User login
Do fellowship programs help prepare general surgery residents for board exams?
WASHINGTON – Pass rates on the general surgery board exams were significantly higher for programs in which residents trained alongside surgical fellows, according to a retrospective study presented at the annual clinical congress of the American College of Surgeons.
The study adds another perspective to the ongoing debate about the impact of fellowships on general surgery residency and residency programs. Mohammed J. Al Fayyadh, MD, of the University of Texas Health Science Center at San Antonio and his associates investigated the impact of fellowships on program pass rates for general surgery boards. They reviewed American Board of Surgery exam data for the classes of 2010-2014, which included 242 programs (a total of 5,191 resident examinees), of which 148 had fellows participating (3,767 resident examinees).
The findings suggest that having fellows in a program has a positive impact on the pass rates of those taking the ABS general surgery exam. Pass rates were significantly higher for general surgery programs with fellows. This trend held for all measures studied: Qualifying (written) exam (88% vs. 86%), certifying (oral) exam (83% vs. 80%), and combined exams (74% vs. 69%). Differences between the groups were statistically significant.
The pass rates tended to be higher in programs with higher Fel:Res ratios. For example, programs with a 1.5:1 Fel:Res ratio had the highest pass rates for the qualifying, certifying, and combined exams.
The impact of subspecialty fellowships on general surgery residencies has been the subject of research and debate in recent years. Some studies have suggested that subspecialty training for fellows has meant that general surgery residents have less opportunity to operate in areas such as trauma (“Trauma operative training declining for general surgery residents,” ACS Surgery News, Oct. 4, 2016) and vascular surgery (Ann Vasc Surg. 2016;33;98-102).
Taken in this context, it is possible that the programs with more fellows simply recruit more competitive residents who tend to do better on exams. However, this could also reflect more resources in fellowship programs that are available to all trainees or better mentorship opportunities, said Dr. Al Fayyadh. He concluded that more research is needed to tease out the impact of fellowship programs on general surgery resident education.
Dr. Al Fayyadh had no disclosures.
WASHINGTON – Pass rates on the general surgery board exams were significantly higher for programs in which residents trained alongside surgical fellows, according to a retrospective study presented at the annual clinical congress of the American College of Surgeons.
The study adds another perspective to the ongoing debate about the impact of fellowships on general surgery residency and residency programs. Mohammed J. Al Fayyadh, MD, of the University of Texas Health Science Center at San Antonio and his associates investigated the impact of fellowships on program pass rates for general surgery boards. They reviewed American Board of Surgery exam data for the classes of 2010-2014, which included 242 programs (a total of 5,191 resident examinees), of which 148 had fellows participating (3,767 resident examinees).
The findings suggest that having fellows in a program has a positive impact on the pass rates of those taking the ABS general surgery exam. Pass rates were significantly higher for general surgery programs with fellows. This trend held for all measures studied: Qualifying (written) exam (88% vs. 86%), certifying (oral) exam (83% vs. 80%), and combined exams (74% vs. 69%). Differences between the groups were statistically significant.
The pass rates tended to be higher in programs with higher Fel:Res ratios. For example, programs with a 1.5:1 Fel:Res ratio had the highest pass rates for the qualifying, certifying, and combined exams.
The impact of subspecialty fellowships on general surgery residencies has been the subject of research and debate in recent years. Some studies have suggested that subspecialty training for fellows has meant that general surgery residents have less opportunity to operate in areas such as trauma (“Trauma operative training declining for general surgery residents,” ACS Surgery News, Oct. 4, 2016) and vascular surgery (Ann Vasc Surg. 2016;33;98-102).
Taken in this context, it is possible that the programs with more fellows simply recruit more competitive residents who tend to do better on exams. However, this could also reflect more resources in fellowship programs that are available to all trainees or better mentorship opportunities, said Dr. Al Fayyadh. He concluded that more research is needed to tease out the impact of fellowship programs on general surgery resident education.
Dr. Al Fayyadh had no disclosures.
WASHINGTON – Pass rates on the general surgery board exams were significantly higher for programs in which residents trained alongside surgical fellows, according to a retrospective study presented at the annual clinical congress of the American College of Surgeons.
The study adds another perspective to the ongoing debate about the impact of fellowships on general surgery residency and residency programs. Mohammed J. Al Fayyadh, MD, of the University of Texas Health Science Center at San Antonio and his associates investigated the impact of fellowships on program pass rates for general surgery boards. They reviewed American Board of Surgery exam data for the classes of 2010-2014, which included 242 programs (a total of 5,191 resident examinees), of which 148 had fellows participating (3,767 resident examinees).
The findings suggest that having fellows in a program has a positive impact on the pass rates of those taking the ABS general surgery exam. Pass rates were significantly higher for general surgery programs with fellows. This trend held for all measures studied: Qualifying (written) exam (88% vs. 86%), certifying (oral) exam (83% vs. 80%), and combined exams (74% vs. 69%). Differences between the groups were statistically significant.
The pass rates tended to be higher in programs with higher Fel:Res ratios. For example, programs with a 1.5:1 Fel:Res ratio had the highest pass rates for the qualifying, certifying, and combined exams.
The impact of subspecialty fellowships on general surgery residencies has been the subject of research and debate in recent years. Some studies have suggested that subspecialty training for fellows has meant that general surgery residents have less opportunity to operate in areas such as trauma (“Trauma operative training declining for general surgery residents,” ACS Surgery News, Oct. 4, 2016) and vascular surgery (Ann Vasc Surg. 2016;33;98-102).
Taken in this context, it is possible that the programs with more fellows simply recruit more competitive residents who tend to do better on exams. However, this could also reflect more resources in fellowship programs that are available to all trainees or better mentorship opportunities, said Dr. Al Fayyadh. He concluded that more research is needed to tease out the impact of fellowship programs on general surgery resident education.
Dr. Al Fayyadh had no disclosures.
AT THE ACS CLINICAL CONGRESS
It’s elementary: Watson aids in breast cancer decisions
SAN ANTONIO – When oncologists at Manipal Hospitals in Bangalore, India, need help with a breast cancer conundrum, they can make the electronic equivalent of the famous cry for help “Watson, come here, I want you!”
In this case, Watson is not the real-life sidekick of Alexander Graham Bell or the fictional companion of Sherlock Holmes, but Watson for Oncology (WFO), an IBM-created artificial intelligence platform being developed in the United States and used in clinical practice in India to help guide clinical decision making but not to replace clinicians’ judgment, explained S.P. Somashekhar, MBBS, MS, MCH, FRCS, chairman of the Manipal Comprehensive Cancer Center.
This is no game
WFO is the clinical cousin of the artificial intelligence platform, also named Watson, that beat all-time champion Ken Jennings on the television game show “Jeopardy!” The system was named after Thomas J. Watson, IBM’s first chief executive officer.
The version of Watson used in India was developed by physicians and investigators at Memorial Sloan Kettering Cancer Center (MSKCC) in New York and by IBM. Watson for Oncology is fed national treatment guidelines, more than 1,000 training cases, MSKCC internal guidelines, and medical literature curated by MSKCC; it then chews over the data and spits out evidence-based recommendations.
At Manipal Hospitals, the electronic medical record contains an “Ask Watson” button that allows clinicians to interrogate the artificial entity’s vast stores of medical data to offer recommendations that clinicians can then use, modify, or reject at their discretion.
The system analyzes more than 100 patient attributes and then offers options with a green label for “recommended,” amber label “for consideration,” or a red label for “not recommended.”
Concordance study
At SABCS, Dr. Somashekhar presented results of a study evaluating the concordance of treatment recommendations between WFO and the members of the Manipal Multidisciplinary Tumor Board (MTB).
They looked at data on 638 patients treated in their hospital system over the three prior years, assessing the MTBs initial, best joint decision at the time of the original treatment decision (T1), and compared it with WFO’s recommendation made in 2016 (T2) and with a blinded MTB re-review of nonconcordant cases, also made in 2016.
Evaluating concordance by stage, they found that, for 514 cases of nonmetastatic disease, there was 79% concordance between the original MTB decisions and WFO’s recommendations for therapies in the combined “for consideration” and “recommended” categories.
For 124 cases of metastatic disease, however, Watson and its human counterparts were more frequently at odds, with only a 46% concordance.
In a subset analysis by receptor status, the investigators found that Watson was best – that is, most highly concordant – in triple-negative disease, with a 67.9% concordance in regard to MTB choices. In contrast, for patients with metastatic disease negative for the human epidermal growth factor receptor-2 (HER2), the concordance rate was a low 35%.
Dr. Somashekhar commented that part of the explanation for the discordant concordance rates by tumor subtype can be attributed to the fact that patients with triple-negative breast cancers have fewer treatment options than patients with HER2 negative–only tumors, who have a much broader array of possibilities.
One area where WFO has its two-legged colleagues beaten hands down, however, is in the time it takes to capture and analyze data: humans took a mean of 20 minutes, Watson a median of 40 seconds.
The investigators acknowledged that WFO represents a further step toward personalized medicine, but emphasized that software will never replace the doctor-patient relationship or override the treating physician’s decisions.
‘This one would actually help’
At the briefing, moderator C. Kent Osborne, MD, from Baylor College of Medicine, Houston, commented, “I guess this not going to put us out of business as physicians – at least I hope not.”
Asked whether WFO could be a useful clinical tool or just another nuisance task added to an already overcrowded clinic schedule, Dr. Osborne responded, “I think this one would actually help.”
This study was investigative and received no external funding. Dr. Somashekhar, Dr. Osborne, and Dr. Blaes reported no conflicts of interest.
SAN ANTONIO – When oncologists at Manipal Hospitals in Bangalore, India, need help with a breast cancer conundrum, they can make the electronic equivalent of the famous cry for help “Watson, come here, I want you!”
In this case, Watson is not the real-life sidekick of Alexander Graham Bell or the fictional companion of Sherlock Holmes, but Watson for Oncology (WFO), an IBM-created artificial intelligence platform being developed in the United States and used in clinical practice in India to help guide clinical decision making but not to replace clinicians’ judgment, explained S.P. Somashekhar, MBBS, MS, MCH, FRCS, chairman of the Manipal Comprehensive Cancer Center.
This is no game
WFO is the clinical cousin of the artificial intelligence platform, also named Watson, that beat all-time champion Ken Jennings on the television game show “Jeopardy!” The system was named after Thomas J. Watson, IBM’s first chief executive officer.
The version of Watson used in India was developed by physicians and investigators at Memorial Sloan Kettering Cancer Center (MSKCC) in New York and by IBM. Watson for Oncology is fed national treatment guidelines, more than 1,000 training cases, MSKCC internal guidelines, and medical literature curated by MSKCC; it then chews over the data and spits out evidence-based recommendations.
At Manipal Hospitals, the electronic medical record contains an “Ask Watson” button that allows clinicians to interrogate the artificial entity’s vast stores of medical data to offer recommendations that clinicians can then use, modify, or reject at their discretion.
The system analyzes more than 100 patient attributes and then offers options with a green label for “recommended,” amber label “for consideration,” or a red label for “not recommended.”
Concordance study
At SABCS, Dr. Somashekhar presented results of a study evaluating the concordance of treatment recommendations between WFO and the members of the Manipal Multidisciplinary Tumor Board (MTB).
They looked at data on 638 patients treated in their hospital system over the three prior years, assessing the MTBs initial, best joint decision at the time of the original treatment decision (T1), and compared it with WFO’s recommendation made in 2016 (T2) and with a blinded MTB re-review of nonconcordant cases, also made in 2016.
Evaluating concordance by stage, they found that, for 514 cases of nonmetastatic disease, there was 79% concordance between the original MTB decisions and WFO’s recommendations for therapies in the combined “for consideration” and “recommended” categories.
For 124 cases of metastatic disease, however, Watson and its human counterparts were more frequently at odds, with only a 46% concordance.
In a subset analysis by receptor status, the investigators found that Watson was best – that is, most highly concordant – in triple-negative disease, with a 67.9% concordance in regard to MTB choices. In contrast, for patients with metastatic disease negative for the human epidermal growth factor receptor-2 (HER2), the concordance rate was a low 35%.
Dr. Somashekhar commented that part of the explanation for the discordant concordance rates by tumor subtype can be attributed to the fact that patients with triple-negative breast cancers have fewer treatment options than patients with HER2 negative–only tumors, who have a much broader array of possibilities.
One area where WFO has its two-legged colleagues beaten hands down, however, is in the time it takes to capture and analyze data: humans took a mean of 20 minutes, Watson a median of 40 seconds.
The investigators acknowledged that WFO represents a further step toward personalized medicine, but emphasized that software will never replace the doctor-patient relationship or override the treating physician’s decisions.
