User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
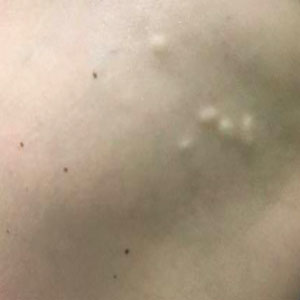
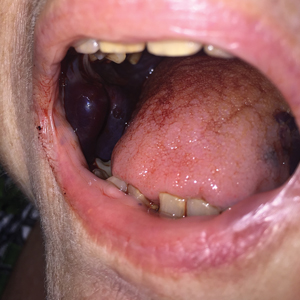
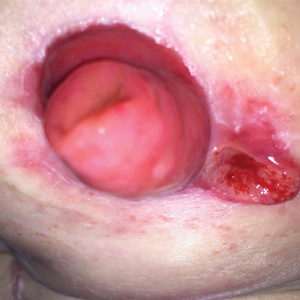
FDA panel urges caution with skin cancer–detecting tools
A and how to address longstanding issues of racial equity in this field of medicine.
The Food and Drug Administration has scheduled two meetings to gather expert feedback about managing an expected expansion in the use of skin lesion apps and devices. Outside of the United States, there are apps promoted as being able to help spot skin lesions that should trigger a medical visit.
The general and plastic surgery devices panel of the FDA’s Medical Devices Advisory Committee began work on this topic on July 28, with a wide-ranging discussion about potential expanded use of computer-aided, skin lesion analyzer (SLA) devices. On Friday, the panel is considering an FDA proposal to shift the designation for an approved device for aiding dermatologists in skin cancer diagnoses from the most stringent regulatory category, class III, to the less restrictive class II.
The FDA called the meeting amid growing interest in using technology to aid in finding cancers, with some of these products already marketed to consumers outside of the United States. There are presently no legally marketed, FDA-cleared or FDA-approved SLA devices indicated for use by clinicians other than dermatologists or the lay public, the agency said in a briefing memo for the meeting. There are two devices with FDA approval, though, for aiding dermatologists. The FDA approved SciBase’s Nevisense in 2017 and Mela Sciences’ MelaFind, which has fallen out of use, in 2012. Both are class III devices.
But some companies intend to offer products for consumers in the United States. The company SkinVision, for example, has developed an app of the same name, which is intended to detect suspicious-looking skin spots via smartphone photos. SkinVision’s website says the product has been offered to consumers in Australia for remote skin checks since 2015. People in the Netherlands and United Kingdom also can use SkinVision, according to the company’s website. SkinVision says the company is working on providing the app for U.S. customers, “but we are not quite there yet.”
During the meeting, FDA panelists repeatedly emphasized the potential risks of these devices in terms of sensitivity (how often a test correctly generates a positive result) and of specificity (how often a test correctly generates a negative result).
New tools intended to aid in detection of skin cancer might produce too many false positives and thus trigger floods of worried patients seeking care and often facing unnecessary biopsies, the FDA panelists said. But more worrisome would be FDA clearance of tools that delivered too many false negative results, leaving people unaware of their cancers.
The standards would have to be set very high for new products, especially those intended for consumers, said FDA panelist Murad Alam, MD, a dermatologist and vice chair of the department of dermatology at Northwestern University, Chicago. Current technologies for analyzing skin lesions are not yet up to that task. Dr. Alam likened the situation to the hopes for self-driving cars.
“It sounds great in principle. If you read the predictions from 20 years ago, it should already have happened,” Dr. Alam said. “But we’re still struggling with that because there are serious points of failure.”
FDA panelist Veronica Rotemberg, MD, PhD, a dermatologist at Memorial Sloan Kettering Cancer Center, New York, also argued for well-designed studies to understand how consumers and clinicians would react to new tools.
“We have to define what prospective information, in the intended use setting, we need to feel comfortable saying that these tools could be in a layperson’s hand or a primary care person’s hand,” Dr. Rotemberg said.
The studies would not need to be large, especially in the case of nonmelanoma skin cancer, which is common, she added.
“There’s too much nuance here for us to be able to say: ‘This is what would happen,’ without testing it,” Dr. Rotemberg said. “I do not think these prospective studies would be very burdensome, but they would help us understand what the burden would be and what the costs would be and what the potential harms would be.”
Because of rules against disclosing corporate information, the FDA cannot tell the public about the kinds of inquiries it already may have fielded from companies interested in selling skin cancer detection tools.
But in response to a question during the FDA meeting, Binita Ashar, MD, a top official in the FDA’s Center for Devices and Radiological Health, said there is interest in having these kinds of products sold in the United States as well.
“I can tell you that this a very timely discussion and questions that we’re posing to you are the questions that we’re encountering or that we have been grappling with,” Dr. Ashar said.
FDA panelists noted that many patients cannot get access easily to dermatology visits.
Companies seeking to develop SLA devices likely will market their tools as attempts to fill a gap that now exists in medical care.
But there will be challenges ahead in explaining to patients how to interpret readings from these tools, the FDA panelists said. Consumers should know these tools are meant to assist in diagnosis, and not to make it.
“I’m not sure the layperson will hear that,” said FDA panelist Paula E. Bourelly, MD, a dermatologist from Olney, Md.
As a result, use of SLA tools could create tension between physicians and patients, with consumers demanding biopsies after seeing readings they don’t understand.
“I do have great concerns about the layperson feeling overly confident and reducing the provider to a technician,” she said.
The FDA panelists were not asked to cast formal votes on any issues discussed during the meeting They instead engaged in broad discussions around questions posed by the FDA in three key areas:
- What standards should be used to confirm lesion diagnosis in clinical testing of the accuracy of SLA devices?
- What would be acceptable true false-positive and false-negative results (sensitivity and specificity) for different diagnoses and users?
- How can the FDA address health equity considerations based on variable incidence of skin lesions?
Developing standards
The FDA asked the panel to consider several scenarios for SLA devices and to discuss how standards might vary depending on the user of the device, whether it would be dermatologists, other clinicians, or consumers.
The agency sought comments in particular about using histological diagnosis (core specimen processing with a consensus diagnosis from an expert dermatopathologist panel). In the briefing document for the meeting, the FDA argued that this approach provides the greatest certainty in the diagnosis.
“Device developers, however, cite concerns, both practical and ethical, in requiring biopsy of all lesions, particularly those that appear benign,” the FDA said. “They have proposed alternate means of defining ground truth, including consensus opinion of experts (of visual or dermoscopic examination of the lesion[s]), opinion of one expert (visual or dermoscopic examination), or other methods.”
In summarizing the discussion on this question, the FDA panel chairman, Hobart W. Harris, MD, MPH, a surgeon from the University of California, San Francisco, noted that there was broad support for histological data in clinical trials of SLA devices, with some allowance for cases where more hybrid approaches would be used.
There were also suggestions offered about designing trials and the need for biopsies of lesions that are clearly benign, as this would help gather data to help in developing algorithms.
Dr. Alam said care should be taken in explaining to study participants that they might have to undergo biopsies that they didn’t need, as part of the larger effort to gather data. This should be detailed in the consent form, he said.
“But I also think this is a relatively minor risk,” Dr. Alam said, comparing these biopsies to the blood samples that patients in many clinical studies routinely give.
“Are all of those blood draws necessary to track the change in whatever parameters that are being tracked? Probably not,” Dr. Alam said. “I think it would be possible to explain to a reasonable patient what this entails.”
Dr. Alam noted that companies might face extra hurdles in enrolling study participants and keeping them in the trials if the FDA seeks this kind of biopsy data. “But I don’t think inconvenience to the study sponsor is a good argument” for not seeking this kind of data, he added.
Leaving a loophole where certain kinds of clearly benign lesions don’t require a biopsy would eventually erode the quality of the research done on these devices. “That bar will be moved to accommodate the convenience of the sponsor, to make the study feasible,” Dr. Alam said. “And pretty soon, you’ll be missing a lot of patients that really should have biopsies.”
Acceptable rates of false positives, false negatives
The FDA panel chair noted that his colleagues had strongly urged review standards that would require that the devices improve on the rates of successful catches of suspicious lesions and lower false positives. But they did not endorse specific targets regarding the sensitivity and specificity rates.
“No one seems to be comfortable with providing or preordaining” these targets, Dr. Harris said.
Panelist Deneen Hesser, MSHSA, RN, urged a deep recognition of the power of a FDA clearance in the view of consumers.
“We need to be cognizant of what the term ‘FDA approved’ means to the lay individual,” said Ms. Hesser, who served as the patient representative on the panel. “A patient who sees that those tools are FDA approved will assume that each of those is the gold standard” in terms of expectations for delivering accurate results.
Like many of the panelists, Dr. Rotemberg urged the FDA to gather data about how patients would react to different messages encoded in consumer-oriented products.
“If the device says: ‘You should see a dermatologist for this’ and no other information, that’s very different from [saying]: ‘That lesion is suspicious for melanoma,’ ” Dr. Rotemberg said.
Despite the likely difficulties in conducting trials, the FDA needs to have the data to answer key questions about patient and physician reactions to readings from new tools, Dr. Rotemberg said.
“We don’t know how many additional biopsies we would cause with a specificity of 80%” for a new SLA tool, Dr. Rotemberg said, giving an example. “We don’t know how confident a dermatologist might be to say: ‘Actually, I’m not suspicious about that lesion and we can just fudge it or not biopsy it.’ We don’t know any of that until we study it in real life.”
The panelists also urged the FDA to seek to ensure that new tools used in analyzing skin lesions improve the quality of diagnosis.
Addressing equity
The FDA also asked the panel to weigh in on whether the agency should clear SLA tools in cases where the existing study data is drawn heavily from people considered to be at higher risk for skin cancer.
“To ensure generalizability across the entire U.S. population, should FDA require SLAs indicated for use beyond cancerous lesions be tested in a representative U.S. population?” the FDA asked.
The three most common skin cancers – melanoma, basal cell carcinoma, and squamous cell carcinoma – are more prevalent in people with Fitzpatrick I and II skin types, who tend to get sunburns, not tans. But people of color are more likely to develop melanoma in areas that are not sun exposed, such as the sole of the foot or under fingernails or toenails.
“Due in part to lower expected risk and screening, these melanomas are often detected late,” the FDA said in the briefing document.
There was broad consensus among panelists that the FDA should encourage companies to enroll people with all skin types and tones.
But they also looked for ways that the FDA could clear devices based on initial studies conducted largely with people considered to be at higher risk, with the agency then requiring follow-up trials to see how these products would work for the general U.S. population.
A version of this article first appeared on Medscape.com.
A and how to address longstanding issues of racial equity in this field of medicine.
The Food and Drug Administration has scheduled two meetings to gather expert feedback about managing an expected expansion in the use of skin lesion apps and devices. Outside of the United States, there are apps promoted as being able to help spot skin lesions that should trigger a medical visit.
The general and plastic surgery devices panel of the FDA’s Medical Devices Advisory Committee began work on this topic on July 28, with a wide-ranging discussion about potential expanded use of computer-aided, skin lesion analyzer (SLA) devices. On Friday, the panel is considering an FDA proposal to shift the designation for an approved device for aiding dermatologists in skin cancer diagnoses from the most stringent regulatory category, class III, to the less restrictive class II.
The FDA called the meeting amid growing interest in using technology to aid in finding cancers, with some of these products already marketed to consumers outside of the United States. There are presently no legally marketed, FDA-cleared or FDA-approved SLA devices indicated for use by clinicians other than dermatologists or the lay public, the agency said in a briefing memo for the meeting. There are two devices with FDA approval, though, for aiding dermatologists. The FDA approved SciBase’s Nevisense in 2017 and Mela Sciences’ MelaFind, which has fallen out of use, in 2012. Both are class III devices.
But some companies intend to offer products for consumers in the United States. The company SkinVision, for example, has developed an app of the same name, which is intended to detect suspicious-looking skin spots via smartphone photos. SkinVision’s website says the product has been offered to consumers in Australia for remote skin checks since 2015. People in the Netherlands and United Kingdom also can use SkinVision, according to the company’s website. SkinVision says the company is working on providing the app for U.S. customers, “but we are not quite there yet.”
During the meeting, FDA panelists repeatedly emphasized the potential risks of these devices in terms of sensitivity (how often a test correctly generates a positive result) and of specificity (how often a test correctly generates a negative result).
New tools intended to aid in detection of skin cancer might produce too many false positives and thus trigger floods of worried patients seeking care and often facing unnecessary biopsies, the FDA panelists said. But more worrisome would be FDA clearance of tools that delivered too many false negative results, leaving people unaware of their cancers.
The standards would have to be set very high for new products, especially those intended for consumers, said FDA panelist Murad Alam, MD, a dermatologist and vice chair of the department of dermatology at Northwestern University, Chicago. Current technologies for analyzing skin lesions are not yet up to that task. Dr. Alam likened the situation to the hopes for self-driving cars.
“It sounds great in principle. If you read the predictions from 20 years ago, it should already have happened,” Dr. Alam said. “But we’re still struggling with that because there are serious points of failure.”
FDA panelist Veronica Rotemberg, MD, PhD, a dermatologist at Memorial Sloan Kettering Cancer Center, New York, also argued for well-designed studies to understand how consumers and clinicians would react to new tools.
“We have to define what prospective information, in the intended use setting, we need to feel comfortable saying that these tools could be in a layperson’s hand or a primary care person’s hand,” Dr. Rotemberg said.
The studies would not need to be large, especially in the case of nonmelanoma skin cancer, which is common, she added.
“There’s too much nuance here for us to be able to say: ‘This is what would happen,’ without testing it,” Dr. Rotemberg said. “I do not think these prospective studies would be very burdensome, but they would help us understand what the burden would be and what the costs would be and what the potential harms would be.”
Because of rules against disclosing corporate information, the FDA cannot tell the public about the kinds of inquiries it already may have fielded from companies interested in selling skin cancer detection tools.
But in response to a question during the FDA meeting, Binita Ashar, MD, a top official in the FDA’s Center for Devices and Radiological Health, said there is interest in having these kinds of products sold in the United States as well.
“I can tell you that this a very timely discussion and questions that we’re posing to you are the questions that we’re encountering or that we have been grappling with,” Dr. Ashar said.
FDA panelists noted that many patients cannot get access easily to dermatology visits.
Companies seeking to develop SLA devices likely will market their tools as attempts to fill a gap that now exists in medical care.
But there will be challenges ahead in explaining to patients how to interpret readings from these tools, the FDA panelists said. Consumers should know these tools are meant to assist in diagnosis, and not to make it.
“I’m not sure the layperson will hear that,” said FDA panelist Paula E. Bourelly, MD, a dermatologist from Olney, Md.
As a result, use of SLA tools could create tension between physicians and patients, with consumers demanding biopsies after seeing readings they don’t understand.
“I do have great concerns about the layperson feeling overly confident and reducing the provider to a technician,” she said.
The FDA panelists were not asked to cast formal votes on any issues discussed during the meeting They instead engaged in broad discussions around questions posed by the FDA in three key areas:
- What standards should be used to confirm lesion diagnosis in clinical testing of the accuracy of SLA devices?
- What would be acceptable true false-positive and false-negative results (sensitivity and specificity) for different diagnoses and users?
- How can the FDA address health equity considerations based on variable incidence of skin lesions?
Developing standards
The FDA asked the panel to consider several scenarios for SLA devices and to discuss how standards might vary depending on the user of the device, whether it would be dermatologists, other clinicians, or consumers.
The agency sought comments in particular about using histological diagnosis (core specimen processing with a consensus diagnosis from an expert dermatopathologist panel). In the briefing document for the meeting, the FDA argued that this approach provides the greatest certainty in the diagnosis.
“Device developers, however, cite concerns, both practical and ethical, in requiring biopsy of all lesions, particularly those that appear benign,” the FDA said. “They have proposed alternate means of defining ground truth, including consensus opinion of experts (of visual or dermoscopic examination of the lesion[s]), opinion of one expert (visual or dermoscopic examination), or other methods.”
In summarizing the discussion on this question, the FDA panel chairman, Hobart W. Harris, MD, MPH, a surgeon from the University of California, San Francisco, noted that there was broad support for histological data in clinical trials of SLA devices, with some allowance for cases where more hybrid approaches would be used.
There were also suggestions offered about designing trials and the need for biopsies of lesions that are clearly benign, as this would help gather data to help in developing algorithms.
Dr. Alam said care should be taken in explaining to study participants that they might have to undergo biopsies that they didn’t need, as part of the larger effort to gather data. This should be detailed in the consent form, he said.
“But I also think this is a relatively minor risk,” Dr. Alam said, comparing these biopsies to the blood samples that patients in many clinical studies routinely give.
“Are all of those blood draws necessary to track the change in whatever parameters that are being tracked? Probably not,” Dr. Alam said. “I think it would be possible to explain to a reasonable patient what this entails.”
Dr. Alam noted that companies might face extra hurdles in enrolling study participants and keeping them in the trials if the FDA seeks this kind of biopsy data. “But I don’t think inconvenience to the study sponsor is a good argument” for not seeking this kind of data, he added.
Leaving a loophole where certain kinds of clearly benign lesions don’t require a biopsy would eventually erode the quality of the research done on these devices. “That bar will be moved to accommodate the convenience of the sponsor, to make the study feasible,” Dr. Alam said. “And pretty soon, you’ll be missing a lot of patients that really should have biopsies.”
Acceptable rates of false positives, false negatives
The FDA panel chair noted that his colleagues had strongly urged review standards that would require that the devices improve on the rates of successful catches of suspicious lesions and lower false positives. But they did not endorse specific targets regarding the sensitivity and specificity rates.
“No one seems to be comfortable with providing or preordaining” these targets, Dr. Harris said.
Panelist Deneen Hesser, MSHSA, RN, urged a deep recognition of the power of a FDA clearance in the view of consumers.
“We need to be cognizant of what the term ‘FDA approved’ means to the lay individual,” said Ms. Hesser, who served as the patient representative on the panel. “A patient who sees that those tools are FDA approved will assume that each of those is the gold standard” in terms of expectations for delivering accurate results.
Like many of the panelists, Dr. Rotemberg urged the FDA to gather data about how patients would react to different messages encoded in consumer-oriented products.
“If the device says: ‘You should see a dermatologist for this’ and no other information, that’s very different from [saying]: ‘That lesion is suspicious for melanoma,’ ” Dr. Rotemberg said.
Despite the likely difficulties in conducting trials, the FDA needs to have the data to answer key questions about patient and physician reactions to readings from new tools, Dr. Rotemberg said.
“We don’t know how many additional biopsies we would cause with a specificity of 80%” for a new SLA tool, Dr. Rotemberg said, giving an example. “We don’t know how confident a dermatologist might be to say: ‘Actually, I’m not suspicious about that lesion and we can just fudge it or not biopsy it.’ We don’t know any of that until we study it in real life.”
The panelists also urged the FDA to seek to ensure that new tools used in analyzing skin lesions improve the quality of diagnosis.
Addressing equity
The FDA also asked the panel to weigh in on whether the agency should clear SLA tools in cases where the existing study data is drawn heavily from people considered to be at higher risk for skin cancer.
“To ensure generalizability across the entire U.S. population, should FDA require SLAs indicated for use beyond cancerous lesions be tested in a representative U.S. population?” the FDA asked.
The three most common skin cancers – melanoma, basal cell carcinoma, and squamous cell carcinoma – are more prevalent in people with Fitzpatrick I and II skin types, who tend to get sunburns, not tans. But people of color are more likely to develop melanoma in areas that are not sun exposed, such as the sole of the foot or under fingernails or toenails.
“Due in part to lower expected risk and screening, these melanomas are often detected late,” the FDA said in the briefing document.
There was broad consensus among panelists that the FDA should encourage companies to enroll people with all skin types and tones.
But they also looked for ways that the FDA could clear devices based on initial studies conducted largely with people considered to be at higher risk, with the agency then requiring follow-up trials to see how these products would work for the general U.S. population.
A version of this article first appeared on Medscape.com.
A and how to address longstanding issues of racial equity in this field of medicine.
The Food and Drug Administration has scheduled two meetings to gather expert feedback about managing an expected expansion in the use of skin lesion apps and devices. Outside of the United States, there are apps promoted as being able to help spot skin lesions that should trigger a medical visit.
The general and plastic surgery devices panel of the FDA’s Medical Devices Advisory Committee began work on this topic on July 28, with a wide-ranging discussion about potential expanded use of computer-aided, skin lesion analyzer (SLA) devices. On Friday, the panel is considering an FDA proposal to shift the designation for an approved device for aiding dermatologists in skin cancer diagnoses from the most stringent regulatory category, class III, to the less restrictive class II.
