User login
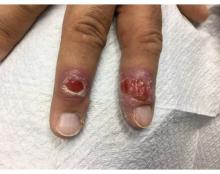
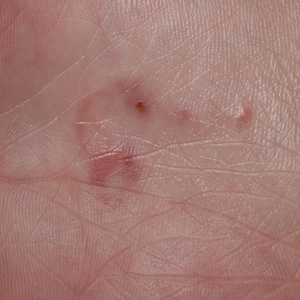
A 58-year-old White male presented with lesions on his index and middle finger for 3 months
Syphilis
Two biopsies by punch technique were performed; one for pathology and one for tissue culture (fungal and atypical mycobacteria). Tissue cultures showed no growth at 4 and 6 weeks, respectively. The lesions were swabbed for bacterial and viral cultures. Bacterial culture was positive for methicillin-resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa, and group C Streptococcus. Viral culture for herpes simplex virus (HSV) and varicella zoster virus (VZV) was negative. Histopathology confirmed the diagnosis of syphilis. Immunoperoxidase stain was positive for Treponema pallidum, and negative for HSV-1, HSV-2, and VZV. Special stains for PAS, GMS, Fite, and AFB were negative for organisms.
Syphilis, also known as Lues disease, is a contagious, sexually acquired disease caused by the spirochete T pallidum. The skin and mucous membranes are primarily infected. There are primary, secondary, and tertiary stages. In the primary or initial stage of syphilis, a chancre appears, usually 3-4 weeks after infection. The chancre is a painless papule or erosion that progresses to a firm ulceration. Lymphadenopathy may be present. Less often, multiple chancres may be present. Primary chancre on the finger has been reported in the literature, although it is far less common to have extragenital primary syphilis. The incidence ranges from 2% to 10%. Other extragenital areas that can be affected include lips, intraoral lesions, and the anus. Atypical chancres can be formed when other microbial agents are also present. Generally, an untreated chancre will heal spontaneously within a few months.
The patient referred to the department of health for treatment with penicillin G and further workup of sexually transmitted diseases. He was also seen by infectious disease for treatment of the superimposed bacterial infections and treated with an antibiotic regimen.
The case and photo were submitted by Dr. Bilu Martin.
Dr Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Ramoni S et al. Sex Transm Dis. 2010 Jul;37(7):468. doi: 10.1097/OLQ.0b013e3181e2cfac.
Starzycki Z. Br J Vener Dis. 1983 Jun;59(3):169-71. doi: 10.1136/sti.59.3.169.
Syphilis
Two biopsies by punch technique were performed; one for pathology and one for tissue culture (fungal and atypical mycobacteria). Tissue cultures showed no growth at 4 and 6 weeks, respectively. The lesions were swabbed for bacterial and viral cultures. Bacterial culture was positive for methicillin-resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa, and group C Streptococcus. Viral culture for herpes simplex virus (HSV) and varicella zoster virus (VZV) was negative. Histopathology confirmed the diagnosis of syphilis. Immunoperoxidase stain was positive for Treponema pallidum, and negative for HSV-1, HSV-2, and VZV. Special stains for PAS, GMS, Fite, and AFB were negative for organisms.
Syphilis, also known as Lues disease, is a contagious, sexually acquired disease caused by the spirochete T pallidum. The skin and mucous membranes are primarily infected. There are primary, secondary, and tertiary stages. In the primary or initial stage of syphilis, a chancre appears, usually 3-4 weeks after infection. The chancre is a painless papule or erosion that progresses to a firm ulceration. Lymphadenopathy may be present. Less often, multiple chancres may be present. Primary chancre on the finger has been reported in the literature, although it is far less common to have extragenital primary syphilis. The incidence ranges from 2% to 10%. Other extragenital areas that can be affected include lips, intraoral lesions, and the anus. Atypical chancres can be formed when other microbial agents are also present. Generally, an untreated chancre will heal spontaneously within a few months.
