User login
BRAIN Initiative could help end mental illness stigma
BETHESDA, MD – Top researchers are optimistic that billions in funding for basic neuroscience research made possible by the 21st Century Cures Act will lead to breakthroughs in the understanding of brain function and dysfunction that could help end the stigma of mental illness.
Under the new law, $6.3 billion are slated for health initiatives over the next decade. The Cancer Moonshot will receive $1.8 billion, precision medicine will receive just under $1.5 billion, and the BRAIN Initiative (Brain Research Through Advancing Innovative Neurotechnologies) will receive just over that amount.
“I know there have been some anxieties about the program’s fragilities given the change in administration,” Dr. Collins told an audience at a meeting of the NIH Brain Institute investigators. “But [passage of the 21st Century Cures Act] should be a source of great reassurance.”
The actual cost of the BRAIN Initiative is expected to top $4.5 billion by the year 2025, funded by a mix of federal and private monies. The National Institute of Mental Health’s typical annual budget is just under $1.5 billion – essentially the same as the amount provided for in the new law. The funds will benefit neuroscience generally, and if successful, understanding of the brain specifically.
The BRAIN Initiative funding is not for clinical studies, however. Instead, it will be spread across what Dr. Collins dubbed, “Team Science,” a multidisciplinary band of investigators, all of whom are expected to help not only identify the brain’s biologic processes but also create the tools needed to reengineer the brain’s circuitry when it malfunctions. Some current BRAIN projects Dr. Collins singled out as worth watching were the use of ultrasound neuromodulation in nonhuman primates, the analysis of individual methylone in cell nuclei, and the development of ultrahigh-resolution 3D brain imaging technology.
“We are moving toward a full bore effort toward technologies that allow us to see what is happening in real time in circuits in the brain,” Dr. Collins said. “Maybe there are certain fundamental operating principles that may have eluded us in the past but which may start to appear.”
In 2016, there were as many neuroscientists as there were engineers of all types – 141 each – named as the principal investigators of projects funded by the initiative, according to NIMH Director Joshua A. Gordon, MD, PhD, who also spoke at the meeting. Among the other disciplines Dr. Gordon said were represented in the 2016 roster of investigators were 61 radiologists, 33 biostatisticians, 47 neurosurgeons, and 69 psychiatrists and psychologists.
The enormous focus on bench science – about 85% of all NIMH funding is for nonclinical research – is not without its critics. In a recent editorial, several prominent academic clinicians urged the NIMH to spend less on futuristic neuroscience and more on the application of therapies for those with mental illness, and on their psychosocial needs (Br J Psychiatry. 2016;208[6]507-9). The last-minute inclusion in the 21st Century Cures Act of several provisions that will have a direct impact in the near future on the plight of those with severe mental illness has the potential to allay some concerns, however.
“My hope is that the [law] will be interpreted from the perspective of making effective treatments available for ... those who are already ill,” Susan M. Essock, PhD, the Edna L. Edison Professor of Medical Psychology (in Psychiatry) at Columbia University, New York, and a cosigner of the editorial, said in an interview.
The law aims to drive evidence-based grant making for programs such as the Recovery After an Initial Schizophrenia Episode (RAISE) integrated care model and expands assisted outpatient treatment for children with serious emotional disturbance, or adults with serious mental illness, among several other provisions.
For some, the initiative’s cross-specialty structure and hefty price tag are worthwhile if it means filling the void of basic science necessary to explain the mechanics of mental illness, a void often filled with attitudes leading to the stigmatization of people who are ill.
Dr. Mayberg said she believes that a “systems approach” to behavior will emerge over time as interdisciplinary BRAIN Initiative researchers are given the freedom to make necessary discoveries, often when least expected, and are urged to share what they find. “Real fundamental changes happen by accident, in my experience. They are rarely planned. It happens when there are a lot of clues. A diversified portfolio has to be the answer, because nobody is smart enough to do it all [alone]. The goal is diversified, evidence-based approaches. ... Science has to make fundamental progress on how the brain works, because we’re obviously missing something clinically,” she said.
The ultimate goals of the initiative are to understand human beings and to improve the human condition by alleviating suffering, according to Brian Litt, MD, professor of neurology and bioengineering at the University of Pennsylvania, Philadelphia, and a moderator of a panel focused on the clinical implications of BRAIN research.
“If you think about the fundamental problems that affect our society and many around the world, they’re not just diseases like Parkinson’s or epilepsy, they are the disorders of mood, addiction, violence, anger, and a number of other conditions that affect behavior that are part of the neurological spectrum that affects the way our society functions,” Dr. Litt, a clinician specializing in treating people with epilepsy, told the audience.
Rather than create a neuroengineered super race, he said, fundamental neuroscience is about helping humans reach their fullest potential. For his support of this, Dr. Litt said he makes “no apologies. As a clinician, I am not sorry to be involved in fundamental neuroscience where ... incremental discoveries can create cures.”
As science favors cures, policy inevitably will evolve, perhaps even making current interventions relics of ignorance. “Psychosocial intervention is what you do when you have an absence of knowing how to cure,” said Dr. Mayberg. “It might be that [currently] what we treat is maladaptation to the primary problem.”
The key for clinicians, she said, will be to keep up with the latest literature. “Clinicians have to be aware of what’s exciting, of what should be in their armamentarium.”
Dr. Collins, Dr. Gordon, Dr. Essock, and Dr. Mayberg had no disclosures. Dr. Litt has cofounded several companies, including Blackfynn and Neuropace, and has served as a consultant and licensed technology to several companies.
[email protected]
On Twitter @whitneymcknight
BETHESDA, MD – Top researchers are optimistic that billions in funding for basic neuroscience research made possible by the 21st Century Cures Act will lead to breakthroughs in the understanding of brain function and dysfunction that could help end the stigma of mental illness.
Under the new law, $6.3 billion are slated for health initiatives over the next decade. The Cancer Moonshot will receive $1.8 billion, precision medicine will receive just under $1.5 billion, and the BRAIN Initiative (Brain Research Through Advancing Innovative Neurotechnologies) will receive just over that amount.
“I know there have been some anxieties about the program’s fragilities given the change in administration,” Dr. Collins told an audience at a meeting of the NIH Brain Institute investigators. “But [passage of the 21st Century Cures Act] should be a source of great reassurance.”
The actual cost of the BRAIN Initiative is expected to top $4.5 billion by the year 2025, funded by a mix of federal and private monies. The National Institute of Mental Health’s typical annual budget is just under $1.5 billion – essentially the same as the amount provided for in the new law. The funds will benefit neuroscience generally, and if successful, understanding of the brain specifically.
The BRAIN Initiative funding is not for clinical studies, however. Instead, it will be spread across what Dr. Collins dubbed, “Team Science,” a multidisciplinary band of investigators, all of whom are expected to help not only identify the brain’s biologic processes but also create the tools needed to reengineer the brain’s circuitry when it malfunctions. Some current BRAIN projects Dr. Collins singled out as worth watching were the use of ultrasound neuromodulation in nonhuman primates, the analysis of individual methylone in cell nuclei, and the development of ultrahigh-resolution 3D brain imaging technology.
“We are moving toward a full bore effort toward technologies that allow us to see what is happening in real time in circuits in the brain,” Dr. Collins said. “Maybe there are certain fundamental operating principles that may have eluded us in the past but which may start to appear.”
In 2016, there were as many neuroscientists as there were engineers of all types – 141 each – named as the principal investigators of projects funded by the initiative, according to NIMH Director Joshua A. Gordon, MD, PhD, who also spoke at the meeting. Among the other disciplines Dr. Gordon said were represented in the 2016 roster of investigators were 61 radiologists, 33 biostatisticians, 47 neurosurgeons, and 69 psychiatrists and psychologists.
The enormous focus on bench science – about 85% of all NIMH funding is for nonclinical research – is not without its critics. In a recent editorial, several prominent academic clinicians urged the NIMH to spend less on futuristic neuroscience and more on the application of therapies for those with mental illness, and on their psychosocial needs (Br J Psychiatry. 2016;208[6]507-9). The last-minute inclusion in the 21st Century Cures Act of several provisions that will have a direct impact in the near future on the plight of those with severe mental illness has the potential to allay some concerns, however.
“My hope is that the [law] will be interpreted from the perspective of making effective treatments available for ... those who are already ill,” Susan M. Essock, PhD, the Edna L. Edison Professor of Medical Psychology (in Psychiatry) at Columbia University, New York, and a cosigner of the editorial, said in an interview.
The law aims to drive evidence-based grant making for programs such as the Recovery After an Initial Schizophrenia Episode (RAISE) integrated care model and expands assisted outpatient treatment for children with serious emotional disturbance, or adults with serious mental illness, among several other provisions.
For some, the initiative’s cross-specialty structure and hefty price tag are worthwhile if it means filling the void of basic science necessary to explain the mechanics of mental illness, a void often filled with attitudes leading to the stigmatization of people who are ill.
Dr. Mayberg said she believes that a “systems approach” to behavior will emerge over time as interdisciplinary BRAIN Initiative researchers are given the freedom to make necessary discoveries, often when least expected, and are urged to share what they find. “Real fundamental changes happen by accident, in my experience. They are rarely planned. It happens when there are a lot of clues. A diversified portfolio has to be the answer, because nobody is smart enough to do it all [alone]. The goal is diversified, evidence-based approaches. ... Science has to make fundamental progress on how the brain works, because we’re obviously missing something clinically,” she said.
The ultimate goals of the initiative are to understand human beings and to improve the human condition by alleviating suffering, according to Brian Litt, MD, professor of neurology and bioengineering at the University of Pennsylvania, Philadelphia, and a moderator of a panel focused on the clinical implications of BRAIN research.
“If you think about the fundamental problems that affect our society and many around the world, they’re not just diseases like Parkinson’s or epilepsy, they are the disorders of mood, addiction, violence, anger, and a number of other conditions that affect behavior that are part of the neurological spectrum that affects the way our society functions,” Dr. Litt, a clinician specializing in treating people with epilepsy, told the audience.
Rather than create a neuroengineered super race, he said, fundamental neuroscience is about helping humans reach their fullest potential. For his support of this, Dr. Litt said he makes “no apologies. As a clinician, I am not sorry to be involved in fundamental neuroscience where ... incremental discoveries can create cures.”
As science favors cures, policy inevitably will evolve, perhaps even making current interventions relics of ignorance. “Psychosocial intervention is what you do when you have an absence of knowing how to cure,” said Dr. Mayberg. “It might be that [currently] what we treat is maladaptation to the primary problem.”
The key for clinicians, she said, will be to keep up with the latest literature. “Clinicians have to be aware of what’s exciting, of what should be in their armamentarium.”
Dr. Collins, Dr. Gordon, Dr. Essock, and Dr. Mayberg had no disclosures. Dr. Litt has cofounded several companies, including Blackfynn and Neuropace, and has served as a consultant and licensed technology to several companies.
[email protected]
On Twitter @whitneymcknight
BETHESDA, MD – Top researchers are optimistic that billions in funding for basic neuroscience research made possible by the 21st Century Cures Act will lead to breakthroughs in the understanding of brain function and dysfunction that could help end the stigma of mental illness.
Under the new law, $6.3 billion are slated for health initiatives over the next decade. The Cancer Moonshot will receive $1.8 billion, precision medicine will receive just under $1.5 billion, and the BRAIN Initiative (Brain Research Through Advancing Innovative Neurotechnologies) will receive just over that amount.
“I know there have been some anxieties about the program’s fragilities given the change in administration,” Dr. Collins told an audience at a meeting of the NIH Brain Institute investigators. “But [passage of the 21st Century Cures Act] should be a source of great reassurance.”
The actual cost of the BRAIN Initiative is expected to top $4.5 billion by the year 2025, funded by a mix of federal and private monies. The National Institute of Mental Health’s typical annual budget is just under $1.5 billion – essentially the same as the amount provided for in the new law. The funds will benefit neuroscience generally, and if successful, understanding of the brain specifically.
The BRAIN Initiative funding is not for clinical studies, however. Instead, it will be spread across what Dr. Collins dubbed, “Team Science,” a multidisciplinary band of investigators, all of whom are expected to help not only identify the brain’s biologic processes but also create the tools needed to reengineer the brain’s circuitry when it malfunctions. Some current BRAIN projects Dr. Collins singled out as worth watching were the use of ultrasound neuromodulation in nonhuman primates, the analysis of individual methylone in cell nuclei, and the development of ultrahigh-resolution 3D brain imaging technology.
“We are moving toward a full bore effort toward technologies that allow us to see what is happening in real time in circuits in the brain,” Dr. Collins said. “Maybe there are certain fundamental operating principles that may have eluded us in the past but which may start to appear.”
In 2016, there were as many neuroscientists as there were engineers of all types – 141 each – named as the principal investigators of projects funded by the initiative, according to NIMH Director Joshua A. Gordon, MD, PhD, who also spoke at the meeting. Among the other disciplines Dr. Gordon said were represented in the 2016 roster of investigators were 61 radiologists, 33 biostatisticians, 47 neurosurgeons, and 69 psychiatrists and psychologists.
The enormous focus on bench science – about 85% of all NIMH funding is for nonclinical research – is not without its critics. In a recent editorial, several prominent academic clinicians urged the NIMH to spend less on futuristic neuroscience and more on the application of therapies for those with mental illness, and on their psychosocial needs (Br J Psychiatry. 2016;208[6]507-9). The last-minute inclusion in the 21st Century Cures Act of several provisions that will have a direct impact in the near future on the plight of those with severe mental illness has the potential to allay some concerns, however.
“My hope is that the [law] will be interpreted from the perspective of making effective treatments available for ... those who are already ill,” Susan M. Essock, PhD, the Edna L. Edison Professor of Medical Psychology (in Psychiatry) at Columbia University, New York, and a cosigner of the editorial, said in an interview.
The law aims to drive evidence-based grant making for programs such as the Recovery After an Initial Schizophrenia Episode (RAISE) integrated care model and expands assisted outpatient treatment for children with serious emotional disturbance, or adults with serious mental illness, among several other provisions.
For some, the initiative’s cross-specialty structure and hefty price tag are worthwhile if it means filling the void of basic science necessary to explain the mechanics of mental illness, a void often filled with attitudes leading to the stigmatization of people who are ill.
Dr. Mayberg said she believes that a “systems approach” to behavior will emerge over time as interdisciplinary BRAIN Initiative researchers are given the freedom to make necessary discoveries, often when least expected, and are urged to share what they find. “Real fundamental changes happen by accident, in my experience. They are rarely planned. It happens when there are a lot of clues. A diversified portfolio has to be the answer, because nobody is smart enough to do it all [alone]. The goal is diversified, evidence-based approaches. ... Science has to make fundamental progress on how the brain works, because we’re obviously missing something clinically,” she said.
The ultimate goals of the initiative are to understand human beings and to improve the human condition by alleviating suffering, according to Brian Litt, MD, professor of neurology and bioengineering at the University of Pennsylvania, Philadelphia, and a moderator of a panel focused on the clinical implications of BRAIN research.
“If you think about the fundamental problems that affect our society and many around the world, they’re not just diseases like Parkinson’s or epilepsy, they are the disorders of mood, addiction, violence, anger, and a number of other conditions that affect behavior that are part of the neurological spectrum that affects the way our society functions,” Dr. Litt, a clinician specializing in treating people with epilepsy, told the audience.
Rather than create a neuroengineered super race, he said, fundamental neuroscience is about helping humans reach their fullest potential. For his support of this, Dr. Litt said he makes “no apologies. As a clinician, I am not sorry to be involved in fundamental neuroscience where ... incremental discoveries can create cures.”
As science favors cures, policy inevitably will evolve, perhaps even making current interventions relics of ignorance. “Psychosocial intervention is what you do when you have an absence of knowing how to cure,” said Dr. Mayberg. “It might be that [currently] what we treat is maladaptation to the primary problem.”
The key for clinicians, she said, will be to keep up with the latest literature. “Clinicians have to be aware of what’s exciting, of what should be in their armamentarium.”