The FDA called the meeting amid growing interest in using technology to aid in finding cancers, with some of these products already marketed to consumers outside of the United States. There are presently no legally marketed, FDA-cleared or FDA-approved SLA devices indicated for use by clinicians other than dermatologists or the lay public, the agency said in a briefing memo for the meeting. There are two devices with FDA approval, though, for aiding dermatologists. The FDA approved SciBase’s Nevisense in 2017 and Mela Sciences’ MelaFind, which has fallen out of use, in 2012. Both are class III devices.
But some companies intend to offer products for consumers in the United States. The company SkinVision, for example, has developed an app of the same name, which is intended to detect suspicious-looking skin spots via smartphone photos. SkinVision’s website says the product has been offered to consumers in Australia for remote skin checks since 2015. People in the Netherlands and United Kingdom also can use SkinVision, according to the company’s website. SkinVision says the company is working on providing the app for U.S. customers, “but we are not quite there yet.”
During the meeting, FDA panelists repeatedly emphasized the potential risks of these devices in terms of sensitivity (how often a test correctly generates a positive result) and of specificity (how often a test correctly generates a negative result).
New tools intended to aid in detection of skin cancer might produce too many false positives and thus trigger floods of worried patients seeking care and often facing unnecessary biopsies, the FDA panelists said. But more worrisome would be FDA clearance of tools that delivered too many false negative results, leaving people unaware of their cancers.
The standards would have to be set very high for new products, especially those intended for consumers, said FDA panelist Murad Alam, MD, a dermatologist and vice chair of the department of dermatology at Northwestern University, Chicago. Current technologies for analyzing skin lesions are not yet up to that task. Dr. Alam likened the situation to the hopes for self-driving cars.
“It sounds great in principle. If you read the predictions from 20 years ago, it should already have happened,” Dr. Alam said. “But we’re still struggling with that because there are serious points of failure.”
FDA panelist Veronica Rotemberg, MD, PhD, a dermatologist at Memorial Sloan Kettering Cancer Center, New York, also argued for well-designed studies to understand how consumers and clinicians would react to new tools.
“We have to define what prospective information, in the intended use setting, we need to feel comfortable saying that these tools could be in a layperson’s hand or a primary care person’s hand,” Dr. Rotemberg said.
The studies would not need to be large, especially in the case of nonmelanoma skin cancer, which is common, she added.
“There’s too much nuance here for us to be able to say: ‘This is what would happen,’ without testing it,” Dr. Rotemberg said. “I do not think these prospective studies would be very burdensome, but they would help us understand what the burden would be and what the costs would be and what the potential harms would be.”
Because of rules against disclosing corporate information, the FDA cannot tell the public about the kinds of inquiries it already may have fielded from companies interested in selling skin cancer detection tools.
But in response to a question during the FDA meeting, Binita Ashar, MD, a top official in the FDA’s Center for Devices and Radiological Health, said there is interest in having these kinds of products sold in the United States as well.
“I can tell you that this a very timely discussion and questions that we’re posing to you are the questions that we’re encountering or that we have been grappling with,” Dr. Ashar said.
FDA panelists noted that many patients cannot get access easily to dermatology visits.
Companies seeking to develop SLA devices likely will market their tools as attempts to fill a gap that now exists in medical care.
But there will be challenges ahead in explaining to patients how to interpret readings from these tools, the FDA panelists said. Consumers should know these tools are meant to assist in diagnosis, and not to make it.
“I’m not sure the layperson will hear that,” said FDA panelist Paula E. Bourelly, MD, a dermatologist from Olney, Md.
As a result, use of SLA tools could create tension between physicians and patients, with consumers demanding biopsies after seeing readings they don’t understand.
“I do have great concerns about the layperson feeling overly confident and reducing the provider to a technician,” she said.
The FDA panelists were not asked to cast formal votes on any issues discussed during the meeting They instead engaged in broad discussions around questions posed by the FDA in three key areas:
- What standards should be used to confirm lesion diagnosis in clinical testing of the accuracy of SLA devices?
- What would be acceptable true false-positive and false-negative results (sensitivity and specificity) for different diagnoses and users?
- How can the FDA address health equity considerations based on variable incidence of skin lesions?
Developing standards
The FDA asked the panel to consider several scenarios for SLA devices and to discuss how standards might vary depending on the user of the device, whether it would be dermatologists, other clinicians, or consumers.
The agency sought comments in particular about using histological diagnosis (core specimen processing with a consensus diagnosis from an expert dermatopathologist panel). In the briefing document for the meeting, the FDA argued that this approach provides the greatest certainty in the diagnosis.
“Device developers, however, cite concerns, both practical and ethical, in requiring biopsy of all lesions, particularly those that appear benign,” the FDA said. “They have proposed alternate means of defining ground truth, including consensus opinion of experts (of visual or dermoscopic examination of the lesion[s]), opinion of one expert (visual or dermoscopic examination), or other methods.”
In summarizing the discussion on this question, the FDA panel chairman, Hobart W. Harris, MD, MPH, a surgeon from the University of California, San Francisco, noted that there was broad support for histological data in clinical trials of SLA devices, with some allowance for cases where more hybrid approaches would be used.
There were also suggestions offered about designing trials and the need for biopsies of lesions that are clearly benign, as this would help gather data to help in developing algorithms.
Dr. Alam said care should be taken in explaining to study participants that they might have to undergo biopsies that they didn’t need, as part of the larger effort to gather data. This should be detailed in the consent form, he said.
“But I also think this is a relatively minor risk,” Dr. Alam said, comparing these biopsies to the blood samples that patients in many clinical studies routinely give.
“Are all of those blood draws necessary to track the change in whatever parameters that are being tracked? Probably not,” Dr. Alam said. “I think it would be possible to explain to a reasonable patient what this entails.”
Dr. Alam noted that companies might face extra hurdles in enrolling study participants and keeping them in the trials if the FDA seeks this kind of biopsy data. “But I don’t think inconvenience to the study sponsor is a good argument” for not seeking this kind of data, he added.
Leaving a loophole where certain kinds of clearly benign lesions don’t require a biopsy would eventually erode the quality of the research done on these devices. “That bar will be moved to accommodate the convenience of the sponsor, to make the study feasible,” Dr. Alam said. “And pretty soon, you’ll be missing a lot of patients that really should have biopsies.”
Acceptable rates of false positives, false negatives
The FDA panel chair noted that his colleagues had strongly urged review standards that would require that the devices improve on the rates of successful catches of suspicious lesions and lower false positives. But they did not endorse specific targets regarding the sensitivity and specificity rates.
“No one seems to be comfortable with providing or preordaining” these targets, Dr. Harris said.
Panelist Deneen Hesser, MSHSA, RN, urged a deep recognition of the power of a FDA clearance in the view of consumers.
“We need to be cognizant of what the term ‘FDA approved’ means to the lay individual,” said Ms. Hesser, who served as the patient representative on the panel. “A patient who sees that those tools are FDA approved will assume that each of those is the gold standard” in terms of expectations for delivering accurate results.
Like many of the panelists, Dr. Rotemberg urged the FDA to gather data about how patients would react to different messages encoded in consumer-oriented products.
“If the device says: ‘You should see a dermatologist for this’ and no other information, that’s very different from [saying]: ‘That lesion is suspicious for melanoma,’ ” Dr. Rotemberg said.
Despite the likely difficulties in conducting trials, the FDA needs to have the data to answer key questions about patient and physician reactions to readings from new tools, Dr. Rotemberg said.
“We don’t know how many additional biopsies we would cause with a specificity of 80%” for a new SLA tool, Dr. Rotemberg said, giving an example. “We don’t know how confident a dermatologist might be to say: ‘Actually, I’m not suspicious about that lesion and we can just fudge it or not biopsy it.’ We don’t know any of that until we study it in real life.”
The panelists also urged the FDA to seek to ensure that new tools used in analyzing skin lesions improve the quality of diagnosis.
Addressing equity
The FDA also asked the panel to weigh in on whether the agency should clear SLA tools in cases where the existing study data is drawn heavily from people considered to be at higher risk for skin cancer.
“To ensure generalizability across the entire U.S. population, should FDA require SLAs indicated for use beyond cancerous lesions be tested in a representative U.S. population?” the FDA asked.
The three most common skin cancers – melanoma, basal cell carcinoma, and squamous cell carcinoma – are more prevalent in people with Fitzpatrick I and II skin types, who tend to get sunburns, not tans. But people of color are more likely to develop melanoma in areas that are not sun exposed, such as the sole of the foot or under fingernails or toenails.
“Due in part to lower expected risk and screening, these melanomas are often detected late,” the FDA said in the briefing document.
There was broad consensus among panelists that the FDA should encourage companies to enroll people with all skin types and tones.
But they also looked for ways that the FDA could clear devices based on initial studies conducted largely with people considered to be at higher risk, with the agency then requiring follow-up trials to see how these products would work for the general U.S. population.
A version of this article first appeared on Medscape.com.
Avoiding harm in the diagnosis and treatment of food allergies
INDIANAPOLIS – If there’s one truth that David R. Stukus, MD, has come to realize from his 2 years as director of a food allergy treatment center, it’s that
“When they’re given a diagnosis of food allergy, many families do not receive proper education to help them understand the risk as well as self-management and prognosis,” he said at the annual meeting of the Society for Pediatric Dermatology. “They are left to fend for themselves, which leads to increased anxiety. If they don’t understand what it means to manage their child’s food allergy, they’re going to think that they’re a ticking time bomb,” said Dr. Stukus, director of the Food Allergy Treatment Center and professor of pediatrics in the division of allergy and immunology at Nationwide Children’s Hospital in Columbus, Ohio.
During his presentation, he toured clinicians through best practices to diagnose and treat food allergies and shared cautionary tales of unsupported claims, unnecessary testing, and potential harm to misdiagnosed patients.
While food allergies can be serious and life-threatening, they are also manageable, he continued. It doesn’t mean that children with food allergies can’t go to school, attend baseball games, or participate in activities that any other child would. “Telling someone to adopt a restricted diet is not a benign recommendation,” he said. “That can cause real harm.”
Dr. Stukus defined food allergy as an immunologic response to an allergen that results in reproducible symptoms with every exposure. “Most commonly we’re going to see IgE-mediated food allergies, which often occur within minutes of eating certain foods,” he said.
Food intolerance, on the other hand, is a nonimmunologic response to a food that causes gastrointestinal symptoms with exposure. “This can come and go over time,” he said. “The most common example is lactose intolerance.”
Then there’s food sensitivity, which Dr. Stukus said is not a medical term but a marketing term often applied to a variety of symptoms without evidence to support its use.
“On the Internet you will find many companies marketing food sensitivity tests,” he said. “Gluten-free foods are now a billion-dollar industry. There are no validated tests to diagnose food sensitivity. All the blood tests measure IgG, which is memory antibody. If you eat a food, it is a normal response to produce IgG to it, but these companies will test all these things and when it comes back elevated, they say ‘Aha! This is your food sensitivity and this is why you’re not sleeping well at night.’ ” To illustrate the harm that can come from food allergy tests he discussed a 6-year-old girl who presented to his clinic several years ago with typical symptoms of allergic rhinitis. The parent reported a history of sneezing around dogs, itchy, watery eyes in the spring, recurrent cough, and frequent upper respiratory infections.
The referring physician had ordered an allergy panel, which flagged a long list of foods that the girl was supposedly allergic to, including banana, egg white, cod, and peanut. “This family was told to take all of these foods out of her diet,” Dr. Stukus said. “Interestingly, she had been seen by this physician for evaluation of environmental allergies, but the only ones included in the test were cat, cockroach, dog, and dust mite. They didn’t even include the spring pollen allergies. You want to avoid tests like this.”
Food sensitization is not the same as food allergy, he continued, noting that about 30% of all children will have detectable IgE toward peanuts, milk, egg, and shrimp, but that only about 5% are truly allergic to those foods.
“If we go by IgE testing alone, we’re going to overdiagnose the vast majority of people with food allergies that they don’t actually have,” he said. “Food allergy is diagnosed by the history and then confirmed by testing. With IgE-mediated food allergies we know that milk, egg, wheat, soy, finned fish, shellfish, and peanuts account for more than 90% of all food allergy reactions. Can any food potentially cause a food allergy? Yes, potentially, but we know that most fruits and vegetables and grains are very unlikely to cause an allergy.”
IgE-mediated food allergies are objective, immediate onset, and reproducible with every exposure to the offending food, no matter what form. Typical symptoms include hives, swelling, vomiting, runny nose/congestion, wheezing, hypotension, and anaphylaxis.
“We can also accurately identify infants that are more at risk to develop food allergies,” Dr. Stukus said. Infants with refractory atopic dermatitis often progress from eczema to food allergies to allergic rhinitis and asthma, the so-called “allergic march.” “Family history does have a role as well, but it’s not as significant,” he said. As for diagnostic tools, skin prick testing detects the presence of specific IgE bound to cutaneous mast cells and has a high negative predictive value and a low positive predictive value (around 50%).
With serum-specific IgE testing, levels of IgE for food and/or inhalant allergen can be obtained conveniently through routine venipuncture. Results are reported in ranges from 0.1 kU/L to 100 kU/L, and some are reported as arbitrary classes in levels of severity from 1 to 5.
“I highly discourage anybody from paying attention to arbitrary classes [on these reports],” Dr. Stukus said. “Those are meaningless. The absolute value is all that matters.”
He added that both skin and blood testing have high rates of false positive results. “We really need to use the history to help guide what tests we do; they were never designed to be used as screening tests, yet they’re used as screening tests on a regular basis,” he said. “There is also no indication to do shotgun testing. The reason why is because we see lots of cross reactivity on testing. If we have someone with peanut allergy and we start doing specific IgE testing for all legumes, more often than not we’re going to find detectable IgE, but it’s much less likely that they actually have clinical reactivity to foods like soy and beans.”
Dr. Stukus advises clinicians to consider certain questions before they order an allergen panel, the first being: Do I have the knowledge and experience to properly interpret the results?
“If you don’t know how to interpret the test, you probably shouldn’t order it in the first place,” he said. “If you do have the knowledge to interpret the results, will the results help to determine the diagnosis or change management? If not, why are you testing just to test? There is zero clinical indication to order a food allergy panel.” Dr. Stukus recommended a review of unproven tests for adverse reactions to foods published in 2018 in The Journal of Allergy and Clinical Immunology.
According to Dr. Stukus, potential harms from unproven food allergy tests include cost, unnecessary dietary avoidance, and a delay in diagnosis for the underlying condition. During the COVID-19 pandemic, he observed an increase in the number of patients with orthorexia, which he described as an eating disorder characterized by having an unsafe obsession with healthy food that becomes deeply rooted in the individual’s way of thinking to the point that it interferes with daily life.
“If you take someone who has anxiety at baseline, and then you give them a list of foods that they allegedly can’t eat, that’s going to cause worse anxiety,” he added. “We’re seeing that from the results of these tests.”
Dr. Stukus disclosed that he is a consultant for Before Brands, Kaleo, and Novartis. He is also associate editor of the Annals of Allergy, Asthma and Immunology.
INDIANAPOLIS – If there’s one truth that David R. Stukus, MD, has come to realize from his 2 years as director of a food allergy treatment center, it’s that
“When they’re given a diagnosis of food allergy, many families do not receive proper education to help them understand the risk as well as self-management and prognosis,” he said at the annual meeting of the Society for Pediatric Dermatology. “They are left to fend for themselves, which leads to increased anxiety. If they don’t understand what it means to manage their child’s food allergy, they’re going to think that they’re a ticking time bomb,” said Dr. Stukus, director of the Food Allergy Treatment Center and professor of pediatrics in the division of allergy and immunology at Nationwide Children’s Hospital in Columbus, Ohio.
During his presentation, he toured clinicians through best practices to diagnose and treat food allergies and shared cautionary tales of unsupported claims, unnecessary testing, and potential harm to misdiagnosed patients.
While food allergies can be serious and life-threatening, they are also manageable, he continued. It doesn’t mean that children with food allergies can’t go to school, attend baseball games, or participate in activities that any other child would. “Telling someone to adopt a restricted diet is not a benign recommendation,” he said. “That can cause real harm.”
Dr. Stukus defined food allergy as an immunologic response to an allergen that results in reproducible symptoms with every exposure. “Most commonly we’re going to see IgE-mediated food allergies, which often occur within minutes of eating certain foods,” he said.
Food intolerance, on the other hand, is a nonimmunologic response to a food that causes gastrointestinal symptoms with exposure. “This can come and go over time,” he said. “The most common example is lactose intolerance.”
Then there’s food sensitivity, which Dr. Stukus said is not a medical term but a marketing term often applied to a variety of symptoms without evidence to support its use.
“On the Internet you will find many companies marketing food sensitivity tests,” he said. “Gluten-free foods are now a billion-dollar industry. There are no validated tests to diagnose food sensitivity. All the blood tests measure IgG, which is memory antibody. If you eat a food, it is a normal response to produce IgG to it, but these companies will test all these things and when it comes back elevated, they say ‘Aha! This is your food sensitivity and this is why you’re not sleeping well at night.’ ” To illustrate the harm that can come from food allergy tests he discussed a 6-year-old girl who presented to his clinic several years ago with typical symptoms of allergic rhinitis. The parent reported a history of sneezing around dogs, itchy, watery eyes in the spring, recurrent cough, and frequent upper respiratory infections.
The referring physician had ordered an allergy panel, which flagged a long list of foods that the girl was supposedly allergic to, including banana, egg white, cod, and peanut. “This family was told to take all of these foods out of her diet,” Dr. Stukus said. “Interestingly, she had been seen by this physician for evaluation of environmental allergies, but the only ones included in the test were cat, cockroach, dog, and dust mite. They didn’t even include the spring pollen allergies. You want to avoid tests like this.”
Food sensitization is not the same as food allergy, he continued, noting that about 30% of all children will have detectable IgE toward peanuts, milk, egg, and shrimp, but that only about 5% are truly allergic to those foods.
“If we go by IgE testing alone, we’re going to overdiagnose the vast majority of people with food allergies that they don’t actually have,” he said. “Food allergy is diagnosed by the history and then confirmed by testing. With IgE-mediated food allergies we know that milk, egg, wheat, soy, finned fish, shellfish, and peanuts account for more than 90% of all food allergy reactions. Can any food potentially cause a food allergy? Yes, potentially, but we know that most fruits and vegetables and grains are very unlikely to cause an allergy.”
IgE-mediated food allergies are objective, immediate onset, and reproducible with every exposure to the offending food, no matter what form. Typical symptoms include hives, swelling, vomiting, runny nose/congestion, wheezing, hypotension, and anaphylaxis.
“We can also accurately identify infants that are more at risk to develop food allergies,” Dr. Stukus said. Infants with refractory atopic dermatitis often progress from eczema to food allergies to allergic rhinitis and asthma, the so-called “allergic march.” “Family history does have a role as well, but it’s not as significant,” he said. As for diagnostic tools, skin prick testing detects the presence of specific IgE bound to cutaneous mast cells and has a high negative predictive value and a low positive predictive value (around 50%).
With serum-specific IgE testing, levels of IgE for food and/or inhalant allergen can be obtained conveniently through routine venipuncture. Results are reported in ranges from 0.1 kU/L to 100 kU/L, and some are reported as arbitrary classes in levels of severity from 1 to 5.
“I highly discourage anybody from paying attention to arbitrary classes [on these reports],” Dr. Stukus said. “Those are meaningless. The absolute value is all that matters.”
He added that both skin and blood testing have high rates of false positive results. “We really need to use the history to help guide what tests we do; they were never designed to be used as screening tests, yet they’re used as screening tests on a regular basis,” he said. “There is also no indication to do shotgun testing. The reason why is because we see lots of cross reactivity on testing. If we have someone with peanut allergy and we start doing specific IgE testing for all legumes, more often than not we’re going to find detectable IgE, but it’s much less likely that they actually have clinical reactivity to foods like soy and beans.”
Dr. Stukus advises clinicians to consider certain questions before they order an allergen panel, the first being: Do I have the knowledge and experience to properly interpret the results?
“If you don’t know how to interpret the test, you probably shouldn’t order it in the first place,” he said. “If you do have the knowledge to interpret the results, will the results help to determine the diagnosis or change management? If not, why are you testing just to test? There is zero clinical indication to order a food allergy panel.” Dr. Stukus recommended a review of unproven tests for adverse reactions to foods published in 2018 in The Journal of Allergy and Clinical Immunology.
According to Dr. Stukus, potential harms from unproven food allergy tests include cost, unnecessary dietary avoidance, and a delay in diagnosis for the underlying condition. During the COVID-19 pandemic, he observed an increase in the number of patients with orthorexia, which he described as an eating disorder characterized by having an unsafe obsession with healthy food that becomes deeply rooted in the individual’s way of thinking to the point that it interferes with daily life.
