User login
The Changing Face of MOC More information
On April 8, 2106, CHEST joined 29 other medical specialty societies at the American Board of Internal Medicine’s (ABIM’s) biannual Liaison Committee on Certification and Recertification (LCCR) meeting in Philadelphia. The meeting focused on the changing face of Maintenance of Certification (MOC) and the vision ABIM has for the future of MOC.
President Richard J. Baron, MD, responded to a letter signed by several medical specialty societies, asking for clarification on ABIM’s vision and philosophy for MOC and the future changes ABIM is considering for the MOC program. Dr. Baron articulated the desire for MOC to be relevant to physicians’ practices in collaboration with the ABIM and the medical specialty societies, to produce, “a credential that speaks to whether physicians are staying current in knowledge and practice over the course of their career in their specialty.”
Dr. Richard G. Battaglia, ABIM Chief Medical Officer, and Eric McKeeby, ABIM Director of Community Engagement, reported the results of a membership survey and focus group discussions regarding ABIM’s Assessment 2020 Report, published in September 2015. They highlighted the challenges, opportunities, and future plans for the MOC program, in light of diplomates’ input through these mechanisms. In the feedback received, the majority of diplomates favored a move away from the secure 10-year MOC recertification examination. Several options were presented, including smaller, more frequent exams, secure exams taken from home or office, and the ability to “test out” of the 10-year exam by successfully completing smaller assessments along the way. Ultimately, participants favored a move away from assessment and toward learning and improvement through a mechanism that is relevant to their real-world practices. Dr. Baron noted the ABIM survey results in which 76.3% of diplomates said they wanted the MOC credential to mean “I am staying current in the knowledge I need to practice,” and he reiterated the Board’s commitment to developing assessment approaches that would result in a meaningful credential based on performance. The ABIM Board of Directors met in April, with the exam format being a priority for them.
Regarding the future of MOC, while the practice assessment requirement is on hold through 2018, ABIM recognizes the work in this area many physicians are currently doing. By early 2017, ABIM is planning on extending the partnership with ACCME to recognize practice assessment activities, along with current medical knowledge activities, for both CME and MOC. This expansion would include blended activities that meet both medical knowledge and practice assessment requirements.
In addition to ABIM staff’s perspectives, three medical societies, including CHEST, reported on their MOC efforts. Heather Dethloff, CHEST Education and Accreditation Specialist, participated in a panel discussion, along with the Endocrine Society and the American Academy of Hospice and Palliative Medicine regarding the ongoing efforts to incorporate MOC into educational activities within their organizations. Successes and challenges encountered through the ABIM MOC certification process were the topics for discussion. During this presentation, CHEST announced its plan to incorporate ABIM MOC into the entire CHEST Annual Meeting 2016; details will be communicated to CHEST members and meeting registrants in coming months.
Throughout these changes to the MOC program, CHEST has been, and will continue to be, in communication with ABIM, advocating for our members. Any questions or concerns about this process can be directed to Heather Dethloff, Education and Accreditation Specialist, at [email protected].
More information
New ABIM Survey Indicates Physician Interest in Potential Changes to MOC Assessment: www.abim.org/news/new-abim-survey-indicates-physician-interest-in-potential-changes-to-moc-assessment.aspx
On April 8, 2106, CHEST joined 29 other medical specialty societies at the American Board of Internal Medicine’s (ABIM’s) biannual Liaison Committee on Certification and Recertification (LCCR) meeting in Philadelphia. The meeting focused on the changing face of Maintenance of Certification (MOC) and the vision ABIM has for the future of MOC.
President Richard J. Baron, MD, responded to a letter signed by several medical specialty societies, asking for clarification on ABIM’s vision and philosophy for MOC and the future changes ABIM is considering for the MOC program. Dr. Baron articulated the desire for MOC to be relevant to physicians’ practices in collaboration with the ABIM and the medical specialty societies, to produce, “a credential that speaks to whether physicians are staying current in knowledge and practice over the course of their career in their specialty.”
Dr. Richard G. Battaglia, ABIM Chief Medical Officer, and Eric McKeeby, ABIM Director of Community Engagement, reported the results of a membership survey and focus group discussions regarding ABIM’s Assessment 2020 Report, published in September 2015. They highlighted the challenges, opportunities, and future plans for the MOC program, in light of diplomates’ input through these mechanisms. In the feedback received, the majority of diplomates favored a move away from the secure 10-year MOC recertification examination. Several options were presented, including smaller, more frequent exams, secure exams taken from home or office, and the ability to “test out” of the 10-year exam by successfully completing smaller assessments along the way. Ultimately, participants favored a move away from assessment and toward learning and improvement through a mechanism that is relevant to their real-world practices. Dr. Baron noted the ABIM survey results in which 76.3% of diplomates said they wanted the MOC credential to mean “I am staying current in the knowledge I need to practice,” and he reiterated the Board’s commitment to developing assessment approaches that would result in a meaningful credential based on performance. The ABIM Board of Directors met in April, with the exam format being a priority for them.
Regarding the future of MOC, while the practice assessment requirement is on hold through 2018, ABIM recognizes the work in this area many physicians are currently doing. By early 2017, ABIM is planning on extending the partnership with ACCME to recognize practice assessment activities, along with current medical knowledge activities, for both CME and MOC. This expansion would include blended activities that meet both medical knowledge and practice assessment requirements.
In addition to ABIM staff’s perspectives, three medical societies, including CHEST, reported on their MOC efforts. Heather Dethloff, CHEST Education and Accreditation Specialist, participated in a panel discussion, along with the Endocrine Society and the American Academy of Hospice and Palliative Medicine regarding the ongoing efforts to incorporate MOC into educational activities within their organizations. Successes and challenges encountered through the ABIM MOC certification process were the topics for discussion. During this presentation, CHEST announced its plan to incorporate ABIM MOC into the entire CHEST Annual Meeting 2016; details will be communicated to CHEST members and meeting registrants in coming months.
Throughout these changes to the MOC program, CHEST has been, and will continue to be, in communication with ABIM, advocating for our members. Any questions or concerns about this process can be directed to Heather Dethloff, Education and Accreditation Specialist, at [email protected].
More information
New ABIM Survey Indicates Physician Interest in Potential Changes to MOC Assessment: www.abim.org/news/new-abim-survey-indicates-physician-interest-in-potential-changes-to-moc-assessment.aspx
On April 8, 2106, CHEST joined 29 other medical specialty societies at the American Board of Internal Medicine’s (ABIM’s) biannual Liaison Committee on Certification and Recertification (LCCR) meeting in Philadelphia. The meeting focused on the changing face of Maintenance of Certification (MOC) and the vision ABIM has for the future of MOC.
President Richard J. Baron, MD, responded to a letter signed by several medical specialty societies, asking for clarification on ABIM’s vision and philosophy for MOC and the future changes ABIM is considering for the MOC program. Dr. Baron articulated the desire for MOC to be relevant to physicians’ practices in collaboration with the ABIM and the medical specialty societies, to produce, “a credential that speaks to whether physicians are staying current in knowledge and practice over the course of their career in their specialty.”
Dr. Richard G. Battaglia, ABIM Chief Medical Officer, and Eric McKeeby, ABIM Director of Community Engagement, reported the results of a membership survey and focus group discussions regarding ABIM’s Assessment 2020 Report, published in September 2015. They highlighted the challenges, opportunities, and future plans for the MOC program, in light of diplomates’ input through these mechanisms. In the feedback received, the majority of diplomates favored a move away from the secure 10-year MOC recertification examination. Several options were presented, including smaller, more frequent exams, secure exams taken from home or office, and the ability to “test out” of the 10-year exam by successfully completing smaller assessments along the way. Ultimately, participants favored a move away from assessment and toward learning and improvement through a mechanism that is relevant to their real-world practices. Dr. Baron noted the ABIM survey results in which 76.3% of diplomates said they wanted the MOC credential to mean “I am staying current in the knowledge I need to practice,” and he reiterated the Board’s commitment to developing assessment approaches that would result in a meaningful credential based on performance. The ABIM Board of Directors met in April, with the exam format being a priority for them.
