User login
Leadless dual-chamber pacemaker clears early safety, performance hurdles
Cardiology, well into the age of leadless pacemakers, could be headed for an age of leadless pacemaker systems in which various pacing functions are achieved by multiple implants that “talk” to each other.
Even now, a leadless two-part pacemaker system has shown it can safely achieve atrioventricular (AV) synchrony in patients with standard indications for a dual-chamber device, at least over the short term, suggests a prospective observational study. Currently available leadless pacemakers can stimulate only the right ventricle.
Experienced operators achieved a 98% implantation success rate in 300 patients who received an investigational dual-chamber leadless system, the AVEIR DR i2i (Abbott).
Its two separately implanted miniature pulse generators achieve AV synchrony via “beat-to-beat wireless bidirectional communication,” Daniel J. Cantillon, MD, said when presenting the study at the annual scientific sessions of the Heart Rhythm Societyin New Orleans.
The system seemed to work well regardless of the patient’s body orientation. “Sitting, supine, left lateral, right lateral, standing, normal walk, fast walk – we demonstrated robust AV synchrony in all of those positions and with movement,” said Dr. Cantillon, of the Cleveland Clinic.
Should the device be approved, it could “expand the use case for leadless cardiac pacing” to include atrial-only, ventricular-only, fully functional dual-chamber pacing scenarios.”
Dr. Cantillon is senior author on the study’s online publication in the New England Journal of Medicine, timed to coincide with his HRS presentation, with first author Reinoud E. Knops, MD, PhD, Amsterdam University Medical Center.
“The electrical performance of both the atrial and ventricular leadless pacemakers appears to be similar to that of transvenous dual-chamber pacemakers,” the published report states.
More data needed
The study is important and has “significant implications for our pacing field,” Jonathan P. Piccini, MD, MHS, said in an interview. It suggests that “dual-chamber pacing can be achieved with leadless technology” and “with a very high degree” of AV synchrony.
“Obviously, more data as the technology moves into clinical practice will be critical,” said Dr. Piccini, who directs cardiac electrophysiology at Duke University Medical Center, Durham, N.C. “We will also need to understand which patients are best served by leadless technology and which will be better served with traditional transvenous devices.”
The AVEIR DR i2i system consists of two leadless pulse generators for percutaneous implantation in the right atrium and right ventricle, respectively. They link like components of a wireless network to coordinate their separate sensing and rate-adaptive, AV-synchronous pacing functions.
The right ventricular implant “is physically identical to a commercially available single-chamber leadless pacemaker” from Abbott, the published report states.
Leadless pacemaker systems inherently avoid the two main sources of transvenous devices’ major complication – infection – by not requiring such leads or surgery for creating a pulse-generator subcutaneous pocket.
The first such systems consisted of one implant that could provide single-chamber ventricular pacing but not atrial pacing or AV synchronous pacing. The transcatheter single-chamber leadless Micra (Medtronic) for example, was approved in the United States in April 2016 for ventricular-only pacing.
A successor, the Micra AV, approved in 2020, was designed to simulate AV-synchronous pacing by stimulating the ventricle in sequence with mechanically sensed atrial contractions, as described by Dr. Cantillon and associates. But it could not directly pace the atrium, “rendering it inappropriate for patients with sinus-node dysfunction.”
The AVEIR DR i2i system doesn’t have those limitations. It was, however, associated with 35 device- or procedure-related complications in the study, of which the most common was procedural arrhythmia, “namely atrial fibrillation,” Dr. Cantillon said.
Atrial fibrillation can develop during implantation of pacemakers with transvenous leads but is generally terminated without being considered an important event. Yet the study classified it as a serious complication, inflating the complication rate, because “the patients had to be restored to sinus rhythm so we could assess the AV synchrony and also the atrial electrical performance,” he said.
Some of the devices dislodged from their implantation site within a month of the procedure, but “all of those patients were successfully managed percutaneously,” said Dr. Cantillon.
“The 1.7% dislodgement rate is something that we will need to keep an eye on, as embolization of devices is always a significant concern,” Dr. Piccini said. Still, the observed total complication rate “was certainly in line” with rates associated with conventional pacemaker implantation.
Reliable AV synchrony
Fred M. Kusumoto, MD, Mayo Clinic, Jacksonville, Fla., lauded what seems to be the system’s “incredibly reliable AV synchrony in different conditions, albeit in a very controlled environment.”
Of interest will be whether its performance, including maintenance of AV synchrony, holds up in “a more long-term evaluation in the outpatient setting,” said Dr. Kusumoto, speaking as the invited discussant for Dr. Cantillon’s presentation.
Also missing or in short supply from the study, he observed, are insights about long-term efficacy and complications, battery longevity, effectiveness of its rate-responsive capability, and any effect on clinical outcomes.
Local body network
Of the study’s 300 patients (mean age 69 years; 38% female) at 55 sites in Canada, Europe, and the United States, 63.3% had sinus-node dysfunction and 33.3% had AV block as their primary dual-chamber pacing indication; 298 were successfully implanted with both devices.
About 45% had a history of supraventricular arrhythmia, 4.3% had prior ventricular arrhythmia, and 20% had a history of arrhythmia ablation.
By 3 months, the group reported, the primary safety endpoint (freedom from device- or procedure-related serious adverse events) occurred in 90.3%, compared with the performance goal of 78% (P < .001).
The first of two primary performance endpoints (adequate atrial capture threshold and sensing amplitude by predefined criteria) was met in 90.2%, surpassing the 82.5% performance goal (P < .001).
The second primary performance goal (at least 70% AV synchrony with the patient sitting) was seen in 97.3% against the performance goal of 83% (P < .001).
What shouldn’t be “glossed over” from the study, Dr. Kusumoto offered, is that it’s possible to achieve a wireless connection “between two devices that are actually intracardiac.” That raises the prospect of a “local body network” that could be “expanded even more dramatically with other types of devices. I mean, think of the paradigm shift.”
The AVEIR DR i2i trial was funded by Abbott. Dr. Cantillon discloses receiving honoraria or fees for speaking or consulting from Abbott Laboratories, Boston Scientific, Biosense Webster, and Shockwave Medical, as well as holding royalty rights with AirStrip. Dr. Piccini has disclosed relationships with Abbott, Medtronic, Biotronik, Boston Scientific, and other drug and medical device companies. Dr. Kusumoto reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cardiology, well into the age of leadless pacemakers, could be headed for an age of leadless pacemaker systems in which various pacing functions are achieved by multiple implants that “talk” to each other.
Even now, a leadless two-part pacemaker system has shown it can safely achieve atrioventricular (AV) synchrony in patients with standard indications for a dual-chamber device, at least over the short term, suggests a prospective observational study. Currently available leadless pacemakers can stimulate only the right ventricle.
Experienced operators achieved a 98% implantation success rate in 300 patients who received an investigational dual-chamber leadless system, the AVEIR DR i2i (Abbott).
Its two separately implanted miniature pulse generators achieve AV synchrony via “beat-to-beat wireless bidirectional communication,” Daniel J. Cantillon, MD, said when presenting the study at the annual scientific sessions of the Heart Rhythm Societyin New Orleans.
The system seemed to work well regardless of the patient’s body orientation. “Sitting, supine, left lateral, right lateral, standing, normal walk, fast walk – we demonstrated robust AV synchrony in all of those positions and with movement,” said Dr. Cantillon, of the Cleveland Clinic.
Should the device be approved, it could “expand the use case for leadless cardiac pacing” to include atrial-only, ventricular-only, fully functional dual-chamber pacing scenarios.”
Dr. Cantillon is senior author on the study’s online publication in the New England Journal of Medicine, timed to coincide with his HRS presentation, with first author Reinoud E. Knops, MD, PhD, Amsterdam University Medical Center.
“The electrical performance of both the atrial and ventricular leadless pacemakers appears to be similar to that of transvenous dual-chamber pacemakers,” the published report states.
More data needed
The study is important and has “significant implications for our pacing field,” Jonathan P. Piccini, MD, MHS, said in an interview. It suggests that “dual-chamber pacing can be achieved with leadless technology” and “with a very high degree” of AV synchrony.
“Obviously, more data as the technology moves into clinical practice will be critical,” said Dr. Piccini, who directs cardiac electrophysiology at Duke University Medical Center, Durham, N.C. “We will also need to understand which patients are best served by leadless technology and which will be better served with traditional transvenous devices.”
The AVEIR DR i2i system consists of two leadless pulse generators for percutaneous implantation in the right atrium and right ventricle, respectively. They link like components of a wireless network to coordinate their separate sensing and rate-adaptive, AV-synchronous pacing functions.
The right ventricular implant “is physically identical to a commercially available single-chamber leadless pacemaker” from Abbott, the published report states.
Leadless pacemaker systems inherently avoid the two main sources of transvenous devices’ major complication – infection – by not requiring such leads or surgery for creating a pulse-generator subcutaneous pocket.
The first such systems consisted of one implant that could provide single-chamber ventricular pacing but not atrial pacing or AV synchronous pacing. The transcatheter single-chamber leadless Micra (Medtronic) for example, was approved in the United States in April 2016 for ventricular-only pacing.
A successor, the Micra AV, approved in 2020, was designed to simulate AV-synchronous pacing by stimulating the ventricle in sequence with mechanically sensed atrial contractions, as described by Dr. Cantillon and associates. But it could not directly pace the atrium, “rendering it inappropriate for patients with sinus-node dysfunction.”
The AVEIR DR i2i system doesn’t have those limitations. It was, however, associated with 35 device- or procedure-related complications in the study, of which the most common was procedural arrhythmia, “namely atrial fibrillation,” Dr. Cantillon said.
Atrial fibrillation can develop during implantation of pacemakers with transvenous leads but is generally terminated without being considered an important event. Yet the study classified it as a serious complication, inflating the complication rate, because “the patients had to be restored to sinus rhythm so we could assess the AV synchrony and also the atrial electrical performance,” he said.
Some of the devices dislodged from their implantation site within a month of the procedure, but “all of those patients were successfully managed percutaneously,” said Dr. Cantillon.
“The 1.7% dislodgement rate is something that we will need to keep an eye on, as embolization of devices is always a significant concern,” Dr. Piccini said. Still, the observed total complication rate “was certainly in line” with rates associated with conventional pacemaker implantation.
Reliable AV synchrony
Fred M. Kusumoto, MD, Mayo Clinic, Jacksonville, Fla., lauded what seems to be the system’s “incredibly reliable AV synchrony in different conditions, albeit in a very controlled environment.”
Of interest will be whether its performance, including maintenance of AV synchrony, holds up in “a more long-term evaluation in the outpatient setting,” said Dr. Kusumoto, speaking as the invited discussant for Dr. Cantillon’s presentation.
Also missing or in short supply from the study, he observed, are insights about long-term efficacy and complications, battery longevity, effectiveness of its rate-responsive capability, and any effect on clinical outcomes.
Local body network
Of the study’s 300 patients (mean age 69 years; 38% female) at 55 sites in Canada, Europe, and the United States, 63.3% had sinus-node dysfunction and 33.3% had AV block as their primary dual-chamber pacing indication; 298 were successfully implanted with both devices.
About 45% had a history of supraventricular arrhythmia, 4.3% had prior ventricular arrhythmia, and 20% had a history of arrhythmia ablation.
By 3 months, the group reported, the primary safety endpoint (freedom from device- or procedure-related serious adverse events) occurred in 90.3%, compared with the performance goal of 78% (P < .001).
The first of two primary performance endpoints (adequate atrial capture threshold and sensing amplitude by predefined criteria) was met in 90.2%, surpassing the 82.5% performance goal (P < .001).
The second primary performance goal (at least 70% AV synchrony with the patient sitting) was seen in 97.3% against the performance goal of 83% (P < .001).
What shouldn’t be “glossed over” from the study, Dr. Kusumoto offered, is that it’s possible to achieve a wireless connection “between two devices that are actually intracardiac.” That raises the prospect of a “local body network” that could be “expanded even more dramatically with other types of devices. I mean, think of the paradigm shift.”
The AVEIR DR i2i trial was funded by Abbott. Dr. Cantillon discloses receiving honoraria or fees for speaking or consulting from Abbott Laboratories, Boston Scientific, Biosense Webster, and Shockwave Medical, as well as holding royalty rights with AirStrip. Dr. Piccini has disclosed relationships with Abbott, Medtronic, Biotronik, Boston Scientific, and other drug and medical device companies. Dr. Kusumoto reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cardiology, well into the age of leadless pacemakers, could be headed for an age of leadless pacemaker systems in which various pacing functions are achieved by multiple implants that “talk” to each other.
Even now, a leadless two-part pacemaker system has shown it can safely achieve atrioventricular (AV) synchrony in patients with standard indications for a dual-chamber device, at least over the short term, suggests a prospective observational study. Currently available leadless pacemakers can stimulate only the right ventricle.
Experienced operators achieved a 98% implantation success rate in 300 patients who received an investigational dual-chamber leadless system, the AVEIR DR i2i (Abbott).
Its two separately implanted miniature pulse generators achieve AV synchrony via “beat-to-beat wireless bidirectional communication,” Daniel J. Cantillon, MD, said when presenting the study at the annual scientific sessions of the Heart Rhythm Societyin New Orleans.
The system seemed to work well regardless of the patient’s body orientation. “Sitting, supine, left lateral, right lateral, standing, normal walk, fast walk – we demonstrated robust AV synchrony in all of those positions and with movement,” said Dr. Cantillon, of the Cleveland Clinic.
Should the device be approved, it could “expand the use case for leadless cardiac pacing” to include atrial-only, ventricular-only, fully functional dual-chamber pacing scenarios.”
Dr. Cantillon is senior author on the study’s online publication in the New England Journal of Medicine, timed to coincide with his HRS presentation, with first author Reinoud E. Knops, MD, PhD, Amsterdam University Medical Center.
“The electrical performance of both the atrial and ventricular leadless pacemakers appears to be similar to that of transvenous dual-chamber pacemakers,” the published report states.
More data needed
The study is important and has “significant implications for our pacing field,” Jonathan P. Piccini, MD, MHS, said in an interview. It suggests that “dual-chamber pacing can be achieved with leadless technology” and “with a very high degree” of AV synchrony.
“Obviously, more data as the technology moves into clinical practice will be critical,” said Dr. Piccini, who directs cardiac electrophysiology at Duke University Medical Center, Durham, N.C. “We will also need to understand which patients are best served by leadless technology and which will be better served with traditional transvenous devices.”
The AVEIR DR i2i system consists of two leadless pulse generators for percutaneous implantation in the right atrium and right ventricle, respectively. They link like components of a wireless network to coordinate their separate sensing and rate-adaptive, AV-synchronous pacing functions.
The right ventricular implant “is physically identical to a commercially available single-chamber leadless pacemaker” from Abbott, the published report states.
Leadless pacemaker systems inherently avoid the two main sources of transvenous devices’ major complication – infection – by not requiring such leads or surgery for creating a pulse-generator subcutaneous pocket.
The first such systems consisted of one implant that could provide single-chamber ventricular pacing but not atrial pacing or AV synchronous pacing. The transcatheter single-chamber leadless Micra (Medtronic) for example, was approved in the United States in April 2016 for ventricular-only pacing.
A successor, the Micra AV, approved in 2020, was designed to simulate AV-synchronous pacing by stimulating the ventricle in sequence with mechanically sensed atrial contractions, as described by Dr. Cantillon and associates. But it could not directly pace the atrium, “rendering it inappropriate for patients with sinus-node dysfunction.”
The AVEIR DR i2i system doesn’t have those limitations. It was, however, associated with 35 device- or procedure-related complications in the study, of which the most common was procedural arrhythmia, “namely atrial fibrillation,” Dr. Cantillon said.
Atrial fibrillation can develop during implantation of pacemakers with transvenous leads but is generally terminated without being considered an important event. Yet the study classified it as a serious complication, inflating the complication rate, because “the patients had to be restored to sinus rhythm so we could assess the AV synchrony and also the atrial electrical performance,” he said.
Some of the devices dislodged from their implantation site within a month of the procedure, but “all of those patients were successfully managed percutaneously,” said Dr. Cantillon.
“The 1.7% dislodgement rate is something that we will need to keep an eye on, as embolization of devices is always a significant concern,” Dr. Piccini said. Still, the observed total complication rate “was certainly in line” with rates associated with conventional pacemaker implantation.
Reliable AV synchrony
Fred M. Kusumoto, MD, Mayo Clinic, Jacksonville, Fla., lauded what seems to be the system’s “incredibly reliable AV synchrony in different conditions, albeit in a very controlled environment.”
Of interest will be whether its performance, including maintenance of AV synchrony, holds up in “a more long-term evaluation in the outpatient setting,” said Dr. Kusumoto, speaking as the invited discussant for Dr. Cantillon’s presentation.
Also missing or in short supply from the study, he observed, are insights about long-term efficacy and complications, battery longevity, effectiveness of its rate-responsive capability, and any effect on clinical outcomes.
Local body network
Of the study’s 300 patients (mean age 69 years; 38% female) at 55 sites in Canada, Europe, and the United States, 63.3% had sinus-node dysfunction and 33.3% had AV block as their primary dual-chamber pacing indication; 298 were successfully implanted with both devices.
About 45% had a history of supraventricular arrhythmia, 4.3% had prior ventricular arrhythmia, and 20% had a history of arrhythmia ablation.
By 3 months, the group reported, the primary safety endpoint (freedom from device- or procedure-related serious adverse events) occurred in 90.3%, compared with the performance goal of 78% (P < .001).
The first of two primary performance endpoints (adequate atrial capture threshold and sensing amplitude by predefined criteria) was met in 90.2%, surpassing the 82.5% performance goal (P < .001).
The second primary performance goal (at least 70% AV synchrony with the patient sitting) was seen in 97.3% against the performance goal of 83% (P < .001).
What shouldn’t be “glossed over” from the study, Dr. Kusumoto offered, is that it’s possible to achieve a wireless connection “between two devices that are actually intracardiac.” That raises the prospect of a “local body network” that could be “expanded even more dramatically with other types of devices. I mean, think of the paradigm shift.”
The AVEIR DR i2i trial was funded by Abbott. Dr. Cantillon discloses receiving honoraria or fees for speaking or consulting from Abbott Laboratories, Boston Scientific, Biosense Webster, and Shockwave Medical, as well as holding royalty rights with AirStrip. Dr. Piccini has disclosed relationships with Abbott, Medtronic, Biotronik, Boston Scientific, and other drug and medical device companies. Dr. Kusumoto reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM HRS 2023
Researchers locate signals in brain related to chronic pain
a new study in Nature Neuroscience concluded.
