User login
Analysis of Hospitalist Discontinuity
Studies examining the importance of continuity of care have shown that patients who maintain a continuous relationship with a single physician have improved outcomes.[1, 2] However, most of these studies were performed in the outpatient, rather than the inpatient setting. With over 35 million patients admitted to hospitals in 2013, along with the significant increase in hospital discontinuity over recent years, the impact of inpatient continuity of care on quality outcomes and patient satisfaction is becoming increasingly relevant.[3, 4]
Service handoffs, when a physician hands over treatment responsibility for a panel of patients and is not expected to return, are a type of handoff that contributes to inpatient discontinuity. In particular, service handoffs between hospitalists are an especially common and inherently risky type of transition, as there is a severing of an established relationship during a patient's hospitalization. Unfortunately, due to the lack of evidence on the effects of service handoffs, current guidelines are limited in their recommendations.[5] Whereas several recent studies have begun to explore the effects of these handoffs, no prior study has examined this issue from a patient's perspective.[6, 7, 8]
Patients are uniquely positioned to inform us about their experiences in care transitions. Furthermore, with patient satisfaction now affecting Medicare reimbursement rates, patient experiences while in the hospital are becoming even more significant.[9] Despite this emphasis toward more patient‐centered care, no study has explored the hospitalized patient's experience with hospitalist service handoffs. Our goal was to qualitatively assess the hospitalized patients' experiences with transitions between hospitalists to develop a conceptual model to inform future work on improving inpatient transitions of care.
METHODS
Sampling and Recruitment
We conducted bedside interviews of hospitalized patients at an urban academic medical center from October 2014 through December 2014. The hospitalist service consists of a physician and an advanced nurse practitioner (ANP) who divide a panel of patients that consist of general medicine and subspecialty patients who are often comanaged with hepatology, oncology, and nephrology subspecialists. We performed a purposive selection of patients who could potentially comment on their experience with a hospitalist service transition using the following method: 48 hours after a service handoff (ie, an outgoing physician completing 1 week on service, then transfers the care of the patient to a new oncoming hospitalist), oncoming hospitalists were approached and asked if any patient on their service had experienced a service handoff and still remained in the hospital. A 48‐hour time period was chosen to give the patients time to familiarize themselves with their new hospitalist, allowing them to properly comment on the handoff. Patients who were managed by the ANP, who were non‐English speaking, or who were deemed to have an altered mental status based on clinical suspicion by the interviewing physician (C.M.W.) were excluded from participation. Following each weekly service transition, a list of patients who met the above criteria was collected from 4 nonteaching hospitalist services, and were approached by the primary investigator (C.M.W.) and asked if they would be willing to participate. All patients were general medicine patients and no exclusions were made based on physical location within the hospital. Those who agreed provided signed written consent prior to participation to allow access to the electronic health records (EHRs) by study personnel.
Data Collection
Patients were administered a 9‐question, semistructured interview that was informed by expert opinion and existing literature, which was developed to elicit their perspective regarding their transition between hospitalists.[10, 11] No formal changes were made to the interview guide during the study period, and all patients were asked the same questions. Outcomes from interim analysis guided further questioning in subsequent interviews so as to increase the depth of patient responses (ie, Can you explain your response in greater depth?). Prior to the interview, patients were read a description of a hospitalist, and were reminded which hospitalists had cared for them during their stay (see Supporting Information, Appendix 1, in the online version of this article). If family members or a caregiver were present at the time of interview, they were asked not to comment. No repeat interviews were carried out.
All interviews were performed privately in single‐occupancy rooms, digitally recorded using an iPad (Apple, Cupertino, CA) and professionally transcribed verbatim (Rev, San Francisco, CA). All analysis was performed using MAXQDA Software (VERBI Software GmbH, Berlin, Germany). We obtained demographic information about each patient through chart review
Data Analysis
Grounded theory was utilized, with an inductive approach with no a priori hypothesis.[12] The constant comparative method was used to generate emerging and reoccurring themes.[13] Units of analysis were sentences and phrases. Our research team consisted of 4 academic hospitalists, 2 with backgrounds in clinical medicine, medical education, and qualitative analysis (J.M.F., V.M.A.), 1 as a clinician (C.M.W.), and 1 in health economics (D.O.M.). Interim analysis was performed on a weekly basis (C.M.W.), during which time a coding template was created and refined through an iterative process (C.M.W., J.M.F.). All disagreements in coded themes were resolved through group discussion until full consensus was reached. Each week, responses were assessed for thematic saturation.[14] Interviews were continued if new themes arose during this analysis. Data collection was ended once we ceased to extract new topics from participants. A summary of all themes was then presented to a group of 10 patients who met the same inclusion criteria for respondent validation and member checking. All reporting was performed within the Standards for Reporting Qualitative Research, with additional guidance derived from the Consolidated Criteria for Reporting Qualitative Research.[15, 16] The University of Chicago Institutional Review Board approved this protocol.
RESULTS
In total, 43 eligible patients were recruited, and 40 (93%) agreed to participate. Interviewed patients were between 51 and 65 (39%) years old, had a mean age of 54.5 (15) years, were predominantly female (65%), African American (58%), had a median length of stay at the time of interview of 6.5 days (interquartile range [IQR]: 48), and had an average of 2.0 (IQR: 13) hospitalists oversee their care at the time of interview (Table 1). Interview times ranged from 10:25 to 25:48 minutes, with an average of 15:32 minutes.
| Value | |
|---|---|
| |
| Response rate, n (%) | 40/43 (93) |
| Age, mean SD | 54.5 15 |
| Sex, n (%) | |
| Female | 26 (65) |
| Male | 14 (35) |
| Race, n (%) | |
| African American | 23 (58) |
| White | 16 (40) |
| Hispanic | 1 (2) |
| Median LOS at time of interview, d (IQR) | 6.5 (48) |
| Median no. of hospitalists at time of interview, n (IQR) | 2.0 (13) |
We identified 6 major themes on patient perceptions of hospitalist service handoffs including (1) physician‐patient communication, (2) transparency in the hospitalist transition process, (3) indifference toward the hospitalist transition, (4) hospitalist‐subspecialist communication, (5) recognition of new opportunities due to a transition, and (6) hospitalists' bedside manner (Table 2).
| Themes | Subthemes | Representative Quotes |
|---|---|---|
| Physician‐patient communication | Patients dislike redundant communication with oncoming hospitalist. | I mean it's just you always have to explain your situation over and over and over again. (patient 14) |
| When I said it once already, then you're repeating it to another doctor. I feel as if that hospitalist didn't talk to the other hospitalist. (patient 7) | ||
| Poor communication can negatively affect the doctor‐patient relationship. | They don't really want to explain things. They don't think I'll understand. I think & yeah, I'm okay. You don't even have to put it in layman's terms. I know medical. I'm in nursing school. I have a year left. But even if you didn't know that, I would still hope you would try to tell me what was going on instead of just doing it in your head, and treating it. (patient 2) | |
| I mean it's just you always have to explain your situation over and over and over again. After a while you just stop trusting them. (patient 20) | ||
| Good communication can positively affect the doctor‐patient relationship. | Just continue with the communication, the open communication, and always stress to me that I have a voice and just going out of their way to do whatever they can to help me through whatever I'm going through. (patient 1) | |
| Transparency in transition | Patients want to be informed prior to a service changeover. | I think they should be told immediately, even maybe given prior notice, like this may happen, just so you're not surprised when it happens. (patient 15) |
| When the doctor approached me, he let me know that he wasn't going to be here the next day and there was going to be another doctor coming in. That made me feel comfortable. (patient 9) | ||
| Patients desire a more formalized process in the service changeover. | People want things to be consistent. People don't like change. They like routine. So, if he's leaving, you're coming on, I'd like for him to bring you in, introduce you to me, and for you just assure me that I'll take care of you. (patient 4) | |
| Just like when you get a new medication, you're given all this information on it. So when you get a new hospitalist, shouldn't I get all the information on them? Like where they went to school, what they look like. (patient 23) | ||
| Patients want clearer definition of the roles the physicians will play in their care. | The first time I was hospitalized for the first time I had all these different doctors coming in, and I had the residency, and the specialists, and the department, and I don't know who's who. What I asked them to do is when they come in the room, which they did, but introduce it a little more for me. Write it down like these are the special team and these are the doctors because even though they come in and give me their name, I have no idea what they're doing. (patient 5) | |
| Someone should explain the setup and who people are. Someone would say, Okay when you're in a hospital this is your [doctor's] role. Like they should have booklets and everything. (patient 19) | ||
| Indifference toward transition | Many patients have trust in service changeovers. | [S]o as long as everybody's on board and communicates well and efficiently, I don't have a problem with it. (patient 6) |
| To me, it really wasn't no preference, as long as I was getting the care that I needed. (patient 21) | ||
| It's not a concern as long as they're on the same page. (patient 17) | ||
| Hospitalist‐specialist communication | Patients are concerned about communication between their hospitalist and their subspecialists. | The more cooks you get in the kitchen, the more things get to get lost, so I'm always concerned that they're not sharing the same information, especially when you're getting asked the same questions that you might have just answered the last hour ago. (patient 9) |
| I don't know if the hospitalist are talking to them [subspecialist]. They haven't got time. (patient 35) | ||
| Patients place trust in the communication between hospitalist and subspecialist. | I think among the teams themselveswhich is my pain doctor, Dr. K's group, the oncology group itself, they switch off and trade with each other and they all speak the same language so that works out good. (patient 3) | |
| Lack of interprofessional communication can lead to patient concern. | I was afraid that one was going to drop the ball on something and not pass something on, or you know. (patient 11) | |
| I had numerous doctors who all seemed to not communicate with each other at all or did so by email or whatever. They didn't just sit down together and say we feel this way and we feel that way. I didn't like that at all. (patient 10) | ||
| New opportunities due to transition | Patients see new doctor as opportunity for medical reevaluation. | I see it as two heads are better than one, three heads are better than one, four heads are better than one. When people put their heads together to work towards a common goal, especially when they're, you know, people working their craft, it can't be bad. (patient 9) |
| I finally got my ears looked atbecause I've asked to have my ears looked at since Mondayand the new doc is trying to make an effort to look at them. (patient 39) | ||
| Patients see service changeover as an opportunity to form a better personal relationship. | Having a new hospitalist it gives you opportunity for a new beginning. (patient 11) | |
| Bedside manner | Good bedside manner can assist in a service changeover. | Some of them are all business‐like but some of them are, Well how do you feel today? Hi, how are you? So this made a little difference. You feel more comfortable. You're going to be more comfortable with them. Their bedside manner helps. (patient 16) |
| It's just like when a doctor sits down and talks to you, they just seem more relaxed and more .... I know they're very busy and they have lots of things to do and other patients to see, but while they're in there with you, you know, you don't get too much time with them. So bedside manner is just so important. (patient 24) | ||
| Poor bedside manner can be detrimental in transition. | [B]ecause they be so busy they claim they don't have time just to sit and talk to a patient, and make sure they all right. (patient 17) |
Physician‐Patient Communication
Communication between the physician and the patient was an important element in patients' assessment of their experience. Patient's tended to divide physician‐patient communication into 2 categories: good communication, which consisted of open communication (patient 1) and patient engagement, and bad communication, which was described as physicians not sharing information or taking the time to explain the course of care in words that I'll understand (patient 2). Patients also described dissatisfaction with redundant communication between multiple hospitalists and the frustration of often having to describe their clinical course to multiple providers.
Transparency in Communication
The desire to have greater transparency in the handoff process was another common theme. This was likely due to the fact that 34/40 (85%) of surveyed patients were unaware that a service changeover had ever taken place. This lack of transparency was viewed to have further downstream consequences as patients stated that there should be a level of transparency, and when it's not, then there is always trust issues (patient 1). Upon further questioning as to how to make the process more transparent, many patients recommended a formalized, face‐to‐face introduction involving the patient and both hospitalists, in which the outgoing hospitalist would, bring you [oncoming hospitalist] in, and introduce you to me (patient 4).
Patients often stated that given the large spectrum of physicians they might encounter during their stay (ie, medical student, resident, hospitalist attending, subspecialty fellow, subspecialist attending), clearer definitions of physicians' roles are needed.
Hospitalist‐Specialist Communication
Concern about the communication between their hospitalist and subspecialist was another predominant theme. Conflicting and unclear directions from multiple services were especially frustrating, as a patient stated, One guy took me off this pill, the other guy wants me on that pill, I'm like okay, I can't do both (patient 8). Furthermore, a subset of patients referenced their subspecialist as their primary care provider and preferred their subspecialist for guidance in their hospital course, rather than their hospitalist. This often appeared in cases where the patient had an established relationship with the subspecialist prior to their hospitalization.
New Opportunities Due to Transition
Patients expressed positive feelings toward service handoffs by viewing the transition as an opportunity for medical reevaluation by a new physician. Patients told of instances in which a specific complaint was not being addressed by the first physician, but would be addressed by the second (oncoming) physician. A commonly expressed idea was that the oncoming physician might know something that he [Dr. B] didn't know, and since Dr. B was only here for a week, why not give him [oncoming hospitalist] a chance (patient 10). Patients would also describe the transition as an opportunity to form, and possibly improve, therapeutic alliances with a new hospitalist.
Bedside Manner
Bedside manner was another commonly mentioned thematic element. Patients were often quick to forget prior problems or issues that they may have suffered because of the transition if the oncoming physician was perceived to have a good bedside manner, often described as someone who formally introduced themselves, was considered relaxed, and would take the time to sit and talk with the patient. As a patient put it, [S]he sat down and got to know meand asked me what I wanted to do (patient 12). Conversely, patients described instances in which a perceived bad bedside manner led to a poor relationship between the physician and the patient, in which trust and comfort (patient 11) were sacrificed.
Indifference Toward Transition
In contrast to some of the previous findings, which called for improved interactions between physicians and patients, we also discovered a theme of indifference toward the transition. Several patients stated feelings of trust with the medical system, and were content with the service changeover as long as they felt that their medical needs were being met. Patients also tended to express a level of acceptance with the transition, and tended to believe that this was the price we pay for being here [in the hospital] (patient 7).
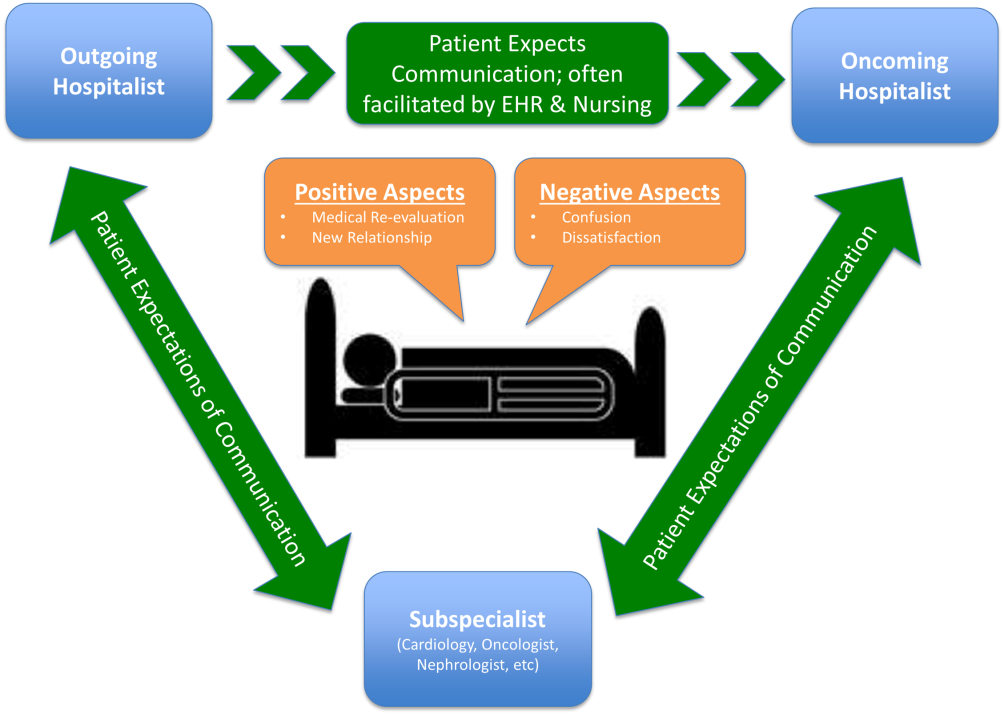
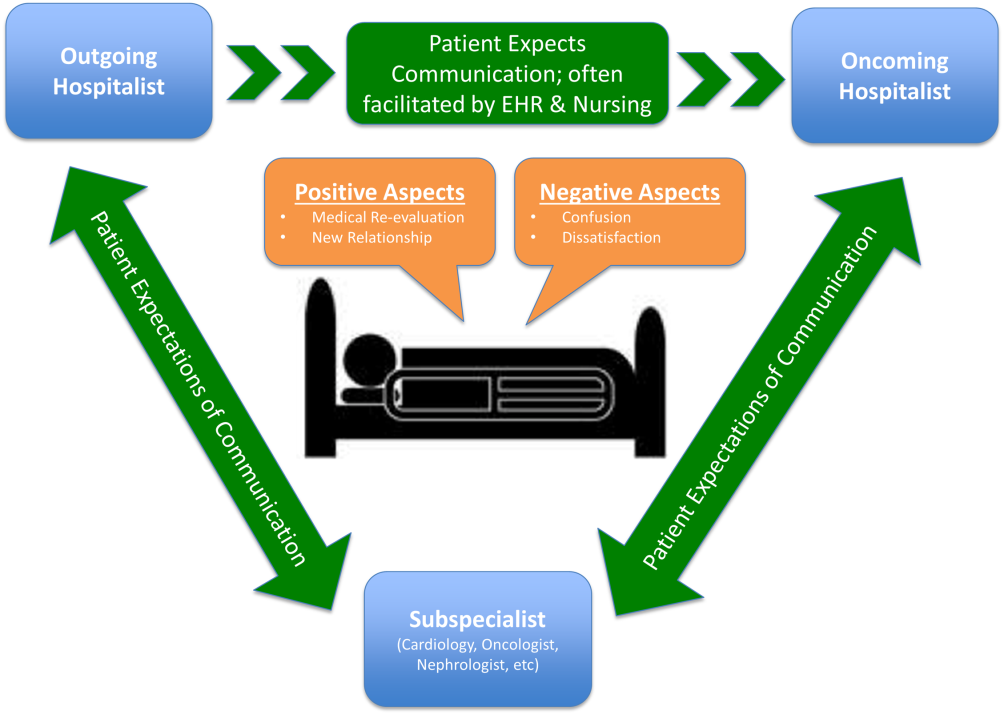
Conceptual Model
Following the collection and analysis of all patient responses, all themes were utilized to construct the ideal patient‐centered service handoff. The ideal transition describes open lines of communication between all involved parties, is facilitated by multiple modalities, such as the EHRs and nursing staff, and recognizes the patient as the primary stakeholder (Figure 1).
DISCUSSION
To our knowledge, this is the first qualitative investigation of the hospitalized patient's experience with service handoffs between hospitalists. The patient perspective adds a personal and first‐hand description of how fragmented care may impact the hospitalized patient experience.
Of the 6 themes, communication was found to be the most pertinent to our respondents. Because much of patient care is an inherently communicative activity, it is not surprising that patients, as well as patient safety experts, have focused on communication as an area in need of improvement in transition processes.[17, 18] Moreover, multiple medical societies have directly called for improvements within this area, and have specifically recommended clear and direct communication of treatment plans between the patient and physician, timely exchange of information, and knowledge of who is primarily in charge of the patients care.[11] Not surprisingly, each of these recommendations appears to be echoed by our participants. This theme is especially important given that good physician‐patient communication has been noted to be a major goal in achieving patient‐centered care, and has been positively correlated to medication adherence, patient satisfaction, and physical health outcomes.[19, 20, 21, 22, 23]
Although not a substitute for face‐to‐face interactions, other communication interventions between physicians and patients should be considered. For example, get to know me posters placed in patient rooms have been shown to encourage communication between patients and physicians.[24] Additionally, physician face cards have been used to improve patients' abilities to identify and clarify physicians' roles in patient care.[25] As a patient put it, If they got a new one [hospitalist], just as if I got a new medicationprint out information on themlike where they went to med school, and stuff(patient 13). These modalities may represent highly implementable, cost‐effective adjuncts to current handoff methods that may improve lines of communication between physicians and patients.
In addition to the importance placed on physician‐patient communication, interprofessional communication between hospitalists and subspecialists was also highly regarded. Studies have shown that practice‐based interprofessional communication, such as daily interdisciplinary rounds and the use of external facilitators, can improve healthcare processes and outcomes.[26] However, these interventions must be weighed with the many conflicting factors that both hospitalists and subspecialists face on daily basis, including high patient volumes, time limitations, patient availability, and scheduling conflicts.[27] None the less, the strong emphasis patients placed on this line of communication highlights this domain as an area in which hospitalist and subspecialist can work together for systematic improvement.
Patients also recognized the complexity of the transfer process between hospitalists and called for improved transparency. For example, patients repeatedly requested to be informed prior to any changes in their hospitalists, a request that remains consistent with current guidelines.[11] There also existed a strong desire for a more formalized process of transitioning between hospitalists, which often described a handoff procedure that would occur at the patient's bedside. This desire seems to be mirrored in the data that show that patients prefer to interact with their care team at the bedside and report higher satisfaction when they are involved with their care.[28, 29] Unfortunately, this desire for more direct interaction with physicians runs counter to the current paradigm of patient care, where most activities on rounds do not take place at the bedside.[30]
In contrast to patient's calls for improved transparency, an equally large portion of patients expressed relative indifference to the transition. Whereas on the surface this may seem salutary, some studies suggest that a lack of patient activation and engagement may have adverse effects toward patients' overall care.[31] Furthermore, others have shown evidence of better healthcare experiences, improved health outcomes, and lower costs in patients who are more active in their care.[30, 31] Altogether, this suggests that despite some patients' indifference, physicians should continue to engage patients in their hospital care.[32]
Although prevailing sentiments among patient safety advocates are that patient handoffs are inherently dangerous and place patients at increased risk of adverse events, patients did not always share this concern. A frequently occurring theme was that the transition is an opportunity for medical reevaluation or the establishment of a new, possibly improved therapeutic alliance. Recognizing this viewpoint offers oncoming hospitalists the opportunity to focus on issues that the patient may have felt were not being properly addressed with their prior physician.
