User login
Dual Radial Styloid and Volar Plating for Unstable Fractures of the Distal Radius
ABSTRACT
As the operative management of displaced distal radius fractures evolves, intraoperative techniques and fixation strategies evolve as well. Achieving and maintaining an acceptable reduction is paramount but can be difficult with particular fracture patterns. In this article, we describe the use of a radial column plate as a reduction tool in the management of unstable distal radius fractures, along with clinical and radiographic clinical outcomes. This technique can be useful in situations where multiplanar instability exists, or simply when intraoperative assistance is limited. Surgeons can expect acceptable radiographic and clinical outcomes when using this technique, although effects on scar formation and wrist range of motion are currently not known.
Continue to: Distal radius fractures...
Distal radius fractures are among the most common orthopedic injuries encountered; their reported incidence is >640,000 annually and is estimated to increase.1-4 The management of these injuries has evolved from closed reduction and casting to percutaneous pinning and internal fixation, as the importance of achieving and maintaining an anatomic reduction has become more apparent.5-7 More recently, volar locking plates have emerged as a way to prevent complications associated with dorsal plating. Most authors agree that volar locked plating achieves stable fixation and allows for early postoperative wrist range of motion (ROM).5,8-11 However, a volar approach to a dorsally unstable fracture creates difficulty with regard to reduction at the time of surgery and several reports have noted mechanical failure with utilization of locked volar plating alone.12-15
Dual plating of unstable distal radius fractures with a volar locking plate and a radial column plate has been described in the past in the setting of severely comminuted fractures or in patterns with a large radial styloid fragment that was not addressed with a volar locking plate alone.16-19 The purpose of this study is to present the use of the radial column plate as a tool that allows a surgeon to achieve and maintain reduction during open reduction and internal fixation (ORIF) of an unstable distal radius fracture.
OPERATIVE TECHNIQUE
Patients for whom ORIF is indicated include those with unstable distal radius fractures, with or without intra-articular extension and involvement of both the intermediate and lateral columns.
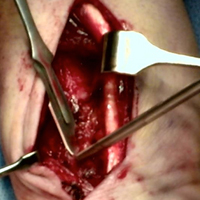
The patient is positioned supine on the operating table with the operative hand placed palm-up on a radiolucent hand table. A volar approach to the distal radius is undertaken, utilizing the interval between the flexor carpi radialis (FCR) tendon and the radial artery. The floor of the FCR sheath is incised, and a self-retaining retractor with blunt tips can be placed to permit visualization. The pronator quadratus (PQ) is sharply reflected off the radial boarder of the distal radius and approximately 1 mm to 2 mm proximal to the radiocarpal joint with an L-shaped incision for fracture site exposure. The brachioradialis is then identified and tenotomized with a scalpel (Figure 1).
A preliminary reduction is then performed using a combination of axial traction and palmar translation of the carpus. The surgeon should not be concerned with radial height or inclination at this point; however, volar tilt should be established as best as possible. A rolled towel is placed dorsal to the metacarpals, holding the wrist in a flexed position as it is placed back onto the radiolucent hand table.
Continue to: A 7 to 8 hole...
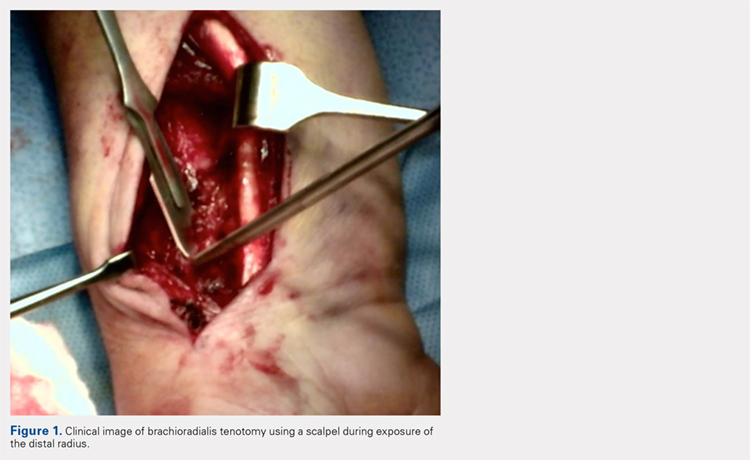
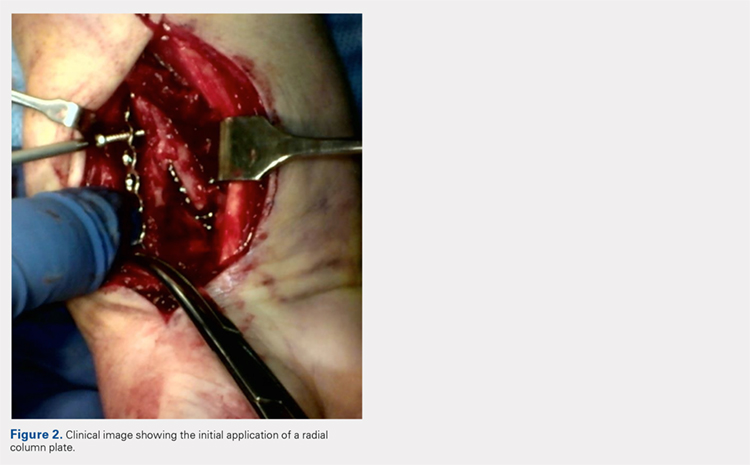
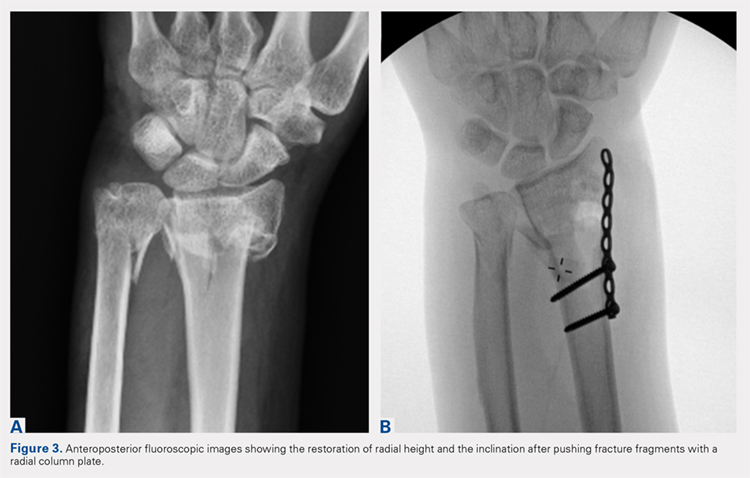
A 7 to 8 hole 2.0-mm reconstruction plate (DePuy Synthes) is bent to the shape of the radial boarder of the distal radius. Undercontouring of the plate is necessary to allow for its use as a reduction tool. The plate is then applied to the radial column ensuring that the distal aspect of the plate engages the distal fracture fragment(s) (Figure 2). A single 2.4-mm fully threaded cortical screw in the radial to ulnar direction is then placed bicortically in the proximal fragment in the hole nearest the fracture site. As the screw is tightened, the plate will push the distal fragment(s) due to its undercontoured shape, and in doing so, will restore radial height and inclination (Figure 3). As this screw is being used as a “working screw,” it will be longer than needed after final tightening. A second screw is then placed proximally to prevent rotation of the plate, and the initial screw can be replaced if its length is of concern. If it is the intention of the surgeon to remove the plate prior to wound closure, the second screw is typically not necessary, and there is no indication for exchanging the first screw if it is long.
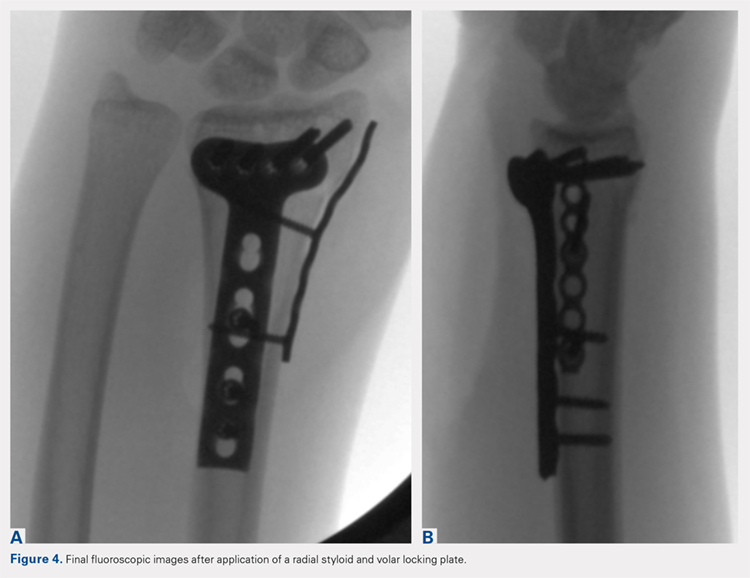
At this point, final changes to the reduction can still be performed, as the distal fragment(s) has no fixation except for a buttress plate on its radial border. However, the pressure applied by this plate is still typically adequate to maintain a reduction without the use of percutaneous pins or an assistant holding the reduction. Volar fixation is then applied and positioned under both direct visualization and fluoroscopic aid, and cortical and locking screws are inserted as needed (Figure 4). The radial styloid plate can then be removed; however, it is our preference to leave it in place, as we have not seen any postoperative issues that we can attribute to this technique. The PQ is then repaired over the volar locking plate directly to the radial column plate.
At our institution, patients are maintained in a plaster volar-based wrist splint for a period of 2 weeks postoperatively. After splint and suture removal, active and passive ROM exercises of the wrist and hand are initiated, and a custom thermoplast volar wrist splint is manufactured. This splint is to be worn at all times except during physical therapy. At the 6-week postoperative visit, all restrictions are lifted, assuming there are no complications or unexpected issues. Patients are then seen for follow-up at 3 and 6 months postoperatively. Continued follow-up is indicated if patients are following an abnormal clinical or radiographic course.
MATERIALS AND METHODS
After Institutional Review Board approval was obtained, a clinical outcomes registry was queried to identify all patients treated operatively by the senior author (DGL) for a distal radius fracture at our Level 1 trauma center between August 2002 and December 2013. Adult (age >18 years) patients with isolated distal radius fractures treated with a radial styloid plate were included for initial review (N = 261). Patients for whom 6-month clinical or radiographic outcomes were unknown were then excluded (n = 225).
Patient demographics were recorded from the existing database along with visual analog scale, Quick Disabilities of the Arm, Shoulder and Hand (DASH), and short form 36 (SF-36) physical component scores (PCS) and mental component scores (MCS) from the final follow-up visit. Injury and intraoperative and final radiographs were assessed by a single reviewer (MRG) using calibrated radiographs on our institution’s picture archiving and communication system. Radial height, radial inclination, and volar tilt were documented for each time point except for radial height, which was not recorded for intraoperative fluoroscopy images due to lack of calibration. Intra-articular extension was noted on injury films. Wound complications, the presence of a deep or superficial infection, and removal of implants after union were recorded.
Continue to: RESULTS
RESULTS
Thirty-six patients met the inclusion criteria and were therefore included in the study. The average age at the time of surgery was 60.6 years (range, 25-87 years), 27 patients (75%) were female, and 21 (58%) had left-sided injuries. Patient comorbidities can be seen in Table 1. Twenty-six fractures (72.2%) had intra-articular extension. Average follow-up was 15.6 months (range, 6-53.9 months).
Table 1. Comorbidities of Patients Treated with Radial Column Plating
| Total No. of patients | 36 | |
| Diabetes mellitus | 2 | 5.6% |
| Hyperlipidemia | 7 | 19.4% |
| Hypertension | 11 | 30.6% |
| Current smoker | 4 | 11.1% |
| Current alcohol abuse | 1 | 2.8% |
| Peripheral vascular disease | 0 | 0.0% |
| Mean body mass index | 27.0 | Range: 19-34.5 |
Radiographic measurements at the time of injury, surgery, and final follow-up can be seen in Table 2. As previously noted, radial height could not be recorded on intraoperative films due to the use of fluoroscopy, which is not calibrated at our institution. The average changes in radial inclination and volar tilt from the time of surgery (intraoperative fluoroscopy) to final follow-up were 0.46° (range, −4.4°-4.3°) and 0.24° (range, −10.6°-9.6°), respectively. All patients had acceptable radial height, radial inclination, and volar tilt at final follow-up. Clinical outcomes were obtained at a mean of 15.6 months (range, 6-54 months) and were generally good, with a mean DASH score of 20.7 (range, 0-57.5), SF-36 PCS of 45.4 (range, 22.7-68.0), and SF-36 MCS of 50.5 (range, 22.3-64.1) (Table 3). Of the 36 patients with 6-month outcome scores, 13 (36.1%) elected for implant removal after fracture union at a mean of 7.6 months after index surgery (range, 3.2-49.8 months). No infections or wound complications were noted.
Table 2. Radiographic Measurements for Patients Treated with Radial Column Plating
| Mean Measurement | Range | |
| Injury radiographs | ||
| Radial inclination (degrees) | 7.3 | −22.9-22 |
| Radial height (mm) | 3.3 | −14.9-11.5 |
| Volar tilt (degrees) | −10.4 | −49.2-33.9 |
| Intraoperative fluoroscopy | ||
| Radial inclination (degrees) | 21.1 | 13.1-26.6 |
| Volar tilt (degrees) | 6.2 | −3.6-12.2 |
| Final radiographs | ||
| Radial inclination (degrees) | 21.5 | 14.5-29.2 |
| Radial height (mm) | 11.0 | 7.6-14.6 |
| Volar tilt (degrees) | 6.8 | −12.4-18.8 |
DISCUSSION
In this article, we described the use of a radial column plate as a tool to achieve and maintain a reduction during the surgical fixation of an unstable distal radius fracture with a volar locking plate. We have further presented a series of 36 patients treated in this manner and their clinical and radiographic outcomes. This technique permits the maintenance of coronal alignment, thereby limiting the use of percutaneous techniques or the need to manually hold fracture fragments in a reduced position, which may be valuable to the surgeon who is operating without a surgical assistant.
