User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Regular vitamin D supplements may lower melanoma risk
. They also found a trend for benefit with occasional use.
The study, published in Melanoma Research, involved almost 500 individuals attending a dermatology clinic who reported on their use of vitamin D supplements.
Regular users had a significant 55% reduction in the odds of having a past or present melanoma diagnosis, while occasional use was associated with a nonsignificant 46% reduction. The reduction was similar for all skin cancer types.
However, senior author Ilkka T. Harvima, MD, PhD, department of dermatology, University of Eastern Finland and Kuopio (Finland) University Hospital, warned there are limitations to the study.
Despite adjustment for several possible confounding factors, “it is still possible that some other, yet unidentified or untested, factors can still confound the present result,” he said.
Consequently, “the causal link between vitamin D and melanoma cannot be confirmed by the present results,” Dr. Harvima said in a statement.
Even if the link were to be proven, “the question about the optimal dose of oral vitamin D in order to for it to have beneficial effects remains to be answered,” he said.
“Until we know more, national intake recommendations should be followed.”
The incidence of cutaneous malignant melanoma and other skin cancers has been increasing steadily in Western populations, particularly in immunosuppressed individuals, the authors pointed out, and they attributed the rise to an increased exposure to ultraviolet radiation.
While ultraviolet radiation exposure is a well-known risk factor, “the other side of the coin is that public sun protection campaigns have led to alerts that insufficient sun exposure is a significant public health problem, resulting in insufficient vitamin D status.”
For their study, the team reviewed the records of 498 patients aged 21-79 years at a dermatology outpatient clinic who were deemed by an experienced dermatologist to be at risk of any type of skin cancer.
Among these patients, 295 individuals had a history of past or present cutaneous malignancy, with 100 diagnosed with melanoma, 213 with basal cell carcinoma, and 41 with squamous cell carcinoma. A further 70 subjects had cancer elsewhere, including breast, prostate, kidney, bladder, intestine, and blood cancers.
A subgroup of 96 patients were immunocompromised and were considered separately.
The 402 remaining patients were categorized, based on their self-reported use of oral vitamin D preparations, as nonusers (n = 99), occasional users (n = 126), and regular users (n = 177).
Regular use of vitamin D was associated with being more educated (P = .032), less frequent outdoor working (P = .003), lower tobacco pack years (P = .001), and more frequent solarium exposure (P = .002).
There was no significant association between vitamin D use and photoaging, actinic keratoses, nevi, basal or squamous cell carcinoma, body mass index, or self-estimated lifetime exposure to sunlight or sunburns.
However, there were significant associations between regular use of vitamin D and a lower incidence of melanoma and other cancer types.
There were significantly fewer individuals in the regular vitamin D use group with a past or present history of melanoma when compared with the nonuse group, at 18.1% vs. 32.3% (P = .021), or any type of skin cancer, at 62.1% vs. 74.7% (P = .027).
Multivariate logistic regression analysis revealed that regular vitamin D use was significantly associated with a reduced melanoma risk, at an odds ratio vs. nonuse of 0.447 (P = .016).
Occasional use was associated with a reduced, albeit nonsignificant, risk, with an odds ratio versus nonuse of 0.540 (P = .08).
For any type of skin cancers, regular vitamin D use was associated with an odds ratio vs. nonuse of 0.478 (P = .032), while that for occasional vitamin D use was 0.543 (P = .061).
“Somewhat similar” results were obtained when the investigators looked at the subgroup of immunocompromised individuals, although they note that “the number of subjects was low.”
The study was supported by the Cancer Center of Eastern Finland of the University of Eastern Finland, the Finnish Cancer Research Foundation, and the VTR-funding of Kuopio University Hospital. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
. They also found a trend for benefit with occasional use.
The study, published in Melanoma Research, involved almost 500 individuals attending a dermatology clinic who reported on their use of vitamin D supplements.
Regular users had a significant 55% reduction in the odds of having a past or present melanoma diagnosis, while occasional use was associated with a nonsignificant 46% reduction. The reduction was similar for all skin cancer types.
However, senior author Ilkka T. Harvima, MD, PhD, department of dermatology, University of Eastern Finland and Kuopio (Finland) University Hospital, warned there are limitations to the study.
Despite adjustment for several possible confounding factors, “it is still possible that some other, yet unidentified or untested, factors can still confound the present result,” he said.
Consequently, “the causal link between vitamin D and melanoma cannot be confirmed by the present results,” Dr. Harvima said in a statement.
Even if the link were to be proven, “the question about the optimal dose of oral vitamin D in order to for it to have beneficial effects remains to be answered,” he said.
“Until we know more, national intake recommendations should be followed.”
The incidence of cutaneous malignant melanoma and other skin cancers has been increasing steadily in Western populations, particularly in immunosuppressed individuals, the authors pointed out, and they attributed the rise to an increased exposure to ultraviolet radiation.
While ultraviolet radiation exposure is a well-known risk factor, “the other side of the coin is that public sun protection campaigns have led to alerts that insufficient sun exposure is a significant public health problem, resulting in insufficient vitamin D status.”
For their study, the team reviewed the records of 498 patients aged 21-79 years at a dermatology outpatient clinic who were deemed by an experienced dermatologist to be at risk of any type of skin cancer.
Among these patients, 295 individuals had a history of past or present cutaneous malignancy, with 100 diagnosed with melanoma, 213 with basal cell carcinoma, and 41 with squamous cell carcinoma. A further 70 subjects had cancer elsewhere, including breast, prostate, kidney, bladder, intestine, and blood cancers.
A subgroup of 96 patients were immunocompromised and were considered separately.
The 402 remaining patients were categorized, based on their self-reported use of oral vitamin D preparations, as nonusers (n = 99), occasional users (n = 126), and regular users (n = 177).
Regular use of vitamin D was associated with being more educated (P = .032), less frequent outdoor working (P = .003), lower tobacco pack years (P = .001), and more frequent solarium exposure (P = .002).
There was no significant association between vitamin D use and photoaging, actinic keratoses, nevi, basal or squamous cell carcinoma, body mass index, or self-estimated lifetime exposure to sunlight or sunburns.
However, there were significant associations between regular use of vitamin D and a lower incidence of melanoma and other cancer types.
There were significantly fewer individuals in the regular vitamin D use group with a past or present history of melanoma when compared with the nonuse group, at 18.1% vs. 32.3% (P = .021), or any type of skin cancer, at 62.1% vs. 74.7% (P = .027).
Multivariate logistic regression analysis revealed that regular vitamin D use was significantly associated with a reduced melanoma risk, at an odds ratio vs. nonuse of 0.447 (P = .016).
Occasional use was associated with a reduced, albeit nonsignificant, risk, with an odds ratio versus nonuse of 0.540 (P = .08).
For any type of skin cancers, regular vitamin D use was associated with an odds ratio vs. nonuse of 0.478 (P = .032), while that for occasional vitamin D use was 0.543 (P = .061).
“Somewhat similar” results were obtained when the investigators looked at the subgroup of immunocompromised individuals, although they note that “the number of subjects was low.”
The study was supported by the Cancer Center of Eastern Finland of the University of Eastern Finland, the Finnish Cancer Research Foundation, and the VTR-funding of Kuopio University Hospital. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
. They also found a trend for benefit with occasional use.
The study, published in Melanoma Research, involved almost 500 individuals attending a dermatology clinic who reported on their use of vitamin D supplements.
Regular users had a significant 55% reduction in the odds of having a past or present melanoma diagnosis, while occasional use was associated with a nonsignificant 46% reduction. The reduction was similar for all skin cancer types.
However, senior author Ilkka T. Harvima, MD, PhD, department of dermatology, University of Eastern Finland and Kuopio (Finland) University Hospital, warned there are limitations to the study.
Despite adjustment for several possible confounding factors, “it is still possible that some other, yet unidentified or untested, factors can still confound the present result,” he said.
Consequently, “the causal link between vitamin D and melanoma cannot be confirmed by the present results,” Dr. Harvima said in a statement.
Even if the link were to be proven, “the question about the optimal dose of oral vitamin D in order to for it to have beneficial effects remains to be answered,” he said.
“Until we know more, national intake recommendations should be followed.”
The incidence of cutaneous malignant melanoma and other skin cancers has been increasing steadily in Western populations, particularly in immunosuppressed individuals, the authors pointed out, and they attributed the rise to an increased exposure to ultraviolet radiation.
While ultraviolet radiation exposure is a well-known risk factor, “the other side of the coin is that public sun protection campaigns have led to alerts that insufficient sun exposure is a significant public health problem, resulting in insufficient vitamin D status.”
For their study, the team reviewed the records of 498 patients aged 21-79 years at a dermatology outpatient clinic who were deemed by an experienced dermatologist to be at risk of any type of skin cancer.
Among these patients, 295 individuals had a history of past or present cutaneous malignancy, with 100 diagnosed with melanoma, 213 with basal cell carcinoma, and 41 with squamous cell carcinoma. A further 70 subjects had cancer elsewhere, including breast, prostate, kidney, bladder, intestine, and blood cancers.
A subgroup of 96 patients were immunocompromised and were considered separately.
The 402 remaining patients were categorized, based on their self-reported use of oral vitamin D preparations, as nonusers (n = 99), occasional users (n = 126), and regular users (n = 177).
Regular use of vitamin D was associated with being more educated (P = .032), less frequent outdoor working (P = .003), lower tobacco pack years (P = .001), and more frequent solarium exposure (P = .002).
There was no significant association between vitamin D use and photoaging, actinic keratoses, nevi, basal or squamous cell carcinoma, body mass index, or self-estimated lifetime exposure to sunlight or sunburns.
However, there were significant associations between regular use of vitamin D and a lower incidence of melanoma and other cancer types.
There were significantly fewer individuals in the regular vitamin D use group with a past or present history of melanoma when compared with the nonuse group, at 18.1% vs. 32.3% (P = .021), or any type of skin cancer, at 62.1% vs. 74.7% (P = .027).
Multivariate logistic regression analysis revealed that regular vitamin D use was significantly associated with a reduced melanoma risk, at an odds ratio vs. nonuse of 0.447 (P = .016).
Occasional use was associated with a reduced, albeit nonsignificant, risk, with an odds ratio versus nonuse of 0.540 (P = .08).
For any type of skin cancers, regular vitamin D use was associated with an odds ratio vs. nonuse of 0.478 (P = .032), while that for occasional vitamin D use was 0.543 (P = .061).
“Somewhat similar” results were obtained when the investigators looked at the subgroup of immunocompromised individuals, although they note that “the number of subjects was low.”
The study was supported by the Cancer Center of Eastern Finland of the University of Eastern Finland, the Finnish Cancer Research Foundation, and the VTR-funding of Kuopio University Hospital. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM MELANOMA RESEARCH
Early retirement and the terrible, horrible, no good, very bad cognitive decline
The ‘scheme’ in the name should have been a clue
Retirement. The shiny reward to a lifetime’s worth of working and saving. We’re all literally working to get there, some of us more to get there early, but current research reveals that early retirement isn’t the relaxing finish line we dream about, cognitively speaking.
Researchers at Binghamton (N.Y.) University set out to examine just how retirement plans affect cognitive performance. They started off with China’s New Rural Pension Scheme (scheme probably has a less negative connotation in Chinese), a plan that financially aids the growing rural retirement-age population in the country. Then they looked at data from the Chinese Health and Retirement Longitudinal Survey, which tests cognition with a focus on episodic memory and parts of intact mental status.
What they found was the opposite of what you would expect out of retirees with nothing but time on their hands.
The pension program, which had been in place for almost a decade, led to delayed recall, especially among women, supporting “the mental retirement hypothesis that decreased mental activity results in worsening cognitive skills,” the investigators said in a written statement.
There also was a drop in social engagement, with lower rates of volunteering and social interaction than people who didn’t receive the pension. Some behaviors, like regular alcohol consumption, did improve over the previous year, as did total health in general, but “the adverse effects of early retirement on mental and social engagement significantly outweigh the program’s protective effect on various health behaviors,” Plamen Nikolov, PhD, said about his research.
So if you’re looking to retire early, don’t skimp on the crosswords and the bingo nights. Stay busy in a good way. Your brain will thank you.
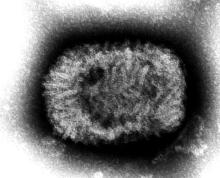
Indiana Jones and the First Smallpox Ancestor
Smallpox was, not that long ago, one of the most devastating diseases known to humanity, killing 300 million people in the 20th century alone. Eradicating it has to be one of medicine’s crowning achievements. Now it can only be found in museums, which is where it belongs.
Here’s the thing with smallpox though: For all it did to us, we know frustratingly little about where it came from. Until very recently, the best available genetic evidence placed its emergence in the 17th century, which clashes with historical data. You know what that means, right? It’s time to dig out the fedora and whip, cue the music, and dig into a recently published study spanning continents in search of the mythical smallpox origin story.
We pick up in 2020, when genetic evidence definitively showed smallpox in a Viking burial site, moving the disease’s emergence a thousand years earlier. Which is all well and good, but there’s solid visual evidence that Egyptian pharaohs were dying of smallpox, as their bodies show the signature scarring. Historians were pretty sure smallpox went back about 4,000 years, but there was no genetic material to prove it.
Since there aren’t any 4,000-year-old smallpox germs laying around, the researchers chose to attack the problem another way – by burning down a Venetian catacomb, er, conducting a analysis of historical smallpox genetics to find the virus’s origin. By analyzing the genomes of various strains at different periods of time, they were able to determine that the variola virus had a definitive common ancestor. Some of the genetic components in the Viking-age sample, for example, persisted until the 18th century.
Armed with this information, the scientists determined that the first smallpox ancestor emerged about 3,800 years ago. That’s very close to the historians’ estimate for the disease’s emergence. Proof at last of smallpox’s truly ancient origin. One might even say the researchers chose wisely.
The only hall of fame that really matters
LOTME loves the holiday season – the food, the gifts, the radio stations that play nothing but Christmas music – but for us the most wonderful time of the year comes just a bit later. No, it’s not our annual Golden Globes slap bet. Nope, not even the “excitement” of the College Football Playoff National Championship. It’s time for the National Inventors Hall of Fame to announce its latest inductees, and we could hardly sleep last night after putting cookies out for Thomas Edison. Fasten your seatbelts!
- Robert G. Bryant is a NASA chemist who developed Langley Research Center-Soluble Imide (yes, that’s the actual name) a polymer used as an insulation material for leads in implantable cardiac resynchronization therapy devices.
- Rory Cooper is a biomedical engineer who was paralyzed in a bicycle accident. His work has improved manual and electric wheelchairs and advanced the health, mobility, and social inclusion of people with disabilities and older adults. He is also the first NIHF inductee named Rory.
- Katalin Karikó, a biochemist, and Drew Weissman, an immunologist, “discovered how to enable messenger ribonucleic acid (mRNA) to enter cells without triggering the body’s immune system,” NIHF said, and that laid the foundation for the mRNA COVID-19 vaccines developed by Pfizer-BioNTech and Moderna. That, of course, led to the antivax movement, which has provided so much LOTME fodder over the years.
- Angela Hartley Brodie was a biochemist who discovered and developed a class of drugs called aromatase inhibitors, which can stop the production of hormones that fuel cancer cell growth and are used to treat breast cancer in 500,000 women worldwide each year.
We can’t mention all of the inductees for 2023 (our editor made that very clear), but we would like to offer a special shout-out to brothers Cyril (the first Cyril in the NIHF, by the way) and Louis Keller, who invented the world’s first compact loader, which eventually became the Bobcat skid-steer loader. Not really medical, you’re probably thinking, but we’re sure that someone, somewhere, at some time, used one to build a hospital, landscape a hospital, or clean up after the demolition of a hospital.
The ‘scheme’ in the name should have been a clue
Retirement. The shiny reward to a lifetime’s worth of working and saving. We’re all literally working to get there, some of us more to get there early, but current research reveals that early retirement isn’t the relaxing finish line we dream about, cognitively speaking.
Researchers at Binghamton (N.Y.) University set out to examine just how retirement plans affect cognitive performance. They started off with China’s New Rural Pension Scheme (scheme probably has a less negative connotation in Chinese), a plan that financially aids the growing rural retirement-age population in the country. Then they looked at data from the Chinese Health and Retirement Longitudinal Survey, which tests cognition with a focus on episodic memory and parts of intact mental status.
What they found was the opposite of what you would expect out of retirees with nothing but time on their hands.
The pension program, which had been in place for almost a decade, led to delayed recall, especially among women, supporting “the mental retirement hypothesis that decreased mental activity results in worsening cognitive skills,” the investigators said in a written statement.
There also was a drop in social engagement, with lower rates of volunteering and social interaction than people who didn’t receive the pension. Some behaviors, like regular alcohol consumption, did improve over the previous year, as did total health in general, but “the adverse effects of early retirement on mental and social engagement significantly outweigh the program’s protective effect on various health behaviors,” Plamen Nikolov, PhD, said about his research.
So if you’re looking to retire early, don’t skimp on the crosswords and the bingo nights. Stay busy in a good way. Your brain will thank you.
Indiana Jones and the First Smallpox Ancestor
Smallpox was, not that long ago, one of the most devastating diseases known to humanity, killing 300 million people in the 20th century alone. Eradicating it has to be one of medicine’s crowning achievements. Now it can only be found in museums, which is where it belongs.
Here’s the thing with smallpox though: For all it did to us, we know frustratingly little about where it came from. Until very recently, the best available genetic evidence placed its emergence in the 17th century, which clashes with historical data. You know what that means, right? It’s time to dig out the fedora and whip, cue the music, and dig into a recently published study spanning continents in search of the mythical smallpox origin story.
We pick up in 2020, when genetic evidence definitively showed smallpox in a Viking burial site, moving the disease’s emergence a thousand years earlier. Which is all well and good, but there’s solid visual evidence that Egyptian pharaohs were dying of smallpox, as their bodies show the signature scarring. Historians were pretty sure smallpox went back about 4,000 years, but there was no genetic material to prove it.
Since there aren’t any 4,000-year-old smallpox germs laying around, the researchers chose to attack the problem another way – by burning down a Venetian catacomb, er, conducting a analysis of historical smallpox genetics to find the virus’s origin. By analyzing the genomes of various strains at different periods of time, they were able to determine that the variola virus had a definitive common ancestor. Some of the genetic components in the Viking-age sample, for example, persisted until the 18th century.
Armed with this information, the scientists determined that the first smallpox ancestor emerged about 3,800 years ago. That’s very close to the historians’ estimate for the disease’s emergence. Proof at last of smallpox’s truly ancient origin. One might even say the researchers chose wisely.
The only hall of fame that really matters
LOTME loves the holiday season – the food, the gifts, the radio stations that play nothing but Christmas music – but for us the most wonderful time of the year comes just a bit later. No, it’s not our annual Golden Globes slap bet. Nope, not even the “excitement” of the College Football Playoff National Championship. It’s time for the National Inventors Hall of Fame to announce its latest inductees, and we could hardly sleep last night after putting cookies out for Thomas Edison. Fasten your seatbelts!
- Robert G. Bryant is a NASA chemist who developed Langley Research Center-Soluble Imide (yes, that’s the actual name) a polymer used as an insulation material for leads in implantable cardiac resynchronization therapy devices.