Finally, although our conceptual model is not a strict guideline, we believe that any future studies should consider this framework when constructing interventions to improve service‐level handoffs. Several interventions already exist. For instance, educational interventions, such as patient‐centered interviewing, have been shown to improve patient satisfaction, compliance with medications, lead to fewer lawsuits, and improve health outcomes.[33, 34, 35] Additional methods of keeping the patient more informed include physician face sheets and performance of the handoff at the patient's bedside. Although well known in nursing literature, the idea of physicians performing handoffs at the patient's bedside is a particularly patient‐centric process.[36] This type of intervention may have the ability to transform the handoff from the current state of a 2‐way street, in which information is passed between 2 hospitalists, to a 3‐way stop, in which both hospitalists and the patient are able to communicate at this critical junction of care.
Although our study does offer new insight into the effects of discontinuous care, its exploratory nature does have limitations. First, being performed at a single academic center limits our ability to generalize our findings. Second, perspectives of those who did not wish to participate, patients' family members or caregivers, and those who were not queried, could highly differ from those we interviewed. Additionally, we did not collect data on patients' diagnoses or reason for admission, thus limiting our ability to assess if certain diagnosis or subpopulations predispose patients to experiencing a service handoff. Third, although our study was restricted to English‐speaking patients only, we must consider that non‐English speakers would likely suffer from even greater communication barriers than those who took part in our study. Finally, our interviews and data analysis were conducted by hospitalists, which could have subconsciously influenced the interview process, and the interpretation of patient responses. However, we tried to mitigate these issues by having the same individual interview all participants, by using an interview guide to ensure cross‐cohort consistency, by using open‐ended questions, and by attempting to give patients every opportunity to express themselves.
CONCLUSIONS
From a patients' perspective, inpatient service handoffs are often opaque experiences that are highlighted by poor communication between physicians and patients. Although deficits in communication and transparency acted as barriers to a patient‐centered handoff, physicians should recognize that service handoffs may also represent opportunities for improvement, and should focus on these domains when they start on a new service.
Disclosures
All funding for this project was provided by the Section of Hospital Medicine at The University of Chicago Medical Center. The data from this article were presented at the Society of Hospital Medicine Annual Conference, National Harbor, March 31, 2015, and at the Society of General Internal Medicine National Meeting in Toronto, Canada, April 23, 2015. The authors report that no conflicts of interest, financial or otherwise, exist.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- , , , et al. Continuity of care and the risk of preventable hospitalization in older adults. JAMA Intern Med. 2013;173(20):1879–1885.
- Agency for Healthcare Research and Quality. HCUPnet: a tool for identifying, tracking, and analyzing national hospital statistics. Available at: http://hcupnet.ahrq.gov/HCUPnet.jsp?Id=82B37DA366A36BAD6(8):438–444.
- , , , , , . Hospitalist handoffs: a systematic review and task force recommendations. J Hosp Med. 2009;4(7):433–440.
- , , , , . The impact of fragmentation of hospitalist care on length of stay. J Hosp Med. 2010;5(6):335–338.
- , , , et al. The impact of hospitalist discontinuity on hospital cost, readmissions, and patient satisfaction. J Gen Intern Med. 2014;29(7):1004–1008.
- , , , et al. The effect of hospitalist discontinuity on adverse events. J Hosp Med. 2015;10(3):147–151.
- Agency for Healthcare Research and Quality. HCAHPS Fact Sheet. CAHPS Hospital Survey August 2013. Available at: http://www.hcahpsonline.org/files/August_2013_HCAHPS_Fact_Sheet3.pdf. Accessed February 2, 2015.
- , , , et al. A conceptual framework for studying the safety of transitions in emergency care. In: Henriksen K, Battles JB, Marks ES, eds. Advances in Patient Safety: From Research to Implementation. Rockville, MD: Agency for Healthcare Research and Quality; 2005:309–321. Concepts and Methodology; vol 2. Available at: http://www.ncbi.nlm.nih.gov/books/NBK20522. Accessed January 15, 2015.
- , , , et al. Transitions of care consensus policy statement American College of Physicians‐Society of General Internal Medicine‐Society of Hospital Medicine‐American Geriatrics Society‐American College of Emergency Physicians‐Society of Academic Emergency Medicine. J Gen Intern Med. 2009;24(8):971–976.
- , . Grounded theory in medical education research: AMEE guide no. 70. Med Teach. 2012;34(10):850–861.
- . A purposeful approach to the constant comparative method in the analysis of qualitative interviews. Qual Quant. 2002;36(4):391–409.
- . The significance of saturation. Qual Health Res. 1995;5(2):147–149.
- , , , , . Standards for reporting qualitative research: a synthesis of recommendations. Acad Med. 2014;89(9):1245–1251.
- , , . Consolidated criteria for reporting qualitative research (COREQ): a 32‐item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–357.
- , , , . Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists. J Hosp Med. 2007;2(5):314–323.
- The Joint Commission. Hot Topics in Healthcare, Issue 2. Transitions of care: the need for collaboration across entire care continuum. Available at: http://www.jointcommission.org/assets/1/6/TOC_Hot_Topics.pdf. Accessed April 9, 2015.
- , . Physician communication and patient adherence to treatment: a meta‐analysis. Med Care. 2009;47(8):826–834.
- , . Impediments to adherence to post myocardial infarction medications. Curr Cardiol Rep. 2013;15(1):322.
- , , , , . Medical specialists' patient‐centered communication and patient‐reported outcomes. Med Care. 2007;45(4):330–339.
- , , , . Does doctor‐patient communication affect patient satisfaction with hospital care? Results of an analysis with a novel instrumental variable. Health Serv Res. 2008;43(5 pt 1):1505–1519.
- , , . Patient‐centredness in chronic illness: what is it and does it matter? Patient Educ Couns. 2003;51(3):197–206.
- , , , et al. Merging cultures: palliative care specialists in the medical intensive care unit. Crit Care Med. 2006;34(11 suppl):S388–S393.
- , , , et al. Improving inpatients' identification of their doctors: use of FACE cards. Jt Comm J Qual Patient Saf. 2009;35(12):613–619.
- , , . Interprofessional collaboration: effects of practice‐based interventions on professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2009;(3):CD000072.
- , , , et al. Identifying and overcoming the barriers to bedside rounds: a multicenter qualitative study. Acad Med. 2014;89(2):326–334.
- , , , , . The effect of bedside case presentations on patients' perceptions of their medical care. N Engl J Med. 1997;336(16):1150–1155.
- , , , . Patient‐centered interprofessional collaborative care: factors associated with bedside interprofessional rounds. J Gen Intern Med. 2014;29(7):1040–1047.
- , , , et al. Attending rounds in the current era: what is and is not happening. JAMA Intern Med. 2013;173(12):1084–1089.
- , . What the evidence shows about patient activation: better health outcomes and care experiences; fewer data on costs. Health Aff (Millwood). 2013;32(2):207–214.
- , , , , . When patient activation levels change, health outcomes and costs change, too. Health Aff Proj Hope. 2015;34(3):431–437.
- , , , et al. Evidence‐based guidelines for teaching patient‐centered interviewing. Patient Educ Couns. 2000;39(1):27–36.
- , , . Meta‐analysis of correlates of provider behavior in medical encounters. Med Care. 1988;26(7):657–675.
- , . Characteristics of potential plaintiffs in malpractice litigation. Ann Intern Med. 1994;120(9):792–798.
- , , , , . Bedside shift reports: what does the evidence say? J Nurs Adm. 2014;44(10):541–545.
Studies examining the importance of continuity of care have shown that patients who maintain a continuous relationship with a single physician have improved outcomes.[1, 2] However, most of these studies were performed in the outpatient, rather than the inpatient setting. With over 35 million patients admitted to hospitals in 2013, along with the significant increase in hospital discontinuity over recent years, the impact of inpatient continuity of care on quality outcomes and patient satisfaction is becoming increasingly relevant.[3, 4]
Service handoffs, when a physician hands over treatment responsibility for a panel of patients and is not expected to return, are a type of handoff that contributes to inpatient discontinuity. In particular, service handoffs between hospitalists are an especially common and inherently risky type of transition, as there is a severing of an established relationship during a patient's hospitalization. Unfortunately, due to the lack of evidence on the effects of service handoffs, current guidelines are limited in their recommendations.[5] Whereas several recent studies have begun to explore the effects of these handoffs, no prior study has examined this issue from a patient's perspective.[6, 7, 8]
Patients are uniquely positioned to inform us about their experiences in care transitions. Furthermore, with patient satisfaction now affecting Medicare reimbursement rates, patient experiences while in the hospital are becoming even more significant.[9] Despite this emphasis toward more patient‐centered care, no study has explored the hospitalized patient's experience with hospitalist service handoffs. Our goal was to qualitatively assess the hospitalized patients' experiences with transitions between hospitalists to develop a conceptual model to inform future work on improving inpatient transitions of care.
METHODS
Sampling and Recruitment
We conducted bedside interviews of hospitalized patients at an urban academic medical center from October 2014 through December 2014. The hospitalist service consists of a physician and an advanced nurse practitioner (ANP) who divide a panel of patients that consist of general medicine and subspecialty patients who are often comanaged with hepatology, oncology, and nephrology subspecialists. We performed a purposive selection of patients who could potentially comment on their experience with a hospitalist service transition using the following method: 48 hours after a service handoff (ie, an outgoing physician completing 1 week on service, then transfers the care of the patient to a new oncoming hospitalist), oncoming hospitalists were approached and asked if any patient on their service had experienced a service handoff and still remained in the hospital. A 48‐hour time period was chosen to give the patients time to familiarize themselves with their new hospitalist, allowing them to properly comment on the handoff. Patients who were managed by the ANP, who were non‐English speaking, or who were deemed to have an altered mental status based on clinical suspicion by the interviewing physician (C.M.W.) were excluded from participation. Following each weekly service transition, a list of patients who met the above criteria was collected from 4 nonteaching hospitalist services, and were approached by the primary investigator (C.M.W.) and asked if they would be willing to participate. All patients were general medicine patients and no exclusions were made based on physical location within the hospital. Those who agreed provided signed written consent prior to participation to allow access to the electronic health records (EHRs) by study personnel.
Data Collection
Patients were administered a 9‐question, semistructured interview that was informed by expert opinion and existing literature, which was developed to elicit their perspective regarding their transition between hospitalists.[10, 11] No formal changes were made to the interview guide during the study period, and all patients were asked the same questions. Outcomes from interim analysis guided further questioning in subsequent interviews so as to increase the depth of patient responses (ie, Can you explain your response in greater depth?). Prior to the interview, patients were read a description of a hospitalist, and were reminded which hospitalists had cared for them during their stay (see Supporting Information, Appendix 1, in the online version of this article). If family members or a caregiver were present at the time of interview, they were asked not to comment. No repeat interviews were carried out.
All interviews were performed privately in single‐occupancy rooms, digitally recorded using an iPad (Apple, Cupertino, CA) and professionally transcribed verbatim (Rev, San Francisco, CA). All analysis was performed using MAXQDA Software (VERBI Software GmbH, Berlin, Germany). We obtained demographic information about each patient through chart review
Data Analysis
Grounded theory was utilized, with an inductive approach with no a priori hypothesis.[12] The constant comparative method was used to generate emerging and reoccurring themes.[13] Units of analysis were sentences and phrases. Our research team consisted of 4 academic hospitalists, 2 with backgrounds in clinical medicine, medical education, and qualitative analysis (J.M.F., V.M.A.), 1 as a clinician (C.M.W.), and 1 in health economics (D.O.M.). Interim analysis was performed on a weekly basis (C.M.W.), during which time a coding template was created and refined through an iterative process (C.M.W., J.M.F.). All disagreements in coded themes were resolved through group discussion until full consensus was reached. Each week, responses were assessed for thematic saturation.[14] Interviews were continued if new themes arose during this analysis. Data collection was ended once we ceased to extract new topics from participants. A summary of all themes was then presented to a group of 10 patients who met the same inclusion criteria for respondent validation and member checking. All reporting was performed within the Standards for Reporting Qualitative Research, with additional guidance derived from the Consolidated Criteria for Reporting Qualitative Research.[15, 16] The University of Chicago Institutional Review Board approved this protocol.
RESULTS
In total, 43 eligible patients were recruited, and 40 (93%) agreed to participate. Interviewed patients were between 51 and 65 (39%) years old, had a mean age of 54.5 (15) years, were predominantly female (65%), African American (58%), had a median length of stay at the time of interview of 6.5 days (interquartile range [IQR]: 48), and had an average of 2.0 (IQR: 13) hospitalists oversee their care at the time of interview (Table 1). Interview times ranged from 10:25 to 25:48 minutes, with an average of 15:32 minutes.
| Value | |
|---|---|
| |
| Response rate, n (%) | 40/43 (93) |
| Age, mean SD | 54.5 15 |
| Sex, n (%) | |
| Female | 26 (65) |
| Male | 14 (35) |
| Race, n (%) | |
| African American | 23 (58) |
| White | 16 (40) |
| Hispanic | 1 (2) |
| Median LOS at time of interview, d (IQR) | 6.5 (48) |
| Median no. of hospitalists at time of interview, n (IQR) | 2.0 (13) |
We identified 6 major themes on patient perceptions of hospitalist service handoffs including (1) physician‐patient communication, (2) transparency in the hospitalist transition process, (3) indifference toward the hospitalist transition, (4) hospitalist‐subspecialist communication, (5) recognition of new opportunities due to a transition, and (6) hospitalists' bedside manner (Table 2).
| Themes | Subthemes | Representative Quotes |
|---|---|---|
| Physician‐patient communication | Patients dislike redundant communication with oncoming hospitalist. | I mean it's just you always have to explain your situation over and over and over again. (patient 14) |
| When I said it once already, then you're repeating it to another doctor. I feel as if that hospitalist didn't talk to the other hospitalist. (patient 7) | ||
| Poor communication can negatively affect the doctor‐patient relationship. | They don't really want to explain things. They don't think I'll understand. I think & yeah, I'm okay. You don't even have to put it in layman's terms. I know medical. I'm in nursing school. I have a year left. But even if you didn't know that, I would still hope you would try to tell me what was going on instead of just doing it in your head, and treating it. (patient 2) | |
| I mean it's just you always have to explain your situation over and over and over again. After a while you just stop trusting them. (patient 20) | ||
| Good communication can positively affect the doctor‐patient relationship. | Just continue with the communication, the open communication, and always stress to me that I have a voice and just going out of their way to do whatever they can to help me through whatever I'm going through. (patient 1) | |
| Transparency in transition | Patients want to be informed prior to a service changeover. | I think they should be told immediately, even maybe given prior notice, like this may happen, just so you're not surprised when it happens. (patient 15) |
| When the doctor approached me, he let me know that he wasn't going to be here the next day and there was going to be another doctor coming in. That made me feel comfortable. (patient 9) | ||
| Patients desire a more formalized process in the service changeover. | People want things to be consistent. People don't like change. They like routine. So, if he's leaving, you're coming on, I'd like for him to bring you in, introduce you to me, and for you just assure me that I'll take care of you. (patient 4) | |
| Just like when you get a new medication, you're given all this information on it. So when you get a new hospitalist, shouldn't I get all the information on them? Like where they went to school, what they look like. (patient 23) | ||
| Patients want clearer definition of the roles the physicians will play in their care. | The first time I was hospitalized for the first time I had all these different doctors coming in, and I had the residency, and the specialists, and the department, and I don't know who's who. What I asked them to do is when they come in the room, which they did, but introduce it a little more for me. Write it down like these are the special team and these are the doctors because even though they come in and give me their name, I have no idea what they're doing. (patient 5) | |
| Someone should explain the setup and who people are. Someone would say, Okay when you're in a hospital this is your [doctor's] role. Like they should have booklets and everything. (patient 19) | ||
| Indifference toward transition | Many patients have trust in service changeovers. | [S]o as long as everybody's on board and communicates well and efficiently, I don't have a problem with it. (patient 6) |
| To me, it really wasn't no preference, as long as I was getting the care that I needed. (patient 21) | ||
| It's not a concern as long as they're on the same page. (patient 17) | ||
| Hospitalist‐specialist communication | Patients are concerned about communication between their hospitalist and their subspecialists. | The more cooks you get in the kitchen, the more things get to get lost, so I'm always concerned that they're not sharing the same information, especially when you're getting asked the same questions that you might have just answered the last hour ago. (patient 9) |
| I don't know if the hospitalist are talking to them [subspecialist]. They haven't got time. (patient 35) | ||
| Patients place trust in the communication between hospitalist and subspecialist. | I think among the teams themselveswhich is my pain doctor, Dr. K's group, the oncology group itself, they switch off and trade with each other and they all speak the same language so that works out good. (patient 3) | |
| Lack of interprofessional communication can lead to patient concern. | I was afraid that one was going to drop the ball on something and not pass something on, or you know. (patient 11) | |
| I had numerous doctors who all seemed to not communicate with each other at all or did so by email or whatever. They didn't just sit down together and say we feel this way and we feel that way. I didn't like that at all. (patient 10) | ||
| New opportunities due to transition | Patients see new doctor as opportunity for medical reevaluation. | I see it as two heads are better than one, three heads are better than one, four heads are better than one. When people put their heads together to work towards a common goal, especially when they're, you know, people working their craft, it can't be bad. (patient 9) |
| I finally got my ears looked atbecause I've asked to have my ears looked at since Mondayand the new doc is trying to make an effort to look at them. (patient 39) | ||
| Patients see service changeover as an opportunity to form a better personal relationship. | Having a new hospitalist it gives you opportunity for a new beginning. (patient 11) | |
| Bedside manner | Good bedside manner can assist in a service changeover. | Some of them are all business‐like but some of them are, Well how do you feel today? Hi, how are you? So this made a little difference. You feel more comfortable. You're going to be more comfortable with them. Their bedside manner helps. (patient 16) |
| It's just like when a doctor sits down and talks to you, they just seem more relaxed and more .... I know they're very busy and they have lots of things to do and other patients to see, but while they're in there with you, you know, you don't get too much time with them. So bedside manner is just so important. (patient 24) | ||
| Poor bedside manner can be detrimental in transition. | [B]ecause they be so busy they claim they don't have time just to sit and talk to a patient, and make sure they all right. (patient 17) |
Physician‐Patient Communication
Communication between the physician and the patient was an important element in patients' assessment of their experience. Patient's tended to divide physician‐patient communication into 2 categories: good communication, which consisted of open communication (patient 1) and patient engagement, and bad communication, which was described as physicians not sharing information or taking the time to explain the course of care in words that I'll understand (patient 2). Patients also described dissatisfaction with redundant communication between multiple hospitalists and the frustration of often having to describe their clinical course to multiple providers.
Transparency in Communication
The desire to have greater transparency in the handoff process was another common theme. This was likely due to the fact that 34/40 (85%) of surveyed patients were unaware that a service changeover had ever taken place. This lack of transparency was viewed to have further downstream consequences as patients stated that there should be a level of transparency, and when it's not, then there is always trust issues (patient 1). Upon further questioning as to how to make the process more transparent, many patients recommended a formalized, face‐to‐face introduction involving the patient and both hospitalists, in which the outgoing hospitalist would, bring you [oncoming hospitalist] in, and introduce you to me (patient 4).
Patients often stated that given the large spectrum of physicians they might encounter during their stay (ie, medical student, resident, hospitalist attending, subspecialty fellow, subspecialist attending), clearer definitions of physicians' roles are needed.
Hospitalist‐Specialist Communication
Concern about the communication between their hospitalist and subspecialist was another predominant theme. Conflicting and unclear directions from multiple services were especially frustrating, as a patient stated, One guy took me off this pill, the other guy wants me on that pill, I'm like okay, I can't do both (patient 8). Furthermore, a subset of patients referenced their subspecialist as their primary care provider and preferred their subspecialist for guidance in their hospital course, rather than their hospitalist. This often appeared in cases where the patient had an established relationship with the subspecialist prior to their hospitalization.
New Opportunities Due to Transition
Patients expressed positive feelings toward service handoffs by viewing the transition as an opportunity for medical reevaluation by a new physician. Patients told of instances in which a specific complaint was not being addressed by the first physician, but would be addressed by the second (oncoming) physician. A commonly expressed idea was that the oncoming physician might know something that he [Dr. B] didn't know, and since Dr. B was only here for a week, why not give him [oncoming hospitalist] a chance (patient 10). Patients would also describe the transition as an opportunity to form, and possibly improve, therapeutic alliances with a new hospitalist.
Bedside Manner
Bedside manner was another commonly mentioned thematic element. Patients were often quick to forget prior problems or issues that they may have suffered because of the transition if the oncoming physician was perceived to have a good bedside manner, often described as someone who formally introduced themselves, was considered relaxed, and would take the time to sit and talk with the patient. As a patient put it, [S]he sat down and got to know meand asked me what I wanted to do (patient 12). Conversely, patients described instances in which a perceived bad bedside manner led to a poor relationship between the physician and the patient, in which trust and comfort (patient 11) were sacrificed.