Table 3. Clinical Outcome Scores at Final Follow-Up for Patients Treated with Radial Column Plating
| Outcome Score | Mean Score | Range |
| VAS | 1.4 | 0-7.5 |
| DASH | 20.7 | 0-57.5 |
| PCS | 45.4 | 22.7-68 |
| MCS | 50.5 | 22.3-64.1 |
Abbreviations: DASH, Quick Disabilities of the Arm, Shoulder and Hand; MCS, mental component scores; PCS, physical component scores; VAS, visual analog scale.
In addition to its value as a reduction tool, unlike traditional temporary k-wire fixation, we believe that the utilization of a radial styloid plate allows for increased stability until fracture union is achieved. Biomechanical studies have demonstrated favorable results with the use of a radial column plate. Grindel and colleagues20 evaluated dual radial styloid and volar radius plating vs volar plating alone in their biomechanical comparison of 8 cadaveric matched hand and forearm pairs. Specimens were fixated with a volar locking plate, and a 1-cm wedge osteotomy was created dorsally approximately 2 cm from the articular margin. The distal fragment was then osteotomized longitudinally between the 2 ulnar and 2 radial distal locking screws to create a fracture pattern that mimics a dorsally unstable injury with intra-articular extension. Half of the specimens then underwent radial styloid plating with 2 screws securing the construct proximally, and load-to-failure testing was performed. The authors found that utilization of both the volar and radial styloid plates resulted in 50% increased stiffness and 76% increased force-to-failure as compared with radial styloid plating alone. Similar, although not statistically significant, results were found by Blythe and colleagues.21 In their cadaveric study, dorsal and volar plating with an additional radial column plate resulted in improved stiffness with axial loading compared to volar or dorsal plating alone 21.
Two prior studies have presented outcome data after fixation of distal radius fractures with radial column and volar radius dual plating. Tang and colleagues16 described this technique and presented postoperative outcomes in 8 patients followed for an average of 35 weeks. They reported a 100% union rate, no loss of reduction, and a mean DASH score of 19.9. Jacobi and colleagues17 also described this technique in their 2010 report. In their cohort of 10 patients treated by multiple surgeons, they found a mean of 39° of flexion, 49° of extension, 75° of pronation, and 75° of supination at 24 months postoperatively. Eight patients were rated as “excellent,” 1 as “good,” and 1 as “fair” according to the Gartland and Werley score, with all 10 cases achieving bony union. No cases demonstrated loss of volar tilt, radial length, or radial inclination. In both studies, however, the use of the radial column plate was advocated as a fragment-specific fixation tool and not as a reduction tool.
Continue to: Although 1-year DASH scores...
Although 1-year DASH scores for volar plating alone have been shown in the literature to be consistently within 6 and 13, 3-month and 6-month scores have historically been >18.22-27 Our short-term clinical results (Table 3) are comparable to these historic controls. Further, within our cohort there were no cases of nonunion, postoperative infection, or wound complications, and radiographic measures show maintenance of reduction at final follow-up (Table 2).
We do recognize that 36.1% (13/36) of our cohort had their distal radius implants removed. Although this incidence is high, it stems from the fact that patients who elect for implant removal are more likely to have had an atypical postoperative course and are therefore followed for longer than 6 months. Those who do not elect for removal are typically discharged from care after their 3-month postoperative visit, and were therefore not eligible for inclusion in this study. Overall, a total of 261 patients have been treated with this technique by the senior surgeon. Of those patients, only 28 (10.7%) underwent removal of surgical implants. If the remaining patients had been followed for the full 6 months, it is likely that outcome scores would have been skewed in a more favorable direction.
Surgeons electing to utilize radial styloid plating for displaced distal radius fractures should recognize that the required increased surgical dissection might lead to increased scar formation and postoperative stiffness. A limitation of this study is the lack of quantitative wrist ROM data. Future studies may compare final clinical outcomes and ROM for patients treated with and without radial column fixation.
CONCLUSION
We advocate for the use of a radial column plate as a tool to help achieve and maintain fracture reduction in the setting of an unstable distal radius fracture being treated with ORIF. This technique may be particularly useful when a surgical assistant is not available. Surgeons can expect clinical and radiographic results that are similar to those of volar locked plating alone.
1. Larsen CF, Lauritsen J. Epidemiology of acute wrist trauma. Int J Epidemiol. 1993;22(5):911-916.
2. Chung KC, Spilson SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908-915. doi:10.1053/jhsu.2001.26322.
3. Melton LJ 3rd, Amadio PC, Crowson CS, O'Fallon WM. Long-term trends in the incidence of distal forearm fractures. Osteoporos Int. 1998;8(4):341-348.
4. Hagino H, Yamamoto K, Ohshiro H, Nakamura T, Kishimoto H, Nose T. Changing incidence of hip, distal radius, and proximal humerus fractures in Tottori Prefecture, Japan. Bone. 1999;24(3):265-270.
5. Diaz-Garcia RJ, Chung KC. The evolution of distal radius fracture management: A historical treatise. Hand Clin. 2012;28(2):105-111. doi:10.1016/j.hcl.2012.02.007.
6. McQueen M, Caspers J. Colles fracture: Does the anatomical result affect the final function? J Bone Joint Surg Br. 1988;70(4):649-651.
7. Stewart HD, Innes AR, Burke FD. Factors affecting the outcome of Colles' fracture: an anatomical and functional study. Injury. 1985;16(5):289-295.
8. Knight D, Hajducka C, Will E, McQueen M. Locked volar plating for unstable distal radial fractures: Clinical and radiological outcomes. Injury. 2010;41(2):184-189. doi:10.1016/j.injury.2009.08.024.
9. Anakwe R, Khan L, Cook R, McEachan J. Locked volar plating for complex distal radius fractures: patient reported outcomes and satisfaction. J Orthop Surg Res. 2010;5:51. doi:10.1186/1749799X-5-51.
10. Gruber G, Gruber K, Giessauf C, et al. Volar plate fixation of AO type C2 and C3 distal radius fractures, a single-center study of 55 patients. J Orthop Trauma. 2008;22(7):467-472. doi:10.1097/BOT.0b013e318180db09.
11. Koval KJ, Harrast JJ, Anglen JO, Weinstein JN. Fractures of the distal part of the radius. The evolution of practice over time. Where’s the evidence? J Bone Joint Surg Am. 2008;90(9):1855-1861. doi:10.2106/JBJS.G.01569.
12. Foo TL, Gan AW, Soh T, Chew WY. Mechanical failure of the distal radius volar locking plate. J Orthop Surg (Hong Kong). 2013;21(3):332-336. doi:10.11777/230949901302100314.
13. Ward CM, Kuhl TL, Adams BD. Early complications of volar plating of distal radius fractures and their relationship to surgeon experience. Hand (N Y). 2011;6(2):185-189. doi:10.1007/s11552-010-9313-5.
14. Min W, Kaplan K, Miyamoto R, Tejwani NC. A unique failure mechanism of a distal radius fracture fixed with volar plating--a case report. Bull NYU Hosp Jt Dis. 2010;68(4):304-306.
15. Cao J, Ozer K. Failure of volar locking plate fixation of an extraarticular distal radius fracture: A case report. Patient Saf Surg. 2010;4(1):19. doi:10.1186/1754-9493-4-19.
16. Tang P, Ding A, Uzumcugil A. Radial column and volar plating (RCVP) for distal radius fractures with a radial styloid component or severe comminution. Tech Hand Up Extrem Surg. 2010;14(3):143-149. doi:10.1097/BTH.0b013e3181cae14d.
17. Jacobi M, Wahl P, Kohut G. Repositioning and stabilization of the radial styloid process in comminuted fractures of the distal radius using a single approach: The radio-volar double plating technique. J Orthop Surg Res. 2010;5:55. doi:10.1186/1749-799X-5-55.
18. Rikli DA, Regazzoni P. The double plating technique for distal radius fractures. Tech Hand Up Extrem Surg. 2000;4(2):107-114.
19. Rikli DA, Regazzoni P. Fractures of the distal end of the radius treated by internal fixation and early function. A preliminary report of 20 cases. J Bone Joint Surg Br. 1996;78(4):588-592.
20. Grindel SI, Wang M, Gerlach M, McGrady LM, Brown S. Biomechanical comparison of fixed-angle volar plate versus fixed-angle volar plate plus fragment-specific fixation in a cadaveric distal radius fracture model. J Hand Surg Am. 2007;32(2):194-199. doi:10.1016/j.jhsa.2006.12.003.
21. Blythe M, Stoffel K, Jarrett P, Kuster M. Volar versus dorsal locking plates with and without radial styloid locking plates for the fixation of dorsally comminuted distal radius fractures: A biomechanical study in cadavers. J Hand Surg Am. 2006;31(10):1587-1593. doi:10.1016/j.jhsa.2006.09.011.
22. Loveridge J, Ahearn N, Gee C, Pearson D, Sivaloganathan S, Bhatia R. Treatment of distal radial fractures with the DVR-A plate--the early bristol experience. Hand Surg. 2013;18(2):159-167. doi:10.1142/S0218810413500184.
23. Karantana A, Downing ND, Forward DP, et al. Surgical treatment of distal radial fractures with a volar locking plate versus conventional percutaneous methods: a randomized controlled trial. J Bone Joint Surg Am. 2013;95(19):1737-1744. doi:10.2106/JBJS.L.00232.
24. Egol K, Walsh M, Tejwani N, McLaurin T, Wynn C, Paksima N. Bridging external fixation and supplementary kirschner-wire fixation versus volar locked plating for unstable fractures of the distal radius: A randomised, prospective trial. J Bone Joint Surg Br. 2008;90(9):1214-1221. doi:10.1302/0301-620X.90B9.20521.
25. von Recum J, Matschke S, Jupiter JB, et al. Characteristics of two different locking compression plates in the volar fixation of complex articular distal radius fractures. Bone Joint Res. 2012;1(6):111-117. doi:10.1302/2046-3758.16.2000008.
26. Safi A, Hart R, Těknědžjan B, Kozák T. Treatment of extra-articular and simple articular distal radial fractures with intramedullary nail versus volar locking plate. J Hand Surg Eur Vol. 2013;38(7):774-779. doi:10.1177/1753193413478715.
27. Kim JK, Park SD. Outcomes after volar plate fixation of low-grade open and closed distal radius fractures are similar. Clin Orthop Relat Res. 2013;471(6):2030-2035. doi:10.1007/s11999-013-2798-9.
ABSTRACT
As the operative management of displaced distal radius fractures evolves, intraoperative techniques and fixation strategies evolve as well. Achieving and maintaining an acceptable reduction is paramount but can be difficult with particular fracture patterns. In this article, we describe the use of a radial column plate as a reduction tool in the management of unstable distal radius fractures, along with clinical and radiographic clinical outcomes. This technique can be useful in situations where multiplanar instability exists, or simply when intraoperative assistance is limited. Surgeons can expect acceptable radiographic and clinical outcomes when using this technique, although effects on scar formation and wrist range of motion are currently not known.
Continue to: Distal radius fractures...
Distal radius fractures are among the most common orthopedic injuries encountered; their reported incidence is >640,000 annually and is estimated to increase.1-4 The management of these injuries has evolved from closed reduction and casting to percutaneous pinning and internal fixation, as the importance of achieving and maintaining an anatomic reduction has become more apparent.5-7 More recently, volar locking plates have emerged as a way to prevent complications associated with dorsal plating. Most authors agree that volar locked plating achieves stable fixation and allows for early postoperative wrist range of motion (ROM).5,8-11 However, a volar approach to a dorsally unstable fracture creates difficulty with regard to reduction at the time of surgery and several reports have noted mechanical failure with utilization of locked volar plating alone.12-15
Dual plating of unstable distal radius fractures with a volar locking plate and a radial column plate has been described in the past in the setting of severely comminuted fractures or in patterns with a large radial styloid fragment that was not addressed with a volar locking plate alone.16-19 The purpose of this study is to present the use of the radial column plate as a tool that allows a surgeon to achieve and maintain reduction during open reduction and internal fixation (ORIF) of an unstable distal radius fracture.
OPERATIVE TECHNIQUE
Patients for whom ORIF is indicated include those with unstable distal radius fractures, with or without intra-articular extension and involvement of both the intermediate and lateral columns.
The patient is positioned supine on the operating table with the operative hand placed palm-up on a radiolucent hand table. A volar approach to the distal radius is undertaken, utilizing the interval between the flexor carpi radialis (FCR) tendon and the radial artery. The floor of the FCR sheath is incised, and a self-retaining retractor with blunt tips can be placed to permit visualization. The pronator quadratus (PQ) is sharply reflected off the radial boarder of the distal radius and approximately 1 mm to 2 mm proximal to the radiocarpal joint with an L-shaped incision for fracture site exposure. The brachioradialis is then identified and tenotomized with a scalpel (Figure 1).

A preliminary reduction is then performed using a combination of axial traction and palmar translation of the carpus. The surgeon should not be concerned with radial height or inclination at this point; however, volar tilt should be established as best as possible. A rolled towel is placed dorsal to the metacarpals, holding the wrist in a flexed position as it is placed back onto the radiolucent hand table.
Continue to: A 7 to 8 hole...
A 7 to 8 hole 2.0-mm reconstruction plate (DePuy Synthes) is bent to the shape of the radial boarder of the distal radius. Undercontouring of the plate is necessary to allow for its use as a reduction tool. The plate is then applied to the radial column ensuring that the distal aspect of the plate engages the distal fracture fragment(s) (Figure 2). A single 2.4-mm fully threaded cortical screw in the radial to ulnar direction is then placed bicortically in the proximal fragment in the hole nearest the fracture site. As the screw is tightened, the plate will push the distal fragment(s) due to its undercontoured shape, and in doing so, will restore radial height and inclination (Figure 3). As this screw is being used as a “working screw,” it will be longer than needed after final tightening. A second screw is then placed proximally to prevent rotation of the plate, and the initial screw can be replaced if its length is of concern. If it is the intention of the surgeon to remove the plate prior to wound closure, the second screw is typically not necessary, and there is no indication for exchanging the first screw if it is long.

