User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Artificial Intelligence vs Medical Providers in the Dermoscopic Diagnosis of Melanoma
The incidence of skin cancer continues to increase, and it is by far the most common malignancy in the United States. Based on the sheer incidence and prevalence of skin cancer, early detection and treatment are critical. Looking at melanoma alone, the 5-year survival rate is greater than 99% when detected early but falls to 71% when the disease reaches the lymph nodes and 32% with metastasis to distant organs.1 Furthermore, a 2018 study found stage I melanoma patients who were treated 4 months after biopsy had a 41% increased risk of death compared with those treated within the first month.2 However, many patients are not seen by a dermatologist first for examination of suspicious skin lesions and instead are referred by a general practitioner or primary care mid-level provider. Therefore, many patients experience a longer time to diagnosis or treatment, which directly correlates with survival rate.
Dermoscopy is a noninvasive diagnostic tool for skin lesions, including melanoma. Using a handheld dermoscope (or dermatoscope), a transilluminating light source magnifies skin lesions and allows for the visualization of subsurface skin structures within the epidermis, dermoepidermal junction, and papillary dermis.3 Dermoscopy has been shown to improve a dermatologist’s accuracy in diagnosing malignant melanoma vs clinical evaluation with the unaided eye.4,5 More recently, dermoscopy has been digitized, allowing for the collection and documentation of case photographs. Dermoscopy also has expanded past the scope of dermatologists and has become increasingly useful in primary care.6 Among family physicians, dermoscopy also has been shown to have a higher sensitivity for melanoma detection compared to gross examination.7 Therefore, both the increased diagnostic performance of malignant melanoma using a dermoscope and the expanded use of dermoscopy in medical care validate the evaluation of an artificial intelligence (AI) algorithm in diagnosing malignant melanoma using dermoscopic images.
Triage (Triage Technologies Inc) is an AI application that uses a web interface and combines a pretrained convolutional neural network (CNN) with a reinforcement learning agent as a question-answering model. The CNN algorithm can classify 133 different skin diseases, 7 of which it is able to classify using dermoscopic images. This study sought to evaluate the performance of Triage’s dermoscopic classifier in identifying lesions as benign or malignant to determine whether AI could assist in the triage of skin cancer cases to shorten time to diagnosis.
Materials and Methods
The MClass-D test set from the International Skin Imaging Collaboration was assessed by both AI and practicing medical providers. The set was composed of 80 benign nevi and 20 biopsy-verified malignant melanomas. Board-certified US dermatologists (n=23), family physicians (n=7), and primary care mid-level providers (n=12)(ie, nurse practitioners, physician assistants) were asked to label the images as benign or malignant. The results from the medical providers were then compared to the performance of the AI application by looking at the sensitivity, specificity, accuracy, positive predictive value (PPV), and negative predictive value (NPV). Statistical significance was determined with a 1 sample t test run through RStudio (Posit Software, PBC), and P<.05 was considered significant.
Results
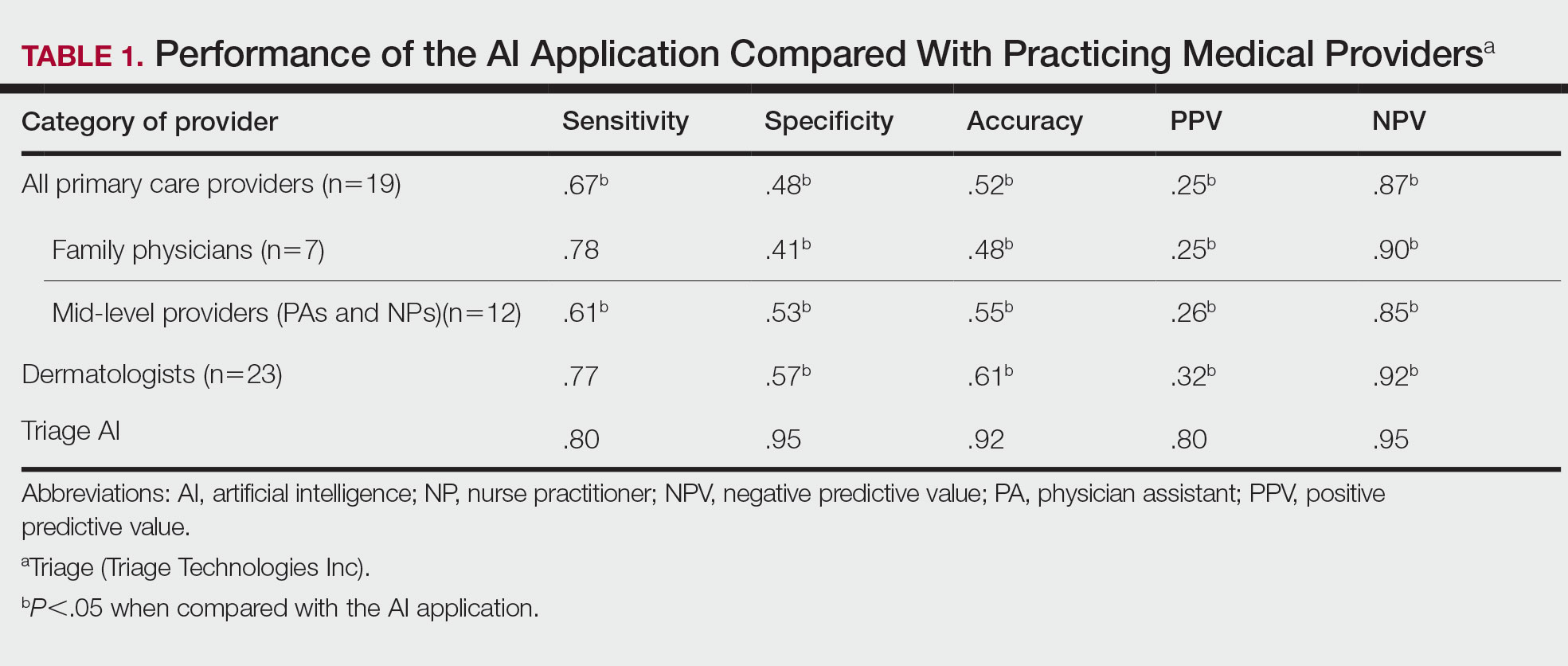
The AI application performed extremely well in differentiating between benign nevi and malignant melanomas, with a sensitivity of 80%, specificity of 95%, accuracy of 92%, PPV of 80%, and NPV of 95% (Table 1). When compared with practicing medical providers, the AI performed significantly better in almost all categories (P<.05)(Figure 1). With all medical providers combined, the AI had significantly higher accuracy, sensitivity, and specificity (P<.05). The accuracy of the individual medical providers ranged from 32% to 78%.
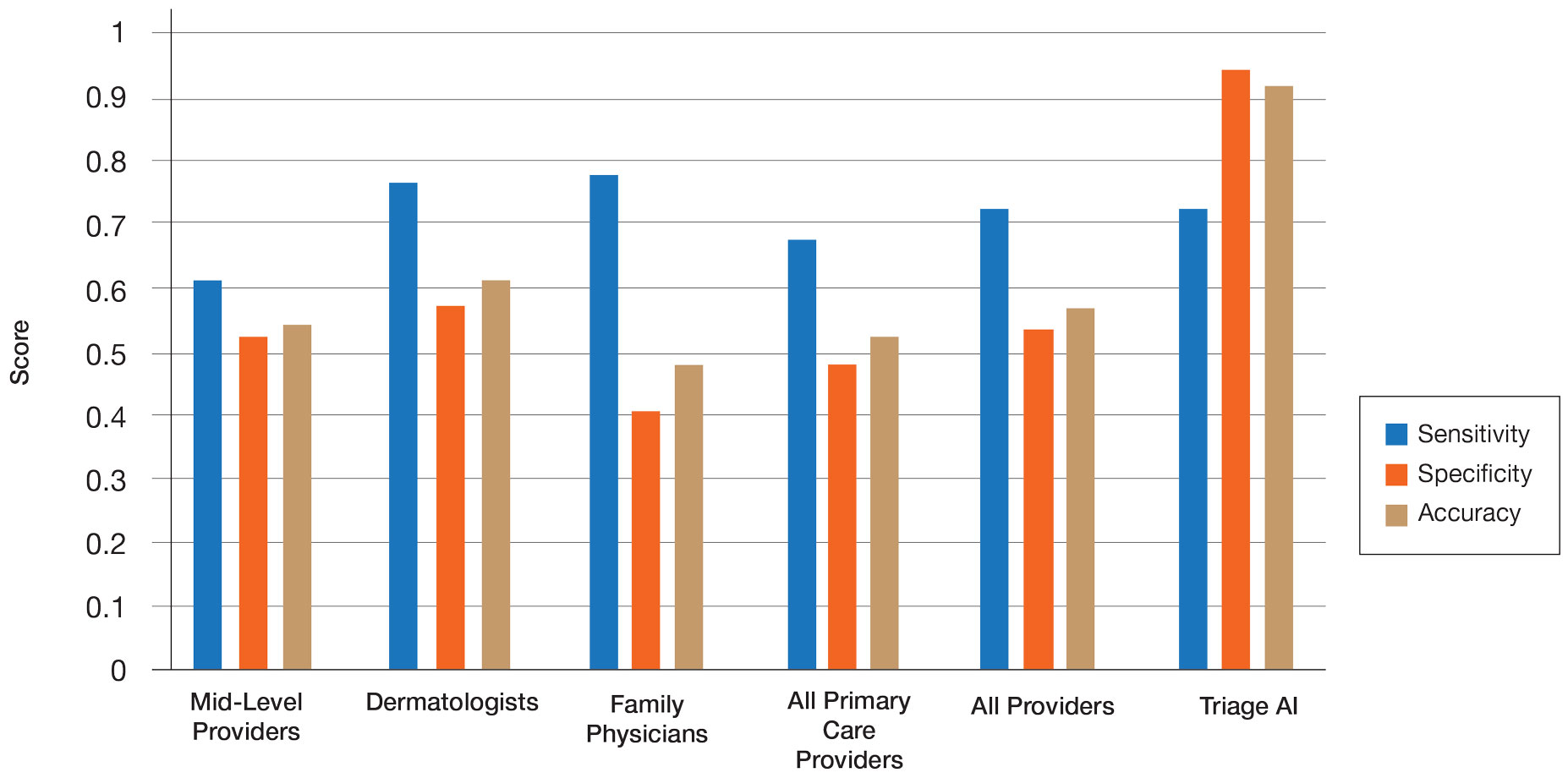
Compared with dermatologists, the AI was significantly more specific and accurate and demonstrated a higher PPV and NPV (P<.05). There was no significant difference between the AI and dermatologists in sensitivity or labeling the true malignant lesions as malignant. The dermatologists who participated had been practicing from 1.5 years to 44 years, with an average of 16 years of dermatologic experience. There was no correlation between years practicing and performance in determining the malignancy of lesions. Of 14 dermatologists, dermoscopy was used daily by 10 and occasionally by 3, but only 6 dermatologists had any formal training. Dermatologists who used dermoscopy averaged 11 years of use.
The AI also performed significantly better than the primary care providers, including both family physicians and mid-level providers (P<.05). With the family physicians and mid-level provider scores combined, the AI showed a statistically significantly better performance in all categories examined, including sensitivity, specificity, accuracy, PPV, and NPV (P<.05). However, when compared with family physicians alone, the AI did not demonstrate a statistically significant difference in sensitivity.
Comment
Automatic Visual Recognition Development—The AI application we studied was developed by dermatologists as a tool to assist in the screening of skin lesions suspicious for melanoma or a benign neoplasm.8 Developing AI applications that can reliably recognize objects in photographs has been the subject of considerable research. Notable progress in automatic visual recognition was shown in 2012 when a deep learning model won the ImageNet object recognition challenge and outperformed competing approaches by a large margin.9,10 The ImageNet competition, which has been held annually since 2010, required participants to build a visual classification system that distinguished among 1000 object categories using 1.2 million labeled images as training data. In 2017, participants developed automated visual systems that surpassed the estimated human performance.11 Given this success, the organization decided to deliver a more challenging competition involving 3D imaging—Medical ImageNet, a petabyte-scale, cloud-based, open repository project—with goals including image classification and annotation.12
Convolutional Neural Networks—Convolutional neural networks are computer system architectures commonly employed for making predictions from images.13 Convolutional neural networks are based on a set of layers of learned filters that perform convolution, a mathematical operation that reflects the relationship between the 2 functions. The main algorithm that makes the learning possible is called backpropagation, wherein an error is computed at the output and distributed backward through the neural network’s layers.14 Although CNNs and backpropagation methods have existed since 1989, recent technologic advances have allowed for deep learning–based algorithms to be widely integrated with everyday applications.15 Advances in computational power in the form of graphics processing units and parallelization, the existence of large data sets such as the ImageNet database, and the rise of software frameworks have allowed for quick prototyping and deployment of deep learning models.16,17
Convolutional neural networks have demonstrated potential to excel at a wide range of visual tasks. In dermatology, visual recognition methods often rely on using either a pretrained CNN as a feature extractor for further classification or fine-tuning a pretrained network on dermoscopic images.18-20 In 2017, a model was trained on 130,000 clinical images of benign and malignant skin lesions. Its performance was found to be in line with that of 21 US board-certified dermatology experts when diagnosing skin cancers from clinical images confirmed by biopsy.21
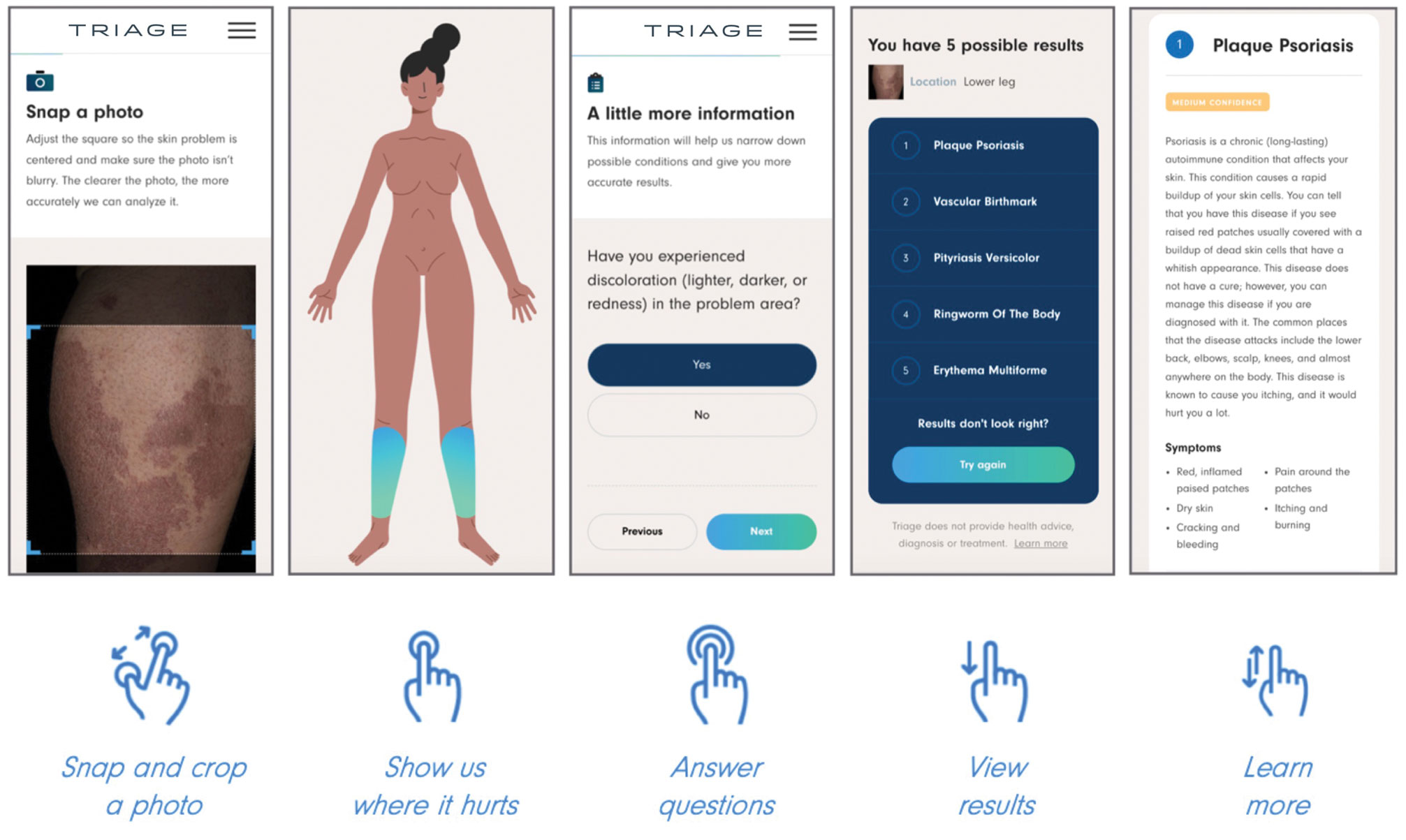
Triage—The AI application Triage is composed of several components contained in a web interface (Figure 2). To use the interface, the user must sign up and upload a photograph to the website. The image first passes through a gated-logic visual classifier that rejects any images that do not contain a visible skin condition. If the image contains a skin condition, the image is passed to a skin classifier that predicts the probability of the image containing 1 of 133 classes of skin conditions, 7 of which the application can diagnose with a dermoscopic image.
The AI application uses several techniques when training a CNN model. To address skin condition class imbalances (when more examples exist for 1 class than the others) in the training data, additional weights are applied to mistakes made on underrepresented classes, which encourages the model to better detect cases with low prevalence in the data set. Data augmentation techniques such as rotating, zooming, and flipping the training images are applied to allow the model to become more familiar with variability in the input images. Convolutional neural networks are trained using a well-known neural network optimization method called Stochastic gradient descent with momentum.22
The final predictions are refined by a question-and-answer system that encodes dermatology knowledge and is currently under active development. Finally, the top k most probable conditions are displayed to the user, where k≤5. An initial prototype of the system was described in a published research paper in the 2019 medical imaging workshop of the Neural Information Systems conference.23
The prototype demonstrated that combining a pretrained CNN with a reinforcement learning agent as a question-answering model increased the classification confidence and accuracy of its visual symptom checker and decreased the average number of questions asked to narrow down the differential diagnosis. The reinforcement learning approach increases the accuracy more than 20% compared with the CNN-only approach, which only uses visual information to predict the condition.23
This application’s current visual question-answering system is trained on a diverse set of data that includes more than 20 years of clinical encounters and user-uploaded cases submitted by more than 150,000 patients and 10,000 clinicians in more than 150 countries. All crowdsourced images used for training the dermoscopy classifier are biopsy-verified images contributed by dermatologists. These data are made up of case photographs that are tagged with metadata around the patient’s age, sex, symptoms, and diagnoses. The CNN algorithm used covers 133 skin disease classes, representing 588 clinical conditions. It also can automatically detect 7 malignant, premalignant, and benign dermoscopic categories, which is the focus of this study (Table 2). Diagnoses are verified by patient response to treatment, biopsy results, and dermatologist consensus.

In addition to having improved performance, supporting more than 130 disease classes, and having a diverse data set, the application used has beat competing technologies.20,24 The application currently is available on the internet in more than 30 countries after it received Health Canada Class I medical device approval and the CE mark in Europe.
Can AI Reliably Detect Melanoma?—In our study, of the lesions labeled benign, the higher PPV and NPV of the AI algorithm means that the lesions were more reliably true benign lesions, and the lesions labeled as malignant were more likely to be true malignant lesions. Therefore, the diagnosis given by the AI compared with the medical provider was significantly more likely to be correct. These findings demonstrate that this AI application can reliably detect malignant melanoma using dermoscopic images. However, this study was limited by the small sample size of medical providers. Further studies are necessary to assess whether the high diagnostic accuracy of the application translates to expedited referrals and a decrease in unnecessary biopsies.
Dermoscopy Training—This study looked at dermoscopic images instead of gross examination, as is often done in clinic, which draws into question the dermoscopic training dermatologists receive. The diagnostic accuracy using dermoscopic images has been shown to be higher than evaluation with the naked eye.5,6 However, there currently is no standard for dermoscopic training in dermatology residencies, and education varies widely.25 These data suggest that there may be a lack of dermoscopic training among dermatologists, which could accentuate the difference in performance between dermatologists and AI. Most primary care providers also lack formal dermoscopy training. Although dermoscopy has been shown to increase the diagnostic efficacy of primary care providers, this increase does not become apparent until the medical provider has had years of formal training in addition to clinical experience, which is not commonly provided in the medical training that primary care providers receive.8,26
Conclusion
It is anticipated that AI will shape the future of medicine and become incorporated into daily practice.27 Artificial intelligence will not replace physicians but rather assist clinicians and help to streamline medical care. Clinicians will take on the role of interpreting AI output and integrate it into patient care. With this advancement, it is important to highlight that for AI to improve the quality, efficiency, and accessibility of health care, clinicians must be equipped with the right training.27-29
- Cancer facts & figures 2023. American Cancer Society. Accessed April 20, 2023. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Conic RZ, Cabrera CI, Khorana AA, et al. Determination of the impact of melanoma surgical timing on survival using the National Cancer Database. J Am Acad Dermatol. 2018;78:40-46.e7. doi:10.1016/j.jaad.2017.08.039
- Lallas A, Zalaudek I, Argenziano G, et al. Dermoscopy in general dermatology. Dermatol Clin. 2013;31:679-694, x. doi:10.1016/j.det.2013.06.008
- Bafounta M-L, Beauchet A, Aegerter P, et al. Is dermoscopy (epiluminescence microscopy) useful for the diagnosis of melanoma?: results of a meta-analysis using techniques adapted to the evaluation of diagnostic tests. Arch Dermatol. 2001;137:1343-1350. doi:10.1001/archderm.137.10.1343
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676. doi:10.1111/j.1365-2133.2008.08713.x
- Marghoob AA, Usatine RP, Jaimes N. Dermoscopy for the family physician. Am Fam Physician. 2013;88:441-450.
- Herschorn A. Dermoscopy for melanoma detection in family practice. Can Fam Physician. 2012;58:740-745, e372-8.
- Instructions for use for the Triage app. Triage website. Accessed April 20, 2023. https://www.triage.com/pdf/en/Instructions%20for%20Use.pdf
- Krizhevsky A, Sutskever I, Hinton GE. ImageNet classification with deep convolutional neural networks. In: Pereira F, Burges CJC, Bottou L, et al, eds. Advances in Neural Information Processing Systems. Vol 25. Curran Associates, Inc; 2012. Accessed April 17, 2023. https://proceedings.neurips.cc/paper/2012/file/c399862d3b9d6b76c8436e924a68c45b-Paper.pdf
- Russakovsky O, Deng J, Su H, et al. ImageNet large scale visualrecognition challenge. Int J Comput Vis. 2015;115:211-252. doi:10.1007/s11263-015-0816-y
- Hu J, Shen L, Albanie S, et al. Squeeze-and-excitation networks. IEEE Trans Patt Anal Mach Intell. 2020;42:2011-2023. doi:10.1109/TPAMI.2019.2913372
- Medical image net-radiology informatics. Stanford University Center for Artificial Intelligence in Medicine & Imaging website. Accessed April 20, 2023. https://aimi.stanford.edu/medical-imagenet
- LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521:436-444. doi:10.1038/nature14539
- Le Cun Yet al. A theoretical framework for back-propagation. In:Touretzky D, Honton G, Sejnowski T, eds. Proceedings of the 1988 Connect Models Summer School. Morgan Kaufmann; 1988:21-28.
- Lecun Y, Bottou L, Bengio Y, et al. Gradient-based learning applied to document recognition. Proc IEEE. 1998;86:2278-2324. doi:10.1109/5.726791
- Chollet E. About Keras. Keras website. Accessed April 21, 2023. https://keras.io/about/
- Introduction to TensorFlow. TensorFlow website. Accessed April 21, 2023. https://www.tensorflow.org/learn
- Kawahara J, BenTaieb A, Hamarneh G. Deep features to classify skin lesions. 2016 IEEE 13th International Symposium on Biomedical Imaging. 2016. doi:10.1109/ISBI.2016.7493528
- Lopez AR, Giro-i-Nieto X, Burdick J, et al. Skin lesion classification from dermoscopic images using deep learning techniques. doi:10.2316/P.2017.852-053
- Codella NCF, Nguyen QB, Pankanti S, et al. Deep learning ensembles for melanoma recognition in dermoscopy images. IBM J Res Dev. 2017;61:1-28. doi:10.1147/JRD.2017.2708299
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Sutskever I, Martens J, Dahl G, et al. On the importance of initialization and momentum in deep learning. ICML’13: Proceedings of the 30th International Conference on International Conference on Machine Learning. 2013;28:1139-1147.
- Akrout M, Farahmand AM, Jarmain T, et al. Improving skin condition classification with a visual symptom checker trained using reinforcement learning. In: Medical Image Computing and Computer Assisted Intervention – MICCAI 2019: 22nd International Conference. October 13-17, 2019. Shenzhen, China. Proceedings, Part IV. Springer-Verlag; 549-557. doi:10.1007/978-3-030-32251-9_60
- Liu Y, Jain A, Eng C, et al. A deep learning system for differential diagnosis of skin diseases. Nat Med. 2020;26:900-908. doi:10.1038/s41591-020-0842-3
- Fried LJ, Tan A, Berry EG, et al. Dermoscopy proficiency expectations for US dermatology resident physicians: results of a modified delphi survey of pigmented lesion experts. JAMA Dermatol. 2021;157:189-197. doi:10.1001/jamadermatol.2020.5213
- Fee JA, McGrady FP, Rosendahl C, et al. Training primary care physicians in dermoscopy for skin cancer detection: a scoping review. J Cancer Educ. 2020;35:643-650. doi:10.1007/s13187-019-01647-7
- James CA, Wachter RM, Woolliscroft JO. Preparing clinicians for a clinical world influenced by artificial intelligence. JAMA. 2022;327:1333-1334. doi:10.1001/jama.2022.3580
- Yu K-H, Beam AL, Kohane IS. Artificial intelligence in healthcare. Nat Biomed Eng. 2018;2:719-731. doi:10.1038/s41551-018-0305-z
- Chen M, Decary M. Artificial intelligence in healthcare: an essential guide for health leaders. Healthc Manag Forum. 2020;33:10-18. doi:10.1177/0840470419873123
The incidence of skin cancer continues to increase, and it is by far the most common malignancy in the United States. Based on the sheer incidence and prevalence of skin cancer, early detection and treatment are critical. Looking at melanoma alone, the 5-year survival rate is greater than 99% when detected early but falls to 71% when the disease reaches the lymph nodes and 32% with metastasis to distant organs.1 Furthermore, a 2018 study found stage I melanoma patients who were treated 4 months after biopsy had a 41% increased risk of death compared with those treated within the first month.2 However, many patients are not seen by a dermatologist first for examination of suspicious skin lesions and instead are referred by a general practitioner or primary care mid-level provider. Therefore, many patients experience a longer time to diagnosis or treatment, which directly correlates with survival rate.
Dermoscopy is a noninvasive diagnostic tool for skin lesions, including melanoma. Using a handheld dermoscope (or dermatoscope), a transilluminating light source magnifies skin lesions and allows for the visualization of subsurface skin structures within the epidermis, dermoepidermal junction, and papillary dermis.3 Dermoscopy has been shown to improve a dermatologist’s accuracy in diagnosing malignant melanoma vs clinical evaluation with the unaided eye.4,5 More recently, dermoscopy has been digitized, allowing for the collection and documentation of case photographs. Dermoscopy also has expanded past the scope of dermatologists and has become increasingly useful in primary care.6 Among family physicians, dermoscopy also has been shown to have a higher sensitivity for melanoma detection compared to gross examination.7 Therefore, both the increased diagnostic performance of malignant melanoma using a dermoscope and the expanded use of dermoscopy in medical care validate the evaluation of an artificial intelligence (AI) algorithm in diagnosing malignant melanoma using dermoscopic images.
Triage (Triage Technologies Inc) is an AI application that uses a web interface and combines a pretrained convolutional neural network (CNN) with a reinforcement learning agent as a question-answering model. The CNN algorithm can classify 133 different skin diseases, 7 of which it is able to classify using dermoscopic images. This study sought to evaluate the performance of Triage’s dermoscopic classifier in identifying lesions as benign or malignant to determine whether AI could assist in the triage of skin cancer cases to shorten time to diagnosis.
Materials and Methods
The MClass-D test set from the International Skin Imaging Collaboration was assessed by both AI and practicing medical providers. The set was composed of 80 benign nevi and 20 biopsy-verified malignant melanomas. Board-certified US dermatologists (n=23), family physicians (n=7), and primary care mid-level providers (n=12)(ie, nurse practitioners, physician assistants) were asked to label the images as benign or malignant. The results from the medical providers were then compared to the performance of the AI application by looking at the sensitivity, specificity, accuracy, positive predictive value (PPV), and negative predictive value (NPV). Statistical significance was determined with a 1 sample t test run through RStudio (Posit Software, PBC), and P<.05 was considered significant.

Results
The AI application performed extremely well in differentiating between benign nevi and malignant melanomas, with a sensitivity of 80%, specificity of 95%, accuracy of 92%, PPV of 80%, and NPV of 95% (Table 1). When compared with practicing medical providers, the AI performed significantly better in almost all categories (P<.05)(Figure 1). With all medical providers combined, the AI had significantly higher accuracy, sensitivity, and specificity (P<.05). The accuracy of the individual medical providers ranged from 32% to 78%.

Compared with dermatologists, the AI was significantly more specific and accurate and demonstrated a higher PPV and NPV (P<.05). There was no significant difference between the AI and dermatologists in sensitivity or labeling the true malignant lesions as malignant. The dermatologists who participated had been practicing from 1.5 years to 44 years, with an average of 16 years of dermatologic experience. There was no correlation between years practicing and performance in determining the malignancy of lesions. Of 14 dermatologists, dermoscopy was used daily by 10 and occasionally by 3, but only 6 dermatologists had any formal training. Dermatologists who used dermoscopy averaged 11 years of use.
The AI also performed significantly better than the primary care providers, including both family physicians and mid-level providers (P<.05). With the family physicians and mid-level provider scores combined, the AI showed a statistically significantly better performance in all categories examined, including sensitivity, specificity, accuracy, PPV, and NPV (P<.05). However, when compared with family physicians alone, the AI did not demonstrate a statistically significant difference in sensitivity.
Comment
Automatic Visual Recognition Development—The AI application we studied was developed by dermatologists as a tool to assist in the screening of skin lesions suspicious for melanoma or a benign neoplasm.8 Developing AI applications that can reliably recognize objects in photographs has been the subject of considerable research. Notable progress in automatic visual recognition was shown in 2012 when a deep learning model won the ImageNet object recognition challenge and outperformed competing approaches by a large margin.9,10 The ImageNet competition, which has been held annually since 2010, required participants to build a visual classification system that distinguished among 1000 object categories using 1.2 million labeled images as training data. In 2017, participants developed automated visual systems that surpassed the estimated human performance.11 Given this success, the organization decided to deliver a more challenging competition involving 3D imaging—Medical ImageNet, a petabyte-scale, cloud-based, open repository project—with goals including image classification and annotation.12
Convolutional Neural Networks—Convolutional neural networks are computer system architectures commonly employed for making predictions from images.13 Convolutional neural networks are based on a set of layers of learned filters that perform convolution, a mathematical operation that reflects the relationship between the 2 functions. The main algorithm that makes the learning possible is called backpropagation, wherein an error is computed at the output and distributed backward through the neural network’s layers.14 Although CNNs and backpropagation methods have existed since 1989, recent technologic advances have allowed for deep learning–based algorithms to be widely integrated with everyday applications.15 Advances in computational power in the form of graphics processing units and parallelization, the existence of large data sets such as the ImageNet database, and the rise of software frameworks have allowed for quick prototyping and deployment of deep learning models.16,17
Convolutional neural networks have demonstrated potential to excel at a wide range of visual tasks. In dermatology, visual recognition methods often rely on using either a pretrained CNN as a feature extractor for further classification or fine-tuning a pretrained network on dermoscopic images.18-20 In 2017, a model was trained on 130,000 clinical images of benign and malignant skin lesions. Its performance was found to be in line with that of 21 US board-certified dermatology experts when diagnosing skin cancers from clinical images confirmed by biopsy.21
Triage—The AI application Triage is composed of several components contained in a web interface (Figure 2). To use the interface, the user must sign up and upload a photograph to the website. The image first passes through a gated-logic visual classifier that rejects any images that do not contain a visible skin condition. If the image contains a skin condition, the image is passed to a skin classifier that predicts the probability of the image containing 1 of 133 classes of skin conditions, 7 of which the application can diagnose with a dermoscopic image.

The AI application uses several techniques when training a CNN model. To address skin condition class imbalances (when more examples exist for 1 class than the others) in the training data, additional weights are applied to mistakes made on underrepresented classes, which encourages the model to better detect cases with low prevalence in the data set. Data augmentation techniques such as rotating, zooming, and flipping the training images are applied to allow the model to become more familiar with variability in the input images. Convolutional neural networks are trained using a well-known neural network optimization method called Stochastic gradient descent with momentum.22
The final predictions are refined by a question-and-answer system that encodes dermatology knowledge and is currently under active development. Finally, the top k most probable conditions are displayed to the user, where k≤5. An initial prototype of the system was described in a published research paper in the 2019 medical imaging workshop of the Neural Information Systems conference.23
The prototype demonstrated that combining a pretrained CNN with a reinforcement learning agent as a question-answering model increased the classification confidence and accuracy of its visual symptom checker and decreased the average number of questions asked to narrow down the differential diagnosis. The reinforcement learning approach increases the accuracy more than 20% compared with the CNN-only approach, which only uses visual information to predict the condition.23
This application’s current visual question-answering system is trained on a diverse set of data that includes more than 20 years of clinical encounters and user-uploaded cases submitted by more than 150,000 patients and 10,000 clinicians in more than 150 countries. All crowdsourced images used for training the dermoscopy classifier are biopsy-verified images contributed by dermatologists. These data are made up of case photographs that are tagged with metadata around the patient’s age, sex, symptoms, and diagnoses. The CNN algorithm used covers 133 skin disease classes, representing 588 clinical conditions. It also can automatically detect 7 malignant, premalignant, and benign dermoscopic categories, which is the focus of this study (Table 2). Diagnoses are verified by patient response to treatment, biopsy results, and dermatologist consensus.

