User login
Twincretin ‘impressive’: Topline data from phase 3 trial in diabetes
Tirzepatide, a novel subcutaneously injected drug that acts via two related but separate pathways of glucose control, produced strikingly positive effects in top-line results from the phase 3, placebo-controlled study SURPASS-1 in 478 adults with type 2 diabetes, according to a Dec. 9 press release from the manufacturer, Lilly.
The tirzepatide molecule exerts agonist effects at both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor, and has been called a “twincretin” for its activity encompassing two different incretins. Phase 2 trial results caused excitement, with one physician calling the data “unbelievable” when reported in 2018.
SURPASS-1 enrolled patients who were very early in the course of their disease, had on average relatively mild elevation in glucose levels, and few metabolic comorbidities. They took one of three doses of the agent (5, 10, or 15 mg) as monotherapy or placebo for 40 weeks.
Julio Rosenstock, MD, said in the Lilly statement: “The study took a bold approach in assessing A1c targets. Not only did nearly 90% of all participants taking tirzepatide meet the standard A1c goal of less than 7%, more than half taking the highest dose also achieved an A1c less than 5.7%, the level seen in people without diabetes.”
Dr. Rosenstock is principal investigator of SURPASS-1 and director of the Dallas Diabetes Research Center in Texas.
The discontinuation rate in the high-dose group was 21.5% compared with less than 10% in the two lower-dose cohorts. Lilly said most of the dropouts “were due to the pandemic and family or work reasons.” The dropout rate in the placebo group was 14.8%.
These data were not included in the efficacy analysis, however, which “muddied” the analysis somewhat, one pharma analyst told BioPharma Dive.
Commenting on the new trial data, Ildiko Lingvay, MD, said in an interview: “I am very impressed with these results,” which are “unprecedented for any glucose-lowering medication that has ever been tested.”
Dr. Lingvay, of the department of internal medicine/endocrinology, and medical director, office of clinical trials management at UT Southwestern Medical Center, Dallas, was not involved in the study.
She added that the weight loss seen with tirzepatide “is equally impressive with greater than 10% of body weight loss above placebo achieved within 40 weeks of treatment and without any directed weight loss efforts.”
If the agent is eventually approved, “I am enthusiastic about the prospect of having another very powerful tool to address both diabetes and obesity,” she added.
The full results of SURPASS-1 will be presented at the American Diabetes Association 81st Scientific Sessions and published in a peer-reviewed journal in 2021.
SURPASS-1 is one of eight phase 3 studies of the drug, including five registration studies and one large 12,500-patient cardiovascular outcomes trial.
Tirzepatide patients lost up to 20 lb, side effect profile ‘reassuring’
In the study, patients had been recently diagnosed with type 2 diabetes (average duration, 4.8 years) and 54% were treatment-naive. Average baseline hemoglobin A1c was 7.9% and mean weight was 85.9 kg (189 pounds).
Patients started on a subcutaneous injectable dose of tirzepatide of 2.5 mg per week, which was titrated up to the final dose – 5, 10, or 15 mg – in 2.5-mg increments given as monotherapy for 40 weeks and compared with placebo.
Treatment with tirzepatide resulted in average reductions in A1c from baseline that ranged from 1.87% to 2.07%, depending on the dose, and were all significant compared with an increase of 0.4% with placebo.
The percentage of patients whose A1c fell to normal levels (less than 5.7%) ranged from 30.5% to 51.7%, compared with 0.9% among controls, and again, was significant for all doses.
Patients treated with tirzepatide also lost weight. Average weight reductions after 40 weeks were significant and ranged from 7.0 to 9.5 kg (15-21 pounds) compared with an average loss of 0.7 kg (1.5 pounds) among patients who received placebo.
The most common adverse events were gastrointestinal-related and mild to moderate in severity, and usually occurred during dose escalation.
Dr. Lingvay said the safety data reported are “reassuring, with side effects in the anticipated range and comparable with other medications in the GLP-1 agonist class.”
And no hypoglycemic (level 2, < 54 mg/dL) events were reported, “which is impressive considering the overall glucose level achieved,” she noted.
“I am eagerly awaiting the results of the other studies within the SURPASS program and hope those will confirm these initial findings and provide additional safety and efficacy information in a wider range of patients with type 2 diabetes,” she concluded.
Dr. Lingvay has reported receiving research funding, advisory/consulting fees, and/or other support from Novo Nordisk, Eli Lilly, Sanofi, AstraZeneca, Boehringer Ingelheim, Janssen, Intercept, Intarcia, Target Pharma, Merck, Pfizer, Novartis, GI Dynamics, Mylan, MannKind, Valeritas, Bayer, and Zealand Pharma.
A version of this article originally appeared on Medscape.com.
Tirzepatide, a novel subcutaneously injected drug that acts via two related but separate pathways of glucose control, produced strikingly positive effects in top-line results from the phase 3, placebo-controlled study SURPASS-1 in 478 adults with type 2 diabetes, according to a Dec. 9 press release from the manufacturer, Lilly.
The tirzepatide molecule exerts agonist effects at both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor, and has been called a “twincretin” for its activity encompassing two different incretins. Phase 2 trial results caused excitement, with one physician calling the data “unbelievable” when reported in 2018.
SURPASS-1 enrolled patients who were very early in the course of their disease, had on average relatively mild elevation in glucose levels, and few metabolic comorbidities. They took one of three doses of the agent (5, 10, or 15 mg) as monotherapy or placebo for 40 weeks.
Julio Rosenstock, MD, said in the Lilly statement: “The study took a bold approach in assessing A1c targets. Not only did nearly 90% of all participants taking tirzepatide meet the standard A1c goal of less than 7%, more than half taking the highest dose also achieved an A1c less than 5.7%, the level seen in people without diabetes.”
Dr. Rosenstock is principal investigator of SURPASS-1 and director of the Dallas Diabetes Research Center in Texas.
The discontinuation rate in the high-dose group was 21.5% compared with less than 10% in the two lower-dose cohorts. Lilly said most of the dropouts “were due to the pandemic and family or work reasons.” The dropout rate in the placebo group was 14.8%.
These data were not included in the efficacy analysis, however, which “muddied” the analysis somewhat, one pharma analyst told BioPharma Dive.
Commenting on the new trial data, Ildiko Lingvay, MD, said in an interview: “I am very impressed with these results,” which are “unprecedented for any glucose-lowering medication that has ever been tested.”
Dr. Lingvay, of the department of internal medicine/endocrinology, and medical director, office of clinical trials management at UT Southwestern Medical Center, Dallas, was not involved in the study.
She added that the weight loss seen with tirzepatide “is equally impressive with greater than 10% of body weight loss above placebo achieved within 40 weeks of treatment and without any directed weight loss efforts.”
If the agent is eventually approved, “I am enthusiastic about the prospect of having another very powerful tool to address both diabetes and obesity,” she added.
The full results of SURPASS-1 will be presented at the American Diabetes Association 81st Scientific Sessions and published in a peer-reviewed journal in 2021.
SURPASS-1 is one of eight phase 3 studies of the drug, including five registration studies and one large 12,500-patient cardiovascular outcomes trial.
Tirzepatide patients lost up to 20 lb, side effect profile ‘reassuring’
In the study, patients had been recently diagnosed with type 2 diabetes (average duration, 4.8 years) and 54% were treatment-naive. Average baseline hemoglobin A1c was 7.9% and mean weight was 85.9 kg (189 pounds).
Patients started on a subcutaneous injectable dose of tirzepatide of 2.5 mg per week, which was titrated up to the final dose – 5, 10, or 15 mg – in 2.5-mg increments given as monotherapy for 40 weeks and compared with placebo.
Treatment with tirzepatide resulted in average reductions in A1c from baseline that ranged from 1.87% to 2.07%, depending on the dose, and were all significant compared with an increase of 0.4% with placebo.
The percentage of patients whose A1c fell to normal levels (less than 5.7%) ranged from 30.5% to 51.7%, compared with 0.9% among controls, and again, was significant for all doses.
Patients treated with tirzepatide also lost weight. Average weight reductions after 40 weeks were significant and ranged from 7.0 to 9.5 kg (15-21 pounds) compared with an average loss of 0.7 kg (1.5 pounds) among patients who received placebo.
The most common adverse events were gastrointestinal-related and mild to moderate in severity, and usually occurred during dose escalation.
Dr. Lingvay said the safety data reported are “reassuring, with side effects in the anticipated range and comparable with other medications in the GLP-1 agonist class.”
And no hypoglycemic (level 2, < 54 mg/dL) events were reported, “which is impressive considering the overall glucose level achieved,” she noted.
“I am eagerly awaiting the results of the other studies within the SURPASS program and hope those will confirm these initial findings and provide additional safety and efficacy information in a wider range of patients with type 2 diabetes,” she concluded.
Dr. Lingvay has reported receiving research funding, advisory/consulting fees, and/or other support from Novo Nordisk, Eli Lilly, Sanofi, AstraZeneca, Boehringer Ingelheim, Janssen, Intercept, Intarcia, Target Pharma, Merck, Pfizer, Novartis, GI Dynamics, Mylan, MannKind, Valeritas, Bayer, and Zealand Pharma.
A version of this article originally appeared on Medscape.com.
Tirzepatide, a novel subcutaneously injected drug that acts via two related but separate pathways of glucose control, produced strikingly positive effects in top-line results from the phase 3, placebo-controlled study SURPASS-1 in 478 adults with type 2 diabetes, according to a Dec. 9 press release from the manufacturer, Lilly.
The tirzepatide molecule exerts agonist effects at both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor, and has been called a “twincretin” for its activity encompassing two different incretins. Phase 2 trial results caused excitement, with one physician calling the data “unbelievable” when reported in 2018.
SURPASS-1 enrolled patients who were very early in the course of their disease, had on average relatively mild elevation in glucose levels, and few metabolic comorbidities. They took one of three doses of the agent (5, 10, or 15 mg) as monotherapy or placebo for 40 weeks.
Julio Rosenstock, MD, said in the Lilly statement: “The study took a bold approach in assessing A1c targets. Not only did nearly 90% of all participants taking tirzepatide meet the standard A1c goal of less than 7%, more than half taking the highest dose also achieved an A1c less than 5.7%, the level seen in people without diabetes.”
Dr. Rosenstock is principal investigator of SURPASS-1 and director of the Dallas Diabetes Research Center in Texas.
The discontinuation rate in the high-dose group was 21.5% compared with less than 10% in the two lower-dose cohorts. Lilly said most of the dropouts “were due to the pandemic and family or work reasons.” The dropout rate in the placebo group was 14.8%.
These data were not included in the efficacy analysis, however, which “muddied” the analysis somewhat, one pharma analyst told BioPharma Dive.
Commenting on the new trial data, Ildiko Lingvay, MD, said in an interview: “I am very impressed with these results,” which are “unprecedented for any glucose-lowering medication that has ever been tested.”
Dr. Lingvay, of the department of internal medicine/endocrinology, and medical director, office of clinical trials management at UT Southwestern Medical Center, Dallas, was not involved in the study.
She added that the weight loss seen with tirzepatide “is equally impressive with greater than 10% of body weight loss above placebo achieved within 40 weeks of treatment and without any directed weight loss efforts.”
If the agent is eventually approved, “I am enthusiastic about the prospect of having another very powerful tool to address both diabetes and obesity,” she added.
The full results of SURPASS-1 will be presented at the American Diabetes Association 81st Scientific Sessions and published in a peer-reviewed journal in 2021.
SURPASS-1 is one of eight phase 3 studies of the drug, including five registration studies and one large 12,500-patient cardiovascular outcomes trial.
Tirzepatide patients lost up to 20 lb, side effect profile ‘reassuring’
In the study, patients had been recently diagnosed with type 2 diabetes (average duration, 4.8 years) and 54% were treatment-naive. Average baseline hemoglobin A1c was 7.9% and mean weight was 85.9 kg (189 pounds).
Patients started on a subcutaneous injectable dose of tirzepatide of 2.5 mg per week, which was titrated up to the final dose – 5, 10, or 15 mg – in 2.5-mg increments given as monotherapy for 40 weeks and compared with placebo.
Treatment with tirzepatide resulted in average reductions in A1c from baseline that ranged from 1.87% to 2.07%, depending on the dose, and were all significant compared with an increase of 0.4% with placebo.
The percentage of patients whose A1c fell to normal levels (less than 5.7%) ranged from 30.5% to 51.7%, compared with 0.9% among controls, and again, was significant for all doses.
Patients treated with tirzepatide also lost weight. Average weight reductions after 40 weeks were significant and ranged from 7.0 to 9.5 kg (15-21 pounds) compared with an average loss of 0.7 kg (1.5 pounds) among patients who received placebo.
The most common adverse events were gastrointestinal-related and mild to moderate in severity, and usually occurred during dose escalation.
Dr. Lingvay said the safety data reported are “reassuring, with side effects in the anticipated range and comparable with other medications in the GLP-1 agonist class.”
And no hypoglycemic (level 2, < 54 mg/dL) events were reported, “which is impressive considering the overall glucose level achieved,” she noted.
“I am eagerly awaiting the results of the other studies within the SURPASS program and hope those will confirm these initial findings and provide additional safety and efficacy information in a wider range of patients with type 2 diabetes,” she concluded.
Dr. Lingvay has reported receiving research funding, advisory/consulting fees, and/or other support from Novo Nordisk, Eli Lilly, Sanofi, AstraZeneca, Boehringer Ingelheim, Janssen, Intercept, Intarcia, Target Pharma, Merck, Pfizer, Novartis, GI Dynamics, Mylan, MannKind, Valeritas, Bayer, and Zealand Pharma.
A version of this article originally appeared on Medscape.com.
Type 2 Diabetes 2021
This supplement to Clinician Reviews brings together key updates in the field of T2D to help care for patients who have not only T2D, but also other interconnected diseases.
Supplementary Materials:
Chapter 1: Evolution of Type 2 Diabetes Treatment
Plain Language Patient Summary:

Infographic:
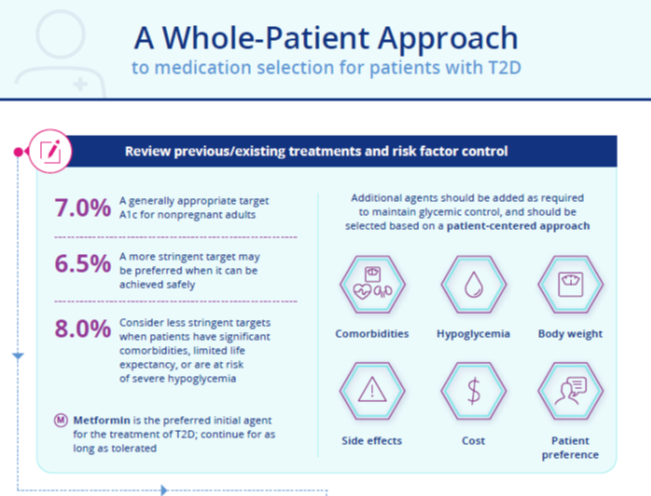
Chapter 2: A Practical Approach to Managing Kidney Disease in Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 3: Heart Failure in Patients with Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

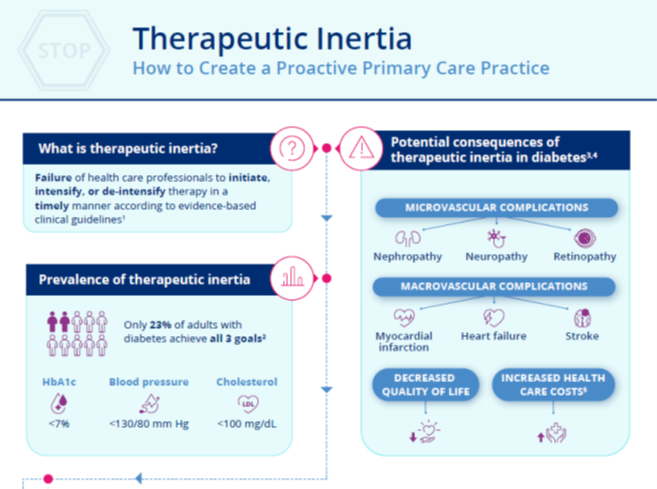
Chapter 4: Overcoming Therapeutic Inertia: Practical Approaches
Plain Language Patient Summary:

Infographic:
Supplemental Materials are joint copyright © 2020 Frontline Medical Communications and Boehringer Ingelheim Pharmaceuticals, Inc.
This supplement to Clinician Reviews brings together key updates in the field of T2D to help care for patients who have not only T2D, but also other interconnected diseases.
Supplementary Materials:
Chapter 1: Evolution of Type 2 Diabetes Treatment
Plain Language Patient Summary:

Infographic:

Chapter 2: A Practical Approach to Managing Kidney Disease in Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 3: Heart Failure in Patients with Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 4: Overcoming Therapeutic Inertia: Practical Approaches
Plain Language Patient Summary:

Infographic:

Supplemental Materials are joint copyright © 2020 Frontline Medical Communications and Boehringer Ingelheim Pharmaceuticals, Inc.
This supplement to Clinician Reviews brings together key updates in the field of T2D to help care for patients who have not only T2D, but also other interconnected diseases.
Supplementary Materials:
Chapter 1: Evolution of Type 2 Diabetes Treatment
Plain Language Patient Summary:

Infographic:

Chapter 2: A Practical Approach to Managing Kidney Disease in Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 3: Heart Failure in Patients with Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 4: Overcoming Therapeutic Inertia: Practical Approaches
Plain Language Patient Summary:

Infographic:

