improvement and showed a trend to be more effective than mycophenolate mofetil alone for treating the signs and symptoms of scleroderma-related interstitial lung disease, but the combination therapy came with an increase in side effects, according to results from the Scleroderma Lung Study III.
Dinesh Khanna, MBBS, MSc, of the University of Michigan, Ann Arbor, presented the results at the annual meeting of the American College of Rheumatology. He noted some problems with the study – namely its small size, enrolling only 51 patients, about one-third of its original goal. But he also said it showed a potential signal for efficacy and that the study itself could serve as a “template” for future studies of combination mycophenolate mofetil (MMF) plus pirfenidone therapy for scleroderma-related interstitial lung disease (SSc-ILD).
“The pirfenidone patients had quite a bit more GI side effects and photosensitivity, and those are known side effects,” Dr. Khanna said in an interview. “So the combination therapy had more side effects but trends to higher efficacy.”
The design of SLS-III, a phase 2 clinical trial, was a challenge, Dr. Khanna explained. The goal was to enroll 150 SSc-ILD patients who hadn’t had any previous treatment for their disease. Finding those patients proved difficult. “In fact, if you look at the recent history, 70% of the patients with early diffuse scleroderma are on MMF,” he said in his presentation. Compounding low study enrollment was the intervening COVID-19 pandemic, he added.
Testing a faster-acting combination
Nonetheless, the trial managed to enroll 27 patients in the combination therapy group and 24 in the MMF-plus-placebo group and compared their outcomes over 18 months. Study dosing was 1,500 mg MMF twice daily and pirfenidone 801 mg three times daily, titrated to the tolerable dose.
Despite the study’s being underpowered, Dr. Khanna said, it still reported some notable outcomes that merit further investigation. “I think what was intriguing in the study was the long-term benefit in the patient-reported outcomes and the structural changes,” he said in the interview.
Among those notable outcomes was a clinically significant change in forced vital capacity (FVC) percentage for the combination vs. the placebo groups: 2.24% vs. 2.09%. He also noted that the combination group saw a somewhat more robust improvement in FVC at six months: 2.59% (± 0.98%) vs. 0.92% (± 1.1%) in the placebo group.
The combination group showed greater improvements in high-resolution computed tomography-evaluated lung involvement and lung fibrosis and patient-reported outcomes, including a statistically significant 3.67-point greater improvement in PROMIS-29 physical function score (4.42 vs. 0.75).
The patients on combination therapy had higher rates of serious adverse events (SAEs), and seven discontinued one or both study drugs early, all in the combined arm. Four combination therapy patients had six SAEs, compared to two placebo patients with three SAEs. In the combination group, SAEs included chest pain, herpes zoster ophthalmicus, nodular basal cell cancer, marginal zone B cell lymphoma, renal crisis, and dyspnea. SAEs in the placebo group were colitis, COVID-19 and hypoxic respiratory failure.
Study design challenges
Nonetheless, Dr. Khanna said the SLS-III data are consistent with the SLS-II findings, with mean improvements in FVC of 2.24% and 2.1%, respectively.
“The next study may be able to replicate what we tried to do, keeping in mind that there are really no MMF-naive patients who are walking around,” Dr. Khanna said. “So the challenge is about the feasibility of recruiting within a trial vs. trying to show a statistical difference between the drug and placebo.”
This study could serve as a foundation for future studies of MMF in patients with SSc-ILD, Robert Spiera, MD, of the Hospital for Special Surgery in New York, said in an interview. “There are lessons to be learned both from the study but also from prior studies looking at MMF use in the background in patients treated with other drugs in clinical trials,” he said.
Dr. Spiera noted that the study had other challenges besides the difficulty in recruiting patients who hadn’t been on MMF therapy. “A great challenge is that the benefit with regard to the impact on the lungs from MMF seems most prominent in the first 6 months to a year to even 2 years that somebody is on the drug,” he said.
The other challenge with this study is that a large proportion of patients had limited systemic disease and relatively lower levels of skin disease compared with other studies of patients on MMF, Dr. Spiera said.
“The optimal treatment of scleroderma-associated lung disease remains a very important and not-adequately met need,” he said. “Particularly, we’re looking for drugs that are tolerable in a patient population that are very prone to GI side effects in general. This study and others have taught us a lot about trial design, and I think more globally this will allow us to move this field forward.”
Dr. Khanna disclosed relationships with Actelion, Boehringer Ingelheim, Bristol-Myers Squibb, CSL Behring, Horizon Therapeutics USA, Janssen Global Services, Prometheus Biosciences, Mitsubishi Tanabe Pharma Corp., Genentech/Roche, Theraly, and Pfizer. Genentech provided funding for the study and pirfenidone and placebo drugs at no cost.
Dr. Spiera disclosed relationships with GlaxoSmithKline, Boehringer-Ingelheim, Corbus Pharmaceutical, InflaRx, AbbVie/Abbott, Sanofi, Novartis, Chemocentryx, Roche and Vera.
AT ACR 2022
Starting a podcast
In my last column, I discussed . At this writing (November 2022), more than 600 million blogs are online, compared with about 2 million podcasts, and relatively few of them are run by physicians. With podcasts, you have a better chance of standing out in a crowded online world.
Starting a podcast is not difficult, but there are several steps you need to go through before launching one.
As with blogging, start by outlining a long-range plan. Your general topic will probably be your specialty, but you will need to narrow your focus to a few specific subjects, such as the problems you see most often, or a subspecialty that you concentrate on. You can always expand your topic later, as you get more popular. Choose a name for your podcast, and purchase a domain name that accurately describes it.
You will also need to choose a hosting service. Numerous inexpensive hosting platforms are available, and a simple Google search will find them for you. Many of them provide free learning materials, helpful creative tools, and customer support to get you through the confusing technical aspects. They can also help you choose a music introduction (to add a bit of polish), and help you piece together your audio segments. Buzzsprout, RSS.com, and Podbean get good reviews on many sites. (As always, I have no financial interest in any company or service mentioned herein.)
Hosting services can assist you in creating a template – a framework that you can reuse each time you record an episode – containing your intro and exit music, tracks for your conversations, etc. This will make your podcasts instantly recognizable each time your listeners tune in.
Many podcasting experts recommend recruiting a co-host. This can be an associate within your practice, a friend who practices elsewhere, or perhaps a resident in an academic setting. You will be able to spread the workload of creating, editing, and promoting. Plus, it is much easier to generate interesting content when two people are having a conversation, rather than one person lecturing from a prepared script. You might also consider having multiple co-hosts, either to expand episodes into group discussions, or to take turns working with you in covering different subjects.
How long you make your podcast is entirely up to you. Some consultants recommend specific time frames, such as 5 minutes (because that’s an average attention span), or 28 minutes (because that’s the average driving commute time). There are short podcasts and long ones; whatever works for you is fine, as long as you don’t drift off the topic. Furthermore, no one says they must all be the same length; when you are finished talking, you are done. And no one says you must stick with one subject throughout. Combining several short segments might hold more listeners’ interest and will make it easier to share small clips on social media.
Content guidelines are similar to those for blogs. Give people content that will be of interest or benefit to them. Talk about subjects – medical and otherwise – that are relevant to your practice or are prominent in the news.
As with blogs, try to avoid polarizing political discussions, and while it’s fine to discuss treatments and procedures that you offer, aggressive solicitation tends to make viewers look elsewhere. Keep any medical advice in general terms; don’t portray any specific patients as examples.
When your podcast is ready, your hosting platform will show you how to submit it to iTunes, and how to submit your podcast RSS feed to other podcast directories. As you upload new episodes, your host will automatically update your RSS feed, so that any directory you are listed on will receive the new episode.
Once you are uploaded, you can use your host’s social sharing tools to spread the word. As with blogs, use social media, such as your practice’s Facebook page, to push podcast updates into patients’ feeds and track relevant Twitter hashtags to find online communities that might be interested in your subject matter. You should also find your episode embed code (which your host will have) and place it in a prominent place on your website so patients can listen directly from there.
Transcriptions are another excellent promotional tool. Search engines will “read” your podcasts and list them in searches. Some podcast hosts will do transcribing for a fee, but there are independent transcription services as well.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
In my last column, I discussed . At this writing (November 2022), more than 600 million blogs are online, compared with about 2 million podcasts, and relatively few of them are run by physicians. With podcasts, you have a better chance of standing out in a crowded online world.
Starting a podcast is not difficult, but there are several steps you need to go through before launching one.
As with blogging, start by outlining a long-range plan. Your general topic will probably be your specialty, but you will need to narrow your focus to a few specific subjects, such as the problems you see most often, or a subspecialty that you concentrate on. You can always expand your topic later, as you get more popular. Choose a name for your podcast, and purchase a domain name that accurately describes it.
You will also need to choose a hosting service. Numerous inexpensive hosting platforms are available, and a simple Google search will find them for you. Many of them provide free learning materials, helpful creative tools, and customer support to get you through the confusing technical aspects. They can also help you choose a music introduction (to add a bit of polish), and help you piece together your audio segments. Buzzsprout, RSS.com, and Podbean get good reviews on many sites. (As always, I have no financial interest in any company or service mentioned herein.)
Hosting services can assist you in creating a template – a framework that you can reuse each time you record an episode – containing your intro and exit music, tracks for your conversations, etc. This will make your podcasts instantly recognizable each time your listeners tune in.
