User login
COVID-19 and psoriasis: Is there a link?
.
Psoriasis has several well-established triggers, including stress, skin injury, cold or warm air, and allergies. Illnesses like strep throat can also cause a psoriasis flare in some people – and it appears COVID may also do so. “Psoriasis flares have long been associated with bacterial and viral infections, particularly a form of psoriasis called guttate, which is characterized by tons of tiny red scaly bumps all over the body,” said Joel M. Gelfand, MD, a professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia. “Infection with COVID-19 has been associated with flares of guttate and pustular psoriasis, and even psoriasis that affects 100% of the skin ... in many published case reports.”
Israeli researchers recently found that psoriasis patients have a slightly higher risk of getting COVID, although they are not at higher risk of hospitalization or death, which could be related to treatment with immune-modulating therapy, which can increase their risk of infections.
How could COVID cause psoriasis to flare?
Psoriasis is an autoimmune condition, and inflammation can cause symptoms.
Investigators for a study from Albany (N.Y.) Medical College and Weirton (Pa.) Medical Center found that people in the study who were already diagnosed with the skin condition had an unexpected flare within a week to a month after testing positive for COVID. New psoriasis after a COVID infection was also found. The researchers think this could be because COVID causes inflammation in the body, which negatively affects previously well-controlled psoriasis. They also think it’s possible that COVID-related inflammation could trigger a genetic tendency to have psoriasis, which may explain why it can appear for the first time after a positive test.
“A viral infection like COVID-19 can signal the release of proinflammatory factors that can appear as rashes, such as with psoriasis.” said Robert O. Carpenter, MD, director of wellness at Texas A&M University in Bryan.
What are the symptoms of COVID-related psoriasis?
The signs are the same as those of any form of psoriasis.
For a patient with psoriasis, will COVID automatically make it worse?
Not necessarily.
“Psoriasis is a common condition, so people should be aware that new psoriasis that develops may not be related to COVID-19,” said Esther Freeman MD, PhD, director of global health dermatology at Massachusetts General Hospital in Boston.
As with every aspect of COVID, doctors and scientists are still learning about how serious and widespread a problem psoriasis after COVID-19 may be. “We have seen case reports that psoriasis can flare after COVID-19,” said Dr. Freeman, who is also an associate professor of dermatology at Harvard Medical School. “I will say, this has not been a tidal wave – more like sporadic cases here and there. So I do not think psoriasis flares are a major post-COVID finding, nor do they necessarily mean you have long COVID. That being said, we know that many different infections can cause psoriasis flares, and so, in that respect, it’s not that surprising that SARS-CoV-2, like other infections, could trigger a psoriasis flare.”
Could getting COVID more than once cause psoriasis to flare? It’s possible.
“Your body can change after having COVID-19,” said Dr. Carpenter. “We don’t know the long-term implications, but having COVID-19 repeatedly can increase the risk of long COVID, which can cause many systemic changes in your body.”
Another important point: For patients who take biologics for treating psoriasis, getting vaccinated and boosted for COVID is an important step to take to help protect themselves.
Is psoriasis itself a potential symptom of COVID?
“Yes, but we don’t know the frequency at which this may occur, and a causal relationship is difficult to establish from just case reports,” said Dr. Gelfand, who’s also medical director of the clinical studies unit in the department of dermatology at his university. “Typically, if a patient presents with a flare of psoriasis, particularly guttate, pustular, or erythrodermic forms, an infectious trigger should be considered, and testing for strep and possibly COVID-19 may be appropriate.”
A version of this article first appeared on Medscape.com.
.
Psoriasis has several well-established triggers, including stress, skin injury, cold or warm air, and allergies. Illnesses like strep throat can also cause a psoriasis flare in some people – and it appears COVID may also do so. “Psoriasis flares have long been associated with bacterial and viral infections, particularly a form of psoriasis called guttate, which is characterized by tons of tiny red scaly bumps all over the body,” said Joel M. Gelfand, MD, a professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia. “Infection with COVID-19 has been associated with flares of guttate and pustular psoriasis, and even psoriasis that affects 100% of the skin ... in many published case reports.”
Israeli researchers recently found that psoriasis patients have a slightly higher risk of getting COVID, although they are not at higher risk of hospitalization or death, which could be related to treatment with immune-modulating therapy, which can increase their risk of infections.
How could COVID cause psoriasis to flare?
Psoriasis is an autoimmune condition, and inflammation can cause symptoms.
Investigators for a study from Albany (N.Y.) Medical College and Weirton (Pa.) Medical Center found that people in the study who were already diagnosed with the skin condition had an unexpected flare within a week to a month after testing positive for COVID. New psoriasis after a COVID infection was also found. The researchers think this could be because COVID causes inflammation in the body, which negatively affects previously well-controlled psoriasis. They also think it’s possible that COVID-related inflammation could trigger a genetic tendency to have psoriasis, which may explain why it can appear for the first time after a positive test.
“A viral infection like COVID-19 can signal the release of proinflammatory factors that can appear as rashes, such as with psoriasis.” said Robert O. Carpenter, MD, director of wellness at Texas A&M University in Bryan.
What are the symptoms of COVID-related psoriasis?
The signs are the same as those of any form of psoriasis.
For a patient with psoriasis, will COVID automatically make it worse?
Not necessarily.
“Psoriasis is a common condition, so people should be aware that new psoriasis that develops may not be related to COVID-19,” said Esther Freeman MD, PhD, director of global health dermatology at Massachusetts General Hospital in Boston.
As with every aspect of COVID, doctors and scientists are still learning about how serious and widespread a problem psoriasis after COVID-19 may be. “We have seen case reports that psoriasis can flare after COVID-19,” said Dr. Freeman, who is also an associate professor of dermatology at Harvard Medical School. “I will say, this has not been a tidal wave – more like sporadic cases here and there. So I do not think psoriasis flares are a major post-COVID finding, nor do they necessarily mean you have long COVID. That being said, we know that many different infections can cause psoriasis flares, and so, in that respect, it’s not that surprising that SARS-CoV-2, like other infections, could trigger a psoriasis flare.”
Could getting COVID more than once cause psoriasis to flare? It’s possible.
“Your body can change after having COVID-19,” said Dr. Carpenter. “We don’t know the long-term implications, but having COVID-19 repeatedly can increase the risk of long COVID, which can cause many systemic changes in your body.”
Another important point: For patients who take biologics for treating psoriasis, getting vaccinated and boosted for COVID is an important step to take to help protect themselves.
Is psoriasis itself a potential symptom of COVID?
“Yes, but we don’t know the frequency at which this may occur, and a causal relationship is difficult to establish from just case reports,” said Dr. Gelfand, who’s also medical director of the clinical studies unit in the department of dermatology at his university. “Typically, if a patient presents with a flare of psoriasis, particularly guttate, pustular, or erythrodermic forms, an infectious trigger should be considered, and testing for strep and possibly COVID-19 may be appropriate.”
A version of this article first appeared on Medscape.com.
.
Psoriasis has several well-established triggers, including stress, skin injury, cold or warm air, and allergies. Illnesses like strep throat can also cause a psoriasis flare in some people – and it appears COVID may also do so. “Psoriasis flares have long been associated with bacterial and viral infections, particularly a form of psoriasis called guttate, which is characterized by tons of tiny red scaly bumps all over the body,” said Joel M. Gelfand, MD, a professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia. “Infection with COVID-19 has been associated with flares of guttate and pustular psoriasis, and even psoriasis that affects 100% of the skin ... in many published case reports.”
Israeli researchers recently found that psoriasis patients have a slightly higher risk of getting COVID, although they are not at higher risk of hospitalization or death, which could be related to treatment with immune-modulating therapy, which can increase their risk of infections.
How could COVID cause psoriasis to flare?
Psoriasis is an autoimmune condition, and inflammation can cause symptoms.
Investigators for a study from Albany (N.Y.) Medical College and Weirton (Pa.) Medical Center found that people in the study who were already diagnosed with the skin condition had an unexpected flare within a week to a month after testing positive for COVID. New psoriasis after a COVID infection was also found. The researchers think this could be because COVID causes inflammation in the body, which negatively affects previously well-controlled psoriasis. They also think it’s possible that COVID-related inflammation could trigger a genetic tendency to have psoriasis, which may explain why it can appear for the first time after a positive test.
“A viral infection like COVID-19 can signal the release of proinflammatory factors that can appear as rashes, such as with psoriasis.” said Robert O. Carpenter, MD, director of wellness at Texas A&M University in Bryan.
What are the symptoms of COVID-related psoriasis?
The signs are the same as those of any form of psoriasis.
For a patient with psoriasis, will COVID automatically make it worse?
Not necessarily.
“Psoriasis is a common condition, so people should be aware that new psoriasis that develops may not be related to COVID-19,” said Esther Freeman MD, PhD, director of global health dermatology at Massachusetts General Hospital in Boston.
As with every aspect of COVID, doctors and scientists are still learning about how serious and widespread a problem psoriasis after COVID-19 may be. “We have seen case reports that psoriasis can flare after COVID-19,” said Dr. Freeman, who is also an associate professor of dermatology at Harvard Medical School. “I will say, this has not been a tidal wave – more like sporadic cases here and there. So I do not think psoriasis flares are a major post-COVID finding, nor do they necessarily mean you have long COVID. That being said, we know that many different infections can cause psoriasis flares, and so, in that respect, it’s not that surprising that SARS-CoV-2, like other infections, could trigger a psoriasis flare.”
Could getting COVID more than once cause psoriasis to flare? It’s possible.
“Your body can change after having COVID-19,” said Dr. Carpenter. “We don’t know the long-term implications, but having COVID-19 repeatedly can increase the risk of long COVID, which can cause many systemic changes in your body.”
Another important point: For patients who take biologics for treating psoriasis, getting vaccinated and boosted for COVID is an important step to take to help protect themselves.
Is psoriasis itself a potential symptom of COVID?
“Yes, but we don’t know the frequency at which this may occur, and a causal relationship is difficult to establish from just case reports,” said Dr. Gelfand, who’s also medical director of the clinical studies unit in the department of dermatology at his university. “Typically, if a patient presents with a flare of psoriasis, particularly guttate, pustular, or erythrodermic forms, an infectious trigger should be considered, and testing for strep and possibly COVID-19 may be appropriate.”
A version of this article first appeared on Medscape.com.
FDA puts partial hold on investigational alopecia areata drug deuruxolitinib
The in a press release on May 2.
The announcement came after a pulmonary embolism occurred with the 12-mg twice-daily dose in one of the long-term open-label extension (OLE) studies, the company, Sun Pharmaceutical Industries, said.
The company stated that the FDA has placed the Investigational New Drug testing for deuruxolitinib on partial clinical hold, and the agency is requiring that study participants who are currently on the 12-mg twice-daily dose in the OLE studies stop taking that dose. The hold covers only the 12-mg dose.
No hold on 8-mg dose
“There have been no thrombotic events reported to date for the 8-mg b.i.d. dose and U.S. FDA has not placed the 8-mg b.i.d. dose on hold,” the company said in the statement.
The statement added, “We are taking immediate steps to transition the patients in the OLE studies to the 8-mg b.i.d. dose arm in the ongoing studies.”
The company said that no thromboembolic events were observed in the phase 2 or phase 3 trials and said that it will work closely with the FDA to address its concerns. A formal letter detailing the FDA’s concerns is expected within 30 days.
Deuruxolitinib is an investigational oral selective inhibitor of Janus kinase 1 (JAK1) and JAK2 enzymes.
The FDA has granted deuruxolitinib breakthrough therapy designation for the treatment of adult patients with moderate to severe alopecia areata as well as fast-track designation for the treatment of alopecia areata.
In March, this news organization reported from the annual meeting of the American Academy of Dermatology that, based on phase 3 studies that demonstrate robust hair growth in about one-third of patients, deuruxolitinib has the potential to become the second JAK inhibitor available for the treatment of alopecia areata. If approved, it will join baricitinib (Olumiant), which received FDA approval almost 1 year ago.
