User login
Workforce Shortage of Pediatric Dermatologists: A Medical Student’s Perspective
Workforce Shortage of Pediatric Dermatologists: A Medical Student’s Perspective
There is a shortage of pediatric dermatologists in the United States, with fewer than 2% of practicing dermatologists specializing in pediatrics.1 Pediatric dermatology has the third highest referral rate by pediatricians but also is the third most challenging specialty to access, with an average appointment wait time of 92 days.2,3 Another factor leading to increased appointment wait times is the specificity of care required for pediatric patients. Frequently, pediatric patients evaluated by a general dermatologist will be referred to their pediatric dermatology colleagues. As medical students, we were introduced to the field of pediatric dermatology through different avenues—personal experience, research mentorship, or a clinical rotation in medical school. We found ourselves curious about the discrepancy between the supply of and demand for pediatric dermatologists and wondered what could be done to increase awareness of this subspecialty among medical students. We believe this workforce shortage can be ameliorated by improving early exposure to pediatric dermatology. In this article, we explore the existing framework surrounding pediatric dermatology in medical education and offer feasible recommendations and solutions to realistically combat this problem.
Pediatric dermatologists are essential to the greater dermatology community. Pediatric dermatologists receive advanced training in complex pediatric skin conditions that often is lacking in general dermatology residency. A large percentage of pediatric dermatology patients seen in academic medical centers have already been seen by general dermatologists who subsequently referred them to specialty care. In one study, 9.6% (10/108) of practicing pediatric dermatologists noted that their referrals were from general dermatologists.4 In another study, 42% (19/45) of referrals to a multidisciplinary pediatric dermatology-genetics were from general dermatologists.5 Given the shortage of pediatric dermatologists, these referrals undoubtedly overwhelm the system, and the results of these studies underscore the reality that general dermatologists do not necessarily feel adequately trained in complex pediatric conditions, creating an intrinsic need for pediatric dermatologists.
Admani et al6 reported that early mentorship was the single most important factor to 84% (91/109) of survey respondents who pursued pediatric dermatology. Forty percent (40/100) of survey respondents chose their specialty of pediatric dermatology during pediatrics residency, 34% (34/100) during medical school, 17% (17/100) during dermatology residency, and 5% (5/100) during internship, indicating that medical school is a crucial time for recruitment.6 It has been noted in the literature that more medical students matched to dermatology residency from schools with dermatology clerkships built into the curriculum than from schools without dedicated dermatology rotations, suggesting that early clinical exposure to dermatology fields has a predictable influence in matching.7 Currently, only about 10% (15/155) of allopathic medical schools in the United States offer a formal elective in pediatric dermatology via the Association of American Medical College’s Visiting Student Learning Opportunities program.8 When this information was cross-referenced with the most recently matched pediatric dermatology fellowship class (2023-2024), provided by the Fellowship Directors Chair of the Society for Pediatric Dermatology, we found that 17% (4/24) of the matched fellows attended one of these 15 medical schools. We also found that the 2023-2024 pediatric dermatology fellowship class had 12 unmatched spots out of 36 total positions nationwide (33%), highlighting a gap in pediatric dermatology care and placing further strain on an already underserved subspecialty. These data suggest that, while dermatologists may decide to pursue pediatric dermatology fellowships during residency, there is an opportunity to foster interest during medical school training and improve the fellowship match rate.
Several medical schools in the United States incorporate pediatric dermatology into their curricula, including lectures in preclinical courses and career panels to pediatric dermatology electives in the third and fourth years. These institutions can serve as models for other medical schools. Within preclinical content, we recommend creating a designated dermatology unit that can incorporate common pediatric dermatology pathologies also seen by general practitioners, such as common childhood rashes, atopic dermatitis, alopecia areata, seborrheic dermatitis, and acne. Rare pediatric diseases such as epidermolysis bullosa, tuberous sclerosis, and Ehlers-Danlos syndrome also may be included in the unit. If schools are not able to offer a stand-alone dermatology preclinical course, this content can be added to the immunology, musculoskeletal, infectious diseases, or genetics courses to account for the multisystemic effects of some of these conditions. Ideally, schools would offer elective exposure to pediatric dermatology during the clinical years of medical school to increase knowledge of the field; for example, pediatric dermatology materials could be included in core clerkships, as much of this content is applicable to the general pediatrics rotation. In particular, a lecture on common rashes in pediatric patients could be given before starting the core pediatric rotation. Additionally, problem-based pediatric dermatology cases could be implemented during the core pediatrics rotation. If students are offered an independent dermatology clinical elective, the already formatted 2- and 4-week basic dermatology courses designed by the American Academy of Dermatology could serve as suggested teaching guides or as self-teaching resources that could complement the dermatology rotation.9,10 Pediatric topics (eg, pediatric cutaneous fungal infections) are included within the American Academy of Dermatology basic dermatology curriculum.8,9
Increasing access to pediatric dermatology resources such as lecture series and mentorship opportunities could further broaden the pediatric dermatology knowledge base of medical students. Within medical school dermatology interest groups, there is an opportunity to have a pediatric dermatology lead to help coordinate lecture series and journal club sessions for interested students. The Society for Pediatric Dermatology and the Pediatric Dermatology Research Alliance have created programs to support students, and we encourage schools to raise awareness of these organizations as well as conference and grant opportunities. These initiatives foster meaningful mentor-mentee relationships, and more medical students may be interested if they are aware of these support networks.
There also may be opportunities to create residency tracks that increase the number of dermatology residency applicants. Programs such as the newly implemented pediatric dermatology track at the University of Pennsylvania and New York University allow medical students who are interested in pursuing pediatric dermatology to have a more focused and linear training path.11,12 Due to the inherent competition in matching into dermatology, we surmise that many students with interest in pediatric dermatology are lost to pediatric residencies. Given the large percentage of pediatric residents who ultimately develop an interest in pediatric dermatology, holding a spot for pediatric dermatology applicants—akin to the combined medical-dermatology spots—may be an avenue to increase the pool of pediatric dermatology fellows.1,6 Another avenue is to encourage the development of first-year pediatric internship tracks that lead directly into dermatology residency, such as newly established programs at the University of Pennsylvania and New York University.11,12
As a group of both aspiring and practicing pediatric dermatologists, we have identified opportunities for formalized education in and early exposure to this subspecialty during medical training instead of leaving the discovery of the field to chance. The gaps in medical education that we have identified have already led us to present potential curricular changes to the medical education committee at our home institution. We hope to inspire the development of strong pediatric dermatology education at the medical school level.
While the solution to the pediatric dermatology workforce shortage is complex and multifaceted, there is a unique opportunity to target medical students through mentorship, access to education, and clinical experiences. We recommend that medical schools implement these educational methods and track the efficacy of these interventions to quantify the predicted association between an increased workforce and early exposure to pediatric dermatology. Addressing a lack of exposure to the field and increasing support of students pursuing pediatric dermatology can help to alleviate the shortage at the earliest point in training.
- Prindaville B, Antaya RJ, Siegfried EC. Pediatric dermatology: past, present, and future. Pediatr Dermatol. 2015;32:1-12. doi:10.1111/pde.12362
- Wright TS. Update on the pediatric dermatology workforce shortage. Cutis. 2021;108:237-238. doi:10.12788/cutis.0379
- Stephens MR, Murthy AS, McMahon PJ. Wait times, health care touchpoints, and nonattendance in an academic pediatric dermatology clinic. ediatr Dermatol. 2019;36:893-897. doi:10.1111/pde.13943
- Fogel AL, Teng JM. A survey to assess perceived differences in referral pathways to board-certified pediatric dermatologists. Pediatr Dermatol. 2015;32:e314-e315. doi:10.1111/pde.12703
- Parker JC, Rangu S, Grand KL, et al. Genetic skin disorders: the value of a multidisciplinary clinic. Am J Med Genet A. 2021;185:1159-1167. doi:10.1002/ajmg.a.62095
- Admani S, Caufield M, Kim SS, et al. Understanding the pediatric dermatology workforce shortage: mentoring matters. J Pediatr. 2014;164:372-5.e1. doi:10.1016/j.jpeds.2013.10.004
- Ogidi P, Ahmed F, Cahn BA, et al. Medical schools as gatekeepers: a survey and analysis of factors predicting dermatology residency placement. J Am Acad Dermatol. 2022;86:490-492. doi:10.1016 /j.jaad.2021.09.027
- Visiting Student Learning Opportunities (VSLO). Accessed May 30, 2025. https://students-residents.aamc.org/visiting-student-learning-opportunities/visiting-student-learning-opportunities-vslo
- American Academy of Dermatology Association. AAD Learning Center. Basic dermatology curriculum (2-week rotation). Accessed May 12, 2025. https://learning.aad.org/Listing/Basic-Dermatology-Curriculum-2-Week-Rotation-5395
- American Academy of Dermatology Association. AAD Learning Center. Basic dermatology curriculum (4-week rotation). Accessed May 12, 2025. https://learning.aad.org/Public/Catalog/Details.aspx?id=YPssTVIbBO3Zb%2bOuf%2fM7Kg%3d%3d&returnurl=%2fUsers%2fUserOnlineCourse.aspx%3fLearningActivityID%3dYPssTVIbBO3Zb%252bOuf%252fM7Kg%253d%253d
- Penn Medicine Dermatology Residency Training Program. Residency tracks. Accessed May 12, 2025. https://dermatology.upenn.edu/residents/residency-tracks/
- Pediatric Dermatology Residency Track at NYU Grossman School of Medicine. Pediatric Track. Accessed May 30, 2025. https://med.nyu.edu/departments-institutes/dermatology/education/residency/pediatric-track
There is a shortage of pediatric dermatologists in the United States, with fewer than 2% of practicing dermatologists specializing in pediatrics.1 Pediatric dermatology has the third highest referral rate by pediatricians but also is the third most challenging specialty to access, with an average appointment wait time of 92 days.2,3 Another factor leading to increased appointment wait times is the specificity of care required for pediatric patients. Frequently, pediatric patients evaluated by a general dermatologist will be referred to their pediatric dermatology colleagues. As medical students, we were introduced to the field of pediatric dermatology through different avenues—personal experience, research mentorship, or a clinical rotation in medical school. We found ourselves curious about the discrepancy between the supply of and demand for pediatric dermatologists and wondered what could be done to increase awareness of this subspecialty among medical students. We believe this workforce shortage can be ameliorated by improving early exposure to pediatric dermatology. In this article, we explore the existing framework surrounding pediatric dermatology in medical education and offer feasible recommendations and solutions to realistically combat this problem.
Pediatric dermatologists are essential to the greater dermatology community. Pediatric dermatologists receive advanced training in complex pediatric skin conditions that often is lacking in general dermatology residency. A large percentage of pediatric dermatology patients seen in academic medical centers have already been seen by general dermatologists who subsequently referred them to specialty care. In one study, 9.6% (10/108) of practicing pediatric dermatologists noted that their referrals were from general dermatologists.4 In another study, 42% (19/45) of referrals to a multidisciplinary pediatric dermatology-genetics were from general dermatologists.5 Given the shortage of pediatric dermatologists, these referrals undoubtedly overwhelm the system, and the results of these studies underscore the reality that general dermatologists do not necessarily feel adequately trained in complex pediatric conditions, creating an intrinsic need for pediatric dermatologists.
Admani et al6 reported that early mentorship was the single most important factor to 84% (91/109) of survey respondents who pursued pediatric dermatology. Forty percent (40/100) of survey respondents chose their specialty of pediatric dermatology during pediatrics residency, 34% (34/100) during medical school, 17% (17/100) during dermatology residency, and 5% (5/100) during internship, indicating that medical school is a crucial time for recruitment.6 It has been noted in the literature that more medical students matched to dermatology residency from schools with dermatology clerkships built into the curriculum than from schools without dedicated dermatology rotations, suggesting that early clinical exposure to dermatology fields has a predictable influence in matching.7 Currently, only about 10% (15/155) of allopathic medical schools in the United States offer a formal elective in pediatric dermatology via the Association of American Medical College’s Visiting Student Learning Opportunities program.8 When this information was cross-referenced with the most recently matched pediatric dermatology fellowship class (2023-2024), provided by the Fellowship Directors Chair of the Society for Pediatric Dermatology, we found that 17% (4/24) of the matched fellows attended one of these 15 medical schools. We also found that the 2023-2024 pediatric dermatology fellowship class had 12 unmatched spots out of 36 total positions nationwide (33%), highlighting a gap in pediatric dermatology care and placing further strain on an already underserved subspecialty. These data suggest that, while dermatologists may decide to pursue pediatric dermatology fellowships during residency, there is an opportunity to foster interest during medical school training and improve the fellowship match rate.
Several medical schools in the United States incorporate pediatric dermatology into their curricula, including lectures in preclinical courses and career panels to pediatric dermatology electives in the third and fourth years. These institutions can serve as models for other medical schools. Within preclinical content, we recommend creating a designated dermatology unit that can incorporate common pediatric dermatology pathologies also seen by general practitioners, such as common childhood rashes, atopic dermatitis, alopecia areata, seborrheic dermatitis, and acne. Rare pediatric diseases such as epidermolysis bullosa, tuberous sclerosis, and Ehlers-Danlos syndrome also may be included in the unit. If schools are not able to offer a stand-alone dermatology preclinical course, this content can be added to the immunology, musculoskeletal, infectious diseases, or genetics courses to account for the multisystemic effects of some of these conditions. Ideally, schools would offer elective exposure to pediatric dermatology during the clinical years of medical school to increase knowledge of the field; for example, pediatric dermatology materials could be included in core clerkships, as much of this content is applicable to the general pediatrics rotation. In particular, a lecture on common rashes in pediatric patients could be given before starting the core pediatric rotation. Additionally, problem-based pediatric dermatology cases could be implemented during the core pediatrics rotation. If students are offered an independent dermatology clinical elective, the already formatted 2- and 4-week basic dermatology courses designed by the American Academy of Dermatology could serve as suggested teaching guides or as self-teaching resources that could complement the dermatology rotation.9,10 Pediatric topics (eg, pediatric cutaneous fungal infections) are included within the American Academy of Dermatology basic dermatology curriculum.8,9
Increasing access to pediatric dermatology resources such as lecture series and mentorship opportunities could further broaden the pediatric dermatology knowledge base of medical students. Within medical school dermatology interest groups, there is an opportunity to have a pediatric dermatology lead to help coordinate lecture series and journal club sessions for interested students. The Society for Pediatric Dermatology and the Pediatric Dermatology Research Alliance have created programs to support students, and we encourage schools to raise awareness of these organizations as well as conference and grant opportunities. These initiatives foster meaningful mentor-mentee relationships, and more medical students may be interested if they are aware of these support networks.
There also may be opportunities to create residency tracks that increase the number of dermatology residency applicants. Programs such as the newly implemented pediatric dermatology track at the University of Pennsylvania and New York University allow medical students who are interested in pursuing pediatric dermatology to have a more focused and linear training path.11,12 Due to the inherent competition in matching into dermatology, we surmise that many students with interest in pediatric dermatology are lost to pediatric residencies. Given the large percentage of pediatric residents who ultimately develop an interest in pediatric dermatology, holding a spot for pediatric dermatology applicants—akin to the combined medical-dermatology spots—may be an avenue to increase the pool of pediatric dermatology fellows.1,6 Another avenue is to encourage the development of first-year pediatric internship tracks that lead directly into dermatology residency, such as newly established programs at the University of Pennsylvania and New York University.11,12
As a group of both aspiring and practicing pediatric dermatologists, we have identified opportunities for formalized education in and early exposure to this subspecialty during medical training instead of leaving the discovery of the field to chance. The gaps in medical education that we have identified have already led us to present potential curricular changes to the medical education committee at our home institution. We hope to inspire the development of strong pediatric dermatology education at the medical school level.
While the solution to the pediatric dermatology workforce shortage is complex and multifaceted, there is a unique opportunity to target medical students through mentorship, access to education, and clinical experiences. We recommend that medical schools implement these educational methods and track the efficacy of these interventions to quantify the predicted association between an increased workforce and early exposure to pediatric dermatology. Addressing a lack of exposure to the field and increasing support of students pursuing pediatric dermatology can help to alleviate the shortage at the earliest point in training.
There is a shortage of pediatric dermatologists in the United States, with fewer than 2% of practicing dermatologists specializing in pediatrics.1 Pediatric dermatology has the third highest referral rate by pediatricians but also is the third most challenging specialty to access, with an average appointment wait time of 92 days.2,3 Another factor leading to increased appointment wait times is the specificity of care required for pediatric patients. Frequently, pediatric patients evaluated by a general dermatologist will be referred to their pediatric dermatology colleagues. As medical students, we were introduced to the field of pediatric dermatology through different avenues—personal experience, research mentorship, or a clinical rotation in medical school. We found ourselves curious about the discrepancy between the supply of and demand for pediatric dermatologists and wondered what could be done to increase awareness of this subspecialty among medical students. We believe this workforce shortage can be ameliorated by improving early exposure to pediatric dermatology. In this article, we explore the existing framework surrounding pediatric dermatology in medical education and offer feasible recommendations and solutions to realistically combat this problem.
Pediatric dermatologists are essential to the greater dermatology community. Pediatric dermatologists receive advanced training in complex pediatric skin conditions that often is lacking in general dermatology residency. A large percentage of pediatric dermatology patients seen in academic medical centers have already been seen by general dermatologists who subsequently referred them to specialty care. In one study, 9.6% (10/108) of practicing pediatric dermatologists noted that their referrals were from general dermatologists.4 In another study, 42% (19/45) of referrals to a multidisciplinary pediatric dermatology-genetics were from general dermatologists.5 Given the shortage of pediatric dermatologists, these referrals undoubtedly overwhelm the system, and the results of these studies underscore the reality that general dermatologists do not necessarily feel adequately trained in complex pediatric conditions, creating an intrinsic need for pediatric dermatologists.
Admani et al6 reported that early mentorship was the single most important factor to 84% (91/109) of survey respondents who pursued pediatric dermatology. Forty percent (40/100) of survey respondents chose their specialty of pediatric dermatology during pediatrics residency, 34% (34/100) during medical school, 17% (17/100) during dermatology residency, and 5% (5/100) during internship, indicating that medical school is a crucial time for recruitment.6 It has been noted in the literature that more medical students matched to dermatology residency from schools with dermatology clerkships built into the curriculum than from schools without dedicated dermatology rotations, suggesting that early clinical exposure to dermatology fields has a predictable influence in matching.7 Currently, only about 10% (15/155) of allopathic medical schools in the United States offer a formal elective in pediatric dermatology via the Association of American Medical College’s Visiting Student Learning Opportunities program.8 When this information was cross-referenced with the most recently matched pediatric dermatology fellowship class (2023-2024), provided by the Fellowship Directors Chair of the Society for Pediatric Dermatology, we found that 17% (4/24) of the matched fellows attended one of these 15 medical schools. We also found that the 2023-2024 pediatric dermatology fellowship class had 12 unmatched spots out of 36 total positions nationwide (33%), highlighting a gap in pediatric dermatology care and placing further strain on an already underserved subspecialty. These data suggest that, while dermatologists may decide to pursue pediatric dermatology fellowships during residency, there is an opportunity to foster interest during medical school training and improve the fellowship match rate.
Several medical schools in the United States incorporate pediatric dermatology into their curricula, including lectures in preclinical courses and career panels to pediatric dermatology electives in the third and fourth years. These institutions can serve as models for other medical schools. Within preclinical content, we recommend creating a designated dermatology unit that can incorporate common pediatric dermatology pathologies also seen by general practitioners, such as common childhood rashes, atopic dermatitis, alopecia areata, seborrheic dermatitis, and acne. Rare pediatric diseases such as epidermolysis bullosa, tuberous sclerosis, and Ehlers-Danlos syndrome also may be included in the unit. If schools are not able to offer a stand-alone dermatology preclinical course, this content can be added to the immunology, musculoskeletal, infectious diseases, or genetics courses to account for the multisystemic effects of some of these conditions. Ideally, schools would offer elective exposure to pediatric dermatology during the clinical years of medical school to increase knowledge of the field; for example, pediatric dermatology materials could be included in core clerkships, as much of this content is applicable to the general pediatrics rotation. In particular, a lecture on common rashes in pediatric patients could be given before starting the core pediatric rotation. Additionally, problem-based pediatric dermatology cases could be implemented during the core pediatrics rotation. If students are offered an independent dermatology clinical elective, the already formatted 2- and 4-week basic dermatology courses designed by the American Academy of Dermatology could serve as suggested teaching guides or as self-teaching resources that could complement the dermatology rotation.9,10 Pediatric topics (eg, pediatric cutaneous fungal infections) are included within the American Academy of Dermatology basic dermatology curriculum.8,9
Increasing access to pediatric dermatology resources such as lecture series and mentorship opportunities could further broaden the pediatric dermatology knowledge base of medical students. Within medical school dermatology interest groups, there is an opportunity to have a pediatric dermatology lead to help coordinate lecture series and journal club sessions for interested students. The Society for Pediatric Dermatology and the Pediatric Dermatology Research Alliance have created programs to support students, and we encourage schools to raise awareness of these organizations as well as conference and grant opportunities. These initiatives foster meaningful mentor-mentee relationships, and more medical students may be interested if they are aware of these support networks.
There also may be opportunities to create residency tracks that increase the number of dermatology residency applicants. Programs such as the newly implemented pediatric dermatology track at the University of Pennsylvania and New York University allow medical students who are interested in pursuing pediatric dermatology to have a more focused and linear training path.11,12 Due to the inherent competition in matching into dermatology, we surmise that many students with interest in pediatric dermatology are lost to pediatric residencies. Given the large percentage of pediatric residents who ultimately develop an interest in pediatric dermatology, holding a spot for pediatric dermatology applicants—akin to the combined medical-dermatology spots—may be an avenue to increase the pool of pediatric dermatology fellows.1,6 Another avenue is to encourage the development of first-year pediatric internship tracks that lead directly into dermatology residency, such as newly established programs at the University of Pennsylvania and New York University.11,12
As a group of both aspiring and practicing pediatric dermatologists, we have identified opportunities for formalized education in and early exposure to this subspecialty during medical training instead of leaving the discovery of the field to chance. The gaps in medical education that we have identified have already led us to present potential curricular changes to the medical education committee at our home institution. We hope to inspire the development of strong pediatric dermatology education at the medical school level.
While the solution to the pediatric dermatology workforce shortage is complex and multifaceted, there is a unique opportunity to target medical students through mentorship, access to education, and clinical experiences. We recommend that medical schools implement these educational methods and track the efficacy of these interventions to quantify the predicted association between an increased workforce and early exposure to pediatric dermatology. Addressing a lack of exposure to the field and increasing support of students pursuing pediatric dermatology can help to alleviate the shortage at the earliest point in training.
- Prindaville B, Antaya RJ, Siegfried EC. Pediatric dermatology: past, present, and future. Pediatr Dermatol. 2015;32:1-12. doi:10.1111/pde.12362
- Wright TS. Update on the pediatric dermatology workforce shortage. Cutis. 2021;108:237-238. doi:10.12788/cutis.0379
- Stephens MR, Murthy AS, McMahon PJ. Wait times, health care touchpoints, and nonattendance in an academic pediatric dermatology clinic. ediatr Dermatol. 2019;36:893-897. doi:10.1111/pde.13943
- Fogel AL, Teng JM. A survey to assess perceived differences in referral pathways to board-certified pediatric dermatologists. Pediatr Dermatol. 2015;32:e314-e315. doi:10.1111/pde.12703
- Parker JC, Rangu S, Grand KL, et al. Genetic skin disorders: the value of a multidisciplinary clinic. Am J Med Genet A. 2021;185:1159-1167. doi:10.1002/ajmg.a.62095
- Admani S, Caufield M, Kim SS, et al. Understanding the pediatric dermatology workforce shortage: mentoring matters. J Pediatr. 2014;164:372-5.e1. doi:10.1016/j.jpeds.2013.10.004
- Ogidi P, Ahmed F, Cahn BA, et al. Medical schools as gatekeepers: a survey and analysis of factors predicting dermatology residency placement. J Am Acad Dermatol. 2022;86:490-492. doi:10.1016 /j.jaad.2021.09.027
- Visiting Student Learning Opportunities (VSLO). Accessed May 30, 2025. https://students-residents.aamc.org/visiting-student-learning-opportunities/visiting-student-learning-opportunities-vslo
- American Academy of Dermatology Association. AAD Learning Center. Basic dermatology curriculum (2-week rotation). Accessed May 12, 2025. https://learning.aad.org/Listing/Basic-Dermatology-Curriculum-2-Week-Rotation-5395
- American Academy of Dermatology Association. AAD Learning Center. Basic dermatology curriculum (4-week rotation). Accessed May 12, 2025. https://learning.aad.org/Public/Catalog/Details.aspx?id=YPssTVIbBO3Zb%2bOuf%2fM7Kg%3d%3d&returnurl=%2fUsers%2fUserOnlineCourse.aspx%3fLearningActivityID%3dYPssTVIbBO3Zb%252bOuf%252fM7Kg%253d%253d
- Penn Medicine Dermatology Residency Training Program. Residency tracks. Accessed May 12, 2025. https://dermatology.upenn.edu/residents/residency-tracks/
- Pediatric Dermatology Residency Track at NYU Grossman School of Medicine. Pediatric Track. Accessed May 30, 2025. https://med.nyu.edu/departments-institutes/dermatology/education/residency/pediatric-track
- Prindaville B, Antaya RJ, Siegfried EC. Pediatric dermatology: past, present, and future. Pediatr Dermatol. 2015;32:1-12. doi:10.1111/pde.12362
- Wright TS. Update on the pediatric dermatology workforce shortage. Cutis. 2021;108:237-238. doi:10.12788/cutis.0379
- Stephens MR, Murthy AS, McMahon PJ. Wait times, health care touchpoints, and nonattendance in an academic pediatric dermatology clinic. ediatr Dermatol. 2019;36:893-897. doi:10.1111/pde.13943
- Fogel AL, Teng JM. A survey to assess perceived differences in referral pathways to board-certified pediatric dermatologists. Pediatr Dermatol. 2015;32:e314-e315. doi:10.1111/pde.12703
- Parker JC, Rangu S, Grand KL, et al. Genetic skin disorders: the value of a multidisciplinary clinic. Am J Med Genet A. 2021;185:1159-1167. doi:10.1002/ajmg.a.62095
- Admani S, Caufield M, Kim SS, et al. Understanding the pediatric dermatology workforce shortage: mentoring matters. J Pediatr. 2014;164:372-5.e1. doi:10.1016/j.jpeds.2013.10.004
- Ogidi P, Ahmed F, Cahn BA, et al. Medical schools as gatekeepers: a survey and analysis of factors predicting dermatology residency placement. J Am Acad Dermatol. 2022;86:490-492. doi:10.1016 /j.jaad.2021.09.027
- Visiting Student Learning Opportunities (VSLO). Accessed May 30, 2025. https://students-residents.aamc.org/visiting-student-learning-opportunities/visiting-student-learning-opportunities-vslo
- American Academy of Dermatology Association. AAD Learning Center. Basic dermatology curriculum (2-week rotation). Accessed May 12, 2025. https://learning.aad.org/Listing/Basic-Dermatology-Curriculum-2-Week-Rotation-5395
- American Academy of Dermatology Association. AAD Learning Center. Basic dermatology curriculum (4-week rotation). Accessed May 12, 2025. https://learning.aad.org/Public/Catalog/Details.aspx?id=YPssTVIbBO3Zb%2bOuf%2fM7Kg%3d%3d&returnurl=%2fUsers%2fUserOnlineCourse.aspx%3fLearningActivityID%3dYPssTVIbBO3Zb%252bOuf%252fM7Kg%253d%253d
- Penn Medicine Dermatology Residency Training Program. Residency tracks. Accessed May 12, 2025. https://dermatology.upenn.edu/residents/residency-tracks/
- Pediatric Dermatology Residency Track at NYU Grossman School of Medicine. Pediatric Track. Accessed May 30, 2025. https://med.nyu.edu/departments-institutes/dermatology/education/residency/pediatric-track
Workforce Shortage of Pediatric Dermatologists: A Medical Student’s Perspective
Workforce Shortage of Pediatric Dermatologists: A Medical Student’s Perspective
PRACTICE POINTS
- Addressing a lack of exposure to pediatric dermatology in medical school and increasing support for students who are interested in the field can help alleviate the shortage of physicians at the earliest point in training.
- Increasing access to pediatric dermatology resources, such as lecture series and mentorship opportunities, could further broaden the medical student knowledge base.
- There is an opportunity to create residency tracks that increase the number of dermatology residency applicants who are medical students interested in pursuing pediatric dermatology.
Eruptive Erythematous Papules on the Forearms
Eruptive Erythematous Papules on the Forearms
THE DIAGNOSIS: Acral Eruptive Syringoma
Syringomas are small, benign, often asymptomatic eccrine tumors that originate in the intraepidermal portion of eccrine sweat ducts.1 Clinically, they present as multiple symmetric white-to-yellow or discrete flesh-colored papules measuring 1 to 3 mm in diameter, often located on the face (most commonly on the eyelids), with a greater prevalence in middle-aged women. Occasionally, they manifest in other locations such as the cheeks, chest, axillae, abdomen, and groin.2
In 1987, Friedman and Butler3 developed a classification system categorizing syringomas into 4 clinical subtypes: familial syringoma, localized syringoma, Down syndrome–related syringoma, and generalized syringoma. The fourth subtype includes the variant of eruptive syringoma,3 a rare clinical manifestation that often develops before or during puberty with several flesh-colored or lightly pigmented papules on the neck, anterior chest, upper abdomen, axillae, periumbilical region, and/or genital region.1,4,5 The etiology of eruptive syringomas is unclear, although it has been linked to abnormal proliferation of sweat glands due to an underlying local inflammatory process.6
Acral distribution of syringomas is a rare variant that can manifest as part of generalized eruptive syringoma with consequent involvement of the arms and other areas.5,7 There are limited case reports on eruptive syringomas with predominant acral distribution.8 Compared to classic syringomas, the acral variant is associated with an older age of onset as well as a similar prevalence between men and women.9 Acral eruptive syringoma (AES) usually is isolated to the distal arms and legs. The most commonly affected region is the anterior surface of the forearms, although involvement of the dorsal hands, wrists, and feet also has been reported.10-16
The first known case of AES, which was reported in 1977, described eruptive syringomas on the dorsal hands of a healthy 31-year-old man.17 Several cases have been reported since then, mostly in patients aged 30 to 60 years, with predominant involvement of the dorsal hands and forearms.18-24 A review of Embase as well as PubMed articles indexed for MEDLINE using the search terms syringoma OR eccrine ductal tumor and eruptive OR acral OR arms OR forearms OR extremities identified 19 reported cases of AES between 1977 and 2023. For the reported AES cases, the mean (SD) age at diagnosis was 45.1 years (15.96 years), with patient ages ranging from 19 to 76 years. Notably, most cases occurred in individuals aged between 30 and 60 years, which deviates from the typical age of onset of localized syringomas, commonly seen during puberty or early adulthood.
Currently, AES is categorized within the clinical presentation of eruptive syringoma. Nevertheless, some authors have proposed classifying it as a distinct fifth clinical group due to specific features that distinguish it from generalized eruptive syringoma.9 This reclassification has considerable implications for the differential diagnosis, particularly because exclusive acral involvement poses a substantial diagnostic challenge and often requires histologic confirmation.
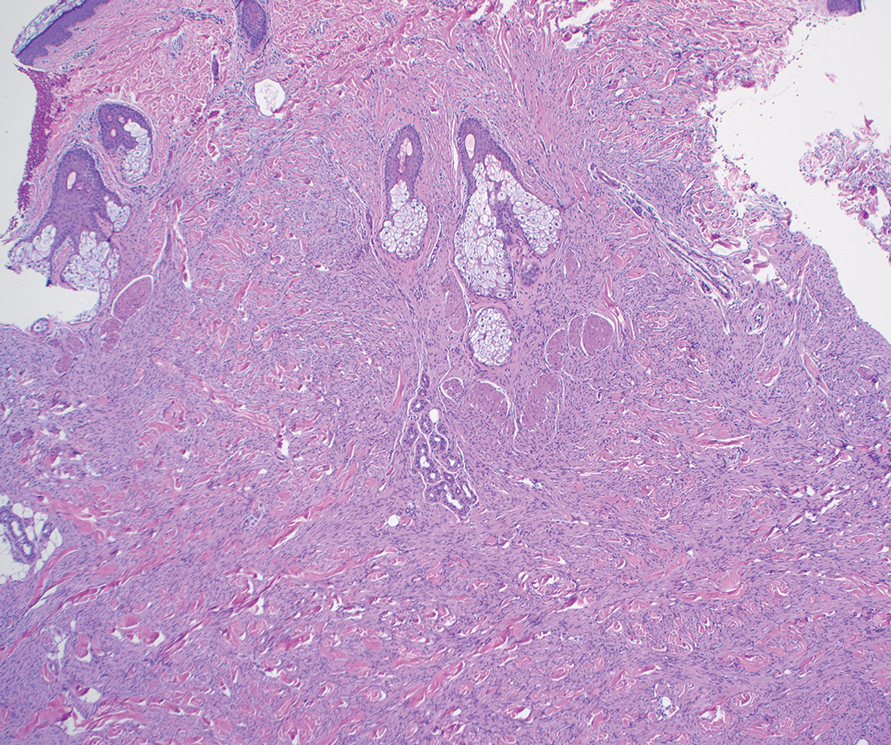
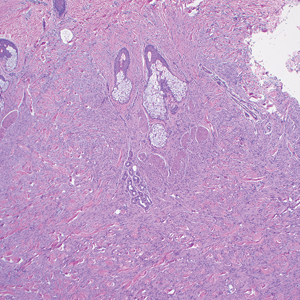
As shown in the Figure, histopathologic examination revealed tubular structures in the upper dermis with characteristic comma-shaped extensions. Some of these structures were lined with cuboidal cells and contained eosinophilic material within the lumen. There was no involvement of the epidermis or deeper dermis. The histologic features were consistent with syringoma, which is distinguished by its predominant involvement of the upper dermis and the presence of enlarged, dilated eccrine ducts, as observed in our case.
Treatment of syringomas often is challenging due to the high rate of recurrence and the risk for postinflammatory hyperpigmentation. Since the condition is benign, treatment typically is pursued for aesthetic reasons. Various therapeutic approaches have been reported, each with diverse response rates. The most common method involves surgical intervention, either with electrodesiccation or CO2 laser—both of which have shown satisfactory resolution of lesions without recurrence at 1-year follow-up, with no major scarring reported.25,26 Alternatively, topical management with retinoids daily over a 4-month period leads to flattening of the tumors with no further appearance of new lesions.27 Despite the availability of numerous management options, establishing a first-line treatment remains controversial due to the high risk for recurrence and the variability in the number and location of lesions among individual patients. In our case, given the benign nature of syringomas, the asymptomatic nature of the lesions, the involvement of noncritical aesthetic areas, and the limited response to noninvasive therapeutic options, the patient was informed of the diagnosis, and no further pharmacologic or surgical intervention was pursued.
- Williams K, Shinkai K. Evaluation and management of the patient with multiple syringomas: a systematic review of the literature. J Am Acad Dermatol. 2016;74:1234-1240.E9. doi:10.1016 /j.jaad.2015.12.006
- Resende C, Araújo C, Santos R, et al. Late-onset of eruptive syringomas: a diagnostic challenge. An Bras Dermatol. 2015;90(3 suppl 1):239-241. doi:10.1590/abd1806-4841.20153899
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol. 1987;16:310-314.
- Avhad G, Ghuge P, Jerajani HR. Generalized eruptive syringoma. Indian J Dermatol. 2015;60:214. doi:10.4103/0019-5154.152586
- Ning WV, Bashey S, Cole C, et al. Multiple eruptive syringomas on the penis. Cutis. 2019;103:E15-E16.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Jamalipour M, Heidarpour M, Rajabi P. Generalized eruptive syringomas. Indian J Dermatol. 2009;54:65-67. doi:10.4103/0019-5154.48992
- Mohaghegh F, Amiri A, Fatemi Naeini F, et al. Acral eruptive syringoma: an unusual presentation with misdiagnosis. Case Rep Dermatol Med. 2020;2020:5416285. doi:10.1155/2020/5416285
- Valdivielso-Ramos M, de la Cueva P, Gimeno M, et al. Acral syringomas. Actas Dermosifiliogr. 2010;101:458-460.
- Patel K, Lundgren AD, Ahmed AM, et al. Disseminated syringomas of the upper extremities in a young woman. Cureus. 2018;10:E3619. doi:10.7759/cureus.3619
- Balci DD, Atik E, Altintas S. Coexistence of acral syringomas and multiple trichoepitheliomas on the face. J Cutan Med Surg. 2009;13:169-171. doi:10.2310/7750.2008.08011
- Martín-García RF, Muñoz CM. Acral syringomas presenting as a photosensitive papular eruption. Cutis. 2006;77:33-36.
- Varas-Meis E, Prada-García C, Samaniego-González E, et al. Acral syringomas associated with hematological neoplasm. Indian J Dermatol Venereol Leprol. 2017;83:136. doi:10.4103/0378-6323.192961
- Berbis P, Fabre JF, Jancovici E, et al. Late-onset syringomas of the upper extremities associated with a carcinoid tumor. Arch Dermatol. 1989;125:848-849.
- Metze D, Jurecka W, Gebhart W. Disseminated syringomas of the upper extremities. case history and immunohistochemical and ultrastructural study. Dermatologica. 1990;180:228-235. doi:10.1159/000248036
- Gómez-de Castro C, Vivanco Allende B, García-García B. Multiple acral syringomas. siringomas acrales múltiples. Actas Dermosifiliogr (Engl Ed). 2018;109:834-836. doi:10.1016/j.ad.2017.10.014
- Hughes PS, Apisarnthanarax P. Acral syringoma. Arch Dermatol. 1977;113:1435-1436.
- Asai Y, Ishii M, Hamada T. Acral syringoma: electron microscopic studies on its origin. Acta Derm Venereol. 1982;62:64-68.
- van den Broek H, Lundquist CD. Syringomas of the upper extremities with onset in the sixth decade. J Am Acad Dermatol. 1982,6:534-536. doi:10.1016/S0190-9622(82)80368-X
- Garcia C, Krunic AL, Grichnik J, et al. Multiple acral syringomata with uniform involvement of the hands and feet. Cutis. 1997;59:213-214, 216.
- Patrizi A, Neri I, Marzaduri S, et al. Syringoma: a review of twenty-nine cases. Acta Derm Venereol. 1998;78:460-462.
- Iglesias Sancho M, Serra Llobet J, Salleras Redonnet M, et al. Siringomas disem- inados de inicio acral, aparecidos en la octava década. Actas Dermosifiliofr. 1999;90:253-257.
- Muniesa C, Fortuño Y, Moreno A, et al. Papules on the dorsum of the fingers. Actas Dermosifiliogr. 2008;99:812-813. doi:10.1016 /S1578-2190(08)70371-8
- Koh MJ. Multiple acral syringomas involving the hands. Clin Exp Dermatol. 2009;34:E438. doi:10.1111/j.1365-2230.2009.03462.x
- Karam P, Benedetto AV. Syringomas: new approach to an old technique. Int J Dermatol. 1996;35:219-220. doi:10.1111/j.1365-4362 .1996.tb01647.x
- Wang JI, Roenigk HH. Treatment of multiple facial syringomas with the carbon dioxide (CO2) laser. Dermatol Surg. 1999;25:136-139. doi:10.1046/j.1524-4725.1999.08111.x
- Gómez MI, Pérez B, Azaña JM, et al. Eruptive syringoma: treatment with topical tretinoin. Dermatology. 2009;189:105-106. doi:10.1159/000246803
THE DIAGNOSIS: Acral Eruptive Syringoma
Syringomas are small, benign, often asymptomatic eccrine tumors that originate in the intraepidermal portion of eccrine sweat ducts.1 Clinically, they present as multiple symmetric white-to-yellow or discrete flesh-colored papules measuring 1 to 3 mm in diameter, often located on the face (most commonly on the eyelids), with a greater prevalence in middle-aged women. Occasionally, they manifest in other locations such as the cheeks, chest, axillae, abdomen, and groin.2
In 1987, Friedman and Butler3 developed a classification system categorizing syringomas into 4 clinical subtypes: familial syringoma, localized syringoma, Down syndrome–related syringoma, and generalized syringoma. The fourth subtype includes the variant of eruptive syringoma,3 a rare clinical manifestation that often develops before or during puberty with several flesh-colored or lightly pigmented papules on the neck, anterior chest, upper abdomen, axillae, periumbilical region, and/or genital region.1,4,5 The etiology of eruptive syringomas is unclear, although it has been linked to abnormal proliferation of sweat glands due to an underlying local inflammatory process.6
Acral distribution of syringomas is a rare variant that can manifest as part of generalized eruptive syringoma with consequent involvement of the arms and other areas.5,7 There are limited case reports on eruptive syringomas with predominant acral distribution.8 Compared to classic syringomas, the acral variant is associated with an older age of onset as well as a similar prevalence between men and women.9 Acral eruptive syringoma (AES) usually is isolated to the distal arms and legs. The most commonly affected region is the anterior surface of the forearms, although involvement of the dorsal hands, wrists, and feet also has been reported.10-16
The first known case of AES, which was reported in 1977, described eruptive syringomas on the dorsal hands of a healthy 31-year-old man.17 Several cases have been reported since then, mostly in patients aged 30 to 60 years, with predominant involvement of the dorsal hands and forearms.18-24 A review of Embase as well as PubMed articles indexed for MEDLINE using the search terms syringoma OR eccrine ductal tumor and eruptive OR acral OR arms OR forearms OR extremities identified 19 reported cases of AES between 1977 and 2023. For the reported AES cases, the mean (SD) age at diagnosis was 45.1 years (15.96 years), with patient ages ranging from 19 to 76 years. Notably, most cases occurred in individuals aged between 30 and 60 years, which deviates from the typical age of onset of localized syringomas, commonly seen during puberty or early adulthood.
Currently, AES is categorized within the clinical presentation of eruptive syringoma. Nevertheless, some authors have proposed classifying it as a distinct fifth clinical group due to specific features that distinguish it from generalized eruptive syringoma.9 This reclassification has considerable implications for the differential diagnosis, particularly because exclusive acral involvement poses a substantial diagnostic challenge and often requires histologic confirmation.
As shown in the Figure, histopathologic examination revealed tubular structures in the upper dermis with characteristic comma-shaped extensions. Some of these structures were lined with cuboidal cells and contained eosinophilic material within the lumen. There was no involvement of the epidermis or deeper dermis. The histologic features were consistent with syringoma, which is distinguished by its predominant involvement of the upper dermis and the presence of enlarged, dilated eccrine ducts, as observed in our case.

Treatment of syringomas often is challenging due to the high rate of recurrence and the risk for postinflammatory hyperpigmentation. Since the condition is benign, treatment typically is pursued for aesthetic reasons. Various therapeutic approaches have been reported, each with diverse response rates. The most common method involves surgical intervention, either with electrodesiccation or CO2 laser—both of which have shown satisfactory resolution of lesions without recurrence at 1-year follow-up, with no major scarring reported.25,26 Alternatively, topical management with retinoids daily over a 4-month period leads to flattening of the tumors with no further appearance of new lesions.27 Despite the availability of numerous management options, establishing a first-line treatment remains controversial due to the high risk for recurrence and the variability in the number and location of lesions among individual patients. In our case, given the benign nature of syringomas, the asymptomatic nature of the lesions, the involvement of noncritical aesthetic areas, and the limited response to noninvasive therapeutic options, the patient was informed of the diagnosis, and no further pharmacologic or surgical intervention was pursued.
THE DIAGNOSIS: Acral Eruptive Syringoma
Syringomas are small, benign, often asymptomatic eccrine tumors that originate in the intraepidermal portion of eccrine sweat ducts.1 Clinically, they present as multiple symmetric white-to-yellow or discrete flesh-colored papules measuring 1 to 3 mm in diameter, often located on the face (most commonly on the eyelids), with a greater prevalence in middle-aged women. Occasionally, they manifest in other locations such as the cheeks, chest, axillae, abdomen, and groin.2
In 1987, Friedman and Butler3 developed a classification system categorizing syringomas into 4 clinical subtypes: familial syringoma, localized syringoma, Down syndrome–related syringoma, and generalized syringoma. The fourth subtype includes the variant of eruptive syringoma,3 a rare clinical manifestation that often develops before or during puberty with several flesh-colored or lightly pigmented papules on the neck, anterior chest, upper abdomen, axillae, periumbilical region, and/or genital region.1,4,5 The etiology of eruptive syringomas is unclear, although it has been linked to abnormal proliferation of sweat glands due to an underlying local inflammatory process.6
Acral distribution of syringomas is a rare variant that can manifest as part of generalized eruptive syringoma with consequent involvement of the arms and other areas.5,7 There are limited case reports on eruptive syringomas with predominant acral distribution.8 Compared to classic syringomas, the acral variant is associated with an older age of onset as well as a similar prevalence between men and women.9 Acral eruptive syringoma (AES) usually is isolated to the distal arms and legs. The most commonly affected region is the anterior surface of the forearms, although involvement of the dorsal hands, wrists, and feet also has been reported.10-16
The first known case of AES, which was reported in 1977, described eruptive syringomas on the dorsal hands of a healthy 31-year-old man.17 Several cases have been reported since then, mostly in patients aged 30 to 60 years, with predominant involvement of the dorsal hands and forearms.18-24 A review of Embase as well as PubMed articles indexed for MEDLINE using the search terms syringoma OR eccrine ductal tumor and eruptive OR acral OR arms OR forearms OR extremities identified 19 reported cases of AES between 1977 and 2023. For the reported AES cases, the mean (SD) age at diagnosis was 45.1 years (15.96 years), with patient ages ranging from 19 to 76 years. Notably, most cases occurred in individuals aged between 30 and 60 years, which deviates from the typical age of onset of localized syringomas, commonly seen during puberty or early adulthood.
Currently, AES is categorized within the clinical presentation of eruptive syringoma. Nevertheless, some authors have proposed classifying it as a distinct fifth clinical group due to specific features that distinguish it from generalized eruptive syringoma.9 This reclassification has considerable implications for the differential diagnosis, particularly because exclusive acral involvement poses a substantial diagnostic challenge and often requires histologic confirmation.
As shown in the Figure, histopathologic examination revealed tubular structures in the upper dermis with characteristic comma-shaped extensions. Some of these structures were lined with cuboidal cells and contained eosinophilic material within the lumen. There was no involvement of the epidermis or deeper dermis. The histologic features were consistent with syringoma, which is distinguished by its predominant involvement of the upper dermis and the presence of enlarged, dilated eccrine ducts, as observed in our case.

