User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Sleep disturbances more profound in older adults with atopic dermatitis
especially trouble staying asleep.
Those are key findings from a cross-sectional study that Jaya Manjunath, BS, and Jonathan I. Silverberg, MD, PhD, MPH, presented during a poster session at the Revolutionizing Atopic Dermatitis symposium.
“Atopic dermatitis is a chronic, pruritic skin disease associated with sleep disturbance and fatigue affecting adults of all ages,” they wrote. “When caring for geriatric patients, several factors such as sleep disturbance, polypharmacy, cognition, social support, and mobility should be considered. However, little is known about the characteristics of atopic dermatitis in the geriatric population.”
Ms. Manjunath, a student at George Washington University, Washington, and Dr. Silverberg, director of clinical research in the department of dermatology at GWU, recruited patients with AD aged 18 years and older diagnosed by Hanifin-Rajka criteria who were evaluated at an academic medical center between 2014 and 2019. They underwent full body skin exams and completed electronic questionnaires. AD severity was assessed with the Eczema Area and Severity Index (EASI), Scoring Atopic Dermatitis (SCORAD) total and itch subscores, Investigator’s Global Assessment (IGA), patient-reported Global Assessment of AD severity, and the Patient-Oriented Eczema Measure (POEM).
The researchers also assessed the frequency of sleep disturbances, including difficulty falling asleep and staying asleep, and used multivariable logistic regression models to evaluate associations of age (65 and older vs. 18-64 years) with AD severity, sleep disturbance or fatigue, controlling for total POEM score, sex, and race.
Using adjusted odds ratios, Ms. Manjunath and Dr. Silverberg found that being 65 or older was not associated with AD severity on the EASI (adjusted odds ratio, 1.47); total SCORAD (aOR, 1.10), and itch subscore (aOR, 1.00); IGA (aOR, 1.87); patient-reported Global Assessment of AD severity (aOR, 0.80), or the patient-oriented eczema measure (aOR, 0.55), associations that were not statistically significant.
However, the researchers found that older adult age was associated with an increased number of nights of sleep disturbance from AD in the past week (aOR, 2.14; P = .0142), as well as increased fatigue in the past 7 days (aOR, 1.81; P = .0313), trouble sleeping in the past 7 days (aOR, 1.98; P = .0118), and trouble staying asleep in the past 7 days (aOR, 2.26; P = .0030), but not with difficulty falling asleep in the last 7 days (aOR, 1.16; P = .5996).
“Future studies are needed to determine why geriatric AD patients have increased sleep disturbance and optimal interventions to address their sleep disturbance,” the researchers concluded.
The study was supported by the Agency for Healthcare Research and Quality, the Dermatology Foundation, and by an unrestricted grant from Galderma. Ms. Manjunath disclosed no relevant financial relationships. Dr. Silverberg reported that he is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
A version of this article first appeared on Medscape.com.
especially trouble staying asleep.
Those are key findings from a cross-sectional study that Jaya Manjunath, BS, and Jonathan I. Silverberg, MD, PhD, MPH, presented during a poster session at the Revolutionizing Atopic Dermatitis symposium.
“Atopic dermatitis is a chronic, pruritic skin disease associated with sleep disturbance and fatigue affecting adults of all ages,” they wrote. “When caring for geriatric patients, several factors such as sleep disturbance, polypharmacy, cognition, social support, and mobility should be considered. However, little is known about the characteristics of atopic dermatitis in the geriatric population.”
Ms. Manjunath, a student at George Washington University, Washington, and Dr. Silverberg, director of clinical research in the department of dermatology at GWU, recruited patients with AD aged 18 years and older diagnosed by Hanifin-Rajka criteria who were evaluated at an academic medical center between 2014 and 2019. They underwent full body skin exams and completed electronic questionnaires. AD severity was assessed with the Eczema Area and Severity Index (EASI), Scoring Atopic Dermatitis (SCORAD) total and itch subscores, Investigator’s Global Assessment (IGA), patient-reported Global Assessment of AD severity, and the Patient-Oriented Eczema Measure (POEM).
The researchers also assessed the frequency of sleep disturbances, including difficulty falling asleep and staying asleep, and used multivariable logistic regression models to evaluate associations of age (65 and older vs. 18-64 years) with AD severity, sleep disturbance or fatigue, controlling for total POEM score, sex, and race.
Using adjusted odds ratios, Ms. Manjunath and Dr. Silverberg found that being 65 or older was not associated with AD severity on the EASI (adjusted odds ratio, 1.47); total SCORAD (aOR, 1.10), and itch subscore (aOR, 1.00); IGA (aOR, 1.87); patient-reported Global Assessment of AD severity (aOR, 0.80), or the patient-oriented eczema measure (aOR, 0.55), associations that were not statistically significant.
However, the researchers found that older adult age was associated with an increased number of nights of sleep disturbance from AD in the past week (aOR, 2.14; P = .0142), as well as increased fatigue in the past 7 days (aOR, 1.81; P = .0313), trouble sleeping in the past 7 days (aOR, 1.98; P = .0118), and trouble staying asleep in the past 7 days (aOR, 2.26; P = .0030), but not with difficulty falling asleep in the last 7 days (aOR, 1.16; P = .5996).
“Future studies are needed to determine why geriatric AD patients have increased sleep disturbance and optimal interventions to address their sleep disturbance,” the researchers concluded.
The study was supported by the Agency for Healthcare Research and Quality, the Dermatology Foundation, and by an unrestricted grant from Galderma. Ms. Manjunath disclosed no relevant financial relationships. Dr. Silverberg reported that he is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
A version of this article first appeared on Medscape.com.
especially trouble staying asleep.
Those are key findings from a cross-sectional study that Jaya Manjunath, BS, and Jonathan I. Silverberg, MD, PhD, MPH, presented during a poster session at the Revolutionizing Atopic Dermatitis symposium.
“Atopic dermatitis is a chronic, pruritic skin disease associated with sleep disturbance and fatigue affecting adults of all ages,” they wrote. “When caring for geriatric patients, several factors such as sleep disturbance, polypharmacy, cognition, social support, and mobility should be considered. However, little is known about the characteristics of atopic dermatitis in the geriatric population.”
Ms. Manjunath, a student at George Washington University, Washington, and Dr. Silverberg, director of clinical research in the department of dermatology at GWU, recruited patients with AD aged 18 years and older diagnosed by Hanifin-Rajka criteria who were evaluated at an academic medical center between 2014 and 2019. They underwent full body skin exams and completed electronic questionnaires. AD severity was assessed with the Eczema Area and Severity Index (EASI), Scoring Atopic Dermatitis (SCORAD) total and itch subscores, Investigator’s Global Assessment (IGA), patient-reported Global Assessment of AD severity, and the Patient-Oriented Eczema Measure (POEM).
The researchers also assessed the frequency of sleep disturbances, including difficulty falling asleep and staying asleep, and used multivariable logistic regression models to evaluate associations of age (65 and older vs. 18-64 years) with AD severity, sleep disturbance or fatigue, controlling for total POEM score, sex, and race.
Using adjusted odds ratios, Ms. Manjunath and Dr. Silverberg found that being 65 or older was not associated with AD severity on the EASI (adjusted odds ratio, 1.47); total SCORAD (aOR, 1.10), and itch subscore (aOR, 1.00); IGA (aOR, 1.87); patient-reported Global Assessment of AD severity (aOR, 0.80), or the patient-oriented eczema measure (aOR, 0.55), associations that were not statistically significant.
However, the researchers found that older adult age was associated with an increased number of nights of sleep disturbance from AD in the past week (aOR, 2.14; P = .0142), as well as increased fatigue in the past 7 days (aOR, 1.81; P = .0313), trouble sleeping in the past 7 days (aOR, 1.98; P = .0118), and trouble staying asleep in the past 7 days (aOR, 2.26; P = .0030), but not with difficulty falling asleep in the last 7 days (aOR, 1.16; P = .5996).
“Future studies are needed to determine why geriatric AD patients have increased sleep disturbance and optimal interventions to address their sleep disturbance,” the researchers concluded.
The study was supported by the Agency for Healthcare Research and Quality, the Dermatology Foundation, and by an unrestricted grant from Galderma. Ms. Manjunath disclosed no relevant financial relationships. Dr. Silverberg reported that he is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
A version of this article first appeared on Medscape.com.
FROM REVOLUTIONIZING AD 2021
Secretan Syndrome: A Fluctuating Case of Factitious Lymphedema
Secretan syndrome (SS) represents a recurrent or chronic form of factitious lymphedema, usually affecting the dorsal aspect of the hand.1-3 It is accepted as a subtype of Munchausen syndrome whereby the patient self-inflicts and simulates lymphedema.1,2 Historically, many of the cases reported with the term Charcot’s oedème bleu are now believed to represent clinical variants of SS.4-6
Case Report
A 38-year-old Turkish woman presented with progressive swelling of the right hand of 2 years’ duration that had caused difficulty in manual work and reduction in manual dexterity. She previously had sought medical treatment for this condition by visiting several hospitals. According to her medical record, the following laboratory or radiologic tests had revealed negative or normal findings, except for obvious soft-tissue edema: bacterial and fungal cultures, plain radiography, Doppler ultrasonography, lymphoscintigraphy, magnetic resonance imaging, fine needle aspiration, and punch biopsy. Reflex sympathetic dystrophy, compartment syndrome, filariasis, tuberculosis, and lymphatic and venous obstruction were all excluded by appropriate testing. Our patient was in good health prior to onset of this disorder, and her medical history was unremarkable. There was no family history of a similar condition.
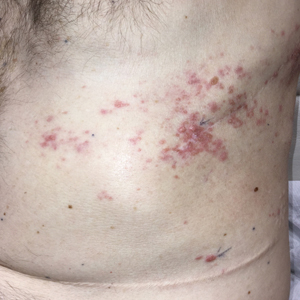
Dermatologic examination revealed brawny, soft, pitting edema; erythema; and crusts affecting the dorsal aspect of the right hand and proximal parts of the fingers (Figure 1). The yellow discoloration of the skin and nails was attributed to potassium permanganate wet dressings. Under an elastic bandage at the wrist, which the patient unrolled herself, a sharp line of demarcation was evident, separating the lymphedematous and normal parts of the arm. There was no axillary lymphadenopathy.

The patient’s affect was discordant to the manifestation of the cutaneous findings. She wanted to show every physician in the department how swollen her hand was and seemed to be happy with this condition. Although she displayed no signs of disturbance when the affected extremity was touched or handled, she reported severe pain and tenderness as well as difficulty in housework. She noted that she normally resided in a city and that the swelling had started at the time she had relocated to a rural village to take care of her bedridden mother-in-law. She was under an intensive workload in the village, and the condition of the hand was impeding manual work.
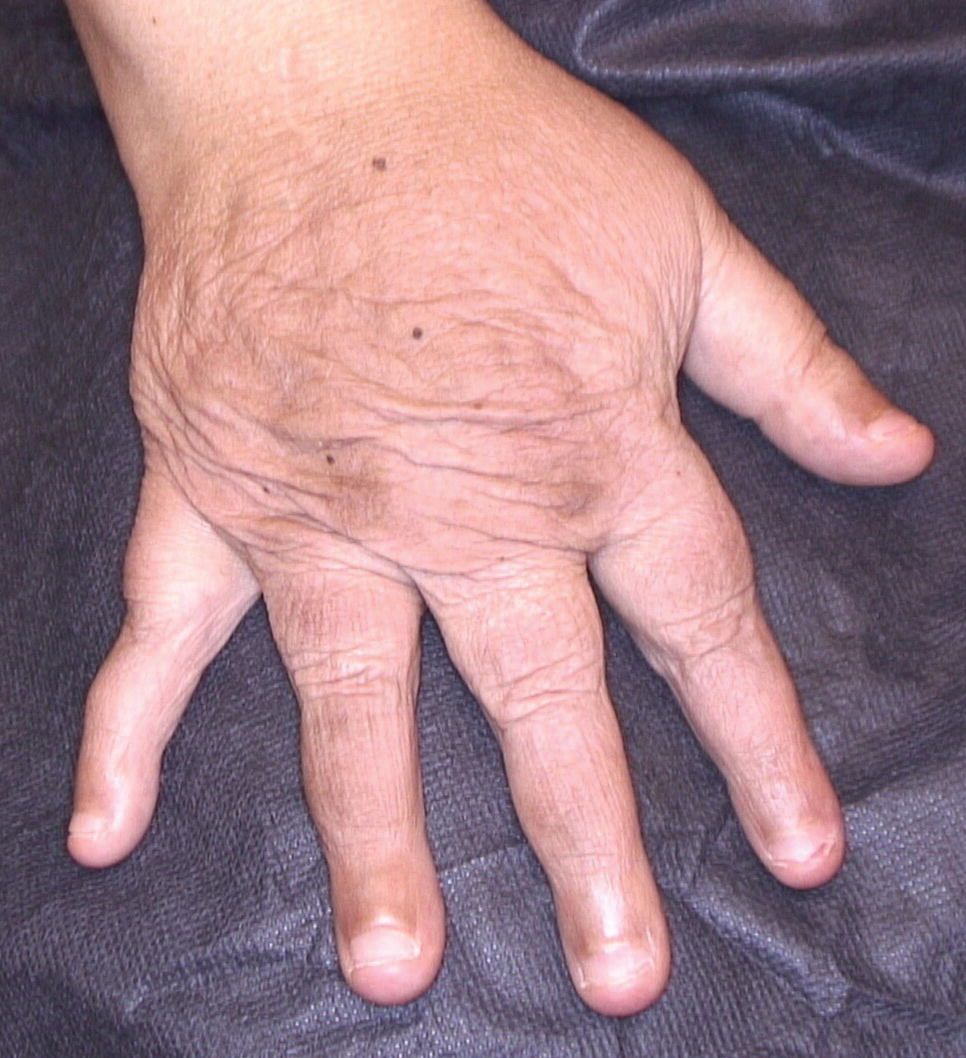
Factitious lymphedema was considered, and hospitalization was recommended. The patient was then lost to follow-up; however, one of her relatives noted that the patient had returned to the city. When she presented again 1 year later, almost all physical signs had disappeared (Figure 2), and a psychiatric referral was recommended. A Minnesota Multiphasic Personality Inventory test yielded an invalid result due to the patient’s exaggeration of her preexisting physical symptoms. Further psychiatric workup was rejected by the patient.
Almost a year after the psychiatric referral, the patient’s follow-up photographs revealed that the lymphedema recurred when she went to visit her mother-in-law in the rural village and that it was completely ameliorated when she returned to the city. Thus, a positive “mother-in-law provocation test” was accepted as final proof of the self-inflicted nature of the condition.
Comment
In 1901, Henri Francois Secretan, a Swiss physician, reported workmen who had persistent hard swellings on the dorsal aspect of the hands after minor work-related trauma for which they had compensation claims.7 In his original report, Secretan did not suggest self-inflicted trauma in the etiology of this disorder.5,8,9 In 1890, Jean Martin Charcot, a French neurologist, described oedème bleu, a term that is now believed to denote a condition similar to SS.4-6 Currently, SS is attributed to self-inflicted injury and is considered a form of factitious lymphedema.9 As in dermatitis artefacta, most patients with SS are young women, and male patients with the condition tend to be older.3,8
The mechanism used to provoke this factitious lymphedema might be of traumatic or obstructive nature. Secretan syndrome either is induced by intermittent or constant application of a tourniquet, ligature, cord, elastic bandage, scarf, kerchief, rubber band, or compress around the affected extremity, or by repetitive blunt trauma, force, or skin irritation.1,4,5,8-10 There was an underlying psychopathology in all reported cases.1,8,11 Factitious lymphedema is unconsciously motivated and consciously produced.4,12 The affected patient often is experiencing a serious emotional conflict and is unlikely to be a malingerer, although exaggeration of symptoms may occur, as in our patient.12 Psychiatric evaluation in SS may uncover neurosis, hysteria, frank psychosis, schizophrenia, masochism, depression, or an abnormal personality disorder.1,12
Patients with SS present with recurrent or chronic lymphedema, usually affecting the dominant hand.1 Involvement usually is unilateral; bilateral cases are rare.3,6 Secretan syndrome is not solely limited to the hands; it also may involve the upper and lower extremities, including the feet.3,11 There may be a clear line of demarcation, a ring, sulcus, distinct circumferential linear bands of erythema, discoloration, or ecchymoses, separating the normal and lymphedematous parts of the extremity.1,4,6,8-10,12 Patients usually attempt to hide the constricted areas from sight.1 Over time, flexion contractures may develop due to peritendinous fibrosis.6 Histopathology displays a hematoma with adhesions to the extensor tendons; a hematoma surrounded by a thickened scar; or changes similar to ganglion tissue with cystic areas of mucin, fibrosis, and myxoid degeneration.4,6
Factitious lymphedema can only be definitively diagnosed when the patient confesses or is caught self-inflicting the injury. Nevertheless, a diagnosis by exclusion is possible.4 Lymphangiography, lymphoscintigraphy, vascular Doppler ultrasonography, and magnetic resonance imaging may be helpful in excluding congenital and acquired causes of lymphedema and venous obstruction.1,3,9,11 Magnetic resonance imaging may show soft tissue and tendon edema as well as diffuse peritendinous fibrosis extending to the fascia of the dorsal interosseous muscles.3,4
Factitious lymphedema should be suspected in all patients with recurrent or chronic unilateral lymphedema without an explicable or apparent predisposing factor.4,11,12 Patients with SS typically visit several hospitals or institutions; see many physicians; and willingly accept, request, and undergo unnecessary extensive, invasive, and costly diagnostic and therapeutic procedures and prolonged hospitalizations.1,2,5,12 The disorder promptly responds to immobilization and elevation of the limb.2,4 Plaster casts may prove useful in prevention of compression and thus amelioration of the lymphedema.1,4,6 Once the diagnosis is confirmed, direct confrontation should be avoided and ideally the patient should be referred for psychiatric evaluation.1,2,4,5,8,12 If the patient admits self-inflicting behavior, psychotherapy and/or behavior modification therapy along with psychotropic medications may be helpful to relieve emotional and behavioral symptoms.12 Unfortunately, if the patient denies a self-inflicting role in the occurrence of lymphedema and persists in self-injurious behavior, psychotherapy or psychotropic medications will be futile.9
1. Miyamoto Y, Hamanaka T, Yokoyama S, et al. Factitious lymphedema of the upper limb. Kawasaki Med J. 1979;5:39-45.
2. de Oliveira RK, Bayer LR, Lauxen D, et al. Factitious lesions of the hand. Rev Bras Ortop. 2013;48:381-386.
3. Hahm MH, Yi JH. A case report of Secretan’s disease in both hands. J Korean Soc Radiol. 2013;68:511-514.
4. Eldridge MP, Grunert BK, Matloub HS. Streamlined classification of psychopathological hand disorders: a literature review. Hand (NY). 2008;3:118-128.
5. Ostlere LS, Harris D, Denton C, et al. Boxing-glove hand: an unusual presentation of dermatitis artefacta. J Am Acad Dermatol. 1993;28:120-122.
6. Winkelmann RK, Barker SM. Factitial traumatic panniculitis. J Am Acad Dermatol. 1985;13:988-994.
7. Secretan H. Oederne dur et hyperplasie traumatique du metacarpe dorsal. RevMed Suisse Romande. 1901;21:409-416.
8. Barth JH, Pegum JS. The case of the speckled band: acquired lymphedema due to constriction bands. J Am Acad Dermatol. 1986;15:296-297.