Also at the AAD annual meeting, this news organization reported that principal investigator Brett A. King, MD, PhD, associate professor of dermatology, Yale University, New Haven, Conn., in his presentation on the results of THRIVE-AA2, one of the two phase 3 trials of deuruxolitinib, displayed several before-and-after photos and said, “The photos tell the whole story. This is why there is so much excitement about these drugs.” Dr King also was a principal investigator in studies of baricitinib.
With one exception, labeling for baricitinib and other JAK inhibitors with dermatologic indications includes a boxed warning listing serious adverse events including the risk for major adverse cardiac events and thrombosis, including pulmonary embolism, based on the risks in a rheumatoid arthritis study.
A version of this article first appeared on Medscape.com.
The in a press release on May 2.
The announcement came after a pulmonary embolism occurred with the 12-mg twice-daily dose in one of the long-term open-label extension (OLE) studies, the company, Sun Pharmaceutical Industries, said.
The company stated that the FDA has placed the Investigational New Drug testing for deuruxolitinib on partial clinical hold, and the agency is requiring that study participants who are currently on the 12-mg twice-daily dose in the OLE studies stop taking that dose. The hold covers only the 12-mg dose.
No hold on 8-mg dose
“There have been no thrombotic events reported to date for the 8-mg b.i.d. dose and U.S. FDA has not placed the 8-mg b.i.d. dose on hold,” the company said in the statement.
The statement added, “We are taking immediate steps to transition the patients in the OLE studies to the 8-mg b.i.d. dose arm in the ongoing studies.”
The company said that no thromboembolic events were observed in the phase 2 or phase 3 trials and said that it will work closely with the FDA to address its concerns. A formal letter detailing the FDA’s concerns is expected within 30 days.
Deuruxolitinib is an investigational oral selective inhibitor of Janus kinase 1 (JAK1) and JAK2 enzymes.
The FDA has granted deuruxolitinib breakthrough therapy designation for the treatment of adult patients with moderate to severe alopecia areata as well as fast-track designation for the treatment of alopecia areata.
In March, this news organization reported from the annual meeting of the American Academy of Dermatology that, based on phase 3 studies that demonstrate robust hair growth in about one-third of patients, deuruxolitinib has the potential to become the second JAK inhibitor available for the treatment of alopecia areata. If approved, it will join baricitinib (Olumiant), which received FDA approval almost 1 year ago.
Also at the AAD annual meeting, this news organization reported that principal investigator Brett A. King, MD, PhD, associate professor of dermatology, Yale University, New Haven, Conn., in his presentation on the results of THRIVE-AA2, one of the two phase 3 trials of deuruxolitinib, displayed several before-and-after photos and said, “The photos tell the whole story. This is why there is so much excitement about these drugs.” Dr King also was a principal investigator in studies of baricitinib.
With one exception, labeling for baricitinib and other JAK inhibitors with dermatologic indications includes a boxed warning listing serious adverse events including the risk for major adverse cardiac events and thrombosis, including pulmonary embolism, based on the risks in a rheumatoid arthritis study.
A version of this article first appeared on Medscape.com.
The in a press release on May 2.
The announcement came after a pulmonary embolism occurred with the 12-mg twice-daily dose in one of the long-term open-label extension (OLE) studies, the company, Sun Pharmaceutical Industries, said.
The company stated that the FDA has placed the Investigational New Drug testing for deuruxolitinib on partial clinical hold, and the agency is requiring that study participants who are currently on the 12-mg twice-daily dose in the OLE studies stop taking that dose. The hold covers only the 12-mg dose.
No hold on 8-mg dose
“There have been no thrombotic events reported to date for the 8-mg b.i.d. dose and U.S. FDA has not placed the 8-mg b.i.d. dose on hold,” the company said in the statement.
The statement added, “We are taking immediate steps to transition the patients in the OLE studies to the 8-mg b.i.d. dose arm in the ongoing studies.”
The company said that no thromboembolic events were observed in the phase 2 or phase 3 trials and said that it will work closely with the FDA to address its concerns. A formal letter detailing the FDA’s concerns is expected within 30 days.
Deuruxolitinib is an investigational oral selective inhibitor of Janus kinase 1 (JAK1) and JAK2 enzymes.
The FDA has granted deuruxolitinib breakthrough therapy designation for the treatment of adult patients with moderate to severe alopecia areata as well as fast-track designation for the treatment of alopecia areata.
In March, this news organization reported from the annual meeting of the American Academy of Dermatology that, based on phase 3 studies that demonstrate robust hair growth in about one-third of patients, deuruxolitinib has the potential to become the second JAK inhibitor available for the treatment of alopecia areata. If approved, it will join baricitinib (Olumiant), which received FDA approval almost 1 year ago.
Also at the AAD annual meeting, this news organization reported that principal investigator Brett A. King, MD, PhD, associate professor of dermatology, Yale University, New Haven, Conn., in his presentation on the results of THRIVE-AA2, one of the two phase 3 trials of deuruxolitinib, displayed several before-and-after photos and said, “The photos tell the whole story. This is why there is so much excitement about these drugs.” Dr King also was a principal investigator in studies of baricitinib.
With one exception, labeling for baricitinib and other JAK inhibitors with dermatologic indications includes a boxed warning listing serious adverse events including the risk for major adverse cardiac events and thrombosis, including pulmonary embolism, based on the risks in a rheumatoid arthritis study.
A version of this article first appeared on Medscape.com.
Gray hair and aging: Could ‘stuck’ stem cells be to blame?
New evidence points more to a cycle wherein undifferentiated stem cells mature to perform their hair-coloring duties and then transform back to their primitive form. To accomplish this, they need to stay on the move.
When these special stem cells get “stuck” in the follicle, gray hair is the result, according to a new study reported online in Nature.
The regeneration cycle of melanocyte stem cells (McSCs) to melanocytes and back again can last for years. However, McSCs die sooner than do other cells nearby, such as hair follicle stem cells. This difference can explain why people go gray but still grow hair.
“It was thought that melanocyte stem cells are maintained in an undifferentiated state, instead of repeating differentiation and de-differentiation,” said the study’s senior investigator Mayumi Ito, PhD, professor in the departments of dermatology and cell biology at NYU Langone Health, New York.
The process involves different compartments in the hair follicle – the germ area is where the stem cells regenerate; the follicle bulge is where they get stuck. A different microenvironment in each location dictates how they change. This “chameleon-like” property surprised researchers.
Now that investigators figured out how gray hair might get started, a next step will be to search for a way to stop it.
The research has been performed in mice to date but could translate to humans. “Because the structure of the hair follicle is similar between mice and humans, we speculate that human melanocytes may also demonstrate the plasticity during hair regeneration,” Dr. Ito told this news organization.
Future findings could also lead to new therapies. “Our study suggests that moving melanocytes to a proper location within the hair follicle may help prevent gray hair,” Dr. Ito said.
Given the known effects of ultraviolet B (UVB) radiation on melanocytes, Dr. Ito and colleagues wanted to see what effect it might have on this cycle. So in the study, they exposed hair follicles of mice to UVB radiation and report it speeds up the process for McSCs to transform to color-producing melanocytes. They found that these McSCs can regenerate or change back to undifferentiated stem cells, so UVB radiation does not interrupt the process.
A melanoma clue?
The study also could have implications for melanoma. Unlike other tumors, melanocytes that cause cancer can self-renew even from a fully differentiated, pigmented form, the researchers note.
This makes melanomas more difficult to eliminate.
“Our study suggests normal melanocytes are very plastic and can reverse a differentiation state. Melanoma cells are known to be very plastic,” Dr. Ito said. “We consider this feature of melanoma may be related to the high plasticity of original melanocytes.”
The finding that melanocyte stem cells “are more plastic than maybe previously given credit for … certainly has implications in melanoma,” agreed Melissa Harris, PhD, associate professor, department of biology at the University of Alabama, Birmingham, when asked to comment on the study.
Small technology, big insights?
The advanced technology used by Dr. Ito and colleagues in the study included 3D-intravital imaging and single-cell RNA sequencing to track the stem cells in almost real time as they aged and moved within each hair follicle.
“This paper uses a nice mix of classic and modern techniques to help answer a question that many in the field of pigmentation biology have suspected for a long time. Not all dormant melanocyte stem cells are created equal,” Dr. Harris said.
“The one question not answered in this paper is how to reverse the dysfunction of the melanocyte stem cell ‘stuck’ in the hair bulge,” Dr. Harris added. “There are numerous clinical case studies in humans showing medicine-induced hair repigmentation, and perhaps these cases are examples of dysfunctional melanocyte stem cells becoming ‘unstuck.’ ”
‘Very interesting’ findings
The study and its results “are very interesting from a mechanistic perspective and basic science view,” said Anthony M. Rossi, MD, a private practice dermatologist and assistant attending dermatologist at Memorial Sloan Kettering Cancer Center in New York, when asked to comment on the results.
The research provides another view of how melanocyte stem cells can pigment the hair shaft, Dr. Rossi added. “It gives insight into the behavior of stem cells and how they can travel and change state, something not well-known before.”
Dr. Rossi cautioned that other mechanisms are likely taking place. He pointed out that graying of hair can actually occur after a sudden stress event, as well as with vitamin B12 deficiency, thyroid disease, vitiligo-related autoimmune destruction, neurofibromatosis, tuberous sclerosis, and alopecia areata.
The “standout concept” in this paper is that the melanocyte stem cells are stranded and are not getting the right signal from the microenvironment to amplify and appropriately migrate to provide pigment to the hair shaft, said Paradi Mirmirani, MD, a private practice dermatologist in Vallejo, Calif.
It could be challenging to find the right signaling to reverse the graying process, Dr. Mirmirani added. “But the first step is always to understand the underlying basic mechanism. It would be interesting to see if other factors such as smoking, stress … influence the melanocyte stem cells in the same way.”
Grants from the National Institutes of Health and the Department of Defense supported the study. Dr. Ito, Dr. Harris, Dr. Mirmirani, and Dr. Rossi had no relevant disclosures.
A version of this article first appeared on Medscape.com.
New evidence points more to a cycle wherein undifferentiated stem cells mature to perform their hair-coloring duties and then transform back to their primitive form. To accomplish this, they need to stay on the move.
When these special stem cells get “stuck” in the follicle, gray hair is the result, according to a new study reported online in Nature.
The regeneration cycle of melanocyte stem cells (McSCs) to melanocytes and back again can last for years. However, McSCs die sooner than do other cells nearby, such as hair follicle stem cells. This difference can explain why people go gray but still grow hair.
“It was thought that melanocyte stem cells are maintained in an undifferentiated state, instead of repeating differentiation and de-differentiation,” said the study’s senior investigator Mayumi Ito, PhD, professor in the departments of dermatology and cell biology at NYU Langone Health, New York.
The process involves different compartments in the hair follicle – the germ area is where the stem cells regenerate; the follicle bulge is where they get stuck. A different microenvironment in each location dictates how they change. This “chameleon-like” property surprised researchers.
Now that investigators figured out how gray hair might get started, a next step will be to search for a way to stop it.
The research has been performed in mice to date but could translate to humans. “Because the structure of the hair follicle is similar between mice and humans, we speculate that human melanocytes may also demonstrate the plasticity during hair regeneration,” Dr. Ito told this news organization.
Future findings could also lead to new therapies. “Our study suggests that moving melanocytes to a proper location within the hair follicle may help prevent gray hair,” Dr. Ito said.
Given the known effects of ultraviolet B (UVB) radiation on melanocytes, Dr. Ito and colleagues wanted to see what effect it might have on this cycle. So in the study, they exposed hair follicles of mice to UVB radiation and report it speeds up the process for McSCs to transform to color-producing melanocytes. They found that these McSCs can regenerate or change back to undifferentiated stem cells, so UVB radiation does not interrupt the process.
A melanoma clue?
The study also could have implications for melanoma. Unlike other tumors, melanocytes that cause cancer can self-renew even from a fully differentiated, pigmented form, the researchers note.
This makes melanomas more difficult to eliminate.
“Our study suggests normal melanocytes are very plastic and can reverse a differentiation state. Melanoma cells are known to be very plastic,” Dr. Ito said. “We consider this feature of melanoma may be related to the high plasticity of original melanocytes.”
