User login
Multiple atopic dermatitis therapies completed or close to completing phase 3 studies
Major , Jonathan I. Silverberg, MD, PhD, said during a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled due to the COVID-19 pandemic.
“In the next 2-3 years, we may have nine new treatments approved for atopic dermatitis,” said Dr. Silverberg, director of clinical research and contact dermatitis at the University.
All nine medications he discussed are either in ongoing pivotal phase 3 randomized controlled trials or have completed their phase 3 developmental programs. “This is not theoretical; these are things you’re going to be using in your toolbox imminently,” he stressed.
Oral JAK inhibitors
The Janus kinase (JAK) pathway is the intracellular signaling mediator that interacts with extracellular inflammatory cytokines, including interleukin-4, -13, and -31, which are familiar to dermatologists because they’re targeted by potent biologic monoclonal antibody therapies. For example, IL-4 goes through JAK1 and 3, while IL-31 signals through JAK1 and 2.
“You really need to know the key JAK and STAT pathways involved in atopic dermatitis because it will help you determine the selectivity of the agents you’re going to be using,” the dermatologist advised.
Three oral, once-daily JAK inhibitors – abrocitinib, upadacitinib, and baricitinib – are in an advanced stage of development.
“Upadacitinib and abrocitinib may be the two most potent options coming to market soon for us to be thinking about,” Dr. Silverberg said.
Abrocitinib: Three positive phase 3 studies featuring this selective JAK1 inhibitor have been completed in adults with moderate to severe atopic dermatitis (AD). The most recent, JADE COMPARE, featured a head-to-head randomized comparison of abrocitinib and the injectable IL-4/IL-13 inhibitor dupilumab. The results of this 837-patient study haven’t yet been formally presented at a conference because of the COVID-19 pandemic. However, Pfizer recently announced that abrocitinib at 200 mg/day achieved significantly greater improvements than dupilumab (Dupixent) in the coprimary endpoints of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (that is, clear or almost clear) and disease extent based upon 75% reduction from baseline in Eczema Area and Severity Index (EASI 75) at 12 weeks. The same was true at 16 weeks.
Also, a significantly larger proportion of abrocitinib-treated patients achieved at least a 4-point reduction in itch severity as measured using the Peak Pruritus Numerical Rating Scale at week 2. The company plans to file for regulatory approval later this year.
The JADE COMPARE data are exciting because of a pressing unmet need for treatment options that are even more powerful than dupilumab, Dr. Silverberg said.
Upadacitinib: This is selective JAK1 inhibitor is not as far along in the developmental pipeline as abrocitinib, but the efficacy appears to be comparable. In a phase 2 study of 126 adults with moderate to severe AD, upadacitinib at the top dose of 30 mg/day achieved efficacy results Dr. Silverberg deemed “quite extraordinary,” with a rate of IGA score of 0 or 1 of 50% at 16 weeks and an EASI 75 response rate of 69%. Those findings numerically eclipsed results seen in an earlier phase 3 pivotal trial for dupilumab, in which the IGA 0/1 rate was 37% and EASI 75 was 48%, albeit with the caveat that cross-trial comparisons must be taken with a large grain of salt.
Baricitinib: Multiple phase 3 studies of this JAK 1/2 inhibitor have reported positive results. At the top dose of 4 mg/day, baricitinib appears to be less effective than dupilumab in its earlier pivotal trials.
“This may be a good oral option for our patients. It could be similar to the Otezla [apremilast] story in psoriasis: It’s perhaps not as effective as a lot of the biologics, but patients often prefer an oral option,” Dr. Silverberg said.
Of note, in one large, placebo-controlled, phase 3 study of baricitinib on top of background low- or medium-potency topical steroids, the IGA 0/1 rate at 16 weeks with placebo plus topical steroids was a modest 14.7%, which underscores that this long-time workhorse topical therapy is objectively less effective than most physicians think. In contrast, the IGA 0/1 rate with baricitinib at 4 mg/day plus topical steroids was a more respectable 30.6%.
All three oral JAK inhibitors have rapid onset of efficacy, a key advantage over the biologic agents.
“The issue you have to keep in mind is safety. The safety in the atopic dermatitis population was overall quite good for all three drugs. However, safety concerns have come up with JAK inhibitors in rheumatoid arthritis. I think that’s the part we watch the most in this. The efficacy has become clear. Now the question is where does the safety take us,” he said.
Novel injectable biologics
Nemolizumab: This humanized monoclonal antibody inhibits IL-31 receptor alpha. Mounting evidence implicates IL-31 as both a proinflammatory and immunomodulatory cytokine linking the immune and neural systems.
Early on, most researchers pigeonholed IL-31 as being a key player only in the itch factor in AD. Not so. Indeed, Dr. Silverberg was the lead investigator in a recent phase 2b study of nemolizumab that demonstrated the biologic is also effective at rapidly clearing AD lesions. The study, which evaluated three different doses in 226 adults with moderate to severe AD and severe pruritus who were on background topical corticosteroids, showed that nemolizumab at 30 mg every 4 weeks trounced placebo in terms of itch reduction: The 69% drop from baseline in Peak Pruritus Numeric Rating Scale at week 16 was twice that in controls, with a significant difference apparent even at week 1.
But in addition, the 33% IGA 0/1 rate at the same time point bested the 12% rate in controls. The EASI 75 response rate was significantly higher as well – 49% versus 19% – as was the EASI 90 response of 33%, compared with 9% in controls. Moreover, nemolizumab-treated patients used close to 40% less topical steroids during the study (J Allergy Clin Immunol. 2020 Jan;145[1]:173-82).
“This is something that’s fascinating. The study gets into the idea that a subset of atopic dermatitis patients have the itch that rashes, and perhaps if you break the itch/scratch cycle you can modify the lesions. Or the effect may even be due to the direct anti-inflammatory action of IL-31 blockade,” Dr. Silverberg observed.
It appeared that a plateau hadn’t been reached for some endpoints out at week 24, when the study ended. Japanese phase 3 studies have been completed, with what he called “great results,” and others are ongoing in the United States.
Tralokinumab: This fully human monoclonal antibody binds to IL-13, but unlike dupilumab, it doesn’t also inhibit IL-4. Tralokinumab met all primary and secondary endpoints in three pivotal phase 3 clinical trials, known as ECZTRA 1-3, that assessed it as treatment for moderate to severe AD in adults and showed an overall adverse event rate comparable with placebo. Leo Pharma, the Danish company developing the biologic, has announced it will file for marketing approval before the end of 2020. Phase 3 data would have been presented at the annual meeting of the American Academy of Dermatology in Denver, had it not been canceled. Dr. Silverberg said that, based upon phase 2 results, it appears tralokinumab may not be quite as effective as dupilumab in the overall AD population, but he predicted the newcomer will still play a useful role.
“The complexities of the immune system are such that some patients will respond better to one drug than another. I think we still have a lot to learn about who the patients are for these novel assets,” he said.
Lebrikizumab: This is another selective IL-13 inhibitor, but this one binds to IL-13 in a slightly different way than tralokinumab. The Food and Drug Administration granted it Fast Track status in December 2019. Twin placebo-controlled phase 3 studies of lebrikizumab as monotherapy for moderate to severe AD are ongoing, and another phase 3 trial of the biologic in combination with topical steroids is planned. Based upon the results of a phase 2b study, the highest dose studied – 250 mg every 2 weeks – appears to be at least as effective as dupilumab.
Nonsteroidal topical agents
These three late-stage topical creams – ruxolitinib, delgocitinib, and tapinarof – have previously received considerable coverage in Dermatology News. Ruxolitinib, a selective JAK1/2 inhibitor, has completed a positive phase 3 trial in adolescents and adults with mild to moderate AD. Delgocitinib, a pan-JAK1/2/3 and Tyrosine kinase 2 inhibitor, is already approved in an ointment formulation in Japan, and the cream formulation is in phase 2 studies in the United States and Europe. Tapinarof has a unique mechanism of action – it’s an aryl hydrocarbon receptor modulator – and is now in phase 3 in adolescents and adults with moderate to severe AD.
These three drugs appear to offer efficacy that’s comparable to or even better than medium-potency topical steroids, and without the notorious steroidal side effects that have caused widespread parental steroid-phobia. Potential applications for other inflammatory diseases, including vitiligo and psoriasis, are under study.
Dr. Silverberg reported receiving research grants from Galderma and GlaxoSmithKline and serving as a consultant to those pharmaceutical companies and more than a dozen others.
Major , Jonathan I. Silverberg, MD, PhD, said during a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled due to the COVID-19 pandemic.
“In the next 2-3 years, we may have nine new treatments approved for atopic dermatitis,” said Dr. Silverberg, director of clinical research and contact dermatitis at the University.
All nine medications he discussed are either in ongoing pivotal phase 3 randomized controlled trials or have completed their phase 3 developmental programs. “This is not theoretical; these are things you’re going to be using in your toolbox imminently,” he stressed.
Oral JAK inhibitors
The Janus kinase (JAK) pathway is the intracellular signaling mediator that interacts with extracellular inflammatory cytokines, including interleukin-4, -13, and -31, which are familiar to dermatologists because they’re targeted by potent biologic monoclonal antibody therapies. For example, IL-4 goes through JAK1 and 3, while IL-31 signals through JAK1 and 2.
“You really need to know the key JAK and STAT pathways involved in atopic dermatitis because it will help you determine the selectivity of the agents you’re going to be using,” the dermatologist advised.
Three oral, once-daily JAK inhibitors – abrocitinib, upadacitinib, and baricitinib – are in an advanced stage of development.
“Upadacitinib and abrocitinib may be the two most potent options coming to market soon for us to be thinking about,” Dr. Silverberg said.
Abrocitinib: Three positive phase 3 studies featuring this selective JAK1 inhibitor have been completed in adults with moderate to severe atopic dermatitis (AD). The most recent, JADE COMPARE, featured a head-to-head randomized comparison of abrocitinib and the injectable IL-4/IL-13 inhibitor dupilumab. The results of this 837-patient study haven’t yet been formally presented at a conference because of the COVID-19 pandemic. However, Pfizer recently announced that abrocitinib at 200 mg/day achieved significantly greater improvements than dupilumab (Dupixent) in the coprimary endpoints of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (that is, clear or almost clear) and disease extent based upon 75% reduction from baseline in Eczema Area and Severity Index (EASI 75) at 12 weeks. The same was true at 16 weeks.
Also, a significantly larger proportion of abrocitinib-treated patients achieved at least a 4-point reduction in itch severity as measured using the Peak Pruritus Numerical Rating Scale at week 2. The company plans to file for regulatory approval later this year.
The JADE COMPARE data are exciting because of a pressing unmet need for treatment options that are even more powerful than dupilumab, Dr. Silverberg said.
Upadacitinib: This is selective JAK1 inhibitor is not as far along in the developmental pipeline as abrocitinib, but the efficacy appears to be comparable. In a phase 2 study of 126 adults with moderate to severe AD, upadacitinib at the top dose of 30 mg/day achieved efficacy results Dr. Silverberg deemed “quite extraordinary,” with a rate of IGA score of 0 or 1 of 50% at 16 weeks and an EASI 75 response rate of 69%. Those findings numerically eclipsed results seen in an earlier phase 3 pivotal trial for dupilumab, in which the IGA 0/1 rate was 37% and EASI 75 was 48%, albeit with the caveat that cross-trial comparisons must be taken with a large grain of salt.
Baricitinib: Multiple phase 3 studies of this JAK 1/2 inhibitor have reported positive results. At the top dose of 4 mg/day, baricitinib appears to be less effective than dupilumab in its earlier pivotal trials.
“This may be a good oral option for our patients. It could be similar to the Otezla [apremilast] story in psoriasis: It’s perhaps not as effective as a lot of the biologics, but patients often prefer an oral option,” Dr. Silverberg said.
Of note, in one large, placebo-controlled, phase 3 study of baricitinib on top of background low- or medium-potency topical steroids, the IGA 0/1 rate at 16 weeks with placebo plus topical steroids was a modest 14.7%, which underscores that this long-time workhorse topical therapy is objectively less effective than most physicians think. In contrast, the IGA 0/1 rate with baricitinib at 4 mg/day plus topical steroids was a more respectable 30.6%.
All three oral JAK inhibitors have rapid onset of efficacy, a key advantage over the biologic agents.
“The issue you have to keep in mind is safety. The safety in the atopic dermatitis population was overall quite good for all three drugs. However, safety concerns have come up with JAK inhibitors in rheumatoid arthritis. I think that’s the part we watch the most in this. The efficacy has become clear. Now the question is where does the safety take us,” he said.
Novel injectable biologics
Nemolizumab: This humanized monoclonal antibody inhibits IL-31 receptor alpha. Mounting evidence implicates IL-31 as both a proinflammatory and immunomodulatory cytokine linking the immune and neural systems.
Early on, most researchers pigeonholed IL-31 as being a key player only in the itch factor in AD. Not so. Indeed, Dr. Silverberg was the lead investigator in a recent phase 2b study of nemolizumab that demonstrated the biologic is also effective at rapidly clearing AD lesions. The study, which evaluated three different doses in 226 adults with moderate to severe AD and severe pruritus who were on background topical corticosteroids, showed that nemolizumab at 30 mg every 4 weeks trounced placebo in terms of itch reduction: The 69% drop from baseline in Peak Pruritus Numeric Rating Scale at week 16 was twice that in controls, with a significant difference apparent even at week 1.
But in addition, the 33% IGA 0/1 rate at the same time point bested the 12% rate in controls. The EASI 75 response rate was significantly higher as well – 49% versus 19% – as was the EASI 90 response of 33%, compared with 9% in controls. Moreover, nemolizumab-treated patients used close to 40% less topical steroids during the study (J Allergy Clin Immunol. 2020 Jan;145[1]:173-82).
“This is something that’s fascinating. The study gets into the idea that a subset of atopic dermatitis patients have the itch that rashes, and perhaps if you break the itch/scratch cycle you can modify the lesions. Or the effect may even be due to the direct anti-inflammatory action of IL-31 blockade,” Dr. Silverberg observed.