Dr. Collins, Dr. Gordon, Dr. Essock, and Dr. Mayberg had no disclosures. Dr. Litt has cofounded several companies, including Blackfynn and Neuropace, and has served as a consultant and licensed technology to several companies.
[email protected]
On Twitter @whitneymcknight
EXPERT ANALYSIS AT AN NIH ADVISORY COUNCIL MEETING
Pig-in-a-poke health insurance, dermatologists never say never!
One of the most acute issues facing dermatologists is the implementation of narrow networks by health insurers. Inaccurate physician rosters have long been a problem that insurers have been indifferent to, but this was not an acute problem since almost all providers were included in the networks.
However, the recent “ramp up” because of increased costs and cuts associated with the Affordable Care Act (ACA) has resulted in the delisting of many thousands of physicians (including hundreds of dermatologists) from insurance plans and refusal to allow them to enroll in new plans. All in a time of physician shortages and an acute shortage of dermatologists.
When I was president of the American Academy of Dermatology, I made it a priority that patients and the government, who pay or subsidize insurance companies for this hollow insurance, understand how they were being cheated on their health care dollars – or, as we say in Kentucky, being sold a “pig in a poke.” A pig in a poke is a pig (or another small farm animal) carried to market in a bag with only the head sticking out, disguising the skinny product within.
The AAD staff and I went to the White House, Congress, the Centers for Medicare and Medicaid Services (CMS), AHIP (America’s Health Insurance Plans trade association), PhRMA (Pharmaceutical Research and Manufacturers of America), and the Medicare Payment Advisory Commission (MedPac); and spoke to the Office of Inspector General (OIG), which agreed to investigate. We and the dermatology patient advocacy groups organized a letter writing campaign and solicited letters and complaints from members. We contacted the state health insurance commissioners and testified at their hearings. Our fellow dermatologist and AMA board of trustee member Jack Resneck, MD, researched and wrote an article in JAMA Dermatology, demonstrating how pathetically inaccurate the insurance company physician networks are (2014;150[12]:1290-7). We beat the drum loud and long, and it worked!
The OIG issued a scathing report about the lack of follow through by CMS on billions of health care dollars being paid by the federal government to Medicare Advantage plans. CMS agreed with their report, and agreed to investigate. A CMS survey reported in October 2016 that almost half of the doctor listings in their Medicare Advantage (Humana, Aetna, United, and Anthem senior plans) directories contained incorrect information, including inaccurate names and addresses; multiple, duplicate listings; and doctors who have died. CMS concluded that they expect this performance to improve over the next three study periods – or they will start to assess $25,000 a day in penalties.
Also published last year, a “secret shopper” survey of primary care providers in five insurance marketplace pricing regions found that fewer than 30% of consumers were able to schedule an appointment with the physician they had first selected (Health Aff [Millwood]. 2016 Jul 1;35[7]:1160-6).
Folks, the only way to improve the accuracy is to delete duplicated listings, delete physicians no longer seeing new patients, delete physicians incorrectly listed, and delete those who have died or moved on. We have to continue this effort on a state by state level, or insurers will quickly backslide, because if there are no doctors to see, there are no bills for them to pay.
Accurate rosters will reveal huge gaps in their directories, and insurers will be forced to recontract with delisted physicians. Physicians should watch for and anticipate this, and should not sign up at a disadvantage. Patients must continue to pressure their health insurers both personally and through their human resources departments at work. No one should have to buy “a pig in a poke.” This is huge national victory, for patients and physicians. Dermatologists, you should be proud of your AAD leading the charge, and following through on getting this issue addressed and fixed.
Dr. Coldiron is a past president of the American Academy of Dermatology. He is currently in private practice, but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics.
One of the most acute issues facing dermatologists is the implementation of narrow networks by health insurers. Inaccurate physician rosters have long been a problem that insurers have been indifferent to, but this was not an acute problem since almost all providers were included in the networks.
However, the recent “ramp up” because of increased costs and cuts associated with the Affordable Care Act (ACA) has resulted in the delisting of many thousands of physicians (including hundreds of dermatologists) from insurance plans and refusal to allow them to enroll in new plans. All in a time of physician shortages and an acute shortage of dermatologists.
When I was president of the American Academy of Dermatology, I made it a priority that patients and the government, who pay or subsidize insurance companies for this hollow insurance, understand how they were being cheated on their health care dollars – or, as we say in Kentucky, being sold a “pig in a poke.” A pig in a poke is a pig (or another small farm animal) carried to market in a bag with only the head sticking out, disguising the skinny product within.
The AAD staff and I went to the White House, Congress, the Centers for Medicare and Medicaid Services (CMS), AHIP (America’s Health Insurance Plans trade association), PhRMA (Pharmaceutical Research and Manufacturers of America), and the Medicare Payment Advisory Commission (MedPac); and spoke to the Office of Inspector General (OIG), which agreed to investigate. We and the dermatology patient advocacy groups organized a letter writing campaign and solicited letters and complaints from members. We contacted the state health insurance commissioners and testified at their hearings. Our fellow dermatologist and AMA board of trustee member Jack Resneck, MD, researched and wrote an article in JAMA Dermatology, demonstrating how pathetically inaccurate the insurance company physician networks are (2014;150[12]:1290-7). We beat the drum loud and long, and it worked!
The OIG issued a scathing report about the lack of follow through by CMS on billions of health care dollars being paid by the federal government to Medicare Advantage plans. CMS agreed with their report, and agreed to investigate. A CMS survey reported in October 2016 that almost half of the doctor listings in their Medicare Advantage (Humana, Aetna, United, and Anthem senior plans) directories contained incorrect information, including inaccurate names and addresses; multiple, duplicate listings; and doctors who have died. CMS concluded that they expect this performance to improve over the next three study periods – or they will start to assess $25,000 a day in penalties.
Also published last year, a “secret shopper” survey of primary care providers in five insurance marketplace pricing regions found that fewer than 30% of consumers were able to schedule an appointment with the physician they had first selected (Health Aff [Millwood]. 2016 Jul 1;35[7]:1160-6).
Folks, the only way to improve the accuracy is to delete duplicated listings, delete physicians no longer seeing new patients, delete physicians incorrectly listed, and delete those who have died or moved on. We have to continue this effort on a state by state level, or insurers will quickly backslide, because if there are no doctors to see, there are no bills for them to pay.
Accurate rosters will reveal huge gaps in their directories, and insurers will be forced to recontract with delisted physicians. Physicians should watch for and anticipate this, and should not sign up at a disadvantage. Patients must continue to pressure their health insurers both personally and through their human resources departments at work. No one should have to buy “a pig in a poke.” This is huge national victory, for patients and physicians. Dermatologists, you should be proud of your AAD leading the charge, and following through on getting this issue addressed and fixed.
Dr. Coldiron is a past president of the American Academy of Dermatology. He is currently in private practice, but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics.
One of the most acute issues facing dermatologists is the implementation of narrow networks by health insurers. Inaccurate physician rosters have long been a problem that insurers have been indifferent to, but this was not an acute problem since almost all providers were included in the networks.
However, the recent “ramp up” because of increased costs and cuts associated with the Affordable Care Act (ACA) has resulted in the delisting of many thousands of physicians (including hundreds of dermatologists) from insurance plans and refusal to allow them to enroll in new plans. All in a time of physician shortages and an acute shortage of dermatologists.
When I was president of the American Academy of Dermatology, I made it a priority that patients and the government, who pay or subsidize insurance companies for this hollow insurance, understand how they were being cheated on their health care dollars – or, as we say in Kentucky, being sold a “pig in a poke.” A pig in a poke is a pig (or another small farm animal) carried to market in a bag with only the head sticking out, disguising the skinny product within.
The AAD staff and I went to the White House, Congress, the Centers for Medicare and Medicaid Services (CMS), AHIP (America’s Health Insurance Plans trade association), PhRMA (Pharmaceutical Research and Manufacturers of America), and the Medicare Payment Advisory Commission (MedPac); and spoke to the Office of Inspector General (OIG), which agreed to investigate. We and the dermatology patient advocacy groups organized a letter writing campaign and solicited letters and complaints from members. We contacted the state health insurance commissioners and testified at their hearings. Our fellow dermatologist and AMA board of trustee member Jack Resneck, MD, researched and wrote an article in JAMA Dermatology, demonstrating how pathetically inaccurate the insurance company physician networks are (2014;150[12]:1290-7). We beat the drum loud and long, and it worked!
The OIG issued a scathing report about the lack of follow through by CMS on billions of health care dollars being paid by the federal government to Medicare Advantage plans. CMS agreed with their report, and agreed to investigate. A CMS survey reported in October 2016 that almost half of the doctor listings in their Medicare Advantage (Humana, Aetna, United, and Anthem senior plans) directories contained incorrect information, including inaccurate names and addresses; multiple, duplicate listings; and doctors who have died. CMS concluded that they expect this performance to improve over the next three study periods – or they will start to assess $25,000 a day in penalties.
Also published last year, a “secret shopper” survey of primary care providers in five insurance marketplace pricing regions found that fewer than 30% of consumers were able to schedule an appointment with the physician they had first selected (Health Aff [Millwood]. 2016 Jul 1;35[7]:1160-6).
Folks, the only way to improve the accuracy is to delete duplicated listings, delete physicians no longer seeing new patients, delete physicians incorrectly listed, and delete those who have died or moved on. We have to continue this effort on a state by state level, or insurers will quickly backslide, because if there are no doctors to see, there are no bills for them to pay.
Accurate rosters will reveal huge gaps in their directories, and insurers will be forced to recontract with delisted physicians. Physicians should watch for and anticipate this, and should not sign up at a disadvantage. Patients must continue to pressure their health insurers both personally and through their human resources departments at work. No one should have to buy “a pig in a poke.” This is huge national victory, for patients and physicians. Dermatologists, you should be proud of your AAD leading the charge, and following through on getting this issue addressed and fixed.
Dr. Coldiron is a past president of the American Academy of Dermatology. He is currently in private practice, but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics.
In NSCLC, delayed chemo yields survival benefit comparable to early chemo
Patients with non–small-cell lung cancer (NSCLC) for whom adjuvant chemotherapy must be delayed for as long as 18 weeks have mortality outcomes that are no worse than those of patients who start chemotherapy soon after surgery, and those who undergo delayed chemotherapy have a significantly lower risk for death than patients who have no chemotherapy at all, investigators report.
A retrospective review of data on 12,473 patients with previously untreated NSCLC showed that there were no significant differences in 5-year overall survival (OS) estimates among patients who started multi-agent chemotherapy at 18-38 days postoperatively, from 39 to 56 days after surgery (the reference interval), or from 57 to 127 days after surgery, reported Daniel J. Boffa, MD, of Yale University, New Haven, Conn., and his colleagues.
In addition, when they used propensity score matching to pair patients who received chemotherapy with patients who did not undergo chemotherapy, they found that even late chemotherapy was associated with a significantly lower risk for death.
“Clinicians should still consider chemotherapy in appropriately selected patients that are healthy enough to tolerate it, up to 4 months after NSCLC resection. Further study is warranted to confirm these findings,” the investigators concluded (JAMA Oncol. 2017 Jan. 5 doi: 10.1001/jamaoncol.2016.5829).
In the retrospective review of records from the National Cancer Database, the investigators limited the study to patients for whom chemotherapy is typically prescribed: those with lymph node metastases, tumors 4 cm or larger, and/or local extension of disease. They looked at the association between the time to initiation of adjuvant chemotherapy and survival using Cox modeling with restricted cubic splines, a validated statistical method for evaluating links between survival and independent variables.
Dr. Boffa and his associates found that the unadjusted Kaplan-Meier 5-year OS estimates did not differ between the groups, at 53% for the early chemotherapy group (hazard ratio [HR] vs. the reference group, 1.009, P = .79), 55% for the reference group, and 53% for the later chemotherapy group (HR 1.037, P = .27).
Comparing adjuvant chemotherapy timing on the efficacy of surgery alone in patients matched by tumor stage and other features, the researchers found that chemotherapy started during any of the three intervals was associated with an approximately 34% reduction in risk of death compared with no chemotherapy (HR for the respective time intervals 0.672, 0.645, and 0.664; P less than .001 for each comparison).
The study helps to clarify for clinicians the benefits of adjuvant chemotherapy in select patients with NSCLC in a real-world setting, Howard (Jack) West, MD, of the Swedish Cancer Institute, Seattle, said in an accompanying editorial (JAMA Oncol. 2017 Jan. 5 doi: 10.1001/jamaoncol.2016.5798).
“While retrospective data cannot define the benefit of delayed adjuvant chemotherapy with the clarity of a prospective randomized trial, we must remember that in the land of the blind, the one-eyed man is king; these limited data inject an evidence-based answer for a very common clinical question for which we have been forced by necessity to rely only on our best judgments,” he wrote.
The study was internally supported. The authors and Dr. West reported no conflict of interest disclosures.
Patients with non–small-cell lung cancer (NSCLC) for whom adjuvant chemotherapy must be delayed for as long as 18 weeks have mortality outcomes that are no worse than those of patients who start chemotherapy soon after surgery, and those who undergo delayed chemotherapy have a significantly lower risk for death than patients who have no chemotherapy at all, investigators report.
A retrospective review of data on 12,473 patients with previously untreated NSCLC showed that there were no significant differences in 5-year overall survival (OS) estimates among patients who started multi-agent chemotherapy at 18-38 days postoperatively, from 39 to 56 days after surgery (the reference interval), or from 57 to 127 days after surgery, reported Daniel J. Boffa, MD, of Yale University, New Haven, Conn., and his colleagues.
In addition, when they used propensity score matching to pair patients who received chemotherapy with patients who did not undergo chemotherapy, they found that even late chemotherapy was associated with a significantly lower risk for death.
“Clinicians should still consider chemotherapy in appropriately selected patients that are healthy enough to tolerate it, up to 4 months after NSCLC resection. Further study is warranted to confirm these findings,” the investigators concluded (JAMA Oncol. 2017 Jan. 5 doi: 10.1001/jamaoncol.2016.5829).
In the retrospective review of records from the National Cancer Database, the investigators limited the study to patients for whom chemotherapy is typically prescribed: those with lymph node metastases, tumors 4 cm or larger, and/or local extension of disease. They looked at the association between the time to initiation of adjuvant chemotherapy and survival using Cox modeling with restricted cubic splines, a validated statistical method for evaluating links between survival and independent variables.
Dr. Boffa and his associates found that the unadjusted Kaplan-Meier 5-year OS estimates did not differ between the groups, at 53% for the early chemotherapy group (hazard ratio [HR] vs. the reference group, 1.009, P = .79), 55% for the reference group, and 53% for the later chemotherapy group (HR 1.037, P = .27).
Comparing adjuvant chemotherapy timing on the efficacy of surgery alone in patients matched by tumor stage and other features, the researchers found that chemotherapy started during any of the three intervals was associated with an approximately 34% reduction in risk of death compared with no chemotherapy (HR for the respective time intervals 0.672, 0.645, and 0.664; P less than .001 for each comparison).
The study helps to clarify for clinicians the benefits of adjuvant chemotherapy in select patients with NSCLC in a real-world setting, Howard (Jack) West, MD, of the Swedish Cancer Institute, Seattle, said in an accompanying editorial (JAMA Oncol. 2017 Jan. 5 doi: 10.1001/jamaoncol.2016.5798).
“While retrospective data cannot define the benefit of delayed adjuvant chemotherapy with the clarity of a prospective randomized trial, we must remember that in the land of the blind, the one-eyed man is king; these limited data inject an evidence-based answer for a very common clinical question for which we have been forced by necessity to rely only on our best judgments,” he wrote.
The study was internally supported. The authors and Dr. West reported no conflict of interest disclosures.
Patients with non–small-cell lung cancer (NSCLC) for whom adjuvant chemotherapy must be delayed for as long as 18 weeks have mortality outcomes that are no worse than those of patients who start chemotherapy soon after surgery, and those who undergo delayed chemotherapy have a significantly lower risk for death than patients who have no chemotherapy at all, investigators report.