In addition to having improved performance, supporting more than 130 disease classes, and having a diverse data set, the application used has beat competing technologies.20,24 The application currently is available on the internet in more than 30 countries after it received Health Canada Class I medical device approval and the CE mark in Europe.
Can AI Reliably Detect Melanoma?—In our study, of the lesions labeled benign, the higher PPV and NPV of the AI algorithm means that the lesions were more reliably true benign lesions, and the lesions labeled as malignant were more likely to be true malignant lesions. Therefore, the diagnosis given by the AI compared with the medical provider was significantly more likely to be correct. These findings demonstrate that this AI application can reliably detect malignant melanoma using dermoscopic images. However, this study was limited by the small sample size of medical providers. Further studies are necessary to assess whether the high diagnostic accuracy of the application translates to expedited referrals and a decrease in unnecessary biopsies.
Dermoscopy Training—This study looked at dermoscopic images instead of gross examination, as is often done in clinic, which draws into question the dermoscopic training dermatologists receive. The diagnostic accuracy using dermoscopic images has been shown to be higher than evaluation with the naked eye.5,6 However, there currently is no standard for dermoscopic training in dermatology residencies, and education varies widely.25 These data suggest that there may be a lack of dermoscopic training among dermatologists, which could accentuate the difference in performance between dermatologists and AI. Most primary care providers also lack formal dermoscopy training. Although dermoscopy has been shown to increase the diagnostic efficacy of primary care providers, this increase does not become apparent until the medical provider has had years of formal training in addition to clinical experience, which is not commonly provided in the medical training that primary care providers receive.8,26
Conclusion
It is anticipated that AI will shape the future of medicine and become incorporated into daily practice.27 Artificial intelligence will not replace physicians but rather assist clinicians and help to streamline medical care. Clinicians will take on the role of interpreting AI output and integrate it into patient care. With this advancement, it is important to highlight that for AI to improve the quality, efficiency, and accessibility of health care, clinicians must be equipped with the right training.27-29
The incidence of skin cancer continues to increase, and it is by far the most common malignancy in the United States. Based on the sheer incidence and prevalence of skin cancer, early detection and treatment are critical. Looking at melanoma alone, the 5-year survival rate is greater than 99% when detected early but falls to 71% when the disease reaches the lymph nodes and 32% with metastasis to distant organs.1 Furthermore, a 2018 study found stage I melanoma patients who were treated 4 months after biopsy had a 41% increased risk of death compared with those treated within the first month.2 However, many patients are not seen by a dermatologist first for examination of suspicious skin lesions and instead are referred by a general practitioner or primary care mid-level provider. Therefore, many patients experience a longer time to diagnosis or treatment, which directly correlates with survival rate.
Dermoscopy is a noninvasive diagnostic tool for skin lesions, including melanoma. Using a handheld dermoscope (or dermatoscope), a transilluminating light source magnifies skin lesions and allows for the visualization of subsurface skin structures within the epidermis, dermoepidermal junction, and papillary dermis.3 Dermoscopy has been shown to improve a dermatologist’s accuracy in diagnosing malignant melanoma vs clinical evaluation with the unaided eye.4,5 More recently, dermoscopy has been digitized, allowing for the collection and documentation of case photographs. Dermoscopy also has expanded past the scope of dermatologists and has become increasingly useful in primary care.6 Among family physicians, dermoscopy also has been shown to have a higher sensitivity for melanoma detection compared to gross examination.7 Therefore, both the increased diagnostic performance of malignant melanoma using a dermoscope and the expanded use of dermoscopy in medical care validate the evaluation of an artificial intelligence (AI) algorithm in diagnosing malignant melanoma using dermoscopic images.
Triage (Triage Technologies Inc) is an AI application that uses a web interface and combines a pretrained convolutional neural network (CNN) with a reinforcement learning agent as a question-answering model. The CNN algorithm can classify 133 different skin diseases, 7 of which it is able to classify using dermoscopic images. This study sought to evaluate the performance of Triage’s dermoscopic classifier in identifying lesions as benign or malignant to determine whether AI could assist in the triage of skin cancer cases to shorten time to diagnosis.
Materials and Methods
The MClass-D test set from the International Skin Imaging Collaboration was assessed by both AI and practicing medical providers. The set was composed of 80 benign nevi and 20 biopsy-verified malignant melanomas. Board-certified US dermatologists (n=23), family physicians (n=7), and primary care mid-level providers (n=12)(ie, nurse practitioners, physician assistants) were asked to label the images as benign or malignant. The results from the medical providers were then compared to the performance of the AI application by looking at the sensitivity, specificity, accuracy, positive predictive value (PPV), and negative predictive value (NPV). Statistical significance was determined with a 1 sample t test run through RStudio (Posit Software, PBC), and P<.05 was considered significant.

Results
The AI application performed extremely well in differentiating between benign nevi and malignant melanomas, with a sensitivity of 80%, specificity of 95%, accuracy of 92%, PPV of 80%, and NPV of 95% (Table 1). When compared with practicing medical providers, the AI performed significantly better in almost all categories (P<.05)(Figure 1). With all medical providers combined, the AI had significantly higher accuracy, sensitivity, and specificity (P<.05). The accuracy of the individual medical providers ranged from 32% to 78%.

Compared with dermatologists, the AI was significantly more specific and accurate and demonstrated a higher PPV and NPV (P<.05). There was no significant difference between the AI and dermatologists in sensitivity or labeling the true malignant lesions as malignant. The dermatologists who participated had been practicing from 1.5 years to 44 years, with an average of 16 years of dermatologic experience. There was no correlation between years practicing and performance in determining the malignancy of lesions. Of 14 dermatologists, dermoscopy was used daily by 10 and occasionally by 3, but only 6 dermatologists had any formal training. Dermatologists who used dermoscopy averaged 11 years of use.
The AI also performed significantly better than the primary care providers, including both family physicians and mid-level providers (P<.05). With the family physicians and mid-level provider scores combined, the AI showed a statistically significantly better performance in all categories examined, including sensitivity, specificity, accuracy, PPV, and NPV (P<.05). However, when compared with family physicians alone, the AI did not demonstrate a statistically significant difference in sensitivity.
Comment
Automatic Visual Recognition Development—The AI application we studied was developed by dermatologists as a tool to assist in the screening of skin lesions suspicious for melanoma or a benign neoplasm.8 Developing AI applications that can reliably recognize objects in photographs has been the subject of considerable research. Notable progress in automatic visual recognition was shown in 2012 when a deep learning model won the ImageNet object recognition challenge and outperformed competing approaches by a large margin.9,10 The ImageNet competition, which has been held annually since 2010, required participants to build a visual classification system that distinguished among 1000 object categories using 1.2 million labeled images as training data. In 2017, participants developed automated visual systems that surpassed the estimated human performance.11 Given this success, the organization decided to deliver a more challenging competition involving 3D imaging—Medical ImageNet, a petabyte-scale, cloud-based, open repository project—with goals including image classification and annotation.12
Convolutional Neural Networks—Convolutional neural networks are computer system architectures commonly employed for making predictions from images.13 Convolutional neural networks are based on a set of layers of learned filters that perform convolution, a mathematical operation that reflects the relationship between the 2 functions. The main algorithm that makes the learning possible is called backpropagation, wherein an error is computed at the output and distributed backward through the neural network’s layers.14 Although CNNs and backpropagation methods have existed since 1989, recent technologic advances have allowed for deep learning–based algorithms to be widely integrated with everyday applications.15 Advances in computational power in the form of graphics processing units and parallelization, the existence of large data sets such as the ImageNet database, and the rise of software frameworks have allowed for quick prototyping and deployment of deep learning models.16,17
Convolutional neural networks have demonstrated potential to excel at a wide range of visual tasks. In dermatology, visual recognition methods often rely on using either a pretrained CNN as a feature extractor for further classification or fine-tuning a pretrained network on dermoscopic images.18-20 In 2017, a model was trained on 130,000 clinical images of benign and malignant skin lesions. Its performance was found to be in line with that of 21 US board-certified dermatology experts when diagnosing skin cancers from clinical images confirmed by biopsy.21
Triage—The AI application Triage is composed of several components contained in a web interface (Figure 2). To use the interface, the user must sign up and upload a photograph to the website. The image first passes through a gated-logic visual classifier that rejects any images that do not contain a visible skin condition. If the image contains a skin condition, the image is passed to a skin classifier that predicts the probability of the image containing 1 of 133 classes of skin conditions, 7 of which the application can diagnose with a dermoscopic image.

The AI application uses several techniques when training a CNN model. To address skin condition class imbalances (when more examples exist for 1 class than the others) in the training data, additional weights are applied to mistakes made on underrepresented classes, which encourages the model to better detect cases with low prevalence in the data set. Data augmentation techniques such as rotating, zooming, and flipping the training images are applied to allow the model to become more familiar with variability in the input images. Convolutional neural networks are trained using a well-known neural network optimization method called Stochastic gradient descent with momentum.22
The final predictions are refined by a question-and-answer system that encodes dermatology knowledge and is currently under active development. Finally, the top k most probable conditions are displayed to the user, where k≤5. An initial prototype of the system was described in a published research paper in the 2019 medical imaging workshop of the Neural Information Systems conference.23
The prototype demonstrated that combining a pretrained CNN with a reinforcement learning agent as a question-answering model increased the classification confidence and accuracy of its visual symptom checker and decreased the average number of questions asked to narrow down the differential diagnosis. The reinforcement learning approach increases the accuracy more than 20% compared with the CNN-only approach, which only uses visual information to predict the condition.23
This application’s current visual question-answering system is trained on a diverse set of data that includes more than 20 years of clinical encounters and user-uploaded cases submitted by more than 150,000 patients and 10,000 clinicians in more than 150 countries. All crowdsourced images used for training the dermoscopy classifier are biopsy-verified images contributed by dermatologists. These data are made up of case photographs that are tagged with metadata around the patient’s age, sex, symptoms, and diagnoses. The CNN algorithm used covers 133 skin disease classes, representing 588 clinical conditions. It also can automatically detect 7 malignant, premalignant, and benign dermoscopic categories, which is the focus of this study (Table 2). Diagnoses are verified by patient response to treatment, biopsy results, and dermatologist consensus.

In addition to having improved performance, supporting more than 130 disease classes, and having a diverse data set, the application used has beat competing technologies.20,24 The application currently is available on the internet in more than 30 countries after it received Health Canada Class I medical device approval and the CE mark in Europe.
Can AI Reliably Detect Melanoma?—In our study, of the lesions labeled benign, the higher PPV and NPV of the AI algorithm means that the lesions were more reliably true benign lesions, and the lesions labeled as malignant were more likely to be true malignant lesions. Therefore, the diagnosis given by the AI compared with the medical provider was significantly more likely to be correct. These findings demonstrate that this AI application can reliably detect malignant melanoma using dermoscopic images. However, this study was limited by the small sample size of medical providers. Further studies are necessary to assess whether the high diagnostic accuracy of the application translates to expedited referrals and a decrease in unnecessary biopsies.
Dermoscopy Training—This study looked at dermoscopic images instead of gross examination, as is often done in clinic, which draws into question the dermoscopic training dermatologists receive. The diagnostic accuracy using dermoscopic images has been shown to be higher than evaluation with the naked eye.5,6 However, there currently is no standard for dermoscopic training in dermatology residencies, and education varies widely.25 These data suggest that there may be a lack of dermoscopic training among dermatologists, which could accentuate the difference in performance between dermatologists and AI. Most primary care providers also lack formal dermoscopy training. Although dermoscopy has been shown to increase the diagnostic efficacy of primary care providers, this increase does not become apparent until the medical provider has had years of formal training in addition to clinical experience, which is not commonly provided in the medical training that primary care providers receive.8,26
Conclusion
It is anticipated that AI will shape the future of medicine and become incorporated into daily practice.27 Artificial intelligence will not replace physicians but rather assist clinicians and help to streamline medical care. Clinicians will take on the role of interpreting AI output and integrate it into patient care. With this advancement, it is important to highlight that for AI to improve the quality, efficiency, and accessibility of health care, clinicians must be equipped with the right training.27-29
- Cancer facts & figures 2023. American Cancer Society. Accessed April 20, 2023. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Conic RZ, Cabrera CI, Khorana AA, et al. Determination of the impact of melanoma surgical timing on survival using the National Cancer Database. J Am Acad Dermatol. 2018;78:40-46.e7. doi:10.1016/j.jaad.2017.08.039
- Lallas A, Zalaudek I, Argenziano G, et al. Dermoscopy in general dermatology. Dermatol Clin. 2013;31:679-694, x. doi:10.1016/j.det.2013.06.008
- Bafounta M-L, Beauchet A, Aegerter P, et al. Is dermoscopy (epiluminescence microscopy) useful for the diagnosis of melanoma?: results of a meta-analysis using techniques adapted to the evaluation of diagnostic tests. Arch Dermatol. 2001;137:1343-1350. doi:10.1001/archderm.137.10.1343
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676. doi:10.1111/j.1365-2133.2008.08713.x
- Marghoob AA, Usatine RP, Jaimes N. Dermoscopy for the family physician. Am Fam Physician. 2013;88:441-450.
- Herschorn A. Dermoscopy for melanoma detection in family practice. Can Fam Physician. 2012;58:740-745, e372-8.
- Instructions for use for the Triage app. Triage website. Accessed April 20, 2023. https://www.triage.com/pdf/en/Instructions%20for%20Use.pdf
- Krizhevsky A, Sutskever I, Hinton GE. ImageNet classification with deep convolutional neural networks. In: Pereira F, Burges CJC, Bottou L, et al, eds. Advances in Neural Information Processing Systems. Vol 25. Curran Associates, Inc; 2012. Accessed April 17, 2023. https://proceedings.neurips.cc/paper/2012/file/c399862d3b9d6b76c8436e924a68c45b-Paper.pdf
- Russakovsky O, Deng J, Su H, et al. ImageNet large scale visualrecognition challenge. Int J Comput Vis. 2015;115:211-252. doi:10.1007/s11263-015-0816-y
- Hu J, Shen L, Albanie S, et al. Squeeze-and-excitation networks. IEEE Trans Patt Anal Mach Intell. 2020;42:2011-2023. doi:10.1109/TPAMI.2019.2913372
- Medical image net-radiology informatics. Stanford University Center for Artificial Intelligence in Medicine & Imaging website. Accessed April 20, 2023. https://aimi.stanford.edu/medical-imagenet
- LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521:436-444. doi:10.1038/nature14539
- Le Cun Yet al. A theoretical framework for back-propagation. In:Touretzky D, Honton G, Sejnowski T, eds. Proceedings of the 1988 Connect Models Summer School. Morgan Kaufmann; 1988:21-28.
- Lecun Y, Bottou L, Bengio Y, et al. Gradient-based learning applied to document recognition. Proc IEEE. 1998;86:2278-2324. doi:10.1109/5.726791
- Chollet E. About Keras. Keras website. Accessed April 21, 2023. https://keras.io/about/
- Introduction to TensorFlow. TensorFlow website. Accessed April 21, 2023. https://www.tensorflow.org/learn
- Kawahara J, BenTaieb A, Hamarneh G. Deep features to classify skin lesions. 2016 IEEE 13th International Symposium on Biomedical Imaging. 2016. doi:10.1109/ISBI.2016.7493528
- Lopez AR, Giro-i-Nieto X, Burdick J, et al. Skin lesion classification from dermoscopic images using deep learning techniques. doi:10.2316/P.2017.852-053
- Codella NCF, Nguyen QB, Pankanti S, et al. Deep learning ensembles for melanoma recognition in dermoscopy images. IBM J Res Dev. 2017;61:1-28. doi:10.1147/JRD.2017.2708299
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Sutskever I, Martens J, Dahl G, et al. On the importance of initialization and momentum in deep learning. ICML’13: Proceedings of the 30th International Conference on International Conference on Machine Learning. 2013;28:1139-1147.
- Akrout M, Farahmand AM, Jarmain T, et al. Improving skin condition classification with a visual symptom checker trained using reinforcement learning. In: Medical Image Computing and Computer Assisted Intervention – MICCAI 2019: 22nd International Conference. October 13-17, 2019. Shenzhen, China. Proceedings, Part IV. Springer-Verlag; 549-557. doi:10.1007/978-3-030-32251-9_60
- Liu Y, Jain A, Eng C, et al. A deep learning system for differential diagnosis of skin diseases. Nat Med. 2020;26:900-908. doi:10.1038/s41591-020-0842-3
- Fried LJ, Tan A, Berry EG, et al. Dermoscopy proficiency expectations for US dermatology resident physicians: results of a modified delphi survey of pigmented lesion experts. JAMA Dermatol. 2021;157:189-197. doi:10.1001/jamadermatol.2020.5213
- Fee JA, McGrady FP, Rosendahl C, et al. Training primary care physicians in dermoscopy for skin cancer detection: a scoping review. J Cancer Educ. 2020;35:643-650. doi:10.1007/s13187-019-01647-7
- James CA, Wachter RM, Woolliscroft JO. Preparing clinicians for a clinical world influenced by artificial intelligence. JAMA. 2022;327:1333-1334. doi:10.1001/jama.2022.3580
- Yu K-H, Beam AL, Kohane IS. Artificial intelligence in healthcare. Nat Biomed Eng. 2018;2:719-731. doi:10.1038/s41551-018-0305-z
- Chen M, Decary M. Artificial intelligence in healthcare: an essential guide for health leaders. Healthc Manag Forum. 2020;33:10-18. doi:10.1177/0840470419873123
- Cancer facts & figures 2023. American Cancer Society. Accessed April 20, 2023. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Conic RZ, Cabrera CI, Khorana AA, et al. Determination of the impact of melanoma surgical timing on survival using the National Cancer Database. J Am Acad Dermatol. 2018;78:40-46.e7. doi:10.1016/j.jaad.2017.08.039
- Lallas A, Zalaudek I, Argenziano G, et al. Dermoscopy in general dermatology. Dermatol Clin. 2013;31:679-694, x. doi:10.1016/j.det.2013.06.008
- Bafounta M-L, Beauchet A, Aegerter P, et al. Is dermoscopy (epiluminescence microscopy) useful for the diagnosis of melanoma?: results of a meta-analysis using techniques adapted to the evaluation of diagnostic tests. Arch Dermatol. 2001;137:1343-1350. doi:10.1001/archderm.137.10.1343
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676. doi:10.1111/j.1365-2133.2008.08713.x
- Marghoob AA, Usatine RP, Jaimes N. Dermoscopy for the family physician. Am Fam Physician. 2013;88:441-450.
- Herschorn A. Dermoscopy for melanoma detection in family practice. Can Fam Physician. 2012;58:740-745, e372-8.
- Instructions for use for the Triage app. Triage website. Accessed April 20, 2023. https://www.triage.com/pdf/en/Instructions%20for%20Use.pdf
- Krizhevsky A, Sutskever I, Hinton GE. ImageNet classification with deep convolutional neural networks. In: Pereira F, Burges CJC, Bottou L, et al, eds. Advances in Neural Information Processing Systems. Vol 25. Curran Associates, Inc; 2012. Accessed April 17, 2023. https://proceedings.neurips.cc/paper/2012/file/c399862d3b9d6b76c8436e924a68c45b-Paper.pdf
- Russakovsky O, Deng J, Su H, et al. ImageNet large scale visualrecognition challenge. Int J Comput Vis. 2015;115:211-252. doi:10.1007/s11263-015-0816-y
- Hu J, Shen L, Albanie S, et al. Squeeze-and-excitation networks. IEEE Trans Patt Anal Mach Intell. 2020;42:2011-2023. doi:10.1109/TPAMI.2019.2913372
- Medical image net-radiology informatics. Stanford University Center for Artificial Intelligence in Medicine & Imaging website. Accessed April 20, 2023. https://aimi.stanford.edu/medical-imagenet
- LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521:436-444. doi:10.1038/nature14539
- Le Cun Yet al. A theoretical framework for back-propagation. In:Touretzky D, Honton G, Sejnowski T, eds. Proceedings of the 1988 Connect Models Summer School. Morgan Kaufmann; 1988:21-28.
- Lecun Y, Bottou L, Bengio Y, et al. Gradient-based learning applied to document recognition. Proc IEEE. 1998;86:2278-2324. doi:10.1109/5.726791
- Chollet E. About Keras. Keras website. Accessed April 21, 2023. https://keras.io/about/
- Introduction to TensorFlow. TensorFlow website. Accessed April 21, 2023. https://www.tensorflow.org/learn
- Kawahara J, BenTaieb A, Hamarneh G. Deep features to classify skin lesions. 2016 IEEE 13th International Symposium on Biomedical Imaging. 2016. doi:10.1109/ISBI.2016.7493528
- Lopez AR, Giro-i-Nieto X, Burdick J, et al. Skin lesion classification from dermoscopic images using deep learning techniques. doi:10.2316/P.2017.852-053
- Codella NCF, Nguyen QB, Pankanti S, et al. Deep learning ensembles for melanoma recognition in dermoscopy images. IBM J Res Dev. 2017;61:1-28. doi:10.1147/JRD.2017.2708299
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Sutskever I, Martens J, Dahl G, et al. On the importance of initialization and momentum in deep learning. ICML’13: Proceedings of the 30th International Conference on International Conference on Machine Learning. 2013;28:1139-1147.
- Akrout M, Farahmand AM, Jarmain T, et al. Improving skin condition classification with a visual symptom checker trained using reinforcement learning. In: Medical Image Computing and Computer Assisted Intervention – MICCAI 2019: 22nd International Conference. October 13-17, 2019. Shenzhen, China. Proceedings, Part IV. Springer-Verlag; 549-557. doi:10.1007/978-3-030-32251-9_60
- Liu Y, Jain A, Eng C, et al. A deep learning system for differential diagnosis of skin diseases. Nat Med. 2020;26:900-908. doi:10.1038/s41591-020-0842-3
- Fried LJ, Tan A, Berry EG, et al. Dermoscopy proficiency expectations for US dermatology resident physicians: results of a modified delphi survey of pigmented lesion experts. JAMA Dermatol. 2021;157:189-197. doi:10.1001/jamadermatol.2020.5213
- Fee JA, McGrady FP, Rosendahl C, et al. Training primary care physicians in dermoscopy for skin cancer detection: a scoping review. J Cancer Educ. 2020;35:643-650. doi:10.1007/s13187-019-01647-7
- James CA, Wachter RM, Woolliscroft JO. Preparing clinicians for a clinical world influenced by artificial intelligence. JAMA. 2022;327:1333-1334. doi:10.1001/jama.2022.3580
- Yu K-H, Beam AL, Kohane IS. Artificial intelligence in healthcare. Nat Biomed Eng. 2018;2:719-731. doi:10.1038/s41551-018-0305-z
- Chen M, Decary M. Artificial intelligence in healthcare: an essential guide for health leaders. Healthc Manag Forum. 2020;33:10-18. doi:10.1177/0840470419873123
Practice Points
- Artificial intelligence (AI) has the potential to facilitate the diagnosis of pigmented lesions and expedite the management of malignant melanoma.
- Further studies should be done to see if the high diagnostic accuracy of the AI application we studied translates to a decrease in unnecessary biopsies or expedited referral for pigmented lesions.
- The large variability of formal dermoscopy training among board-certified dermatologists may contribute to the decreased ability to identify pigmented lesions with dermoscopic imaging compared to AI.
Treatment of Angiosarcoma of the Head and Neck: A Systematic Review
Cutaneous angiosarcoma (cAS) is a rare malignancy arising from vascular or lymphatic tissue. It classically presents during the sixth or seventh decades of life as a raised purple papule or plaque on the head and neck areas.1 Primary cAS frequently mimics benign conditions, leading to delays in care. Such delays coupled with the aggressive nature of angiosarcomas leads to a poor prognosis. Five-year survival rates range from 11% to 50%, and more than half of patients die within 1 year of diagnosis.2-7
Currently, there is no consensus on the most effective treatments, as the rare nature of cAS has made the development of controlled clinical trials difficult. Wide local excision (WLE) is most frequently employed; however, the tumor’s infiltrative growth makes complete resection and negative surgical margins difficult to achieve.8 Recently, Mohs micrographic surgery (MMS) has been postulated as a treatment option. The tissue-sparing nature and intraoperative margin control of MMS may provide tumor eradication and cosmesis benefits reported with other cutaneous malignancies.9
Nearly all localized cASs are treated with surgical excision with or without adjuvant treatment modalities; however, it is unclear which of these modalities provide a survival benefit. We conducted a systematic review of the literature to compare treatment modalities for localized cAS of the head and neck regions and to compare treatments based on tumor stage.
METHODS
A literature search was performed to identify published studies indexed by MEDLINE, Cochrane Central Register of Controlled Trials (CENTRAL), Embase, and PubMed from January 1, 1977, to May 8, 2020, reporting on cAS and treatment modalities used. The search was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines.5 Data extracted included patient demographics, tumor characteristics (including T1 [≤5 cm] and T2 [>5 cm and ≤10 cm] based on the American Joint Committee on Cancer soft tissue sarcoma staging criteria), treatments used, follow-up time, overall survival (OS) rates, and complications.10,11
Studies were required to (1) include participants with head and neck cAS; (2) report original patient data following cAS treatment with surgical (WLE or MMS) and/or nonsurgical modalities (chemotherapy [CT], radiotherapy [RT], immunotherapy [IT]); (3) report outcome data related to OS rates following treatment; and (4) have articles published in English. Given the rare nature of cAS, there was no limitation on the number of participants needed.
The Newcastle-Ottawa scale for observational studies was used to assess the quality of studies.12 Higher scores indicate low risk of bias, while lower scores represent high risk of bias.
Continuous data were reported with means and SDs, while categorical variables were reported as percentages. Overall survival means and SDs were compared between treatment modalities using an independent sample t test with P<.05 considered statistically significant. Due to the heterogeneity of the data, a meta-analysis was not reported.
RESULTS
Literature Search and Risk of Bias Assessment
There were 283 manuscripts identified, 56 articles read in full, and 40 articles included in the review (Figure). Among the 16 studies not meeting inclusion criteria, 7 did not provide enough data to isolate head and neck cAS cases,1,13-18 6 did not report outcomes related to the current review,19-24 and 3 did not provide enough data to isolate different treatment outcomes.25-27 Among the included studies, 32 reported use of WLE: WLE alone (n=21)2,7,11,28-45; WLE with RT (n=24)2,3,11,28-31,33-36,38-41,43-51; WLE with CT (n=7)2,31,35,39,41,48,52; WLE with RT and CT (n=11)2,29,31,33-35,39,40,48,52,53; WLE with RT and IT (n=3)35,54,55; and WLE with RT, CT, and IT (n=1).53 Nine studies reported MMS: MMS alone (n=5)39,56-59; MMS with RT (n=3)32,50,60,61; and MMS with RT and CT (n=1).51
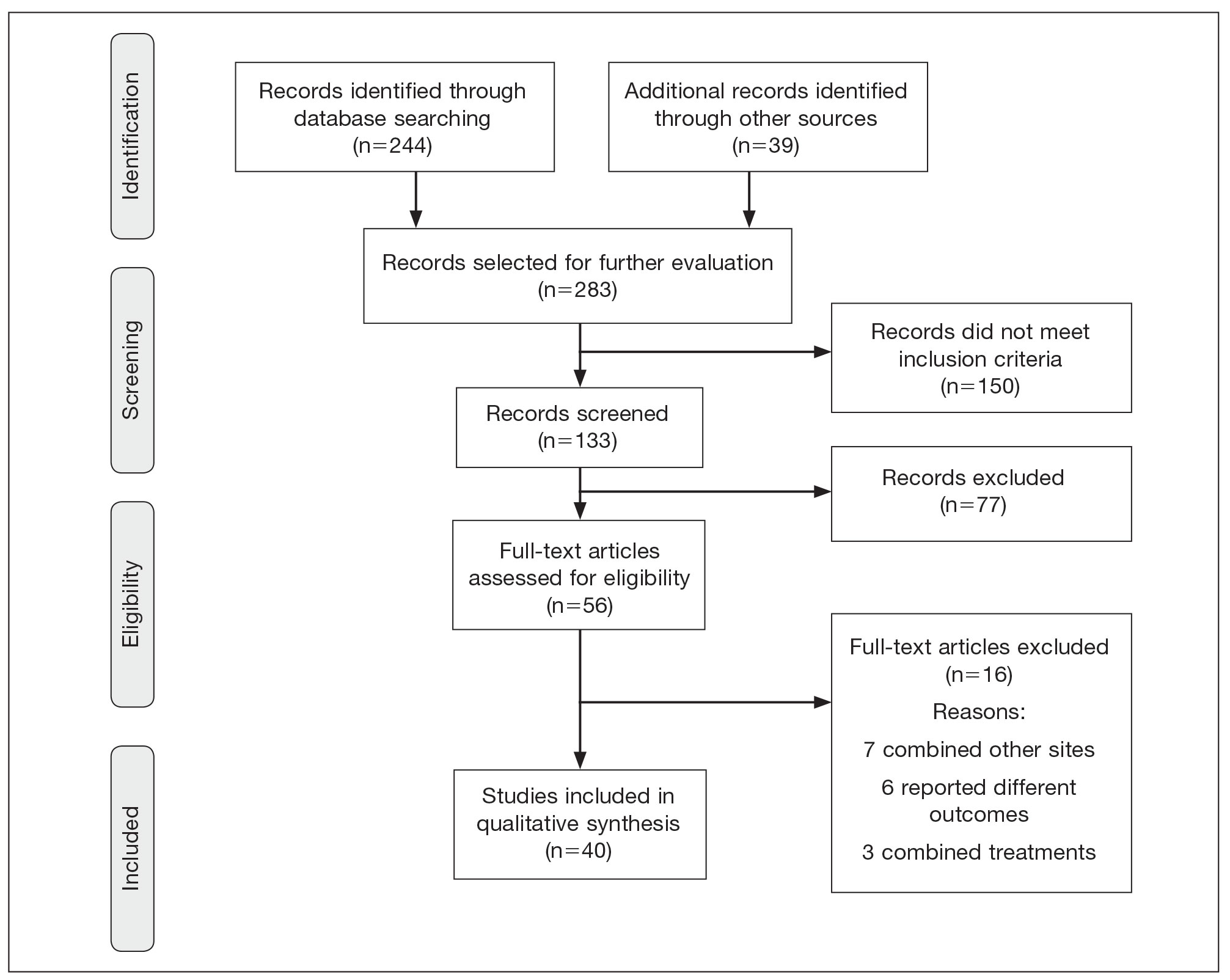
Risk of bias assessment identified low risk in 3 articles. High risk was identified in 5 case reports,57-61 and 1 study did not describe patient selection.43 Clayton et al56 showed intermediate risk, given the study controlled for 1 factor.
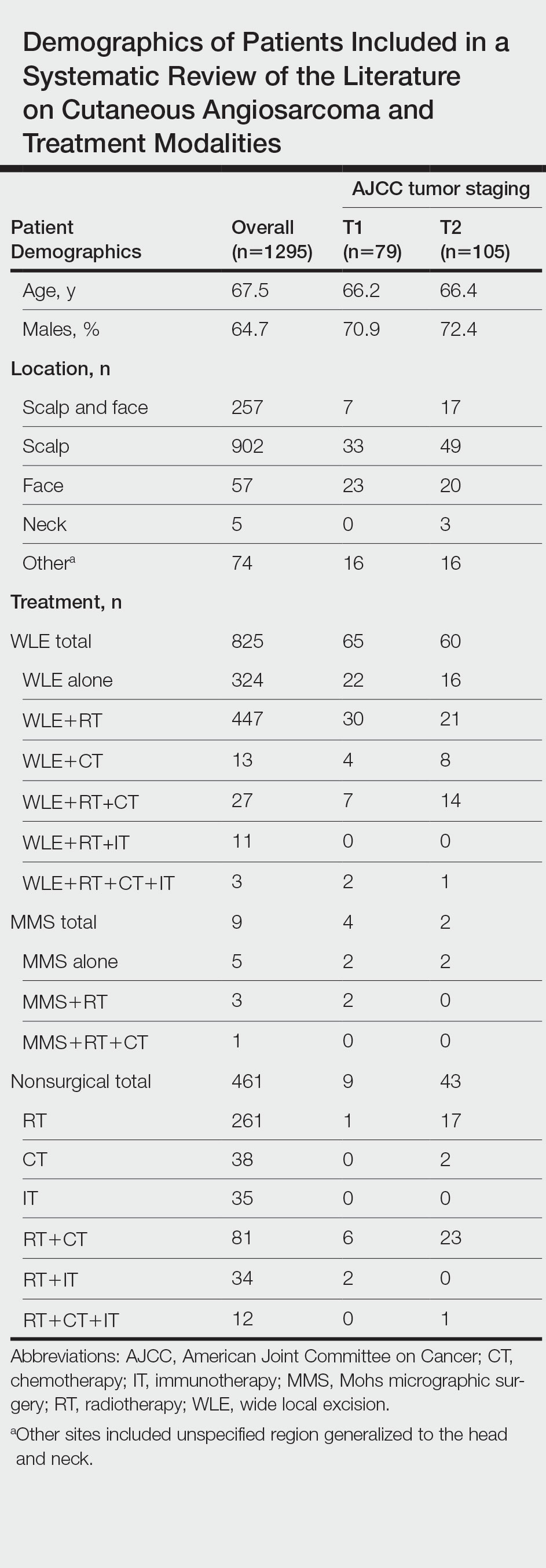
Patient Demographics
A total of 1295 patients were included. The pooled mean age of the patients was 67.5 years (range, 3–88 years), and 64.7% were male. There were 79 cases identified as T1 and 105 as T2. A total of 825 cases were treated using WLE with or without adjuvant therapy, while a total of 9 cases were treated using MMS with and without adjuvant therapies (Table). There were 461 cases treated without surgical excision: RT alone (n=261), CT alone (n=38), IT alone (n=35), RT with CT (n=81), RT with IT (n=34), and RT with CT and IT (n=12)(Table). The median follow-up period across all studies was 23.5 months (range, 1–228 months).
Comparison Between Surgical and Nonsurgical Modalities
Wide Local Excision—Wide local excision (n=825; 63.7%) alone or in combination with other therapies was the most frequently used treatment modality. The mean (SD) OS was longest for WLE with RT, CT, and IT (n=3; 39.3 [24.1]), followed by WLE with RT (n=447; 35.9 [34.3] months), WLE with CT (n=13; 32.4 [30.2] months), WLE alone (n=324; 29.6 [34.1] months), WLE with RT and IT (n=11; 23.5 [4.9] months), and WLE with RT and CT (n=27; 20.7 [13.1] months).
Nonsurgical Modalities—Nonsurgical methods were used less frequently than surgical methods (n=461; 35.6%). The mean (SD) OS time in descending order was as follows: RT with CT and IT (n=12; 34.9 [1.2] months), RT with CT (n=81; 30.4 [37.8] months), IT alone (n=35; 25.7 [no SD reported] months), RT with IT (n=34; 20.5 [8.6] months), CT alone (n=38; 20.1 [15.9] months), and RT alone (n=261; 12.8 [8.3] months).
When comparing mean (SD) OS outcomes between surgical and nonsurgical treatment modalities, only the addition of WLE to RT significantly increased OS when compared with RT alone (WLE, 35.9 [34.3] months; RT alone, 12.8 [8.3] months; P=.001). When WLE was added to CT or both RT and CT, there was no significant difference with OS when compared with CT alone (WLE with CT, 32.4 [30.2] months; CT alone, 20.1 [15.9] months; P=.065); or both RT and CT in combination (WLE with RT and CT, 20.7 [13.1] months; RT and CT, 30.4 [37.8] months; P=.204).
Comparison Between T1 and T2 cAS
T1 Angiosarcoma—There were 79 patients identified as having T1 tumors across 16 studies.2,31,32,34,39-41,46,48-50,53,58-60,62 The mean (SD) OS was longest for WLE with RT, CT, and IT (n=2; 56.0 [6.0] months), followed by WLE with CT (n=4; 54.5 [41.0] months); WLE with RT (n=30; 39.7 [41.2] months); WLE alone (n=22; 37.2 [37.3] months); WLE with both RT and CT (n=7; 25.5 [18.7] months); RT with IT (n=2; 20.0 [11.0] months); RT with CT (n=6; 15.7 [6.8] months); and RT alone (n=1; 13 [no SD]) months)(eTable).
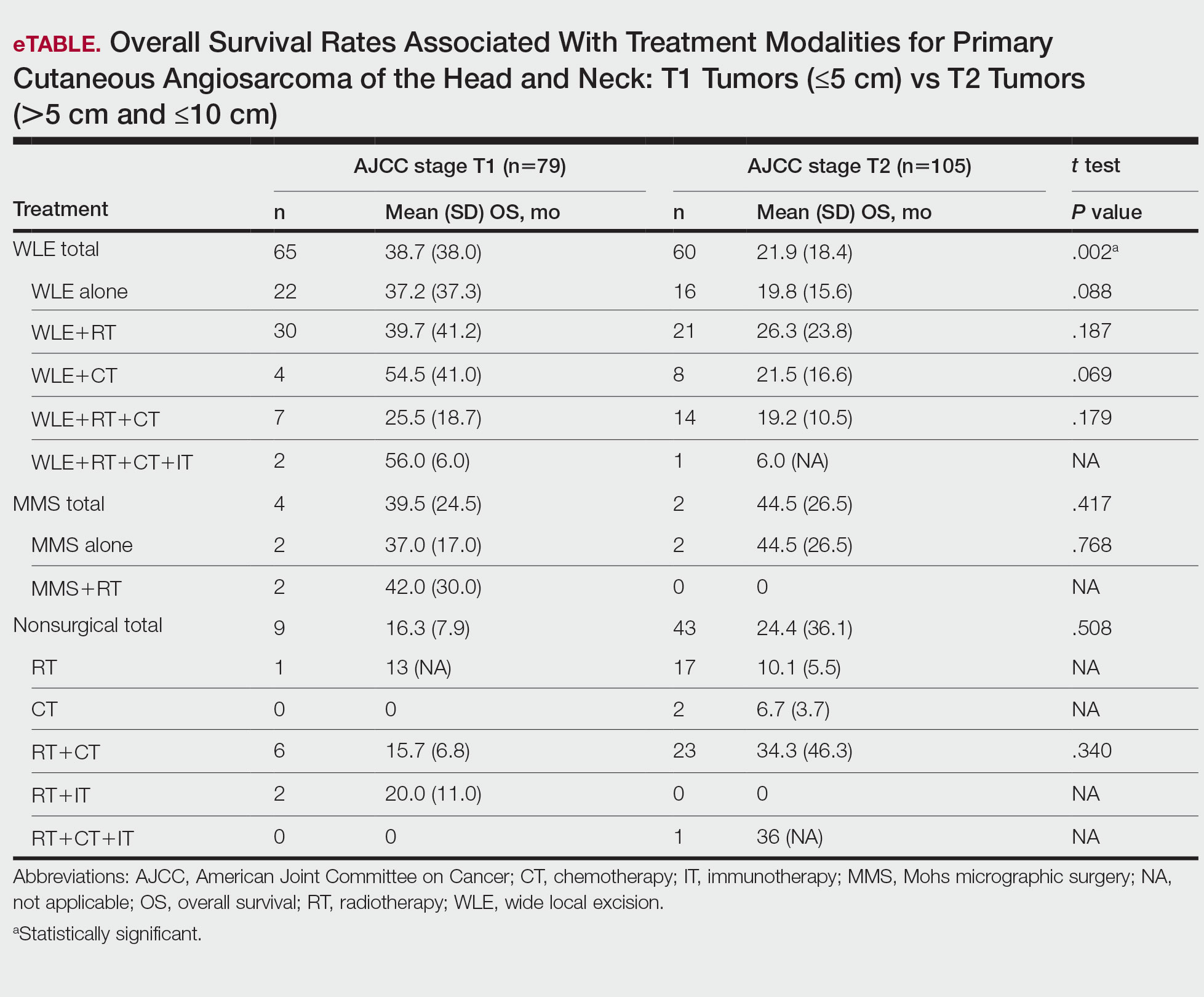
T2 Angiosarcoma—There were 105 patients with T2 tumors in 15 studies.2,31,32,34,39-41,46,48-50,52,53,57,62 The mean (SD) OS for each treatment modality in descending order was as follows: RT with CT and IT (n=1; 36 [no SD reported] months); RT with CT (n=23; 34.3 [46.3] months); WLE with RT (n=21; 26.3 [23.8] months); WLE with CT (n=8; 21.5 [16.6] months); WLE alone (n=16; 19.8 [15.6] months); WLE with RT and CT (n=14; 19.2 [10.5] months); RT alone (n=17; 10.1 [5.5] months); CT alone (n=2; 6.7 [3.7] months); and WLE with RT, CT, and IT (n=1; 6.0 [no SD] months)(eTable).
Mohs Micrographic Surgery—The use of MMS was only identified in case reports or small observational studies for a total of 9 patients. Five cASs were treated with MMS alone for a mean (SD) OS of 37 (21.5) months, with 4 reporting cAS staging: 2 were T158,59 (mean [SD] OS, 37.0 [17.0] months) and 2 were T2 tumors39,57 (mean [SD] OS, 44.5 [26.5] months). Mohs micrographic surgery with RT was used for 3 tumors (mean [SD] OS, 34.0 [26.9] months); 2 were T150,60 (mean [SD] OS, 42.0 [30.0] months) and 1 unreported staging (eTable).56 Mohs micrographic surgery with both RT and CT was used in 1 patient (unreported staging; OS, 82 months).51
Complications
Complications were rare and mainly associated with CT and RT. Four studies reported radiation dermatitis with RT.53,55,62,63 Two studies reported peripheral neuropathy and myelotoxicity with CT.35,51 Only 1 study reported poor wound healing due to surgical complications.29
COMMENT
Cutaneous angiosarcomas are rare and have limited treatment guidelines. Surgical excision does appear to be an effective adjunct to nonsurgical treatments, particularly WLE combined with RT, CT, and IT. Although MMS ultimately may be useful for cAS, the limited number and substantial heterogeneity of reported cases precludes definitive conclusions at this time.
Achieving margin control during WLE is associated with higher OS when treating angiosarcoma,36,46 which is particularly true for T1 tumors where margin control is imperative, and many cases are treated with a combination of WLE and RT. Overall survival times are lower for T2 tumors, as these tumors are larger and most likely have spread; therefore, more aggressive combination treatments were more prevalent. In these cases, complete margin control may be difficult to achieve and may not be as critical to the outcome if another form of adjuvant therapy can be administered promptly.24,64
When surgery is contraindicated, RT with or without CT was the most commonly reported treatment modality. However, these treatments were notably less effective than when used in combination with surgical resection. The use of RT alone has a recurrence rate reported up to 100% in certain studies, suggesting the need to utilize RT in combination with other modalities.23,39 It is important to note that RT often is used as monotherapy in palliative treatment, which may indirectly skew survival rates.2
Limitations of the study include a lack of randomized controlled trials. Most reports were retrospective reviews or case series, and tumor staging was sparsely reported. Finally, although MMS may provide utility in the treatment of cAS, the sample size of 9 precluded definitive conclusions from being formed about its efficacy.
CONCLUSION
Cutaneous angiosarcoma is rare and has limited data comparing different treatment modalities. The paucity of data currently limits definitive recommendations; however, both surgical and nonsurgical modalities have demonstrated potential efficacy in the treatment of cAS and may benefit from additional research. Clinicians should consider a multidisciplinary approach for patients with a diagnosis of cAS to tailor treatments on a case-by-case basis.
- Rodríguez-Jiménez P, Jimenez YD, Reolid A, et al. State of the art of Mohs surgery for rare cutaneous tumors in the Spanish Registry of Mohs Surgery (REGESMOHS). Int J Dermatol. 2020;59:321-325.
- Alqumber NA, Choi JW, Kang MK. The management and prognosis of facial and scalp angiosarcoma: a retrospective analysis of 15 patients. Ann Plast Surg. 2019;83:55-62.
- Pawlik TM, Paulino AF, McGinn CJ, et al. Cutaneous angiosarcoma of the scalp: a multidisciplinary approach. Cancer. 2003;98:1716-1726.
- Deyrup AT, McKenney JK, Tighiouart M, et al. Sporadic cutaneous angiosarcomas: a proposal for risk stratification based on 69 cases. Am J Surg Pathol. 2008;32:72-77.
- Meis-Kindblom JM, Kindblom LG. Angiosarcoma of soft tissue: a study of 80 cases. Am J Surg Pathol. 1998;22:683-697.
- Harbour P, Song DH. The skin and subcutaneous tissue. In: Brunicardi FC, Andersen DK, Billiar TR, et al, eds. Schwartz’s Principles of Surgery. 11th ed. McGraw-Hill Education; 2019. Accessed April 24, 2023. https://accesssurgery.mhmedical.com/content.aspx?bookid=2576§ionid=216206374
- Oashi K, Namikawa K, Tsutsumida A, et al. Surgery with curative intent is associated with prolonged survival in patients with cutaneous angiosarcoma of the scalp and face—a retrospective study of 38 untreated cases in the Japanese population. Eur J Surg Oncol. 2018;44:823-829.
- Young RJ, Brown NJ, Reed MW, et al. Angiosarcoma. Lancet Oncol. 2010;11:983-991.
- Tolkachjov SN, Brodland DG, Coldiron BM, et al. Understanding Mohs micrographic surgery: a review and practical guide for the nondermatologist. Mayo Clin Proc. 2017;92:1261-1271.
- Amin M, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. Springer; 2017.
- Holden CA, Spittle MF, Jones EW. Angiosarcoma of the face and scalp, prognosis and treatment. Cancer. 1987;59:1046-1057.
- Cook DA, Reed DA. Appraising the quality of medical education research methods: the Medical Education Research Study Quality Instrument and the Newcastle-Ottawa Scale-Education. Acad Med. 2015;90:1067-1076.
- Lee BL, Chen CF, Chen PC, et al. Investigation of prognostic features in primary cutaneous and soft tissue angiosarcoma after surgical resection: a retrospective study. Ann Plast Surg. 2017;78(3 suppl 2):S41-S46.
- Shen CJ, Parzuchowski AS, Kummerlowe MN, et al. Combined modality therapy improves overall survival for angiosarcoma. Acta Oncol. 2017;56:1235-1238.
- Breakey RW, Crowley TP, Anderson IB, et al. The surgical management of head and neck sarcoma: the Newcastle experience. J Plast Reconstr Aesthet Surg. 2017;70:78-84.
- Singla S, Papavasiliou P, Powers B, et al. Challenges in the treatment of angiosarcoma: a single institution experience. Am J Surg. 2014;208:254-259.
- Sasaki R, Soejima T, Kishi K, et al. Angiosarcoma treated with radiotherapy: impact of tumor type and size on outcome. Int J Radiat Oncol Biol Phys. 2002;52:1032-1040.
- Naka N, Ohsawa M, Tomita Y, et al. Angiosarcoma in Japan. A review of 99 cases. Cancer. 1995;75:989-996.
- DeMartelaere SL, Roberts D, Burgess MA, et al. Neoadjuvant chemotherapy-specific and overall treatment outcomes in patients with cutaneous angiosarcoma of the face with periorbital involvement. Head Neck. 2008;30:639-646.
- Ward JR, Feigenberg SJ, Mendenhall NP, et al. Radiation therapy for angiosarcoma. Head Neck. 2003;25:873-878.
- Letsa I, Benson C, Al-Muderis O, et al. Angiosarcoma of the face and scalp: effective systemic treatment in the older patient. J Geriatr Oncol. 2014;5:276-280.
- Buehler D, Rice SR, Moody JS, et al. Angiosarcoma outcomes and prognostic factors: a 25-year single institution experience. Am J Clin Oncol. 2014;37:473-479.
- Patel SH, Hayden RE, Hinni ML, et al. Angiosarcoma of the scalp and face: the Mayo Clinic experience. JAMA Otolaryngol Head Neck Surg. 2015;141:335-340.
- Guadagnolo BA, Zagars GK, Araujo D, et al. Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck. 2011;33:661-667.
- Zhang Y, Yan Y, Zhu M, et al. Clinical outcomes in primary scalp angiosarcoma. Oncol Lett. 2019;18:5091-5096.
- Kamo R, Ishii M. Histological differentiation, histogenesis and prognosis of cutaneous angiosarcoma. Osaka City Med J. 2011;57:31-44.
- Ito T, Uchi H, Nakahara T, et al. Cutaneous angiosarcoma of the head and face: a single-center analysis of treatment outcomes in 43 patients in Japan. J Cancer Res Clin Oncol. 2016;142:1387-1394.
- Aust MR, Olsen KD, Lewis JE, et al. Angiosarcomas of the head and neck: clinical and pathologic characteristics. Ann Otol Rhinol Laryngol. 1997;106:943-951.
- Buschmann A, Lehnhardt M, Toman N, et al. Surgical treatment of angiosarcoma of the scalp: less is more. Ann Plast Surg. 2008;61:399-403.
- Cassidy RJ, Switchenko JM, Yushak ML, et al. The importance of surgery in scalp angiosarcomas. Surg Oncol. 2018;27:A3-A8.
- Choi JH, Ahn KC, Chang H, et al. Surgical treatment and prognosis of angiosarcoma of the scalp: a retrospective analysis of 14 patients in a single institution. Biomed Res Int. 2015;2015:321896.
- Chow TL, Kwan WW, Kwan CK. Treatment of cutaneous angiosarcoma of the scalp and face in Chinese patients: local experience at a regional hospital in Hong Kong. Hong Kong Med J. 2018;24:25-31.
- Donghi D, Kerl K, Dummer R, et al. Cutaneous angiosarcoma: own experience over 13 years. clinical features, disease course and immunohistochemical profile. J Eur Acad Dermatol Venereol. 2010;24:1230-1234.
- Ferrari A, Casanova M, Bisogno G, et al. Malignant vascular tumors in children and adolescents: a report from the Italian and German Soft Tissue Sarcoma Cooperative Group. Med Pediatr Oncol. 2002;39:109-114.
- Fujisawa Y, Nakamura Y, Kawachi Y, et al. Comparison between taxane-based chemotherapy with conventional surgery-based therapy for cutaneous angiosarcoma: a single-center experience. J Dermatolog Treat. 2014;25:419-423.
- Hodgkinson DJ, Soule EH, Woods JE. Cutaneous angiosarcoma of the head and neck. Cancer. 1979;44:1106-1113.
- Lim SY, Pyon JK, Mun GH, et al. Surgical treatment of angiosarcoma of the scalp with superficial parotidectomy. Ann Plast Surg. 2010;64:180-182.
- Maddox JC, Evans HL. Angiosarcoma of skin and soft tissue: a study of forty-four cases. Cancer. 1981;48:1907-1921.
- Mark RJ, Tran LM, Sercarz J, et al. Angiosarcoma of the head and neck. The UCLA experience 1955 through 1990. Arch Otolaryngol Head Neck Surg. 1993;119:973-978.
- Morgan MB, Swann M, Somach S, et al. Cutaneous angiosarcoma: a case series with prognostic correlation. J Am Acad Dermatol. 2004;50:867-874.
- Mullins B, Hackman T. Angiosarcoma of the head and neck. Int Arch Otorhinolaryngol. 2015;19:191-195.
- Ogawa K, Takahashi K, Asato Y, et al. Treatment and prognosis of angiosarcoma of the scalp and face: a retrospective analysis of 48 patients. Br J Radiol. 2012;85:E1127-E1133.
- Panje WR, Moran WJ, Bostwick DG, et al. Angiosarcoma of the head and neck: review of 11 cases. Laryngoscope. 1986;96:1381-1384.
- Perez MC, Padhya TA, Messina JL, et al. Cutaneous angiosarcoma: a single-institution experience. Ann Surg Oncol. 2013;20:3391-3397.
- Veness M, Cooper S. Treatment of cutaneous angiosarcomas of the head and neck. Australas Radiol. 1995;39:277-281.
- Barttelbort SW, Stahl R, Ariyan S. Cutaneous angiosarcoma of the face and scalp. Plast Reconstr Surg. 1989;84:55-59.
- Bernstein JM, Irish JC, Brown DH, et al. Survival outcomes for cutaneous angiosarcoma of the scalp versus face. Head Neck. 2017;39:1205-1211.
- Köhler HF, Neves RI, Brechtbühl ER, et al. Cutaneous angiosarcoma of the head and neck: report of 23 cases from a single institution. Otolaryngol Head Neck Surg. 2008;139:519-524.
- Morales PH, Lindberg RD, Barkley HT Jr. Soft tissue angiosarcomas. Int J Radiat Oncol Biol Phys. 1981;7:1655-1659.
- Wollina U, Hansel G, Schönlebe J, et al. Cutaneous angiosarcoma is a rare aggressive malignant vascular tumour of the skin. J Eur Acad Dermatol Venereol. 2011;25:964-968.
- Wollina U, Koch A, Hansel G, et al. A 10-year analysis of cutaneous mesenchymal tumors (sarcomas and related entities) in a skin cancer center. Int J Dermatol. 2013;52:1189-1197.
- Bien E, Stachowicz-Stencel T, Balcerska A, et al. Angiosarcoma in children - still uncontrollable oncological problem. The report of the Polish Paediatric Rare Tumours Study. Eur J Cancer Care (Engl). 2009;18:411-420.
- Suzuki G, Yamazaki H, Takenaka H, et al. Definitive radiation therapy for angiosarcoma of the face and scalp. In Vivo. 2016;30:921-926.
- Miki Y, Tada T, Kamo R, et al. Single institutional experience of the treatment of angiosarcoma of the face and scalp. Br J Radiol. 2013;86:20130439.
- Ohguri T, Imada H, Nomoto S, et al. Angiosarcoma of the scalp treated with curative radiotherapy plus recombinant interleukin-2 immunotherapy. Int J Radiat Oncol Biol Phys. 2005;61:1446-1453.
- Clayton BD, Leshin B, Hitchcock MG, et al. Utility of rush paraffin-embedded tangential sections in the management of cutaneous neoplasms. Dermatol Surg. 2000;26:671-678.
- Goldberg DJ, Kim YA. Angiosarcoma of the scalp treated with Mohs micrographic surgery. J Dermatol Surg Oncol. 1993;19:156-158.
- Mikhail GR, Kelly AP Jr. Malignant angioendothelioma of the face. J Dermatol Surg Oncol. 1977;3:181-183.
- Muscarella VA. Angiosarcoma treated by Mohs micrographic surgery. J Dermatol Surg Oncol. 1993;19:1132-1133.
- Bullen R, Larson PO, Landeck AE, et al. Angiosarcoma of the head and neck managed by a combination of multiple biopsies to determine tumor margin and radiation therapy. report of three cases and review of the literature. Dermatol Surg. 1998;24:1105-1110.
- Wiwatwongwana D, White VA, Dolman PJ. Two cases of periocular cutaneous angiosarcoma. Ophthalmic Plast Reconstr Surg. 2010;26:365-366.
- Morrison WH, Byers RM, Garden AS, et al. Cutaneous angiosarcoma of the head and neck. A therapeutic dilemma. Cancer. 1995;76:319-327.
- Hata M, Wada H, Ogino I, et al. Radiation therapy for angiosarcoma of the scalp: treatment outcomes of total scalp irradiation with X-rays and electrons. Strahlenther Onkol. 2014;190:899-904.
- Hwang K, Kim MY, Lee SH. Recommendations for therapeutic decisions of angiosarcoma of the scalp and face. J Craniofac Surg. 2015;26:E253-E256.
Cutaneous angiosarcoma (cAS) is a rare malignancy arising from vascular or lymphatic tissue. It classically presents during the sixth or seventh decades of life as a raised purple papule or plaque on the head and neck areas.1 Primary cAS frequently mimics benign conditions, leading to delays in care. Such delays coupled with the aggressive nature of angiosarcomas leads to a poor prognosis. Five-year survival rates range from 11% to 50%, and more than half of patients die within 1 year of diagnosis.2-7
Currently, there is no consensus on the most effective treatments, as the rare nature of cAS has made the development of controlled clinical trials difficult. Wide local excision (WLE) is most frequently employed; however, the tumor’s infiltrative growth makes complete resection and negative surgical margins difficult to achieve.8 Recently, Mohs micrographic surgery (MMS) has been postulated as a treatment option. The tissue-sparing nature and intraoperative margin control of MMS may provide tumor eradication and cosmesis benefits reported with other cutaneous malignancies.9
Nearly all localized cASs are treated with surgical excision with or without adjuvant treatment modalities; however, it is unclear which of these modalities provide a survival benefit. We conducted a systematic review of the literature to compare treatment modalities for localized cAS of the head and neck regions and to compare treatments based on tumor stage.
METHODS
A literature search was performed to identify published studies indexed by MEDLINE, Cochrane Central Register of Controlled Trials (CENTRAL), Embase, and PubMed from January 1, 1977, to May 8, 2020, reporting on cAS and treatment modalities used. The search was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines.5 Data extracted included patient demographics, tumor characteristics (including T1 [≤5 cm] and T2 [>5 cm and ≤10 cm] based on the American Joint Committee on Cancer soft tissue sarcoma staging criteria), treatments used, follow-up time, overall survival (OS) rates, and complications.10,11
Studies were required to (1) include participants with head and neck cAS; (2) report original patient data following cAS treatment with surgical (WLE or MMS) and/or nonsurgical modalities (chemotherapy [CT], radiotherapy [RT], immunotherapy [IT]); (3) report outcome data related to OS rates following treatment; and (4) have articles published in English. Given the rare nature of cAS, there was no limitation on the number of participants needed.
The Newcastle-Ottawa scale for observational studies was used to assess the quality of studies.12 Higher scores indicate low risk of bias, while lower scores represent high risk of bias.
Continuous data were reported with means and SDs, while categorical variables were reported as percentages. Overall survival means and SDs were compared between treatment modalities using an independent sample t test with P<.05 considered statistically significant. Due to the heterogeneity of the data, a meta-analysis was not reported.
RESULTS
Literature Search and Risk of Bias Assessment
There were 283 manuscripts identified, 56 articles read in full, and 40 articles included in the review (Figure). Among the 16 studies not meeting inclusion criteria, 7 did not provide enough data to isolate head and neck cAS cases,1,13-18 6 did not report outcomes related to the current review,19-24 and 3 did not provide enough data to isolate different treatment outcomes.25-27 Among the included studies, 32 reported use of WLE: WLE alone (n=21)2,7,11,28-45; WLE with RT (n=24)2,3,11,28-31,33-36,38-41,43-51; WLE with CT (n=7)2,31,35,39,41,48,52; WLE with RT and CT (n=11)2,29,31,33-35,39,40,48,52,53; WLE with RT and IT (n=3)35,54,55; and WLE with RT, CT, and IT (n=1).53 Nine studies reported MMS: MMS alone (n=5)39,56-59; MMS with RT (n=3)32,50,60,61; and MMS with RT and CT (n=1).51