It appeared that a plateau hadn’t been reached for some endpoints out at week 24, when the study ended. Japanese phase 3 studies have been completed, with what he called “great results,” and others are ongoing in the United States.
Tralokinumab: This fully human monoclonal antibody binds to IL-13, but unlike dupilumab, it doesn’t also inhibit IL-4. Tralokinumab met all primary and secondary endpoints in three pivotal phase 3 clinical trials, known as ECZTRA 1-3, that assessed it as treatment for moderate to severe AD in adults and showed an overall adverse event rate comparable with placebo. Leo Pharma, the Danish company developing the biologic, has announced it will file for marketing approval before the end of 2020. Phase 3 data would have been presented at the annual meeting of the American Academy of Dermatology in Denver, had it not been canceled. Dr. Silverberg said that, based upon phase 2 results, it appears tralokinumab may not be quite as effective as dupilumab in the overall AD population, but he predicted the newcomer will still play a useful role.
“The complexities of the immune system are such that some patients will respond better to one drug than another. I think we still have a lot to learn about who the patients are for these novel assets,” he said.
Lebrikizumab: This is another selective IL-13 inhibitor, but this one binds to IL-13 in a slightly different way than tralokinumab. The Food and Drug Administration granted it Fast Track status in December 2019. Twin placebo-controlled phase 3 studies of lebrikizumab as monotherapy for moderate to severe AD are ongoing, and another phase 3 trial of the biologic in combination with topical steroids is planned. Based upon the results of a phase 2b study, the highest dose studied – 250 mg every 2 weeks – appears to be at least as effective as dupilumab.
Nonsteroidal topical agents
These three late-stage topical creams – ruxolitinib, delgocitinib, and tapinarof – have previously received considerable coverage in Dermatology News. Ruxolitinib, a selective JAK1/2 inhibitor, has completed a positive phase 3 trial in adolescents and adults with mild to moderate AD. Delgocitinib, a pan-JAK1/2/3 and Tyrosine kinase 2 inhibitor, is already approved in an ointment formulation in Japan, and the cream formulation is in phase 2 studies in the United States and Europe. Tapinarof has a unique mechanism of action – it’s an aryl hydrocarbon receptor modulator – and is now in phase 3 in adolescents and adults with moderate to severe AD.
These three drugs appear to offer efficacy that’s comparable to or even better than medium-potency topical steroids, and without the notorious steroidal side effects that have caused widespread parental steroid-phobia. Potential applications for other inflammatory diseases, including vitiligo and psoriasis, are under study.
Dr. Silverberg reported receiving research grants from Galderma and GlaxoSmithKline and serving as a consultant to those pharmaceutical companies and more than a dozen others.
Major , Jonathan I. Silverberg, MD, PhD, said during a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled due to the COVID-19 pandemic.
“In the next 2-3 years, we may have nine new treatments approved for atopic dermatitis,” said Dr. Silverberg, director of clinical research and contact dermatitis at the University.
All nine medications he discussed are either in ongoing pivotal phase 3 randomized controlled trials or have completed their phase 3 developmental programs. “This is not theoretical; these are things you’re going to be using in your toolbox imminently,” he stressed.
Oral JAK inhibitors
The Janus kinase (JAK) pathway is the intracellular signaling mediator that interacts with extracellular inflammatory cytokines, including interleukin-4, -13, and -31, which are familiar to dermatologists because they’re targeted by potent biologic monoclonal antibody therapies. For example, IL-4 goes through JAK1 and 3, while IL-31 signals through JAK1 and 2.
“You really need to know the key JAK and STAT pathways involved in atopic dermatitis because it will help you determine the selectivity of the agents you’re going to be using,” the dermatologist advised.
Three oral, once-daily JAK inhibitors – abrocitinib, upadacitinib, and baricitinib – are in an advanced stage of development.
“Upadacitinib and abrocitinib may be the two most potent options coming to market soon for us to be thinking about,” Dr. Silverberg said.
Abrocitinib: Three positive phase 3 studies featuring this selective JAK1 inhibitor have been completed in adults with moderate to severe atopic dermatitis (AD). The most recent, JADE COMPARE, featured a head-to-head randomized comparison of abrocitinib and the injectable IL-4/IL-13 inhibitor dupilumab. The results of this 837-patient study haven’t yet been formally presented at a conference because of the COVID-19 pandemic. However, Pfizer recently announced that abrocitinib at 200 mg/day achieved significantly greater improvements than dupilumab (Dupixent) in the coprimary endpoints of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (that is, clear or almost clear) and disease extent based upon 75% reduction from baseline in Eczema Area and Severity Index (EASI 75) at 12 weeks. The same was true at 16 weeks.
Also, a significantly larger proportion of abrocitinib-treated patients achieved at least a 4-point reduction in itch severity as measured using the Peak Pruritus Numerical Rating Scale at week 2. The company plans to file for regulatory approval later this year.
The JADE COMPARE data are exciting because of a pressing unmet need for treatment options that are even more powerful than dupilumab, Dr. Silverberg said.
Upadacitinib: This is selective JAK1 inhibitor is not as far along in the developmental pipeline as abrocitinib, but the efficacy appears to be comparable. In a phase 2 study of 126 adults with moderate to severe AD, upadacitinib at the top dose of 30 mg/day achieved efficacy results Dr. Silverberg deemed “quite extraordinary,” with a rate of IGA score of 0 or 1 of 50% at 16 weeks and an EASI 75 response rate of 69%. Those findings numerically eclipsed results seen in an earlier phase 3 pivotal trial for dupilumab, in which the IGA 0/1 rate was 37% and EASI 75 was 48%, albeit with the caveat that cross-trial comparisons must be taken with a large grain of salt.
Baricitinib: Multiple phase 3 studies of this JAK 1/2 inhibitor have reported positive results. At the top dose of 4 mg/day, baricitinib appears to be less effective than dupilumab in its earlier pivotal trials.
“This may be a good oral option for our patients. It could be similar to the Otezla [apremilast] story in psoriasis: It’s perhaps not as effective as a lot of the biologics, but patients often prefer an oral option,” Dr. Silverberg said.
Of note, in one large, placebo-controlled, phase 3 study of baricitinib on top of background low- or medium-potency topical steroids, the IGA 0/1 rate at 16 weeks with placebo plus topical steroids was a modest 14.7%, which underscores that this long-time workhorse topical therapy is objectively less effective than most physicians think. In contrast, the IGA 0/1 rate with baricitinib at 4 mg/day plus topical steroids was a more respectable 30.6%.
All three oral JAK inhibitors have rapid onset of efficacy, a key advantage over the biologic agents.
“The issue you have to keep in mind is safety. The safety in the atopic dermatitis population was overall quite good for all three drugs. However, safety concerns have come up with JAK inhibitors in rheumatoid arthritis. I think that’s the part we watch the most in this. The efficacy has become clear. Now the question is where does the safety take us,” he said.
Novel injectable biologics
Nemolizumab: This humanized monoclonal antibody inhibits IL-31 receptor alpha. Mounting evidence implicates IL-31 as both a proinflammatory and immunomodulatory cytokine linking the immune and neural systems.
Early on, most researchers pigeonholed IL-31 as being a key player only in the itch factor in AD. Not so. Indeed, Dr. Silverberg was the lead investigator in a recent phase 2b study of nemolizumab that demonstrated the biologic is also effective at rapidly clearing AD lesions. The study, which evaluated three different doses in 226 adults with moderate to severe AD and severe pruritus who were on background topical corticosteroids, showed that nemolizumab at 30 mg every 4 weeks trounced placebo in terms of itch reduction: The 69% drop from baseline in Peak Pruritus Numeric Rating Scale at week 16 was twice that in controls, with a significant difference apparent even at week 1.
But in addition, the 33% IGA 0/1 rate at the same time point bested the 12% rate in controls. The EASI 75 response rate was significantly higher as well – 49% versus 19% – as was the EASI 90 response of 33%, compared with 9% in controls. Moreover, nemolizumab-treated patients used close to 40% less topical steroids during the study (J Allergy Clin Immunol. 2020 Jan;145[1]:173-82).
“This is something that’s fascinating. The study gets into the idea that a subset of atopic dermatitis patients have the itch that rashes, and perhaps if you break the itch/scratch cycle you can modify the lesions. Or the effect may even be due to the direct anti-inflammatory action of IL-31 blockade,” Dr. Silverberg observed.
It appeared that a plateau hadn’t been reached for some endpoints out at week 24, when the study ended. Japanese phase 3 studies have been completed, with what he called “great results,” and others are ongoing in the United States.
Tralokinumab: This fully human monoclonal antibody binds to IL-13, but unlike dupilumab, it doesn’t also inhibit IL-4. Tralokinumab met all primary and secondary endpoints in three pivotal phase 3 clinical trials, known as ECZTRA 1-3, that assessed it as treatment for moderate to severe AD in adults and showed an overall adverse event rate comparable with placebo. Leo Pharma, the Danish company developing the biologic, has announced it will file for marketing approval before the end of 2020. Phase 3 data would have been presented at the annual meeting of the American Academy of Dermatology in Denver, had it not been canceled. Dr. Silverberg said that, based upon phase 2 results, it appears tralokinumab may not be quite as effective as dupilumab in the overall AD population, but he predicted the newcomer will still play a useful role.
“The complexities of the immune system are such that some patients will respond better to one drug than another. I think we still have a lot to learn about who the patients are for these novel assets,” he said.
Lebrikizumab: This is another selective IL-13 inhibitor, but this one binds to IL-13 in a slightly different way than tralokinumab. The Food and Drug Administration granted it Fast Track status in December 2019. Twin placebo-controlled phase 3 studies of lebrikizumab as monotherapy for moderate to severe AD are ongoing, and another phase 3 trial of the biologic in combination with topical steroids is planned. Based upon the results of a phase 2b study, the highest dose studied – 250 mg every 2 weeks – appears to be at least as effective as dupilumab.
Nonsteroidal topical agents
These three late-stage topical creams – ruxolitinib, delgocitinib, and tapinarof – have previously received considerable coverage in Dermatology News. Ruxolitinib, a selective JAK1/2 inhibitor, has completed a positive phase 3 trial in adolescents and adults with mild to moderate AD. Delgocitinib, a pan-JAK1/2/3 and Tyrosine kinase 2 inhibitor, is already approved in an ointment formulation in Japan, and the cream formulation is in phase 2 studies in the United States and Europe. Tapinarof has a unique mechanism of action – it’s an aryl hydrocarbon receptor modulator – and is now in phase 3 in adolescents and adults with moderate to severe AD.
These three drugs appear to offer efficacy that’s comparable to or even better than medium-potency topical steroids, and without the notorious steroidal side effects that have caused widespread parental steroid-phobia. Potential applications for other inflammatory diseases, including vitiligo and psoriasis, are under study.
Dr. Silverberg reported receiving research grants from Galderma and GlaxoSmithKline and serving as a consultant to those pharmaceutical companies and more than a dozen others.
Case series suggests biologics, JAK inhibitors safe during pandemic
Use of biologics and Janus kinase (JAK) inhibitors was not associated with worse outcomes in 86 people with inflammatory diseases who contracted COVID-19, according to a case series from New York University Langone Health.
“We are not seeing worse outcomes with overall use of either. It’s reassuring” that the data support continued use during the pandemic, said rheumatologist and senior investigator Jose Scher, MD, an associate professor at New York University.
There have been concerns among rheumatologists, gastroenterologists, and dermatologists that underlying inflammatory diseases and the agents used to treat them would impact outcomes in COVID-19.
Dr. Scher and colleagues, including lead author and rheumatologist Rebecca Haberman, MD, wanted to address the issue, so they reviewed the experience in their own health system of patients with inflammatory diseases – most commonly psoriatic arthritis, RA, and Crohn’s disease – who were assessed for COVID-19 from March 3 to April 3.
Fever, cough, and shortness of breath were the most common symptoms. The infection was confirmed by polymerase chain reaction in 59 (69%) and highly suspected in 27.
A total of 62 patients (72%) were on JAK inhibitors or biologics at baseline, including 38 (44%) on tumor necrosis factor inhibitors.
Overall, 14 patients (16%) were hospitalized with COVID-19, which is consistent the 26% hospitalization rate among the general population in New York City.
Baseline biologic and JAK inhibitor use was actually lower among hospitalized patients than among those who weren’t hospitalized (50% vs. 76%), and the hospitalization rate was only 11% among 62 subjects who had been on the agents long term, more than a year among most.
Hospitalized patients tended to be slightly older (mean, 50 vs. 46 years) with a higher prevalence of hypertension, diabetes, and chronic obstructive pulmonary disease. They also had a higher prevalence of RA (43% vs. 19%), methotrexate use (43% vs. 15%), and use of hydroxychloroquine (21% vs. 7%) and oral glucocorticoids (29% vs. 6%).
It’s unknown what to make of those findings for now, Dr. Scher said. The study didn’t address differences in the severity of the underlying inflammatory illness, but a new and significantly larger case series is in the works that will analyze that and other potential confounders.
Dr. Scher noted that he’s particularly interested in drilling down further on the higher prevalence of RA and methotrexate in hospitalized patients. “We want to understand those signals better. All of this needs further validation,” he said.
Of the 14 hospitalized patients, 11 (79%) were discharged after a mean of 5.6 days. One died in the ED, and two remained hospitalized as of April 3, including one in the ICU.
The investigators are contributing to COVID-19 registries for inflammatory disease patients. The registries are tending to report higher hospitalization rates, but Dr. Scher noted they might be biased towards more severe cases, among other issues.
As for the current situation in New York City, he said that the “last week in March and first 3 in April were indescribable in terms of admissions, intubations, and deaths. Over the last week or so, it has calmed down significantly.”
There was no external funding. Dr. Haberman reported ties to Janssen, and Dr. Scher reported ties to Janssen, Novartis, Pfizer, and other companies.
SOURCE: Haberman R et al. N Engl J Med. 2020 Apr 29. doi: 10.1056/NEJMc2009567.
Use of biologics and Janus kinase (JAK) inhibitors was not associated with worse outcomes in 86 people with inflammatory diseases who contracted COVID-19, according to a case series from New York University Langone Health.