Indifference Toward Transition
In contrast to some of the previous findings, which called for improved interactions between physicians and patients, we also discovered a theme of indifference toward the transition. Several patients stated feelings of trust with the medical system, and were content with the service changeover as long as they felt that their medical needs were being met. Patients also tended to express a level of acceptance with the transition, and tended to believe that this was the price we pay for being here [in the hospital] (patient 7).
Conceptual Model
Following the collection and analysis of all patient responses, all themes were utilized to construct the ideal patient‐centered service handoff. The ideal transition describes open lines of communication between all involved parties, is facilitated by multiple modalities, such as the EHRs and nursing staff, and recognizes the patient as the primary stakeholder (Figure 1).

DISCUSSION
To our knowledge, this is the first qualitative investigation of the hospitalized patient's experience with service handoffs between hospitalists. The patient perspective adds a personal and first‐hand description of how fragmented care may impact the hospitalized patient experience.
Of the 6 themes, communication was found to be the most pertinent to our respondents. Because much of patient care is an inherently communicative activity, it is not surprising that patients, as well as patient safety experts, have focused on communication as an area in need of improvement in transition processes.[17, 18] Moreover, multiple medical societies have directly called for improvements within this area, and have specifically recommended clear and direct communication of treatment plans between the patient and physician, timely exchange of information, and knowledge of who is primarily in charge of the patients care.[11] Not surprisingly, each of these recommendations appears to be echoed by our participants. This theme is especially important given that good physician‐patient communication has been noted to be a major goal in achieving patient‐centered care, and has been positively correlated to medication adherence, patient satisfaction, and physical health outcomes.[19, 20, 21, 22, 23]
Although not a substitute for face‐to‐face interactions, other communication interventions between physicians and patients should be considered. For example, get to know me posters placed in patient rooms have been shown to encourage communication between patients and physicians.[24] Additionally, physician face cards have been used to improve patients' abilities to identify and clarify physicians' roles in patient care.[25] As a patient put it, If they got a new one [hospitalist], just as if I got a new medicationprint out information on themlike where they went to med school, and stuff(patient 13). These modalities may represent highly implementable, cost‐effective adjuncts to current handoff methods that may improve lines of communication between physicians and patients.
In addition to the importance placed on physician‐patient communication, interprofessional communication between hospitalists and subspecialists was also highly regarded. Studies have shown that practice‐based interprofessional communication, such as daily interdisciplinary rounds and the use of external facilitators, can improve healthcare processes and outcomes.[26] However, these interventions must be weighed with the many conflicting factors that both hospitalists and subspecialists face on daily basis, including high patient volumes, time limitations, patient availability, and scheduling conflicts.[27] None the less, the strong emphasis patients placed on this line of communication highlights this domain as an area in which hospitalist and subspecialist can work together for systematic improvement.
Patients also recognized the complexity of the transfer process between hospitalists and called for improved transparency. For example, patients repeatedly requested to be informed prior to any changes in their hospitalists, a request that remains consistent with current guidelines.[11] There also existed a strong desire for a more formalized process of transitioning between hospitalists, which often described a handoff procedure that would occur at the patient's bedside. This desire seems to be mirrored in the data that show that patients prefer to interact with their care team at the bedside and report higher satisfaction when they are involved with their care.[28, 29] Unfortunately, this desire for more direct interaction with physicians runs counter to the current paradigm of patient care, where most activities on rounds do not take place at the bedside.[30]
In contrast to patient's calls for improved transparency, an equally large portion of patients expressed relative indifference to the transition. Whereas on the surface this may seem salutary, some studies suggest that a lack of patient activation and engagement may have adverse effects toward patients' overall care.[31] Furthermore, others have shown evidence of better healthcare experiences, improved health outcomes, and lower costs in patients who are more active in their care.[30, 31] Altogether, this suggests that despite some patients' indifference, physicians should continue to engage patients in their hospital care.[32]
Although prevailing sentiments among patient safety advocates are that patient handoffs are inherently dangerous and place patients at increased risk of adverse events, patients did not always share this concern. A frequently occurring theme was that the transition is an opportunity for medical reevaluation or the establishment of a new, possibly improved therapeutic alliance. Recognizing this viewpoint offers oncoming hospitalists the opportunity to focus on issues that the patient may have felt were not being properly addressed with their prior physician.
Finally, although our conceptual model is not a strict guideline, we believe that any future studies should consider this framework when constructing interventions to improve service‐level handoffs. Several interventions already exist. For instance, educational interventions, such as patient‐centered interviewing, have been shown to improve patient satisfaction, compliance with medications, lead to fewer lawsuits, and improve health outcomes.[33, 34, 35] Additional methods of keeping the patient more informed include physician face sheets and performance of the handoff at the patient's bedside. Although well known in nursing literature, the idea of physicians performing handoffs at the patient's bedside is a particularly patient‐centric process.[36] This type of intervention may have the ability to transform the handoff from the current state of a 2‐way street, in which information is passed between 2 hospitalists, to a 3‐way stop, in which both hospitalists and the patient are able to communicate at this critical junction of care.
Although our study does offer new insight into the effects of discontinuous care, its exploratory nature does have limitations. First, being performed at a single academic center limits our ability to generalize our findings. Second, perspectives of those who did not wish to participate, patients' family members or caregivers, and those who were not queried, could highly differ from those we interviewed. Additionally, we did not collect data on patients' diagnoses or reason for admission, thus limiting our ability to assess if certain diagnosis or subpopulations predispose patients to experiencing a service handoff. Third, although our study was restricted to English‐speaking patients only, we must consider that non‐English speakers would likely suffer from even greater communication barriers than those who took part in our study. Finally, our interviews and data analysis were conducted by hospitalists, which could have subconsciously influenced the interview process, and the interpretation of patient responses. However, we tried to mitigate these issues by having the same individual interview all participants, by using an interview guide to ensure cross‐cohort consistency, by using open‐ended questions, and by attempting to give patients every opportunity to express themselves.
CONCLUSIONS
From a patients' perspective, inpatient service handoffs are often opaque experiences that are highlighted by poor communication between physicians and patients. Although deficits in communication and transparency acted as barriers to a patient‐centered handoff, physicians should recognize that service handoffs may also represent opportunities for improvement, and should focus on these domains when they start on a new service.
Disclosures
All funding for this project was provided by the Section of Hospital Medicine at The University of Chicago Medical Center. The data from this article were presented at the Society of Hospital Medicine Annual Conference, National Harbor, March 31, 2015, and at the Society of General Internal Medicine National Meeting in Toronto, Canada, April 23, 2015. The authors report that no conflicts of interest, financial or otherwise, exist.
Studies examining the importance of continuity of care have shown that patients who maintain a continuous relationship with a single physician have improved outcomes.[1, 2] However, most of these studies were performed in the outpatient, rather than the inpatient setting. With over 35 million patients admitted to hospitals in 2013, along with the significant increase in hospital discontinuity over recent years, the impact of inpatient continuity of care on quality outcomes and patient satisfaction is becoming increasingly relevant.[3, 4]
Service handoffs, when a physician hands over treatment responsibility for a panel of patients and is not expected to return, are a type of handoff that contributes to inpatient discontinuity. In particular, service handoffs between hospitalists are an especially common and inherently risky type of transition, as there is a severing of an established relationship during a patient's hospitalization. Unfortunately, due to the lack of evidence on the effects of service handoffs, current guidelines are limited in their recommendations.[5] Whereas several recent studies have begun to explore the effects of these handoffs, no prior study has examined this issue from a patient's perspective.[6, 7, 8]
Patients are uniquely positioned to inform us about their experiences in care transitions. Furthermore, with patient satisfaction now affecting Medicare reimbursement rates, patient experiences while in the hospital are becoming even more significant.[9] Despite this emphasis toward more patient‐centered care, no study has explored the hospitalized patient's experience with hospitalist service handoffs. Our goal was to qualitatively assess the hospitalized patients' experiences with transitions between hospitalists to develop a conceptual model to inform future work on improving inpatient transitions of care.
METHODS
Sampling and Recruitment
We conducted bedside interviews of hospitalized patients at an urban academic medical center from October 2014 through December 2014. The hospitalist service consists of a physician and an advanced nurse practitioner (ANP) who divide a panel of patients that consist of general medicine and subspecialty patients who are often comanaged with hepatology, oncology, and nephrology subspecialists. We performed a purposive selection of patients who could potentially comment on their experience with a hospitalist service transition using the following method: 48 hours after a service handoff (ie, an outgoing physician completing 1 week on service, then transfers the care of the patient to a new oncoming hospitalist), oncoming hospitalists were approached and asked if any patient on their service had experienced a service handoff and still remained in the hospital. A 48‐hour time period was chosen to give the patients time to familiarize themselves with their new hospitalist, allowing them to properly comment on the handoff. Patients who were managed by the ANP, who were non‐English speaking, or who were deemed to have an altered mental status based on clinical suspicion by the interviewing physician (C.M.W.) were excluded from participation. Following each weekly service transition, a list of patients who met the above criteria was collected from 4 nonteaching hospitalist services, and were approached by the primary investigator (C.M.W.) and asked if they would be willing to participate. All patients were general medicine patients and no exclusions were made based on physical location within the hospital. Those who agreed provided signed written consent prior to participation to allow access to the electronic health records (EHRs) by study personnel.
Data Collection
Patients were administered a 9‐question, semistructured interview that was informed by expert opinion and existing literature, which was developed to elicit their perspective regarding their transition between hospitalists.[10, 11] No formal changes were made to the interview guide during the study period, and all patients were asked the same questions. Outcomes from interim analysis guided further questioning in subsequent interviews so as to increase the depth of patient responses (ie, Can you explain your response in greater depth?). Prior to the interview, patients were read a description of a hospitalist, and were reminded which hospitalists had cared for them during their stay (see Supporting Information, Appendix 1, in the online version of this article). If family members or a caregiver were present at the time of interview, they were asked not to comment. No repeat interviews were carried out.
All interviews were performed privately in single‐occupancy rooms, digitally recorded using an iPad (Apple, Cupertino, CA) and professionally transcribed verbatim (Rev, San Francisco, CA). All analysis was performed using MAXQDA Software (VERBI Software GmbH, Berlin, Germany). We obtained demographic information about each patient through chart review
Data Analysis
Grounded theory was utilized, with an inductive approach with no a priori hypothesis.[12] The constant comparative method was used to generate emerging and reoccurring themes.[13] Units of analysis were sentences and phrases. Our research team consisted of 4 academic hospitalists, 2 with backgrounds in clinical medicine, medical education, and qualitative analysis (J.M.F., V.M.A.), 1 as a clinician (C.M.W.), and 1 in health economics (D.O.M.). Interim analysis was performed on a weekly basis (C.M.W.), during which time a coding template was created and refined through an iterative process (C.M.W., J.M.F.). All disagreements in coded themes were resolved through group discussion until full consensus was reached. Each week, responses were assessed for thematic saturation.[14] Interviews were continued if new themes arose during this analysis. Data collection was ended once we ceased to extract new topics from participants. A summary of all themes was then presented to a group of 10 patients who met the same inclusion criteria for respondent validation and member checking. All reporting was performed within the Standards for Reporting Qualitative Research, with additional guidance derived from the Consolidated Criteria for Reporting Qualitative Research.[15, 16] The University of Chicago Institutional Review Board approved this protocol.
RESULTS
In total, 43 eligible patients were recruited, and 40 (93%) agreed to participate. Interviewed patients were between 51 and 65 (39%) years old, had a mean age of 54.5 (15) years, were predominantly female (65%), African American (58%), had a median length of stay at the time of interview of 6.5 days (interquartile range [IQR]: 48), and had an average of 2.0 (IQR: 13) hospitalists oversee their care at the time of interview (Table 1). Interview times ranged from 10:25 to 25:48 minutes, with an average of 15:32 minutes.
| Value | |
|---|---|
| |
| Response rate, n (%) | 40/43 (93) |
| Age, mean SD | 54.5 15 |
| Sex, n (%) | |
| Female | 26 (65) |
| Male | 14 (35) |
| Race, n (%) | |
| African American | 23 (58) |
| White | 16 (40) |
| Hispanic | 1 (2) |
| Median LOS at time of interview, d (IQR) | 6.5 (48) |
| Median no. of hospitalists at time of interview, n (IQR) | 2.0 (13) |
We identified 6 major themes on patient perceptions of hospitalist service handoffs including (1) physician‐patient communication, (2) transparency in the hospitalist transition process, (3) indifference toward the hospitalist transition, (4) hospitalist‐subspecialist communication, (5) recognition of new opportunities due to a transition, and (6) hospitalists' bedside manner (Table 2).
| Themes | Subthemes | Representative Quotes |
|---|---|---|
| Physician‐patient communication | Patients dislike redundant communication with oncoming hospitalist. | I mean it's just you always have to explain your situation over and over and over again. (patient 14) |
| When I said it once already, then you're repeating it to another doctor. I feel as if that hospitalist didn't talk to the other hospitalist. (patient 7) | ||
| Poor communication can negatively affect the doctor‐patient relationship. | They don't really want to explain things. They don't think I'll understand. I think & yeah, I'm okay. You don't even have to put it in layman's terms. I know medical. I'm in nursing school. I have a year left. But even if you didn't know that, I would still hope you would try to tell me what was going on instead of just doing it in your head, and treating it. (patient 2) | |
| I mean it's just you always have to explain your situation over and over and over again. After a while you just stop trusting them. (patient 20) | ||
| Good communication can positively affect the doctor‐patient relationship. | Just continue with the communication, the open communication, and always stress to me that I have a voice and just going out of their way to do whatever they can to help me through whatever I'm going through. (patient 1) | |
| Transparency in transition | Patients want to be informed prior to a service changeover. | I think they should be told immediately, even maybe given prior notice, like this may happen, just so you're not surprised when it happens. (patient 15) |
| When the doctor approached me, he let me know that he wasn't going to be here the next day and there was going to be another doctor coming in. That made me feel comfortable. (patient 9) | ||
| Patients desire a more formalized process in the service changeover. | People want things to be consistent. People don't like change. They like routine. So, if he's leaving, you're coming on, I'd like for him to bring you in, introduce you to me, and for you just assure me that I'll take care of you. (patient 4) | |
| Just like when you get a new medication, you're given all this information on it. So when you get a new hospitalist, shouldn't I get all the information on them? Like where they went to school, what they look like. (patient 23) | ||
| Patients want clearer definition of the roles the physicians will play in their care. | The first time I was hospitalized for the first time I had all these different doctors coming in, and I had the residency, and the specialists, and the department, and I don't know who's who. What I asked them to do is when they come in the room, which they did, but introduce it a little more for me. Write it down like these are the special team and these are the doctors because even though they come in and give me their name, I have no idea what they're doing. (patient 5) | |
| Someone should explain the setup and who people are. Someone would say, Okay when you're in a hospital this is your [doctor's] role. Like they should have booklets and everything. (patient 19) | ||
| Indifference toward transition | Many patients have trust in service changeovers. | [S]o as long as everybody's on board and communicates well and efficiently, I don't have a problem with it. (patient 6) |
| To me, it really wasn't no preference, as long as I was getting the care that I needed. (patient 21) | ||
| It's not a concern as long as they're on the same page. (patient 17) | ||
| Hospitalist‐specialist communication | Patients are concerned about communication between their hospitalist and their subspecialists. | The more cooks you get in the kitchen, the more things get to get lost, so I'm always concerned that they're not sharing the same information, especially when you're getting asked the same questions that you might have just answered the last hour ago. (patient 9) |
| I don't know if the hospitalist are talking to them [subspecialist]. They haven't got time. (patient 35) | ||
| Patients place trust in the communication between hospitalist and subspecialist. | I think among the teams themselveswhich is my pain doctor, Dr. K's group, the oncology group itself, they switch off and trade with each other and they all speak the same language so that works out good. (patient 3) | |
| Lack of interprofessional communication can lead to patient concern. | I was afraid that one was going to drop the ball on something and not pass something on, or you know. (patient 11) | |
| I had numerous doctors who all seemed to not communicate with each other at all or did so by email or whatever. They didn't just sit down together and say we feel this way and we feel that way. I didn't like that at all. (patient 10) | ||
| New opportunities due to transition | Patients see new doctor as opportunity for medical reevaluation. | I see it as two heads are better than one, three heads are better than one, four heads are better than one. When people put their heads together to work towards a common goal, especially when they're, you know, people working their craft, it can't be bad. (patient 9) |
| I finally got my ears looked atbecause I've asked to have my ears looked at since Mondayand the new doc is trying to make an effort to look at them. (patient 39) | ||
| Patients see service changeover as an opportunity to form a better personal relationship. | Having a new hospitalist it gives you opportunity for a new beginning. (patient 11) | |
| Bedside manner | Good bedside manner can assist in a service changeover. | Some of them are all business‐like but some of them are, Well how do you feel today? Hi, how are you? So this made a little difference. You feel more comfortable. You're going to be more comfortable with them. Their bedside manner helps. (patient 16) |
| It's just like when a doctor sits down and talks to you, they just seem more relaxed and more .... I know they're very busy and they have lots of things to do and other patients to see, but while they're in there with you, you know, you don't get too much time with them. So bedside manner is just so important. (patient 24) | ||
| Poor bedside manner can be detrimental in transition. | [B]ecause they be so busy they claim they don't have time just to sit and talk to a patient, and make sure they all right. (patient 17) |
Physician‐Patient Communication
Communication between the physician and the patient was an important element in patients' assessment of their experience. Patient's tended to divide physician‐patient communication into 2 categories: good communication, which consisted of open communication (patient 1) and patient engagement, and bad communication, which was described as physicians not sharing information or taking the time to explain the course of care in words that I'll understand (patient 2). Patients also described dissatisfaction with redundant communication between multiple hospitalists and the frustration of often having to describe their clinical course to multiple providers.
Transparency in Communication
The desire to have greater transparency in the handoff process was another common theme. This was likely due to the fact that 34/40 (85%) of surveyed patients were unaware that a service changeover had ever taken place. This lack of transparency was viewed to have further downstream consequences as patients stated that there should be a level of transparency, and when it's not, then there is always trust issues (patient 1). Upon further questioning as to how to make the process more transparent, many patients recommended a formalized, face‐to‐face introduction involving the patient and both hospitalists, in which the outgoing hospitalist would, bring you [oncoming hospitalist] in, and introduce you to me (patient 4).
Patients often stated that given the large spectrum of physicians they might encounter during their stay (ie, medical student, resident, hospitalist attending, subspecialty fellow, subspecialist attending), clearer definitions of physicians' roles are needed.
Hospitalist‐Specialist Communication
Concern about the communication between their hospitalist and subspecialist was another predominant theme. Conflicting and unclear directions from multiple services were especially frustrating, as a patient stated, One guy took me off this pill, the other guy wants me on that pill, I'm like okay, I can't do both (patient 8). Furthermore, a subset of patients referenced their subspecialist as their primary care provider and preferred their subspecialist for guidance in their hospital course, rather than their hospitalist. This often appeared in cases where the patient had an established relationship with the subspecialist prior to their hospitalization.
New Opportunities Due to Transition
Patients expressed positive feelings toward service handoffs by viewing the transition as an opportunity for medical reevaluation by a new physician. Patients told of instances in which a specific complaint was not being addressed by the first physician, but would be addressed by the second (oncoming) physician. A commonly expressed idea was that the oncoming physician might know something that he [Dr. B] didn't know, and since Dr. B was only here for a week, why not give him [oncoming hospitalist] a chance (patient 10). Patients would also describe the transition as an opportunity to form, and possibly improve, therapeutic alliances with a new hospitalist.
Bedside Manner
Bedside manner was another commonly mentioned thematic element. Patients were often quick to forget prior problems or issues that they may have suffered because of the transition if the oncoming physician was perceived to have a good bedside manner, often described as someone who formally introduced themselves, was considered relaxed, and would take the time to sit and talk with the patient. As a patient put it, [S]he sat down and got to know meand asked me what I wanted to do (patient 12). Conversely, patients described instances in which a perceived bad bedside manner led to a poor relationship between the physician and the patient, in which trust and comfort (patient 11) were sacrificed.
Indifference Toward Transition
In contrast to some of the previous findings, which called for improved interactions between physicians and patients, we also discovered a theme of indifference toward the transition. Several patients stated feelings of trust with the medical system, and were content with the service changeover as long as they felt that their medical needs were being met. Patients also tended to express a level of acceptance with the transition, and tended to believe that this was the price we pay for being here [in the hospital] (patient 7).
Conceptual Model
Following the collection and analysis of all patient responses, all themes were utilized to construct the ideal patient‐centered service handoff. The ideal transition describes open lines of communication between all involved parties, is facilitated by multiple modalities, such as the EHRs and nursing staff, and recognizes the patient as the primary stakeholder (Figure 1).

DISCUSSION
To our knowledge, this is the first qualitative investigation of the hospitalized patient's experience with service handoffs between hospitalists. The patient perspective adds a personal and first‐hand description of how fragmented care may impact the hospitalized patient experience.
Of the 6 themes, communication was found to be the most pertinent to our respondents. Because much of patient care is an inherently communicative activity, it is not surprising that patients, as well as patient safety experts, have focused on communication as an area in need of improvement in transition processes.[17, 18] Moreover, multiple medical societies have directly called for improvements within this area, and have specifically recommended clear and direct communication of treatment plans between the patient and physician, timely exchange of information, and knowledge of who is primarily in charge of the patients care.[11] Not surprisingly, each of these recommendations appears to be echoed by our participants. This theme is especially important given that good physician‐patient communication has been noted to be a major goal in achieving patient‐centered care, and has been positively correlated to medication adherence, patient satisfaction, and physical health outcomes.[19, 20, 21, 22, 23]
Although not a substitute for face‐to‐face interactions, other communication interventions between physicians and patients should be considered. For example, get to know me posters placed in patient rooms have been shown to encourage communication between patients and physicians.[24] Additionally, physician face cards have been used to improve patients' abilities to identify and clarify physicians' roles in patient care.[25] As a patient put it, If they got a new one [hospitalist], just as if I got a new medicationprint out information on themlike where they went to med school, and stuff(patient 13). These modalities may represent highly implementable, cost‐effective adjuncts to current handoff methods that may improve lines of communication between physicians and patients.