Treatment of syringomas often is challenging due to the high rate of recurrence and the risk for postinflammatory hyperpigmentation. Since the condition is benign, treatment typically is pursued for aesthetic reasons. Various therapeutic approaches have been reported, each with diverse response rates. The most common method involves surgical intervention, either with electrodesiccation or CO2 laser—both of which have shown satisfactory resolution of lesions without recurrence at 1-year follow-up, with no major scarring reported.25,26 Alternatively, topical management with retinoids daily over a 4-month period leads to flattening of the tumors with no further appearance of new lesions.27 Despite the availability of numerous management options, establishing a first-line treatment remains controversial due to the high risk for recurrence and the variability in the number and location of lesions among individual patients. In our case, given the benign nature of syringomas, the asymptomatic nature of the lesions, the involvement of noncritical aesthetic areas, and the limited response to noninvasive therapeutic options, the patient was informed of the diagnosis, and no further pharmacologic or surgical intervention was pursued.
- Williams K, Shinkai K. Evaluation and management of the patient with multiple syringomas: a systematic review of the literature. J Am Acad Dermatol. 2016;74:1234-1240.E9. doi:10.1016 /j.jaad.2015.12.006
- Resende C, Araújo C, Santos R, et al. Late-onset of eruptive syringomas: a diagnostic challenge. An Bras Dermatol. 2015;90(3 suppl 1):239-241. doi:10.1590/abd1806-4841.20153899
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol. 1987;16:310-314.
- Avhad G, Ghuge P, Jerajani HR. Generalized eruptive syringoma. Indian J Dermatol. 2015;60:214. doi:10.4103/0019-5154.152586
- Ning WV, Bashey S, Cole C, et al. Multiple eruptive syringomas on the penis. Cutis. 2019;103:E15-E16.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Jamalipour M, Heidarpour M, Rajabi P. Generalized eruptive syringomas. Indian J Dermatol. 2009;54:65-67. doi:10.4103/0019-5154.48992
- Mohaghegh F, Amiri A, Fatemi Naeini F, et al. Acral eruptive syringoma: an unusual presentation with misdiagnosis. Case Rep Dermatol Med. 2020;2020:5416285. doi:10.1155/2020/5416285
- Valdivielso-Ramos M, de la Cueva P, Gimeno M, et al. Acral syringomas. Actas Dermosifiliogr. 2010;101:458-460.
- Patel K, Lundgren AD, Ahmed AM, et al. Disseminated syringomas of the upper extremities in a young woman. Cureus. 2018;10:E3619. doi:10.7759/cureus.3619
- Balci DD, Atik E, Altintas S. Coexistence of acral syringomas and multiple trichoepitheliomas on the face. J Cutan Med Surg. 2009;13:169-171. doi:10.2310/7750.2008.08011
- Martín-García RF, Muñoz CM. Acral syringomas presenting as a photosensitive papular eruption. Cutis. 2006;77:33-36.
- Varas-Meis E, Prada-García C, Samaniego-González E, et al. Acral syringomas associated with hematological neoplasm. Indian J Dermatol Venereol Leprol. 2017;83:136. doi:10.4103/0378-6323.192961
- Berbis P, Fabre JF, Jancovici E, et al. Late-onset syringomas of the upper extremities associated with a carcinoid tumor. Arch Dermatol. 1989;125:848-849.
- Metze D, Jurecka W, Gebhart W. Disseminated syringomas of the upper extremities. case history and immunohistochemical and ultrastructural study. Dermatologica. 1990;180:228-235. doi:10.1159/000248036
- Gómez-de Castro C, Vivanco Allende B, García-García B. Multiple acral syringomas. siringomas acrales múltiples. Actas Dermosifiliogr (Engl Ed). 2018;109:834-836. doi:10.1016/j.ad.2017.10.014
- Hughes PS, Apisarnthanarax P. Acral syringoma. Arch Dermatol. 1977;113:1435-1436.
- Asai Y, Ishii M, Hamada T. Acral syringoma: electron microscopic studies on its origin. Acta Derm Venereol. 1982;62:64-68.
- van den Broek H, Lundquist CD. Syringomas of the upper extremities with onset in the sixth decade. J Am Acad Dermatol. 1982,6:534-536. doi:10.1016/S0190-9622(82)80368-X
- Garcia C, Krunic AL, Grichnik J, et al. Multiple acral syringomata with uniform involvement of the hands and feet. Cutis. 1997;59:213-214, 216.
- Patrizi A, Neri I, Marzaduri S, et al. Syringoma: a review of twenty-nine cases. Acta Derm Venereol. 1998;78:460-462.
- Iglesias Sancho M, Serra Llobet J, Salleras Redonnet M, et al. Siringomas disem- inados de inicio acral, aparecidos en la octava década. Actas Dermosifiliofr. 1999;90:253-257.
- Muniesa C, Fortuño Y, Moreno A, et al. Papules on the dorsum of the fingers. Actas Dermosifiliogr. 2008;99:812-813. doi:10.1016 /S1578-2190(08)70371-8
- Koh MJ. Multiple acral syringomas involving the hands. Clin Exp Dermatol. 2009;34:E438. doi:10.1111/j.1365-2230.2009.03462.x
- Karam P, Benedetto AV. Syringomas: new approach to an old technique. Int J Dermatol. 1996;35:219-220. doi:10.1111/j.1365-4362 .1996.tb01647.x
- Wang JI, Roenigk HH. Treatment of multiple facial syringomas with the carbon dioxide (CO2) laser. Dermatol Surg. 1999;25:136-139. doi:10.1046/j.1524-4725.1999.08111.x
- Gómez MI, Pérez B, Azaña JM, et al. Eruptive syringoma: treatment with topical tretinoin. Dermatology. 2009;189:105-106. doi:10.1159/000246803
- Williams K, Shinkai K. Evaluation and management of the patient with multiple syringomas: a systematic review of the literature. J Am Acad Dermatol. 2016;74:1234-1240.E9. doi:10.1016 /j.jaad.2015.12.006
- Resende C, Araújo C, Santos R, et al. Late-onset of eruptive syringomas: a diagnostic challenge. An Bras Dermatol. 2015;90(3 suppl 1):239-241. doi:10.1590/abd1806-4841.20153899
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol. 1987;16:310-314.
- Avhad G, Ghuge P, Jerajani HR. Generalized eruptive syringoma. Indian J Dermatol. 2015;60:214. doi:10.4103/0019-5154.152586
- Ning WV, Bashey S, Cole C, et al. Multiple eruptive syringomas on the penis. Cutis. 2019;103:E15-E16.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Jamalipour M, Heidarpour M, Rajabi P. Generalized eruptive syringomas. Indian J Dermatol. 2009;54:65-67. doi:10.4103/0019-5154.48992
- Mohaghegh F, Amiri A, Fatemi Naeini F, et al. Acral eruptive syringoma: an unusual presentation with misdiagnosis. Case Rep Dermatol Med. 2020;2020:5416285. doi:10.1155/2020/5416285
- Valdivielso-Ramos M, de la Cueva P, Gimeno M, et al. Acral syringomas. Actas Dermosifiliogr. 2010;101:458-460.
- Patel K, Lundgren AD, Ahmed AM, et al. Disseminated syringomas of the upper extremities in a young woman. Cureus. 2018;10:E3619. doi:10.7759/cureus.3619
- Balci DD, Atik E, Altintas S. Coexistence of acral syringomas and multiple trichoepitheliomas on the face. J Cutan Med Surg. 2009;13:169-171. doi:10.2310/7750.2008.08011
- Martín-García RF, Muñoz CM. Acral syringomas presenting as a photosensitive papular eruption. Cutis. 2006;77:33-36.
- Varas-Meis E, Prada-García C, Samaniego-González E, et al. Acral syringomas associated with hematological neoplasm. Indian J Dermatol Venereol Leprol. 2017;83:136. doi:10.4103/0378-6323.192961
- Berbis P, Fabre JF, Jancovici E, et al. Late-onset syringomas of the upper extremities associated with a carcinoid tumor. Arch Dermatol. 1989;125:848-849.
- Metze D, Jurecka W, Gebhart W. Disseminated syringomas of the upper extremities. case history and immunohistochemical and ultrastructural study. Dermatologica. 1990;180:228-235. doi:10.1159/000248036
- Gómez-de Castro C, Vivanco Allende B, García-García B. Multiple acral syringomas. siringomas acrales múltiples. Actas Dermosifiliogr (Engl Ed). 2018;109:834-836. doi:10.1016/j.ad.2017.10.014
- Hughes PS, Apisarnthanarax P. Acral syringoma. Arch Dermatol. 1977;113:1435-1436.
- Asai Y, Ishii M, Hamada T. Acral syringoma: electron microscopic studies on its origin. Acta Derm Venereol. 1982;62:64-68.
- van den Broek H, Lundquist CD. Syringomas of the upper extremities with onset in the sixth decade. J Am Acad Dermatol. 1982,6:534-536. doi:10.1016/S0190-9622(82)80368-X
- Garcia C, Krunic AL, Grichnik J, et al. Multiple acral syringomata with uniform involvement of the hands and feet. Cutis. 1997;59:213-214, 216.
- Patrizi A, Neri I, Marzaduri S, et al. Syringoma: a review of twenty-nine cases. Acta Derm Venereol. 1998;78:460-462.
- Iglesias Sancho M, Serra Llobet J, Salleras Redonnet M, et al. Siringomas disem- inados de inicio acral, aparecidos en la octava década. Actas Dermosifiliofr. 1999;90:253-257.
- Muniesa C, Fortuño Y, Moreno A, et al. Papules on the dorsum of the fingers. Actas Dermosifiliogr. 2008;99:812-813. doi:10.1016 /S1578-2190(08)70371-8
- Koh MJ. Multiple acral syringomas involving the hands. Clin Exp Dermatol. 2009;34:E438. doi:10.1111/j.1365-2230.2009.03462.x
- Karam P, Benedetto AV. Syringomas: new approach to an old technique. Int J Dermatol. 1996;35:219-220. doi:10.1111/j.1365-4362 .1996.tb01647.x
- Wang JI, Roenigk HH. Treatment of multiple facial syringomas with the carbon dioxide (CO2) laser. Dermatol Surg. 1999;25:136-139. doi:10.1046/j.1524-4725.1999.08111.x
- Gómez MI, Pérez B, Azaña JM, et al. Eruptive syringoma: treatment with topical tretinoin. Dermatology. 2009;189:105-106. doi:10.1159/000246803
Eruptive Erythematous Papules on the Forearms
Eruptive Erythematous Papules on the Forearms
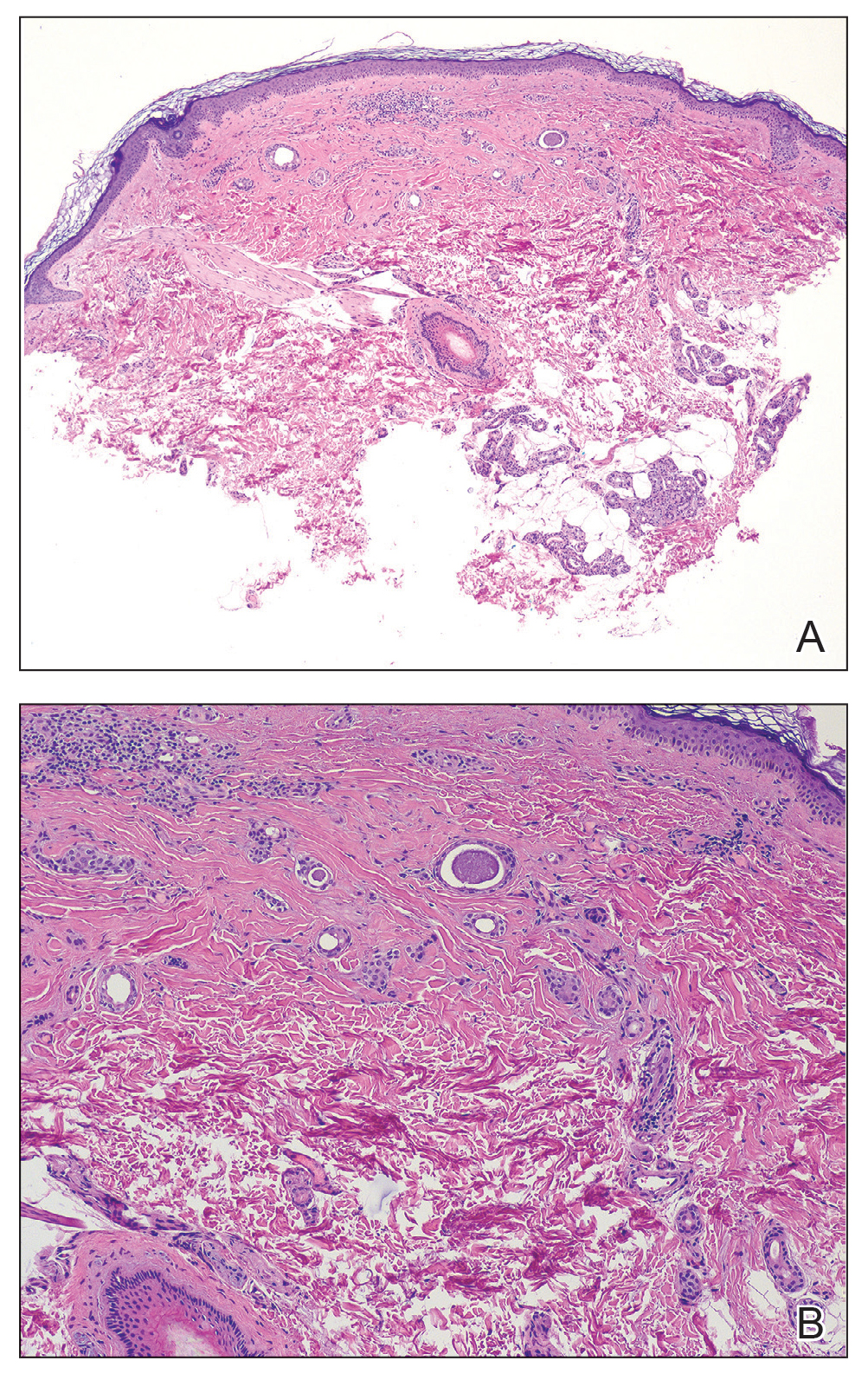
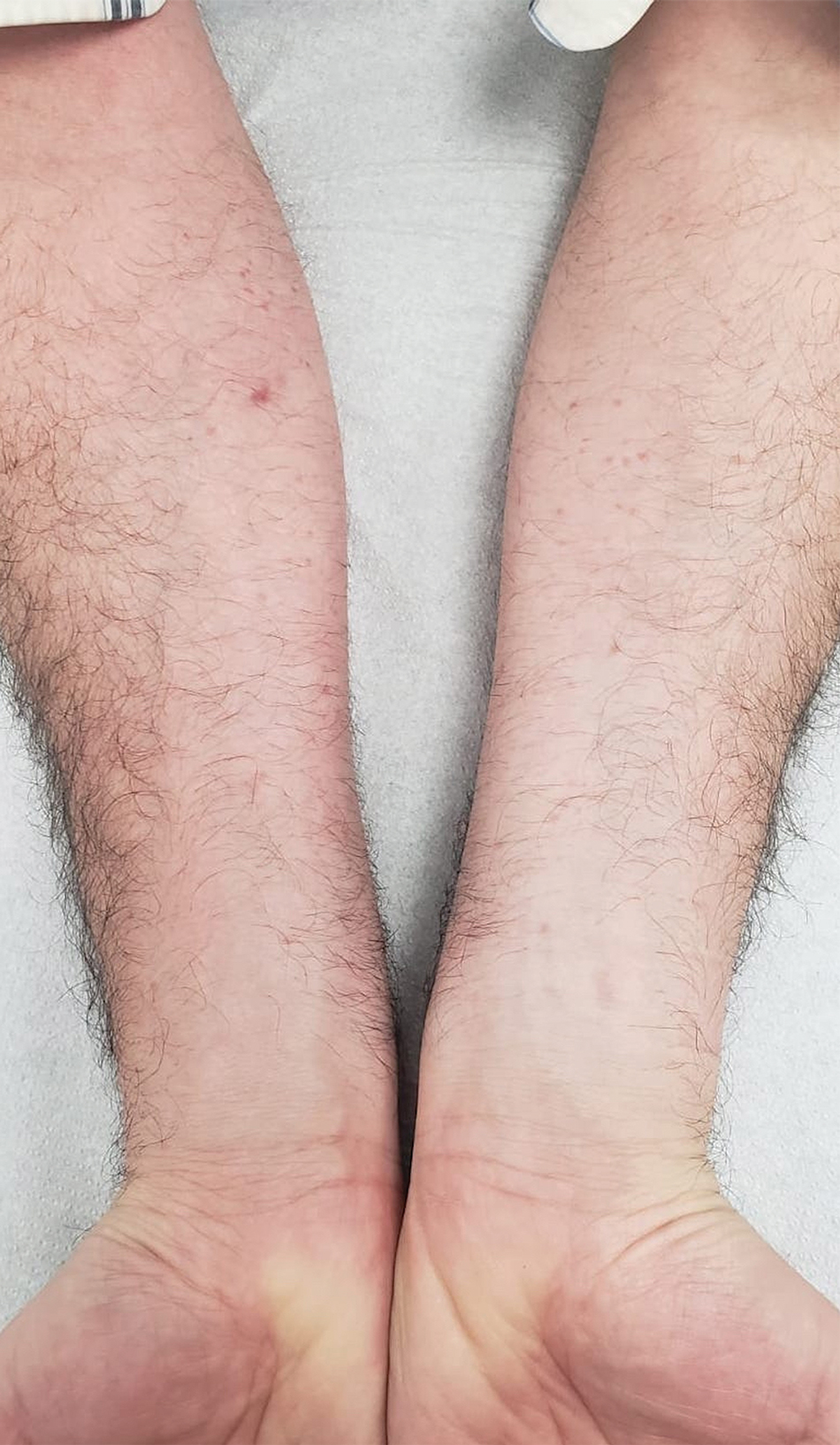
A 44-year-old man presented to the dermatology department with multiple eruptive, nonconfluent, erythematous papules on the anterior forearms of 2 years’ duration. The patient’s medical history was notable for right-sided testicular cancer diagnosed in childhood and 3 excised basal cell carcinomas, the most recent of which was concurrent with the present case. The patient denied any recent pruritus, exposure to irritants, or use of over-the-counter medications. Physical examination was remarkable for numerous monomorphic, symmetric, nonconfluent, flesh-colored to slightly pigmented papules on the dorsal aspect of the forearms. No involvement of the fingers or lower extremities was observed. Two punch biopsies of representative lesions on the right and left forearms were taken. Histopathologic examination revealed eccrine ductal proliferations lined by cuboidal cells embedded within bundles of sclerotic collagen.
Actinic Keratosis Treatment With Diclofenac Gel 1%
Actinic Keratosis Treatment With Diclofenac Gel 1%
To the Editor:
Actinic keratoses (AKs) are keratinocyte neoplasms that manifest as rough, scaly, erythematous papules with ill-defined borders (commonly known as precancers) and develop due to long-term UV light exposure.1 They must be treated promptly due to the risk for progression to squamous cell carcinoma (SCC). One US Department of Veterans Affairs study reported that 0.6% of AKs progress to SCC in 1 year and 2.6% progressed to SCC in 4 years.2 In 10% of AKs that will progress to SCC, one study reported progression in approximately 2 years.3
The risk for progression also increases in patients with multiple AKs; the risk is 4-fold higher in patients with 6 to 20 AKs and 11-fold higher in patients with more than 20 AKs.4 Common treatment options include lesion-directed therapies such as cryotherapy, laser therapy, surgery, and curettage, as well as field-directed therapies such as topical 5-fluorouracil (5-FU), diclofenac gel 3%, chemical peeling, topical imiquimod, and photodynamic therapy (PDT).4 When diclofenac gel is chosen as a treatment modality, it is commonly prescribed in the 3% formulation. Diclofenac gel 3% has been shown to be effective in the treatment of AKs,5,6 but diclofenac gel 1% has not been well described in the literature. We report the case of a patient with AKs on the lower legs who was treated with diclofenac gel after other therapies failed.
A 55-year-old woman presented for a routine skin check due to a history of nonmelanoma skin cancer. Her medical history also included palmar hyperhidrosis, disseminated superficial actinic porokeratosis, and extensive actinic damage, as well as numerous biopsy-proven AKs. She had been evaluated every 3 months up to presentation due to the frequency of AK development over the past 5 years. The lesions were mainly localized to both lower legs, where the patient had acquired considerable lifetime sun exposure from tanning beds and sunbathing while boating. She also noted exposure to well water as a child, but none of her family members had a similar issue with AKs.
Prior to this visit, the patient had undergone 5 years of therapy for AKs. She initially was treated with multiple courses of topical 5-FU, but she consequently developed severe allergic contact dermatitis. Subsequent treatments included cryotherapy as well as application of tretinoin cream nightly for 2 weeks followed by PDT. She was unable to tolerate the tretinoin, which she reported led to dryness and irritation. She reported mild improvement after her first session of PDT but only minimal improvement after the next session. Ingenol mebutate was then prescribed for topical use on the legs for 2 days, which did not result in improvement. The patient continued to follow up for unresolved AKs on the legs and was prescribed acitretin to help reduce the risk for progression to SCC. At follow-up 3 months later, she reported decreased soreness from AKs after starting the acitretin and, aside from mild dryness, she tolerated the medication well; however, with continued use of acitretin, she began to experience adverse effects 6 months later, including thyroid suppression and hair loss, leading to discontinuation. Instead, 3 months later, she was recommended to start nicotinamide supplementation for prevention of SCC.
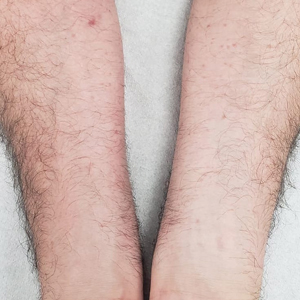
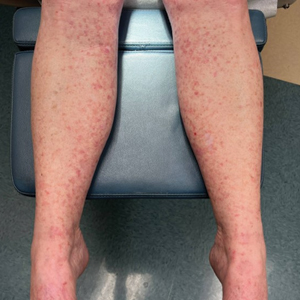
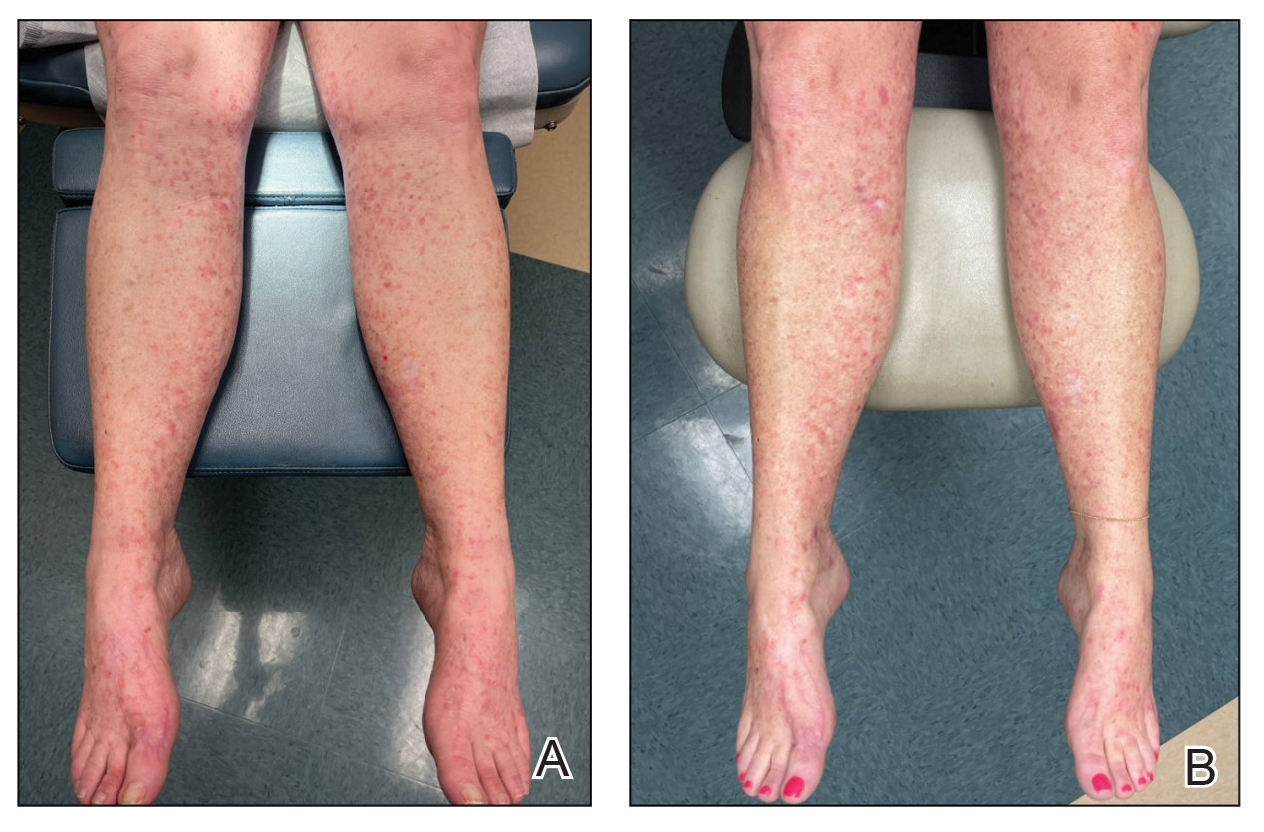
Due to continued AK development (Figure, A), we eventually prescribed diclofenac gel 3% twice daily for both legs 9 months after prescribing nicotinamide. This regimen was cost prohibitive, as the medication was not covered by her insurance and the cost was $300 for one tube. We recommended the patient instead apply the 3% gel to the right leg only due to greater severity of AKs on this leg and over-the-counter diclofenac gel 1% twice daily to the left leg. Approximately 5 months later, she reported a reduction in the discomfort from AKs as well as a reduction in the total number of AKs. She applied the 2 different products as instructed for the first month but did not notice a difference between them. She then continued to apply only the 1% gel on both legs for a total of 8 months with excellent response (Figure, B). At subsequent follow-up visits over a 2-year period, she has only required cryotherapy as spot treatment for AKs.
For 1 to a few discrete AKs, liquid nitrogen cryotherapy is considered first-line therapy.7 However, if multiple AKs are present, surrounding photodamaged skin also should be treated with field-directed therapy due to surrounding keratinocytes bearing a high mutational burden and risk of cancerization.8 Common field-directed therapies include topical 5-FU, topical imiquimod, topical tirbanibulin, PDT, retinoids, and topical diclofenac 3%.
One challenge in field-directed treatment of AKs is the side-effect profile seen in some patients, causing them to prematurely discontinue treatment. In our patient, 5-FU cream, tretinoin cream, and oral acitretin were not well tolerated. Topical diclofenac generally is well tolerated, with mostly mild local skin reactions and low risk for systemic adverse events. Adverse effects mainly consist of mild local skin reactions including pruritus (reported in 31%-52% of patients who used topical diclofenac), dryness (25%-27%), and irritation (less than 1%).9,10 Although diclofenac carries a black-box warning for serious cardiovascular thrombotic events and serious gastrointestinal tract bleeding, systemic absorption of topical diclofenac has been proven to be substantially lower (5- to 17-fold) compared to the oral formulation, and resulting serious adverse effects have been found to be largely reduced compared to the oral formulation.11,12 If allergic contact dermatitis develops, diclofenac should be discontinued.9,13
Diclofenac’s antineoplastic mechanism of action of cyclooxygenase-2 inhibition involves induction of apoptosis as well as reduction in tumor cell proliferation and tumor angiogenesis.14,15 Topical diclofenac may result in decreased levels of lactate and amino acid in AK lesions, particularly in lesions responding to treatment.16 Topical diclofenac may alter immune infiltration by inducing infiltration of dermal CD8+ T cells along with high IFN-γ messenger RNA expression, suggesting improvement of T-cell function after topical diclofenac treatment.16
Although diclofenac gel 3% has been shown to be effective in treatment of AKs,5,6 diclofenac gel 1% has not yet been well studied. Use of the 1% gel is indicated for osteoarthritis and musculoskeletal pain by the US Food and Drug Administration.10,17 Efficacy of the 1% gel has been documented for these and other conditions including seborrheic keratoses.18-20
Because the 1% diclofenac formulation is available over-the-counter, it is more accessible to patients compared to the 3% formulation and often substantially decreases the cost of the medication for the patient. The cost of diclofenac gel 1% in the United States ranges from $0.04 to $0.31 per gram compared to $1.07 to $11.79 per gram for the 3% gel prescription formulation.17 Efficacy of the 1% formulation compared to the 3% formulation could represent an avenue to increase accessibility to field-directed therapy in the population for the treatment of AKs with a potentially well-tolerated, effective, and low-cost medication formulation.
This case represents the effectiveness of diclofenac gel 1% in treating AKs. Several treatment modalities failed in our case, but she experienced improvement with use of over-the-counter diclofenac gel 1%. She also noted no difference in response between the prescription 3% diclofenac formulation and the over-the-counter 1% formulation. Diclofenac gel 1% may represent an excellent therapeutic option in treatment-refractory cases of AKs. Larger randomized trials should be considered to assess safety and efficacy.
- FEisen DB, Asgari MM, Bennett DD, et al. Guidelines of care for the management of actinic keratosis. J Am Acad Dermatol. 2021;85:e209-e233.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Fuchs A, Marmur E. The kinetics of skin cancer: progression of actinic keratosis to squamous cell carcinoma. Dermatol Surg. 2007;33: 1099-1101.
- Dianzani C, Conforti C, Giuffrida R, et al. Current therapies for actinic keratosis. Int J Dermatol. 2020;59:677-684.
- Javor S, Cozzani E, Parodi A. Topical treatment of actinic keratosis with 3.0% diclofenac in 2.5% hyaluronan gel: review of the literature about the cumulative evidence of its efficacy and safety. G Ital Dermatol Venereol. 2016;151:275-280.
- Martin GM, Stockfleth E. Diclofenac sodium 3% gel for the management of actinic keratosis: 10+ years of cumulative evidence of efficacy and safety. J Drugs Dermatol. 2012;11:600-608.
- Arisi M, Guasco Pisani E, et al. Cryotherapy for actinic keratosis: basic principles and literature review. Clin Cosmet Investig Dermatol. 2022;15:357-365.
- Calzavara-Pinton P, Calzavara-Pinton I, Rovati C, et al. Topical pharmacotherapy for actinic keratoses in older adults. Drugs Aging. 2022;39:143-152.
- Beutner C, Forkel S, Kreipe K, et al. Contact allergy to topical diclofenac with systemic tolerance. Contact Dermatitis. 2022;86:41-43.
- Voltaren gel (diclofenac sodium topical gel). Prescribing information. Novartis Consumer Health, Inc; 2009. Accessed May 21, 2025. https:// www.accessdata.fda.gov/drugsatfda_docs/label/2009/022122s006lbl.pdf
- Moreira SA, Liu DJ. Diclofenac systemic bioavailability of a topical 1% diclofenac + 3% menthol combination gel vs. an oral diclofenac tablet in healthy volunteers: a randomized, open-label, crossover study. Int J Clin Pharmacol Ther. 2017;55:368-372.
- Kienzler JL, Gold M, Nollevaux F. Systemic bioavailability of topical diclofenac sodium gel 1% versus oral diclofenac sodium in healthy volunteers. J Clin Pharmacol. 2010;50:50-61.
- Gulin SJ, Chiriac A. Diclofenac-induced allergic contact dermatitis: a series of four patients. Drug Saf Case Rep. 2016;3:15.
- Fecker LF, Stockfleth E, Nindl I, et al. The role of apoptosis in therapy and prophylaxis of epithelial tumours by nonsteroidal antiinflammatory drugs (NSAIDs). Br J Dermatol. 2007;156(Suppl 3):25-33.
- Thomas GJ, Herranz P, Cruz SB, et al. Treatment of actinic keratosis through inhibition of cyclooxygenase-2: potential mechanism of action of diclofenac sodium 3% in hyaluronic acid 2.5. Dermatol Ther. 2019;32:e12800.
- Singer K, Dettmer K, Unger P, et al. Topical diclofenac reprograms metabolism and immune cell infiltration in actinic keratosis. Front Oncol. 2019;9:605.
- Diclofenac (topical). Drug information. UpToDate. https://www-uptodate-com.libraryaccess.elpaso.ttuhsc.edu/contents/diclofenac-topical-drug-information?source=auto_suggest&selectedTitle=1~3---3~4---diclofenac&search=diclofenac%20topical#F8017265
- Afify AA, Hana MR. Comparative evaluation of topical diclofenac sodium versus topical ibuprofen in the treatment of seborrheic keratosis. Dermatol Ther. 2020;33:e14370.
- Yin F, Ma J, Xiao H, et al. Randomized, double-blind, noninferiority study of diclofenac diethylamine 2.32% gel applied twice daily versus diclofenac diethylamine 1.16% gel applied four times daily in patients with acute ankle sprain. BMC Musculoskelet Disord. 2022;23:1125.
- van Herwaarden N, van den Elsen GAH, de Jong ICA, et al. Topical NSAIDs: ineffective or undervalued? [in Dutch]. Ned Tijdschr Geneeskd. 2021;165:D5317.
To the Editor:
Actinic keratoses (AKs) are keratinocyte neoplasms that manifest as rough, scaly, erythematous papules with ill-defined borders (commonly known as precancers) and develop due to long-term UV light exposure.1 They must be treated promptly due to the risk for progression to squamous cell carcinoma (SCC). One US Department of Veterans Affairs study reported that 0.6% of AKs progress to SCC in 1 year and 2.6% progressed to SCC in 4 years.2 In 10% of AKs that will progress to SCC, one study reported progression in approximately 2 years.3
The risk for progression also increases in patients with multiple AKs; the risk is 4-fold higher in patients with 6 to 20 AKs and 11-fold higher in patients with more than 20 AKs.4 Common treatment options include lesion-directed therapies such as cryotherapy, laser therapy, surgery, and curettage, as well as field-directed therapies such as topical 5-fluorouracil (5-FU), diclofenac gel 3%, chemical peeling, topical imiquimod, and photodynamic therapy (PDT).4 When diclofenac gel is chosen as a treatment modality, it is commonly prescribed in the 3% formulation. Diclofenac gel 3% has been shown to be effective in the treatment of AKs,5,6 but diclofenac gel 1% has not been well described in the literature. We report the case of a patient with AKs on the lower legs who was treated with diclofenac gel after other therapies failed.
A 55-year-old woman presented for a routine skin check due to a history of nonmelanoma skin cancer. Her medical history also included palmar hyperhidrosis, disseminated superficial actinic porokeratosis, and extensive actinic damage, as well as numerous biopsy-proven AKs. She had been evaluated every 3 months up to presentation due to the frequency of AK development over the past 5 years. The lesions were mainly localized to both lower legs, where the patient had acquired considerable lifetime sun exposure from tanning beds and sunbathing while boating. She also noted exposure to well water as a child, but none of her family members had a similar issue with AKs.
Prior to this visit, the patient had undergone 5 years of therapy for AKs. She initially was treated with multiple courses of topical 5-FU, but she consequently developed severe allergic contact dermatitis. Subsequent treatments included cryotherapy as well as application of tretinoin cream nightly for 2 weeks followed by PDT. She was unable to tolerate the tretinoin, which she reported led to dryness and irritation. She reported mild improvement after her first session of PDT but only minimal improvement after the next session. Ingenol mebutate was then prescribed for topical use on the legs for 2 days, which did not result in improvement. The patient continued to follow up for unresolved AKs on the legs and was prescribed acitretin to help reduce the risk for progression to SCC. At follow-up 3 months later, she reported decreased soreness from AKs after starting the acitretin and, aside from mild dryness, she tolerated the medication well; however, with continued use of acitretin, she began to experience adverse effects 6 months later, including thyroid suppression and hair loss, leading to discontinuation. Instead, 3 months later, she was recommended to start nicotinamide supplementation for prevention of SCC.
Due to continued AK development (Figure, A), we eventually prescribed diclofenac gel 3% twice daily for both legs 9 months after prescribing nicotinamide. This regimen was cost prohibitive, as the medication was not covered by her insurance and the cost was $300 for one tube. We recommended the patient instead apply the 3% gel to the right leg only due to greater severity of AKs on this leg and over-the-counter diclofenac gel 1% twice daily to the left leg. Approximately 5 months later, she reported a reduction in the discomfort from AKs as well as a reduction in the total number of AKs. She applied the 2 different products as instructed for the first month but did not notice a difference between them. She then continued to apply only the 1% gel on both legs for a total of 8 months with excellent response (Figure, B). At subsequent follow-up visits over a 2-year period, she has only required cryotherapy as spot treatment for AKs.

For 1 to a few discrete AKs, liquid nitrogen cryotherapy is considered first-line therapy.7 However, if multiple AKs are present, surrounding photodamaged skin also should be treated with field-directed therapy due to surrounding keratinocytes bearing a high mutational burden and risk of cancerization.8 Common field-directed therapies include topical 5-FU, topical imiquimod, topical tirbanibulin, PDT, retinoids, and topical diclofenac 3%.
One challenge in field-directed treatment of AKs is the side-effect profile seen in some patients, causing them to prematurely discontinue treatment. In our patient, 5-FU cream, tretinoin cream, and oral acitretin were not well tolerated. Topical diclofenac generally is well tolerated, with mostly mild local skin reactions and low risk for systemic adverse events. Adverse effects mainly consist of mild local skin reactions including pruritus (reported in 31%-52% of patients who used topical diclofenac), dryness (25%-27%), and irritation (less than 1%).9,10 Although diclofenac carries a black-box warning for serious cardiovascular thrombotic events and serious gastrointestinal tract bleeding, systemic absorption of topical diclofenac has been proven to be substantially lower (5- to 17-fold) compared to the oral formulation, and resulting serious adverse effects have been found to be largely reduced compared to the oral formulation.11,12 If allergic contact dermatitis develops, diclofenac should be discontinued.9,13
Diclofenac’s antineoplastic mechanism of action of cyclooxygenase-2 inhibition involves induction of apoptosis as well as reduction in tumor cell proliferation and tumor angiogenesis.14,15 Topical diclofenac may result in decreased levels of lactate and amino acid in AK lesions, particularly in lesions responding to treatment.16 Topical diclofenac may alter immune infiltration by inducing infiltration of dermal CD8+ T cells along with high IFN-γ messenger RNA expression, suggesting improvement of T-cell function after topical diclofenac treatment.16
Although diclofenac gel 3% has been shown to be effective in treatment of AKs,5,6 diclofenac gel 1% has not yet been well studied. Use of the 1% gel is indicated for osteoarthritis and musculoskeletal pain by the US Food and Drug Administration.10,17 Efficacy of the 1% gel has been documented for these and other conditions including seborrheic keratoses.18-20
Because the 1% diclofenac formulation is available over-the-counter, it is more accessible to patients compared to the 3% formulation and often substantially decreases the cost of the medication for the patient. The cost of diclofenac gel 1% in the United States ranges from $0.04 to $0.31 per gram compared to $1.07 to $11.79 per gram for the 3% gel prescription formulation.17 Efficacy of the 1% formulation compared to the 3% formulation could represent an avenue to increase accessibility to field-directed therapy in the population for the treatment of AKs with a potentially well-tolerated, effective, and low-cost medication formulation.
This case represents the effectiveness of diclofenac gel 1% in treating AKs. Several treatment modalities failed in our case, but she experienced improvement with use of over-the-counter diclofenac gel 1%. She also noted no difference in response between the prescription 3% diclofenac formulation and the over-the-counter 1% formulation. Diclofenac gel 1% may represent an excellent therapeutic option in treatment-refractory cases of AKs. Larger randomized trials should be considered to assess safety and efficacy.
To the Editor:
Actinic keratoses (AKs) are keratinocyte neoplasms that manifest as rough, scaly, erythematous papules with ill-defined borders (commonly known as precancers) and develop due to long-term UV light exposure.1 They must be treated promptly due to the risk for progression to squamous cell carcinoma (SCC). One US Department of Veterans Affairs study reported that 0.6% of AKs progress to SCC in 1 year and 2.6% progressed to SCC in 4 years.2 In 10% of AKs that will progress to SCC, one study reported progression in approximately 2 years.3
The risk for progression also increases in patients with multiple AKs; the risk is 4-fold higher in patients with 6 to 20 AKs and 11-fold higher in patients with more than 20 AKs.4 Common treatment options include lesion-directed therapies such as cryotherapy, laser therapy, surgery, and curettage, as well as field-directed therapies such as topical 5-fluorouracil (5-FU), diclofenac gel 3%, chemical peeling, topical imiquimod, and photodynamic therapy (PDT).4 When diclofenac gel is chosen as a treatment modality, it is commonly prescribed in the 3% formulation. Diclofenac gel 3% has been shown to be effective in the treatment of AKs,5,6 but diclofenac gel 1% has not been well described in the literature. We report the case of a patient with AKs on the lower legs who was treated with diclofenac gel after other therapies failed.
A 55-year-old woman presented for a routine skin check due to a history of nonmelanoma skin cancer. Her medical history also included palmar hyperhidrosis, disseminated superficial actinic porokeratosis, and extensive actinic damage, as well as numerous biopsy-proven AKs. She had been evaluated every 3 months up to presentation due to the frequency of AK development over the past 5 years. The lesions were mainly localized to both lower legs, where the patient had acquired considerable lifetime sun exposure from tanning beds and sunbathing while boating. She also noted exposure to well water as a child, but none of her family members had a similar issue with AKs.
Prior to this visit, the patient had undergone 5 years of therapy for AKs. She initially was treated with multiple courses of topical 5-FU, but she consequently developed severe allergic contact dermatitis. Subsequent treatments included cryotherapy as well as application of tretinoin cream nightly for 2 weeks followed by PDT. She was unable to tolerate the tretinoin, which she reported led to dryness and irritation. She reported mild improvement after her first session of PDT but only minimal improvement after the next session. Ingenol mebutate was then prescribed for topical use on the legs for 2 days, which did not result in improvement. The patient continued to follow up for unresolved AKs on the legs and was prescribed acitretin to help reduce the risk for progression to SCC. At follow-up 3 months later, she reported decreased soreness from AKs after starting the acitretin and, aside from mild dryness, she tolerated the medication well; however, with continued use of acitretin, she began to experience adverse effects 6 months later, including thyroid suppression and hair loss, leading to discontinuation. Instead, 3 months later, she was recommended to start nicotinamide supplementation for prevention of SCC.
Due to continued AK development (Figure, A), we eventually prescribed diclofenac gel 3% twice daily for both legs 9 months after prescribing nicotinamide. This regimen was cost prohibitive, as the medication was not covered by her insurance and the cost was $300 for one tube. We recommended the patient instead apply the 3% gel to the right leg only due to greater severity of AKs on this leg and over-the-counter diclofenac gel 1% twice daily to the left leg. Approximately 5 months later, she reported a reduction in the discomfort from AKs as well as a reduction in the total number of AKs. She applied the 2 different products as instructed for the first month but did not notice a difference between them. She then continued to apply only the 1% gel on both legs for a total of 8 months with excellent response (Figure, B). At subsequent follow-up visits over a 2-year period, she has only required cryotherapy as spot treatment for AKs.