- Rory Cooper is a biomedical engineer who was paralyzed in a bicycle accident. His work has improved manual and electric wheelchairs and advanced the health, mobility, and social inclusion of people with disabilities and older adults. He is also the first NIHF inductee named Rory.
- Katalin Karikó, a biochemist, and Drew Weissman, an immunologist, “discovered how to enable messenger ribonucleic acid (mRNA) to enter cells without triggering the body’s immune system,” NIHF said, and that laid the foundation for the mRNA COVID-19 vaccines developed by Pfizer-BioNTech and Moderna. That, of course, led to the antivax movement, which has provided so much LOTME fodder over the years.
- Angela Hartley Brodie was a biochemist who discovered and developed a class of drugs called aromatase inhibitors, which can stop the production of hormones that fuel cancer cell growth and are used to treat breast cancer in 500,000 women worldwide each year.
We can’t mention all of the inductees for 2023 (our editor made that very clear), but we would like to offer a special shout-out to brothers Cyril (the first Cyril in the NIHF, by the way) and Louis Keller, who invented the world’s first compact loader, which eventually became the Bobcat skid-steer loader. Not really medical, you’re probably thinking, but we’re sure that someone, somewhere, at some time, used one to build a hospital, landscape a hospital, or clean up after the demolition of a hospital.
The ‘scheme’ in the name should have been a clue
Retirement. The shiny reward to a lifetime’s worth of working and saving. We’re all literally working to get there, some of us more to get there early, but current research reveals that early retirement isn’t the relaxing finish line we dream about, cognitively speaking.
Researchers at Binghamton (N.Y.) University set out to examine just how retirement plans affect cognitive performance. They started off with China’s New Rural Pension Scheme (scheme probably has a less negative connotation in Chinese), a plan that financially aids the growing rural retirement-age population in the country. Then they looked at data from the Chinese Health and Retirement Longitudinal Survey, which tests cognition with a focus on episodic memory and parts of intact mental status.
What they found was the opposite of what you would expect out of retirees with nothing but time on their hands.
The pension program, which had been in place for almost a decade, led to delayed recall, especially among women, supporting “the mental retirement hypothesis that decreased mental activity results in worsening cognitive skills,” the investigators said in a written statement.
There also was a drop in social engagement, with lower rates of volunteering and social interaction than people who didn’t receive the pension. Some behaviors, like regular alcohol consumption, did improve over the previous year, as did total health in general, but “the adverse effects of early retirement on mental and social engagement significantly outweigh the program’s protective effect on various health behaviors,” Plamen Nikolov, PhD, said about his research.
So if you’re looking to retire early, don’t skimp on the crosswords and the bingo nights. Stay busy in a good way. Your brain will thank you.
Indiana Jones and the First Smallpox Ancestor
Smallpox was, not that long ago, one of the most devastating diseases known to humanity, killing 300 million people in the 20th century alone. Eradicating it has to be one of medicine’s crowning achievements. Now it can only be found in museums, which is where it belongs.
Here’s the thing with smallpox though: For all it did to us, we know frustratingly little about where it came from. Until very recently, the best available genetic evidence placed its emergence in the 17th century, which clashes with historical data. You know what that means, right? It’s time to dig out the fedora and whip, cue the music, and dig into a recently published study spanning continents in search of the mythical smallpox origin story.
We pick up in 2020, when genetic evidence definitively showed smallpox in a Viking burial site, moving the disease’s emergence a thousand years earlier. Which is all well and good, but there’s solid visual evidence that Egyptian pharaohs were dying of smallpox, as their bodies show the signature scarring. Historians were pretty sure smallpox went back about 4,000 years, but there was no genetic material to prove it.
Since there aren’t any 4,000-year-old smallpox germs laying around, the researchers chose to attack the problem another way – by burning down a Venetian catacomb, er, conducting a analysis of historical smallpox genetics to find the virus’s origin. By analyzing the genomes of various strains at different periods of time, they were able to determine that the variola virus had a definitive common ancestor. Some of the genetic components in the Viking-age sample, for example, persisted until the 18th century.
Armed with this information, the scientists determined that the first smallpox ancestor emerged about 3,800 years ago. That’s very close to the historians’ estimate for the disease’s emergence. Proof at last of smallpox’s truly ancient origin. One might even say the researchers chose wisely.
The only hall of fame that really matters
LOTME loves the holiday season – the food, the gifts, the radio stations that play nothing but Christmas music – but for us the most wonderful time of the year comes just a bit later. No, it’s not our annual Golden Globes slap bet. Nope, not even the “excitement” of the College Football Playoff National Championship. It’s time for the National Inventors Hall of Fame to announce its latest inductees, and we could hardly sleep last night after putting cookies out for Thomas Edison. Fasten your seatbelts!
- Robert G. Bryant is a NASA chemist who developed Langley Research Center-Soluble Imide (yes, that’s the actual name) a polymer used as an insulation material for leads in implantable cardiac resynchronization therapy devices.
- Rory Cooper is a biomedical engineer who was paralyzed in a bicycle accident. His work has improved manual and electric wheelchairs and advanced the health, mobility, and social inclusion of people with disabilities and older adults. He is also the first NIHF inductee named Rory.
- Katalin Karikó, a biochemist, and Drew Weissman, an immunologist, “discovered how to enable messenger ribonucleic acid (mRNA) to enter cells without triggering the body’s immune system,” NIHF said, and that laid the foundation for the mRNA COVID-19 vaccines developed by Pfizer-BioNTech and Moderna. That, of course, led to the antivax movement, which has provided so much LOTME fodder over the years.
- Angela Hartley Brodie was a biochemist who discovered and developed a class of drugs called aromatase inhibitors, which can stop the production of hormones that fuel cancer cell growth and are used to treat breast cancer in 500,000 women worldwide each year.
We can’t mention all of the inductees for 2023 (our editor made that very clear), but we would like to offer a special shout-out to brothers Cyril (the first Cyril in the NIHF, by the way) and Louis Keller, who invented the world’s first compact loader, which eventually became the Bobcat skid-steer loader. Not really medical, you’re probably thinking, but we’re sure that someone, somewhere, at some time, used one to build a hospital, landscape a hospital, or clean up after the demolition of a hospital.
Abnormal bleeding common among youth with joint hypermobility
A small cohort study of pediatric rheumatology patients with generalized joint hypermobility (GJH) who presented to a specialized rheumatology* clinic suggests that many such patients have abnormal bleeding symptoms, in comparison with health control patients.
The study of 81 patients with GJH found that about three quarters had significantly elevated median bleeding scores, but only 12% had been assessed by hematology for bleeding.
“We propose that screening for bleeding symptoms should be integrated into the routine care for all patients with GJH, with hematology referrals for patients with increased bleeding concerns,” wrote a research team led by Nicole E. Kendel, MD, a pediatric hematologist-oncologist at Akron Children’s Hospital in Ohio, in a study published online in Arthritis Care and Research.
“Further studies are needed to understand the mechanism of bleeding, evaluate comorbidities associated with these bleeding symptoms, and potentially allow for tailored pharmacologic therapy,” the authors stated.
Background
Dr. Kendel’s team had reported moderate menstruation-associated limitations in school, social, and physical activities among female adolescents with GJH. “This cohort also experienced nonreproductive bleeding symptoms and demonstrated minimal hemostatic laboratory abnormalities, indicating that this population may be underdiagnosed and subsequently poorly managed,” she said in an interview. “As excessive bleeding symptoms could have a significant impact on overall health and quality of life, we thought it was important to define the incidence and natural course of bleeding symptoms in a more generalized subset of this population.”
Although the investigators hypothesized that there would be a statistically significant increase in bleeding scores, “we were still impressed by the frequency of abnormal scores, particularly when looking at the low percentage of patients [12%] who had previously been referred to hematology,” she said.
Study results
The median age of the study cohort was 13 years (interquartile range, 10-16 years), and 72.8% were female. The mean Beighton score, which measures joint flexibility, was 6.2 (range, 4-9). All participants were seen by rheumatologists and were diagnosed for conditions on the hypermobility spectrum. Those conditions ranged from GJH to hypermobile Ehlers-Danlos syndrome (hEDS).
Abnormal bleeding, as measured by the International Society on Thrombosis and Haemostasis Bleeding Assessment Tool, was found in 75% (95% confidence interval [CI], 64%-84%). Overall mean and median bleeding scores were 5.2 and 4, respectively; scores ranged from 0 to 16. Abnormal scores of ≥ 3 were observed for patients < 8 years of age, ≥ 4 for men ≥ 18 years of age, and ≥ 6 for women ≥ 18 years of age. These measures were significantly elevated compared with those reported for historical healthy pediatric control persons (P < .001).
The most common hemorrhagic symptom was oral bleeding (74.1%) that occurred with tooth brushing, flossing, tooth loss, or eruption. Others reported easy bruising (59.3%) and bleeding from minor wounds (42%). In terms of procedures, tooth extraction requiring additional packing was reported by 25.9%, and 22.2% reported significant bleeding after otolaryngologic procedures, such as tonsillectomy/adenoidectomy, septoplasty, and nasal turbinate reduction.
Prolonged or heavy menstrual periods were reported by 37.3% of female patients.
Bleeding scores did not differ by biological sex or NSAID use, nor did any correlation emerge between patients’ bleeding and Beighton scores. However, there was a positive correlation with increasing age, a phenomenon observed with other bleeding disorders and in the healthy population, the authors noted.
Of the 10 study participants who had previously undergone hematologic assessment, one had been diagnosed with acquired, heart disease–related von Willebrand disease, and another with mild bleeding disorder.
Severe connective tissue disorders are associated with increased bleeding symptoms in the adult population, Dr. Kendel said, but few studies have assessed bleeding across the GJH spectrum, particularly in children.
Bleeding is thought to be due to modifications of collagen in the blood vessels. “These modifications create mechanical weakness of the vessel wall, as well as defective subendothelial connective tissue supporting those blood vessels,” Dr. Kendel explained. She noted that altered collagen creates defective interactions between collagen and other coagulation factors.
“Even in the presence of a normal laboratory evaluation, GJH can lead to symptoms consistent with a mild bleeding disorder,” she continued. “These symptoms are both preventable and treatable. I’m hopeful more centers will start routinely evaluating for increased bleeding symptoms, with referral to hematology for those with increased bleeding concerns.”
Commenting on the study’s recommendation, Beth S. Gottlieb, MD, chief of the division of pediatric rheumatology at Northwell Health in New Hyde Park, N.Y., who was not involved in the investigation, said a brief questionnaire on bleeding risk is a reasonable addition to a rheumatology office visit.
“Joint hypermobility is very common, but not all affected children meet the criteria for the hypermobile form of hEDS,” she told this news organization. “Screening for bleeding tendency is often done as routine medical history questions. Once a child is identified as hypermobile, these screening questions are usually asked, but utilizing one of the formal bleeding risk questionnaires is not currently routine.”
According to Dr. Gottlieb, it remains unclear whether screening would have a significant impact on children who have been diagnosed with hypermobility. “Most of these children are young and may not yet have a significant history for bleeding tendency,” she said. “Education of families is always important, and it will be essential to educate without adding unnecessary stress. Screening guidelines may be an important tool that is easy to incorporate into routine clinical practice.”
Limitation
The study was limited by selection bias, as patients had all been referred to a specialized rheumatology clinic.
The study was supported by the Clinical and Translational Intramural Funding Program of the Abigail Wexner Research Institute. The authors and Dr. Gottlieb have disclosed no relevant financial relationships.
*Correction, 1/11/2023: An earlier version of this story misstated the type of specialty clinic where patients were first seen.
A version of this article first appeared on Medscape.com.
A small cohort study of pediatric rheumatology patients with generalized joint hypermobility (GJH) who presented to a specialized rheumatology* clinic suggests that many such patients have abnormal bleeding symptoms, in comparison with health control patients.
The study of 81 patients with GJH found that about three quarters had significantly elevated median bleeding scores, but only 12% had been assessed by hematology for bleeding.
“We propose that screening for bleeding symptoms should be integrated into the routine care for all patients with GJH, with hematology referrals for patients with increased bleeding concerns,” wrote a research team led by Nicole E. Kendel, MD, a pediatric hematologist-oncologist at Akron Children’s Hospital in Ohio, in a study published online in Arthritis Care and Research.
“Further studies are needed to understand the mechanism of bleeding, evaluate comorbidities associated with these bleeding symptoms, and potentially allow for tailored pharmacologic therapy,” the authors stated.
Background
Dr. Kendel’s team had reported moderate menstruation-associated limitations in school, social, and physical activities among female adolescents with GJH. “This cohort also experienced nonreproductive bleeding symptoms and demonstrated minimal hemostatic laboratory abnormalities, indicating that this population may be underdiagnosed and subsequently poorly managed,” she said in an interview. “As excessive bleeding symptoms could have a significant impact on overall health and quality of life, we thought it was important to define the incidence and natural course of bleeding symptoms in a more generalized subset of this population.”
Although the investigators hypothesized that there would be a statistically significant increase in bleeding scores, “we were still impressed by the frequency of abnormal scores, particularly when looking at the low percentage of patients [12%] who had previously been referred to hematology,” she said.
Study results
The median age of the study cohort was 13 years (interquartile range, 10-16 years), and 72.8% were female. The mean Beighton score, which measures joint flexibility, was 6.2 (range, 4-9). All participants were seen by rheumatologists and were diagnosed for conditions on the hypermobility spectrum. Those conditions ranged from GJH to hypermobile Ehlers-Danlos syndrome (hEDS).
Abnormal bleeding, as measured by the International Society on Thrombosis and Haemostasis Bleeding Assessment Tool, was found in 75% (95% confidence interval [CI], 64%-84%). Overall mean and median bleeding scores were 5.2 and 4, respectively; scores ranged from 0 to 16. Abnormal scores of ≥ 3 were observed for patients < 8 years of age, ≥ 4 for men ≥ 18 years of age, and ≥ 6 for women ≥ 18 years of age. These measures were significantly elevated compared with those reported for historical healthy pediatric control persons (P < .001).
The most common hemorrhagic symptom was oral bleeding (74.1%) that occurred with tooth brushing, flossing, tooth loss, or eruption. Others reported easy bruising (59.3%) and bleeding from minor wounds (42%). In terms of procedures, tooth extraction requiring additional packing was reported by 25.9%, and 22.2% reported significant bleeding after otolaryngologic procedures, such as tonsillectomy/adenoidectomy, septoplasty, and nasal turbinate reduction.
Prolonged or heavy menstrual periods were reported by 37.3% of female patients.
Bleeding scores did not differ by biological sex or NSAID use, nor did any correlation emerge between patients’ bleeding and Beighton scores. However, there was a positive correlation with increasing age, a phenomenon observed with other bleeding disorders and in the healthy population, the authors noted.
Of the 10 study participants who had previously undergone hematologic assessment, one had been diagnosed with acquired, heart disease–related von Willebrand disease, and another with mild bleeding disorder.
Severe connective tissue disorders are associated with increased bleeding symptoms in the adult population, Dr. Kendel said, but few studies have assessed bleeding across the GJH spectrum, particularly in children.
Bleeding is thought to be due to modifications of collagen in the blood vessels. “These modifications create mechanical weakness of the vessel wall, as well as defective subendothelial connective tissue supporting those blood vessels,” Dr. Kendel explained. She noted that altered collagen creates defective interactions between collagen and other coagulation factors.
“Even in the presence of a normal laboratory evaluation, GJH can lead to symptoms consistent with a mild bleeding disorder,” she continued. “These symptoms are both preventable and treatable. I’m hopeful more centers will start routinely evaluating for increased bleeding symptoms, with referral to hematology for those with increased bleeding concerns.”
Commenting on the study’s recommendation, Beth S. Gottlieb, MD, chief of the division of pediatric rheumatology at Northwell Health in New Hyde Park, N.Y., who was not involved in the investigation, said a brief questionnaire on bleeding risk is a reasonable addition to a rheumatology office visit.
“Joint hypermobility is very common, but not all affected children meet the criteria for the hypermobile form of hEDS,” she told this news organization. “Screening for bleeding tendency is often done as routine medical history questions. Once a child is identified as hypermobile, these screening questions are usually asked, but utilizing one of the formal bleeding risk questionnaires is not currently routine.”
According to Dr. Gottlieb, it remains unclear whether screening would have a significant impact on children who have been diagnosed with hypermobility. “Most of these children are young and may not yet have a significant history for bleeding tendency,” she said. “Education of families is always important, and it will be essential to educate without adding unnecessary stress. Screening guidelines may be an important tool that is easy to incorporate into routine clinical practice.”
Limitation
The study was limited by selection bias, as patients had all been referred to a specialized rheumatology clinic.
The study was supported by the Clinical and Translational Intramural Funding Program of the Abigail Wexner Research Institute. The authors and Dr. Gottlieb have disclosed no relevant financial relationships.
*Correction, 1/11/2023: An earlier version of this story misstated the type of specialty clinic where patients were first seen.
A version of this article first appeared on Medscape.com.
A small cohort study of pediatric rheumatology patients with generalized joint hypermobility (GJH) who presented to a specialized rheumatology* clinic suggests that many such patients have abnormal bleeding symptoms, in comparison with health control patients.
The study of 81 patients with GJH found that about three quarters had significantly elevated median bleeding scores, but only 12% had been assessed by hematology for bleeding.
“We propose that screening for bleeding symptoms should be integrated into the routine care for all patients with GJH, with hematology referrals for patients with increased bleeding concerns,” wrote a research team led by Nicole E. Kendel, MD, a pediatric hematologist-oncologist at Akron Children’s Hospital in Ohio, in a study published online in Arthritis Care and Research.
“Further studies are needed to understand the mechanism of bleeding, evaluate comorbidities associated with these bleeding symptoms, and potentially allow for tailored pharmacologic therapy,” the authors stated.
Background
Dr. Kendel’s team had reported moderate menstruation-associated limitations in school, social, and physical activities among female adolescents with GJH. “This cohort also experienced nonreproductive bleeding symptoms and demonstrated minimal hemostatic laboratory abnormalities, indicating that this population may be underdiagnosed and subsequently poorly managed,” she said in an interview. “As excessive bleeding symptoms could have a significant impact on overall health and quality of life, we thought it was important to define the incidence and natural course of bleeding symptoms in a more generalized subset of this population.”
Although the investigators hypothesized that there would be a statistically significant increase in bleeding scores, “we were still impressed by the frequency of abnormal scores, particularly when looking at the low percentage of patients [12%] who had previously been referred to hematology,” she said.
Study results
The median age of the study cohort was 13 years (interquartile range, 10-16 years), and 72.8% were female. The mean Beighton score, which measures joint flexibility, was 6.2 (range, 4-9). All participants were seen by rheumatologists and were diagnosed for conditions on the hypermobility spectrum. Those conditions ranged from GJH to hypermobile Ehlers-Danlos syndrome (hEDS).
Abnormal bleeding, as measured by the International Society on Thrombosis and Haemostasis Bleeding Assessment Tool, was found in 75% (95% confidence interval [CI], 64%-84%). Overall mean and median bleeding scores were 5.2 and 4, respectively; scores ranged from 0 to 16. Abnormal scores of ≥ 3 were observed for patients < 8 years of age, ≥ 4 for men ≥ 18 years of age, and ≥ 6 for women ≥ 18 years of age. These measures were significantly elevated compared with those reported for historical healthy pediatric control persons (P < .001).