Regarding the future of MOC, while the practice assessment requirement is on hold through 2018, ABIM recognizes the work in this area many physicians are currently doing. By early 2017, ABIM is planning on extending the partnership with ACCME to recognize practice assessment activities, along with current medical knowledge activities, for both CME and MOC. This expansion would include blended activities that meet both medical knowledge and practice assessment requirements.
In addition to ABIM staff’s perspectives, three medical societies, including CHEST, reported on their MOC efforts. Heather Dethloff, CHEST Education and Accreditation Specialist, participated in a panel discussion, along with the Endocrine Society and the American Academy of Hospice and Palliative Medicine regarding the ongoing efforts to incorporate MOC into educational activities within their organizations. Successes and challenges encountered through the ABIM MOC certification process were the topics for discussion. During this presentation, CHEST announced its plan to incorporate ABIM MOC into the entire CHEST Annual Meeting 2016; details will be communicated to CHEST members and meeting registrants in coming months.
Throughout these changes to the MOC program, CHEST has been, and will continue to be, in communication with ABIM, advocating for our members. Any questions or concerns about this process can be directed to Heather Dethloff, Education and Accreditation Specialist, at [email protected].
More information
New ABIM Survey Indicates Physician Interest in Potential Changes to MOC Assessment: www.abim.org/news/new-abim-survey-indicates-physician-interest-in-potential-changes-to-moc-assessment.aspx
Catching up with our past presidents
Where are they now? What have they been up to? CHEST’s Past Presidents each forged the way for the many successes of the American College of Chest Physicians (CHEST), leading to enhanced patient care around the globe. Their outstanding leadership and vision are evidenced today in many of CHEST’s current initiatives, and now it is time to check in with these past leaders to give us a look at what’s new in their lives.
Kalpalatha K. Guntupalli, MD, Master FCCP, MACP, FCCM
Frances K. Friedman and Oscar Friedman, MD ’36 Endowed Professor for Pulmonary Disorders; and Chief, Section of Pulmonary, Critical Care, and Sleep Medicine, Department of Medicine, Baylor College of Medicine.
President 2009-2010
November 1, 2009, is clearly etched into my memory. I was sworn in as the 73rd President, and 3rd woman President, of the American College of Chest Physicians during CHEST Annual Meeting 2009 in San Diego.
I consider the 2 years leading up to the presidency and the year following my term as the best years of my professional career. They were action packed, full of excitement that gave immense satisfaction. During my year, the longtime EVP/CEO, Al Lever, retired, and we welcomed the new EVP/CEO Mr. Paul Markowski.
To make the transition easier, I started the “4 Ps” call of the 4 Presidents (President-Designate, President-Elect, President, and the Past President), a weekly call to catch up and keep everyone in the loop. This has since become a tradition in the organization.
My theme for the year was “Act local, Think global.” We started many international initiatives, in the Middle East, India, China, and South America, that have since evolved into successful programs. In 2010, as members of the Federation of International Respiratory Societies (FIRS), we celebrated the “Year of the Lung.” We participated in the “world spirometry day” (102,487 spirometries were done globally) and did many other programs to increase awareness of lung disease. We held a long-term strategic retreat developing goals for the College. We implemented many other process-driven initiatives under the new CEO’s leadership.
Land was acquired where the new beautiful headquarters building now stands.
What is life like after CHEST Presidency?
I was very honored in 2012 to receive the “Pravasi Bharatiya Samman,” the highest award bestowed on a nonresident Indian by the President of India or distinguished members of the Indian Diaspora to “honor exceptional and meritorious contributions in their chosen field/profession and enhancing the image of India.” In 2013, I spent 4 months as a Fulbright Scholar forming an ARDS network in India, and in 2015, I was honored as a Master FCCP by our own organization.
As the Section Chief, I have been busy building a “Lung Institute” and ICU services along with many new initiatives in our very active Pulmonary/CC/Sleep section at Baylor College of Medicine.
Where are they now? What have they been up to? CHEST’s Past Presidents each forged the way for the many successes of the American College of Chest Physicians (CHEST), leading to enhanced patient care around the globe. Their outstanding leadership and vision are evidenced today in many of CHEST’s current initiatives, and now it is time to check in with these past leaders to give us a look at what’s new in their lives.
Kalpalatha K. Guntupalli, MD, Master FCCP, MACP, FCCM
Frances K. Friedman and Oscar Friedman, MD ’36 Endowed Professor for Pulmonary Disorders; and Chief, Section of Pulmonary, Critical Care, and Sleep Medicine, Department of Medicine, Baylor College of Medicine.
President 2009-2010
November 1, 2009, is clearly etched into my memory. I was sworn in as the 73rd President, and 3rd woman President, of the American College of Chest Physicians during CHEST Annual Meeting 2009 in San Diego.
I consider the 2 years leading up to the presidency and the year following my term as the best years of my professional career. They were action packed, full of excitement that gave immense satisfaction. During my year, the longtime EVP/CEO, Al Lever, retired, and we welcomed the new EVP/CEO Mr. Paul Markowski.
To make the transition easier, I started the “4 Ps” call of the 4 Presidents (President-Designate, President-Elect, President, and the Past President), a weekly call to catch up and keep everyone in the loop. This has since become a tradition in the organization.
My theme for the year was “Act local, Think global.” We started many international initiatives, in the Middle East, India, China, and South America, that have since evolved into successful programs. In 2010, as members of the Federation of International Respiratory Societies (FIRS), we celebrated the “Year of the Lung.” We participated in the “world spirometry day” (102,487 spirometries were done globally) and did many other programs to increase awareness of lung disease. We held a long-term strategic retreat developing goals for the College. We implemented many other process-driven initiatives under the new CEO’s leadership.
Land was acquired where the new beautiful headquarters building now stands.
What is life like after CHEST Presidency?
I was very honored in 2012 to receive the “Pravasi Bharatiya Samman,” the highest award bestowed on a nonresident Indian by the President of India or distinguished members of the Indian Diaspora to “honor exceptional and meritorious contributions in their chosen field/profession and enhancing the image of India.” In 2013, I spent 4 months as a Fulbright Scholar forming an ARDS network in India, and in 2015, I was honored as a Master FCCP by our own organization.
As the Section Chief, I have been busy building a “Lung Institute” and ICU services along with many new initiatives in our very active Pulmonary/CC/Sleep section at Baylor College of Medicine.
Where are they now? What have they been up to? CHEST’s Past Presidents each forged the way for the many successes of the American College of Chest Physicians (CHEST), leading to enhanced patient care around the globe. Their outstanding leadership and vision are evidenced today in many of CHEST’s current initiatives, and now it is time to check in with these past leaders to give us a look at what’s new in their lives.
Kalpalatha K. Guntupalli, MD, Master FCCP, MACP, FCCM
Frances K. Friedman and Oscar Friedman, MD ’36 Endowed Professor for Pulmonary Disorders; and Chief, Section of Pulmonary, Critical Care, and Sleep Medicine, Department of Medicine, Baylor College of Medicine.
President 2009-2010
November 1, 2009, is clearly etched into my memory. I was sworn in as the 73rd President, and 3rd woman President, of the American College of Chest Physicians during CHEST Annual Meeting 2009 in San Diego.