9. Birman MV, Lee DH. Factitious disorders of the upper extremity. J Am Acad Orthop Surg. 2012;20:78-85.
10. Nwaejike N, Archbold H, Wilson DS. Factitious lymphoedema as a psychiatric condition mimicking reflex sympathetic dystrophy: a case report. J Med Case Rep. 2008;2:216.
11. De Fátima Guerreiro Godoy M, Pereira De Godoy JM. Factitious lymphedema of the arm: case report and review of publications. Eur J Phys Rehabil Med. 2015;51:337-339.
12. Abhari SAA, Alimalayeri N, Abhari SSA, et al. Factitious lymphedema of the hand. Iran J Psychiatry. 2006;1:166-168.
Secretan syndrome (SS) represents a recurrent or chronic form of factitious lymphedema, usually affecting the dorsal aspect of the hand.1-3 It is accepted as a subtype of Munchausen syndrome whereby the patient self-inflicts and simulates lymphedema.1,2 Historically, many of the cases reported with the term Charcot’s oedème bleu are now believed to represent clinical variants of SS.4-6
Case Report
A 38-year-old Turkish woman presented with progressive swelling of the right hand of 2 years’ duration that had caused difficulty in manual work and reduction in manual dexterity. She previously had sought medical treatment for this condition by visiting several hospitals. According to her medical record, the following laboratory or radiologic tests had revealed negative or normal findings, except for obvious soft-tissue edema: bacterial and fungal cultures, plain radiography, Doppler ultrasonography, lymphoscintigraphy, magnetic resonance imaging, fine needle aspiration, and punch biopsy. Reflex sympathetic dystrophy, compartment syndrome, filariasis, tuberculosis, and lymphatic and venous obstruction were all excluded by appropriate testing. Our patient was in good health prior to onset of this disorder, and her medical history was unremarkable. There was no family history of a similar condition.
Dermatologic examination revealed brawny, soft, pitting edema; erythema; and crusts affecting the dorsal aspect of the right hand and proximal parts of the fingers (Figure 1). The yellow discoloration of the skin and nails was attributed to potassium permanganate wet dressings. Under an elastic bandage at the wrist, which the patient unrolled herself, a sharp line of demarcation was evident, separating the lymphedematous and normal parts of the arm. There was no axillary lymphadenopathy.

The patient’s affect was discordant to the manifestation of the cutaneous findings. She wanted to show every physician in the department how swollen her hand was and seemed to be happy with this condition. Although she displayed no signs of disturbance when the affected extremity was touched or handled, she reported severe pain and tenderness as well as difficulty in housework. She noted that she normally resided in a city and that the swelling had started at the time she had relocated to a rural village to take care of her bedridden mother-in-law. She was under an intensive workload in the village, and the condition of the hand was impeding manual work.
Factitious lymphedema was considered, and hospitalization was recommended. The patient was then lost to follow-up; however, one of her relatives noted that the patient had returned to the city. When she presented again 1 year later, almost all physical signs had disappeared (Figure 2), and a psychiatric referral was recommended. A Minnesota Multiphasic Personality Inventory test yielded an invalid result due to the patient’s exaggeration of her preexisting physical symptoms. Further psychiatric workup was rejected by the patient.

Almost a year after the psychiatric referral, the patient’s follow-up photographs revealed that the lymphedema recurred when she went to visit her mother-in-law in the rural village and that it was completely ameliorated when she returned to the city. Thus, a positive “mother-in-law provocation test” was accepted as final proof of the self-inflicted nature of the condition.
Comment
In 1901, Henri Francois Secretan, a Swiss physician, reported workmen who had persistent hard swellings on the dorsal aspect of the hands after minor work-related trauma for which they had compensation claims.7 In his original report, Secretan did not suggest self-inflicted trauma in the etiology of this disorder.5,8,9 In 1890, Jean Martin Charcot, a French neurologist, described oedème bleu, a term that is now believed to denote a condition similar to SS.4-6 Currently, SS is attributed to self-inflicted injury and is considered a form of factitious lymphedema.9 As in dermatitis artefacta, most patients with SS are young women, and male patients with the condition tend to be older.3,8
The mechanism used to provoke this factitious lymphedema might be of traumatic or obstructive nature. Secretan syndrome either is induced by intermittent or constant application of a tourniquet, ligature, cord, elastic bandage, scarf, kerchief, rubber band, or compress around the affected extremity, or by repetitive blunt trauma, force, or skin irritation.1,4,5,8-10 There was an underlying psychopathology in all reported cases.1,8,11 Factitious lymphedema is unconsciously motivated and consciously produced.4,12 The affected patient often is experiencing a serious emotional conflict and is unlikely to be a malingerer, although exaggeration of symptoms may occur, as in our patient.12 Psychiatric evaluation in SS may uncover neurosis, hysteria, frank psychosis, schizophrenia, masochism, depression, or an abnormal personality disorder.1,12
Patients with SS present with recurrent or chronic lymphedema, usually affecting the dominant hand.1 Involvement usually is unilateral; bilateral cases are rare.3,6 Secretan syndrome is not solely limited to the hands; it also may involve the upper and lower extremities, including the feet.3,11 There may be a clear line of demarcation, a ring, sulcus, distinct circumferential linear bands of erythema, discoloration, or ecchymoses, separating the normal and lymphedematous parts of the extremity.1,4,6,8-10,12 Patients usually attempt to hide the constricted areas from sight.1 Over time, flexion contractures may develop due to peritendinous fibrosis.6 Histopathology displays a hematoma with adhesions to the extensor tendons; a hematoma surrounded by a thickened scar; or changes similar to ganglion tissue with cystic areas of mucin, fibrosis, and myxoid degeneration.4,6
Factitious lymphedema can only be definitively diagnosed when the patient confesses or is caught self-inflicting the injury. Nevertheless, a diagnosis by exclusion is possible.4 Lymphangiography, lymphoscintigraphy, vascular Doppler ultrasonography, and magnetic resonance imaging may be helpful in excluding congenital and acquired causes of lymphedema and venous obstruction.1,3,9,11 Magnetic resonance imaging may show soft tissue and tendon edema as well as diffuse peritendinous fibrosis extending to the fascia of the dorsal interosseous muscles.3,4
Factitious lymphedema should be suspected in all patients with recurrent or chronic unilateral lymphedema without an explicable or apparent predisposing factor.4,11,12 Patients with SS typically visit several hospitals or institutions; see many physicians; and willingly accept, request, and undergo unnecessary extensive, invasive, and costly diagnostic and therapeutic procedures and prolonged hospitalizations.1,2,5,12 The disorder promptly responds to immobilization and elevation of the limb.2,4 Plaster casts may prove useful in prevention of compression and thus amelioration of the lymphedema.1,4,6 Once the diagnosis is confirmed, direct confrontation should be avoided and ideally the patient should be referred for psychiatric evaluation.1,2,4,5,8,12 If the patient admits self-inflicting behavior, psychotherapy and/or behavior modification therapy along with psychotropic medications may be helpful to relieve emotional and behavioral symptoms.12 Unfortunately, if the patient denies a self-inflicting role in the occurrence of lymphedema and persists in self-injurious behavior, psychotherapy or psychotropic medications will be futile.9
Secretan syndrome (SS) represents a recurrent or chronic form of factitious lymphedema, usually affecting the dorsal aspect of the hand.1-3 It is accepted as a subtype of Munchausen syndrome whereby the patient self-inflicts and simulates lymphedema.1,2 Historically, many of the cases reported with the term Charcot’s oedème bleu are now believed to represent clinical variants of SS.4-6
Case Report
A 38-year-old Turkish woman presented with progressive swelling of the right hand of 2 years’ duration that had caused difficulty in manual work and reduction in manual dexterity. She previously had sought medical treatment for this condition by visiting several hospitals. According to her medical record, the following laboratory or radiologic tests had revealed negative or normal findings, except for obvious soft-tissue edema: bacterial and fungal cultures, plain radiography, Doppler ultrasonography, lymphoscintigraphy, magnetic resonance imaging, fine needle aspiration, and punch biopsy. Reflex sympathetic dystrophy, compartment syndrome, filariasis, tuberculosis, and lymphatic and venous obstruction were all excluded by appropriate testing. Our patient was in good health prior to onset of this disorder, and her medical history was unremarkable. There was no family history of a similar condition.
Dermatologic examination revealed brawny, soft, pitting edema; erythema; and crusts affecting the dorsal aspect of the right hand and proximal parts of the fingers (Figure 1). The yellow discoloration of the skin and nails was attributed to potassium permanganate wet dressings. Under an elastic bandage at the wrist, which the patient unrolled herself, a sharp line of demarcation was evident, separating the lymphedematous and normal parts of the arm. There was no axillary lymphadenopathy.

The patient’s affect was discordant to the manifestation of the cutaneous findings. She wanted to show every physician in the department how swollen her hand was and seemed to be happy with this condition. Although she displayed no signs of disturbance when the affected extremity was touched or handled, she reported severe pain and tenderness as well as difficulty in housework. She noted that she normally resided in a city and that the swelling had started at the time she had relocated to a rural village to take care of her bedridden mother-in-law. She was under an intensive workload in the village, and the condition of the hand was impeding manual work.
Factitious lymphedema was considered, and hospitalization was recommended. The patient was then lost to follow-up; however, one of her relatives noted that the patient had returned to the city. When she presented again 1 year later, almost all physical signs had disappeared (Figure 2), and a psychiatric referral was recommended. A Minnesota Multiphasic Personality Inventory test yielded an invalid result due to the patient’s exaggeration of her preexisting physical symptoms. Further psychiatric workup was rejected by the patient.

Almost a year after the psychiatric referral, the patient’s follow-up photographs revealed that the lymphedema recurred when she went to visit her mother-in-law in the rural village and that it was completely ameliorated when she returned to the city. Thus, a positive “mother-in-law provocation test” was accepted as final proof of the self-inflicted nature of the condition.
Comment
In 1901, Henri Francois Secretan, a Swiss physician, reported workmen who had persistent hard swellings on the dorsal aspect of the hands after minor work-related trauma for which they had compensation claims.7 In his original report, Secretan did not suggest self-inflicted trauma in the etiology of this disorder.5,8,9 In 1890, Jean Martin Charcot, a French neurologist, described oedème bleu, a term that is now believed to denote a condition similar to SS.4-6 Currently, SS is attributed to self-inflicted injury and is considered a form of factitious lymphedema.9 As in dermatitis artefacta, most patients with SS are young women, and male patients with the condition tend to be older.3,8
The mechanism used to provoke this factitious lymphedema might be of traumatic or obstructive nature. Secretan syndrome either is induced by intermittent or constant application of a tourniquet, ligature, cord, elastic bandage, scarf, kerchief, rubber band, or compress around the affected extremity, or by repetitive blunt trauma, force, or skin irritation.1,4,5,8-10 There was an underlying psychopathology in all reported cases.1,8,11 Factitious lymphedema is unconsciously motivated and consciously produced.4,12 The affected patient often is experiencing a serious emotional conflict and is unlikely to be a malingerer, although exaggeration of symptoms may occur, as in our patient.12 Psychiatric evaluation in SS may uncover neurosis, hysteria, frank psychosis, schizophrenia, masochism, depression, or an abnormal personality disorder.1,12
Patients with SS present with recurrent or chronic lymphedema, usually affecting the dominant hand.1 Involvement usually is unilateral; bilateral cases are rare.3,6 Secretan syndrome is not solely limited to the hands; it also may involve the upper and lower extremities, including the feet.3,11 There may be a clear line of demarcation, a ring, sulcus, distinct circumferential linear bands of erythema, discoloration, or ecchymoses, separating the normal and lymphedematous parts of the extremity.1,4,6,8-10,12 Patients usually attempt to hide the constricted areas from sight.1 Over time, flexion contractures may develop due to peritendinous fibrosis.6 Histopathology displays a hematoma with adhesions to the extensor tendons; a hematoma surrounded by a thickened scar; or changes similar to ganglion tissue with cystic areas of mucin, fibrosis, and myxoid degeneration.4,6
Factitious lymphedema can only be definitively diagnosed when the patient confesses or is caught self-inflicting the injury. Nevertheless, a diagnosis by exclusion is possible.4 Lymphangiography, lymphoscintigraphy, vascular Doppler ultrasonography, and magnetic resonance imaging may be helpful in excluding congenital and acquired causes of lymphedema and venous obstruction.1,3,9,11 Magnetic resonance imaging may show soft tissue and tendon edema as well as diffuse peritendinous fibrosis extending to the fascia of the dorsal interosseous muscles.3,4
Factitious lymphedema should be suspected in all patients with recurrent or chronic unilateral lymphedema without an explicable or apparent predisposing factor.4,11,12 Patients with SS typically visit several hospitals or institutions; see many physicians; and willingly accept, request, and undergo unnecessary extensive, invasive, and costly diagnostic and therapeutic procedures and prolonged hospitalizations.1,2,5,12 The disorder promptly responds to immobilization and elevation of the limb.2,4 Plaster casts may prove useful in prevention of compression and thus amelioration of the lymphedema.1,4,6 Once the diagnosis is confirmed, direct confrontation should be avoided and ideally the patient should be referred for psychiatric evaluation.1,2,4,5,8,12 If the patient admits self-inflicting behavior, psychotherapy and/or behavior modification therapy along with psychotropic medications may be helpful to relieve emotional and behavioral symptoms.12 Unfortunately, if the patient denies a self-inflicting role in the occurrence of lymphedema and persists in self-injurious behavior, psychotherapy or psychotropic medications will be futile.9
1. Miyamoto Y, Hamanaka T, Yokoyama S, et al. Factitious lymphedema of the upper limb. Kawasaki Med J. 1979;5:39-45.
2. de Oliveira RK, Bayer LR, Lauxen D, et al. Factitious lesions of the hand. Rev Bras Ortop. 2013;48:381-386.
3. Hahm MH, Yi JH. A case report of Secretan’s disease in both hands. J Korean Soc Radiol. 2013;68:511-514.
4. Eldridge MP, Grunert BK, Matloub HS. Streamlined classification of psychopathological hand disorders: a literature review. Hand (NY). 2008;3:118-128.
5. Ostlere LS, Harris D, Denton C, et al. Boxing-glove hand: an unusual presentation of dermatitis artefacta. J Am Acad Dermatol. 1993;28:120-122.
6. Winkelmann RK, Barker SM. Factitial traumatic panniculitis. J Am Acad Dermatol. 1985;13:988-994.
7. Secretan H. Oederne dur et hyperplasie traumatique du metacarpe dorsal. RevMed Suisse Romande. 1901;21:409-416.
8. Barth JH, Pegum JS. The case of the speckled band: acquired lymphedema due to constriction bands. J Am Acad Dermatol. 1986;15:296-297.
9. Birman MV, Lee DH. Factitious disorders of the upper extremity. J Am Acad Orthop Surg. 2012;20:78-85.
10. Nwaejike N, Archbold H, Wilson DS. Factitious lymphoedema as a psychiatric condition mimicking reflex sympathetic dystrophy: a case report. J Med Case Rep. 2008;2:216.
11. De Fátima Guerreiro Godoy M, Pereira De Godoy JM. Factitious lymphedema of the arm: case report and review of publications. Eur J Phys Rehabil Med. 2015;51:337-339.
12. Abhari SAA, Alimalayeri N, Abhari SSA, et al. Factitious lymphedema of the hand. Iran J Psychiatry. 2006;1:166-168.
1. Miyamoto Y, Hamanaka T, Yokoyama S, et al. Factitious lymphedema of the upper limb. Kawasaki Med J. 1979;5:39-45.
2. de Oliveira RK, Bayer LR, Lauxen D, et al. Factitious lesions of the hand. Rev Bras Ortop. 2013;48:381-386.
3. Hahm MH, Yi JH. A case report of Secretan’s disease in both hands. J Korean Soc Radiol. 2013;68:511-514.
4. Eldridge MP, Grunert BK, Matloub HS. Streamlined classification of psychopathological hand disorders: a literature review. Hand (NY). 2008;3:118-128.
5. Ostlere LS, Harris D, Denton C, et al. Boxing-glove hand: an unusual presentation of dermatitis artefacta. J Am Acad Dermatol. 1993;28:120-122.
6. Winkelmann RK, Barker SM. Factitial traumatic panniculitis. J Am Acad Dermatol. 1985;13:988-994.
7. Secretan H. Oederne dur et hyperplasie traumatique du metacarpe dorsal. RevMed Suisse Romande. 1901;21:409-416.
8. Barth JH, Pegum JS. The case of the speckled band: acquired lymphedema due to constriction bands. J Am Acad Dermatol. 1986;15:296-297.
9. Birman MV, Lee DH. Factitious disorders of the upper extremity. J Am Acad Orthop Surg. 2012;20:78-85.
10. Nwaejike N, Archbold H, Wilson DS. Factitious lymphoedema as a psychiatric condition mimicking reflex sympathetic dystrophy: a case report. J Med Case Rep. 2008;2:216.
11. De Fátima Guerreiro Godoy M, Pereira De Godoy JM. Factitious lymphedema of the arm: case report and review of publications. Eur J Phys Rehabil Med. 2015;51:337-339.
12. Abhari SAA, Alimalayeri N, Abhari SSA, et al. Factitious lymphedema of the hand. Iran J Psychiatry. 2006;1:166-168.
Practice Points
- Secretan syndrome is a recurrent or chronic form of factitious lymphedema that usually affects the dorsal aspect of the hand; it is accepted as a subtype of Munchausen syndrome.
- Secretan syndrome usually is induced by compression of the extremity by tourniquets, ligatures, cords, or similar equipment.
- This unconsciously motivated and consciously produced lymphedema is an expression of underlying psychiatric disease.
Skin imaging working group releases first guidelines for AI algorithms used in dermatology
The
The guidelines, published in JAMA Dermatology on Dec. 1, 2021, contain a broad range of recommendations stakeholders should consider when developing and assessing image-based AI algorithms in dermatology. The recommendations are divided into categories of data, technique, technical assessment, and application. ISIC is “an academia and industry partnership designed to facilitate the application of digital skin imaging to help reduce melanoma mortality,” and is organized into different working groups, including the AI working group, according to its website.
“Our goal with these guidelines was to create higher-quality reporting of dataset and algorithm characteristics for dermatology AI,” first author Roxana Daneshjou, MD, PhD, clinical scholar in dermatology, in the department of dermatology at Stanford (Calif.) University, said in an interview. “We hope these guidelines also aid regulatory bodies around the world when they are assessing algorithms to be used in dermatology.”
Recommendations for data
The authors recommended that datasets used by AI algorithms have image descriptions and details on image artifacts. “For photography, these include the type of camera used; whether images were taken under standardized or varying conditions; whether they were taken by professional photographers, laymen, or health care professionals; and image quality,” they wrote. They also recommended that developers include in an image description the type of lighting used and whether the photo contains pen markings, hair, tattoos, injuries, surgical effects, or other “physical perturbations.”
Exchangeable image file format data obtained from the camera, and preprocessing procedures like color normalization and “postprocessing” of images, such as filtering, should also be disclosed. In addition, developers should disclose and justify inclusion of images that have been created by an algorithm within a dataset. Any public images used in the datasets should have references, and privately used images should be made public where possible, the authors said.