The most common hemorrhagic symptom was oral bleeding (74.1%) that occurred with tooth brushing, flossing, tooth loss, or eruption. Others reported easy bruising (59.3%) and bleeding from minor wounds (42%). In terms of procedures, tooth extraction requiring additional packing was reported by 25.9%, and 22.2% reported significant bleeding after otolaryngologic procedures, such as tonsillectomy/adenoidectomy, septoplasty, and nasal turbinate reduction.
Prolonged or heavy menstrual periods were reported by 37.3% of female patients.
Bleeding scores did not differ by biological sex or NSAID use, nor did any correlation emerge between patients’ bleeding and Beighton scores. However, there was a positive correlation with increasing age, a phenomenon observed with other bleeding disorders and in the healthy population, the authors noted.
Of the 10 study participants who had previously undergone hematologic assessment, one had been diagnosed with acquired, heart disease–related von Willebrand disease, and another with mild bleeding disorder.
Severe connective tissue disorders are associated with increased bleeding symptoms in the adult population, Dr. Kendel said, but few studies have assessed bleeding across the GJH spectrum, particularly in children.
Bleeding is thought to be due to modifications of collagen in the blood vessels. “These modifications create mechanical weakness of the vessel wall, as well as defective subendothelial connective tissue supporting those blood vessels,” Dr. Kendel explained. She noted that altered collagen creates defective interactions between collagen and other coagulation factors.
“Even in the presence of a normal laboratory evaluation, GJH can lead to symptoms consistent with a mild bleeding disorder,” she continued. “These symptoms are both preventable and treatable. I’m hopeful more centers will start routinely evaluating for increased bleeding symptoms, with referral to hematology for those with increased bleeding concerns.”
Commenting on the study’s recommendation, Beth S. Gottlieb, MD, chief of the division of pediatric rheumatology at Northwell Health in New Hyde Park, N.Y., who was not involved in the investigation, said a brief questionnaire on bleeding risk is a reasonable addition to a rheumatology office visit.
“Joint hypermobility is very common, but not all affected children meet the criteria for the hypermobile form of hEDS,” she told this news organization. “Screening for bleeding tendency is often done as routine medical history questions. Once a child is identified as hypermobile, these screening questions are usually asked, but utilizing one of the formal bleeding risk questionnaires is not currently routine.”
According to Dr. Gottlieb, it remains unclear whether screening would have a significant impact on children who have been diagnosed with hypermobility. “Most of these children are young and may not yet have a significant history for bleeding tendency,” she said. “Education of families is always important, and it will be essential to educate without adding unnecessary stress. Screening guidelines may be an important tool that is easy to incorporate into routine clinical practice.”
Limitation
The study was limited by selection bias, as patients had all been referred to a specialized rheumatology clinic.
The study was supported by the Clinical and Translational Intramural Funding Program of the Abigail Wexner Research Institute. The authors and Dr. Gottlieb have disclosed no relevant financial relationships.
*Correction, 1/11/2023: An earlier version of this story misstated the type of specialty clinic where patients were first seen.
A version of this article first appeared on Medscape.com.
FROM ARTHRITIS CARE AND RESEARCH
What to do when patients don’t listen
The term “nonadherent” has gradually replaced “noncompliant” in the physician lexicon as a nod to the evolving doctor-patient relationship. Noncompliance implies that a patient isn’t following their doctor’s orders. Adherence, on the other hand, is a measure of how closely your patient’s behavior matches the recommendations you’ve made. It’s a subtle difference but an important distinction in approaching care.
“Noncompliance is inherently negative feedback to the patient, whereas there’s a reason for nonadherence, and it’s usually external,” said Sharon Rabinovitz, MD, president of the Georgia Academy of Family Physicians.
Why won’t patients listen?
The reasons behind a patient’s nonadherence are multifaceted, but they are often driven by social determinants of health, such as transportation, poor health literacy, finances, and lack of access to pharmacies.
Other times, patients don’t want to take medicine, don’t prioritize their health, or they find the dietary and lifestyle modifications doctors suggest too hard to make or they struggle at losing weight, eating more healthfully, or cutting back on alcohol, for instance.
“When you come down to it, the big hindrance of it all is cost and the ability for the patient to be able to afford some of the things that we think they should be able to do,” said Teresa Lovins, MD, a physician in private practice Columbus, Ind., and a member of the board of directors of the American Academy of Family Physicians.
Another common deterrent to treatment is undesired side effects that a patient may not want to mention.
“For example, a lot of patients who are taking antidepressants have sexual dysfunction associated with those medications,” said Dr. Rabinovitz. “If you don’t ask the right questions, you’re not going to be able to fully assess the experience the patient is having and a reason why they might not take it [the medication].”
Much nonadherence is intentional and is based on experience, belief systems, and knowledge. For example, the American Medical Association finds that patients may not understand why they need a certain treatment (and therefore dismiss it), or they may be overloaded with multiple medications, fear dependency on a drug, have a mistrust of pharmaceutical companies or the medical system as a whole, or have symptoms of depression that make taking healthy actions more difficult. In addition, patients may be unable to afford their medication, or their lack of symptoms may lead them to believe they don’t really need the prescription, as occurs with disorders such as hypertension or high cholesterol.
“In my training, we did something called Balint training, where we would get together as a group with attendings and discuss cases that were difficult from a biopsychosocial perspective and consider all the factors in the patient perspective, including family dynamics, social systems, and economic realities,” said Russell Blackwelder, MD, director of geriatric education and associate professor of family medicine at the Medical University of South Carolina, Charleston.
“That training was, for me, very helpful for opening up and being more empathetic and really examining the patient’s point of view and everything that impacts them.”
Dr. Lovins agreed that it’s crucial to establish a good rapport and build mutual trust.
“If you don’t know the patient, you have a harder time asking the right questions to get to the meat of why they’re not taking their medicine or what they’re not doing to help their health,” she said. “It takes a little bit of trust on both parts to get to that question that really gets to the heart of why they’re not doing what you’re asking them to do.”
How to encourage adherence
Although there may not be a one-size-fits-all approach for achieving general adherence or adherence to a medication regimen, some methods may increase success.
Kenneth Zweig, MD, an internist at Northern Virginia Family Practice Associates, Alexandria, said that convincing patients to make one small change that they can sustain can get the ball rolling.
“I had one patient who was very overweight and had high blood pressure, high cholesterol, back pain, insomnia, and depression, who was also drinking three to four beers a night,” Dr. Zweig said. “After a long discussion, I challenged him to stop all alcohol for 1 week. At the end of the week, he noticed that he slept better, lost some weight, had lower blood pressure, and had more energy. Once he saw the benefits of this one change, he was motivated to improve other aspects of his health as well. He improved his diet, started exercising, and lost over 50 pounds. He has persisted with these lifestyle changes ever since.”
A team-based approach may also increase treatment understanding and adherence. In one older study, patients who were assigned to team-based care, including care by pharmacists, were significantly more adherent to medication regimens. Patients were more comfortable asking questions and raising concerns when they felt their treatment plan was a collaboration between several providers and themselves.
Dr. Lovins said to always approach the patient with a positive. “Say, what can we do together to make this work? What are your questions about this medication? And try and focus on the positive things that you can change instead of leaving the patient with a negative feeling or that you’re angry with them or that you’re unhappy with their choices. Patients respond better when they are treated as part of the team.”
Fear of judgment can also be a barrier to honesty between patients and their doctors. Shame creates a reluctance to admit nonadherence. Dr. Lovins said in an interview that it’s the physician’s responsibility to create a blame-free space for patients to speak openly about their struggles with treatment and reasons for nonadherence.
When should you redirect care?
Ultimately, the goal is good care and treatment of disease. However, if you and your patient are at an impasse and progress is stalling or failing, it may be appropriate to encourage the patient to seek care elsewhere.
“Just like any relationship, some physician-patient relationships are just not a good fit,” said Dr. Blackwelder. And this may be the reason why the patient is nonadherent — something between the two of you doesn’t click.
While there are ethical considerations for this decision, most medical boards have guidelines on how to go about it, Dr. Blackwelder said in an interview. “In the state of South Carolina, we have to be available to provide urgent coverage for at least 30 days and notify the patient in writing that they need to find somebody else and to help them find somebody else if we can.”
Just as with care, a clear conversation is the best practice if you’re proposing a potential shift away from a physician-patient relationship. You might say: We’re not making the kind of progress I’d like to see, and I’m wondering if you think working with another doctor may help you.
“The most important thing is being very honest and transparent with the patient that you’re concerned you’re not making the appropriate strides forward,” said Dr. Rabinovitz. Then you can ask, ‘Am I the right doctor to help you reach your goals? And if not, how can I help you get to where you need to be?’ ”
A version of this article first appeared on Medscape.com.
The term “nonadherent” has gradually replaced “noncompliant” in the physician lexicon as a nod to the evolving doctor-patient relationship. Noncompliance implies that a patient isn’t following their doctor’s orders. Adherence, on the other hand, is a measure of how closely your patient’s behavior matches the recommendations you’ve made. It’s a subtle difference but an important distinction in approaching care.
“Noncompliance is inherently negative feedback to the patient, whereas there’s a reason for nonadherence, and it’s usually external,” said Sharon Rabinovitz, MD, president of the Georgia Academy of Family Physicians.
Why won’t patients listen?
The reasons behind a patient’s nonadherence are multifaceted, but they are often driven by social determinants of health, such as transportation, poor health literacy, finances, and lack of access to pharmacies.
Other times, patients don’t want to take medicine, don’t prioritize their health, or they find the dietary and lifestyle modifications doctors suggest too hard to make or they struggle at losing weight, eating more healthfully, or cutting back on alcohol, for instance.
“When you come down to it, the big hindrance of it all is cost and the ability for the patient to be able to afford some of the things that we think they should be able to do,” said Teresa Lovins, MD, a physician in private practice Columbus, Ind., and a member of the board of directors of the American Academy of Family Physicians.
Another common deterrent to treatment is undesired side effects that a patient may not want to mention.
“For example, a lot of patients who are taking antidepressants have sexual dysfunction associated with those medications,” said Dr. Rabinovitz. “If you don’t ask the right questions, you’re not going to be able to fully assess the experience the patient is having and a reason why they might not take it [the medication].”
Much nonadherence is intentional and is based on experience, belief systems, and knowledge. For example, the American Medical Association finds that patients may not understand why they need a certain treatment (and therefore dismiss it), or they may be overloaded with multiple medications, fear dependency on a drug, have a mistrust of pharmaceutical companies or the medical system as a whole, or have symptoms of depression that make taking healthy actions more difficult. In addition, patients may be unable to afford their medication, or their lack of symptoms may lead them to believe they don’t really need the prescription, as occurs with disorders such as hypertension or high cholesterol.
“In my training, we did something called Balint training, where we would get together as a group with attendings and discuss cases that were difficult from a biopsychosocial perspective and consider all the factors in the patient perspective, including family dynamics, social systems, and economic realities,” said Russell Blackwelder, MD, director of geriatric education and associate professor of family medicine at the Medical University of South Carolina, Charleston.
“That training was, for me, very helpful for opening up and being more empathetic and really examining the patient’s point of view and everything that impacts them.”
Dr. Lovins agreed that it’s crucial to establish a good rapport and build mutual trust.
“If you don’t know the patient, you have a harder time asking the right questions to get to the meat of why they’re not taking their medicine or what they’re not doing to help their health,” she said. “It takes a little bit of trust on both parts to get to that question that really gets to the heart of why they’re not doing what you’re asking them to do.”
How to encourage adherence
Although there may not be a one-size-fits-all approach for achieving general adherence or adherence to a medication regimen, some methods may increase success.
Kenneth Zweig, MD, an internist at Northern Virginia Family Practice Associates, Alexandria, said that convincing patients to make one small change that they can sustain can get the ball rolling.
“I had one patient who was very overweight and had high blood pressure, high cholesterol, back pain, insomnia, and depression, who was also drinking three to four beers a night,” Dr. Zweig said. “After a long discussion, I challenged him to stop all alcohol for 1 week. At the end of the week, he noticed that he slept better, lost some weight, had lower blood pressure, and had more energy. Once he saw the benefits of this one change, he was motivated to improve other aspects of his health as well. He improved his diet, started exercising, and lost over 50 pounds. He has persisted with these lifestyle changes ever since.”
A team-based approach may also increase treatment understanding and adherence. In one older study, patients who were assigned to team-based care, including care by pharmacists, were significantly more adherent to medication regimens. Patients were more comfortable asking questions and raising concerns when they felt their treatment plan was a collaboration between several providers and themselves.
Dr. Lovins said to always approach the patient with a positive. “Say, what can we do together to make this work? What are your questions about this medication? And try and focus on the positive things that you can change instead of leaving the patient with a negative feeling or that you’re angry with them or that you’re unhappy with their choices. Patients respond better when they are treated as part of the team.”
Fear of judgment can also be a barrier to honesty between patients and their doctors. Shame creates a reluctance to admit nonadherence. Dr. Lovins said in an interview that it’s the physician’s responsibility to create a blame-free space for patients to speak openly about their struggles with treatment and reasons for nonadherence.
When should you redirect care?
Ultimately, the goal is good care and treatment of disease. However, if you and your patient are at an impasse and progress is stalling or failing, it may be appropriate to encourage the patient to seek care elsewhere.
“Just like any relationship, some physician-patient relationships are just not a good fit,” said Dr. Blackwelder. And this may be the reason why the patient is nonadherent — something between the two of you doesn’t click.
While there are ethical considerations for this decision, most medical boards have guidelines on how to go about it, Dr. Blackwelder said in an interview. “In the state of South Carolina, we have to be available to provide urgent coverage for at least 30 days and notify the patient in writing that they need to find somebody else and to help them find somebody else if we can.”
Just as with care, a clear conversation is the best practice if you’re proposing a potential shift away from a physician-patient relationship. You might say: We’re not making the kind of progress I’d like to see, and I’m wondering if you think working with another doctor may help you.
“The most important thing is being very honest and transparent with the patient that you’re concerned you’re not making the appropriate strides forward,” said Dr. Rabinovitz. Then you can ask, ‘Am I the right doctor to help you reach your goals? And if not, how can I help you get to where you need to be?’ ”
A version of this article first appeared on Medscape.com.
The term “nonadherent” has gradually replaced “noncompliant” in the physician lexicon as a nod to the evolving doctor-patient relationship. Noncompliance implies that a patient isn’t following their doctor’s orders. Adherence, on the other hand, is a measure of how closely your patient’s behavior matches the recommendations you’ve made. It’s a subtle difference but an important distinction in approaching care.
“Noncompliance is inherently negative feedback to the patient, whereas there’s a reason for nonadherence, and it’s usually external,” said Sharon Rabinovitz, MD, president of the Georgia Academy of Family Physicians.
Why won’t patients listen?
The reasons behind a patient’s nonadherence are multifaceted, but they are often driven by social determinants of health, such as transportation, poor health literacy, finances, and lack of access to pharmacies.
Other times, patients don’t want to take medicine, don’t prioritize their health, or they find the dietary and lifestyle modifications doctors suggest too hard to make or they struggle at losing weight, eating more healthfully, or cutting back on alcohol, for instance.
“When you come down to it, the big hindrance of it all is cost and the ability for the patient to be able to afford some of the things that we think they should be able to do,” said Teresa Lovins, MD, a physician in private practice Columbus, Ind., and a member of the board of directors of the American Academy of Family Physicians.
Another common deterrent to treatment is undesired side effects that a patient may not want to mention.
“For example, a lot of patients who are taking antidepressants have sexual dysfunction associated with those medications,” said Dr. Rabinovitz. “If you don’t ask the right questions, you’re not going to be able to fully assess the experience the patient is having and a reason why they might not take it [the medication].”
Much nonadherence is intentional and is based on experience, belief systems, and knowledge. For example, the American Medical Association finds that patients may not understand why they need a certain treatment (and therefore dismiss it), or they may be overloaded with multiple medications, fear dependency on a drug, have a mistrust of pharmaceutical companies or the medical system as a whole, or have symptoms of depression that make taking healthy actions more difficult. In addition, patients may be unable to afford their medication, or their lack of symptoms may lead them to believe they don’t really need the prescription, as occurs with disorders such as hypertension or high cholesterol.
“In my training, we did something called Balint training, where we would get together as a group with attendings and discuss cases that were difficult from a biopsychosocial perspective and consider all the factors in the patient perspective, including family dynamics, social systems, and economic realities,” said Russell Blackwelder, MD, director of geriatric education and associate professor of family medicine at the Medical University of South Carolina, Charleston.
“That training was, for me, very helpful for opening up and being more empathetic and really examining the patient’s point of view and everything that impacts them.”
Dr. Lovins agreed that it’s crucial to establish a good rapport and build mutual trust.
“If you don’t know the patient, you have a harder time asking the right questions to get to the meat of why they’re not taking their medicine or what they’re not doing to help their health,” she said. “It takes a little bit of trust on both parts to get to that question that really gets to the heart of why they’re not doing what you’re asking them to do.”
How to encourage adherence
Although there may not be a one-size-fits-all approach for achieving general adherence or adherence to a medication regimen, some methods may increase success.
Kenneth Zweig, MD, an internist at Northern Virginia Family Practice Associates, Alexandria, said that convincing patients to make one small change that they can sustain can get the ball rolling.
“I had one patient who was very overweight and had high blood pressure, high cholesterol, back pain, insomnia, and depression, who was also drinking three to four beers a night,” Dr. Zweig said. “After a long discussion, I challenged him to stop all alcohol for 1 week. At the end of the week, he noticed that he slept better, lost some weight, had lower blood pressure, and had more energy. Once he saw the benefits of this one change, he was motivated to improve other aspects of his health as well. He improved his diet, started exercising, and lost over 50 pounds. He has persisted with these lifestyle changes ever since.”
A team-based approach may also increase treatment understanding and adherence. In one older study, patients who were assigned to team-based care, including care by pharmacists, were significantly more adherent to medication regimens. Patients were more comfortable asking questions and raising concerns when they felt their treatment plan was a collaboration between several providers and themselves.
Dr. Lovins said to always approach the patient with a positive. “Say, what can we do together to make this work? What are your questions about this medication? And try and focus on the positive things that you can change instead of leaving the patient with a negative feeling or that you’re angry with them or that you’re unhappy with their choices. Patients respond better when they are treated as part of the team.”
Fear of judgment can also be a barrier to honesty between patients and their doctors. Shame creates a reluctance to admit nonadherence. Dr. Lovins said in an interview that it’s the physician’s responsibility to create a blame-free space for patients to speak openly about their struggles with treatment and reasons for nonadherence.
When should you redirect care?
Ultimately, the goal is good care and treatment of disease. However, if you and your patient are at an impasse and progress is stalling or failing, it may be appropriate to encourage the patient to seek care elsewhere.
“Just like any relationship, some physician-patient relationships are just not a good fit,” said Dr. Blackwelder. And this may be the reason why the patient is nonadherent — something between the two of you doesn’t click.
