User login
COVID-19 vaccinations lag in youngest children
Case: A 3-year-old girl presented to the emergency department after a brief seizure at home. She looked well on physical exam except for a fever of 103° F and thick rhinorrhea.
The intern on duty methodically worked through the standard list of questions. “Immunizations up to date?” she asked.
“Absolutely,” the child’s mom responded. “She’s had everything that’s recommended.”
“Including COVID-19 vaccine?” the intern prompted.
“No.” The mom responded with a shake of her head. “We don’t do that vaccine.”
That mom is not alone.
COVID-19 vaccines for children as young as 6 months were given emergency-use authorization by the Food and Drug Administration in June 2022 and in February 2023, the Advisory Committee on Immunization Practices included COVID-19 vaccine on the routine childhood immunization schedule.
COVID-19 vaccines are safe in young children, and they prevent the most severe outcomes associated with infection, including hospitalization. Newly released data confirm that the COVID-19 vaccines produced by Moderna and Pfizer also provide protection against symptomatic infection for at least 4 months after completion of the monovalent primary series.
In a Morbidity and Mortality Weekly Report released on Feb. 17, 2023, the Centers for Disease Control and Prevention reported the results of a test-negative design case-control study that enrolled symptomatic children tested for SARS-CoV-2 infection through Feb. 5, 2023, as part of the Increasing Community Access to Testing (ICATT) program.1 ICATT provides SARS-CoV-2 testing to persons aged at least 3 years at pharmacy and community-based testing sites nationwide.
Two doses of monovalent Moderna vaccine (complete primary series) was 60% effective against symptomatic infection (95% confidence interval, 49%-68%) 2 weeks to 2 months after receipt of the second dose. Vaccine effectiveness dropped to 36% (95% CI, 15%-52%) 3-4 months after the second dose. Three doses of monovalent Pfizer-BioNTech vaccine (complete primary series) was 31% effective (95% CI, 7%-49%) at preventing symptomatic infection 2 weeks to 4 months after receipt of the third dose. A bivalent vaccine dose for eligible children is expected to provide more protection against currently circulating SARS-CoV-2 variants.
Despite evidence of vaccine efficacy, very few parents are opting to protect their young children with the COVID-19 vaccine. The CDC reports that, as of March 1, 2023, only 8% of children under 2 years and 10.5% of children aged 2-4 years have initiated a COVID vaccine series. The American Academy of Pediatrics has emphasized that 15.0 million children between the ages of 6 months and 4 years have not yet received their first COVID-19 vaccine dose.
While the reasons underlying low COVID-19 vaccination rates in young children are complex, themes emerge. Socioeconomic disparities contributing to low vaccination rates in young children were highlighted in another recent MMWR article.2 Through Dec. 1, 2022, vaccination coverage was lower in rural counties (3.4%) than in urban counties (10.5%). Rates were lower in Black and Hispanic children than in White and Asian children.
According to the CDC, high rates of poverty in Black and Hispanic communities may affect vaccination coverage by affecting caregivers’ access to vaccination sites or ability to leave work to take their child to be vaccinated. Pediatric care providers have repeatedly been identified by parents as a source of trusted vaccine information and a strong provider recommendation is associated with vaccination, but not all families are receiving vaccine advice. In a 2022 Kaiser Family Foundation survey, parents of young children with annual household incomes above $90,000 were more likely to talk to their pediatrician about a COVID-19 vaccine than families with lower incomes.3Vaccine hesitancy, fueled by general confusion and skepticism, is another factor contributing to low vaccination rates. Admittedly, the recommendations are complex and on March 14, 2023, the FDA again revised the emergency-use authorization for young children. Some caregivers continue to express concerns about vaccine side effects as well as the belief that the vaccine won’t prevent their child from getting sick.
Kendall Purcell, MD, a pediatrician with Norton Children’s Medical Group in Louisville, Ky., recommends COVID-19 vaccination for her patients because it reduces the risk of severe disease. That factored into her own decision to vaccinate her 4-year-old son and 1-year-old daughter, but she hasn’t been able to convince the parents of all her patients. “Some feel that COVID-19 is not as severe for children, so the risks don’t outweigh the benefits when it comes to vaccinating their children.” Back to our case: In the ED the intern reviewed the laboratory testing she had ordered. She then sat down with the mother of the 3-year-old girl to discuss the diagnosis: febrile seizure associated with COVID-19 infection. Febrile seizures are a well-recognized but uncommon complication of COVID-19 in children. In a retrospective cohort study using electronic health record data, febrile seizures occurred in 0.5% of 8,854 children aged 0-5 years with COVID-19 infection.4 About 9% of these children required critical care services. In another cohort of hospitalized children, neurologic complications occurred in 7% of children hospitalized with COVID-19.5 Febrile and nonfebrile seizures were most commonly observed.
“I really thought COVID-19 was no big deal in young kids,” the mom said. “Parents need the facts.”
The facts are these: Through Dec. 2, 2022, more than 3 million cases of COVID-19 have been reported in children aged younger than 5 years. While COVID is generally less severe in young children than older adults, it is difficult to predict which children will become seriously ill. When children are hospitalized, one in four requires intensive care. COVID-19 is now a vaccine-preventable disease, but too many children remain unprotected.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected]. Ms. Ezell is a recent graduate from Indiana University Southeast with a Bachelor of Arts in English. They have no conflicts of interest.
References
1. Fleming-Dutra KE et al. Morb Mortal Wkly Rep. 2023;72:177-182.
2. Murthy BP et al. Morb Mortal Wkly Rep. 2023;72:183-9.
3. Lopes L et al. KFF COVID-19 vaccine monitor: July 2022. San Francisco: Kaiser Family Foundation, 2022.
4. Cadet K et al. J Child Neurol. 2022 Apr;37(5):410-5.
5. Antoon JW et al. Pediatrics. 2022 Nov 1;150(5):e2022058167.
Case: A 3-year-old girl presented to the emergency department after a brief seizure at home. She looked well on physical exam except for a fever of 103° F and thick rhinorrhea.
The intern on duty methodically worked through the standard list of questions. “Immunizations up to date?” she asked.
“Absolutely,” the child’s mom responded. “She’s had everything that’s recommended.”
“Including COVID-19 vaccine?” the intern prompted.
“No.” The mom responded with a shake of her head. “We don’t do that vaccine.”
That mom is not alone.
COVID-19 vaccines for children as young as 6 months were given emergency-use authorization by the Food and Drug Administration in June 2022 and in February 2023, the Advisory Committee on Immunization Practices included COVID-19 vaccine on the routine childhood immunization schedule.
COVID-19 vaccines are safe in young children, and they prevent the most severe outcomes associated with infection, including hospitalization. Newly released data confirm that the COVID-19 vaccines produced by Moderna and Pfizer also provide protection against symptomatic infection for at least 4 months after completion of the monovalent primary series.
In a Morbidity and Mortality Weekly Report released on Feb. 17, 2023, the Centers for Disease Control and Prevention reported the results of a test-negative design case-control study that enrolled symptomatic children tested for SARS-CoV-2 infection through Feb. 5, 2023, as part of the Increasing Community Access to Testing (ICATT) program.1 ICATT provides SARS-CoV-2 testing to persons aged at least 3 years at pharmacy and community-based testing sites nationwide.
Two doses of monovalent Moderna vaccine (complete primary series) was 60% effective against symptomatic infection (95% confidence interval, 49%-68%) 2 weeks to 2 months after receipt of the second dose. Vaccine effectiveness dropped to 36% (95% CI, 15%-52%) 3-4 months after the second dose. Three doses of monovalent Pfizer-BioNTech vaccine (complete primary series) was 31% effective (95% CI, 7%-49%) at preventing symptomatic infection 2 weeks to 4 months after receipt of the third dose. A bivalent vaccine dose for eligible children is expected to provide more protection against currently circulating SARS-CoV-2 variants.
Despite evidence of vaccine efficacy, very few parents are opting to protect their young children with the COVID-19 vaccine. The CDC reports that, as of March 1, 2023, only 8% of children under 2 years and 10.5% of children aged 2-4 years have initiated a COVID vaccine series. The American Academy of Pediatrics has emphasized that 15.0 million children between the ages of 6 months and 4 years have not yet received their first COVID-19 vaccine dose.
While the reasons underlying low COVID-19 vaccination rates in young children are complex, themes emerge. Socioeconomic disparities contributing to low vaccination rates in young children were highlighted in another recent MMWR article.2 Through Dec. 1, 2022, vaccination coverage was lower in rural counties (3.4%) than in urban counties (10.5%). Rates were lower in Black and Hispanic children than in White and Asian children.
According to the CDC, high rates of poverty in Black and Hispanic communities may affect vaccination coverage by affecting caregivers’ access to vaccination sites or ability to leave work to take their child to be vaccinated. Pediatric care providers have repeatedly been identified by parents as a source of trusted vaccine information and a strong provider recommendation is associated with vaccination, but not all families are receiving vaccine advice. In a 2022 Kaiser Family Foundation survey, parents of young children with annual household incomes above $90,000 were more likely to talk to their pediatrician about a COVID-19 vaccine than families with lower incomes.3Vaccine hesitancy, fueled by general confusion and skepticism, is another factor contributing to low vaccination rates. Admittedly, the recommendations are complex and on March 14, 2023, the FDA again revised the emergency-use authorization for young children. Some caregivers continue to express concerns about vaccine side effects as well as the belief that the vaccine won’t prevent their child from getting sick.
Kendall Purcell, MD, a pediatrician with Norton Children’s Medical Group in Louisville, Ky., recommends COVID-19 vaccination for her patients because it reduces the risk of severe disease. That factored into her own decision to vaccinate her 4-year-old son and 1-year-old daughter, but she hasn’t been able to convince the parents of all her patients. “Some feel that COVID-19 is not as severe for children, so the risks don’t outweigh the benefits when it comes to vaccinating their children.” Back to our case: In the ED the intern reviewed the laboratory testing she had ordered. She then sat down with the mother of the 3-year-old girl to discuss the diagnosis: febrile seizure associated with COVID-19 infection. Febrile seizures are a well-recognized but uncommon complication of COVID-19 in children. In a retrospective cohort study using electronic health record data, febrile seizures occurred in 0.5% of 8,854 children aged 0-5 years with COVID-19 infection.4 About 9% of these children required critical care services. In another cohort of hospitalized children, neurologic complications occurred in 7% of children hospitalized with COVID-19.5 Febrile and nonfebrile seizures were most commonly observed.
“I really thought COVID-19 was no big deal in young kids,” the mom said. “Parents need the facts.”
The facts are these: Through Dec. 2, 2022, more than 3 million cases of COVID-19 have been reported in children aged younger than 5 years. While COVID is generally less severe in young children than older adults, it is difficult to predict which children will become seriously ill. When children are hospitalized, one in four requires intensive care. COVID-19 is now a vaccine-preventable disease, but too many children remain unprotected.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected]. Ms. Ezell is a recent graduate from Indiana University Southeast with a Bachelor of Arts in English. They have no conflicts of interest.
References
1. Fleming-Dutra KE et al. Morb Mortal Wkly Rep. 2023;72:177-182.
2. Murthy BP et al. Morb Mortal Wkly Rep. 2023;72:183-9.
3. Lopes L et al. KFF COVID-19 vaccine monitor: July 2022. San Francisco: Kaiser Family Foundation, 2022.
4. Cadet K et al. J Child Neurol. 2022 Apr;37(5):410-5.
5. Antoon JW et al. Pediatrics. 2022 Nov 1;150(5):e2022058167.
Case: A 3-year-old girl presented to the emergency department after a brief seizure at home. She looked well on physical exam except for a fever of 103° F and thick rhinorrhea.
The intern on duty methodically worked through the standard list of questions. “Immunizations up to date?” she asked.
“Absolutely,” the child’s mom responded. “She’s had everything that’s recommended.”
“Including COVID-19 vaccine?” the intern prompted.
“No.” The mom responded with a shake of her head. “We don’t do that vaccine.”
That mom is not alone.
COVID-19 vaccines for children as young as 6 months were given emergency-use authorization by the Food and Drug Administration in June 2022 and in February 2023, the Advisory Committee on Immunization Practices included COVID-19 vaccine on the routine childhood immunization schedule.
COVID-19 vaccines are safe in young children, and they prevent the most severe outcomes associated with infection, including hospitalization. Newly released data confirm that the COVID-19 vaccines produced by Moderna and Pfizer also provide protection against symptomatic infection for at least 4 months after completion of the monovalent primary series.
In a Morbidity and Mortality Weekly Report released on Feb. 17, 2023, the Centers for Disease Control and Prevention reported the results of a test-negative design case-control study that enrolled symptomatic children tested for SARS-CoV-2 infection through Feb. 5, 2023, as part of the Increasing Community Access to Testing (ICATT) program.1 ICATT provides SARS-CoV-2 testing to persons aged at least 3 years at pharmacy and community-based testing sites nationwide.
Two doses of monovalent Moderna vaccine (complete primary series) was 60% effective against symptomatic infection (95% confidence interval, 49%-68%) 2 weeks to 2 months after receipt of the second dose. Vaccine effectiveness dropped to 36% (95% CI, 15%-52%) 3-4 months after the second dose. Three doses of monovalent Pfizer-BioNTech vaccine (complete primary series) was 31% effective (95% CI, 7%-49%) at preventing symptomatic infection 2 weeks to 4 months after receipt of the third dose. A bivalent vaccine dose for eligible children is expected to provide more protection against currently circulating SARS-CoV-2 variants.
Despite evidence of vaccine efficacy, very few parents are opting to protect their young children with the COVID-19 vaccine. The CDC reports that, as of March 1, 2023, only 8% of children under 2 years and 10.5% of children aged 2-4 years have initiated a COVID vaccine series. The American Academy of Pediatrics has emphasized that 15.0 million children between the ages of 6 months and 4 years have not yet received their first COVID-19 vaccine dose.
While the reasons underlying low COVID-19 vaccination rates in young children are complex, themes emerge. Socioeconomic disparities contributing to low vaccination rates in young children were highlighted in another recent MMWR article.2 Through Dec. 1, 2022, vaccination coverage was lower in rural counties (3.4%) than in urban counties (10.5%). Rates were lower in Black and Hispanic children than in White and Asian children.
According to the CDC, high rates of poverty in Black and Hispanic communities may affect vaccination coverage by affecting caregivers’ access to vaccination sites or ability to leave work to take their child to be vaccinated. Pediatric care providers have repeatedly been identified by parents as a source of trusted vaccine information and a strong provider recommendation is associated with vaccination, but not all families are receiving vaccine advice. In a 2022 Kaiser Family Foundation survey, parents of young children with annual household incomes above $90,000 were more likely to talk to their pediatrician about a COVID-19 vaccine than families with lower incomes.3Vaccine hesitancy, fueled by general confusion and skepticism, is another factor contributing to low vaccination rates. Admittedly, the recommendations are complex and on March 14, 2023, the FDA again revised the emergency-use authorization for young children. Some caregivers continue to express concerns about vaccine side effects as well as the belief that the vaccine won’t prevent their child from getting sick.
Kendall Purcell, MD, a pediatrician with Norton Children’s Medical Group in Louisville, Ky., recommends COVID-19 vaccination for her patients because it reduces the risk of severe disease. That factored into her own decision to vaccinate her 4-year-old son and 1-year-old daughter, but she hasn’t been able to convince the parents of all her patients. “Some feel that COVID-19 is not as severe for children, so the risks don’t outweigh the benefits when it comes to vaccinating their children.” Back to our case: In the ED the intern reviewed the laboratory testing she had ordered. She then sat down with the mother of the 3-year-old girl to discuss the diagnosis: febrile seizure associated with COVID-19 infection. Febrile seizures are a well-recognized but uncommon complication of COVID-19 in children. In a retrospective cohort study using electronic health record data, febrile seizures occurred in 0.5% of 8,854 children aged 0-5 years with COVID-19 infection.4 About 9% of these children required critical care services. In another cohort of hospitalized children, neurologic complications occurred in 7% of children hospitalized with COVID-19.5 Febrile and nonfebrile seizures were most commonly observed.
“I really thought COVID-19 was no big deal in young kids,” the mom said. “Parents need the facts.”
The facts are these: Through Dec. 2, 2022, more than 3 million cases of COVID-19 have been reported in children aged younger than 5 years. While COVID is generally less severe in young children than older adults, it is difficult to predict which children will become seriously ill. When children are hospitalized, one in four requires intensive care. COVID-19 is now a vaccine-preventable disease, but too many children remain unprotected.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead. Email her at [email protected]. Ms. Ezell is a recent graduate from Indiana University Southeast with a Bachelor of Arts in English. They have no conflicts of interest.