“We are not seeing worse outcomes with overall use of either. It’s reassuring” that the data support continued use during the pandemic, said rheumatologist and senior investigator Jose Scher, MD, an associate professor at New York University.
There have been concerns among rheumatologists, gastroenterologists, and dermatologists that underlying inflammatory diseases and the agents used to treat them would impact outcomes in COVID-19.
Dr. Scher and colleagues, including lead author and rheumatologist Rebecca Haberman, MD, wanted to address the issue, so they reviewed the experience in their own health system of patients with inflammatory diseases – most commonly psoriatic arthritis, RA, and Crohn’s disease – who were assessed for COVID-19 from March 3 to April 3.
Fever, cough, and shortness of breath were the most common symptoms. The infection was confirmed by polymerase chain reaction in 59 (69%) and highly suspected in 27.
A total of 62 patients (72%) were on JAK inhibitors or biologics at baseline, including 38 (44%) on tumor necrosis factor inhibitors.
Overall, 14 patients (16%) were hospitalized with COVID-19, which is consistent the 26% hospitalization rate among the general population in New York City.
Baseline biologic and JAK inhibitor use was actually lower among hospitalized patients than among those who weren’t hospitalized (50% vs. 76%), and the hospitalization rate was only 11% among 62 subjects who had been on the agents long term, more than a year among most.
Hospitalized patients tended to be slightly older (mean, 50 vs. 46 years) with a higher prevalence of hypertension, diabetes, and chronic obstructive pulmonary disease. They also had a higher prevalence of RA (43% vs. 19%), methotrexate use (43% vs. 15%), and use of hydroxychloroquine (21% vs. 7%) and oral glucocorticoids (29% vs. 6%).
It’s unknown what to make of those findings for now, Dr. Scher said. The study didn’t address differences in the severity of the underlying inflammatory illness, but a new and significantly larger case series is in the works that will analyze that and other potential confounders.
Dr. Scher noted that he’s particularly interested in drilling down further on the higher prevalence of RA and methotrexate in hospitalized patients. “We want to understand those signals better. All of this needs further validation,” he said.
Of the 14 hospitalized patients, 11 (79%) were discharged after a mean of 5.6 days. One died in the ED, and two remained hospitalized as of April 3, including one in the ICU.
The investigators are contributing to COVID-19 registries for inflammatory disease patients. The registries are tending to report higher hospitalization rates, but Dr. Scher noted they might be biased towards more severe cases, among other issues.
As for the current situation in New York City, he said that the “last week in March and first 3 in April were indescribable in terms of admissions, intubations, and deaths. Over the last week or so, it has calmed down significantly.”
There was no external funding. Dr. Haberman reported ties to Janssen, and Dr. Scher reported ties to Janssen, Novartis, Pfizer, and other companies.
SOURCE: Haberman R et al. N Engl J Med. 2020 Apr 29. doi: 10.1056/NEJMc2009567.
Use of biologics and Janus kinase (JAK) inhibitors was not associated with worse outcomes in 86 people with inflammatory diseases who contracted COVID-19, according to a case series from New York University Langone Health.
“We are not seeing worse outcomes with overall use of either. It’s reassuring” that the data support continued use during the pandemic, said rheumatologist and senior investigator Jose Scher, MD, an associate professor at New York University.
There have been concerns among rheumatologists, gastroenterologists, and dermatologists that underlying inflammatory diseases and the agents used to treat them would impact outcomes in COVID-19.
Dr. Scher and colleagues, including lead author and rheumatologist Rebecca Haberman, MD, wanted to address the issue, so they reviewed the experience in their own health system of patients with inflammatory diseases – most commonly psoriatic arthritis, RA, and Crohn’s disease – who were assessed for COVID-19 from March 3 to April 3.
Fever, cough, and shortness of breath were the most common symptoms. The infection was confirmed by polymerase chain reaction in 59 (69%) and highly suspected in 27.
A total of 62 patients (72%) were on JAK inhibitors or biologics at baseline, including 38 (44%) on tumor necrosis factor inhibitors.
Overall, 14 patients (16%) were hospitalized with COVID-19, which is consistent the 26% hospitalization rate among the general population in New York City.
Baseline biologic and JAK inhibitor use was actually lower among hospitalized patients than among those who weren’t hospitalized (50% vs. 76%), and the hospitalization rate was only 11% among 62 subjects who had been on the agents long term, more than a year among most.
Hospitalized patients tended to be slightly older (mean, 50 vs. 46 years) with a higher prevalence of hypertension, diabetes, and chronic obstructive pulmonary disease. They also had a higher prevalence of RA (43% vs. 19%), methotrexate use (43% vs. 15%), and use of hydroxychloroquine (21% vs. 7%) and oral glucocorticoids (29% vs. 6%).
It’s unknown what to make of those findings for now, Dr. Scher said. The study didn’t address differences in the severity of the underlying inflammatory illness, but a new and significantly larger case series is in the works that will analyze that and other potential confounders.
Dr. Scher noted that he’s particularly interested in drilling down further on the higher prevalence of RA and methotrexate in hospitalized patients. “We want to understand those signals better. All of this needs further validation,” he said.
Of the 14 hospitalized patients, 11 (79%) were discharged after a mean of 5.6 days. One died in the ED, and two remained hospitalized as of April 3, including one in the ICU.
The investigators are contributing to COVID-19 registries for inflammatory disease patients. The registries are tending to report higher hospitalization rates, but Dr. Scher noted they might be biased towards more severe cases, among other issues.
As for the current situation in New York City, he said that the “last week in March and first 3 in April were indescribable in terms of admissions, intubations, and deaths. Over the last week or so, it has calmed down significantly.”
There was no external funding. Dr. Haberman reported ties to Janssen, and Dr. Scher reported ties to Janssen, Novartis, Pfizer, and other companies.
SOURCE: Haberman R et al. N Engl J Med. 2020 Apr 29. doi: 10.1056/NEJMc2009567.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
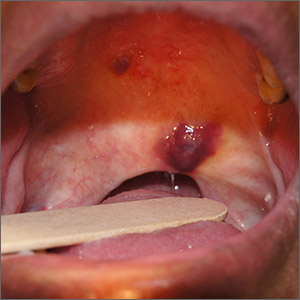
Purple oral plaques
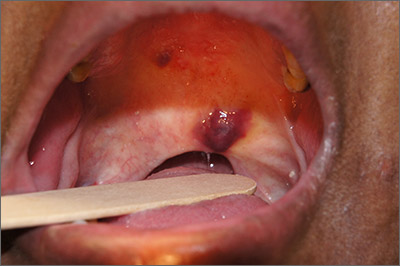
The papules were clinically consistent with Kaposi sarcoma (KS) and confirmed with a biopsy from the buccal mucosa. The patient had not been given a diagnosis of human immunodeficiency virus (HIV) prior to this presentation. However, the physician confirmed the diagnosis with RNA titers. A CD4 count < 40 cells/mm3 (reference range, 500-1200 cells/mm3) pointed to her progression to AIDS. A chest X-ray revealed multiple nodules; a subsequent biopsy indicated that they were consistent with KS.
KS is a low-grade tumor of vascular origin associated with human herpesvirus-8 (HHV-8). It most often presents on the skin as flat to raised pink to deep purple lesions. It can manifest in the oral mucosa, viscera, and other organs, which can portend a worse prognosis because of the risks associated with bleeding and organ perforation.
There are 4 types of KS.
- Classic KS occurs most often in elderly men of Mediterranean descent.
- HIV-associated KS can occur at any time during HIV infection but is more common as CD4 counts fall. HIV-associated KS increased in frequency dramatically in the United States during the early years of the HIV pandemic prior to effective antiretroviral therapy (ART).
- Endemic KS occurs in equatorial Africa, where there is a natural increased transmission rate of HHV-8.
- Iatrogenic KS can occur following treatment with immunosuppressive therapies.
Our patient was admitted to the Infectious Disease Service and given ART. Chemotherapy was discussed (and sometimes is warranted in extensive visceral disease) but the patient and her specialists opted for ART alone. In addition to ART, she was started on daily trimethoprim-sulfamethoxazole for pneumocystis prophylaxis.
At 6 months’ follow-up with Infectious Disease, the patient’s oral lesions resolved, CD4 count increased above 200 cells/mm3, and HIV RNA titers fell.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Thariat J, Kirova Y, Sio T, et al. Mucosal Kaposi sarcoma, a Rare Cancer Network study. Rare Tumors. 2012;4:E49.

The papules were clinically consistent with Kaposi sarcoma (KS) and confirmed with a biopsy from the buccal mucosa. The patient had not been given a diagnosis of human immunodeficiency virus (HIV) prior to this presentation. However, the physician confirmed the diagnosis with RNA titers. A CD4 count < 40 cells/mm3 (reference range, 500-1200 cells/mm3) pointed to her progression to AIDS. A chest X-ray revealed multiple nodules; a subsequent biopsy indicated that they were consistent with KS.
KS is a low-grade tumor of vascular origin associated with human herpesvirus-8 (HHV-8). It most often presents on the skin as flat to raised pink to deep purple lesions. It can manifest in the oral mucosa, viscera, and other organs, which can portend a worse prognosis because of the risks associated with bleeding and organ perforation.
There are 4 types of KS.
- Classic KS occurs most often in elderly men of Mediterranean descent.
- HIV-associated KS can occur at any time during HIV infection but is more common as CD4 counts fall. HIV-associated KS increased in frequency dramatically in the United States during the early years of the HIV pandemic prior to effective antiretroviral therapy (ART).
- Endemic KS occurs in equatorial Africa, where there is a natural increased transmission rate of HHV-8.
- Iatrogenic KS can occur following treatment with immunosuppressive therapies.
Our patient was admitted to the Infectious Disease Service and given ART. Chemotherapy was discussed (and sometimes is warranted in extensive visceral disease) but the patient and her specialists opted for ART alone. In addition to ART, she was started on daily trimethoprim-sulfamethoxazole for pneumocystis prophylaxis.
At 6 months’ follow-up with Infectious Disease, the patient’s oral lesions resolved, CD4 count increased above 200 cells/mm3, and HIV RNA titers fell.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The papules were clinically consistent with Kaposi sarcoma (KS) and confirmed with a biopsy from the buccal mucosa. The patient had not been given a diagnosis of human immunodeficiency virus (HIV) prior to this presentation. However, the physician confirmed the diagnosis with RNA titers. A CD4 count < 40 cells/mm3 (reference range, 500-1200 cells/mm3) pointed to her progression to AIDS. A chest X-ray revealed multiple nodules; a subsequent biopsy indicated that they were consistent with KS.
KS is a low-grade tumor of vascular origin associated with human herpesvirus-8 (HHV-8). It most often presents on the skin as flat to raised pink to deep purple lesions. It can manifest in the oral mucosa, viscera, and other organs, which can portend a worse prognosis because of the risks associated with bleeding and organ perforation.
There are 4 types of KS.
- Classic KS occurs most often in elderly men of Mediterranean descent.
- HIV-associated KS can occur at any time during HIV infection but is more common as CD4 counts fall. HIV-associated KS increased in frequency dramatically in the United States during the early years of the HIV pandemic prior to effective antiretroviral therapy (ART).
- Endemic KS occurs in equatorial Africa, where there is a natural increased transmission rate of HHV-8.
- Iatrogenic KS can occur following treatment with immunosuppressive therapies.
Our patient was admitted to the Infectious Disease Service and given ART. Chemotherapy was discussed (and sometimes is warranted in extensive visceral disease) but the patient and her specialists opted for ART alone. In addition to ART, she was started on daily trimethoprim-sulfamethoxazole for pneumocystis prophylaxis.
At 6 months’ follow-up with Infectious Disease, the patient’s oral lesions resolved, CD4 count increased above 200 cells/mm3, and HIV RNA titers fell.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Thariat J, Kirova Y, Sio T, et al. Mucosal Kaposi sarcoma, a Rare Cancer Network study. Rare Tumors. 2012;4:E49.
Thariat J, Kirova Y, Sio T, et al. Mucosal Kaposi sarcoma, a Rare Cancer Network study. Rare Tumors. 2012;4:E49.
COVID-19: Frequently asked clinical questions
Question
How should patients on immunosuppressive therapy be advised during the COVID-19 pandemic?
Answer
In general, those patients who have not tested positive, have not been exposed, and are asymptomatic should continue their medications as prescribed.
The American College of Rheumatology issued a statement on April 14, recommending that stable patients continue their medications. Those with known exposure but without confirmed infection may continue hydroxychloroquine, sulfasalazine, and NSAIDs.
Immunosuppressants, non–IL-6 biologics, and JAK inhibitors should be stopped temporarily, pending a negative test or after two weeks without symptoms. In patients with confirmed positive COVID-19 infection, sulfasalazine, methotrexate, leflunomide, immunosuppressants, non-IL-6 biologics, and JAK inhibitors should be stopped temporarily, pending a negative test or after two weeks without symptoms. In patients with confirmed positive COVID-19 infection, sulfasalazine, methotrexate, leflunomide, immunosuppressants, non-IL-6 biologics, and JAK inhibitors should be stopped temporarily. Anti-malarial therapies (hydroxycholoroquine and chloroquine) may be continued and IL-6 inhibitors may be continued in select circumstances.1
The American Academy of Dermatology recommends that the discussion of continuation of biologics be based on a case-by-case basis, citing insufficient evidence to recommend against discontinuation at this time in those patients who have not tested positive. In patients who have tested positive for COVID-19 it is recommended that biologic therapy be suspended until symptoms have resolved.2
Question
Should I continue preventive services during peak COVID-19?
Answer
The Centers for Disease Control and Prevention recommends delaying all elective ambulatory provider visits. In general, preventative services, such as adult immunizations, lipid screening, and cancer screenings, should be delayed. Additionally, the CDC recommends reaching out to patients who are at high risk for complications from respiratory diseases to ensure medication adherence and provide resources if these patients become ill. Facilities can reduce transmission of COVID-19 by triaging and assessing patients through virtual visits through phone calls, video conferences, text-monitoring systems, and other telemedicine tools. Physicians should try to provide routine and chronic care through virtual visits when possible over in-person visits.3
Question
Should I continue to vaccinate my pediatric population during peak COVID-19?