A retrospective review of data on 12,473 patients with previously untreated NSCLC showed that there were no significant differences in 5-year overall survival (OS) estimates among patients who started multi-agent chemotherapy at 18-38 days postoperatively, from 39 to 56 days after surgery (the reference interval), or from 57 to 127 days after surgery, reported Daniel J. Boffa, MD, of Yale University, New Haven, Conn., and his colleagues.
In addition, when they used propensity score matching to pair patients who received chemotherapy with patients who did not undergo chemotherapy, they found that even late chemotherapy was associated with a significantly lower risk for death.
“Clinicians should still consider chemotherapy in appropriately selected patients that are healthy enough to tolerate it, up to 4 months after NSCLC resection. Further study is warranted to confirm these findings,” the investigators concluded (JAMA Oncol. 2017 Jan. 5 doi: 10.1001/jamaoncol.2016.5829).
In the retrospective review of records from the National Cancer Database, the investigators limited the study to patients for whom chemotherapy is typically prescribed: those with lymph node metastases, tumors 4 cm or larger, and/or local extension of disease. They looked at the association between the time to initiation of adjuvant chemotherapy and survival using Cox modeling with restricted cubic splines, a validated statistical method for evaluating links between survival and independent variables.
Dr. Boffa and his associates found that the unadjusted Kaplan-Meier 5-year OS estimates did not differ between the groups, at 53% for the early chemotherapy group (hazard ratio [HR] vs. the reference group, 1.009, P = .79), 55% for the reference group, and 53% for the later chemotherapy group (HR 1.037, P = .27).
Comparing adjuvant chemotherapy timing on the efficacy of surgery alone in patients matched by tumor stage and other features, the researchers found that chemotherapy started during any of the three intervals was associated with an approximately 34% reduction in risk of death compared with no chemotherapy (HR for the respective time intervals 0.672, 0.645, and 0.664; P less than .001 for each comparison).
The study helps to clarify for clinicians the benefits of adjuvant chemotherapy in select patients with NSCLC in a real-world setting, Howard (Jack) West, MD, of the Swedish Cancer Institute, Seattle, said in an accompanying editorial (JAMA Oncol. 2017 Jan. 5 doi: 10.1001/jamaoncol.2016.5798).
“While retrospective data cannot define the benefit of delayed adjuvant chemotherapy with the clarity of a prospective randomized trial, we must remember that in the land of the blind, the one-eyed man is king; these limited data inject an evidence-based answer for a very common clinical question for which we have been forced by necessity to rely only on our best judgments,” he wrote.
The study was internally supported. The authors and Dr. West reported no conflict of interest disclosures.
FROM JAMA ONCOLOGY
Key clinical point: Chemotherapy delayed for up to 18 weeks after surgery offers survival benefits comparable to those of earlier chemotherapy in non–small-cell lung cancer.
Major finding: There were no statistical differences in 5-year survival of patients with NSCLC started on chemotherapy either 18-38, 39-56, or 57-127 days after surgery.
Data source: Retrospective observational study of 12,473 patients with untreated NSCLC in the National Cancer Database.
Disclosures: The study was internally supported. The authors and Dr. West reported no conflict of interest disclosures.
Diagnosis and treatment of global endometrial ablation failure
One in seven women suffer with abnormal uterine bleeding during their reproductive years, according to Fraser et al. (Exp Rev Obstet Gynecol. 2009;4:179-89). Heavy menstrual bleeding (menorrhagia) is the most common pattern. Global endometrial ablation has become a very popular surgical technique for women complaining of menorrhagia, disinterested in either medical management or definitive therapy – hysterectomy – or where medical management has failed. With proper patient selection, endometrial ablation yields an 80%-90% success rate in reducing heavy menstrual flow and is associated with a 90% patient satisfaction rate (Cochrane Database Syst Rev. 2009 Oct 7;[4]:CD001501).
Literature is replete with conditions believed to increase risk of endometrial ablation failure. This list includes untreated uterine cornua, endometrial regrowth, the presence of submucous leiomyomas or polyps, abnormal uterine cavity, enlarged uterine cavity (width and/or length), endometrial ablation in a young patient, parity of five or greater, unsuspected adhesiolysis, postablation tubal sterilization syndrome, history of dysmenorrhea, smoking, obesity, prior cesarean section, previous gynecologic surgery, and procedure length. Interestingly, type of global endometrial ablation procedure or original bleeding pattern does not influence failure rate.
In this edition of the Master Class in Gynecologic Surgery, Dr. Morris Wortman discusses not only the prevention of endometrial ablation failure, but also how to treat the problem via conservative surgical management.
Dr. Wortman is a clinical associate professor of obstetrics and gynecology at the University of Rochester (N.Y.) and is the director at the Center for Menstrual Disorders and Reproductive Choice, also in Rochester. Dr. Wortman has lectured extensively on endometrial ablation and has authored several scientific articles in peer reviewed journals.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, and past president of the AAGL and the International Society for Gynecologic Endoscopy. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/SRS fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. He reported being a subinvestigator on a study sponsored by Channel Medsystems. Email him at [email protected].
Why failures occur and how to correct them
BY MORRIS WORTMAN, MD
Since the introduction almost 20 years ago of devices for nonresectoscopic – or “global” – endometrial ablation, the procedure has been widely adopted as the treatment of choice for abnormal uterine bleeding that is refractory to medical management.
Between 400,000 and 500,000 endometrial ablations are done in the United States every year in women who have completed childbearing, and it probably won’t be long before the procedure surpasses hysterectomy in prevalence for the management of abnormal bleeding.
In recent years, the literature has begun to address the incidence of these delayed complications and the requirement for subsequent hysterectomy. A 2007 practice bulletin issued by the American College of Obstetricians and Gynecologists stated that hysterectomy rates within 4 years of endometrial ablation are at least 24% (Obstet Gynecol. 2007 May;109[5]:1233-48). And a study published the following year reported that 26% of 3,681 women undergoing EA at Kaiser Permanente facilities in Northern California required hysterectomy within 8 years (Obstet Gynecol. 2008 Dec;112[6]:1214-20).
It appears that the vast majority of what we now refer to as late-onset EA failures – complications attributable to EA that occur beyond a perioperative period of 1 month – will occur within 5 years. Some EA failures have occurred over 5-10 years, however, and in my practice we have seen late-onset complications occurring 17 or more years after the initial ablation.
In our practice, we are successfully managing delayed complications after GEA using ultrasound-guided reoperative hysteroscopy to fully explore the uterine cavity and excise areas of endometrial growth and other disease. In 2014, we published a retrospective review of 50 women whom we treated for delayed complications after a variety of GEA techniques; almost 90% avoided hysterectomy during a mean follow-up period of 18 months (J Minim Invasive Gynecol. 2014 Mar-Apr;21[2]:238-44).
Our experience since then has included reoperative surgery on more than 115 GEA failures. Additionally, we’ve managed 220 patients who have undergone various hysteroscopic and resectoscopic endometrial ablations, some of which date back to the use of the Nd:YAG laser in the late 1980s.
The fact that late-onset EA failures occur does not mean that hysterectomy should routinely be performed as a first-line treatment for intractable uterine bleeding. Overall, there is much more morbidity associated with hysterectomy than with EA.
What failures do suggest is that there are certain risk factors for late-onset EA complications. Our experience in treating women who have experienced late-onset EA failure has provided us with insight into who may be at greatest risk for late-onset EA failure and how patients can best be selected for the procedure. We’ve also learned more about the diagnosis of delayed complications.
Causes of EA failure
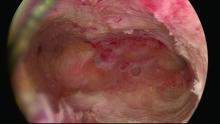
Untreated uterine cornua, and untreated submucous leiomyomas and endometrial polyps, are common causes of EA failure. Among the 50 women included in our retrospective review of ultrasound-guided reoperative hysteroscopy after GEA failure, 44% had intraoperative evidence of untreated cornua and nearly one-fourth had persistent or enlarging submucous leiomyomas.
Contrary to what some believe, most endometrial ablations will not adequately destroy submucous or intramural leiomyomas. Therefore, we recommend that these fibroids be entirely removed immediately before EA.
Moreover, GEA will not always provide adequate thermal destruction to the entire endometrial cavity. The cornua regions are particularly at risk; they are difficult to reach under ideal circumstances, and especially difficult to treat in patients who have a uterine septum or a T-shaped uterus (with the ostia and cornua deeply recessed). We have also seen late-onset EA failures in patients with an extended uterine transverse diameter. The limits of GEA are greatest when a device with a fixed configuration or geometry is used.
A history of abnormal hysteroscopy or other evidence of such anatomic distortions are therefore among the reported risk factors for GEA failure (J Minim Invasive Gynecol. 2015 Mar-Apr;22[3]:323-31). A history of tubal ligation also confers risk; the procedure further increases susceptibility for failure when functioning endometrial tissue remains or regrows at the cornua, because any retrograde menstrual bleeding that occurs will be constrained by the obstructed proximal portion of the fallopian tubes.
Obesity is another risk factor for GEA failure in that the condition increases the risk of endometrial cancer, making the need for reliable biopsies in the case of spotting or other signs or symptoms even more important. On the other hand, obesity may also worsen a patient’s status as a candidate for hysterectomy.
There is much to consider with these patients. For some obese patients, GEA may be less risky than hysterectomy while for others, such as those who also have polycystic ovarian syndrome (in whom the risk for developing endometrial cancer is further increased) the scale may tip in favor of hysterectomy.
Age at the time of the primary GEA may be the single most important risk factor for GEA failure and is an important predictor of success in patient selection. Numerous investigators have shown that women younger than 35 years of age at the time of their EA had a significantly increased risk for hysterectomy, compared with women who were at least 45 years old. The younger the patient, the longer the “bridge” to menopause and the greater the likelihood that bridge will fail.
While age is not necessarily a contraindication, it is worthy of serious consideration. We generally discourage GEA for patients younger than 35. We also advise ensuring that each patient undergoing initial EA is highly self-motivated to have a uterine-sparing procedure; if not, symptoms she may experience later will likely drive her toward hysterectomy anyway.
Additionally, we caution against performing GEA in patients who have chronic pelvic pain; these patients tend to have poorer outcomes with any type of hysteroscopic surgery.
Diagnosing failed EA
Delayed complications manifest in several ways: Renewed and increasing vaginal bleeding after a period of improvement, cyclic pelvic pain (unilateral, bilateral, or suprapubic), or both bleeding and pain. Some women – likely an underreported number of them – present with postmenopausal bleeding and proceed to have unsuccessful attempts at an endometrial biopsy due to EA-associated endometrial scarring.
The cyclic pelvic pain associated with endometrial persistence or regrowth tends to worsen over time and is often described as sharp or laborlike. In our experience, a description of “laborlike” pain and a history of EA is almost fully predictive of a finding of endometrial growth. Often a hematometra can be demonstrated on transvaginal ultrasound, but this isn’t always the case.
Pain typically precedes bleeding in patients who demonstrate both. In such cases, blood from functioning endometrial tissue or other sources becomes blocked from exiting the uterine cavity by EA-induced intrauterine scarring and contracture. Painful uterine contractions then aim to expel the pooled blood. In other cases of pain – mainly those without significant vaginal bleeding – the pain is often attributed to cornual and central hematometra.
For the majority of EA failures, the diagnosis lies in the history and current symptoms. Unfortunately, the traditional methods of assessing the endometrial cavity have little merit for women presenting with delayed-onset EA complications. A sonographically assisted pelvic examination can be useful in evaluating complications, but the interpretation of ultrasounds in women with a prior EA can be challenging and is often beyond the training of most radiologists and gynecologists.
It is not uncommon for images to be incorrectly interpreted in the emergency department or physicians’ offices as “normal” and for such readings to set off a chain of CT scans, MRIs, laparoscopies, ovarian cystectomies, and other procedures that miss the root causes of pain.
Unfortunately, there is little in the literature that describes and defines ultrasound findings after EA. We do know that sonography should be timed with episodes of pain, and that the absence of a demonstrable hematometra does not exclude a diagnosis of EA failure.
Correcting late-onset failures
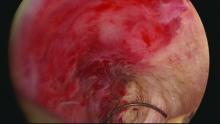
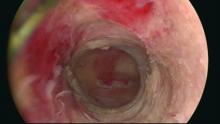
Our office-based operating room is fitted with side-by-side monitors that enable simultaneous sonographic and hysteroscopic views for correction of GEA failures; the rest of the set-up is similar to that of other operative hysteroscopies. However, we do employ a wide variety of resectoscopes with diameters ranging from 13 to 28 Fr. The smaller-diameter scopes are particularly useful for evaluating postmenopausal bleeding in women with a prior EA.
For those inexperienced with ultrasound-guided surgery, the initial resection is often the most challenging. The initial tissue removal is carried out on the thickest observed uterine wall – usually the posterior or anterior wall – and is done with near complete reliance on the ultrasound image. Hysteroscopic visualization is poor at this time because the outflow ports of the continuous flow resectoscope are obstructed by tissue in the narrow tubular cavity.
We then actually remove the resectoscope and clean the outflow ports of clots and debris that may have accumulated. When the scope is reinserted, there is typically sufficient room in the uterine cavity for continuous flow and excellent hysteroscopic visualization.
The sequence of resection from this point on will vary. If we’ve begun on the anterior wall, we’ll move to the posterior and then the two lateral walls to further restore the cavity. Areas of endometrial regrowth will typically be identified at this point and resected. The dissection then will extend upward, usually to within 10 mm of the fundus in the midline as measured by ultrasound. Reconfiguring the loop electrode to a 135- to 160-degree angle can be helpful in the delicate dissection that is required at the fundus.
Once all areas of endometrium have been identified and excised, we will deeply coagulate exposed myometrium with a ball-end electrode. Rarely, we will reach our maximum allowable fluid absorption limit prior to completing the case, a scenario seen in less than 1% of our patients.
In more than 330 reoperative hysteroscopic procedures, we’ve had only one uterine perforation that occurred when we switched ultrasound machines. Very likely, we were too aggressive in removing tissue at the fundus. The patient required a diagnostic laparoscopy but sustained no visceral injury.
Dr. Wortman is a clinical associate professor of obstetrics and gynecology at the University of Rochester (N.Y.) and the director of the Center for Menstrual Disorders and Reproductive Choice in Rochester. He reported having no relevant financial disclosures.
One in seven women suffer with abnormal uterine bleeding during their reproductive years, according to Fraser et al. (Exp Rev Obstet Gynecol. 2009;4:179-89). Heavy menstrual bleeding (menorrhagia) is the most common pattern. Global endometrial ablation has become a very popular surgical technique for women complaining of menorrhagia, disinterested in either medical management or definitive therapy – hysterectomy – or where medical management has failed. With proper patient selection, endometrial ablation yields an 80%-90% success rate in reducing heavy menstrual flow and is associated with a 90% patient satisfaction rate (Cochrane Database Syst Rev. 2009 Oct 7;[4]:CD001501).
Literature is replete with conditions believed to increase risk of endometrial ablation failure. This list includes untreated uterine cornua, endometrial regrowth, the presence of submucous leiomyomas or polyps, abnormal uterine cavity, enlarged uterine cavity (width and/or length), endometrial ablation in a young patient, parity of five or greater, unsuspected adhesiolysis, postablation tubal sterilization syndrome, history of dysmenorrhea, smoking, obesity, prior cesarean section, previous gynecologic surgery, and procedure length. Interestingly, type of global endometrial ablation procedure or original bleeding pattern does not influence failure rate.
In this edition of the Master Class in Gynecologic Surgery, Dr. Morris Wortman discusses not only the prevention of endometrial ablation failure, but also how to treat the problem via conservative surgical management.
Dr. Wortman is a clinical associate professor of obstetrics and gynecology at the University of Rochester (N.Y.) and is the director at the Center for Menstrual Disorders and Reproductive Choice, also in Rochester. Dr. Wortman has lectured extensively on endometrial ablation and has authored several scientific articles in peer reviewed journals.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, and past president of the AAGL and the International Society for Gynecologic Endoscopy. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/SRS fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. He reported being a subinvestigator on a study sponsored by Channel Medsystems. Email him at [email protected].