I consider the 2 years leading up to the presidency and the year following my term as the best years of my professional career. They were action packed, full of excitement that gave immense satisfaction. During my year, the longtime EVP/CEO, Al Lever, retired, and we welcomed the new EVP/CEO Mr. Paul Markowski.
To make the transition easier, I started the “4 Ps” call of the 4 Presidents (President-Designate, President-Elect, President, and the Past President), a weekly call to catch up and keep everyone in the loop. This has since become a tradition in the organization.
My theme for the year was “Act local, Think global.” We started many international initiatives, in the Middle East, India, China, and South America, that have since evolved into successful programs. In 2010, as members of the Federation of International Respiratory Societies (FIRS), we celebrated the “Year of the Lung.” We participated in the “world spirometry day” (102,487 spirometries were done globally) and did many other programs to increase awareness of lung disease. We held a long-term strategic retreat developing goals for the College. We implemented many other process-driven initiatives under the new CEO’s leadership.
Land was acquired where the new beautiful headquarters building now stands.
What is life like after CHEST Presidency?
I was very honored in 2012 to receive the “Pravasi Bharatiya Samman,” the highest award bestowed on a nonresident Indian by the President of India or distinguished members of the Indian Diaspora to “honor exceptional and meritorious contributions in their chosen field/profession and enhancing the image of India.” In 2013, I spent 4 months as a Fulbright Scholar forming an ARDS network in India, and in 2015, I was honored as a Master FCCP by our own organization.
As the Section Chief, I have been busy building a “Lung Institute” and ICU services along with many new initiatives in our very active Pulmonary/CC/Sleep section at Baylor College of Medicine.
Predictors of abnormal longitudinal patterns of lung-function growth and decline
When it comes to predictive demographic and clinical factors associated with abnormal patterns of lung growth and decline in those with persistent, mild-to-moderate asthma in childhood, male sex and childhood levels of lung function as assessed by the forced expiratory volume in 1 second (FEV1) make all the difference, according to the results of a study published in the New England Journal of Medicine.
“Determinants of abnormal patterns of FEV1 growth and decline are multifactorial and complex, and identification of factors associated with the timing of a decline from the maximal level requires longitudinal data,” said author Michael McGeachie, Ph.D., of Brigham and Women’s Hospital, Boston, and his colleagues.
To identify these determinants, particularly in those with asthma, Dr. McGeachie and colleagues analyzed longitudinal data from a subset of participants from the Childhood Asthma Management Program (CAMP) cohort who were followed from enrollment at the age of 5 to 12 years, into the third decade of life. The trajectory of lung growth and the decline from maximum growth in this large cohort of persons who had persistent, mild-to-moderate asthma in childhood were compared against those from persons without asthma who were participants in the third National Health and Nutrition Examination Survey. (N Engl J Med. 2016;374:1842-52).
Data from 684 participants from the CAMP cohort were assessed and 25% had normal lung-function growth without an early decline, 26% had normal growth and an early decline, 23% had reduced growth without an early decline, and 26% had reduced growth and an early decline. Results of the multinomial logistic-regression analysis of risk factors for abnormal patterns of lung growth and decline showed that the 26% of participants with reduced growth and an early decline had lower FEV1 lung function at enrollment and were more likely to be male, compared with those who had normal growth.
Additional study results indicated that 18% of the participants who had reduced lung-function growth, with or without an early decline, met the case definition for chronic obstructive pulmonary disease (COPD) based on the postbronchodilator spirometric criteria at an age of less than 30 years.
Based on their data, Dr. McGeachie and his colleagues said that detection of an abnormal trajectory by means of early and ongoing serial FEV1 monitoring may help identify children and young adults at risk for abnormal lung-function growth that could lead to chronic airflow obstruction in adulthood.
Funding for this project was provided by grants from the Parker B. Francis Foundation, the National Institutes of Health, the National Human Genome Research Institute, and the Human Frontier Science Program Organization. Dr. McGeachie reported grant support from one of the funding sources during the conduct of the study. Nineteen coauthors reported they had nothing to disclose and the remainder either reported grant support or ties to industry sources.
When it comes to predictive demographic and clinical factors associated with abnormal patterns of lung growth and decline in those with persistent, mild-to-moderate asthma in childhood, male sex and childhood levels of lung function as assessed by the forced expiratory volume in 1 second (FEV1) make all the difference, according to the results of a study published in the New England Journal of Medicine.
“Determinants of abnormal patterns of FEV1 growth and decline are multifactorial and complex, and identification of factors associated with the timing of a decline from the maximal level requires longitudinal data,” said author Michael McGeachie, Ph.D., of Brigham and Women’s Hospital, Boston, and his colleagues.
To identify these determinants, particularly in those with asthma, Dr. McGeachie and colleagues analyzed longitudinal data from a subset of participants from the Childhood Asthma Management Program (CAMP) cohort who were followed from enrollment at the age of 5 to 12 years, into the third decade of life. The trajectory of lung growth and the decline from maximum growth in this large cohort of persons who had persistent, mild-to-moderate asthma in childhood were compared against those from persons without asthma who were participants in the third National Health and Nutrition Examination Survey. (N Engl J Med. 2016;374:1842-52).
Data from 684 participants from the CAMP cohort were assessed and 25% had normal lung-function growth without an early decline, 26% had normal growth and an early decline, 23% had reduced growth without an early decline, and 26% had reduced growth and an early decline. Results of the multinomial logistic-regression analysis of risk factors for abnormal patterns of lung growth and decline showed that the 26% of participants with reduced growth and an early decline had lower FEV1 lung function at enrollment and were more likely to be male, compared with those who had normal growth.
Additional study results indicated that 18% of the participants who had reduced lung-function growth, with or without an early decline, met the case definition for chronic obstructive pulmonary disease (COPD) based on the postbronchodilator spirometric criteria at an age of less than 30 years.
Based on their data, Dr. McGeachie and his colleagues said that detection of an abnormal trajectory by means of early and ongoing serial FEV1 monitoring may help identify children and young adults at risk for abnormal lung-function growth that could lead to chronic airflow obstruction in adulthood.
Funding for this project was provided by grants from the Parker B. Francis Foundation, the National Institutes of Health, the National Human Genome Research Institute, and the Human Frontier Science Program Organization. Dr. McGeachie reported grant support from one of the funding sources during the conduct of the study. Nineteen coauthors reported they had nothing to disclose and the remainder either reported grant support or ties to industry sources.
When it comes to predictive demographic and clinical factors associated with abnormal patterns of lung growth and decline in those with persistent, mild-to-moderate asthma in childhood, male sex and childhood levels of lung function as assessed by the forced expiratory volume in 1 second (FEV1) make all the difference, according to the results of a study published in the New England Journal of Medicine.
“Determinants of abnormal patterns of FEV1 growth and decline are multifactorial and complex, and identification of factors associated with the timing of a decline from the maximal level requires longitudinal data,” said author Michael McGeachie, Ph.D., of Brigham and Women’s Hospital, Boston, and his colleagues.
To identify these determinants, particularly in those with asthma, Dr. McGeachie and colleagues analyzed longitudinal data from a subset of participants from the Childhood Asthma Management Program (CAMP) cohort who were followed from enrollment at the age of 5 to 12 years, into the third decade of life. The trajectory of lung growth and the decline from maximum growth in this large cohort of persons who had persistent, mild-to-moderate asthma in childhood were compared against those from persons without asthma who were participants in the third National Health and Nutrition Examination Survey. (N Engl J Med. 2016;374:1842-52).