Many podcasting experts recommend recruiting a co-host. This can be an associate within your practice, a friend who practices elsewhere, or perhaps a resident in an academic setting. You will be able to spread the workload of creating, editing, and promoting. Plus, it is much easier to generate interesting content when two people are having a conversation, rather than one person lecturing from a prepared script. You might also consider having multiple co-hosts, either to expand episodes into group discussions, or to take turns working with you in covering different subjects.
How long you make your podcast is entirely up to you. Some consultants recommend specific time frames, such as 5 minutes (because that’s an average attention span), or 28 minutes (because that’s the average driving commute time). There are short podcasts and long ones; whatever works for you is fine, as long as you don’t drift off the topic. Furthermore, no one says they must all be the same length; when you are finished talking, you are done. And no one says you must stick with one subject throughout. Combining several short segments might hold more listeners’ interest and will make it easier to share small clips on social media.
Content guidelines are similar to those for blogs. Give people content that will be of interest or benefit to them. Talk about subjects – medical and otherwise – that are relevant to your practice or are prominent in the news.
As with blogs, try to avoid polarizing political discussions, and while it’s fine to discuss treatments and procedures that you offer, aggressive solicitation tends to make viewers look elsewhere. Keep any medical advice in general terms; don’t portray any specific patients as examples.
When your podcast is ready, your hosting platform will show you how to submit it to iTunes, and how to submit your podcast RSS feed to other podcast directories. As you upload new episodes, your host will automatically update your RSS feed, so that any directory you are listed on will receive the new episode.
Once you are uploaded, you can use your host’s social sharing tools to spread the word. As with blogs, use social media, such as your practice’s Facebook page, to push podcast updates into patients’ feeds and track relevant Twitter hashtags to find online communities that might be interested in your subject matter. You should also find your episode embed code (which your host will have) and place it in a prominent place on your website so patients can listen directly from there.
Transcriptions are another excellent promotional tool. Search engines will “read” your podcasts and list them in searches. Some podcast hosts will do transcribing for a fee, but there are independent transcription services as well.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
In my last column, I discussed . At this writing (November 2022), more than 600 million blogs are online, compared with about 2 million podcasts, and relatively few of them are run by physicians. With podcasts, you have a better chance of standing out in a crowded online world.
Starting a podcast is not difficult, but there are several steps you need to go through before launching one.
As with blogging, start by outlining a long-range plan. Your general topic will probably be your specialty, but you will need to narrow your focus to a few specific subjects, such as the problems you see most often, or a subspecialty that you concentrate on. You can always expand your topic later, as you get more popular. Choose a name for your podcast, and purchase a domain name that accurately describes it.
You will also need to choose a hosting service. Numerous inexpensive hosting platforms are available, and a simple Google search will find them for you. Many of them provide free learning materials, helpful creative tools, and customer support to get you through the confusing technical aspects. They can also help you choose a music introduction (to add a bit of polish), and help you piece together your audio segments. Buzzsprout, RSS.com, and Podbean get good reviews on many sites. (As always, I have no financial interest in any company or service mentioned herein.)
Hosting services can assist you in creating a template – a framework that you can reuse each time you record an episode – containing your intro and exit music, tracks for your conversations, etc. This will make your podcasts instantly recognizable each time your listeners tune in.
Many podcasting experts recommend recruiting a co-host. This can be an associate within your practice, a friend who practices elsewhere, or perhaps a resident in an academic setting. You will be able to spread the workload of creating, editing, and promoting. Plus, it is much easier to generate interesting content when two people are having a conversation, rather than one person lecturing from a prepared script. You might also consider having multiple co-hosts, either to expand episodes into group discussions, or to take turns working with you in covering different subjects.
How long you make your podcast is entirely up to you. Some consultants recommend specific time frames, such as 5 minutes (because that’s an average attention span), or 28 minutes (because that’s the average driving commute time). There are short podcasts and long ones; whatever works for you is fine, as long as you don’t drift off the topic. Furthermore, no one says they must all be the same length; when you are finished talking, you are done. And no one says you must stick with one subject throughout. Combining several short segments might hold more listeners’ interest and will make it easier to share small clips on social media.
Content guidelines are similar to those for blogs. Give people content that will be of interest or benefit to them. Talk about subjects – medical and otherwise – that are relevant to your practice or are prominent in the news.
As with blogs, try to avoid polarizing political discussions, and while it’s fine to discuss treatments and procedures that you offer, aggressive solicitation tends to make viewers look elsewhere. Keep any medical advice in general terms; don’t portray any specific patients as examples.
When your podcast is ready, your hosting platform will show you how to submit it to iTunes, and how to submit your podcast RSS feed to other podcast directories. As you upload new episodes, your host will automatically update your RSS feed, so that any directory you are listed on will receive the new episode.
Once you are uploaded, you can use your host’s social sharing tools to spread the word. As with blogs, use social media, such as your practice’s Facebook page, to push podcast updates into patients’ feeds and track relevant Twitter hashtags to find online communities that might be interested in your subject matter. You should also find your episode embed code (which your host will have) and place it in a prominent place on your website so patients can listen directly from there.
Transcriptions are another excellent promotional tool. Search engines will “read” your podcasts and list them in searches. Some podcast hosts will do transcribing for a fee, but there are independent transcription services as well.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Infant anaphylaxis: Study characterizes symptoms, treatment
LOUISVILLE, KY. – , research findings indicate.
Given that early administration of epinephrine can be potentially lifesaving for infants with anaphylaxis, the study highlighted the real-world successes in increased uptake of treatment in this vulnerable patient population.
Most infants in the study who presented to the ED and received epinephrine were able to be discharged home after just a few hours, with only 1 out of 10 requiring hospitalization.
The study also reported that most symptoms were in the skin/mucosal, gastrointestinal, respiratory, and cardiovascular (CV) systems, providing improved characterization of anaphylaxis symptoms in the infant population.
Nearly “all episodes were triggered by food – especially egg, peanut, milk, and cashew,” commented Colleen Shannon, MD, a pediatrician at Children’s Hospital of Philadelphia, who presented the research findings at the annual meeting of the American College of Allergy, Asthma, and Immunology.
Dr. Shannon noted that despite previous research demonstrating age-based differences in the presentation of anaphylaxis, the symptomatology of anaphylaxis in infants has not been robustly characterized. Better characterization of anaphylaxis in infants with allergies may help ensure earlier and more accurate diagnosis and management, she said.
For the study, the researchers performed a retrospective chart review of 169 patients between 0 and 24 months of age (mean age, 1.0 years) who presented to the emergency department of a pediatric tertiary referral center between 2019 and 2022.
All patients in the study met diagnostic criteria for anaphylaxis. The investigators used the medical records of patients to evaluate for demographics, as well as presenting symptoms and treatment.
More than half (56.2%) of infants in the study were 12 months of age or younger, and 64.5% were male.
Nearly all (96.5%) anaphylaxis episodes presenting to the ED were triggered by food. The most common foods triggering these episodes were egg (26.6%), peanut (25.4%), milk (13.6%), and cashew (10.1%).
Most symptoms involved the skin/mucosal (97.6%) and GI (74.6%) systems, followed by respiratory (56.8%) and CV (34.3%) systems. Isolated tachycardia was recorded in 84.5% of patients with CV-related symptoms.
Epinephrine was administered to 86.4% of infants who presented to the ED with anaphylaxis. Nearly a third (30.1%) of these infants received epinephrine before arriving to the ED, and 9.5% required more than 1 dose.
The researchers also found that 10.1% of patients required hospital admission, but none had symptoms severe enough to require intensive care.
Jennifer Hoffmann, MD, an emergency medicine physician at the Lurie Children’s Hospital of Chicago, told this news organization that while characterizing anaphylaxis symptoms is relevant for clinicians, it also remains vitally important “to teach parents of infants how to recognize the signs of anaphylaxis, particularly as they begin to introduce new foods,” to ensure timely treatment.
She added that since most infants in the study improved after a single dose of epinephrine, most infants presenting to the ED with anaphylaxis can therefore be safely discharged home after only a brief period of observation. “That is, age alone should not be a reason for admission,” explained Dr. Hoffmann, who wasn’t involved in the research study.
The study was independently supported. Dr. Shannon and Dr. Hoffmann report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LOUISVILLE, KY. – , research findings indicate.
Given that early administration of epinephrine can be potentially lifesaving for infants with anaphylaxis, the study highlighted the real-world successes in increased uptake of treatment in this vulnerable patient population.
Most infants in the study who presented to the ED and received epinephrine were able to be discharged home after just a few hours, with only 1 out of 10 requiring hospitalization.
The study also reported that most symptoms were in the skin/mucosal, gastrointestinal, respiratory, and cardiovascular (CV) systems, providing improved characterization of anaphylaxis symptoms in the infant population.
Nearly “all episodes were triggered by food – especially egg, peanut, milk, and cashew,” commented Colleen Shannon, MD, a pediatrician at Children’s Hospital of Philadelphia, who presented the research findings at the annual meeting of the American College of Allergy, Asthma, and Immunology.
Dr. Shannon noted that despite previous research demonstrating age-based differences in the presentation of anaphylaxis, the symptomatology of anaphylaxis in infants has not been robustly characterized. Better characterization of anaphylaxis in infants with allergies may help ensure earlier and more accurate diagnosis and management, she said.
For the study, the researchers performed a retrospective chart review of 169 patients between 0 and 24 months of age (mean age, 1.0 years) who presented to the emergency department of a pediatric tertiary referral center between 2019 and 2022.
All patients in the study met diagnostic criteria for anaphylaxis. The investigators used the medical records of patients to evaluate for demographics, as well as presenting symptoms and treatment.
