User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Children who are overweight at risk for chronic kidney disease
TOPLINE
, with the association, though weaker, still significant among those who do not develop type 2 diabetes or hypertension, in a large cohort study.
METHODOLOGY
- The study included data on 593,660 adolescents aged 16-20, born after January 1, 1975, who had medical assessments as part of mandatory military service in Israel.
- The mean age at study entry was 17.2 and 54.5% were male.
- Early CKD was defined as stage 1 to 2 CKD with moderately or severely increased albuminuria, with an estimated glomerular filtration rate of 60 mL/min/1.73 m2 or higher.
- The study excluded those with kidney pathology, albuminuria, hypertension, dysglycemia, or missing blood pressure or BMI data.
- Participants were followed up until early CKD onset, death, the last day insured, or August 23, 2020.
TAKEAWAY
- With a mean follow-up of 13.4 years, 1963 adolescents (0.3%) overall developed early chronic kidney disease. Among males, an increased risk of developing CKD was observed with a high-normal BMI in adolescence (hazard ratio [HR], 1.8); with overweight BMI (HR, 4.0); with mild obesity (HR, 6.7); and severe obesity (HR, 9.4).
- Among females, the increased risk was also observed with high-normal BMI (HR 1.4); overweight (HR, 2.3); mild obesity (HR, 2.7); and severe obesity (HR, 4.3).
- In excluding those who developed diabetes or hypertension, the overall rate of early CKD in the cohort was 0.2%.
- For males without diabetes or hypertension, the adjusted HR for early CKD with high-normal weight was 1.2; for overweight, HR 1.6; for mild obesity, HR 2.2; and for severe obesity, HR 2.7.
- For females without diabetes or hypertension, the corresponding increased risk for early CKD was HR 1.2 for high-normal BMI; HR 1.8 for overweight; 1.5 for mild obesity and 2.3 for severe obesity.
IN PRACTICE
“These findings suggest that adolescent obesity is a major risk factor for early CKD in young adulthood; this underscores the importance of mitigating adolescent obesity rates and managing risk factors for kidney disease in adolescents with high BMI,” the authors report.
“The association was evident even in persons with high-normal BMI in adolescence, was more pronounced in men, and appeared before the age of 30 years,” they say.
“Given the increasing obesity rates among adolescents, our findings are a harbinger of the potentially preventable increasing burden of CKD and subsequent cardiovascular disease.”
SOURCE
The study was conducted by first author Avishai M. Tsur, MD, of the Israel Defense Forces, Medical Corps, Tel Hashomer, Ramat Gan, Israel and Department of Military Medicine, Hebrew University of Jerusalem Faculty of Medicine, Jerusalem, Israel, and colleagues. The study was published online in JAMA Pediatrics.
LIMITATIONS
The study lacked longitudinal data on clinical and lifestyle factors, including stress, diet and physical activity. While adolescents were screened using urine dipstick, a lack of serum creatinine measurements could have missed some adolescents with reduced eGFR at the study entry. The generalizability of the results is limited by the lack of people from West Africa and East Asia in the study population.
DISCLOSURES
Coauthor Josef Coresh, MD, reported receiving grants from the National Institutes of Health outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE
, with the association, though weaker, still significant among those who do not develop type 2 diabetes or hypertension, in a large cohort study.
METHODOLOGY
- The study included data on 593,660 adolescents aged 16-20, born after January 1, 1975, who had medical assessments as part of mandatory military service in Israel.
- The mean age at study entry was 17.2 and 54.5% were male.
- Early CKD was defined as stage 1 to 2 CKD with moderately or severely increased albuminuria, with an estimated glomerular filtration rate of 60 mL/min/1.73 m2 or higher.
- The study excluded those with kidney pathology, albuminuria, hypertension, dysglycemia, or missing blood pressure or BMI data.
- Participants were followed up until early CKD onset, death, the last day insured, or August 23, 2020.
TAKEAWAY
- With a mean follow-up of 13.4 years, 1963 adolescents (0.3%) overall developed early chronic kidney disease. Among males, an increased risk of developing CKD was observed with a high-normal BMI in adolescence (hazard ratio [HR], 1.8); with overweight BMI (HR, 4.0); with mild obesity (HR, 6.7); and severe obesity (HR, 9.4).
- Among females, the increased risk was also observed with high-normal BMI (HR 1.4); overweight (HR, 2.3); mild obesity (HR, 2.7); and severe obesity (HR, 4.3).
- In excluding those who developed diabetes or hypertension, the overall rate of early CKD in the cohort was 0.2%.
- For males without diabetes or hypertension, the adjusted HR for early CKD with high-normal weight was 1.2; for overweight, HR 1.6; for mild obesity, HR 2.2; and for severe obesity, HR 2.7.
- For females without diabetes or hypertension, the corresponding increased risk for early CKD was HR 1.2 for high-normal BMI; HR 1.8 for overweight; 1.5 for mild obesity and 2.3 for severe obesity.
IN PRACTICE
“These findings suggest that adolescent obesity is a major risk factor for early CKD in young adulthood; this underscores the importance of mitigating adolescent obesity rates and managing risk factors for kidney disease in adolescents with high BMI,” the authors report.
“The association was evident even in persons with high-normal BMI in adolescence, was more pronounced in men, and appeared before the age of 30 years,” they say.
“Given the increasing obesity rates among adolescents, our findings are a harbinger of the potentially preventable increasing burden of CKD and subsequent cardiovascular disease.”
SOURCE
The study was conducted by first author Avishai M. Tsur, MD, of the Israel Defense Forces, Medical Corps, Tel Hashomer, Ramat Gan, Israel and Department of Military Medicine, Hebrew University of Jerusalem Faculty of Medicine, Jerusalem, Israel, and colleagues. The study was published online in JAMA Pediatrics.
LIMITATIONS
The study lacked longitudinal data on clinical and lifestyle factors, including stress, diet and physical activity. While adolescents were screened using urine dipstick, a lack of serum creatinine measurements could have missed some adolescents with reduced eGFR at the study entry. The generalizability of the results is limited by the lack of people from West Africa and East Asia in the study population.
DISCLOSURES
Coauthor Josef Coresh, MD, reported receiving grants from the National Institutes of Health outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE
, with the association, though weaker, still significant among those who do not develop type 2 diabetes or hypertension, in a large cohort study.
METHODOLOGY
- The study included data on 593,660 adolescents aged 16-20, born after January 1, 1975, who had medical assessments as part of mandatory military service in Israel.
- The mean age at study entry was 17.2 and 54.5% were male.
- Early CKD was defined as stage 1 to 2 CKD with moderately or severely increased albuminuria, with an estimated glomerular filtration rate of 60 mL/min/1.73 m2 or higher.
- The study excluded those with kidney pathology, albuminuria, hypertension, dysglycemia, or missing blood pressure or BMI data.
- Participants were followed up until early CKD onset, death, the last day insured, or August 23, 2020.
TAKEAWAY
- With a mean follow-up of 13.4 years, 1963 adolescents (0.3%) overall developed early chronic kidney disease. Among males, an increased risk of developing CKD was observed with a high-normal BMI in adolescence (hazard ratio [HR], 1.8); with overweight BMI (HR, 4.0); with mild obesity (HR, 6.7); and severe obesity (HR, 9.4).
- Among females, the increased risk was also observed with high-normal BMI (HR 1.4); overweight (HR, 2.3); mild obesity (HR, 2.7); and severe obesity (HR, 4.3).
- In excluding those who developed diabetes or hypertension, the overall rate of early CKD in the cohort was 0.2%.
- For males without diabetes or hypertension, the adjusted HR for early CKD with high-normal weight was 1.2; for overweight, HR 1.6; for mild obesity, HR 2.2; and for severe obesity, HR 2.7.
- For females without diabetes or hypertension, the corresponding increased risk for early CKD was HR 1.2 for high-normal BMI; HR 1.8 for overweight; 1.5 for mild obesity and 2.3 for severe obesity.
IN PRACTICE
“These findings suggest that adolescent obesity is a major risk factor for early CKD in young adulthood; this underscores the importance of mitigating adolescent obesity rates and managing risk factors for kidney disease in adolescents with high BMI,” the authors report.
“The association was evident even in persons with high-normal BMI in adolescence, was more pronounced in men, and appeared before the age of 30 years,” they say.
“Given the increasing obesity rates among adolescents, our findings are a harbinger of the potentially preventable increasing burden of CKD and subsequent cardiovascular disease.”
SOURCE
The study was conducted by first author Avishai M. Tsur, MD, of the Israel Defense Forces, Medical Corps, Tel Hashomer, Ramat Gan, Israel and Department of Military Medicine, Hebrew University of Jerusalem Faculty of Medicine, Jerusalem, Israel, and colleagues. The study was published online in JAMA Pediatrics.
LIMITATIONS
The study lacked longitudinal data on clinical and lifestyle factors, including stress, diet and physical activity. While adolescents were screened using urine dipstick, a lack of serum creatinine measurements could have missed some adolescents with reduced eGFR at the study entry. The generalizability of the results is limited by the lack of people from West Africa and East Asia in the study population.
DISCLOSURES
Coauthor Josef Coresh, MD, reported receiving grants from the National Institutes of Health outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
Technology for primary care — terrific, terrifying, or both?
We have all been using technology in our primary care practices for a long time but newer formats have been emerging so fast that our minds, much less our staff’s minds, may be spinning.
Our old friend the telephone, a time-soaking nemesis for scheduling, checking coverage, questions calls, prescribing, quick consults, and follow-up is being replaced by EHR portals and SMS for messaging (e.g. DoctorConnect, SimplePractice), drop-in televisits and patient education links on our websites (e.g. Schmitt Pediatric Care, Remedy Connect), and chatbots for scheduling (e.g. CHEC-UP). While time is saved, what is lost may be hearing the subtext of anxiety or misperceptions in parents’ voices that would change our advice and the empathetic human connection in conversations with our patients. A hybrid approach may be better.
The paper appointment book has been replaced by scheduling systems sometimes lacking in flexibility for double booking, sibling visits, and variable length or extremely valuable multi-professional visits. Allowing patients to book their own visits may place complex problems in inappropriate slots, so only allowing online requests for visits is safer. On the other hand, many of us can now squeeze in “same day” televisits (e.g. Blueberry Pediatrics), sometimes from outside our practice (e.g., zocdoc), to increase payments and even entice new patients to enroll.
Amazing advances in technology are being made in specialty care such as genetic modifications (CRISPR), immunotherapies (mRNA vaccines and AI drug design), robot-assisted surgery, and 3-D printing of body parts and prosthetics. Technology as treatment such as transcranial magnetic stimulation and vagal stimulation are finding value in psychiatry.
But beside being aware of and able to order such specialty technologies, innovations are now extending our senses in primary care such as amplified or visual stethoscopes, bedside ultrasound (e.g. Butterfly), remote visualization (oto-, endo-)scopes, photographic vision screens (e.g. iScreen) for skin lesion (VisualDx) and genetic syndrome facial recognition. We need to be sure that technologies are tested and calibrated for children and different racial groups and genders to provide safe and equitable care. Early adoption may not always be the best approach. Costs of technology, as usual, may limit access to these advanced care aids especially, as usual, in practices serving low income and rural communities.
Patients, especially younger parents and youth, now expect to participate and can directly benefit from technology as part of their health care. Validated parent or self-report screens (e.g. EHRs, Phreesia) can detect important issues early for more effective intervention. Such questionnaires typically provide a pass/fail result or score, but other delivery systems (e.g. CHADIS) include interpretation, assist patients/parents in setting visit priorities and health goals, and even chain results of one questionnaire to secondary screens to hone in on problems, sometimes obviating a time-consuming second visit. Patient-completed comprehensive questionnaires (e.g. Well Visit Planner, CHADIS) allow us time to use our skills to focus on concerns, education, and management rather than asking myriad routine questions. Some (e.g. CHADIS) even create visit documentation reducing our “pajama time” write ups (and burnout); automate repeated online measures to track progress; and use questionnaire results to trigger related patient-specific education and resources rather than the often-ignored generic EHR handouts.
Digital therapeutics such as apps for anxiety (e.g. Calm), depression (e.g. SparkRx, Cass), weight control (e.g. Noom, Lose it), fitness, or sleep tracking (e.g. Whoop) help educate and, in some cases, provide real-time feedback to personalize discovery of contributing factors in order to maintain motivation for positive health behavior change. Some video games improve ADHD symptoms (e.g. EndeavorRX). Virtual reality scenarios have been shown to desensitize those with PTSD and social anxiety or teach social skills to children with autism.
Systems that trigger resource listings (including apps) from screen results can help, but now with over 10,000 apps for mental health, knowing what to recommend for what conditions is a challenge for which ratings (e.g. MINDapps.org) can help. With few product reps visiting to tell us what’s new, we need to read critically about innovations, search the web, subscribe to the AAP SOAPM LISTSERV, visit exhibitors at professional meetings, and talk with peers.
All the digital data collected from health care technology, if assembled with privacy constraints and analyzed with advanced statistical methods, have the possibility, with or without inclusion of genomic data, to allow for more accurate diagnostic and treatment decision support. While AI can search widely for patterns, it needs to be “trained” on appropriate data to make correct conclusions. We are all aware that the history determines 85% of both diagnosis and treatment decisions, particularly in primary care where x-rays or lab tests are not often needed.
But history in EHR notes is often idiosyncratic, entered hours after the visit by the clinician, and does not include the information needed to define diagnostic or guideline criteria, even if the clinician knows and considered those criteria. EHR templates are presented blank and are onerous and time consuming for clinicians. In addition, individual patient barriers to care, preferences, and environmental or subjective concerns are infrequently documented even though they may make the biggest difference to adherence and/or outcomes.