In addition to the importance placed on physician‐patient communication, interprofessional communication between hospitalists and subspecialists was also highly regarded. Studies have shown that practice‐based interprofessional communication, such as daily interdisciplinary rounds and the use of external facilitators, can improve healthcare processes and outcomes.[26] However, these interventions must be weighed with the many conflicting factors that both hospitalists and subspecialists face on daily basis, including high patient volumes, time limitations, patient availability, and scheduling conflicts.[27] None the less, the strong emphasis patients placed on this line of communication highlights this domain as an area in which hospitalist and subspecialist can work together for systematic improvement.
Patients also recognized the complexity of the transfer process between hospitalists and called for improved transparency. For example, patients repeatedly requested to be informed prior to any changes in their hospitalists, a request that remains consistent with current guidelines.[11] There also existed a strong desire for a more formalized process of transitioning between hospitalists, which often described a handoff procedure that would occur at the patient's bedside. This desire seems to be mirrored in the data that show that patients prefer to interact with their care team at the bedside and report higher satisfaction when they are involved with their care.[28, 29] Unfortunately, this desire for more direct interaction with physicians runs counter to the current paradigm of patient care, where most activities on rounds do not take place at the bedside.[30]
In contrast to patient's calls for improved transparency, an equally large portion of patients expressed relative indifference to the transition. Whereas on the surface this may seem salutary, some studies suggest that a lack of patient activation and engagement may have adverse effects toward patients' overall care.[31] Furthermore, others have shown evidence of better healthcare experiences, improved health outcomes, and lower costs in patients who are more active in their care.[30, 31] Altogether, this suggests that despite some patients' indifference, physicians should continue to engage patients in their hospital care.[32]
Although prevailing sentiments among patient safety advocates are that patient handoffs are inherently dangerous and place patients at increased risk of adverse events, patients did not always share this concern. A frequently occurring theme was that the transition is an opportunity for medical reevaluation or the establishment of a new, possibly improved therapeutic alliance. Recognizing this viewpoint offers oncoming hospitalists the opportunity to focus on issues that the patient may have felt were not being properly addressed with their prior physician.
Finally, although our conceptual model is not a strict guideline, we believe that any future studies should consider this framework when constructing interventions to improve service‐level handoffs. Several interventions already exist. For instance, educational interventions, such as patient‐centered interviewing, have been shown to improve patient satisfaction, compliance with medications, lead to fewer lawsuits, and improve health outcomes.[33, 34, 35] Additional methods of keeping the patient more informed include physician face sheets and performance of the handoff at the patient's bedside. Although well known in nursing literature, the idea of physicians performing handoffs at the patient's bedside is a particularly patient‐centric process.[36] This type of intervention may have the ability to transform the handoff from the current state of a 2‐way street, in which information is passed between 2 hospitalists, to a 3‐way stop, in which both hospitalists and the patient are able to communicate at this critical junction of care.
Although our study does offer new insight into the effects of discontinuous care, its exploratory nature does have limitations. First, being performed at a single academic center limits our ability to generalize our findings. Second, perspectives of those who did not wish to participate, patients' family members or caregivers, and those who were not queried, could highly differ from those we interviewed. Additionally, we did not collect data on patients' diagnoses or reason for admission, thus limiting our ability to assess if certain diagnosis or subpopulations predispose patients to experiencing a service handoff. Third, although our study was restricted to English‐speaking patients only, we must consider that non‐English speakers would likely suffer from even greater communication barriers than those who took part in our study. Finally, our interviews and data analysis were conducted by hospitalists, which could have subconsciously influenced the interview process, and the interpretation of patient responses. However, we tried to mitigate these issues by having the same individual interview all participants, by using an interview guide to ensure cross‐cohort consistency, by using open‐ended questions, and by attempting to give patients every opportunity to express themselves.
CONCLUSIONS
From a patients' perspective, inpatient service handoffs are often opaque experiences that are highlighted by poor communication between physicians and patients. Although deficits in communication and transparency acted as barriers to a patient‐centered handoff, physicians should recognize that service handoffs may also represent opportunities for improvement, and should focus on these domains when they start on a new service.
Disclosures
All funding for this project was provided by the Section of Hospital Medicine at The University of Chicago Medical Center. The data from this article were presented at the Society of Hospital Medicine Annual Conference, National Harbor, March 31, 2015, and at the Society of General Internal Medicine National Meeting in Toronto, Canada, April 23, 2015. The authors report that no conflicts of interest, financial or otherwise, exist.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- , , , et al. Continuity of care and the risk of preventable hospitalization in older adults. JAMA Intern Med. 2013;173(20):1879–1885.
- Agency for Healthcare Research and Quality. HCUPnet: a tool for identifying, tracking, and analyzing national hospital statistics. Available at: http://hcupnet.ahrq.gov/HCUPnet.jsp?Id=82B37DA366A36BAD6(8):438–444.
- , , , , , . Hospitalist handoffs: a systematic review and task force recommendations. J Hosp Med. 2009;4(7):433–440.
- , , , , . The impact of fragmentation of hospitalist care on length of stay. J Hosp Med. 2010;5(6):335–338.
- , , , et al. The impact of hospitalist discontinuity on hospital cost, readmissions, and patient satisfaction. J Gen Intern Med. 2014;29(7):1004–1008.
- , , , et al. The effect of hospitalist discontinuity on adverse events. J Hosp Med. 2015;10(3):147–151.
- Agency for Healthcare Research and Quality. HCAHPS Fact Sheet. CAHPS Hospital Survey August 2013. Available at: http://www.hcahpsonline.org/files/August_2013_HCAHPS_Fact_Sheet3.pdf. Accessed February 2, 2015.
- , , , et al. A conceptual framework for studying the safety of transitions in emergency care. In: Henriksen K, Battles JB, Marks ES, eds. Advances in Patient Safety: From Research to Implementation. Rockville, MD: Agency for Healthcare Research and Quality; 2005:309–321. Concepts and Methodology; vol 2. Available at: http://www.ncbi.nlm.nih.gov/books/NBK20522. Accessed January 15, 2015.
- , , , et al. Transitions of care consensus policy statement American College of Physicians‐Society of General Internal Medicine‐Society of Hospital Medicine‐American Geriatrics Society‐American College of Emergency Physicians‐Society of Academic Emergency Medicine. J Gen Intern Med. 2009;24(8):971–976.
- , . Grounded theory in medical education research: AMEE guide no. 70. Med Teach. 2012;34(10):850–861.
- . A purposeful approach to the constant comparative method in the analysis of qualitative interviews. Qual Quant. 2002;36(4):391–409.
- . The significance of saturation. Qual Health Res. 1995;5(2):147–149.
- , , , , . Standards for reporting qualitative research: a synthesis of recommendations. Acad Med. 2014;89(9):1245–1251.
- , , . Consolidated criteria for reporting qualitative research (COREQ): a 32‐item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–357.
- , , , . Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists. J Hosp Med. 2007;2(5):314–323.
- The Joint Commission. Hot Topics in Healthcare, Issue 2. Transitions of care: the need for collaboration across entire care continuum. Available at: http://www.jointcommission.org/assets/1/6/TOC_Hot_Topics.pdf. Accessed April 9, 2015.
- , . Physician communication and patient adherence to treatment: a meta‐analysis. Med Care. 2009;47(8):826–834.
- , . Impediments to adherence to post myocardial infarction medications. Curr Cardiol Rep. 2013;15(1):322.
- , , , , . Medical specialists' patient‐centered communication and patient‐reported outcomes. Med Care. 2007;45(4):330–339.
- , , , . Does doctor‐patient communication affect patient satisfaction with hospital care? Results of an analysis with a novel instrumental variable. Health Serv Res. 2008;43(5 pt 1):1505–1519.
- , , . Patient‐centredness in chronic illness: what is it and does it matter? Patient Educ Couns. 2003;51(3):197–206.
- , , , et al. Merging cultures: palliative care specialists in the medical intensive care unit. Crit Care Med. 2006;34(11 suppl):S388–S393.
- , , , et al. Improving inpatients' identification of their doctors: use of FACE cards. Jt Comm J Qual Patient Saf. 2009;35(12):613–619.
- , , . Interprofessional collaboration: effects of practice‐based interventions on professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2009;(3):CD000072.
- , , , et al. Identifying and overcoming the barriers to bedside rounds: a multicenter qualitative study. Acad Med. 2014;89(2):326–334.
- , , , , . The effect of bedside case presentations on patients' perceptions of their medical care. N Engl J Med. 1997;336(16):1150–1155.
- , , , . Patient‐centered interprofessional collaborative care: factors associated with bedside interprofessional rounds. J Gen Intern Med. 2014;29(7):1040–1047.
- , , , et al. Attending rounds in the current era: what is and is not happening. JAMA Intern Med. 2013;173(12):1084–1089.
- , . What the evidence shows about patient activation: better health outcomes and care experiences; fewer data on costs. Health Aff (Millwood). 2013;32(2):207–214.
- , , , , . When patient activation levels change, health outcomes and costs change, too. Health Aff Proj Hope. 2015;34(3):431–437.
- , , , et al. Evidence‐based guidelines for teaching patient‐centered interviewing. Patient Educ Couns. 2000;39(1):27–36.
- , , . Meta‐analysis of correlates of provider behavior in medical encounters. Med Care. 1988;26(7):657–675.
- , . Characteristics of potential plaintiffs in malpractice litigation. Ann Intern Med. 1994;120(9):792–798.
- , , , , . Bedside shift reports: what does the evidence say? J Nurs Adm. 2014;44(10):541–545.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- , , , et al. Continuity of care and the risk of preventable hospitalization in older adults. JAMA Intern Med. 2013;173(20):1879–1885.
- Agency for Healthcare Research and Quality. HCUPnet: a tool for identifying, tracking, and analyzing national hospital statistics. Available at: http://hcupnet.ahrq.gov/HCUPnet.jsp?Id=82B37DA366A36BAD6(8):438–444.
- , , , , , . Hospitalist handoffs: a systematic review and task force recommendations. J Hosp Med. 2009;4(7):433–440.
- , , , , . The impact of fragmentation of hospitalist care on length of stay. J Hosp Med. 2010;5(6):335–338.
- , , , et al. The impact of hospitalist discontinuity on hospital cost, readmissions, and patient satisfaction. J Gen Intern Med. 2014;29(7):1004–1008.
- , , , et al. The effect of hospitalist discontinuity on adverse events. J Hosp Med. 2015;10(3):147–151.
- Agency for Healthcare Research and Quality. HCAHPS Fact Sheet. CAHPS Hospital Survey August 2013. Available at: http://www.hcahpsonline.org/files/August_2013_HCAHPS_Fact_Sheet3.pdf. Accessed February 2, 2015.
- , , , et al. A conceptual framework for studying the safety of transitions in emergency care. In: Henriksen K, Battles JB, Marks ES, eds. Advances in Patient Safety: From Research to Implementation. Rockville, MD: Agency for Healthcare Research and Quality; 2005:309–321. Concepts and Methodology; vol 2. Available at: http://www.ncbi.nlm.nih.gov/books/NBK20522. Accessed January 15, 2015.
- , , , et al. Transitions of care consensus policy statement American College of Physicians‐Society of General Internal Medicine‐Society of Hospital Medicine‐American Geriatrics Society‐American College of Emergency Physicians‐Society of Academic Emergency Medicine. J Gen Intern Med. 2009;24(8):971–976.
- , . Grounded theory in medical education research: AMEE guide no. 70. Med Teach. 2012;34(10):850–861.
- . A purposeful approach to the constant comparative method in the analysis of qualitative interviews. Qual Quant. 2002;36(4):391–409.
- . The significance of saturation. Qual Health Res. 1995;5(2):147–149.
- , , , , . Standards for reporting qualitative research: a synthesis of recommendations. Acad Med. 2014;89(9):1245–1251.
- , , . Consolidated criteria for reporting qualitative research (COREQ): a 32‐item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–357.
- , , , . Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists. J Hosp Med. 2007;2(5):314–323.
- The Joint Commission. Hot Topics in Healthcare, Issue 2. Transitions of care: the need for collaboration across entire care continuum. Available at: http://www.jointcommission.org/assets/1/6/TOC_Hot_Topics.pdf. Accessed April 9, 2015.
- , . Physician communication and patient adherence to treatment: a meta‐analysis. Med Care. 2009;47(8):826–834.
- , . Impediments to adherence to post myocardial infarction medications. Curr Cardiol Rep. 2013;15(1):322.
- , , , , . Medical specialists' patient‐centered communication and patient‐reported outcomes. Med Care. 2007;45(4):330–339.
- , , , . Does doctor‐patient communication affect patient satisfaction with hospital care? Results of an analysis with a novel instrumental variable. Health Serv Res. 2008;43(5 pt 1):1505–1519.
- , , . Patient‐centredness in chronic illness: what is it and does it matter? Patient Educ Couns. 2003;51(3):197–206.
- , , , et al. Merging cultures: palliative care specialists in the medical intensive care unit. Crit Care Med. 2006;34(11 suppl):S388–S393.
- , , , et al. Improving inpatients' identification of their doctors: use of FACE cards. Jt Comm J Qual Patient Saf. 2009;35(12):613–619.
- , , . Interprofessional collaboration: effects of practice‐based interventions on professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2009;(3):CD000072.
- , , , et al. Identifying and overcoming the barriers to bedside rounds: a multicenter qualitative study. Acad Med. 2014;89(2):326–334.
- , , , , . The effect of bedside case presentations on patients' perceptions of their medical care. N Engl J Med. 1997;336(16):1150–1155.
- , , , . Patient‐centered interprofessional collaborative care: factors associated with bedside interprofessional rounds. J Gen Intern Med. 2014;29(7):1040–1047.
- , , , et al. Attending rounds in the current era: what is and is not happening. JAMA Intern Med. 2013;173(12):1084–1089.
- , . What the evidence shows about patient activation: better health outcomes and care experiences; fewer data on costs. Health Aff (Millwood). 2013;32(2):207–214.
- , , , , . When patient activation levels change, health outcomes and costs change, too. Health Aff Proj Hope. 2015;34(3):431–437.
- , , , et al. Evidence‐based guidelines for teaching patient‐centered interviewing. Patient Educ Couns. 2000;39(1):27–36.
- , , . Meta‐analysis of correlates of provider behavior in medical encounters. Med Care. 1988;26(7):657–675.
- , . Characteristics of potential plaintiffs in malpractice litigation. Ann Intern Med. 1994;120(9):792–798.
- , , , , . Bedside shift reports: what does the evidence say? J Nurs Adm. 2014;44(10):541–545.
CAR T-cell start-up launched
Dr. Siddhartha Mukherjee has partnered with Puretech Health to launch a new biotechnology and immuno-oncology company to broaden the use of chimeric antigen receptor (CAR) T-cell therapy. Dr. Mukherjee, a Columbia University researcher, hematologist, oncologist, and Pulitzer Prize–winning author of “The Emperor of All Maladies: A Biography of Cancer,” (New York: Scribner, a division of Simon & Schuster, 2011) is licensing his CAR T-cell technology to the joint venture, called Vor BioPharma.
Vor BioPharma will focus on advancing and expanding CAR T-cell therapy, a relatively new cancer treatment where T cells are first collected from a patient’s blood and then genetically engineered to produce CAR proteins on their surface. The CAR proteins are designed to bind specific antigens found on the patient’s cancer cells. These genetically engineered T cells are grown in a laboratory and then infused into the patient. As of now, CAR T-cell therapy is primarily used to treat B-cell leukemias and other chronic lymphocytic leukemia.
“We continue to make great strides in developing new ways to treat cancer using the body’s immune system,” said Dr. Mukherjee in a written statement announcing the partnership. “The positive clinical response researchers have achieved with CAR T-cell therapies in B-cell leukemias has led to great interest within the oncology community and is something we hope to achieve in other cancers over time,” he said.
“CAR T-cell therapies have shown remarkable progress in the clinic, yet their applicability beyond a small subset of cancers is currently very limited,” said Dr. Sanjiv Sam Gambhir of Stanford University and a member of the Vor Scientific Advisory Board. “This technology seeks to address bottlenecks that prevent CAR T-cell therapy from becoming more broadly useful in treating cancers outside of B-cell cancers.”
Other Vor BioPharma employees and Scientific Advisory Board members include Dr. Joseph Bolen, former President and Chief Scientific Officer of Moderna Therapeutics; Dr. Dan Littman of the Howard Hughes Medical Institute; and Dr. Derrick Rossi of Harvard University.
On Twitter @jess_craig94
Dr. Siddhartha Mukherjee has partnered with Puretech Health to launch a new biotechnology and immuno-oncology company to broaden the use of chimeric antigen receptor (CAR) T-cell therapy. Dr. Mukherjee, a Columbia University researcher, hematologist, oncologist, and Pulitzer Prize–winning author of “The Emperor of All Maladies: A Biography of Cancer,” (New York: Scribner, a division of Simon & Schuster, 2011) is licensing his CAR T-cell technology to the joint venture, called Vor BioPharma.
Vor BioPharma will focus on advancing and expanding CAR T-cell therapy, a relatively new cancer treatment where T cells are first collected from a patient’s blood and then genetically engineered to produce CAR proteins on their surface. The CAR proteins are designed to bind specific antigens found on the patient’s cancer cells. These genetically engineered T cells are grown in a laboratory and then infused into the patient. As of now, CAR T-cell therapy is primarily used to treat B-cell leukemias and other chronic lymphocytic leukemia.
“We continue to make great strides in developing new ways to treat cancer using the body’s immune system,” said Dr. Mukherjee in a written statement announcing the partnership. “The positive clinical response researchers have achieved with CAR T-cell therapies in B-cell leukemias has led to great interest within the oncology community and is something we hope to achieve in other cancers over time,” he said.
“CAR T-cell therapies have shown remarkable progress in the clinic, yet their applicability beyond a small subset of cancers is currently very limited,” said Dr. Sanjiv Sam Gambhir of Stanford University and a member of the Vor Scientific Advisory Board. “This technology seeks to address bottlenecks that prevent CAR T-cell therapy from becoming more broadly useful in treating cancers outside of B-cell cancers.”
Other Vor BioPharma employees and Scientific Advisory Board members include Dr. Joseph Bolen, former President and Chief Scientific Officer of Moderna Therapeutics; Dr. Dan Littman of the Howard Hughes Medical Institute; and Dr. Derrick Rossi of Harvard University.
On Twitter @jess_craig94
Dr. Siddhartha Mukherjee has partnered with Puretech Health to launch a new biotechnology and immuno-oncology company to broaden the use of chimeric antigen receptor (CAR) T-cell therapy. Dr. Mukherjee, a Columbia University researcher, hematologist, oncologist, and Pulitzer Prize–winning author of “The Emperor of All Maladies: A Biography of Cancer,” (New York: Scribner, a division of Simon & Schuster, 2011) is licensing his CAR T-cell technology to the joint venture, called Vor BioPharma.
Vor BioPharma will focus on advancing and expanding CAR T-cell therapy, a relatively new cancer treatment where T cells are first collected from a patient’s blood and then genetically engineered to produce CAR proteins on their surface. The CAR proteins are designed to bind specific antigens found on the patient’s cancer cells. These genetically engineered T cells are grown in a laboratory and then infused into the patient. As of now, CAR T-cell therapy is primarily used to treat B-cell leukemias and other chronic lymphocytic leukemia.
“We continue to make great strides in developing new ways to treat cancer using the body’s immune system,” said Dr. Mukherjee in a written statement announcing the partnership. “The positive clinical response researchers have achieved with CAR T-cell therapies in B-cell leukemias has led to great interest within the oncology community and is something we hope to achieve in other cancers over time,” he said.
“CAR T-cell therapies have shown remarkable progress in the clinic, yet their applicability beyond a small subset of cancers is currently very limited,” said Dr. Sanjiv Sam Gambhir of Stanford University and a member of the Vor Scientific Advisory Board. “This technology seeks to address bottlenecks that prevent CAR T-cell therapy from becoming more broadly useful in treating cancers outside of B-cell cancers.”
Other Vor BioPharma employees and Scientific Advisory Board members include Dr. Joseph Bolen, former President and Chief Scientific Officer of Moderna Therapeutics; Dr. Dan Littman of the Howard Hughes Medical Institute; and Dr. Derrick Rossi of Harvard University.
On Twitter @jess_craig94
Exercise is protective but underutilized in atrial fib patients
CHICAGO – Efforts to encourage even modest amounts of physical activity in sedentary patients with atrial fibrillation are likely to pay off in reduced risks of cardiovascular and all-cause mortality, according to a report from the EurObservational Research Program Pilot Survey on Atrial Fibrillation General Registry.
“Clearly we would recommend regular physical activity for patients with atrial fibrillation on the basis of the mortality benefit we see in the registry. If we give patients with atrial fibrillation oral anticoagulation, they are protected against stroke risk, but clearly they are still dying a lot,” Dr. Marco Proietti said at the annual meeting of the American College of Cardiology.
He presented 1-year follow-up data on 2,442 “real world” patients enrolled in the nine-country, observational, prospective registry, known as EORP-AF, shortly after being diagnosed with AF. One of the goals of EORP-AF is to learn whether physical exercise protects against cardiovascular events and all-cause mortality in AF patients, as has been well established in the general population and in patients at high cardiovascular risk.
One striking finding was that nearly 40% of patients in EORP-AF reported engaging in no physical activity, defined for study purposes as zero to less than 3 hours of physical activity per week for less than 2 years.