While there are ethical considerations for this decision, most medical boards have guidelines on how to go about it, Dr. Blackwelder said in an interview. “In the state of South Carolina, we have to be available to provide urgent coverage for at least 30 days and notify the patient in writing that they need to find somebody else and to help them find somebody else if we can.”
Just as with care, a clear conversation is the best practice if you’re proposing a potential shift away from a physician-patient relationship. You might say: We’re not making the kind of progress I’d like to see, and I’m wondering if you think working with another doctor may help you.
“The most important thing is being very honest and transparent with the patient that you’re concerned you’re not making the appropriate strides forward,” said Dr. Rabinovitz. Then you can ask, ‘Am I the right doctor to help you reach your goals? And if not, how can I help you get to where you need to be?’ ”
A version of this article first appeared on Medscape.com.
Warfarin best for thrombotic antiphospholipid syndrome?
Patients with thrombotic antiphospholipid syndrome are better treated with a vitamin K antagonist, such as warfarin, rather than a direct oral anticoagulant (DOAC), a new systematic review and meta-analysis suggests.
“Our study is showing that in randomized controlled trials in patients with thrombotic antiphospholipid syndrome, the risk of arterial thrombotic events, particularly stroke, is significantly increased with DOACs vs. vitamin K antagonists,” senior author, Behnood Bikdeli, MD, Brigham and Women’s Hospital, Boston, told this news organization. “These results probably suggest that DOACs are not the optimal regimen for patients with thrombotic antiphospholipid syndrome.”
The study was published online in the Journal of the American College of Cardiology.
Autoimmune disorder
Thrombotic antiphospholipid syndrome is a systemic autoimmune disorder characterized by recurrent arterial and/or venous thrombotic events.
Dr. Bikdeli estimates that antiphospholipid syndrome is the cause of 50,000-100,000 strokes, 100,000 cases of myocardial infarction, and 30,000 cases of deep vein thrombosis every year.
“It is a serious condition, and these are a high-risk and complex group of patients,” he said.
The standard treatment has been anticoagulation with a vitamin K antagonist such as warfarin. “But this is a cumbersome treatment, with many drug interactions and the need for INR [International Normalized Ratio] monitoring, which can be difficult to manage in patients with antiphospholipid syndrome as there can sometimes be falsely abnormal numbers,” Dr. Bikdeli noted. “Because of these challenges, it looked very promising to explore the use of DOACs in this population.”
Four main randomized trials have been conducted to investigate the use of DOACs in antiphospholipid syndrome – three with rivaroxaban and one with apixaban. “These trials were all quite small and, while they did not show definite results, some of them suggested nonsignificant findings of slightly worse outcomes for DOACs vs. vitamin K antagonists. But there is a lot of uncertainty, and it is difficult to look at subgroups in such small trials,” Dr. Bikdeli said. “There are many questions remaining about whether we should use DOACs in patients with antiphospholipid syndrome and, if so, which particular subgroups.”
The authors therefore performed a systematic review and meta-analysis of randomized controlled trials that compared DOACs with vitamin K antagonists in patients with antiphospholipid syndrome. They also contacted the principal investigators of the trials to obtain additional unpublished aggregate level data on specific subgroups.
Four open-label randomized controlled trials involving 472 patients were included in the meta-analysis.
Overall, the use of DOACs, compared with vitamin K antagonists, was associated with increased odds of subsequent arterial thrombotic events (odds ratio, 5.43; P < .001), especially stroke.
The odds of subsequent venous thrombotic events or major bleeding were not significantly different between the two groups. Most findings were consistent within subgroups.
“Our results show that use of DOACs vs. vitamin K antagonists is associated with increased risk of arterial thrombotic events – a risk that is primarily driven by a significant increase in the risk of stroke,” Dr. Bikdeli commented.
When looking at subgroups of interest, it was previously thought that DOACs may not be so effective in the so-called “triple-positive” antiphospholipid patients. These patients have three different types of antibodies and have the highest risk of thrombosis, Dr. Bikdeli noted.
“But one of the interesting findings of our study is that the results are actually consistent in women vs. men and in people who have triple-positive antibodies and those who had double- or single-positive antibodies,” he said. “Our analyses did not show effect modification by antibody subgroups. They suggest similar trends towards worse outcomes in all subgroups.”
“From these results, I would be similarly concerned to use DOACs even if someone has double-positive or single-positive antiphospholipid antibodies,” he added.
Dr. Bikdeli said he would still recommend shared decision-making with patients. “If I have a patient who has thrombotic antiphospholipid syndrome, I would share my reservation about DOACs, but there are multiple factors that come into decision-making. If someone has difficulty with checking INRs, we may make an informed choice and still use a DOAC, but patients need to know that there is likely an excess risk of subsequent arterial events with DOACs, compared with a vitamin K antagonist.”
He noted that it is still not completely clear on the situation for people with single-positive antiphospholipid syndrome or the type of antibody that is present. It is also possible that a higher dose of DOAC could be more effective, a strategy that is being investigated in a separate randomized trial currently ongoing.
“But for routine practice I would have concerns about using DOACs in antiphospholipid syndrome patients in general,” he said. “For triple positive there is more data and greater concern, but I wouldn’t give a pass for a double- or single-positive patient either.”
The reason why DOACs would be less effective than vitamin K antagonists in antiphospholipid syndrome is not known.
“That is the million-dollar question,” Dr. Bikdeli commented. “DOACs have been such helpful drugs for many patients and clinicians as well. But we have seen that they are not optimal in a series of scenarios now – patients with mechanical heart valves, patients with rheumatic [atrial fibrillaton], and now patients with thrombotic antiphospholipid syndrome.”
One hypothesis is that these patients have some more components of inflammation and are more prone to blood clots, and because vitamin K antagonists work at several parts of the coagulation cascade, they might be more successful, compared with the more targeted DOAC therapy. “But I think we need more studies to fully understand this,” he said.
‘Important implications’
In an accompanying editorial,Mark A. Crowther, MD, McMaster University, Hamilton, Ont., and Aubrey E. Jones, PharmD, and Daniel M. Witt, PharmD, both of the University of Utah College of Pharmacy, Salt Lake City, say that: “As the quality of the evidence was rated ‘high’ for the arterial thrombosis outcome and ‘moderate’ for the venous thrombosis and bleeding outcomes, these results should lead to a revision of evidence-based guidelines to recommend against using DOACs as an option for most patients with thrombotic antiphospholipid syndrome.”
They add that this recommendation for vitamin K antagonists also applies to patients previously thought to be at lower risk from antiphospholipid syndrome – including those with only one or two positive serological tests and those with only prior venous thrombosis.
The editorialists point out that this will have important implications, particularly for the accurate diagnosis of antiphospholipid syndrome, including confirmation and documentation of positive laboratory tests at least 12 weeks after the initial positive test.
They recommend that while awaiting confirmatory testing, patients with suspected antiphospholipid syndrome should avoid DOACs, and that “strong consideration” should be given to switching essentially all antiphospholipid syndrome patients currently receiving DOACs to vitamin K antagonists.
Dr. Bikdeli is a consulting expert, on behalf of the plaintiff, for litigation related to two specific brand models of IVC filters and is supported by the Scott Schoen and Nancy Adams IGNITE Award from the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital and a Career Development Award from the American Heart Association and VIVA Physicians. Dr. Crowther has received personal funding from AstraZeneca, Precision Biologics, Hemostasis Reference Laboratories, Syneos Health, Bayer, Pfizer, and CSL Behring; and holds the Leo Pharma Chair in Thromboembolism Research, which is endowed at McMaster University. Dr. Jones is supported by a career development award from the National Heart, Lung, and Blood Institute; and Dr. Witt is supported by grant funding from the Agency for Healthcare Research and Quality.
A version of this article first appeared on Medscape.com.
Patients with thrombotic antiphospholipid syndrome are better treated with a vitamin K antagonist, such as warfarin, rather than a direct oral anticoagulant (DOAC), a new systematic review and meta-analysis suggests.
“Our study is showing that in randomized controlled trials in patients with thrombotic antiphospholipid syndrome, the risk of arterial thrombotic events, particularly stroke, is significantly increased with DOACs vs. vitamin K antagonists,” senior author, Behnood Bikdeli, MD, Brigham and Women’s Hospital, Boston, told this news organization. “These results probably suggest that DOACs are not the optimal regimen for patients with thrombotic antiphospholipid syndrome.”
The study was published online in the Journal of the American College of Cardiology.
Autoimmune disorder
Thrombotic antiphospholipid syndrome is a systemic autoimmune disorder characterized by recurrent arterial and/or venous thrombotic events.
Dr. Bikdeli estimates that antiphospholipid syndrome is the cause of 50,000-100,000 strokes, 100,000 cases of myocardial infarction, and 30,000 cases of deep vein thrombosis every year.
“It is a serious condition, and these are a high-risk and complex group of patients,” he said.
The standard treatment has been anticoagulation with a vitamin K antagonist such as warfarin. “But this is a cumbersome treatment, with many drug interactions and the need for INR [International Normalized Ratio] monitoring, which can be difficult to manage in patients with antiphospholipid syndrome as there can sometimes be falsely abnormal numbers,” Dr. Bikdeli noted. “Because of these challenges, it looked very promising to explore the use of DOACs in this population.”
Four main randomized trials have been conducted to investigate the use of DOACs in antiphospholipid syndrome – three with rivaroxaban and one with apixaban. “These trials were all quite small and, while they did not show definite results, some of them suggested nonsignificant findings of slightly worse outcomes for DOACs vs. vitamin K antagonists. But there is a lot of uncertainty, and it is difficult to look at subgroups in such small trials,” Dr. Bikdeli said. “There are many questions remaining about whether we should use DOACs in patients with antiphospholipid syndrome and, if so, which particular subgroups.”
The authors therefore performed a systematic review and meta-analysis of randomized controlled trials that compared DOACs with vitamin K antagonists in patients with antiphospholipid syndrome. They also contacted the principal investigators of the trials to obtain additional unpublished aggregate level data on specific subgroups.
Four open-label randomized controlled trials involving 472 patients were included in the meta-analysis.
Overall, the use of DOACs, compared with vitamin K antagonists, was associated with increased odds of subsequent arterial thrombotic events (odds ratio, 5.43; P < .001), especially stroke.
The odds of subsequent venous thrombotic events or major bleeding were not significantly different between the two groups. Most findings were consistent within subgroups.
“Our results show that use of DOACs vs. vitamin K antagonists is associated with increased risk of arterial thrombotic events – a risk that is primarily driven by a significant increase in the risk of stroke,” Dr. Bikdeli commented.
When looking at subgroups of interest, it was previously thought that DOACs may not be so effective in the so-called “triple-positive” antiphospholipid patients. These patients have three different types of antibodies and have the highest risk of thrombosis, Dr. Bikdeli noted.
“But one of the interesting findings of our study is that the results are actually consistent in women vs. men and in people who have triple-positive antibodies and those who had double- or single-positive antibodies,” he said. “Our analyses did not show effect modification by antibody subgroups. They suggest similar trends towards worse outcomes in all subgroups.”
“From these results, I would be similarly concerned to use DOACs even if someone has double-positive or single-positive antiphospholipid antibodies,” he added.
Dr. Bikdeli said he would still recommend shared decision-making with patients. “If I have a patient who has thrombotic antiphospholipid syndrome, I would share my reservation about DOACs, but there are multiple factors that come into decision-making. If someone has difficulty with checking INRs, we may make an informed choice and still use a DOAC, but patients need to know that there is likely an excess risk of subsequent arterial events with DOACs, compared with a vitamin K antagonist.”
He noted that it is still not completely clear on the situation for people with single-positive antiphospholipid syndrome or the type of antibody that is present. It is also possible that a higher dose of DOAC could be more effective, a strategy that is being investigated in a separate randomized trial currently ongoing.
“But for routine practice I would have concerns about using DOACs in antiphospholipid syndrome patients in general,” he said. “For triple positive there is more data and greater concern, but I wouldn’t give a pass for a double- or single-positive patient either.”
The reason why DOACs would be less effective than vitamin K antagonists in antiphospholipid syndrome is not known.
“That is the million-dollar question,” Dr. Bikdeli commented. “DOACs have been such helpful drugs for many patients and clinicians as well. But we have seen that they are not optimal in a series of scenarios now – patients with mechanical heart valves, patients with rheumatic [atrial fibrillaton], and now patients with thrombotic antiphospholipid syndrome.”
One hypothesis is that these patients have some more components of inflammation and are more prone to blood clots, and because vitamin K antagonists work at several parts of the coagulation cascade, they might be more successful, compared with the more targeted DOAC therapy. “But I think we need more studies to fully understand this,” he said.
‘Important implications’
In an accompanying editorial,Mark A. Crowther, MD, McMaster University, Hamilton, Ont., and Aubrey E. Jones, PharmD, and Daniel M. Witt, PharmD, both of the University of Utah College of Pharmacy, Salt Lake City, say that: “As the quality of the evidence was rated ‘high’ for the arterial thrombosis outcome and ‘moderate’ for the venous thrombosis and bleeding outcomes, these results should lead to a revision of evidence-based guidelines to recommend against using DOACs as an option for most patients with thrombotic antiphospholipid syndrome.”
They add that this recommendation for vitamin K antagonists also applies to patients previously thought to be at lower risk from antiphospholipid syndrome – including those with only one or two positive serological tests and those with only prior venous thrombosis.
The editorialists point out that this will have important implications, particularly for the accurate diagnosis of antiphospholipid syndrome, including confirmation and documentation of positive laboratory tests at least 12 weeks after the initial positive test.
They recommend that while awaiting confirmatory testing, patients with suspected antiphospholipid syndrome should avoid DOACs, and that “strong consideration” should be given to switching essentially all antiphospholipid syndrome patients currently receiving DOACs to vitamin K antagonists.
Dr. Bikdeli is a consulting expert, on behalf of the plaintiff, for litigation related to two specific brand models of IVC filters and is supported by the Scott Schoen and Nancy Adams IGNITE Award from the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital and a Career Development Award from the American Heart Association and VIVA Physicians. Dr. Crowther has received personal funding from AstraZeneca, Precision Biologics, Hemostasis Reference Laboratories, Syneos Health, Bayer, Pfizer, and CSL Behring; and holds the Leo Pharma Chair in Thromboembolism Research, which is endowed at McMaster University. Dr. Jones is supported by a career development award from the National Heart, Lung, and Blood Institute; and Dr. Witt is supported by grant funding from the Agency for Healthcare Research and Quality.
A version of this article first appeared on Medscape.com.
Patients with thrombotic antiphospholipid syndrome are better treated with a vitamin K antagonist, such as warfarin, rather than a direct oral anticoagulant (DOAC), a new systematic review and meta-analysis suggests.
“Our study is showing that in randomized controlled trials in patients with thrombotic antiphospholipid syndrome, the risk of arterial thrombotic events, particularly stroke, is significantly increased with DOACs vs. vitamin K antagonists,” senior author, Behnood Bikdeli, MD, Brigham and Women’s Hospital, Boston, told this news organization. “These results probably suggest that DOACs are not the optimal regimen for patients with thrombotic antiphospholipid syndrome.”
The study was published online in the Journal of the American College of Cardiology.
Autoimmune disorder
Thrombotic antiphospholipid syndrome is a systemic autoimmune disorder characterized by recurrent arterial and/or venous thrombotic events.
Dr. Bikdeli estimates that antiphospholipid syndrome is the cause of 50,000-100,000 strokes, 100,000 cases of myocardial infarction, and 30,000 cases of deep vein thrombosis every year.
“It is a serious condition, and these are a high-risk and complex group of patients,” he said.
The standard treatment has been anticoagulation with a vitamin K antagonist such as warfarin. “But this is a cumbersome treatment, with many drug interactions and the need for INR [International Normalized Ratio] monitoring, which can be difficult to manage in patients with antiphospholipid syndrome as there can sometimes be falsely abnormal numbers,” Dr. Bikdeli noted. “Because of these challenges, it looked very promising to explore the use of DOACs in this population.”
Four main randomized trials have been conducted to investigate the use of DOACs in antiphospholipid syndrome – three with rivaroxaban and one with apixaban. “These trials were all quite small and, while they did not show definite results, some of them suggested nonsignificant findings of slightly worse outcomes for DOACs vs. vitamin K antagonists. But there is a lot of uncertainty, and it is difficult to look at subgroups in such small trials,” Dr. Bikdeli said. “There are many questions remaining about whether we should use DOACs in patients with antiphospholipid syndrome and, if so, which particular subgroups.”
The authors therefore performed a systematic review and meta-analysis of randomized controlled trials that compared DOACs with vitamin K antagonists in patients with antiphospholipid syndrome. They also contacted the principal investigators of the trials to obtain additional unpublished aggregate level data on specific subgroups.
Four open-label randomized controlled trials involving 472 patients were included in the meta-analysis.
Overall, the use of DOACs, compared with vitamin K antagonists, was associated with increased odds of subsequent arterial thrombotic events (odds ratio, 5.43; P < .001), especially stroke.
The odds of subsequent venous thrombotic events or major bleeding were not significantly different between the two groups. Most findings were consistent within subgroups.
“Our results show that use of DOACs vs. vitamin K antagonists is associated with increased risk of arterial thrombotic events – a risk that is primarily driven by a significant increase in the risk of stroke,” Dr. Bikdeli commented.
When looking at subgroups of interest, it was previously thought that DOACs may not be so effective in the so-called “triple-positive” antiphospholipid patients. These patients have three different types of antibodies and have the highest risk of thrombosis, Dr. Bikdeli noted.
“But one of the interesting findings of our study is that the results are actually consistent in women vs. men and in people who have triple-positive antibodies and those who had double- or single-positive antibodies,” he said. “Our analyses did not show effect modification by antibody subgroups. They suggest similar trends towards worse outcomes in all subgroups.”
“From these results, I would be similarly concerned to use DOACs even if someone has double-positive or single-positive antiphospholipid antibodies,” he added.
Dr. Bikdeli said he would still recommend shared decision-making with patients. “If I have a patient who has thrombotic antiphospholipid syndrome, I would share my reservation about DOACs, but there are multiple factors that come into decision-making. If someone has difficulty with checking INRs, we may make an informed choice and still use a DOAC, but patients need to know that there is likely an excess risk of subsequent arterial events with DOACs, compared with a vitamin K antagonist.”
He noted that it is still not completely clear on the situation for people with single-positive antiphospholipid syndrome or the type of antibody that is present. It is also possible that a higher dose of DOAC could be more effective, a strategy that is being investigated in a separate randomized trial currently ongoing.
“But for routine practice I would have concerns about using DOACs in antiphospholipid syndrome patients in general,” he said. “For triple positive there is more data and greater concern, but I wouldn’t give a pass for a double- or single-positive patient either.”
The reason why DOACs would be less effective than vitamin K antagonists in antiphospholipid syndrome is not known.
“That is the million-dollar question,” Dr. Bikdeli commented. “DOACs have been such helpful drugs for many patients and clinicians as well. But we have seen that they are not optimal in a series of scenarios now – patients with mechanical heart valves, patients with rheumatic [atrial fibrillaton], and now patients with thrombotic antiphospholipid syndrome.”
One hypothesis is that these patients have some more components of inflammation and are more prone to blood clots, and because vitamin K antagonists work at several parts of the coagulation cascade, they might be more successful, compared with the more targeted DOAC therapy. “But I think we need more studies to fully understand this,” he said.