The finding that melanocyte stem cells “are more plastic than maybe previously given credit for … certainly has implications in melanoma,” agreed Melissa Harris, PhD, associate professor, department of biology at the University of Alabama, Birmingham, when asked to comment on the study.
Small technology, big insights?
The advanced technology used by Dr. Ito and colleagues in the study included 3D-intravital imaging and single-cell RNA sequencing to track the stem cells in almost real time as they aged and moved within each hair follicle.
“This paper uses a nice mix of classic and modern techniques to help answer a question that many in the field of pigmentation biology have suspected for a long time. Not all dormant melanocyte stem cells are created equal,” Dr. Harris said.
“The one question not answered in this paper is how to reverse the dysfunction of the melanocyte stem cell ‘stuck’ in the hair bulge,” Dr. Harris added. “There are numerous clinical case studies in humans showing medicine-induced hair repigmentation, and perhaps these cases are examples of dysfunctional melanocyte stem cells becoming ‘unstuck.’ ”
‘Very interesting’ findings
The study and its results “are very interesting from a mechanistic perspective and basic science view,” said Anthony M. Rossi, MD, a private practice dermatologist and assistant attending dermatologist at Memorial Sloan Kettering Cancer Center in New York, when asked to comment on the results.
The research provides another view of how melanocyte stem cells can pigment the hair shaft, Dr. Rossi added. “It gives insight into the behavior of stem cells and how they can travel and change state, something not well-known before.”
Dr. Rossi cautioned that other mechanisms are likely taking place. He pointed out that graying of hair can actually occur after a sudden stress event, as well as with vitamin B12 deficiency, thyroid disease, vitiligo-related autoimmune destruction, neurofibromatosis, tuberous sclerosis, and alopecia areata.
The “standout concept” in this paper is that the melanocyte stem cells are stranded and are not getting the right signal from the microenvironment to amplify and appropriately migrate to provide pigment to the hair shaft, said Paradi Mirmirani, MD, a private practice dermatologist in Vallejo, Calif.
It could be challenging to find the right signaling to reverse the graying process, Dr. Mirmirani added. “But the first step is always to understand the underlying basic mechanism. It would be interesting to see if other factors such as smoking, stress … influence the melanocyte stem cells in the same way.”
Grants from the National Institutes of Health and the Department of Defense supported the study. Dr. Ito, Dr. Harris, Dr. Mirmirani, and Dr. Rossi had no relevant disclosures.
A version of this article first appeared on Medscape.com.
New evidence points more to a cycle wherein undifferentiated stem cells mature to perform their hair-coloring duties and then transform back to their primitive form. To accomplish this, they need to stay on the move.
When these special stem cells get “stuck” in the follicle, gray hair is the result, according to a new study reported online in Nature.
The regeneration cycle of melanocyte stem cells (McSCs) to melanocytes and back again can last for years. However, McSCs die sooner than do other cells nearby, such as hair follicle stem cells. This difference can explain why people go gray but still grow hair.
“It was thought that melanocyte stem cells are maintained in an undifferentiated state, instead of repeating differentiation and de-differentiation,” said the study’s senior investigator Mayumi Ito, PhD, professor in the departments of dermatology and cell biology at NYU Langone Health, New York.
The process involves different compartments in the hair follicle – the germ area is where the stem cells regenerate; the follicle bulge is where they get stuck. A different microenvironment in each location dictates how they change. This “chameleon-like” property surprised researchers.
Now that investigators figured out how gray hair might get started, a next step will be to search for a way to stop it.
The research has been performed in mice to date but could translate to humans. “Because the structure of the hair follicle is similar between mice and humans, we speculate that human melanocytes may also demonstrate the plasticity during hair regeneration,” Dr. Ito told this news organization.
Future findings could also lead to new therapies. “Our study suggests that moving melanocytes to a proper location within the hair follicle may help prevent gray hair,” Dr. Ito said.
Given the known effects of ultraviolet B (UVB) radiation on melanocytes, Dr. Ito and colleagues wanted to see what effect it might have on this cycle. So in the study, they exposed hair follicles of mice to UVB radiation and report it speeds up the process for McSCs to transform to color-producing melanocytes. They found that these McSCs can regenerate or change back to undifferentiated stem cells, so UVB radiation does not interrupt the process.
A melanoma clue?
The study also could have implications for melanoma. Unlike other tumors, melanocytes that cause cancer can self-renew even from a fully differentiated, pigmented form, the researchers note.
This makes melanomas more difficult to eliminate.
“Our study suggests normal melanocytes are very plastic and can reverse a differentiation state. Melanoma cells are known to be very plastic,” Dr. Ito said. “We consider this feature of melanoma may be related to the high plasticity of original melanocytes.”
The finding that melanocyte stem cells “are more plastic than maybe previously given credit for … certainly has implications in melanoma,” agreed Melissa Harris, PhD, associate professor, department of biology at the University of Alabama, Birmingham, when asked to comment on the study.
Small technology, big insights?
The advanced technology used by Dr. Ito and colleagues in the study included 3D-intravital imaging and single-cell RNA sequencing to track the stem cells in almost real time as they aged and moved within each hair follicle.
“This paper uses a nice mix of classic and modern techniques to help answer a question that many in the field of pigmentation biology have suspected for a long time. Not all dormant melanocyte stem cells are created equal,” Dr. Harris said.
“The one question not answered in this paper is how to reverse the dysfunction of the melanocyte stem cell ‘stuck’ in the hair bulge,” Dr. Harris added. “There are numerous clinical case studies in humans showing medicine-induced hair repigmentation, and perhaps these cases are examples of dysfunctional melanocyte stem cells becoming ‘unstuck.’ ”
‘Very interesting’ findings
The study and its results “are very interesting from a mechanistic perspective and basic science view,” said Anthony M. Rossi, MD, a private practice dermatologist and assistant attending dermatologist at Memorial Sloan Kettering Cancer Center in New York, when asked to comment on the results.
The research provides another view of how melanocyte stem cells can pigment the hair shaft, Dr. Rossi added. “It gives insight into the behavior of stem cells and how they can travel and change state, something not well-known before.”
Dr. Rossi cautioned that other mechanisms are likely taking place. He pointed out that graying of hair can actually occur after a sudden stress event, as well as with vitamin B12 deficiency, thyroid disease, vitiligo-related autoimmune destruction, neurofibromatosis, tuberous sclerosis, and alopecia areata.
The “standout concept” in this paper is that the melanocyte stem cells are stranded and are not getting the right signal from the microenvironment to amplify and appropriately migrate to provide pigment to the hair shaft, said Paradi Mirmirani, MD, a private practice dermatologist in Vallejo, Calif.
It could be challenging to find the right signaling to reverse the graying process, Dr. Mirmirani added. “But the first step is always to understand the underlying basic mechanism. It would be interesting to see if other factors such as smoking, stress … influence the melanocyte stem cells in the same way.”
Grants from the National Institutes of Health and the Department of Defense supported the study. Dr. Ito, Dr. Harris, Dr. Mirmirani, and Dr. Rossi had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE
Pustules on face
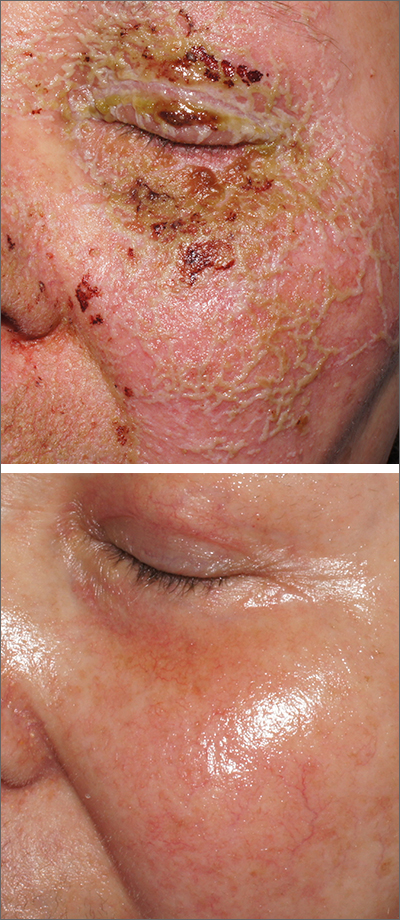
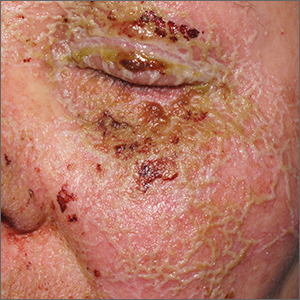
A review of the patient’s chemotherapy medications revealed that 4 weeks earlier, panitumumab had been added to her folinic acid, fluorouracil, and irinotecan (FOLFIRI) regimen. The physician diagnosed this acneiform eruption as an adverse effect of the panitumumab.
Panitumumab is a monoclonal antibody that works to inhibit epidermal growth factor receptor (EGFR) proteins that are overexpressed on some solid tumors and responsible for cancer cell proliferation. EGFR inhibitor–induced acneiform eruptions are common in patients receiving panitumumab.
EGFR proteins have been a target of chemotherapy since the approval of the small molecule erlotinib in 2004. Panitumumab and cetuximab are monoclonal antibodies targeting EGFR and improve long-term survival in patients with metastatic colorectal cancer when added to other standard chemotherapy regimens. EGFR is found throughout the epidermis and all EGFR inhibitors may cause unique skin toxicity not seen with other chemotherapy agents. In 1 study of 229 patients, 59% of patients exhibited skin toxicity at Day 15; the most common examples included widespread acne-like papules and pustules or an eczema-like manifestation.1 Eruptions may be worsened by significant sun exposure while on panitumumab. In this case, the acneiform eruption occurred more intensely along visible facial telangiectasias.
When EGFR inhibitor–induced acneiform eruption occurs, patients commonly develop skin toxicity within the first 2 to 4 weeks of therapy. Pre-therapy doxycycline or minocycline and/or topical steroids may help prevent toxicities from occurring. These same therapies may be used to treat symptoms after they have occurred. More severe cases with systemic symptoms or failure to improve with the above measures may need prednisone or cessation of therapy.
This patient was started on topical hydrocortisone 2.5% ointment twice daily and oral doxycycline 100 mg bid for 6 weeks. She had dramatic improvement within 3 weeks. Doxycycline was subsequently continued at a dose of 100 mg/d and the patient was able to continue with her chemotherapy combination for several more months. Unfortunately, her colon cancer progressed despite therapy and she ultimately died from cancer-related complications.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Bouché O, Ben Abdelghani M, Labourey JL, et al. Management of skin toxicities during panitumumab treatment in metastatic colorectal cancer. World J Gastroenterol. 2019;25:4007-4018. doi: 10.3748/wjg.v25.i29.4007

A review of the patient’s chemotherapy medications revealed that 4 weeks earlier, panitumumab had been added to her folinic acid, fluorouracil, and irinotecan (FOLFIRI) regimen. The physician diagnosed this acneiform eruption as an adverse effect of the panitumumab.
Panitumumab is a monoclonal antibody that works to inhibit epidermal growth factor receptor (EGFR) proteins that are overexpressed on some solid tumors and responsible for cancer cell proliferation. EGFR inhibitor–induced acneiform eruptions are common in patients receiving panitumumab.
EGFR proteins have been a target of chemotherapy since the approval of the small molecule erlotinib in 2004. Panitumumab and cetuximab are monoclonal antibodies targeting EGFR and improve long-term survival in patients with metastatic colorectal cancer when added to other standard chemotherapy regimens. EGFR is found throughout the epidermis and all EGFR inhibitors may cause unique skin toxicity not seen with other chemotherapy agents. In 1 study of 229 patients, 59% of patients exhibited skin toxicity at Day 15; the most common examples included widespread acne-like papules and pustules or an eczema-like manifestation.1 Eruptions may be worsened by significant sun exposure while on panitumumab. In this case, the acneiform eruption occurred more intensely along visible facial telangiectasias.