‘This one would actually help’
At the briefing, moderator C. Kent Osborne, MD, from Baylor College of Medicine, Houston, commented, “I guess this not going to put us out of business as physicians – at least I hope not.”
Asked whether WFO could be a useful clinical tool or just another nuisance task added to an already overcrowded clinic schedule, Dr. Osborne responded, “I think this one would actually help.”
This study was investigative and received no external funding. Dr. Somashekhar, Dr. Osborne, and Dr. Blaes reported no conflicts of interest.
SAN ANTONIO – When oncologists at Manipal Hospitals in Bangalore, India, need help with a breast cancer conundrum, they can make the electronic equivalent of the famous cry for help “Watson, come here, I want you!”
In this case, Watson is not the real-life sidekick of Alexander Graham Bell or the fictional companion of Sherlock Holmes, but Watson for Oncology (WFO), an IBM-created artificial intelligence platform being developed in the United States and used in clinical practice in India to help guide clinical decision making but not to replace clinicians’ judgment, explained S.P. Somashekhar, MBBS, MS, MCH, FRCS, chairman of the Manipal Comprehensive Cancer Center.
This is no game
WFO is the clinical cousin of the artificial intelligence platform, also named Watson, that beat all-time champion Ken Jennings on the television game show “Jeopardy!” The system was named after Thomas J. Watson, IBM’s first chief executive officer.
The version of Watson used in India was developed by physicians and investigators at Memorial Sloan Kettering Cancer Center (MSKCC) in New York and by IBM. Watson for Oncology is fed national treatment guidelines, more than 1,000 training cases, MSKCC internal guidelines, and medical literature curated by MSKCC; it then chews over the data and spits out evidence-based recommendations.
At Manipal Hospitals, the electronic medical record contains an “Ask Watson” button that allows clinicians to interrogate the artificial entity’s vast stores of medical data to offer recommendations that clinicians can then use, modify, or reject at their discretion.
The system analyzes more than 100 patient attributes and then offers options with a green label for “recommended,” amber label “for consideration,” or a red label for “not recommended.”
Concordance study
At SABCS, Dr. Somashekhar presented results of a study evaluating the concordance of treatment recommendations between WFO and the members of the Manipal Multidisciplinary Tumor Board (MTB).
They looked at data on 638 patients treated in their hospital system over the three prior years, assessing the MTBs initial, best joint decision at the time of the original treatment decision (T1), and compared it with WFO’s recommendation made in 2016 (T2) and with a blinded MTB re-review of nonconcordant cases, also made in 2016.
Evaluating concordance by stage, they found that, for 514 cases of nonmetastatic disease, there was 79% concordance between the original MTB decisions and WFO’s recommendations for therapies in the combined “for consideration” and “recommended” categories.
For 124 cases of metastatic disease, however, Watson and its human counterparts were more frequently at odds, with only a 46% concordance.
In a subset analysis by receptor status, the investigators found that Watson was best – that is, most highly concordant – in triple-negative disease, with a 67.9% concordance in regard to MTB choices. In contrast, for patients with metastatic disease negative for the human epidermal growth factor receptor-2 (HER2), the concordance rate was a low 35%.
Dr. Somashekhar commented that part of the explanation for the discordant concordance rates by tumor subtype can be attributed to the fact that patients with triple-negative breast cancers have fewer treatment options than patients with HER2 negative–only tumors, who have a much broader array of possibilities.
One area where WFO has its two-legged colleagues beaten hands down, however, is in the time it takes to capture and analyze data: humans took a mean of 20 minutes, Watson a median of 40 seconds.
The investigators acknowledged that WFO represents a further step toward personalized medicine, but emphasized that software will never replace the doctor-patient relationship or override the treating physician’s decisions.
‘This one would actually help’
At the briefing, moderator C. Kent Osborne, MD, from Baylor College of Medicine, Houston, commented, “I guess this not going to put us out of business as physicians – at least I hope not.”
Asked whether WFO could be a useful clinical tool or just another nuisance task added to an already overcrowded clinic schedule, Dr. Osborne responded, “I think this one would actually help.”
This study was investigative and received no external funding. Dr. Somashekhar, Dr. Osborne, and Dr. Blaes reported no conflicts of interest.
AT SABCS 2016
Key clinical point: There was good, if imperfect, concordance in breast cancer treatment decisions between the software platform Watson for Oncology and human tumor board members.
Major finding: Concordance on treatment decisions for nonmetastatic breast cancers was 79%; concordance for metastatic cancers was 46%.
Data source: Single-institution study comparing treatment-decision concordance rates between a computerized decision tool and human reviewers.
Disclosures: This study was investigative and received no external funding. Dr. Somashekhar, Dr. Osborne, and Dr. Blaes reported no conflicts of interest.
VIDEO: Watson for Oncology offers electronic curbside consults in breast cancer
SAN ANTONIO – Watson for Oncology or WFO, is the clinical cousin of the IBM-created cognitive computing system best known as the machine that defeated all-time champions on the TV quiz show “Jeopardy!”
WFO, however, has the much more important function of providing oncologists with evidence-based support for clinical decisions. Data being presented here at the San Antonio Breast Cancer Symposium show that in Bangalore, India, where WFO is used in a large hospital system, there is good concordance between tumor board recommendations and WFO’s recommendations about the treatment of breast cancer, although much more work needs to be done. The investigators emphasize that WFO is a highly useful tool that can augment but will not replace clinical judgment, and can never replace the physician-patient relationship.
In this video interview, Andrew Norden, MD, deputy chief health officer for the IBM Watson project, based in Cambridge, Mass., describes how Watson for Oncology works, and how different versions of the Watson platform are being used in medicine throughout the world.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Watson for Oncology or WFO, is the clinical cousin of the IBM-created cognitive computing system best known as the machine that defeated all-time champions on the TV quiz show “Jeopardy!”
WFO, however, has the much more important function of providing oncologists with evidence-based support for clinical decisions. Data being presented here at the San Antonio Breast Cancer Symposium show that in Bangalore, India, where WFO is used in a large hospital system, there is good concordance between tumor board recommendations and WFO’s recommendations about the treatment of breast cancer, although much more work needs to be done. The investigators emphasize that WFO is a highly useful tool that can augment but will not replace clinical judgment, and can never replace the physician-patient relationship.
In this video interview, Andrew Norden, MD, deputy chief health officer for the IBM Watson project, based in Cambridge, Mass., describes how Watson for Oncology works, and how different versions of the Watson platform are being used in medicine throughout the world.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Watson for Oncology or WFO, is the clinical cousin of the IBM-created cognitive computing system best known as the machine that defeated all-time champions on the TV quiz show “Jeopardy!”
WFO, however, has the much more important function of providing oncologists with evidence-based support for clinical decisions. Data being presented here at the San Antonio Breast Cancer Symposium show that in Bangalore, India, where WFO is used in a large hospital system, there is good concordance between tumor board recommendations and WFO’s recommendations about the treatment of breast cancer, although much more work needs to be done. The investigators emphasize that WFO is a highly useful tool that can augment but will not replace clinical judgment, and can never replace the physician-patient relationship.
In this video interview, Andrew Norden, MD, deputy chief health officer for the IBM Watson project, based in Cambridge, Mass., describes how Watson for Oncology works, and how different versions of the Watson platform are being used in medicine throughout the world.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SABCS 2016
VIDEO: Abemaciclib reduces Ki67 expression in early HR+/HER2– breast cancer
SAN ANTONIO – Abemaciclib, both alone and in combination with anastrozole, significantly reduced Ki67 expression vs. anastrozole monotherapy after 2 weeks of treatment in the NeoMONARCH phase II neoadjuvant clinical trial of postmenopausal patients with hormone receptor–positive, HER2-negative early-stage breast cancer.
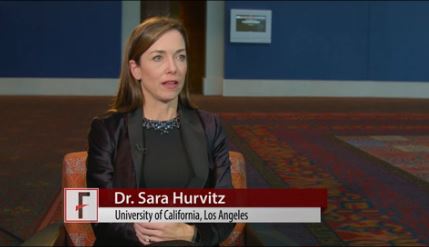
The findings, given that a change in Ki67 at 2 weeks in neoadjuvant studies appears to predict improved disease-free survival in adjuvant studies, support continued evaluation of the cyclin-dependent kinase-4 (CDK4) inhibitor for the treatment of patients with early-stage breast cancer, Sara Hurvitz, MD, reported at the San Antonio Breast Cancer Symposium.
“In hormone receptor–positive breast cancers, estrogen stimulates D-type cyclins, resulting in increased activity of CDK4 and CDK6, and then phosphorylate RB – the tumor suppressor protein retinoblastoma – which releases the E2F transcription factor,” explained Dr. Hurvitz of the University of California, Los Angeles.
This in turn ultimately leads to cell cycle progression from G1 to S.
“This increased rate of proliferation can be observed in tumor tissue samples by measuring the expression of Ki67. Blocking CDK4 and CDK6 should lead to a decrease in E2F expression, as well as a drop in the cell cycling and a drop in Ki67,” she said, adding that cell cycle arrest may induce senescence, which may also induce a phenotype that’s characterized by an immune cell infiltrate.
Indeed, in study subjects randomized to receive abemaciclib, treatment was shown to induce profound cell cycle arrest defined by decreased Ki67 and E2F targeted proliferation messenger RNAs, and reduction of expression of genes associated with senescence.
“Abemaciclib alone or in combination with anastrozole significantly reduced the Ki67 expression compared to anastrozole alone after 2 weeks of therapy, based on the geometric mean change and complete cell cycle arrest, and the study did meet its primary endpoint,” she said.
In a video interview, Dr. Hurvitz discussed the study methodology, results, and safety findings, as well as an intriguing observation regarding the effects of treatment on tumor differentiation and immune infiltrates over time.
This study was sponsored by Eli Lilly. Dr. Hurvitz has received renumeration for research and/or travel from Amgen, Bayer, BioMarin, Boehringer Ingelheim, Dignitana, Eli Lilly, Genentech, GSK, Medivation, Merrimack, Novartis, OBI Pharma, Pfizer, Puma Biotechnology, and Roche.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Abemaciclib, both alone and in combination with anastrozole, significantly reduced Ki67 expression vs. anastrozole monotherapy after 2 weeks of treatment in the NeoMONARCH phase II neoadjuvant clinical trial of postmenopausal patients with hormone receptor–positive, HER2-negative early-stage breast cancer.
The findings, given that a change in Ki67 at 2 weeks in neoadjuvant studies appears to predict improved disease-free survival in adjuvant studies, support continued evaluation of the cyclin-dependent kinase-4 (CDK4) inhibitor for the treatment of patients with early-stage breast cancer, Sara Hurvitz, MD, reported at the San Antonio Breast Cancer Symposium.
“In hormone receptor–positive breast cancers, estrogen stimulates D-type cyclins, resulting in increased activity of CDK4 and CDK6, and then phosphorylate RB – the tumor suppressor protein retinoblastoma – which releases the E2F transcription factor,” explained Dr. Hurvitz of the University of California, Los Angeles.
This in turn ultimately leads to cell cycle progression from G1 to S.
“This increased rate of proliferation can be observed in tumor tissue samples by measuring the expression of Ki67. Blocking CDK4 and CDK6 should lead to a decrease in E2F expression, as well as a drop in the cell cycling and a drop in Ki67,” she said, adding that cell cycle arrest may induce senescence, which may also induce a phenotype that’s characterized by an immune cell infiltrate.
Indeed, in study subjects randomized to receive abemaciclib, treatment was shown to induce profound cell cycle arrest defined by decreased Ki67 and E2F targeted proliferation messenger RNAs, and reduction of expression of genes associated with senescence.
“Abemaciclib alone or in combination with anastrozole significantly reduced the Ki67 expression compared to anastrozole alone after 2 weeks of therapy, based on the geometric mean change and complete cell cycle arrest, and the study did meet its primary endpoint,” she said.
In a video interview, Dr. Hurvitz discussed the study methodology, results, and safety findings, as well as an intriguing observation regarding the effects of treatment on tumor differentiation and immune infiltrates over time.
This study was sponsored by Eli Lilly. Dr. Hurvitz has received renumeration for research and/or travel from Amgen, Bayer, BioMarin, Boehringer Ingelheim, Dignitana, Eli Lilly, Genentech, GSK, Medivation, Merrimack, Novartis, OBI Pharma, Pfizer, Puma Biotechnology, and Roche.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Abemaciclib, both alone and in combination with anastrozole, significantly reduced Ki67 expression vs. anastrozole monotherapy after 2 weeks of treatment in the NeoMONARCH phase II neoadjuvant clinical trial of postmenopausal patients with hormone receptor–positive, HER2-negative early-stage breast cancer.