Notes made from voice to text digital AI translation of the encounter (e.g. Nuance DAX) are even less likely to include diagnostic criteria as it would be inappropriate to speak these. To use EHR history data to train AI and to test efficacy of care using variations of guidelines, guideline-related data is needed from online patient entries in questionnaires that are transformed to fill in templates along with some structured choices for clinician entries forming visit notes (e.g. CHADIS). New apps to facilitate clinician documentation of guidelines (e.g. AvoMD) could streamline visits as well as help document guideline criteria. The resulting combination of guideline-relevant patient histories and objective data to test and iteratively refine guidelines will allow a process known as a “Learning Health System.”
Technology to collect this kind of data can allow for the aspirational American Academy of Pediatrics CHILD Registry to approach this goal. Population-level data can provide surveillance for illness, toxins, effects of climate change, social drivers of health, and even effects of technologies themselves such as social media and remote learning so that we can attempt to make the best choices for the future.
Clinicians, staff, and patients will need to develop trust in technology as it infiltrates all aspects of health care. Professionals need both evidence and experience to trust a technology, which takes time and effort. Disinformation in the media may reduce trust or evoke unwarranted trust, as we have all seen regarding vaccines. Clear and coherent public health messaging can help but is no longer a panacea for developing trust in health care. Our nonjudgmental listening and informed opinions are needed more than ever.
The biggest issues for new technology are likely to be the need for workflow adjustments, changing our habit patterns, training, and cost/benefit analyses. With today’s high staff churn, confusion and even chaos can ensue when adopting new technology.
Staff need to be part of the selection process, if at all possible, and discuss how roles and flow will need to change. Having one staff member be a champion and expert for new tech can move adoption to a shared process rather than imposing “one more thing.” It is crucial to discuss the benefits for patients and staff even if the change is required. Sometimes cost savings can include a bonus for staff or free group lunches. Providing a certificate of achievement or title promotion for mastering new tech may be appropriate. Giving some time off from other tasks to learn new workflows can reduce resistance rather than just adding it on to a regular workload. Office “huddles” going forward can include examples of benefits staff have observed or heard about from the adoption. There are quality improvement processes that engage the team — some that earn MOC-4 or CEU credits — that apply to making workflow changes and measuring them iteratively.
If technology takes over important aspects of the work of medical professionals, even if it is faster and/or more accurate, it may degrade clinical observational, interactional, and decision-making skills through lack of use. It may also remove the sense of self-efficacy that motivates professionals to endure onerous training and desire to enter the field. Using technology may reduce empathetic interactions that are basic to humanistic motivation, work satisfaction, and even community respect. Moral injury is already rampant in medicine from restrictions on freedom to do what we see as important for our patients. Technology has great potential and already is enhancing our ability to provide the best care for patients but the risks need to be watched for and ameliorated.
When technology automates comprehensive visit documentation that highlights priority and risk areas from patient input and individualizes decision support, it can facilitate the personalized care that we and our patients want to experience. We must not be so awed, intrigued, or wary of new technology to miss its benefits nor give up our good clinical judgment about the technology or about our patients.
Dr. Howard is assistant professor of pediatrics at The Johns Hopkins University School of Medicine, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
We have all been using technology in our primary care practices for a long time but newer formats have been emerging so fast that our minds, much less our staff’s minds, may be spinning.
Our old friend the telephone, a time-soaking nemesis for scheduling, checking coverage, questions calls, prescribing, quick consults, and follow-up is being replaced by EHR portals and SMS for messaging (e.g. DoctorConnect, SimplePractice), drop-in televisits and patient education links on our websites (e.g. Schmitt Pediatric Care, Remedy Connect), and chatbots for scheduling (e.g. CHEC-UP). While time is saved, what is lost may be hearing the subtext of anxiety or misperceptions in parents’ voices that would change our advice and the empathetic human connection in conversations with our patients. A hybrid approach may be better.
The paper appointment book has been replaced by scheduling systems sometimes lacking in flexibility for double booking, sibling visits, and variable length or extremely valuable multi-professional visits. Allowing patients to book their own visits may place complex problems in inappropriate slots, so only allowing online requests for visits is safer. On the other hand, many of us can now squeeze in “same day” televisits (e.g. Blueberry Pediatrics), sometimes from outside our practice (e.g., zocdoc), to increase payments and even entice new patients to enroll.
Amazing advances in technology are being made in specialty care such as genetic modifications (CRISPR), immunotherapies (mRNA vaccines and AI drug design), robot-assisted surgery, and 3-D printing of body parts and prosthetics. Technology as treatment such as transcranial magnetic stimulation and vagal stimulation are finding value in psychiatry.
But beside being aware of and able to order such specialty technologies, innovations are now extending our senses in primary care such as amplified or visual stethoscopes, bedside ultrasound (e.g. Butterfly), remote visualization (oto-, endo-)scopes, photographic vision screens (e.g. iScreen) for skin lesion (VisualDx) and genetic syndrome facial recognition. We need to be sure that technologies are tested and calibrated for children and different racial groups and genders to provide safe and equitable care. Early adoption may not always be the best approach. Costs of technology, as usual, may limit access to these advanced care aids especially, as usual, in practices serving low income and rural communities.
Patients, especially younger parents and youth, now expect to participate and can directly benefit from technology as part of their health care. Validated parent or self-report screens (e.g. EHRs, Phreesia) can detect important issues early for more effective intervention. Such questionnaires typically provide a pass/fail result or score, but other delivery systems (e.g. CHADIS) include interpretation, assist patients/parents in setting visit priorities and health goals, and even chain results of one questionnaire to secondary screens to hone in on problems, sometimes obviating a time-consuming second visit. Patient-completed comprehensive questionnaires (e.g. Well Visit Planner, CHADIS) allow us time to use our skills to focus on concerns, education, and management rather than asking myriad routine questions. Some (e.g. CHADIS) even create visit documentation reducing our “pajama time” write ups (and burnout); automate repeated online measures to track progress; and use questionnaire results to trigger related patient-specific education and resources rather than the often-ignored generic EHR handouts.
Digital therapeutics such as apps for anxiety (e.g. Calm), depression (e.g. SparkRx, Cass), weight control (e.g. Noom, Lose it), fitness, or sleep tracking (e.g. Whoop) help educate and, in some cases, provide real-time feedback to personalize discovery of contributing factors in order to maintain motivation for positive health behavior change. Some video games improve ADHD symptoms (e.g. EndeavorRX). Virtual reality scenarios have been shown to desensitize those with PTSD and social anxiety or teach social skills to children with autism.
Systems that trigger resource listings (including apps) from screen results can help, but now with over 10,000 apps for mental health, knowing what to recommend for what conditions is a challenge for which ratings (e.g. MINDapps.org) can help. With few product reps visiting to tell us what’s new, we need to read critically about innovations, search the web, subscribe to the AAP SOAPM LISTSERV, visit exhibitors at professional meetings, and talk with peers.
All the digital data collected from health care technology, if assembled with privacy constraints and analyzed with advanced statistical methods, have the possibility, with or without inclusion of genomic data, to allow for more accurate diagnostic and treatment decision support. While AI can search widely for patterns, it needs to be “trained” on appropriate data to make correct conclusions. We are all aware that the history determines 85% of both diagnosis and treatment decisions, particularly in primary care where x-rays or lab tests are not often needed.
But history in EHR notes is often idiosyncratic, entered hours after the visit by the clinician, and does not include the information needed to define diagnostic or guideline criteria, even if the clinician knows and considered those criteria. EHR templates are presented blank and are onerous and time consuming for clinicians. In addition, individual patient barriers to care, preferences, and environmental or subjective concerns are infrequently documented even though they may make the biggest difference to adherence and/or outcomes.
Notes made from voice to text digital AI translation of the encounter (e.g. Nuance DAX) are even less likely to include diagnostic criteria as it would be inappropriate to speak these. To use EHR history data to train AI and to test efficacy of care using variations of guidelines, guideline-related data is needed from online patient entries in questionnaires that are transformed to fill in templates along with some structured choices for clinician entries forming visit notes (e.g. CHADIS). New apps to facilitate clinician documentation of guidelines (e.g. AvoMD) could streamline visits as well as help document guideline criteria. The resulting combination of guideline-relevant patient histories and objective data to test and iteratively refine guidelines will allow a process known as a “Learning Health System.”
Technology to collect this kind of data can allow for the aspirational American Academy of Pediatrics CHILD Registry to approach this goal. Population-level data can provide surveillance for illness, toxins, effects of climate change, social drivers of health, and even effects of technologies themselves such as social media and remote learning so that we can attempt to make the best choices for the future.
Clinicians, staff, and patients will need to develop trust in technology as it infiltrates all aspects of health care. Professionals need both evidence and experience to trust a technology, which takes time and effort. Disinformation in the media may reduce trust or evoke unwarranted trust, as we have all seen regarding vaccines. Clear and coherent public health messaging can help but is no longer a panacea for developing trust in health care. Our nonjudgmental listening and informed opinions are needed more than ever.
The biggest issues for new technology are likely to be the need for workflow adjustments, changing our habit patterns, training, and cost/benefit analyses. With today’s high staff churn, confusion and even chaos can ensue when adopting new technology.
Staff need to be part of the selection process, if at all possible, and discuss how roles and flow will need to change. Having one staff member be a champion and expert for new tech can move adoption to a shared process rather than imposing “one more thing.” It is crucial to discuss the benefits for patients and staff even if the change is required. Sometimes cost savings can include a bonus for staff or free group lunches. Providing a certificate of achievement or title promotion for mastering new tech may be appropriate. Giving some time off from other tasks to learn new workflows can reduce resistance rather than just adding it on to a regular workload. Office “huddles” going forward can include examples of benefits staff have observed or heard about from the adoption. There are quality improvement processes that engage the team — some that earn MOC-4 or CEU credits — that apply to making workflow changes and measuring them iteratively.
If technology takes over important aspects of the work of medical professionals, even if it is faster and/or more accurate, it may degrade clinical observational, interactional, and decision-making skills through lack of use. It may also remove the sense of self-efficacy that motivates professionals to endure onerous training and desire to enter the field. Using technology may reduce empathetic interactions that are basic to humanistic motivation, work satisfaction, and even community respect. Moral injury is already rampant in medicine from restrictions on freedom to do what we see as important for our patients. Technology has great potential and already is enhancing our ability to provide the best care for patients but the risks need to be watched for and ameliorated.
When technology automates comprehensive visit documentation that highlights priority and risk areas from patient input and individualizes decision support, it can facilitate the personalized care that we and our patients want to experience. We must not be so awed, intrigued, or wary of new technology to miss its benefits nor give up our good clinical judgment about the technology or about our patients.
Dr. Howard is assistant professor of pediatrics at The Johns Hopkins University School of Medicine, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
We have all been using technology in our primary care practices for a long time but newer formats have been emerging so fast that our minds, much less our staff’s minds, may be spinning.
Our old friend the telephone, a time-soaking nemesis for scheduling, checking coverage, questions calls, prescribing, quick consults, and follow-up is being replaced by EHR portals and SMS for messaging (e.g. DoctorConnect, SimplePractice), drop-in televisits and patient education links on our websites (e.g. Schmitt Pediatric Care, Remedy Connect), and chatbots for scheduling (e.g. CHEC-UP). While time is saved, what is lost may be hearing the subtext of anxiety or misperceptions in parents’ voices that would change our advice and the empathetic human connection in conversations with our patients. A hybrid approach may be better.
The paper appointment book has been replaced by scheduling systems sometimes lacking in flexibility for double booking, sibling visits, and variable length or extremely valuable multi-professional visits. Allowing patients to book their own visits may place complex problems in inappropriate slots, so only allowing online requests for visits is safer. On the other hand, many of us can now squeeze in “same day” televisits (e.g. Blueberry Pediatrics), sometimes from outside our practice (e.g., zocdoc), to increase payments and even entice new patients to enroll.
Amazing advances in technology are being made in specialty care such as genetic modifications (CRISPR), immunotherapies (mRNA vaccines and AI drug design), robot-assisted surgery, and 3-D printing of body parts and prosthetics. Technology as treatment such as transcranial magnetic stimulation and vagal stimulation are finding value in psychiatry.
But beside being aware of and able to order such specialty technologies, innovations are now extending our senses in primary care such as amplified or visual stethoscopes, bedside ultrasound (e.g. Butterfly), remote visualization (oto-, endo-)scopes, photographic vision screens (e.g. iScreen) for skin lesion (VisualDx) and genetic syndrome facial recognition. We need to be sure that technologies are tested and calibrated for children and different racial groups and genders to provide safe and equitable care. Early adoption may not always be the best approach. Costs of technology, as usual, may limit access to these advanced care aids especially, as usual, in practices serving low income and rural communities.
Patients, especially younger parents and youth, now expect to participate and can directly benefit from technology as part of their health care. Validated parent or self-report screens (e.g. EHRs, Phreesia) can detect important issues early for more effective intervention. Such questionnaires typically provide a pass/fail result or score, but other delivery systems (e.g. CHADIS) include interpretation, assist patients/parents in setting visit priorities and health goals, and even chain results of one questionnaire to secondary screens to hone in on problems, sometimes obviating a time-consuming second visit. Patient-completed comprehensive questionnaires (e.g. Well Visit Planner, CHADIS) allow us time to use our skills to focus on concerns, education, and management rather than asking myriad routine questions. Some (e.g. CHADIS) even create visit documentation reducing our “pajama time” write ups (and burnout); automate repeated online measures to track progress; and use questionnaire results to trigger related patient-specific education and resources rather than the often-ignored generic EHR handouts.