For 1 to a few discrete AKs, liquid nitrogen cryotherapy is considered first-line therapy.7 However, if multiple AKs are present, surrounding photodamaged skin also should be treated with field-directed therapy due to surrounding keratinocytes bearing a high mutational burden and risk of cancerization.8 Common field-directed therapies include topical 5-FU, topical imiquimod, topical tirbanibulin, PDT, retinoids, and topical diclofenac 3%.
One challenge in field-directed treatment of AKs is the side-effect profile seen in some patients, causing them to prematurely discontinue treatment. In our patient, 5-FU cream, tretinoin cream, and oral acitretin were not well tolerated. Topical diclofenac generally is well tolerated, with mostly mild local skin reactions and low risk for systemic adverse events. Adverse effects mainly consist of mild local skin reactions including pruritus (reported in 31%-52% of patients who used topical diclofenac), dryness (25%-27%), and irritation (less than 1%).9,10 Although diclofenac carries a black-box warning for serious cardiovascular thrombotic events and serious gastrointestinal tract bleeding, systemic absorption of topical diclofenac has been proven to be substantially lower (5- to 17-fold) compared to the oral formulation, and resulting serious adverse effects have been found to be largely reduced compared to the oral formulation.11,12 If allergic contact dermatitis develops, diclofenac should be discontinued.9,13
Diclofenac’s antineoplastic mechanism of action of cyclooxygenase-2 inhibition involves induction of apoptosis as well as reduction in tumor cell proliferation and tumor angiogenesis.14,15 Topical diclofenac may result in decreased levels of lactate and amino acid in AK lesions, particularly in lesions responding to treatment.16 Topical diclofenac may alter immune infiltration by inducing infiltration of dermal CD8+ T cells along with high IFN-γ messenger RNA expression, suggesting improvement of T-cell function after topical diclofenac treatment.16
Although diclofenac gel 3% has been shown to be effective in treatment of AKs,5,6 diclofenac gel 1% has not yet been well studied. Use of the 1% gel is indicated for osteoarthritis and musculoskeletal pain by the US Food and Drug Administration.10,17 Efficacy of the 1% gel has been documented for these and other conditions including seborrheic keratoses.18-20
Because the 1% diclofenac formulation is available over-the-counter, it is more accessible to patients compared to the 3% formulation and often substantially decreases the cost of the medication for the patient. The cost of diclofenac gel 1% in the United States ranges from $0.04 to $0.31 per gram compared to $1.07 to $11.79 per gram for the 3% gel prescription formulation.17 Efficacy of the 1% formulation compared to the 3% formulation could represent an avenue to increase accessibility to field-directed therapy in the population for the treatment of AKs with a potentially well-tolerated, effective, and low-cost medication formulation.
This case represents the effectiveness of diclofenac gel 1% in treating AKs. Several treatment modalities failed in our case, but she experienced improvement with use of over-the-counter diclofenac gel 1%. She also noted no difference in response between the prescription 3% diclofenac formulation and the over-the-counter 1% formulation. Diclofenac gel 1% may represent an excellent therapeutic option in treatment-refractory cases of AKs. Larger randomized trials should be considered to assess safety and efficacy.
- FEisen DB, Asgari MM, Bennett DD, et al. Guidelines of care for the management of actinic keratosis. J Am Acad Dermatol. 2021;85:e209-e233.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Fuchs A, Marmur E. The kinetics of skin cancer: progression of actinic keratosis to squamous cell carcinoma. Dermatol Surg. 2007;33: 1099-1101.
- Dianzani C, Conforti C, Giuffrida R, et al. Current therapies for actinic keratosis. Int J Dermatol. 2020;59:677-684.
- Javor S, Cozzani E, Parodi A. Topical treatment of actinic keratosis with 3.0% diclofenac in 2.5% hyaluronan gel: review of the literature about the cumulative evidence of its efficacy and safety. G Ital Dermatol Venereol. 2016;151:275-280.
- Martin GM, Stockfleth E. Diclofenac sodium 3% gel for the management of actinic keratosis: 10+ years of cumulative evidence of efficacy and safety. J Drugs Dermatol. 2012;11:600-608.
- Arisi M, Guasco Pisani E, et al. Cryotherapy for actinic keratosis: basic principles and literature review. Clin Cosmet Investig Dermatol. 2022;15:357-365.
- Calzavara-Pinton P, Calzavara-Pinton I, Rovati C, et al. Topical pharmacotherapy for actinic keratoses in older adults. Drugs Aging. 2022;39:143-152.
- Beutner C, Forkel S, Kreipe K, et al. Contact allergy to topical diclofenac with systemic tolerance. Contact Dermatitis. 2022;86:41-43.
- Voltaren gel (diclofenac sodium topical gel). Prescribing information. Novartis Consumer Health, Inc; 2009. Accessed May 21, 2025. https:// www.accessdata.fda.gov/drugsatfda_docs/label/2009/022122s006lbl.pdf
- Moreira SA, Liu DJ. Diclofenac systemic bioavailability of a topical 1% diclofenac + 3% menthol combination gel vs. an oral diclofenac tablet in healthy volunteers: a randomized, open-label, crossover study. Int J Clin Pharmacol Ther. 2017;55:368-372.
- Kienzler JL, Gold M, Nollevaux F. Systemic bioavailability of topical diclofenac sodium gel 1% versus oral diclofenac sodium in healthy volunteers. J Clin Pharmacol. 2010;50:50-61.
- Gulin SJ, Chiriac A. Diclofenac-induced allergic contact dermatitis: a series of four patients. Drug Saf Case Rep. 2016;3:15.
- Fecker LF, Stockfleth E, Nindl I, et al. The role of apoptosis in therapy and prophylaxis of epithelial tumours by nonsteroidal antiinflammatory drugs (NSAIDs). Br J Dermatol. 2007;156(Suppl 3):25-33.
- Thomas GJ, Herranz P, Cruz SB, et al. Treatment of actinic keratosis through inhibition of cyclooxygenase-2: potential mechanism of action of diclofenac sodium 3% in hyaluronic acid 2.5. Dermatol Ther. 2019;32:e12800.
- Singer K, Dettmer K, Unger P, et al. Topical diclofenac reprograms metabolism and immune cell infiltration in actinic keratosis. Front Oncol. 2019;9:605.
- Diclofenac (topical). Drug information. UpToDate. https://www-uptodate-com.libraryaccess.elpaso.ttuhsc.edu/contents/diclofenac-topical-drug-information?source=auto_suggest&selectedTitle=1~3---3~4---diclofenac&search=diclofenac%20topical#F8017265
- Afify AA, Hana MR. Comparative evaluation of topical diclofenac sodium versus topical ibuprofen in the treatment of seborrheic keratosis. Dermatol Ther. 2020;33:e14370.
- Yin F, Ma J, Xiao H, et al. Randomized, double-blind, noninferiority study of diclofenac diethylamine 2.32% gel applied twice daily versus diclofenac diethylamine 1.16% gel applied four times daily in patients with acute ankle sprain. BMC Musculoskelet Disord. 2022;23:1125.
- van Herwaarden N, van den Elsen GAH, de Jong ICA, et al. Topical NSAIDs: ineffective or undervalued? [in Dutch]. Ned Tijdschr Geneeskd. 2021;165:D5317.
- FEisen DB, Asgari MM, Bennett DD, et al. Guidelines of care for the management of actinic keratosis. J Am Acad Dermatol. 2021;85:e209-e233.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Fuchs A, Marmur E. The kinetics of skin cancer: progression of actinic keratosis to squamous cell carcinoma. Dermatol Surg. 2007;33: 1099-1101.
- Dianzani C, Conforti C, Giuffrida R, et al. Current therapies for actinic keratosis. Int J Dermatol. 2020;59:677-684.
- Javor S, Cozzani E, Parodi A. Topical treatment of actinic keratosis with 3.0% diclofenac in 2.5% hyaluronan gel: review of the literature about the cumulative evidence of its efficacy and safety. G Ital Dermatol Venereol. 2016;151:275-280.
- Martin GM, Stockfleth E. Diclofenac sodium 3% gel for the management of actinic keratosis: 10+ years of cumulative evidence of efficacy and safety. J Drugs Dermatol. 2012;11:600-608.
- Arisi M, Guasco Pisani E, et al. Cryotherapy for actinic keratosis: basic principles and literature review. Clin Cosmet Investig Dermatol. 2022;15:357-365.
- Calzavara-Pinton P, Calzavara-Pinton I, Rovati C, et al. Topical pharmacotherapy for actinic keratoses in older adults. Drugs Aging. 2022;39:143-152.
- Beutner C, Forkel S, Kreipe K, et al. Contact allergy to topical diclofenac with systemic tolerance. Contact Dermatitis. 2022;86:41-43.
- Voltaren gel (diclofenac sodium topical gel). Prescribing information. Novartis Consumer Health, Inc; 2009. Accessed May 21, 2025. https:// www.accessdata.fda.gov/drugsatfda_docs/label/2009/022122s006lbl.pdf
- Moreira SA, Liu DJ. Diclofenac systemic bioavailability of a topical 1% diclofenac + 3% menthol combination gel vs. an oral diclofenac tablet in healthy volunteers: a randomized, open-label, crossover study. Int J Clin Pharmacol Ther. 2017;55:368-372.
- Kienzler JL, Gold M, Nollevaux F. Systemic bioavailability of topical diclofenac sodium gel 1% versus oral diclofenac sodium in healthy volunteers. J Clin Pharmacol. 2010;50:50-61.
- Gulin SJ, Chiriac A. Diclofenac-induced allergic contact dermatitis: a series of four patients. Drug Saf Case Rep. 2016;3:15.
- Fecker LF, Stockfleth E, Nindl I, et al. The role of apoptosis in therapy and prophylaxis of epithelial tumours by nonsteroidal antiinflammatory drugs (NSAIDs). Br J Dermatol. 2007;156(Suppl 3):25-33.
- Thomas GJ, Herranz P, Cruz SB, et al. Treatment of actinic keratosis through inhibition of cyclooxygenase-2: potential mechanism of action of diclofenac sodium 3% in hyaluronic acid 2.5. Dermatol Ther. 2019;32:e12800.
- Singer K, Dettmer K, Unger P, et al. Topical diclofenac reprograms metabolism and immune cell infiltration in actinic keratosis. Front Oncol. 2019;9:605.
- Diclofenac (topical). Drug information. UpToDate. https://www-uptodate-com.libraryaccess.elpaso.ttuhsc.edu/contents/diclofenac-topical-drug-information?source=auto_suggest&selectedTitle=1~3---3~4---diclofenac&search=diclofenac%20topical#F8017265
- Afify AA, Hana MR. Comparative evaluation of topical diclofenac sodium versus topical ibuprofen in the treatment of seborrheic keratosis. Dermatol Ther. 2020;33:e14370.
- Yin F, Ma J, Xiao H, et al. Randomized, double-blind, noninferiority study of diclofenac diethylamine 2.32% gel applied twice daily versus diclofenac diethylamine 1.16% gel applied four times daily in patients with acute ankle sprain. BMC Musculoskelet Disord. 2022;23:1125.
- van Herwaarden N, van den Elsen GAH, de Jong ICA, et al. Topical NSAIDs: ineffective or undervalued? [in Dutch]. Ned Tijdschr Geneeskd. 2021;165:D5317.
Actinic Keratosis Treatment With Diclofenac Gel 1%
Actinic Keratosis Treatment With Diclofenac Gel 1%
PRACTICE POINTS
- There are numerous field-directed therapies for actinic keratoses (AKs); however, efficacy and tolerability vary among the available treatments.
- Diclofenac gel 1% is an affordable option that could potentially increase accessibility and decrease cost of field therapy for the treatment of AKs, while maintaining therapeutic efficacy.
Behavioral Health Trainee Satisfaction at the US Department of Veterans Affairs During the COVID-19 Pandemic
Behavioral Health Trainee Satisfaction at the US Department of Veterans Affairs During the COVID-19 Pandemic
The COVID-19 pandemic changed the education and training experiences of health care students and those set to comprise the future workforce. Apart from general training disruptions or delays due to the pandemic, behavioral health trainees such as psychologists and social workers faced limited opportunities to provide in-person services.1-5 Trainees also experienced fewer referrals to mental health services from primary care and more disrupted, no-show, or cancelled appointments.4-6 Behavioral health trainees experienced a limited ability to establish rapport and more difficulty providing effective services because of the limited in-person interaction presented by telehealth.6 The pandemic also resulted in feelings of increased isolation and decreased teamwork.1,7 The virtual or remote setting made it more difficult for trainees to feel as if they were a member of a team or community of behavioral health professionals.1,7
Behavioral health trainees had to adapt to conducting patient visits and educational didactics through virtual platforms.1,3-7 Challenges included access or technological problems with online platforms and a lack of telehealth training use.3,4,6 One study found that while both behavioral health trainees and licensed practitioners reported similar rates of telehealth use for mental health services by early April 2020, trainees had more difficulties implementing telehealth compared with licensed practitioners. This study found that US Department of Veteran Affairs (VA) facilities reported higher use of telehealth in February 2020.5
A mission of the VA is to provide education and training to health care professionals through partnerships with affiliated academic institutions. The VA is the largest education and training supplier for health care professions in the US. As many as 50% of psychologists in the US received some training at the VA.8 Additionally, more graduate-level social work students are trained at the VA than at any other organization.9 The VA is a major contributor to not only its own behavioral health workforce, but that of the entire country.
The VA is also the largest employer of psychologists and social workers in the US.10,11 The VA Office of Academic Affiliations (OAA) oversees health care profession education and training at all VA facilities. In 2012, OAA began the Mental Health Education Expansion program to increase training for behavioral health professionals, including psychologists and social workers. 12 The OAA initiative was aligned with VA training and workforce priorities.8,12 To gauge the effectiveness of VA education and training, OAA encourages VA trainees to complete the Trainee Satisfaction Survey (TSS), which measures trainee satisfaction and the likelihood of a trainee to consider the VA for future employment.
Researchers at the Veterans Emergency Management Evaluation Center sought to understand the impact the COVID-19 pandemic had on behavioral health trainees’ experiences by examining TSS data from before and after the onset of the pandemic. This study expands on prior research among physician residents and fellows which found associations between VA training experiences and the COVID- 19 pandemic. The previous study found declines in trainee satisfaction and a decreased likelihood to consider the VA for future employment.13
It is important to understand the effects the pandemic had on the professional development and wellness for both physician and behavioral health professional trainees. Identifying how the pandemic impacted trainee satisfaction may help improve education programs and mitigate the impact of future public health emergencies. This is particularly important due to the shortage of behavioral health professionals in the VA and the US.12,14
METHODS
This study used TSS data collected from August 2018 to July 2021 from 153 VA facilities. A behavioral health trainee was defined as any psychology or social work trainee who completed 1 rotation at a VA facility. Psychiatric trainees were excluded because as physicians their training programs differ markedly from those for psychology and social work. Excluding psychiatry, psychology and social work comprise the 2 largest mental health care training groups.
This study was reviewed and approved as a quality improvement project by the VA Greater Los Angeles Healthcare System (VAGLAHS) Institutional Review Board, which waived informed consent requirements. The OAA granted access to data using a process open to all VA researchers. At the time of data collection, respondents were assured their anonymity; participation was voluntary.
Measures
Any response provided before February 29, 2020, was defined as the prepandemic period. The pandemic period included any response from April 1, 2020, to July 31, 2021. Responses collected in March 2020 were excluded as it would be unclear from the survey whether the training period occurred before or after the onset of the pandemic.
To measure overall trainee satisfaction with the VA training experience, responses were grouped as satisfied (satisfied/ very satisfied) and dissatisfied (dissatisfied/ very dissatisfied). To measure a trainee’s likelihood to consider the VA for future employment as a result of their training experience, responses were grouped as likely (likely/very likely) and unlikely (unlikely/very unlikely).
Other components of satisfaction were also collected including onboarding, clinical faculty/preceptors, clinical learning environment, physical environment, working environment, and respect felt at work. If a respondent chose very dissatisfied or dissatisfied, they were subsequently asked to specify the reason for their dissatisfaction with an open-ended response. Open-ended responses were not permitted if a respondent indicated a satisfied or very satisfied response.
Statistical Analyses
Stata SE 17 was used for statistical analyses. To test the relationship between the pandemic group and the 2 separate outcome variables, logistic regressions were conducted to measure overall satisfaction and likelihood of future VA employment. Margin commands were used to calculate the difference in the probability of reporting satisfied/very satisfied and likely/very likely for the prepandemic and pandemic groups. The association of the COVID-19 group with each outcome variable was expressed as the difference in the percentage of the outcome between the prepandemic and pandemic groups. Preliminary analyses demonstrated similar effects of the pandemic on psychology and social work trainees; therefore, the groups were combined.
Rapid Coding and Thematic Analyses
Qualitative data were based on open-ended responses from behavioral health trainees when they were asked to specify the cause of dissatisfaction in the aforementioned areas of satisfaction. Methods for qualitative data included rapid coding and thematic content analyses.15,16 Additional general information regarding the qualitative data analyses is described elsewhere.13 A keyword search was completed to identify all open-ended responses related to COVID-19 pandemic causes of dissatisfaction. Keywords included: virus, COVID, corona, pandemic, PPE, N95, mask, social distance, and safety. All open-ended responses were reviewed to ensure keywords were appropriately identifying pandemic-related causes of dissatisfaction and did not overlook other references to the pandemic, and to identify initial themes and corresponding definitions based on survey questions. After review, additional keywords were included in the content analyses that were related to providing mental health services using remote or telehealth options. This included the following keywords: remote, video, VVC (VA Video Connect), and tele. The research team completed a review of the initial themes and definitions and created a final coding construct with definitions before completing an independent coding of all identified pandemic-related responses. Frequency counts of each code were provided to identify which pandemic-related causes of dissatisfaction were mentioned most.
RESULTS
A total of 3950 behavioral health trainees responded to the TSS, including 2715 psychology trainees and 1235 social work trainees who indicated they received training at the VA in academic years 2018/2019, 2019/2020, or 2020/2021. The academic year 2018/2019 was considered in an effort to provide a larger sample of prepandemic trainees.
The percentage of trainees reporting satisfaction with their training decreased across prepandemic to pandemic groups. In the pandemic group, 2166 of 2324 respondents (93.2%) reported satisfaction compared to 1474 of 1555 (94.8%) in the prepandemic trainee group (P = .04; 95% CI, -3.10 to -0.08). There was no association between the pandemic group and behavioral health trainees’ reported willingness to consider the VA for future employment (Table 1). Preliminary analyses demonstrated similar effects of the pandemic on psychology and social work trainees, therefore the groups were combined, and overall effects were reported.
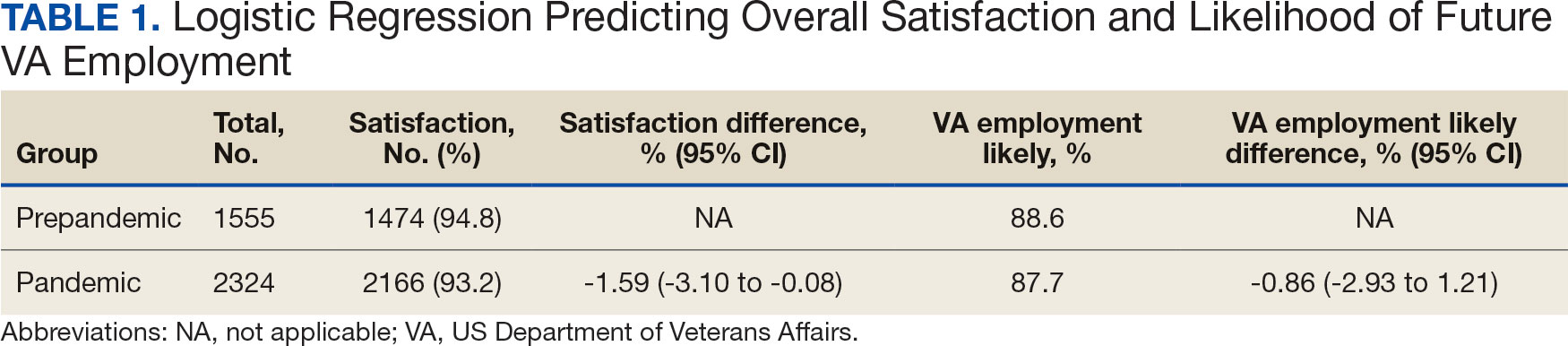
Pandemic-Related Dissatisfaction
Of the 3950 psychology and social work trainees who responded to the survey, 75 (1.9%) indicated dissatisfaction with their VA training experience using pandemic-related keywords. Open-ended responses were generally short (range, 1-32 words; median, 19 words). Qualitative analyses revealed 7 themes (Table 2).
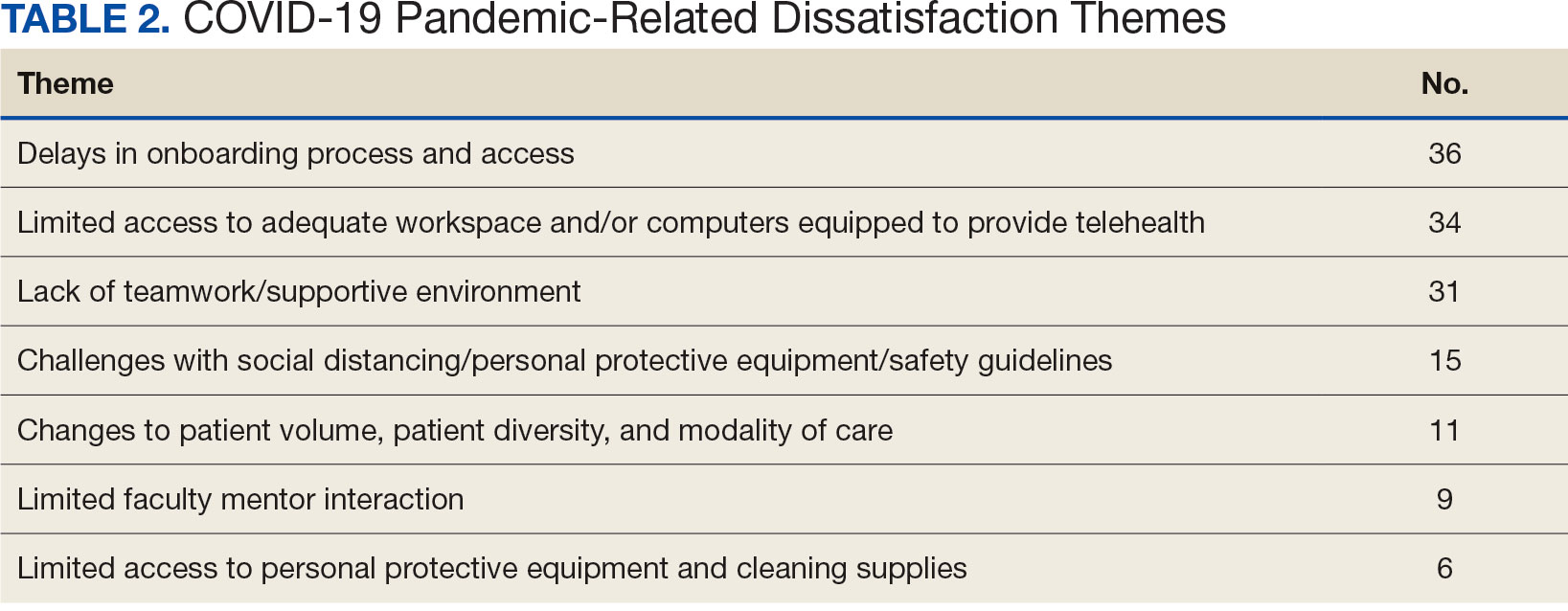
The most frequently identified theme was challenges with onboarding. One respondent indicated the modified onboarding procedures in place due to the pandemic were difficult to understand and resulted in delays. Another frequently mentioned cause of dissatisfaction was limited work or office space and insufficient computer availability. This was often noted to relate to a lack of private space to conduct telehealth visits or computers that were not equipped to provide telehealth. Several respondents also noted technological issues when attempting to use VVC to provide telehealth.
Another common theme was that the pandemic diminished teamwork, generated feelings of isolation, and created unsupportive environments for trainees. For instance, some trainees indicated that COVID-19 decreased the inclusion of trainees as part of the regular staff groups and accordingly resulted in limited networking opportunities. Other causes of dissatisfaction included the pandemic’s impacts on the learning environment, such as decreases in patient volume, decreased diversity of patient cases, and a limited presence of faculty mentors. Several respondents indicated that the pandemic limited their caseloads and indicated that most patients were seen virtually. Open-ended responses from a few respondents indicated their training environments were noncompliant with social distancing, personal protective equipment requirements, or other safety guidelines.
DISCUSSION
This study illustrates the impact of the COVID-19 pandemic on the behavioral health trainee experience, which was expressed through decreased satisfaction with their clinical training at the VA. The narrative data indicated that the observed pandemic-related dissatisfaction was linked specifically to onboarding, a lack of safe and private workspaces and computers, as well as a lack of a supportive work environment.
Although the reported decrease in satisfaction was statistically significant, the effect size was not large. Additionally, while satisfaction did decrease, the trainees’ reported likelihood to consider the VA for future employment was not impacted. This may suggest psychologist and social work trainees’ perseverance and dedication to their chosen profession persisted despite the challenges presented by the pandemic. Furthermore, the qualitative data suggest potential ways to mitigate health care profession trainee challenges that can follow a crisis like the COVID-19 pandemic, although further study is warranted.
While narrative responses with pandemic-related keywords did indicate challenges specific to COVID-19 (ie, limited access to workspaces and/or computers equipped for telehealth), the overall frequency of pandemic-related responses was low. This may indicate these are institutional challenges trainees face independent of the pandemic. These findings warrant longterm attention to the satisfaction of psychology and social worker trainees’ during the pandemic. For example, additional training for the use of telehealth could be provided. One study indicated that < 61% of psychology postdoctoral fellows received telepsychology training during the pandemic, and of those who did receive training, less than half were satisfied with it.3
Similarly, strategies could be developed to ensure a more supportive learning and work environment, and provide additional networking opportunities for trainees, despite social distancing. Education specific to disaster response should be incorporated into behavioral health care professionals’ training, especially because behavioral health care professionals provided major contributions during the pandemic due to reported increases in mental health concerns (eg, anxiety and depression) during the period.7,17,18 As the pandemic progressed, policies and procedures were established or modified to address some of these concerns because they were not necessarily limited to trainees. For example, additional training resources were developed to support the use of various telehealth technologies, virtual resources were used more often for meetings, and supervisors developed more comfort and familiarity with how to manage in a virtual or hybrid environment.
Limitations
Although the TSS data provide a large national sample of behavioral health care trainees, it only includes VA trainees, and therefore may not be completely generalizable across health care. However, because many psychologists and social workers throughout the US train at the VA, and because the VA is the largest employer of practicing psychologists and social workers, understanding the impacts felt at the VA informs institutions nationally.8-11 The TSS has limited demographic data (eg, age, race, ethnicity, and sex), so it is unclear whether the respondent groups before and during the pandemic differed in ways that could relate to outcomes. The data also do not specify exact training dates; however, anecdotal evidence suggests respondents generally complete the survey close to the end of their training.
Additionally, open-ended narrative responses were only asked for replies that indicated dissatisfaction, precluding a more nuanced understanding of potential positive outcomes. Furthermore, the TSS is limited to questions about the trainees’ clinical experiences, but because the pandemic created many stressors, there may have been personal issues that affected their work. It is possible that changes in overall satisfaction may have been rooted in something outside of their clinical experience. Finally, the response rate for the TSS is consistently low both before and during the pandemic and includes a limited number of narrative responses.
CONCLUSIONS
The VA is an important contributor to the education, training, and composition of the behavioral health care workforce. A deeper understanding of the VA trainee experience is important to identify how to improve behavioral health care professional education and training. This is especially true as behavioral health care faces shortages within the VA and nationwide.8,12,19
This study reinforces research that found health care trainees experienced decreased learning opportunities and telehealth-related challenges during the COVID-19 pandemic. 13,20 Despite the observed decline in trainee satisfaction, the lack of a corresponding change in likelihood to seek employment with the VA is encouraging for VA efforts to maintain and grow its behavioral health care workforce and for similar efforts outside VA. This resilience may relate to the substantial prepandemic time invested in their professional development. Future studies should examine long term impacts of the pandemic on trainee’s clinical experience and whether the pipeline of behavioral health care workers declines over time as students that are earlier in their career paths instead chose other professions. Future research should also explore ways to improve professional development and wellness of behavioral health care trainees during disasters (eg, telehealth training, additional networking, and social support).
- Muddle S, Rettie H, Harris O, Lawes A, Robinson R. Trainee life under COVID-19: a systemic case report. J Fam Ther. 2022;44(2):239-249. doi:10.1111/1467-6427.12354
- Valenzuela J, Crosby LE, Harrison RR. Commentary: reflections on the COVID-19 pandemic and health disparities in pediatric psychology. J Pediatr Psychol. 2020;45(8):839- 841. doi:10.1093/jpepsy/jsaa063
- Frye WS, Feldman M, Katzenstein J, Gardner L. Modified training experiences for psychology interns and fellows during COVID-19: use of telepsychology and telesupervision by child and adolescent training programs. J Clin Psychol Med Settings. 2022;29(4):840- 848. doi:10.1007/s10880-021-09839-4
- Perrin PB, Rybarczyk BD, Pierce BS, Jones HA, Shaffer C, Islam L. Rapid telepsychology deployment during the COVID-19 pandemic: a special issue commentary and lessons from primary care psychology training. J Clin Psychol. 2020;76(6):1173-1185. doi:10.1002/jclp.22969
- Reilly SE, Zane KL, McCuddy WT, et al. Mental health practitioners’ immediate practical response during the COVID-19 pandemic: observational questionnaire study. JMIR Ment Health. 2020;7(9):e21237. doi:10.2196/21237
- Sadicario JS, Parlier-Ahmad AB, Brechbiel JK, Islam LZ, Martin CE. Caring for women with substance use disorders through pregnancy and postpartum during the COVID-19 pandemic: lessons learned from psychology trainees in an integrated OBGYN/substance use disorder outpatient treatment program. J Subst Abuse Treat. 2021;122:108200. doi:10.1016/j.jsat.2020.108200
- Schneider NM, Steinberg DM, Garcia AM, et al. Pediatric consultation-liaison psychology: insights and lessons learned during the COVID-19 pandemic. J Clin Psychol Med Settings. 2023;30(1):51-60. doi:10.1007/s10880-022-09887-4
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee to Evaluate the Department of Veterans Affairs Mental Health. Mental Health Workforce and Facilities Infrastructure. In: Evaluation of the Department of Veterans Affairs Mental Health Services. National Academies Press (US); 2018. https://www.ncbi.nlm.nih.gov/books/NBK499512/
- U.S. Department of Veterans Affairs Veterans Health Administration. Career as a VA social worker. Updated March 3, 2025. Accessed May 6, 2025. https://www.socialwork.va.gov/VA_Employment.asp
- United States Senate Committee on Veterans Affairs hearing on “Making the VA the Workplace of Choice for Health Care Providers.” News release. American Psychological Association. April 9, 2008. Accessed April 9, 2025. https:// www.apa.org/news/press/releases/2008/04/testimony
- VA National Professional Social Work Month Planning Committee. The diverse, far-reaching VA social worker profession. March 17, 2023. Accessed April 9, 2025. https://news.va.gov/116804/diverse-far-reaching-social-worker-profession/
- Patel EL, Bates JM, Holguin JK, et al. Program profile: the expansion of associated health training in the VA. Fed Pract. 2021;38(8):374-380. doi:10.12788/fp.0163
- Northcraft H, Bai J, Griffin AR, Hovsepian S, Dobalian A. Association of the COVID-19 pandemic on VA resident and fellow training satisfaction and future VA employment: a mixed methods study. J Grad Med Educ. 2022;14(5):593- 598. doi:10.4300/JGME-D-22-00168.1
- Health Resources and Services Administration. Health workforce shortage areas. Accessed April 9, 2025. https://data.hrsa.gov/topics/health-workforce/shortage-areas
- Gale RC, Wu J, Erhardt T, et al. Comparison of rapid vs in-depth qualitative analytic methods from a process evaluation of academic detailing in the Veterans Health Administration. Implement Sci. 2019;14(1):11. doi:10.1186/s13012-019-0853-y
- Taylor B, Henshall C, Kenyon S, Litchfield I, Greenfield S. Can rapid approaches to qualitative analysis deliver timely, valid findings to clinical leaders? A mixed methods study comparing rapid and thematic analysis. BMJ Open. 2018;8(10):e019993. doi:10.1136/bmjopen-2017-019993
- Kranke D, Der-Martirosian C, Hovsepian S, et al. Social workers being effective in disaster settings. Soc Work Public Health. 2020;35(8):664-668. doi:10.1080/19371918.20 20.1820928
- Kranke D, Gin JL, Der-Martirosian C, Weiss EL, Dobalian A. VA social work leadership and compassion fatigue during the 2017 hurricane season. Soc Work Ment Health. 2020;18:188-199. doi:10.1080/15332985.2019.1700873
- Health Resources and Services Administration. Workforce projections. Accessed April 9, 2025. https://data.hrsa.gov/topics/health-workforce/workforce-projections
- Der-Martirosian C, Wyte-Lake T, Balut M, et al. Implementation of telehealth services at the US Department of Veterans Affairs during the COVID-19 pandemic: mixed methods study. JMIR Form Res. 2021;5(9):e29429. doi:10.2196/29429
The COVID-19 pandemic changed the education and training experiences of health care students and those set to comprise the future workforce. Apart from general training disruptions or delays due to the pandemic, behavioral health trainees such as psychologists and social workers faced limited opportunities to provide in-person services.1-5 Trainees also experienced fewer referrals to mental health services from primary care and more disrupted, no-show, or cancelled appointments.4-6 Behavioral health trainees experienced a limited ability to establish rapport and more difficulty providing effective services because of the limited in-person interaction presented by telehealth.6 The pandemic also resulted in feelings of increased isolation and decreased teamwork.1,7 The virtual or remote setting made it more difficult for trainees to feel as if they were a member of a team or community of behavioral health professionals.1,7
Behavioral health trainees had to adapt to conducting patient visits and educational didactics through virtual platforms.1,3-7 Challenges included access or technological problems with online platforms and a lack of telehealth training use.3,4,6 One study found that while both behavioral health trainees and licensed practitioners reported similar rates of telehealth use for mental health services by early April 2020, trainees had more difficulties implementing telehealth compared with licensed practitioners. This study found that US Department of Veteran Affairs (VA) facilities reported higher use of telehealth in February 2020.5
A mission of the VA is to provide education and training to health care professionals through partnerships with affiliated academic institutions. The VA is the largest education and training supplier for health care professions in the US. As many as 50% of psychologists in the US received some training at the VA.8 Additionally, more graduate-level social work students are trained at the VA than at any other organization.9 The VA is a major contributor to not only its own behavioral health workforce, but that of the entire country.
The VA is also the largest employer of psychologists and social workers in the US.10,11 The VA Office of Academic Affiliations (OAA) oversees health care profession education and training at all VA facilities. In 2012, OAA began the Mental Health Education Expansion program to increase training for behavioral health professionals, including psychologists and social workers. 12 The OAA initiative was aligned with VA training and workforce priorities.8,12 To gauge the effectiveness of VA education and training, OAA encourages VA trainees to complete the Trainee Satisfaction Survey (TSS), which measures trainee satisfaction and the likelihood of a trainee to consider the VA for future employment.
Researchers at the Veterans Emergency Management Evaluation Center sought to understand the impact the COVID-19 pandemic had on behavioral health trainees’ experiences by examining TSS data from before and after the onset of the pandemic. This study expands on prior research among physician residents and fellows which found associations between VA training experiences and the COVID- 19 pandemic. The previous study found declines in trainee satisfaction and a decreased likelihood to consider the VA for future employment.13
It is important to understand the effects the pandemic had on the professional development and wellness for both physician and behavioral health professional trainees. Identifying how the pandemic impacted trainee satisfaction may help improve education programs and mitigate the impact of future public health emergencies. This is particularly important due to the shortage of behavioral health professionals in the VA and the US.12,14
METHODS
This study used TSS data collected from August 2018 to July 2021 from 153 VA facilities. A behavioral health trainee was defined as any psychology or social work trainee who completed 1 rotation at a VA facility. Psychiatric trainees were excluded because as physicians their training programs differ markedly from those for psychology and social work. Excluding psychiatry, psychology and social work comprise the 2 largest mental health care training groups.
This study was reviewed and approved as a quality improvement project by the VA Greater Los Angeles Healthcare System (VAGLAHS) Institutional Review Board, which waived informed consent requirements. The OAA granted access to data using a process open to all VA researchers. At the time of data collection, respondents were assured their anonymity; participation was voluntary.
Measures
Any response provided before February 29, 2020, was defined as the prepandemic period. The pandemic period included any response from April 1, 2020, to July 31, 2021. Responses collected in March 2020 were excluded as it would be unclear from the survey whether the training period occurred before or after the onset of the pandemic.
To measure overall trainee satisfaction with the VA training experience, responses were grouped as satisfied (satisfied/ very satisfied) and dissatisfied (dissatisfied/ very dissatisfied). To measure a trainee’s likelihood to consider the VA for future employment as a result of their training experience, responses were grouped as likely (likely/very likely) and unlikely (unlikely/very unlikely).
Other components of satisfaction were also collected including onboarding, clinical faculty/preceptors, clinical learning environment, physical environment, working environment, and respect felt at work. If a respondent chose very dissatisfied or dissatisfied, they were subsequently asked to specify the reason for their dissatisfaction with an open-ended response. Open-ended responses were not permitted if a respondent indicated a satisfied or very satisfied response.
Statistical Analyses
Stata SE 17 was used for statistical analyses. To test the relationship between the pandemic group and the 2 separate outcome variables, logistic regressions were conducted to measure overall satisfaction and likelihood of future VA employment. Margin commands were used to calculate the difference in the probability of reporting satisfied/very satisfied and likely/very likely for the prepandemic and pandemic groups. The association of the COVID-19 group with each outcome variable was expressed as the difference in the percentage of the outcome between the prepandemic and pandemic groups. Preliminary analyses demonstrated similar effects of the pandemic on psychology and social work trainees; therefore, the groups were combined.
Rapid Coding and Thematic Analyses
Qualitative data were based on open-ended responses from behavioral health trainees when they were asked to specify the cause of dissatisfaction in the aforementioned areas of satisfaction. Methods for qualitative data included rapid coding and thematic content analyses.15,16 Additional general information regarding the qualitative data analyses is described elsewhere.13 A keyword search was completed to identify all open-ended responses related to COVID-19 pandemic causes of dissatisfaction. Keywords included: virus, COVID, corona, pandemic, PPE, N95, mask, social distance, and safety. All open-ended responses were reviewed to ensure keywords were appropriately identifying pandemic-related causes of dissatisfaction and did not overlook other references to the pandemic, and to identify initial themes and corresponding definitions based on survey questions. After review, additional keywords were included in the content analyses that were related to providing mental health services using remote or telehealth options. This included the following keywords: remote, video, VVC (VA Video Connect), and tele. The research team completed a review of the initial themes and definitions and created a final coding construct with definitions before completing an independent coding of all identified pandemic-related responses. Frequency counts of each code were provided to identify which pandemic-related causes of dissatisfaction were mentioned most.
RESULTS
A total of 3950 behavioral health trainees responded to the TSS, including 2715 psychology trainees and 1235 social work trainees who indicated they received training at the VA in academic years 2018/2019, 2019/2020, or 2020/2021. The academic year 2018/2019 was considered in an effort to provide a larger sample of prepandemic trainees.
The percentage of trainees reporting satisfaction with their training decreased across prepandemic to pandemic groups. In the pandemic group, 2166 of 2324 respondents (93.2%) reported satisfaction compared to 1474 of 1555 (94.8%) in the prepandemic trainee group (P = .04; 95% CI, -3.10 to -0.08). There was no association between the pandemic group and behavioral health trainees’ reported willingness to consider the VA for future employment (Table 1). Preliminary analyses demonstrated similar effects of the pandemic on psychology and social work trainees, therefore the groups were combined, and overall effects were reported.

Pandemic-Related Dissatisfaction
Of the 3950 psychology and social work trainees who responded to the survey, 75 (1.9%) indicated dissatisfaction with their VA training experience using pandemic-related keywords. Open-ended responses were generally short (range, 1-32 words; median, 19 words). Qualitative analyses revealed 7 themes (Table 2).

The most frequently identified theme was challenges with onboarding. One respondent indicated the modified onboarding procedures in place due to the pandemic were difficult to understand and resulted in delays. Another frequently mentioned cause of dissatisfaction was limited work or office space and insufficient computer availability. This was often noted to relate to a lack of private space to conduct telehealth visits or computers that were not equipped to provide telehealth. Several respondents also noted technological issues when attempting to use VVC to provide telehealth.
Another common theme was that the pandemic diminished teamwork, generated feelings of isolation, and created unsupportive environments for trainees. For instance, some trainees indicated that COVID-19 decreased the inclusion of trainees as part of the regular staff groups and accordingly resulted in limited networking opportunities. Other causes of dissatisfaction included the pandemic’s impacts on the learning environment, such as decreases in patient volume, decreased diversity of patient cases, and a limited presence of faculty mentors. Several respondents indicated that the pandemic limited their caseloads and indicated that most patients were seen virtually. Open-ended responses from a few respondents indicated their training environments were noncompliant with social distancing, personal protective equipment requirements, or other safety guidelines.
DISCUSSION
This study illustrates the impact of the COVID-19 pandemic on the behavioral health trainee experience, which was expressed through decreased satisfaction with their clinical training at the VA. The narrative data indicated that the observed pandemic-related dissatisfaction was linked specifically to onboarding, a lack of safe and private workspaces and computers, as well as a lack of a supportive work environment.
Although the reported decrease in satisfaction was statistically significant, the effect size was not large. Additionally, while satisfaction did decrease, the trainees’ reported likelihood to consider the VA for future employment was not impacted. This may suggest psychologist and social work trainees’ perseverance and dedication to their chosen profession persisted despite the challenges presented by the pandemic. Furthermore, the qualitative data suggest potential ways to mitigate health care profession trainee challenges that can follow a crisis like the COVID-19 pandemic, although further study is warranted.
While narrative responses with pandemic-related keywords did indicate challenges specific to COVID-19 (ie, limited access to workspaces and/or computers equipped for telehealth), the overall frequency of pandemic-related responses was low. This may indicate these are institutional challenges trainees face independent of the pandemic. These findings warrant longterm attention to the satisfaction of psychology and social worker trainees’ during the pandemic. For example, additional training for the use of telehealth could be provided. One study indicated that < 61% of psychology postdoctoral fellows received telepsychology training during the pandemic, and of those who did receive training, less than half were satisfied with it.3
Similarly, strategies could be developed to ensure a more supportive learning and work environment, and provide additional networking opportunities for trainees, despite social distancing. Education specific to disaster response should be incorporated into behavioral health care professionals’ training, especially because behavioral health care professionals provided major contributions during the pandemic due to reported increases in mental health concerns (eg, anxiety and depression) during the period.7,17,18 As the pandemic progressed, policies and procedures were established or modified to address some of these concerns because they were not necessarily limited to trainees. For example, additional training resources were developed to support the use of various telehealth technologies, virtual resources were used more often for meetings, and supervisors developed more comfort and familiarity with how to manage in a virtual or hybrid environment.
Limitations
Although the TSS data provide a large national sample of behavioral health care trainees, it only includes VA trainees, and therefore may not be completely generalizable across health care. However, because many psychologists and social workers throughout the US train at the VA, and because the VA is the largest employer of practicing psychologists and social workers, understanding the impacts felt at the VA informs institutions nationally.8-11 The TSS has limited demographic data (eg, age, race, ethnicity, and sex), so it is unclear whether the respondent groups before and during the pandemic differed in ways that could relate to outcomes. The data also do not specify exact training dates; however, anecdotal evidence suggests respondents generally complete the survey close to the end of their training.
Additionally, open-ended narrative responses were only asked for replies that indicated dissatisfaction, precluding a more nuanced understanding of potential positive outcomes. Furthermore, the TSS is limited to questions about the trainees’ clinical experiences, but because the pandemic created many stressors, there may have been personal issues that affected their work. It is possible that changes in overall satisfaction may have been rooted in something outside of their clinical experience. Finally, the response rate for the TSS is consistently low both before and during the pandemic and includes a limited number of narrative responses.
CONCLUSIONS
The VA is an important contributor to the education, training, and composition of the behavioral health care workforce. A deeper understanding of the VA trainee experience is important to identify how to improve behavioral health care professional education and training. This is especially true as behavioral health care faces shortages within the VA and nationwide.8,12,19
This study reinforces research that found health care trainees experienced decreased learning opportunities and telehealth-related challenges during the COVID-19 pandemic. 13,20 Despite the observed decline in trainee satisfaction, the lack of a corresponding change in likelihood to seek employment with the VA is encouraging for VA efforts to maintain and grow its behavioral health care workforce and for similar efforts outside VA. This resilience may relate to the substantial prepandemic time invested in their professional development. Future studies should examine long term impacts of the pandemic on trainee’s clinical experience and whether the pipeline of behavioral health care workers declines over time as students that are earlier in their career paths instead chose other professions. Future research should also explore ways to improve professional development and wellness of behavioral health care trainees during disasters (eg, telehealth training, additional networking, and social support).
The COVID-19 pandemic changed the education and training experiences of health care students and those set to comprise the future workforce. Apart from general training disruptions or delays due to the pandemic, behavioral health trainees such as psychologists and social workers faced limited opportunities to provide in-person services.1-5 Trainees also experienced fewer referrals to mental health services from primary care and more disrupted, no-show, or cancelled appointments.4-6 Behavioral health trainees experienced a limited ability to establish rapport and more difficulty providing effective services because of the limited in-person interaction presented by telehealth.6 The pandemic also resulted in feelings of increased isolation and decreased teamwork.1,7 The virtual or remote setting made it more difficult for trainees to feel as if they were a member of a team or community of behavioral health professionals.1,7
Behavioral health trainees had to adapt to conducting patient visits and educational didactics through virtual platforms.1,3-7 Challenges included access or technological problems with online platforms and a lack of telehealth training use.3,4,6 One study found that while both behavioral health trainees and licensed practitioners reported similar rates of telehealth use for mental health services by early April 2020, trainees had more difficulties implementing telehealth compared with licensed practitioners. This study found that US Department of Veteran Affairs (VA) facilities reported higher use of telehealth in February 2020.5
A mission of the VA is to provide education and training to health care professionals through partnerships with affiliated academic institutions. The VA is the largest education and training supplier for health care professions in the US. As many as 50% of psychologists in the US received some training at the VA.8 Additionally, more graduate-level social work students are trained at the VA than at any other organization.9 The VA is a major contributor to not only its own behavioral health workforce, but that of the entire country.
The VA is also the largest employer of psychologists and social workers in the US.10,11 The VA Office of Academic Affiliations (OAA) oversees health care profession education and training at all VA facilities. In 2012, OAA began the Mental Health Education Expansion program to increase training for behavioral health professionals, including psychologists and social workers. 12 The OAA initiative was aligned with VA training and workforce priorities.8,12 To gauge the effectiveness of VA education and training, OAA encourages VA trainees to complete the Trainee Satisfaction Survey (TSS), which measures trainee satisfaction and the likelihood of a trainee to consider the VA for future employment.
Researchers at the Veterans Emergency Management Evaluation Center sought to understand the impact the COVID-19 pandemic had on behavioral health trainees’ experiences by examining TSS data from before and after the onset of the pandemic. This study expands on prior research among physician residents and fellows which found associations between VA training experiences and the COVID- 19 pandemic. The previous study found declines in trainee satisfaction and a decreased likelihood to consider the VA for future employment.13
It is important to understand the effects the pandemic had on the professional development and wellness for both physician and behavioral health professional trainees. Identifying how the pandemic impacted trainee satisfaction may help improve education programs and mitigate the impact of future public health emergencies. This is particularly important due to the shortage of behavioral health professionals in the VA and the US.12,14
METHODS
This study used TSS data collected from August 2018 to July 2021 from 153 VA facilities. A behavioral health trainee was defined as any psychology or social work trainee who completed 1 rotation at a VA facility. Psychiatric trainees were excluded because as physicians their training programs differ markedly from those for psychology and social work. Excluding psychiatry, psychology and social work comprise the 2 largest mental health care training groups.
This study was reviewed and approved as a quality improvement project by the VA Greater Los Angeles Healthcare System (VAGLAHS) Institutional Review Board, which waived informed consent requirements. The OAA granted access to data using a process open to all VA researchers. At the time of data collection, respondents were assured their anonymity; participation was voluntary.
Measures
Any response provided before February 29, 2020, was defined as the prepandemic period. The pandemic period included any response from April 1, 2020, to July 31, 2021. Responses collected in March 2020 were excluded as it would be unclear from the survey whether the training period occurred before or after the onset of the pandemic.
To measure overall trainee satisfaction with the VA training experience, responses were grouped as satisfied (satisfied/ very satisfied) and dissatisfied (dissatisfied/ very dissatisfied). To measure a trainee’s likelihood to consider the VA for future employment as a result of their training experience, responses were grouped as likely (likely/very likely) and unlikely (unlikely/very unlikely).
Other components of satisfaction were also collected including onboarding, clinical faculty/preceptors, clinical learning environment, physical environment, working environment, and respect felt at work. If a respondent chose very dissatisfied or dissatisfied, they were subsequently asked to specify the reason for their dissatisfaction with an open-ended response. Open-ended responses were not permitted if a respondent indicated a satisfied or very satisfied response.
Statistical Analyses
Stata SE 17 was used for statistical analyses. To test the relationship between the pandemic group and the 2 separate outcome variables, logistic regressions were conducted to measure overall satisfaction and likelihood of future VA employment. Margin commands were used to calculate the difference in the probability of reporting satisfied/very satisfied and likely/very likely for the prepandemic and pandemic groups. The association of the COVID-19 group with each outcome variable was expressed as the difference in the percentage of the outcome between the prepandemic and pandemic groups. Preliminary analyses demonstrated similar effects of the pandemic on psychology and social work trainees; therefore, the groups were combined.
Rapid Coding and Thematic Analyses
Qualitative data were based on open-ended responses from behavioral health trainees when they were asked to specify the cause of dissatisfaction in the aforementioned areas of satisfaction. Methods for qualitative data included rapid coding and thematic content analyses.15,16 Additional general information regarding the qualitative data analyses is described elsewhere.13 A keyword search was completed to identify all open-ended responses related to COVID-19 pandemic causes of dissatisfaction. Keywords included: virus, COVID, corona, pandemic, PPE, N95, mask, social distance, and safety. All open-ended responses were reviewed to ensure keywords were appropriately identifying pandemic-related causes of dissatisfaction and did not overlook other references to the pandemic, and to identify initial themes and corresponding definitions based on survey questions. After review, additional keywords were included in the content analyses that were related to providing mental health services using remote or telehealth options. This included the following keywords: remote, video, VVC (VA Video Connect), and tele. The research team completed a review of the initial themes and definitions and created a final coding construct with definitions before completing an independent coding of all identified pandemic-related responses. Frequency counts of each code were provided to identify which pandemic-related causes of dissatisfaction were mentioned most.
RESULTS
A total of 3950 behavioral health trainees responded to the TSS, including 2715 psychology trainees and 1235 social work trainees who indicated they received training at the VA in academic years 2018/2019, 2019/2020, or 2020/2021. The academic year 2018/2019 was considered in an effort to provide a larger sample of prepandemic trainees.
The percentage of trainees reporting satisfaction with their training decreased across prepandemic to pandemic groups. In the pandemic group, 2166 of 2324 respondents (93.2%) reported satisfaction compared to 1474 of 1555 (94.8%) in the prepandemic trainee group (P = .04; 95% CI, -3.10 to -0.08). There was no association between the pandemic group and behavioral health trainees’ reported willingness to consider the VA for future employment (Table 1). Preliminary analyses demonstrated similar effects of the pandemic on psychology and social work trainees, therefore the groups were combined, and overall effects were reported.

Pandemic-Related Dissatisfaction
Of the 3950 psychology and social work trainees who responded to the survey, 75 (1.9%) indicated dissatisfaction with their VA training experience using pandemic-related keywords. Open-ended responses were generally short (range, 1-32 words; median, 19 words). Qualitative analyses revealed 7 themes (Table 2).