The other three categories employed by investigators were “occasional,” meaning less than 3 hours per week but for 2 years or more; “regular,” defined as at least 3 hours weekly for at least 2 years; and “intense,” which required more than 7 hours of physical activity per week for at least 2 years. Levels of cardiovascular and stroke risk factors decreased progressively with increasing levels of physical activity. Only 5% of the AF patients met the ‘intense’ standard, noted Dr. Proietti of the University of Birmingham (England).
The 1-year cardiovascular mortality rate approached 6% in the no physical activity group and hovered around 1% in the other three groups. The 1-year all-cause mortality rate exceeded 12% in the no-exercise group, was 4%% in the occasional exercisers, and 1%-2% in the groups reporting regular or intense physical activity.
The 1-year composite endpoint of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% of the sedentary patients, a rate two-to-three times higher than in the others.
Updated outcomes are to be reported from the EORP-AF pilot registry after 2 and 3 years of follow-up. Meanwhile, on the basis of the success of the pilot registry, more than 10,000 patients with AF have been enrolled in the EORP-AF main registry, according to Dr. Proietti.
A study limitation, he conceded, is that the registry includes no objective measure of physical capacity, such as METS.
Session co-chair Dr. Brian Olshansky, emeritus professor of internal medicine at the University of Iowa, Iowa City, observed that the registry data raise a classic chicken-versus-egg issue: Do the sedentary patients do worse because they’re inactive, or are they inactive because they are sicker and hence have worse outcomes?
Dr. Proietti said the registry data provide some support for the latter idea, since the no-physical-activity group had higher prevalences of coronary artery disease and heart failure.
Dr. Olshansky raised another point: “It’s interesting to me that there’s a whole bunch of literature showing that elite endurance athletes – bike racers, cross country skiers – have a very high incidence of atrial fibrillation. It seems to be either an inflammatory or an autonomic issue.”
Dr. Proietti replied that he’s familiar with that extensive literature, but the EORP-AF data through 1 year don’t provide validation. While the intense physical activity group tended to have more symptomatic AF than the other groups, they were no more likely to show progression from paroxysmal to permanent AF. The much larger main registry now underway may be able to better clarify the relationship between physical activity and incidence and progression of AF, including the possibility of a U-shaped dose-response curve.
The EORP-AF registry is supported by the European Society of Cardiology. Dr. Proietti reported having no financial conflicts of interest.
CHICAGO – Efforts to encourage even modest amounts of physical activity in sedentary patients with atrial fibrillation are likely to pay off in reduced risks of cardiovascular and all-cause mortality, according to a report from the EurObservational Research Program Pilot Survey on Atrial Fibrillation General Registry.
“Clearly we would recommend regular physical activity for patients with atrial fibrillation on the basis of the mortality benefit we see in the registry. If we give patients with atrial fibrillation oral anticoagulation, they are protected against stroke risk, but clearly they are still dying a lot,” Dr. Marco Proietti said at the annual meeting of the American College of Cardiology.
He presented 1-year follow-up data on 2,442 “real world” patients enrolled in the nine-country, observational, prospective registry, known as EORP-AF, shortly after being diagnosed with AF. One of the goals of EORP-AF is to learn whether physical exercise protects against cardiovascular events and all-cause mortality in AF patients, as has been well established in the general population and in patients at high cardiovascular risk.
One striking finding was that nearly 40% of patients in EORP-AF reported engaging in no physical activity, defined for study purposes as zero to less than 3 hours of physical activity per week for less than 2 years.
The other three categories employed by investigators were “occasional,” meaning less than 3 hours per week but for 2 years or more; “regular,” defined as at least 3 hours weekly for at least 2 years; and “intense,” which required more than 7 hours of physical activity per week for at least 2 years. Levels of cardiovascular and stroke risk factors decreased progressively with increasing levels of physical activity. Only 5% of the AF patients met the ‘intense’ standard, noted Dr. Proietti of the University of Birmingham (England).
The 1-year cardiovascular mortality rate approached 6% in the no physical activity group and hovered around 1% in the other three groups. The 1-year all-cause mortality rate exceeded 12% in the no-exercise group, was 4%% in the occasional exercisers, and 1%-2% in the groups reporting regular or intense physical activity.
The 1-year composite endpoint of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% of the sedentary patients, a rate two-to-three times higher than in the others.
Updated outcomes are to be reported from the EORP-AF pilot registry after 2 and 3 years of follow-up. Meanwhile, on the basis of the success of the pilot registry, more than 10,000 patients with AF have been enrolled in the EORP-AF main registry, according to Dr. Proietti.
A study limitation, he conceded, is that the registry includes no objective measure of physical capacity, such as METS.
Session co-chair Dr. Brian Olshansky, emeritus professor of internal medicine at the University of Iowa, Iowa City, observed that the registry data raise a classic chicken-versus-egg issue: Do the sedentary patients do worse because they’re inactive, or are they inactive because they are sicker and hence have worse outcomes?
Dr. Proietti said the registry data provide some support for the latter idea, since the no-physical-activity group had higher prevalences of coronary artery disease and heart failure.
Dr. Olshansky raised another point: “It’s interesting to me that there’s a whole bunch of literature showing that elite endurance athletes – bike racers, cross country skiers – have a very high incidence of atrial fibrillation. It seems to be either an inflammatory or an autonomic issue.”
Dr. Proietti replied that he’s familiar with that extensive literature, but the EORP-AF data through 1 year don’t provide validation. While the intense physical activity group tended to have more symptomatic AF than the other groups, they were no more likely to show progression from paroxysmal to permanent AF. The much larger main registry now underway may be able to better clarify the relationship between physical activity and incidence and progression of AF, including the possibility of a U-shaped dose-response curve.
The EORP-AF registry is supported by the European Society of Cardiology. Dr. Proietti reported having no financial conflicts of interest.
CHICAGO – Efforts to encourage even modest amounts of physical activity in sedentary patients with atrial fibrillation are likely to pay off in reduced risks of cardiovascular and all-cause mortality, according to a report from the EurObservational Research Program Pilot Survey on Atrial Fibrillation General Registry.
“Clearly we would recommend regular physical activity for patients with atrial fibrillation on the basis of the mortality benefit we see in the registry. If we give patients with atrial fibrillation oral anticoagulation, they are protected against stroke risk, but clearly they are still dying a lot,” Dr. Marco Proietti said at the annual meeting of the American College of Cardiology.
He presented 1-year follow-up data on 2,442 “real world” patients enrolled in the nine-country, observational, prospective registry, known as EORP-AF, shortly after being diagnosed with AF. One of the goals of EORP-AF is to learn whether physical exercise protects against cardiovascular events and all-cause mortality in AF patients, as has been well established in the general population and in patients at high cardiovascular risk.
One striking finding was that nearly 40% of patients in EORP-AF reported engaging in no physical activity, defined for study purposes as zero to less than 3 hours of physical activity per week for less than 2 years.
The other three categories employed by investigators were “occasional,” meaning less than 3 hours per week but for 2 years or more; “regular,” defined as at least 3 hours weekly for at least 2 years; and “intense,” which required more than 7 hours of physical activity per week for at least 2 years. Levels of cardiovascular and stroke risk factors decreased progressively with increasing levels of physical activity. Only 5% of the AF patients met the ‘intense’ standard, noted Dr. Proietti of the University of Birmingham (England).
The 1-year cardiovascular mortality rate approached 6% in the no physical activity group and hovered around 1% in the other three groups. The 1-year all-cause mortality rate exceeded 12% in the no-exercise group, was 4%% in the occasional exercisers, and 1%-2% in the groups reporting regular or intense physical activity.
The 1-year composite endpoint of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% of the sedentary patients, a rate two-to-three times higher than in the others.
Updated outcomes are to be reported from the EORP-AF pilot registry after 2 and 3 years of follow-up. Meanwhile, on the basis of the success of the pilot registry, more than 10,000 patients with AF have been enrolled in the EORP-AF main registry, according to Dr. Proietti.
A study limitation, he conceded, is that the registry includes no objective measure of physical capacity, such as METS.
Session co-chair Dr. Brian Olshansky, emeritus professor of internal medicine at the University of Iowa, Iowa City, observed that the registry data raise a classic chicken-versus-egg issue: Do the sedentary patients do worse because they’re inactive, or are they inactive because they are sicker and hence have worse outcomes?
Dr. Proietti said the registry data provide some support for the latter idea, since the no-physical-activity group had higher prevalences of coronary artery disease and heart failure.
Dr. Olshansky raised another point: “It’s interesting to me that there’s a whole bunch of literature showing that elite endurance athletes – bike racers, cross country skiers – have a very high incidence of atrial fibrillation. It seems to be either an inflammatory or an autonomic issue.”
Dr. Proietti replied that he’s familiar with that extensive literature, but the EORP-AF data through 1 year don’t provide validation. While the intense physical activity group tended to have more symptomatic AF than the other groups, they were no more likely to show progression from paroxysmal to permanent AF. The much larger main registry now underway may be able to better clarify the relationship between physical activity and incidence and progression of AF, including the possibility of a U-shaped dose-response curve.
The EORP-AF registry is supported by the European Society of Cardiology. Dr. Proietti reported having no financial conflicts of interest.
AT ACC 16
Key clinical point: Atrial fibrillation patients who report engaging in even occasional physical activity have a markedly lower risk of all-cause mortality than those who are sedentary.
Major finding: The 1-year composite outcome of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% in patients with atrial fibrillation who were sedentary, a rate two to three times greater than in those who engaged in various amounts of physical activity.
Data source: An analysis of 1-year outcomes in 2,442 patients with AF enrolled in the prospective, observational EORP-AF pilot registry.
Disclosures: The EORP-AF registry is supported by the European Society of Cardiology. The presenter reported having no financial conflicts of interest.
Optimal timing of CRC postop colonoscopy studied
LOS ANGELES – The detection rate of significant polyps was highest for the first postoperative surveillance colonoscopies performed at 1 year following curative resection for colorectal cancer, results from a single-center study demonstrated.
“There’s no consensus on when to perform the first surveillance colonoscopy post curative resection for colorectal cancer,” lead study author Dr. Noura Alhassan said at the annual meeting of the American Society of Colon and Rectal Surgeons. For example, the American Society of Colon and Rectal Surgeons and National Carcinoma Comprehensive Network guidelines recommend a colonoscopy at 1 year, while the Canadian Association of Gastroenterology recommends surveillance at 3 years postoperatively.
In an effort to determine the optimal timing of the first surveillance colonoscopy following curative colorectal carcinoma resection, Dr. Alhassan and her associates retrospectively reviewed the charts of all patients who underwent colorectal resection from 2007 to 2012 at Jewish General Hospital, a tertiary care center affiliated with McGill University, Montreal. The study included patients who had a complete preoperative colonoscopy, those who had a complete postoperative colonoscopy performed by one of the Jewish General Hospital colorectal surgeons, and those who had colorectal cancer resection with curative intent. Excluded from the study were patients with stage IV colorectal cancer, those with a prior history of colorectal cancer, those who underwent total abdominal colectomies or proctocolectomies, those who underwent local excision, and those with familial cancer syndromes and inflammatory bowel disease.
Dr. Alhassan, a fourth-year resident in the division of general surgery at McGill University, said that the researchers classified the colonoscopic findings as normal, nonsignificant polyps, significant polyps, and recurrence. Significant polyps consisted of adenomas 1 cm or greater in size, villous or tubulovillous adenoma, adenoma with high-grade dysplasia, three or more adenomas, or sessile serrated polyps at least 1 cm in size or with dysplasia. Of the 857 colorectal resections performed during the study period, 181 met inclusion criteria. The tumor stage was evenly distributed among study participants and 57% of the resections were colon operations, while the remaining 43% were proctectomies.
The preoperative colonoscopy was done by one of the Jewish General Hospital gastroenterologists 43% of the time, by one of the Jewish General Hospital colorectal surgeons 41% of the time, and by an outside hospital 16% of the time. The median time to postoperative colonoscopy was 421 days (1.1 years). Specifically, 25.90% of patients underwent their first surveillance colonoscopy in the first postoperative year, 48.10% in the second year, 14.40% in the third year, 8.5% in the fourth year, and 2.7% in the fifth year.
Dr. Alhassan reported that the all-polyp detection rate was 30.1%; 21.3% were detected in postoperative year 1, 33.3% in year 2, and 34.6% in year 3.
The overall significant polyp detection rate was 10.5%, but the detection rate was 12.8% in postoperative year 1, 8% in postoperative year 2, and 7.7% in postoperative year 3. There were two anastomotic recurrences: one in year 1 (2.1%) and one in year 3 (3.8%).
On univariate analysis, factors associated with significant polyp detection were male gender, poor bowel preparation on preoperative colonoscopy, and concomitant use of metformin, while having stage III disease was associated with a lower significant polyp detection rate.
On multivariate analysis only male gender was associated with a higher significant polyp detection rate, while stage III disease was associated with a lower significant polyp detection rate.
“Significant polyp detection rate of 12.8% at postoperative year 1 justifies surveillance colonoscopy at 1 year post curative colon cancer resection,” Dr. Alhassan concluded. She reported having no financial disclosures.
LOS ANGELES – The detection rate of significant polyps was highest for the first postoperative surveillance colonoscopies performed at 1 year following curative resection for colorectal cancer, results from a single-center study demonstrated.
“There’s no consensus on when to perform the first surveillance colonoscopy post curative resection for colorectal cancer,” lead study author Dr. Noura Alhassan said at the annual meeting of the American Society of Colon and Rectal Surgeons. For example, the American Society of Colon and Rectal Surgeons and National Carcinoma Comprehensive Network guidelines recommend a colonoscopy at 1 year, while the Canadian Association of Gastroenterology recommends surveillance at 3 years postoperatively.
In an effort to determine the optimal timing of the first surveillance colonoscopy following curative colorectal carcinoma resection, Dr. Alhassan and her associates retrospectively reviewed the charts of all patients who underwent colorectal resection from 2007 to 2012 at Jewish General Hospital, a tertiary care center affiliated with McGill University, Montreal. The study included patients who had a complete preoperative colonoscopy, those who had a complete postoperative colonoscopy performed by one of the Jewish General Hospital colorectal surgeons, and those who had colorectal cancer resection with curative intent. Excluded from the study were patients with stage IV colorectal cancer, those with a prior history of colorectal cancer, those who underwent total abdominal colectomies or proctocolectomies, those who underwent local excision, and those with familial cancer syndromes and inflammatory bowel disease.
Dr. Alhassan, a fourth-year resident in the division of general surgery at McGill University, said that the researchers classified the colonoscopic findings as normal, nonsignificant polyps, significant polyps, and recurrence. Significant polyps consisted of adenomas 1 cm or greater in size, villous or tubulovillous adenoma, adenoma with high-grade dysplasia, three or more adenomas, or sessile serrated polyps at least 1 cm in size or with dysplasia. Of the 857 colorectal resections performed during the study period, 181 met inclusion criteria. The tumor stage was evenly distributed among study participants and 57% of the resections were colon operations, while the remaining 43% were proctectomies.
The preoperative colonoscopy was done by one of the Jewish General Hospital gastroenterologists 43% of the time, by one of the Jewish General Hospital colorectal surgeons 41% of the time, and by an outside hospital 16% of the time. The median time to postoperative colonoscopy was 421 days (1.1 years). Specifically, 25.90% of patients underwent their first surveillance colonoscopy in the first postoperative year, 48.10% in the second year, 14.40% in the third year, 8.5% in the fourth year, and 2.7% in the fifth year.
Dr. Alhassan reported that the all-polyp detection rate was 30.1%; 21.3% were detected in postoperative year 1, 33.3% in year 2, and 34.6% in year 3.
The overall significant polyp detection rate was 10.5%, but the detection rate was 12.8% in postoperative year 1, 8% in postoperative year 2, and 7.7% in postoperative year 3. There were two anastomotic recurrences: one in year 1 (2.1%) and one in year 3 (3.8%).
On univariate analysis, factors associated with significant polyp detection were male gender, poor bowel preparation on preoperative colonoscopy, and concomitant use of metformin, while having stage III disease was associated with a lower significant polyp detection rate.
On multivariate analysis only male gender was associated with a higher significant polyp detection rate, while stage III disease was associated with a lower significant polyp detection rate.
“Significant polyp detection rate of 12.8% at postoperative year 1 justifies surveillance colonoscopy at 1 year post curative colon cancer resection,” Dr. Alhassan concluded. She reported having no financial disclosures.
LOS ANGELES – The detection rate of significant polyps was highest for the first postoperative surveillance colonoscopies performed at 1 year following curative resection for colorectal cancer, results from a single-center study demonstrated.
“There’s no consensus on when to perform the first surveillance colonoscopy post curative resection for colorectal cancer,” lead study author Dr. Noura Alhassan said at the annual meeting of the American Society of Colon and Rectal Surgeons. For example, the American Society of Colon and Rectal Surgeons and National Carcinoma Comprehensive Network guidelines recommend a colonoscopy at 1 year, while the Canadian Association of Gastroenterology recommends surveillance at 3 years postoperatively.
In an effort to determine the optimal timing of the first surveillance colonoscopy following curative colorectal carcinoma resection, Dr. Alhassan and her associates retrospectively reviewed the charts of all patients who underwent colorectal resection from 2007 to 2012 at Jewish General Hospital, a tertiary care center affiliated with McGill University, Montreal. The study included patients who had a complete preoperative colonoscopy, those who had a complete postoperative colonoscopy performed by one of the Jewish General Hospital colorectal surgeons, and those who had colorectal cancer resection with curative intent. Excluded from the study were patients with stage IV colorectal cancer, those with a prior history of colorectal cancer, those who underwent total abdominal colectomies or proctocolectomies, those who underwent local excision, and those with familial cancer syndromes and inflammatory bowel disease.
Dr. Alhassan, a fourth-year resident in the division of general surgery at McGill University, said that the researchers classified the colonoscopic findings as normal, nonsignificant polyps, significant polyps, and recurrence. Significant polyps consisted of adenomas 1 cm or greater in size, villous or tubulovillous adenoma, adenoma with high-grade dysplasia, three or more adenomas, or sessile serrated polyps at least 1 cm in size or with dysplasia. Of the 857 colorectal resections performed during the study period, 181 met inclusion criteria. The tumor stage was evenly distributed among study participants and 57% of the resections were colon operations, while the remaining 43% were proctectomies.
The preoperative colonoscopy was done by one of the Jewish General Hospital gastroenterologists 43% of the time, by one of the Jewish General Hospital colorectal surgeons 41% of the time, and by an outside hospital 16% of the time. The median time to postoperative colonoscopy was 421 days (1.1 years). Specifically, 25.90% of patients underwent their first surveillance colonoscopy in the first postoperative year, 48.10% in the second year, 14.40% in the third year, 8.5% in the fourth year, and 2.7% in the fifth year.
Dr. Alhassan reported that the all-polyp detection rate was 30.1%; 21.3% were detected in postoperative year 1, 33.3% in year 2, and 34.6% in year 3.
The overall significant polyp detection rate was 10.5%, but the detection rate was 12.8% in postoperative year 1, 8% in postoperative year 2, and 7.7% in postoperative year 3. There were two anastomotic recurrences: one in year 1 (2.1%) and one in year 3 (3.8%).
On univariate analysis, factors associated with significant polyp detection were male gender, poor bowel preparation on preoperative colonoscopy, and concomitant use of metformin, while having stage III disease was associated with a lower significant polyp detection rate.
On multivariate analysis only male gender was associated with a higher significant polyp detection rate, while stage III disease was associated with a lower significant polyp detection rate.
“Significant polyp detection rate of 12.8% at postoperative year 1 justifies surveillance colonoscopy at 1 year post curative colon cancer resection,” Dr. Alhassan concluded. She reported having no financial disclosures.
AT THE ASCRS ANNUAL MEETING
Key clinical point: The highest proportion of significant polyps on surveillance colonoscopy after curative resection was detected in postoperative year 1.
Major finding: The overall significant polyp detection rate was 10.5%, but 12.8% were detected in postoperative year 1, 8% in postoperative year 2, and 7.7% in postoperative year 3.
Data source: A retrospective study of 181 patients who underwent colorectal resection from 2007 to 2012 at Jewish General Hospital, Montreal.
Disclosures: Dr. Alhassan reported having no financial disclosures.
Anticoagulation therapy after VT ablation yields fewer thrombotic events
San Francisco – Anticoagulation therapy is probably a good idea after ventricular tachycardia ablation in patients with risk factors or stroke, even if they don’t have atrial fibrillation, according to investigators from the University of Kansas Medical Center in Kansas City.
The advice comes from a review of 2,235 ventricular tachycardia (VT) ablation cases from the university and other members of the International VT Ablation Center Collaborative; about a quarter of the patients (604) were prescribed oral anticoagulation therapy at baseline and at discharge, nearly all for atrial fibrillation (AF) and most with warfarin. Over the next year, just 0.3% (2) had a subsequent thromboembolic complication, one of which was an ischemic stroke.
The remaining patients (1,631) did not have a diagnosis of AF and were not on anticoagulants at baseline or after discharge. They were more likely to have New York Heart Association class I or II heart failure and higher ejection fractions, and to otherwise be in better shape compared with the patients who received anticoagulation therapy. Even so, within a year, 1.3% (21) had a thromboembolic event, almost half of which were ischemic strokes, a substantial increase in relative risk (P = .05).
Maybe those patients had undiagnosed AF at baseline, or perhaps a clot formed over the ablation scar, Dr. Rizwan Afzal said at the annual scientific sessions of the Heart Rhythm Society. Regardless, “this observation has changed our practice. If VT ablation patients have low ejection fractions, if they’re elderly, or have other risk factors for stroke, we put them on blood thinners [afterward] “even if they don’t have atrial fibrillation. We are not sure how long they should be on anticoagulation [therapy] to counteract the increased risk of stroke,” but probably at least for a few weeks, he said.