Why failures occur and how to correct them
BY MORRIS WORTMAN, MD
Since the introduction almost 20 years ago of devices for nonresectoscopic – or “global” – endometrial ablation, the procedure has been widely adopted as the treatment of choice for abnormal uterine bleeding that is refractory to medical management.
Between 400,000 and 500,000 endometrial ablations are done in the United States every year in women who have completed childbearing, and it probably won’t be long before the procedure surpasses hysterectomy in prevalence for the management of abnormal bleeding.
In recent years, the literature has begun to address the incidence of these delayed complications and the requirement for subsequent hysterectomy. A 2007 practice bulletin issued by the American College of Obstetricians and Gynecologists stated that hysterectomy rates within 4 years of endometrial ablation are at least 24% (Obstet Gynecol. 2007 May;109[5]:1233-48). And a study published the following year reported that 26% of 3,681 women undergoing EA at Kaiser Permanente facilities in Northern California required hysterectomy within 8 years (Obstet Gynecol. 2008 Dec;112[6]:1214-20).
It appears that the vast majority of what we now refer to as late-onset EA failures – complications attributable to EA that occur beyond a perioperative period of 1 month – will occur within 5 years. Some EA failures have occurred over 5-10 years, however, and in my practice we have seen late-onset complications occurring 17 or more years after the initial ablation.
In our practice, we are successfully managing delayed complications after GEA using ultrasound-guided reoperative hysteroscopy to fully explore the uterine cavity and excise areas of endometrial growth and other disease. In 2014, we published a retrospective review of 50 women whom we treated for delayed complications after a variety of GEA techniques; almost 90% avoided hysterectomy during a mean follow-up period of 18 months (J Minim Invasive Gynecol. 2014 Mar-Apr;21[2]:238-44).
Our experience since then has included reoperative surgery on more than 115 GEA failures. Additionally, we’ve managed 220 patients who have undergone various hysteroscopic and resectoscopic endometrial ablations, some of which date back to the use of the Nd:YAG laser in the late 1980s.
The fact that late-onset EA failures occur does not mean that hysterectomy should routinely be performed as a first-line treatment for intractable uterine bleeding. Overall, there is much more morbidity associated with hysterectomy than with EA.
What failures do suggest is that there are certain risk factors for late-onset EA complications. Our experience in treating women who have experienced late-onset EA failure has provided us with insight into who may be at greatest risk for late-onset EA failure and how patients can best be selected for the procedure. We’ve also learned more about the diagnosis of delayed complications.
Causes of EA failure
Untreated uterine cornua, and untreated submucous leiomyomas and endometrial polyps, are common causes of EA failure. Among the 50 women included in our retrospective review of ultrasound-guided reoperative hysteroscopy after GEA failure, 44% had intraoperative evidence of untreated cornua and nearly one-fourth had persistent or enlarging submucous leiomyomas.
Contrary to what some believe, most endometrial ablations will not adequately destroy submucous or intramural leiomyomas. Therefore, we recommend that these fibroids be entirely removed immediately before EA.
Moreover, GEA will not always provide adequate thermal destruction to the entire endometrial cavity. The cornua regions are particularly at risk; they are difficult to reach under ideal circumstances, and especially difficult to treat in patients who have a uterine septum or a T-shaped uterus (with the ostia and cornua deeply recessed). We have also seen late-onset EA failures in patients with an extended uterine transverse diameter. The limits of GEA are greatest when a device with a fixed configuration or geometry is used.
A history of abnormal hysteroscopy or other evidence of such anatomic distortions are therefore among the reported risk factors for GEA failure (J Minim Invasive Gynecol. 2015 Mar-Apr;22[3]:323-31). A history of tubal ligation also confers risk; the procedure further increases susceptibility for failure when functioning endometrial tissue remains or regrows at the cornua, because any retrograde menstrual bleeding that occurs will be constrained by the obstructed proximal portion of the fallopian tubes.
Obesity is another risk factor for GEA failure in that the condition increases the risk of endometrial cancer, making the need for reliable biopsies in the case of spotting or other signs or symptoms even more important. On the other hand, obesity may also worsen a patient’s status as a candidate for hysterectomy.
There is much to consider with these patients. For some obese patients, GEA may be less risky than hysterectomy while for others, such as those who also have polycystic ovarian syndrome (in whom the risk for developing endometrial cancer is further increased) the scale may tip in favor of hysterectomy.
Age at the time of the primary GEA may be the single most important risk factor for GEA failure and is an important predictor of success in patient selection. Numerous investigators have shown that women younger than 35 years of age at the time of their EA had a significantly increased risk for hysterectomy, compared with women who were at least 45 years old. The younger the patient, the longer the “bridge” to menopause and the greater the likelihood that bridge will fail.
While age is not necessarily a contraindication, it is worthy of serious consideration. We generally discourage GEA for patients younger than 35. We also advise ensuring that each patient undergoing initial EA is highly self-motivated to have a uterine-sparing procedure; if not, symptoms she may experience later will likely drive her toward hysterectomy anyway.
Additionally, we caution against performing GEA in patients who have chronic pelvic pain; these patients tend to have poorer outcomes with any type of hysteroscopic surgery.
Diagnosing failed EA
Delayed complications manifest in several ways: Renewed and increasing vaginal bleeding after a period of improvement, cyclic pelvic pain (unilateral, bilateral, or suprapubic), or both bleeding and pain. Some women – likely an underreported number of them – present with postmenopausal bleeding and proceed to have unsuccessful attempts at an endometrial biopsy due to EA-associated endometrial scarring.
The cyclic pelvic pain associated with endometrial persistence or regrowth tends to worsen over time and is often described as sharp or laborlike. In our experience, a description of “laborlike” pain and a history of EA is almost fully predictive of a finding of endometrial growth. Often a hematometra can be demonstrated on transvaginal ultrasound, but this isn’t always the case.
Pain typically precedes bleeding in patients who demonstrate both. In such cases, blood from functioning endometrial tissue or other sources becomes blocked from exiting the uterine cavity by EA-induced intrauterine scarring and contracture. Painful uterine contractions then aim to expel the pooled blood. In other cases of pain – mainly those without significant vaginal bleeding – the pain is often attributed to cornual and central hematometra.
For the majority of EA failures, the diagnosis lies in the history and current symptoms. Unfortunately, the traditional methods of assessing the endometrial cavity have little merit for women presenting with delayed-onset EA complications. A sonographically assisted pelvic examination can be useful in evaluating complications, but the interpretation of ultrasounds in women with a prior EA can be challenging and is often beyond the training of most radiologists and gynecologists.
It is not uncommon for images to be incorrectly interpreted in the emergency department or physicians’ offices as “normal” and for such readings to set off a chain of CT scans, MRIs, laparoscopies, ovarian cystectomies, and other procedures that miss the root causes of pain.
Unfortunately, there is little in the literature that describes and defines ultrasound findings after EA. We do know that sonography should be timed with episodes of pain, and that the absence of a demonstrable hematometra does not exclude a diagnosis of EA failure.
Correcting late-onset failures
Our office-based operating room is fitted with side-by-side monitors that enable simultaneous sonographic and hysteroscopic views for correction of GEA failures; the rest of the set-up is similar to that of other operative hysteroscopies. However, we do employ a wide variety of resectoscopes with diameters ranging from 13 to 28 Fr. The smaller-diameter scopes are particularly useful for evaluating postmenopausal bleeding in women with a prior EA.
For those inexperienced with ultrasound-guided surgery, the initial resection is often the most challenging. The initial tissue removal is carried out on the thickest observed uterine wall – usually the posterior or anterior wall – and is done with near complete reliance on the ultrasound image. Hysteroscopic visualization is poor at this time because the outflow ports of the continuous flow resectoscope are obstructed by tissue in the narrow tubular cavity.
We then actually remove the resectoscope and clean the outflow ports of clots and debris that may have accumulated. When the scope is reinserted, there is typically sufficient room in the uterine cavity for continuous flow and excellent hysteroscopic visualization.
The sequence of resection from this point on will vary. If we’ve begun on the anterior wall, we’ll move to the posterior and then the two lateral walls to further restore the cavity. Areas of endometrial regrowth will typically be identified at this point and resected. The dissection then will extend upward, usually to within 10 mm of the fundus in the midline as measured by ultrasound. Reconfiguring the loop electrode to a 135- to 160-degree angle can be helpful in the delicate dissection that is required at the fundus.
Once all areas of endometrium have been identified and excised, we will deeply coagulate exposed myometrium with a ball-end electrode. Rarely, we will reach our maximum allowable fluid absorption limit prior to completing the case, a scenario seen in less than 1% of our patients.
In more than 330 reoperative hysteroscopic procedures, we’ve had only one uterine perforation that occurred when we switched ultrasound machines. Very likely, we were too aggressive in removing tissue at the fundus. The patient required a diagnostic laparoscopy but sustained no visceral injury.
Dr. Wortman is a clinical associate professor of obstetrics and gynecology at the University of Rochester (N.Y.) and the director of the Center for Menstrual Disorders and Reproductive Choice in Rochester. He reported having no relevant financial disclosures.
One in seven women suffer with abnormal uterine bleeding during their reproductive years, according to Fraser et al. (Exp Rev Obstet Gynecol. 2009;4:179-89). Heavy menstrual bleeding (menorrhagia) is the most common pattern. Global endometrial ablation has become a very popular surgical technique for women complaining of menorrhagia, disinterested in either medical management or definitive therapy – hysterectomy – or where medical management has failed. With proper patient selection, endometrial ablation yields an 80%-90% success rate in reducing heavy menstrual flow and is associated with a 90% patient satisfaction rate (Cochrane Database Syst Rev. 2009 Oct 7;[4]:CD001501).
Literature is replete with conditions believed to increase risk of endometrial ablation failure. This list includes untreated uterine cornua, endometrial regrowth, the presence of submucous leiomyomas or polyps, abnormal uterine cavity, enlarged uterine cavity (width and/or length), endometrial ablation in a young patient, parity of five or greater, unsuspected adhesiolysis, postablation tubal sterilization syndrome, history of dysmenorrhea, smoking, obesity, prior cesarean section, previous gynecologic surgery, and procedure length. Interestingly, type of global endometrial ablation procedure or original bleeding pattern does not influence failure rate.
In this edition of the Master Class in Gynecologic Surgery, Dr. Morris Wortman discusses not only the prevention of endometrial ablation failure, but also how to treat the problem via conservative surgical management.
Dr. Wortman is a clinical associate professor of obstetrics and gynecology at the University of Rochester (N.Y.) and is the director at the Center for Menstrual Disorders and Reproductive Choice, also in Rochester. Dr. Wortman has lectured extensively on endometrial ablation and has authored several scientific articles in peer reviewed journals.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, and past president of the AAGL and the International Society for Gynecologic Endoscopy. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/SRS fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. He reported being a subinvestigator on a study sponsored by Channel Medsystems. Email him at [email protected].
Why failures occur and how to correct them
BY MORRIS WORTMAN, MD
Since the introduction almost 20 years ago of devices for nonresectoscopic – or “global” – endometrial ablation, the procedure has been widely adopted as the treatment of choice for abnormal uterine bleeding that is refractory to medical management.
Between 400,000 and 500,000 endometrial ablations are done in the United States every year in women who have completed childbearing, and it probably won’t be long before the procedure surpasses hysterectomy in prevalence for the management of abnormal bleeding.
In recent years, the literature has begun to address the incidence of these delayed complications and the requirement for subsequent hysterectomy. A 2007 practice bulletin issued by the American College of Obstetricians and Gynecologists stated that hysterectomy rates within 4 years of endometrial ablation are at least 24% (Obstet Gynecol. 2007 May;109[5]:1233-48). And a study published the following year reported that 26% of 3,681 women undergoing EA at Kaiser Permanente facilities in Northern California required hysterectomy within 8 years (Obstet Gynecol. 2008 Dec;112[6]:1214-20).
It appears that the vast majority of what we now refer to as late-onset EA failures – complications attributable to EA that occur beyond a perioperative period of 1 month – will occur within 5 years. Some EA failures have occurred over 5-10 years, however, and in my practice we have seen late-onset complications occurring 17 or more years after the initial ablation.
In our practice, we are successfully managing delayed complications after GEA using ultrasound-guided reoperative hysteroscopy to fully explore the uterine cavity and excise areas of endometrial growth and other disease. In 2014, we published a retrospective review of 50 women whom we treated for delayed complications after a variety of GEA techniques; almost 90% avoided hysterectomy during a mean follow-up period of 18 months (J Minim Invasive Gynecol. 2014 Mar-Apr;21[2]:238-44).
Our experience since then has included reoperative surgery on more than 115 GEA failures. Additionally, we’ve managed 220 patients who have undergone various hysteroscopic and resectoscopic endometrial ablations, some of which date back to the use of the Nd:YAG laser in the late 1980s.
The fact that late-onset EA failures occur does not mean that hysterectomy should routinely be performed as a first-line treatment for intractable uterine bleeding. Overall, there is much more morbidity associated with hysterectomy than with EA.
What failures do suggest is that there are certain risk factors for late-onset EA complications. Our experience in treating women who have experienced late-onset EA failure has provided us with insight into who may be at greatest risk for late-onset EA failure and how patients can best be selected for the procedure. We’ve also learned more about the diagnosis of delayed complications.
Causes of EA failure
Untreated uterine cornua, and untreated submucous leiomyomas and endometrial polyps, are common causes of EA failure. Among the 50 women included in our retrospective review of ultrasound-guided reoperative hysteroscopy after GEA failure, 44% had intraoperative evidence of untreated cornua and nearly one-fourth had persistent or enlarging submucous leiomyomas.
Contrary to what some believe, most endometrial ablations will not adequately destroy submucous or intramural leiomyomas. Therefore, we recommend that these fibroids be entirely removed immediately before EA.
Moreover, GEA will not always provide adequate thermal destruction to the entire endometrial cavity. The cornua regions are particularly at risk; they are difficult to reach under ideal circumstances, and especially difficult to treat in patients who have a uterine septum or a T-shaped uterus (with the ostia and cornua deeply recessed). We have also seen late-onset EA failures in patients with an extended uterine transverse diameter. The limits of GEA are greatest when a device with a fixed configuration or geometry is used.
A history of abnormal hysteroscopy or other evidence of such anatomic distortions are therefore among the reported risk factors for GEA failure (J Minim Invasive Gynecol. 2015 Mar-Apr;22[3]:323-31). A history of tubal ligation also confers risk; the procedure further increases susceptibility for failure when functioning endometrial tissue remains or regrows at the cornua, because any retrograde menstrual bleeding that occurs will be constrained by the obstructed proximal portion of the fallopian tubes.
Obesity is another risk factor for GEA failure in that the condition increases the risk of endometrial cancer, making the need for reliable biopsies in the case of spotting or other signs or symptoms even more important. On the other hand, obesity may also worsen a patient’s status as a candidate for hysterectomy.
There is much to consider with these patients. For some obese patients, GEA may be less risky than hysterectomy while for others, such as those who also have polycystic ovarian syndrome (in whom the risk for developing endometrial cancer is further increased) the scale may tip in favor of hysterectomy.
Age at the time of the primary GEA may be the single most important risk factor for GEA failure and is an important predictor of success in patient selection. Numerous investigators have shown that women younger than 35 years of age at the time of their EA had a significantly increased risk for hysterectomy, compared with women who were at least 45 years old. The younger the patient, the longer the “bridge” to menopause and the greater the likelihood that bridge will fail.
While age is not necessarily a contraindication, it is worthy of serious consideration. We generally discourage GEA for patients younger than 35. We also advise ensuring that each patient undergoing initial EA is highly self-motivated to have a uterine-sparing procedure; if not, symptoms she may experience later will likely drive her toward hysterectomy anyway.
Additionally, we caution against performing GEA in patients who have chronic pelvic pain; these patients tend to have poorer outcomes with any type of hysteroscopic surgery.