Digital therapeutics such as apps for anxiety (e.g. Calm), depression (e.g. SparkRx, Cass), weight control (e.g. Noom, Lose it), fitness, or sleep tracking (e.g. Whoop) help educate and, in some cases, provide real-time feedback to personalize discovery of contributing factors in order to maintain motivation for positive health behavior change. Some video games improve ADHD symptoms (e.g. EndeavorRX). Virtual reality scenarios have been shown to desensitize those with PTSD and social anxiety or teach social skills to children with autism.
Systems that trigger resource listings (including apps) from screen results can help, but now with over 10,000 apps for mental health, knowing what to recommend for what conditions is a challenge for which ratings (e.g. MINDapps.org) can help. With few product reps visiting to tell us what’s new, we need to read critically about innovations, search the web, subscribe to the AAP SOAPM LISTSERV, visit exhibitors at professional meetings, and talk with peers.
All the digital data collected from health care technology, if assembled with privacy constraints and analyzed with advanced statistical methods, have the possibility, with or without inclusion of genomic data, to allow for more accurate diagnostic and treatment decision support. While AI can search widely for patterns, it needs to be “trained” on appropriate data to make correct conclusions. We are all aware that the history determines 85% of both diagnosis and treatment decisions, particularly in primary care where x-rays or lab tests are not often needed.
But history in EHR notes is often idiosyncratic, entered hours after the visit by the clinician, and does not include the information needed to define diagnostic or guideline criteria, even if the clinician knows and considered those criteria. EHR templates are presented blank and are onerous and time consuming for clinicians. In addition, individual patient barriers to care, preferences, and environmental or subjective concerns are infrequently documented even though they may make the biggest difference to adherence and/or outcomes.
Notes made from voice to text digital AI translation of the encounter (e.g. Nuance DAX) are even less likely to include diagnostic criteria as it would be inappropriate to speak these. To use EHR history data to train AI and to test efficacy of care using variations of guidelines, guideline-related data is needed from online patient entries in questionnaires that are transformed to fill in templates along with some structured choices for clinician entries forming visit notes (e.g. CHADIS). New apps to facilitate clinician documentation of guidelines (e.g. AvoMD) could streamline visits as well as help document guideline criteria. The resulting combination of guideline-relevant patient histories and objective data to test and iteratively refine guidelines will allow a process known as a “Learning Health System.”
Technology to collect this kind of data can allow for the aspirational American Academy of Pediatrics CHILD Registry to approach this goal. Population-level data can provide surveillance for illness, toxins, effects of climate change, social drivers of health, and even effects of technologies themselves such as social media and remote learning so that we can attempt to make the best choices for the future.
Clinicians, staff, and patients will need to develop trust in technology as it infiltrates all aspects of health care. Professionals need both evidence and experience to trust a technology, which takes time and effort. Disinformation in the media may reduce trust or evoke unwarranted trust, as we have all seen regarding vaccines. Clear and coherent public health messaging can help but is no longer a panacea for developing trust in health care. Our nonjudgmental listening and informed opinions are needed more than ever.
The biggest issues for new technology are likely to be the need for workflow adjustments, changing our habit patterns, training, and cost/benefit analyses. With today’s high staff churn, confusion and even chaos can ensue when adopting new technology.
Staff need to be part of the selection process, if at all possible, and discuss how roles and flow will need to change. Having one staff member be a champion and expert for new tech can move adoption to a shared process rather than imposing “one more thing.” It is crucial to discuss the benefits for patients and staff even if the change is required. Sometimes cost savings can include a bonus for staff or free group lunches. Providing a certificate of achievement or title promotion for mastering new tech may be appropriate. Giving some time off from other tasks to learn new workflows can reduce resistance rather than just adding it on to a regular workload. Office “huddles” going forward can include examples of benefits staff have observed or heard about from the adoption. There are quality improvement processes that engage the team — some that earn MOC-4 or CEU credits — that apply to making workflow changes and measuring them iteratively.
If technology takes over important aspects of the work of medical professionals, even if it is faster and/or more accurate, it may degrade clinical observational, interactional, and decision-making skills through lack of use. It may also remove the sense of self-efficacy that motivates professionals to endure onerous training and desire to enter the field. Using technology may reduce empathetic interactions that are basic to humanistic motivation, work satisfaction, and even community respect. Moral injury is already rampant in medicine from restrictions on freedom to do what we see as important for our patients. Technology has great potential and already is enhancing our ability to provide the best care for patients but the risks need to be watched for and ameliorated.
When technology automates comprehensive visit documentation that highlights priority and risk areas from patient input and individualizes decision support, it can facilitate the personalized care that we and our patients want to experience. We must not be so awed, intrigued, or wary of new technology to miss its benefits nor give up our good clinical judgment about the technology or about our patients.
Dr. Howard is assistant professor of pediatrics at The Johns Hopkins University School of Medicine, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Acne stigma persists across social and professional settings
from more than 1300 individuals.

Self-stigma among people with acne has been examined in previous studies; however, “little is known about the prevalence and magnitude of stigmatizing attitudes of the general public toward individuals with acne,” wrote Ali Shields of Drexel University, Philadelphia, Pennsylvania, and her coauthors.
In the study, recently published in JAMA Dermatology, they reviewed survey data from 1357 adults aged 18 years and older who were identified through an online national research registry (ResearchMatch). The mean age of the participants was 42.4 years range). 67.7% were female.
Participants were randomly shown 1 of 12 standardized portraits of individuals that varied in skin tone, sex, and acne severity. They responded to questions about stigmatizing attitudes with respect to the portrait, including stereotype endorsement and desire for social distance.
With regard to social distance, survey participants were significantly less comfortable being friends with people with severe acne, compared with those who did not have acne (adjusted coefficient [aC], -0.28, P = .003). Compared with people without acne, participants also reported significantly less comfort in hiring someone with severe acne (aC, -0.33; P < .001), having physical contact (aC, -0.26; P = .006), dating (aC, -0.44; P = .004), and posting photos with that person on social media (aC, -0.50; P < .001).
With regard to common acne stereotypes, survey participants also rated individuals with severe acne as significantly more likely than those without acne to have poor hygiene and to be unattractive, unintelligent, unlikeable, immature, and untrustworthy (aCs, -1.04, -0.89, -0.42, -0.36, -0.52, and -0.40, respectively; P < .001 for all).
In a linear regression analysis, the researchers found no evidence of association modification by sex of the portraits presented, but found evidence that “the effect size of association of acne with stereotype endorsement was greater for individuals with dark skin.”
The findings were limited by several factors including the potential differences in degree of severity between images after the addition of acne because the baseline images were not exact controls for each other: Therefore comparisons between image sets based on skin tone or sex should be interpreted cautiously, the researchers noted. Other limitations included the homogeneous population of survey respondents and the inability to account for all aspects of stigma, they said.
However, the results illustrate the persistent stigma associated with acne and “highlight the need to identify approaches to reduce stigmatizing attitudes in the community and for adequate access to care, which might prevent negative downstream effects related to these stigmatizing attitudes,” the authors concluded.
The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal Diseases to corresponding author John S. Barbieri, MD. Coauthor Arash Mostaghimi, MD, disclosed personal fees from hims & hers, AbbVie, Sun Pharmaceutical Industries, Pfizer, Digital Diagnostics, Lilly, Equillium, ASLAN Pharmaceuticals, Boehringer Ingelheim, Fig.1 Beauty, Acom Healthcare, and Olaplex outside the current study. Dr. Barbieri disclosed personal fees from Dexcel Pharma for consulting outside the current study.
from more than 1300 individuals.

Self-stigma among people with acne has been examined in previous studies; however, “little is known about the prevalence and magnitude of stigmatizing attitudes of the general public toward individuals with acne,” wrote Ali Shields of Drexel University, Philadelphia, Pennsylvania, and her coauthors.
In the study, recently published in JAMA Dermatology, they reviewed survey data from 1357 adults aged 18 years and older who were identified through an online national research registry (ResearchMatch). The mean age of the participants was 42.4 years range). 67.7% were female.
Participants were randomly shown 1 of 12 standardized portraits of individuals that varied in skin tone, sex, and acne severity. They responded to questions about stigmatizing attitudes with respect to the portrait, including stereotype endorsement and desire for social distance.
With regard to social distance, survey participants were significantly less comfortable being friends with people with severe acne, compared with those who did not have acne (adjusted coefficient [aC], -0.28, P = .003). Compared with people without acne, participants also reported significantly less comfort in hiring someone with severe acne (aC, -0.33; P < .001), having physical contact (aC, -0.26; P = .006), dating (aC, -0.44; P = .004), and posting photos with that person on social media (aC, -0.50; P < .001).
With regard to common acne stereotypes, survey participants also rated individuals with severe acne as significantly more likely than those without acne to have poor hygiene and to be unattractive, unintelligent, unlikeable, immature, and untrustworthy (aCs, -1.04, -0.89, -0.42, -0.36, -0.52, and -0.40, respectively; P < .001 for all).
In a linear regression analysis, the researchers found no evidence of association modification by sex of the portraits presented, but found evidence that “the effect size of association of acne with stereotype endorsement was greater for individuals with dark skin.”
The findings were limited by several factors including the potential differences in degree of severity between images after the addition of acne because the baseline images were not exact controls for each other: Therefore comparisons between image sets based on skin tone or sex should be interpreted cautiously, the researchers noted. Other limitations included the homogeneous population of survey respondents and the inability to account for all aspects of stigma, they said.
However, the results illustrate the persistent stigma associated with acne and “highlight the need to identify approaches to reduce stigmatizing attitudes in the community and for adequate access to care, which might prevent negative downstream effects related to these stigmatizing attitudes,” the authors concluded.
The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal Diseases to corresponding author John S. Barbieri, MD. Coauthor Arash Mostaghimi, MD, disclosed personal fees from hims & hers, AbbVie, Sun Pharmaceutical Industries, Pfizer, Digital Diagnostics, Lilly, Equillium, ASLAN Pharmaceuticals, Boehringer Ingelheim, Fig.1 Beauty, Acom Healthcare, and Olaplex outside the current study. Dr. Barbieri disclosed personal fees from Dexcel Pharma for consulting outside the current study.
from more than 1300 individuals.

Self-stigma among people with acne has been examined in previous studies; however, “little is known about the prevalence and magnitude of stigmatizing attitudes of the general public toward individuals with acne,” wrote Ali Shields of Drexel University, Philadelphia, Pennsylvania, and her coauthors.
In the study, recently published in JAMA Dermatology, they reviewed survey data from 1357 adults aged 18 years and older who were identified through an online national research registry (ResearchMatch). The mean age of the participants was 42.4 years range). 67.7% were female.
Participants were randomly shown 1 of 12 standardized portraits of individuals that varied in skin tone, sex, and acne severity. They responded to questions about stigmatizing attitudes with respect to the portrait, including stereotype endorsement and desire for social distance.
With regard to social distance, survey participants were significantly less comfortable being friends with people with severe acne, compared with those who did not have acne (adjusted coefficient [aC], -0.28, P = .003). Compared with people without acne, participants also reported significantly less comfort in hiring someone with severe acne (aC, -0.33; P < .001), having physical contact (aC, -0.26; P = .006), dating (aC, -0.44; P = .004), and posting photos with that person on social media (aC, -0.50; P < .001).
With regard to common acne stereotypes, survey participants also rated individuals with severe acne as significantly more likely than those without acne to have poor hygiene and to be unattractive, unintelligent, unlikeable, immature, and untrustworthy (aCs, -1.04, -0.89, -0.42, -0.36, -0.52, and -0.40, respectively; P < .001 for all).
In a linear regression analysis, the researchers found no evidence of association modification by sex of the portraits presented, but found evidence that “the effect size of association of acne with stereotype endorsement was greater for individuals with dark skin.”
The findings were limited by several factors including the potential differences in degree of severity between images after the addition of acne because the baseline images were not exact controls for each other: Therefore comparisons between image sets based on skin tone or sex should be interpreted cautiously, the researchers noted. Other limitations included the homogeneous population of survey respondents and the inability to account for all aspects of stigma, they said.
However, the results illustrate the persistent stigma associated with acne and “highlight the need to identify approaches to reduce stigmatizing attitudes in the community and for adequate access to care, which might prevent negative downstream effects related to these stigmatizing attitudes,” the authors concluded.
The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal Diseases to corresponding author John S. Barbieri, MD. Coauthor Arash Mostaghimi, MD, disclosed personal fees from hims & hers, AbbVie, Sun Pharmaceutical Industries, Pfizer, Digital Diagnostics, Lilly, Equillium, ASLAN Pharmaceuticals, Boehringer Ingelheim, Fig.1 Beauty, Acom Healthcare, and Olaplex outside the current study. Dr. Barbieri disclosed personal fees from Dexcel Pharma for consulting outside the current study.