Dr. Afzal and his colleagues generally opt for warfarin; the use is off label for newer oral anticoagulants, and a tough sell to insurance companies.
There were no predictors of increased thromboembolic risk in the group that was not on anticoagulation therapy. During follow-up, about 2.2% (13) of patients on anticoagulation therapy had bleeding complications, including one intracranial hemorrhage, compared with 2.5% (41) of the patients not treated with an anticoagulant; most of them were on aspirin after the procedure, and the rest were on dual antiplatelet therapy (P = .7), reported Dr. Afzal, a cardiology fellow at the University of Kansas.
The median age of the study patients was 65 years, and 87% were men. In the group on anticoagulation therapy, the mean baseline left ventricular ejection fraction was 31%; 35% had prior cardiac surgery, 29% were on cardiac resynchronization therapy, and 44% had NYHA class III or IV heart failure. The mean baseline ejection fraction among patients who were not on anticoagulation therapy was 35%; 29% had prior heart surgery, 24% were on CRT, and 32.5% had NYHA class III or IV heart failure.
There was no industry funding for the work, and the investigators had no disclosures.
San Francisco – Anticoagulation therapy is probably a good idea after ventricular tachycardia ablation in patients with risk factors or stroke, even if they don’t have atrial fibrillation, according to investigators from the University of Kansas Medical Center in Kansas City.
The advice comes from a review of 2,235 ventricular tachycardia (VT) ablation cases from the university and other members of the International VT Ablation Center Collaborative; about a quarter of the patients (604) were prescribed oral anticoagulation therapy at baseline and at discharge, nearly all for atrial fibrillation (AF) and most with warfarin. Over the next year, just 0.3% (2) had a subsequent thromboembolic complication, one of which was an ischemic stroke.
The remaining patients (1,631) did not have a diagnosis of AF and were not on anticoagulants at baseline or after discharge. They were more likely to have New York Heart Association class I or II heart failure and higher ejection fractions, and to otherwise be in better shape compared with the patients who received anticoagulation therapy. Even so, within a year, 1.3% (21) had a thromboembolic event, almost half of which were ischemic strokes, a substantial increase in relative risk (P = .05).
Maybe those patients had undiagnosed AF at baseline, or perhaps a clot formed over the ablation scar, Dr. Rizwan Afzal said at the annual scientific sessions of the Heart Rhythm Society. Regardless, “this observation has changed our practice. If VT ablation patients have low ejection fractions, if they’re elderly, or have other risk factors for stroke, we put them on blood thinners [afterward] “even if they don’t have atrial fibrillation. We are not sure how long they should be on anticoagulation [therapy] to counteract the increased risk of stroke,” but probably at least for a few weeks, he said.
Dr. Afzal and his colleagues generally opt for warfarin; the use is off label for newer oral anticoagulants, and a tough sell to insurance companies.
There were no predictors of increased thromboembolic risk in the group that was not on anticoagulation therapy. During follow-up, about 2.2% (13) of patients on anticoagulation therapy had bleeding complications, including one intracranial hemorrhage, compared with 2.5% (41) of the patients not treated with an anticoagulant; most of them were on aspirin after the procedure, and the rest were on dual antiplatelet therapy (P = .7), reported Dr. Afzal, a cardiology fellow at the University of Kansas.
The median age of the study patients was 65 years, and 87% were men. In the group on anticoagulation therapy, the mean baseline left ventricular ejection fraction was 31%; 35% had prior cardiac surgery, 29% were on cardiac resynchronization therapy, and 44% had NYHA class III or IV heart failure. The mean baseline ejection fraction among patients who were not on anticoagulation therapy was 35%; 29% had prior heart surgery, 24% were on CRT, and 32.5% had NYHA class III or IV heart failure.
There was no industry funding for the work, and the investigators had no disclosures.
San Francisco – Anticoagulation therapy is probably a good idea after ventricular tachycardia ablation in patients with risk factors or stroke, even if they don’t have atrial fibrillation, according to investigators from the University of Kansas Medical Center in Kansas City.
The advice comes from a review of 2,235 ventricular tachycardia (VT) ablation cases from the university and other members of the International VT Ablation Center Collaborative; about a quarter of the patients (604) were prescribed oral anticoagulation therapy at baseline and at discharge, nearly all for atrial fibrillation (AF) and most with warfarin. Over the next year, just 0.3% (2) had a subsequent thromboembolic complication, one of which was an ischemic stroke.
The remaining patients (1,631) did not have a diagnosis of AF and were not on anticoagulants at baseline or after discharge. They were more likely to have New York Heart Association class I or II heart failure and higher ejection fractions, and to otherwise be in better shape compared with the patients who received anticoagulation therapy. Even so, within a year, 1.3% (21) had a thromboembolic event, almost half of which were ischemic strokes, a substantial increase in relative risk (P = .05).
Maybe those patients had undiagnosed AF at baseline, or perhaps a clot formed over the ablation scar, Dr. Rizwan Afzal said at the annual scientific sessions of the Heart Rhythm Society. Regardless, “this observation has changed our practice. If VT ablation patients have low ejection fractions, if they’re elderly, or have other risk factors for stroke, we put them on blood thinners [afterward] “even if they don’t have atrial fibrillation. We are not sure how long they should be on anticoagulation [therapy] to counteract the increased risk of stroke,” but probably at least for a few weeks, he said.
Dr. Afzal and his colleagues generally opt for warfarin; the use is off label for newer oral anticoagulants, and a tough sell to insurance companies.
There were no predictors of increased thromboembolic risk in the group that was not on anticoagulation therapy. During follow-up, about 2.2% (13) of patients on anticoagulation therapy had bleeding complications, including one intracranial hemorrhage, compared with 2.5% (41) of the patients not treated with an anticoagulant; most of them were on aspirin after the procedure, and the rest were on dual antiplatelet therapy (P = .7), reported Dr. Afzal, a cardiology fellow at the University of Kansas.
The median age of the study patients was 65 years, and 87% were men. In the group on anticoagulation therapy, the mean baseline left ventricular ejection fraction was 31%; 35% had prior cardiac surgery, 29% were on cardiac resynchronization therapy, and 44% had NYHA class III or IV heart failure. The mean baseline ejection fraction among patients who were not on anticoagulation therapy was 35%; 29% had prior heart surgery, 24% were on CRT, and 32.5% had NYHA class III or IV heart failure.
There was no industry funding for the work, and the investigators had no disclosures.
AT HEART RHYTHM 2016
Key clinical point: Anticoagulant therapy may be a good idea after ventricular tachycardia ablation in patients with risk factors for stroke, even if they don’t have atrial fibrillation.
Major finding: About 0.3% of patients on oral anticoagulant therapy after VT ablation had a thromboembolic event within a year, compared with 1.3% of those who were not on such therapy.
Data source: Review of 2,245 VT ablation cases.
Disclosures: There was no industry funding for the work, and the investigators had no disclosures.
Low vasculitis risk with TNF inhibitors
GLASGOW – Treatment with tumor necrosis factor (TNF) inhibitors for rheumatoid arthritis is associated with a low risk of vasculitis-like events, according to a large analysis of data from the United Kingdom.
Investigators using data from the British Society for Rheumatology Biologics Register for Rheumatoid Arthritis (BSRBR-RA) found that the crude incidence rate was 16 cases per 10,000 person-years among TNF-inhibitor users versus seven cases per 10,000 person-years among users of nonbiologic disease-modifying antirheumatic drugs (nbDMARDs) such as methotrexate and sulfasalazine.
Although the risk was slightly higher among anti-TNF than nbDMARD users, the propensity score fully adjusted hazard ratio for a first vasculitis-like event was 1.27, comparing the anti-TNF drugs with nbDMARDs, with a 95% confidence interval of 0.40-4.04.
“This is the first prospective observational study to systematically look at the risk of vasculitis-like events” in patients with RA treated with anti-TNF agents, Dr. Meghna Jani said at the British Society for Rheumatology annual conference.
Dr. Jani of the Arthritis Research UK Centre for Epidemiology at the University of Manchester (England) explained that the reason for looking at this topic was that vasculitis-like events had been reported in case series and single-center studies, but these prior reports were too small to be able to estimate exactly how big a problem this was.
“Anti-TNF agents are associated with the development of a number of autoantibodies, including antinuclear antibodies and antidrug antibodies, and ANCA [antineutrophil cytoplasmic antibody],” she observed.
“We know that a small proportion of these patients may then go on to develop autoimmune diseases, some independent of autoantibodies,” she added. The most common of these is vasculitis, including cutaneous vasculitis.
Vasculitis is a somewhat paradoxical adverse event, she noted, in that it has been associated with anti-TNF therapy, but these drugs can also be used to treat it.
Now in its 15th year, the BSRBR-RA is the largest ongoing cohort of patients treated with biologic agents for rheumatic disease and provides one of the best sources of data to examine the risk for vasculitis-like events Dr. Jani observed. The aims were to look at the respective risks as well as to see if there were any particular predictive factors.
The current analysis included more than 16,000 patients enrolled in the BSRBR-RA between 2001 and 2015, of whom 12,745 were newly started on an anti-TNF drug and 3,640 were receiving nbDMARDs and were also biologic naive. The mean age of patients in the two groups was 56 and 60 years, 76% and 72% were female, with a mean Disease Activity Score (DAS28) of 6.5 and 5.1 and median disease duration of 11 and 6 years, respectively.
After more than 52,428 person-years of exposure and a median of 5.1 years of follow-up, 81 vasculitis-like events occurred in the anti-TNF therapy group. Vasculitis-like events were attributed to treatment only if they had occurred within 90 days of starting the drug. Follow-up stopped after a first event; if there was a switch to another biologic drug; and at death, the last clinical follow-up, or the end of the analysis period (May 31, 2015).
In comparison, there were 20,635 person-years of exposure and 6.5 years’ follow-up in the nbDMARD group, with 14 vasculitis-like events reported during this time.
A sensitivity analysis was performed excluding patients who had nail-fold vasculitis at baseline, had vasculitis due to a possible secondary cause such as infection, and were taking any other medications associated with vasculitis-like events. Results showed a similar risk for a first vasculitis event between anti-TNF and nbDMARD users (aHR = 1.05; 95% CI, 0.32-3.45).
Looking at the risk of vasculitis events for individual anti-TNF drugs, there initially appeared to be a higher risk for patients taking infliximab (n = 3,292) and etanercept (n = 4,450) but not for those taking adalimumab (n = 4,312) versus nbDMARDs, with crude incidence rates of 10, 17, and 11 per 10,000 person-years; after adjustment, these differences were not significant (aHRs of 1.55, 1.72, and 0.77, respectively, with 95% CIs crossing 1.0). A crude rate for certolizumab could not be calculated as there were no vasculitis events reported but there were only 691 patients enrolled in the BSRBR-RA at the time of the analysis who had been exposed to the drug.
“The risk of the event was highest in the first year of treatment, followed by reduction over time,” Dr. Jani reported. “Reassuringly, up to two-thirds of patients in both cohorts had manifestations that were just limited to cutaneous involvement,” she said.
The most common systemic presentation was digital ischemia, affecting 14% of patients treated with anti-TNFs and 14% of those given nbDMARDs. Neurologic involvement was also seen in both groups of patients (7% vs. 7%), but new nail-fold vasculitis (17% vs. 0%), respiratory involvement (4% vs. 0%), associated thrombotic events (5% vs. 0%), and renal involvement (2.5% vs. 0%) were seen in TNF inhibitor-treated patients only.
Ten anti-TNF–treated patients and one nbDMARD-treated patient needed treatment for the vasculitis-like event, and three patients in the anti-TNF cohort died as a result of the event, all of whom had multisystem organ involvement and one of whom had cytoplasmic ANCA-positive vasculitis.
Treatment with methotrexate or sulfasalazine at baseline was associated with a lower risk for vasculitis-like events, while seropositive status, disease duration, DAS28, and HAQ scores were associated with an increased risk for such events.
The BSRBR-RA receives restricted income financial support from Abbvie, Amgen, Swedish Orphan Biovitram (SOBI), Merck, Pfizer, Roche, and UCB Pharma. Dr. Jani disclosed she has received honoraria from Pfizer, Abbvie, and UCB Pharma.
GLASGOW – Treatment with tumor necrosis factor (TNF) inhibitors for rheumatoid arthritis is associated with a low risk of vasculitis-like events, according to a large analysis of data from the United Kingdom.
Investigators using data from the British Society for Rheumatology Biologics Register for Rheumatoid Arthritis (BSRBR-RA) found that the crude incidence rate was 16 cases per 10,000 person-years among TNF-inhibitor users versus seven cases per 10,000 person-years among users of nonbiologic disease-modifying antirheumatic drugs (nbDMARDs) such as methotrexate and sulfasalazine.
Although the risk was slightly higher among anti-TNF than nbDMARD users, the propensity score fully adjusted hazard ratio for a first vasculitis-like event was 1.27, comparing the anti-TNF drugs with nbDMARDs, with a 95% confidence interval of 0.40-4.04.
“This is the first prospective observational study to systematically look at the risk of vasculitis-like events” in patients with RA treated with anti-TNF agents, Dr. Meghna Jani said at the British Society for Rheumatology annual conference.
Dr. Jani of the Arthritis Research UK Centre for Epidemiology at the University of Manchester (England) explained that the reason for looking at this topic was that vasculitis-like events had been reported in case series and single-center studies, but these prior reports were too small to be able to estimate exactly how big a problem this was.
“Anti-TNF agents are associated with the development of a number of autoantibodies, including antinuclear antibodies and antidrug antibodies, and ANCA [antineutrophil cytoplasmic antibody],” she observed.
“We know that a small proportion of these patients may then go on to develop autoimmune diseases, some independent of autoantibodies,” she added. The most common of these is vasculitis, including cutaneous vasculitis.
Vasculitis is a somewhat paradoxical adverse event, she noted, in that it has been associated with anti-TNF therapy, but these drugs can also be used to treat it.
Now in its 15th year, the BSRBR-RA is the largest ongoing cohort of patients treated with biologic agents for rheumatic disease and provides one of the best sources of data to examine the risk for vasculitis-like events Dr. Jani observed. The aims were to look at the respective risks as well as to see if there were any particular predictive factors.
The current analysis included more than 16,000 patients enrolled in the BSRBR-RA between 2001 and 2015, of whom 12,745 were newly started on an anti-TNF drug and 3,640 were receiving nbDMARDs and were also biologic naive. The mean age of patients in the two groups was 56 and 60 years, 76% and 72% were female, with a mean Disease Activity Score (DAS28) of 6.5 and 5.1 and median disease duration of 11 and 6 years, respectively.
After more than 52,428 person-years of exposure and a median of 5.1 years of follow-up, 81 vasculitis-like events occurred in the anti-TNF therapy group. Vasculitis-like events were attributed to treatment only if they had occurred within 90 days of starting the drug. Follow-up stopped after a first event; if there was a switch to another biologic drug; and at death, the last clinical follow-up, or the end of the analysis period (May 31, 2015).
In comparison, there were 20,635 person-years of exposure and 6.5 years’ follow-up in the nbDMARD group, with 14 vasculitis-like events reported during this time.
A sensitivity analysis was performed excluding patients who had nail-fold vasculitis at baseline, had vasculitis due to a possible secondary cause such as infection, and were taking any other medications associated with vasculitis-like events. Results showed a similar risk for a first vasculitis event between anti-TNF and nbDMARD users (aHR = 1.05; 95% CI, 0.32-3.45).
Looking at the risk of vasculitis events for individual anti-TNF drugs, there initially appeared to be a higher risk for patients taking infliximab (n = 3,292) and etanercept (n = 4,450) but not for those taking adalimumab (n = 4,312) versus nbDMARDs, with crude incidence rates of 10, 17, and 11 per 10,000 person-years; after adjustment, these differences were not significant (aHRs of 1.55, 1.72, and 0.77, respectively, with 95% CIs crossing 1.0). A crude rate for certolizumab could not be calculated as there were no vasculitis events reported but there were only 691 patients enrolled in the BSRBR-RA at the time of the analysis who had been exposed to the drug.
“The risk of the event was highest in the first year of treatment, followed by reduction over time,” Dr. Jani reported. “Reassuringly, up to two-thirds of patients in both cohorts had manifestations that were just limited to cutaneous involvement,” she said.
The most common systemic presentation was digital ischemia, affecting 14% of patients treated with anti-TNFs and 14% of those given nbDMARDs. Neurologic involvement was also seen in both groups of patients (7% vs. 7%), but new nail-fold vasculitis (17% vs. 0%), respiratory involvement (4% vs. 0%), associated thrombotic events (5% vs. 0%), and renal involvement (2.5% vs. 0%) were seen in TNF inhibitor-treated patients only.
Ten anti-TNF–treated patients and one nbDMARD-treated patient needed treatment for the vasculitis-like event, and three patients in the anti-TNF cohort died as a result of the event, all of whom had multisystem organ involvement and one of whom had cytoplasmic ANCA-positive vasculitis.
Treatment with methotrexate or sulfasalazine at baseline was associated with a lower risk for vasculitis-like events, while seropositive status, disease duration, DAS28, and HAQ scores were associated with an increased risk for such events.
The BSRBR-RA receives restricted income financial support from Abbvie, Amgen, Swedish Orphan Biovitram (SOBI), Merck, Pfizer, Roche, and UCB Pharma. Dr. Jani disclosed she has received honoraria from Pfizer, Abbvie, and UCB Pharma.
GLASGOW – Treatment with tumor necrosis factor (TNF) inhibitors for rheumatoid arthritis is associated with a low risk of vasculitis-like events, according to a large analysis of data from the United Kingdom.
Investigators using data from the British Society for Rheumatology Biologics Register for Rheumatoid Arthritis (BSRBR-RA) found that the crude incidence rate was 16 cases per 10,000 person-years among TNF-inhibitor users versus seven cases per 10,000 person-years among users of nonbiologic disease-modifying antirheumatic drugs (nbDMARDs) such as methotrexate and sulfasalazine.
Although the risk was slightly higher among anti-TNF than nbDMARD users, the propensity score fully adjusted hazard ratio for a first vasculitis-like event was 1.27, comparing the anti-TNF drugs with nbDMARDs, with a 95% confidence interval of 0.40-4.04.
“This is the first prospective observational study to systematically look at the risk of vasculitis-like events” in patients with RA treated with anti-TNF agents, Dr. Meghna Jani said at the British Society for Rheumatology annual conference.
Dr. Jani of the Arthritis Research UK Centre for Epidemiology at the University of Manchester (England) explained that the reason for looking at this topic was that vasculitis-like events had been reported in case series and single-center studies, but these prior reports were too small to be able to estimate exactly how big a problem this was.
“Anti-TNF agents are associated with the development of a number of autoantibodies, including antinuclear antibodies and antidrug antibodies, and ANCA [antineutrophil cytoplasmic antibody],” she observed.
“We know that a small proportion of these patients may then go on to develop autoimmune diseases, some independent of autoantibodies,” she added. The most common of these is vasculitis, including cutaneous vasculitis.
Vasculitis is a somewhat paradoxical adverse event, she noted, in that it has been associated with anti-TNF therapy, but these drugs can also be used to treat it.
Now in its 15th year, the BSRBR-RA is the largest ongoing cohort of patients treated with biologic agents for rheumatic disease and provides one of the best sources of data to examine the risk for vasculitis-like events Dr. Jani observed. The aims were to look at the respective risks as well as to see if there were any particular predictive factors.
The current analysis included more than 16,000 patients enrolled in the BSRBR-RA between 2001 and 2015, of whom 12,745 were newly started on an anti-TNF drug and 3,640 were receiving nbDMARDs and were also biologic naive. The mean age of patients in the two groups was 56 and 60 years, 76% and 72% were female, with a mean Disease Activity Score (DAS28) of 6.5 and 5.1 and median disease duration of 11 and 6 years, respectively.
After more than 52,428 person-years of exposure and a median of 5.1 years of follow-up, 81 vasculitis-like events occurred in the anti-TNF therapy group. Vasculitis-like events were attributed to treatment only if they had occurred within 90 days of starting the drug. Follow-up stopped after a first event; if there was a switch to another biologic drug; and at death, the last clinical follow-up, or the end of the analysis period (May 31, 2015).
In comparison, there were 20,635 person-years of exposure and 6.5 years’ follow-up in the nbDMARD group, with 14 vasculitis-like events reported during this time.
A sensitivity analysis was performed excluding patients who had nail-fold vasculitis at baseline, had vasculitis due to a possible secondary cause such as infection, and were taking any other medications associated with vasculitis-like events. Results showed a similar risk for a first vasculitis event between anti-TNF and nbDMARD users (aHR = 1.05; 95% CI, 0.32-3.45).
Looking at the risk of vasculitis events for individual anti-TNF drugs, there initially appeared to be a higher risk for patients taking infliximab (n = 3,292) and etanercept (n = 4,450) but not for those taking adalimumab (n = 4,312) versus nbDMARDs, with crude incidence rates of 10, 17, and 11 per 10,000 person-years; after adjustment, these differences were not significant (aHRs of 1.55, 1.72, and 0.77, respectively, with 95% CIs crossing 1.0). A crude rate for certolizumab could not be calculated as there were no vasculitis events reported but there were only 691 patients enrolled in the BSRBR-RA at the time of the analysis who had been exposed to the drug.
“The risk of the event was highest in the first year of treatment, followed by reduction over time,” Dr. Jani reported. “Reassuringly, up to two-thirds of patients in both cohorts had manifestations that were just limited to cutaneous involvement,” she said.