The researchers used the devices on four patients who had felt endless nerve pain for more than a year. The devices recorded several times a day, which could pave “the way for implanted devices to one day predict pain signals or even short-circuit them,” The New York Times reported.
The study says the pain “was associated with electrical fluctuations in the orbitofrontal cortex, an area involved in emotion regulation, self-evaluation, and decision-making,” The Times reported. “The research suggests that such patterns of brain activity could serve as biomarkers to guide diagnosis and treatment for millions of people with shooting or burning chronic pain linked to a damaged nervous system.”
Ajay Wasan, MD, and a pain specialist at the University of Pittsburgh who was not involved in the study praised it to the Times.
“The study really advances a whole generation of research that has shown that the functioning of the brain is really important to processing and perceiving pain,” he said.
Chronic pain is defined as persistent or recurring and lasting more than three months. The Centers for Disease Control and Prevention says about 20% of Americans experience it. It has been linked with depression, Alzheimer’s disease and other dementias, suicide, and substance use.
Yet, the study’s authors noted, “pain severity is often measured through subjective report, while objective biomarkers that may guide diagnosis and treatment are lacking.”
Medtronic provided devices for the study. The study authors reported no conflicts of interest.
A version of this article first appeared on WebMD.com.
a new study in Nature Neuroscience concluded.
The researchers used the devices on four patients who had felt endless nerve pain for more than a year. The devices recorded several times a day, which could pave “the way for implanted devices to one day predict pain signals or even short-circuit them,” The New York Times reported.
The study says the pain “was associated with electrical fluctuations in the orbitofrontal cortex, an area involved in emotion regulation, self-evaluation, and decision-making,” The Times reported. “The research suggests that such patterns of brain activity could serve as biomarkers to guide diagnosis and treatment for millions of people with shooting or burning chronic pain linked to a damaged nervous system.”
Ajay Wasan, MD, and a pain specialist at the University of Pittsburgh who was not involved in the study praised it to the Times.
“The study really advances a whole generation of research that has shown that the functioning of the brain is really important to processing and perceiving pain,” he said.
Chronic pain is defined as persistent or recurring and lasting more than three months. The Centers for Disease Control and Prevention says about 20% of Americans experience it. It has been linked with depression, Alzheimer’s disease and other dementias, suicide, and substance use.
Yet, the study’s authors noted, “pain severity is often measured through subjective report, while objective biomarkers that may guide diagnosis and treatment are lacking.”
Medtronic provided devices for the study. The study authors reported no conflicts of interest.
A version of this article first appeared on WebMD.com.
a new study in Nature Neuroscience concluded.
The researchers used the devices on four patients who had felt endless nerve pain for more than a year. The devices recorded several times a day, which could pave “the way for implanted devices to one day predict pain signals or even short-circuit them,” The New York Times reported.
The study says the pain “was associated with electrical fluctuations in the orbitofrontal cortex, an area involved in emotion regulation, self-evaluation, and decision-making,” The Times reported. “The research suggests that such patterns of brain activity could serve as biomarkers to guide diagnosis and treatment for millions of people with shooting or burning chronic pain linked to a damaged nervous system.”
Ajay Wasan, MD, and a pain specialist at the University of Pittsburgh who was not involved in the study praised it to the Times.
“The study really advances a whole generation of research that has shown that the functioning of the brain is really important to processing and perceiving pain,” he said.
Chronic pain is defined as persistent or recurring and lasting more than three months. The Centers for Disease Control and Prevention says about 20% of Americans experience it. It has been linked with depression, Alzheimer’s disease and other dementias, suicide, and substance use.
Yet, the study’s authors noted, “pain severity is often measured through subjective report, while objective biomarkers that may guide diagnosis and treatment are lacking.”
Medtronic provided devices for the study. The study authors reported no conflicts of interest.
A version of this article first appeared on WebMD.com.
FROM NATURE NEUROSCIENCE
ARNI bests ARB to reduce NT-proBNP in stabilized preserved-EF HF
Patients with an ejection fraction (EF) greater than 40% who were stabilized after recent worsening or de novo heart failure (HF) had a greater reduction in natriuretic peptides and less worsening renal function, but a higher rate of hypotension over 8 weeks with sacubitril-valsartan (Entresto) versus valsartan (Diovan) in the PARAGLIDE-HF trial.
A subgroup analysis showed evidence of a larger treatment effect among those with an EF of 60% or less, said Robert Mentz, MD, of the Duke Clinical Research Institute, Durham, N.C.
Dr. Mentz presented the findings at the Heart Failure Association of the European Society of Cardiology (HFA-ESC) scientific sessions. The study was also published online simultaneously in the Journal of the American College of Cardiology.
“Next steps will involve further assessment of the cardiovascular and renal benefits, as well as further exploration of the symptomatic hypotension that we observed,” Dr. Mentz said in an interview.
Meanwhile, he said, “clinicians should be aware of these new data – specifically, the incremental reduction in natriuretic peptide level, compared with valsartan, and potential benefits on cardiovascular and renal events,” particularly in those with an EF greater than 40% to 60% or less.
Larger benefit for EF > 40% to < 60%
PARAGLIDE-HF was a double-blind, randomized controlled trial with 466 patients with EF greater than 40% enrolled within 30 days of a worsening HF event. The median age was 71 years, 52% were women, and 22% were Black.
The trial was a follow-up to PARAGON-HF, which had shown that, in patients with an EF of at least 45%, sacubitril-valsartan did not result in a significantly lower rate of total hospitalizations for HF or death from cardiovascular causes, compared with valsartan.
The primary endpoint for PARAGLIDE was the time-averaged proportional change in N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) from baseline through weeks 4 and 8, as in the PIONEER-HF trial. That trial showed that among patients hospitalized for acute decompensated HF with reduced EF (< 40%), the angiotensin receptor/neprilysin inhibitor (ARNI) led to a greater reduction in NT-proBNP concentration than the angiotensin receptor blocker (ARB).
Similarly, for PARAGLIDE, the time-averaged reduction in NT-proBNP was greater with sacubitril-valsartan, with a change ratio of 0.85 (15% greater reduction).
A secondary hierarchical outcome for PARAGLIDE, using the win ratio, consisted of time to cardiovascular death, number and timing of HF hospitalizations, number and timing of urgent HF visits, and time-averaged proportional change in NT-proBNP from baseline to weeks 4 and 8.
The hierarchical outcome favored sacubitril-valsartan, but was not significant (unmatched win ratio, 1.19).
As noted, sacubitril-valsartan reduced worsening renal function, compared with valsartan (odds ratio, 0.61), but increased symptomatic hypotension (OR, 1.73).
“We will work to better characterize the hypotension events that were observed to help identify those patients at greater risk and to provide further clarity around the timing and implications of these events,” Dr. Mentz said in an interview.
The team hypothesized that such events may be prevented by optimizing volume status and background therapies commonly used to treat hypertension in these patients.
“For instance,” Dr. Mentz suggested, “calcium channel blockers like amlodipine could be dose reduced or discontinued in patients with lower baseline blood pressures to better support sacubitril/valsartan initiation and titration.”
He highlighted the subgroup analysis showing evidence of a larger treatment effect in study patients with an EF of 60% or less for the NT-proBNP change (0.78) and the hierarchical outcome (win ratio, 1.46).
“These data may influence future guidance for sacubitril-valsartan in HF with EF greater than 40%, regardless of HF chronicity [acute or chronic vs. de novo] and treatment setting [hospital vs. clinic],” Dr. Mentz concluded.
Data ‘far from conclusive’
In a comment, Sean Pinney, MD, chief of cardiology at Mount Sinai Morningside, New York, said that the study results “help expand the current evidence base supporting the use of an ARNI in patients” with an EF greater than 40% up to 60%, and “provide confidence that ARNIs help to lower natriuretic peptides.
“It comes as little surprise that not everyone was able to tolerate these medications due to intolerable side effects like dizziness or hypotension,” he said.
Nevertheless, he added, “hopefully, these trial data help strengthen clinicians’ resolve to prescribe sacubitril/valsartan to a growing population of vulnerable patients.”
In a related editorial, Hector O. Ventura, MD, of the Ochsner Clinical School–University of Queensland, New Orleans, and colleagues express several concerns about the study.
Although the trial achieved significance for the primary endpoint, the margin of benefit was less than expected and the magnitude of the NT-proBNP reduction may not have been enough to reach the threshold for clinical benefit, they wrote.
Diuretic dosing in the two groups was not reported, and between-group differences may have contributed to both the differential NT-proBNP reduction and the rates of hypotension.
Furthermore, the sacubitril-valsartan group had a higher proportion of missing NT-proBNP data, which may have biased the results.
“Clinicians who elect to use sacubitril-valsartan in this population should be mindful of the risk for hypotension and select patients carefully, while providing close ambulatory follow up to ensure stability and adherence,” they noted.
“This important trial provides some wins that support selective use of sacubitril-valsartan in HFpEF [as well as] observed losses, which too may help to define better implementation strategies in appropriately selected patients,” the editorialists concluded.
The study was funded by Novartis. Dr. Mentz and other coauthors have received fees from Novartis. Dr. Pinney, Dr. Ventura, and the other editorialists disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with an ejection fraction (EF) greater than 40% who were stabilized after recent worsening or de novo heart failure (HF) had a greater reduction in natriuretic peptides and less worsening renal function, but a higher rate of hypotension over 8 weeks with sacubitril-valsartan (Entresto) versus valsartan (Diovan) in the PARAGLIDE-HF trial.
A subgroup analysis showed evidence of a larger treatment effect among those with an EF of 60% or less, said Robert Mentz, MD, of the Duke Clinical Research Institute, Durham, N.C.
Dr. Mentz presented the findings at the Heart Failure Association of the European Society of Cardiology (HFA-ESC) scientific sessions. The study was also published online simultaneously in the Journal of the American College of Cardiology.
“Next steps will involve further assessment of the cardiovascular and renal benefits, as well as further exploration of the symptomatic hypotension that we observed,” Dr. Mentz said in an interview.
Meanwhile, he said, “clinicians should be aware of these new data – specifically, the incremental reduction in natriuretic peptide level, compared with valsartan, and potential benefits on cardiovascular and renal events,” particularly in those with an EF greater than 40% to 60% or less.
Larger benefit for EF > 40% to < 60%
PARAGLIDE-HF was a double-blind, randomized controlled trial with 466 patients with EF greater than 40% enrolled within 30 days of a worsening HF event. The median age was 71 years, 52% were women, and 22% were Black.
The trial was a follow-up to PARAGON-HF, which had shown that, in patients with an EF of at least 45%, sacubitril-valsartan did not result in a significantly lower rate of total hospitalizations for HF or death from cardiovascular causes, compared with valsartan.
The primary endpoint for PARAGLIDE was the time-averaged proportional change in N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) from baseline through weeks 4 and 8, as in the PIONEER-HF trial. That trial showed that among patients hospitalized for acute decompensated HF with reduced EF (< 40%), the angiotensin receptor/neprilysin inhibitor (ARNI) led to a greater reduction in NT-proBNP concentration than the angiotensin receptor blocker (ARB).
Similarly, for PARAGLIDE, the time-averaged reduction in NT-proBNP was greater with sacubitril-valsartan, with a change ratio of 0.85 (15% greater reduction).
A secondary hierarchical outcome for PARAGLIDE, using the win ratio, consisted of time to cardiovascular death, number and timing of HF hospitalizations, number and timing of urgent HF visits, and time-averaged proportional change in NT-proBNP from baseline to weeks 4 and 8.
The hierarchical outcome favored sacubitril-valsartan, but was not significant (unmatched win ratio, 1.19).
As noted, sacubitril-valsartan reduced worsening renal function, compared with valsartan (odds ratio, 0.61), but increased symptomatic hypotension (OR, 1.73).
“We will work to better characterize the hypotension events that were observed to help identify those patients at greater risk and to provide further clarity around the timing and implications of these events,” Dr. Mentz said in an interview.
The team hypothesized that such events may be prevented by optimizing volume status and background therapies commonly used to treat hypertension in these patients.
“For instance,” Dr. Mentz suggested, “calcium channel blockers like amlodipine could be dose reduced or discontinued in patients with lower baseline blood pressures to better support sacubitril/valsartan initiation and titration.”
He highlighted the subgroup analysis showing evidence of a larger treatment effect in study patients with an EF of 60% or less for the NT-proBNP change (0.78) and the hierarchical outcome (win ratio, 1.46).
“These data may influence future guidance for sacubitril-valsartan in HF with EF greater than 40%, regardless of HF chronicity [acute or chronic vs. de novo] and treatment setting [hospital vs. clinic],” Dr. Mentz concluded.
Data ‘far from conclusive’
In a comment, Sean Pinney, MD, chief of cardiology at Mount Sinai Morningside, New York, said that the study results “help expand the current evidence base supporting the use of an ARNI in patients” with an EF greater than 40% up to 60%, and “provide confidence that ARNIs help to lower natriuretic peptides.
“It comes as little surprise that not everyone was able to tolerate these medications due to intolerable side effects like dizziness or hypotension,” he said.
Nevertheless, he added, “hopefully, these trial data help strengthen clinicians’ resolve to prescribe sacubitril/valsartan to a growing population of vulnerable patients.”
In a related editorial, Hector O. Ventura, MD, of the Ochsner Clinical School–University of Queensland, New Orleans, and colleagues express several concerns about the study.
Although the trial achieved significance for the primary endpoint, the margin of benefit was less than expected and the magnitude of the NT-proBNP reduction may not have been enough to reach the threshold for clinical benefit, they wrote.
Diuretic dosing in the two groups was not reported, and between-group differences may have contributed to both the differential NT-proBNP reduction and the rates of hypotension.
Furthermore, the sacubitril-valsartan group had a higher proportion of missing NT-proBNP data, which may have biased the results.
“Clinicians who elect to use sacubitril-valsartan in this population should be mindful of the risk for hypotension and select patients carefully, while providing close ambulatory follow up to ensure stability and adherence,” they noted.
“This important trial provides some wins that support selective use of sacubitril-valsartan in HFpEF [as well as] observed losses, which too may help to define better implementation strategies in appropriately selected patients,” the editorialists concluded.
The study was funded by Novartis. Dr. Mentz and other coauthors have received fees from Novartis. Dr. Pinney, Dr. Ventura, and the other editorialists disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with an ejection fraction (EF) greater than 40% who were stabilized after recent worsening or de novo heart failure (HF) had a greater reduction in natriuretic peptides and less worsening renal function, but a higher rate of hypotension over 8 weeks with sacubitril-valsartan (Entresto) versus valsartan (Diovan) in the PARAGLIDE-HF trial.
A subgroup analysis showed evidence of a larger treatment effect among those with an EF of 60% or less, said Robert Mentz, MD, of the Duke Clinical Research Institute, Durham, N.C.
Dr. Mentz presented the findings at the Heart Failure Association of the European Society of Cardiology (HFA-ESC) scientific sessions. The study was also published online simultaneously in the Journal of the American College of Cardiology.
“Next steps will involve further assessment of the cardiovascular and renal benefits, as well as further exploration of the symptomatic hypotension that we observed,” Dr. Mentz said in an interview.
Meanwhile, he said, “clinicians should be aware of these new data – specifically, the incremental reduction in natriuretic peptide level, compared with valsartan, and potential benefits on cardiovascular and renal events,” particularly in those with an EF greater than 40% to 60% or less.
Larger benefit for EF > 40% to < 60%
PARAGLIDE-HF was a double-blind, randomized controlled trial with 466 patients with EF greater than 40% enrolled within 30 days of a worsening HF event. The median age was 71 years, 52% were women, and 22% were Black.
The trial was a follow-up to PARAGON-HF, which had shown that, in patients with an EF of at least 45%, sacubitril-valsartan did not result in a significantly lower rate of total hospitalizations for HF or death from cardiovascular causes, compared with valsartan.
The primary endpoint for PARAGLIDE was the time-averaged proportional change in N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) from baseline through weeks 4 and 8, as in the PIONEER-HF trial. That trial showed that among patients hospitalized for acute decompensated HF with reduced EF (< 40%), the angiotensin receptor/neprilysin inhibitor (ARNI) led to a greater reduction in NT-proBNP concentration than the angiotensin receptor blocker (ARB).
Similarly, for PARAGLIDE, the time-averaged reduction in NT-proBNP was greater with sacubitril-valsartan, with a change ratio of 0.85 (15% greater reduction).
A secondary hierarchical outcome for PARAGLIDE, using the win ratio, consisted of time to cardiovascular death, number and timing of HF hospitalizations, number and timing of urgent HF visits, and time-averaged proportional change in NT-proBNP from baseline to weeks 4 and 8.
The hierarchical outcome favored sacubitril-valsartan, but was not significant (unmatched win ratio, 1.19).
As noted, sacubitril-valsartan reduced worsening renal function, compared with valsartan (odds ratio, 0.61), but increased symptomatic hypotension (OR, 1.73).
“We will work to better characterize the hypotension events that were observed to help identify those patients at greater risk and to provide further clarity around the timing and implications of these events,” Dr. Mentz said in an interview.
The team hypothesized that such events may be prevented by optimizing volume status and background therapies commonly used to treat hypertension in these patients.
“For instance,” Dr. Mentz suggested, “calcium channel blockers like amlodipine could be dose reduced or discontinued in patients with lower baseline blood pressures to better support sacubitril/valsartan initiation and titration.”
He highlighted the subgroup analysis showing evidence of a larger treatment effect in study patients with an EF of 60% or less for the NT-proBNP change (0.78) and the hierarchical outcome (win ratio, 1.46).
“These data may influence future guidance for sacubitril-valsartan in HF with EF greater than 40%, regardless of HF chronicity [acute or chronic vs. de novo] and treatment setting [hospital vs. clinic],” Dr. Mentz concluded.
Data ‘far from conclusive’
In a comment, Sean Pinney, MD, chief of cardiology at Mount Sinai Morningside, New York, said that the study results “help expand the current evidence base supporting the use of an ARNI in patients” with an EF greater than 40% up to 60%, and “provide confidence that ARNIs help to lower natriuretic peptides.
“It comes as little surprise that not everyone was able to tolerate these medications due to intolerable side effects like dizziness or hypotension,” he said.
Nevertheless, he added, “hopefully, these trial data help strengthen clinicians’ resolve to prescribe sacubitril/valsartan to a growing population of vulnerable patients.”
In a related editorial, Hector O. Ventura, MD, of the Ochsner Clinical School–University of Queensland, New Orleans, and colleagues express several concerns about the study.