More than half (56.2%) of infants in the study were 12 months of age or younger, and 64.5% were male.
Nearly all (96.5%) anaphylaxis episodes presenting to the ED were triggered by food. The most common foods triggering these episodes were egg (26.6%), peanut (25.4%), milk (13.6%), and cashew (10.1%).
Most symptoms involved the skin/mucosal (97.6%) and GI (74.6%) systems, followed by respiratory (56.8%) and CV (34.3%) systems. Isolated tachycardia was recorded in 84.5% of patients with CV-related symptoms.
Epinephrine was administered to 86.4% of infants who presented to the ED with anaphylaxis. Nearly a third (30.1%) of these infants received epinephrine before arriving to the ED, and 9.5% required more than 1 dose.
The researchers also found that 10.1% of patients required hospital admission, but none had symptoms severe enough to require intensive care.
Jennifer Hoffmann, MD, an emergency medicine physician at the Lurie Children’s Hospital of Chicago, told this news organization that while characterizing anaphylaxis symptoms is relevant for clinicians, it also remains vitally important “to teach parents of infants how to recognize the signs of anaphylaxis, particularly as they begin to introduce new foods,” to ensure timely treatment.
She added that since most infants in the study improved after a single dose of epinephrine, most infants presenting to the ED with anaphylaxis can therefore be safely discharged home after only a brief period of observation. “That is, age alone should not be a reason for admission,” explained Dr. Hoffmann, who wasn’t involved in the research study.
The study was independently supported. Dr. Shannon and Dr. Hoffmann report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LOUISVILLE, KY. – , research findings indicate.
Given that early administration of epinephrine can be potentially lifesaving for infants with anaphylaxis, the study highlighted the real-world successes in increased uptake of treatment in this vulnerable patient population.
Most infants in the study who presented to the ED and received epinephrine were able to be discharged home after just a few hours, with only 1 out of 10 requiring hospitalization.
The study also reported that most symptoms were in the skin/mucosal, gastrointestinal, respiratory, and cardiovascular (CV) systems, providing improved characterization of anaphylaxis symptoms in the infant population.
Nearly “all episodes were triggered by food – especially egg, peanut, milk, and cashew,” commented Colleen Shannon, MD, a pediatrician at Children’s Hospital of Philadelphia, who presented the research findings at the annual meeting of the American College of Allergy, Asthma, and Immunology.
Dr. Shannon noted that despite previous research demonstrating age-based differences in the presentation of anaphylaxis, the symptomatology of anaphylaxis in infants has not been robustly characterized. Better characterization of anaphylaxis in infants with allergies may help ensure earlier and more accurate diagnosis and management, she said.
For the study, the researchers performed a retrospective chart review of 169 patients between 0 and 24 months of age (mean age, 1.0 years) who presented to the emergency department of a pediatric tertiary referral center between 2019 and 2022.
All patients in the study met diagnostic criteria for anaphylaxis. The investigators used the medical records of patients to evaluate for demographics, as well as presenting symptoms and treatment.
More than half (56.2%) of infants in the study were 12 months of age or younger, and 64.5% were male.
Nearly all (96.5%) anaphylaxis episodes presenting to the ED were triggered by food. The most common foods triggering these episodes were egg (26.6%), peanut (25.4%), milk (13.6%), and cashew (10.1%).
Most symptoms involved the skin/mucosal (97.6%) and GI (74.6%) systems, followed by respiratory (56.8%) and CV (34.3%) systems. Isolated tachycardia was recorded in 84.5% of patients with CV-related symptoms.
Epinephrine was administered to 86.4% of infants who presented to the ED with anaphylaxis. Nearly a third (30.1%) of these infants received epinephrine before arriving to the ED, and 9.5% required more than 1 dose.
The researchers also found that 10.1% of patients required hospital admission, but none had symptoms severe enough to require intensive care.
Jennifer Hoffmann, MD, an emergency medicine physician at the Lurie Children’s Hospital of Chicago, told this news organization that while characterizing anaphylaxis symptoms is relevant for clinicians, it also remains vitally important “to teach parents of infants how to recognize the signs of anaphylaxis, particularly as they begin to introduce new foods,” to ensure timely treatment.
She added that since most infants in the study improved after a single dose of epinephrine, most infants presenting to the ED with anaphylaxis can therefore be safely discharged home after only a brief period of observation. “That is, age alone should not be a reason for admission,” explained Dr. Hoffmann, who wasn’t involved in the research study.
The study was independently supported. Dr. Shannon and Dr. Hoffmann report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACAAI
Lower hydroxychloroquine dose for lupus tied to hospitalizations for flares
PHILADELPHIA – Patients with systemic lupus erythematosus treated with lower doses of hydroxychloroquine (HCQ) had an increased risk for hospitalization for flares, according to study results presented during a press conference at the annual meeting of the American College of Rheumatology.
HCQ is a cornerstone treatment for SLE as it has been shown to increase survival and decrease disease flares.
Doses decreased with changing guidelines
Guidelines over the years have recommended decreasing doses of HCQ. In 2011, ophthalmology guidelines recommended limiting HCQ dosing to 6.5 mg/kg per day or less of ideal body weight to reduce the chance of retinopathy. For many patients, this required a dose lower than 400 mg/day, an amount frequently used to treat lupus.
In 2016, updated guidelines further lowered the dosage of HCQ, recommending 5 mg/kg or less of patient’s actual body weight.
The effects that lower dosing has had on SLE-associated hospitalizations was unknown, which inspired Dr. Nestor’s research.
The team conducted a case-crossover study within the Mass General Brigham SLE cohort.
Hospitalizations studied over a decade
Dr. Nestor and colleagues identified patients with SLE (via electronic health records) who had at least one visit for SLE and were prescribed HCQ between January 2011 and December 2021, the period over which the recommendations were made.
They identified patients who had been hospitalized during that decade with SLE as the primary discharge diagnosis.
Patients were excluded if they had non-SLE indications, such as kidney transplant or infection without a concomitant SLE flare.
Of 2,971 patients with SLE who used HCQ, 576 had at least one hospitalization with primary discharge diagnosis of SLE.
Of these, 108 were hospitalized for an SLE flare and had used HCQ prior to that hospitalization and had at least one control period with HCQ use during the study period.
All of the patients in the study had to have a case period and a control period, Dr. Nestor explained. The case period was 6 months on HCQ ending in hospitalization for lupus and the control period was 6 months on HCQ that did not end in hospitalization for lupus.
Significantly increased hospitalizations
Low-dose HCQ by weight-based dose (≤ 5 vs. > 5 mg/kg per day) and by non–weight-based dose (< 400 vs. 400 mg per day) were both associated with significantly increased hospitalizations for SLE (adjusted odds ratio, 4.41; 95% confidence interval, 1.50-12.98; and AOR, 3.48; 95% CI, 1.33-9.13, respectively).
The average age of the hospitalized group was 36 years. Most patients (92%) were female, 43.5% were White, and 32.4% were Black.
In calling for reassessment of the dosing, Dr. Nestor said, “We are protecting our patients against a very long-term side effect of hydroxychloroquine retinopathy. [It] typically takes 10-20 years to develop in our patients. But by doing that, we’re missing many of the short-term benefits from hydroxychloroquine in our patients, leading to more lupus flares, which leads to more end-organ damage.”
She said patients taking HCQ for lupus are asked to see an ophthalmologist once a year to monitor for the side effect, adding that rheumatologists and ophthalmologists could work together to adjust the guidelines.
Dr. Nestor suggested it’s possible that patients need higher doses of HCQ earlier in their disease and lower doses later. “Perhaps it’s just the patients who are particularly active who need the higher doses,” she said.
“I don’t think this question is settled,” he told this news organization. “The 5 mg/kg dose recommendation was based on terms of safety but not of effectiveness. We don’t know what the effective dose of HCQ is, and this study shows that low dose is less effective.”
He agreed there needs to be a risk/benefit balance, but noted, “HCQ retinopathy is very rare and we have great tools to screen for it.”
Study limitations include incomplete information on whether patients adhered to treatment plans and reasons for using lower-dose HCQ.
The study authors and Dr. Duarte Garcia report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – Patients with systemic lupus erythematosus treated with lower doses of hydroxychloroquine (HCQ) had an increased risk for hospitalization for flares, according to study results presented during a press conference at the annual meeting of the American College of Rheumatology.
HCQ is a cornerstone treatment for SLE as it has been shown to increase survival and decrease disease flares.
Doses decreased with changing guidelines
Guidelines over the years have recommended decreasing doses of HCQ. In 2011, ophthalmology guidelines recommended limiting HCQ dosing to 6.5 mg/kg per day or less of ideal body weight to reduce the chance of retinopathy. For many patients, this required a dose lower than 400 mg/day, an amount frequently used to treat lupus.
In 2016, updated guidelines further lowered the dosage of HCQ, recommending 5 mg/kg or less of patient’s actual body weight.
The effects that lower dosing has had on SLE-associated hospitalizations was unknown, which inspired Dr. Nestor’s research.
The team conducted a case-crossover study within the Mass General Brigham SLE cohort.
Hospitalizations studied over a decade
Dr. Nestor and colleagues identified patients with SLE (via electronic health records) who had at least one visit for SLE and were prescribed HCQ between January 2011 and December 2021, the period over which the recommendations were made.
They identified patients who had been hospitalized during that decade with SLE as the primary discharge diagnosis.
Patients were excluded if they had non-SLE indications, such as kidney transplant or infection without a concomitant SLE flare.
Of 2,971 patients with SLE who used HCQ, 576 had at least one hospitalization with primary discharge diagnosis of SLE.