The ISIC working group guidelines also provided recommendations for patient-level metadata. Each image should include a patient’s geographical location and medical center they visited as well as their age, sex and gender, ethnicity and/or race, and skin tone. Dr. Daneshjou said this was one area where she and her colleagues found a lack of transparency in AI datasets in algorithms in a recent review. “We found that many AI papers provided sparse details about the images used to train and test their algorithms,” Dr. Daneshjou explained. “For example, only 7 out of 70 papers had any information about the skin tones in the images used for developing and/or testing AI algorithms. Understanding the diversity of images used to train and test algorithms is important because algorithms that are developed on images of predominantly white skin likely won’t work as well on Black and brown skin.”
The guideline authors also asked algorithm developers to describe the limitations of not including patient-level metadata information when it is incomplete or unavailable. In addition, “we ask that algorithm developers comment on potential biases of their algorithms,” Dr. Daneshjou said. “For example, an algorithm based only on telemedicine images may not capture the full range of diseases seen within an in-person clinic.”
When describing their AI algorithm, developers should detail their reasoning for the dataset size and partitions, inclusion and exclusion criteria for images, and use of any external samples for test sets. “Authors should consider any differences between the image characteristics used for algorithm development and those that might be encountered in the real world,” the guidelines stated.
Recommendations for technique
How the images in a dataset are labeled is a unique challenge in developing AI algorithms for dermatology, the authors noted. Developers should use histopathological diagnosis in their labeling, but this can sometimes result in label noise.
“Many of the AI algorithms in dermatology use supervised learning, which requires labeled examples to help the algorithm ‘learn’ features for discriminating between lesions. We found that some papers use consensus labeling – dermatologists providing a label – to label skin cancers; however, the standard for diagnosing skin cancer is using histopathology from a biopsy,” she said. “Dermatologists can biopsy seven to eight suspected melanomas before discovering a true melanoma, so dermatologist labeling of skin cancers is prone to label noise.”
ISIC’s guidelines stated a gold standard of labeling for dermatologic images is one area that still needs future research, but currently, “diagnoses, labels and diagnostic groups used in data repositories as well as public ontologies” such as ICD-11, AnatomyMapper, and SNOMED-CT should be included in dermatologic image datasets.
AI developers should also provide a detailed description of their algorithm, which includes methods, work flows, mathematical formulas as well as the generalizability of the algorithm across more than one dataset.
Recommendations for technical assessment
“Another important recommendation is that algorithm developers should provide a way for algorithms to be publicly evaluable by researchers,” Dr. Daneshjou said. “Many dermatology AI algorithms do not share either their data or their algorithm. Algorithm sharing is important for assessing reproducibility and robustness.”
Google’s recently announced AI-powered dermatology assistant tool, for example, “has made claims about its accuracy and ability to diagnose skin disease at a dermatologist level, but there is no way for researchers to independently test these claims,” she said. Other options like Model Dermatology, developed by Seung Seog Han, MD, PhD, of the Dermatology Clinic in Seoul, South Korea, and colleagues, offer an application programming interface “that allows researchers to test the algorithm,” Dr. Daneshjou said. “This kind of openness is key for assessing algorithm robustness.”
Developers should also note in their algorithm explanations how performance markers and benchmarks would translate to proposed clinical application. “In this context,” the use case – the context in which the AI application is being used – “should be clearly described – who are the intended users and under what clinical scenario are they using the algorithm,” the authors wrote.
Recommendations for application
The guidelines note that use case for the model should also be described by the AI developers. “Our checklist includes delineating use cases for algorithms and describing what use cases may be within the scope of the algorithm versus which use cases are out of scope,” Dr. Daneshjou said. “For example, an algorithm developed to provide decision support to dermatologists, with a human in the loop, may not be accurate enough to release directly to consumers.”
As the goal of AI algorithms in dermatology is eventual implementation for clinicians and patients, the authors asked developers to consider shortcomings and potential harms of the algorithm during implementation. “Ethical considerations and impact on vulnerable populations should also be considered and discussed,” they wrote. An algorithm “suggesting aesthetic medical treatments may have negative effects given the biased nature of beauty standards,” and “an algorithm that diagnoses basal cell carcinomas but lacks any pigmented basal cell carcinomas, which are more often seen in skin of color, will not perform equitably across populations.”
Prior to implementing an AI algorithm, the ISIC working group recommended developers perform prospective clinical trials for validation. Checklists and guidelines like SPIRIT-AI and CONSORT-AI “provide guidance on how to design clinical trials to test AI algorithms,” Dr. Daneshjou said.
After implementation, “I believe we need additional research in how we monitor algorithms after they are deployed clinically, Dr. Daneshjou said. “Currently there are no [Food and Drug Administration]–approved AI algorithms in dermatology; however, there are several applications that have CE mark in Europe, and there are no mechanisms for postmarket surveillance there.
'Timely' recommendations
Commenting on the ISIC working group guidelines, Justin M. Ko, MD, MBA, director and chief of medical dermatology for Stanford Health Care, who was not involved with the work, said that the recommendations are timely and provide “a framework for a ‘common language’ around AI datasets specifically tailored to dermatology.” Dr. Ko, chair of the American Academy of Dermatology’s Ad Hoc Task Force on Augmented Intelligence, noted the work by Dr. Daneshjou and colleagues “is consistent with and builds further details” on the position statement released by the AAD AI task force in 2019.
“As machine-learning capabilities and commercial efforts continue to mature, it becomes increasingly important that we are able to ‘look under the hood,’ and evaluate all the critical factors that influence development of these capabilities,” he said in an interview. “A standard set of reporting guidelines not only allows for transparency in evaluating data and performance of models and algorithms, but also forces the consideration of issues of equity, fairness, mitigation of bias, and clinically meaningful outcomes.”
One concern is the impact of AI algorithms on societal or health systems, he noted, which is brought up in the guidelines. “The last thing we would want is the development of robust AI systems that exacerbate access challenges, or generate patient anxiety/worry, or drive low-value utilization, or adds to care team burden, or create a technological barrier to care, or increases inequity in dermatologic care,” he said.
In developing AI algorithms for dermatology, a “major practical issue” is how performance on paper will translate to real-world use, Dr. Ko explained, and the ISIC guidelines “provide a critical step in empowering clinicians, practices, and our field to shape the advent of the AI and augmented intelligence tools and systems to promote and enhance meaningful clinical outcomes, and augment the core patient-clinician relationship and ensure they are grounded in principles of fairness, equity and transparency.”
This research was funded by awards and grants to individual authors from the Charina Fund, a Google Research Award, Melanoma Research Alliance, National Health and Medical Research Council, National Institutes of Health/National Cancer Institute, National Science Foundation, and the Department of Veterans Affairs. The authors disclosed relationships with governmental entities, pharmaceutical companies, technology startups, medical publishers, charitable trusts, consulting firms, dermatology training companies, providers of medical devices, manufacturers of dermatologic products, and other organizations related to the paper in the form of supplied equipment, having founded a company; receiving grants, patents, or personal fees; holding shares; and medical reporting. Dr. Ko reported that he serves as a clinical advisor for Skin Analytics, and has an ongoing research collaboration with Google.
The
The guidelines, published in JAMA Dermatology on Dec. 1, 2021, contain a broad range of recommendations stakeholders should consider when developing and assessing image-based AI algorithms in dermatology. The recommendations are divided into categories of data, technique, technical assessment, and application. ISIC is “an academia and industry partnership designed to facilitate the application of digital skin imaging to help reduce melanoma mortality,” and is organized into different working groups, including the AI working group, according to its website.
“Our goal with these guidelines was to create higher-quality reporting of dataset and algorithm characteristics for dermatology AI,” first author Roxana Daneshjou, MD, PhD, clinical scholar in dermatology, in the department of dermatology at Stanford (Calif.) University, said in an interview. “We hope these guidelines also aid regulatory bodies around the world when they are assessing algorithms to be used in dermatology.”
Recommendations for data
The authors recommended that datasets used by AI algorithms have image descriptions and details on image artifacts. “For photography, these include the type of camera used; whether images were taken under standardized or varying conditions; whether they were taken by professional photographers, laymen, or health care professionals; and image quality,” they wrote. They also recommended that developers include in an image description the type of lighting used and whether the photo contains pen markings, hair, tattoos, injuries, surgical effects, or other “physical perturbations.”
Exchangeable image file format data obtained from the camera, and preprocessing procedures like color normalization and “postprocessing” of images, such as filtering, should also be disclosed. In addition, developers should disclose and justify inclusion of images that have been created by an algorithm within a dataset. Any public images used in the datasets should have references, and privately used images should be made public where possible, the authors said.
The ISIC working group guidelines also provided recommendations for patient-level metadata. Each image should include a patient’s geographical location and medical center they visited as well as their age, sex and gender, ethnicity and/or race, and skin tone. Dr. Daneshjou said this was one area where she and her colleagues found a lack of transparency in AI datasets in algorithms in a recent review. “We found that many AI papers provided sparse details about the images used to train and test their algorithms,” Dr. Daneshjou explained. “For example, only 7 out of 70 papers had any information about the skin tones in the images used for developing and/or testing AI algorithms. Understanding the diversity of images used to train and test algorithms is important because algorithms that are developed on images of predominantly white skin likely won’t work as well on Black and brown skin.”
The guideline authors also asked algorithm developers to describe the limitations of not including patient-level metadata information when it is incomplete or unavailable. In addition, “we ask that algorithm developers comment on potential biases of their algorithms,” Dr. Daneshjou said. “For example, an algorithm based only on telemedicine images may not capture the full range of diseases seen within an in-person clinic.”
When describing their AI algorithm, developers should detail their reasoning for the dataset size and partitions, inclusion and exclusion criteria for images, and use of any external samples for test sets. “Authors should consider any differences between the image characteristics used for algorithm development and those that might be encountered in the real world,” the guidelines stated.
Recommendations for technique
How the images in a dataset are labeled is a unique challenge in developing AI algorithms for dermatology, the authors noted. Developers should use histopathological diagnosis in their labeling, but this can sometimes result in label noise.
“Many of the AI algorithms in dermatology use supervised learning, which requires labeled examples to help the algorithm ‘learn’ features for discriminating between lesions. We found that some papers use consensus labeling – dermatologists providing a label – to label skin cancers; however, the standard for diagnosing skin cancer is using histopathology from a biopsy,” she said. “Dermatologists can biopsy seven to eight suspected melanomas before discovering a true melanoma, so dermatologist labeling of skin cancers is prone to label noise.”
ISIC’s guidelines stated a gold standard of labeling for dermatologic images is one area that still needs future research, but currently, “diagnoses, labels and diagnostic groups used in data repositories as well as public ontologies” such as ICD-11, AnatomyMapper, and SNOMED-CT should be included in dermatologic image datasets.
AI developers should also provide a detailed description of their algorithm, which includes methods, work flows, mathematical formulas as well as the generalizability of the algorithm across more than one dataset.
Recommendations for technical assessment
“Another important recommendation is that algorithm developers should provide a way for algorithms to be publicly evaluable by researchers,” Dr. Daneshjou said. “Many dermatology AI algorithms do not share either their data or their algorithm. Algorithm sharing is important for assessing reproducibility and robustness.”
Google’s recently announced AI-powered dermatology assistant tool, for example, “has made claims about its accuracy and ability to diagnose skin disease at a dermatologist level, but there is no way for researchers to independently test these claims,” she said. Other options like Model Dermatology, developed by Seung Seog Han, MD, PhD, of the Dermatology Clinic in Seoul, South Korea, and colleagues, offer an application programming interface “that allows researchers to test the algorithm,” Dr. Daneshjou said. “This kind of openness is key for assessing algorithm robustness.”
Developers should also note in their algorithm explanations how performance markers and benchmarks would translate to proposed clinical application. “In this context,” the use case – the context in which the AI application is being used – “should be clearly described – who are the intended users and under what clinical scenario are they using the algorithm,” the authors wrote.
Recommendations for application
The guidelines note that use case for the model should also be described by the AI developers. “Our checklist includes delineating use cases for algorithms and describing what use cases may be within the scope of the algorithm versus which use cases are out of scope,” Dr. Daneshjou said. “For example, an algorithm developed to provide decision support to dermatologists, with a human in the loop, may not be accurate enough to release directly to consumers.”
As the goal of AI algorithms in dermatology is eventual implementation for clinicians and patients, the authors asked developers to consider shortcomings and potential harms of the algorithm during implementation. “Ethical considerations and impact on vulnerable populations should also be considered and discussed,” they wrote. An algorithm “suggesting aesthetic medical treatments may have negative effects given the biased nature of beauty standards,” and “an algorithm that diagnoses basal cell carcinomas but lacks any pigmented basal cell carcinomas, which are more often seen in skin of color, will not perform equitably across populations.”
Prior to implementing an AI algorithm, the ISIC working group recommended developers perform prospective clinical trials for validation. Checklists and guidelines like SPIRIT-AI and CONSORT-AI “provide guidance on how to design clinical trials to test AI algorithms,” Dr. Daneshjou said.
After implementation, “I believe we need additional research in how we monitor algorithms after they are deployed clinically, Dr. Daneshjou said. “Currently there are no [Food and Drug Administration]–approved AI algorithms in dermatology; however, there are several applications that have CE mark in Europe, and there are no mechanisms for postmarket surveillance there.
'Timely' recommendations
Commenting on the ISIC working group guidelines, Justin M. Ko, MD, MBA, director and chief of medical dermatology for Stanford Health Care, who was not involved with the work, said that the recommendations are timely and provide “a framework for a ‘common language’ around AI datasets specifically tailored to dermatology.” Dr. Ko, chair of the American Academy of Dermatology’s Ad Hoc Task Force on Augmented Intelligence, noted the work by Dr. Daneshjou and colleagues “is consistent with and builds further details” on the position statement released by the AAD AI task force in 2019.
“As machine-learning capabilities and commercial efforts continue to mature, it becomes increasingly important that we are able to ‘look under the hood,’ and evaluate all the critical factors that influence development of these capabilities,” he said in an interview. “A standard set of reporting guidelines not only allows for transparency in evaluating data and performance of models and algorithms, but also forces the consideration of issues of equity, fairness, mitigation of bias, and clinically meaningful outcomes.”
One concern is the impact of AI algorithms on societal or health systems, he noted, which is brought up in the guidelines. “The last thing we would want is the development of robust AI systems that exacerbate access challenges, or generate patient anxiety/worry, or drive low-value utilization, or adds to care team burden, or create a technological barrier to care, or increases inequity in dermatologic care,” he said.
In developing AI algorithms for dermatology, a “major practical issue” is how performance on paper will translate to real-world use, Dr. Ko explained, and the ISIC guidelines “provide a critical step in empowering clinicians, practices, and our field to shape the advent of the AI and augmented intelligence tools and systems to promote and enhance meaningful clinical outcomes, and augment the core patient-clinician relationship and ensure they are grounded in principles of fairness, equity and transparency.”
This research was funded by awards and grants to individual authors from the Charina Fund, a Google Research Award, Melanoma Research Alliance, National Health and Medical Research Council, National Institutes of Health/National Cancer Institute, National Science Foundation, and the Department of Veterans Affairs. The authors disclosed relationships with governmental entities, pharmaceutical companies, technology startups, medical publishers, charitable trusts, consulting firms, dermatology training companies, providers of medical devices, manufacturers of dermatologic products, and other organizations related to the paper in the form of supplied equipment, having founded a company; receiving grants, patents, or personal fees; holding shares; and medical reporting. Dr. Ko reported that he serves as a clinical advisor for Skin Analytics, and has an ongoing research collaboration with Google.
The
The guidelines, published in JAMA Dermatology on Dec. 1, 2021, contain a broad range of recommendations stakeholders should consider when developing and assessing image-based AI algorithms in dermatology. The recommendations are divided into categories of data, technique, technical assessment, and application. ISIC is “an academia and industry partnership designed to facilitate the application of digital skin imaging to help reduce melanoma mortality,” and is organized into different working groups, including the AI working group, according to its website.
“Our goal with these guidelines was to create higher-quality reporting of dataset and algorithm characteristics for dermatology AI,” first author Roxana Daneshjou, MD, PhD, clinical scholar in dermatology, in the department of dermatology at Stanford (Calif.) University, said in an interview. “We hope these guidelines also aid regulatory bodies around the world when they are assessing algorithms to be used in dermatology.”
Recommendations for data
The authors recommended that datasets used by AI algorithms have image descriptions and details on image artifacts. “For photography, these include the type of camera used; whether images were taken under standardized or varying conditions; whether they were taken by professional photographers, laymen, or health care professionals; and image quality,” they wrote. They also recommended that developers include in an image description the type of lighting used and whether the photo contains pen markings, hair, tattoos, injuries, surgical effects, or other “physical perturbations.”
Exchangeable image file format data obtained from the camera, and preprocessing procedures like color normalization and “postprocessing” of images, such as filtering, should also be disclosed. In addition, developers should disclose and justify inclusion of images that have been created by an algorithm within a dataset. Any public images used in the datasets should have references, and privately used images should be made public where possible, the authors said.
The ISIC working group guidelines also provided recommendations for patient-level metadata. Each image should include a patient’s geographical location and medical center they visited as well as their age, sex and gender, ethnicity and/or race, and skin tone. Dr. Daneshjou said this was one area where she and her colleagues found a lack of transparency in AI datasets in algorithms in a recent review. “We found that many AI papers provided sparse details about the images used to train and test their algorithms,” Dr. Daneshjou explained. “For example, only 7 out of 70 papers had any information about the skin tones in the images used for developing and/or testing AI algorithms. Understanding the diversity of images used to train and test algorithms is important because algorithms that are developed on images of predominantly white skin likely won’t work as well on Black and brown skin.”
The guideline authors also asked algorithm developers to describe the limitations of not including patient-level metadata information when it is incomplete or unavailable. In addition, “we ask that algorithm developers comment on potential biases of their algorithms,” Dr. Daneshjou said. “For example, an algorithm based only on telemedicine images may not capture the full range of diseases seen within an in-person clinic.”
When describing their AI algorithm, developers should detail their reasoning for the dataset size and partitions, inclusion and exclusion criteria for images, and use of any external samples for test sets. “Authors should consider any differences between the image characteristics used for algorithm development and those that might be encountered in the real world,” the guidelines stated.
Recommendations for technique
How the images in a dataset are labeled is a unique challenge in developing AI algorithms for dermatology, the authors noted. Developers should use histopathological diagnosis in their labeling, but this can sometimes result in label noise.
“Many of the AI algorithms in dermatology use supervised learning, which requires labeled examples to help the algorithm ‘learn’ features for discriminating between lesions. We found that some papers use consensus labeling – dermatologists providing a label – to label skin cancers; however, the standard for diagnosing skin cancer is using histopathology from a biopsy,” she said. “Dermatologists can biopsy seven to eight suspected melanomas before discovering a true melanoma, so dermatologist labeling of skin cancers is prone to label noise.”
ISIC’s guidelines stated a gold standard of labeling for dermatologic images is one area that still needs future research, but currently, “diagnoses, labels and diagnostic groups used in data repositories as well as public ontologies” such as ICD-11, AnatomyMapper, and SNOMED-CT should be included in dermatologic image datasets.
AI developers should also provide a detailed description of their algorithm, which includes methods, work flows, mathematical formulas as well as the generalizability of the algorithm across more than one dataset.
Recommendations for technical assessment
“Another important recommendation is that algorithm developers should provide a way for algorithms to be publicly evaluable by researchers,” Dr. Daneshjou said. “Many dermatology AI algorithms do not share either their data or their algorithm. Algorithm sharing is important for assessing reproducibility and robustness.”