References
1. Fleming-Dutra KE et al. Morb Mortal Wkly Rep. 2023;72:177-182.
2. Murthy BP et al. Morb Mortal Wkly Rep. 2023;72:183-9.
3. Lopes L et al. KFF COVID-19 vaccine monitor: July 2022. San Francisco: Kaiser Family Foundation, 2022.
4. Cadet K et al. J Child Neurol. 2022 Apr;37(5):410-5.
5. Antoon JW et al. Pediatrics. 2022 Nov 1;150(5):e2022058167.
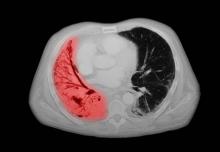
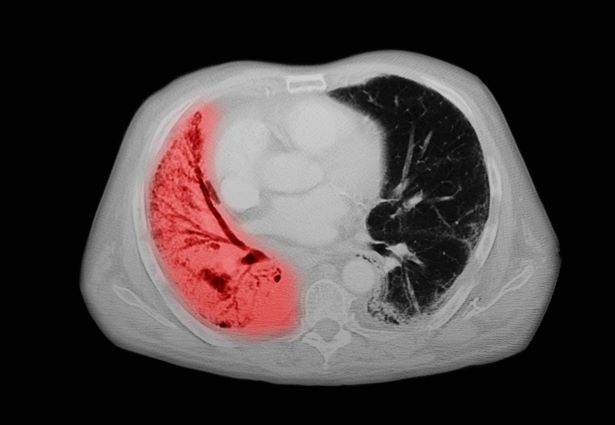
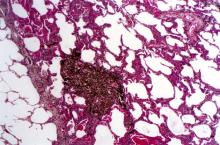
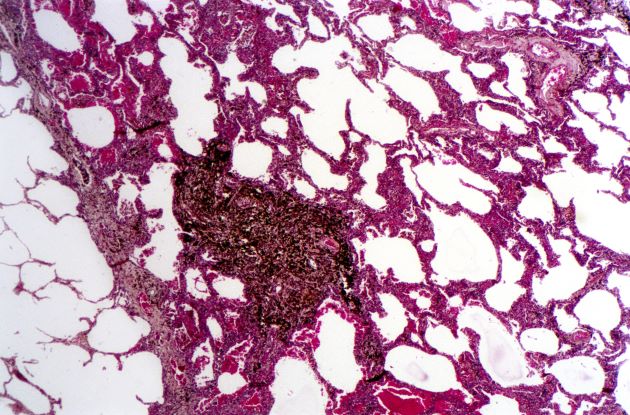
SCLC: Presentation and Diagnosis
Older woman presents with shortness of breath
Small cell lung cancer (SCLC) is the most aggressive lung cancer subtype, has a poor prognosis, and is highly associated with smoking. It accounts for 10%-15% of all lung cancers. Rapid tumor growth may lead to obstruction of major airways, with distal collapse leading to postobstructive pneumonitis, infection, and fever. SCLCs usually grow rapidly and metastasize to mediastinal lymph nodes relatively early in the course of the disease. At presentation, patients may have very large intrathoracic tumors, and distinguishing the primary tumor from lymph node metastases may be difficult.
CT of all common sites of metastasis should be performed to stage the disease. CT scanning of the thorax (lungs and mediastinum) and commonly involved abdominal viscera is the minimum requirement in standard staging workup of SCLC. Intravenous contrast agents should be used whenever possible. In the United States, CT scans of the chest and upper abdomen to include the liver and adrenal glands are standard. The liver is a common site of metastasis.
Most cases of SCLC will present in advanced stages, be inoperable, and have a dismal prognosis. Only about 5% of patients present at an early stage (Ia, Ib, or IIa). The National Comprehensive Cancer Network (NCCN) guidelines recommend that these patients be managed with aggressive chemoradiation therapy; in some, lobectomy associated with mediastinal lymph node dissection may be performed. The NCCN notes that advanced disease is managed primarily with chemotherapy, mainly for palliation and symptom control. Older patients, such as the current patient, who have a good performance status (ECOG 0 or 1) and intact organ function should receive standard carboplatin-based chemotherapy. However, even those who have poor prognostic factors (eg, poor performance status, medically significant concomitant conditions) may still be considered for chemotherapy if appropriate precautions are taken to avoid excessive toxicity and further decline in performance status.
Unlike non-SCLC, which has seen waves of new drug approvals from the US Food and Drug Administration (FDA), the prognosis for SCLC has not changed substantially in the past two decades and remains poor. However, the FDA approved atezolizumab in combination with carboplatin and etoposide for first-line treatment of adult patients with extensive-stage SCLC. Further, the approval of durvalumab in combination with chemotherapy; and the approval of lurbinectedin, a novel chemotherapy agent approved for second-line treatment of SCLC, have added to the therapeutic options for patients with SCLC.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships
Small cell lung cancer (SCLC) is the most aggressive lung cancer subtype, has a poor prognosis, and is highly associated with smoking. It accounts for 10%-15% of all lung cancers. Rapid tumor growth may lead to obstruction of major airways, with distal collapse leading to postobstructive pneumonitis, infection, and fever. SCLCs usually grow rapidly and metastasize to mediastinal lymph nodes relatively early in the course of the disease. At presentation, patients may have very large intrathoracic tumors, and distinguishing the primary tumor from lymph node metastases may be difficult.
CT of all common sites of metastasis should be performed to stage the disease. CT scanning of the thorax (lungs and mediastinum) and commonly involved abdominal viscera is the minimum requirement in standard staging workup of SCLC. Intravenous contrast agents should be used whenever possible. In the United States, CT scans of the chest and upper abdomen to include the liver and adrenal glands are standard. The liver is a common site of metastasis.
Most cases of SCLC will present in advanced stages, be inoperable, and have a dismal prognosis. Only about 5% of patients present at an early stage (Ia, Ib, or IIa). The National Comprehensive Cancer Network (NCCN) guidelines recommend that these patients be managed with aggressive chemoradiation therapy; in some, lobectomy associated with mediastinal lymph node dissection may be performed. The NCCN notes that advanced disease is managed primarily with chemotherapy, mainly for palliation and symptom control. Older patients, such as the current patient, who have a good performance status (ECOG 0 or 1) and intact organ function should receive standard carboplatin-based chemotherapy. However, even those who have poor prognostic factors (eg, poor performance status, medically significant concomitant conditions) may still be considered for chemotherapy if appropriate precautions are taken to avoid excessive toxicity and further decline in performance status.
Unlike non-SCLC, which has seen waves of new drug approvals from the US Food and Drug Administration (FDA), the prognosis for SCLC has not changed substantially in the past two decades and remains poor. However, the FDA approved atezolizumab in combination with carboplatin and etoposide for first-line treatment of adult patients with extensive-stage SCLC. Further, the approval of durvalumab in combination with chemotherapy; and the approval of lurbinectedin, a novel chemotherapy agent approved for second-line treatment of SCLC, have added to the therapeutic options for patients with SCLC.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships
Small cell lung cancer (SCLC) is the most aggressive lung cancer subtype, has a poor prognosis, and is highly associated with smoking. It accounts for 10%-15% of all lung cancers. Rapid tumor growth may lead to obstruction of major airways, with distal collapse leading to postobstructive pneumonitis, infection, and fever. SCLCs usually grow rapidly and metastasize to mediastinal lymph nodes relatively early in the course of the disease. At presentation, patients may have very large intrathoracic tumors, and distinguishing the primary tumor from lymph node metastases may be difficult.
CT of all common sites of metastasis should be performed to stage the disease. CT scanning of the thorax (lungs and mediastinum) and commonly involved abdominal viscera is the minimum requirement in standard staging workup of SCLC. Intravenous contrast agents should be used whenever possible. In the United States, CT scans of the chest and upper abdomen to include the liver and adrenal glands are standard. The liver is a common site of metastasis.
Most cases of SCLC will present in advanced stages, be inoperable, and have a dismal prognosis. Only about 5% of patients present at an early stage (Ia, Ib, or IIa). The National Comprehensive Cancer Network (NCCN) guidelines recommend that these patients be managed with aggressive chemoradiation therapy; in some, lobectomy associated with mediastinal lymph node dissection may be performed. The NCCN notes that advanced disease is managed primarily with chemotherapy, mainly for palliation and symptom control. Older patients, such as the current patient, who have a good performance status (ECOG 0 or 1) and intact organ function should receive standard carboplatin-based chemotherapy. However, even those who have poor prognostic factors (eg, poor performance status, medically significant concomitant conditions) may still be considered for chemotherapy if appropriate precautions are taken to avoid excessive toxicity and further decline in performance status.
Unlike non-SCLC, which has seen waves of new drug approvals from the US Food and Drug Administration (FDA), the prognosis for SCLC has not changed substantially in the past two decades and remains poor. However, the FDA approved atezolizumab in combination with carboplatin and etoposide for first-line treatment of adult patients with extensive-stage SCLC. Further, the approval of durvalumab in combination with chemotherapy; and the approval of lurbinectedin, a novel chemotherapy agent approved for second-line treatment of SCLC, have added to the therapeutic options for patients with SCLC.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships
A 72-year-old woman presents with shortness of breath, a productive cough, chest pain, some fatigue, anorexia, a recent 18-pound weight loss, and a history of hypertension. She is currently a smoker and has a 45–pack-year smoking history. On physical examination she has some dullness to percussion with some decreased breath sounds. She has a distended abdomen and complains of itchy skin. The chest x-ray shows a left hilar mass and a 5.4-cm left upper-lobe mass. A CT scan reveals a hilar mass with a mediastinal extension.
Shortness of breath and chest pain
Liver biopsy will reveal the cause of the liver dysfunction and confirm whether this patient has small cell lung cancer (SCLC), as suspected, and whether it has spread to the liver. This will affect chemotherapy options.
Most cases of SCLC will present in advanced stages, be inoperable, and have a dismal prognosis. Only about 5% of patients present at an early stage (Ia, Ib, or IIa). The National Comprehensive Cancer Network (NCCN) guidelines recommend that patients with advanced disease be managed with aggressive chemoradiation therapy and immunotherapy.
SCLC is the most aggressive lung cancer subtype. It is almost always associated with smoking. It accounts for 10%-15% of all lung cancers. Rapid tumor growth may lead to obstruction of major airways, with distal collapse leading to postobstructive pneumonitis, infection, and fever. About 70% of patients show metastatic spread at presentation, and the liver is one of the most common sites of metastasis.
Unlike non-SCLC, for which the prognosis has improved in part because of several new drug approvals from the US Food and Drug Administration, the prognosis for SCLC has not changed substantially in the past two decades and remains poor. However, with immunotherapy, the outlook may be improving. The NCCN recommends atezolizumab in combination with carboplatin and etoposide for first-line treatment of adult patients with extensive-stage SCLC. Furthermore, the approval of durvalumab in combination with chemotherapy and the approval of lurbinectedin, a novel chemotherapy agent approved for second-line treatment of SCLC, have added to the therapeutic options for patients with advanced SCLC.
Maurie Markman, MD, President, Department of Medical Oncology, Cancer Treatment Centers of America.
Maurie Markman, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Merck
Serve(d) as a speaker or a member of a speakers bureau for: AstraZeneca; Novis; Glaxo Smith Kline
Received research grants from: AstraZeneca; Novis; GSK; Merck
Liver biopsy will reveal the cause of the liver dysfunction and confirm whether this patient has small cell lung cancer (SCLC), as suspected, and whether it has spread to the liver. This will affect chemotherapy options.
Most cases of SCLC will present in advanced stages, be inoperable, and have a dismal prognosis. Only about 5% of patients present at an early stage (Ia, Ib, or IIa). The National Comprehensive Cancer Network (NCCN) guidelines recommend that patients with advanced disease be managed with aggressive chemoradiation therapy and immunotherapy.
SCLC is the most aggressive lung cancer subtype. It is almost always associated with smoking. It accounts for 10%-15% of all lung cancers. Rapid tumor growth may lead to obstruction of major airways, with distal collapse leading to postobstructive pneumonitis, infection, and fever. About 70% of patients show metastatic spread at presentation, and the liver is one of the most common sites of metastasis.
Unlike non-SCLC, for which the prognosis has improved in part because of several new drug approvals from the US Food and Drug Administration, the prognosis for SCLC has not changed substantially in the past two decades and remains poor. However, with immunotherapy, the outlook may be improving. The NCCN recommends atezolizumab in combination with carboplatin and etoposide for first-line treatment of adult patients with extensive-stage SCLC. Furthermore, the approval of durvalumab in combination with chemotherapy and the approval of lurbinectedin, a novel chemotherapy agent approved for second-line treatment of SCLC, have added to the therapeutic options for patients with advanced SCLC.
Maurie Markman, MD, President, Department of Medical Oncology, Cancer Treatment Centers of America.
Maurie Markman, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Merck
Serve(d) as a speaker or a member of a speakers bureau for: AstraZeneca; Novis; Glaxo Smith Kline
Received research grants from: AstraZeneca; Novis; GSK; Merck
Liver biopsy will reveal the cause of the liver dysfunction and confirm whether this patient has small cell lung cancer (SCLC), as suspected, and whether it has spread to the liver. This will affect chemotherapy options.
Most cases of SCLC will present in advanced stages, be inoperable, and have a dismal prognosis. Only about 5% of patients present at an early stage (Ia, Ib, or IIa). The National Comprehensive Cancer Network (NCCN) guidelines recommend that patients with advanced disease be managed with aggressive chemoradiation therapy and immunotherapy.
SCLC is the most aggressive lung cancer subtype. It is almost always associated with smoking. It accounts for 10%-15% of all lung cancers. Rapid tumor growth may lead to obstruction of major airways, with distal collapse leading to postobstructive pneumonitis, infection, and fever. About 70% of patients show metastatic spread at presentation, and the liver is one of the most common sites of metastasis.
Unlike non-SCLC, for which the prognosis has improved in part because of several new drug approvals from the US Food and Drug Administration, the prognosis for SCLC has not changed substantially in the past two decades and remains poor. However, with immunotherapy, the outlook may be improving. The NCCN recommends atezolizumab in combination with carboplatin and etoposide for first-line treatment of adult patients with extensive-stage SCLC. Furthermore, the approval of durvalumab in combination with chemotherapy and the approval of lurbinectedin, a novel chemotherapy agent approved for second-line treatment of SCLC, have added to the therapeutic options for patients with advanced SCLC.
Maurie Markman, MD, President, Department of Medical Oncology, Cancer Treatment Centers of America.
Maurie Markman, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Merck
Serve(d) as a speaker or a member of a speakers bureau for: AstraZeneca; Novis; Glaxo Smith Kline
Received research grants from: AstraZeneca; Novis; GSK; Merck
A 64-year-old man presents with shortness of breath, a productive cough, chest pain, some fatigue, anorexia, a recent 15-lb weight loss, jaundice, and a history of type 2 diabetes. He is a college professor and has a 35–pack-year smoking history; he quit 3 years ago.
On physical examination, he has dullness to percussion and decreased breath sounds. His laboratory data reveal elevated serum liver enzyme levels. Chest radiography shows a left hilar mass and a 5.6-cm left upper lobe mass. CT reveals a hilar mass with a bilateral mediastinal extension and pneumonia. PET shows activity in the left upper lobe mass, with supraclavicular nodal areas and liver lesions. MRI shows that there are no metastases to the brain.
Woman presents with cough and bronchorrhea
Bronchioalveolar cell carcinoma (BAC) is a variant of non–small cell lung cancer (NSCLC) that, in recent years, has received a new identity in some of the literature. Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma (MIA) are relatively new entities that in some published literature have replaced the term BAC. The National Comprehensive Cancer Network recognizes these terms. AIS is defined as a localized adenocarcinoma of < 3 cm that exhibits a lepidic growth pattern, with neoplastic cells along the alveolar structures but without stromal, vascular, or pleural invasion. MIA refers to small, solitary adenocarcinomas < 3 cm with either pure lepidic growth or predominant lepidic growth with ≤ 5 mm of stromal invasion. BAC has unique epidemiologic, pathologic, and clinical features compared with other NSCLC subtypes. For example, although it is smoking-related, the relationship of BAC to smoking is less strong than with other types of NSCLC. About a third of patients with BAC are never-smokers.
There are also some unique radiographic features — its presentation may be confused with pneumonia or other inflammatory conditions in the lung, and only after a patient does not improve after a course of antibiotics should a diagnosis of BAC be considered. Unlike other types of lung cancer where chemotherapy may be the first plan of attack, surgery is often the first choice for treating BAC, particularly when there is no mediastinal node involvement (10%-25% of cases) or distal metastases (5% of cases). BAC usually harbors EGFR mutation. It is responsive to new targeted therapies for lung cancer, particularly osimertinib, afatinib, erlotinib, and gefitinib. Thus, people with BAC are good candidates for genetic testing.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Bronchioalveolar cell carcinoma (BAC) is a variant of non–small cell lung cancer (NSCLC) that, in recent years, has received a new identity in some of the literature. Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma (MIA) are relatively new entities that in some published literature have replaced the term BAC. The National Comprehensive Cancer Network recognizes these terms. AIS is defined as a localized adenocarcinoma of < 3 cm that exhibits a lepidic growth pattern, with neoplastic cells along the alveolar structures but without stromal, vascular, or pleural invasion. MIA refers to small, solitary adenocarcinomas < 3 cm with either pure lepidic growth or predominant lepidic growth with ≤ 5 mm of stromal invasion. BAC has unique epidemiologic, pathologic, and clinical features compared with other NSCLC subtypes. For example, although it is smoking-related, the relationship of BAC to smoking is less strong than with other types of NSCLC. About a third of patients with BAC are never-smokers.