At this point, final changes to the reduction can still be performed, as the distal fragment(s) has no fixation except for a buttress plate on its radial border. However, the pressure applied by this plate is still typically adequate to maintain a reduction without the use of percutaneous pins or an assistant holding the reduction. Volar fixation is then applied and positioned under both direct visualization and fluoroscopic aid, and cortical and locking screws are inserted as needed (Figure 4). The radial styloid plate can then be removed; however, it is our preference to leave it in place, as we have not seen any postoperative issues that we can attribute to this technique. The PQ is then repaired over the volar locking plate directly to the radial column plate.

At our institution, patients are maintained in a plaster volar-based wrist splint for a period of 2 weeks postoperatively. After splint and suture removal, active and passive ROM exercises of the wrist and hand are initiated, and a custom thermoplast volar wrist splint is manufactured. This splint is to be worn at all times except during physical therapy. At the 6-week postoperative visit, all restrictions are lifted, assuming there are no complications or unexpected issues. Patients are then seen for follow-up at 3 and 6 months postoperatively. Continued follow-up is indicated if patients are following an abnormal clinical or radiographic course.

MATERIALS AND METHODS
After Institutional Review Board approval was obtained, a clinical outcomes registry was queried to identify all patients treated operatively by the senior author (DGL) for a distal radius fracture at our Level 1 trauma center between August 2002 and December 2013. Adult (age >18 years) patients with isolated distal radius fractures treated with a radial styloid plate were included for initial review (N = 261). Patients for whom 6-month clinical or radiographic outcomes were unknown were then excluded (n = 225).
Patient demographics were recorded from the existing database along with visual analog scale, Quick Disabilities of the Arm, Shoulder and Hand (DASH), and short form 36 (SF-36) physical component scores (PCS) and mental component scores (MCS) from the final follow-up visit. Injury and intraoperative and final radiographs were assessed by a single reviewer (MRG) using calibrated radiographs on our institution’s picture archiving and communication system. Radial height, radial inclination, and volar tilt were documented for each time point except for radial height, which was not recorded for intraoperative fluoroscopy images due to lack of calibration. Intra-articular extension was noted on injury films. Wound complications, the presence of a deep or superficial infection, and removal of implants after union were recorded.
Continue to: RESULTS
RESULTS
Thirty-six patients met the inclusion criteria and were therefore included in the study. The average age at the time of surgery was 60.6 years (range, 25-87 years), 27 patients (75%) were female, and 21 (58%) had left-sided injuries. Patient comorbidities can be seen in Table 1. Twenty-six fractures (72.2%) had intra-articular extension. Average follow-up was 15.6 months (range, 6-53.9 months).
Table 1. Comorbidities of Patients Treated with Radial Column Plating
| Total No. of patients | 36 | |
| Diabetes mellitus | 2 | 5.6% |
| Hyperlipidemia | 7 | 19.4% |
| Hypertension | 11 | 30.6% |
| Current smoker | 4 | 11.1% |
| Current alcohol abuse | 1 | 2.8% |
| Peripheral vascular disease | 0 | 0.0% |
| Mean body mass index | 27.0 | Range: 19-34.5 |
Radiographic measurements at the time of injury, surgery, and final follow-up can be seen in Table 2. As previously noted, radial height could not be recorded on intraoperative films due to the use of fluoroscopy, which is not calibrated at our institution. The average changes in radial inclination and volar tilt from the time of surgery (intraoperative fluoroscopy) to final follow-up were 0.46° (range, −4.4°-4.3°) and 0.24° (range, −10.6°-9.6°), respectively. All patients had acceptable radial height, radial inclination, and volar tilt at final follow-up. Clinical outcomes were obtained at a mean of 15.6 months (range, 6-54 months) and were generally good, with a mean DASH score of 20.7 (range, 0-57.5), SF-36 PCS of 45.4 (range, 22.7-68.0), and SF-36 MCS of 50.5 (range, 22.3-64.1) (Table 3). Of the 36 patients with 6-month outcome scores, 13 (36.1%) elected for implant removal after fracture union at a mean of 7.6 months after index surgery (range, 3.2-49.8 months). No infections or wound complications were noted.
Table 2. Radiographic Measurements for Patients Treated with Radial Column Plating
| Mean Measurement | Range | |
| Injury radiographs | ||
| Radial inclination (degrees) | 7.3 | −22.9-22 |
| Radial height (mm) | 3.3 | −14.9-11.5 |
| Volar tilt (degrees) | −10.4 | −49.2-33.9 |
| Intraoperative fluoroscopy | ||
| Radial inclination (degrees) | 21.1 | 13.1-26.6 |
| Volar tilt (degrees) | 6.2 | −3.6-12.2 |
| Final radiographs | ||
| Radial inclination (degrees) | 21.5 | 14.5-29.2 |
| Radial height (mm) | 11.0 | 7.6-14.6 |
| Volar tilt (degrees) | 6.8 | −12.4-18.8 |
DISCUSSION
In this article, we described the use of a radial column plate as a tool to achieve and maintain a reduction during the surgical fixation of an unstable distal radius fracture with a volar locking plate. We have further presented a series of 36 patients treated in this manner and their clinical and radiographic outcomes. This technique permits the maintenance of coronal alignment, thereby limiting the use of percutaneous techniques or the need to manually hold fracture fragments in a reduced position, which may be valuable to the surgeon who is operating without a surgical assistant.
Table 3. Clinical Outcome Scores at Final Follow-Up for Patients Treated with Radial Column Plating
| Outcome Score | Mean Score | Range |
| VAS | 1.4 | 0-7.5 |
| DASH | 20.7 | 0-57.5 |
| PCS | 45.4 | 22.7-68 |
| MCS | 50.5 | 22.3-64.1 |
Abbreviations: DASH, Quick Disabilities of the Arm, Shoulder and Hand; MCS, mental component scores; PCS, physical component scores; VAS, visual analog scale.
In addition to its value as a reduction tool, unlike traditional temporary k-wire fixation, we believe that the utilization of a radial styloid plate allows for increased stability until fracture union is achieved. Biomechanical studies have demonstrated favorable results with the use of a radial column plate. Grindel and colleagues20 evaluated dual radial styloid and volar radius plating vs volar plating alone in their biomechanical comparison of 8 cadaveric matched hand and forearm pairs. Specimens were fixated with a volar locking plate, and a 1-cm wedge osteotomy was created dorsally approximately 2 cm from the articular margin. The distal fragment was then osteotomized longitudinally between the 2 ulnar and 2 radial distal locking screws to create a fracture pattern that mimics a dorsally unstable injury with intra-articular extension. Half of the specimens then underwent radial styloid plating with 2 screws securing the construct proximally, and load-to-failure testing was performed. The authors found that utilization of both the volar and radial styloid plates resulted in 50% increased stiffness and 76% increased force-to-failure as compared with radial styloid plating alone. Similar, although not statistically significant, results were found by Blythe and colleagues.21 In their cadaveric study, dorsal and volar plating with an additional radial column plate resulted in improved stiffness with axial loading compared to volar or dorsal plating alone 21.
Two prior studies have presented outcome data after fixation of distal radius fractures with radial column and volar radius dual plating. Tang and colleagues16 described this technique and presented postoperative outcomes in 8 patients followed for an average of 35 weeks. They reported a 100% union rate, no loss of reduction, and a mean DASH score of 19.9. Jacobi and colleagues17 also described this technique in their 2010 report. In their cohort of 10 patients treated by multiple surgeons, they found a mean of 39° of flexion, 49° of extension, 75° of pronation, and 75° of supination at 24 months postoperatively. Eight patients were rated as “excellent,” 1 as “good,” and 1 as “fair” according to the Gartland and Werley score, with all 10 cases achieving bony union. No cases demonstrated loss of volar tilt, radial length, or radial inclination. In both studies, however, the use of the radial column plate was advocated as a fragment-specific fixation tool and not as a reduction tool.
Continue to: Although 1-year DASH scores...
Although 1-year DASH scores for volar plating alone have been shown in the literature to be consistently within 6 and 13, 3-month and 6-month scores have historically been >18.22-27 Our short-term clinical results (Table 3) are comparable to these historic controls. Further, within our cohort there were no cases of nonunion, postoperative infection, or wound complications, and radiographic measures show maintenance of reduction at final follow-up (Table 2).
We do recognize that 36.1% (13/36) of our cohort had their distal radius implants removed. Although this incidence is high, it stems from the fact that patients who elect for implant removal are more likely to have had an atypical postoperative course and are therefore followed for longer than 6 months. Those who do not elect for removal are typically discharged from care after their 3-month postoperative visit, and were therefore not eligible for inclusion in this study. Overall, a total of 261 patients have been treated with this technique by the senior surgeon. Of those patients, only 28 (10.7%) underwent removal of surgical implants. If the remaining patients had been followed for the full 6 months, it is likely that outcome scores would have been skewed in a more favorable direction.
Surgeons electing to utilize radial styloid plating for displaced distal radius fractures should recognize that the required increased surgical dissection might lead to increased scar formation and postoperative stiffness. A limitation of this study is the lack of quantitative wrist ROM data. Future studies may compare final clinical outcomes and ROM for patients treated with and without radial column fixation.
CONCLUSION
We advocate for the use of a radial column plate as a tool to help achieve and maintain fracture reduction in the setting of an unstable distal radius fracture being treated with ORIF. This technique may be particularly useful when a surgical assistant is not available. Surgeons can expect clinical and radiographic results that are similar to those of volar locked plating alone.
ABSTRACT
As the operative management of displaced distal radius fractures evolves, intraoperative techniques and fixation strategies evolve as well. Achieving and maintaining an acceptable reduction is paramount but can be difficult with particular fracture patterns. In this article, we describe the use of a radial column plate as a reduction tool in the management of unstable distal radius fractures, along with clinical and radiographic clinical outcomes. This technique can be useful in situations where multiplanar instability exists, or simply when intraoperative assistance is limited. Surgeons can expect acceptable radiographic and clinical outcomes when using this technique, although effects on scar formation and wrist range of motion are currently not known.
Continue to: Distal radius fractures...
Distal radius fractures are among the most common orthopedic injuries encountered; their reported incidence is >640,000 annually and is estimated to increase.1-4 The management of these injuries has evolved from closed reduction and casting to percutaneous pinning and internal fixation, as the importance of achieving and maintaining an anatomic reduction has become more apparent.5-7 More recently, volar locking plates have emerged as a way to prevent complications associated with dorsal plating. Most authors agree that volar locked plating achieves stable fixation and allows for early postoperative wrist range of motion (ROM).5,8-11 However, a volar approach to a dorsally unstable fracture creates difficulty with regard to reduction at the time of surgery and several reports have noted mechanical failure with utilization of locked volar plating alone.12-15
Dual plating of unstable distal radius fractures with a volar locking plate and a radial column plate has been described in the past in the setting of severely comminuted fractures or in patterns with a large radial styloid fragment that was not addressed with a volar locking plate alone.16-19 The purpose of this study is to present the use of the radial column plate as a tool that allows a surgeon to achieve and maintain reduction during open reduction and internal fixation (ORIF) of an unstable distal radius fracture.
OPERATIVE TECHNIQUE
Patients for whom ORIF is indicated include those with unstable distal radius fractures, with or without intra-articular extension and involvement of both the intermediate and lateral columns.
The patient is positioned supine on the operating table with the operative hand placed palm-up on a radiolucent hand table. A volar approach to the distal radius is undertaken, utilizing the interval between the flexor carpi radialis (FCR) tendon and the radial artery. The floor of the FCR sheath is incised, and a self-retaining retractor with blunt tips can be placed to permit visualization. The pronator quadratus (PQ) is sharply reflected off the radial boarder of the distal radius and approximately 1 mm to 2 mm proximal to the radiocarpal joint with an L-shaped incision for fracture site exposure. The brachioradialis is then identified and tenotomized with a scalpel (Figure 1).

A preliminary reduction is then performed using a combination of axial traction and palmar translation of the carpus. The surgeon should not be concerned with radial height or inclination at this point; however, volar tilt should be established as best as possible. A rolled towel is placed dorsal to the metacarpals, holding the wrist in a flexed position as it is placed back onto the radiolucent hand table.
Continue to: A 7 to 8 hole...
A 7 to 8 hole 2.0-mm reconstruction plate (DePuy Synthes) is bent to the shape of the radial boarder of the distal radius. Undercontouring of the plate is necessary to allow for its use as a reduction tool. The plate is then applied to the radial column ensuring that the distal aspect of the plate engages the distal fracture fragment(s) (Figure 2). A single 2.4-mm fully threaded cortical screw in the radial to ulnar direction is then placed bicortically in the proximal fragment in the hole nearest the fracture site. As the screw is tightened, the plate will push the distal fragment(s) due to its undercontoured shape, and in doing so, will restore radial height and inclination (Figure 3). As this screw is being used as a “working screw,” it will be longer than needed after final tightening. A second screw is then placed proximally to prevent rotation of the plate, and the initial screw can be replaced if its length is of concern. If it is the intention of the surgeon to remove the plate prior to wound closure, the second screw is typically not necessary, and there is no indication for exchanging the first screw if it is long.

At this point, final changes to the reduction can still be performed, as the distal fragment(s) has no fixation except for a buttress plate on its radial border. However, the pressure applied by this plate is still typically adequate to maintain a reduction without the use of percutaneous pins or an assistant holding the reduction. Volar fixation is then applied and positioned under both direct visualization and fluoroscopic aid, and cortical and locking screws are inserted as needed (Figure 4). The radial styloid plate can then be removed; however, it is our preference to leave it in place, as we have not seen any postoperative issues that we can attribute to this technique. The PQ is then repaired over the volar locking plate directly to the radial column plate.

