User login
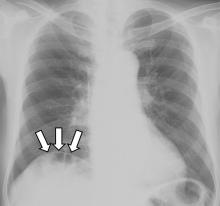
Gas under the right diaphragm
A 66-year-old man presented to the hospital with 3 days of nausea, vomiting, and abdominal pain. He had come to the emergency department several times during this period, but the cause of his symptoms had not been determined.
The patient was successfully treated with urgent right hemicolectomy.
THE CHILAIDITI SIGN AND SYNDROME
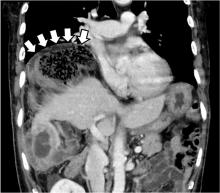
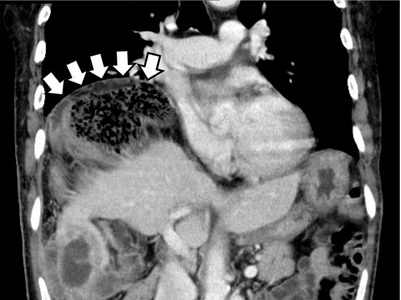
The Chilaiditi sign is an infrequent anomaly found incidentally on chest or abdominal radiography as a colonic interposition between the liver and right hemidiaphragm.1 It is often asymptomatic but is sometimes accompanied by nausea, vomiting, abdominal pain, and constipation, ie, Chilaiditi syndrome.
Generally, after conservative treatment with fasting and pain control, symptoms may subside and follow-up should be sufficient. However, nasogastric decompression and laxatives are occasionally needed and are often effective in patients with Chilaiditi syndrome. Urgent abdominal surgery is indicated for patients with symptoms of volvulus of the colon, stomach, or small intestine.2
DISTINGUISHING CHARACTERISTICS
The Chilaiditi sign is often confused with pneumoperitoneum, which usually requires urgent abdominal surgery. But the presence of haustration or valvulae conniventes (folds in the small bowel mucosa) in the hepatodiaphragmatic space helps distinguish between intraluminal gas and free air. If the patient presents with abdominal pain without signs of peritonitis, and if imaging indicates the Chilaiditi sign, then supplementary imaging (eg, decubitus radiography, chest CT, abdominal CT) is recommended to make the definitive diagnosis and to avoid unnecessary surgery.
Gas under the diaphragm on standing chest radiography without signs of peritonitis may also be seen after laparotomy and after scuba diving as well as in cases of biliary enteric fistula, incompetent sphincter of Oddi, gallstone ileus, and pneumatosis cystoides intestinalis. The incidence rate of the Chilaiditi sign detected by radiography is between 0.025% and 0.28%.3
PREDISPOSING FACTORS
The cause of the Chilaiditi sign remains unknown. Predisposing factors can be categorized as diaphragmatic (diaphragmatic thinning, phrenic nerve injury, expanded thoracic cavity), intestinal (megacolon, increased intra-abdominal pressure), and hepatic (hepatic atrophy, cirrhosis, ascites).
In healthy people, Chilaiditi syndrome is usually attributed to a congenital abnormal lengthening of the colon or to undue looseness of ligaments of the colon and liver.
Recognizing the Chilaiditi sign is particularly important in patients scheduled to undergo a percutaneous transhepatic procedure or colonoscopic examination, as these procedures increase the risk of perforation.
- Chilaiditi D. Zur Frage der Hepatoptose und Ptose im allegemeinen im Anschluss an drei Fälle von temporärer, partieller Leberverlagerung. Fortschritte auf dem Gebiete der Röntgenstrahlen 1910; 16:173–208.
- Williams A, Cox R, Palaniappan B, Woodward A. Chilaiditi’s syndrome associated with colonic volvulus and intestinal malrotation—a rare case. Int J Surg Case Rep 2014; 5:335–338.
- Orangio GR, Fazio VW, Winkelman E, McGonagle BA. The Chilaiditi syndrome and associated volvulus of the transverse colon. An indication for surgical therapy. Dis Colon Rectum 1986; 29:653-656.
A 66-year-old man presented to the hospital with 3 days of nausea, vomiting, and abdominal pain. He had come to the emergency department several times during this period, but the cause of his symptoms had not been determined.
The patient was successfully treated with urgent right hemicolectomy.
THE CHILAIDITI SIGN AND SYNDROME
The Chilaiditi sign is an infrequent anomaly found incidentally on chest or abdominal radiography as a colonic interposition between the liver and right hemidiaphragm.1 It is often asymptomatic but is sometimes accompanied by nausea, vomiting, abdominal pain, and constipation, ie, Chilaiditi syndrome.
Generally, after conservative treatment with fasting and pain control, symptoms may subside and follow-up should be sufficient. However, nasogastric decompression and laxatives are occasionally needed and are often effective in patients with Chilaiditi syndrome. Urgent abdominal surgery is indicated for patients with symptoms of volvulus of the colon, stomach, or small intestine.2
DISTINGUISHING CHARACTERISTICS
The Chilaiditi sign is often confused with pneumoperitoneum, which usually requires urgent abdominal surgery. But the presence of haustration or valvulae conniventes (folds in the small bowel mucosa) in the hepatodiaphragmatic space helps distinguish between intraluminal gas and free air. If the patient presents with abdominal pain without signs of peritonitis, and if imaging indicates the Chilaiditi sign, then supplementary imaging (eg, decubitus radiography, chest CT, abdominal CT) is recommended to make the definitive diagnosis and to avoid unnecessary surgery.
Gas under the diaphragm on standing chest radiography without signs of peritonitis may also be seen after laparotomy and after scuba diving as well as in cases of biliary enteric fistula, incompetent sphincter of Oddi, gallstone ileus, and pneumatosis cystoides intestinalis. The incidence rate of the Chilaiditi sign detected by radiography is between 0.025% and 0.28%.3
PREDISPOSING FACTORS
The cause of the Chilaiditi sign remains unknown. Predisposing factors can be categorized as diaphragmatic (diaphragmatic thinning, phrenic nerve injury, expanded thoracic cavity), intestinal (megacolon, increased intra-abdominal pressure), and hepatic (hepatic atrophy, cirrhosis, ascites).
In healthy people, Chilaiditi syndrome is usually attributed to a congenital abnormal lengthening of the colon or to undue looseness of ligaments of the colon and liver.
Recognizing the Chilaiditi sign is particularly important in patients scheduled to undergo a percutaneous transhepatic procedure or colonoscopic examination, as these procedures increase the risk of perforation.
A 66-year-old man presented to the hospital with 3 days of nausea, vomiting, and abdominal pain. He had come to the emergency department several times during this period, but the cause of his symptoms had not been determined.
The patient was successfully treated with urgent right hemicolectomy.
THE CHILAIDITI SIGN AND SYNDROME
The Chilaiditi sign is an infrequent anomaly found incidentally on chest or abdominal radiography as a colonic interposition between the liver and right hemidiaphragm.1 It is often asymptomatic but is sometimes accompanied by nausea, vomiting, abdominal pain, and constipation, ie, Chilaiditi syndrome.
Generally, after conservative treatment with fasting and pain control, symptoms may subside and follow-up should be sufficient. However, nasogastric decompression and laxatives are occasionally needed and are often effective in patients with Chilaiditi syndrome. Urgent abdominal surgery is indicated for patients with symptoms of volvulus of the colon, stomach, or small intestine.2
DISTINGUISHING CHARACTERISTICS
The Chilaiditi sign is often confused with pneumoperitoneum, which usually requires urgent abdominal surgery. But the presence of haustration or valvulae conniventes (folds in the small bowel mucosa) in the hepatodiaphragmatic space helps distinguish between intraluminal gas and free air. If the patient presents with abdominal pain without signs of peritonitis, and if imaging indicates the Chilaiditi sign, then supplementary imaging (eg, decubitus radiography, chest CT, abdominal CT) is recommended to make the definitive diagnosis and to avoid unnecessary surgery.
Gas under the diaphragm on standing chest radiography without signs of peritonitis may also be seen after laparotomy and after scuba diving as well as in cases of biliary enteric fistula, incompetent sphincter of Oddi, gallstone ileus, and pneumatosis cystoides intestinalis. The incidence rate of the Chilaiditi sign detected by radiography is between 0.025% and 0.28%.3
PREDISPOSING FACTORS
The cause of the Chilaiditi sign remains unknown. Predisposing factors can be categorized as diaphragmatic (diaphragmatic thinning, phrenic nerve injury, expanded thoracic cavity), intestinal (megacolon, increased intra-abdominal pressure), and hepatic (hepatic atrophy, cirrhosis, ascites).
In healthy people, Chilaiditi syndrome is usually attributed to a congenital abnormal lengthening of the colon or to undue looseness of ligaments of the colon and liver.
Recognizing the Chilaiditi sign is particularly important in patients scheduled to undergo a percutaneous transhepatic procedure or colonoscopic examination, as these procedures increase the risk of perforation.
- Chilaiditi D. Zur Frage der Hepatoptose und Ptose im allegemeinen im Anschluss an drei Fälle von temporärer, partieller Leberverlagerung. Fortschritte auf dem Gebiete der Röntgenstrahlen 1910; 16:173–208.
- Williams A, Cox R, Palaniappan B, Woodward A. Chilaiditi’s syndrome associated with colonic volvulus and intestinal malrotation—a rare case. Int J Surg Case Rep 2014; 5:335–338.
- Orangio GR, Fazio VW, Winkelman E, McGonagle BA. The Chilaiditi syndrome and associated volvulus of the transverse colon. An indication for surgical therapy. Dis Colon Rectum 1986; 29:653-656.
- Chilaiditi D. Zur Frage der Hepatoptose und Ptose im allegemeinen im Anschluss an drei Fälle von temporärer, partieller Leberverlagerung. Fortschritte auf dem Gebiete der Röntgenstrahlen 1910; 16:173–208.
- Williams A, Cox R, Palaniappan B, Woodward A. Chilaiditi’s syndrome associated with colonic volvulus and intestinal malrotation—a rare case. Int J Surg Case Rep 2014; 5:335–338.
- Orangio GR, Fazio VW, Winkelman E, McGonagle BA. The Chilaiditi syndrome and associated volvulus of the transverse colon. An indication for surgical therapy. Dis Colon Rectum 1986; 29:653-656.
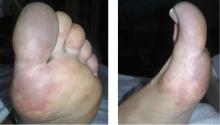
Paraneoplastic acral vascular syndrome
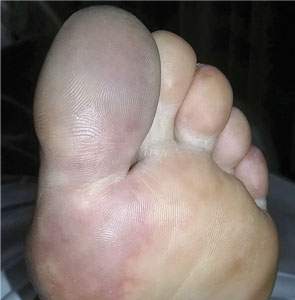
A 66-year-old woman presented to the emergency room with pain and bluish discoloration of her left great toe for the past 2 days. She had no history of trauma to the toe or of peripheral vascular disease. She also complained of intermittent abdominal pain for the last 5 months and unintentional weight loss of 8 pounds.
Vascular disease was suspected, and the patient was started on systemic anticoagulation. However, chest computed tomography (CT) and conventional angiography showed no aortic disease; vessels were of normal caliber, and no distal filling defects were noted.
A routine evaluation with complete blood cell count, peripheral smear, renal function testing, and urinalysis with eosinophil smear was also unrevealing. An extensive investigation followed, including serum complement testing, assays for antinuclear, antineutrophil cytoplasmic, and anti-Sjögren syndrome A and B antibodies, cryoglobulin testing, and hepatitis B and C virus serology, as well as screening for syphilis and lupus anticoagulant, anticardiolipin, and beta-2 glycoprotein antibodies. The results for all these tests were also unremarkable.
Results of coagulation testing and venous duplex ultrasonography of the legs were normal, and electrocardiography and echocardiography showed no signs of valvular vegetation, myxoma, or patent foramen ovale.
Given our patient’s age, abdominal pain, and weight loss but negative vascular evaluation, we considered a diagnosis of paraneoplastic acral vascular syndrome. Abdominal CT revealed a tumor of the pancreatic head with multiple liver lesions, and cytologic study confirmed pancreatic adenocarcinoma.
DISTANT MARKERS OF MALIGNANCY
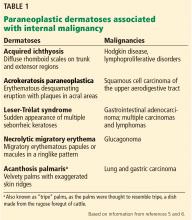
Causes of blue toe syndrome to consider in the differential diagnosis include arterial thromboembolism, vasoconstrictive drug use or disorders, vasculitis, and venous thrombosis.1 These are common and deserve prompt investigation. However, if they are ruled out, a peripheral acral vascular syndrome should be considered. These syndromes present as Raynaud phenomenon, gangrene, or acrocyanosis of the fingers or toes with underlying neoplasia. Unusual features such as sudden onset in a patient over age 50, an acral distribution, and associated symptoms such as unrelated pain and weight loss should spark concern for underlying malignancy.
Paraneoplastic syndromes are defined as signs and symptoms that present distant from the site of malignancy. Dermatoses as markers of internal malignancy are well-established but perplexing clinical entities whose exact causes remain unknown.2
Paraneoplastic dermatoses are well recognized as harbingers of metastatic disease.5,6 Our patient’s story demonstrates need for a thorough diagnostic investigation.
- Hirschmann JV, Raugi GJ. Blue (or purple) toe syndrome. J Am Acad Dermatol 2009; 60:1–20.
- Naschitz JE, Rosner I, Rozenbaum M, Zuckerman E, Yeshurun D. Rheumatic syndromes: clues to occult neoplasia. Semin Arthritis Rheum 1999; 29:43–55.
- Poszepczynska-Guigné E, Viguier M, Chosidow O, Orcel B, Emmerich J, Dubertret L. Paraneoplastic acral vascular syndrome: epidemiologic features, clinical manifestations, and disease sequelae. J Am Acad Dermatol 2002; 47:47–52.
- DeCross AJ, Sahasrabudhe DM. Paraneoplastic Raynaud's phenomenon. Am J Med 1992; 92:571–572.
- Ramos-E-Silva M, Carvalho JC, Carneiro SC. Cutaneous paraneoplasia. Clin Dermatol 2011; 29:541–547.
- Chung VQ, Moschella SL, Zembowicz A, Liu V. Clinical and pathologic findings of paraneoplastic dermatoses. J Am Acad Dermatol 2006; 54:745–762.
A 66-year-old woman presented to the emergency room with pain and bluish discoloration of her left great toe for the past 2 days. She had no history of trauma to the toe or of peripheral vascular disease. She also complained of intermittent abdominal pain for the last 5 months and unintentional weight loss of 8 pounds.
Vascular disease was suspected, and the patient was started on systemic anticoagulation. However, chest computed tomography (CT) and conventional angiography showed no aortic disease; vessels were of normal caliber, and no distal filling defects were noted.
A routine evaluation with complete blood cell count, peripheral smear, renal function testing, and urinalysis with eosinophil smear was also unrevealing. An extensive investigation followed, including serum complement testing, assays for antinuclear, antineutrophil cytoplasmic, and anti-Sjögren syndrome A and B antibodies, cryoglobulin testing, and hepatitis B and C virus serology, as well as screening for syphilis and lupus anticoagulant, anticardiolipin, and beta-2 glycoprotein antibodies. The results for all these tests were also unremarkable.
Results of coagulation testing and venous duplex ultrasonography of the legs were normal, and electrocardiography and echocardiography showed no signs of valvular vegetation, myxoma, or patent foramen ovale.
Given our patient’s age, abdominal pain, and weight loss but negative vascular evaluation, we considered a diagnosis of paraneoplastic acral vascular syndrome. Abdominal CT revealed a tumor of the pancreatic head with multiple liver lesions, and cytologic study confirmed pancreatic adenocarcinoma.
DISTANT MARKERS OF MALIGNANCY
Causes of blue toe syndrome to consider in the differential diagnosis include arterial thromboembolism, vasoconstrictive drug use or disorders, vasculitis, and venous thrombosis.1 These are common and deserve prompt investigation. However, if they are ruled out, a peripheral acral vascular syndrome should be considered. These syndromes present as Raynaud phenomenon, gangrene, or acrocyanosis of the fingers or toes with underlying neoplasia. Unusual features such as sudden onset in a patient over age 50, an acral distribution, and associated symptoms such as unrelated pain and weight loss should spark concern for underlying malignancy.
Paraneoplastic syndromes are defined as signs and symptoms that present distant from the site of malignancy. Dermatoses as markers of internal malignancy are well-established but perplexing clinical entities whose exact causes remain unknown.2
Paraneoplastic dermatoses are well recognized as harbingers of metastatic disease.5,6 Our patient’s story demonstrates need for a thorough diagnostic investigation.
A 66-year-old woman presented to the emergency room with pain and bluish discoloration of her left great toe for the past 2 days. She had no history of trauma to the toe or of peripheral vascular disease. She also complained of intermittent abdominal pain for the last 5 months and unintentional weight loss of 8 pounds.
Vascular disease was suspected, and the patient was started on systemic anticoagulation. However, chest computed tomography (CT) and conventional angiography showed no aortic disease; vessels were of normal caliber, and no distal filling defects were noted.
A routine evaluation with complete blood cell count, peripheral smear, renal function testing, and urinalysis with eosinophil smear was also unrevealing. An extensive investigation followed, including serum complement testing, assays for antinuclear, antineutrophil cytoplasmic, and anti-Sjögren syndrome A and B antibodies, cryoglobulin testing, and hepatitis B and C virus serology, as well as screening for syphilis and lupus anticoagulant, anticardiolipin, and beta-2 glycoprotein antibodies. The results for all these tests were also unremarkable.
Results of coagulation testing and venous duplex ultrasonography of the legs were normal, and electrocardiography and echocardiography showed no signs of valvular vegetation, myxoma, or patent foramen ovale.
Given our patient’s age, abdominal pain, and weight loss but negative vascular evaluation, we considered a diagnosis of paraneoplastic acral vascular syndrome. Abdominal CT revealed a tumor of the pancreatic head with multiple liver lesions, and cytologic study confirmed pancreatic adenocarcinoma.
DISTANT MARKERS OF MALIGNANCY
Causes of blue toe syndrome to consider in the differential diagnosis include arterial thromboembolism, vasoconstrictive drug use or disorders, vasculitis, and venous thrombosis.1 These are common and deserve prompt investigation. However, if they are ruled out, a peripheral acral vascular syndrome should be considered. These syndromes present as Raynaud phenomenon, gangrene, or acrocyanosis of the fingers or toes with underlying neoplasia. Unusual features such as sudden onset in a patient over age 50, an acral distribution, and associated symptoms such as unrelated pain and weight loss should spark concern for underlying malignancy.
Paraneoplastic syndromes are defined as signs and symptoms that present distant from the site of malignancy. Dermatoses as markers of internal malignancy are well-established but perplexing clinical entities whose exact causes remain unknown.2
Paraneoplastic dermatoses are well recognized as harbingers of metastatic disease.5,6 Our patient’s story demonstrates need for a thorough diagnostic investigation.
- Hirschmann JV, Raugi GJ. Blue (or purple) toe syndrome. J Am Acad Dermatol 2009; 60:1–20.
- Naschitz JE, Rosner I, Rozenbaum M, Zuckerman E, Yeshurun D. Rheumatic syndromes: clues to occult neoplasia. Semin Arthritis Rheum 1999; 29:43–55.
- Poszepczynska-Guigné E, Viguier M, Chosidow O, Orcel B, Emmerich J, Dubertret L. Paraneoplastic acral vascular syndrome: epidemiologic features, clinical manifestations, and disease sequelae. J Am Acad Dermatol 2002; 47:47–52.
- DeCross AJ, Sahasrabudhe DM. Paraneoplastic Raynaud's phenomenon. Am J Med 1992; 92:571–572.
- Ramos-E-Silva M, Carvalho JC, Carneiro SC. Cutaneous paraneoplasia. Clin Dermatol 2011; 29:541–547.
- Chung VQ, Moschella SL, Zembowicz A, Liu V. Clinical and pathologic findings of paraneoplastic dermatoses. J Am Acad Dermatol 2006; 54:745–762.