The findings, given that a change in Ki67 at 2 weeks in neoadjuvant studies appears to predict improved disease-free survival in adjuvant studies, support continued evaluation of the cyclin-dependent kinase-4 (CDK4) inhibitor for the treatment of patients with early-stage breast cancer, Sara Hurvitz, MD, reported at the San Antonio Breast Cancer Symposium.
“In hormone receptor–positive breast cancers, estrogen stimulates D-type cyclins, resulting in increased activity of CDK4 and CDK6, and then phosphorylate RB – the tumor suppressor protein retinoblastoma – which releases the E2F transcription factor,” explained Dr. Hurvitz of the University of California, Los Angeles.
This in turn ultimately leads to cell cycle progression from G1 to S.
“This increased rate of proliferation can be observed in tumor tissue samples by measuring the expression of Ki67. Blocking CDK4 and CDK6 should lead to a decrease in E2F expression, as well as a drop in the cell cycling and a drop in Ki67,” she said, adding that cell cycle arrest may induce senescence, which may also induce a phenotype that’s characterized by an immune cell infiltrate.
Indeed, in study subjects randomized to receive abemaciclib, treatment was shown to induce profound cell cycle arrest defined by decreased Ki67 and E2F targeted proliferation messenger RNAs, and reduction of expression of genes associated with senescence.
“Abemaciclib alone or in combination with anastrozole significantly reduced the Ki67 expression compared to anastrozole alone after 2 weeks of therapy, based on the geometric mean change and complete cell cycle arrest, and the study did meet its primary endpoint,” she said.
In a video interview, Dr. Hurvitz discussed the study methodology, results, and safety findings, as well as an intriguing observation regarding the effects of treatment on tumor differentiation and immune infiltrates over time.
This study was sponsored by Eli Lilly. Dr. Hurvitz has received renumeration for research and/or travel from Amgen, Bayer, BioMarin, Boehringer Ingelheim, Dignitana, Eli Lilly, Genentech, GSK, Medivation, Merrimack, Novartis, OBI Pharma, Pfizer, Puma Biotechnology, and Roche.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SABCS 2016
Study reinforces lenalidomide maintenance in newly diagnosed multiple myeloma
SAN DIEGO – Maintenance therapy with lenalidomide significantly improved progression-free survival in patients of all ages with myeloma, regardless of their risk or response status at the end of induction, Gareth Morgan, MD, PhD, said during an oral session at the annual meeting of the American Society of Hematology.
“The very important point is that maintenance therapy with lenalidomide worked across a range of different risk groups,” said Dr. Morgan, director of the Myeloma Institute at the University of Arkansas for Medical Sciences in Little Rock. “It worked independent of gender, age, [International Staging System] disease stage, and response at baseline,” he added. ‘It also worked irrespective of genetic risk status, which is contrary to what you hear very frequently. All of the curves are consistent with better outcomes if you continue lenalidomide long-term.”
In the overall cohort analysis, half of the patients who received lenalidomide (Revlimid) maintenance were alive and progression-free after 36 months (95% confidence interval, 31-39 months), twice the median PFS of observation-only patients, for a hazard ratio of 0.45 (95% CI, 0.39-0.52; P less than .0001).
This effect held up across numerous subgroups. For example, among 828 transplant-eligible patients, median PFS was 50 months with lenalidomide maintenance and 28 months with observation only (HR, 0.47; P less than .0001). Among 724 transplant-ineligible patients, median PFS was 24 months with lenalidomide and 11 months with observation only (HR, 0.42; P less than .0001), Dr. Morgan reported.
Lenalidomide maintenance did not fully overcome the effects of high-risk cytogenetics but still increased PFS by a median of 10 months, compared with no maintenance (median PFS, 23 months vs. 13 months, respectively; P less than .0001). For patients with standard-risk cytogenetics, median PFS was 44 months on lenalidomide maintenance and 25 months otherwise (P less than .0001).
When patients had minimal residual disease after induction, their median PFS on lenalidomide was 17 months longer if they received maintenance lenalidomide (30 vs. 13 months; P less than .0001). Not surprisingly, the best overall outcomes occurred in MRD-negative patients who received lenalidomide maintenance (median PFS, 44 months, vs. 31 months without lenalidomide; P less than .0001), he said.
Responses also were more likely to deepen over time if patients received lenalidomide maintenance (HR, 1.74; 95% CI, 1.2-2.6; P = .004). “This continued down to about 24 months, which is compatible with conventional response rates,” Dr. Morgan noted.
Safety results reflected prior studies and were unremarkable, he added. “I treat a lot of people with lenalidomide for long periods of time, and the worst thing I usually see is some fatigue.” About one-third of patients developed grade 3-4 neutropenia on lenalidomide maintenance, but less than 5% developed grade 3-4 thrombocytopenia, anemia, deep vein thromboses, or neuropathies. Rates of primary and second malignancies were no worse with maintenance than without it. “All investigators are now in agreement on this finding,” Dr. Morgan emphasized.
The researchers also performed a whole exosome study of 70 paired specimens collected when patients were randomized and again when they relapsed. They found no evidence that lenalidomide induced excess mutations and no significant difference between groups in mutational patterns or genomic copy number variants that alter risk status.
Dr. Morgan and his associates will present overall survival data when the number of events reaches 458, he said. For now, the PFS data reinforce lenalidomide as the standard of care for patients of all ages with newly diagnosed multiple myeloma, he concluded.
The Myeloma XI trial is funded by Cancer Research UK, the Experimental Cancer Medicine Centre, NIHR Clinical Research Network: Cancer, and the University of Leeds. Dr. Morgan disclosed consulting and other relationships with Celgene, the maker of lenalidomide.
SAN DIEGO – Maintenance therapy with lenalidomide significantly improved progression-free survival in patients of all ages with myeloma, regardless of their risk or response status at the end of induction, Gareth Morgan, MD, PhD, said during an oral session at the annual meeting of the American Society of Hematology.
“The very important point is that maintenance therapy with lenalidomide worked across a range of different risk groups,” said Dr. Morgan, director of the Myeloma Institute at the University of Arkansas for Medical Sciences in Little Rock. “It worked independent of gender, age, [International Staging System] disease stage, and response at baseline,” he added. ‘It also worked irrespective of genetic risk status, which is contrary to what you hear very frequently. All of the curves are consistent with better outcomes if you continue lenalidomide long-term.”
In the overall cohort analysis, half of the patients who received lenalidomide (Revlimid) maintenance were alive and progression-free after 36 months (95% confidence interval, 31-39 months), twice the median PFS of observation-only patients, for a hazard ratio of 0.45 (95% CI, 0.39-0.52; P less than .0001).
This effect held up across numerous subgroups. For example, among 828 transplant-eligible patients, median PFS was 50 months with lenalidomide maintenance and 28 months with observation only (HR, 0.47; P less than .0001). Among 724 transplant-ineligible patients, median PFS was 24 months with lenalidomide and 11 months with observation only (HR, 0.42; P less than .0001), Dr. Morgan reported.
Lenalidomide maintenance did not fully overcome the effects of high-risk cytogenetics but still increased PFS by a median of 10 months, compared with no maintenance (median PFS, 23 months vs. 13 months, respectively; P less than .0001). For patients with standard-risk cytogenetics, median PFS was 44 months on lenalidomide maintenance and 25 months otherwise (P less than .0001).
When patients had minimal residual disease after induction, their median PFS on lenalidomide was 17 months longer if they received maintenance lenalidomide (30 vs. 13 months; P less than .0001). Not surprisingly, the best overall outcomes occurred in MRD-negative patients who received lenalidomide maintenance (median PFS, 44 months, vs. 31 months without lenalidomide; P less than .0001), he said.
Responses also were more likely to deepen over time if patients received lenalidomide maintenance (HR, 1.74; 95% CI, 1.2-2.6; P = .004). “This continued down to about 24 months, which is compatible with conventional response rates,” Dr. Morgan noted.
Safety results reflected prior studies and were unremarkable, he added. “I treat a lot of people with lenalidomide for long periods of time, and the worst thing I usually see is some fatigue.” About one-third of patients developed grade 3-4 neutropenia on lenalidomide maintenance, but less than 5% developed grade 3-4 thrombocytopenia, anemia, deep vein thromboses, or neuropathies. Rates of primary and second malignancies were no worse with maintenance than without it. “All investigators are now in agreement on this finding,” Dr. Morgan emphasized.
The researchers also performed a whole exosome study of 70 paired specimens collected when patients were randomized and again when they relapsed. They found no evidence that lenalidomide induced excess mutations and no significant difference between groups in mutational patterns or genomic copy number variants that alter risk status.
Dr. Morgan and his associates will present overall survival data when the number of events reaches 458, he said. For now, the PFS data reinforce lenalidomide as the standard of care for patients of all ages with newly diagnosed multiple myeloma, he concluded.
The Myeloma XI trial is funded by Cancer Research UK, the Experimental Cancer Medicine Centre, NIHR Clinical Research Network: Cancer, and the University of Leeds. Dr. Morgan disclosed consulting and other relationships with Celgene, the maker of lenalidomide.
SAN DIEGO – Maintenance therapy with lenalidomide significantly improved progression-free survival in patients of all ages with myeloma, regardless of their risk or response status at the end of induction, Gareth Morgan, MD, PhD, said during an oral session at the annual meeting of the American Society of Hematology.
“The very important point is that maintenance therapy with lenalidomide worked across a range of different risk groups,” said Dr. Morgan, director of the Myeloma Institute at the University of Arkansas for Medical Sciences in Little Rock. “It worked independent of gender, age, [International Staging System] disease stage, and response at baseline,” he added. ‘It also worked irrespective of genetic risk status, which is contrary to what you hear very frequently. All of the curves are consistent with better outcomes if you continue lenalidomide long-term.”
In the overall cohort analysis, half of the patients who received lenalidomide (Revlimid) maintenance were alive and progression-free after 36 months (95% confidence interval, 31-39 months), twice the median PFS of observation-only patients, for a hazard ratio of 0.45 (95% CI, 0.39-0.52; P less than .0001).
This effect held up across numerous subgroups. For example, among 828 transplant-eligible patients, median PFS was 50 months with lenalidomide maintenance and 28 months with observation only (HR, 0.47; P less than .0001). Among 724 transplant-ineligible patients, median PFS was 24 months with lenalidomide and 11 months with observation only (HR, 0.42; P less than .0001), Dr. Morgan reported.
Lenalidomide maintenance did not fully overcome the effects of high-risk cytogenetics but still increased PFS by a median of 10 months, compared with no maintenance (median PFS, 23 months vs. 13 months, respectively; P less than .0001). For patients with standard-risk cytogenetics, median PFS was 44 months on lenalidomide maintenance and 25 months otherwise (P less than .0001).
When patients had minimal residual disease after induction, their median PFS on lenalidomide was 17 months longer if they received maintenance lenalidomide (30 vs. 13 months; P less than .0001). Not surprisingly, the best overall outcomes occurred in MRD-negative patients who received lenalidomide maintenance (median PFS, 44 months, vs. 31 months without lenalidomide; P less than .0001), he said.
Responses also were more likely to deepen over time if patients received lenalidomide maintenance (HR, 1.74; 95% CI, 1.2-2.6; P = .004). “This continued down to about 24 months, which is compatible with conventional response rates,” Dr. Morgan noted.
Safety results reflected prior studies and were unremarkable, he added. “I treat a lot of people with lenalidomide for long periods of time, and the worst thing I usually see is some fatigue.” About one-third of patients developed grade 3-4 neutropenia on lenalidomide maintenance, but less than 5% developed grade 3-4 thrombocytopenia, anemia, deep vein thromboses, or neuropathies. Rates of primary and second malignancies were no worse with maintenance than without it. “All investigators are now in agreement on this finding,” Dr. Morgan emphasized.
The researchers also performed a whole exosome study of 70 paired specimens collected when patients were randomized and again when they relapsed. They found no evidence that lenalidomide induced excess mutations and no significant difference between groups in mutational patterns or genomic copy number variants that alter risk status.
Dr. Morgan and his associates will present overall survival data when the number of events reaches 458, he said. For now, the PFS data reinforce lenalidomide as the standard of care for patients of all ages with newly diagnosed multiple myeloma, he concluded.
The Myeloma XI trial is funded by Cancer Research UK, the Experimental Cancer Medicine Centre, NIHR Clinical Research Network: Cancer, and the University of Leeds. Dr. Morgan disclosed consulting and other relationships with Celgene, the maker of lenalidomide.