Answer
Practices that schedule separate well visits and sick visits in different sessions or locations can continue to provide well child visits. A practice could, for example, schedule well visits in the morning and sick visits in the afternoon if a single facility is used. These practices should prioritize newborn care and vaccinations of children, especially for those under the age of 24 months.4
Question
Can physicians use telehealth (phone only or audiovisual) to conduct visits with Medicare patients even if they are new patients?
Answer
Effective March 1 through the duration of the pandemic, Medicare will pay physicians for telehealth services at the same rate as an in-office visit. On March 30th, the Centers for Medicare & Medcaid Services announced new policies for physicians and hospitals during the COVID-19 pandemic. These guidelines were updated on April 9.
Audio-only visits are now permitted and the limit on the number of these kinds of visits allowed per month has been waived. Controlled substances can be prescribed via telehealth; however, complying with each state’s individual laws is still required.
Use of any two-way, audiovisual device is permitted. The level of service billed for visits with both audio and visual components is the same as an in-office visit. Telemedicine can be used for both new and existing patients.5
A list of services that may be rendered via telehealth are available on the CMS website.6
It will be important to regularly check the references given, as information on some of these topics is updated frequently.
Dr. Chuong is a second-year resident in the family medicine residency, Dr. Flanagan is a third-year resident, and Dr. Matthews is an intern, all at Abington (Pa.) Jefferson Health. Dr. Skolnik is professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
References
1. ACR issues COVID-19 treatment guidance for rheumatic disease patients.
2. American Academy of Dermatology: Guidance on the use of biologic agents during COVID-19 outbreak.
3. Centers for Disease Control and Prevention. Actions to take in response to community transmission of COVID-19.
4. Centers for Disease Control and Prevention. Maintaining childhood immunizations during COVID19 pandemic.
5. Centers for Medicare & Medcaid Services. COVID-19 frequently asked questions (FAQs) on Medicare Fee-for-Service (FFS) billing.
6. Centers for Medicare & Medcaid Services. List of telehealth services.
Question
How should patients on immunosuppressive therapy be advised during the COVID-19 pandemic?
Answer
In general, those patients who have not tested positive, have not been exposed, and are asymptomatic should continue their medications as prescribed.
The American College of Rheumatology issued a statement on April 14, recommending that stable patients continue their medications. Those with known exposure but without confirmed infection may continue hydroxychloroquine, sulfasalazine, and NSAIDs.
Immunosuppressants, non–IL-6 biologics, and JAK inhibitors should be stopped temporarily, pending a negative test or after two weeks without symptoms. In patients with confirmed positive COVID-19 infection, sulfasalazine, methotrexate, leflunomide, immunosuppressants, non-IL-6 biologics, and JAK inhibitors should be stopped temporarily, pending a negative test or after two weeks without symptoms. In patients with confirmed positive COVID-19 infection, sulfasalazine, methotrexate, leflunomide, immunosuppressants, non-IL-6 biologics, and JAK inhibitors should be stopped temporarily. Anti-malarial therapies (hydroxycholoroquine and chloroquine) may be continued and IL-6 inhibitors may be continued in select circumstances.1
The American Academy of Dermatology recommends that the discussion of continuation of biologics be based on a case-by-case basis, citing insufficient evidence to recommend against discontinuation at this time in those patients who have not tested positive. In patients who have tested positive for COVID-19 it is recommended that biologic therapy be suspended until symptoms have resolved.2
Question
Should I continue preventive services during peak COVID-19?
Answer
The Centers for Disease Control and Prevention recommends delaying all elective ambulatory provider visits. In general, preventative services, such as adult immunizations, lipid screening, and cancer screenings, should be delayed. Additionally, the CDC recommends reaching out to patients who are at high risk for complications from respiratory diseases to ensure medication adherence and provide resources if these patients become ill. Facilities can reduce transmission of COVID-19 by triaging and assessing patients through virtual visits through phone calls, video conferences, text-monitoring systems, and other telemedicine tools. Physicians should try to provide routine and chronic care through virtual visits when possible over in-person visits.3
Question
Should I continue to vaccinate my pediatric population during peak COVID-19?
Answer
Practices that schedule separate well visits and sick visits in different sessions or locations can continue to provide well child visits. A practice could, for example, schedule well visits in the morning and sick visits in the afternoon if a single facility is used. These practices should prioritize newborn care and vaccinations of children, especially for those under the age of 24 months.4
Question
Can physicians use telehealth (phone only or audiovisual) to conduct visits with Medicare patients even if they are new patients?
Answer
Effective March 1 through the duration of the pandemic, Medicare will pay physicians for telehealth services at the same rate as an in-office visit. On March 30th, the Centers for Medicare & Medcaid Services announced new policies for physicians and hospitals during the COVID-19 pandemic. These guidelines were updated on April 9.
Audio-only visits are now permitted and the limit on the number of these kinds of visits allowed per month has been waived. Controlled substances can be prescribed via telehealth; however, complying with each state’s individual laws is still required.
Use of any two-way, audiovisual device is permitted. The level of service billed for visits with both audio and visual components is the same as an in-office visit. Telemedicine can be used for both new and existing patients.5
A list of services that may be rendered via telehealth are available on the CMS website.6
It will be important to regularly check the references given, as information on some of these topics is updated frequently.
Dr. Chuong is a second-year resident in the family medicine residency, Dr. Flanagan is a third-year resident, and Dr. Matthews is an intern, all at Abington (Pa.) Jefferson Health. Dr. Skolnik is professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
References
1. ACR issues COVID-19 treatment guidance for rheumatic disease patients.
2. American Academy of Dermatology: Guidance on the use of biologic agents during COVID-19 outbreak.
3. Centers for Disease Control and Prevention. Actions to take in response to community transmission of COVID-19.
4. Centers for Disease Control and Prevention. Maintaining childhood immunizations during COVID19 pandemic.
5. Centers for Medicare & Medcaid Services. COVID-19 frequently asked questions (FAQs) on Medicare Fee-for-Service (FFS) billing.
6. Centers for Medicare & Medcaid Services. List of telehealth services.
Question
How should patients on immunosuppressive therapy be advised during the COVID-19 pandemic?
Answer
In general, those patients who have not tested positive, have not been exposed, and are asymptomatic should continue their medications as prescribed.
The American College of Rheumatology issued a statement on April 14, recommending that stable patients continue their medications. Those with known exposure but without confirmed infection may continue hydroxychloroquine, sulfasalazine, and NSAIDs.
Immunosuppressants, non–IL-6 biologics, and JAK inhibitors should be stopped temporarily, pending a negative test or after two weeks without symptoms. In patients with confirmed positive COVID-19 infection, sulfasalazine, methotrexate, leflunomide, immunosuppressants, non-IL-6 biologics, and JAK inhibitors should be stopped temporarily, pending a negative test or after two weeks without symptoms. In patients with confirmed positive COVID-19 infection, sulfasalazine, methotrexate, leflunomide, immunosuppressants, non-IL-6 biologics, and JAK inhibitors should be stopped temporarily. Anti-malarial therapies (hydroxycholoroquine and chloroquine) may be continued and IL-6 inhibitors may be continued in select circumstances.1
The American Academy of Dermatology recommends that the discussion of continuation of biologics be based on a case-by-case basis, citing insufficient evidence to recommend against discontinuation at this time in those patients who have not tested positive. In patients who have tested positive for COVID-19 it is recommended that biologic therapy be suspended until symptoms have resolved.2
Question
Should I continue preventive services during peak COVID-19?
Answer
The Centers for Disease Control and Prevention recommends delaying all elective ambulatory provider visits. In general, preventative services, such as adult immunizations, lipid screening, and cancer screenings, should be delayed. Additionally, the CDC recommends reaching out to patients who are at high risk for complications from respiratory diseases to ensure medication adherence and provide resources if these patients become ill. Facilities can reduce transmission of COVID-19 by triaging and assessing patients through virtual visits through phone calls, video conferences, text-monitoring systems, and other telemedicine tools. Physicians should try to provide routine and chronic care through virtual visits when possible over in-person visits.3
Question
Should I continue to vaccinate my pediatric population during peak COVID-19?
Answer
Practices that schedule separate well visits and sick visits in different sessions or locations can continue to provide well child visits. A practice could, for example, schedule well visits in the morning and sick visits in the afternoon if a single facility is used. These practices should prioritize newborn care and vaccinations of children, especially for those under the age of 24 months.4
Question
Can physicians use telehealth (phone only or audiovisual) to conduct visits with Medicare patients even if they are new patients?
Answer
Effective March 1 through the duration of the pandemic, Medicare will pay physicians for telehealth services at the same rate as an in-office visit. On March 30th, the Centers for Medicare & Medcaid Services announced new policies for physicians and hospitals during the COVID-19 pandemic. These guidelines were updated on April 9.
Audio-only visits are now permitted and the limit on the number of these kinds of visits allowed per month has been waived. Controlled substances can be prescribed via telehealth; however, complying with each state’s individual laws is still required.
Use of any two-way, audiovisual device is permitted. The level of service billed for visits with both audio and visual components is the same as an in-office visit. Telemedicine can be used for both new and existing patients.5
A list of services that may be rendered via telehealth are available on the CMS website.6
It will be important to regularly check the references given, as information on some of these topics is updated frequently.
Dr. Chuong is a second-year resident in the family medicine residency, Dr. Flanagan is a third-year resident, and Dr. Matthews is an intern, all at Abington (Pa.) Jefferson Health. Dr. Skolnik is professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
References
1. ACR issues COVID-19 treatment guidance for rheumatic disease patients.
2. American Academy of Dermatology: Guidance on the use of biologic agents during COVID-19 outbreak.
3. Centers for Disease Control and Prevention. Actions to take in response to community transmission of COVID-19.
4. Centers for Disease Control and Prevention. Maintaining childhood immunizations during COVID19 pandemic.
5. Centers for Medicare & Medcaid Services. COVID-19 frequently asked questions (FAQs) on Medicare Fee-for-Service (FFS) billing.
6. Centers for Medicare & Medcaid Services. List of telehealth services.
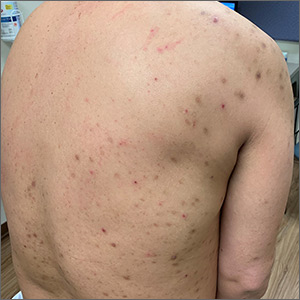
Itchy bumps on back
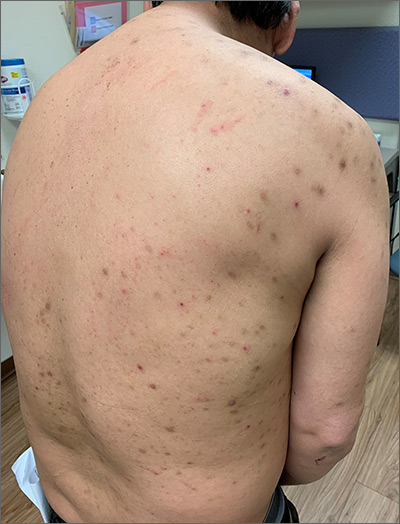
This patient had prurigo nodularis (PN). The diagnosis usually is made clinically by the appearance of the lesions and the cycle of severe pruritus and scratching. In this case, the patient had acutely excoriated lesions in addition to more chronic lesions that had become hyperpigmented nodules. The distribution pattern on his back was typical and highlighted the clinical course of PN. There were no lesions present where the patient was unable to scratch; however, lesions were present where he could reach, hence the term Picker’s nodules. Often, these patients have a history of atopic dermatitis, and anxiety may play a role in patients nervously scratching the lesions.
Biopsy is indicated if there is suspicion of bullous pemphigoid or cutaneous T-cell lymphoma. Pathology of PN shows increased density of nerve fibers in the dermis along with an increased number of T cells, mast cells, and eosinophilic granulocytes. Most patients do not require biopsy unless the diagnosis is in doubt.
Treatment can be difficult due to the severe pruritis and subsequent scratching that appears to prolong the chronic cycle of inflammation. Daily use of nonsedating antihistamines (eg, loratadine, cetirizine) may help reduce pruritus and break the cycle. Sedating antihistamines (eg, diphenhydramine, hydroxyzine) can be used cautiously at bedtime; cotton gloves worn while sleeping may reduce nocturnal scratching and excoriations.
Topical steroids (eg, triamcinolone, betamethasone) can reduce the itching and local inflammation. Emollients can help with associated dyshidrosis and eczema, if present.
Second line therapies include topical calcineurin inhibitors (eg, tacrolimus, pimecrolimus), calcipotriene, and narrow beam UVB therapy.
This patient had done reasonably well with cetirizine and triamcinolone in the past, so treatment was restarted. He was counseled regarding the nature and chronicity of his PN and told that if he could achieve symptom control and stop scratching the lesions, his condition might resolve.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Zeidler C, Yosipovitch G, Ständer S. Prurigo nodularis and its management. Dermatol Clin. 2018;36:189-197.

This patient had prurigo nodularis (PN). The diagnosis usually is made clinically by the appearance of the lesions and the cycle of severe pruritus and scratching. In this case, the patient had acutely excoriated lesions in addition to more chronic lesions that had become hyperpigmented nodules. The distribution pattern on his back was typical and highlighted the clinical course of PN. There were no lesions present where the patient was unable to scratch; however, lesions were present where he could reach, hence the term Picker’s nodules. Often, these patients have a history of atopic dermatitis, and anxiety may play a role in patients nervously scratching the lesions.
Biopsy is indicated if there is suspicion of bullous pemphigoid or cutaneous T-cell lymphoma. Pathology of PN shows increased density of nerve fibers in the dermis along with an increased number of T cells, mast cells, and eosinophilic granulocytes. Most patients do not require biopsy unless the diagnosis is in doubt.
Treatment can be difficult due to the severe pruritis and subsequent scratching that appears to prolong the chronic cycle of inflammation. Daily use of nonsedating antihistamines (eg, loratadine, cetirizine) may help reduce pruritus and break the cycle. Sedating antihistamines (eg, diphenhydramine, hydroxyzine) can be used cautiously at bedtime; cotton gloves worn while sleeping may reduce nocturnal scratching and excoriations.