- Hirschmann JV, Raugi GJ. Blue (or purple) toe syndrome. J Am Acad Dermatol 2009; 60:1–20.
- Naschitz JE, Rosner I, Rozenbaum M, Zuckerman E, Yeshurun D. Rheumatic syndromes: clues to occult neoplasia. Semin Arthritis Rheum 1999; 29:43–55.
- Poszepczynska-Guigné E, Viguier M, Chosidow O, Orcel B, Emmerich J, Dubertret L. Paraneoplastic acral vascular syndrome: epidemiologic features, clinical manifestations, and disease sequelae. J Am Acad Dermatol 2002; 47:47–52.
- DeCross AJ, Sahasrabudhe DM. Paraneoplastic Raynaud's phenomenon. Am J Med 1992; 92:571–572.
- Ramos-E-Silva M, Carvalho JC, Carneiro SC. Cutaneous paraneoplasia. Clin Dermatol 2011; 29:541–547.
- Chung VQ, Moschella SL, Zembowicz A, Liu V. Clinical and pathologic findings of paraneoplastic dermatoses. J Am Acad Dermatol 2006; 54:745–762.
A 75-year-old with abdominal pain, hypoxia, and weak pulses in the left leg
A 75-year-old man presented to the emergency department for evaluation of abdominal pain. He had stage 3 chronic obstructive pulmonary disease (COPD), with a forced expiratory volume in 1 second of 33%.
PREVIOUS HOSPITALIZATION
Aside from his COPD, he had been healthy until 1 month earlier, when he had been hospitalized because of shortness of breath and chest pressure with exertion. His troponin T level had been elevated, peaking at 0.117 ng/mL (reference range 0–0.029).
Left heart catheterization had shown no significant coronary artery disease. A myocardial bridge of the distal left anterior descending coronary artery had been seen, so that the artery appeared to be narrowed by 50% to 60% with ventricular contraction. But this was not thought to have been the cause of his presentation.
On discharge, he required oxygen 4 L/min by nasal cannula. Previously, he had not needed supplemental oxygen.
CURRENT PRESENTATION
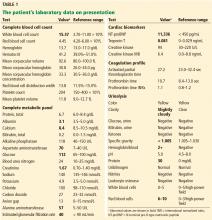
The patient described persistent and severe periumbilical abdominal pain during the previous day. It was not associated with eating, and he denied diarrhea, constipation, hematemesis, hematochezia, bright red blood per rectum, or melena. He continued to describe persistent shortness of breath and pleuritic chest pain. His vital signs were as follows:
- Heart rate 104 beats per minute
- Respiratory rate 16 to 20 breaths per minute
- Blood pressure 101–142/62–84 mm Hg
- Oxygen saturation 78% on room air.
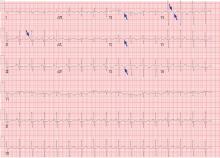
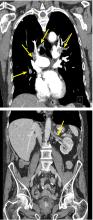
His laboratory findings on presentation are shown in Table 1, and his electrocardiogram is shown in Figure 1.
WHAT DOES HIS ELECTROCARDIOGRAM SHOW?
1. Which of the following is the most accurate description of this patient’s electrocardiogram?
- Sinus tachycardia, peaked P waves (P pulmonale) in lead II, and T-wave inversions in the right precordial leads
- Sinus tachycardia and left bundle branch block
- Sinus tachycardia and poor R-wave progression
- Sinus tachycardia and ST elevation in the precordial leads
Our patient’s electrocardiogram shows sinus tachycardia, P pulmonale, T-wave inversion in the right precordial leads (V1–V3), and biphasic T waves in lead V4,, which suggest right ventricular strain.
The rhythm most commonly seen in patients with pulmonary embolism is sinus tachycardia, followed by nonspecific ST-segment or T-wave abnormalities. In one series of patients with acute pulmonary embolism, the classic findings of P pulmonale, right ventricular hypertrophy, right axis deviation, and right bundle branch block were rare (< 6%).1 Thus, these classic findings are not sensitive for the diagnosis of pulmonary embolism, and their absence does not rule it out.
Further studies for our patient
Transthoracic echocardiography was performed to look for evidence of right ventricular strain secondary to the pulmonary embolism.
ECHOCARDIOGRAPHIC SIGNS OF PULMONARY EMBOLISM
2. Which of the following findings on transthoracic echocardiography would not suggest acute pulmonary embolism?
- Midright ventricular wall hypokinesis with apical sparing
- Severe tricuspid regurgitation
- Left ventricular dilation
- Lack of respiratory variation of the inferior vena cava
- Septal wall motion toward the left ventricle
Left ventricular dilation does not suggest acute pulmonary embolism. Echocardiograms of patients with acute submassive pulmonary embolism typically show evidence of right ventricular strain, such as the other entities listed above (midright ventricular hypokinesis with apical sparing, severe tricuspid regurgitation, lack of respiratory variation of the inferior vena cava, and septal wall motion toward the left ventricle).
The degree of right ventricular dysfunction is related to the extent of acute pulmonary vascular occlusion and aids in risk-stratification of patients with acute pulmonary embolism. Midright ventricular wall hypokinesis with apical sparing has been termed the McConnell sign.2
In our patient, transthoracic echocardiography showed:
- Normal left ventricular ejection fraction
- Mild diastolic dysfunction
- Right ventricular dilation with moderately decreased right ventricular systolic function and apical sparing
- Right ventricular systolic pressure 54 mm Hg, consistent with moderate pulmonary hypertension
- Right atrial pressure 10 mm Hg
- No inspiratory collapse of a dilated inferior vena cava
- Mild tricuspid valve regurgitation.
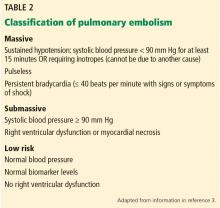
CLASSIFICATION OF ACUTE PULMONARY EMBOLISM
3. Given the above information, how would you classify the patient’s pulmonary embolism?
- Massive
- Submassive
- Low-risk
- Clinically stable
The patient’s pulmonary embolism is submassive.
Historically, the classification of pulmonary embolism was determined by the angiographic thrombus burden. However, this has limited utility because clinical factors (eg, hypotension on initial presentation) have been shown to be better predictors of short-term mortality risk.3
Our patient is characterized as having a submassive pulmonary embolism based on elevated biomarkers (troponin T, N-terminal pro-B-type natriuretic peptide) and right ventricular dysfunction in the absence of hypotension.
ULTRASONOGRAPHY FOR DIAGNOSIS OF DEEP VEIN THROMBOSIS
Venous duplex ultrasonography has become the standard for diagnosis of lower extremity deep vein thrombosis. However, its quality and diagnostic accuracy depend on the skill of the person performing the examination. It is further limited by certain patient characteristics, including severe obesity, edema, and wounds and dressings at the site being examined.5
Our patient underwent duplex ultrasonography of the lower extremities, which demonstrated acute proximal and calf deep vein thrombosis in the right femoral, popliteal, and peroneal veins and no deep vein thrombosis in the left leg.
RISK STRATIFICATION IN ACUTE PULMONARY EMBOLISM
Multiple models exist to estimate the risk of complications in patients with acute pulmonary embolism.
The Bova score6 is based on the following factors:
- Systolic blood pressure 90–100 mm Hg (2 points) (patients with systolic blood pressure lower than 90 mm Hg were excluded from the study from which this score was derived)
- Cardiac troponin elevation (2 points)
- Right ventricular dysfunction on echocardiography or computed tomography (2 points)
- Heart rate 100 beats/min or greater (1 point).
A total score of 0, 1, or 2 (stage I) denotes low risk, 3 or 4 points (stage II) intermediate risk, and more than 4 points (stage III) high risk.
The PESI score (Pulmonary Embolism Severity Index)7 is based on:
- Age (1 point per year)
- Sex (10 points for being male)
- Heart rate 110 per minute or greater (20 points)
- Cancer (30 points)
- Heart failure (10 points)
- Chronic lung disease (10 points)
- Systolic blood pressure less than 100 mm Hg (30 points)
- Respiratory rate at least 30 per minute (20 points)
- Temperature less than 36ºC (20 points)
- Altered mental status (60 points)
- Arterial oxygen saturation less than 90% (20 points).
The total score is broken down into 5 classes: I (< 65 points), II (65–85), III (86–105), IV (106–125), and V (> 126). Classes I and II are low risk, and the higher ones are high risk.
The simplified PESI score8 was developed to more rapidly risk-stratify patients and has been found to be similar to the PESI score in prognostic accuracy. Patients get 1 point for each of the following:
- Age over 80
- Cancer
- Chronic cardiopulmonary disease (heart failure or chronic lung disease)
- Heart rate 110 per minute or greater
- Systolic blood pressure less than 100 mm Hg
- Arterial oxygen saturation less than 90%.
A total score of 0 is low risk; anything higher is high risk.
Back to our patient
Our patient had proximal and calf deep vein thrombosis of the right leg, bilateral submassive pulmonary emboli with associated biomarker elevation and right ventricular dysfunction, and left renal artery thrombosis with infarction. Using the PESI score, his risk of death in the next 30 days was 13.7% and his 30-day risk of a complicated course was 27%. Using the Bova score, his 30-day risk of death was 15.5% and his 30-day risk of a complicated course was 29.2%.6,7
Notably, the patient’s right ventricular function had also been impaired on the echocardiogram performed during his admission 1 month previously. On transthoracic echocardiography during the current admission, the patient was found to have a similar degree of right ventricular dysfunction. This finding, along with the oxygen requirement that developed during the earlier admission, suggested that his pulmonary embolism may have been subacute and that the diagnosis may have been missed during the earlier hospital stay.
The patient was treated with unfractionated heparin. After the hospital’s multidisciplinary pulmonary embolism response team discussed and weighed the above factors, they recommended to not pursue thrombolytic therapy or inferior vena cava filter placement.
Of note, the patient’s pulses in the left lower extremity continued to be weak but palpable, and the left leg was cooler to touch than the right leg.
ASSESSING PERIPHERAL ARTERY DISEASE
4. How should the finding of weak pulses in this patient’s left leg be initially investigated?
- Computed tomographic angiography with runoff
- Ankle-brachial indices with pulse-volume recordings
- Arterial duplex ultrasonography
- Magnetic resonance angiography of the lower extremities
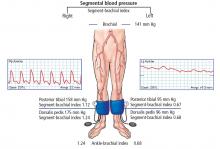
The ankle-brachial index is the initial diagnostic test for assessment of pulse abnormalities and for diagnosis of lower-extremity peripheral artery disease. It is calculated by dividing the higher of the ankle systolic pressures (posterior tibial or dorsalis pedis) by the higher of the 2 brachial pressures (left or right).9 Normal values are between 1.00 and 1.40.
Ankle-brachial indices in our patient
Our patient underwent measurement of his brachial, dorsalis pedis, and posterior tibial artery systolic pressures using blood pressure cuffs and continuous-wave Doppler. Ankle pulse-volume recordings were also obtained.
Given the patient’s poor renal function and concern for acute renal infarction, we thought it best to avoid iodinated or gadolinium contrast, such as with magnetic resonance or computed tomographic angiography.
Segmental leg pressures and pulse-volume recordings can be performed to help localize the level of arterial disease in the extremities, but were not done in this case because of the extensive deep vein thrombosis in the right leg.10,11
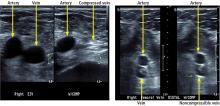
Arterial ultrasonography in our patient
Arterial duplex ultrasonography was performed to help determine the location of arterial disease. It showed patent arteries in the right leg. In the left lower extremity there was slow, monophasic blood flow in the distal superficial femoral artery. The popliteal artery was occluded. The posterior tibial artery was occluded at the origin, with reconstitution distally. The peroneal artery was occluded throughout. The anterior tibial artery was patent throughout. The ultrasonographic findings were thought to be suspicious for arterial thromboembolism.
WHAT CAN CAUSE BOTH ARTERIAL AND VENOUS THROMBOSIS?
5. Given that the patient had both arterial thrombosis (renal artery, lower-extremity arteries) and venous thromboembolism (deep vein thrombosis and pulmonary embolism), which of the following would be included in the differential diagnosis?
- Antiphospholipid antibody syndrome
- Protein C or protein S deficiency
- Malignancy
- Paradoxical embolization
- Factor V Leiden mutation
Correct answers include antiphospholipid antibody syndrome, malignancy, and paradoxical embolization.
The differential diagnosis for concomitant venous and arterial thrombosis is broad,12 and includes the following:
- Structural factors: patent foramen ovale, popliteal artery aneurysm
- Malignancy
- Inflammatory diseases: Behçet disease, Buerger disease, inflammatory bowel disease, antiphospholipid antibody syndrome, elevated lipoprotein(a), elevated homocysteine
- Hematologic diseases: myelodysplastic syndrome, disseminated intravascular coagulation, paroxysmal nocturnal hemoglobinuria, heparin-induced thrombocytopenia.
Traditional risk factors for venous thromboembolism include protein C deficiency, protein S deficiency, factor V Leiden mutation, the prothrombin G20210A gene mutation, and others. These are relatively minor risk factors for venous thrombosis and do not pose a risk for arterial thrombosis.12 In contrast, antiphospholipid antibody syndrome and malignancy pose a risk for both venous and arterial thrombosis. Paradoxical embolism is a mechanism by which arterial thrombosis (emboli) can develop in the setting of existing venous thrombosis.12
Our patient underwent testing for antiphospholipid antibodies and lupus anticoagulant, and he was encouraged to undergo age-appropriate cancer screening as an outpatient.12
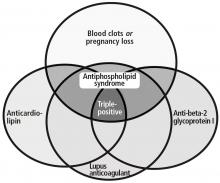
ANTIPHOSPHOLIPID ANTIBODY SYNDROME
Antiphospholipid antibody syndrome is defined by both clinical and laboratory criteria. Clinical symptoms include vascular thrombosis (arterial, venous, or both) and pregnancy-related complications.13
Laboratory criteria require the presence of antiphospholipid antibodies or lupus anticoagulant. These must be confirmed with repeat testing in 12 weeks. Antiphospholipid antibodies are detected by an enzyme-linked immunosorbent assay; laboratory assessment for the presence of lupus anticoagulant is a stepwise process and relies on 4 criteria:
- There should be prolongation of a phospholipid-dependent clotting test (eg, activated partial thromboplastin time, dilute Russell viper venom time test).
- There must be evidence of an inhibitory activity with mixing study.
- The inhibitor must exhibit phospholipid dependence; that is, with more phospholipid there is shortening of clotting time.
- Specific inhibitors must be excluded, including factor VIII and anticoagulant drugs such as heparin.14–17
Clinically, one should consider antiphospholipid antibody syndrome in patients who have arterial thrombosis, a history of pregnancy morbidity, or unexplained prolongation of activated partial thromboplastin time.13
Antiphospholipid antibodies may be present in up to a quarter of patients with venous thromboembolism, but it is persistent positivity of antibody assays that is associated with increased future risk of venous thromboembolism.19 Of note, the risk of venous thromboembolism in patients with confirmed antiphospholipid antibody syndrome is 10 times higher than in the general population.20
ANTIPHOSPHOLIPID ANTIBODIES ARE NOT ALL THE SAME
6. Which of the following antiphospholipid antibodies have not been associated with an increased thrombotic risk?
- Anti-beta-2-glycoprotein I IgG
- Lupus anticoagulant
- Antiphosphatidylserine
- Anticardiolipin IgM
- Anticardiolipin IgG
The correct answer is antiphosphatidylserine.15
Antiphospholipid antibodies are directed against a portion of select plasma proteins that are uncovered upon phospholipid binding. While lupus anticoagulant, anti-beta-2-glycoprotein I, and anticardiolipin antibodies are associated with thrombosis, antiprothrombin antibodies (including antiprothrombin and antiphosphatidylserine antibodies) are not.15,21
PARADOXICAL EMBOLISM
Patent foramen ovale, a communication between the right and left atrium in the interatrial septum, is associated with an increased risk of paradoxical embolization. The prevalence of patent foramen ovale is estimated to be 27% to 29% in the general population.22 Noncerebral systemic paradoxical embolism occurs less frequently than cerebral embolism, accounting for approximately 5% to 10% of paradoxical emboli.22
To evaluate for patent foramen ovale, transthoracic echocardiography is performed with a bubble (agitated saline contrast) study to assess for interatrial shunting. Transesophageal echocardiography or transcranial Doppler bubble studies may also be performed.
Although patent foramen ovale is most commonly associated with cerebral embolism, peripheral emboli can occur. Some research suggests that this may be a more common cause of arterial thromboembolism in younger patients. There have also been reports of other sites of systemic embolization, including the renal artery.12
Back to our patient
Initial antiphospholipid antibody testing was positive for lupus anticoagulant. Anticardiolipin and anti-beta-2-glycoprotein I antibodies were not detected.
Transesophageal echocardiography revealed a patent foramen ovale with a highly mobile atrial septum (atrial septal aneurysm).
The patient was treated with intravenous unfractionated heparin with bridging to warfarin with a target international normalized ratio (INR) of 2 to 3. His renal artery infarction and his lower-extremity arterial thromboembolic event were conservatively managed. His respiratory status improved, and he no longer required supplemental oxygen. His creatinine peaked at 1.7 mg/dL during his admission and improved to 1.2 mg/dL before he was discharged.
At follow-up, repeat echocardiography showed that his right ventricular systolic pressure had improved (decreased) to 37 mm Hg from 54 mm Hg. Repeat confirmatory testing was positive for lupus anticoagulant 12 weeks later. He has been maintained on warfarin with an INR goal of 2 to 3 as well as low-dose aspirin with plans for long-term anticoagulation. We decided to keep the patient on anticoagulation indefinitely with warfarin; he was not a candidate for a direct oral anticoagulant, given limited data on the use of these agents in the setting of lupus anticoagulant and antiphospholipid antibody syndrome.
SUMMARY OF CASE
In summary, this patient was a 75-year-old man with COPD who presented with abdominal pain. He was noted to have a left renal infarction, extensive unprovoked lower-extremity deep vein thrombosis with pulmonary emboli, and lower limb arterial thromboembolism.
He also had an underlying hypercoagulable state—antiphospholipid antibody syndrome—that predisposed him to both arterial and venous thrombosis. He was ultimately found to have a patent foramen ovale, which further increased the risk of arterial thrombosis by facilitating paradoxical embolization of venous thrombi. It is not certain whether the renal infarction and leg artery thrombi were due to paradoxical embolism or to in situ thrombosis, but we believe that it was most likely paradoxical embolization.
- Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest 1991; 100:598–603.
- Alsoos F, Khaddam A. Echocardiographic evaluation methods for right ventricular function. J Echocardiogr 2015; 13:43–51.
- Jaff MR, McMurtry MS, Archer SL, et al; American Heart Association Council on Cardiopulmonary, Critical Care, Perioperative and Resuscitation; American Heart Association Council on Peripheral Vascular Disease; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation 2011; 123:1788–1830.
- Heit JA, Silverstein MD, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ 3rd. Risk factors for deep vein thrombosis and pulmonary embolism: a population-based case-control study. Arch Intern Med 2000; 160:809–815.
- Gornik HL, Sharma AM. Duplex ultrasound in the diagnosis of lower-extremity deep venous thrombosis. Circulation 2014; 129:917–921.
- Fernández C, Bova C, Sanchez O, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest 2015; 148:211–218.
- Aujesky D, Perrier A, Roy PM, et al. Validation of a clinical prognostic model to identify low-risk patients with pulmonary embolism. J Intern Med 2007; 261:597–604.
- Jiménez D, Aujesky D, Moores L, et al; RIETE Investigators. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med 2010; 170:1383–1389.
- Kim ES, Wattanakit K, Gornik HL. Using the ankle-brachial index to diagnose peripheral artery disease and assess cardiovascular risk. Cleve Clin J Med 2012; 79:651–661.