Data from 684 participants from the CAMP cohort were assessed and 25% had normal lung-function growth without an early decline, 26% had normal growth and an early decline, 23% had reduced growth without an early decline, and 26% had reduced growth and an early decline. Results of the multinomial logistic-regression analysis of risk factors for abnormal patterns of lung growth and decline showed that the 26% of participants with reduced growth and an early decline had lower FEV1 lung function at enrollment and were more likely to be male, compared with those who had normal growth.
Additional study results indicated that 18% of the participants who had reduced lung-function growth, with or without an early decline, met the case definition for chronic obstructive pulmonary disease (COPD) based on the postbronchodilator spirometric criteria at an age of less than 30 years.
Based on their data, Dr. McGeachie and his colleagues said that detection of an abnormal trajectory by means of early and ongoing serial FEV1 monitoring may help identify children and young adults at risk for abnormal lung-function growth that could lead to chronic airflow obstruction in adulthood.
Funding for this project was provided by grants from the Parker B. Francis Foundation, the National Institutes of Health, the National Human Genome Research Institute, and the Human Frontier Science Program Organization. Dr. McGeachie reported grant support from one of the funding sources during the conduct of the study. Nineteen coauthors reported they had nothing to disclose and the remainder either reported grant support or ties to industry sources.
Key clinical point: Abnormal longitudinal patterns of lung-function growth in young asthma patients are associated with specific risk factors and may be related to development of COPD.
Major finding: Those with reduced lung growth and an early decline had a lower forced expiratory volume in 1 second lung function in childhood and were more likely to be male.
Data sources: A subset of participants from the Childhood Asthma Management Program cohort.
Disclosures: Funding for this project was provided by grants from the Parker B. Francis Foundation, the National Institutes of Health, the National Human Genome Research Institute, and the Human Frontier Science Program Organization. Dr. McGeachie reported grant support from one of the funding sources during the conduct of the study. Nineteen coauthors reported they had nothing to disclose and the remainder either reported grant support or ties to industry sources.
Fresh Press: ACS Surgery News May issue is live on the website!
The digital May issue of ACS Surgery News is available online. Use the mobile app to download or view as a pdf.
The growing problem with reproducibility and sloppy use of statistical tools in biomedical research is the topic of this month’s feature. Even lab mice can be the sources of misleading research results. Dr. Peter Angelos reflects on what all this can mean for surgical research.
Don’t miss Dr. Tyler Hughes’s lighthearted look at a fictional surgeon of the future, Dr. ‘Bones’ McCoy, and how some of Dr. McCoy’s challenges are all too familiar to today’s surgeons.
This issue has news from on-site coverage of the annual meetings of the American Association of Endocrine Surgeons, the American Surgical Association, and the Central Surgical Association.
The digital May issue of ACS Surgery News is available online. Use the mobile app to download or view as a pdf.
The growing problem with reproducibility and sloppy use of statistical tools in biomedical research is the topic of this month’s feature. Even lab mice can be the sources of misleading research results. Dr. Peter Angelos reflects on what all this can mean for surgical research.
Don’t miss Dr. Tyler Hughes’s lighthearted look at a fictional surgeon of the future, Dr. ‘Bones’ McCoy, and how some of Dr. McCoy’s challenges are all too familiar to today’s surgeons.
This issue has news from on-site coverage of the annual meetings of the American Association of Endocrine Surgeons, the American Surgical Association, and the Central Surgical Association.
The digital May issue of ACS Surgery News is available online. Use the mobile app to download or view as a pdf.
The growing problem with reproducibility and sloppy use of statistical tools in biomedical research is the topic of this month’s feature. Even lab mice can be the sources of misleading research results. Dr. Peter Angelos reflects on what all this can mean for surgical research.
Don’t miss Dr. Tyler Hughes’s lighthearted look at a fictional surgeon of the future, Dr. ‘Bones’ McCoy, and how some of Dr. McCoy’s challenges are all too familiar to today’s surgeons.
This issue has news from on-site coverage of the annual meetings of the American Association of Endocrine Surgeons, the American Surgical Association, and the Central Surgical Association.
Alastair Noyce, MBBS, PhD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Rhythm control may be best for atrial fib in HFpEF
CHICAGO – Atrial fibrillation with good heart rate control in patients who have heart failure with preserved ejection fraction is independently associated with exercise intolerance, impaired contractile reserve, and a sharply higher mortality rate than in matched HFpEF patients without the arrhythmia, a retrospective analysis showed.
“Our study, the largest of its kind, provides mechanistic evidence from cardiopulmonary testing that a rhythm control strategy may potentially improve peak exercise capacity and survival in this patient population, a finding that of course requires future prospective appraisal in randomized trials comparing rate and rhythm control of atrial fibrillation in HFpEF,” Dr. Mohamed Badreldin Elshazly reported at the annual meeting of the American College of Cardiology.
In the meantime, his study also shows the useful role cardiopulmonary stress testing can play in the setting of atrial fibrillation (AF) in HFpEF, he added.
“Cardiopulmonary stress tests are cheap and easy to do. They’re a big asset for personalized medicine. Using an objective measure like cardiopulmonary stress testing to define the physiologic and hemodynamic consequences of atrial fibrillation in individual patients may help identify those in whom rhythm control may improve exercise tolerance and quality of life, and those who may be okay with rate control,” according to Dr. Elshazly of the Cleveland Clinic.
He noted that while it’s well established that atrial fibrillation is associated with exercise intolerance in patients with heart failure with reduced ejection fraction (HFrEF) and that restoration of sinus rhythm in such patients has a positive impact on exercise hemodynamics, symptom severity, and quality of life, the situation is murkier regarding AF in patients with HFpEF. Prior studies were generally small and unable to establish whether AF was independently associated with exercise intolerance or if HFpEF patients who developed AF were sicker and higher risk.
He presented a retrospective, case-control study in a cohort of 1,825 patients with HFpEF referred for maximal, symptom-limited cardiopulmonary stress testing at the Cleveland Clinic. Among these were 242 patients with AF. They were extensively propensity matched – including on the basis of heart failure etiology – to 484 HFpEF patients without AF.
“That’s what makes our study strong. We were the first to be able to do propensity matching and therefore account for other risk factors in our analysis,” Dr. Elshazly explained.
The investigators measured peak oxygen uptake (VO2), the minute ventilation–carbon dioxide production relationship (VE/VCO2) as an indicator of ventilatory efficiency, metabolic equivalents (METS), ventilatory anaerobic threshold, circulatory power as a proxy for cardiac power, peak oxygen pulse as a surrogate for stroke volume, and resting and peak heart rate and systolic blood pressure. The patients with AF were in fibrillation at the time of their cardiopulmonary stress testing.
The HFpEF patients with AF had a mean resting heart rate of 70 beats per minute and a peak rate of 130 bpm. This group showed evidence of impaired peak exercise tolerance as reflected in lower peak VO2, oxygen pulse, and circulatory power at peak exercise. Their VE/VCO2 was higher, indicating impaired ventilatory efficiency. Notably, however, their submaximal exercise capacity was similar to the non-AF controls.
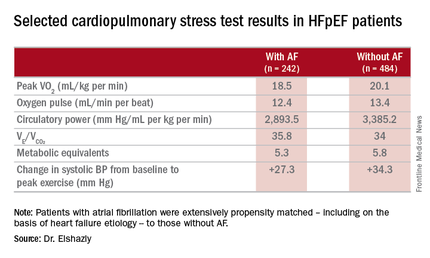
“Atrial fibrillation in these patients is really more of a disease that shows itself in patients when you take them to their peak exercise capacity,” he observed.
All-cause mortality was significantly higher in the AF as compared with no-AF patients with HFpEF. The mortality curves separated early and the divergence grew larger over the course of 8 years of follow-up.
One audience member pointed out that the large mortality difference between the two groups seems disproportionate to the rather modest differences in exercise capacity.