When EGFR inhibitor–induced acneiform eruption occurs, patients commonly develop skin toxicity within the first 2 to 4 weeks of therapy. Pre-therapy doxycycline or minocycline and/or topical steroids may help prevent toxicities from occurring. These same therapies may be used to treat symptoms after they have occurred. More severe cases with systemic symptoms or failure to improve with the above measures may need prednisone or cessation of therapy.
This patient was started on topical hydrocortisone 2.5% ointment twice daily and oral doxycycline 100 mg bid for 6 weeks. She had dramatic improvement within 3 weeks. Doxycycline was subsequently continued at a dose of 100 mg/d and the patient was able to continue with her chemotherapy combination for several more months. Unfortunately, her colon cancer progressed despite therapy and she ultimately died from cancer-related complications.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

A review of the patient’s chemotherapy medications revealed that 4 weeks earlier, panitumumab had been added to her folinic acid, fluorouracil, and irinotecan (FOLFIRI) regimen. The physician diagnosed this acneiform eruption as an adverse effect of the panitumumab.
Panitumumab is a monoclonal antibody that works to inhibit epidermal growth factor receptor (EGFR) proteins that are overexpressed on some solid tumors and responsible for cancer cell proliferation. EGFR inhibitor–induced acneiform eruptions are common in patients receiving panitumumab.
EGFR proteins have been a target of chemotherapy since the approval of the small molecule erlotinib in 2004. Panitumumab and cetuximab are monoclonal antibodies targeting EGFR and improve long-term survival in patients with metastatic colorectal cancer when added to other standard chemotherapy regimens. EGFR is found throughout the epidermis and all EGFR inhibitors may cause unique skin toxicity not seen with other chemotherapy agents. In 1 study of 229 patients, 59% of patients exhibited skin toxicity at Day 15; the most common examples included widespread acne-like papules and pustules or an eczema-like manifestation.1 Eruptions may be worsened by significant sun exposure while on panitumumab. In this case, the acneiform eruption occurred more intensely along visible facial telangiectasias.
When EGFR inhibitor–induced acneiform eruption occurs, patients commonly develop skin toxicity within the first 2 to 4 weeks of therapy. Pre-therapy doxycycline or minocycline and/or topical steroids may help prevent toxicities from occurring. These same therapies may be used to treat symptoms after they have occurred. More severe cases with systemic symptoms or failure to improve with the above measures may need prednisone or cessation of therapy.
This patient was started on topical hydrocortisone 2.5% ointment twice daily and oral doxycycline 100 mg bid for 6 weeks. She had dramatic improvement within 3 weeks. Doxycycline was subsequently continued at a dose of 100 mg/d and the patient was able to continue with her chemotherapy combination for several more months. Unfortunately, her colon cancer progressed despite therapy and she ultimately died from cancer-related complications.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Bouché O, Ben Abdelghani M, Labourey JL, et al. Management of skin toxicities during panitumumab treatment in metastatic colorectal cancer. World J Gastroenterol. 2019;25:4007-4018. doi: 10.3748/wjg.v25.i29.4007
1. Bouché O, Ben Abdelghani M, Labourey JL, et al. Management of skin toxicities during panitumumab treatment in metastatic colorectal cancer. World J Gastroenterol. 2019;25:4007-4018. doi: 10.3748/wjg.v25.i29.4007
Could combining topical antioxidants with a nonablative laser prevent acne scars?
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
AT ASLMS 2023
Meta-analysis examines cancer risk concern for JAK inhibitors
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
AT BSR 2023
Small study finds IPL-radiofrequency combination effective for dry eye disease
PHOENIX – and improved meibum quality in both upper and lower eyelids, results from an ongoing, novel study showed.
Dry eye disease affects a large proportion of people in the United States “and the factors that contribute to that are certainly not going away,” lead study author James G. Chelnis MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where he presented the results during an abstract session. “Prepandemic, we used to have meetings in person; now most are on a computer screen,” a common risk factor for worsening dry eyes, he said. Telltale dry eye symptoms include blurry vision, irritation, and corneal damage – mostly caused by meibomian gland dysfunction – which impacts the quality and quantity of meibum secreted. Common treatments include warm compresses, doxycycline, and artificial tears.
While some studies have shown IPL is helpful in treating dry eye disease caused by meibomian gland dysfunction, little information is available on its use alone or in combination with topical RF to preserve and improve the function of meibomian glands, said Dr. Chelnis, an ophthalmic plastic surgeon in New York City. “The theory here is that the radiofrequency would be able to vibrate the water molecules inside the meibomian glands, which would allow you to turn over the meibum faster, as well as improve the blink reflex response by building supporting collagen,” he said. “Our novel study explores the ability of this combined modality treatment to improve upon meibomian gland health.”
Study design, results
Dr. Chelnis and his colleagues enrolled 11 individuals with a previous diagnosis of dry eye disease and meibomian gland dysfunction with Ocular Surface Disease Index (OSDI) survey scores higher than 23, which indicate at least moderate dry eye symptoms. Inclusion criteria were being 22 years of age or older, signs of meibomian gland dysfunction as detected by biomicroscopy, a modified meibomian gland score over 12 in the lower eyelid of at least one eye, and type I-IV skin.
All patients received four treatments (each 2 weeks apart) of IPL to the lower eyelid, surrounding malar region, and nose, followed by 7 minutes of topical RF treatments at 1 MHz and 4 MHz extending to the inferior, lateral, and superior orbital rim. Evaluation of meibomian gland expression and quality of meibum upon expression was conducted following each treatment session, with a final evaluation 4 weeks after the final treatment session.
Meibum quality was evaluated on a scale of 0-3 representing clear (0), cloudy (1), inspissated (2), and blocked (3) meibum, respectively.
Following treatment, meibomian gland expression and meibum quality improved in all eyelids in all 11 patients. Specifically, in the right eye, the number of upper lid expressible glands increased from an average of 13 to 27.9 and the number of lower lid expressible glands increased from an average of 14.6 to 28.2; and in the left eye, the number of upper lid expressible glands increased from an average of 13.3 to 27.3 and the number of lower lid expressible glands increased from an average of 14.8 to 26.8 (P < .001 for all associations).
The overall percentage improvement in meibomian gland expression in the right eye was 82.7% for the upper lids and 136.6% for the lower lids, and in the left eye, 82.9% for the upper lids, and 112.2% for the lower lids.
When comparing upper against lower lids, meibomian gland expression increased 124.4% and 82.8%, respectively. Meibum quality improved in all four eyelids, although upper eyelids displayed a superior improvement compared with lower eyelids.
“We are finding that combining IPL plus RF produces a more complete and comprehensive improvement in the quality of their meibomian gland health, and as such, their dry eyes,” with “a large decrease in their symptom profile,” he concluded.
More patients to be studied
Dr. Chelnis acknowledged certain limitations of the study, including the small number of patients, but he and his colleagues have added an additional clinical site to expand the sample size. “Larger scale studies are needed to evaluate long-term effectiveness of IPL plus RF as well as a comparison with other treatment options.”
During a question-and-answer session Mathew M. Avram, MD, JD, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital, Boston, who served as one of the abstract session moderators, asked Dr. Chelnis to comment on what the mechanism of action of the IPL-RF combination in improving meibomian gland health.
“It’s not fully understood, but part of it is improved vascularity at the lid margin,” said Dr. Chelnis, who holds a faculty position in the department of ophthalmology at Icahn School of Medicine at Mount Sinai, New York. “Your ocular surface is sort of like your screen door; it catches everything that’s in the environment. An increase in vascularity and immunologic cytokines occurs in response to that. If you’re looking at the eye with a slit lamp, you can see a lot of vascularity that occurs at the lid margin and crowds the meibomian glands. When you decrease that crowding and immunogenic response, you move towards a normally functioning lid margin.”
Dr. Chelnis disclosed that he is a consultant to or an adviser for Lumenis, Horizon Therapeutics, and Soniquence.
PHOENIX – and improved meibum quality in both upper and lower eyelids, results from an ongoing, novel study showed.
Dry eye disease affects a large proportion of people in the United States “and the factors that contribute to that are certainly not going away,” lead study author James G. Chelnis MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where he presented the results during an abstract session. “Prepandemic, we used to have meetings in person; now most are on a computer screen,” a common risk factor for worsening dry eyes, he said. Telltale dry eye symptoms include blurry vision, irritation, and corneal damage – mostly caused by meibomian gland dysfunction – which impacts the quality and quantity of meibum secreted. Common treatments include warm compresses, doxycycline, and artificial tears.
While some studies have shown IPL is helpful in treating dry eye disease caused by meibomian gland dysfunction, little information is available on its use alone or in combination with topical RF to preserve and improve the function of meibomian glands, said Dr. Chelnis, an ophthalmic plastic surgeon in New York City. “The theory here is that the radiofrequency would be able to vibrate the water molecules inside the meibomian glands, which would allow you to turn over the meibum faster, as well as improve the blink reflex response by building supporting collagen,” he said. “Our novel study explores the ability of this combined modality treatment to improve upon meibomian gland health.”
Study design, results
Dr. Chelnis and his colleagues enrolled 11 individuals with a previous diagnosis of dry eye disease and meibomian gland dysfunction with Ocular Surface Disease Index (OSDI) survey scores higher than 23, which indicate at least moderate dry eye symptoms. Inclusion criteria were being 22 years of age or older, signs of meibomian gland dysfunction as detected by biomicroscopy, a modified meibomian gland score over 12 in the lower eyelid of at least one eye, and type I-IV skin.
All patients received four treatments (each 2 weeks apart) of IPL to the lower eyelid, surrounding malar region, and nose, followed by 7 minutes of topical RF treatments at 1 MHz and 4 MHz extending to the inferior, lateral, and superior orbital rim. Evaluation of meibomian gland expression and quality of meibum upon expression was conducted following each treatment session, with a final evaluation 4 weeks after the final treatment session.
Meibum quality was evaluated on a scale of 0-3 representing clear (0), cloudy (1), inspissated (2), and blocked (3) meibum, respectively.
Following treatment, meibomian gland expression and meibum quality improved in all eyelids in all 11 patients. Specifically, in the right eye, the number of upper lid expressible glands increased from an average of 13 to 27.9 and the number of lower lid expressible glands increased from an average of 14.6 to 28.2; and in the left eye, the number of upper lid expressible glands increased from an average of 13.3 to 27.3 and the number of lower lid expressible glands increased from an average of 14.8 to 26.8 (P < .001 for all associations).
The overall percentage improvement in meibomian gland expression in the right eye was 82.7% for the upper lids and 136.6% for the lower lids, and in the left eye, 82.9% for the upper lids, and 112.2% for the lower lids.
When comparing upper against lower lids, meibomian gland expression increased 124.4% and 82.8%, respectively. Meibum quality improved in all four eyelids, although upper eyelids displayed a superior improvement compared with lower eyelids.
“We are finding that combining IPL plus RF produces a more complete and comprehensive improvement in the quality of their meibomian gland health, and as such, their dry eyes,” with “a large decrease in their symptom profile,” he concluded.
More patients to be studied
Dr. Chelnis acknowledged certain limitations of the study, including the small number of patients, but he and his colleagues have added an additional clinical site to expand the sample size. “Larger scale studies are needed to evaluate long-term effectiveness of IPL plus RF as well as a comparison with other treatment options.”
During a question-and-answer session Mathew M. Avram, MD, JD, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital, Boston, who served as one of the abstract session moderators, asked Dr. Chelnis to comment on what the mechanism of action of the IPL-RF combination in improving meibomian gland health.
“It’s not fully understood, but part of it is improved vascularity at the lid margin,” said Dr. Chelnis, who holds a faculty position in the department of ophthalmology at Icahn School of Medicine at Mount Sinai, New York. “Your ocular surface is sort of like your screen door; it catches everything that’s in the environment. An increase in vascularity and immunologic cytokines occurs in response to that. If you’re looking at the eye with a slit lamp, you can see a lot of vascularity that occurs at the lid margin and crowds the meibomian glands. When you decrease that crowding and immunogenic response, you move towards a normally functioning lid margin.”
Dr. Chelnis disclosed that he is a consultant to or an adviser for Lumenis, Horizon Therapeutics, and Soniquence.