There are also some unique radiographic features — its presentation may be confused with pneumonia or other inflammatory conditions in the lung, and only after a patient does not improve after a course of antibiotics should a diagnosis of BAC be considered. Unlike other types of lung cancer where chemotherapy may be the first plan of attack, surgery is often the first choice for treating BAC, particularly when there is no mediastinal node involvement (10%-25% of cases) or distal metastases (5% of cases). BAC usually harbors EGFR mutation. It is responsive to new targeted therapies for lung cancer, particularly osimertinib, afatinib, erlotinib, and gefitinib. Thus, people with BAC are good candidates for genetic testing.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Bronchioalveolar cell carcinoma (BAC) is a variant of non–small cell lung cancer (NSCLC) that, in recent years, has received a new identity in some of the literature. Adenocarcinoma in situ (AIS) and minimally invasive adenocarcinoma (MIA) are relatively new entities that in some published literature have replaced the term BAC. The National Comprehensive Cancer Network recognizes these terms. AIS is defined as a localized adenocarcinoma of < 3 cm that exhibits a lepidic growth pattern, with neoplastic cells along the alveolar structures but without stromal, vascular, or pleural invasion. MIA refers to small, solitary adenocarcinomas < 3 cm with either pure lepidic growth or predominant lepidic growth with ≤ 5 mm of stromal invasion. BAC has unique epidemiologic, pathologic, and clinical features compared with other NSCLC subtypes. For example, although it is smoking-related, the relationship of BAC to smoking is less strong than with other types of NSCLC. About a third of patients with BAC are never-smokers.
There are also some unique radiographic features — its presentation may be confused with pneumonia or other inflammatory conditions in the lung, and only after a patient does not improve after a course of antibiotics should a diagnosis of BAC be considered. Unlike other types of lung cancer where chemotherapy may be the first plan of attack, surgery is often the first choice for treating BAC, particularly when there is no mediastinal node involvement (10%-25% of cases) or distal metastases (5% of cases). BAC usually harbors EGFR mutation. It is responsive to new targeted therapies for lung cancer, particularly osimertinib, afatinib, erlotinib, and gefitinib. Thus, people with BAC are good candidates for genetic testing.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
A 50-year-old woman, a never-smoker, presented with complaints of intermittent cough and shortness of breath for 3 months, associated with bronchorrhea (copious watery sputum production). She had lost 15 pounds in the past 2 months and had dyspnea on exertion for 1 month. Her pulse rate was 88/min, respiratory rate 18/min, and oxygen saturation 96% on room air. A chest x-ray (posteroanterior view) showed dense opacity in the right lower zone. Contrast-enhanced CT of the thorax showed diffuse ground-glass opacities around nodules and consolidation involving the apical and basal segments of the right lower lobe. Despite several courses of antimicrobials, bronchodilators, and IV corticosteroid therapy, the patient's condition worsened.
SCLC: The Basics
Older men more at risk as dangerous falls rise for all seniors
When Senate Minority Leader Mitch McConnell (R-Ky.) fell recently at a dinner event in Washington, he unfortunately joined a large group of his senior citizen peers.
This wasn’t the first tumble the 81-year-old has taken. In 2019, he fell in his home, fracturing his shoulder. This time, he got a concussion and was recently released to an in-patient rehabilitation facility. While Sen. McConnell didn’t fracture his skull, in falling and hitting his head, he became part of an emerging statistic: One that reveals falls are more dangerous for senior men than senior women.
This new research, which appeared in the American Journal of Emergency Medicine, came as a surprise to lead researcher Scott Alter, MD, associate professor of emergency medicine at the Florida Atlantic University, Boca Raton.
“We always hear about lower bone density rates among females, so we didn’t expect to see males with more skull fractures,” he said.
Dr. Alter said that as a clinician in a southern Florida facility, his emergency department was the perfect study grounds to evaluate incoming geriatric patients due to falls. Older “patients are at higher risk of skull fractures and intercranial bleeding, and we wanted to look at any patient presenting with a head injury. Some 80% were fall related, however.”
The statistics bear out the fact that falls of all types are common among the elderly: Some 800,000 seniors wind up in the hospital each year because of falls.
The numbers show death rates from falls are on the rise in the senior citizen age group, too, up 30% from 2007 to 2016. Falls account for 70% of accidental deaths in people 75 and older. They are the leading cause of injury-related visits to emergency departments in the country, too.
Jennifer Stevens, MD, a gerontologist and executive director at Florida-based Abbey Delray South, is aware of the dire numbers and sees their consequences regularly. “The reasons seniors are at a high fall risk are many,” she said. “They include balance issues, declining strength, diseases like Parkinson’s and Alzheimer’s, side effects of their medications, and more.”
In addition, many seniors live in spaces that are not necessarily equipped for their limitations, and hazards exist all over their homes. Put together, and the risks for falls are everywhere. But there are steps seniors, their families, and even middle-aged people can take to mitigate and hopefully prevent dangerous falls.
Starting early
While in many cases the journey to lessen fall risks begins after a fall, the time to begin addressing the issue is long before you hit your senior years. Mary Therese Cole, a physical therapist and certified dementia practitioner at Manual Edge Physical Therapy in Colorado Springs, Colo., says that age 50 is a good time to start paying attention and addressing physical declines.
“This is an age where your vision might begin deteriorating,” she said. “It’s a big reason why elderly people trip and fall.”
As our brains begin to age in our middle years, the neural pathways from brain to extremities start to decline, too. The result is that many people stop picking up their feet as well as they used to do, making them more likely to trip.
“You’re not elderly yet, but you’re not a spring chicken, either,” Ms. Cole said. “Any issues you have now will only get worse if you’re not working on them.”
A good starting point in middle age, then, is to work on both strength training and balance exercises. A certified personal trainer or physical therapist can help get you on a program to ward off many of these declines.
If you’ve reached your later years, however, and are experiencing physical declines, it’s smart to check in with your primary care doctor for an assessment. “He or she can get your started on regular PT to evaluate any shortcomings and then address them,” Ms. Cole said.
She noted that when she’s working with senior patients, she’ll test their strength getting into and out of a chair, do a manual strength test to check on lower extremities, check their walking stride, and ask about conditions such as diabetes, former surgeries, and other conditions.
From there, Ms. Cole said she can write up a plan for the patient. Likewise, Dr. Stevens uses a program called Be Active that allows her to test seniors on a variety of measurements, including flexibility, balance, hand strength, and more.
“Then we match them with classes to address their shortcomings,” she said. “It’s critical that seniors have the ability to recover and not fall if they get knocked off balance.”
Beyond working on your physical limitations, taking a good look at your home is essential, too. “You can have an occupational therapist come to your home and do an evaluation,” Dr. Stevens said. “They can help you rearrange and reorganize for a safer environment.”
Big, common household fall hazards include throw rugs, lack of nightlights for middle-of-the-night visits to the bathroom, a lack of grab bars in the shower/bathtub, and furniture that blocks pathways.
For his part, Dr. Alter likes to point seniors and their doctors to the CDC’s STEADI program, which is aimed at stopping elderly accidents, deaths, and injuries.
“It includes screening for fall risk, assessing factors you can modify or improve, and more tools,” he said.
Dr. Alter also recommended seniors talk to their doctors about medications, particularly blood thinners.
“At a certain point, you need to weigh the benefits of disease prevention with the risk of injury if you fall,” he said. “The bleeding risk might be too high if the patient is at a high risk of falls.”
A version of this article originally appeared on WebMD.com.
When Senate Minority Leader Mitch McConnell (R-Ky.) fell recently at a dinner event in Washington, he unfortunately joined a large group of his senior citizen peers.
This wasn’t the first tumble the 81-year-old has taken. In 2019, he fell in his home, fracturing his shoulder. This time, he got a concussion and was recently released to an in-patient rehabilitation facility. While Sen. McConnell didn’t fracture his skull, in falling and hitting his head, he became part of an emerging statistic: One that reveals falls are more dangerous for senior men than senior women.
This new research, which appeared in the American Journal of Emergency Medicine, came as a surprise to lead researcher Scott Alter, MD, associate professor of emergency medicine at the Florida Atlantic University, Boca Raton.
“We always hear about lower bone density rates among females, so we didn’t expect to see males with more skull fractures,” he said.
Dr. Alter said that as a clinician in a southern Florida facility, his emergency department was the perfect study grounds to evaluate incoming geriatric patients due to falls. Older “patients are at higher risk of skull fractures and intercranial bleeding, and we wanted to look at any patient presenting with a head injury. Some 80% were fall related, however.”
The statistics bear out the fact that falls of all types are common among the elderly: Some 800,000 seniors wind up in the hospital each year because of falls.
The numbers show death rates from falls are on the rise in the senior citizen age group, too, up 30% from 2007 to 2016. Falls account for 70% of accidental deaths in people 75 and older. They are the leading cause of injury-related visits to emergency departments in the country, too.
Jennifer Stevens, MD, a gerontologist and executive director at Florida-based Abbey Delray South, is aware of the dire numbers and sees their consequences regularly. “The reasons seniors are at a high fall risk are many,” she said. “They include balance issues, declining strength, diseases like Parkinson’s and Alzheimer’s, side effects of their medications, and more.”
In addition, many seniors live in spaces that are not necessarily equipped for their limitations, and hazards exist all over their homes. Put together, and the risks for falls are everywhere. But there are steps seniors, their families, and even middle-aged people can take to mitigate and hopefully prevent dangerous falls.
Starting early
While in many cases the journey to lessen fall risks begins after a fall, the time to begin addressing the issue is long before you hit your senior years. Mary Therese Cole, a physical therapist and certified dementia practitioner at Manual Edge Physical Therapy in Colorado Springs, Colo., says that age 50 is a good time to start paying attention and addressing physical declines.
“This is an age where your vision might begin deteriorating,” she said. “It’s a big reason why elderly people trip and fall.”
As our brains begin to age in our middle years, the neural pathways from brain to extremities start to decline, too. The result is that many people stop picking up their feet as well as they used to do, making them more likely to trip.
“You’re not elderly yet, but you’re not a spring chicken, either,” Ms. Cole said. “Any issues you have now will only get worse if you’re not working on them.”
A good starting point in middle age, then, is to work on both strength training and balance exercises. A certified personal trainer or physical therapist can help get you on a program to ward off many of these declines.
If you’ve reached your later years, however, and are experiencing physical declines, it’s smart to check in with your primary care doctor for an assessment. “He or she can get your started on regular PT to evaluate any shortcomings and then address them,” Ms. Cole said.
She noted that when she’s working with senior patients, she’ll test their strength getting into and out of a chair, do a manual strength test to check on lower extremities, check their walking stride, and ask about conditions such as diabetes, former surgeries, and other conditions.
From there, Ms. Cole said she can write up a plan for the patient. Likewise, Dr. Stevens uses a program called Be Active that allows her to test seniors on a variety of measurements, including flexibility, balance, hand strength, and more.
“Then we match them with classes to address their shortcomings,” she said. “It’s critical that seniors have the ability to recover and not fall if they get knocked off balance.”
Beyond working on your physical limitations, taking a good look at your home is essential, too. “You can have an occupational therapist come to your home and do an evaluation,” Dr. Stevens said. “They can help you rearrange and reorganize for a safer environment.”
Big, common household fall hazards include throw rugs, lack of nightlights for middle-of-the-night visits to the bathroom, a lack of grab bars in the shower/bathtub, and furniture that blocks pathways.
For his part, Dr. Alter likes to point seniors and their doctors to the CDC’s STEADI program, which is aimed at stopping elderly accidents, deaths, and injuries.
“It includes screening for fall risk, assessing factors you can modify or improve, and more tools,” he said.
Dr. Alter also recommended seniors talk to their doctors about medications, particularly blood thinners.
“At a certain point, you need to weigh the benefits of disease prevention with the risk of injury if you fall,” he said. “The bleeding risk might be too high if the patient is at a high risk of falls.”
A version of this article originally appeared on WebMD.com.
When Senate Minority Leader Mitch McConnell (R-Ky.) fell recently at a dinner event in Washington, he unfortunately joined a large group of his senior citizen peers.
This wasn’t the first tumble the 81-year-old has taken. In 2019, he fell in his home, fracturing his shoulder. This time, he got a concussion and was recently released to an in-patient rehabilitation facility. While Sen. McConnell didn’t fracture his skull, in falling and hitting his head, he became part of an emerging statistic: One that reveals falls are more dangerous for senior men than senior women.
This new research, which appeared in the American Journal of Emergency Medicine, came as a surprise to lead researcher Scott Alter, MD, associate professor of emergency medicine at the Florida Atlantic University, Boca Raton.
“We always hear about lower bone density rates among females, so we didn’t expect to see males with more skull fractures,” he said.
Dr. Alter said that as a clinician in a southern Florida facility, his emergency department was the perfect study grounds to evaluate incoming geriatric patients due to falls. Older “patients are at higher risk of skull fractures and intercranial bleeding, and we wanted to look at any patient presenting with a head injury. Some 80% were fall related, however.”
The statistics bear out the fact that falls of all types are common among the elderly: Some 800,000 seniors wind up in the hospital each year because of falls.
The numbers show death rates from falls are on the rise in the senior citizen age group, too, up 30% from 2007 to 2016. Falls account for 70% of accidental deaths in people 75 and older. They are the leading cause of injury-related visits to emergency departments in the country, too.
Jennifer Stevens, MD, a gerontologist and executive director at Florida-based Abbey Delray South, is aware of the dire numbers and sees their consequences regularly. “The reasons seniors are at a high fall risk are many,” she said. “They include balance issues, declining strength, diseases like Parkinson’s and Alzheimer’s, side effects of their medications, and more.”
In addition, many seniors live in spaces that are not necessarily equipped for their limitations, and hazards exist all over their homes. Put together, and the risks for falls are everywhere. But there are steps seniors, their families, and even middle-aged people can take to mitigate and hopefully prevent dangerous falls.
Starting early
While in many cases the journey to lessen fall risks begins after a fall, the time to begin addressing the issue is long before you hit your senior years. Mary Therese Cole, a physical therapist and certified dementia practitioner at Manual Edge Physical Therapy in Colorado Springs, Colo., says that age 50 is a good time to start paying attention and addressing physical declines.
“This is an age where your vision might begin deteriorating,” she said. “It’s a big reason why elderly people trip and fall.”
As our brains begin to age in our middle years, the neural pathways from brain to extremities start to decline, too. The result is that many people stop picking up their feet as well as they used to do, making them more likely to trip.
“You’re not elderly yet, but you’re not a spring chicken, either,” Ms. Cole said. “Any issues you have now will only get worse if you’re not working on them.”
A good starting point in middle age, then, is to work on both strength training and balance exercises. A certified personal trainer or physical therapist can help get you on a program to ward off many of these declines.
If you’ve reached your later years, however, and are experiencing physical declines, it’s smart to check in with your primary care doctor for an assessment. “He or she can get your started on regular PT to evaluate any shortcomings and then address them,” Ms. Cole said.
She noted that when she’s working with senior patients, she’ll test their strength getting into and out of a chair, do a manual strength test to check on lower extremities, check their walking stride, and ask about conditions such as diabetes, former surgeries, and other conditions.
From there, Ms. Cole said she can write up a plan for the patient. Likewise, Dr. Stevens uses a program called Be Active that allows her to test seniors on a variety of measurements, including flexibility, balance, hand strength, and more.
“Then we match them with classes to address their shortcomings,” she said. “It’s critical that seniors have the ability to recover and not fall if they get knocked off balance.”
Beyond working on your physical limitations, taking a good look at your home is essential, too. “You can have an occupational therapist come to your home and do an evaluation,” Dr. Stevens said. “They can help you rearrange and reorganize for a safer environment.”
Big, common household fall hazards include throw rugs, lack of nightlights for middle-of-the-night visits to the bathroom, a lack of grab bars in the shower/bathtub, and furniture that blocks pathways.
For his part, Dr. Alter likes to point seniors and their doctors to the CDC’s STEADI program, which is aimed at stopping elderly accidents, deaths, and injuries.
“It includes screening for fall risk, assessing factors you can modify or improve, and more tools,” he said.
Dr. Alter also recommended seniors talk to their doctors about medications, particularly blood thinners.
“At a certain point, you need to weigh the benefits of disease prevention with the risk of injury if you fall,” he said. “The bleeding risk might be too high if the patient is at a high risk of falls.”
A version of this article originally appeared on WebMD.com.
Melanoma screening: Consensus statement offers greater clarity
That is why a group of expert panelists evaluated the existing evidence and a range of clinical scenarios to help clarify the optimal strategies for early detection and assessment of cutaneous melanoma.
Overall, the panelists agreed that a risk-stratified approach is likely the most appropriate strategy for melanoma screening and follow-up and supported the use of visual and dermoscopic examination. However, the panelists did not reach consensus on the role for gene expression profile (GEP) testing in clinical decision-making, citing the need for these assays to be validated in large randomized clinical trials.
In an accompanying editorial, two experts highlighted the importance of carefully evaluating the role of diagnostic tests.
“Diagnostic tests such as GEP must face critical scrutiny; if not, there are immediate concerns for patient care, such as the patient being erroneously informed that they do not have cancer or told that they do have cancer when they do not,” write Alan C. Geller, MPH, RN, from the Harvard T.H. Chan School of Public Health, Boston, and Marvin A. Weinstock, MD, PhD, from Brown University, Providence, R.I.
The consensus statement was published online in JAMA Dermatology.
The need for guidance
Although focusing melanoma screening on higher-risk populations may be cost effective, compared with population-based screening, the major guidelines lack consistent guidance to support a risk-stratified approach to skin cancer screening and best practices on diagnosing cutaneous melanoma.