‘Important implications’
In an accompanying editorial,Mark A. Crowther, MD, McMaster University, Hamilton, Ont., and Aubrey E. Jones, PharmD, and Daniel M. Witt, PharmD, both of the University of Utah College of Pharmacy, Salt Lake City, say that: “As the quality of the evidence was rated ‘high’ for the arterial thrombosis outcome and ‘moderate’ for the venous thrombosis and bleeding outcomes, these results should lead to a revision of evidence-based guidelines to recommend against using DOACs as an option for most patients with thrombotic antiphospholipid syndrome.”
They add that this recommendation for vitamin K antagonists also applies to patients previously thought to be at lower risk from antiphospholipid syndrome – including those with only one or two positive serological tests and those with only prior venous thrombosis.
The editorialists point out that this will have important implications, particularly for the accurate diagnosis of antiphospholipid syndrome, including confirmation and documentation of positive laboratory tests at least 12 weeks after the initial positive test.
They recommend that while awaiting confirmatory testing, patients with suspected antiphospholipid syndrome should avoid DOACs, and that “strong consideration” should be given to switching essentially all antiphospholipid syndrome patients currently receiving DOACs to vitamin K antagonists.
Dr. Bikdeli is a consulting expert, on behalf of the plaintiff, for litigation related to two specific brand models of IVC filters and is supported by the Scott Schoen and Nancy Adams IGNITE Award from the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital and a Career Development Award from the American Heart Association and VIVA Physicians. Dr. Crowther has received personal funding from AstraZeneca, Precision Biologics, Hemostasis Reference Laboratories, Syneos Health, Bayer, Pfizer, and CSL Behring; and holds the Leo Pharma Chair in Thromboembolism Research, which is endowed at McMaster University. Dr. Jones is supported by a career development award from the National Heart, Lung, and Blood Institute; and Dr. Witt is supported by grant funding from the Agency for Healthcare Research and Quality.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Age competency exams for physicians – yes or no?
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert Glatter, medical advisor for Medscape Emergency Medicine. Joining me today is Sandeep Jauhar, a practicing cardiologist and professor of medicine at Northwell Health, a frequent New York Times op-ed contributor, and highly regarded author of the upcoming book “My Father’s Brain: Life in the Shadow of Alzheimer’s.”
Sandeep Jauhar, MD: Thanks for having me.
Dr. Glatter: Your recent op-ed piece in the New York Times caught my eye. In your piece, you refer to a 2020 survey in which almost one-third of licensed doctors in the United States were 60 years of age or older, up from a quarter in 2010. You also state that, due to a 20% prevalence of mild cognitive impairment in persons older than 65, practicing physicians above this age should probably be screened by a battery of tests to ensure that their reasoning and cognitive abilities are intact. The title of the article is “How Would You Feel About a 100-Year-Old Doctor?”
How would you envision such a process? What aspects of day-to-day functioning would the exams truly be evaluating?
Dr. Jauhar: A significant number of people over 65 have measurable cognitive impairment. By cognitive impairment, we’re not talking about dementia. The best estimates are that 1 in 10 people over age 65 have dementia, and roughly 1 in 5 have what’s called MCI, or mild cognitive impairment, which is cognitive impairment out of proportion to what you’d expect from normal aging. It’s a significant issue.
The argument that I made in the op-ed is that neurocognitive assessment is important. That’s not to say that everyone over age 65 has significant cognitive impairment or that older doctors can’t practice medicine safely and effectively. They absolutely can. The question is, do we leave neurocognitive assessment to physicians who may possibly be suffering from impairment?
In dementia, people very often have impaired self-awareness, a condition called anosognosia, which is a neurological term for not being aware of your own impairment because of your impairment.
I would argue that, instead of having voluntary neurocognitive screening, it should be mandated. The question is how to do that effectively, fairly, and transparently.
One could argue a gerontocracy in medicine today, where there are so many older physicians. What do we do about that? That really is something that I think needs to be debated.
Dr. Glatter: The question I have is, if we (that is, physicians and the health care profession) don’t take care of this, someone’s going to do it for us. We need to jump on this now while we have the opportunity. The AMA has been opposed to this, except when you have reason to suspect cognitive decline or are concerned about patient safety. A mandatory age of retirement is certainly something they’re not for, and we know this.
Your argument in your op-ed piece is very well thought out, and you lay the groundwork for testing (looking at someone’s memory, coordination, processing speed, and other executive functions). Certainly, for a psychiatrist, hearing is important, and for a dermatologist, vision is important. For a surgeon, there are other issues. Based on the specialty, we must be careful to see the important aspects of functioning. I am sure you would agree with this.
Dr. Jauhar: Obviously, the hand skills that are important for ophthalmological surgery certainly aren’t required for office-based psychological counseling, for example. We have to be smart about how we assess impairment.
You describe the spectrum of actions. On the one hand, there’s mandatory retirement at the age of 65 or 70 years. We know that commercial pilots are mandated to essentially retire at 65, and air-traffic controllers must retire in their late 50s.
We know that there’s a large amount of variability in competence. There are internists in their 80s with whom I’ve worked, and I’m absolutely wowed by their experience and judgment. There are new medical resident graduates who don’t really seem to have the requisite level of competence that would make me feel comfortable to have them as my doctor or a doctor for a member of my family.
To mandate retirement, I think the AMA is absolutely right. To not call for any kind of competency testing, to me, seems equally unwise. Because at the end of the day, you have to balance individual physician needs or wants to continue practicing with patient safety. I haven’t really come across too many physicians who say, “There’s absolutely no need for a competency testing.”
We have to meet somewhere in the middle. The middle is either voluntary cognitive competency testing or mandatory. I would argue that, because we know that as the brain changes we have cognitive impairment, but we’re not always aware that we need help, mandatory testing is the way.
One other thing that you mentioned was about having the solution imposed on us. You and I are doctors. We deal with bureaucracy. We deal with poorly thought-out solutions to issues in health care that make our lives that much more difficult. I don’t want that solution imposed on us by some outside agency. I think we need to figure this out within medicine and figure out the right way of doing it.
The AMA is on board with this. They haven’t called for mandatory testing, but they have said that if testing were to occur, these are the guidelines. The guidelines are fair and equitable, not too time-consuming, transparent, and not punitive. If someone comes out and doesn’t test well, we shouldn’t force them out of the profession. We can find ways to use their experience to help train younger doctors, for example.
Dr. Glatter: I wanted to segue to an area where there has been some challenge to the legality of these mandatory types of age restrictions and imposing the exams as well. There’s been a lawsuit as well by the EEOC [Equal Employment Opportunity Commission], on behalf of Yale. Basically, there’s been a concern that ageism is part of what’s going on. Yale now screens their providers beginning at age 70, and they have a program. UCSD [University of California, San Diego] has a program in place. Obviously, these institutions are looking at it. This is a very small part of the overall picture.
Health care systems overall, we’re talking about a fraction of them in the country are really addressing the issue of competency exams. The question is, where do we go from here? How do we get engagement or adoption and get physicians as a whole to embrace this concept?
Dr. Jauhar: The EEOC filed a lawsuit on behalf of the Yale medical staff that argued that Yale’s plan to do vision testing and neurocognitive screening – there may be a physical exam also – constitutes age discrimination because it’s reserved for doctors over the age of 70. Those are the physicians who are most likely to have cognitive impairment.
We have rules already for impaired physicians who are, for example, addicted to illicit drugs or have alcohol abuse. We already have some of those measures in place. This is focused on cognitive impairment in aging physicians because cognitive impairment is an issue that arises with aging. We have to be clear about that.
Most younger physicians will not have measurable cognitive impairment that would impair their ability to practice. To force young physicians (for example, physicians in their forties) to undergo such screening, all in the name of preventing age discrimination, doesn’t strike me as being a good use of resources. They’re more likely to be false positives, as you know from Bayesian statistics. When you have low pretest probability, you’re more likely to get false positives.
How are we going to screen hundreds of thousands of physicians? We have to make a choice about the group that really is more likely to benefit from such screening. Very few hospitals are addressing this issue and it’s going to become more important.
Dr. Glatter: Surgeons have been particularly active in pushing for age-based screening. In 2016, the American College of Surgeons started making surgeons at age 65-70 undergo voluntary health and neurocognitive assessments, and encouraged physicians to disclose any concerning findings as part of their professional obligation, which is pretty impressive in my mind.
Surgeons’ skill set is quite demanding physically and technically. That the Society of Surgical Chairs took it upon themselves to institute this is pretty telling.
Dr. Jauhar: The overall society called for screening, but then in a separate survey of surgical chairs, the idea was advanced that we should have mandatory retirement. Now, I don’t particularly agree with that.
I’ve seen it, where you have the aging surgeon who was a star in their day, and no one wants to say anything when their skills have visibly degraded, and no one wants to carry that torch and tell them that they need to retire. What happens is people whisper, and unfortunately, bad outcomes have to occur before people tend to get involved, and that’s what I’m trying to prevent.
Dr. Glatter: The question is whether older physicians have worse patient outcomes. The evidence is inconclusive, but studies have shown higher mortality rates for cardiovascular surgeons in terms of the procedures that they do. On the flip side, there are also higher mortality rates for GI surgery performed by younger surgeons. It’s a mixed bag.
Dr. Jauhar: For specialized surgery, you need the accrual of a certain amount of experience. The optimal age is about 60, because they’ve seen many things and they’ve seen complications. They don’t have a hand tremor yet so they’re still functioning well, and they’ve accrued a lot of experience. We have to be smart about who we screen.
There’s a learning curve in surgery. By no means am I arguing that younger surgeons are better surgeons. I would say that there’s probably a tipping point where once you get past a certain age and physical deterioration starts to take effect, that can overshadow the accrual of cognitive and surgical experience. We have to balance those things.
I would say neurocognitive screening and vision testing are important, but exactly what do you measure? How much of a hand tremor would constitute a risk? These things have to be figured out. I just want doctors to be leading the charge here and not have this imposed by bureaucrats.
Dr. Glatter: I was reading that some doctors have had these exams administered and they can really pass cognitive aspects of the exam, but there have been nuances in the actual practicing of medicine, day-to-day functioning, which they’re not good at.
Someone made a comment that the only way to know if a doctor can do well in practice is to observe their practice and observe them taking care of patients. In other words, you can game the system and pass the cognitive exam in some form but then have a problem practicing medicine.
Dr. Jauhar: Ultimately, outcomes have to be measured. We can’t adopt such a granular approach for every aging physician. There has to be some sort of screening that maybe raises a red flag and then hospitals and department chairs need to investigate further. What are the outcomes? What are people saying in the operating room? I think the screening is just that; it’s a way of opening the door to further investigation, but it’s not a witch hunt.
I have the highest respect for older physicians, and I learn from them every day, honestly, especially in my field (cardiology), because some of the older physicians can hear and see things on physical exam that I didn’t even know existed. There’s much to be learned from them.
This is not intended to be a witch hunt or to try to get rid of older physicians – by any means. We want to avoid some of the outcomes that I read about in the New York Times comments section. It’s not fair to our patients not to do at least some sort of screening to prevent those kinds of mistakes.
Dr. Glatter: I wanted to go back to data from Yale between October 2016 and January 2019, where 141 Yale clinicians who ranged in age from 69 to 92 years completed cognitive assessments. Of those, 18 clinicians, or about 13% of those tested, demonstrated cognitive deficits that were “deemed likely to impair their ability to practice medicine independently.” That’s telling. These are subtleties, but they’re important to identify. I would love to get your comment on that.
Dr. Jauhar: It’s in keeping with what we know about the proportion of our older citizens who have cognitive impairment. About 10% have dementia and about 20% have at least mild cognitive impairment. That’s in keeping with what we know, and this was a general screening.
There are certain programs, like in San Diego, for example, where physicians are referred, and so there’s a selection bias. But this was just general screening. It’s worrisome. I’m an aging physician myself. I want fairness in this process because I’m going to be assessed as well.
I just don’t really understand yet why there’s so much circling of the wagons and so much resistance. It seems like it would be good for physicians also to be removed from situations where they might get into potential litigation because of mistakes and physical or visual impairment. It seems like it’d be good for patients and physicians alike.
Dr. Glatter: It’s difficult to give up your profession, change fields, or become administrative at some point, and [decide] when to make that transition. As we all get older, we’re not going to have the ability to do what we did in our 20s, 30s, and so forth.
Dr. Jauhar: Much of the resistance is coming from doctors who are used to high levels of autonomy. I’m certainly sympathetic to that because I don’t want anyone telling me how to practice. The reason this is coming up and hasn’t come up in the past is not because of loss of autonomy but because of an actual demographic change. Many physicians were trained in the 1960s, ’70s, or ’80s. They’re getting to retirement age but they’re not retiring, and we can speculate as to why that is.
In America’s educational system, doctors incur a huge amount of debt. I know physicians who are still paying off their debt and they’re in their 50s and 60s, so I’m very sympathetic to that. I’m not trying to force doctors out of practicing. I just want whoever is practicing to be competent and to practice safely. We have to figure out how to do that.
Dr. Glatter: The fact that there is a shortage of physicians forecast in the next 10-15 years makes many physicians reluctant to retire. They feel like they want to be part of that support network and we don’t want to have a dire situation, especially in the rural areas. We’re not immune from aging. We’re human beings. We all have to realize that.
Dr. Jauhar: I know that the ACC is starting to debate this issue, in part because of my op-ed. My hope is that it will start a conversation and we will institute a plan that comes from physicians and serves our patients, and doesn’t serve some cottage industry of testing or serve the needs of insurers or bureaucrats. It has to serve the doctor-patient relationship.
Dr. Glatter: In some random surveys that I’ve read, up to 30%-40% of physicians do support some type of age-based screening or competency assessment. The needle’s moving. It’s just not there yet. I think that wider adoption is coming.
Dr. Jauhar: Data are coming as more hospitals start to adopt these late practitioner programs. Some of the data that came out of Yale, for example, are very important. We’re going to see more published data in this area, and it will clarify what we need to do and how big the problem is.
Dr. Glatter: I want to thank you again for your time and for writing the op-ed because it certainly was well read and opened the eyes of not only physicians, but also the public at large. It’s a conversation that has to be had. Thank you for doing this.
Dr. Jauhar: Thanks for inviting me, Robert. It was a pleasure to talk to you.
Dr. Glatter is assistant professor of emergency medicine, department of emergency medicine, at Hofstra University, Hempstead, N.Y. Dr. Jauhar is director of the heart failure program, Long Island Jewish Medical Center, New Hyde Park, N.Y. Neither Dr. Glatter nor Dr. Jauhar reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert Glatter, medical advisor for Medscape Emergency Medicine. Joining me today is Sandeep Jauhar, a practicing cardiologist and professor of medicine at Northwell Health, a frequent New York Times op-ed contributor, and highly regarded author of the upcoming book “My Father’s Brain: Life in the Shadow of Alzheimer’s.”
Sandeep Jauhar, MD: Thanks for having me.
Dr. Glatter: Your recent op-ed piece in the New York Times caught my eye. In your piece, you refer to a 2020 survey in which almost one-third of licensed doctors in the United States were 60 years of age or older, up from a quarter in 2010. You also state that, due to a 20% prevalence of mild cognitive impairment in persons older than 65, practicing physicians above this age should probably be screened by a battery of tests to ensure that their reasoning and cognitive abilities are intact. The title of the article is “How Would You Feel About a 100-Year-Old Doctor?”
How would you envision such a process? What aspects of day-to-day functioning would the exams truly be evaluating?
Dr. Jauhar: A significant number of people over 65 have measurable cognitive impairment. By cognitive impairment, we’re not talking about dementia. The best estimates are that 1 in 10 people over age 65 have dementia, and roughly 1 in 5 have what’s called MCI, or mild cognitive impairment, which is cognitive impairment out of proportion to what you’d expect from normal aging. It’s a significant issue.
The argument that I made in the op-ed is that neurocognitive assessment is important. That’s not to say that everyone over age 65 has significant cognitive impairment or that older doctors can’t practice medicine safely and effectively. They absolutely can. The question is, do we leave neurocognitive assessment to physicians who may possibly be suffering from impairment?
In dementia, people very often have impaired self-awareness, a condition called anosognosia, which is a neurological term for not being aware of your own impairment because of your impairment.
I would argue that, instead of having voluntary neurocognitive screening, it should be mandated. The question is how to do that effectively, fairly, and transparently.
One could argue a gerontocracy in medicine today, where there are so many older physicians. What do we do about that? That really is something that I think needs to be debated.
Dr. Glatter: The question I have is, if we (that is, physicians and the health care profession) don’t take care of this, someone’s going to do it for us. We need to jump on this now while we have the opportunity. The AMA has been opposed to this, except when you have reason to suspect cognitive decline or are concerned about patient safety. A mandatory age of retirement is certainly something they’re not for, and we know this.
Your argument in your op-ed piece is very well thought out, and you lay the groundwork for testing (looking at someone’s memory, coordination, processing speed, and other executive functions). Certainly, for a psychiatrist, hearing is important, and for a dermatologist, vision is important. For a surgeon, there are other issues. Based on the specialty, we must be careful to see the important aspects of functioning. I am sure you would agree with this.
Dr. Jauhar: Obviously, the hand skills that are important for ophthalmological surgery certainly aren’t required for office-based psychological counseling, for example. We have to be smart about how we assess impairment.
You describe the spectrum of actions. On the one hand, there’s mandatory retirement at the age of 65 or 70 years. We know that commercial pilots are mandated to essentially retire at 65, and air-traffic controllers must retire in their late 50s.
We know that there’s a large amount of variability in competence. There are internists in their 80s with whom I’ve worked, and I’m absolutely wowed by their experience and judgment. There are new medical resident graduates who don’t really seem to have the requisite level of competence that would make me feel comfortable to have them as my doctor or a doctor for a member of my family.
To mandate retirement, I think the AMA is absolutely right. To not call for any kind of competency testing, to me, seems equally unwise. Because at the end of the day, you have to balance individual physician needs or wants to continue practicing with patient safety. I haven’t really come across too many physicians who say, “There’s absolutely no need for a competency testing.”
We have to meet somewhere in the middle. The middle is either voluntary cognitive competency testing or mandatory. I would argue that, because we know that as the brain changes we have cognitive impairment, but we’re not always aware that we need help, mandatory testing is the way.
One other thing that you mentioned was about having the solution imposed on us. You and I are doctors. We deal with bureaucracy. We deal with poorly thought-out solutions to issues in health care that make our lives that much more difficult. I don’t want that solution imposed on us by some outside agency. I think we need to figure this out within medicine and figure out the right way of doing it.
The AMA is on board with this. They haven’t called for mandatory testing, but they have said that if testing were to occur, these are the guidelines. The guidelines are fair and equitable, not too time-consuming, transparent, and not punitive. If someone comes out and doesn’t test well, we shouldn’t force them out of the profession. We can find ways to use their experience to help train younger doctors, for example.