AT ASH 2016
Key clinical point: Maintenance therapy with lenalidomide significantly improved progression-free survival in patients of all ages with myeloma, regardless of response to induction or baseline risk status.
Major finding: Median PFS for patients on lenalidomide maintenance was 36 months (95% confidence interval, 31-39 months), twice that of observation-only patients (hazard ratio, 0.45; P less than .0001).
Data source: A phase III, multicenter, open-label, parallel-group, randomized controlled trial of 1,551 patients with newly diagnosed multiple myeloma.
Disclosures: The Myeloma XI trial is funded by Cancer Research UK, the Experimental Cancer Medicine Centre, NIHR Clinical Research Network: Cancer, and the University of Leeds. Dr. Morgan disclosed consulting and other relationships with Celgene, the maker of lenalidomide.
SHM Commits to Help Hospitals with Judicious Opioid Prescribing
SHM recently enrolled 10 hospitals in the United States in a one-year mentored implementation program to reduce adverse events associated with opioid prescribing. Each hospital will have an assigned physician mentor to guide them through:
- A needs assessment.
- Formal selection of data collection measures.
- Outcome and process data collection on intervention units.
- Design and implementation of key interventions to enhance safety for patients in the hospital who are prescribed opioid medications.
The one-year program includes monthly calls, a site visit with the SHM physician mentor, and a formal assessment of the program’s implementation.
Visit www.hospitalmedicine.org/RADEO to view the online toolkit or download the Reducing Adverse Drug Events Related to Opioids (RADEO) Implementation Guide.
SHM recently enrolled 10 hospitals in the United States in a one-year mentored implementation program to reduce adverse events associated with opioid prescribing. Each hospital will have an assigned physician mentor to guide them through:
- A needs assessment.
- Formal selection of data collection measures.
- Outcome and process data collection on intervention units.
- Design and implementation of key interventions to enhance safety for patients in the hospital who are prescribed opioid medications.
The one-year program includes monthly calls, a site visit with the SHM physician mentor, and a formal assessment of the program’s implementation.
Visit www.hospitalmedicine.org/RADEO to view the online toolkit or download the Reducing Adverse Drug Events Related to Opioids (RADEO) Implementation Guide.
SHM recently enrolled 10 hospitals in the United States in a one-year mentored implementation program to reduce adverse events associated with opioid prescribing. Each hospital will have an assigned physician mentor to guide them through:
- A needs assessment.
- Formal selection of data collection measures.
- Outcome and process data collection on intervention units.
- Design and implementation of key interventions to enhance safety for patients in the hospital who are prescribed opioid medications.
The one-year program includes monthly calls, a site visit with the SHM physician mentor, and a formal assessment of the program’s implementation.
Visit www.hospitalmedicine.org/RADEO to view the online toolkit or download the Reducing Adverse Drug Events Related to Opioids (RADEO) Implementation Guide.
Track List Available for Hospital Medicine 2017
Looking for more? Seize the opportunity to learn from an elite group of experts. Earn additional CME credits, broaden your skills, and fine-tune your practice. Immerse yourself in a day of learning by enrolling in one six pre-courses on May 1. Visit www.hospitalmedicine2017.org/precourse to view course topics, agendas, faculty, and more.
Looking for more? Seize the opportunity to learn from an elite group of experts. Earn additional CME credits, broaden your skills, and fine-tune your practice. Immerse yourself in a day of learning by enrolling in one six pre-courses on May 1. Visit www.hospitalmedicine2017.org/precourse to view course topics, agendas, faculty, and more.
Looking for more? Seize the opportunity to learn from an elite group of experts. Earn additional CME credits, broaden your skills, and fine-tune your practice. Immerse yourself in a day of learning by enrolling in one six pre-courses on May 1. Visit www.hospitalmedicine2017.org/precourse to view course topics, agendas, faculty, and more.
Potential treatment for cGVHD after steroid failure

2016 ASH Annual Meeting
SAN DIEGO—Ibrutinib, a Bruton’s tyrosine kinase inhibitor approved to treat chronic lymphocytic leukemia and other hematologic diseases, appears to provide relief for patients suffering from chronic graft-versus-host disease (cGVHD) after failing corticosteroid therapy.
At present, no approved therapy exists for these patients. Ibrutinib reduced the severity of cGVHD in preclinical models and has been used successfully in the post-allogeneic transplant setting.
The US Food and Drug Administration granted ibrutinib breakthrough therapy and orphan drug designations as a potential treatment for cGVHD.
David Miklos, MD, of Stanford University in California, explained at the 2016 ASH Annual Meeting that, in cGVHD, healthy B cells have been corrupted to produce self-reactive antibody complexes, and the T cells are killing healthy tissues and cells.
This destructive process involves the Bruton’s tyrosine kinase molecule, which can be inhibited and thereby block some of the downstream cGVHD pathogenesis.
“And to this aim, we went about testing the benefits of ibrutinib in the treatment of steroid-refractory chronic graft-versus-host disease,” Dr Miklos said.
In phase 1 of the study, investigators tested the 420 mg oral once-daily dose. They found no dose-limiting toxicities.
“So this dose was carried forward into the phase 2 study,” Dr Miklos said.
He presented results of the phase 2 study at the meeting as a late-breaking abstract (LBA-3).
Study design
Patients were eligible for the study if they had steroid-dependent or -refractory cGVHD. They had to have 3 or fewer prior treatments, and they could continue other systemic immunosuppression if they were using it.
They had to have erythematous rash on more than 25% of their body surface or a total mouth score of more than 4 as defined by National Institutes of Health (NIH) criteria.
Patients with cGVHD had to have failed frontline therapy.
They were treated with the phase 1 dose until progression of cGVHD or unacceptable toxicity.
The primary endpoint was cGVHD response per NIH 2005 response criteria.
Secondary endpoints included rate of sustained response, change in Lee cGVHD symptom scale, changes in corticosteroid requirement over time, and safety endpoints.
Investigators enrolled 42 patients, the first of whom was dosed in July 2014.
Patient demographics
Patients were a median age of 56 (range, 19–74), and 52% were male.
The median time from allogeneic transplant to the diagnosis of cGVHD was 7.6 months (range, 1.5–76.0), and the median time from initial cGVHD diagnosis to start of ibrutinib therapy was 13.7 months (range, 1.1–63.2).
Most patients had mouth (86%), skin (81%), or gastrointestinal (33%) cGVHD involvement.
And most patients had received matched (88%), unrelated (60%), nonmyeloablative (57%) peripheral blood stem cell (88%) transplants.
“This was a heavily treated patient population,” Dr Miklos said.
They had received a median of 2 (range, 1–3) prior regimens, with a median prednisone dose at enrollment of 0.3 mg/kg/day.
Prior cGVHD therapies included corticosteroids (100%), tacrolimus (50%), extracorporeal photopheresis (33%), rituximab (26%), mycophenolate mofetil (24%), cyclosporine (19%), sirolimus (17%), and other immunosuppressants (5%).
Results
The overall response rate was 67%, including 9 complete responses and 19 partial responses. Seventy-nine percent responded by the first assessment, and 71% of the 28 responders had a sustained cGVHD response of at least 5 months.
Investigators observed responses across multiple organs. Eighty percent (20/25) of patients with at least 2 involved organs at baseline responded in at least 2 organs, and 56% (5/9) of patients with 3 or more involved organs at baseline responded in at least 3 organs.
“These responses seen across all organs and in multiple organs suggest that the ibrutinib is actually targeting the underlying process of chronic GVHD and not simply masking the symptoms of chronic GVHD,” Dr Miklos noted.
Median corticosteroid use decreased throughout the ibrutinib treatment period. Twenty-six patients (62%) reduced steroid doses to less than 0.15/mg/kg/day while on ibrutinib.
Five responders discontinued all corticosteroid treatment.
Dr Miklos pointed out that baseline steroid dose did not vary between those patients who had responses and those who did not.
And ibrutinib produced clinically meaning improvement in the Lee symptom scale score among patients who responded.
Discontinuation and toxicity
At a median follow-up of 14 months, 12 patients were still on ibrutinib therapy.
“Only 5 patients discontinued for the progression of chronic GVHD,” Dr Miklos noted.
Other reasons for discontinuation included adverse events (AEs, n=14), patient decision (n=6), investigator decision (n=2), recurrence or progression of original malignancy (n=2), and noncompliance with study drug (n=1).
“The AE profile largely reflects what has been seen with ibrutinib use in the patients being treated for malignancies,” Dr Miklos said. “They also reflect adverse events that we see in patients receiving corticosteroids for the treatment of chronic GVHD.”
Treatment-emergent AEs occurring in more than 15% of patients included fatigue, diarrhea, muscle spasms, nausea, bruising, upper respiratory tract infection, pneumonia, pyrexia, headache, and fall.
Serious AEs occurred in 22 patients (52%), including pneumonia (n=6), septic shock (n=2), and pyrexia (n=2).
Two patients died while on study due to multilobular pneumonia and bronchopulmonary aspergillosis.
Exploratory endpoints
Investigators measured plasma levels of various factors following ibrutinib therapy through the first 90 days. Proinflammatory, chemotactic, and fibrotic factors decreased significantly while patients were on ibrutinib.
“This indicates that the cellular inflammation, the immune recruitment, and the fibrosis at the root of chronic GVHD was improving,” Dr Mikos said.
These factors included IFNγ, TNFα, IP-10, and CXCL9—biomarkers associated with cGVHD.
“We believe the efficacy of ibrutinib in this population supports further study in frontline treatment of chronic GVHD in a randomized, double-blinded study,” Dr Miklos concluded.
The current study was sponsored by Pharmacyclics, Inc. ![]()

2016 ASH Annual Meeting
SAN DIEGO—Ibrutinib, a Bruton’s tyrosine kinase inhibitor approved to treat chronic lymphocytic leukemia and other hematologic diseases, appears to provide relief for patients suffering from chronic graft-versus-host disease (cGVHD) after failing corticosteroid therapy.
At present, no approved therapy exists for these patients. Ibrutinib reduced the severity of cGVHD in preclinical models and has been used successfully in the post-allogeneic transplant setting.
The US Food and Drug Administration granted ibrutinib breakthrough therapy and orphan drug designations as a potential treatment for cGVHD.
David Miklos, MD, of Stanford University in California, explained at the 2016 ASH Annual Meeting that, in cGVHD, healthy B cells have been corrupted to produce self-reactive antibody complexes, and the T cells are killing healthy tissues and cells.
This destructive process involves the Bruton’s tyrosine kinase molecule, which can be inhibited and thereby block some of the downstream cGVHD pathogenesis.
“And to this aim, we went about testing the benefits of ibrutinib in the treatment of steroid-refractory chronic graft-versus-host disease,” Dr Miklos said.
In phase 1 of the study, investigators tested the 420 mg oral once-daily dose. They found no dose-limiting toxicities.
“So this dose was carried forward into the phase 2 study,” Dr Miklos said.
He presented results of the phase 2 study at the meeting as a late-breaking abstract (LBA-3).
Study design
Patients were eligible for the study if they had steroid-dependent or -refractory cGVHD. They had to have 3 or fewer prior treatments, and they could continue other systemic immunosuppression if they were using it.
They had to have erythematous rash on more than 25% of their body surface or a total mouth score of more than 4 as defined by National Institutes of Health (NIH) criteria.
Patients with cGVHD had to have failed frontline therapy.
They were treated with the phase 1 dose until progression of cGVHD or unacceptable toxicity.
The primary endpoint was cGVHD response per NIH 2005 response criteria.
Secondary endpoints included rate of sustained response, change in Lee cGVHD symptom scale, changes in corticosteroid requirement over time, and safety endpoints.
Investigators enrolled 42 patients, the first of whom was dosed in July 2014.
Patient demographics
Patients were a median age of 56 (range, 19–74), and 52% were male.
The median time from allogeneic transplant to the diagnosis of cGVHD was 7.6 months (range, 1.5–76.0), and the median time from initial cGVHD diagnosis to start of ibrutinib therapy was 13.7 months (range, 1.1–63.2).
Most patients had mouth (86%), skin (81%), or gastrointestinal (33%) cGVHD involvement.
And most patients had received matched (88%), unrelated (60%), nonmyeloablative (57%) peripheral blood stem cell (88%) transplants.
“This was a heavily treated patient population,” Dr Miklos said.
They had received a median of 2 (range, 1–3) prior regimens, with a median prednisone dose at enrollment of 0.3 mg/kg/day.
Prior cGVHD therapies included corticosteroids (100%), tacrolimus (50%), extracorporeal photopheresis (33%), rituximab (26%), mycophenolate mofetil (24%), cyclosporine (19%), sirolimus (17%), and other immunosuppressants (5%).