The most common systemic presentation was digital ischemia, affecting 14% of patients treated with anti-TNFs and 14% of those given nbDMARDs. Neurologic involvement was also seen in both groups of patients (7% vs. 7%), but new nail-fold vasculitis (17% vs. 0%), respiratory involvement (4% vs. 0%), associated thrombotic events (5% vs. 0%), and renal involvement (2.5% vs. 0%) were seen in TNF inhibitor-treated patients only.
Ten anti-TNF–treated patients and one nbDMARD-treated patient needed treatment for the vasculitis-like event, and three patients in the anti-TNF cohort died as a result of the event, all of whom had multisystem organ involvement and one of whom had cytoplasmic ANCA-positive vasculitis.
Treatment with methotrexate or sulfasalazine at baseline was associated with a lower risk for vasculitis-like events, while seropositive status, disease duration, DAS28, and HAQ scores were associated with an increased risk for such events.
The BSRBR-RA receives restricted income financial support from Abbvie, Amgen, Swedish Orphan Biovitram (SOBI), Merck, Pfizer, Roche, and UCB Pharma. Dr. Jani disclosed she has received honoraria from Pfizer, Abbvie, and UCB Pharma.
AT RHEUMATOLOGY 2016
Key clinical point: There is a low risk of vasculitis-like events with tumor necrosis factor inhibitors.
Major finding: Crude incidence rates for vasculitis-like events were 16/10,000 person-years with TNF-inhibitor therapy and 7/10,000 person-years with nonbiologic disease-modifying antirheumatic drugs.
Data source: British Society for Rheumatology Biologics Register for Rheumatoid Arthritis of 12,745 TNF-inhibitor and 3,640 nbDMARD users.
Disclosures: The BSRBR-RA receives restricted income financial support from Abbvie, Amgen, Swedish Orphan Biovitram (SOBI), Merck, Pfizer, Roche, and UCB Pharma. Dr. Jani disclosed she has received honoraria from Pfizer, Abbvie, and UCB Pharma.
Erythematous Atrophic Plaque in the Inguinal Fold
The Diagnosis: Granulomatous Slack Skin Disease
Initial biopsy revealed a lichenoid lymphohistiocytic infiltrate with scattered epidermotropism, papillary dermal sclerosis, and lymphocyte atypia (Figure 1). A repeat biopsy showed a lichenoid granulomatous infiltrate with histiocytes and rare giant cells, superficially located in the dermis, without a deeper dense infiltration. Focal lymphocytic epidermotropism also was present (Figure 2). The infiltrate was CD3+CD4+ with a minority of cells also staining for CD8. An elastin stain demonstrated diminished elastin fibers in the superficial dermis. A clonal T-cell receptor gene rearrangement was identified by polymerase chain reaction. One group of pink and brown papules was present on the dorsal aspect of the right foot (Figure 3). A biopsy of this area showed similar findings. The patient was treated with a trial of carmustine 20-mg% ointment over the following year with some improvement of the mild pruritus but without notable change in the clinical findings.
Granulomatous slack skin disease (GSSD) is a rare form of mycosis fungoides–type cutaneous T-cell lymphoma. It usually presents as well-demarcated, atrophic, poikilodermatous patches and plaques with a predilection for the inguinal and axillary regions.1 The affected areas tend to be asymptomatic and enlarge gradually over years to become pendulous with lax skin and wrinkles. In contrast to other forms of cutaneous T-cell lymphoma, extracutaneous spread is rare. The disease shows a slow progression over many years and by itself is not life threatening. However, affected patients have a risk for developing secondary lymphoproliferative neoplasms, which have been documented in approximately 50% of reported cases.2 These lymphoproliferative neoplasms may arise concurrently, precede, or follow the development of GSSD lesions. Hodgkin lymphoma, seen in 33% of cases, is the most common association, with others being non-Hodgkin lymphoma, mycosis fungoides, acute myeloid leukemia, and Langerhans cell histiocytosis.1-3
Histologically, GSSD is characterized by a dense, dermal, granulomatous proliferation of atypical T lymphocytes with scattered multinucleated giant cells.1,4 There is a loss of elastin fibers in the infiltrated areas, and occasional elastophagocytosis can be seen.1,2,4 Immunoprofiling of the infiltrate has shown CD3+CD4+CD45RO+ T-helper cells with occasional loss of CD5 and CD7.3 A clonal T-cell receptor rearrangement of the g and b genes frequently is described.1,4,5
At this time no treatment has been found to be reliably curative. Varying success in treating GSSD has been achieved with topical nitrogen mustard, carmustine, topical and systemic corticosteroids, psoralen plus UVA, radiotherapy, azathioprine, IFN-g, and combinations of these agents.1-3,6-9 Excision of the diseased skin has been performed for cosmetically or functionally disturbing lesions, but in all but one case the lesions recurred within months.1,10 A consistently reliable treatment of GSSD has not been established; treatment should be tailored to the individual patient.
- Shah A, Safaya A. Granulomatous slack skin disease: a review, in comparison with mycosis fungoides. J Eur Acad Dermatol Venereol. 2012;26:1472-1478.
- Teixeira M, Alves R, Lima M, et al. Granulomatous slack skin. Eur J Dermatol. 2007;17:435-438.
- van Haselen CW, Toonstra J, van der Putte SJ, et al. Granulomatous slack skin: report of three patients with an updated review of the literature. Dermatology. 1998;196:382-391.
- Kempf W, Ostheeren-Michaelis S, Paulli M, et al. Granulomatous mycosis fungoides and granulomatous slack skin: a multicenter study of the Cutaneous Lymphoma Histopathology Task Force Group of the European Organization for Research and Treatment of Cancer (EORTC). Arch Dermatol. 2008;144:1609-1617.
- LeBoit PE, Zackheim HS, White CR Jr. Granulomatous variants of cutaneous t-cell lymphoma: the histopathology of granulomatous mycosis fungoides and granulomatous slack skin. Am J Surg Pathol. 1988;12:83-95.
- Hultgren TL, Jones D, Duvic M. Topical nitrogen mustard for the treatment of granulomatous slack skin. Am J Clin Dermatol. 2007;8:51-54.
- Camacho FM, Burg G, Moreno JC, et al. Granulomatous slack skin in childhood. Pediatr Dermatol. 1997;14:204-208.
- Liu Z, Huang C, Li J. Prednisone combined with interferon for the treatment of one case of generalized granulomatous slack skin. J Huazhong Univ Sci Technolo Med Sci. 2005;25:617-618.
- Oberholzer PA, Cozzio A, Dummer R, et al. Granulomatous slack skin responds to UVA1 phototherapy. Dermatology. 2009;219:268-271.
- Clarijis M, Poot F, Laka A, et al. Granulomatous slack skin: treatment with extensive surgery and review of the literature. Dermatology. 2003;206:393-397.
The Diagnosis: Granulomatous Slack Skin Disease
Initial biopsy revealed a lichenoid lymphohistiocytic infiltrate with scattered epidermotropism, papillary dermal sclerosis, and lymphocyte atypia (Figure 1). A repeat biopsy showed a lichenoid granulomatous infiltrate with histiocytes and rare giant cells, superficially located in the dermis, without a deeper dense infiltration. Focal lymphocytic epidermotropism also was present (Figure 2). The infiltrate was CD3+CD4+ with a minority of cells also staining for CD8. An elastin stain demonstrated diminished elastin fibers in the superficial dermis. A clonal T-cell receptor gene rearrangement was identified by polymerase chain reaction. One group of pink and brown papules was present on the dorsal aspect of the right foot (Figure 3). A biopsy of this area showed similar findings. The patient was treated with a trial of carmustine 20-mg% ointment over the following year with some improvement of the mild pruritus but without notable change in the clinical findings.



Granulomatous slack skin disease (GSSD) is a rare form of mycosis fungoides–type cutaneous T-cell lymphoma. It usually presents as well-demarcated, atrophic, poikilodermatous patches and plaques with a predilection for the inguinal and axillary regions.1 The affected areas tend to be asymptomatic and enlarge gradually over years to become pendulous with lax skin and wrinkles. In contrast to other forms of cutaneous T-cell lymphoma, extracutaneous spread is rare. The disease shows a slow progression over many years and by itself is not life threatening. However, affected patients have a risk for developing secondary lymphoproliferative neoplasms, which have been documented in approximately 50% of reported cases.2 These lymphoproliferative neoplasms may arise concurrently, precede, or follow the development of GSSD lesions. Hodgkin lymphoma, seen in 33% of cases, is the most common association, with others being non-Hodgkin lymphoma, mycosis fungoides, acute myeloid leukemia, and Langerhans cell histiocytosis.1-3
Histologically, GSSD is characterized by a dense, dermal, granulomatous proliferation of atypical T lymphocytes with scattered multinucleated giant cells.1,4 There is a loss of elastin fibers in the infiltrated areas, and occasional elastophagocytosis can be seen.1,2,4 Immunoprofiling of the infiltrate has shown CD3+CD4+CD45RO+ T-helper cells with occasional loss of CD5 and CD7.3 A clonal T-cell receptor rearrangement of the g and b genes frequently is described.1,4,5
At this time no treatment has been found to be reliably curative. Varying success in treating GSSD has been achieved with topical nitrogen mustard, carmustine, topical and systemic corticosteroids, psoralen plus UVA, radiotherapy, azathioprine, IFN-g, and combinations of these agents.1-3,6-9 Excision of the diseased skin has been performed for cosmetically or functionally disturbing lesions, but in all but one case the lesions recurred within months.1,10 A consistently reliable treatment of GSSD has not been established; treatment should be tailored to the individual patient.
The Diagnosis: Granulomatous Slack Skin Disease
Initial biopsy revealed a lichenoid lymphohistiocytic infiltrate with scattered epidermotropism, papillary dermal sclerosis, and lymphocyte atypia (Figure 1). A repeat biopsy showed a lichenoid granulomatous infiltrate with histiocytes and rare giant cells, superficially located in the dermis, without a deeper dense infiltration. Focal lymphocytic epidermotropism also was present (Figure 2). The infiltrate was CD3+CD4+ with a minority of cells also staining for CD8. An elastin stain demonstrated diminished elastin fibers in the superficial dermis. A clonal T-cell receptor gene rearrangement was identified by polymerase chain reaction. One group of pink and brown papules was present on the dorsal aspect of the right foot (Figure 3). A biopsy of this area showed similar findings. The patient was treated with a trial of carmustine 20-mg% ointment over the following year with some improvement of the mild pruritus but without notable change in the clinical findings.



Granulomatous slack skin disease (GSSD) is a rare form of mycosis fungoides–type cutaneous T-cell lymphoma. It usually presents as well-demarcated, atrophic, poikilodermatous patches and plaques with a predilection for the inguinal and axillary regions.1 The affected areas tend to be asymptomatic and enlarge gradually over years to become pendulous with lax skin and wrinkles. In contrast to other forms of cutaneous T-cell lymphoma, extracutaneous spread is rare. The disease shows a slow progression over many years and by itself is not life threatening. However, affected patients have a risk for developing secondary lymphoproliferative neoplasms, which have been documented in approximately 50% of reported cases.2 These lymphoproliferative neoplasms may arise concurrently, precede, or follow the development of GSSD lesions. Hodgkin lymphoma, seen in 33% of cases, is the most common association, with others being non-Hodgkin lymphoma, mycosis fungoides, acute myeloid leukemia, and Langerhans cell histiocytosis.1-3
Histologically, GSSD is characterized by a dense, dermal, granulomatous proliferation of atypical T lymphocytes with scattered multinucleated giant cells.1,4 There is a loss of elastin fibers in the infiltrated areas, and occasional elastophagocytosis can be seen.1,2,4 Immunoprofiling of the infiltrate has shown CD3+CD4+CD45RO+ T-helper cells with occasional loss of CD5 and CD7.3 A clonal T-cell receptor rearrangement of the g and b genes frequently is described.1,4,5
At this time no treatment has been found to be reliably curative. Varying success in treating GSSD has been achieved with topical nitrogen mustard, carmustine, topical and systemic corticosteroids, psoralen plus UVA, radiotherapy, azathioprine, IFN-g, and combinations of these agents.1-3,6-9 Excision of the diseased skin has been performed for cosmetically or functionally disturbing lesions, but in all but one case the lesions recurred within months.1,10 A consistently reliable treatment of GSSD has not been established; treatment should be tailored to the individual patient.
- Shah A, Safaya A. Granulomatous slack skin disease: a review, in comparison with mycosis fungoides. J Eur Acad Dermatol Venereol. 2012;26:1472-1478.
- Teixeira M, Alves R, Lima M, et al. Granulomatous slack skin. Eur J Dermatol. 2007;17:435-438.
- van Haselen CW, Toonstra J, van der Putte SJ, et al. Granulomatous slack skin: report of three patients with an updated review of the literature. Dermatology. 1998;196:382-391.
- Kempf W, Ostheeren-Michaelis S, Paulli M, et al. Granulomatous mycosis fungoides and granulomatous slack skin: a multicenter study of the Cutaneous Lymphoma Histopathology Task Force Group of the European Organization for Research and Treatment of Cancer (EORTC). Arch Dermatol. 2008;144:1609-1617.
- LeBoit PE, Zackheim HS, White CR Jr. Granulomatous variants of cutaneous t-cell lymphoma: the histopathology of granulomatous mycosis fungoides and granulomatous slack skin. Am J Surg Pathol. 1988;12:83-95.
- Hultgren TL, Jones D, Duvic M. Topical nitrogen mustard for the treatment of granulomatous slack skin. Am J Clin Dermatol. 2007;8:51-54.
- Camacho FM, Burg G, Moreno JC, et al. Granulomatous slack skin in childhood. Pediatr Dermatol. 1997;14:204-208.
- Liu Z, Huang C, Li J. Prednisone combined with interferon for the treatment of one case of generalized granulomatous slack skin. J Huazhong Univ Sci Technolo Med Sci. 2005;25:617-618.
- Oberholzer PA, Cozzio A, Dummer R, et al. Granulomatous slack skin responds to UVA1 phototherapy. Dermatology. 2009;219:268-271.
- Clarijis M, Poot F, Laka A, et al. Granulomatous slack skin: treatment with extensive surgery and review of the literature. Dermatology. 2003;206:393-397.
- Shah A, Safaya A. Granulomatous slack skin disease: a review, in comparison with mycosis fungoides. J Eur Acad Dermatol Venereol. 2012;26:1472-1478.
- Teixeira M, Alves R, Lima M, et al. Granulomatous slack skin. Eur J Dermatol. 2007;17:435-438.
- van Haselen CW, Toonstra J, van der Putte SJ, et al. Granulomatous slack skin: report of three patients with an updated review of the literature. Dermatology. 1998;196:382-391.
- Kempf W, Ostheeren-Michaelis S, Paulli M, et al. Granulomatous mycosis fungoides and granulomatous slack skin: a multicenter study of the Cutaneous Lymphoma Histopathology Task Force Group of the European Organization for Research and Treatment of Cancer (EORTC). Arch Dermatol. 2008;144:1609-1617.
- LeBoit PE, Zackheim HS, White CR Jr. Granulomatous variants of cutaneous t-cell lymphoma: the histopathology of granulomatous mycosis fungoides and granulomatous slack skin. Am J Surg Pathol. 1988;12:83-95.
- Hultgren TL, Jones D, Duvic M. Topical nitrogen mustard for the treatment of granulomatous slack skin. Am J Clin Dermatol. 2007;8:51-54.
- Camacho FM, Burg G, Moreno JC, et al. Granulomatous slack skin in childhood. Pediatr Dermatol. 1997;14:204-208.
- Liu Z, Huang C, Li J. Prednisone combined with interferon for the treatment of one case of generalized granulomatous slack skin. J Huazhong Univ Sci Technolo Med Sci. 2005;25:617-618.
- Oberholzer PA, Cozzio A, Dummer R, et al. Granulomatous slack skin responds to UVA1 phototherapy. Dermatology. 2009;219:268-271.
- Clarijis M, Poot F, Laka A, et al. Granulomatous slack skin: treatment with extensive surgery and review of the literature. Dermatology. 2003;206:393-397.
A 66-year-old man presented with a rash on the groin of more than 6 years’ duration. The eruption was asymptomatic, except for occasional pruritus during the summer months. Numerous over-the-counter ointments, creams, and powders, as well as prescription topical corticosteroids, had failed to provide improvement. An outside biopsy performed 1 year earlier was considered nondiagnostic. Physical examination revealed a pink to violaceous, pendulous, atrophic plaque with slight scale on the right side of the lower abdomen running just superior to the right inguinal fold; the left inguinal fold was unaffected. Inguinal lymph nodes were not palpable. A 4-mm punch biopsy of the plaque in the inguinal fold was performed.
Summer colds
Most viral infections in summer months are caused by enteroviruses. We studied illnesses in about 400 kids aged 4-18 years seen in private pediatric practice and were surprised by what we found.
Our impression was that summer colds lasted for a shorter time span than winter colds. What we found was that the median duration of illness was about 8 days. Among the various syndromes, the most common was stomatitis (viral blisters in the throat), accounting for 58% of all cases seen. A flulike illness with fever, myalgias, and malaise was second most common (28% of cases), followed by hand/foot/mouth syndrome (8%), pleurodynia (3%), fever with viral rash (3%), and aseptic meningitis (1%). Most of the cases occurred among children 4-12 years old.
The most prevalent symptoms were fever, headache, sore throat, tiredness, muscle aches, and crankiness. Fever was present in about 85% of cases of children with stomatitis, in 95% of cases with myalgias and malaise, but in only 50% of cases of hand/foot/mouth. Headache was very common as well, occurring in about 40% of children with stomatitis, 70% of children with myalgias and malaise, and in 30% of children with hand/foot/mouth.
Illness within a household was quite common. About 50% of the children who came for care had a sibling or parent ill with a summer cold. However, while the symptoms of the family members often were the same as the child who presented for care, that was not always the case. As anticipated, most illness within a household occurred within a 2-week time span. Hand/foot/mouth was most easily recognized by parents to have spread among their children. When a parent became ill, it was almost always the mother because she was almost always the primary parent caretaker.
Summer colds took a toll on families in terms of loss of work by parents. Most of the children were ill enough to stay out of day care or school for about 2-4 days. Virtually all the children with hand/foot/mouth and stomatitis with classic viral blister lesions had a single visit to the pediatric practice, and very limited or no tests done or medications prescribed other than acetaminophen or ibuprofen. But for the children with higher fevers without hand/foot/mouth or stomatitis, the costs of care escalated as tests were much more often performed (CBC, chest x-ray), and medications prescribed (antibiotics for uncertain diagnosis in the context of high fever), and occasional referrals made to the emergency department for further work-up (100% of cases of aseptic meningitis and 50% of cases of pleurodynia).
Overall, summer colds are not so insignificant as presumed at first glance. What interests me now is why summer colds so infrequently are followed by an acute otitis media or sinusitis, whereas winter colds caused by respiratory syncytial virus, influenza, and rhinoviruses are followed by an acute otitis media in about one-third of cases. A new study is underway!
Dr. Pichichero, a specialist in pediatric infectious diseases, is director of the Research Institute, Rochester (N.Y.) General Hospital. He is also a pediatrician at Legacy Pediatrics in Rochester. He has no disclosures.
Most viral infections in summer months are caused by enteroviruses. We studied illnesses in about 400 kids aged 4-18 years seen in private pediatric practice and were surprised by what we found.
Our impression was that summer colds lasted for a shorter time span than winter colds. What we found was that the median duration of illness was about 8 days. Among the various syndromes, the most common was stomatitis (viral blisters in the throat), accounting for 58% of all cases seen. A flulike illness with fever, myalgias, and malaise was second most common (28% of cases), followed by hand/foot/mouth syndrome (8%), pleurodynia (3%), fever with viral rash (3%), and aseptic meningitis (1%). Most of the cases occurred among children 4-12 years old.
The most prevalent symptoms were fever, headache, sore throat, tiredness, muscle aches, and crankiness. Fever was present in about 85% of cases of children with stomatitis, in 95% of cases with myalgias and malaise, but in only 50% of cases of hand/foot/mouth. Headache was very common as well, occurring in about 40% of children with stomatitis, 70% of children with myalgias and malaise, and in 30% of children with hand/foot/mouth.
Illness within a household was quite common. About 50% of the children who came for care had a sibling or parent ill with a summer cold. However, while the symptoms of the family members often were the same as the child who presented for care, that was not always the case. As anticipated, most illness within a household occurred within a 2-week time span. Hand/foot/mouth was most easily recognized by parents to have spread among their children. When a parent became ill, it was almost always the mother because she was almost always the primary parent caretaker.
Summer colds took a toll on families in terms of loss of work by parents. Most of the children were ill enough to stay out of day care or school for about 2-4 days. Virtually all the children with hand/foot/mouth and stomatitis with classic viral blister lesions had a single visit to the pediatric practice, and very limited or no tests done or medications prescribed other than acetaminophen or ibuprofen. But for the children with higher fevers without hand/foot/mouth or stomatitis, the costs of care escalated as tests were much more often performed (CBC, chest x-ray), and medications prescribed (antibiotics for uncertain diagnosis in the context of high fever), and occasional referrals made to the emergency department for further work-up (100% of cases of aseptic meningitis and 50% of cases of pleurodynia).
Overall, summer colds are not so insignificant as presumed at first glance. What interests me now is why summer colds so infrequently are followed by an acute otitis media or sinusitis, whereas winter colds caused by respiratory syncytial virus, influenza, and rhinoviruses are followed by an acute otitis media in about one-third of cases. A new study is underway!
Dr. Pichichero, a specialist in pediatric infectious diseases, is director of the Research Institute, Rochester (N.Y.) General Hospital. He is also a pediatrician at Legacy Pediatrics in Rochester. He has no disclosures.
Most viral infections in summer months are caused by enteroviruses. We studied illnesses in about 400 kids aged 4-18 years seen in private pediatric practice and were surprised by what we found.