“If you take someone who has anxiety at baseline, and then you give them a list of foods that they allegedly can’t eat, that’s going to cause worse anxiety,” he added. “We’re seeing that from the results of these tests.”
Dr. Stukus disclosed that he is a consultant for Before Brands, Kaleo, and Novartis. He is also associate editor of the Annals of Allergy, Asthma and Immunology.
INDIANAPOLIS – If there’s one truth that David R. Stukus, MD, has come to realize from his 2 years as director of a food allergy treatment center, it’s that
“When they’re given a diagnosis of food allergy, many families do not receive proper education to help them understand the risk as well as self-management and prognosis,” he said at the annual meeting of the Society for Pediatric Dermatology. “They are left to fend for themselves, which leads to increased anxiety. If they don’t understand what it means to manage their child’s food allergy, they’re going to think that they’re a ticking time bomb,” said Dr. Stukus, director of the Food Allergy Treatment Center and professor of pediatrics in the division of allergy and immunology at Nationwide Children’s Hospital in Columbus, Ohio.
During his presentation, he toured clinicians through best practices to diagnose and treat food allergies and shared cautionary tales of unsupported claims, unnecessary testing, and potential harm to misdiagnosed patients.
While food allergies can be serious and life-threatening, they are also manageable, he continued. It doesn’t mean that children with food allergies can’t go to school, attend baseball games, or participate in activities that any other child would. “Telling someone to adopt a restricted diet is not a benign recommendation,” he said. “That can cause real harm.”
Dr. Stukus defined food allergy as an immunologic response to an allergen that results in reproducible symptoms with every exposure. “Most commonly we’re going to see IgE-mediated food allergies, which often occur within minutes of eating certain foods,” he said.
Food intolerance, on the other hand, is a nonimmunologic response to a food that causes gastrointestinal symptoms with exposure. “This can come and go over time,” he said. “The most common example is lactose intolerance.”
Then there’s food sensitivity, which Dr. Stukus said is not a medical term but a marketing term often applied to a variety of symptoms without evidence to support its use.
“On the Internet you will find many companies marketing food sensitivity tests,” he said. “Gluten-free foods are now a billion-dollar industry. There are no validated tests to diagnose food sensitivity. All the blood tests measure IgG, which is memory antibody. If you eat a food, it is a normal response to produce IgG to it, but these companies will test all these things and when it comes back elevated, they say ‘Aha! This is your food sensitivity and this is why you’re not sleeping well at night.’ ” To illustrate the harm that can come from food allergy tests he discussed a 6-year-old girl who presented to his clinic several years ago with typical symptoms of allergic rhinitis. The parent reported a history of sneezing around dogs, itchy, watery eyes in the spring, recurrent cough, and frequent upper respiratory infections.
The referring physician had ordered an allergy panel, which flagged a long list of foods that the girl was supposedly allergic to, including banana, egg white, cod, and peanut. “This family was told to take all of these foods out of her diet,” Dr. Stukus said. “Interestingly, she had been seen by this physician for evaluation of environmental allergies, but the only ones included in the test were cat, cockroach, dog, and dust mite. They didn’t even include the spring pollen allergies. You want to avoid tests like this.”
Food sensitization is not the same as food allergy, he continued, noting that about 30% of all children will have detectable IgE toward peanuts, milk, egg, and shrimp, but that only about 5% are truly allergic to those foods.
“If we go by IgE testing alone, we’re going to overdiagnose the vast majority of people with food allergies that they don’t actually have,” he said. “Food allergy is diagnosed by the history and then confirmed by testing. With IgE-mediated food allergies we know that milk, egg, wheat, soy, finned fish, shellfish, and peanuts account for more than 90% of all food allergy reactions. Can any food potentially cause a food allergy? Yes, potentially, but we know that most fruits and vegetables and grains are very unlikely to cause an allergy.”
IgE-mediated food allergies are objective, immediate onset, and reproducible with every exposure to the offending food, no matter what form. Typical symptoms include hives, swelling, vomiting, runny nose/congestion, wheezing, hypotension, and anaphylaxis.
“We can also accurately identify infants that are more at risk to develop food allergies,” Dr. Stukus said. Infants with refractory atopic dermatitis often progress from eczema to food allergies to allergic rhinitis and asthma, the so-called “allergic march.” “Family history does have a role as well, but it’s not as significant,” he said. As for diagnostic tools, skin prick testing detects the presence of specific IgE bound to cutaneous mast cells and has a high negative predictive value and a low positive predictive value (around 50%).
With serum-specific IgE testing, levels of IgE for food and/or inhalant allergen can be obtained conveniently through routine venipuncture. Results are reported in ranges from 0.1 kU/L to 100 kU/L, and some are reported as arbitrary classes in levels of severity from 1 to 5.
“I highly discourage anybody from paying attention to arbitrary classes [on these reports],” Dr. Stukus said. “Those are meaningless. The absolute value is all that matters.”
He added that both skin and blood testing have high rates of false positive results. “We really need to use the history to help guide what tests we do; they were never designed to be used as screening tests, yet they’re used as screening tests on a regular basis,” he said. “There is also no indication to do shotgun testing. The reason why is because we see lots of cross reactivity on testing. If we have someone with peanut allergy and we start doing specific IgE testing for all legumes, more often than not we’re going to find detectable IgE, but it’s much less likely that they actually have clinical reactivity to foods like soy and beans.”
Dr. Stukus advises clinicians to consider certain questions before they order an allergen panel, the first being: Do I have the knowledge and experience to properly interpret the results?
“If you don’t know how to interpret the test, you probably shouldn’t order it in the first place,” he said. “If you do have the knowledge to interpret the results, will the results help to determine the diagnosis or change management? If not, why are you testing just to test? There is zero clinical indication to order a food allergy panel.” Dr. Stukus recommended a review of unproven tests for adverse reactions to foods published in 2018 in The Journal of Allergy and Clinical Immunology.
According to Dr. Stukus, potential harms from unproven food allergy tests include cost, unnecessary dietary avoidance, and a delay in diagnosis for the underlying condition. During the COVID-19 pandemic, he observed an increase in the number of patients with orthorexia, which he described as an eating disorder characterized by having an unsafe obsession with healthy food that becomes deeply rooted in the individual’s way of thinking to the point that it interferes with daily life.
“If you take someone who has anxiety at baseline, and then you give them a list of foods that they allegedly can’t eat, that’s going to cause worse anxiety,” he added. “We’re seeing that from the results of these tests.”
Dr. Stukus disclosed that he is a consultant for Before Brands, Kaleo, and Novartis. He is also associate editor of the Annals of Allergy, Asthma and Immunology.
AT SPD 2022
Aesthetics abound for the aging face
At the MedscapeLive’s Women’s and Pediatric Dermatology Seminar, Jacqueline Watchmaker, MD, a dermatologist in Scottsdale, Ariz., provided an overview of current options, along with advice on how to keep patients’ expectations realistic and how to properly choose the best candidates for the best procedures.
“One of the most common concerns patients come to me with are wrinkles on the upper face,” but this is far from their only concern, Dr. Watchmaker said. Wrinkles and sagging of the lower face, areas under the eyes, nasolabial folds, marionette lines, and the neck also draw concern. Uneven coloration is another common concern, she said.
“So, what can we do for all of this?” she asked. The options are plentiful. Wrinkles of the upper face are easy to address with neuromodulators, she said, and soft-tissue fillers help the jawline and cheek areas.
“For the lower face, skin tightening devices really shine,” she added. And lasers can help correct uneven coloration. Surgery, of course, can also produce good results, but many patients want to stick with noninvasive or minimally invasive procedures.
Case: 83-year-old woman
Dr. Watchmaker discussed an 83-year old patient, who had malar mounds and accentuation of the infraorbital hollowness resulting from changes in subcutaneous fat and ligament laxity. She also had uneven coloration from photo damage, wrinkles on the upper face, linear appearance of zygoma related to underlying bony changes and fat compartment descent, and nasolabial folds and jowls related to decreased bony compartments, ligament laxity, and shifting of fat. She was naive to any cosmetic procedure.
Despite her age, this patient had no wrinkling on the upper forehead. Dr. Watchmaker did not inject neuromodulator in the upper forehead, as this patient also had a slightly heavy eyelid. “If you inject too much, it can cause some drooping of the eyelid and eyebrow,” she said.
For filler, she used a combination of high G (firmness, support) hyaluronic acid filler, a medium G acid filler, and a low G filler. The result: The woman’s face became more balanced, the mid-face volumization lifted the lower face, and the glabellar and periocular lines were softer, although still present. “It’s important to counsel patients that neuromodulators won’t make the lines go away the first time, but they will be softened.”
Practice tips
It’s important to titrate neuromodulators to fit the patient, Dr. Watchmaker said. Ask: What are their goals: Reversal of static lines? Softening wrinkles? Maintaining current status? “There’s not one dosing regimen,” and both dosing and frequency of neuromodulators can be titrated to fit each patient’s aesthetic goals, she said. For older patients who want to soften or maintain appearance, she suggested treatment every 4-6 months. And some patients just want to maintain the status quo, she noted.
Ideal candidates
For neuromodulators and fillers, who is an ideal candidate? “I think it’s anyone who has realistic expectations,” she said. Patients need to know how many treatments are needed and how much it will cost. For patients with extensive wrinkling and sagging, she said, she does extensive counseling about what results to expect “because I don’t want them to feel like they wasted their time or their money.”
She also suggests a surgical consult, as some may opt for that route after learning about the options and expected results.
Skin tightening
Both radiofrequency and microfocused ultrasound are noninvasive and additional options. Radiofrequency uses radio waves, with electromagnetic energy to stimulate heat. Ultrasound uses ultrasound waves to stimulate heat. Both approaches cause collagen contraction, neocollagenesis, and skin tightening.
These procedures do well for the lower face, Dr. Watchmaker said, but “I am relatively unimpressed for how well they do for the upper face.” Ideal candidates have mild to moderate skin laxity and want to avoid surgery. She also tells patients that collagen isn’t made overnight. “You won’t see much for 3-6 months after.” The good news? Usually the treatments need to be repeated only every 1.5-2 years, she said.
Lasers
“There are so many lasers out there,” said Dr. Watchmaker, who groups them into three categories: those used for wrinkles, dyschromia, and erythema. Her picks: ablative lasers (CO2 and erbium) and erbium-doped YAG 1550 nm laser for rhytids. Thulium 1927 and QS and picosecond lasers are her picks for dyschromia, and for erythema, pulsed dye and KTP lasers.
Some laser treatments are not a “walk in the park,” as she warns patients. For example, after treatment with ablative lasers, there is pain, post-procedure redness, and crusting.
Take-home points
A combination of noninvasive and minimally invasive procedures can produce appearance-improving results. That’s more likely if dermatologists choose ideal candidates, personalize the treatment, and set realistic expectations. “We have a finite number of tools,” she said, but they can be used in a variety of ways.
At the interactive panel discussion following her presentation, Dr. Watchmaker was asked what she tells patients about sun protection. “I talk a lot about sunscreens,’’ she said, always urging patients to use them. While the options for rejuvenation are numerous, taking care of the skin is still crucial.
Dr. Watchmaker had no disclosures. MedscapeLive and this news organization are owned by the same parent company.
At the MedscapeLive’s Women’s and Pediatric Dermatology Seminar, Jacqueline Watchmaker, MD, a dermatologist in Scottsdale, Ariz., provided an overview of current options, along with advice on how to keep patients’ expectations realistic and how to properly choose the best candidates for the best procedures.
“One of the most common concerns patients come to me with are wrinkles on the upper face,” but this is far from their only concern, Dr. Watchmaker said. Wrinkles and sagging of the lower face, areas under the eyes, nasolabial folds, marionette lines, and the neck also draw concern. Uneven coloration is another common concern, she said.
“So, what can we do for all of this?” she asked. The options are plentiful. Wrinkles of the upper face are easy to address with neuromodulators, she said, and soft-tissue fillers help the jawline and cheek areas.
“For the lower face, skin tightening devices really shine,” she added. And lasers can help correct uneven coloration. Surgery, of course, can also produce good results, but many patients want to stick with noninvasive or minimally invasive procedures.
Case: 83-year-old woman
Dr. Watchmaker discussed an 83-year old patient, who had malar mounds and accentuation of the infraorbital hollowness resulting from changes in subcutaneous fat and ligament laxity. She also had uneven coloration from photo damage, wrinkles on the upper face, linear appearance of zygoma related to underlying bony changes and fat compartment descent, and nasolabial folds and jowls related to decreased bony compartments, ligament laxity, and shifting of fat. She was naive to any cosmetic procedure.
Despite her age, this patient had no wrinkling on the upper forehead. Dr. Watchmaker did not inject neuromodulator in the upper forehead, as this patient also had a slightly heavy eyelid. “If you inject too much, it can cause some drooping of the eyelid and eyebrow,” she said.
For filler, she used a combination of high G (firmness, support) hyaluronic acid filler, a medium G acid filler, and a low G filler. The result: The woman’s face became more balanced, the mid-face volumization lifted the lower face, and the glabellar and periocular lines were softer, although still present. “It’s important to counsel patients that neuromodulators won’t make the lines go away the first time, but they will be softened.”
Practice tips
It’s important to titrate neuromodulators to fit the patient, Dr. Watchmaker said. Ask: What are their goals: Reversal of static lines? Softening wrinkles? Maintaining current status? “There’s not one dosing regimen,” and both dosing and frequency of neuromodulators can be titrated to fit each patient’s aesthetic goals, she said. For older patients who want to soften or maintain appearance, she suggested treatment every 4-6 months. And some patients just want to maintain the status quo, she noted.
Ideal candidates
For neuromodulators and fillers, who is an ideal candidate? “I think it’s anyone who has realistic expectations,” she said. Patients need to know how many treatments are needed and how much it will cost. For patients with extensive wrinkling and sagging, she said, she does extensive counseling about what results to expect “because I don’t want them to feel like they wasted their time or their money.”
She also suggests a surgical consult, as some may opt for that route after learning about the options and expected results.
Skin tightening
Both radiofrequency and microfocused ultrasound are noninvasive and additional options. Radiofrequency uses radio waves, with electromagnetic energy to stimulate heat. Ultrasound uses ultrasound waves to stimulate heat. Both approaches cause collagen contraction, neocollagenesis, and skin tightening.
These procedures do well for the lower face, Dr. Watchmaker said, but “I am relatively unimpressed for how well they do for the upper face.” Ideal candidates have mild to moderate skin laxity and want to avoid surgery. She also tells patients that collagen isn’t made overnight. “You won’t see much for 3-6 months after.” The good news? Usually the treatments need to be repeated only every 1.5-2 years, she said.
Lasers
“There are so many lasers out there,” said Dr. Watchmaker, who groups them into three categories: those used for wrinkles, dyschromia, and erythema. Her picks: ablative lasers (CO2 and erbium) and erbium-doped YAG 1550 nm laser for rhytids. Thulium 1927 and QS and picosecond lasers are her picks for dyschromia, and for erythema, pulsed dye and KTP lasers.
Some laser treatments are not a “walk in the park,” as she warns patients. For example, after treatment with ablative lasers, there is pain, post-procedure redness, and crusting.
Take-home points
A combination of noninvasive and minimally invasive procedures can produce appearance-improving results. That’s more likely if dermatologists choose ideal candidates, personalize the treatment, and set realistic expectations. “We have a finite number of tools,” she said, but they can be used in a variety of ways.
At the interactive panel discussion following her presentation, Dr. Watchmaker was asked what she tells patients about sun protection. “I talk a lot about sunscreens,’’ she said, always urging patients to use them. While the options for rejuvenation are numerous, taking care of the skin is still crucial.
Dr. Watchmaker had no disclosures. MedscapeLive and this news organization are owned by the same parent company.
At the MedscapeLive’s Women’s and Pediatric Dermatology Seminar, Jacqueline Watchmaker, MD, a dermatologist in Scottsdale, Ariz., provided an overview of current options, along with advice on how to keep patients’ expectations realistic and how to properly choose the best candidates for the best procedures.
“One of the most common concerns patients come to me with are wrinkles on the upper face,” but this is far from their only concern, Dr. Watchmaker said. Wrinkles and sagging of the lower face, areas under the eyes, nasolabial folds, marionette lines, and the neck also draw concern. Uneven coloration is another common concern, she said.
“So, what can we do for all of this?” she asked. The options are plentiful. Wrinkles of the upper face are easy to address with neuromodulators, she said, and soft-tissue fillers help the jawline and cheek areas.
“For the lower face, skin tightening devices really shine,” she added. And lasers can help correct uneven coloration. Surgery, of course, can also produce good results, but many patients want to stick with noninvasive or minimally invasive procedures.
Case: 83-year-old woman
Dr. Watchmaker discussed an 83-year old patient, who had malar mounds and accentuation of the infraorbital hollowness resulting from changes in subcutaneous fat and ligament laxity. She also had uneven coloration from photo damage, wrinkles on the upper face, linear appearance of zygoma related to underlying bony changes and fat compartment descent, and nasolabial folds and jowls related to decreased bony compartments, ligament laxity, and shifting of fat. She was naive to any cosmetic procedure.
Despite her age, this patient had no wrinkling on the upper forehead. Dr. Watchmaker did not inject neuromodulator in the upper forehead, as this patient also had a slightly heavy eyelid. “If you inject too much, it can cause some drooping of the eyelid and eyebrow,” she said.
For filler, she used a combination of high G (firmness, support) hyaluronic acid filler, a medium G acid filler, and a low G filler. The result: The woman’s face became more balanced, the mid-face volumization lifted the lower face, and the glabellar and periocular lines were softer, although still present. “It’s important to counsel patients that neuromodulators won’t make the lines go away the first time, but they will be softened.”
Practice tips
It’s important to titrate neuromodulators to fit the patient, Dr. Watchmaker said. Ask: What are their goals: Reversal of static lines? Softening wrinkles? Maintaining current status? “There’s not one dosing regimen,” and both dosing and frequency of neuromodulators can be titrated to fit each patient’s aesthetic goals, she said. For older patients who want to soften or maintain appearance, she suggested treatment every 4-6 months. And some patients just want to maintain the status quo, she noted.
Ideal candidates
For neuromodulators and fillers, who is an ideal candidate? “I think it’s anyone who has realistic expectations,” she said. Patients need to know how many treatments are needed and how much it will cost. For patients with extensive wrinkling and sagging, she said, she does extensive counseling about what results to expect “because I don’t want them to feel like they wasted their time or their money.”
She also suggests a surgical consult, as some may opt for that route after learning about the options and expected results.
Skin tightening
Both radiofrequency and microfocused ultrasound are noninvasive and additional options. Radiofrequency uses radio waves, with electromagnetic energy to stimulate heat. Ultrasound uses ultrasound waves to stimulate heat. Both approaches cause collagen contraction, neocollagenesis, and skin tightening.
These procedures do well for the lower face, Dr. Watchmaker said, but “I am relatively unimpressed for how well they do for the upper face.” Ideal candidates have mild to moderate skin laxity and want to avoid surgery. She also tells patients that collagen isn’t made overnight. “You won’t see much for 3-6 months after.” The good news? Usually the treatments need to be repeated only every 1.5-2 years, she said.
Lasers
“There are so many lasers out there,” said Dr. Watchmaker, who groups them into three categories: those used for wrinkles, dyschromia, and erythema. Her picks: ablative lasers (CO2 and erbium) and erbium-doped YAG 1550 nm laser for rhytids. Thulium 1927 and QS and picosecond lasers are her picks for dyschromia, and for erythema, pulsed dye and KTP lasers.
Some laser treatments are not a “walk in the park,” as she warns patients. For example, after treatment with ablative lasers, there is pain, post-procedure redness, and crusting.
Take-home points
A combination of noninvasive and minimally invasive procedures can produce appearance-improving results. That’s more likely if dermatologists choose ideal candidates, personalize the treatment, and set realistic expectations. “We have a finite number of tools,” she said, but they can be used in a variety of ways.
At the interactive panel discussion following her presentation, Dr. Watchmaker was asked what she tells patients about sun protection. “I talk a lot about sunscreens,’’ she said, always urging patients to use them. While the options for rejuvenation are numerous, taking care of the skin is still crucial.
Dr. Watchmaker had no disclosures. MedscapeLive and this news organization are owned by the same parent company.
FROM MEDSCAPELIVE WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
U.S. clears 786,000 monkeypox vaccine doses for distribution
More than 780,000 doses of the JYNNEOS monkeypox vaccine will be available in the United States beginning July 29, the Department of Health & Human Services announced on July 28 in a press call.