In the prebiopsy setting, the appropriate use of diagnostic tools for evaluating the need for biopsy remain poorly defined, and, in the post-biopsy setting, questions remain concerning the diagnostic accuracy of molecular techniques, diagnostic GEP testing, next-generation sequencing, and immunohistochemical assessment for various markers of melanoma.
To provide consensus recommendations on optimal screening practices, prebiopsy and postbiopsy diagnostics, and prognostic assessment of cutaneous melanoma, a group of 42 panelists voted on hypothetical scenarios via an emailed survey. The panel then came together for a consensus conference, which included 51 experts who discussed their approach to the various clinical case scenarios. Most attendees (45 of the 51) answered a follow-up survey for their final recommendations.
The panelists reached a consensus, with 70% agreement, to support a risk-stratified approach to melanoma screening in clinical settings and public screening events. The experts agreed that higher-risk individuals (those with a relative risk of 5 or greater) could be appropriately screened by a general dermatologist or pigmented lesion evaluation. Higher-risk individuals included those with severe skin damage from the sun, systemic immunosuppression, or a personal history of nonmelanoma or melanoma skin cancer.
Panelists agreed that those at general or lower risk (RR < 2) could be screened by a primary care provider or through regular self- or partner examinations, whereas those at moderate risk could be screened by their primary care clinician or general dermatologist. The experts observed “a shift in acceptance” of primary care physicians screening the general population, and an acknowledgement of the importance of self- and partner examinations as screening adjuncts for all populations.
In the prebiopsy setting, panelists reached consensus that visual and dermoscopic examination was appropriate for evaluating patients with “no new, changing, or unusual skin lesions or with a new lesion that is not visually concerning.”
The panelists also reached consensus that lesions deemed clinically suspicious for cancer or showing features of cancer on reflectance confocal microscopy should be biopsied. Although most respondents (86%) did not currently use epidermal tape stripping routinely, they agreed that, in a hypothetical situation where epidermal tape stripping was used, that lesions positive for PRAME or LINC should be biopsied.
In the postbiopsy setting, views on the use of GEP scores varied. Although panelists agreed that a low-risk prognostic GEP score should not outweigh concerning histologic features when patients are selected to undergo sentinel lymph node biopsy (SLNB), they did not reach consensus for imaging recommendations in the setting of a high-risk prognostic GEP score and low-risk histology and/or negative nodal status.
“The panelists await future, well-designed prospective studies to determine if use of these and newer technologies improves the care of patients with melanoma,” the panelists write.
In the editorial, Mr. Geller and Dr. Weinstock highlighted concerns about the cost and potential access issues associated with these newer technologies, given that the current cost of GEP testing exceeds $7,000.
The editorialists also emphasize that “going forward, the field should be advanced by tackling one of the more pressing, common, potentially morbid, and costly procedures – the prognostic use of sentinel lymph node biopsy.”
Of critical importance is “whether GEP can reduce morbidity and cost by safely reducing the number of SLNBs performed,” Mr. Geller and Dr. Weinstock write.
The funding for the administration and facilitation of the consensus development conference and the development of the manuscript was provided by Dermtech, in an unrestricted award overseen by the Melanoma Research Foundation and managed and executed at UPMC by the principal investigator. Several of the coauthors disclosed relationships with industry. Mr. Geller is a contributor to UptoDate for which he receives royalties. Dr. Weinstock receives consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
That is why a group of expert panelists evaluated the existing evidence and a range of clinical scenarios to help clarify the optimal strategies for early detection and assessment of cutaneous melanoma.
Overall, the panelists agreed that a risk-stratified approach is likely the most appropriate strategy for melanoma screening and follow-up and supported the use of visual and dermoscopic examination. However, the panelists did not reach consensus on the role for gene expression profile (GEP) testing in clinical decision-making, citing the need for these assays to be validated in large randomized clinical trials.
In an accompanying editorial, two experts highlighted the importance of carefully evaluating the role of diagnostic tests.
“Diagnostic tests such as GEP must face critical scrutiny; if not, there are immediate concerns for patient care, such as the patient being erroneously informed that they do not have cancer or told that they do have cancer when they do not,” write Alan C. Geller, MPH, RN, from the Harvard T.H. Chan School of Public Health, Boston, and Marvin A. Weinstock, MD, PhD, from Brown University, Providence, R.I.
The consensus statement was published online in JAMA Dermatology.
The need for guidance
Although focusing melanoma screening on higher-risk populations may be cost effective, compared with population-based screening, the major guidelines lack consistent guidance to support a risk-stratified approach to skin cancer screening and best practices on diagnosing cutaneous melanoma.
In the prebiopsy setting, the appropriate use of diagnostic tools for evaluating the need for biopsy remain poorly defined, and, in the post-biopsy setting, questions remain concerning the diagnostic accuracy of molecular techniques, diagnostic GEP testing, next-generation sequencing, and immunohistochemical assessment for various markers of melanoma.
To provide consensus recommendations on optimal screening practices, prebiopsy and postbiopsy diagnostics, and prognostic assessment of cutaneous melanoma, a group of 42 panelists voted on hypothetical scenarios via an emailed survey. The panel then came together for a consensus conference, which included 51 experts who discussed their approach to the various clinical case scenarios. Most attendees (45 of the 51) answered a follow-up survey for their final recommendations.
The panelists reached a consensus, with 70% agreement, to support a risk-stratified approach to melanoma screening in clinical settings and public screening events. The experts agreed that higher-risk individuals (those with a relative risk of 5 or greater) could be appropriately screened by a general dermatologist or pigmented lesion evaluation. Higher-risk individuals included those with severe skin damage from the sun, systemic immunosuppression, or a personal history of nonmelanoma or melanoma skin cancer.
Panelists agreed that those at general or lower risk (RR < 2) could be screened by a primary care provider or through regular self- or partner examinations, whereas those at moderate risk could be screened by their primary care clinician or general dermatologist. The experts observed “a shift in acceptance” of primary care physicians screening the general population, and an acknowledgement of the importance of self- and partner examinations as screening adjuncts for all populations.
In the prebiopsy setting, panelists reached consensus that visual and dermoscopic examination was appropriate for evaluating patients with “no new, changing, or unusual skin lesions or with a new lesion that is not visually concerning.”
The panelists also reached consensus that lesions deemed clinically suspicious for cancer or showing features of cancer on reflectance confocal microscopy should be biopsied. Although most respondents (86%) did not currently use epidermal tape stripping routinely, they agreed that, in a hypothetical situation where epidermal tape stripping was used, that lesions positive for PRAME or LINC should be biopsied.
In the postbiopsy setting, views on the use of GEP scores varied. Although panelists agreed that a low-risk prognostic GEP score should not outweigh concerning histologic features when patients are selected to undergo sentinel lymph node biopsy (SLNB), they did not reach consensus for imaging recommendations in the setting of a high-risk prognostic GEP score and low-risk histology and/or negative nodal status.
“The panelists await future, well-designed prospective studies to determine if use of these and newer technologies improves the care of patients with melanoma,” the panelists write.
In the editorial, Mr. Geller and Dr. Weinstock highlighted concerns about the cost and potential access issues associated with these newer technologies, given that the current cost of GEP testing exceeds $7,000.
The editorialists also emphasize that “going forward, the field should be advanced by tackling one of the more pressing, common, potentially morbid, and costly procedures – the prognostic use of sentinel lymph node biopsy.”
Of critical importance is “whether GEP can reduce morbidity and cost by safely reducing the number of SLNBs performed,” Mr. Geller and Dr. Weinstock write.
The funding for the administration and facilitation of the consensus development conference and the development of the manuscript was provided by Dermtech, in an unrestricted award overseen by the Melanoma Research Foundation and managed and executed at UPMC by the principal investigator. Several of the coauthors disclosed relationships with industry. Mr. Geller is a contributor to UptoDate for which he receives royalties. Dr. Weinstock receives consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
That is why a group of expert panelists evaluated the existing evidence and a range of clinical scenarios to help clarify the optimal strategies for early detection and assessment of cutaneous melanoma.
Overall, the panelists agreed that a risk-stratified approach is likely the most appropriate strategy for melanoma screening and follow-up and supported the use of visual and dermoscopic examination. However, the panelists did not reach consensus on the role for gene expression profile (GEP) testing in clinical decision-making, citing the need for these assays to be validated in large randomized clinical trials.
In an accompanying editorial, two experts highlighted the importance of carefully evaluating the role of diagnostic tests.
“Diagnostic tests such as GEP must face critical scrutiny; if not, there are immediate concerns for patient care, such as the patient being erroneously informed that they do not have cancer or told that they do have cancer when they do not,” write Alan C. Geller, MPH, RN, from the Harvard T.H. Chan School of Public Health, Boston, and Marvin A. Weinstock, MD, PhD, from Brown University, Providence, R.I.
The consensus statement was published online in JAMA Dermatology.
The need for guidance
Although focusing melanoma screening on higher-risk populations may be cost effective, compared with population-based screening, the major guidelines lack consistent guidance to support a risk-stratified approach to skin cancer screening and best practices on diagnosing cutaneous melanoma.
In the prebiopsy setting, the appropriate use of diagnostic tools for evaluating the need for biopsy remain poorly defined, and, in the post-biopsy setting, questions remain concerning the diagnostic accuracy of molecular techniques, diagnostic GEP testing, next-generation sequencing, and immunohistochemical assessment for various markers of melanoma.
To provide consensus recommendations on optimal screening practices, prebiopsy and postbiopsy diagnostics, and prognostic assessment of cutaneous melanoma, a group of 42 panelists voted on hypothetical scenarios via an emailed survey. The panel then came together for a consensus conference, which included 51 experts who discussed their approach to the various clinical case scenarios. Most attendees (45 of the 51) answered a follow-up survey for their final recommendations.
The panelists reached a consensus, with 70% agreement, to support a risk-stratified approach to melanoma screening in clinical settings and public screening events. The experts agreed that higher-risk individuals (those with a relative risk of 5 or greater) could be appropriately screened by a general dermatologist or pigmented lesion evaluation. Higher-risk individuals included those with severe skin damage from the sun, systemic immunosuppression, or a personal history of nonmelanoma or melanoma skin cancer.
Panelists agreed that those at general or lower risk (RR < 2) could be screened by a primary care provider or through regular self- or partner examinations, whereas those at moderate risk could be screened by their primary care clinician or general dermatologist. The experts observed “a shift in acceptance” of primary care physicians screening the general population, and an acknowledgement of the importance of self- and partner examinations as screening adjuncts for all populations.
In the prebiopsy setting, panelists reached consensus that visual and dermoscopic examination was appropriate for evaluating patients with “no new, changing, or unusual skin lesions or with a new lesion that is not visually concerning.”
The panelists also reached consensus that lesions deemed clinically suspicious for cancer or showing features of cancer on reflectance confocal microscopy should be biopsied. Although most respondents (86%) did not currently use epidermal tape stripping routinely, they agreed that, in a hypothetical situation where epidermal tape stripping was used, that lesions positive for PRAME or LINC should be biopsied.
In the postbiopsy setting, views on the use of GEP scores varied. Although panelists agreed that a low-risk prognostic GEP score should not outweigh concerning histologic features when patients are selected to undergo sentinel lymph node biopsy (SLNB), they did not reach consensus for imaging recommendations in the setting of a high-risk prognostic GEP score and low-risk histology and/or negative nodal status.
“The panelists await future, well-designed prospective studies to determine if use of these and newer technologies improves the care of patients with melanoma,” the panelists write.
In the editorial, Mr. Geller and Dr. Weinstock highlighted concerns about the cost and potential access issues associated with these newer technologies, given that the current cost of GEP testing exceeds $7,000.
The editorialists also emphasize that “going forward, the field should be advanced by tackling one of the more pressing, common, potentially morbid, and costly procedures – the prognostic use of sentinel lymph node biopsy.”
Of critical importance is “whether GEP can reduce morbidity and cost by safely reducing the number of SLNBs performed,” Mr. Geller and Dr. Weinstock write.
The funding for the administration and facilitation of the consensus development conference and the development of the manuscript was provided by Dermtech, in an unrestricted award overseen by the Melanoma Research Foundation and managed and executed at UPMC by the principal investigator. Several of the coauthors disclosed relationships with industry. Mr. Geller is a contributor to UptoDate for which he receives royalties. Dr. Weinstock receives consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
A love letter to Black birthing people from Black birth workers, midwives, and physicians
A few years ago, my partner emailed me about a consult.
“Dr. Carter, I had the pleasure of seeing Mrs. Smith today for a preconception consult for chronic hypertension. As a high-risk Black woman, she wants to know what we’re going to do to make sure that she doesn’t die in pregnancy or childbirth. I told her that you’re better equipped to answer this question.”
I was early in my career, and the only thing I could assume that equipped me to answer this question over my partners was my identity as a Black woman living in America.
Mrs. Smith was copied on the message and replied with a long list of follow-up questions and a request for an in-person meeting with me. I was conflicted. As a friend, daughter, and mother, I understood her fear and wanted to be there for her. As a newly appointed assistant professor on the tenure track with 20% clinical time, my clinical responsibilities easily exceeded 50% (in part, because I failed to set boundaries). I spent countless hours of uncompensated time serving on diversity, equity, and inclusion initiatives and mentoring and volunteering for multiple community organizations; I was acutely aware that I would be measured against colleagues who rise through the ranks, unencumbered by these social, moral, and ethical responsibilities, collectively known as the “Black tax.”1
I knew from prior experiences and the tone of Mrs. Smith’s email that it would be a tough, long meeting that would set a precedent of concierge level care that only promised to intensify once she became pregnant. I agonized over my reply. How could I balance providing compassionate care for this patient with my young research program, which I hoped to nurture so that it would one day grow to have population-level impact?
It took me 2 days to finally reply to the message with a kind, but firm, email stating that I would be happy to see her for a follow-up preconception visit. It was my attempt to balance accessibility with boundaries. She did not reply.
Did I fail her?
The fact that I still think of Mrs. Smith may indicate that I did the wrong thing. In fact, writing the first draft of this letter was a therapeutic experience, and I addressed it to Mrs. Smith. As I shared the experience and letter with friends in the field, however, everyone had similar stories. The letter continued to pass between colleagues, who each made it infinitely better. This collective process created the beautiful love letter to Black birthing people that we share here.
We call upon all of our obstetric clinician colleagues to educate themselves to be equally, ethically, and equitably equipped to care for and serve historically marginalized women and birthing people. We hope that this letter will aid in the journey, and we encourage you to share it with patients to open conversations that are too often left closed.

Continue to: Our love letter to Black women and birthing people...
Our love letter to Black women and birthing people
We see you, we hear you, we know you are scared, and we are you. In recent years, the press has amplified gross inequities in maternal care and outcomes that we, as Black birth workers, midwives, and physicians, already knew to be true. We grieve, along with you regarding the recently reported pregnancy-related deaths of Mrs. Kira Johnson,2 Dr. Shalon Irving,3 Dr. Chaniece Wallace,4 and so many other names we do not know because their stories did not receive national attention, but we know that they represented the best of us, and they are gone too soon. As Black birth workers, midwives, physicians, and more, we have a front-row seat to the United States’ serious obstetric racism, manifested in biased clinical interactions, unjust hospital policies, and an inequitable health care system that leads to disparities in maternal morbidity and mortality for Black women.
Unfortunately, this is not anything new, and the legacy dates back to slavery and the disregard for Black people in this country. What has changed is our increased awareness of these health injustices. This collective consciousness of the risk that is carried with our pregnancies casts a shadow of fear over a period that should be full of the joy and promise of new life. We fear that our personhood will be disregarded, our pain will be ignored, and our voices silenced by a medical system that has sought to dominate our bodies and experiment on them without our permission.5 While this history is reprehensible, and our collective risk as Black people is disproportionately high, our purpose in writing this letter is to help Black birthing people recapture the joy and celebration that should be theirs in pregnancy and in the journey to parenthood.
As Black birth workers, we see Black pregnant patients desperately seeking safety, security, and breaking down barriers to find us for their pregnancy care. Often, they are terrified and looking for kinship and community in our offices. In rural areas patients may drive up to 4 hours in distance for an appointment, and during appointments entrust us with their stories of feeling unheard in the medical system. When we anecdotally asked about what they feared about pregnancy, childbirth, and the postpartum period and thought was their risk of dying during pregnancy or childbirth, answers ranged from 1% to 60%. Our actual risk of dying from a pregnancy-related cause, as a Black woman, is 0.0414% (41.4 Black maternal deaths per 100,000 live births).6 To put that in perspective, our risk of dying is higher walking down the street or driving a car.7
What is the source of the fear? Based on past and present injustices inflicted on people with historically marginalized identities, we have every right to be scared; but, make no mistake that fear comes at a cost, and Black birthing people are the ones paying the bill! Stress and chronic worry are associated with poor pregnancy outcomes, and so this completely justifiable fear, at the population level, is not serving us well personally.8 Unfortunately, lost in the messaging about racial inequities in maternal mortality is the reality that the vast majority of Black people and babies will survive, thrive, and have healthy pregnancy outcomes, despite the terrifying population-level statistics and horrific stories of discrimination and neglect that make us feel like our pregnancies and personal peril are synonymous.