The patient referred to the department of health for treatment with penicillin G and further workup of sexually transmitted diseases. He was also seen by infectious disease for treatment of the superimposed bacterial infections and treated with an antibiotic regimen.
The case and photo were submitted by Dr. Bilu Martin.
Dr Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Ramoni S et al. Sex Transm Dis. 2010 Jul;37(7):468. doi: 10.1097/OLQ.0b013e3181e2cfac.
Starzycki Z. Br J Vener Dis. 1983 Jun;59(3):169-71. doi: 10.1136/sti.59.3.169.
Syphilis
Two biopsies by punch technique were performed; one for pathology and one for tissue culture (fungal and atypical mycobacteria). Tissue cultures showed no growth at 4 and 6 weeks, respectively. The lesions were swabbed for bacterial and viral cultures. Bacterial culture was positive for methicillin-resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa, and group C Streptococcus. Viral culture for herpes simplex virus (HSV) and varicella zoster virus (VZV) was negative. Histopathology confirmed the diagnosis of syphilis. Immunoperoxidase stain was positive for Treponema pallidum, and negative for HSV-1, HSV-2, and VZV. Special stains for PAS, GMS, Fite, and AFB were negative for organisms.
Syphilis, also known as Lues disease, is a contagious, sexually acquired disease caused by the spirochete T pallidum. The skin and mucous membranes are primarily infected. There are primary, secondary, and tertiary stages. In the primary or initial stage of syphilis, a chancre appears, usually 3-4 weeks after infection. The chancre is a painless papule or erosion that progresses to a firm ulceration. Lymphadenopathy may be present. Less often, multiple chancres may be present. Primary chancre on the finger has been reported in the literature, although it is far less common to have extragenital primary syphilis. The incidence ranges from 2% to 10%. Other extragenital areas that can be affected include lips, intraoral lesions, and the anus. Atypical chancres can be formed when other microbial agents are also present. Generally, an untreated chancre will heal spontaneously within a few months.
The patient referred to the department of health for treatment with penicillin G and further workup of sexually transmitted diseases. He was also seen by infectious disease for treatment of the superimposed bacterial infections and treated with an antibiotic regimen.
The case and photo were submitted by Dr. Bilu Martin.
Dr Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Ramoni S et al. Sex Transm Dis. 2010 Jul;37(7):468. doi: 10.1097/OLQ.0b013e3181e2cfac.
Starzycki Z. Br J Vener Dis. 1983 Jun;59(3):169-71. doi: 10.1136/sti.59.3.169.
A 58-year-old White male with no significant past medical history presented with lesions on his right index and middle fingers, which had been present for 3 months. The lesions were painless. The patient has a history of hand dermatitis. Upon questioning, the patient said he had not fished or cleaned fish tanks. He did garden occasionally (no roses). He has been using Neosporin on the lesions. He denied any fever or systemic symptoms and had no lymphadenopathy.
What's your diagnosis?
Slim Silver Lining Appears for STI Rates
The persistent epidemic of sexually transmitted infections (STIs) in the United States is showing signs of a slowdown in cases of syphilis, gonorrhea, and chlamydia, according to the latest data from the Centers for Disease Control and Prevention (CDC).
More than 2.4 million cases of these three nationally notifiable STIs were reported in the United States in 2023 but represent a 1.8% decrease from 2022, based on a new CDC report, Sexually Transmitted Infections Surveillance, 2023.
The 2023 report indicates a 7.2% decrease in gonorrhea, which accounts for most of the decrease.
Although syphilis cases increased overall, they did so by only 1% compared with double-digit increases in previous years, according to the report. Primary and secondary syphilis decreased by 10%, compared with 2022 overall, with a 13% decrease in cases among gay and bisexual men.
Congenital syphilis rates increased by 3%. However, the 3% increase represents a significant drop from the 30% increases each year in recent years, according to the report.
Chlamydia rates remained essentially stable, with a decrease of less than 1.0% overall. Reported chlamydia rates increased by 1.3% among men and decreased by 1.7% among women.