HHS Secretary Xavier Becerra urged local and state public health departments to use these doses for preventive vaccination efforts to stay ahead of the virus and end the outbreak, noting that the HHS and Centers for Disease Control and Prevention do not control how vaccines are distributed at state and local levels. “We don’t have the authority to tell them what to do,” he said during the call. “We need them to work with us.”
As of July 28, there were 4,907 reported cases of monkeypox in the United States and officials expect cases will continue to rise in the coming weeks.
The vaccine is manufactured by the small Danish company Bavarian Nordic. These additional 786,000 doses were previously stored at a plant in Denmark, awaiting the completion of an inspection and authorization of the vaccine plant by the Food and Drug Administration. The agency announced on July 27 that both the vaccine doses and the manufacturing plant met standards.
With the announcement of these additional doses, the vaccine allocation plan is also being updated to take into account two important factors: the number of people at high risk in a jurisdiction and the number of new cases reported since the last vaccine allocation.
“This update gives greater weight to prioritizing vaccines to areas with the greatest number of people at risk, which includes men who have sex with men who have HIV or who are eligible for HIV pre-exposure prophylaxis, while still considering where we are seeing cases increase,” said Capt. Jennifer McQuiston, DVM, deputy director of the division of high consequence pathogens and pathology at the CDC.
Capt.McQuiston also provided additional demographic information on the U.S. outbreak. The median age of people with confirmed cases is 35 years old, with a range from 17 to 76. (This does not include the two cases in children reported on July 22.) Of the cases where sex at birth was provided, 99% were individuals assigned male sex at birth. In cases with reported ethnicity and race, 37% were non-Hispanic White people, 31% were Hispanic/Latino, 27% were Black or African American, and 4% were of Asian descent. The most common symptoms were rash – present in 99% of cases – malaise, fever, and swollen lymph nodes.
HHS and CDC did not have data on how many people have received at least one dose of the monkeypox vaccine. When asked how many people need to be fully vaccinated against monkeypox to contain the outbreak, Mr. Becerra did not provide an estimate but implied that preventive vaccination could help limit the number of vaccines needed and expressed optimism about quelling the outbreak in the United States. “We believe that we have done everything we can at the federal level to work with our state and local partners and communities affected to make sure we can stay ahead of this and end this outbreak,” he said, “but everybody’s got to do their part.”
A version of this article first appeared on Medscape.com.
More than 780,000 doses of the JYNNEOS monkeypox vaccine will be available in the United States beginning July 29, the Department of Health & Human Services announced on July 28 in a press call.
HHS Secretary Xavier Becerra urged local and state public health departments to use these doses for preventive vaccination efforts to stay ahead of the virus and end the outbreak, noting that the HHS and Centers for Disease Control and Prevention do not control how vaccines are distributed at state and local levels. “We don’t have the authority to tell them what to do,” he said during the call. “We need them to work with us.”
As of July 28, there were 4,907 reported cases of monkeypox in the United States and officials expect cases will continue to rise in the coming weeks.
The vaccine is manufactured by the small Danish company Bavarian Nordic. These additional 786,000 doses were previously stored at a plant in Denmark, awaiting the completion of an inspection and authorization of the vaccine plant by the Food and Drug Administration. The agency announced on July 27 that both the vaccine doses and the manufacturing plant met standards.
With the announcement of these additional doses, the vaccine allocation plan is also being updated to take into account two important factors: the number of people at high risk in a jurisdiction and the number of new cases reported since the last vaccine allocation.
“This update gives greater weight to prioritizing vaccines to areas with the greatest number of people at risk, which includes men who have sex with men who have HIV or who are eligible for HIV pre-exposure prophylaxis, while still considering where we are seeing cases increase,” said Capt. Jennifer McQuiston, DVM, deputy director of the division of high consequence pathogens and pathology at the CDC.
Capt.McQuiston also provided additional demographic information on the U.S. outbreak. The median age of people with confirmed cases is 35 years old, with a range from 17 to 76. (This does not include the two cases in children reported on July 22.) Of the cases where sex at birth was provided, 99% were individuals assigned male sex at birth. In cases with reported ethnicity and race, 37% were non-Hispanic White people, 31% were Hispanic/Latino, 27% were Black or African American, and 4% were of Asian descent. The most common symptoms were rash – present in 99% of cases – malaise, fever, and swollen lymph nodes.
HHS and CDC did not have data on how many people have received at least one dose of the monkeypox vaccine. When asked how many people need to be fully vaccinated against monkeypox to contain the outbreak, Mr. Becerra did not provide an estimate but implied that preventive vaccination could help limit the number of vaccines needed and expressed optimism about quelling the outbreak in the United States. “We believe that we have done everything we can at the federal level to work with our state and local partners and communities affected to make sure we can stay ahead of this and end this outbreak,” he said, “but everybody’s got to do their part.”
A version of this article first appeared on Medscape.com.
More than 780,000 doses of the JYNNEOS monkeypox vaccine will be available in the United States beginning July 29, the Department of Health & Human Services announced on July 28 in a press call.
HHS Secretary Xavier Becerra urged local and state public health departments to use these doses for preventive vaccination efforts to stay ahead of the virus and end the outbreak, noting that the HHS and Centers for Disease Control and Prevention do not control how vaccines are distributed at state and local levels. “We don’t have the authority to tell them what to do,” he said during the call. “We need them to work with us.”
As of July 28, there were 4,907 reported cases of monkeypox in the United States and officials expect cases will continue to rise in the coming weeks.
The vaccine is manufactured by the small Danish company Bavarian Nordic. These additional 786,000 doses were previously stored at a plant in Denmark, awaiting the completion of an inspection and authorization of the vaccine plant by the Food and Drug Administration. The agency announced on July 27 that both the vaccine doses and the manufacturing plant met standards.
With the announcement of these additional doses, the vaccine allocation plan is also being updated to take into account two important factors: the number of people at high risk in a jurisdiction and the number of new cases reported since the last vaccine allocation.
“This update gives greater weight to prioritizing vaccines to areas with the greatest number of people at risk, which includes men who have sex with men who have HIV or who are eligible for HIV pre-exposure prophylaxis, while still considering where we are seeing cases increase,” said Capt. Jennifer McQuiston, DVM, deputy director of the division of high consequence pathogens and pathology at the CDC.
Capt.McQuiston also provided additional demographic information on the U.S. outbreak. The median age of people with confirmed cases is 35 years old, with a range from 17 to 76. (This does not include the two cases in children reported on July 22.) Of the cases where sex at birth was provided, 99% were individuals assigned male sex at birth. In cases with reported ethnicity and race, 37% were non-Hispanic White people, 31% were Hispanic/Latino, 27% were Black or African American, and 4% were of Asian descent. The most common symptoms were rash – present in 99% of cases – malaise, fever, and swollen lymph nodes.
HHS and CDC did not have data on how many people have received at least one dose of the monkeypox vaccine. When asked how many people need to be fully vaccinated against monkeypox to contain the outbreak, Mr. Becerra did not provide an estimate but implied that preventive vaccination could help limit the number of vaccines needed and expressed optimism about quelling the outbreak in the United States. “We believe that we have done everything we can at the federal level to work with our state and local partners and communities affected to make sure we can stay ahead of this and end this outbreak,” he said, “but everybody’s got to do their part.”
A version of this article first appeared on Medscape.com.
Potentially deadly bacteria detected in U.S. soil
, according to a new alert from the Centers for Disease Control and Prevention.
The bacteria, Burkholderia pseudomallei, was found along the Gulf Coast region in southern Mississippi. Typically, the bacteria are in tropical and subtropical climates, especially in parts of Southeast Asia, northern Australia, Central America, South America, Puerto Rico, and the U.S. Virgin Islands.
The bacteria can cause melioidosis, a rare and serious infectious disease that spreads to animals and humans through contact with contaminated soil and water via cuts, wounds, mucous membranes, breathing the bacteria in, or eating or drinking it. Worldwide, the disease is fatal in 10%-50% of those who become infected.
CDC and state officials are investigating the samples to find out how widespread the bacteria are within the United States. So far, modeling suggests that the environmental conditions on the Gulf Coast support the growth of B. pseudomallei.
“It is unclear how long the bacteria has been in the environment and where else it might be found in the U.S.,” according to the CDC statement. “CDC is alerting clinicians throughout the country of this discovery through a national health advisory, reminding them to be aware of the signs and symptoms of melioidosis and to consider melioidosis in patients that present with symptoms of the disease.”
Two unrelated people who live near the Gulf Coast region of Mississippi became sick with melioidosis recently – one in July 2020 and one in May 2022. Neither had traveled outside of the United States. The cases led the CDC and the Mississippi State Department of Health to collect environmental samples and test household products at the patients’ homes in June 2022. Three of the samples taken from soil and puddle water in the 2020 case tested positive for the bacteria.
Genomic sequencing revealed that both patients were infected with the same strain of the bacteria from the Western Hemisphere. They were hospitalized with sepsis due to pneumonia and had known risk factors for melioidosis. Both patients recovered after they were treated with antibiotics.
An average of 12 melioidosis cases are diagnosed in the United States each year, with most in people with recent travel to a country where the bacteria is endemic, or regularly found. Cases have also been linked to contaminated products imported from endemic countries. In late 2021, four cases in four states – Georgia, Kansas, Minnesota, and Texas – were linked to a contaminated aromatherapy spray that was imported, and Walmart issued a recall in November of that year, according to a CDC announcement. Two of the four people died.
Given the small number of cases found in the United States, the CDC believes the risk of melioidosis for the general population continues to be “very low,” and the risk of person-to-person spread is considered “extremely low.” But people who live on the Gulf Coast of Mississippi and who have health conditions that may put them at a higher risk, such as diabetes, chronic kidney disease, chronic lung disease, excessive alcohol use, and immunosuppressive conditions, should protect themselves.
The CDC recommends avoiding contact with soil or muddy water, particularly after heavy rains, and protecting open wounds with waterproof bandages. People should also wear waterproof boots when gardening, working in the yard, or doing agricultural work, which can prevent infection through the feet and lower legs, especially after flooding or storms. People should also wear gloves to protect their hands when working directly with soil.
Melioidosis has a wide range of symptoms, including fever, joint pain, headaches, coughing, chest pain, and belly pain. It can also cause conditions such as pneumonia, abscesses, and blood infections. The disease can infect any organ, including the brain. In most cases, symptoms appear within 1-21 days after exposure, with an average of 7 days after exposure.
The CDC’s health advisory for health professionals and public health officials shows that melioidosis is now considered to be locally endemic in areas of the Gulf Coast region in Mississippi.
“Once well-established in the soil, B. pseudomallei cannot feasibly be removed from the soil,” according to the advisory. “Public health efforts should focus primarily on improving identification of cases so that appropriate treatment can be administered.”
A version of this article first appeared on WebMD.com.
, according to a new alert from the Centers for Disease Control and Prevention.
The bacteria, Burkholderia pseudomallei, was found along the Gulf Coast region in southern Mississippi. Typically, the bacteria are in tropical and subtropical climates, especially in parts of Southeast Asia, northern Australia, Central America, South America, Puerto Rico, and the U.S. Virgin Islands.
The bacteria can cause melioidosis, a rare and serious infectious disease that spreads to animals and humans through contact with contaminated soil and water via cuts, wounds, mucous membranes, breathing the bacteria in, or eating or drinking it. Worldwide, the disease is fatal in 10%-50% of those who become infected.
CDC and state officials are investigating the samples to find out how widespread the bacteria are within the United States. So far, modeling suggests that the environmental conditions on the Gulf Coast support the growth of B. pseudomallei.
“It is unclear how long the bacteria has been in the environment and where else it might be found in the U.S.,” according to the CDC statement. “CDC is alerting clinicians throughout the country of this discovery through a national health advisory, reminding them to be aware of the signs and symptoms of melioidosis and to consider melioidosis in patients that present with symptoms of the disease.”
Two unrelated people who live near the Gulf Coast region of Mississippi became sick with melioidosis recently – one in July 2020 and one in May 2022. Neither had traveled outside of the United States. The cases led the CDC and the Mississippi State Department of Health to collect environmental samples and test household products at the patients’ homes in June 2022. Three of the samples taken from soil and puddle water in the 2020 case tested positive for the bacteria.
Genomic sequencing revealed that both patients were infected with the same strain of the bacteria from the Western Hemisphere. They were hospitalized with sepsis due to pneumonia and had known risk factors for melioidosis. Both patients recovered after they were treated with antibiotics.
An average of 12 melioidosis cases are diagnosed in the United States each year, with most in people with recent travel to a country where the bacteria is endemic, or regularly found. Cases have also been linked to contaminated products imported from endemic countries. In late 2021, four cases in four states – Georgia, Kansas, Minnesota, and Texas – were linked to a contaminated aromatherapy spray that was imported, and Walmart issued a recall in November of that year, according to a CDC announcement. Two of the four people died.
Given the small number of cases found in the United States, the CDC believes the risk of melioidosis for the general population continues to be “very low,” and the risk of person-to-person spread is considered “extremely low.” But people who live on the Gulf Coast of Mississippi and who have health conditions that may put them at a higher risk, such as diabetes, chronic kidney disease, chronic lung disease, excessive alcohol use, and immunosuppressive conditions, should protect themselves.
The CDC recommends avoiding contact with soil or muddy water, particularly after heavy rains, and protecting open wounds with waterproof bandages. People should also wear waterproof boots when gardening, working in the yard, or doing agricultural work, which can prevent infection through the feet and lower legs, especially after flooding or storms. People should also wear gloves to protect their hands when working directly with soil.
Melioidosis has a wide range of symptoms, including fever, joint pain, headaches, coughing, chest pain, and belly pain. It can also cause conditions such as pneumonia, abscesses, and blood infections. The disease can infect any organ, including the brain. In most cases, symptoms appear within 1-21 days after exposure, with an average of 7 days after exposure.
The CDC’s health advisory for health professionals and public health officials shows that melioidosis is now considered to be locally endemic in areas of the Gulf Coast region in Mississippi.
“Once well-established in the soil, B. pseudomallei cannot feasibly be removed from the soil,” according to the advisory. “Public health efforts should focus primarily on improving identification of cases so that appropriate treatment can be administered.”
A version of this article first appeared on WebMD.com.
, according to a new alert from the Centers for Disease Control and Prevention.
The bacteria, Burkholderia pseudomallei, was found along the Gulf Coast region in southern Mississippi. Typically, the bacteria are in tropical and subtropical climates, especially in parts of Southeast Asia, northern Australia, Central America, South America, Puerto Rico, and the U.S. Virgin Islands.
The bacteria can cause melioidosis, a rare and serious infectious disease that spreads to animals and humans through contact with contaminated soil and water via cuts, wounds, mucous membranes, breathing the bacteria in, or eating or drinking it. Worldwide, the disease is fatal in 10%-50% of those who become infected.
CDC and state officials are investigating the samples to find out how widespread the bacteria are within the United States. So far, modeling suggests that the environmental conditions on the Gulf Coast support the growth of B. pseudomallei.
“It is unclear how long the bacteria has been in the environment and where else it might be found in the U.S.,” according to the CDC statement. “CDC is alerting clinicians throughout the country of this discovery through a national health advisory, reminding them to be aware of the signs and symptoms of melioidosis and to consider melioidosis in patients that present with symptoms of the disease.”
Two unrelated people who live near the Gulf Coast region of Mississippi became sick with melioidosis recently – one in July 2020 and one in May 2022. Neither had traveled outside of the United States. The cases led the CDC and the Mississippi State Department of Health to collect environmental samples and test household products at the patients’ homes in June 2022. Three of the samples taken from soil and puddle water in the 2020 case tested positive for the bacteria.
Genomic sequencing revealed that both patients were infected with the same strain of the bacteria from the Western Hemisphere. They were hospitalized with sepsis due to pneumonia and had known risk factors for melioidosis. Both patients recovered after they were treated with antibiotics.
An average of 12 melioidosis cases are diagnosed in the United States each year, with most in people with recent travel to a country where the bacteria is endemic, or regularly found. Cases have also been linked to contaminated products imported from endemic countries. In late 2021, four cases in four states – Georgia, Kansas, Minnesota, and Texas – were linked to a contaminated aromatherapy spray that was imported, and Walmart issued a recall in November of that year, according to a CDC announcement. Two of the four people died.
Given the small number of cases found in the United States, the CDC believes the risk of melioidosis for the general population continues to be “very low,” and the risk of person-to-person spread is considered “extremely low.” But people who live on the Gulf Coast of Mississippi and who have health conditions that may put them at a higher risk, such as diabetes, chronic kidney disease, chronic lung disease, excessive alcohol use, and immunosuppressive conditions, should protect themselves.
The CDC recommends avoiding contact with soil or muddy water, particularly after heavy rains, and protecting open wounds with waterproof bandages. People should also wear waterproof boots when gardening, working in the yard, or doing agricultural work, which can prevent infection through the feet and lower legs, especially after flooding or storms. People should also wear gloves to protect their hands when working directly with soil.
Melioidosis has a wide range of symptoms, including fever, joint pain, headaches, coughing, chest pain, and belly pain. It can also cause conditions such as pneumonia, abscesses, and blood infections. The disease can infect any organ, including the brain. In most cases, symptoms appear within 1-21 days after exposure, with an average of 7 days after exposure.
The CDC’s health advisory for health professionals and public health officials shows that melioidosis is now considered to be locally endemic in areas of the Gulf Coast region in Mississippi.
“Once well-established in the soil, B. pseudomallei cannot feasibly be removed from the soil,” according to the advisory. “Public health efforts should focus primarily on improving identification of cases so that appropriate treatment can be administered.”
A version of this article first appeared on WebMD.com.
Calcinosis Cutis Associated With Subcutaneous Glatiramer Acetate
To the Editor:
Calcinosis cutis is a condition characterized by the deposition of insoluble calcium salts in the skin. Dystrophic calcinosis cutis is the most common type, occurring in previously traumatized skin in the absence of abnormal blood calcium levels. It commonly is seen in patients with connective tissue diseases and is thought to be precipitated by chronic inflammation and vascular hypoxia.1 Herein, we describe a case of calcinosis cutis arising after treatment with subcutaneous glatiramer acetate, an agent that is effective for the treatment of relapsing-remitting multiple sclerosis (MS). Diagnostic workup and treatment modalities for calcinosis cutis in this patient population should be considered in the context of minimizing interruption or discontinuation of this disease-modifying agent.
A 53-year-old woman with a history of relapsing-remitting MS and systemic lupus erythematosus (SLE) presented with multiple firm asymptomatic subcutaneous nodules on the thighs of 1 year’s duration that were increasing in number. The involved areas were the injection sites of subcutaneous glatiramer acetate, an immunomodulator for the treatment of MS, which our patient self-administered 3 times weekly. Physical examination revealed multiple flesh-colored to white, firm, and nontender nodules on the thighs (Figure). There was no epidermal change, and she had no other skin involvement. A punch biopsy of one of the nodules revealed calcium deposits in collagen bundles of the deep dermis. Calcium, phosphorus, parathyroid hormone, and vitamin D levels were within reference range. She declined further treatment for the calcinosis cutis and opted to continue treatment with glatiramer acetate, as her MS was well controlled on this medication.