At our institution, patients are maintained in a plaster volar-based wrist splint for a period of 2 weeks postoperatively. After splint and suture removal, active and passive ROM exercises of the wrist and hand are initiated, and a custom thermoplast volar wrist splint is manufactured. This splint is to be worn at all times except during physical therapy. At the 6-week postoperative visit, all restrictions are lifted, assuming there are no complications or unexpected issues. Patients are then seen for follow-up at 3 and 6 months postoperatively. Continued follow-up is indicated if patients are following an abnormal clinical or radiographic course.

MATERIALS AND METHODS
After Institutional Review Board approval was obtained, a clinical outcomes registry was queried to identify all patients treated operatively by the senior author (DGL) for a distal radius fracture at our Level 1 trauma center between August 2002 and December 2013. Adult (age >18 years) patients with isolated distal radius fractures treated with a radial styloid plate were included for initial review (N = 261). Patients for whom 6-month clinical or radiographic outcomes were unknown were then excluded (n = 225).
Patient demographics were recorded from the existing database along with visual analog scale, Quick Disabilities of the Arm, Shoulder and Hand (DASH), and short form 36 (SF-36) physical component scores (PCS) and mental component scores (MCS) from the final follow-up visit. Injury and intraoperative and final radiographs were assessed by a single reviewer (MRG) using calibrated radiographs on our institution’s picture archiving and communication system. Radial height, radial inclination, and volar tilt were documented for each time point except for radial height, which was not recorded for intraoperative fluoroscopy images due to lack of calibration. Intra-articular extension was noted on injury films. Wound complications, the presence of a deep or superficial infection, and removal of implants after union were recorded.
Continue to: RESULTS
RESULTS
Thirty-six patients met the inclusion criteria and were therefore included in the study. The average age at the time of surgery was 60.6 years (range, 25-87 years), 27 patients (75%) were female, and 21 (58%) had left-sided injuries. Patient comorbidities can be seen in Table 1. Twenty-six fractures (72.2%) had intra-articular extension. Average follow-up was 15.6 months (range, 6-53.9 months).
Table 1. Comorbidities of Patients Treated with Radial Column Plating
| Total No. of patients | 36 | |
| Diabetes mellitus | 2 | 5.6% |
| Hyperlipidemia | 7 | 19.4% |
| Hypertension | 11 | 30.6% |
| Current smoker | 4 | 11.1% |
| Current alcohol abuse | 1 | 2.8% |
| Peripheral vascular disease | 0 | 0.0% |
| Mean body mass index | 27.0 | Range: 19-34.5 |
Radiographic measurements at the time of injury, surgery, and final follow-up can be seen in Table 2. As previously noted, radial height could not be recorded on intraoperative films due to the use of fluoroscopy, which is not calibrated at our institution. The average changes in radial inclination and volar tilt from the time of surgery (intraoperative fluoroscopy) to final follow-up were 0.46° (range, −4.4°-4.3°) and 0.24° (range, −10.6°-9.6°), respectively. All patients had acceptable radial height, radial inclination, and volar tilt at final follow-up. Clinical outcomes were obtained at a mean of 15.6 months (range, 6-54 months) and were generally good, with a mean DASH score of 20.7 (range, 0-57.5), SF-36 PCS of 45.4 (range, 22.7-68.0), and SF-36 MCS of 50.5 (range, 22.3-64.1) (Table 3). Of the 36 patients with 6-month outcome scores, 13 (36.1%) elected for implant removal after fracture union at a mean of 7.6 months after index surgery (range, 3.2-49.8 months). No infections or wound complications were noted.
Table 2. Radiographic Measurements for Patients Treated with Radial Column Plating
| Mean Measurement | Range | |
| Injury radiographs | ||
| Radial inclination (degrees) | 7.3 | −22.9-22 |
| Radial height (mm) | 3.3 | −14.9-11.5 |
| Volar tilt (degrees) | −10.4 | −49.2-33.9 |
| Intraoperative fluoroscopy | ||
| Radial inclination (degrees) | 21.1 | 13.1-26.6 |
| Volar tilt (degrees) | 6.2 | −3.6-12.2 |
| Final radiographs | ||
| Radial inclination (degrees) | 21.5 | 14.5-29.2 |
| Radial height (mm) | 11.0 | 7.6-14.6 |
| Volar tilt (degrees) | 6.8 | −12.4-18.8 |
DISCUSSION
In this article, we described the use of a radial column plate as a tool to achieve and maintain a reduction during the surgical fixation of an unstable distal radius fracture with a volar locking plate. We have further presented a series of 36 patients treated in this manner and their clinical and radiographic outcomes. This technique permits the maintenance of coronal alignment, thereby limiting the use of percutaneous techniques or the need to manually hold fracture fragments in a reduced position, which may be valuable to the surgeon who is operating without a surgical assistant.
Table 3. Clinical Outcome Scores at Final Follow-Up for Patients Treated with Radial Column Plating
| Outcome Score | Mean Score | Range |
| VAS | 1.4 | 0-7.5 |
| DASH | 20.7 | 0-57.5 |
| PCS | 45.4 | 22.7-68 |
| MCS | 50.5 | 22.3-64.1 |
Abbreviations: DASH, Quick Disabilities of the Arm, Shoulder and Hand; MCS, mental component scores; PCS, physical component scores; VAS, visual analog scale.
In addition to its value as a reduction tool, unlike traditional temporary k-wire fixation, we believe that the utilization of a radial styloid plate allows for increased stability until fracture union is achieved. Biomechanical studies have demonstrated favorable results with the use of a radial column plate. Grindel and colleagues20 evaluated dual radial styloid and volar radius plating vs volar plating alone in their biomechanical comparison of 8 cadaveric matched hand and forearm pairs. Specimens were fixated with a volar locking plate, and a 1-cm wedge osteotomy was created dorsally approximately 2 cm from the articular margin. The distal fragment was then osteotomized longitudinally between the 2 ulnar and 2 radial distal locking screws to create a fracture pattern that mimics a dorsally unstable injury with intra-articular extension. Half of the specimens then underwent radial styloid plating with 2 screws securing the construct proximally, and load-to-failure testing was performed. The authors found that utilization of both the volar and radial styloid plates resulted in 50% increased stiffness and 76% increased force-to-failure as compared with radial styloid plating alone. Similar, although not statistically significant, results were found by Blythe and colleagues.21 In their cadaveric study, dorsal and volar plating with an additional radial column plate resulted in improved stiffness with axial loading compared to volar or dorsal plating alone 21.
Two prior studies have presented outcome data after fixation of distal radius fractures with radial column and volar radius dual plating. Tang and colleagues16 described this technique and presented postoperative outcomes in 8 patients followed for an average of 35 weeks. They reported a 100% union rate, no loss of reduction, and a mean DASH score of 19.9. Jacobi and colleagues17 also described this technique in their 2010 report. In their cohort of 10 patients treated by multiple surgeons, they found a mean of 39° of flexion, 49° of extension, 75° of pronation, and 75° of supination at 24 months postoperatively. Eight patients were rated as “excellent,” 1 as “good,” and 1 as “fair” according to the Gartland and Werley score, with all 10 cases achieving bony union. No cases demonstrated loss of volar tilt, radial length, or radial inclination. In both studies, however, the use of the radial column plate was advocated as a fragment-specific fixation tool and not as a reduction tool.
Continue to: Although 1-year DASH scores...
Although 1-year DASH scores for volar plating alone have been shown in the literature to be consistently within 6 and 13, 3-month and 6-month scores have historically been >18.22-27 Our short-term clinical results (Table 3) are comparable to these historic controls. Further, within our cohort there were no cases of nonunion, postoperative infection, or wound complications, and radiographic measures show maintenance of reduction at final follow-up (Table 2).
We do recognize that 36.1% (13/36) of our cohort had their distal radius implants removed. Although this incidence is high, it stems from the fact that patients who elect for implant removal are more likely to have had an atypical postoperative course and are therefore followed for longer than 6 months. Those who do not elect for removal are typically discharged from care after their 3-month postoperative visit, and were therefore not eligible for inclusion in this study. Overall, a total of 261 patients have been treated with this technique by the senior surgeon. Of those patients, only 28 (10.7%) underwent removal of surgical implants. If the remaining patients had been followed for the full 6 months, it is likely that outcome scores would have been skewed in a more favorable direction.
Surgeons electing to utilize radial styloid plating for displaced distal radius fractures should recognize that the required increased surgical dissection might lead to increased scar formation and postoperative stiffness. A limitation of this study is the lack of quantitative wrist ROM data. Future studies may compare final clinical outcomes and ROM for patients treated with and without radial column fixation.
CONCLUSION
We advocate for the use of a radial column plate as a tool to help achieve and maintain fracture reduction in the setting of an unstable distal radius fracture being treated with ORIF. This technique may be particularly useful when a surgical assistant is not available. Surgeons can expect clinical and radiographic results that are similar to those of volar locked plating alone.
1. Larsen CF, Lauritsen J. Epidemiology of acute wrist trauma. Int J Epidemiol. 1993;22(5):911-916.
2. Chung KC, Spilson SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908-915. doi:10.1053/jhsu.2001.26322.
3. Melton LJ 3rd, Amadio PC, Crowson CS, O'Fallon WM. Long-term trends in the incidence of distal forearm fractures. Osteoporos Int. 1998;8(4):341-348.
4. Hagino H, Yamamoto K, Ohshiro H, Nakamura T, Kishimoto H, Nose T. Changing incidence of hip, distal radius, and proximal humerus fractures in Tottori Prefecture, Japan. Bone. 1999;24(3):265-270.
5. Diaz-Garcia RJ, Chung KC. The evolution of distal radius fracture management: A historical treatise. Hand Clin. 2012;28(2):105-111. doi:10.1016/j.hcl.2012.02.007.
6. McQueen M, Caspers J. Colles fracture: Does the anatomical result affect the final function? J Bone Joint Surg Br. 1988;70(4):649-651.
7. Stewart HD, Innes AR, Burke FD. Factors affecting the outcome of Colles' fracture: an anatomical and functional study. Injury. 1985;16(5):289-295.
8. Knight D, Hajducka C, Will E, McQueen M. Locked volar plating for unstable distal radial fractures: Clinical and radiological outcomes. Injury. 2010;41(2):184-189. doi:10.1016/j.injury.2009.08.024.
9. Anakwe R, Khan L, Cook R, McEachan J. Locked volar plating for complex distal radius fractures: patient reported outcomes and satisfaction. J Orthop Surg Res. 2010;5:51. doi:10.1186/1749799X-5-51.
10. Gruber G, Gruber K, Giessauf C, et al. Volar plate fixation of AO type C2 and C3 distal radius fractures, a single-center study of 55 patients. J Orthop Trauma. 2008;22(7):467-472. doi:10.1097/BOT.0b013e318180db09.
11. Koval KJ, Harrast JJ, Anglen JO, Weinstein JN. Fractures of the distal part of the radius. The evolution of practice over time. Where’s the evidence? J Bone Joint Surg Am. 2008;90(9):1855-1861. doi:10.2106/JBJS.G.01569.
12. Foo TL, Gan AW, Soh T, Chew WY. Mechanical failure of the distal radius volar locking plate. J Orthop Surg (Hong Kong). 2013;21(3):332-336. doi:10.11777/230949901302100314.
13. Ward CM, Kuhl TL, Adams BD. Early complications of volar plating of distal radius fractures and their relationship to surgeon experience. Hand (N Y). 2011;6(2):185-189. doi:10.1007/s11552-010-9313-5.
14. Min W, Kaplan K, Miyamoto R, Tejwani NC. A unique failure mechanism of a distal radius fracture fixed with volar plating--a case report. Bull NYU Hosp Jt Dis. 2010;68(4):304-306.
15. Cao J, Ozer K. Failure of volar locking plate fixation of an extraarticular distal radius fracture: A case report. Patient Saf Surg. 2010;4(1):19. doi:10.1186/1754-9493-4-19.
16. Tang P, Ding A, Uzumcugil A. Radial column and volar plating (RCVP) for distal radius fractures with a radial styloid component or severe comminution. Tech Hand Up Extrem Surg. 2010;14(3):143-149. doi:10.1097/BTH.0b013e3181cae14d.
17. Jacobi M, Wahl P, Kohut G. Repositioning and stabilization of the radial styloid process in comminuted fractures of the distal radius using a single approach: The radio-volar double plating technique. J Orthop Surg Res. 2010;5:55. doi:10.1186/1749-799X-5-55.
18. Rikli DA, Regazzoni P. The double plating technique for distal radius fractures. Tech Hand Up Extrem Surg. 2000;4(2):107-114.
19. Rikli DA, Regazzoni P. Fractures of the distal end of the radius treated by internal fixation and early function. A preliminary report of 20 cases. J Bone Joint Surg Br. 1996;78(4):588-592.
20. Grindel SI, Wang M, Gerlach M, McGrady LM, Brown S. Biomechanical comparison of fixed-angle volar plate versus fixed-angle volar plate plus fragment-specific fixation in a cadaveric distal radius fracture model. J Hand Surg Am. 2007;32(2):194-199. doi:10.1016/j.jhsa.2006.12.003.
21. Blythe M, Stoffel K, Jarrett P, Kuster M. Volar versus dorsal locking plates with and without radial styloid locking plates for the fixation of dorsally comminuted distal radius fractures: A biomechanical study in cadavers. J Hand Surg Am. 2006;31(10):1587-1593. doi:10.1016/j.jhsa.2006.09.011.
22. Loveridge J, Ahearn N, Gee C, Pearson D, Sivaloganathan S, Bhatia R. Treatment of distal radial fractures with the DVR-A plate--the early bristol experience. Hand Surg. 2013;18(2):159-167. doi:10.1142/S0218810413500184.