Supplemental Materials are joint copyright © 2020 Frontline Medical Communications and Boehringer Ingelheim Pharmaceuticals, Inc.
ACC/AHA update two atrial fibrillation performance measures
The American College of Cardiology and American Heart Association Task Force on Performance Measures have made two changes to performance measures for adults with atrial fibrillation or atrial flutter.
The 2020 Update to the 2016 ACC/AHA Clinical Performance and Quality Measures for Adults With Atrial Fibrillation or Atrial Flutter was published online Dec. 7 in the Journal of the American College of Cardiology and Circulation: Cardiovascular Quality and Outcomes. It was developed in collaboration with the Heart Rhythm Society.
Both performance measure changes were prompted by, and are in accordance with, the 2019 ACC/AHA/Heart Rhythm Society atrial fibrillation guideline focused update issued in January 2019, and reported by this news organization at that time.
The first change is the clarification that valvular atrial fibrillation is atrial fibrillation with either moderate or severe mitral stenosis or a mechanical heart valve. This change is incorporated into all the performance measures.
The second change, which only applies to the performance measure of anticoagulation prescribed, is the separation of a male and female threshold for the CHA2DS2-VASc score.
This threshold is now a score higher than 1 for men and higher than 2 for women, further demonstrating that the risk for stroke differs for men and women with atrial fibrillation or atrial flutter, the ACC/AHA noted in a press release.
“Successful implementation of these updated performance measures by clinicians and healthcare organizations will lead to quality improvement for adult patients with atrial fibrillation or atrial flutter,” they said.
A version of this article originally appeared on Medscape.com.
The American College of Cardiology and American Heart Association Task Force on Performance Measures have made two changes to performance measures for adults with atrial fibrillation or atrial flutter.
The 2020 Update to the 2016 ACC/AHA Clinical Performance and Quality Measures for Adults With Atrial Fibrillation or Atrial Flutter was published online Dec. 7 in the Journal of the American College of Cardiology and Circulation: Cardiovascular Quality and Outcomes. It was developed in collaboration with the Heart Rhythm Society.
Both performance measure changes were prompted by, and are in accordance with, the 2019 ACC/AHA/Heart Rhythm Society atrial fibrillation guideline focused update issued in January 2019, and reported by this news organization at that time.
The first change is the clarification that valvular atrial fibrillation is atrial fibrillation with either moderate or severe mitral stenosis or a mechanical heart valve. This change is incorporated into all the performance measures.
The second change, which only applies to the performance measure of anticoagulation prescribed, is the separation of a male and female threshold for the CHA2DS2-VASc score.
This threshold is now a score higher than 1 for men and higher than 2 for women, further demonstrating that the risk for stroke differs for men and women with atrial fibrillation or atrial flutter, the ACC/AHA noted in a press release.
“Successful implementation of these updated performance measures by clinicians and healthcare organizations will lead to quality improvement for adult patients with atrial fibrillation or atrial flutter,” they said.
A version of this article originally appeared on Medscape.com.
The American College of Cardiology and American Heart Association Task Force on Performance Measures have made two changes to performance measures for adults with atrial fibrillation or atrial flutter.
The 2020 Update to the 2016 ACC/AHA Clinical Performance and Quality Measures for Adults With Atrial Fibrillation or Atrial Flutter was published online Dec. 7 in the Journal of the American College of Cardiology and Circulation: Cardiovascular Quality and Outcomes. It was developed in collaboration with the Heart Rhythm Society.
Both performance measure changes were prompted by, and are in accordance with, the 2019 ACC/AHA/Heart Rhythm Society atrial fibrillation guideline focused update issued in January 2019, and reported by this news organization at that time.
The first change is the clarification that valvular atrial fibrillation is atrial fibrillation with either moderate or severe mitral stenosis or a mechanical heart valve. This change is incorporated into all the performance measures.
The second change, which only applies to the performance measure of anticoagulation prescribed, is the separation of a male and female threshold for the CHA2DS2-VASc score.
This threshold is now a score higher than 1 for men and higher than 2 for women, further demonstrating that the risk for stroke differs for men and women with atrial fibrillation or atrial flutter, the ACC/AHA noted in a press release.
“Successful implementation of these updated performance measures by clinicians and healthcare organizations will lead to quality improvement for adult patients with atrial fibrillation or atrial flutter,” they said.
A version of this article originally appeared on Medscape.com.
Type 2 Diabetes 2021
This supplement to The Journal of Family Practice brings together key updates in the field of T2D to help care for patients who have not only T2D, but also other interconnected diseases.
Supplementary Materials:
Chapter 1: Evolution of Type 2 Diabetes Treatment
Plain Language Patient Summary:

Infographic:

Chapter 2: A Practical Approach to Managing Kidney Disease in Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 3: Heart Failure in Patients with Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 4: Overcoming Therapeutic Inertia: Practical Approaches
Plain Language Patient Summary:

Infographic:

Supplemental Materials are joint copyright © 2020 Frontline Medical Communications and Boehringer Ingelheim Pharmaceuticals, Inc.
This supplement to The Journal of Family Practice brings together key updates in the field of T2D to help care for patients who have not only T2D, but also other interconnected diseases.
Supplementary Materials:
Chapter 1: Evolution of Type 2 Diabetes Treatment
Plain Language Patient Summary:

Infographic:

Chapter 2: A Practical Approach to Managing Kidney Disease in Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 3: Heart Failure in Patients with Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 4: Overcoming Therapeutic Inertia: Practical Approaches
Plain Language Patient Summary:

Infographic:

Supplemental Materials are joint copyright © 2020 Frontline Medical Communications and Boehringer Ingelheim Pharmaceuticals, Inc.
This supplement to The Journal of Family Practice brings together key updates in the field of T2D to help care for patients who have not only T2D, but also other interconnected diseases.
Supplementary Materials:
Chapter 1: Evolution of Type 2 Diabetes Treatment
Plain Language Patient Summary:

Infographic:

Chapter 2: A Practical Approach to Managing Kidney Disease in Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 3: Heart Failure in Patients with Type 2 Diabetes
Plain Language Patient Summary:

Infographic:

Chapter 4: Overcoming Therapeutic Inertia: Practical Approaches
Plain Language Patient Summary:

Infographic:

