User login
Methacrylate Polymer Powder Dressing for a Lower Leg Surgical Defect
To the Editor:
Surgical wounds on the lower leg are challenging to manage because venous stasis, bacterial colonization, and high tension may contribute to protracted healing. Advances in technology led to the development of novel, polymer-based wound-healing modalities that hold promise for the management of these wounds.
A 75-year-old man presented with a well-differentiated squamous cell carcinoma with a 3-mm depth of invasion on the left pretibial region. His comorbidities were notable for hypertension, hypercholesterolemia, varicose veins, myocardial infarction, peripheral vascular disease, and a 32 pack-year cigarette smoking history. Current medications included clopidogrel bisulfate and warfarin sodium to manage a recently placed coronary artery stent.
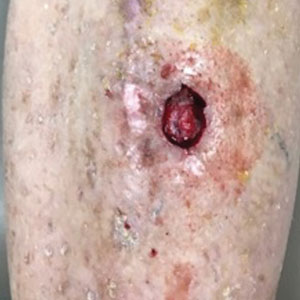
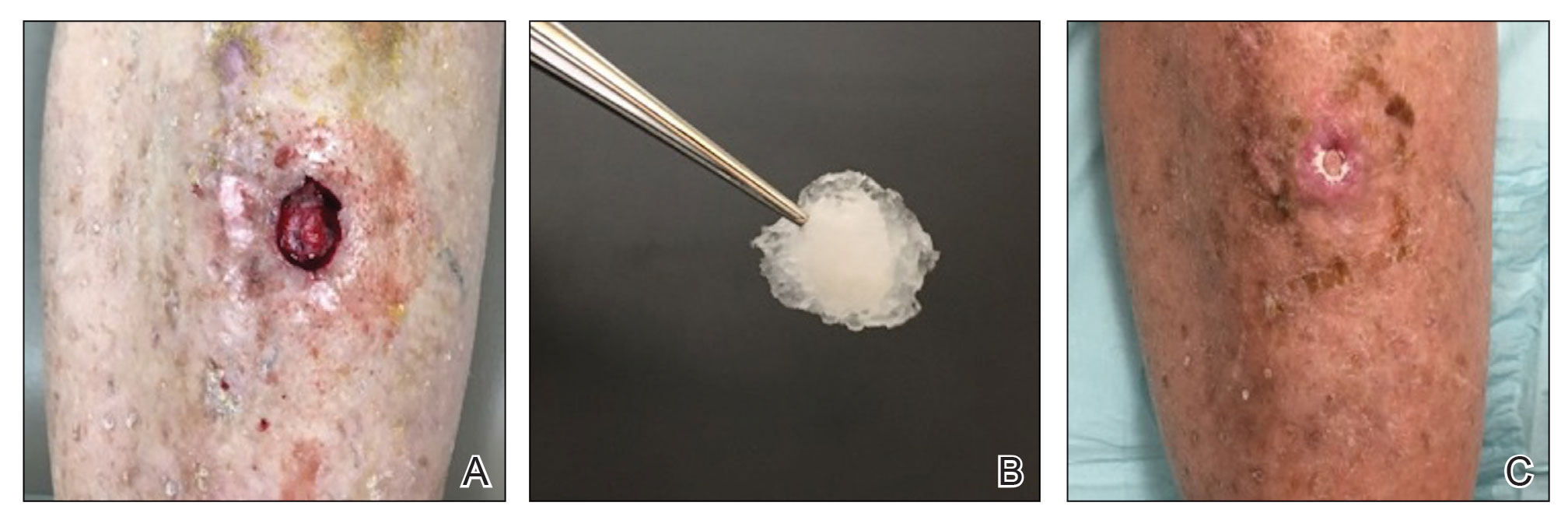
The tumor was cleared after 2 stages of Mohs micrographic surgery with excision down to tibialis anterior fascia (Figure 1A). The resultant defect measured 43×33 mm in area and 9 mm in depth (wound size, 12,771 mm3). Reconstructive options were discussed, including random-pattern flap repair and skin graft. Given the patient’s risk of bleeding, the decision was made to forego a flap repair. Additionally, the patient was a heavy smoker and could not comply with the wound care and elevation and ambulation restrictions required for optimal skin graft care. Therefore, a decision was made to proceed with secondary intention healing using a methacrylate polymer powder dressing.
After achieving hemostasis, a novel 10-mg sterile, biologically inert methacrylate polymer powder dressing was poured over the wound in a uniform layer to fill and seal the entire wound surface (Figure 1B). Sterile normal saline 0.1 mL was sprayed onto the powder to activate particle aggregation. No secondary dressing was used, and the patient was permitted to get the dressing wet after 48 hours.
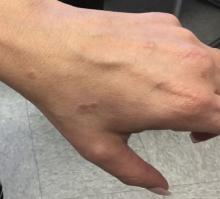
The dressing was changed in a similar fashion 4 weeks after application, following gentle debridement with gauze and normal saline. Eight weeks after surgery, the wound exhibited healthy granulation tissue and measured 5×6 mm in area and 2 mm in depth (wound size, 60 mm3), which represented a 99.5% reduction in wound size (Figure 1C). The dressing was not painful, and there were no reported adverse effects. The patient continued to smoke and ambulate fully throughout this period. No antibiotics were used.
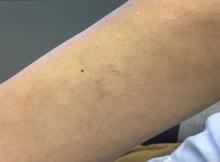
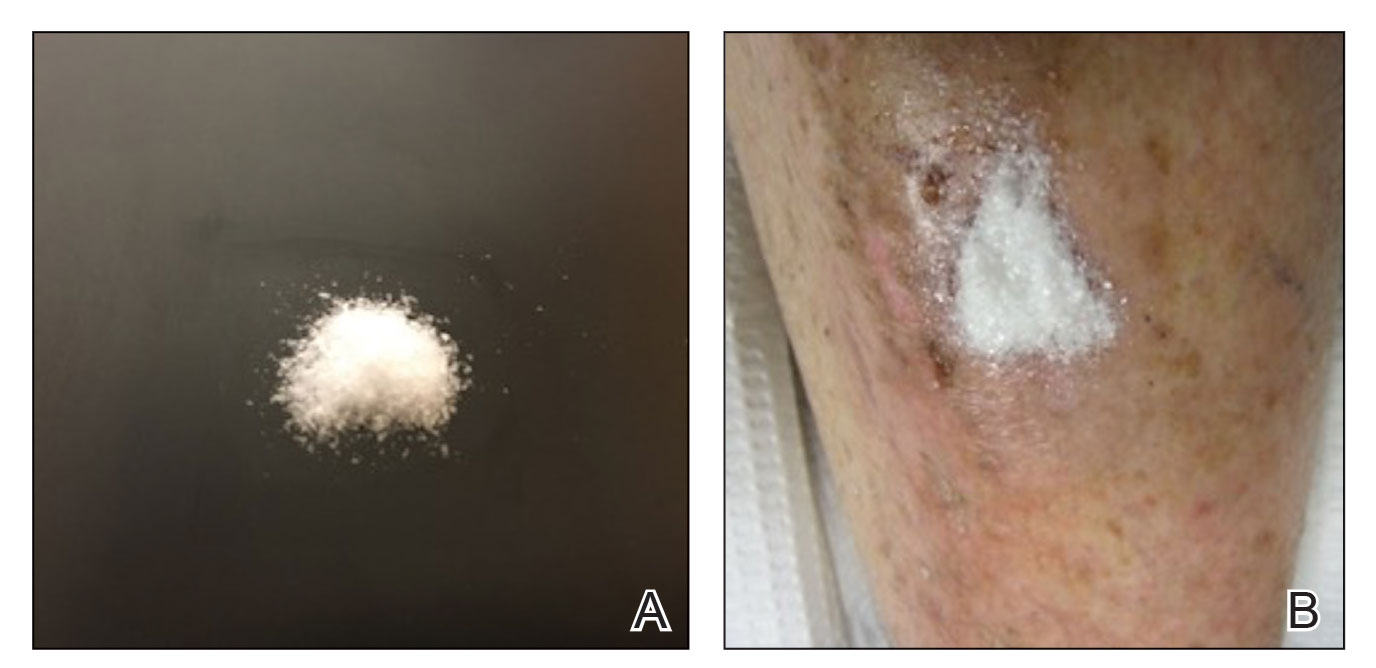
Methacrylate polymer powder dressings are a novel and sophisticated dressing modality with great promise for the management of surgical wounds on the lower limb. The dressing is a sterile powder consisting of 84.8% poly-2-hydroxyethylmethacrylate, 14.9% poly-2-hydroxypropylmethacrylate, and 0.3% sodium deoxycholate. These hydrophilic polymers have a covalent methacrylate backbone with a hydroxyl aliphatic side chain. When saline or wound exudate contacts the powder, the spheres hydrate and nonreversibly aggregate to form a moist, flexible dressing that conforms to the topography of the wound and seals it (Figure 2).1
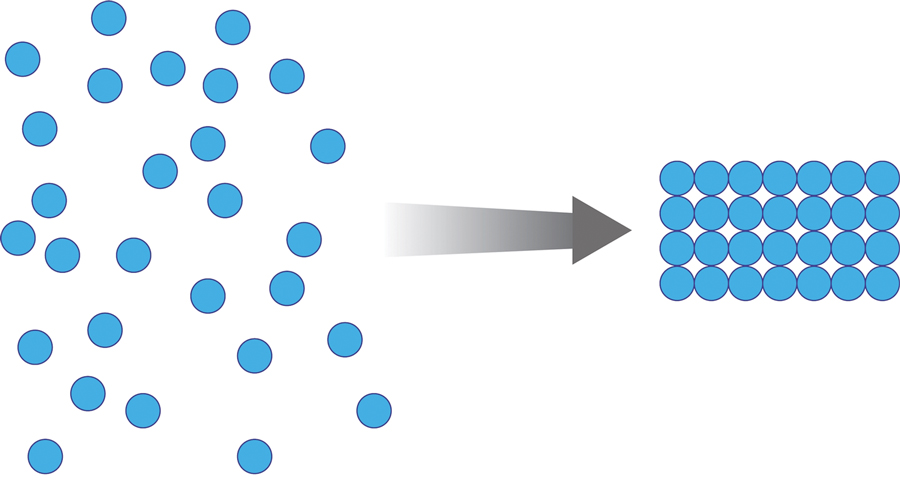
Once the spheres have aggregated, they are designed to orient in a honeycomb formation with 4- to 10-nm openings that serve as capillary channels (Figure 3). This porous architecture of the polymer is essential for adequate moisture management. It allows for vapor transpiration at a rate of 12 L/m2 per day, which ensures the capillary flow from the moist wound surface is evenly distributed through the dressing, contributing to its 68% water content. Notably, this approximately three-fifths water composition is similar to the water makeup of human skin. Optimized moisture management is theorized to enhance epithelial migration, stimulate angiogenesis, retain growth factors, promote autolytic debridement, and maintain ideal voltage and oxygen gradients for wound healing. The risk for infection is not increased by the existence of these pores, as their small size does not allow for bacterial migration.1
This case demonstrates the effectiveness of using a methacrylate polymer powder dressing to promote timely wound healing in a poorly vascularized lower leg surgical wound. The low maintenance, user-friendly dressing was changed at monthly intervals, which spared the patient the inconvenience and pain associated with the repeated application of more conventional primary and secondary dressings. The dressing was well tolerated and resulted in a 99.5% reduction in wound size. Further studies are needed to investigate the utility of this promising technology.
1. Fitzgerald RH, Bharara M, Mills JL, et al. Use of a nanoflex powder dressing for wound management following debridement for necrotising fasciitis in the diabetic foot. Int Wound J. 2009;6:133-139.
To the Editor:
Surgical wounds on the lower leg are challenging to manage because venous stasis, bacterial colonization, and high tension may contribute to protracted healing. Advances in technology led to the development of novel, polymer-based wound-healing modalities that hold promise for the management of these wounds.
A 75-year-old man presented with a well-differentiated squamous cell carcinoma with a 3-mm depth of invasion on the left pretibial region. His comorbidities were notable for hypertension, hypercholesterolemia, varicose veins, myocardial infarction, peripheral vascular disease, and a 32 pack-year cigarette smoking history. Current medications included clopidogrel bisulfate and warfarin sodium to manage a recently placed coronary artery stent.
The tumor was cleared after 2 stages of Mohs micrographic surgery with excision down to tibialis anterior fascia (Figure 1A). The resultant defect measured 43×33 mm in area and 9 mm in depth (wound size, 12,771 mm3). Reconstructive options were discussed, including random-pattern flap repair and skin graft. Given the patient’s risk of bleeding, the decision was made to forego a flap repair. Additionally, the patient was a heavy smoker and could not comply with the wound care and elevation and ambulation restrictions required for optimal skin graft care. Therefore, a decision was made to proceed with secondary intention healing using a methacrylate polymer powder dressing.