- Jaff MR. Lower extremity arterial disease. Diagnostic aspects. Cardiol Clin 2002; 20:491–500.
- Rooke TW, Hirsch AT, Misra S, et al; American College of Cardiology Foundation Task Force; American Heart Association Task Force. Management of patients with peripheral artery disease (compilation of 2005 and 2011 ACCF/AHA Guideline Recommendations): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 61:1555–1570.
- Lichtin A, Bartholomew J. The coagulation consult: a case-based guide. New York, NY: Springer; 2014.
- Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med 2002; 346:752–763.
- Brandt JT, Triplett DA, Alving B, Scharrer I. Criteria for the diagnosis of lupus anticoagulants: an update. On behalf of the Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the ISTH. Thromb Haemost 1995; 74:1185–1190.
- Miyakis S, Lockshin M, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost 2006; 4:295–306.
- Pengo V, Tripodi A, Reber G, et al; Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis. Update of the guidelines for lupus anticoagulant detection. Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis. J Thromb Haemost 2009; 7:1737–1740.
- Nichols WL, Kottke-Marchant K, Ledford-Kraemer MR, Homburger HA, Cardel LK. Lupus anticoagulants, antiphospholipid antibodies, and antiphospholipid syndrome. In: Kottke-Marchant K, Davis BH, editors. Laboratory Hematology Practice. Hoboken, New Jersey: Blackwell Publishing, Ltd.; 2012:509–525.
- Houghton DE, Moll S. Antiphospholipid antibodies. Vasc Med 2017; 22:545–550.
- Roldan V, Lecumberri R, Muñoz-Torrero JFS, et al; RIETE Investigators. Thrombophilia testing in patients with venous thromboembolism. Findings from the RIETE registry. Thromb Res 2009; 124:174–177.
- Wahl DG, Guillemin F, de Maistre E, Perret-Guillaume C, Lecompte T, Thibaut G. Meta-analysis of the risk of venous thrombosis in individuals with antiphospholipid antibodies without underlying autoimmune disease or previous thrombosis. Lupus 1998; 7:15–22.
- Love PE, Santoro SA. Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders. Prevalence and clinical significance. Ann Intern Med 1990; 112:682–698.
- Thompson T, Evans W. Paradoxical embolism. QJM 1930; os-23:135–150.
A 75-year-old man presented to the emergency department for evaluation of abdominal pain. He had stage 3 chronic obstructive pulmonary disease (COPD), with a forced expiratory volume in 1 second of 33%.
PREVIOUS HOSPITALIZATION
Aside from his COPD, he had been healthy until 1 month earlier, when he had been hospitalized because of shortness of breath and chest pressure with exertion. His troponin T level had been elevated, peaking at 0.117 ng/mL (reference range 0–0.029).
Left heart catheterization had shown no significant coronary artery disease. A myocardial bridge of the distal left anterior descending coronary artery had been seen, so that the artery appeared to be narrowed by 50% to 60% with ventricular contraction. But this was not thought to have been the cause of his presentation.
On discharge, he required oxygen 4 L/min by nasal cannula. Previously, he had not needed supplemental oxygen.
CURRENT PRESENTATION
The patient described persistent and severe periumbilical abdominal pain during the previous day. It was not associated with eating, and he denied diarrhea, constipation, hematemesis, hematochezia, bright red blood per rectum, or melena. He continued to describe persistent shortness of breath and pleuritic chest pain. His vital signs were as follows:
- Heart rate 104 beats per minute
- Respiratory rate 16 to 20 breaths per minute
- Blood pressure 101–142/62–84 mm Hg
- Oxygen saturation 78% on room air.
His laboratory findings on presentation are shown in Table 1, and his electrocardiogram is shown in Figure 1.
WHAT DOES HIS ELECTROCARDIOGRAM SHOW?
1. Which of the following is the most accurate description of this patient’s electrocardiogram?
- Sinus tachycardia, peaked P waves (P pulmonale) in lead II, and T-wave inversions in the right precordial leads
- Sinus tachycardia and left bundle branch block
- Sinus tachycardia and poor R-wave progression
- Sinus tachycardia and ST elevation in the precordial leads
Our patient’s electrocardiogram shows sinus tachycardia, P pulmonale, T-wave inversion in the right precordial leads (V1–V3), and biphasic T waves in lead V4,, which suggest right ventricular strain.
The rhythm most commonly seen in patients with pulmonary embolism is sinus tachycardia, followed by nonspecific ST-segment or T-wave abnormalities. In one series of patients with acute pulmonary embolism, the classic findings of P pulmonale, right ventricular hypertrophy, right axis deviation, and right bundle branch block were rare (< 6%).1 Thus, these classic findings are not sensitive for the diagnosis of pulmonary embolism, and their absence does not rule it out.
Further studies for our patient
Transthoracic echocardiography was performed to look for evidence of right ventricular strain secondary to the pulmonary embolism.
ECHOCARDIOGRAPHIC SIGNS OF PULMONARY EMBOLISM
2. Which of the following findings on transthoracic echocardiography would not suggest acute pulmonary embolism?
- Midright ventricular wall hypokinesis with apical sparing
- Severe tricuspid regurgitation
- Left ventricular dilation
- Lack of respiratory variation of the inferior vena cava
- Septal wall motion toward the left ventricle
Left ventricular dilation does not suggest acute pulmonary embolism. Echocardiograms of patients with acute submassive pulmonary embolism typically show evidence of right ventricular strain, such as the other entities listed above (midright ventricular hypokinesis with apical sparing, severe tricuspid regurgitation, lack of respiratory variation of the inferior vena cava, and septal wall motion toward the left ventricle).
The degree of right ventricular dysfunction is related to the extent of acute pulmonary vascular occlusion and aids in risk-stratification of patients with acute pulmonary embolism. Midright ventricular wall hypokinesis with apical sparing has been termed the McConnell sign.2
In our patient, transthoracic echocardiography showed:
- Normal left ventricular ejection fraction
- Mild diastolic dysfunction
- Right ventricular dilation with moderately decreased right ventricular systolic function and apical sparing
- Right ventricular systolic pressure 54 mm Hg, consistent with moderate pulmonary hypertension
- Right atrial pressure 10 mm Hg
- No inspiratory collapse of a dilated inferior vena cava
- Mild tricuspid valve regurgitation.
CLASSIFICATION OF ACUTE PULMONARY EMBOLISM
3. Given the above information, how would you classify the patient’s pulmonary embolism?
- Massive
- Submassive
- Low-risk
- Clinically stable
The patient’s pulmonary embolism is submassive.
Historically, the classification of pulmonary embolism was determined by the angiographic thrombus burden. However, this has limited utility because clinical factors (eg, hypotension on initial presentation) have been shown to be better predictors of short-term mortality risk.3
Our patient is characterized as having a submassive pulmonary embolism based on elevated biomarkers (troponin T, N-terminal pro-B-type natriuretic peptide) and right ventricular dysfunction in the absence of hypotension.
ULTRASONOGRAPHY FOR DIAGNOSIS OF DEEP VEIN THROMBOSIS
Venous duplex ultrasonography has become the standard for diagnosis of lower extremity deep vein thrombosis. However, its quality and diagnostic accuracy depend on the skill of the person performing the examination. It is further limited by certain patient characteristics, including severe obesity, edema, and wounds and dressings at the site being examined.5
Our patient underwent duplex ultrasonography of the lower extremities, which demonstrated acute proximal and calf deep vein thrombosis in the right femoral, popliteal, and peroneal veins and no deep vein thrombosis in the left leg.
RISK STRATIFICATION IN ACUTE PULMONARY EMBOLISM
Multiple models exist to estimate the risk of complications in patients with acute pulmonary embolism.
The Bova score6 is based on the following factors:
- Systolic blood pressure 90–100 mm Hg (2 points) (patients with systolic blood pressure lower than 90 mm Hg were excluded from the study from which this score was derived)
- Cardiac troponin elevation (2 points)
- Right ventricular dysfunction on echocardiography or computed tomography (2 points)
- Heart rate 100 beats/min or greater (1 point).
A total score of 0, 1, or 2 (stage I) denotes low risk, 3 or 4 points (stage II) intermediate risk, and more than 4 points (stage III) high risk.
The PESI score (Pulmonary Embolism Severity Index)7 is based on:
- Age (1 point per year)
- Sex (10 points for being male)
- Heart rate 110 per minute or greater (20 points)
- Cancer (30 points)
- Heart failure (10 points)
- Chronic lung disease (10 points)
- Systolic blood pressure less than 100 mm Hg (30 points)
- Respiratory rate at least 30 per minute (20 points)
- Temperature less than 36ºC (20 points)
- Altered mental status (60 points)
- Arterial oxygen saturation less than 90% (20 points).
The total score is broken down into 5 classes: I (< 65 points), II (65–85), III (86–105), IV (106–125), and V (> 126). Classes I and II are low risk, and the higher ones are high risk.
The simplified PESI score8 was developed to more rapidly risk-stratify patients and has been found to be similar to the PESI score in prognostic accuracy. Patients get 1 point for each of the following:
- Age over 80
- Cancer
- Chronic cardiopulmonary disease (heart failure or chronic lung disease)
- Heart rate 110 per minute or greater
- Systolic blood pressure less than 100 mm Hg
- Arterial oxygen saturation less than 90%.
A total score of 0 is low risk; anything higher is high risk.
Back to our patient
Our patient had proximal and calf deep vein thrombosis of the right leg, bilateral submassive pulmonary emboli with associated biomarker elevation and right ventricular dysfunction, and left renal artery thrombosis with infarction. Using the PESI score, his risk of death in the next 30 days was 13.7% and his 30-day risk of a complicated course was 27%. Using the Bova score, his 30-day risk of death was 15.5% and his 30-day risk of a complicated course was 29.2%.6,7
Notably, the patient’s right ventricular function had also been impaired on the echocardiogram performed during his admission 1 month previously. On transthoracic echocardiography during the current admission, the patient was found to have a similar degree of right ventricular dysfunction. This finding, along with the oxygen requirement that developed during the earlier admission, suggested that his pulmonary embolism may have been subacute and that the diagnosis may have been missed during the earlier hospital stay.
The patient was treated with unfractionated heparin. After the hospital’s multidisciplinary pulmonary embolism response team discussed and weighed the above factors, they recommended to not pursue thrombolytic therapy or inferior vena cava filter placement.
Of note, the patient’s pulses in the left lower extremity continued to be weak but palpable, and the left leg was cooler to touch than the right leg.
ASSESSING PERIPHERAL ARTERY DISEASE
4. How should the finding of weak pulses in this patient’s left leg be initially investigated?
- Computed tomographic angiography with runoff
- Ankle-brachial indices with pulse-volume recordings
- Arterial duplex ultrasonography
- Magnetic resonance angiography of the lower extremities
The ankle-brachial index is the initial diagnostic test for assessment of pulse abnormalities and for diagnosis of lower-extremity peripheral artery disease. It is calculated by dividing the higher of the ankle systolic pressures (posterior tibial or dorsalis pedis) by the higher of the 2 brachial pressures (left or right).9 Normal values are between 1.00 and 1.40.
Ankle-brachial indices in our patient
Our patient underwent measurement of his brachial, dorsalis pedis, and posterior tibial artery systolic pressures using blood pressure cuffs and continuous-wave Doppler. Ankle pulse-volume recordings were also obtained.
Given the patient’s poor renal function and concern for acute renal infarction, we thought it best to avoid iodinated or gadolinium contrast, such as with magnetic resonance or computed tomographic angiography.
Segmental leg pressures and pulse-volume recordings can be performed to help localize the level of arterial disease in the extremities, but were not done in this case because of the extensive deep vein thrombosis in the right leg.10,11
Arterial ultrasonography in our patient
Arterial duplex ultrasonography was performed to help determine the location of arterial disease. It showed patent arteries in the right leg. In the left lower extremity there was slow, monophasic blood flow in the distal superficial femoral artery. The popliteal artery was occluded. The posterior tibial artery was occluded at the origin, with reconstitution distally. The peroneal artery was occluded throughout. The anterior tibial artery was patent throughout. The ultrasonographic findings were thought to be suspicious for arterial thromboembolism.
WHAT CAN CAUSE BOTH ARTERIAL AND VENOUS THROMBOSIS?
5. Given that the patient had both arterial thrombosis (renal artery, lower-extremity arteries) and venous thromboembolism (deep vein thrombosis and pulmonary embolism), which of the following would be included in the differential diagnosis?
- Antiphospholipid antibody syndrome
- Protein C or protein S deficiency
- Malignancy
- Paradoxical embolization
- Factor V Leiden mutation
Correct answers include antiphospholipid antibody syndrome, malignancy, and paradoxical embolization.
The differential diagnosis for concomitant venous and arterial thrombosis is broad,12 and includes the following:
- Structural factors: patent foramen ovale, popliteal artery aneurysm
- Malignancy
- Inflammatory diseases: Behçet disease, Buerger disease, inflammatory bowel disease, antiphospholipid antibody syndrome, elevated lipoprotein(a), elevated homocysteine
- Hematologic diseases: myelodysplastic syndrome, disseminated intravascular coagulation, paroxysmal nocturnal hemoglobinuria, heparin-induced thrombocytopenia.
Traditional risk factors for venous thromboembolism include protein C deficiency, protein S deficiency, factor V Leiden mutation, the prothrombin G20210A gene mutation, and others. These are relatively minor risk factors for venous thrombosis and do not pose a risk for arterial thrombosis.12 In contrast, antiphospholipid antibody syndrome and malignancy pose a risk for both venous and arterial thrombosis. Paradoxical embolism is a mechanism by which arterial thrombosis (emboli) can develop in the setting of existing venous thrombosis.12
Our patient underwent testing for antiphospholipid antibodies and lupus anticoagulant, and he was encouraged to undergo age-appropriate cancer screening as an outpatient.12
ANTIPHOSPHOLIPID ANTIBODY SYNDROME
Antiphospholipid antibody syndrome is defined by both clinical and laboratory criteria. Clinical symptoms include vascular thrombosis (arterial, venous, or both) and pregnancy-related complications.13
Laboratory criteria require the presence of antiphospholipid antibodies or lupus anticoagulant. These must be confirmed with repeat testing in 12 weeks. Antiphospholipid antibodies are detected by an enzyme-linked immunosorbent assay; laboratory assessment for the presence of lupus anticoagulant is a stepwise process and relies on 4 criteria:
- There should be prolongation of a phospholipid-dependent clotting test (eg, activated partial thromboplastin time, dilute Russell viper venom time test).
- There must be evidence of an inhibitory activity with mixing study.
- The inhibitor must exhibit phospholipid dependence; that is, with more phospholipid there is shortening of clotting time.
- Specific inhibitors must be excluded, including factor VIII and anticoagulant drugs such as heparin.14–17
Clinically, one should consider antiphospholipid antibody syndrome in patients who have arterial thrombosis, a history of pregnancy morbidity, or unexplained prolongation of activated partial thromboplastin time.13
Antiphospholipid antibodies may be present in up to a quarter of patients with venous thromboembolism, but it is persistent positivity of antibody assays that is associated with increased future risk of venous thromboembolism.19 Of note, the risk of venous thromboembolism in patients with confirmed antiphospholipid antibody syndrome is 10 times higher than in the general population.20
ANTIPHOSPHOLIPID ANTIBODIES ARE NOT ALL THE SAME
6. Which of the following antiphospholipid antibodies have not been associated with an increased thrombotic risk?
- Anti-beta-2-glycoprotein I IgG
- Lupus anticoagulant
- Antiphosphatidylserine
- Anticardiolipin IgM
- Anticardiolipin IgG
The correct answer is antiphosphatidylserine.15
Antiphospholipid antibodies are directed against a portion of select plasma proteins that are uncovered upon phospholipid binding. While lupus anticoagulant, anti-beta-2-glycoprotein I, and anticardiolipin antibodies are associated with thrombosis, antiprothrombin antibodies (including antiprothrombin and antiphosphatidylserine antibodies) are not.15,21
PARADOXICAL EMBOLISM
Patent foramen ovale, a communication between the right and left atrium in the interatrial septum, is associated with an increased risk of paradoxical embolization. The prevalence of patent foramen ovale is estimated to be 27% to 29% in the general population.22 Noncerebral systemic paradoxical embolism occurs less frequently than cerebral embolism, accounting for approximately 5% to 10% of paradoxical emboli.22
To evaluate for patent foramen ovale, transthoracic echocardiography is performed with a bubble (agitated saline contrast) study to assess for interatrial shunting. Transesophageal echocardiography or transcranial Doppler bubble studies may also be performed.
Although patent foramen ovale is most commonly associated with cerebral embolism, peripheral emboli can occur. Some research suggests that this may be a more common cause of arterial thromboembolism in younger patients. There have also been reports of other sites of systemic embolization, including the renal artery.12
Back to our patient
Initial antiphospholipid antibody testing was positive for lupus anticoagulant. Anticardiolipin and anti-beta-2-glycoprotein I antibodies were not detected.
Transesophageal echocardiography revealed a patent foramen ovale with a highly mobile atrial septum (atrial septal aneurysm).
The patient was treated with intravenous unfractionated heparin with bridging to warfarin with a target international normalized ratio (INR) of 2 to 3. His renal artery infarction and his lower-extremity arterial thromboembolic event were conservatively managed. His respiratory status improved, and he no longer required supplemental oxygen. His creatinine peaked at 1.7 mg/dL during his admission and improved to 1.2 mg/dL before he was discharged.
At follow-up, repeat echocardiography showed that his right ventricular systolic pressure had improved (decreased) to 37 mm Hg from 54 mm Hg. Repeat confirmatory testing was positive for lupus anticoagulant 12 weeks later. He has been maintained on warfarin with an INR goal of 2 to 3 as well as low-dose aspirin with plans for long-term anticoagulation. We decided to keep the patient on anticoagulation indefinitely with warfarin; he was not a candidate for a direct oral anticoagulant, given limited data on the use of these agents in the setting of lupus anticoagulant and antiphospholipid antibody syndrome.
SUMMARY OF CASE
In summary, this patient was a 75-year-old man with COPD who presented with abdominal pain. He was noted to have a left renal infarction, extensive unprovoked lower-extremity deep vein thrombosis with pulmonary emboli, and lower limb arterial thromboembolism.
He also had an underlying hypercoagulable state—antiphospholipid antibody syndrome—that predisposed him to both arterial and venous thrombosis. He was ultimately found to have a patent foramen ovale, which further increased the risk of arterial thrombosis by facilitating paradoxical embolization of venous thrombi. It is not certain whether the renal infarction and leg artery thrombi were due to paradoxical embolism or to in situ thrombosis, but we believe that it was most likely paradoxical embolization.
A 75-year-old man presented to the emergency department for evaluation of abdominal pain. He had stage 3 chronic obstructive pulmonary disease (COPD), with a forced expiratory volume in 1 second of 33%.
PREVIOUS HOSPITALIZATION
Aside from his COPD, he had been healthy until 1 month earlier, when he had been hospitalized because of shortness of breath and chest pressure with exertion. His troponin T level had been elevated, peaking at 0.117 ng/mL (reference range 0–0.029).
Left heart catheterization had shown no significant coronary artery disease. A myocardial bridge of the distal left anterior descending coronary artery had been seen, so that the artery appeared to be narrowed by 50% to 60% with ventricular contraction. But this was not thought to have been the cause of his presentation.
On discharge, he required oxygen 4 L/min by nasal cannula. Previously, he had not needed supplemental oxygen.
CURRENT PRESENTATION
The patient described persistent and severe periumbilical abdominal pain during the previous day. It was not associated with eating, and he denied diarrhea, constipation, hematemesis, hematochezia, bright red blood per rectum, or melena. He continued to describe persistent shortness of breath and pleuritic chest pain. His vital signs were as follows:
- Heart rate 104 beats per minute
- Respiratory rate 16 to 20 breaths per minute
- Blood pressure 101–142/62–84 mm Hg
- Oxygen saturation 78% on room air.