Despite the declines, overall disparities persist, with higher rates of STIs among gay and bisexual men, as well as American Indian/Alaska Native, Black/African American, and Hispanic/Latino populations, according to the report.
CDC Cautiously Optimistic
The CDC is “guardedly optimistic that the new data represent a turning point in terms of syphilis and gonorrhea,” said Bradley Stoner, MD, director of the CDC’s Division of STD Prevention, in an interview.
However, a tremendous amount of work remains to be done, notably in addressing disparities in care, said Stoner.
New techniques for diagnosis and treatment, such as the increased use of doxycycline (doxy PEP) for the prevention of STIs after sex for high-risk populations with a history of STIs, are likely contributing to the overall decrease, Stoner said. Other contributing factors include improved communication and awareness of STI treatment options at the community level in emergency departments, substance use facilities, and syringe use programs.
Although the United States has not yet turned the corner in reducing STIs, “We are at an inflection point in the epidemic after years of increases,” Stoner told this news organization. “The CDC is committed to keeping the momentum going and turning things around.” Although congenital syphilis rates are slowing down, it remains a significant problem with severe outcomes for mothers and infants, he noted.
The message to healthcare providers on the front lines is to increase awareness, screen widely, and provide effective treatments, Stoner emphasized.
Looking ahead, more research is needed to identify the settings in which prevention tools can be best utilized, such as urgent care or other programs, said Stoner. “My hope is that implementation science research will give us some clues.” In addition, better tools for detection and treatment of STIs are always needed, notably better diagnostics for syphilis, which still requires a blood test, although research is underway for more efficient testing.
Spotlight on Disparities, Syphilis
“I think these are very nuanced results,” said David J. Cennimo, MD, associate professor of medicine and pediatrics in the Division of Infectious Disease at Rutgers New Jersey Medical School, Newark, in an interview. “I am happy, on first pass, to see that STI rates have declined.” However, a closer look reveals that most of the improvements are driven by the 7% drop in gonorrhea, while chlamydia and syphilis rates are relatively stable.
The decreases may reflect that the public is receiving the messaging about the need for screening and safer sex. “Clinicians also have been educated on the need for screening,” Cennimo said. However, “we are still 90% above the [STI] rates from 20 years ago.”
Clinicians also must recognize the disparities in STI rates by race and other demographics, Cennimo said. The current report “is a call to make sure that STI and other medical services are targeted to specific groups as needed and are widely available, especially in under-resourced areas.”
“I am still dismayed by the high syphilis rates, which are also resulting in congenital syphilis,” Cennimo said. “Syphilis in pregnancy is very dangerous, and any case of congenital syphilis is a failure of preventive care and screening; it is a potentially devastating disease.
“We have good treatments for STIs, but we must continue to monitor for resistance,” said Cennimo. “More work is needed to reach high-risk individuals and to provide preventive care and screening.”
The research was supported by the CDC. Stoner and Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
The persistent epidemic of sexually transmitted infections (STIs) in the United States is showing signs of a slowdown in cases of syphilis, gonorrhea, and chlamydia, according to the latest data from the Centers for Disease Control and Prevention (CDC).
More than 2.4 million cases of these three nationally notifiable STIs were reported in the United States in 2023 but represent a 1.8% decrease from 2022, based on a new CDC report, Sexually Transmitted Infections Surveillance, 2023.
The 2023 report indicates a 7.2% decrease in gonorrhea, which accounts for most of the decrease.
Although syphilis cases increased overall, they did so by only 1% compared with double-digit increases in previous years, according to the report. Primary and secondary syphilis decreased by 10%, compared with 2022 overall, with a 13% decrease in cases among gay and bisexual men.
Congenital syphilis rates increased by 3%. However, the 3% increase represents a significant drop from the 30% increases each year in recent years, according to the report.
Chlamydia rates remained essentially stable, with a decrease of less than 1.0% overall. Reported chlamydia rates increased by 1.3% among men and decreased by 1.7% among women.
Despite the declines, overall disparities persist, with higher rates of STIs among gay and bisexual men, as well as American Indian/Alaska Native, Black/African American, and Hispanic/Latino populations, according to the report.