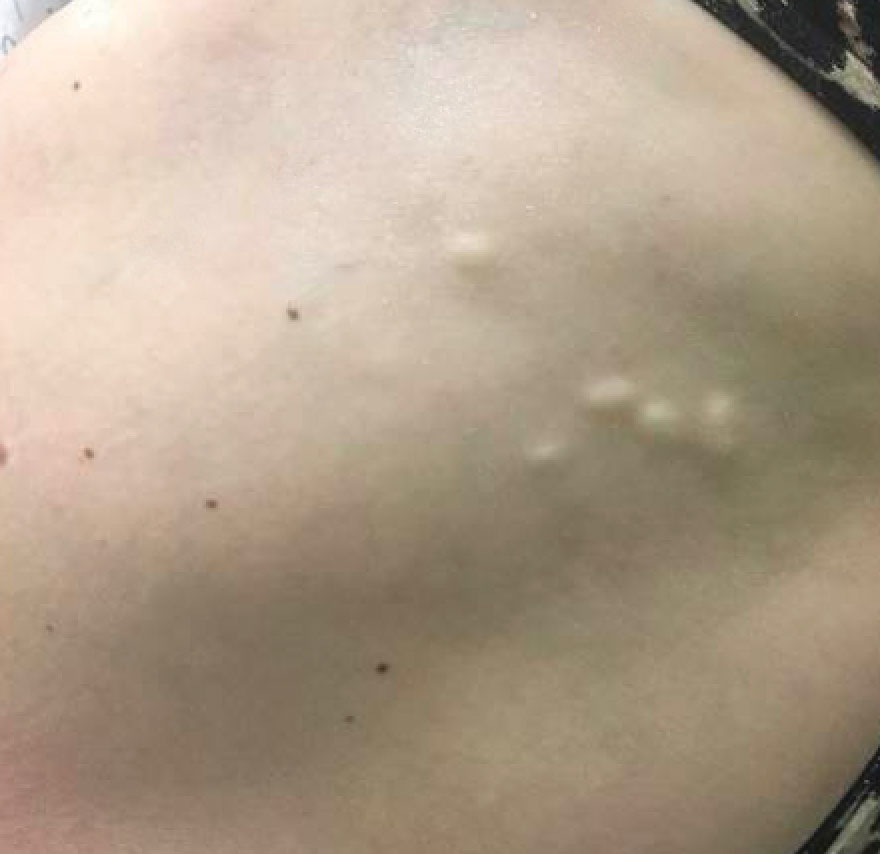
Glatiramer acetate is an immunogenic polypeptide injectable that is approved by the US Food and Drug Administration for the treatment of relapsing-remitting MS.2 It is composed of synthetic polypeptides and contains 4 naturally occurring amino acids. Glatiramer acetate is administered subcutaneously as 20 mg/mL/d or 40 mg/mL 3 times weekly. Transient injection-site reactions are the most common cutaneous adverse events and include localized edema, induration, erythema, pain, and pruritus.3 There have been multiple reports of lobular panniculitis and skin necrosis as well as embolia cutis medicamentosa (Nicolau syndrome).4,5 Our case of calcinosis cutis related to glatiramer acetate is unique. The mechanism of calcinosis cutis in our patient likely was dystrophic due to tissue damage, rather than due to the injection of a calcium-containing substance. Our patient’s history of SLE is a notable risk factor for the development of calcinosis cutis, likely incited by the trauma occurring with subcutaneous injections.6
The mainstay of treatment for localized calcinosis cutis in the setting of connective tissue disease is surgical excision as well as treatment of the underlying disorder. Potential therapies include calcium channel blockers, warfarin, bisphosphonates, intravenous immunoglobulin, minocycline, colchicine, anti–tumor necrosis factor agents, intralesional corticosteroids, intravenous sodium thiosulfate, and CO2 laser.1,6 Our patient was already on intravenous immunoglobulin for MS and hydroxychloroquine for SLE. In select cases where the patient is asymptomatic and prefers not to pursue treatment, no treatment is necessary.
Although calcinosis cutis may occur in SLE alone, it is uncommon and usually is seen in chronic severe SLE, where calcification usually occurs in the setting of pre-existing cutaneous lupus.4 This case report of calcinosis cutis following treatment with glatiramer acetate highlights some of the cutaneous side effects associated with glatiramer acetate injections and should prompt practitioners to consider dystrophic calcinosis cutis in patients requiring subcutaneous medications, particularly in those with pre-existing connective tissue disease.
- Valenzuela A, Chung L. Calcinosis: pathophysiology and management. Curr Opin Rheumatol. 2015;27:542-548.
- Copaxone. Prescribing information. Teva Neuroscience, Inc; 2022. Accessed July 15, 2022. https://www.copaxone.com/globalassets/copaxone/prescribing-information.pdf
- McKeage K. Glatiramer acetate 40 mg/mL in relapsing-remitting multiple sclerosis: a review. CNS Drugs. 2015;29:425-432.
- Balak DMW, Hengstman GJD, Çakmak A, et al. Cutaneous adverse events associated with disease-modifying treatment in multiple sclerosis: a systematic review. Mult Scler. 2012;18:1705-1717.
- Watkins CE, Litchfield J, Youngberg G, et al. Glatiramer acetate-induced lobular panniculitis and skin necrosis. Cutis. 2015;95:E26-E30.
- Reiter N, El-Shabrawi L, Leinweber B, et al. Calcinosis cutis. J Am Acad Dermatol. 2011;65:1-12.
To the Editor:
Calcinosis cutis is a condition characterized by the deposition of insoluble calcium salts in the skin. Dystrophic calcinosis cutis is the most common type, occurring in previously traumatized skin in the absence of abnormal blood calcium levels. It commonly is seen in patients with connective tissue diseases and is thought to be precipitated by chronic inflammation and vascular hypoxia.1 Herein, we describe a case of calcinosis cutis arising after treatment with subcutaneous glatiramer acetate, an agent that is effective for the treatment of relapsing-remitting multiple sclerosis (MS). Diagnostic workup and treatment modalities for calcinosis cutis in this patient population should be considered in the context of minimizing interruption or discontinuation of this disease-modifying agent.
A 53-year-old woman with a history of relapsing-remitting MS and systemic lupus erythematosus (SLE) presented with multiple firm asymptomatic subcutaneous nodules on the thighs of 1 year’s duration that were increasing in number. The involved areas were the injection sites of subcutaneous glatiramer acetate, an immunomodulator for the treatment of MS, which our patient self-administered 3 times weekly. Physical examination revealed multiple flesh-colored to white, firm, and nontender nodules on the thighs (Figure). There was no epidermal change, and she had no other skin involvement. A punch biopsy of one of the nodules revealed calcium deposits in collagen bundles of the deep dermis. Calcium, phosphorus, parathyroid hormone, and vitamin D levels were within reference range. She declined further treatment for the calcinosis cutis and opted to continue treatment with glatiramer acetate, as her MS was well controlled on this medication.

Glatiramer acetate is an immunogenic polypeptide injectable that is approved by the US Food and Drug Administration for the treatment of relapsing-remitting MS.2 It is composed of synthetic polypeptides and contains 4 naturally occurring amino acids. Glatiramer acetate is administered subcutaneously as 20 mg/mL/d or 40 mg/mL 3 times weekly. Transient injection-site reactions are the most common cutaneous adverse events and include localized edema, induration, erythema, pain, and pruritus.3 There have been multiple reports of lobular panniculitis and skin necrosis as well as embolia cutis medicamentosa (Nicolau syndrome).4,5 Our case of calcinosis cutis related to glatiramer acetate is unique. The mechanism of calcinosis cutis in our patient likely was dystrophic due to tissue damage, rather than due to the injection of a calcium-containing substance. Our patient’s history of SLE is a notable risk factor for the development of calcinosis cutis, likely incited by the trauma occurring with subcutaneous injections.6
The mainstay of treatment for localized calcinosis cutis in the setting of connective tissue disease is surgical excision as well as treatment of the underlying disorder. Potential therapies include calcium channel blockers, warfarin, bisphosphonates, intravenous immunoglobulin, minocycline, colchicine, anti–tumor necrosis factor agents, intralesional corticosteroids, intravenous sodium thiosulfate, and CO2 laser.1,6 Our patient was already on intravenous immunoglobulin for MS and hydroxychloroquine for SLE. In select cases where the patient is asymptomatic and prefers not to pursue treatment, no treatment is necessary.
Although calcinosis cutis may occur in SLE alone, it is uncommon and usually is seen in chronic severe SLE, where calcification usually occurs in the setting of pre-existing cutaneous lupus.4 This case report of calcinosis cutis following treatment with glatiramer acetate highlights some of the cutaneous side effects associated with glatiramer acetate injections and should prompt practitioners to consider dystrophic calcinosis cutis in patients requiring subcutaneous medications, particularly in those with pre-existing connective tissue disease.
To the Editor:
Calcinosis cutis is a condition characterized by the deposition of insoluble calcium salts in the skin. Dystrophic calcinosis cutis is the most common type, occurring in previously traumatized skin in the absence of abnormal blood calcium levels. It commonly is seen in patients with connective tissue diseases and is thought to be precipitated by chronic inflammation and vascular hypoxia.1 Herein, we describe a case of calcinosis cutis arising after treatment with subcutaneous glatiramer acetate, an agent that is effective for the treatment of relapsing-remitting multiple sclerosis (MS). Diagnostic workup and treatment modalities for calcinosis cutis in this patient population should be considered in the context of minimizing interruption or discontinuation of this disease-modifying agent.
A 53-year-old woman with a history of relapsing-remitting MS and systemic lupus erythematosus (SLE) presented with multiple firm asymptomatic subcutaneous nodules on the thighs of 1 year’s duration that were increasing in number. The involved areas were the injection sites of subcutaneous glatiramer acetate, an immunomodulator for the treatment of MS, which our patient self-administered 3 times weekly. Physical examination revealed multiple flesh-colored to white, firm, and nontender nodules on the thighs (Figure). There was no epidermal change, and she had no other skin involvement. A punch biopsy of one of the nodules revealed calcium deposits in collagen bundles of the deep dermis. Calcium, phosphorus, parathyroid hormone, and vitamin D levels were within reference range. She declined further treatment for the calcinosis cutis and opted to continue treatment with glatiramer acetate, as her MS was well controlled on this medication.

Glatiramer acetate is an immunogenic polypeptide injectable that is approved by the US Food and Drug Administration for the treatment of relapsing-remitting MS.2 It is composed of synthetic polypeptides and contains 4 naturally occurring amino acids. Glatiramer acetate is administered subcutaneously as 20 mg/mL/d or 40 mg/mL 3 times weekly. Transient injection-site reactions are the most common cutaneous adverse events and include localized edema, induration, erythema, pain, and pruritus.3 There have been multiple reports of lobular panniculitis and skin necrosis as well as embolia cutis medicamentosa (Nicolau syndrome).4,5 Our case of calcinosis cutis related to glatiramer acetate is unique. The mechanism of calcinosis cutis in our patient likely was dystrophic due to tissue damage, rather than due to the injection of a calcium-containing substance. Our patient’s history of SLE is a notable risk factor for the development of calcinosis cutis, likely incited by the trauma occurring with subcutaneous injections.6
The mainstay of treatment for localized calcinosis cutis in the setting of connective tissue disease is surgical excision as well as treatment of the underlying disorder. Potential therapies include calcium channel blockers, warfarin, bisphosphonates, intravenous immunoglobulin, minocycline, colchicine, anti–tumor necrosis factor agents, intralesional corticosteroids, intravenous sodium thiosulfate, and CO2 laser.1,6 Our patient was already on intravenous immunoglobulin for MS and hydroxychloroquine for SLE. In select cases where the patient is asymptomatic and prefers not to pursue treatment, no treatment is necessary.
Although calcinosis cutis may occur in SLE alone, it is uncommon and usually is seen in chronic severe SLE, where calcification usually occurs in the setting of pre-existing cutaneous lupus.4 This case report of calcinosis cutis following treatment with glatiramer acetate highlights some of the cutaneous side effects associated with glatiramer acetate injections and should prompt practitioners to consider dystrophic calcinosis cutis in patients requiring subcutaneous medications, particularly in those with pre-existing connective tissue disease.
- Valenzuela A, Chung L. Calcinosis: pathophysiology and management. Curr Opin Rheumatol. 2015;27:542-548.
- Copaxone. Prescribing information. Teva Neuroscience, Inc; 2022. Accessed July 15, 2022. https://www.copaxone.com/globalassets/copaxone/prescribing-information.pdf
- McKeage K. Glatiramer acetate 40 mg/mL in relapsing-remitting multiple sclerosis: a review. CNS Drugs. 2015;29:425-432.
- Balak DMW, Hengstman GJD, Çakmak A, et al. Cutaneous adverse events associated with disease-modifying treatment in multiple sclerosis: a systematic review. Mult Scler. 2012;18:1705-1717.
- Watkins CE, Litchfield J, Youngberg G, et al. Glatiramer acetate-induced lobular panniculitis and skin necrosis. Cutis. 2015;95:E26-E30.
- Reiter N, El-Shabrawi L, Leinweber B, et al. Calcinosis cutis. J Am Acad Dermatol. 2011;65:1-12.
- Valenzuela A, Chung L. Calcinosis: pathophysiology and management. Curr Opin Rheumatol. 2015;27:542-548.
- Copaxone. Prescribing information. Teva Neuroscience, Inc; 2022. Accessed July 15, 2022. https://www.copaxone.com/globalassets/copaxone/prescribing-information.pdf
- McKeage K. Glatiramer acetate 40 mg/mL in relapsing-remitting multiple sclerosis: a review. CNS Drugs. 2015;29:425-432.
- Balak DMW, Hengstman GJD, Çakmak A, et al. Cutaneous adverse events associated with disease-modifying treatment in multiple sclerosis: a systematic review. Mult Scler. 2012;18:1705-1717.
- Watkins CE, Litchfield J, Youngberg G, et al. Glatiramer acetate-induced lobular panniculitis and skin necrosis. Cutis. 2015;95:E26-E30.
- Reiter N, El-Shabrawi L, Leinweber B, et al. Calcinosis cutis. J Am Acad Dermatol. 2011;65:1-12.
Practice Points
- Glatiramer acetate is a subcutaneous injection utilized for relapsing-remitting multiple sclerosis, and common adverse effects include injection-site reactions such as calcinosis cutis.
- Development of calcinosis cutis in association with glatiramer acetate is not an indication for medication discontinuation.
- Dermatologists should be aware of this potential association, and treatment should be considered in cases of symptomatic calcinosis cutis.
Rituximab for Acquired Hemophilia A in the Setting of Bullous Pemphigoid
To the Editor:
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by the formation of antihemidesmosomal antibodies, resulting in tense bullae concentrated on the extremities and trunk that often are preceded by a pruritic urticarial phase.1 A rare complication of BP is the subsequent development of acquired hemophilia A. We report a case of BP with associated factor VIII–neutralizing antibodies in a patient who improved with prednisone and rituximab therapy.
A 78-year-old woman presented with red-orange pruritic plaques on the right heel that spread to involve the arms and legs, abdomen, and trunk with new-onset bullae over the course of 2 weeks (Figure 1). Dermatology was consulted, and a diagnosis of BP was confirmed via biopsy and direct immunofluorescence.
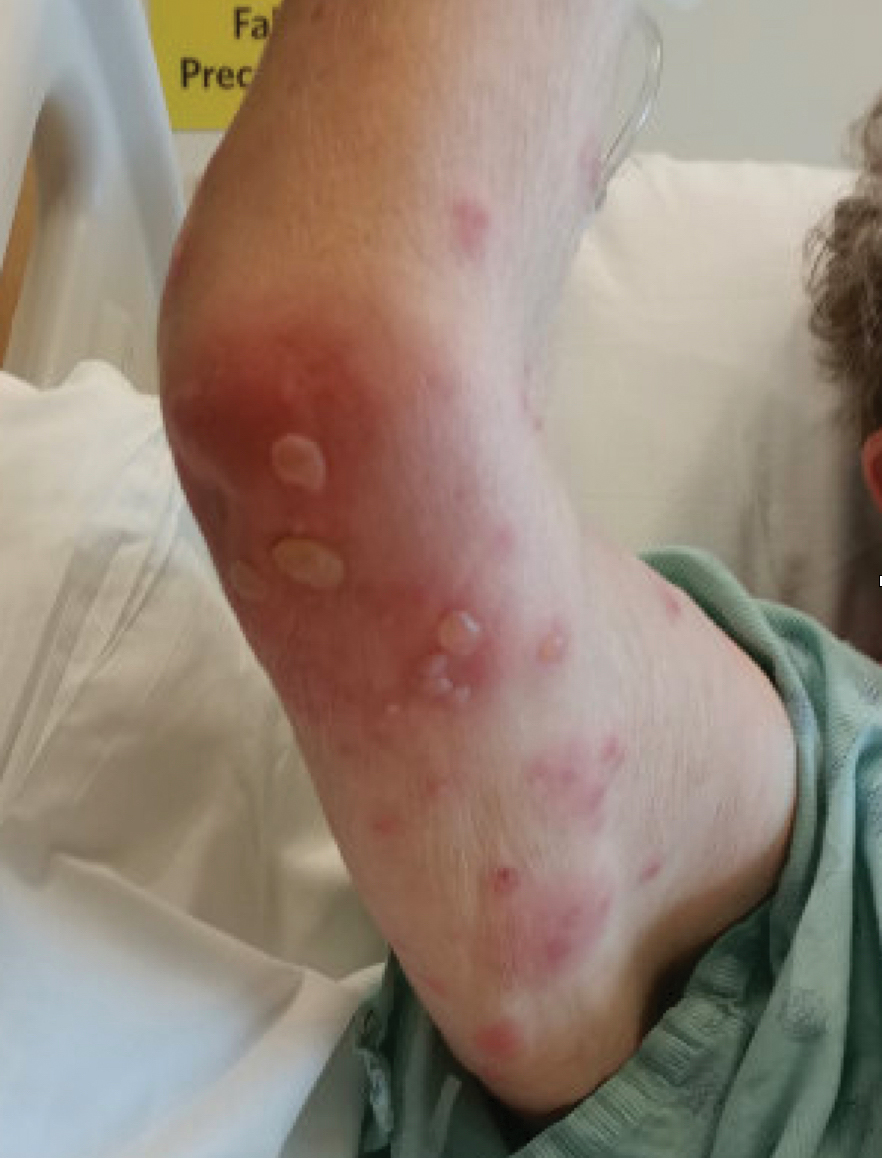
Despite treatment with prednisone 40 mg/d and clobetasol ointment 0.05%, she continued to develop extensive cutaneous bullae and new hemorrhagic bullae on the buccal mucosae (Figure 2), necessitating hospital admission. She clinically improved after prednisone was increased to 60 mg/d and mycophenolate mofetil 500 mg twice daily was added; however, she returned 8 days after discharge from the hospital with altered mental status, new-onset hematomas of the abdomen and right leg, and a hemoglobin level of 5.8 g/dL (reference range, 14.0–17.5 g/dL). Activated prothrombin time was prolonged without correction on mixing studies, raising concern for coagulation factor inhibition. Factor VIII activity was diminished to 9% and then 1% three days later. Mycophenolate mofetil was discontinued, and the patient was acutely stabilized with blood transfusions, intravenous immunoglobulin, tranexamic acid, and aminocaproic acid. Rituximab was initiated at 1000 mg and then administered again 2 weeks later. At 7-week follow-up, coagulation studies normalized, and there was no evidence of blistering dermatosis on examination.