Risk of bias assessment identified low risk in 3 articles. High risk was identified in 5 case reports,57-61 and 1 study did not describe patient selection.43 Clayton et al56 showed intermediate risk, given the study controlled for 1 factor.
Patient Demographics
A total of 1295 patients were included. The pooled mean age of the patients was 67.5 years (range, 3–88 years), and 64.7% were male. There were 79 cases identified as T1 and 105 as T2. A total of 825 cases were treated using WLE with or without adjuvant therapy, while a total of 9 cases were treated using MMS with and without adjuvant therapies (Table). There were 461 cases treated without surgical excision: RT alone (n=261), CT alone (n=38), IT alone (n=35), RT with CT (n=81), RT with IT (n=34), and RT with CT and IT (n=12)(Table). The median follow-up period across all studies was 23.5 months (range, 1–228 months).

Comparison Between Surgical and Nonsurgical Modalities
Wide Local Excision—Wide local excision (n=825; 63.7%) alone or in combination with other therapies was the most frequently used treatment modality. The mean (SD) OS was longest for WLE with RT, CT, and IT (n=3; 39.3 [24.1]), followed by WLE with RT (n=447; 35.9 [34.3] months), WLE with CT (n=13; 32.4 [30.2] months), WLE alone (n=324; 29.6 [34.1] months), WLE with RT and IT (n=11; 23.5 [4.9] months), and WLE with RT and CT (n=27; 20.7 [13.1] months).
Nonsurgical Modalities—Nonsurgical methods were used less frequently than surgical methods (n=461; 35.6%). The mean (SD) OS time in descending order was as follows: RT with CT and IT (n=12; 34.9 [1.2] months), RT with CT (n=81; 30.4 [37.8] months), IT alone (n=35; 25.7 [no SD reported] months), RT with IT (n=34; 20.5 [8.6] months), CT alone (n=38; 20.1 [15.9] months), and RT alone (n=261; 12.8 [8.3] months).
When comparing mean (SD) OS outcomes between surgical and nonsurgical treatment modalities, only the addition of WLE to RT significantly increased OS when compared with RT alone (WLE, 35.9 [34.3] months; RT alone, 12.8 [8.3] months; P=.001). When WLE was added to CT or both RT and CT, there was no significant difference with OS when compared with CT alone (WLE with CT, 32.4 [30.2] months; CT alone, 20.1 [15.9] months; P=.065); or both RT and CT in combination (WLE with RT and CT, 20.7 [13.1] months; RT and CT, 30.4 [37.8] months; P=.204).
Comparison Between T1 and T2 cAS
T1 Angiosarcoma—There were 79 patients identified as having T1 tumors across 16 studies.2,31,32,34,39-41,46,48-50,53,58-60,62 The mean (SD) OS was longest for WLE with RT, CT, and IT (n=2; 56.0 [6.0] months), followed by WLE with CT (n=4; 54.5 [41.0] months); WLE with RT (n=30; 39.7 [41.2] months); WLE alone (n=22; 37.2 [37.3] months); WLE with both RT and CT (n=7; 25.5 [18.7] months); RT with IT (n=2; 20.0 [11.0] months); RT with CT (n=6; 15.7 [6.8] months); and RT alone (n=1; 13 [no SD]) months)(eTable).

T2 Angiosarcoma—There were 105 patients with T2 tumors in 15 studies.2,31,32,34,39-41,46,48-50,52,53,57,62 The mean (SD) OS for each treatment modality in descending order was as follows: RT with CT and IT (n=1; 36 [no SD reported] months); RT with CT (n=23; 34.3 [46.3] months); WLE with RT (n=21; 26.3 [23.8] months); WLE with CT (n=8; 21.5 [16.6] months); WLE alone (n=16; 19.8 [15.6] months); WLE with RT and CT (n=14; 19.2 [10.5] months); RT alone (n=17; 10.1 [5.5] months); CT alone (n=2; 6.7 [3.7] months); and WLE with RT, CT, and IT (n=1; 6.0 [no SD] months)(eTable).
Mohs Micrographic Surgery—The use of MMS was only identified in case reports or small observational studies for a total of 9 patients. Five cASs were treated with MMS alone for a mean (SD) OS of 37 (21.5) months, with 4 reporting cAS staging: 2 were T158,59 (mean [SD] OS, 37.0 [17.0] months) and 2 were T2 tumors39,57 (mean [SD] OS, 44.5 [26.5] months). Mohs micrographic surgery with RT was used for 3 tumors (mean [SD] OS, 34.0 [26.9] months); 2 were T150,60 (mean [SD] OS, 42.0 [30.0] months) and 1 unreported staging (eTable).56 Mohs micrographic surgery with both RT and CT was used in 1 patient (unreported staging; OS, 82 months).51
Complications
Complications were rare and mainly associated with CT and RT. Four studies reported radiation dermatitis with RT.53,55,62,63 Two studies reported peripheral neuropathy and myelotoxicity with CT.35,51 Only 1 study reported poor wound healing due to surgical complications.29
COMMENT
Cutaneous angiosarcomas are rare and have limited treatment guidelines. Surgical excision does appear to be an effective adjunct to nonsurgical treatments, particularly WLE combined with RT, CT, and IT. Although MMS ultimately may be useful for cAS, the limited number and substantial heterogeneity of reported cases precludes definitive conclusions at this time.
Achieving margin control during WLE is associated with higher OS when treating angiosarcoma,36,46 which is particularly true for T1 tumors where margin control is imperative, and many cases are treated with a combination of WLE and RT. Overall survival times are lower for T2 tumors, as these tumors are larger and most likely have spread; therefore, more aggressive combination treatments were more prevalent. In these cases, complete margin control may be difficult to achieve and may not be as critical to the outcome if another form of adjuvant therapy can be administered promptly.24,64
When surgery is contraindicated, RT with or without CT was the most commonly reported treatment modality. However, these treatments were notably less effective than when used in combination with surgical resection. The use of RT alone has a recurrence rate reported up to 100% in certain studies, suggesting the need to utilize RT in combination with other modalities.23,39 It is important to note that RT often is used as monotherapy in palliative treatment, which may indirectly skew survival rates.2
Limitations of the study include a lack of randomized controlled trials. Most reports were retrospective reviews or case series, and tumor staging was sparsely reported. Finally, although MMS may provide utility in the treatment of cAS, the sample size of 9 precluded definitive conclusions from being formed about its efficacy.
CONCLUSION
Cutaneous angiosarcoma is rare and has limited data comparing different treatment modalities. The paucity of data currently limits definitive recommendations; however, both surgical and nonsurgical modalities have demonstrated potential efficacy in the treatment of cAS and may benefit from additional research. Clinicians should consider a multidisciplinary approach for patients with a diagnosis of cAS to tailor treatments on a case-by-case basis.
Cutaneous angiosarcoma (cAS) is a rare malignancy arising from vascular or lymphatic tissue. It classically presents during the sixth or seventh decades of life as a raised purple papule or plaque on the head and neck areas.1 Primary cAS frequently mimics benign conditions, leading to delays in care. Such delays coupled with the aggressive nature of angiosarcomas leads to a poor prognosis. Five-year survival rates range from 11% to 50%, and more than half of patients die within 1 year of diagnosis.2-7
Currently, there is no consensus on the most effective treatments, as the rare nature of cAS has made the development of controlled clinical trials difficult. Wide local excision (WLE) is most frequently employed; however, the tumor’s infiltrative growth makes complete resection and negative surgical margins difficult to achieve.8 Recently, Mohs micrographic surgery (MMS) has been postulated as a treatment option. The tissue-sparing nature and intraoperative margin control of MMS may provide tumor eradication and cosmesis benefits reported with other cutaneous malignancies.9
Nearly all localized cASs are treated with surgical excision with or without adjuvant treatment modalities; however, it is unclear which of these modalities provide a survival benefit. We conducted a systematic review of the literature to compare treatment modalities for localized cAS of the head and neck regions and to compare treatments based on tumor stage.
METHODS
A literature search was performed to identify published studies indexed by MEDLINE, Cochrane Central Register of Controlled Trials (CENTRAL), Embase, and PubMed from January 1, 1977, to May 8, 2020, reporting on cAS and treatment modalities used. The search was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines.5 Data extracted included patient demographics, tumor characteristics (including T1 [≤5 cm] and T2 [>5 cm and ≤10 cm] based on the American Joint Committee on Cancer soft tissue sarcoma staging criteria), treatments used, follow-up time, overall survival (OS) rates, and complications.10,11
Studies were required to (1) include participants with head and neck cAS; (2) report original patient data following cAS treatment with surgical (WLE or MMS) and/or nonsurgical modalities (chemotherapy [CT], radiotherapy [RT], immunotherapy [IT]); (3) report outcome data related to OS rates following treatment; and (4) have articles published in English. Given the rare nature of cAS, there was no limitation on the number of participants needed.
The Newcastle-Ottawa scale for observational studies was used to assess the quality of studies.12 Higher scores indicate low risk of bias, while lower scores represent high risk of bias.
Continuous data were reported with means and SDs, while categorical variables were reported as percentages. Overall survival means and SDs were compared between treatment modalities using an independent sample t test with P<.05 considered statistically significant. Due to the heterogeneity of the data, a meta-analysis was not reported.
RESULTS
Literature Search and Risk of Bias Assessment
There were 283 manuscripts identified, 56 articles read in full, and 40 articles included in the review (Figure). Among the 16 studies not meeting inclusion criteria, 7 did not provide enough data to isolate head and neck cAS cases,1,13-18 6 did not report outcomes related to the current review,19-24 and 3 did not provide enough data to isolate different treatment outcomes.25-27 Among the included studies, 32 reported use of WLE: WLE alone (n=21)2,7,11,28-45; WLE with RT (n=24)2,3,11,28-31,33-36,38-41,43-51; WLE with CT (n=7)2,31,35,39,41,48,52; WLE with RT and CT (n=11)2,29,31,33-35,39,40,48,52,53; WLE with RT and IT (n=3)35,54,55; and WLE with RT, CT, and IT (n=1).53 Nine studies reported MMS: MMS alone (n=5)39,56-59; MMS with RT (n=3)32,50,60,61; and MMS with RT and CT (n=1).51

Risk of bias assessment identified low risk in 3 articles. High risk was identified in 5 case reports,57-61 and 1 study did not describe patient selection.43 Clayton et al56 showed intermediate risk, given the study controlled for 1 factor.
Patient Demographics
A total of 1295 patients were included. The pooled mean age of the patients was 67.5 years (range, 3–88 years), and 64.7% were male. There were 79 cases identified as T1 and 105 as T2. A total of 825 cases were treated using WLE with or without adjuvant therapy, while a total of 9 cases were treated using MMS with and without adjuvant therapies (Table). There were 461 cases treated without surgical excision: RT alone (n=261), CT alone (n=38), IT alone (n=35), RT with CT (n=81), RT with IT (n=34), and RT with CT and IT (n=12)(Table). The median follow-up period across all studies was 23.5 months (range, 1–228 months).