Of these, 108 were hospitalized for an SLE flare and had used HCQ prior to that hospitalization and had at least one control period with HCQ use during the study period.
All of the patients in the study had to have a case period and a control period, Dr. Nestor explained. The case period was 6 months on HCQ ending in hospitalization for lupus and the control period was 6 months on HCQ that did not end in hospitalization for lupus.
Significantly increased hospitalizations
Low-dose HCQ by weight-based dose (≤ 5 vs. > 5 mg/kg per day) and by non–weight-based dose (< 400 vs. 400 mg per day) were both associated with significantly increased hospitalizations for SLE (adjusted odds ratio, 4.41; 95% confidence interval, 1.50-12.98; and AOR, 3.48; 95% CI, 1.33-9.13, respectively).
The average age of the hospitalized group was 36 years. Most patients (92%) were female, 43.5% were White, and 32.4% were Black.
In calling for reassessment of the dosing, Dr. Nestor said, “We are protecting our patients against a very long-term side effect of hydroxychloroquine retinopathy. [It] typically takes 10-20 years to develop in our patients. But by doing that, we’re missing many of the short-term benefits from hydroxychloroquine in our patients, leading to more lupus flares, which leads to more end-organ damage.”
She said patients taking HCQ for lupus are asked to see an ophthalmologist once a year to monitor for the side effect, adding that rheumatologists and ophthalmologists could work together to adjust the guidelines.
Dr. Nestor suggested it’s possible that patients need higher doses of HCQ earlier in their disease and lower doses later. “Perhaps it’s just the patients who are particularly active who need the higher doses,” she said.
“I don’t think this question is settled,” he told this news organization. “The 5 mg/kg dose recommendation was based on terms of safety but not of effectiveness. We don’t know what the effective dose of HCQ is, and this study shows that low dose is less effective.”
He agreed there needs to be a risk/benefit balance, but noted, “HCQ retinopathy is very rare and we have great tools to screen for it.”
Study limitations include incomplete information on whether patients adhered to treatment plans and reasons for using lower-dose HCQ.
The study authors and Dr. Duarte Garcia report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – Patients with systemic lupus erythematosus treated with lower doses of hydroxychloroquine (HCQ) had an increased risk for hospitalization for flares, according to study results presented during a press conference at the annual meeting of the American College of Rheumatology.
HCQ is a cornerstone treatment for SLE as it has been shown to increase survival and decrease disease flares.
Doses decreased with changing guidelines
Guidelines over the years have recommended decreasing doses of HCQ. In 2011, ophthalmology guidelines recommended limiting HCQ dosing to 6.5 mg/kg per day or less of ideal body weight to reduce the chance of retinopathy. For many patients, this required a dose lower than 400 mg/day, an amount frequently used to treat lupus.
In 2016, updated guidelines further lowered the dosage of HCQ, recommending 5 mg/kg or less of patient’s actual body weight.
The effects that lower dosing has had on SLE-associated hospitalizations was unknown, which inspired Dr. Nestor’s research.
The team conducted a case-crossover study within the Mass General Brigham SLE cohort.
Hospitalizations studied over a decade
Dr. Nestor and colleagues identified patients with SLE (via electronic health records) who had at least one visit for SLE and were prescribed HCQ between January 2011 and December 2021, the period over which the recommendations were made.
They identified patients who had been hospitalized during that decade with SLE as the primary discharge diagnosis.
Patients were excluded if they had non-SLE indications, such as kidney transplant or infection without a concomitant SLE flare.
Of 2,971 patients with SLE who used HCQ, 576 had at least one hospitalization with primary discharge diagnosis of SLE.
Of these, 108 were hospitalized for an SLE flare and had used HCQ prior to that hospitalization and had at least one control period with HCQ use during the study period.
All of the patients in the study had to have a case period and a control period, Dr. Nestor explained. The case period was 6 months on HCQ ending in hospitalization for lupus and the control period was 6 months on HCQ that did not end in hospitalization for lupus.
Significantly increased hospitalizations
Low-dose HCQ by weight-based dose (≤ 5 vs. > 5 mg/kg per day) and by non–weight-based dose (< 400 vs. 400 mg per day) were both associated with significantly increased hospitalizations for SLE (adjusted odds ratio, 4.41; 95% confidence interval, 1.50-12.98; and AOR, 3.48; 95% CI, 1.33-9.13, respectively).
The average age of the hospitalized group was 36 years. Most patients (92%) were female, 43.5% were White, and 32.4% were Black.
In calling for reassessment of the dosing, Dr. Nestor said, “We are protecting our patients against a very long-term side effect of hydroxychloroquine retinopathy. [It] typically takes 10-20 years to develop in our patients. But by doing that, we’re missing many of the short-term benefits from hydroxychloroquine in our patients, leading to more lupus flares, which leads to more end-organ damage.”
She said patients taking HCQ for lupus are asked to see an ophthalmologist once a year to monitor for the side effect, adding that rheumatologists and ophthalmologists could work together to adjust the guidelines.
Dr. Nestor suggested it’s possible that patients need higher doses of HCQ earlier in their disease and lower doses later. “Perhaps it’s just the patients who are particularly active who need the higher doses,” she said.
“I don’t think this question is settled,” he told this news organization. “The 5 mg/kg dose recommendation was based on terms of safety but not of effectiveness. We don’t know what the effective dose of HCQ is, and this study shows that low dose is less effective.”
He agreed there needs to be a risk/benefit balance, but noted, “HCQ retinopathy is very rare and we have great tools to screen for it.”
Study limitations include incomplete information on whether patients adhered to treatment plans and reasons for using lower-dose HCQ.
The study authors and Dr. Duarte Garcia report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACR 2022
Remibrutinib safe for Sjögren’s in phase 2
PHILADELPHIA – Oral remibrutinib was well tolerated and had a good safety profile over 24 weeks among patients with moderate to severe Sjögren syndrome (SS), according to new phase 2 data presented at the annual meeting of the American College of Rheumatology.
Thomas Dörner, MD, with the department of rheumatology and clinical immunology at Charité Universitätsmedizin Berlin, presented the data from the double-blind, randomized, placebo-controlled, proof-of-concept study.
The authors said the results of the study suggest that remibrutinib, a highly specific inhibitor of Bruton tyrosine kinase, has the potential to become the first effective oral disease-modifying therapy for SS.
The 73 participants in the study had moderate to severe SS. The baseline EULAR Sjögren Syndrome Disease Activity Index (ESSDAI) score was at least 5, EULAR Sjögren Syndrome Patient Reported Index (ESSPRI) was at least 5, and anti-Ro/SSA antibody positivity was 3 months or less before screening. The patients’ unstimulated whole salivary flow rate was > 0 mL/min.
Overall, 73 patients (71 women) were randomly assigned to receive either remibrutinib 100 mg twice a day (n = 24), remibrutinib 100 mg four times a day (n = 25), or placebo (n = 24) between August 2019 and May 2021.
Remibrutinib met the primary endpoint and resulted in a statistically significant improvement in ESSDAI score for both regimens combined compared with placebo at week 24 (ESSDAI, –2.86).
Patient-reported outcomes similar to placebo
Patient-reported outcomes, including scores on ESSPRI, Functional Assessment of Chronic Illness Therapy–Fatigue, and EuroQol-5 Dimension, were similar in the treatment groups and the placebo group.
“All of the patients, including the placebo patients, improved over the time of the study,” Dr. Dörner said.
The average age of the patients was 51.8 years (range, 18-75 years). Groups were generally balanced with regard to demographic qualities and disease severity at baseline, and the patients represented the SS population well, Dr. Dörner said.
No severe adverse events were reported. Infections were the most frequently reported adverse events, and the rates were similar with the study drug and placebo. No notable liver abnormalities were reported in any of the groups.
Chrisanna Dobrowolski, MD, assistant professor of medicine at Icahn School of Medicine at Mount Sinai in New York, told this news organization, “Preliminary results are promising, but they failed to show improvements in patient-reported quality-of-life measures.
“Having statistical improvements in disease activity measures without clinically meaningful improvement in patient quality of life may limit the value of this treatment,” she said.
Dr. Dobrowolski added that the follow-up period of 6 months is short, and larger studies over a longer period are needed to better assess the effect on patients’ quality of life.
“Regardless, this is the first oral medication which has shown disease-modifying potential for the glandular symptoms of SS and is an exciting new avenue of investigation to be further explored,” she said.
Patients with SS 15 to 20 times more likely to develop B-cell lymphoma as a life-threatening complication. SS is a systemic autoimmune disease characterized by B-cell hyperactivation, lymphoid infiltration, progressive destruction of exocrine glands, and various complications outside the glands, the study authors wrote in the abstract.
Nearly 4 million in U.S. live with the disease
Nearly 4 million people in the United States live with the disease. Common symptoms include light sensitivity, dry eye, dry mouth, fatigue, and joint pain.
SS can be difficult to diagnose because the symptoms vary from person to person and can be confused with those caused by other diseases.
Ardy Fenando, MD, a rheumatology fellow with the University of Kansas Medical Center, said in an interview, “We need more therapies for Sjögren’s. Heterogeneity complicates the way we set the primary endpoints. Therefore, we haven’t had a proven treatment for Sjögren’s. This is supported by previous RCTs [randomized controlled trials] that failed to meet the primary end points.”