Supplemental Materials are joint copyright © 2020 Frontline Medical Communications and Boehringer Ingelheim Pharmaceuticals, Inc.
Raising psychiatry up ‘from depths of the asylums’
New biography captures Dr. Anthony Clare’s complexity
In “Psychiatrist in the Chair,” authors Brendan Kelly and Muiris Houston tell the story of a fellow Irishman, Anthony Clare, MD, who brought intelligence and eloquence to psychiatry. They tell a well-measured, well-referenced story of Anthony Clare’s personal and professional life. They capture his eloquence, wit, charm, and success in psychiatry as well as alluding to Dr. Clare’s self-reported “some kind of Irish darkness.”
In 1983, I was a young Scottish psychiatrist entering a fusty profession. Suddenly, there was Dr. Anthony Clare on the BBC! In “In the Psychiatrist’s Chair,” Dr. Clare interviewed celebrities. In addition to describing his Irish darkness, Brendan Kelly, MD, PhD and Muiris Houston, MD, FRCGP, both of whom are affiliated with Trinity College Dublin, note that Dr. Clare said: “I’m better at destroying systems than I am at putting them together – I do rather look for people to interview who will not live up to the prediction; there’s an element of destructiveness that’s still in me.”
I still listen to his talks on YouTube. His delicate probing questioning of B.F. Skinner, PhD, is one of my favorites, as he expertly and in an ever-so-friendly manner, teases out Dr. Skinner’s views of his upbringing and tags them to his behavorialism. It is this skill as an interviewer that captured us; can psychiatrists really be this clever? Yes, we can. All of the young and hopeful psychiatrists could see a future.
Dr. Clare raised psychiatry up from the depths of the asylums. He showed that a psychiatrist can be kind, charming, and sophisticated – handsome and helpful, not the ghouls of old movies. He did what needed to be done to psychiatry at that time: He set us on a footing that was not scary to the public. His vision for psychiatry was to improve services to those in need, reduce stigma, and show the public that there is a continuum between health and illness. He also took on the push for diagnoses, which he felt separated the normal from the abnormal, us from them.
He wrote his seminal work 10 years after graduating from the University College of Dublin. “Psychiatry in Dissent: Controversial Issues in Thought and Practice” was published in 1976, and is still considered one of the most influential texts in psychiatry. Dr. Clare “legitimized psychiatry not only in the eyes of the public but in the eyes of psychiatrists too,” the authors wrote. He did not support psychoanalysis and eschewed the rigor attached to the learning of new psychotherapies. He took renowned experts to task, but in ever such an elegant way. He successfully took on Hans Eysenck, PhD, I think because Dr. Eysenck insulted the intelligence of the Irish. He had a measured response to the anti-psychiatrists Thomas Szasz, MD, and R.D. Laing, MD, incorporating their ideas into his view of psychiatry. Dr. Clare was a social psychiatrist who highlighted the role of poverty and lack of access to mental health services. He stated that psychiatry was a “shambles, a mess and at a very primitive level.”
I enjoyed learning about his fight to make the membership exam for entrance into the Royal College of Psychiatry worthy of its name. Dr. Clare helped found the Association of Psychiatrists in Training (APIT) and wrote eloquently about the difference between training and indoctrination, which he described as having people fit a predetermined paradigm of how psychiatry should be constructed and practiced, versus education, which he defined as forming the mind. He highlighted the lack of good training facilities, and teaching staff in many parts of the United Kingdom. When Dr. Clare studied candidates in Edinburgh, he found that 70% had no child, forensic, or intellectual disability training. By the time I did my training there, I was able to get experience in all three subspecialties. He opposed the granting of automatic membership to current consultants, many of whom he considered to be “dunderheads.” . I can attest to that!
In the later phase of his life, Dr. Clare likened his self-punishing regime at the height of his hyperproductive fame to an addiction – a fix, with its risk/reward, pain/pleasure kick. He identified fear as being an unacknowledged presence in most of his life. There are vague hints from Dr. Clare’s friends and colleagues that something drove him back to Ireland from a successful life in London. Although his wife was described as being fully supportive of him, her words on his tombstone indicate something: What they indicate you can decide. She called him “a loving husband, father and grandfather, orator, physician, writer and broadcaster.” No mention was made of his being one of the greatest psychiatrists of his generation.
In 2000, he wrote “On Men: Masculinity in Crisis,” about men and the patriarchy, and highlighted the concept of “performance-based self-worth” in men. He stated: “What is the point of an awful lot of what I do. I’m in my 50s. I think one should be spending a good deal of your time doing things you want to do ... and what is that? I want to see much more of my family and friends. I want to continue making a contribution, but how can I best do that? ... I am contaminated by patriarchy; there is no man who isn’t. There is hope for men only if they ‘acknowledge the end of patriarchal power and participate in the discussion of how the post-patriarchal age is to be negotiated”. He opined whether it is still the case that, if men do not reevaluate their roles, they will soon be entirely irrelevant as social beings. The value of men is less in income generation but more in cultivating involvement, awareness, consistency, and caring, he stated.
As always with famous and talented people, we are interested not only in their professional gifts to us but in their personal journeys, and the authors, Dr. Kelly and Dr. Houston have given us this rich profile of one of my lifelong heroes, Anthony Clare. Anthony Clare makes you feel good about being a psychiatrist, and that is such an important gift.
Dr. Heru is professor of psychiatry at the University of Colorado at Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest.
New biography captures Dr. Anthony Clare’s complexity
New biography captures Dr. Anthony Clare’s complexity
In “Psychiatrist in the Chair,” authors Brendan Kelly and Muiris Houston tell the story of a fellow Irishman, Anthony Clare, MD, who brought intelligence and eloquence to psychiatry. They tell a well-measured, well-referenced story of Anthony Clare’s personal and professional life. They capture his eloquence, wit, charm, and success in psychiatry as well as alluding to Dr. Clare’s self-reported “some kind of Irish darkness.”
In 1983, I was a young Scottish psychiatrist entering a fusty profession. Suddenly, there was Dr. Anthony Clare on the BBC! In “In the Psychiatrist’s Chair,” Dr. Clare interviewed celebrities. In addition to describing his Irish darkness, Brendan Kelly, MD, PhD and Muiris Houston, MD, FRCGP, both of whom are affiliated with Trinity College Dublin, note that Dr. Clare said: “I’m better at destroying systems than I am at putting them together – I do rather look for people to interview who will not live up to the prediction; there’s an element of destructiveness that’s still in me.”
I still listen to his talks on YouTube. His delicate probing questioning of B.F. Skinner, PhD, is one of my favorites, as he expertly and in an ever-so-friendly manner, teases out Dr. Skinner’s views of his upbringing and tags them to his behavorialism. It is this skill as an interviewer that captured us; can psychiatrists really be this clever? Yes, we can. All of the young and hopeful psychiatrists could see a future.
Dr. Clare raised psychiatry up from the depths of the asylums. He showed that a psychiatrist can be kind, charming, and sophisticated – handsome and helpful, not the ghouls of old movies. He did what needed to be done to psychiatry at that time: He set us on a footing that was not scary to the public. His vision for psychiatry was to improve services to those in need, reduce stigma, and show the public that there is a continuum between health and illness. He also took on the push for diagnoses, which he felt separated the normal from the abnormal, us from them.
He wrote his seminal work 10 years after graduating from the University College of Dublin. “Psychiatry in Dissent: Controversial Issues in Thought and Practice” was published in 1976, and is still considered one of the most influential texts in psychiatry. Dr. Clare “legitimized psychiatry not only in the eyes of the public but in the eyes of psychiatrists too,” the authors wrote. He did not support psychoanalysis and eschewed the rigor attached to the learning of new psychotherapies. He took renowned experts to task, but in ever such an elegant way. He successfully took on Hans Eysenck, PhD, I think because Dr. Eysenck insulted the intelligence of the Irish. He had a measured response to the anti-psychiatrists Thomas Szasz, MD, and R.D. Laing, MD, incorporating their ideas into his view of psychiatry. Dr. Clare was a social psychiatrist who highlighted the role of poverty and lack of access to mental health services. He stated that psychiatry was a “shambles, a mess and at a very primitive level.”
I enjoyed learning about his fight to make the membership exam for entrance into the Royal College of Psychiatry worthy of its name. Dr. Clare helped found the Association of Psychiatrists in Training (APIT) and wrote eloquently about the difference between training and indoctrination, which he described as having people fit a predetermined paradigm of how psychiatry should be constructed and practiced, versus education, which he defined as forming the mind. He highlighted the lack of good training facilities, and teaching staff in many parts of the United Kingdom. When Dr. Clare studied candidates in Edinburgh, he found that 70% had no child, forensic, or intellectual disability training. By the time I did my training there, I was able to get experience in all three subspecialties. He opposed the granting of automatic membership to current consultants, many of whom he considered to be “dunderheads.” . I can attest to that!
In the later phase of his life, Dr. Clare likened his self-punishing regime at the height of his hyperproductive fame to an addiction – a fix, with its risk/reward, pain/pleasure kick. He identified fear as being an unacknowledged presence in most of his life. There are vague hints from Dr. Clare’s friends and colleagues that something drove him back to Ireland from a successful life in London. Although his wife was described as being fully supportive of him, her words on his tombstone indicate something: What they indicate you can decide. She called him “a loving husband, father and grandfather, orator, physician, writer and broadcaster.” No mention was made of his being one of the greatest psychiatrists of his generation.
In 2000, he wrote “On Men: Masculinity in Crisis,” about men and the patriarchy, and highlighted the concept of “performance-based self-worth” in men. He stated: “What is the point of an awful lot of what I do. I’m in my 50s. I think one should be spending a good deal of your time doing things you want to do ... and what is that? I want to see much more of my family and friends. I want to continue making a contribution, but how can I best do that? ... I am contaminated by patriarchy; there is no man who isn’t. There is hope for men only if they ‘acknowledge the end of patriarchal power and participate in the discussion of how the post-patriarchal age is to be negotiated”. He opined whether it is still the case that, if men do not reevaluate their roles, they will soon be entirely irrelevant as social beings. The value of men is less in income generation but more in cultivating involvement, awareness, consistency, and caring, he stated.
As always with famous and talented people, we are interested not only in their professional gifts to us but in their personal journeys, and the authors, Dr. Kelly and Dr. Houston have given us this rich profile of one of my lifelong heroes, Anthony Clare. Anthony Clare makes you feel good about being a psychiatrist, and that is such an important gift.
Dr. Heru is professor of psychiatry at the University of Colorado at Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest.
In “Psychiatrist in the Chair,” authors Brendan Kelly and Muiris Houston tell the story of a fellow Irishman, Anthony Clare, MD, who brought intelligence and eloquence to psychiatry. They tell a well-measured, well-referenced story of Anthony Clare’s personal and professional life. They capture his eloquence, wit, charm, and success in psychiatry as well as alluding to Dr. Clare’s self-reported “some kind of Irish darkness.”
In 1983, I was a young Scottish psychiatrist entering a fusty profession. Suddenly, there was Dr. Anthony Clare on the BBC! In “In the Psychiatrist’s Chair,” Dr. Clare interviewed celebrities. In addition to describing his Irish darkness, Brendan Kelly, MD, PhD and Muiris Houston, MD, FRCGP, both of whom are affiliated with Trinity College Dublin, note that Dr. Clare said: “I’m better at destroying systems than I am at putting them together – I do rather look for people to interview who will not live up to the prediction; there’s an element of destructiveness that’s still in me.”
I still listen to his talks on YouTube. His delicate probing questioning of B.F. Skinner, PhD, is one of my favorites, as he expertly and in an ever-so-friendly manner, teases out Dr. Skinner’s views of his upbringing and tags them to his behavorialism. It is this skill as an interviewer that captured us; can psychiatrists really be this clever? Yes, we can. All of the young and hopeful psychiatrists could see a future.
Dr. Clare raised psychiatry up from the depths of the asylums. He showed that a psychiatrist can be kind, charming, and sophisticated – handsome and helpful, not the ghouls of old movies. He did what needed to be done to psychiatry at that time: He set us on a footing that was not scary to the public. His vision for psychiatry was to improve services to those in need, reduce stigma, and show the public that there is a continuum between health and illness. He also took on the push for diagnoses, which he felt separated the normal from the abnormal, us from them.
He wrote his seminal work 10 years after graduating from the University College of Dublin. “Psychiatry in Dissent: Controversial Issues in Thought and Practice” was published in 1976, and is still considered one of the most influential texts in psychiatry. Dr. Clare “legitimized psychiatry not only in the eyes of the public but in the eyes of psychiatrists too,” the authors wrote. He did not support psychoanalysis and eschewed the rigor attached to the learning of new psychotherapies. He took renowned experts to task, but in ever such an elegant way. He successfully took on Hans Eysenck, PhD, I think because Dr. Eysenck insulted the intelligence of the Irish. He had a measured response to the anti-psychiatrists Thomas Szasz, MD, and R.D. Laing, MD, incorporating their ideas into his view of psychiatry. Dr. Clare was a social psychiatrist who highlighted the role of poverty and lack of access to mental health services. He stated that psychiatry was a “shambles, a mess and at a very primitive level.”
I enjoyed learning about his fight to make the membership exam for entrance into the Royal College of Psychiatry worthy of its name. Dr. Clare helped found the Association of Psychiatrists in Training (APIT) and wrote eloquently about the difference between training and indoctrination, which he described as having people fit a predetermined paradigm of how psychiatry should be constructed and practiced, versus education, which he defined as forming the mind. He highlighted the lack of good training facilities, and teaching staff in many parts of the United Kingdom. When Dr. Clare studied candidates in Edinburgh, he found that 70% had no child, forensic, or intellectual disability training. By the time I did my training there, I was able to get experience in all three subspecialties. He opposed the granting of automatic membership to current consultants, many of whom he considered to be “dunderheads.” . I can attest to that!
In the later phase of his life, Dr. Clare likened his self-punishing regime at the height of his hyperproductive fame to an addiction – a fix, with its risk/reward, pain/pleasure kick. He identified fear as being an unacknowledged presence in most of his life. There are vague hints from Dr. Clare’s friends and colleagues that something drove him back to Ireland from a successful life in London. Although his wife was described as being fully supportive of him, her words on his tombstone indicate something: What they indicate you can decide. She called him “a loving husband, father and grandfather, orator, physician, writer and broadcaster.” No mention was made of his being one of the greatest psychiatrists of his generation.
In 2000, he wrote “On Men: Masculinity in Crisis,” about men and the patriarchy, and highlighted the concept of “performance-based self-worth” in men. He stated: “What is the point of an awful lot of what I do. I’m in my 50s. I think one should be spending a good deal of your time doing things you want to do ... and what is that? I want to see much more of my family and friends. I want to continue making a contribution, but how can I best do that? ... I am contaminated by patriarchy; there is no man who isn’t. There is hope for men only if they ‘acknowledge the end of patriarchal power and participate in the discussion of how the post-patriarchal age is to be negotiated”. He opined whether it is still the case that, if men do not reevaluate their roles, they will soon be entirely irrelevant as social beings. The value of men is less in income generation but more in cultivating involvement, awareness, consistency, and caring, he stated.
As always with famous and talented people, we are interested not only in their professional gifts to us but in their personal journeys, and the authors, Dr. Kelly and Dr. Houston have given us this rich profile of one of my lifelong heroes, Anthony Clare. Anthony Clare makes you feel good about being a psychiatrist, and that is such an important gift.
Dr. Heru is professor of psychiatry at the University of Colorado at Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest.
RARE DISEASES REPORT: Cancers
Rare cancers, though individually rare by definition, impose a tremendous burden on adult and pediatric patient populations, especially when considering hematological cancers. In this Rare Diseases Report: Cancers, we bring you the latest information on new and ongoing developments in the treatment of some of these cancers through interviews with frontline researchers in the field.
- Survey reveals special impact of COVID-19 on patients with rare disorders
- New agents boost survival – and complexity – in chronic lymphocytic leukemia
- Targeted therapies may alter the landscape of MCL treatment
- Will CAR T push beyond lymphoma? There’s no guarantee
Rare cancers, though individually rare by definition, impose a tremendous burden on adult and pediatric patient populations, especially when considering hematological cancers. In this Rare Diseases Report: Cancers, we bring you the latest information on new and ongoing developments in the treatment of some of these cancers through interviews with frontline researchers in the field.
- Survey reveals special impact of COVID-19 on patients with rare disorders
- New agents boost survival – and complexity – in chronic lymphocytic leukemia
- Targeted therapies may alter the landscape of MCL treatment
- Will CAR T push beyond lymphoma? There’s no guarantee
Rare cancers, though individually rare by definition, impose a tremendous burden on adult and pediatric patient populations, especially when considering hematological cancers. In this Rare Diseases Report: Cancers, we bring you the latest information on new and ongoing developments in the treatment of some of these cancers through interviews with frontline researchers in the field.
- Survey reveals special impact of COVID-19 on patients with rare disorders
- New agents boost survival – and complexity – in chronic lymphocytic leukemia
- Targeted therapies may alter the landscape of MCL treatment
- Will CAR T push beyond lymphoma? There’s no guarantee
BTK Inhibitors: Researchers Eye Next-Generation Treatment Options for MS
Bruton tyrosine kinase (BTK) inhibitors effectively treat certain leukemias and lymphomas, due to their ability to inhibit a protein kinase that is critical for B cell receptor signaling. These agents can inhibit antigen-triggered activation and maturation of B cells and their release of pro-inflammatory cytokines. BTK is also a key signaling protein that controls activation of monocytes, macrophages and neutrophils and its inhibition reduces the activation of these cells.
Judging from the papers presented at the recent MS Virtual 2020—the 8th Joint ACTRIMS-ECTRIMS Meeting, BTK inhibitors are showing promise in the management of multiple sclerosis (MS), as well, as researchers eye next-generation treatment options. They may prove to be effective not only in relapsing remitting multiple sclerosis, but also on progressive disease through their effect on brain macrophages and microglial cells.
Leading the way at MS Virtual 2020 was a presentation by Patrick Vermersch, MD, PhD, who summarized the results to date of masitinib’s role in primary progressive MS (PPMS) and non-active secondar progressive MS (nSPMS). The professor of neurology at Lille University in Lille, France, and his colleagues conducted a phase 3 study to assess masitinib’s efficacy at two dosage levels – 4.5 mg/kg/day (n=199) and a 6.0 mg/kg/day (n=199) -- vs placebo (n=101). The randomized, double-blinded, placebo controlled, two-parallel group trial involved adult participants with EDSS scores between 2.0 and 6.0 with either PPMS or nSPMS. Investigators looked at overall EDSS change from baseline using a variety of repeated measures, with an emphasis on determining whether participants improved, were stable, or worsened. Among the results
- In the lower-dose contingent, the odds of either a reduction in EDSS progression or increased EDSS improvement improved by 39% for patients taking masitinib, vs those receiving placebo
- Also, among those receiving the lower dose, the odds of experiencing disability progression decreased by 37% in the treatment group, compared with placebo
- Results using the higher dose of masitinib were inconclusive
Based on these results, investigators concluded that masitinib 4.5 mg/kg/day is very likely to emerge as a new treatment option for individuals with for PPMS and nSPMS.
Another MS Virtual 2020 presentation focused on fenebrutinib, an investigational, noncovalent investigational BTK inhibitor being studies as a possible treatment for MS. Investigators assessed the potency, selectivity, and kinetics of inhibition of the BTK enzyme via fenebrutinib, compared with two other BTK inhibitors, evobrutinib and tolebrutinib, in a panel of 219 human kinases.
Fenebrutinib was found to be a more potent inhibitor, compared with the other two BTK inhibitors. Additionally, fenebrutinib effectively blocked B cell and basophil activation. Moreover, fenebrutinib inhibited fewer off target kinases >50%, compared with the other two BTK inhibitors. Investigators concluded that fenebrutinib’s high selectivity and potency shows promise as a drug linked with fewer adverse events and a better treatment profile than other investigational BTK inhibitors.
These encouraging findings are leading to the initiation of three phase 3 clinical trials involving fenebrutinib, announced at MS Virtual 2020 by Stephen L. Hauser, MD, professor of neurology, and director of the UCSF Weill Institute for Neurosciences. The trials will evaluate fenebrutinib’s impact on disease progression in relapsing MS and primary progressive MS. The primary endpoint of the studies will be 12-week composite Confirmed Disability Progression, which investigators hope will provide a stronger, more thorough assessment of disability progression, vs EDSS score alone.
Other BTK inhibitors worth watching include:
- Evobrutinib: An open-label phase 2 study has shown that the drug is linked with reduced annualized relapse rate through 108 weeks
- BIIB091: Safety and tolerability has been demonstrated in phase 1, clearing the way for further investigation
- Tolebrutinib: Under assessment for its ability to inhibit microglia-driven inflammation in murine models
Torke S, Pretzch R, Hausler D, et al. Inhibition of Bruton’s tyrosine kinase interferes with pathogenic B-cell development in inflammatory CNS demyelinating disease. Acta Neuropathol. 2020;140;535-548. doi: https://doi.org/10.1007/
Bruton tyrosine kinase (BTK) inhibitors effectively treat certain leukemias and lymphomas, due to their ability to inhibit a protein kinase that is critical for B cell receptor signaling. These agents can inhibit antigen-triggered activation and maturation of B cells and their release of pro-inflammatory cytokines. BTK is also a key signaling protein that controls activation of monocytes, macrophages and neutrophils and its inhibition reduces the activation of these cells.
Judging from the papers presented at the recent MS Virtual 2020—the 8th Joint ACTRIMS-ECTRIMS Meeting, BTK inhibitors are showing promise in the management of multiple sclerosis (MS), as well, as researchers eye next-generation treatment options. They may prove to be effective not only in relapsing remitting multiple sclerosis, but also on progressive disease through their effect on brain macrophages and microglial cells.
Leading the way at MS Virtual 2020 was a presentation by Patrick Vermersch, MD, PhD, who summarized the results to date of masitinib’s role in primary progressive MS (PPMS) and non-active secondar progressive MS (nSPMS). The professor of neurology at Lille University in Lille, France, and his colleagues conducted a phase 3 study to assess masitinib’s efficacy at two dosage levels – 4.5 mg/kg/day (n=199) and a 6.0 mg/kg/day (n=199) -- vs placebo (n=101). The randomized, double-blinded, placebo controlled, two-parallel group trial involved adult participants with EDSS scores between 2.0 and 6.0 with either PPMS or nSPMS. Investigators looked at overall EDSS change from baseline using a variety of repeated measures, with an emphasis on determining whether participants improved, were stable, or worsened. Among the results
- In the lower-dose contingent, the odds of either a reduction in EDSS progression or increased EDSS improvement improved by 39% for patients taking masitinib, vs those receiving placebo
- Also, among those receiving the lower dose, the odds of experiencing disability progression decreased by 37% in the treatment group, compared with placebo
- Results using the higher dose of masitinib were inconclusive
Based on these results, investigators concluded that masitinib 4.5 mg/kg/day is very likely to emerge as a new treatment option for individuals with for PPMS and nSPMS.
Another MS Virtual 2020 presentation focused on fenebrutinib, an investigational, noncovalent investigational BTK inhibitor being studies as a possible treatment for MS. Investigators assessed the potency, selectivity, and kinetics of inhibition of the BTK enzyme via fenebrutinib, compared with two other BTK inhibitors, evobrutinib and tolebrutinib, in a panel of 219 human kinases.
Fenebrutinib was found to be a more potent inhibitor, compared with the other two BTK inhibitors. Additionally, fenebrutinib effectively blocked B cell and basophil activation. Moreover, fenebrutinib inhibited fewer off target kinases >50%, compared with the other two BTK inhibitors. Investigators concluded that fenebrutinib’s high selectivity and potency shows promise as a drug linked with fewer adverse events and a better treatment profile than other investigational BTK inhibitors.
These encouraging findings are leading to the initiation of three phase 3 clinical trials involving fenebrutinib, announced at MS Virtual 2020 by Stephen L. Hauser, MD, professor of neurology, and director of the UCSF Weill Institute for Neurosciences. The trials will evaluate fenebrutinib’s impact on disease progression in relapsing MS and primary progressive MS. The primary endpoint of the studies will be 12-week composite Confirmed Disability Progression, which investigators hope will provide a stronger, more thorough assessment of disability progression, vs EDSS score alone.
Other BTK inhibitors worth watching include:
- Evobrutinib: An open-label phase 2 study has shown that the drug is linked with reduced annualized relapse rate through 108 weeks
- BIIB091: Safety and tolerability has been demonstrated in phase 1, clearing the way for further investigation
- Tolebrutinib: Under assessment for its ability to inhibit microglia-driven inflammation in murine models
Bruton tyrosine kinase (BTK) inhibitors effectively treat certain leukemias and lymphomas, due to their ability to inhibit a protein kinase that is critical for B cell receptor signaling. These agents can inhibit antigen-triggered activation and maturation of B cells and their release of pro-inflammatory cytokines. BTK is also a key signaling protein that controls activation of monocytes, macrophages and neutrophils and its inhibition reduces the activation of these cells.
Judging from the papers presented at the recent MS Virtual 2020—the 8th Joint ACTRIMS-ECTRIMS Meeting, BTK inhibitors are showing promise in the management of multiple sclerosis (MS), as well, as researchers eye next-generation treatment options. They may prove to be effective not only in relapsing remitting multiple sclerosis, but also on progressive disease through their effect on brain macrophages and microglial cells.
Leading the way at MS Virtual 2020 was a presentation by Patrick Vermersch, MD, PhD, who summarized the results to date of masitinib’s role in primary progressive MS (PPMS) and non-active secondar progressive MS (nSPMS). The professor of neurology at Lille University in Lille, France, and his colleagues conducted a phase 3 study to assess masitinib’s efficacy at two dosage levels – 4.5 mg/kg/day (n=199) and a 6.0 mg/kg/day (n=199) -- vs placebo (n=101). The randomized, double-blinded, placebo controlled, two-parallel group trial involved adult participants with EDSS scores between 2.0 and 6.0 with either PPMS or nSPMS. Investigators looked at overall EDSS change from baseline using a variety of repeated measures, with an emphasis on determining whether participants improved, were stable, or worsened. Among the results
- In the lower-dose contingent, the odds of either a reduction in EDSS progression or increased EDSS improvement improved by 39% for patients taking masitinib, vs those receiving placebo
- Also, among those receiving the lower dose, the odds of experiencing disability progression decreased by 37% in the treatment group, compared with placebo
- Results using the higher dose of masitinib were inconclusive
Based on these results, investigators concluded that masitinib 4.5 mg/kg/day is very likely to emerge as a new treatment option for individuals with for PPMS and nSPMS.
Another MS Virtual 2020 presentation focused on fenebrutinib, an investigational, noncovalent investigational BTK inhibitor being studies as a possible treatment for MS. Investigators assessed the potency, selectivity, and kinetics of inhibition of the BTK enzyme via fenebrutinib, compared with two other BTK inhibitors, evobrutinib and tolebrutinib, in a panel of 219 human kinases.
Fenebrutinib was found to be a more potent inhibitor, compared with the other two BTK inhibitors. Additionally, fenebrutinib effectively blocked B cell and basophil activation. Moreover, fenebrutinib inhibited fewer off target kinases >50%, compared with the other two BTK inhibitors. Investigators concluded that fenebrutinib’s high selectivity and potency shows promise as a drug linked with fewer adverse events and a better treatment profile than other investigational BTK inhibitors.
These encouraging findings are leading to the initiation of three phase 3 clinical trials involving fenebrutinib, announced at MS Virtual 2020 by Stephen L. Hauser, MD, professor of neurology, and director of the UCSF Weill Institute for Neurosciences. The trials will evaluate fenebrutinib’s impact on disease progression in relapsing MS and primary progressive MS. The primary endpoint of the studies will be 12-week composite Confirmed Disability Progression, which investigators hope will provide a stronger, more thorough assessment of disability progression, vs EDSS score alone.
Other BTK inhibitors worth watching include:
- Evobrutinib: An open-label phase 2 study has shown that the drug is linked with reduced annualized relapse rate through 108 weeks
- BIIB091: Safety and tolerability has been demonstrated in phase 1, clearing the way for further investigation
- Tolebrutinib: Under assessment for its ability to inhibit microglia-driven inflammation in murine models
Torke S, Pretzch R, Hausler D, et al. Inhibition of Bruton’s tyrosine kinase interferes with pathogenic B-cell development in inflammatory CNS demyelinating disease. Acta Neuropathol. 2020;140;535-548. doi: https://doi.org/10.1007/
Torke S, Pretzch R, Hausler D, et al. Inhibition of Bruton’s tyrosine kinase interferes with pathogenic B-cell development in inflammatory CNS demyelinating disease. Acta Neuropathol. 2020;140;535-548. doi: https://doi.org/10.1007/
Adding atezolizumab to chemo doesn’t worsen QOL in early TNBC
In the randomized phase 3 trial, patients received neoadjuvant atezolizumab or placebo plus chemotherapy, followed by surgery and adjuvant atezolizumab or observation.
An analysis of patient-reported outcomes showed that, after a brief decline in health-related quality of life (HRQOL) in both the atezolizumab and control arms, the burden of treatment eased and then remained similar throughout follow-up.
“Treatment-related symptoms in both arms were similar, and the addition of atezolizumab to chemotherapy was tolerable, with no additional treatment side-effect bother reported,” said Elizabeth Mittendorf, MD, PhD, of the Dana-Farber Cancer Institute in Boston.
She presented these results in an oral abstract presentation during the 2020 San Antonio Breast Cancer Symposium.
Initial results
Primary results of the IMpassion031 trial were reported at ESMO 2020 and published in The Lancet.
The trial enrolled patients with treatment-naive early TNBC, and they were randomized to receive chemotherapy plus atezolizumab or placebo. Atezolizumab was given at 840 mg intravenously every 2 weeks.
Chemotherapy consisted of nab-paclitaxel at 125 mg/m2 every week for 12 weeks followed by doxorubicin at 60 mg/m2 and cyclophosphamide at 600 mg/m2 every 2 weeks for 8 weeks. Patients then underwent surgery, which was followed by either atezolizumab maintenance or observation.
The addition of atezolizumab was associated with a 17% improvement in the rate of pathological complete response (pCR) in the intention-to-treat population.
Among patients positive for PD-L1, atezolizumab was associated with a 20% improvement in pCR rate.
Patients have their say
At SABCS 2020, Dr. Mittendorf presented patient-reported outcomes (PRO) for 161 patients randomized to atezolizumab plus chemotherapy and 167 assigned to placebo plus chemotherapy.
The outcome measures – including role function (the ability to work or pursue common everyday activities), physical function, emotional and social function, and HRQOL – came from scores on the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Core 30 (QLQ-C30).
The exploratory PRO endpoints were mean function and disease- or treatment-related symptoms as well as mean change from baseline in these symptoms.
At baseline, mean physical function scores were high in both arms, at approximately 90%. They dropped to about 65% in each arm by cycle 5 and rebounded to about 80% by cycle 7.
“The change was clinically meaningful, in that the dip was greater than 10 points,” Dr. Mittendorf said. “As patients completed neoadjuvant therapy and were in the adjuvant phase of the study, there was an increase and stabilization with respect to their physical function.”
Similarly, role function declined from 89% in each arm at baseline to a nadir of about 55% in the placebo arm and 50% in the atezolizumab arm. After cycle 5, role function levels began to increase in both arms and remained above 70% from cycle 7 onward.