The most frequently identified theme was challenges with onboarding. One respondent indicated the modified onboarding procedures in place due to the pandemic were difficult to understand and resulted in delays. Another frequently mentioned cause of dissatisfaction was limited work or office space and insufficient computer availability. This was often noted to relate to a lack of private space to conduct telehealth visits or computers that were not equipped to provide telehealth. Several respondents also noted technological issues when attempting to use VVC to provide telehealth.
Another common theme was that the pandemic diminished teamwork, generated feelings of isolation, and created unsupportive environments for trainees. For instance, some trainees indicated that COVID-19 decreased the inclusion of trainees as part of the regular staff groups and accordingly resulted in limited networking opportunities. Other causes of dissatisfaction included the pandemic’s impacts on the learning environment, such as decreases in patient volume, decreased diversity of patient cases, and a limited presence of faculty mentors. Several respondents indicated that the pandemic limited their caseloads and indicated that most patients were seen virtually. Open-ended responses from a few respondents indicated their training environments were noncompliant with social distancing, personal protective equipment requirements, or other safety guidelines.
DISCUSSION
This study illustrates the impact of the COVID-19 pandemic on the behavioral health trainee experience, which was expressed through decreased satisfaction with their clinical training at the VA. The narrative data indicated that the observed pandemic-related dissatisfaction was linked specifically to onboarding, a lack of safe and private workspaces and computers, as well as a lack of a supportive work environment.
Although the reported decrease in satisfaction was statistically significant, the effect size was not large. Additionally, while satisfaction did decrease, the trainees’ reported likelihood to consider the VA for future employment was not impacted. This may suggest psychologist and social work trainees’ perseverance and dedication to their chosen profession persisted despite the challenges presented by the pandemic. Furthermore, the qualitative data suggest potential ways to mitigate health care profession trainee challenges that can follow a crisis like the COVID-19 pandemic, although further study is warranted.
While narrative responses with pandemic-related keywords did indicate challenges specific to COVID-19 (ie, limited access to workspaces and/or computers equipped for telehealth), the overall frequency of pandemic-related responses was low. This may indicate these are institutional challenges trainees face independent of the pandemic. These findings warrant longterm attention to the satisfaction of psychology and social worker trainees’ during the pandemic. For example, additional training for the use of telehealth could be provided. One study indicated that < 61% of psychology postdoctoral fellows received telepsychology training during the pandemic, and of those who did receive training, less than half were satisfied with it.3
Similarly, strategies could be developed to ensure a more supportive learning and work environment, and provide additional networking opportunities for trainees, despite social distancing. Education specific to disaster response should be incorporated into behavioral health care professionals’ training, especially because behavioral health care professionals provided major contributions during the pandemic due to reported increases in mental health concerns (eg, anxiety and depression) during the period.7,17,18 As the pandemic progressed, policies and procedures were established or modified to address some of these concerns because they were not necessarily limited to trainees. For example, additional training resources were developed to support the use of various telehealth technologies, virtual resources were used more often for meetings, and supervisors developed more comfort and familiarity with how to manage in a virtual or hybrid environment.
Limitations
Although the TSS data provide a large national sample of behavioral health care trainees, it only includes VA trainees, and therefore may not be completely generalizable across health care. However, because many psychologists and social workers throughout the US train at the VA, and because the VA is the largest employer of practicing psychologists and social workers, understanding the impacts felt at the VA informs institutions nationally.8-11 The TSS has limited demographic data (eg, age, race, ethnicity, and sex), so it is unclear whether the respondent groups before and during the pandemic differed in ways that could relate to outcomes. The data also do not specify exact training dates; however, anecdotal evidence suggests respondents generally complete the survey close to the end of their training.
Additionally, open-ended narrative responses were only asked for replies that indicated dissatisfaction, precluding a more nuanced understanding of potential positive outcomes. Furthermore, the TSS is limited to questions about the trainees’ clinical experiences, but because the pandemic created many stressors, there may have been personal issues that affected their work. It is possible that changes in overall satisfaction may have been rooted in something outside of their clinical experience. Finally, the response rate for the TSS is consistently low both before and during the pandemic and includes a limited number of narrative responses.
CONCLUSIONS
The VA is an important contributor to the education, training, and composition of the behavioral health care workforce. A deeper understanding of the VA trainee experience is important to identify how to improve behavioral health care professional education and training. This is especially true as behavioral health care faces shortages within the VA and nationwide.8,12,19
This study reinforces research that found health care trainees experienced decreased learning opportunities and telehealth-related challenges during the COVID-19 pandemic. 13,20 Despite the observed decline in trainee satisfaction, the lack of a corresponding change in likelihood to seek employment with the VA is encouraging for VA efforts to maintain and grow its behavioral health care workforce and for similar efforts outside VA. This resilience may relate to the substantial prepandemic time invested in their professional development. Future studies should examine long term impacts of the pandemic on trainee’s clinical experience and whether the pipeline of behavioral health care workers declines over time as students that are earlier in their career paths instead chose other professions. Future research should also explore ways to improve professional development and wellness of behavioral health care trainees during disasters (eg, telehealth training, additional networking, and social support).
- Muddle S, Rettie H, Harris O, Lawes A, Robinson R. Trainee life under COVID-19: a systemic case report. J Fam Ther. 2022;44(2):239-249. doi:10.1111/1467-6427.12354
- Valenzuela J, Crosby LE, Harrison RR. Commentary: reflections on the COVID-19 pandemic and health disparities in pediatric psychology. J Pediatr Psychol. 2020;45(8):839- 841. doi:10.1093/jpepsy/jsaa063
- Frye WS, Feldman M, Katzenstein J, Gardner L. Modified training experiences for psychology interns and fellows during COVID-19: use of telepsychology and telesupervision by child and adolescent training programs. J Clin Psychol Med Settings. 2022;29(4):840- 848. doi:10.1007/s10880-021-09839-4
- Perrin PB, Rybarczyk BD, Pierce BS, Jones HA, Shaffer C, Islam L. Rapid telepsychology deployment during the COVID-19 pandemic: a special issue commentary and lessons from primary care psychology training. J Clin Psychol. 2020;76(6):1173-1185. doi:10.1002/jclp.22969
- Reilly SE, Zane KL, McCuddy WT, et al. Mental health practitioners’ immediate practical response during the COVID-19 pandemic: observational questionnaire study. JMIR Ment Health. 2020;7(9):e21237. doi:10.2196/21237
- Sadicario JS, Parlier-Ahmad AB, Brechbiel JK, Islam LZ, Martin CE. Caring for women with substance use disorders through pregnancy and postpartum during the COVID-19 pandemic: lessons learned from psychology trainees in an integrated OBGYN/substance use disorder outpatient treatment program. J Subst Abuse Treat. 2021;122:108200. doi:10.1016/j.jsat.2020.108200
- Schneider NM, Steinberg DM, Garcia AM, et al. Pediatric consultation-liaison psychology: insights and lessons learned during the COVID-19 pandemic. J Clin Psychol Med Settings. 2023;30(1):51-60. doi:10.1007/s10880-022-09887-4
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee to Evaluate the Department of Veterans Affairs Mental Health. Mental Health Workforce and Facilities Infrastructure. In: Evaluation of the Department of Veterans Affairs Mental Health Services. National Academies Press (US); 2018. https://www.ncbi.nlm.nih.gov/books/NBK499512/
- U.S. Department of Veterans Affairs Veterans Health Administration. Career as a VA social worker. Updated March 3, 2025. Accessed May 6, 2025. https://www.socialwork.va.gov/VA_Employment.asp
- United States Senate Committee on Veterans Affairs hearing on “Making the VA the Workplace of Choice for Health Care Providers.” News release. American Psychological Association. April 9, 2008. Accessed April 9, 2025. https:// www.apa.org/news/press/releases/2008/04/testimony
- VA National Professional Social Work Month Planning Committee. The diverse, far-reaching VA social worker profession. March 17, 2023. Accessed April 9, 2025. https://news.va.gov/116804/diverse-far-reaching-social-worker-profession/
- Patel EL, Bates JM, Holguin JK, et al. Program profile: the expansion of associated health training in the VA. Fed Pract. 2021;38(8):374-380. doi:10.12788/fp.0163
- Northcraft H, Bai J, Griffin AR, Hovsepian S, Dobalian A. Association of the COVID-19 pandemic on VA resident and fellow training satisfaction and future VA employment: a mixed methods study. J Grad Med Educ. 2022;14(5):593- 598. doi:10.4300/JGME-D-22-00168.1
- Health Resources and Services Administration. Health workforce shortage areas. Accessed April 9, 2025. https://data.hrsa.gov/topics/health-workforce/shortage-areas
- Gale RC, Wu J, Erhardt T, et al. Comparison of rapid vs in-depth qualitative analytic methods from a process evaluation of academic detailing in the Veterans Health Administration. Implement Sci. 2019;14(1):11. doi:10.1186/s13012-019-0853-y
- Taylor B, Henshall C, Kenyon S, Litchfield I, Greenfield S. Can rapid approaches to qualitative analysis deliver timely, valid findings to clinical leaders? A mixed methods study comparing rapid and thematic analysis. BMJ Open. 2018;8(10):e019993. doi:10.1136/bmjopen-2017-019993
- Kranke D, Der-Martirosian C, Hovsepian S, et al. Social workers being effective in disaster settings. Soc Work Public Health. 2020;35(8):664-668. doi:10.1080/19371918.20 20.1820928
- Kranke D, Gin JL, Der-Martirosian C, Weiss EL, Dobalian A. VA social work leadership and compassion fatigue during the 2017 hurricane season. Soc Work Ment Health. 2020;18:188-199. doi:10.1080/15332985.2019.1700873
- Health Resources and Services Administration. Workforce projections. Accessed April 9, 2025. https://data.hrsa.gov/topics/health-workforce/workforce-projections
- Der-Martirosian C, Wyte-Lake T, Balut M, et al. Implementation of telehealth services at the US Department of Veterans Affairs during the COVID-19 pandemic: mixed methods study. JMIR Form Res. 2021;5(9):e29429. doi:10.2196/29429
- Muddle S, Rettie H, Harris O, Lawes A, Robinson R. Trainee life under COVID-19: a systemic case report. J Fam Ther. 2022;44(2):239-249. doi:10.1111/1467-6427.12354
- Valenzuela J, Crosby LE, Harrison RR. Commentary: reflections on the COVID-19 pandemic and health disparities in pediatric psychology. J Pediatr Psychol. 2020;45(8):839- 841. doi:10.1093/jpepsy/jsaa063
- Frye WS, Feldman M, Katzenstein J, Gardner L. Modified training experiences for psychology interns and fellows during COVID-19: use of telepsychology and telesupervision by child and adolescent training programs. J Clin Psychol Med Settings. 2022;29(4):840- 848. doi:10.1007/s10880-021-09839-4
- Perrin PB, Rybarczyk BD, Pierce BS, Jones HA, Shaffer C, Islam L. Rapid telepsychology deployment during the COVID-19 pandemic: a special issue commentary and lessons from primary care psychology training. J Clin Psychol. 2020;76(6):1173-1185. doi:10.1002/jclp.22969
- Reilly SE, Zane KL, McCuddy WT, et al. Mental health practitioners’ immediate practical response during the COVID-19 pandemic: observational questionnaire study. JMIR Ment Health. 2020;7(9):e21237. doi:10.2196/21237
- Sadicario JS, Parlier-Ahmad AB, Brechbiel JK, Islam LZ, Martin CE. Caring for women with substance use disorders through pregnancy and postpartum during the COVID-19 pandemic: lessons learned from psychology trainees in an integrated OBGYN/substance use disorder outpatient treatment program. J Subst Abuse Treat. 2021;122:108200. doi:10.1016/j.jsat.2020.108200
- Schneider NM, Steinberg DM, Garcia AM, et al. Pediatric consultation-liaison psychology: insights and lessons learned during the COVID-19 pandemic. J Clin Psychol Med Settings. 2023;30(1):51-60. doi:10.1007/s10880-022-09887-4
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; Committee to Evaluate the Department of Veterans Affairs Mental Health. Mental Health Workforce and Facilities Infrastructure. In: Evaluation of the Department of Veterans Affairs Mental Health Services. National Academies Press (US); 2018. https://www.ncbi.nlm.nih.gov/books/NBK499512/
- U.S. Department of Veterans Affairs Veterans Health Administration. Career as a VA social worker. Updated March 3, 2025. Accessed May 6, 2025. https://www.socialwork.va.gov/VA_Employment.asp
- United States Senate Committee on Veterans Affairs hearing on “Making the VA the Workplace of Choice for Health Care Providers.” News release. American Psychological Association. April 9, 2008. Accessed April 9, 2025. https:// www.apa.org/news/press/releases/2008/04/testimony
- VA National Professional Social Work Month Planning Committee. The diverse, far-reaching VA social worker profession. March 17, 2023. Accessed April 9, 2025. https://news.va.gov/116804/diverse-far-reaching-social-worker-profession/
- Patel EL, Bates JM, Holguin JK, et al. Program profile: the expansion of associated health training in the VA. Fed Pract. 2021;38(8):374-380. doi:10.12788/fp.0163
- Northcraft H, Bai J, Griffin AR, Hovsepian S, Dobalian A. Association of the COVID-19 pandemic on VA resident and fellow training satisfaction and future VA employment: a mixed methods study. J Grad Med Educ. 2022;14(5):593- 598. doi:10.4300/JGME-D-22-00168.1
- Health Resources and Services Administration. Health workforce shortage areas. Accessed April 9, 2025. https://data.hrsa.gov/topics/health-workforce/shortage-areas
- Gale RC, Wu J, Erhardt T, et al. Comparison of rapid vs in-depth qualitative analytic methods from a process evaluation of academic detailing in the Veterans Health Administration. Implement Sci. 2019;14(1):11. doi:10.1186/s13012-019-0853-y
- Taylor B, Henshall C, Kenyon S, Litchfield I, Greenfield S. Can rapid approaches to qualitative analysis deliver timely, valid findings to clinical leaders? A mixed methods study comparing rapid and thematic analysis. BMJ Open. 2018;8(10):e019993. doi:10.1136/bmjopen-2017-019993
- Kranke D, Der-Martirosian C, Hovsepian S, et al. Social workers being effective in disaster settings. Soc Work Public Health. 2020;35(8):664-668. doi:10.1080/19371918.20 20.1820928
- Kranke D, Gin JL, Der-Martirosian C, Weiss EL, Dobalian A. VA social work leadership and compassion fatigue during the 2017 hurricane season. Soc Work Ment Health. 2020;18:188-199. doi:10.1080/15332985.2019.1700873
- Health Resources and Services Administration. Workforce projections. Accessed April 9, 2025. https://data.hrsa.gov/topics/health-workforce/workforce-projections
- Der-Martirosian C, Wyte-Lake T, Balut M, et al. Implementation of telehealth services at the US Department of Veterans Affairs during the COVID-19 pandemic: mixed methods study. JMIR Form Res. 2021;5(9):e29429. doi:10.2196/29429
Behavioral Health Trainee Satisfaction at the US Department of Veterans Affairs During the COVID-19 Pandemic
Behavioral Health Trainee Satisfaction at the US Department of Veterans Affairs During the COVID-19 Pandemic
Impact of Initial Specimen Diversion Technique on Blood Culture Contamination Rates
Impact of Initial Specimen Diversion Technique on Blood Culture Contamination Rates
Blood cultures provide crucial evidence for diagnostic medicine, specifically aimed at identifying the presence of microbial infections in the bloodstream. Blood culturing is instrumental in diagnosing conditions such as sepsis, bacteremia, or fungemia, where the identification of the causative agent is necessary for targeted and effective treatment.1
The process involves aseptically drawing blood into sterile culture bottles, minimizing the risk of contamination with well-established guidelines. These culture bottles contain specific growth media that support the replication of microorganisms if they are present. Once the blood specimen is collected, it incubates, allowing any potential pathogens to grow. Subsequent analysis and identification of these microorganisms enable health care professionals (HCPs) to prescribe appropriate antimicrobial therapies to treat specific infections, contributing to more effective and targeted patient care.2
The reliability of blood culture results depends on minimizing contamination risk, a challenge inherent in the procedure. Contamination can lead to false-positive results, potentially misguiding treatment.3 HCPs must adhere to strict aseptic techniques during blood draws, ensuring proper skin preparation with antiseptic solutions. The use of sterile equipment and avoiding prolonged tourniquet application helps maintain the integrity of the blood specimen. Timely inoculation of blood into culture bottles and careful handling are essential to mitigate contamination risk.2 Regular training and reinforcement of proper techniques is important to uphold the accuracy of blood culture results and enhance the reliability of diagnoses and treatment decisions.3 Despite diligent contamination prevention efforts, health care systems struggle to maintain contamination rates below the 3.0% national benchmark set by the Clinical & Laboratory Standards Institute (CLSI).4
Blood culture contamination is a critical concern in clinical practice; it can lead to misdiagnosis, prolonged hospital stays, unnecessary antibiotic use, and increased health care costs.5 Monitoring blood culture contamination is integral to patient safety, avoiding inappropriate and potentially harmful treatment, providing efficient care, contributing to antibiotic stewardship, supporting cost efficiency, and maintaining quality assurance and clinical research practices for public health.6
The initial specimen diversion technique (ISDT) recently emerged as a potential strategy to reduce blood culture contamination rates. This technique involves diverting a small portion of the initial blood plus the skin plug from the hollow needle away from the primary collection site before filling the culture bottles. This process minimizes skin surface contaminants, providing a cleaner blood specimen for culturing.7
The ISDT was introduced as a result of historically elevated contamination rates.8 Despite implementing various mitigation methods, the US Department of Veterans Affairs (VA) Central Texas Healthcare System (VACTHCS) has struggled to meet the national benchmark of maintaining blood culture contamination < 3.0%. The VACTHCS is a 146-bed teaching hospital with about 30,000 annual visits at the Olin E. Teague Veterans Affairs Medical Center (OETVMC) emergency department (ED). VACTHCS conducted a 16-month pilot study using 2 commercially available ISDT devices and published the findings.8
The Military Construction, Veterans Affairs, and Related Agencies Appropriations Act, 2022 (MilCon-VA Act) committee report prioritized the reduction of blood culture contamination to < 1% to prevent health risks and harm to veterans undergoing blood testing for the diagnosis of sepsis.9 Because it had been 5 years since OETVMC began using an ISDT in the ED, the ISDT adaptation strategy for mitigating blood culture contamination was revisited per institution policy.
The objective of this quality improvement project was to analyze retrospective data to understand the long-term impact of ISDT use on blood culture contamination rates. We hypothesized that ISDT use would contribute to efforts to maintain OETVMC ED blood culture contamination rate below the national (3.0%) and VACTHCS (2.5%) thresholds. This project assessed the progress for reducing blood culture contamination compared with the pre-ISDT era.8
METHODS
This retrospective analysis compared the blood culture contamination rates 36 months before and after the introduction of the ISDT device at the OETVMC ED. The preimplementation period was from December 2014 through November 2017 (36 months) and the postimplementation period was December 2017 through November 2020 (36 months). Data were collected from the Department of Pathology and Microbiology blood culture records of all adult patients admitted to the hospital through the ED and required blood cultures for suspicion of infection. Protected health information and VA sensitive information were not collected: all data were deidentified. A total of 18,541 blood cultures were collected 36 months preimplementation and 14,865 blood cultures were collected up to 36 months postimplementation. For comparison purposes, a similar dataset was collected from patients’ blood samples drawn by phlebotomists in the laboratory, where there had been no previous issues with overcontamination; no ISDT devices were used in the collection of these samples.
Blood Culture Contamination Variable
Blood cultures were monitored using the BACT/ALERT 3D (bioMérieux) and subsequently BACT/ALERT VIRTUO (bioMérieux), with positive bottles characterized by VITEK MS Matrix Assisted Laser Desorption Ionization Time-of-Flight technology (bioMérieux) and automated susceptibility testing (VITEK 2 [bioMérieux]).10 In an updated review of blood culture contamination, the American Society for Microbiology used the College of American Pathologists' Q-Probes quality improvement studies as a guideline for classifying contamination. A sample was determined to be contaminated if ≥ 1 of the following organisms were found in only 1 bottle in a series of blood culture sets: coagulase-negative staphylococci, Micrococcus species, α-hemolytic viridans group streptococci, Corynebacterium species, Propionibacterium acnes, and Bacillus species.11 The contamination assessment criteria remained unchanged, except for use of an ISDT device in blood culture collection at the ED.
The VACTHCS Infection Prevention Department ensured that the ISDT device was available and that ED nurses were trained annually on its use to collect blood cultures. Monthly reports of contamination were sent to the nursing supervisor for corrective action and retraining. The initial performance improvement project was slated for 16 months but was expanded to a 6-year period of retrospective data to obtain strong correlation.
Statistical Analysis
Contamination rates were recorded monthly from the hospital laboratory information management system for 36 months both before and after ISDT adoption. Statistical analysis was performed using a 2-tailed unpaired t-test to compare monthly contamination rates for the 2 periods with GraphPad Prism version 10.0.0 for Windows.
RESULTS
Prior to 2017, the ED reported contamination rates above the national (3.0%) and OETVMC thresholds (2.5%), with a mean of 4.5% (95% CI, 3.90-4.90).8 After ISDT implementation, the ED showed significant improvement with a reduction to mean 2.6% (95% CI, 2.10-3.20) (P < .001) (Figure 1). Figure 2 shows monthly blood culture contamination rates at the ED from December 2014 through November 2020. Month 36 (November 2017) shows a clear dip in contamination rate when the ISDT was introduced and month 37 to month 44 show remarkably low contamination rates. During this time, the institute experimented with 2 ISDT devices, and closer scrutiny may reveal this period as an outlier due to the monitoring of ISDT application, as previously reported.8
The blood culture contamination rate for samples drawn by the phlebotomists in the laboratory (excluding the ED) was calculated during the same time period (Figure 3). Non-ED contamination rates remained below 2.5% for 69 of 72 months.
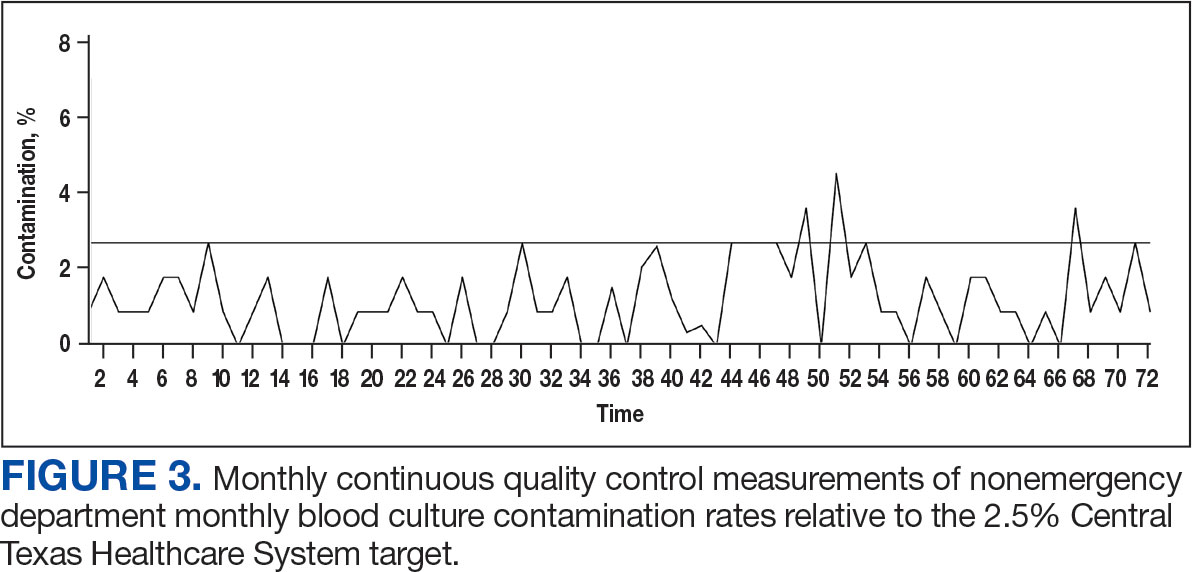
DISCUSSION
The blood culture contamination rate in the OETVMC ED dropped following ISDT implementation and continued to show long-term benefits. For the 36-month period following ISDT implementation, the mean contamination rate was 2.6%, which was below the national target threshold of 3.0% and close to the OETVMC target of 2.5%. These results suggest that ISDT can have a positive impact on patient care and laboratory efficiency. Improvements in the blood contamination rates in the ED can have a positive impact on the overall hospital contamination rates.
Blood drawn by phlebotomists in the hospital laboratory infrequently had contamination rates that exceeded the 2.5% target threshold. Because the non-ED contamination rates did not change throughout the comparison period, other factors were likely not involved in the improvements seen in the ED. The decision to implement ISDT exclusively in the ED was based on its historically elevated contamination rate.8 Issues with blood culture contamination in EDs across various hospital systems are well documented and not unique to VACTHCS.12
Contamination in blood cultures can be a significant issue in the hospital. It occurs when microorganisms from the skin or environment enter the blood culture sample during collection. Moreover, it can contribute to antibiotic resistance when patients are prescribed inappropriate antibiotics. It is also important to ensure HCPs are well-trained and consistently follow standardized protocols and understand the implications of false-positive results.13
ISDT helps reduce false-positive results and is a significant advancement in the field of blood culture collection.8,14 By discarding the initial blood, it ensures that only the true bloodstream sample is cultured, leading to more accurate results.15 It also may minimize the risk of contamination-related delays in diagnosis and treatment and benefits patients and health care institutions by potentially reducing hospital stays, unnecessary antibiotic use, and health care costs.
One of the ISDT device manufacturers estimated the financial impact on OETVMC based on the pilot project.8 While this study did not calculate the direct and indirect cost savings associated with this process improvement, the manufacturer’s website suggests that VACTHCS could annually save about $486,000.16 Furthermore, implementation of ISDT may improve laboratory efficiency, as they reduce the workload associated with identifying and reporting false-positive cultures. 6 ISDT devices represent a valuable tool in the efforts to reduce blood culture contamination and its wide-ranging implications in clinical settings. While ISDT alone will not be sufficient in achieving a lower threshold (< 1%) of blood culture contamination, it can be part of a multiprong effort that optimizes best practices in the collection, handling, and management of blood cultures.
Continuous quality improvement efforts and monitoring of blood culture contamination rates can help health care institutions identify problem areas and implement necessary changes. Addressing blood culture contamination can improve patient care, reduce costs, and address antibiotic resistance.
Limitations
This study was limited by its study design, which did not use a side-by-side comparison of blood cultures from groups with and without ISDT. All blood cultures from patients in the region were processed at OETVMC, which may not be representative of non-VA EDs. Part of this study took place during the COVID-19 pandemic, which may have skewed data. Additionally, hospital data were collected from a veteran population in Central Texas, and the lack of demographic diversity may not be generalizable to the greater population.
CONCLUSIONS
The findings of this study suggest ISDT may be effective in reducing blood culture contamination rates in the high-risk ED environment, which aligns with previous research. 5,14 The ISDT may help reduce blood culture contamination rates, improving the quality of patient care and reducing health care costs. MilCon-VA mandated that all VA facilities have blood culture contamination as a metric with a goal of blood culture contamination rates < 1%.8 However, achieving this goal remains a challenge. Further research and continuous quality improvement efforts are necessary to achieve it. Consistently achieving a contamination threshold of < 1% may require minimizing human error. An automated robotic venipuncture device, as recently designed and reported, may be necessary to reduce human error in blood draw and contamination.16
- Chela HK, Vasudevan A, Rojas-Moreno C, Naqvi SH. Approach to positive blood cultures in the hospitalized patient: a review. Mo Med. 2019;116(4):313-317.
- Lamy B, Dargère S, Arendrup MC, Parienti JJ, Tattevin P. How to optimize the use of blood cultures for the diagnosis of bloodstream infections? A state-of-the art. Front Microbiol. 2016;7:697. doi:10.3389/fmicb.2016.00697
- Doern GV, Carroll KC, Diekema DJ, et al. Practical guidance for clinical microbiology laboratories: a comprehensive update on the problem of blood culture contamination and a discussion of methods for addressing the problem. Clin Microbiol Rev. 2019;33:e00009-19. doi:10.1128/CMR.00009-19
- Wilson ML, Kirn Jr TJ, Antonara S, et al. Clinical and Laboratory Standards Institute Guideline M47—Principles and Procedures for Blood Cultures. Clinical and Laboratory Standards Institute. April 22, 2022. Accessed May 21, 2025. https://clsi.org/shop/standards/m47/
- Hancock JA, Campbell S, Jones MM, Wang-Rodriguez J, VHA Microbiology SME Workgroup, Klutts JS. Development and validation of a standardized blood culture contamination definition and metric dashboard for a large health care system. Am J Clin Pathol. 2023;160(3):255-260. doi:10.1093/ajcp/aqad044
- Shinozaki T, Deane RS, Mazuzan JE Jr, Hamel AJ, Hazelton D. Bacterial contamination of arterial lines. A prospective study. JAMA. 1983;249(2):223-225.
- Al Mohajer M, Lasco T. The impact of initial specimen diversion systems on blood culture contamination. Open Forum Infect Dis. 2023;10:ofad182. doi:10.1093/ofid/ofad182
- Arenas M, Boseman GM, Coppin JD, Lukey J, Jinadatha C, Navarathna DH. Asynchronous testing of 2 specimen-diversion devices to reduce blood culture contamination: a single-site product supply quality improvement project. J Emerg Nurs. 2021;47(2):256-264. e6. doi:10.1016/j.jen.2020.11.008
- Military Construction, Veterans Affairs, and Related Agencies Appropriations Act, 2022, HR 4355, 117th Cong (2021-2022). Accessed May 12, 2025. https://www.congress.gov/bill/117th-congress/house-bill/4355?
- Altun O, Almuhayawi M, Lüthje P, Taha R, Ullberg M, Özenci V. Controlled evaluation of the New BacT/ Alert Virtuo blood culture system for detection and time to detection of bacteria and yeasts. J Clin Microbiol. 2016;54(4):1148-1151. doi:10.1128/JCM.03362-15
- Hall KK, Lyman JA. Updated review of blood culture contamination. Clin Microbiol Rev. 2006;19(4):788-802. doi:10.1128/CMR.00062-05
- Gander RM, Byrd L, DeCrescenzo M, Hirany S, Bowen M, Baughman J. Impact of blood cultures drawn by phlebotomy on contamination rates and health care costs in a hospital emergency department. J Clin Microbiol. 2009;47(4):1021-1024. doi:10.1128/JCM.02162-08
- Garcia RA, Spitzer ED, Beaudry J, et al. Multidisciplinary team review of best practices for collection and handling of blood cultures to determine effective interventions for increasing the yield of true-positive bacteremias, reducing contamination, and eliminating false-positive central lineassociated bloodstream infections. Am J Infect Control. 2015;43(11):1222-1237. doi:10.1016/j.ajic.2015.06.030
- Callado GY, Lin V, Thottacherry E, et al. Diagnostic stewardship: a systematic review and meta-analysis of blood collection diversion devices used to reduce blood culture contamination and improve the accuracy of diagnosis in clinical settings. Open Forum Infect Dis. 2023;10(9):ofad433. doi:10.1093/ofid/ofad433
- Patton RG, Schmitt T. Innovation for reducing blood culture contamination: initial specimen diversion technique. J Clin Microbiol. 2010;48:4501-4503. doi:10.1128/JCM.00910-10
- Kurin. Clinical evidence: published Kurin studies. 2024. Accessed May 12, 2025. https://www.kurin.com/studies
- Leipheimer JM, Balter ML, Chen AI, et al. First-in-human evaluation of a hand-held automated venipuncture device for rapid venous blood draws. Technology (Singap World Sci). 2019;7(3-4):98-107. doi:10.1142/S2339547819500067?
Blood cultures provide crucial evidence for diagnostic medicine, specifically aimed at identifying the presence of microbial infections in the bloodstream. Blood culturing is instrumental in diagnosing conditions such as sepsis, bacteremia, or fungemia, where the identification of the causative agent is necessary for targeted and effective treatment.1
The process involves aseptically drawing blood into sterile culture bottles, minimizing the risk of contamination with well-established guidelines. These culture bottles contain specific growth media that support the replication of microorganisms if they are present. Once the blood specimen is collected, it incubates, allowing any potential pathogens to grow. Subsequent analysis and identification of these microorganisms enable health care professionals (HCPs) to prescribe appropriate antimicrobial therapies to treat specific infections, contributing to more effective and targeted patient care.2
The reliability of blood culture results depends on minimizing contamination risk, a challenge inherent in the procedure. Contamination can lead to false-positive results, potentially misguiding treatment.3 HCPs must adhere to strict aseptic techniques during blood draws, ensuring proper skin preparation with antiseptic solutions. The use of sterile equipment and avoiding prolonged tourniquet application helps maintain the integrity of the blood specimen. Timely inoculation of blood into culture bottles and careful handling are essential to mitigate contamination risk.2 Regular training and reinforcement of proper techniques is important to uphold the accuracy of blood culture results and enhance the reliability of diagnoses and treatment decisions.3 Despite diligent contamination prevention efforts, health care systems struggle to maintain contamination rates below the 3.0% national benchmark set by the Clinical & Laboratory Standards Institute (CLSI).4
Blood culture contamination is a critical concern in clinical practice; it can lead to misdiagnosis, prolonged hospital stays, unnecessary antibiotic use, and increased health care costs.5 Monitoring blood culture contamination is integral to patient safety, avoiding inappropriate and potentially harmful treatment, providing efficient care, contributing to antibiotic stewardship, supporting cost efficiency, and maintaining quality assurance and clinical research practices for public health.6
The initial specimen diversion technique (ISDT) recently emerged as a potential strategy to reduce blood culture contamination rates. This technique involves diverting a small portion of the initial blood plus the skin plug from the hollow needle away from the primary collection site before filling the culture bottles. This process minimizes skin surface contaminants, providing a cleaner blood specimen for culturing.7
The ISDT was introduced as a result of historically elevated contamination rates.8 Despite implementing various mitigation methods, the US Department of Veterans Affairs (VA) Central Texas Healthcare System (VACTHCS) has struggled to meet the national benchmark of maintaining blood culture contamination < 3.0%. The VACTHCS is a 146-bed teaching hospital with about 30,000 annual visits at the Olin E. Teague Veterans Affairs Medical Center (OETVMC) emergency department (ED). VACTHCS conducted a 16-month pilot study using 2 commercially available ISDT devices and published the findings.8
The Military Construction, Veterans Affairs, and Related Agencies Appropriations Act, 2022 (MilCon-VA Act) committee report prioritized the reduction of blood culture contamination to < 1% to prevent health risks and harm to veterans undergoing blood testing for the diagnosis of sepsis.9 Because it had been 5 years since OETVMC began using an ISDT in the ED, the ISDT adaptation strategy for mitigating blood culture contamination was revisited per institution policy.
The objective of this quality improvement project was to analyze retrospective data to understand the long-term impact of ISDT use on blood culture contamination rates. We hypothesized that ISDT use would contribute to efforts to maintain OETVMC ED blood culture contamination rate below the national (3.0%) and VACTHCS (2.5%) thresholds. This project assessed the progress for reducing blood culture contamination compared with the pre-ISDT era.8
METHODS
This retrospective analysis compared the blood culture contamination rates 36 months before and after the introduction of the ISDT device at the OETVMC ED. The preimplementation period was from December 2014 through November 2017 (36 months) and the postimplementation period was December 2017 through November 2020 (36 months). Data were collected from the Department of Pathology and Microbiology blood culture records of all adult patients admitted to the hospital through the ED and required blood cultures for suspicion of infection. Protected health information and VA sensitive information were not collected: all data were deidentified. A total of 18,541 blood cultures were collected 36 months preimplementation and 14,865 blood cultures were collected up to 36 months postimplementation. For comparison purposes, a similar dataset was collected from patients’ blood samples drawn by phlebotomists in the laboratory, where there had been no previous issues with overcontamination; no ISDT devices were used in the collection of these samples.
Blood Culture Contamination Variable
Blood cultures were monitored using the BACT/ALERT 3D (bioMérieux) and subsequently BACT/ALERT VIRTUO (bioMérieux), with positive bottles characterized by VITEK MS Matrix Assisted Laser Desorption Ionization Time-of-Flight technology (bioMérieux) and automated susceptibility testing (VITEK 2 [bioMérieux]).10 In an updated review of blood culture contamination, the American Society for Microbiology used the College of American Pathologists' Q-Probes quality improvement studies as a guideline for classifying contamination. A sample was determined to be contaminated if ≥ 1 of the following organisms were found in only 1 bottle in a series of blood culture sets: coagulase-negative staphylococci, Micrococcus species, α-hemolytic viridans group streptococci, Corynebacterium species, Propionibacterium acnes, and Bacillus species.11 The contamination assessment criteria remained unchanged, except for use of an ISDT device in blood culture collection at the ED.
The VACTHCS Infection Prevention Department ensured that the ISDT device was available and that ED nurses were trained annually on its use to collect blood cultures. Monthly reports of contamination were sent to the nursing supervisor for corrective action and retraining. The initial performance improvement project was slated for 16 months but was expanded to a 6-year period of retrospective data to obtain strong correlation.
Statistical Analysis
Contamination rates were recorded monthly from the hospital laboratory information management system for 36 months both before and after ISDT adoption. Statistical analysis was performed using a 2-tailed unpaired t-test to compare monthly contamination rates for the 2 periods with GraphPad Prism version 10.0.0 for Windows.
RESULTS
Prior to 2017, the ED reported contamination rates above the national (3.0%) and OETVMC thresholds (2.5%), with a mean of 4.5% (95% CI, 3.90-4.90).8 After ISDT implementation, the ED showed significant improvement with a reduction to mean 2.6% (95% CI, 2.10-3.20) (P < .001) (Figure 1). Figure 2 shows monthly blood culture contamination rates at the ED from December 2014 through November 2020. Month 36 (November 2017) shows a clear dip in contamination rate when the ISDT was introduced and month 37 to month 44 show remarkably low contamination rates. During this time, the institute experimented with 2 ISDT devices, and closer scrutiny may reveal this period as an outlier due to the monitoring of ISDT application, as previously reported.8


The blood culture contamination rate for samples drawn by the phlebotomists in the laboratory (excluding the ED) was calculated during the same time period (Figure 3). Non-ED contamination rates remained below 2.5% for 69 of 72 months.

DISCUSSION
The blood culture contamination rate in the OETVMC ED dropped following ISDT implementation and continued to show long-term benefits. For the 36-month period following ISDT implementation, the mean contamination rate was 2.6%, which was below the national target threshold of 3.0% and close to the OETVMC target of 2.5%. These results suggest that ISDT can have a positive impact on patient care and laboratory efficiency. Improvements in the blood contamination rates in the ED can have a positive impact on the overall hospital contamination rates.
Blood drawn by phlebotomists in the hospital laboratory infrequently had contamination rates that exceeded the 2.5% target threshold. Because the non-ED contamination rates did not change throughout the comparison period, other factors were likely not involved in the improvements seen in the ED. The decision to implement ISDT exclusively in the ED was based on its historically elevated contamination rate.8 Issues with blood culture contamination in EDs across various hospital systems are well documented and not unique to VACTHCS.12
Contamination in blood cultures can be a significant issue in the hospital. It occurs when microorganisms from the skin or environment enter the blood culture sample during collection. Moreover, it can contribute to antibiotic resistance when patients are prescribed inappropriate antibiotics. It is also important to ensure HCPs are well-trained and consistently follow standardized protocols and understand the implications of false-positive results.13
ISDT helps reduce false-positive results and is a significant advancement in the field of blood culture collection.8,14 By discarding the initial blood, it ensures that only the true bloodstream sample is cultured, leading to more accurate results.15 It also may minimize the risk of contamination-related delays in diagnosis and treatment and benefits patients and health care institutions by potentially reducing hospital stays, unnecessary antibiotic use, and health care costs.
One of the ISDT device manufacturers estimated the financial impact on OETVMC based on the pilot project.8 While this study did not calculate the direct and indirect cost savings associated with this process improvement, the manufacturer’s website suggests that VACTHCS could annually save about $486,000.16 Furthermore, implementation of ISDT may improve laboratory efficiency, as they reduce the workload associated with identifying and reporting false-positive cultures. 6 ISDT devices represent a valuable tool in the efforts to reduce blood culture contamination and its wide-ranging implications in clinical settings. While ISDT alone will not be sufficient in achieving a lower threshold (< 1%) of blood culture contamination, it can be part of a multiprong effort that optimizes best practices in the collection, handling, and management of blood cultures.
Continuous quality improvement efforts and monitoring of blood culture contamination rates can help health care institutions identify problem areas and implement necessary changes. Addressing blood culture contamination can improve patient care, reduce costs, and address antibiotic resistance.
Limitations
This study was limited by its study design, which did not use a side-by-side comparison of blood cultures from groups with and without ISDT. All blood cultures from patients in the region were processed at OETVMC, which may not be representative of non-VA EDs. Part of this study took place during the COVID-19 pandemic, which may have skewed data. Additionally, hospital data were collected from a veteran population in Central Texas, and the lack of demographic diversity may not be generalizable to the greater population.
CONCLUSIONS
The findings of this study suggest ISDT may be effective in reducing blood culture contamination rates in the high-risk ED environment, which aligns with previous research. 5,14 The ISDT may help reduce blood culture contamination rates, improving the quality of patient care and reducing health care costs. MilCon-VA mandated that all VA facilities have blood culture contamination as a metric with a goal of blood culture contamination rates < 1%.8 However, achieving this goal remains a challenge. Further research and continuous quality improvement efforts are necessary to achieve it. Consistently achieving a contamination threshold of < 1% may require minimizing human error. An automated robotic venipuncture device, as recently designed and reported, may be necessary to reduce human error in blood draw and contamination.16
Blood cultures provide crucial evidence for diagnostic medicine, specifically aimed at identifying the presence of microbial infections in the bloodstream. Blood culturing is instrumental in diagnosing conditions such as sepsis, bacteremia, or fungemia, where the identification of the causative agent is necessary for targeted and effective treatment.1
The process involves aseptically drawing blood into sterile culture bottles, minimizing the risk of contamination with well-established guidelines. These culture bottles contain specific growth media that support the replication of microorganisms if they are present. Once the blood specimen is collected, it incubates, allowing any potential pathogens to grow. Subsequent analysis and identification of these microorganisms enable health care professionals (HCPs) to prescribe appropriate antimicrobial therapies to treat specific infections, contributing to more effective and targeted patient care.2
The reliability of blood culture results depends on minimizing contamination risk, a challenge inherent in the procedure. Contamination can lead to false-positive results, potentially misguiding treatment.3 HCPs must adhere to strict aseptic techniques during blood draws, ensuring proper skin preparation with antiseptic solutions. The use of sterile equipment and avoiding prolonged tourniquet application helps maintain the integrity of the blood specimen. Timely inoculation of blood into culture bottles and careful handling are essential to mitigate contamination risk.2 Regular training and reinforcement of proper techniques is important to uphold the accuracy of blood culture results and enhance the reliability of diagnoses and treatment decisions.3 Despite diligent contamination prevention efforts, health care systems struggle to maintain contamination rates below the 3.0% national benchmark set by the Clinical & Laboratory Standards Institute (CLSI).4
Blood culture contamination is a critical concern in clinical practice; it can lead to misdiagnosis, prolonged hospital stays, unnecessary antibiotic use, and increased health care costs.5 Monitoring blood culture contamination is integral to patient safety, avoiding inappropriate and potentially harmful treatment, providing efficient care, contributing to antibiotic stewardship, supporting cost efficiency, and maintaining quality assurance and clinical research practices for public health.6
The initial specimen diversion technique (ISDT) recently emerged as a potential strategy to reduce blood culture contamination rates. This technique involves diverting a small portion of the initial blood plus the skin plug from the hollow needle away from the primary collection site before filling the culture bottles. This process minimizes skin surface contaminants, providing a cleaner blood specimen for culturing.7
The ISDT was introduced as a result of historically elevated contamination rates.8 Despite implementing various mitigation methods, the US Department of Veterans Affairs (VA) Central Texas Healthcare System (VACTHCS) has struggled to meet the national benchmark of maintaining blood culture contamination < 3.0%. The VACTHCS is a 146-bed teaching hospital with about 30,000 annual visits at the Olin E. Teague Veterans Affairs Medical Center (OETVMC) emergency department (ED). VACTHCS conducted a 16-month pilot study using 2 commercially available ISDT devices and published the findings.8
The Military Construction, Veterans Affairs, and Related Agencies Appropriations Act, 2022 (MilCon-VA Act) committee report prioritized the reduction of blood culture contamination to < 1% to prevent health risks and harm to veterans undergoing blood testing for the diagnosis of sepsis.9 Because it had been 5 years since OETVMC began using an ISDT in the ED, the ISDT adaptation strategy for mitigating blood culture contamination was revisited per institution policy.
The objective of this quality improvement project was to analyze retrospective data to understand the long-term impact of ISDT use on blood culture contamination rates. We hypothesized that ISDT use would contribute to efforts to maintain OETVMC ED blood culture contamination rate below the national (3.0%) and VACTHCS (2.5%) thresholds. This project assessed the progress for reducing blood culture contamination compared with the pre-ISDT era.8
METHODS
This retrospective analysis compared the blood culture contamination rates 36 months before and after the introduction of the ISDT device at the OETVMC ED. The preimplementation period was from December 2014 through November 2017 (36 months) and the postimplementation period was December 2017 through November 2020 (36 months). Data were collected from the Department of Pathology and Microbiology blood culture records of all adult patients admitted to the hospital through the ED and required blood cultures for suspicion of infection. Protected health information and VA sensitive information were not collected: all data were deidentified. A total of 18,541 blood cultures were collected 36 months preimplementation and 14,865 blood cultures were collected up to 36 months postimplementation. For comparison purposes, a similar dataset was collected from patients’ blood samples drawn by phlebotomists in the laboratory, where there had been no previous issues with overcontamination; no ISDT devices were used in the collection of these samples.
Blood Culture Contamination Variable
Blood cultures were monitored using the BACT/ALERT 3D (bioMérieux) and subsequently BACT/ALERT VIRTUO (bioMérieux), with positive bottles characterized by VITEK MS Matrix Assisted Laser Desorption Ionization Time-of-Flight technology (bioMérieux) and automated susceptibility testing (VITEK 2 [bioMérieux]).10 In an updated review of blood culture contamination, the American Society for Microbiology used the College of American Pathologists' Q-Probes quality improvement studies as a guideline for classifying contamination. A sample was determined to be contaminated if ≥ 1 of the following organisms were found in only 1 bottle in a series of blood culture sets: coagulase-negative staphylococci, Micrococcus species, α-hemolytic viridans group streptococci, Corynebacterium species, Propionibacterium acnes, and Bacillus species.11 The contamination assessment criteria remained unchanged, except for use of an ISDT device in blood culture collection at the ED.
The VACTHCS Infection Prevention Department ensured that the ISDT device was available and that ED nurses were trained annually on its use to collect blood cultures. Monthly reports of contamination were sent to the nursing supervisor for corrective action and retraining. The initial performance improvement project was slated for 16 months but was expanded to a 6-year period of retrospective data to obtain strong correlation.
Statistical Analysis
Contamination rates were recorded monthly from the hospital laboratory information management system for 36 months both before and after ISDT adoption. Statistical analysis was performed using a 2-tailed unpaired t-test to compare monthly contamination rates for the 2 periods with GraphPad Prism version 10.0.0 for Windows.
RESULTS
Prior to 2017, the ED reported contamination rates above the national (3.0%) and OETVMC thresholds (2.5%), with a mean of 4.5% (95% CI, 3.90-4.90).8 After ISDT implementation, the ED showed significant improvement with a reduction to mean 2.6% (95% CI, 2.10-3.20) (P < .001) (Figure 1). Figure 2 shows monthly blood culture contamination rates at the ED from December 2014 through November 2020. Month 36 (November 2017) shows a clear dip in contamination rate when the ISDT was introduced and month 37 to month 44 show remarkably low contamination rates. During this time, the institute experimented with 2 ISDT devices, and closer scrutiny may reveal this period as an outlier due to the monitoring of ISDT application, as previously reported.8


The blood culture contamination rate for samples drawn by the phlebotomists in the laboratory (excluding the ED) was calculated during the same time period (Figure 3). Non-ED contamination rates remained below 2.5% for 69 of 72 months.