CDC Cautiously Optimistic
The CDC is “guardedly optimistic that the new data represent a turning point in terms of syphilis and gonorrhea,” said Bradley Stoner, MD, director of the CDC’s Division of STD Prevention, in an interview.
However, a tremendous amount of work remains to be done, notably in addressing disparities in care, said Stoner.
New techniques for diagnosis and treatment, such as the increased use of doxycycline (doxy PEP) for the prevention of STIs after sex for high-risk populations with a history of STIs, are likely contributing to the overall decrease, Stoner said. Other contributing factors include improved communication and awareness of STI treatment options at the community level in emergency departments, substance use facilities, and syringe use programs.
Although the United States has not yet turned the corner in reducing STIs, “We are at an inflection point in the epidemic after years of increases,” Stoner told this news organization. “The CDC is committed to keeping the momentum going and turning things around.” Although congenital syphilis rates are slowing down, it remains a significant problem with severe outcomes for mothers and infants, he noted.
The message to healthcare providers on the front lines is to increase awareness, screen widely, and provide effective treatments, Stoner emphasized.
Looking ahead, more research is needed to identify the settings in which prevention tools can be best utilized, such as urgent care or other programs, said Stoner. “My hope is that implementation science research will give us some clues.” In addition, better tools for detection and treatment of STIs are always needed, notably better diagnostics for syphilis, which still requires a blood test, although research is underway for more efficient testing.
Spotlight on Disparities, Syphilis
“I think these are very nuanced results,” said David J. Cennimo, MD, associate professor of medicine and pediatrics in the Division of Infectious Disease at Rutgers New Jersey Medical School, Newark, in an interview. “I am happy, on first pass, to see that STI rates have declined.” However, a closer look reveals that most of the improvements are driven by the 7% drop in gonorrhea, while chlamydia and syphilis rates are relatively stable.
The decreases may reflect that the public is receiving the messaging about the need for screening and safer sex. “Clinicians also have been educated on the need for screening,” Cennimo said. However, “we are still 90% above the [STI] rates from 20 years ago.”
Clinicians also must recognize the disparities in STI rates by race and other demographics, Cennimo said. The current report “is a call to make sure that STI and other medical services are targeted to specific groups as needed and are widely available, especially in under-resourced areas.”
“I am still dismayed by the high syphilis rates, which are also resulting in congenital syphilis,” Cennimo said. “Syphilis in pregnancy is very dangerous, and any case of congenital syphilis is a failure of preventive care and screening; it is a potentially devastating disease.
“We have good treatments for STIs, but we must continue to monitor for resistance,” said Cennimo. “More work is needed to reach high-risk individuals and to provide preventive care and screening.”
The research was supported by the CDC. Stoner and Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
The persistent epidemic of sexually transmitted infections (STIs) in the United States is showing signs of a slowdown in cases of syphilis, gonorrhea, and chlamydia, according to the latest data from the Centers for Disease Control and Prevention (CDC).
More than 2.4 million cases of these three nationally notifiable STIs were reported in the United States in 2023 but represent a 1.8% decrease from 2022, based on a new CDC report, Sexually Transmitted Infections Surveillance, 2023.
The 2023 report indicates a 7.2% decrease in gonorrhea, which accounts for most of the decrease.
Although syphilis cases increased overall, they did so by only 1% compared with double-digit increases in previous years, according to the report. Primary and secondary syphilis decreased by 10%, compared with 2022 overall, with a 13% decrease in cases among gay and bisexual men.
Congenital syphilis rates increased by 3%. However, the 3% increase represents a significant drop from the 30% increases each year in recent years, according to the report.
Chlamydia rates remained essentially stable, with a decrease of less than 1.0% overall. Reported chlamydia rates increased by 1.3% among men and decreased by 1.7% among women.
Despite the declines, overall disparities persist, with higher rates of STIs among gay and bisexual men, as well as American Indian/Alaska Native, Black/African American, and Hispanic/Latino populations, according to the report.