Diagnosing failed EA
Delayed complications manifest in several ways: Renewed and increasing vaginal bleeding after a period of improvement, cyclic pelvic pain (unilateral, bilateral, or suprapubic), or both bleeding and pain. Some women – likely an underreported number of them – present with postmenopausal bleeding and proceed to have unsuccessful attempts at an endometrial biopsy due to EA-associated endometrial scarring.
The cyclic pelvic pain associated with endometrial persistence or regrowth tends to worsen over time and is often described as sharp or laborlike. In our experience, a description of “laborlike” pain and a history of EA is almost fully predictive of a finding of endometrial growth. Often a hematometra can be demonstrated on transvaginal ultrasound, but this isn’t always the case.
Pain typically precedes bleeding in patients who demonstrate both. In such cases, blood from functioning endometrial tissue or other sources becomes blocked from exiting the uterine cavity by EA-induced intrauterine scarring and contracture. Painful uterine contractions then aim to expel the pooled blood. In other cases of pain – mainly those without significant vaginal bleeding – the pain is often attributed to cornual and central hematometra.
For the majority of EA failures, the diagnosis lies in the history and current symptoms. Unfortunately, the traditional methods of assessing the endometrial cavity have little merit for women presenting with delayed-onset EA complications. A sonographically assisted pelvic examination can be useful in evaluating complications, but the interpretation of ultrasounds in women with a prior EA can be challenging and is often beyond the training of most radiologists and gynecologists.
It is not uncommon for images to be incorrectly interpreted in the emergency department or physicians’ offices as “normal” and for such readings to set off a chain of CT scans, MRIs, laparoscopies, ovarian cystectomies, and other procedures that miss the root causes of pain.
Unfortunately, there is little in the literature that describes and defines ultrasound findings after EA. We do know that sonography should be timed with episodes of pain, and that the absence of a demonstrable hematometra does not exclude a diagnosis of EA failure.
Correcting late-onset failures
Our office-based operating room is fitted with side-by-side monitors that enable simultaneous sonographic and hysteroscopic views for correction of GEA failures; the rest of the set-up is similar to that of other operative hysteroscopies. However, we do employ a wide variety of resectoscopes with diameters ranging from 13 to 28 Fr. The smaller-diameter scopes are particularly useful for evaluating postmenopausal bleeding in women with a prior EA.
For those inexperienced with ultrasound-guided surgery, the initial resection is often the most challenging. The initial tissue removal is carried out on the thickest observed uterine wall – usually the posterior or anterior wall – and is done with near complete reliance on the ultrasound image. Hysteroscopic visualization is poor at this time because the outflow ports of the continuous flow resectoscope are obstructed by tissue in the narrow tubular cavity.
We then actually remove the resectoscope and clean the outflow ports of clots and debris that may have accumulated. When the scope is reinserted, there is typically sufficient room in the uterine cavity for continuous flow and excellent hysteroscopic visualization.
The sequence of resection from this point on will vary. If we’ve begun on the anterior wall, we’ll move to the posterior and then the two lateral walls to further restore the cavity. Areas of endometrial regrowth will typically be identified at this point and resected. The dissection then will extend upward, usually to within 10 mm of the fundus in the midline as measured by ultrasound. Reconfiguring the loop electrode to a 135- to 160-degree angle can be helpful in the delicate dissection that is required at the fundus.
Once all areas of endometrium have been identified and excised, we will deeply coagulate exposed myometrium with a ball-end electrode. Rarely, we will reach our maximum allowable fluid absorption limit prior to completing the case, a scenario seen in less than 1% of our patients.
In more than 330 reoperative hysteroscopic procedures, we’ve had only one uterine perforation that occurred when we switched ultrasound machines. Very likely, we were too aggressive in removing tissue at the fundus. The patient required a diagnostic laparoscopy but sustained no visceral injury.
Dr. Wortman is a clinical associate professor of obstetrics and gynecology at the University of Rochester (N.Y.) and the director of the Center for Menstrual Disorders and Reproductive Choice in Rochester. He reported having no relevant financial disclosures.
Urinary incontinence
Editor’s Note: This is the first installment of a six-part series that will review key concepts and articles that ob.gyns. can use to prepare for the American Board of Obstetrics and Gynecology Maintenance of Certification examination. The series is adapted from Ob/Gyn Board Master (obgynboardmaster.com), an online board review course created by Erudyte Inc. The series will cover key topics across the specialty, as well as general test-taking and study tips. This month’s edition of the Board Exam Corner focuses on urinary incontinence.
The literature
The American College of Obstetricians and Gynecologists and the American Urogynecologic Society recently published an updated Practice Bulletin regarding the diagnosis and management of urinary incontinence. Practice Bulletin No. 155 was also a maintenance of certification (MOC) assignment for 2016. The contents of this article are critical as urinary incontinence is a prevalent and treatable condition, and it will likely be part of this year’s board examination. This month’s Board Corner will review the key elements of the updated guidelines.
Let’s begin with a possible medical board question: An 80-year old patient has urinary leakage as a result of severe arthritis and poor vision, and is unable to reach the bathroom in time. What is the diagnosis?
A. Mixed urinary incontinence
B. Extraurethral incontinence
C. Functional incontinence
D. Postural incontinence
E. Ectopic ureter
The answer is C.
Functional incontinence occurs because of cognitive or mobility impairments in the presence of an intact lower urinary tract. The patient in the vignette has “restricted mobility” and a physical impairment that leads to urinary incontinence.
A is incorrect because mixed incontinence is an involuntary loss of urine associated with urgency and with physical exertion.
B and E are incorrect because extraurethral incontinence is urine leakage through channels other than the urethral meatus (e.g., vesicovaginal fistula or ectopic ureter).
D is incorrect because postural incontinence is the involuntary loss of urine associated with change in body positions (e.g., from sitting to standing).
The key points to remember are:
• Urinary incontinence is a prevalent condition that is treatable despite what many women believe.
• There are several types of incontinence but the main types are stress, urgency, and mixed incontinence.
• Basic office evaluation includes urinalysis and postvoid residual, but not urodynamics or cystoscopy.
• Treatment of stress incontinence includes weight loss, fluid management, pelvic floor muscle exercises (with or without assistance from a physical therapist), and suburethral sling or bulking agent injections.
• Treatment of urgency urinary incontinence includes bladder training, fluid management, decreasing caffeine, antimuscarinic and beta-3 agonists, and third-line treatments (botulinum toxin A injection, sacral neuromodulation, or tibial nerve stimulation).
• The mnemonic DIAPPERS is useful in remembering all of the reversible, nongenitourinary (i.e., functional) causes of incontinence. 
Test-taking tip of the month
If you finish the test early, take the time to go back to any question for which you made an educated guess. Reread the question carefully. Don’t change your answer unless there is a very good reason for doing so because your first impression is usually the right one. Don’t change an answer just because you have a gut feeling. However, there are good reasons to change your answers when you go back. For instance, consider changing your answer if rereading the question helped you to better understand what is being asked, if you reconsidered and conceptualized a better answer, if you remembered a new piece of information, or if you learned something from a later question that will help you better answer the earlier question.
Reference
Obstet Gynecol. 2015 Nov;126(5): e66-81.
Dr. Sam Siddighi is editor-in-chief of the Ob/Gyn Board Master and director of female pelvic medicine and reconstructive surgery and director of grand rounds at Loma Linda University Health in California. Ob.Gyn. News and Ob/Gyn Board Master are owned by the same parent company, Frontline Medical Communications.
Editor’s Note: This is the first installment of a six-part series that will review key concepts and articles that ob.gyns. can use to prepare for the American Board of Obstetrics and Gynecology Maintenance of Certification examination. The series is adapted from Ob/Gyn Board Master (obgynboardmaster.com), an online board review course created by Erudyte Inc. The series will cover key topics across the specialty, as well as general test-taking and study tips. This month’s edition of the Board Exam Corner focuses on urinary incontinence.
The literature
The American College of Obstetricians and Gynecologists and the American Urogynecologic Society recently published an updated Practice Bulletin regarding the diagnosis and management of urinary incontinence. Practice Bulletin No. 155 was also a maintenance of certification (MOC) assignment for 2016. The contents of this article are critical as urinary incontinence is a prevalent and treatable condition, and it will likely be part of this year’s board examination. This month’s Board Corner will review the key elements of the updated guidelines.
Let’s begin with a possible medical board question: An 80-year old patient has urinary leakage as a result of severe arthritis and poor vision, and is unable to reach the bathroom in time. What is the diagnosis?
A. Mixed urinary incontinence
B. Extraurethral incontinence
C. Functional incontinence
D. Postural incontinence
E. Ectopic ureter
The answer is C.
Functional incontinence occurs because of cognitive or mobility impairments in the presence of an intact lower urinary tract. The patient in the vignette has “restricted mobility” and a physical impairment that leads to urinary incontinence.
A is incorrect because mixed incontinence is an involuntary loss of urine associated with urgency and with physical exertion.
B and E are incorrect because extraurethral incontinence is urine leakage through channels other than the urethral meatus (e.g., vesicovaginal fistula or ectopic ureter).
D is incorrect because postural incontinence is the involuntary loss of urine associated with change in body positions (e.g., from sitting to standing).
The key points to remember are:
• Urinary incontinence is a prevalent condition that is treatable despite what many women believe.
• There are several types of incontinence but the main types are stress, urgency, and mixed incontinence.
• Basic office evaluation includes urinalysis and postvoid residual, but not urodynamics or cystoscopy.
• Treatment of stress incontinence includes weight loss, fluid management, pelvic floor muscle exercises (with or without assistance from a physical therapist), and suburethral sling or bulking agent injections.
• Treatment of urgency urinary incontinence includes bladder training, fluid management, decreasing caffeine, antimuscarinic and beta-3 agonists, and third-line treatments (botulinum toxin A injection, sacral neuromodulation, or tibial nerve stimulation).
• The mnemonic DIAPPERS is useful in remembering all of the reversible, nongenitourinary (i.e., functional) causes of incontinence. 
Test-taking tip of the month
If you finish the test early, take the time to go back to any question for which you made an educated guess. Reread the question carefully. Don’t change your answer unless there is a very good reason for doing so because your first impression is usually the right one. Don’t change an answer just because you have a gut feeling. However, there are good reasons to change your answers when you go back. For instance, consider changing your answer if rereading the question helped you to better understand what is being asked, if you reconsidered and conceptualized a better answer, if you remembered a new piece of information, or if you learned something from a later question that will help you better answer the earlier question.
Reference
Obstet Gynecol. 2015 Nov;126(5): e66-81.
Dr. Sam Siddighi is editor-in-chief of the Ob/Gyn Board Master and director of female pelvic medicine and reconstructive surgery and director of grand rounds at Loma Linda University Health in California. Ob.Gyn. News and Ob/Gyn Board Master are owned by the same parent company, Frontline Medical Communications.
Editor’s Note: This is the first installment of a six-part series that will review key concepts and articles that ob.gyns. can use to prepare for the American Board of Obstetrics and Gynecology Maintenance of Certification examination. The series is adapted from Ob/Gyn Board Master (obgynboardmaster.com), an online board review course created by Erudyte Inc. The series will cover key topics across the specialty, as well as general test-taking and study tips. This month’s edition of the Board Exam Corner focuses on urinary incontinence.
The literature
The American College of Obstetricians and Gynecologists and the American Urogynecologic Society recently published an updated Practice Bulletin regarding the diagnosis and management of urinary incontinence. Practice Bulletin No. 155 was also a maintenance of certification (MOC) assignment for 2016. The contents of this article are critical as urinary incontinence is a prevalent and treatable condition, and it will likely be part of this year’s board examination. This month’s Board Corner will review the key elements of the updated guidelines.
Let’s begin with a possible medical board question: An 80-year old patient has urinary leakage as a result of severe arthritis and poor vision, and is unable to reach the bathroom in time. What is the diagnosis?
A. Mixed urinary incontinence
B. Extraurethral incontinence
C. Functional incontinence
D. Postural incontinence
E. Ectopic ureter
The answer is C.
Functional incontinence occurs because of cognitive or mobility impairments in the presence of an intact lower urinary tract. The patient in the vignette has “restricted mobility” and a physical impairment that leads to urinary incontinence.
A is incorrect because mixed incontinence is an involuntary loss of urine associated with urgency and with physical exertion.
B and E are incorrect because extraurethral incontinence is urine leakage through channels other than the urethral meatus (e.g., vesicovaginal fistula or ectopic ureter).
D is incorrect because postural incontinence is the involuntary loss of urine associated with change in body positions (e.g., from sitting to standing).
The key points to remember are:
• Urinary incontinence is a prevalent condition that is treatable despite what many women believe.
• There are several types of incontinence but the main types are stress, urgency, and mixed incontinence.
• Basic office evaluation includes urinalysis and postvoid residual, but not urodynamics or cystoscopy.
• Treatment of stress incontinence includes weight loss, fluid management, pelvic floor muscle exercises (with or without assistance from a physical therapist), and suburethral sling or bulking agent injections.
• Treatment of urgency urinary incontinence includes bladder training, fluid management, decreasing caffeine, antimuscarinic and beta-3 agonists, and third-line treatments (botulinum toxin A injection, sacral neuromodulation, or tibial nerve stimulation).
• The mnemonic DIAPPERS is useful in remembering all of the reversible, nongenitourinary (i.e., functional) causes of incontinence. 
Test-taking tip of the month
If you finish the test early, take the time to go back to any question for which you made an educated guess. Reread the question carefully. Don’t change your answer unless there is a very good reason for doing so because your first impression is usually the right one. Don’t change an answer just because you have a gut feeling. However, there are good reasons to change your answers when you go back. For instance, consider changing your answer if rereading the question helped you to better understand what is being asked, if you reconsidered and conceptualized a better answer, if you remembered a new piece of information, or if you learned something from a later question that will help you better answer the earlier question.
Reference
Obstet Gynecol. 2015 Nov;126(5): e66-81.
Dr. Sam Siddighi is editor-in-chief of the Ob/Gyn Board Master and director of female pelvic medicine and reconstructive surgery and director of grand rounds at Loma Linda University Health in California. Ob.Gyn. News and Ob/Gyn Board Master are owned by the same parent company, Frontline Medical Communications.
VIDEO: New antisense inhibitor nets impressive reductions in lipids
NEW ORLEANS – A new antisense inhibitor to angiopoietinlike protein 3 (ANGPTL3) reduces lipids in healthy adults with elevated triglyceride levels, according to results of a phase I/IIa ascending-dose trial reported at the American Heart Association scientific sessions.
ANGPTL3 regulates lipid and possibly general metabolism through actions in the liver, gut, muscle, and adipose tissue, explained presenting author Sotirios Tsimikas, MD, an investigator with Ionis Pharmaceuticals, Carlsbad, Calif., and a professor of medicine and director of vascular medicine at the University of California, San Diego. Individuals having loss-of-function mutations in the gene encoding this protein have very low plasma levels of cholesterol and triglycerides.
In the trial, sponsored by Ionis Pharmaceuticals, 32 healthy volunteers with elevated triglyceride levels were treated with various doses of the antisense inhibitor, called IONIS-ANGPTL3-LRx, or a placebo, given by weekly subcutaneous injections for 6 weeks.
Results showed that participants treated at the higher-dose levels had a reduction from baseline of 66% in triglycerides, 68% in apoliprotein C-III (ApoC-III), 35% in LDL cholesterol, 36% in total cholesterol, and 40% in non-HDL cholesterol, as well as 25% in HDL cholesterol, Dr. Tsimikas reported. They also had reductions in apolipoprotein B (ApoB).
“All these lipid parameters are really going in the right direction in terms of postulating potential clinical benefit,” he commented.
Safety and tolerability results showed that only a single participant experienced a local injection site adverse event related to the inhibitor (erythema and pruritus). None experienced flulike symptoms, platelet reductions, or serious adverse events, and none left the study because of adverse events.