After achieving hemostasis, a novel 10-mg sterile, biologically inert methacrylate polymer powder dressing was poured over the wound in a uniform layer to fill and seal the entire wound surface (Figure 1B). Sterile normal saline 0.1 mL was sprayed onto the powder to activate particle aggregation. No secondary dressing was used, and the patient was permitted to get the dressing wet after 48 hours.
The dressing was changed in a similar fashion 4 weeks after application, following gentle debridement with gauze and normal saline. Eight weeks after surgery, the wound exhibited healthy granulation tissue and measured 5×6 mm in area and 2 mm in depth (wound size, 60 mm3), which represented a 99.5% reduction in wound size (Figure 1C). The dressing was not painful, and there were no reported adverse effects. The patient continued to smoke and ambulate fully throughout this period. No antibiotics were used.

Methacrylate polymer powder dressings are a novel and sophisticated dressing modality with great promise for the management of surgical wounds on the lower limb. The dressing is a sterile powder consisting of 84.8% poly-2-hydroxyethylmethacrylate, 14.9% poly-2-hydroxypropylmethacrylate, and 0.3% sodium deoxycholate. These hydrophilic polymers have a covalent methacrylate backbone with a hydroxyl aliphatic side chain. When saline or wound exudate contacts the powder, the spheres hydrate and nonreversibly aggregate to form a moist, flexible dressing that conforms to the topography of the wound and seals it (Figure 2).1

Once the spheres have aggregated, they are designed to orient in a honeycomb formation with 4- to 10-nm openings that serve as capillary channels (Figure 3). This porous architecture of the polymer is essential for adequate moisture management. It allows for vapor transpiration at a rate of 12 L/m2 per day, which ensures the capillary flow from the moist wound surface is evenly distributed through the dressing, contributing to its 68% water content. Notably, this approximately three-fifths water composition is similar to the water makeup of human skin. Optimized moisture management is theorized to enhance epithelial migration, stimulate angiogenesis, retain growth factors, promote autolytic debridement, and maintain ideal voltage and oxygen gradients for wound healing. The risk for infection is not increased by the existence of these pores, as their small size does not allow for bacterial migration.1

This case demonstrates the effectiveness of using a methacrylate polymer powder dressing to promote timely wound healing in a poorly vascularized lower leg surgical wound. The low maintenance, user-friendly dressing was changed at monthly intervals, which spared the patient the inconvenience and pain associated with the repeated application of more conventional primary and secondary dressings. The dressing was well tolerated and resulted in a 99.5% reduction in wound size. Further studies are needed to investigate the utility of this promising technology.
To the Editor:
Surgical wounds on the lower leg are challenging to manage because venous stasis, bacterial colonization, and high tension may contribute to protracted healing. Advances in technology led to the development of novel, polymer-based wound-healing modalities that hold promise for the management of these wounds.
A 75-year-old man presented with a well-differentiated squamous cell carcinoma with a 3-mm depth of invasion on the left pretibial region. His comorbidities were notable for hypertension, hypercholesterolemia, varicose veins, myocardial infarction, peripheral vascular disease, and a 32 pack-year cigarette smoking history. Current medications included clopidogrel bisulfate and warfarin sodium to manage a recently placed coronary artery stent.
The tumor was cleared after 2 stages of Mohs micrographic surgery with excision down to tibialis anterior fascia (Figure 1A). The resultant defect measured 43×33 mm in area and 9 mm in depth (wound size, 12,771 mm3). Reconstructive options were discussed, including random-pattern flap repair and skin graft. Given the patient’s risk of bleeding, the decision was made to forego a flap repair. Additionally, the patient was a heavy smoker and could not comply with the wound care and elevation and ambulation restrictions required for optimal skin graft care. Therefore, a decision was made to proceed with secondary intention healing using a methacrylate polymer powder dressing.
After achieving hemostasis, a novel 10-mg sterile, biologically inert methacrylate polymer powder dressing was poured over the wound in a uniform layer to fill and seal the entire wound surface (Figure 1B). Sterile normal saline 0.1 mL was sprayed onto the powder to activate particle aggregation. No secondary dressing was used, and the patient was permitted to get the dressing wet after 48 hours.
The dressing was changed in a similar fashion 4 weeks after application, following gentle debridement with gauze and normal saline. Eight weeks after surgery, the wound exhibited healthy granulation tissue and measured 5×6 mm in area and 2 mm in depth (wound size, 60 mm3), which represented a 99.5% reduction in wound size (Figure 1C). The dressing was not painful, and there were no reported adverse effects. The patient continued to smoke and ambulate fully throughout this period. No antibiotics were used.

Methacrylate polymer powder dressings are a novel and sophisticated dressing modality with great promise for the management of surgical wounds on the lower limb. The dressing is a sterile powder consisting of 84.8% poly-2-hydroxyethylmethacrylate, 14.9% poly-2-hydroxypropylmethacrylate, and 0.3% sodium deoxycholate. These hydrophilic polymers have a covalent methacrylate backbone with a hydroxyl aliphatic side chain. When saline or wound exudate contacts the powder, the spheres hydrate and nonreversibly aggregate to form a moist, flexible dressing that conforms to the topography of the wound and seals it (Figure 2).1

Once the spheres have aggregated, they are designed to orient in a honeycomb formation with 4- to 10-nm openings that serve as capillary channels (Figure 3). This porous architecture of the polymer is essential for adequate moisture management. It allows for vapor transpiration at a rate of 12 L/m2 per day, which ensures the capillary flow from the moist wound surface is evenly distributed through the dressing, contributing to its 68% water content. Notably, this approximately three-fifths water composition is similar to the water makeup of human skin. Optimized moisture management is theorized to enhance epithelial migration, stimulate angiogenesis, retain growth factors, promote autolytic debridement, and maintain ideal voltage and oxygen gradients for wound healing. The risk for infection is not increased by the existence of these pores, as their small size does not allow for bacterial migration.1

This case demonstrates the effectiveness of using a methacrylate polymer powder dressing to promote timely wound healing in a poorly vascularized lower leg surgical wound. The low maintenance, user-friendly dressing was changed at monthly intervals, which spared the patient the inconvenience and pain associated with the repeated application of more conventional primary and secondary dressings. The dressing was well tolerated and resulted in a 99.5% reduction in wound size. Further studies are needed to investigate the utility of this promising technology.
1. Fitzgerald RH, Bharara M, Mills JL, et al. Use of a nanoflex powder dressing for wound management following debridement for necrotising fasciitis in the diabetic foot. Int Wound J. 2009;6:133-139.
1. Fitzgerald RH, Bharara M, Mills JL, et al. Use of a nanoflex powder dressing for wound management following debridement for necrotising fasciitis in the diabetic foot. Int Wound J. 2009;6:133-139.
PRACTICE POINTS
- Lower leg surgical wounds are difficult to manage, as venous stasis, bacterial colonization, and high tension may contribute to protracted healing.
- A methacrylate polymer powder dressing is user friendly and facilitates granulation and reduction in size of difficult lower leg wounds.
After PCI, 1-month beats 12-month DAPT in high-risk patients
Replacing dual-antiplatelet therapy (DAPT) with clopidogrel alone 1 month after percutaneous intervention (PCI) offers a lower risk of bleeding with comparable protection against cardiovascular events, according to two subgroup analyses of the Japanese STOPDAPT-2 and STOPDAPT-2 ACS trials.
The objective of these two analyses was to evaluate whether there was a benefit-to-risk ratio advantage for those who entered the study with high bleeding risk or who had undergone a complex PCI. Overall, bleeding risk was reduced without a major increase in cardiovascular events regardless of subgroup, according to results published by a multicenter group of Japanese investigators.
In this substudy, like the previously published studies from which the data were drawn, the primary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, stroke, and Thrombolysis In Myocardial Infarction bleeding (major or minor).
The proportion of patients in the 1-month and 12-month DAPT groups reaching this composite endpoint at 1 year was not significantly different among patients stratified by baseline bleeding risk or by PCI complexity, according to a multicenter group of authors led by Takeshi Kimura, MD, department of cardiovascular medicine, Kyoto University.
Shortened DAPT is focus of multiple trials
The new analysis, published in JACC Asia, is a follow-up to the 2019 STOPDAPT-2 trial, published in JAMA, and the 2022 STOPDAPT-2 ACS trial, published in JAMA Cardiology. The first tested 1- versus 12-month DAPT in PCI patients receiving a drug-eluting stent. The second study compared the same strategies in patients undergoing PCI to treat an acute coronary syndrome (ACS).
Both studies were conducted in Japan. DAPT consisted of the P2Y12 receptor inhibitor clopidogrel plus aspirin. The experimental arm received this regimen for 1 month followed by clopidogrel monotherapy. The control arm remained on DAPT for 12 months.
The study is potentially important because it addresses the challenge of finding “the sweet spot of antiplatelet therapy in East Asian patients,” according to the coauthors of an accompanying editorial in the same issue of JACC Asia.
Previous data suggest East Asians have a higher risk of bleeding but lower anti-ischemic benefits from DAPT therapy, explained the coauthors, Antonio Greco, MD and Davide Capodanno, MD, PhD, both from the University of Catania (Italy). They praised the effort to explore this question.
In the STOPDAPT-2 trial, the shortened DAPT regimen was associated with a significantly lower rate of a composite endpoint of cardiovascular and bleeding events than standard DAPT, meeting criteria for superiority as well as noninferiority. In the STOPDAPT-2 ACS trial, shortened DAPT failed to achieve noninferiority to standard DAPT because of an increase in cardiovascular events despite a reduction in bleeding events.
Neither of these studies specifically compared shortened to standard DAPT in patients with high bleeding risk or in patients who underwent complex PCI, which are among the most common patient groups in which to consider a modified DAPT regimen. To do this, two new substudies were performed with the combined data from 5,997 patients in the two STOPDAPT-2 trials.
Two candidate groups for shortened DAPT evaluated
In the first substudy, the 1,893 patients who met criteria for high bleeding risk were compared with the 4,104 who did not. In those with a high risk of bleeding, the proportion reaching a primary endpoint at 1 year was lower, but not significantly different, for those on 1-month versus standard DAPT (5.01% vs. 5.14%). This was also true in those without an elevated bleeding risk (1.90% vs. 2.02%).
In the second substudy, 999 patients who had a complex PCI, defined by such characteristics as implantation of at least three stents or chronic total occlusion in the target lesions, were compared with the 4,998 who did not. Again, the primary endpoint was lower in both those who had a complex PCI (3.15% vs. 4.07%) and those who did not (2.78% vs. 2.82%).
Not surprisingly, patients with a high bleeding risk benefited from a substantially lower risk of bleeding events on the 1-month DAPT regimen (0.66% vs. 2.27%). The cost was a higher risk of cardiovascular events (4.35% vs. 3.52%), but this difference did not reach significance. Those without an elevated bleeding risk also had a lower risk of bleeding events (0.43% vs. 0.85%) but a higher risk of cardiovascular events (1.56% vs. 1.22%). Again, differences were nonsignificant. In the substudy evaluating DAPT duration in relation to complex PCI, the rate of cardiovascular events at 1 year in those treated with short versus 12-month DAPT was nearly identical (2.53% vs. 2.52%). In the non–complex PCI patients, event rates were nonsignificantly greater on the shortened DAPT regimen (2.38% vs. 1.86%), but the bleeding rate was lower on shortened DAPT whether PCI had been complex (0.63% vs. 1.75%) or not (0.48% vs. 1.22%).
In the absence of any major signal that complex PCI benefited from longer duration DAPT, “complex PCI might not be an appropriate determinant for DAPT durations,” according to Dr. Kimura and coinvestigators.
Study data might not be generalizable
Dr. Greco and Dr. Capodanno pointed out that there are differences between patients and PCI practices in Japan relative to other areas of the world, limiting the generalizability of these findings even if the question is relevant.
“This is an approach that might be suggested for patients at high bleeding risk who have the characteristics of the patients enrolled in the STOPDAPT-2 trials,” Dr. Capodanno said in an interview. In his own PCI practice treating ACS patients, “I would not feel safe enough with clopidogrel monotherapy after only 1 month.”
He considers the ACS population to have a particularly “delicate bleeding-ischemia trade-off,” which is why he thinks this question is relevant and needs to be explored further in additional populations. However, he might design trials differently in his own practice setting. For example, he would at the very least be interested in testing a more potent P2Y12 inhibitor such as ticagrelor when considering a single antiplatelet agent after a limited course of DAPT.
One message from this study is that “bleeding risk trumps PCI complexity,” according to Deepak L. Bhatt, MD, who recently assumed the position of director of Mount Sinai Heart in New York. He liked the approach the investigators took to address a complex and relevant clinical issue, but he also expressed reservations about the clinical applicability of this subgroup analysis.
“We really need more data before uniformly shortening DAPT duration in all patients,” Dr. Bhatt said in an interview. He considers this a hot clinical issue that is likely to generate more trials. He hopes these will provide more definitive evidence of when and how DAPT duration can be reduced. Overall, he anticipates progress toward tailoring therapy in specific populations in order to achieve the best risk-to-benefit balance.