Results
The overall response rate was 67%, including 9 complete responses and 19 partial responses. Seventy-nine percent responded by the first assessment, and 71% of the 28 responders had a sustained cGVHD response of at least 5 months.
Investigators observed responses across multiple organs. Eighty percent (20/25) of patients with at least 2 involved organs at baseline responded in at least 2 organs, and 56% (5/9) of patients with 3 or more involved organs at baseline responded in at least 3 organs.
“These responses seen across all organs and in multiple organs suggest that the ibrutinib is actually targeting the underlying process of chronic GVHD and not simply masking the symptoms of chronic GVHD,” Dr Miklos noted.
Median corticosteroid use decreased throughout the ibrutinib treatment period. Twenty-six patients (62%) reduced steroid doses to less than 0.15/mg/kg/day while on ibrutinib.
Five responders discontinued all corticosteroid treatment.
Dr Miklos pointed out that baseline steroid dose did not vary between those patients who had responses and those who did not.
And ibrutinib produced clinically meaning improvement in the Lee symptom scale score among patients who responded.
Discontinuation and toxicity
At a median follow-up of 14 months, 12 patients were still on ibrutinib therapy.
“Only 5 patients discontinued for the progression of chronic GVHD,” Dr Miklos noted.
Other reasons for discontinuation included adverse events (AEs, n=14), patient decision (n=6), investigator decision (n=2), recurrence or progression of original malignancy (n=2), and noncompliance with study drug (n=1).
“The AE profile largely reflects what has been seen with ibrutinib use in the patients being treated for malignancies,” Dr Miklos said. “They also reflect adverse events that we see in patients receiving corticosteroids for the treatment of chronic GVHD.”
Treatment-emergent AEs occurring in more than 15% of patients included fatigue, diarrhea, muscle spasms, nausea, bruising, upper respiratory tract infection, pneumonia, pyrexia, headache, and fall.
Serious AEs occurred in 22 patients (52%), including pneumonia (n=6), septic shock (n=2), and pyrexia (n=2).
Two patients died while on study due to multilobular pneumonia and bronchopulmonary aspergillosis.
Exploratory endpoints
Investigators measured plasma levels of various factors following ibrutinib therapy through the first 90 days. Proinflammatory, chemotactic, and fibrotic factors decreased significantly while patients were on ibrutinib.
“This indicates that the cellular inflammation, the immune recruitment, and the fibrosis at the root of chronic GVHD was improving,” Dr Mikos said.
These factors included IFNγ, TNFα, IP-10, and CXCL9—biomarkers associated with cGVHD.
“We believe the efficacy of ibrutinib in this population supports further study in frontline treatment of chronic GVHD in a randomized, double-blinded study,” Dr Miklos concluded.
The current study was sponsored by Pharmacyclics, Inc. ![]()

2016 ASH Annual Meeting
SAN DIEGO—Ibrutinib, a Bruton’s tyrosine kinase inhibitor approved to treat chronic lymphocytic leukemia and other hematologic diseases, appears to provide relief for patients suffering from chronic graft-versus-host disease (cGVHD) after failing corticosteroid therapy.
At present, no approved therapy exists for these patients. Ibrutinib reduced the severity of cGVHD in preclinical models and has been used successfully in the post-allogeneic transplant setting.
The US Food and Drug Administration granted ibrutinib breakthrough therapy and orphan drug designations as a potential treatment for cGVHD.
David Miklos, MD, of Stanford University in California, explained at the 2016 ASH Annual Meeting that, in cGVHD, healthy B cells have been corrupted to produce self-reactive antibody complexes, and the T cells are killing healthy tissues and cells.
This destructive process involves the Bruton’s tyrosine kinase molecule, which can be inhibited and thereby block some of the downstream cGVHD pathogenesis.
“And to this aim, we went about testing the benefits of ibrutinib in the treatment of steroid-refractory chronic graft-versus-host disease,” Dr Miklos said.
In phase 1 of the study, investigators tested the 420 mg oral once-daily dose. They found no dose-limiting toxicities.
“So this dose was carried forward into the phase 2 study,” Dr Miklos said.
He presented results of the phase 2 study at the meeting as a late-breaking abstract (LBA-3).
Study design
Patients were eligible for the study if they had steroid-dependent or -refractory cGVHD. They had to have 3 or fewer prior treatments, and they could continue other systemic immunosuppression if they were using it.
They had to have erythematous rash on more than 25% of their body surface or a total mouth score of more than 4 as defined by National Institutes of Health (NIH) criteria.
Patients with cGVHD had to have failed frontline therapy.
They were treated with the phase 1 dose until progression of cGVHD or unacceptable toxicity.
The primary endpoint was cGVHD response per NIH 2005 response criteria.
Secondary endpoints included rate of sustained response, change in Lee cGVHD symptom scale, changes in corticosteroid requirement over time, and safety endpoints.
Investigators enrolled 42 patients, the first of whom was dosed in July 2014.
Patient demographics
Patients were a median age of 56 (range, 19–74), and 52% were male.
The median time from allogeneic transplant to the diagnosis of cGVHD was 7.6 months (range, 1.5–76.0), and the median time from initial cGVHD diagnosis to start of ibrutinib therapy was 13.7 months (range, 1.1–63.2).
Most patients had mouth (86%), skin (81%), or gastrointestinal (33%) cGVHD involvement.
And most patients had received matched (88%), unrelated (60%), nonmyeloablative (57%) peripheral blood stem cell (88%) transplants.
“This was a heavily treated patient population,” Dr Miklos said.
They had received a median of 2 (range, 1–3) prior regimens, with a median prednisone dose at enrollment of 0.3 mg/kg/day.
Prior cGVHD therapies included corticosteroids (100%), tacrolimus (50%), extracorporeal photopheresis (33%), rituximab (26%), mycophenolate mofetil (24%), cyclosporine (19%), sirolimus (17%), and other immunosuppressants (5%).
Results
The overall response rate was 67%, including 9 complete responses and 19 partial responses. Seventy-nine percent responded by the first assessment, and 71% of the 28 responders had a sustained cGVHD response of at least 5 months.
Investigators observed responses across multiple organs. Eighty percent (20/25) of patients with at least 2 involved organs at baseline responded in at least 2 organs, and 56% (5/9) of patients with 3 or more involved organs at baseline responded in at least 3 organs.
“These responses seen across all organs and in multiple organs suggest that the ibrutinib is actually targeting the underlying process of chronic GVHD and not simply masking the symptoms of chronic GVHD,” Dr Miklos noted.
Median corticosteroid use decreased throughout the ibrutinib treatment period. Twenty-six patients (62%) reduced steroid doses to less than 0.15/mg/kg/day while on ibrutinib.
Five responders discontinued all corticosteroid treatment.
Dr Miklos pointed out that baseline steroid dose did not vary between those patients who had responses and those who did not.
And ibrutinib produced clinically meaning improvement in the Lee symptom scale score among patients who responded.
Discontinuation and toxicity
At a median follow-up of 14 months, 12 patients were still on ibrutinib therapy.
“Only 5 patients discontinued for the progression of chronic GVHD,” Dr Miklos noted.
Other reasons for discontinuation included adverse events (AEs, n=14), patient decision (n=6), investigator decision (n=2), recurrence or progression of original malignancy (n=2), and noncompliance with study drug (n=1).
“The AE profile largely reflects what has been seen with ibrutinib use in the patients being treated for malignancies,” Dr Miklos said. “They also reflect adverse events that we see in patients receiving corticosteroids for the treatment of chronic GVHD.”
Treatment-emergent AEs occurring in more than 15% of patients included fatigue, diarrhea, muscle spasms, nausea, bruising, upper respiratory tract infection, pneumonia, pyrexia, headache, and fall.
Serious AEs occurred in 22 patients (52%), including pneumonia (n=6), septic shock (n=2), and pyrexia (n=2).
Two patients died while on study due to multilobular pneumonia and bronchopulmonary aspergillosis.
Exploratory endpoints
Investigators measured plasma levels of various factors following ibrutinib therapy through the first 90 days. Proinflammatory, chemotactic, and fibrotic factors decreased significantly while patients were on ibrutinib.
“This indicates that the cellular inflammation, the immune recruitment, and the fibrosis at the root of chronic GVHD was improving,” Dr Mikos said.
These factors included IFNγ, TNFα, IP-10, and CXCL9—biomarkers associated with cGVHD.
“We believe the efficacy of ibrutinib in this population supports further study in frontline treatment of chronic GVHD in a randomized, double-blinded study,” Dr Miklos concluded.
The current study was sponsored by Pharmacyclics, Inc. ![]()
Aromatase inhibitor effect on endothelial function may lead to CVD
SAN ANTONIO – Aromatase inhibitors, a mainstay of therapy in postmenopausal women with operable hormone receptor–positive breast cancers, are associated with reductions in endothelial function that could contribute to the development of cardiovascular disease, independent of the duration of therapy, investigators have found.
In a cross-sectional study examining endothelial function among postmenopausal women with locally advanced breast cancer on an aromatase inhibitor (AI), there were trends toward reduction in large and small artery elasticity and a significant decrement in vascular tone, compared with the vessels of healthy controls, reported Anne Blaes, MD, from the Masonic Cancer Center at the University of Minnesota in Minneapolis.
“Other studies have suggested that the cardiac risk from aromatase inhibitors is increased further in those with a previous diagnosis of cardiovascular disease. In this study we did not include this patient population, but I really think further work needs to be done in this area,” she said at the San Antonio Breast Cancer Symposium.
Her group’s findings suggest that prospective breast cancer trials need biomarkers to predict cardiovascular risk for patients who are on chronic AI therapy, she said.
CV incidence modest, deaths lows
The incidence rates of cardiovascular disease in clinical trials of adjuvant AI therapy have ranged from 3% to 17%, although the incidence of death from cardiovascular disease was relatively low in these trials, on the order of 1%-2%. Data on cardiovascular risk factors, however, were inconsistently collected across the various studies, Dr. Blaes noted.
“More recently, a lot of discussion has gone on about both the use of prolonged endocrine therapy using aromatase inhibitors – whether to consider 5 or 10 years – and in addition, as our population is aging, competing risks for mortality, whether that’s breast cancer or cardiovascular risk,” she said.
The investigators examined endothelial function in 36 postmenopausal women with locally advanced, operable breast cancer treated with curative intent with adjuvant AI therapy, and compared results with those of 25 healthy postmenopausal volunteers, five of whom were excluded from the final analysis due to prior use of exogenous estrogen.
About half of the patients had received chemotherapy, and two-thirds had received radiation therapy. The AIs used for most patients were anastrozole (Arimidex) and letrozole (Femara). Seven of the 36 cases had previously received tamoxifen.
The authors measured endothelial function using the EndoPAT (Itamar Medical) system that measures peripheral arterial tone (PAT) to identify reactive hyperemia. Endothelial dysfunction measured this way has been associated with an increased risk of cardiac adverse events independent of the Framingham Risk Score, Dr. Blaes said.
The participants underwent biomarker analysis and pulse wave analysis using a cardiovascular profiling system, and pulse contour analysis using the Endo-PAT2000 system. The investigators then compared biomarkers and functional test markers between cases and controls using T-tests and Wilcoxon Rank-Sum tests.
Biomarkers included inflammatory markers (high-sensitivity C-reactive protein, white blood cell count, interleukin 6), markers of hemostasis (fibrinogen, d-dimer, plasminogen-activator inhibitor-1, tissue-type plasminogen activator), and endothelial function markers (von Willebrand factor, circulating endothelial cells, soluble vascular cell adhesion molecule-1, and others).
They measured large-artery elasticity (LAE), small-artery elasticity (SAE), and the EndoPAT ratio, or reactive hyperemia index (RHI), the post-to-pre occlusion PAT signal ratio in the occluded side, normalized to the control side and further corrected for baseline vascular tone. An RHI score above 1.67 is considered normal, and a score of 1.67 or below is considered abnormal.
They found that both LAE and SAE trended toward significantly worse vascular tone in cases, compared with controls, but the differences were not statistically significant. The EndoPAT ratio, however, was significantly worse among cases, at 0.8, compared with 2.6 for controls, a difference that remained significant after controlling for systolic blood pressure (P less than .0001).
Hemostatic and endothelial biomarkers were significantly elevated in cases, compared with controls, but there were no significant differences in inflammatory markers.
When the investigators looked at the association between vascular function and cancer treatment characteristics, they found no differences in the use of chemotherapy, radiation, or left vs. right breast treated.
The use of anastrozole was associated with a significant reduction in LAE, compared with either letrozole or exemestane (P = .03). There was no association between duration of AI therapy and EndoPAT ratio.