PHOENIX – and improved meibum quality in both upper and lower eyelids, results from an ongoing, novel study showed.
Dry eye disease affects a large proportion of people in the United States “and the factors that contribute to that are certainly not going away,” lead study author James G. Chelnis MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where he presented the results during an abstract session. “Prepandemic, we used to have meetings in person; now most are on a computer screen,” a common risk factor for worsening dry eyes, he said. Telltale dry eye symptoms include blurry vision, irritation, and corneal damage – mostly caused by meibomian gland dysfunction – which impacts the quality and quantity of meibum secreted. Common treatments include warm compresses, doxycycline, and artificial tears.
While some studies have shown IPL is helpful in treating dry eye disease caused by meibomian gland dysfunction, little information is available on its use alone or in combination with topical RF to preserve and improve the function of meibomian glands, said Dr. Chelnis, an ophthalmic plastic surgeon in New York City. “The theory here is that the radiofrequency would be able to vibrate the water molecules inside the meibomian glands, which would allow you to turn over the meibum faster, as well as improve the blink reflex response by building supporting collagen,” he said. “Our novel study explores the ability of this combined modality treatment to improve upon meibomian gland health.”
Study design, results
Dr. Chelnis and his colleagues enrolled 11 individuals with a previous diagnosis of dry eye disease and meibomian gland dysfunction with Ocular Surface Disease Index (OSDI) survey scores higher than 23, which indicate at least moderate dry eye symptoms. Inclusion criteria were being 22 years of age or older, signs of meibomian gland dysfunction as detected by biomicroscopy, a modified meibomian gland score over 12 in the lower eyelid of at least one eye, and type I-IV skin.
All patients received four treatments (each 2 weeks apart) of IPL to the lower eyelid, surrounding malar region, and nose, followed by 7 minutes of topical RF treatments at 1 MHz and 4 MHz extending to the inferior, lateral, and superior orbital rim. Evaluation of meibomian gland expression and quality of meibum upon expression was conducted following each treatment session, with a final evaluation 4 weeks after the final treatment session.
Meibum quality was evaluated on a scale of 0-3 representing clear (0), cloudy (1), inspissated (2), and blocked (3) meibum, respectively.
Following treatment, meibomian gland expression and meibum quality improved in all eyelids in all 11 patients. Specifically, in the right eye, the number of upper lid expressible glands increased from an average of 13 to 27.9 and the number of lower lid expressible glands increased from an average of 14.6 to 28.2; and in the left eye, the number of upper lid expressible glands increased from an average of 13.3 to 27.3 and the number of lower lid expressible glands increased from an average of 14.8 to 26.8 (P < .001 for all associations).
The overall percentage improvement in meibomian gland expression in the right eye was 82.7% for the upper lids and 136.6% for the lower lids, and in the left eye, 82.9% for the upper lids, and 112.2% for the lower lids.
When comparing upper against lower lids, meibomian gland expression increased 124.4% and 82.8%, respectively. Meibum quality improved in all four eyelids, although upper eyelids displayed a superior improvement compared with lower eyelids.
“We are finding that combining IPL plus RF produces a more complete and comprehensive improvement in the quality of their meibomian gland health, and as such, their dry eyes,” with “a large decrease in their symptom profile,” he concluded.
More patients to be studied
Dr. Chelnis acknowledged certain limitations of the study, including the small number of patients, but he and his colleagues have added an additional clinical site to expand the sample size. “Larger scale studies are needed to evaluate long-term effectiveness of IPL plus RF as well as a comparison with other treatment options.”
During a question-and-answer session Mathew M. Avram, MD, JD, director of laser, cosmetics, and dermatologic surgery at Massachusetts General Hospital, Boston, who served as one of the abstract session moderators, asked Dr. Chelnis to comment on what the mechanism of action of the IPL-RF combination in improving meibomian gland health.
“It’s not fully understood, but part of it is improved vascularity at the lid margin,” said Dr. Chelnis, who holds a faculty position in the department of ophthalmology at Icahn School of Medicine at Mount Sinai, New York. “Your ocular surface is sort of like your screen door; it catches everything that’s in the environment. An increase in vascularity and immunologic cytokines occurs in response to that. If you’re looking at the eye with a slit lamp, you can see a lot of vascularity that occurs at the lid margin and crowds the meibomian glands. When you decrease that crowding and immunogenic response, you move towards a normally functioning lid margin.”
Dr. Chelnis disclosed that he is a consultant to or an adviser for Lumenis, Horizon Therapeutics, and Soniquence.
AT ASLMS 2023
Guidelines for assessing cancer risk may need updating
The authors of the clinical trial suggest that these guidelines may need to be revised.
Individuals with hereditary breast and ovarian cancer (HBOC) have an 80% lifetime risk of breast cancer and are at greater risk of ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Those with Lynch syndrome (LS) have an 80% lifetime risk of colorectal cancer, a 60% lifetime risk of endometrial cancer, and heightened risk of upper gastrointestinal, urinary tract, skin, and other tumors, said study coauthor N. Jewel Samadder, MD in a statement.
The National Cancer Control Network has guidelines for determining family risk for colorectal cancer and breast, ovarian, and pancreatic cancer to identify individuals who should be screened for LS and HBOC, but these rely on personal and family health histories.
“These criteria were created at a time when genetic testing was cost prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing. However, [LS and HBOC] are poorly identified in current practice, and many patients are not aware of their cancer risk,” said Dr. Samadder, professor of medicine and coleader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center, Phoenix, in the statement.
Whole-exome sequencing covers only protein-coding regions of the genome, which is less than 2% of the total genome but includes more than 85% of known disease-related genetic variants, according to Emily Gay, who presented the trial results (Abstract 5768) on April 18 at the annual meeting of the American Association for Cancer Research.
“In recent years, the cost of whole-exome sequencing has been rapidly decreasing, allowing us to complete this test on saliva samples from thousands, if not tens of thousands of patients covering large populations and large health systems,” said Ms. Gay, a genetic counseling graduate student at the University of Arizona, during her presentation.
She described results from the TAPESTRY clinical trial, with 44,306 participants from Mayo Clinic centers in Arizona, Florida, and Minnesota, who were identified as definitely or likely to be harboring pathogenic mutations and consented to whole-exome sequencing from saliva samples. They used electronic health records to determine whether patients would satisfy the testing criteria from NCCN guidelines.
The researchers identified 1.24% of participants to be carriers of HBOC or LS. Of the HBOC carriers, 62.8% were female, and of the LS carriers, 62.6% were female. The percentages of HBOC and LS carriers who were White were 88.6 and 94.5, respectively. The median age of both groups was 57 years. Of HBOC carriers, 47.3% had personal histories of cancers; for LS carries, the percentage was 44.2.
Of HBOC carriers, 49.1% had been previously unaware of their genetic condition, while an even higher percentage of patients with LS – 59.3% – fell into that category. Thirty-two percent of those with HBOC and 56.2% of those with LS would not have qualified for screening using the relevant NCCN guidelines.
“Most strikingly,” 63.8% of individuals with mutations in the MSH6 gene and 83.7% of those mutations in the PMS2 gene would not have met NCCN criteria, Ms. Gay said.
Having a cancer type not known to be related to a genetic syndrome was a reason for 58.6% of individuals failing to meet NCCN guidelines, while 60.5% did not meet the guidelines because of an insufficient number of relatives known to have a history of cancer, and 63.3% did not because they had no personal history of cancer. Among individuals with a pathogenic mutation who met NCCN criteria, 34% were not aware of their condition.
“This suggests that the NCCN guidelines are underutilized in clinical practice, potentially due to the busy schedule of clinicians or because the complexity of using these criteria,” said Ms. Gay.
The numbers were even more striking among minorities: “There is additional data analysis and research needed in this area, but based on our preliminary findings, we saw that nearly 50% of the individuals who are [part of an underrepresented minority group] did not meet criteria, compared with 32% of the white cohort,” said Ms. Gay.
Asked what new NCCN guidelines should be, Ms. Gay replied: “I think maybe limiting the number of relatives that you have to have with a certain type of cancer, especially as we see families get smaller and smaller, especially in the United States – that family data isn’t necessarily available or as useful. And then also, I think, incorporating in the size of a family into the calculation, so more of maybe a point-based system like we see with other genetic conditions rather than a ‘yes you meet or no, you don’t.’ More of a range to say ‘you fall on the low-risk, medium-risk, or high-risk stage,’” said Ms. Gay.
During the Q&A period, session cochair Andrew Godwin, PhD, who is a professor of molecular oncology and pathology at University of Kansas Medical Center, Kansas City, said he wondered if whole-exome sequencing was capable of picking up cancer risk mutations that standard targeted tests don’t look for.
Dr. Samadder, who was in the audience, answered the question, saying that targeted tests are actually better at picking up some types of mutations like intronic mutations, single-nucleotide polymorphisms, and deletions.
“There are some limitations to whole-exome sequencing. Our estimate here of 1.2% [of participants carrying HBOC or LS mutations] is probably an underestimate. There are additional variants that exome sequencing probably doesn’t pick up easily or as well. That’s why we qualify that exome sequencing is a screening test, not a diagnostic,” he continued.
Ms. Gay and Dr. Samadder have no relevant financial disclosures. Dr. Godwin has financial relationships with Clara Biotech, VITRAC Therapeutics, and Sinochips Diagnostics.
The authors of the clinical trial suggest that these guidelines may need to be revised.
Individuals with hereditary breast and ovarian cancer (HBOC) have an 80% lifetime risk of breast cancer and are at greater risk of ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Those with Lynch syndrome (LS) have an 80% lifetime risk of colorectal cancer, a 60% lifetime risk of endometrial cancer, and heightened risk of upper gastrointestinal, urinary tract, skin, and other tumors, said study coauthor N. Jewel Samadder, MD in a statement.
The National Cancer Control Network has guidelines for determining family risk for colorectal cancer and breast, ovarian, and pancreatic cancer to identify individuals who should be screened for LS and HBOC, but these rely on personal and family health histories.
“These criteria were created at a time when genetic testing was cost prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing. However, [LS and HBOC] are poorly identified in current practice, and many patients are not aware of their cancer risk,” said Dr. Samadder, professor of medicine and coleader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center, Phoenix, in the statement.
Whole-exome sequencing covers only protein-coding regions of the genome, which is less than 2% of the total genome but includes more than 85% of known disease-related genetic variants, according to Emily Gay, who presented the trial results (Abstract 5768) on April 18 at the annual meeting of the American Association for Cancer Research.
“In recent years, the cost of whole-exome sequencing has been rapidly decreasing, allowing us to complete this test on saliva samples from thousands, if not tens of thousands of patients covering large populations and large health systems,” said Ms. Gay, a genetic counseling graduate student at the University of Arizona, during her presentation.
She described results from the TAPESTRY clinical trial, with 44,306 participants from Mayo Clinic centers in Arizona, Florida, and Minnesota, who were identified as definitely or likely to be harboring pathogenic mutations and consented to whole-exome sequencing from saliva samples. They used electronic health records to determine whether patients would satisfy the testing criteria from NCCN guidelines.
The researchers identified 1.24% of participants to be carriers of HBOC or LS. Of the HBOC carriers, 62.8% were female, and of the LS carriers, 62.6% were female. The percentages of HBOC and LS carriers who were White were 88.6 and 94.5, respectively. The median age of both groups was 57 years. Of HBOC carriers, 47.3% had personal histories of cancers; for LS carries, the percentage was 44.2.
Of HBOC carriers, 49.1% had been previously unaware of their genetic condition, while an even higher percentage of patients with LS – 59.3% – fell into that category. Thirty-two percent of those with HBOC and 56.2% of those with LS would not have qualified for screening using the relevant NCCN guidelines.
“Most strikingly,” 63.8% of individuals with mutations in the MSH6 gene and 83.7% of those mutations in the PMS2 gene would not have met NCCN criteria, Ms. Gay said.
Having a cancer type not known to be related to a genetic syndrome was a reason for 58.6% of individuals failing to meet NCCN guidelines, while 60.5% did not meet the guidelines because of an insufficient number of relatives known to have a history of cancer, and 63.3% did not because they had no personal history of cancer. Among individuals with a pathogenic mutation who met NCCN criteria, 34% were not aware of their condition.