Comparison Between Surgical and Nonsurgical Modalities
Wide Local Excision—Wide local excision (n=825; 63.7%) alone or in combination with other therapies was the most frequently used treatment modality. The mean (SD) OS was longest for WLE with RT, CT, and IT (n=3; 39.3 [24.1]), followed by WLE with RT (n=447; 35.9 [34.3] months), WLE with CT (n=13; 32.4 [30.2] months), WLE alone (n=324; 29.6 [34.1] months), WLE with RT and IT (n=11; 23.5 [4.9] months), and WLE with RT and CT (n=27; 20.7 [13.1] months).
Nonsurgical Modalities—Nonsurgical methods were used less frequently than surgical methods (n=461; 35.6%). The mean (SD) OS time in descending order was as follows: RT with CT and IT (n=12; 34.9 [1.2] months), RT with CT (n=81; 30.4 [37.8] months), IT alone (n=35; 25.7 [no SD reported] months), RT with IT (n=34; 20.5 [8.6] months), CT alone (n=38; 20.1 [15.9] months), and RT alone (n=261; 12.8 [8.3] months).
When comparing mean (SD) OS outcomes between surgical and nonsurgical treatment modalities, only the addition of WLE to RT significantly increased OS when compared with RT alone (WLE, 35.9 [34.3] months; RT alone, 12.8 [8.3] months; P=.001). When WLE was added to CT or both RT and CT, there was no significant difference with OS when compared with CT alone (WLE with CT, 32.4 [30.2] months; CT alone, 20.1 [15.9] months; P=.065); or both RT and CT in combination (WLE with RT and CT, 20.7 [13.1] months; RT and CT, 30.4 [37.8] months; P=.204).
Comparison Between T1 and T2 cAS
T1 Angiosarcoma—There were 79 patients identified as having T1 tumors across 16 studies.2,31,32,34,39-41,46,48-50,53,58-60,62 The mean (SD) OS was longest for WLE with RT, CT, and IT (n=2; 56.0 [6.0] months), followed by WLE with CT (n=4; 54.5 [41.0] months); WLE with RT (n=30; 39.7 [41.2] months); WLE alone (n=22; 37.2 [37.3] months); WLE with both RT and CT (n=7; 25.5 [18.7] months); RT with IT (n=2; 20.0 [11.0] months); RT with CT (n=6; 15.7 [6.8] months); and RT alone (n=1; 13 [no SD]) months)(eTable).

T2 Angiosarcoma—There were 105 patients with T2 tumors in 15 studies.2,31,32,34,39-41,46,48-50,52,53,57,62 The mean (SD) OS for each treatment modality in descending order was as follows: RT with CT and IT (n=1; 36 [no SD reported] months); RT with CT (n=23; 34.3 [46.3] months); WLE with RT (n=21; 26.3 [23.8] months); WLE with CT (n=8; 21.5 [16.6] months); WLE alone (n=16; 19.8 [15.6] months); WLE with RT and CT (n=14; 19.2 [10.5] months); RT alone (n=17; 10.1 [5.5] months); CT alone (n=2; 6.7 [3.7] months); and WLE with RT, CT, and IT (n=1; 6.0 [no SD] months)(eTable).
Mohs Micrographic Surgery—The use of MMS was only identified in case reports or small observational studies for a total of 9 patients. Five cASs were treated with MMS alone for a mean (SD) OS of 37 (21.5) months, with 4 reporting cAS staging: 2 were T158,59 (mean [SD] OS, 37.0 [17.0] months) and 2 were T2 tumors39,57 (mean [SD] OS, 44.5 [26.5] months). Mohs micrographic surgery with RT was used for 3 tumors (mean [SD] OS, 34.0 [26.9] months); 2 were T150,60 (mean [SD] OS, 42.0 [30.0] months) and 1 unreported staging (eTable).56 Mohs micrographic surgery with both RT and CT was used in 1 patient (unreported staging; OS, 82 months).51
Complications
Complications were rare and mainly associated with CT and RT. Four studies reported radiation dermatitis with RT.53,55,62,63 Two studies reported peripheral neuropathy and myelotoxicity with CT.35,51 Only 1 study reported poor wound healing due to surgical complications.29
COMMENT
Cutaneous angiosarcomas are rare and have limited treatment guidelines. Surgical excision does appear to be an effective adjunct to nonsurgical treatments, particularly WLE combined with RT, CT, and IT. Although MMS ultimately may be useful for cAS, the limited number and substantial heterogeneity of reported cases precludes definitive conclusions at this time.
Achieving margin control during WLE is associated with higher OS when treating angiosarcoma,36,46 which is particularly true for T1 tumors where margin control is imperative, and many cases are treated with a combination of WLE and RT. Overall survival times are lower for T2 tumors, as these tumors are larger and most likely have spread; therefore, more aggressive combination treatments were more prevalent. In these cases, complete margin control may be difficult to achieve and may not be as critical to the outcome if another form of adjuvant therapy can be administered promptly.24,64
When surgery is contraindicated, RT with or without CT was the most commonly reported treatment modality. However, these treatments were notably less effective than when used in combination with surgical resection. The use of RT alone has a recurrence rate reported up to 100% in certain studies, suggesting the need to utilize RT in combination with other modalities.23,39 It is important to note that RT often is used as monotherapy in palliative treatment, which may indirectly skew survival rates.2
Limitations of the study include a lack of randomized controlled trials. Most reports were retrospective reviews or case series, and tumor staging was sparsely reported. Finally, although MMS may provide utility in the treatment of cAS, the sample size of 9 precluded definitive conclusions from being formed about its efficacy.
CONCLUSION
Cutaneous angiosarcoma is rare and has limited data comparing different treatment modalities. The paucity of data currently limits definitive recommendations; however, both surgical and nonsurgical modalities have demonstrated potential efficacy in the treatment of cAS and may benefit from additional research. Clinicians should consider a multidisciplinary approach for patients with a diagnosis of cAS to tailor treatments on a case-by-case basis.
- Rodríguez-Jiménez P, Jimenez YD, Reolid A, et al. State of the art of Mohs surgery for rare cutaneous tumors in the Spanish Registry of Mohs Surgery (REGESMOHS). Int J Dermatol. 2020;59:321-325.
- Alqumber NA, Choi JW, Kang MK. The management and prognosis of facial and scalp angiosarcoma: a retrospective analysis of 15 patients. Ann Plast Surg. 2019;83:55-62.
- Pawlik TM, Paulino AF, McGinn CJ, et al. Cutaneous angiosarcoma of the scalp: a multidisciplinary approach. Cancer. 2003;98:1716-1726.
- Deyrup AT, McKenney JK, Tighiouart M, et al. Sporadic cutaneous angiosarcomas: a proposal for risk stratification based on 69 cases. Am J Surg Pathol. 2008;32:72-77.
- Meis-Kindblom JM, Kindblom LG. Angiosarcoma of soft tissue: a study of 80 cases. Am J Surg Pathol. 1998;22:683-697.
- Harbour P, Song DH. The skin and subcutaneous tissue. In: Brunicardi FC, Andersen DK, Billiar TR, et al, eds. Schwartz’s Principles of Surgery. 11th ed. McGraw-Hill Education; 2019. Accessed April 24, 2023. https://accesssurgery.mhmedical.com/content.aspx?bookid=2576§ionid=216206374
- Oashi K, Namikawa K, Tsutsumida A, et al. Surgery with curative intent is associated with prolonged survival in patients with cutaneous angiosarcoma of the scalp and face—a retrospective study of 38 untreated cases in the Japanese population. Eur J Surg Oncol. 2018;44:823-829.
- Young RJ, Brown NJ, Reed MW, et al. Angiosarcoma. Lancet Oncol. 2010;11:983-991.
- Tolkachjov SN, Brodland DG, Coldiron BM, et al. Understanding Mohs micrographic surgery: a review and practical guide for the nondermatologist. Mayo Clin Proc. 2017;92:1261-1271.
- Amin M, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. Springer; 2017.
- Holden CA, Spittle MF, Jones EW. Angiosarcoma of the face and scalp, prognosis and treatment. Cancer. 1987;59:1046-1057.
- Cook DA, Reed DA. Appraising the quality of medical education research methods: the Medical Education Research Study Quality Instrument and the Newcastle-Ottawa Scale-Education. Acad Med. 2015;90:1067-1076.
- Lee BL, Chen CF, Chen PC, et al. Investigation of prognostic features in primary cutaneous and soft tissue angiosarcoma after surgical resection: a retrospective study. Ann Plast Surg. 2017;78(3 suppl 2):S41-S46.
- Shen CJ, Parzuchowski AS, Kummerlowe MN, et al. Combined modality therapy improves overall survival for angiosarcoma. Acta Oncol. 2017;56:1235-1238.
- Breakey RW, Crowley TP, Anderson IB, et al. The surgical management of head and neck sarcoma: the Newcastle experience. J Plast Reconstr Aesthet Surg. 2017;70:78-84.
- Singla S, Papavasiliou P, Powers B, et al. Challenges in the treatment of angiosarcoma: a single institution experience. Am J Surg. 2014;208:254-259.
- Sasaki R, Soejima T, Kishi K, et al. Angiosarcoma treated with radiotherapy: impact of tumor type and size on outcome. Int J Radiat Oncol Biol Phys. 2002;52:1032-1040.
- Naka N, Ohsawa M, Tomita Y, et al. Angiosarcoma in Japan. A review of 99 cases. Cancer. 1995;75:989-996.
- DeMartelaere SL, Roberts D, Burgess MA, et al. Neoadjuvant chemotherapy-specific and overall treatment outcomes in patients with cutaneous angiosarcoma of the face with periorbital involvement. Head Neck. 2008;30:639-646.
- Ward JR, Feigenberg SJ, Mendenhall NP, et al. Radiation therapy for angiosarcoma. Head Neck. 2003;25:873-878.
- Letsa I, Benson C, Al-Muderis O, et al. Angiosarcoma of the face and scalp: effective systemic treatment in the older patient. J Geriatr Oncol. 2014;5:276-280.
- Buehler D, Rice SR, Moody JS, et al. Angiosarcoma outcomes and prognostic factors: a 25-year single institution experience. Am J Clin Oncol. 2014;37:473-479.
- Patel SH, Hayden RE, Hinni ML, et al. Angiosarcoma of the scalp and face: the Mayo Clinic experience. JAMA Otolaryngol Head Neck Surg. 2015;141:335-340.
- Guadagnolo BA, Zagars GK, Araujo D, et al. Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck. 2011;33:661-667.
- Zhang Y, Yan Y, Zhu M, et al. Clinical outcomes in primary scalp angiosarcoma. Oncol Lett. 2019;18:5091-5096.
- Kamo R, Ishii M. Histological differentiation, histogenesis and prognosis of cutaneous angiosarcoma. Osaka City Med J. 2011;57:31-44.
- Ito T, Uchi H, Nakahara T, et al. Cutaneous angiosarcoma of the head and face: a single-center analysis of treatment outcomes in 43 patients in Japan. J Cancer Res Clin Oncol. 2016;142:1387-1394.
- Aust MR, Olsen KD, Lewis JE, et al. Angiosarcomas of the head and neck: clinical and pathologic characteristics. Ann Otol Rhinol Laryngol. 1997;106:943-951.
- Buschmann A, Lehnhardt M, Toman N, et al. Surgical treatment of angiosarcoma of the scalp: less is more. Ann Plast Surg. 2008;61:399-403.
- Cassidy RJ, Switchenko JM, Yushak ML, et al. The importance of surgery in scalp angiosarcomas. Surg Oncol. 2018;27:A3-A8.
- Choi JH, Ahn KC, Chang H, et al. Surgical treatment and prognosis of angiosarcoma of the scalp: a retrospective analysis of 14 patients in a single institution. Biomed Res Int. 2015;2015:321896.
- Chow TL, Kwan WW, Kwan CK. Treatment of cutaneous angiosarcoma of the scalp and face in Chinese patients: local experience at a regional hospital in Hong Kong. Hong Kong Med J. 2018;24:25-31.
- Donghi D, Kerl K, Dummer R, et al. Cutaneous angiosarcoma: own experience over 13 years. clinical features, disease course and immunohistochemical profile. J Eur Acad Dermatol Venereol. 2010;24:1230-1234.
- Ferrari A, Casanova M, Bisogno G, et al. Malignant vascular tumors in children and adolescents: a report from the Italian and German Soft Tissue Sarcoma Cooperative Group. Med Pediatr Oncol. 2002;39:109-114.
- Fujisawa Y, Nakamura Y, Kawachi Y, et al. Comparison between taxane-based chemotherapy with conventional surgery-based therapy for cutaneous angiosarcoma: a single-center experience. J Dermatolog Treat. 2014;25:419-423.
- Hodgkinson DJ, Soule EH, Woods JE. Cutaneous angiosarcoma of the head and neck. Cancer. 1979;44:1106-1113.
- Lim SY, Pyon JK, Mun GH, et al. Surgical treatment of angiosarcoma of the scalp with superficial parotidectomy. Ann Plast Surg. 2010;64:180-182.
- Maddox JC, Evans HL. Angiosarcoma of skin and soft tissue: a study of forty-four cases. Cancer. 1981;48:1907-1921.
- Mark RJ, Tran LM, Sercarz J, et al. Angiosarcoma of the head and neck. The UCLA experience 1955 through 1990. Arch Otolaryngol Head Neck Surg. 1993;119:973-978.
- Morgan MB, Swann M, Somach S, et al. Cutaneous angiosarcoma: a case series with prognostic correlation. J Am Acad Dermatol. 2004;50:867-874.
- Mullins B, Hackman T. Angiosarcoma of the head and neck. Int Arch Otorhinolaryngol. 2015;19:191-195.
- Ogawa K, Takahashi K, Asato Y, et al. Treatment and prognosis of angiosarcoma of the scalp and face: a retrospective analysis of 48 patients. Br J Radiol. 2012;85:E1127-E1133.
- Panje WR, Moran WJ, Bostwick DG, et al. Angiosarcoma of the head and neck: review of 11 cases. Laryngoscope. 1986;96:1381-1384.
- Perez MC, Padhya TA, Messina JL, et al. Cutaneous angiosarcoma: a single-institution experience. Ann Surg Oncol. 2013;20:3391-3397.
- Veness M, Cooper S. Treatment of cutaneous angiosarcomas of the head and neck. Australas Radiol. 1995;39:277-281.
- Barttelbort SW, Stahl R, Ariyan S. Cutaneous angiosarcoma of the face and scalp. Plast Reconstr Surg. 1989;84:55-59.
- Bernstein JM, Irish JC, Brown DH, et al. Survival outcomes for cutaneous angiosarcoma of the scalp versus face. Head Neck. 2017;39:1205-1211.
- Köhler HF, Neves RI, Brechtbühl ER, et al. Cutaneous angiosarcoma of the head and neck: report of 23 cases from a single institution. Otolaryngol Head Neck Surg. 2008;139:519-524.
- Morales PH, Lindberg RD, Barkley HT Jr. Soft tissue angiosarcomas. Int J Radiat Oncol Biol Phys. 1981;7:1655-1659.
- Wollina U, Hansel G, Schönlebe J, et al. Cutaneous angiosarcoma is a rare aggressive malignant vascular tumour of the skin. J Eur Acad Dermatol Venereol. 2011;25:964-968.
- Wollina U, Koch A, Hansel G, et al. A 10-year analysis of cutaneous mesenchymal tumors (sarcomas and related entities) in a skin cancer center. Int J Dermatol. 2013;52:1189-1197.
- Bien E, Stachowicz-Stencel T, Balcerska A, et al. Angiosarcoma in children - still uncontrollable oncological problem. The report of the Polish Paediatric Rare Tumours Study. Eur J Cancer Care (Engl). 2009;18:411-420.
- Suzuki G, Yamazaki H, Takenaka H, et al. Definitive radiation therapy for angiosarcoma of the face and scalp. In Vivo. 2016;30:921-926.
- Miki Y, Tada T, Kamo R, et al. Single institutional experience of the treatment of angiosarcoma of the face and scalp. Br J Radiol. 2013;86:20130439.
- Ohguri T, Imada H, Nomoto S, et al. Angiosarcoma of the scalp treated with curative radiotherapy plus recombinant interleukin-2 immunotherapy. Int J Radiat Oncol Biol Phys. 2005;61:1446-1453.
- Clayton BD, Leshin B, Hitchcock MG, et al. Utility of rush paraffin-embedded tangential sections in the management of cutaneous neoplasms. Dermatol Surg. 2000;26:671-678.
- Goldberg DJ, Kim YA. Angiosarcoma of the scalp treated with Mohs micrographic surgery. J Dermatol Surg Oncol. 1993;19:156-158.
- Mikhail GR, Kelly AP Jr. Malignant angioendothelioma of the face. J Dermatol Surg Oncol. 1977;3:181-183.
- Muscarella VA. Angiosarcoma treated by Mohs micrographic surgery. J Dermatol Surg Oncol. 1993;19:1132-1133.
- Bullen R, Larson PO, Landeck AE, et al. Angiosarcoma of the head and neck managed by a combination of multiple biopsies to determine tumor margin and radiation therapy. report of three cases and review of the literature. Dermatol Surg. 1998;24:1105-1110.
- Wiwatwongwana D, White VA, Dolman PJ. Two cases of periocular cutaneous angiosarcoma. Ophthalmic Plast Reconstr Surg. 2010;26:365-366.
- Morrison WH, Byers RM, Garden AS, et al. Cutaneous angiosarcoma of the head and neck. A therapeutic dilemma. Cancer. 1995;76:319-327.
- Hata M, Wada H, Ogino I, et al. Radiation therapy for angiosarcoma of the scalp: treatment outcomes of total scalp irradiation with X-rays and electrons. Strahlenther Onkol. 2014;190:899-904.
- Hwang K, Kim MY, Lee SH. Recommendations for therapeutic decisions of angiosarcoma of the scalp and face. J Craniofac Surg. 2015;26:E253-E256.
- Rodríguez-Jiménez P, Jimenez YD, Reolid A, et al. State of the art of Mohs surgery for rare cutaneous tumors in the Spanish Registry of Mohs Surgery (REGESMOHS). Int J Dermatol. 2020;59:321-325.
- Alqumber NA, Choi JW, Kang MK. The management and prognosis of facial and scalp angiosarcoma: a retrospective analysis of 15 patients. Ann Plast Surg. 2019;83:55-62.
- Pawlik TM, Paulino AF, McGinn CJ, et al. Cutaneous angiosarcoma of the scalp: a multidisciplinary approach. Cancer. 2003;98:1716-1726.
- Deyrup AT, McKenney JK, Tighiouart M, et al. Sporadic cutaneous angiosarcomas: a proposal for risk stratification based on 69 cases. Am J Surg Pathol. 2008;32:72-77.
- Meis-Kindblom JM, Kindblom LG. Angiosarcoma of soft tissue: a study of 80 cases. Am J Surg Pathol. 1998;22:683-697.
- Harbour P, Song DH. The skin and subcutaneous tissue. In: Brunicardi FC, Andersen DK, Billiar TR, et al, eds. Schwartz’s Principles of Surgery. 11th ed. McGraw-Hill Education; 2019. Accessed April 24, 2023. https://accesssurgery.mhmedical.com/content.aspx?bookid=2576§ionid=216206374
- Oashi K, Namikawa K, Tsutsumida A, et al. Surgery with curative intent is associated with prolonged survival in patients with cutaneous angiosarcoma of the scalp and face—a retrospective study of 38 untreated cases in the Japanese population. Eur J Surg Oncol. 2018;44:823-829.
- Young RJ, Brown NJ, Reed MW, et al. Angiosarcoma. Lancet Oncol. 2010;11:983-991.
- Tolkachjov SN, Brodland DG, Coldiron BM, et al. Understanding Mohs micrographic surgery: a review and practical guide for the nondermatologist. Mayo Clin Proc. 2017;92:1261-1271.
- Amin M, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. Springer; 2017.
- Holden CA, Spittle MF, Jones EW. Angiosarcoma of the face and scalp, prognosis and treatment. Cancer. 1987;59:1046-1057.
- Cook DA, Reed DA. Appraising the quality of medical education research methods: the Medical Education Research Study Quality Instrument and the Newcastle-Ottawa Scale-Education. Acad Med. 2015;90:1067-1076.
- Lee BL, Chen CF, Chen PC, et al. Investigation of prognostic features in primary cutaneous and soft tissue angiosarcoma after surgical resection: a retrospective study. Ann Plast Surg. 2017;78(3 suppl 2):S41-S46.
- Shen CJ, Parzuchowski AS, Kummerlowe MN, et al. Combined modality therapy improves overall survival for angiosarcoma. Acta Oncol. 2017;56:1235-1238.
- Breakey RW, Crowley TP, Anderson IB, et al. The surgical management of head and neck sarcoma: the Newcastle experience. J Plast Reconstr Aesthet Surg. 2017;70:78-84.
- Singla S, Papavasiliou P, Powers B, et al. Challenges in the treatment of angiosarcoma: a single institution experience. Am J Surg. 2014;208:254-259.
- Sasaki R, Soejima T, Kishi K, et al. Angiosarcoma treated with radiotherapy: impact of tumor type and size on outcome. Int J Radiat Oncol Biol Phys. 2002;52:1032-1040.
- Naka N, Ohsawa M, Tomita Y, et al. Angiosarcoma in Japan. A review of 99 cases. Cancer. 1995;75:989-996.
- DeMartelaere SL, Roberts D, Burgess MA, et al. Neoadjuvant chemotherapy-specific and overall treatment outcomes in patients with cutaneous angiosarcoma of the face with periorbital involvement. Head Neck. 2008;30:639-646.
- Ward JR, Feigenberg SJ, Mendenhall NP, et al. Radiation therapy for angiosarcoma. Head Neck. 2003;25:873-878.
- Letsa I, Benson C, Al-Muderis O, et al. Angiosarcoma of the face and scalp: effective systemic treatment in the older patient. J Geriatr Oncol. 2014;5:276-280.
- Buehler D, Rice SR, Moody JS, et al. Angiosarcoma outcomes and prognostic factors: a 25-year single institution experience. Am J Clin Oncol. 2014;37:473-479.
- Patel SH, Hayden RE, Hinni ML, et al. Angiosarcoma of the scalp and face: the Mayo Clinic experience. JAMA Otolaryngol Head Neck Surg. 2015;141:335-340.
- Guadagnolo BA, Zagars GK, Araujo D, et al. Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck. 2011;33:661-667.
- Zhang Y, Yan Y, Zhu M, et al. Clinical outcomes in primary scalp angiosarcoma. Oncol Lett. 2019;18:5091-5096.
- Kamo R, Ishii M. Histological differentiation, histogenesis and prognosis of cutaneous angiosarcoma. Osaka City Med J. 2011;57:31-44.
- Ito T, Uchi H, Nakahara T, et al. Cutaneous angiosarcoma of the head and face: a single-center analysis of treatment outcomes in 43 patients in Japan. J Cancer Res Clin Oncol. 2016;142:1387-1394.
- Aust MR, Olsen KD, Lewis JE, et al. Angiosarcomas of the head and neck: clinical and pathologic characteristics. Ann Otol Rhinol Laryngol. 1997;106:943-951.
- Buschmann A, Lehnhardt M, Toman N, et al. Surgical treatment of angiosarcoma of the scalp: less is more. Ann Plast Surg. 2008;61:399-403.
- Cassidy RJ, Switchenko JM, Yushak ML, et al. The importance of surgery in scalp angiosarcomas. Surg Oncol. 2018;27:A3-A8.
- Choi JH, Ahn KC, Chang H, et al. Surgical treatment and prognosis of angiosarcoma of the scalp: a retrospective analysis of 14 patients in a single institution. Biomed Res Int. 2015;2015:321896.
- Chow TL, Kwan WW, Kwan CK. Treatment of cutaneous angiosarcoma of the scalp and face in Chinese patients: local experience at a regional hospital in Hong Kong. Hong Kong Med J. 2018;24:25-31.
- Donghi D, Kerl K, Dummer R, et al. Cutaneous angiosarcoma: own experience over 13 years. clinical features, disease course and immunohistochemical profile. J Eur Acad Dermatol Venereol. 2010;24:1230-1234.
- Ferrari A, Casanova M, Bisogno G, et al. Malignant vascular tumors in children and adolescents: a report from the Italian and German Soft Tissue Sarcoma Cooperative Group. Med Pediatr Oncol. 2002;39:109-114.
- Fujisawa Y, Nakamura Y, Kawachi Y, et al. Comparison between taxane-based chemotherapy with conventional surgery-based therapy for cutaneous angiosarcoma: a single-center experience. J Dermatolog Treat. 2014;25:419-423.
- Hodgkinson DJ, Soule EH, Woods JE. Cutaneous angiosarcoma of the head and neck. Cancer. 1979;44:1106-1113.
- Lim SY, Pyon JK, Mun GH, et al. Surgical treatment of angiosarcoma of the scalp with superficial parotidectomy. Ann Plast Surg. 2010;64:180-182.
- Maddox JC, Evans HL. Angiosarcoma of skin and soft tissue: a study of forty-four cases. Cancer. 1981;48:1907-1921.
- Mark RJ, Tran LM, Sercarz J, et al. Angiosarcoma of the head and neck. The UCLA experience 1955 through 1990. Arch Otolaryngol Head Neck Surg. 1993;119:973-978.
- Morgan MB, Swann M, Somach S, et al. Cutaneous angiosarcoma: a case series with prognostic correlation. J Am Acad Dermatol. 2004;50:867-874.
- Mullins B, Hackman T. Angiosarcoma of the head and neck. Int Arch Otorhinolaryngol. 2015;19:191-195.
- Ogawa K, Takahashi K, Asato Y, et al. Treatment and prognosis of angiosarcoma of the scalp and face: a retrospective analysis of 48 patients. Br J Radiol. 2012;85:E1127-E1133.
- Panje WR, Moran WJ, Bostwick DG, et al. Angiosarcoma of the head and neck: review of 11 cases. Laryngoscope. 1986;96:1381-1384.
- Perez MC, Padhya TA, Messina JL, et al. Cutaneous angiosarcoma: a single-institution experience. Ann Surg Oncol. 2013;20:3391-3397.
- Veness M, Cooper S. Treatment of cutaneous angiosarcomas of the head and neck. Australas Radiol. 1995;39:277-281.
- Barttelbort SW, Stahl R, Ariyan S. Cutaneous angiosarcoma of the face and scalp. Plast Reconstr Surg. 1989;84:55-59.
- Bernstein JM, Irish JC, Brown DH, et al. Survival outcomes for cutaneous angiosarcoma of the scalp versus face. Head Neck. 2017;39:1205-1211.
- Köhler HF, Neves RI, Brechtbühl ER, et al. Cutaneous angiosarcoma of the head and neck: report of 23 cases from a single institution. Otolaryngol Head Neck Surg. 2008;139:519-524.
- Morales PH, Lindberg RD, Barkley HT Jr. Soft tissue angiosarcomas. Int J Radiat Oncol Biol Phys. 1981;7:1655-1659.
- Wollina U, Hansel G, Schönlebe J, et al. Cutaneous angiosarcoma is a rare aggressive malignant vascular tumour of the skin. J Eur Acad Dermatol Venereol. 2011;25:964-968.
- Wollina U, Koch A, Hansel G, et al. A 10-year analysis of cutaneous mesenchymal tumors (sarcomas and related entities) in a skin cancer center. Int J Dermatol. 2013;52:1189-1197.
- Bien E, Stachowicz-Stencel T, Balcerska A, et al. Angiosarcoma in children - still uncontrollable oncological problem. The report of the Polish Paediatric Rare Tumours Study. Eur J Cancer Care (Engl). 2009;18:411-420.
- Suzuki G, Yamazaki H, Takenaka H, et al. Definitive radiation therapy for angiosarcoma of the face and scalp. In Vivo. 2016;30:921-926.
- Miki Y, Tada T, Kamo R, et al. Single institutional experience of the treatment of angiosarcoma of the face and scalp. Br J Radiol. 2013;86:20130439.
- Ohguri T, Imada H, Nomoto S, et al. Angiosarcoma of the scalp treated with curative radiotherapy plus recombinant interleukin-2 immunotherapy. Int J Radiat Oncol Biol Phys. 2005;61:1446-1453.
- Clayton BD, Leshin B, Hitchcock MG, et al. Utility of rush paraffin-embedded tangential sections in the management of cutaneous neoplasms. Dermatol Surg. 2000;26:671-678.
- Goldberg DJ, Kim YA. Angiosarcoma of the scalp treated with Mohs micrographic surgery. J Dermatol Surg Oncol. 1993;19:156-158.
- Mikhail GR, Kelly AP Jr. Malignant angioendothelioma of the face. J Dermatol Surg Oncol. 1977;3:181-183.
- Muscarella VA. Angiosarcoma treated by Mohs micrographic surgery. J Dermatol Surg Oncol. 1993;19:1132-1133.
- Bullen R, Larson PO, Landeck AE, et al. Angiosarcoma of the head and neck managed by a combination of multiple biopsies to determine tumor margin and radiation therapy. report of three cases and review of the literature. Dermatol Surg. 1998;24:1105-1110.
- Wiwatwongwana D, White VA, Dolman PJ. Two cases of periocular cutaneous angiosarcoma. Ophthalmic Plast Reconstr Surg. 2010;26:365-366.
- Morrison WH, Byers RM, Garden AS, et al. Cutaneous angiosarcoma of the head and neck. A therapeutic dilemma. Cancer. 1995;76:319-327.
- Hata M, Wada H, Ogino I, et al. Radiation therapy for angiosarcoma of the scalp: treatment outcomes of total scalp irradiation with X-rays and electrons. Strahlenther Onkol. 2014;190:899-904.
- Hwang K, Kim MY, Lee SH. Recommendations for therapeutic decisions of angiosarcoma of the scalp and face. J Craniofac Surg. 2015;26:E253-E256.
Practice Points
- Angiosarcoma is a rare tumor that is difficult to treat, with multiple treatment options being utilized.
- Within this systematic review, wide local excision (WLE) combined with radiotherapy (RT), chemotherapy, and immunotherapy, as well as Mohs micrographic surgery (MMS), offered the longest mean (SD) overall survival time.
- When clinicians are tasked with treating primary cutaneous angiosarcoma of the head and neck, they should consider MMS or WLE combined with RT.
Scalp Nodule Associated With Hair Loss
The Diagnosis: Alopecic and Aseptic Nodule of the Scalp
Alopecic and aseptic nodule of the scalp (AANS) is an underdiagnosed condition presenting with one or few inflammatory nodules on the scalp with overlying nonscarring alopecia. The nodules can be soft, fluctuant, or firm and are characterized by negative fungal and bacterial stains as well as cultures.1 Trichoscopic features such as black or yellow dots, fine vellus hairs, and broken hairs have been reported.1-3 Dilated follicular openings may be seen and are termed the Eastern pancake sign, as they resemble the bubble cavities formed during the cooking of atayef.2 The histologic features of AANS often are nonspecific but show a nodular or pseudocystic, lymphohistiocytic to acute inflammatory component centered in the dermis.1 Granulomatous inflammation or isolated giant cells have been reported within the deep dermis.1,4 In our patient, histopathology revealed admixed acute and granulomatous inflammation within the deep dermis (Figure). Treatment of AANS includes oral antibiotics such as doxycycline, intralesional corticosteroids, or excision.1
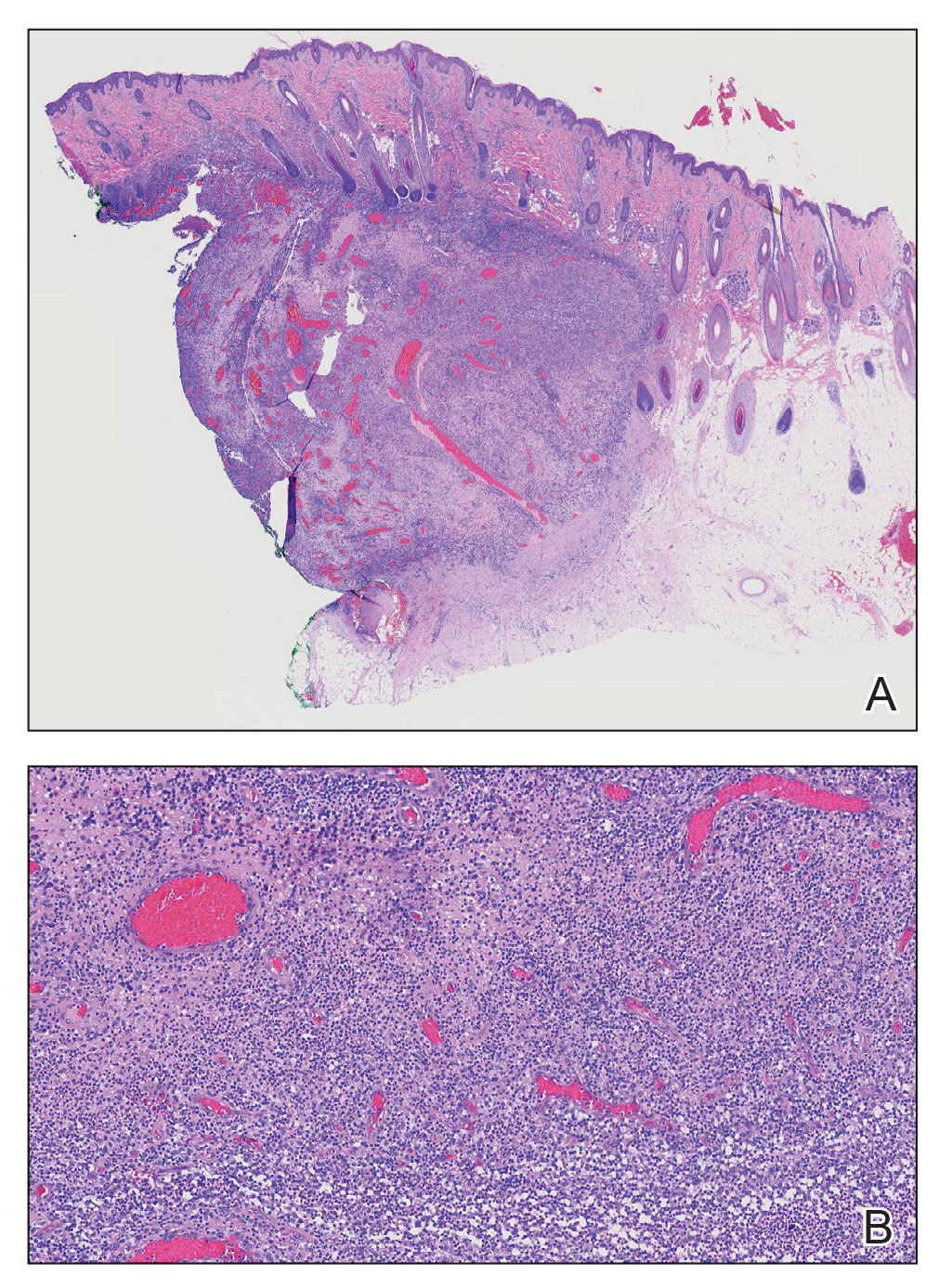
Although the etiology of AANS currently is unclear, a process of follicular plugging or a deep folliculitis sparing the bulge stem cells has been theorized. Young males are disproportionately affected.1 It is uncertain how much overlap there is, if any, between AANS and pseudocyst of the scalp, the latter of which primarily is reported in the Japanese literature and demonstrates alopecic nodules between the forehead and vertex of the scalp with pseudocystic architecture and granulomatous infiltration on histopathology.4-7
There are several clinical and histologic differences between AANS and other diagnoses in the differential. Dermoid cysts tend to present at birth, with 70% of cases presenting before the age of 6 years, and without overlying skin changes.8 They represent a benign entrapment of ectoderm along embryonic closure lines during development.9 Histologic examination typically will show a squamous-lined cyst within the dermis with associated adnexal structures.10 Cylindromas are benign neoplasms of eccrine sweat glands named after the histologic presentation of cylinder-shaped basaloid cell populations when cross-sectioned.11,12 When cylindromas coalesce on the scalp, they form a distinctive morphology sometimes loosely resembling a turban, giving them the previously more common name turban tumors.11,13 Cylindromas appear as slow-growing protuberant tumors that are erythematous or flesh colored. Cylindromas are 9 times more common in females.13 Pilar cysts have a stratified squamous epithelium lining with a palisaded outer layer and are derived from the outer root sheath of hair follicles.14 Clinically, pilar cysts are smooth mobile cysts that favor skin with a dense concentration of hair follicles.14,15 On palpation, pilar cysts are firm due to their keratinous contents and typically are nontender unless inflamed.15 Lipomas are benign mesenchymal tumors with mature adipocytes that often appear as subcutaneous nodules without overlying skin changes, though they can involve deep fascia. On palpation, lipomas generally are soft, mobile, and nontender.16
- Bellinato F, Maurelli M, Colato C, et al. Alopecic and aseptic nodules of the scalp: a new case with a systematic review of the literature [published online May 1, 2021]. Clin Case Rep. 2021;9:E04153. doi:10.1002/ccr3.4153
- Lázaro-Simó AI, Sancho MI, Quintana-Codina M, et al. Alopecic and aseptic nodules of the scalp with trichoscopic and ultrasonographic findings. Indian J Dermatol. 2017;62:515-518.
- Garrido-Colmenero C, Arias-Santiago S, Aneiros Fernández J, et al. Trichoscopy and ultrasonography features of aseptic and alopecic nodules of the scalp. J Eur Acad Dermatol Venereol. 2016;30:507-509. doi:10.1111/jdv.12903
- Seol JE, Park IH, Kim DH, et al. Alopecic and aseptic nodules of the scalp/pseudocyst of the scalp: clinicopathological and therapeutic analyses in 11 Korean patients. Dermatology. 2016;232:165-170.
- Lee SS, Kim SY, Im M, et al. Pseudocyst of the scalp. Ann Dermatol. 2011;23(suppl 2):S267-S269.
- Eisenberg EL. Alopecia-associated pseudocyst of the scalp. J Am Acad Dermatol. 2012;67:E114-E116.
- Tsuruta D, Hayashi A, Kobayashi H, et al. Pseudocyst of the scalp. Dermatology. 2005;210:333-335.
- Orozco-Covarrubias L, Lara-Carpio R, Saez-De-Ocariz M, et al. Dermoid cysts: a report of 75 pediatric patients. Pediatr Dermatol. 2013;30:706-711.
- Julapalli MR, Cohen BA, Hollier LH, et al. Congenital, ill-defined, yellowish plaque: the nasal dermoid. Pediatr Dermatol. 2006;23:556-559.
- Reissis D, Pfaff MJ, Patel A, et al. Craniofacial dermoid cysts: histological analysis and inter-site comparison. Yale J Biol Med. 2014;87:349-357.
- Chauhan DS, Guruprasad Y. Dermal cylindroma of the scalp. Natl J Maxillofac Surg. 2012;3:59-61.
- Albores-Saavedra J, Heard SC, McLaren B, et al. Cylindroma (dermal analog tumor) of the breast: a comparison with cylindroma of the skin and adenoid cystic carcinoma of the breast. Am J Clin Pathol. 2005;123:866-873.
- Myers DJ, Fillman EP. Cylindroma. StatPearls. StatPearls Publishing; 2022.
- Ramaswamy AS, Manjunatha HK, Sunilkumar B, et al. Morphological spectrum of pilar cysts. N Am J Med Sci. 2013;5:124-128. doi:10.4103/1947-2714.107532
- Al Aboud DM, Yarrarapu SNS, Patel BC. Pilar cyst. StatPearls. StatPearls Publishing; 2022. 16. Kolb L, Yarrarapu SNS, Ameer MA, et al. Lipoma. StatPearls. StatPearls Publishing; 2022.
The Diagnosis: Alopecic and Aseptic Nodule of the Scalp
Alopecic and aseptic nodule of the scalp (AANS) is an underdiagnosed condition presenting with one or few inflammatory nodules on the scalp with overlying nonscarring alopecia. The nodules can be soft, fluctuant, or firm and are characterized by negative fungal and bacterial stains as well as cultures.1 Trichoscopic features such as black or yellow dots, fine vellus hairs, and broken hairs have been reported.1-3 Dilated follicular openings may be seen and are termed the Eastern pancake sign, as they resemble the bubble cavities formed during the cooking of atayef.2 The histologic features of AANS often are nonspecific but show a nodular or pseudocystic, lymphohistiocytic to acute inflammatory component centered in the dermis.1 Granulomatous inflammation or isolated giant cells have been reported within the deep dermis.1,4 In our patient, histopathology revealed admixed acute and granulomatous inflammation within the deep dermis (Figure). Treatment of AANS includes oral antibiotics such as doxycycline, intralesional corticosteroids, or excision.1