Dr. Glatter: I wanted to segue to an area where there has been some challenge to the legality of these mandatory types of age restrictions and imposing the exams as well. There’s been a lawsuit as well by the EEOC [Equal Employment Opportunity Commission], on behalf of Yale. Basically, there’s been a concern that ageism is part of what’s going on. Yale now screens their providers beginning at age 70, and they have a program. UCSD [University of California, San Diego] has a program in place. Obviously, these institutions are looking at it. This is a very small part of the overall picture.
Health care systems overall, we’re talking about a fraction of them in the country are really addressing the issue of competency exams. The question is, where do we go from here? How do we get engagement or adoption and get physicians as a whole to embrace this concept?
Dr. Jauhar: The EEOC filed a lawsuit on behalf of the Yale medical staff that argued that Yale’s plan to do vision testing and neurocognitive screening – there may be a physical exam also – constitutes age discrimination because it’s reserved for doctors over the age of 70. Those are the physicians who are most likely to have cognitive impairment.
We have rules already for impaired physicians who are, for example, addicted to illicit drugs or have alcohol abuse. We already have some of those measures in place. This is focused on cognitive impairment in aging physicians because cognitive impairment is an issue that arises with aging. We have to be clear about that.
Most younger physicians will not have measurable cognitive impairment that would impair their ability to practice. To force young physicians (for example, physicians in their forties) to undergo such screening, all in the name of preventing age discrimination, doesn’t strike me as being a good use of resources. They’re more likely to be false positives, as you know from Bayesian statistics. When you have low pretest probability, you’re more likely to get false positives.
How are we going to screen hundreds of thousands of physicians? We have to make a choice about the group that really is more likely to benefit from such screening. Very few hospitals are addressing this issue and it’s going to become more important.
Dr. Glatter: Surgeons have been particularly active in pushing for age-based screening. In 2016, the American College of Surgeons started making surgeons at age 65-70 undergo voluntary health and neurocognitive assessments, and encouraged physicians to disclose any concerning findings as part of their professional obligation, which is pretty impressive in my mind.
Surgeons’ skill set is quite demanding physically and technically. That the Society of Surgical Chairs took it upon themselves to institute this is pretty telling.
Dr. Jauhar: The overall society called for screening, but then in a separate survey of surgical chairs, the idea was advanced that we should have mandatory retirement. Now, I don’t particularly agree with that.
I’ve seen it, where you have the aging surgeon who was a star in their day, and no one wants to say anything when their skills have visibly degraded, and no one wants to carry that torch and tell them that they need to retire. What happens is people whisper, and unfortunately, bad outcomes have to occur before people tend to get involved, and that’s what I’m trying to prevent.
Dr. Glatter: The question is whether older physicians have worse patient outcomes. The evidence is inconclusive, but studies have shown higher mortality rates for cardiovascular surgeons in terms of the procedures that they do. On the flip side, there are also higher mortality rates for GI surgery performed by younger surgeons. It’s a mixed bag.
Dr. Jauhar: For specialized surgery, you need the accrual of a certain amount of experience. The optimal age is about 60, because they’ve seen many things and they’ve seen complications. They don’t have a hand tremor yet so they’re still functioning well, and they’ve accrued a lot of experience. We have to be smart about who we screen.
There’s a learning curve in surgery. By no means am I arguing that younger surgeons are better surgeons. I would say that there’s probably a tipping point where once you get past a certain age and physical deterioration starts to take effect, that can overshadow the accrual of cognitive and surgical experience. We have to balance those things.
I would say neurocognitive screening and vision testing are important, but exactly what do you measure? How much of a hand tremor would constitute a risk? These things have to be figured out. I just want doctors to be leading the charge here and not have this imposed by bureaucrats.
Dr. Glatter: I was reading that some doctors have had these exams administered and they can really pass cognitive aspects of the exam, but there have been nuances in the actual practicing of medicine, day-to-day functioning, which they’re not good at.
Someone made a comment that the only way to know if a doctor can do well in practice is to observe their practice and observe them taking care of patients. In other words, you can game the system and pass the cognitive exam in some form but then have a problem practicing medicine.
Dr. Jauhar: Ultimately, outcomes have to be measured. We can’t adopt such a granular approach for every aging physician. There has to be some sort of screening that maybe raises a red flag and then hospitals and department chairs need to investigate further. What are the outcomes? What are people saying in the operating room? I think the screening is just that; it’s a way of opening the door to further investigation, but it’s not a witch hunt.
I have the highest respect for older physicians, and I learn from them every day, honestly, especially in my field (cardiology), because some of the older physicians can hear and see things on physical exam that I didn’t even know existed. There’s much to be learned from them.
This is not intended to be a witch hunt or to try to get rid of older physicians – by any means. We want to avoid some of the outcomes that I read about in the New York Times comments section. It’s not fair to our patients not to do at least some sort of screening to prevent those kinds of mistakes.
Dr. Glatter: I wanted to go back to data from Yale between October 2016 and January 2019, where 141 Yale clinicians who ranged in age from 69 to 92 years completed cognitive assessments. Of those, 18 clinicians, or about 13% of those tested, demonstrated cognitive deficits that were “deemed likely to impair their ability to practice medicine independently.” That’s telling. These are subtleties, but they’re important to identify. I would love to get your comment on that.
Dr. Jauhar: It’s in keeping with what we know about the proportion of our older citizens who have cognitive impairment. About 10% have dementia and about 20% have at least mild cognitive impairment. That’s in keeping with what we know, and this was a general screening.
There are certain programs, like in San Diego, for example, where physicians are referred, and so there’s a selection bias. But this was just general screening. It’s worrisome. I’m an aging physician myself. I want fairness in this process because I’m going to be assessed as well.
I just don’t really understand yet why there’s so much circling of the wagons and so much resistance. It seems like it would be good for physicians also to be removed from situations where they might get into potential litigation because of mistakes and physical or visual impairment. It seems like it’d be good for patients and physicians alike.
Dr. Glatter: It’s difficult to give up your profession, change fields, or become administrative at some point, and [decide] when to make that transition. As we all get older, we’re not going to have the ability to do what we did in our 20s, 30s, and so forth.
Dr. Jauhar: Much of the resistance is coming from doctors who are used to high levels of autonomy. I’m certainly sympathetic to that because I don’t want anyone telling me how to practice. The reason this is coming up and hasn’t come up in the past is not because of loss of autonomy but because of an actual demographic change. Many physicians were trained in the 1960s, ’70s, or ’80s. They’re getting to retirement age but they’re not retiring, and we can speculate as to why that is.
In America’s educational system, doctors incur a huge amount of debt. I know physicians who are still paying off their debt and they’re in their 50s and 60s, so I’m very sympathetic to that. I’m not trying to force doctors out of practicing. I just want whoever is practicing to be competent and to practice safely. We have to figure out how to do that.
Dr. Glatter: The fact that there is a shortage of physicians forecast in the next 10-15 years makes many physicians reluctant to retire. They feel like they want to be part of that support network and we don’t want to have a dire situation, especially in the rural areas. We’re not immune from aging. We’re human beings. We all have to realize that.
Dr. Jauhar: I know that the ACC is starting to debate this issue, in part because of my op-ed. My hope is that it will start a conversation and we will institute a plan that comes from physicians and serves our patients, and doesn’t serve some cottage industry of testing or serve the needs of insurers or bureaucrats. It has to serve the doctor-patient relationship.
Dr. Glatter: In some random surveys that I’ve read, up to 30%-40% of physicians do support some type of age-based screening or competency assessment. The needle’s moving. It’s just not there yet. I think that wider adoption is coming.
Dr. Jauhar: Data are coming as more hospitals start to adopt these late practitioner programs. Some of the data that came out of Yale, for example, are very important. We’re going to see more published data in this area, and it will clarify what we need to do and how big the problem is.
Dr. Glatter: I want to thank you again for your time and for writing the op-ed because it certainly was well read and opened the eyes of not only physicians, but also the public at large. It’s a conversation that has to be had. Thank you for doing this.
Dr. Jauhar: Thanks for inviting me, Robert. It was a pleasure to talk to you.
Dr. Glatter is assistant professor of emergency medicine, department of emergency medicine, at Hofstra University, Hempstead, N.Y. Dr. Jauhar is director of the heart failure program, Long Island Jewish Medical Center, New Hyde Park, N.Y. Neither Dr. Glatter nor Dr. Jauhar reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert Glatter, medical advisor for Medscape Emergency Medicine. Joining me today is Sandeep Jauhar, a practicing cardiologist and professor of medicine at Northwell Health, a frequent New York Times op-ed contributor, and highly regarded author of the upcoming book “My Father’s Brain: Life in the Shadow of Alzheimer’s.”
Sandeep Jauhar, MD: Thanks for having me.
Dr. Glatter: Your recent op-ed piece in the New York Times caught my eye. In your piece, you refer to a 2020 survey in which almost one-third of licensed doctors in the United States were 60 years of age or older, up from a quarter in 2010. You also state that, due to a 20% prevalence of mild cognitive impairment in persons older than 65, practicing physicians above this age should probably be screened by a battery of tests to ensure that their reasoning and cognitive abilities are intact. The title of the article is “How Would You Feel About a 100-Year-Old Doctor?”
How would you envision such a process? What aspects of day-to-day functioning would the exams truly be evaluating?
Dr. Jauhar: A significant number of people over 65 have measurable cognitive impairment. By cognitive impairment, we’re not talking about dementia. The best estimates are that 1 in 10 people over age 65 have dementia, and roughly 1 in 5 have what’s called MCI, or mild cognitive impairment, which is cognitive impairment out of proportion to what you’d expect from normal aging. It’s a significant issue.
The argument that I made in the op-ed is that neurocognitive assessment is important. That’s not to say that everyone over age 65 has significant cognitive impairment or that older doctors can’t practice medicine safely and effectively. They absolutely can. The question is, do we leave neurocognitive assessment to physicians who may possibly be suffering from impairment?
In dementia, people very often have impaired self-awareness, a condition called anosognosia, which is a neurological term for not being aware of your own impairment because of your impairment.
I would argue that, instead of having voluntary neurocognitive screening, it should be mandated. The question is how to do that effectively, fairly, and transparently.
One could argue a gerontocracy in medicine today, where there are so many older physicians. What do we do about that? That really is something that I think needs to be debated.
Dr. Glatter: The question I have is, if we (that is, physicians and the health care profession) don’t take care of this, someone’s going to do it for us. We need to jump on this now while we have the opportunity. The AMA has been opposed to this, except when you have reason to suspect cognitive decline or are concerned about patient safety. A mandatory age of retirement is certainly something they’re not for, and we know this.
Your argument in your op-ed piece is very well thought out, and you lay the groundwork for testing (looking at someone’s memory, coordination, processing speed, and other executive functions). Certainly, for a psychiatrist, hearing is important, and for a dermatologist, vision is important. For a surgeon, there are other issues. Based on the specialty, we must be careful to see the important aspects of functioning. I am sure you would agree with this.
Dr. Jauhar: Obviously, the hand skills that are important for ophthalmological surgery certainly aren’t required for office-based psychological counseling, for example. We have to be smart about how we assess impairment.
You describe the spectrum of actions. On the one hand, there’s mandatory retirement at the age of 65 or 70 years. We know that commercial pilots are mandated to essentially retire at 65, and air-traffic controllers must retire in their late 50s.
We know that there’s a large amount of variability in competence. There are internists in their 80s with whom I’ve worked, and I’m absolutely wowed by their experience and judgment. There are new medical resident graduates who don’t really seem to have the requisite level of competence that would make me feel comfortable to have them as my doctor or a doctor for a member of my family.
To mandate retirement, I think the AMA is absolutely right. To not call for any kind of competency testing, to me, seems equally unwise. Because at the end of the day, you have to balance individual physician needs or wants to continue practicing with patient safety. I haven’t really come across too many physicians who say, “There’s absolutely no need for a competency testing.”
We have to meet somewhere in the middle. The middle is either voluntary cognitive competency testing or mandatory. I would argue that, because we know that as the brain changes we have cognitive impairment, but we’re not always aware that we need help, mandatory testing is the way.
One other thing that you mentioned was about having the solution imposed on us. You and I are doctors. We deal with bureaucracy. We deal with poorly thought-out solutions to issues in health care that make our lives that much more difficult. I don’t want that solution imposed on us by some outside agency. I think we need to figure this out within medicine and figure out the right way of doing it.
The AMA is on board with this. They haven’t called for mandatory testing, but they have said that if testing were to occur, these are the guidelines. The guidelines are fair and equitable, not too time-consuming, transparent, and not punitive. If someone comes out and doesn’t test well, we shouldn’t force them out of the profession. We can find ways to use their experience to help train younger doctors, for example.
Dr. Glatter: I wanted to segue to an area where there has been some challenge to the legality of these mandatory types of age restrictions and imposing the exams as well. There’s been a lawsuit as well by the EEOC [Equal Employment Opportunity Commission], on behalf of Yale. Basically, there’s been a concern that ageism is part of what’s going on. Yale now screens their providers beginning at age 70, and they have a program. UCSD [University of California, San Diego] has a program in place. Obviously, these institutions are looking at it. This is a very small part of the overall picture.
Health care systems overall, we’re talking about a fraction of them in the country are really addressing the issue of competency exams. The question is, where do we go from here? How do we get engagement or adoption and get physicians as a whole to embrace this concept?
Dr. Jauhar: The EEOC filed a lawsuit on behalf of the Yale medical staff that argued that Yale’s plan to do vision testing and neurocognitive screening – there may be a physical exam also – constitutes age discrimination because it’s reserved for doctors over the age of 70. Those are the physicians who are most likely to have cognitive impairment.
We have rules already for impaired physicians who are, for example, addicted to illicit drugs or have alcohol abuse. We already have some of those measures in place. This is focused on cognitive impairment in aging physicians because cognitive impairment is an issue that arises with aging. We have to be clear about that.
Most younger physicians will not have measurable cognitive impairment that would impair their ability to practice. To force young physicians (for example, physicians in their forties) to undergo such screening, all in the name of preventing age discrimination, doesn’t strike me as being a good use of resources. They’re more likely to be false positives, as you know from Bayesian statistics. When you have low pretest probability, you’re more likely to get false positives.
How are we going to screen hundreds of thousands of physicians? We have to make a choice about the group that really is more likely to benefit from such screening. Very few hospitals are addressing this issue and it’s going to become more important.
Dr. Glatter: Surgeons have been particularly active in pushing for age-based screening. In 2016, the American College of Surgeons started making surgeons at age 65-70 undergo voluntary health and neurocognitive assessments, and encouraged physicians to disclose any concerning findings as part of their professional obligation, which is pretty impressive in my mind.
Surgeons’ skill set is quite demanding physically and technically. That the Society of Surgical Chairs took it upon themselves to institute this is pretty telling.
Dr. Jauhar: The overall society called for screening, but then in a separate survey of surgical chairs, the idea was advanced that we should have mandatory retirement. Now, I don’t particularly agree with that.
I’ve seen it, where you have the aging surgeon who was a star in their day, and no one wants to say anything when their skills have visibly degraded, and no one wants to carry that torch and tell them that they need to retire. What happens is people whisper, and unfortunately, bad outcomes have to occur before people tend to get involved, and that’s what I’m trying to prevent.
Dr. Glatter: The question is whether older physicians have worse patient outcomes. The evidence is inconclusive, but studies have shown higher mortality rates for cardiovascular surgeons in terms of the procedures that they do. On the flip side, there are also higher mortality rates for GI surgery performed by younger surgeons. It’s a mixed bag.
Dr. Jauhar: For specialized surgery, you need the accrual of a certain amount of experience. The optimal age is about 60, because they’ve seen many things and they’ve seen complications. They don’t have a hand tremor yet so they’re still functioning well, and they’ve accrued a lot of experience. We have to be smart about who we screen.
There’s a learning curve in surgery. By no means am I arguing that younger surgeons are better surgeons. I would say that there’s probably a tipping point where once you get past a certain age and physical deterioration starts to take effect, that can overshadow the accrual of cognitive and surgical experience. We have to balance those things.
I would say neurocognitive screening and vision testing are important, but exactly what do you measure? How much of a hand tremor would constitute a risk? These things have to be figured out. I just want doctors to be leading the charge here and not have this imposed by bureaucrats.
Dr. Glatter: I was reading that some doctors have had these exams administered and they can really pass cognitive aspects of the exam, but there have been nuances in the actual practicing of medicine, day-to-day functioning, which they’re not good at.
Someone made a comment that the only way to know if a doctor can do well in practice is to observe their practice and observe them taking care of patients. In other words, you can game the system and pass the cognitive exam in some form but then have a problem practicing medicine.
Dr. Jauhar: Ultimately, outcomes have to be measured. We can’t adopt such a granular approach for every aging physician. There has to be some sort of screening that maybe raises a red flag and then hospitals and department chairs need to investigate further. What are the outcomes? What are people saying in the operating room? I think the screening is just that; it’s a way of opening the door to further investigation, but it’s not a witch hunt.
I have the highest respect for older physicians, and I learn from them every day, honestly, especially in my field (cardiology), because some of the older physicians can hear and see things on physical exam that I didn’t even know existed. There’s much to be learned from them.
This is not intended to be a witch hunt or to try to get rid of older physicians – by any means. We want to avoid some of the outcomes that I read about in the New York Times comments section. It’s not fair to our patients not to do at least some sort of screening to prevent those kinds of mistakes.
Dr. Glatter: I wanted to go back to data from Yale between October 2016 and January 2019, where 141 Yale clinicians who ranged in age from 69 to 92 years completed cognitive assessments. Of those, 18 clinicians, or about 13% of those tested, demonstrated cognitive deficits that were “deemed likely to impair their ability to practice medicine independently.” That’s telling. These are subtleties, but they’re important to identify. I would love to get your comment on that.
Dr. Jauhar: It’s in keeping with what we know about the proportion of our older citizens who have cognitive impairment. About 10% have dementia and about 20% have at least mild cognitive impairment. That’s in keeping with what we know, and this was a general screening.
There are certain programs, like in San Diego, for example, where physicians are referred, and so there’s a selection bias. But this was just general screening. It’s worrisome. I’m an aging physician myself. I want fairness in this process because I’m going to be assessed as well.
I just don’t really understand yet why there’s so much circling of the wagons and so much resistance. It seems like it would be good for physicians also to be removed from situations where they might get into potential litigation because of mistakes and physical or visual impairment. It seems like it’d be good for patients and physicians alike.
Dr. Glatter: It’s difficult to give up your profession, change fields, or become administrative at some point, and [decide] when to make that transition. As we all get older, we’re not going to have the ability to do what we did in our 20s, 30s, and so forth.
Dr. Jauhar: Much of the resistance is coming from doctors who are used to high levels of autonomy. I’m certainly sympathetic to that because I don’t want anyone telling me how to practice. The reason this is coming up and hasn’t come up in the past is not because of loss of autonomy but because of an actual demographic change. Many physicians were trained in the 1960s, ’70s, or ’80s. They’re getting to retirement age but they’re not retiring, and we can speculate as to why that is.
In America’s educational system, doctors incur a huge amount of debt. I know physicians who are still paying off their debt and they’re in their 50s and 60s, so I’m very sympathetic to that. I’m not trying to force doctors out of practicing. I just want whoever is practicing to be competent and to practice safely. We have to figure out how to do that.