CDC Cautiously Optimistic
The CDC is “guardedly optimistic that the new data represent a turning point in terms of syphilis and gonorrhea,” said Bradley Stoner, MD, director of the CDC’s Division of STD Prevention, in an interview.
However, a tremendous amount of work remains to be done, notably in addressing disparities in care, said Stoner.
New techniques for diagnosis and treatment, such as the increased use of doxycycline (doxy PEP) for the prevention of STIs after sex for high-risk populations with a history of STIs, are likely contributing to the overall decrease, Stoner said. Other contributing factors include improved communication and awareness of STI treatment options at the community level in emergency departments, substance use facilities, and syringe use programs.
Although the United States has not yet turned the corner in reducing STIs, “We are at an inflection point in the epidemic after years of increases,” Stoner told this news organization. “The CDC is committed to keeping the momentum going and turning things around.” Although congenital syphilis rates are slowing down, it remains a significant problem with severe outcomes for mothers and infants, he noted.
The message to healthcare providers on the front lines is to increase awareness, screen widely, and provide effective treatments, Stoner emphasized.
Looking ahead, more research is needed to identify the settings in which prevention tools can be best utilized, such as urgent care or other programs, said Stoner. “My hope is that implementation science research will give us some clues.” In addition, better tools for detection and treatment of STIs are always needed, notably better diagnostics for syphilis, which still requires a blood test, although research is underway for more efficient testing.
Spotlight on Disparities, Syphilis
“I think these are very nuanced results,” said David J. Cennimo, MD, associate professor of medicine and pediatrics in the Division of Infectious Disease at Rutgers New Jersey Medical School, Newark, in an interview. “I am happy, on first pass, to see that STI rates have declined.” However, a closer look reveals that most of the improvements are driven by the 7% drop in gonorrhea, while chlamydia and syphilis rates are relatively stable.
The decreases may reflect that the public is receiving the messaging about the need for screening and safer sex. “Clinicians also have been educated on the need for screening,” Cennimo said. However, “we are still 90% above the [STI] rates from 20 years ago.”
Clinicians also must recognize the disparities in STI rates by race and other demographics, Cennimo said. The current report “is a call to make sure that STI and other medical services are targeted to specific groups as needed and are widely available, especially in under-resourced areas.”
“I am still dismayed by the high syphilis rates, which are also resulting in congenital syphilis,” Cennimo said. “Syphilis in pregnancy is very dangerous, and any case of congenital syphilis is a failure of preventive care and screening; it is a potentially devastating disease.
“We have good treatments for STIs, but we must continue to monitor for resistance,” said Cennimo. “More work is needed to reach high-risk individuals and to provide preventive care and screening.”
The research was supported by the CDC. Stoner and Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Post COVID-19, Long-term Risk for Autoimmune, Autoinflammatory Skin Disorders Increased, Study Finds
In addition, the authors reported that COVID-19 vaccination appears to reduce these risks.
The study was published in JAMA Dermatology.
‘Compelling Evidence’
“This well-executed study by Heo et al provides compelling evidence to support an association between COVID-19 infection and the development of subsequent autoimmune and autoinflammatory skin diseases,” wrote authors led by Lisa M. Arkin, MD, of the Department of Dermatology, University of Wisconsin School of Medicine and Public Health in Madison, in an accompanying editorial.
Using databases from Korea’s National Health Insurance Service and the Korea Disease Control and Prevention Agency, investigators led by Yeon-Woo Heo, MD, a dermatology resident at Yonsei University Wonju College of Medicine, Wonju, Republic of Korea, compared 3.1 million people who had COVID-19 with 3.8 million controls, all with at least 180 days’ follow-up through December 31, 2022.
At a mean follow-up of 287 days in both cohorts, authors found significantly elevated risks for AA and vitiligo (adjusted hazard ratio [aHR], 1.11 for both), AT (aHR, 1.24), Behçet disease (aHR, 1.45), and BP (aHR, 1.62) in the post–COVID-19 cohort. The infection also raised the risk for other conditions such as systemic lupus erythematosus (aHR, 1.14) and Crohn’s disease (aHR, 1.35).