Google’s recently announced AI-powered dermatology assistant tool, for example, “has made claims about its accuracy and ability to diagnose skin disease at a dermatologist level, but there is no way for researchers to independently test these claims,” she said. Other options like Model Dermatology, developed by Seung Seog Han, MD, PhD, of the Dermatology Clinic in Seoul, South Korea, and colleagues, offer an application programming interface “that allows researchers to test the algorithm,” Dr. Daneshjou said. “This kind of openness is key for assessing algorithm robustness.”
Developers should also note in their algorithm explanations how performance markers and benchmarks would translate to proposed clinical application. “In this context,” the use case – the context in which the AI application is being used – “should be clearly described – who are the intended users and under what clinical scenario are they using the algorithm,” the authors wrote.
Recommendations for application
The guidelines note that use case for the model should also be described by the AI developers. “Our checklist includes delineating use cases for algorithms and describing what use cases may be within the scope of the algorithm versus which use cases are out of scope,” Dr. Daneshjou said. “For example, an algorithm developed to provide decision support to dermatologists, with a human in the loop, may not be accurate enough to release directly to consumers.”
As the goal of AI algorithms in dermatology is eventual implementation for clinicians and patients, the authors asked developers to consider shortcomings and potential harms of the algorithm during implementation. “Ethical considerations and impact on vulnerable populations should also be considered and discussed,” they wrote. An algorithm “suggesting aesthetic medical treatments may have negative effects given the biased nature of beauty standards,” and “an algorithm that diagnoses basal cell carcinomas but lacks any pigmented basal cell carcinomas, which are more often seen in skin of color, will not perform equitably across populations.”
Prior to implementing an AI algorithm, the ISIC working group recommended developers perform prospective clinical trials for validation. Checklists and guidelines like SPIRIT-AI and CONSORT-AI “provide guidance on how to design clinical trials to test AI algorithms,” Dr. Daneshjou said.
After implementation, “I believe we need additional research in how we monitor algorithms after they are deployed clinically, Dr. Daneshjou said. “Currently there are no [Food and Drug Administration]–approved AI algorithms in dermatology; however, there are several applications that have CE mark in Europe, and there are no mechanisms for postmarket surveillance there.
'Timely' recommendations
Commenting on the ISIC working group guidelines, Justin M. Ko, MD, MBA, director and chief of medical dermatology for Stanford Health Care, who was not involved with the work, said that the recommendations are timely and provide “a framework for a ‘common language’ around AI datasets specifically tailored to dermatology.” Dr. Ko, chair of the American Academy of Dermatology’s Ad Hoc Task Force on Augmented Intelligence, noted the work by Dr. Daneshjou and colleagues “is consistent with and builds further details” on the position statement released by the AAD AI task force in 2019.
“As machine-learning capabilities and commercial efforts continue to mature, it becomes increasingly important that we are able to ‘look under the hood,’ and evaluate all the critical factors that influence development of these capabilities,” he said in an interview. “A standard set of reporting guidelines not only allows for transparency in evaluating data and performance of models and algorithms, but also forces the consideration of issues of equity, fairness, mitigation of bias, and clinically meaningful outcomes.”
One concern is the impact of AI algorithms on societal or health systems, he noted, which is brought up in the guidelines. “The last thing we would want is the development of robust AI systems that exacerbate access challenges, or generate patient anxiety/worry, or drive low-value utilization, or adds to care team burden, or create a technological barrier to care, or increases inequity in dermatologic care,” he said.
In developing AI algorithms for dermatology, a “major practical issue” is how performance on paper will translate to real-world use, Dr. Ko explained, and the ISIC guidelines “provide a critical step in empowering clinicians, practices, and our field to shape the advent of the AI and augmented intelligence tools and systems to promote and enhance meaningful clinical outcomes, and augment the core patient-clinician relationship and ensure they are grounded in principles of fairness, equity and transparency.”
This research was funded by awards and grants to individual authors from the Charina Fund, a Google Research Award, Melanoma Research Alliance, National Health and Medical Research Council, National Institutes of Health/National Cancer Institute, National Science Foundation, and the Department of Veterans Affairs. The authors disclosed relationships with governmental entities, pharmaceutical companies, technology startups, medical publishers, charitable trusts, consulting firms, dermatology training companies, providers of medical devices, manufacturers of dermatologic products, and other organizations related to the paper in the form of supplied equipment, having founded a company; receiving grants, patents, or personal fees; holding shares; and medical reporting. Dr. Ko reported that he serves as a clinical advisor for Skin Analytics, and has an ongoing research collaboration with Google.
FROM JAMA DERMATOLOGY
Bamlanivimab’s effects in COVID-19 depend on antibodies
In the randomized controlled trial, in both the group who received bamlanivimab and the group who received placebo, higher antigen and viral RNA levels were associated with a lower proportion of patients achieving recovery.
Other studies have shown that the use of monoclonal antibodies reduces hospitalization risk in outpatients with early COVID-19, and appears to promote viral load decline in the nasopharynx, wrote Jens D. Lundgren, MD, of the University of Copenhagen and colleagues in their article published in the Annals of Internal Medicine. What had been missing prior to this new research was final results from hospitalized patients, the authors said.
In the new study, the researchers randomized 314 adults hospitalized with COVID-19 but without end-organ failure to receive 7,000 mg bamlanivimab (163 patients) or a placebo (151 patients). All patients received study-supplied remdesivir unless contraindicated. The researchers compared the efficacy of bamlanivimab versus placebo, but considered remdesivir the standard of care in this study.
At baseline, 50% of patients overall had antispike endogenous neutralizing antibodies (nAbs), and 50% had SARS-CoV-2 nucleocapsid plasma antigen levels of at least 1,000 ng/L.
The median time to sustained recovery, 19 days, was not significantly different between the bamlanivimab and placebo groups (subhazard ratio, 0.99).
“As hypothesized, among those who were negative for nAb, the difference between bamlanivimab and placebo was more evident if levels of plasma antigen or nasal-swab viral RNA were above the median entry levels,” with subhazard ratios of 1.48 and 1.89, respectively, the researchers explained.
However, the hazard ratio for death for bamlanivimab vs. placebo was 0.45 for patients negative for nAb vs. 3.53 for those positive for nAb. These differences with respect to nAb status were similar across all 90 elements of a composite safety outcome, the researchers said.
Potential benefits remain unclear
The use of neutralizing monoclonal antibodies has been extensively documented as an effective treatment for COVID-19 among ambulatory patients, corresponding author Dr. Lundgren said in an interview.
“Conversely, among admitted patients with COVID-19 pneumonia, the benefit has been questionable,” he said.
The researchers examined a hypothesis that the null finding in hospitalized patients may stem from differences in underlying mechanisms, “either from uncontrolled viral replication – which would be predicted to occur in particular among those not yet been able to mount an endogenous immune response – or from hyperinflammation among those that have mounted such a response,” Dr. Lundgren said.
The study findings supported the stated hypothesis, said Dr. Lundgren. “However, it was surprising that not only was the neutralizing antibody without any benefit among those that had mounted an endogenous immune response, but it actually may have been harmful,” he said.
Bamlanivimab was effective against the viral strain that circulated at the time of enrollment in the study, but subsequent viral strains have appeared to be unaffected by the neutralizing activity of the antibody, said Dr. Lundgren.
From a practical standpoint, “the findings would suggest that use of neutralizing monoclonal antibodies for patients admitted to a hospital with COVID pneumonia should be restricted to those that have not yet mounted an endogenous immune response, as determined by lack of detectable neutralizing antibodies at the time of admission,” Dr. Lundgren said.
Looking ahead, studies are currently underway to examine how the findings translate to vaccinated patients, he added. Other questions to be addressed include whether the benefits and harms apply to some or all neutralizing antibody products, he said.
In addition, “our research consortium is currently doing field testing of several point-of-care test candidates to examine their reliability and functionality,” for how quickly they might identify an endogenous neutralizing antibody response in an admitted COVID pneumonia patient,” Dr. Lundgren noted.
Findings show bamlanivimab’s limits
“Based on the findings of the current study, no clear subgroup of patients could be identified who would benefit from bamlanivimab when hospitalized with COVID-19,” said Suman Pal, MD, of the University of New Mexico, Albuquerque, in an interview.
“The study findings also show possible harm of using bamlanivimab in hospitalized COVID-19 patients who were seropositive for neutralizing antibodies prior to receiving therapy,” Dr. Pal emphasized. “Moreover, the study did not include participants with COVID-19 from variant strains, such as delta and omicron, which currently account for a large number of cases.” “Therefore, the results of this study do not support the use of bamlanivimab in the clinical setting until further evidence is available to guide the selection of patients who may benefit from therapy,” he explained.
“The possible benefit of bamlanivimab does not outweigh the risks in patients hospitalized with COVID-19,” he concluded.
Dr. Pal emphasized the need for larger prospective studies to establish whether bamlanivimab may have benefits in a subgroup of patients, but “well-validated point-of-care tests to identify such patients need to be readily available before this therapy can be considered by clinicians at the bedside,” he concluded.
Diligent screening required before use
Monoclonal antibody treatment has been administered to individuals with diagnosis of COVID-19 infection as outpatients as well as for hospitalized inpatients, said Noel Deep, MD, an internist in Antigo, Wisc., in an interview. “This study is important because it helps physicians and health care institutions to evaluate whether continued use of the monoclonal antibodies would be beneficial and, if so, in what patient populations,” he said.
The findings present interesting implications for the care of COVID-19 patients, said Dr. Deep. “This study indicates that bamlanivimab does not provide the benefit that was initially envisioned when the monoclonal antibody infusions were initially initiated in the treatment of COVID-19 infections. “Serological screening of the patients would help to identify that subgroup of individuals who could benefit from this monoclonal antibody rather than administering it to every COVID-19–positive individual,” he explained.
However, “it is important to note that the emergency use authorization (EUA) for single-agent bamlanivimab has been revoked,” Dr. Deep said.
“The potential benefits of bamlanivimab can be realized only if adequate attention is paid to identifying the appropriate candidates based on serological screening, and administering bamlanivimab to those who are already producing endogenous antibodies could lead to increased risk to those individuals,” he said. Dr. Deep added that he would favor administration of bamlanivimab “in those appropriately screened and eligible candidates, and it is my opinion that the benefits outweigh the risks in those individuals.”
Although the EUA for single-agent bamlanivimab has been revoked, “alternative monoclonal antibody therapies remain available under EUA, including REGEN-COV (casirivimab and imdevimab, administered together), and bamlanivimab and etesevimab administered together, for the same uses as previously authorized for bamlanivimab alone,” Dr. Deep said. “The FDA believes that these alternative monoclonal antibody therapies remain appropriate to treat patients with COVID-19, and I would like to see some data about the benefits and risks of these agents,” he noted.
Limitations, funding, and disclosures
The main limitation of the study was the small size and the fact that it was a subgroup analysis of a trial that ended early because of futility, the researchers wrote. However, the Therapeutics for Inpatients With COVID-19 (TICO) platform will proceed with clinical evaluation of additional COVID-19 treatments, they said.
The study was supported primarily by the U.S. government Operation Warp Speed and the National Institute of Allergy and Infectious Diseases. Other funding sources included the Division of Clinical Research and Leidos Biomedical Research for the INSIGHT (International Network for Strategic Initiatives in Global HIV Trials) Network, as well as an agreement between the National Heart, Lung, and Blood Institute and the Research Triangle Institute for the PETAL (Prevention & Early Treatment of Acute Lung Injury) Network and CTSN (Cardiothoracic Surgical Trials Network). Other support came from the U.S. Department of Veterans Affairs and the governments of Denmark (National Research Foundation), Australia (National Health and Medical Research Council), and the United Kingdom (Medical Research Council).
The medications used in the study were donated by Gilead Sciences and Eli Lilly.
The researchers had no financial conflicts do disclose. Dr. Deep and Dr. Pal had no relevant financial conflicts to disclose.
In the randomized controlled trial, in both the group who received bamlanivimab and the group who received placebo, higher antigen and viral RNA levels were associated with a lower proportion of patients achieving recovery.
Other studies have shown that the use of monoclonal antibodies reduces hospitalization risk in outpatients with early COVID-19, and appears to promote viral load decline in the nasopharynx, wrote Jens D. Lundgren, MD, of the University of Copenhagen and colleagues in their article published in the Annals of Internal Medicine. What had been missing prior to this new research was final results from hospitalized patients, the authors said.
In the new study, the researchers randomized 314 adults hospitalized with COVID-19 but without end-organ failure to receive 7,000 mg bamlanivimab (163 patients) or a placebo (151 patients). All patients received study-supplied remdesivir unless contraindicated. The researchers compared the efficacy of bamlanivimab versus placebo, but considered remdesivir the standard of care in this study.
At baseline, 50% of patients overall had antispike endogenous neutralizing antibodies (nAbs), and 50% had SARS-CoV-2 nucleocapsid plasma antigen levels of at least 1,000 ng/L.
The median time to sustained recovery, 19 days, was not significantly different between the bamlanivimab and placebo groups (subhazard ratio, 0.99).
“As hypothesized, among those who were negative for nAb, the difference between bamlanivimab and placebo was more evident if levels of plasma antigen or nasal-swab viral RNA were above the median entry levels,” with subhazard ratios of 1.48 and 1.89, respectively, the researchers explained.
However, the hazard ratio for death for bamlanivimab vs. placebo was 0.45 for patients negative for nAb vs. 3.53 for those positive for nAb. These differences with respect to nAb status were similar across all 90 elements of a composite safety outcome, the researchers said.
Potential benefits remain unclear
The use of neutralizing monoclonal antibodies has been extensively documented as an effective treatment for COVID-19 among ambulatory patients, corresponding author Dr. Lundgren said in an interview.
“Conversely, among admitted patients with COVID-19 pneumonia, the benefit has been questionable,” he said.
The researchers examined a hypothesis that the null finding in hospitalized patients may stem from differences in underlying mechanisms, “either from uncontrolled viral replication – which would be predicted to occur in particular among those not yet been able to mount an endogenous immune response – or from hyperinflammation among those that have mounted such a response,” Dr. Lundgren said.
The study findings supported the stated hypothesis, said Dr. Lundgren. “However, it was surprising that not only was the neutralizing antibody without any benefit among those that had mounted an endogenous immune response, but it actually may have been harmful,” he said.
Bamlanivimab was effective against the viral strain that circulated at the time of enrollment in the study, but subsequent viral strains have appeared to be unaffected by the neutralizing activity of the antibody, said Dr. Lundgren.
From a practical standpoint, “the findings would suggest that use of neutralizing monoclonal antibodies for patients admitted to a hospital with COVID pneumonia should be restricted to those that have not yet mounted an endogenous immune response, as determined by lack of detectable neutralizing antibodies at the time of admission,” Dr. Lundgren said.
Looking ahead, studies are currently underway to examine how the findings translate to vaccinated patients, he added. Other questions to be addressed include whether the benefits and harms apply to some or all neutralizing antibody products, he said.
In addition, “our research consortium is currently doing field testing of several point-of-care test candidates to examine their reliability and functionality,” for how quickly they might identify an endogenous neutralizing antibody response in an admitted COVID pneumonia patient,” Dr. Lundgren noted.
Findings show bamlanivimab’s limits
“Based on the findings of the current study, no clear subgroup of patients could be identified who would benefit from bamlanivimab when hospitalized with COVID-19,” said Suman Pal, MD, of the University of New Mexico, Albuquerque, in an interview.
“The study findings also show possible harm of using bamlanivimab in hospitalized COVID-19 patients who were seropositive for neutralizing antibodies prior to receiving therapy,” Dr. Pal emphasized. “Moreover, the study did not include participants with COVID-19 from variant strains, such as delta and omicron, which currently account for a large number of cases.” “Therefore, the results of this study do not support the use of bamlanivimab in the clinical setting until further evidence is available to guide the selection of patients who may benefit from therapy,” he explained.
“The possible benefit of bamlanivimab does not outweigh the risks in patients hospitalized with COVID-19,” he concluded.
Dr. Pal emphasized the need for larger prospective studies to establish whether bamlanivimab may have benefits in a subgroup of patients, but “well-validated point-of-care tests to identify such patients need to be readily available before this therapy can be considered by clinicians at the bedside,” he concluded.
Diligent screening required before use
Monoclonal antibody treatment has been administered to individuals with diagnosis of COVID-19 infection as outpatients as well as for hospitalized inpatients, said Noel Deep, MD, an internist in Antigo, Wisc., in an interview. “This study is important because it helps physicians and health care institutions to evaluate whether continued use of the monoclonal antibodies would be beneficial and, if so, in what patient populations,” he said.
The findings present interesting implications for the care of COVID-19 patients, said Dr. Deep. “This study indicates that bamlanivimab does not provide the benefit that was initially envisioned when the monoclonal antibody infusions were initially initiated in the treatment of COVID-19 infections. “Serological screening of the patients would help to identify that subgroup of individuals who could benefit from this monoclonal antibody rather than administering it to every COVID-19–positive individual,” he explained.
However, “it is important to note that the emergency use authorization (EUA) for single-agent bamlanivimab has been revoked,” Dr. Deep said.
“The potential benefits of bamlanivimab can be realized only if adequate attention is paid to identifying the appropriate candidates based on serological screening, and administering bamlanivimab to those who are already producing endogenous antibodies could lead to increased risk to those individuals,” he said. Dr. Deep added that he would favor administration of bamlanivimab “in those appropriately screened and eligible candidates, and it is my opinion that the benefits outweigh the risks in those individuals.”
Although the EUA for single-agent bamlanivimab has been revoked, “alternative monoclonal antibody therapies remain available under EUA, including REGEN-COV (casirivimab and imdevimab, administered together), and bamlanivimab and etesevimab administered together, for the same uses as previously authorized for bamlanivimab alone,” Dr. Deep said. “The FDA believes that these alternative monoclonal antibody therapies remain appropriate to treat patients with COVID-19, and I would like to see some data about the benefits and risks of these agents,” he noted.
Limitations, funding, and disclosures
The main limitation of the study was the small size and the fact that it was a subgroup analysis of a trial that ended early because of futility, the researchers wrote. However, the Therapeutics for Inpatients With COVID-19 (TICO) platform will proceed with clinical evaluation of additional COVID-19 treatments, they said.
The study was supported primarily by the U.S. government Operation Warp Speed and the National Institute of Allergy and Infectious Diseases. Other funding sources included the Division of Clinical Research and Leidos Biomedical Research for the INSIGHT (International Network for Strategic Initiatives in Global HIV Trials) Network, as well as an agreement between the National Heart, Lung, and Blood Institute and the Research Triangle Institute for the PETAL (Prevention & Early Treatment of Acute Lung Injury) Network and CTSN (Cardiothoracic Surgical Trials Network). Other support came from the U.S. Department of Veterans Affairs and the governments of Denmark (National Research Foundation), Australia (National Health and Medical Research Council), and the United Kingdom (Medical Research Council).
The medications used in the study were donated by Gilead Sciences and Eli Lilly.
The researchers had no financial conflicts do disclose. Dr. Deep and Dr. Pal had no relevant financial conflicts to disclose.
In the randomized controlled trial, in both the group who received bamlanivimab and the group who received placebo, higher antigen and viral RNA levels were associated with a lower proportion of patients achieving recovery.
Other studies have shown that the use of monoclonal antibodies reduces hospitalization risk in outpatients with early COVID-19, and appears to promote viral load decline in the nasopharynx, wrote Jens D. Lundgren, MD, of the University of Copenhagen and colleagues in their article published in the Annals of Internal Medicine. What had been missing prior to this new research was final results from hospitalized patients, the authors said.