While it is true that our absolute individual, personal risk is lower than population-level statistics convey, let us be clear: We are furious about what is happening to Black people! It is immoral that Black patients in the richest country in the world are 3-4 times more likely to die of a pregnancy-related cause than White women,9 and we are more likely to experience pregnancy complications and “near misses” when death is narrowly avoided. Research has done an excellent job defining reproductive health disparities in this country, but prioritizing and funding meaningful strategies, policies, and programs to close this gap have not taken precedence—especially initiatives and research that are headed by Black women.10–12 This is largely because researchers and health care systems continue evaluating strategies that focus on behavior change and narratives that identify individual responsibility as a sole cause of inequity.
Let us be clear, Black people and our behaviors are not the problem.13 The problems are White supremacy, classism, sexism, heteropatriarchy, and obstetric racism.1-21 These must be recognized and addressed across all levels of power. We endorse systems-level changes that are at the root of promoting health equity in our reproductive outcomes. These changes include paid parental leave, Medicaid expansion/extension, reimbursement for doula and lactation services, increased access to perinatal mental health and wellness services, and so much more. (See the Black Mamas Matter Alliance Toolkit: https://blackmamas matter.org/our-work/toolkits/.)
Continue to: Pearls for reassurance...
Pearls for reassurance
While the inequities and their solutions are grounded in the need for systemic change,22 we realize that these population-level solutions feel abstract when our sisters and siblings ask us, “So what can I do to advocate for myself and my baby, right now in this pregnancy?” To be clear, no amount of personal hypervigilance on our part as Black pregnancy-capable people is going to fix these problems, which are systemic; however, we want to provide a few pearls that may be helpful for patient self-advocacy and reassurance:
- Seek culturally and ethnically congruent care. We intuitively want to find a clinician who looks like us, but sadly, in the United States only 5% of physicians and 2% of midwives are Black. Demand exceeds supply for Black patients who are seeking racially congruent care. Nonetheless, it is critical that you find a physician or midwife who centers you and provides support and care that affirms the strengths and assets of you, your family, and your community when cultural and ethnic congruency are not possible for you and your pregnancy.
- Ask how your clinicians are actively working to ensure optimal and equitable experiences for Black birthing individuals. We recommend asking your clinician and/or hospital what, if anything, they are doing to address health care inequities, obstetric racism, or implicit bias in their pregnancy and postpartum care. Many groups (including some authors of this letter) are working on measures to address obstetric racism. An acknowledgement of initiatives to mitigate inequities is a meaningful first step. You can suggest that they look into it while you explore your options, as this work is rapidly emerging in many areas of the country.
- Plan for well-person care. The best time to optimize pregnancy and birth outcomes is before you get pregnant. Set up an appointment with a midwife, ObGyn, or your primary care physician before you get pregnant. Discuss your concerns about pregnancy and use this time to optimize your health. This also provides an opportunity to build a relationship with your physician/ midwife and their group to evaluate whether they curate an environment where you feel seen, heard, and valued when you go for annual exams or problem visits. If you do not get that sense after a couple of visits, find a place where you do.
- Advocate for a second opinion. If something does not sound right to you or you have questions that were not adequately answered, it is your prerogative to seek a second opinion; a clinician should never be offended by this.
- Consider these factors, for those who deliver in a hospital (by choice or necessity):
a. 24/7 access to obstetricians and dedicated anesthesiologists in the hospital
b. trauma-informed medical/mental health/social services
c. lactation consultation
d. supportive trial of labor after cesarean delivery policy
e. massive blood transfusion protocol.
- Seek doula support! It always helps to have another set of eyes and ears to help advocate for you, especially when you are in pain during pregnancy, childbirth, or in the postpartum period, or are having difficulty advocating for yourself. There is also evidence that women supported by doulas have better pregnancy-related outcomes and experiences.23 Many major cities in the United States have started to provide race-concordant doula care for Black birthing people for free.24
- Don’t forget about your mental health. As stated, chronic stress from racism impacts birth outcomes. Having a mental health clinician is a great way to mitigate adverse effects of prolonged tension.25–27
- Ask your clinician, hospital, or insurance company about participating in group prenatal care and/or nurse home visiting models28 because both are associated with improved birth outcomes.29 Many institutions are implementing group care that provides race-concordant care.30,31
- Ask your clinician, hospital, or local health department for recommendations to a lactation consultant or educator who can support your efforts in breast/ chest/body-feeding.
We invite you to consider this truth
You, alone, do not carry the entire population-level risk of Black birthing people on your shoulders. We all carry a piece of it. We, along with many allies, advocates, and activists, are outraged and angered by generations of racism and mistreatment of Black birthing people in our health systems and hospitals. We are channeling our frustration and disgust to demand substantive and sustainable change.
Our purpose here is to provide love and reassurance to our sisters and siblings who are going through their pregnancies with thoughts about our nation’s past and present failures to promote health equity for us and our babies. Our purpose is neither to minimize the public health crisis of Black infant and maternal morbidity and mortality nor is it to absolve clinicians, health systems, or governments from taking responsibility for these shameful outcomes or making meaningful changes to address them. In fact, we love taking care of our community by providing the best clinical care we can to our patients. We call upon all of our clinical colleagues to educate themselves to be ethically and equitably equipped to provide health care for Black pregnant patients. Finally, to birthing Black families, please remember this: If you choose to have a baby, the outcome and experience must align with what is right for you and your baby to survive and thrive. So much of the joys of pregnancy have been stolen, but we will recapture the celebration that should be ours in pregnancy and the journey to parenthood.
Sincerely,
Ebony B. Carter, MD, MPH
Maternal Fetal Medicine
Washington University School of Medicine
St. Louis, Missouri
Karen A. Scott, MD, MPH
Birthing Cultural Rigor, LLC
Nashville, Tennessee
Andrea Jackson, MD, MAS
ObGyn
University of California,
San Francisco
Sara Whetstone, MD, MHS
ObGyn
University of California,
San Francisco
Traci Johnson, MD
ObGyn
University of Missouri
School of Medicine
Kansas City, Missouri
Sarahn Wheeler, MD
Maternal Fetal Medicine
Duke University School of Medicine
Durham, North Carolina
Asmara Gebre, CNM
Midwife
Zuckerberg San Francisco General Hospital
San Francisco, California
Joia Crear-Perry, MD
ObGyn
National Birth Equity Collaborative
New Orleans, Louisiana
Dineo Khabele, MD
Gynecologic Oncology
Washington University School of Medicine
St. Louis, Missouri
Judette Louis, MD, MPH
Maternal Fetal Medicine
University of South Florida College of Medicine
Tampa, Florida
Yvonne Smith, MSN, RN
Director
Barnes-Jewish Hospital
St. Louis, Missouri
Laura Riley, MD
Maternal Fetal Medicine
Weill Cornell Medicine
New York, New York
Antoinette Liddell, MSN, RN
Care Coordinator
Barnes-Jewish Hospital
St. Louis, Missouri
Cynthia Gyamfi-Bannerman, MD
Maternal Fetal Medicine
Columbia University Irving Medical Center
New York, New York
Rasheda Pippens, MSN, RN
Nurse Educator
Barnes-Jewish Hospital
St. Louis, Missouri
Ayaba Worjoloh-Clemens, MD
ObGyn
Atlanta, Georgia
Allison Bryant, MD, MPH
Maternal Fetal Medicine
Massachusetts General Hospital
Boston, Massachusetts
Sheri L. Foote, CNM
Midwife
Zuckerberg San Francisco General Hospital
San Francisco, California
J. Lindsay Sillas, MD
ObGyn
Bella OB/GYN
Houston, Texas
Cynthia Rogers, MD
Psychiatrist
Washington University School of Medicine
St. Louis, Missouri
Audra R. Meadows, MD, MPH
ObGyn
University of California, San Diego
AeuMuro G. Lake, MD
Urogynecologist
Urogynecology and Healing Arts
Seattle, Washington
Nancy Moore, MSN, RN, WHNP-BC
Nurse Practitioner
Barnes-Jewish Hospital
St. Louis, Missouri
Zoë Julian, MD, MPH
ObGyn
University of Alabama at Birmingham
Janice M. Tinsley, MN, RNC-OB
Zuckerberg San Francisco General Hospital
San Francisco, California
Jamila B. Perritt, MD, MPH
ObGyn
Washington, DC
Joy A. Cooper, MD, MSc
ObGyn
Culture Care
Oakland, California
Arthurine K. Zakama, MD
ObGyn
University of California,San Francisco
Alissa Erogbogbo, MD
OB Hospitalist
Los Altos, California
Sanithia L. Williams, MD
ObGyn
Huntsville, Alabama
Audra Williams, MD, MPH
ObGyn
University of Alabama, Birmingham
Hedwige “Didi” Saint Louis, MD, MPH
OB Hospitalist
Morehouse School of Medicine
Atlanta, Georgia
Cherise Cokley, MD
OB Hospitalist
Community Hospital
Munster, Indiana
J’Leise Sosa, MD, MPH
ObGyn
Buffalo, New York
- Rodríguez JE, Campbell KM, Pololi LH. Addressing disparities in academic medicine: what of the minority tax? BMC Med Educ. 2015;15:6. https ://doi.org/10.1186/s12909-015-0290-9.
- Helm A. Yet another beautiful Black woman dies in childbirth. Kira Johnson spoke 5 languages, raced cars, was daughter in law of Judge Glenda Hatchett. She still died in childbirth. October 19, 2018. https://www.theroot.com/kira-johnson-spoke- 5-languages-raced-cars-was-daughter-18298 62323. Accessed February 27, 2027.
- Shock after Black pediatrics doctor dies after giving birth to first child. November 6, 2020. https ://www.bet.com/article/rvyskv/black-pediatrics -doctor-dies-after-giving-birth#! Accessed February 24, 2023.
- Dr. Shalon’s maternal action project. https ://www.drshalonsmap.org/. Accessed February 24, 2023.
- Verdantam S, Penman M. Remembering Anarcha, Lucy, and Betsey: The mothers of modern gynecology. https://www.npr .org/2016/02/16/466942135/remembering -anarcha-lucy-and-betsey-the-mothers-of -modern-gynecology. February 16, 2016. Accessed February 24, 2023.
- Centers for Disease Control and Prevention website. Pregnancy Mortality Surveillance System. Last reviewed June 22, 2022. Accessed March 8, 2023.
- Odds of dying. NSC injury facts. https ://injuryfacts.nsc.org/all-injuries/preventable -death-overview/odds-of-dying/data-details /#:~:text=Statements%20about%20the%20 odds%20or%20chances%20of%20dying,in% 20%28value%20given%20in%20the%20lifetime %20odds%20column%29. Accessed February 24, 2023.
- Gembruch U, Baschat AA. True knot of the umbilical cord: transient constrictive effect to umbilical venous blood flow demonstrated by Doppler sonography. Ultrasound Obstet Gynecol. 1996;8:53-56. doi: 10.1046/j.14690705.1996.08010053.x.
- MacDorman MF, Thoma M, Declcerq E, et al. Racial and ethnic disparities in maternal mortality in the United States using enhanced vital records, 2016-2017. Am J Public Health. 2012;111:16731681.
- Taffe MA, Gilpin NW. Racial inequity in grant funding from the US National Institutes of Health. Elife. 2021;10. doi: 10.7554/eLife.65697.
- Black Women Scholars and Research Working Group for the Black Mamas Matter Alliance. Black maternal health research re-envisioned: best practices for the conduct of research with, for, and by Black mamas. Harvard Law Policy Rev. 2020;14:393.
- Sullivan P. In philanthropy, race is still a factor in who gets what, study shows. NY Times. https ://www.nytimes.com/2020/05/01/your-money /philanthropy-race.html. May 5, 2020.
- Scott KA, Britton L, McLemore MR. The ethics of perinatal care for Black women: dismantling the structural racism in “Mother Blame” narratives. J Perinat Neonatal Nurs. 2019;33:108-115. doi: 10.1097/jpn.0000000000000394.
- Dominguez TP, Dunkel-Schetter C, Glynn LM, Hobel C, Sandman CA. Racial Differences in Birth Outcomes: The Role of General, Pregnancy, and Racism Stress. Health Psychology. 2008;27(2):194203. doi: 10.1037/0278-6133.27.2.194.
- Hardeman RR, Murphy KA, Karbeah J, et al. Naming institutionalized racism in the public health literature: a systematic literature review. Public Health Rep. 2018;133:240-249. doi: 10.1177/0033354918760574.
- Hardeman RR, Karbeah J. Examining racism in health services research: a disciplinary self- critique. Health Serv Res. 2020;55 Suppl 2:777-780. doi: 10.1111/1475-6773.13558.
- Hardeman RR, Karbeah J, Kozhimannil KB. Applying a critical race lens to relationship-centered care in pregnancy and childbirth: an antidote to structural racism. Birth. 2020;47:3-7. doi: 10.1111/birt.12462.
- Scott KA, Davis D-A. Obstetric racism: naming and identifying a way out of Black women’s adverse medical experiences. Am Anthropologist. 2021;123:681-684. doi: https://doi.org/10.1111 /aman.13559.
- Mullings L. Resistance and resilience the sojourner syndrome and the social context of reproduction in central Harlem. Schulz AJ, Mullings L, eds. Gender, Race, Class, & Health: Intersectional Approaches. Jossey-Bass/Wiley: Hoboken, NJ; 2006:345-370.
- Chambers BD, Arabia SE, Arega HA, et al. Exposures to structural racism and racial discrimination among pregnant and early post-partum Black women living in Oakland, California. Stress Health. 2020;36:213-219. doi: 10.1002/smi.2922.
- Chambers BD, Arega HA, Arabia SE, et al. Black women’s perspectives on structural racism across the reproductive lifespan: a conceptual framework for measurement development. Maternal Child Health J. 2021;25:402-413. doi: 10.1007 /s10995-020-03074-3.
- Julian Z, Robles D, Whetstone S, et al. Community-informed models of perinatal and reproductive health services provision: A justice-centered paradigm toward equity among Black birthing communities. Seminar Perinatol. 2020;44:151267. doi: 10.1016/j.semperi.2020.151267.
- Bohren MA, Hofmeyr GJ, Sakala C, et al. Continuous support for women during childbirth. Cochrane Database System Rev. 2017;7:Cd003766. doi: 10.1002/14651858.CD003766.pub6.
- National Black doulas association. https://www .blackdoulas.org/. Accessed February 24, 2023.
- Therapy for Black girls. https://therapyforblack girls.com/. Accessed February 24, 2023.
- National Queer and Trans Therapists of Color Network. https://www.nqttcn.com/. Accessed February 24, 2023.
- Shades of Blue Project. http://cbww.org. Accessed February 24, 2023.
- Centering Healthcare Institute. https://www .centeringhealthcare.org/. Accessed February 24, 2023.
- Carter EB, Temming LA, Akin J, et al. Group prenatal care compared with traditional prenatal care: a systematic review and meta-analysis. Obstet Gynecol. 2016;128:551-561. doi: 10.1097 /aog.0000000000001560.
- National Center of Excellence in Women’s Health. https://womenshealth.ucsf.edu/coe/embrace -perinatal-care-black-families. Accessed February 24, 2023.
- Alameda Health System. http://www.alamedahealthsystem.org/family-birthing-center/black -centering/. Accessed February 24, 2023.
A few years ago, my partner emailed me about a consult.
“Dr. Carter, I had the pleasure of seeing Mrs. Smith today for a preconception consult for chronic hypertension. As a high-risk Black woman, she wants to know what we’re going to do to make sure that she doesn’t die in pregnancy or childbirth. I told her that you’re better equipped to answer this question.”
I was early in my career, and the only thing I could assume that equipped me to answer this question over my partners was my identity as a Black woman living in America.
Mrs. Smith was copied on the message and replied with a long list of follow-up questions and a request for an in-person meeting with me. I was conflicted. As a friend, daughter, and mother, I understood her fear and wanted to be there for her. As a newly appointed assistant professor on the tenure track with 20% clinical time, my clinical responsibilities easily exceeded 50% (in part, because I failed to set boundaries). I spent countless hours of uncompensated time serving on diversity, equity, and inclusion initiatives and mentoring and volunteering for multiple community organizations; I was acutely aware that I would be measured against colleagues who rise through the ranks, unencumbered by these social, moral, and ethical responsibilities, collectively known as the “Black tax.”1
I knew from prior experiences and the tone of Mrs. Smith’s email that it would be a tough, long meeting that would set a precedent of concierge level care that only promised to intensify once she became pregnant. I agonized over my reply. How could I balance providing compassionate care for this patient with my young research program, which I hoped to nurture so that it would one day grow to have population-level impact?
It took me 2 days to finally reply to the message with a kind, but firm, email stating that I would be happy to see her for a follow-up preconception visit. It was my attempt to balance accessibility with boundaries. She did not reply.
Did I fail her?
The fact that I still think of Mrs. Smith may indicate that I did the wrong thing. In fact, writing the first draft of this letter was a therapeutic experience, and I addressed it to Mrs. Smith. As I shared the experience and letter with friends in the field, however, everyone had similar stories. The letter continued to pass between colleagues, who each made it infinitely better. This collective process created the beautiful love letter to Black birthing people that we share here.
We call upon all of our obstetric clinician colleagues to educate themselves to be equally, ethically, and equitably equipped to care for and serve historically marginalized women and birthing people. We hope that this letter will aid in the journey, and we encourage you to share it with patients to open conversations that are too often left closed.