In subgroup analyses, demographic factors were associated with diverse effects: COVID-19 infection was associated with significantly higher odds of developing AA (for both men and women), vitiligo (men), Behçet disease (men and women), Crohn’s disease (men), ulcerative colitis (men), rheumatoid arthritis (men and women), systemic lupus erythematosus (men), ankylosing spondylitis (men), AT (women), and BP (women) than controls.
Those aged under 40 years were more likely to develop AA, primary cicatricial alopecia, Behçet disease, and ulcerative colitis, while those aged 40 years or older were more likely to develop AA, AT, vitiligo, Behçet disease, Crohn’s disease, rheumatoid arthritis, systemic lupus erythematosus, Sjögren’s syndrome, ankylosing spondylitis, and BP.
Additionally, severe COVID-19 requiring intensive care unit admission was associated with a significantly increased risk for autoimmune diseases, including AA, psoriasis, BP, and sarcoidosis. By timeframe, risks for AA, AT, and psoriasis were significantly higher during the initial Delta-dominant period.
Vaccination Effect
Moreover, vaccinated individuals were less likely to develop AA, AT, psoriasis, Behçet disease, and various nondermatologic conditions than were those who were unvaccinated. This finding, wrote Heo and colleagues, “may provide evidence to support the hypothesis that COVID-19 vaccines can help prevent autoimmune diseases.”
“That’s the part we all need to take into our offices tomorrow,” said Brett King, MD, PhD, a Fairfield, Connecticut–based dermatologist in private practice. He was not involved with the study but was asked to comment.
Overall, King said, the study carries two main messages. “The first is that COVID-19 infection increases the likelihood of developing an autoimmune or autoinflammatory disease in a large population.” The second and very important message is that being vaccinated against COVID-19 provides protection against developing an autoimmune or autoinflammatory disease.
“My concern is that the popular media highlights the first part,” said King, “and everybody who develops alopecia areata, vitiligo, or sarcoidosis blames COVID-19. That’s not what this work says.”
The foregoing distinction is especially important during the fall and winter, he added, when people getting influenza vaccines are routinely offered COVID-19 vaccines. “Many patients have said, ‘I got the COVID vaccine and developed alopecia areata 6 months later.’ Nearly everybody who has developed a new or worsening health condition in the last almost 5 years has had the perfect fall guy — the COVID vaccine or infection.”
With virtually all patients asking if they should get an updated COVID-19 vaccine or booster, he added, many report having heard that such vaccines cause AA, vitiligo, or other diseases. “To anchor these conversations in real data and not just anecdotes from a blog or Facebook is very useful,” said King, “and now we have very good data saying that the COVID vaccine is protective against these disorders.”
George Han, MD, PhD, associate professor of dermatology at the Donald and Barbara Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York, applauds investigators’ use of a large, robust database but suggests interpreting results cautiously. He was not involved with the study but was asked to comment.
“You could do a large, well-done study,” Han said, “but it could still not necessarily be generalizable. These autoimmune conditions they’re looking at have clear ethnic and racial biases.” Heo and colleagues acknowledged shortcomings including their study population’s monomorphic nature.
Additional issues that limit the study’s impact, said Han, include the difficulty of conceptualizing a 10%-20% increase in conditions that at baseline are rare. And many of the findings reflected natural patterns, he said. For instance, BP more commonly affects older people, COVID-19 notwithstanding.
Han said that for him, the study’s main value going forward is helping to explain a rash of worsening inflammatory skin disease that many dermatologists saw early in the pandemic. “We would regularly see patients who were well controlled with, for example, psoriasis or eczema. But after COVID-19 infection or a vaccine (usually mRNA-type), in some cases they would come in flaring badly.” This happened at least a dozen times during the first year of post-shutdown appointments, he said.
“We’ve seen patients who have flared multiple times — they get the booster, then flare again,” Han added. Similar patterns occurred with pyoderma gangrenosum and other inflammatory skin diseases, he said.