Dr. Dörner has relationships with AbbVie, Eli Lilly, Roche/Genentech, Janssen, Novartis, Bristol-Myers Squibb), and UCB. Other authors have various relationships with industry. Dr. Fenando and Dr. Dobrowolski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – Oral remibrutinib was well tolerated and had a good safety profile over 24 weeks among patients with moderate to severe Sjögren syndrome (SS), according to new phase 2 data presented at the annual meeting of the American College of Rheumatology.
Thomas Dörner, MD, with the department of rheumatology and clinical immunology at Charité Universitätsmedizin Berlin, presented the data from the double-blind, randomized, placebo-controlled, proof-of-concept study.
The authors said the results of the study suggest that remibrutinib, a highly specific inhibitor of Bruton tyrosine kinase, has the potential to become the first effective oral disease-modifying therapy for SS.
The 73 participants in the study had moderate to severe SS. The baseline EULAR Sjögren Syndrome Disease Activity Index (ESSDAI) score was at least 5, EULAR Sjögren Syndrome Patient Reported Index (ESSPRI) was at least 5, and anti-Ro/SSA antibody positivity was 3 months or less before screening. The patients’ unstimulated whole salivary flow rate was > 0 mL/min.
Overall, 73 patients (71 women) were randomly assigned to receive either remibrutinib 100 mg twice a day (n = 24), remibrutinib 100 mg four times a day (n = 25), or placebo (n = 24) between August 2019 and May 2021.
Remibrutinib met the primary endpoint and resulted in a statistically significant improvement in ESSDAI score for both regimens combined compared with placebo at week 24 (ESSDAI, –2.86).
Patient-reported outcomes similar to placebo
Patient-reported outcomes, including scores on ESSPRI, Functional Assessment of Chronic Illness Therapy–Fatigue, and EuroQol-5 Dimension, were similar in the treatment groups and the placebo group.
“All of the patients, including the placebo patients, improved over the time of the study,” Dr. Dörner said.
The average age of the patients was 51.8 years (range, 18-75 years). Groups were generally balanced with regard to demographic qualities and disease severity at baseline, and the patients represented the SS population well, Dr. Dörner said.
No severe adverse events were reported. Infections were the most frequently reported adverse events, and the rates were similar with the study drug and placebo. No notable liver abnormalities were reported in any of the groups.
Chrisanna Dobrowolski, MD, assistant professor of medicine at Icahn School of Medicine at Mount Sinai in New York, told this news organization, “Preliminary results are promising, but they failed to show improvements in patient-reported quality-of-life measures.
“Having statistical improvements in disease activity measures without clinically meaningful improvement in patient quality of life may limit the value of this treatment,” she said.
Dr. Dobrowolski added that the follow-up period of 6 months is short, and larger studies over a longer period are needed to better assess the effect on patients’ quality of life.
“Regardless, this is the first oral medication which has shown disease-modifying potential for the glandular symptoms of SS and is an exciting new avenue of investigation to be further explored,” she said.
Patients with SS 15 to 20 times more likely to develop B-cell lymphoma as a life-threatening complication. SS is a systemic autoimmune disease characterized by B-cell hyperactivation, lymphoid infiltration, progressive destruction of exocrine glands, and various complications outside the glands, the study authors wrote in the abstract.
Nearly 4 million in U.S. live with the disease
Nearly 4 million people in the United States live with the disease. Common symptoms include light sensitivity, dry eye, dry mouth, fatigue, and joint pain.
SS can be difficult to diagnose because the symptoms vary from person to person and can be confused with those caused by other diseases.
Ardy Fenando, MD, a rheumatology fellow with the University of Kansas Medical Center, said in an interview, “We need more therapies for Sjögren’s. Heterogeneity complicates the way we set the primary endpoints. Therefore, we haven’t had a proven treatment for Sjögren’s. This is supported by previous RCTs [randomized controlled trials] that failed to meet the primary end points.”
Dr. Dörner has relationships with AbbVie, Eli Lilly, Roche/Genentech, Janssen, Novartis, Bristol-Myers Squibb), and UCB. Other authors have various relationships with industry. Dr. Fenando and Dr. Dobrowolski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – Oral remibrutinib was well tolerated and had a good safety profile over 24 weeks among patients with moderate to severe Sjögren syndrome (SS), according to new phase 2 data presented at the annual meeting of the American College of Rheumatology.
Thomas Dörner, MD, with the department of rheumatology and clinical immunology at Charité Universitätsmedizin Berlin, presented the data from the double-blind, randomized, placebo-controlled, proof-of-concept study.
The authors said the results of the study suggest that remibrutinib, a highly specific inhibitor of Bruton tyrosine kinase, has the potential to become the first effective oral disease-modifying therapy for SS.
The 73 participants in the study had moderate to severe SS. The baseline EULAR Sjögren Syndrome Disease Activity Index (ESSDAI) score was at least 5, EULAR Sjögren Syndrome Patient Reported Index (ESSPRI) was at least 5, and anti-Ro/SSA antibody positivity was 3 months or less before screening. The patients’ unstimulated whole salivary flow rate was > 0 mL/min.
Overall, 73 patients (71 women) were randomly assigned to receive either remibrutinib 100 mg twice a day (n = 24), remibrutinib 100 mg four times a day (n = 25), or placebo (n = 24) between August 2019 and May 2021.
Remibrutinib met the primary endpoint and resulted in a statistically significant improvement in ESSDAI score for both regimens combined compared with placebo at week 24 (ESSDAI, –2.86).
Patient-reported outcomes similar to placebo
Patient-reported outcomes, including scores on ESSPRI, Functional Assessment of Chronic Illness Therapy–Fatigue, and EuroQol-5 Dimension, were similar in the treatment groups and the placebo group.
“All of the patients, including the placebo patients, improved over the time of the study,” Dr. Dörner said.
The average age of the patients was 51.8 years (range, 18-75 years). Groups were generally balanced with regard to demographic qualities and disease severity at baseline, and the patients represented the SS population well, Dr. Dörner said.
No severe adverse events were reported. Infections were the most frequently reported adverse events, and the rates were similar with the study drug and placebo. No notable liver abnormalities were reported in any of the groups.
Chrisanna Dobrowolski, MD, assistant professor of medicine at Icahn School of Medicine at Mount Sinai in New York, told this news organization, “Preliminary results are promising, but they failed to show improvements in patient-reported quality-of-life measures.
“Having statistical improvements in disease activity measures without clinically meaningful improvement in patient quality of life may limit the value of this treatment,” she said.
Dr. Dobrowolski added that the follow-up period of 6 months is short, and larger studies over a longer period are needed to better assess the effect on patients’ quality of life.
“Regardless, this is the first oral medication which has shown disease-modifying potential for the glandular symptoms of SS and is an exciting new avenue of investigation to be further explored,” she said.
Patients with SS 15 to 20 times more likely to develop B-cell lymphoma as a life-threatening complication. SS is a systemic autoimmune disease characterized by B-cell hyperactivation, lymphoid infiltration, progressive destruction of exocrine glands, and various complications outside the glands, the study authors wrote in the abstract.
Nearly 4 million in U.S. live with the disease
Nearly 4 million people in the United States live with the disease. Common symptoms include light sensitivity, dry eye, dry mouth, fatigue, and joint pain.
SS can be difficult to diagnose because the symptoms vary from person to person and can be confused with those caused by other diseases.
Ardy Fenando, MD, a rheumatology fellow with the University of Kansas Medical Center, said in an interview, “We need more therapies for Sjögren’s. Heterogeneity complicates the way we set the primary endpoints. Therefore, we haven’t had a proven treatment for Sjögren’s. This is supported by previous RCTs [randomized controlled trials] that failed to meet the primary end points.”
Dr. Dörner has relationships with AbbVie, Eli Lilly, Roche/Genentech, Janssen, Novartis, Bristol-Myers Squibb), and UCB. Other authors have various relationships with industry. Dr. Fenando and Dr. Dobrowolski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACR 2022
First recommendations for cancer screening in myositis issued
AT ACR 2022
PHILADELPHIA – The first consensus screening guidelines for patients with idiopathic inflammatory myopathy (IIM) provide recommendations on risk stratification for individuals, basic and enhanced screening protocols, and screening frequency.
The recommendations, issued by the International Myositis Assessment and Clinical Studies Group (IMACS), stratify cancer risk for individual patients into low, intermediate, or high categories based on the IIM disease subtype, autoantibody status, and clinical features, reported Alexander Oldroyd, PhD, MSc, MBChB of the University of Manchester, England.
“There’s a big unmet need for cancer screening. One in four adults with myositis has cancer, either 3 years before or after a diagnosis of myositis. It’s one of the leading causes of death in these patients, and they’re overwhelmingly diagnosed at a late stage, so we need standardized approaches to get early diagnosis,” he said in an interview at the annual meeting of the American College of Rheumatology.
Sharon Kolasinski, MD, of the University of Pennsylvania in Philadelphia, said in an interview that the guideline is a welcome development for rheumatologists. Dr. Kolasinski moderated the session where Dr. Oldroyd described the guideline, but she was not involved in its formulation.
“I think that we all have wondered for a very long time: What is the optimal cancer screening for myositis patients? We all worry that the onset of their diseases is associated with a coincident cancer, or that they will develop it soon,” she said.
Dr. Oldroyd emphasized that all patients with myositis have elevated risk for cancer compared with the general population and that the guideline categories of low, intermediate, and high are relative only to patients with IIM.
International consensus
The data on which the recommendations are based come from a systematic review and meta-analysis by Dr. Oldroyd and colleagues of 69 studies on cancer risk factors and 9 on IIM-specific cancer screening.