“It brings up an interesting point,” Dr. Elshazly replied. “Maybe the increase in total mortality that we see is being driven by other things besides cardiovascular mortality. Our data doesn’t capture the specific cause of death, be it cancer, for example, but it does raise the idea that this mortality difference is not all driven by cardiovascular mortality, but by atrial fibrillation.”
Dr. Elshazly reported having no financial conflicts of interest regarding his institutionally supported study.
CHICAGO – Atrial fibrillation with good heart rate control in patients who have heart failure with preserved ejection fraction is independently associated with exercise intolerance, impaired contractile reserve, and a sharply higher mortality rate than in matched HFpEF patients without the arrhythmia, a retrospective analysis showed.
“Our study, the largest of its kind, provides mechanistic evidence from cardiopulmonary testing that a rhythm control strategy may potentially improve peak exercise capacity and survival in this patient population, a finding that of course requires future prospective appraisal in randomized trials comparing rate and rhythm control of atrial fibrillation in HFpEF,” Dr. Mohamed Badreldin Elshazly reported at the annual meeting of the American College of Cardiology.
In the meantime, his study also shows the useful role cardiopulmonary stress testing can play in the setting of atrial fibrillation (AF) in HFpEF, he added.
“Cardiopulmonary stress tests are cheap and easy to do. They’re a big asset for personalized medicine. Using an objective measure like cardiopulmonary stress testing to define the physiologic and hemodynamic consequences of atrial fibrillation in individual patients may help identify those in whom rhythm control may improve exercise tolerance and quality of life, and those who may be okay with rate control,” according to Dr. Elshazly of the Cleveland Clinic.
He noted that while it’s well established that atrial fibrillation is associated with exercise intolerance in patients with heart failure with reduced ejection fraction (HFrEF) and that restoration of sinus rhythm in such patients has a positive impact on exercise hemodynamics, symptom severity, and quality of life, the situation is murkier regarding AF in patients with HFpEF. Prior studies were generally small and unable to establish whether AF was independently associated with exercise intolerance or if HFpEF patients who developed AF were sicker and higher risk.
He presented a retrospective, case-control study in a cohort of 1,825 patients with HFpEF referred for maximal, symptom-limited cardiopulmonary stress testing at the Cleveland Clinic. Among these were 242 patients with AF. They were extensively propensity matched – including on the basis of heart failure etiology – to 484 HFpEF patients without AF.
“That’s what makes our study strong. We were the first to be able to do propensity matching and therefore account for other risk factors in our analysis,” Dr. Elshazly explained.
The investigators measured peak oxygen uptake (VO2), the minute ventilation–carbon dioxide production relationship (VE/VCO2) as an indicator of ventilatory efficiency, metabolic equivalents (METS), ventilatory anaerobic threshold, circulatory power as a proxy for cardiac power, peak oxygen pulse as a surrogate for stroke volume, and resting and peak heart rate and systolic blood pressure. The patients with AF were in fibrillation at the time of their cardiopulmonary stress testing.
The HFpEF patients with AF had a mean resting heart rate of 70 beats per minute and a peak rate of 130 bpm. This group showed evidence of impaired peak exercise tolerance as reflected in lower peak VO2, oxygen pulse, and circulatory power at peak exercise. Their VE/VCO2 was higher, indicating impaired ventilatory efficiency. Notably, however, their submaximal exercise capacity was similar to the non-AF controls.

“Atrial fibrillation in these patients is really more of a disease that shows itself in patients when you take them to their peak exercise capacity,” he observed.
All-cause mortality was significantly higher in the AF as compared with no-AF patients with HFpEF. The mortality curves separated early and the divergence grew larger over the course of 8 years of follow-up.
One audience member pointed out that the large mortality difference between the two groups seems disproportionate to the rather modest differences in exercise capacity.
“It brings up an interesting point,” Dr. Elshazly replied. “Maybe the increase in total mortality that we see is being driven by other things besides cardiovascular mortality. Our data doesn’t capture the specific cause of death, be it cancer, for example, but it does raise the idea that this mortality difference is not all driven by cardiovascular mortality, but by atrial fibrillation.”
Dr. Elshazly reported having no financial conflicts of interest regarding his institutionally supported study.
CHICAGO – Atrial fibrillation with good heart rate control in patients who have heart failure with preserved ejection fraction is independently associated with exercise intolerance, impaired contractile reserve, and a sharply higher mortality rate than in matched HFpEF patients without the arrhythmia, a retrospective analysis showed.
“Our study, the largest of its kind, provides mechanistic evidence from cardiopulmonary testing that a rhythm control strategy may potentially improve peak exercise capacity and survival in this patient population, a finding that of course requires future prospective appraisal in randomized trials comparing rate and rhythm control of atrial fibrillation in HFpEF,” Dr. Mohamed Badreldin Elshazly reported at the annual meeting of the American College of Cardiology.
In the meantime, his study also shows the useful role cardiopulmonary stress testing can play in the setting of atrial fibrillation (AF) in HFpEF, he added.
“Cardiopulmonary stress tests are cheap and easy to do. They’re a big asset for personalized medicine. Using an objective measure like cardiopulmonary stress testing to define the physiologic and hemodynamic consequences of atrial fibrillation in individual patients may help identify those in whom rhythm control may improve exercise tolerance and quality of life, and those who may be okay with rate control,” according to Dr. Elshazly of the Cleveland Clinic.
He noted that while it’s well established that atrial fibrillation is associated with exercise intolerance in patients with heart failure with reduced ejection fraction (HFrEF) and that restoration of sinus rhythm in such patients has a positive impact on exercise hemodynamics, symptom severity, and quality of life, the situation is murkier regarding AF in patients with HFpEF. Prior studies were generally small and unable to establish whether AF was independently associated with exercise intolerance or if HFpEF patients who developed AF were sicker and higher risk.
He presented a retrospective, case-control study in a cohort of 1,825 patients with HFpEF referred for maximal, symptom-limited cardiopulmonary stress testing at the Cleveland Clinic. Among these were 242 patients with AF. They were extensively propensity matched – including on the basis of heart failure etiology – to 484 HFpEF patients without AF.
“That’s what makes our study strong. We were the first to be able to do propensity matching and therefore account for other risk factors in our analysis,” Dr. Elshazly explained.
The investigators measured peak oxygen uptake (VO2), the minute ventilation–carbon dioxide production relationship (VE/VCO2) as an indicator of ventilatory efficiency, metabolic equivalents (METS), ventilatory anaerobic threshold, circulatory power as a proxy for cardiac power, peak oxygen pulse as a surrogate for stroke volume, and resting and peak heart rate and systolic blood pressure. The patients with AF were in fibrillation at the time of their cardiopulmonary stress testing.
The HFpEF patients with AF had a mean resting heart rate of 70 beats per minute and a peak rate of 130 bpm. This group showed evidence of impaired peak exercise tolerance as reflected in lower peak VO2, oxygen pulse, and circulatory power at peak exercise. Their VE/VCO2 was higher, indicating impaired ventilatory efficiency. Notably, however, their submaximal exercise capacity was similar to the non-AF controls.

“Atrial fibrillation in these patients is really more of a disease that shows itself in patients when you take them to their peak exercise capacity,” he observed.
All-cause mortality was significantly higher in the AF as compared with no-AF patients with HFpEF. The mortality curves separated early and the divergence grew larger over the course of 8 years of follow-up.
One audience member pointed out that the large mortality difference between the two groups seems disproportionate to the rather modest differences in exercise capacity.