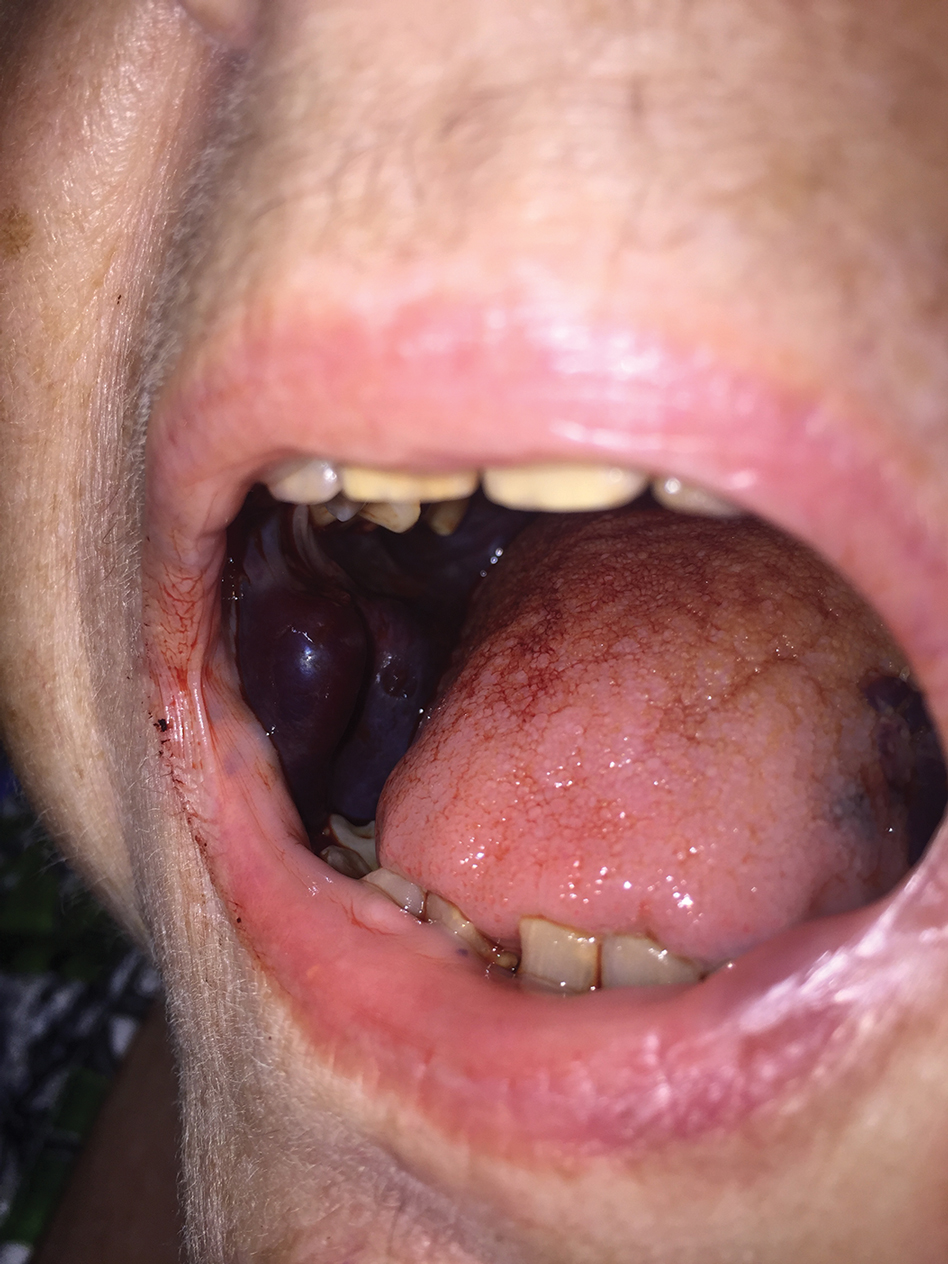
Bullous pemphigoid generally is seen in patients older than 60 years, and the incidence increases with age. The disease course follows formation of IgG antibodies against BP180 or BP230, leading to localized activation of the complement cascade at the basement membrane zone.1 Medications, vaccinations, UV radiation, and burns have been implicated in disease induction.2
Identification of antihemidesmosomal antibodies on lesional biopsy via direct immunofluorescence is the gold standard for diagnosis, though indirect antibodies measured via enzyme-linked immunosorbent assay may provide information regarding disease severity.1 Patients with milder disease may be treated with topical corticosteroids, doxycycline, and nicotinamide; however, severe disease requires treatment with systemic glucocorticoids and steroid-sparing agents.3 Rituximab initially was approved by the US Food and Drug Administration for the treatment of pemphigus vulgaris, and mounting evidence for the use of rituximab in BP is promising. Although data are limited to retrospective studies, rituximab has shown notable remission rates and steroid-sparing effects in those with moderate to severe BP.4
Acquired hemophilia A (AHA) is caused by the production of IgG autoantibodies, which block physiologic interactions between factor VIII and factor IX, phospholipids, and von Willebrand factor.5 Acquired hemophilia A often is diagnosed by prolonged activated prothrombin time and decreased factor VIII activity after a previously unaffected patient develops severe bleeding. Treatment involves re-establishing hemostasis and the use of corticosteroids and immunosuppressive agents to diminish autoantibody production.4
Bullous pemphigoid–associated AHA likely is due to antigenic similarity between BP180 and factor VIII, leading to concomitant neutralization of factor VIII with the production of BP-associated autoantibodies.5 Bullous pemphigoid–associated AHA has been reported with manifestations of bleeding concurrent with or after the development of dermatologic disease. Rituximab use has been reported with clinical efficacy in several cases, including our patient.6 Continued hematologic monitoring is recommended, as recurrences are common within the first 2 years.5
- Bağcı IS, Horváth ON, Ruzicka T, et al. Bullous pemphigoid. Autoimmun Rev. 2017;16:445-455.
- Schiavo AL, Ruocco E, Brancaccio G, et al. Bullous pemphigoid: etiology, pathogenesis, and inducing factors: facts and controversies. Clin Dermatol 2013;31:391-399.
- Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-332.
- Cho Y, Chu C, Wang L. First-line combination therapy with rituximab and corticosteroids provides a high complete remission rate in moderate-to-severe bullous pemphigoid. Br J Dermatol. 2015;173:302-304.
- Zdziarska J, Musial J. Acquired hemophilia A: an underdiagnosed severe bleeding disorder. Pol Arch Med Wewn. 2014;124:200-206.
- Binet Q, Lambert C, Sacré L, et al. Successful management of acquired hemophilia associated with bullous pemphigoid: a case report and review of the literature [published online March 28, 2017]. Case Rep Hematol. 2017;2017:2057019.
To the Editor:
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by the formation of antihemidesmosomal antibodies, resulting in tense bullae concentrated on the extremities and trunk that often are preceded by a pruritic urticarial phase.1 A rare complication of BP is the subsequent development of acquired hemophilia A. We report a case of BP with associated factor VIII–neutralizing antibodies in a patient who improved with prednisone and rituximab therapy.
A 78-year-old woman presented with red-orange pruritic plaques on the right heel that spread to involve the arms and legs, abdomen, and trunk with new-onset bullae over the course of 2 weeks (Figure 1). Dermatology was consulted, and a diagnosis of BP was confirmed via biopsy and direct immunofluorescence.

Despite treatment with prednisone 40 mg/d and clobetasol ointment 0.05%, she continued to develop extensive cutaneous bullae and new hemorrhagic bullae on the buccal mucosae (Figure 2), necessitating hospital admission. She clinically improved after prednisone was increased to 60 mg/d and mycophenolate mofetil 500 mg twice daily was added; however, she returned 8 days after discharge from the hospital with altered mental status, new-onset hematomas of the abdomen and right leg, and a hemoglobin level of 5.8 g/dL (reference range, 14.0–17.5 g/dL). Activated prothrombin time was prolonged without correction on mixing studies, raising concern for coagulation factor inhibition. Factor VIII activity was diminished to 9% and then 1% three days later. Mycophenolate mofetil was discontinued, and the patient was acutely stabilized with blood transfusions, intravenous immunoglobulin, tranexamic acid, and aminocaproic acid. Rituximab was initiated at 1000 mg and then administered again 2 weeks later. At 7-week follow-up, coagulation studies normalized, and there was no evidence of blistering dermatosis on examination.

Bullous pemphigoid generally is seen in patients older than 60 years, and the incidence increases with age. The disease course follows formation of IgG antibodies against BP180 or BP230, leading to localized activation of the complement cascade at the basement membrane zone.1 Medications, vaccinations, UV radiation, and burns have been implicated in disease induction.2
Identification of antihemidesmosomal antibodies on lesional biopsy via direct immunofluorescence is the gold standard for diagnosis, though indirect antibodies measured via enzyme-linked immunosorbent assay may provide information regarding disease severity.1 Patients with milder disease may be treated with topical corticosteroids, doxycycline, and nicotinamide; however, severe disease requires treatment with systemic glucocorticoids and steroid-sparing agents.3 Rituximab initially was approved by the US Food and Drug Administration for the treatment of pemphigus vulgaris, and mounting evidence for the use of rituximab in BP is promising. Although data are limited to retrospective studies, rituximab has shown notable remission rates and steroid-sparing effects in those with moderate to severe BP.4
Acquired hemophilia A (AHA) is caused by the production of IgG autoantibodies, which block physiologic interactions between factor VIII and factor IX, phospholipids, and von Willebrand factor.5 Acquired hemophilia A often is diagnosed by prolonged activated prothrombin time and decreased factor VIII activity after a previously unaffected patient develops severe bleeding. Treatment involves re-establishing hemostasis and the use of corticosteroids and immunosuppressive agents to diminish autoantibody production.4
Bullous pemphigoid–associated AHA likely is due to antigenic similarity between BP180 and factor VIII, leading to concomitant neutralization of factor VIII with the production of BP-associated autoantibodies.5 Bullous pemphigoid–associated AHA has been reported with manifestations of bleeding concurrent with or after the development of dermatologic disease. Rituximab use has been reported with clinical efficacy in several cases, including our patient.6 Continued hematologic monitoring is recommended, as recurrences are common within the first 2 years.5
To the Editor:
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by the formation of antihemidesmosomal antibodies, resulting in tense bullae concentrated on the extremities and trunk that often are preceded by a pruritic urticarial phase.1 A rare complication of BP is the subsequent development of acquired hemophilia A. We report a case of BP with associated factor VIII–neutralizing antibodies in a patient who improved with prednisone and rituximab therapy.
A 78-year-old woman presented with red-orange pruritic plaques on the right heel that spread to involve the arms and legs, abdomen, and trunk with new-onset bullae over the course of 2 weeks (Figure 1). Dermatology was consulted, and a diagnosis of BP was confirmed via biopsy and direct immunofluorescence.

Despite treatment with prednisone 40 mg/d and clobetasol ointment 0.05%, she continued to develop extensive cutaneous bullae and new hemorrhagic bullae on the buccal mucosae (Figure 2), necessitating hospital admission. She clinically improved after prednisone was increased to 60 mg/d and mycophenolate mofetil 500 mg twice daily was added; however, she returned 8 days after discharge from the hospital with altered mental status, new-onset hematomas of the abdomen and right leg, and a hemoglobin level of 5.8 g/dL (reference range, 14.0–17.5 g/dL). Activated prothrombin time was prolonged without correction on mixing studies, raising concern for coagulation factor inhibition. Factor VIII activity was diminished to 9% and then 1% three days later. Mycophenolate mofetil was discontinued, and the patient was acutely stabilized with blood transfusions, intravenous immunoglobulin, tranexamic acid, and aminocaproic acid. Rituximab was initiated at 1000 mg and then administered again 2 weeks later. At 7-week follow-up, coagulation studies normalized, and there was no evidence of blistering dermatosis on examination.

Bullous pemphigoid generally is seen in patients older than 60 years, and the incidence increases with age. The disease course follows formation of IgG antibodies against BP180 or BP230, leading to localized activation of the complement cascade at the basement membrane zone.1 Medications, vaccinations, UV radiation, and burns have been implicated in disease induction.2
Identification of antihemidesmosomal antibodies on lesional biopsy via direct immunofluorescence is the gold standard for diagnosis, though indirect antibodies measured via enzyme-linked immunosorbent assay may provide information regarding disease severity.1 Patients with milder disease may be treated with topical corticosteroids, doxycycline, and nicotinamide; however, severe disease requires treatment with systemic glucocorticoids and steroid-sparing agents.3 Rituximab initially was approved by the US Food and Drug Administration for the treatment of pemphigus vulgaris, and mounting evidence for the use of rituximab in BP is promising. Although data are limited to retrospective studies, rituximab has shown notable remission rates and steroid-sparing effects in those with moderate to severe BP.4
Acquired hemophilia A (AHA) is caused by the production of IgG autoantibodies, which block physiologic interactions between factor VIII and factor IX, phospholipids, and von Willebrand factor.5 Acquired hemophilia A often is diagnosed by prolonged activated prothrombin time and decreased factor VIII activity after a previously unaffected patient develops severe bleeding. Treatment involves re-establishing hemostasis and the use of corticosteroids and immunosuppressive agents to diminish autoantibody production.4
Bullous pemphigoid–associated AHA likely is due to antigenic similarity between BP180 and factor VIII, leading to concomitant neutralization of factor VIII with the production of BP-associated autoantibodies.5 Bullous pemphigoid–associated AHA has been reported with manifestations of bleeding concurrent with or after the development of dermatologic disease. Rituximab use has been reported with clinical efficacy in several cases, including our patient.6 Continued hematologic monitoring is recommended, as recurrences are common within the first 2 years.5
- Bağcı IS, Horváth ON, Ruzicka T, et al. Bullous pemphigoid. Autoimmun Rev. 2017;16:445-455.
- Schiavo AL, Ruocco E, Brancaccio G, et al. Bullous pemphigoid: etiology, pathogenesis, and inducing factors: facts and controversies. Clin Dermatol 2013;31:391-399.
- Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-332.
- Cho Y, Chu C, Wang L. First-line combination therapy with rituximab and corticosteroids provides a high complete remission rate in moderate-to-severe bullous pemphigoid. Br J Dermatol. 2015;173:302-304.
- Zdziarska J, Musial J. Acquired hemophilia A: an underdiagnosed severe bleeding disorder. Pol Arch Med Wewn. 2014;124:200-206.
- Binet Q, Lambert C, Sacré L, et al. Successful management of acquired hemophilia associated with bullous pemphigoid: a case report and review of the literature [published online March 28, 2017]. Case Rep Hematol. 2017;2017:2057019.
- Bağcı IS, Horváth ON, Ruzicka T, et al. Bullous pemphigoid. Autoimmun Rev. 2017;16:445-455.
- Schiavo AL, Ruocco E, Brancaccio G, et al. Bullous pemphigoid: etiology, pathogenesis, and inducing factors: facts and controversies. Clin Dermatol 2013;31:391-399.
- Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-332.
- Cho Y, Chu C, Wang L. First-line combination therapy with rituximab and corticosteroids provides a high complete remission rate in moderate-to-severe bullous pemphigoid. Br J Dermatol. 2015;173:302-304.
- Zdziarska J, Musial J. Acquired hemophilia A: an underdiagnosed severe bleeding disorder. Pol Arch Med Wewn. 2014;124:200-206.
- Binet Q, Lambert C, Sacré L, et al. Successful management of acquired hemophilia associated with bullous pemphigoid: a case report and review of the literature [published online March 28, 2017]. Case Rep Hematol. 2017;2017:2057019.
Practice Points
- Physicians must be aware of the potential for acquired hemophilia A in patients with bullous pemphigoid (BP).
- Rituximab is an effective therapy for BP and should be considered for patients in this cohort.
Peristomal Pyoderma Gangrenosum at an Ileostomy Site
To the Editor:
Peristomal pyoderma gangrenosum (PPG) is a rare entity first described in 1984.1 Lesions usually begin as pustules that coalesce into an erythematous skin ulceration that contains purulent material. The lesion appears on the skin that surrounds an abdominal stoma. Peristomal pyoderma gangrenosum typically is associated with Crohn disease and ulcerative colitis, cancer, blood dyscrasia, diabetes mellitus, and hepatitis.2 We describe a case of PPG following an ileostomy in a patient with colon cancer and a related history of Crohn disease.
A 32-year-old woman presented to a dermatology office with a spontaneously painful, 3.2-cm ulceration that was extremely tender to palpation, located immediately adjacent to the site of an ileostomy (Figure). The patient had a history of refractory constipation that failed to respond to standard conservative measures 4 years prior. She underwent a colonoscopy, which revealed a 6.5-cm, irregularly shaped, exophytic mass in the rectosigmoid portion of the colon. Histopathologic examination of several biopsies confirmed the diagnosis of moderately well-differentiated adenocarcinoma, and additional evaluation determined the cancer to be stage IIB. She had a medical history of pancolonic Crohn disease since high school that was treated with periodic infusions of infliximab at the standard dose of 5 mg/kg. Colon cancer treatment consisted of preoperative radiotherapy, complete colectomy with ileoanal anastomosis, and creation of a J-pouch and formation of a temporary ileostomy, along with postoperative capecitabine chemotherapy.
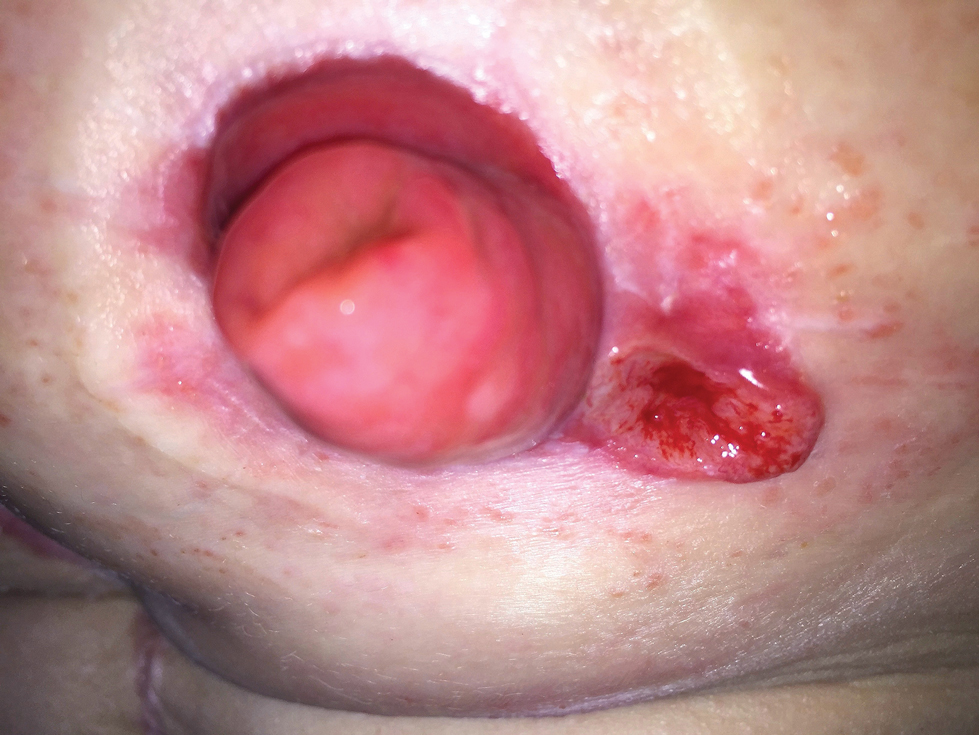
The ileostomy eventually was reversed, and the patient did well for 3 years. When the patient developed severe abdominal pain, the J-pouch was examined and found to be remarkably involved with Crohn disease. However, during the colonoscopy, the J-pouch was inadvertently punctured, leading to the formation of a large pelvic abscess. The latter necessitated diversion of stool, and the patient had the original ileostomy recreated.
Prior to presentation to dermatology, various consultants suspected the ulceration was possibly a deep fungal infection, cutaneous Crohn disease, a factitious ulceration, or acute allergic contact dermatitis related to some element of ostomy care. However, dermatologic consultation suggested that the troublesome lesion was classic PPG and recommended administration of a tumor necrosis factor (TNF) α–blocking agent and concomitant intralesional injections of dilute triamcinolone acetonide.
The patient was treated with subcutaneous adalimumab 40 mg once weekly, and received near weekly subcutaneous injections of triamcinolone acetonide 10 mg/mL. After 2 months, the discomfort subsided, and the ulceration gradually resolved into a depressed scar. Eighteen months later, the scar was barely perceptible as a minimally erythematous depression. Adalimumab ultimately was discontinued, as the residual J-pouch was removed, and the biologic drug was associated with extensive alopecia areata–like hair loss. There has been no recurrence of PPG in the 40 months since clinical resolution.
Peristomal pyoderma gangrenosum is an uncommon subtype of pyoderma gangrenosum, which is characterized by chronic, persistent, or recurrent painful ulceration(s) close to an abdominal stoma. In total, fewer than 100 cases of PPG have been reported thus far in the readily available medical literature.3 Inflammatory bowel disease (IBD) is the most frequently diagnosed systemic condition associated with PPG, though other associated conditions include diverticular disease, abdominal malignancy, and neurologic dysfunction. Approximately 2% to 4.3% of all patients who have stoma creation surgery related to underlying IBD develop PPG. It is estimated that the yearly incidence rate of PPG in all abdominal stomas is quite low (approximately 0.6%).4
Peristomal pyoderma gangrenosum can occur at any age, but it tends to predominate in young to middle-aged adults, with a slight female predilection. The etiology and pathogenesis of PPG are largely unknown, though studies have shown that an abnormal immune response may be critical to its development. Risk factors for PPG are not well defined but potentially include autoimmune disorders, a high body mass index, and females or African Americans with IBD.4 Because PPG does not have characteristic histopathologic features, it is a diagnosis of exclusion that is based on the clinical examination and histologic findings that rule out other potential disorders.