Continue to: Our love letter to Black women and birthing people...
Our love letter to Black women and birthing people
We see you, we hear you, we know you are scared, and we are you. In recent years, the press has amplified gross inequities in maternal care and outcomes that we, as Black birth workers, midwives, and physicians, already knew to be true. We grieve, along with you regarding the recently reported pregnancy-related deaths of Mrs. Kira Johnson,2 Dr. Shalon Irving,3 Dr. Chaniece Wallace,4 and so many other names we do not know because their stories did not receive national attention, but we know that they represented the best of us, and they are gone too soon. As Black birth workers, midwives, physicians, and more, we have a front-row seat to the United States’ serious obstetric racism, manifested in biased clinical interactions, unjust hospital policies, and an inequitable health care system that leads to disparities in maternal morbidity and mortality for Black women.
Unfortunately, this is not anything new, and the legacy dates back to slavery and the disregard for Black people in this country. What has changed is our increased awareness of these health injustices. This collective consciousness of the risk that is carried with our pregnancies casts a shadow of fear over a period that should be full of the joy and promise of new life. We fear that our personhood will be disregarded, our pain will be ignored, and our voices silenced by a medical system that has sought to dominate our bodies and experiment on them without our permission.5 While this history is reprehensible, and our collective risk as Black people is disproportionately high, our purpose in writing this letter is to help Black birthing people recapture the joy and celebration that should be theirs in pregnancy and in the journey to parenthood.
As Black birth workers, we see Black pregnant patients desperately seeking safety, security, and breaking down barriers to find us for their pregnancy care. Often, they are terrified and looking for kinship and community in our offices. In rural areas patients may drive up to 4 hours in distance for an appointment, and during appointments entrust us with their stories of feeling unheard in the medical system. When we anecdotally asked about what they feared about pregnancy, childbirth, and the postpartum period and thought was their risk of dying during pregnancy or childbirth, answers ranged from 1% to 60%. Our actual risk of dying from a pregnancy-related cause, as a Black woman, is 0.0414% (41.4 Black maternal deaths per 100,000 live births).6 To put that in perspective, our risk of dying is higher walking down the street or driving a car.7
What is the source of the fear? Based on past and present injustices inflicted on people with historically marginalized identities, we have every right to be scared; but, make no mistake that fear comes at a cost, and Black birthing people are the ones paying the bill! Stress and chronic worry are associated with poor pregnancy outcomes, and so this completely justifiable fear, at the population level, is not serving us well personally.8 Unfortunately, lost in the messaging about racial inequities in maternal mortality is the reality that the vast majority of Black people and babies will survive, thrive, and have healthy pregnancy outcomes, despite the terrifying population-level statistics and horrific stories of discrimination and neglect that make us feel like our pregnancies and personal peril are synonymous.
While it is true that our absolute individual, personal risk is lower than population-level statistics convey, let us be clear: We are furious about what is happening to Black people! It is immoral that Black patients in the richest country in the world are 3-4 times more likely to die of a pregnancy-related cause than White women,9 and we are more likely to experience pregnancy complications and “near misses” when death is narrowly avoided. Research has done an excellent job defining reproductive health disparities in this country, but prioritizing and funding meaningful strategies, policies, and programs to close this gap have not taken precedence—especially initiatives and research that are headed by Black women.10–12 This is largely because researchers and health care systems continue evaluating strategies that focus on behavior change and narratives that identify individual responsibility as a sole cause of inequity.
Let us be clear, Black people and our behaviors are not the problem.13 The problems are White supremacy, classism, sexism, heteropatriarchy, and obstetric racism.1-21 These must be recognized and addressed across all levels of power. We endorse systems-level changes that are at the root of promoting health equity in our reproductive outcomes. These changes include paid parental leave, Medicaid expansion/extension, reimbursement for doula and lactation services, increased access to perinatal mental health and wellness services, and so much more. (See the Black Mamas Matter Alliance Toolkit: https://blackmamas matter.org/our-work/toolkits/.)
Continue to: Pearls for reassurance...
Pearls for reassurance
While the inequities and their solutions are grounded in the need for systemic change,22 we realize that these population-level solutions feel abstract when our sisters and siblings ask us, “So what can I do to advocate for myself and my baby, right now in this pregnancy?” To be clear, no amount of personal hypervigilance on our part as Black pregnancy-capable people is going to fix these problems, which are systemic; however, we want to provide a few pearls that may be helpful for patient self-advocacy and reassurance:
- Seek culturally and ethnically congruent care. We intuitively want to find a clinician who looks like us, but sadly, in the United States only 5% of physicians and 2% of midwives are Black. Demand exceeds supply for Black patients who are seeking racially congruent care. Nonetheless, it is critical that you find a physician or midwife who centers you and provides support and care that affirms the strengths and assets of you, your family, and your community when cultural and ethnic congruency are not possible for you and your pregnancy.
- Ask how your clinicians are actively working to ensure optimal and equitable experiences for Black birthing individuals. We recommend asking your clinician and/or hospital what, if anything, they are doing to address health care inequities, obstetric racism, or implicit bias in their pregnancy and postpartum care. Many groups (including some authors of this letter) are working on measures to address obstetric racism. An acknowledgement of initiatives to mitigate inequities is a meaningful first step. You can suggest that they look into it while you explore your options, as this work is rapidly emerging in many areas of the country.
- Plan for well-person care. The best time to optimize pregnancy and birth outcomes is before you get pregnant. Set up an appointment with a midwife, ObGyn, or your primary care physician before you get pregnant. Discuss your concerns about pregnancy and use this time to optimize your health. This also provides an opportunity to build a relationship with your physician/ midwife and their group to evaluate whether they curate an environment where you feel seen, heard, and valued when you go for annual exams or problem visits. If you do not get that sense after a couple of visits, find a place where you do.
- Advocate for a second opinion. If something does not sound right to you or you have questions that were not adequately answered, it is your prerogative to seek a second opinion; a clinician should never be offended by this.
- Consider these factors, for those who deliver in a hospital (by choice or necessity):
a. 24/7 access to obstetricians and dedicated anesthesiologists in the hospital
b. trauma-informed medical/mental health/social services
c. lactation consultation
d. supportive trial of labor after cesarean delivery policy
e. massive blood transfusion protocol.
- Seek doula support! It always helps to have another set of eyes and ears to help advocate for you, especially when you are in pain during pregnancy, childbirth, or in the postpartum period, or are having difficulty advocating for yourself. There is also evidence that women supported by doulas have better pregnancy-related outcomes and experiences.23 Many major cities in the United States have started to provide race-concordant doula care for Black birthing people for free.24
- Don’t forget about your mental health. As stated, chronic stress from racism impacts birth outcomes. Having a mental health clinician is a great way to mitigate adverse effects of prolonged tension.25–27
- Ask your clinician, hospital, or insurance company about participating in group prenatal care and/or nurse home visiting models28 because both are associated with improved birth outcomes.29 Many institutions are implementing group care that provides race-concordant care.30,31
- Ask your clinician, hospital, or local health department for recommendations to a lactation consultant or educator who can support your efforts in breast/ chest/body-feeding.
We invite you to consider this truth
You, alone, do not carry the entire population-level risk of Black birthing people on your shoulders. We all carry a piece of it. We, along with many allies, advocates, and activists, are outraged and angered by generations of racism and mistreatment of Black birthing people in our health systems and hospitals. We are channeling our frustration and disgust to demand substantive and sustainable change.
Our purpose here is to provide love and reassurance to our sisters and siblings who are going through their pregnancies with thoughts about our nation’s past and present failures to promote health equity for us and our babies. Our purpose is neither to minimize the public health crisis of Black infant and maternal morbidity and mortality nor is it to absolve clinicians, health systems, or governments from taking responsibility for these shameful outcomes or making meaningful changes to address them. In fact, we love taking care of our community by providing the best clinical care we can to our patients. We call upon all of our clinical colleagues to educate themselves to be ethically and equitably equipped to provide health care for Black pregnant patients. Finally, to birthing Black families, please remember this: If you choose to have a baby, the outcome and experience must align with what is right for you and your baby to survive and thrive. So much of the joys of pregnancy have been stolen, but we will recapture the celebration that should be ours in pregnancy and the journey to parenthood.
Sincerely,
Ebony B. Carter, MD, MPH
Maternal Fetal Medicine
Washington University School of Medicine
St. Louis, Missouri
Karen A. Scott, MD, MPH
Birthing Cultural Rigor, LLC
Nashville, Tennessee
Andrea Jackson, MD, MAS
ObGyn
University of California,
San Francisco
Sara Whetstone, MD, MHS
ObGyn
University of California,
San Francisco
Traci Johnson, MD
ObGyn
University of Missouri
School of Medicine
Kansas City, Missouri
Sarahn Wheeler, MD
Maternal Fetal Medicine
Duke University School of Medicine
Durham, North Carolina
Asmara Gebre, CNM
Midwife
Zuckerberg San Francisco General Hospital
San Francisco, California
Joia Crear-Perry, MD
ObGyn
National Birth Equity Collaborative
New Orleans, Louisiana
Dineo Khabele, MD
Gynecologic Oncology
Washington University School of Medicine
St. Louis, Missouri
Judette Louis, MD, MPH
Maternal Fetal Medicine
University of South Florida College of Medicine
Tampa, Florida
Yvonne Smith, MSN, RN
Director
Barnes-Jewish Hospital
St. Louis, Missouri
Laura Riley, MD
Maternal Fetal Medicine
Weill Cornell Medicine
New York, New York
Antoinette Liddell, MSN, RN
Care Coordinator
Barnes-Jewish Hospital
St. Louis, Missouri
Cynthia Gyamfi-Bannerman, MD
Maternal Fetal Medicine
Columbia University Irving Medical Center
New York, New York
Rasheda Pippens, MSN, RN
Nurse Educator
Barnes-Jewish Hospital
St. Louis, Missouri
Ayaba Worjoloh-Clemens, MD
ObGyn
Atlanta, Georgia
Allison Bryant, MD, MPH
Maternal Fetal Medicine
Massachusetts General Hospital
Boston, Massachusetts
Sheri L. Foote, CNM
Midwife
Zuckerberg San Francisco General Hospital
San Francisco, California
J. Lindsay Sillas, MD
ObGyn
Bella OB/GYN
Houston, Texas
Cynthia Rogers, MD
Psychiatrist
Washington University School of Medicine
St. Louis, Missouri
Audra R. Meadows, MD, MPH
ObGyn
University of California, San Diego
AeuMuro G. Lake, MD
Urogynecologist
Urogynecology and Healing Arts
Seattle, Washington
Nancy Moore, MSN, RN, WHNP-BC
Nurse Practitioner
Barnes-Jewish Hospital
St. Louis, Missouri
Zoë Julian, MD, MPH
ObGyn
University of Alabama at Birmingham
Janice M. Tinsley, MN, RNC-OB
Zuckerberg San Francisco General Hospital
San Francisco, California
Jamila B. Perritt, MD, MPH
ObGyn
Washington, DC
Joy A. Cooper, MD, MSc
ObGyn
Culture Care
Oakland, California
Arthurine K. Zakama, MD
ObGyn
University of California,San Francisco
Alissa Erogbogbo, MD
OB Hospitalist
Los Altos, California
Sanithia L. Williams, MD
ObGyn
Huntsville, Alabama
Audra Williams, MD, MPH
ObGyn
University of Alabama, Birmingham
Hedwige “Didi” Saint Louis, MD, MPH
OB Hospitalist
Morehouse School of Medicine
Atlanta, Georgia
Cherise Cokley, MD
OB Hospitalist
Community Hospital
Munster, Indiana
J’Leise Sosa, MD, MPH
ObGyn
Buffalo, New York
A few years ago, my partner emailed me about a consult.
“Dr. Carter, I had the pleasure of seeing Mrs. Smith today for a preconception consult for chronic hypertension. As a high-risk Black woman, she wants to know what we’re going to do to make sure that she doesn’t die in pregnancy or childbirth. I told her that you’re better equipped to answer this question.”
I was early in my career, and the only thing I could assume that equipped me to answer this question over my partners was my identity as a Black woman living in America.
Mrs. Smith was copied on the message and replied with a long list of follow-up questions and a request for an in-person meeting with me. I was conflicted. As a friend, daughter, and mother, I understood her fear and wanted to be there for her. As a newly appointed assistant professor on the tenure track with 20% clinical time, my clinical responsibilities easily exceeded 50% (in part, because I failed to set boundaries). I spent countless hours of uncompensated time serving on diversity, equity, and inclusion initiatives and mentoring and volunteering for multiple community organizations; I was acutely aware that I would be measured against colleagues who rise through the ranks, unencumbered by these social, moral, and ethical responsibilities, collectively known as the “Black tax.”1
I knew from prior experiences and the tone of Mrs. Smith’s email that it would be a tough, long meeting that would set a precedent of concierge level care that only promised to intensify once she became pregnant. I agonized over my reply. How could I balance providing compassionate care for this patient with my young research program, which I hoped to nurture so that it would one day grow to have population-level impact?
It took me 2 days to finally reply to the message with a kind, but firm, email stating that I would be happy to see her for a follow-up preconception visit. It was my attempt to balance accessibility with boundaries. She did not reply.
Did I fail her?
The fact that I still think of Mrs. Smith may indicate that I did the wrong thing. In fact, writing the first draft of this letter was a therapeutic experience, and I addressed it to Mrs. Smith. As I shared the experience and letter with friends in the field, however, everyone had similar stories. The letter continued to pass between colleagues, who each made it infinitely better. This collective process created the beautiful love letter to Black birthing people that we share here.
We call upon all of our obstetric clinician colleagues to educate themselves to be equally, ethically, and equitably equipped to care for and serve historically marginalized women and birthing people. We hope that this letter will aid in the journey, and we encourage you to share it with patients to open conversations that are too often left closed.

Continue to: Our love letter to Black women and birthing people...
Our love letter to Black women and birthing people
We see you, we hear you, we know you are scared, and we are you. In recent years, the press has amplified gross inequities in maternal care and outcomes that we, as Black birth workers, midwives, and physicians, already knew to be true. We grieve, along with you regarding the recently reported pregnancy-related deaths of Mrs. Kira Johnson,2 Dr. Shalon Irving,3 Dr. Chaniece Wallace,4 and so many other names we do not know because their stories did not receive national attention, but we know that they represented the best of us, and they are gone too soon. As Black birth workers, midwives, physicians, and more, we have a front-row seat to the United States’ serious obstetric racism, manifested in biased clinical interactions, unjust hospital policies, and an inequitable health care system that leads to disparities in maternal morbidity and mortality for Black women.
Unfortunately, this is not anything new, and the legacy dates back to slavery and the disregard for Black people in this country. What has changed is our increased awareness of these health injustices. This collective consciousness of the risk that is carried with our pregnancies casts a shadow of fear over a period that should be full of the joy and promise of new life. We fear that our personhood will be disregarded, our pain will be ignored, and our voices silenced by a medical system that has sought to dominate our bodies and experiment on them without our permission.5 While this history is reprehensible, and our collective risk as Black people is disproportionately high, our purpose in writing this letter is to help Black birthing people recapture the joy and celebration that should be theirs in pregnancy and in the journey to parenthood.
As Black birth workers, we see Black pregnant patients desperately seeking safety, security, and breaking down barriers to find us for their pregnancy care. Often, they are terrified and looking for kinship and community in our offices. In rural areas patients may drive up to 4 hours in distance for an appointment, and during appointments entrust us with their stories of feeling unheard in the medical system. When we anecdotally asked about what they feared about pregnancy, childbirth, and the postpartum period and thought was their risk of dying during pregnancy or childbirth, answers ranged from 1% to 60%. Our actual risk of dying from a pregnancy-related cause, as a Black woman, is 0.0414% (41.4 Black maternal deaths per 100,000 live births).6 To put that in perspective, our risk of dying is higher walking down the street or driving a car.7
What is the source of the fear? Based on past and present injustices inflicted on people with historically marginalized identities, we have every right to be scared; but, make no mistake that fear comes at a cost, and Black birthing people are the ones paying the bill! Stress and chronic worry are associated with poor pregnancy outcomes, and so this completely justifiable fear, at the population level, is not serving us well personally.8 Unfortunately, lost in the messaging about racial inequities in maternal mortality is the reality that the vast majority of Black people and babies will survive, thrive, and have healthy pregnancy outcomes, despite the terrifying population-level statistics and horrific stories of discrimination and neglect that make us feel like our pregnancies and personal peril are synonymous.
While it is true that our absolute individual, personal risk is lower than population-level statistics convey, let us be clear: We are furious about what is happening to Black people! It is immoral that Black patients in the richest country in the world are 3-4 times more likely to die of a pregnancy-related cause than White women,9 and we are more likely to experience pregnancy complications and “near misses” when death is narrowly avoided. Research has done an excellent job defining reproductive health disparities in this country, but prioritizing and funding meaningful strategies, policies, and programs to close this gap have not taken precedence—especially initiatives and research that are headed by Black women.10–12 This is largely because researchers and health care systems continue evaluating strategies that focus on behavior change and narratives that identify individual responsibility as a sole cause of inequity.