Role function did not completely recover or stabilize among patients in the atezolizumab arm, “and we attribute this to the fact that these patients continue to receive atezolizumab, which requires that they come to clinic for that therapy, thereby potentially impacting role function,” Dr. Mittendorf said.
She reported that HRQOL declined from a median of nearly 80% at baseline in both arms to a nadir of 62% in the placebo arm and 52% in the atezolizumab arm by cycle 5. HRQOL rebounded starting at cycle 6 and stabilized thereafter, with little daylight between the treatment arms at the most recent follow-up.
The investigators saw worsening of the treatment-related symptoms fatigue, nausea/vomiting, and diarrhea in each arm, with the highest level of symptoms except pain reported at cycle 5. The highest reported pain level was seen at cycle 4.
In each arm, the symptoms declined and stabilized in the adjuvant setting, with mean values at week 16 similar to those reported at baseline for most symptoms. The exception was fatigue, which remained slightly elevated in both arms.
Regarding side-effect bother, a similar proportion of patients in each arm reported increased level of bother by visit during the neoadjuvant phase. However, no additional side-effect bother was reported by patients receiving maintenance atezolizumab, compared with patients in the placebo arm, who were followed with observation alone.
‘Reassuring’ data
“What we can conclude from the PRO in IMpassion031 is that early breast cancer patients are relatively asymptomatic. They do have a good baseline quality of life and very good functioning. The neoadjuvant therapy certainly has deterioration in quality of life and functioning, but this is transient, and there was no evident burden from the atezolizumab,” said invited discussant Sylvia Adams, MD, of New York University.
She said it’s reassuring to see the addition of atezolizumab did not adversely affect HRQOL but added that the study “also shows that chemotherapy has a major impact on well-being, as expected.”
Questions and problems that still need to be addressed regarding the use of immunotherapies in early TNBC include whether every patient needs chemotherapy or immunotherapy, a lack of predictive biomarkers, whether increases in pCR rates after neoadjuvant immunotherapy and chemotherapy will translate into improved survival, and the optimal chemotherapy backbone and schedule, she said.
IMpassion031 is sponsored by F. Hoffman-LaRoche. Dr. Mittendorf disclosed relationships with Roche/Genentech, GlaxoSmithKline, Physicians’ Education Resource, AstraZeneca, Exact Sciences, Merck, Peregrine Pharmaceuticals, SELLAS Life Sciences, TapImmune, EMD Serono, Galena Biopharma, Bristol Myers Squibb, and Lilly. Dr. Adams disclosed relationships with Genentech, Bristol Myers Squibb, Merck, Amgen, Celgene, and Novartis.
SOURCE: Mittendorf E at al. SABCS 2020, Abstract GS3-02.
In the randomized phase 3 trial, patients received neoadjuvant atezolizumab or placebo plus chemotherapy, followed by surgery and adjuvant atezolizumab or observation.
An analysis of patient-reported outcomes showed that, after a brief decline in health-related quality of life (HRQOL) in both the atezolizumab and control arms, the burden of treatment eased and then remained similar throughout follow-up.
“Treatment-related symptoms in both arms were similar, and the addition of atezolizumab to chemotherapy was tolerable, with no additional treatment side-effect bother reported,” said Elizabeth Mittendorf, MD, PhD, of the Dana-Farber Cancer Institute in Boston.
She presented these results in an oral abstract presentation during the 2020 San Antonio Breast Cancer Symposium.
Initial results
Primary results of the IMpassion031 trial were reported at ESMO 2020 and published in The Lancet.
The trial enrolled patients with treatment-naive early TNBC, and they were randomized to receive chemotherapy plus atezolizumab or placebo. Atezolizumab was given at 840 mg intravenously every 2 weeks.
Chemotherapy consisted of nab-paclitaxel at 125 mg/m2 every week for 12 weeks followed by doxorubicin at 60 mg/m2 and cyclophosphamide at 600 mg/m2 every 2 weeks for 8 weeks. Patients then underwent surgery, which was followed by either atezolizumab maintenance or observation.
The addition of atezolizumab was associated with a 17% improvement in the rate of pathological complete response (pCR) in the intention-to-treat population.
Among patients positive for PD-L1, atezolizumab was associated with a 20% improvement in pCR rate.
Patients have their say
At SABCS 2020, Dr. Mittendorf presented patient-reported outcomes (PRO) for 161 patients randomized to atezolizumab plus chemotherapy and 167 assigned to placebo plus chemotherapy.
The outcome measures – including role function (the ability to work or pursue common everyday activities), physical function, emotional and social function, and HRQOL – came from scores on the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Core 30 (QLQ-C30).
The exploratory PRO endpoints were mean function and disease- or treatment-related symptoms as well as mean change from baseline in these symptoms.
At baseline, mean physical function scores were high in both arms, at approximately 90%. They dropped to about 65% in each arm by cycle 5 and rebounded to about 80% by cycle 7.
“The change was clinically meaningful, in that the dip was greater than 10 points,” Dr. Mittendorf said. “As patients completed neoadjuvant therapy and were in the adjuvant phase of the study, there was an increase and stabilization with respect to their physical function.”
Similarly, role function declined from 89% in each arm at baseline to a nadir of about 55% in the placebo arm and 50% in the atezolizumab arm. After cycle 5, role function levels began to increase in both arms and remained above 70% from cycle 7 onward.
Role function did not completely recover or stabilize among patients in the atezolizumab arm, “and we attribute this to the fact that these patients continue to receive atezolizumab, which requires that they come to clinic for that therapy, thereby potentially impacting role function,” Dr. Mittendorf said.
She reported that HRQOL declined from a median of nearly 80% at baseline in both arms to a nadir of 62% in the placebo arm and 52% in the atezolizumab arm by cycle 5. HRQOL rebounded starting at cycle 6 and stabilized thereafter, with little daylight between the treatment arms at the most recent follow-up.
The investigators saw worsening of the treatment-related symptoms fatigue, nausea/vomiting, and diarrhea in each arm, with the highest level of symptoms except pain reported at cycle 5. The highest reported pain level was seen at cycle 4.
In each arm, the symptoms declined and stabilized in the adjuvant setting, with mean values at week 16 similar to those reported at baseline for most symptoms. The exception was fatigue, which remained slightly elevated in both arms.
Regarding side-effect bother, a similar proportion of patients in each arm reported increased level of bother by visit during the neoadjuvant phase. However, no additional side-effect bother was reported by patients receiving maintenance atezolizumab, compared with patients in the placebo arm, who were followed with observation alone.
‘Reassuring’ data
“What we can conclude from the PRO in IMpassion031 is that early breast cancer patients are relatively asymptomatic. They do have a good baseline quality of life and very good functioning. The neoadjuvant therapy certainly has deterioration in quality of life and functioning, but this is transient, and there was no evident burden from the atezolizumab,” said invited discussant Sylvia Adams, MD, of New York University.
She said it’s reassuring to see the addition of atezolizumab did not adversely affect HRQOL but added that the study “also shows that chemotherapy has a major impact on well-being, as expected.”
Questions and problems that still need to be addressed regarding the use of immunotherapies in early TNBC include whether every patient needs chemotherapy or immunotherapy, a lack of predictive biomarkers, whether increases in pCR rates after neoadjuvant immunotherapy and chemotherapy will translate into improved survival, and the optimal chemotherapy backbone and schedule, she said.
IMpassion031 is sponsored by F. Hoffman-LaRoche. Dr. Mittendorf disclosed relationships with Roche/Genentech, GlaxoSmithKline, Physicians’ Education Resource, AstraZeneca, Exact Sciences, Merck, Peregrine Pharmaceuticals, SELLAS Life Sciences, TapImmune, EMD Serono, Galena Biopharma, Bristol Myers Squibb, and Lilly. Dr. Adams disclosed relationships with Genentech, Bristol Myers Squibb, Merck, Amgen, Celgene, and Novartis.
SOURCE: Mittendorf E at al. SABCS 2020, Abstract GS3-02.
In the randomized phase 3 trial, patients received neoadjuvant atezolizumab or placebo plus chemotherapy, followed by surgery and adjuvant atezolizumab or observation.
An analysis of patient-reported outcomes showed that, after a brief decline in health-related quality of life (HRQOL) in both the atezolizumab and control arms, the burden of treatment eased and then remained similar throughout follow-up.
“Treatment-related symptoms in both arms were similar, and the addition of atezolizumab to chemotherapy was tolerable, with no additional treatment side-effect bother reported,” said Elizabeth Mittendorf, MD, PhD, of the Dana-Farber Cancer Institute in Boston.
She presented these results in an oral abstract presentation during the 2020 San Antonio Breast Cancer Symposium.
Initial results
Primary results of the IMpassion031 trial were reported at ESMO 2020 and published in The Lancet.
The trial enrolled patients with treatment-naive early TNBC, and they were randomized to receive chemotherapy plus atezolizumab or placebo. Atezolizumab was given at 840 mg intravenously every 2 weeks.
Chemotherapy consisted of nab-paclitaxel at 125 mg/m2 every week for 12 weeks followed by doxorubicin at 60 mg/m2 and cyclophosphamide at 600 mg/m2 every 2 weeks for 8 weeks. Patients then underwent surgery, which was followed by either atezolizumab maintenance or observation.
The addition of atezolizumab was associated with a 17% improvement in the rate of pathological complete response (pCR) in the intention-to-treat population.
Among patients positive for PD-L1, atezolizumab was associated with a 20% improvement in pCR rate.
Patients have their say
At SABCS 2020, Dr. Mittendorf presented patient-reported outcomes (PRO) for 161 patients randomized to atezolizumab plus chemotherapy and 167 assigned to placebo plus chemotherapy.
The outcome measures – including role function (the ability to work or pursue common everyday activities), physical function, emotional and social function, and HRQOL – came from scores on the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Core 30 (QLQ-C30).
The exploratory PRO endpoints were mean function and disease- or treatment-related symptoms as well as mean change from baseline in these symptoms.
At baseline, mean physical function scores were high in both arms, at approximately 90%. They dropped to about 65% in each arm by cycle 5 and rebounded to about 80% by cycle 7.
“The change was clinically meaningful, in that the dip was greater than 10 points,” Dr. Mittendorf said. “As patients completed neoadjuvant therapy and were in the adjuvant phase of the study, there was an increase and stabilization with respect to their physical function.”
Similarly, role function declined from 89% in each arm at baseline to a nadir of about 55% in the placebo arm and 50% in the atezolizumab arm. After cycle 5, role function levels began to increase in both arms and remained above 70% from cycle 7 onward.
Role function did not completely recover or stabilize among patients in the atezolizumab arm, “and we attribute this to the fact that these patients continue to receive atezolizumab, which requires that they come to clinic for that therapy, thereby potentially impacting role function,” Dr. Mittendorf said.
She reported that HRQOL declined from a median of nearly 80% at baseline in both arms to a nadir of 62% in the placebo arm and 52% in the atezolizumab arm by cycle 5. HRQOL rebounded starting at cycle 6 and stabilized thereafter, with little daylight between the treatment arms at the most recent follow-up.
The investigators saw worsening of the treatment-related symptoms fatigue, nausea/vomiting, and diarrhea in each arm, with the highest level of symptoms except pain reported at cycle 5. The highest reported pain level was seen at cycle 4.
In each arm, the symptoms declined and stabilized in the adjuvant setting, with mean values at week 16 similar to those reported at baseline for most symptoms. The exception was fatigue, which remained slightly elevated in both arms.
Regarding side-effect bother, a similar proportion of patients in each arm reported increased level of bother by visit during the neoadjuvant phase. However, no additional side-effect bother was reported by patients receiving maintenance atezolizumab, compared with patients in the placebo arm, who were followed with observation alone.
‘Reassuring’ data
“What we can conclude from the PRO in IMpassion031 is that early breast cancer patients are relatively asymptomatic. They do have a good baseline quality of life and very good functioning. The neoadjuvant therapy certainly has deterioration in quality of life and functioning, but this is transient, and there was no evident burden from the atezolizumab,” said invited discussant Sylvia Adams, MD, of New York University.
She said it’s reassuring to see the addition of atezolizumab did not adversely affect HRQOL but added that the study “also shows that chemotherapy has a major impact on well-being, as expected.”
Questions and problems that still need to be addressed regarding the use of immunotherapies in early TNBC include whether every patient needs chemotherapy or immunotherapy, a lack of predictive biomarkers, whether increases in pCR rates after neoadjuvant immunotherapy and chemotherapy will translate into improved survival, and the optimal chemotherapy backbone and schedule, she said.
IMpassion031 is sponsored by F. Hoffman-LaRoche. Dr. Mittendorf disclosed relationships with Roche/Genentech, GlaxoSmithKline, Physicians’ Education Resource, AstraZeneca, Exact Sciences, Merck, Peregrine Pharmaceuticals, SELLAS Life Sciences, TapImmune, EMD Serono, Galena Biopharma, Bristol Myers Squibb, and Lilly. Dr. Adams disclosed relationships with Genentech, Bristol Myers Squibb, Merck, Amgen, Celgene, and Novartis.
SOURCE: Mittendorf E at al. SABCS 2020, Abstract GS3-02.
FROM SABCS 2020
Four factors may predict better survival with cabozantinib in mRCC
Starting cabozantinib at 60 mg/day, prior nephrectomy, favorable- or intermediate-risk disease, and body mass index of 25 kg/m2 or higher were all significantly associated with better OS.
These findings were based on data from the early access program of the CABOREAL study and were recently published in the European Journal of Cancer.
“The CABOREAL study describes cabozantinib use in a real-life setting in the largest unselected population to date of patients with mRCC,” lead author Laurence Albiges, MD, PhD, of the Gustave Roussy Cancer Center in Villejuif, France, and colleagues wrote.
The retrospective study enrolled 410 patients with mRCC who were treated with at least one dose of cabozantinib between September 2016 and February 2018. Clinical data were collected from medical records at 26 oncology centers in France.
The researchers evaluated the real-world use of cabozantinib, including duration of therapy, treatment discontinuations, and dose changes. OS and predictive factors of OS were assessed as well.
The median age of study participants was 63.0 years (range, 56.0-70.0 years). Roughly a third of patients (33.4%) received two prior lines of therapy (33.4%), and 41.2% received three or more lines of therapy before cabozantinib. Overall, 85.6% of patients had clear cell histology.
The median duration of cabozantinib treatment was 7.6 months (range, 3.2-15.7 months). The starting dose was 60 mg, 40 mg, and 20 mg in 70.9%, 26.7%, and 2.0% of patients, respectively.
The dose was decreased in 57.0% of patients, 58.7% required a dose modification, and 15.6% required a modified dose schedule. The median average daily dose was 40.0 mg (range, 13.9-60.0 mg).
Adverse events were the main reason for dose modification or treatment interruption. In all, 92.5% of patients had a modification because of an adverse event, and 85.0% had an interruption because of an adverse event.
Upon permanent discontinuation of cabozantinib, more than half of patients (54.4%) received subsequent therapy, including nivolumab (47.8%), axitinib (21.7%), and everolimus (19.0%).
The median OS was 14.4 months (95% confidence interval, 12.4-16.2 months), and the 1-year OS rate was 56.5% (95% CI, 51.5-61.2%).
Factors significantly associated with longer OS included cabozantinib initiation at 60 mg/day (P = .0486), prior nephrectomy (P = .0109), favorable or intermediate risk according to the International Metastatic RCC Database Consortium (P < .0001), and body mass index of 25 kg/m2 or higher (P = .0021).
“We report, for the first time, that the daily dose of 60 mg cabozantinib at initiation is an independent predictive factor of OS in a multivariate analysis,” the researchers wrote.
“It is interesting to see real-world studies like this to help to widen our understanding of how to utilize drugs like cabozantinib,” commented Simon Crabb, MBBS, PhD, of the University of Southampton (England).
“In general, we would expect a less favorable prognosis in patients with non-clear cell histology, likely in part part due to the underlying biology of the disease,” he added.
Dr. Albiges and colleagues acknowledged that the retrospective design and lack of a prospective safety evaluation were two key limitations of their study. However, the authors maintain that the reported cabozantinib use and exposure rates are indicative of the real-world setting.
This study was sponsored by Ipsen. Several authors disclosed financial relationships with Amgen, Astellas Pharma, AstraZeneca, Bristol-Myers Squibb, Exelixis, Ipsen, and numerous other companies. Dr. Crabb reported having no conflicts of interest related to this work.
SOURCE: Albiges L et al. Eur J Cancer. 2020 Nov 27. doi: 10.1016/j.ejca.2020.09.030.
Starting cabozantinib at 60 mg/day, prior nephrectomy, favorable- or intermediate-risk disease, and body mass index of 25 kg/m2 or higher were all significantly associated with better OS.
These findings were based on data from the early access program of the CABOREAL study and were recently published in the European Journal of Cancer.
“The CABOREAL study describes cabozantinib use in a real-life setting in the largest unselected population to date of patients with mRCC,” lead author Laurence Albiges, MD, PhD, of the Gustave Roussy Cancer Center in Villejuif, France, and colleagues wrote.
The retrospective study enrolled 410 patients with mRCC who were treated with at least one dose of cabozantinib between September 2016 and February 2018. Clinical data were collected from medical records at 26 oncology centers in France.
The researchers evaluated the real-world use of cabozantinib, including duration of therapy, treatment discontinuations, and dose changes. OS and predictive factors of OS were assessed as well.
The median age of study participants was 63.0 years (range, 56.0-70.0 years). Roughly a third of patients (33.4%) received two prior lines of therapy (33.4%), and 41.2% received three or more lines of therapy before cabozantinib. Overall, 85.6% of patients had clear cell histology.
The median duration of cabozantinib treatment was 7.6 months (range, 3.2-15.7 months). The starting dose was 60 mg, 40 mg, and 20 mg in 70.9%, 26.7%, and 2.0% of patients, respectively.
The dose was decreased in 57.0% of patients, 58.7% required a dose modification, and 15.6% required a modified dose schedule. The median average daily dose was 40.0 mg (range, 13.9-60.0 mg).
Adverse events were the main reason for dose modification or treatment interruption. In all, 92.5% of patients had a modification because of an adverse event, and 85.0% had an interruption because of an adverse event.
Upon permanent discontinuation of cabozantinib, more than half of patients (54.4%) received subsequent therapy, including nivolumab (47.8%), axitinib (21.7%), and everolimus (19.0%).
The median OS was 14.4 months (95% confidence interval, 12.4-16.2 months), and the 1-year OS rate was 56.5% (95% CI, 51.5-61.2%).
Factors significantly associated with longer OS included cabozantinib initiation at 60 mg/day (P = .0486), prior nephrectomy (P = .0109), favorable or intermediate risk according to the International Metastatic RCC Database Consortium (P < .0001), and body mass index of 25 kg/m2 or higher (P = .0021).
“We report, for the first time, that the daily dose of 60 mg cabozantinib at initiation is an independent predictive factor of OS in a multivariate analysis,” the researchers wrote.
“It is interesting to see real-world studies like this to help to widen our understanding of how to utilize drugs like cabozantinib,” commented Simon Crabb, MBBS, PhD, of the University of Southampton (England).
“In general, we would expect a less favorable prognosis in patients with non-clear cell histology, likely in part part due to the underlying biology of the disease,” he added.
Dr. Albiges and colleagues acknowledged that the retrospective design and lack of a prospective safety evaluation were two key limitations of their study. However, the authors maintain that the reported cabozantinib use and exposure rates are indicative of the real-world setting.
This study was sponsored by Ipsen. Several authors disclosed financial relationships with Amgen, Astellas Pharma, AstraZeneca, Bristol-Myers Squibb, Exelixis, Ipsen, and numerous other companies. Dr. Crabb reported having no conflicts of interest related to this work.
SOURCE: Albiges L et al. Eur J Cancer. 2020 Nov 27. doi: 10.1016/j.ejca.2020.09.030.
Starting cabozantinib at 60 mg/day, prior nephrectomy, favorable- or intermediate-risk disease, and body mass index of 25 kg/m2 or higher were all significantly associated with better OS.
These findings were based on data from the early access program of the CABOREAL study and were recently published in the European Journal of Cancer.
“The CABOREAL study describes cabozantinib use in a real-life setting in the largest unselected population to date of patients with mRCC,” lead author Laurence Albiges, MD, PhD, of the Gustave Roussy Cancer Center in Villejuif, France, and colleagues wrote.
The retrospective study enrolled 410 patients with mRCC who were treated with at least one dose of cabozantinib between September 2016 and February 2018. Clinical data were collected from medical records at 26 oncology centers in France.
The researchers evaluated the real-world use of cabozantinib, including duration of therapy, treatment discontinuations, and dose changes. OS and predictive factors of OS were assessed as well.
The median age of study participants was 63.0 years (range, 56.0-70.0 years). Roughly a third of patients (33.4%) received two prior lines of therapy (33.4%), and 41.2% received three or more lines of therapy before cabozantinib. Overall, 85.6% of patients had clear cell histology.
The median duration of cabozantinib treatment was 7.6 months (range, 3.2-15.7 months). The starting dose was 60 mg, 40 mg, and 20 mg in 70.9%, 26.7%, and 2.0% of patients, respectively.
The dose was decreased in 57.0% of patients, 58.7% required a dose modification, and 15.6% required a modified dose schedule. The median average daily dose was 40.0 mg (range, 13.9-60.0 mg).
Adverse events were the main reason for dose modification or treatment interruption. In all, 92.5% of patients had a modification because of an adverse event, and 85.0% had an interruption because of an adverse event.
Upon permanent discontinuation of cabozantinib, more than half of patients (54.4%) received subsequent therapy, including nivolumab (47.8%), axitinib (21.7%), and everolimus (19.0%).
The median OS was 14.4 months (95% confidence interval, 12.4-16.2 months), and the 1-year OS rate was 56.5% (95% CI, 51.5-61.2%).
Factors significantly associated with longer OS included cabozantinib initiation at 60 mg/day (P = .0486), prior nephrectomy (P = .0109), favorable or intermediate risk according to the International Metastatic RCC Database Consortium (P < .0001), and body mass index of 25 kg/m2 or higher (P = .0021).
“We report, for the first time, that the daily dose of 60 mg cabozantinib at initiation is an independent predictive factor of OS in a multivariate analysis,” the researchers wrote.
“It is interesting to see real-world studies like this to help to widen our understanding of how to utilize drugs like cabozantinib,” commented Simon Crabb, MBBS, PhD, of the University of Southampton (England).
“In general, we would expect a less favorable prognosis in patients with non-clear cell histology, likely in part part due to the underlying biology of the disease,” he added.
Dr. Albiges and colleagues acknowledged that the retrospective design and lack of a prospective safety evaluation were two key limitations of their study. However, the authors maintain that the reported cabozantinib use and exposure rates are indicative of the real-world setting.
This study was sponsored by Ipsen. Several authors disclosed financial relationships with Amgen, Astellas Pharma, AstraZeneca, Bristol-Myers Squibb, Exelixis, Ipsen, and numerous other companies. Dr. Crabb reported having no conflicts of interest related to this work.
SOURCE: Albiges L et al. Eur J Cancer. 2020 Nov 27. doi: 10.1016/j.ejca.2020.09.030.
FROM THE EUROPEAN JOURNAL OF CANCER
At-home exercises for 4 common musculoskeletal complaints
The mainstay of treatment for many musculoskeletal (MSK) complaints is physical or occupational therapy. But often an individual’s underlying biomechanical issue is one that can be easily addressed with a home exercise plan, and, in light of the COVID-19 pandemic, patients may wish to avoid in-person physical therapy. This article describes the rationale for, and methods of providing, home exercises for several MSK conditions commonly seen in the primary care setting.
General rehabilitation principles: First things first
With basic MSK complaints, focus on controlling pain and swelling before undertaking restoration of function. Tailor pharmacologic and nonpharmacologic options to the patient’s needs, using first-line modalities such as ice and compression to reduce inflammation, and prescribing scheduled doses of an anti-inflammatory medication to help with both pain and inflammation.
Once pain is sufficiently controlled, have patients begin basic rehabilitation with simple range-of-motion exercises that move the injured region through normal patterns, as tolerated. Later, the patient can progress through more specific exercises to return the injured region to full functional capacity.
Explain to patients that it takes about 7 to 10 days of consistent care to decrease inflammation, but that they should begin prescribed exercises once they are able to tolerate them. Plan a follow-up visit in 2 to 3 weeks to check on the patient’s response to prescribed care.
Which is better, ice or heat?
Ice and heat are both commonly used to treat MSK injuries and pain, although scrutiny of the use of either intervention has increased. Despite the widespread use of these modalities, there is little evidence to support their effect on patient outcomes. The historical consensus has been that ice decreases pain, inflammation, and edema,while heat can facilitate movement in rehabilitation by improving blood flow and decreasing stiffness.1-3 In our practice, we encourage use of both topical modalities as a way to start exercise therapy when pain from the acute injury limits participation. Patients often ask which modality they should use. Ice is generally applied in the acute injury phase (48-72 hours after injury), while heat has been thought to be more beneficial in the chronic stages.
Ccontinue to: When and how to apply ice
When and how to apply ice. Applying an ice pack or a bag of frozen vegetables directly to the affected area will help control pain and swelling. Ice should be applied for 15 to 20 minutes at a time, once an hour. If a patient has sensitivity to cold or if the ice pack is a gel-type, have the patient place a layer (eg, towel) between the ice and skin to avoid injury to the skin. Additional caution should be exercised in patients with peripheral vascular disease, cryoglobulinemia, Raynaud disease, or a history of frostbite at the site.4
An alternative method we sometimes recommend is ice-cup massage. The patient can fill a small paper cup with water and freeze it. The cup is then used to massage the injured area, providing a more active method of icing whereby the cold can penetrate more quickly. Ice-cup massage should be done for 5 to 10 minutes, 3 to 4 times a day.
When and how to apply heat. Heat will help relax and loosen muscles and is a preferred treatment for older injuries, chronic pain, muscle tension, and spasms.5 Because heat can increase blood flow and, likely, inflammation, it should not be used in the acute injury phase. A heating pad or a warm, wet towel can be applied for up to 20 minutes at a time to help relieve pain and tension. Heat is also beneficial before participating in rehab activities as a method of “warming up” a recently injured area.6 However, ice should still be used following activity to prevent any new inflammation.
Anti-inflammatory medications
For an acute injury, nonsteroidal anti-inflammatory drugs (NSAIDs) not only can decrease inflammation and aid in healing but can alleviate pain. We typically start with over-the-counter (OTC) NSAIDs taken on a schedule. A good suggestion is to have the patient take the scheduled NSAID with food for 7 to 10 days or until symptoms subside.
Topical analgesics
Because oral medications can occasionally cause adverse effects or be contraindicated in some patients, topical analgesics can be a good substitute due to their minimal adverse effects. Acceptable topical medications include NSAIDs, lidocaine, menthol, and arnica. Other than prescribed topical NSAIDs, these products can be applied directly to the painful area on an as-needed basis. Often, a topical patch is a nice option to recommend for use during work or school, and a topical cream or ointment can be used at bedtime.
Continue to: Graduated rehabilitation
Graduated rehabilitation
The following 4 common MSK injuries are ones that can benefit from a graduated approach to rehabilitation at home.
Lateral ankle sprain
Lateral ankle sprain, usually resulting from an inversion mechanism, is the most common type of acute ankle sprain seen in primary care and sports medicine settings.7-9 The injury causes lateral ankle pain and swelling, decreased range of motion and strength, and pain with weight-bearing activities.
Treatment and rehabilitation after this type of injury are critical to restoring normal function and increasing the likelihood of returning to pre-injury levels of activity.9,10 Goals for an acute ankle sprain include controlling swelling, regaining full range of motion, increasing muscle strength and power, and improving balance.
Phase 1: Immediately following injury, have the patient protect the injured area with rest, ice, compression, and elevation (RICE). This will help to decrease swelling and pain. Exercises to regain range of motion, such as stretching and doing ankle “ABCs,” should begin within 48 to 72 hours of the initial injury (TABLE 1).9-11
Continue to: Phase 2
Phase 2: Once the patient has achieved full range of motion and pain is controlled, begin the process of regaining strength. The 4-way ankle exercise program (with elastic tubing) is an easy at-home exercise that has been shown to improve strength in plantar flexion, dorsiflexion, eversion, and inversion (TABLE 1).9-11
Phase 3: Once your patient is able to bear full weight with little to no pain, begin a balance program (TABLE 19-11). This is the most frequently neglected component of rehabilitation and the most common reason patients return with chronic ankle pain or repeat ankle injuries. Deficits in postural stability and balance have been reported in unstable ankles following acute ankle sprains,10,12-15 and studies have shown that individuals with poor stability are at a greater risk of injury.13-16
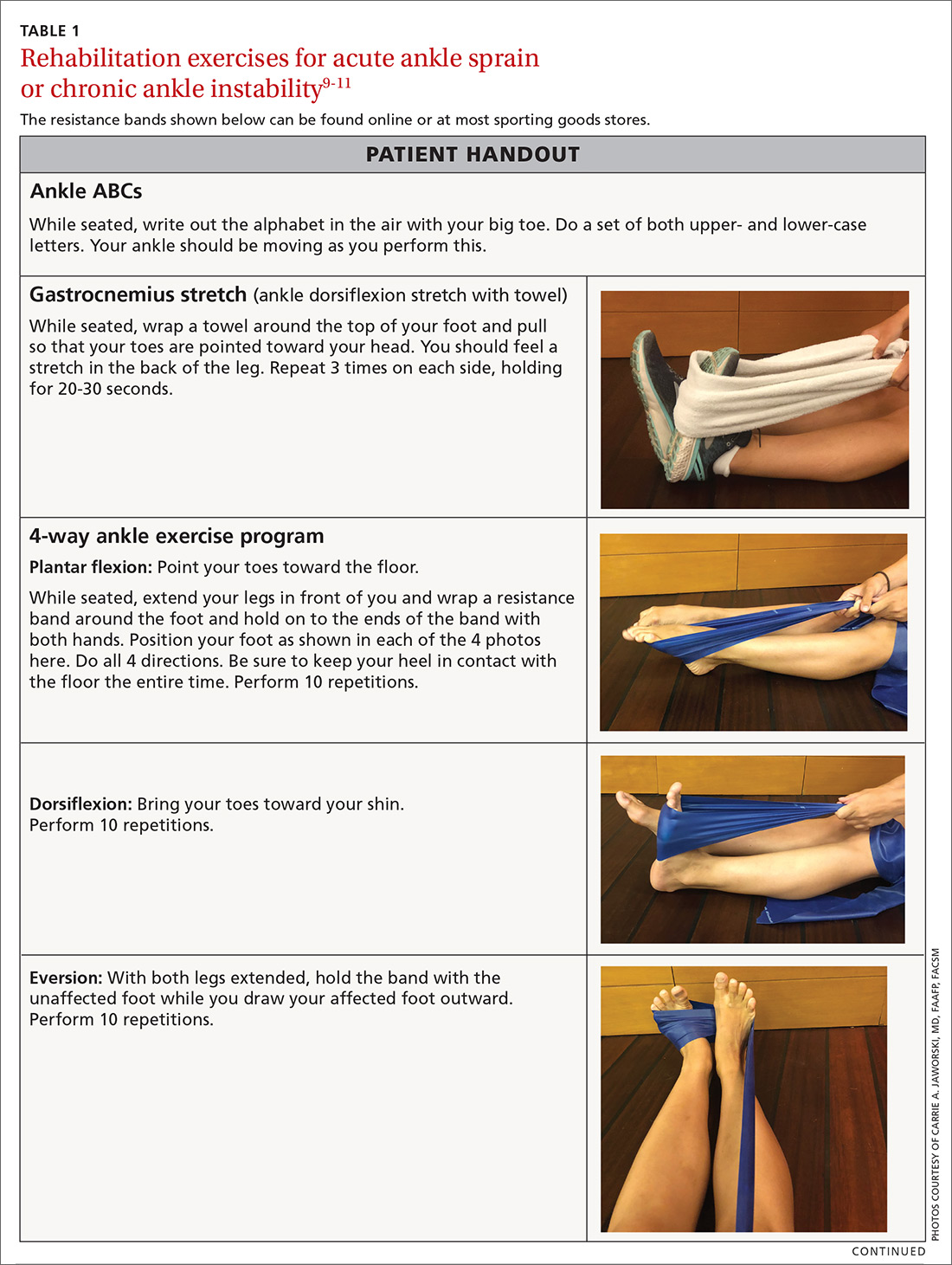
For most lateral ankle sprains, patients can expect time to recovery to range from 2 to 8 weeks. Longer recoveries are associated with more severe injuries or those that involve the syndesmosis.
Plantar fasciitis
Plantar fasciitis (PF) of the foot can be frustrating for a patient due to its chronic nature. Most patients will present with pain in the heel that is aggravated by weight-bearing activities. A conservative management program that focuses on reducing pain and inflammation, reducing tissue stress, and restoring strength and flexibility has been shown to be effective for this type of injury.17,18
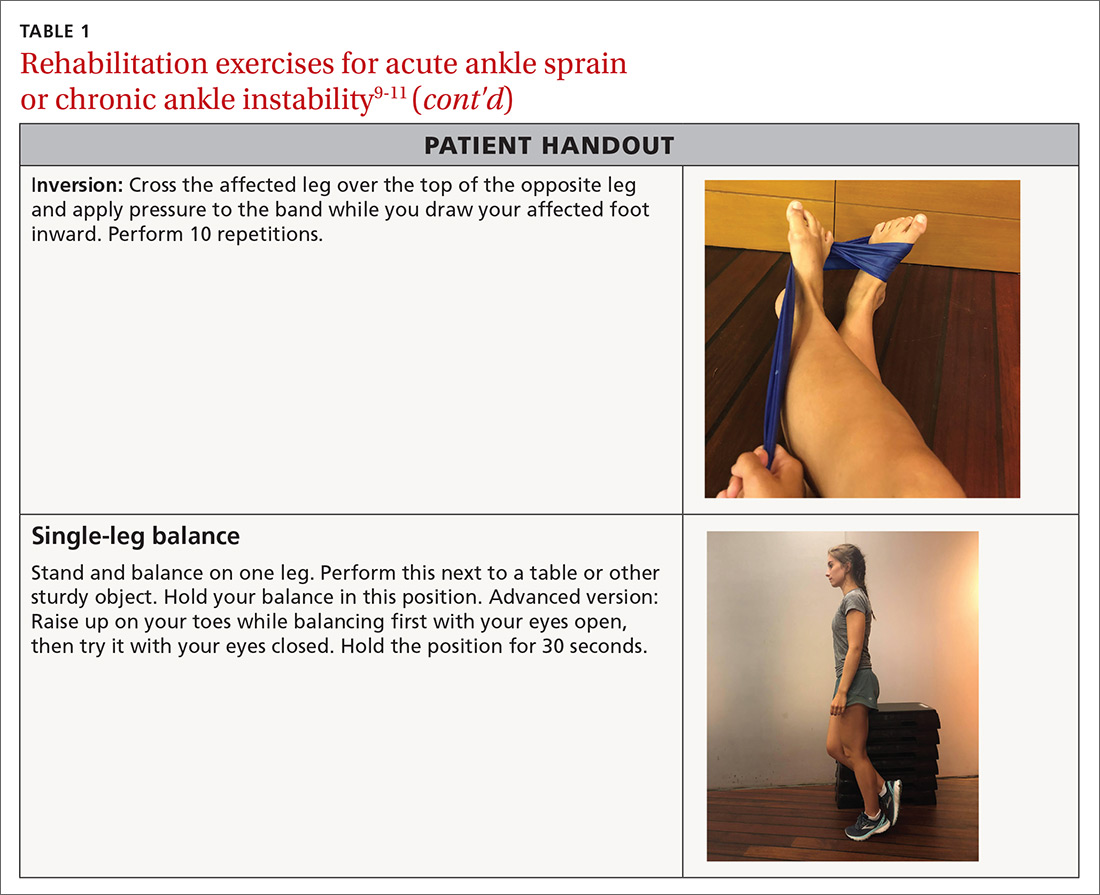
Step 1: Reduce pain and inflammation. Deep-tissue massage and cryotherapy are easy ways to help with pain and inflammation. Deep-tissue massage can be accomplished by rolling the bottom of the foot on a golf or lacrosse ball. A favorite recommendation of ours to reduce inflammation is to use the ice-cup massage, mentioned earlier, for 5 minutes. Or rolling the bottom of the foot on a frozen water bottle will accomplish both tasks at once (TABLE 217,18).
Step 2: Reduce tissue stress. Management tools commonly used to reduce tissue stress are OTC orthotics and night splints. The night splint has been shown to improve symptoms,but patients often stop using it due to discomfort.19 Many kinds of night splints are available, but we have found that the sock variety with a strap to keep the foot in dorsiflexion is best tolerated, and it should be covered by most care plans.
Continue to: Step 3
Step 3: Restore muscle strength and flexibility. Restoring flexibility of the gastrocnemius and soleus is most frequently recommended for treating PF. Strengthening exercises that involve intrinsic and extrinsic muscles of the foot and ankle are also essential.17,18 Helpful exercises include those listed in TABLE 1.9-11 Additionally, an eccentric heel stretch can help to alleviate PF symptoms (TABLE 217,18).
A reasonable timeline for follow-up on newly diagnosed PF is 4 to 6 weeks. While many patients will not have recovered in that time, the goal is to document progress in recovery. If no progress is made, consider other treatment modalities.
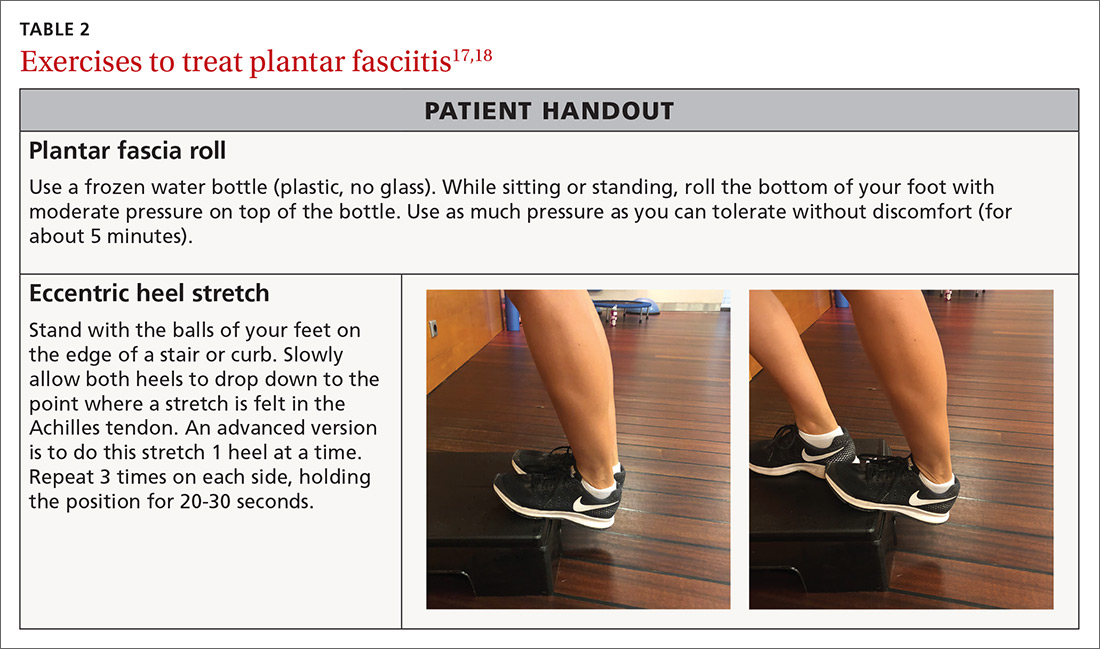
Patellofemoral pain syndrome
Patellofemoral pain syndrome (PFPS) is one of the most common orthopedic complaints, estimated to comprise 7.3% of all orthopedic visits.20 Commonly called “runner’s knee,” PFPS is the leading cause of anterior knee pain in active individuals. Studies suggest a gender bias, with PFPS being diagnosed more frequently in females than in males, particularly between the ages of 10 and 19.20 Often, there is vague anterior knee pain, or pain that worsens with activities such as climbing hills or stairs, or with long sitting or when fatigued.
In general, unbalanced patellar tracking within the trochlear groove likely leads to this pain. Multiple contributory factors have been described; however, evidence increasingly has shown that deficiencies in hip strength may contribute significantly to maltracking of the patella with resultant pain. Specifically, weakness in hip external rotators and abductors is associated with abnormal lower extremity mechanics.21 One randomized controlled trial by Ferber et al found that therapy protocols directed at hip and core strength showed earlier resolution of pain and greater strength when compared with knee protocols alone.22
We routinely talk to patients about how the knee is the “victim” caught between weak hips and/or flat feet. It is prudent to look for both in the office visit. This can be done with one simple maneuver: Ask your patient to do a squat followed by 3 or 4 single-leg squats on each side. This will often reveal dysfunction at the foot/ankle or weakness in the hips/core as demonstrated by pronated feet (along with valgus tracking of the knees inward) or loss of balance upon squatting.
There is general consensus that a nonsurgical approach is the mainstay of treatment for PFPS.23 Pelvic stabilization and hip strengthening are standard components along with treatment protocols of exercises tailored to one’s individual weaknesses.
Numerous types of exercises do not require specialized equipment and can be taught in the office (TABLE 324). Explain to patients that the recovery process may take several months. Monthly follow-up to document progress is essential and helps to ensure compliance with one’s home program.