DISCUSSION
The blood culture contamination rate in the OETVMC ED dropped following ISDT implementation and continued to show long-term benefits. For the 36-month period following ISDT implementation, the mean contamination rate was 2.6%, which was below the national target threshold of 3.0% and close to the OETVMC target of 2.5%. These results suggest that ISDT can have a positive impact on patient care and laboratory efficiency. Improvements in the blood contamination rates in the ED can have a positive impact on the overall hospital contamination rates.
Blood drawn by phlebotomists in the hospital laboratory infrequently had contamination rates that exceeded the 2.5% target threshold. Because the non-ED contamination rates did not change throughout the comparison period, other factors were likely not involved in the improvements seen in the ED. The decision to implement ISDT exclusively in the ED was based on its historically elevated contamination rate.8 Issues with blood culture contamination in EDs across various hospital systems are well documented and not unique to VACTHCS.12
Contamination in blood cultures can be a significant issue in the hospital. It occurs when microorganisms from the skin or environment enter the blood culture sample during collection. Moreover, it can contribute to antibiotic resistance when patients are prescribed inappropriate antibiotics. It is also important to ensure HCPs are well-trained and consistently follow standardized protocols and understand the implications of false-positive results.13
ISDT helps reduce false-positive results and is a significant advancement in the field of blood culture collection.8,14 By discarding the initial blood, it ensures that only the true bloodstream sample is cultured, leading to more accurate results.15 It also may minimize the risk of contamination-related delays in diagnosis and treatment and benefits patients and health care institutions by potentially reducing hospital stays, unnecessary antibiotic use, and health care costs.
One of the ISDT device manufacturers estimated the financial impact on OETVMC based on the pilot project.8 While this study did not calculate the direct and indirect cost savings associated with this process improvement, the manufacturer’s website suggests that VACTHCS could annually save about $486,000.16 Furthermore, implementation of ISDT may improve laboratory efficiency, as they reduce the workload associated with identifying and reporting false-positive cultures. 6 ISDT devices represent a valuable tool in the efforts to reduce blood culture contamination and its wide-ranging implications in clinical settings. While ISDT alone will not be sufficient in achieving a lower threshold (< 1%) of blood culture contamination, it can be part of a multiprong effort that optimizes best practices in the collection, handling, and management of blood cultures.
Continuous quality improvement efforts and monitoring of blood culture contamination rates can help health care institutions identify problem areas and implement necessary changes. Addressing blood culture contamination can improve patient care, reduce costs, and address antibiotic resistance.
Limitations
This study was limited by its study design, which did not use a side-by-side comparison of blood cultures from groups with and without ISDT. All blood cultures from patients in the region were processed at OETVMC, which may not be representative of non-VA EDs. Part of this study took place during the COVID-19 pandemic, which may have skewed data. Additionally, hospital data were collected from a veteran population in Central Texas, and the lack of demographic diversity may not be generalizable to the greater population.
CONCLUSIONS
The findings of this study suggest ISDT may be effective in reducing blood culture contamination rates in the high-risk ED environment, which aligns with previous research. 5,14 The ISDT may help reduce blood culture contamination rates, improving the quality of patient care and reducing health care costs. MilCon-VA mandated that all VA facilities have blood culture contamination as a metric with a goal of blood culture contamination rates < 1%.8 However, achieving this goal remains a challenge. Further research and continuous quality improvement efforts are necessary to achieve it. Consistently achieving a contamination threshold of < 1% may require minimizing human error. An automated robotic venipuncture device, as recently designed and reported, may be necessary to reduce human error in blood draw and contamination.16
- Chela HK, Vasudevan A, Rojas-Moreno C, Naqvi SH. Approach to positive blood cultures in the hospitalized patient: a review. Mo Med. 2019;116(4):313-317.
- Lamy B, Dargère S, Arendrup MC, Parienti JJ, Tattevin P. How to optimize the use of blood cultures for the diagnosis of bloodstream infections? A state-of-the art. Front Microbiol. 2016;7:697. doi:10.3389/fmicb.2016.00697
- Doern GV, Carroll KC, Diekema DJ, et al. Practical guidance for clinical microbiology laboratories: a comprehensive update on the problem of blood culture contamination and a discussion of methods for addressing the problem. Clin Microbiol Rev. 2019;33:e00009-19. doi:10.1128/CMR.00009-19
- Wilson ML, Kirn Jr TJ, Antonara S, et al. Clinical and Laboratory Standards Institute Guideline M47—Principles and Procedures for Blood Cultures. Clinical and Laboratory Standards Institute. April 22, 2022. Accessed May 21, 2025. https://clsi.org/shop/standards/m47/
- Hancock JA, Campbell S, Jones MM, Wang-Rodriguez J, VHA Microbiology SME Workgroup, Klutts JS. Development and validation of a standardized blood culture contamination definition and metric dashboard for a large health care system. Am J Clin Pathol. 2023;160(3):255-260. doi:10.1093/ajcp/aqad044
- Shinozaki T, Deane RS, Mazuzan JE Jr, Hamel AJ, Hazelton D. Bacterial contamination of arterial lines. A prospective study. JAMA. 1983;249(2):223-225.
- Al Mohajer M, Lasco T. The impact of initial specimen diversion systems on blood culture contamination. Open Forum Infect Dis. 2023;10:ofad182. doi:10.1093/ofid/ofad182
- Arenas M, Boseman GM, Coppin JD, Lukey J, Jinadatha C, Navarathna DH. Asynchronous testing of 2 specimen-diversion devices to reduce blood culture contamination: a single-site product supply quality improvement project. J Emerg Nurs. 2021;47(2):256-264. e6. doi:10.1016/j.jen.2020.11.008
- Military Construction, Veterans Affairs, and Related Agencies Appropriations Act, 2022, HR 4355, 117th Cong (2021-2022). Accessed May 12, 2025. https://www.congress.gov/bill/117th-congress/house-bill/4355?
- Altun O, Almuhayawi M, Lüthje P, Taha R, Ullberg M, Özenci V. Controlled evaluation of the New BacT/ Alert Virtuo blood culture system for detection and time to detection of bacteria and yeasts. J Clin Microbiol. 2016;54(4):1148-1151. doi:10.1128/JCM.03362-15
- Hall KK, Lyman JA. Updated review of blood culture contamination. Clin Microbiol Rev. 2006;19(4):788-802. doi:10.1128/CMR.00062-05
- Gander RM, Byrd L, DeCrescenzo M, Hirany S, Bowen M, Baughman J. Impact of blood cultures drawn by phlebotomy on contamination rates and health care costs in a hospital emergency department. J Clin Microbiol. 2009;47(4):1021-1024. doi:10.1128/JCM.02162-08
- Garcia RA, Spitzer ED, Beaudry J, et al. Multidisciplinary team review of best practices for collection and handling of blood cultures to determine effective interventions for increasing the yield of true-positive bacteremias, reducing contamination, and eliminating false-positive central lineassociated bloodstream infections. Am J Infect Control. 2015;43(11):1222-1237. doi:10.1016/j.ajic.2015.06.030
- Callado GY, Lin V, Thottacherry E, et al. Diagnostic stewardship: a systematic review and meta-analysis of blood collection diversion devices used to reduce blood culture contamination and improve the accuracy of diagnosis in clinical settings. Open Forum Infect Dis. 2023;10(9):ofad433. doi:10.1093/ofid/ofad433
- Patton RG, Schmitt T. Innovation for reducing blood culture contamination: initial specimen diversion technique. J Clin Microbiol. 2010;48:4501-4503. doi:10.1128/JCM.00910-10
- Kurin. Clinical evidence: published Kurin studies. 2024. Accessed May 12, 2025. https://www.kurin.com/studies
- Leipheimer JM, Balter ML, Chen AI, et al. First-in-human evaluation of a hand-held automated venipuncture device for rapid venous blood draws. Technology (Singap World Sci). 2019;7(3-4):98-107. doi:10.1142/S2339547819500067?
- Chela HK, Vasudevan A, Rojas-Moreno C, Naqvi SH. Approach to positive blood cultures in the hospitalized patient: a review. Mo Med. 2019;116(4):313-317.
- Lamy B, Dargère S, Arendrup MC, Parienti JJ, Tattevin P. How to optimize the use of blood cultures for the diagnosis of bloodstream infections? A state-of-the art. Front Microbiol. 2016;7:697. doi:10.3389/fmicb.2016.00697
- Doern GV, Carroll KC, Diekema DJ, et al. Practical guidance for clinical microbiology laboratories: a comprehensive update on the problem of blood culture contamination and a discussion of methods for addressing the problem. Clin Microbiol Rev. 2019;33:e00009-19. doi:10.1128/CMR.00009-19
- Wilson ML, Kirn Jr TJ, Antonara S, et al. Clinical and Laboratory Standards Institute Guideline M47—Principles and Procedures for Blood Cultures. Clinical and Laboratory Standards Institute. April 22, 2022. Accessed May 21, 2025. https://clsi.org/shop/standards/m47/
- Hancock JA, Campbell S, Jones MM, Wang-Rodriguez J, VHA Microbiology SME Workgroup, Klutts JS. Development and validation of a standardized blood culture contamination definition and metric dashboard for a large health care system. Am J Clin Pathol. 2023;160(3):255-260. doi:10.1093/ajcp/aqad044
- Shinozaki T, Deane RS, Mazuzan JE Jr, Hamel AJ, Hazelton D. Bacterial contamination of arterial lines. A prospective study. JAMA. 1983;249(2):223-225.
- Al Mohajer M, Lasco T. The impact of initial specimen diversion systems on blood culture contamination. Open Forum Infect Dis. 2023;10:ofad182. doi:10.1093/ofid/ofad182
- Arenas M, Boseman GM, Coppin JD, Lukey J, Jinadatha C, Navarathna DH. Asynchronous testing of 2 specimen-diversion devices to reduce blood culture contamination: a single-site product supply quality improvement project. J Emerg Nurs. 2021;47(2):256-264. e6. doi:10.1016/j.jen.2020.11.008
- Military Construction, Veterans Affairs, and Related Agencies Appropriations Act, 2022, HR 4355, 117th Cong (2021-2022). Accessed May 12, 2025. https://www.congress.gov/bill/117th-congress/house-bill/4355?
- Altun O, Almuhayawi M, Lüthje P, Taha R, Ullberg M, Özenci V. Controlled evaluation of the New BacT/ Alert Virtuo blood culture system for detection and time to detection of bacteria and yeasts. J Clin Microbiol. 2016;54(4):1148-1151. doi:10.1128/JCM.03362-15
- Hall KK, Lyman JA. Updated review of blood culture contamination. Clin Microbiol Rev. 2006;19(4):788-802. doi:10.1128/CMR.00062-05
- Gander RM, Byrd L, DeCrescenzo M, Hirany S, Bowen M, Baughman J. Impact of blood cultures drawn by phlebotomy on contamination rates and health care costs in a hospital emergency department. J Clin Microbiol. 2009;47(4):1021-1024. doi:10.1128/JCM.02162-08
- Garcia RA, Spitzer ED, Beaudry J, et al. Multidisciplinary team review of best practices for collection and handling of blood cultures to determine effective interventions for increasing the yield of true-positive bacteremias, reducing contamination, and eliminating false-positive central lineassociated bloodstream infections. Am J Infect Control. 2015;43(11):1222-1237. doi:10.1016/j.ajic.2015.06.030
- Callado GY, Lin V, Thottacherry E, et al. Diagnostic stewardship: a systematic review and meta-analysis of blood collection diversion devices used to reduce blood culture contamination and improve the accuracy of diagnosis in clinical settings. Open Forum Infect Dis. 2023;10(9):ofad433. doi:10.1093/ofid/ofad433
- Patton RG, Schmitt T. Innovation for reducing blood culture contamination: initial specimen diversion technique. J Clin Microbiol. 2010;48:4501-4503. doi:10.1128/JCM.00910-10
- Kurin. Clinical evidence: published Kurin studies. 2024. Accessed May 12, 2025. https://www.kurin.com/studies
- Leipheimer JM, Balter ML, Chen AI, et al. First-in-human evaluation of a hand-held automated venipuncture device for rapid venous blood draws. Technology (Singap World Sci). 2019;7(3-4):98-107. doi:10.1142/S2339547819500067?
Impact of Initial Specimen Diversion Technique on Blood Culture Contamination Rates
Impact of Initial Specimen Diversion Technique on Blood Culture Contamination Rates
Elusive Edema: A Case of Nephrotic Syndrome Mimicking Decompensated Cirrhosis
Elusive Edema: A Case of Nephrotic Syndrome Mimicking Decompensated Cirrhosis
Histology is the gold standard for cirrhosis diagnosis. However, a combination of clinical history, physical examination findings, and supportive laboratory and radiographic features is generally sufficient to make the diagnosis. Routine ultrasound and computed tomography (CT) imaging often identifies a nodular liver contour with sequelae of portal hypertension, including splenomegaly, varices, and ascites, which can suggest cirrhosis when supported by laboratory parameters and clinical features. As a result, the diagnosis is typically made clinically.1 Many patients with compensated cirrhosis go undetected. The presence of a decompensation event (ascites, spontaneous bacterial peritonitis, variceal hemorrhage, or hepatic encephalopathy) often leads to index diagnosis when patients were previously compensated. When a patient presents with suspected decompensated cirrhosis, it is important to consider other diagnoses with similar presentations and ensure that multiple disease processes are not contributing to the symptoms.
CASE PRESENTATION
A 64-year-old male with a history of intravenous (IV) methamphetamine use and prior incarceration presented with a 3-week history of progressively worsening generalized swelling. Prior to the onset of his symptoms, the patient injured his right lower extremity (RLE) in a bicycle accident, resulting in edema that progressed to bilateral lower extremity (BLE) edema and worsening fatigue, despite resolution of the initial injury. The patient gained weight though he could not quantify the amount. He experienced progressive hunger, thirst, and fatigue as well as increased sleep. Additionally, the patient experienced worsening dyspnea on exertion and orthopnea. He started using 2 pillows instead of 1 pillow at night.
The patient reported no fevers, chills, sputum production, chest pain, or paroxysmal nocturnal dyspnea. He had no known history of sexually transmitted infections, no significant history of alcohol use, and occasional tobacco and marijuana use. He had been incarcerated > 10 years before and last used IV methamphetamine 3 years before. He did not regularly take any medications.
The patient’s vital signs included a temperature of 98.2 °F; 78/min heart rate; 15/min respiratory rate; 159/109 mm Hg blood pressure; and 98% oxygen saturation on room air. He had gained 20 lbs in the past 4 months. He had pitting edema in both legs and arms, as well as periorbital swelling, but no jugular venous distention, abnormal heart sounds, or murmurs. Breath sounds were distant but clear to auscultation. His abdomen was distended with normal bowel sounds and no fluid wave; mild epigastric tenderness was present, but no intra-abdominal masses were palpated. He had spider angiomata on the upper chest but no other stigmata of cirrhosis, such as caput medusae or jaundice. Tattoos were noted.
Laboratory test results showed a platelet count of 178 x 103/μL (reference range, 140- 440 ~ 103μL).Creatinine was 0.80 mg/dL (reference range, < 1.28 mg/dL), with an estimated glomerular filtration rate (eGFR) of 99 mL/min/1.73 m2 using the Chronic Kidney Disease-Epidemiology equation (reference range, > 60 mL/min/1.73 m2), (reference range, > 60 mL/min/1.73 m2), and Cystatin C was 1.14 mg/L (reference range, < 1.15 mg/L). His electrolytes and complete blood count were within normal limits, including sodium, 134 mmol/L; potassium, 4.4 mmol/L; chloride, 108 mmol/L; and carbon dioxide, 22.5 mmol/L.
Additional test results included alkaline phosphatase, 126 U/L (reference range, < 94 U/L); alanine transaminase, 41 U/L (reference range, < 45 U/L); aspartate aminotransferase, 70 U/L (reference range, < 35 U/L); total bilirubin, 0.6 mg/dL (reference range, < 1 mg/dL); albumin, 1.8 g/dL (reference range, 3.2-4.8 g/dL); and total protein, 6.3 g/dL (reference range, 5.9-8.3 g/dL). The patient’s international normalized ratio was 0.96 (reference range, 0.8-1.1), and brain natriuretic peptide was normal at 56 pg/mL. No prior laboratory results were available for comparison.
Urine toxicology was positive for amphetamines. Urinalysis demonstrated large occult blood, with a red blood cell count of 26/ HPF (reference range, 0/HPF) and proteinuria (100 mg/dL; reference range, negative), without bacteria, nitrites, or leukocyte esterase. Urine white blood cell count was 10/ HPF (reference range, 0/HPF), and fine granular casts and hyaline casts were present.
A noncontrast CT of the abdomen and pelvis in the emergency department showed an irregular liver contour with diffuse nodularity, multiple portosystemic collaterals, moderate abdominal and pelvic ascites, small bilateral pleural effusions with associated atelectasis, and anasarca consistent with cirrhosis (Figure 1). The patient was admitted to the internal medicine service for workup and management of newly diagnosed cirrhosis.
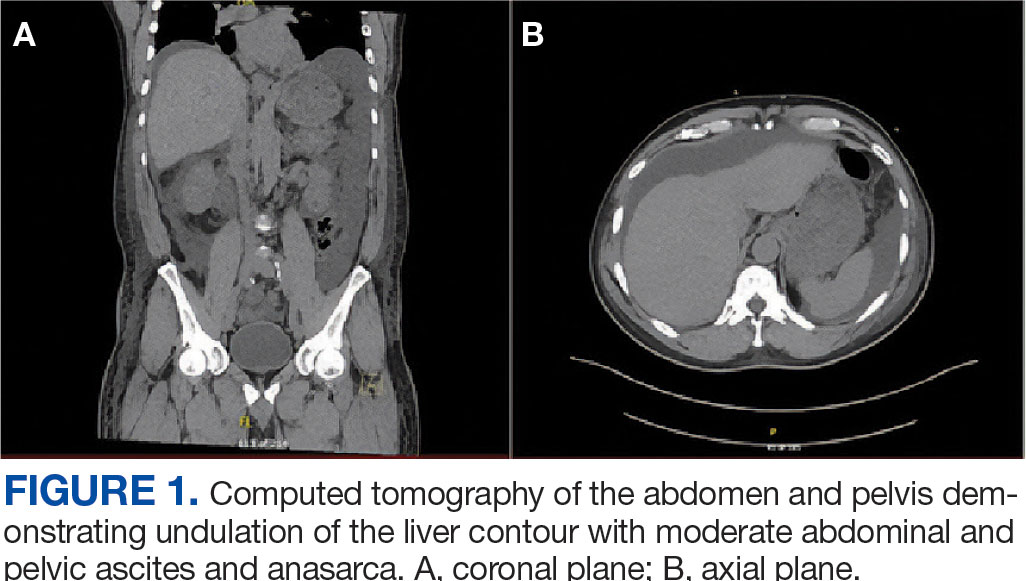
Paracentesis revealed straw-colored fluid with an ascitic fluid neutrophil count of 17/μL, a protein level of < 3 g/dL and albumin level of < 1.5 g/dL. Gram stain of the ascitic fluid showed a moderate white blood cell count with no organisms. Fluid culture showed no microbial growth.
Initial workup for cirrhosis demonstrated a positive total hepatitis A antibody. The patient had a nonreactive hepatitis B surface antigen and surface antibody, but a reactive hepatitis B core antibody; a hepatitis B DNA level was not ordered. He had a reactive hepatitis C antibody with a viral load of 4,490,000 II/mL (genotype 1a). The patient’s iron level was 120 μg/dL, with a calculated total iron-binding capacity (TIBC) of 126.2 μg/dL. His transferrin saturation (TSAT) (serum iron divided by TIBC) was 95%. The patient had nonreactive antinuclear antibody and antimitochondrial antibody tests and a positive antismooth muscle antibody test with a titer of 1:40. His α-fetoprotein (AFP) level was 505 ng/mL (reference range, < 8 ng/mL).
Follow-up MRI of the abdomen and pelvis showed cirrhotic morphology with large volume ascites and portosystemic collaterals, consistent with portal hypertension. Additionally, it showed multiple scattered peripheral sub centimeter hyperenhancing foci, most likely representing benign lesions.
The patient's spot urine protein-creatinine ratio was 3.76. To better quantify proteinuria, a 24-hour urine collection was performed and revealed 12.8 g/d of urine protein (reference range, 0-0.17 g/d). His serum triglyceride level was 175 mg/dL (reference range, 40-60 mg/dL); total cholesterol was 177 mg/ dL (reference range, ≤ 200 mg/dL); low density lipoprotein cholesterol was 98 mg/ dL (reference range, ≤ 130 mg/dL); and highdensity lipoprotein cholesterol was 43.8 mg/ dL (reference range, ≥ 40 mg/dL); C3 complement level was 71 mg/dL (reference range, 82-185 mg/dL); and C4 complement level was 22 mg/dL (reference range, 15-53 mg/ dL). His rheumatoid factor was < 14 IU/mL. Tests for rapid plasma reagin and HIV antigen- antibody were nonreactive, and the phospholipase A2 receptor antibody test was negative. The patient tested positive for QuantiFERON-TB Gold and qualitative cryoglobulin, which indicated a cryocrit of 1%.
A renal biopsy was performed, revealing diffuse podocyte foot process effacement and glomerulonephritis with low-grade C3 and immunoglobulin (Ig) G deposits, consistent with early membranoproliferative glomerulonephritis (MPGN) (Figures 2 and 3).
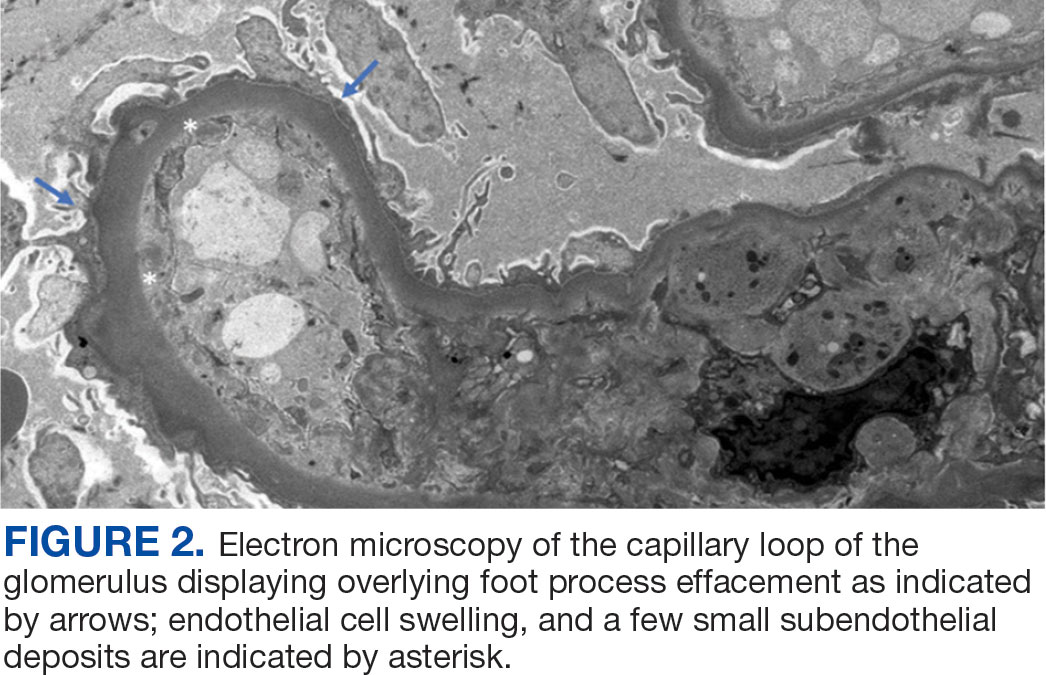
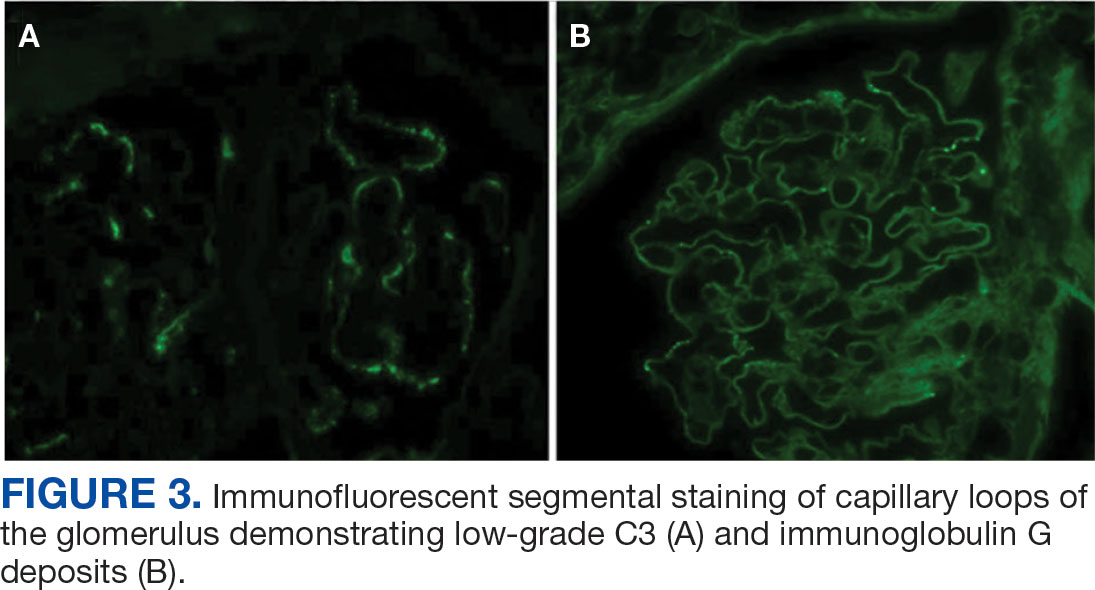
The patient was initially diuresed with IV furosemide without significant urine output. He was then diuresed with IV 25% albumin (total, 25 g), followed by IV furosemide 40 mg twice daily, which led to significant urine output and resolution of his anasarca. Given the patient’s hypoalbuminemic state, IV albumin was necessary to deliver furosemide to the proximal tubule. He was started on lisinopril for renal protection and discharged with spironolactone and furosemide for fluid management in the context of cirrhosis.
The patient was evaluated by the Liver Nodule Clinic, which includes specialists from hepatology, medical oncology, radiation oncology, interventional radiology, and diagnostic radiology. The team considered the patient’s medical history and characteristics of the nodules on imaging. Notable aspects of the patient’s history included hepatitis C virus (HCV) infection and an elevated AFP level, although imaging showed no lesion concerning for malignancy. Given these findings, the patient was scheduled for a liver biopsy to establish a tissue diagnosis of cirrhosis. Hepatology, nephrology, and infectious disease specialists coordinated to plan the management and treatment of latent tuberculosis (TB), chronic HCV, MPGN, compensated cirrhosis, and suspicious liver lesions.
The patient chose to handle management and treatment as an outpatient. He was discharged with furosemide and spironolactone for anasarca management, and amlodipine and lisinopril for his hypertension and MPGN. Follow-up appointments were scheduled with infectious disease for management of latent TB and HCV, nephrology for MPGN, gastroenterology for cirrhosis, and interventional radiology for liver biopsy. Unfortunately, the patient was unhoused with limited access to transportation, which prevented timely follow-up. Given these social factors, immunosuppression was not started. Additionally, he did not start on HCV therapy because the viral load was still pending at time of discharge.
DISCUSSION
The diagnosis of decompensated cirrhosis was prematurely established, resulting in a diagnostic delay, a form of diagnostic error. However, on hospital day 2, the initial hypothesis of decompensated cirrhosis as the sole driver of the patient’s presentation was reconsidered due to the disconnect between the severity of hypoalbuminemia and diffuse edema (anasarca), and the absence of laboratory evidence of hepatic decompensation (normal international normalized ratio, bilirubin, and low but normal platelet count). Although image findings supported cirrhosis, laboratory markers did not indicate hepatic decompensation. The severity of hypoalbuminemia and anasarca, along with an indeterminate Serum-Ascites Albumin Gradient, prompted the patient’s care team to consider other causes, specifically, nephrotic syndrome.
The patien’s spot protein-to-creatinine ratio was 3.76 (reference range < 0.2 mg/mg creatinine), but a 24-hour urine protein collection was 12.8 g/day (reference range < 150 mg/day). While most spot urine protein- to-creatinine ratios (UPCR) correlate with a 24-hour urine collection, discrepancies can occur, as in this case. It is important to recognize that the spot UPCR assumes that patients are excreting 1000 mg of creatinine daily in their urine, which is not always the case. In addition, changes in urine osmolality can lead to different values. The gold standard for proteinuria is a 24-hour urine collection for protein and creatinine.
The patient’s nephrotic-range proteinuria and severe hypoalbuminemia are not solely explained by cirrhosis. In addition, the patient’s lower extremity edema pointed to nephrotic syndrome. The differential diagnosis for nephrotic syndrome includes both primary and secondary forms of membranous nephropathy, minimal change disease, focal segmental glomerulosclerosis, and MPGN, a histopathological diagnosis that requires distinguishing between immune complex-mediated and complement-mediated forms. Other causes of nephrotic syndrome that do not fit in any of these buckets include amyloidosis, IgA nephropathy, and diabetes mellitus (DM). Despite DM being a common cause of nephrotic range proteinuria, it rarely leads to full nephrotic syndrome.
When considering the diagnosis, we reframed the patient’s clinical syndrome as compensated cirrhosis plus nephrotic syndrome. This approach prioritized identifying a cause that could explain both cirrhosis (from any cause) leading to IgA nephropathy or injection drug use serving as a risk factor for cirrhosis and nephrotic syndrome through HCV or AA amyloidosis, respectively. This problem representation guided us to the correct diagnosis. There are multiple renal diseases associated with HCV infection, including MPGN, membranous nephropathy, focal segmental glomerulosclerosis, and IgA nephropathy.2 MPGN and mixed cryoglobulinemia are the most common. In the past, MPGN was classified as type I, II, and III.
The patient’s urine toxicology revealed recent amphetamine use, which can also lead to acute kidney injury through rhabdomyolysis or acute interstitial nephritis (AIN).3 In the cases of rhabdomyolysis, urinalysis would show positive heme without any red blood cell on microscopic analysis, which was not present in this case. AIN commonly manifests as acute kidney injury, pyuria, and proteinuria but without a decrease in complement levels.4 While the patient’s urine sediment included white blood cell (10/high-power field), the presence of microscopic hematuria, decreased complement levels, and proteinuria in the context of HCV positivity makes MPGN more likely than AIN.
Recently, there has been greater emphasis on using immunofluorescence for kidney biopsies. MPGN is now classified into 2 main categories: MPGN with mesangial immunoglobulins and C3 deposits in the capillary walls, and MPGN with C3 deposits but without Ig.5 MPGN with Ig-complement deposits is seen in autoimmune diseases and infections and is associated with dysproteinemias.
The renal biopsy in this patient was consistent with MPGN with immunofluorescence, a common finding in patients with infection. By synthesizing these data, we concluded that the patient represented a case of chronic HCV infection that led to MPGN with cryoglobulinemia. The normal C4 and negative RF do not suggest cryoglobulinemic crisis. Compensated cirrhosis was seen on imaging, pending liver biopsy.
Treatment
The management of MPGN secondary to HCV infection relies on the treatment of the underlying infection and clearance of viral load. Direct-acting antivirals have been used successfully in the treatment of HCV-associated MPGN. When cryoglobulinemia is present, immunosuppressive therapy is recommended. These regimens commonly include rituximab and steroids.5 Rituximab is also used for nephrotic syndrome associated with MPGN, as recommended in the 2018 Kidney Disease: Improving Global Outcomes guidelines.6
When initiating rituximab therapy in a patient who tests positive for hepatitis B (HBcAb positive or HBsAb positive), it is recommended to follow the established guidelines, which include treating them with entecavir for prophylaxis to prevent reactivation or a flare of hepatitis B.7 The patient in this case needed close follow-up in the nephrology and hepatology clinic. Immunosuppressive therapy was not pursued while the patient was admitted to the hospital due to instability with housing, transportation, and difficulty in ensuring close follow-up.
CONCLUSIONS
Clinicians should maintain a broad differential even in the face of confirmatory imaging and other objective findings. In the case of anasarca, nephrotic syndrome should be considered. Key causes of nephrotic syndromes include MPGN, membranous nephropathy, minimal change disease, and focal segmental glomerulosclerosis. MPGN is a histopathological diagnosis, and it is essential to identify if it is secondary to immune complexes or only complement mediated because Ig-complement deposits are seen in autoimmune disease and infection. The management of MPGN due to HCV infection relies on antiviral therapy. In the presence of cryoglobulinemia, immunosuppressive therapy is recommended.
- Tapper EB, Parikh ND. Diagnosis and management of cirrhosis and its complications: a review. JAMA. 2023;329(18):1589–1602. doi:10.1001/jama.2023.5997
- Ozkok A, Yildiz A. Hepatitis C virus associated glomerulopathies. World J Gastroenterol. 2014;20(24):7544-7554. doi:10.3748/wjg.v20.i24.7544
- Foley RJ, Kapatkin K, Vrani R, Weinman EJ. Amphetamineinduced acute renal failure. South Med J. 1984;77(2):258- 260. doi:10.1097/00007611-198402000-00035
- Rossert J. Drug - induced acute interstitial nephritis. Kidney Int. 2001;60(2):804-817. doi:10.1046/j.1523-1755.2001.060002804.x
- Sethi S, Fervenza FC. Membranoproliferative glomerulonephritis: pathogenetic heterogeneity and proposal for a new classification. Semin Nephrol. 2011;31(4):341-348. doi:10.1016/j.semnephrol.2011.06.005
- Jadoul M, Berenguer MC, Doss W, et al. Executive summary of the 2018 KDIGO hepatitis C in CKD guideline: welcoming advances in evaluation and management. Kidney Int. 2018;94(4):663-673. doi:10.1016/j.kint.2018.06.011
- Myint A, Tong MJ, Beaven SW. Reactivation of hepatitis b virus: a review of clinical guidelines. Clin Liver Dis (Hoboken). 2020;15(4):162-167. doi:10.1002/cld.883
Histology is the gold standard for cirrhosis diagnosis. However, a combination of clinical history, physical examination findings, and supportive laboratory and radiographic features is generally sufficient to make the diagnosis. Routine ultrasound and computed tomography (CT) imaging often identifies a nodular liver contour with sequelae of portal hypertension, including splenomegaly, varices, and ascites, which can suggest cirrhosis when supported by laboratory parameters and clinical features. As a result, the diagnosis is typically made clinically.1 Many patients with compensated cirrhosis go undetected. The presence of a decompensation event (ascites, spontaneous bacterial peritonitis, variceal hemorrhage, or hepatic encephalopathy) often leads to index diagnosis when patients were previously compensated. When a patient presents with suspected decompensated cirrhosis, it is important to consider other diagnoses with similar presentations and ensure that multiple disease processes are not contributing to the symptoms.
CASE PRESENTATION
A 64-year-old male with a history of intravenous (IV) methamphetamine use and prior incarceration presented with a 3-week history of progressively worsening generalized swelling. Prior to the onset of his symptoms, the patient injured his right lower extremity (RLE) in a bicycle accident, resulting in edema that progressed to bilateral lower extremity (BLE) edema and worsening fatigue, despite resolution of the initial injury. The patient gained weight though he could not quantify the amount. He experienced progressive hunger, thirst, and fatigue as well as increased sleep. Additionally, the patient experienced worsening dyspnea on exertion and orthopnea. He started using 2 pillows instead of 1 pillow at night.
The patient reported no fevers, chills, sputum production, chest pain, or paroxysmal nocturnal dyspnea. He had no known history of sexually transmitted infections, no significant history of alcohol use, and occasional tobacco and marijuana use. He had been incarcerated > 10 years before and last used IV methamphetamine 3 years before. He did not regularly take any medications.
The patient’s vital signs included a temperature of 98.2 °F; 78/min heart rate; 15/min respiratory rate; 159/109 mm Hg blood pressure; and 98% oxygen saturation on room air. He had gained 20 lbs in the past 4 months. He had pitting edema in both legs and arms, as well as periorbital swelling, but no jugular venous distention, abnormal heart sounds, or murmurs. Breath sounds were distant but clear to auscultation. His abdomen was distended with normal bowel sounds and no fluid wave; mild epigastric tenderness was present, but no intra-abdominal masses were palpated. He had spider angiomata on the upper chest but no other stigmata of cirrhosis, such as caput medusae or jaundice. Tattoos were noted.
Laboratory test results showed a platelet count of 178 x 103/μL (reference range, 140- 440 ~ 103μL).Creatinine was 0.80 mg/dL (reference range, < 1.28 mg/dL), with an estimated glomerular filtration rate (eGFR) of 99 mL/min/1.73 m2 using the Chronic Kidney Disease-Epidemiology equation (reference range, > 60 mL/min/1.73 m2), (reference range, > 60 mL/min/1.73 m2), and Cystatin C was 1.14 mg/L (reference range, < 1.15 mg/L). His electrolytes and complete blood count were within normal limits, including sodium, 134 mmol/L; potassium, 4.4 mmol/L; chloride, 108 mmol/L; and carbon dioxide, 22.5 mmol/L.
Additional test results included alkaline phosphatase, 126 U/L (reference range, < 94 U/L); alanine transaminase, 41 U/L (reference range, < 45 U/L); aspartate aminotransferase, 70 U/L (reference range, < 35 U/L); total bilirubin, 0.6 mg/dL (reference range, < 1 mg/dL); albumin, 1.8 g/dL (reference range, 3.2-4.8 g/dL); and total protein, 6.3 g/dL (reference range, 5.9-8.3 g/dL). The patient’s international normalized ratio was 0.96 (reference range, 0.8-1.1), and brain natriuretic peptide was normal at 56 pg/mL. No prior laboratory results were available for comparison.
Urine toxicology was positive for amphetamines. Urinalysis demonstrated large occult blood, with a red blood cell count of 26/ HPF (reference range, 0/HPF) and proteinuria (100 mg/dL; reference range, negative), without bacteria, nitrites, or leukocyte esterase. Urine white blood cell count was 10/ HPF (reference range, 0/HPF), and fine granular casts and hyaline casts were present.
A noncontrast CT of the abdomen and pelvis in the emergency department showed an irregular liver contour with diffuse nodularity, multiple portosystemic collaterals, moderate abdominal and pelvic ascites, small bilateral pleural effusions with associated atelectasis, and anasarca consistent with cirrhosis (Figure 1). The patient was admitted to the internal medicine service for workup and management of newly diagnosed cirrhosis.

Paracentesis revealed straw-colored fluid with an ascitic fluid neutrophil count of 17/μL, a protein level of < 3 g/dL and albumin level of < 1.5 g/dL. Gram stain of the ascitic fluid showed a moderate white blood cell count with no organisms. Fluid culture showed no microbial growth.
Initial workup for cirrhosis demonstrated a positive total hepatitis A antibody. The patient had a nonreactive hepatitis B surface antigen and surface antibody, but a reactive hepatitis B core antibody; a hepatitis B DNA level was not ordered. He had a reactive hepatitis C antibody with a viral load of 4,490,000 II/mL (genotype 1a). The patient’s iron level was 120 μg/dL, with a calculated total iron-binding capacity (TIBC) of 126.2 μg/dL. His transferrin saturation (TSAT) (serum iron divided by TIBC) was 95%. The patient had nonreactive antinuclear antibody and antimitochondrial antibody tests and a positive antismooth muscle antibody test with a titer of 1:40. His α-fetoprotein (AFP) level was 505 ng/mL (reference range, < 8 ng/mL).
Follow-up MRI of the abdomen and pelvis showed cirrhotic morphology with large volume ascites and portosystemic collaterals, consistent with portal hypertension. Additionally, it showed multiple scattered peripheral sub centimeter hyperenhancing foci, most likely representing benign lesions.
The patient's spot urine protein-creatinine ratio was 3.76. To better quantify proteinuria, a 24-hour urine collection was performed and revealed 12.8 g/d of urine protein (reference range, 0-0.17 g/d). His serum triglyceride level was 175 mg/dL (reference range, 40-60 mg/dL); total cholesterol was 177 mg/ dL (reference range, ≤ 200 mg/dL); low density lipoprotein cholesterol was 98 mg/ dL (reference range, ≤ 130 mg/dL); and highdensity lipoprotein cholesterol was 43.8 mg/ dL (reference range, ≥ 40 mg/dL); C3 complement level was 71 mg/dL (reference range, 82-185 mg/dL); and C4 complement level was 22 mg/dL (reference range, 15-53 mg/ dL). His rheumatoid factor was < 14 IU/mL. Tests for rapid plasma reagin and HIV antigen- antibody were nonreactive, and the phospholipase A2 receptor antibody test was negative. The patient tested positive for QuantiFERON-TB Gold and qualitative cryoglobulin, which indicated a cryocrit of 1%.
A renal biopsy was performed, revealing diffuse podocyte foot process effacement and glomerulonephritis with low-grade C3 and immunoglobulin (Ig) G deposits, consistent with early membranoproliferative glomerulonephritis (MPGN) (Figures 2 and 3).


The patient was initially diuresed with IV furosemide without significant urine output. He was then diuresed with IV 25% albumin (total, 25 g), followed by IV furosemide 40 mg twice daily, which led to significant urine output and resolution of his anasarca. Given the patient’s hypoalbuminemic state, IV albumin was necessary to deliver furosemide to the proximal tubule. He was started on lisinopril for renal protection and discharged with spironolactone and furosemide for fluid management in the context of cirrhosis.
The patient was evaluated by the Liver Nodule Clinic, which includes specialists from hepatology, medical oncology, radiation oncology, interventional radiology, and diagnostic radiology. The team considered the patient’s medical history and characteristics of the nodules on imaging. Notable aspects of the patient’s history included hepatitis C virus (HCV) infection and an elevated AFP level, although imaging showed no lesion concerning for malignancy. Given these findings, the patient was scheduled for a liver biopsy to establish a tissue diagnosis of cirrhosis. Hepatology, nephrology, and infectious disease specialists coordinated to plan the management and treatment of latent tuberculosis (TB), chronic HCV, MPGN, compensated cirrhosis, and suspicious liver lesions.
The patient chose to handle management and treatment as an outpatient. He was discharged with furosemide and spironolactone for anasarca management, and amlodipine and lisinopril for his hypertension and MPGN. Follow-up appointments were scheduled with infectious disease for management of latent TB and HCV, nephrology for MPGN, gastroenterology for cirrhosis, and interventional radiology for liver biopsy. Unfortunately, the patient was unhoused with limited access to transportation, which prevented timely follow-up. Given these social factors, immunosuppression was not started. Additionally, he did not start on HCV therapy because the viral load was still pending at time of discharge.
DISCUSSION
The diagnosis of decompensated cirrhosis was prematurely established, resulting in a diagnostic delay, a form of diagnostic error. However, on hospital day 2, the initial hypothesis of decompensated cirrhosis as the sole driver of the patient’s presentation was reconsidered due to the disconnect between the severity of hypoalbuminemia and diffuse edema (anasarca), and the absence of laboratory evidence of hepatic decompensation (normal international normalized ratio, bilirubin, and low but normal platelet count). Although image findings supported cirrhosis, laboratory markers did not indicate hepatic decompensation. The severity of hypoalbuminemia and anasarca, along with an indeterminate Serum-Ascites Albumin Gradient, prompted the patient’s care team to consider other causes, specifically, nephrotic syndrome.
The patien’s spot protein-to-creatinine ratio was 3.76 (reference range < 0.2 mg/mg creatinine), but a 24-hour urine protein collection was 12.8 g/day (reference range < 150 mg/day). While most spot urine protein- to-creatinine ratios (UPCR) correlate with a 24-hour urine collection, discrepancies can occur, as in this case. It is important to recognize that the spot UPCR assumes that patients are excreting 1000 mg of creatinine daily in their urine, which is not always the case. In addition, changes in urine osmolality can lead to different values. The gold standard for proteinuria is a 24-hour urine collection for protein and creatinine.
The patient’s nephrotic-range proteinuria and severe hypoalbuminemia are not solely explained by cirrhosis. In addition, the patient’s lower extremity edema pointed to nephrotic syndrome. The differential diagnosis for nephrotic syndrome includes both primary and secondary forms of membranous nephropathy, minimal change disease, focal segmental glomerulosclerosis, and MPGN, a histopathological diagnosis that requires distinguishing between immune complex-mediated and complement-mediated forms. Other causes of nephrotic syndrome that do not fit in any of these buckets include amyloidosis, IgA nephropathy, and diabetes mellitus (DM). Despite DM being a common cause of nephrotic range proteinuria, it rarely leads to full nephrotic syndrome.
When considering the diagnosis, we reframed the patient’s clinical syndrome as compensated cirrhosis plus nephrotic syndrome. This approach prioritized identifying a cause that could explain both cirrhosis (from any cause) leading to IgA nephropathy or injection drug use serving as a risk factor for cirrhosis and nephrotic syndrome through HCV or AA amyloidosis, respectively. This problem representation guided us to the correct diagnosis. There are multiple renal diseases associated with HCV infection, including MPGN, membranous nephropathy, focal segmental glomerulosclerosis, and IgA nephropathy.2 MPGN and mixed cryoglobulinemia are the most common. In the past, MPGN was classified as type I, II, and III.
The patient’s urine toxicology revealed recent amphetamine use, which can also lead to acute kidney injury through rhabdomyolysis or acute interstitial nephritis (AIN).3 In the cases of rhabdomyolysis, urinalysis would show positive heme without any red blood cell on microscopic analysis, which was not present in this case. AIN commonly manifests as acute kidney injury, pyuria, and proteinuria but without a decrease in complement levels.4 While the patient’s urine sediment included white blood cell (10/high-power field), the presence of microscopic hematuria, decreased complement levels, and proteinuria in the context of HCV positivity makes MPGN more likely than AIN.
Recently, there has been greater emphasis on using immunofluorescence for kidney biopsies. MPGN is now classified into 2 main categories: MPGN with mesangial immunoglobulins and C3 deposits in the capillary walls, and MPGN with C3 deposits but without Ig.5 MPGN with Ig-complement deposits is seen in autoimmune diseases and infections and is associated with dysproteinemias.
The renal biopsy in this patient was consistent with MPGN with immunofluorescence, a common finding in patients with infection. By synthesizing these data, we concluded that the patient represented a case of chronic HCV infection that led to MPGN with cryoglobulinemia. The normal C4 and negative RF do not suggest cryoglobulinemic crisis. Compensated cirrhosis was seen on imaging, pending liver biopsy.
Treatment
The management of MPGN secondary to HCV infection relies on the treatment of the underlying infection and clearance of viral load. Direct-acting antivirals have been used successfully in the treatment of HCV-associated MPGN. When cryoglobulinemia is present, immunosuppressive therapy is recommended. These regimens commonly include rituximab and steroids.5 Rituximab is also used for nephrotic syndrome associated with MPGN, as recommended in the 2018 Kidney Disease: Improving Global Outcomes guidelines.6
When initiating rituximab therapy in a patient who tests positive for hepatitis B (HBcAb positive or HBsAb positive), it is recommended to follow the established guidelines, which include treating them with entecavir for prophylaxis to prevent reactivation or a flare of hepatitis B.7 The patient in this case needed close follow-up in the nephrology and hepatology clinic. Immunosuppressive therapy was not pursued while the patient was admitted to the hospital due to instability with housing, transportation, and difficulty in ensuring close follow-up.
CONCLUSIONS
Clinicians should maintain a broad differential even in the face of confirmatory imaging and other objective findings. In the case of anasarca, nephrotic syndrome should be considered. Key causes of nephrotic syndromes include MPGN, membranous nephropathy, minimal change disease, and focal segmental glomerulosclerosis. MPGN is a histopathological diagnosis, and it is essential to identify if it is secondary to immune complexes or only complement mediated because Ig-complement deposits are seen in autoimmune disease and infection. The management of MPGN due to HCV infection relies on antiviral therapy. In the presence of cryoglobulinemia, immunosuppressive therapy is recommended.
Histology is the gold standard for cirrhosis diagnosis. However, a combination of clinical history, physical examination findings, and supportive laboratory and radiographic features is generally sufficient to make the diagnosis. Routine ultrasound and computed tomography (CT) imaging often identifies a nodular liver contour with sequelae of portal hypertension, including splenomegaly, varices, and ascites, which can suggest cirrhosis when supported by laboratory parameters and clinical features. As a result, the diagnosis is typically made clinically.1 Many patients with compensated cirrhosis go undetected. The presence of a decompensation event (ascites, spontaneous bacterial peritonitis, variceal hemorrhage, or hepatic encephalopathy) often leads to index diagnosis when patients were previously compensated. When a patient presents with suspected decompensated cirrhosis, it is important to consider other diagnoses with similar presentations and ensure that multiple disease processes are not contributing to the symptoms.
CASE PRESENTATION
A 64-year-old male with a history of intravenous (IV) methamphetamine use and prior incarceration presented with a 3-week history of progressively worsening generalized swelling. Prior to the onset of his symptoms, the patient injured his right lower extremity (RLE) in a bicycle accident, resulting in edema that progressed to bilateral lower extremity (BLE) edema and worsening fatigue, despite resolution of the initial injury. The patient gained weight though he could not quantify the amount. He experienced progressive hunger, thirst, and fatigue as well as increased sleep. Additionally, the patient experienced worsening dyspnea on exertion and orthopnea. He started using 2 pillows instead of 1 pillow at night.
The patient reported no fevers, chills, sputum production, chest pain, or paroxysmal nocturnal dyspnea. He had no known history of sexually transmitted infections, no significant history of alcohol use, and occasional tobacco and marijuana use. He had been incarcerated > 10 years before and last used IV methamphetamine 3 years before. He did not regularly take any medications.
The patient’s vital signs included a temperature of 98.2 °F; 78/min heart rate; 15/min respiratory rate; 159/109 mm Hg blood pressure; and 98% oxygen saturation on room air. He had gained 20 lbs in the past 4 months. He had pitting edema in both legs and arms, as well as periorbital swelling, but no jugular venous distention, abnormal heart sounds, or murmurs. Breath sounds were distant but clear to auscultation. His abdomen was distended with normal bowel sounds and no fluid wave; mild epigastric tenderness was present, but no intra-abdominal masses were palpated. He had spider angiomata on the upper chest but no other stigmata of cirrhosis, such as caput medusae or jaundice. Tattoos were noted.
Laboratory test results showed a platelet count of 178 x 103/μL (reference range, 140- 440 ~ 103μL).Creatinine was 0.80 mg/dL (reference range, < 1.28 mg/dL), with an estimated glomerular filtration rate (eGFR) of 99 mL/min/1.73 m2 using the Chronic Kidney Disease-Epidemiology equation (reference range, > 60 mL/min/1.73 m2), (reference range, > 60 mL/min/1.73 m2), and Cystatin C was 1.14 mg/L (reference range, < 1.15 mg/L). His electrolytes and complete blood count were within normal limits, including sodium, 134 mmol/L; potassium, 4.4 mmol/L; chloride, 108 mmol/L; and carbon dioxide, 22.5 mmol/L.
Additional test results included alkaline phosphatase, 126 U/L (reference range, < 94 U/L); alanine transaminase, 41 U/L (reference range, < 45 U/L); aspartate aminotransferase, 70 U/L (reference range, < 35 U/L); total bilirubin, 0.6 mg/dL (reference range, < 1 mg/dL); albumin, 1.8 g/dL (reference range, 3.2-4.8 g/dL); and total protein, 6.3 g/dL (reference range, 5.9-8.3 g/dL). The patient’s international normalized ratio was 0.96 (reference range, 0.8-1.1), and brain natriuretic peptide was normal at 56 pg/mL. No prior laboratory results were available for comparison.
Urine toxicology was positive for amphetamines. Urinalysis demonstrated large occult blood, with a red blood cell count of 26/ HPF (reference range, 0/HPF) and proteinuria (100 mg/dL; reference range, negative), without bacteria, nitrites, or leukocyte esterase. Urine white blood cell count was 10/ HPF (reference range, 0/HPF), and fine granular casts and hyaline casts were present.
A noncontrast CT of the abdomen and pelvis in the emergency department showed an irregular liver contour with diffuse nodularity, multiple portosystemic collaterals, moderate abdominal and pelvic ascites, small bilateral pleural effusions with associated atelectasis, and anasarca consistent with cirrhosis (Figure 1). The patient was admitted to the internal medicine service for workup and management of newly diagnosed cirrhosis.

Paracentesis revealed straw-colored fluid with an ascitic fluid neutrophil count of 17/μL, a protein level of < 3 g/dL and albumin level of < 1.5 g/dL. Gram stain of the ascitic fluid showed a moderate white blood cell count with no organisms. Fluid culture showed no microbial growth.
Initial workup for cirrhosis demonstrated a positive total hepatitis A antibody. The patient had a nonreactive hepatitis B surface antigen and surface antibody, but a reactive hepatitis B core antibody; a hepatitis B DNA level was not ordered. He had a reactive hepatitis C antibody with a viral load of 4,490,000 II/mL (genotype 1a). The patient’s iron level was 120 μg/dL, with a calculated total iron-binding capacity (TIBC) of 126.2 μg/dL. His transferrin saturation (TSAT) (serum iron divided by TIBC) was 95%. The patient had nonreactive antinuclear antibody and antimitochondrial antibody tests and a positive antismooth muscle antibody test with a titer of 1:40. His α-fetoprotein (AFP) level was 505 ng/mL (reference range, < 8 ng/mL).
Follow-up MRI of the abdomen and pelvis showed cirrhotic morphology with large volume ascites and portosystemic collaterals, consistent with portal hypertension. Additionally, it showed multiple scattered peripheral sub centimeter hyperenhancing foci, most likely representing benign lesions.
The patient's spot urine protein-creatinine ratio was 3.76. To better quantify proteinuria, a 24-hour urine collection was performed and revealed 12.8 g/d of urine protein (reference range, 0-0.17 g/d). His serum triglyceride level was 175 mg/dL (reference range, 40-60 mg/dL); total cholesterol was 177 mg/ dL (reference range, ≤ 200 mg/dL); low density lipoprotein cholesterol was 98 mg/ dL (reference range, ≤ 130 mg/dL); and highdensity lipoprotein cholesterol was 43.8 mg/ dL (reference range, ≥ 40 mg/dL); C3 complement level was 71 mg/dL (reference range, 82-185 mg/dL); and C4 complement level was 22 mg/dL (reference range, 15-53 mg/ dL). His rheumatoid factor was < 14 IU/mL. Tests for rapid plasma reagin and HIV antigen- antibody were nonreactive, and the phospholipase A2 receptor antibody test was negative. The patient tested positive for QuantiFERON-TB Gold and qualitative cryoglobulin, which indicated a cryocrit of 1%.
A renal biopsy was performed, revealing diffuse podocyte foot process effacement and glomerulonephritis with low-grade C3 and immunoglobulin (Ig) G deposits, consistent with early membranoproliferative glomerulonephritis (MPGN) (Figures 2 and 3).