Although the trial achieved significance for the primary endpoint, the margin of benefit was less than expected and the magnitude of the NT-proBNP reduction may not have been enough to reach the threshold for clinical benefit, they wrote.
Diuretic dosing in the two groups was not reported, and between-group differences may have contributed to both the differential NT-proBNP reduction and the rates of hypotension.
Furthermore, the sacubitril-valsartan group had a higher proportion of missing NT-proBNP data, which may have biased the results.
“Clinicians who elect to use sacubitril-valsartan in this population should be mindful of the risk for hypotension and select patients carefully, while providing close ambulatory follow up to ensure stability and adherence,” they noted.
“This important trial provides some wins that support selective use of sacubitril-valsartan in HFpEF [as well as] observed losses, which too may help to define better implementation strategies in appropriately selected patients,” the editorialists concluded.
The study was funded by Novartis. Dr. Mentz and other coauthors have received fees from Novartis. Dr. Pinney, Dr. Ventura, and the other editorialists disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ESC HEART FAILURE 2023
Adult tonsillectomies work and they’re cost effective
A new randomized trial offers rare insight into outcomes in adult tonsillectomy, a surgical procedure that’s commonly performed in the United States yet falling out of favor. Tonsillectomies are both clinically effective and cost-effective in adult patients with recurrent acute tonsillitis, a British team reports.
The researchers declined to weigh in on whether the procedure is actually better than nonsurgical management. Still, “here at last, we have a substantial piece of scientific evidence which shows that, compared with nonsurgical management, removal of tonsils has a significant impact on the number of sore throat days and on the cost of managing sore throat disease in adults,” said study lead author Janet A. Wilson, MBChB, MD, an emerita professor of otolaryngology at Newcastle University (England), in an interview.
The study was published in The Lancet.
Tonsillectomies have become much less common over the past several decades as questions have arisen about their value. In the United States, the number of procedures performed each year plunged from a high of 1.4 million in 1959 to an estimated 286,000 tonsillectomies performed in children under 15 and 120,000 in people aged 15 in 2010.
It’s harder for adults to tolerate tonsillectomies than children, Dr. Wilson said. In children, surgeons can easily remove tonsils by scraping them off the throat’s side walls. But, she said, “an adult tonsillectomy is more akin to taking off the skin of an unripe orange, so it’s harder work for the surgeon and more traumatizing for the wall of the adult patient’s pharynx. We can only assume that this greater amount of fibrous tissue reflects the cumulative effect of infections over a period of years.”
While tonsillectomies are still performed hundreds of times a day in adults in the United States, a 2014 Cochrane Library review found there’s “insufficient information “to support them versus nonsurgical care as treatments to reduce sore throats.”
For the new multicenter, open-label, randomized study, researchers randomly assigned patients aged 16 and older with recurrent acute tonsillitis to immediate tonsillectomy or nonsurgical management, which Dr. Wilson said can include cold fluids, honey, analgesics/anti-inflammatories. and anesthetic throat lozenges. The study was conducted between 2015 and 2018.
Ultimately, there were 224 and 204 patients, respectively, in the two groups (average age = 23, [19-30], 78% female, 90% White).
Patients who underwent tonsillectomies versus nonsurgical treatment had fewer sore throats over 2 years (median 23 days [IQR 11-46 days] vs. 30 days [14-65 days]) with an incident rate ratio of 0.53 (95% confidence interval, 0.43-0.65, P < 0.0001) after adjustment for clinic site and baseline severity.
The study also shows that “adults who have severe recurrent throat infections with a frequency of seven episodes within 1 year, five or more for 2 consecutive years, or three or more in 3 consecutive years will suffer fewer days of sore throat in the 2 years following tonsillectomy than if they had kept their tonsils,” Dr. Wilson said.
The study doesn’t examine longer-term consequences. A 2018 study of children linked tonsillectomies to “significantly increased relative risk of later respiratory, allergic, and infectious diseases.”
In the new study, nearly 4 in 10 (39%) of the tonsillectomy patients had adverse events linked to the surgeries, and bleeding (19%) was the most common adverse effect. The researchers also estimated that “tonsillectomy has a high probability of being considered cost-effective.”
“Whichever way the results were analyzed and confounding variables allowed for, the result always seems to be the same: Tonsillectomy applied using current qualifying criteria was a worthwhile procedure,” Dr. Wilson said.
Dr. Wilson noted that tonsillectomy patients will suffer a persistent sore throat after surgery, “about the same as a bad episode of tonsillitis.” And she said patients will need to adjust their diet for a few days and take 1-2 weeks off work.
In an interview, internal medicine physician Noel Deep, MD, of Antigo, Wisc., said antibiotics are a common treatment for tonsillitis in primary care clinics. According to him, the United States doesn’t have guidelines for tonsillectomies in adults. He believes they can be considered if tonsillitis keeps recurring three to five times a year and disrupts quality of life.
Dr. Deep said the new study “reinforces the benefit of tonsillectomies. Several studies from Germany, Sweden, Finland, and the United Kingdom have demonstrated benefits of tonsillectomies, but they were only for short periods of less than a year and lacked long-term data.”
He noted that “there is no clear evidence as to when to recommend tonsillectomies.” Clinicians should talk to patients about the potential that tonsillectomies will reduce sore throat episodes and cost the patient less in the long run, he said. It’s also important, he said, to make sure tonsillitis is bacterial before prescribing antibiotics.
The United Kingdom’s National Institute for Health Research funded the study. Dr. Wilson disclosed support for meetings/travel from ENT Scotland, and the other authors report various disclosures, including grants and contracts. Dr. Deep serves on the editorial advisory board of Internal Medicine News and is chair of the American Medical Association Council on Science and Public Health.
A new randomized trial offers rare insight into outcomes in adult tonsillectomy, a surgical procedure that’s commonly performed in the United States yet falling out of favor. Tonsillectomies are both clinically effective and cost-effective in adult patients with recurrent acute tonsillitis, a British team reports.
The researchers declined to weigh in on whether the procedure is actually better than nonsurgical management. Still, “here at last, we have a substantial piece of scientific evidence which shows that, compared with nonsurgical management, removal of tonsils has a significant impact on the number of sore throat days and on the cost of managing sore throat disease in adults,” said study lead author Janet A. Wilson, MBChB, MD, an emerita professor of otolaryngology at Newcastle University (England), in an interview.
The study was published in The Lancet.
Tonsillectomies have become much less common over the past several decades as questions have arisen about their value. In the United States, the number of procedures performed each year plunged from a high of 1.4 million in 1959 to an estimated 286,000 tonsillectomies performed in children under 15 and 120,000 in people aged 15 in 2010.
It’s harder for adults to tolerate tonsillectomies than children, Dr. Wilson said. In children, surgeons can easily remove tonsils by scraping them off the throat’s side walls. But, she said, “an adult tonsillectomy is more akin to taking off the skin of an unripe orange, so it’s harder work for the surgeon and more traumatizing for the wall of the adult patient’s pharynx. We can only assume that this greater amount of fibrous tissue reflects the cumulative effect of infections over a period of years.”
While tonsillectomies are still performed hundreds of times a day in adults in the United States, a 2014 Cochrane Library review found there’s “insufficient information “to support them versus nonsurgical care as treatments to reduce sore throats.”
For the new multicenter, open-label, randomized study, researchers randomly assigned patients aged 16 and older with recurrent acute tonsillitis to immediate tonsillectomy or nonsurgical management, which Dr. Wilson said can include cold fluids, honey, analgesics/anti-inflammatories. and anesthetic throat lozenges. The study was conducted between 2015 and 2018.
Ultimately, there were 224 and 204 patients, respectively, in the two groups (average age = 23, [19-30], 78% female, 90% White).
Patients who underwent tonsillectomies versus nonsurgical treatment had fewer sore throats over 2 years (median 23 days [IQR 11-46 days] vs. 30 days [14-65 days]) with an incident rate ratio of 0.53 (95% confidence interval, 0.43-0.65, P < 0.0001) after adjustment for clinic site and baseline severity.
The study also shows that “adults who have severe recurrent throat infections with a frequency of seven episodes within 1 year, five or more for 2 consecutive years, or three or more in 3 consecutive years will suffer fewer days of sore throat in the 2 years following tonsillectomy than if they had kept their tonsils,” Dr. Wilson said.
The study doesn’t examine longer-term consequences. A 2018 study of children linked tonsillectomies to “significantly increased relative risk of later respiratory, allergic, and infectious diseases.”
In the new study, nearly 4 in 10 (39%) of the tonsillectomy patients had adverse events linked to the surgeries, and bleeding (19%) was the most common adverse effect. The researchers also estimated that “tonsillectomy has a high probability of being considered cost-effective.”
“Whichever way the results were analyzed and confounding variables allowed for, the result always seems to be the same: Tonsillectomy applied using current qualifying criteria was a worthwhile procedure,” Dr. Wilson said.
Dr. Wilson noted that tonsillectomy patients will suffer a persistent sore throat after surgery, “about the same as a bad episode of tonsillitis.” And she said patients will need to adjust their diet for a few days and take 1-2 weeks off work.
In an interview, internal medicine physician Noel Deep, MD, of Antigo, Wisc., said antibiotics are a common treatment for tonsillitis in primary care clinics. According to him, the United States doesn’t have guidelines for tonsillectomies in adults. He believes they can be considered if tonsillitis keeps recurring three to five times a year and disrupts quality of life.
Dr. Deep said the new study “reinforces the benefit of tonsillectomies. Several studies from Germany, Sweden, Finland, and the United Kingdom have demonstrated benefits of tonsillectomies, but they were only for short periods of less than a year and lacked long-term data.”
He noted that “there is no clear evidence as to when to recommend tonsillectomies.” Clinicians should talk to patients about the potential that tonsillectomies will reduce sore throat episodes and cost the patient less in the long run, he said. It’s also important, he said, to make sure tonsillitis is bacterial before prescribing antibiotics.
The United Kingdom’s National Institute for Health Research funded the study. Dr. Wilson disclosed support for meetings/travel from ENT Scotland, and the other authors report various disclosures, including grants and contracts. Dr. Deep serves on the editorial advisory board of Internal Medicine News and is chair of the American Medical Association Council on Science and Public Health.
A new randomized trial offers rare insight into outcomes in adult tonsillectomy, a surgical procedure that’s commonly performed in the United States yet falling out of favor. Tonsillectomies are both clinically effective and cost-effective in adult patients with recurrent acute tonsillitis, a British team reports.
The researchers declined to weigh in on whether the procedure is actually better than nonsurgical management. Still, “here at last, we have a substantial piece of scientific evidence which shows that, compared with nonsurgical management, removal of tonsils has a significant impact on the number of sore throat days and on the cost of managing sore throat disease in adults,” said study lead author Janet A. Wilson, MBChB, MD, an emerita professor of otolaryngology at Newcastle University (England), in an interview.
The study was published in The Lancet.
Tonsillectomies have become much less common over the past several decades as questions have arisen about their value. In the United States, the number of procedures performed each year plunged from a high of 1.4 million in 1959 to an estimated 286,000 tonsillectomies performed in children under 15 and 120,000 in people aged 15 in 2010.
It’s harder for adults to tolerate tonsillectomies than children, Dr. Wilson said. In children, surgeons can easily remove tonsils by scraping them off the throat’s side walls. But, she said, “an adult tonsillectomy is more akin to taking off the skin of an unripe orange, so it’s harder work for the surgeon and more traumatizing for the wall of the adult patient’s pharynx. We can only assume that this greater amount of fibrous tissue reflects the cumulative effect of infections over a period of years.”
While tonsillectomies are still performed hundreds of times a day in adults in the United States, a 2014 Cochrane Library review found there’s “insufficient information “to support them versus nonsurgical care as treatments to reduce sore throats.”
For the new multicenter, open-label, randomized study, researchers randomly assigned patients aged 16 and older with recurrent acute tonsillitis to immediate tonsillectomy or nonsurgical management, which Dr. Wilson said can include cold fluids, honey, analgesics/anti-inflammatories. and anesthetic throat lozenges. The study was conducted between 2015 and 2018.
Ultimately, there were 224 and 204 patients, respectively, in the two groups (average age = 23, [19-30], 78% female, 90% White).
Patients who underwent tonsillectomies versus nonsurgical treatment had fewer sore throats over 2 years (median 23 days [IQR 11-46 days] vs. 30 days [14-65 days]) with an incident rate ratio of 0.53 (95% confidence interval, 0.43-0.65, P < 0.0001) after adjustment for clinic site and baseline severity.
The study also shows that “adults who have severe recurrent throat infections with a frequency of seven episodes within 1 year, five or more for 2 consecutive years, or three or more in 3 consecutive years will suffer fewer days of sore throat in the 2 years following tonsillectomy than if they had kept their tonsils,” Dr. Wilson said.
The study doesn’t examine longer-term consequences. A 2018 study of children linked tonsillectomies to “significantly increased relative risk of later respiratory, allergic, and infectious diseases.”
In the new study, nearly 4 in 10 (39%) of the tonsillectomy patients had adverse events linked to the surgeries, and bleeding (19%) was the most common adverse effect. The researchers also estimated that “tonsillectomy has a high probability of being considered cost-effective.”
“Whichever way the results were analyzed and confounding variables allowed for, the result always seems to be the same: Tonsillectomy applied using current qualifying criteria was a worthwhile procedure,” Dr. Wilson said.
Dr. Wilson noted that tonsillectomy patients will suffer a persistent sore throat after surgery, “about the same as a bad episode of tonsillitis.” And she said patients will need to adjust their diet for a few days and take 1-2 weeks off work.
In an interview, internal medicine physician Noel Deep, MD, of Antigo, Wisc., said antibiotics are a common treatment for tonsillitis in primary care clinics. According to him, the United States doesn’t have guidelines for tonsillectomies in adults. He believes they can be considered if tonsillitis keeps recurring three to five times a year and disrupts quality of life.
Dr. Deep said the new study “reinforces the benefit of tonsillectomies. Several studies from Germany, Sweden, Finland, and the United Kingdom have demonstrated benefits of tonsillectomies, but they were only for short periods of less than a year and lacked long-term data.”
He noted that “there is no clear evidence as to when to recommend tonsillectomies.” Clinicians should talk to patients about the potential that tonsillectomies will reduce sore throat episodes and cost the patient less in the long run, he said. It’s also important, he said, to make sure tonsillitis is bacterial before prescribing antibiotics.
The United Kingdom’s National Institute for Health Research funded the study. Dr. Wilson disclosed support for meetings/travel from ENT Scotland, and the other authors report various disclosures, including grants and contracts. Dr. Deep serves on the editorial advisory board of Internal Medicine News and is chair of the American Medical Association Council on Science and Public Health.
FROM THE LANCET
The weird world of hydrogels: How they’ll change health care
Imagine a day when a simple injection prompts a broken bone to heal. When tiny, ingestible devices linger in the body, unnoticed, tracking our health or delivering life-saving medications. When brain and heart implants mesh with flesh so seamlessly that the body thinks they’ve been there all along.
These are the dreams of materials scientists who have toiled for decades to mimic the complex architecture of the human body in hopes of replacing broken parts or treating disease.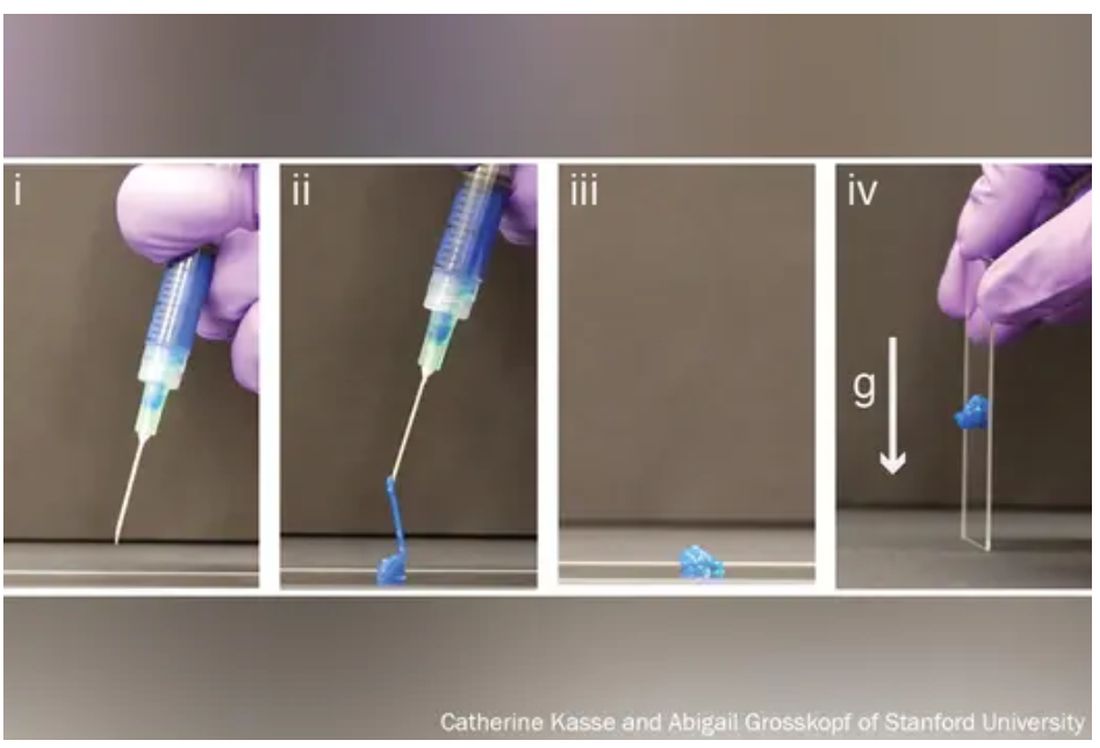
The problem, say bioengineers, is that most replacement and corrective parts – from prosthetics to pacemakers – are made of hard, dry, lifeless materials, like metal or plastic, while biological tissue is soft, wet, and living.
The body knows the difference and tends to reject imitations.
Enter hydrogels, three-dimensional networks of molecules swollen with – by definition – water.
First described in 1960 by creators of soft contact lenses, these weird, shape-shifting substances are able to morph from liquid to solid to a squishy in-between. (Early, simple uses include hair gel or Jell-O.). Slow to gain attention, growing to just 1,000 studies published by 1982, they’ve become the subject of intense study recently, with 100,000 papers published by 2020, and 3,800 already this year alone.