There are 4 types of PPG based on the clinical and histopathologic characteristics: ulcerative, pustular, bullous, and vegetative. Peristomal pyoderma gangrenosum tends to be either ulcerative or vegetative, with ulcerative being by far the predominant type. The onset of PPG is quite variable, occurring a few weeks to several years after stoma formation.5 Ulcer size can range from less than 3 cm to 30 cm.4 Lesions begin as deep painful nodules or as superficial hemorrhagic pustules, either idiopathic or following ostensibly minimal trauma. Subsequently, they become necrotic and form an ulceration. The ulcers can be single or multiple lesions, typically with erythematous raised borders and purulent discharge. The ulcers are extremely painful and rapidly progressive. After the ulcers heal, they often leave a characteristic weblike atrophic scar that can break down further following any form of irritation or trauma.5
A prompt diagnosis of PPG is important. A diagnosis of PPG should be considered when dealing with a noninfectious ulcer surrounding a stoma in patients with IBD or other autoimmune conditions.6 Because PPG is a rare skin disorder, it is likely to be missed and lead to unnecessary diagnostic workup and a delay in proper therapy. In our patient, a diagnosis of PPG was overlooked for other infectious and autoimmune causes. The diagnostic evaluation of a patient with PPG is based on 3 principles: (1) ruling out other causes of a peristomal ulcer, such as an abscess, contact dermatitis, or wound infection; (2) determining whether there is an underlying intestinal bowel disease in the stoma; and (3) identifying associated systemic disorders such as vasculitis, erythema nodosum, or similar processes.4 The differential diagnosis depends on the type and stage of PPG and can include malignancy, vasculitis, extraintestinal IBD, infectious disease, and insect bites. A review of the history of the ulcer is helpful in ruling out other diseases, and a colonoscopy or ileoscopy can identify if patients have an underlying active IBD. Swabs for smear and both bacterial and fungal cultures should be taken from the exudate and directly from the ulcer base. Biopsy of the ulcer also helps to exclude alternative diagnoses.6
The primary goals of treating PPG include to reduce pain and the risk for secondary infection, increase pouch adherence, and decrease purulent exudate.7 Although there is not one well-defined optimal therapeutic intervention, there are a variety of effective approaches that may be considered and used. In mild cases, management methods such as dressings, topical agents, or intralesional steroids may be capable of controlling the disease. Daily wound care is important. Moisture-retentive dressings can control pain, induce collagen formation, promote angiogenesis, and prevent contamination. Cleaning the wound with sterile saline and applying an anti-infective agent also may be effective. Application of ultrapotent topical steroids and tacrolimus ointment 0.3% can be used in patients without concomitant secondary infection. In patients who are in remission, human platelet-derived growth factor may be used. Intralesional injections of dilute triamcinolone acetonide or cyclosporine solution also can be helpful. Cyclosporin A was used as a systemic monotherapy to treat a 48-year-old man and 50-year-old woman with the idiopathic form of PPG. After 3 months of treatment, PPG had completely resolved and there were no major side effects.8 Other potential topical therapies that control inflammation and promote wound healing include benzoyl peroxide, chlormethine (topical alkylating agent and nitrogen mustard that has anti-inflammatory properties), nicotine, and 5-aminosalicylic acid. If an ulcer becomes infected, empiric antibiotic therapy should be given immediately and adjusted based on culture and sensitivity results.4
Systemic therapy should be considered in patients who do not respond to topical or local interventions, have a rapid and severe course, or have an active underlying bowel disease. Oral prednisone (1 mg/kg/d) has proved to be one of the most successful drugs used to treat PPG. Treatment should be continued until complete lesion healing, and low-dose maintenance therapy should be administered in recurrent cases. Intravenous corticosteroid therapy—hydrocortisone 100 mg 4 times daily or pulse therapy with intravenous methylprednisolone 1 g/d)—can be used for up to 5 days and may be effective. Oral minocycline 100 mg twice daily may be helpful as an adjunctive therapy to corticosteroids. When corticosteroids fail, oral cyclosporine 3 to 5 mg/kg/d often is prescribed. Studies have shown that patients demonstrate clinical improvement within 3 weeks of cyclosporine initiation, and it has been shown further to be more effective than either azathioprine or methotrexate.4,8
Infliximab, a chimeric antibody that binds both circulating and tissue-bound TNF-α, has been shown to effectively treat PPG. A clinical trial conducted by Brooklyn et al9 found that 46% of patients (6/13) treated with infliximab responded compared with only 6% in a placebo control group (1/17). Although infliximab may result in sepsis, the benefits far outweigh the risks, especially for patients with steroid-refractory PPG.4 Adalimumab is a human monoclonal IgG1 antibody to TNF-α that neutralizes its function by blocking the interaction between the molecule and its receptor. Many clinical studies have shown that adalimumab induces and maintains a clinical response in patients with active Crohn disease. The biologic proved to be effective in our patient, but it is associated with potential side effects that should be monitored including injection-site reactions, pruritus, leukopenia, urticaria, and rare instances of alopecia.10 Etanercept is another potentially effective biologic agent.7 Plasma exchange, immunoglobulin infusion, and interferon-alfa therapy also can be used in refractory PPG cases, though data on these treatments are very limited.4
Unlike routine pyoderma gangrenosum—for which surgical intervention is contraindicated—surgical intervention may be appropriate for the peristomal variant. Surgical treatment options include stoma revision and/or relocation; however, both of these procedures are accompanied by failure rates ranging from 40% to 100%.5 Removal of a diseased intestinal segment, especially one with active IBD, may result in healing of the skin lesion. In our patient, removal of the residual and diseased J-pouch was part of the management plan. However,it generally is recommended that any surgical intervention be accompanied by medical therapy including oral metronidazole 500 mg/d and concomitant administration of an immunosuppressant.1,3
Because PPG tends to recur, long-term maintenance therapy should always be considered. Pain reduction, anemia correction, proper nutrition, and management of associated and underlying diseases should be performed. Meticulous care of the stoma and prevention of leaks also should be emphasized. Overall, if PPG is detected and diagnosed early as well as treated appropriately and aggressively, the patient likely will have a good prognosis.4
- Sheldon DG, Sawchuk LL, Kozarek RA, et al. Twenty cases of peristomal pyoderma gangrenosum: diagnostic implications and management. Arch Surg. 2000;135:564-569.
- Hughes AP, Jackson JM, Callen JP. Clinical features and treatment of peristomal pyoderma gangrenosum. JAMA. 2000;284:1546-1548.
- Afifi L, Sanchez IM, Wallace MM, et al. Diagnosis and management of peristomal pyoderma gangrenosum: a systematic review. J Am Acad Dermatol. 2018;78:1195-1204.
- Wu XR, Shen B. Diagnosis and management of parastomal pyoderma gangrenosum. Gastroenterol Rep (Oxf). 2013;1:1-8.
- Javed A, Pal S, Ahuja V, et al. Management of peristomal pyoderma gangrenosum: two different approaches for the same clinical problem. Trop Gastroenterol. 2011;32:153-156.
- Toh JW, Whiteley I. Devastating peristomal pyoderma gangrenosum: challenges in diagnosis and management. Clin Gastroenterol Hepatol. 2017;15:A19-A20.
- DeMartyn LE, Faller NA, Miller L. Treating peristomal pyoderma gangrenosum with topical crushed prednisone: a report of three cases. Ostomy Wound Manage. 2014;60:50-54.
- V’lckova-Laskoska MT, Laskoski DS, Caca-Biljanovska NG, et al. Pyoderma gangrenosum successfully treated with cyclosporin A.Adv Exp Med Biol. 1999;455:541-555.
- Brooklyn TN, Dunnill MGS, Shetty A, at al. Infliximab for the treatment of pyoderma gangrenosum: a randomised, double blind, placebo controlled trial. Gut. 2006;55:505-509.
- Alkhouri N, Hupertz V, Mahajan L. Adalimumab treatment for peristomal pyoderma gangrenosum associated with Crohn’s disease. Inflamm Bowel Dis. 2009;15:803-806.
To the Editor:
Peristomal pyoderma gangrenosum (PPG) is a rare entity first described in 1984.1 Lesions usually begin as pustules that coalesce into an erythematous skin ulceration that contains purulent material. The lesion appears on the skin that surrounds an abdominal stoma. Peristomal pyoderma gangrenosum typically is associated with Crohn disease and ulcerative colitis, cancer, blood dyscrasia, diabetes mellitus, and hepatitis.2 We describe a case of PPG following an ileostomy in a patient with colon cancer and a related history of Crohn disease.
A 32-year-old woman presented to a dermatology office with a spontaneously painful, 3.2-cm ulceration that was extremely tender to palpation, located immediately adjacent to the site of an ileostomy (Figure). The patient had a history of refractory constipation that failed to respond to standard conservative measures 4 years prior. She underwent a colonoscopy, which revealed a 6.5-cm, irregularly shaped, exophytic mass in the rectosigmoid portion of the colon. Histopathologic examination of several biopsies confirmed the diagnosis of moderately well-differentiated adenocarcinoma, and additional evaluation determined the cancer to be stage IIB. She had a medical history of pancolonic Crohn disease since high school that was treated with periodic infusions of infliximab at the standard dose of 5 mg/kg. Colon cancer treatment consisted of preoperative radiotherapy, complete colectomy with ileoanal anastomosis, and creation of a J-pouch and formation of a temporary ileostomy, along with postoperative capecitabine chemotherapy.

The ileostomy eventually was reversed, and the patient did well for 3 years. When the patient developed severe abdominal pain, the J-pouch was examined and found to be remarkably involved with Crohn disease. However, during the colonoscopy, the J-pouch was inadvertently punctured, leading to the formation of a large pelvic abscess. The latter necessitated diversion of stool, and the patient had the original ileostomy recreated.
Prior to presentation to dermatology, various consultants suspected the ulceration was possibly a deep fungal infection, cutaneous Crohn disease, a factitious ulceration, or acute allergic contact dermatitis related to some element of ostomy care. However, dermatologic consultation suggested that the troublesome lesion was classic PPG and recommended administration of a tumor necrosis factor (TNF) α–blocking agent and concomitant intralesional injections of dilute triamcinolone acetonide.
The patient was treated with subcutaneous adalimumab 40 mg once weekly, and received near weekly subcutaneous injections of triamcinolone acetonide 10 mg/mL. After 2 months, the discomfort subsided, and the ulceration gradually resolved into a depressed scar. Eighteen months later, the scar was barely perceptible as a minimally erythematous depression. Adalimumab ultimately was discontinued, as the residual J-pouch was removed, and the biologic drug was associated with extensive alopecia areata–like hair loss. There has been no recurrence of PPG in the 40 months since clinical resolution.
Peristomal pyoderma gangrenosum is an uncommon subtype of pyoderma gangrenosum, which is characterized by chronic, persistent, or recurrent painful ulceration(s) close to an abdominal stoma. In total, fewer than 100 cases of PPG have been reported thus far in the readily available medical literature.3 Inflammatory bowel disease (IBD) is the most frequently diagnosed systemic condition associated with PPG, though other associated conditions include diverticular disease, abdominal malignancy, and neurologic dysfunction. Approximately 2% to 4.3% of all patients who have stoma creation surgery related to underlying IBD develop PPG. It is estimated that the yearly incidence rate of PPG in all abdominal stomas is quite low (approximately 0.6%).4
Peristomal pyoderma gangrenosum can occur at any age, but it tends to predominate in young to middle-aged adults, with a slight female predilection. The etiology and pathogenesis of PPG are largely unknown, though studies have shown that an abnormal immune response may be critical to its development. Risk factors for PPG are not well defined but potentially include autoimmune disorders, a high body mass index, and females or African Americans with IBD.4 Because PPG does not have characteristic histopathologic features, it is a diagnosis of exclusion that is based on the clinical examination and histologic findings that rule out other potential disorders.
There are 4 types of PPG based on the clinical and histopathologic characteristics: ulcerative, pustular, bullous, and vegetative. Peristomal pyoderma gangrenosum tends to be either ulcerative or vegetative, with ulcerative being by far the predominant type. The onset of PPG is quite variable, occurring a few weeks to several years after stoma formation.5 Ulcer size can range from less than 3 cm to 30 cm.4 Lesions begin as deep painful nodules or as superficial hemorrhagic pustules, either idiopathic or following ostensibly minimal trauma. Subsequently, they become necrotic and form an ulceration. The ulcers can be single or multiple lesions, typically with erythematous raised borders and purulent discharge. The ulcers are extremely painful and rapidly progressive. After the ulcers heal, they often leave a characteristic weblike atrophic scar that can break down further following any form of irritation or trauma.5
A prompt diagnosis of PPG is important. A diagnosis of PPG should be considered when dealing with a noninfectious ulcer surrounding a stoma in patients with IBD or other autoimmune conditions.6 Because PPG is a rare skin disorder, it is likely to be missed and lead to unnecessary diagnostic workup and a delay in proper therapy. In our patient, a diagnosis of PPG was overlooked for other infectious and autoimmune causes. The diagnostic evaluation of a patient with PPG is based on 3 principles: (1) ruling out other causes of a peristomal ulcer, such as an abscess, contact dermatitis, or wound infection; (2) determining whether there is an underlying intestinal bowel disease in the stoma; and (3) identifying associated systemic disorders such as vasculitis, erythema nodosum, or similar processes.4 The differential diagnosis depends on the type and stage of PPG and can include malignancy, vasculitis, extraintestinal IBD, infectious disease, and insect bites. A review of the history of the ulcer is helpful in ruling out other diseases, and a colonoscopy or ileoscopy can identify if patients have an underlying active IBD. Swabs for smear and both bacterial and fungal cultures should be taken from the exudate and directly from the ulcer base. Biopsy of the ulcer also helps to exclude alternative diagnoses.6
The primary goals of treating PPG include to reduce pain and the risk for secondary infection, increase pouch adherence, and decrease purulent exudate.7 Although there is not one well-defined optimal therapeutic intervention, there are a variety of effective approaches that may be considered and used. In mild cases, management methods such as dressings, topical agents, or intralesional steroids may be capable of controlling the disease. Daily wound care is important. Moisture-retentive dressings can control pain, induce collagen formation, promote angiogenesis, and prevent contamination. Cleaning the wound with sterile saline and applying an anti-infective agent also may be effective. Application of ultrapotent topical steroids and tacrolimus ointment 0.3% can be used in patients without concomitant secondary infection. In patients who are in remission, human platelet-derived growth factor may be used. Intralesional injections of dilute triamcinolone acetonide or cyclosporine solution also can be helpful. Cyclosporin A was used as a systemic monotherapy to treat a 48-year-old man and 50-year-old woman with the idiopathic form of PPG. After 3 months of treatment, PPG had completely resolved and there were no major side effects.8 Other potential topical therapies that control inflammation and promote wound healing include benzoyl peroxide, chlormethine (topical alkylating agent and nitrogen mustard that has anti-inflammatory properties), nicotine, and 5-aminosalicylic acid. If an ulcer becomes infected, empiric antibiotic therapy should be given immediately and adjusted based on culture and sensitivity results.4
Systemic therapy should be considered in patients who do not respond to topical or local interventions, have a rapid and severe course, or have an active underlying bowel disease. Oral prednisone (1 mg/kg/d) has proved to be one of the most successful drugs used to treat PPG. Treatment should be continued until complete lesion healing, and low-dose maintenance therapy should be administered in recurrent cases. Intravenous corticosteroid therapy—hydrocortisone 100 mg 4 times daily or pulse therapy with intravenous methylprednisolone 1 g/d)—can be used for up to 5 days and may be effective. Oral minocycline 100 mg twice daily may be helpful as an adjunctive therapy to corticosteroids. When corticosteroids fail, oral cyclosporine 3 to 5 mg/kg/d often is prescribed. Studies have shown that patients demonstrate clinical improvement within 3 weeks of cyclosporine initiation, and it has been shown further to be more effective than either azathioprine or methotrexate.4,8
Infliximab, a chimeric antibody that binds both circulating and tissue-bound TNF-α, has been shown to effectively treat PPG. A clinical trial conducted by Brooklyn et al9 found that 46% of patients (6/13) treated with infliximab responded compared with only 6% in a placebo control group (1/17). Although infliximab may result in sepsis, the benefits far outweigh the risks, especially for patients with steroid-refractory PPG.4 Adalimumab is a human monoclonal IgG1 antibody to TNF-α that neutralizes its function by blocking the interaction between the molecule and its receptor. Many clinical studies have shown that adalimumab induces and maintains a clinical response in patients with active Crohn disease. The biologic proved to be effective in our patient, but it is associated with potential side effects that should be monitored including injection-site reactions, pruritus, leukopenia, urticaria, and rare instances of alopecia.10 Etanercept is another potentially effective biologic agent.7 Plasma exchange, immunoglobulin infusion, and interferon-alfa therapy also can be used in refractory PPG cases, though data on these treatments are very limited.4
Unlike routine pyoderma gangrenosum—for which surgical intervention is contraindicated—surgical intervention may be appropriate for the peristomal variant. Surgical treatment options include stoma revision and/or relocation; however, both of these procedures are accompanied by failure rates ranging from 40% to 100%.5 Removal of a diseased intestinal segment, especially one with active IBD, may result in healing of the skin lesion. In our patient, removal of the residual and diseased J-pouch was part of the management plan. However,it generally is recommended that any surgical intervention be accompanied by medical therapy including oral metronidazole 500 mg/d and concomitant administration of an immunosuppressant.1,3
Because PPG tends to recur, long-term maintenance therapy should always be considered. Pain reduction, anemia correction, proper nutrition, and management of associated and underlying diseases should be performed. Meticulous care of the stoma and prevention of leaks also should be emphasized. Overall, if PPG is detected and diagnosed early as well as treated appropriately and aggressively, the patient likely will have a good prognosis.4
To the Editor:
Peristomal pyoderma gangrenosum (PPG) is a rare entity first described in 1984.1 Lesions usually begin as pustules that coalesce into an erythematous skin ulceration that contains purulent material. The lesion appears on the skin that surrounds an abdominal stoma. Peristomal pyoderma gangrenosum typically is associated with Crohn disease and ulcerative colitis, cancer, blood dyscrasia, diabetes mellitus, and hepatitis.2 We describe a case of PPG following an ileostomy in a patient with colon cancer and a related history of Crohn disease.
A 32-year-old woman presented to a dermatology office with a spontaneously painful, 3.2-cm ulceration that was extremely tender to palpation, located immediately adjacent to the site of an ileostomy (Figure). The patient had a history of refractory constipation that failed to respond to standard conservative measures 4 years prior. She underwent a colonoscopy, which revealed a 6.5-cm, irregularly shaped, exophytic mass in the rectosigmoid portion of the colon. Histopathologic examination of several biopsies confirmed the diagnosis of moderately well-differentiated adenocarcinoma, and additional evaluation determined the cancer to be stage IIB. She had a medical history of pancolonic Crohn disease since high school that was treated with periodic infusions of infliximab at the standard dose of 5 mg/kg. Colon cancer treatment consisted of preoperative radiotherapy, complete colectomy with ileoanal anastomosis, and creation of a J-pouch and formation of a temporary ileostomy, along with postoperative capecitabine chemotherapy.

The ileostomy eventually was reversed, and the patient did well for 3 years. When the patient developed severe abdominal pain, the J-pouch was examined and found to be remarkably involved with Crohn disease. However, during the colonoscopy, the J-pouch was inadvertently punctured, leading to the formation of a large pelvic abscess. The latter necessitated diversion of stool, and the patient had the original ileostomy recreated.
Prior to presentation to dermatology, various consultants suspected the ulceration was possibly a deep fungal infection, cutaneous Crohn disease, a factitious ulceration, or acute allergic contact dermatitis related to some element of ostomy care. However, dermatologic consultation suggested that the troublesome lesion was classic PPG and recommended administration of a tumor necrosis factor (TNF) α–blocking agent and concomitant intralesional injections of dilute triamcinolone acetonide.
The patient was treated with subcutaneous adalimumab 40 mg once weekly, and received near weekly subcutaneous injections of triamcinolone acetonide 10 mg/mL. After 2 months, the discomfort subsided, and the ulceration gradually resolved into a depressed scar. Eighteen months later, the scar was barely perceptible as a minimally erythematous depression. Adalimumab ultimately was discontinued, as the residual J-pouch was removed, and the biologic drug was associated with extensive alopecia areata–like hair loss. There has been no recurrence of PPG in the 40 months since clinical resolution.
Peristomal pyoderma gangrenosum is an uncommon subtype of pyoderma gangrenosum, which is characterized by chronic, persistent, or recurrent painful ulceration(s) close to an abdominal stoma. In total, fewer than 100 cases of PPG have been reported thus far in the readily available medical literature.3 Inflammatory bowel disease (IBD) is the most frequently diagnosed systemic condition associated with PPG, though other associated conditions include diverticular disease, abdominal malignancy, and neurologic dysfunction. Approximately 2% to 4.3% of all patients who have stoma creation surgery related to underlying IBD develop PPG. It is estimated that the yearly incidence rate of PPG in all abdominal stomas is quite low (approximately 0.6%).4
Peristomal pyoderma gangrenosum can occur at any age, but it tends to predominate in young to middle-aged adults, with a slight female predilection. The etiology and pathogenesis of PPG are largely unknown, though studies have shown that an abnormal immune response may be critical to its development. Risk factors for PPG are not well defined but potentially include autoimmune disorders, a high body mass index, and females or African Americans with IBD.4 Because PPG does not have characteristic histopathologic features, it is a diagnosis of exclusion that is based on the clinical examination and histologic findings that rule out other potential disorders.