23. Karantana A, Downing ND, Forward DP, et al. Surgical treatment of distal radial fractures with a volar locking plate versus conventional percutaneous methods: a randomized controlled trial. J Bone Joint Surg Am. 2013;95(19):1737-1744. doi:10.2106/JBJS.L.00232.
24. Egol K, Walsh M, Tejwani N, McLaurin T, Wynn C, Paksima N. Bridging external fixation and supplementary kirschner-wire fixation versus volar locked plating for unstable fractures of the distal radius: A randomised, prospective trial. J Bone Joint Surg Br. 2008;90(9):1214-1221. doi:10.1302/0301-620X.90B9.20521.
25. von Recum J, Matschke S, Jupiter JB, et al. Characteristics of two different locking compression plates in the volar fixation of complex articular distal radius fractures. Bone Joint Res. 2012;1(6):111-117. doi:10.1302/2046-3758.16.2000008.
26. Safi A, Hart R, Těknědžjan B, Kozák T. Treatment of extra-articular and simple articular distal radial fractures with intramedullary nail versus volar locking plate. J Hand Surg Eur Vol. 2013;38(7):774-779. doi:10.1177/1753193413478715.
27. Kim JK, Park SD. Outcomes after volar plate fixation of low-grade open and closed distal radius fractures are similar. Clin Orthop Relat Res. 2013;471(6):2030-2035. doi:10.1007/s11999-013-2798-9.
1. Larsen CF, Lauritsen J. Epidemiology of acute wrist trauma. Int J Epidemiol. 1993;22(5):911-916.
2. Chung KC, Spilson SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908-915. doi:10.1053/jhsu.2001.26322.
3. Melton LJ 3rd, Amadio PC, Crowson CS, O'Fallon WM. Long-term trends in the incidence of distal forearm fractures. Osteoporos Int. 1998;8(4):341-348.
4. Hagino H, Yamamoto K, Ohshiro H, Nakamura T, Kishimoto H, Nose T. Changing incidence of hip, distal radius, and proximal humerus fractures in Tottori Prefecture, Japan. Bone. 1999;24(3):265-270.
5. Diaz-Garcia RJ, Chung KC. The evolution of distal radius fracture management: A historical treatise. Hand Clin. 2012;28(2):105-111. doi:10.1016/j.hcl.2012.02.007.
6. McQueen M, Caspers J. Colles fracture: Does the anatomical result affect the final function? J Bone Joint Surg Br. 1988;70(4):649-651.
7. Stewart HD, Innes AR, Burke FD. Factors affecting the outcome of Colles' fracture: an anatomical and functional study. Injury. 1985;16(5):289-295.
8. Knight D, Hajducka C, Will E, McQueen M. Locked volar plating for unstable distal radial fractures: Clinical and radiological outcomes. Injury. 2010;41(2):184-189. doi:10.1016/j.injury.2009.08.024.
9. Anakwe R, Khan L, Cook R, McEachan J. Locked volar plating for complex distal radius fractures: patient reported outcomes and satisfaction. J Orthop Surg Res. 2010;5:51. doi:10.1186/1749799X-5-51.
10. Gruber G, Gruber K, Giessauf C, et al. Volar plate fixation of AO type C2 and C3 distal radius fractures, a single-center study of 55 patients. J Orthop Trauma. 2008;22(7):467-472. doi:10.1097/BOT.0b013e318180db09.
11. Koval KJ, Harrast JJ, Anglen JO, Weinstein JN. Fractures of the distal part of the radius. The evolution of practice over time. Where’s the evidence? J Bone Joint Surg Am. 2008;90(9):1855-1861. doi:10.2106/JBJS.G.01569.
12. Foo TL, Gan AW, Soh T, Chew WY. Mechanical failure of the distal radius volar locking plate. J Orthop Surg (Hong Kong). 2013;21(3):332-336. doi:10.11777/230949901302100314.
13. Ward CM, Kuhl TL, Adams BD. Early complications of volar plating of distal radius fractures and their relationship to surgeon experience. Hand (N Y). 2011;6(2):185-189. doi:10.1007/s11552-010-9313-5.
14. Min W, Kaplan K, Miyamoto R, Tejwani NC. A unique failure mechanism of a distal radius fracture fixed with volar plating--a case report. Bull NYU Hosp Jt Dis. 2010;68(4):304-306.
15. Cao J, Ozer K. Failure of volar locking plate fixation of an extraarticular distal radius fracture: A case report. Patient Saf Surg. 2010;4(1):19. doi:10.1186/1754-9493-4-19.
16. Tang P, Ding A, Uzumcugil A. Radial column and volar plating (RCVP) for distal radius fractures with a radial styloid component or severe comminution. Tech Hand Up Extrem Surg. 2010;14(3):143-149. doi:10.1097/BTH.0b013e3181cae14d.
17. Jacobi M, Wahl P, Kohut G. Repositioning and stabilization of the radial styloid process in comminuted fractures of the distal radius using a single approach: The radio-volar double plating technique. J Orthop Surg Res. 2010;5:55. doi:10.1186/1749-799X-5-55.
18. Rikli DA, Regazzoni P. The double plating technique for distal radius fractures. Tech Hand Up Extrem Surg. 2000;4(2):107-114.
19. Rikli DA, Regazzoni P. Fractures of the distal end of the radius treated by internal fixation and early function. A preliminary report of 20 cases. J Bone Joint Surg Br. 1996;78(4):588-592.
20. Grindel SI, Wang M, Gerlach M, McGrady LM, Brown S. Biomechanical comparison of fixed-angle volar plate versus fixed-angle volar plate plus fragment-specific fixation in a cadaveric distal radius fracture model. J Hand Surg Am. 2007;32(2):194-199. doi:10.1016/j.jhsa.2006.12.003.
21. Blythe M, Stoffel K, Jarrett P, Kuster M. Volar versus dorsal locking plates with and without radial styloid locking plates for the fixation of dorsally comminuted distal radius fractures: A biomechanical study in cadavers. J Hand Surg Am. 2006;31(10):1587-1593. doi:10.1016/j.jhsa.2006.09.011.
22. Loveridge J, Ahearn N, Gee C, Pearson D, Sivaloganathan S, Bhatia R. Treatment of distal radial fractures with the DVR-A plate--the early bristol experience. Hand Surg. 2013;18(2):159-167. doi:10.1142/S0218810413500184.
23. Karantana A, Downing ND, Forward DP, et al. Surgical treatment of distal radial fractures with a volar locking plate versus conventional percutaneous methods: a randomized controlled trial. J Bone Joint Surg Am. 2013;95(19):1737-1744. doi:10.2106/JBJS.L.00232.
24. Egol K, Walsh M, Tejwani N, McLaurin T, Wynn C, Paksima N. Bridging external fixation and supplementary kirschner-wire fixation versus volar locked plating for unstable fractures of the distal radius: A randomised, prospective trial. J Bone Joint Surg Br. 2008;90(9):1214-1221. doi:10.1302/0301-620X.90B9.20521.
25. von Recum J, Matschke S, Jupiter JB, et al. Characteristics of two different locking compression plates in the volar fixation of complex articular distal radius fractures. Bone Joint Res. 2012;1(6):111-117. doi:10.1302/2046-3758.16.2000008.
26. Safi A, Hart R, Těknědžjan B, Kozák T. Treatment of extra-articular and simple articular distal radial fractures with intramedullary nail versus volar locking plate. J Hand Surg Eur Vol. 2013;38(7):774-779. doi:10.1177/1753193413478715.
27. Kim JK, Park SD. Outcomes after volar plate fixation of low-grade open and closed distal radius fractures are similar. Clin Orthop Relat Res. 2013;471(6):2030-2035. doi:10.1007/s11999-013-2798-9.
TAKE-HOME POINTS
- Radial column fixation can be used as a reduction tool in unstable distal radius fractures.
- Radial column fixation can help maintain reduction until union in unstable distal radius fractures when combined with volar plating.
- When operating without an assistant, radial column plating can assist in reduction maintenance when other techniques are not successful and holding a reduction manually is not possible.
- Acceptable clinical and radiographic outcomes can be achieved with the use of dual radial styloid and volar plating for unstable distal radius fractures.
- The effects of increased dissection during radial column fixation in distal radius fractures with regard to scar formation and wrist ROM is currently not known.
Many VTE patients live in fear of the next event
SAN DIEGO – An estimated 41% of patients who experienced a venous thromboembolism (VTE) fear another clot often or almost all the time. In addition, about 25% report abnormal levels of anxiety, and 12% have abnormal depression scores.
Those are key findings from a large survey that set out to estimate the number of bleeding harms and emotional harms experienced by a U.S. population of adults who have experienced a VTE.
“There is emerging research in Europe that shows high levels of stress and anxiety in people who have a thrombosis event,” lead study author Michael Feehan, PhD, said in an interview at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “We interviewed people around the country and found that a lot of them were living with fear, anxiety, and distress. We did a projective exercise and asked, ‘If VTE was an animal, what would it be?’ Many responded with snakes and bears, hostile things. Snakes came up a lot. Snakes can be dormant, and then they can suddenly come out and bite you. That was the kind of language they were using.”
In what he said is the largest study of its kind, Dr. Feehan, a psychologist in the College of Pharmacy at the University of Utah, Salt Lake City, and his associates conducted an online survey of 907 patients aged 18 and older who had experienced a VTE event in the previous 24 months.
The survey was administered in May 2016 and excluded patients with cancer-related VTE. It took about 30 minutes to complete and included questions about the bleeding harms that have occurred since their VTE diagnosis, such as nosebleeds or a cut difficult to control, excessive bruising, vomiting blood, bloody urine, and blood in stools. It also included standardized measures of anxiety, depression, cognitive function, and ratings on eight items of current distress, feeling tense, anxious, confused, depressed, afraid, angry, frustrated, or annoyed.
Self-reported bleeding events included excessive bruising (45%), bleeding from cuts difficult to control (33%), and epistaxis (16%). As for emotional harm, 41% of respondents lived in fear of getting another clot “often” or “almost all the time,” while 25% experienced abnormally high levels of anxiety, and 12% experienced abnormally high levels of depression.
A multivariate structural equation model revealed the following principal factors significantly associated with a composite latent variable of emotional harm: poor health literacy, younger age, the lack of perceived self-control over one’s health, history of medical mistakes in care, and overt barriers to health care access such as transportation limitations and financial limitations (P less than .05 for all associations).
“If you’re working with patients who believe they don’t have any control over their own care, or if they’re younger or have other disease states, or if they have difficulty getting to and from the hospital, all of those things contribute to elevated emotional harms,” Dr. Feehan said. “That level of emotional harm is clinically relevant.”
After the research team shared the study results with staff of the university’s thrombosis services, clinicians started changing how they interview patients. “For example, instead of asking just ‘Have you experienced any VTE symptoms?’ they now ask things like, ‘How are you feeling?’ or ‘How are things going for you living with the disease?’” Dr. Feehan noted. “Then, patients might say, ‘I’m actually quite worried.’ Such questions can help patients open up about how they feel and foster a better relationship with their provider. A better relationship with their provider might help them feel more in control.”
The study was supported by Pfizer Independent Grants for Learning & Change, Bristol-Myers Squibb Independent Medical Education, the Joint Commission, the National Eye Institute, and an unrestricted grant from Research to Prevent Blindness. Dr. Feehan disclosed that he has consulted for Pfizer in the past.
SOURCE: Feehan et al. THSNA 2018, Poster 75.
SAN DIEGO – An estimated 41% of patients who experienced a venous thromboembolism (VTE) fear another clot often or almost all the time. In addition, about 25% report abnormal levels of anxiety, and 12% have abnormal depression scores.
Those are key findings from a large survey that set out to estimate the number of bleeding harms and emotional harms experienced by a U.S. population of adults who have experienced a VTE.
“There is emerging research in Europe that shows high levels of stress and anxiety in people who have a thrombosis event,” lead study author Michael Feehan, PhD, said in an interview at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “We interviewed people around the country and found that a lot of them were living with fear, anxiety, and distress. We did a projective exercise and asked, ‘If VTE was an animal, what would it be?’ Many responded with snakes and bears, hostile things. Snakes came up a lot. Snakes can be dormant, and then they can suddenly come out and bite you. That was the kind of language they were using.”
In what he said is the largest study of its kind, Dr. Feehan, a psychologist in the College of Pharmacy at the University of Utah, Salt Lake City, and his associates conducted an online survey of 907 patients aged 18 and older who had experienced a VTE event in the previous 24 months.
The survey was administered in May 2016 and excluded patients with cancer-related VTE. It took about 30 minutes to complete and included questions about the bleeding harms that have occurred since their VTE diagnosis, such as nosebleeds or a cut difficult to control, excessive bruising, vomiting blood, bloody urine, and blood in stools. It also included standardized measures of anxiety, depression, cognitive function, and ratings on eight items of current distress, feeling tense, anxious, confused, depressed, afraid, angry, frustrated, or annoyed.
Self-reported bleeding events included excessive bruising (45%), bleeding from cuts difficult to control (33%), and epistaxis (16%). As for emotional harm, 41% of respondents lived in fear of getting another clot “often” or “almost all the time,” while 25% experienced abnormally high levels of anxiety, and 12% experienced abnormally high levels of depression.
A multivariate structural equation model revealed the following principal factors significantly associated with a composite latent variable of emotional harm: poor health literacy, younger age, the lack of perceived self-control over one’s health, history of medical mistakes in care, and overt barriers to health care access such as transportation limitations and financial limitations (P less than .05 for all associations).
“If you’re working with patients who believe they don’t have any control over their own care, or if they’re younger or have other disease states, or if they have difficulty getting to and from the hospital, all of those things contribute to elevated emotional harms,” Dr. Feehan said. “That level of emotional harm is clinically relevant.”