As chemists, biologists, and engineers begin to work more with one another and with medical doctors,
“We are, essentially, hydrogels,” said Benjamin Wiley, PhD, a chemistry professor at Duke University in Durham, N.C. “As people develop new hydrogels that more closely match the tissues in our body, we’ll be able to treat a whole host of ailments we couldn’t treat before.”
From contact lenses to brain implants
Put simply, a hydrogel is like a mesh bag of water.
The mesh is made of polymers, or spaghetti-like strands of molecules, stitched together in a repeating pattern and swollen with H2O, much like the way 3D matrixes in our body surround, support, and give structure to our cells and tissues.
“Imagine a soccer net, with all of these long fibers woven together to create the net,” said Eric Appel, PhD, associate professor of materials science and engineering at Stanford (Calif.) University.
While the broader category of “gels” could be filled with anything, including chemical solvents, water is the key ingredient that sets hydrogels apart, making them ideal for, as some scientists put it, “merging humans and machines.”
Human bones are about 25% water, while muscles hover around 70% and the brain is 85%. The precious liquid plays a host of critical roles, from shuttling nutrients in and waste out to helping cells talk to each other.
Lab-made hydrogels can be loaded with cargo (like a ball in the net), including cells or drugs that help mimic some of those functions.
Hydrogels are soft and pliable like flesh. So, if used in implants, they may be less likely to damage surrounding tissue.
“Think about a metal spoon in your bowl of pudding. As you’re shaking the bowl, the spoon doesn’t stay in place, and you get scarring around the spoon,” said Christina Tringides, PhD, a materials scientist who studies neural engineering. That, she says, is exactly what happens to brain implants when patients breathe or move. “It’s a mechanical mismatch. But with hydrogels, you could get perfect mechanical matching.”
Hydrogels also tend to be nontoxic, so the immune system may be less likely to attack them as foreign bodies.
All this has made hydrogels the new darling of the bioengineering world.
“There has been an absolute explosion of interest in these materials,” Dr. Appel said.
Smarter drug delivery and ingestible electronics
Early versions of hydrogels were thick and gooey, making it hard to get them inside the body.
“Think of a block of Jell-O. You couldn’t inject something like that,” Dr. Appel said.
But Dr. Appel, whose lab develops new drug delivery systems, has been tinkering with gel formulas for years in hopes that these high-tech globs could someday ferry timed-release drugs to just the right spot in the body.
His new hydrogels start as fully formed gels (which help preserve the drug contents) inside a syringe. But once the plunger is pushed, they magically shape-shift to a liquid thin enough to flow easily through a standard needle. Upon exit, they immediately reform into gels, protecting the inherent cargo from degrading.
This could be a game changer at a time when many cutting-edge drugs – think Humira for arthritis or Ozempic for type 2 diabetes – are made of quickly degrading proteins too large and complex to simply jam into a pill. Instead, they must be injected, often frequently.
“Because the gel takes months to dissolve, it slowly delivers the drug over time,” Dr. Appel said. “You could conceivably go from a shot once a week to once every 4 months.”
Such slow-release hydrogels could make vaccines last longer, in turn teaching the body to better resist emerging virus variants, and deliver tumor-busting therapies more precisely, said Dr. Appel, who has formed a startup and hopes to fast-track the first hydrogel drug delivery system to clinical trials within a few years.
Meanwhile, another team at the Massachusetts Institute of Technology has taken a different approach, developing a standard-sized ingestible hydrogel pill that swells up like a puffer fish in the stomach, lasting a month and slowly releasing drugs all the while. To remove the pill, a patient simply drinks a salt-based solution that shrivels the ping-pong ball–sized device so it can be passed out of the body.
In a paper in Nature Communications, the scientists showed the puffer fish pill could also be loaded with tiny cameras or monitors to track conditions like ulcers or cancer.
“The dream is to have a Jell-O-like smart pill that, once swallowed, stays in the stomach and monitors the patient’s health,” said Xuanhe Zhao, PhD, a researcher on the project and associate professor of mechanical engineering at MIT.
Building joints and regrowing bones
Since the 1970s, researchers have mulled using hydrogels to replace human cartilage, a remarkably strong and flexible tissue made of about 90% water but able to withstand the weight of a car on an area about the size of a coin.
Until recently, those efforts have largely failed. Meaning when knee cartilage wears down, things like cartilage transplants, drilling holes to stimulate new growth, or total joint replacements – all of which require lengthy rehab – are the only options.
But that may be about to change.
Dr. Wiley and his colleagues at Duke recently reported that they’d developed the first gel-based cartilage substitute even stronger and more durable than the real thing.
By attaching their hydrogel to a titanium backing to help stick it in place, they hope to repair damaged cartilage “much like a dentist fills a cavity” long before surgery is necessary.
They too have partnered with industry to bring their hydrogel to market – starting with knees.
“Ultimately, the goal is to do any joint – hips, ankles, fingers, and toes,” Dr. Wiley said.
At the University of Toronto, chemist Karina Carneiro, PhD, and dentist Christopher McCulloch, DDS, are also thinking big.
In a recent paper in Proceedings of the National Academy of Sciences, they describe a hydrogel, designed by Dr. Carneiro and made of DNA, that can be injected, migrate to a defect in bone – an irreparable break, hole from surgery, or jawbone withered by age – and fill in the gap like putty. But not only does it patch the hole, it prompts the bone to regenerate.
In rats with holes in their skulls due to surgery, they found that the treatment did not work as well as the existing gold standard for repairing holes in bone – grafting bone from elsewhere in the body. But it did work.
“These are very early days for DNA hydrogels,” cautioned Dr. McCulloch, a study coauthor and professor in the Faculty of Dentistry, noting that it will likely be a decade or more before such technology could be available to patients. “But there is the potential that DNA hydrogel could someday grow bone without having to have highly invasive surgical procedures. That’s a significant advancement.”
A sci-fi future
Perhaps the wildest, and weirdest, potential applications of hydrogels come in the realm of human-machine interaction.
Numerous companies are already dabbling in neural prosthetic or brain computer interfaces that might someday, for instance, let someone who is paralyzed and can’t speak write on a laptop using their thoughts.
The spoon-in-the-Jell-O problem has been a major stumbling block.
But Dr. Tringides, who recently earned her PhD in biophysics from Harvard, is working on it.
She and her team have developed a seaweed-based hydrogel loaded with tiny flecks of nanomaterials that can not only meld nicely into squishy brain tissue but also conduct electricity.
Within a decade, she says, this could replace the clunky platinum metal discs used for electrocorticography – recording electrical activity in the brain to identify where seizures start or doing precise brain surgery.
In 30 to 50 years? Let your imagination run wild.
“I’m a skeptic. I like to take research step by step,” she said. “But things are definitely progressing in an interesting direction.”
A version of this article first appeared on WebMD.com.
Imagine a day when a simple injection prompts a broken bone to heal. When tiny, ingestible devices linger in the body, unnoticed, tracking our health or delivering life-saving medications. When brain and heart implants mesh with flesh so seamlessly that the body thinks they’ve been there all along.
These are the dreams of materials scientists who have toiled for decades to mimic the complex architecture of the human body in hopes of replacing broken parts or treating disease.
The problem, say bioengineers, is that most replacement and corrective parts – from prosthetics to pacemakers – are made of hard, dry, lifeless materials, like metal or plastic, while biological tissue is soft, wet, and living.
The body knows the difference and tends to reject imitations.
Enter hydrogels, three-dimensional networks of molecules swollen with – by definition – water.
First described in 1960 by creators of soft contact lenses, these weird, shape-shifting substances are able to morph from liquid to solid to a squishy in-between. (Early, simple uses include hair gel or Jell-O.). Slow to gain attention, growing to just 1,000 studies published by 1982, they’ve become the subject of intense study recently, with 100,000 papers published by 2020, and 3,800 already this year alone.
As chemists, biologists, and engineers begin to work more with one another and with medical doctors,
“We are, essentially, hydrogels,” said Benjamin Wiley, PhD, a chemistry professor at Duke University in Durham, N.C. “As people develop new hydrogels that more closely match the tissues in our body, we’ll be able to treat a whole host of ailments we couldn’t treat before.”
From contact lenses to brain implants
Put simply, a hydrogel is like a mesh bag of water.
The mesh is made of polymers, or spaghetti-like strands of molecules, stitched together in a repeating pattern and swollen with H2O, much like the way 3D matrixes in our body surround, support, and give structure to our cells and tissues.
“Imagine a soccer net, with all of these long fibers woven together to create the net,” said Eric Appel, PhD, associate professor of materials science and engineering at Stanford (Calif.) University.
While the broader category of “gels” could be filled with anything, including chemical solvents, water is the key ingredient that sets hydrogels apart, making them ideal for, as some scientists put it, “merging humans and machines.”
Human bones are about 25% water, while muscles hover around 70% and the brain is 85%. The precious liquid plays a host of critical roles, from shuttling nutrients in and waste out to helping cells talk to each other.
Lab-made hydrogels can be loaded with cargo (like a ball in the net), including cells or drugs that help mimic some of those functions.
Hydrogels are soft and pliable like flesh. So, if used in implants, they may be less likely to damage surrounding tissue.
“Think about a metal spoon in your bowl of pudding. As you’re shaking the bowl, the spoon doesn’t stay in place, and you get scarring around the spoon,” said Christina Tringides, PhD, a materials scientist who studies neural engineering. That, she says, is exactly what happens to brain implants when patients breathe or move. “It’s a mechanical mismatch. But with hydrogels, you could get perfect mechanical matching.”
Hydrogels also tend to be nontoxic, so the immune system may be less likely to attack them as foreign bodies.
All this has made hydrogels the new darling of the bioengineering world.
“There has been an absolute explosion of interest in these materials,” Dr. Appel said.
Smarter drug delivery and ingestible electronics
Early versions of hydrogels were thick and gooey, making it hard to get them inside the body.
“Think of a block of Jell-O. You couldn’t inject something like that,” Dr. Appel said.
But Dr. Appel, whose lab develops new drug delivery systems, has been tinkering with gel formulas for years in hopes that these high-tech globs could someday ferry timed-release drugs to just the right spot in the body.
His new hydrogels start as fully formed gels (which help preserve the drug contents) inside a syringe. But once the plunger is pushed, they magically shape-shift to a liquid thin enough to flow easily through a standard needle. Upon exit, they immediately reform into gels, protecting the inherent cargo from degrading.
This could be a game changer at a time when many cutting-edge drugs – think Humira for arthritis or Ozempic for type 2 diabetes – are made of quickly degrading proteins too large and complex to simply jam into a pill. Instead, they must be injected, often frequently.
“Because the gel takes months to dissolve, it slowly delivers the drug over time,” Dr. Appel said. “You could conceivably go from a shot once a week to once every 4 months.”
Such slow-release hydrogels could make vaccines last longer, in turn teaching the body to better resist emerging virus variants, and deliver tumor-busting therapies more precisely, said Dr. Appel, who has formed a startup and hopes to fast-track the first hydrogel drug delivery system to clinical trials within a few years.
Meanwhile, another team at the Massachusetts Institute of Technology has taken a different approach, developing a standard-sized ingestible hydrogel pill that swells up like a puffer fish in the stomach, lasting a month and slowly releasing drugs all the while. To remove the pill, a patient simply drinks a salt-based solution that shrivels the ping-pong ball–sized device so it can be passed out of the body.
In a paper in Nature Communications, the scientists showed the puffer fish pill could also be loaded with tiny cameras or monitors to track conditions like ulcers or cancer.
“The dream is to have a Jell-O-like smart pill that, once swallowed, stays in the stomach and monitors the patient’s health,” said Xuanhe Zhao, PhD, a researcher on the project and associate professor of mechanical engineering at MIT.
Building joints and regrowing bones
Since the 1970s, researchers have mulled using hydrogels to replace human cartilage, a remarkably strong and flexible tissue made of about 90% water but able to withstand the weight of a car on an area about the size of a coin.
Until recently, those efforts have largely failed. Meaning when knee cartilage wears down, things like cartilage transplants, drilling holes to stimulate new growth, or total joint replacements – all of which require lengthy rehab – are the only options.
But that may be about to change.
Dr. Wiley and his colleagues at Duke recently reported that they’d developed the first gel-based cartilage substitute even stronger and more durable than the real thing.
By attaching their hydrogel to a titanium backing to help stick it in place, they hope to repair damaged cartilage “much like a dentist fills a cavity” long before surgery is necessary.
They too have partnered with industry to bring their hydrogel to market – starting with knees.
“Ultimately, the goal is to do any joint – hips, ankles, fingers, and toes,” Dr. Wiley said.
At the University of Toronto, chemist Karina Carneiro, PhD, and dentist Christopher McCulloch, DDS, are also thinking big.
In a recent paper in Proceedings of the National Academy of Sciences, they describe a hydrogel, designed by Dr. Carneiro and made of DNA, that can be injected, migrate to a defect in bone – an irreparable break, hole from surgery, or jawbone withered by age – and fill in the gap like putty. But not only does it patch the hole, it prompts the bone to regenerate.
In rats with holes in their skulls due to surgery, they found that the treatment did not work as well as the existing gold standard for repairing holes in bone – grafting bone from elsewhere in the body. But it did work.
“These are very early days for DNA hydrogels,” cautioned Dr. McCulloch, a study coauthor and professor in the Faculty of Dentistry, noting that it will likely be a decade or more before such technology could be available to patients. “But there is the potential that DNA hydrogel could someday grow bone without having to have highly invasive surgical procedures. That’s a significant advancement.”
A sci-fi future
Perhaps the wildest, and weirdest, potential applications of hydrogels come in the realm of human-machine interaction.
Numerous companies are already dabbling in neural prosthetic or brain computer interfaces that might someday, for instance, let someone who is paralyzed and can’t speak write on a laptop using their thoughts.
The spoon-in-the-Jell-O problem has been a major stumbling block.
But Dr. Tringides, who recently earned her PhD in biophysics from Harvard, is working on it.
She and her team have developed a seaweed-based hydrogel loaded with tiny flecks of nanomaterials that can not only meld nicely into squishy brain tissue but also conduct electricity.
Within a decade, she says, this could replace the clunky platinum metal discs used for electrocorticography – recording electrical activity in the brain to identify where seizures start or doing precise brain surgery.
In 30 to 50 years? Let your imagination run wild.
“I’m a skeptic. I like to take research step by step,” she said. “But things are definitely progressing in an interesting direction.”
A version of this article first appeared on WebMD.com.
Imagine a day when a simple injection prompts a broken bone to heal. When tiny, ingestible devices linger in the body, unnoticed, tracking our health or delivering life-saving medications. When brain and heart implants mesh with flesh so seamlessly that the body thinks they’ve been there all along.
These are the dreams of materials scientists who have toiled for decades to mimic the complex architecture of the human body in hopes of replacing broken parts or treating disease.
The problem, say bioengineers, is that most replacement and corrective parts – from prosthetics to pacemakers – are made of hard, dry, lifeless materials, like metal or plastic, while biological tissue is soft, wet, and living.
The body knows the difference and tends to reject imitations.
Enter hydrogels, three-dimensional networks of molecules swollen with – by definition – water.
First described in 1960 by creators of soft contact lenses, these weird, shape-shifting substances are able to morph from liquid to solid to a squishy in-between. (Early, simple uses include hair gel or Jell-O.). Slow to gain attention, growing to just 1,000 studies published by 1982, they’ve become the subject of intense study recently, with 100,000 papers published by 2020, and 3,800 already this year alone.
As chemists, biologists, and engineers begin to work more with one another and with medical doctors,
“We are, essentially, hydrogels,” said Benjamin Wiley, PhD, a chemistry professor at Duke University in Durham, N.C. “As people develop new hydrogels that more closely match the tissues in our body, we’ll be able to treat a whole host of ailments we couldn’t treat before.”
From contact lenses to brain implants
Put simply, a hydrogel is like a mesh bag of water.
The mesh is made of polymers, or spaghetti-like strands of molecules, stitched together in a repeating pattern and swollen with H2O, much like the way 3D matrixes in our body surround, support, and give structure to our cells and tissues.
“Imagine a soccer net, with all of these long fibers woven together to create the net,” said Eric Appel, PhD, associate professor of materials science and engineering at Stanford (Calif.) University.
While the broader category of “gels” could be filled with anything, including chemical solvents, water is the key ingredient that sets hydrogels apart, making them ideal for, as some scientists put it, “merging humans and machines.”
Human bones are about 25% water, while muscles hover around 70% and the brain is 85%. The precious liquid plays a host of critical roles, from shuttling nutrients in and waste out to helping cells talk to each other.
Lab-made hydrogels can be loaded with cargo (like a ball in the net), including cells or drugs that help mimic some of those functions.
Hydrogels are soft and pliable like flesh. So, if used in implants, they may be less likely to damage surrounding tissue.
“Think about a metal spoon in your bowl of pudding. As you’re shaking the bowl, the spoon doesn’t stay in place, and you get scarring around the spoon,” said Christina Tringides, PhD, a materials scientist who studies neural engineering. That, she says, is exactly what happens to brain implants when patients breathe or move. “It’s a mechanical mismatch. But with hydrogels, you could get perfect mechanical matching.”
Hydrogels also tend to be nontoxic, so the immune system may be less likely to attack them as foreign bodies.
All this has made hydrogels the new darling of the bioengineering world.
“There has been an absolute explosion of interest in these materials,” Dr. Appel said.
Smarter drug delivery and ingestible electronics
Early versions of hydrogels were thick and gooey, making it hard to get them inside the body.
“Think of a block of Jell-O. You couldn’t inject something like that,” Dr. Appel said.
But Dr. Appel, whose lab develops new drug delivery systems, has been tinkering with gel formulas for years in hopes that these high-tech globs could someday ferry timed-release drugs to just the right spot in the body.
His new hydrogels start as fully formed gels (which help preserve the drug contents) inside a syringe. But once the plunger is pushed, they magically shape-shift to a liquid thin enough to flow easily through a standard needle. Upon exit, they immediately reform into gels, protecting the inherent cargo from degrading.
This could be a game changer at a time when many cutting-edge drugs – think Humira for arthritis or Ozempic for type 2 diabetes – are made of quickly degrading proteins too large and complex to simply jam into a pill. Instead, they must be injected, often frequently.
“Because the gel takes months to dissolve, it slowly delivers the drug over time,” Dr. Appel said. “You could conceivably go from a shot once a week to once every 4 months.”
Such slow-release hydrogels could make vaccines last longer, in turn teaching the body to better resist emerging virus variants, and deliver tumor-busting therapies more precisely, said Dr. Appel, who has formed a startup and hopes to fast-track the first hydrogel drug delivery system to clinical trials within a few years.