Topical steroids (eg, triamcinolone, betamethasone) can reduce the itching and local inflammation. Emollients can help with associated dyshidrosis and eczema, if present.
Second line therapies include topical calcineurin inhibitors (eg, tacrolimus, pimecrolimus), calcipotriene, and narrow beam UVB therapy.
This patient had done reasonably well with cetirizine and triamcinolone in the past, so treatment was restarted. He was counseled regarding the nature and chronicity of his PN and told that if he could achieve symptom control and stop scratching the lesions, his condition might resolve.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This patient had prurigo nodularis (PN). The diagnosis usually is made clinically by the appearance of the lesions and the cycle of severe pruritus and scratching. In this case, the patient had acutely excoriated lesions in addition to more chronic lesions that had become hyperpigmented nodules. The distribution pattern on his back was typical and highlighted the clinical course of PN. There were no lesions present where the patient was unable to scratch; however, lesions were present where he could reach, hence the term Picker’s nodules. Often, these patients have a history of atopic dermatitis, and anxiety may play a role in patients nervously scratching the lesions.
Biopsy is indicated if there is suspicion of bullous pemphigoid or cutaneous T-cell lymphoma. Pathology of PN shows increased density of nerve fibers in the dermis along with an increased number of T cells, mast cells, and eosinophilic granulocytes. Most patients do not require biopsy unless the diagnosis is in doubt.
Treatment can be difficult due to the severe pruritis and subsequent scratching that appears to prolong the chronic cycle of inflammation. Daily use of nonsedating antihistamines (eg, loratadine, cetirizine) may help reduce pruritus and break the cycle. Sedating antihistamines (eg, diphenhydramine, hydroxyzine) can be used cautiously at bedtime; cotton gloves worn while sleeping may reduce nocturnal scratching and excoriations.
Topical steroids (eg, triamcinolone, betamethasone) can reduce the itching and local inflammation. Emollients can help with associated dyshidrosis and eczema, if present.
Second line therapies include topical calcineurin inhibitors (eg, tacrolimus, pimecrolimus), calcipotriene, and narrow beam UVB therapy.
This patient had done reasonably well with cetirizine and triamcinolone in the past, so treatment was restarted. He was counseled regarding the nature and chronicity of his PN and told that if he could achieve symptom control and stop scratching the lesions, his condition might resolve.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Zeidler C, Yosipovitch G, Ständer S. Prurigo nodularis and its management. Dermatol Clin. 2018;36:189-197.
Zeidler C, Yosipovitch G, Ständer S. Prurigo nodularis and its management. Dermatol Clin. 2018;36:189-197.
EU panel review supports decision to pull Picato from market
Picato was cleared for marketing in the European Union in November 2012. The European Commission requested a safety review of the drug in September 2019 after data suggested a higher number of skin cancer cases, including cases of squamous cell carcinoma, in patients using it, as reported by Medscape Medical News.
In January 2020, use of Picato was suspended as a precaution while the PRAC review was underway. One month later, marketing authorization was withdrawn at the request of Leo Laboratories Ltd, which marketed the medicine.
The PRAC has now concluded its review of all available data on the risk for skin cancer in patients using Picato, including results of a study that compared Picato with imiquimod.
The review found “a higher occurrence of skin cancers, especially squamous cell carcinoma, in areas of skin treated with Picato than in areas treated with imiquimod,” the EMA said Friday in a news release.
“The committee also considered that Picato’s effectiveness is not maintained over time and noted that other treatment options are available for actinic keratosis,” the EMA said.
The agency recommends that patients who have used Picato watch for unusual skin changes or growths, which may occur weeks to months after use, and seek medical advice if any occur.
Picato continues to be available in the United States, although the US Food and Drug Administration is also looking into its safety and risks.
This article first appeared on Medscape.com.
Picato was cleared for marketing in the European Union in November 2012. The European Commission requested a safety review of the drug in September 2019 after data suggested a higher number of skin cancer cases, including cases of squamous cell carcinoma, in patients using it, as reported by Medscape Medical News.
In January 2020, use of Picato was suspended as a precaution while the PRAC review was underway. One month later, marketing authorization was withdrawn at the request of Leo Laboratories Ltd, which marketed the medicine.
The PRAC has now concluded its review of all available data on the risk for skin cancer in patients using Picato, including results of a study that compared Picato with imiquimod.
The review found “a higher occurrence of skin cancers, especially squamous cell carcinoma, in areas of skin treated with Picato than in areas treated with imiquimod,” the EMA said Friday in a news release.
“The committee also considered that Picato’s effectiveness is not maintained over time and noted that other treatment options are available for actinic keratosis,” the EMA said.
The agency recommends that patients who have used Picato watch for unusual skin changes or growths, which may occur weeks to months after use, and seek medical advice if any occur.
Picato continues to be available in the United States, although the US Food and Drug Administration is also looking into its safety and risks.
This article first appeared on Medscape.com.
Picato was cleared for marketing in the European Union in November 2012. The European Commission requested a safety review of the drug in September 2019 after data suggested a higher number of skin cancer cases, including cases of squamous cell carcinoma, in patients using it, as reported by Medscape Medical News.
In January 2020, use of Picato was suspended as a precaution while the PRAC review was underway. One month later, marketing authorization was withdrawn at the request of Leo Laboratories Ltd, which marketed the medicine.
The PRAC has now concluded its review of all available data on the risk for skin cancer in patients using Picato, including results of a study that compared Picato with imiquimod.
The review found “a higher occurrence of skin cancers, especially squamous cell carcinoma, in areas of skin treated with Picato than in areas treated with imiquimod,” the EMA said Friday in a news release.
“The committee also considered that Picato’s effectiveness is not maintained over time and noted that other treatment options are available for actinic keratosis,” the EMA said.
The agency recommends that patients who have used Picato watch for unusual skin changes or growths, which may occur weeks to months after use, and seek medical advice if any occur.
Picato continues to be available in the United States, although the US Food and Drug Administration is also looking into its safety and risks.
This article first appeared on Medscape.com.
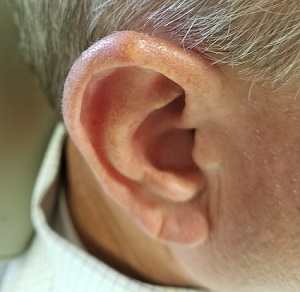
Lesion Has Been Giving Him an Earful
ANSWER
The correct answer is gouty tophus (choice “b”).
DISCUSSION
Gout is a defect of purine metabolism, usually caused by underexcretion of uric acid. Diet and heredity also play parts in gout’s development. Gouty tophi usually develop after years of hyperuricemia. As uric acid builds up in the bloodstream over time, it can then begin to be deposited into joints—most commonly the first metatarsal-phalangeal—as well as cartilage or even bones.
On further questioning, the patient recalled having been told on several occasions that his serum uric acid was elevated. In retrospect, his arthritis was most likely gouty in nature.
In terms of the differential, BCC (choice “a”) is common on helical rims, but it would not have contained the type of material found in this patient’s lesion. Also, it would not have waxed and waned as this lesion had done.
Epidermal cysts (choice “c”) can certainly come and go in prominence, but they are filled with a cheesy, pasty material—not the dry crystalline substance found in this lesion. Moreover, most epidermal cysts will have a small comedonal punctum over the center of the lesion. Dystrophic calcification (choice “d”) can mimic gouty tophi, but it is usually rough, firm, and fixed. It certainly would not be coming and going as it pleases.
TREATMENT
Surgical excision of the tophus was offered, but the patient was content with knowing the correct diagnosis. His PCP had previously explained therapeutic options—such as medication and dietary changes—that could address the overall problem. The patient elected to pursue treatment with his PCP.
ANSWER
The correct answer is gouty tophus (choice “b”).
DISCUSSION
Gout is a defect of purine metabolism, usually caused by underexcretion of uric acid. Diet and heredity also play parts in gout’s development. Gouty tophi usually develop after years of hyperuricemia. As uric acid builds up in the bloodstream over time, it can then begin to be deposited into joints—most commonly the first metatarsal-phalangeal—as well as cartilage or even bones.
On further questioning, the patient recalled having been told on several occasions that his serum uric acid was elevated. In retrospect, his arthritis was most likely gouty in nature.
In terms of the differential, BCC (choice “a”) is common on helical rims, but it would not have contained the type of material found in this patient’s lesion. Also, it would not have waxed and waned as this lesion had done.
Epidermal cysts (choice “c”) can certainly come and go in prominence, but they are filled with a cheesy, pasty material—not the dry crystalline substance found in this lesion. Moreover, most epidermal cysts will have a small comedonal punctum over the center of the lesion. Dystrophic calcification (choice “d”) can mimic gouty tophi, but it is usually rough, firm, and fixed. It certainly would not be coming and going as it pleases.
TREATMENT
Surgical excision of the tophus was offered, but the patient was content with knowing the correct diagnosis. His PCP had previously explained therapeutic options—such as medication and dietary changes—that could address the overall problem. The patient elected to pursue treatment with his PCP.
ANSWER
The correct answer is gouty tophus (choice “b”).
DISCUSSION
Gout is a defect of purine metabolism, usually caused by underexcretion of uric acid. Diet and heredity also play parts in gout’s development. Gouty tophi usually develop after years of hyperuricemia. As uric acid builds up in the bloodstream over time, it can then begin to be deposited into joints—most commonly the first metatarsal-phalangeal—as well as cartilage or even bones.
On further questioning, the patient recalled having been told on several occasions that his serum uric acid was elevated. In retrospect, his arthritis was most likely gouty in nature.
In terms of the differential, BCC (choice “a”) is common on helical rims, but it would not have contained the type of material found in this patient’s lesion. Also, it would not have waxed and waned as this lesion had done.
Epidermal cysts (choice “c”) can certainly come and go in prominence, but they are filled with a cheesy, pasty material—not the dry crystalline substance found in this lesion. Moreover, most epidermal cysts will have a small comedonal punctum over the center of the lesion. Dystrophic calcification (choice “d”) can mimic gouty tophi, but it is usually rough, firm, and fixed. It certainly would not be coming and going as it pleases.
TREATMENT
Surgical excision of the tophus was offered, but the patient was content with knowing the correct diagnosis. His PCP had previously explained therapeutic options—such as medication and dietary changes—that could address the overall problem. The patient elected to pursue treatment with his PCP.
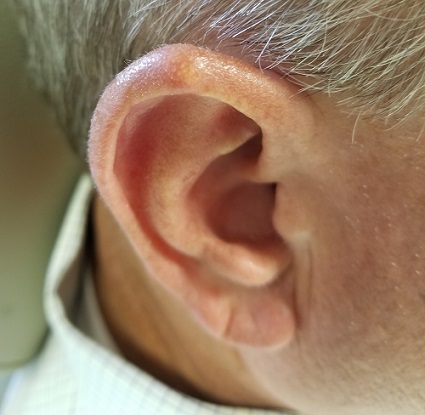
Over the years, the lesion on this 49-year-old man’s right ear has waxed and waned in prominence. Although it never causes pain, its unrelenting existence coupled with a history of basal cell carcinoma (BCC) on his face has caused him to worry. He has had no other lesions and is in otherwise good health, except for occasional bouts of arthritis, for which he takes ibuprofen, with good results.
While his family had suggested that the lesion could be a cyst, his primary care provider (PCP) disagreed and referred him to dermatology.
A 5-mm firm papule is located at the right helical crest. The lesion is skin-colored, with no redness, and nontender on palpation, although it is moderately firm and mobile. No punctum is noted.
The surrounding skin has no signs of sun damage, although the patient is quite fair. As a young man, he had far too much sun exposure, burning easily and tanning only with difficulty.
After consultation with the patient, the decision is made to incise the lesion using a #11 blade to examine its contents. The incision reveals a dry crystalline substance, which is easily cleared with brief curettage. This effectively leads to a flattening of the lesion.
Protean manifestations of COVID-19: “Our ignorance is profound”
Although a cause-and-effect relationship is unknown, people with the virus have presented with or developed heart disease, acute liver injury, ongoing GI issues, skin manifestations, neurologic damage, and other problems, especially among sicker people.
For example, French physicians described an association with encephalopathy, agitation, confusion, and corticospinal tract signs among 58 people hospitalized with acute respiratory distress (N Engl J Med. 2020 Apr 15. doi: 10.1056/NEJMc2008597).
In particular, Yale New Haven (Conn.) Hospital is dealing with unexpected complications up close. Almost half of the beds there are occupied by COVID-19 patients. Over 100 people are in the ICU, and almost 70 intubated. Of the more than 750 COVID admissions so far, only about 350 have been discharged. “Even in a bad flu season, you never see something like this; it’s just unheard of,” said Harlan Krumholz, MD, a Yale cardiologist and professor of medicine helping lead the efforts there.
Kidney injuries prominent
“When they get to the ICU, we are seeing lots of people with acute kidney injuries; lots of people developing endocrine problems; people having blood sugar control issues, coagulation issues, blood clots. We are just waking up to the wide range of ways this virus can affect people. Our ignorance is profound,” Dr. Krumholz said, but physicians “recognize that this thing has the capability of attacking almost every single organ system, and it may or may not present with respiratory symptoms.”
It’s a similar story at Mt. Sinai South Nassau, a hospital in Oceanside, N.Y. “We’ve seen a lot of renal injury in people having complications, a lot of acute dialysis,” but it’s unclear how much is caused by the virus and how much is simply because people are so sick, said Aaron Glatt, MD, infectious disease professor and chair of medicine at the hospital. However, he said things are looking brighter than at Yale.
“We are not seeing the same level of increase in cases that we had previously, and we are starting to see extubations and discharges. We’ve treated a number of patients with plasma therapy, and hopefully that will be of benefit. We’ve seen some response to” the immunosuppressive “tocilizumab [Actemra], and a lot of response to very good respiratory therapy. I think we are starting to flatten the curve,” Dr. Glatt said.
“Look for tricky symptoms”
The growing awareness of COVID’s protean manifestations is evident in Medscape’s Consult forum, an online community where physicians and medical students share information and seek advice; there’s been over 200 COVID-19 cases and questions since January.