FROM JAMA DERMATOLOGY
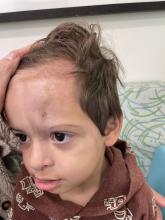
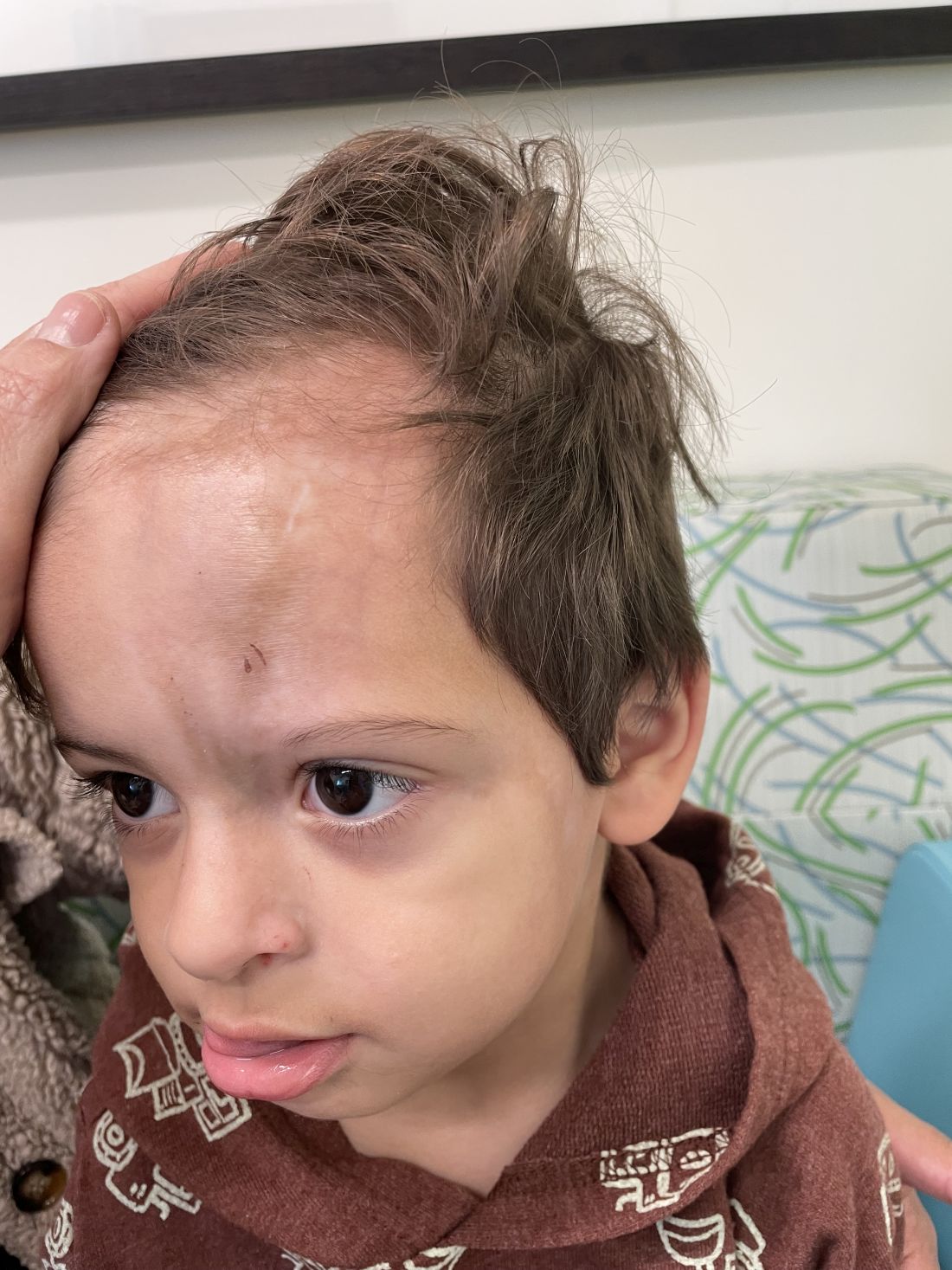
An 18-month-old male presents with a red mark on the forehead and nose
Following the initial presentation, the lesion was initially considered an acquired port wine stain and the child was referred for laser treatment. Upon reassessment during laser treatment a few months later, the lesion had progressed to hyper- and hypopigmented plaques with associated tissue sclerosis and bone atrophy on the mid forehead, nose, and scalp. Patches of alopecia and atrophy were observed on the frontal scalp. The diagnosis was revised to linear morphea en coup de sabre and the child was referred to pediatric rheumatology and commenced treatment with methotrexate and oral corticosteroids.
Linear morphea, a rare connective tissue disorder, primarily affects girls in the first 2 decades of life. Lesions can initially present in many ways. Usually, they present as hypo- or hyperpigmented patches, but may also present as lichenoid uncolored or pink plaques resembling lichen striatus. There may also be erythematous patches mimicking a capillary malformation, as seen in our patient. A recent article reviewing the progression of the lesions from erythematous patches to sclerosis suggests it occurs between 3 and 7 months of age. Subsequent stages manifest as significant atrophy, hypo- and hyperpigmentation, and in severe cases, bone atrophy and deformity, often causing substantial cosmetic disfigurement and functional impairment.
Pathophysiologically, linear morphea involves a complex interplay of immunologic, vascular, and fibrotic processes. While the initial triggers remain elusive, dysregulated immune responses leading to endothelial injury, subsequent activation of fibroblasts and myofibroblasts, and excessive collagen deposition are implicated. Angiogenic disturbances exacerbate tissue ischemia, perpetuating the fibrotic cascade. Alterations in cytokine signaling pathways, particularly TGF-beta and interleukin-6, play pivotal roles in promoting fibrosis and modulating the inflammatory milieu.
Diagnosis of linear morphea en coup de sabre relies on clinical examination, imaging (ultrasonography, MRI, CT scan), and skin biopsy for histopathological analysis. Imaging helps evaluate tissue involvement, while histology reveals characteristic dermal sclerosis, collagen deposition, and inflammation. Early-stage histology may show telangiectatic changes, complicating its differentiation from capillary malformation.
Treatment aims to mitigate symptoms, halt disease progression, and improve cosmesis and functionality. This involves a multidisciplinary approach with systemic medications, phototherapy, physical therapy, and surgical interventions in severe cases. Early identification is crucial for systemic treatments such as methotrexate and systemic corticosteroids to arrest disease progression. Other adjunctive therapies include topical corticosteroids, calcineurin inhibitors, and phototherapy. Surgical procedures like tissue expansion or autologous fat grafting may address tissue atrophy and deformities.
Linear morphea en coup de sabre presents diagnostic and therapeutic challenges because of its rarity and variable clinical course. Collaborative efforts among dermatologists, rheumatologists, radiologists, and surgeons are essential for accurate diagnosis, evaluation, and tailored management. Continued research into pathogenesis and novel therapeutic agents is pivotal to enhance understanding and improve outcomes for those affected by this enigmatic dermatologic condition.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Gomez-Garcia LA et al. Pediatr Dermatol. 2022 Mar;39(2):275-80.
Ng SS, Tay YK. J Cosmet Laser Ther. 2015;17(5):277-80.
Nijhawan RI et al. J Am Acad Dermatol. 2011 Apr;64(4):779-82.
Following the initial presentation, the lesion was initially considered an acquired port wine stain and the child was referred for laser treatment. Upon reassessment during laser treatment a few months later, the lesion had progressed to hyper- and hypopigmented plaques with associated tissue sclerosis and bone atrophy on the mid forehead, nose, and scalp. Patches of alopecia and atrophy were observed on the frontal scalp. The diagnosis was revised to linear morphea en coup de sabre and the child was referred to pediatric rheumatology and commenced treatment with methotrexate and oral corticosteroids.
Linear morphea, a rare connective tissue disorder, primarily affects girls in the first 2 decades of life. Lesions can initially present in many ways. Usually, they present as hypo- or hyperpigmented patches, but may also present as lichenoid uncolored or pink plaques resembling lichen striatus. There may also be erythematous patches mimicking a capillary malformation, as seen in our patient. A recent article reviewing the progression of the lesions from erythematous patches to sclerosis suggests it occurs between 3 and 7 months of age. Subsequent stages manifest as significant atrophy, hypo- and hyperpigmentation, and in severe cases, bone atrophy and deformity, often causing substantial cosmetic disfigurement and functional impairment.
Pathophysiologically, linear morphea involves a complex interplay of immunologic, vascular, and fibrotic processes. While the initial triggers remain elusive, dysregulated immune responses leading to endothelial injury, subsequent activation of fibroblasts and myofibroblasts, and excessive collagen deposition are implicated. Angiogenic disturbances exacerbate tissue ischemia, perpetuating the fibrotic cascade. Alterations in cytokine signaling pathways, particularly TGF-beta and interleukin-6, play pivotal roles in promoting fibrosis and modulating the inflammatory milieu.
Diagnosis of linear morphea en coup de sabre relies on clinical examination, imaging (ultrasonography, MRI, CT scan), and skin biopsy for histopathological analysis. Imaging helps evaluate tissue involvement, while histology reveals characteristic dermal sclerosis, collagen deposition, and inflammation. Early-stage histology may show telangiectatic changes, complicating its differentiation from capillary malformation.
Treatment aims to mitigate symptoms, halt disease progression, and improve cosmesis and functionality. This involves a multidisciplinary approach with systemic medications, phototherapy, physical therapy, and surgical interventions in severe cases. Early identification is crucial for systemic treatments such as methotrexate and systemic corticosteroids to arrest disease progression. Other adjunctive therapies include topical corticosteroids, calcineurin inhibitors, and phototherapy. Surgical procedures like tissue expansion or autologous fat grafting may address tissue atrophy and deformities.
Linear morphea en coup de sabre presents diagnostic and therapeutic challenges because of its rarity and variable clinical course. Collaborative efforts among dermatologists, rheumatologists, radiologists, and surgeons are essential for accurate diagnosis, evaluation, and tailored management. Continued research into pathogenesis and novel therapeutic agents is pivotal to enhance understanding and improve outcomes for those affected by this enigmatic dermatologic condition.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Gomez-Garcia LA et al. Pediatr Dermatol. 2022 Mar;39(2):275-80.
Ng SS, Tay YK. J Cosmet Laser Ther. 2015;17(5):277-80.
Nijhawan RI et al. J Am Acad Dermatol. 2011 Apr;64(4):779-82.
Following the initial presentation, the lesion was initially considered an acquired port wine stain and the child was referred for laser treatment. Upon reassessment during laser treatment a few months later, the lesion had progressed to hyper- and hypopigmented plaques with associated tissue sclerosis and bone atrophy on the mid forehead, nose, and scalp. Patches of alopecia and atrophy were observed on the frontal scalp. The diagnosis was revised to linear morphea en coup de sabre and the child was referred to pediatric rheumatology and commenced treatment with methotrexate and oral corticosteroids.
Linear morphea, a rare connective tissue disorder, primarily affects girls in the first 2 decades of life. Lesions can initially present in many ways. Usually, they present as hypo- or hyperpigmented patches, but may also present as lichenoid uncolored or pink plaques resembling lichen striatus. There may also be erythematous patches mimicking a capillary malformation, as seen in our patient. A recent article reviewing the progression of the lesions from erythematous patches to sclerosis suggests it occurs between 3 and 7 months of age. Subsequent stages manifest as significant atrophy, hypo- and hyperpigmentation, and in severe cases, bone atrophy and deformity, often causing substantial cosmetic disfigurement and functional impairment.
Pathophysiologically, linear morphea involves a complex interplay of immunologic, vascular, and fibrotic processes. While the initial triggers remain elusive, dysregulated immune responses leading to endothelial injury, subsequent activation of fibroblasts and myofibroblasts, and excessive collagen deposition are implicated. Angiogenic disturbances exacerbate tissue ischemia, perpetuating the fibrotic cascade. Alterations in cytokine signaling pathways, particularly TGF-beta and interleukin-6, play pivotal roles in promoting fibrosis and modulating the inflammatory milieu.
Diagnosis of linear morphea en coup de sabre relies on clinical examination, imaging (ultrasonography, MRI, CT scan), and skin biopsy for histopathological analysis. Imaging helps evaluate tissue involvement, while histology reveals characteristic dermal sclerosis, collagen deposition, and inflammation. Early-stage histology may show telangiectatic changes, complicating its differentiation from capillary malformation.
Treatment aims to mitigate symptoms, halt disease progression, and improve cosmesis and functionality. This involves a multidisciplinary approach with systemic medications, phototherapy, physical therapy, and surgical interventions in severe cases. Early identification is crucial for systemic treatments such as methotrexate and systemic corticosteroids to arrest disease progression. Other adjunctive therapies include topical corticosteroids, calcineurin inhibitors, and phototherapy. Surgical procedures like tissue expansion or autologous fat grafting may address tissue atrophy and deformities.
Linear morphea en coup de sabre presents diagnostic and therapeutic challenges because of its rarity and variable clinical course. Collaborative efforts among dermatologists, rheumatologists, radiologists, and surgeons are essential for accurate diagnosis, evaluation, and tailored management. Continued research into pathogenesis and novel therapeutic agents is pivotal to enhance understanding and improve outcomes for those affected by this enigmatic dermatologic condition.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Gomez-Garcia LA et al. Pediatr Dermatol. 2022 Mar;39(2):275-80.
Ng SS, Tay YK. J Cosmet Laser Ther. 2015;17(5):277-80.
Nijhawan RI et al. J Am Acad Dermatol. 2011 Apr;64(4):779-82.
On examination, a faint pink patch was observed on the right forehead, frontal scalp, and nose. The lesion paled under pressure, with small areas of hair loss on the scalp. No atrophy was noted.
Sickle Cell Gene Therapy ‘Truly Transformative’
.
More specifically, a single infusion of lovo-cel led to complete resolution of vaso-occlusive events in 88% of patients, with 94% achieving complete resolution of severe events. All 10 adolescents in the study achieved complete resolution of vaso-occlusive events. Most patients remained free of vaso-occlusive events at their last follow-up.
“This is a one-time, truly transformative treatment with lovo-cel,” lead author Julie Kanter, MD, director of the adult sickle cell clinic at the University of Alabama in Birmingham, said in a media briefing at the annual meeting of the American Society of Hematology. The gene therapy can essentially eliminate vaso-occlusive events in patients with sickle cell disease and lead to normal hemoglobin levels, Dr. Kanter added.