Meanwhile, another team at the Massachusetts Institute of Technology has taken a different approach, developing a standard-sized ingestible hydrogel pill that swells up like a puffer fish in the stomach, lasting a month and slowly releasing drugs all the while. To remove the pill, a patient simply drinks a salt-based solution that shrivels the ping-pong ball–sized device so it can be passed out of the body.
In a paper in Nature Communications, the scientists showed the puffer fish pill could also be loaded with tiny cameras or monitors to track conditions like ulcers or cancer.
“The dream is to have a Jell-O-like smart pill that, once swallowed, stays in the stomach and monitors the patient’s health,” said Xuanhe Zhao, PhD, a researcher on the project and associate professor of mechanical engineering at MIT.
Building joints and regrowing bones
Since the 1970s, researchers have mulled using hydrogels to replace human cartilage, a remarkably strong and flexible tissue made of about 90% water but able to withstand the weight of a car on an area about the size of a coin.
Until recently, those efforts have largely failed. Meaning when knee cartilage wears down, things like cartilage transplants, drilling holes to stimulate new growth, or total joint replacements – all of which require lengthy rehab – are the only options.
But that may be about to change.
Dr. Wiley and his colleagues at Duke recently reported that they’d developed the first gel-based cartilage substitute even stronger and more durable than the real thing.
By attaching their hydrogel to a titanium backing to help stick it in place, they hope to repair damaged cartilage “much like a dentist fills a cavity” long before surgery is necessary.
They too have partnered with industry to bring their hydrogel to market – starting with knees.
“Ultimately, the goal is to do any joint – hips, ankles, fingers, and toes,” Dr. Wiley said.
At the University of Toronto, chemist Karina Carneiro, PhD, and dentist Christopher McCulloch, DDS, are also thinking big.
In a recent paper in Proceedings of the National Academy of Sciences, they describe a hydrogel, designed by Dr. Carneiro and made of DNA, that can be injected, migrate to a defect in bone – an irreparable break, hole from surgery, or jawbone withered by age – and fill in the gap like putty. But not only does it patch the hole, it prompts the bone to regenerate.
In rats with holes in their skulls due to surgery, they found that the treatment did not work as well as the existing gold standard for repairing holes in bone – grafting bone from elsewhere in the body. But it did work.
“These are very early days for DNA hydrogels,” cautioned Dr. McCulloch, a study coauthor and professor in the Faculty of Dentistry, noting that it will likely be a decade or more before such technology could be available to patients. “But there is the potential that DNA hydrogel could someday grow bone without having to have highly invasive surgical procedures. That’s a significant advancement.”
A sci-fi future
Perhaps the wildest, and weirdest, potential applications of hydrogels come in the realm of human-machine interaction.
Numerous companies are already dabbling in neural prosthetic or brain computer interfaces that might someday, for instance, let someone who is paralyzed and can’t speak write on a laptop using their thoughts.
The spoon-in-the-Jell-O problem has been a major stumbling block.
But Dr. Tringides, who recently earned her PhD in biophysics from Harvard, is working on it.
She and her team have developed a seaweed-based hydrogel loaded with tiny flecks of nanomaterials that can not only meld nicely into squishy brain tissue but also conduct electricity.
Within a decade, she says, this could replace the clunky platinum metal discs used for electrocorticography – recording electrical activity in the brain to identify where seizures start or doing precise brain surgery.
In 30 to 50 years? Let your imagination run wild.
“I’m a skeptic. I like to take research step by step,” she said. “But things are definitely progressing in an interesting direction.”
A version of this article first appeared on WebMD.com.
Should you prescribe bioidentical hormones for menopause?
BALTIMORE – according to an expert at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists (ACOG).
Clinicians write an estimated 26 to 33 million prescriptions for compounded bioidentical hormone therapy (cBHT) every year, and almost 41% of menopausal women who need treatment try cBHT during their lives. But these drugs lack the approval for this indication from the Food and Drug Administration.
“There is a public perception that this is natural, safer, and anti-aging,” said Robert Kauffman, MD, a professor of obstetrics and gynecology and assistant dean for research at Texas Tech University Health Sciences Center in Amarillo.
Following the 2002 Women’s Health Initiative report showing a link between hormone therapy (HT) and an increase in the incidence of breast cancer, medical schools have slowed or paused instructing trainees on the traditional treatment, Dr. Kauffman said. The association was later determined to be spurious: HT is not associated with a risk for all-cause mortality or deaths from cardiovascular disease or cancer. However, HT still is largely ignored by younger physicians, Dr. Kauffman said, because of unsubstantiated “dangers” such as heart attack, stroke, and deep vein thrombosis.
The lack of education on HT for medical school students and residents has “opened the door to unsubstantiated marketing claims and practices” for cBHT, Dr. Kauffman said. “Hence, the use of compounded bioidentical hormone therapy has increased” as clinicians look for alternatives.
Groups including ACOG, the North American Menopause Society (NAMS), and the U.S. Preventive Services Task Force recommend against the use of Non–FDA-approved therapies such as cBHT, except for narrow indications. Dr. Kauffman said that drug manufacturers have not conducted randomized controlled trials or observational studies on cBHT in treating menopause.
He cited studies showing quality problems with the compounding process of these drugs, and wide variations in the amount of actual ingredients from product labels. One 2021 study published in Menopause comparing patients taking cBHT or FDA-approved HT found that side effects were significantly higher in the cBHT group (57.6% vs. 14.8%; P < .0001).
But manufacturers of cBHT claim that their products prevent cardiovascular disease and Alzheimer’s disease and decrease the risk for breast cancer and stroke – assertions that are at best unproven, according to Dr. Kauffman.
The National Academies of Sciences, Engineering, and Medicine in 2020 said that clinicians have a duty to inform patients of the insufficient evidence to support clinical use of cBHT and should prescribe the products only to patients with documented allergies to an active ingredient in an FDA-approved agent or who require an alternative dosage.
Patients may also have to pay much more out of pocket for cBHT products because they often are not covered by insurance. Generic HT products, meanwhile, are relatively inexpensive and typically are covered, he noted.
“We have to be careful to avoid financial harm to patients by prescribing things, which are much more expensive than those which are usually available,” Dr. Kauffman said.
Prescribing any non–FDA-approved product, especially when biosimilars are available, places physicians at legal risk, Dr. Kauffman said. Physicians who recommend cBHT should inform patients that the products are not FDA approved and carefully document this discussion in the patient’s electronic health record. State boards of medicine can sanction physicians for “coercion” for prescribing cBHT products without mentioning alternatives, he added.
JoAnn Pinkerton, MD, professor of obstetrics and gynecology at the University of Virginia, Charlottesville, and executive director emeritus of NAMS, who attended the session, praised Dr. Kauffman for providing a balanced and evidence-based overview of the subject.
“There are issues concerning safety, contaminants, and not knowing exactly what dose you’re getting,” with compounded hormones, Dr. Pinkerton said. “They’re being hyped as safer and more effective when in reality, we don’t have any studies that show that information.”
Dr. Pinkerton noted that while a compounded form of physiological testosterone might be relatively reliable, “if you’re using something like a pellet that is super physiologic with incredibly high doses, that you really don’t have any information to stand on that it’s safe or effective ... it might be putting your license at risk.”
A version of this article first appeared on Medscape.com.
BALTIMORE – according to an expert at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists (ACOG).
Clinicians write an estimated 26 to 33 million prescriptions for compounded bioidentical hormone therapy (cBHT) every year, and almost 41% of menopausal women who need treatment try cBHT during their lives. But these drugs lack the approval for this indication from the Food and Drug Administration.
“There is a public perception that this is natural, safer, and anti-aging,” said Robert Kauffman, MD, a professor of obstetrics and gynecology and assistant dean for research at Texas Tech University Health Sciences Center in Amarillo.
Following the 2002 Women’s Health Initiative report showing a link between hormone therapy (HT) and an increase in the incidence of breast cancer, medical schools have slowed or paused instructing trainees on the traditional treatment, Dr. Kauffman said. The association was later determined to be spurious: HT is not associated with a risk for all-cause mortality or deaths from cardiovascular disease or cancer. However, HT still is largely ignored by younger physicians, Dr. Kauffman said, because of unsubstantiated “dangers” such as heart attack, stroke, and deep vein thrombosis.
The lack of education on HT for medical school students and residents has “opened the door to unsubstantiated marketing claims and practices” for cBHT, Dr. Kauffman said. “Hence, the use of compounded bioidentical hormone therapy has increased” as clinicians look for alternatives.
Groups including ACOG, the North American Menopause Society (NAMS), and the U.S. Preventive Services Task Force recommend against the use of Non–FDA-approved therapies such as cBHT, except for narrow indications. Dr. Kauffman said that drug manufacturers have not conducted randomized controlled trials or observational studies on cBHT in treating menopause.
He cited studies showing quality problems with the compounding process of these drugs, and wide variations in the amount of actual ingredients from product labels. One 2021 study published in Menopause comparing patients taking cBHT or FDA-approved HT found that side effects were significantly higher in the cBHT group (57.6% vs. 14.8%; P < .0001).
But manufacturers of cBHT claim that their products prevent cardiovascular disease and Alzheimer’s disease and decrease the risk for breast cancer and stroke – assertions that are at best unproven, according to Dr. Kauffman.
The National Academies of Sciences, Engineering, and Medicine in 2020 said that clinicians have a duty to inform patients of the insufficient evidence to support clinical use of cBHT and should prescribe the products only to patients with documented allergies to an active ingredient in an FDA-approved agent or who require an alternative dosage.
Patients may also have to pay much more out of pocket for cBHT products because they often are not covered by insurance. Generic HT products, meanwhile, are relatively inexpensive and typically are covered, he noted.
“We have to be careful to avoid financial harm to patients by prescribing things, which are much more expensive than those which are usually available,” Dr. Kauffman said.
Prescribing any non–FDA-approved product, especially when biosimilars are available, places physicians at legal risk, Dr. Kauffman said. Physicians who recommend cBHT should inform patients that the products are not FDA approved and carefully document this discussion in the patient’s electronic health record. State boards of medicine can sanction physicians for “coercion” for prescribing cBHT products without mentioning alternatives, he added.
JoAnn Pinkerton, MD, professor of obstetrics and gynecology at the University of Virginia, Charlottesville, and executive director emeritus of NAMS, who attended the session, praised Dr. Kauffman for providing a balanced and evidence-based overview of the subject.
“There are issues concerning safety, contaminants, and not knowing exactly what dose you’re getting,” with compounded hormones, Dr. Pinkerton said. “They’re being hyped as safer and more effective when in reality, we don’t have any studies that show that information.”
Dr. Pinkerton noted that while a compounded form of physiological testosterone might be relatively reliable, “if you’re using something like a pellet that is super physiologic with incredibly high doses, that you really don’t have any information to stand on that it’s safe or effective ... it might be putting your license at risk.”
A version of this article first appeared on Medscape.com.
BALTIMORE – according to an expert at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists (ACOG).
Clinicians write an estimated 26 to 33 million prescriptions for compounded bioidentical hormone therapy (cBHT) every year, and almost 41% of menopausal women who need treatment try cBHT during their lives. But these drugs lack the approval for this indication from the Food and Drug Administration.
“There is a public perception that this is natural, safer, and anti-aging,” said Robert Kauffman, MD, a professor of obstetrics and gynecology and assistant dean for research at Texas Tech University Health Sciences Center in Amarillo.
Following the 2002 Women’s Health Initiative report showing a link between hormone therapy (HT) and an increase in the incidence of breast cancer, medical schools have slowed or paused instructing trainees on the traditional treatment, Dr. Kauffman said. The association was later determined to be spurious: HT is not associated with a risk for all-cause mortality or deaths from cardiovascular disease or cancer. However, HT still is largely ignored by younger physicians, Dr. Kauffman said, because of unsubstantiated “dangers” such as heart attack, stroke, and deep vein thrombosis.
The lack of education on HT for medical school students and residents has “opened the door to unsubstantiated marketing claims and practices” for cBHT, Dr. Kauffman said. “Hence, the use of compounded bioidentical hormone therapy has increased” as clinicians look for alternatives.
Groups including ACOG, the North American Menopause Society (NAMS), and the U.S. Preventive Services Task Force recommend against the use of Non–FDA-approved therapies such as cBHT, except for narrow indications. Dr. Kauffman said that drug manufacturers have not conducted randomized controlled trials or observational studies on cBHT in treating menopause.
He cited studies showing quality problems with the compounding process of these drugs, and wide variations in the amount of actual ingredients from product labels. One 2021 study published in Menopause comparing patients taking cBHT or FDA-approved HT found that side effects were significantly higher in the cBHT group (57.6% vs. 14.8%; P < .0001).
But manufacturers of cBHT claim that their products prevent cardiovascular disease and Alzheimer’s disease and decrease the risk for breast cancer and stroke – assertions that are at best unproven, according to Dr. Kauffman.
The National Academies of Sciences, Engineering, and Medicine in 2020 said that clinicians have a duty to inform patients of the insufficient evidence to support clinical use of cBHT and should prescribe the products only to patients with documented allergies to an active ingredient in an FDA-approved agent or who require an alternative dosage.
Patients may also have to pay much more out of pocket for cBHT products because they often are not covered by insurance. Generic HT products, meanwhile, are relatively inexpensive and typically are covered, he noted.
“We have to be careful to avoid financial harm to patients by prescribing things, which are much more expensive than those which are usually available,” Dr. Kauffman said.
Prescribing any non–FDA-approved product, especially when biosimilars are available, places physicians at legal risk, Dr. Kauffman said. Physicians who recommend cBHT should inform patients that the products are not FDA approved and carefully document this discussion in the patient’s electronic health record. State boards of medicine can sanction physicians for “coercion” for prescribing cBHT products without mentioning alternatives, he added.
JoAnn Pinkerton, MD, professor of obstetrics and gynecology at the University of Virginia, Charlottesville, and executive director emeritus of NAMS, who attended the session, praised Dr. Kauffman for providing a balanced and evidence-based overview of the subject.
“There are issues concerning safety, contaminants, and not knowing exactly what dose you’re getting,” with compounded hormones, Dr. Pinkerton said. “They’re being hyped as safer and more effective when in reality, we don’t have any studies that show that information.”
Dr. Pinkerton noted that while a compounded form of physiological testosterone might be relatively reliable, “if you’re using something like a pellet that is super physiologic with incredibly high doses, that you really don’t have any information to stand on that it’s safe or effective ... it might be putting your license at risk.”
A version of this article first appeared on Medscape.com.
AT ACOG 2023
Investigational lupus drug cenerimod moves to phase 3 studies after equivocal phase 2 results
SEOUL, SOUTH KOREA – A once-daily, oral dose of the investigational drug cenerimod, developed for the treatment of systemic lupus erythematosus, has shown a greater response rate among individuals with more severe disease, according to data presented at an international congress on SLE.
Cenerimod is a potent, highly-selective sphingosine 1–phosphate receptor 1 (S1P1) modulator with attenuated calcium signaling, which targets an important signaling molecule in immunity and cell migration, said rheumatologist Sandra Navarra, MD, of the University of Santo Tomas Hospital and St. Luke’s Medical Center in Manila, Philippines.
“It reduces the migration of T cells and B cells from the lymph nodes to the circulation into the tissues,” Dr. Navarra told the conference. S1P1 receptor modulators are already approved for treatment of multiple sclerosis, but cenerimod is the first to be explored for the treatment of lupus.
Dr. Navarra presented data from the international CARE study, a randomized, placebo-controlled, phase 2 study involving 427 patients with moderate to severe SLE.
Patients had to have been diagnosed at least 6 months before screening, be on stable lupus medications, and have abnormal antinuclear or anti–double stranded DNA antibodies. They were randomized to either 0.5 mg, 1 mg, 2 mg, or 4 mg of cenerimod daily or placebo for 12 months. At 6 months, the patients who had initially been randomized to 4 mg daily were rerandomized either to 2 mg daily or placebo.
While the study found that 4 mg of cenerimod was associated with a reduction in disease activity from baseline to month 6 on the modified Systemic Lupus Erythematosus Disease Activity Index–2000 score (excluding leukopenia), compared with placebo (P = .029). However, the final result was not statistically significant after adjustment for the multiplicity of tests for the four doses against placebo.
But the researchers saw a greater response among individuals with higher levels of interferon type 1 gene expression at baseline, as well as those with higher anti-dsDNA and lower C4 levels, which “makes sense,” Dr. Navarra said in an interview, because those were the sicker patients with “more inflammatory, more active disease.”
The study did exclude patients with active lupus nephritis, severe active central nervous system lupus, or severe cardiovascular disorders.
Dr. Navarra said the findings are now factored into patient selection for two phase 3 trials, called OPUS-1 and OPUS-2, which are now underway. The OPUS trials have revised eligibility criteria, as well as a screening period of up to 60 days to ensure that only patients with true moderate to severe SLE are enrolled.
The drug was well tolerated, with the rate of adverse events similar across all study groups. The adverse events of particular interest – hypertension, infections and infestations, and eye disorders – were all mild and transient. There were a greater number of reports of hypertension among those taking 1-mg and 4-mg doses of cenerimod, but Dr. Navarra said monthly measurements of systolic or diastolic blood pressure didn’t show any change.
The study was funded by cenerimod manufacturer Idorsia Pharmaceuticals. Dr. Navarra has financial relationships with Biogen, Astellas, Janssen, Novartis, Pfizer, Boehringer-Ingelheim, and GlaxoSmithKline.
SEOUL, SOUTH KOREA – A once-daily, oral dose of the investigational drug cenerimod, developed for the treatment of systemic lupus erythematosus, has shown a greater response rate among individuals with more severe disease, according to data presented at an international congress on SLE.
Cenerimod is a potent, highly-selective sphingosine 1–phosphate receptor 1 (S1P1) modulator with attenuated calcium signaling, which targets an important signaling molecule in immunity and cell migration, said rheumatologist Sandra Navarra, MD, of the University of Santo Tomas Hospital and St. Luke’s Medical Center in Manila, Philippines.
“It reduces the migration of T cells and B cells from the lymph nodes to the circulation into the tissues,” Dr. Navarra told the conference. S1P1 receptor modulators are already approved for treatment of multiple sclerosis, but cenerimod is the first to be explored for the treatment of lupus.
Dr. Navarra presented data from the international CARE study, a randomized, placebo-controlled, phase 2 study involving 427 patients with moderate to severe SLE.