Dr. Kimura has financial relationships with Boston Scientific, Daiichi Sankyo, Sanofi, Terumo, and Abbott Medical Japan, which provided funding for the STOPDAPT-2 and STOPDAPT-2 ACS trials. Dr. Capodanno reported financial relationships with Amgen, Arena, Chiesi, Daiichi Sakyo, Sanofi Aventis, and Terumo. Dr. Bhatt reported financial relationships with more than 20 pharmaceutical companies, including Abbott Medical.
Replacing dual-antiplatelet therapy (DAPT) with clopidogrel alone 1 month after percutaneous intervention (PCI) offers a lower risk of bleeding with comparable protection against cardiovascular events, according to two subgroup analyses of the Japanese STOPDAPT-2 and STOPDAPT-2 ACS trials.
The objective of these two analyses was to evaluate whether there was a benefit-to-risk ratio advantage for those who entered the study with high bleeding risk or who had undergone a complex PCI. Overall, bleeding risk was reduced without a major increase in cardiovascular events regardless of subgroup, according to results published by a multicenter group of Japanese investigators.
In this substudy, like the previously published studies from which the data were drawn, the primary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, stroke, and Thrombolysis In Myocardial Infarction bleeding (major or minor).
The proportion of patients in the 1-month and 12-month DAPT groups reaching this composite endpoint at 1 year was not significantly different among patients stratified by baseline bleeding risk or by PCI complexity, according to a multicenter group of authors led by Takeshi Kimura, MD, department of cardiovascular medicine, Kyoto University.
Shortened DAPT is focus of multiple trials
The new analysis, published in JACC Asia, is a follow-up to the 2019 STOPDAPT-2 trial, published in JAMA, and the 2022 STOPDAPT-2 ACS trial, published in JAMA Cardiology. The first tested 1- versus 12-month DAPT in PCI patients receiving a drug-eluting stent. The second study compared the same strategies in patients undergoing PCI to treat an acute coronary syndrome (ACS).
Both studies were conducted in Japan. DAPT consisted of the P2Y12 receptor inhibitor clopidogrel plus aspirin. The experimental arm received this regimen for 1 month followed by clopidogrel monotherapy. The control arm remained on DAPT for 12 months.
The study is potentially important because it addresses the challenge of finding “the sweet spot of antiplatelet therapy in East Asian patients,” according to the coauthors of an accompanying editorial in the same issue of JACC Asia.
Previous data suggest East Asians have a higher risk of bleeding but lower anti-ischemic benefits from DAPT therapy, explained the coauthors, Antonio Greco, MD and Davide Capodanno, MD, PhD, both from the University of Catania (Italy). They praised the effort to explore this question.
In the STOPDAPT-2 trial, the shortened DAPT regimen was associated with a significantly lower rate of a composite endpoint of cardiovascular and bleeding events than standard DAPT, meeting criteria for superiority as well as noninferiority. In the STOPDAPT-2 ACS trial, shortened DAPT failed to achieve noninferiority to standard DAPT because of an increase in cardiovascular events despite a reduction in bleeding events.
Neither of these studies specifically compared shortened to standard DAPT in patients with high bleeding risk or in patients who underwent complex PCI, which are among the most common patient groups in which to consider a modified DAPT regimen. To do this, two new substudies were performed with the combined data from 5,997 patients in the two STOPDAPT-2 trials.
Two candidate groups for shortened DAPT evaluated
In the first substudy, the 1,893 patients who met criteria for high bleeding risk were compared with the 4,104 who did not. In those with a high risk of bleeding, the proportion reaching a primary endpoint at 1 year was lower, but not significantly different, for those on 1-month versus standard DAPT (5.01% vs. 5.14%). This was also true in those without an elevated bleeding risk (1.90% vs. 2.02%).
In the second substudy, 999 patients who had a complex PCI, defined by such characteristics as implantation of at least three stents or chronic total occlusion in the target lesions, were compared with the 4,998 who did not. Again, the primary endpoint was lower in both those who had a complex PCI (3.15% vs. 4.07%) and those who did not (2.78% vs. 2.82%).
Not surprisingly, patients with a high bleeding risk benefited from a substantially lower risk of bleeding events on the 1-month DAPT regimen (0.66% vs. 2.27%). The cost was a higher risk of cardiovascular events (4.35% vs. 3.52%), but this difference did not reach significance. Those without an elevated bleeding risk also had a lower risk of bleeding events (0.43% vs. 0.85%) but a higher risk of cardiovascular events (1.56% vs. 1.22%). Again, differences were nonsignificant. In the substudy evaluating DAPT duration in relation to complex PCI, the rate of cardiovascular events at 1 year in those treated with short versus 12-month DAPT was nearly identical (2.53% vs. 2.52%). In the non–complex PCI patients, event rates were nonsignificantly greater on the shortened DAPT regimen (2.38% vs. 1.86%), but the bleeding rate was lower on shortened DAPT whether PCI had been complex (0.63% vs. 1.75%) or not (0.48% vs. 1.22%).
In the absence of any major signal that complex PCI benefited from longer duration DAPT, “complex PCI might not be an appropriate determinant for DAPT durations,” according to Dr. Kimura and coinvestigators.
Study data might not be generalizable
Dr. Greco and Dr. Capodanno pointed out that there are differences between patients and PCI practices in Japan relative to other areas of the world, limiting the generalizability of these findings even if the question is relevant.
“This is an approach that might be suggested for patients at high bleeding risk who have the characteristics of the patients enrolled in the STOPDAPT-2 trials,” Dr. Capodanno said in an interview. In his own PCI practice treating ACS patients, “I would not feel safe enough with clopidogrel monotherapy after only 1 month.”
He considers the ACS population to have a particularly “delicate bleeding-ischemia trade-off,” which is why he thinks this question is relevant and needs to be explored further in additional populations. However, he might design trials differently in his own practice setting. For example, he would at the very least be interested in testing a more potent P2Y12 inhibitor such as ticagrelor when considering a single antiplatelet agent after a limited course of DAPT.
One message from this study is that “bleeding risk trumps PCI complexity,” according to Deepak L. Bhatt, MD, who recently assumed the position of director of Mount Sinai Heart in New York. He liked the approach the investigators took to address a complex and relevant clinical issue, but he also expressed reservations about the clinical applicability of this subgroup analysis.
“We really need more data before uniformly shortening DAPT duration in all patients,” Dr. Bhatt said in an interview. He considers this a hot clinical issue that is likely to generate more trials. He hopes these will provide more definitive evidence of when and how DAPT duration can be reduced. Overall, he anticipates progress toward tailoring therapy in specific populations in order to achieve the best risk-to-benefit balance.
Dr. Kimura has financial relationships with Boston Scientific, Daiichi Sankyo, Sanofi, Terumo, and Abbott Medical Japan, which provided funding for the STOPDAPT-2 and STOPDAPT-2 ACS trials. Dr. Capodanno reported financial relationships with Amgen, Arena, Chiesi, Daiichi Sakyo, Sanofi Aventis, and Terumo. Dr. Bhatt reported financial relationships with more than 20 pharmaceutical companies, including Abbott Medical.
Replacing dual-antiplatelet therapy (DAPT) with clopidogrel alone 1 month after percutaneous intervention (PCI) offers a lower risk of bleeding with comparable protection against cardiovascular events, according to two subgroup analyses of the Japanese STOPDAPT-2 and STOPDAPT-2 ACS trials.
The objective of these two analyses was to evaluate whether there was a benefit-to-risk ratio advantage for those who entered the study with high bleeding risk or who had undergone a complex PCI. Overall, bleeding risk was reduced without a major increase in cardiovascular events regardless of subgroup, according to results published by a multicenter group of Japanese investigators.
In this substudy, like the previously published studies from which the data were drawn, the primary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, stroke, and Thrombolysis In Myocardial Infarction bleeding (major or minor).
The proportion of patients in the 1-month and 12-month DAPT groups reaching this composite endpoint at 1 year was not significantly different among patients stratified by baseline bleeding risk or by PCI complexity, according to a multicenter group of authors led by Takeshi Kimura, MD, department of cardiovascular medicine, Kyoto University.
Shortened DAPT is focus of multiple trials
The new analysis, published in JACC Asia, is a follow-up to the 2019 STOPDAPT-2 trial, published in JAMA, and the 2022 STOPDAPT-2 ACS trial, published in JAMA Cardiology. The first tested 1- versus 12-month DAPT in PCI patients receiving a drug-eluting stent. The second study compared the same strategies in patients undergoing PCI to treat an acute coronary syndrome (ACS).
Both studies were conducted in Japan. DAPT consisted of the P2Y12 receptor inhibitor clopidogrel plus aspirin. The experimental arm received this regimen for 1 month followed by clopidogrel monotherapy. The control arm remained on DAPT for 12 months.
The study is potentially important because it addresses the challenge of finding “the sweet spot of antiplatelet therapy in East Asian patients,” according to the coauthors of an accompanying editorial in the same issue of JACC Asia.
Previous data suggest East Asians have a higher risk of bleeding but lower anti-ischemic benefits from DAPT therapy, explained the coauthors, Antonio Greco, MD and Davide Capodanno, MD, PhD, both from the University of Catania (Italy). They praised the effort to explore this question.
In the STOPDAPT-2 trial, the shortened DAPT regimen was associated with a significantly lower rate of a composite endpoint of cardiovascular and bleeding events than standard DAPT, meeting criteria for superiority as well as noninferiority. In the STOPDAPT-2 ACS trial, shortened DAPT failed to achieve noninferiority to standard DAPT because of an increase in cardiovascular events despite a reduction in bleeding events.
Neither of these studies specifically compared shortened to standard DAPT in patients with high bleeding risk or in patients who underwent complex PCI, which are among the most common patient groups in which to consider a modified DAPT regimen. To do this, two new substudies were performed with the combined data from 5,997 patients in the two STOPDAPT-2 trials.
Two candidate groups for shortened DAPT evaluated
In the first substudy, the 1,893 patients who met criteria for high bleeding risk were compared with the 4,104 who did not. In those with a high risk of bleeding, the proportion reaching a primary endpoint at 1 year was lower, but not significantly different, for those on 1-month versus standard DAPT (5.01% vs. 5.14%). This was also true in those without an elevated bleeding risk (1.90% vs. 2.02%).
In the second substudy, 999 patients who had a complex PCI, defined by such characteristics as implantation of at least three stents or chronic total occlusion in the target lesions, were compared with the 4,998 who did not. Again, the primary endpoint was lower in both those who had a complex PCI (3.15% vs. 4.07%) and those who did not (2.78% vs. 2.82%).
Not surprisingly, patients with a high bleeding risk benefited from a substantially lower risk of bleeding events on the 1-month DAPT regimen (0.66% vs. 2.27%). The cost was a higher risk of cardiovascular events (4.35% vs. 3.52%), but this difference did not reach significance. Those without an elevated bleeding risk also had a lower risk of bleeding events (0.43% vs. 0.85%) but a higher risk of cardiovascular events (1.56% vs. 1.22%). Again, differences were nonsignificant. In the substudy evaluating DAPT duration in relation to complex PCI, the rate of cardiovascular events at 1 year in those treated with short versus 12-month DAPT was nearly identical (2.53% vs. 2.52%). In the non–complex PCI patients, event rates were nonsignificantly greater on the shortened DAPT regimen (2.38% vs. 1.86%), but the bleeding rate was lower on shortened DAPT whether PCI had been complex (0.63% vs. 1.75%) or not (0.48% vs. 1.22%).
In the absence of any major signal that complex PCI benefited from longer duration DAPT, “complex PCI might not be an appropriate determinant for DAPT durations,” according to Dr. Kimura and coinvestigators.
Study data might not be generalizable
Dr. Greco and Dr. Capodanno pointed out that there are differences between patients and PCI practices in Japan relative to other areas of the world, limiting the generalizability of these findings even if the question is relevant.
“This is an approach that might be suggested for patients at high bleeding risk who have the characteristics of the patients enrolled in the STOPDAPT-2 trials,” Dr. Capodanno said in an interview. In his own PCI practice treating ACS patients, “I would not feel safe enough with clopidogrel monotherapy after only 1 month.”
He considers the ACS population to have a particularly “delicate bleeding-ischemia trade-off,” which is why he thinks this question is relevant and needs to be explored further in additional populations. However, he might design trials differently in his own practice setting. For example, he would at the very least be interested in testing a more potent P2Y12 inhibitor such as ticagrelor when considering a single antiplatelet agent after a limited course of DAPT.
One message from this study is that “bleeding risk trumps PCI complexity,” according to Deepak L. Bhatt, MD, who recently assumed the position of director of Mount Sinai Heart in New York. He liked the approach the investigators took to address a complex and relevant clinical issue, but he also expressed reservations about the clinical applicability of this subgroup analysis.
“We really need more data before uniformly shortening DAPT duration in all patients,” Dr. Bhatt said in an interview. He considers this a hot clinical issue that is likely to generate more trials. He hopes these will provide more definitive evidence of when and how DAPT duration can be reduced. Overall, he anticipates progress toward tailoring therapy in specific populations in order to achieve the best risk-to-benefit balance.