His laboratory findings on presentation are shown in Table 1, and his electrocardiogram is shown in Figure 1.
WHAT DOES HIS ELECTROCARDIOGRAM SHOW?
1. Which of the following is the most accurate description of this patient’s electrocardiogram?
- Sinus tachycardia, peaked P waves (P pulmonale) in lead II, and T-wave inversions in the right precordial leads
- Sinus tachycardia and left bundle branch block
- Sinus tachycardia and poor R-wave progression
- Sinus tachycardia and ST elevation in the precordial leads
Our patient’s electrocardiogram shows sinus tachycardia, P pulmonale, T-wave inversion in the right precordial leads (V1–V3), and biphasic T waves in lead V4,, which suggest right ventricular strain.
The rhythm most commonly seen in patients with pulmonary embolism is sinus tachycardia, followed by nonspecific ST-segment or T-wave abnormalities. In one series of patients with acute pulmonary embolism, the classic findings of P pulmonale, right ventricular hypertrophy, right axis deviation, and right bundle branch block were rare (< 6%).1 Thus, these classic findings are not sensitive for the diagnosis of pulmonary embolism, and their absence does not rule it out.
Further studies for our patient
Transthoracic echocardiography was performed to look for evidence of right ventricular strain secondary to the pulmonary embolism.
ECHOCARDIOGRAPHIC SIGNS OF PULMONARY EMBOLISM
2. Which of the following findings on transthoracic echocardiography would not suggest acute pulmonary embolism?
- Midright ventricular wall hypokinesis with apical sparing
- Severe tricuspid regurgitation
- Left ventricular dilation
- Lack of respiratory variation of the inferior vena cava
- Septal wall motion toward the left ventricle
Left ventricular dilation does not suggest acute pulmonary embolism. Echocardiograms of patients with acute submassive pulmonary embolism typically show evidence of right ventricular strain, such as the other entities listed above (midright ventricular hypokinesis with apical sparing, severe tricuspid regurgitation, lack of respiratory variation of the inferior vena cava, and septal wall motion toward the left ventricle).
The degree of right ventricular dysfunction is related to the extent of acute pulmonary vascular occlusion and aids in risk-stratification of patients with acute pulmonary embolism. Midright ventricular wall hypokinesis with apical sparing has been termed the McConnell sign.2
In our patient, transthoracic echocardiography showed:
- Normal left ventricular ejection fraction
- Mild diastolic dysfunction
- Right ventricular dilation with moderately decreased right ventricular systolic function and apical sparing
- Right ventricular systolic pressure 54 mm Hg, consistent with moderate pulmonary hypertension
- Right atrial pressure 10 mm Hg
- No inspiratory collapse of a dilated inferior vena cava
- Mild tricuspid valve regurgitation.
CLASSIFICATION OF ACUTE PULMONARY EMBOLISM
3. Given the above information, how would you classify the patient’s pulmonary embolism?
- Massive
- Submassive
- Low-risk
- Clinically stable
The patient’s pulmonary embolism is submassive.
Historically, the classification of pulmonary embolism was determined by the angiographic thrombus burden. However, this has limited utility because clinical factors (eg, hypotension on initial presentation) have been shown to be better predictors of short-term mortality risk.3
Our patient is characterized as having a submassive pulmonary embolism based on elevated biomarkers (troponin T, N-terminal pro-B-type natriuretic peptide) and right ventricular dysfunction in the absence of hypotension.
ULTRASONOGRAPHY FOR DIAGNOSIS OF DEEP VEIN THROMBOSIS
Venous duplex ultrasonography has become the standard for diagnosis of lower extremity deep vein thrombosis. However, its quality and diagnostic accuracy depend on the skill of the person performing the examination. It is further limited by certain patient characteristics, including severe obesity, edema, and wounds and dressings at the site being examined.5
Our patient underwent duplex ultrasonography of the lower extremities, which demonstrated acute proximal and calf deep vein thrombosis in the right femoral, popliteal, and peroneal veins and no deep vein thrombosis in the left leg.
RISK STRATIFICATION IN ACUTE PULMONARY EMBOLISM
Multiple models exist to estimate the risk of complications in patients with acute pulmonary embolism.
The Bova score6 is based on the following factors:
- Systolic blood pressure 90–100 mm Hg (2 points) (patients with systolic blood pressure lower than 90 mm Hg were excluded from the study from which this score was derived)
- Cardiac troponin elevation (2 points)
- Right ventricular dysfunction on echocardiography or computed tomography (2 points)
- Heart rate 100 beats/min or greater (1 point).
A total score of 0, 1, or 2 (stage I) denotes low risk, 3 or 4 points (stage II) intermediate risk, and more than 4 points (stage III) high risk.
The PESI score (Pulmonary Embolism Severity Index)7 is based on:
- Age (1 point per year)
- Sex (10 points for being male)
- Heart rate 110 per minute or greater (20 points)
- Cancer (30 points)
- Heart failure (10 points)
- Chronic lung disease (10 points)
- Systolic blood pressure less than 100 mm Hg (30 points)
- Respiratory rate at least 30 per minute (20 points)
- Temperature less than 36ºC (20 points)
- Altered mental status (60 points)
- Arterial oxygen saturation less than 90% (20 points).
The total score is broken down into 5 classes: I (< 65 points), II (65–85), III (86–105), IV (106–125), and V (> 126). Classes I and II are low risk, and the higher ones are high risk.
The simplified PESI score8 was developed to more rapidly risk-stratify patients and has been found to be similar to the PESI score in prognostic accuracy. Patients get 1 point for each of the following:
- Age over 80
- Cancer
- Chronic cardiopulmonary disease (heart failure or chronic lung disease)
- Heart rate 110 per minute or greater
- Systolic blood pressure less than 100 mm Hg
- Arterial oxygen saturation less than 90%.
A total score of 0 is low risk; anything higher is high risk.
Back to our patient
Our patient had proximal and calf deep vein thrombosis of the right leg, bilateral submassive pulmonary emboli with associated biomarker elevation and right ventricular dysfunction, and left renal artery thrombosis with infarction. Using the PESI score, his risk of death in the next 30 days was 13.7% and his 30-day risk of a complicated course was 27%. Using the Bova score, his 30-day risk of death was 15.5% and his 30-day risk of a complicated course was 29.2%.6,7
Notably, the patient’s right ventricular function had also been impaired on the echocardiogram performed during his admission 1 month previously. On transthoracic echocardiography during the current admission, the patient was found to have a similar degree of right ventricular dysfunction. This finding, along with the oxygen requirement that developed during the earlier admission, suggested that his pulmonary embolism may have been subacute and that the diagnosis may have been missed during the earlier hospital stay.
The patient was treated with unfractionated heparin. After the hospital’s multidisciplinary pulmonary embolism response team discussed and weighed the above factors, they recommended to not pursue thrombolytic therapy or inferior vena cava filter placement.
Of note, the patient’s pulses in the left lower extremity continued to be weak but palpable, and the left leg was cooler to touch than the right leg.
ASSESSING PERIPHERAL ARTERY DISEASE
4. How should the finding of weak pulses in this patient’s left leg be initially investigated?
- Computed tomographic angiography with runoff
- Ankle-brachial indices with pulse-volume recordings
- Arterial duplex ultrasonography
- Magnetic resonance angiography of the lower extremities
The ankle-brachial index is the initial diagnostic test for assessment of pulse abnormalities and for diagnosis of lower-extremity peripheral artery disease. It is calculated by dividing the higher of the ankle systolic pressures (posterior tibial or dorsalis pedis) by the higher of the 2 brachial pressures (left or right).9 Normal values are between 1.00 and 1.40.
Ankle-brachial indices in our patient
Our patient underwent measurement of his brachial, dorsalis pedis, and posterior tibial artery systolic pressures using blood pressure cuffs and continuous-wave Doppler. Ankle pulse-volume recordings were also obtained.
Given the patient’s poor renal function and concern for acute renal infarction, we thought it best to avoid iodinated or gadolinium contrast, such as with magnetic resonance or computed tomographic angiography.
Segmental leg pressures and pulse-volume recordings can be performed to help localize the level of arterial disease in the extremities, but were not done in this case because of the extensive deep vein thrombosis in the right leg.10,11
Arterial ultrasonography in our patient
Arterial duplex ultrasonography was performed to help determine the location of arterial disease. It showed patent arteries in the right leg. In the left lower extremity there was slow, monophasic blood flow in the distal superficial femoral artery. The popliteal artery was occluded. The posterior tibial artery was occluded at the origin, with reconstitution distally. The peroneal artery was occluded throughout. The anterior tibial artery was patent throughout. The ultrasonographic findings were thought to be suspicious for arterial thromboembolism.
WHAT CAN CAUSE BOTH ARTERIAL AND VENOUS THROMBOSIS?
5. Given that the patient had both arterial thrombosis (renal artery, lower-extremity arteries) and venous thromboembolism (deep vein thrombosis and pulmonary embolism), which of the following would be included in the differential diagnosis?
- Antiphospholipid antibody syndrome
- Protein C or protein S deficiency
- Malignancy
- Paradoxical embolization
- Factor V Leiden mutation
Correct answers include antiphospholipid antibody syndrome, malignancy, and paradoxical embolization.
The differential diagnosis for concomitant venous and arterial thrombosis is broad,12 and includes the following:
- Structural factors: patent foramen ovale, popliteal artery aneurysm
- Malignancy
- Inflammatory diseases: Behçet disease, Buerger disease, inflammatory bowel disease, antiphospholipid antibody syndrome, elevated lipoprotein(a), elevated homocysteine
- Hematologic diseases: myelodysplastic syndrome, disseminated intravascular coagulation, paroxysmal nocturnal hemoglobinuria, heparin-induced thrombocytopenia.
Traditional risk factors for venous thromboembolism include protein C deficiency, protein S deficiency, factor V Leiden mutation, the prothrombin G20210A gene mutation, and others. These are relatively minor risk factors for venous thrombosis and do not pose a risk for arterial thrombosis.12 In contrast, antiphospholipid antibody syndrome and malignancy pose a risk for both venous and arterial thrombosis. Paradoxical embolism is a mechanism by which arterial thrombosis (emboli) can develop in the setting of existing venous thrombosis.12
Our patient underwent testing for antiphospholipid antibodies and lupus anticoagulant, and he was encouraged to undergo age-appropriate cancer screening as an outpatient.12
ANTIPHOSPHOLIPID ANTIBODY SYNDROME
Antiphospholipid antibody syndrome is defined by both clinical and laboratory criteria. Clinical symptoms include vascular thrombosis (arterial, venous, or both) and pregnancy-related complications.13
Laboratory criteria require the presence of antiphospholipid antibodies or lupus anticoagulant. These must be confirmed with repeat testing in 12 weeks. Antiphospholipid antibodies are detected by an enzyme-linked immunosorbent assay; laboratory assessment for the presence of lupus anticoagulant is a stepwise process and relies on 4 criteria:
- There should be prolongation of a phospholipid-dependent clotting test (eg, activated partial thromboplastin time, dilute Russell viper venom time test).
- There must be evidence of an inhibitory activity with mixing study.
- The inhibitor must exhibit phospholipid dependence; that is, with more phospholipid there is shortening of clotting time.
- Specific inhibitors must be excluded, including factor VIII and anticoagulant drugs such as heparin.14–17
Clinically, one should consider antiphospholipid antibody syndrome in patients who have arterial thrombosis, a history of pregnancy morbidity, or unexplained prolongation of activated partial thromboplastin time.13
Antiphospholipid antibodies may be present in up to a quarter of patients with venous thromboembolism, but it is persistent positivity of antibody assays that is associated with increased future risk of venous thromboembolism.19 Of note, the risk of venous thromboembolism in patients with confirmed antiphospholipid antibody syndrome is 10 times higher than in the general population.20
ANTIPHOSPHOLIPID ANTIBODIES ARE NOT ALL THE SAME
6. Which of the following antiphospholipid antibodies have not been associated with an increased thrombotic risk?
- Anti-beta-2-glycoprotein I IgG
- Lupus anticoagulant
- Antiphosphatidylserine
- Anticardiolipin IgM
- Anticardiolipin IgG
The correct answer is antiphosphatidylserine.15
Antiphospholipid antibodies are directed against a portion of select plasma proteins that are uncovered upon phospholipid binding. While lupus anticoagulant, anti-beta-2-glycoprotein I, and anticardiolipin antibodies are associated with thrombosis, antiprothrombin antibodies (including antiprothrombin and antiphosphatidylserine antibodies) are not.15,21
PARADOXICAL EMBOLISM
Patent foramen ovale, a communication between the right and left atrium in the interatrial septum, is associated with an increased risk of paradoxical embolization. The prevalence of patent foramen ovale is estimated to be 27% to 29% in the general population.22 Noncerebral systemic paradoxical embolism occurs less frequently than cerebral embolism, accounting for approximately 5% to 10% of paradoxical emboli.22
To evaluate for patent foramen ovale, transthoracic echocardiography is performed with a bubble (agitated saline contrast) study to assess for interatrial shunting. Transesophageal echocardiography or transcranial Doppler bubble studies may also be performed.
Although patent foramen ovale is most commonly associated with cerebral embolism, peripheral emboli can occur. Some research suggests that this may be a more common cause of arterial thromboembolism in younger patients. There have also been reports of other sites of systemic embolization, including the renal artery.12
Back to our patient
Initial antiphospholipid antibody testing was positive for lupus anticoagulant. Anticardiolipin and anti-beta-2-glycoprotein I antibodies were not detected.
Transesophageal echocardiography revealed a patent foramen ovale with a highly mobile atrial septum (atrial septal aneurysm).
The patient was treated with intravenous unfractionated heparin with bridging to warfarin with a target international normalized ratio (INR) of 2 to 3. His renal artery infarction and his lower-extremity arterial thromboembolic event were conservatively managed. His respiratory status improved, and he no longer required supplemental oxygen. His creatinine peaked at 1.7 mg/dL during his admission and improved to 1.2 mg/dL before he was discharged.
At follow-up, repeat echocardiography showed that his right ventricular systolic pressure had improved (decreased) to 37 mm Hg from 54 mm Hg. Repeat confirmatory testing was positive for lupus anticoagulant 12 weeks later. He has been maintained on warfarin with an INR goal of 2 to 3 as well as low-dose aspirin with plans for long-term anticoagulation. We decided to keep the patient on anticoagulation indefinitely with warfarin; he was not a candidate for a direct oral anticoagulant, given limited data on the use of these agents in the setting of lupus anticoagulant and antiphospholipid antibody syndrome.
SUMMARY OF CASE
In summary, this patient was a 75-year-old man with COPD who presented with abdominal pain. He was noted to have a left renal infarction, extensive unprovoked lower-extremity deep vein thrombosis with pulmonary emboli, and lower limb arterial thromboembolism.
He also had an underlying hypercoagulable state—antiphospholipid antibody syndrome—that predisposed him to both arterial and venous thrombosis. He was ultimately found to have a patent foramen ovale, which further increased the risk of arterial thrombosis by facilitating paradoxical embolization of venous thrombi. It is not certain whether the renal infarction and leg artery thrombi were due to paradoxical embolism or to in situ thrombosis, but we believe that it was most likely paradoxical embolization.
- Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest 1991; 100:598–603.
- Alsoos F, Khaddam A. Echocardiographic evaluation methods for right ventricular function. J Echocardiogr 2015; 13:43–51.
- Jaff MR, McMurtry MS, Archer SL, et al; American Heart Association Council on Cardiopulmonary, Critical Care, Perioperative and Resuscitation; American Heart Association Council on Peripheral Vascular Disease; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation 2011; 123:1788–1830.
- Heit JA, Silverstein MD, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ 3rd. Risk factors for deep vein thrombosis and pulmonary embolism: a population-based case-control study. Arch Intern Med 2000; 160:809–815.
- Gornik HL, Sharma AM. Duplex ultrasound in the diagnosis of lower-extremity deep venous thrombosis. Circulation 2014; 129:917–921.
- Fernández C, Bova C, Sanchez O, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest 2015; 148:211–218.
- Aujesky D, Perrier A, Roy PM, et al. Validation of a clinical prognostic model to identify low-risk patients with pulmonary embolism. J Intern Med 2007; 261:597–604.
- Jiménez D, Aujesky D, Moores L, et al; RIETE Investigators. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med 2010; 170:1383–1389.
- Kim ES, Wattanakit K, Gornik HL. Using the ankle-brachial index to diagnose peripheral artery disease and assess cardiovascular risk. Cleve Clin J Med 2012; 79:651–661.
- Jaff MR. Lower extremity arterial disease. Diagnostic aspects. Cardiol Clin 2002; 20:491–500.
- Rooke TW, Hirsch AT, Misra S, et al; American College of Cardiology Foundation Task Force; American Heart Association Task Force. Management of patients with peripheral artery disease (compilation of 2005 and 2011 ACCF/AHA Guideline Recommendations): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 61:1555–1570.
- Lichtin A, Bartholomew J. The coagulation consult: a case-based guide. New York, NY: Springer; 2014.
- Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med 2002; 346:752–763.
- Brandt JT, Triplett DA, Alving B, Scharrer I. Criteria for the diagnosis of lupus anticoagulants: an update. On behalf of the Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the ISTH. Thromb Haemost 1995; 74:1185–1190.
- Miyakis S, Lockshin M, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost 2006; 4:295–306.
- Pengo V, Tripodi A, Reber G, et al; Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis. Update of the guidelines for lupus anticoagulant detection. Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis. J Thromb Haemost 2009; 7:1737–1740.
- Nichols WL, Kottke-Marchant K, Ledford-Kraemer MR, Homburger HA, Cardel LK. Lupus anticoagulants, antiphospholipid antibodies, and antiphospholipid syndrome. In: Kottke-Marchant K, Davis BH, editors. Laboratory Hematology Practice. Hoboken, New Jersey: Blackwell Publishing, Ltd.; 2012:509–525.
- Houghton DE, Moll S. Antiphospholipid antibodies. Vasc Med 2017; 22:545–550.
- Roldan V, Lecumberri R, Muñoz-Torrero JFS, et al; RIETE Investigators. Thrombophilia testing in patients with venous thromboembolism. Findings from the RIETE registry. Thromb Res 2009; 124:174–177.
- Wahl DG, Guillemin F, de Maistre E, Perret-Guillaume C, Lecompte T, Thibaut G. Meta-analysis of the risk of venous thrombosis in individuals with antiphospholipid antibodies without underlying autoimmune disease or previous thrombosis. Lupus 1998; 7:15–22.
- Love PE, Santoro SA. Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders. Prevalence and clinical significance. Ann Intern Med 1990; 112:682–698.
- Thompson T, Evans W. Paradoxical embolism. QJM 1930; os-23:135–150.
- Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest 1991; 100:598–603.
- Alsoos F, Khaddam A. Echocardiographic evaluation methods for right ventricular function. J Echocardiogr 2015; 13:43–51.
- Jaff MR, McMurtry MS, Archer SL, et al; American Heart Association Council on Cardiopulmonary, Critical Care, Perioperative and Resuscitation; American Heart Association Council on Peripheral Vascular Disease; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation 2011; 123:1788–1830.
- Heit JA, Silverstein MD, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ 3rd. Risk factors for deep vein thrombosis and pulmonary embolism: a population-based case-control study. Arch Intern Med 2000; 160:809–815.
- Gornik HL, Sharma AM. Duplex ultrasound in the diagnosis of lower-extremity deep venous thrombosis. Circulation 2014; 129:917–921.
- Fernández C, Bova C, Sanchez O, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest 2015; 148:211–218.
- Aujesky D, Perrier A, Roy PM, et al. Validation of a clinical prognostic model to identify low-risk patients with pulmonary embolism. J Intern Med 2007; 261:597–604.
- Jiménez D, Aujesky D, Moores L, et al; RIETE Investigators. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med 2010; 170:1383–1389.