Let us be clear, Black people and our behaviors are not the problem.13 The problems are White supremacy, classism, sexism, heteropatriarchy, and obstetric racism.1-21 These must be recognized and addressed across all levels of power. We endorse systems-level changes that are at the root of promoting health equity in our reproductive outcomes. These changes include paid parental leave, Medicaid expansion/extension, reimbursement for doula and lactation services, increased access to perinatal mental health and wellness services, and so much more. (See the Black Mamas Matter Alliance Toolkit: https://blackmamas matter.org/our-work/toolkits/.)
Continue to: Pearls for reassurance...
Pearls for reassurance
While the inequities and their solutions are grounded in the need for systemic change,22 we realize that these population-level solutions feel abstract when our sisters and siblings ask us, “So what can I do to advocate for myself and my baby, right now in this pregnancy?” To be clear, no amount of personal hypervigilance on our part as Black pregnancy-capable people is going to fix these problems, which are systemic; however, we want to provide a few pearls that may be helpful for patient self-advocacy and reassurance:
- Seek culturally and ethnically congruent care. We intuitively want to find a clinician who looks like us, but sadly, in the United States only 5% of physicians and 2% of midwives are Black. Demand exceeds supply for Black patients who are seeking racially congruent care. Nonetheless, it is critical that you find a physician or midwife who centers you and provides support and care that affirms the strengths and assets of you, your family, and your community when cultural and ethnic congruency are not possible for you and your pregnancy.
- Ask how your clinicians are actively working to ensure optimal and equitable experiences for Black birthing individuals. We recommend asking your clinician and/or hospital what, if anything, they are doing to address health care inequities, obstetric racism, or implicit bias in their pregnancy and postpartum care. Many groups (including some authors of this letter) are working on measures to address obstetric racism. An acknowledgement of initiatives to mitigate inequities is a meaningful first step. You can suggest that they look into it while you explore your options, as this work is rapidly emerging in many areas of the country.
- Plan for well-person care. The best time to optimize pregnancy and birth outcomes is before you get pregnant. Set up an appointment with a midwife, ObGyn, or your primary care physician before you get pregnant. Discuss your concerns about pregnancy and use this time to optimize your health. This also provides an opportunity to build a relationship with your physician/ midwife and their group to evaluate whether they curate an environment where you feel seen, heard, and valued when you go for annual exams or problem visits. If you do not get that sense after a couple of visits, find a place where you do.
- Advocate for a second opinion. If something does not sound right to you or you have questions that were not adequately answered, it is your prerogative to seek a second opinion; a clinician should never be offended by this.
- Consider these factors, for those who deliver in a hospital (by choice or necessity):
a. 24/7 access to obstetricians and dedicated anesthesiologists in the hospital
b. trauma-informed medical/mental health/social services
c. lactation consultation
d. supportive trial of labor after cesarean delivery policy
e. massive blood transfusion protocol.
- Seek doula support! It always helps to have another set of eyes and ears to help advocate for you, especially when you are in pain during pregnancy, childbirth, or in the postpartum period, or are having difficulty advocating for yourself. There is also evidence that women supported by doulas have better pregnancy-related outcomes and experiences.23 Many major cities in the United States have started to provide race-concordant doula care for Black birthing people for free.24
- Don’t forget about your mental health. As stated, chronic stress from racism impacts birth outcomes. Having a mental health clinician is a great way to mitigate adverse effects of prolonged tension.25–27
- Ask your clinician, hospital, or insurance company about participating in group prenatal care and/or nurse home visiting models28 because both are associated with improved birth outcomes.29 Many institutions are implementing group care that provides race-concordant care.30,31
- Ask your clinician, hospital, or local health department for recommendations to a lactation consultant or educator who can support your efforts in breast/ chest/body-feeding.
We invite you to consider this truth
You, alone, do not carry the entire population-level risk of Black birthing people on your shoulders. We all carry a piece of it. We, along with many allies, advocates, and activists, are outraged and angered by generations of racism and mistreatment of Black birthing people in our health systems and hospitals. We are channeling our frustration and disgust to demand substantive and sustainable change.
Our purpose here is to provide love and reassurance to our sisters and siblings who are going through their pregnancies with thoughts about our nation’s past and present failures to promote health equity for us and our babies. Our purpose is neither to minimize the public health crisis of Black infant and maternal morbidity and mortality nor is it to absolve clinicians, health systems, or governments from taking responsibility for these shameful outcomes or making meaningful changes to address them. In fact, we love taking care of our community by providing the best clinical care we can to our patients. We call upon all of our clinical colleagues to educate themselves to be ethically and equitably equipped to provide health care for Black pregnant patients. Finally, to birthing Black families, please remember this: If you choose to have a baby, the outcome and experience must align with what is right for you and your baby to survive and thrive. So much of the joys of pregnancy have been stolen, but we will recapture the celebration that should be ours in pregnancy and the journey to parenthood.
Sincerely,
Ebony B. Carter, MD, MPH
Maternal Fetal Medicine
Washington University School of Medicine
St. Louis, Missouri
Karen A. Scott, MD, MPH
Birthing Cultural Rigor, LLC
Nashville, Tennessee
Andrea Jackson, MD, MAS
ObGyn
University of California,
San Francisco
Sara Whetstone, MD, MHS
ObGyn
University of California,
San Francisco
Traci Johnson, MD
ObGyn
University of Missouri
School of Medicine
Kansas City, Missouri
Sarahn Wheeler, MD
Maternal Fetal Medicine
Duke University School of Medicine
Durham, North Carolina
Asmara Gebre, CNM
Midwife
Zuckerberg San Francisco General Hospital
San Francisco, California
Joia Crear-Perry, MD
ObGyn
National Birth Equity Collaborative
New Orleans, Louisiana
Dineo Khabele, MD
Gynecologic Oncology
Washington University School of Medicine
St. Louis, Missouri
Judette Louis, MD, MPH
Maternal Fetal Medicine
University of South Florida College of Medicine
Tampa, Florida
Yvonne Smith, MSN, RN
Director
Barnes-Jewish Hospital
St. Louis, Missouri
Laura Riley, MD
Maternal Fetal Medicine
Weill Cornell Medicine
New York, New York
Antoinette Liddell, MSN, RN
Care Coordinator
Barnes-Jewish Hospital
St. Louis, Missouri
Cynthia Gyamfi-Bannerman, MD
Maternal Fetal Medicine
Columbia University Irving Medical Center
New York, New York
Rasheda Pippens, MSN, RN
Nurse Educator
Barnes-Jewish Hospital
St. Louis, Missouri
Ayaba Worjoloh-Clemens, MD
ObGyn
Atlanta, Georgia
Allison Bryant, MD, MPH
Maternal Fetal Medicine
Massachusetts General Hospital
Boston, Massachusetts
Sheri L. Foote, CNM
Midwife
Zuckerberg San Francisco General Hospital
San Francisco, California
J. Lindsay Sillas, MD
ObGyn
Bella OB/GYN
Houston, Texas
Cynthia Rogers, MD
Psychiatrist
Washington University School of Medicine
St. Louis, Missouri
Audra R. Meadows, MD, MPH
ObGyn
University of California, San Diego
AeuMuro G. Lake, MD
Urogynecologist
Urogynecology and Healing Arts
Seattle, Washington
Nancy Moore, MSN, RN, WHNP-BC
Nurse Practitioner
Barnes-Jewish Hospital
St. Louis, Missouri
Zoë Julian, MD, MPH
ObGyn
University of Alabama at Birmingham
Janice M. Tinsley, MN, RNC-OB
Zuckerberg San Francisco General Hospital
San Francisco, California
Jamila B. Perritt, MD, MPH
ObGyn
Washington, DC
Joy A. Cooper, MD, MSc
ObGyn
Culture Care
Oakland, California
Arthurine K. Zakama, MD
ObGyn
University of California,San Francisco
Alissa Erogbogbo, MD
OB Hospitalist
Los Altos, California
Sanithia L. Williams, MD
ObGyn
Huntsville, Alabama
Audra Williams, MD, MPH
ObGyn
University of Alabama, Birmingham
Hedwige “Didi” Saint Louis, MD, MPH
OB Hospitalist
Morehouse School of Medicine
Atlanta, Georgia
Cherise Cokley, MD
OB Hospitalist
Community Hospital
Munster, Indiana
J’Leise Sosa, MD, MPH
ObGyn
Buffalo, New York
- Rodríguez JE, Campbell KM, Pololi LH. Addressing disparities in academic medicine: what of the minority tax? BMC Med Educ. 2015;15:6. https ://doi.org/10.1186/s12909-015-0290-9.
- Helm A. Yet another beautiful Black woman dies in childbirth. Kira Johnson spoke 5 languages, raced cars, was daughter in law of Judge Glenda Hatchett. She still died in childbirth. October 19, 2018. https://www.theroot.com/kira-johnson-spoke- 5-languages-raced-cars-was-daughter-18298 62323. Accessed February 27, 2027.
- Shock after Black pediatrics doctor dies after giving birth to first child. November 6, 2020. https ://www.bet.com/article/rvyskv/black-pediatrics -doctor-dies-after-giving-birth#! Accessed February 24, 2023.
- Dr. Shalon’s maternal action project. https ://www.drshalonsmap.org/. Accessed February 24, 2023.
- Verdantam S, Penman M. Remembering Anarcha, Lucy, and Betsey: The mothers of modern gynecology. https://www.npr .org/2016/02/16/466942135/remembering -anarcha-lucy-and-betsey-the-mothers-of -modern-gynecology. February 16, 2016. Accessed February 24, 2023.
- Centers for Disease Control and Prevention website. Pregnancy Mortality Surveillance System. Last reviewed June 22, 2022. Accessed March 8, 2023.
- Odds of dying. NSC injury facts. https ://injuryfacts.nsc.org/all-injuries/preventable -death-overview/odds-of-dying/data-details /#:~:text=Statements%20about%20the%20 odds%20or%20chances%20of%20dying,in% 20%28value%20given%20in%20the%20lifetime %20odds%20column%29. Accessed February 24, 2023.
- Gembruch U, Baschat AA. True knot of the umbilical cord: transient constrictive effect to umbilical venous blood flow demonstrated by Doppler sonography. Ultrasound Obstet Gynecol. 1996;8:53-56. doi: 10.1046/j.14690705.1996.08010053.x.
- MacDorman MF, Thoma M, Declcerq E, et al. Racial and ethnic disparities in maternal mortality in the United States using enhanced vital records, 2016-2017. Am J Public Health. 2012;111:16731681.
- Taffe MA, Gilpin NW. Racial inequity in grant funding from the US National Institutes of Health. Elife. 2021;10. doi: 10.7554/eLife.65697.
- Black Women Scholars and Research Working Group for the Black Mamas Matter Alliance. Black maternal health research re-envisioned: best practices for the conduct of research with, for, and by Black mamas. Harvard Law Policy Rev. 2020;14:393.
- Sullivan P. In philanthropy, race is still a factor in who gets what, study shows. NY Times. https ://www.nytimes.com/2020/05/01/your-money /philanthropy-race.html. May 5, 2020.
- Scott KA, Britton L, McLemore MR. The ethics of perinatal care for Black women: dismantling the structural racism in “Mother Blame” narratives. J Perinat Neonatal Nurs. 2019;33:108-115. doi: 10.1097/jpn.0000000000000394.
- Dominguez TP, Dunkel-Schetter C, Glynn LM, Hobel C, Sandman CA. Racial Differences in Birth Outcomes: The Role of General, Pregnancy, and Racism Stress. Health Psychology. 2008;27(2):194203. doi: 10.1037/0278-6133.27.2.194.
- Hardeman RR, Murphy KA, Karbeah J, et al. Naming institutionalized racism in the public health literature: a systematic literature review. Public Health Rep. 2018;133:240-249. doi: 10.1177/0033354918760574.
- Hardeman RR, Karbeah J. Examining racism in health services research: a disciplinary self- critique. Health Serv Res. 2020;55 Suppl 2:777-780. doi: 10.1111/1475-6773.13558.
- Hardeman RR, Karbeah J, Kozhimannil KB. Applying a critical race lens to relationship-centered care in pregnancy and childbirth: an antidote to structural racism. Birth. 2020;47:3-7. doi: 10.1111/birt.12462.
- Scott KA, Davis D-A. Obstetric racism: naming and identifying a way out of Black women’s adverse medical experiences. Am Anthropologist. 2021;123:681-684. doi: https://doi.org/10.1111 /aman.13559.
- Mullings L. Resistance and resilience the sojourner syndrome and the social context of reproduction in central Harlem. Schulz AJ, Mullings L, eds. Gender, Race, Class, & Health: Intersectional Approaches. Jossey-Bass/Wiley: Hoboken, NJ; 2006:345-370.
- Chambers BD, Arabia SE, Arega HA, et al. Exposures to structural racism and racial discrimination among pregnant and early post-partum Black women living in Oakland, California. Stress Health. 2020;36:213-219. doi: 10.1002/smi.2922.
- Chambers BD, Arega HA, Arabia SE, et al. Black women’s perspectives on structural racism across the reproductive lifespan: a conceptual framework for measurement development. Maternal Child Health J. 2021;25:402-413. doi: 10.1007 /s10995-020-03074-3.
- Julian Z, Robles D, Whetstone S, et al. Community-informed models of perinatal and reproductive health services provision: A justice-centered paradigm toward equity among Black birthing communities. Seminar Perinatol. 2020;44:151267. doi: 10.1016/j.semperi.2020.151267.
- Bohren MA, Hofmeyr GJ, Sakala C, et al. Continuous support for women during childbirth. Cochrane Database System Rev. 2017;7:Cd003766. doi: 10.1002/14651858.CD003766.pub6.
- National Black doulas association. https://www .blackdoulas.org/. Accessed February 24, 2023.
- Therapy for Black girls. https://therapyforblack girls.com/. Accessed February 24, 2023.
- National Queer and Trans Therapists of Color Network. https://www.nqttcn.com/. Accessed February 24, 2023.
- Shades of Blue Project. http://cbww.org. Accessed February 24, 2023.
- Centering Healthcare Institute. https://www .centeringhealthcare.org/. Accessed February 24, 2023.
- Carter EB, Temming LA, Akin J, et al. Group prenatal care compared with traditional prenatal care: a systematic review and meta-analysis. Obstet Gynecol. 2016;128:551-561. doi: 10.1097 /aog.0000000000001560.
- National Center of Excellence in Women’s Health. https://womenshealth.ucsf.edu/coe/embrace -perinatal-care-black-families. Accessed February 24, 2023.
- Alameda Health System. http://www.alamedahealthsystem.org/family-birthing-center/black -centering/. Accessed February 24, 2023.
- Rodríguez JE, Campbell KM, Pololi LH. Addressing disparities in academic medicine: what of the minority tax? BMC Med Educ. 2015;15:6. https ://doi.org/10.1186/s12909-015-0290-9.
- Helm A. Yet another beautiful Black woman dies in childbirth. Kira Johnson spoke 5 languages, raced cars, was daughter in law of Judge Glenda Hatchett. She still died in childbirth. October 19, 2018. https://www.theroot.com/kira-johnson-spoke- 5-languages-raced-cars-was-daughter-18298 62323. Accessed February 27, 2027.
- Shock after Black pediatrics doctor dies after giving birth to first child. November 6, 2020. https ://www.bet.com/article/rvyskv/black-pediatrics -doctor-dies-after-giving-birth#! Accessed February 24, 2023.
- Dr. Shalon’s maternal action project. https ://www.drshalonsmap.org/. Accessed February 24, 2023.
- Verdantam S, Penman M. Remembering Anarcha, Lucy, and Betsey: The mothers of modern gynecology. https://www.npr .org/2016/02/16/466942135/remembering -anarcha-lucy-and-betsey-the-mothers-of -modern-gynecology. February 16, 2016. Accessed February 24, 2023.
- Centers for Disease Control and Prevention website. Pregnancy Mortality Surveillance System. Last reviewed June 22, 2022. Accessed March 8, 2023.
- Odds of dying. NSC injury facts. https ://injuryfacts.nsc.org/all-injuries/preventable -death-overview/odds-of-dying/data-details /#:~:text=Statements%20about%20the%20 odds%20or%20chances%20of%20dying,in% 20%28value%20given%20in%20the%20lifetime %20odds%20column%29. Accessed February 24, 2023.
- Gembruch U, Baschat AA. True knot of the umbilical cord: transient constrictive effect to umbilical venous blood flow demonstrated by Doppler sonography. Ultrasound Obstet Gynecol. 1996;8:53-56. doi: 10.1046/j.14690705.1996.08010053.x.
- MacDorman MF, Thoma M, Declcerq E, et al. Racial and ethnic disparities in maternal mortality in the United States using enhanced vital records, 2016-2017. Am J Public Health. 2012;111:16731681.
- Taffe MA, Gilpin NW. Racial inequity in grant funding from the US National Institutes of Health. Elife. 2021;10. doi: 10.7554/eLife.65697.
- Black Women Scholars and Research Working Group for the Black Mamas Matter Alliance. Black maternal health research re-envisioned: best practices for the conduct of research with, for, and by Black mamas. Harvard Law Policy Rev. 2020;14:393.
- Sullivan P. In philanthropy, race is still a factor in who gets what, study shows. NY Times. https ://www.nytimes.com/2020/05/01/your-money /philanthropy-race.html. May 5, 2020.