Given the modest effect sizes of the associations reported in the Korean study, Arkin and colleagues wrote in their JAMA Dermatology editorial that surveillance for autoimmune disease is probably not warranted without new examination findings or symptoms. “For certain,” King said, “we should not go hunting for things that aren’t obviously there.”
Rather, Arkin and colleagues wrote, the higher autoimmunity rates seen among the unvaccinated, as well as during the Delta phase (when patients were sicker and hospitalizations were more likely) and in patients requiring intensive care, suggest that “interventions that reduce disease severity could also potentially reduce long-term risk of subsequent autoimmune sequelae.”
Future research addressing whether people with preexisting autoimmune conditions are at greater risk for flares or developing new autoimmune diseases following COVID-19 infection “would help to frame an evidence-based approach for patients with autoimmune disorders who develop COVID-19 infection, including the role for antiviral treatments,” they added.
The study was supported by grants from the Research Program of the Korea Medical Institute, the Korea Health Industry Development Institute, and the National Research Foundation of Korea. Han and King reported no relevant financial relationships. Arkin disclosed receiving research grants to her institution from Amgen and Eli Lilly, personal fees from Sanofi/Regeneron for consulting, and personal consulting fees from Merck outside the submitted work. Another author reported personal consulting fees from Dexcel Pharma and Honeydew outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
In addition, the authors reported that COVID-19 vaccination appears to reduce these risks.
The study was published in JAMA Dermatology.
‘Compelling Evidence’
“This well-executed study by Heo et al provides compelling evidence to support an association between COVID-19 infection and the development of subsequent autoimmune and autoinflammatory skin diseases,” wrote authors led by Lisa M. Arkin, MD, of the Department of Dermatology, University of Wisconsin School of Medicine and Public Health in Madison, in an accompanying editorial.
Using databases from Korea’s National Health Insurance Service and the Korea Disease Control and Prevention Agency, investigators led by Yeon-Woo Heo, MD, a dermatology resident at Yonsei University Wonju College of Medicine, Wonju, Republic of Korea, compared 3.1 million people who had COVID-19 with 3.8 million controls, all with at least 180 days’ follow-up through December 31, 2022.
At a mean follow-up of 287 days in both cohorts, authors found significantly elevated risks for AA and vitiligo (adjusted hazard ratio [aHR], 1.11 for both), AT (aHR, 1.24), Behçet disease (aHR, 1.45), and BP (aHR, 1.62) in the post–COVID-19 cohort. The infection also raised the risk for other conditions such as systemic lupus erythematosus (aHR, 1.14) and Crohn’s disease (aHR, 1.35).
In subgroup analyses, demographic factors were associated with diverse effects: COVID-19 infection was associated with significantly higher odds of developing AA (for both men and women), vitiligo (men), Behçet disease (men and women), Crohn’s disease (men), ulcerative colitis (men), rheumatoid arthritis (men and women), systemic lupus erythematosus (men), ankylosing spondylitis (men), AT (women), and BP (women) than controls.
Those aged under 40 years were more likely to develop AA, primary cicatricial alopecia, Behçet disease, and ulcerative colitis, while those aged 40 years or older were more likely to develop AA, AT, vitiligo, Behçet disease, Crohn’s disease, rheumatoid arthritis, systemic lupus erythematosus, Sjögren’s syndrome, ankylosing spondylitis, and BP.
Additionally, severe COVID-19 requiring intensive care unit admission was associated with a significantly increased risk for autoimmune diseases, including AA, psoriasis, BP, and sarcoidosis. By timeframe, risks for AA, AT, and psoriasis were significantly higher during the initial Delta-dominant period.
Vaccination Effect
Moreover, vaccinated individuals were less likely to develop AA, AT, psoriasis, Behçet disease, and various nondermatologic conditions than were those who were unvaccinated. This finding, wrote Heo and colleagues, “may provide evidence to support the hypothesis that COVID-19 vaccines can help prevent autoimmune diseases.”