Although the etiology of AANS currently is unclear, a process of follicular plugging or a deep folliculitis sparing the bulge stem cells has been theorized. Young males are disproportionately affected.1 It is uncertain how much overlap there is, if any, between AANS and pseudocyst of the scalp, the latter of which primarily is reported in the Japanese literature and demonstrates alopecic nodules between the forehead and vertex of the scalp with pseudocystic architecture and granulomatous infiltration on histopathology.4-7
There are several clinical and histologic differences between AANS and other diagnoses in the differential. Dermoid cysts tend to present at birth, with 70% of cases presenting before the age of 6 years, and without overlying skin changes.8 They represent a benign entrapment of ectoderm along embryonic closure lines during development.9 Histologic examination typically will show a squamous-lined cyst within the dermis with associated adnexal structures.10 Cylindromas are benign neoplasms of eccrine sweat glands named after the histologic presentation of cylinder-shaped basaloid cell populations when cross-sectioned.11,12 When cylindromas coalesce on the scalp, they form a distinctive morphology sometimes loosely resembling a turban, giving them the previously more common name turban tumors.11,13 Cylindromas appear as slow-growing protuberant tumors that are erythematous or flesh colored. Cylindromas are 9 times more common in females.13 Pilar cysts have a stratified squamous epithelium lining with a palisaded outer layer and are derived from the outer root sheath of hair follicles.14 Clinically, pilar cysts are smooth mobile cysts that favor skin with a dense concentration of hair follicles.14,15 On palpation, pilar cysts are firm due to their keratinous contents and typically are nontender unless inflamed.15 Lipomas are benign mesenchymal tumors with mature adipocytes that often appear as subcutaneous nodules without overlying skin changes, though they can involve deep fascia. On palpation, lipomas generally are soft, mobile, and nontender.16
The Diagnosis: Alopecic and Aseptic Nodule of the Scalp
Alopecic and aseptic nodule of the scalp (AANS) is an underdiagnosed condition presenting with one or few inflammatory nodules on the scalp with overlying nonscarring alopecia. The nodules can be soft, fluctuant, or firm and are characterized by negative fungal and bacterial stains as well as cultures.1 Trichoscopic features such as black or yellow dots, fine vellus hairs, and broken hairs have been reported.1-3 Dilated follicular openings may be seen and are termed the Eastern pancake sign, as they resemble the bubble cavities formed during the cooking of atayef.2 The histologic features of AANS often are nonspecific but show a nodular or pseudocystic, lymphohistiocytic to acute inflammatory component centered in the dermis.1 Granulomatous inflammation or isolated giant cells have been reported within the deep dermis.1,4 In our patient, histopathology revealed admixed acute and granulomatous inflammation within the deep dermis (Figure). Treatment of AANS includes oral antibiotics such as doxycycline, intralesional corticosteroids, or excision.1

Although the etiology of AANS currently is unclear, a process of follicular plugging or a deep folliculitis sparing the bulge stem cells has been theorized. Young males are disproportionately affected.1 It is uncertain how much overlap there is, if any, between AANS and pseudocyst of the scalp, the latter of which primarily is reported in the Japanese literature and demonstrates alopecic nodules between the forehead and vertex of the scalp with pseudocystic architecture and granulomatous infiltration on histopathology.4-7
There are several clinical and histologic differences between AANS and other diagnoses in the differential. Dermoid cysts tend to present at birth, with 70% of cases presenting before the age of 6 years, and without overlying skin changes.8 They represent a benign entrapment of ectoderm along embryonic closure lines during development.9 Histologic examination typically will show a squamous-lined cyst within the dermis with associated adnexal structures.10 Cylindromas are benign neoplasms of eccrine sweat glands named after the histologic presentation of cylinder-shaped basaloid cell populations when cross-sectioned.11,12 When cylindromas coalesce on the scalp, they form a distinctive morphology sometimes loosely resembling a turban, giving them the previously more common name turban tumors.11,13 Cylindromas appear as slow-growing protuberant tumors that are erythematous or flesh colored. Cylindromas are 9 times more common in females.13 Pilar cysts have a stratified squamous epithelium lining with a palisaded outer layer and are derived from the outer root sheath of hair follicles.14 Clinically, pilar cysts are smooth mobile cysts that favor skin with a dense concentration of hair follicles.14,15 On palpation, pilar cysts are firm due to their keratinous contents and typically are nontender unless inflamed.15 Lipomas are benign mesenchymal tumors with mature adipocytes that often appear as subcutaneous nodules without overlying skin changes, though they can involve deep fascia. On palpation, lipomas generally are soft, mobile, and nontender.16
- Bellinato F, Maurelli M, Colato C, et al. Alopecic and aseptic nodules of the scalp: a new case with a systematic review of the literature [published online May 1, 2021]. Clin Case Rep. 2021;9:E04153. doi:10.1002/ccr3.4153
- Lázaro-Simó AI, Sancho MI, Quintana-Codina M, et al. Alopecic and aseptic nodules of the scalp with trichoscopic and ultrasonographic findings. Indian J Dermatol. 2017;62:515-518.
- Garrido-Colmenero C, Arias-Santiago S, Aneiros Fernández J, et al. Trichoscopy and ultrasonography features of aseptic and alopecic nodules of the scalp. J Eur Acad Dermatol Venereol. 2016;30:507-509. doi:10.1111/jdv.12903
- Seol JE, Park IH, Kim DH, et al. Alopecic and aseptic nodules of the scalp/pseudocyst of the scalp: clinicopathological and therapeutic analyses in 11 Korean patients. Dermatology. 2016;232:165-170.
- Lee SS, Kim SY, Im M, et al. Pseudocyst of the scalp. Ann Dermatol. 2011;23(suppl 2):S267-S269.
- Eisenberg EL. Alopecia-associated pseudocyst of the scalp. J Am Acad Dermatol. 2012;67:E114-E116.
- Tsuruta D, Hayashi A, Kobayashi H, et al. Pseudocyst of the scalp. Dermatology. 2005;210:333-335.
- Orozco-Covarrubias L, Lara-Carpio R, Saez-De-Ocariz M, et al. Dermoid cysts: a report of 75 pediatric patients. Pediatr Dermatol. 2013;30:706-711.
- Julapalli MR, Cohen BA, Hollier LH, et al. Congenital, ill-defined, yellowish plaque: the nasal dermoid. Pediatr Dermatol. 2006;23:556-559.
- Reissis D, Pfaff MJ, Patel A, et al. Craniofacial dermoid cysts: histological analysis and inter-site comparison. Yale J Biol Med. 2014;87:349-357.
- Chauhan DS, Guruprasad Y. Dermal cylindroma of the scalp. Natl J Maxillofac Surg. 2012;3:59-61.
- Albores-Saavedra J, Heard SC, McLaren B, et al. Cylindroma (dermal analog tumor) of the breast: a comparison with cylindroma of the skin and adenoid cystic carcinoma of the breast. Am J Clin Pathol. 2005;123:866-873.
- Myers DJ, Fillman EP. Cylindroma. StatPearls. StatPearls Publishing; 2022.
- Ramaswamy AS, Manjunatha HK, Sunilkumar B, et al. Morphological spectrum of pilar cysts. N Am J Med Sci. 2013;5:124-128. doi:10.4103/1947-2714.107532
- Al Aboud DM, Yarrarapu SNS, Patel BC. Pilar cyst. StatPearls. StatPearls Publishing; 2022. 16. Kolb L, Yarrarapu SNS, Ameer MA, et al. Lipoma. StatPearls. StatPearls Publishing; 2022.
- Bellinato F, Maurelli M, Colato C, et al. Alopecic and aseptic nodules of the scalp: a new case with a systematic review of the literature [published online May 1, 2021]. Clin Case Rep. 2021;9:E04153. doi:10.1002/ccr3.4153
- Lázaro-Simó AI, Sancho MI, Quintana-Codina M, et al. Alopecic and aseptic nodules of the scalp with trichoscopic and ultrasonographic findings. Indian J Dermatol. 2017;62:515-518.
- Garrido-Colmenero C, Arias-Santiago S, Aneiros Fernández J, et al. Trichoscopy and ultrasonography features of aseptic and alopecic nodules of the scalp. J Eur Acad Dermatol Venereol. 2016;30:507-509. doi:10.1111/jdv.12903
- Seol JE, Park IH, Kim DH, et al. Alopecic and aseptic nodules of the scalp/pseudocyst of the scalp: clinicopathological and therapeutic analyses in 11 Korean patients. Dermatology. 2016;232:165-170.
- Lee SS, Kim SY, Im M, et al. Pseudocyst of the scalp. Ann Dermatol. 2011;23(suppl 2):S267-S269.
- Eisenberg EL. Alopecia-associated pseudocyst of the scalp. J Am Acad Dermatol. 2012;67:E114-E116.
- Tsuruta D, Hayashi A, Kobayashi H, et al. Pseudocyst of the scalp. Dermatology. 2005;210:333-335.
- Orozco-Covarrubias L, Lara-Carpio R, Saez-De-Ocariz M, et al. Dermoid cysts: a report of 75 pediatric patients. Pediatr Dermatol. 2013;30:706-711.
- Julapalli MR, Cohen BA, Hollier LH, et al. Congenital, ill-defined, yellowish plaque: the nasal dermoid. Pediatr Dermatol. 2006;23:556-559.
- Reissis D, Pfaff MJ, Patel A, et al. Craniofacial dermoid cysts: histological analysis and inter-site comparison. Yale J Biol Med. 2014;87:349-357.
- Chauhan DS, Guruprasad Y. Dermal cylindroma of the scalp. Natl J Maxillofac Surg. 2012;3:59-61.
- Albores-Saavedra J, Heard SC, McLaren B, et al. Cylindroma (dermal analog tumor) of the breast: a comparison with cylindroma of the skin and adenoid cystic carcinoma of the breast. Am J Clin Pathol. 2005;123:866-873.
- Myers DJ, Fillman EP. Cylindroma. StatPearls. StatPearls Publishing; 2022.
- Ramaswamy AS, Manjunatha HK, Sunilkumar B, et al. Morphological spectrum of pilar cysts. N Am J Med Sci. 2013;5:124-128. doi:10.4103/1947-2714.107532
- Al Aboud DM, Yarrarapu SNS, Patel BC. Pilar cyst. StatPearls. StatPearls Publishing; 2022. 16. Kolb L, Yarrarapu SNS, Ameer MA, et al. Lipoma. StatPearls. StatPearls Publishing; 2022.
A 9-year-old boy presented with a soft subcutaneous nodule with overlying alopecia on the right parietal scalp of 5 months’ duration that had grown in size, became increasingly alopecic, and was complicated by intermittent pain. An excisional biopsy of the nodule revealed deep dermal mixed inflammation with scattered granulomas. No foreign material, definitive cystic spaces, or cyst wall lining was identified. Special stains including periodic acid– Schiff, Fite acid-fast, and Twort Gram were negative for infectious organisms. His postoperative course was uneventful, and no recurrence of the nodule was reported.