The authors of that paper found that the dermatomyositis subtype, older age, male sex, dysphagia, cutaneous ulceration and antitranscriptional intermediary factor-1 gamma (anti-TIF1-gamma) positivity were associated with significantly increased risk of cancer.
In contrast, polymyositis and clinically amyopathic dermatomyositis subtypes, Raynaud’s phenomenon, interstitial lung disease, very high serum creatine kinase or lactate dehydrogenase levels, and positivity for anti-Jo1 or anti-EJ antibodies were associated with significantly reduced risk of cancer.
The consensus recommendations were developed with anonymous contributions from 75 expert participants in 22 countries, with additional input from 3 patient partners.
Do this
The guideline lists 18 recommendations, of which 13 are strong and 5 are conditional.
An example of a strong recommendation is number 3, based on a moderate level of evidences:
“All adult IIM patients, irrespective of cancer risk, should continue to participate in country/region-specific age and sex appropriate cancer screening programs,” the guideline recommends.
Patients with verified inclusion body myositis or juvenile-onset IIM do not, however, require routine screening for myositis-associated cancer, the guideline says (recommendations 1 and 2).
There are also recommendations that all adults with new-onset IIM be tested for myositis-specific and myositis-associated autoantibodies to assist in stratifying patients by risk category.
The guideline divides screening recommendations into basic and enhanced. The basic screening should include a comprehensive history and physical exam, complete blood count, liver functions tests, erythrocyte sedimentation rates/plasma viscosity, serum protein electrophoresis, urinalysis, and chest x-ray.
Adults with IIM who are determined to be at low risk for IIM-related cancer should have basic cancer screening at the time of IIM diagnosis. Adults with intermediate risk should undergo both basic and enhanced screening at the time of IIM diagnosis, and those with high risk should undergo enhanced screening at the time of myositis diagnosis, with basic screening annually for 3 years, the recommendations say.
Consider doing this
Conditional recommendations (“clinicians should consider ...”) include the use of PET/CT for adults at high risk for cancer when an underlying cancer has not been detected at the time of IIM diagnosis. They also include a single screening test for anti-TIF1-gamma positive dermatomyositis patients whose disease onset was after age 40 and who have at least one additional risk factor.
Also conditionally recommended are upper and lower gastrointestinal endoscopy for patients at high risk when an underlying cancer is not found at the time of IIM diagnosis, nasoendoscopy in geographical regions with elevated risk for nasopharyngeal cancers, and screening for all IIM patients with red-flag symptoms or clinical features of cancer, including unexplained weight loss, family history of cancer, smoking, unexplained fever, or night sweats.
Guided steps
“I think clinicians have a lot of questions such as, ‘well, what should I do, when should I do it?’ These are important clinical questions, and we need guidance about this. We need to balance comprehensiveness with cost-effectiveness, and we need expert opinion about what steps we should take now and which should we take later,” Dr. Kolasinski said.
The guideline development process was supported by the University of Manchester, IMACS, National Institute for Health Research (United Kingdom), National Institutes of Health, National Health Service Northern Care Alliance, The Myositis Association, Myositis UK, University of Pittsburgh, Versus Arthritis, and the Center for Musculoskeletal Research. Dr. Oldroyd and Dr. Kolasinski reported having no relevant conflicts of interest.
AT ACR 2022
PHILADELPHIA – The first consensus screening guidelines for patients with idiopathic inflammatory myopathy (IIM) provide recommendations on risk stratification for individuals, basic and enhanced screening protocols, and screening frequency.
The recommendations, issued by the International Myositis Assessment and Clinical Studies Group (IMACS), stratify cancer risk for individual patients into low, intermediate, or high categories based on the IIM disease subtype, autoantibody status, and clinical features, reported Alexander Oldroyd, PhD, MSc, MBChB of the University of Manchester, England.
“There’s a big unmet need for cancer screening. One in four adults with myositis has cancer, either 3 years before or after a diagnosis of myositis. It’s one of the leading causes of death in these patients, and they’re overwhelmingly diagnosed at a late stage, so we need standardized approaches to get early diagnosis,” he said in an interview at the annual meeting of the American College of Rheumatology.
Sharon Kolasinski, MD, of the University of Pennsylvania in Philadelphia, said in an interview that the guideline is a welcome development for rheumatologists. Dr. Kolasinski moderated the session where Dr. Oldroyd described the guideline, but she was not involved in its formulation.
“I think that we all have wondered for a very long time: What is the optimal cancer screening for myositis patients? We all worry that the onset of their diseases is associated with a coincident cancer, or that they will develop it soon,” she said.
Dr. Oldroyd emphasized that all patients with myositis have elevated risk for cancer compared with the general population and that the guideline categories of low, intermediate, and high are relative only to patients with IIM.
International consensus
The data on which the recommendations are based come from a systematic review and meta-analysis by Dr. Oldroyd and colleagues of 69 studies on cancer risk factors and 9 on IIM-specific cancer screening.
The authors of that paper found that the dermatomyositis subtype, older age, male sex, dysphagia, cutaneous ulceration and antitranscriptional intermediary factor-1 gamma (anti-TIF1-gamma) positivity were associated with significantly increased risk of cancer.
In contrast, polymyositis and clinically amyopathic dermatomyositis subtypes, Raynaud’s phenomenon, interstitial lung disease, very high serum creatine kinase or lactate dehydrogenase levels, and positivity for anti-Jo1 or anti-EJ antibodies were associated with significantly reduced risk of cancer.
The consensus recommendations were developed with anonymous contributions from 75 expert participants in 22 countries, with additional input from 3 patient partners.
Do this
The guideline lists 18 recommendations, of which 13 are strong and 5 are conditional.
An example of a strong recommendation is number 3, based on a moderate level of evidences:
“All adult IIM patients, irrespective of cancer risk, should continue to participate in country/region-specific age and sex appropriate cancer screening programs,” the guideline recommends.
Patients with verified inclusion body myositis or juvenile-onset IIM do not, however, require routine screening for myositis-associated cancer, the guideline says (recommendations 1 and 2).
There are also recommendations that all adults with new-onset IIM be tested for myositis-specific and myositis-associated autoantibodies to assist in stratifying patients by risk category.
The guideline divides screening recommendations into basic and enhanced. The basic screening should include a comprehensive history and physical exam, complete blood count, liver functions tests, erythrocyte sedimentation rates/plasma viscosity, serum protein electrophoresis, urinalysis, and chest x-ray.
Adults with IIM who are determined to be at low risk for IIM-related cancer should have basic cancer screening at the time of IIM diagnosis. Adults with intermediate risk should undergo both basic and enhanced screening at the time of IIM diagnosis, and those with high risk should undergo enhanced screening at the time of myositis diagnosis, with basic screening annually for 3 years, the recommendations say.
Consider doing this
Conditional recommendations (“clinicians should consider ...”) include the use of PET/CT for adults at high risk for cancer when an underlying cancer has not been detected at the time of IIM diagnosis. They also include a single screening test for anti-TIF1-gamma positive dermatomyositis patients whose disease onset was after age 40 and who have at least one additional risk factor.
Also conditionally recommended are upper and lower gastrointestinal endoscopy for patients at high risk when an underlying cancer is not found at the time of IIM diagnosis, nasoendoscopy in geographical regions with elevated risk for nasopharyngeal cancers, and screening for all IIM patients with red-flag symptoms or clinical features of cancer, including unexplained weight loss, family history of cancer, smoking, unexplained fever, or night sweats.
Guided steps
“I think clinicians have a lot of questions such as, ‘well, what should I do, when should I do it?’ These are important clinical questions, and we need guidance about this. We need to balance comprehensiveness with cost-effectiveness, and we need expert opinion about what steps we should take now and which should we take later,” Dr. Kolasinski said.
The guideline development process was supported by the University of Manchester, IMACS, National Institute for Health Research (United Kingdom), National Institutes of Health, National Health Service Northern Care Alliance, The Myositis Association, Myositis UK, University of Pittsburgh, Versus Arthritis, and the Center for Musculoskeletal Research. Dr. Oldroyd and Dr. Kolasinski reported having no relevant conflicts of interest.
AT ACR 2022
PHILADELPHIA – The first consensus screening guidelines for patients with idiopathic inflammatory myopathy (IIM) provide recommendations on risk stratification for individuals, basic and enhanced screening protocols, and screening frequency.
The recommendations, issued by the International Myositis Assessment and Clinical Studies Group (IMACS), stratify cancer risk for individual patients into low, intermediate, or high categories based on the IIM disease subtype, autoantibody status, and clinical features, reported Alexander Oldroyd, PhD, MSc, MBChB of the University of Manchester, England.
“There’s a big unmet need for cancer screening. One in four adults with myositis has cancer, either 3 years before or after a diagnosis of myositis. It’s one of the leading causes of death in these patients, and they’re overwhelmingly diagnosed at a late stage, so we need standardized approaches to get early diagnosis,” he said in an interview at the annual meeting of the American College of Rheumatology.
Sharon Kolasinski, MD, of the University of Pennsylvania in Philadelphia, said in an interview that the guideline is a welcome development for rheumatologists. Dr. Kolasinski moderated the session where Dr. Oldroyd described the guideline, but she was not involved in its formulation.
“I think that we all have wondered for a very long time: What is the optimal cancer screening for myositis patients? We all worry that the onset of their diseases is associated with a coincident cancer, or that they will develop it soon,” she said.
Dr. Oldroyd emphasized that all patients with myositis have elevated risk for cancer compared with the general population and that the guideline categories of low, intermediate, and high are relative only to patients with IIM.