Dr. Kimura has financial relationships with Boston Scientific, Daiichi Sankyo, Sanofi, Terumo, and Abbott Medical Japan, which provided funding for the STOPDAPT-2 and STOPDAPT-2 ACS trials. Dr. Capodanno reported financial relationships with Amgen, Arena, Chiesi, Daiichi Sakyo, Sanofi Aventis, and Terumo. Dr. Bhatt reported financial relationships with more than 20 pharmaceutical companies, including Abbott Medical.
FROM JACC ASIA
Can 6 minutes of intense cycling put the brakes on Alzheimer’s?
new research suggests.
In a small study of healthy adults, 6 minutes of high-intensity cycling increased circulating levels of brain-derived neurotrophic factor (BDNF) to a significantly greater extent than prolonged light cycling or fasting.
However, the data do not suggest that 6 minutes of high-intensity exercise “wards off dementia,” cautioned lead investigator Travis Gibbons, MSc, PhD candidate in environmental physiology at the University of Otago (New Zealand), Dunedin, and now postdoctoral fellow at the University of British Columbia – Okanagan, Kelowna.
“Like all science, this is just a small piece that supports a potential mechanistic role for how exercise might improve brain health,” Dr. Gibbons told this news organization.
The findings were published online in the Journal of Physiology.
Targeting BDNF
Both intermittent fasting and exercise have previously been shown to have potent neuroprotective effects; and an acute upregulation of BDNF appears to be a common mechanistic link.
To tease apart the influence of fasting and exercise on BDNF production, Dr. Gibbons and colleagues studied 12 aerobically fit, healthy men (n = 6) and women (n = 6) aged 20-40 years.
In a study that employed a repeated-measures crossover design, they assessed circulating BDNF levels after a 20-hour fast, prolonged (90-min) light cycling, short (6-min) high-intensity cycling, and combined fasting and exercise.
Six minutes of high-intensity exercise appeared to be the most efficient way to increase BDNF.
Fasting for 20 hours led to a ninefold increase in ketone body delivery to the brain but had no effect on any metric of BDNF in peripheral circulation at rest or during exercise.
Six minutes of high-intensity exercise increased every metric of circulating BDNF four to five times more than prolonged low-intensity exercise.
In addition, the increase in plasma-derived BDNF correlated with a sixfold increase in circulating lactate irrespective of feeding or fasting state.
Lactate delivery?
“My leading theory is that, during and following intense exercise, lactate produced by muscles is delivered and consumed by the brain,” Dr. Gibbons noted.
“It takes high-intensity exercise to provoke this ‘cerebral substrate switch’ from glucose to lactate. Critically, this cerebral substrate switch has been shown to contribute to the early processes that upregulate BDNF production in the brain,” he said.
However, “Whether this translates to ‘warding off dementia’ is not clear,” Dr. Gibbons added.
The study also suggests that increases in plasma volume and platelet concentration appear to play a role in concentrating BDNF in the circulation during exercise.
The investigators note that BDNF and other neurotrophic-based pharmaceutical therapies have shown “great promise” in slowing and even arresting neurodegenerative processes in animals, but attempts to harness the protective power of BDNF in human neurodegeneration have thus far failed.
“Whether episodically upregulating BDNF production with intense exercise is an effective strategy to curb age-related cognitive decline in humans is unknown, but animal models indicate that it is and that BDNF plays a primary role,” the researchers write.
Funding for the study was provided by the Healthcare Otago Charitable Trust. The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In a small study of healthy adults, 6 minutes of high-intensity cycling increased circulating levels of brain-derived neurotrophic factor (BDNF) to a significantly greater extent than prolonged light cycling or fasting.
However, the data do not suggest that 6 minutes of high-intensity exercise “wards off dementia,” cautioned lead investigator Travis Gibbons, MSc, PhD candidate in environmental physiology at the University of Otago (New Zealand), Dunedin, and now postdoctoral fellow at the University of British Columbia – Okanagan, Kelowna.
“Like all science, this is just a small piece that supports a potential mechanistic role for how exercise might improve brain health,” Dr. Gibbons told this news organization.
The findings were published online in the Journal of Physiology.
Targeting BDNF
Both intermittent fasting and exercise have previously been shown to have potent neuroprotective effects; and an acute upregulation of BDNF appears to be a common mechanistic link.
To tease apart the influence of fasting and exercise on BDNF production, Dr. Gibbons and colleagues studied 12 aerobically fit, healthy men (n = 6) and women (n = 6) aged 20-40 years.
In a study that employed a repeated-measures crossover design, they assessed circulating BDNF levels after a 20-hour fast, prolonged (90-min) light cycling, short (6-min) high-intensity cycling, and combined fasting and exercise.
Six minutes of high-intensity exercise appeared to be the most efficient way to increase BDNF.
Fasting for 20 hours led to a ninefold increase in ketone body delivery to the brain but had no effect on any metric of BDNF in peripheral circulation at rest or during exercise.
Six minutes of high-intensity exercise increased every metric of circulating BDNF four to five times more than prolonged low-intensity exercise.
In addition, the increase in plasma-derived BDNF correlated with a sixfold increase in circulating lactate irrespective of feeding or fasting state.
Lactate delivery?
“My leading theory is that, during and following intense exercise, lactate produced by muscles is delivered and consumed by the brain,” Dr. Gibbons noted.
“It takes high-intensity exercise to provoke this ‘cerebral substrate switch’ from glucose to lactate. Critically, this cerebral substrate switch has been shown to contribute to the early processes that upregulate BDNF production in the brain,” he said.
However, “Whether this translates to ‘warding off dementia’ is not clear,” Dr. Gibbons added.
The study also suggests that increases in plasma volume and platelet concentration appear to play a role in concentrating BDNF in the circulation during exercise.
The investigators note that BDNF and other neurotrophic-based pharmaceutical therapies have shown “great promise” in slowing and even arresting neurodegenerative processes in animals, but attempts to harness the protective power of BDNF in human neurodegeneration have thus far failed.
“Whether episodically upregulating BDNF production with intense exercise is an effective strategy to curb age-related cognitive decline in humans is unknown, but animal models indicate that it is and that BDNF plays a primary role,” the researchers write.
Funding for the study was provided by the Healthcare Otago Charitable Trust. The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In a small study of healthy adults, 6 minutes of high-intensity cycling increased circulating levels of brain-derived neurotrophic factor (BDNF) to a significantly greater extent than prolonged light cycling or fasting.
However, the data do not suggest that 6 minutes of high-intensity exercise “wards off dementia,” cautioned lead investigator Travis Gibbons, MSc, PhD candidate in environmental physiology at the University of Otago (New Zealand), Dunedin, and now postdoctoral fellow at the University of British Columbia – Okanagan, Kelowna.
“Like all science, this is just a small piece that supports a potential mechanistic role for how exercise might improve brain health,” Dr. Gibbons told this news organization.
The findings were published online in the Journal of Physiology.
Targeting BDNF
Both intermittent fasting and exercise have previously been shown to have potent neuroprotective effects; and an acute upregulation of BDNF appears to be a common mechanistic link.
To tease apart the influence of fasting and exercise on BDNF production, Dr. Gibbons and colleagues studied 12 aerobically fit, healthy men (n = 6) and women (n = 6) aged 20-40 years.
In a study that employed a repeated-measures crossover design, they assessed circulating BDNF levels after a 20-hour fast, prolonged (90-min) light cycling, short (6-min) high-intensity cycling, and combined fasting and exercise.
Six minutes of high-intensity exercise appeared to be the most efficient way to increase BDNF.
Fasting for 20 hours led to a ninefold increase in ketone body delivery to the brain but had no effect on any metric of BDNF in peripheral circulation at rest or during exercise.
Six minutes of high-intensity exercise increased every metric of circulating BDNF four to five times more than prolonged low-intensity exercise.
In addition, the increase in plasma-derived BDNF correlated with a sixfold increase in circulating lactate irrespective of feeding or fasting state.
Lactate delivery?
“My leading theory is that, during and following intense exercise, lactate produced by muscles is delivered and consumed by the brain,” Dr. Gibbons noted.
“It takes high-intensity exercise to provoke this ‘cerebral substrate switch’ from glucose to lactate. Critically, this cerebral substrate switch has been shown to contribute to the early processes that upregulate BDNF production in the brain,” he said.
However, “Whether this translates to ‘warding off dementia’ is not clear,” Dr. Gibbons added.
The study also suggests that increases in plasma volume and platelet concentration appear to play a role in concentrating BDNF in the circulation during exercise.
The investigators note that BDNF and other neurotrophic-based pharmaceutical therapies have shown “great promise” in slowing and even arresting neurodegenerative processes in animals, but attempts to harness the protective power of BDNF in human neurodegeneration have thus far failed.
“Whether episodically upregulating BDNF production with intense exercise is an effective strategy to curb age-related cognitive decline in humans is unknown, but animal models indicate that it is and that BDNF plays a primary role,” the researchers write.
Funding for the study was provided by the Healthcare Otago Charitable Trust. The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF PHYSIOLOGY
Singer is paralyzed after delay in care; hospital must pay
Delay in treatment will cost hospital millions
according to a report on WFAA.com, among other news sites.
On March 21, 2019, Judy “Jessie” Adams, then part of a singing-songwriting duo with her husband, Richard, went to Premier Interventional Pain Management, in Flower Mound, Tex., prior to the couple’s drive to Ohio for a funeral. At Premier, Jesse received an epidural steroid injection (ESI) that she hoped would ease her back pain during the long drive.
Instead, the injection ended up increasing her pain.
“He [the pain physician] gave me the shot, but I couldn’t feel my legs. They were tingling, but I couldn’t feel them,” Mrs. Adams explained. “The pain was so bad in my back.” In their suit, Adams and her husband alleged that the doctor had probably “nicked a blood vessel during the ESI procedure, causing Jessie to hemorrhage.” (The couple’s suit against the doctor was settled prior to trial.)
Mrs. Adams remained under observation at the pain facility for about 1½ hours, at which point she was taken by ambulance to nearby Texas Health Presbyterian Hospital. There, in the emergency department, staff ordered a “STAT MRI” in preparation for an emergency laminectomy.
For reasons that remain murky, the MRI wasn’t performed for 1 hour and 37 minutes. The emergency laminectomy itself wasn’t started until more than 5 hours after Adams had been admitted to the ED. This was a direct violation of hospital protocol, which required that emergency surgeries be performed within 1 hour of admittance in the first available surgical suite. (At trial, Mrs. Adams’s attorneys from Lyons & Simmons offered evidence that a suite became available 49 minutes after Adams had arrived at the ED.)
During the wait, Mrs. Adams continued to experience excruciating pain. “I kept screaming: ‘Help me,’ ” she recalled. At trial, her attorneys argued that the hospital’s delay in addressing her spinal emergency led directly to her current paralysis, which keeps her confined to a wheelchair and renders her incontinent.
The hospital disagreed. In court, it maintained that Mrs. Adams was already paralyzed when she arrived at the ED and that there was no delay in care.
The jury saw things differently, however. Siding with the plaintiffs, it awarded Mrs. Adams and her husband $10.1 million, including $500,000 for Mr. Adams’s loss of future earnings and $1 million for his “loss of consortium” with his wife.
Their music career now effectively over, Mr. Adams spends most of his time taking care of Mrs. Adams.
“Music was our lifeblood for so many years, and he can’t do it anymore,” Mrs. Adams said. “He goes upstairs to play his guitar and write, and suddenly I need him to come and cath me. I just feel like I’m going to wake up from this bad dream, but it’s the same routine.”
Two doctors are absolved in woman’s sudden death
In a 3-2 decision in December 2022, the Pennsylvania Supreme Court ruled that the state’s 2-year statute of limitations in wrongful-death cases applies even in cases in which plaintiffs fail to identify the cause of death in a timely manner, as a report in the Claims Journal indicates.
The decision stems from a lawsuit filed by Linda Reibenstein on behalf of her mother, Mary Ann Whitman, who died in late April 2010 from a ruptured aortic aneurysm.
On April 12, 2010, Ms. Whitman visited Patrick D. Conaboy, MD, a Scranton family physician, complaining of a persistent cough, fever, and lower-back pain. Following an initial examination, Dr. Conaboy ordered an aortic duplex ultrasound scan and a CT scan of the patient’s abdomen.
The ultrasound was performed by radiologist Charles Barax, MD, who reviewed both scans. He identified a “poorly visualized aortic aneurysm.” At this point, Dr. Conaboy referred Ms. Whitman to a vascular surgeon. But before this visit could take place, Whitman’s aneurysm ruptured, killing her. This was listed as the medical cause of death on the patient’s death certificate.
In April 2011, Ms. Reibenstein filed a claim against Dr. Barax, alleging that he had failed to gauge the severity of her mother’s condition. Ms. Reibenstein’s attorney wasn’t able to question Dr. Barax on the record until well after the state’s 2-year statute of limitations had elapsed. When he did testify, Dr. Barax explained that the scans’ image quality prevented him from determining whether Whitman’s aneurysm was rupturing or simply bleeding. Despite this, he insisted that he had warned Dr. Conaboy of the potential for Ms. Whitman’s aneurysm to rupture.