Continue to : Neck pain
Neck pain
The annual prevalence of nonspecific neck pain ranges from 27% to 48%, with 70% of individuals being afflicted at some time in their lives.25 First rule out any neurologic factors that might suggest cervical disc disease or spinal stenosis. If a patient describes weakness or sensory changes along one or both upper extremities, obtain imaging and consider more formalized therapy with a physical therapist.
In patients without any red flags, investigate possible biomechanical causes. It is essential to review the patient’s work and home habits, particularly in light of COVID-19, to determine if adjustments may be needed. Factors to consider are desk and computer setups at work or home, reading or laptop use in bed, sleep habits, and frequency of cellular phone calls/texting.26 A formal ergonomic assessment of the patient’s workplace may be helpful.
A mainstay in treating mechanical neck pain is alleviating trapezial tightness or spasm. Manipulative therapies such as osteopathic manipulation, massage, and chiropractic care can provide pain relief in the acute setting as well as help with control of chronic symptoms.27 A simple self-care tool is using a tennis ball to massage the trapezial muscles. This can be accomplished by having the patient position the tennis ball along the upper trapezial muscles, holding it in place by leaning against a wall, and initiating self-massage. Another method of self-massage is to put 2 tennis balls in an athletic tube sock and tie off the end, place the sock on the floor, and lie on it in the supine position.
There is also evidence that exercise of any kind can help control neck pain.28,29 The easiest exercises one can offer a patient with neck stiffness, or even mild cervical strains, is self-directed stretching through gentle pressure applied in all 4 directions on the neck. This technique can be repeated hourly both at work and at home (TABLE 4).
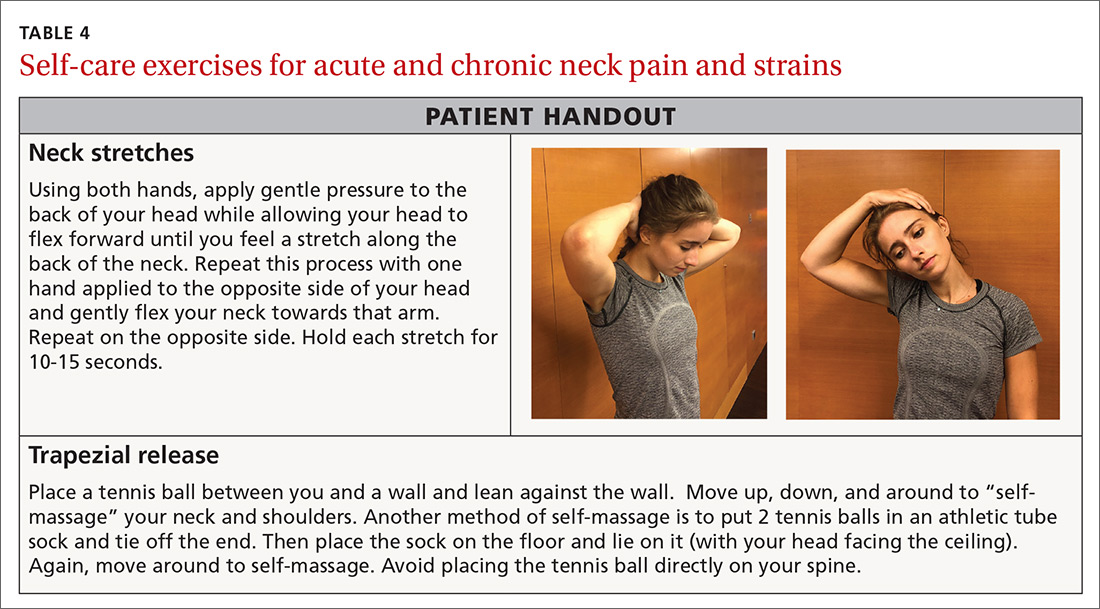
Reminders that can help ensure success
You can use the approaches described here for numerous other MSK conditions in helping patients on the road to recovery.
After the acute phase, advise patients to
• apply heat to the affected area before exercising. This can help bring blood flow to the region and promote ease of movement.
• continue icing the area following rehabilitation exercises in order to control exercise-induced inflammation.
• report any changing symptoms such as worsening pain, numbness, or weakness.
These techniques are one step in the recovery process. A home program can benefit the patient either alone or in combination with more advanced techniques that are best accomplished under the watchful eye of a physical or occupational therapist.
CORRESPONDENCE
Carrie A. Jaworski, MD, FAAFP, FACSM, 2180 Pfingsten Road, Suite 3100, Glenview, IL 60026; [email protected]
1. Hubbard TJ, Aronson SL, Denegar CR. Does cryotherapy hasten return to participation? A systematic review. J Athl Train. 2004;39:88-94.
2. Ho SS, Coel MN, Kagawa R, et al. The effects of ice on blood flow and bone metabolism in knees. Am J Sports Med. 1994;22:537-540.
3. Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65.
4. Bleakley CM, O’Connor S, Tully MA, et al. The PRICE study (Protection Rest Ice Compression Elevation): design of a randomised controlled trial comparing standard versus cryokinetic ice applications in the management of acute ankle sprain. BMC Musculoskelet Disord. 2007;8:125.
5. Mayer JM, Ralph L, Look M, et al. Treating acute low back pain with continuous low-level heat wrap therapy and/or exercise: a randomized controlled trial. Spine J. 2005;5:395-403.
6. Cetin N, Aytar A, Atalay A, et al. Comparing hot pack, short-wave diathermy, ultrasound, and TENS on isokinetic strength, pain, and functional status of women with osteoarthritic knees: a single-blind, randomized, controlled trial. Am J Phys Med Rehabil. 2008;87:443-451.
7. Waterman BR, Owens BD, Davey S, et al. The epidemiology of ankle sprains in the United States. J Bone Joint Surg Am. 2010;92:2279-2284.
8. Fong DT, Hong Y, Chan LK, et al. A systematic review on ankle injury and ankle sprain in sports. Sports Med. 2007;37:73-94.
9. Kerkhoffs GM, Rowe BH, Assendelft WJ, et al. Immobilisation and functional treatment for acute lateral ankle ligament injuries in adults. Cochrane Database Syst Rev. 2002(3):CD003762.
10. Mattacola CG, Dwyer MK. Rehabilitation of the ankle after acute sprain or chronic instability. J Ath Train. 2002;37:413-429.
11. Hü bscher M, Zech A, Pfeifer K, et al. Neuromuscular training for sports injury prevention: a systematic review. Med Sci Sports Exerc. 2010;42:413-421.
12. Emery CA, Meeuwisse WH. The effectiveness of a neuromuscular prevention strategy to reduce injuries in youth soccer: a cluster-randomised controlled trial. Br J Sports Med. 2010;44:555-562.
13. Tiemstra JD. Update on acute ankle sprains. Am Fam Physician. 2012;85:1170-1176.
14. Beynnon BD, Murphy DF, Alosa DM. Predictive factors for lateral ankle sprains: a literature review. J Ath Train. 2002;37:376-380.
15. Schiftan GS, Ross LA, Hahne AJ. The effectiveness of proprioceptive training in preventing ankle sprains in sporting populations: a systematic review and meta-analysis. J Sci Med Sport. 2015;18:238–244.
16. Hupperets MD, Verhagen EA, van Mechelen W. Effect of unsupervised home based proprioceptive training on recurrences of ankle sprain: randomised controlled trial. BMJ. 2009;339:b2684
17. Thompson JV, Saini SS, Reb CW, et al. Diagnosis and management of plantar fasciitis. J Am Osteopath Assoc. 2014;114:900-906.
18. DiGiovanni BF, Nawoczenski DA, Malay DP, et al. Plantar fascia-specific stretching exercise improves outcomes in patients with chronic plantar fasciitis. A prospective clinical trial with two-year follow-up. J Bone Joint Surg Am. 2006;88:1775-1781.