In the new study, the researchers randomized 314 adults hospitalized with COVID-19 but without end-organ failure to receive 7,000 mg bamlanivimab (163 patients) or a placebo (151 patients). All patients received study-supplied remdesivir unless contraindicated. The researchers compared the efficacy of bamlanivimab versus placebo, but considered remdesivir the standard of care in this study.
At baseline, 50% of patients overall had antispike endogenous neutralizing antibodies (nAbs), and 50% had SARS-CoV-2 nucleocapsid plasma antigen levels of at least 1,000 ng/L.
The median time to sustained recovery, 19 days, was not significantly different between the bamlanivimab and placebo groups (subhazard ratio, 0.99).
“As hypothesized, among those who were negative for nAb, the difference between bamlanivimab and placebo was more evident if levels of plasma antigen or nasal-swab viral RNA were above the median entry levels,” with subhazard ratios of 1.48 and 1.89, respectively, the researchers explained.
However, the hazard ratio for death for bamlanivimab vs. placebo was 0.45 for patients negative for nAb vs. 3.53 for those positive for nAb. These differences with respect to nAb status were similar across all 90 elements of a composite safety outcome, the researchers said.
Potential benefits remain unclear
The use of neutralizing monoclonal antibodies has been extensively documented as an effective treatment for COVID-19 among ambulatory patients, corresponding author Dr. Lundgren said in an interview.
“Conversely, among admitted patients with COVID-19 pneumonia, the benefit has been questionable,” he said.
The researchers examined a hypothesis that the null finding in hospitalized patients may stem from differences in underlying mechanisms, “either from uncontrolled viral replication – which would be predicted to occur in particular among those not yet been able to mount an endogenous immune response – or from hyperinflammation among those that have mounted such a response,” Dr. Lundgren said.
The study findings supported the stated hypothesis, said Dr. Lundgren. “However, it was surprising that not only was the neutralizing antibody without any benefit among those that had mounted an endogenous immune response, but it actually may have been harmful,” he said.
Bamlanivimab was effective against the viral strain that circulated at the time of enrollment in the study, but subsequent viral strains have appeared to be unaffected by the neutralizing activity of the antibody, said Dr. Lundgren.
From a practical standpoint, “the findings would suggest that use of neutralizing monoclonal antibodies for patients admitted to a hospital with COVID pneumonia should be restricted to those that have not yet mounted an endogenous immune response, as determined by lack of detectable neutralizing antibodies at the time of admission,” Dr. Lundgren said.
Looking ahead, studies are currently underway to examine how the findings translate to vaccinated patients, he added. Other questions to be addressed include whether the benefits and harms apply to some or all neutralizing antibody products, he said.
In addition, “our research consortium is currently doing field testing of several point-of-care test candidates to examine their reliability and functionality,” for how quickly they might identify an endogenous neutralizing antibody response in an admitted COVID pneumonia patient,” Dr. Lundgren noted.
Findings show bamlanivimab’s limits
“Based on the findings of the current study, no clear subgroup of patients could be identified who would benefit from bamlanivimab when hospitalized with COVID-19,” said Suman Pal, MD, of the University of New Mexico, Albuquerque, in an interview.
“The study findings also show possible harm of using bamlanivimab in hospitalized COVID-19 patients who were seropositive for neutralizing antibodies prior to receiving therapy,” Dr. Pal emphasized. “Moreover, the study did not include participants with COVID-19 from variant strains, such as delta and omicron, which currently account for a large number of cases.” “Therefore, the results of this study do not support the use of bamlanivimab in the clinical setting until further evidence is available to guide the selection of patients who may benefit from therapy,” he explained.
“The possible benefit of bamlanivimab does not outweigh the risks in patients hospitalized with COVID-19,” he concluded.
Dr. Pal emphasized the need for larger prospective studies to establish whether bamlanivimab may have benefits in a subgroup of patients, but “well-validated point-of-care tests to identify such patients need to be readily available before this therapy can be considered by clinicians at the bedside,” he concluded.
Diligent screening required before use
Monoclonal antibody treatment has been administered to individuals with diagnosis of COVID-19 infection as outpatients as well as for hospitalized inpatients, said Noel Deep, MD, an internist in Antigo, Wisc., in an interview. “This study is important because it helps physicians and health care institutions to evaluate whether continued use of the monoclonal antibodies would be beneficial and, if so, in what patient populations,” he said.
The findings present interesting implications for the care of COVID-19 patients, said Dr. Deep. “This study indicates that bamlanivimab does not provide the benefit that was initially envisioned when the monoclonal antibody infusions were initially initiated in the treatment of COVID-19 infections. “Serological screening of the patients would help to identify that subgroup of individuals who could benefit from this monoclonal antibody rather than administering it to every COVID-19–positive individual,” he explained.
However, “it is important to note that the emergency use authorization (EUA) for single-agent bamlanivimab has been revoked,” Dr. Deep said.
“The potential benefits of bamlanivimab can be realized only if adequate attention is paid to identifying the appropriate candidates based on serological screening, and administering bamlanivimab to those who are already producing endogenous antibodies could lead to increased risk to those individuals,” he said. Dr. Deep added that he would favor administration of bamlanivimab “in those appropriately screened and eligible candidates, and it is my opinion that the benefits outweigh the risks in those individuals.”
Although the EUA for single-agent bamlanivimab has been revoked, “alternative monoclonal antibody therapies remain available under EUA, including REGEN-COV (casirivimab and imdevimab, administered together), and bamlanivimab and etesevimab administered together, for the same uses as previously authorized for bamlanivimab alone,” Dr. Deep said. “The FDA believes that these alternative monoclonal antibody therapies remain appropriate to treat patients with COVID-19, and I would like to see some data about the benefits and risks of these agents,” he noted.
Limitations, funding, and disclosures
The main limitation of the study was the small size and the fact that it was a subgroup analysis of a trial that ended early because of futility, the researchers wrote. However, the Therapeutics for Inpatients With COVID-19 (TICO) platform will proceed with clinical evaluation of additional COVID-19 treatments, they said.
The study was supported primarily by the U.S. government Operation Warp Speed and the National Institute of Allergy and Infectious Diseases. Other funding sources included the Division of Clinical Research and Leidos Biomedical Research for the INSIGHT (International Network for Strategic Initiatives in Global HIV Trials) Network, as well as an agreement between the National Heart, Lung, and Blood Institute and the Research Triangle Institute for the PETAL (Prevention & Early Treatment of Acute Lung Injury) Network and CTSN (Cardiothoracic Surgical Trials Network). Other support came from the U.S. Department of Veterans Affairs and the governments of Denmark (National Research Foundation), Australia (National Health and Medical Research Council), and the United Kingdom (Medical Research Council).
The medications used in the study were donated by Gilead Sciences and Eli Lilly.
The researchers had no financial conflicts do disclose. Dr. Deep and Dr. Pal had no relevant financial conflicts to disclose.
FROM ANNALS OF INTERNAL MEDICINE
Is it safe to pair low-power fractional diode lasers with cosmetic injectables in a single session?
, results from a 6-year, single-center review showed.
“These treatments can be complementary in single-session treatments and can offer increased convenience for both patients and physicians,” primary study author Jordan V. Wang, MD, MBE, MBA, said during a virtual abstract session at the annual meeting of the American Society for Dermatologic Surgery.
To date, limited studies have demonstrated the safety of pairing botulinum neurotoxin type A and soft-tissue fillers with laser and other energy-based devices during the same treatment session on the same day, said Dr. Wang, medical research director at the Laser & Skin Surgery Center of New York. “Some concerns remain, though, regarding patient safety and efficacy,” he said. “Data on single-session treatment with low-power, low-density 1,927-nm and 1,440-nm fractional diode lasers and either botulinum neurotoxin or fillers are lacking.”
In a retrospective review of electronic medical records conducted from May 2015 to April 2021, Dr. Wang, Roy G. Geronemus, MD, and Carolyn Kushner, MD evaluated patients who received a single-session facial treatment with either BoNT-A or soft-tissue fillers and the low-power, low-density 1,927-nm and 1,440-nm fractional diode lasers (Clear+Brilliant Perméa and Original, Solta, Pleasanton, Calif.). Safety was assessed by documenting adverse events related to the spread of BoNT-A and fillers or laser treatment of filled areas within 4 weeks.
Adverse events they looked for related to botulinum neurotoxin use included eyelid ptosis; neck weakness or spasms; impairments in chewing, swallowing, speech, and respiration; and prescriptions of apraclonidine eye drops. Filler-related adverse events they looked for included product migration, unexpected loss of filler volume, vascular occlusion, acute pain, necrosis, blindness, and burn. “For both, we looked at hospital or emergency room transfers or admissions and referrals to ENT or ophthalmology,” Dr. Wang said.
During the 6-year study period, 525 patients had 1,562 single-session laser treatments with a mean 46.4 units of BoNT-A, and 398 patients had 1,237 single-session treatments with a mean 1.6 soft-tissue filler syringes. Among those who received BoNT-A, most (93%) were female, their mean age was 51 years, and 99% were treated with a 1,927-nm wavelength at a medium setting in 87% of cases. The top five injection sites were glabella (82%), forehead (69%), periorbital area (64%), neck (40%), and jawline and/or masseters (13%).
The researchers noted one case (0.06%) where apraclonidine eye drops were prescribed for ptosis. The patient had undergone eight other single-session treatments without issue. There were no other documented adverse events directly related to spread of BoNT-A. According to Dr. Wang, this rate of ptosis is lower than the incidence with BoNT-A alone in two landmark trials studying its effects on glabellar lines, which was reported as 5.4% and 1.0%.
Among the 398 patients who received soft-tissue fillers, most (94%) were female, their mean age was 54 years, and 99% were treated with a 1927nm wavelength at a medium setting in 97% of cases. The top five injection sites were cheeks and/or tear troughs (89%), perioral area and/or marionette lines (77%), lips (34%), nasolabial folds (19%), and temples (11%), and the mean number of filler syringes per treatment was 1.6. Slightly more than half (51%) had 1 session, while the remainder had 2 to greater than 10 sessions. The researchers observed no documented adverse events related to spread of fillers or laser treatment of filled areas.
“This laser is a low-powered device that creates small, superficial, and transient microchannels, which likely contributes to the safety of single-session treatments with cosmetic injectables,” Dr. Wang said. However, prospective studies are needed to further validate these results, he added.
“With this very mild laser, it is not surprising that combined treatment had no effect,” said Eric F. Bernstein, MD, MSE, director of the Main Line Center for Laser Surgery in Ardmore, Pa., who was asked to comment on the study results. “There have been numerous anecdotal reports of spreading of botulinum toxin effect to areas not in the target area for treatment following a variety of lasers, including the more powerful version of the laser used in this study. In addition, spread following vascular and other lasers has been reported,” he noted
The laser used in this study, Dr. Bernstein continued, “is low powered and emits a wavelength that is very superficially absorbed, resulting in injury to the stratum corneum, superficial epidermis, or possibly the very superficial dermis, and is often used by physician extenders and not physicians – although I suspect this is not the case in the current study. One can have a reasonable degree of confidence when combining this laser with injectables, but these results cannot be extrapolated to other devices.”
The abstract received the annual ASDS Carruthers Award during the meeting. Dr. Wang reported that he is a consultant or advisor to Allergan, Alastin, AVAVA, Cynosure, Lutronic, Novoxel, Sofwave, and Solta. Dr. Bernstein reported having received research funding from Cynosure, Candela, and Acclaro. He also has received consulting fees from Cynosure and holds ownership interest in Candela, Novoxel, OnSite, Joylux, and Acclaro and has served on the advisory board for Novoxel, Cynosure, and Acclaro.
, results from a 6-year, single-center review showed.
“These treatments can be complementary in single-session treatments and can offer increased convenience for both patients and physicians,” primary study author Jordan V. Wang, MD, MBE, MBA, said during a virtual abstract session at the annual meeting of the American Society for Dermatologic Surgery.
To date, limited studies have demonstrated the safety of pairing botulinum neurotoxin type A and soft-tissue fillers with laser and other energy-based devices during the same treatment session on the same day, said Dr. Wang, medical research director at the Laser & Skin Surgery Center of New York. “Some concerns remain, though, regarding patient safety and efficacy,” he said. “Data on single-session treatment with low-power, low-density 1,927-nm and 1,440-nm fractional diode lasers and either botulinum neurotoxin or fillers are lacking.”
In a retrospective review of electronic medical records conducted from May 2015 to April 2021, Dr. Wang, Roy G. Geronemus, MD, and Carolyn Kushner, MD evaluated patients who received a single-session facial treatment with either BoNT-A or soft-tissue fillers and the low-power, low-density 1,927-nm and 1,440-nm fractional diode lasers (Clear+Brilliant Perméa and Original, Solta, Pleasanton, Calif.). Safety was assessed by documenting adverse events related to the spread of BoNT-A and fillers or laser treatment of filled areas within 4 weeks.
Adverse events they looked for related to botulinum neurotoxin use included eyelid ptosis; neck weakness or spasms; impairments in chewing, swallowing, speech, and respiration; and prescriptions of apraclonidine eye drops. Filler-related adverse events they looked for included product migration, unexpected loss of filler volume, vascular occlusion, acute pain, necrosis, blindness, and burn. “For both, we looked at hospital or emergency room transfers or admissions and referrals to ENT or ophthalmology,” Dr. Wang said.
During the 6-year study period, 525 patients had 1,562 single-session laser treatments with a mean 46.4 units of BoNT-A, and 398 patients had 1,237 single-session treatments with a mean 1.6 soft-tissue filler syringes. Among those who received BoNT-A, most (93%) were female, their mean age was 51 years, and 99% were treated with a 1,927-nm wavelength at a medium setting in 87% of cases. The top five injection sites were glabella (82%), forehead (69%), periorbital area (64%), neck (40%), and jawline and/or masseters (13%).
The researchers noted one case (0.06%) where apraclonidine eye drops were prescribed for ptosis. The patient had undergone eight other single-session treatments without issue. There were no other documented adverse events directly related to spread of BoNT-A. According to Dr. Wang, this rate of ptosis is lower than the incidence with BoNT-A alone in two landmark trials studying its effects on glabellar lines, which was reported as 5.4% and 1.0%.
Among the 398 patients who received soft-tissue fillers, most (94%) were female, their mean age was 54 years, and 99% were treated with a 1927nm wavelength at a medium setting in 97% of cases. The top five injection sites were cheeks and/or tear troughs (89%), perioral area and/or marionette lines (77%), lips (34%), nasolabial folds (19%), and temples (11%), and the mean number of filler syringes per treatment was 1.6. Slightly more than half (51%) had 1 session, while the remainder had 2 to greater than 10 sessions. The researchers observed no documented adverse events related to spread of fillers or laser treatment of filled areas.
“This laser is a low-powered device that creates small, superficial, and transient microchannels, which likely contributes to the safety of single-session treatments with cosmetic injectables,” Dr. Wang said. However, prospective studies are needed to further validate these results, he added.
“With this very mild laser, it is not surprising that combined treatment had no effect,” said Eric F. Bernstein, MD, MSE, director of the Main Line Center for Laser Surgery in Ardmore, Pa., who was asked to comment on the study results. “There have been numerous anecdotal reports of spreading of botulinum toxin effect to areas not in the target area for treatment following a variety of lasers, including the more powerful version of the laser used in this study. In addition, spread following vascular and other lasers has been reported,” he noted
The laser used in this study, Dr. Bernstein continued, “is low powered and emits a wavelength that is very superficially absorbed, resulting in injury to the stratum corneum, superficial epidermis, or possibly the very superficial dermis, and is often used by physician extenders and not physicians – although I suspect this is not the case in the current study. One can have a reasonable degree of confidence when combining this laser with injectables, but these results cannot be extrapolated to other devices.”
The abstract received the annual ASDS Carruthers Award during the meeting. Dr. Wang reported that he is a consultant or advisor to Allergan, Alastin, AVAVA, Cynosure, Lutronic, Novoxel, Sofwave, and Solta. Dr. Bernstein reported having received research funding from Cynosure, Candela, and Acclaro. He also has received consulting fees from Cynosure and holds ownership interest in Candela, Novoxel, OnSite, Joylux, and Acclaro and has served on the advisory board for Novoxel, Cynosure, and Acclaro.
, results from a 6-year, single-center review showed.
“These treatments can be complementary in single-session treatments and can offer increased convenience for both patients and physicians,” primary study author Jordan V. Wang, MD, MBE, MBA, said during a virtual abstract session at the annual meeting of the American Society for Dermatologic Surgery.
To date, limited studies have demonstrated the safety of pairing botulinum neurotoxin type A and soft-tissue fillers with laser and other energy-based devices during the same treatment session on the same day, said Dr. Wang, medical research director at the Laser & Skin Surgery Center of New York. “Some concerns remain, though, regarding patient safety and efficacy,” he said. “Data on single-session treatment with low-power, low-density 1,927-nm and 1,440-nm fractional diode lasers and either botulinum neurotoxin or fillers are lacking.”
In a retrospective review of electronic medical records conducted from May 2015 to April 2021, Dr. Wang, Roy G. Geronemus, MD, and Carolyn Kushner, MD evaluated patients who received a single-session facial treatment with either BoNT-A or soft-tissue fillers and the low-power, low-density 1,927-nm and 1,440-nm fractional diode lasers (Clear+Brilliant Perméa and Original, Solta, Pleasanton, Calif.). Safety was assessed by documenting adverse events related to the spread of BoNT-A and fillers or laser treatment of filled areas within 4 weeks.
Adverse events they looked for related to botulinum neurotoxin use included eyelid ptosis; neck weakness or spasms; impairments in chewing, swallowing, speech, and respiration; and prescriptions of apraclonidine eye drops. Filler-related adverse events they looked for included product migration, unexpected loss of filler volume, vascular occlusion, acute pain, necrosis, blindness, and burn. “For both, we looked at hospital or emergency room transfers or admissions and referrals to ENT or ophthalmology,” Dr. Wang said.
During the 6-year study period, 525 patients had 1,562 single-session laser treatments with a mean 46.4 units of BoNT-A, and 398 patients had 1,237 single-session treatments with a mean 1.6 soft-tissue filler syringes. Among those who received BoNT-A, most (93%) were female, their mean age was 51 years, and 99% were treated with a 1,927-nm wavelength at a medium setting in 87% of cases. The top five injection sites were glabella (82%), forehead (69%), periorbital area (64%), neck (40%), and jawline and/or masseters (13%).
The researchers noted one case (0.06%) where apraclonidine eye drops were prescribed for ptosis. The patient had undergone eight other single-session treatments without issue. There were no other documented adverse events directly related to spread of BoNT-A. According to Dr. Wang, this rate of ptosis is lower than the incidence with BoNT-A alone in two landmark trials studying its effects on glabellar lines, which was reported as 5.4% and 1.0%.
Among the 398 patients who received soft-tissue fillers, most (94%) were female, their mean age was 54 years, and 99% were treated with a 1927nm wavelength at a medium setting in 97% of cases. The top five injection sites were cheeks and/or tear troughs (89%), perioral area and/or marionette lines (77%), lips (34%), nasolabial folds (19%), and temples (11%), and the mean number of filler syringes per treatment was 1.6. Slightly more than half (51%) had 1 session, while the remainder had 2 to greater than 10 sessions. The researchers observed no documented adverse events related to spread of fillers or laser treatment of filled areas.
“This laser is a low-powered device that creates small, superficial, and transient microchannels, which likely contributes to the safety of single-session treatments with cosmetic injectables,” Dr. Wang said. However, prospective studies are needed to further validate these results, he added.