- Kim ES, Wattanakit K, Gornik HL. Using the ankle-brachial index to diagnose peripheral artery disease and assess cardiovascular risk. Cleve Clin J Med 2012; 79:651–661.
- Jaff MR. Lower extremity arterial disease. Diagnostic aspects. Cardiol Clin 2002; 20:491–500.
- Rooke TW, Hirsch AT, Misra S, et al; American College of Cardiology Foundation Task Force; American Heart Association Task Force. Management of patients with peripheral artery disease (compilation of 2005 and 2011 ACCF/AHA Guideline Recommendations): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 61:1555–1570.
- Lichtin A, Bartholomew J. The coagulation consult: a case-based guide. New York, NY: Springer; 2014.
- Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med 2002; 346:752–763.
- Brandt JT, Triplett DA, Alving B, Scharrer I. Criteria for the diagnosis of lupus anticoagulants: an update. On behalf of the Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the ISTH. Thromb Haemost 1995; 74:1185–1190.
- Miyakis S, Lockshin M, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost 2006; 4:295–306.
- Pengo V, Tripodi A, Reber G, et al; Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis. Update of the guidelines for lupus anticoagulant detection. Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the International Society on Thrombosis and Haemostasis. J Thromb Haemost 2009; 7:1737–1740.
- Nichols WL, Kottke-Marchant K, Ledford-Kraemer MR, Homburger HA, Cardel LK. Lupus anticoagulants, antiphospholipid antibodies, and antiphospholipid syndrome. In: Kottke-Marchant K, Davis BH, editors. Laboratory Hematology Practice. Hoboken, New Jersey: Blackwell Publishing, Ltd.; 2012:509–525.
- Houghton DE, Moll S. Antiphospholipid antibodies. Vasc Med 2017; 22:545–550.
- Roldan V, Lecumberri R, Muñoz-Torrero JFS, et al; RIETE Investigators. Thrombophilia testing in patients with venous thromboembolism. Findings from the RIETE registry. Thromb Res 2009; 124:174–177.
- Wahl DG, Guillemin F, de Maistre E, Perret-Guillaume C, Lecompte T, Thibaut G. Meta-analysis of the risk of venous thrombosis in individuals with antiphospholipid antibodies without underlying autoimmune disease or previous thrombosis. Lupus 1998; 7:15–22.
- Love PE, Santoro SA. Antiphospholipid antibodies: anticardiolipin and the lupus anticoagulant in systemic lupus erythematosus (SLE) and in non-SLE disorders. Prevalence and clinical significance. Ann Intern Med 1990; 112:682–698.
- Thompson T, Evans W. Paradoxical embolism. QJM 1930; os-23:135–150.
Erythema ab igne
A 42-year-old man presented to his primary care physician with chief complaints of fatigue and worsening of severe chronic thoracic and lumbar back pain that he attributed to routine daily activity at his job, not to any specific antecedent trauma. Over-the-counter nonsteroidal anti-inflammatory agents and analgesics had provided moderate pain relief. A heating pad, which he had been using daily, often for several hours at a time, offered significant pain relief.
AN UNCOMMON CAUSE OF HYPERPIGMENTATION
Erythema ab igne is an uncommon thermally induced eruption associated with periods of repeated exposure of the skin to warm stimuli. It has been reported after holding a kerosene stove between the knees1 and after balancing a laptop computer on the thighs,2 but hot water bottles and heating pads3 are the most commonly reported causes.
Clinical findings and a compatible history are critical to the diagnosis. The involved area should have a history of heat exposure, followed by the development of asymptomatic persistent erythema and, in long-standing cases, hyperpigmentation in a distinct reticulate (netlike) distribution. Telangiectasias and atrophy may occur in advanced disease. In extreme presentations, bullae may develop.4
OTHER POTENTIAL CAUSES
Based on the history and findings on physical examination, conditions to consider in the differential diagnosis can include reticular eruptions such as livedo reticularis (associated with cold temperature changes), hypercoagulable states (antiphospholipid antibody syndrome, Sneddon syndrome), and connective tissue diseases (systemic lupus erythematosus).
Other causes of hyperpigmentation such as postinflammatory hyperpigmentation (associated with a prior eruption) and stasis dermatitis (found in areas of poor venous drainage) may also be considered.
The frequent application of an external agent such as a drug or lotion, or even the use of a heating pad if it is made from rubber or is finished with an allergenic dye, raises the possibility of allergic contact dermatitis, but this was not likely in our patient because of the lack of pruritus and active dermatitis. Repeated application of a stimulus such as a heating pad with frequent rubbing can also precipitate lichen simplex chronicus, with thickened, hyperkeratotic skin. Another cause of hyperpigmentation on the back is notalgia paresthetica, which is typically unilateral, over the shoulder blades, and is associated with pain, pruritus, or other neurologic symptoms. Again, these were not present in our patient.
TREATMENT OPTIONS
The first-line treatment for erythema ab igne is to stop exposure to the offending heat source. The hyperpigmentation may slowly fade over several years. Cosmetic treatments such as laser therapy and depigmenting creams can be tried for persistent hyperpigmentation. Patients may be monitored for the small increased risk of cutaneous malignancies—particularly squamous cell carcinoma—arising within regions of erythema ab igne, particularly if atrophic or nonhealing in nature.
Clinicians should recognize the differential diagnosis of erythema ab igne in the appropriate clinical setting and provide the patient counseling regarding this preventable dermatosis.
- Arnold AW, Itin PH. Laptop computer-induced erythema ab igne in a child and review of the literature. Pediatrics 2010; 126:e1227–e1230.
- Milgrom Y, Sabag T, Zlotogorski A, Heyman SN. Erythema ab igne of shins: a kerosene stove-induced prototype in diabetics. J Postgrad Med 2013; 59:56–57.
- Waldorf DS, Rast MF, Garofalo VJ. Heating-pad erythematous dermatitis ‘erythema ab igne.’ JAMA 1971; 218:1704.
- Asilian A, Abtahi-Naeini B, Pourazizi M, Rakhshanpour M. Rapid onset of bullous erythema ab igne: a case report of atypical presentation. Indian J Dermatol 2015; 60:325.
A 42-year-old man presented to his primary care physician with chief complaints of fatigue and worsening of severe chronic thoracic and lumbar back pain that he attributed to routine daily activity at his job, not to any specific antecedent trauma. Over-the-counter nonsteroidal anti-inflammatory agents and analgesics had provided moderate pain relief. A heating pad, which he had been using daily, often for several hours at a time, offered significant pain relief.
AN UNCOMMON CAUSE OF HYPERPIGMENTATION
Erythema ab igne is an uncommon thermally induced eruption associated with periods of repeated exposure of the skin to warm stimuli. It has been reported after holding a kerosene stove between the knees1 and after balancing a laptop computer on the thighs,2 but hot water bottles and heating pads3 are the most commonly reported causes.
Clinical findings and a compatible history are critical to the diagnosis. The involved area should have a history of heat exposure, followed by the development of asymptomatic persistent erythema and, in long-standing cases, hyperpigmentation in a distinct reticulate (netlike) distribution. Telangiectasias and atrophy may occur in advanced disease. In extreme presentations, bullae may develop.4
OTHER POTENTIAL CAUSES
Based on the history and findings on physical examination, conditions to consider in the differential diagnosis can include reticular eruptions such as livedo reticularis (associated with cold temperature changes), hypercoagulable states (antiphospholipid antibody syndrome, Sneddon syndrome), and connective tissue diseases (systemic lupus erythematosus).
Other causes of hyperpigmentation such as postinflammatory hyperpigmentation (associated with a prior eruption) and stasis dermatitis (found in areas of poor venous drainage) may also be considered.
The frequent application of an external agent such as a drug or lotion, or even the use of a heating pad if it is made from rubber or is finished with an allergenic dye, raises the possibility of allergic contact dermatitis, but this was not likely in our patient because of the lack of pruritus and active dermatitis. Repeated application of a stimulus such as a heating pad with frequent rubbing can also precipitate lichen simplex chronicus, with thickened, hyperkeratotic skin. Another cause of hyperpigmentation on the back is notalgia paresthetica, which is typically unilateral, over the shoulder blades, and is associated with pain, pruritus, or other neurologic symptoms. Again, these were not present in our patient.
TREATMENT OPTIONS
The first-line treatment for erythema ab igne is to stop exposure to the offending heat source. The hyperpigmentation may slowly fade over several years. Cosmetic treatments such as laser therapy and depigmenting creams can be tried for persistent hyperpigmentation. Patients may be monitored for the small increased risk of cutaneous malignancies—particularly squamous cell carcinoma—arising within regions of erythema ab igne, particularly if atrophic or nonhealing in nature.
Clinicians should recognize the differential diagnosis of erythema ab igne in the appropriate clinical setting and provide the patient counseling regarding this preventable dermatosis.
A 42-year-old man presented to his primary care physician with chief complaints of fatigue and worsening of severe chronic thoracic and lumbar back pain that he attributed to routine daily activity at his job, not to any specific antecedent trauma. Over-the-counter nonsteroidal anti-inflammatory agents and analgesics had provided moderate pain relief. A heating pad, which he had been using daily, often for several hours at a time, offered significant pain relief.
AN UNCOMMON CAUSE OF HYPERPIGMENTATION
Erythema ab igne is an uncommon thermally induced eruption associated with periods of repeated exposure of the skin to warm stimuli. It has been reported after holding a kerosene stove between the knees1 and after balancing a laptop computer on the thighs,2 but hot water bottles and heating pads3 are the most commonly reported causes.
Clinical findings and a compatible history are critical to the diagnosis. The involved area should have a history of heat exposure, followed by the development of asymptomatic persistent erythema and, in long-standing cases, hyperpigmentation in a distinct reticulate (netlike) distribution. Telangiectasias and atrophy may occur in advanced disease. In extreme presentations, bullae may develop.4
OTHER POTENTIAL CAUSES
Based on the history and findings on physical examination, conditions to consider in the differential diagnosis can include reticular eruptions such as livedo reticularis (associated with cold temperature changes), hypercoagulable states (antiphospholipid antibody syndrome, Sneddon syndrome), and connective tissue diseases (systemic lupus erythematosus).
Other causes of hyperpigmentation such as postinflammatory hyperpigmentation (associated with a prior eruption) and stasis dermatitis (found in areas of poor venous drainage) may also be considered.
The frequent application of an external agent such as a drug or lotion, or even the use of a heating pad if it is made from rubber or is finished with an allergenic dye, raises the possibility of allergic contact dermatitis, but this was not likely in our patient because of the lack of pruritus and active dermatitis. Repeated application of a stimulus such as a heating pad with frequent rubbing can also precipitate lichen simplex chronicus, with thickened, hyperkeratotic skin. Another cause of hyperpigmentation on the back is notalgia paresthetica, which is typically unilateral, over the shoulder blades, and is associated with pain, pruritus, or other neurologic symptoms. Again, these were not present in our patient.
TREATMENT OPTIONS
The first-line treatment for erythema ab igne is to stop exposure to the offending heat source. The hyperpigmentation may slowly fade over several years. Cosmetic treatments such as laser therapy and depigmenting creams can be tried for persistent hyperpigmentation. Patients may be monitored for the small increased risk of cutaneous malignancies—particularly squamous cell carcinoma—arising within regions of erythema ab igne, particularly if atrophic or nonhealing in nature.
Clinicians should recognize the differential diagnosis of erythema ab igne in the appropriate clinical setting and provide the patient counseling regarding this preventable dermatosis.
- Arnold AW, Itin PH. Laptop computer-induced erythema ab igne in a child and review of the literature. Pediatrics 2010; 126:e1227–e1230.
- Milgrom Y, Sabag T, Zlotogorski A, Heyman SN. Erythema ab igne of shins: a kerosene stove-induced prototype in diabetics. J Postgrad Med 2013; 59:56–57.
- Waldorf DS, Rast MF, Garofalo VJ. Heating-pad erythematous dermatitis ‘erythema ab igne.’ JAMA 1971; 218:1704.
- Asilian A, Abtahi-Naeini B, Pourazizi M, Rakhshanpour M. Rapid onset of bullous erythema ab igne: a case report of atypical presentation. Indian J Dermatol 2015; 60:325.
- Arnold AW, Itin PH. Laptop computer-induced erythema ab igne in a child and review of the literature. Pediatrics 2010; 126:e1227–e1230.
- Milgrom Y, Sabag T, Zlotogorski A, Heyman SN. Erythema ab igne of shins: a kerosene stove-induced prototype in diabetics. J Postgrad Med 2013; 59:56–57.
- Waldorf DS, Rast MF, Garofalo VJ. Heating-pad erythematous dermatitis ‘erythema ab igne.’ JAMA 1971; 218:1704.
- Asilian A, Abtahi-Naeini B, Pourazizi M, Rakhshanpour M. Rapid onset of bullous erythema ab igne: a case report of atypical presentation. Indian J Dermatol 2015; 60:325.
Peer-reviewers for 2017
We thank those who reviewed manuscripts submitted to the Cleveland Clinic Journal of Medicine in 2017. Reviewing papers for the Journal—both for specialty content and for relevance to our readership—is an arduous task that involves considerable time and effort. Our publication decisions depend in no small part on the timely efforts of reviewers, and we are indebted to them for contributing their expertise this past year.
—Brian F. Mandell, MD, PhD, Editor in Chief
We thank those who reviewed manuscripts submitted to the Cleveland Clinic Journal of Medicine in 2017. Reviewing papers for the Journal—both for specialty content and for relevance to our readership—is an arduous task that involves considerable time and effort. Our publication decisions depend in no small part on the timely efforts of reviewers, and we are indebted to them for contributing their expertise this past year.
—Brian F. Mandell, MD, PhD, Editor in Chief
We thank those who reviewed manuscripts submitted to the Cleveland Clinic Journal of Medicine in 2017. Reviewing papers for the Journal—both for specialty content and for relevance to our readership—is an arduous task that involves considerable time and effort. Our publication decisions depend in no small part on the timely efforts of reviewers, and we are indebted to them for contributing their expertise this past year.
—Brian F. Mandell, MD, PhD, Editor in Chief
MDedge Daily News: State of the Union gives health care little love
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Health care gets little love in the State of the Union, the head of the CDC resigns, cancer rates after colonoscopy may heading in the wrong direction, and the Senate has its say on a 20-week abortion ban.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Health care gets little love in the State of the Union, the head of the CDC resigns, cancer rates after colonoscopy may heading in the wrong direction, and the Senate has its say on a 20-week abortion ban.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Health care gets little love in the State of the Union, the head of the CDC resigns, cancer rates after colonoscopy may heading in the wrong direction, and the Senate has its say on a 20-week abortion ban.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
Fight statin phobia with hard facts
SNOWMASS, COLO. – One of the toughest tasks in all of preventive cardiology is to convince statin-phobic patients at increased cardiovascular risk that they should take a statin, Robert A. Vogel, MD, observed.
“The two hardest challenges I have in my practice are getting people to stop smoking and getting people who have fear of statins to take statins. That’s half my practice. ,” declared Dr. Vogel, a cardiologist at the University of Colorado, Denver.
“There is one true harm of a statin that I always worry about, and that’s hemorrhagic stroke. It’s rare, but it does occur,” the cardiologist said at the Annual Cardiovascular Conference at Snowmass.
Dr. Vogel highlighted key studies that he believes have convincingly addressed impaired cognition and other proposed statin side effects. He also provided an update on the safety profile of the proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors.
Neurocognitive problems: The Food and Drug Administration really ratcheted up patient fretting when it mandated in 2012 that the labeling for all statins must include a warning of postmarketing reports of adverse events involving ill-defined memory impairment and confusion that were reversed upon drug discontinuation.
“I can tell you, I’ve had dozens of patients come in and say, ‘What about this warning? I’m afraid of dementia,’ ” Dr. Vogel said.
It’s tough to refute anecdotal case reports, but Dr. Vogel pointed to several published meta-analyses of prospective cohort studies, randomized controlled trials, and cross-sectional studies to illustrate his point. For example, investigators at Johns Hopkins University in Baltimore analyzed the results of 16 high-quality randomized trials and prospective cohort studies and found that the short-term studies showed no effect of statin therapy on measurable cognitive endpoints. Moreover, the pooled results of eight long-term studies, including more than 23,000 patients, showed a significant 29% reduction in new-onset dementia in statin-treated patients (Mayo Clin Proc. 2013 Nov;88[11]1213-21).
Another meta-analysis, this one including 27 studies, concluded there is “moderate-quality evidence” to suggest statin users have no increased incidence of dementia, mild cognitive impairment, or any change in neurocognitive scores related to executive function, declarative memory, processing speed, or global cognitive performance.
In this same report, the investigators delved into the FDA’s adverse event reporting database and determined that the rate of reported cognitive-related adverse events was 1.9 cases per 1 million statin prescriptions, identical to the rate for clopidogrel and essentially the same as the 1.6 cases per 1 million rate for losartan (Ann Intern Med. 2013 Nov 19;159[10]:688-97).
“It shows that if you take any drug and put it in the type of population we give these drugs to, you’re going to see about the same frequency of these anecdotal reports, with no signal that statins are any worse than any other drugs we use in cardiology. Is this proof that statins don’t cause cognitive impairment? No, but it’s suggestive that if you give drugs, people have adverse events that may or may not be related to those drugs. So this was reassuring to me that we’ll see this stuff anecdotally, but it probably isn’t due to the statin itself,” Dr. Vogel continued.
Myalgia: In the STOMP study (Effect of Statins on Skeletal Muscle Function and Performance), investigators at Hartford (Conn.) Hospital randomized 420 healthy, statin-naive subjects in a double-blind fashion to 80 mg/day of atorvastatin or placebo for 6 months. The incidence of myalgia was 9.4% in the atorvastatin group compared with 4.6% in placebo-treated controls. Of note, muscle strength on formal testing wasn’t reduced to a greater extent in myalgic patients on atorvastatin than in myalgic patients on placebo (Circulation. 2013 Jan 1;127[1]:96-103).
“There is a signal there,” Dr. Vogel commented. “So the true [placebo-subtracted] incidence of myalgias on a high-dose statin is about 5%. It’s not 20%, it’s not 30%, it’s about 1 patient in 20. It’s real, but it’s not everybody. Those are the numbers you have to think about. If half your patients on statin therapy are getting myalgias, you need to go into a different practice because you’ve got a bunch of Web-searching patients.”
Diabetes: A meta-analysis of 13 randomized controlled trials of statins with more than 91,000 participants and a mean of 4 years of follow-up concluded that for every 255 patients treated with a statin for 4 years, there would be one extra case of new-onset type 2 diabetes, a harm dwarfed by the reduction in cardiovascular events (Lancet. 2010 Feb 27;375[9716]:735-42).
The mechanism for this statin-related, slightly increased risk of developing type 2 diabetes has been clarified by a genetic analysis involving more than 223,000 participants in 43 genetic studies. A large multicenter team of investigators showed that genetic polymorphisms resulting in a less active 3-hydroxy-3-methylglutaryl-coenzyme A reductase gene are associated with lower LDL cholesterol, slightly higher body weight and waist circumference, and increased plasma insulin and plasma glucose. The investigators showed that the more of these alleles an individual possessed, the greater the risk of type 2 diabetes (Lancet. 2015 Jan 24;385[9965]:351-61).
Hemorrhagic stroke: In the SPARCL study (Stroke Prevention by Aggressive Reduction in Cholesterol Levels), 4,731 patients with a recent stroke or transient ischemic attack – 67% of which were ischemic strokes, 2% hemorrhagic strokes – were randomized to high-dose atorvastatin or placebo. Atorvastatin for secondary prevention markedly reduced the overall stroke risk. But this was due to a dramatic decrease in ischemic strokes. The incidence of hemorrhagic stroke was 2.3% in patients on atorvastatin at 80 mg/day for secondary prevention, compared with 1.4% in placebo-treated controls.