“Among all known therapies that lower triglycerides, this [IONIS-ANGPTL3-LRx] is also associated with a reduction not only in LDL cholesterol levels, but also in ApoB as well, which portends well for future outcomes trials,” noted Dr. Tsimikas.
“We think this is going to be a promising candidate for patients who have uncontrolled LDL cholesterol, elevated triglycerides, and possibly patients who have hepatic steatosis and NASH [nonalcoholic steatohepatitis],” he concluded.
Dr. Tsimikas, who is an employee of and shareholder in the trial’s sponsor, Ionis Pharmaceuticals, discussed his findings in a video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The trial shows a very clear proof of concept in regards to the effects on lipids.
In contrast to agents that increase LDL receptor levels (statins, proprotein convertase subtilisin/kexin type 9 [PCSK9] inhibitors, bile acid–binding resins, and ezetimibe), IONIS-ANGPTL3-LRx produces greater reductions in non-HDL cholesterol than in LDL cholesterol, suggesting different mechanisms are at work.
But the key issue at this point is to ascertain the longer-term safety of IONIS-ANGPTL3-LRx in the liver and other organs.
One of the most impressive aspects of the combination of biotechnology and genetics is how rapidly you can come to a proof of concept in humans, and this is unprecedented, compared with what we have seen over the last 20 or 30 years. Unfortunately, the determination of safety is not quite as rapid, and it takes large numbers and long duration of follow-up. Obviously, we need that in terms of understanding benefits and risks.
Christie M. Ballantyne, MD, director of the Center for Cardiovascular Disease Prevention at the Methodist DeBakey Heart Center, Baylor College of Medicine in Houston, made these comments as the invited discussant. He has been a consultant to and received research support from trial sponsor Ionis Pharmaceuticals.
The trial shows a very clear proof of concept in regards to the effects on lipids.
In contrast to agents that increase LDL receptor levels (statins, proprotein convertase subtilisin/kexin type 9 [PCSK9] inhibitors, bile acid–binding resins, and ezetimibe), IONIS-ANGPTL3-LRx produces greater reductions in non-HDL cholesterol than in LDL cholesterol, suggesting different mechanisms are at work.
But the key issue at this point is to ascertain the longer-term safety of IONIS-ANGPTL3-LRx in the liver and other organs.
One of the most impressive aspects of the combination of biotechnology and genetics is how rapidly you can come to a proof of concept in humans, and this is unprecedented, compared with what we have seen over the last 20 or 30 years. Unfortunately, the determination of safety is not quite as rapid, and it takes large numbers and long duration of follow-up. Obviously, we need that in terms of understanding benefits and risks.
Christie M. Ballantyne, MD, director of the Center for Cardiovascular Disease Prevention at the Methodist DeBakey Heart Center, Baylor College of Medicine in Houston, made these comments as the invited discussant. He has been a consultant to and received research support from trial sponsor Ionis Pharmaceuticals.
The trial shows a very clear proof of concept in regards to the effects on lipids.
In contrast to agents that increase LDL receptor levels (statins, proprotein convertase subtilisin/kexin type 9 [PCSK9] inhibitors, bile acid–binding resins, and ezetimibe), IONIS-ANGPTL3-LRx produces greater reductions in non-HDL cholesterol than in LDL cholesterol, suggesting different mechanisms are at work.
But the key issue at this point is to ascertain the longer-term safety of IONIS-ANGPTL3-LRx in the liver and other organs.
One of the most impressive aspects of the combination of biotechnology and genetics is how rapidly you can come to a proof of concept in humans, and this is unprecedented, compared with what we have seen over the last 20 or 30 years. Unfortunately, the determination of safety is not quite as rapid, and it takes large numbers and long duration of follow-up. Obviously, we need that in terms of understanding benefits and risks.
Christie M. Ballantyne, MD, director of the Center for Cardiovascular Disease Prevention at the Methodist DeBakey Heart Center, Baylor College of Medicine in Houston, made these comments as the invited discussant. He has been a consultant to and received research support from trial sponsor Ionis Pharmaceuticals.
NEW ORLEANS – A new antisense inhibitor to angiopoietinlike protein 3 (ANGPTL3) reduces lipids in healthy adults with elevated triglyceride levels, according to results of a phase I/IIa ascending-dose trial reported at the American Heart Association scientific sessions.
ANGPTL3 regulates lipid and possibly general metabolism through actions in the liver, gut, muscle, and adipose tissue, explained presenting author Sotirios Tsimikas, MD, an investigator with Ionis Pharmaceuticals, Carlsbad, Calif., and a professor of medicine and director of vascular medicine at the University of California, San Diego. Individuals having loss-of-function mutations in the gene encoding this protein have very low plasma levels of cholesterol and triglycerides.
In the trial, sponsored by Ionis Pharmaceuticals, 32 healthy volunteers with elevated triglyceride levels were treated with various doses of the antisense inhibitor, called IONIS-ANGPTL3-LRx, or a placebo, given by weekly subcutaneous injections for 6 weeks.
Results showed that participants treated at the higher-dose levels had a reduction from baseline of 66% in triglycerides, 68% in apoliprotein C-III (ApoC-III), 35% in LDL cholesterol, 36% in total cholesterol, and 40% in non-HDL cholesterol, as well as 25% in HDL cholesterol, Dr. Tsimikas reported. They also had reductions in apolipoprotein B (ApoB).
“All these lipid parameters are really going in the right direction in terms of postulating potential clinical benefit,” he commented.
Safety and tolerability results showed that only a single participant experienced a local injection site adverse event related to the inhibitor (erythema and pruritus). None experienced flulike symptoms, platelet reductions, or serious adverse events, and none left the study because of adverse events.
“Among all known therapies that lower triglycerides, this [IONIS-ANGPTL3-LRx] is also associated with a reduction not only in LDL cholesterol levels, but also in ApoB as well, which portends well for future outcomes trials,” noted Dr. Tsimikas.
“We think this is going to be a promising candidate for patients who have uncontrolled LDL cholesterol, elevated triglycerides, and possibly patients who have hepatic steatosis and NASH [nonalcoholic steatohepatitis],” he concluded.
Dr. Tsimikas, who is an employee of and shareholder in the trial’s sponsor, Ionis Pharmaceuticals, discussed his findings in a video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – A new antisense inhibitor to angiopoietinlike protein 3 (ANGPTL3) reduces lipids in healthy adults with elevated triglyceride levels, according to results of a phase I/IIa ascending-dose trial reported at the American Heart Association scientific sessions.
ANGPTL3 regulates lipid and possibly general metabolism through actions in the liver, gut, muscle, and adipose tissue, explained presenting author Sotirios Tsimikas, MD, an investigator with Ionis Pharmaceuticals, Carlsbad, Calif., and a professor of medicine and director of vascular medicine at the University of California, San Diego. Individuals having loss-of-function mutations in the gene encoding this protein have very low plasma levels of cholesterol and triglycerides.
In the trial, sponsored by Ionis Pharmaceuticals, 32 healthy volunteers with elevated triglyceride levels were treated with various doses of the antisense inhibitor, called IONIS-ANGPTL3-LRx, or a placebo, given by weekly subcutaneous injections for 6 weeks.
Results showed that participants treated at the higher-dose levels had a reduction from baseline of 66% in triglycerides, 68% in apoliprotein C-III (ApoC-III), 35% in LDL cholesterol, 36% in total cholesterol, and 40% in non-HDL cholesterol, as well as 25% in HDL cholesterol, Dr. Tsimikas reported. They also had reductions in apolipoprotein B (ApoB).
“All these lipid parameters are really going in the right direction in terms of postulating potential clinical benefit,” he commented.
Safety and tolerability results showed that only a single participant experienced a local injection site adverse event related to the inhibitor (erythema and pruritus). None experienced flulike symptoms, platelet reductions, or serious adverse events, and none left the study because of adverse events.
“Among all known therapies that lower triglycerides, this [IONIS-ANGPTL3-LRx] is also associated with a reduction not only in LDL cholesterol levels, but also in ApoB as well, which portends well for future outcomes trials,” noted Dr. Tsimikas.
“We think this is going to be a promising candidate for patients who have uncontrolled LDL cholesterol, elevated triglycerides, and possibly patients who have hepatic steatosis and NASH [nonalcoholic steatohepatitis],” he concluded.
Dr. Tsimikas, who is an employee of and shareholder in the trial’s sponsor, Ionis Pharmaceuticals, discussed his findings in a video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: Treatment was associated with reductions in levels of triglycerides (–66%), ApoC-III (–68%), LDL cholesterol (–35%), total cholesterol (–36%), and non-HDL cholesterol (–40%), as well as levels of HDL cholesterol (–25%).
Data source: A phase I/IIa ascending-dose trial among 32 healthy adults with hypertriglyceridemia.
Disclosures: Dr. Tsimikas is an employee of and shareholder in Ionis Pharmaceuticals, which sponsored the trial.
Addressing Sexual Health With Patients
Why start this new column on mental health with an article that focuses on sexual health? Surely that is the domain of family practice, urology, gynecology, endocrinology, or some other discipline. While it is true that all these disciplines and many others are central to the diagnosis and treatment of sexual health, mental health providers need to be an integral part of the conversations.
I have a problem with arbitrarily separating mental health and behavioral health from physical health. Mental health is directly affected by physical health and vice versa. If a woman has a urinary tract infection, she is not feeling mentally or sexually healthy. If a man has erectile dysfunction (ED), he seldom is at the top of his game emotionally. For our war-wounded who lack limbs or who have genitourinary injuries, optimal sexual functioning can be a challenge.
I am probably preaching to the choir here, so I will not belabor the point. However, I will develop this point by using lessons learned from combat-injured service members, the psychiatric adverse effects (AEs) of commonly used psychiatric medications, and most important, asking about sexual health as part of taking a mental health history.
The War-Wounded
Since 9/11, 2.7 million U.S. service members have served in wars. Much discussion has been focused on posttraumatic stress disorder (PTSD) and traumatic brain injury (TBI), the invisible wounds of war. Probably about 25% of service members who have been deployed have PTSD, and about 300,000 service members have TBI.1 About 50,000 have other physical injuries. These injuries directly affect intimate relationships.
The signature weapon of the wars in Iraq and Afghanistan was the bomb, or improvised explosive device. Motor vehicle accidents and gunshot wounds also have added to the myriad of injuries. Physical war wounds involve sexual functioning. These include, but are not limited to, lower extremity amputations, genitourinary injuries, and facial disfigurement or burns. All of these may involve multiple surgeries, pain, and disability and can significantly impact self-esteem.
One of the many lessons I have learned in putting together my most recent book, Intimacy Post Injury: Combat Trauma and Sexual Health, is that there is a void in the recent general medical and popular literature about optimizing sexual health in those with injuries or diseases.2 The majority of wounded warriors are men, but injuries happen to women service members as well. Of course, sexual assault causes severe harm to intimate relationships as well.
At this time, most injured personnel are still in the Military Health System (MHS) or have transitioned to the VHA. However, many service members also will be treated in the civilian health system.
Psychiatric Medications and Sexual Adverse Effects
The treatment of PTSD, depression, and other psychiatric conditions often involves medications that have sexual AEs. Sexual AEs usually refer to problems with erectile function (impotence), difficulties with ejaculation, lack of orgasm or desire, or lack of lubrication for women. Selective serotonin reuptake inhibitors (SSRIs) have a very high incidence of sexual AEs, ranging from 30% to 70%.3 When I speak with clinicians about their anecdotal experience, the percentage always is high, usually more than 50%. Other psychiatric medications, such as antipsychotics, older antidepressants, and stimulants also have sexual AEs, and narcotics are notorious for their sexual AEs.
Fortunately, many solutions exist. For SSRIs, the solutions include lowering the dose of the offending agent, switching to another agent, drug holidays, or adding other medications, such as bupropion or cyproheptadine. Family practice and other physicians are very familiar with phosphodiesterase inhibitors, such as sildenafil, tadalafil, and vardenafil. Although these medications have AEs and are very expensive, they work well for impotence.
Other solutions are nonpharmacologic: Setting aside time for intimacy can be crucial. Gels and creams can help with lubrication. Communication with providers and between partners and families is the most important ingredient.
Asking About Sexual Health
I encourage all medical personnel who treat active-duty service members or veterans to (1) discuss sexual health with their patients; (2) learn the basics of how to evaluate, treat, or refer ED, including SSRI AEs; and (3) understand how to discuss the effects of physical injury, pain, and disability on sexual functioning.
The conversation should touch on sexual activity, satisfaction with intimacy, exposure to sexually transmitted diseases, and if appropriate, previous sexual abuse. The appropriate time and place for a conversation about sexual health depends on the setting. In the outpatient setting, I bring up the subject after I ask about sleep and appetite and before I ask about suicidal and homicidal thoughts; others may choose elsewhere in the patient history. However, asking about sexual issues may or may not be appropriate in an emergency department situation.
Providers often are uncomfortable with asking about sexual issues, perhaps more so if they are young and female and the patient is older and male. Therefore, I encourage expanded training in medical school and throughout residency.
Sexual Difficulties and Suicide
In the military, the suicide rate has been rising from about 10 per 100,000 per year in 2004 to about 20 per 20,000 in this decade.4,5 According to the VA Office of Suicide Prevention, about 20 veterans die by suicide daily.6 One question that has received little attention is the relationship between sexual difficulties and suicide. Although there has been an important focus on causes of suicide in the military and veterans, little is known about the important issue of how many service members die by suicide because of impotence.
We do know a lot about the big picture as to why service members die by suicide. In about two-thirds of the completed suicides, there were relationship issues. In addition, there are often legal, occupational, and financial difficulties. About two-thirds of service members die by suicide using firearms. Jumping and strangulation are other common methods.4,5
But there is much we do not know. What percentage of relationship difficulties are related to sexual dysfunction? Is ED the straw that breaks the camel’s back and leads to the shot to the chest? Other subjects outside the scope of this column (but included in Intimacy Post Injury) include sexual therapy, fertility, adaptations for those with disabilities, reproductive AEs of toxin exposure, and surgeries that include penile transplantation.
My hypothesis is that sexual problems, specifically ED or impotence, contribute to feelings of failure and inadequacy and thus to suicidal or homicidal thoughts.
Conclusion
Health care providers do not always talk to patients about their sexual health and may barely mention the sexual AEs of psychiatric or other medications. In whatever setting you practice, you should not neglect asking questions about sexual health, as it is a critical issue for many of our patients and should be for us.
1. Committee on the Assessment of Ongoing Efforts in the Treatment of Posttraumatic Stress Disorder, Board of Health of Selected Populations. Treatment of Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment. Washington, DC: National Academies Press; 2014.
2. Ritchie EC, ed. Intimacy Post-Injury: Combat Trauma and Sexual Health. New York: Oxford University Press; 2016.
3. Higgins A, Nash M, Lynch AM. Antidepressant-associated sexual dysfunction: impact, effects and treatment. Drug Healthc Patient Saf. 2010;2:141-150.
4. Black SA, Gallaway S, Bell MR, Ritchie EC. Prevalence and risk factors associated with suicides of army soldiers 2001-2009. Mil Psychol. 2011;23:433-451.
5. Ritchie EC. Suicides and the United States army: perspectives from the former psychiatry consultant to the army surgeon general. Cerebrum. 2012;2012:1.
6. U.S. Department of Veterans Affairs. Suicide among veterans and other Americans 2001-2014. http://www.mentalhealth.va.gov/docs/2016suicidedatareport.pdf. Published August 3, 2016. Accessed December 27, 2016.
Why start this new column on mental health with an article that focuses on sexual health? Surely that is the domain of family practice, urology, gynecology, endocrinology, or some other discipline. While it is true that all these disciplines and many others are central to the diagnosis and treatment of sexual health, mental health providers need to be an integral part of the conversations.
I have a problem with arbitrarily separating mental health and behavioral health from physical health. Mental health is directly affected by physical health and vice versa. If a woman has a urinary tract infection, she is not feeling mentally or sexually healthy. If a man has erectile dysfunction (ED), he seldom is at the top of his game emotionally. For our war-wounded who lack limbs or who have genitourinary injuries, optimal sexual functioning can be a challenge.