Patients had to have been diagnosed at least 6 months before screening, be on stable lupus medications, and have abnormal antinuclear or anti–double stranded DNA antibodies. They were randomized to either 0.5 mg, 1 mg, 2 mg, or 4 mg of cenerimod daily or placebo for 12 months. At 6 months, the patients who had initially been randomized to 4 mg daily were rerandomized either to 2 mg daily or placebo.
While the study found that 4 mg of cenerimod was associated with a reduction in disease activity from baseline to month 6 on the modified Systemic Lupus Erythematosus Disease Activity Index–2000 score (excluding leukopenia), compared with placebo (P = .029). However, the final result was not statistically significant after adjustment for the multiplicity of tests for the four doses against placebo.
But the researchers saw a greater response among individuals with higher levels of interferon type 1 gene expression at baseline, as well as those with higher anti-dsDNA and lower C4 levels, which “makes sense,” Dr. Navarra said in an interview, because those were the sicker patients with “more inflammatory, more active disease.”
The study did exclude patients with active lupus nephritis, severe active central nervous system lupus, or severe cardiovascular disorders.
Dr. Navarra said the findings are now factored into patient selection for two phase 3 trials, called OPUS-1 and OPUS-2, which are now underway. The OPUS trials have revised eligibility criteria, as well as a screening period of up to 60 days to ensure that only patients with true moderate to severe SLE are enrolled.
The drug was well tolerated, with the rate of adverse events similar across all study groups. The adverse events of particular interest – hypertension, infections and infestations, and eye disorders – were all mild and transient. There were a greater number of reports of hypertension among those taking 1-mg and 4-mg doses of cenerimod, but Dr. Navarra said monthly measurements of systolic or diastolic blood pressure didn’t show any change.
The study was funded by cenerimod manufacturer Idorsia Pharmaceuticals. Dr. Navarra has financial relationships with Biogen, Astellas, Janssen, Novartis, Pfizer, Boehringer-Ingelheim, and GlaxoSmithKline.
SEOUL, SOUTH KOREA – A once-daily, oral dose of the investigational drug cenerimod, developed for the treatment of systemic lupus erythematosus, has shown a greater response rate among individuals with more severe disease, according to data presented at an international congress on SLE.
Cenerimod is a potent, highly-selective sphingosine 1–phosphate receptor 1 (S1P1) modulator with attenuated calcium signaling, which targets an important signaling molecule in immunity and cell migration, said rheumatologist Sandra Navarra, MD, of the University of Santo Tomas Hospital and St. Luke’s Medical Center in Manila, Philippines.
“It reduces the migration of T cells and B cells from the lymph nodes to the circulation into the tissues,” Dr. Navarra told the conference. S1P1 receptor modulators are already approved for treatment of multiple sclerosis, but cenerimod is the first to be explored for the treatment of lupus.
Dr. Navarra presented data from the international CARE study, a randomized, placebo-controlled, phase 2 study involving 427 patients with moderate to severe SLE.
Patients had to have been diagnosed at least 6 months before screening, be on stable lupus medications, and have abnormal antinuclear or anti–double stranded DNA antibodies. They were randomized to either 0.5 mg, 1 mg, 2 mg, or 4 mg of cenerimod daily or placebo for 12 months. At 6 months, the patients who had initially been randomized to 4 mg daily were rerandomized either to 2 mg daily or placebo.
While the study found that 4 mg of cenerimod was associated with a reduction in disease activity from baseline to month 6 on the modified Systemic Lupus Erythematosus Disease Activity Index–2000 score (excluding leukopenia), compared with placebo (P = .029). However, the final result was not statistically significant after adjustment for the multiplicity of tests for the four doses against placebo.
But the researchers saw a greater response among individuals with higher levels of interferon type 1 gene expression at baseline, as well as those with higher anti-dsDNA and lower C4 levels, which “makes sense,” Dr. Navarra said in an interview, because those were the sicker patients with “more inflammatory, more active disease.”
The study did exclude patients with active lupus nephritis, severe active central nervous system lupus, or severe cardiovascular disorders.
Dr. Navarra said the findings are now factored into patient selection for two phase 3 trials, called OPUS-1 and OPUS-2, which are now underway. The OPUS trials have revised eligibility criteria, as well as a screening period of up to 60 days to ensure that only patients with true moderate to severe SLE are enrolled.
The drug was well tolerated, with the rate of adverse events similar across all study groups. The adverse events of particular interest – hypertension, infections and infestations, and eye disorders – were all mild and transient. There were a greater number of reports of hypertension among those taking 1-mg and 4-mg doses of cenerimod, but Dr. Navarra said monthly measurements of systolic or diastolic blood pressure didn’t show any change.
The study was funded by cenerimod manufacturer Idorsia Pharmaceuticals. Dr. Navarra has financial relationships with Biogen, Astellas, Janssen, Novartis, Pfizer, Boehringer-Ingelheim, and GlaxoSmithKline.
AT LUPUS 2023
Serious mental illness not a factor in most mass school shootings
Mass shootings, often on school campuses, have become a regular and sad reality in the United States.
The statistics are grim. Every day 12 children die from gun violence in America and another 32 are shot and injured. Since the Columbine High School shooting in 1999, more than 338,000 students in the United States have experienced school gun violence, according to the nonprofit organization Sandy Hook Promise.
A new analysis from the Columbia Mass Murder Database (CMMD) sheds fresh light on the debate over whether mental illness or easy access to guns is the key driver of mass shootings.
The findings, which are published in the Journal of Forensic Sciences, show that most perpetrators of mass school shootings are young, White men without serious mental illness.
A ‘straw man’
Mental health is often used as a “straw man” in debates about mass shootings, lead investigator Ragy Girgis, MD, told this news organization.
“There are many factors that contribute to the mass shooting epidemic, including gun access, criminality, substance use and misuse, and many others. Mental illness is incidental in the vast majority of cases,” said Dr. Girgis, with Columbia University Irving Medical Center, New York, and the New York State Psychiatric Institute.
“People with serious mental illness constitute only a small portion of the perpetrators of gun violence in this country,” coinvestigator Paul Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University, New York, told this news organization.
Using the CMMD, the researchers examined 82 incidents of mass murder perpetrated in academic settings including schools, colleges, and universities. The average number of victims of these incidents was eight. More than half (60%) of mass school shootings involved at least one semi- or fully automatic firearm.
All 82 incidents were initiated by men (mean age, 28), and 67% were White. About two-thirds (63%) involved guns.
More than three-quarters (77%) of all perpetrators of mass murders in academic settings had no recorded history of psychotic symptoms.
Despite the absence of serious mental illness, almost half (46%) of the mass school shooters took their own lives at the scene, suggesting that they viewed themselves as engaging in some form of “final act,” the researchers note.
“The major difference between mass shooters in school settings and elsewhere is the higher rate of suicide by the perpetrators in school settings. That suggests that the shootings are often part of a preexisting intent to die on the part of the shooter,” said Dr. Appelbaum.
Epidemic of emptiness
He noted that the typical profile of a mass school shooter is that of “a young male with anger problems, often as a result of bullying or abuse, frequently described as a loner, who has signaled a desire to kill other people.”
“If we only focus on mental illness, we will miss the warning signs in the majority of cases associated with victimization (such as bullying) and consequent anger,” Dr. Appelbaum said.
Dr. Girgis said there is a need to deal with the “epidemic of emptiness, narcissism, anger, and societal rejection felt by many young men/boys who, when combined with a desire to take their own lives and a great need for notoriety, feel that perpetrating a mass school shooting is their only option.”
“We also need to understand why it is so easy for so many mass school shooters to obtain firearms that are not theirs – either illegally or from someone else who themselves may have obtained the firearm legally,” Dr. Girgis said.
“All countries have people with mental illness,” Dr. Appelbaum said, “but among developed countries the U.S. is unique in the easy availability of weapons and in our disproportionate rate of murders.”
He also noted that school shootings are not a problem that clinicians are going to solve.
“Although they can be alert to signals from their patients of an intent to harm people in a school (or other) setting, the vast majority of shooters are not receiving treatment for a mental disorder,” Dr. Appelbaum said.
“This is a problem that can only be substantially diminished by reducing access to firearms, which includes requirements for safe storage, universal background checks, waiting periods to purchase firearms, and similar means-oriented interventions,” he added.
Need for regular mental health checks
Thea Gallagher, PsyD, who was not involved in the study, noted that mass school shooters may not have a psychotic illness, but with mental health there is a “spectrum, and obviously, that individual is struggling to some extent, most likely, mentally, if they are at a place where they are willing to take the lives of others and themselves.”
“We need to understand more about how people get to this place and the issues people are struggling with. We need to push for yearly mental health checks just like the yearly physical,” Dr. Gallagher, with the department of psychiatry at NYU Langone Health, New York, told this news organization.
“The more that we create conversation and moments to talk about how people are feeling internally, the better chance we have to give people who are struggling healthy coping strategies and the opportunity to process their emotions and not bury them,” Dr. Gallagher said.
Support for the study was provided in part by the New York State Office of Mental Hygiene, and the Elizabeth K. Dollard Charitable Trust. Dr. Girgis has received royalties and/or advances from books on mental health published by Wipf and Stock, and Routledge/Taylor and Francis. He has consulted for Noble Insights, IMS Expert Services, and Fowler White Burnett. Dr. Appelbaum and Dr. Gallagher report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mass shootings, often on school campuses, have become a regular and sad reality in the United States.
The statistics are grim. Every day 12 children die from gun violence in America and another 32 are shot and injured. Since the Columbine High School shooting in 1999, more than 338,000 students in the United States have experienced school gun violence, according to the nonprofit organization Sandy Hook Promise.
A new analysis from the Columbia Mass Murder Database (CMMD) sheds fresh light on the debate over whether mental illness or easy access to guns is the key driver of mass shootings.
The findings, which are published in the Journal of Forensic Sciences, show that most perpetrators of mass school shootings are young, White men without serious mental illness.
A ‘straw man’
Mental health is often used as a “straw man” in debates about mass shootings, lead investigator Ragy Girgis, MD, told this news organization.
“There are many factors that contribute to the mass shooting epidemic, including gun access, criminality, substance use and misuse, and many others. Mental illness is incidental in the vast majority of cases,” said Dr. Girgis, with Columbia University Irving Medical Center, New York, and the New York State Psychiatric Institute.
“People with serious mental illness constitute only a small portion of the perpetrators of gun violence in this country,” coinvestigator Paul Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University, New York, told this news organization.
Using the CMMD, the researchers examined 82 incidents of mass murder perpetrated in academic settings including schools, colleges, and universities. The average number of victims of these incidents was eight. More than half (60%) of mass school shootings involved at least one semi- or fully automatic firearm.
All 82 incidents were initiated by men (mean age, 28), and 67% were White. About two-thirds (63%) involved guns.
More than three-quarters (77%) of all perpetrators of mass murders in academic settings had no recorded history of psychotic symptoms.
Despite the absence of serious mental illness, almost half (46%) of the mass school shooters took their own lives at the scene, suggesting that they viewed themselves as engaging in some form of “final act,” the researchers note.
“The major difference between mass shooters in school settings and elsewhere is the higher rate of suicide by the perpetrators in school settings. That suggests that the shootings are often part of a preexisting intent to die on the part of the shooter,” said Dr. Appelbaum.
Epidemic of emptiness
He noted that the typical profile of a mass school shooter is that of “a young male with anger problems, often as a result of bullying or abuse, frequently described as a loner, who has signaled a desire to kill other people.”
“If we only focus on mental illness, we will miss the warning signs in the majority of cases associated with victimization (such as bullying) and consequent anger,” Dr. Appelbaum said.
Dr. Girgis said there is a need to deal with the “epidemic of emptiness, narcissism, anger, and societal rejection felt by many young men/boys who, when combined with a desire to take their own lives and a great need for notoriety, feel that perpetrating a mass school shooting is their only option.”
“We also need to understand why it is so easy for so many mass school shooters to obtain firearms that are not theirs – either illegally or from someone else who themselves may have obtained the firearm legally,” Dr. Girgis said.
“All countries have people with mental illness,” Dr. Appelbaum said, “but among developed countries the U.S. is unique in the easy availability of weapons and in our disproportionate rate of murders.”
He also noted that school shootings are not a problem that clinicians are going to solve.
“Although they can be alert to signals from their patients of an intent to harm people in a school (or other) setting, the vast majority of shooters are not receiving treatment for a mental disorder,” Dr. Appelbaum said.
“This is a problem that can only be substantially diminished by reducing access to firearms, which includes requirements for safe storage, universal background checks, waiting periods to purchase firearms, and similar means-oriented interventions,” he added.
Need for regular mental health checks
Thea Gallagher, PsyD, who was not involved in the study, noted that mass school shooters may not have a psychotic illness, but with mental health there is a “spectrum, and obviously, that individual is struggling to some extent, most likely, mentally, if they are at a place where they are willing to take the lives of others and themselves.”
“We need to understand more about how people get to this place and the issues people are struggling with. We need to push for yearly mental health checks just like the yearly physical,” Dr. Gallagher, with the department of psychiatry at NYU Langone Health, New York, told this news organization.
“The more that we create conversation and moments to talk about how people are feeling internally, the better chance we have to give people who are struggling healthy coping strategies and the opportunity to process their emotions and not bury them,” Dr. Gallagher said.
Support for the study was provided in part by the New York State Office of Mental Hygiene, and the Elizabeth K. Dollard Charitable Trust. Dr. Girgis has received royalties and/or advances from books on mental health published by Wipf and Stock, and Routledge/Taylor and Francis. He has consulted for Noble Insights, IMS Expert Services, and Fowler White Burnett. Dr. Appelbaum and Dr. Gallagher report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mass shootings, often on school campuses, have become a regular and sad reality in the United States.
The statistics are grim. Every day 12 children die from gun violence in America and another 32 are shot and injured. Since the Columbine High School shooting in 1999, more than 338,000 students in the United States have experienced school gun violence, according to the nonprofit organization Sandy Hook Promise.
A new analysis from the Columbia Mass Murder Database (CMMD) sheds fresh light on the debate over whether mental illness or easy access to guns is the key driver of mass shootings.
The findings, which are published in the Journal of Forensic Sciences, show that most perpetrators of mass school shootings are young, White men without serious mental illness.
A ‘straw man’
Mental health is often used as a “straw man” in debates about mass shootings, lead investigator Ragy Girgis, MD, told this news organization.
“There are many factors that contribute to the mass shooting epidemic, including gun access, criminality, substance use and misuse, and many others. Mental illness is incidental in the vast majority of cases,” said Dr. Girgis, with Columbia University Irving Medical Center, New York, and the New York State Psychiatric Institute.
“People with serious mental illness constitute only a small portion of the perpetrators of gun violence in this country,” coinvestigator Paul Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University, New York, told this news organization.
Using the CMMD, the researchers examined 82 incidents of mass murder perpetrated in academic settings including schools, colleges, and universities. The average number of victims of these incidents was eight. More than half (60%) of mass school shootings involved at least one semi- or fully automatic firearm.
All 82 incidents were initiated by men (mean age, 28), and 67% were White. About two-thirds (63%) involved guns.
More than three-quarters (77%) of all perpetrators of mass murders in academic settings had no recorded history of psychotic symptoms.
Despite the absence of serious mental illness, almost half (46%) of the mass school shooters took their own lives at the scene, suggesting that they viewed themselves as engaging in some form of “final act,” the researchers note.
“The major difference between mass shooters in school settings and elsewhere is the higher rate of suicide by the perpetrators in school settings. That suggests that the shootings are often part of a preexisting intent to die on the part of the shooter,” said Dr. Appelbaum.
Epidemic of emptiness
He noted that the typical profile of a mass school shooter is that of “a young male with anger problems, often as a result of bullying or abuse, frequently described as a loner, who has signaled a desire to kill other people.”
“If we only focus on mental illness, we will miss the warning signs in the majority of cases associated with victimization (such as bullying) and consequent anger,” Dr. Appelbaum said.
Dr. Girgis said there is a need to deal with the “epidemic of emptiness, narcissism, anger, and societal rejection felt by many young men/boys who, when combined with a desire to take their own lives and a great need for notoriety, feel that perpetrating a mass school shooting is their only option.”
“We also need to understand why it is so easy for so many mass school shooters to obtain firearms that are not theirs – either illegally or from someone else who themselves may have obtained the firearm legally,” Dr. Girgis said.
“All countries have people with mental illness,” Dr. Appelbaum said, “but among developed countries the U.S. is unique in the easy availability of weapons and in our disproportionate rate of murders.”
He also noted that school shootings are not a problem that clinicians are going to solve.
“Although they can be alert to signals from their patients of an intent to harm people in a school (or other) setting, the vast majority of shooters are not receiving treatment for a mental disorder,” Dr. Appelbaum said.
“This is a problem that can only be substantially diminished by reducing access to firearms, which includes requirements for safe storage, universal background checks, waiting periods to purchase firearms, and similar means-oriented interventions,” he added.
Need for regular mental health checks
Thea Gallagher, PsyD, who was not involved in the study, noted that mass school shooters may not have a psychotic illness, but with mental health there is a “spectrum, and obviously, that individual is struggling to some extent, most likely, mentally, if they are at a place where they are willing to take the lives of others and themselves.”
“We need to understand more about how people get to this place and the issues people are struggling with. We need to push for yearly mental health checks just like the yearly physical,” Dr. Gallagher, with the department of psychiatry at NYU Langone Health, New York, told this news organization.
“The more that we create conversation and moments to talk about how people are feeling internally, the better chance we have to give people who are struggling healthy coping strategies and the opportunity to process their emotions and not bury them,” Dr. Gallagher said.
Support for the study was provided in part by the New York State Office of Mental Hygiene, and the Elizabeth K. Dollard Charitable Trust. Dr. Girgis has received royalties and/or advances from books on mental health published by Wipf and Stock, and Routledge/Taylor and Francis. He has consulted for Noble Insights, IMS Expert Services, and Fowler White Burnett. Dr. Appelbaum and Dr. Gallagher report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Care bundle improves outcome in ICH: INTERACT3
The INTERACT3 study showed that timely administration of a care bundle that included early intensive lowering of systolic blood pressure, strict glucose control, treatment of fever, and rapid reversal of abnormal anticoagulation led to less disability, lower rates of death, and better overall quality of life.
“This is a groundbreaking result. It is the first-ever published trial in ICH patients to show a clear benefit on functional outcomes and on mortality,” lead investigator Craig Anderson, MD, director of global brain health at the George Institute for Global Health, Sydney, said in an interview.
“These results show that, if we can organize care and focus on optimal management of these four aspects of the health of the patient, they do better,” Dr. Anderson said.