19. Lee SY, McKeon P, Hertel J. Does the use of orthoses improve self-reported pain and function measures in patients with plantar fasciitis? A meta-analysis. Phys Ther Sport. 2009;10:12-18.
20. Glaviano NR, Key M, Hart JM, et al. Demographic and epidemiological trends in patellofemoral pain. J Sports Phys Ther. 2015;10: 281-290.
21. Louden JK. Biomechanics and pathomechanics of the patellofemoral joint. Int J Sports Phys Ther. 2016;11: 820-830.
22. Ferber R, Bolgla L, Earl-Boehm JE, et al. Strengthening of hip and core versus knee muscles for the treatment of patellofemoral pain: a multicenter randomized controlled trial. J Ath Train. 2015;50: 366-377.
23. Collins NJ, Bisset LM, Crossley KM, et al. Efficacy of nonsurgical interventions for anterior knee pain: systematic review and meta-analysis of randomized trials. Sports Med. 2013;41:31-49.
24. Bolgla LA. Hip strength and kinematics in patellofemoral syndrome. In: Brotzman SB, Manske RC eds. Clinical Orthopaedic Rehabilitation. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2011:273-274.
25. Hogg-Johnson S, van der Velde G, Carroll LJ, et al. The burden and determinants of neck pain in the general population: results of the Bone and Joint Decade 2000-2010 Task Force on Neck Pain and Its Associated Disorders. Spine. 2008;33(suppl 4):S39-S51.
26. Larsson B, Søgaard K, Rosendal L. Work related neck-shoulder pain: a review on magnitude, risk factors, biochemical characteristics, clinical picture and preventive interventions. Best Pract Res Clin Rheumatol. 2007; 21:447-463.
27. Giles LG, Muller R. Chronic spinal pain: a randomized clinical trial comparing medication, acupuncture, and spinal manipulation. Spine. 2003;28:1490-1502.
28. Bronfort G, Evans R, Anderson A, et al. Spinal manipulation, medication, or home exercise with advice for acute and subacute neck pain: a randomized trial. Ann Intern Med. 2012;156:1-10.
29. Evans R, Bronfort G, Bittell S, et al. A pilot study for a randomized clinical trial assessing chiropractic care, medical care, and self-care education for acute and subacute neck pain patients. J Manipulative Physiol Ther. 2003;26:403-411.
The mainstay of treatment for many musculoskeletal (MSK) complaints is physical or occupational therapy. But often an individual’s underlying biomechanical issue is one that can be easily addressed with a home exercise plan, and, in light of the COVID-19 pandemic, patients may wish to avoid in-person physical therapy. This article describes the rationale for, and methods of providing, home exercises for several MSK conditions commonly seen in the primary care setting.
General rehabilitation principles: First things first
With basic MSK complaints, focus on controlling pain and swelling before undertaking restoration of function. Tailor pharmacologic and nonpharmacologic options to the patient’s needs, using first-line modalities such as ice and compression to reduce inflammation, and prescribing scheduled doses of an anti-inflammatory medication to help with both pain and inflammation.
Once pain is sufficiently controlled, have patients begin basic rehabilitation with simple range-of-motion exercises that move the injured region through normal patterns, as tolerated. Later, the patient can progress through more specific exercises to return the injured region to full functional capacity.
Explain to patients that it takes about 7 to 10 days of consistent care to decrease inflammation, but that they should begin prescribed exercises once they are able to tolerate them. Plan a follow-up visit in 2 to 3 weeks to check on the patient’s response to prescribed care.
Which is better, ice or heat?
Ice and heat are both commonly used to treat MSK injuries and pain, although scrutiny of the use of either intervention has increased. Despite the widespread use of these modalities, there is little evidence to support their effect on patient outcomes. The historical consensus has been that ice decreases pain, inflammation, and edema,while heat can facilitate movement in rehabilitation by improving blood flow and decreasing stiffness.1-3 In our practice, we encourage use of both topical modalities as a way to start exercise therapy when pain from the acute injury limits participation. Patients often ask which modality they should use. Ice is generally applied in the acute injury phase (48-72 hours after injury), while heat has been thought to be more beneficial in the chronic stages.
Ccontinue to: When and how to apply ice
When and how to apply ice. Applying an ice pack or a bag of frozen vegetables directly to the affected area will help control pain and swelling. Ice should be applied for 15 to 20 minutes at a time, once an hour. If a patient has sensitivity to cold or if the ice pack is a gel-type, have the patient place a layer (eg, towel) between the ice and skin to avoid injury to the skin. Additional caution should be exercised in patients with peripheral vascular disease, cryoglobulinemia, Raynaud disease, or a history of frostbite at the site.4
An alternative method we sometimes recommend is ice-cup massage. The patient can fill a small paper cup with water and freeze it. The cup is then used to massage the injured area, providing a more active method of icing whereby the cold can penetrate more quickly. Ice-cup massage should be done for 5 to 10 minutes, 3 to 4 times a day.
When and how to apply heat. Heat will help relax and loosen muscles and is a preferred treatment for older injuries, chronic pain, muscle tension, and spasms.5 Because heat can increase blood flow and, likely, inflammation, it should not be used in the acute injury phase. A heating pad or a warm, wet towel can be applied for up to 20 minutes at a time to help relieve pain and tension. Heat is also beneficial before participating in rehab activities as a method of “warming up” a recently injured area.6 However, ice should still be used following activity to prevent any new inflammation.
Anti-inflammatory medications
For an acute injury, nonsteroidal anti-inflammatory drugs (NSAIDs) not only can decrease inflammation and aid in healing but can alleviate pain. We typically start with over-the-counter (OTC) NSAIDs taken on a schedule. A good suggestion is to have the patient take the scheduled NSAID with food for 7 to 10 days or until symptoms subside.
Topical analgesics
Because oral medications can occasionally cause adverse effects or be contraindicated in some patients, topical analgesics can be a good substitute due to their minimal adverse effects. Acceptable topical medications include NSAIDs, lidocaine, menthol, and arnica. Other than prescribed topical NSAIDs, these products can be applied directly to the painful area on an as-needed basis. Often, a topical patch is a nice option to recommend for use during work or school, and a topical cream or ointment can be used at bedtime.
Continue to: Graduated rehabilitation
Graduated rehabilitation
The following 4 common MSK injuries are ones that can benefit from a graduated approach to rehabilitation at home.
Lateral ankle sprain
Lateral ankle sprain, usually resulting from an inversion mechanism, is the most common type of acute ankle sprain seen in primary care and sports medicine settings.7-9 The injury causes lateral ankle pain and swelling, decreased range of motion and strength, and pain with weight-bearing activities.
Treatment and rehabilitation after this type of injury are critical to restoring normal function and increasing the likelihood of returning to pre-injury levels of activity.9,10 Goals for an acute ankle sprain include controlling swelling, regaining full range of motion, increasing muscle strength and power, and improving balance.
Phase 1: Immediately following injury, have the patient protect the injured area with rest, ice, compression, and elevation (RICE). This will help to decrease swelling and pain. Exercises to regain range of motion, such as stretching and doing ankle “ABCs,” should begin within 48 to 72 hours of the initial injury (TABLE 1).9-11
Continue to: Phase 2
Phase 2: Once the patient has achieved full range of motion and pain is controlled, begin the process of regaining strength. The 4-way ankle exercise program (with elastic tubing) is an easy at-home exercise that has been shown to improve strength in plantar flexion, dorsiflexion, eversion, and inversion (TABLE 1).9-11
Phase 3: Once your patient is able to bear full weight with little to no pain, begin a balance program (TABLE 19-11). This is the most frequently neglected component of rehabilitation and the most common reason patients return with chronic ankle pain or repeat ankle injuries. Deficits in postural stability and balance have been reported in unstable ankles following acute ankle sprains,10,12-15 and studies have shown that individuals with poor stability are at a greater risk of injury.13-16