Early on, traffic was mostly about typical pulmonary presentations, but lately it’s shifted to nonrespiratory involvement. Physicians want to know if what they are seeing is related to the virus, and if other people are seeing the same things.
There’s a case on Consult of a 37-year-old man with stomach pain, vomiting, and diarrhea, but no respiratory symptoms and a positive COVID test. A chest CT incidental to his abdominal scan revealed significant bilateral lung involvement.
A 69-year-old woman with a history of laparotomy and new onset intestinal subocclusion had only adhesions on a subsequent exploratory laparotomy, and was doing okay otherwise. She suddenly went into respiratory failure with progressive bradycardia and died 3 days later. Aspiration pneumonia, pulmonary embolism, and MI had been ruled out. “The pattern of cardiovascular failure was in favor of myocarditis, but we don’t have any other clue,” the physician said after describing a second similar case.
Another doctor on the forum reported elevated cardiac enzymes without coronary artery obstruction in a positive patient who went into shock, with an ejection fraction of 40% and markedly increased heart wall thickness, but no lung involvement. There are also two cases of idiopathic thrombocytopenia without fever of hypoxia.
An Italian gastroenterologist said: “Look for tricky symptoms.” Expand “patient history, asking about the sudden occurrence of dysgeusia and/or anosmia. These symptoms have become my guiding diagnostic light” in Verona. “Most patients become nauseated, [and] the taste of any food is unbearable. When I find these symptoms by history, the patient is COVID positive 100%.”
‘Make sure that they didn’t die in vain’
There was interest in those and other reports on Consult, and comments from physicians who have theories, but no certain answers about what is, and is not, caused by the virus.
Direct viral attack is likely a part of it, said Stanley Perlman, MD, PhD, a professor of microbiology and immunology at the University of Iowa, Iowa City.
The ACE2 receptor the virus uses to enter cells is common in many organs, plus there were extrapulmonary manifestations with severe acute respiratory syndrome (SARS), another pandemic caused by a zoonotic coronavirus almost 20 years ago. At least with SARS, “many organs were infected when examined at autopsy,” he said.
The body’s inflammatory response is almost certainly also in play. Progressive derangements in inflammatory markers – C-reactive protein, D-dimer, ferritin – correlate with worse prognosis, and “the cytokine storm that occurs in these patients can lead to a degree of encephalopathy, myocarditis, liver impairment, and kidney impairment; multiorgan dysfunction, in other words,” said William Shaffner, MD, a professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
But in some cases, the virus might simply be a bystander to an unrelated disease process; in others, the experimental treatments being used might cause problems. Indeed, cardiology groups recently warned of torsade de pointes – a dangerously abnormal heart rhythm – with hydroxychloroquine and azithromycin.
“We think it’s some combination,” but don’t really know, Dr. Krumholz said. In the meantime, “we are forced to treat patients by instinct and first principles,” and long-term sequelae are unknown. “We don’t want to be in this position for long.”
To that end, he said, “this is the time for us all to hold hands and be together because we need to learn rapidly from each other. Our job is both to care for the people in front of us and make sure that they didn’t die in vain, that the experience they had is funneled into a larger set of data to make sure the next person is better off.”
Although a cause-and-effect relationship is unknown, people with the virus have presented with or developed heart disease, acute liver injury, ongoing GI issues, skin manifestations, neurologic damage, and other problems, especially among sicker people.
For example, French physicians described an association with encephalopathy, agitation, confusion, and corticospinal tract signs among 58 people hospitalized with acute respiratory distress (N Engl J Med. 2020 Apr 15. doi: 10.1056/NEJMc2008597).
In particular, Yale New Haven (Conn.) Hospital is dealing with unexpected complications up close. Almost half of the beds there are occupied by COVID-19 patients. Over 100 people are in the ICU, and almost 70 intubated. Of the more than 750 COVID admissions so far, only about 350 have been discharged. “Even in a bad flu season, you never see something like this; it’s just unheard of,” said Harlan Krumholz, MD, a Yale cardiologist and professor of medicine helping lead the efforts there.
Kidney injuries prominent
“When they get to the ICU, we are seeing lots of people with acute kidney injuries; lots of people developing endocrine problems; people having blood sugar control issues, coagulation issues, blood clots. We are just waking up to the wide range of ways this virus can affect people. Our ignorance is profound,” Dr. Krumholz said, but physicians “recognize that this thing has the capability of attacking almost every single organ system, and it may or may not present with respiratory symptoms.”
It’s a similar story at Mt. Sinai South Nassau, a hospital in Oceanside, N.Y. “We’ve seen a lot of renal injury in people having complications, a lot of acute dialysis,” but it’s unclear how much is caused by the virus and how much is simply because people are so sick, said Aaron Glatt, MD, infectious disease professor and chair of medicine at the hospital. However, he said things are looking brighter than at Yale.
“We are not seeing the same level of increase in cases that we had previously, and we are starting to see extubations and discharges. We’ve treated a number of patients with plasma therapy, and hopefully that will be of benefit. We’ve seen some response to” the immunosuppressive “tocilizumab [Actemra], and a lot of response to very good respiratory therapy. I think we are starting to flatten the curve,” Dr. Glatt said.
“Look for tricky symptoms”
The growing awareness of COVID’s protean manifestations is evident in Medscape’s Consult forum, an online community where physicians and medical students share information and seek advice; there’s been over 200 COVID-19 cases and questions since January.
Early on, traffic was mostly about typical pulmonary presentations, but lately it’s shifted to nonrespiratory involvement. Physicians want to know if what they are seeing is related to the virus, and if other people are seeing the same things.
There’s a case on Consult of a 37-year-old man with stomach pain, vomiting, and diarrhea, but no respiratory symptoms and a positive COVID test. A chest CT incidental to his abdominal scan revealed significant bilateral lung involvement.
A 69-year-old woman with a history of laparotomy and new onset intestinal subocclusion had only adhesions on a subsequent exploratory laparotomy, and was doing okay otherwise. She suddenly went into respiratory failure with progressive bradycardia and died 3 days later. Aspiration pneumonia, pulmonary embolism, and MI had been ruled out. “The pattern of cardiovascular failure was in favor of myocarditis, but we don’t have any other clue,” the physician said after describing a second similar case.
Another doctor on the forum reported elevated cardiac enzymes without coronary artery obstruction in a positive patient who went into shock, with an ejection fraction of 40% and markedly increased heart wall thickness, but no lung involvement. There are also two cases of idiopathic thrombocytopenia without fever of hypoxia.
An Italian gastroenterologist said: “Look for tricky symptoms.” Expand “patient history, asking about the sudden occurrence of dysgeusia and/or anosmia. These symptoms have become my guiding diagnostic light” in Verona. “Most patients become nauseated, [and] the taste of any food is unbearable. When I find these symptoms by history, the patient is COVID positive 100%.”
‘Make sure that they didn’t die in vain’
There was interest in those and other reports on Consult, and comments from physicians who have theories, but no certain answers about what is, and is not, caused by the virus.
Direct viral attack is likely a part of it, said Stanley Perlman, MD, PhD, a professor of microbiology and immunology at the University of Iowa, Iowa City.
The ACE2 receptor the virus uses to enter cells is common in many organs, plus there were extrapulmonary manifestations with severe acute respiratory syndrome (SARS), another pandemic caused by a zoonotic coronavirus almost 20 years ago. At least with SARS, “many organs were infected when examined at autopsy,” he said.
The body’s inflammatory response is almost certainly also in play. Progressive derangements in inflammatory markers – C-reactive protein, D-dimer, ferritin – correlate with worse prognosis, and “the cytokine storm that occurs in these patients can lead to a degree of encephalopathy, myocarditis, liver impairment, and kidney impairment; multiorgan dysfunction, in other words,” said William Shaffner, MD, a professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
But in some cases, the virus might simply be a bystander to an unrelated disease process; in others, the experimental treatments being used might cause problems. Indeed, cardiology groups recently warned of torsade de pointes – a dangerously abnormal heart rhythm – with hydroxychloroquine and azithromycin.
“We think it’s some combination,” but don’t really know, Dr. Krumholz said. In the meantime, “we are forced to treat patients by instinct and first principles,” and long-term sequelae are unknown. “We don’t want to be in this position for long.”
To that end, he said, “this is the time for us all to hold hands and be together because we need to learn rapidly from each other. Our job is both to care for the people in front of us and make sure that they didn’t die in vain, that the experience they had is funneled into a larger set of data to make sure the next person is better off.”
Although a cause-and-effect relationship is unknown, people with the virus have presented with or developed heart disease, acute liver injury, ongoing GI issues, skin manifestations, neurologic damage, and other problems, especially among sicker people.
For example, French physicians described an association with encephalopathy, agitation, confusion, and corticospinal tract signs among 58 people hospitalized with acute respiratory distress (N Engl J Med. 2020 Apr 15. doi: 10.1056/NEJMc2008597).
In particular, Yale New Haven (Conn.) Hospital is dealing with unexpected complications up close. Almost half of the beds there are occupied by COVID-19 patients. Over 100 people are in the ICU, and almost 70 intubated. Of the more than 750 COVID admissions so far, only about 350 have been discharged. “Even in a bad flu season, you never see something like this; it’s just unheard of,” said Harlan Krumholz, MD, a Yale cardiologist and professor of medicine helping lead the efforts there.
Kidney injuries prominent
“When they get to the ICU, we are seeing lots of people with acute kidney injuries; lots of people developing endocrine problems; people having blood sugar control issues, coagulation issues, blood clots. We are just waking up to the wide range of ways this virus can affect people. Our ignorance is profound,” Dr. Krumholz said, but physicians “recognize that this thing has the capability of attacking almost every single organ system, and it may or may not present with respiratory symptoms.”
It’s a similar story at Mt. Sinai South Nassau, a hospital in Oceanside, N.Y. “We’ve seen a lot of renal injury in people having complications, a lot of acute dialysis,” but it’s unclear how much is caused by the virus and how much is simply because people are so sick, said Aaron Glatt, MD, infectious disease professor and chair of medicine at the hospital. However, he said things are looking brighter than at Yale.
“We are not seeing the same level of increase in cases that we had previously, and we are starting to see extubations and discharges. We’ve treated a number of patients with plasma therapy, and hopefully that will be of benefit. We’ve seen some response to” the immunosuppressive “tocilizumab [Actemra], and a lot of response to very good respiratory therapy. I think we are starting to flatten the curve,” Dr. Glatt said.
“Look for tricky symptoms”
The growing awareness of COVID’s protean manifestations is evident in Medscape’s Consult forum, an online community where physicians and medical students share information and seek advice; there’s been over 200 COVID-19 cases and questions since January.
Early on, traffic was mostly about typical pulmonary presentations, but lately it’s shifted to nonrespiratory involvement. Physicians want to know if what they are seeing is related to the virus, and if other people are seeing the same things.
There’s a case on Consult of a 37-year-old man with stomach pain, vomiting, and diarrhea, but no respiratory symptoms and a positive COVID test. A chest CT incidental to his abdominal scan revealed significant bilateral lung involvement.
A 69-year-old woman with a history of laparotomy and new onset intestinal subocclusion had only adhesions on a subsequent exploratory laparotomy, and was doing okay otherwise. She suddenly went into respiratory failure with progressive bradycardia and died 3 days later. Aspiration pneumonia, pulmonary embolism, and MI had been ruled out. “The pattern of cardiovascular failure was in favor of myocarditis, but we don’t have any other clue,” the physician said after describing a second similar case.
Another doctor on the forum reported elevated cardiac enzymes without coronary artery obstruction in a positive patient who went into shock, with an ejection fraction of 40% and markedly increased heart wall thickness, but no lung involvement. There are also two cases of idiopathic thrombocytopenia without fever of hypoxia.
An Italian gastroenterologist said: “Look for tricky symptoms.” Expand “patient history, asking about the sudden occurrence of dysgeusia and/or anosmia. These symptoms have become my guiding diagnostic light” in Verona. “Most patients become nauseated, [and] the taste of any food is unbearable. When I find these symptoms by history, the patient is COVID positive 100%.”
‘Make sure that they didn’t die in vain’
There was interest in those and other reports on Consult, and comments from physicians who have theories, but no certain answers about what is, and is not, caused by the virus.
Direct viral attack is likely a part of it, said Stanley Perlman, MD, PhD, a professor of microbiology and immunology at the University of Iowa, Iowa City.
The ACE2 receptor the virus uses to enter cells is common in many organs, plus there were extrapulmonary manifestations with severe acute respiratory syndrome (SARS), another pandemic caused by a zoonotic coronavirus almost 20 years ago. At least with SARS, “many organs were infected when examined at autopsy,” he said.
The body’s inflammatory response is almost certainly also in play. Progressive derangements in inflammatory markers – C-reactive protein, D-dimer, ferritin – correlate with worse prognosis, and “the cytokine storm that occurs in these patients can lead to a degree of encephalopathy, myocarditis, liver impairment, and kidney impairment; multiorgan dysfunction, in other words,” said William Shaffner, MD, a professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
But in some cases, the virus might simply be a bystander to an unrelated disease process; in others, the experimental treatments being used might cause problems. Indeed, cardiology groups recently warned of torsade de pointes – a dangerously abnormal heart rhythm – with hydroxychloroquine and azithromycin.
“We think it’s some combination,” but don’t really know, Dr. Krumholz said. In the meantime, “we are forced to treat patients by instinct and first principles,” and long-term sequelae are unknown. “We don’t want to be in this position for long.”
To that end, he said, “this is the time for us all to hold hands and be together because we need to learn rapidly from each other. Our job is both to care for the people in front of us and make sure that they didn’t die in vain, that the experience they had is funneled into a larger set of data to make sure the next person is better off.”
Financial incentives affect the adoption of biosimilars
during the same time period in 2015-2019, according to an analysis published in Arthritis and Rheumatology.
The use of the biosimilars also was associated with cost savings at the VAMC, but not at the academic medical center, which illustrates that insufficient financial incentives can delay the adoption of biosimilars and the health care system’s realization of cost savings, according to the authors.