“It brings up an interesting point,” Dr. Elshazly replied. “Maybe the increase in total mortality that we see is being driven by other things besides cardiovascular mortality. Our data doesn’t capture the specific cause of death, be it cancer, for example, but it does raise the idea that this mortality difference is not all driven by cardiovascular mortality, but by atrial fibrillation.”
Dr. Elshazly reported having no financial conflicts of interest regarding his institutionally supported study.
AT ACC 16
Key clinical point: Atrial fibrillation in patients with heart failure with preserved ejection fraction is associated with exercise intolerance and increased mortality.
Major finding: Mean peak VO2 was 18.5 mL/kg per minute in patients with HFpEF and atrial fibrillation, significantly less than the 20.1 mL/kg per minute in controls.
Data source: A retrospective, single-institution study of cardiopulmonary stress test findings and 8-year mortality in 242 patients with HFpEF and atrial fibrillation and 484 propensity-matched controls with HFpEF and no arrhythmia.
Disclosures: The presenter reported having no financial conflicts of interest regarding his institutionally supported study.
Refined technique eliminates phrenic nerve palsy with second-generation cryoablation device
SAN FRANCISCO – Lower doses and conservative applications eliminated phrenic nerve palsy with Medtronic’s Arctic Front Advance cryoballoon ablation catheter at the University of Florida, Gainesville.
Cardiologists there were accustomed to performing atrial fibrillation pulmonary vein isolation with the first-generation device – the Arctic Front – when they switched to the second-generation Advance catheter in 2012; they had 2 phrenic nerve palsies in the first 33 patients (6%), a doubling from the 2 cases in 74 patients (2.7%) with the first-generation device.
The second-generation catheter is more powerful, with a cooling jet in the front of the balloon that delivers colder temperatures deeper into pulmonary veins. “We realized we needed to change the way we were using these catheters. You can use them safely, but you have to respect” their power, said lead investigator Dr. Robert Gibson.
“We reduced freezing times from 240 seconds to 180 seconds, and made that a hard rule. We limited the number of ablations” to two complete occlusions per vein, down from four to seven with the first-generation Arctic Front. “We also implemented a nadir cutoff to stop ablation when catheter temperatures fell below –55° C, and we tried to stay as proximal as possible to the pulmonary vein antra while still maintaining complete occlusion.” To help with that, cardiologists stopped using the 23-mm catheter, opting instead for the 28-mm catheter, Dr. Gibson said at the Heart Rhythm Society annual meeting.
Since making the changes, the team has performed 140 ablations, and there has not been a single phrenic nerve palsy. “We haven’t experienced a diaphragm paralysis” with the new approach. “We are very happy with these results. The changes make physiologic sense. You stay back; you use fewer freezes,” he said.
Phrenic nerve injury also decreased, from 3 cases in the first 33 second-generation patients (9%) to 9 in the 140 (6.4%) with the refined technique. Total phrenic nerve complications are now fewer in Gainesville than with the original, less powerful first-generation Arctic Front.
The team didn’t report ablation success with their new approach, but a 2015 review of Arctic Front Advance in more than 3,000 patients suggested long-term success with similar refinements (Heart Rhythm. 2015 Jul;12[7]:1658-66).
Patients in the University of Florida review were in their early 60s, on average, and about two-thirds were men. About 30% had prior ablations. Phrenic nerve injury was determined by continuous phrenic nerve stimulation and manual diaphragm palpation during cryoablation.
The investigators had no relevant financial disclosures. Medtronic helped with the statistical analysis.
SAN FRANCISCO – Lower doses and conservative applications eliminated phrenic nerve palsy with Medtronic’s Arctic Front Advance cryoballoon ablation catheter at the University of Florida, Gainesville.
Cardiologists there were accustomed to performing atrial fibrillation pulmonary vein isolation with the first-generation device – the Arctic Front – when they switched to the second-generation Advance catheter in 2012; they had 2 phrenic nerve palsies in the first 33 patients (6%), a doubling from the 2 cases in 74 patients (2.7%) with the first-generation device.
The second-generation catheter is more powerful, with a cooling jet in the front of the balloon that delivers colder temperatures deeper into pulmonary veins. “We realized we needed to change the way we were using these catheters. You can use them safely, but you have to respect” their power, said lead investigator Dr. Robert Gibson.
“We reduced freezing times from 240 seconds to 180 seconds, and made that a hard rule. We limited the number of ablations” to two complete occlusions per vein, down from four to seven with the first-generation Arctic Front. “We also implemented a nadir cutoff to stop ablation when catheter temperatures fell below –55° C, and we tried to stay as proximal as possible to the pulmonary vein antra while still maintaining complete occlusion.” To help with that, cardiologists stopped using the 23-mm catheter, opting instead for the 28-mm catheter, Dr. Gibson said at the Heart Rhythm Society annual meeting.
Since making the changes, the team has performed 140 ablations, and there has not been a single phrenic nerve palsy. “We haven’t experienced a diaphragm paralysis” with the new approach. “We are very happy with these results. The changes make physiologic sense. You stay back; you use fewer freezes,” he said.
Phrenic nerve injury also decreased, from 3 cases in the first 33 second-generation patients (9%) to 9 in the 140 (6.4%) with the refined technique. Total phrenic nerve complications are now fewer in Gainesville than with the original, less powerful first-generation Arctic Front.
The team didn’t report ablation success with their new approach, but a 2015 review of Arctic Front Advance in more than 3,000 patients suggested long-term success with similar refinements (Heart Rhythm. 2015 Jul;12[7]:1658-66).
Patients in the University of Florida review were in their early 60s, on average, and about two-thirds were men. About 30% had prior ablations. Phrenic nerve injury was determined by continuous phrenic nerve stimulation and manual diaphragm palpation during cryoablation.
The investigators had no relevant financial disclosures. Medtronic helped with the statistical analysis.
SAN FRANCISCO – Lower doses and conservative applications eliminated phrenic nerve palsy with Medtronic’s Arctic Front Advance cryoballoon ablation catheter at the University of Florida, Gainesville.
Cardiologists there were accustomed to performing atrial fibrillation pulmonary vein isolation with the first-generation device – the Arctic Front – when they switched to the second-generation Advance catheter in 2012; they had 2 phrenic nerve palsies in the first 33 patients (6%), a doubling from the 2 cases in 74 patients (2.7%) with the first-generation device.
The second-generation catheter is more powerful, with a cooling jet in the front of the balloon that delivers colder temperatures deeper into pulmonary veins. “We realized we needed to change the way we were using these catheters. You can use them safely, but you have to respect” their power, said lead investigator Dr. Robert Gibson.
“We reduced freezing times from 240 seconds to 180 seconds, and made that a hard rule. We limited the number of ablations” to two complete occlusions per vein, down from four to seven with the first-generation Arctic Front. “We also implemented a nadir cutoff to stop ablation when catheter temperatures fell below –55° C, and we tried to stay as proximal as possible to the pulmonary vein antra while still maintaining complete occlusion.” To help with that, cardiologists stopped using the 23-mm catheter, opting instead for the 28-mm catheter, Dr. Gibson said at the Heart Rhythm Society annual meeting.
Since making the changes, the team has performed 140 ablations, and there has not been a single phrenic nerve palsy. “We haven’t experienced a diaphragm paralysis” with the new approach. “We are very happy with these results. The changes make physiologic sense. You stay back; you use fewer freezes,” he said.
Phrenic nerve injury also decreased, from 3 cases in the first 33 second-generation patients (9%) to 9 in the 140 (6.4%) with the refined technique. Total phrenic nerve complications are now fewer in Gainesville than with the original, less powerful first-generation Arctic Front.
The team didn’t report ablation success with their new approach, but a 2015 review of Arctic Front Advance in more than 3,000 patients suggested long-term success with similar refinements (Heart Rhythm. 2015 Jul;12[7]:1658-66).