For most lateral ankle sprains, patients can expect time to recovery to range from 2 to 8 weeks. Longer recoveries are associated with more severe injuries or those that involve the syndesmosis.
Plantar fasciitis
Plantar fasciitis (PF) of the foot can be frustrating for a patient due to its chronic nature. Most patients will present with pain in the heel that is aggravated by weight-bearing activities. A conservative management program that focuses on reducing pain and inflammation, reducing tissue stress, and restoring strength and flexibility has been shown to be effective for this type of injury.17,18

Step 1: Reduce pain and inflammation. Deep-tissue massage and cryotherapy are easy ways to help with pain and inflammation. Deep-tissue massage can be accomplished by rolling the bottom of the foot on a golf or lacrosse ball. A favorite recommendation of ours to reduce inflammation is to use the ice-cup massage, mentioned earlier, for 5 minutes. Or rolling the bottom of the foot on a frozen water bottle will accomplish both tasks at once (TABLE 217,18).
Step 2: Reduce tissue stress. Management tools commonly used to reduce tissue stress are OTC orthotics and night splints. The night splint has been shown to improve symptoms,but patients often stop using it due to discomfort.19 Many kinds of night splints are available, but we have found that the sock variety with a strap to keep the foot in dorsiflexion is best tolerated, and it should be covered by most care plans.
Continue to: Step 3
Step 3: Restore muscle strength and flexibility. Restoring flexibility of the gastrocnemius and soleus is most frequently recommended for treating PF. Strengthening exercises that involve intrinsic and extrinsic muscles of the foot and ankle are also essential.17,18 Helpful exercises include those listed in TABLE 1.9-11 Additionally, an eccentric heel stretch can help to alleviate PF symptoms (TABLE 217,18).
A reasonable timeline for follow-up on newly diagnosed PF is 4 to 6 weeks. While many patients will not have recovered in that time, the goal is to document progress in recovery. If no progress is made, consider other treatment modalities.

Patellofemoral pain syndrome
Patellofemoral pain syndrome (PFPS) is one of the most common orthopedic complaints, estimated to comprise 7.3% of all orthopedic visits.20 Commonly called “runner’s knee,” PFPS is the leading cause of anterior knee pain in active individuals. Studies suggest a gender bias, with PFPS being diagnosed more frequently in females than in males, particularly between the ages of 10 and 19.20 Often, there is vague anterior knee pain, or pain that worsens with activities such as climbing hills or stairs, or with long sitting or when fatigued.
In general, unbalanced patellar tracking within the trochlear groove likely leads to this pain. Multiple contributory factors have been described; however, evidence increasingly has shown that deficiencies in hip strength may contribute significantly to maltracking of the patella with resultant pain. Specifically, weakness in hip external rotators and abductors is associated with abnormal lower extremity mechanics.21 One randomized controlled trial by Ferber et al found that therapy protocols directed at hip and core strength showed earlier resolution of pain and greater strength when compared with knee protocols alone.22
We routinely talk to patients about how the knee is the “victim” caught between weak hips and/or flat feet. It is prudent to look for both in the office visit. This can be done with one simple maneuver: Ask your patient to do a squat followed by 3 or 4 single-leg squats on each side. This will often reveal dysfunction at the foot/ankle or weakness in the hips/core as demonstrated by pronated feet (along with valgus tracking of the knees inward) or loss of balance upon squatting.
There is general consensus that a nonsurgical approach is the mainstay of treatment for PFPS.23 Pelvic stabilization and hip strengthening are standard components along with treatment protocols of exercises tailored to one’s individual weaknesses.
Numerous types of exercises do not require specialized equipment and can be taught in the office (TABLE 324). Explain to patients that the recovery process may take several months. Monthly follow-up to document progress is essential and helps to ensure compliance with one’s home program.


Continue to : Neck pain
Neck pain
The annual prevalence of nonspecific neck pain ranges from 27% to 48%, with 70% of individuals being afflicted at some time in their lives.25 First rule out any neurologic factors that might suggest cervical disc disease or spinal stenosis. If a patient describes weakness or sensory changes along one or both upper extremities, obtain imaging and consider more formalized therapy with a physical therapist.
In patients without any red flags, investigate possible biomechanical causes. It is essential to review the patient’s work and home habits, particularly in light of COVID-19, to determine if adjustments may be needed. Factors to consider are desk and computer setups at work or home, reading or laptop use in bed, sleep habits, and frequency of cellular phone calls/texting.26 A formal ergonomic assessment of the patient’s workplace may be helpful.
A mainstay in treating mechanical neck pain is alleviating trapezial tightness or spasm. Manipulative therapies such as osteopathic manipulation, massage, and chiropractic care can provide pain relief in the acute setting as well as help with control of chronic symptoms.27 A simple self-care tool is using a tennis ball to massage the trapezial muscles. This can be accomplished by having the patient position the tennis ball along the upper trapezial muscles, holding it in place by leaning against a wall, and initiating self-massage. Another method of self-massage is to put 2 tennis balls in an athletic tube sock and tie off the end, place the sock on the floor, and lie on it in the supine position.
There is also evidence that exercise of any kind can help control neck pain.28,29 The easiest exercises one can offer a patient with neck stiffness, or even mild cervical strains, is self-directed stretching through gentle pressure applied in all 4 directions on the neck. This technique can be repeated hourly both at work and at home (TABLE 4).

Reminders that can help ensure success
You can use the approaches described here for numerous other MSK conditions in helping patients on the road to recovery.
After the acute phase, advise patients to
• apply heat to the affected area before exercising. This can help bring blood flow to the region and promote ease of movement.
• continue icing the area following rehabilitation exercises in order to control exercise-induced inflammation.
• report any changing symptoms such as worsening pain, numbness, or weakness.
These techniques are one step in the recovery process. A home program can benefit the patient either alone or in combination with more advanced techniques that are best accomplished under the watchful eye of a physical or occupational therapist.
CORRESPONDENCE
Carrie A. Jaworski, MD, FAAFP, FACSM, 2180 Pfingsten Road, Suite 3100, Glenview, IL 60026; [email protected]
The mainstay of treatment for many musculoskeletal (MSK) complaints is physical or occupational therapy. But often an individual’s underlying biomechanical issue is one that can be easily addressed with a home exercise plan, and, in light of the COVID-19 pandemic, patients may wish to avoid in-person physical therapy. This article describes the rationale for, and methods of providing, home exercises for several MSK conditions commonly seen in the primary care setting.
General rehabilitation principles: First things first
With basic MSK complaints, focus on controlling pain and swelling before undertaking restoration of function. Tailor pharmacologic and nonpharmacologic options to the patient’s needs, using first-line modalities such as ice and compression to reduce inflammation, and prescribing scheduled doses of an anti-inflammatory medication to help with both pain and inflammation.
Once pain is sufficiently controlled, have patients begin basic rehabilitation with simple range-of-motion exercises that move the injured region through normal patterns, as tolerated. Later, the patient can progress through more specific exercises to return the injured region to full functional capacity.
Explain to patients that it takes about 7 to 10 days of consistent care to decrease inflammation, but that they should begin prescribed exercises once they are able to tolerate them. Plan a follow-up visit in 2 to 3 weeks to check on the patient’s response to prescribed care.
Which is better, ice or heat?
Ice and heat are both commonly used to treat MSK injuries and pain, although scrutiny of the use of either intervention has increased. Despite the widespread use of these modalities, there is little evidence to support their effect on patient outcomes. The historical consensus has been that ice decreases pain, inflammation, and edema,while heat can facilitate movement in rehabilitation by improving blood flow and decreasing stiffness.1-3 In our practice, we encourage use of both topical modalities as a way to start exercise therapy when pain from the acute injury limits participation. Patients often ask which modality they should use. Ice is generally applied in the acute injury phase (48-72 hours after injury), while heat has been thought to be more beneficial in the chronic stages.
Ccontinue to: When and how to apply ice
When and how to apply ice. Applying an ice pack or a bag of frozen vegetables directly to the affected area will help control pain and swelling. Ice should be applied for 15 to 20 minutes at a time, once an hour. If a patient has sensitivity to cold or if the ice pack is a gel-type, have the patient place a layer (eg, towel) between the ice and skin to avoid injury to the skin. Additional caution should be exercised in patients with peripheral vascular disease, cryoglobulinemia, Raynaud disease, or a history of frostbite at the site.4
An alternative method we sometimes recommend is ice-cup massage. The patient can fill a small paper cup with water and freeze it. The cup is then used to massage the injured area, providing a more active method of icing whereby the cold can penetrate more quickly. Ice-cup massage should be done for 5 to 10 minutes, 3 to 4 times a day.
When and how to apply heat. Heat will help relax and loosen muscles and is a preferred treatment for older injuries, chronic pain, muscle tension, and spasms.5 Because heat can increase blood flow and, likely, inflammation, it should not be used in the acute injury phase. A heating pad or a warm, wet towel can be applied for up to 20 minutes at a time to help relieve pain and tension. Heat is also beneficial before participating in rehab activities as a method of “warming up” a recently injured area.6 However, ice should still be used following activity to prevent any new inflammation.
Anti-inflammatory medications
For an acute injury, nonsteroidal anti-inflammatory drugs (NSAIDs) not only can decrease inflammation and aid in healing but can alleviate pain. We typically start with over-the-counter (OTC) NSAIDs taken on a schedule. A good suggestion is to have the patient take the scheduled NSAID with food for 7 to 10 days or until symptoms subside.
Topical analgesics
Because oral medications can occasionally cause adverse effects or be contraindicated in some patients, topical analgesics can be a good substitute due to their minimal adverse effects. Acceptable topical medications include NSAIDs, lidocaine, menthol, and arnica. Other than prescribed topical NSAIDs, these products can be applied directly to the painful area on an as-needed basis. Often, a topical patch is a nice option to recommend for use during work or school, and a topical cream or ointment can be used at bedtime.
Continue to: Graduated rehabilitation
Graduated rehabilitation
The following 4 common MSK injuries are ones that can benefit from a graduated approach to rehabilitation at home.
Lateral ankle sprain
Lateral ankle sprain, usually resulting from an inversion mechanism, is the most common type of acute ankle sprain seen in primary care and sports medicine settings.7-9 The injury causes lateral ankle pain and swelling, decreased range of motion and strength, and pain with weight-bearing activities.
Treatment and rehabilitation after this type of injury are critical to restoring normal function and increasing the likelihood of returning to pre-injury levels of activity.9,10 Goals for an acute ankle sprain include controlling swelling, regaining full range of motion, increasing muscle strength and power, and improving balance.
Phase 1: Immediately following injury, have the patient protect the injured area with rest, ice, compression, and elevation (RICE). This will help to decrease swelling and pain. Exercises to regain range of motion, such as stretching and doing ankle “ABCs,” should begin within 48 to 72 hours of the initial injury (TABLE 1).9-11
Continue to: Phase 2
Phase 2: Once the patient has achieved full range of motion and pain is controlled, begin the process of regaining strength. The 4-way ankle exercise program (with elastic tubing) is an easy at-home exercise that has been shown to improve strength in plantar flexion, dorsiflexion, eversion, and inversion (TABLE 1).9-11
Phase 3: Once your patient is able to bear full weight with little to no pain, begin a balance program (TABLE 19-11). This is the most frequently neglected component of rehabilitation and the most common reason patients return with chronic ankle pain or repeat ankle injuries. Deficits in postural stability and balance have been reported in unstable ankles following acute ankle sprains,10,12-15 and studies have shown that individuals with poor stability are at a greater risk of injury.13-16

For most lateral ankle sprains, patients can expect time to recovery to range from 2 to 8 weeks. Longer recoveries are associated with more severe injuries or those that involve the syndesmosis.
Plantar fasciitis
Plantar fasciitis (PF) of the foot can be frustrating for a patient due to its chronic nature. Most patients will present with pain in the heel that is aggravated by weight-bearing activities. A conservative management program that focuses on reducing pain and inflammation, reducing tissue stress, and restoring strength and flexibility has been shown to be effective for this type of injury.17,18

Step 1: Reduce pain and inflammation. Deep-tissue massage and cryotherapy are easy ways to help with pain and inflammation. Deep-tissue massage can be accomplished by rolling the bottom of the foot on a golf or lacrosse ball. A favorite recommendation of ours to reduce inflammation is to use the ice-cup massage, mentioned earlier, for 5 minutes. Or rolling the bottom of the foot on a frozen water bottle will accomplish both tasks at once (TABLE 217,18).
Step 2: Reduce tissue stress. Management tools commonly used to reduce tissue stress are OTC orthotics and night splints. The night splint has been shown to improve symptoms,but patients often stop using it due to discomfort.19 Many kinds of night splints are available, but we have found that the sock variety with a strap to keep the foot in dorsiflexion is best tolerated, and it should be covered by most care plans.
Continue to: Step 3
Step 3: Restore muscle strength and flexibility. Restoring flexibility of the gastrocnemius and soleus is most frequently recommended for treating PF. Strengthening exercises that involve intrinsic and extrinsic muscles of the foot and ankle are also essential.17,18 Helpful exercises include those listed in TABLE 1.9-11 Additionally, an eccentric heel stretch can help to alleviate PF symptoms (TABLE 217,18).
A reasonable timeline for follow-up on newly diagnosed PF is 4 to 6 weeks. While many patients will not have recovered in that time, the goal is to document progress in recovery. If no progress is made, consider other treatment modalities.