In multivariate analysis, the SPARCL investigators found that hemorrhagic stroke risk was increased by an adjusted 68% in patients on atorvastatin, 465% in patients whose prior stroke was hemorrhagic, and 519% in patients with a blood pressure reading of 160-179/100-109 mm Hg at their last clinic visit prior to the hemorrhagic stroke (Neurology. 2008 Jun 10;70[24 Pt 2]:2364-70).
Hepatic dysfunction: This event is extremely rare, so much so that it’s not listed as a side effect in statin labeling. Monitoring of liver function tests is no longer recommended in patients on statin therapy. If elevated tests are seen, find out about the patient’s alcohol consumption – the explanation is far more likely to lie there, according to Dr. Vogel.
What about the safety of the PCSK9 inhibitors?
Dr. Vogel is the U.S. national coordinator for the ongoing phase 3 ODYSSEY Outcomes Study, which is due to report initial results at the 2018 annual meeting of the American College of Cardiology. So far, albeit with only a couple of years worth of data in the large randomized outcome trials, the PCSK9 inhibitors haven’t been associated with muscle problems, cognitive impairment, hepatic dysfunction, or hemorrhagic stroke.
“I think we will eventually see an increased risk of hemorrhagic stroke. I don’t know why we wouldn’t because the mechanism is an antiplatelet mechanism, as with statins. But I don’t know what the incidence will be,” he said.
The EBBINGHAUS substudy of the ongoing FOURIER trial (Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk) of the PCSK9 inhibitor evolocumab(Repatha) provides welcome data on cognition. EBBINGHAUS included 1,974 patients with atherosclerotic cardiovascular disease and normal cognition at baseline who underwent serial testing using the Cambridge Neuropsychological Test Automated Battery over the course of 2 years of prospective follow-up. No signal was seen of any impairment in spatial working memory, learning ability, or the elements of executive function, including planning, organization, and attention. Nor did structured assessment of patient self-reported everyday cognition differ between the active treatment and control arms. Ditto for investigators’ assessment of cognitive adverse events. Moreover, when it did occur, measurable cognitive decline proved unrelated to achieved LDL cholesterol level.
“Two-year data is not 20-year data. And cognitive decline is of great concern. Those of us in this field are going to remain vigilant and look for this, but at least for the present, when you put a patient on a PCSK9 inhibitor and see an LDL drop to 15 or 10 mg/dL – and you will see that happen – the data so far say we do not see cognitive decline,” Dr. Vogel said.
He reported serving as a paid consultant to the National Football League and receiving a research grant from and serving on the speakers bureau for Sanofi.
SNOWMASS, COLO. – One of the toughest tasks in all of preventive cardiology is to convince statin-phobic patients at increased cardiovascular risk that they should take a statin, Robert A. Vogel, MD, observed.
“The two hardest challenges I have in my practice are getting people to stop smoking and getting people who have fear of statins to take statins. That’s half my practice. ,” declared Dr. Vogel, a cardiologist at the University of Colorado, Denver.
“There is one true harm of a statin that I always worry about, and that’s hemorrhagic stroke. It’s rare, but it does occur,” the cardiologist said at the Annual Cardiovascular Conference at Snowmass.
Dr. Vogel highlighted key studies that he believes have convincingly addressed impaired cognition and other proposed statin side effects. He also provided an update on the safety profile of the proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors.
Neurocognitive problems: The Food and Drug Administration really ratcheted up patient fretting when it mandated in 2012 that the labeling for all statins must include a warning of postmarketing reports of adverse events involving ill-defined memory impairment and confusion that were reversed upon drug discontinuation.
“I can tell you, I’ve had dozens of patients come in and say, ‘What about this warning? I’m afraid of dementia,’ ” Dr. Vogel said.
It’s tough to refute anecdotal case reports, but Dr. Vogel pointed to several published meta-analyses of prospective cohort studies, randomized controlled trials, and cross-sectional studies to illustrate his point. For example, investigators at Johns Hopkins University in Baltimore analyzed the results of 16 high-quality randomized trials and prospective cohort studies and found that the short-term studies showed no effect of statin therapy on measurable cognitive endpoints. Moreover, the pooled results of eight long-term studies, including more than 23,000 patients, showed a significant 29% reduction in new-onset dementia in statin-treated patients (Mayo Clin Proc. 2013 Nov;88[11]1213-21).
Another meta-analysis, this one including 27 studies, concluded there is “moderate-quality evidence” to suggest statin users have no increased incidence of dementia, mild cognitive impairment, or any change in neurocognitive scores related to executive function, declarative memory, processing speed, or global cognitive performance.
In this same report, the investigators delved into the FDA’s adverse event reporting database and determined that the rate of reported cognitive-related adverse events was 1.9 cases per 1 million statin prescriptions, identical to the rate for clopidogrel and essentially the same as the 1.6 cases per 1 million rate for losartan (Ann Intern Med. 2013 Nov 19;159[10]:688-97).
“It shows that if you take any drug and put it in the type of population we give these drugs to, you’re going to see about the same frequency of these anecdotal reports, with no signal that statins are any worse than any other drugs we use in cardiology. Is this proof that statins don’t cause cognitive impairment? No, but it’s suggestive that if you give drugs, people have adverse events that may or may not be related to those drugs. So this was reassuring to me that we’ll see this stuff anecdotally, but it probably isn’t due to the statin itself,” Dr. Vogel continued.
Myalgia: In the STOMP study (Effect of Statins on Skeletal Muscle Function and Performance), investigators at Hartford (Conn.) Hospital randomized 420 healthy, statin-naive subjects in a double-blind fashion to 80 mg/day of atorvastatin or placebo for 6 months. The incidence of myalgia was 9.4% in the atorvastatin group compared with 4.6% in placebo-treated controls. Of note, muscle strength on formal testing wasn’t reduced to a greater extent in myalgic patients on atorvastatin than in myalgic patients on placebo (Circulation. 2013 Jan 1;127[1]:96-103).
“There is a signal there,” Dr. Vogel commented. “So the true [placebo-subtracted] incidence of myalgias on a high-dose statin is about 5%. It’s not 20%, it’s not 30%, it’s about 1 patient in 20. It’s real, but it’s not everybody. Those are the numbers you have to think about. If half your patients on statin therapy are getting myalgias, you need to go into a different practice because you’ve got a bunch of Web-searching patients.”
Diabetes: A meta-analysis of 13 randomized controlled trials of statins with more than 91,000 participants and a mean of 4 years of follow-up concluded that for every 255 patients treated with a statin for 4 years, there would be one extra case of new-onset type 2 diabetes, a harm dwarfed by the reduction in cardiovascular events (Lancet. 2010 Feb 27;375[9716]:735-42).
The mechanism for this statin-related, slightly increased risk of developing type 2 diabetes has been clarified by a genetic analysis involving more than 223,000 participants in 43 genetic studies. A large multicenter team of investigators showed that genetic polymorphisms resulting in a less active 3-hydroxy-3-methylglutaryl-coenzyme A reductase gene are associated with lower LDL cholesterol, slightly higher body weight and waist circumference, and increased plasma insulin and plasma glucose. The investigators showed that the more of these alleles an individual possessed, the greater the risk of type 2 diabetes (Lancet. 2015 Jan 24;385[9965]:351-61).
Hemorrhagic stroke: In the SPARCL study (Stroke Prevention by Aggressive Reduction in Cholesterol Levels), 4,731 patients with a recent stroke or transient ischemic attack – 67% of which were ischemic strokes, 2% hemorrhagic strokes – were randomized to high-dose atorvastatin or placebo. Atorvastatin for secondary prevention markedly reduced the overall stroke risk. But this was due to a dramatic decrease in ischemic strokes. The incidence of hemorrhagic stroke was 2.3% in patients on atorvastatin at 80 mg/day for secondary prevention, compared with 1.4% in placebo-treated controls.
In multivariate analysis, the SPARCL investigators found that hemorrhagic stroke risk was increased by an adjusted 68% in patients on atorvastatin, 465% in patients whose prior stroke was hemorrhagic, and 519% in patients with a blood pressure reading of 160-179/100-109 mm Hg at their last clinic visit prior to the hemorrhagic stroke (Neurology. 2008 Jun 10;70[24 Pt 2]:2364-70).
Hepatic dysfunction: This event is extremely rare, so much so that it’s not listed as a side effect in statin labeling. Monitoring of liver function tests is no longer recommended in patients on statin therapy. If elevated tests are seen, find out about the patient’s alcohol consumption – the explanation is far more likely to lie there, according to Dr. Vogel.
What about the safety of the PCSK9 inhibitors?
Dr. Vogel is the U.S. national coordinator for the ongoing phase 3 ODYSSEY Outcomes Study, which is due to report initial results at the 2018 annual meeting of the American College of Cardiology. So far, albeit with only a couple of years worth of data in the large randomized outcome trials, the PCSK9 inhibitors haven’t been associated with muscle problems, cognitive impairment, hepatic dysfunction, or hemorrhagic stroke.
“I think we will eventually see an increased risk of hemorrhagic stroke. I don’t know why we wouldn’t because the mechanism is an antiplatelet mechanism, as with statins. But I don’t know what the incidence will be,” he said.
The EBBINGHAUS substudy of the ongoing FOURIER trial (Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk) of the PCSK9 inhibitor evolocumab(Repatha) provides welcome data on cognition. EBBINGHAUS included 1,974 patients with atherosclerotic cardiovascular disease and normal cognition at baseline who underwent serial testing using the Cambridge Neuropsychological Test Automated Battery over the course of 2 years of prospective follow-up. No signal was seen of any impairment in spatial working memory, learning ability, or the elements of executive function, including planning, organization, and attention. Nor did structured assessment of patient self-reported everyday cognition differ between the active treatment and control arms. Ditto for investigators’ assessment of cognitive adverse events. Moreover, when it did occur, measurable cognitive decline proved unrelated to achieved LDL cholesterol level.
“Two-year data is not 20-year data. And cognitive decline is of great concern. Those of us in this field are going to remain vigilant and look for this, but at least for the present, when you put a patient on a PCSK9 inhibitor and see an LDL drop to 15 or 10 mg/dL – and you will see that happen – the data so far say we do not see cognitive decline,” Dr. Vogel said.
He reported serving as a paid consultant to the National Football League and receiving a research grant from and serving on the speakers bureau for Sanofi.
SNOWMASS, COLO. – One of the toughest tasks in all of preventive cardiology is to convince statin-phobic patients at increased cardiovascular risk that they should take a statin, Robert A. Vogel, MD, observed.
“The two hardest challenges I have in my practice are getting people to stop smoking and getting people who have fear of statins to take statins. That’s half my practice. ,” declared Dr. Vogel, a cardiologist at the University of Colorado, Denver.
“There is one true harm of a statin that I always worry about, and that’s hemorrhagic stroke. It’s rare, but it does occur,” the cardiologist said at the Annual Cardiovascular Conference at Snowmass.
Dr. Vogel highlighted key studies that he believes have convincingly addressed impaired cognition and other proposed statin side effects. He also provided an update on the safety profile of the proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors.
Neurocognitive problems: The Food and Drug Administration really ratcheted up patient fretting when it mandated in 2012 that the labeling for all statins must include a warning of postmarketing reports of adverse events involving ill-defined memory impairment and confusion that were reversed upon drug discontinuation.
“I can tell you, I’ve had dozens of patients come in and say, ‘What about this warning? I’m afraid of dementia,’ ” Dr. Vogel said.
It’s tough to refute anecdotal case reports, but Dr. Vogel pointed to several published meta-analyses of prospective cohort studies, randomized controlled trials, and cross-sectional studies to illustrate his point. For example, investigators at Johns Hopkins University in Baltimore analyzed the results of 16 high-quality randomized trials and prospective cohort studies and found that the short-term studies showed no effect of statin therapy on measurable cognitive endpoints. Moreover, the pooled results of eight long-term studies, including more than 23,000 patients, showed a significant 29% reduction in new-onset dementia in statin-treated patients (Mayo Clin Proc. 2013 Nov;88[11]1213-21).
Another meta-analysis, this one including 27 studies, concluded there is “moderate-quality evidence” to suggest statin users have no increased incidence of dementia, mild cognitive impairment, or any change in neurocognitive scores related to executive function, declarative memory, processing speed, or global cognitive performance.
In this same report, the investigators delved into the FDA’s adverse event reporting database and determined that the rate of reported cognitive-related adverse events was 1.9 cases per 1 million statin prescriptions, identical to the rate for clopidogrel and essentially the same as the 1.6 cases per 1 million rate for losartan (Ann Intern Med. 2013 Nov 19;159[10]:688-97).
“It shows that if you take any drug and put it in the type of population we give these drugs to, you’re going to see about the same frequency of these anecdotal reports, with no signal that statins are any worse than any other drugs we use in cardiology. Is this proof that statins don’t cause cognitive impairment? No, but it’s suggestive that if you give drugs, people have adverse events that may or may not be related to those drugs. So this was reassuring to me that we’ll see this stuff anecdotally, but it probably isn’t due to the statin itself,” Dr. Vogel continued.
Myalgia: In the STOMP study (Effect of Statins on Skeletal Muscle Function and Performance), investigators at Hartford (Conn.) Hospital randomized 420 healthy, statin-naive subjects in a double-blind fashion to 80 mg/day of atorvastatin or placebo for 6 months. The incidence of myalgia was 9.4% in the atorvastatin group compared with 4.6% in placebo-treated controls. Of note, muscle strength on formal testing wasn’t reduced to a greater extent in myalgic patients on atorvastatin than in myalgic patients on placebo (Circulation. 2013 Jan 1;127[1]:96-103).
“There is a signal there,” Dr. Vogel commented. “So the true [placebo-subtracted] incidence of myalgias on a high-dose statin is about 5%. It’s not 20%, it’s not 30%, it’s about 1 patient in 20. It’s real, but it’s not everybody. Those are the numbers you have to think about. If half your patients on statin therapy are getting myalgias, you need to go into a different practice because you’ve got a bunch of Web-searching patients.”
Diabetes: A meta-analysis of 13 randomized controlled trials of statins with more than 91,000 participants and a mean of 4 years of follow-up concluded that for every 255 patients treated with a statin for 4 years, there would be one extra case of new-onset type 2 diabetes, a harm dwarfed by the reduction in cardiovascular events (Lancet. 2010 Feb 27;375[9716]:735-42).
The mechanism for this statin-related, slightly increased risk of developing type 2 diabetes has been clarified by a genetic analysis involving more than 223,000 participants in 43 genetic studies. A large multicenter team of investigators showed that genetic polymorphisms resulting in a less active 3-hydroxy-3-methylglutaryl-coenzyme A reductase gene are associated with lower LDL cholesterol, slightly higher body weight and waist circumference, and increased plasma insulin and plasma glucose. The investigators showed that the more of these alleles an individual possessed, the greater the risk of type 2 diabetes (Lancet. 2015 Jan 24;385[9965]:351-61).
Hemorrhagic stroke: In the SPARCL study (Stroke Prevention by Aggressive Reduction in Cholesterol Levels), 4,731 patients with a recent stroke or transient ischemic attack – 67% of which were ischemic strokes, 2% hemorrhagic strokes – were randomized to high-dose atorvastatin or placebo. Atorvastatin for secondary prevention markedly reduced the overall stroke risk. But this was due to a dramatic decrease in ischemic strokes. The incidence of hemorrhagic stroke was 2.3% in patients on atorvastatin at 80 mg/day for secondary prevention, compared with 1.4% in placebo-treated controls.
In multivariate analysis, the SPARCL investigators found that hemorrhagic stroke risk was increased by an adjusted 68% in patients on atorvastatin, 465% in patients whose prior stroke was hemorrhagic, and 519% in patients with a blood pressure reading of 160-179/100-109 mm Hg at their last clinic visit prior to the hemorrhagic stroke (Neurology. 2008 Jun 10;70[24 Pt 2]:2364-70).
Hepatic dysfunction: This event is extremely rare, so much so that it’s not listed as a side effect in statin labeling. Monitoring of liver function tests is no longer recommended in patients on statin therapy. If elevated tests are seen, find out about the patient’s alcohol consumption – the explanation is far more likely to lie there, according to Dr. Vogel.
What about the safety of the PCSK9 inhibitors?
Dr. Vogel is the U.S. national coordinator for the ongoing phase 3 ODYSSEY Outcomes Study, which is due to report initial results at the 2018 annual meeting of the American College of Cardiology. So far, albeit with only a couple of years worth of data in the large randomized outcome trials, the PCSK9 inhibitors haven’t been associated with muscle problems, cognitive impairment, hepatic dysfunction, or hemorrhagic stroke.
“I think we will eventually see an increased risk of hemorrhagic stroke. I don’t know why we wouldn’t because the mechanism is an antiplatelet mechanism, as with statins. But I don’t know what the incidence will be,” he said.
The EBBINGHAUS substudy of the ongoing FOURIER trial (Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk) of the PCSK9 inhibitor evolocumab(Repatha) provides welcome data on cognition. EBBINGHAUS included 1,974 patients with atherosclerotic cardiovascular disease and normal cognition at baseline who underwent serial testing using the Cambridge Neuropsychological Test Automated Battery over the course of 2 years of prospective follow-up. No signal was seen of any impairment in spatial working memory, learning ability, or the elements of executive function, including planning, organization, and attention. Nor did structured assessment of patient self-reported everyday cognition differ between the active treatment and control arms. Ditto for investigators’ assessment of cognitive adverse events. Moreover, when it did occur, measurable cognitive decline proved unrelated to achieved LDL cholesterol level.
“Two-year data is not 20-year data. And cognitive decline is of great concern. Those of us in this field are going to remain vigilant and look for this, but at least for the present, when you put a patient on a PCSK9 inhibitor and see an LDL drop to 15 or 10 mg/dL – and you will see that happen – the data so far say we do not see cognitive decline,” Dr. Vogel said.
He reported serving as a paid consultant to the National Football League and receiving a research grant from and serving on the speakers bureau for Sanofi.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
A Year 3 Progress Report on Graduate Medical Education Expansion in the Veterans Choice Act
The VHA is the largest healthcare delivery system in the U.S. It includes 146 medical centers (VAMCs), 1,063 community-based outpatient centers (CBOCs) and various other sites of care. General Omar Bradley, the first VA Secretary, established education as one of VA’s 4 statutory missions in Policy Memorandum No.2.1 In addition to training physicians to care for active-duty service members and veterans, 38 USC §7302 directs the VA to assist in providing an adequate supply of health personnel. The 4 statutory missions of the VA are inclusive of not only developing, operating, and maintaining a health care system for veterans, but also including contingency support services as part of emergency preparedness, conducting research, and offering a program of education for health professions.
Background
Today, with few exceptions, the VHA does not act as a graduate medical education (GME) sponsoring institution. Through its Office of Academic Affiliations (OAA), the VHA develops partnerships with Liaison Committee for Medical Education (LCME)/American Osteopathic Association (AOA)-approved medical colleges/universities and with institutions that sponsor Accreditation Council for Graduate Medical Education (ACGME)/AOA-accredited residency program-sponsoring institutions. These collaborations include 144 out of 149 allopathic medical schools and all 34 osteopathic medical schools. The VHA provided training to 43,565 medical residents and 24,683 medical students through these partnerships in 2017.2 Since funding of the GME positions is not provided through the Centers for Medicare & Medicaid Services (CMS), program sponsors may use these partnerships to expand GME positions beyond their funding (but not ACGME) cap.