For “anybody who has rounded on the inpatient floor and taken care of adolescents admitted with a pain crisis multiple times a year,” seeing these results “is so compelling,” commented Sarah O’Brien, MD, a pediatric hematologist at Nationwide Children’s Hospital in Columbus, Ohio, who moderated the briefing but was not involved in the study.
One and Done
Sickle cell disease, a debilitating and potentially life-threatening blood disorder, affects an estimated 100,000 people in the US.
People with the condition have a mutation in hemoglobin, which causes red blood cells to develop an abnormal sickle shape. These sickled cells block the flow of blood, ultimately depriving tissues of oxygen and leading to organ damage and severe pain, known as vaso-occlusive events.
On Dec. 8, the U.S. Food and Drug Administration (FDA) approved lovo-cel for patients aged 12 years or older with severe sickle cell disease alongside another gene-editing therapy called exagamglogene autotemcel or exa-cel (Casgevy, Vertex Pharmaceuticals and Crispr Therapeutics). The two therapies use different gene-editing approaches — exa-cel is the first to use the gene-editing tool CRISPR while lovo-cel uses a lentiviral vector.
Both are one-time, single-dose cell-based gene therapies.
With lovo-cel, patients first undergo a transfusion regimen and myeloablative conditioning with busulfan to collect cells that can then be genetically modified. A patient’s harvested cells are modified with an anti-sickling version of hemoglobin A, HbAT87Q. Patients then receive an infusion of these edited cells and remain in the hospital during engraftment and reconstitution.
Dr. Kanter presented long-term follow-up data on 47 patients enrolled in phase 1/2 and phase 3 studies of lovo-cel.
All patients had stable HbAT87Q levels from 6 months to their last follow-up at a median of 35.5 months.
Most patients achieved a durable globin response through their final follow-up visit.
Among the 34 evaluable patients, 88% had complete resolution of vaso-occlusive events 6 to 18 months after their infusion, including all 10 adolescent patients. Almost all patients (94%) achieved complete resolution of serious vaso-occlusive events.
In the few patients who experienced posttreatment vaso-occlusive events, these individuals still achieved major reductions in hospital admissions and hospital days.
Among 20 patients followed for at least 3 years, more than half had clinically meaningful improvements in pain intensity, pain interference, and fatigue.
Most treatment-related adverse events occurred within 1 year of lovo-cel infusions and were primarily related to busulfan conditioning. No cases of veno-occlusive liver disease, graft failure, or graft vs host disease occurred, and patients did not have complications related to the viral vector. No patients who had a history of stroke prior to lovo-cel therapy experienced a post-therapy stroke.
One patient died at baseline from significant cardiopulmonary disease related to sickle cell disease, but the death was considered unrelated to lovo-cel therapy.
To see a one-time treatment that essentially eradicates vaso-occlusive events is “really unparalleled,” said Steven Pipe, MD, from the University of Michigan School of Medicine in Ann Arbor, who presented data on a different study at the briefing.
However, Dr. Kanter noted, “it’s important to highlight that many of these individuals come into this therapy with significant disease and end-organ complications, and this will be something we will really need to follow long-term to understand how much this therapy can stabilize or reverse these complications.”
The studies were funded by bluebird bio. Dr. Kanter disclosed honoraria from the company and consulting/advising activities and receipt of research funding from multiple other entities. Dr. O’Brien disclosed consultancy for AstraZeneca, honoraria from Pharmacosmos, and research funding from Bristol Myers Squibb. Dr. Pipe disclosed consulting activities from multiple companies, not including bluebird bio.
A version of this article appeared on Medscape.com.
.
More specifically, a single infusion of lovo-cel led to complete resolution of vaso-occlusive events in 88% of patients, with 94% achieving complete resolution of severe events. All 10 adolescents in the study achieved complete resolution of vaso-occlusive events. Most patients remained free of vaso-occlusive events at their last follow-up.
“This is a one-time, truly transformative treatment with lovo-cel,” lead author Julie Kanter, MD, director of the adult sickle cell clinic at the University of Alabama in Birmingham, said in a media briefing at the annual meeting of the American Society of Hematology. The gene therapy can essentially eliminate vaso-occlusive events in patients with sickle cell disease and lead to normal hemoglobin levels, Dr. Kanter added.
For “anybody who has rounded on the inpatient floor and taken care of adolescents admitted with a pain crisis multiple times a year,” seeing these results “is so compelling,” commented Sarah O’Brien, MD, a pediatric hematologist at Nationwide Children’s Hospital in Columbus, Ohio, who moderated the briefing but was not involved in the study.
One and Done
Sickle cell disease, a debilitating and potentially life-threatening blood disorder, affects an estimated 100,000 people in the US.
People with the condition have a mutation in hemoglobin, which causes red blood cells to develop an abnormal sickle shape. These sickled cells block the flow of blood, ultimately depriving tissues of oxygen and leading to organ damage and severe pain, known as vaso-occlusive events.
On Dec. 8, the U.S. Food and Drug Administration (FDA) approved lovo-cel for patients aged 12 years or older with severe sickle cell disease alongside another gene-editing therapy called exagamglogene autotemcel or exa-cel (Casgevy, Vertex Pharmaceuticals and Crispr Therapeutics). The two therapies use different gene-editing approaches — exa-cel is the first to use the gene-editing tool CRISPR while lovo-cel uses a lentiviral vector.
Both are one-time, single-dose cell-based gene therapies.
With lovo-cel, patients first undergo a transfusion regimen and myeloablative conditioning with busulfan to collect cells that can then be genetically modified. A patient’s harvested cells are modified with an anti-sickling version of hemoglobin A, HbAT87Q. Patients then receive an infusion of these edited cells and remain in the hospital during engraftment and reconstitution.
Dr. Kanter presented long-term follow-up data on 47 patients enrolled in phase 1/2 and phase 3 studies of lovo-cel.
All patients had stable HbAT87Q levels from 6 months to their last follow-up at a median of 35.5 months.
Most patients achieved a durable globin response through their final follow-up visit.
Among the 34 evaluable patients, 88% had complete resolution of vaso-occlusive events 6 to 18 months after their infusion, including all 10 adolescent patients. Almost all patients (94%) achieved complete resolution of serious vaso-occlusive events.
In the few patients who experienced posttreatment vaso-occlusive events, these individuals still achieved major reductions in hospital admissions and hospital days.
Among 20 patients followed for at least 3 years, more than half had clinically meaningful improvements in pain intensity, pain interference, and fatigue.
Most treatment-related adverse events occurred within 1 year of lovo-cel infusions and were primarily related to busulfan conditioning. No cases of veno-occlusive liver disease, graft failure, or graft vs host disease occurred, and patients did not have complications related to the viral vector. No patients who had a history of stroke prior to lovo-cel therapy experienced a post-therapy stroke.
One patient died at baseline from significant cardiopulmonary disease related to sickle cell disease, but the death was considered unrelated to lovo-cel therapy.
To see a one-time treatment that essentially eradicates vaso-occlusive events is “really unparalleled,” said Steven Pipe, MD, from the University of Michigan School of Medicine in Ann Arbor, who presented data on a different study at the briefing.
However, Dr. Kanter noted, “it’s important to highlight that many of these individuals come into this therapy with significant disease and end-organ complications, and this will be something we will really need to follow long-term to understand how much this therapy can stabilize or reverse these complications.”
The studies were funded by bluebird bio. Dr. Kanter disclosed honoraria from the company and consulting/advising activities and receipt of research funding from multiple other entities. Dr. O’Brien disclosed consultancy for AstraZeneca, honoraria from Pharmacosmos, and research funding from Bristol Myers Squibb. Dr. Pipe disclosed consulting activities from multiple companies, not including bluebird bio.
A version of this article appeared on Medscape.com.
.
More specifically, a single infusion of lovo-cel led to complete resolution of vaso-occlusive events in 88% of patients, with 94% achieving complete resolution of severe events. All 10 adolescents in the study achieved complete resolution of vaso-occlusive events. Most patients remained free of vaso-occlusive events at their last follow-up.
“This is a one-time, truly transformative treatment with lovo-cel,” lead author Julie Kanter, MD, director of the adult sickle cell clinic at the University of Alabama in Birmingham, said in a media briefing at the annual meeting of the American Society of Hematology. The gene therapy can essentially eliminate vaso-occlusive events in patients with sickle cell disease and lead to normal hemoglobin levels, Dr. Kanter added.
For “anybody who has rounded on the inpatient floor and taken care of adolescents admitted with a pain crisis multiple times a year,” seeing these results “is so compelling,” commented Sarah O’Brien, MD, a pediatric hematologist at Nationwide Children’s Hospital in Columbus, Ohio, who moderated the briefing but was not involved in the study.
One and Done
Sickle cell disease, a debilitating and potentially life-threatening blood disorder, affects an estimated 100,000 people in the US.
People with the condition have a mutation in hemoglobin, which causes red blood cells to develop an abnormal sickle shape. These sickled cells block the flow of blood, ultimately depriving tissues of oxygen and leading to organ damage and severe pain, known as vaso-occlusive events.
On Dec. 8, the U.S. Food and Drug Administration (FDA) approved lovo-cel for patients aged 12 years or older with severe sickle cell disease alongside another gene-editing therapy called exagamglogene autotemcel or exa-cel (Casgevy, Vertex Pharmaceuticals and Crispr Therapeutics). The two therapies use different gene-editing approaches — exa-cel is the first to use the gene-editing tool CRISPR while lovo-cel uses a lentiviral vector.
Both are one-time, single-dose cell-based gene therapies.
With lovo-cel, patients first undergo a transfusion regimen and myeloablative conditioning with busulfan to collect cells that can then be genetically modified. A patient’s harvested cells are modified with an anti-sickling version of hemoglobin A, HbAT87Q. Patients then receive an infusion of these edited cells and remain in the hospital during engraftment and reconstitution.
Dr. Kanter presented long-term follow-up data on 47 patients enrolled in phase 1/2 and phase 3 studies of lovo-cel.
All patients had stable HbAT87Q levels from 6 months to their last follow-up at a median of 35.5 months.
Most patients achieved a durable globin response through their final follow-up visit.
Among the 34 evaluable patients, 88% had complete resolution of vaso-occlusive events 6 to 18 months after their infusion, including all 10 adolescent patients. Almost all patients (94%) achieved complete resolution of serious vaso-occlusive events.
In the few patients who experienced posttreatment vaso-occlusive events, these individuals still achieved major reductions in hospital admissions and hospital days.
Among 20 patients followed for at least 3 years, more than half had clinically meaningful improvements in pain intensity, pain interference, and fatigue.
Most treatment-related adverse events occurred within 1 year of lovo-cel infusions and were primarily related to busulfan conditioning. No cases of veno-occlusive liver disease, graft failure, or graft vs host disease occurred, and patients did not have complications related to the viral vector. No patients who had a history of stroke prior to lovo-cel therapy experienced a post-therapy stroke.
One patient died at baseline from significant cardiopulmonary disease related to sickle cell disease, but the death was considered unrelated to lovo-cel therapy.
To see a one-time treatment that essentially eradicates vaso-occlusive events is “really unparalleled,” said Steven Pipe, MD, from the University of Michigan School of Medicine in Ann Arbor, who presented data on a different study at the briefing.
However, Dr. Kanter noted, “it’s important to highlight that many of these individuals come into this therapy with significant disease and end-organ complications, and this will be something we will really need to follow long-term to understand how much this therapy can stabilize or reverse these complications.”
The studies were funded by bluebird bio. Dr. Kanter disclosed honoraria from the company and consulting/advising activities and receipt of research funding from multiple other entities. Dr. O’Brien disclosed consultancy for AstraZeneca, honoraria from Pharmacosmos, and research funding from Bristol Myers Squibb. Dr. Pipe disclosed consulting activities from multiple companies, not including bluebird bio.
A version of this article appeared on Medscape.com.
FROM ASH 2023
This test may guide AML therapy for Black pediatric patients
.
The score, dubbed ACS10 and initially highlighted in a 2022 report, predicts how well patients will respond to cytarabine based on their genetic make-up, and has the potential to personalize treatment for Black pediatric patients, a group that often has worse outcomes than White patients.
In the current study, presented at the annual meeting of the American Society of Hematology (ASH) , Black patients with low ACS10 scores had significantly worse outcomes compared with those with high scores when initially treated with low-dose cytarabine, daunorubicin, and etoposide.
The difference in outcomes disappeared, however, for patients who received high-dose cytarabine, daunorubicin, and etoposide or clofarabine and cytarabine.
The genetic traits revealed by the test likely help explain why Black patients with AML typically fare worse on certain regimens, Cynthia E. Dunbar, MD, chief of the Translational Stem Cell Biology Branch at the National Heart, Lung, and Blood Institute, commented in an ASH press preview briefing.
This study also suggests that clinicians should perform testing for genetic variants and biomarkers that impact outcomes “instead of assuming that a certain dose should be given simply based on perceived or reported race or ethnicity,” said Dr. Dunbar, also secretary of ASH.
The ACS10 test, derived from a combination of 10 single nucleotide polymorphisms, is not yet available, but one could be developed to help guide treatment decisions for clinicians, especially those in developing countries where AML treatment can be very expensive, said study lead author Jatinder Lamba, PhD, MSc, of the University of Florida College of Pharmacy, Gainesville, at an ASH press briefing on Thursday.
Prior research shows that Black pediatric patients with AML often have worse outcomes than White patients. A recent study , for instance, found Black patients with AML, especially those aged 18 to 29 years, had a higher early death rate compared with White patients (16% vs 3%) and significantly lower 5-year overall survival rates (22% vs 51%). The authors of this study suggested that genetic differences between young Black and White patients could help explain the disparity.