The patient was initially diuresed with IV furosemide without significant urine output. He was then diuresed with IV 25% albumin (total, 25 g), followed by IV furosemide 40 mg twice daily, which led to significant urine output and resolution of his anasarca. Given the patient’s hypoalbuminemic state, IV albumin was necessary to deliver furosemide to the proximal tubule. He was started on lisinopril for renal protection and discharged with spironolactone and furosemide for fluid management in the context of cirrhosis.
The patient was evaluated by the Liver Nodule Clinic, which includes specialists from hepatology, medical oncology, radiation oncology, interventional radiology, and diagnostic radiology. The team considered the patient’s medical history and characteristics of the nodules on imaging. Notable aspects of the patient’s history included hepatitis C virus (HCV) infection and an elevated AFP level, although imaging showed no lesion concerning for malignancy. Given these findings, the patient was scheduled for a liver biopsy to establish a tissue diagnosis of cirrhosis. Hepatology, nephrology, and infectious disease specialists coordinated to plan the management and treatment of latent tuberculosis (TB), chronic HCV, MPGN, compensated cirrhosis, and suspicious liver lesions.
The patient chose to handle management and treatment as an outpatient. He was discharged with furosemide and spironolactone for anasarca management, and amlodipine and lisinopril for his hypertension and MPGN. Follow-up appointments were scheduled with infectious disease for management of latent TB and HCV, nephrology for MPGN, gastroenterology for cirrhosis, and interventional radiology for liver biopsy. Unfortunately, the patient was unhoused with limited access to transportation, which prevented timely follow-up. Given these social factors, immunosuppression was not started. Additionally, he did not start on HCV therapy because the viral load was still pending at time of discharge.
DISCUSSION
The diagnosis of decompensated cirrhosis was prematurely established, resulting in a diagnostic delay, a form of diagnostic error. However, on hospital day 2, the initial hypothesis of decompensated cirrhosis as the sole driver of the patient’s presentation was reconsidered due to the disconnect between the severity of hypoalbuminemia and diffuse edema (anasarca), and the absence of laboratory evidence of hepatic decompensation (normal international normalized ratio, bilirubin, and low but normal platelet count). Although image findings supported cirrhosis, laboratory markers did not indicate hepatic decompensation. The severity of hypoalbuminemia and anasarca, along with an indeterminate Serum-Ascites Albumin Gradient, prompted the patient’s care team to consider other causes, specifically, nephrotic syndrome.
The patien’s spot protein-to-creatinine ratio was 3.76 (reference range < 0.2 mg/mg creatinine), but a 24-hour urine protein collection was 12.8 g/day (reference range < 150 mg/day). While most spot urine protein- to-creatinine ratios (UPCR) correlate with a 24-hour urine collection, discrepancies can occur, as in this case. It is important to recognize that the spot UPCR assumes that patients are excreting 1000 mg of creatinine daily in their urine, which is not always the case. In addition, changes in urine osmolality can lead to different values. The gold standard for proteinuria is a 24-hour urine collection for protein and creatinine.
The patient’s nephrotic-range proteinuria and severe hypoalbuminemia are not solely explained by cirrhosis. In addition, the patient’s lower extremity edema pointed to nephrotic syndrome. The differential diagnosis for nephrotic syndrome includes both primary and secondary forms of membranous nephropathy, minimal change disease, focal segmental glomerulosclerosis, and MPGN, a histopathological diagnosis that requires distinguishing between immune complex-mediated and complement-mediated forms. Other causes of nephrotic syndrome that do not fit in any of these buckets include amyloidosis, IgA nephropathy, and diabetes mellitus (DM). Despite DM being a common cause of nephrotic range proteinuria, it rarely leads to full nephrotic syndrome.
When considering the diagnosis, we reframed the patient’s clinical syndrome as compensated cirrhosis plus nephrotic syndrome. This approach prioritized identifying a cause that could explain both cirrhosis (from any cause) leading to IgA nephropathy or injection drug use serving as a risk factor for cirrhosis and nephrotic syndrome through HCV or AA amyloidosis, respectively. This problem representation guided us to the correct diagnosis. There are multiple renal diseases associated with HCV infection, including MPGN, membranous nephropathy, focal segmental glomerulosclerosis, and IgA nephropathy.2 MPGN and mixed cryoglobulinemia are the most common. In the past, MPGN was classified as type I, II, and III.
The patient’s urine toxicology revealed recent amphetamine use, which can also lead to acute kidney injury through rhabdomyolysis or acute interstitial nephritis (AIN).3 In the cases of rhabdomyolysis, urinalysis would show positive heme without any red blood cell on microscopic analysis, which was not present in this case. AIN commonly manifests as acute kidney injury, pyuria, and proteinuria but without a decrease in complement levels.4 While the patient’s urine sediment included white blood cell (10/high-power field), the presence of microscopic hematuria, decreased complement levels, and proteinuria in the context of HCV positivity makes MPGN more likely than AIN.
Recently, there has been greater emphasis on using immunofluorescence for kidney biopsies. MPGN is now classified into 2 main categories: MPGN with mesangial immunoglobulins and C3 deposits in the capillary walls, and MPGN with C3 deposits but without Ig.5 MPGN with Ig-complement deposits is seen in autoimmune diseases and infections and is associated with dysproteinemias.
The renal biopsy in this patient was consistent with MPGN with immunofluorescence, a common finding in patients with infection. By synthesizing these data, we concluded that the patient represented a case of chronic HCV infection that led to MPGN with cryoglobulinemia. The normal C4 and negative RF do not suggest cryoglobulinemic crisis. Compensated cirrhosis was seen on imaging, pending liver biopsy.
Treatment
The management of MPGN secondary to HCV infection relies on the treatment of the underlying infection and clearance of viral load. Direct-acting antivirals have been used successfully in the treatment of HCV-associated MPGN. When cryoglobulinemia is present, immunosuppressive therapy is recommended. These regimens commonly include rituximab and steroids.5 Rituximab is also used for nephrotic syndrome associated with MPGN, as recommended in the 2018 Kidney Disease: Improving Global Outcomes guidelines.6
When initiating rituximab therapy in a patient who tests positive for hepatitis B (HBcAb positive or HBsAb positive), it is recommended to follow the established guidelines, which include treating them with entecavir for prophylaxis to prevent reactivation or a flare of hepatitis B.7 The patient in this case needed close follow-up in the nephrology and hepatology clinic. Immunosuppressive therapy was not pursued while the patient was admitted to the hospital due to instability with housing, transportation, and difficulty in ensuring close follow-up.
CONCLUSIONS
Clinicians should maintain a broad differential even in the face of confirmatory imaging and other objective findings. In the case of anasarca, nephrotic syndrome should be considered. Key causes of nephrotic syndromes include MPGN, membranous nephropathy, minimal change disease, and focal segmental glomerulosclerosis. MPGN is a histopathological diagnosis, and it is essential to identify if it is secondary to immune complexes or only complement mediated because Ig-complement deposits are seen in autoimmune disease and infection. The management of MPGN due to HCV infection relies on antiviral therapy. In the presence of cryoglobulinemia, immunosuppressive therapy is recommended.
- Tapper EB, Parikh ND. Diagnosis and management of cirrhosis and its complications: a review. JAMA. 2023;329(18):1589–1602. doi:10.1001/jama.2023.5997
- Ozkok A, Yildiz A. Hepatitis C virus associated glomerulopathies. World J Gastroenterol. 2014;20(24):7544-7554. doi:10.3748/wjg.v20.i24.7544
- Foley RJ, Kapatkin K, Vrani R, Weinman EJ. Amphetamineinduced acute renal failure. South Med J. 1984;77(2):258- 260. doi:10.1097/00007611-198402000-00035
- Rossert J. Drug - induced acute interstitial nephritis. Kidney Int. 2001;60(2):804-817. doi:10.1046/j.1523-1755.2001.060002804.x
- Sethi S, Fervenza FC. Membranoproliferative glomerulonephritis: pathogenetic heterogeneity and proposal for a new classification. Semin Nephrol. 2011;31(4):341-348. doi:10.1016/j.semnephrol.2011.06.005
- Jadoul M, Berenguer MC, Doss W, et al. Executive summary of the 2018 KDIGO hepatitis C in CKD guideline: welcoming advances in evaluation and management. Kidney Int. 2018;94(4):663-673. doi:10.1016/j.kint.2018.06.011
- Myint A, Tong MJ, Beaven SW. Reactivation of hepatitis b virus: a review of clinical guidelines. Clin Liver Dis (Hoboken). 2020;15(4):162-167. doi:10.1002/cld.883
- Tapper EB, Parikh ND. Diagnosis and management of cirrhosis and its complications: a review. JAMA. 2023;329(18):1589–1602. doi:10.1001/jama.2023.5997
- Ozkok A, Yildiz A. Hepatitis C virus associated glomerulopathies. World J Gastroenterol. 2014;20(24):7544-7554. doi:10.3748/wjg.v20.i24.7544
- Foley RJ, Kapatkin K, Vrani R, Weinman EJ. Amphetamineinduced acute renal failure. South Med J. 1984;77(2):258- 260. doi:10.1097/00007611-198402000-00035
- Rossert J. Drug - induced acute interstitial nephritis. Kidney Int. 2001;60(2):804-817. doi:10.1046/j.1523-1755.2001.060002804.x
- Sethi S, Fervenza FC. Membranoproliferative glomerulonephritis: pathogenetic heterogeneity and proposal for a new classification. Semin Nephrol. 2011;31(4):341-348. doi:10.1016/j.semnephrol.2011.06.005
- Jadoul M, Berenguer MC, Doss W, et al. Executive summary of the 2018 KDIGO hepatitis C in CKD guideline: welcoming advances in evaluation and management. Kidney Int. 2018;94(4):663-673. doi:10.1016/j.kint.2018.06.011
- Myint A, Tong MJ, Beaven SW. Reactivation of hepatitis b virus: a review of clinical guidelines. Clin Liver Dis (Hoboken). 2020;15(4):162-167. doi:10.1002/cld.883
Elusive Edema: A Case of Nephrotic Syndrome Mimicking Decompensated Cirrhosis
Elusive Edema: A Case of Nephrotic Syndrome Mimicking Decompensated Cirrhosis
What About Stolen Valor is Actually Illegal?
What About Stolen Valor is Actually Illegal?
Memorial Day is the most solemn of all American military commemorations. It is the day when we honor those who sacrificed their lives so that their fellow citizens could flourish in freedom. At 3 PM, a grateful nation is called to observe 2 minutes of silence in remembrance of the heroes who died in battle or of the wounds they sustained in combat. Communities across the country will carry out ceremonies, lining national cemeteries with flags, holding patriotic parades, and conducting spiritual observances.1
Sadly, almost as long as there has been a United States, there has been a parallel practice dishonoring the uniform and deceiving veterans and the public alike known as stolen valor. Stolen valor is a persistent, yet strange, psychological behavior: individuals who never served in the US Armed Forces claim they have done heroic deeds for which they often sustained serious injuries in the line of duty and almost always won medals for their heroism.2 This editorial will trace the US legal history of stolen valor cases to provide the background for next month’s editorial examining its clinical and ethical aspects.
While many cases of stolen valor do not receive media attention, the experience of Sarah Cavanaugh, a former VA social worker who claimed to be a marine veteran who served in Iraq and Afghanistan, was the subject of the Deep Cover podcast series.3 Cavanaugh had claimed that an improvised explosive device blew up her Humvee, crushing her hip. Still she somehow was able to help her fellow Marines and earned the Bronze Star among other decorations for her heroism. That was not the only lie Cavanaugh told: she also told her friends and wife that she had advanced lung cancer due to burn pit exposure. In line with the best-worst of those who have stolen valor, her mastery of manipulation enabled her to become the commander of a local Veterans of Foreign Wars post. Using stolen identities and fraudulent documents, Cavanaugh was able to purloin veteran benefits, donated leave from other VA employees and money, and stole goods and services from various charitable organizations whose mission was to help wounded veterans and those struggling to adjust to civilian life. Before law enforcement unraveled her sordid tale, she misappropriated hundreds of thousands of dollars in VA benefits and donations and exploited dozens of generous veterans and compassionate civilians.4
Cavanaugh’s story was so sordidly compelling that I kept saying out loud to myself (and my spouse), “This has to be illegal.” The truth about stolen valor law is far more ambivalent and frustrating than I had anticipated or wanted. The first insult to my sense of justice was that lying about military service is not in itself illegal: you can pad your military resume with unearned decorations or impress a future partner or employer with your combat exploits without much fear of legal repercussions. The legal history of attempting to make stealing valor a crime has almost as many twists and turns as the fallacious narratives of military imposters and illustrates the uniquely American experiment in balancing freedom and fairness.
The Stolen Valor Act of 2005 made it a federal misdemeanor to wear, manufacture, or sell military decorations, or medals (Cavanaugh bought her medals online) without legal authorization. It also made it a crime to falsely represent oneself as having been the recipient of a decoration, medical, or service badge that Congress or the Armed Forces authorized. There were even stiffer penalties if the medal was a Silver Star, Distinguished Service Cross, US Air Force or US Navy Cross, or Purple Heart. Punishments include fines and imprisonment. The stated legislative purpose was to prohibit fraud that devalued military awards and the dignity of those who legitimately earned them.5
Next comes a distinctly American reaction to the initial Congressional attempt to protect the legacy of those who served—a lawsuit. Xavier Alvarez was an official on a California district water board claimed to be a 25-year veteran of the US Marine Corps wounded in combat and received the Congressional Medal of Honor. The Federal Bureau of Investigation exposed the lie and instead of the nation’s highest honor, Alvarez was the first to be convicted under the Stolen Valor Act of 2005. Alvarez appealed the decision, ironically claiming the law violated his free speech rights. The case landed in the Supreme Court, which ruled that the Stolen Valor Act did indeed violate the Free Speech Clause of the First Amendment. The majority opinion found the Act as passed was too encompassing of all speech and needed to target only cases in which false statements resulted in actual harm.6
The Stolen Valor Act of 2013 amends the criminal code regarding fraudulent claims about military service to include those who don’t only lie but also profit from it, as Cavanaugh did. The revised act specifically focuses on individuals who claim to have earned military honors for the intended purpose of obtaining money, property, or any other tangible benefit.7
Despite the complicated nature of Stolen Valor Law, it did prevail in Cavanaugh’s case. A US District Court Judge in Rhode Island found her guilty of stolen valor in all its permutations, along with identity theft of other veterans’ military and medical records and fraud in obtaining benefits and services intended for real veterans. Cavanaugh was sentenced to 70 months in federal prison, 3 years of supervised release, ordered to pay $284,796.82 in restitution, and to restore 261 hours of donated leave to the federal government, charitable organizations, and good Samaritans she duped and swindled.8
The revised law under which Cavanaugh was punished lasted 10 years until another classically American ethical concern—privacy—motivated additional legislative effort. A 2023/2024 US House of Representatives proposal to amend the Stolen Valor Act would have strengthened the privacy protections afforded military records. It would have required the information to only be accessed with the permission of the individual who served or their family or through a Freedom of Information Act request. This would make the kind of journalistic and law enforcement investigations that eventually caught Cavanaugh in her lies far more laborious for false valor hunters while at the same time preventing unscrupulous inquiries into service members’ personal information. Advocates for free speech and defenders of military honor are both lobbying Congress; as of this writing the legislation has not been passed.9
As we close part 1 of this review of stolen valor, we return to Memorial Day. This day provides the somber recognition that without the brave men and women of integrity who died in defense of a democracy that promotes the political activity of its citizens, we would not even be able to have this debate over justice, freedom, and truth.
- US Department of Veterans Affairs. The difference between Veterans Day and Memorial Day. October 30, 2023. Accessed May 27, 2025. https://news.va.gov/125549/difference-between-veterans-day-memorial-day/
- Home of Heroes. Stolen valor. Accessed May 27, 2025. https://homeofheroes.com/stolen-valor
- Halpern J. Deep cover: the truth about Sarah. May 2025. Accessed May 27, 2025. https://www.pushkin.fm/podcasts/deep-cover
- Stillwell B. The latest season of the ‘deep cover’ podcast dives into one of the biggest stolen valor cases ever. Military. com. May 22, 2025. Accessed May 27, 2025. https:// www.military.com/off-duty/2025/05/22/latest-season-of-deep-cover-podcast-dives-one-of-biggest-stolen-valor-cases-ever.html
- The Stolen Valor Act of 2005. Pub L No: 109-437. 120 Stat 3266
- Alvarez v United States. 567 US 2012.
- The Stolen Valor Act of 2013. 18 USC § 704(b)
- US Attorney’s Office, District of Rhode Island. Rhode Island woman sentenced to federal prison for falsifying military service; false use of military medals; identify theft, and fraudulently collecting more than $250,000, in veteran benefits and charitable contributions. March 14, 2023. Accessed May 27, 2025. https://www.justice.gov/usao-ri/pr/rhode-island-woman-sentenced-federal-prison-falsifying-military-service-false-use
- Armed Forces Benefit Association. Stolen Valor Act: all you need to know. February 21, 2024. Accessed May 27, 2025. https://www.afba.com/military-life/active-duty-and-veterans/stolen-valor-act-all-you-need-to-know/
Memorial Day is the most solemn of all American military commemorations. It is the day when we honor those who sacrificed their lives so that their fellow citizens could flourish in freedom. At 3 PM, a grateful nation is called to observe 2 minutes of silence in remembrance of the heroes who died in battle or of the wounds they sustained in combat. Communities across the country will carry out ceremonies, lining national cemeteries with flags, holding patriotic parades, and conducting spiritual observances.1
Sadly, almost as long as there has been a United States, there has been a parallel practice dishonoring the uniform and deceiving veterans and the public alike known as stolen valor. Stolen valor is a persistent, yet strange, psychological behavior: individuals who never served in the US Armed Forces claim they have done heroic deeds for which they often sustained serious injuries in the line of duty and almost always won medals for their heroism.2 This editorial will trace the US legal history of stolen valor cases to provide the background for next month’s editorial examining its clinical and ethical aspects.
While many cases of stolen valor do not receive media attention, the experience of Sarah Cavanaugh, a former VA social worker who claimed to be a marine veteran who served in Iraq and Afghanistan, was the subject of the Deep Cover podcast series.3 Cavanaugh had claimed that an improvised explosive device blew up her Humvee, crushing her hip. Still she somehow was able to help her fellow Marines and earned the Bronze Star among other decorations for her heroism. That was not the only lie Cavanaugh told: she also told her friends and wife that she had advanced lung cancer due to burn pit exposure. In line with the best-worst of those who have stolen valor, her mastery of manipulation enabled her to become the commander of a local Veterans of Foreign Wars post. Using stolen identities and fraudulent documents, Cavanaugh was able to purloin veteran benefits, donated leave from other VA employees and money, and stole goods and services from various charitable organizations whose mission was to help wounded veterans and those struggling to adjust to civilian life. Before law enforcement unraveled her sordid tale, she misappropriated hundreds of thousands of dollars in VA benefits and donations and exploited dozens of generous veterans and compassionate civilians.4
Cavanaugh’s story was so sordidly compelling that I kept saying out loud to myself (and my spouse), “This has to be illegal.” The truth about stolen valor law is far more ambivalent and frustrating than I had anticipated or wanted. The first insult to my sense of justice was that lying about military service is not in itself illegal: you can pad your military resume with unearned decorations or impress a future partner or employer with your combat exploits without much fear of legal repercussions. The legal history of attempting to make stealing valor a crime has almost as many twists and turns as the fallacious narratives of military imposters and illustrates the uniquely American experiment in balancing freedom and fairness.
The Stolen Valor Act of 2005 made it a federal misdemeanor to wear, manufacture, or sell military decorations, or medals (Cavanaugh bought her medals online) without legal authorization. It also made it a crime to falsely represent oneself as having been the recipient of a decoration, medical, or service badge that Congress or the Armed Forces authorized. There were even stiffer penalties if the medal was a Silver Star, Distinguished Service Cross, US Air Force or US Navy Cross, or Purple Heart. Punishments include fines and imprisonment. The stated legislative purpose was to prohibit fraud that devalued military awards and the dignity of those who legitimately earned them.5
Next comes a distinctly American reaction to the initial Congressional attempt to protect the legacy of those who served—a lawsuit. Xavier Alvarez was an official on a California district water board claimed to be a 25-year veteran of the US Marine Corps wounded in combat and received the Congressional Medal of Honor. The Federal Bureau of Investigation exposed the lie and instead of the nation’s highest honor, Alvarez was the first to be convicted under the Stolen Valor Act of 2005. Alvarez appealed the decision, ironically claiming the law violated his free speech rights. The case landed in the Supreme Court, which ruled that the Stolen Valor Act did indeed violate the Free Speech Clause of the First Amendment. The majority opinion found the Act as passed was too encompassing of all speech and needed to target only cases in which false statements resulted in actual harm.6
The Stolen Valor Act of 2013 amends the criminal code regarding fraudulent claims about military service to include those who don’t only lie but also profit from it, as Cavanaugh did. The revised act specifically focuses on individuals who claim to have earned military honors for the intended purpose of obtaining money, property, or any other tangible benefit.7
Despite the complicated nature of Stolen Valor Law, it did prevail in Cavanaugh’s case. A US District Court Judge in Rhode Island found her guilty of stolen valor in all its permutations, along with identity theft of other veterans’ military and medical records and fraud in obtaining benefits and services intended for real veterans. Cavanaugh was sentenced to 70 months in federal prison, 3 years of supervised release, ordered to pay $284,796.82 in restitution, and to restore 261 hours of donated leave to the federal government, charitable organizations, and good Samaritans she duped and swindled.8
The revised law under which Cavanaugh was punished lasted 10 years until another classically American ethical concern—privacy—motivated additional legislative effort. A 2023/2024 US House of Representatives proposal to amend the Stolen Valor Act would have strengthened the privacy protections afforded military records. It would have required the information to only be accessed with the permission of the individual who served or their family or through a Freedom of Information Act request. This would make the kind of journalistic and law enforcement investigations that eventually caught Cavanaugh in her lies far more laborious for false valor hunters while at the same time preventing unscrupulous inquiries into service members’ personal information. Advocates for free speech and defenders of military honor are both lobbying Congress; as of this writing the legislation has not been passed.9
As we close part 1 of this review of stolen valor, we return to Memorial Day. This day provides the somber recognition that without the brave men and women of integrity who died in defense of a democracy that promotes the political activity of its citizens, we would not even be able to have this debate over justice, freedom, and truth.
Memorial Day is the most solemn of all American military commemorations. It is the day when we honor those who sacrificed their lives so that their fellow citizens could flourish in freedom. At 3 PM, a grateful nation is called to observe 2 minutes of silence in remembrance of the heroes who died in battle or of the wounds they sustained in combat. Communities across the country will carry out ceremonies, lining national cemeteries with flags, holding patriotic parades, and conducting spiritual observances.1
Sadly, almost as long as there has been a United States, there has been a parallel practice dishonoring the uniform and deceiving veterans and the public alike known as stolen valor. Stolen valor is a persistent, yet strange, psychological behavior: individuals who never served in the US Armed Forces claim they have done heroic deeds for which they often sustained serious injuries in the line of duty and almost always won medals for their heroism.2 This editorial will trace the US legal history of stolen valor cases to provide the background for next month’s editorial examining its clinical and ethical aspects.
While many cases of stolen valor do not receive media attention, the experience of Sarah Cavanaugh, a former VA social worker who claimed to be a marine veteran who served in Iraq and Afghanistan, was the subject of the Deep Cover podcast series.3 Cavanaugh had claimed that an improvised explosive device blew up her Humvee, crushing her hip. Still she somehow was able to help her fellow Marines and earned the Bronze Star among other decorations for her heroism. That was not the only lie Cavanaugh told: she also told her friends and wife that she had advanced lung cancer due to burn pit exposure. In line with the best-worst of those who have stolen valor, her mastery of manipulation enabled her to become the commander of a local Veterans of Foreign Wars post. Using stolen identities and fraudulent documents, Cavanaugh was able to purloin veteran benefits, donated leave from other VA employees and money, and stole goods and services from various charitable organizations whose mission was to help wounded veterans and those struggling to adjust to civilian life. Before law enforcement unraveled her sordid tale, she misappropriated hundreds of thousands of dollars in VA benefits and donations and exploited dozens of generous veterans and compassionate civilians.4
Cavanaugh’s story was so sordidly compelling that I kept saying out loud to myself (and my spouse), “This has to be illegal.” The truth about stolen valor law is far more ambivalent and frustrating than I had anticipated or wanted. The first insult to my sense of justice was that lying about military service is not in itself illegal: you can pad your military resume with unearned decorations or impress a future partner or employer with your combat exploits without much fear of legal repercussions. The legal history of attempting to make stealing valor a crime has almost as many twists and turns as the fallacious narratives of military imposters and illustrates the uniquely American experiment in balancing freedom and fairness.
The Stolen Valor Act of 2005 made it a federal misdemeanor to wear, manufacture, or sell military decorations, or medals (Cavanaugh bought her medals online) without legal authorization. It also made it a crime to falsely represent oneself as having been the recipient of a decoration, medical, or service badge that Congress or the Armed Forces authorized. There were even stiffer penalties if the medal was a Silver Star, Distinguished Service Cross, US Air Force or US Navy Cross, or Purple Heart. Punishments include fines and imprisonment. The stated legislative purpose was to prohibit fraud that devalued military awards and the dignity of those who legitimately earned them.5
Next comes a distinctly American reaction to the initial Congressional attempt to protect the legacy of those who served—a lawsuit. Xavier Alvarez was an official on a California district water board claimed to be a 25-year veteran of the US Marine Corps wounded in combat and received the Congressional Medal of Honor. The Federal Bureau of Investigation exposed the lie and instead of the nation’s highest honor, Alvarez was the first to be convicted under the Stolen Valor Act of 2005. Alvarez appealed the decision, ironically claiming the law violated his free speech rights. The case landed in the Supreme Court, which ruled that the Stolen Valor Act did indeed violate the Free Speech Clause of the First Amendment. The majority opinion found the Act as passed was too encompassing of all speech and needed to target only cases in which false statements resulted in actual harm.6
The Stolen Valor Act of 2013 amends the criminal code regarding fraudulent claims about military service to include those who don’t only lie but also profit from it, as Cavanaugh did. The revised act specifically focuses on individuals who claim to have earned military honors for the intended purpose of obtaining money, property, or any other tangible benefit.7
Despite the complicated nature of Stolen Valor Law, it did prevail in Cavanaugh’s case. A US District Court Judge in Rhode Island found her guilty of stolen valor in all its permutations, along with identity theft of other veterans’ military and medical records and fraud in obtaining benefits and services intended for real veterans. Cavanaugh was sentenced to 70 months in federal prison, 3 years of supervised release, ordered to pay $284,796.82 in restitution, and to restore 261 hours of donated leave to the federal government, charitable organizations, and good Samaritans she duped and swindled.8
The revised law under which Cavanaugh was punished lasted 10 years until another classically American ethical concern—privacy—motivated additional legislative effort. A 2023/2024 US House of Representatives proposal to amend the Stolen Valor Act would have strengthened the privacy protections afforded military records. It would have required the information to only be accessed with the permission of the individual who served or their family or through a Freedom of Information Act request. This would make the kind of journalistic and law enforcement investigations that eventually caught Cavanaugh in her lies far more laborious for false valor hunters while at the same time preventing unscrupulous inquiries into service members’ personal information. Advocates for free speech and defenders of military honor are both lobbying Congress; as of this writing the legislation has not been passed.9
As we close part 1 of this review of stolen valor, we return to Memorial Day. This day provides the somber recognition that without the brave men and women of integrity who died in defense of a democracy that promotes the political activity of its citizens, we would not even be able to have this debate over justice, freedom, and truth.
- US Department of Veterans Affairs. The difference between Veterans Day and Memorial Day. October 30, 2023. Accessed May 27, 2025. https://news.va.gov/125549/difference-between-veterans-day-memorial-day/
- Home of Heroes. Stolen valor. Accessed May 27, 2025. https://homeofheroes.com/stolen-valor
- Halpern J. Deep cover: the truth about Sarah. May 2025. Accessed May 27, 2025. https://www.pushkin.fm/podcasts/deep-cover
- Stillwell B. The latest season of the ‘deep cover’ podcast dives into one of the biggest stolen valor cases ever. Military. com. May 22, 2025. Accessed May 27, 2025. https:// www.military.com/off-duty/2025/05/22/latest-season-of-deep-cover-podcast-dives-one-of-biggest-stolen-valor-cases-ever.html
- The Stolen Valor Act of 2005. Pub L No: 109-437. 120 Stat 3266
- Alvarez v United States. 567 US 2012.
- The Stolen Valor Act of 2013. 18 USC § 704(b)
- US Attorney’s Office, District of Rhode Island. Rhode Island woman sentenced to federal prison for falsifying military service; false use of military medals; identify theft, and fraudulently collecting more than $250,000, in veteran benefits and charitable contributions. March 14, 2023. Accessed May 27, 2025. https://www.justice.gov/usao-ri/pr/rhode-island-woman-sentenced-federal-prison-falsifying-military-service-false-use
- Armed Forces Benefit Association. Stolen Valor Act: all you need to know. February 21, 2024. Accessed May 27, 2025. https://www.afba.com/military-life/active-duty-and-veterans/stolen-valor-act-all-you-need-to-know/
- US Department of Veterans Affairs. The difference between Veterans Day and Memorial Day. October 30, 2023. Accessed May 27, 2025. https://news.va.gov/125549/difference-between-veterans-day-memorial-day/
- Home of Heroes. Stolen valor. Accessed May 27, 2025. https://homeofheroes.com/stolen-valor
- Halpern J. Deep cover: the truth about Sarah. May 2025. Accessed May 27, 2025. https://www.pushkin.fm/podcasts/deep-cover
- Stillwell B. The latest season of the ‘deep cover’ podcast dives into one of the biggest stolen valor cases ever. Military. com. May 22, 2025. Accessed May 27, 2025. https:// www.military.com/off-duty/2025/05/22/latest-season-of-deep-cover-podcast-dives-one-of-biggest-stolen-valor-cases-ever.html
- The Stolen Valor Act of 2005. Pub L No: 109-437. 120 Stat 3266
- Alvarez v United States. 567 US 2012.
- The Stolen Valor Act of 2013. 18 USC § 704(b)
- US Attorney’s Office, District of Rhode Island. Rhode Island woman sentenced to federal prison for falsifying military service; false use of military medals; identify theft, and fraudulently collecting more than $250,000, in veteran benefits and charitable contributions. March 14, 2023. Accessed May 27, 2025. https://www.justice.gov/usao-ri/pr/rhode-island-woman-sentenced-federal-prison-falsifying-military-service-false-use
- Armed Forces Benefit Association. Stolen Valor Act: all you need to know. February 21, 2024. Accessed May 27, 2025. https://www.afba.com/military-life/active-duty-and-veterans/stolen-valor-act-all-you-need-to-know/
What About Stolen Valor is Actually Illegal?
What About Stolen Valor is Actually Illegal?
The Use of Lung Cancer Screening to Increase Chronic Obstructive Pulmonary Disease Diagnosis in Veterans Affairs Primary Care
The Use of Lung Cancer Screening to Increase Chronic Obstructive Pulmonary Disease Diagnosis in Veterans Affairs Primary Care
Primary care practitioners (PCPs) in the US Department of Veterans Affairs (VA) provide care for patients with higher rates of many diseases—diabetes, heart disease, cancer, chronic obstructive pulmonary disease (COPD), and stroke—compared to the nonveteran population. 1 Due to the medical complexities of these diseases, they are often misdiagnosed or not diagnosed at all.
COPD is hiding in plain sight, impacting quality of life and burdening US health care systems.2 Research has yielded new treatments and evidence-based guidelines; however, COPD remains underdiagnosed. Only 13 million of the estimated 79 million US adults with COPD aged 20 to 79 years have been formally diagnosed.3 By the time patients are diagnosed, the disease is often advanced, and therapies are less effective. In 2 large studies of patients with COPD symptoms, later diagnosis was associated with worse outcomes.4,5
Veterans have a higher prevalence of COPD (8%-19%) than nonveterans (6%), likely due to higher rates of smoking and service-related exposures, especially among veterans of post-9/11 conflicts.6,7 Veterans do not always report symptoms and PCPs may not ask about symptoms, leading to underdiagnosis.8 The combination of high likelihood and underdetection of COPD presents a challenge and a target for VA quality improvement (QI).
The US Preventive Services Task Force (USPSTF) recommends against screening asymptomatic patients for COPD. However, both the USPSTF and the Global Initiative for Chronic Obstructive Lung Disease Report advocate for active case finding in primary care clinics to determine whether high-risk patients, such as smokers, experience COPD symptoms and warrant spirometry. 9,10 To make early COPD diagnoses, clinicians may use questionnaires alone or in combination with handheld peak expiratory flow rate measurements.11,12 Formal spirometry, considered the gold standard for COPD diagnosis, is ordered for patients who report COPD symptoms (ie, shortness of breath with exertion) or who have both COPD symptoms and reduced peak flow rates.
A systematic review and meta-analysis found that while the combination of questionnaires and peak flows was the more effective strategy overall, questionnaires alone were also valuable for identifying patients with possible COPD.13 Implementation of either screening method in primary care practices would be challenging. In a simulation study that applied chronic disease and preventive care guidelines to hypothetical patient panels, the time required for PCPs to provide guideline-recommended chronic and preventive care in addition to acute care far exceeded 8 hours per day, even in team-based settings.14 Overburdened PCPs are therefore unlikely to accept additional tasks like COPD case finding.
Why don’t patients report their pulmonary symptoms? Patients may not recognize the symptoms as evidence of COPD. Others may be afraid of a COPD diagnosis or the stigma that is associated with it.15 Perhaps they believe COPD treatment is ineffective because of lung damage from smoking. Some patients may not want to know if they have COPD, while others reduce activity levels to avoid symptoms.16
QUALITY IMPROVEMENT PROJECT
Given the high prevalence of COPD among veterans and the potential for underdiagnosis, VA Northeast Ohio Healthcare System (VANEOHS) internal medicine residents and faculty assessed the state of COPD diagnosis in its primary care clinic with a QI project in 2022. Patients in the clinic between August 1, 2015, and November 30, 2022, with an International Classification of Diseases-10 (ICD-10) COPD diagnosis code (J44) in the electronic health record were included. Of 157 included patients, 105 patients who had prior spirometry testing were excluded. Of the 52 patients with diagnosed COPD and no spirometry testing, 30 patients had computed tomography (CT) findings consistent with COPD (ie, airway thickening, emphysema, air trapping) that was performed for CT lung cancer screening (LCS).17 Twenty-three of these 30 patients were contacted by phone. All 23 were ever smokers and 13 reported COPD symptoms. The PCPs of the symptomatic patients were then contacted. Spirometry was ordered for all 13 patients and completed by 7. Three spirometry tests confirmed the COPD diagnosis. One PCP initiated inhaler therapy for a patient with newly diagnosed COPD.
All 11 PCPs of symptomatic patients were interviewed (many had > 1 symptomatic patient). They reported being unaware of patients’ COPD symptoms because the patients did not mention them, noting that screening for COPD was not a priority.
Role of Lung Cancer Screening
VA PCPs use electronic health record clinical reminders to track tests, consults, chronic disease education, cancer screenings, and routine health maintenance. A clinical reminder already exists (based on USPSTF recommendations) for LCS for patients aged 50 to 80 years who have a smoking history of 20 pack years. Patients who meet these criteria would also be considered high risk for COPD.
The VANEOHS QI project suggests that previously undiagnosed patients with findings of COPD on LCS may also have symptoms of COPD. Therefore, we wondered whether the LCS clinical reminder could serve a second purpose by prompting PCPs to ask veterans who meet LCS criteria about their COPD symptoms.
In 2022, about 13 million patients were eligible for LCS.18 Patients who qualify for LCS are at high risk for other cardiopulmonary disorders, such as COPD and coronary artery disease. Lung cancer is detected in only 1% of patients screened with CT at baseline. However, more often LCS yields evidence of additional cardiopulmonary disorders, such as emphysema or coronary artery calcifications. The International Early Lung Cancer Program (I-ELCAP) and the National Lung Cancer Screening Trial (NLST), which included > 79,000 patients, found evidence of emphysema on CT imaging in 24% and 31% of cases, respectively.19,20 In both cohorts, > 80% of patients with emphysema on CT imaging had no prior history of COPD.
In a 2022 article summarizing the potential impact of CT LCS on COPD diagnosis, Mulshine et al suggest that detection of emphysema on CT LCS provides “earlier recognition for PCPs to identify patients who would benefit from detailed symptom screening to prompt spirometry for COPD detection” and additional motivation for tobacco cessation.21 The VANEOHS QI project was developed and implemented prior to I-ELCAP or NLST reporting results but reinforces the value of CT LCS for COPD diagnosis.
Early diagnosis of COPD remains challenging because PCPs do not ask, patients do not tell, and symptoms can easily be dismissed. However, earlier diagnosis of COPD in symptomatic patients improves outcomes.3,4 To bridge this gap, VA PCPs and primary care patient aligned care teams (PACTs) need to commit to probing high-risk patients for COPD symptoms and ordering spirometry for those who are symptomatic. To accomplish this task, primary care teams need help.
The VANEOHS QI project confirmed that some patients with evidence of COPD on CT have symptoms of COPD that they did not share with their PCPs and suggests that LCS can be used as a dual action case finding method to screen both for lung cancer and COPD. We propose that patients who are eligible for LCS should also be probed for COPD symptoms at their clinic visits; for symptomatic patients, spirometry should be ordered, and COPD evidence-based management should be initiated when spirometry results are consistent with COPD. Annual probing for COPD symptoms could be considered in asymptomatic patients with ongoing tobacco use or emphysema on CT, since they may develop symptoms in the future. This new case-finding method bypasses the need for time-prohibitive questionnaires or peak flow measurements.
Future Opportunities
VA PCPs juggle many priorities and despite the simplicity of this new case finding COPD method, it may be unintentionally overlooked. PCPs often run out of time or may forget to ask patients about COPD symptoms when ordering LCS.
Future innovations to increase COPD diagnosis could include the creation of a yearly VA clinical reminder linked to the tobacco use reminder that has check boxes asking about symptoms of COPD in current and prior smokers. If patients have COPD symptoms, the reminder can prompt the ordering of spirometry. Similar reminders could be implemented to identify veterans with exposures to burn pits or other military environmental exposures who may have COPD symptoms. Another possible way to increase COPD diagnosis would be a partnership between primary care and the VA LCS program where patients receiving screening are asked about COPD symptoms during their LCS interviews and PACTs are alerted to order spirometry for symptomatic patients.
Elusive no longer! We can pull the veil back on COPD diagnosis and identify patients with possible COPD earlier in their course using their eligibility for LCS as a yearly reminder to probe them for symptoms. While not all patients who undergo LCS—even those with evidence of COPD on CT—will have COPD symptoms, symptoms may develop over time. LCS provides the possibility of 2 diagnoses from 1 test. This is an opportunity we cannot afford to miss.
- Betancourt JA, Granados PS, Pacheco GJ, et al. Exploring health outcomes for U.S. veterans compared to non-veterans from 2003 to 2019. Healthcare (Basel). 2021;9(5):604. doi:10.3390/healthcare90506064
- Bamonti PM, Fischer I, Moye J, Poghosyan H, Pietrzak RH. Obstructive respiratory disease in U.S. veterans: prevalence, characteristics, and health burden. J Psychiatr Res. 2024;176:140-147. doi:10.1016/j.jpsychires.2024.05.053
- Criner RN, Han MK. COPD care in the 21st century: a public health priority. Respir Care. 2018;63(5):591-600. doi:10.4187/respcare.06276
- Larsson K, Janson C, Ställberg B, et al. Impact of COPD diagnosis timing on clinical and economic outcomes: the ARCTIC observational cohort study. Int J Chron Obstruct Pulmon Dis. 2019;14:995-1008. doi:10.2147/COPD.S195382
- Kostikas K, Price D, Gutzwiller FS, et al. Clinical impact and healthcare resource utilization associated with early versus late COPD diagnosis in patients from UK CPRD Database. Int J Chron Obstruct Pulmon Dis. 2020;15:1729- 1738. doi:10.2147/COPD.S255414
- Bamonti PM, Robinson SA, Wan ES, Moy ML. Improving physiological, physical, and psychological health outcomes: a narrative review in US veterans with COPD. Int J Chron Obstruct Pulmon Dis. 2022;17:1269-1283. doi:10.2147/COPD.S339323
- Savitz DA, Woskie SR, Bello A, et al. Deployment to military bases with open burn pits and respiratory and cardiovascular disease. JAMA Netw Open. 2024;7(4):e247629. doi:10.1001/jamanetworkopen.2024.7629
- Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
- Guirguis-Blake JM, Senger CA, Webber EM, Mularski RA, Whitlock EP. Screening for chronic obstructive pulmonary disease: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2016;315(13):1378-1393. doi:10.1001/jama.2016.2654
- Capriotti T, Tomy R, Morales M. COPD updates: 2023 GOLD Report for primary care providers. Clinical Advisor. May 9, 2023. Accessed May 14, 2025. https://www.clinicaladvisor.com/features/copd-updates-2023-gold-report-primary-care/
- Leidy NK, Martinez FJ, Malley KG, et al. Can CAPTURE be used to identify undiagnosed patients with mild- to- moderate COPD likely to benefit from treatment? Int J Chron Obstruct Pulmon Dis. 2018;13:1901-1912. doi:10.2147/COPD.S152226
- Jithoo A, Enright PL, Burney P, et al. Case-finding options for COPD: results from the burden of obstructive lung disease study. Eur Respir J. 2013;41(3):548-555. doi:10.1183/09031936.00132011
- Haroon SM, Jordan RE, O’Beirne-Elliman J, Adab P. Effectiveness of case finding strategies for COPD in primary care: a systematic review and meta-analysis. NPJ Prim Care Respir Med. 2015;25:15056. doi:10.1038/npjpcrm.2015.56
- Porter J, Boyd C, Skandari MR, Laiteerapong N. Revisiting the time needed to provide adult primary care. J Gen Intern Med. 2023;38(1)147-155. doi:10.1007/s11606-022-07707-x
- Woo S, Zhou W, Larson JL. Stigma experiences in people with chronic obstructive pulmonary disease: an integrative review. Int J Chron Obstruct Pulmon Dis. 2021;16:1647- 1659. doi:10.2147/COPD.S306874
- Aaron SD, Montes de Oca M, Celli B, et al. Early diagnosis and treatment of COPD: the costs and benefits of case finding. Am J Respir Crit Care Med. 2024;209(8):928-937. doi:10.1164/rccm.202311-2120PP
- Kwon A, Lee C, Arafah A, Klein M, Namboodiri S, Lee C. Increasing chronic obstructive pulmonary disease (COPD) diagnosis with pulmonary function testing for patients with chest imaging evidence of COPD. Poster presented at: Society of General Internal Medicine Midwest Regional Meeting; October 19-20, 2023; Chicago, IL.
- Henderson LM, Su I, Rivera MP, et al. Prevalence of lung cancer screening in the US, 2022. JAMA Netw Open. 2024;7(3):e243190. doi:10.1001/jamanetworkopen.2024.3190
- Steiger D, Siddiqi MF, Yip R, Yankelevitz DF, Henschke CI; I-ELCAP investigators. The importance of low-dose CT screening to identify emphysema in asymptomatic participants with and without a prior diagnosis of COPD. Clin Imaging. 2021;78:136-141. doi:10.1016/j.clinimag.2021.03.012
- Pinsky PF, Lynch DA, Gierada DS. Incidental findings on low-dose CT scan lung cancer screenings and deaths from respiratory diseases. Chest. 2022;161(4):1092-1100. doi:10.1016/j.chest.2021.11.015
- Mulshine JL, Aldigé CR, Ambrose LF, et al. Emphysema detection in the course of lung cancer screening: optimizing a rare opportunity to impact population health. Ann Am Thorac Soc. 2023;20(4):499- 503. doi:10.1513/AnnalsATS.202207-631PS
Primary care practitioners (PCPs) in the US Department of Veterans Affairs (VA) provide care for patients with higher rates of many diseases—diabetes, heart disease, cancer, chronic obstructive pulmonary disease (COPD), and stroke—compared to the nonveteran population. 1 Due to the medical complexities of these diseases, they are often misdiagnosed or not diagnosed at all.
COPD is hiding in plain sight, impacting quality of life and burdening US health care systems.2 Research has yielded new treatments and evidence-based guidelines; however, COPD remains underdiagnosed. Only 13 million of the estimated 79 million US adults with COPD aged 20 to 79 years have been formally diagnosed.3 By the time patients are diagnosed, the disease is often advanced, and therapies are less effective. In 2 large studies of patients with COPD symptoms, later diagnosis was associated with worse outcomes.4,5
Veterans have a higher prevalence of COPD (8%-19%) than nonveterans (6%), likely due to higher rates of smoking and service-related exposures, especially among veterans of post-9/11 conflicts.6,7 Veterans do not always report symptoms and PCPs may not ask about symptoms, leading to underdiagnosis.8 The combination of high likelihood and underdetection of COPD presents a challenge and a target for VA quality improvement (QI).
The US Preventive Services Task Force (USPSTF) recommends against screening asymptomatic patients for COPD. However, both the USPSTF and the Global Initiative for Chronic Obstructive Lung Disease Report advocate for active case finding in primary care clinics to determine whether high-risk patients, such as smokers, experience COPD symptoms and warrant spirometry. 9,10 To make early COPD diagnoses, clinicians may use questionnaires alone or in combination with handheld peak expiratory flow rate measurements.11,12 Formal spirometry, considered the gold standard for COPD diagnosis, is ordered for patients who report COPD symptoms (ie, shortness of breath with exertion) or who have both COPD symptoms and reduced peak flow rates.
A systematic review and meta-analysis found that while the combination of questionnaires and peak flows was the more effective strategy overall, questionnaires alone were also valuable for identifying patients with possible COPD.13 Implementation of either screening method in primary care practices would be challenging. In a simulation study that applied chronic disease and preventive care guidelines to hypothetical patient panels, the time required for PCPs to provide guideline-recommended chronic and preventive care in addition to acute care far exceeded 8 hours per day, even in team-based settings.14 Overburdened PCPs are therefore unlikely to accept additional tasks like COPD case finding.
Why don’t patients report their pulmonary symptoms? Patients may not recognize the symptoms as evidence of COPD. Others may be afraid of a COPD diagnosis or the stigma that is associated with it.15 Perhaps they believe COPD treatment is ineffective because of lung damage from smoking. Some patients may not want to know if they have COPD, while others reduce activity levels to avoid symptoms.16
QUALITY IMPROVEMENT PROJECT
Given the high prevalence of COPD among veterans and the potential for underdiagnosis, VA Northeast Ohio Healthcare System (VANEOHS) internal medicine residents and faculty assessed the state of COPD diagnosis in its primary care clinic with a QI project in 2022. Patients in the clinic between August 1, 2015, and November 30, 2022, with an International Classification of Diseases-10 (ICD-10) COPD diagnosis code (J44) in the electronic health record were included. Of 157 included patients, 105 patients who had prior spirometry testing were excluded. Of the 52 patients with diagnosed COPD and no spirometry testing, 30 patients had computed tomography (CT) findings consistent with COPD (ie, airway thickening, emphysema, air trapping) that was performed for CT lung cancer screening (LCS).17 Twenty-three of these 30 patients were contacted by phone. All 23 were ever smokers and 13 reported COPD symptoms. The PCPs of the symptomatic patients were then contacted. Spirometry was ordered for all 13 patients and completed by 7. Three spirometry tests confirmed the COPD diagnosis. One PCP initiated inhaler therapy for a patient with newly diagnosed COPD.
All 11 PCPs of symptomatic patients were interviewed (many had > 1 symptomatic patient). They reported being unaware of patients’ COPD symptoms because the patients did not mention them, noting that screening for COPD was not a priority.
Role of Lung Cancer Screening
VA PCPs use electronic health record clinical reminders to track tests, consults, chronic disease education, cancer screenings, and routine health maintenance. A clinical reminder already exists (based on USPSTF recommendations) for LCS for patients aged 50 to 80 years who have a smoking history of 20 pack years. Patients who meet these criteria would also be considered high risk for COPD.
The VANEOHS QI project suggests that previously undiagnosed patients with findings of COPD on LCS may also have symptoms of COPD. Therefore, we wondered whether the LCS clinical reminder could serve a second purpose by prompting PCPs to ask veterans who meet LCS criteria about their COPD symptoms.
In 2022, about 13 million patients were eligible for LCS.18 Patients who qualify for LCS are at high risk for other cardiopulmonary disorders, such as COPD and coronary artery disease. Lung cancer is detected in only 1% of patients screened with CT at baseline. However, more often LCS yields evidence of additional cardiopulmonary disorders, such as emphysema or coronary artery calcifications. The International Early Lung Cancer Program (I-ELCAP) and the National Lung Cancer Screening Trial (NLST), which included > 79,000 patients, found evidence of emphysema on CT imaging in 24% and 31% of cases, respectively.19,20 In both cohorts, > 80% of patients with emphysema on CT imaging had no prior history of COPD.
In a 2022 article summarizing the potential impact of CT LCS on COPD diagnosis, Mulshine et al suggest that detection of emphysema on CT LCS provides “earlier recognition for PCPs to identify patients who would benefit from detailed symptom screening to prompt spirometry for COPD detection” and additional motivation for tobacco cessation.21 The VANEOHS QI project was developed and implemented prior to I-ELCAP or NLST reporting results but reinforces the value of CT LCS for COPD diagnosis.
Early diagnosis of COPD remains challenging because PCPs do not ask, patients do not tell, and symptoms can easily be dismissed. However, earlier diagnosis of COPD in symptomatic patients improves outcomes.3,4 To bridge this gap, VA PCPs and primary care patient aligned care teams (PACTs) need to commit to probing high-risk patients for COPD symptoms and ordering spirometry for those who are symptomatic. To accomplish this task, primary care teams need help.
The VANEOHS QI project confirmed that some patients with evidence of COPD on CT have symptoms of COPD that they did not share with their PCPs and suggests that LCS can be used as a dual action case finding method to screen both for lung cancer and COPD. We propose that patients who are eligible for LCS should also be probed for COPD symptoms at their clinic visits; for symptomatic patients, spirometry should be ordered, and COPD evidence-based management should be initiated when spirometry results are consistent with COPD. Annual probing for COPD symptoms could be considered in asymptomatic patients with ongoing tobacco use or emphysema on CT, since they may develop symptoms in the future. This new case-finding method bypasses the need for time-prohibitive questionnaires or peak flow measurements.
Future Opportunities
VA PCPs juggle many priorities and despite the simplicity of this new case finding COPD method, it may be unintentionally overlooked. PCPs often run out of time or may forget to ask patients about COPD symptoms when ordering LCS.
Future innovations to increase COPD diagnosis could include the creation of a yearly VA clinical reminder linked to the tobacco use reminder that has check boxes asking about symptoms of COPD in current and prior smokers. If patients have COPD symptoms, the reminder can prompt the ordering of spirometry. Similar reminders could be implemented to identify veterans with exposures to burn pits or other military environmental exposures who may have COPD symptoms. Another possible way to increase COPD diagnosis would be a partnership between primary care and the VA LCS program where patients receiving screening are asked about COPD symptoms during their LCS interviews and PACTs are alerted to order spirometry for symptomatic patients.
Elusive no longer! We can pull the veil back on COPD diagnosis and identify patients with possible COPD earlier in their course using their eligibility for LCS as a yearly reminder to probe them for symptoms. While not all patients who undergo LCS—even those with evidence of COPD on CT—will have COPD symptoms, symptoms may develop over time. LCS provides the possibility of 2 diagnoses from 1 test. This is an opportunity we cannot afford to miss.
Primary care practitioners (PCPs) in the US Department of Veterans Affairs (VA) provide care for patients with higher rates of many diseases—diabetes, heart disease, cancer, chronic obstructive pulmonary disease (COPD), and stroke—compared to the nonveteran population. 1 Due to the medical complexities of these diseases, they are often misdiagnosed or not diagnosed at all.
COPD is hiding in plain sight, impacting quality of life and burdening US health care systems.2 Research has yielded new treatments and evidence-based guidelines; however, COPD remains underdiagnosed. Only 13 million of the estimated 79 million US adults with COPD aged 20 to 79 years have been formally diagnosed.3 By the time patients are diagnosed, the disease is often advanced, and therapies are less effective. In 2 large studies of patients with COPD symptoms, later diagnosis was associated with worse outcomes.4,5
Veterans have a higher prevalence of COPD (8%-19%) than nonveterans (6%), likely due to higher rates of smoking and service-related exposures, especially among veterans of post-9/11 conflicts.6,7 Veterans do not always report symptoms and PCPs may not ask about symptoms, leading to underdiagnosis.8 The combination of high likelihood and underdetection of COPD presents a challenge and a target for VA quality improvement (QI).
The US Preventive Services Task Force (USPSTF) recommends against screening asymptomatic patients for COPD. However, both the USPSTF and the Global Initiative for Chronic Obstructive Lung Disease Report advocate for active case finding in primary care clinics to determine whether high-risk patients, such as smokers, experience COPD symptoms and warrant spirometry. 9,10 To make early COPD diagnoses, clinicians may use questionnaires alone or in combination with handheld peak expiratory flow rate measurements.11,12 Formal spirometry, considered the gold standard for COPD diagnosis, is ordered for patients who report COPD symptoms (ie, shortness of breath with exertion) or who have both COPD symptoms and reduced peak flow rates.
A systematic review and meta-analysis found that while the combination of questionnaires and peak flows was the more effective strategy overall, questionnaires alone were also valuable for identifying patients with possible COPD.13 Implementation of either screening method in primary care practices would be challenging. In a simulation study that applied chronic disease and preventive care guidelines to hypothetical patient panels, the time required for PCPs to provide guideline-recommended chronic and preventive care in addition to acute care far exceeded 8 hours per day, even in team-based settings.14 Overburdened PCPs are therefore unlikely to accept additional tasks like COPD case finding.
Why don’t patients report their pulmonary symptoms? Patients may not recognize the symptoms as evidence of COPD. Others may be afraid of a COPD diagnosis or the stigma that is associated with it.15 Perhaps they believe COPD treatment is ineffective because of lung damage from smoking. Some patients may not want to know if they have COPD, while others reduce activity levels to avoid symptoms.16
QUALITY IMPROVEMENT PROJECT
Given the high prevalence of COPD among veterans and the potential for underdiagnosis, VA Northeast Ohio Healthcare System (VANEOHS) internal medicine residents and faculty assessed the state of COPD diagnosis in its primary care clinic with a QI project in 2022. Patients in the clinic between August 1, 2015, and November 30, 2022, with an International Classification of Diseases-10 (ICD-10) COPD diagnosis code (J44) in the electronic health record were included. Of 157 included patients, 105 patients who had prior spirometry testing were excluded. Of the 52 patients with diagnosed COPD and no spirometry testing, 30 patients had computed tomography (CT) findings consistent with COPD (ie, airway thickening, emphysema, air trapping) that was performed for CT lung cancer screening (LCS).17 Twenty-three of these 30 patients were contacted by phone. All 23 were ever smokers and 13 reported COPD symptoms. The PCPs of the symptomatic patients were then contacted. Spirometry was ordered for all 13 patients and completed by 7. Three spirometry tests confirmed the COPD diagnosis. One PCP initiated inhaler therapy for a patient with newly diagnosed COPD.
All 11 PCPs of symptomatic patients were interviewed (many had > 1 symptomatic patient). They reported being unaware of patients’ COPD symptoms because the patients did not mention them, noting that screening for COPD was not a priority.
Role of Lung Cancer Screening
VA PCPs use electronic health record clinical reminders to track tests, consults, chronic disease education, cancer screenings, and routine health maintenance. A clinical reminder already exists (based on USPSTF recommendations) for LCS for patients aged 50 to 80 years who have a smoking history of 20 pack years. Patients who meet these criteria would also be considered high risk for COPD.
The VANEOHS QI project suggests that previously undiagnosed patients with findings of COPD on LCS may also have symptoms of COPD. Therefore, we wondered whether the LCS clinical reminder could serve a second purpose by prompting PCPs to ask veterans who meet LCS criteria about their COPD symptoms.
In 2022, about 13 million patients were eligible for LCS.18 Patients who qualify for LCS are at high risk for other cardiopulmonary disorders, such as COPD and coronary artery disease. Lung cancer is detected in only 1% of patients screened with CT at baseline. However, more often LCS yields evidence of additional cardiopulmonary disorders, such as emphysema or coronary artery calcifications. The International Early Lung Cancer Program (I-ELCAP) and the National Lung Cancer Screening Trial (NLST), which included > 79,000 patients, found evidence of emphysema on CT imaging in 24% and 31% of cases, respectively.19,20 In both cohorts, > 80% of patients with emphysema on CT imaging had no prior history of COPD.
In a 2022 article summarizing the potential impact of CT LCS on COPD diagnosis, Mulshine et al suggest that detection of emphysema on CT LCS provides “earlier recognition for PCPs to identify patients who would benefit from detailed symptom screening to prompt spirometry for COPD detection” and additional motivation for tobacco cessation.21 The VANEOHS QI project was developed and implemented prior to I-ELCAP or NLST reporting results but reinforces the value of CT LCS for COPD diagnosis.
Early diagnosis of COPD remains challenging because PCPs do not ask, patients do not tell, and symptoms can easily be dismissed. However, earlier diagnosis of COPD in symptomatic patients improves outcomes.3,4 To bridge this gap, VA PCPs and primary care patient aligned care teams (PACTs) need to commit to probing high-risk patients for COPD symptoms and ordering spirometry for those who are symptomatic. To accomplish this task, primary care teams need help.
The VANEOHS QI project confirmed that some patients with evidence of COPD on CT have symptoms of COPD that they did not share with their PCPs and suggests that LCS can be used as a dual action case finding method to screen both for lung cancer and COPD. We propose that patients who are eligible for LCS should also be probed for COPD symptoms at their clinic visits; for symptomatic patients, spirometry should be ordered, and COPD evidence-based management should be initiated when spirometry results are consistent with COPD. Annual probing for COPD symptoms could be considered in asymptomatic patients with ongoing tobacco use or emphysema on CT, since they may develop symptoms in the future. This new case-finding method bypasses the need for time-prohibitive questionnaires or peak flow measurements.
Future Opportunities
VA PCPs juggle many priorities and despite the simplicity of this new case finding COPD method, it may be unintentionally overlooked. PCPs often run out of time or may forget to ask patients about COPD symptoms when ordering LCS.
Future innovations to increase COPD diagnosis could include the creation of a yearly VA clinical reminder linked to the tobacco use reminder that has check boxes asking about symptoms of COPD in current and prior smokers. If patients have COPD symptoms, the reminder can prompt the ordering of spirometry. Similar reminders could be implemented to identify veterans with exposures to burn pits or other military environmental exposures who may have COPD symptoms. Another possible way to increase COPD diagnosis would be a partnership between primary care and the VA LCS program where patients receiving screening are asked about COPD symptoms during their LCS interviews and PACTs are alerted to order spirometry for symptomatic patients.
Elusive no longer! We can pull the veil back on COPD diagnosis and identify patients with possible COPD earlier in their course using their eligibility for LCS as a yearly reminder to probe them for symptoms. While not all patients who undergo LCS—even those with evidence of COPD on CT—will have COPD symptoms, symptoms may develop over time. LCS provides the possibility of 2 diagnoses from 1 test. This is an opportunity we cannot afford to miss.
- Betancourt JA, Granados PS, Pacheco GJ, et al. Exploring health outcomes for U.S. veterans compared to non-veterans from 2003 to 2019. Healthcare (Basel). 2021;9(5):604. doi:10.3390/healthcare90506064
- Bamonti PM, Fischer I, Moye J, Poghosyan H, Pietrzak RH. Obstructive respiratory disease in U.S. veterans: prevalence, characteristics, and health burden. J Psychiatr Res. 2024;176:140-147. doi:10.1016/j.jpsychires.2024.05.053
- Criner RN, Han MK. COPD care in the 21st century: a public health priority. Respir Care. 2018;63(5):591-600. doi:10.4187/respcare.06276
- Larsson K, Janson C, Ställberg B, et al. Impact of COPD diagnosis timing on clinical and economic outcomes: the ARCTIC observational cohort study. Int J Chron Obstruct Pulmon Dis. 2019;14:995-1008. doi:10.2147/COPD.S195382
- Kostikas K, Price D, Gutzwiller FS, et al. Clinical impact and healthcare resource utilization associated with early versus late COPD diagnosis in patients from UK CPRD Database. Int J Chron Obstruct Pulmon Dis. 2020;15:1729- 1738. doi:10.2147/COPD.S255414
- Bamonti PM, Robinson SA, Wan ES, Moy ML. Improving physiological, physical, and psychological health outcomes: a narrative review in US veterans with COPD. Int J Chron Obstruct Pulmon Dis. 2022;17:1269-1283. doi:10.2147/COPD.S339323
- Savitz DA, Woskie SR, Bello A, et al. Deployment to military bases with open burn pits and respiratory and cardiovascular disease. JAMA Netw Open. 2024;7(4):e247629. doi:10.1001/jamanetworkopen.2024.7629
- Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
- Guirguis-Blake JM, Senger CA, Webber EM, Mularski RA, Whitlock EP. Screening for chronic obstructive pulmonary disease: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2016;315(13):1378-1393. doi:10.1001/jama.2016.2654
- Capriotti T, Tomy R, Morales M. COPD updates: 2023 GOLD Report for primary care providers. Clinical Advisor. May 9, 2023. Accessed May 14, 2025. https://www.clinicaladvisor.com/features/copd-updates-2023-gold-report-primary-care/
- Leidy NK, Martinez FJ, Malley KG, et al. Can CAPTURE be used to identify undiagnosed patients with mild- to- moderate COPD likely to benefit from treatment? Int J Chron Obstruct Pulmon Dis. 2018;13:1901-1912. doi:10.2147/COPD.S152226
- Jithoo A, Enright PL, Burney P, et al. Case-finding options for COPD: results from the burden of obstructive lung disease study. Eur Respir J. 2013;41(3):548-555. doi:10.1183/09031936.00132011
- Haroon SM, Jordan RE, O’Beirne-Elliman J, Adab P. Effectiveness of case finding strategies for COPD in primary care: a systematic review and meta-analysis. NPJ Prim Care Respir Med. 2015;25:15056. doi:10.1038/npjpcrm.2015.56
- Porter J, Boyd C, Skandari MR, Laiteerapong N. Revisiting the time needed to provide adult primary care. J Gen Intern Med. 2023;38(1)147-155. doi:10.1007/s11606-022-07707-x
- Woo S, Zhou W, Larson JL. Stigma experiences in people with chronic obstructive pulmonary disease: an integrative review. Int J Chron Obstruct Pulmon Dis. 2021;16:1647- 1659. doi:10.2147/COPD.S306874
- Aaron SD, Montes de Oca M, Celli B, et al. Early diagnosis and treatment of COPD: the costs and benefits of case finding. Am J Respir Crit Care Med. 2024;209(8):928-937. doi:10.1164/rccm.202311-2120PP
- Kwon A, Lee C, Arafah A, Klein M, Namboodiri S, Lee C. Increasing chronic obstructive pulmonary disease (COPD) diagnosis with pulmonary function testing for patients with chest imaging evidence of COPD. Poster presented at: Society of General Internal Medicine Midwest Regional Meeting; October 19-20, 2023; Chicago, IL.
- Henderson LM, Su I, Rivera MP, et al. Prevalence of lung cancer screening in the US, 2022. JAMA Netw Open. 2024;7(3):e243190. doi:10.1001/jamanetworkopen.2024.3190
- Steiger D, Siddiqi MF, Yip R, Yankelevitz DF, Henschke CI; I-ELCAP investigators. The importance of low-dose CT screening to identify emphysema in asymptomatic participants with and without a prior diagnosis of COPD. Clin Imaging. 2021;78:136-141. doi:10.1016/j.clinimag.2021.03.012
- Pinsky PF, Lynch DA, Gierada DS. Incidental findings on low-dose CT scan lung cancer screenings and deaths from respiratory diseases. Chest. 2022;161(4):1092-1100. doi:10.1016/j.chest.2021.11.015
- Mulshine JL, Aldigé CR, Ambrose LF, et al. Emphysema detection in the course of lung cancer screening: optimizing a rare opportunity to impact population health. Ann Am Thorac Soc. 2023;20(4):499- 503. doi:10.1513/AnnalsATS.202207-631PS
- Betancourt JA, Granados PS, Pacheco GJ, et al. Exploring health outcomes for U.S. veterans compared to non-veterans from 2003 to 2019. Healthcare (Basel). 2021;9(5):604. doi:10.3390/healthcare90506064
- Bamonti PM, Fischer I, Moye J, Poghosyan H, Pietrzak RH. Obstructive respiratory disease in U.S. veterans: prevalence, characteristics, and health burden. J Psychiatr Res. 2024;176:140-147. doi:10.1016/j.jpsychires.2024.05.053
- Criner RN, Han MK. COPD care in the 21st century: a public health priority. Respir Care. 2018;63(5):591-600. doi:10.4187/respcare.06276
- Larsson K, Janson C, Ställberg B, et al. Impact of COPD diagnosis timing on clinical and economic outcomes: the ARCTIC observational cohort study. Int J Chron Obstruct Pulmon Dis. 2019;14:995-1008. doi:10.2147/COPD.S195382
- Kostikas K, Price D, Gutzwiller FS, et al. Clinical impact and healthcare resource utilization associated with early versus late COPD diagnosis in patients from UK CPRD Database. Int J Chron Obstruct Pulmon Dis. 2020;15:1729- 1738. doi:10.2147/COPD.S255414
- Bamonti PM, Robinson SA, Wan ES, Moy ML. Improving physiological, physical, and psychological health outcomes: a narrative review in US veterans with COPD. Int J Chron Obstruct Pulmon Dis. 2022;17:1269-1283. doi:10.2147/COPD.S339323
- Savitz DA, Woskie SR, Bello A, et al. Deployment to military bases with open burn pits and respiratory and cardiovascular disease. JAMA Netw Open. 2024;7(4):e247629. doi:10.1001/jamanetworkopen.2024.7629
- Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
- Guirguis-Blake JM, Senger CA, Webber EM, Mularski RA, Whitlock EP. Screening for chronic obstructive pulmonary disease: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2016;315(13):1378-1393. doi:10.1001/jama.2016.2654
- Capriotti T, Tomy R, Morales M. COPD updates: 2023 GOLD Report for primary care providers. Clinical Advisor. May 9, 2023. Accessed May 14, 2025. https://www.clinicaladvisor.com/features/copd-updates-2023-gold-report-primary-care/
- Leidy NK, Martinez FJ, Malley KG, et al. Can CAPTURE be used to identify undiagnosed patients with mild- to- moderate COPD likely to benefit from treatment? Int J Chron Obstruct Pulmon Dis. 2018;13:1901-1912. doi:10.2147/COPD.S152226
- Jithoo A, Enright PL, Burney P, et al. Case-finding options for COPD: results from the burden of obstructive lung disease study. Eur Respir J. 2013;41(3):548-555. doi:10.1183/09031936.00132011
- Haroon SM, Jordan RE, O’Beirne-Elliman J, Adab P. Effectiveness of case finding strategies for COPD in primary care: a systematic review and meta-analysis. NPJ Prim Care Respir Med. 2015;25:15056. doi:10.1038/npjpcrm.2015.56
- Porter J, Boyd C, Skandari MR, Laiteerapong N. Revisiting the time needed to provide adult primary care. J Gen Intern Med. 2023;38(1)147-155. doi:10.1007/s11606-022-07707-x
- Woo S, Zhou W, Larson JL. Stigma experiences in people with chronic obstructive pulmonary disease: an integrative review. Int J Chron Obstruct Pulmon Dis. 2021;16:1647- 1659. doi:10.2147/COPD.S306874
- Aaron SD, Montes de Oca M, Celli B, et al. Early diagnosis and treatment of COPD: the costs and benefits of case finding. Am J Respir Crit Care Med. 2024;209(8):928-937. doi:10.1164/rccm.202311-2120PP
- Kwon A, Lee C, Arafah A, Klein M, Namboodiri S, Lee C. Increasing chronic obstructive pulmonary disease (COPD) diagnosis with pulmonary function testing for patients with chest imaging evidence of COPD. Poster presented at: Society of General Internal Medicine Midwest Regional Meeting; October 19-20, 2023; Chicago, IL.
- Henderson LM, Su I, Rivera MP, et al. Prevalence of lung cancer screening in the US, 2022. JAMA Netw Open. 2024;7(3):e243190. doi:10.1001/jamanetworkopen.2024.3190
- Steiger D, Siddiqi MF, Yip R, Yankelevitz DF, Henschke CI; I-ELCAP investigators. The importance of low-dose CT screening to identify emphysema in asymptomatic participants with and without a prior diagnosis of COPD. Clin Imaging. 2021;78:136-141. doi:10.1016/j.clinimag.2021.03.012
- Pinsky PF, Lynch DA, Gierada DS. Incidental findings on low-dose CT scan lung cancer screenings and deaths from respiratory diseases. Chest. 2022;161(4):1092-1100. doi:10.1016/j.chest.2021.11.015
- Mulshine JL, Aldigé CR, Ambrose LF, et al. Emphysema detection in the course of lung cancer screening: optimizing a rare opportunity to impact population health. Ann Am Thorac Soc. 2023;20(4):499- 503. doi:10.1513/AnnalsATS.202207-631PS
The Use of Lung Cancer Screening to Increase Chronic Obstructive Pulmonary Disease Diagnosis in Veterans Affairs Primary Care
The Use of Lung Cancer Screening to Increase Chronic Obstructive Pulmonary Disease Diagnosis in Veterans Affairs Primary Care
When Patient-Centered Care Initiatives Align: Integrating VA Whole Health and Shared Decision-Making for Lung Cancer Screening
When Patient-Centered Care Initiatives Align: Integrating VA Whole Health and Shared Decision-Making for Lung Cancer Screening
The landmark Crossing the Quality Chasm report from the National Academy of Medicine identified patient- centered care as essential to health care quality. The report defines patientcentered care as “respectful of and responsive to individual patient preferences, needs, and values.”1 Many health care systems, including the Veterans Health Administration, are transforming to a patient-centered model of care.2 The US Department of Veterans Affairs (VA) Whole Health System of Care initiative is a system-wide, cultural transformation. Within whole health, what matters most to the patient—including their preferences, needs, and values—is foundational to health care and meant to be essential in every clinical encounter. Whole health implementation includes a progressive rollout with health care practitioner (HCP) trainings across the VA.2
Shared decision-making (SDM) is a different but aligned patient-centered care concept. SDM is a process through which a decision or care plan, based on patients’ preferences, needs, and values, is made or developed.3-5 SDM is ideal in situations with equipoise (decisions with equivalent choices), individualized risks, and/or greater uncertainty of the net benefit, such as with lung cancer screening (LCS).3 SDM for LCS is required by the US Centers for Medicare and Medicaid Services and has been adopted by many US health care systems, including the VA.6,7 Early detection of lung cancer can reduce death by 20% at the population level.8 However, at the patient level there is wide variation in the risk of developing lung cancer and a range of potential harms.8 LCS follow-up procedures may be more invasive than with other cancer screenings. Thus, there is concern about the risk of false-positive results leading to unnecessary care or complications.8 Given this balance between benefit and harm and the differing patient value on the trade-offs of LCS, an individualized, patient-centered approach is essential when deciding whether LCS is the right choice for a specific patient.
Despite the importance of LCS SDM, observational studies have shown poor implementation in clinical encounters.9,10 HCP barriers include competing demands, limited time, lack of familiarity with and training in SDM, and beliefs biasing screening over no screening.11-13 Additionally, HCPs may assume that patients want them to make the decision. However, research has shown that patients actually want to be more involved in their health care decisions.14 One suggested strategy to overcome these barriers is aligning SDM for LCS within an organization’s broader patient-centered initiatives.15
This project sought to align the need for SDM for LCS and the broader VA whole health initiative as part of a multilevel strategy to implement SDM for LCS across Veterans Integrated Service Network (VISN) 1.16
This article addresses HCP-level barriers. HCPs targeted are those typically involved in LCS. The VA utilizes LCS coordinators (LCSCs) in both centralized or consult models (in which LCSCs are involved in all aspects of screening) and hybrid models (in which primary care practitioners and LCSCs are both engaged in LCS tasks). The goal of this program was to generate areas of conceptual alignment between SDM and whole health as a first step in integrating these VA initiatives. This work was conducted as a foundation for an SDM for lung cancer HCP training and consultation initiative.
ALIGNMENT PROCESS
We reviewed relevant literature and resources for SDM and whole health. In reviewing the SDM literature, we included a sample of the most widely cited literature on the topic, and focused primarily on the systematic review by Bomhof-Roordink et al.4,5,17,18 This review provided a synthesis of SDM elements across SDM models and identified 53 different elements clustered into 24 components.4 The most common components were present in at least half of all SDM published models, including: make the decision, patient preferences, tailor information, deliberate, create choice awareness, and learn about the patient. Bomhof-Roordink et al provided the guiding framework for this conceptualization of SDM because that study included the available recent published SDM models.4
Second, published literature on VA whole health along with supplemental promotional and training materials were reviewed. The whole health materials included 2 sets of training slides developed for VA HCPs (available to VA employees): Implementing Whole Health in Clinical Care, which is focused on HCPs’ work with patients, and Whole Health for You and Me, which is about HCPs’ personal well-being.19 We also reviewed a publication describing the history of whole health and patient-facing online whole health tools.2,19
Each document was reviewed for key elements related to SDM, patient-centered care, and whole health. Using the 53 elements identified by Bomhof-Roordink et al, we reviewed and compared each element to the whole health materials to create the integrated model of SDM and whole health. We iteratively discussed and organized the elements until we reached consensus.
SDM and Whole Health Alignment
We created an integrated model of SDM for LCS within the context of the VA whole health initiative. This integrated model is directed at HCPs who would likely engage patients in discussions of LCS, including primary care practitioners and nurse coordinators. The model includes 3 steps for HCPs to follow that align SDM within whole health: (1) frame the conversation and partner with the patient; (2) share clinical perspective and elicit patient values; and (3) deliberate and decide together. For each step, the SDM elements, whole health elements, and integration of SDM and whole health are provided. Table 1 provides an overview of the similarities and differences between SDM and whole health. Example phrases that merge SDM and whole health for HCPs to use in patient conversations about LCS are included in Table 2.
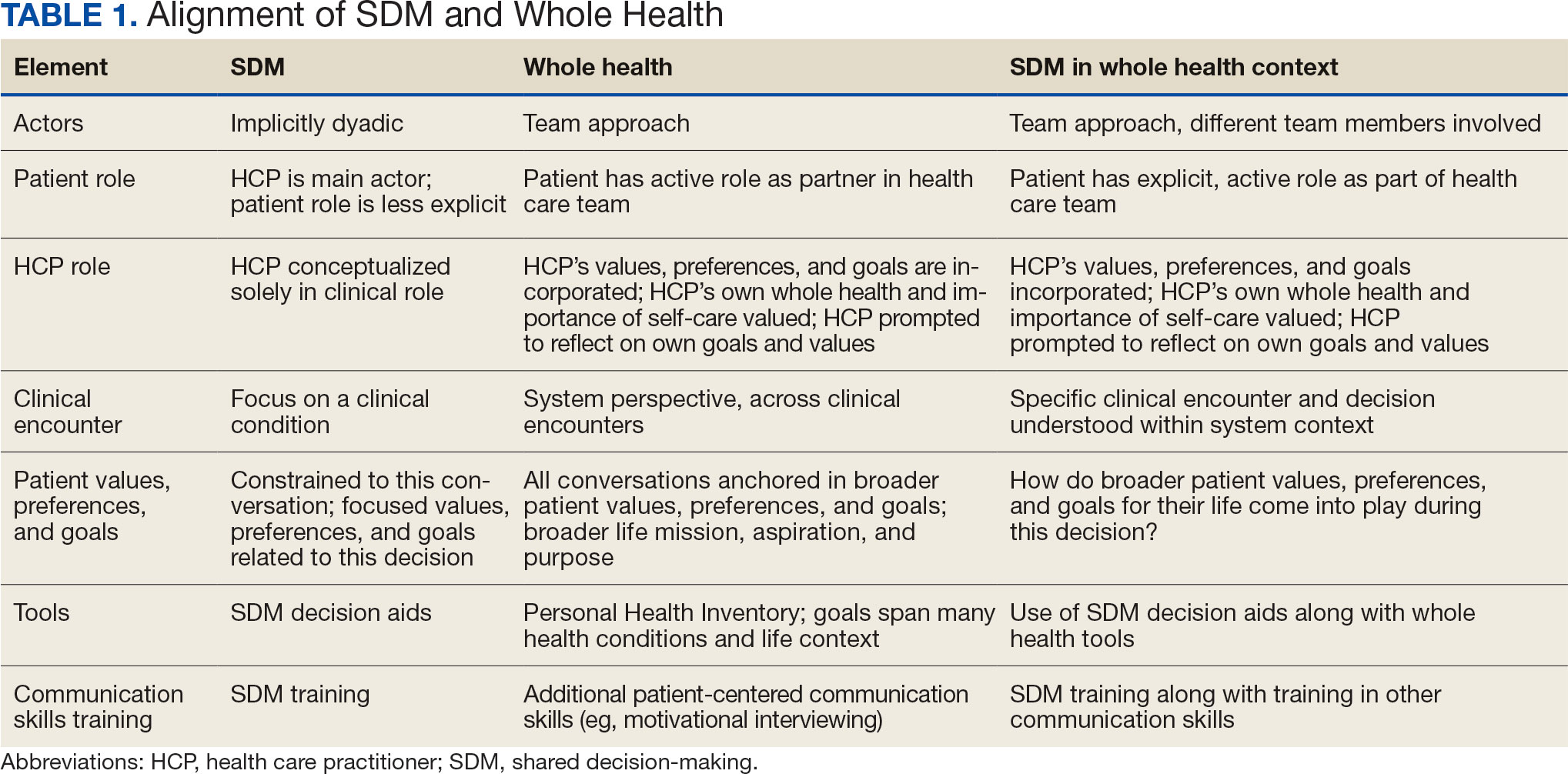