After the research team shared the study results with staff of the university’s thrombosis services, clinicians started changing how they interview patients. “For example, instead of asking just ‘Have you experienced any VTE symptoms?’ they now ask things like, ‘How are you feeling?’ or ‘How are things going for you living with the disease?’” Dr. Feehan noted. “Then, patients might say, ‘I’m actually quite worried.’ Such questions can help patients open up about how they feel and foster a better relationship with their provider. A better relationship with their provider might help them feel more in control.”
The study was supported by Pfizer Independent Grants for Learning & Change, Bristol-Myers Squibb Independent Medical Education, the Joint Commission, the National Eye Institute, and an unrestricted grant from Research to Prevent Blindness. Dr. Feehan disclosed that he has consulted for Pfizer in the past.
SOURCE: Feehan et al. THSNA 2018, Poster 75.
SAN DIEGO – An estimated 41% of patients who experienced a venous thromboembolism (VTE) fear another clot often or almost all the time. In addition, about 25% report abnormal levels of anxiety, and 12% have abnormal depression scores.
Those are key findings from a large survey that set out to estimate the number of bleeding harms and emotional harms experienced by a U.S. population of adults who have experienced a VTE.
“There is emerging research in Europe that shows high levels of stress and anxiety in people who have a thrombosis event,” lead study author Michael Feehan, PhD, said in an interview at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “We interviewed people around the country and found that a lot of them were living with fear, anxiety, and distress. We did a projective exercise and asked, ‘If VTE was an animal, what would it be?’ Many responded with snakes and bears, hostile things. Snakes came up a lot. Snakes can be dormant, and then they can suddenly come out and bite you. That was the kind of language they were using.”
In what he said is the largest study of its kind, Dr. Feehan, a psychologist in the College of Pharmacy at the University of Utah, Salt Lake City, and his associates conducted an online survey of 907 patients aged 18 and older who had experienced a VTE event in the previous 24 months.
The survey was administered in May 2016 and excluded patients with cancer-related VTE. It took about 30 minutes to complete and included questions about the bleeding harms that have occurred since their VTE diagnosis, such as nosebleeds or a cut difficult to control, excessive bruising, vomiting blood, bloody urine, and blood in stools. It also included standardized measures of anxiety, depression, cognitive function, and ratings on eight items of current distress, feeling tense, anxious, confused, depressed, afraid, angry, frustrated, or annoyed.
Self-reported bleeding events included excessive bruising (45%), bleeding from cuts difficult to control (33%), and epistaxis (16%). As for emotional harm, 41% of respondents lived in fear of getting another clot “often” or “almost all the time,” while 25% experienced abnormally high levels of anxiety, and 12% experienced abnormally high levels of depression.
A multivariate structural equation model revealed the following principal factors significantly associated with a composite latent variable of emotional harm: poor health literacy, younger age, the lack of perceived self-control over one’s health, history of medical mistakes in care, and overt barriers to health care access such as transportation limitations and financial limitations (P less than .05 for all associations).
“If you’re working with patients who believe they don’t have any control over their own care, or if they’re younger or have other disease states, or if they have difficulty getting to and from the hospital, all of those things contribute to elevated emotional harms,” Dr. Feehan said. “That level of emotional harm is clinically relevant.”
After the research team shared the study results with staff of the university’s thrombosis services, clinicians started changing how they interview patients. “For example, instead of asking just ‘Have you experienced any VTE symptoms?’ they now ask things like, ‘How are you feeling?’ or ‘How are things going for you living with the disease?’” Dr. Feehan noted. “Then, patients might say, ‘I’m actually quite worried.’ Such questions can help patients open up about how they feel and foster a better relationship with their provider. A better relationship with their provider might help them feel more in control.”
The study was supported by Pfizer Independent Grants for Learning & Change, Bristol-Myers Squibb Independent Medical Education, the Joint Commission, the National Eye Institute, and an unrestricted grant from Research to Prevent Blindness. Dr. Feehan disclosed that he has consulted for Pfizer in the past.
SOURCE: Feehan et al. THSNA 2018, Poster 75.
REPORTING FROM THSNA 2018
Key clinical point: Consider asking VTE patients how living with their disease affects them from an emotional standpoint.
Major finding: Of 907 survey respondents, 41% said they live in fear of getting another clot “often” or “almost all the time.”
Study details: An online survey of 907 patients aged 18 and older who had experienced a VTE event in the previous 24 months.
Disclosures: The study was supported by Pfizer Independent Grants for Learning & Change, Bristol-Myers Squibb Independent Medical Education, the Joint Commission, the National Eye Institute, and an unrestricted grant from Research to Prevent Blindness. Dr. Feehan disclosed that he has consulted for Pfizer in the past.
Source: Feehan M et al. THSNA 2018, Poster 75.
Ranking points physicians toward South Dakota
South Dakota’s nickname may be the Mount Rushmore State, but for physicians it’s the land of opportunity, according to personal finance website WalletHub.
South Dakota took the top spot in the 2018 list of the “best states to practice medicine” with 75.97 out of a possible 100 points while New Jersey took up residence at the other end of the list by finishing 51st (the District of Columbia was included) with a score of 40.24, WalletHub reported.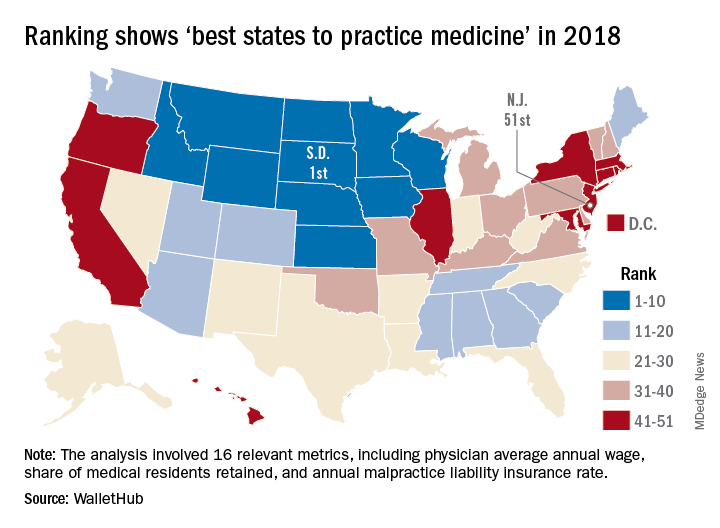
South Dakota headed a solid block of states in the upper Midwest and Rocky Mountain regions that made up the entire top 10, with Nebraska second overall, Idaho third, Iowa (last year’s winner) fourth, and Minnesota fifth. Finishing just above New Jersey were Rhode Island in 50th, New York in 49th, Hawaii in 48th, and D.C. in 47th.
WalletHub compared the 50 states and D.C. using 16 different metrics across two broad categories: “opportunity and competition” (11 metrics worth 70 points) and “medical environment” (5 metrics worth 30 points). Metrics included physicians’ average annual wage (adjusted for cost of living), employer-based insurance rates, projected share of elderly population, physician assistants per capita, and malpractice award payout amount per capita.
South Dakota, which ranked first in the medical environment category and third in opportunity and competition, posted top-5 scores in such areas as adjusted physician annual wage, lowest projected competition, and annual malpractice liability insurance rate, according to the survey.
South Dakota’s nickname may be the Mount Rushmore State, but for physicians it’s the land of opportunity, according to personal finance website WalletHub.
South Dakota took the top spot in the 2018 list of the “best states to practice medicine” with 75.97 out of a possible 100 points while New Jersey took up residence at the other end of the list by finishing 51st (the District of Columbia was included) with a score of 40.24, WalletHub reported.
South Dakota headed a solid block of states in the upper Midwest and Rocky Mountain regions that made up the entire top 10, with Nebraska second overall, Idaho third, Iowa (last year’s winner) fourth, and Minnesota fifth. Finishing just above New Jersey were Rhode Island in 50th, New York in 49th, Hawaii in 48th, and D.C. in 47th.
WalletHub compared the 50 states and D.C. using 16 different metrics across two broad categories: “opportunity and competition” (11 metrics worth 70 points) and “medical environment” (5 metrics worth 30 points). Metrics included physicians’ average annual wage (adjusted for cost of living), employer-based insurance rates, projected share of elderly population, physician assistants per capita, and malpractice award payout amount per capita.
South Dakota, which ranked first in the medical environment category and third in opportunity and competition, posted top-5 scores in such areas as adjusted physician annual wage, lowest projected competition, and annual malpractice liability insurance rate, according to the survey.
South Dakota’s nickname may be the Mount Rushmore State, but for physicians it’s the land of opportunity, according to personal finance website WalletHub.
South Dakota took the top spot in the 2018 list of the “best states to practice medicine” with 75.97 out of a possible 100 points while New Jersey took up residence at the other end of the list by finishing 51st (the District of Columbia was included) with a score of 40.24, WalletHub reported.
South Dakota headed a solid block of states in the upper Midwest and Rocky Mountain regions that made up the entire top 10, with Nebraska second overall, Idaho third, Iowa (last year’s winner) fourth, and Minnesota fifth. Finishing just above New Jersey were Rhode Island in 50th, New York in 49th, Hawaii in 48th, and D.C. in 47th.
WalletHub compared the 50 states and D.C. using 16 different metrics across two broad categories: “opportunity and competition” (11 metrics worth 70 points) and “medical environment” (5 metrics worth 30 points). Metrics included physicians’ average annual wage (adjusted for cost of living), employer-based insurance rates, projected share of elderly population, physician assistants per capita, and malpractice award payout amount per capita.
South Dakota, which ranked first in the medical environment category and third in opportunity and competition, posted top-5 scores in such areas as adjusted physician annual wage, lowest projected competition, and annual malpractice liability insurance rate, according to the survey.
Cenk Ayata, MD, & Messoud Ashina, MD



Bridget Mueller, MD, PhD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Paul Rizzoli, MD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Christopher Gottschalk, MD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Are higher rifampin doses for pulmonary TB more effective?
BOSTON – Current daily doses of rifampin for treating pulmonary tuberculosis may be too low and could be safely increased, results of a randomized phase 2 study suggest.
“Back in the 1970s, rifampin was an expensive drug, and attempts to shorten TB therapy using higher but intermittent doses of rifampin were unsuccessful at that time because of increased toxicity. That line of inquiry was essentially dormant for 40 years,” said Gustavo Velásquez, MD, from Brigham & Women’s Hospital in Boston.
To get a better idea of optimal rifampin dosing for the treatment of pulmonary TB, Dr. Velásquez and his colleagues conducted the HIRIF (High-Dose Rifampin in Patients With TB) trial. The phase 2 study was designed to evaluate the pharmacokinetics, efficacy, and safety of higher daily rifampin doses for pulmonary TB.
They looked at the three parameters across three treatments arms: 10 mg/kg rifampin (the current standard of care), 15 mg/kg, or 20 mg/kg.
Patients in Peru were screened, enrolled, and randomized in cohorts of 60 patients each to one of the three specified dose levels, which they received either as additional rifampin tablets or placebo for the first 8 weeks of treatment, after which all patients were continued on rifampin 10 mg/kg to complete a 6-month regimen. All patients were followed for an additional 6 months to for assessment of TB recurrence.
Rifampin total doses ranged from as low as 300 mg for patients in the 30 kg-37 kg weight range, to as high as 1,500 mg for those weighing more than 70 kg.
The efficacy analysis was by modified intention to treat, excluding 6 patients who had insufficient log10 colony-forming units (CFUs) of TB, and a per-protocol analysis excluding an additional 42 patients whose doses of rifampin were affected by three study halts for adverse events. After each halt and review by the data-safety monitoring board, the trial was allowed to resume, but because enrollment and experimental dosing also were suspended, patients in the 15- and 20-mg/kg arms received 10 mg/kg during the 2-5 week halts. The number of patients in the 10-, 15-, and 20-mg/kg doses included in the per-protocol analysis were 56, 38, and 38, respectively,
Pharmacokinetic evidence from this study, previously published, showed that the median maximum drug concentration (Cmax) in serum in the experimental arms reached the lower end of the targeted range of 8 mcg/mL or greater, whereas the median in the standard-of-care arm was 6.2 mcg/mL. Only 33% of patients in the 10-mg/kg arm reached the minimum 8-mcg/mL level, Dr. Velásquez noted, vs. 72% and 81% of patients in the 15- and 20-mg/kg doses, respectively.
In the modified intention-to-treat population, for every 5-mg/kg increase in rifampin dose, there was a nonsignificant trend toward faster decline in TB CFUs in sputum. Similarly, for every 1-log increase in rifampin AUC/MIC, there was a trend, albeit nonsignificant, toward faster decline.
However, in patients in the per-protocol analysis, every 5-mg/kg dose increase and 1-log increase in rifampin AUC was associated with significantly faster declines in CFUs (P = .022 and .011, respectively).
An analysis of treatment outcomes at 12 months, a secondary endpoint, showed that there were five cases of treatment failure, including three in the control arm and one each in 15- and 20-mg/kg arms, and six cases of recurrence after cure, which occurred in three, one, and two patients, respectively,
The safety analysis by intention-to-treat showed that the incidence of grade 2 or greater rifampin-related adverse events (AEs) were 43.3%, 51.7%, and 38.3% in the 10-, 15-, and 20-mg/kg doses, differences that were not statistically significant.
In addition, there were no significant differences among the treatment arms in either time to first grade 2 or greater rifampin-related AEs, the occurrence of one or more grade 2 or greater hepatic rifampin-AEs, or time to first hepatic rifampin-related AEs of grade 2 or above.
Dr. Velásquez noted that the study was limited by the possibility that the study halts could have biased efficacy effect estimates toward null and by differences in weight distribution among the three treatment arms.
“This actually is the first trial that shows not only a dose response of rifampin but also an exposure response of rifampin in combination therapy,” he said. “Our study supports that even higher doses of rifampin beyond what we studied of 20 mg/kg should be studied for potential treatment shortening.” The evidence also suggests that the current 10-mg/kg dose is low and could be safely increased to a 15- or 20-mg/kg dose, he concluded.