I am probably preaching to the choir here, so I will not belabor the point. However, I will develop this point by using lessons learned from combat-injured service members, the psychiatric adverse effects (AEs) of commonly used psychiatric medications, and most important, asking about sexual health as part of taking a mental health history.
The War-Wounded
Since 9/11, 2.7 million U.S. service members have served in wars. Much discussion has been focused on posttraumatic stress disorder (PTSD) and traumatic brain injury (TBI), the invisible wounds of war. Probably about 25% of service members who have been deployed have PTSD, and about 300,000 service members have TBI.1 About 50,000 have other physical injuries. These injuries directly affect intimate relationships.
The signature weapon of the wars in Iraq and Afghanistan was the bomb, or improvised explosive device. Motor vehicle accidents and gunshot wounds also have added to the myriad of injuries. Physical war wounds involve sexual functioning. These include, but are not limited to, lower extremity amputations, genitourinary injuries, and facial disfigurement or burns. All of these may involve multiple surgeries, pain, and disability and can significantly impact self-esteem.
One of the many lessons I have learned in putting together my most recent book, Intimacy Post Injury: Combat Trauma and Sexual Health, is that there is a void in the recent general medical and popular literature about optimizing sexual health in those with injuries or diseases.2 The majority of wounded warriors are men, but injuries happen to women service members as well. Of course, sexual assault causes severe harm to intimate relationships as well.
At this time, most injured personnel are still in the Military Health System (MHS) or have transitioned to the VHA. However, many service members also will be treated in the civilian health system.
Psychiatric Medications and Sexual Adverse Effects
The treatment of PTSD, depression, and other psychiatric conditions often involves medications that have sexual AEs. Sexual AEs usually refer to problems with erectile function (impotence), difficulties with ejaculation, lack of orgasm or desire, or lack of lubrication for women. Selective serotonin reuptake inhibitors (SSRIs) have a very high incidence of sexual AEs, ranging from 30% to 70%.3 When I speak with clinicians about their anecdotal experience, the percentage always is high, usually more than 50%. Other psychiatric medications, such as antipsychotics, older antidepressants, and stimulants also have sexual AEs, and narcotics are notorious for their sexual AEs.
Fortunately, many solutions exist. For SSRIs, the solutions include lowering the dose of the offending agent, switching to another agent, drug holidays, or adding other medications, such as bupropion or cyproheptadine. Family practice and other physicians are very familiar with phosphodiesterase inhibitors, such as sildenafil, tadalafil, and vardenafil. Although these medications have AEs and are very expensive, they work well for impotence.
Other solutions are nonpharmacologic: Setting aside time for intimacy can be crucial. Gels and creams can help with lubrication. Communication with providers and between partners and families is the most important ingredient.
Asking About Sexual Health
I encourage all medical personnel who treat active-duty service members or veterans to (1) discuss sexual health with their patients; (2) learn the basics of how to evaluate, treat, or refer ED, including SSRI AEs; and (3) understand how to discuss the effects of physical injury, pain, and disability on sexual functioning.
The conversation should touch on sexual activity, satisfaction with intimacy, exposure to sexually transmitted diseases, and if appropriate, previous sexual abuse. The appropriate time and place for a conversation about sexual health depends on the setting. In the outpatient setting, I bring up the subject after I ask about sleep and appetite and before I ask about suicidal and homicidal thoughts; others may choose elsewhere in the patient history. However, asking about sexual issues may or may not be appropriate in an emergency department situation.
Providers often are uncomfortable with asking about sexual issues, perhaps more so if they are young and female and the patient is older and male. Therefore, I encourage expanded training in medical school and throughout residency.
Sexual Difficulties and Suicide
In the military, the suicide rate has been rising from about 10 per 100,000 per year in 2004 to about 20 per 20,000 in this decade.4,5 According to the VA Office of Suicide Prevention, about 20 veterans die by suicide daily.6 One question that has received little attention is the relationship between sexual difficulties and suicide. Although there has been an important focus on causes of suicide in the military and veterans, little is known about the important issue of how many service members die by suicide because of impotence.
We do know a lot about the big picture as to why service members die by suicide. In about two-thirds of the completed suicides, there were relationship issues. In addition, there are often legal, occupational, and financial difficulties. About two-thirds of service members die by suicide using firearms. Jumping and strangulation are other common methods.4,5
But there is much we do not know. What percentage of relationship difficulties are related to sexual dysfunction? Is ED the straw that breaks the camel’s back and leads to the shot to the chest? Other subjects outside the scope of this column (but included in Intimacy Post Injury) include sexual therapy, fertility, adaptations for those with disabilities, reproductive AEs of toxin exposure, and surgeries that include penile transplantation.
My hypothesis is that sexual problems, specifically ED or impotence, contribute to feelings of failure and inadequacy and thus to suicidal or homicidal thoughts.
Conclusion
Health care providers do not always talk to patients about their sexual health and may barely mention the sexual AEs of psychiatric or other medications. In whatever setting you practice, you should not neglect asking questions about sexual health, as it is a critical issue for many of our patients and should be for us.
Why start this new column on mental health with an article that focuses on sexual health? Surely that is the domain of family practice, urology, gynecology, endocrinology, or some other discipline. While it is true that all these disciplines and many others are central to the diagnosis and treatment of sexual health, mental health providers need to be an integral part of the conversations.
I have a problem with arbitrarily separating mental health and behavioral health from physical health. Mental health is directly affected by physical health and vice versa. If a woman has a urinary tract infection, she is not feeling mentally or sexually healthy. If a man has erectile dysfunction (ED), he seldom is at the top of his game emotionally. For our war-wounded who lack limbs or who have genitourinary injuries, optimal sexual functioning can be a challenge.
I am probably preaching to the choir here, so I will not belabor the point. However, I will develop this point by using lessons learned from combat-injured service members, the psychiatric adverse effects (AEs) of commonly used psychiatric medications, and most important, asking about sexual health as part of taking a mental health history.
The War-Wounded
Since 9/11, 2.7 million U.S. service members have served in wars. Much discussion has been focused on posttraumatic stress disorder (PTSD) and traumatic brain injury (TBI), the invisible wounds of war. Probably about 25% of service members who have been deployed have PTSD, and about 300,000 service members have TBI.1 About 50,000 have other physical injuries. These injuries directly affect intimate relationships.
The signature weapon of the wars in Iraq and Afghanistan was the bomb, or improvised explosive device. Motor vehicle accidents and gunshot wounds also have added to the myriad of injuries. Physical war wounds involve sexual functioning. These include, but are not limited to, lower extremity amputations, genitourinary injuries, and facial disfigurement or burns. All of these may involve multiple surgeries, pain, and disability and can significantly impact self-esteem.
One of the many lessons I have learned in putting together my most recent book, Intimacy Post Injury: Combat Trauma and Sexual Health, is that there is a void in the recent general medical and popular literature about optimizing sexual health in those with injuries or diseases.2 The majority of wounded warriors are men, but injuries happen to women service members as well. Of course, sexual assault causes severe harm to intimate relationships as well.
At this time, most injured personnel are still in the Military Health System (MHS) or have transitioned to the VHA. However, many service members also will be treated in the civilian health system.
Psychiatric Medications and Sexual Adverse Effects
The treatment of PTSD, depression, and other psychiatric conditions often involves medications that have sexual AEs. Sexual AEs usually refer to problems with erectile function (impotence), difficulties with ejaculation, lack of orgasm or desire, or lack of lubrication for women. Selective serotonin reuptake inhibitors (SSRIs) have a very high incidence of sexual AEs, ranging from 30% to 70%.3 When I speak with clinicians about their anecdotal experience, the percentage always is high, usually more than 50%. Other psychiatric medications, such as antipsychotics, older antidepressants, and stimulants also have sexual AEs, and narcotics are notorious for their sexual AEs.
Fortunately, many solutions exist. For SSRIs, the solutions include lowering the dose of the offending agent, switching to another agent, drug holidays, or adding other medications, such as bupropion or cyproheptadine. Family practice and other physicians are very familiar with phosphodiesterase inhibitors, such as sildenafil, tadalafil, and vardenafil. Although these medications have AEs and are very expensive, they work well for impotence.
Other solutions are nonpharmacologic: Setting aside time for intimacy can be crucial. Gels and creams can help with lubrication. Communication with providers and between partners and families is the most important ingredient.
Asking About Sexual Health
I encourage all medical personnel who treat active-duty service members or veterans to (1) discuss sexual health with their patients; (2) learn the basics of how to evaluate, treat, or refer ED, including SSRI AEs; and (3) understand how to discuss the effects of physical injury, pain, and disability on sexual functioning.
The conversation should touch on sexual activity, satisfaction with intimacy, exposure to sexually transmitted diseases, and if appropriate, previous sexual abuse. The appropriate time and place for a conversation about sexual health depends on the setting. In the outpatient setting, I bring up the subject after I ask about sleep and appetite and before I ask about suicidal and homicidal thoughts; others may choose elsewhere in the patient history. However, asking about sexual issues may or may not be appropriate in an emergency department situation.
Providers often are uncomfortable with asking about sexual issues, perhaps more so if they are young and female and the patient is older and male. Therefore, I encourage expanded training in medical school and throughout residency.
Sexual Difficulties and Suicide
In the military, the suicide rate has been rising from about 10 per 100,000 per year in 2004 to about 20 per 20,000 in this decade.4,5 According to the VA Office of Suicide Prevention, about 20 veterans die by suicide daily.6 One question that has received little attention is the relationship between sexual difficulties and suicide. Although there has been an important focus on causes of suicide in the military and veterans, little is known about the important issue of how many service members die by suicide because of impotence.
We do know a lot about the big picture as to why service members die by suicide. In about two-thirds of the completed suicides, there were relationship issues. In addition, there are often legal, occupational, and financial difficulties. About two-thirds of service members die by suicide using firearms. Jumping and strangulation are other common methods.4,5
But there is much we do not know. What percentage of relationship difficulties are related to sexual dysfunction? Is ED the straw that breaks the camel’s back and leads to the shot to the chest? Other subjects outside the scope of this column (but included in Intimacy Post Injury) include sexual therapy, fertility, adaptations for those with disabilities, reproductive AEs of toxin exposure, and surgeries that include penile transplantation.
My hypothesis is that sexual problems, specifically ED or impotence, contribute to feelings of failure and inadequacy and thus to suicidal or homicidal thoughts.
Conclusion
Health care providers do not always talk to patients about their sexual health and may barely mention the sexual AEs of psychiatric or other medications. In whatever setting you practice, you should not neglect asking questions about sexual health, as it is a critical issue for many of our patients and should be for us.
1. Committee on the Assessment of Ongoing Efforts in the Treatment of Posttraumatic Stress Disorder, Board of Health of Selected Populations. Treatment of Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment. Washington, DC: National Academies Press; 2014.
2. Ritchie EC, ed. Intimacy Post-Injury: Combat Trauma and Sexual Health. New York: Oxford University Press; 2016.
3. Higgins A, Nash M, Lynch AM. Antidepressant-associated sexual dysfunction: impact, effects and treatment. Drug Healthc Patient Saf. 2010;2:141-150.
4. Black SA, Gallaway S, Bell MR, Ritchie EC. Prevalence and risk factors associated with suicides of army soldiers 2001-2009. Mil Psychol. 2011;23:433-451.
5. Ritchie EC. Suicides and the United States army: perspectives from the former psychiatry consultant to the army surgeon general. Cerebrum. 2012;2012:1.
6. U.S. Department of Veterans Affairs. Suicide among veterans and other Americans 2001-2014. http://www.mentalhealth.va.gov/docs/2016suicidedatareport.pdf. Published August 3, 2016. Accessed December 27, 2016.
1. Committee on the Assessment of Ongoing Efforts in the Treatment of Posttraumatic Stress Disorder, Board of Health of Selected Populations. Treatment of Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment. Washington, DC: National Academies Press; 2014.
2. Ritchie EC, ed. Intimacy Post-Injury: Combat Trauma and Sexual Health. New York: Oxford University Press; 2016.
3. Higgins A, Nash M, Lynch AM. Antidepressant-associated sexual dysfunction: impact, effects and treatment. Drug Healthc Patient Saf. 2010;2:141-150.
4. Black SA, Gallaway S, Bell MR, Ritchie EC. Prevalence and risk factors associated with suicides of army soldiers 2001-2009. Mil Psychol. 2011;23:433-451.
5. Ritchie EC. Suicides and the United States army: perspectives from the former psychiatry consultant to the army surgeon general. Cerebrum. 2012;2012:1.
6. U.S. Department of Veterans Affairs. Suicide among veterans and other Americans 2001-2014. http://www.mentalhealth.va.gov/docs/2016suicidedatareport.pdf. Published August 3, 2016. Accessed December 27, 2016.
Yoga may improve QOL in kids with cancer

Photo by Bill Branson
A yoga program for children with cancer can be carried out during cancer treatment and has quality of life (QOL) benefits for the children as well as their caregivers, according to research published in Rehabilitation Oncology.
However, the program was not feasible for all patients. More than half of those initially enrolled could not complete the study due to treatment toxicity or scheduling conflicts.
Andrea Orsey, MD, of Connecticut Children’s Medical Center in Hartford, and her colleagues conducted this research to evaluate the feasibility and effectiveness of a yoga intervention for children with cancer and their families.
The team began by conducting a survey of 20 children and adolescents with cancer and their parents/guardians.
Survey respondents expressed interest in a yoga program. But they also perceived several barriers to such a program, including concerns about side effects, pain/discomfort, and physical limitations.
With these barriers in mind, Dr Orsey and her colleagues developed a yoga intervention for pediatric cancer patients, delivered by certified yoga instructors.
The program was designed to be performed in a variety of settings and tailored to the children’s physical condition or mobility issues.
A pilot evaluation included 10 children with cancer and their caregivers. Twenty-two patient/caregiver pairs were actually enrolled, but 6 pairs withdrew because of treatment toxicity, and 6 had the study window lapse due to scheduling conflicts.
Although limited by its small size, the study suggested that yoga improved health-related QOL for both caregivers and children.
The children had significant improvements in both social and emotional QOL. They had an overall improvement in fatigue, but this was not statistically significant.
Caregivers had a significant improvement in mental health but not physical health or caregiver burden.
Both caregivers and children said they were satisfied with the yoga program and would recommend it to others.
Dr Orsey and her colleagues hope this pilot study will help guide future efforts to provide yoga to children with cancer and their families.
The researchers noted that a key issue will be coordinating yoga sessions with the medical demands of chemotherapy. ![]()

Photo by Bill Branson
A yoga program for children with cancer can be carried out during cancer treatment and has quality of life (QOL) benefits for the children as well as their caregivers, according to research published in Rehabilitation Oncology.
However, the program was not feasible for all patients. More than half of those initially enrolled could not complete the study due to treatment toxicity or scheduling conflicts.
Andrea Orsey, MD, of Connecticut Children’s Medical Center in Hartford, and her colleagues conducted this research to evaluate the feasibility and effectiveness of a yoga intervention for children with cancer and their families.
The team began by conducting a survey of 20 children and adolescents with cancer and their parents/guardians.
Survey respondents expressed interest in a yoga program. But they also perceived several barriers to such a program, including concerns about side effects, pain/discomfort, and physical limitations.
With these barriers in mind, Dr Orsey and her colleagues developed a yoga intervention for pediatric cancer patients, delivered by certified yoga instructors.
The program was designed to be performed in a variety of settings and tailored to the children’s physical condition or mobility issues.
A pilot evaluation included 10 children with cancer and their caregivers. Twenty-two patient/caregiver pairs were actually enrolled, but 6 pairs withdrew because of treatment toxicity, and 6 had the study window lapse due to scheduling conflicts.
Although limited by its small size, the study suggested that yoga improved health-related QOL for both caregivers and children.
The children had significant improvements in both social and emotional QOL. They had an overall improvement in fatigue, but this was not statistically significant.
Caregivers had a significant improvement in mental health but not physical health or caregiver burden.