STEP 1. FRAME THE CONVERSATION AND PARTNER WITH THE PATIENT
Shared decision-making. Traditional SDM literature includes an initial step of letting patients know that there is a choice to be made between ≥ 2 clinical options.4 Ancillary elements of this first step include asking patients their preferences about the degree to which they want to be involved in SDM and about how they like to receive information (eg, verbal, written, video). These steps open the SDM conversation and ensure the patient and HCP are on the same page before moving forward. For example, the US Agency for Healthcare Research and Quality SHARE model’s first step is for HCPs to communicate that choices exist and to invite the patient to be involved in decisions.20 Similarly, Elwyn’s 3-step SDM model begins with establishing that a choice exists and inviting patient input on making that choice.17
Whole health. Patients are encouraged to play an active role in their health care. Through whole health programs such as Taking Charge of My Life and Health, patients explore their values and set self-care goals.21 HCP whole health trainings teach and reinforce communication skills, including SDM, listening skills, and motivational interviewing.19
Shared decision-making/whole health integration. SDM and whole health both prioritize respect, compassion, and patients’ expertise. They focus on the patient-HCP relationship with an emphasis on fostering egalitarian interactions. HCPs frame the SDM conversation and partner with the patient so they know what to expect and who will be involved. This conversation is framed from the outset as a collaborative discussion. HCPs empower the patient to play an active role in decision-making and help them understand why their engagement is critical.
STEP 2. SHARE CLINICAL PERSPECTIVE AND ELICIT PATIENT VALUES
Shared decision-making. HCPs share clinical perspective on LCS tailored to individual patients while explicitly inviting the patient to share their preferences and values when thinking about whether to undergo LCS. HCPs give a balanced description of LCS, including the benefits and harms, tailored to the patient’s unique information needs and questions. Sharing clinical perspective also includes describing treatment options, the most common element across SDM models.4 Decision aids, which provide unbiased information and include a values clarification exercise, may be helpful in sharing clinical perspectives and clarifying patient values related to the trade-offs of LCS.22 For example, the VA National Center for Health Promotion and Disease Prevention developed a LCS decision aid to be used for SDM for LCS.
Whole health. The conversation shifts from “What is the matter with you?” to “What matters to you?” starting with the patient’s goals and priorities rather than disease prevention, diagnosis, and treatment.2 Several whole health tools exist, including the Personal Health Inventory, used to identify what matters most to patients and understand their current well-being and self-care.23 Using the inventory, the patient and their health care team develop the patient’s personal health plan.24 Additionally, whole health trains HCPs to reflect on their own attitudes and biases when providing clinical care.
Shared decision-making/whole health integration. The LCS conversation can build on other whole health-related conversations with a HCP or other team members. HCPs can reference the patient’s personal health plan for documentation of the patient’s preferences, values, and goals in the electronic medical record. During this process, HCPs can give space for patients to discuss factors in their life and experiences that impact their perspective and decision-making. For example, patient concerns could be explored here, including fear of a cancer diagnosis, stigma around smoking, and fears around the screening and/or treatment process. HCPs may ask, “What matters most to you when making this decision?” Finally, by sharing clinical information, HCPs will focus on patient values to help overcome their own biases toward a desire for LCS. HCPs, similar to the rest of the US public, tend to hold highly favorable attitudes toward cancer screening as well as misconceptions about the magnitude of benefits from screening.13
STEP 3. DELIBERATE AND DECIDE TOGETHER
Shared decision-making. Decision-making is almost always considered the last SDM step.4 In the final step, the patient and HCP discuss the options (ie, to screen or not to screen) considering the patient’s values and preferences, and patients decide with their HCP whether they will undergo LCS. Patients may decide they need more time to think about these options. As part of deliberation, HCPs assess what other information patients may need to arrive at a decision. Family members, friends, or peers may be included in making the final decision.
Whole health. In Whole health, decisions also may include the entire health care team and other individuals important to the patient (eg, family, friends). Integration across different health care settings is also considered a key whole health element. Finally, whole health focuses on long-term relationships with patients; thus, the LCS SDM process is situated within longer term relationship building and patient empowerment, both of which will facilitate partnering with the patient in future conversations about other decisions.
Shared decision-making/whole health integration. Both SDM and whole health emphasize partnership with the patient in making a final decision. There is also focus on decision-making as an ongoing process. Deciding whether LCS is the best choice might include naming and addressing emotions, voicing questions not raised, and exploring whether screening fits the patient’s goals, values, and life context. HCPs may give guidance, but patients retain the authority to make decisions. The goal is to empower patients to know that the only right decision is the one right for them and they will be supported.
Limitations
This article describes a VA practice program and was not a formal research study. Further work is needed to evaluate the presented strategies. Additionally, we did not conduct a systematic literature review and thus elements of SDM and whole health may not be exhaustive.
CONCLUSIONS
This article describes the alignment of 2 distinct VA initiatives, whole health and SDM for LCS. The goal was to reduce known barriers to SDM, such as competing demands, limited time, and lack of familiarity with and training in SDM.11-13 These concepts are well aligned. This integrated model is the first step in informing the development of a HCP training program and materials as part of a multilevel strategy that our team is using to implement SDM for LCS in VISN 1.16 The final training and materials resulting from this work were delivered to LCSCs in 3 ways: (1) a series of 3 interactive group training sessions, including didactic elements, role play, and time for open discussion; (2) 1-on-1 academic detailing; and (3) educational handouts. In academic detailing, a member of the research team trained in academic detailing met virtually with each nurse coordinator, identified that individual’s barriers to SDM, and used the training materials to highlight messages to overcome those barriers; follow-up calls provided a forum for discussing progress and overcoming additional challenges. Although this article focused specifically on whole health and SDM, the conceptual alignment process strategy can be applied to other implementations of multiple initiatives.
- Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. The National Academies Press; 2001. doi:10.17226/10027
- Bokhour BG, Haun JN, Hyde J, Charns M, Kligler B. Transforming the Veterans Affairs to a whole health system of care: time for action and research. Med Care. 2020;58:295- 300. doi:10.1097/MLR.0000000000001316
- Elwyn G, Frosch D, Rollnick S. Dual equipoise shared decision making: definitions for decision and behaviour support interventions. Implement Sci. 2009;4:75. doi:7510.1186/1748-5908-4-75
- Bomhof-Roordink H, Gärtner FR, Stiggelbout AM, Pieterse AH. Key components of shared decision making models: a systematic review. BMJ Open. 2019;9:e031763. doi:10.1136/bmjopen-2019-031763
- Charles C, Gafni A, Whelan T. Decision-making in the physician- patient encounter: revisiting the shared treatment decision-making model. Soc Sci Med. 1999;49:651-661. doi:10.1016/s0277-9536(99)00145-8
- Moyer VA; US Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160:330- 338. doi:10.7326/m13-2771
- Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT). February 10, 2022. Accessed February 7, 2025. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=304
- Aberle DR, Adams AM, Berg CD, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365:395-409. doi:10.1056/NEJMoa1102873
- Slatore CG, Wiener RS. Pulmonary nodules: a small problem for many, severe distress for some, and how to communicate about it. Chest. 2018;153:1004-1015. doi:10.1016/j.chest.2017.10.013
- Nishi SPE, Lowenstein LM, Mendoza TR, et al. Shared decision-making for lung cancer screening: how well are we “sharing”? Chest. 2021;160:330-340. doi:10.1016/j.chest.2021.01.041
- Wiener RS, Koppelman E, Bolton R, et al. Patient and clinician perspectives on shared decision-making in early adopting lung cancer screening programs: a qualitative study. J Gen Intern Med. 2018;33:1035-1042. doi:10.1007/s11606-018-4350-9
- Melzer AC, Golden SE, Ono SS, Datta S, Triplette M, Slatore CG. “We just never have enough time”: clinician views of lung cancer screening processes and implementation. Ann Am Thorac Soc. 2020. doi:10.1513/AnnalsATS.202003-262OC
- Schwartz LM, Woloshin S, Fowler FJ Jr, Welch HG. Enthusiasm for cancer screening in the United States. JAMA. 2004;291:71-78. doi:10.1001/jama.291.1.71
- Lown BA, Rosen J, Marttila J. An agenda for improving compassionate care: a survey shows about half of patients say such care is missing. Health Aff (Millwood). 2011;30:1772-1778. doi:10.1377/hlthaff.2011.0539
- Scholl I, LaRussa A, Hahlweg P, Kobrin S, Elwyn G. Organizational- and system-level characteristics that influence implementation of shared decision-making and strategies to address them - a scoping review. Implement Sci. 2018;13:40. doi:10.1186/s13012-018-0731-z
- Khanna A, Fix GM, Anderson E, et al. Towards a framework for patient-centred care coordination: a scoping review protocol. BMJ Open. 2022;12:e066808. doi:10.1136/bmjopen-2022-066808
- Elwyn G, Durand MA, Song J, et al. A three-talk model for shared decision making: multistage consultation process. BMJ. 2017;359:j4891. doi:10.1136/bmj.j4891
- Makoul G, Clayman ML. An integrative model of shared decision making in medical encounters. Patient Educ Couns. 2006;60:301-312. doi:10.1016/j.pec.2005.06.010
- Whole Health. US Department of Veterans Affairs. Accessed April 14, 2025. https://www.va.gov/wholehealth/
- Agency for Healthcare Research and Quality. The SHARE approach. Accessed April 14, 2025. https://www.ahrq.gov/health-literacy/professional-training/shared-decision/index.html
- Abadi MH, Barker AM, Rao SR, Orner M, Rychener D, Bokhour BG. Examining the impact of a peer-led group program for veteran engagement and well-being. J Altern Complement Med. 2021;27:S37-S44. doi:10.1089/acm.2020.0124
- Stacey D, Lewis KB, Smith M, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2024;1:CD001431. doi:10.1002/14651858.CD001431.pub6
- US Department of Veterans Affairs, Veterans Health Administration, Office of Patient Centered Care and Cultural Transformation. Personal health inventory. Revised April 2019. Accessed April 14, 2025. https://www.va.gov/wholehealth/docs/10-773_PHI_July2019_508.pdf
- US Department of Veterans Affairs. Build your personal health plan. Updated July 24, 2024. Accessed April 14, 2025. https://www.va.gov/wholehealth/phi.asp
The landmark Crossing the Quality Chasm report from the National Academy of Medicine identified patient- centered care as essential to health care quality. The report defines patientcentered care as “respectful of and responsive to individual patient preferences, needs, and values.”1 Many health care systems, including the Veterans Health Administration, are transforming to a patient-centered model of care.2 The US Department of Veterans Affairs (VA) Whole Health System of Care initiative is a system-wide, cultural transformation. Within whole health, what matters most to the patient—including their preferences, needs, and values—is foundational to health care and meant to be essential in every clinical encounter. Whole health implementation includes a progressive rollout with health care practitioner (HCP) trainings across the VA.2
Shared decision-making (SDM) is a different but aligned patient-centered care concept. SDM is a process through which a decision or care plan, based on patients’ preferences, needs, and values, is made or developed.3-5 SDM is ideal in situations with equipoise (decisions with equivalent choices), individualized risks, and/or greater uncertainty of the net benefit, such as with lung cancer screening (LCS).3 SDM for LCS is required by the US Centers for Medicare and Medicaid Services and has been adopted by many US health care systems, including the VA.6,7 Early detection of lung cancer can reduce death by 20% at the population level.8 However, at the patient level there is wide variation in the risk of developing lung cancer and a range of potential harms.8 LCS follow-up procedures may be more invasive than with other cancer screenings. Thus, there is concern about the risk of false-positive results leading to unnecessary care or complications.8 Given this balance between benefit and harm and the differing patient value on the trade-offs of LCS, an individualized, patient-centered approach is essential when deciding whether LCS is the right choice for a specific patient.
Despite the importance of LCS SDM, observational studies have shown poor implementation in clinical encounters.9,10 HCP barriers include competing demands, limited time, lack of familiarity with and training in SDM, and beliefs biasing screening over no screening.11-13 Additionally, HCPs may assume that patients want them to make the decision. However, research has shown that patients actually want to be more involved in their health care decisions.14 One suggested strategy to overcome these barriers is aligning SDM for LCS within an organization’s broader patient-centered initiatives.15
This project sought to align the need for SDM for LCS and the broader VA whole health initiative as part of a multilevel strategy to implement SDM for LCS across Veterans Integrated Service Network (VISN) 1.16
This article addresses HCP-level barriers. HCPs targeted are those typically involved in LCS. The VA utilizes LCS coordinators (LCSCs) in both centralized or consult models (in which LCSCs are involved in all aspects of screening) and hybrid models (in which primary care practitioners and LCSCs are both engaged in LCS tasks). The goal of this program was to generate areas of conceptual alignment between SDM and whole health as a first step in integrating these VA initiatives. This work was conducted as a foundation for an SDM for lung cancer HCP training and consultation initiative.
ALIGNMENT PROCESS
We reviewed relevant literature and resources for SDM and whole health. In reviewing the SDM literature, we included a sample of the most widely cited literature on the topic, and focused primarily on the systematic review by Bomhof-Roordink et al.4,5,17,18 This review provided a synthesis of SDM elements across SDM models and identified 53 different elements clustered into 24 components.4 The most common components were present in at least half of all SDM published models, including: make the decision, patient preferences, tailor information, deliberate, create choice awareness, and learn about the patient. Bomhof-Roordink et al provided the guiding framework for this conceptualization of SDM because that study included the available recent published SDM models.4
Second, published literature on VA whole health along with supplemental promotional and training materials were reviewed. The whole health materials included 2 sets of training slides developed for VA HCPs (available to VA employees): Implementing Whole Health in Clinical Care, which is focused on HCPs’ work with patients, and Whole Health for You and Me, which is about HCPs’ personal well-being.19 We also reviewed a publication describing the history of whole health and patient-facing online whole health tools.2,19
Each document was reviewed for key elements related to SDM, patient-centered care, and whole health. Using the 53 elements identified by Bomhof-Roordink et al, we reviewed and compared each element to the whole health materials to create the integrated model of SDM and whole health. We iteratively discussed and organized the elements until we reached consensus.
SDM and Whole Health Alignment
We created an integrated model of SDM for LCS within the context of the VA whole health initiative. This integrated model is directed at HCPs who would likely engage patients in discussions of LCS, including primary care practitioners and nurse coordinators. The model includes 3 steps for HCPs to follow that align SDM within whole health: (1) frame the conversation and partner with the patient; (2) share clinical perspective and elicit patient values; and (3) deliberate and decide together. For each step, the SDM elements, whole health elements, and integration of SDM and whole health are provided. Table 1 provides an overview of the similarities and differences between SDM and whole health. Example phrases that merge SDM and whole health for HCPs to use in patient conversations about LCS are included in Table 2.


STEP 1. FRAME THE CONVERSATION AND PARTNER WITH THE PATIENT
Shared decision-making. Traditional SDM literature includes an initial step of letting patients know that there is a choice to be made between ≥ 2 clinical options.4 Ancillary elements of this first step include asking patients their preferences about the degree to which they want to be involved in SDM and about how they like to receive information (eg, verbal, written, video). These steps open the SDM conversation and ensure the patient and HCP are on the same page before moving forward. For example, the US Agency for Healthcare Research and Quality SHARE model’s first step is for HCPs to communicate that choices exist and to invite the patient to be involved in decisions.20 Similarly, Elwyn’s 3-step SDM model begins with establishing that a choice exists and inviting patient input on making that choice.17
Whole health. Patients are encouraged to play an active role in their health care. Through whole health programs such as Taking Charge of My Life and Health, patients explore their values and set self-care goals.21 HCP whole health trainings teach and reinforce communication skills, including SDM, listening skills, and motivational interviewing.19
Shared decision-making/whole health integration. SDM and whole health both prioritize respect, compassion, and patients’ expertise. They focus on the patient-HCP relationship with an emphasis on fostering egalitarian interactions. HCPs frame the SDM conversation and partner with the patient so they know what to expect and who will be involved. This conversation is framed from the outset as a collaborative discussion. HCPs empower the patient to play an active role in decision-making and help them understand why their engagement is critical.
STEP 2. SHARE CLINICAL PERSPECTIVE AND ELICIT PATIENT VALUES
Shared decision-making. HCPs share clinical perspective on LCS tailored to individual patients while explicitly inviting the patient to share their preferences and values when thinking about whether to undergo LCS. HCPs give a balanced description of LCS, including the benefits and harms, tailored to the patient’s unique information needs and questions. Sharing clinical perspective also includes describing treatment options, the most common element across SDM models.4 Decision aids, which provide unbiased information and include a values clarification exercise, may be helpful in sharing clinical perspectives and clarifying patient values related to the trade-offs of LCS.22 For example, the VA National Center for Health Promotion and Disease Prevention developed a LCS decision aid to be used for SDM for LCS.
Whole health. The conversation shifts from “What is the matter with you?” to “What matters to you?” starting with the patient’s goals and priorities rather than disease prevention, diagnosis, and treatment.2 Several whole health tools exist, including the Personal Health Inventory, used to identify what matters most to patients and understand their current well-being and self-care.23 Using the inventory, the patient and their health care team develop the patient’s personal health plan.24 Additionally, whole health trains HCPs to reflect on their own attitudes and biases when providing clinical care.
Shared decision-making/whole health integration. The LCS conversation can build on other whole health-related conversations with a HCP or other team members. HCPs can reference the patient’s personal health plan for documentation of the patient’s preferences, values, and goals in the electronic medical record. During this process, HCPs can give space for patients to discuss factors in their life and experiences that impact their perspective and decision-making. For example, patient concerns could be explored here, including fear of a cancer diagnosis, stigma around smoking, and fears around the screening and/or treatment process. HCPs may ask, “What matters most to you when making this decision?” Finally, by sharing clinical information, HCPs will focus on patient values to help overcome their own biases toward a desire for LCS. HCPs, similar to the rest of the US public, tend to hold highly favorable attitudes toward cancer screening as well as misconceptions about the magnitude of benefits from screening.13
STEP 3. DELIBERATE AND DECIDE TOGETHER
Shared decision-making. Decision-making is almost always considered the last SDM step.4 In the final step, the patient and HCP discuss the options (ie, to screen or not to screen) considering the patient’s values and preferences, and patients decide with their HCP whether they will undergo LCS. Patients may decide they need more time to think about these options. As part of deliberation, HCPs assess what other information patients may need to arrive at a decision. Family members, friends, or peers may be included in making the final decision.
Whole health. In Whole health, decisions also may include the entire health care team and other individuals important to the patient (eg, family, friends). Integration across different health care settings is also considered a key whole health element. Finally, whole health focuses on long-term relationships with patients; thus, the LCS SDM process is situated within longer term relationship building and patient empowerment, both of which will facilitate partnering with the patient in future conversations about other decisions.
Shared decision-making/whole health integration. Both SDM and whole health emphasize partnership with the patient in making a final decision. There is also focus on decision-making as an ongoing process. Deciding whether LCS is the best choice might include naming and addressing emotions, voicing questions not raised, and exploring whether screening fits the patient’s goals, values, and life context. HCPs may give guidance, but patients retain the authority to make decisions. The goal is to empower patients to know that the only right decision is the one right for them and they will be supported.
Limitations
This article describes a VA practice program and was not a formal research study. Further work is needed to evaluate the presented strategies. Additionally, we did not conduct a systematic literature review and thus elements of SDM and whole health may not be exhaustive.
CONCLUSIONS
This article describes the alignment of 2 distinct VA initiatives, whole health and SDM for LCS. The goal was to reduce known barriers to SDM, such as competing demands, limited time, and lack of familiarity with and training in SDM.11-13 These concepts are well aligned. This integrated model is the first step in informing the development of a HCP training program and materials as part of a multilevel strategy that our team is using to implement SDM for LCS in VISN 1.16 The final training and materials resulting from this work were delivered to LCSCs in 3 ways: (1) a series of 3 interactive group training sessions, including didactic elements, role play, and time for open discussion; (2) 1-on-1 academic detailing; and (3) educational handouts. In academic detailing, a member of the research team trained in academic detailing met virtually with each nurse coordinator, identified that individual’s barriers to SDM, and used the training materials to highlight messages to overcome those barriers; follow-up calls provided a forum for discussing progress and overcoming additional challenges. Although this article focused specifically on whole health and SDM, the conceptual alignment process strategy can be applied to other implementations of multiple initiatives.
The landmark Crossing the Quality Chasm report from the National Academy of Medicine identified patient- centered care as essential to health care quality. The report defines patientcentered care as “respectful of and responsive to individual patient preferences, needs, and values.”1 Many health care systems, including the Veterans Health Administration, are transforming to a patient-centered model of care.2 The US Department of Veterans Affairs (VA) Whole Health System of Care initiative is a system-wide, cultural transformation. Within whole health, what matters most to the patient—including their preferences, needs, and values—is foundational to health care and meant to be essential in every clinical encounter. Whole health implementation includes a progressive rollout with health care practitioner (HCP) trainings across the VA.2
Shared decision-making (SDM) is a different but aligned patient-centered care concept. SDM is a process through which a decision or care plan, based on patients’ preferences, needs, and values, is made or developed.3-5 SDM is ideal in situations with equipoise (decisions with equivalent choices), individualized risks, and/or greater uncertainty of the net benefit, such as with lung cancer screening (LCS).3 SDM for LCS is required by the US Centers for Medicare and Medicaid Services and has been adopted by many US health care systems, including the VA.6,7 Early detection of lung cancer can reduce death by 20% at the population level.8 However, at the patient level there is wide variation in the risk of developing lung cancer and a range of potential harms.8 LCS follow-up procedures may be more invasive than with other cancer screenings. Thus, there is concern about the risk of false-positive results leading to unnecessary care or complications.8 Given this balance between benefit and harm and the differing patient value on the trade-offs of LCS, an individualized, patient-centered approach is essential when deciding whether LCS is the right choice for a specific patient.
Despite the importance of LCS SDM, observational studies have shown poor implementation in clinical encounters.9,10 HCP barriers include competing demands, limited time, lack of familiarity with and training in SDM, and beliefs biasing screening over no screening.11-13 Additionally, HCPs may assume that patients want them to make the decision. However, research has shown that patients actually want to be more involved in their health care decisions.14 One suggested strategy to overcome these barriers is aligning SDM for LCS within an organization’s broader patient-centered initiatives.15
This project sought to align the need for SDM for LCS and the broader VA whole health initiative as part of a multilevel strategy to implement SDM for LCS across Veterans Integrated Service Network (VISN) 1.16
This article addresses HCP-level barriers. HCPs targeted are those typically involved in LCS. The VA utilizes LCS coordinators (LCSCs) in both centralized or consult models (in which LCSCs are involved in all aspects of screening) and hybrid models (in which primary care practitioners and LCSCs are both engaged in LCS tasks). The goal of this program was to generate areas of conceptual alignment between SDM and whole health as a first step in integrating these VA initiatives. This work was conducted as a foundation for an SDM for lung cancer HCP training and consultation initiative.
ALIGNMENT PROCESS
We reviewed relevant literature and resources for SDM and whole health. In reviewing the SDM literature, we included a sample of the most widely cited literature on the topic, and focused primarily on the systematic review by Bomhof-Roordink et al.4,5,17,18 This review provided a synthesis of SDM elements across SDM models and identified 53 different elements clustered into 24 components.4 The most common components were present in at least half of all SDM published models, including: make the decision, patient preferences, tailor information, deliberate, create choice awareness, and learn about the patient. Bomhof-Roordink et al provided the guiding framework for this conceptualization of SDM because that study included the available recent published SDM models.4
Second, published literature on VA whole health along with supplemental promotional and training materials were reviewed. The whole health materials included 2 sets of training slides developed for VA HCPs (available to VA employees): Implementing Whole Health in Clinical Care, which is focused on HCPs’ work with patients, and Whole Health for You and Me, which is about HCPs’ personal well-being.19 We also reviewed a publication describing the history of whole health and patient-facing online whole health tools.2,19
Each document was reviewed for key elements related to SDM, patient-centered care, and whole health. Using the 53 elements identified by Bomhof-Roordink et al, we reviewed and compared each element to the whole health materials to create the integrated model of SDM and whole health. We iteratively discussed and organized the elements until we reached consensus.
SDM and Whole Health Alignment
We created an integrated model of SDM for LCS within the context of the VA whole health initiative. This integrated model is directed at HCPs who would likely engage patients in discussions of LCS, including primary care practitioners and nurse coordinators. The model includes 3 steps for HCPs to follow that align SDM within whole health: (1) frame the conversation and partner with the patient; (2) share clinical perspective and elicit patient values; and (3) deliberate and decide together. For each step, the SDM elements, whole health elements, and integration of SDM and whole health are provided. Table 1 provides an overview of the similarities and differences between SDM and whole health. Example phrases that merge SDM and whole health for HCPs to use in patient conversations about LCS are included in Table 2.