Medicare, which is not allowed to negotiate drug prices, is one of the largest payers for infused therapies. Medicare reimbursement for infused therapies is based on the latter’s average selling price (ASP) during the previous quarter. Institutions may negotiate purchase prices with drug manufacturers and receive Medicare reimbursement. Biosimilars generally have lower ASPs than their corresponding reference therapies, and biosimilar manufacturers may have less room to negotiate prices than reference therapy manufacturers. Consequently, a given institution might have a greater incentive to use reference products than to use biosimilars.
An examination of pharmacy data
The VA negotiates drug prices for all of its medical centers and has mandated that clinicians prefer biosimilars to their corresponding reference therapies, so Joshua F. Baker, MD, of the University of Pennsylvania and the Corporal Michael J. Crescenz VAMC, both in Philadelphia, and his colleagues hypothesized that the adoption of biosimilars had proceeded more quickly at a VAMC than at a nearby academic medical center.
The investigators examined pharmacy data from the University of Pennsylvania Health System (UPHS) electronic medical record and the Corporal Michael J. Crescenz VAMC to compare the frequency of prescribing biosimilars at these sites between Jan. 1, 2015, and May 31, 2019. Dr. Baker and his associates focused specifically on reference infliximab (Remicade) and the reference noninfusion therapies filgrastim (Neupogen) and pegfilgrastim (Neulasta) and on biosimilars of these therapies (infliximab-dyyb [Inflectra], infliximab-abda [Renflexis], filgrastim-sndz [Zarxio], and pegfilgrastim-jmdb [Fulphila]).
Because Medicare was the predominant payer, the researchers estimated reimbursement for reference and biosimilar infliximabs according to the Medicare Part B reimbursement policy. They defined an institution’s incentive to use a given therapy as the difference between the reimbursement and acquisition cost for that therapy. Dr. Baker and colleagues compared the incentives for UPHS with those for the VAMC.
VAMC saved 81% of reference product cost
The researchers identified 15,761 infusions of infliximab at UPHS and 446 at the VAMC during the study period. The proportion of infusions that used the reference product was 99% at UPHS and 62% at the VAMC. ASPs for biosimilar infliximab have been consistently lower than those for the reference product since July 2017. In December 2017, the VAMC switched to the biosimilar infliximab.
Institutional incentives based on Medicare Part B reimbursement and acquisitions costs for reference and biosimilar infliximab have been similar since 2018. In 2019, the institutional incentive favored the reference product by $49-$64 per 100-mg vial. But at the VAMC, the cost per 100-mg vial was $623.48 for the reference product and $115.58 for the biosimilar Renflexis. Purchasing the biosimilar thus yielded a savings of 81%. The current costs for the therapies are $546 and $116, respectively.
In addition, Dr. Baker and colleagues identified 46,683 orders for filgrastim or pegfilgrastim at UPHS. Approximately 90% of the orders were for either of the two reference products despite the ASP of biosimilar filgrastim being approximately 40% lower than that of its reference product. At the VAMC, about 88% of orders were for the reference products. Biosimilars became available in 2016. UPHS began using them at a modest rate, but their adoption was greater at the VAMC, which designated them as preferred products.
Tendering and a nationwide policy mandating use of biosimilars have resulted in financial savings for the VAMC, wrote Dr. Baker and colleagues. “These data suggest that, with current Medicare Part B reimbursement policy, the absence of financial incentives to encourage use of infliximab biosimilars has resulted in slower uptake of biosimilar use at institutions outside of the VA system. The implications of this are a slower reduction in costs to the health care system, since decreases in ASP over time are predicated on negotiations at the institutional level, which have been gradual and stepwise. ...
“Although some of our results may not be applicable to other geographical regions of the U.S., the comparison of two affiliated institutions in geographical proximity and with shared health care providers is a strength,” they continued. “Our findings should be replicated using national VAMC data or data from other health care systems.”
The researchers said that their findings may not apply to noninfused therapies, which are not covered under Medicare Part B, and they did not directly study the impact of pharmacy benefit managers. However, they noted that their data on filgrastim and pegfilgrastim support the hypothesis that pharmacy benefit managers receive “incentives that continue to promote the use of reference products that have higher manufacturer’s list prices, which likely will limit the uptake of both infused and injectable biosimilar therapies over time.” They said that “this finding has important implications for when noninfused biosimilars (e.g. etanercept and adalimumab) are eventually introduced to the U.S. market.”
European governments incentivize use of biosimilars
Government and institutional incentives have increased the adoption of biosimilars in Europe, wrote Guro Lovik Goll, MD, and Tore Kristian Kvien, MD, of the department of rheumatology at Diakonhjemmet Hospital in Oslo, in an accompanying editorial. Norway and Denmark have annual national tender systems in which biosimilars and reference products compete. The price of infliximab biosimilar was 39% lower than the reference product in 2014 and 69% lower in 2015. “Competition has caused dramatically lower prices both for biosimilars and also for the originator drugs competing with them,” wrote the authors.
In 2015, the government of Denmark mandated that patients on infliximab be switched to a biosimilar, and patients in Norway also have been switched to biosimilars. The use of etanercept in Norway increased by 40% from 2016 to 2019, and the use of infliximab has increased by more than threefold since 2015. “In Norway, the consequence of competition, national tenders, and availability of biosimilars have led to better access to therapy for more people in need of biologic drugs, while at the same time showing a total cost reduction of biologics for use in rheumatology, gastroenterology, and dermatology,” wrote the authors.
Health care costs $10,000 per capita in the United States, compared with $5,300 for other wealthy countries in the Organization for Economic Cooperation and Development. Low life expectancy and high infant mortality in the U.S. indicate that high costs are not associated with better outcomes. “As Americans seem to lose out on the cost-cutting potential of biosimilars, this missed opportunity is set to get even more expensive,” the authors concluded.
The U.S. Department of Veterans Affairs, the National Institutes of Health, and the American Diabetes Association contributed funding for the study. Dr. Baker reported receiving consulting fees from Bristol-Myers Squibb and Gilead, and another author reported receiving research support paid to his institution by Pfizer and UCB, as well as receiving consulting fees from nine pharmaceutical companies. Dr. Goll and Dr. Kvien both reported receiving fees for speaking and/or consulting from numerous pharmaceutical companies, including Pfizer.
SOURCES: Baker JF et al. Arthritis Rheumatol. 2020 Apr 6. doi: 10.1002/art.41277.
during the same time period in 2015-2019, according to an analysis published in Arthritis and Rheumatology.
The use of the biosimilars also was associated with cost savings at the VAMC, but not at the academic medical center, which illustrates that insufficient financial incentives can delay the adoption of biosimilars and the health care system’s realization of cost savings, according to the authors.
Medicare, which is not allowed to negotiate drug prices, is one of the largest payers for infused therapies. Medicare reimbursement for infused therapies is based on the latter’s average selling price (ASP) during the previous quarter. Institutions may negotiate purchase prices with drug manufacturers and receive Medicare reimbursement. Biosimilars generally have lower ASPs than their corresponding reference therapies, and biosimilar manufacturers may have less room to negotiate prices than reference therapy manufacturers. Consequently, a given institution might have a greater incentive to use reference products than to use biosimilars.
An examination of pharmacy data
The VA negotiates drug prices for all of its medical centers and has mandated that clinicians prefer biosimilars to their corresponding reference therapies, so Joshua F. Baker, MD, of the University of Pennsylvania and the Corporal Michael J. Crescenz VAMC, both in Philadelphia, and his colleagues hypothesized that the adoption of biosimilars had proceeded more quickly at a VAMC than at a nearby academic medical center.
The investigators examined pharmacy data from the University of Pennsylvania Health System (UPHS) electronic medical record and the Corporal Michael J. Crescenz VAMC to compare the frequency of prescribing biosimilars at these sites between Jan. 1, 2015, and May 31, 2019. Dr. Baker and his associates focused specifically on reference infliximab (Remicade) and the reference noninfusion therapies filgrastim (Neupogen) and pegfilgrastim (Neulasta) and on biosimilars of these therapies (infliximab-dyyb [Inflectra], infliximab-abda [Renflexis], filgrastim-sndz [Zarxio], and pegfilgrastim-jmdb [Fulphila]).
Because Medicare was the predominant payer, the researchers estimated reimbursement for reference and biosimilar infliximabs according to the Medicare Part B reimbursement policy. They defined an institution’s incentive to use a given therapy as the difference between the reimbursement and acquisition cost for that therapy. Dr. Baker and colleagues compared the incentives for UPHS with those for the VAMC.
VAMC saved 81% of reference product cost
The researchers identified 15,761 infusions of infliximab at UPHS and 446 at the VAMC during the study period. The proportion of infusions that used the reference product was 99% at UPHS and 62% at the VAMC. ASPs for biosimilar infliximab have been consistently lower than those for the reference product since July 2017. In December 2017, the VAMC switched to the biosimilar infliximab.
Institutional incentives based on Medicare Part B reimbursement and acquisitions costs for reference and biosimilar infliximab have been similar since 2018. In 2019, the institutional incentive favored the reference product by $49-$64 per 100-mg vial. But at the VAMC, the cost per 100-mg vial was $623.48 for the reference product and $115.58 for the biosimilar Renflexis. Purchasing the biosimilar thus yielded a savings of 81%. The current costs for the therapies are $546 and $116, respectively.
In addition, Dr. Baker and colleagues identified 46,683 orders for filgrastim or pegfilgrastim at UPHS. Approximately 90% of the orders were for either of the two reference products despite the ASP of biosimilar filgrastim being approximately 40% lower than that of its reference product. At the VAMC, about 88% of orders were for the reference products. Biosimilars became available in 2016. UPHS began using them at a modest rate, but their adoption was greater at the VAMC, which designated them as preferred products.
Tendering and a nationwide policy mandating use of biosimilars have resulted in financial savings for the VAMC, wrote Dr. Baker and colleagues. “These data suggest that, with current Medicare Part B reimbursement policy, the absence of financial incentives to encourage use of infliximab biosimilars has resulted in slower uptake of biosimilar use at institutions outside of the VA system. The implications of this are a slower reduction in costs to the health care system, since decreases in ASP over time are predicated on negotiations at the institutional level, which have been gradual and stepwise. ...
“Although some of our results may not be applicable to other geographical regions of the U.S., the comparison of two affiliated institutions in geographical proximity and with shared health care providers is a strength,” they continued. “Our findings should be replicated using national VAMC data or data from other health care systems.”
The researchers said that their findings may not apply to noninfused therapies, which are not covered under Medicare Part B, and they did not directly study the impact of pharmacy benefit managers. However, they noted that their data on filgrastim and pegfilgrastim support the hypothesis that pharmacy benefit managers receive “incentives that continue to promote the use of reference products that have higher manufacturer’s list prices, which likely will limit the uptake of both infused and injectable biosimilar therapies over time.” They said that “this finding has important implications for when noninfused biosimilars (e.g. etanercept and adalimumab) are eventually introduced to the U.S. market.”
European governments incentivize use of biosimilars
Government and institutional incentives have increased the adoption of biosimilars in Europe, wrote Guro Lovik Goll, MD, and Tore Kristian Kvien, MD, of the department of rheumatology at Diakonhjemmet Hospital in Oslo, in an accompanying editorial. Norway and Denmark have annual national tender systems in which biosimilars and reference products compete. The price of infliximab biosimilar was 39% lower than the reference product in 2014 and 69% lower in 2015. “Competition has caused dramatically lower prices both for biosimilars and also for the originator drugs competing with them,” wrote the authors.
In 2015, the government of Denmark mandated that patients on infliximab be switched to a biosimilar, and patients in Norway also have been switched to biosimilars. The use of etanercept in Norway increased by 40% from 2016 to 2019, and the use of infliximab has increased by more than threefold since 2015. “In Norway, the consequence of competition, national tenders, and availability of biosimilars have led to better access to therapy for more people in need of biologic drugs, while at the same time showing a total cost reduction of biologics for use in rheumatology, gastroenterology, and dermatology,” wrote the authors.
Health care costs $10,000 per capita in the United States, compared with $5,300 for other wealthy countries in the Organization for Economic Cooperation and Development. Low life expectancy and high infant mortality in the U.S. indicate that high costs are not associated with better outcomes. “As Americans seem to lose out on the cost-cutting potential of biosimilars, this missed opportunity is set to get even more expensive,” the authors concluded.
The U.S. Department of Veterans Affairs, the National Institutes of Health, and the American Diabetes Association contributed funding for the study. Dr. Baker reported receiving consulting fees from Bristol-Myers Squibb and Gilead, and another author reported receiving research support paid to his institution by Pfizer and UCB, as well as receiving consulting fees from nine pharmaceutical companies. Dr. Goll and Dr. Kvien both reported receiving fees for speaking and/or consulting from numerous pharmaceutical companies, including Pfizer.
SOURCES: Baker JF et al. Arthritis Rheumatol. 2020 Apr 6. doi: 10.1002/art.41277.
during the same time period in 2015-2019, according to an analysis published in Arthritis and Rheumatology.
The use of the biosimilars also was associated with cost savings at the VAMC, but not at the academic medical center, which illustrates that insufficient financial incentives can delay the adoption of biosimilars and the health care system’s realization of cost savings, according to the authors.
Medicare, which is not allowed to negotiate drug prices, is one of the largest payers for infused therapies. Medicare reimbursement for infused therapies is based on the latter’s average selling price (ASP) during the previous quarter. Institutions may negotiate purchase prices with drug manufacturers and receive Medicare reimbursement. Biosimilars generally have lower ASPs than their corresponding reference therapies, and biosimilar manufacturers may have less room to negotiate prices than reference therapy manufacturers. Consequently, a given institution might have a greater incentive to use reference products than to use biosimilars.
An examination of pharmacy data
The VA negotiates drug prices for all of its medical centers and has mandated that clinicians prefer biosimilars to their corresponding reference therapies, so Joshua F. Baker, MD, of the University of Pennsylvania and the Corporal Michael J. Crescenz VAMC, both in Philadelphia, and his colleagues hypothesized that the adoption of biosimilars had proceeded more quickly at a VAMC than at a nearby academic medical center.