Our impression was that summer colds lasted for a shorter time span than winter colds. What we found was that the median duration of illness was about 8 days. Among the various syndromes, the most common was stomatitis (viral blisters in the throat), accounting for 58% of all cases seen. A flulike illness with fever, myalgias, and malaise was second most common (28% of cases), followed by hand/foot/mouth syndrome (8%), pleurodynia (3%), fever with viral rash (3%), and aseptic meningitis (1%). Most of the cases occurred among children 4-12 years old.
The most prevalent symptoms were fever, headache, sore throat, tiredness, muscle aches, and crankiness. Fever was present in about 85% of cases of children with stomatitis, in 95% of cases with myalgias and malaise, but in only 50% of cases of hand/foot/mouth. Headache was very common as well, occurring in about 40% of children with stomatitis, 70% of children with myalgias and malaise, and in 30% of children with hand/foot/mouth.
Illness within a household was quite common. About 50% of the children who came for care had a sibling or parent ill with a summer cold. However, while the symptoms of the family members often were the same as the child who presented for care, that was not always the case. As anticipated, most illness within a household occurred within a 2-week time span. Hand/foot/mouth was most easily recognized by parents to have spread among their children. When a parent became ill, it was almost always the mother because she was almost always the primary parent caretaker.
Summer colds took a toll on families in terms of loss of work by parents. Most of the children were ill enough to stay out of day care or school for about 2-4 days. Virtually all the children with hand/foot/mouth and stomatitis with classic viral blister lesions had a single visit to the pediatric practice, and very limited or no tests done or medications prescribed other than acetaminophen or ibuprofen. But for the children with higher fevers without hand/foot/mouth or stomatitis, the costs of care escalated as tests were much more often performed (CBC, chest x-ray), and medications prescribed (antibiotics for uncertain diagnosis in the context of high fever), and occasional referrals made to the emergency department for further work-up (100% of cases of aseptic meningitis and 50% of cases of pleurodynia).
Overall, summer colds are not so insignificant as presumed at first glance. What interests me now is why summer colds so infrequently are followed by an acute otitis media or sinusitis, whereas winter colds caused by respiratory syncytial virus, influenza, and rhinoviruses are followed by an acute otitis media in about one-third of cases. A new study is underway!
Dr. Pichichero, a specialist in pediatric infectious diseases, is director of the Research Institute, Rochester (N.Y.) General Hospital. He is also a pediatrician at Legacy Pediatrics in Rochester. He has no disclosures.
Sports safety
The National Athletic Trainers Association and the American Medical Society for Sports Medicine have developed a new program they call Collaborative Solutions for Safety in Sports, with the goal of establishing a suite of safety rules, policies, and possibly laws to protect high school athletes from injury (“School Athletes Often Lack Adequate Protection” by Jane Brody, New York Times, April 18, 2016).
After its second meeting, Dr. Jonathan Drezner, director of the Center for Sports Cardiology at the University of Washington, Seattle, said that the collaborative hopes that eventually every high school in the country will have an athletic trainer at every practice and game; an emergency action plan to respond appropriately to an athlete in distress; a publicly accessible automated external defibrillator (AED) and a school-based program in its use; and climatization policies to prevent heat injury and heat stroke.
I suspect that in communities in which the athletic facilities are located on a single campus that these guideline might be achievable. But here in Brunswick, Maine, and all of the other communities that I am familiar with, having a trainer at every practice and game is logistically impractical and financially unsustainable.
For example, on a given weekday afternoon in the spring here in Brunswick, there may be boys and girls varsity and junior varsity lacrosse, baseball, and softball practices or games on fields scattered around town – many of which are miles apart. The track team may be running on the roads and in the woods, who knows where. Staffing all of these events might be a financial windfall for the athletic trainers, if they could be found. But the money just isn’t there. Although the 50 high school athletes who died last year according to the National Athletic Trainers Association is 50 too many, I doubt that having a trainer at every practice and game would be a cost-effective solution.
When I opened my practice, Brunswick’s only pediatrician eagerly relinquished the job of school physician. Along with doing annual physical assessments on the bus drivers, I was expected to attend all of the home varsity football games. Trotting out on the field to evaluate the bumped and bruised players provided good visibility and helped build my practice. But I wondered who was going to attend to the junior varsity players and to the soccer players, who in my experience were more likely to be injured than the varsity football players. I certainly didn’t have the time. Nor could I be in five places at once.
After a couple of years, the athletic director and I hatched a plan to ask the school board to require that the coaches in every sport be certified in CPR. Our plan was quickly adopted, in part because a 15-year-old in a neighboring town had recently suffered a cardiac event during a track practice. Later, it was discovered that she had short QT syndrome, but there was an unfortunate delay in finding someone skilled in CPR.
In the 35 years since the CPR requirement was initially adopted, there has not been a single player who required resuscitation. However, last year one of the track coaches was running with a friend at dawn on a rural road when his friend dropped, pulseless. The coach’s school-required CPR training saved the man’s life.
While having a trainer at every high school practice and game is an unrealistic goal, educating coaches and players on how to identify and manage an athlete in distress makes a lot of sense. But the collaborative’s recommendation that is my personal favorite is having AEDs publicly accessible at games and practices. As a grandparent who spends a lot of time watching his grandchildren compete, I want the equipment available should I get a little too excited.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
The National Athletic Trainers Association and the American Medical Society for Sports Medicine have developed a new program they call Collaborative Solutions for Safety in Sports, with the goal of establishing a suite of safety rules, policies, and possibly laws to protect high school athletes from injury (“School Athletes Often Lack Adequate Protection” by Jane Brody, New York Times, April 18, 2016).
After its second meeting, Dr. Jonathan Drezner, director of the Center for Sports Cardiology at the University of Washington, Seattle, said that the collaborative hopes that eventually every high school in the country will have an athletic trainer at every practice and game; an emergency action plan to respond appropriately to an athlete in distress; a publicly accessible automated external defibrillator (AED) and a school-based program in its use; and climatization policies to prevent heat injury and heat stroke.
I suspect that in communities in which the athletic facilities are located on a single campus that these guideline might be achievable. But here in Brunswick, Maine, and all of the other communities that I am familiar with, having a trainer at every practice and game is logistically impractical and financially unsustainable.
For example, on a given weekday afternoon in the spring here in Brunswick, there may be boys and girls varsity and junior varsity lacrosse, baseball, and softball practices or games on fields scattered around town – many of which are miles apart. The track team may be running on the roads and in the woods, who knows where. Staffing all of these events might be a financial windfall for the athletic trainers, if they could be found. But the money just isn’t there. Although the 50 high school athletes who died last year according to the National Athletic Trainers Association is 50 too many, I doubt that having a trainer at every practice and game would be a cost-effective solution.
When I opened my practice, Brunswick’s only pediatrician eagerly relinquished the job of school physician. Along with doing annual physical assessments on the bus drivers, I was expected to attend all of the home varsity football games. Trotting out on the field to evaluate the bumped and bruised players provided good visibility and helped build my practice. But I wondered who was going to attend to the junior varsity players and to the soccer players, who in my experience were more likely to be injured than the varsity football players. I certainly didn’t have the time. Nor could I be in five places at once.
After a couple of years, the athletic director and I hatched a plan to ask the school board to require that the coaches in every sport be certified in CPR. Our plan was quickly adopted, in part because a 15-year-old in a neighboring town had recently suffered a cardiac event during a track practice. Later, it was discovered that she had short QT syndrome, but there was an unfortunate delay in finding someone skilled in CPR.
In the 35 years since the CPR requirement was initially adopted, there has not been a single player who required resuscitation. However, last year one of the track coaches was running with a friend at dawn on a rural road when his friend dropped, pulseless. The coach’s school-required CPR training saved the man’s life.
While having a trainer at every high school practice and game is an unrealistic goal, educating coaches and players on how to identify and manage an athlete in distress makes a lot of sense. But the collaborative’s recommendation that is my personal favorite is having AEDs publicly accessible at games and practices. As a grandparent who spends a lot of time watching his grandchildren compete, I want the equipment available should I get a little too excited.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
The National Athletic Trainers Association and the American Medical Society for Sports Medicine have developed a new program they call Collaborative Solutions for Safety in Sports, with the goal of establishing a suite of safety rules, policies, and possibly laws to protect high school athletes from injury (“School Athletes Often Lack Adequate Protection” by Jane Brody, New York Times, April 18, 2016).
After its second meeting, Dr. Jonathan Drezner, director of the Center for Sports Cardiology at the University of Washington, Seattle, said that the collaborative hopes that eventually every high school in the country will have an athletic trainer at every practice and game; an emergency action plan to respond appropriately to an athlete in distress; a publicly accessible automated external defibrillator (AED) and a school-based program in its use; and climatization policies to prevent heat injury and heat stroke.
I suspect that in communities in which the athletic facilities are located on a single campus that these guideline might be achievable. But here in Brunswick, Maine, and all of the other communities that I am familiar with, having a trainer at every practice and game is logistically impractical and financially unsustainable.
For example, on a given weekday afternoon in the spring here in Brunswick, there may be boys and girls varsity and junior varsity lacrosse, baseball, and softball practices or games on fields scattered around town – many of which are miles apart. The track team may be running on the roads and in the woods, who knows where. Staffing all of these events might be a financial windfall for the athletic trainers, if they could be found. But the money just isn’t there. Although the 50 high school athletes who died last year according to the National Athletic Trainers Association is 50 too many, I doubt that having a trainer at every practice and game would be a cost-effective solution.
When I opened my practice, Brunswick’s only pediatrician eagerly relinquished the job of school physician. Along with doing annual physical assessments on the bus drivers, I was expected to attend all of the home varsity football games. Trotting out on the field to evaluate the bumped and bruised players provided good visibility and helped build my practice. But I wondered who was going to attend to the junior varsity players and to the soccer players, who in my experience were more likely to be injured than the varsity football players. I certainly didn’t have the time. Nor could I be in five places at once.
After a couple of years, the athletic director and I hatched a plan to ask the school board to require that the coaches in every sport be certified in CPR. Our plan was quickly adopted, in part because a 15-year-old in a neighboring town had recently suffered a cardiac event during a track practice. Later, it was discovered that she had short QT syndrome, but there was an unfortunate delay in finding someone skilled in CPR.
In the 35 years since the CPR requirement was initially adopted, there has not been a single player who required resuscitation. However, last year one of the track coaches was running with a friend at dawn on a rural road when his friend dropped, pulseless. The coach’s school-required CPR training saved the man’s life.
While having a trainer at every high school practice and game is an unrealistic goal, educating coaches and players on how to identify and manage an athlete in distress makes a lot of sense. But the collaborative’s recommendation that is my personal favorite is having AEDs publicly accessible at games and practices. As a grandparent who spends a lot of time watching his grandchildren compete, I want the equipment available should I get a little too excited.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
Focus on patient-level factors, postop complications to reduce readmissions
CHICAGO – Preadmission and postdischarge factors were important predictors of postoperative readmission in a large cohort of surgical patients, but the hospital course had little incremental impact on either readmissions or postdischarge complications in the cohort, according to a retrospective study of Veterans Affairs data.
The findings suggest that efforts to reduce postoperative readmissions should focus on enhanced postdischarge surveillance and early intervention, Dr. Melanie S. Morris of the University of Alabama at Birmingham reported at the annual meeting of the American Surgical Association.
To assess the relative contributions of patient factors, operative characteristics, and postoperative hospital course on readmissions, she and her colleagues evaluated 243,956 general, vascular, and orthopedic surgery patients in 121 VA hospitals. The overall readmission rate among the cohort was 11.1%, and for general, vascular, and orthopedic surgeries, the rates were 12.9%, 15.4%, and 7.6%, respectively; the average postoperative length of stay was 6.9 days, and 6.1% of patients experienced a predischarge complication.
Almost all readmissions occurred within 2 weeks of discharge, and for general surgery patients, most occurred within 1 week. The readmission rate for vascular surgery patients remained high beyond the 2-week mark.
An examination of the reasons for readmission showed that wound complications were the most common reason for readmission, and this was particularly true for vascular surgery patients, in whom 44% of readmissions were for wound complications, Dr. Morris said.
Gastrointestinal complications including ileus and obstruction were also common, accounting for nearly 28% of readmissions among general surgery patients, she said.
Importantly, when including preoperative data (such as demographics, comorbidities, social and behavioral factors, labs and vital signs, and planned procedure type), the variability in readmissions could only be explained 8.6% of the time, she said.
“Adding in operative data, such as procedure complexity and intraoperative blood transfusions, as well as postoperative course, added very little to our predictive ability. Including both of those groups, we could only explain 10% of the variation in readmission,” she said.
Including postdischarge data such as complications and emergency department utilization in the model increased predictive ability to 18%.
R2 and C-statistics comparing the sequentially built model showed that demographics and comorbidities contributed the most to predicting readmission risk, Dr. Morris said.
Modeling based on readmission reason and specialty improved predictive ability. For example, almost 12% of readmissions for wound complications among vascular surgery patients were predictable.
“Our best predictive ability was for orthopedic patients who were readmitted with pneumonia. We were able to predict that 14% of the time,” she said.
The findings were derived by merging VA Surgical Quality Improvement Program data from inpatient operations performed between 2007 and 2014 and involving at least a 2-day postoperative hospital stay, with clinical data including laboratory findings, vitals, prior health care utilization, and postoperative complications.
“We then grouped our variables of interest into the following categories: preoperative, operative, postoperative but predischarge, and postdischarge,” she explained, noting that logistic models predicting 30-day readmission were constructed by sequentially adding groups into the model. Models were compared by way of adjusted R2 and C-statistics.
Assuming postoperative readmissions are preventable suggests that they are linked to the quality of care during the index hospitalization. The current findings demonstrate the challenges in predicting readmissions, and are important given that hospitals with higher-than-expected readmission rates for certain diagnoses and procedures are fined by the Centers for Medicare & Medicaid Services; 54% of hospitals were fined in 2015, she said.
“Readmission is difficult to predict at the time of discharge despite exhaustive statistical modeling with very granular clinical patient-level detail. Preoperative patient factors and postdischarge complications contribute the most to predictive models. Efforts to decrease readmissions should focus on modifiable patient-level factors, transitions of care, and minimizing postoperative complications,” she concluded.
Dr. Morris reported having no disclosures.
The complete manuscript of this presentation is anticipated to be published in Annals of Surgery pending editorial review.
CHICAGO – Preadmission and postdischarge factors were important predictors of postoperative readmission in a large cohort of surgical patients, but the hospital course had little incremental impact on either readmissions or postdischarge complications in the cohort, according to a retrospective study of Veterans Affairs data.
The findings suggest that efforts to reduce postoperative readmissions should focus on enhanced postdischarge surveillance and early intervention, Dr. Melanie S. Morris of the University of Alabama at Birmingham reported at the annual meeting of the American Surgical Association.
To assess the relative contributions of patient factors, operative characteristics, and postoperative hospital course on readmissions, she and her colleagues evaluated 243,956 general, vascular, and orthopedic surgery patients in 121 VA hospitals. The overall readmission rate among the cohort was 11.1%, and for general, vascular, and orthopedic surgeries, the rates were 12.9%, 15.4%, and 7.6%, respectively; the average postoperative length of stay was 6.9 days, and 6.1% of patients experienced a predischarge complication.
Almost all readmissions occurred within 2 weeks of discharge, and for general surgery patients, most occurred within 1 week. The readmission rate for vascular surgery patients remained high beyond the 2-week mark.
An examination of the reasons for readmission showed that wound complications were the most common reason for readmission, and this was particularly true for vascular surgery patients, in whom 44% of readmissions were for wound complications, Dr. Morris said.
Gastrointestinal complications including ileus and obstruction were also common, accounting for nearly 28% of readmissions among general surgery patients, she said.
Importantly, when including preoperative data (such as demographics, comorbidities, social and behavioral factors, labs and vital signs, and planned procedure type), the variability in readmissions could only be explained 8.6% of the time, she said.
“Adding in operative data, such as procedure complexity and intraoperative blood transfusions, as well as postoperative course, added very little to our predictive ability. Including both of those groups, we could only explain 10% of the variation in readmission,” she said.
Including postdischarge data such as complications and emergency department utilization in the model increased predictive ability to 18%.
R2 and C-statistics comparing the sequentially built model showed that demographics and comorbidities contributed the most to predicting readmission risk, Dr. Morris said.
Modeling based on readmission reason and specialty improved predictive ability. For example, almost 12% of readmissions for wound complications among vascular surgery patients were predictable.
“Our best predictive ability was for orthopedic patients who were readmitted with pneumonia. We were able to predict that 14% of the time,” she said.
The findings were derived by merging VA Surgical Quality Improvement Program data from inpatient operations performed between 2007 and 2014 and involving at least a 2-day postoperative hospital stay, with clinical data including laboratory findings, vitals, prior health care utilization, and postoperative complications.
“We then grouped our variables of interest into the following categories: preoperative, operative, postoperative but predischarge, and postdischarge,” she explained, noting that logistic models predicting 30-day readmission were constructed by sequentially adding groups into the model. Models were compared by way of adjusted R2 and C-statistics.
Assuming postoperative readmissions are preventable suggests that they are linked to the quality of care during the index hospitalization. The current findings demonstrate the challenges in predicting readmissions, and are important given that hospitals with higher-than-expected readmission rates for certain diagnoses and procedures are fined by the Centers for Medicare & Medicaid Services; 54% of hospitals were fined in 2015, she said.
“Readmission is difficult to predict at the time of discharge despite exhaustive statistical modeling with very granular clinical patient-level detail. Preoperative patient factors and postdischarge complications contribute the most to predictive models. Efforts to decrease readmissions should focus on modifiable patient-level factors, transitions of care, and minimizing postoperative complications,” she concluded.
Dr. Morris reported having no disclosures.
The complete manuscript of this presentation is anticipated to be published in Annals of Surgery pending editorial review.
CHICAGO – Preadmission and postdischarge factors were important predictors of postoperative readmission in a large cohort of surgical patients, but the hospital course had little incremental impact on either readmissions or postdischarge complications in the cohort, according to a retrospective study of Veterans Affairs data.
The findings suggest that efforts to reduce postoperative readmissions should focus on enhanced postdischarge surveillance and early intervention, Dr. Melanie S. Morris of the University of Alabama at Birmingham reported at the annual meeting of the American Surgical Association.
To assess the relative contributions of patient factors, operative characteristics, and postoperative hospital course on readmissions, she and her colleagues evaluated 243,956 general, vascular, and orthopedic surgery patients in 121 VA hospitals. The overall readmission rate among the cohort was 11.1%, and for general, vascular, and orthopedic surgeries, the rates were 12.9%, 15.4%, and 7.6%, respectively; the average postoperative length of stay was 6.9 days, and 6.1% of patients experienced a predischarge complication.
Almost all readmissions occurred within 2 weeks of discharge, and for general surgery patients, most occurred within 1 week. The readmission rate for vascular surgery patients remained high beyond the 2-week mark.
An examination of the reasons for readmission showed that wound complications were the most common reason for readmission, and this was particularly true for vascular surgery patients, in whom 44% of readmissions were for wound complications, Dr. Morris said.
Gastrointestinal complications including ileus and obstruction were also common, accounting for nearly 28% of readmissions among general surgery patients, she said.
Importantly, when including preoperative data (such as demographics, comorbidities, social and behavioral factors, labs and vital signs, and planned procedure type), the variability in readmissions could only be explained 8.6% of the time, she said.
“Adding in operative data, such as procedure complexity and intraoperative blood transfusions, as well as postoperative course, added very little to our predictive ability. Including both of those groups, we could only explain 10% of the variation in readmission,” she said.
Including postdischarge data such as complications and emergency department utilization in the model increased predictive ability to 18%.
R2 and C-statistics comparing the sequentially built model showed that demographics and comorbidities contributed the most to predicting readmission risk, Dr. Morris said.
Modeling based on readmission reason and specialty improved predictive ability. For example, almost 12% of readmissions for wound complications among vascular surgery patients were predictable.
“Our best predictive ability was for orthopedic patients who were readmitted with pneumonia. We were able to predict that 14% of the time,” she said.
The findings were derived by merging VA Surgical Quality Improvement Program data from inpatient operations performed between 2007 and 2014 and involving at least a 2-day postoperative hospital stay, with clinical data including laboratory findings, vitals, prior health care utilization, and postoperative complications.
“We then grouped our variables of interest into the following categories: preoperative, operative, postoperative but predischarge, and postdischarge,” she explained, noting that logistic models predicting 30-day readmission were constructed by sequentially adding groups into the model. Models were compared by way of adjusted R2 and C-statistics.
Assuming postoperative readmissions are preventable suggests that they are linked to the quality of care during the index hospitalization. The current findings demonstrate the challenges in predicting readmissions, and are important given that hospitals with higher-than-expected readmission rates for certain diagnoses and procedures are fined by the Centers for Medicare & Medicaid Services; 54% of hospitals were fined in 2015, she said.
“Readmission is difficult to predict at the time of discharge despite exhaustive statistical modeling with very granular clinical patient-level detail. Preoperative patient factors and postdischarge complications contribute the most to predictive models. Efforts to decrease readmissions should focus on modifiable patient-level factors, transitions of care, and minimizing postoperative complications,” she concluded.
Dr. Morris reported having no disclosures.
The complete manuscript of this presentation is anticipated to be published in Annals of Surgery pending editorial review.
AT THE ASA ANNUAL MEETING
Key clinical point: Preadmission and postdischarge factors were important predictors of postoperative readmission in a large cohort of surgical patients, but the hospital course had little incremental impact on either readmissions or postdischarge complications.
Major finding: Including both preoperative and operative data in the model predicted only 10% of the variability in readmission rates.
Data source: A retrospective study of data for nearly 244,000 VA patients.
Disclosures: Dr. Morris reported having no disclosures.