Estradiol levels implicated
Not surprisingly, women on endocrine therapy in the study had significantly lower levels of estradiol than controls. Estradiol appears to be important for regulating healthy endothelial function, commented Patricia A. Ganz, MD, of the Jonsson Comprehensive Cancer Center at the University of California, Los Angeles, who was the invited discussant.
“I think these are very provocative, hypothesis-generating findings, and I think they really fit what we expect the physiology should be in terms of endothelial function, even within this postmenopausal group of women where we’re looking at two discrete groups in terms of the estradiol level,” she said.
The study was funded by Building Interdisciplinary Research Careers in Women’s Health and a Masonic Scholar Award. Dr. Blaes and Dr. Ganz reported no relevant conflicts of interest.
SAN ANTONIO – Aromatase inhibitors, a mainstay of therapy in postmenopausal women with operable hormone receptor–positive breast cancers, are associated with reductions in endothelial function that could contribute to the development of cardiovascular disease, independent of the duration of therapy, investigators have found.
In a cross-sectional study examining endothelial function among postmenopausal women with locally advanced breast cancer on an aromatase inhibitor (AI), there were trends toward reduction in large and small artery elasticity and a significant decrement in vascular tone, compared with the vessels of healthy controls, reported Anne Blaes, MD, from the Masonic Cancer Center at the University of Minnesota in Minneapolis.
“Other studies have suggested that the cardiac risk from aromatase inhibitors is increased further in those with a previous diagnosis of cardiovascular disease. In this study we did not include this patient population, but I really think further work needs to be done in this area,” she said at the San Antonio Breast Cancer Symposium.
Her group’s findings suggest that prospective breast cancer trials need biomarkers to predict cardiovascular risk for patients who are on chronic AI therapy, she said.
CV incidence modest, deaths lows
The incidence rates of cardiovascular disease in clinical trials of adjuvant AI therapy have ranged from 3% to 17%, although the incidence of death from cardiovascular disease was relatively low in these trials, on the order of 1%-2%. Data on cardiovascular risk factors, however, were inconsistently collected across the various studies, Dr. Blaes noted.
“More recently, a lot of discussion has gone on about both the use of prolonged endocrine therapy using aromatase inhibitors – whether to consider 5 or 10 years – and in addition, as our population is aging, competing risks for mortality, whether that’s breast cancer or cardiovascular risk,” she said.
The investigators examined endothelial function in 36 postmenopausal women with locally advanced, operable breast cancer treated with curative intent with adjuvant AI therapy, and compared results with those of 25 healthy postmenopausal volunteers, five of whom were excluded from the final analysis due to prior use of exogenous estrogen.
About half of the patients had received chemotherapy, and two-thirds had received radiation therapy. The AIs used for most patients were anastrozole (Arimidex) and letrozole (Femara). Seven of the 36 cases had previously received tamoxifen.
The authors measured endothelial function using the EndoPAT (Itamar Medical) system that measures peripheral arterial tone (PAT) to identify reactive hyperemia. Endothelial dysfunction measured this way has been associated with an increased risk of cardiac adverse events independent of the Framingham Risk Score, Dr. Blaes said.
The participants underwent biomarker analysis and pulse wave analysis using a cardiovascular profiling system, and pulse contour analysis using the Endo-PAT2000 system. The investigators then compared biomarkers and functional test markers between cases and controls using T-tests and Wilcoxon Rank-Sum tests.
Biomarkers included inflammatory markers (high-sensitivity C-reactive protein, white blood cell count, interleukin 6), markers of hemostasis (fibrinogen, d-dimer, plasminogen-activator inhibitor-1, tissue-type plasminogen activator), and endothelial function markers (von Willebrand factor, circulating endothelial cells, soluble vascular cell adhesion molecule-1, and others).
They measured large-artery elasticity (LAE), small-artery elasticity (SAE), and the EndoPAT ratio, or reactive hyperemia index (RHI), the post-to-pre occlusion PAT signal ratio in the occluded side, normalized to the control side and further corrected for baseline vascular tone. An RHI score above 1.67 is considered normal, and a score of 1.67 or below is considered abnormal.
They found that both LAE and SAE trended toward significantly worse vascular tone in cases, compared with controls, but the differences were not statistically significant. The EndoPAT ratio, however, was significantly worse among cases, at 0.8, compared with 2.6 for controls, a difference that remained significant after controlling for systolic blood pressure (P less than .0001).
Hemostatic and endothelial biomarkers were significantly elevated in cases, compared with controls, but there were no significant differences in inflammatory markers.
When the investigators looked at the association between vascular function and cancer treatment characteristics, they found no differences in the use of chemotherapy, radiation, or left vs. right breast treated.
The use of anastrozole was associated with a significant reduction in LAE, compared with either letrozole or exemestane (P = .03). There was no association between duration of AI therapy and EndoPAT ratio.
Estradiol levels implicated
Not surprisingly, women on endocrine therapy in the study had significantly lower levels of estradiol than controls. Estradiol appears to be important for regulating healthy endothelial function, commented Patricia A. Ganz, MD, of the Jonsson Comprehensive Cancer Center at the University of California, Los Angeles, who was the invited discussant.
“I think these are very provocative, hypothesis-generating findings, and I think they really fit what we expect the physiology should be in terms of endothelial function, even within this postmenopausal group of women where we’re looking at two discrete groups in terms of the estradiol level,” she said.
The study was funded by Building Interdisciplinary Research Careers in Women’s Health and a Masonic Scholar Award. Dr. Blaes and Dr. Ganz reported no relevant conflicts of interest.
SAN ANTONIO – Aromatase inhibitors, a mainstay of therapy in postmenopausal women with operable hormone receptor–positive breast cancers, are associated with reductions in endothelial function that could contribute to the development of cardiovascular disease, independent of the duration of therapy, investigators have found.
In a cross-sectional study examining endothelial function among postmenopausal women with locally advanced breast cancer on an aromatase inhibitor (AI), there were trends toward reduction in large and small artery elasticity and a significant decrement in vascular tone, compared with the vessels of healthy controls, reported Anne Blaes, MD, from the Masonic Cancer Center at the University of Minnesota in Minneapolis.
“Other studies have suggested that the cardiac risk from aromatase inhibitors is increased further in those with a previous diagnosis of cardiovascular disease. In this study we did not include this patient population, but I really think further work needs to be done in this area,” she said at the San Antonio Breast Cancer Symposium.
Her group’s findings suggest that prospective breast cancer trials need biomarkers to predict cardiovascular risk for patients who are on chronic AI therapy, she said.
CV incidence modest, deaths lows
The incidence rates of cardiovascular disease in clinical trials of adjuvant AI therapy have ranged from 3% to 17%, although the incidence of death from cardiovascular disease was relatively low in these trials, on the order of 1%-2%. Data on cardiovascular risk factors, however, were inconsistently collected across the various studies, Dr. Blaes noted.
“More recently, a lot of discussion has gone on about both the use of prolonged endocrine therapy using aromatase inhibitors – whether to consider 5 or 10 years – and in addition, as our population is aging, competing risks for mortality, whether that’s breast cancer or cardiovascular risk,” she said.
The investigators examined endothelial function in 36 postmenopausal women with locally advanced, operable breast cancer treated with curative intent with adjuvant AI therapy, and compared results with those of 25 healthy postmenopausal volunteers, five of whom were excluded from the final analysis due to prior use of exogenous estrogen.
About half of the patients had received chemotherapy, and two-thirds had received radiation therapy. The AIs used for most patients were anastrozole (Arimidex) and letrozole (Femara). Seven of the 36 cases had previously received tamoxifen.
The authors measured endothelial function using the EndoPAT (Itamar Medical) system that measures peripheral arterial tone (PAT) to identify reactive hyperemia. Endothelial dysfunction measured this way has been associated with an increased risk of cardiac adverse events independent of the Framingham Risk Score, Dr. Blaes said.
The participants underwent biomarker analysis and pulse wave analysis using a cardiovascular profiling system, and pulse contour analysis using the Endo-PAT2000 system. The investigators then compared biomarkers and functional test markers between cases and controls using T-tests and Wilcoxon Rank-Sum tests.
Biomarkers included inflammatory markers (high-sensitivity C-reactive protein, white blood cell count, interleukin 6), markers of hemostasis (fibrinogen, d-dimer, plasminogen-activator inhibitor-1, tissue-type plasminogen activator), and endothelial function markers (von Willebrand factor, circulating endothelial cells, soluble vascular cell adhesion molecule-1, and others).
They measured large-artery elasticity (LAE), small-artery elasticity (SAE), and the EndoPAT ratio, or reactive hyperemia index (RHI), the post-to-pre occlusion PAT signal ratio in the occluded side, normalized to the control side and further corrected for baseline vascular tone. An RHI score above 1.67 is considered normal, and a score of 1.67 or below is considered abnormal.
They found that both LAE and SAE trended toward significantly worse vascular tone in cases, compared with controls, but the differences were not statistically significant. The EndoPAT ratio, however, was significantly worse among cases, at 0.8, compared with 2.6 for controls, a difference that remained significant after controlling for systolic blood pressure (P less than .0001).
Hemostatic and endothelial biomarkers were significantly elevated in cases, compared with controls, but there were no significant differences in inflammatory markers.
When the investigators looked at the association between vascular function and cancer treatment characteristics, they found no differences in the use of chemotherapy, radiation, or left vs. right breast treated.
The use of anastrozole was associated with a significant reduction in LAE, compared with either letrozole or exemestane (P = .03). There was no association between duration of AI therapy and EndoPAT ratio.
Estradiol levels implicated
Not surprisingly, women on endocrine therapy in the study had significantly lower levels of estradiol than controls. Estradiol appears to be important for regulating healthy endothelial function, commented Patricia A. Ganz, MD, of the Jonsson Comprehensive Cancer Center at the University of California, Los Angeles, who was the invited discussant.
“I think these are very provocative, hypothesis-generating findings, and I think they really fit what we expect the physiology should be in terms of endothelial function, even within this postmenopausal group of women where we’re looking at two discrete groups in terms of the estradiol level,” she said.
The study was funded by Building Interdisciplinary Research Careers in Women’s Health and a Masonic Scholar Award. Dr. Blaes and Dr. Ganz reported no relevant conflicts of interest.
AT SABCS 2016
Key clinical point: Aromatase inhibitors appear to have a decremental effect on vascular endothelial function, which could contribute to cardiovascular disease.
Major finding: Postmenopausal breast cancer survivors had significantly worse endothelial function than healthy postmenopausal controls, as measured by the EndoPAT ratio.
Data source: Case-control study with 36 postmenopausal breast cancer survivors on aromatase inhibitors and 25 healthy controls.
Disclosures: The study was funded by Building Interdisciplinary Research Careers in Women’s Health and a Masonic Scholar Award. Dr. Blaes and Dr. Ganz reported no relevant conflicts of interest.
TauRx still rooting for its methylene blue AD drug, despite controversial study results
SAN DIEGO – An anti-tau compound that has stirred scientific controversy for 8 years will continue along its developmental pathway at a much lower dose, despite yet another study that has Alzheimer’s researchers scratching their heads.
The drug, dubbed LMTM, is a derivative of the dye methylene blue. Its most recent phase III study, reported at the Clinical Trials on Alzheimer’s Disease conference, found that 100 mg twice a day conferred no cognitive or functional benefit upon patients with mild AD, compared with a control dose of 4 mg.
Some significant differences, however, did emerge in two prespecified subanalyses of the 4-mg control group. Patients who took the low dose, intended to be a placebo comparator, did better than those on the high dose – but only if they were not taking any standard symptomatic AD medications.
Based on these findings, TauRx, which is developing LMTM, will abandon the 100-mg dose and refocus on the 4-mg dose, said Claude Wischik, MD, chairman and chief executive officer of the Singapore-based company.
“I think it looks effective and there’s no advantage to going to a higher dose,” Dr. Wischik said in an interview. “The 100-mg dose doesn’t offer anything above the 4-mg dose, and we saw more dropouts in the higher-dose group. We will go forward with a new trial using 4 mg.”
The new commitment to 4 mg turns LMTM’s prior development trajectory on its head, as nothing lower than 75 mg has been investigated in a phase III study. The 4-mg control dose was used as a placebo stand-in, since LMTM colors urine blue or green. The low dose was considered biologically inactive and used to maintain the study blind.
Dr. Wischik has been investigating LMTM as a tau anti-aggregant for 10 years, first publicly reporting clinical data in 2008. LMTM has never posted significant cognitive or functional benefits in any primary analysis. Instead, it has moved forward based on a series of subanalyses that showed significant or near-significant benefits in smaller, meticulously constructed subgroups – conclusions that critics have called questionable at best. The most recent of these examined the drug’s effect in patients with mild to moderate disease and was presented last July at the Alzheimer’s Association’s International Conference (AAIC).