“This suggests that the NCCN guidelines are underutilized in clinical practice, potentially due to the busy schedule of clinicians or because the complexity of using these criteria,” said Ms. Gay.
The numbers were even more striking among minorities: “There is additional data analysis and research needed in this area, but based on our preliminary findings, we saw that nearly 50% of the individuals who are [part of an underrepresented minority group] did not meet criteria, compared with 32% of the white cohort,” said Ms. Gay.
Asked what new NCCN guidelines should be, Ms. Gay replied: “I think maybe limiting the number of relatives that you have to have with a certain type of cancer, especially as we see families get smaller and smaller, especially in the United States – that family data isn’t necessarily available or as useful. And then also, I think, incorporating in the size of a family into the calculation, so more of maybe a point-based system like we see with other genetic conditions rather than a ‘yes you meet or no, you don’t.’ More of a range to say ‘you fall on the low-risk, medium-risk, or high-risk stage,’” said Ms. Gay.
During the Q&A period, session cochair Andrew Godwin, PhD, who is a professor of molecular oncology and pathology at University of Kansas Medical Center, Kansas City, said he wondered if whole-exome sequencing was capable of picking up cancer risk mutations that standard targeted tests don’t look for.
Dr. Samadder, who was in the audience, answered the question, saying that targeted tests are actually better at picking up some types of mutations like intronic mutations, single-nucleotide polymorphisms, and deletions.
“There are some limitations to whole-exome sequencing. Our estimate here of 1.2% [of participants carrying HBOC or LS mutations] is probably an underestimate. There are additional variants that exome sequencing probably doesn’t pick up easily or as well. That’s why we qualify that exome sequencing is a screening test, not a diagnostic,” he continued.
Ms. Gay and Dr. Samadder have no relevant financial disclosures. Dr. Godwin has financial relationships with Clara Biotech, VITRAC Therapeutics, and Sinochips Diagnostics.
The authors of the clinical trial suggest that these guidelines may need to be revised.
Individuals with hereditary breast and ovarian cancer (HBOC) have an 80% lifetime risk of breast cancer and are at greater risk of ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Those with Lynch syndrome (LS) have an 80% lifetime risk of colorectal cancer, a 60% lifetime risk of endometrial cancer, and heightened risk of upper gastrointestinal, urinary tract, skin, and other tumors, said study coauthor N. Jewel Samadder, MD in a statement.
The National Cancer Control Network has guidelines for determining family risk for colorectal cancer and breast, ovarian, and pancreatic cancer to identify individuals who should be screened for LS and HBOC, but these rely on personal and family health histories.
“These criteria were created at a time when genetic testing was cost prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing. However, [LS and HBOC] are poorly identified in current practice, and many patients are not aware of their cancer risk,” said Dr. Samadder, professor of medicine and coleader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center, Phoenix, in the statement.
Whole-exome sequencing covers only protein-coding regions of the genome, which is less than 2% of the total genome but includes more than 85% of known disease-related genetic variants, according to Emily Gay, who presented the trial results (Abstract 5768) on April 18 at the annual meeting of the American Association for Cancer Research.
“In recent years, the cost of whole-exome sequencing has been rapidly decreasing, allowing us to complete this test on saliva samples from thousands, if not tens of thousands of patients covering large populations and large health systems,” said Ms. Gay, a genetic counseling graduate student at the University of Arizona, during her presentation.
She described results from the TAPESTRY clinical trial, with 44,306 participants from Mayo Clinic centers in Arizona, Florida, and Minnesota, who were identified as definitely or likely to be harboring pathogenic mutations and consented to whole-exome sequencing from saliva samples. They used electronic health records to determine whether patients would satisfy the testing criteria from NCCN guidelines.
The researchers identified 1.24% of participants to be carriers of HBOC or LS. Of the HBOC carriers, 62.8% were female, and of the LS carriers, 62.6% were female. The percentages of HBOC and LS carriers who were White were 88.6 and 94.5, respectively. The median age of both groups was 57 years. Of HBOC carriers, 47.3% had personal histories of cancers; for LS carries, the percentage was 44.2.
Of HBOC carriers, 49.1% had been previously unaware of their genetic condition, while an even higher percentage of patients with LS – 59.3% – fell into that category. Thirty-two percent of those with HBOC and 56.2% of those with LS would not have qualified for screening using the relevant NCCN guidelines.
“Most strikingly,” 63.8% of individuals with mutations in the MSH6 gene and 83.7% of those mutations in the PMS2 gene would not have met NCCN criteria, Ms. Gay said.
Having a cancer type not known to be related to a genetic syndrome was a reason for 58.6% of individuals failing to meet NCCN guidelines, while 60.5% did not meet the guidelines because of an insufficient number of relatives known to have a history of cancer, and 63.3% did not because they had no personal history of cancer. Among individuals with a pathogenic mutation who met NCCN criteria, 34% were not aware of their condition.
“This suggests that the NCCN guidelines are underutilized in clinical practice, potentially due to the busy schedule of clinicians or because the complexity of using these criteria,” said Ms. Gay.
The numbers were even more striking among minorities: “There is additional data analysis and research needed in this area, but based on our preliminary findings, we saw that nearly 50% of the individuals who are [part of an underrepresented minority group] did not meet criteria, compared with 32% of the white cohort,” said Ms. Gay.
Asked what new NCCN guidelines should be, Ms. Gay replied: “I think maybe limiting the number of relatives that you have to have with a certain type of cancer, especially as we see families get smaller and smaller, especially in the United States – that family data isn’t necessarily available or as useful. And then also, I think, incorporating in the size of a family into the calculation, so more of maybe a point-based system like we see with other genetic conditions rather than a ‘yes you meet or no, you don’t.’ More of a range to say ‘you fall on the low-risk, medium-risk, or high-risk stage,’” said Ms. Gay.
During the Q&A period, session cochair Andrew Godwin, PhD, who is a professor of molecular oncology and pathology at University of Kansas Medical Center, Kansas City, said he wondered if whole-exome sequencing was capable of picking up cancer risk mutations that standard targeted tests don’t look for.
Dr. Samadder, who was in the audience, answered the question, saying that targeted tests are actually better at picking up some types of mutations like intronic mutations, single-nucleotide polymorphisms, and deletions.
“There are some limitations to whole-exome sequencing. Our estimate here of 1.2% [of participants carrying HBOC or LS mutations] is probably an underestimate. There are additional variants that exome sequencing probably doesn’t pick up easily or as well. That’s why we qualify that exome sequencing is a screening test, not a diagnostic,” he continued.
Ms. Gay and Dr. Samadder have no relevant financial disclosures. Dr. Godwin has financial relationships with Clara Biotech, VITRAC Therapeutics, and Sinochips Diagnostics.
FROM AACR 2023
Study focuses on adolescent data in upadacitinib AD trials
(AD), an analysis of three clinical trials reports.
Upadacitinib (Rinvoq) was approved by the Food and Drug Administration for treating adults and pediatric patients 12 years of age and older with refractory, moderate to severe AD, in January 2022. This study analyzed the adolescent data in three double-blind, placebo-controlled phase 3 randomized clinical trials, which included adults and 552 adolescents between 12 and 17 years of age with moderate to severe AD in more than 20 countries in Europe, North and South America, the Middle East, Oceania, and the Asia-Pacific region from July 2018 through December 2020.
In the studies, “treatment of moderate to severe AD in adolescents with upadacitinib was effective and generally well tolerated, with an overall efficacy and safety profile similar to that observed in adults, and patient-reported outcomes indicated an overall better health-related quality of life compared with placebo,” lead study author Amy S. Paller, MD, chair of the department of dermatology and professor of dermatology and pediatrics, at Northwestern University, Chicago, and her colleagues write in JAMA Dermatology.
Adolescents in the three studies – Measure Up 1, Measure Up 2, and AD Up – received once-daily oral upadacitinib 15 mg, 30 mg, or placebo. All participants in AD Up used topical corticosteroids.
At 16 weeks, in Measure Up 1, Measure Up 2, and AD Up, respectively, a greater proportion of adolescents improved by at least 75% in the Eczema Area and Severity Index (EASI 75) with upadacitinib 15 mg (73%, 69%, 63%); and with upadacitinib 30 mg (78%, 73%, 84%), compared with placebo (12%, 13%, 30%), (P < .001 for all comparisons vs. placebo).
Upadacitinib was generally well tolerated among the adolescents, with mild or moderate acne being the most common adverse event, reported in 10%-13% of those on 15 mg and 15%-16% of those on 30 mg vs. 2%-3% of those on placebo.
Asked to comment on the study, Peck Ong, MD, a pediatric allergist and immunologist at Children’s Hospital Los Angeles, said that he was not surprised by the drug’s effectiveness because JAK inhibitors are potent immunosuppressants. Strengths of the studies include the many pediatric participants, its international reach, and its use of standardized and validated measures, said Dr. Ong, who was not involved in the study.
“The effect of JAK inhibitors is more specific than traditional immunosuppressants such as cyclosporine and methotrexate but not as specific as biologics; therefore, long-term safety data are needed,” he advised. “16 weeks is a very short time to study a chronic disease like atopic dermatitis. We need safety data longer than 1 year.”
Given the disease’s potential impact on self-esteem, sleep, and other important areas of life, Sean Reynolds, MBBCH, a pediatric dermatologist at Children’s Mercy Kansas City (Mo.), welcomed the data on the newer pharmacologic agents.
“FDA-approved systemic treatment options for adolescents with AD are currently limited, which necessitates studies such as this that explore additional treatment options,” said Dr. Reynolds, who also was not involved in the study, told this news organization.
He added that oral upadacitinib may especially help patients who have not found relief with other topical or systemic treatments or who are needle phobic. While the overall efficacy and relatively mild side effects for most patients taking upadacitinib in the trials are encouraging, “the long-term efficacy and side effects in this population require further study, especially considering the limited systemic AD treatment options available in this age group,” he added.
“Given the reported use of other JAK inhibitors to treat myriad inflammatory skin conditions beyond atopic dermatitis, the potential use of upadacitinib and other JAK inhibitors to treat these skin diseases in children and adolescents represents an exciting area for future study in the field of pediatric dermatology,” Dr. Reynolds noted.
The study was funded by AbbVie, the developer and manufacturer of upadacitinib. Dr. Paller and almost all other authors reported relevant financial relationships with AbbVie and other pharmaceutical companies. Dr. Ong reported serving on an AbbVie advisory board, and Dr. Reynolds reported no conflict of interest with the study.
(AD), an analysis of three clinical trials reports.
Upadacitinib (Rinvoq) was approved by the Food and Drug Administration for treating adults and pediatric patients 12 years of age and older with refractory, moderate to severe AD, in January 2022. This study analyzed the adolescent data in three double-blind, placebo-controlled phase 3 randomized clinical trials, which included adults and 552 adolescents between 12 and 17 years of age with moderate to severe AD in more than 20 countries in Europe, North and South America, the Middle East, Oceania, and the Asia-Pacific region from July 2018 through December 2020.
In the studies, “treatment of moderate to severe AD in adolescents with upadacitinib was effective and generally well tolerated, with an overall efficacy and safety profile similar to that observed in adults, and patient-reported outcomes indicated an overall better health-related quality of life compared with placebo,” lead study author Amy S. Paller, MD, chair of the department of dermatology and professor of dermatology and pediatrics, at Northwestern University, Chicago, and her colleagues write in JAMA Dermatology.
Adolescents in the three studies – Measure Up 1, Measure Up 2, and AD Up – received once-daily oral upadacitinib 15 mg, 30 mg, or placebo. All participants in AD Up used topical corticosteroids.
At 16 weeks, in Measure Up 1, Measure Up 2, and AD Up, respectively, a greater proportion of adolescents improved by at least 75% in the Eczema Area and Severity Index (EASI 75) with upadacitinib 15 mg (73%, 69%, 63%); and with upadacitinib 30 mg (78%, 73%, 84%), compared with placebo (12%, 13%, 30%), (P < .001 for all comparisons vs. placebo).