Dr. Glatter: The fact that there is a shortage of physicians forecast in the next 10-15 years makes many physicians reluctant to retire. They feel like they want to be part of that support network and we don’t want to have a dire situation, especially in the rural areas. We’re not immune from aging. We’re human beings. We all have to realize that.
Dr. Jauhar: I know that the ACC is starting to debate this issue, in part because of my op-ed. My hope is that it will start a conversation and we will institute a plan that comes from physicians and serves our patients, and doesn’t serve some cottage industry of testing or serve the needs of insurers or bureaucrats. It has to serve the doctor-patient relationship.
Dr. Glatter: In some random surveys that I’ve read, up to 30%-40% of physicians do support some type of age-based screening or competency assessment. The needle’s moving. It’s just not there yet. I think that wider adoption is coming.
Dr. Jauhar: Data are coming as more hospitals start to adopt these late practitioner programs. Some of the data that came out of Yale, for example, are very important. We’re going to see more published data in this area, and it will clarify what we need to do and how big the problem is.
Dr. Glatter: I want to thank you again for your time and for writing the op-ed because it certainly was well read and opened the eyes of not only physicians, but also the public at large. It’s a conversation that has to be had. Thank you for doing this.
Dr. Jauhar: Thanks for inviting me, Robert. It was a pleasure to talk to you.
Dr. Glatter is assistant professor of emergency medicine, department of emergency medicine, at Hofstra University, Hempstead, N.Y. Dr. Jauhar is director of the heart failure program, Long Island Jewish Medical Center, New Hyde Park, N.Y. Neither Dr. Glatter nor Dr. Jauhar reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Chronic pain patients swapping opioids for medical cannabis
new research shows.
“That patients report substituting cannabis for pain medicines so much really underscores the need for research on the benefits and risks of using cannabis for chronic pain,” lead author Mark C. Bicket, MD, PhD, assistant professor, department of anesthesiology, and director, Opioid Prescribing Engagement Network, University of Michigan, Ann Arbor, said in an interview.
However, he added, the question is whether they’re turning to cannabis and away from other pain treatments. “What’s not clear and one of the gaps that we wanted to address in the study was if medical cannabis use is changing the use of other treatments for chronic pain,” said Dr. Bicket.
The study was published online in JAMA Network Open.
Decreased opioid use
The survey included a representative sample of 1724 American adults aged 18 years or older with chronic noncancer pain living in areas with a medical cannabis program.
Respondents were asked about their use of three categories of pain treatments. This included medical cannabis; pharmacologic treatments including prescription opioids, nonopioid analgesics, and over-the-counter analgesics; and common nonpharmacologic treatments such as physical therapy, meditation, and cognitive-behavioral therapy (CBT).
Just over 96% of respondents completed the full survey. About 57% of the sample was female and the mean age of the study sample was 52.3 years.
Among study participants, 31% (95% CI, 28.2% - 34.1%) reported having ever used cannabis to manage pain; 25.9% (95% confidence interval, 23.2%-28.8%) reported use in the past 12 months, and 23.2% (95% CI, 20.6%-26%) reported use in the past 30 days.
“This translates into a large number of individuals who are using cannabis in an intended medical way” to treat chronic condition such as low back pain, migraine, and fibromyalgia, said Dr. Bicket.
More than half of survey respondents reported their medical cannabis use led to a decrease in prescription opioid use, prescription nonopioid use and use of over-the-counter medications.
Dr. Bicket noted “almost no one” said medical cannabis use led to higher use of these drugs.
As for nonpharmacologic treatments, 38.7% reported their use of cannabis led to decreased use of physical therapy, 19.1% to lower use of meditation, and 26% to less CBT. At the same time, 5.9%, 23.7% and 17.1%, respectively, reported it led to increased use of physical therapy, meditation, and CBT.
Medical cannabis is regulated at a state level. On a federal level, it’s considered a Schedule I substance, which means it’s deemed not to have a therapeutic use, although some groups are trying to change that categorization, said Dr. Bicket.
As a result, cannabis products “are quite variable” in terms of how they’re used (smoked, eaten etc.) and in their composition, including percentage of cannabidiol and tetrahydrocannabinol.
“We really don’t have a good sense of the relative risks and benefits that could come from cannabis as a treatment for chronic pain,” said Dr. Bicket. “As a physician, it’s difficult to have discussions with patients because I’m not able to understand the products they’re using based on this regulatory environment we have.”
He added clinicians “are operating in an area of uncertainty right now.”
What’s needed is research to determine how safe and effective medical cannabis is for chronic pain, he said.
Pain a leading indication
Commenting on the findings, Jason W. Busse, PhD, professor, department of anesthesia, and associate director, Centre for Medicinal Cannabis Research, McMaster University, Hamilton, Ont., said the study reinforces results of some prior research.
“It gives us current information certainly highlighting the high rate of use of medical cannabis among individuals with chronic pain once it becomes legally available.”
In addition, this high rate of use “means we desperately need information about the benefits and harms” of medical marijuana, he said.
Dr. Busse noted the survey didn’t provide information on the types of cannabis being used or the mode of administration. Oil drops and sprays cause less pulmonary harm than smoked versions, he said. It’s also not clear from the survey if participants are taking formulations with high levels of tetrahydrocannabinol that are associated with greater risk of harm.
He noted cannabis may interact with prescription drugs to make them less effective or, in some cases, to augment their adverse effects.
Dr. Busse pointed out some patients could be using fewer opioids because providers are under “enormous pressure” to reduce prescriptions of these drugs in the wake of spikes in opioid overdoses and deaths.
Chronic pain is “absolutely the leading indication” for medical marijuana, said Dr. Busse. U.S. reimbursement data suggest up to 65% of individuals get cannabis to treat a listed indication for chronic pain.
He said he hopes this new study will increase interest in funding new trials “so we can have better evidence to guide practice to help patients make decisions.”
The study received support from the National Institute on Drug Abuse. Dr. Bicket reported receiving personal fees from Axial Healthcare as well as grants from the National Institutes of Health, the Centers for Disease Control and Prevention, Michigan Department of Health and Human Services, Arnold Foundation, and the Patient-Centered Outcomes Research Institute outside the submitted work. Dr. Busse reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research shows.
“That patients report substituting cannabis for pain medicines so much really underscores the need for research on the benefits and risks of using cannabis for chronic pain,” lead author Mark C. Bicket, MD, PhD, assistant professor, department of anesthesiology, and director, Opioid Prescribing Engagement Network, University of Michigan, Ann Arbor, said in an interview.
However, he added, the question is whether they’re turning to cannabis and away from other pain treatments. “What’s not clear and one of the gaps that we wanted to address in the study was if medical cannabis use is changing the use of other treatments for chronic pain,” said Dr. Bicket.
The study was published online in JAMA Network Open.
Decreased opioid use
The survey included a representative sample of 1724 American adults aged 18 years or older with chronic noncancer pain living in areas with a medical cannabis program.
Respondents were asked about their use of three categories of pain treatments. This included medical cannabis; pharmacologic treatments including prescription opioids, nonopioid analgesics, and over-the-counter analgesics; and common nonpharmacologic treatments such as physical therapy, meditation, and cognitive-behavioral therapy (CBT).
Just over 96% of respondents completed the full survey. About 57% of the sample was female and the mean age of the study sample was 52.3 years.
Among study participants, 31% (95% CI, 28.2% - 34.1%) reported having ever used cannabis to manage pain; 25.9% (95% confidence interval, 23.2%-28.8%) reported use in the past 12 months, and 23.2% (95% CI, 20.6%-26%) reported use in the past 30 days.
“This translates into a large number of individuals who are using cannabis in an intended medical way” to treat chronic condition such as low back pain, migraine, and fibromyalgia, said Dr. Bicket.
More than half of survey respondents reported their medical cannabis use led to a decrease in prescription opioid use, prescription nonopioid use and use of over-the-counter medications.
Dr. Bicket noted “almost no one” said medical cannabis use led to higher use of these drugs.
As for nonpharmacologic treatments, 38.7% reported their use of cannabis led to decreased use of physical therapy, 19.1% to lower use of meditation, and 26% to less CBT. At the same time, 5.9%, 23.7% and 17.1%, respectively, reported it led to increased use of physical therapy, meditation, and CBT.
Medical cannabis is regulated at a state level. On a federal level, it’s considered a Schedule I substance, which means it’s deemed not to have a therapeutic use, although some groups are trying to change that categorization, said Dr. Bicket.
As a result, cannabis products “are quite variable” in terms of how they’re used (smoked, eaten etc.) and in their composition, including percentage of cannabidiol and tetrahydrocannabinol.
“We really don’t have a good sense of the relative risks and benefits that could come from cannabis as a treatment for chronic pain,” said Dr. Bicket. “As a physician, it’s difficult to have discussions with patients because I’m not able to understand the products they’re using based on this regulatory environment we have.”
He added clinicians “are operating in an area of uncertainty right now.”
What’s needed is research to determine how safe and effective medical cannabis is for chronic pain, he said.
Pain a leading indication
Commenting on the findings, Jason W. Busse, PhD, professor, department of anesthesia, and associate director, Centre for Medicinal Cannabis Research, McMaster University, Hamilton, Ont., said the study reinforces results of some prior research.
“It gives us current information certainly highlighting the high rate of use of medical cannabis among individuals with chronic pain once it becomes legally available.”
In addition, this high rate of use “means we desperately need information about the benefits and harms” of medical marijuana, he said.
Dr. Busse noted the survey didn’t provide information on the types of cannabis being used or the mode of administration. Oil drops and sprays cause less pulmonary harm than smoked versions, he said. It’s also not clear from the survey if participants are taking formulations with high levels of tetrahydrocannabinol that are associated with greater risk of harm.
He noted cannabis may interact with prescription drugs to make them less effective or, in some cases, to augment their adverse effects.
Dr. Busse pointed out some patients could be using fewer opioids because providers are under “enormous pressure” to reduce prescriptions of these drugs in the wake of spikes in opioid overdoses and deaths.
Chronic pain is “absolutely the leading indication” for medical marijuana, said Dr. Busse. U.S. reimbursement data suggest up to 65% of individuals get cannabis to treat a listed indication for chronic pain.
He said he hopes this new study will increase interest in funding new trials “so we can have better evidence to guide practice to help patients make decisions.”
The study received support from the National Institute on Drug Abuse. Dr. Bicket reported receiving personal fees from Axial Healthcare as well as grants from the National Institutes of Health, the Centers for Disease Control and Prevention, Michigan Department of Health and Human Services, Arnold Foundation, and the Patient-Centered Outcomes Research Institute outside the submitted work. Dr. Busse reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research shows.
“That patients report substituting cannabis for pain medicines so much really underscores the need for research on the benefits and risks of using cannabis for chronic pain,” lead author Mark C. Bicket, MD, PhD, assistant professor, department of anesthesiology, and director, Opioid Prescribing Engagement Network, University of Michigan, Ann Arbor, said in an interview.
However, he added, the question is whether they’re turning to cannabis and away from other pain treatments. “What’s not clear and one of the gaps that we wanted to address in the study was if medical cannabis use is changing the use of other treatments for chronic pain,” said Dr. Bicket.
The study was published online in JAMA Network Open.
Decreased opioid use
The survey included a representative sample of 1724 American adults aged 18 years or older with chronic noncancer pain living in areas with a medical cannabis program.
Respondents were asked about their use of three categories of pain treatments. This included medical cannabis; pharmacologic treatments including prescription opioids, nonopioid analgesics, and over-the-counter analgesics; and common nonpharmacologic treatments such as physical therapy, meditation, and cognitive-behavioral therapy (CBT).
Just over 96% of respondents completed the full survey. About 57% of the sample was female and the mean age of the study sample was 52.3 years.
Among study participants, 31% (95% CI, 28.2% - 34.1%) reported having ever used cannabis to manage pain; 25.9% (95% confidence interval, 23.2%-28.8%) reported use in the past 12 months, and 23.2% (95% CI, 20.6%-26%) reported use in the past 30 days.
“This translates into a large number of individuals who are using cannabis in an intended medical way” to treat chronic condition such as low back pain, migraine, and fibromyalgia, said Dr. Bicket.
More than half of survey respondents reported their medical cannabis use led to a decrease in prescription opioid use, prescription nonopioid use and use of over-the-counter medications.
Dr. Bicket noted “almost no one” said medical cannabis use led to higher use of these drugs.
As for nonpharmacologic treatments, 38.7% reported their use of cannabis led to decreased use of physical therapy, 19.1% to lower use of meditation, and 26% to less CBT. At the same time, 5.9%, 23.7% and 17.1%, respectively, reported it led to increased use of physical therapy, meditation, and CBT.
Medical cannabis is regulated at a state level. On a federal level, it’s considered a Schedule I substance, which means it’s deemed not to have a therapeutic use, although some groups are trying to change that categorization, said Dr. Bicket.
As a result, cannabis products “are quite variable” in terms of how they’re used (smoked, eaten etc.) and in their composition, including percentage of cannabidiol and tetrahydrocannabinol.
“We really don’t have a good sense of the relative risks and benefits that could come from cannabis as a treatment for chronic pain,” said Dr. Bicket. “As a physician, it’s difficult to have discussions with patients because I’m not able to understand the products they’re using based on this regulatory environment we have.”
He added clinicians “are operating in an area of uncertainty right now.”
What’s needed is research to determine how safe and effective medical cannabis is for chronic pain, he said.
Pain a leading indication
Commenting on the findings, Jason W. Busse, PhD, professor, department of anesthesia, and associate director, Centre for Medicinal Cannabis Research, McMaster University, Hamilton, Ont., said the study reinforces results of some prior research.
“It gives us current information certainly highlighting the high rate of use of medical cannabis among individuals with chronic pain once it becomes legally available.”
In addition, this high rate of use “means we desperately need information about the benefits and harms” of medical marijuana, he said.
Dr. Busse noted the survey didn’t provide information on the types of cannabis being used or the mode of administration. Oil drops and sprays cause less pulmonary harm than smoked versions, he said. It’s also not clear from the survey if participants are taking formulations with high levels of tetrahydrocannabinol that are associated with greater risk of harm.
He noted cannabis may interact with prescription drugs to make them less effective or, in some cases, to augment their adverse effects.
Dr. Busse pointed out some patients could be using fewer opioids because providers are under “enormous pressure” to reduce prescriptions of these drugs in the wake of spikes in opioid overdoses and deaths.
Chronic pain is “absolutely the leading indication” for medical marijuana, said Dr. Busse. U.S. reimbursement data suggest up to 65% of individuals get cannabis to treat a listed indication for chronic pain.
He said he hopes this new study will increase interest in funding new trials “so we can have better evidence to guide practice to help patients make decisions.”
The study received support from the National Institute on Drug Abuse. Dr. Bicket reported receiving personal fees from Axial Healthcare as well as grants from the National Institutes of Health, the Centers for Disease Control and Prevention, Michigan Department of Health and Human Services, Arnold Foundation, and the Patient-Centered Outcomes Research Institute outside the submitted work. Dr. Busse reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
New Omicron subvariant is ‘crazy infectious,’ COVID expert warns
“It’s crazy infectious,” said Paula Cannon, PhD, a virologist at the University of Southern California, Los Angeles. “All the things that have protected you for the past couple of years, I don’t think are going to protect you against this new crop of variants.”
XBB.1.5 is spreading quickly in the United States. It accounted for 27.6% of cases in the country in the week ending on Jan. 7, up from about 1% of cases at one point in December, according to the Centers for Disease Control and Prevention. It’s especially prevalent in the Northeast, now accounting for more than 70% of the cases in that region.
It’s spreading across the globe, too. Maria Van Kerkhove, PhD, technical lead of the World Health Organization, has called XBB.1.5 is “the most transmissible subvariant that has been detected yet.”
Ashish Jha, MD, the White House COVID-19 response coordinator, tweeted a few days ago that the spread of XBB.1.5 is “stunning” but cautioned that it’s unclear if the symptoms of infection will be more severe than for previous variants.
“Whether we’ll have an XBB.1.5 wave (and if yes, how big) will depend on many factors including immunity of the population, people’s actions, etc.,” he tweeted.
He urged people to get up to date on their boosters, wear a snug-fitting mask, and avoid crowded indoor spaces. He noted that people who haven’t been infected recently or haven’t gotten the bivalent booster likely have little protection against infection.
The symptoms for XBB.1.5 appear to be the same as for other versions of COVID-19. However, it’s less common for people infected with XBB.1.5 to report losing their sense of taste and smell, USA Today reported.
A version of this article first appeared on WebMD.com.
“It’s crazy infectious,” said Paula Cannon, PhD, a virologist at the University of Southern California, Los Angeles. “All the things that have protected you for the past couple of years, I don’t think are going to protect you against this new crop of variants.”
XBB.1.5 is spreading quickly in the United States. It accounted for 27.6% of cases in the country in the week ending on Jan. 7, up from about 1% of cases at one point in December, according to the Centers for Disease Control and Prevention. It’s especially prevalent in the Northeast, now accounting for more than 70% of the cases in that region.
It’s spreading across the globe, too. Maria Van Kerkhove, PhD, technical lead of the World Health Organization, has called XBB.1.5 is “the most transmissible subvariant that has been detected yet.”
Ashish Jha, MD, the White House COVID-19 response coordinator, tweeted a few days ago that the spread of XBB.1.5 is “stunning” but cautioned that it’s unclear if the symptoms of infection will be more severe than for previous variants.
“Whether we’ll have an XBB.1.5 wave (and if yes, how big) will depend on many factors including immunity of the population, people’s actions, etc.,” he tweeted.
He urged people to get up to date on their boosters, wear a snug-fitting mask, and avoid crowded indoor spaces. He noted that people who haven’t been infected recently or haven’t gotten the bivalent booster likely have little protection against infection.
The symptoms for XBB.1.5 appear to be the same as for other versions of COVID-19. However, it’s less common for people infected with XBB.1.5 to report losing their sense of taste and smell, USA Today reported.
A version of this article first appeared on WebMD.com.
“It’s crazy infectious,” said Paula Cannon, PhD, a virologist at the University of Southern California, Los Angeles. “All the things that have protected you for the past couple of years, I don’t think are going to protect you against this new crop of variants.”
XBB.1.5 is spreading quickly in the United States. It accounted for 27.6% of cases in the country in the week ending on Jan. 7, up from about 1% of cases at one point in December, according to the Centers for Disease Control and Prevention. It’s especially prevalent in the Northeast, now accounting for more than 70% of the cases in that region.
It’s spreading across the globe, too. Maria Van Kerkhove, PhD, technical lead of the World Health Organization, has called XBB.1.5 is “the most transmissible subvariant that has been detected yet.”
Ashish Jha, MD, the White House COVID-19 response coordinator, tweeted a few days ago that the spread of XBB.1.5 is “stunning” but cautioned that it’s unclear if the symptoms of infection will be more severe than for previous variants.
“Whether we’ll have an XBB.1.5 wave (and if yes, how big) will depend on many factors including immunity of the population, people’s actions, etc.,” he tweeted.
He urged people to get up to date on their boosters, wear a snug-fitting mask, and avoid crowded indoor spaces. He noted that people who haven’t been infected recently or haven’t gotten the bivalent booster likely have little protection against infection.
The symptoms for XBB.1.5 appear to be the same as for other versions of COVID-19. However, it’s less common for people infected with XBB.1.5 to report losing their sense of taste and smell, USA Today reported.