The gap between supply and demand of physicians continues to grow nationally.3,4 This gap is particularly significant in rural and other underserved areas. U.S. Census Bureau data show that about 5 million veterans (24%) live in rural areas.5 Compared with the urban veteran population, the rural veteran experiences higher disease prevalence and lower physical and mental quality-of-life scores.6 Addressing the problem of physician shortages is a mission-critical priority for the VHA.7
With an eye toward enhancing 2 of the 4 statutory missions of the VA and to mitigate the shortage of physicians and improve the access of veterans to VHA medical services, on August 7, 2014, the Veterans Access, Choice, and Accountability Act of 2014 (Public Law [PL] 113-146), known as the Choice Act was enacted.8 Title III, §301(b) of the Choice Act requires VHA to increase GME residency positions by:
Establishing new medical residency programs, or ensuring that already established medical residency programs have a sufficient number of residency positions, at any VHA medical facility that is: (a) experiencing a shortage of physicians and (b) located in a community that is designated as a health professional shortage area.
The legislation specifies that priority must be placed on medical occupations that experience the largest staffing shortages throughout the VHA and “programs in primary care, mental health, and any other specialty that the Secretary of the VA determines appropriate.” The Choice Act authorized the VHA to increase the number of GME residency positions by up to 1,500 over a 5-year period. In December 2016, as amended by PL 114–315, Title VI, §617(a), this authorization was extended by another 5 years for a total of 10 years and will run through 2024.9
GME Development/Distribution
To distribute these newly created GME positions as mandated by Congress, the OAA is using a system with 3 types of request for proposal (RFP) applications. These include planning, infrastructure, and position grants. This phased approach was taken with the understanding that the development of new training sites requires a properly staffed education office and dedicated faculty time. Planning and infrastructure grants provide start-up funds for smaller VAMCs, allowing them to keep facility resources focused on their clinical mission.
Planning grants (of up to $250,000 over 2 years) primarily were designed for VA facilities with no or low numbers of physician residents at the desired teaching location. Priority was given to facilities in rural and/or underserved areas as well as those developing new affiliations. Applications were reviewed by OAA staff along with peer-selected Designated Education Officers (DEOs) from VA facilities across the nation that were not applying for the grants. Awards were based on the priorities mentioned earlier, with additional credit for programs focused on 2 VHA fundamental services areas—primary care and/or mental health training. Facilities receiving planning grants were mentored by an OAA physician staff member, anticipating a 2- to 3-year time line to request positions and begin GME training.
Infrastructure grants (of up to $520,000 used over 2-3 years) were designed as bridge funds after approval of Veterans Access, Choice, and Accountability Act (VACAA) GME positions. Infrastructure grants are appropriate to sustain a local education office, develop VA faculty, purchase equipment, and make minor modifications to the clinical space in the VAMCs or CBOCs to enhance the learning environment during the period before VA supportive funds from the Veterans Equitable Resource Allocation (VERA) (similar to indirect GME funds from CMS) become available. Applications were managed the same as planning grant submissions.
Position RFPs, unlike planning and infrastructure RFPs, are available to all VAMCs. The primary purpose of the VACAA Position RFP is to fund new positions in primary care and psychiatry. Graduate medical education positions in subspecialty programs also are considered when there is documentation of critical need to improve access to these services. Applications were reviewed by OAA staff along with selected DEOs from VA facilities around the U.S. Award criteria prioritized primary care (family medicine, internal medicine, geriatrics), and mental health (psychiatry and psychiatry subspecialties). Priority also was given to positions in areas with a documented shortage of physicians and areas with high concentrations of veterans.
Current Progress
To date the OAA has offered 3 RFP cycles consisting of planning/infrastructure grants, and 4 RFP cycles for salary/benefit support for additional resident full-time equivalent (FTE) positions. Resident positions were defined as residency or fellowship FTEs that were part of an ACGME or AOA-accredited training program. Figure 1 illustrates the geographic distribution of awarded GME positions.
In primary care specialties (family medicine, internal medicine, and geriatrics, a total of 349.4 FTE positions have been approved (Table 1). Due to a low number of applications, only 6.3 of these positions were awarded in geriatrics. In mental health, 167.6 FTE positions have been approved, whereas in critical needs specialties (needed to support rural/underserved healthcare and improve specialty access) 256.5 FTE positions have been added.
Discussion
There are several important desired short-term outcomes from VACAA. The first is improved access to high-quality care for both rural and urban veterans. There is an emphasis on primary care and mental health because shortages in these areas across the U.S. are well established.3,4,10 Likewise, rural areas have been prioritized because often there is a disparity of care.
One area of concern is the small number of applicants in geriatrics. Even with VACAA specifically targeting geriatrics as a primary care specialty, we have only received enough applications to approve 6.3 positions over the first 3 years of the program. As the veteran and overall population in the U.S. ages, it is important to develop a medical workforce that is willing and able to address their needs.
The VACAA statute is not intended to alter medical students’ career choice but rather to provide funded positions for those choosing primary care, geriatrics, psychiatry (including psychiatric subspecialties), and experience in the VA clinical settings. The hope is that this experience will encourage practitioners to competently care for veterans after training in the VA and/or other civilian settings.
By enabling smaller VA facilities to become training sites through planning and infrastructure grants, residents have the opportunity to gain experience in more rural settings. Physicians who choose to train in rural areas are likely to spend time practicing in those areas after they complete training.15 The process of developing facilities with no GME into training sitestakes time and resources. Establishing an education office and choosing site directors and core faculty are all important steps that must be done before resident rotations begin. Resources provided through VACAA have enabled the VHA to reduce the number of VAMCs with no GME activity to just 3.
Another benefit of VACAA GME expansion is the opportunity to engage new LCME/AOA-accredited medical schools and ACGME/AOA-accredited residency-sponsoring institutions.16,17 Representatives of these institutions may have perceived a reluctance of their local VAs to develop GME affiliations in the past. This statute has enabled many VAMCs to use nontraditional training sites and modalities to overcome barriers and create new academic affiliations.
However, VACAA only provides funds for training that occurs in established VA sites of care. This can hinder the development of partnerships where other funding sources are required for non-VA rotations. Another VACAA limitation is that it does not fund undergraduate medical education as does the Armed Forces Health Professional Scholarship Program (HPSP). In addition, the primary financial relationship is between the VA and the sponsoring institution, thus VHA cannot send residents to underserved locations.
Conclusion
The VHA has a rich tradition of educating physician and other health care providers in the U.S. More than 60% of U.S. trained physicians received a portion of their training through VHA.2 Through VACAA GME expansion initiative, the 113th Congress has asked VHA to continue its important training mission “to bind up the Nations wounds” and “to care for him who shall have borne the battle.”18
Acknowledgments
In memoriam – Robert Louis Jesse MD, PhD. Dr. Jesse, the Chief of the Office of Academic Affiliations passed away on September 2, 2017, at age 64. He had an illustrious medical career as a cardiologist and served in many leadership roles including Principal Deputy Under Secretary for Health in the U.S. Department of Veterans Affairs. His expertise, visionary leadership, and friendship will be missed by all involved in the VA’s academic training mission but particularly by those of us who worked for and with him at OAA.
1. U.S. Department of Veteran Affairs. Policy Memorandum No. 2. Policy in association of veterans’ hospitals with medical schools. https://www.va.gov/oaa/Archive/PolicyMemo2.pdf. Published January 30, 1947. Accessed December 13, 2017.
2. U.S. Department of Veteran Affairs, Office of Academic Affiliations. 2017 statistics: health professions trainees. https://www .va.gov/OAA/docs/OAA_Statistics.pdf. Accessed January 8, 2018.
3. IHS, Inc. The complexities of physician supply and demand 2016 update: projections from 2014 to 2025, final report. https://www.aamc.org/download/458082/data/2016_complexities_of_supply_and_demand_projections.pdf. Published April 5, 2016. Accessed December 13, 2017.
4. Petterson SM, Liaw WR, Tran C, Bazemore AW. Estimating the residency expansion required to avoid projected primary care physician shortages by 2035. Ann Fam Med. 2015;13(2):107-114.
5. Holder KA. Veterans in rural America 2011-2015. https://www.census.gov/content/dam/Census/library/publica tions/2017/acs/acs-36.pdf. Published January 2017. Accessed January 18, 2018.
6. Weeks WB, Wallace AE, Wang S, Lee A, Kazis LE. Rural-urban disparities in health-related quality of life within disease categories of veterans. J Rural Health. 2006;22(3):204-211.
7. U.S. Government Accountability Office. GAO-18-124. VHA Physician Staffing and Recruitment. https://www.gao.gov/assets/690/687853.pdf. Published October 19, 2017. Accessed January 23, 2018.
8. Veterans Access, Choice, and Accountability Act, section 301 (b): Increase of graduate medical education residency positions, 38 USC § 74 (2014) .
9. Jeff Miller and Richard Blumenthal Veterans Health Care and Benefits Improvement Act of 2016, 38 USC §101 (2016).
10. Thomas KC, Ellis AR, Konrad TR, Holzer CE, Morrissey JP. County-level estimates of mental health professional shortage in the United States. Psychiatr Serv. 2009;60(10):1323-1328.
11. Garibaldi RA, Popkave C, Bylsma W. Career plans for trainees in internal medicine residency programs. Acad Med. 2005;80(5):507-512.
12. West CP, Dupras DM. General medicine vs subspecialty career plans among internal medicine residents. JAMA. 2012;308(21):2241-2247.
13. Stimmel B, Haddow S, Smith L. The practice of general internal medicine by subspecialists. J Urban Health. 1998;75(1):184-190.
14. Shea JA, Kleetke PR, Wozniak GD, Polsky D, Escarce JJ. Self-reported physician specialties and the primary care content of medical practice: a study of the AMA physician masterfile. American Medical Association. Med Care. 1999;37(4):333-338.
15. Rabinowitz HK, Diamond JJ, Markham FW, Paynter NP. Critical factors for designing
16. Accredited MD programs in the United States. http://lcme.org /directory/accredited-u-s-programs/. Updated December 12, 2017. Accessed January 8, 2018.
17. Osteopathic medical schools. http://www.osteopathic.org/in side-aoa/about/affiliates/Pages/osteopathic-medical-schools.aspx Published 2017. Accessed January 8, 2018.
18. Lincoln A. Second inaugural address. https://www.va.gov/opa/publications/celebrate/vamotto.pdf. Accessed January 8. 2018.
The VHA is the largest healthcare delivery system in the U.S. It includes 146 medical centers (VAMCs), 1,063 community-based outpatient centers (CBOCs) and various other sites of care. General Omar Bradley, the first VA Secretary, established education as one of VA’s 4 statutory missions in Policy Memorandum No.2.1 In addition to training physicians to care for active-duty service members and veterans, 38 USC §7302 directs the VA to assist in providing an adequate supply of health personnel. The 4 statutory missions of the VA are inclusive of not only developing, operating, and maintaining a health care system for veterans, but also including contingency support services as part of emergency preparedness, conducting research, and offering a program of education for health professions.
Background
Today, with few exceptions, the VHA does not act as a graduate medical education (GME) sponsoring institution. Through its Office of Academic Affiliations (OAA), the VHA develops partnerships with Liaison Committee for Medical Education (LCME)/American Osteopathic Association (AOA)-approved medical colleges/universities and with institutions that sponsor Accreditation Council for Graduate Medical Education (ACGME)/AOA-accredited residency program-sponsoring institutions. These collaborations include 144 out of 149 allopathic medical schools and all 34 osteopathic medical schools. The VHA provided training to 43,565 medical residents and 24,683 medical students through these partnerships in 2017.2 Since funding of the GME positions is not provided through the Centers for Medicare & Medicaid Services (CMS), program sponsors may use these partnerships to expand GME positions beyond their funding (but not ACGME) cap.
The gap between supply and demand of physicians continues to grow nationally.3,4 This gap is particularly significant in rural and other underserved areas. U.S. Census Bureau data show that about 5 million veterans (24%) live in rural areas.5 Compared with the urban veteran population, the rural veteran experiences higher disease prevalence and lower physical and mental quality-of-life scores.6 Addressing the problem of physician shortages is a mission-critical priority for the VHA.7
With an eye toward enhancing 2 of the 4 statutory missions of the VA and to mitigate the shortage of physicians and improve the access of veterans to VHA medical services, on August 7, 2014, the Veterans Access, Choice, and Accountability Act of 2014 (Public Law [PL] 113-146), known as the Choice Act was enacted.8 Title III, §301(b) of the Choice Act requires VHA to increase GME residency positions by:
Establishing new medical residency programs, or ensuring that already established medical residency programs have a sufficient number of residency positions, at any VHA medical facility that is: (a) experiencing a shortage of physicians and (b) located in a community that is designated as a health professional shortage area.
The legislation specifies that priority must be placed on medical occupations that experience the largest staffing shortages throughout the VHA and “programs in primary care, mental health, and any other specialty that the Secretary of the VA determines appropriate.” The Choice Act authorized the VHA to increase the number of GME residency positions by up to 1,500 over a 5-year period. In December 2016, as amended by PL 114–315, Title VI, §617(a), this authorization was extended by another 5 years for a total of 10 years and will run through 2024.9
GME Development/Distribution
To distribute these newly created GME positions as mandated by Congress, the OAA is using a system with 3 types of request for proposal (RFP) applications. These include planning, infrastructure, and position grants. This phased approach was taken with the understanding that the development of new training sites requires a properly staffed education office and dedicated faculty time. Planning and infrastructure grants provide start-up funds for smaller VAMCs, allowing them to keep facility resources focused on their clinical mission.
Planning grants (of up to $250,000 over 2 years) primarily were designed for VA facilities with no or low numbers of physician residents at the desired teaching location. Priority was given to facilities in rural and/or underserved areas as well as those developing new affiliations. Applications were reviewed by OAA staff along with peer-selected Designated Education Officers (DEOs) from VA facilities across the nation that were not applying for the grants. Awards were based on the priorities mentioned earlier, with additional credit for programs focused on 2 VHA fundamental services areas—primary care and/or mental health training. Facilities receiving planning grants were mentored by an OAA physician staff member, anticipating a 2- to 3-year time line to request positions and begin GME training.
Infrastructure grants (of up to $520,000 used over 2-3 years) were designed as bridge funds after approval of Veterans Access, Choice, and Accountability Act (VACAA) GME positions. Infrastructure grants are appropriate to sustain a local education office, develop VA faculty, purchase equipment, and make minor modifications to the clinical space in the VAMCs or CBOCs to enhance the learning environment during the period before VA supportive funds from the Veterans Equitable Resource Allocation (VERA) (similar to indirect GME funds from CMS) become available. Applications were managed the same as planning grant submissions.
Position RFPs, unlike planning and infrastructure RFPs, are available to all VAMCs. The primary purpose of the VACAA Position RFP is to fund new positions in primary care and psychiatry. Graduate medical education positions in subspecialty programs also are considered when there is documentation of critical need to improve access to these services. Applications were reviewed by OAA staff along with selected DEOs from VA facilities around the U.S. Award criteria prioritized primary care (family medicine, internal medicine, geriatrics), and mental health (psychiatry and psychiatry subspecialties). Priority also was given to positions in areas with a documented shortage of physicians and areas with high concentrations of veterans.
Current Progress
To date the OAA has offered 3 RFP cycles consisting of planning/infrastructure grants, and 4 RFP cycles for salary/benefit support for additional resident full-time equivalent (FTE) positions. Resident positions were defined as residency or fellowship FTEs that were part of an ACGME or AOA-accredited training program. Figure 1 illustrates the geographic distribution of awarded GME positions.
In primary care specialties (family medicine, internal medicine, and geriatrics, a total of 349.4 FTE positions have been approved (Table 1). Due to a low number of applications, only 6.3 of these positions were awarded in geriatrics. In mental health, 167.6 FTE positions have been approved, whereas in critical needs specialties (needed to support rural/underserved healthcare and improve specialty access) 256.5 FTE positions have been added.
Discussion
There are several important desired short-term outcomes from VACAA. The first is improved access to high-quality care for both rural and urban veterans. There is an emphasis on primary care and mental health because shortages in these areas across the U.S. are well established.3,4,10 Likewise, rural areas have been prioritized because often there is a disparity of care.
One area of concern is the small number of applicants in geriatrics. Even with VACAA specifically targeting geriatrics as a primary care specialty, we have only received enough applications to approve 6.3 positions over the first 3 years of the program. As the veteran and overall population in the U.S. ages, it is important to develop a medical workforce that is willing and able to address their needs.
The VACAA statute is not intended to alter medical students’ career choice but rather to provide funded positions for those choosing primary care, geriatrics, psychiatry (including psychiatric subspecialties), and experience in the VA clinical settings. The hope is that this experience will encourage practitioners to competently care for veterans after training in the VA and/or other civilian settings.
By enabling smaller VA facilities to become training sites through planning and infrastructure grants, residents have the opportunity to gain experience in more rural settings. Physicians who choose to train in rural areas are likely to spend time practicing in those areas after they complete training.15 The process of developing facilities with no GME into training sitestakes time and resources. Establishing an education office and choosing site directors and core faculty are all important steps that must be done before resident rotations begin. Resources provided through VACAA have enabled the VHA to reduce the number of VAMCs with no GME activity to just 3.
Another benefit of VACAA GME expansion is the opportunity to engage new LCME/AOA-accredited medical schools and ACGME/AOA-accredited residency-sponsoring institutions.16,17 Representatives of these institutions may have perceived a reluctance of their local VAs to develop GME affiliations in the past. This statute has enabled many VAMCs to use nontraditional training sites and modalities to overcome barriers and create new academic affiliations.
However, VACAA only provides funds for training that occurs in established VA sites of care. This can hinder the development of partnerships where other funding sources are required for non-VA rotations. Another VACAA limitation is that it does not fund undergraduate medical education as does the Armed Forces Health Professional Scholarship Program (HPSP). In addition, the primary financial relationship is between the VA and the sponsoring institution, thus VHA cannot send residents to underserved locations.
Conclusion
The VHA has a rich tradition of educating physician and other health care providers in the U.S. More than 60% of U.S. trained physicians received a portion of their training through VHA.2 Through VACAA GME expansion initiative, the 113th Congress has asked VHA to continue its important training mission “to bind up the Nations wounds” and “to care for him who shall have borne the battle.”18
Acknowledgments
In memoriam – Robert Louis Jesse MD, PhD. Dr. Jesse, the Chief of the Office of Academic Affiliations passed away on September 2, 2017, at age 64. He had an illustrious medical career as a cardiologist and served in many leadership roles including Principal Deputy Under Secretary for Health in the U.S. Department of Veterans Affairs. His expertise, visionary leadership, and friendship will be missed by all involved in the VA’s academic training mission but particularly by those of us who worked for and with him at OAA.
The VHA is the largest healthcare delivery system in the U.S. It includes 146 medical centers (VAMCs), 1,063 community-based outpatient centers (CBOCs) and various other sites of care. General Omar Bradley, the first VA Secretary, established education as one of VA’s 4 statutory missions in Policy Memorandum No.2.1 In addition to training physicians to care for active-duty service members and veterans, 38 USC §7302 directs the VA to assist in providing an adequate supply of health personnel. The 4 statutory missions of the VA are inclusive of not only developing, operating, and maintaining a health care system for veterans, but also including contingency support services as part of emergency preparedness, conducting research, and offering a program of education for health professions.
Background
Today, with few exceptions, the VHA does not act as a graduate medical education (GME) sponsoring institution. Through its Office of Academic Affiliations (OAA), the VHA develops partnerships with Liaison Committee for Medical Education (LCME)/American Osteopathic Association (AOA)-approved medical colleges/universities and with institutions that sponsor Accreditation Council for Graduate Medical Education (ACGME)/AOA-accredited residency program-sponsoring institutions. These collaborations include 144 out of 149 allopathic medical schools and all 34 osteopathic medical schools. The VHA provided training to 43,565 medical residents and 24,683 medical students through these partnerships in 2017.2 Since funding of the GME positions is not provided through the Centers for Medicare & Medicaid Services (CMS), program sponsors may use these partnerships to expand GME positions beyond their funding (but not ACGME) cap.