Two Canadian provinces lift licensing barriers for U.S. doctors
They’ll no longer have to start with a limited license and take additional exams or be supervised for up to a year to become fully licensed.
Canada is experiencing an acute shortage of licensed physicians that’s expected to intensify over the next decade. The shortfall is estimated to be about 44,000 physicians by 2028, with family doctors accounting for 72% of the deficit.
“Reducing licensing barriers should make Canada a more attractive option for U.S. doctors who may be considering a move north,” said Tom Florence, president of AMN Healthcare’s Physician Solutions division, which recruits American physicians to work in Canada.
“Canada also has a truly expedited work visa process for qualifying physicians who have a job offer and wish to practice there,” said Mr. Florence. It usually takes about 6 months compared with at least 18 months for Canadian physicians who want to work in the United States, he said.
Few U.S.-trained physicians work in Canada, which has a population of nearly 39 million. Just 812 of them practiced in Canada in 2019, the last year data was collected, according to the Canadian Medical Association.
But Canada may attract American physicians who find U.S. medicine to be fraught with ethical dilemmas and restrictions from insurance companies and elected officials, said Theresa Rohr-Kirchgraber, MD, an internist and immediate past president of the American Medical Women’s Association.
“Rather than give up practicing medicine, a move to Canada may be a welcome respite for some U.S. physicians,” she said.
Physician recruiters in Ontario and Nova Scotia welcomed the news. About 13% of the population is without a family doctor, according to news reports.
A number of U.S. physicians have started practices in Nova Scotia in recent years, said Katrina Philopoulos, Nova Scotia Health’s director of physician recruitment. “I think this momentum will help us,” she said.
Other Canadian provinces with physician shortages are also considering making similar changes. Alberta recently announced a 5-year pilot project to waive some licensing requirements for family doctors and general practitioners trained in Australia, Ireland, United Kingdom, and the United States.
What are the pros and cons of working in Canada?
“Some U.S. physicians may be attracted by a single-payer system in which all patients have access to coverage, but there are a range of drawbacks and benefits to consider in both systems,” said Mr. Florence.
U.S. physicians generally earn more than their Canadian counterparts, so income is not likely to be a draw, he said.
That appears to be the case for both family medicine physicians and specialists when comparing average net annual salaries. (To obtain Canadian salaries, 2021 gross income data from the Canadian Institute for Health Information were used; 20% was deducted for operation costs; and Canadian dollars were converted into U.S. dollars based on the current exchange rate.)
A family medicine doctor in Canada will earn an annual average salary of $195,853 USD compared with $236,000 in the United States. A cardiologist in Canada will earn $314,051 USD annually compared with $459,000 in the United States. A dermatologist in Canada will earn $270,018 annually compared with $394,000 in the United States.
Everett Fuller, MD, an emergency medicine physician who moved from Texas to Nova Scotia in 2015 for his Canadian wife, recently wrote about the pros and cons of working there compared with the United States. For him, it was a worthwhile move.
“It’s getting back to making medicine and patient care the priority instead of the business of medicine,” Dr. Fuller wrote.
“I have the comfort of knowing that a patient and their family will not go bankrupt trying to pay medical bills if I make a catastrophic diagnosis. There’s no out-of-pocket cost, other than prescriptions (depending on their drug plan).”
Dr. Fuller also doesn’t have to fight insurers for reimbursement or preapprovals, and he pays much less for medical malpractice premiums in a less litigious environment, he said.
But he mentioned a few negatives. Some treatment is rationed, which can lead to long wait times for patients to get appointments. Also, “hospitals aren’t in it for the profit, so you’re not going to get a CT, MRI, and cath lab in every hospital,” he noted.
Mr. Florence doesn’t think either system “offers a panacea for many of the challenges physicians face today. Even with reduced barriers to licensure, we do not anticipate an exodus to U.S. physicians to the north.”
A version of this article first appeared on Medscape.com.
They’ll no longer have to start with a limited license and take additional exams or be supervised for up to a year to become fully licensed.
Canada is experiencing an acute shortage of licensed physicians that’s expected to intensify over the next decade. The shortfall is estimated to be about 44,000 physicians by 2028, with family doctors accounting for 72% of the deficit.
“Reducing licensing barriers should make Canada a more attractive option for U.S. doctors who may be considering a move north,” said Tom Florence, president of AMN Healthcare’s Physician Solutions division, which recruits American physicians to work in Canada.
“Canada also has a truly expedited work visa process for qualifying physicians who have a job offer and wish to practice there,” said Mr. Florence. It usually takes about 6 months compared with at least 18 months for Canadian physicians who want to work in the United States, he said.
Few U.S.-trained physicians work in Canada, which has a population of nearly 39 million. Just 812 of them practiced in Canada in 2019, the last year data was collected, according to the Canadian Medical Association.
But Canada may attract American physicians who find U.S. medicine to be fraught with ethical dilemmas and restrictions from insurance companies and elected officials, said Theresa Rohr-Kirchgraber, MD, an internist and immediate past president of the American Medical Women’s Association.
“Rather than give up practicing medicine, a move to Canada may be a welcome respite for some U.S. physicians,” she said.
Physician recruiters in Ontario and Nova Scotia welcomed the news. About 13% of the population is without a family doctor, according to news reports.
A number of U.S. physicians have started practices in Nova Scotia in recent years, said Katrina Philopoulos, Nova Scotia Health’s director of physician recruitment. “I think this momentum will help us,” she said.
Other Canadian provinces with physician shortages are also considering making similar changes. Alberta recently announced a 5-year pilot project to waive some licensing requirements for family doctors and general practitioners trained in Australia, Ireland, United Kingdom, and the United States.
What are the pros and cons of working in Canada?
“Some U.S. physicians may be attracted by a single-payer system in which all patients have access to coverage, but there are a range of drawbacks and benefits to consider in both systems,” said Mr. Florence.
U.S. physicians generally earn more than their Canadian counterparts, so income is not likely to be a draw, he said.
That appears to be the case for both family medicine physicians and specialists when comparing average net annual salaries. (To obtain Canadian salaries, 2021 gross income data from the Canadian Institute for Health Information were used; 20% was deducted for operation costs; and Canadian dollars were converted into U.S. dollars based on the current exchange rate.)
A family medicine doctor in Canada will earn an annual average salary of $195,853 USD compared with $236,000 in the United States. A cardiologist in Canada will earn $314,051 USD annually compared with $459,000 in the United States. A dermatologist in Canada will earn $270,018 annually compared with $394,000 in the United States.
Everett Fuller, MD, an emergency medicine physician who moved from Texas to Nova Scotia in 2015 for his Canadian wife, recently wrote about the pros and cons of working there compared with the United States. For him, it was a worthwhile move.
“It’s getting back to making medicine and patient care the priority instead of the business of medicine,” Dr. Fuller wrote.
“I have the comfort of knowing that a patient and their family will not go bankrupt trying to pay medical bills if I make a catastrophic diagnosis. There’s no out-of-pocket cost, other than prescriptions (depending on their drug plan).”
Dr. Fuller also doesn’t have to fight insurers for reimbursement or preapprovals, and he pays much less for medical malpractice premiums in a less litigious environment, he said.
But he mentioned a few negatives. Some treatment is rationed, which can lead to long wait times for patients to get appointments. Also, “hospitals aren’t in it for the profit, so you’re not going to get a CT, MRI, and cath lab in every hospital,” he noted.
Mr. Florence doesn’t think either system “offers a panacea for many of the challenges physicians face today. Even with reduced barriers to licensure, we do not anticipate an exodus to U.S. physicians to the north.”
A version of this article first appeared on Medscape.com.
They’ll no longer have to start with a limited license and take additional exams or be supervised for up to a year to become fully licensed.
Canada is experiencing an acute shortage of licensed physicians that’s expected to intensify over the next decade. The shortfall is estimated to be about 44,000 physicians by 2028, with family doctors accounting for 72% of the deficit.
“Reducing licensing barriers should make Canada a more attractive option for U.S. doctors who may be considering a move north,” said Tom Florence, president of AMN Healthcare’s Physician Solutions division, which recruits American physicians to work in Canada.
“Canada also has a truly expedited work visa process for qualifying physicians who have a job offer and wish to practice there,” said Mr. Florence. It usually takes about 6 months compared with at least 18 months for Canadian physicians who want to work in the United States, he said.
Few U.S.-trained physicians work in Canada, which has a population of nearly 39 million. Just 812 of them practiced in Canada in 2019, the last year data was collected, according to the Canadian Medical Association.
But Canada may attract American physicians who find U.S. medicine to be fraught with ethical dilemmas and restrictions from insurance companies and elected officials, said Theresa Rohr-Kirchgraber, MD, an internist and immediate past president of the American Medical Women’s Association.
“Rather than give up practicing medicine, a move to Canada may be a welcome respite for some U.S. physicians,” she said.
Physician recruiters in Ontario and Nova Scotia welcomed the news. About 13% of the population is without a family doctor, according to news reports.
A number of U.S. physicians have started practices in Nova Scotia in recent years, said Katrina Philopoulos, Nova Scotia Health’s director of physician recruitment. “I think this momentum will help us,” she said.
Other Canadian provinces with physician shortages are also considering making similar changes. Alberta recently announced a 5-year pilot project to waive some licensing requirements for family doctors and general practitioners trained in Australia, Ireland, United Kingdom, and the United States.
What are the pros and cons of working in Canada?
“Some U.S. physicians may be attracted by a single-payer system in which all patients have access to coverage, but there are a range of drawbacks and benefits to consider in both systems,” said Mr. Florence.
U.S. physicians generally earn more than their Canadian counterparts, so income is not likely to be a draw, he said.
That appears to be the case for both family medicine physicians and specialists when comparing average net annual salaries. (To obtain Canadian salaries, 2021 gross income data from the Canadian Institute for Health Information were used; 20% was deducted for operation costs; and Canadian dollars were converted into U.S. dollars based on the current exchange rate.)
A family medicine doctor in Canada will earn an annual average salary of $195,853 USD compared with $236,000 in the United States. A cardiologist in Canada will earn $314,051 USD annually compared with $459,000 in the United States. A dermatologist in Canada will earn $270,018 annually compared with $394,000 in the United States.
Everett Fuller, MD, an emergency medicine physician who moved from Texas to Nova Scotia in 2015 for his Canadian wife, recently wrote about the pros and cons of working there compared with the United States. For him, it was a worthwhile move.
“It’s getting back to making medicine and patient care the priority instead of the business of medicine,” Dr. Fuller wrote.
“I have the comfort of knowing that a patient and their family will not go bankrupt trying to pay medical bills if I make a catastrophic diagnosis. There’s no out-of-pocket cost, other than prescriptions (depending on their drug plan).”
Dr. Fuller also doesn’t have to fight insurers for reimbursement or preapprovals, and he pays much less for medical malpractice premiums in a less litigious environment, he said.
But he mentioned a few negatives. Some treatment is rationed, which can lead to long wait times for patients to get appointments. Also, “hospitals aren’t in it for the profit, so you’re not going to get a CT, MRI, and cath lab in every hospital,” he noted.
Mr. Florence doesn’t think either system “offers a panacea for many of the challenges physicians face today. Even with reduced barriers to licensure, we do not anticipate an exodus to U.S. physicians to the north.”
A version of this article first appeared on Medscape.com.
Researchers seek to understand post-COVID autoimmune disease risk
Since the COVID-19 pandemic started more than 3 years ago, the longer-lasting effects of SARS-CoV-2 infection have continued to reveal themselves. Approximately 28% of Americans report having ever experienced post-COVID conditions, such as brain fog, postexertional malaise, and joint pain, and 11% say they are still experiencing these long-term effects. Now, new research is showing that people who have had COVID are more likely to newly develop an autoimmune disease. Exactly why this is happening is less clear, experts say.
Two preprint studies and one study published in a peer-reviewed journal provide strong evidence that patients who have been infected with SARS-CoV-2 are at elevated risk of developing an autoimmune disease. The studies retrospectively reviewed medical records from three countries and compared the incidence of new-onset autoimmune disease among patients who had polymerase chain reaction–confirmed COVID-19 and those who had never been diagnosed with the virus.
A study analyzing the health records of 3.8 million U.S. patients – more than 888,460 with confirmed COVID-19 – found that the COVID-19 group was two to three times as likely to develop various autoimmune diseases, such as rheumatoid arthritis, systemic lupus erythematosus, and systemic sclerosis. A U.K. preprint study that included more than 458,000 people with confirmed COVID found that those who had previously been infected with SARS-CoV-2 were 22% more likely to develop an autoimmune disease compared with the control group. In this cohort, the diseases most strongly associated with COVID-19 were type 1 diabetes, inflammatory bowel disease, and psoriasis. A preprint study from German researchers found that COVID-19 patients were almost 43% more likely to develop an autoimmune disease, compared with those who had never been infected. COVID-19 was most strongly linked to vasculitis.
These large studies are telling us, “Yes, this link is there, so we have to accept it,” Sonia Sharma, PhD, of the Center for Autoimmunity and Inflammation at the La Jolla (Calif.) Institute for Immunology, told this news organization. But this is not the first time that autoimmune diseases have been linked to previous infections.
Researchers have known for decades that Epstein-Barr virus infection is linked to several autoimmune diseases, including systemic lupus erythematosus, multiple sclerosis, and rheumatoid arthritis. More recent research suggests the virus may activate certain genes associated with these immune disorders. Hepatitis C virus can induce cryoglobulinemia, and infection with cytomegalovirus has been implicated in several autoimmune diseases. Bacterial infections have also been linked to autoimmunity, such as group A streptococcus and rheumatic fever, as well as salmonella and reactive arthritis, to name only a few.
“In a way, this isn’t necessarily a new concept to physicians, particularly rheumatologists,” said Jeffrey A. Sparks, MD, a rheumatologist at Brigham and Women’s Hospital in Boston. “There’s a fine line between appropriately clearing an infection and the body overreacting and setting off a cascade where the immune system is chronically overactive that can manifest as an autoimmune disease,” he told this news organization.
A dysregulated response to infection
It takes the immune system a week or two to develop antigen-specific antibodies to a new pathogen. But for patients with serious infections – in this instance, COVID-19 – that’s time they don’t have. Therefore, the immune system has an alternative pathway, called extrafollicular activation, that creates fast-acting antibodies, explained Matthew Woodruff, PhD, an instructor of immunology and rheumatology at Emory University, Atlanta.
The trade-off is that these antibodies are not as specific and can target the body’s own tissues. This dysregulation of antibody selection is generally short lived and fades when more targeted antibodies are produced and take over, but in some cases, this process can lead to high levels of self-targeting antibodies that can harm the body’s organs and tissues. Research also suggests that for patients who experience long COVID, the same autoantibodies that drive the initial immune response are detectable in the body months after infection, though it is not known whether these lingering immune cells cause these longer-lasting symptoms.
“If you have a virus that causes hyperinflammation plus organ damage, that is a recipe for disaster,” Dr. Sharma said. “It’s a recipe for autoantibodies and autoreactive T cells that down the road can attack the body’s own tissues, especially in people whose immune system is trained in such a way to cause self-reactivity,” she added.
This hyperinflammation can result in rare but serious complications, such as multisystem inflammatory syndrome in children and adults, which can occur 2-6 weeks after SARS-CoV-2 infection. But even in these patients with severe illness, organ-specific complications tend to resolve in 6 months with “no significant sequelae 1 year after diagnosis,” according to the Centers for Disease Control and Prevention. And while long COVID can last for a year or longer, data suggest that symptoms do eventually resolve for most people. What is not clear is why acute autoimmunity triggered by COVID-19 can become a chronic condition in certain patients.
Predisposition to autoimmunity
P. J. Utz, MD, PhD, professor of immunology and rheumatology at Stanford (Calif.) University, said that people who develop autoimmune disease after SARS-CoV-2 infection may have already been predisposed toward autoimmunity. Especially for autoimmune diseases such as type 1 diabetes and lupus, autoantibodies can appear and circulate in the body for more than a decade in some people before they present with any clinical symptoms. “Their immune system is primed such that if they get infected with something – or they have some other environmental trigger that maybe we don’t know about yet – that is enough to then push them over the edge so that they get full-blown autoimmunity,” he said. What is not known is whether these patients’ conditions would have advanced to true clinical disease had they not been infected, he said.
He also noted that the presence of autoantibodies does not necessarily mean someone has autoimmune disease; healthy people can also have autoantibodies, and everyone develops them with age. “My advice would be, ‘Don’t lose sleep over this,’ “ he said.
Dr. Sparks agreed that while these retrospective studies did show an elevated risk of autoimmune disease after COVID-19, that risk appears to be relatively small. “As a practicing rheumatologist, we aren’t seeing a stampede of patients with new-onset rheumatic diseases,” he said. “It’s not like we’re overwhelmed with autoimmune patients, even though almost everyone’s had COVID. So, if there is a risk, it’s very modest.”
Dr. Sparks is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Utz receives research funding from Pfizer. Dr. Sharma and Dr. Woodruff have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Since the COVID-19 pandemic started more than 3 years ago, the longer-lasting effects of SARS-CoV-2 infection have continued to reveal themselves. Approximately 28% of Americans report having ever experienced post-COVID conditions, such as brain fog, postexertional malaise, and joint pain, and 11% say they are still experiencing these long-term effects. Now, new research is showing that people who have had COVID are more likely to newly develop an autoimmune disease. Exactly why this is happening is less clear, experts say.
Two preprint studies and one study published in a peer-reviewed journal provide strong evidence that patients who have been infected with SARS-CoV-2 are at elevated risk of developing an autoimmune disease. The studies retrospectively reviewed medical records from three countries and compared the incidence of new-onset autoimmune disease among patients who had polymerase chain reaction–confirmed COVID-19 and those who had never been diagnosed with the virus.
A study analyzing the health records of 3.8 million U.S. patients – more than 888,460 with confirmed COVID-19 – found that the COVID-19 group was two to three times as likely to develop various autoimmune diseases, such as rheumatoid arthritis, systemic lupus erythematosus, and systemic sclerosis. A U.K. preprint study that included more than 458,000 people with confirmed COVID found that those who had previously been infected with SARS-CoV-2 were 22% more likely to develop an autoimmune disease compared with the control group. In this cohort, the diseases most strongly associated with COVID-19 were type 1 diabetes, inflammatory bowel disease, and psoriasis. A preprint study from German researchers found that COVID-19 patients were almost 43% more likely to develop an autoimmune disease, compared with those who had never been infected. COVID-19 was most strongly linked to vasculitis.
These large studies are telling us, “Yes, this link is there, so we have to accept it,” Sonia Sharma, PhD, of the Center for Autoimmunity and Inflammation at the La Jolla (Calif.) Institute for Immunology, told this news organization. But this is not the first time that autoimmune diseases have been linked to previous infections.
Researchers have known for decades that Epstein-Barr virus infection is linked to several autoimmune diseases, including systemic lupus erythematosus, multiple sclerosis, and rheumatoid arthritis. More recent research suggests the virus may activate certain genes associated with these immune disorders. Hepatitis C virus can induce cryoglobulinemia, and infection with cytomegalovirus has been implicated in several autoimmune diseases. Bacterial infections have also been linked to autoimmunity, such as group A streptococcus and rheumatic fever, as well as salmonella and reactive arthritis, to name only a few.
“In a way, this isn’t necessarily a new concept to physicians, particularly rheumatologists,” said Jeffrey A. Sparks, MD, a rheumatologist at Brigham and Women’s Hospital in Boston. “There’s a fine line between appropriately clearing an infection and the body overreacting and setting off a cascade where the immune system is chronically overactive that can manifest as an autoimmune disease,” he told this news organization.
A dysregulated response to infection
It takes the immune system a week or two to develop antigen-specific antibodies to a new pathogen. But for patients with serious infections – in this instance, COVID-19 – that’s time they don’t have. Therefore, the immune system has an alternative pathway, called extrafollicular activation, that creates fast-acting antibodies, explained Matthew Woodruff, PhD, an instructor of immunology and rheumatology at Emory University, Atlanta.
The trade-off is that these antibodies are not as specific and can target the body’s own tissues. This dysregulation of antibody selection is generally short lived and fades when more targeted antibodies are produced and take over, but in some cases, this process can lead to high levels of self-targeting antibodies that can harm the body’s organs and tissues. Research also suggests that for patients who experience long COVID, the same autoantibodies that drive the initial immune response are detectable in the body months after infection, though it is not known whether these lingering immune cells cause these longer-lasting symptoms.
“If you have a virus that causes hyperinflammation plus organ damage, that is a recipe for disaster,” Dr. Sharma said. “It’s a recipe for autoantibodies and autoreactive T cells that down the road can attack the body’s own tissues, especially in people whose immune system is trained in such a way to cause self-reactivity,” she added.
This hyperinflammation can result in rare but serious complications, such as multisystem inflammatory syndrome in children and adults, which can occur 2-6 weeks after SARS-CoV-2 infection. But even in these patients with severe illness, organ-specific complications tend to resolve in 6 months with “no significant sequelae 1 year after diagnosis,” according to the Centers for Disease Control and Prevention. And while long COVID can last for a year or longer, data suggest that symptoms do eventually resolve for most people. What is not clear is why acute autoimmunity triggered by COVID-19 can become a chronic condition in certain patients.
Predisposition to autoimmunity
P. J. Utz, MD, PhD, professor of immunology and rheumatology at Stanford (Calif.) University, said that people who develop autoimmune disease after SARS-CoV-2 infection may have already been predisposed toward autoimmunity. Especially for autoimmune diseases such as type 1 diabetes and lupus, autoantibodies can appear and circulate in the body for more than a decade in some people before they present with any clinical symptoms. “Their immune system is primed such that if they get infected with something – or they have some other environmental trigger that maybe we don’t know about yet – that is enough to then push them over the edge so that they get full-blown autoimmunity,” he said. What is not known is whether these patients’ conditions would have advanced to true clinical disease had they not been infected, he said.
He also noted that the presence of autoantibodies does not necessarily mean someone has autoimmune disease; healthy people can also have autoantibodies, and everyone develops them with age. “My advice would be, ‘Don’t lose sleep over this,’ “ he said.
Dr. Sparks agreed that while these retrospective studies did show an elevated risk of autoimmune disease after COVID-19, that risk appears to be relatively small. “As a practicing rheumatologist, we aren’t seeing a stampede of patients with new-onset rheumatic diseases,” he said. “It’s not like we’re overwhelmed with autoimmune patients, even though almost everyone’s had COVID. So, if there is a risk, it’s very modest.”
Dr. Sparks is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Utz receives research funding from Pfizer. Dr. Sharma and Dr. Woodruff have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Since the COVID-19 pandemic started more than 3 years ago, the longer-lasting effects of SARS-CoV-2 infection have continued to reveal themselves. Approximately 28% of Americans report having ever experienced post-COVID conditions, such as brain fog, postexertional malaise, and joint pain, and 11% say they are still experiencing these long-term effects. Now, new research is showing that people who have had COVID are more likely to newly develop an autoimmune disease. Exactly why this is happening is less clear, experts say.
Two preprint studies and one study published in a peer-reviewed journal provide strong evidence that patients who have been infected with SARS-CoV-2 are at elevated risk of developing an autoimmune disease. The studies retrospectively reviewed medical records from three countries and compared the incidence of new-onset autoimmune disease among patients who had polymerase chain reaction–confirmed COVID-19 and those who had never been diagnosed with the virus.
A study analyzing the health records of 3.8 million U.S. patients – more than 888,460 with confirmed COVID-19 – found that the COVID-19 group was two to three times as likely to develop various autoimmune diseases, such as rheumatoid arthritis, systemic lupus erythematosus, and systemic sclerosis. A U.K. preprint study that included more than 458,000 people with confirmed COVID found that those who had previously been infected with SARS-CoV-2 were 22% more likely to develop an autoimmune disease compared with the control group. In this cohort, the diseases most strongly associated with COVID-19 were type 1 diabetes, inflammatory bowel disease, and psoriasis. A preprint study from German researchers found that COVID-19 patients were almost 43% more likely to develop an autoimmune disease, compared with those who had never been infected. COVID-19 was most strongly linked to vasculitis.
These large studies are telling us, “Yes, this link is there, so we have to accept it,” Sonia Sharma, PhD, of the Center for Autoimmunity and Inflammation at the La Jolla (Calif.) Institute for Immunology, told this news organization. But this is not the first time that autoimmune diseases have been linked to previous infections.
Researchers have known for decades that Epstein-Barr virus infection is linked to several autoimmune diseases, including systemic lupus erythematosus, multiple sclerosis, and rheumatoid arthritis. More recent research suggests the virus may activate certain genes associated with these immune disorders. Hepatitis C virus can induce cryoglobulinemia, and infection with cytomegalovirus has been implicated in several autoimmune diseases. Bacterial infections have also been linked to autoimmunity, such as group A streptococcus and rheumatic fever, as well as salmonella and reactive arthritis, to name only a few.
“In a way, this isn’t necessarily a new concept to physicians, particularly rheumatologists,” said Jeffrey A. Sparks, MD, a rheumatologist at Brigham and Women’s Hospital in Boston. “There’s a fine line between appropriately clearing an infection and the body overreacting and setting off a cascade where the immune system is chronically overactive that can manifest as an autoimmune disease,” he told this news organization.
A dysregulated response to infection
It takes the immune system a week or two to develop antigen-specific antibodies to a new pathogen. But for patients with serious infections – in this instance, COVID-19 – that’s time they don’t have. Therefore, the immune system has an alternative pathway, called extrafollicular activation, that creates fast-acting antibodies, explained Matthew Woodruff, PhD, an instructor of immunology and rheumatology at Emory University, Atlanta.
The trade-off is that these antibodies are not as specific and can target the body’s own tissues. This dysregulation of antibody selection is generally short lived and fades when more targeted antibodies are produced and take over, but in some cases, this process can lead to high levels of self-targeting antibodies that can harm the body’s organs and tissues. Research also suggests that for patients who experience long COVID, the same autoantibodies that drive the initial immune response are detectable in the body months after infection, though it is not known whether these lingering immune cells cause these longer-lasting symptoms.
“If you have a virus that causes hyperinflammation plus organ damage, that is a recipe for disaster,” Dr. Sharma said. “It’s a recipe for autoantibodies and autoreactive T cells that down the road can attack the body’s own tissues, especially in people whose immune system is trained in such a way to cause self-reactivity,” she added.
This hyperinflammation can result in rare but serious complications, such as multisystem inflammatory syndrome in children and adults, which can occur 2-6 weeks after SARS-CoV-2 infection. But even in these patients with severe illness, organ-specific complications tend to resolve in 6 months with “no significant sequelae 1 year after diagnosis,” according to the Centers for Disease Control and Prevention. And while long COVID can last for a year or longer, data suggest that symptoms do eventually resolve for most people. What is not clear is why acute autoimmunity triggered by COVID-19 can become a chronic condition in certain patients.
Predisposition to autoimmunity
P. J. Utz, MD, PhD, professor of immunology and rheumatology at Stanford (Calif.) University, said that people who develop autoimmune disease after SARS-CoV-2 infection may have already been predisposed toward autoimmunity. Especially for autoimmune diseases such as type 1 diabetes and lupus, autoantibodies can appear and circulate in the body for more than a decade in some people before they present with any clinical symptoms. “Their immune system is primed such that if they get infected with something – or they have some other environmental trigger that maybe we don’t know about yet – that is enough to then push them over the edge so that they get full-blown autoimmunity,” he said. What is not known is whether these patients’ conditions would have advanced to true clinical disease had they not been infected, he said.
He also noted that the presence of autoantibodies does not necessarily mean someone has autoimmune disease; healthy people can also have autoantibodies, and everyone develops them with age. “My advice would be, ‘Don’t lose sleep over this,’ “ he said.
Dr. Sparks agreed that while these retrospective studies did show an elevated risk of autoimmune disease after COVID-19, that risk appears to be relatively small. “As a practicing rheumatologist, we aren’t seeing a stampede of patients with new-onset rheumatic diseases,” he said. “It’s not like we’re overwhelmed with autoimmune patients, even though almost everyone’s had COVID. So, if there is a risk, it’s very modest.”
Dr. Sparks is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Utz receives research funding from Pfizer. Dr. Sharma and Dr. Woodruff have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FMT in a pill: FDA approves second product to prevent C. diff recurrence
The recent approval of the first oral fecal-derived microbiota therapy to prevent the recurrence of Clostridioides difficile (C. diff) infection in patients was welcome news for physicians who’ve struggled under the weight of having too few treatment options for the prevention of C. diff recurrence.
The product, developed by Massachusetts-based Seres Therepeutics and marketed as Vowst, was approved by the U.S. Food and Drug Administration on April 26. It is approved for use in adults who have already been treated with antibiotics for a recurrent infection with C. diff bacteria.
and is designed to be delivered in four capsules taken daily for 3 days.
Gastroenterologist Phillip I. Tarr, MD, division chief of gastroenterology at Washington University, St. Louis, and chair of the American Gastroenterological Association Center for Gut Microbiome Research and Education, said that prevention of recurrent C. diff infection “remains challenging,” and that Vowst “provides the first FDA-approved, orally administered microbiome therapeutic with which to achieve this goal. This advance also makes us optimistic we might soon be able to prevent other disorders by managing gut microbial communities.”
Vowst is the second therapy derived from human stool to be approved for the indication in less than 6 months. In December, the FDA approved Rebyota (Ferring), a rectally delivered treatment that also uses microbes from donor feces. Both products were given priority review, orphan drug, and breakthrough therapy designations by the agency.
C. diff infection can be aggravated by an alteration of normal gut flora associated with antibiotics treatment, leading to cycles of repeated infections. Infection can produce diarrhea, abdominal pain, fever, and severe morbidity. In the United States, an estimated 15,000 to 30,000 deaths per year are linked to C. diff. Risk factors for recurrent infection include being 65 or older, hospitalization, being in a nursing home, a weakened immune system, and previous infection with C. diff.
Therapies transplanting fecal microbiota from donors have been used since the 1950s as treatments for recurrent C. diff infection, and in the past decade, as stool banks recruiting screened donors have made fecal microbiota transplants, or FMT, standard of care. However, only in recent years have fecal-derived therapies become subject to standardized safety and efficacy testing.
Both the current FDA-approved products, Rebyota and Vowst, were shown in randomized controlled trials to reduce recurrence of C. diff infection, compared with placebo. In a phase 3 clinical trial of Rebyota (n = 262) in antibiotic-treated patients, one rectally administered dose reduced recurrence of C. diff infection by 70.6% at 8 weeks, compared with 57.5% for placebo. A phase 3 study of Vowst (n = 281) showed recurrence in treated subjects to be 12.4% at 8 weeks, compared with nearly 40% of those receiving placebo (relative risk, 0.32; 95% confidence interval, 0.18-0.58; P less than .001).
Despite screening protocols that have become increasingly homogenized and rigorous, FMT is associated with the risk of introducing pathogens. Vowst is manufactured with purified bacterial spores derived from donor feces, not whole stool. Nonetheless, FDA noted in its statement that Vowst could still potentially introduce infectious agents or allergens.
Antibiotics are still first-line treatment
In an interview, Jessica Allegretti, MD, MPH, AGAF, medical director of the Crohn’s and Colitis Center at Brigham & Women’s Hospital, Boston, said that having two FDA-approved therapies with different means of administration “is great for the field and great for patients. These are both meant to be used after a course of antibiotics, so antibiotics are still the mainstay of treatment for C. diff and recurrent C. diff, but we now have more options to prevent recurrence.”
The convenience of an oral therapy that can be taken at home is “very attractive,” Dr. Allegretti added, noting that there will also be patients “who either don’t want to or can’t take capsules, for whom a rectal administration [in a health care setting] may be preferred.”
Dr. Allegretti, who has used FMT to treat recurrent C. difficile for more than a decade, said that she expected traditional FMT using screened donor stool to remain available even as the new products are adopted by clinicians. FMT centers like OpenBiome “will continue to provide access for patients who either don’t have the ability to get the FDA-approved products because of insurance coverage, or for financial reasons, or maybe neither of the new products is appropriate for them,” she said. “I do think there will always be a need for the traditional option. The more options that we have available the better.”
TD Cowen analyst Joseph Thome told Reuters that the drug could be priced close to $20,000 per course, expecting peak sales of $750 million in the U.S. in 2033.
Dr. Allegretti disclosed consulting work for Seres Therapeutics, Ferring, and other manufacturers. She is a member of OpenBiome’s clinical advisory board.
The recent approval of the first oral fecal-derived microbiota therapy to prevent the recurrence of Clostridioides difficile (C. diff) infection in patients was welcome news for physicians who’ve struggled under the weight of having too few treatment options for the prevention of C. diff recurrence.
The product, developed by Massachusetts-based Seres Therepeutics and marketed as Vowst, was approved by the U.S. Food and Drug Administration on April 26. It is approved for use in adults who have already been treated with antibiotics for a recurrent infection with C. diff bacteria.
and is designed to be delivered in four capsules taken daily for 3 days.
Gastroenterologist Phillip I. Tarr, MD, division chief of gastroenterology at Washington University, St. Louis, and chair of the American Gastroenterological Association Center for Gut Microbiome Research and Education, said that prevention of recurrent C. diff infection “remains challenging,” and that Vowst “provides the first FDA-approved, orally administered microbiome therapeutic with which to achieve this goal. This advance also makes us optimistic we might soon be able to prevent other disorders by managing gut microbial communities.”
Vowst is the second therapy derived from human stool to be approved for the indication in less than 6 months. In December, the FDA approved Rebyota (Ferring), a rectally delivered treatment that also uses microbes from donor feces. Both products were given priority review, orphan drug, and breakthrough therapy designations by the agency.
C. diff infection can be aggravated by an alteration of normal gut flora associated with antibiotics treatment, leading to cycles of repeated infections. Infection can produce diarrhea, abdominal pain, fever, and severe morbidity. In the United States, an estimated 15,000 to 30,000 deaths per year are linked to C. diff. Risk factors for recurrent infection include being 65 or older, hospitalization, being in a nursing home, a weakened immune system, and previous infection with C. diff.
Therapies transplanting fecal microbiota from donors have been used since the 1950s as treatments for recurrent C. diff infection, and in the past decade, as stool banks recruiting screened donors have made fecal microbiota transplants, or FMT, standard of care. However, only in recent years have fecal-derived therapies become subject to standardized safety and efficacy testing.
Both the current FDA-approved products, Rebyota and Vowst, were shown in randomized controlled trials to reduce recurrence of C. diff infection, compared with placebo. In a phase 3 clinical trial of Rebyota (n = 262) in antibiotic-treated patients, one rectally administered dose reduced recurrence of C. diff infection by 70.6% at 8 weeks, compared with 57.5% for placebo. A phase 3 study of Vowst (n = 281) showed recurrence in treated subjects to be 12.4% at 8 weeks, compared with nearly 40% of those receiving placebo (relative risk, 0.32; 95% confidence interval, 0.18-0.58; P less than .001).
Despite screening protocols that have become increasingly homogenized and rigorous, FMT is associated with the risk of introducing pathogens. Vowst is manufactured with purified bacterial spores derived from donor feces, not whole stool. Nonetheless, FDA noted in its statement that Vowst could still potentially introduce infectious agents or allergens.
Antibiotics are still first-line treatment
In an interview, Jessica Allegretti, MD, MPH, AGAF, medical director of the Crohn’s and Colitis Center at Brigham & Women’s Hospital, Boston, said that having two FDA-approved therapies with different means of administration “is great for the field and great for patients. These are both meant to be used after a course of antibiotics, so antibiotics are still the mainstay of treatment for C. diff and recurrent C. diff, but we now have more options to prevent recurrence.”
The convenience of an oral therapy that can be taken at home is “very attractive,” Dr. Allegretti added, noting that there will also be patients “who either don’t want to or can’t take capsules, for whom a rectal administration [in a health care setting] may be preferred.”
Dr. Allegretti, who has used FMT to treat recurrent C. difficile for more than a decade, said that she expected traditional FMT using screened donor stool to remain available even as the new products are adopted by clinicians. FMT centers like OpenBiome “will continue to provide access for patients who either don’t have the ability to get the FDA-approved products because of insurance coverage, or for financial reasons, or maybe neither of the new products is appropriate for them,” she said. “I do think there will always be a need for the traditional option. The more options that we have available the better.”
TD Cowen analyst Joseph Thome told Reuters that the drug could be priced close to $20,000 per course, expecting peak sales of $750 million in the U.S. in 2033.
Dr. Allegretti disclosed consulting work for Seres Therapeutics, Ferring, and other manufacturers. She is a member of OpenBiome’s clinical advisory board.
The recent approval of the first oral fecal-derived microbiota therapy to prevent the recurrence of Clostridioides difficile (C. diff) infection in patients was welcome news for physicians who’ve struggled under the weight of having too few treatment options for the prevention of C. diff recurrence.
The product, developed by Massachusetts-based Seres Therepeutics and marketed as Vowst, was approved by the U.S. Food and Drug Administration on April 26. It is approved for use in adults who have already been treated with antibiotics for a recurrent infection with C. diff bacteria.
and is designed to be delivered in four capsules taken daily for 3 days.
Gastroenterologist Phillip I. Tarr, MD, division chief of gastroenterology at Washington University, St. Louis, and chair of the American Gastroenterological Association Center for Gut Microbiome Research and Education, said that prevention of recurrent C. diff infection “remains challenging,” and that Vowst “provides the first FDA-approved, orally administered microbiome therapeutic with which to achieve this goal. This advance also makes us optimistic we might soon be able to prevent other disorders by managing gut microbial communities.”
Vowst is the second therapy derived from human stool to be approved for the indication in less than 6 months. In December, the FDA approved Rebyota (Ferring), a rectally delivered treatment that also uses microbes from donor feces. Both products were given priority review, orphan drug, and breakthrough therapy designations by the agency.
C. diff infection can be aggravated by an alteration of normal gut flora associated with antibiotics treatment, leading to cycles of repeated infections. Infection can produce diarrhea, abdominal pain, fever, and severe morbidity. In the United States, an estimated 15,000 to 30,000 deaths per year are linked to C. diff. Risk factors for recurrent infection include being 65 or older, hospitalization, being in a nursing home, a weakened immune system, and previous infection with C. diff.
Therapies transplanting fecal microbiota from donors have been used since the 1950s as treatments for recurrent C. diff infection, and in the past decade, as stool banks recruiting screened donors have made fecal microbiota transplants, or FMT, standard of care. However, only in recent years have fecal-derived therapies become subject to standardized safety and efficacy testing.
Both the current FDA-approved products, Rebyota and Vowst, were shown in randomized controlled trials to reduce recurrence of C. diff infection, compared with placebo. In a phase 3 clinical trial of Rebyota (n = 262) in antibiotic-treated patients, one rectally administered dose reduced recurrence of C. diff infection by 70.6% at 8 weeks, compared with 57.5% for placebo. A phase 3 study of Vowst (n = 281) showed recurrence in treated subjects to be 12.4% at 8 weeks, compared with nearly 40% of those receiving placebo (relative risk, 0.32; 95% confidence interval, 0.18-0.58; P less than .001).
Despite screening protocols that have become increasingly homogenized and rigorous, FMT is associated with the risk of introducing pathogens. Vowst is manufactured with purified bacterial spores derived from donor feces, not whole stool. Nonetheless, FDA noted in its statement that Vowst could still potentially introduce infectious agents or allergens.
Antibiotics are still first-line treatment
In an interview, Jessica Allegretti, MD, MPH, AGAF, medical director of the Crohn’s and Colitis Center at Brigham & Women’s Hospital, Boston, said that having two FDA-approved therapies with different means of administration “is great for the field and great for patients. These are both meant to be used after a course of antibiotics, so antibiotics are still the mainstay of treatment for C. diff and recurrent C. diff, but we now have more options to prevent recurrence.”
The convenience of an oral therapy that can be taken at home is “very attractive,” Dr. Allegretti added, noting that there will also be patients “who either don’t want to or can’t take capsules, for whom a rectal administration [in a health care setting] may be preferred.”
Dr. Allegretti, who has used FMT to treat recurrent C. difficile for more than a decade, said that she expected traditional FMT using screened donor stool to remain available even as the new products are adopted by clinicians. FMT centers like OpenBiome “will continue to provide access for patients who either don’t have the ability to get the FDA-approved products because of insurance coverage, or for financial reasons, or maybe neither of the new products is appropriate for them,” she said. “I do think there will always be a need for the traditional option. The more options that we have available the better.”
TD Cowen analyst Joseph Thome told Reuters that the drug could be priced close to $20,000 per course, expecting peak sales of $750 million in the U.S. in 2033.
Dr. Allegretti disclosed consulting work for Seres Therapeutics, Ferring, and other manufacturers. She is a member of OpenBiome’s clinical advisory board.
FDA approves injectable treatment for cheek lines, wrinkles
The treatment, marketed as Sculptra, is the first FDA-approved PLLA collagen stimulator that, “when injected into the cheek area, helps stimulate natural collagen production to smooth wrinkles and improve skin quality such as firmness and glow,” according to a press release from the manufacturer, Galderma. Sculptra was first approved for aesthetic use in 2009 in the United States and is now available in more than 40 countries.
With this expanded approval, PLLA-SCA is now indicated for use in people with healthy immune systems for correcting shallow to deep nasolabial fold contour deficiencies, fine lines, and wrinkles in the cheeks and other facial areas.
94% have enduring improvement at 2 years
In a clinical trial, PLLA-SCA achieved the primary efficacy endpoint of at least a 1-grade improvement in wrinkles on both cheeks concurrently at rest and its secondary endpoint of improving cheek wrinkles when smiling for up to 2 years, the company states.
According to Galderma, patients showed aesthetic improvement in cheek wrinkles throughout the study; 96% showed improvement at 3 months, 94% showed improvement at 1 year, and 94% showed improvement at 2 years.
The most common side effects after initial treatment are injection site swelling, tenderness, redness, pain, bruising, bleeding, itching, and lumps, according to the company. Other side effects may include small lumps under the skin that are sometimes noticeable when pressing on the treated area.
PLLA-SCA is available only through a licensed practitioner and should not be used by people allergic to any ingredient of the product or who have a history of keloid formation or hypertrophic scarring. The company notes that safety has not been established in patients who are pregnant, lactating, breastfeeding, or younger than 18.
In its instruction to clinicians, the company warns the treatment should not be injected into the blood vessels “as it may cause vascular occlusion, infarction, or embolic phenomena.”
Skin sores, cysts, pimples, rashes, hives, or infection should be healed completely before injecting the treatment, the company cautions. PLLA-SCA should not be injected into the red area of the lip or in the periorbital area.
A version of this article first appeared on Medscape.com.
The treatment, marketed as Sculptra, is the first FDA-approved PLLA collagen stimulator that, “when injected into the cheek area, helps stimulate natural collagen production to smooth wrinkles and improve skin quality such as firmness and glow,” according to a press release from the manufacturer, Galderma. Sculptra was first approved for aesthetic use in 2009 in the United States and is now available in more than 40 countries.
With this expanded approval, PLLA-SCA is now indicated for use in people with healthy immune systems for correcting shallow to deep nasolabial fold contour deficiencies, fine lines, and wrinkles in the cheeks and other facial areas.
94% have enduring improvement at 2 years
In a clinical trial, PLLA-SCA achieved the primary efficacy endpoint of at least a 1-grade improvement in wrinkles on both cheeks concurrently at rest and its secondary endpoint of improving cheek wrinkles when smiling for up to 2 years, the company states.
According to Galderma, patients showed aesthetic improvement in cheek wrinkles throughout the study; 96% showed improvement at 3 months, 94% showed improvement at 1 year, and 94% showed improvement at 2 years.
The most common side effects after initial treatment are injection site swelling, tenderness, redness, pain, bruising, bleeding, itching, and lumps, according to the company. Other side effects may include small lumps under the skin that are sometimes noticeable when pressing on the treated area.
PLLA-SCA is available only through a licensed practitioner and should not be used by people allergic to any ingredient of the product or who have a history of keloid formation or hypertrophic scarring. The company notes that safety has not been established in patients who are pregnant, lactating, breastfeeding, or younger than 18.
In its instruction to clinicians, the company warns the treatment should not be injected into the blood vessels “as it may cause vascular occlusion, infarction, or embolic phenomena.”
Skin sores, cysts, pimples, rashes, hives, or infection should be healed completely before injecting the treatment, the company cautions. PLLA-SCA should not be injected into the red area of the lip or in the periorbital area.
A version of this article first appeared on Medscape.com.
The treatment, marketed as Sculptra, is the first FDA-approved PLLA collagen stimulator that, “when injected into the cheek area, helps stimulate natural collagen production to smooth wrinkles and improve skin quality such as firmness and glow,” according to a press release from the manufacturer, Galderma. Sculptra was first approved for aesthetic use in 2009 in the United States and is now available in more than 40 countries.
With this expanded approval, PLLA-SCA is now indicated for use in people with healthy immune systems for correcting shallow to deep nasolabial fold contour deficiencies, fine lines, and wrinkles in the cheeks and other facial areas.
94% have enduring improvement at 2 years
In a clinical trial, PLLA-SCA achieved the primary efficacy endpoint of at least a 1-grade improvement in wrinkles on both cheeks concurrently at rest and its secondary endpoint of improving cheek wrinkles when smiling for up to 2 years, the company states.
According to Galderma, patients showed aesthetic improvement in cheek wrinkles throughout the study; 96% showed improvement at 3 months, 94% showed improvement at 1 year, and 94% showed improvement at 2 years.
The most common side effects after initial treatment are injection site swelling, tenderness, redness, pain, bruising, bleeding, itching, and lumps, according to the company. Other side effects may include small lumps under the skin that are sometimes noticeable when pressing on the treated area.
PLLA-SCA is available only through a licensed practitioner and should not be used by people allergic to any ingredient of the product or who have a history of keloid formation or hypertrophic scarring. The company notes that safety has not been established in patients who are pregnant, lactating, breastfeeding, or younger than 18.
In its instruction to clinicians, the company warns the treatment should not be injected into the blood vessels “as it may cause vascular occlusion, infarction, or embolic phenomena.”
Skin sores, cysts, pimples, rashes, hives, or infection should be healed completely before injecting the treatment, the company cautions. PLLA-SCA should not be injected into the red area of the lip or in the periorbital area.
A version of this article first appeared on Medscape.com.
Erythema Ab Igne: A Clinical Review
Erythema ab igne (EAI)(also known as toasted skin syndrome) was first described in the British Journal of Dermatology in the 20th century, 1 though it was known by physicians long before. Reticular netlike skin changes were seen in association with patients who spent extended time directly next to a heat source. This association led to the name of this condition, which literally means “redness by fire.” Indeed, EAI induced by chronic heat exposure has been described across the world for centuries. For example, in the cold regions of northern China, people used to sleep on beds of hot bricks called kang to stay warm at night. The people of India’s Kashmir district carried pots of hot coals called kangri next to the skin under large woven shawls to stay warm. In the past, Irish women often spent much time by a turf- or peat-burning fire. Chronic heat exposure in these cases can lead not only to EAI but also to aggressive types of cancer, often with a latency of 30 years or more. 2
More recently, the invention of home central heating led to a stark decrease in the number of cases associated with combustion-based heat, with a transition to etiologies such as use of hot water bottles, electric blankets, and electric space heaters. Over time, technological advances led to ever-increasing potential causes for EAI, such as laptops or cell phones, car heaters and heated seats, heated blankets,3,4 infrared lamps for food, and even medical devices such as ultrasound-based heating products and convective temperature management systems for hospitalized patients. As technology evolves, so do the potential causes of EAI, requiring clinicians to diagnose and deduce the cause through a thorough social and medical history as well as a workup on the present illness with considerations for the anatomical location.5-7 Herein, we describe the etiology of EAI, diagnosis, and treatment options.
Clinical Characteristics
Erythema ab igne begins as mild, transient, and erythematous macules and patches in a reticular pattern that resolve minutes to hours after removal of the heat source. With weeks to months of continued or repeated application of the heat source, the affected area eventually becomes hyperpigmented where there once was erythema (Figures 1 and 2). Sometimes papules, bullae, telangiectasia, and hyperkeratosis also form. The rash usually is asymptomatic, though pain, pruritus, and dysesthesia have been reported.7 Dermoscopy of EAI in the hyperpigmented stage can reveal diffuse superficial dark pigmentation, telangiectasia, and mild whitish scaling.8 Although the pathogenesis has remained elusive over the years, lesions do seem to be mostly associated with cumulative exposure to heat rather than length of exposure.7
Etiology of EAI
Anatomic Location—The affected site depends on the source of heat (Table). Classic examples of this condition include a patient with EAI presenting on the anterior thighs after working in front of a hot oven or a patient with chronic back pain presenting with lower-back EAI secondary to frequent use of a hot water bottle or heating pad.7 With evolving technology over the last few decades, new etiologies have become more common—teenagers are presenting with anterior thigh EAI secondary to frequent laptop use2-29; patients are holding warm cell phones in their pant pockets, leading to unilateral geometric EAI on the anterior thigh (front pocket) or buttock (back pocket)30; plug-in radiators under computer desks are causing EAI on the lower legs31-34; and automobile seat heaters have been shown to cause EAI on the posterior legs.5,35-37 Clinicians should consider anatomic location a critical clue for etiology.
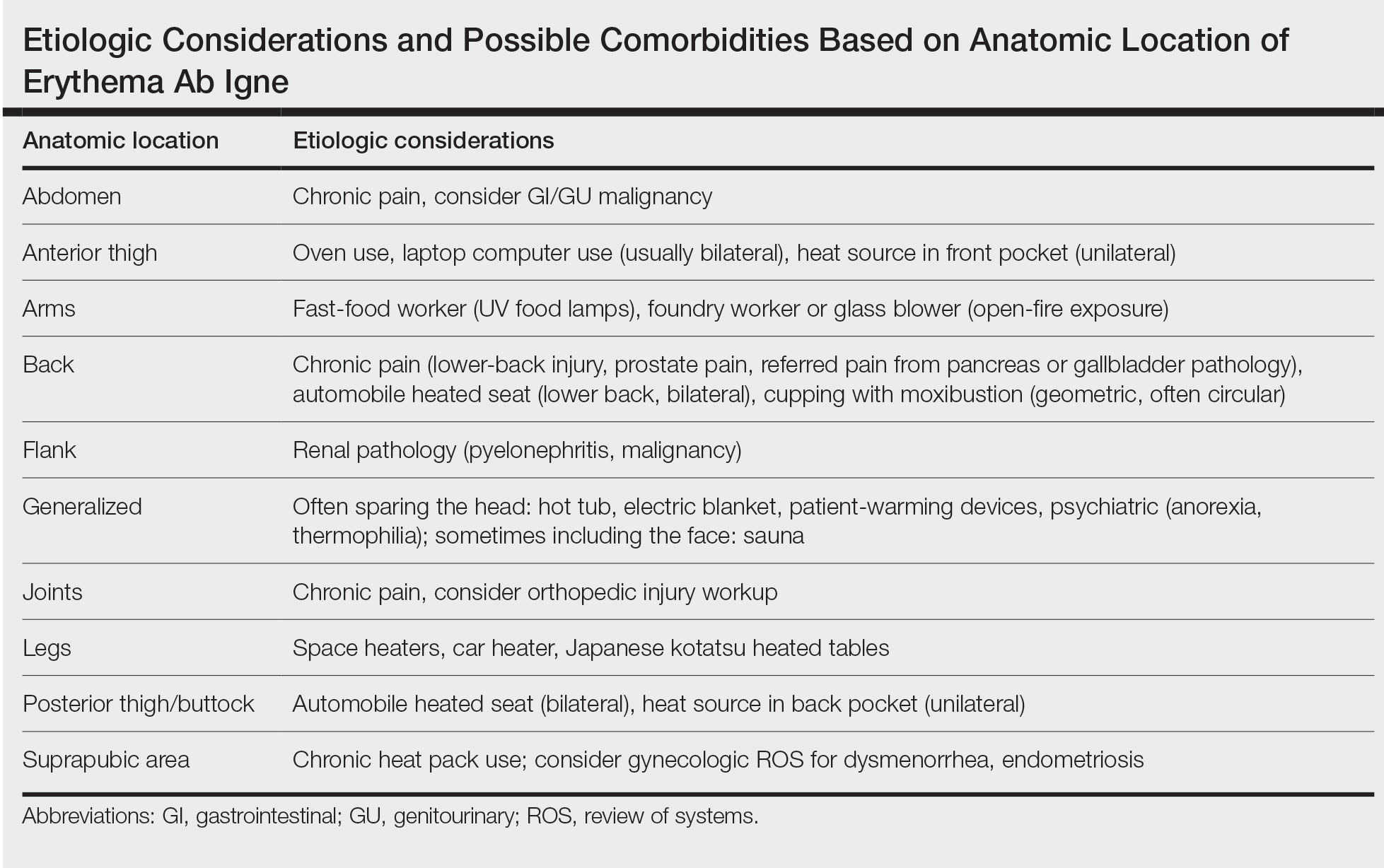
Social History—There are rarer and more highly specific causes of EAI than simple heat exposure that can be parsed from a patient’s social history. Occupational exposure has been documented, such as bakers with exposure to ovens, foundry workers with exposure to heated metals, or fast-food workers with chronic exposure to infrared food lamps.6,7 There also are cultural practices that can cause EAI. For example, the practice of cupping with moxibustion was shown to create a specific pattern in the shape of the cultural tool used.38 When footbaths with Chinese herbal remedies are performed frequently with high heat, they can lead to EAI on the feet with a linear border at the ankles. There also have been reports of kotatsu (heated tables in Japan) leading to lower-body EAI.39,40 These cultural practices also are more common in patients with darker skin types, which can lead to hyperpigmentation that is difficult to treat, making early diagnosis important.7
Medical History—Case reports have shown EAI caused by patients attempting to use heat-based methods for pain relief of an underlying serious disease such as cancer, bowel pathology (abdominal EAI), spinal disc prolapse (midline back EAI),41 sickle cell anemia, and renal pathology (posterior upper flank EAI).6,7,40-49 Patients with hypothyroidism or anorexia have been noted to have generalized EAI sparing the face secondary to repeated and extended hot baths or showers.50-53 One patient with schizophrenia was shown to have associated thermophilia due to a delusion that led the patient to soak in hot baths for long periods of time, leading to EAI.54 Finally, all physicians should be aware of iatrogenic causes of EAI, such as use of warming devices, ultrasound-based warming techniques, and laser therapy for lipolysis. Inquire about the patient’s surgical history or intensive care unit stays as well as alternative medicine or chiropractic visits. Obtaining a history of medical procedures can be enlightening when an etiology is not immediately clear.7,55,56
Diagnosis
Erythema ab igne is a clinical diagnosis based on recognizable cutaneous findings and a clear history of moderate heat exposure. However, when a clinical diagnosis of EAI is not certain (eg, when unable to obtain a clear history from the patient) or when malignant transformation is suspected, a biopsy can be performed. Pathologically, hematoxylin and eosin staining of EAI classically reveals dilated small vascular channels in the superficial dermis, hence a clinically reticular rash; interface dermatitis clinically manifesting as erythema; and pigment incontinence with melanin-laden macrophages consistent with clinical hyperpigmentation. Finally, for unclear reasons, increased numbers of elastic fibers classically are seen in biopsies of EAI.7
Differential Diagnosis
The differential diagnosis for a reticular patch includes livedo reticularis (Figure 3), which usually manifests as a more generalized rash in patients with chronic disease or coagulopathy such as systemic lupus erythematosus, cryoglobulinemia, or Raynaud phenomenon. When differentiating EAI from livedo reticularis or cutis marmorata, consider that both alternative diagnoses are more vascular appearing and are associated with cold exposure rather than heat exposure. In cases that are less reticular, livedo racemosa can be considered in the differential diagnosis. Finally, poikiloderma of Civatte can be reticular, particularly on dermoscopy, but the distribution on the neck with submental sparing should help to distinguish it from EAI unless a heat source around the neck is identified while taking the patient’s history.7
In babies, a reticular generalized rash is most likely to be cutis marmorata (Figure 4), which is a physiologic response to cold exposure that resolves with rewarming of the skin. A more serious condition—cutis marmorata telangiectatica congenita (Figure 5)—usually is present at birth, most frequently involves a single extremity, and notably does not resolve with rewarming. This is an important differential for EAI in children because it can be associated with vascular and neurologic anomalies as well as limb asymmetry. Finally, port-wine stains can sometimes be reticular in appearance and can mimic the early erythematous stages of EAI. However, unlike the erythematous stage of EAI, the port-wine stains will be present at birth.7
Emerging in 2020, an important differential diagnosis to consider is a cutaneous manifestation of COVID-19 infection. An erythematous, reticular, chilblainlike or transient livedo reticularis–like rash has been described as a cutaneous manifestation of COVID-19. Although the pathophysiology is still being elucidated, it is suspected that this is caused by a major vaso-occlusive crisis secondary to COVID-19–induced thrombotic vasculopathy. Interestingly, the majority of patients with this COVID-related exanthem also displayed symptoms of COVID-19 (eg, fever, cough) at the time of presentation,57-60 but there also have been cases in patients who were asymptomatic or mildly symptomatic.60
In some cases, EAI is an indication to screen for an underlying disease. For example, uncontrolled pain is an opportunity to improve interventions such as modifying the patient’s pain-control regimen, placing a palliative care pain consultation, or checking if the patient has had age-appropriate screenings for malignancy. New focal pain in a patient with a prior diagnosis of cancer may be a sign of a new metastasis. A thermophilic patient leaves opportunity to assess for underlying medical causes such as thyroid abnormalities or social/psychological issues. Geriatric patients who are diagnosed with EAI may need to be assessed for dementia or home safety issues. Patients with a history of diabetes mellitus can unknowingly develop EAI on the lower extremities, which may signal a need to assess the patient for peripheral neuropathy. Patients with gastroparesis secondary to diabetes also may develop EAI on the abdomen secondary to heating pad use for discomfort. These examples are a reminder to consider possible secondary comorbidities in all diagnoses of EAI.7
Prognosis
Although the prognosis of EAI is excellent if caught early, failure to diagnose this condition can lead to permanent discoloration of the skin and even malignancy.6 A rare sequela includes squamous cell carcinoma, most commonly seen in chronic cases of the lower leg, which is likely related to chronic inflammation of the skin.61-65 Rare cases of poorly differentiated carcinoma,66 cutaneous marginal zone lymphoma,67 and Merkel cell carcinoma68 have been reported. Patients diagnosed with EAI should receive normal periodic surveillance of the skin based on their medical history, though the physician should have an increased suspicion and plan for biopsy of any nodules or ulcerations found on the skin of the affected area.7
Treatments
Once the diagnosis of EAI is made, treatment starts with removal of the heat source causing the rash. Because the rash usually is asymptomatic, further treatment typically is not required. The discoloration can resolve over months or years, but permanent hyperpigmentation is not uncommon. If hyperpigmentation persists despite removal of the heat source and the patient desires further treatment for discoloration, there are few treatment options, none of which are approved by the US Food and Drug Administration for this condition.7 There is some evidence for the use of Nd:YAG lasers to reduce hyperpigmentation in EAI.69 There have been some reports of treatment using topical hydroquinone and topical tretinoin in an attempt to lighten the skin. If associated hyperkeratosis or other epithelial atypia is present, the use of 5-fluorouracil may show some improvement.70 One case report has been published of successful treatment with systemic mesoglycan and topical bioflavonoids.71 It also is conceivable that medications used to treat postinflammatory hyperpigmentation may be helpful in this condition (eg, kojic acid, arbutin, mild topical steroids, azelaic acid). Patients with darker skin may experience permanent discoloration and may not be good candidates for alternative treatments such as laser therapy due to the risk for inducible hyperpigmentation.7
Conclusion
No matter the etiology, EAI usually is a benign skin condition that is treated by removal of the causative heat source. Once a diagnosis is made, the clinician must work with the patient to determine the etiology. Care must be taken to ensure that there are no underlying signs, such as chronic pain or psychiatric illness, that could point to associated conditions. Rarely, sequalae such as cancers have been documented in areas of chronic EAI. Once the heat source is identified and removed, any remaining hyperpigmentation usually will self-resolve over months to years, though this may take longer in patients with darker skin types. If more aggressive treatment is preferred by the patient, laser therapy, topical medications, and oral over-the-counter vitamins have been tried with minimal responses.
- Perry. Case of erythema ab igne. Br J Dermatol. 1900;xxiii:375.
- Bose S, Ortonee JP. Diseases affected by heat. In: Parish LC, Millikan LE, Amer M, et al. Global Dermatology Diagnosis and Management According to Geography, Climate, and Culture. Springer-Varlag; 1994:83-92.
- Leal-Lobato MM, Blasco-Morente G. Electric blanket induced erythema ab igne [in Spanish]. Semergen. 2015;41:456-457. doi:10.1016/j.semerg.2014.12.008
- Huynh N, Sarma D, Huerter C. Erythema ab igne: a case report and review of the literature. Cutis. 2011;88:290-292.
- Kesty K, Feldman SR. Erythema ab igne: evolving technology, evolving presentation. Dermatol Online J. 2014;20. doi:10.5070/D32011024689
- Miller K, Hunt R, Chu J, et al. Erythema ab igne. Dermatol Online J. 2011;17:28.
- Smith ML. Environmental and sports-related skin diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1569-1594.
- Errichetti E, Stinco G. Dermoscopy in general dermatology: a practical overview. Dermatol Ther (Heidelb). 2016;6:471-507. doi:10.1007/s13555-016-0141-6
- Guarneri C, Tchernev G, Wollina U, et al. Erythema ab igne caused by laptop computer. Open Access Maced J Med Sci. 2017;5:490-492. doi:10.3889/oamjms.2017.137
- Arnold AW, Itin PH. Laptop computer-induced erythema ab igne in a child and review of the literature. Pediatrics. 2010;126:E1227-E1230. doi:10.1542/peds.2010-1390
- Dickman J, Kessler S. Unilateral reticulated patch localized to the anterior thigh. JAAD Case Rep. 2018;4:746-748. doi:10.1016/j.jdcr.2018.06.007
- Boffa MJ. Laptop computer-induced erythema ab igne on the left breast. Cutis. 2011;87:175-176.
- Li K, Barankin B. Cutaneous manifestations of modern technology use. J Cutan Med Surg. 2011;15:347-353. doi:10.2310/7750.2011.10053
- Riahi RR, Cohen PR. Laptop-induced erythema ab igne: report and review of literature. Dermatol Online J. 2012;18:5.
- Andersen F. Laptop-thighs--laptop-induced erythema ab igne [in Danish]. Ugeskr Laeger. 2010;172:635.
- Jagtman BA. Erythema ab igne due to a laptop computer. Contact Dermatitis. 2004;50:105. doi:10.1111/j.0105-1873.2004.0295g.x
- Olechowska M, Kisiel K, Ruszkowska L, et al. Erythema ab igne (EAI) induced by a laptop computer: report of two cases. J Dtsch Dermatol Ges. doi:10.1111/j.1610-0387.2014.12387
- Nayak SUK, Shenoi SD, Prabhu S. Laptop induced erythema ab igne. Indian J Dermatol. 2012;57:131-132. doi:10.4103/0019-5154.94284
- Salvio AG, Nunes AJ, Angarita DPR. Laptop computer induced erythema ab igne: a new presentation of an old disease. An Bras Dermatol. 2016;91:79-80. doi:10.1590/abd1806-4841.20165139
- Schummer C, Tittelbach J, Elsner P. Right-sided laptop dermatitis [in German]. Dtsch Med Wochenschr. 2015;140:1376-1377. doi:10.1055/s-0041-103615
- Manoharan D. Erythema ab igne: usual site, unusual cause. J Pharm Bioallied Sci. 2015;7(suppl 1):S74-S75. doi:10.4103/0975-7406.155811
- Giraldi S, Diettrich F, Abbage KT, et al. Erythema ab igne induced by a laptop computer in an adolescent. An Bras Dermatol. 2011;86:128-130. doi:10.1590/S0365-05962011000100018
- Secher LLS, Vind-Kezunovic D, Zachariae COC. Side-effects to the use of laptop computers: erythema ab igne. Dermatol Reports. 2010;31:E11. doi:10.4081/dr.2010.e11
- Botten D, Langley RGB, Webb A. Academic branding: erythema ab igne and use of laptop computers. CMAJ. 2010;182:E857. doi:10.1503/cmaj.091868
- Bilic M, Adams BB. Erythema ab igne induced by a laptop computer. J Am Acad Dermatol. 2004;50:973-974. doi:10.1016/j.jaad.2003.08.007
- Fu LW, Vender R. Erythema ab igne caused by laptop computer gaming - a case report. Int J Dermatol. 2012;51:716-717. doi:10.1111/j.1365-4632.2011.05033.x
- Levinbook WS, Mallett J, Grant-Kels JM. Laptop computer-associated erythema ab igne. Cutis. 2007;80:319-320.
- Mohr MR, Scott KA, Pariser RM, et al. Laptop computer-induced erythema ab igne: a case report. Cutis. 2007;79:59-60.
- Cantor AS, Bartling SJ. Laptop computer-induced hyperpigmentation. Dermatol Online J. 2018;24:13030/qt6k37r9wm.
- Kaptanog˘lu AF, Mullaaziz D. Erythema ab igne in the palmar area induced by smart phone: case report. Turkiye Klin J Med Sci. 2015;35:284-286. doi:10.5336/medsci.2015-46976
- Redding KS, Watts AN, Lee J, et al. Space heater-induced bullous erythema ab igne. Cutis. 2017;100:E9-E10.
- Goorland J, Edens MA, Baudoin TD. An emergency department presentation of erythema ab igne caused by repeated heater exposure. J La State Med Soc. 2016;168:33-34.
- Kokturk A, Kaya TI, Baz K, et al. Bullous erythema ab igne. Dermatol Online J. 2003;9:18.
- Brzezinski P, Ismail S, Chiriac A. Radiator-induced erythema ab igne in 8-year-old girl. Rev Chil Pediatr. 2014;85:239-240. doi:10.4067/S0370-41062014000200015
- Adams BB. Heated car seat-induced erythema ab igne. Arch Dermatol. 2012;148:265-266. doi:10.1001/archdermatol.2011.2207
- Helm TN, Spigel GT, Helm KF. Erythema ab igne caused by a car heater. Cutis. 1997;59:81-82.
- Gregory JF, Beute TC. Erythema ab igne. J Spec Oper Med. 2013;13:115-119. doi:10.55460/5AVH-NZHY
- Chua S, Chen Q, Lee HY. Erythema ab igne and dermal scarring caused by cupping and moxibustion treatment. J Dtsch Dermatol Ges. 2015;13:337-338. doi:10.1111/ddg.12581
- Chen JF, Liu YC, Chen YF, et al. Erythema ab igne after footbath with Chinese herbal remedies. J Chinese Med Assoc. 2011;74:51-53. doi:10.1016/j.jcma.2011.01.009
- Baltazar D, Brockman R, Simpson E. Kotatsu-induced erythema ab igne. An Bras Dermatol. 2019;94:253-254. doi:10.1590/abd1806-4841.20198792
- Baig M, Byrne F. Erythema ab igne and its relation to spinal pathology. Cureus. 2018;10:e2914. doi:10.7759/cureus.2914
- Aria AB, Chen L, Silapunt S. Erythema ab igne from heating pad use: a report of three clinical cases and a differential diagnosis. Cureus. 2018;10:e2635. doi:10.7759/cureus.2635
- Milchak M, Smucker J, Chung CG, et al. Erythema ab igne due to heating pad use: a case report and review of clinical presentation, prevention, and complications. Case Rep Med. 2016;1862480. doi:10.1155/2016/1862480
- Gmuca S, Yu J, Weiss PF, et al. Erythema ab igne in an adolescent with chronic pain: an alarming cutaneous eruption from heat exposure. Pediatr Emerg Care. 2020;36:e236-e238. doi:10.1097/PEC.0000000000001460
- Dizdarevic A, Karim OA, Bygum A. A reddish brown reticulated hyperpigmented erythema on the abdomen of a girl. Erythema ab igne, also known as toasted skin syndrome, caused by a heating pad onthe abdomen. Acta Derm Venereol. 2014;94:365-367. doi:10.2340/00015555-1722
- Chatterjee S. Erythema ab igne from prolonged use of a heating pad. Mayo Clin Proc. 2005;80:1500. doi:10.4065/80.11.1500
- Waldorf DS, Rast MF, Garofalo VJ. Heating-pad erythematous dermatitis “erythema ab igne.” JAMA. 1971;218:1704. doi:10.1001/jama.1971.03190240056023
- South AM, Crispin MK, Marqueling AL, et al. A hyperpigmented reticular rash in a patient on peritoneal dialysis. Perit Dial Int. 2016;36:677-700. doi:10.3747/pdi.2016.00042
- Ravindran R. Erythema ab igne in an individual with diabetes and gastroparesis. BMJ Case Rep. 2017;2017:bcr2014203856. doi:10.1136/bcr-2014-203856
- Dessinioti C, Katsambas A, Tzavela E, et al. Erythema ab igne in three girls with anorexia nervosa. Pediatr Dermatol. 2016;33:e149-e150. doi:10.1111/pde.12770
- Fischer J, Rein K, Erfurt-Berge C, et al. Three cases of erythema ab igne (EAI) in patients with eating disorders. Neuropsychiatr. 2010;24:141-143.
- Docx MKF, Simons A, Ramet J, et al. Erythema ab igne in an adolescent with anorexia nervosa. Int J Eat Disord. 2013;46:381-383. doi:10.1002/eat.22075
- Turan E, Cimen V, Haytoglu NSK, et al. A case of bullous erythema ab igne accompanied by anemia and subclinical hypothyroidism. Dermatol Online J. 2014;20:223366.
- Pavithran K. Erythema ab igne, schizophrenia and thermophilia. Indian J Dermatol Venereol Leprol. 1987;53:181-182.
- Dellavelle R, Gillum P. Erythema ab igne following heating/cooling blanket use in the intensive care unit. Cutis. 2000;66:136-138.
- Park SY, Kim SM, Yoon TJ. Erythema ab igne caused by weight loss heating pad. Korean J Dermatol. 2007;45:489-491.
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98:75-81. doi:10.1016/j.jdermsci.2020.04.011
- Gisondi P, Plaserico S, Bordin C, et al. Cutaneous manifestations of SARS‐CoV‐2 infection: a clinical update. J Eur Acad Dermatol Venereol. 2020;34:2499-2504. doi:10.1111/jdv.16774
- Manalo IF, Smith MK, Cheeley J, et al. A dermatologic manifestation of COVID-19: transient livedo reticularis. J Am Acad Dermatol. 2020;83:700. doi:10.1016/j.jaad.2020.04.018
- Zhao Q, Fang X, Pang Z, et al. COVID‐19 and cutaneous manifestations: a systematic review. J Eur Acad Dermatol Venereol. 2020;34:2505-2510. doi:10.1111/jdv.16778
- Akasaka T, Kon S. Two cases of squamous cell carcinoma arising from erythema ab igne. Nihon Hifuka Gakkai Zasshi. 1989;99:735-742.
- Arrington JH 3rd, Lockman DS. Thermal keratoses and squamous cell carcinoma in situ associated with erythema ab igne. Arch Dermatol. 1979;115:1226-1228.
- Wharton JB, Sheehan DJ, Lesher JL Jr. Squamous cell carcinoma in situ arising in the setting of erythema ab igne. J Drugs Dermatol. 2008;7:488-489.
- Wollina U, Helm C, Hansel G, et al. Two cases of erythema ab igne, one with a squamous cell carcinoma. G Ital Dermatol Venereol. 2007;142:415-418.
- Rudolph CM, Soyer HP, Wolf P, et al. Squamous cell carcinoma arising in erythema ab igne. Hautarzt. 2000;51:260-263. doi:10.1007/s001050051115
- Sigmon JR, Cantrell J, Teague D, et al. Poorly differentiated carcinoma arising in the setting of erythema ab igne. Am J Dermatopathol. 2013;35:676-678. doi:10.1097/DAD.0b013e3182871648
- Wharton J, Roffwarg D, Miller J, et al. Cutaneous marginal zone lymphoma arising in the setting of erythema ab igne. J Am Acad Dermatol. 2010;62:1080-1081. doi:10.1016/j.jaad.2009.08.005
- Jones CS, Tyring SK, Lee PC, et al. Development of neuroendocrine (Merkel cell) carcinoma mixed with squamous cell carcinoma in erythema ab igne. Arch Dermatol. 1988;124:110-113.
- Kim HW, Kim EJ, Park HC, et al. Erythema ab igne successfully treated with low fluenced 1,064-nm Q-switched neodymium-doped yttrium aluminum garnet laser. J Cosmet Laser Ther. 2014;16:147-148. doi:10.3109/14764172.2013.854623
- Tan S, Bertucci V. Erythema ab igne: an old condition new again. CMAJ. 2000;62:77-78.
- Gianfaldoni S, Gianfaldoni R, Tchernev G, et al. Erythema ab igne successfully treated with mesoglycan and bioflavonoids: a case-report. Open Access Maced J Med Sci. 2017;5:432-435. doi:10.3889/oamjms.2017.123
Erythema ab igne (EAI)(also known as toasted skin syndrome) was first described in the British Journal of Dermatology in the 20th century, 1 though it was known by physicians long before. Reticular netlike skin changes were seen in association with patients who spent extended time directly next to a heat source. This association led to the name of this condition, which literally means “redness by fire.” Indeed, EAI induced by chronic heat exposure has been described across the world for centuries. For example, in the cold regions of northern China, people used to sleep on beds of hot bricks called kang to stay warm at night. The people of India’s Kashmir district carried pots of hot coals called kangri next to the skin under large woven shawls to stay warm. In the past, Irish women often spent much time by a turf- or peat-burning fire. Chronic heat exposure in these cases can lead not only to EAI but also to aggressive types of cancer, often with a latency of 30 years or more. 2
More recently, the invention of home central heating led to a stark decrease in the number of cases associated with combustion-based heat, with a transition to etiologies such as use of hot water bottles, electric blankets, and electric space heaters. Over time, technological advances led to ever-increasing potential causes for EAI, such as laptops or cell phones, car heaters and heated seats, heated blankets,3,4 infrared lamps for food, and even medical devices such as ultrasound-based heating products and convective temperature management systems for hospitalized patients. As technology evolves, so do the potential causes of EAI, requiring clinicians to diagnose and deduce the cause through a thorough social and medical history as well as a workup on the present illness with considerations for the anatomical location.5-7 Herein, we describe the etiology of EAI, diagnosis, and treatment options.