In a media briefing following the presentation, moderator Constance Benson, MD, from the University of California San Diego, who was not involved in the study, commented that with “high-dose rifampin, I think we have a really very robust body of literature to which this study can be added, demonstrating the safety of high-dose rifampin in the context of TB treatment.”
“There are some circumstances where I think using a much higher dose than we’ve been using would be an appropriate thing to do,” she added.
Examples of patients who might benefit include patients with disseminated TB or people with more serious TB than the average case, she said.
The study was supported by the National Institute of Allergy and Infectious Diseases. Dr. Velásquez and Dr. Benson reported no relevant conflicts of interest.
SOURCE: Velásquez GE et al. CROI 2018, Abstract 39LB.
BOSTON – Current daily doses of rifampin for treating pulmonary tuberculosis may be too low and could be safely increased, results of a randomized phase 2 study suggest.
“Back in the 1970s, rifampin was an expensive drug, and attempts to shorten TB therapy using higher but intermittent doses of rifampin were unsuccessful at that time because of increased toxicity. That line of inquiry was essentially dormant for 40 years,” said Gustavo Velásquez, MD, from Brigham & Women’s Hospital in Boston.
To get a better idea of optimal rifampin dosing for the treatment of pulmonary TB, Dr. Velásquez and his colleagues conducted the HIRIF (High-Dose Rifampin in Patients With TB) trial. The phase 2 study was designed to evaluate the pharmacokinetics, efficacy, and safety of higher daily rifampin doses for pulmonary TB.
They looked at the three parameters across three treatments arms: 10 mg/kg rifampin (the current standard of care), 15 mg/kg, or 20 mg/kg.
Patients in Peru were screened, enrolled, and randomized in cohorts of 60 patients each to one of the three specified dose levels, which they received either as additional rifampin tablets or placebo for the first 8 weeks of treatment, after which all patients were continued on rifampin 10 mg/kg to complete a 6-month regimen. All patients were followed for an additional 6 months to for assessment of TB recurrence.
Rifampin total doses ranged from as low as 300 mg for patients in the 30 kg-37 kg weight range, to as high as 1,500 mg for those weighing more than 70 kg.
The efficacy analysis was by modified intention to treat, excluding 6 patients who had insufficient log10 colony-forming units (CFUs) of TB, and a per-protocol analysis excluding an additional 42 patients whose doses of rifampin were affected by three study halts for adverse events. After each halt and review by the data-safety monitoring board, the trial was allowed to resume, but because enrollment and experimental dosing also were suspended, patients in the 15- and 20-mg/kg arms received 10 mg/kg during the 2-5 week halts. The number of patients in the 10-, 15-, and 20-mg/kg doses included in the per-protocol analysis were 56, 38, and 38, respectively,
Pharmacokinetic evidence from this study, previously published, showed that the median maximum drug concentration (Cmax) in serum in the experimental arms reached the lower end of the targeted range of 8 mcg/mL or greater, whereas the median in the standard-of-care arm was 6.2 mcg/mL. Only 33% of patients in the 10-mg/kg arm reached the minimum 8-mcg/mL level, Dr. Velásquez noted, vs. 72% and 81% of patients in the 15- and 20-mg/kg doses, respectively.
In the modified intention-to-treat population, for every 5-mg/kg increase in rifampin dose, there was a nonsignificant trend toward faster decline in TB CFUs in sputum. Similarly, for every 1-log increase in rifampin AUC/MIC, there was a trend, albeit nonsignificant, toward faster decline.
However, in patients in the per-protocol analysis, every 5-mg/kg dose increase and 1-log increase in rifampin AUC was associated with significantly faster declines in CFUs (P = .022 and .011, respectively).
An analysis of treatment outcomes at 12 months, a secondary endpoint, showed that there were five cases of treatment failure, including three in the control arm and one each in 15- and 20-mg/kg arms, and six cases of recurrence after cure, which occurred in three, one, and two patients, respectively,
The safety analysis by intention-to-treat showed that the incidence of grade 2 or greater rifampin-related adverse events (AEs) were 43.3%, 51.7%, and 38.3% in the 10-, 15-, and 20-mg/kg doses, differences that were not statistically significant.
In addition, there were no significant differences among the treatment arms in either time to first grade 2 or greater rifampin-related AEs, the occurrence of one or more grade 2 or greater hepatic rifampin-AEs, or time to first hepatic rifampin-related AEs of grade 2 or above.
Dr. Velásquez noted that the study was limited by the possibility that the study halts could have biased efficacy effect estimates toward null and by differences in weight distribution among the three treatment arms.
“This actually is the first trial that shows not only a dose response of rifampin but also an exposure response of rifampin in combination therapy,” he said. “Our study supports that even higher doses of rifampin beyond what we studied of 20 mg/kg should be studied for potential treatment shortening.” The evidence also suggests that the current 10-mg/kg dose is low and could be safely increased to a 15- or 20-mg/kg dose, he concluded.
In a media briefing following the presentation, moderator Constance Benson, MD, from the University of California San Diego, who was not involved in the study, commented that with “high-dose rifampin, I think we have a really very robust body of literature to which this study can be added, demonstrating the safety of high-dose rifampin in the context of TB treatment.”
“There are some circumstances where I think using a much higher dose than we’ve been using would be an appropriate thing to do,” she added.
Examples of patients who might benefit include patients with disseminated TB or people with more serious TB than the average case, she said.
The study was supported by the National Institute of Allergy and Infectious Diseases. Dr. Velásquez and Dr. Benson reported no relevant conflicts of interest.
SOURCE: Velásquez GE et al. CROI 2018, Abstract 39LB.
BOSTON – Current daily doses of rifampin for treating pulmonary tuberculosis may be too low and could be safely increased, results of a randomized phase 2 study suggest.
“Back in the 1970s, rifampin was an expensive drug, and attempts to shorten TB therapy using higher but intermittent doses of rifampin were unsuccessful at that time because of increased toxicity. That line of inquiry was essentially dormant for 40 years,” said Gustavo Velásquez, MD, from Brigham & Women’s Hospital in Boston.
To get a better idea of optimal rifampin dosing for the treatment of pulmonary TB, Dr. Velásquez and his colleagues conducted the HIRIF (High-Dose Rifampin in Patients With TB) trial. The phase 2 study was designed to evaluate the pharmacokinetics, efficacy, and safety of higher daily rifampin doses for pulmonary TB.
They looked at the three parameters across three treatments arms: 10 mg/kg rifampin (the current standard of care), 15 mg/kg, or 20 mg/kg.
Patients in Peru were screened, enrolled, and randomized in cohorts of 60 patients each to one of the three specified dose levels, which they received either as additional rifampin tablets or placebo for the first 8 weeks of treatment, after which all patients were continued on rifampin 10 mg/kg to complete a 6-month regimen. All patients were followed for an additional 6 months to for assessment of TB recurrence.
Rifampin total doses ranged from as low as 300 mg for patients in the 30 kg-37 kg weight range, to as high as 1,500 mg for those weighing more than 70 kg.
The efficacy analysis was by modified intention to treat, excluding 6 patients who had insufficient log10 colony-forming units (CFUs) of TB, and a per-protocol analysis excluding an additional 42 patients whose doses of rifampin were affected by three study halts for adverse events. After each halt and review by the data-safety monitoring board, the trial was allowed to resume, but because enrollment and experimental dosing also were suspended, patients in the 15- and 20-mg/kg arms received 10 mg/kg during the 2-5 week halts. The number of patients in the 10-, 15-, and 20-mg/kg doses included in the per-protocol analysis were 56, 38, and 38, respectively,
Pharmacokinetic evidence from this study, previously published, showed that the median maximum drug concentration (Cmax) in serum in the experimental arms reached the lower end of the targeted range of 8 mcg/mL or greater, whereas the median in the standard-of-care arm was 6.2 mcg/mL. Only 33% of patients in the 10-mg/kg arm reached the minimum 8-mcg/mL level, Dr. Velásquez noted, vs. 72% and 81% of patients in the 15- and 20-mg/kg doses, respectively.
In the modified intention-to-treat population, for every 5-mg/kg increase in rifampin dose, there was a nonsignificant trend toward faster decline in TB CFUs in sputum. Similarly, for every 1-log increase in rifampin AUC/MIC, there was a trend, albeit nonsignificant, toward faster decline.
However, in patients in the per-protocol analysis, every 5-mg/kg dose increase and 1-log increase in rifampin AUC was associated with significantly faster declines in CFUs (P = .022 and .011, respectively).
An analysis of treatment outcomes at 12 months, a secondary endpoint, showed that there were five cases of treatment failure, including three in the control arm and one each in 15- and 20-mg/kg arms, and six cases of recurrence after cure, which occurred in three, one, and two patients, respectively,
The safety analysis by intention-to-treat showed that the incidence of grade 2 or greater rifampin-related adverse events (AEs) were 43.3%, 51.7%, and 38.3% in the 10-, 15-, and 20-mg/kg doses, differences that were not statistically significant.
In addition, there were no significant differences among the treatment arms in either time to first grade 2 or greater rifampin-related AEs, the occurrence of one or more grade 2 or greater hepatic rifampin-AEs, or time to first hepatic rifampin-related AEs of grade 2 or above.
Dr. Velásquez noted that the study was limited by the possibility that the study halts could have biased efficacy effect estimates toward null and by differences in weight distribution among the three treatment arms.
“This actually is the first trial that shows not only a dose response of rifampin but also an exposure response of rifampin in combination therapy,” he said. “Our study supports that even higher doses of rifampin beyond what we studied of 20 mg/kg should be studied for potential treatment shortening.” The evidence also suggests that the current 10-mg/kg dose is low and could be safely increased to a 15- or 20-mg/kg dose, he concluded.
In a media briefing following the presentation, moderator Constance Benson, MD, from the University of California San Diego, who was not involved in the study, commented that with “high-dose rifampin, I think we have a really very robust body of literature to which this study can be added, demonstrating the safety of high-dose rifampin in the context of TB treatment.”
“There are some circumstances where I think using a much higher dose than we’ve been using would be an appropriate thing to do,” she added.
Examples of patients who might benefit include patients with disseminated TB or people with more serious TB than the average case, she said.
The study was supported by the National Institute of Allergy and Infectious Diseases. Dr. Velásquez and Dr. Benson reported no relevant conflicts of interest.
SOURCE: Velásquez GE et al. CROI 2018, Abstract 39LB.
REPORTING FROM CROI 2018
Key clinical point: Higher rifampin doses than customarily used offer better control of TB with safety, investigators say.
Major finding: In a per-protocol analysis, reduction in TB colony-forming units was significantly faster with 15- or 20-mg/kg rifampin dose vs. the standard 10-mg/kg dose.
Data source: Randomized, controlled trial in 180 Peruvian patients with pulmonary tuberculosis.
Disclosures: The study was supported by the National Institute of Allergy and Infectious Diseases. Dr. Velásquez and Dr. Benson reported no relevant conflicts of interest.
Source: Velásquez GE et al. CROI 2018, Abstract 39LB.
Bacteria seen as potential lupus triggers
A new study points to bacteria commonly found in the human microbiome as possible culprits in the quest to identify the elusive triggers of autoantibodies present in patients with systemic lupus erythematosus (SLE).
Anti-Ro antibodies, including anti-Ro60 antibodies, are found in about half of SLE patients, and are the most common antinuclear antibodies that can be seen preclinically. They are also pathogenic.
In a proof-of-concept study, a Yale-led research team collected microbiome samples from eight SLE patients who were positive for anti-Ro60 autoantibodies, five SLE patients who were anti-Ro60-negative, and seven healthy controls. The researchers took samples from the mouth, sternum, and stool. They found that commensal bacteria containing orthologs to Ro60 were found commonly in all of the patient groups. These included Propionibacterium propionicus, Corynebacterium amycolatum, Actinomyces massiliensis, and Bacteroides thetaiotaomicron, reported Teri M. Greiling, MD, PhD, of Yale University, New Haven, Conn., and her associates.
They also found that CD4 memory T-cell clones from lupus patients that were specific to Ro60 autoantigen were stimulated by Ro60-containing bacteria.
Researchers also colonized two groups of mice with B. thetaiotaomicron – mice with a lupuslike syndrome induced with the immune-response modifier imiquimod and those without this syndrome induced. The levels of anti-Ro60 antibodies were increased in those with the syndrome. Plus, lupus nephritislike complex depositions were seen in 88% of the glomeruli of the imiquimod-treated mice and in just 8% of those not treated with imiquimod.
“These data,” the researchers said, “suggest that Ro60 orthologs from human commensal bacteria may initiate and drive lupus pathogenesis.”
The study authors reported they had nothing to disclose. The work was supported by various grants from the National Institutes of Health, Arthritis National Research Foundation, the Arthritis Foundation, and the Lupus Research Institute, as well as NIH Intramural Research Programs.
SOURCE: Greiling TM et al. Sci Transl Med. 2018;10(434). doi: 10.1126/scitranslmed.aan2306.
A new study points to bacteria commonly found in the human microbiome as possible culprits in the quest to identify the elusive triggers of autoantibodies present in patients with systemic lupus erythematosus (SLE).
Anti-Ro antibodies, including anti-Ro60 antibodies, are found in about half of SLE patients, and are the most common antinuclear antibodies that can be seen preclinically. They are also pathogenic.
In a proof-of-concept study, a Yale-led research team collected microbiome samples from eight SLE patients who were positive for anti-Ro60 autoantibodies, five SLE patients who were anti-Ro60-negative, and seven healthy controls. The researchers took samples from the mouth, sternum, and stool. They found that commensal bacteria containing orthologs to Ro60 were found commonly in all of the patient groups. These included Propionibacterium propionicus, Corynebacterium amycolatum, Actinomyces massiliensis, and Bacteroides thetaiotaomicron, reported Teri M. Greiling, MD, PhD, of Yale University, New Haven, Conn., and her associates.