In March 2016, nearly 6 years after her mother’s death, Ms. Reibenstein filed a new lawsuit, this one against Dr. Conaboy, whom she alleged had failed to properly treat her mother’s condition. Dr. Conaboy, in turn, asked the court for summary judgment – that is, a judgment in his favor without a full trial – arguing that the state’s window for filing a wrongful-death claim had long since closed. For their part, Ms. Reibenstein and her attorney argued that the state’s 2-year statute of limitations didn’t start until the plaintiff had discovered the cause of her mother’s death.
Initially refusing to dismiss the case, a lower court reconsidered Dr. Conaboy’s motion for summary judgment and ruled that Ms. Reibenstein had failed to present any evidence of “affirmative misrepresentation or fraudulent concealment.” In other words, in the absence of any willful attempt on the part of the defendant to hide the legal cause of death, which includes “acts, omissions, or events having some causative connection with the death,” the statute of limitations remained in effect, and the defendant’s motion was thereby granted.
Continuing the legal seesaw, a state appeals court reversed the lower-court ruling. Noting that the Pennsylvania malpractice statute was ambiguous, the court argued that it should be interpreted in a way that protects plaintiffs who seek “fair compensation” but encounter willfully erected obstacles in pursuit of their claim.
Dr. Conaboy then took his case to the state’s highest court. In its majority decision, the Pennsylvania Supreme Court staked out a narrow definition of cause of death – one based on the death certificate – and ruled that only willful fraud in that document would constitute the necessary condition for halting the claim’s clock. Furthermore, the high court said, when lawmakers adopted the Medical Care Availability and Reduction of Error Act in 2002, they did so with no guarantee “that all of the information necessary to sustain a claim will be gathered in the limitations period.”
Similarly, the court ruled, “at some point the clock must run out, lest health care providers remain subject to liability exposure indefinitely, with the prospect of a trial marred by the death or diminished memory of material witnesses or the loss of critical evidence.”
A version of this article first appeared on Medscape.com.
Delay in treatment will cost hospital millions
according to a report on WFAA.com, among other news sites.
On March 21, 2019, Judy “Jessie” Adams, then part of a singing-songwriting duo with her husband, Richard, went to Premier Interventional Pain Management, in Flower Mound, Tex., prior to the couple’s drive to Ohio for a funeral. At Premier, Jesse received an epidural steroid injection (ESI) that she hoped would ease her back pain during the long drive.
Instead, the injection ended up increasing her pain.
“He [the pain physician] gave me the shot, but I couldn’t feel my legs. They were tingling, but I couldn’t feel them,” Mrs. Adams explained. “The pain was so bad in my back.” In their suit, Adams and her husband alleged that the doctor had probably “nicked a blood vessel during the ESI procedure, causing Jessie to hemorrhage.” (The couple’s suit against the doctor was settled prior to trial.)
Mrs. Adams remained under observation at the pain facility for about 1½ hours, at which point she was taken by ambulance to nearby Texas Health Presbyterian Hospital. There, in the emergency department, staff ordered a “STAT MRI” in preparation for an emergency laminectomy.
For reasons that remain murky, the MRI wasn’t performed for 1 hour and 37 minutes. The emergency laminectomy itself wasn’t started until more than 5 hours after Adams had been admitted to the ED. This was a direct violation of hospital protocol, which required that emergency surgeries be performed within 1 hour of admittance in the first available surgical suite. (At trial, Mrs. Adams’s attorneys from Lyons & Simmons offered evidence that a suite became available 49 minutes after Adams had arrived at the ED.)
During the wait, Mrs. Adams continued to experience excruciating pain. “I kept screaming: ‘Help me,’ ” she recalled. At trial, her attorneys argued that the hospital’s delay in addressing her spinal emergency led directly to her current paralysis, which keeps her confined to a wheelchair and renders her incontinent.
The hospital disagreed. In court, it maintained that Mrs. Adams was already paralyzed when she arrived at the ED and that there was no delay in care.
The jury saw things differently, however. Siding with the plaintiffs, it awarded Mrs. Adams and her husband $10.1 million, including $500,000 for Mr. Adams’s loss of future earnings and $1 million for his “loss of consortium” with his wife.
Their music career now effectively over, Mr. Adams spends most of his time taking care of Mrs. Adams.
“Music was our lifeblood for so many years, and he can’t do it anymore,” Mrs. Adams said. “He goes upstairs to play his guitar and write, and suddenly I need him to come and cath me. I just feel like I’m going to wake up from this bad dream, but it’s the same routine.”
Two doctors are absolved in woman’s sudden death
In a 3-2 decision in December 2022, the Pennsylvania Supreme Court ruled that the state’s 2-year statute of limitations in wrongful-death cases applies even in cases in which plaintiffs fail to identify the cause of death in a timely manner, as a report in the Claims Journal indicates.
The decision stems from a lawsuit filed by Linda Reibenstein on behalf of her mother, Mary Ann Whitman, who died in late April 2010 from a ruptured aortic aneurysm.
On April 12, 2010, Ms. Whitman visited Patrick D. Conaboy, MD, a Scranton family physician, complaining of a persistent cough, fever, and lower-back pain. Following an initial examination, Dr. Conaboy ordered an aortic duplex ultrasound scan and a CT scan of the patient’s abdomen.
The ultrasound was performed by radiologist Charles Barax, MD, who reviewed both scans. He identified a “poorly visualized aortic aneurysm.” At this point, Dr. Conaboy referred Ms. Whitman to a vascular surgeon. But before this visit could take place, Whitman’s aneurysm ruptured, killing her. This was listed as the medical cause of death on the patient’s death certificate.
In April 2011, Ms. Reibenstein filed a claim against Dr. Barax, alleging that he had failed to gauge the severity of her mother’s condition. Ms. Reibenstein’s attorney wasn’t able to question Dr. Barax on the record until well after the state’s 2-year statute of limitations had elapsed. When he did testify, Dr. Barax explained that the scans’ image quality prevented him from determining whether Whitman’s aneurysm was rupturing or simply bleeding. Despite this, he insisted that he had warned Dr. Conaboy of the potential for Ms. Whitman’s aneurysm to rupture.
In March 2016, nearly 6 years after her mother’s death, Ms. Reibenstein filed a new lawsuit, this one against Dr. Conaboy, whom she alleged had failed to properly treat her mother’s condition. Dr. Conaboy, in turn, asked the court for summary judgment – that is, a judgment in his favor without a full trial – arguing that the state’s window for filing a wrongful-death claim had long since closed. For their part, Ms. Reibenstein and her attorney argued that the state’s 2-year statute of limitations didn’t start until the plaintiff had discovered the cause of her mother’s death.
Initially refusing to dismiss the case, a lower court reconsidered Dr. Conaboy’s motion for summary judgment and ruled that Ms. Reibenstein had failed to present any evidence of “affirmative misrepresentation or fraudulent concealment.” In other words, in the absence of any willful attempt on the part of the defendant to hide the legal cause of death, which includes “acts, omissions, or events having some causative connection with the death,” the statute of limitations remained in effect, and the defendant’s motion was thereby granted.
Continuing the legal seesaw, a state appeals court reversed the lower-court ruling. Noting that the Pennsylvania malpractice statute was ambiguous, the court argued that it should be interpreted in a way that protects plaintiffs who seek “fair compensation” but encounter willfully erected obstacles in pursuit of their claim.
Dr. Conaboy then took his case to the state’s highest court. In its majority decision, the Pennsylvania Supreme Court staked out a narrow definition of cause of death – one based on the death certificate – and ruled that only willful fraud in that document would constitute the necessary condition for halting the claim’s clock. Furthermore, the high court said, when lawmakers adopted the Medical Care Availability and Reduction of Error Act in 2002, they did so with no guarantee “that all of the information necessary to sustain a claim will be gathered in the limitations period.”
Similarly, the court ruled, “at some point the clock must run out, lest health care providers remain subject to liability exposure indefinitely, with the prospect of a trial marred by the death or diminished memory of material witnesses or the loss of critical evidence.”
A version of this article first appeared on Medscape.com.
Delay in treatment will cost hospital millions
according to a report on WFAA.com, among other news sites.
On March 21, 2019, Judy “Jessie” Adams, then part of a singing-songwriting duo with her husband, Richard, went to Premier Interventional Pain Management, in Flower Mound, Tex., prior to the couple’s drive to Ohio for a funeral. At Premier, Jesse received an epidural steroid injection (ESI) that she hoped would ease her back pain during the long drive.
Instead, the injection ended up increasing her pain.
“He [the pain physician] gave me the shot, but I couldn’t feel my legs. They were tingling, but I couldn’t feel them,” Mrs. Adams explained. “The pain was so bad in my back.” In their suit, Adams and her husband alleged that the doctor had probably “nicked a blood vessel during the ESI procedure, causing Jessie to hemorrhage.” (The couple’s suit against the doctor was settled prior to trial.)
Mrs. Adams remained under observation at the pain facility for about 1½ hours, at which point she was taken by ambulance to nearby Texas Health Presbyterian Hospital. There, in the emergency department, staff ordered a “STAT MRI” in preparation for an emergency laminectomy.
For reasons that remain murky, the MRI wasn’t performed for 1 hour and 37 minutes. The emergency laminectomy itself wasn’t started until more than 5 hours after Adams had been admitted to the ED. This was a direct violation of hospital protocol, which required that emergency surgeries be performed within 1 hour of admittance in the first available surgical suite. (At trial, Mrs. Adams’s attorneys from Lyons & Simmons offered evidence that a suite became available 49 minutes after Adams had arrived at the ED.)
During the wait, Mrs. Adams continued to experience excruciating pain. “I kept screaming: ‘Help me,’ ” she recalled. At trial, her attorneys argued that the hospital’s delay in addressing her spinal emergency led directly to her current paralysis, which keeps her confined to a wheelchair and renders her incontinent.
The hospital disagreed. In court, it maintained that Mrs. Adams was already paralyzed when she arrived at the ED and that there was no delay in care.
The jury saw things differently, however. Siding with the plaintiffs, it awarded Mrs. Adams and her husband $10.1 million, including $500,000 for Mr. Adams’s loss of future earnings and $1 million for his “loss of consortium” with his wife.
Their music career now effectively over, Mr. Adams spends most of his time taking care of Mrs. Adams.
“Music was our lifeblood for so many years, and he can’t do it anymore,” Mrs. Adams said. “He goes upstairs to play his guitar and write, and suddenly I need him to come and cath me. I just feel like I’m going to wake up from this bad dream, but it’s the same routine.”
Two doctors are absolved in woman’s sudden death
In a 3-2 decision in December 2022, the Pennsylvania Supreme Court ruled that the state’s 2-year statute of limitations in wrongful-death cases applies even in cases in which plaintiffs fail to identify the cause of death in a timely manner, as a report in the Claims Journal indicates.
The decision stems from a lawsuit filed by Linda Reibenstein on behalf of her mother, Mary Ann Whitman, who died in late April 2010 from a ruptured aortic aneurysm.
On April 12, 2010, Ms. Whitman visited Patrick D. Conaboy, MD, a Scranton family physician, complaining of a persistent cough, fever, and lower-back pain. Following an initial examination, Dr. Conaboy ordered an aortic duplex ultrasound scan and a CT scan of the patient’s abdomen.
The ultrasound was performed by radiologist Charles Barax, MD, who reviewed both scans. He identified a “poorly visualized aortic aneurysm.” At this point, Dr. Conaboy referred Ms. Whitman to a vascular surgeon. But before this visit could take place, Whitman’s aneurysm ruptured, killing her. This was listed as the medical cause of death on the patient’s death certificate.
In April 2011, Ms. Reibenstein filed a claim against Dr. Barax, alleging that he had failed to gauge the severity of her mother’s condition. Ms. Reibenstein’s attorney wasn’t able to question Dr. Barax on the record until well after the state’s 2-year statute of limitations had elapsed. When he did testify, Dr. Barax explained that the scans’ image quality prevented him from determining whether Whitman’s aneurysm was rupturing or simply bleeding. Despite this, he insisted that he had warned Dr. Conaboy of the potential for Ms. Whitman’s aneurysm to rupture.
In March 2016, nearly 6 years after her mother’s death, Ms. Reibenstein filed a new lawsuit, this one against Dr. Conaboy, whom she alleged had failed to properly treat her mother’s condition. Dr. Conaboy, in turn, asked the court for summary judgment – that is, a judgment in his favor without a full trial – arguing that the state’s window for filing a wrongful-death claim had long since closed. For their part, Ms. Reibenstein and her attorney argued that the state’s 2-year statute of limitations didn’t start until the plaintiff had discovered the cause of her mother’s death.
Initially refusing to dismiss the case, a lower court reconsidered Dr. Conaboy’s motion for summary judgment and ruled that Ms. Reibenstein had failed to present any evidence of “affirmative misrepresentation or fraudulent concealment.” In other words, in the absence of any willful attempt on the part of the defendant to hide the legal cause of death, which includes “acts, omissions, or events having some causative connection with the death,” the statute of limitations remained in effect, and the defendant’s motion was thereby granted.