In the new analysis, Dr. Lamba and colleagues explored how outcomes by race and cytarabine pharmacogenomics varied in pediatric patients with AML.
The study included 86 Black patients and 359 White patients with newly diagnosed AML treated on two multi-institutional clinical trials. The patients received one of three initial treatments that included cytarabine: high-dose or low-dose cytarabine, daunorubicin, and etoposide, or clofarabine and cytarabine.
Most Black patients in the analysis (73%) had low ACS10 scores compared with 30% of White patients.
Unlike other recent reports, this study found that Black and White patients had similar complete remission rates following two courses of induction therapy (92.6% vs 95%) as well as similar rates of minimal residual disease negativity after one course (55.8% vs 55.4%).
Event-free survival (EFS) and overall survival rates were also similar, with 5-year EFS estimates at 58.3% for Black patients and 58.2% for White patients and overall survival rates at 63.8% vs 69.4%, respectively (P = .24).
However, when separating outcomes by ACS10 scores, Black patients with low scores had significantly worse EFS following low-dose cytarabine, daunorubicin, and etoposide compared with those with high ACS10 scores. And when these patients received high-dose cytarabine, daunorubicin, and etoposide or clofarabine and cytarabine induction therapy instead, the differences went away.
Overall, Black patients demonstrated significantly better EFS following treatment with clofarabine and cytarabine compared with the low-dose cytarabine triple therapy (hazard ratio, 0.17; P = .01). After adjusting for cofounders, clofarabine and cytarabine induction was the best treatment for Black patients with low ACS10 scores (HR for EFS, 0.2).
“Our results suggest that pharmacogenomics differences between Black and White patients should be considered when tailoring induction regimens to improve outcomes of Black patients and bridge the racial disparity gap in AML treatment,” the researchers concluded.
In developing countries, especially in Africa, starting patients on high-dose cytarabine, daunorubicin, and etoposide can lead to better results “without increasing much of the economic burden” since this treatment is the cheapest, Dr. Lamba said. “At the same time, if the patients have high ACS10 score, you can reduce their economic burden by giving them standard dose” cytarabine, daunorubicin, and etoposide and achieve similar results.
No study funding was reported. Dr. Lamba reported no relevant financial relationships, and three other authors reported various disclosures. Disclosures for Dr. Dunbar were unavailable..
A version of this article appeared on Medscape.com.
.
The score, dubbed ACS10 and initially highlighted in a 2022 report, predicts how well patients will respond to cytarabine based on their genetic make-up, and has the potential to personalize treatment for Black pediatric patients, a group that often has worse outcomes than White patients.
In the current study, presented at the annual meeting of the American Society of Hematology (ASH) , Black patients with low ACS10 scores had significantly worse outcomes compared with those with high scores when initially treated with low-dose cytarabine, daunorubicin, and etoposide.
The difference in outcomes disappeared, however, for patients who received high-dose cytarabine, daunorubicin, and etoposide or clofarabine and cytarabine.
The genetic traits revealed by the test likely help explain why Black patients with AML typically fare worse on certain regimens, Cynthia E. Dunbar, MD, chief of the Translational Stem Cell Biology Branch at the National Heart, Lung, and Blood Institute, commented in an ASH press preview briefing.
This study also suggests that clinicians should perform testing for genetic variants and biomarkers that impact outcomes “instead of assuming that a certain dose should be given simply based on perceived or reported race or ethnicity,” said Dr. Dunbar, also secretary of ASH.
The ACS10 test, derived from a combination of 10 single nucleotide polymorphisms, is not yet available, but one could be developed to help guide treatment decisions for clinicians, especially those in developing countries where AML treatment can be very expensive, said study lead author Jatinder Lamba, PhD, MSc, of the University of Florida College of Pharmacy, Gainesville, at an ASH press briefing on Thursday.
Prior research shows that Black pediatric patients with AML often have worse outcomes than White patients. A recent study , for instance, found Black patients with AML, especially those aged 18 to 29 years, had a higher early death rate compared with White patients (16% vs 3%) and significantly lower 5-year overall survival rates (22% vs 51%). The authors of this study suggested that genetic differences between young Black and White patients could help explain the disparity.
In the new analysis, Dr. Lamba and colleagues explored how outcomes by race and cytarabine pharmacogenomics varied in pediatric patients with AML.
The study included 86 Black patients and 359 White patients with newly diagnosed AML treated on two multi-institutional clinical trials. The patients received one of three initial treatments that included cytarabine: high-dose or low-dose cytarabine, daunorubicin, and etoposide, or clofarabine and cytarabine.
Most Black patients in the analysis (73%) had low ACS10 scores compared with 30% of White patients.
Unlike other recent reports, this study found that Black and White patients had similar complete remission rates following two courses of induction therapy (92.6% vs 95%) as well as similar rates of minimal residual disease negativity after one course (55.8% vs 55.4%).
Event-free survival (EFS) and overall survival rates were also similar, with 5-year EFS estimates at 58.3% for Black patients and 58.2% for White patients and overall survival rates at 63.8% vs 69.4%, respectively (P = .24).
However, when separating outcomes by ACS10 scores, Black patients with low scores had significantly worse EFS following low-dose cytarabine, daunorubicin, and etoposide compared with those with high ACS10 scores. And when these patients received high-dose cytarabine, daunorubicin, and etoposide or clofarabine and cytarabine induction therapy instead, the differences went away.
Overall, Black patients demonstrated significantly better EFS following treatment with clofarabine and cytarabine compared with the low-dose cytarabine triple therapy (hazard ratio, 0.17; P = .01). After adjusting for cofounders, clofarabine and cytarabine induction was the best treatment for Black patients with low ACS10 scores (HR for EFS, 0.2).
“Our results suggest that pharmacogenomics differences between Black and White patients should be considered when tailoring induction regimens to improve outcomes of Black patients and bridge the racial disparity gap in AML treatment,” the researchers concluded.
In developing countries, especially in Africa, starting patients on high-dose cytarabine, daunorubicin, and etoposide can lead to better results “without increasing much of the economic burden” since this treatment is the cheapest, Dr. Lamba said. “At the same time, if the patients have high ACS10 score, you can reduce their economic burden by giving them standard dose” cytarabine, daunorubicin, and etoposide and achieve similar results.
No study funding was reported. Dr. Lamba reported no relevant financial relationships, and three other authors reported various disclosures. Disclosures for Dr. Dunbar were unavailable..
A version of this article appeared on Medscape.com.
.
The score, dubbed ACS10 and initially highlighted in a 2022 report, predicts how well patients will respond to cytarabine based on their genetic make-up, and has the potential to personalize treatment for Black pediatric patients, a group that often has worse outcomes than White patients.
In the current study, presented at the annual meeting of the American Society of Hematology (ASH) , Black patients with low ACS10 scores had significantly worse outcomes compared with those with high scores when initially treated with low-dose cytarabine, daunorubicin, and etoposide.
The difference in outcomes disappeared, however, for patients who received high-dose cytarabine, daunorubicin, and etoposide or clofarabine and cytarabine.
The genetic traits revealed by the test likely help explain why Black patients with AML typically fare worse on certain regimens, Cynthia E. Dunbar, MD, chief of the Translational Stem Cell Biology Branch at the National Heart, Lung, and Blood Institute, commented in an ASH press preview briefing.
This study also suggests that clinicians should perform testing for genetic variants and biomarkers that impact outcomes “instead of assuming that a certain dose should be given simply based on perceived or reported race or ethnicity,” said Dr. Dunbar, also secretary of ASH.
The ACS10 test, derived from a combination of 10 single nucleotide polymorphisms, is not yet available, but one could be developed to help guide treatment decisions for clinicians, especially those in developing countries where AML treatment can be very expensive, said study lead author Jatinder Lamba, PhD, MSc, of the University of Florida College of Pharmacy, Gainesville, at an ASH press briefing on Thursday.
Prior research shows that Black pediatric patients with AML often have worse outcomes than White patients. A recent study , for instance, found Black patients with AML, especially those aged 18 to 29 years, had a higher early death rate compared with White patients (16% vs 3%) and significantly lower 5-year overall survival rates (22% vs 51%). The authors of this study suggested that genetic differences between young Black and White patients could help explain the disparity.
In the new analysis, Dr. Lamba and colleagues explored how outcomes by race and cytarabine pharmacogenomics varied in pediatric patients with AML.
The study included 86 Black patients and 359 White patients with newly diagnosed AML treated on two multi-institutional clinical trials. The patients received one of three initial treatments that included cytarabine: high-dose or low-dose cytarabine, daunorubicin, and etoposide, or clofarabine and cytarabine.
Most Black patients in the analysis (73%) had low ACS10 scores compared with 30% of White patients.
Unlike other recent reports, this study found that Black and White patients had similar complete remission rates following two courses of induction therapy (92.6% vs 95%) as well as similar rates of minimal residual disease negativity after one course (55.8% vs 55.4%).
Event-free survival (EFS) and overall survival rates were also similar, with 5-year EFS estimates at 58.3% for Black patients and 58.2% for White patients and overall survival rates at 63.8% vs 69.4%, respectively (P = .24).
However, when separating outcomes by ACS10 scores, Black patients with low scores had significantly worse EFS following low-dose cytarabine, daunorubicin, and etoposide compared with those with high ACS10 scores. And when these patients received high-dose cytarabine, daunorubicin, and etoposide or clofarabine and cytarabine induction therapy instead, the differences went away.
Overall, Black patients demonstrated significantly better EFS following treatment with clofarabine and cytarabine compared with the low-dose cytarabine triple therapy (hazard ratio, 0.17; P = .01). After adjusting for cofounders, clofarabine and cytarabine induction was the best treatment for Black patients with low ACS10 scores (HR for EFS, 0.2).
“Our results suggest that pharmacogenomics differences between Black and White patients should be considered when tailoring induction regimens to improve outcomes of Black patients and bridge the racial disparity gap in AML treatment,” the researchers concluded.
In developing countries, especially in Africa, starting patients on high-dose cytarabine, daunorubicin, and etoposide can lead to better results “without increasing much of the economic burden” since this treatment is the cheapest, Dr. Lamba said. “At the same time, if the patients have high ACS10 score, you can reduce their economic burden by giving them standard dose” cytarabine, daunorubicin, and etoposide and achieve similar results.
No study funding was reported. Dr. Lamba reported no relevant financial relationships, and three other authors reported various disclosures. Disclosures for Dr. Dunbar were unavailable..
A version of this article appeared on Medscape.com.
FROM ASH 2023
Which migraine medications are most effective?
TOPLINE:
new results from large, real-world analysis of self-reported patient data show.
METHODOLOGY:
- Researchers analyzed nearly 11 million migraine attack records extracted from Migraine Buddy, an e-diary smartphone app, over a 6-year period.
- They evaluated self-reported treatment effectiveness for 25 acute migraine medications among seven classes: acetaminophen, NSAIDs, triptans, combination analgesics, ergots, antiemetics, and opioids.
- A two-level nested multivariate logistic regression model adjusted for within-subject dependency and for concomitant medications taken within each analyzed migraine attack.
- The final analysis included nearly 5 million medication-outcome pairs from 3.1 million migraine attacks in 278,000 medication users.
TAKEAWAY:
- Using ibuprofen as the reference, triptans, ergots, and antiemetics were the top three medication classes with the highest effectiveness (mean odds ratios [OR] 4.80, 3.02, and 2.67, respectively).
- The next most effective medication classes were opioids (OR, 2.49), NSAIDs other than ibuprofen (OR, 1.94), combination analgesics acetaminophen/acetylsalicylic acid/caffeine (OR, 1.69), and others (OR, 1.49).
- Acetaminophen (OR, 0.83) was considered to be the least effective.
- The most effective individual medications were eletriptan (Relpax) (OR, 6.1); zolmitriptan (Zomig) (OR, 5.7); and sumatriptan (Imitrex) (OR, 5.2).
IN PRACTICE:
“Our findings that triptans, ergots, and antiemetics are the most effective classes of medications align with the guideline recommendations and offer generalizable insights to complement clinical practice,” the authors wrote.
SOURCE:
The study, with first author Chia-Chun Chiang, MD, Department of Neurology, Mayo Clinic, Rochester, Minnesota, was published online November 29 in Neurology.
LIMITATIONS:
The findings are based on subjective user-reported ratings of effectiveness and information on side effects, dosages, and formulations were not available. The newer migraine medication classes, gepants and ditans, were not included due to the relatively lower number of treated attacks. The regression model did not include age, gender, pain intensity, and other migraine-associated symptoms, which could potentially affect treatment effectiveness.
DISCLOSURES:
Funding for the study was provided by the Kanagawa University of Human Service research fund. A full list of author disclosures can be found with the original article.
A version of this article first appeared on Medscape.com.
TOPLINE:
new results from large, real-world analysis of self-reported patient data show.
METHODOLOGY:
- Researchers analyzed nearly 11 million migraine attack records extracted from Migraine Buddy, an e-diary smartphone app, over a 6-year period.
- They evaluated self-reported treatment effectiveness for 25 acute migraine medications among seven classes: acetaminophen, NSAIDs, triptans, combination analgesics, ergots, antiemetics, and opioids.
- A two-level nested multivariate logistic regression model adjusted for within-subject dependency and for concomitant medications taken within each analyzed migraine attack.
- The final analysis included nearly 5 million medication-outcome pairs from 3.1 million migraine attacks in 278,000 medication users.
TAKEAWAY:
- Using ibuprofen as the reference, triptans, ergots, and antiemetics were the top three medication classes with the highest effectiveness (mean odds ratios [OR] 4.80, 3.02, and 2.67, respectively).