- Scott KA, Britton L, McLemore MR. The ethics of perinatal care for Black women: dismantling the structural racism in “Mother Blame” narratives. J Perinat Neonatal Nurs. 2019;33:108-115. doi: 10.1097/jpn.0000000000000394.
- Dominguez TP, Dunkel-Schetter C, Glynn LM, Hobel C, Sandman CA. Racial Differences in Birth Outcomes: The Role of General, Pregnancy, and Racism Stress. Health Psychology. 2008;27(2):194203. doi: 10.1037/0278-6133.27.2.194.
- Hardeman RR, Murphy KA, Karbeah J, et al. Naming institutionalized racism in the public health literature: a systematic literature review. Public Health Rep. 2018;133:240-249. doi: 10.1177/0033354918760574.
- Hardeman RR, Karbeah J. Examining racism in health services research: a disciplinary self- critique. Health Serv Res. 2020;55 Suppl 2:777-780. doi: 10.1111/1475-6773.13558.
- Hardeman RR, Karbeah J, Kozhimannil KB. Applying a critical race lens to relationship-centered care in pregnancy and childbirth: an antidote to structural racism. Birth. 2020;47:3-7. doi: 10.1111/birt.12462.
- Scott KA, Davis D-A. Obstetric racism: naming and identifying a way out of Black women’s adverse medical experiences. Am Anthropologist. 2021;123:681-684. doi: https://doi.org/10.1111 /aman.13559.
- Mullings L. Resistance and resilience the sojourner syndrome and the social context of reproduction in central Harlem. Schulz AJ, Mullings L, eds. Gender, Race, Class, & Health: Intersectional Approaches. Jossey-Bass/Wiley: Hoboken, NJ; 2006:345-370.
- Chambers BD, Arabia SE, Arega HA, et al. Exposures to structural racism and racial discrimination among pregnant and early post-partum Black women living in Oakland, California. Stress Health. 2020;36:213-219. doi: 10.1002/smi.2922.
- Chambers BD, Arega HA, Arabia SE, et al. Black women’s perspectives on structural racism across the reproductive lifespan: a conceptual framework for measurement development. Maternal Child Health J. 2021;25:402-413. doi: 10.1007 /s10995-020-03074-3.
- Julian Z, Robles D, Whetstone S, et al. Community-informed models of perinatal and reproductive health services provision: A justice-centered paradigm toward equity among Black birthing communities. Seminar Perinatol. 2020;44:151267. doi: 10.1016/j.semperi.2020.151267.
- Bohren MA, Hofmeyr GJ, Sakala C, et al. Continuous support for women during childbirth. Cochrane Database System Rev. 2017;7:Cd003766. doi: 10.1002/14651858.CD003766.pub6.
- National Black doulas association. https://www .blackdoulas.org/. Accessed February 24, 2023.
- Therapy for Black girls. https://therapyforblack girls.com/. Accessed February 24, 2023.
- National Queer and Trans Therapists of Color Network. https://www.nqttcn.com/. Accessed February 24, 2023.
- Shades of Blue Project. http://cbww.org. Accessed February 24, 2023.
- Centering Healthcare Institute. https://www .centeringhealthcare.org/. Accessed February 24, 2023.
- Carter EB, Temming LA, Akin J, et al. Group prenatal care compared with traditional prenatal care: a systematic review and meta-analysis. Obstet Gynecol. 2016;128:551-561. doi: 10.1097 /aog.0000000000001560.
- National Center of Excellence in Women’s Health. https://womenshealth.ucsf.edu/coe/embrace -perinatal-care-black-families. Accessed February 24, 2023.
- Alameda Health System. http://www.alamedahealthsystem.org/family-birthing-center/black -centering/. Accessed February 24, 2023.
Novel single-use patch shows promise for primary axillary hyperhidrosis
NEW ORLEANS – , results from a pivotal randomized trial showed.
“This is a new kind of device that is going to be a nice tool to have for treating patients who have hyperhidrosis of the axilla,” the study’s lead investigator, David M. Pariser, MD, who practices dermatology in Norfolk, Va., said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology.
In a study known as SAHARA, investigators at 11 sites evaluated the efficacy of the targeted alkali thermolysis (TAT) patch, a single-use disposable device. The patch consists of a thin sodium layer on an adhesive overlay. It’s applied to the dry axilla, and as the patient sweats during treatment, the sweat reacts with the sodium. According to Dr. Pariser, this interaction generates precisely targeted thermal energy that targets sweat glands, leading to a reduction in excessive sweat production for up to three months.
The researchers enrolled 110 individuals with Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4 and randomized them to either an active TAT or a sham patch for up to 3 minutes. Their mean age was about 33 years, and slightly more than half were women. “If significant discomfort or pain was noted, [the patch] treatment was halted; otherwise, it was left on for 3 minutes,” said Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk. “The treated area was thoroughly cleaned after treatment, and the TAT patch was deactivated. This process was repeated on the other axilla.”
The HDSS, Gravimetric Sweat Production (GSP), and quality of life assessments for bother and impact were measured through 12 weeks. The quality of life assessments were an exploratory endpoint and scored from 0 to 4, with 4 being extremely bothered or impacted and 0 not being bothered or impacted at all. The primary efficacy endpoint was the proportion of treated patients achieving a 1 or 2 on the HDSS at week 4, compared with sham treatment.
Secondary endpoints included the proportion of patients with an improvement of at least 2 grades from baseline to 4 weeks in HDSS by treatment group; mean improvement in the quality of life scale bother by treatment group; mean improvement in the quality of life scale impact by treatment group; and the proportion of subjects with at least 50% improvement in GSP from baseline to 4 weeks in the active patch group only.
Adverse events (AEs) were divided into 3 categories: AEs at the treatment site (or skin reactions within the treated part of the axilla); procedure-related AEs (those that are the result of treatment, but not in the treated part of the axilla), and non-axillary AEs.
Dr. Pariser reported that at 4 weeks, 63.6% of patients in the active patch group versus 44.2% of those in the sham group improved to an HDSS score of 1 or 2 (P = .0332) and that 43.2% of those in the active patch group versus 16.3% of those in the sham group (P = .0107) achieved a 2-point or greater HDSS improvement. In addition, 9.1% of those in the active patch group achieved a 3-point improvement on the HDSS, compared with none in the sham group. “That’s an amazing improvement; you’re basically going from moderate or severe to none,” he commented.
In other findings, 60.5% of patients in the active patch group showed at least a 50% reduction in GSP, compared with 32.6% of those in the sham group (P = .0102), with mean reductions of 57.3 mg/5min and 18.2 mg/5min, respectively (P = .0036). As for quality-of-life outcome scores, bother associated with hyperhidrosis was reduced by 1.52 points in active versus 0.61 in sham subjects (P = .0005), while impact was reduced by 1.44 in active versus 0.57 in sham subjects (P = .0004).
Adverse events
A total of 13 patients in the active patch group experienced AEs at the treatment site, including six with erythema; four with erosion; two with burning, itching or stinging; and one with underarm odor. “The two procedure-related AEs in the TAT-treated group were compensatory sweating and irritant contact dermatitis due to the adhesive,” said Dr. Pariser said.
Most adverse events resolved in fewer than 2 weeks, and all were mild to moderate. No serious adverse events occurred. Only five adverse events occurred in the sham group.
The TAT patch is currently undergoing review by the Food and Drug Administration, and according to Dr. Pariser, no other body sites have been treated with the device.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, characterized hyperhidrosis as “an exceedingly common medical condition that is commonly overlooked even though it has a tremendous burden on quality of life. I should know, as both someone who manages a large cohort of these patients but also as someone who suffers from it.”
Treatment options “have historically been limited, many of which are off-label and some which are difficult to access due to cost and/or duration/frequency of treatment,” added Dr. Friedman, who was not involved with the study. “The TAT patch offers a new, targeted, in-office, practical procedure-based approach to treat primary axillary hyperhidrosis. Innovation is certainly welcomed and needed, and I am curious to see how this technology is employed in practice once approved.”
The device is being developed by Candesant Biomedical. Dr. Pariser disclosed that he is a consultant or investigator for Bickel Biotechnology, Biofrontera AG, Bristol Myers Squibb, Celgene Corporation, Novartis Pharmaceuticals, Pfizer, Regeneron, and Sanofi.
Dr. Friedman reported having no relevant disclosures.
NEW ORLEANS – , results from a pivotal randomized trial showed.
“This is a new kind of device that is going to be a nice tool to have for treating patients who have hyperhidrosis of the axilla,” the study’s lead investigator, David M. Pariser, MD, who practices dermatology in Norfolk, Va., said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology.
In a study known as SAHARA, investigators at 11 sites evaluated the efficacy of the targeted alkali thermolysis (TAT) patch, a single-use disposable device. The patch consists of a thin sodium layer on an adhesive overlay. It’s applied to the dry axilla, and as the patient sweats during treatment, the sweat reacts with the sodium. According to Dr. Pariser, this interaction generates precisely targeted thermal energy that targets sweat glands, leading to a reduction in excessive sweat production for up to three months.
The researchers enrolled 110 individuals with Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4 and randomized them to either an active TAT or a sham patch for up to 3 minutes. Their mean age was about 33 years, and slightly more than half were women. “If significant discomfort or pain was noted, [the patch] treatment was halted; otherwise, it was left on for 3 minutes,” said Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk. “The treated area was thoroughly cleaned after treatment, and the TAT patch was deactivated. This process was repeated on the other axilla.”
The HDSS, Gravimetric Sweat Production (GSP), and quality of life assessments for bother and impact were measured through 12 weeks. The quality of life assessments were an exploratory endpoint and scored from 0 to 4, with 4 being extremely bothered or impacted and 0 not being bothered or impacted at all. The primary efficacy endpoint was the proportion of treated patients achieving a 1 or 2 on the HDSS at week 4, compared with sham treatment.
Secondary endpoints included the proportion of patients with an improvement of at least 2 grades from baseline to 4 weeks in HDSS by treatment group; mean improvement in the quality of life scale bother by treatment group; mean improvement in the quality of life scale impact by treatment group; and the proportion of subjects with at least 50% improvement in GSP from baseline to 4 weeks in the active patch group only.
Adverse events (AEs) were divided into 3 categories: AEs at the treatment site (or skin reactions within the treated part of the axilla); procedure-related AEs (those that are the result of treatment, but not in the treated part of the axilla), and non-axillary AEs.
Dr. Pariser reported that at 4 weeks, 63.6% of patients in the active patch group versus 44.2% of those in the sham group improved to an HDSS score of 1 or 2 (P = .0332) and that 43.2% of those in the active patch group versus 16.3% of those in the sham group (P = .0107) achieved a 2-point or greater HDSS improvement. In addition, 9.1% of those in the active patch group achieved a 3-point improvement on the HDSS, compared with none in the sham group. “That’s an amazing improvement; you’re basically going from moderate or severe to none,” he commented.
In other findings, 60.5% of patients in the active patch group showed at least a 50% reduction in GSP, compared with 32.6% of those in the sham group (P = .0102), with mean reductions of 57.3 mg/5min and 18.2 mg/5min, respectively (P = .0036). As for quality-of-life outcome scores, bother associated with hyperhidrosis was reduced by 1.52 points in active versus 0.61 in sham subjects (P = .0005), while impact was reduced by 1.44 in active versus 0.57 in sham subjects (P = .0004).
Adverse events
A total of 13 patients in the active patch group experienced AEs at the treatment site, including six with erythema; four with erosion; two with burning, itching or stinging; and one with underarm odor. “The two procedure-related AEs in the TAT-treated group were compensatory sweating and irritant contact dermatitis due to the adhesive,” said Dr. Pariser said.
Most adverse events resolved in fewer than 2 weeks, and all were mild to moderate. No serious adverse events occurred. Only five adverse events occurred in the sham group.
The TAT patch is currently undergoing review by the Food and Drug Administration, and according to Dr. Pariser, no other body sites have been treated with the device.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, characterized hyperhidrosis as “an exceedingly common medical condition that is commonly overlooked even though it has a tremendous burden on quality of life. I should know, as both someone who manages a large cohort of these patients but also as someone who suffers from it.”
Treatment options “have historically been limited, many of which are off-label and some which are difficult to access due to cost and/or duration/frequency of treatment,” added Dr. Friedman, who was not involved with the study. “The TAT patch offers a new, targeted, in-office, practical procedure-based approach to treat primary axillary hyperhidrosis. Innovation is certainly welcomed and needed, and I am curious to see how this technology is employed in practice once approved.”
The device is being developed by Candesant Biomedical. Dr. Pariser disclosed that he is a consultant or investigator for Bickel Biotechnology, Biofrontera AG, Bristol Myers Squibb, Celgene Corporation, Novartis Pharmaceuticals, Pfizer, Regeneron, and Sanofi.
Dr. Friedman reported having no relevant disclosures.
NEW ORLEANS – , results from a pivotal randomized trial showed.
“This is a new kind of device that is going to be a nice tool to have for treating patients who have hyperhidrosis of the axilla,” the study’s lead investigator, David M. Pariser, MD, who practices dermatology in Norfolk, Va., said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology.
In a study known as SAHARA, investigators at 11 sites evaluated the efficacy of the targeted alkali thermolysis (TAT) patch, a single-use disposable device. The patch consists of a thin sodium layer on an adhesive overlay. It’s applied to the dry axilla, and as the patient sweats during treatment, the sweat reacts with the sodium. According to Dr. Pariser, this interaction generates precisely targeted thermal energy that targets sweat glands, leading to a reduction in excessive sweat production for up to three months.
The researchers enrolled 110 individuals with Hyperhidrosis Disease Severity Scale (HDSS) scores of 3 or 4 and randomized them to either an active TAT or a sham patch for up to 3 minutes. Their mean age was about 33 years, and slightly more than half were women. “If significant discomfort or pain was noted, [the patch] treatment was halted; otherwise, it was left on for 3 minutes,” said Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk. “The treated area was thoroughly cleaned after treatment, and the TAT patch was deactivated. This process was repeated on the other axilla.”
The HDSS, Gravimetric Sweat Production (GSP), and quality of life assessments for bother and impact were measured through 12 weeks. The quality of life assessments were an exploratory endpoint and scored from 0 to 4, with 4 being extremely bothered or impacted and 0 not being bothered or impacted at all. The primary efficacy endpoint was the proportion of treated patients achieving a 1 or 2 on the HDSS at week 4, compared with sham treatment.
Secondary endpoints included the proportion of patients with an improvement of at least 2 grades from baseline to 4 weeks in HDSS by treatment group; mean improvement in the quality of life scale bother by treatment group; mean improvement in the quality of life scale impact by treatment group; and the proportion of subjects with at least 50% improvement in GSP from baseline to 4 weeks in the active patch group only.
Adverse events (AEs) were divided into 3 categories: AEs at the treatment site (or skin reactions within the treated part of the axilla); procedure-related AEs (those that are the result of treatment, but not in the treated part of the axilla), and non-axillary AEs.
Dr. Pariser reported that at 4 weeks, 63.6% of patients in the active patch group versus 44.2% of those in the sham group improved to an HDSS score of 1 or 2 (P = .0332) and that 43.2% of those in the active patch group versus 16.3% of those in the sham group (P = .0107) achieved a 2-point or greater HDSS improvement. In addition, 9.1% of those in the active patch group achieved a 3-point improvement on the HDSS, compared with none in the sham group. “That’s an amazing improvement; you’re basically going from moderate or severe to none,” he commented.
In other findings, 60.5% of patients in the active patch group showed at least a 50% reduction in GSP, compared with 32.6% of those in the sham group (P = .0102), with mean reductions of 57.3 mg/5min and 18.2 mg/5min, respectively (P = .0036). As for quality-of-life outcome scores, bother associated with hyperhidrosis was reduced by 1.52 points in active versus 0.61 in sham subjects (P = .0005), while impact was reduced by 1.44 in active versus 0.57 in sham subjects (P = .0004).
Adverse events
A total of 13 patients in the active patch group experienced AEs at the treatment site, including six with erythema; four with erosion; two with burning, itching or stinging; and one with underarm odor. “The two procedure-related AEs in the TAT-treated group were compensatory sweating and irritant contact dermatitis due to the adhesive,” said Dr. Pariser said.
Most adverse events resolved in fewer than 2 weeks, and all were mild to moderate. No serious adverse events occurred. Only five adverse events occurred in the sham group.
The TAT patch is currently undergoing review by the Food and Drug Administration, and according to Dr. Pariser, no other body sites have been treated with the device.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, characterized hyperhidrosis as “an exceedingly common medical condition that is commonly overlooked even though it has a tremendous burden on quality of life. I should know, as both someone who manages a large cohort of these patients but also as someone who suffers from it.”
Treatment options “have historically been limited, many of which are off-label and some which are difficult to access due to cost and/or duration/frequency of treatment,” added Dr. Friedman, who was not involved with the study. “The TAT patch offers a new, targeted, in-office, practical procedure-based approach to treat primary axillary hyperhidrosis. Innovation is certainly welcomed and needed, and I am curious to see how this technology is employed in practice once approved.”
The device is being developed by Candesant Biomedical. Dr. Pariser disclosed that he is a consultant or investigator for Bickel Biotechnology, Biofrontera AG, Bristol Myers Squibb, Celgene Corporation, Novartis Pharmaceuticals, Pfizer, Regeneron, and Sanofi.
Dr. Friedman reported having no relevant disclosures.
AT AAD 2023