Clinical Characteristics
Erythema ab igne begins as mild, transient, and erythematous macules and patches in a reticular pattern that resolve minutes to hours after removal of the heat source. With weeks to months of continued or repeated application of the heat source, the affected area eventually becomes hyperpigmented where there once was erythema (Figures 1 and 2). Sometimes papules, bullae, telangiectasia, and hyperkeratosis also form. The rash usually is asymptomatic, though pain, pruritus, and dysesthesia have been reported.7 Dermoscopy of EAI in the hyperpigmented stage can reveal diffuse superficial dark pigmentation, telangiectasia, and mild whitish scaling.8 Although the pathogenesis has remained elusive over the years, lesions do seem to be mostly associated with cumulative exposure to heat rather than length of exposure.7

Etiology of EAI
Anatomic Location—The affected site depends on the source of heat (Table). Classic examples of this condition include a patient with EAI presenting on the anterior thighs after working in front of a hot oven or a patient with chronic back pain presenting with lower-back EAI secondary to frequent use of a hot water bottle or heating pad.7 With evolving technology over the last few decades, new etiologies have become more common—teenagers are presenting with anterior thigh EAI secondary to frequent laptop use2-29; patients are holding warm cell phones in their pant pockets, leading to unilateral geometric EAI on the anterior thigh (front pocket) or buttock (back pocket)30; plug-in radiators under computer desks are causing EAI on the lower legs31-34; and automobile seat heaters have been shown to cause EAI on the posterior legs.5,35-37 Clinicians should consider anatomic location a critical clue for etiology.

Social History—There are rarer and more highly specific causes of EAI than simple heat exposure that can be parsed from a patient’s social history. Occupational exposure has been documented, such as bakers with exposure to ovens, foundry workers with exposure to heated metals, or fast-food workers with chronic exposure to infrared food lamps.6,7 There also are cultural practices that can cause EAI. For example, the practice of cupping with moxibustion was shown to create a specific pattern in the shape of the cultural tool used.38 When footbaths with Chinese herbal remedies are performed frequently with high heat, they can lead to EAI on the feet with a linear border at the ankles. There also have been reports of kotatsu (heated tables in Japan) leading to lower-body EAI.39,40 These cultural practices also are more common in patients with darker skin types, which can lead to hyperpigmentation that is difficult to treat, making early diagnosis important.7
Medical History—Case reports have shown EAI caused by patients attempting to use heat-based methods for pain relief of an underlying serious disease such as cancer, bowel pathology (abdominal EAI), spinal disc prolapse (midline back EAI),41 sickle cell anemia, and renal pathology (posterior upper flank EAI).6,7,40-49 Patients with hypothyroidism or anorexia have been noted to have generalized EAI sparing the face secondary to repeated and extended hot baths or showers.50-53 One patient with schizophrenia was shown to have associated thermophilia due to a delusion that led the patient to soak in hot baths for long periods of time, leading to EAI.54 Finally, all physicians should be aware of iatrogenic causes of EAI, such as use of warming devices, ultrasound-based warming techniques, and laser therapy for lipolysis. Inquire about the patient’s surgical history or intensive care unit stays as well as alternative medicine or chiropractic visits. Obtaining a history of medical procedures can be enlightening when an etiology is not immediately clear.7,55,56
Diagnosis
Erythema ab igne is a clinical diagnosis based on recognizable cutaneous findings and a clear history of moderate heat exposure. However, when a clinical diagnosis of EAI is not certain (eg, when unable to obtain a clear history from the patient) or when malignant transformation is suspected, a biopsy can be performed. Pathologically, hematoxylin and eosin staining of EAI classically reveals dilated small vascular channels in the superficial dermis, hence a clinically reticular rash; interface dermatitis clinically manifesting as erythema; and pigment incontinence with melanin-laden macrophages consistent with clinical hyperpigmentation. Finally, for unclear reasons, increased numbers of elastic fibers classically are seen in biopsies of EAI.7
Differential Diagnosis
The differential diagnosis for a reticular patch includes livedo reticularis (Figure 3), which usually manifests as a more generalized rash in patients with chronic disease or coagulopathy such as systemic lupus erythematosus, cryoglobulinemia, or Raynaud phenomenon. When differentiating EAI from livedo reticularis or cutis marmorata, consider that both alternative diagnoses are more vascular appearing and are associated with cold exposure rather than heat exposure. In cases that are less reticular, livedo racemosa can be considered in the differential diagnosis. Finally, poikiloderma of Civatte can be reticular, particularly on dermoscopy, but the distribution on the neck with submental sparing should help to distinguish it from EAI unless a heat source around the neck is identified while taking the patient’s history.7

In babies, a reticular generalized rash is most likely to be cutis marmorata (Figure 4), which is a physiologic response to cold exposure that resolves with rewarming of the skin. A more serious condition—cutis marmorata telangiectatica congenita (Figure 5)—usually is present at birth, most frequently involves a single extremity, and notably does not resolve with rewarming. This is an important differential for EAI in children because it can be associated with vascular and neurologic anomalies as well as limb asymmetry. Finally, port-wine stains can sometimes be reticular in appearance and can mimic the early erythematous stages of EAI. However, unlike the erythematous stage of EAI, the port-wine stains will be present at birth.7

Emerging in 2020, an important differential diagnosis to consider is a cutaneous manifestation of COVID-19 infection. An erythematous, reticular, chilblainlike or transient livedo reticularis–like rash has been described as a cutaneous manifestation of COVID-19. Although the pathophysiology is still being elucidated, it is suspected that this is caused by a major vaso-occlusive crisis secondary to COVID-19–induced thrombotic vasculopathy. Interestingly, the majority of patients with this COVID-related exanthem also displayed symptoms of COVID-19 (eg, fever, cough) at the time of presentation,57-60 but there also have been cases in patients who were asymptomatic or mildly symptomatic.60

In some cases, EAI is an indication to screen for an underlying disease. For example, uncontrolled pain is an opportunity to improve interventions such as modifying the patient’s pain-control regimen, placing a palliative care pain consultation, or checking if the patient has had age-appropriate screenings for malignancy. New focal pain in a patient with a prior diagnosis of cancer may be a sign of a new metastasis. A thermophilic patient leaves opportunity to assess for underlying medical causes such as thyroid abnormalities or social/psychological issues. Geriatric patients who are diagnosed with EAI may need to be assessed for dementia or home safety issues. Patients with a history of diabetes mellitus can unknowingly develop EAI on the lower extremities, which may signal a need to assess the patient for peripheral neuropathy. Patients with gastroparesis secondary to diabetes also may develop EAI on the abdomen secondary to heating pad use for discomfort. These examples are a reminder to consider possible secondary comorbidities in all diagnoses of EAI.7
Prognosis
Although the prognosis of EAI is excellent if caught early, failure to diagnose this condition can lead to permanent discoloration of the skin and even malignancy.6 A rare sequela includes squamous cell carcinoma, most commonly seen in chronic cases of the lower leg, which is likely related to chronic inflammation of the skin.61-65 Rare cases of poorly differentiated carcinoma,66 cutaneous marginal zone lymphoma,67 and Merkel cell carcinoma68 have been reported. Patients diagnosed with EAI should receive normal periodic surveillance of the skin based on their medical history, though the physician should have an increased suspicion and plan for biopsy of any nodules or ulcerations found on the skin of the affected area.7
Treatments
Once the diagnosis of EAI is made, treatment starts with removal of the heat source causing the rash. Because the rash usually is asymptomatic, further treatment typically is not required. The discoloration can resolve over months or years, but permanent hyperpigmentation is not uncommon. If hyperpigmentation persists despite removal of the heat source and the patient desires further treatment for discoloration, there are few treatment options, none of which are approved by the US Food and Drug Administration for this condition.7 There is some evidence for the use of Nd:YAG lasers to reduce hyperpigmentation in EAI.69 There have been some reports of treatment using topical hydroquinone and topical tretinoin in an attempt to lighten the skin. If associated hyperkeratosis or other epithelial atypia is present, the use of 5-fluorouracil may show some improvement.70 One case report has been published of successful treatment with systemic mesoglycan and topical bioflavonoids.71 It also is conceivable that medications used to treat postinflammatory hyperpigmentation may be helpful in this condition (eg, kojic acid, arbutin, mild topical steroids, azelaic acid). Patients with darker skin may experience permanent discoloration and may not be good candidates for alternative treatments such as laser therapy due to the risk for inducible hyperpigmentation.7
Conclusion
No matter the etiology, EAI usually is a benign skin condition that is treated by removal of the causative heat source. Once a diagnosis is made, the clinician must work with the patient to determine the etiology. Care must be taken to ensure that there are no underlying signs, such as chronic pain or psychiatric illness, that could point to associated conditions. Rarely, sequalae such as cancers have been documented in areas of chronic EAI. Once the heat source is identified and removed, any remaining hyperpigmentation usually will self-resolve over months to years, though this may take longer in patients with darker skin types. If more aggressive treatment is preferred by the patient, laser therapy, topical medications, and oral over-the-counter vitamins have been tried with minimal responses.
Erythema ab igne (EAI)(also known as toasted skin syndrome) was first described in the British Journal of Dermatology in the 20th century, 1 though it was known by physicians long before. Reticular netlike skin changes were seen in association with patients who spent extended time directly next to a heat source. This association led to the name of this condition, which literally means “redness by fire.” Indeed, EAI induced by chronic heat exposure has been described across the world for centuries. For example, in the cold regions of northern China, people used to sleep on beds of hot bricks called kang to stay warm at night. The people of India’s Kashmir district carried pots of hot coals called kangri next to the skin under large woven shawls to stay warm. In the past, Irish women often spent much time by a turf- or peat-burning fire. Chronic heat exposure in these cases can lead not only to EAI but also to aggressive types of cancer, often with a latency of 30 years or more. 2
More recently, the invention of home central heating led to a stark decrease in the number of cases associated with combustion-based heat, with a transition to etiologies such as use of hot water bottles, electric blankets, and electric space heaters. Over time, technological advances led to ever-increasing potential causes for EAI, such as laptops or cell phones, car heaters and heated seats, heated blankets,3,4 infrared lamps for food, and even medical devices such as ultrasound-based heating products and convective temperature management systems for hospitalized patients. As technology evolves, so do the potential causes of EAI, requiring clinicians to diagnose and deduce the cause through a thorough social and medical history as well as a workup on the present illness with considerations for the anatomical location.5-7 Herein, we describe the etiology of EAI, diagnosis, and treatment options.

Clinical Characteristics
Erythema ab igne begins as mild, transient, and erythematous macules and patches in a reticular pattern that resolve minutes to hours after removal of the heat source. With weeks to months of continued or repeated application of the heat source, the affected area eventually becomes hyperpigmented where there once was erythema (Figures 1 and 2). Sometimes papules, bullae, telangiectasia, and hyperkeratosis also form. The rash usually is asymptomatic, though pain, pruritus, and dysesthesia have been reported.7 Dermoscopy of EAI in the hyperpigmented stage can reveal diffuse superficial dark pigmentation, telangiectasia, and mild whitish scaling.8 Although the pathogenesis has remained elusive over the years, lesions do seem to be mostly associated with cumulative exposure to heat rather than length of exposure.7

Etiology of EAI
Anatomic Location—The affected site depends on the source of heat (Table). Classic examples of this condition include a patient with EAI presenting on the anterior thighs after working in front of a hot oven or a patient with chronic back pain presenting with lower-back EAI secondary to frequent use of a hot water bottle or heating pad.7 With evolving technology over the last few decades, new etiologies have become more common—teenagers are presenting with anterior thigh EAI secondary to frequent laptop use2-29; patients are holding warm cell phones in their pant pockets, leading to unilateral geometric EAI on the anterior thigh (front pocket) or buttock (back pocket)30; plug-in radiators under computer desks are causing EAI on the lower legs31-34; and automobile seat heaters have been shown to cause EAI on the posterior legs.5,35-37 Clinicians should consider anatomic location a critical clue for etiology.