International consensus
The data on which the recommendations are based come from a systematic review and meta-analysis by Dr. Oldroyd and colleagues of 69 studies on cancer risk factors and 9 on IIM-specific cancer screening.
The authors of that paper found that the dermatomyositis subtype, older age, male sex, dysphagia, cutaneous ulceration and antitranscriptional intermediary factor-1 gamma (anti-TIF1-gamma) positivity were associated with significantly increased risk of cancer.
In contrast, polymyositis and clinically amyopathic dermatomyositis subtypes, Raynaud’s phenomenon, interstitial lung disease, very high serum creatine kinase or lactate dehydrogenase levels, and positivity for anti-Jo1 or anti-EJ antibodies were associated with significantly reduced risk of cancer.
The consensus recommendations were developed with anonymous contributions from 75 expert participants in 22 countries, with additional input from 3 patient partners.
Do this
The guideline lists 18 recommendations, of which 13 are strong and 5 are conditional.
An example of a strong recommendation is number 3, based on a moderate level of evidences:
“All adult IIM patients, irrespective of cancer risk, should continue to participate in country/region-specific age and sex appropriate cancer screening programs,” the guideline recommends.
Patients with verified inclusion body myositis or juvenile-onset IIM do not, however, require routine screening for myositis-associated cancer, the guideline says (recommendations 1 and 2).
There are also recommendations that all adults with new-onset IIM be tested for myositis-specific and myositis-associated autoantibodies to assist in stratifying patients by risk category.
The guideline divides screening recommendations into basic and enhanced. The basic screening should include a comprehensive history and physical exam, complete blood count, liver functions tests, erythrocyte sedimentation rates/plasma viscosity, serum protein electrophoresis, urinalysis, and chest x-ray.
Adults with IIM who are determined to be at low risk for IIM-related cancer should have basic cancer screening at the time of IIM diagnosis. Adults with intermediate risk should undergo both basic and enhanced screening at the time of IIM diagnosis, and those with high risk should undergo enhanced screening at the time of myositis diagnosis, with basic screening annually for 3 years, the recommendations say.
Consider doing this
Conditional recommendations (“clinicians should consider ...”) include the use of PET/CT for adults at high risk for cancer when an underlying cancer has not been detected at the time of IIM diagnosis. They also include a single screening test for anti-TIF1-gamma positive dermatomyositis patients whose disease onset was after age 40 and who have at least one additional risk factor.
Also conditionally recommended are upper and lower gastrointestinal endoscopy for patients at high risk when an underlying cancer is not found at the time of IIM diagnosis, nasoendoscopy in geographical regions with elevated risk for nasopharyngeal cancers, and screening for all IIM patients with red-flag symptoms or clinical features of cancer, including unexplained weight loss, family history of cancer, smoking, unexplained fever, or night sweats.
Guided steps
“I think clinicians have a lot of questions such as, ‘well, what should I do, when should I do it?’ These are important clinical questions, and we need guidance about this. We need to balance comprehensiveness with cost-effectiveness, and we need expert opinion about what steps we should take now and which should we take later,” Dr. Kolasinski said.
The guideline development process was supported by the University of Manchester, IMACS, National Institute for Health Research (United Kingdom), National Institutes of Health, National Health Service Northern Care Alliance, The Myositis Association, Myositis UK, University of Pittsburgh, Versus Arthritis, and the Center for Musculoskeletal Research. Dr. Oldroyd and Dr. Kolasinski reported having no relevant conflicts of interest.
Serum dupilumab levels do not predict clinical response
The finding that serum levels, according to a study published in JAMA Dermatology.
The study results mean that researchers should continue exploring potential AD drugs with novel mechanisms to help patients who fail type 2 inflammatory inhibition, experts told this news organization. The search for accurate augurs of clinical performance also must continue.
Addressing inadequate response
Quantifying nonresponse and incomplete response levels with dupilumab is difficult, said Jonathan I. Silverberg, MD, PhD, MPH, offering perspective on the study. “True nonresponse is probably less than 20%, but many other patients are inadequate responders even if they are having partial response.” Dr. Silverberg, professor of dermatology and director of clinical research, at George Washington University, Washington, was not an investigator.
Robert Sidbury, MD, MPH, added, “When a patient doesn’t respond to a medication that you expect they should, we always ask ourselves why.” Dermatologists have long assumed that, as with biologics for psoriasis, low blood levels were to blame for dupilumab nonresponse, said Dr. Sidbury, who is division chief of dermatology at Seattle Children’s Hospital and was not involved with the study. “This study showed that there was no correlation between response and blood levels.”
In the study, Lotte S. Spekhorst, MD, of National Expertise Center for Atopic Dermatitis, department of dermatology and allergology, University Medical Center Utrecht (the Netherlands) and coinvestigators prospectively followed 295 consecutive adult patients with moderate AD who were treated with dupilumab for 1 year. All patients received the same loading (600 mg) and biweekly (300 mg) doses.
The median dupilumab level at 16 weeks was 86.6 mcg/mL, which is higher than serum levels observed with other monoclonal antibodies used for other indications, such as psoriasis and inflammatory bowel disease, the authors noted. More importantly, researchers found no significant relationship between median week 16 dupilumab levels and 1-year clinical responses measured either discretely (Eczema Area and Severity Index [EASI] < 50, 50, 75, or 90; P = .18) or as quartiles (P = .06).
“It may be that response is dependent on target availability of the IL-4R-alpha, with an interpatient variability producing heterogeneity in response,” the authors wrote. But because serum dupilumab levels were relatively high, they said, all patients’ IL-4R-alpha “was likely fully saturated” at 16 weeks.
“This would explain why serum dupilumab levels were not related to effectiveness,” they noted, “although we cannot rule out differential effects in the tissue associated with heterogeneity in serum dupilumab levels.”
The study helps explain why some patients do not fully respond to dupilumab, said Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, who was not involved with the study.
“One hypothesis would be that drug serum levels differ due to metabolism or absorption reasons,” Dr. Simpson said in an interview. Results also suggest that heterogeneity in disease biology, such as other uninhibited cytokine pathways, might explain differences in clinical results. “Thus, more therapeutics that target different inflammatory pathways are needed to capture responses in patients not adequately responding to type 2 inflammatory blockade,” he said.
Assessing AEs
As with response levels, serum dupilumab levels at week 16 did not predict AEs including dupilumab-associated ocular surface disease (DAOSD), which impacted 46.4% of 216 patients who reported AEs. These findings also contradict what happens with biologics in other diseases such as psoriasis and inflammatory bowel disease, said Dr. Sidbury, wherein serum drug levels may predict both clinical response and side-effect risks.
A previous study showed that lowering dupilumab levels led to improvement in DAOSD. Authors of the current study therefore surmised that DAOSD development might be more associated with interpatient variability in IL-4R-alpha expression than with serum drug levels. “More research is necessary to confirm the hypothesis of interpatient variability of the IL-4Ra and the pharmacokinetics of dupilumab,” they concluded.
For now, said Dr. Sidbury, the study helps clinicians look beyond serum drug levels when patients respond inadequately to dupilumab. Moreover, added Dr. Silverberg, study results mean that physicians must find other ways to predict dupilumab response levels. “We need better predictors of clinical response – theranostic markers that we could test the patient to and understand how well they’re going to do,” he said.
Be it dupilumab or any other medication, he said, physicians lack even confirmatory biomarkers to reflect when a drug is working well. “Right now, we go with clinical assessments. But if it’s not drug levels, we have to figure out why some patients do markedly better than others.”
It was not unreasonable, Dr. Silverberg said, for the investigators to seek a biomarker in blood rather than tissue. “But in this disease, we believe that the more important place to look for biomarkers and drug levels would be in the skin itself. So we are still left with the issue” that drug levels in tissue might reflect response when serum levels do not.
The study was supported by grants from AbbVie, Eli Lilly, Leo Pharma, Pfizer, and Sanofi. Study patients participated in the BioDay Registry, which is sponsored by Sanofi, Regeneron, AbbVie, Eli Lilly, LEO Pharma, and Pfizer; the sponsors had no role in the study design and conduct. Dr. Spekhorst discloses receiving speaking fees from Abbvie outside the work; disclosures of other authors included receiving advisory, speaking consulting, and/or investigator fees from Sanofi Genzyme during the study. Several authors had no disclosures.
Dr. Simpson has been an investigator and consultant for Regeneron and Sanofi, makers of dupilumab. Dr. Silverberg has been an investigator, consultant, and speaker for Regeneron and Sanofi. Dr. Sidbury has been a clinical investigator for all dupilumab pediatric trials. (His institution has a contract with Regeneron and Sanofi, but he receives no money from the arrangement.)
The finding that serum levels, according to a study published in JAMA Dermatology.
The study results mean that researchers should continue exploring potential AD drugs with novel mechanisms to help patients who fail type 2 inflammatory inhibition, experts told this news organization. The search for accurate augurs of clinical performance also must continue.
Addressing inadequate response
Quantifying nonresponse and incomplete response levels with dupilumab is difficult, said Jonathan I. Silverberg, MD, PhD, MPH, offering perspective on the study. “True nonresponse is probably less than 20%, but many other patients are inadequate responders even if they are having partial response.” Dr. Silverberg, professor of dermatology and director of clinical research, at George Washington University, Washington, was not an investigator.
Robert Sidbury, MD, MPH, added, “When a patient doesn’t respond to a medication that you expect they should, we always ask ourselves why.” Dermatologists have long assumed that, as with biologics for psoriasis, low blood levels were to blame for dupilumab nonresponse, said Dr. Sidbury, who is division chief of dermatology at Seattle Children’s Hospital and was not involved with the study. “This study showed that there was no correlation between response and blood levels.”