There are 4 types of PPG based on the clinical and histopathologic characteristics: ulcerative, pustular, bullous, and vegetative. Peristomal pyoderma gangrenosum tends to be either ulcerative or vegetative, with ulcerative being by far the predominant type. The onset of PPG is quite variable, occurring a few weeks to several years after stoma formation.5 Ulcer size can range from less than 3 cm to 30 cm.4 Lesions begin as deep painful nodules or as superficial hemorrhagic pustules, either idiopathic or following ostensibly minimal trauma. Subsequently, they become necrotic and form an ulceration. The ulcers can be single or multiple lesions, typically with erythematous raised borders and purulent discharge. The ulcers are extremely painful and rapidly progressive. After the ulcers heal, they often leave a characteristic weblike atrophic scar that can break down further following any form of irritation or trauma.5
A prompt diagnosis of PPG is important. A diagnosis of PPG should be considered when dealing with a noninfectious ulcer surrounding a stoma in patients with IBD or other autoimmune conditions.6 Because PPG is a rare skin disorder, it is likely to be missed and lead to unnecessary diagnostic workup and a delay in proper therapy. In our patient, a diagnosis of PPG was overlooked for other infectious and autoimmune causes. The diagnostic evaluation of a patient with PPG is based on 3 principles: (1) ruling out other causes of a peristomal ulcer, such as an abscess, contact dermatitis, or wound infection; (2) determining whether there is an underlying intestinal bowel disease in the stoma; and (3) identifying associated systemic disorders such as vasculitis, erythema nodosum, or similar processes.4 The differential diagnosis depends on the type and stage of PPG and can include malignancy, vasculitis, extraintestinal IBD, infectious disease, and insect bites. A review of the history of the ulcer is helpful in ruling out other diseases, and a colonoscopy or ileoscopy can identify if patients have an underlying active IBD. Swabs for smear and both bacterial and fungal cultures should be taken from the exudate and directly from the ulcer base. Biopsy of the ulcer also helps to exclude alternative diagnoses.6
The primary goals of treating PPG include to reduce pain and the risk for secondary infection, increase pouch adherence, and decrease purulent exudate.7 Although there is not one well-defined optimal therapeutic intervention, there are a variety of effective approaches that may be considered and used. In mild cases, management methods such as dressings, topical agents, or intralesional steroids may be capable of controlling the disease. Daily wound care is important. Moisture-retentive dressings can control pain, induce collagen formation, promote angiogenesis, and prevent contamination. Cleaning the wound with sterile saline and applying an anti-infective agent also may be effective. Application of ultrapotent topical steroids and tacrolimus ointment 0.3% can be used in patients without concomitant secondary infection. In patients who are in remission, human platelet-derived growth factor may be used. Intralesional injections of dilute triamcinolone acetonide or cyclosporine solution also can be helpful. Cyclosporin A was used as a systemic monotherapy to treat a 48-year-old man and 50-year-old woman with the idiopathic form of PPG. After 3 months of treatment, PPG had completely resolved and there were no major side effects.8 Other potential topical therapies that control inflammation and promote wound healing include benzoyl peroxide, chlormethine (topical alkylating agent and nitrogen mustard that has anti-inflammatory properties), nicotine, and 5-aminosalicylic acid. If an ulcer becomes infected, empiric antibiotic therapy should be given immediately and adjusted based on culture and sensitivity results.4
Systemic therapy should be considered in patients who do not respond to topical or local interventions, have a rapid and severe course, or have an active underlying bowel disease. Oral prednisone (1 mg/kg/d) has proved to be one of the most successful drugs used to treat PPG. Treatment should be continued until complete lesion healing, and low-dose maintenance therapy should be administered in recurrent cases. Intravenous corticosteroid therapy—hydrocortisone 100 mg 4 times daily or pulse therapy with intravenous methylprednisolone 1 g/d)—can be used for up to 5 days and may be effective. Oral minocycline 100 mg twice daily may be helpful as an adjunctive therapy to corticosteroids. When corticosteroids fail, oral cyclosporine 3 to 5 mg/kg/d often is prescribed. Studies have shown that patients demonstrate clinical improvement within 3 weeks of cyclosporine initiation, and it has been shown further to be more effective than either azathioprine or methotrexate.4,8
Infliximab, a chimeric antibody that binds both circulating and tissue-bound TNF-α, has been shown to effectively treat PPG. A clinical trial conducted by Brooklyn et al9 found that 46% of patients (6/13) treated with infliximab responded compared with only 6% in a placebo control group (1/17). Although infliximab may result in sepsis, the benefits far outweigh the risks, especially for patients with steroid-refractory PPG.4 Adalimumab is a human monoclonal IgG1 antibody to TNF-α that neutralizes its function by blocking the interaction between the molecule and its receptor. Many clinical studies have shown that adalimumab induces and maintains a clinical response in patients with active Crohn disease. The biologic proved to be effective in our patient, but it is associated with potential side effects that should be monitored including injection-site reactions, pruritus, leukopenia, urticaria, and rare instances of alopecia.10 Etanercept is another potentially effective biologic agent.7 Plasma exchange, immunoglobulin infusion, and interferon-alfa therapy also can be used in refractory PPG cases, though data on these treatments are very limited.4
Unlike routine pyoderma gangrenosum—for which surgical intervention is contraindicated—surgical intervention may be appropriate for the peristomal variant. Surgical treatment options include stoma revision and/or relocation; however, both of these procedures are accompanied by failure rates ranging from 40% to 100%.5 Removal of a diseased intestinal segment, especially one with active IBD, may result in healing of the skin lesion. In our patient, removal of the residual and diseased J-pouch was part of the management plan. However,it generally is recommended that any surgical intervention be accompanied by medical therapy including oral metronidazole 500 mg/d and concomitant administration of an immunosuppressant.1,3
Because PPG tends to recur, long-term maintenance therapy should always be considered. Pain reduction, anemia correction, proper nutrition, and management of associated and underlying diseases should be performed. Meticulous care of the stoma and prevention of leaks also should be emphasized. Overall, if PPG is detected and diagnosed early as well as treated appropriately and aggressively, the patient likely will have a good prognosis.4
- Sheldon DG, Sawchuk LL, Kozarek RA, et al. Twenty cases of peristomal pyoderma gangrenosum: diagnostic implications and management. Arch Surg. 2000;135:564-569.
- Hughes AP, Jackson JM, Callen JP. Clinical features and treatment of peristomal pyoderma gangrenosum. JAMA. 2000;284:1546-1548.
- Afifi L, Sanchez IM, Wallace MM, et al. Diagnosis and management of peristomal pyoderma gangrenosum: a systematic review. J Am Acad Dermatol. 2018;78:1195-1204.
- Wu XR, Shen B. Diagnosis and management of parastomal pyoderma gangrenosum. Gastroenterol Rep (Oxf). 2013;1:1-8.
- Javed A, Pal S, Ahuja V, et al. Management of peristomal pyoderma gangrenosum: two different approaches for the same clinical problem. Trop Gastroenterol. 2011;32:153-156.
- Toh JW, Whiteley I. Devastating peristomal pyoderma gangrenosum: challenges in diagnosis and management. Clin Gastroenterol Hepatol. 2017;15:A19-A20.
- DeMartyn LE, Faller NA, Miller L. Treating peristomal pyoderma gangrenosum with topical crushed prednisone: a report of three cases. Ostomy Wound Manage. 2014;60:50-54.
- V’lckova-Laskoska MT, Laskoski DS, Caca-Biljanovska NG, et al. Pyoderma gangrenosum successfully treated with cyclosporin A.Adv Exp Med Biol. 1999;455:541-555.
- Brooklyn TN, Dunnill MGS, Shetty A, at al. Infliximab for the treatment of pyoderma gangrenosum: a randomised, double blind, placebo controlled trial. Gut. 2006;55:505-509.
- Alkhouri N, Hupertz V, Mahajan L. Adalimumab treatment for peristomal pyoderma gangrenosum associated with Crohn’s disease. Inflamm Bowel Dis. 2009;15:803-806.
- Sheldon DG, Sawchuk LL, Kozarek RA, et al. Twenty cases of peristomal pyoderma gangrenosum: diagnostic implications and management. Arch Surg. 2000;135:564-569.
- Hughes AP, Jackson JM, Callen JP. Clinical features and treatment of peristomal pyoderma gangrenosum. JAMA. 2000;284:1546-1548.
- Afifi L, Sanchez IM, Wallace MM, et al. Diagnosis and management of peristomal pyoderma gangrenosum: a systematic review. J Am Acad Dermatol. 2018;78:1195-1204.
- Wu XR, Shen B. Diagnosis and management of parastomal pyoderma gangrenosum. Gastroenterol Rep (Oxf). 2013;1:1-8.
- Javed A, Pal S, Ahuja V, et al. Management of peristomal pyoderma gangrenosum: two different approaches for the same clinical problem. Trop Gastroenterol. 2011;32:153-156.
- Toh JW, Whiteley I. Devastating peristomal pyoderma gangrenosum: challenges in diagnosis and management. Clin Gastroenterol Hepatol. 2017;15:A19-A20.
- DeMartyn LE, Faller NA, Miller L. Treating peristomal pyoderma gangrenosum with topical crushed prednisone: a report of three cases. Ostomy Wound Manage. 2014;60:50-54.
- V’lckova-Laskoska MT, Laskoski DS, Caca-Biljanovska NG, et al. Pyoderma gangrenosum successfully treated with cyclosporin A.Adv Exp Med Biol. 1999;455:541-555.
- Brooklyn TN, Dunnill MGS, Shetty A, at al. Infliximab for the treatment of pyoderma gangrenosum: a randomised, double blind, placebo controlled trial. Gut. 2006;55:505-509.
- Alkhouri N, Hupertz V, Mahajan L. Adalimumab treatment for peristomal pyoderma gangrenosum associated with Crohn’s disease. Inflamm Bowel Dis. 2009;15:803-806.
Practice Points
- A pyoderma gangrenosum subtype occurs in close proximity to an abdominal stoma.
- Peristomal pyoderma gangrenosum is a diagnosis of exclusion.
- Peristomal pyoderma gangrenosum typically responds best to tumor necrosis factor α blockers and corticosteroid therapy (intralesional and systemic).
Coming to a pill near you: The exercise molecule
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
How does radiofrequency microneedling work?
Technology in the field of aesthetic dermatology continues to advance over time. Microneedling, largely used to improve textural changes of the skin associated with photoaging and acne scarring, has evolved over time from the use of dermarollers and microneedling skin pens to energy-based devices that deliver radiofrequency (RF) energy though microneedles that are used today.
.
Unlike prior radiofrequency energy-based devices that deliver radiofrequency energy on the skin surface to allow bulk thermal energy (or heat) to stimulate collagen remodeling and tissue tightening, RF microneedling devices deliver the same RF or thermal energy via needles. RF, measured in Hertz (Hz) is part of the electromagnetic spectrum, with most devices delivering thermal energy at around 1-2 MHz, which is less than most typical RF only devices (at around 4-6 MHz), but with potentially more precise depth and delivery. For comparison, the RF of household electrical currents are around 60 Hz; traditional electrosurgical units, 50Hz -300 kHz; AM radio, 500 KHz; and microwaves, 2500 MHz.
When delivered to the skin, RF energy produces a change in the electrical charge of the skin, resulting in movement of electrons. The impedance (or resistance) of the tissue to the electron movement is what generates heat. Different factors, including tissue thickness, pressure applied to the tissue, hydration, bipolar versus monopolar delivery, and the number of needles are several factors than can affect the impedance.
Bipolar RF means that the current passes between two electrodes, whereas monopolar RF means that the electrical current is between an active treatment electrode and a passive grounding electrode (or grounding pad typically placed on the patient’s back). With bipolar RF, the current is limited to the area between the two electrodes. The depth of penetration is half of the distance between the electrodes, thus resulting in shallow (but potentially more aggressive) tissue heating. With monopolar RF, deeper tissue penetration occurs that is also often less uncomfortable to the patient.
The desired result of the energy delivery is collagen remodeling and strengthening of elastin. RF microneedling and microneedling in general may also have potential for use in enhancing topical product delivery.
Depending on the device, settings can be tailored to affect the energy delivery, including the type of needle (insulated vs. uninsulated vs. semi-insulated), Hz, number of needles, depth of needles, and time of exposure. In general, insulated needle tips provide less heat accumulation and potential injury to the skin surface, whereas uninsulated needles allow for more heat accumulation. Insulated needles, longer time of exposure, and lower energies (Hz) are safer options for darker skin types and those who hyperpigment easily.
Immediately after treatment, expected clinical endpoints can include erythema, edema, and possibly pinpoint bleeding that may last approximately several days to 2 weeks depending on the intensity of treatment. Potential side effects include infection, pigmentary alteration, folliculitis, prolonged grid marks, and scarring. Contraindications to treatment include having a pacemaker, history of keloid formation, active skin infections, prior gold threads in the treatment area, pregnancy and breastfeeding, metal implants in the treatment area, embedded electronic devices that cannot be turned off, isotretinoin use in the past 6 months, and allergy to any of the components of treatment.
Caution should be taken with tattoos in the treatment area or grounding pad (including cosmetic tattoos as tattoo ink may often contain metals that may absorb some of the heat, increasing the risk for injury or extrusion of the ink), a history of cold sores or herpes simplex virus in the treatment area (if so, a prophylactic antiviral would be indicated prior to treatment), use of topical retinoids in the past 7 days, having received neurotoxin or fillers in the prior 2 weeks, autoimmune disease, bleeding disorders, neuropathy, and history of poor healing.
Depending on the device and area being treated, most RF microneedling treatments require two to five treatments, typically 4-6 weeks apart. If improvement is seen, it may be noticeable after one to two treatments, and as with laser resurfacing, continued improvement may be noticeable over the following 6-12 months post treatment.
Dr. Wesley and Dr. Lily Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. Dr. Wesley has no relevant disclosures.
Technology in the field of aesthetic dermatology continues to advance over time. Microneedling, largely used to improve textural changes of the skin associated with photoaging and acne scarring, has evolved over time from the use of dermarollers and microneedling skin pens to energy-based devices that deliver radiofrequency (RF) energy though microneedles that are used today.
.
Unlike prior radiofrequency energy-based devices that deliver radiofrequency energy on the skin surface to allow bulk thermal energy (or heat) to stimulate collagen remodeling and tissue tightening, RF microneedling devices deliver the same RF or thermal energy via needles. RF, measured in Hertz (Hz) is part of the electromagnetic spectrum, with most devices delivering thermal energy at around 1-2 MHz, which is less than most typical RF only devices (at around 4-6 MHz), but with potentially more precise depth and delivery. For comparison, the RF of household electrical currents are around 60 Hz; traditional electrosurgical units, 50Hz -300 kHz; AM radio, 500 KHz; and microwaves, 2500 MHz.
When delivered to the skin, RF energy produces a change in the electrical charge of the skin, resulting in movement of electrons. The impedance (or resistance) of the tissue to the electron movement is what generates heat. Different factors, including tissue thickness, pressure applied to the tissue, hydration, bipolar versus monopolar delivery, and the number of needles are several factors than can affect the impedance.
Bipolar RF means that the current passes between two electrodes, whereas monopolar RF means that the electrical current is between an active treatment electrode and a passive grounding electrode (or grounding pad typically placed on the patient’s back). With bipolar RF, the current is limited to the area between the two electrodes. The depth of penetration is half of the distance between the electrodes, thus resulting in shallow (but potentially more aggressive) tissue heating. With monopolar RF, deeper tissue penetration occurs that is also often less uncomfortable to the patient.
The desired result of the energy delivery is collagen remodeling and strengthening of elastin. RF microneedling and microneedling in general may also have potential for use in enhancing topical product delivery.
Depending on the device, settings can be tailored to affect the energy delivery, including the type of needle (insulated vs. uninsulated vs. semi-insulated), Hz, number of needles, depth of needles, and time of exposure. In general, insulated needle tips provide less heat accumulation and potential injury to the skin surface, whereas uninsulated needles allow for more heat accumulation. Insulated needles, longer time of exposure, and lower energies (Hz) are safer options for darker skin types and those who hyperpigment easily.
Immediately after treatment, expected clinical endpoints can include erythema, edema, and possibly pinpoint bleeding that may last approximately several days to 2 weeks depending on the intensity of treatment. Potential side effects include infection, pigmentary alteration, folliculitis, prolonged grid marks, and scarring. Contraindications to treatment include having a pacemaker, history of keloid formation, active skin infections, prior gold threads in the treatment area, pregnancy and breastfeeding, metal implants in the treatment area, embedded electronic devices that cannot be turned off, isotretinoin use in the past 6 months, and allergy to any of the components of treatment.
Caution should be taken with tattoos in the treatment area or grounding pad (including cosmetic tattoos as tattoo ink may often contain metals that may absorb some of the heat, increasing the risk for injury or extrusion of the ink), a history of cold sores or herpes simplex virus in the treatment area (if so, a prophylactic antiviral would be indicated prior to treatment), use of topical retinoids in the past 7 days, having received neurotoxin or fillers in the prior 2 weeks, autoimmune disease, bleeding disorders, neuropathy, and history of poor healing.
Depending on the device and area being treated, most RF microneedling treatments require two to five treatments, typically 4-6 weeks apart. If improvement is seen, it may be noticeable after one to two treatments, and as with laser resurfacing, continued improvement may be noticeable over the following 6-12 months post treatment.
Dr. Wesley and Dr. Lily Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. Dr. Wesley has no relevant disclosures.
Technology in the field of aesthetic dermatology continues to advance over time. Microneedling, largely used to improve textural changes of the skin associated with photoaging and acne scarring, has evolved over time from the use of dermarollers and microneedling skin pens to energy-based devices that deliver radiofrequency (RF) energy though microneedles that are used today.
.
Unlike prior radiofrequency energy-based devices that deliver radiofrequency energy on the skin surface to allow bulk thermal energy (or heat) to stimulate collagen remodeling and tissue tightening, RF microneedling devices deliver the same RF or thermal energy via needles. RF, measured in Hertz (Hz) is part of the electromagnetic spectrum, with most devices delivering thermal energy at around 1-2 MHz, which is less than most typical RF only devices (at around 4-6 MHz), but with potentially more precise depth and delivery. For comparison, the RF of household electrical currents are around 60 Hz; traditional electrosurgical units, 50Hz -300 kHz; AM radio, 500 KHz; and microwaves, 2500 MHz.
When delivered to the skin, RF energy produces a change in the electrical charge of the skin, resulting in movement of electrons. The impedance (or resistance) of the tissue to the electron movement is what generates heat. Different factors, including tissue thickness, pressure applied to the tissue, hydration, bipolar versus monopolar delivery, and the number of needles are several factors than can affect the impedance.
Bipolar RF means that the current passes between two electrodes, whereas monopolar RF means that the electrical current is between an active treatment electrode and a passive grounding electrode (or grounding pad typically placed on the patient’s back). With bipolar RF, the current is limited to the area between the two electrodes. The depth of penetration is half of the distance between the electrodes, thus resulting in shallow (but potentially more aggressive) tissue heating. With monopolar RF, deeper tissue penetration occurs that is also often less uncomfortable to the patient.
The desired result of the energy delivery is collagen remodeling and strengthening of elastin. RF microneedling and microneedling in general may also have potential for use in enhancing topical product delivery.
Depending on the device, settings can be tailored to affect the energy delivery, including the type of needle (insulated vs. uninsulated vs. semi-insulated), Hz, number of needles, depth of needles, and time of exposure. In general, insulated needle tips provide less heat accumulation and potential injury to the skin surface, whereas uninsulated needles allow for more heat accumulation. Insulated needles, longer time of exposure, and lower energies (Hz) are safer options for darker skin types and those who hyperpigment easily.
Immediately after treatment, expected clinical endpoints can include erythema, edema, and possibly pinpoint bleeding that may last approximately several days to 2 weeks depending on the intensity of treatment. Potential side effects include infection, pigmentary alteration, folliculitis, prolonged grid marks, and scarring. Contraindications to treatment include having a pacemaker, history of keloid formation, active skin infections, prior gold threads in the treatment area, pregnancy and breastfeeding, metal implants in the treatment area, embedded electronic devices that cannot be turned off, isotretinoin use in the past 6 months, and allergy to any of the components of treatment.
Caution should be taken with tattoos in the treatment area or grounding pad (including cosmetic tattoos as tattoo ink may often contain metals that may absorb some of the heat, increasing the risk for injury or extrusion of the ink), a history of cold sores or herpes simplex virus in the treatment area (if so, a prophylactic antiviral would be indicated prior to treatment), use of topical retinoids in the past 7 days, having received neurotoxin or fillers in the prior 2 weeks, autoimmune disease, bleeding disorders, neuropathy, and history of poor healing.
Depending on the device and area being treated, most RF microneedling treatments require two to five treatments, typically 4-6 weeks apart. If improvement is seen, it may be noticeable after one to two treatments, and as with laser resurfacing, continued improvement may be noticeable over the following 6-12 months post treatment.
Dr. Wesley and Dr. Lily Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. Dr. Wesley has no relevant disclosures.