The gap between supply and demand of physicians continues to grow nationally.3,4 This gap is particularly significant in rural and other underserved areas. U.S. Census Bureau data show that about 5 million veterans (24%) live in rural areas.5 Compared with the urban veteran population, the rural veteran experiences higher disease prevalence and lower physical and mental quality-of-life scores.6 Addressing the problem of physician shortages is a mission-critical priority for the VHA.7
With an eye toward enhancing 2 of the 4 statutory missions of the VA and to mitigate the shortage of physicians and improve the access of veterans to VHA medical services, on August 7, 2014, the Veterans Access, Choice, and Accountability Act of 2014 (Public Law [PL] 113-146), known as the Choice Act was enacted.8 Title III, §301(b) of the Choice Act requires VHA to increase GME residency positions by:
Establishing new medical residency programs, or ensuring that already established medical residency programs have a sufficient number of residency positions, at any VHA medical facility that is: (a) experiencing a shortage of physicians and (b) located in a community that is designated as a health professional shortage area.
The legislation specifies that priority must be placed on medical occupations that experience the largest staffing shortages throughout the VHA and “programs in primary care, mental health, and any other specialty that the Secretary of the VA determines appropriate.” The Choice Act authorized the VHA to increase the number of GME residency positions by up to 1,500 over a 5-year period. In December 2016, as amended by PL 114–315, Title VI, §617(a), this authorization was extended by another 5 years for a total of 10 years and will run through 2024.9
GME Development/Distribution
To distribute these newly created GME positions as mandated by Congress, the OAA is using a system with 3 types of request for proposal (RFP) applications. These include planning, infrastructure, and position grants. This phased approach was taken with the understanding that the development of new training sites requires a properly staffed education office and dedicated faculty time. Planning and infrastructure grants provide start-up funds for smaller VAMCs, allowing them to keep facility resources focused on their clinical mission.
Planning grants (of up to $250,000 over 2 years) primarily were designed for VA facilities with no or low numbers of physician residents at the desired teaching location. Priority was given to facilities in rural and/or underserved areas as well as those developing new affiliations. Applications were reviewed by OAA staff along with peer-selected Designated Education Officers (DEOs) from VA facilities across the nation that were not applying for the grants. Awards were based on the priorities mentioned earlier, with additional credit for programs focused on 2 VHA fundamental services areas—primary care and/or mental health training. Facilities receiving planning grants were mentored by an OAA physician staff member, anticipating a 2- to 3-year time line to request positions and begin GME training.
Infrastructure grants (of up to $520,000 used over 2-3 years) were designed as bridge funds after approval of Veterans Access, Choice, and Accountability Act (VACAA) GME positions. Infrastructure grants are appropriate to sustain a local education office, develop VA faculty, purchase equipment, and make minor modifications to the clinical space in the VAMCs or CBOCs to enhance the learning environment during the period before VA supportive funds from the Veterans Equitable Resource Allocation (VERA) (similar to indirect GME funds from CMS) become available. Applications were managed the same as planning grant submissions.
Position RFPs, unlike planning and infrastructure RFPs, are available to all VAMCs. The primary purpose of the VACAA Position RFP is to fund new positions in primary care and psychiatry. Graduate medical education positions in subspecialty programs also are considered when there is documentation of critical need to improve access to these services. Applications were reviewed by OAA staff along with selected DEOs from VA facilities around the U.S. Award criteria prioritized primary care (family medicine, internal medicine, geriatrics), and mental health (psychiatry and psychiatry subspecialties). Priority also was given to positions in areas with a documented shortage of physicians and areas with high concentrations of veterans.
Current Progress
To date the OAA has offered 3 RFP cycles consisting of planning/infrastructure grants, and 4 RFP cycles for salary/benefit support for additional resident full-time equivalent (FTE) positions. Resident positions were defined as residency or fellowship FTEs that were part of an ACGME or AOA-accredited training program. Figure 1 illustrates the geographic distribution of awarded GME positions.
In primary care specialties (family medicine, internal medicine, and geriatrics, a total of 349.4 FTE positions have been approved (Table 1). Due to a low number of applications, only 6.3 of these positions were awarded in geriatrics. In mental health, 167.6 FTE positions have been approved, whereas in critical needs specialties (needed to support rural/underserved healthcare and improve specialty access) 256.5 FTE positions have been added.
Discussion
There are several important desired short-term outcomes from VACAA. The first is improved access to high-quality care for both rural and urban veterans. There is an emphasis on primary care and mental health because shortages in these areas across the U.S. are well established.3,4,10 Likewise, rural areas have been prioritized because often there is a disparity of care.
One area of concern is the small number of applicants in geriatrics. Even with VACAA specifically targeting geriatrics as a primary care specialty, we have only received enough applications to approve 6.3 positions over the first 3 years of the program. As the veteran and overall population in the U.S. ages, it is important to develop a medical workforce that is willing and able to address their needs.
The VACAA statute is not intended to alter medical students’ career choice but rather to provide funded positions for those choosing primary care, geriatrics, psychiatry (including psychiatric subspecialties), and experience in the VA clinical settings. The hope is that this experience will encourage practitioners to competently care for veterans after training in the VA and/or other civilian settings.
By enabling smaller VA facilities to become training sites through planning and infrastructure grants, residents have the opportunity to gain experience in more rural settings. Physicians who choose to train in rural areas are likely to spend time practicing in those areas after they complete training.15 The process of developing facilities with no GME into training sitestakes time and resources. Establishing an education office and choosing site directors and core faculty are all important steps that must be done before resident rotations begin. Resources provided through VACAA have enabled the VHA to reduce the number of VAMCs with no GME activity to just 3.
Another benefit of VACAA GME expansion is the opportunity to engage new LCME/AOA-accredited medical schools and ACGME/AOA-accredited residency-sponsoring institutions.16,17 Representatives of these institutions may have perceived a reluctance of their local VAs to develop GME affiliations in the past. This statute has enabled many VAMCs to use nontraditional training sites and modalities to overcome barriers and create new academic affiliations.
However, VACAA only provides funds for training that occurs in established VA sites of care. This can hinder the development of partnerships where other funding sources are required for non-VA rotations. Another VACAA limitation is that it does not fund undergraduate medical education as does the Armed Forces Health Professional Scholarship Program (HPSP). In addition, the primary financial relationship is between the VA and the sponsoring institution, thus VHA cannot send residents to underserved locations.
Conclusion
The VHA has a rich tradition of educating physician and other health care providers in the U.S. More than 60% of U.S. trained physicians received a portion of their training through VHA.2 Through VACAA GME expansion initiative, the 113th Congress has asked VHA to continue its important training mission “to bind up the Nations wounds” and “to care for him who shall have borne the battle.”18
Acknowledgments
In memoriam – Robert Louis Jesse MD, PhD. Dr. Jesse, the Chief of the Office of Academic Affiliations passed away on September 2, 2017, at age 64. He had an illustrious medical career as a cardiologist and served in many leadership roles including Principal Deputy Under Secretary for Health in the U.S. Department of Veterans Affairs. His expertise, visionary leadership, and friendship will be missed by all involved in the VA’s academic training mission but particularly by those of us who worked for and with him at OAA.
1. U.S. Department of Veteran Affairs. Policy Memorandum No. 2. Policy in association of veterans’ hospitals with medical schools. https://www.va.gov/oaa/Archive/PolicyMemo2.pdf. Published January 30, 1947. Accessed December 13, 2017.
2. U.S. Department of Veteran Affairs, Office of Academic Affiliations. 2017 statistics: health professions trainees. https://www .va.gov/OAA/docs/OAA_Statistics.pdf. Accessed January 8, 2018.
3. IHS, Inc. The complexities of physician supply and demand 2016 update: projections from 2014 to 2025, final report. https://www.aamc.org/download/458082/data/2016_complexities_of_supply_and_demand_projections.pdf. Published April 5, 2016. Accessed December 13, 2017.
4. Petterson SM, Liaw WR, Tran C, Bazemore AW. Estimating the residency expansion required to avoid projected primary care physician shortages by 2035. Ann Fam Med. 2015;13(2):107-114.
5. Holder KA. Veterans in rural America 2011-2015. https://www.census.gov/content/dam/Census/library/publica tions/2017/acs/acs-36.pdf. Published January 2017. Accessed January 18, 2018.
6. Weeks WB, Wallace AE, Wang S, Lee A, Kazis LE. Rural-urban disparities in health-related quality of life within disease categories of veterans. J Rural Health. 2006;22(3):204-211.
7. U.S. Government Accountability Office. GAO-18-124. VHA Physician Staffing and Recruitment. https://www.gao.gov/assets/690/687853.pdf. Published October 19, 2017. Accessed January 23, 2018.
8. Veterans Access, Choice, and Accountability Act, section 301 (b): Increase of graduate medical education residency positions, 38 USC § 74 (2014) .
9. Jeff Miller and Richard Blumenthal Veterans Health Care and Benefits Improvement Act of 2016, 38 USC §101 (2016).
10. Thomas KC, Ellis AR, Konrad TR, Holzer CE, Morrissey JP. County-level estimates of mental health professional shortage in the United States. Psychiatr Serv. 2009;60(10):1323-1328.
11. Garibaldi RA, Popkave C, Bylsma W. Career plans for trainees in internal medicine residency programs. Acad Med. 2005;80(5):507-512.
12. West CP, Dupras DM. General medicine vs subspecialty career plans among internal medicine residents. JAMA. 2012;308(21):2241-2247.
13. Stimmel B, Haddow S, Smith L. The practice of general internal medicine by subspecialists. J Urban Health. 1998;75(1):184-190.
14. Shea JA, Kleetke PR, Wozniak GD, Polsky D, Escarce JJ. Self-reported physician specialties and the primary care content of medical practice: a study of the AMA physician masterfile. American Medical Association. Med Care. 1999;37(4):333-338.
15. Rabinowitz HK, Diamond JJ, Markham FW, Paynter NP. Critical factors for designing
16. Accredited MD programs in the United States. http://lcme.org /directory/accredited-u-s-programs/. Updated December 12, 2017. Accessed January 8, 2018.
17. Osteopathic medical schools. http://www.osteopathic.org/in side-aoa/about/affiliates/Pages/osteopathic-medical-schools.aspx Published 2017. Accessed January 8, 2018.
18. Lincoln A. Second inaugural address. https://www.va.gov/opa/publications/celebrate/vamotto.pdf. Accessed January 8. 2018.
1. U.S. Department of Veteran Affairs. Policy Memorandum No. 2. Policy in association of veterans’ hospitals with medical schools. https://www.va.gov/oaa/Archive/PolicyMemo2.pdf. Published January 30, 1947. Accessed December 13, 2017.
2. U.S. Department of Veteran Affairs, Office of Academic Affiliations. 2017 statistics: health professions trainees. https://www .va.gov/OAA/docs/OAA_Statistics.pdf. Accessed January 8, 2018.
3. IHS, Inc. The complexities of physician supply and demand 2016 update: projections from 2014 to 2025, final report. https://www.aamc.org/download/458082/data/2016_complexities_of_supply_and_demand_projections.pdf. Published April 5, 2016. Accessed December 13, 2017.
4. Petterson SM, Liaw WR, Tran C, Bazemore AW. Estimating the residency expansion required to avoid projected primary care physician shortages by 2035. Ann Fam Med. 2015;13(2):107-114.
5. Holder KA. Veterans in rural America 2011-2015. https://www.census.gov/content/dam/Census/library/publica tions/2017/acs/acs-36.pdf. Published January 2017. Accessed January 18, 2018.
6. Weeks WB, Wallace AE, Wang S, Lee A, Kazis LE. Rural-urban disparities in health-related quality of life within disease categories of veterans. J Rural Health. 2006;22(3):204-211.
7. U.S. Government Accountability Office. GAO-18-124. VHA Physician Staffing and Recruitment. https://www.gao.gov/assets/690/687853.pdf. Published October 19, 2017. Accessed January 23, 2018.
8. Veterans Access, Choice, and Accountability Act, section 301 (b): Increase of graduate medical education residency positions, 38 USC § 74 (2014) .
9. Jeff Miller and Richard Blumenthal Veterans Health Care and Benefits Improvement Act of 2016, 38 USC §101 (2016).
10. Thomas KC, Ellis AR, Konrad TR, Holzer CE, Morrissey JP. County-level estimates of mental health professional shortage in the United States. Psychiatr Serv. 2009;60(10):1323-1328.
11. Garibaldi RA, Popkave C, Bylsma W. Career plans for trainees in internal medicine residency programs. Acad Med. 2005;80(5):507-512.
12. West CP, Dupras DM. General medicine vs subspecialty career plans among internal medicine residents. JAMA. 2012;308(21):2241-2247.
13. Stimmel B, Haddow S, Smith L. The practice of general internal medicine by subspecialists. J Urban Health. 1998;75(1):184-190.
14. Shea JA, Kleetke PR, Wozniak GD, Polsky D, Escarce JJ. Self-reported physician specialties and the primary care content of medical practice: a study of the AMA physician masterfile. American Medical Association. Med Care. 1999;37(4):333-338.
15. Rabinowitz HK, Diamond JJ, Markham FW, Paynter NP. Critical factors for designing
16. Accredited MD programs in the United States. http://lcme.org /directory/accredited-u-s-programs/. Updated December 12, 2017. Accessed January 8, 2018.
17. Osteopathic medical schools. http://www.osteopathic.org/in side-aoa/about/affiliates/Pages/osteopathic-medical-schools.aspx Published 2017. Accessed January 8, 2018.
18. Lincoln A. Second inaugural address. https://www.va.gov/opa/publications/celebrate/vamotto.pdf. Accessed January 8. 2018.
February 2018 Digital Edition
Click here to access the February 2018 Digital Edition.
Table of Contents
- A Year 3 Progress Report on Graduate Medical Education Expansion in the Veterans Choice Act
- Postpartum Psychosis in a Young VA Patient: Diagnosis, Implications, and Treatment Recommendations
- Veteran With Alcohol Use Disorder and Acute Pancreatitis
- Misleading Diagnosis of Idiopathic Pulmonary Fibrosis: A Clinical Concern
- Addressing the Needs of Patients With Chronic Pain
- Singing Praises, Naming Names
- The Clinicians’ Role in Building a System of Care: Army Behavioral Health Since 2001

Click here to access the February 2018 Digital Edition.
Table of Contents
- A Year 3 Progress Report on Graduate Medical Education Expansion in the Veterans Choice Act
- Postpartum Psychosis in a Young VA Patient: Diagnosis, Implications, and Treatment Recommendations
- Veteran With Alcohol Use Disorder and Acute Pancreatitis
- Misleading Diagnosis of Idiopathic Pulmonary Fibrosis: A Clinical Concern
- Addressing the Needs of Patients With Chronic Pain
- Singing Praises, Naming Names
- The Clinicians’ Role in Building a System of Care: Army Behavioral Health Since 2001

Click here to access the February 2018 Digital Edition.
Table of Contents
- A Year 3 Progress Report on Graduate Medical Education Expansion in the Veterans Choice Act
- Postpartum Psychosis in a Young VA Patient: Diagnosis, Implications, and Treatment Recommendations
- Veteran With Alcohol Use Disorder and Acute Pancreatitis
- Misleading Diagnosis of Idiopathic Pulmonary Fibrosis: A Clinical Concern
- Addressing the Needs of Patients With Chronic Pain
- Singing Praises, Naming Names
- The Clinicians’ Role in Building a System of Care: Army Behavioral Health Since 2001

Rituximab Still Proves Safe Long Term
Rituximab, a B-cell–depleting agent, has been found safe and effective in clinical trials of patients with multiple sclerosis and patients with rheumatoid arthritis, among others. However, progressive multifocal leukoencephalopathy (PML) and malignancies have been reported in patients with lymphoma, rheumatoid arthritis, and lupus who also received multiple immunosuppressive therapies, say researchers from Wayne State University in Michigan and University of Chicago in Illinois. In studies with ocrelizumab, which also depletes B-cells, adverse effects (AEs) have included infections, such as, herpes virus-associated infection, and neoplasms.
Although most research has found rituximab and ocrelizumab safe and effective, there is a “paucity of literature” on the safety of continuous B-cell depletion over a long period, the researchers say. They conducted a retrospective study involving 29 patients with immune-mediated neurologic disorders who received continuous cycles of rituximab infusions every 6 to 9 months for up to 7 years. Although small, the study was longer than the trials with ocrelizumab in multiple sclerosis . The mean duration of treatment was 51 months; with a mean of 9 treatment cycles.
The researchers found a low incidence of adverse events and prolonged rituximab-induced B-cell depletion did not lead to any life-threatening AEs, including malignancy. Overall, 32 AEs were reported. Four were serious; 3 were noted after 9 cycles (48 months), and 1 after 11 cycles (60 months). There were no cases of PML or malignancies. Repeated rituximab infusions were well tolerated The rate of AEs remained low over the 7-year observation period.
Source:
Memon AB, Javed A, Caon C, et al. PLoS ONE. 2018;13(1):e0190425.
doi: 10.1371/journal.pone.0190425.
Rituximab, a B-cell–depleting agent, has been found safe and effective in clinical trials of patients with multiple sclerosis and patients with rheumatoid arthritis, among others. However, progressive multifocal leukoencephalopathy (PML) and malignancies have been reported in patients with lymphoma, rheumatoid arthritis, and lupus who also received multiple immunosuppressive therapies, say researchers from Wayne State University in Michigan and University of Chicago in Illinois. In studies with ocrelizumab, which also depletes B-cells, adverse effects (AEs) have included infections, such as, herpes virus-associated infection, and neoplasms.
Although most research has found rituximab and ocrelizumab safe and effective, there is a “paucity of literature” on the safety of continuous B-cell depletion over a long period, the researchers say. They conducted a retrospective study involving 29 patients with immune-mediated neurologic disorders who received continuous cycles of rituximab infusions every 6 to 9 months for up to 7 years. Although small, the study was longer than the trials with ocrelizumab in multiple sclerosis . The mean duration of treatment was 51 months; with a mean of 9 treatment cycles.
The researchers found a low incidence of adverse events and prolonged rituximab-induced B-cell depletion did not lead to any life-threatening AEs, including malignancy. Overall, 32 AEs were reported. Four were serious; 3 were noted after 9 cycles (48 months), and 1 after 11 cycles (60 months). There were no cases of PML or malignancies. Repeated rituximab infusions were well tolerated The rate of AEs remained low over the 7-year observation period.
Source:
Memon AB, Javed A, Caon C, et al. PLoS ONE. 2018;13(1):e0190425.
doi: 10.1371/journal.pone.0190425.
Rituximab, a B-cell–depleting agent, has been found safe and effective in clinical trials of patients with multiple sclerosis and patients with rheumatoid arthritis, among others. However, progressive multifocal leukoencephalopathy (PML) and malignancies have been reported in patients with lymphoma, rheumatoid arthritis, and lupus who also received multiple immunosuppressive therapies, say researchers from Wayne State University in Michigan and University of Chicago in Illinois. In studies with ocrelizumab, which also depletes B-cells, adverse effects (AEs) have included infections, such as, herpes virus-associated infection, and neoplasms.
Although most research has found rituximab and ocrelizumab safe and effective, there is a “paucity of literature” on the safety of continuous B-cell depletion over a long period, the researchers say. They conducted a retrospective study involving 29 patients with immune-mediated neurologic disorders who received continuous cycles of rituximab infusions every 6 to 9 months for up to 7 years. Although small, the study was longer than the trials with ocrelizumab in multiple sclerosis . The mean duration of treatment was 51 months; with a mean of 9 treatment cycles.
The researchers found a low incidence of adverse events and prolonged rituximab-induced B-cell depletion did not lead to any life-threatening AEs, including malignancy. Overall, 32 AEs were reported. Four were serious; 3 were noted after 9 cycles (48 months), and 1 after 11 cycles (60 months). There were no cases of PML or malignancies. Repeated rituximab infusions were well tolerated The rate of AEs remained low over the 7-year observation period.
Source:
Memon AB, Javed A, Caon C, et al. PLoS ONE. 2018;13(1):e0190425.
doi: 10.1371/journal.pone.0190425.