‘Game changer’
“This is a game changer because now we have level A evidence showing something is definitely beneficial for these patients,” Dr. Anderson added. “That means hospitals have the imperative to organize their systems to do these things and maximize care. We have never had that before.”
Dr. Anderson noted that, while some previous studies have suggested benefit from various interventions, such as early lowering of blood pressure, the results have not been conclusive.
“This means the intervention has not always been implemented, leading to large variations in clinical practice. But now we have a package that is proven to work; this should become a guideline-recommended practice,” he commented.
The INTERACT-3 results were presented at the European Stroke Organisation conference in Munich. They were also simultaneously published online in The Lancet.
Dr. Anderson explained that, until now, there haven’t been any proven treatments for ICH. “There has been a lot of energy and research put into the field, but this has resulted in several interventions that are ‘probably useful’ or which have a level B recommendation,” he said. “No therapy has been shown to be beneficial in a totally conclusive way, so we are still not entirely sure exactly whether the treatments we use actually make a difference.”
The INTERACT3 researchers therefore decided to evaluate a care package consisting of a bundle of several treatments in the hope that they may have additive or synergistic effects.
The study involved 7,036 patients with imaging-confirmed spontaneous ICH who presented within 6 hours of symptom onset to one of 121 hospitals in 10 mainly low- and middle-income countries: Brazil, China, India, Mexico, Nigeria, Pakistan, Peru, Sri Lanka, Vietnam, and Chile.
Using a cluster design, all hospitals started with usual care as a control and then at some point during the study started using the care bundle intervention.
The care-bundle protocol included the early intensive lowering of systolic blood pressure (target, < 140 mm Hg), strict glucose control (target, 6.1-7.8 mmol/L in those without diabetes and 7.8-10.0 mmol/L in those with diabetes), antipyrexia treatment (target body temperature, ≤ 37.5° C), and rapid reversal of warfarin-related anticoagulation (target international normalized ratio, 1.5) in patients for whom these variables were abnormal.
Overall, the modified intention-to-treat population included 3,221 patients who were assigned to the care-bundle group and 3,815 who were assigned to the usual-care group. Primary outcome data were available for 2,892 patients in the care-bundle group and 3,363 patients in the usual-care group.
The primary outcome was functional recovery, measured with the Modified Rankin Scale at 6 months. Results show that the likelihood of a poor functional outcome was lower in the care-bundle group (common odds ratio, 0.86; P = .015).
Patients who received the interventional care bundle also had a significantly lower rate of serious adverse events (16.0% vs. 20.1%) and mortality (14.1% vs. 17.0%).
NNT of 35 to save one life free of disability
“The number needed to treat (NNT) is just 35 to save a life free of disability,” Dr. Anderson commented. “That’s pretty good. We estimate that this care bundle would save tens of thousands of lives a year if universally adopted.”
The intervention group also spent less time in hospital and had improved health-related quality of life.
Dr. Anderson pointed out that the interventions included in the care bundle were all relatively easy to perform.
“They just require a bit more nursing time and the use of a few inexpensive medicines and maybe infusion pumps, but we’re not talking about the need for skilled surgery or a new therapy costing hundreds of thousands of dollars, so this care bundle should be very straightforward to implement. While we haven’t done a formal cost-effectiveness analysis, I would say it will definitely be good value for money.”
Dr. Anderson believes the rapid lowering of blood pressure is a very important part of the care bundle. He noted that target levels were achieved, on average, in 2.3 hours, compared with 4.0 hours in the control group. But he stressed that this was not just a trial of blood pressure reduction and that the whole package is important.
He gave a couple of possible reasons why this trial was successful whereas previous trials did not show a clear benefit of blood pressure lowering in ICH.
“Firstly, it was a very large trial with more than 7,000 patients – that is more than three times larger than any other trial in ICH. And secondly, the package of care means there are several different interventions that together show a real benefit,” he said. “It’s like the polypill, or a rehabilitation program – if you put several different things together, the whole package can show really positive results.”
Dr. Anderson also pointed out that the study included a wide spectrum of ICH patients, and the benefit of the care bundle was seen across all groups and all stroke severities.
“There were a lot of patients with a large ICH, and if anything, they showed an even larger benefit with the bundle of care,” he said.
The researchers note that the burden of ICH is greatest in low- and middle-income countries. In 2019, 30% of all stroke cases in these countries were ICH, almost double the proportion seen in high-income countries (16%). This is in part attributable to high rates of hypertension and limited resources for primary prevention, including identification and management of stroke risk factors by health care services.
‘Outstanding example’ of less therapeutic negativity
Lili Song, MD, PhD, joint lead author and head of the Stroke Program at the George Institute China, Beijing, said, “A lack of proven treatments for ICH has led to a pessimistic view that not much can be done for these patients.
“However, with INTERACT3, we demonstrate on a large scale how readily available treatments can be used to improve outcomes in resource-limited settings,” she said. “We hope this evidence will inform clinical practice guidelines across the globe and help save many lives.”
In a comment that accompanied the article, Wendy Ziai, MD, Matthew Bower, MD, and Daniel Hanley, MD, Johns Hopkins University, Baltimore, say the INTERACT3 study shows that “an intracerebral hemorrhage care bundle focused on physiological control interventions, whether synergistic or not, might promote better outcomes in hospitals where care has not previously optimized sustained interventions.”
Pointing out that the care bundle has minimal risks of cost and coordination and a high public health effect, they conclude: “This effort is an outstanding example of why less therapeutic negativity, and more intervention might benefit survivors of intracerebral hemorrhage.”
The INTERACT3 study was funded by the Department of Health and Social Care, the Foreign, Commonwealth and Development Office, the Medical Research Council, and the Wellcome Trust (all in the United Kingdom), the West China Hospital Outstanding Discipline Development 1–3-5 Programme, the National Health and Medical Research Council of Australia, Sichuan Credit Pharmaceutical, and Takeda (China).
A version of this article first appeared on Medscape.com.
The INTERACT3 study showed that timely administration of a care bundle that included early intensive lowering of systolic blood pressure, strict glucose control, treatment of fever, and rapid reversal of abnormal anticoagulation led to less disability, lower rates of death, and better overall quality of life.
“This is a groundbreaking result. It is the first-ever published trial in ICH patients to show a clear benefit on functional outcomes and on mortality,” lead investigator Craig Anderson, MD, director of global brain health at the George Institute for Global Health, Sydney, said in an interview.
“These results show that, if we can organize care and focus on optimal management of these four aspects of the health of the patient, they do better,” Dr. Anderson said.
‘Game changer’
“This is a game changer because now we have level A evidence showing something is definitely beneficial for these patients,” Dr. Anderson added. “That means hospitals have the imperative to organize their systems to do these things and maximize care. We have never had that before.”
Dr. Anderson noted that, while some previous studies have suggested benefit from various interventions, such as early lowering of blood pressure, the results have not been conclusive.
“This means the intervention has not always been implemented, leading to large variations in clinical practice. But now we have a package that is proven to work; this should become a guideline-recommended practice,” he commented.
The INTERACT-3 results were presented at the European Stroke Organisation conference in Munich. They were also simultaneously published online in The Lancet.
Dr. Anderson explained that, until now, there haven’t been any proven treatments for ICH. “There has been a lot of energy and research put into the field, but this has resulted in several interventions that are ‘probably useful’ or which have a level B recommendation,” he said. “No therapy has been shown to be beneficial in a totally conclusive way, so we are still not entirely sure exactly whether the treatments we use actually make a difference.”
The INTERACT3 researchers therefore decided to evaluate a care package consisting of a bundle of several treatments in the hope that they may have additive or synergistic effects.
The study involved 7,036 patients with imaging-confirmed spontaneous ICH who presented within 6 hours of symptom onset to one of 121 hospitals in 10 mainly low- and middle-income countries: Brazil, China, India, Mexico, Nigeria, Pakistan, Peru, Sri Lanka, Vietnam, and Chile.
Using a cluster design, all hospitals started with usual care as a control and then at some point during the study started using the care bundle intervention.
The care-bundle protocol included the early intensive lowering of systolic blood pressure (target, < 140 mm Hg), strict glucose control (target, 6.1-7.8 mmol/L in those without diabetes and 7.8-10.0 mmol/L in those with diabetes), antipyrexia treatment (target body temperature, ≤ 37.5° C), and rapid reversal of warfarin-related anticoagulation (target international normalized ratio, 1.5) in patients for whom these variables were abnormal.
Overall, the modified intention-to-treat population included 3,221 patients who were assigned to the care-bundle group and 3,815 who were assigned to the usual-care group. Primary outcome data were available for 2,892 patients in the care-bundle group and 3,363 patients in the usual-care group.
The primary outcome was functional recovery, measured with the Modified Rankin Scale at 6 months. Results show that the likelihood of a poor functional outcome was lower in the care-bundle group (common odds ratio, 0.86; P = .015).
Patients who received the interventional care bundle also had a significantly lower rate of serious adverse events (16.0% vs. 20.1%) and mortality (14.1% vs. 17.0%).
NNT of 35 to save one life free of disability
“The number needed to treat (NNT) is just 35 to save a life free of disability,” Dr. Anderson commented. “That’s pretty good. We estimate that this care bundle would save tens of thousands of lives a year if universally adopted.”
The intervention group also spent less time in hospital and had improved health-related quality of life.
Dr. Anderson pointed out that the interventions included in the care bundle were all relatively easy to perform.
“They just require a bit more nursing time and the use of a few inexpensive medicines and maybe infusion pumps, but we’re not talking about the need for skilled surgery or a new therapy costing hundreds of thousands of dollars, so this care bundle should be very straightforward to implement. While we haven’t done a formal cost-effectiveness analysis, I would say it will definitely be good value for money.”
Dr. Anderson believes the rapid lowering of blood pressure is a very important part of the care bundle. He noted that target levels were achieved, on average, in 2.3 hours, compared with 4.0 hours in the control group. But he stressed that this was not just a trial of blood pressure reduction and that the whole package is important.
He gave a couple of possible reasons why this trial was successful whereas previous trials did not show a clear benefit of blood pressure lowering in ICH.
“Firstly, it was a very large trial with more than 7,000 patients – that is more than three times larger than any other trial in ICH. And secondly, the package of care means there are several different interventions that together show a real benefit,” he said. “It’s like the polypill, or a rehabilitation program – if you put several different things together, the whole package can show really positive results.”
Dr. Anderson also pointed out that the study included a wide spectrum of ICH patients, and the benefit of the care bundle was seen across all groups and all stroke severities.
“There were a lot of patients with a large ICH, and if anything, they showed an even larger benefit with the bundle of care,” he said.
The researchers note that the burden of ICH is greatest in low- and middle-income countries. In 2019, 30% of all stroke cases in these countries were ICH, almost double the proportion seen in high-income countries (16%). This is in part attributable to high rates of hypertension and limited resources for primary prevention, including identification and management of stroke risk factors by health care services.
‘Outstanding example’ of less therapeutic negativity
Lili Song, MD, PhD, joint lead author and head of the Stroke Program at the George Institute China, Beijing, said, “A lack of proven treatments for ICH has led to a pessimistic view that not much can be done for these patients.
“However, with INTERACT3, we demonstrate on a large scale how readily available treatments can be used to improve outcomes in resource-limited settings,” she said. “We hope this evidence will inform clinical practice guidelines across the globe and help save many lives.”
In a comment that accompanied the article, Wendy Ziai, MD, Matthew Bower, MD, and Daniel Hanley, MD, Johns Hopkins University, Baltimore, say the INTERACT3 study shows that “an intracerebral hemorrhage care bundle focused on physiological control interventions, whether synergistic or not, might promote better outcomes in hospitals where care has not previously optimized sustained interventions.”
Pointing out that the care bundle has minimal risks of cost and coordination and a high public health effect, they conclude: “This effort is an outstanding example of why less therapeutic negativity, and more intervention might benefit survivors of intracerebral hemorrhage.”
The INTERACT3 study was funded by the Department of Health and Social Care, the Foreign, Commonwealth and Development Office, the Medical Research Council, and the Wellcome Trust (all in the United Kingdom), the West China Hospital Outstanding Discipline Development 1–3-5 Programme, the National Health and Medical Research Council of Australia, Sichuan Credit Pharmaceutical, and Takeda (China).
A version of this article first appeared on Medscape.com.
The INTERACT3 study showed that timely administration of a care bundle that included early intensive lowering of systolic blood pressure, strict glucose control, treatment of fever, and rapid reversal of abnormal anticoagulation led to less disability, lower rates of death, and better overall quality of life.
“This is a groundbreaking result. It is the first-ever published trial in ICH patients to show a clear benefit on functional outcomes and on mortality,” lead investigator Craig Anderson, MD, director of global brain health at the George Institute for Global Health, Sydney, said in an interview.
“These results show that, if we can organize care and focus on optimal management of these four aspects of the health of the patient, they do better,” Dr. Anderson said.
‘Game changer’
“This is a game changer because now we have level A evidence showing something is definitely beneficial for these patients,” Dr. Anderson added. “That means hospitals have the imperative to organize their systems to do these things and maximize care. We have never had that before.”
Dr. Anderson noted that, while some previous studies have suggested benefit from various interventions, such as early lowering of blood pressure, the results have not been conclusive.
“This means the intervention has not always been implemented, leading to large variations in clinical practice. But now we have a package that is proven to work; this should become a guideline-recommended practice,” he commented.
The INTERACT-3 results were presented at the European Stroke Organisation conference in Munich. They were also simultaneously published online in The Lancet.
Dr. Anderson explained that, until now, there haven’t been any proven treatments for ICH. “There has been a lot of energy and research put into the field, but this has resulted in several interventions that are ‘probably useful’ or which have a level B recommendation,” he said. “No therapy has been shown to be beneficial in a totally conclusive way, so we are still not entirely sure exactly whether the treatments we use actually make a difference.”
The INTERACT3 researchers therefore decided to evaluate a care package consisting of a bundle of several treatments in the hope that they may have additive or synergistic effects.
The study involved 7,036 patients with imaging-confirmed spontaneous ICH who presented within 6 hours of symptom onset to one of 121 hospitals in 10 mainly low- and middle-income countries: Brazil, China, India, Mexico, Nigeria, Pakistan, Peru, Sri Lanka, Vietnam, and Chile.
Using a cluster design, all hospitals started with usual care as a control and then at some point during the study started using the care bundle intervention.
The care-bundle protocol included the early intensive lowering of systolic blood pressure (target, < 140 mm Hg), strict glucose control (target, 6.1-7.8 mmol/L in those without diabetes and 7.8-10.0 mmol/L in those with diabetes), antipyrexia treatment (target body temperature, ≤ 37.5° C), and rapid reversal of warfarin-related anticoagulation (target international normalized ratio, 1.5) in patients for whom these variables were abnormal.
Overall, the modified intention-to-treat population included 3,221 patients who were assigned to the care-bundle group and 3,815 who were assigned to the usual-care group. Primary outcome data were available for 2,892 patients in the care-bundle group and 3,363 patients in the usual-care group.
The primary outcome was functional recovery, measured with the Modified Rankin Scale at 6 months. Results show that the likelihood of a poor functional outcome was lower in the care-bundle group (common odds ratio, 0.86; P = .015).
Patients who received the interventional care bundle also had a significantly lower rate of serious adverse events (16.0% vs. 20.1%) and mortality (14.1% vs. 17.0%).
NNT of 35 to save one life free of disability
“The number needed to treat (NNT) is just 35 to save a life free of disability,” Dr. Anderson commented. “That’s pretty good. We estimate that this care bundle would save tens of thousands of lives a year if universally adopted.”
The intervention group also spent less time in hospital and had improved health-related quality of life.
Dr. Anderson pointed out that the interventions included in the care bundle were all relatively easy to perform.
“They just require a bit more nursing time and the use of a few inexpensive medicines and maybe infusion pumps, but we’re not talking about the need for skilled surgery or a new therapy costing hundreds of thousands of dollars, so this care bundle should be very straightforward to implement. While we haven’t done a formal cost-effectiveness analysis, I would say it will definitely be good value for money.”
Dr. Anderson believes the rapid lowering of blood pressure is a very important part of the care bundle. He noted that target levels were achieved, on average, in 2.3 hours, compared with 4.0 hours in the control group. But he stressed that this was not just a trial of blood pressure reduction and that the whole package is important.
He gave a couple of possible reasons why this trial was successful whereas previous trials did not show a clear benefit of blood pressure lowering in ICH.
“Firstly, it was a very large trial with more than 7,000 patients – that is more than three times larger than any other trial in ICH. And secondly, the package of care means there are several different interventions that together show a real benefit,” he said. “It’s like the polypill, or a rehabilitation program – if you put several different things together, the whole package can show really positive results.”
Dr. Anderson also pointed out that the study included a wide spectrum of ICH patients, and the benefit of the care bundle was seen across all groups and all stroke severities.
“There were a lot of patients with a large ICH, and if anything, they showed an even larger benefit with the bundle of care,” he said.
The researchers note that the burden of ICH is greatest in low- and middle-income countries. In 2019, 30% of all stroke cases in these countries were ICH, almost double the proportion seen in high-income countries (16%). This is in part attributable to high rates of hypertension and limited resources for primary prevention, including identification and management of stroke risk factors by health care services.
‘Outstanding example’ of less therapeutic negativity
Lili Song, MD, PhD, joint lead author and head of the Stroke Program at the George Institute China, Beijing, said, “A lack of proven treatments for ICH has led to a pessimistic view that not much can be done for these patients.
“However, with INTERACT3, we demonstrate on a large scale how readily available treatments can be used to improve outcomes in resource-limited settings,” she said. “We hope this evidence will inform clinical practice guidelines across the globe and help save many lives.”
In a comment that accompanied the article, Wendy Ziai, MD, Matthew Bower, MD, and Daniel Hanley, MD, Johns Hopkins University, Baltimore, say the INTERACT3 study shows that “an intracerebral hemorrhage care bundle focused on physiological control interventions, whether synergistic or not, might promote better outcomes in hospitals where care has not previously optimized sustained interventions.”
Pointing out that the care bundle has minimal risks of cost and coordination and a high public health effect, they conclude: “This effort is an outstanding example of why less therapeutic negativity, and more intervention might benefit survivors of intracerebral hemorrhage.”
The INTERACT3 study was funded by the Department of Health and Social Care, the Foreign, Commonwealth and Development Office, the Medical Research Council, and the Wellcome Trust (all in the United Kingdom), the West China Hospital Outstanding Discipline Development 1–3-5 Programme, the National Health and Medical Research Council of Australia, Sichuan Credit Pharmaceutical, and Takeda (China).
A version of this article first appeared on Medscape.com.