They also found that CD4 memory T-cell clones from lupus patients that were specific to Ro60 autoantigen were stimulated by Ro60-containing bacteria.
Researchers also colonized two groups of mice with B. thetaiotaomicron – mice with a lupuslike syndrome induced with the immune-response modifier imiquimod and those without this syndrome induced. The levels of anti-Ro60 antibodies were increased in those with the syndrome. Plus, lupus nephritislike complex depositions were seen in 88% of the glomeruli of the imiquimod-treated mice and in just 8% of those not treated with imiquimod.
“These data,” the researchers said, “suggest that Ro60 orthologs from human commensal bacteria may initiate and drive lupus pathogenesis.”
The study authors reported they had nothing to disclose. The work was supported by various grants from the National Institutes of Health, Arthritis National Research Foundation, the Arthritis Foundation, and the Lupus Research Institute, as well as NIH Intramural Research Programs.
SOURCE: Greiling TM et al. Sci Transl Med. 2018;10(434). doi: 10.1126/scitranslmed.aan2306.
A new study points to bacteria commonly found in the human microbiome as possible culprits in the quest to identify the elusive triggers of autoantibodies present in patients with systemic lupus erythematosus (SLE).
Anti-Ro antibodies, including anti-Ro60 antibodies, are found in about half of SLE patients, and are the most common antinuclear antibodies that can be seen preclinically. They are also pathogenic.
In a proof-of-concept study, a Yale-led research team collected microbiome samples from eight SLE patients who were positive for anti-Ro60 autoantibodies, five SLE patients who were anti-Ro60-negative, and seven healthy controls. The researchers took samples from the mouth, sternum, and stool. They found that commensal bacteria containing orthologs to Ro60 were found commonly in all of the patient groups. These included Propionibacterium propionicus, Corynebacterium amycolatum, Actinomyces massiliensis, and Bacteroides thetaiotaomicron, reported Teri M. Greiling, MD, PhD, of Yale University, New Haven, Conn., and her associates.
They also found that CD4 memory T-cell clones from lupus patients that were specific to Ro60 autoantigen were stimulated by Ro60-containing bacteria.
Researchers also colonized two groups of mice with B. thetaiotaomicron – mice with a lupuslike syndrome induced with the immune-response modifier imiquimod and those without this syndrome induced. The levels of anti-Ro60 antibodies were increased in those with the syndrome. Plus, lupus nephritislike complex depositions were seen in 88% of the glomeruli of the imiquimod-treated mice and in just 8% of those not treated with imiquimod.
“These data,” the researchers said, “suggest that Ro60 orthologs from human commensal bacteria may initiate and drive lupus pathogenesis.”
The study authors reported they had nothing to disclose. The work was supported by various grants from the National Institutes of Health, Arthritis National Research Foundation, the Arthritis Foundation, and the Lupus Research Institute, as well as NIH Intramural Research Programs.
SOURCE: Greiling TM et al. Sci Transl Med. 2018;10(434). doi: 10.1126/scitranslmed.aan2306.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point:
Major finding: Autoreactive human lupus CD4-positive T cells cross-reacted with Ro60 ortholog–containing commensal bacteria.
Study details: A proof-of-concept study including microbiota samples from eight anti–Ro60-positive lupus patients, five anti–Ro60-negative lupus patients, and seven healthy controls.
Disclosures: The study authors reported they had nothing to disclose. The work was supported by various grants from the National Institutes of Health, Arthritis National Research Foundation, the Arthritis Foundation, and the Lupus Research Institute, as well as NIH Intramural Research Programs.
Source: Greiling TM et al. Sci Transl Med. 2018;10(434). doi: 10.1126/scitranslmed.aan2306.
Shulkin: Privatization Fight Led to Ouster
After weeks of speculation, President Donald Trump fired VA Secretary David J. Shulkin, MD, on March 28 and proposed White House physician US Navy RADM Ronny L. Jackson, MD, as his replacement. In another unusual move, President Trump did not ask Deputy Secretary of Veterans Affairs Thomas G. Bowman to manage the agency during the confirmation process, but instead asked Robert Leon Wilkie Jr who was serving as the US Department of Defense Undersecretary of Defense for Personnel and Readiness.
While acknowledging that all cabinet members serve at the pleasure of the President, Shulkin placed the blame for his dismissal squarely on other VA political appointees. “There are many political appointees in the VA that believe that we are moving in the wrong direction or weren’t moving fast enough toward privatizing the VA,” Shulkin told NPR in an early morning interview. “I think that it’s essential for national security and for the country that we honor our commitment by having a strong VA. I was not against reforming VA, but I was against privatization.”
Shulkin was clearly prepared and quick to indentify his oponents in the administration. He wrote a New York Times op-ed that was published just hours after his firing. “Successes within the department have intensified the ambitions of people who want to put VA health care in the hands of the private sector,” Dr. Shulkin wrote. “I believe differences in philosophy deserve robust debate, and solutions should be determined based on the merits of the arguments. The advocates within the administration for privatizing VA health services, however, reject this approach. They saw me as an obstacle to privatization who had to be removed. That is because I am convinced that privatization is a political issue aimed at rewarding select people and companies with profits, even if it undermines care for veterans.”
“As I prepare to leave government,” Shulkin concluded, “I am struck by a recurring thought: It should not be this hard to serve your country.”
Although controversy has swirled around Dr. Shulkin since the release of a VA Inspector General report that was highly critical of a trip to Europe, he dismissed the trip as a cause of his dismissal. According to Shulkin, the White House had refused to allow him to defend himself and point out that the trip was for the White House and the VA. “This was the five allies conference, a trip that the VA secretary has participated in for 40 years with major allies,” he told NPR “The single expenditure spent was on a coach airfare for my wife who was officially invited. Everything was pre-approved by our ethics committee. When the Inspector General didn't like the way that my staff had handled the approval, I wrote a check back to the government.”
Despite the controversy, veteran service organizations (VSOs) and congressional leaders have remained supportive of Dr. Shulkin and skeptical of Dr. Jackson. Most of the VSOs were quick to issue statements. “We are grateful for [Dr. Shulkin’s] efforts to steer VA toward sensible, lasting transformation of veterans health care and hope to see those plans continue moving forward,” said Commander Delphine Metcalf-Foster of Disabled American Veterans in a statement. “While we look forward to learning more about the qualifications and views of the new nominee, we are extremely concerned about the existing leadership vacuum in VA. At a time of critical negotiations over the future of veterans health care reform, VA today has no Secretary, no Under Secretary of Health or Benefits, and the named Acting Secretary has no background in health care and no apparent experience working in or with the Department.”
In his statement, AMVETS Executive Director Joe Chenelly raised a number of issues that are likely to take center stage in the confirmation hearings, asking “is it appropriate for an active-duty military officer to run a federal agency?” and “with an official bio that does not seem to contain any indication that he’s held a command, is the president’s nominee fully prepared to lead such a massive bureaucracy?” Chenelly added, “I am deeply concerned about the nominee. Veterans’ lives depend on this decision, and the Trump administration needs to substantiate that this active-duty Navy officer is qualified to run a $200 billion bureaucracy, the second largest agency in the government.”
After weeks of speculation, President Donald Trump fired VA Secretary David J. Shulkin, MD, on March 28 and proposed White House physician US Navy RADM Ronny L. Jackson, MD, as his replacement. In another unusual move, President Trump did not ask Deputy Secretary of Veterans Affairs Thomas G. Bowman to manage the agency during the confirmation process, but instead asked Robert Leon Wilkie Jr who was serving as the US Department of Defense Undersecretary of Defense for Personnel and Readiness.
While acknowledging that all cabinet members serve at the pleasure of the President, Shulkin placed the blame for his dismissal squarely on other VA political appointees. “There are many political appointees in the VA that believe that we are moving in the wrong direction or weren’t moving fast enough toward privatizing the VA,” Shulkin told NPR in an early morning interview. “I think that it’s essential for national security and for the country that we honor our commitment by having a strong VA. I was not against reforming VA, but I was against privatization.”
Shulkin was clearly prepared and quick to indentify his oponents in the administration. He wrote a New York Times op-ed that was published just hours after his firing. “Successes within the department have intensified the ambitions of people who want to put VA health care in the hands of the private sector,” Dr. Shulkin wrote. “I believe differences in philosophy deserve robust debate, and solutions should be determined based on the merits of the arguments. The advocates within the administration for privatizing VA health services, however, reject this approach. They saw me as an obstacle to privatization who had to be removed. That is because I am convinced that privatization is a political issue aimed at rewarding select people and companies with profits, even if it undermines care for veterans.”
“As I prepare to leave government,” Shulkin concluded, “I am struck by a recurring thought: It should not be this hard to serve your country.”
Although controversy has swirled around Dr. Shulkin since the release of a VA Inspector General report that was highly critical of a trip to Europe, he dismissed the trip as a cause of his dismissal. According to Shulkin, the White House had refused to allow him to defend himself and point out that the trip was for the White House and the VA. “This was the five allies conference, a trip that the VA secretary has participated in for 40 years with major allies,” he told NPR “The single expenditure spent was on a coach airfare for my wife who was officially invited. Everything was pre-approved by our ethics committee. When the Inspector General didn't like the way that my staff had handled the approval, I wrote a check back to the government.”
Despite the controversy, veteran service organizations (VSOs) and congressional leaders have remained supportive of Dr. Shulkin and skeptical of Dr. Jackson. Most of the VSOs were quick to issue statements. “We are grateful for [Dr. Shulkin’s] efforts to steer VA toward sensible, lasting transformation of veterans health care and hope to see those plans continue moving forward,” said Commander Delphine Metcalf-Foster of Disabled American Veterans in a statement. “While we look forward to learning more about the qualifications and views of the new nominee, we are extremely concerned about the existing leadership vacuum in VA. At a time of critical negotiations over the future of veterans health care reform, VA today has no Secretary, no Under Secretary of Health or Benefits, and the named Acting Secretary has no background in health care and no apparent experience working in or with the Department.”
In his statement, AMVETS Executive Director Joe Chenelly raised a number of issues that are likely to take center stage in the confirmation hearings, asking “is it appropriate for an active-duty military officer to run a federal agency?” and “with an official bio that does not seem to contain any indication that he’s held a command, is the president’s nominee fully prepared to lead such a massive bureaucracy?” Chenelly added, “I am deeply concerned about the nominee. Veterans’ lives depend on this decision, and the Trump administration needs to substantiate that this active-duty Navy officer is qualified to run a $200 billion bureaucracy, the second largest agency in the government.”
After weeks of speculation, President Donald Trump fired VA Secretary David J. Shulkin, MD, on March 28 and proposed White House physician US Navy RADM Ronny L. Jackson, MD, as his replacement. In another unusual move, President Trump did not ask Deputy Secretary of Veterans Affairs Thomas G. Bowman to manage the agency during the confirmation process, but instead asked Robert Leon Wilkie Jr who was serving as the US Department of Defense Undersecretary of Defense for Personnel and Readiness.
While acknowledging that all cabinet members serve at the pleasure of the President, Shulkin placed the blame for his dismissal squarely on other VA political appointees. “There are many political appointees in the VA that believe that we are moving in the wrong direction or weren’t moving fast enough toward privatizing the VA,” Shulkin told NPR in an early morning interview. “I think that it’s essential for national security and for the country that we honor our commitment by having a strong VA. I was not against reforming VA, but I was against privatization.”
Shulkin was clearly prepared and quick to indentify his oponents in the administration. He wrote a New York Times op-ed that was published just hours after his firing. “Successes within the department have intensified the ambitions of people who want to put VA health care in the hands of the private sector,” Dr. Shulkin wrote. “I believe differences in philosophy deserve robust debate, and solutions should be determined based on the merits of the arguments. The advocates within the administration for privatizing VA health services, however, reject this approach. They saw me as an obstacle to privatization who had to be removed. That is because I am convinced that privatization is a political issue aimed at rewarding select people and companies with profits, even if it undermines care for veterans.”
“As I prepare to leave government,” Shulkin concluded, “I am struck by a recurring thought: It should not be this hard to serve your country.”
Although controversy has swirled around Dr. Shulkin since the release of a VA Inspector General report that was highly critical of a trip to Europe, he dismissed the trip as a cause of his dismissal. According to Shulkin, the White House had refused to allow him to defend himself and point out that the trip was for the White House and the VA. “This was the five allies conference, a trip that the VA secretary has participated in for 40 years with major allies,” he told NPR “The single expenditure spent was on a coach airfare for my wife who was officially invited. Everything was pre-approved by our ethics committee. When the Inspector General didn't like the way that my staff had handled the approval, I wrote a check back to the government.”
Despite the controversy, veteran service organizations (VSOs) and congressional leaders have remained supportive of Dr. Shulkin and skeptical of Dr. Jackson. Most of the VSOs were quick to issue statements. “We are grateful for [Dr. Shulkin’s] efforts to steer VA toward sensible, lasting transformation of veterans health care and hope to see those plans continue moving forward,” said Commander Delphine Metcalf-Foster of Disabled American Veterans in a statement. “While we look forward to learning more about the qualifications and views of the new nominee, we are extremely concerned about the existing leadership vacuum in VA. At a time of critical negotiations over the future of veterans health care reform, VA today has no Secretary, no Under Secretary of Health or Benefits, and the named Acting Secretary has no background in health care and no apparent experience working in or with the Department.”
In his statement, AMVETS Executive Director Joe Chenelly raised a number of issues that are likely to take center stage in the confirmation hearings, asking “is it appropriate for an active-duty military officer to run a federal agency?” and “with an official bio that does not seem to contain any indication that he’s held a command, is the president’s nominee fully prepared to lead such a massive bureaucracy?” Chenelly added, “I am deeply concerned about the nominee. Veterans’ lives depend on this decision, and the Trump administration needs to substantiate that this active-duty Navy officer is qualified to run a $200 billion bureaucracy, the second largest agency in the government.”