In the study, Lotte S. Spekhorst, MD, of National Expertise Center for Atopic Dermatitis, department of dermatology and allergology, University Medical Center Utrecht (the Netherlands) and coinvestigators prospectively followed 295 consecutive adult patients with moderate AD who were treated with dupilumab for 1 year. All patients received the same loading (600 mg) and biweekly (300 mg) doses.
The median dupilumab level at 16 weeks was 86.6 mcg/mL, which is higher than serum levels observed with other monoclonal antibodies used for other indications, such as psoriasis and inflammatory bowel disease, the authors noted. More importantly, researchers found no significant relationship between median week 16 dupilumab levels and 1-year clinical responses measured either discretely (Eczema Area and Severity Index [EASI] < 50, 50, 75, or 90; P = .18) or as quartiles (P = .06).
“It may be that response is dependent on target availability of the IL-4R-alpha, with an interpatient variability producing heterogeneity in response,” the authors wrote. But because serum dupilumab levels were relatively high, they said, all patients’ IL-4R-alpha “was likely fully saturated” at 16 weeks.
“This would explain why serum dupilumab levels were not related to effectiveness,” they noted, “although we cannot rule out differential effects in the tissue associated with heterogeneity in serum dupilumab levels.”
The study helps explain why some patients do not fully respond to dupilumab, said Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, who was not involved with the study.
“One hypothesis would be that drug serum levels differ due to metabolism or absorption reasons,” Dr. Simpson said in an interview. Results also suggest that heterogeneity in disease biology, such as other uninhibited cytokine pathways, might explain differences in clinical results. “Thus, more therapeutics that target different inflammatory pathways are needed to capture responses in patients not adequately responding to type 2 inflammatory blockade,” he said.
Assessing AEs
As with response levels, serum dupilumab levels at week 16 did not predict AEs including dupilumab-associated ocular surface disease (DAOSD), which impacted 46.4% of 216 patients who reported AEs. These findings also contradict what happens with biologics in other diseases such as psoriasis and inflammatory bowel disease, said Dr. Sidbury, wherein serum drug levels may predict both clinical response and side-effect risks.
A previous study showed that lowering dupilumab levels led to improvement in DAOSD. Authors of the current study therefore surmised that DAOSD development might be more associated with interpatient variability in IL-4R-alpha expression than with serum drug levels. “More research is necessary to confirm the hypothesis of interpatient variability of the IL-4Ra and the pharmacokinetics of dupilumab,” they concluded.
For now, said Dr. Sidbury, the study helps clinicians look beyond serum drug levels when patients respond inadequately to dupilumab. Moreover, added Dr. Silverberg, study results mean that physicians must find other ways to predict dupilumab response levels. “We need better predictors of clinical response – theranostic markers that we could test the patient to and understand how well they’re going to do,” he said.
Be it dupilumab or any other medication, he said, physicians lack even confirmatory biomarkers to reflect when a drug is working well. “Right now, we go with clinical assessments. But if it’s not drug levels, we have to figure out why some patients do markedly better than others.”
It was not unreasonable, Dr. Silverberg said, for the investigators to seek a biomarker in blood rather than tissue. “But in this disease, we believe that the more important place to look for biomarkers and drug levels would be in the skin itself. So we are still left with the issue” that drug levels in tissue might reflect response when serum levels do not.
The study was supported by grants from AbbVie, Eli Lilly, Leo Pharma, Pfizer, and Sanofi. Study patients participated in the BioDay Registry, which is sponsored by Sanofi, Regeneron, AbbVie, Eli Lilly, LEO Pharma, and Pfizer; the sponsors had no role in the study design and conduct. Dr. Spekhorst discloses receiving speaking fees from Abbvie outside the work; disclosures of other authors included receiving advisory, speaking consulting, and/or investigator fees from Sanofi Genzyme during the study. Several authors had no disclosures.
Dr. Simpson has been an investigator and consultant for Regeneron and Sanofi, makers of dupilumab. Dr. Silverberg has been an investigator, consultant, and speaker for Regeneron and Sanofi. Dr. Sidbury has been a clinical investigator for all dupilumab pediatric trials. (His institution has a contract with Regeneron and Sanofi, but he receives no money from the arrangement.)
The finding that serum levels, according to a study published in JAMA Dermatology.
The study results mean that researchers should continue exploring potential AD drugs with novel mechanisms to help patients who fail type 2 inflammatory inhibition, experts told this news organization. The search for accurate augurs of clinical performance also must continue.
Addressing inadequate response
Quantifying nonresponse and incomplete response levels with dupilumab is difficult, said Jonathan I. Silverberg, MD, PhD, MPH, offering perspective on the study. “True nonresponse is probably less than 20%, but many other patients are inadequate responders even if they are having partial response.” Dr. Silverberg, professor of dermatology and director of clinical research, at George Washington University, Washington, was not an investigator.
Robert Sidbury, MD, MPH, added, “When a patient doesn’t respond to a medication that you expect they should, we always ask ourselves why.” Dermatologists have long assumed that, as with biologics for psoriasis, low blood levels were to blame for dupilumab nonresponse, said Dr. Sidbury, who is division chief of dermatology at Seattle Children’s Hospital and was not involved with the study. “This study showed that there was no correlation between response and blood levels.”
In the study, Lotte S. Spekhorst, MD, of National Expertise Center for Atopic Dermatitis, department of dermatology and allergology, University Medical Center Utrecht (the Netherlands) and coinvestigators prospectively followed 295 consecutive adult patients with moderate AD who were treated with dupilumab for 1 year. All patients received the same loading (600 mg) and biweekly (300 mg) doses.
The median dupilumab level at 16 weeks was 86.6 mcg/mL, which is higher than serum levels observed with other monoclonal antibodies used for other indications, such as psoriasis and inflammatory bowel disease, the authors noted. More importantly, researchers found no significant relationship between median week 16 dupilumab levels and 1-year clinical responses measured either discretely (Eczema Area and Severity Index [EASI] < 50, 50, 75, or 90; P = .18) or as quartiles (P = .06).
“It may be that response is dependent on target availability of the IL-4R-alpha, with an interpatient variability producing heterogeneity in response,” the authors wrote. But because serum dupilumab levels were relatively high, they said, all patients’ IL-4R-alpha “was likely fully saturated” at 16 weeks.
“This would explain why serum dupilumab levels were not related to effectiveness,” they noted, “although we cannot rule out differential effects in the tissue associated with heterogeneity in serum dupilumab levels.”
The study helps explain why some patients do not fully respond to dupilumab, said Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, who was not involved with the study.
“One hypothesis would be that drug serum levels differ due to metabolism or absorption reasons,” Dr. Simpson said in an interview. Results also suggest that heterogeneity in disease biology, such as other uninhibited cytokine pathways, might explain differences in clinical results. “Thus, more therapeutics that target different inflammatory pathways are needed to capture responses in patients not adequately responding to type 2 inflammatory blockade,” he said.
Assessing AEs
As with response levels, serum dupilumab levels at week 16 did not predict AEs including dupilumab-associated ocular surface disease (DAOSD), which impacted 46.4% of 216 patients who reported AEs. These findings also contradict what happens with biologics in other diseases such as psoriasis and inflammatory bowel disease, said Dr. Sidbury, wherein serum drug levels may predict both clinical response and side-effect risks.
A previous study showed that lowering dupilumab levels led to improvement in DAOSD. Authors of the current study therefore surmised that DAOSD development might be more associated with interpatient variability in IL-4R-alpha expression than with serum drug levels. “More research is necessary to confirm the hypothesis of interpatient variability of the IL-4Ra and the pharmacokinetics of dupilumab,” they concluded.
For now, said Dr. Sidbury, the study helps clinicians look beyond serum drug levels when patients respond inadequately to dupilumab. Moreover, added Dr. Silverberg, study results mean that physicians must find other ways to predict dupilumab response levels. “We need better predictors of clinical response – theranostic markers that we could test the patient to and understand how well they’re going to do,” he said.
Be it dupilumab or any other medication, he said, physicians lack even confirmatory biomarkers to reflect when a drug is working well. “Right now, we go with clinical assessments. But if it’s not drug levels, we have to figure out why some patients do markedly better than others.”
It was not unreasonable, Dr. Silverberg said, for the investigators to seek a biomarker in blood rather than tissue. “But in this disease, we believe that the more important place to look for biomarkers and drug levels would be in the skin itself. So we are still left with the issue” that drug levels in tissue might reflect response when serum levels do not.
The study was supported by grants from AbbVie, Eli Lilly, Leo Pharma, Pfizer, and Sanofi. Study patients participated in the BioDay Registry, which is sponsored by Sanofi, Regeneron, AbbVie, Eli Lilly, LEO Pharma, and Pfizer; the sponsors had no role in the study design and conduct. Dr. Spekhorst discloses receiving speaking fees from Abbvie outside the work; disclosures of other authors included receiving advisory, speaking consulting, and/or investigator fees from Sanofi Genzyme during the study. Several authors had no disclosures.
Dr. Simpson has been an investigator and consultant for Regeneron and Sanofi, makers of dupilumab. Dr. Silverberg has been an investigator, consultant, and speaker for Regeneron and Sanofi. Dr. Sidbury has been a clinical investigator for all dupilumab pediatric trials. (His institution has a contract with Regeneron and Sanofi, but he receives no money from the arrangement.)
FROM JAMA DERMATOLOGY