That study also didn’t meet its primary endpoints in the overall cohort of 891 patients, but TauRx promoted it as “promising,” based on a subgroup analysis of the 15% of patients who were not taking memantine or cholinesterase inhibitors.
Among these patients, those taking 75 mg twice daily declined 6 points less on the Alzheimer’s Disease Assessment Scale-cognitive subscale (ADAS-cog) than those taking 4 mg. Those taking 125 mg twice daily declined 6 points less than the 4-mg group. On the Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL), patients taking 75 mg twice daily scored 6.5 points higher than did the placebo group, indicating better function, and those taking 125 mg twice daily scored 7 points higher than did the placebo group.
At AAIC, researchers suggested that the monotherapy groups could have had a less aggressive disease course, or might not have had Alzheimer’s disease at all. Others complained about the unorthodox grouping of control patients in the subgroup analysis.
It was after digesting these data that TauRx investigators changed the statistical analysis of the current study, then in its final months, from a randomized trial to a cohort analysis. This was done before data lockdown, but it was still a dramatic shift from the original study design.
“The primary analysis was changed to essentially analyze this as a cohort study,” said Lon Schneider, MD, who presented the results at CTAD. “The comparisons of interest were patients taking 100 mg twice a day who were not on [symptomatic treatment], compared to the original control group of 4 mg. The other comparison was the 4-mg group not on cholinesterase inhibitors to the 4-mg group that was on them.”
The 18-month trial randomized 800 patients with mild AD to 100 mg LMTM twice daily or to 4 mg twice daily. Patients were drawn from two global regions: Canada and the United States, and eastern Europe and Australia.
The study group was a typical one, with a mean age of 70 years and a mean Mini Mental State Exam score of 22. The mean ADAS-cog score was 17. Most (80%) were taking a cholinesterase inhibitor, memantine, or both; 20% were naive to these medications.
Primary endpoints were the ADAS-cog11, ADCS-ADL, and left ventricular volume. Secondary outcomes included the Mini Mental State Exam and Neuropsychiatric Index.
In the primary analysis as the trial was created and conducted, LMTM 100 mg/twice daily did not confer any benefit, compared with the control 4-mg dose. The decline curves were virtually superimposable in the ADAS-cog score, ADCS-ADL score, and in loss of left ventricular volume.
This same nonsignificant pattern occurred in all the secondary endpoints, which Dr. Schneider did not show.
The cohort analyses stratified patients according to whether they were taking any cholinesterase inhibitor or memantine, or both, at baseline. That was where some differences did emerge.
The first compared the entire 4-mg cohort to the subset of patients taking 100 mg as monotherapy (absent any symptomatic medications). Both the 100-mg and 4-mg groups declined linearly on all measures, but compared to the 4 mg group the 100 mg group experienced about a 3-point benefit on both the ADAS-cog and ADS-ADL measures.* The 100-mg group also experienced significantly more dropouts (45% vs. 23%), with 16% of those being due to adverse events.
The second analysis compared the two 4-mg groups: those taking LMTM as monotherapy and those taking it in combination with standard AD medications. Again, both groups declined, but that decline was attenuated in the monotherapy group, with a 4-point advantage in the ADAS-cog and nearly a 5-point advantage in the ADS-ADL. The 3-cc ventricular volume advantage was seen as well.
Again, Dr. Schneider said, these results were recapitulated in the secondary endpoints, which he did not show.
The trial seems to upend TauRx’s earlier firm contention that the previously tested higher doses slow cognitive and functional decline – a view Dr. Wischik clung to after the July data were released. Dr. Schneider attempted to address this by suggesting that “the 4-mg dose may not have been as inactive as the developer thought.”
However, he noted, another possibility is that the patients who took the 4-mg dose but not the symptomatic drugs “may have had a more benign course of disease, compared to those taking cholinesterase inhibitors and memantine.”
This new study has now aroused the same criticism levied last summer. Maria Carrillo, PhD, chief scientific officer of the Alzheimer’s Association, was blunt in her assessment.
“The results of post hoc analyses, even when preplanned, are not valid and could be spurious,” she said. “They may mean nothing. As a field, we have been lured into rabbit holes in the past due to post hoc analyses and wasted too much time and way too much money. Of course, companies can do what they want as next steps in trials if they have the financial backing to do so.”
Dr. Wischik, however, said both the July data and the new data clearly justify taking the 4-mg dose forward in a randomized, placebo-controlled study.
“It’s really very exciting that we got exactly the same results now as we did in the post-hoc analysis [of the July data],” he said in an interview. “We predicted these results based on what we saw, we changed the statistical analysis, and we got the predicted results. This has nothing to do with data scouring.”
The question of whether the monotherapy patients are fundamentally different from those taking standard AD drugs is a valid one, he admitted. “We can’t avoid that criticism until we do another study where people who are not on any AD treatment are randomized.”
Dr. Wischik was then asked whether it would be difficult to recruit an entire cohort of patients with mild Alzheimer’s who are willing to forego approved symptomatic medications while in such a study. He did not think that would be problematic.
“Twenty percent of our cohort was already in that slot,” he said. “This practice pattern is determined somewhat by geography and somewhat by the type of clinician treating the patient. People also go on the drugs and then come off for various reasons. But even in the U.S., only 55% of Alzheimer’s patients are taking them.”
Dr. Wischik didn’t mention the problem of finding an appropriate placebo for such a study. If indeed the 4-mg dose is biologically active, such a placebo would have to be demonstrably inert, as well as provide the appropriate urine color to keep the blinding unbroken.
“That’s a challenge,” Dr. Schneider said.
Dr. Schneider was a coinvestigator on the LMTM phase III program. He has disclosed financial relationships with numerous pharmaceutical companies.
Correction, 12/12/16: An earlier version of this article misstated the results of this study.
[email protected]
On Twitter @Alz_Gal
There are several reasons why this kind of analysis defies scientific credibility.
The term “monotherapy” is really a euphemism for substandard care before the study. These patients on monotherapy were not selected to be so. These were people with mild to moderate AD dementia who should have been on memantine or a cholinesterase inhibitor and were not, for unknown reasons. They represent a health care bias, and on that fact alone this comparison should not have even been mentioned. Calling it monotherapy is an attempt to distract from the fact that this was an indication bias that defines this group.
The claim that this analysis was done before the database lock is true in principal. But because the investigators had already seen this result in their previous study, which was identically designed, they cannot really claim this was truly an ad hoc analysis. They already knew what they were going to see.
In July, the investigators claimed that the 100-mg dose was effective in monotherapy. They have been convinced over the entire course of development that the 4-mg dose was ineffective. Now they are retracting that. To me, this apparent wild goose chase for any kind of effect trivializes the entire process of a clinical trial.
What I believe we are observing here is a profound placebo effect that can occur when people who have been getting substandard care are put in a clinical trial and exposed to good care.
David Knopman, MD, is a clinical neurologist at the Mayo Clinic, Rochester, Minn., and a member of the Alzheimer’s Association Medical and Scientific Advisory Council.
There are several reasons why this kind of analysis defies scientific credibility.
The term “monotherapy” is really a euphemism for substandard care before the study. These patients on monotherapy were not selected to be so. These were people with mild to moderate AD dementia who should have been on memantine or a cholinesterase inhibitor and were not, for unknown reasons. They represent a health care bias, and on that fact alone this comparison should not have even been mentioned. Calling it monotherapy is an attempt to distract from the fact that this was an indication bias that defines this group.
The claim that this analysis was done before the database lock is true in principal. But because the investigators had already seen this result in their previous study, which was identically designed, they cannot really claim this was truly an ad hoc analysis. They already knew what they were going to see.
In July, the investigators claimed that the 100-mg dose was effective in monotherapy. They have been convinced over the entire course of development that the 4-mg dose was ineffective. Now they are retracting that. To me, this apparent wild goose chase for any kind of effect trivializes the entire process of a clinical trial.
What I believe we are observing here is a profound placebo effect that can occur when people who have been getting substandard care are put in a clinical trial and exposed to good care.
David Knopman, MD, is a clinical neurologist at the Mayo Clinic, Rochester, Minn., and a member of the Alzheimer’s Association Medical and Scientific Advisory Council.
There are several reasons why this kind of analysis defies scientific credibility.
The term “monotherapy” is really a euphemism for substandard care before the study. These patients on monotherapy were not selected to be so. These were people with mild to moderate AD dementia who should have been on memantine or a cholinesterase inhibitor and were not, for unknown reasons. They represent a health care bias, and on that fact alone this comparison should not have even been mentioned. Calling it monotherapy is an attempt to distract from the fact that this was an indication bias that defines this group.
The claim that this analysis was done before the database lock is true in principal. But because the investigators had already seen this result in their previous study, which was identically designed, they cannot really claim this was truly an ad hoc analysis. They already knew what they were going to see.
In July, the investigators claimed that the 100-mg dose was effective in monotherapy. They have been convinced over the entire course of development that the 4-mg dose was ineffective. Now they are retracting that. To me, this apparent wild goose chase for any kind of effect trivializes the entire process of a clinical trial.
What I believe we are observing here is a profound placebo effect that can occur when people who have been getting substandard care are put in a clinical trial and exposed to good care.
David Knopman, MD, is a clinical neurologist at the Mayo Clinic, Rochester, Minn., and a member of the Alzheimer’s Association Medical and Scientific Advisory Council.
SAN DIEGO – An anti-tau compound that has stirred scientific controversy for 8 years will continue along its developmental pathway at a much lower dose, despite yet another study that has Alzheimer’s researchers scratching their heads.
The drug, dubbed LMTM, is a derivative of the dye methylene blue. Its most recent phase III study, reported at the Clinical Trials on Alzheimer’s Disease conference, found that 100 mg twice a day conferred no cognitive or functional benefit upon patients with mild AD, compared with a control dose of 4 mg.
Some significant differences, however, did emerge in two prespecified subanalyses of the 4-mg control group. Patients who took the low dose, intended to be a placebo comparator, did better than those on the high dose – but only if they were not taking any standard symptomatic AD medications.
Based on these findings, TauRx, which is developing LMTM, will abandon the 100-mg dose and refocus on the 4-mg dose, said Claude Wischik, MD, chairman and chief executive officer of the Singapore-based company.
“I think it looks effective and there’s no advantage to going to a higher dose,” Dr. Wischik said in an interview. “The 100-mg dose doesn’t offer anything above the 4-mg dose, and we saw more dropouts in the higher-dose group. We will go forward with a new trial using 4 mg.”
The new commitment to 4 mg turns LMTM’s prior development trajectory on its head, as nothing lower than 75 mg has been investigated in a phase III study. The 4-mg control dose was used as a placebo stand-in, since LMTM colors urine blue or green. The low dose was considered biologically inactive and used to maintain the study blind.
Dr. Wischik has been investigating LMTM as a tau anti-aggregant for 10 years, first publicly reporting clinical data in 2008. LMTM has never posted significant cognitive or functional benefits in any primary analysis. Instead, it has moved forward based on a series of subanalyses that showed significant or near-significant benefits in smaller, meticulously constructed subgroups – conclusions that critics have called questionable at best. The most recent of these examined the drug’s effect in patients with mild to moderate disease and was presented last July at the Alzheimer’s Association’s International Conference (AAIC).
That study also didn’t meet its primary endpoints in the overall cohort of 891 patients, but TauRx promoted it as “promising,” based on a subgroup analysis of the 15% of patients who were not taking memantine or cholinesterase inhibitors.
Among these patients, those taking 75 mg twice daily declined 6 points less on the Alzheimer’s Disease Assessment Scale-cognitive subscale (ADAS-cog) than those taking 4 mg. Those taking 125 mg twice daily declined 6 points less than the 4-mg group. On the Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL), patients taking 75 mg twice daily scored 6.5 points higher than did the placebo group, indicating better function, and those taking 125 mg twice daily scored 7 points higher than did the placebo group.
At AAIC, researchers suggested that the monotherapy groups could have had a less aggressive disease course, or might not have had Alzheimer’s disease at all. Others complained about the unorthodox grouping of control patients in the subgroup analysis.
It was after digesting these data that TauRx investigators changed the statistical analysis of the current study, then in its final months, from a randomized trial to a cohort analysis. This was done before data lockdown, but it was still a dramatic shift from the original study design.
“The primary analysis was changed to essentially analyze this as a cohort study,” said Lon Schneider, MD, who presented the results at CTAD. “The comparisons of interest were patients taking 100 mg twice a day who were not on [symptomatic treatment], compared to the original control group of 4 mg. The other comparison was the 4-mg group not on cholinesterase inhibitors to the 4-mg group that was on them.”