“With this very mild laser, it is not surprising that combined treatment had no effect,” said Eric F. Bernstein, MD, MSE, director of the Main Line Center for Laser Surgery in Ardmore, Pa., who was asked to comment on the study results. “There have been numerous anecdotal reports of spreading of botulinum toxin effect to areas not in the target area for treatment following a variety of lasers, including the more powerful version of the laser used in this study. In addition, spread following vascular and other lasers has been reported,” he noted
The laser used in this study, Dr. Bernstein continued, “is low powered and emits a wavelength that is very superficially absorbed, resulting in injury to the stratum corneum, superficial epidermis, or possibly the very superficial dermis, and is often used by physician extenders and not physicians – although I suspect this is not the case in the current study. One can have a reasonable degree of confidence when combining this laser with injectables, but these results cannot be extrapolated to other devices.”
The abstract received the annual ASDS Carruthers Award during the meeting. Dr. Wang reported that he is a consultant or advisor to Allergan, Alastin, AVAVA, Cynosure, Lutronic, Novoxel, Sofwave, and Solta. Dr. Bernstein reported having received research funding from Cynosure, Candela, and Acclaro. He also has received consulting fees from Cynosure and holds ownership interest in Candela, Novoxel, OnSite, Joylux, and Acclaro and has served on the advisory board for Novoxel, Cynosure, and Acclaro.
FROM ASDS 2021
Ophthalmologist who developed medical botox dies at 89
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
BMJ slams ‘incompetent’ Facebook fact-checking of vaccine article
According to an open letter written by outgoing BMJ editor-in-chief Fiona Godlee, MD, and incoming editor-in-chief Kamran Abbasi, MD, Facebook hired a third-party contractor to evaluate the article’s findings. This resulted in “inaccurate, incompetent, and irresponsible” conclusions that “should be of concern to anyone who values and relies on sources such as the BMJ for reliable medical information.”
The article in question investigated data integrity concerns at Pfizer vaccine clinical trial sites. In September 2020, the letter states, a former employee of the research group involved in Pfizer’s main vaccine trials, Ventavia, reached out to the BMJ and “began providing ... dozens of internal company documents, photos, audio recordings, and emails.” According to the company’s website, Ventavia “played a significant part in [COVID-19 clinical trial] recruitment” and “has received recognition by Pfizer for their contribution to vaccine trials.”
It was previously reported that the whistle-blower is a former regional director who was involved in Pfizer’s vaccine trials in Texas during the fall of 2020. She alleges “the company falsified data, unblinded patients, employed inadequately trained vaccinators, and was slow to follow up on adverse events reported in Pfizer’s pivotal phase 3 trial.”
The images provided to the BMJ “showed needles discarded in a plastic biohazard bag instead of a sharps container box” and another displayed “vaccine packaging materials with trial participants’ identification numbers written on them left out in the open, potentially unblinding participants.”
Despite informing Ventavia, the director’s concerns went unaddressed. She then filed a complaint with the Food and Drug Administration and was subsequently fired the same day. The FDA did not investigate the director’s allegations, said Dr. Godlee and Dr. Abbasi, even though the evidence “revealed a host of poor clinical trial research practices occurring at Ventavia that could impact data integrity and patient safety.”
Article labeled as ‘hoax,’ without pointing out errors
The BMJ hired an investigative reporter to follow up on the clinical trial claims. The findings were published in an article on Nov. 2, 2021, after the article “went through ... the usual high-level legal and editorial oversight and peer review,” according to the journal.
However, by Nov. 10, the journal began receiving complaints from readers unable to share the article on social media. Others had their posts flagged with warnings, such as “missing context ... independent fact-checkers say this information could mislead people.” Administrators of various Facebook groups were notified that posts containing the article were “partly false.”
Readers were informed that Facebook contractor Lead Stories performed the article’s “fact check.” Lead Stories is “an award-winning innovative fact checking and debunking website” and “an active part of Facebook’s partnership with third-party fact checkers” – with the latter granting them “access to listings of content that has been flagged as potentially false by Facebook’s systems or its users.” The company said they “decide independently if we want to fact check it or not.”
Lead Stories stated that they “can enter our fact checks into a tool provided by Facebook and Facebook then uses our data to help slow down the spread of false information on its platform.” Although the contractor is compensated, Lead Stories claims they have “no say or influence over what we fact check or what our conclusions are.”
Both editors question the validity of the fact check performed by Lead Stories, as it failed to provide any “assertions of fact” as to what the BMJ got wrong. Moreover, the editors take issue with Lead Stories referring to the journal as a “news blog” and using the phrase “hoax-alert” in the URL when publishing the story on its site.
The BMJ has reached out to Lead Stories and Facebook, said the letter, but Lead Stories refuses to “change anything about their article or actions that have led to Facebook flagging our article.” Requests for Facebook to remove the “fact-checking” label and allow “readers to freely share the article on [Facebook’s] platform” have been unfruitful.
Dr. Godlee and Dr. Abbasi expressed concern that other “high quality information provider[s] have been affected by the incompetence of Meta’s fact checking regime.” In November, Instagram censored Cochrane, an international provider of independent systematic medical reviews. Instagram, also owned by Meta, prohibited users from tagging Cochrane because the organization “repeatedly posted ... false content about COVID-19 or vaccines.” Cochrane refuted the allegations.
While “fact checking has been a staple of good journalism for decades,” said the editors, Meta has “apparently delegated responsibility to people incompetent in carrying out this crucial task.” They urged the company to reconsider its fact-checking strategy and review the issues that contributed to the error.
This news organization reached out to Meta for comment but did not receive a response at press time.
Lead Stories has posted a reply (Lead Stories’ Response To BMJ Open Letter Objecting To A Lead Stories Fact Check) to the BMJ’s complaint on its website.
A version of this article first appeared on Medscape.com.
According to an open letter written by outgoing BMJ editor-in-chief Fiona Godlee, MD, and incoming editor-in-chief Kamran Abbasi, MD, Facebook hired a third-party contractor to evaluate the article’s findings. This resulted in “inaccurate, incompetent, and irresponsible” conclusions that “should be of concern to anyone who values and relies on sources such as the BMJ for reliable medical information.”
The article in question investigated data integrity concerns at Pfizer vaccine clinical trial sites. In September 2020, the letter states, a former employee of the research group involved in Pfizer’s main vaccine trials, Ventavia, reached out to the BMJ and “began providing ... dozens of internal company documents, photos, audio recordings, and emails.” According to the company’s website, Ventavia “played a significant part in [COVID-19 clinical trial] recruitment” and “has received recognition by Pfizer for their contribution to vaccine trials.”
It was previously reported that the whistle-blower is a former regional director who was involved in Pfizer’s vaccine trials in Texas during the fall of 2020. She alleges “the company falsified data, unblinded patients, employed inadequately trained vaccinators, and was slow to follow up on adverse events reported in Pfizer’s pivotal phase 3 trial.”
The images provided to the BMJ “showed needles discarded in a plastic biohazard bag instead of a sharps container box” and another displayed “vaccine packaging materials with trial participants’ identification numbers written on them left out in the open, potentially unblinding participants.”
Despite informing Ventavia, the director’s concerns went unaddressed. She then filed a complaint with the Food and Drug Administration and was subsequently fired the same day. The FDA did not investigate the director’s allegations, said Dr. Godlee and Dr. Abbasi, even though the evidence “revealed a host of poor clinical trial research practices occurring at Ventavia that could impact data integrity and patient safety.”
Article labeled as ‘hoax,’ without pointing out errors
The BMJ hired an investigative reporter to follow up on the clinical trial claims. The findings were published in an article on Nov. 2, 2021, after the article “went through ... the usual high-level legal and editorial oversight and peer review,” according to the journal.
However, by Nov. 10, the journal began receiving complaints from readers unable to share the article on social media. Others had their posts flagged with warnings, such as “missing context ... independent fact-checkers say this information could mislead people.” Administrators of various Facebook groups were notified that posts containing the article were “partly false.”
Readers were informed that Facebook contractor Lead Stories performed the article’s “fact check.” Lead Stories is “an award-winning innovative fact checking and debunking website” and “an active part of Facebook’s partnership with third-party fact checkers” – with the latter granting them “access to listings of content that has been flagged as potentially false by Facebook’s systems or its users.” The company said they “decide independently if we want to fact check it or not.”
Lead Stories stated that they “can enter our fact checks into a tool provided by Facebook and Facebook then uses our data to help slow down the spread of false information on its platform.” Although the contractor is compensated, Lead Stories claims they have “no say or influence over what we fact check or what our conclusions are.”
Both editors question the validity of the fact check performed by Lead Stories, as it failed to provide any “assertions of fact” as to what the BMJ got wrong. Moreover, the editors take issue with Lead Stories referring to the journal as a “news blog” and using the phrase “hoax-alert” in the URL when publishing the story on its site.
The BMJ has reached out to Lead Stories and Facebook, said the letter, but Lead Stories refuses to “change anything about their article or actions that have led to Facebook flagging our article.” Requests for Facebook to remove the “fact-checking” label and allow “readers to freely share the article on [Facebook’s] platform” have been unfruitful.
Dr. Godlee and Dr. Abbasi expressed concern that other “high quality information provider[s] have been affected by the incompetence of Meta’s fact checking regime.” In November, Instagram censored Cochrane, an international provider of independent systematic medical reviews. Instagram, also owned by Meta, prohibited users from tagging Cochrane because the organization “repeatedly posted ... false content about COVID-19 or vaccines.” Cochrane refuted the allegations.
While “fact checking has been a staple of good journalism for decades,” said the editors, Meta has “apparently delegated responsibility to people incompetent in carrying out this crucial task.” They urged the company to reconsider its fact-checking strategy and review the issues that contributed to the error.
This news organization reached out to Meta for comment but did not receive a response at press time.
Lead Stories has posted a reply (Lead Stories’ Response To BMJ Open Letter Objecting To A Lead Stories Fact Check) to the BMJ’s complaint on its website.
A version of this article first appeared on Medscape.com.
According to an open letter written by outgoing BMJ editor-in-chief Fiona Godlee, MD, and incoming editor-in-chief Kamran Abbasi, MD, Facebook hired a third-party contractor to evaluate the article’s findings. This resulted in “inaccurate, incompetent, and irresponsible” conclusions that “should be of concern to anyone who values and relies on sources such as the BMJ for reliable medical information.”
The article in question investigated data integrity concerns at Pfizer vaccine clinical trial sites. In September 2020, the letter states, a former employee of the research group involved in Pfizer’s main vaccine trials, Ventavia, reached out to the BMJ and “began providing ... dozens of internal company documents, photos, audio recordings, and emails.” According to the company’s website, Ventavia “played a significant part in [COVID-19 clinical trial] recruitment” and “has received recognition by Pfizer for their contribution to vaccine trials.”
It was previously reported that the whistle-blower is a former regional director who was involved in Pfizer’s vaccine trials in Texas during the fall of 2020. She alleges “the company falsified data, unblinded patients, employed inadequately trained vaccinators, and was slow to follow up on adverse events reported in Pfizer’s pivotal phase 3 trial.”
The images provided to the BMJ “showed needles discarded in a plastic biohazard bag instead of a sharps container box” and another displayed “vaccine packaging materials with trial participants’ identification numbers written on them left out in the open, potentially unblinding participants.”
Despite informing Ventavia, the director’s concerns went unaddressed. She then filed a complaint with the Food and Drug Administration and was subsequently fired the same day. The FDA did not investigate the director’s allegations, said Dr. Godlee and Dr. Abbasi, even though the evidence “revealed a host of poor clinical trial research practices occurring at Ventavia that could impact data integrity and patient safety.”
Article labeled as ‘hoax,’ without pointing out errors
The BMJ hired an investigative reporter to follow up on the clinical trial claims. The findings were published in an article on Nov. 2, 2021, after the article “went through ... the usual high-level legal and editorial oversight and peer review,” according to the journal.
However, by Nov. 10, the journal began receiving complaints from readers unable to share the article on social media. Others had their posts flagged with warnings, such as “missing context ... independent fact-checkers say this information could mislead people.” Administrators of various Facebook groups were notified that posts containing the article were “partly false.”
Readers were informed that Facebook contractor Lead Stories performed the article’s “fact check.” Lead Stories is “an award-winning innovative fact checking and debunking website” and “an active part of Facebook’s partnership with third-party fact checkers” – with the latter granting them “access to listings of content that has been flagged as potentially false by Facebook’s systems or its users.” The company said they “decide independently if we want to fact check it or not.”
Lead Stories stated that they “can enter our fact checks into a tool provided by Facebook and Facebook then uses our data to help slow down the spread of false information on its platform.” Although the contractor is compensated, Lead Stories claims they have “no say or influence over what we fact check or what our conclusions are.”
Both editors question the validity of the fact check performed by Lead Stories, as it failed to provide any “assertions of fact” as to what the BMJ got wrong. Moreover, the editors take issue with Lead Stories referring to the journal as a “news blog” and using the phrase “hoax-alert” in the URL when publishing the story on its site.
The BMJ has reached out to Lead Stories and Facebook, said the letter, but Lead Stories refuses to “change anything about their article or actions that have led to Facebook flagging our article.” Requests for Facebook to remove the “fact-checking” label and allow “readers to freely share the article on [Facebook’s] platform” have been unfruitful.
Dr. Godlee and Dr. Abbasi expressed concern that other “high quality information provider[s] have been affected by the incompetence of Meta’s fact checking regime.” In November, Instagram censored Cochrane, an international provider of independent systematic medical reviews. Instagram, also owned by Meta, prohibited users from tagging Cochrane because the organization “repeatedly posted ... false content about COVID-19 or vaccines.” Cochrane refuted the allegations.
While “fact checking has been a staple of good journalism for decades,” said the editors, Meta has “apparently delegated responsibility to people incompetent in carrying out this crucial task.” They urged the company to reconsider its fact-checking strategy and review the issues that contributed to the error.
This news organization reached out to Meta for comment but did not receive a response at press time.
Lead Stories has posted a reply (Lead Stories’ Response To BMJ Open Letter Objecting To A Lead Stories Fact Check) to the BMJ’s complaint on its website.
A version of this article first appeared on Medscape.com.
Zosteriform Eruption on the Chest and Abdomen
THE DIAGNOSIS:
Cutaneous Metastatic Mesothelioma
Biopsies of the larger erythematous papules revealed an infiltrate of atypical tumor cells with mitoses (Figure 1) that were immunoreactive for calretinin (Figure 2) and lacked nuclear BRCA1 associated protein-1, BAP1, expression (not shown). The patient’s prior mesothelioma was re-reviewed, and the cutaneous tumor cells were similar to the primary mesothelioma. A diagnosis of cutaneous metastatic mesothelioma (CMM) was made.
Mesothelioma is a rare neoplasm arising from the pleura, pericardium, peritoneum, and tunica vaginalis,1 with an estimated annual incidence of 2500 cases.2 The predominant risk factor for the development of pleural mesothelioma is asbestos exposure, which has been identified in up to 90% of cases. Mesothelioma can give rise to local and less frequently distant hematogenous metastases. Cutaneous involvement of mesothelioma is rare.3 More than 80% of CMM cases are attributed to seeding the skin at procedure sites or by direct infiltration of scars. Distant CMM is rare and typically presents as subcutaneous nodules.4 Few cases of inflammatory CMM have been published,1,4,5 with even fewer mimicking herpes zoster infection (HZI), as seen in our patient.
The most specific stain for mesothelioma is calretinin, which strongly and diffusely stains both the nucleus and cytoplasm. Other markers include Wilms tumor 1, cytokeratin 5/6, thrombomodulin, and HBME-1. Immunohistochemistry to detect the loss of BAP1 staining in the nucleus is important for differentiating between mesothelioma and mesothelial hyperplasia.3
Cutaneous metastases occur in 0.7% to 9% of patients with internal malignant disease. Most commonly, cutaneous metastases present as cutaneous nodules, though other reported inflammatory presentations include erysipeloides, generalized erythematous patches, telangiectasia, and zosteriform distributions.6 Zosteriform distributions are particularly rare and most commonly are due to breast carcinomas or lymphomas. The mechanism of zosteriform metastasis is unknown, but theories include tumoral spread along vessels, invasion of the thoracic perineural sheaths, localized spread of tumor cells from a surgical site, or a Koebner-like reaction at the site of an existing HZI. Regardless of primary tumor type or presentation, cutaneous metastasis is a poor prognostic sign, with survival rates varying based on primary tumor type.7
Other differential diagnoses include herpes zoster granulomatous dermatitis, radiation recall dermatitis, cutaneous Rosai-Dorfman disease, and zosteriform lichen planus, all of which have been reported after HZI.8-10 Herpes zoster granulomatous dermatitis typically presents weeks to years after acute HZI with erythematous to violaceous papules and plaques at the site of the prior HZI. A biopsy reveals interstitial granulomatous dermatitis and multinucleated giant cells.8 Radiation recall dermatitis is a cutaneous inflammatory reaction limited to regions of prior radiation exposure after the administration of a triggering medication. Radiation recall dermatitis can present days to many years after the completion of treatment.9 Although the eruption in our patient was at the site of prior radiation, the pathologic and clinical presentation was not consistent with radiation recall dermatitis. Cutaneous Rosai-Dorfman disease is a non-Langerhans cell histiocytosis that may present as either solitary or numerous papules, plaques, or nodules and has been reported to occur after HZI. Biopsy reveals a diffuse dermal histiocytic infiltration with plasma cells and lymphocytes. In contrast to metastatic disease, mitoses and nuclear atypia are rare in cutaneous RosaiDorfman disease.11 Lichen planus is an inflammatory disease of unknown etiology presenting as flat-topped, violaceous, pruritic papules12 that may present in a zosteriform pattern.13
Although it is uncommon, metastatic spread should be considered in patients with known malignancy presenting with zosteriform eruptions.2 Our patient remained on treatment with immunotherapy, as he was unable to undergo additional radiation and had failed multiple other lines of therapy. He died 3 months after presentation.
- Klebanov N, Reddy BY, Husain S, et al. Cutaneous presentation of mesothelioma with a sarcomatoid transformation. Am J Dermatopathol. 2018;40:378-382.
- Patel SC, Dowell JE. Modern management of malignant pleural mesothelioma. Lung Cancer (Auckl). 2016;7:63-72.
- Ward RE, Ali SA, Kuhar M. Epithelioid malignant mesothelioma metastatic to the skin: a case report and review of the literature. J Cutan Pathol. 2017;44:1057-1063.
- Prieto VG, Kenet BJ, Varghese M. Malignant mesothelioma metastatic to the skin, presenting as inflammatory carcinoma. Am J Dermatopathol. 1997;19:261-265.
- Gaudy-Marqueste C, Dales JP, Collet-Villette AM, et al. Cutaneous metastasis of pleural mesothelioma: two cases [in French]. Ann Dermatol Venereol. 2003;130:455-459.
- Chiang A, Salomon N, Gaikwad R, et al. A case of cutaneous metastasis mimicking herpes zoster rash. IDCases. 2018;12:167-168.
- Thomaidou E, Armon G, Klapholz L, et al. Zosteriform cutaneous metastases. Clin Exp Dermatol. 2018;43:734-736.
- Ferenczi K, Rosenberg AS, McCalmont TH, et al. Herpes zoster granulomatous dermatitis: histopathologic findings in a case series. J Cutan Pathol. 2015;42:739-745.