- The next most effective medication classes were opioids (OR, 2.49), NSAIDs other than ibuprofen (OR, 1.94), combination analgesics acetaminophen/acetylsalicylic acid/caffeine (OR, 1.69), and others (OR, 1.49).
- Acetaminophen (OR, 0.83) was considered to be the least effective.
- The most effective individual medications were eletriptan (Relpax) (OR, 6.1); zolmitriptan (Zomig) (OR, 5.7); and sumatriptan (Imitrex) (OR, 5.2).
IN PRACTICE:
“Our findings that triptans, ergots, and antiemetics are the most effective classes of medications align with the guideline recommendations and offer generalizable insights to complement clinical practice,” the authors wrote.
SOURCE:
The study, with first author Chia-Chun Chiang, MD, Department of Neurology, Mayo Clinic, Rochester, Minnesota, was published online November 29 in Neurology.
LIMITATIONS:
The findings are based on subjective user-reported ratings of effectiveness and information on side effects, dosages, and formulations were not available. The newer migraine medication classes, gepants and ditans, were not included due to the relatively lower number of treated attacks. The regression model did not include age, gender, pain intensity, and other migraine-associated symptoms, which could potentially affect treatment effectiveness.
DISCLOSURES:
Funding for the study was provided by the Kanagawa University of Human Service research fund. A full list of author disclosures can be found with the original article.
A version of this article first appeared on Medscape.com.
TOPLINE:
new results from large, real-world analysis of self-reported patient data show.
METHODOLOGY:
- Researchers analyzed nearly 11 million migraine attack records extracted from Migraine Buddy, an e-diary smartphone app, over a 6-year period.
- They evaluated self-reported treatment effectiveness for 25 acute migraine medications among seven classes: acetaminophen, NSAIDs, triptans, combination analgesics, ergots, antiemetics, and opioids.
- A two-level nested multivariate logistic regression model adjusted for within-subject dependency and for concomitant medications taken within each analyzed migraine attack.
- The final analysis included nearly 5 million medication-outcome pairs from 3.1 million migraine attacks in 278,000 medication users.
TAKEAWAY:
- Using ibuprofen as the reference, triptans, ergots, and antiemetics were the top three medication classes with the highest effectiveness (mean odds ratios [OR] 4.80, 3.02, and 2.67, respectively).
- The next most effective medication classes were opioids (OR, 2.49), NSAIDs other than ibuprofen (OR, 1.94), combination analgesics acetaminophen/acetylsalicylic acid/caffeine (OR, 1.69), and others (OR, 1.49).
- Acetaminophen (OR, 0.83) was considered to be the least effective.
- The most effective individual medications were eletriptan (Relpax) (OR, 6.1); zolmitriptan (Zomig) (OR, 5.7); and sumatriptan (Imitrex) (OR, 5.2).
IN PRACTICE:
“Our findings that triptans, ergots, and antiemetics are the most effective classes of medications align with the guideline recommendations and offer generalizable insights to complement clinical practice,” the authors wrote.
SOURCE:
The study, with first author Chia-Chun Chiang, MD, Department of Neurology, Mayo Clinic, Rochester, Minnesota, was published online November 29 in Neurology.
LIMITATIONS:
The findings are based on subjective user-reported ratings of effectiveness and information on side effects, dosages, and formulations were not available. The newer migraine medication classes, gepants and ditans, were not included due to the relatively lower number of treated attacks. The regression model did not include age, gender, pain intensity, and other migraine-associated symptoms, which could potentially affect treatment effectiveness.
DISCLOSURES:
Funding for the study was provided by the Kanagawa University of Human Service research fund. A full list of author disclosures can be found with the original article.
A version of this article first appeared on Medscape.com.
Tape strips detect hidradenitis suppurativa biomarkers, novel study shows
, results from a novel study showed.
“Tape strips can provide important clues to when and which drugs to use in HS in patients with both early and late disease, which can change clinical practice,” corresponding study author Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York City, said in an interview. “It is noninvasive and nonscarring,” she added.
Tape stripping has been validated in atopic dermatitis, psoriasis, and other dermatologic conditions in recent years. For the current study, which was published online in the Journal of the American Academy of Dermatology, and is believed to be the first of its kind, Dr. Guttman-Yassky and colleagues performed RNA sequencing from large D-Squame tape strips collected from lesional and nonlesional skin of 22 patients with HS and from 21 age- and sex-matched healthy controls. They correlated the expression of skin biomarkers between tape strips and a previously published gene-signature of HS biopsies. The mean age of patients with HS was 43 years, while the mean age of healthy controls was 35. The average International Hidradenitis Suppurativa Severity Score System (IHS4) score of the HS cohort was 36.
Consistent with published studies, the researchers found that tape strips identified an overall higher inflammatory burden in HS. Specifically, they observed an upregulation of known cytokines within the following pathways: Th1 (such as IFNG, CXCL9/10/11, and CCR5); Th17 (such as interleukin [IL]-17A/F, IL12B, IL23A, CAMP, and CCL20); Th2 (such as IL4R, IL13/IL31/IL10, CCR4, CCL7/CCL13/CCL24, TNFSF4/OX40L, and TNFRSF4/OX40); and Th22 (such as IL22 and IL32).
The researchers also found that the expression of Th17 and tumor necrosis factor (TNF)–alpha pathways were highly correlated between tape strips and biopsies and that HS clinical severity was significantly associated with expression of biomarkers, such as TNF-alpha, IL17A/F, OX40, JAK1-3, and IL4R in HS lesional and/or nonlesional skin.
“It was quite unexpected that we are able to identify, using a minimally invasive approach that samples only the upper layers of the epidermis, products and processes that are considered to be deeper-situated, such as IL-17, and other immune markers,” Dr. Guttman-Yassky said in the interview. “We were also surprised to see how well the tape-stripped–derived skin molecular profile correlated with that of biopsies, as well as how well it correlated with the clinical disease severity of HS.”
Also surprising, she added, was that the biomarkers in nonlesional tape-stripped skin, such as IL-17 and TNF alpha, “show high correlations with disease severity and provide clues to early disease.”
If using tape strips in HS is validated in larger cohort studies, the potential cost implications of using this approach in practice remain unclear, Dr. Guttman-Yassky said. “It is currently not cheap, but we are hoping that one day, we can provide a means to diagnose the disease and treat it early, and appropriately, utilizing this approach,” she commented. “We are excited about the applicability of this study to the early treatment and longitudinal follow up of HS with drugs that are targeting specific immune molecules and pathways,” she said, adding that it will also be useful for helping determine which drug should be used for which patient.
She and her co-authors acknowledged certain limitations of the study, including its small sample size and the fact that tape stripping is limited to the epidermis.
Asked to comment on the study, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, said the findings “have important potential implications for our ability to one day personalize treatments for a patient with early HS in a minimally invasive way.”
As the study authors point out, she added, “tape strips only allow sampling of the epidermis, which is limiting in a disease like HS where much of the disruption is in the dermis with deep nodules and dermal tunnels. However, our overall goal should be to catch patients in the early stages of their disease before the occurrence of irreversible tissue damage such as dermal tunnels. Thus, the ongoing campaign for early diagnosis and early intervention by various stakeholders in the field of HS can help mitigate the impact of this inherent limitation of tape strips. It will be exciting to see larger studies that investigate tape strip results in relation to clinical phenotypes, disease progression, and therapeutic responses.”
The study was funded by an International Dermatology Outcome Measures Hidradenitis Suppurativa Grant. Dr. Guttman-Yassky disclosed that she has been a consultant to, an adviser for, and has received research grants from many pharmaceutical companies. Of the remaining authors, 2 also had multiple disclosures and 11 had no disclosures. Dr. Hsiao disclosed that she is a member of the board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
, results from a novel study showed.
“Tape strips can provide important clues to when and which drugs to use in HS in patients with both early and late disease, which can change clinical practice,” corresponding study author Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York City, said in an interview. “It is noninvasive and nonscarring,” she added.
Tape stripping has been validated in atopic dermatitis, psoriasis, and other dermatologic conditions in recent years. For the current study, which was published online in the Journal of the American Academy of Dermatology, and is believed to be the first of its kind, Dr. Guttman-Yassky and colleagues performed RNA sequencing from large D-Squame tape strips collected from lesional and nonlesional skin of 22 patients with HS and from 21 age- and sex-matched healthy controls. They correlated the expression of skin biomarkers between tape strips and a previously published gene-signature of HS biopsies. The mean age of patients with HS was 43 years, while the mean age of healthy controls was 35. The average International Hidradenitis Suppurativa Severity Score System (IHS4) score of the HS cohort was 36.
Consistent with published studies, the researchers found that tape strips identified an overall higher inflammatory burden in HS. Specifically, they observed an upregulation of known cytokines within the following pathways: Th1 (such as IFNG, CXCL9/10/11, and CCR5); Th17 (such as interleukin [IL]-17A/F, IL12B, IL23A, CAMP, and CCL20); Th2 (such as IL4R, IL13/IL31/IL10, CCR4, CCL7/CCL13/CCL24, TNFSF4/OX40L, and TNFRSF4/OX40); and Th22 (such as IL22 and IL32).
The researchers also found that the expression of Th17 and tumor necrosis factor (TNF)–alpha pathways were highly correlated between tape strips and biopsies and that HS clinical severity was significantly associated with expression of biomarkers, such as TNF-alpha, IL17A/F, OX40, JAK1-3, and IL4R in HS lesional and/or nonlesional skin.
“It was quite unexpected that we are able to identify, using a minimally invasive approach that samples only the upper layers of the epidermis, products and processes that are considered to be deeper-situated, such as IL-17, and other immune markers,” Dr. Guttman-Yassky said in the interview. “We were also surprised to see how well the tape-stripped–derived skin molecular profile correlated with that of biopsies, as well as how well it correlated with the clinical disease severity of HS.”
Also surprising, she added, was that the biomarkers in nonlesional tape-stripped skin, such as IL-17 and TNF alpha, “show high correlations with disease severity and provide clues to early disease.”
If using tape strips in HS is validated in larger cohort studies, the potential cost implications of using this approach in practice remain unclear, Dr. Guttman-Yassky said. “It is currently not cheap, but we are hoping that one day, we can provide a means to diagnose the disease and treat it early, and appropriately, utilizing this approach,” she commented. “We are excited about the applicability of this study to the early treatment and longitudinal follow up of HS with drugs that are targeting specific immune molecules and pathways,” she said, adding that it will also be useful for helping determine which drug should be used for which patient.
She and her co-authors acknowledged certain limitations of the study, including its small sample size and the fact that tape stripping is limited to the epidermis.
Asked to comment on the study, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, said the findings “have important potential implications for our ability to one day personalize treatments for a patient with early HS in a minimally invasive way.”
As the study authors point out, she added, “tape strips only allow sampling of the epidermis, which is limiting in a disease like HS where much of the disruption is in the dermis with deep nodules and dermal tunnels. However, our overall goal should be to catch patients in the early stages of their disease before the occurrence of irreversible tissue damage such as dermal tunnels. Thus, the ongoing campaign for early diagnosis and early intervention by various stakeholders in the field of HS can help mitigate the impact of this inherent limitation of tape strips. It will be exciting to see larger studies that investigate tape strip results in relation to clinical phenotypes, disease progression, and therapeutic responses.”
The study was funded by an International Dermatology Outcome Measures Hidradenitis Suppurativa Grant. Dr. Guttman-Yassky disclosed that she has been a consultant to, an adviser for, and has received research grants from many pharmaceutical companies. Of the remaining authors, 2 also had multiple disclosures and 11 had no disclosures. Dr. Hsiao disclosed that she is a member of the board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
, results from a novel study showed.
“Tape strips can provide important clues to when and which drugs to use in HS in patients with both early and late disease, which can change clinical practice,” corresponding study author Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York City, said in an interview. “It is noninvasive and nonscarring,” she added.
Tape stripping has been validated in atopic dermatitis, psoriasis, and other dermatologic conditions in recent years. For the current study, which was published online in the Journal of the American Academy of Dermatology, and is believed to be the first of its kind, Dr. Guttman-Yassky and colleagues performed RNA sequencing from large D-Squame tape strips collected from lesional and nonlesional skin of 22 patients with HS and from 21 age- and sex-matched healthy controls. They correlated the expression of skin biomarkers between tape strips and a previously published gene-signature of HS biopsies. The mean age of patients with HS was 43 years, while the mean age of healthy controls was 35. The average International Hidradenitis Suppurativa Severity Score System (IHS4) score of the HS cohort was 36.
Consistent with published studies, the researchers found that tape strips identified an overall higher inflammatory burden in HS. Specifically, they observed an upregulation of known cytokines within the following pathways: Th1 (such as IFNG, CXCL9/10/11, and CCR5); Th17 (such as interleukin [IL]-17A/F, IL12B, IL23A, CAMP, and CCL20); Th2 (such as IL4R, IL13/IL31/IL10, CCR4, CCL7/CCL13/CCL24, TNFSF4/OX40L, and TNFRSF4/OX40); and Th22 (such as IL22 and IL32).
The researchers also found that the expression of Th17 and tumor necrosis factor (TNF)–alpha pathways were highly correlated between tape strips and biopsies and that HS clinical severity was significantly associated with expression of biomarkers, such as TNF-alpha, IL17A/F, OX40, JAK1-3, and IL4R in HS lesional and/or nonlesional skin.
“It was quite unexpected that we are able to identify, using a minimally invasive approach that samples only the upper layers of the epidermis, products and processes that are considered to be deeper-situated, such as IL-17, and other immune markers,” Dr. Guttman-Yassky said in the interview. “We were also surprised to see how well the tape-stripped–derived skin molecular profile correlated with that of biopsies, as well as how well it correlated with the clinical disease severity of HS.”