Upadacitinib was generally well tolerated among the adolescents, with mild or moderate acne being the most common adverse event, reported in 10%-13% of those on 15 mg and 15%-16% of those on 30 mg vs. 2%-3% of those on placebo.
Asked to comment on the study, Peck Ong, MD, a pediatric allergist and immunologist at Children’s Hospital Los Angeles, said that he was not surprised by the drug’s effectiveness because JAK inhibitors are potent immunosuppressants. Strengths of the studies include the many pediatric participants, its international reach, and its use of standardized and validated measures, said Dr. Ong, who was not involved in the study.
“The effect of JAK inhibitors is more specific than traditional immunosuppressants such as cyclosporine and methotrexate but not as specific as biologics; therefore, long-term safety data are needed,” he advised. “16 weeks is a very short time to study a chronic disease like atopic dermatitis. We need safety data longer than 1 year.”
Given the disease’s potential impact on self-esteem, sleep, and other important areas of life, Sean Reynolds, MBBCH, a pediatric dermatologist at Children’s Mercy Kansas City (Mo.), welcomed the data on the newer pharmacologic agents.
“FDA-approved systemic treatment options for adolescents with AD are currently limited, which necessitates studies such as this that explore additional treatment options,” said Dr. Reynolds, who also was not involved in the study, told this news organization.
He added that oral upadacitinib may especially help patients who have not found relief with other topical or systemic treatments or who are needle phobic. While the overall efficacy and relatively mild side effects for most patients taking upadacitinib in the trials are encouraging, “the long-term efficacy and side effects in this population require further study, especially considering the limited systemic AD treatment options available in this age group,” he added.
“Given the reported use of other JAK inhibitors to treat myriad inflammatory skin conditions beyond atopic dermatitis, the potential use of upadacitinib and other JAK inhibitors to treat these skin diseases in children and adolescents represents an exciting area for future study in the field of pediatric dermatology,” Dr. Reynolds noted.
The study was funded by AbbVie, the developer and manufacturer of upadacitinib. Dr. Paller and almost all other authors reported relevant financial relationships with AbbVie and other pharmaceutical companies. Dr. Ong reported serving on an AbbVie advisory board, and Dr. Reynolds reported no conflict of interest with the study.
(AD), an analysis of three clinical trials reports.
Upadacitinib (Rinvoq) was approved by the Food and Drug Administration for treating adults and pediatric patients 12 years of age and older with refractory, moderate to severe AD, in January 2022. This study analyzed the adolescent data in three double-blind, placebo-controlled phase 3 randomized clinical trials, which included adults and 552 adolescents between 12 and 17 years of age with moderate to severe AD in more than 20 countries in Europe, North and South America, the Middle East, Oceania, and the Asia-Pacific region from July 2018 through December 2020.
In the studies, “treatment of moderate to severe AD in adolescents with upadacitinib was effective and generally well tolerated, with an overall efficacy and safety profile similar to that observed in adults, and patient-reported outcomes indicated an overall better health-related quality of life compared with placebo,” lead study author Amy S. Paller, MD, chair of the department of dermatology and professor of dermatology and pediatrics, at Northwestern University, Chicago, and her colleagues write in JAMA Dermatology.
Adolescents in the three studies – Measure Up 1, Measure Up 2, and AD Up – received once-daily oral upadacitinib 15 mg, 30 mg, or placebo. All participants in AD Up used topical corticosteroids.
At 16 weeks, in Measure Up 1, Measure Up 2, and AD Up, respectively, a greater proportion of adolescents improved by at least 75% in the Eczema Area and Severity Index (EASI 75) with upadacitinib 15 mg (73%, 69%, 63%); and with upadacitinib 30 mg (78%, 73%, 84%), compared with placebo (12%, 13%, 30%), (P < .001 for all comparisons vs. placebo).
Upadacitinib was generally well tolerated among the adolescents, with mild or moderate acne being the most common adverse event, reported in 10%-13% of those on 15 mg and 15%-16% of those on 30 mg vs. 2%-3% of those on placebo.
Asked to comment on the study, Peck Ong, MD, a pediatric allergist and immunologist at Children’s Hospital Los Angeles, said that he was not surprised by the drug’s effectiveness because JAK inhibitors are potent immunosuppressants. Strengths of the studies include the many pediatric participants, its international reach, and its use of standardized and validated measures, said Dr. Ong, who was not involved in the study.
“The effect of JAK inhibitors is more specific than traditional immunosuppressants such as cyclosporine and methotrexate but not as specific as biologics; therefore, long-term safety data are needed,” he advised. “16 weeks is a very short time to study a chronic disease like atopic dermatitis. We need safety data longer than 1 year.”
Given the disease’s potential impact on self-esteem, sleep, and other important areas of life, Sean Reynolds, MBBCH, a pediatric dermatologist at Children’s Mercy Kansas City (Mo.), welcomed the data on the newer pharmacologic agents.
“FDA-approved systemic treatment options for adolescents with AD are currently limited, which necessitates studies such as this that explore additional treatment options,” said Dr. Reynolds, who also was not involved in the study, told this news organization.
He added that oral upadacitinib may especially help patients who have not found relief with other topical or systemic treatments or who are needle phobic. While the overall efficacy and relatively mild side effects for most patients taking upadacitinib in the trials are encouraging, “the long-term efficacy and side effects in this population require further study, especially considering the limited systemic AD treatment options available in this age group,” he added.
“Given the reported use of other JAK inhibitors to treat myriad inflammatory skin conditions beyond atopic dermatitis, the potential use of upadacitinib and other JAK inhibitors to treat these skin diseases in children and adolescents represents an exciting area for future study in the field of pediatric dermatology,” Dr. Reynolds noted.
The study was funded by AbbVie, the developer and manufacturer of upadacitinib. Dr. Paller and almost all other authors reported relevant financial relationships with AbbVie and other pharmaceutical companies. Dr. Ong reported serving on an AbbVie advisory board, and Dr. Reynolds reported no conflict of interest with the study.
FROM JAMA DERMATOLOGY
‘Exciting’ results for cancer vaccine plus pembro in melanoma
according to the latest data from the KEYNOTE-942 trial.
This recurrence-free survival benefit corresponded to a 44% reduced risk of recurrence or death in patients who received the personalized vaccine plus pembrolizumab compared with the immunotherapy alone.
The randomized phase 2b trial is the first to show a positive result for a cancer vaccine in a randomized trial. The results, if confirmed in further studies, hold promise for treating other solid tumors with sensitivity to the programmed death-1 (PD-1) protein, investigators said.
“KEYNOTE-942 is the first randomized study to demonstrate improvement in recurrence-free survival in melanoma, or in any cancer in my view, with an individualized neoantigen vaccine approach,” trial investigator Jeffrey S. Weber, MD, PhD, of NYU Langone Perlmutter Cancer Center in New York, said during an oral abstract session at the annual meeting of the American Association for Cancer Research.
“I have every confidence that this strategy will be expanded to other histologies that are PD-1 sensitive, such as non–small cell lung cancer, renal cell cancer, hepatocellular cancer, gastroesophageal cancer, et cetera,” Dr. Weber said.
Invited discussant Margaret Callahan, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, called the results “exciting,” especially in light of previous results in cancer vaccine trials. “Despite hundreds of formulations and dozens of studies, cancer vaccines have been disappointing so far, and have largely failed to have a meaningful impact in oncology,” she said.
A promising personalized vaccine
The mRNA vaccine is individually tailored and encodes up to 34 patient-specific tumor neoantigens. The vaccine also acts as an adjuvant to strengthen the immune response.
Dr. Weber said that the “mRNA 4157 is what one would call an individualized neoantigen therapy. It will target an individual patient’s unique tumor mutations, and the revelation over the last 5-10 years, is that, for better or worse, virtually all the neoantigens are unique to an individual patient. There are very, very few true universal neoantigens, or at least universal neoantigens that could have clinical utility.”
The vaccines are developed from tumor biopsy tissues that then undergo whole exome and RNA sequencing to identify single nucleotide variants that are present in the tumor but not in normal tissue.
The findings are then fed into a computer algorithm that identifies potential neoepitope peptides that would bind well to the patient’s human leukocyte antigen (HLA) type and could evoke strong T-cell responses.
“Once they’re chosen, you concatenate the sequences together into a single-strand mRNA vaccine, it’s packaged with nanoparticles to encapsulate it, and there you have your mRNA vaccine,” Dr. Weber explained.
In the KEYNOTE-942 trial, the investigators randomly assigned patients with completely resected high-risk cutaneous melanoma on a 2:1 basis to receive mRNA-4157 via intramuscular injection every 3 weeks for a total of nine doses, plus intravenous pembrolizumab every 3 weeks for 18 cycles (107 patients) or pembrolizumab alone (50 patients). Median follow-up was 101 weeks in the combination group and 105 weeks in the pembrolizumab group.
Overall, the 18-month recurrence-free survival rates were 78.6% in the combination arm and 62.2% in the pembrolizumab arm. The recurrence-free survival rates corresponded to a 44% reduced risk of recurrence or death in patients who received the personalized vaccine plus pembrolizumab compared with those who received only pembrolizumab (hazard ratio [HR] for recurrence, 0.561; P =.0266).
Grade 3 or greater adverse events occurred in 25% of patients in the combination group and 18% of patients in the pembrolizumab group. The most common grade 3 event associated with the vaccine was fatigue. No grade 4 adverse events or deaths were associated with the vaccine, and the addition of the vaccine to pembrolizumab did not appear to increase risk for immune-mediated adverse events.
In a subanalysis, Dr. Weber and colleagues explored the relationship between tumor mutational burden and recurrence-free survival. Higher tumor mutational burden may mean more neoepitopes to target, which is helpful when developing personalized neoantigen vaccines, explained coinvestigator Ryan Sullivan, MD, associate director of the melanoma program at Mass General Cancer Center, Boston, who presented the subanalysis results.
The investigators performed whole exome and whole transcriptome sequencing of baseline tumor biopsy samples to determine the mutational burden of tumors and defined a high mutational burden as 10 or more mutations per megabase.
Overall, in the combination group, patients with a higher tumor mutational burden at baseline showed improved outcomes (HR, 0.652; 95% confidence interval [CI], 0.284-1.494), as did patients with a lower tumor mutational burden (HR, 0.586; 95% CI, 0.243-1.415).
The authors found the same was true for patients with high vs. low tumor inflammation scores (high: HR, 0.576; 95% CI, 0.209-1.591 vs. low: HR, 0.528; 95% CI, 0.253-1.101) and higher PD-L1 expression (PD-L1 positive: HR, 0.485; 95% CI, 0.226-1.039 vs. PD-L1 negative: HR, 0.162; 95% CI, 0.038-0.685).
The hazard ratios crossed 1, which suggest that the combination was similarly effective in all patient subsets, said Dr. Sullivan.
Dr. Callahan also highlighted that the P value was based on a one-side log-rank test, “a relatively low bar to jump over” and that there were slight imbalances in both PD-1 expression status and tumor mutational burden – both of which favored the vaccine group and may be associated with better recurrence-free survival.
The 16% difference in recurrence-free survival seen with the combination vs. pembrolizumab alone, if confirmed in further studies, “is clinically meaningful for high-risk patients,” said Dr. Callahan. “The authors are to be congratulated for presenting the first randomized study of a neoantigen vaccine with a clinical efficacy primary endpoint, and this is a trial that incorporates many of the lessons we’ve learned along the years.”
Dr. Sullivan also commented on the promising results. “The field of cancer vaccines is a wasteland of failed clinical trials after some initial promising data, so to have something like this where it does appear that this vaccine strategy works is good not only for patients with melanoma but for those people who have dedicated their lives to trying to develop cancer vaccines,” he said in an interview.
KEYNOTE-942 was funded by Moderna with collaboration from Merck. Dr. Weber has financial relationships with Merck, Moderna, and other companies. Dr. Sullivan has served as a paid consultant for Merck and has received research funding from the company. Dr. Callahan disclosed a consulting/advisory role with Moderna, Merck, and others.
A version of this article first appeared on Medscape.com.
according to the latest data from the KEYNOTE-942 trial.