- Carrasco L, Pastor MA, Izquierdo MJ, et al. Drug eruption secondary to acyclovir with recall phenomenon in a dermatome previously affected by herpes zoster. Clin Exp Dermatol. 2002;27:132-134.
- Malviya N, Marzuka A, Maamed-Tayeb M, et al. Cutaneous involvement of pre-existing Rosai-Dorfman disease via post-herpetic isotopic response. J Cutan Pathol. 2016;43:1211-1214.
- Fang S, Chen AJ. Facial cutaneous Rosai-Dorfman disease: a case report and literature review. Exp Ther Med. 2015;9:1389-1392.
- Le Cleach L, Chosidow O. Clinical practice. lichen planus. N Engl J Med. 2012;366:723-732.
- Fink-Puches R, Hofmann-Wellenhof R, Smolle J. Zosteriform lichen planus. Dermatology. 1996;192:375-377.
THE DIAGNOSIS:
Cutaneous Metastatic Mesothelioma
Biopsies of the larger erythematous papules revealed an infiltrate of atypical tumor cells with mitoses (Figure 1) that were immunoreactive for calretinin (Figure 2) and lacked nuclear BRCA1 associated protein-1, BAP1, expression (not shown). The patient’s prior mesothelioma was re-reviewed, and the cutaneous tumor cells were similar to the primary mesothelioma. A diagnosis of cutaneous metastatic mesothelioma (CMM) was made.


Mesothelioma is a rare neoplasm arising from the pleura, pericardium, peritoneum, and tunica vaginalis,1 with an estimated annual incidence of 2500 cases.2 The predominant risk factor for the development of pleural mesothelioma is asbestos exposure, which has been identified in up to 90% of cases. Mesothelioma can give rise to local and less frequently distant hematogenous metastases. Cutaneous involvement of mesothelioma is rare.3 More than 80% of CMM cases are attributed to seeding the skin at procedure sites or by direct infiltration of scars. Distant CMM is rare and typically presents as subcutaneous nodules.4 Few cases of inflammatory CMM have been published,1,4,5 with even fewer mimicking herpes zoster infection (HZI), as seen in our patient.
The most specific stain for mesothelioma is calretinin, which strongly and diffusely stains both the nucleus and cytoplasm. Other markers include Wilms tumor 1, cytokeratin 5/6, thrombomodulin, and HBME-1. Immunohistochemistry to detect the loss of BAP1 staining in the nucleus is important for differentiating between mesothelioma and mesothelial hyperplasia.3
Cutaneous metastases occur in 0.7% to 9% of patients with internal malignant disease. Most commonly, cutaneous metastases present as cutaneous nodules, though other reported inflammatory presentations include erysipeloides, generalized erythematous patches, telangiectasia, and zosteriform distributions.6 Zosteriform distributions are particularly rare and most commonly are due to breast carcinomas or lymphomas. The mechanism of zosteriform metastasis is unknown, but theories include tumoral spread along vessels, invasion of the thoracic perineural sheaths, localized spread of tumor cells from a surgical site, or a Koebner-like reaction at the site of an existing HZI. Regardless of primary tumor type or presentation, cutaneous metastasis is a poor prognostic sign, with survival rates varying based on primary tumor type.7
Other differential diagnoses include herpes zoster granulomatous dermatitis, radiation recall dermatitis, cutaneous Rosai-Dorfman disease, and zosteriform lichen planus, all of which have been reported after HZI.8-10 Herpes zoster granulomatous dermatitis typically presents weeks to years after acute HZI with erythematous to violaceous papules and plaques at the site of the prior HZI. A biopsy reveals interstitial granulomatous dermatitis and multinucleated giant cells.8 Radiation recall dermatitis is a cutaneous inflammatory reaction limited to regions of prior radiation exposure after the administration of a triggering medication. Radiation recall dermatitis can present days to many years after the completion of treatment.9 Although the eruption in our patient was at the site of prior radiation, the pathologic and clinical presentation was not consistent with radiation recall dermatitis. Cutaneous Rosai-Dorfman disease is a non-Langerhans cell histiocytosis that may present as either solitary or numerous papules, plaques, or nodules and has been reported to occur after HZI. Biopsy reveals a diffuse dermal histiocytic infiltration with plasma cells and lymphocytes. In contrast to metastatic disease, mitoses and nuclear atypia are rare in cutaneous RosaiDorfman disease.11 Lichen planus is an inflammatory disease of unknown etiology presenting as flat-topped, violaceous, pruritic papules12 that may present in a zosteriform pattern.13
Although it is uncommon, metastatic spread should be considered in patients with known malignancy presenting with zosteriform eruptions.2 Our patient remained on treatment with immunotherapy, as he was unable to undergo additional radiation and had failed multiple other lines of therapy. He died 3 months after presentation.
THE DIAGNOSIS:
Cutaneous Metastatic Mesothelioma
Biopsies of the larger erythematous papules revealed an infiltrate of atypical tumor cells with mitoses (Figure 1) that were immunoreactive for calretinin (Figure 2) and lacked nuclear BRCA1 associated protein-1, BAP1, expression (not shown). The patient’s prior mesothelioma was re-reviewed, and the cutaneous tumor cells were similar to the primary mesothelioma. A diagnosis of cutaneous metastatic mesothelioma (CMM) was made.


Mesothelioma is a rare neoplasm arising from the pleura, pericardium, peritoneum, and tunica vaginalis,1 with an estimated annual incidence of 2500 cases.2 The predominant risk factor for the development of pleural mesothelioma is asbestos exposure, which has been identified in up to 90% of cases. Mesothelioma can give rise to local and less frequently distant hematogenous metastases. Cutaneous involvement of mesothelioma is rare.3 More than 80% of CMM cases are attributed to seeding the skin at procedure sites or by direct infiltration of scars. Distant CMM is rare and typically presents as subcutaneous nodules.4 Few cases of inflammatory CMM have been published,1,4,5 with even fewer mimicking herpes zoster infection (HZI), as seen in our patient.
The most specific stain for mesothelioma is calretinin, which strongly and diffusely stains both the nucleus and cytoplasm. Other markers include Wilms tumor 1, cytokeratin 5/6, thrombomodulin, and HBME-1. Immunohistochemistry to detect the loss of BAP1 staining in the nucleus is important for differentiating between mesothelioma and mesothelial hyperplasia.3
Cutaneous metastases occur in 0.7% to 9% of patients with internal malignant disease. Most commonly, cutaneous metastases present as cutaneous nodules, though other reported inflammatory presentations include erysipeloides, generalized erythematous patches, telangiectasia, and zosteriform distributions.6 Zosteriform distributions are particularly rare and most commonly are due to breast carcinomas or lymphomas. The mechanism of zosteriform metastasis is unknown, but theories include tumoral spread along vessels, invasion of the thoracic perineural sheaths, localized spread of tumor cells from a surgical site, or a Koebner-like reaction at the site of an existing HZI. Regardless of primary tumor type or presentation, cutaneous metastasis is a poor prognostic sign, with survival rates varying based on primary tumor type.7
Other differential diagnoses include herpes zoster granulomatous dermatitis, radiation recall dermatitis, cutaneous Rosai-Dorfman disease, and zosteriform lichen planus, all of which have been reported after HZI.8-10 Herpes zoster granulomatous dermatitis typically presents weeks to years after acute HZI with erythematous to violaceous papules and plaques at the site of the prior HZI. A biopsy reveals interstitial granulomatous dermatitis and multinucleated giant cells.8 Radiation recall dermatitis is a cutaneous inflammatory reaction limited to regions of prior radiation exposure after the administration of a triggering medication. Radiation recall dermatitis can present days to many years after the completion of treatment.9 Although the eruption in our patient was at the site of prior radiation, the pathologic and clinical presentation was not consistent with radiation recall dermatitis. Cutaneous Rosai-Dorfman disease is a non-Langerhans cell histiocytosis that may present as either solitary or numerous papules, plaques, or nodules and has been reported to occur after HZI. Biopsy reveals a diffuse dermal histiocytic infiltration with plasma cells and lymphocytes. In contrast to metastatic disease, mitoses and nuclear atypia are rare in cutaneous RosaiDorfman disease.11 Lichen planus is an inflammatory disease of unknown etiology presenting as flat-topped, violaceous, pruritic papules12 that may present in a zosteriform pattern.13
Although it is uncommon, metastatic spread should be considered in patients with known malignancy presenting with zosteriform eruptions.2 Our patient remained on treatment with immunotherapy, as he was unable to undergo additional radiation and had failed multiple other lines of therapy. He died 3 months after presentation.
- Klebanov N, Reddy BY, Husain S, et al. Cutaneous presentation of mesothelioma with a sarcomatoid transformation. Am J Dermatopathol. 2018;40:378-382.
- Patel SC, Dowell JE. Modern management of malignant pleural mesothelioma. Lung Cancer (Auckl). 2016;7:63-72.
- Ward RE, Ali SA, Kuhar M. Epithelioid malignant mesothelioma metastatic to the skin: a case report and review of the literature. J Cutan Pathol. 2017;44:1057-1063.
- Prieto VG, Kenet BJ, Varghese M. Malignant mesothelioma metastatic to the skin, presenting as inflammatory carcinoma. Am J Dermatopathol. 1997;19:261-265.
- Gaudy-Marqueste C, Dales JP, Collet-Villette AM, et al. Cutaneous metastasis of pleural mesothelioma: two cases [in French]. Ann Dermatol Venereol. 2003;130:455-459.
- Chiang A, Salomon N, Gaikwad R, et al. A case of cutaneous metastasis mimicking herpes zoster rash. IDCases. 2018;12:167-168.
- Thomaidou E, Armon G, Klapholz L, et al. Zosteriform cutaneous metastases. Clin Exp Dermatol. 2018;43:734-736.
- Ferenczi K, Rosenberg AS, McCalmont TH, et al. Herpes zoster granulomatous dermatitis: histopathologic findings in a case series. J Cutan Pathol. 2015;42:739-745.
- Carrasco L, Pastor MA, Izquierdo MJ, et al. Drug eruption secondary to acyclovir with recall phenomenon in a dermatome previously affected by herpes zoster. Clin Exp Dermatol. 2002;27:132-134.
- Malviya N, Marzuka A, Maamed-Tayeb M, et al. Cutaneous involvement of pre-existing Rosai-Dorfman disease via post-herpetic isotopic response. J Cutan Pathol. 2016;43:1211-1214.
- Fang S, Chen AJ. Facial cutaneous Rosai-Dorfman disease: a case report and literature review. Exp Ther Med. 2015;9:1389-1392.
- Le Cleach L, Chosidow O. Clinical practice. lichen planus. N Engl J Med. 2012;366:723-732.
- Fink-Puches R, Hofmann-Wellenhof R, Smolle J. Zosteriform lichen planus. Dermatology. 1996;192:375-377.
- Klebanov N, Reddy BY, Husain S, et al. Cutaneous presentation of mesothelioma with a sarcomatoid transformation. Am J Dermatopathol. 2018;40:378-382.
- Patel SC, Dowell JE. Modern management of malignant pleural mesothelioma. Lung Cancer (Auckl). 2016;7:63-72.
- Ward RE, Ali SA, Kuhar M. Epithelioid malignant mesothelioma metastatic to the skin: a case report and review of the literature. J Cutan Pathol. 2017;44:1057-1063.
- Prieto VG, Kenet BJ, Varghese M. Malignant mesothelioma metastatic to the skin, presenting as inflammatory carcinoma. Am J Dermatopathol. 1997;19:261-265.
- Gaudy-Marqueste C, Dales JP, Collet-Villette AM, et al. Cutaneous metastasis of pleural mesothelioma: two cases [in French]. Ann Dermatol Venereol. 2003;130:455-459.
- Chiang A, Salomon N, Gaikwad R, et al. A case of cutaneous metastasis mimicking herpes zoster rash. IDCases. 2018;12:167-168.
- Thomaidou E, Armon G, Klapholz L, et al. Zosteriform cutaneous metastases. Clin Exp Dermatol. 2018;43:734-736.
- Ferenczi K, Rosenberg AS, McCalmont TH, et al. Herpes zoster granulomatous dermatitis: histopathologic findings in a case series. J Cutan Pathol. 2015;42:739-745.
- Carrasco L, Pastor MA, Izquierdo MJ, et al. Drug eruption secondary to acyclovir with recall phenomenon in a dermatome previously affected by herpes zoster. Clin Exp Dermatol. 2002;27:132-134.
- Malviya N, Marzuka A, Maamed-Tayeb M, et al. Cutaneous involvement of pre-existing Rosai-Dorfman disease via post-herpetic isotopic response. J Cutan Pathol. 2016;43:1211-1214.
- Fang S, Chen AJ. Facial cutaneous Rosai-Dorfman disease: a case report and literature review. Exp Ther Med. 2015;9:1389-1392.
- Le Cleach L, Chosidow O. Clinical practice. lichen planus. N Engl J Med. 2012;366:723-732.
- Fink-Puches R, Hofmann-Wellenhof R, Smolle J. Zosteriform lichen planus. Dermatology. 1996;192:375-377.
A 50-year-old man presented with erythematous macules and papules with a dermatomal distribution on the left thoracic region with associated pain of 3 weeks’ duration. The lesions persisted after treatment for herpes zoster. His medical history was notable for mesothelioma that was diagnosed 6 years prior and was treated with ipilimumab and nivolumab following multiple lines of chemotherapy and investigational agents, left thoracotomy, extrapleural pneumonectomy, diaphragmatic reconstruction, and left chest radiation. His medical history also included Hodgkin lymphoma diagnosed 36 years prior that was treated with an appendectomy, splenectomy, systemic chemotherapy, and radiation. Three weeks prior to the current presentation, he was treated by oncology with valacyclovir 1 g 3 times daily for 7 days for presumed herpes zoster without improvement. Physical examination revealed the absence of vesicles, as well as firm, 1- to 6-mm, erythematous papules and plaques, including a few outside of the most affected dermatomes.
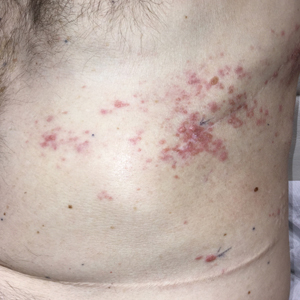
Blastomycosislike Pyoderma: Verrucous Hyperpigmented Plaques on the Pretibial Shins
To the Editor:
Blastomycosislike pyoderma (BLP), also commonly referred to as pyoderma vegetans, is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 It is characterized by a collection of neutrophilic abscesses with pseudoepitheliomatous hyperplasia that coalesce into crusted plaques.
A 15-year-old adolescent girl with a history of type 1 diabetes mellitus was admitted for diabetic ketoacidosis. The patient presented with bilateral pretibial lesions of 6 years’ duration that developed after swimming in a pool following reported trauma to the site. These pruritic plaques had grown slowly and were occasionally tender. Of note, with episodes of hyperglycemia, the lesions developed purulent drainage.
Upon admission to the hospital and subsequent dermatology consultation, physical examination revealed the right pretibial shin had a 15×5-cm, gray-brown, hyperpigmented, verrucous, tender plaque with purulent drainage and overlying crust (Figure 1). The left pretibial shin had a similar smaller lesion (Figure 2). Laboratory test results were notable for a white blood cell count of 41.84 cells/µL (reference range, 3.8–10.5 cells/µL), blood glucose level of 586 mg/dL (reference range, 70–99 mg/dL), and hemoglobin A1c of 11.7% (reference range, 4.0%–5.6%). A biopsy specimen from the right pretibial shin was stained with hematoxylin and eosin for dermatopathologic evaluation as well as sent for tissue culture. Tissue and wound cultures grew Staphylococcus aureus and group B Streptococcus with no fungal or acid-fast bacilli growth.
Blood cultures were negative for bacteria. Results of radiographic imaging were negative for osteomyelitis. Biopsy specimens from the right pretibial plaque showed a markedly inflamed, ruptured follicular unit with a dense dermal lympho-neutrophilic infiltrate and overlying pseudoepitheliomatous hyperplasia (Figure 3). Periodic acid–Schiff, Gomori methenamine-silver, acid-fast bacilli, and Giemsa stains were negative for organisms. No granules consistent with a Splendore-Hoeppli phenomenon were observed. These observations were consistent with a diagnosis of BLP.
Blastomycosislike pyoderma is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 Pediatric cases also are uncommon. Blastomycosislike pyoderma most commonly is caused by infection with S aureus or group A streptococci, but several other organisms have been implicated.2 Clinically, BLP is similar to cutaneous botryomycosis, as both are caused by similar organisms.3 However, while BLP is limited to the skin, botryomycosis may involve visceral organs.
Blastomycosislike pyoderma typically presents as verrucous, hyperkeratotic, purulent plaques with raised borders. It most commonly occurs on the face, scalp, axillae, trunk, and distal extremities. Predisposing factors include immunosuppressed states such as poor nutrition, HIV, malignancy, alcoholism, and diabetes mellitus.3,4 Hyperglycemia is thought to suppress helper T cell (TH1)–dependent immunity, which may explain why our patient’s lesions worsened with hyperglycemic episodes.5Histopathology revealed pseudoepitheliomatous hyperplasia with neutrophilic abscesses.1 The distinguishing feature between botryomycosis and BLP is the development of grains known as the Splendore-Hoeppli phenomenon in botryomycosis.6 The grains are eosinophilic and contain the causative infectious agent. The presence of these grains is consistent with botryomycosis but is not pathognomonic, as it also can be found in several bacterial, fungal, and parasitic infections.3,6
The differential diagnosis of BLP includes atypical mycobacterial infection, pyoderma gangrenosum, fungal infection, and tuberculosis verrucosa cutis.7
Although BLP is caused by bacteria, response to systemic antibiotics is variable. Other treatment modalities include dapsone, systemic and intralesional corticosteroids, retinoids, debridement, CO2 laser, and excision.6,8 Lesions typically start out localized, but it is not uncommon for them to spread to distal or vulnerable tissue, such as sites of trauma or inflammation. Our patient was started on oral trimethoprim-sulfamethoxazole and showed improvement, but she worsened with subsequent hyperglycemic episodes when antibiotics were discontinued.
1. Adis¸en E, Tezel F, Gürer MA. Pyoderma vegetans: a case for discussion. Acta Derm Venereol. 2009;89:186-188.
2. Scuderi S, O’Brien B, Robertson I, et al. Heterogeneity of blastomycosis-like pyoderma: a selection of cases from the last 35 years. Australas J Dermatol. 2017;58:139-141.
3. Marschalko, M. Pyoderma vegetans: report on a case and review of data on pyoderma vegetans and cutaneous botryomycosis. Acta Dermatovenerol Alp Pannonica Adriat. 1995;4:55-59.
4. Cerullo L, Zussman J, Young L. An unusual presentation of blastomycosislike pyoderma (pyoderma vegetans) and a review of the literature. Cutis. 2009;84:201-204.
5. Tanaka Y. Immunosuppressive mechanisms in diabetes mellitus [in Japanese]. Nihon Rinsho. 2008;66:2233-2237.
6. Hussein MR. Mucocutaneous Splendore-Hoeppli phenomenon. J Cutan Pathol. 2008;35:979-988.
7. Lee YS, Jung SW, Sim HS, et al. Blastomycosis-like pyoderma with good response to acitretin. Ann Dermatol. 2011;23:365-368.
8. Kobraei KB, Wesson SK. Blastomycosis-like pyoderma: response to systemic retinoid therapy. Int J Dermatol. 2010;49:1336-1338.