Social History—There are rarer and more highly specific causes of EAI than simple heat exposure that can be parsed from a patient’s social history. Occupational exposure has been documented, such as bakers with exposure to ovens, foundry workers with exposure to heated metals, or fast-food workers with chronic exposure to infrared food lamps.6,7 There also are cultural practices that can cause EAI. For example, the practice of cupping with moxibustion was shown to create a specific pattern in the shape of the cultural tool used.38 When footbaths with Chinese herbal remedies are performed frequently with high heat, they can lead to EAI on the feet with a linear border at the ankles. There also have been reports of kotatsu (heated tables in Japan) leading to lower-body EAI.39,40 These cultural practices also are more common in patients with darker skin types, which can lead to hyperpigmentation that is difficult to treat, making early diagnosis important.7
Medical History—Case reports have shown EAI caused by patients attempting to use heat-based methods for pain relief of an underlying serious disease such as cancer, bowel pathology (abdominal EAI), spinal disc prolapse (midline back EAI),41 sickle cell anemia, and renal pathology (posterior upper flank EAI).6,7,40-49 Patients with hypothyroidism or anorexia have been noted to have generalized EAI sparing the face secondary to repeated and extended hot baths or showers.50-53 One patient with schizophrenia was shown to have associated thermophilia due to a delusion that led the patient to soak in hot baths for long periods of time, leading to EAI.54 Finally, all physicians should be aware of iatrogenic causes of EAI, such as use of warming devices, ultrasound-based warming techniques, and laser therapy for lipolysis. Inquire about the patient’s surgical history or intensive care unit stays as well as alternative medicine or chiropractic visits. Obtaining a history of medical procedures can be enlightening when an etiology is not immediately clear.7,55,56
Diagnosis
Erythema ab igne is a clinical diagnosis based on recognizable cutaneous findings and a clear history of moderate heat exposure. However, when a clinical diagnosis of EAI is not certain (eg, when unable to obtain a clear history from the patient) or when malignant transformation is suspected, a biopsy can be performed. Pathologically, hematoxylin and eosin staining of EAI classically reveals dilated small vascular channels in the superficial dermis, hence a clinically reticular rash; interface dermatitis clinically manifesting as erythema; and pigment incontinence with melanin-laden macrophages consistent with clinical hyperpigmentation. Finally, for unclear reasons, increased numbers of elastic fibers classically are seen in biopsies of EAI.7
Differential Diagnosis
The differential diagnosis for a reticular patch includes livedo reticularis (Figure 3), which usually manifests as a more generalized rash in patients with chronic disease or coagulopathy such as systemic lupus erythematosus, cryoglobulinemia, or Raynaud phenomenon. When differentiating EAI from livedo reticularis or cutis marmorata, consider that both alternative diagnoses are more vascular appearing and are associated with cold exposure rather than heat exposure. In cases that are less reticular, livedo racemosa can be considered in the differential diagnosis. Finally, poikiloderma of Civatte can be reticular, particularly on dermoscopy, but the distribution on the neck with submental sparing should help to distinguish it from EAI unless a heat source around the neck is identified while taking the patient’s history.7

In babies, a reticular generalized rash is most likely to be cutis marmorata (Figure 4), which is a physiologic response to cold exposure that resolves with rewarming of the skin. A more serious condition—cutis marmorata telangiectatica congenita (Figure 5)—usually is present at birth, most frequently involves a single extremity, and notably does not resolve with rewarming. This is an important differential for EAI in children because it can be associated with vascular and neurologic anomalies as well as limb asymmetry. Finally, port-wine stains can sometimes be reticular in appearance and can mimic the early erythematous stages of EAI. However, unlike the erythematous stage of EAI, the port-wine stains will be present at birth.7

Emerging in 2020, an important differential diagnosis to consider is a cutaneous manifestation of COVID-19 infection. An erythematous, reticular, chilblainlike or transient livedo reticularis–like rash has been described as a cutaneous manifestation of COVID-19. Although the pathophysiology is still being elucidated, it is suspected that this is caused by a major vaso-occlusive crisis secondary to COVID-19–induced thrombotic vasculopathy. Interestingly, the majority of patients with this COVID-related exanthem also displayed symptoms of COVID-19 (eg, fever, cough) at the time of presentation,57-60 but there also have been cases in patients who were asymptomatic or mildly symptomatic.60

In some cases, EAI is an indication to screen for an underlying disease. For example, uncontrolled pain is an opportunity to improve interventions such as modifying the patient’s pain-control regimen, placing a palliative care pain consultation, or checking if the patient has had age-appropriate screenings for malignancy. New focal pain in a patient with a prior diagnosis of cancer may be a sign of a new metastasis. A thermophilic patient leaves opportunity to assess for underlying medical causes such as thyroid abnormalities or social/psychological issues. Geriatric patients who are diagnosed with EAI may need to be assessed for dementia or home safety issues. Patients with a history of diabetes mellitus can unknowingly develop EAI on the lower extremities, which may signal a need to assess the patient for peripheral neuropathy. Patients with gastroparesis secondary to diabetes also may develop EAI on the abdomen secondary to heating pad use for discomfort. These examples are a reminder to consider possible secondary comorbidities in all diagnoses of EAI.7
Prognosis
Although the prognosis of EAI is excellent if caught early, failure to diagnose this condition can lead to permanent discoloration of the skin and even malignancy.6 A rare sequela includes squamous cell carcinoma, most commonly seen in chronic cases of the lower leg, which is likely related to chronic inflammation of the skin.61-65 Rare cases of poorly differentiated carcinoma,66 cutaneous marginal zone lymphoma,67 and Merkel cell carcinoma68 have been reported. Patients diagnosed with EAI should receive normal periodic surveillance of the skin based on their medical history, though the physician should have an increased suspicion and plan for biopsy of any nodules or ulcerations found on the skin of the affected area.7
Treatments
Once the diagnosis of EAI is made, treatment starts with removal of the heat source causing the rash. Because the rash usually is asymptomatic, further treatment typically is not required. The discoloration can resolve over months or years, but permanent hyperpigmentation is not uncommon. If hyperpigmentation persists despite removal of the heat source and the patient desires further treatment for discoloration, there are few treatment options, none of which are approved by the US Food and Drug Administration for this condition.7 There is some evidence for the use of Nd:YAG lasers to reduce hyperpigmentation in EAI.69 There have been some reports of treatment using topical hydroquinone and topical tretinoin in an attempt to lighten the skin. If associated hyperkeratosis or other epithelial atypia is present, the use of 5-fluorouracil may show some improvement.70 One case report has been published of successful treatment with systemic mesoglycan and topical bioflavonoids.71 It also is conceivable that medications used to treat postinflammatory hyperpigmentation may be helpful in this condition (eg, kojic acid, arbutin, mild topical steroids, azelaic acid). Patients with darker skin may experience permanent discoloration and may not be good candidates for alternative treatments such as laser therapy due to the risk for inducible hyperpigmentation.7
Conclusion
No matter the etiology, EAI usually is a benign skin condition that is treated by removal of the causative heat source. Once a diagnosis is made, the clinician must work with the patient to determine the etiology. Care must be taken to ensure that there are no underlying signs, such as chronic pain or psychiatric illness, that could point to associated conditions. Rarely, sequalae such as cancers have been documented in areas of chronic EAI. Once the heat source is identified and removed, any remaining hyperpigmentation usually will self-resolve over months to years, though this may take longer in patients with darker skin types. If more aggressive treatment is preferred by the patient, laser therapy, topical medications, and oral over-the-counter vitamins have been tried with minimal responses.
- Perry. Case of erythema ab igne. Br J Dermatol. 1900;xxiii:375.
- Bose S, Ortonee JP. Diseases affected by heat. In: Parish LC, Millikan LE, Amer M, et al. Global Dermatology Diagnosis and Management According to Geography, Climate, and Culture. Springer-Varlag; 1994:83-92.
- Leal-Lobato MM, Blasco-Morente G. Electric blanket induced erythema ab igne [in Spanish]. Semergen. 2015;41:456-457. doi:10.1016/j.semerg.2014.12.008
- Huynh N, Sarma D, Huerter C. Erythema ab igne: a case report and review of the literature. Cutis. 2011;88:290-292.
- Kesty K, Feldman SR. Erythema ab igne: evolving technology, evolving presentation. Dermatol Online J. 2014;20. doi:10.5070/D32011024689
- Miller K, Hunt R, Chu J, et al. Erythema ab igne. Dermatol Online J. 2011;17:28.
- Smith ML. Environmental and sports-related skin diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1569-1594.
- Errichetti E, Stinco G. Dermoscopy in general dermatology: a practical overview. Dermatol Ther (Heidelb). 2016;6:471-507. doi:10.1007/s13555-016-0141-6
- Guarneri C, Tchernev G, Wollina U, et al. Erythema ab igne caused by laptop computer. Open Access Maced J Med Sci. 2017;5:490-492. doi:10.3889/oamjms.2017.137
- Arnold AW, Itin PH. Laptop computer-induced erythema ab igne in a child and review of the literature. Pediatrics. 2010;126:E1227-E1230. doi:10.1542/peds.2010-1390
- Dickman J, Kessler S. Unilateral reticulated patch localized to the anterior thigh. JAAD Case Rep. 2018;4:746-748. doi:10.1016/j.jdcr.2018.06.007
- Boffa MJ. Laptop computer-induced erythema ab igne on the left breast. Cutis. 2011;87:175-176.
- Li K, Barankin B. Cutaneous manifestations of modern technology use. J Cutan Med Surg. 2011;15:347-353. doi:10.2310/7750.2011.10053
- Riahi RR, Cohen PR. Laptop-induced erythema ab igne: report and review of literature. Dermatol Online J. 2012;18:5.
- Andersen F. Laptop-thighs--laptop-induced erythema ab igne [in Danish]. Ugeskr Laeger. 2010;172:635.
- Jagtman BA. Erythema ab igne due to a laptop computer. Contact Dermatitis. 2004;50:105. doi:10.1111/j.0105-1873.2004.0295g.x
- Olechowska M, Kisiel K, Ruszkowska L, et al. Erythema ab igne (EAI) induced by a laptop computer: report of two cases. J Dtsch Dermatol Ges. doi:10.1111/j.1610-0387.2014.12387
- Nayak SUK, Shenoi SD, Prabhu S. Laptop induced erythema ab igne. Indian J Dermatol. 2012;57:131-132. doi:10.4103/0019-5154.94284
- Salvio AG, Nunes AJ, Angarita DPR. Laptop computer induced erythema ab igne: a new presentation of an old disease. An Bras Dermatol. 2016;91:79-80. doi:10.1590/abd1806-4841.20165139
- Schummer C, Tittelbach J, Elsner P. Right-sided laptop dermatitis [in German]. Dtsch Med Wochenschr. 2015;140:1376-1377. doi:10.1055/s-0041-103615
- Manoharan D. Erythema ab igne: usual site, unusual cause. J Pharm Bioallied Sci. 2015;7(suppl 1):S74-S75. doi:10.4103/0975-7406.155811
- Giraldi S, Diettrich F, Abbage KT, et al. Erythema ab igne induced by a laptop computer in an adolescent. An Bras Dermatol. 2011;86:128-130. doi:10.1590/S0365-05962011000100018
- Secher LLS, Vind-Kezunovic D, Zachariae COC. Side-effects to the use of laptop computers: erythema ab igne. Dermatol Reports. 2010;31:E11. doi:10.4081/dr.2010.e11
- Botten D, Langley RGB, Webb A. Academic branding: erythema ab igne and use of laptop computers. CMAJ. 2010;182:E857. doi:10.1503/cmaj.091868
- Bilic M, Adams BB. Erythema ab igne induced by a laptop computer. J Am Acad Dermatol. 2004;50:973-974. doi:10.1016/j.jaad.2003.08.007
- Fu LW, Vender R. Erythema ab igne caused by laptop computer gaming - a case report. Int J Dermatol. 2012;51:716-717. doi:10.1111/j.1365-4632.2011.05033.x
- Levinbook WS, Mallett J, Grant-Kels JM. Laptop computer-associated erythema ab igne. Cutis. 2007;80:319-320.
- Mohr MR, Scott KA, Pariser RM, et al. Laptop computer-induced erythema ab igne: a case report. Cutis. 2007;79:59-60.
- Cantor AS, Bartling SJ. Laptop computer-induced hyperpigmentation. Dermatol Online J. 2018;24:13030/qt6k37r9wm.
- Kaptanog˘lu AF, Mullaaziz D. Erythema ab igne in the palmar area induced by smart phone: case report. Turkiye Klin J Med Sci. 2015;35:284-286. doi:10.5336/medsci.2015-46976
- Redding KS, Watts AN, Lee J, et al. Space heater-induced bullous erythema ab igne. Cutis. 2017;100:E9-E10.
- Goorland J, Edens MA, Baudoin TD. An emergency department presentation of erythema ab igne caused by repeated heater exposure. J La State Med Soc. 2016;168:33-34.
- Kokturk A, Kaya TI, Baz K, et al. Bullous erythema ab igne. Dermatol Online J. 2003;9:18.
- Brzezinski P, Ismail S, Chiriac A. Radiator-induced erythema ab igne in 8-year-old girl. Rev Chil Pediatr. 2014;85:239-240. doi:10.4067/S0370-41062014000200015
- Adams BB. Heated car seat-induced erythema ab igne. Arch Dermatol. 2012;148:265-266. doi:10.1001/archdermatol.2011.2207
- Helm TN, Spigel GT, Helm KF. Erythema ab igne caused by a car heater. Cutis. 1997;59:81-82.
- Gregory JF, Beute TC. Erythema ab igne. J Spec Oper Med. 2013;13:115-119. doi:10.55460/5AVH-NZHY
- Chua S, Chen Q, Lee HY. Erythema ab igne and dermal scarring caused by cupping and moxibustion treatment. J Dtsch Dermatol Ges. 2015;13:337-338. doi:10.1111/ddg.12581
- Chen JF, Liu YC, Chen YF, et al. Erythema ab igne after footbath with Chinese herbal remedies. J Chinese Med Assoc. 2011;74:51-53. doi:10.1016/j.jcma.2011.01.009
- Baltazar D, Brockman R, Simpson E. Kotatsu-induced erythema ab igne. An Bras Dermatol. 2019;94:253-254. doi:10.1590/abd1806-4841.20198792
- Baig M, Byrne F. Erythema ab igne and its relation to spinal pathology. Cureus. 2018;10:e2914. doi:10.7759/cureus.2914
- Aria AB, Chen L, Silapunt S. Erythema ab igne from heating pad use: a report of three clinical cases and a differential diagnosis. Cureus. 2018;10:e2635. doi:10.7759/cureus.2635
- Milchak M, Smucker J, Chung CG, et al. Erythema ab igne due to heating pad use: a case report and review of clinical presentation, prevention, and complications. Case Rep Med. 2016;1862480. doi:10.1155/2016/1862480
- Gmuca S, Yu J, Weiss PF, et al. Erythema ab igne in an adolescent with chronic pain: an alarming cutaneous eruption from heat exposure. Pediatr Emerg Care. 2020;36:e236-e238. doi:10.1097/PEC.0000000000001460
- Dizdarevic A, Karim OA, Bygum A. A reddish brown reticulated hyperpigmented erythema on the abdomen of a girl. Erythema ab igne, also known as toasted skin syndrome, caused by a heating pad onthe abdomen. Acta Derm Venereol. 2014;94:365-367. doi:10.2340/00015555-1722
- Chatterjee S. Erythema ab igne from prolonged use of a heating pad. Mayo Clin Proc. 2005;80:1500. doi:10.4065/80.11.1500
- Waldorf DS, Rast MF, Garofalo VJ. Heating-pad erythematous dermatitis “erythema ab igne.” JAMA. 1971;218:1704. doi:10.1001/jama.1971.03190240056023
- South AM, Crispin MK, Marqueling AL, et al. A hyperpigmented reticular rash in a patient on peritoneal dialysis. Perit Dial Int. 2016;36:677-700. doi:10.3747/pdi.2016.00042
- Ravindran R. Erythema ab igne in an individual with diabetes and gastroparesis. BMJ Case Rep. 2017;2017:bcr2014203856. doi:10.1136/bcr-2014-203856
- Dessinioti C, Katsambas A, Tzavela E, et al. Erythema ab igne in three girls with anorexia nervosa. Pediatr Dermatol. 2016;33:e149-e150. doi:10.1111/pde.12770
- Fischer J, Rein K, Erfurt-Berge C, et al. Three cases of erythema ab igne (EAI) in patients with eating disorders. Neuropsychiatr. 2010;24:141-143.
- Docx MKF, Simons A, Ramet J, et al. Erythema ab igne in an adolescent with anorexia nervosa. Int J Eat Disord. 2013;46:381-383. doi:10.1002/eat.22075
- Turan E, Cimen V, Haytoglu NSK, et al. A case of bullous erythema ab igne accompanied by anemia and subclinical hypothyroidism. Dermatol Online J. 2014;20:223366.
- Pavithran K. Erythema ab igne, schizophrenia and thermophilia. Indian J Dermatol Venereol Leprol. 1987;53:181-182.
- Dellavelle R, Gillum P. Erythema ab igne following heating/cooling blanket use in the intensive care unit. Cutis. 2000;66:136-138.
- Park SY, Kim SM, Yoon TJ. Erythema ab igne caused by weight loss heating pad. Korean J Dermatol. 2007;45:489-491.
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98:75-81. doi:10.1016/j.jdermsci.2020.04.011
- Gisondi P, Plaserico S, Bordin C, et al. Cutaneous manifestations of SARS‐CoV‐2 infection: a clinical update. J Eur Acad Dermatol Venereol. 2020;34:2499-2504. doi:10.1111/jdv.16774
- Manalo IF, Smith MK, Cheeley J, et al. A dermatologic manifestation of COVID-19: transient livedo reticularis. J Am Acad Dermatol. 2020;83:700. doi:10.1016/j.jaad.2020.04.018
- Zhao Q, Fang X, Pang Z, et al. COVID‐19 and cutaneous manifestations: a systematic review. J Eur Acad Dermatol Venereol. 2020;34:2505-2510. doi:10.1111/jdv.16778
- Akasaka T, Kon S. Two cases of squamous cell carcinoma arising from erythema ab igne. Nihon Hifuka Gakkai Zasshi. 1989;99:735-742.
- Arrington JH 3rd, Lockman DS. Thermal keratoses and squamous cell carcinoma in situ associated with erythema ab igne. Arch Dermatol. 1979;115:1226-1228.
- Wharton JB, Sheehan DJ, Lesher JL Jr. Squamous cell carcinoma in situ arising in the setting of erythema ab igne. J Drugs Dermatol. 2008;7:488-489.
- Wollina U, Helm C, Hansel G, et al. Two cases of erythema ab igne, one with a squamous cell carcinoma. G Ital Dermatol Venereol. 2007;142:415-418.
- Rudolph CM, Soyer HP, Wolf P, et al. Squamous cell carcinoma arising in erythema ab igne. Hautarzt. 2000;51:260-263. doi:10.1007/s001050051115
- Sigmon JR, Cantrell J, Teague D, et al. Poorly differentiated carcinoma arising in the setting of erythema ab igne. Am J Dermatopathol. 2013;35:676-678. doi:10.1097/DAD.0b013e3182871648
- Wharton J, Roffwarg D, Miller J, et al. Cutaneous marginal zone lymphoma arising in the setting of erythema ab igne. J Am Acad Dermatol. 2010;62:1080-1081. doi:10.1016/j.jaad.2009.08.005
- Jones CS, Tyring SK, Lee PC, et al. Development of neuroendocrine (Merkel cell) carcinoma mixed with squamous cell carcinoma in erythema ab igne. Arch Dermatol. 1988;124:110-113.
- Kim HW, Kim EJ, Park HC, et al. Erythema ab igne successfully treated with low fluenced 1,064-nm Q-switched neodymium-doped yttrium aluminum garnet laser. J Cosmet Laser Ther. 2014;16:147-148. doi:10.3109/14764172.2013.854623
- Tan S, Bertucci V. Erythema ab igne: an old condition new again. CMAJ. 2000;62:77-78.
- Gianfaldoni S, Gianfaldoni R, Tchernev G, et al. Erythema ab igne successfully treated with mesoglycan and bioflavonoids: a case-report. Open Access Maced J Med Sci. 2017;5:432-435. doi:10.3889/oamjms.2017.123
- Perry. Case of erythema ab igne. Br J Dermatol. 1900;xxiii:375.
- Bose S, Ortonee JP. Diseases affected by heat. In: Parish LC, Millikan LE, Amer M, et al. Global Dermatology Diagnosis and Management According to Geography, Climate, and Culture. Springer-Varlag; 1994:83-92.
- Leal-Lobato MM, Blasco-Morente G. Electric blanket induced erythema ab igne [in Spanish]. Semergen. 2015;41:456-457. doi:10.1016/j.semerg.2014.12.008
- Huynh N, Sarma D, Huerter C. Erythema ab igne: a case report and review of the literature. Cutis. 2011;88:290-292.
- Kesty K, Feldman SR. Erythema ab igne: evolving technology, evolving presentation. Dermatol Online J. 2014;20. doi:10.5070/D32011024689
- Miller K, Hunt R, Chu J, et al. Erythema ab igne. Dermatol Online J. 2011;17:28.
- Smith ML. Environmental and sports-related skin diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1569-1594.
- Errichetti E, Stinco G. Dermoscopy in general dermatology: a practical overview. Dermatol Ther (Heidelb). 2016;6:471-507. doi:10.1007/s13555-016-0141-6
- Guarneri C, Tchernev G, Wollina U, et al. Erythema ab igne caused by laptop computer. Open Access Maced J Med Sci. 2017;5:490-492. doi:10.3889/oamjms.2017.137
- Arnold AW, Itin PH. Laptop computer-induced erythema ab igne in a child and review of the literature. Pediatrics. 2010;126:E1227-E1230. doi:10.1542/peds.2010-1390
- Dickman J, Kessler S. Unilateral reticulated patch localized to the anterior thigh. JAAD Case Rep. 2018;4:746-748. doi:10.1016/j.jdcr.2018.06.007
- Boffa MJ. Laptop computer-induced erythema ab igne on the left breast. Cutis. 2011;87:175-176.
- Li K, Barankin B. Cutaneous manifestations of modern technology use. J Cutan Med Surg. 2011;15:347-353. doi:10.2310/7750.2011.10053
- Riahi RR, Cohen PR. Laptop-induced erythema ab igne: report and review of literature. Dermatol Online J. 2012;18:5.
- Andersen F. Laptop-thighs--laptop-induced erythema ab igne [in Danish]. Ugeskr Laeger. 2010;172:635.
- Jagtman BA. Erythema ab igne due to a laptop computer. Contact Dermatitis. 2004;50:105. doi:10.1111/j.0105-1873.2004.0295g.x
- Olechowska M, Kisiel K, Ruszkowska L, et al. Erythema ab igne (EAI) induced by a laptop computer: report of two cases. J Dtsch Dermatol Ges. doi:10.1111/j.1610-0387.2014.12387
- Nayak SUK, Shenoi SD, Prabhu S. Laptop induced erythema ab igne. Indian J Dermatol. 2012;57:131-132. doi:10.4103/0019-5154.94284
- Salvio AG, Nunes AJ, Angarita DPR. Laptop computer induced erythema ab igne: a new presentation of an old disease. An Bras Dermatol. 2016;91:79-80. doi:10.1590/abd1806-4841.20165139
- Schummer C, Tittelbach J, Elsner P. Right-sided laptop dermatitis [in German]. Dtsch Med Wochenschr. 2015;140:1376-1377. doi:10.1055/s-0041-103615
- Manoharan D. Erythema ab igne: usual site, unusual cause. J Pharm Bioallied Sci. 2015;7(suppl 1):S74-S75. doi:10.4103/0975-7406.155811
- Giraldi S, Diettrich F, Abbage KT, et al. Erythema ab igne induced by a laptop computer in an adolescent. An Bras Dermatol. 2011;86:128-130. doi:10.1590/S0365-05962011000100018
- Secher LLS, Vind-Kezunovic D, Zachariae COC. Side-effects to the use of laptop computers: erythema ab igne. Dermatol Reports. 2010;31:E11. doi:10.4081/dr.2010.e11
- Botten D, Langley RGB, Webb A. Academic branding: erythema ab igne and use of laptop computers. CMAJ. 2010;182:E857. doi:10.1503/cmaj.091868
- Bilic M, Adams BB. Erythema ab igne induced by a laptop computer. J Am Acad Dermatol. 2004;50:973-974. doi:10.1016/j.jaad.2003.08.007
- Fu LW, Vender R. Erythema ab igne caused by laptop computer gaming - a case report. Int J Dermatol. 2012;51:716-717. doi:10.1111/j.1365-4632.2011.05033.x
- Levinbook WS, Mallett J, Grant-Kels JM. Laptop computer-associated erythema ab igne. Cutis. 2007;80:319-320.
- Mohr MR, Scott KA, Pariser RM, et al. Laptop computer-induced erythema ab igne: a case report. Cutis. 2007;79:59-60.
- Cantor AS, Bartling SJ. Laptop computer-induced hyperpigmentation. Dermatol Online J. 2018;24:13030/qt6k37r9wm.
- Kaptanog˘lu AF, Mullaaziz D. Erythema ab igne in the palmar area induced by smart phone: case report. Turkiye Klin J Med Sci. 2015;35:284-286. doi:10.5336/medsci.2015-46976
- Redding KS, Watts AN, Lee J, et al. Space heater-induced bullous erythema ab igne. Cutis. 2017;100:E9-E10.
- Goorland J, Edens MA, Baudoin TD. An emergency department presentation of erythema ab igne caused by repeated heater exposure. J La State Med Soc. 2016;168:33-34.
- Kokturk A, Kaya TI, Baz K, et al. Bullous erythema ab igne. Dermatol Online J. 2003;9:18.
- Brzezinski P, Ismail S, Chiriac A. Radiator-induced erythema ab igne in 8-year-old girl. Rev Chil Pediatr. 2014;85:239-240. doi:10.4067/S0370-41062014000200015
- Adams BB. Heated car seat-induced erythema ab igne. Arch Dermatol. 2012;148:265-266. doi:10.1001/archdermatol.2011.2207
- Helm TN, Spigel GT, Helm KF. Erythema ab igne caused by a car heater. Cutis. 1997;59:81-82.
- Gregory JF, Beute TC. Erythema ab igne. J Spec Oper Med. 2013;13:115-119. doi:10.55460/5AVH-NZHY
- Chua S, Chen Q, Lee HY. Erythema ab igne and dermal scarring caused by cupping and moxibustion treatment. J Dtsch Dermatol Ges. 2015;13:337-338. doi:10.1111/ddg.12581
- Chen JF, Liu YC, Chen YF, et al. Erythema ab igne after footbath with Chinese herbal remedies. J Chinese Med Assoc. 2011;74:51-53. doi:10.1016/j.jcma.2011.01.009
- Baltazar D, Brockman R, Simpson E. Kotatsu-induced erythema ab igne. An Bras Dermatol. 2019;94:253-254. doi:10.1590/abd1806-4841.20198792
- Baig M, Byrne F. Erythema ab igne and its relation to spinal pathology. Cureus. 2018;10:e2914. doi:10.7759/cureus.2914
- Aria AB, Chen L, Silapunt S. Erythema ab igne from heating pad use: a report of three clinical cases and a differential diagnosis. Cureus. 2018;10:e2635. doi:10.7759/cureus.2635
- Milchak M, Smucker J, Chung CG, et al. Erythema ab igne due to heating pad use: a case report and review of clinical presentation, prevention, and complications. Case Rep Med. 2016;1862480. doi:10.1155/2016/1862480
- Gmuca S, Yu J, Weiss PF, et al. Erythema ab igne in an adolescent with chronic pain: an alarming cutaneous eruption from heat exposure. Pediatr Emerg Care. 2020;36:e236-e238. doi:10.1097/PEC.0000000000001460
- Dizdarevic A, Karim OA, Bygum A. A reddish brown reticulated hyperpigmented erythema on the abdomen of a girl. Erythema ab igne, also known as toasted skin syndrome, caused by a heating pad onthe abdomen. Acta Derm Venereol. 2014;94:365-367. doi:10.2340/00015555-1722
- Chatterjee S. Erythema ab igne from prolonged use of a heating pad. Mayo Clin Proc. 2005;80:1500. doi:10.4065/80.11.1500
- Waldorf DS, Rast MF, Garofalo VJ. Heating-pad erythematous dermatitis “erythema ab igne.” JAMA. 1971;218:1704. doi:10.1001/jama.1971.03190240056023
- South AM, Crispin MK, Marqueling AL, et al. A hyperpigmented reticular rash in a patient on peritoneal dialysis. Perit Dial Int. 2016;36:677-700. doi:10.3747/pdi.2016.00042
- Ravindran R. Erythema ab igne in an individual with diabetes and gastroparesis. BMJ Case Rep. 2017;2017:bcr2014203856. doi:10.1136/bcr-2014-203856
- Dessinioti C, Katsambas A, Tzavela E, et al. Erythema ab igne in three girls with anorexia nervosa. Pediatr Dermatol. 2016;33:e149-e150. doi:10.1111/pde.12770
- Fischer J, Rein K, Erfurt-Berge C, et al. Three cases of erythema ab igne (EAI) in patients with eating disorders. Neuropsychiatr. 2010;24:141-143.
- Docx MKF, Simons A, Ramet J, et al. Erythema ab igne in an adolescent with anorexia nervosa. Int J Eat Disord. 2013;46:381-383. doi:10.1002/eat.22075
- Turan E, Cimen V, Haytoglu NSK, et al. A case of bullous erythema ab igne accompanied by anemia and subclinical hypothyroidism. Dermatol Online J. 2014;20:223366.
- Pavithran K. Erythema ab igne, schizophrenia and thermophilia. Indian J Dermatol Venereol Leprol. 1987;53:181-182.
- Dellavelle R, Gillum P. Erythema ab igne following heating/cooling blanket use in the intensive care unit. Cutis. 2000;66:136-138.
- Park SY, Kim SM, Yoon TJ. Erythema ab igne caused by weight loss heating pad. Korean J Dermatol. 2007;45:489-491.
- Sachdeva M, Gianotti R, Shah M, et al. Cutaneous manifestations of COVID-19: report of three cases and a review of literature. J Dermatol Sci. 2020;98:75-81. doi:10.1016/j.jdermsci.2020.04.011
- Gisondi P, Plaserico S, Bordin C, et al. Cutaneous manifestations of SARS‐CoV‐2 infection: a clinical update. J Eur Acad Dermatol Venereol. 2020;34:2499-2504. doi:10.1111/jdv.16774
- Manalo IF, Smith MK, Cheeley J, et al. A dermatologic manifestation of COVID-19: transient livedo reticularis. J Am Acad Dermatol. 2020;83:700. doi:10.1016/j.jaad.2020.04.018
- Zhao Q, Fang X, Pang Z, et al. COVID‐19 and cutaneous manifestations: a systematic review. J Eur Acad Dermatol Venereol. 2020;34:2505-2510. doi:10.1111/jdv.16778
- Akasaka T, Kon S. Two cases of squamous cell carcinoma arising from erythema ab igne. Nihon Hifuka Gakkai Zasshi. 1989;99:735-742.
- Arrington JH 3rd, Lockman DS. Thermal keratoses and squamous cell carcinoma in situ associated with erythema ab igne. Arch Dermatol. 1979;115:1226-1228.
- Wharton JB, Sheehan DJ, Lesher JL Jr. Squamous cell carcinoma in situ arising in the setting of erythema ab igne. J Drugs Dermatol. 2008;7:488-489.
- Wollina U, Helm C, Hansel G, et al. Two cases of erythema ab igne, one with a squamous cell carcinoma. G Ital Dermatol Venereol. 2007;142:415-418.
- Rudolph CM, Soyer HP, Wolf P, et al. Squamous cell carcinoma arising in erythema ab igne. Hautarzt. 2000;51:260-263. doi:10.1007/s001050051115
- Sigmon JR, Cantrell J, Teague D, et al. Poorly differentiated carcinoma arising in the setting of erythema ab igne. Am J Dermatopathol. 2013;35:676-678. doi:10.1097/DAD.0b013e3182871648
- Wharton J, Roffwarg D, Miller J, et al. Cutaneous marginal zone lymphoma arising in the setting of erythema ab igne. J Am Acad Dermatol. 2010;62:1080-1081. doi:10.1016/j.jaad.2009.08.005
- Jones CS, Tyring SK, Lee PC, et al. Development of neuroendocrine (Merkel cell) carcinoma mixed with squamous cell carcinoma in erythema ab igne. Arch Dermatol. 1988;124:110-113.
- Kim HW, Kim EJ, Park HC, et al. Erythema ab igne successfully treated with low fluenced 1,064-nm Q-switched neodymium-doped yttrium aluminum garnet laser. J Cosmet Laser Ther. 2014;16:147-148. doi:10.3109/14764172.2013.854623
- Tan S, Bertucci V. Erythema ab igne: an old condition new again. CMAJ. 2000;62:77-78.
- Gianfaldoni S, Gianfaldoni R, Tchernev G, et al. Erythema ab igne successfully treated with mesoglycan and bioflavonoids: a case-report. Open Access Maced J Med Sci. 2017;5:432-435. doi:10.3889/oamjms.2017.123
Practice Points
- Erythema ab igne (EAI) is a skin condition caused by chronic exposure to heat; removal of the heat source often will result in self-resolution of the rash.
- Erythema ab igne can be a sign of underlying illness in patients self-treating chronic pain with application of heat.
- Recognition and discontinuation of the exposure with close observation are key components in the treatment of EAI.
Could combining topical antioxidants with a nonablative laser prevent acne scars?
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
AT ASLMS 2023
Drive, chip, and putt your way to osteoarthritis relief
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.