A version of this article first appeared on WebMD.com.
New study offers details on post-COVID pediatric illness
Multisystem inflammatory syndrome in children (MIS-C) is more common than previously thought. This pediatric illness occurs 2-6 weeks after being infected with COVID-19.
a new study found. The illness is rare, but it causes dangerous multiorgan dysfunction and frequently requires a stay in the ICU. According to the Centers for Disease Control and Prevention, there have been at least 9,333 cases nationwide and 76 deaths from MIS-C.
Researchers said their findings were in such contrast to previous MIS-C research that it may render the old research “misleading.”
The analysis was powered by improved data extracted from hospital billing systems. Previous analyses of MIS-C were limited to voluntarily reported cases, which is likely the reason for the undercount.
The study reported a mortality rate for people with the most severe cases (affecting six to eight organs) of 5.8%. The authors of a companion editorial to the study said the mortality rate was low when considering the widespread impacts, “reflecting the rapid reversibility of MIS-C” with treatment.
Differences in MIS-C cases were also found based on children’s race and ethnicity. Black patients were more likely to have severe cases affecting more organs, compared to white patients.
The study included 4,107 MIS-C cases, using data from 2021 for patients younger than 21 years old. The median age was 9 years old.
The findings provide direction for further research, the editorial writers suggested.
Questions that need to be answered include asking why Black children with MIS-C are more likely to have a higher number of organ systems affected.
“Identifying patient biological or socioeconomic factors that can be targeted for treatment or prevention should be pursued,” they wrote.
The CDC says symptoms of MIS-C are an ongoing fever plus more than one of the following: stomach pain, bloodshot eyes, diarrhea, dizziness or lightheadedness (signs of low blood pressure), skin rash, or vomiting.
A version of this article first appeared on WebMD.com.
Multisystem inflammatory syndrome in children (MIS-C) is more common than previously thought. This pediatric illness occurs 2-6 weeks after being infected with COVID-19.
a new study found. The illness is rare, but it causes dangerous multiorgan dysfunction and frequently requires a stay in the ICU. According to the Centers for Disease Control and Prevention, there have been at least 9,333 cases nationwide and 76 deaths from MIS-C.
Researchers said their findings were in such contrast to previous MIS-C research that it may render the old research “misleading.”
The analysis was powered by improved data extracted from hospital billing systems. Previous analyses of MIS-C were limited to voluntarily reported cases, which is likely the reason for the undercount.
The study reported a mortality rate for people with the most severe cases (affecting six to eight organs) of 5.8%. The authors of a companion editorial to the study said the mortality rate was low when considering the widespread impacts, “reflecting the rapid reversibility of MIS-C” with treatment.
Differences in MIS-C cases were also found based on children’s race and ethnicity. Black patients were more likely to have severe cases affecting more organs, compared to white patients.
The study included 4,107 MIS-C cases, using data from 2021 for patients younger than 21 years old. The median age was 9 years old.
The findings provide direction for further research, the editorial writers suggested.
Questions that need to be answered include asking why Black children with MIS-C are more likely to have a higher number of organ systems affected.
“Identifying patient biological or socioeconomic factors that can be targeted for treatment or prevention should be pursued,” they wrote.
The CDC says symptoms of MIS-C are an ongoing fever plus more than one of the following: stomach pain, bloodshot eyes, diarrhea, dizziness or lightheadedness (signs of low blood pressure), skin rash, or vomiting.
A version of this article first appeared on WebMD.com.
Multisystem inflammatory syndrome in children (MIS-C) is more common than previously thought. This pediatric illness occurs 2-6 weeks after being infected with COVID-19.
a new study found. The illness is rare, but it causes dangerous multiorgan dysfunction and frequently requires a stay in the ICU. According to the Centers for Disease Control and Prevention, there have been at least 9,333 cases nationwide and 76 deaths from MIS-C.
Researchers said their findings were in such contrast to previous MIS-C research that it may render the old research “misleading.”
The analysis was powered by improved data extracted from hospital billing systems. Previous analyses of MIS-C were limited to voluntarily reported cases, which is likely the reason for the undercount.
The study reported a mortality rate for people with the most severe cases (affecting six to eight organs) of 5.8%. The authors of a companion editorial to the study said the mortality rate was low when considering the widespread impacts, “reflecting the rapid reversibility of MIS-C” with treatment.
Differences in MIS-C cases were also found based on children’s race and ethnicity. Black patients were more likely to have severe cases affecting more organs, compared to white patients.
The study included 4,107 MIS-C cases, using data from 2021 for patients younger than 21 years old. The median age was 9 years old.
The findings provide direction for further research, the editorial writers suggested.
Questions that need to be answered include asking why Black children with MIS-C are more likely to have a higher number of organ systems affected.
“Identifying patient biological or socioeconomic factors that can be targeted for treatment or prevention should be pursued,” they wrote.
The CDC says symptoms of MIS-C are an ongoing fever plus more than one of the following: stomach pain, bloodshot eyes, diarrhea, dizziness or lightheadedness (signs of low blood pressure), skin rash, or vomiting.
A version of this article first appeared on WebMD.com.
FROM JAMA NETWORK OPEN
Five thoughts on the Damar Hamlin collapse
The obvious first statement is that it’s neither wise nor appropriate to speculate on the specifics of Damar Hamlin’s cardiac event during a football game on Jan. 2 (including the possibility of commotio cordis) or his ongoing care. The public nature of his collapse induces intense curiosity but people with illness deserve privacy. Privacy in health care is in short supply. I disagree strongly with those who say his doctors ought to be giving public updates. That’s up to the family.
But there are important general concepts to consider about this incident. These include ...
Cardiac arrest can happen to anyone
People with structural heart disease or other chronic illnesses have a higher risk of arrhythmia, but the notion that athletes are immune from cardiac arrest is wrong. This sentence almost seems too obvious to write, but to this day, I hear clinicians express surprise that an athletic person has heart disease.
Survival turns on rapid and effective intervention
In the old days of electrophysiology, we used to test implantable cardioverter-defibrillators during an implant procedure by inducing ventricular fibrillation (VF) and watching the device convert it. Thankfully, trials have shown that this is no longer necessary for most implants.
When you induce VF In the EP lab, you learn quickly that a) it causes loss of consciousness in a matter of seconds, b) rapid defibrillation restores consciousness, often without the patients knowing or remembering they passed out, and c) the failure of the shock to terminate VF results in deterioration in a matter of 1-2 minutes. Even 1 minute in VF feels so long.
Need is an appropriate word in VF treatment
Clinicians often use the verb need. As in, this patient needs this pill or this procedure. It’s rarely appropriate.
But in the case of treating VF, patients truly need rapid defibrillation. Survival of out-of-hospital cardiac arrest is low because there just aren’t enough automated external defibrillators (AEDs) or people trained to use them. A study of patients who had out-of-hospital cardiac arrest in Denmark found that 30-day survival almost doubled (28.8% vs. 16.4%), when the nearest AED was accessible.
Bystanders must act
The public messages are simple: If a person loses consciousness in front of you, and is not breathing normally, assume it is a cardiac arrest, call 911 to get professional help, and start hands-only chest compressions. Don’t spend time checking for a pulse or trying to wake the person. If this is not a cardiac arrest, they will soon tell you to stop compressing their chest. Seconds matter.
Chest compressions are important but what is really needed is defibrillation. A crucial step in CPR is to send someone to get an AED and get the pads attached. If this is a shockable rhythm, deliver the shock. Hamlin’s collapse emphasizes the importance of the AED; without it, his survival to the hospital would have been unlikely.
Widespread preparticipation screening of young athletes remains a bad idea
Whenever cardiac arrest occurs in an athlete, in such a public way, people think about prevention. Surely it is better to prevent such an event than react to it, goes the thinking. The argument against this idea has four prongs:
The incidence of cardiac disease in a young athlete is extremely low, which sets up a situation where most “positive” tests are false positive. A false positive screening ECG or echocardiogram can create harm in multiple ways. One is the risk from downstream procedures, but worse is the inappropriate disqualification from sport. Healthwise, few harms could be greater than creating long-term fear of exercise in someone.
There is also the problem of false-negative screening tests. An ECG may be normal in the setting of hypertrophic cardiomyopathy. And a normal echocardiogram does not exclude arrhythmogenic right ventricular cardiomyopathy or other genetic causes of cardiac arrest. In a 2018 study from a major sports cardiology center in London, 6 of the 8 sudden cardiac deaths in their series were in athletes who had no detectable abnormalities on screening.
Even when disease is found, it’s not clear that prohibiting participation in sports prevents sudden death. Many previous class III recommendations against participation in sport now carry class II – may be considered – designations.
Finally, screening for any disease loses value as treatments improve. Public education regarding rapid intervention with CPR and AED use is the best treatment option. A great example is the case of Christian Erikson, a Danish soccer player who suffered cardiac arrest during a match at the European Championships in 2021 and was rapidly defibrillated on the field. Therapy was so effective that he was conscious and able to wave to fans on his way out of the stadium. He has now returned to elite competition.
Proponents of screening might oppose my take by saying that National Football League players are intensely screened. But this is different from widespread screening of high school and college athletes. It might sound harsh to say, but professional teams have dualities of interests in the health of their athletes given the million-dollar contracts.
What’s more, professional teams can afford to hire expert cardiologists to perform the testing. This would likely reduce the rate of false-positive findings, compared with screening in the community setting. I often have young people referred to me because of asymptomatic bradycardia found during athletic screening – an obviously normal finding.
Conclusions
As long as there are sports, there will be athletes who suffer cardiac arrest.
We can both hope for Hamlin’s full recovery and learn lessons to help reduce the rate of death from out-of-hospital cardiac arrest. This mostly involves education on how to help fellow humans and a public health commitment to access to AEDs.
John Mandrola, MD, practices cardiac electrophysiology in Louisville, Ky. and is a writer and podcaster for Medscape. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
The obvious first statement is that it’s neither wise nor appropriate to speculate on the specifics of Damar Hamlin’s cardiac event during a football game on Jan. 2 (including the possibility of commotio cordis) or his ongoing care. The public nature of his collapse induces intense curiosity but people with illness deserve privacy. Privacy in health care is in short supply. I disagree strongly with those who say his doctors ought to be giving public updates. That’s up to the family.
But there are important general concepts to consider about this incident. These include ...
Cardiac arrest can happen to anyone
People with structural heart disease or other chronic illnesses have a higher risk of arrhythmia, but the notion that athletes are immune from cardiac arrest is wrong. This sentence almost seems too obvious to write, but to this day, I hear clinicians express surprise that an athletic person has heart disease.
Survival turns on rapid and effective intervention
In the old days of electrophysiology, we used to test implantable cardioverter-defibrillators during an implant procedure by inducing ventricular fibrillation (VF) and watching the device convert it. Thankfully, trials have shown that this is no longer necessary for most implants.
When you induce VF In the EP lab, you learn quickly that a) it causes loss of consciousness in a matter of seconds, b) rapid defibrillation restores consciousness, often without the patients knowing or remembering they passed out, and c) the failure of the shock to terminate VF results in deterioration in a matter of 1-2 minutes. Even 1 minute in VF feels so long.
Need is an appropriate word in VF treatment
Clinicians often use the verb need. As in, this patient needs this pill or this procedure. It’s rarely appropriate.
But in the case of treating VF, patients truly need rapid defibrillation. Survival of out-of-hospital cardiac arrest is low because there just aren’t enough automated external defibrillators (AEDs) or people trained to use them. A study of patients who had out-of-hospital cardiac arrest in Denmark found that 30-day survival almost doubled (28.8% vs. 16.4%), when the nearest AED was accessible.
Bystanders must act
The public messages are simple: If a person loses consciousness in front of you, and is not breathing normally, assume it is a cardiac arrest, call 911 to get professional help, and start hands-only chest compressions. Don’t spend time checking for a pulse or trying to wake the person. If this is not a cardiac arrest, they will soon tell you to stop compressing their chest. Seconds matter.
Chest compressions are important but what is really needed is defibrillation. A crucial step in CPR is to send someone to get an AED and get the pads attached. If this is a shockable rhythm, deliver the shock. Hamlin’s collapse emphasizes the importance of the AED; without it, his survival to the hospital would have been unlikely.
Widespread preparticipation screening of young athletes remains a bad idea
Whenever cardiac arrest occurs in an athlete, in such a public way, people think about prevention. Surely it is better to prevent such an event than react to it, goes the thinking. The argument against this idea has four prongs:
The incidence of cardiac disease in a young athlete is extremely low, which sets up a situation where most “positive” tests are false positive. A false positive screening ECG or echocardiogram can create harm in multiple ways. One is the risk from downstream procedures, but worse is the inappropriate disqualification from sport. Healthwise, few harms could be greater than creating long-term fear of exercise in someone.
There is also the problem of false-negative screening tests. An ECG may be normal in the setting of hypertrophic cardiomyopathy. And a normal echocardiogram does not exclude arrhythmogenic right ventricular cardiomyopathy or other genetic causes of cardiac arrest. In a 2018 study from a major sports cardiology center in London, 6 of the 8 sudden cardiac deaths in their series were in athletes who had no detectable abnormalities on screening.
Even when disease is found, it’s not clear that prohibiting participation in sports prevents sudden death. Many previous class III recommendations against participation in sport now carry class II – may be considered – designations.
Finally, screening for any disease loses value as treatments improve. Public education regarding rapid intervention with CPR and AED use is the best treatment option. A great example is the case of Christian Erikson, a Danish soccer player who suffered cardiac arrest during a match at the European Championships in 2021 and was rapidly defibrillated on the field. Therapy was so effective that he was conscious and able to wave to fans on his way out of the stadium. He has now returned to elite competition.
Proponents of screening might oppose my take by saying that National Football League players are intensely screened. But this is different from widespread screening of high school and college athletes. It might sound harsh to say, but professional teams have dualities of interests in the health of their athletes given the million-dollar contracts.
What’s more, professional teams can afford to hire expert cardiologists to perform the testing. This would likely reduce the rate of false-positive findings, compared with screening in the community setting. I often have young people referred to me because of asymptomatic bradycardia found during athletic screening – an obviously normal finding.
Conclusions
As long as there are sports, there will be athletes who suffer cardiac arrest.
We can both hope for Hamlin’s full recovery and learn lessons to help reduce the rate of death from out-of-hospital cardiac arrest. This mostly involves education on how to help fellow humans and a public health commitment to access to AEDs.
John Mandrola, MD, practices cardiac electrophysiology in Louisville, Ky. and is a writer and podcaster for Medscape. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
The obvious first statement is that it’s neither wise nor appropriate to speculate on the specifics of Damar Hamlin’s cardiac event during a football game on Jan. 2 (including the possibility of commotio cordis) or his ongoing care. The public nature of his collapse induces intense curiosity but people with illness deserve privacy. Privacy in health care is in short supply. I disagree strongly with those who say his doctors ought to be giving public updates. That’s up to the family.
But there are important general concepts to consider about this incident. These include ...
Cardiac arrest can happen to anyone
People with structural heart disease or other chronic illnesses have a higher risk of arrhythmia, but the notion that athletes are immune from cardiac arrest is wrong. This sentence almost seems too obvious to write, but to this day, I hear clinicians express surprise that an athletic person has heart disease.
Survival turns on rapid and effective intervention
In the old days of electrophysiology, we used to test implantable cardioverter-defibrillators during an implant procedure by inducing ventricular fibrillation (VF) and watching the device convert it. Thankfully, trials have shown that this is no longer necessary for most implants.
When you induce VF In the EP lab, you learn quickly that a) it causes loss of consciousness in a matter of seconds, b) rapid defibrillation restores consciousness, often without the patients knowing or remembering they passed out, and c) the failure of the shock to terminate VF results in deterioration in a matter of 1-2 minutes. Even 1 minute in VF feels so long.
Need is an appropriate word in VF treatment
Clinicians often use the verb need. As in, this patient needs this pill or this procedure. It’s rarely appropriate.
But in the case of treating VF, patients truly need rapid defibrillation. Survival of out-of-hospital cardiac arrest is low because there just aren’t enough automated external defibrillators (AEDs) or people trained to use them. A study of patients who had out-of-hospital cardiac arrest in Denmark found that 30-day survival almost doubled (28.8% vs. 16.4%), when the nearest AED was accessible.
Bystanders must act
The public messages are simple: If a person loses consciousness in front of you, and is not breathing normally, assume it is a cardiac arrest, call 911 to get professional help, and start hands-only chest compressions. Don’t spend time checking for a pulse or trying to wake the person. If this is not a cardiac arrest, they will soon tell you to stop compressing their chest. Seconds matter.
Chest compressions are important but what is really needed is defibrillation. A crucial step in CPR is to send someone to get an AED and get the pads attached. If this is a shockable rhythm, deliver the shock. Hamlin’s collapse emphasizes the importance of the AED; without it, his survival to the hospital would have been unlikely.
Widespread preparticipation screening of young athletes remains a bad idea
Whenever cardiac arrest occurs in an athlete, in such a public way, people think about prevention. Surely it is better to prevent such an event than react to it, goes the thinking. The argument against this idea has four prongs:
The incidence of cardiac disease in a young athlete is extremely low, which sets up a situation where most “positive” tests are false positive. A false positive screening ECG or echocardiogram can create harm in multiple ways. One is the risk from downstream procedures, but worse is the inappropriate disqualification from sport. Healthwise, few harms could be greater than creating long-term fear of exercise in someone.
There is also the problem of false-negative screening tests. An ECG may be normal in the setting of hypertrophic cardiomyopathy. And a normal echocardiogram does not exclude arrhythmogenic right ventricular cardiomyopathy or other genetic causes of cardiac arrest. In a 2018 study from a major sports cardiology center in London, 6 of the 8 sudden cardiac deaths in their series were in athletes who had no detectable abnormalities on screening.
Even when disease is found, it’s not clear that prohibiting participation in sports prevents sudden death. Many previous class III recommendations against participation in sport now carry class II – may be considered – designations.
Finally, screening for any disease loses value as treatments improve. Public education regarding rapid intervention with CPR and AED use is the best treatment option. A great example is the case of Christian Erikson, a Danish soccer player who suffered cardiac arrest during a match at the European Championships in 2021 and was rapidly defibrillated on the field. Therapy was so effective that he was conscious and able to wave to fans on his way out of the stadium. He has now returned to elite competition.
Proponents of screening might oppose my take by saying that National Football League players are intensely screened. But this is different from widespread screening of high school and college athletes. It might sound harsh to say, but professional teams have dualities of interests in the health of their athletes given the million-dollar contracts.
What’s more, professional teams can afford to hire expert cardiologists to perform the testing. This would likely reduce the rate of false-positive findings, compared with screening in the community setting. I often have young people referred to me because of asymptomatic bradycardia found during athletic screening – an obviously normal finding.
Conclusions
As long as there are sports, there will be athletes who suffer cardiac arrest.
We can both hope for Hamlin’s full recovery and learn lessons to help reduce the rate of death from out-of-hospital cardiac arrest. This mostly involves education on how to help fellow humans and a public health commitment to access to AEDs.
John Mandrola, MD, practices cardiac electrophysiology in Louisville, Ky. and is a writer and podcaster for Medscape. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.