FROM ESO 2023
'Paradigm shift’: Luspatercept for MDS
“Luspatercept is the first and only therapy to demonstrate superiority in a head-to-head study against ESAs in [transfusion-dependent] LR-MDS,” first author Guillermo Garcia-Manero, MD, chief of the MDS section, department of leukemia, at the University of Texas MD Anderson Cancer Center, Houston, said in a premeeting press briefing in advance of the annual meeting of the American Society of Clinical Oncology.
“It should be considered a paradigm shift in the treatment of LR-MDS–associated anemia,” Dr. Garcia-Manero said.
Commenting on the study, Andrew Artz, MD, a professor at the Hematologic Malignancies Research Institute, City of Hope National Medical Center, Duarte, Calif., agreed that the results could be practice changing.
“We biologically expected luspatercept to best ESA [in ring sideroblast transfusion–dependent MDS], based on luspatercept often rescuing ESA failures in this setting,” Dr. Artz said in an interview.
“The results have the potential to change initial therapy for patients with low-risk red blood cell transfusion-dependent MDS,” he said.
In LR-MDS, which encompasses a variety of bone marrow disorders, chronic anemia is very common, and patients, who are typically elderly, can become burdened by developing dependencies on RBC transfusions.
Transfusion dependency, in addition to creating a host of challenges, can increase the risk of death by as much as 50%, compared with patients who are not transfusion dependent, Dr. Garcia-Manero noted.
While ESAs such as epoetin alfa are the first-line treatment for LR-MDS, patients who are dependent on transfusions are less likely to respond to the agents, hence “there is an unmet need for effective and durable options other than ESAs for treating anemia in patients with LR-MDS,” Dr. Garcia-Manero said.
Luspatercept, a first-in-class monoclonal antibody, has a mechanism of action that is distinct from ESAs, modulating the transforming growth factor–beta pathway and increasing erythrocytosis.
In the previous phase 3 MEDALIST trial, the drug was shown to have efficacy over placebo in reducing the severity of anemia in LR-MDS. In 2020, in what was deemed the first advance in MDS treatment in more than a decade, those results led to approval by the Food and Drug Administration for patients with LR-MDS with ring sideroblasts who are transfusion dependent and are refractory, intolerant, or ineligible to receive ESAs.
To further investigate luspatercept’s efficacy in a head-to-head comparison with an ESA in LR-MDS patients who are ESA naive, Dr. Garcia-Manero and colleagues conducted the phase 3 COMMANDS trial.
For the global, open-label study, patients with LR-MDS who were dependent on RBC transfusions and had no prior use of ESAs were randomized 1:1 to treatment either with subcutaneous luspatercept (starting dose, 1.0 mg/kg with titration up to 1.75 mg/kg; n = 178) once every 3 weeks or subcutaneous epoetin alfa (starting dose, 450 IU/kg with titration up to 1,050 IU/kg; n = 176) once every week, for a minimum of 24 weeks.
Patients in each arm were also able to receive best supportive care, including blood transfusions. Their baseline characteristics were similar in each arm.
For the primary endpoint, patients receiving luspatercept in the intent-to-treat population were nearly twice as likely as those treated with epoetin alfa to become independent of RBC transfusions, with a concurrent mean hemoglobin increase of 1.5 g/dL or more, for at least 12 weeks in the first 24 weeks on study, at a rate of 58.5% with luspatercept versus 31.2% with epoetin alfa (P < .0001).
In addition, patients treated with luspatercept had a longer median duration of transfusion independence, at 126.6 weeks versus 77 weeks in the epoetin alfa group (hazard ratio, 0.456).
Importantly, the statistically significant improvement with luspatercept was consistent among patients with ring sideroblasts (HR, 0.626) as well as without them (HR, 0.492). Dr. Garcia-Manero noted that about 70% of patients in the study had ring sideroblasts, consistent with their common occurrence in LR-MDS.
Luspatercept was also superior in secondary endpoints, including in achieving hematologic improvement, with an erythroid response of at least 8 weeks, per International Working Group 2006 criteria, which was achieved by 74.1% with luspatercept versus 51.3% with epoetin alfa (P < .0001).
The greater improvement with luspatercept was also observed in other subgroups, including based on baseline serum erythropoietin or levels of transfusion dependence, as well as SF381 mutation status.
In terms of safety, treatment emergent adverse events (TEAEs) of any grade were reported among 92.1% of luspatercept and 85.2% of epoetin alfa patients. Longer-term posttreatment safety analyses showed no significant differences between the groups in terms of progression to high-risk MDS, in five (2.8%) with luspatercept and seven(4.0%) epoetin alfa, and progression to acute myeloid leukemia, occurring in four (2.2%) luspatercept and five (2.8%) epoetin alfa patients.
Overall rates of death between the groups were also similar during the treatment and posttreatment periods (32 [18.0%] luspatercept; 32 [18.2%] epoetin alfa patients).
“The toxicity profile was consistent with previous clinical experience,” Dr. Garcia-Manero said.
Dr. Garcia-Manero underscored that “the results of the COMMANDS trial are very important.”
“ESAs are really not optimal agents [for LR-MDS], and these results indicate that luspatercept almost doubles response rates in this patient population, therefore becoming potentially the standard of care for patients with transfusion-dependent LR-MDS who have not received prior ESA treatment,” he said.
Further commenting, Dr. Artz added that the effects in patient subgroups will be of great interest as further data on luspatercept emerges.
“Of highest interest will be the differential responses among patients with and without ring sideroblasts, as well as by SF3B1 mutational status,” he said. Furthermore, “patient-centric data emerge as even more relevant when considering the quantitatively higher rates of treatment-emergent adverse effects in the luspatercept arm.”
“We need to understand how to best sequence anemia therapies in low-risk MDS when we have two active agents, or even if [there is] a role for combined ESA/luspatercept therapy,” he noted.
“The results are exciting, but we need the final data including relevant subsets before declaring luspatercept the winner,” Dr. Artz concluded.
The study was sponsored by Celgene/Bristol-Myers Squibb. Dr. Garcia-Manero reported relationships with Abbvie, Acceleron Pharma, Aprea Therapeutics, Astex Pharmaceuticals, Bristol-Myers Squibb, Genentech, Gilead Sciences, and Novartis. Dr. Artz disclosed previous consulting relationships with Abbvie and Magenta Therapeutics.
“Luspatercept is the first and only therapy to demonstrate superiority in a head-to-head study against ESAs in [transfusion-dependent] LR-MDS,” first author Guillermo Garcia-Manero, MD, chief of the MDS section, department of leukemia, at the University of Texas MD Anderson Cancer Center, Houston, said in a premeeting press briefing in advance of the annual meeting of the American Society of Clinical Oncology.
“It should be considered a paradigm shift in the treatment of LR-MDS–associated anemia,” Dr. Garcia-Manero said.
Commenting on the study, Andrew Artz, MD, a professor at the Hematologic Malignancies Research Institute, City of Hope National Medical Center, Duarte, Calif., agreed that the results could be practice changing.
“We biologically expected luspatercept to best ESA [in ring sideroblast transfusion–dependent MDS], based on luspatercept often rescuing ESA failures in this setting,” Dr. Artz said in an interview.
“The results have the potential to change initial therapy for patients with low-risk red blood cell transfusion-dependent MDS,” he said.
In LR-MDS, which encompasses a variety of bone marrow disorders, chronic anemia is very common, and patients, who are typically elderly, can become burdened by developing dependencies on RBC transfusions.
Transfusion dependency, in addition to creating a host of challenges, can increase the risk of death by as much as 50%, compared with patients who are not transfusion dependent, Dr. Garcia-Manero noted.
While ESAs such as epoetin alfa are the first-line treatment for LR-MDS, patients who are dependent on transfusions are less likely to respond to the agents, hence “there is an unmet need for effective and durable options other than ESAs for treating anemia in patients with LR-MDS,” Dr. Garcia-Manero said.
Luspatercept, a first-in-class monoclonal antibody, has a mechanism of action that is distinct from ESAs, modulating the transforming growth factor–beta pathway and increasing erythrocytosis.
In the previous phase 3 MEDALIST trial, the drug was shown to have efficacy over placebo in reducing the severity of anemia in LR-MDS. In 2020, in what was deemed the first advance in MDS treatment in more than a decade, those results led to approval by the Food and Drug Administration for patients with LR-MDS with ring sideroblasts who are transfusion dependent and are refractory, intolerant, or ineligible to receive ESAs.
To further investigate luspatercept’s efficacy in a head-to-head comparison with an ESA in LR-MDS patients who are ESA naive, Dr. Garcia-Manero and colleagues conducted the phase 3 COMMANDS trial.
For the global, open-label study, patients with LR-MDS who were dependent on RBC transfusions and had no prior use of ESAs were randomized 1:1 to treatment either with subcutaneous luspatercept (starting dose, 1.0 mg/kg with titration up to 1.75 mg/kg; n = 178) once every 3 weeks or subcutaneous epoetin alfa (starting dose, 450 IU/kg with titration up to 1,050 IU/kg; n = 176) once every week, for a minimum of 24 weeks.
Patients in each arm were also able to receive best supportive care, including blood transfusions. Their baseline characteristics were similar in each arm.
For the primary endpoint, patients receiving luspatercept in the intent-to-treat population were nearly twice as likely as those treated with epoetin alfa to become independent of RBC transfusions, with a concurrent mean hemoglobin increase of 1.5 g/dL or more, for at least 12 weeks in the first 24 weeks on study, at a rate of 58.5% with luspatercept versus 31.2% with epoetin alfa (P < .0001).
In addition, patients treated with luspatercept had a longer median duration of transfusion independence, at 126.6 weeks versus 77 weeks in the epoetin alfa group (hazard ratio, 0.456).
Importantly, the statistically significant improvement with luspatercept was consistent among patients with ring sideroblasts (HR, 0.626) as well as without them (HR, 0.492). Dr. Garcia-Manero noted that about 70% of patients in the study had ring sideroblasts, consistent with their common occurrence in LR-MDS.
Luspatercept was also superior in secondary endpoints, including in achieving hematologic improvement, with an erythroid response of at least 8 weeks, per International Working Group 2006 criteria, which was achieved by 74.1% with luspatercept versus 51.3% with epoetin alfa (P < .0001).
The greater improvement with luspatercept was also observed in other subgroups, including based on baseline serum erythropoietin or levels of transfusion dependence, as well as SF381 mutation status.
In terms of safety, treatment emergent adverse events (TEAEs) of any grade were reported among 92.1% of luspatercept and 85.2% of epoetin alfa patients. Longer-term posttreatment safety analyses showed no significant differences between the groups in terms of progression to high-risk MDS, in five (2.8%) with luspatercept and seven(4.0%) epoetin alfa, and progression to acute myeloid leukemia, occurring in four (2.2%) luspatercept and five (2.8%) epoetin alfa patients.
Overall rates of death between the groups were also similar during the treatment and posttreatment periods (32 [18.0%] luspatercept; 32 [18.2%] epoetin alfa patients).
“The toxicity profile was consistent with previous clinical experience,” Dr. Garcia-Manero said.
Dr. Garcia-Manero underscored that “the results of the COMMANDS trial are very important.”
“ESAs are really not optimal agents [for LR-MDS], and these results indicate that luspatercept almost doubles response rates in this patient population, therefore becoming potentially the standard of care for patients with transfusion-dependent LR-MDS who have not received prior ESA treatment,” he said.
Further commenting, Dr. Artz added that the effects in patient subgroups will be of great interest as further data on luspatercept emerges.
“Of highest interest will be the differential responses among patients with and without ring sideroblasts, as well as by SF3B1 mutational status,” he said. Furthermore, “patient-centric data emerge as even more relevant when considering the quantitatively higher rates of treatment-emergent adverse effects in the luspatercept arm.”
“We need to understand how to best sequence anemia therapies in low-risk MDS when we have two active agents, or even if [there is] a role for combined ESA/luspatercept therapy,” he noted.
“The results are exciting, but we need the final data including relevant subsets before declaring luspatercept the winner,” Dr. Artz concluded.
The study was sponsored by Celgene/Bristol-Myers Squibb. Dr. Garcia-Manero reported relationships with Abbvie, Acceleron Pharma, Aprea Therapeutics, Astex Pharmaceuticals, Bristol-Myers Squibb, Genentech, Gilead Sciences, and Novartis. Dr. Artz disclosed previous consulting relationships with Abbvie and Magenta Therapeutics.
“Luspatercept is the first and only therapy to demonstrate superiority in a head-to-head study against ESAs in [transfusion-dependent] LR-MDS,” first author Guillermo Garcia-Manero, MD, chief of the MDS section, department of leukemia, at the University of Texas MD Anderson Cancer Center, Houston, said in a premeeting press briefing in advance of the annual meeting of the American Society of Clinical Oncology.
“It should be considered a paradigm shift in the treatment of LR-MDS–associated anemia,” Dr. Garcia-Manero said.
Commenting on the study, Andrew Artz, MD, a professor at the Hematologic Malignancies Research Institute, City of Hope National Medical Center, Duarte, Calif., agreed that the results could be practice changing.
“We biologically expected luspatercept to best ESA [in ring sideroblast transfusion–dependent MDS], based on luspatercept often rescuing ESA failures in this setting,” Dr. Artz said in an interview.
“The results have the potential to change initial therapy for patients with low-risk red blood cell transfusion-dependent MDS,” he said.
In LR-MDS, which encompasses a variety of bone marrow disorders, chronic anemia is very common, and patients, who are typically elderly, can become burdened by developing dependencies on RBC transfusions.
Transfusion dependency, in addition to creating a host of challenges, can increase the risk of death by as much as 50%, compared with patients who are not transfusion dependent, Dr. Garcia-Manero noted.
While ESAs such as epoetin alfa are the first-line treatment for LR-MDS, patients who are dependent on transfusions are less likely to respond to the agents, hence “there is an unmet need for effective and durable options other than ESAs for treating anemia in patients with LR-MDS,” Dr. Garcia-Manero said.
Luspatercept, a first-in-class monoclonal antibody, has a mechanism of action that is distinct from ESAs, modulating the transforming growth factor–beta pathway and increasing erythrocytosis.
In the previous phase 3 MEDALIST trial, the drug was shown to have efficacy over placebo in reducing the severity of anemia in LR-MDS. In 2020, in what was deemed the first advance in MDS treatment in more than a decade, those results led to approval by the Food and Drug Administration for patients with LR-MDS with ring sideroblasts who are transfusion dependent and are refractory, intolerant, or ineligible to receive ESAs.
To further investigate luspatercept’s efficacy in a head-to-head comparison with an ESA in LR-MDS patients who are ESA naive, Dr. Garcia-Manero and colleagues conducted the phase 3 COMMANDS trial.
For the global, open-label study, patients with LR-MDS who were dependent on RBC transfusions and had no prior use of ESAs were randomized 1:1 to treatment either with subcutaneous luspatercept (starting dose, 1.0 mg/kg with titration up to 1.75 mg/kg; n = 178) once every 3 weeks or subcutaneous epoetin alfa (starting dose, 450 IU/kg with titration up to 1,050 IU/kg; n = 176) once every week, for a minimum of 24 weeks.
Patients in each arm were also able to receive best supportive care, including blood transfusions. Their baseline characteristics were similar in each arm.
For the primary endpoint, patients receiving luspatercept in the intent-to-treat population were nearly twice as likely as those treated with epoetin alfa to become independent of RBC transfusions, with a concurrent mean hemoglobin increase of 1.5 g/dL or more, for at least 12 weeks in the first 24 weeks on study, at a rate of 58.5% with luspatercept versus 31.2% with epoetin alfa (P < .0001).
In addition, patients treated with luspatercept had a longer median duration of transfusion independence, at 126.6 weeks versus 77 weeks in the epoetin alfa group (hazard ratio, 0.456).
Importantly, the statistically significant improvement with luspatercept was consistent among patients with ring sideroblasts (HR, 0.626) as well as without them (HR, 0.492). Dr. Garcia-Manero noted that about 70% of patients in the study had ring sideroblasts, consistent with their common occurrence in LR-MDS.
Luspatercept was also superior in secondary endpoints, including in achieving hematologic improvement, with an erythroid response of at least 8 weeks, per International Working Group 2006 criteria, which was achieved by 74.1% with luspatercept versus 51.3% with epoetin alfa (P < .0001).
The greater improvement with luspatercept was also observed in other subgroups, including based on baseline serum erythropoietin or levels of transfusion dependence, as well as SF381 mutation status.
In terms of safety, treatment emergent adverse events (TEAEs) of any grade were reported among 92.1% of luspatercept and 85.2% of epoetin alfa patients. Longer-term posttreatment safety analyses showed no significant differences between the groups in terms of progression to high-risk MDS, in five (2.8%) with luspatercept and seven(4.0%) epoetin alfa, and progression to acute myeloid leukemia, occurring in four (2.2%) luspatercept and five (2.8%) epoetin alfa patients.
Overall rates of death between the groups were also similar during the treatment and posttreatment periods (32 [18.0%] luspatercept; 32 [18.2%] epoetin alfa patients).
“The toxicity profile was consistent with previous clinical experience,” Dr. Garcia-Manero said.
Dr. Garcia-Manero underscored that “the results of the COMMANDS trial are very important.”
“ESAs are really not optimal agents [for LR-MDS], and these results indicate that luspatercept almost doubles response rates in this patient population, therefore becoming potentially the standard of care for patients with transfusion-dependent LR-MDS who have not received prior ESA treatment,” he said.
Further commenting, Dr. Artz added that the effects in patient subgroups will be of great interest as further data on luspatercept emerges.
“Of highest interest will be the differential responses among patients with and without ring sideroblasts, as well as by SF3B1 mutational status,” he said. Furthermore, “patient-centric data emerge as even more relevant when considering the quantitatively higher rates of treatment-emergent adverse effects in the luspatercept arm.”
“We need to understand how to best sequence anemia therapies in low-risk MDS when we have two active agents, or even if [there is] a role for combined ESA/luspatercept therapy,” he noted.
“The results are exciting, but we need the final data including relevant subsets before declaring luspatercept the winner,” Dr. Artz concluded.
The study was sponsored by Celgene/Bristol-Myers Squibb. Dr. Garcia-Manero reported relationships with Abbvie, Acceleron Pharma, Aprea Therapeutics, Astex Pharmaceuticals, Bristol-Myers Squibb, Genentech, Gilead Sciences, and Novartis. Dr. Artz disclosed previous consulting relationships with Abbvie and Magenta Therapeutics.
FROM ASCO 2023