Both caregivers and children said they were satisfied with the yoga program and would recommend it to others.
Dr Orsey and her colleagues hope this pilot study will help guide future efforts to provide yoga to children with cancer and their families.
The researchers noted that a key issue will be coordinating yoga sessions with the medical demands of chemotherapy. ![]()

Photo by Bill Branson
A yoga program for children with cancer can be carried out during cancer treatment and has quality of life (QOL) benefits for the children as well as their caregivers, according to research published in Rehabilitation Oncology.
However, the program was not feasible for all patients. More than half of those initially enrolled could not complete the study due to treatment toxicity or scheduling conflicts.
Andrea Orsey, MD, of Connecticut Children’s Medical Center in Hartford, and her colleagues conducted this research to evaluate the feasibility and effectiveness of a yoga intervention for children with cancer and their families.
The team began by conducting a survey of 20 children and adolescents with cancer and their parents/guardians.
Survey respondents expressed interest in a yoga program. But they also perceived several barriers to such a program, including concerns about side effects, pain/discomfort, and physical limitations.
With these barriers in mind, Dr Orsey and her colleagues developed a yoga intervention for pediatric cancer patients, delivered by certified yoga instructors.
The program was designed to be performed in a variety of settings and tailored to the children’s physical condition or mobility issues.
A pilot evaluation included 10 children with cancer and their caregivers. Twenty-two patient/caregiver pairs were actually enrolled, but 6 pairs withdrew because of treatment toxicity, and 6 had the study window lapse due to scheduling conflicts.
Although limited by its small size, the study suggested that yoga improved health-related QOL for both caregivers and children.
The children had significant improvements in both social and emotional QOL. They had an overall improvement in fatigue, but this was not statistically significant.
Caregivers had a significant improvement in mental health but not physical health or caregiver burden.
Both caregivers and children said they were satisfied with the yoga program and would recommend it to others.
Dr Orsey and her colleagues hope this pilot study will help guide future efforts to provide yoga to children with cancer and their families.
The researchers noted that a key issue will be coordinating yoga sessions with the medical demands of chemotherapy. ![]()
US cancer cases may near 1.7 million in 2017

patient and her father
Photo by Rhoda Baer
The US may see nearly 1.7 million new cancer cases in 2017 and more than 600,000 cancer-related deaths, according to a report from the American Cancer Society (ACS).
In addition to estimates for 2017, the report, “Cancer Statistics 2017,” includes the most recent data on cancer incidence, mortality, and survival in the US.
The report was published in CA: A Cancer Journal for Clinicians.
The report projects there will be 1,688,780 new cancer cases and 600,920 cancer deaths in the US this year.
This includes:
- 80,500 new cases of lymphoma and 21,210 lymphoma deaths
- 62,130 new cases of leukemia and 24,500 leukemia deaths
- 30,280 new cases of myeloma and 12,590 myeloma deaths.
The report also shows that, from 2004 to 2013, the overall cancer incidence rate was stable in women and declined by about 2% per year in men. From 2005 to 2014, the cancer death rate declined by about 1.5% annually in both men and women.
Overall, the cancer death rate dropped 25% from its peak of 215.1 (per 100,000 population) in 1991 to 161.2 (per 100,000 population) in 2014, the latest year for which data was available. This translates to about 2,143,200 fewer cancer deaths.
“The continuing drops in the cancer death rate are a powerful sign of the potential we have to reduce cancer’s deadly toll,” said Otis W. Brawley, MD, chief medical officer of the ACS.
He said the decrease in cancer death rates is the result of steady reductions in smoking and advances in early detection and treatment. The decrease is driven by decreasing death rates for the 4 major cancer sites—lung, breast, colorectal, and prostate.
The report also shows that racial disparities in cancer death rates continue to decline. The excess risk of cancer death in black men has dropped from 47% in 1990 to 21% in 2014. The black/white disparity declined similarly in women, from a peak of 20% in 1998 to 13% in 2014.
On the other hand, significant gender disparities persist for both cancer incidence and death in the US. For all cancer sites combined, the incidence rate is 20% higher in men than in women, and the cancer death rate is 40% higher in men.
Dr Brawley said the gender gap in cancer mortality largely reflects variation in the distribution of cancers that occur in men and women, much of which is due to differences in the prevalence of cancer risk factors.
The yearly “Cancer Statistics” reports have been published by ACS researchers since 1967 to inform and guide clinicians, investigators, and others in public health in prioritizing efforts to reduce the burden of cancer.
Cancer incidence data for the current report were collected by the Surveillance, Epidemiology, and End Results Program; the National Program of Cancer Registries; and the North American Association of Central Cancer Registries. Mortality data were collected by the National Center for Health Statistics. ![]()

patient and her father
Photo by Rhoda Baer
The US may see nearly 1.7 million new cancer cases in 2017 and more than 600,000 cancer-related deaths, according to a report from the American Cancer Society (ACS).
In addition to estimates for 2017, the report, “Cancer Statistics 2017,” includes the most recent data on cancer incidence, mortality, and survival in the US.
The report was published in CA: A Cancer Journal for Clinicians.
The report projects there will be 1,688,780 new cancer cases and 600,920 cancer deaths in the US this year.
This includes:
- 80,500 new cases of lymphoma and 21,210 lymphoma deaths
- 62,130 new cases of leukemia and 24,500 leukemia deaths
- 30,280 new cases of myeloma and 12,590 myeloma deaths.
The report also shows that, from 2004 to 2013, the overall cancer incidence rate was stable in women and declined by about 2% per year in men. From 2005 to 2014, the cancer death rate declined by about 1.5% annually in both men and women.
Overall, the cancer death rate dropped 25% from its peak of 215.1 (per 100,000 population) in 1991 to 161.2 (per 100,000 population) in 2014, the latest year for which data was available. This translates to about 2,143,200 fewer cancer deaths.
“The continuing drops in the cancer death rate are a powerful sign of the potential we have to reduce cancer’s deadly toll,” said Otis W. Brawley, MD, chief medical officer of the ACS.
He said the decrease in cancer death rates is the result of steady reductions in smoking and advances in early detection and treatment. The decrease is driven by decreasing death rates for the 4 major cancer sites—lung, breast, colorectal, and prostate.
The report also shows that racial disparities in cancer death rates continue to decline. The excess risk of cancer death in black men has dropped from 47% in 1990 to 21% in 2014. The black/white disparity declined similarly in women, from a peak of 20% in 1998 to 13% in 2014.
On the other hand, significant gender disparities persist for both cancer incidence and death in the US. For all cancer sites combined, the incidence rate is 20% higher in men than in women, and the cancer death rate is 40% higher in men.
Dr Brawley said the gender gap in cancer mortality largely reflects variation in the distribution of cancers that occur in men and women, much of which is due to differences in the prevalence of cancer risk factors.
The yearly “Cancer Statistics” reports have been published by ACS researchers since 1967 to inform and guide clinicians, investigators, and others in public health in prioritizing efforts to reduce the burden of cancer.
Cancer incidence data for the current report were collected by the Surveillance, Epidemiology, and End Results Program; the National Program of Cancer Registries; and the North American Association of Central Cancer Registries. Mortality data were collected by the National Center for Health Statistics. ![]()

patient and her father
Photo by Rhoda Baer
The US may see nearly 1.7 million new cancer cases in 2017 and more than 600,000 cancer-related deaths, according to a report from the American Cancer Society (ACS).
In addition to estimates for 2017, the report, “Cancer Statistics 2017,” includes the most recent data on cancer incidence, mortality, and survival in the US.
The report was published in CA: A Cancer Journal for Clinicians.
The report projects there will be 1,688,780 new cancer cases and 600,920 cancer deaths in the US this year.
This includes:
- 80,500 new cases of lymphoma and 21,210 lymphoma deaths
- 62,130 new cases of leukemia and 24,500 leukemia deaths
- 30,280 new cases of myeloma and 12,590 myeloma deaths.
The report also shows that, from 2004 to 2013, the overall cancer incidence rate was stable in women and declined by about 2% per year in men. From 2005 to 2014, the cancer death rate declined by about 1.5% annually in both men and women.
Overall, the cancer death rate dropped 25% from its peak of 215.1 (per 100,000 population) in 1991 to 161.2 (per 100,000 population) in 2014, the latest year for which data was available. This translates to about 2,143,200 fewer cancer deaths.
“The continuing drops in the cancer death rate are a powerful sign of the potential we have to reduce cancer’s deadly toll,” said Otis W. Brawley, MD, chief medical officer of the ACS.
He said the decrease in cancer death rates is the result of steady reductions in smoking and advances in early detection and treatment. The decrease is driven by decreasing death rates for the 4 major cancer sites—lung, breast, colorectal, and prostate.
The report also shows that racial disparities in cancer death rates continue to decline. The excess risk of cancer death in black men has dropped from 47% in 1990 to 21% in 2014. The black/white disparity declined similarly in women, from a peak of 20% in 1998 to 13% in 2014.
On the other hand, significant gender disparities persist for both cancer incidence and death in the US. For all cancer sites combined, the incidence rate is 20% higher in men than in women, and the cancer death rate is 40% higher in men.
Dr Brawley said the gender gap in cancer mortality largely reflects variation in the distribution of cancers that occur in men and women, much of which is due to differences in the prevalence of cancer risk factors.
The yearly “Cancer Statistics” reports have been published by ACS researchers since 1967 to inform and guide clinicians, investigators, and others in public health in prioritizing efforts to reduce the burden of cancer.
Cancer incidence data for the current report were collected by the Surveillance, Epidemiology, and End Results Program; the National Program of Cancer Registries; and the North American Association of Central Cancer Registries. Mortality data were collected by the National Center for Health Statistics. ![]()
Cancer genomic data released to public

Photo courtesy of the
National Institute of
General Medical Sciences
The American Association for Cancer Research (AACR) has announced the first public release of cancer genomic data aggregated through the AACR Project Genomics Evidence Neoplasia Information Exchange (GENIE).
The data set includes nearly 19,000 de-identified genomic records collected from patients who were treated at 8 international institutions, making it one of the largest public cancer genomic data sets released to date.
The release includes data for 59 major cancer types, including leukemias, lymphomas, and multiple myeloma.
The genomic data and a limited amount of linked clinical data for each patient can be accessed via the AACR Project GENIE cBioPortal or from Sage Bionetworks. (Users must create an account for either site to access the data.)
“We are excited to make publicly available this very large set of clinical-grade, next-generation sequencing data obtained during routine patient care,” said Charles L. Sawyers, MD, AACR Project GENIE Steering Committee chairperson.
“These data were generated as part of routine patient care and, without AACR Project GENIE, they would likely never have been shared with the global cancer research community.”
AACR Project GENIE is a multi-phase, international data-sharing project aimed at catalyzing precision oncology through the development of a registry that aggregates and links clinical-grade cancer genomic data with clinical outcomes from tens of thousands of cancer patients treated at multiple institutions.
The newly released data are fully de-identified in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
The data are derived from patients whose tumors were genetically sequenced as part of their care at any of the 8 institutions that participated in the first phase of AACR Project GENIE.
The goal of releasing these data to the cancer research community is to aid new research that will accelerate the pace of progress against cancer.
According to AACR, the data can be used to validate gene signatures of drug response or prognosis, identify new patient populations for drugs that are currently available, and uncover new drug targets and biomarkers.
“I am extremely proud that the American Association for Cancer Research, as the coordinating center for AACR Project GENIE, is delivering on its promise to make these important data publicly available just over a year after unveiling the initiative,” said Margaret Foti, PhD, MD, chief executive officer of the AACR.
To expand the AACR Project GENIE registry, the consortium is accepting applications for new participating centers. Any nonprofit institution that meets certain criteria can submit an application to become a project participant.
For more information on AACR Project GENIE, visit the project website or send an email to [email protected]. ![]()

Photo courtesy of the
National Institute of
General Medical Sciences
The American Association for Cancer Research (AACR) has announced the first public release of cancer genomic data aggregated through the AACR Project Genomics Evidence Neoplasia Information Exchange (GENIE).
The data set includes nearly 19,000 de-identified genomic records collected from patients who were treated at 8 international institutions, making it one of the largest public cancer genomic data sets released to date.
The release includes data for 59 major cancer types, including leukemias, lymphomas, and multiple myeloma.
The genomic data and a limited amount of linked clinical data for each patient can be accessed via the AACR Project GENIE cBioPortal or from Sage Bionetworks. (Users must create an account for either site to access the data.)
“We are excited to make publicly available this very large set of clinical-grade, next-generation sequencing data obtained during routine patient care,” said Charles L. Sawyers, MD, AACR Project GENIE Steering Committee chairperson.
“These data were generated as part of routine patient care and, without AACR Project GENIE, they would likely never have been shared with the global cancer research community.”
AACR Project GENIE is a multi-phase, international data-sharing project aimed at catalyzing precision oncology through the development of a registry that aggregates and links clinical-grade cancer genomic data with clinical outcomes from tens of thousands of cancer patients treated at multiple institutions.
The newly released data are fully de-identified in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
The data are derived from patients whose tumors were genetically sequenced as part of their care at any of the 8 institutions that participated in the first phase of AACR Project GENIE.
The goal of releasing these data to the cancer research community is to aid new research that will accelerate the pace of progress against cancer.
According to AACR, the data can be used to validate gene signatures of drug response or prognosis, identify new patient populations for drugs that are currently available, and uncover new drug targets and biomarkers.
“I am extremely proud that the American Association for Cancer Research, as the coordinating center for AACR Project GENIE, is delivering on its promise to make these important data publicly available just over a year after unveiling the initiative,” said Margaret Foti, PhD, MD, chief executive officer of the AACR.
To expand the AACR Project GENIE registry, the consortium is accepting applications for new participating centers. Any nonprofit institution that meets certain criteria can submit an application to become a project participant.
For more information on AACR Project GENIE, visit the project website or send an email to [email protected]. ![]()

Photo courtesy of the
National Institute of
General Medical Sciences
The American Association for Cancer Research (AACR) has announced the first public release of cancer genomic data aggregated through the AACR Project Genomics Evidence Neoplasia Information Exchange (GENIE).
The data set includes nearly 19,000 de-identified genomic records collected from patients who were treated at 8 international institutions, making it one of the largest public cancer genomic data sets released to date.
The release includes data for 59 major cancer types, including leukemias, lymphomas, and multiple myeloma.
The genomic data and a limited amount of linked clinical data for each patient can be accessed via the AACR Project GENIE cBioPortal or from Sage Bionetworks. (Users must create an account for either site to access the data.)
“We are excited to make publicly available this very large set of clinical-grade, next-generation sequencing data obtained during routine patient care,” said Charles L. Sawyers, MD, AACR Project GENIE Steering Committee chairperson.
“These data were generated as part of routine patient care and, without AACR Project GENIE, they would likely never have been shared with the global cancer research community.”
AACR Project GENIE is a multi-phase, international data-sharing project aimed at catalyzing precision oncology through the development of a registry that aggregates and links clinical-grade cancer genomic data with clinical outcomes from tens of thousands of cancer patients treated at multiple institutions.
The newly released data are fully de-identified in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
The data are derived from patients whose tumors were genetically sequenced as part of their care at any of the 8 institutions that participated in the first phase of AACR Project GENIE.
The goal of releasing these data to the cancer research community is to aid new research that will accelerate the pace of progress against cancer.
According to AACR, the data can be used to validate gene signatures of drug response or prognosis, identify new patient populations for drugs that are currently available, and uncover new drug targets and biomarkers.
“I am extremely proud that the American Association for Cancer Research, as the coordinating center for AACR Project GENIE, is delivering on its promise to make these important data publicly available just over a year after unveiling the initiative,” said Margaret Foti, PhD, MD, chief executive officer of the AACR.
To expand the AACR Project GENIE registry, the consortium is accepting applications for new participating centers. Any nonprofit institution that meets certain criteria can submit an application to become a project participant.
For more information on AACR Project GENIE, visit the project website or send an email to [email protected]. ![]()