The 18-month trial randomized 800 patients with mild AD to 100 mg LMTM twice daily or to 4 mg twice daily. Patients were drawn from two global regions: Canada and the United States, and eastern Europe and Australia.
The study group was a typical one, with a mean age of 70 years and a mean Mini Mental State Exam score of 22. The mean ADAS-cog score was 17. Most (80%) were taking a cholinesterase inhibitor, memantine, or both; 20% were naive to these medications.
Primary endpoints were the ADAS-cog11, ADCS-ADL, and left ventricular volume. Secondary outcomes included the Mini Mental State Exam and Neuropsychiatric Index.
In the primary analysis as the trial was created and conducted, LMTM 100 mg/twice daily did not confer any benefit, compared with the control 4-mg dose. The decline curves were virtually superimposable in the ADAS-cog score, ADCS-ADL score, and in loss of left ventricular volume.
This same nonsignificant pattern occurred in all the secondary endpoints, which Dr. Schneider did not show.
The cohort analyses stratified patients according to whether they were taking any cholinesterase inhibitor or memantine, or both, at baseline. That was where some differences did emerge.
The first compared the entire 4-mg cohort to the subset of patients taking 100 mg as monotherapy (absent any symptomatic medications). Both the 100-mg and 4-mg groups declined linearly on all measures, but compared to the 4 mg group the 100 mg group experienced about a 3-point benefit on both the ADAS-cog and ADS-ADL measures.* The 100-mg group also experienced significantly more dropouts (45% vs. 23%), with 16% of those being due to adverse events.
The second analysis compared the two 4-mg groups: those taking LMTM as monotherapy and those taking it in combination with standard AD medications. Again, both groups declined, but that decline was attenuated in the monotherapy group, with a 4-point advantage in the ADAS-cog and nearly a 5-point advantage in the ADS-ADL. The 3-cc ventricular volume advantage was seen as well.
Again, Dr. Schneider said, these results were recapitulated in the secondary endpoints, which he did not show.
The trial seems to upend TauRx’s earlier firm contention that the previously tested higher doses slow cognitive and functional decline – a view Dr. Wischik clung to after the July data were released. Dr. Schneider attempted to address this by suggesting that “the 4-mg dose may not have been as inactive as the developer thought.”
However, he noted, another possibility is that the patients who took the 4-mg dose but not the symptomatic drugs “may have had a more benign course of disease, compared to those taking cholinesterase inhibitors and memantine.”
This new study has now aroused the same criticism levied last summer. Maria Carrillo, PhD, chief scientific officer of the Alzheimer’s Association, was blunt in her assessment.
“The results of post hoc analyses, even when preplanned, are not valid and could be spurious,” she said. “They may mean nothing. As a field, we have been lured into rabbit holes in the past due to post hoc analyses and wasted too much time and way too much money. Of course, companies can do what they want as next steps in trials if they have the financial backing to do so.”
Dr. Wischik, however, said both the July data and the new data clearly justify taking the 4-mg dose forward in a randomized, placebo-controlled study.
“It’s really very exciting that we got exactly the same results now as we did in the post-hoc analysis [of the July data],” he said in an interview. “We predicted these results based on what we saw, we changed the statistical analysis, and we got the predicted results. This has nothing to do with data scouring.”
The question of whether the monotherapy patients are fundamentally different from those taking standard AD drugs is a valid one, he admitted. “We can’t avoid that criticism until we do another study where people who are not on any AD treatment are randomized.”
Dr. Wischik was then asked whether it would be difficult to recruit an entire cohort of patients with mild Alzheimer’s who are willing to forego approved symptomatic medications while in such a study. He did not think that would be problematic.
“Twenty percent of our cohort was already in that slot,” he said. “This practice pattern is determined somewhat by geography and somewhat by the type of clinician treating the patient. People also go on the drugs and then come off for various reasons. But even in the U.S., only 55% of Alzheimer’s patients are taking them.”
Dr. Wischik didn’t mention the problem of finding an appropriate placebo for such a study. If indeed the 4-mg dose is biologically active, such a placebo would have to be demonstrably inert, as well as provide the appropriate urine color to keep the blinding unbroken.
“That’s a challenge,” Dr. Schneider said.
Dr. Schneider was a coinvestigator on the LMTM phase III program. He has disclosed financial relationships with numerous pharmaceutical companies.
Correction, 12/12/16: An earlier version of this article misstated the results of this study.
[email protected]
On Twitter @Alz_Gal
SAN DIEGO – An anti-tau compound that has stirred scientific controversy for 8 years will continue along its developmental pathway at a much lower dose, despite yet another study that has Alzheimer’s researchers scratching their heads.
The drug, dubbed LMTM, is a derivative of the dye methylene blue. Its most recent phase III study, reported at the Clinical Trials on Alzheimer’s Disease conference, found that 100 mg twice a day conferred no cognitive or functional benefit upon patients with mild AD, compared with a control dose of 4 mg.
Some significant differences, however, did emerge in two prespecified subanalyses of the 4-mg control group. Patients who took the low dose, intended to be a placebo comparator, did better than those on the high dose – but only if they were not taking any standard symptomatic AD medications.
Based on these findings, TauRx, which is developing LMTM, will abandon the 100-mg dose and refocus on the 4-mg dose, said Claude Wischik, MD, chairman and chief executive officer of the Singapore-based company.
“I think it looks effective and there’s no advantage to going to a higher dose,” Dr. Wischik said in an interview. “The 100-mg dose doesn’t offer anything above the 4-mg dose, and we saw more dropouts in the higher-dose group. We will go forward with a new trial using 4 mg.”
The new commitment to 4 mg turns LMTM’s prior development trajectory on its head, as nothing lower than 75 mg has been investigated in a phase III study. The 4-mg control dose was used as a placebo stand-in, since LMTM colors urine blue or green. The low dose was considered biologically inactive and used to maintain the study blind.
Dr. Wischik has been investigating LMTM as a tau anti-aggregant for 10 years, first publicly reporting clinical data in 2008. LMTM has never posted significant cognitive or functional benefits in any primary analysis. Instead, it has moved forward based on a series of subanalyses that showed significant or near-significant benefits in smaller, meticulously constructed subgroups – conclusions that critics have called questionable at best. The most recent of these examined the drug’s effect in patients with mild to moderate disease and was presented last July at the Alzheimer’s Association’s International Conference (AAIC).
That study also didn’t meet its primary endpoints in the overall cohort of 891 patients, but TauRx promoted it as “promising,” based on a subgroup analysis of the 15% of patients who were not taking memantine or cholinesterase inhibitors.
Among these patients, those taking 75 mg twice daily declined 6 points less on the Alzheimer’s Disease Assessment Scale-cognitive subscale (ADAS-cog) than those taking 4 mg. Those taking 125 mg twice daily declined 6 points less than the 4-mg group. On the Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL), patients taking 75 mg twice daily scored 6.5 points higher than did the placebo group, indicating better function, and those taking 125 mg twice daily scored 7 points higher than did the placebo group.
At AAIC, researchers suggested that the monotherapy groups could have had a less aggressive disease course, or might not have had Alzheimer’s disease at all. Others complained about the unorthodox grouping of control patients in the subgroup analysis.
It was after digesting these data that TauRx investigators changed the statistical analysis of the current study, then in its final months, from a randomized trial to a cohort analysis. This was done before data lockdown, but it was still a dramatic shift from the original study design.
“The primary analysis was changed to essentially analyze this as a cohort study,” said Lon Schneider, MD, who presented the results at CTAD. “The comparisons of interest were patients taking 100 mg twice a day who were not on [symptomatic treatment], compared to the original control group of 4 mg. The other comparison was the 4-mg group not on cholinesterase inhibitors to the 4-mg group that was on them.”
The 18-month trial randomized 800 patients with mild AD to 100 mg LMTM twice daily or to 4 mg twice daily. Patients were drawn from two global regions: Canada and the United States, and eastern Europe and Australia.
The study group was a typical one, with a mean age of 70 years and a mean Mini Mental State Exam score of 22. The mean ADAS-cog score was 17. Most (80%) were taking a cholinesterase inhibitor, memantine, or both; 20% were naive to these medications.
Primary endpoints were the ADAS-cog11, ADCS-ADL, and left ventricular volume. Secondary outcomes included the Mini Mental State Exam and Neuropsychiatric Index.
In the primary analysis as the trial was created and conducted, LMTM 100 mg/twice daily did not confer any benefit, compared with the control 4-mg dose. The decline curves were virtually superimposable in the ADAS-cog score, ADCS-ADL score, and in loss of left ventricular volume.
This same nonsignificant pattern occurred in all the secondary endpoints, which Dr. Schneider did not show.
The cohort analyses stratified patients according to whether they were taking any cholinesterase inhibitor or memantine, or both, at baseline. That was where some differences did emerge.
The first compared the entire 4-mg cohort to the subset of patients taking 100 mg as monotherapy (absent any symptomatic medications). Both the 100-mg and 4-mg groups declined linearly on all measures, but compared to the 4 mg group the 100 mg group experienced about a 3-point benefit on both the ADAS-cog and ADS-ADL measures.* The 100-mg group also experienced significantly more dropouts (45% vs. 23%), with 16% of those being due to adverse events.
The second analysis compared the two 4-mg groups: those taking LMTM as monotherapy and those taking it in combination with standard AD medications. Again, both groups declined, but that decline was attenuated in the monotherapy group, with a 4-point advantage in the ADAS-cog and nearly a 5-point advantage in the ADS-ADL. The 3-cc ventricular volume advantage was seen as well.
Again, Dr. Schneider said, these results were recapitulated in the secondary endpoints, which he did not show.
The trial seems to upend TauRx’s earlier firm contention that the previously tested higher doses slow cognitive and functional decline – a view Dr. Wischik clung to after the July data were released. Dr. Schneider attempted to address this by suggesting that “the 4-mg dose may not have been as inactive as the developer thought.”
However, he noted, another possibility is that the patients who took the 4-mg dose but not the symptomatic drugs “may have had a more benign course of disease, compared to those taking cholinesterase inhibitors and memantine.”
This new study has now aroused the same criticism levied last summer. Maria Carrillo, PhD, chief scientific officer of the Alzheimer’s Association, was blunt in her assessment.
“The results of post hoc analyses, even when preplanned, are not valid and could be spurious,” she said. “They may mean nothing. As a field, we have been lured into rabbit holes in the past due to post hoc analyses and wasted too much time and way too much money. Of course, companies can do what they want as next steps in trials if they have the financial backing to do so.”
Dr. Wischik, however, said both the July data and the new data clearly justify taking the 4-mg dose forward in a randomized, placebo-controlled study.
“It’s really very exciting that we got exactly the same results now as we did in the post-hoc analysis [of the July data],” he said in an interview. “We predicted these results based on what we saw, we changed the statistical analysis, and we got the predicted results. This has nothing to do with data scouring.”
The question of whether the monotherapy patients are fundamentally different from those taking standard AD drugs is a valid one, he admitted. “We can’t avoid that criticism until we do another study where people who are not on any AD treatment are randomized.”
Dr. Wischik was then asked whether it would be difficult to recruit an entire cohort of patients with mild Alzheimer’s who are willing to forego approved symptomatic medications while in such a study. He did not think that would be problematic.
“Twenty percent of our cohort was already in that slot,” he said. “This practice pattern is determined somewhat by geography and somewhat by the type of clinician treating the patient. People also go on the drugs and then come off for various reasons. But even in the U.S., only 55% of Alzheimer’s patients are taking them.”
Dr. Wischik didn’t mention the problem of finding an appropriate placebo for such a study. If indeed the 4-mg dose is biologically active, such a placebo would have to be demonstrably inert, as well as provide the appropriate urine color to keep the blinding unbroken.
“That’s a challenge,” Dr. Schneider said.
Dr. Schneider was a coinvestigator on the LMTM phase III program. He has disclosed financial relationships with numerous pharmaceutical companies.
Correction, 12/12/16: An earlier version of this article misstated the results of this study.
[email protected]
On Twitter @Alz_Gal
AT CTAD
Key clinical point:
Major finding: After saying that the placebo dose of drug effected cognitive and clinical benefit in a cohort analysis comparing it to 100 mg twice daily, the company will further develop LMTM in a 4-mg dose.
Data source: The cohort analysis involved 891 patients with mild Alzheimer’s.
Disclosures: Dr. Wischik is the founder and president of Singapore-based TauRx. Dr. Schneider is an investigator in the drug’s phase III trial and has reported financial relationships with numerous pharmaceutical companies.