To the Editor:
Blastomycosislike pyoderma (BLP), also commonly referred to as pyoderma vegetans, is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 It is characterized by a collection of neutrophilic abscesses with pseudoepitheliomatous hyperplasia that coalesce into crusted plaques.
A 15-year-old adolescent girl with a history of type 1 diabetes mellitus was admitted for diabetic ketoacidosis. The patient presented with bilateral pretibial lesions of 6 years’ duration that developed after swimming in a pool following reported trauma to the site. These pruritic plaques had grown slowly and were occasionally tender. Of note, with episodes of hyperglycemia, the lesions developed purulent drainage.
Upon admission to the hospital and subsequent dermatology consultation, physical examination revealed the right pretibial shin had a 15×5-cm, gray-brown, hyperpigmented, verrucous, tender plaque with purulent drainage and overlying crust (Figure 1). The left pretibial shin had a similar smaller lesion (Figure 2). Laboratory test results were notable for a white blood cell count of 41.84 cells/µL (reference range, 3.8–10.5 cells/µL), blood glucose level of 586 mg/dL (reference range, 70–99 mg/dL), and hemoglobin A1c of 11.7% (reference range, 4.0%–5.6%). A biopsy specimen from the right pretibial shin was stained with hematoxylin and eosin for dermatopathologic evaluation as well as sent for tissue culture. Tissue and wound cultures grew Staphylococcus aureus and group B Streptococcus with no fungal or acid-fast bacilli growth.


Blood cultures were negative for bacteria. Results of radiographic imaging were negative for osteomyelitis. Biopsy specimens from the right pretibial plaque showed a markedly inflamed, ruptured follicular unit with a dense dermal lympho-neutrophilic infiltrate and overlying pseudoepitheliomatous hyperplasia (Figure 3). Periodic acid–Schiff, Gomori methenamine-silver, acid-fast bacilli, and Giemsa stains were negative for organisms. No granules consistent with a Splendore-Hoeppli phenomenon were observed. These observations were consistent with a diagnosis of BLP.

Blastomycosislike pyoderma is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 Pediatric cases also are uncommon. Blastomycosislike pyoderma most commonly is caused by infection with S aureus or group A streptococci, but several other organisms have been implicated.2 Clinically, BLP is similar to cutaneous botryomycosis, as both are caused by similar organisms.3 However, while BLP is limited to the skin, botryomycosis may involve visceral organs.
Blastomycosislike pyoderma typically presents as verrucous, hyperkeratotic, purulent plaques with raised borders. It most commonly occurs on the face, scalp, axillae, trunk, and distal extremities. Predisposing factors include immunosuppressed states such as poor nutrition, HIV, malignancy, alcoholism, and diabetes mellitus.3,4 Hyperglycemia is thought to suppress helper T cell (TH1)–dependent immunity, which may explain why our patient’s lesions worsened with hyperglycemic episodes.5Histopathology revealed pseudoepitheliomatous hyperplasia with neutrophilic abscesses.1 The distinguishing feature between botryomycosis and BLP is the development of grains known as the Splendore-Hoeppli phenomenon in botryomycosis.6 The grains are eosinophilic and contain the causative infectious agent. The presence of these grains is consistent with botryomycosis but is not pathognomonic, as it also can be found in several bacterial, fungal, and parasitic infections.3,6
The differential diagnosis of BLP includes atypical mycobacterial infection, pyoderma gangrenosum, fungal infection, and tuberculosis verrucosa cutis.7
Although BLP is caused by bacteria, response to systemic antibiotics is variable. Other treatment modalities include dapsone, systemic and intralesional corticosteroids, retinoids, debridement, CO2 laser, and excision.6,8 Lesions typically start out localized, but it is not uncommon for them to spread to distal or vulnerable tissue, such as sites of trauma or inflammation. Our patient was started on oral trimethoprim-sulfamethoxazole and showed improvement, but she worsened with subsequent hyperglycemic episodes when antibiotics were discontinued.
To the Editor:
Blastomycosislike pyoderma (BLP), also commonly referred to as pyoderma vegetans, is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 It is characterized by a collection of neutrophilic abscesses with pseudoepitheliomatous hyperplasia that coalesce into crusted plaques.
A 15-year-old adolescent girl with a history of type 1 diabetes mellitus was admitted for diabetic ketoacidosis. The patient presented with bilateral pretibial lesions of 6 years’ duration that developed after swimming in a pool following reported trauma to the site. These pruritic plaques had grown slowly and were occasionally tender. Of note, with episodes of hyperglycemia, the lesions developed purulent drainage.
Upon admission to the hospital and subsequent dermatology consultation, physical examination revealed the right pretibial shin had a 15×5-cm, gray-brown, hyperpigmented, verrucous, tender plaque with purulent drainage and overlying crust (Figure 1). The left pretibial shin had a similar smaller lesion (Figure 2). Laboratory test results were notable for a white blood cell count of 41.84 cells/µL (reference range, 3.8–10.5 cells/µL), blood glucose level of 586 mg/dL (reference range, 70–99 mg/dL), and hemoglobin A1c of 11.7% (reference range, 4.0%–5.6%). A biopsy specimen from the right pretibial shin was stained with hematoxylin and eosin for dermatopathologic evaluation as well as sent for tissue culture. Tissue and wound cultures grew Staphylococcus aureus and group B Streptococcus with no fungal or acid-fast bacilli growth.


Blood cultures were negative for bacteria. Results of radiographic imaging were negative for osteomyelitis. Biopsy specimens from the right pretibial plaque showed a markedly inflamed, ruptured follicular unit with a dense dermal lympho-neutrophilic infiltrate and overlying pseudoepitheliomatous hyperplasia (Figure 3). Periodic acid–Schiff, Gomori methenamine-silver, acid-fast bacilli, and Giemsa stains were negative for organisms. No granules consistent with a Splendore-Hoeppli phenomenon were observed. These observations were consistent with a diagnosis of BLP.

Blastomycosislike pyoderma is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 Pediatric cases also are uncommon. Blastomycosislike pyoderma most commonly is caused by infection with S aureus or group A streptococci, but several other organisms have been implicated.2 Clinically, BLP is similar to cutaneous botryomycosis, as both are caused by similar organisms.3 However, while BLP is limited to the skin, botryomycosis may involve visceral organs.
Blastomycosislike pyoderma typically presents as verrucous, hyperkeratotic, purulent plaques with raised borders. It most commonly occurs on the face, scalp, axillae, trunk, and distal extremities. Predisposing factors include immunosuppressed states such as poor nutrition, HIV, malignancy, alcoholism, and diabetes mellitus.3,4 Hyperglycemia is thought to suppress helper T cell (TH1)–dependent immunity, which may explain why our patient’s lesions worsened with hyperglycemic episodes.5Histopathology revealed pseudoepitheliomatous hyperplasia with neutrophilic abscesses.1 The distinguishing feature between botryomycosis and BLP is the development of grains known as the Splendore-Hoeppli phenomenon in botryomycosis.6 The grains are eosinophilic and contain the causative infectious agent. The presence of these grains is consistent with botryomycosis but is not pathognomonic, as it also can be found in several bacterial, fungal, and parasitic infections.3,6
The differential diagnosis of BLP includes atypical mycobacterial infection, pyoderma gangrenosum, fungal infection, and tuberculosis verrucosa cutis.7
Although BLP is caused by bacteria, response to systemic antibiotics is variable. Other treatment modalities include dapsone, systemic and intralesional corticosteroids, retinoids, debridement, CO2 laser, and excision.6,8 Lesions typically start out localized, but it is not uncommon for them to spread to distal or vulnerable tissue, such as sites of trauma or inflammation. Our patient was started on oral trimethoprim-sulfamethoxazole and showed improvement, but she worsened with subsequent hyperglycemic episodes when antibiotics were discontinued.
1. Adis¸en E, Tezel F, Gürer MA. Pyoderma vegetans: a case for discussion. Acta Derm Venereol. 2009;89:186-188.
2. Scuderi S, O’Brien B, Robertson I, et al. Heterogeneity of blastomycosis-like pyoderma: a selection of cases from the last 35 years. Australas J Dermatol. 2017;58:139-141.
3. Marschalko, M. Pyoderma vegetans: report on a case and review of data on pyoderma vegetans and cutaneous botryomycosis. Acta Dermatovenerol Alp Pannonica Adriat. 1995;4:55-59.
4. Cerullo L, Zussman J, Young L. An unusual presentation of blastomycosislike pyoderma (pyoderma vegetans) and a review of the literature. Cutis. 2009;84:201-204.
5. Tanaka Y. Immunosuppressive mechanisms in diabetes mellitus [in Japanese]. Nihon Rinsho. 2008;66:2233-2237.
6. Hussein MR. Mucocutaneous Splendore-Hoeppli phenomenon. J Cutan Pathol. 2008;35:979-988.
7. Lee YS, Jung SW, Sim HS, et al. Blastomycosis-like pyoderma with good response to acitretin. Ann Dermatol. 2011;23:365-368.
8. Kobraei KB, Wesson SK. Blastomycosis-like pyoderma: response to systemic retinoid therapy. Int J Dermatol. 2010;49:1336-1338.
1. Adis¸en E, Tezel F, Gürer MA. Pyoderma vegetans: a case for discussion. Acta Derm Venereol. 2009;89:186-188.
2. Scuderi S, O’Brien B, Robertson I, et al. Heterogeneity of blastomycosis-like pyoderma: a selection of cases from the last 35 years. Australas J Dermatol. 2017;58:139-141.
3. Marschalko, M. Pyoderma vegetans: report on a case and review of data on pyoderma vegetans and cutaneous botryomycosis. Acta Dermatovenerol Alp Pannonica Adriat. 1995;4:55-59.
4. Cerullo L, Zussman J, Young L. An unusual presentation of blastomycosislike pyoderma (pyoderma vegetans) and a review of the literature. Cutis. 2009;84:201-204.
5. Tanaka Y. Immunosuppressive mechanisms in diabetes mellitus [in Japanese]. Nihon Rinsho. 2008;66:2233-2237.
6. Hussein MR. Mucocutaneous Splendore-Hoeppli phenomenon. J Cutan Pathol. 2008;35:979-988.
7. Lee YS, Jung SW, Sim HS, et al. Blastomycosis-like pyoderma with good response to acitretin. Ann Dermatol. 2011;23:365-368.
8. Kobraei KB, Wesson SK. Blastomycosis-like pyoderma: response to systemic retinoid therapy. Int J Dermatol. 2010;49:1336-1338.
Practice Points
- Blastomycosislike pyoderma is a rare condition secondary to bacterial infection, but as the name suggests, it also can resemble cutaneous blastomycosis.
- Blastomycosislike pyoderma most commonly occurs in immunocompromised patients.
- The most common histologic findings include suppurative and neutrophilic inflammation with pseudoepitheliomatous hyperplasia.
COVID cases spike as questions remain about Omicron’s threat
The best way to stay protected is by getting vaccinated and boosted, they said.
“For the unvaccinated, you’re looking at a winter of severe illness and death – for yourselves, families, and the hospitals who may soon overwhelm,” White House COVID-19 Response Coordinator Jeff Zients said at a news briefing Dec. 17. “We need the American people to do their part.”
The Omicron variant has been detected in at least 39 states and 75 countries, according to CDC director Rochelle Walensky, MD.
The strain is more transmissible than the already highly infectious Delta variant, and although there was early evidence that it caused more mild disease, she said that is likely because many of those infected have been vaccinated and boosted.
“Although Delta continues to circulate widely in the United States, Omicron is increasing rapidly and we expect it to become the dominant strain in the United States, as it has in other countries, in the coming weeks,” Dr. Walensky said.
The United States is averaging close to 1,300 deaths from COVID-19 each day. New cases, deaths, and hospitalizations are higher now than in the previous winter – before vaccines were so widely available. The New York Times reported on Dec. 17 that new infections in Connecticut and Maine have grown 150% in the past 2 weeks, and Ohio and Indiana are seeing hospitalization rates nearing the worst of 2020-2021’s winter surge.
Dueling reports released recently gave cause for relief and concern about Omicron.
A study from South Africa released on Dec. 14 shows lower hospitalizations during the first 3 weeks of the Omicron wave than during earlier waves from other variants. That’s the good news.
The concerning news is out of the United Kingdom, where Imperial College London reported Dec. 17 that the risk of reinfection with COVID-19 from Omicron is more than 5 times as high and that cases of Omicron-based COVID-19 are doubling every 2 days.
What’s more, the study “finds no evidence of Omicron having lower severity than Delta, judged by either the proportion of people testing positive who report symptoms, or by the proportion of cases seeking hospital care after infection. However, hospitalization data remains very limited at this time,” the researchers said.
“We have no evidence that the virus itself is more mild,” Eric Topol, MD, executive vice president of Scripps Research and editor-in-chief of Medscape, told PBS NewsHour. “Until we have that, we have to assume that people who don’t have any protection are highly vulnerable to getting very ill.”
The White House COVID-19 team continues to urge parents and guardians to get their children vaccinated, especially in anticipation of a post-holiday spike. Dr. Walensky said the CDC’s vaccine advisory board met on Dec. 16 to continue the safety discussion about COVID-19 vaccinations in children.
So far, 20 million children under 17 and 5 million under 11 have received their shots.
“Looking specifically at vaccine safety data from over 50,000 children 5-11 years old, we found no evidence of serious safety concerns,” Dr. Walensky said.
Top infectious disease expert Anthony S. Fauci, MD, highlighted the importance of getting vaccinated and boosted to avoid serious disease from Delta and Omicron.
“We’re in a situation where we are now facing a very important Delta surge and we are looking over our shoulder at an oncoming Omicron surge,” he said. “The optimum protection is fully vaccinated plus a boost.”
A version of this article first appeared on WebMD.com.
The best way to stay protected is by getting vaccinated and boosted, they said.
“For the unvaccinated, you’re looking at a winter of severe illness and death – for yourselves, families, and the hospitals who may soon overwhelm,” White House COVID-19 Response Coordinator Jeff Zients said at a news briefing Dec. 17. “We need the American people to do their part.”
The Omicron variant has been detected in at least 39 states and 75 countries, according to CDC director Rochelle Walensky, MD.
The strain is more transmissible than the already highly infectious Delta variant, and although there was early evidence that it caused more mild disease, she said that is likely because many of those infected have been vaccinated and boosted.
“Although Delta continues to circulate widely in the United States, Omicron is increasing rapidly and we expect it to become the dominant strain in the United States, as it has in other countries, in the coming weeks,” Dr. Walensky said.
The United States is averaging close to 1,300 deaths from COVID-19 each day. New cases, deaths, and hospitalizations are higher now than in the previous winter – before vaccines were so widely available. The New York Times reported on Dec. 17 that new infections in Connecticut and Maine have grown 150% in the past 2 weeks, and Ohio and Indiana are seeing hospitalization rates nearing the worst of 2020-2021’s winter surge.
Dueling reports released recently gave cause for relief and concern about Omicron.
A study from South Africa released on Dec. 14 shows lower hospitalizations during the first 3 weeks of the Omicron wave than during earlier waves from other variants. That’s the good news.
The concerning news is out of the United Kingdom, where Imperial College London reported Dec. 17 that the risk of reinfection with COVID-19 from Omicron is more than 5 times as high and that cases of Omicron-based COVID-19 are doubling every 2 days.
What’s more, the study “finds no evidence of Omicron having lower severity than Delta, judged by either the proportion of people testing positive who report symptoms, or by the proportion of cases seeking hospital care after infection. However, hospitalization data remains very limited at this time,” the researchers said.
“We have no evidence that the virus itself is more mild,” Eric Topol, MD, executive vice president of Scripps Research and editor-in-chief of Medscape, told PBS NewsHour. “Until we have that, we have to assume that people who don’t have any protection are highly vulnerable to getting very ill.”
The White House COVID-19 team continues to urge parents and guardians to get their children vaccinated, especially in anticipation of a post-holiday spike. Dr. Walensky said the CDC’s vaccine advisory board met on Dec. 16 to continue the safety discussion about COVID-19 vaccinations in children.
So far, 20 million children under 17 and 5 million under 11 have received their shots.
“Looking specifically at vaccine safety data from over 50,000 children 5-11 years old, we found no evidence of serious safety concerns,” Dr. Walensky said.
Top infectious disease expert Anthony S. Fauci, MD, highlighted the importance of getting vaccinated and boosted to avoid serious disease from Delta and Omicron.
“We’re in a situation where we are now facing a very important Delta surge and we are looking over our shoulder at an oncoming Omicron surge,” he said. “The optimum protection is fully vaccinated plus a boost.”
A version of this article first appeared on WebMD.com.
The best way to stay protected is by getting vaccinated and boosted, they said.
“For the unvaccinated, you’re looking at a winter of severe illness and death – for yourselves, families, and the hospitals who may soon overwhelm,” White House COVID-19 Response Coordinator Jeff Zients said at a news briefing Dec. 17. “We need the American people to do their part.”
The Omicron variant has been detected in at least 39 states and 75 countries, according to CDC director Rochelle Walensky, MD.
The strain is more transmissible than the already highly infectious Delta variant, and although there was early evidence that it caused more mild disease, she said that is likely because many of those infected have been vaccinated and boosted.
“Although Delta continues to circulate widely in the United States, Omicron is increasing rapidly and we expect it to become the dominant strain in the United States, as it has in other countries, in the coming weeks,” Dr. Walensky said.
The United States is averaging close to 1,300 deaths from COVID-19 each day. New cases, deaths, and hospitalizations are higher now than in the previous winter – before vaccines were so widely available. The New York Times reported on Dec. 17 that new infections in Connecticut and Maine have grown 150% in the past 2 weeks, and Ohio and Indiana are seeing hospitalization rates nearing the worst of 2020-2021’s winter surge.
Dueling reports released recently gave cause for relief and concern about Omicron.
A study from South Africa released on Dec. 14 shows lower hospitalizations during the first 3 weeks of the Omicron wave than during earlier waves from other variants. That’s the good news.
The concerning news is out of the United Kingdom, where Imperial College London reported Dec. 17 that the risk of reinfection with COVID-19 from Omicron is more than 5 times as high and that cases of Omicron-based COVID-19 are doubling every 2 days.
What’s more, the study “finds no evidence of Omicron having lower severity than Delta, judged by either the proportion of people testing positive who report symptoms, or by the proportion of cases seeking hospital care after infection. However, hospitalization data remains very limited at this time,” the researchers said.
“We have no evidence that the virus itself is more mild,” Eric Topol, MD, executive vice president of Scripps Research and editor-in-chief of Medscape, told PBS NewsHour. “Until we have that, we have to assume that people who don’t have any protection are highly vulnerable to getting very ill.”
The White House COVID-19 team continues to urge parents and guardians to get their children vaccinated, especially in anticipation of a post-holiday spike. Dr. Walensky said the CDC’s vaccine advisory board met on Dec. 16 to continue the safety discussion about COVID-19 vaccinations in children.
So far, 20 million children under 17 and 5 million under 11 have received their shots.
“Looking specifically at vaccine safety data from over 50,000 children 5-11 years old, we found no evidence of serious safety concerns,” Dr. Walensky said.
Top infectious disease expert Anthony S. Fauci, MD, highlighted the importance of getting vaccinated and boosted to avoid serious disease from Delta and Omicron.
“We’re in a situation where we are now facing a very important Delta surge and we are looking over our shoulder at an oncoming Omicron surge,” he said. “The optimum protection is fully vaccinated plus a boost.”
A version of this article first appeared on WebMD.com.