Also surprising, she added, was that the biomarkers in nonlesional tape-stripped skin, such as IL-17 and TNF alpha, “show high correlations with disease severity and provide clues to early disease.”
If using tape strips in HS is validated in larger cohort studies, the potential cost implications of using this approach in practice remain unclear, Dr. Guttman-Yassky said. “It is currently not cheap, but we are hoping that one day, we can provide a means to diagnose the disease and treat it early, and appropriately, utilizing this approach,” she commented. “We are excited about the applicability of this study to the early treatment and longitudinal follow up of HS with drugs that are targeting specific immune molecules and pathways,” she said, adding that it will also be useful for helping determine which drug should be used for which patient.
She and her co-authors acknowledged certain limitations of the study, including its small sample size and the fact that tape stripping is limited to the epidermis.
Asked to comment on the study, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, said the findings “have important potential implications for our ability to one day personalize treatments for a patient with early HS in a minimally invasive way.”
As the study authors point out, she added, “tape strips only allow sampling of the epidermis, which is limiting in a disease like HS where much of the disruption is in the dermis with deep nodules and dermal tunnels. However, our overall goal should be to catch patients in the early stages of their disease before the occurrence of irreversible tissue damage such as dermal tunnels. Thus, the ongoing campaign for early diagnosis and early intervention by various stakeholders in the field of HS can help mitigate the impact of this inherent limitation of tape strips. It will be exciting to see larger studies that investigate tape strip results in relation to clinical phenotypes, disease progression, and therapeutic responses.”
The study was funded by an International Dermatology Outcome Measures Hidradenitis Suppurativa Grant. Dr. Guttman-Yassky disclosed that she has been a consultant to, an adviser for, and has received research grants from many pharmaceutical companies. Of the remaining authors, 2 also had multiple disclosures and 11 had no disclosures. Dr. Hsiao disclosed that she is a member of the board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
New COVID variant JN.1 could disrupt holiday plans
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
FDA approves first 2 gene-editing therapies for sickle cell
These “milestone treatments” mark the first cell-based gene therapies for this debilitating and potentially life-threatening blood disorder that affects about 100,000 people in the United States.
The two therapies are exagamglogene autotemcel, or exa-cel (Casgevy; Vertex Pharmaceuticals and Crispr Therapeutics), and lovotibeglogene autotemcel, or lovo-cel (Lyfgenia; bluebird bio).
“The approval of the first gene therapies for [sickle cell disease] represents a tremendous step forward for the [sickle cell] community, which has been historically overlooked and underfunded,” said Robert A. Brodsky, MD, of Johns Hopkins University School of Medicine, in a statement from the American Society of Hematology, following the approval.
“We are excited to advance the field, especially for individuals whose lives have been severely disrupted by the disease, by approving two cell-based gene therapies today,” Nicole Verdun, MD, of the FDA’s Center for Biologics Evaluation and Research, added in an agency press release.
Sickle cell disease involves a mutation in hemoglobin, a protein in red blood cells that provides oxygen to tissues. The mutation leads red blood cells to develop a crescent or sickle shape, which can restrict blood flow and cause severe pain and organ damage, known as vaso-occlusive events or crises.
Treatment options prior to these approvals primarily included red blood transfusions and hydroxyurea alongside pain management. The only potential curative option has been allogeneic hematopoietic stem cell transplantation, but that comes with significant risks and most patients don’t have an appropriate donor.
Exa-cel
Exa-cel uses CRISPR gene-editing technology. Before the infusion, patients undergo myeloablative conditioning, which removes cells from the bone marrow. These cells are genetically modified to produce fetal hemoglobin. Patients then receive an infusion of the edited cells, which can help restore normal hemoglobin production.
The FDA approval was based on data from the pivotal CLIMB SCD-121 trial. In an October advisory committee meeting, the FDA highlighted trial data demonstrating that 29 of 31 patients reached the trial’s primary endpoint: freedom from severe vaso-occlusive crises over a 12-month period. In addition, 28 of these patients remained free of vaso-occlusive crises for almost 2 years.
The committee noted that one of the 31 patients died about 9 months after receiving an exa-cel infusion.
The cell-based gene therapy also increased both fetal and total hemoglobin, with total hemoglobin levels increasing to > 11 g/dL by month 3 and remaining at that level afterward. No patients experienced graft failure or rejection.
The most common side effects included low platelets and white blood cell counts, mouth sores, nausea, musculoskeletal pain, vomiting, and febrile neutropenia.
Exa-cel could “provide a one-time functional cure” for patients with severe sickle cell disease, according to Franco Locatelli, MD, of Sapienza University of Rome, who presented initial findings last year.
While the current approval is for patients with infusion-dependent sickle cell disease, exa-cel is also being evaluated in patients with another blood disorder, beta-thalassemia.
Lovo-cel
Lovo-cel, a cell-based gene therapy, uses a different technology — a lentiviral vector, or gene delivery vehicle — that can also genetically modify a patient’s blood stem cells.
Like exa-cel, lovo-cel is a one-time, single-dose infusion that contains the patient’s modified cells. Before the infusion, patients undergo myeloablative conditioning. The patient’s stem cells are then genetically modified to allow them to produce the most common form of hemoglobin, HbA
This approval was based on data from a single-arm, 24-month study in patients aged 12-50 years who had sickle cell disease and a history of vaso-occlusive events.
Overall, 88% of patients (28 of 32) achieved complete resolution of vaso-occlusive events 6-18 months after the infusion.
The most common side effects included stomatitis; febrile neutropenia; and low platelet, white blood cell, and red blood cell counts.
The FDA noted that hematologic cancer has occurred in patients treated with lovo-cel, and the label includes a black-box warning about the risk.
Dr. Brodsky noted, however, that “while these new gene therapies are potentially life-changing for individuals living with [sickle cell disease], they must be accessible to be effective.”
Access is a potential concern. Exa-cel and lovo-cel could cost about $2 million.
A version of this article appeared on Medscape.com.
These “milestone treatments” mark the first cell-based gene therapies for this debilitating and potentially life-threatening blood disorder that affects about 100,000 people in the United States.
The two therapies are exagamglogene autotemcel, or exa-cel (Casgevy; Vertex Pharmaceuticals and Crispr Therapeutics), and lovotibeglogene autotemcel, or lovo-cel (Lyfgenia; bluebird bio).
“The approval of the first gene therapies for [sickle cell disease] represents a tremendous step forward for the [sickle cell] community, which has been historically overlooked and underfunded,” said Robert A. Brodsky, MD, of Johns Hopkins University School of Medicine, in a statement from the American Society of Hematology, following the approval.
“We are excited to advance the field, especially for individuals whose lives have been severely disrupted by the disease, by approving two cell-based gene therapies today,” Nicole Verdun, MD, of the FDA’s Center for Biologics Evaluation and Research, added in an agency press release.
Sickle cell disease involves a mutation in hemoglobin, a protein in red blood cells that provides oxygen to tissues. The mutation leads red blood cells to develop a crescent or sickle shape, which can restrict blood flow and cause severe pain and organ damage, known as vaso-occlusive events or crises.
Treatment options prior to these approvals primarily included red blood transfusions and hydroxyurea alongside pain management. The only potential curative option has been allogeneic hematopoietic stem cell transplantation, but that comes with significant risks and most patients don’t have an appropriate donor.
Exa-cel
Exa-cel uses CRISPR gene-editing technology. Before the infusion, patients undergo myeloablative conditioning, which removes cells from the bone marrow. These cells are genetically modified to produce fetal hemoglobin. Patients then receive an infusion of the edited cells, which can help restore normal hemoglobin production.
The FDA approval was based on data from the pivotal CLIMB SCD-121 trial. In an October advisory committee meeting, the FDA highlighted trial data demonstrating that 29 of 31 patients reached the trial’s primary endpoint: freedom from severe vaso-occlusive crises over a 12-month period. In addition, 28 of these patients remained free of vaso-occlusive crises for almost 2 years.
The committee noted that one of the 31 patients died about 9 months after receiving an exa-cel infusion.
The cell-based gene therapy also increased both fetal and total hemoglobin, with total hemoglobin levels increasing to > 11 g/dL by month 3 and remaining at that level afterward. No patients experienced graft failure or rejection.
The most common side effects included low platelets and white blood cell counts, mouth sores, nausea, musculoskeletal pain, vomiting, and febrile neutropenia.
Exa-cel could “provide a one-time functional cure” for patients with severe sickle cell disease, according to Franco Locatelli, MD, of Sapienza University of Rome, who presented initial findings last year.
While the current approval is for patients with infusion-dependent sickle cell disease, exa-cel is also being evaluated in patients with another blood disorder, beta-thalassemia.
Lovo-cel
Lovo-cel, a cell-based gene therapy, uses a different technology — a lentiviral vector, or gene delivery vehicle — that can also genetically modify a patient’s blood stem cells.
Like exa-cel, lovo-cel is a one-time, single-dose infusion that contains the patient’s modified cells. Before the infusion, patients undergo myeloablative conditioning. The patient’s stem cells are then genetically modified to allow them to produce the most common form of hemoglobin, HbA
This approval was based on data from a single-arm, 24-month study in patients aged 12-50 years who had sickle cell disease and a history of vaso-occlusive events.
Overall, 88% of patients (28 of 32) achieved complete resolution of vaso-occlusive events 6-18 months after the infusion.
The most common side effects included stomatitis; febrile neutropenia; and low platelet, white blood cell, and red blood cell counts.
The FDA noted that hematologic cancer has occurred in patients treated with lovo-cel, and the label includes a black-box warning about the risk.
Dr. Brodsky noted, however, that “while these new gene therapies are potentially life-changing for individuals living with [sickle cell disease], they must be accessible to be effective.”
Access is a potential concern. Exa-cel and lovo-cel could cost about $2 million.
A version of this article appeared on Medscape.com.
These “milestone treatments” mark the first cell-based gene therapies for this debilitating and potentially life-threatening blood disorder that affects about 100,000 people in the United States.
The two therapies are exagamglogene autotemcel, or exa-cel (Casgevy; Vertex Pharmaceuticals and Crispr Therapeutics), and lovotibeglogene autotemcel, or lovo-cel (Lyfgenia; bluebird bio).
“The approval of the first gene therapies for [sickle cell disease] represents a tremendous step forward for the [sickle cell] community, which has been historically overlooked and underfunded,” said Robert A. Brodsky, MD, of Johns Hopkins University School of Medicine, in a statement from the American Society of Hematology, following the approval.
“We are excited to advance the field, especially for individuals whose lives have been severely disrupted by the disease, by approving two cell-based gene therapies today,” Nicole Verdun, MD, of the FDA’s Center for Biologics Evaluation and Research, added in an agency press release.
Sickle cell disease involves a mutation in hemoglobin, a protein in red blood cells that provides oxygen to tissues. The mutation leads red blood cells to develop a crescent or sickle shape, which can restrict blood flow and cause severe pain and organ damage, known as vaso-occlusive events or crises.
Treatment options prior to these approvals primarily included red blood transfusions and hydroxyurea alongside pain management. The only potential curative option has been allogeneic hematopoietic stem cell transplantation, but that comes with significant risks and most patients don’t have an appropriate donor.
Exa-cel
Exa-cel uses CRISPR gene-editing technology. Before the infusion, patients undergo myeloablative conditioning, which removes cells from the bone marrow. These cells are genetically modified to produce fetal hemoglobin. Patients then receive an infusion of the edited cells, which can help restore normal hemoglobin production.
The FDA approval was based on data from the pivotal CLIMB SCD-121 trial. In an October advisory committee meeting, the FDA highlighted trial data demonstrating that 29 of 31 patients reached the trial’s primary endpoint: freedom from severe vaso-occlusive crises over a 12-month period. In addition, 28 of these patients remained free of vaso-occlusive crises for almost 2 years.
The committee noted that one of the 31 patients died about 9 months after receiving an exa-cel infusion.
The cell-based gene therapy also increased both fetal and total hemoglobin, with total hemoglobin levels increasing to > 11 g/dL by month 3 and remaining at that level afterward. No patients experienced graft failure or rejection.
The most common side effects included low platelets and white blood cell counts, mouth sores, nausea, musculoskeletal pain, vomiting, and febrile neutropenia.
Exa-cel could “provide a one-time functional cure” for patients with severe sickle cell disease, according to Franco Locatelli, MD, of Sapienza University of Rome, who presented initial findings last year.
While the current approval is for patients with infusion-dependent sickle cell disease, exa-cel is also being evaluated in patients with another blood disorder, beta-thalassemia.
Lovo-cel
Lovo-cel, a cell-based gene therapy, uses a different technology — a lentiviral vector, or gene delivery vehicle — that can also genetically modify a patient’s blood stem cells.
Like exa-cel, lovo-cel is a one-time, single-dose infusion that contains the patient’s modified cells. Before the infusion, patients undergo myeloablative conditioning. The patient’s stem cells are then genetically modified to allow them to produce the most common form of hemoglobin, HbA
This approval was based on data from a single-arm, 24-month study in patients aged 12-50 years who had sickle cell disease and a history of vaso-occlusive events.
Overall, 88% of patients (28 of 32) achieved complete resolution of vaso-occlusive events 6-18 months after the infusion.
The most common side effects included stomatitis; febrile neutropenia; and low platelet, white blood cell, and red blood cell counts.
The FDA noted that hematologic cancer has occurred in patients treated with lovo-cel, and the label includes a black-box warning about the risk.
Dr. Brodsky noted, however, that “while these new gene therapies are potentially life-changing for individuals living with [sickle cell disease], they must be accessible to be effective.”
Access is a potential concern. Exa-cel and lovo-cel could cost about $2 million.
A version of this article appeared on Medscape.com.