Continuing the legal seesaw, a state appeals court reversed the lower-court ruling. Noting that the Pennsylvania malpractice statute was ambiguous, the court argued that it should be interpreted in a way that protects plaintiffs who seek “fair compensation” but encounter willfully erected obstacles in pursuit of their claim.
Dr. Conaboy then took his case to the state’s highest court. In its majority decision, the Pennsylvania Supreme Court staked out a narrow definition of cause of death – one based on the death certificate – and ruled that only willful fraud in that document would constitute the necessary condition for halting the claim’s clock. Furthermore, the high court said, when lawmakers adopted the Medical Care Availability and Reduction of Error Act in 2002, they did so with no guarantee “that all of the information necessary to sustain a claim will be gathered in the limitations period.”
Similarly, the court ruled, “at some point the clock must run out, lest health care providers remain subject to liability exposure indefinitely, with the prospect of a trial marred by the death or diminished memory of material witnesses or the loss of critical evidence.”
A version of this article first appeared on Medscape.com.
Scant evidence for proton pump inhibitor role in gastric cancer
The available evidence suggests that proton pump inhibitors (PPIs) do not cause gastric cancer, researchers say.
A new study could help resolve a controversy over one of the most serious side effects attributed to the widely used medications.
“Our findings are reassuring, especially to all those patients who have an indication for long-term PPI use and need persistent and effective gastric acid suppression to prevent serious health consequences,” said Daniele Piovani, MSc, PhD, an assistant professor of medical statistics at Humanitas University, Milan, in an email to this news organization.
Previous studies did not take into account the probability that the diseases for which the medications were prescribed might have caused the cancer, Dr. Piovani and colleagues write in Alimentary Pharmacology and Therapeutics.
Researchers have worried about the potential of PPIs to cause cancer after finding that they are associated with enterochromaffin-like cells, gastric atrophy, and changes in gut microbiota and gastric mucosal immunology.
Observational studies and meta-analyses showed a link between PPIs and an increased risk for gastric cancer.
“However, the underlying conditions for which PPIs are prescribed are associated with gastric cancer,” said Dr. Piovani. “This may result in an apparent association between PPIs and gastric cancer.”
Another potential confounding factor is that as-yet undiagnosed cancer might also cause symptoms that are treated with PPIs. Patient behavior also may play a role, she noted.
“Let’s imagine a patient with peptic ulcer who takes PPIs,” said Dr. Piovani. “He may not only have peptic ulcer but also be a heavy smoker. He may drink much more alcohol, have a different dietary pattern, be more likely to be exposed to high levels of stress, etc. in respect to a control [patient] who does not have peptic ulcer and does not take PPIs.”
Comparing two drug classes
More recent studies have compared people taking PPIs to people taking histamine-2 receptor antagonists (H2RAs). H2RAs are often used to treat the same conditions as PPIs, but they are not as strongly linked to hypergastrinemia and are not associated with gastric atrophy, so they might serve as good comparators.
Since results of these studies have been conflicting, Dr. Piovani and colleagues attempted to weigh them together in a systematic review and meta-analysis. They identified two randomized clinical trials and 12 observational studies with a total of over 6 million patients.
One randomized controlled trial involved Helicobacter pylori–negative patients with bleeding ulcers. Researchers assigned 138 to 20 mg daily rabeprazole (a PPI) and 132 to 40 mg famotidine (an H2RA). After a year, no cancer occurred.
The other randomized controlled trial involved H. pylori–negative patients with idiopathic peptic ulcers. Investigators assigned 114 to 30 mg lansoprazole (another PPI) and 114 to 40 mg famotidine. In 2 years, one patient receiving famotidine developed cancer.
The researchers found several methodological problems with these trials. One flaw is that the study periods were not long enough to accurately measure what effects the medications might have on gastric cancers, which are a rare outcome, they note. The evidence from these studies was so weak they could not draw conclusions from the results, the investigators conclude.
Pooling data from the 11 observational trials they were able to combine, the researchers found that PPI users had a one-third higher random relative risk of cancer than H2RA users (95% confidence interval, 1.11-1.59). However, these studies were heterogenous, and five of them did not adjust for age and sex, as well as other potentially confounding covariates.
The remaining six observational studies adjusted for age, sex, and at least two other covariates that could affect the risk for gastric cancer. These studies had a total of 2.5 million patients and 7,372 gastric cancers. Combined, these studies showed an RR of gastric cancer in PPI users, compared with H2RA users of 1.07, which was not statistically significant (95% CI, 0.97-1.19).
The researchers found no clear evidence of a dose-response or of an increased risk with longer-term use of PPIs.
Findings support practice guidance
“I found this relatively reassuring,” Mark Lewis, MD, director of gastrointestinal oncology at Intermountain Healthcare, Murray, Utah, told this news organization.
PPIs do dramatically increase the pH of the stomach, stimulating the stomach to try to compensate in a process that can sometimes give rise to tumors, Dr. Lewis said. But these tumors appear to be benign.
Other concerns about PPI use, such as reduction in bone density, remain under investigation, he said.
Some H2RA blockers might actually pose a greater cancer risk than PPIs, said Dr. Lewis, and many clinicians seem to favor PPIs. “I have seen a huge sea change where most patients are on PPIs. And I would say that H2RA blockers are older and increasingly the exception in terms of usage, not the rule.”
The investigators note that observational studies by their nature cannot prove cause and effect, but because gastric cancer is so rare, a randomized controlled trial of PPIs versus H2RAs that is large enough to be definitive may not be feasible.
They conclude that their findings support the American Gastroenterological Association recommendation that “the decision to discontinue PPIs should be based solely on the lack of an indication for use and not because of concern for PPI-associated adverse effects.”
Dr. Piovani and Dr. Lewis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The available evidence suggests that proton pump inhibitors (PPIs) do not cause gastric cancer, researchers say.
A new study could help resolve a controversy over one of the most serious side effects attributed to the widely used medications.
“Our findings are reassuring, especially to all those patients who have an indication for long-term PPI use and need persistent and effective gastric acid suppression to prevent serious health consequences,” said Daniele Piovani, MSc, PhD, an assistant professor of medical statistics at Humanitas University, Milan, in an email to this news organization.
Previous studies did not take into account the probability that the diseases for which the medications were prescribed might have caused the cancer, Dr. Piovani and colleagues write in Alimentary Pharmacology and Therapeutics.
Researchers have worried about the potential of PPIs to cause cancer after finding that they are associated with enterochromaffin-like cells, gastric atrophy, and changes in gut microbiota and gastric mucosal immunology.
Observational studies and meta-analyses showed a link between PPIs and an increased risk for gastric cancer.
“However, the underlying conditions for which PPIs are prescribed are associated with gastric cancer,” said Dr. Piovani. “This may result in an apparent association between PPIs and gastric cancer.”
Another potential confounding factor is that as-yet undiagnosed cancer might also cause symptoms that are treated with PPIs. Patient behavior also may play a role, she noted.
“Let’s imagine a patient with peptic ulcer who takes PPIs,” said Dr. Piovani. “He may not only have peptic ulcer but also be a heavy smoker. He may drink much more alcohol, have a different dietary pattern, be more likely to be exposed to high levels of stress, etc. in respect to a control [patient] who does not have peptic ulcer and does not take PPIs.”
Comparing two drug classes
More recent studies have compared people taking PPIs to people taking histamine-2 receptor antagonists (H2RAs). H2RAs are often used to treat the same conditions as PPIs, but they are not as strongly linked to hypergastrinemia and are not associated with gastric atrophy, so they might serve as good comparators.
Since results of these studies have been conflicting, Dr. Piovani and colleagues attempted to weigh them together in a systematic review and meta-analysis. They identified two randomized clinical trials and 12 observational studies with a total of over 6 million patients.
One randomized controlled trial involved Helicobacter pylori–negative patients with bleeding ulcers. Researchers assigned 138 to 20 mg daily rabeprazole (a PPI) and 132 to 40 mg famotidine (an H2RA). After a year, no cancer occurred.
The other randomized controlled trial involved H. pylori–negative patients with idiopathic peptic ulcers. Investigators assigned 114 to 30 mg lansoprazole (another PPI) and 114 to 40 mg famotidine. In 2 years, one patient receiving famotidine developed cancer.
The researchers found several methodological problems with these trials. One flaw is that the study periods were not long enough to accurately measure what effects the medications might have on gastric cancers, which are a rare outcome, they note. The evidence from these studies was so weak they could not draw conclusions from the results, the investigators conclude.
Pooling data from the 11 observational trials they were able to combine, the researchers found that PPI users had a one-third higher random relative risk of cancer than H2RA users (95% confidence interval, 1.11-1.59). However, these studies were heterogenous, and five of them did not adjust for age and sex, as well as other potentially confounding covariates.
The remaining six observational studies adjusted for age, sex, and at least two other covariates that could affect the risk for gastric cancer. These studies had a total of 2.5 million patients and 7,372 gastric cancers. Combined, these studies showed an RR of gastric cancer in PPI users, compared with H2RA users of 1.07, which was not statistically significant (95% CI, 0.97-1.19).
The researchers found no clear evidence of a dose-response or of an increased risk with longer-term use of PPIs.
Findings support practice guidance
“I found this relatively reassuring,” Mark Lewis, MD, director of gastrointestinal oncology at Intermountain Healthcare, Murray, Utah, told this news organization.
PPIs do dramatically increase the pH of the stomach, stimulating the stomach to try to compensate in a process that can sometimes give rise to tumors, Dr. Lewis said. But these tumors appear to be benign.
Other concerns about PPI use, such as reduction in bone density, remain under investigation, he said.
Some H2RA blockers might actually pose a greater cancer risk than PPIs, said Dr. Lewis, and many clinicians seem to favor PPIs. “I have seen a huge sea change where most patients are on PPIs. And I would say that H2RA blockers are older and increasingly the exception in terms of usage, not the rule.”
The investigators note that observational studies by their nature cannot prove cause and effect, but because gastric cancer is so rare, a randomized controlled trial of PPIs versus H2RAs that is large enough to be definitive may not be feasible.
They conclude that their findings support the American Gastroenterological Association recommendation that “the decision to discontinue PPIs should be based solely on the lack of an indication for use and not because of concern for PPI-associated adverse effects.”
Dr. Piovani and Dr. Lewis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The available evidence suggests that proton pump inhibitors (PPIs) do not cause gastric cancer, researchers say.
A new study could help resolve a controversy over one of the most serious side effects attributed to the widely used medications.
“Our findings are reassuring, especially to all those patients who have an indication for long-term PPI use and need persistent and effective gastric acid suppression to prevent serious health consequences,” said Daniele Piovani, MSc, PhD, an assistant professor of medical statistics at Humanitas University, Milan, in an email to this news organization.
Previous studies did not take into account the probability that the diseases for which the medications were prescribed might have caused the cancer, Dr. Piovani and colleagues write in Alimentary Pharmacology and Therapeutics.
Researchers have worried about the potential of PPIs to cause cancer after finding that they are associated with enterochromaffin-like cells, gastric atrophy, and changes in gut microbiota and gastric mucosal immunology.
Observational studies and meta-analyses showed a link between PPIs and an increased risk for gastric cancer.
“However, the underlying conditions for which PPIs are prescribed are associated with gastric cancer,” said Dr. Piovani. “This may result in an apparent association between PPIs and gastric cancer.”
Another potential confounding factor is that as-yet undiagnosed cancer might also cause symptoms that are treated with PPIs. Patient behavior also may play a role, she noted.
“Let’s imagine a patient with peptic ulcer who takes PPIs,” said Dr. Piovani. “He may not only have peptic ulcer but also be a heavy smoker. He may drink much more alcohol, have a different dietary pattern, be more likely to be exposed to high levels of stress, etc. in respect to a control [patient] who does not have peptic ulcer and does not take PPIs.”
Comparing two drug classes
More recent studies have compared people taking PPIs to people taking histamine-2 receptor antagonists (H2RAs). H2RAs are often used to treat the same conditions as PPIs, but they are not as strongly linked to hypergastrinemia and are not associated with gastric atrophy, so they might serve as good comparators.
Since results of these studies have been conflicting, Dr. Piovani and colleagues attempted to weigh them together in a systematic review and meta-analysis. They identified two randomized clinical trials and 12 observational studies with a total of over 6 million patients.
One randomized controlled trial involved Helicobacter pylori–negative patients with bleeding ulcers. Researchers assigned 138 to 20 mg daily rabeprazole (a PPI) and 132 to 40 mg famotidine (an H2RA). After a year, no cancer occurred.
The other randomized controlled trial involved H. pylori–negative patients with idiopathic peptic ulcers. Investigators assigned 114 to 30 mg lansoprazole (another PPI) and 114 to 40 mg famotidine. In 2 years, one patient receiving famotidine developed cancer.