This recurrence-free survival benefit corresponded to a 44% reduced risk of recurrence or death in patients who received the personalized vaccine plus pembrolizumab compared with the immunotherapy alone.
The randomized phase 2b trial is the first to show a positive result for a cancer vaccine in a randomized trial. The results, if confirmed in further studies, hold promise for treating other solid tumors with sensitivity to the programmed death-1 (PD-1) protein, investigators said.
“KEYNOTE-942 is the first randomized study to demonstrate improvement in recurrence-free survival in melanoma, or in any cancer in my view, with an individualized neoantigen vaccine approach,” trial investigator Jeffrey S. Weber, MD, PhD, of NYU Langone Perlmutter Cancer Center in New York, said during an oral abstract session at the annual meeting of the American Association for Cancer Research.
“I have every confidence that this strategy will be expanded to other histologies that are PD-1 sensitive, such as non–small cell lung cancer, renal cell cancer, hepatocellular cancer, gastroesophageal cancer, et cetera,” Dr. Weber said.
Invited discussant Margaret Callahan, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, called the results “exciting,” especially in light of previous results in cancer vaccine trials. “Despite hundreds of formulations and dozens of studies, cancer vaccines have been disappointing so far, and have largely failed to have a meaningful impact in oncology,” she said.
A promising personalized vaccine
The mRNA vaccine is individually tailored and encodes up to 34 patient-specific tumor neoantigens. The vaccine also acts as an adjuvant to strengthen the immune response.
Dr. Weber said that the “mRNA 4157 is what one would call an individualized neoantigen therapy. It will target an individual patient’s unique tumor mutations, and the revelation over the last 5-10 years, is that, for better or worse, virtually all the neoantigens are unique to an individual patient. There are very, very few true universal neoantigens, or at least universal neoantigens that could have clinical utility.”
The vaccines are developed from tumor biopsy tissues that then undergo whole exome and RNA sequencing to identify single nucleotide variants that are present in the tumor but not in normal tissue.
The findings are then fed into a computer algorithm that identifies potential neoepitope peptides that would bind well to the patient’s human leukocyte antigen (HLA) type and could evoke strong T-cell responses.
“Once they’re chosen, you concatenate the sequences together into a single-strand mRNA vaccine, it’s packaged with nanoparticles to encapsulate it, and there you have your mRNA vaccine,” Dr. Weber explained.
In the KEYNOTE-942 trial, the investigators randomly assigned patients with completely resected high-risk cutaneous melanoma on a 2:1 basis to receive mRNA-4157 via intramuscular injection every 3 weeks for a total of nine doses, plus intravenous pembrolizumab every 3 weeks for 18 cycles (107 patients) or pembrolizumab alone (50 patients). Median follow-up was 101 weeks in the combination group and 105 weeks in the pembrolizumab group.
Overall, the 18-month recurrence-free survival rates were 78.6% in the combination arm and 62.2% in the pembrolizumab arm. The recurrence-free survival rates corresponded to a 44% reduced risk of recurrence or death in patients who received the personalized vaccine plus pembrolizumab compared with those who received only pembrolizumab (hazard ratio [HR] for recurrence, 0.561; P =.0266).
Grade 3 or greater adverse events occurred in 25% of patients in the combination group and 18% of patients in the pembrolizumab group. The most common grade 3 event associated with the vaccine was fatigue. No grade 4 adverse events or deaths were associated with the vaccine, and the addition of the vaccine to pembrolizumab did not appear to increase risk for immune-mediated adverse events.
In a subanalysis, Dr. Weber and colleagues explored the relationship between tumor mutational burden and recurrence-free survival. Higher tumor mutational burden may mean more neoepitopes to target, which is helpful when developing personalized neoantigen vaccines, explained coinvestigator Ryan Sullivan, MD, associate director of the melanoma program at Mass General Cancer Center, Boston, who presented the subanalysis results.
The investigators performed whole exome and whole transcriptome sequencing of baseline tumor biopsy samples to determine the mutational burden of tumors and defined a high mutational burden as 10 or more mutations per megabase.
Overall, in the combination group, patients with a higher tumor mutational burden at baseline showed improved outcomes (HR, 0.652; 95% confidence interval [CI], 0.284-1.494), as did patients with a lower tumor mutational burden (HR, 0.586; 95% CI, 0.243-1.415).
The authors found the same was true for patients with high vs. low tumor inflammation scores (high: HR, 0.576; 95% CI, 0.209-1.591 vs. low: HR, 0.528; 95% CI, 0.253-1.101) and higher PD-L1 expression (PD-L1 positive: HR, 0.485; 95% CI, 0.226-1.039 vs. PD-L1 negative: HR, 0.162; 95% CI, 0.038-0.685).
The hazard ratios crossed 1, which suggest that the combination was similarly effective in all patient subsets, said Dr. Sullivan.
Dr. Callahan also highlighted that the P value was based on a one-side log-rank test, “a relatively low bar to jump over” and that there were slight imbalances in both PD-1 expression status and tumor mutational burden – both of which favored the vaccine group and may be associated with better recurrence-free survival.
The 16% difference in recurrence-free survival seen with the combination vs. pembrolizumab alone, if confirmed in further studies, “is clinically meaningful for high-risk patients,” said Dr. Callahan. “The authors are to be congratulated for presenting the first randomized study of a neoantigen vaccine with a clinical efficacy primary endpoint, and this is a trial that incorporates many of the lessons we’ve learned along the years.”
Dr. Sullivan also commented on the promising results. “The field of cancer vaccines is a wasteland of failed clinical trials after some initial promising data, so to have something like this where it does appear that this vaccine strategy works is good not only for patients with melanoma but for those people who have dedicated their lives to trying to develop cancer vaccines,” he said in an interview.
KEYNOTE-942 was funded by Moderna with collaboration from Merck. Dr. Weber has financial relationships with Merck, Moderna, and other companies. Dr. Sullivan has served as a paid consultant for Merck and has received research funding from the company. Dr. Callahan disclosed a consulting/advisory role with Moderna, Merck, and others.
A version of this article first appeared on Medscape.com.
according to the latest data from the KEYNOTE-942 trial.
This recurrence-free survival benefit corresponded to a 44% reduced risk of recurrence or death in patients who received the personalized vaccine plus pembrolizumab compared with the immunotherapy alone.
The randomized phase 2b trial is the first to show a positive result for a cancer vaccine in a randomized trial. The results, if confirmed in further studies, hold promise for treating other solid tumors with sensitivity to the programmed death-1 (PD-1) protein, investigators said.
“KEYNOTE-942 is the first randomized study to demonstrate improvement in recurrence-free survival in melanoma, or in any cancer in my view, with an individualized neoantigen vaccine approach,” trial investigator Jeffrey S. Weber, MD, PhD, of NYU Langone Perlmutter Cancer Center in New York, said during an oral abstract session at the annual meeting of the American Association for Cancer Research.
“I have every confidence that this strategy will be expanded to other histologies that are PD-1 sensitive, such as non–small cell lung cancer, renal cell cancer, hepatocellular cancer, gastroesophageal cancer, et cetera,” Dr. Weber said.
Invited discussant Margaret Callahan, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, called the results “exciting,” especially in light of previous results in cancer vaccine trials. “Despite hundreds of formulations and dozens of studies, cancer vaccines have been disappointing so far, and have largely failed to have a meaningful impact in oncology,” she said.
A promising personalized vaccine
The mRNA vaccine is individually tailored and encodes up to 34 patient-specific tumor neoantigens. The vaccine also acts as an adjuvant to strengthen the immune response.
Dr. Weber said that the “mRNA 4157 is what one would call an individualized neoantigen therapy. It will target an individual patient’s unique tumor mutations, and the revelation over the last 5-10 years, is that, for better or worse, virtually all the neoantigens are unique to an individual patient. There are very, very few true universal neoantigens, or at least universal neoantigens that could have clinical utility.”
The vaccines are developed from tumor biopsy tissues that then undergo whole exome and RNA sequencing to identify single nucleotide variants that are present in the tumor but not in normal tissue.
The findings are then fed into a computer algorithm that identifies potential neoepitope peptides that would bind well to the patient’s human leukocyte antigen (HLA) type and could evoke strong T-cell responses.
“Once they’re chosen, you concatenate the sequences together into a single-strand mRNA vaccine, it’s packaged with nanoparticles to encapsulate it, and there you have your mRNA vaccine,” Dr. Weber explained.
In the KEYNOTE-942 trial, the investigators randomly assigned patients with completely resected high-risk cutaneous melanoma on a 2:1 basis to receive mRNA-4157 via intramuscular injection every 3 weeks for a total of nine doses, plus intravenous pembrolizumab every 3 weeks for 18 cycles (107 patients) or pembrolizumab alone (50 patients). Median follow-up was 101 weeks in the combination group and 105 weeks in the pembrolizumab group.
Overall, the 18-month recurrence-free survival rates were 78.6% in the combination arm and 62.2% in the pembrolizumab arm. The recurrence-free survival rates corresponded to a 44% reduced risk of recurrence or death in patients who received the personalized vaccine plus pembrolizumab compared with those who received only pembrolizumab (hazard ratio [HR] for recurrence, 0.561; P =.0266).
Grade 3 or greater adverse events occurred in 25% of patients in the combination group and 18% of patients in the pembrolizumab group. The most common grade 3 event associated with the vaccine was fatigue. No grade 4 adverse events or deaths were associated with the vaccine, and the addition of the vaccine to pembrolizumab did not appear to increase risk for immune-mediated adverse events.
In a subanalysis, Dr. Weber and colleagues explored the relationship between tumor mutational burden and recurrence-free survival. Higher tumor mutational burden may mean more neoepitopes to target, which is helpful when developing personalized neoantigen vaccines, explained coinvestigator Ryan Sullivan, MD, associate director of the melanoma program at Mass General Cancer Center, Boston, who presented the subanalysis results.
The investigators performed whole exome and whole transcriptome sequencing of baseline tumor biopsy samples to determine the mutational burden of tumors and defined a high mutational burden as 10 or more mutations per megabase.
Overall, in the combination group, patients with a higher tumor mutational burden at baseline showed improved outcomes (HR, 0.652; 95% confidence interval [CI], 0.284-1.494), as did patients with a lower tumor mutational burden (HR, 0.586; 95% CI, 0.243-1.415).
The authors found the same was true for patients with high vs. low tumor inflammation scores (high: HR, 0.576; 95% CI, 0.209-1.591 vs. low: HR, 0.528; 95% CI, 0.253-1.101) and higher PD-L1 expression (PD-L1 positive: HR, 0.485; 95% CI, 0.226-1.039 vs. PD-L1 negative: HR, 0.162; 95% CI, 0.038-0.685).
The hazard ratios crossed 1, which suggest that the combination was similarly effective in all patient subsets, said Dr. Sullivan.
Dr. Callahan also highlighted that the P value was based on a one-side log-rank test, “a relatively low bar to jump over” and that there were slight imbalances in both PD-1 expression status and tumor mutational burden – both of which favored the vaccine group and may be associated with better recurrence-free survival.
The 16% difference in recurrence-free survival seen with the combination vs. pembrolizumab alone, if confirmed in further studies, “is clinically meaningful for high-risk patients,” said Dr. Callahan. “The authors are to be congratulated for presenting the first randomized study of a neoantigen vaccine with a clinical efficacy primary endpoint, and this is a trial that incorporates many of the lessons we’ve learned along the years.”
Dr. Sullivan also commented on the promising results. “The field of cancer vaccines is a wasteland of failed clinical trials after some initial promising data, so to have something like this where it does appear that this vaccine strategy works is good not only for patients with melanoma but for those people who have dedicated their lives to trying to develop cancer vaccines,” he said in an interview.
KEYNOTE-942 was funded by Moderna with collaboration from Merck. Dr. Weber has financial relationships with Merck, Moderna, and other companies. Dr. Sullivan has served as a paid consultant for Merck and has received research funding from the company. Dr. Callahan disclosed a consulting/advisory role with Moderna, Merck, and others.
A version of this article first appeared on Medscape.com.
FROM AACR 2023