The investigators examined pharmacy data from the University of Pennsylvania Health System (UPHS) electronic medical record and the Corporal Michael J. Crescenz VAMC to compare the frequency of prescribing biosimilars at these sites between Jan. 1, 2015, and May 31, 2019. Dr. Baker and his associates focused specifically on reference infliximab (Remicade) and the reference noninfusion therapies filgrastim (Neupogen) and pegfilgrastim (Neulasta) and on biosimilars of these therapies (infliximab-dyyb [Inflectra], infliximab-abda [Renflexis], filgrastim-sndz [Zarxio], and pegfilgrastim-jmdb [Fulphila]).
Because Medicare was the predominant payer, the researchers estimated reimbursement for reference and biosimilar infliximabs according to the Medicare Part B reimbursement policy. They defined an institution’s incentive to use a given therapy as the difference between the reimbursement and acquisition cost for that therapy. Dr. Baker and colleagues compared the incentives for UPHS with those for the VAMC.
VAMC saved 81% of reference product cost
The researchers identified 15,761 infusions of infliximab at UPHS and 446 at the VAMC during the study period. The proportion of infusions that used the reference product was 99% at UPHS and 62% at the VAMC. ASPs for biosimilar infliximab have been consistently lower than those for the reference product since July 2017. In December 2017, the VAMC switched to the biosimilar infliximab.
Institutional incentives based on Medicare Part B reimbursement and acquisitions costs for reference and biosimilar infliximab have been similar since 2018. In 2019, the institutional incentive favored the reference product by $49-$64 per 100-mg vial. But at the VAMC, the cost per 100-mg vial was $623.48 for the reference product and $115.58 for the biosimilar Renflexis. Purchasing the biosimilar thus yielded a savings of 81%. The current costs for the therapies are $546 and $116, respectively.
In addition, Dr. Baker and colleagues identified 46,683 orders for filgrastim or pegfilgrastim at UPHS. Approximately 90% of the orders were for either of the two reference products despite the ASP of biosimilar filgrastim being approximately 40% lower than that of its reference product. At the VAMC, about 88% of orders were for the reference products. Biosimilars became available in 2016. UPHS began using them at a modest rate, but their adoption was greater at the VAMC, which designated them as preferred products.
Tendering and a nationwide policy mandating use of biosimilars have resulted in financial savings for the VAMC, wrote Dr. Baker and colleagues. “These data suggest that, with current Medicare Part B reimbursement policy, the absence of financial incentives to encourage use of infliximab biosimilars has resulted in slower uptake of biosimilar use at institutions outside of the VA system. The implications of this are a slower reduction in costs to the health care system, since decreases in ASP over time are predicated on negotiations at the institutional level, which have been gradual and stepwise. ...
“Although some of our results may not be applicable to other geographical regions of the U.S., the comparison of two affiliated institutions in geographical proximity and with shared health care providers is a strength,” they continued. “Our findings should be replicated using national VAMC data or data from other health care systems.”
The researchers said that their findings may not apply to noninfused therapies, which are not covered under Medicare Part B, and they did not directly study the impact of pharmacy benefit managers. However, they noted that their data on filgrastim and pegfilgrastim support the hypothesis that pharmacy benefit managers receive “incentives that continue to promote the use of reference products that have higher manufacturer’s list prices, which likely will limit the uptake of both infused and injectable biosimilar therapies over time.” They said that “this finding has important implications for when noninfused biosimilars (e.g. etanercept and adalimumab) are eventually introduced to the U.S. market.”
European governments incentivize use of biosimilars
Government and institutional incentives have increased the adoption of biosimilars in Europe, wrote Guro Lovik Goll, MD, and Tore Kristian Kvien, MD, of the department of rheumatology at Diakonhjemmet Hospital in Oslo, in an accompanying editorial. Norway and Denmark have annual national tender systems in which biosimilars and reference products compete. The price of infliximab biosimilar was 39% lower than the reference product in 2014 and 69% lower in 2015. “Competition has caused dramatically lower prices both for biosimilars and also for the originator drugs competing with them,” wrote the authors.
In 2015, the government of Denmark mandated that patients on infliximab be switched to a biosimilar, and patients in Norway also have been switched to biosimilars. The use of etanercept in Norway increased by 40% from 2016 to 2019, and the use of infliximab has increased by more than threefold since 2015. “In Norway, the consequence of competition, national tenders, and availability of biosimilars have led to better access to therapy for more people in need of biologic drugs, while at the same time showing a total cost reduction of biologics for use in rheumatology, gastroenterology, and dermatology,” wrote the authors.
Health care costs $10,000 per capita in the United States, compared with $5,300 for other wealthy countries in the Organization for Economic Cooperation and Development. Low life expectancy and high infant mortality in the U.S. indicate that high costs are not associated with better outcomes. “As Americans seem to lose out on the cost-cutting potential of biosimilars, this missed opportunity is set to get even more expensive,” the authors concluded.
The U.S. Department of Veterans Affairs, the National Institutes of Health, and the American Diabetes Association contributed funding for the study. Dr. Baker reported receiving consulting fees from Bristol-Myers Squibb and Gilead, and another author reported receiving research support paid to his institution by Pfizer and UCB, as well as receiving consulting fees from nine pharmaceutical companies. Dr. Goll and Dr. Kvien both reported receiving fees for speaking and/or consulting from numerous pharmaceutical companies, including Pfizer.
SOURCES: Baker JF et al. Arthritis Rheumatol. 2020 Apr 6. doi: 10.1002/art.41277.
FROM ARTHRITIS & RHEUMATOLOGY
COVID-19 PPE-related skin effects described in survey of Chinese doctors, nurses
Almost 75% of doctors and nurses in and around Wuhan, China, where the outbreak first emerged, reported skin problems during a single week in early February 2020, in a survey of health care workers (HCW) caring for COVID-19 patients at five university and five regional hospitals. Hands, cheeks, and the nasal bridge were the most commonly affected areas, with skin dryness, maceration, papules, and erythema the most common problems, according to research published in the British Journal of Dermatology.
In New York City, masks in particular are “really an issue,” said Ellen Marmur, MD, a dermatologist in private practice and an associate clinical professor at the Mount Sinai School of Medicine, New York.
She’s dealing with patients who have abrasions and skin infections at the tip of the nose, bruising from the metal strap that goes across the bridge of the nose, and skin irritation from the straps. “Rosacea is [also] definitely flaring up, [and] people’s acne is definitely flaring up, not only because of the stress, but because of the sweat and humidity” that builds up under the masks, she said.
“It’s not a life-threatening thing, but it’s definitely something we’ve been helping people with,” she said. This includes her husband, a cardiologist pulling 12-hour shifts in a New York City hospital wearing an N95 mask; when he comes home, the tip of his nose is red and abraded.
Treatment entails first aid skin care: a dab of a gentle ointment like Aquaphor to prevent abrasions while the mask is on and to help them heal after it’s off, and bacitracin if infection is a worry. For acne and rosacea flares, a course of minocycline or topical clindamycin might help, Dr. Marmur said.
Although almost 75% of the doctors and nurses in the Chinese study reported skin problems, the response rate was low, just 376 of the 1,000 surveyed (37.6%). That might have tilted the results to providers who actually ran into problems, wrote the investigators, led by Ping Lin of the department of dermatology and venereology at Peking University First Hospital, Beijing.
Still, 280 (74.5%) reported adverse skin reactions from caring for COVID-19 patients. “Of note, this rate was much higher than the rate of occupational contact dermatitis (31.5%) in HCWs under normal working condition[s], and that of adverse skin reactions (21.4%-35.5%)” during the outbreak of another coronavirus in 2003, severe acute respiratory syndrome, they wrote.
Most providers in the study washed their hands more than 10 times a day, but only about 22% applied hand cream afterwards, they reported.
On multivariate analysis, working in hospitals harder hit by the pandemic (odds ratio, 2.41; P = .001), working on inpatient wards (OR, 2.44; P = .003), wearing full-body personal protective equipment over 6 hours (OR, 4.26; P < .001), and female sex (OR, 1.87; P = .038) increased the risk of adverse skin reactions. The team suggested moisturizers would help to protect against hand dermatitis, and alcohol-based products instead of soaps “as the former show high antimicrobial activity and low risks of skin damage.” Also, “restricting duration of wearing” of protection gear “to no more than 6 hours would help.”
The study investigators reported that they had no conflicts of interest.
SOURCE: Lin P et al. Br J Dermatol. 2020 Apr 7. doi: 10.1111/bjd.19089.
Almost 75% of doctors and nurses in and around Wuhan, China, where the outbreak first emerged, reported skin problems during a single week in early February 2020, in a survey of health care workers (HCW) caring for COVID-19 patients at five university and five regional hospitals. Hands, cheeks, and the nasal bridge were the most commonly affected areas, with skin dryness, maceration, papules, and erythema the most common problems, according to research published in the British Journal of Dermatology.
In New York City, masks in particular are “really an issue,” said Ellen Marmur, MD, a dermatologist in private practice and an associate clinical professor at the Mount Sinai School of Medicine, New York.
She’s dealing with patients who have abrasions and skin infections at the tip of the nose, bruising from the metal strap that goes across the bridge of the nose, and skin irritation from the straps. “Rosacea is [also] definitely flaring up, [and] people’s acne is definitely flaring up, not only because of the stress, but because of the sweat and humidity” that builds up under the masks, she said.
“It’s not a life-threatening thing, but it’s definitely something we’ve been helping people with,” she said. This includes her husband, a cardiologist pulling 12-hour shifts in a New York City hospital wearing an N95 mask; when he comes home, the tip of his nose is red and abraded.
Treatment entails first aid skin care: a dab of a gentle ointment like Aquaphor to prevent abrasions while the mask is on and to help them heal after it’s off, and bacitracin if infection is a worry. For acne and rosacea flares, a course of minocycline or topical clindamycin might help, Dr. Marmur said.
Although almost 75% of the doctors and nurses in the Chinese study reported skin problems, the response rate was low, just 376 of the 1,000 surveyed (37.6%). That might have tilted the results to providers who actually ran into problems, wrote the investigators, led by Ping Lin of the department of dermatology and venereology at Peking University First Hospital, Beijing.
Still, 280 (74.5%) reported adverse skin reactions from caring for COVID-19 patients. “Of note, this rate was much higher than the rate of occupational contact dermatitis (31.5%) in HCWs under normal working condition[s], and that of adverse skin reactions (21.4%-35.5%)” during the outbreak of another coronavirus in 2003, severe acute respiratory syndrome, they wrote.
Most providers in the study washed their hands more than 10 times a day, but only about 22% applied hand cream afterwards, they reported.
On multivariate analysis, working in hospitals harder hit by the pandemic (odds ratio, 2.41; P = .001), working on inpatient wards (OR, 2.44; P = .003), wearing full-body personal protective equipment over 6 hours (OR, 4.26; P < .001), and female sex (OR, 1.87; P = .038) increased the risk of adverse skin reactions. The team suggested moisturizers would help to protect against hand dermatitis, and alcohol-based products instead of soaps “as the former show high antimicrobial activity and low risks of skin damage.” Also, “restricting duration of wearing” of protection gear “to no more than 6 hours would help.”
The study investigators reported that they had no conflicts of interest.
SOURCE: Lin P et al. Br J Dermatol. 2020 Apr 7. doi: 10.1111/bjd.19089.
Almost 75% of doctors and nurses in and around Wuhan, China, where the outbreak first emerged, reported skin problems during a single week in early February 2020, in a survey of health care workers (HCW) caring for COVID-19 patients at five university and five regional hospitals. Hands, cheeks, and the nasal bridge were the most commonly affected areas, with skin dryness, maceration, papules, and erythema the most common problems, according to research published in the British Journal of Dermatology.
In New York City, masks in particular are “really an issue,” said Ellen Marmur, MD, a dermatologist in private practice and an associate clinical professor at the Mount Sinai School of Medicine, New York.
She’s dealing with patients who have abrasions and skin infections at the tip of the nose, bruising from the metal strap that goes across the bridge of the nose, and skin irritation from the straps. “Rosacea is [also] definitely flaring up, [and] people’s acne is definitely flaring up, not only because of the stress, but because of the sweat and humidity” that builds up under the masks, she said.
“It’s not a life-threatening thing, but it’s definitely something we’ve been helping people with,” she said. This includes her husband, a cardiologist pulling 12-hour shifts in a New York City hospital wearing an N95 mask; when he comes home, the tip of his nose is red and abraded.
Treatment entails first aid skin care: a dab of a gentle ointment like Aquaphor to prevent abrasions while the mask is on and to help them heal after it’s off, and bacitracin if infection is a worry. For acne and rosacea flares, a course of minocycline or topical clindamycin might help, Dr. Marmur said.
Although almost 75% of the doctors and nurses in the Chinese study reported skin problems, the response rate was low, just 376 of the 1,000 surveyed (37.6%). That might have tilted the results to providers who actually ran into problems, wrote the investigators, led by Ping Lin of the department of dermatology and venereology at Peking University First Hospital, Beijing.
Still, 280 (74.5%) reported adverse skin reactions from caring for COVID-19 patients. “Of note, this rate was much higher than the rate of occupational contact dermatitis (31.5%) in HCWs under normal working condition[s], and that of adverse skin reactions (21.4%-35.5%)” during the outbreak of another coronavirus in 2003, severe acute respiratory syndrome, they wrote.
Most providers in the study washed their hands more than 10 times a day, but only about 22% applied hand cream afterwards, they reported.
On multivariate analysis, working in hospitals harder hit by the pandemic (odds ratio, 2.41; P = .001), working on inpatient wards (OR, 2.44; P = .003), wearing full-body personal protective equipment over 6 hours (OR, 4.26; P < .001), and female sex (OR, 1.87; P = .038) increased the risk of adverse skin reactions. The team suggested moisturizers would help to protect against hand dermatitis, and alcohol-based products instead of soaps “as the former show high antimicrobial activity and low risks of skin damage.” Also, “restricting duration of wearing” of protection gear “to no more than 6 hours would help.”
The study investigators reported that they had no conflicts of interest.
SOURCE: Lin P et al. Br J Dermatol. 2020 Apr 7. doi: 10.1111/bjd.19089.
FROM THE BRITISH JOURNAL OF DERMATOLOGY