The researchers found several methodological problems with these trials. One flaw is that the study periods were not long enough to accurately measure what effects the medications might have on gastric cancers, which are a rare outcome, they note. The evidence from these studies was so weak they could not draw conclusions from the results, the investigators conclude.
Pooling data from the 11 observational trials they were able to combine, the researchers found that PPI users had a one-third higher random relative risk of cancer than H2RA users (95% confidence interval, 1.11-1.59). However, these studies were heterogenous, and five of them did not adjust for age and sex, as well as other potentially confounding covariates.
The remaining six observational studies adjusted for age, sex, and at least two other covariates that could affect the risk for gastric cancer. These studies had a total of 2.5 million patients and 7,372 gastric cancers. Combined, these studies showed an RR of gastric cancer in PPI users, compared with H2RA users of 1.07, which was not statistically significant (95% CI, 0.97-1.19).
The researchers found no clear evidence of a dose-response or of an increased risk with longer-term use of PPIs.
Findings support practice guidance
“I found this relatively reassuring,” Mark Lewis, MD, director of gastrointestinal oncology at Intermountain Healthcare, Murray, Utah, told this news organization.
PPIs do dramatically increase the pH of the stomach, stimulating the stomach to try to compensate in a process that can sometimes give rise to tumors, Dr. Lewis said. But these tumors appear to be benign.
Other concerns about PPI use, such as reduction in bone density, remain under investigation, he said.
Some H2RA blockers might actually pose a greater cancer risk than PPIs, said Dr. Lewis, and many clinicians seem to favor PPIs. “I have seen a huge sea change where most patients are on PPIs. And I would say that H2RA blockers are older and increasingly the exception in terms of usage, not the rule.”
The investigators note that observational studies by their nature cannot prove cause and effect, but because gastric cancer is so rare, a randomized controlled trial of PPIs versus H2RAs that is large enough to be definitive may not be feasible.
They conclude that their findings support the American Gastroenterological Association recommendation that “the decision to discontinue PPIs should be based solely on the lack of an indication for use and not because of concern for PPI-associated adverse effects.”
Dr. Piovani and Dr. Lewis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALIMENTARY PHARMACOLOGY AND THERAPEUTICS
A 50-year-old woman with no significant history presented with erythematous, annular plaques, and papules on the dorsal hands and arms
. The prevalence and incidence is approximately 0.1%-0.4%. Although the condition is benign, it may be associated with more serious conditions such as HIV and malignancy. GA affects women more frequently than men but can affect any age group, although it most commonly presents in those ages 30 years and younger. While the exact etiology is unknown, GA has been most strongly associated with diabetes mellitus, hyperlipidemia, and autoimmune diseases.
The disease presents as localized, annular erythematous plaques and papules on the dorsal hands and feet in approximately 75% of cases. However, eruptions may appear on the trunk and extremities and can be categorized into patchy, generalized, interstitial, subcutaneous, or perforating subtypes. The lesions are often asymptomatic and typically not associated with any other symptoms.
The pathogenesis of GA is still under investigation, but recent studies suggest that a Th1-mediated dysregulation of the JAK-STAT pathway may contribute to the disease. Other hypotheses include a delayed hypersensitivity reaction or cell mediated immune response. The mechanism may be multifaceted, and epidemiologic research suggests a genetic predisposition in White individuals, but these findings may be associated with socioeconomic factors and disparities in health care.
GA presents on histology with palisading histiocytes surrounding focal collagen necrobiosis with mucin deposition. Tissue samples also display leukocytic infiltration of the dermis featuring multinucleated giant cells. There are defining features of the different subtypes, but focal collagen necrosis, the presence of histiocytes, and mucin deposition are consistent findings across all presentations.
GA lesions commonly regress on their own, but they tend to recur and can be functionally and visually unappealing to patients. The most common treatments for GA include topical corticosteroids, intralesional corticosteroid injections, and other anti-inflammatory drugs. These interventions can be administered in a variety of ways as the inflammation caused by GA exists on a spectrum, and less severe cases can be managed with topical or intralesional treatment. Systemic therapy may be necessary for severe and recalcitrant cases. Other interventions that have shown promise in smaller studies include phototherapy, hydroxychloroquine, and TNF-alpha inhibitors.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University College of Osteopathic Medicine, Tampa Bay Regional Campus, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Joshi TP and Duvic M. Am J Clin Dermatol. 2022 Jan;23(1):37-50. doi: 10.1007/s40257-021-00636-1.
Muse M et al. Dermatol Online J. 2021 Apr 15;27(4):13030/qt0m50398n.
Schmieder SJ et al. Granuloma Annulare. NIH National Center for Biotechnology Information [Updated 2022 Nov 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan. 7.
. The prevalence and incidence is approximately 0.1%-0.4%. Although the condition is benign, it may be associated with more serious conditions such as HIV and malignancy. GA affects women more frequently than men but can affect any age group, although it most commonly presents in those ages 30 years and younger. While the exact etiology is unknown, GA has been most strongly associated with diabetes mellitus, hyperlipidemia, and autoimmune diseases.
The disease presents as localized, annular erythematous plaques and papules on the dorsal hands and feet in approximately 75% of cases. However, eruptions may appear on the trunk and extremities and can be categorized into patchy, generalized, interstitial, subcutaneous, or perforating subtypes. The lesions are often asymptomatic and typically not associated with any other symptoms.
The pathogenesis of GA is still under investigation, but recent studies suggest that a Th1-mediated dysregulation of the JAK-STAT pathway may contribute to the disease. Other hypotheses include a delayed hypersensitivity reaction or cell mediated immune response. The mechanism may be multifaceted, and epidemiologic research suggests a genetic predisposition in White individuals, but these findings may be associated with socioeconomic factors and disparities in health care.
GA presents on histology with palisading histiocytes surrounding focal collagen necrobiosis with mucin deposition. Tissue samples also display leukocytic infiltration of the dermis featuring multinucleated giant cells. There are defining features of the different subtypes, but focal collagen necrosis, the presence of histiocytes, and mucin deposition are consistent findings across all presentations.
GA lesions commonly regress on their own, but they tend to recur and can be functionally and visually unappealing to patients. The most common treatments for GA include topical corticosteroids, intralesional corticosteroid injections, and other anti-inflammatory drugs. These interventions can be administered in a variety of ways as the inflammation caused by GA exists on a spectrum, and less severe cases can be managed with topical or intralesional treatment. Systemic therapy may be necessary for severe and recalcitrant cases. Other interventions that have shown promise in smaller studies include phototherapy, hydroxychloroquine, and TNF-alpha inhibitors.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University College of Osteopathic Medicine, Tampa Bay Regional Campus, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Joshi TP and Duvic M. Am J Clin Dermatol. 2022 Jan;23(1):37-50. doi: 10.1007/s40257-021-00636-1.
Muse M et al. Dermatol Online J. 2021 Apr 15;27(4):13030/qt0m50398n.
Schmieder SJ et al. Granuloma Annulare. NIH National Center for Biotechnology Information [Updated 2022 Nov 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan. 7.
. The prevalence and incidence is approximately 0.1%-0.4%. Although the condition is benign, it may be associated with more serious conditions such as HIV and malignancy. GA affects women more frequently than men but can affect any age group, although it most commonly presents in those ages 30 years and younger. While the exact etiology is unknown, GA has been most strongly associated with diabetes mellitus, hyperlipidemia, and autoimmune diseases.
The disease presents as localized, annular erythematous plaques and papules on the dorsal hands and feet in approximately 75% of cases. However, eruptions may appear on the trunk and extremities and can be categorized into patchy, generalized, interstitial, subcutaneous, or perforating subtypes. The lesions are often asymptomatic and typically not associated with any other symptoms.
The pathogenesis of GA is still under investigation, but recent studies suggest that a Th1-mediated dysregulation of the JAK-STAT pathway may contribute to the disease. Other hypotheses include a delayed hypersensitivity reaction or cell mediated immune response. The mechanism may be multifaceted, and epidemiologic research suggests a genetic predisposition in White individuals, but these findings may be associated with socioeconomic factors and disparities in health care.
GA presents on histology with palisading histiocytes surrounding focal collagen necrobiosis with mucin deposition. Tissue samples also display leukocytic infiltration of the dermis featuring multinucleated giant cells. There are defining features of the different subtypes, but focal collagen necrosis, the presence of histiocytes, and mucin deposition are consistent findings across all presentations.
GA lesions commonly regress on their own, but they tend to recur and can be functionally and visually unappealing to patients. The most common treatments for GA include topical corticosteroids, intralesional corticosteroid injections, and other anti-inflammatory drugs. These interventions can be administered in a variety of ways as the inflammation caused by GA exists on a spectrum, and less severe cases can be managed with topical or intralesional treatment. Systemic therapy may be necessary for severe and recalcitrant cases. Other interventions that have shown promise in smaller studies include phototherapy, hydroxychloroquine, and TNF-alpha inhibitors.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University College of Osteopathic Medicine, Tampa Bay Regional Campus, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Joshi TP and Duvic M. Am J Clin Dermatol. 2022 Jan;23(1):37-50. doi: 10.1007/s40257-021-00636-1.
Muse M et al. Dermatol Online J. 2021 Apr 15;27(4):13030/qt0m50398n.
Schmieder SJ et al. Granuloma Annulare. NIH National Center for Biotechnology Information [Updated 2022 Nov 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan. 7.
COVID leading cause of death among law enforcement for third year
A new report says 70 officers died of COVID-related causes after getting the virus while on the job. The number is down dramatically from 2021, when 405 officer deaths were attributed to COVID.
The annual count was published Wednesday by the National Law Enforcement Officers Memorial Fund.
In total, 226 officers died in the line of duty in 2022, which is a decrease of 61% from 2021.
The decrease “is almost entirely related to the significant reduction in COVID-19 deaths,” the report stated. The authors said the decline was likely due to “reduced infection rates and the broad availability and use of vaccinations.”
Reported deaths included federal, state, tribal, and local law enforcement officers.
Firearms-related fatalities were the second-leading cause of death among officers, with 64 in 2022. That count sustains a 21% increase seen in 2021, up from the decade-long average of 53 firearms-related deaths annually from 2010 to 2020.
Traffic-related causes ranked third for cause of death in 2022, accounting for 56 deaths.
“While overall line-of-duty deaths are trending down, the continuing trend of greater-than-average firearms-related deaths continues to be a serious concern,” Marcia Ferranto, the organization’s chief executive officer, said in a news release. “Using and reporting on this data allows us to highlight the continuing cost of maintaining our democracy, regrettably measured in the lives of the many law enforcement professionals who sacrifice everything fulfilling their promise to serve and protect.”
A version of this article first appeared on WebMD.com.
A new report says 70 officers died of COVID-related causes after getting the virus while on the job. The number is down dramatically from 2021, when 405 officer deaths were attributed to COVID.
The annual count was published Wednesday by the National Law Enforcement Officers Memorial Fund.
In total, 226 officers died in the line of duty in 2022, which is a decrease of 61% from 2021.
The decrease “is almost entirely related to the significant reduction in COVID-19 deaths,” the report stated. The authors said the decline was likely due to “reduced infection rates and the broad availability and use of vaccinations.”
Reported deaths included federal, state, tribal, and local law enforcement officers.
Firearms-related fatalities were the second-leading cause of death among officers, with 64 in 2022. That count sustains a 21% increase seen in 2021, up from the decade-long average of 53 firearms-related deaths annually from 2010 to 2020.
Traffic-related causes ranked third for cause of death in 2022, accounting for 56 deaths.
“While overall line-of-duty deaths are trending down, the continuing trend of greater-than-average firearms-related deaths continues to be a serious concern,” Marcia Ferranto, the organization’s chief executive officer, said in a news release. “Using and reporting on this data allows us to highlight the continuing cost of maintaining our democracy, regrettably measured in the lives of the many law enforcement professionals who sacrifice everything fulfilling their promise to serve and protect.”
A version of this article first appeared on WebMD.com.
A new report says 70 officers died of COVID-related causes after getting the virus while on the job. The number is down dramatically from 2021, when 405 officer deaths were attributed to COVID.
The annual count was published Wednesday by the National Law Enforcement Officers Memorial Fund.
In total, 226 officers died in the line of duty in 2022, which is a decrease of 61% from 2021.
The decrease “is almost entirely related to the significant reduction in COVID-19 deaths,” the report stated. The authors said the decline was likely due to “reduced infection rates and the broad availability and use of vaccinations.”
Reported deaths included federal, state, tribal, and local law enforcement officers.
Firearms-related fatalities were the second-leading cause of death among officers, with 64 in 2022. That count sustains a 21% increase seen in 2021, up from the decade-long average of 53 firearms-related deaths annually from 2010 to 2020.
Traffic-related causes ranked third for cause of death in 2022, accounting for 56 deaths.
“While overall line-of-duty deaths are trending down, the continuing trend of greater-than-average firearms-related deaths continues to be a serious concern,” Marcia Ferranto, the organization’s chief executive officer, said in a news release. “Using and reporting on this data allows us to highlight the continuing cost of maintaining our democracy, regrettably measured in the lives of the many law enforcement professionals who sacrifice everything fulfilling their promise to serve and protect.”