STEP 1. FRAME THE CONVERSATION AND PARTNER WITH THE PATIENT
Shared decision-making. Traditional SDM literature includes an initial step of letting patients know that there is a choice to be made between ≥ 2 clinical options.4 Ancillary elements of this first step include asking patients their preferences about the degree to which they want to be involved in SDM and about how they like to receive information (eg, verbal, written, video). These steps open the SDM conversation and ensure the patient and HCP are on the same page before moving forward. For example, the US Agency for Healthcare Research and Quality SHARE model’s first step is for HCPs to communicate that choices exist and to invite the patient to be involved in decisions.20 Similarly, Elwyn’s 3-step SDM model begins with establishing that a choice exists and inviting patient input on making that choice.17
Whole health. Patients are encouraged to play an active role in their health care. Through whole health programs such as Taking Charge of My Life and Health, patients explore their values and set self-care goals.21 HCP whole health trainings teach and reinforce communication skills, including SDM, listening skills, and motivational interviewing.19
Shared decision-making/whole health integration. SDM and whole health both prioritize respect, compassion, and patients’ expertise. They focus on the patient-HCP relationship with an emphasis on fostering egalitarian interactions. HCPs frame the SDM conversation and partner with the patient so they know what to expect and who will be involved. This conversation is framed from the outset as a collaborative discussion. HCPs empower the patient to play an active role in decision-making and help them understand why their engagement is critical.
STEP 2. SHARE CLINICAL PERSPECTIVE AND ELICIT PATIENT VALUES
Shared decision-making. HCPs share clinical perspective on LCS tailored to individual patients while explicitly inviting the patient to share their preferences and values when thinking about whether to undergo LCS. HCPs give a balanced description of LCS, including the benefits and harms, tailored to the patient’s unique information needs and questions. Sharing clinical perspective also includes describing treatment options, the most common element across SDM models.4 Decision aids, which provide unbiased information and include a values clarification exercise, may be helpful in sharing clinical perspectives and clarifying patient values related to the trade-offs of LCS.22 For example, the VA National Center for Health Promotion and Disease Prevention developed a LCS decision aid to be used for SDM for LCS.
Whole health. The conversation shifts from “What is the matter with you?” to “What matters to you?” starting with the patient’s goals and priorities rather than disease prevention, diagnosis, and treatment.2 Several whole health tools exist, including the Personal Health Inventory, used to identify what matters most to patients and understand their current well-being and self-care.23 Using the inventory, the patient and their health care team develop the patient’s personal health plan.24 Additionally, whole health trains HCPs to reflect on their own attitudes and biases when providing clinical care.
Shared decision-making/whole health integration. The LCS conversation can build on other whole health-related conversations with a HCP or other team members. HCPs can reference the patient’s personal health plan for documentation of the patient’s preferences, values, and goals in the electronic medical record. During this process, HCPs can give space for patients to discuss factors in their life and experiences that impact their perspective and decision-making. For example, patient concerns could be explored here, including fear of a cancer diagnosis, stigma around smoking, and fears around the screening and/or treatment process. HCPs may ask, “What matters most to you when making this decision?” Finally, by sharing clinical information, HCPs will focus on patient values to help overcome their own biases toward a desire for LCS. HCPs, similar to the rest of the US public, tend to hold highly favorable attitudes toward cancer screening as well as misconceptions about the magnitude of benefits from screening.13
STEP 3. DELIBERATE AND DECIDE TOGETHER
Shared decision-making. Decision-making is almost always considered the last SDM step.4 In the final step, the patient and HCP discuss the options (ie, to screen or not to screen) considering the patient’s values and preferences, and patients decide with their HCP whether they will undergo LCS. Patients may decide they need more time to think about these options. As part of deliberation, HCPs assess what other information patients may need to arrive at a decision. Family members, friends, or peers may be included in making the final decision.
Whole health. In Whole health, decisions also may include the entire health care team and other individuals important to the patient (eg, family, friends). Integration across different health care settings is also considered a key whole health element. Finally, whole health focuses on long-term relationships with patients; thus, the LCS SDM process is situated within longer term relationship building and patient empowerment, both of which will facilitate partnering with the patient in future conversations about other decisions.
Shared decision-making/whole health integration. Both SDM and whole health emphasize partnership with the patient in making a final decision. There is also focus on decision-making as an ongoing process. Deciding whether LCS is the best choice might include naming and addressing emotions, voicing questions not raised, and exploring whether screening fits the patient’s goals, values, and life context. HCPs may give guidance, but patients retain the authority to make decisions. The goal is to empower patients to know that the only right decision is the one right for them and they will be supported.
Limitations
This article describes a VA practice program and was not a formal research study. Further work is needed to evaluate the presented strategies. Additionally, we did not conduct a systematic literature review and thus elements of SDM and whole health may not be exhaustive.
CONCLUSIONS
This article describes the alignment of 2 distinct VA initiatives, whole health and SDM for LCS. The goal was to reduce known barriers to SDM, such as competing demands, limited time, and lack of familiarity with and training in SDM.11-13 These concepts are well aligned. This integrated model is the first step in informing the development of a HCP training program and materials as part of a multilevel strategy that our team is using to implement SDM for LCS in VISN 1.16 The final training and materials resulting from this work were delivered to LCSCs in 3 ways: (1) a series of 3 interactive group training sessions, including didactic elements, role play, and time for open discussion; (2) 1-on-1 academic detailing; and (3) educational handouts. In academic detailing, a member of the research team trained in academic detailing met virtually with each nurse coordinator, identified that individual’s barriers to SDM, and used the training materials to highlight messages to overcome those barriers; follow-up calls provided a forum for discussing progress and overcoming additional challenges. Although this article focused specifically on whole health and SDM, the conceptual alignment process strategy can be applied to other implementations of multiple initiatives.
- Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. The National Academies Press; 2001. doi:10.17226/10027
- Bokhour BG, Haun JN, Hyde J, Charns M, Kligler B. Transforming the Veterans Affairs to a whole health system of care: time for action and research. Med Care. 2020;58:295- 300. doi:10.1097/MLR.0000000000001316
- Elwyn G, Frosch D, Rollnick S. Dual equipoise shared decision making: definitions for decision and behaviour support interventions. Implement Sci. 2009;4:75. doi:7510.1186/1748-5908-4-75
- Bomhof-Roordink H, Gärtner FR, Stiggelbout AM, Pieterse AH. Key components of shared decision making models: a systematic review. BMJ Open. 2019;9:e031763. doi:10.1136/bmjopen-2019-031763
- Charles C, Gafni A, Whelan T. Decision-making in the physician- patient encounter: revisiting the shared treatment decision-making model. Soc Sci Med. 1999;49:651-661. doi:10.1016/s0277-9536(99)00145-8
- Moyer VA; US Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160:330- 338. doi:10.7326/m13-2771
- Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT). February 10, 2022. Accessed February 7, 2025. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=304
- Aberle DR, Adams AM, Berg CD, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365:395-409. doi:10.1056/NEJMoa1102873
- Slatore CG, Wiener RS. Pulmonary nodules: a small problem for many, severe distress for some, and how to communicate about it. Chest. 2018;153:1004-1015. doi:10.1016/j.chest.2017.10.013
- Nishi SPE, Lowenstein LM, Mendoza TR, et al. Shared decision-making for lung cancer screening: how well are we “sharing”? Chest. 2021;160:330-340. doi:10.1016/j.chest.2021.01.041
- Wiener RS, Koppelman E, Bolton R, et al. Patient and clinician perspectives on shared decision-making in early adopting lung cancer screening programs: a qualitative study. J Gen Intern Med. 2018;33:1035-1042. doi:10.1007/s11606-018-4350-9
- Melzer AC, Golden SE, Ono SS, Datta S, Triplette M, Slatore CG. “We just never have enough time”: clinician views of lung cancer screening processes and implementation. Ann Am Thorac Soc. 2020. doi:10.1513/AnnalsATS.202003-262OC
- Schwartz LM, Woloshin S, Fowler FJ Jr, Welch HG. Enthusiasm for cancer screening in the United States. JAMA. 2004;291:71-78. doi:10.1001/jama.291.1.71
- Lown BA, Rosen J, Marttila J. An agenda for improving compassionate care: a survey shows about half of patients say such care is missing. Health Aff (Millwood). 2011;30:1772-1778. doi:10.1377/hlthaff.2011.0539
- Scholl I, LaRussa A, Hahlweg P, Kobrin S, Elwyn G. Organizational- and system-level characteristics that influence implementation of shared decision-making and strategies to address them - a scoping review. Implement Sci. 2018;13:40. doi:10.1186/s13012-018-0731-z
- Khanna A, Fix GM, Anderson E, et al. Towards a framework for patient-centred care coordination: a scoping review protocol. BMJ Open. 2022;12:e066808. doi:10.1136/bmjopen-2022-066808
- Elwyn G, Durand MA, Song J, et al. A three-talk model for shared decision making: multistage consultation process. BMJ. 2017;359:j4891. doi:10.1136/bmj.j4891
- Makoul G, Clayman ML. An integrative model of shared decision making in medical encounters. Patient Educ Couns. 2006;60:301-312. doi:10.1016/j.pec.2005.06.010
- Whole Health. US Department of Veterans Affairs. Accessed April 14, 2025. https://www.va.gov/wholehealth/
- Agency for Healthcare Research and Quality. The SHARE approach. Accessed April 14, 2025. https://www.ahrq.gov/health-literacy/professional-training/shared-decision/index.html
- Abadi MH, Barker AM, Rao SR, Orner M, Rychener D, Bokhour BG. Examining the impact of a peer-led group program for veteran engagement and well-being. J Altern Complement Med. 2021;27:S37-S44. doi:10.1089/acm.2020.0124
- Stacey D, Lewis KB, Smith M, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2024;1:CD001431. doi:10.1002/14651858.CD001431.pub6
- US Department of Veterans Affairs, Veterans Health Administration, Office of Patient Centered Care and Cultural Transformation. Personal health inventory. Revised April 2019. Accessed April 14, 2025. https://www.va.gov/wholehealth/docs/10-773_PHI_July2019_508.pdf
- US Department of Veterans Affairs. Build your personal health plan. Updated July 24, 2024. Accessed April 14, 2025. https://www.va.gov/wholehealth/phi.asp
- Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. The National Academies Press; 2001. doi:10.17226/10027
- Bokhour BG, Haun JN, Hyde J, Charns M, Kligler B. Transforming the Veterans Affairs to a whole health system of care: time for action and research. Med Care. 2020;58:295- 300. doi:10.1097/MLR.0000000000001316
- Elwyn G, Frosch D, Rollnick S. Dual equipoise shared decision making: definitions for decision and behaviour support interventions. Implement Sci. 2009;4:75. doi:7510.1186/1748-5908-4-75
- Bomhof-Roordink H, Gärtner FR, Stiggelbout AM, Pieterse AH. Key components of shared decision making models: a systematic review. BMJ Open. 2019;9:e031763. doi:10.1136/bmjopen-2019-031763
- Charles C, Gafni A, Whelan T. Decision-making in the physician- patient encounter: revisiting the shared treatment decision-making model. Soc Sci Med. 1999;49:651-661. doi:10.1016/s0277-9536(99)00145-8
- Moyer VA; US Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160:330- 338. doi:10.7326/m13-2771
- Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT). February 10, 2022. Accessed February 7, 2025. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=304
- Aberle DR, Adams AM, Berg CD, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365:395-409. doi:10.1056/NEJMoa1102873
- Slatore CG, Wiener RS. Pulmonary nodules: a small problem for many, severe distress for some, and how to communicate about it. Chest. 2018;153:1004-1015. doi:10.1016/j.chest.2017.10.013
- Nishi SPE, Lowenstein LM, Mendoza TR, et al. Shared decision-making for lung cancer screening: how well are we “sharing”? Chest. 2021;160:330-340. doi:10.1016/j.chest.2021.01.041
- Wiener RS, Koppelman E, Bolton R, et al. Patient and clinician perspectives on shared decision-making in early adopting lung cancer screening programs: a qualitative study. J Gen Intern Med. 2018;33:1035-1042. doi:10.1007/s11606-018-4350-9
- Melzer AC, Golden SE, Ono SS, Datta S, Triplette M, Slatore CG. “We just never have enough time”: clinician views of lung cancer screening processes and implementation. Ann Am Thorac Soc. 2020. doi:10.1513/AnnalsATS.202003-262OC
- Schwartz LM, Woloshin S, Fowler FJ Jr, Welch HG. Enthusiasm for cancer screening in the United States. JAMA. 2004;291:71-78. doi:10.1001/jama.291.1.71
- Lown BA, Rosen J, Marttila J. An agenda for improving compassionate care: a survey shows about half of patients say such care is missing. Health Aff (Millwood). 2011;30:1772-1778. doi:10.1377/hlthaff.2011.0539
- Scholl I, LaRussa A, Hahlweg P, Kobrin S, Elwyn G. Organizational- and system-level characteristics that influence implementation of shared decision-making and strategies to address them - a scoping review. Implement Sci. 2018;13:40. doi:10.1186/s13012-018-0731-z
- Khanna A, Fix GM, Anderson E, et al. Towards a framework for patient-centred care coordination: a scoping review protocol. BMJ Open. 2022;12:e066808. doi:10.1136/bmjopen-2022-066808
- Elwyn G, Durand MA, Song J, et al. A three-talk model for shared decision making: multistage consultation process. BMJ. 2017;359:j4891. doi:10.1136/bmj.j4891
- Makoul G, Clayman ML. An integrative model of shared decision making in medical encounters. Patient Educ Couns. 2006;60:301-312. doi:10.1016/j.pec.2005.06.010
- Whole Health. US Department of Veterans Affairs. Accessed April 14, 2025. https://www.va.gov/wholehealth/
- Agency for Healthcare Research and Quality. The SHARE approach. Accessed April 14, 2025. https://www.ahrq.gov/health-literacy/professional-training/shared-decision/index.html
- Abadi MH, Barker AM, Rao SR, Orner M, Rychener D, Bokhour BG. Examining the impact of a peer-led group program for veteran engagement and well-being. J Altern Complement Med. 2021;27:S37-S44. doi:10.1089/acm.2020.0124
- Stacey D, Lewis KB, Smith M, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2024;1:CD001431. doi:10.1002/14651858.CD001431.pub6
- US Department of Veterans Affairs, Veterans Health Administration, Office of Patient Centered Care and Cultural Transformation. Personal health inventory. Revised April 2019. Accessed April 14, 2025. https://www.va.gov/wholehealth/docs/10-773_PHI_July2019_508.pdf
- US Department of Veterans Affairs. Build your personal health plan. Updated July 24, 2024. Accessed April 14, 2025. https://www.va.gov/wholehealth/phi.asp
When Patient-Centered Care Initiatives Align: Integrating VA Whole Health and Shared Decision-Making for Lung Cancer Screening
When Patient-Centered Care Initiatives Align: Integrating VA Whole Health and Shared Decision-Making for Lung Cancer Screening
Painful Flesh-Colored Nodule on the Shoulder
Painful Flesh-Colored Nodule on the Shoulder
THE DIAGNOSIS: Dermatofibrosarcoma Protuberans
The histologic findings showed fascicular proliferation of relatively monomorphic spindle cells with extensive entrapment of collagen and adipocytes. Immunohistochemical staining showed that the lesional cells were diffusely positive for CD34 and negative for SOX10, S100, desmin, and factor XIIIa. The decision was made to perform cytogenetic testing with fluorescence in situ hybridization to evaluate for the presence of platelet-derived growth factor receptor beta (PDGFB) polypeptide rearrangement, a key biomarker known to be positive in most patients with dermatofibrosarcoma protuberans (DFSP).1 This rearrangement results in overproduction of PDGFB, continuous activation of platelet-derived growth factor receptor beta, cellular proliferation, and tumor formation.2 In our patient, results were positive for the PDGFB polypeptide rearrangement, which confirmed suspected diagnosis of DFSP with fibrous histiocytoma like morphology. The patient was referred for Mohs micrographic surgery for proper management.
Dermatofibrosarcoma protuberans is a rare soft-tissue tumor that involves the dermis, subcutaneous fat, and sometimes muscle and fascia.2 Dermatofibrosarcoma protuberans primarily affects young to middle-aged adults, with a slight predilection for individuals in the third to fifth decades of life.3 Lesions preferentially involve the trunk, particularly the shoulder and chest regions, and manifest as poorly circumscribed, locally aggressive mesenchymal neoplasms with a high local recurrence rate but low metastatic potential.4,5 Clinically, the lesions appear as flesh-colored, rubbery plaques or nodules. A diagnosis of DFSP requires a high index of clinical suspicion, and histologic, immunohistochemical, and molecular testing usually are required for confirmation.
On histopathologic examination, DFSP classically demonstrates uniform, spindle-shaped cells that traditionally are arranged in an intersecting pattern and primarily are based in the dermis (Figure 1).5 Infiltration into the underlying tissue is a common feature, with neoplastic extensions causing a classic honeycomb pattern6 that also can be seen in diffuse neurofibroma and may cause diagnostic challenges; however, the immunohistology staining of neurofibroma differs from DFSP in that it stains positive for CD34, SOX-100, and S100, while DFSP has strong and diffuse CD34 immunoreactivity with negative immunostaining for SOX10, S100, desmin, and factor XIIIa.2,6
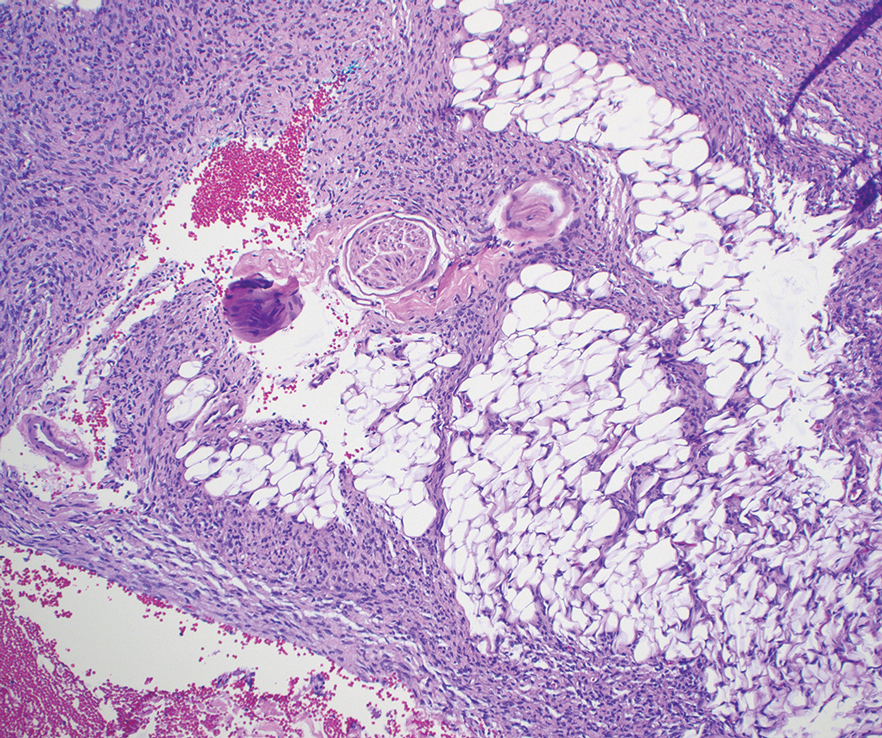
Dermatofibrosarcoma protuberans can cause considerable fat infiltration compared to other soft-tissue neoplasms, making this finding suspicious for—if not characteristic of—DFSP. Collagen trapping also can be observed; however, this is more pathognomonic in cellular fibrous histiocytoma, which is a distinct clinical variant of dermatofibromas. Due to its similarity to other lesions, histopathologic examination along with immunostaining can assist in differentiating and accurately diagnosing DFSP.6
Cellular fibrous histiocytoma (CFH), a distinct clinical variant of dermatofibromas, is a benign tumor of mesenchymal origin that occurs more commonly on the trunk, arms, and legs. On histologic examination, CFH is composed of spindle-shaped cells with variable amounts of eosinophilic cytoplasm and small, oval-shaped eosinophilic nuclei and collagen trapping (Figure 2).7,8 Most CFHs occupy the superficial dermis but can extend into the deep reticular dermis, thus mimicking the honeycomb pattern seen in DFSP. This neoplasm can show a similar architecture to DFSP, which is why further investigation including cytogenetics and immunohistochemical staining can help differentiate the two conditions. Cellular fibrous histiocytoma typically stains negative for CD34 and positive for factor XIIIa.9 However, CD34 can be positive in a subset of CFHs, with a considerable subset showing peripheral CD34 positivity and a smaller subset showing central CD34 the positivity.10 This suggests that CD34 cannot be the only factor differentiating these 2 lesions in making a proper dermatopathologic diagnosis.
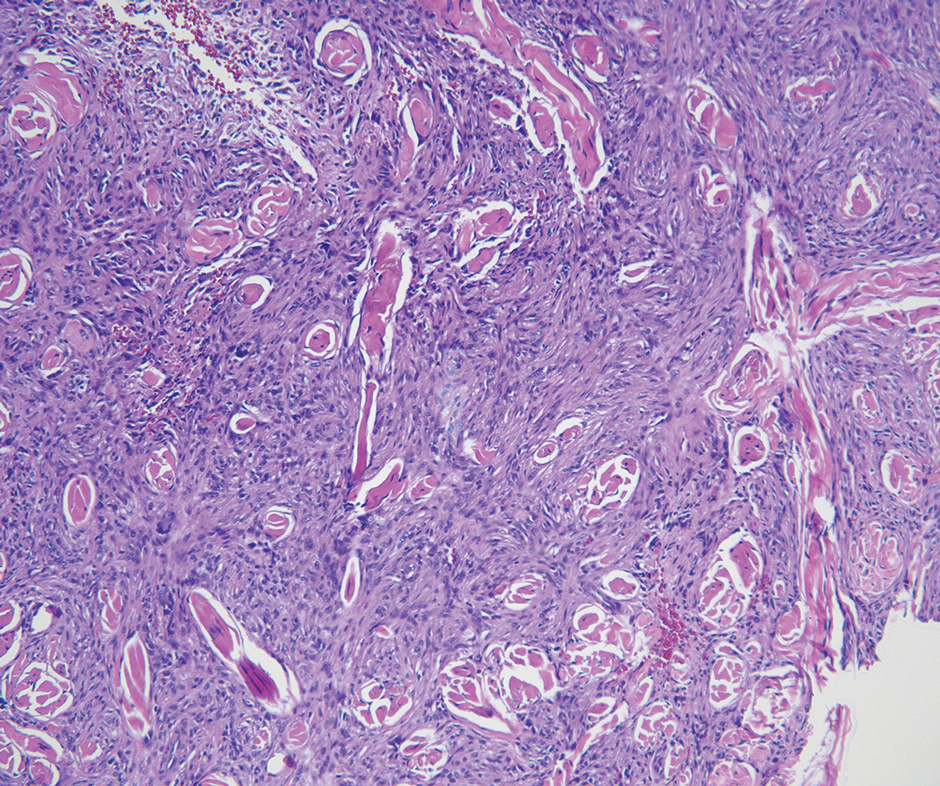
Solitary fibrous tumor (SFT) is a rare mesenchymal tumor that can occur anywhere on the body and typically manifests as a deep, painless, enlarging mass in adults aged 50 to 60 years.11 On histologic examination, SFT consists of randomly arranged cells with a spindle or ovoid shape within a collagenous stroma intermixed with blood vessels with a characteristic staghorn shape (Figure 3).11 Low-grade SFT shows a patternless arrangement with spindle cells, a low number of mitotic figures, and vessels with a staghorn appearance compared to high-grade SFT, which shows hypercellularity with nuclear pleomorphism and a high number of mitotic figures.11 Solitary fibrous tumors are positive for CD34 and STAT-6 and negative for CD31 and typically demonstrate NGFI-A binding protein 2 (NAB2)—signal transducer and activator of transcription 6 (STAT 6) gene fusion.11
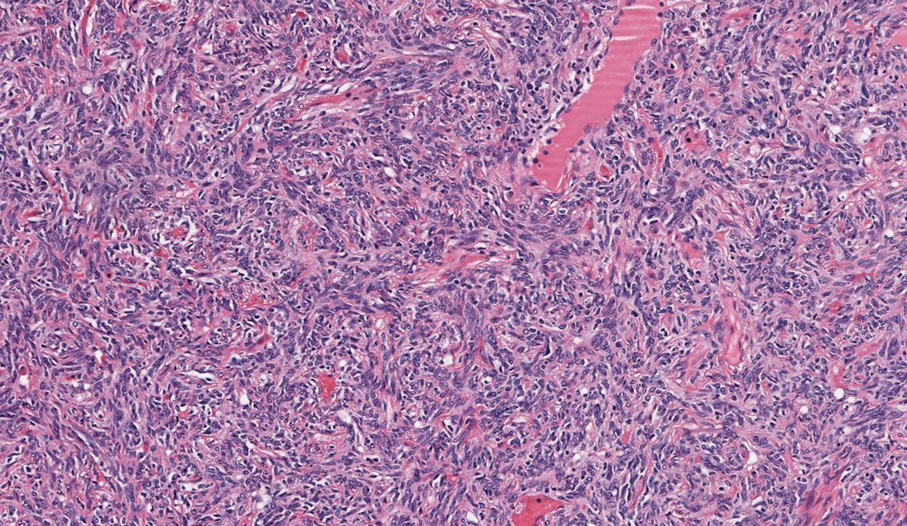
Spindle-cell lipomas are rare, benign, slow-growing, lipomatous tumors that typically manifest in men aged 40 to 70 years.12 These lesions originate most frequently in the subcutaneous tissue of the upper back, posterior neck, and shoulders. The histologic growth pattern of spindle-cell lipomas can mimic other spindle-cell and myxoid tumors, which is why cytogenetic analysis is crucial for differentiating these lesions. On histologic examination, spindle-cell lipomas exhibit a mixture of mature adipocytes, uniform spindle cells, and collagen bundles (eFigure). Spindle-cell lipoma stains positive for CD34 but negative for S100.13 In addition, spindle-cell lipomas tend to show structural rearrangements (mainly deletions) of the long arm of chromosome 13 or even losses of whole chromosome 13, which contains the retinoblastoma (RB1) gene.13
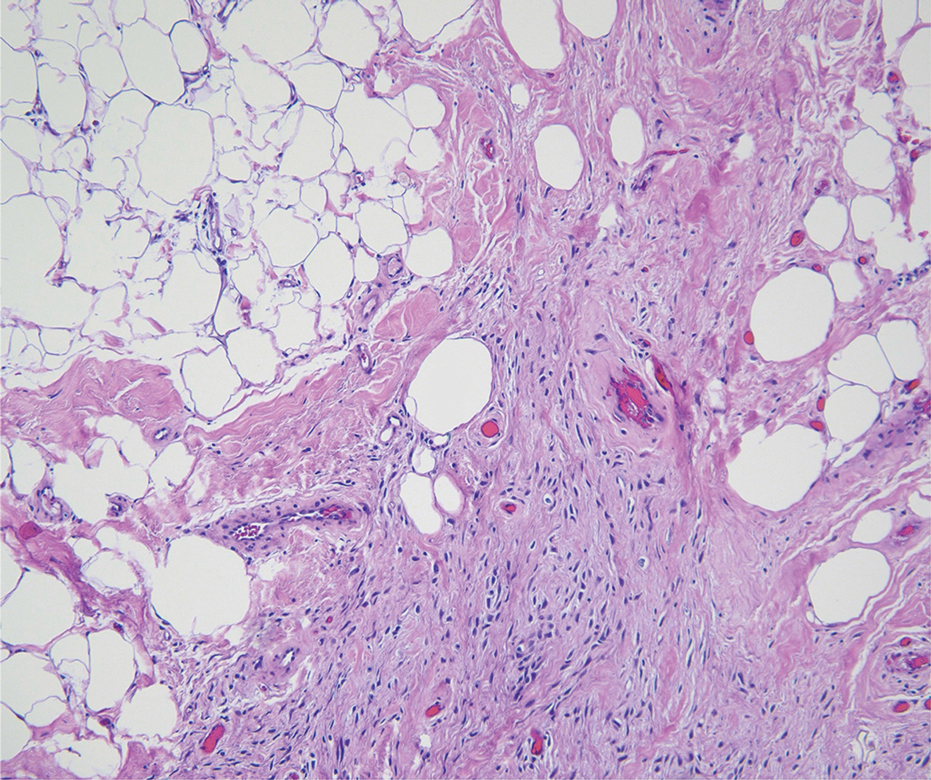
Pleomorphic dermal sarcoma is a rare mesenchymal tumor that can appear clinically and histologically similar to atypical fibroxanthoma.14 This lesion often manifests in elderly patients and is strongly associated with chronic sun exposure.15 Pleomorphic dermal sarcoma is a locally aggressive tumor with metastatic potential to the skin or lymph nodes. On histologic examination, these tumors exhibit pleomorphic atypical epithelioid or spindle cells as well as multinucleated tumor giant cells with possible tumor necrosis, lymphovascular invasion, or perineural infiltration (Figure 4). Pleomorphic dermal sarcoma, typically a diagnosis of exclusion, requires immunohistochemistry to aid in proper identification.16 These lesions stain positive for CD10 and negative for cytokeratins, desmin, HMB45, CD34, p63, p40, SOX10, and S100.15,16
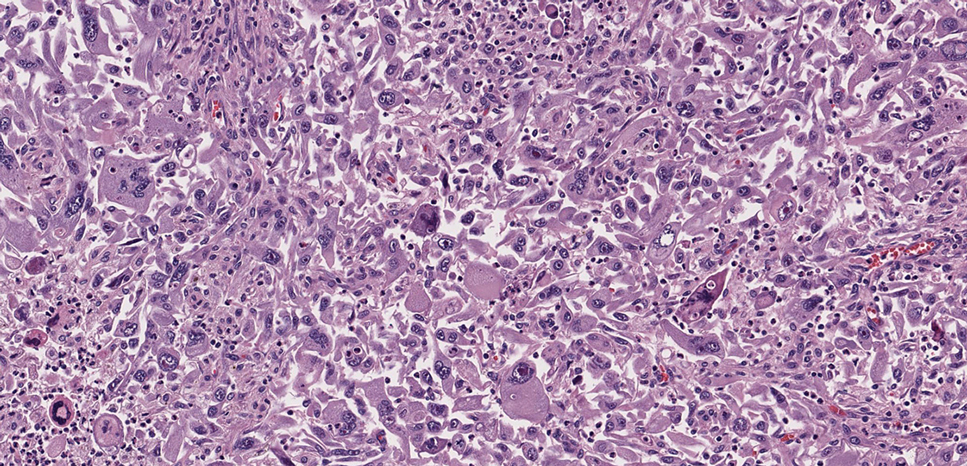
- Ugurel S, Kortmann R, Mohr P, et al. S1 guidelines for dermatofibrosarcoma protuberans (DFSP)—update 2018. J Dtsch Dermatol Ges. 2019;17:663-668. doi:10.1111/ddg.13849
- Brooks J, Ramsey ML. Dermatofibrosarcoma protuberans. StatPearls Publishing; 2024. Updated April 18, 2024. Accessed April 30, 2025.
- Bowne WB, Antonescu CR, Leung DH, et al. Dermatofibrosarcoma protuberans: a clinicopathologic analysis of patients treated and followed at a single institution. Cancer. 2000;88:2711-2720.
- Lim SX, Ramaiya A, Levell NJ, et al. Review of dermatofibrosarcoma protuberans. Clin Exp Dermatol. 2022;48:297-302. doi:10.1093/ced/llac111
- Trinidad CM, Wangsiricharoen S, Prieto VG, et al. Rare variants of dermatofibrosarcoma protuberans: clinical, histologic, and molecular features and diagnostic pitfalls. Dermatopathology. 2023;10:54-62. doi:10.3390/dermatopathology10010008
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752. doi:10.3390/jcm9061752
- Tsunoda K, Oikawa H, Maeda F, et al. A case of cellular fibrous histiocytoma on the right elbow with repeated relapse within a short period. Case Rep Dermatol. 2015;7:10–16. https://doi.org/10.1159/000371790
- Calonje E, Mentzel T, Fletcher CD. Cellular benign fibrous histiocytoma. Clinicopathologic analysis of 74 cases of a distinctive variant of cutaneous fibrous histiocytoma with frequent recurrence. Am J Surg Pathol. 1994;18:668-676.
- Goldblum JR, Tuthill RJ. CD34 and factor-XIIIa immunoreactivity in dermatofibrosarcoma protuberans and dermatofibroma. Am J Dermatopathology. 1997;19:147-153. doi:10.1097/00000372-199704000-00008
- Volpicelli ER, Fletcher CD. Desmin and CD34 positivity in cellular fibrous histiocytoma: an immunohistochemical analysis of 100 cases. J Cutan Pathol. 2012;39:747-752. doi:10.1111/j.1600-0560.2012.01944.x
- Martin-Broto J, Mondaza-Hernandez JL, Moura DS, et al. A comprehensive review on solitary fibrous tumor: new insights for new horizons. Cancers (Basel). 2021;13:2913. doi:10.3390/cancers13122913
- Machol JA, Cusic JG, O’Connor EA, et al. Spindle cell lipoma of the neck: review of the literature and case report. Plast Reconstr Surg Glob Open. 2015;3:E550. doi:10.1097/GOX.0000000000000405
- Domanski HA, Carlén B, Jonsson K, et al. Distinct cytologic features of spindle cell lipoma. a cytologic-histologic study with clinical, radiologic, electron microscopic, and cytogenetic correlations. Cancer. 2001;93:381-389. doi:10.1002/cncr.10142
- Devine RL, Cameron A, Holden AM, et al. The pleomorphic dermal sarcoma: its management, follow-up and the need for more guidance. Adv Oral Maxillofac Surg. 2021;2:100046. doi:10.1016 /j.adoms.2021.100046
- Seretis K, Klaroudas A, Galani V, et al. Pleomorphic dermal sarcoma: it might be rare but it exists [published online August 4, 2023]. J Surg Case Rep. doi:10.1093/jscr/rjad374
- Miller K, Goodlad JR, Brenn T. Pleomorphic dermal sarcoma. Am J Surg Pathol. 2012;36:1317-1326. doi:10.1097/pas.0b013e31825359e1
THE DIAGNOSIS: Dermatofibrosarcoma Protuberans
The histologic findings showed fascicular proliferation of relatively monomorphic spindle cells with extensive entrapment of collagen and adipocytes. Immunohistochemical staining showed that the lesional cells were diffusely positive for CD34 and negative for SOX10, S100, desmin, and factor XIIIa. The decision was made to perform cytogenetic testing with fluorescence in situ hybridization to evaluate for the presence of platelet-derived growth factor receptor beta (PDGFB) polypeptide rearrangement, a key biomarker known to be positive in most patients with dermatofibrosarcoma protuberans (DFSP).1 This rearrangement results in overproduction of PDGFB, continuous activation of platelet-derived growth factor receptor beta, cellular proliferation, and tumor formation.2 In our patient, results were positive for the PDGFB polypeptide rearrangement, which confirmed suspected diagnosis of DFSP with fibrous histiocytoma like morphology. The patient was referred for Mohs micrographic surgery for proper management.
Dermatofibrosarcoma protuberans is a rare soft-tissue tumor that involves the dermis, subcutaneous fat, and sometimes muscle and fascia.2 Dermatofibrosarcoma protuberans primarily affects young to middle-aged adults, with a slight predilection for individuals in the third to fifth decades of life.3 Lesions preferentially involve the trunk, particularly the shoulder and chest regions, and manifest as poorly circumscribed, locally aggressive mesenchymal neoplasms with a high local recurrence rate but low metastatic potential.4,5 Clinically, the lesions appear as flesh-colored, rubbery plaques or nodules. A diagnosis of DFSP requires a high index of clinical suspicion, and histologic, immunohistochemical, and molecular testing usually are required for confirmation.
On histopathologic examination, DFSP classically demonstrates uniform, spindle-shaped cells that traditionally are arranged in an intersecting pattern and primarily are based in the dermis (Figure 1).5 Infiltration into the underlying tissue is a common feature, with neoplastic extensions causing a classic honeycomb pattern6 that also can be seen in diffuse neurofibroma and may cause diagnostic challenges; however, the immunohistology staining of neurofibroma differs from DFSP in that it stains positive for CD34, SOX-100, and S100, while DFSP has strong and diffuse CD34 immunoreactivity with negative immunostaining for SOX10, S100, desmin, and factor XIIIa.2,6

Dermatofibrosarcoma protuberans can cause considerable fat infiltration compared to other soft-tissue neoplasms, making this finding suspicious for—if not characteristic of—DFSP. Collagen trapping also can be observed; however, this is more pathognomonic in cellular fibrous histiocytoma, which is a distinct clinical variant of dermatofibromas. Due to its similarity to other lesions, histopathologic examination along with immunostaining can assist in differentiating and accurately diagnosing DFSP.6
Cellular fibrous histiocytoma (CFH), a distinct clinical variant of dermatofibromas, is a benign tumor of mesenchymal origin that occurs more commonly on the trunk, arms, and legs. On histologic examination, CFH is composed of spindle-shaped cells with variable amounts of eosinophilic cytoplasm and small, oval-shaped eosinophilic nuclei and collagen trapping (Figure 2).7,8 Most CFHs occupy the superficial dermis but can extend into the deep reticular dermis, thus mimicking the honeycomb pattern seen in DFSP. This neoplasm can show a similar architecture to DFSP, which is why further investigation including cytogenetics and immunohistochemical staining can help differentiate the two conditions. Cellular fibrous histiocytoma typically stains negative for CD34 and positive for factor XIIIa.9 However, CD34 can be positive in a subset of CFHs, with a considerable subset showing peripheral CD34 positivity and a smaller subset showing central CD34 the positivity.10 This suggests that CD34 cannot be the only factor differentiating these 2 lesions in making a proper dermatopathologic diagnosis.

Solitary fibrous tumor (SFT) is a rare mesenchymal tumor that can occur anywhere on the body and typically manifests as a deep, painless, enlarging mass in adults aged 50 to 60 years.11 On histologic examination, SFT consists of randomly arranged cells with a spindle or ovoid shape within a collagenous stroma intermixed with blood vessels with a characteristic staghorn shape (Figure 3).11 Low-grade SFT shows a patternless arrangement with spindle cells, a low number of mitotic figures, and vessels with a staghorn appearance compared to high-grade SFT, which shows hypercellularity with nuclear pleomorphism and a high number of mitotic figures.11 Solitary fibrous tumors are positive for CD34 and STAT-6 and negative for CD31 and typically demonstrate NGFI-A binding protein 2 (NAB2)—signal transducer and activator of transcription 6 (STAT 6) gene fusion.11

Spindle-cell lipomas are rare, benign, slow-growing, lipomatous tumors that typically manifest in men aged 40 to 70 years.12 These lesions originate most frequently in the subcutaneous tissue of the upper back, posterior neck, and shoulders. The histologic growth pattern of spindle-cell lipomas can mimic other spindle-cell and myxoid tumors, which is why cytogenetic analysis is crucial for differentiating these lesions. On histologic examination, spindle-cell lipomas exhibit a mixture of mature adipocytes, uniform spindle cells, and collagen bundles (eFigure). Spindle-cell lipoma stains positive for CD34 but negative for S100.13 In addition, spindle-cell lipomas tend to show structural rearrangements (mainly deletions) of the long arm of chromosome 13 or even losses of whole chromosome 13, which contains the retinoblastoma (RB1) gene.13

Pleomorphic dermal sarcoma is a rare mesenchymal tumor that can appear clinically and histologically similar to atypical fibroxanthoma.14 This lesion often manifests in elderly patients and is strongly associated with chronic sun exposure.15 Pleomorphic dermal sarcoma is a locally aggressive tumor with metastatic potential to the skin or lymph nodes. On histologic examination, these tumors exhibit pleomorphic atypical epithelioid or spindle cells as well as multinucleated tumor giant cells with possible tumor necrosis, lymphovascular invasion, or perineural infiltration (Figure 4). Pleomorphic dermal sarcoma, typically a diagnosis of exclusion, requires immunohistochemistry to aid in proper identification.16 These lesions stain positive for CD10 and negative for cytokeratins, desmin, HMB45, CD34, p63, p40, SOX10, and S100.15,16

THE DIAGNOSIS: Dermatofibrosarcoma Protuberans
The histologic findings showed fascicular proliferation of relatively monomorphic spindle cells with extensive entrapment of collagen and adipocytes. Immunohistochemical staining showed that the lesional cells were diffusely positive for CD34 and negative for SOX10, S100, desmin, and factor XIIIa. The decision was made to perform cytogenetic testing with fluorescence in situ hybridization to evaluate for the presence of platelet-derived growth factor receptor beta (PDGFB) polypeptide rearrangement, a key biomarker known to be positive in most patients with dermatofibrosarcoma protuberans (DFSP).1 This rearrangement results in overproduction of PDGFB, continuous activation of platelet-derived growth factor receptor beta, cellular proliferation, and tumor formation.2 In our patient, results were positive for the PDGFB polypeptide rearrangement, which confirmed suspected diagnosis of DFSP with fibrous histiocytoma like morphology. The patient was referred for Mohs micrographic surgery for proper management.
Dermatofibrosarcoma protuberans is a rare soft-tissue tumor that involves the dermis, subcutaneous fat, and sometimes muscle and fascia.2 Dermatofibrosarcoma protuberans primarily affects young to middle-aged adults, with a slight predilection for individuals in the third to fifth decades of life.3 Lesions preferentially involve the trunk, particularly the shoulder and chest regions, and manifest as poorly circumscribed, locally aggressive mesenchymal neoplasms with a high local recurrence rate but low metastatic potential.4,5 Clinically, the lesions appear as flesh-colored, rubbery plaques or nodules. A diagnosis of DFSP requires a high index of clinical suspicion, and histologic, immunohistochemical, and molecular testing usually are required for confirmation.
On histopathologic examination, DFSP classically demonstrates uniform, spindle-shaped cells that traditionally are arranged in an intersecting pattern and primarily are based in the dermis (Figure 1).5 Infiltration into the underlying tissue is a common feature, with neoplastic extensions causing a classic honeycomb pattern6 that also can be seen in diffuse neurofibroma and may cause diagnostic challenges; however, the immunohistology staining of neurofibroma differs from DFSP in that it stains positive for CD34, SOX-100, and S100, while DFSP has strong and diffuse CD34 immunoreactivity with negative immunostaining for SOX10, S100, desmin, and factor XIIIa.2,6

Dermatofibrosarcoma protuberans can cause considerable fat infiltration compared to other soft-tissue neoplasms, making this finding suspicious for—if not characteristic of—DFSP. Collagen trapping also can be observed; however, this is more pathognomonic in cellular fibrous histiocytoma, which is a distinct clinical variant of dermatofibromas. Due to its similarity to other lesions, histopathologic examination along with immunostaining can assist in differentiating and accurately diagnosing DFSP.6
Cellular fibrous histiocytoma (CFH), a distinct clinical variant of dermatofibromas, is a benign tumor of mesenchymal origin that occurs more commonly on the trunk, arms, and legs. On histologic examination, CFH is composed of spindle-shaped cells with variable amounts of eosinophilic cytoplasm and small, oval-shaped eosinophilic nuclei and collagen trapping (Figure 2).7,8 Most CFHs occupy the superficial dermis but can extend into the deep reticular dermis, thus mimicking the honeycomb pattern seen in DFSP. This neoplasm can show a similar architecture to DFSP, which is why further investigation including cytogenetics and immunohistochemical staining can help differentiate the two conditions. Cellular fibrous histiocytoma typically stains negative for CD34 and positive for factor XIIIa.9 However, CD34 can be positive in a subset of CFHs, with a considerable subset showing peripheral CD34 positivity and a smaller subset showing central CD34 the positivity.10 This suggests that CD34 cannot be the only factor differentiating these 2 lesions in making a proper dermatopathologic diagnosis.

Solitary fibrous tumor (SFT) is a rare mesenchymal tumor that can occur anywhere on the body and typically manifests as a deep, painless, enlarging mass in adults aged 50 to 60 years.11 On histologic examination, SFT consists of randomly arranged cells with a spindle or ovoid shape within a collagenous stroma intermixed with blood vessels with a characteristic staghorn shape (Figure 3).11 Low-grade SFT shows a patternless arrangement with spindle cells, a low number of mitotic figures, and vessels with a staghorn appearance compared to high-grade SFT, which shows hypercellularity with nuclear pleomorphism and a high number of mitotic figures.11 Solitary fibrous tumors are positive for CD34 and STAT-6 and negative for CD31 and typically demonstrate NGFI-A binding protein 2 (NAB2)—signal transducer and activator of transcription 6 (STAT 6) gene fusion.11

Spindle-cell lipomas are rare, benign, slow-growing, lipomatous tumors that typically manifest in men aged 40 to 70 years.12 These lesions originate most frequently in the subcutaneous tissue of the upper back, posterior neck, and shoulders. The histologic growth pattern of spindle-cell lipomas can mimic other spindle-cell and myxoid tumors, which is why cytogenetic analysis is crucial for differentiating these lesions. On histologic examination, spindle-cell lipomas exhibit a mixture of mature adipocytes, uniform spindle cells, and collagen bundles (eFigure). Spindle-cell lipoma stains positive for CD34 but negative for S100.13 In addition, spindle-cell lipomas tend to show structural rearrangements (mainly deletions) of the long arm of chromosome 13 or even losses of whole chromosome 13, which contains the retinoblastoma (RB1) gene.13

Pleomorphic dermal sarcoma is a rare mesenchymal tumor that can appear clinically and histologically similar to atypical fibroxanthoma.14 This lesion often manifests in elderly patients and is strongly associated with chronic sun exposure.15 Pleomorphic dermal sarcoma is a locally aggressive tumor with metastatic potential to the skin or lymph nodes. On histologic examination, these tumors exhibit pleomorphic atypical epithelioid or spindle cells as well as multinucleated tumor giant cells with possible tumor necrosis, lymphovascular invasion, or perineural infiltration (Figure 4). Pleomorphic dermal sarcoma, typically a diagnosis of exclusion, requires immunohistochemistry to aid in proper identification.16 These lesions stain positive for CD10 and negative for cytokeratins, desmin, HMB45, CD34, p63, p40, SOX10, and S100.15,16

- Ugurel S, Kortmann R, Mohr P, et al. S1 guidelines for dermatofibrosarcoma protuberans (DFSP)—update 2018. J Dtsch Dermatol Ges. 2019;17:663-668. doi:10.1111/ddg.13849
- Brooks J, Ramsey ML. Dermatofibrosarcoma protuberans. StatPearls Publishing; 2024. Updated April 18, 2024. Accessed April 30, 2025.
- Bowne WB, Antonescu CR, Leung DH, et al. Dermatofibrosarcoma protuberans: a clinicopathologic analysis of patients treated and followed at a single institution. Cancer. 2000;88:2711-2720.
- Lim SX, Ramaiya A, Levell NJ, et al. Review of dermatofibrosarcoma protuberans. Clin Exp Dermatol. 2022;48:297-302. doi:10.1093/ced/llac111
- Trinidad CM, Wangsiricharoen S, Prieto VG, et al. Rare variants of dermatofibrosarcoma protuberans: clinical, histologic, and molecular features and diagnostic pitfalls. Dermatopathology. 2023;10:54-62. doi:10.3390/dermatopathology10010008
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752. doi:10.3390/jcm9061752
- Tsunoda K, Oikawa H, Maeda F, et al. A case of cellular fibrous histiocytoma on the right elbow with repeated relapse within a short period. Case Rep Dermatol. 2015;7:10–16. https://doi.org/10.1159/000371790
- Calonje E, Mentzel T, Fletcher CD. Cellular benign fibrous histiocytoma. Clinicopathologic analysis of 74 cases of a distinctive variant of cutaneous fibrous histiocytoma with frequent recurrence. Am J Surg Pathol. 1994;18:668-676.
- Goldblum JR, Tuthill RJ. CD34 and factor-XIIIa immunoreactivity in dermatofibrosarcoma protuberans and dermatofibroma. Am J Dermatopathology. 1997;19:147-153. doi:10.1097/00000372-199704000-00008
- Volpicelli ER, Fletcher CD. Desmin and CD34 positivity in cellular fibrous histiocytoma: an immunohistochemical analysis of 100 cases. J Cutan Pathol. 2012;39:747-752. doi:10.1111/j.1600-0560.2012.01944.x
- Martin-Broto J, Mondaza-Hernandez JL, Moura DS, et al. A comprehensive review on solitary fibrous tumor: new insights for new horizons. Cancers (Basel). 2021;13:2913. doi:10.3390/cancers13122913
- Machol JA, Cusic JG, O’Connor EA, et al. Spindle cell lipoma of the neck: review of the literature and case report. Plast Reconstr Surg Glob Open. 2015;3:E550. doi:10.1097/GOX.0000000000000405
- Domanski HA, Carlén B, Jonsson K, et al. Distinct cytologic features of spindle cell lipoma. a cytologic-histologic study with clinical, radiologic, electron microscopic, and cytogenetic correlations. Cancer. 2001;93:381-389. doi:10.1002/cncr.10142
- Devine RL, Cameron A, Holden AM, et al. The pleomorphic dermal sarcoma: its management, follow-up and the need for more guidance. Adv Oral Maxillofac Surg. 2021;2:100046. doi:10.1016 /j.adoms.2021.100046
- Seretis K, Klaroudas A, Galani V, et al. Pleomorphic dermal sarcoma: it might be rare but it exists [published online August 4, 2023]. J Surg Case Rep. doi:10.1093/jscr/rjad374
- Miller K, Goodlad JR, Brenn T. Pleomorphic dermal sarcoma. Am J Surg Pathol. 2012;36:1317-1326. doi:10.1097/pas.0b013e31825359e1
- Ugurel S, Kortmann R, Mohr P, et al. S1 guidelines for dermatofibrosarcoma protuberans (DFSP)—update 2018. J Dtsch Dermatol Ges. 2019;17:663-668. doi:10.1111/ddg.13849
- Brooks J, Ramsey ML. Dermatofibrosarcoma protuberans. StatPearls Publishing; 2024. Updated April 18, 2024. Accessed April 30, 2025.
- Bowne WB, Antonescu CR, Leung DH, et al. Dermatofibrosarcoma protuberans: a clinicopathologic analysis of patients treated and followed at a single institution. Cancer. 2000;88:2711-2720.
- Lim SX, Ramaiya A, Levell NJ, et al. Review of dermatofibrosarcoma protuberans. Clin Exp Dermatol. 2022;48:297-302. doi:10.1093/ced/llac111
- Trinidad CM, Wangsiricharoen S, Prieto VG, et al. Rare variants of dermatofibrosarcoma protuberans: clinical, histologic, and molecular features and diagnostic pitfalls. Dermatopathology. 2023;10:54-62. doi:10.3390/dermatopathology10010008
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752. doi:10.3390/jcm9061752
- Tsunoda K, Oikawa H, Maeda F, et al. A case of cellular fibrous histiocytoma on the right elbow with repeated relapse within a short period. Case Rep Dermatol. 2015;7:10–16. https://doi.org/10.1159/000371790
- Calonje E, Mentzel T, Fletcher CD. Cellular benign fibrous histiocytoma. Clinicopathologic analysis of 74 cases of a distinctive variant of cutaneous fibrous histiocytoma with frequent recurrence. Am J Surg Pathol. 1994;18:668-676.
- Goldblum JR, Tuthill RJ. CD34 and factor-XIIIa immunoreactivity in dermatofibrosarcoma protuberans and dermatofibroma. Am J Dermatopathology. 1997;19:147-153. doi:10.1097/00000372-199704000-00008
- Volpicelli ER, Fletcher CD. Desmin and CD34 positivity in cellular fibrous histiocytoma: an immunohistochemical analysis of 100 cases. J Cutan Pathol. 2012;39:747-752. doi:10.1111/j.1600-0560.2012.01944.x
- Martin-Broto J, Mondaza-Hernandez JL, Moura DS, et al. A comprehensive review on solitary fibrous tumor: new insights for new horizons. Cancers (Basel). 2021;13:2913. doi:10.3390/cancers13122913
- Machol JA, Cusic JG, O’Connor EA, et al. Spindle cell lipoma of the neck: review of the literature and case report. Plast Reconstr Surg Glob Open. 2015;3:E550. doi:10.1097/GOX.0000000000000405
- Domanski HA, Carlén B, Jonsson K, et al. Distinct cytologic features of spindle cell lipoma. a cytologic-histologic study with clinical, radiologic, electron microscopic, and cytogenetic correlations. Cancer. 2001;93:381-389. doi:10.1002/cncr.10142
- Devine RL, Cameron A, Holden AM, et al. The pleomorphic dermal sarcoma: its management, follow-up and the need for more guidance. Adv Oral Maxillofac Surg. 2021;2:100046. doi:10.1016 /j.adoms.2021.100046
- Seretis K, Klaroudas A, Galani V, et al. Pleomorphic dermal sarcoma: it might be rare but it exists [published online August 4, 2023]. J Surg Case Rep. doi:10.1093/jscr/rjad374
- Miller K, Goodlad JR, Brenn T. Pleomorphic dermal sarcoma. Am J Surg Pathol. 2012;36:1317-1326. doi:10.1097/pas.0b013e31825359e1
Painful Flesh-Colored Nodule on the Shoulder
Painful Flesh-Colored Nodule on the Shoulder
A 26-year-old man with no notable medical history presented to the dermatology clinic with an inconspicuous, painful, raised lesion on the right posterior shoulder of 6 months’ duration. The patient reported that the lesion was tender to light palpation and bothersome in his daily activities. Physical examination revealed a firm, flesh-colored, 1.8-cm nodule with no erythema or pigmentation on the right shoulder. An elliptical excisional biopsy was performed and submitted for histologic evaluation.