Patients in the University of Florida review were in their early 60s, on average, and about two-thirds were men. About 30% had prior ablations. Phrenic nerve injury was determined by continuous phrenic nerve stimulation and manual diaphragm palpation during cryoablation.
The investigators had no relevant financial disclosures. Medtronic helped with the statistical analysis.
AT HEART RHYTHM 2016
Key clinical point: Second-generation cryoablation devices require a lighter touch to avoid phrenic nerve palsy.
Major finding: A conservative approach dropped the phrenic nerve palsy rate from 6% to 0% (P = .025).
Data source: A review of 247 cryoballoon ablations for paroxysmal atrial fibrillation
Disclosures: The investigators had no relevant financial disclosures. Medtronic helped with the statistical analysis.
Epstein-Barr virus DNA in plasma reliably detects EBV-positive lymphoproliferative disorders
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
FROM BLOOD
Key clinical point: Epstein-Barr virus DNA was a more specific indicator of EBV+ diseases when detected in plasma, as opposed to peripheral blood mononuclear cells.
Major finding: Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but 54% had EBV in PBMCs.
Data source: Viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients.
Disclosures: The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
FDA: Olanzapine can cause serious skin reaction
Olanzapine can cause a rare but serious skin reaction that can affect other parts of the body, according to a Food and Drug Administration safety alert released May 10.
The condition linked to all products containing the second-generation antipsychotic is called Drug Reaction with Eosinophilia and Systemic Symptoms, or DRESS. Symptoms of DRESS include a rash that can spread to all parts of the body, fever, swollen lymph nodes, and swelling. In addition, DRESS can result in injury to the liver, kidneys, lungs, heart, or pancreas, and it also can lead to death. The mortality tied to DRESS can reach 10%, the FDA said.
The FDA Adverse Event Reporting System database has identified 23 cases worldwide of DRESS resulting from olanzapine since 1996, including one patient who died. Currently, the only way to treat DRESS is to withdraw the drug promptly. “Health care professionals should immediately stop treatment with olanzapine if DRESS is suspected,” the safety alert states. “The important ways to manage DRESS are early recognition of the syndrome, discontinuation of the offending agent as soon as possible, and supportive care.”
Olanzapine, used to treat schizophrenia and manic episodes of bipolar disorder, is available in generic versions. The medication also is available under the brand names Zyprexa, Zyprexa Zydis, Zyprexa Relprevv, and Symbyax. The agency said it would add a warning describing DRESS to the labels of drugs containing olanzapine.
Read the full safety alert on the FDA website.
Olanzapine can cause a rare but serious skin reaction that can affect other parts of the body, according to a Food and Drug Administration safety alert released May 10.
The condition linked to all products containing the second-generation antipsychotic is called Drug Reaction with Eosinophilia and Systemic Symptoms, or DRESS. Symptoms of DRESS include a rash that can spread to all parts of the body, fever, swollen lymph nodes, and swelling. In addition, DRESS can result in injury to the liver, kidneys, lungs, heart, or pancreas, and it also can lead to death. The mortality tied to DRESS can reach 10%, the FDA said.
The FDA Adverse Event Reporting System database has identified 23 cases worldwide of DRESS resulting from olanzapine since 1996, including one patient who died. Currently, the only way to treat DRESS is to withdraw the drug promptly. “Health care professionals should immediately stop treatment with olanzapine if DRESS is suspected,” the safety alert states. “The important ways to manage DRESS are early recognition of the syndrome, discontinuation of the offending agent as soon as possible, and supportive care.”
Olanzapine, used to treat schizophrenia and manic episodes of bipolar disorder, is available in generic versions. The medication also is available under the brand names Zyprexa, Zyprexa Zydis, Zyprexa Relprevv, and Symbyax. The agency said it would add a warning describing DRESS to the labels of drugs containing olanzapine.
Read the full safety alert on the FDA website.
Olanzapine can cause a rare but serious skin reaction that can affect other parts of the body, according to a Food and Drug Administration safety alert released May 10.
The condition linked to all products containing the second-generation antipsychotic is called Drug Reaction with Eosinophilia and Systemic Symptoms, or DRESS. Symptoms of DRESS include a rash that can spread to all parts of the body, fever, swollen lymph nodes, and swelling. In addition, DRESS can result in injury to the liver, kidneys, lungs, heart, or pancreas, and it also can lead to death. The mortality tied to DRESS can reach 10%, the FDA said.
The FDA Adverse Event Reporting System database has identified 23 cases worldwide of DRESS resulting from olanzapine since 1996, including one patient who died. Currently, the only way to treat DRESS is to withdraw the drug promptly. “Health care professionals should immediately stop treatment with olanzapine if DRESS is suspected,” the safety alert states. “The important ways to manage DRESS are early recognition of the syndrome, discontinuation of the offending agent as soon as possible, and supportive care.”
Olanzapine, used to treat schizophrenia and manic episodes of bipolar disorder, is available in generic versions. The medication also is available under the brand names Zyprexa, Zyprexa Zydis, Zyprexa Relprevv, and Symbyax. The agency said it would add a warning describing DRESS to the labels of drugs containing olanzapine.
Read the full safety alert on the FDA website.
Red Facial Lesions Are No Day at the Beach
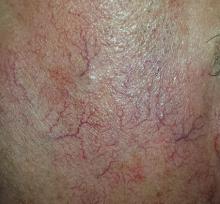
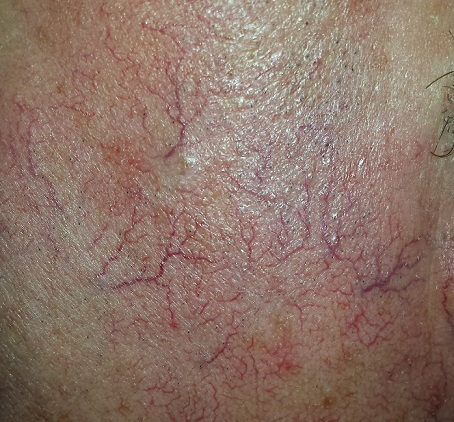
A 70-year-old man is seen for “broken blood vessels” on his face. The lesions were recently noted by a relative who had not seen him in years.
The patient has a pronounced history of sun overexposure as a result of his job and his hobbies, which include fishing and hunting. As a younger man, he enjoyed waterskiing and going to the lake almost every weekend during the warmer months.
He is in decent health apart from the lesions and has no other skin complaints.
EXAMINATION
The sides of the patient’s face are covered with linear, red, dilated blood vessels running in jagged patterns. The lesions are asymptomatic and range from 0.5 to 1.0 mm in diameter; some are several centimeters long. They are readily blanchable with digital pressure, though none are palpable.
Far more blood vessels are seen on the patient’s left side than on his right. The underlying skin is quite fair and thin. In some locations (eg, the lateral forehead), there are patches of white skin free of surface adnexa (ie pores, skin lines, or hair follicles).
His dorsal arms are very rough, with hundreds of solar lentigines, while the skin on his volar arms is relatively unaffected.
What is the diagnosis?
DISCUSSION
This collection of findings is called dermatoheliosis (DHe), literally “sun-skin condition,” caused by overexposure to UV light. Telangiectasias, one manifestation, have many potential causes, but sun exposure is the most common—especially in those whose skin burns easily and frequently.
These blood vessels are simply normal surface vasculature made more obvious by two factors: First, chronic sun exposure overheats the skin for prolonged periods, leading to persistent vasodilatation that eventually becomes fixed. Second, solar atrophy may occur when the skin thins from sun overexposure, making typically invisible structures apparent.