A version of this article first appeared on WebMD.com.
Oramed oral insulin fails to meet goal in type 2 diabetes
Oramed Pharmaceuticals’ investigational oral insulin failed to achieve its primary endpoint in a phase 3 trial, according to top-line results announced by the company.
“Therefore, Oramed expects to discontinue its oral insulin clinical activities for [type 2 diabetes],” according to a company statement.
, ORA-D-013-1, comparing the efficacy of the insulin product ORMD-0801 to placebo in 710 people with type 2 diabetes with inadequate glycemic control on two or three oral glucose-lowering agents.
The participants were randomized 2:2:1:1 into ORMD-0801 dosed at 8 mg once or twice daily, or placebo dosed once or twice daily. They completed a 21-day screening period, followed by a 26-week double-blind treatment period.
The product didn’t achieve the primary endpoint comparing reduction in hemoglobin A1c from baseline to 26 weeks, or the secondary endpoint of mean change in fasting plasma glucose at 26 weeks. There were no serious adverse events.
Oramed Pharmaceuticals specializes in developing oral delivery formulations of drugs currently delivered via injection. The company has offices in the United States and Israel.
Oramed CEO Nadav Kidron commented in the statement, “Today’s outcome is very disappointing, given the positive results from prior trials. Once full data from the studies are available, we expect to share relevant learnings and future plans. We thank all the patients, families, and health care professionals who participated in the trial.”
Insulin manufacturer Novo Nordisk had also been developing an oral insulin product. Successful phase 2a results were presented at the American Diabetes Association’s 2017 Scientific Sessions and full phase 2 feasibility results were published in Lancet Diabetes & Endocrinology in 2019.
However, Novo Nordisk, which manufactures the oral glucagon-like peptide-1 receptor agonist semaglutide (Rybelsus), subsequently discontinued development of their oral insulin product. According to a statement, “Initial results raised questions about truly addressing patients’ unmet needs with insulin therapy. Therefore, we discontinued this work to focus on projects that could in fact improve cardiometabolic outcomes for people living with diabetes.”
A version of this article first appeared on Medscape.com.
Oramed Pharmaceuticals’ investigational oral insulin failed to achieve its primary endpoint in a phase 3 trial, according to top-line results announced by the company.
“Therefore, Oramed expects to discontinue its oral insulin clinical activities for [type 2 diabetes],” according to a company statement.
, ORA-D-013-1, comparing the efficacy of the insulin product ORMD-0801 to placebo in 710 people with type 2 diabetes with inadequate glycemic control on two or three oral glucose-lowering agents.
The participants were randomized 2:2:1:1 into ORMD-0801 dosed at 8 mg once or twice daily, or placebo dosed once or twice daily. They completed a 21-day screening period, followed by a 26-week double-blind treatment period.
The product didn’t achieve the primary endpoint comparing reduction in hemoglobin A1c from baseline to 26 weeks, or the secondary endpoint of mean change in fasting plasma glucose at 26 weeks. There were no serious adverse events.
Oramed Pharmaceuticals specializes in developing oral delivery formulations of drugs currently delivered via injection. The company has offices in the United States and Israel.
Oramed CEO Nadav Kidron commented in the statement, “Today’s outcome is very disappointing, given the positive results from prior trials. Once full data from the studies are available, we expect to share relevant learnings and future plans. We thank all the patients, families, and health care professionals who participated in the trial.”
Insulin manufacturer Novo Nordisk had also been developing an oral insulin product. Successful phase 2a results were presented at the American Diabetes Association’s 2017 Scientific Sessions and full phase 2 feasibility results were published in Lancet Diabetes & Endocrinology in 2019.
However, Novo Nordisk, which manufactures the oral glucagon-like peptide-1 receptor agonist semaglutide (Rybelsus), subsequently discontinued development of their oral insulin product. According to a statement, “Initial results raised questions about truly addressing patients’ unmet needs with insulin therapy. Therefore, we discontinued this work to focus on projects that could in fact improve cardiometabolic outcomes for people living with diabetes.”
A version of this article first appeared on Medscape.com.
Oramed Pharmaceuticals’ investigational oral insulin failed to achieve its primary endpoint in a phase 3 trial, according to top-line results announced by the company.
“Therefore, Oramed expects to discontinue its oral insulin clinical activities for [type 2 diabetes],” according to a company statement.
, ORA-D-013-1, comparing the efficacy of the insulin product ORMD-0801 to placebo in 710 people with type 2 diabetes with inadequate glycemic control on two or three oral glucose-lowering agents.
The participants were randomized 2:2:1:1 into ORMD-0801 dosed at 8 mg once or twice daily, or placebo dosed once or twice daily. They completed a 21-day screening period, followed by a 26-week double-blind treatment period.
The product didn’t achieve the primary endpoint comparing reduction in hemoglobin A1c from baseline to 26 weeks, or the secondary endpoint of mean change in fasting plasma glucose at 26 weeks. There were no serious adverse events.
Oramed Pharmaceuticals specializes in developing oral delivery formulations of drugs currently delivered via injection. The company has offices in the United States and Israel.
Oramed CEO Nadav Kidron commented in the statement, “Today’s outcome is very disappointing, given the positive results from prior trials. Once full data from the studies are available, we expect to share relevant learnings and future plans. We thank all the patients, families, and health care professionals who participated in the trial.”
Insulin manufacturer Novo Nordisk had also been developing an oral insulin product. Successful phase 2a results were presented at the American Diabetes Association’s 2017 Scientific Sessions and full phase 2 feasibility results were published in Lancet Diabetes & Endocrinology in 2019.
However, Novo Nordisk, which manufactures the oral glucagon-like peptide-1 receptor agonist semaglutide (Rybelsus), subsequently discontinued development of their oral insulin product. According to a statement, “Initial results raised questions about truly addressing patients’ unmet needs with insulin therapy. Therefore, we discontinued this work to focus on projects that could in fact improve cardiometabolic outcomes for people living with diabetes.”
A version of this article first appeared on Medscape.com.
Fatigue and blurred vision
Gestational diabetes is a significant health problem worldwide that is associated with immediate and lifelong consequences for the affected woman and her infant. Gestational diabetes increases the risk for pregnancy-related complications, such as induced labor, cesarean delivery, and preeclampsia. There is also an increased risk for neonatal complications, including large-for-gestational-age birth weight, shoulder dystocia, birth injuries, lung disease, jaundice, and hypoglycemia. Regardless of birth weight, neonates born to mothers with gestational diabetes have greater adiposity than do neonates born to mothers without obesity and with normal glucose tolerance, and they have a predilection toward obesity and obesity-related metabolic disorders, including T2D in childhood and adulthood. Similarly, women who develop gestational diabetes have an increased lifetime risk for T2D as well as an increased risk for cardiovascular disease even if they do not progress to T2D.
According to the International Diabetes Federation, 1 in 6 pregnancies is affected by gestational diabetes. Risk factors include higher age and BMI, previous history of gestational diabetes, a family history of T2D, and polycystic ovarian syndrome. Patients may have few, if any, symptoms of gestational diabetes, or they may mistake their symptoms for the normal side effects of pregnancy. Potential symptoms include blurred vision, tingling or numbness in the hands and/or feet, excessive thirst, frequent urination, sores that heal slowly, and excessive fatigue.
The American Diabetes Association (ADA) states that the treatment of gestational diabetes should include medical nutrition therapy, physical activity, and weight management, depending on pregestational weight. Glucose monitoring is essential: Patients should aim for fasting glucose < 95 mg/dL (5.3 mmol/L) and either 1-hour postprandial glucose < 140 mg/dL (7.8 mmol/L) or 2-hour postprandial glucose < 120 mg/dL (6.7 mmol/L). According to the ADA, insulin should be added to lifestyle modifications if needed to achieve glycemic targets. Metformin and glyburide are not recommended as first-line agents because both cross the placenta to the fetus. Long-term safety data are not available for the use of other oral and noninsulin injectable glucose-lowering medications during pregnancy.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Gestational diabetes is a significant health problem worldwide that is associated with immediate and lifelong consequences for the affected woman and her infant. Gestational diabetes increases the risk for pregnancy-related complications, such as induced labor, cesarean delivery, and preeclampsia. There is also an increased risk for neonatal complications, including large-for-gestational-age birth weight, shoulder dystocia, birth injuries, lung disease, jaundice, and hypoglycemia. Regardless of birth weight, neonates born to mothers with gestational diabetes have greater adiposity than do neonates born to mothers without obesity and with normal glucose tolerance, and they have a predilection toward obesity and obesity-related metabolic disorders, including T2D in childhood and adulthood. Similarly, women who develop gestational diabetes have an increased lifetime risk for T2D as well as an increased risk for cardiovascular disease even if they do not progress to T2D.
According to the International Diabetes Federation, 1 in 6 pregnancies is affected by gestational diabetes. Risk factors include higher age and BMI, previous history of gestational diabetes, a family history of T2D, and polycystic ovarian syndrome. Patients may have few, if any, symptoms of gestational diabetes, or they may mistake their symptoms for the normal side effects of pregnancy. Potential symptoms include blurred vision, tingling or numbness in the hands and/or feet, excessive thirst, frequent urination, sores that heal slowly, and excessive fatigue.
The American Diabetes Association (ADA) states that the treatment of gestational diabetes should include medical nutrition therapy, physical activity, and weight management, depending on pregestational weight. Glucose monitoring is essential: Patients should aim for fasting glucose < 95 mg/dL (5.3 mmol/L) and either 1-hour postprandial glucose < 140 mg/dL (7.8 mmol/L) or 2-hour postprandial glucose < 120 mg/dL (6.7 mmol/L). According to the ADA, insulin should be added to lifestyle modifications if needed to achieve glycemic targets. Metformin and glyburide are not recommended as first-line agents because both cross the placenta to the fetus. Long-term safety data are not available for the use of other oral and noninsulin injectable glucose-lowering medications during pregnancy.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Gestational diabetes is a significant health problem worldwide that is associated with immediate and lifelong consequences for the affected woman and her infant. Gestational diabetes increases the risk for pregnancy-related complications, such as induced labor, cesarean delivery, and preeclampsia. There is also an increased risk for neonatal complications, including large-for-gestational-age birth weight, shoulder dystocia, birth injuries, lung disease, jaundice, and hypoglycemia. Regardless of birth weight, neonates born to mothers with gestational diabetes have greater adiposity than do neonates born to mothers without obesity and with normal glucose tolerance, and they have a predilection toward obesity and obesity-related metabolic disorders, including T2D in childhood and adulthood. Similarly, women who develop gestational diabetes have an increased lifetime risk for T2D as well as an increased risk for cardiovascular disease even if they do not progress to T2D.
According to the International Diabetes Federation, 1 in 6 pregnancies is affected by gestational diabetes. Risk factors include higher age and BMI, previous history of gestational diabetes, a family history of T2D, and polycystic ovarian syndrome. Patients may have few, if any, symptoms of gestational diabetes, or they may mistake their symptoms for the normal side effects of pregnancy. Potential symptoms include blurred vision, tingling or numbness in the hands and/or feet, excessive thirst, frequent urination, sores that heal slowly, and excessive fatigue.
The American Diabetes Association (ADA) states that the treatment of gestational diabetes should include medical nutrition therapy, physical activity, and weight management, depending on pregestational weight. Glucose monitoring is essential: Patients should aim for fasting glucose < 95 mg/dL (5.3 mmol/L) and either 1-hour postprandial glucose < 140 mg/dL (7.8 mmol/L) or 2-hour postprandial glucose < 120 mg/dL (6.7 mmol/L). According to the ADA, insulin should be added to lifestyle modifications if needed to achieve glycemic targets. Metformin and glyburide are not recommended as first-line agents because both cross the placenta to the fetus. Long-term safety data are not available for the use of other oral and noninsulin injectable glucose-lowering medications during pregnancy.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 32-year-old Asian American woman (gravida 2 para 1) presents at 26 weeks' gestation for experiencing fatigue and blurred vision. The patient's previous pregnancy 3 years earlier was an uncomplicated vaginal delivery at 38 weeks' gestation. The baby weighed 7 lb 8 oz at delivery. The patient's maternal family history is notable for hypertension and type 2 diabetes (T2D). At the time of presentation, the patient is 5 ft 4 in, and her prepregnancy body mass index (BMI) was 31.8. Physical examination reveals blood pressure of 130/88 beats/min and fetal heart tones at 148 beats/min. She does not report ocular pain and there is no evidence of ocular redness, swelling, or discharge. The patient reports viral gastroenteritis approximately 10 days earlier, which has since resolved. A 1-hour oral glucose tolerance test (OGTT) at 24 weeks was abnormal; a subsequent 3-hour OGTT showed:
• 109 mg/dL (7:30 AM)
• 205 mg/dL (8:30 AM)
• 164 mg/dL (9:30 AM)
• 166 mg/dL (10:30 AM)
Low serum LDH shows potential as depression biomarker
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM GENERAL HOSPITAL PSYCHIATRY