Patellofemoral pain syndrome
Patellofemoral pain syndrome (PFPS) is one of the most common orthopedic complaints, estimated to comprise 7.3% of all orthopedic visits.20 Commonly called “runner’s knee,” PFPS is the leading cause of anterior knee pain in active individuals. Studies suggest a gender bias, with PFPS being diagnosed more frequently in females than in males, particularly between the ages of 10 and 19.20 Often, there is vague anterior knee pain, or pain that worsens with activities such as climbing hills or stairs, or with long sitting or when fatigued.
In general, unbalanced patellar tracking within the trochlear groove likely leads to this pain. Multiple contributory factors have been described; however, evidence increasingly has shown that deficiencies in hip strength may contribute significantly to maltracking of the patella with resultant pain. Specifically, weakness in hip external rotators and abductors is associated with abnormal lower extremity mechanics.21 One randomized controlled trial by Ferber et al found that therapy protocols directed at hip and core strength showed earlier resolution of pain and greater strength when compared with knee protocols alone.22
We routinely talk to patients about how the knee is the “victim” caught between weak hips and/or flat feet. It is prudent to look for both in the office visit. This can be done with one simple maneuver: Ask your patient to do a squat followed by 3 or 4 single-leg squats on each side. This will often reveal dysfunction at the foot/ankle or weakness in the hips/core as demonstrated by pronated feet (along with valgus tracking of the knees inward) or loss of balance upon squatting.
There is general consensus that a nonsurgical approach is the mainstay of treatment for PFPS.23 Pelvic stabilization and hip strengthening are standard components along with treatment protocols of exercises tailored to one’s individual weaknesses.
Numerous types of exercises do not require specialized equipment and can be taught in the office (TABLE 324). Explain to patients that the recovery process may take several months. Monthly follow-up to document progress is essential and helps to ensure compliance with one’s home program.


Continue to : Neck pain
Neck pain
The annual prevalence of nonspecific neck pain ranges from 27% to 48%, with 70% of individuals being afflicted at some time in their lives.25 First rule out any neurologic factors that might suggest cervical disc disease or spinal stenosis. If a patient describes weakness or sensory changes along one or both upper extremities, obtain imaging and consider more formalized therapy with a physical therapist.
In patients without any red flags, investigate possible biomechanical causes. It is essential to review the patient’s work and home habits, particularly in light of COVID-19, to determine if adjustments may be needed. Factors to consider are desk and computer setups at work or home, reading or laptop use in bed, sleep habits, and frequency of cellular phone calls/texting.26 A formal ergonomic assessment of the patient’s workplace may be helpful.
A mainstay in treating mechanical neck pain is alleviating trapezial tightness or spasm. Manipulative therapies such as osteopathic manipulation, massage, and chiropractic care can provide pain relief in the acute setting as well as help with control of chronic symptoms.27 A simple self-care tool is using a tennis ball to massage the trapezial muscles. This can be accomplished by having the patient position the tennis ball along the upper trapezial muscles, holding it in place by leaning against a wall, and initiating self-massage. Another method of self-massage is to put 2 tennis balls in an athletic tube sock and tie off the end, place the sock on the floor, and lie on it in the supine position.
There is also evidence that exercise of any kind can help control neck pain.28,29 The easiest exercises one can offer a patient with neck stiffness, or even mild cervical strains, is self-directed stretching through gentle pressure applied in all 4 directions on the neck. This technique can be repeated hourly both at work and at home (TABLE 4).

Reminders that can help ensure success
You can use the approaches described here for numerous other MSK conditions in helping patients on the road to recovery.
After the acute phase, advise patients to
• apply heat to the affected area before exercising. This can help bring blood flow to the region and promote ease of movement.
• continue icing the area following rehabilitation exercises in order to control exercise-induced inflammation.
• report any changing symptoms such as worsening pain, numbness, or weakness.
These techniques are one step in the recovery process. A home program can benefit the patient either alone or in combination with more advanced techniques that are best accomplished under the watchful eye of a physical or occupational therapist.
CORRESPONDENCE
Carrie A. Jaworski, MD, FAAFP, FACSM, 2180 Pfingsten Road, Suite 3100, Glenview, IL 60026; [email protected]
1. Hubbard TJ, Aronson SL, Denegar CR. Does cryotherapy hasten return to participation? A systematic review. J Athl Train. 2004;39:88-94.
2. Ho SS, Coel MN, Kagawa R, et al. The effects of ice on blood flow and bone metabolism in knees. Am J Sports Med. 1994;22:537-540.
3. Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65.
4. Bleakley CM, O’Connor S, Tully MA, et al. The PRICE study (Protection Rest Ice Compression Elevation): design of a randomised controlled trial comparing standard versus cryokinetic ice applications in the management of acute ankle sprain. BMC Musculoskelet Disord. 2007;8:125.
5. Mayer JM, Ralph L, Look M, et al. Treating acute low back pain with continuous low-level heat wrap therapy and/or exercise: a randomized controlled trial. Spine J. 2005;5:395-403.
6. Cetin N, Aytar A, Atalay A, et al. Comparing hot pack, short-wave diathermy, ultrasound, and TENS on isokinetic strength, pain, and functional status of women with osteoarthritic knees: a single-blind, randomized, controlled trial. Am J Phys Med Rehabil. 2008;87:443-451.
7. Waterman BR, Owens BD, Davey S, et al. The epidemiology of ankle sprains in the United States. J Bone Joint Surg Am. 2010;92:2279-2284.
8. Fong DT, Hong Y, Chan LK, et al. A systematic review on ankle injury and ankle sprain in sports. Sports Med. 2007;37:73-94.
9. Kerkhoffs GM, Rowe BH, Assendelft WJ, et al. Immobilisation and functional treatment for acute lateral ankle ligament injuries in adults. Cochrane Database Syst Rev. 2002(3):CD003762.
10. Mattacola CG, Dwyer MK. Rehabilitation of the ankle after acute sprain or chronic instability. J Ath Train. 2002;37:413-429.
11. Hü bscher M, Zech A, Pfeifer K, et al. Neuromuscular training for sports injury prevention: a systematic review. Med Sci Sports Exerc. 2010;42:413-421.
12. Emery CA, Meeuwisse WH. The effectiveness of a neuromuscular prevention strategy to reduce injuries in youth soccer: a cluster-randomised controlled trial. Br J Sports Med. 2010;44:555-562.
13. Tiemstra JD. Update on acute ankle sprains. Am Fam Physician. 2012;85:1170-1176.
14. Beynnon BD, Murphy DF, Alosa DM. Predictive factors for lateral ankle sprains: a literature review. J Ath Train. 2002;37:376-380.
15. Schiftan GS, Ross LA, Hahne AJ. The effectiveness of proprioceptive training in preventing ankle sprains in sporting populations: a systematic review and meta-analysis. J Sci Med Sport. 2015;18:238–244.
16. Hupperets MD, Verhagen EA, van Mechelen W. Effect of unsupervised home based proprioceptive training on recurrences of ankle sprain: randomised controlled trial. BMJ. 2009;339:b2684
17. Thompson JV, Saini SS, Reb CW, et al. Diagnosis and management of plantar fasciitis. J Am Osteopath Assoc. 2014;114:900-906.
18. DiGiovanni BF, Nawoczenski DA, Malay DP, et al. Plantar fascia-specific stretching exercise improves outcomes in patients with chronic plantar fasciitis. A prospective clinical trial with two-year follow-up. J Bone Joint Surg Am. 2006;88:1775-1781.

19. Lee SY, McKeon P, Hertel J. Does the use of orthoses improve self-reported pain and function measures in patients with plantar fasciitis? A meta-analysis. Phys Ther Sport. 2009;10:12-18.
20. Glaviano NR, Key M, Hart JM, et al. Demographic and epidemiological trends in patellofemoral pain. J Sports Phys Ther. 2015;10: 281-290.
21. Louden JK. Biomechanics and pathomechanics of the patellofemoral joint. Int J Sports Phys Ther. 2016;11: 820-830.
22. Ferber R, Bolgla L, Earl-Boehm JE, et al. Strengthening of hip and core versus knee muscles for the treatment of patellofemoral pain: a multicenter randomized controlled trial. J Ath Train. 2015;50: 366-377.
23. Collins NJ, Bisset LM, Crossley KM, et al. Efficacy of nonsurgical interventions for anterior knee pain: systematic review and meta-analysis of randomized trials. Sports Med. 2013;41:31-49.
24. Bolgla LA. Hip strength and kinematics in patellofemoral syndrome. In: Brotzman SB, Manske RC eds. Clinical Orthopaedic Rehabilitation. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2011:273-274.
25. Hogg-Johnson S, van der Velde G, Carroll LJ, et al. The burden and determinants of neck pain in the general population: results of the Bone and Joint Decade 2000-2010 Task Force on Neck Pain and Its Associated Disorders. Spine. 2008;33(suppl 4):S39-S51.
26. Larsson B, Søgaard K, Rosendal L. Work related neck-shoulder pain: a review on magnitude, risk factors, biochemical characteristics, clinical picture and preventive interventions. Best Pract Res Clin Rheumatol. 2007; 21:447-463.
27. Giles LG, Muller R. Chronic spinal pain: a randomized clinical trial comparing medication, acupuncture, and spinal manipulation. Spine. 2003;28:1490-1502.
28. Bronfort G, Evans R, Anderson A, et al. Spinal manipulation, medication, or home exercise with advice for acute and subacute neck pain: a randomized trial. Ann Intern Med. 2012;156:1-10.
29. Evans R, Bronfort G, Bittell S, et al. A pilot study for a randomized clinical trial assessing chiropractic care, medical care, and self-care education for acute and subacute neck pain patients. J Manipulative Physiol Ther. 2003;26:403-411.
1. Hubbard TJ, Aronson SL, Denegar CR. Does cryotherapy hasten return to participation? A systematic review. J Athl Train. 2004;39:88-94.
2. Ho SS, Coel MN, Kagawa R, et al. The effects of ice on blood flow and bone metabolism in knees. Am J Sports Med. 1994;22:537-540.
3. Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65.
4. Bleakley CM, O’Connor S, Tully MA, et al. The PRICE study (Protection Rest Ice Compression Elevation): design of a randomised controlled trial comparing standard versus cryokinetic ice applications in the management of acute ankle sprain. BMC Musculoskelet Disord. 2007;8:125.
5. Mayer JM, Ralph L, Look M, et al. Treating acute low back pain with continuous low-level heat wrap therapy and/or exercise: a randomized controlled trial. Spine J. 2005;5:395-403.
6. Cetin N, Aytar A, Atalay A, et al. Comparing hot pack, short-wave diathermy, ultrasound, and TENS on isokinetic strength, pain, and functional status of women with osteoarthritic knees: a single-blind, randomized, controlled trial. Am J Phys Med Rehabil. 2008;87:443-451.
7. Waterman BR, Owens BD, Davey S, et al. The epidemiology of ankle sprains in the United States. J Bone Joint Surg Am. 2010;92:2279-2284.
8. Fong DT, Hong Y, Chan LK, et al. A systematic review on ankle injury and ankle sprain in sports. Sports Med. 2007;37:73-94.
9. Kerkhoffs GM, Rowe BH, Assendelft WJ, et al. Immobilisation and functional treatment for acute lateral ankle ligament injuries in adults. Cochrane Database Syst Rev. 2002(3):CD003762.
10. Mattacola CG, Dwyer MK. Rehabilitation of the ankle after acute sprain or chronic instability. J Ath Train. 2002;37:413-429.
11. Hü bscher M, Zech A, Pfeifer K, et al. Neuromuscular training for sports injury prevention: a systematic review. Med Sci Sports Exerc. 2010;42:413-421.
12. Emery CA, Meeuwisse WH. The effectiveness of a neuromuscular prevention strategy to reduce injuries in youth soccer: a cluster-randomised controlled trial. Br J Sports Med. 2010;44:555-562.
13. Tiemstra JD. Update on acute ankle sprains. Am Fam Physician. 2012;85:1170-1176.
14. Beynnon BD, Murphy DF, Alosa DM. Predictive factors for lateral ankle sprains: a literature review. J Ath Train. 2002;37:376-380.
15. Schiftan GS, Ross LA, Hahne AJ. The effectiveness of proprioceptive training in preventing ankle sprains in sporting populations: a systematic review and meta-analysis. J Sci Med Sport. 2015;18:238–244.
16. Hupperets MD, Verhagen EA, van Mechelen W. Effect of unsupervised home based proprioceptive training on recurrences of ankle sprain: randomised controlled trial. BMJ. 2009;339:b2684
17. Thompson JV, Saini SS, Reb CW, et al. Diagnosis and management of plantar fasciitis. J Am Osteopath Assoc. 2014;114:900-906.
18. DiGiovanni BF, Nawoczenski DA, Malay DP, et al. Plantar fascia-specific stretching exercise improves outcomes in patients with chronic plantar fasciitis. A prospective clinical trial with two-year follow-up. J Bone Joint Surg Am. 2006;88:1775-1781.

19. Lee SY, McKeon P, Hertel J. Does the use of orthoses improve self-reported pain and function measures in patients with plantar fasciitis? A meta-analysis. Phys Ther Sport. 2009;10:12-18.
20. Glaviano NR, Key M, Hart JM, et al. Demographic and epidemiological trends in patellofemoral pain. J Sports Phys Ther. 2015;10: 281-290.
21. Louden JK. Biomechanics and pathomechanics of the patellofemoral joint. Int J Sports Phys Ther. 2016;11: 820-830.
22. Ferber R, Bolgla L, Earl-Boehm JE, et al. Strengthening of hip and core versus knee muscles for the treatment of patellofemoral pain: a multicenter randomized controlled trial. J Ath Train. 2015;50: 366-377.
23. Collins NJ, Bisset LM, Crossley KM, et al. Efficacy of nonsurgical interventions for anterior knee pain: systematic review and meta-analysis of randomized trials. Sports Med. 2013;41:31-49.
24. Bolgla LA. Hip strength and kinematics in patellofemoral syndrome. In: Brotzman SB, Manske RC eds. Clinical Orthopaedic Rehabilitation. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2011:273-274.
25. Hogg-Johnson S, van der Velde G, Carroll LJ, et al. The burden and determinants of neck pain in the general population: results of the Bone and Joint Decade 2000-2010 Task Force on Neck Pain and Its Associated Disorders. Spine. 2008;33(suppl 4):S39-S51.
26. Larsson B, Søgaard K, Rosendal L. Work related neck-shoulder pain: a review on magnitude, risk factors, biochemical characteristics, clinical picture and preventive interventions. Best Pract Res Clin Rheumatol. 2007; 21:447-463.
27. Giles LG, Muller R. Chronic spinal pain: a randomized clinical trial comparing medication, acupuncture, and spinal manipulation. Spine. 2003;28:1490-1502.
28. Bronfort G, Evans R, Anderson A, et al. Spinal manipulation, medication, or home exercise with advice for acute and subacute neck pain: a randomized trial. Ann Intern Med. 2012;156:1-10.
29. Evans R, Bronfort G, Bittell S, et al. A pilot study for a randomized clinical trial assessing chiropractic care, medical care, and self-care education for acute and subacute neck pain patients. J Manipulative Physiol Ther. 2003;26:403-411.
PRACTICE RECOMMENDATIONS
❯ Have patients apply ice to an acute injury for 15 to 20 minutes at a time to help control inflammation, and prescribe an anti-inflammatory medication, if indicated. A
❯ Reserve heat application for use following the acute phase of injury to decrease stiffness. A
❯ Instruct patients who have an acute lateral ankle sprain to begin “ankle ABCs” and other range-of-motion exercises once acute pain subsides. C
❯ Consider recommending an eccentric heel stretch to help alleviate plantar fasciitis symptoms. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series