DHe can also manifest as actinic keratosis, weathering of the skin, solar elastosis, and sun-caused skin cancers (basal and squamous cell carcinomas, melanoma, etc). The condition is incredibly common.
Many causes of telangiectasias are unrelated to sun exposure. Other processes that thin the skin—such as radiation therapy or injudicious use of topical steroids—may produce telangiectasias. Further examples include spider vessels on legs due to venous insufficiency and newly acquired vascular lesions resulting from pregnancy or liver cirrhosis. There are also two unusual but significant conditions that involve telangiectasias: a variant of systemic sclerosis called CREST syndrome (calcinosis, Raynaud’s, esophageal dysmotility, sclerodactyly, and telangiectasias) and hereditary hemorrhagic telangiectasias (also known eponymically as Osler-Weber-Rendu syndrome).
Treatment for sun-caused telangiectasias includes laser and electrodessication.
TAKE-HOME LEARNING POINTS
• Telangiectasias are permanently dilated surface vasculature usually seen on the most prominently sun-exposed areas of the skin. The left face and arm are often more heavily affected than the right, since they are closer to the car window during driving.
• Telangiectasias are only one of a number of sun-caused skin changes that are collectively termed dermatoheliosis (DHe).
• Telangiectasias are important because they identify patients at risk for sun-caused skin cancers, such as basal or squamous cell carcinomas and melanoma.
• Other potential causes of telangiectasias include injudicious use of topical steroids, focal radiation therapy, pregnancy, liver disease, and rare conditions such as CREST syndrome and hereditary hemorrhagic telangiectasias.
A 70-year-old man is seen for “broken blood vessels” on his face. The lesions were recently noted by a relative who had not seen him in years.
The patient has a pronounced history of sun overexposure as a result of his job and his hobbies, which include fishing and hunting. As a younger man, he enjoyed waterskiing and going to the lake almost every weekend during the warmer months.
He is in decent health apart from the lesions and has no other skin complaints.
EXAMINATION
The sides of the patient’s face are covered with linear, red, dilated blood vessels running in jagged patterns. The lesions are asymptomatic and range from 0.5 to 1.0 mm in diameter; some are several centimeters long. They are readily blanchable with digital pressure, though none are palpable.
Far more blood vessels are seen on the patient’s left side than on his right. The underlying skin is quite fair and thin. In some locations (eg, the lateral forehead), there are patches of white skin free of surface adnexa (ie pores, skin lines, or hair follicles).
His dorsal arms are very rough, with hundreds of solar lentigines, while the skin on his volar arms is relatively unaffected.
What is the diagnosis?
DISCUSSION
This collection of findings is called dermatoheliosis (DHe), literally “sun-skin condition,” caused by overexposure to UV light. Telangiectasias, one manifestation, have many potential causes, but sun exposure is the most common—especially in those whose skin burns easily and frequently.
These blood vessels are simply normal surface vasculature made more obvious by two factors: First, chronic sun exposure overheats the skin for prolonged periods, leading to persistent vasodilatation that eventually becomes fixed. Second, solar atrophy may occur when the skin thins from sun overexposure, making typically invisible structures apparent.
DHe can also manifest as actinic keratosis, weathering of the skin, solar elastosis, and sun-caused skin cancers (basal and squamous cell carcinomas, melanoma, etc). The condition is incredibly common.
Many causes of telangiectasias are unrelated to sun exposure. Other processes that thin the skin—such as radiation therapy or injudicious use of topical steroids—may produce telangiectasias. Further examples include spider vessels on legs due to venous insufficiency and newly acquired vascular lesions resulting from pregnancy or liver cirrhosis. There are also two unusual but significant conditions that involve telangiectasias: a variant of systemic sclerosis called CREST syndrome (calcinosis, Raynaud’s, esophageal dysmotility, sclerodactyly, and telangiectasias) and hereditary hemorrhagic telangiectasias (also known eponymically as Osler-Weber-Rendu syndrome).
Treatment for sun-caused telangiectasias includes laser and electrodessication.
TAKE-HOME LEARNING POINTS
• Telangiectasias are permanently dilated surface vasculature usually seen on the most prominently sun-exposed areas of the skin. The left face and arm are often more heavily affected than the right, since they are closer to the car window during driving.
• Telangiectasias are only one of a number of sun-caused skin changes that are collectively termed dermatoheliosis (DHe).
• Telangiectasias are important because they identify patients at risk for sun-caused skin cancers, such as basal or squamous cell carcinomas and melanoma.
• Other potential causes of telangiectasias include injudicious use of topical steroids, focal radiation therapy, pregnancy, liver disease, and rare conditions such as CREST syndrome and hereditary hemorrhagic telangiectasias.
A 70-year-old man is seen for “broken blood vessels” on his face. The lesions were recently noted by a relative who had not seen him in years.
The patient has a pronounced history of sun overexposure as a result of his job and his hobbies, which include fishing and hunting. As a younger man, he enjoyed waterskiing and going to the lake almost every weekend during the warmer months.
He is in decent health apart from the lesions and has no other skin complaints.
EXAMINATION
The sides of the patient’s face are covered with linear, red, dilated blood vessels running in jagged patterns. The lesions are asymptomatic and range from 0.5 to 1.0 mm in diameter; some are several centimeters long. They are readily blanchable with digital pressure, though none are palpable.
Far more blood vessels are seen on the patient’s left side than on his right. The underlying skin is quite fair and thin. In some locations (eg, the lateral forehead), there are patches of white skin free of surface adnexa (ie pores, skin lines, or hair follicles).
His dorsal arms are very rough, with hundreds of solar lentigines, while the skin on his volar arms is relatively unaffected.
What is the diagnosis?
DISCUSSION
This collection of findings is called dermatoheliosis (DHe), literally “sun-skin condition,” caused by overexposure to UV light. Telangiectasias, one manifestation, have many potential causes, but sun exposure is the most common—especially in those whose skin burns easily and frequently.
These blood vessels are simply normal surface vasculature made more obvious by two factors: First, chronic sun exposure overheats the skin for prolonged periods, leading to persistent vasodilatation that eventually becomes fixed. Second, solar atrophy may occur when the skin thins from sun overexposure, making typically invisible structures apparent.
DHe can also manifest as actinic keratosis, weathering of the skin, solar elastosis, and sun-caused skin cancers (basal and squamous cell carcinomas, melanoma, etc). The condition is incredibly common.
Many causes of telangiectasias are unrelated to sun exposure. Other processes that thin the skin—such as radiation therapy or injudicious use of topical steroids—may produce telangiectasias. Further examples include spider vessels on legs due to venous insufficiency and newly acquired vascular lesions resulting from pregnancy or liver cirrhosis. There are also two unusual but significant conditions that involve telangiectasias: a variant of systemic sclerosis called CREST syndrome (calcinosis, Raynaud’s, esophageal dysmotility, sclerodactyly, and telangiectasias) and hereditary hemorrhagic telangiectasias (also known eponymically as Osler-Weber-Rendu syndrome).
Treatment for sun-caused telangiectasias includes laser and electrodessication.
TAKE-HOME LEARNING POINTS
• Telangiectasias are permanently dilated surface vasculature usually seen on the most prominently sun-exposed areas of the skin. The left face and arm are often more heavily affected than the right, since they are closer to the car window during driving.
• Telangiectasias are only one of a number of sun-caused skin changes that are collectively termed dermatoheliosis (DHe).
• Telangiectasias are important because they identify patients at risk for sun-caused skin cancers, such as basal or squamous cell carcinomas and melanoma.
• Other potential causes of telangiectasias include injudicious use of topical steroids, focal radiation therapy, pregnancy, liver disease, and rare conditions such as CREST syndrome and hereditary hemorrhagic telangiectasias.