“That’s the part we all need to take into our offices tomorrow,” said Brett King, MD, PhD, a Fairfield, Connecticut–based dermatologist in private practice. He was not involved with the study but was asked to comment.
Overall, King said, the study carries two main messages. “The first is that COVID-19 infection increases the likelihood of developing an autoimmune or autoinflammatory disease in a large population.” The second and very important message is that being vaccinated against COVID-19 provides protection against developing an autoimmune or autoinflammatory disease.
“My concern is that the popular media highlights the first part,” said King, “and everybody who develops alopecia areata, vitiligo, or sarcoidosis blames COVID-19. That’s not what this work says.”
The foregoing distinction is especially important during the fall and winter, he added, when people getting influenza vaccines are routinely offered COVID-19 vaccines. “Many patients have said, ‘I got the COVID vaccine and developed alopecia areata 6 months later.’ Nearly everybody who has developed a new or worsening health condition in the last almost 5 years has had the perfect fall guy — the COVID vaccine or infection.”
With virtually all patients asking if they should get an updated COVID-19 vaccine or booster, he added, many report having heard that such vaccines cause AA, vitiligo, or other diseases. “To anchor these conversations in real data and not just anecdotes from a blog or Facebook is very useful,” said King, “and now we have very good data saying that the COVID vaccine is protective against these disorders.”
George Han, MD, PhD, associate professor of dermatology at the Donald and Barbara Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York, applauds investigators’ use of a large, robust database but suggests interpreting results cautiously. He was not involved with the study but was asked to comment.
“You could do a large, well-done study,” Han said, “but it could still not necessarily be generalizable. These autoimmune conditions they’re looking at have clear ethnic and racial biases.” Heo and colleagues acknowledged shortcomings including their study population’s monomorphic nature.
Additional issues that limit the study’s impact, said Han, include the difficulty of conceptualizing a 10%-20% increase in conditions that at baseline are rare. And many of the findings reflected natural patterns, he said. For instance, BP more commonly affects older people, COVID-19 notwithstanding.
Han said that for him, the study’s main value going forward is helping to explain a rash of worsening inflammatory skin disease that many dermatologists saw early in the pandemic. “We would regularly see patients who were well controlled with, for example, psoriasis or eczema. But after COVID-19 infection or a vaccine (usually mRNA-type), in some cases they would come in flaring badly.” This happened at least a dozen times during the first year of post-shutdown appointments, he said.
“We’ve seen patients who have flared multiple times — they get the booster, then flare again,” Han added. Similar patterns occurred with pyoderma gangrenosum and other inflammatory skin diseases, he said.
Given the modest effect sizes of the associations reported in the Korean study, Arkin and colleagues wrote in their JAMA Dermatology editorial that surveillance for autoimmune disease is probably not warranted without new examination findings or symptoms. “For certain,” King said, “we should not go hunting for things that aren’t obviously there.”
Rather, Arkin and colleagues wrote, the higher autoimmunity rates seen among the unvaccinated, as well as during the Delta phase (when patients were sicker and hospitalizations were more likely) and in patients requiring intensive care, suggest that “interventions that reduce disease severity could also potentially reduce long-term risk of subsequent autoimmune sequelae.”
Future research addressing whether people with preexisting autoimmune conditions are at greater risk for flares or developing new autoimmune diseases following COVID-19 infection “would help to frame an evidence-based approach for patients with autoimmune disorders who develop COVID-19 infection, including the role for antiviral treatments,” they added.
The study was supported by grants from the Research Program of the Korea Medical Institute, the Korea Health Industry Development Institute, and the National Research Foundation of Korea. Han and King reported no relevant financial relationships. Arkin disclosed receiving research grants to her institution from Amgen and Eli Lilly, personal fees from Sanofi/Regeneron for consulting, and personal consulting fees from Merck outside the submitted work. Another author reported personal consulting fees from Dexcel Pharma and Honeydew outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
In addition, the authors reported that COVID-19 vaccination appears to reduce these risks.
The study was published in JAMA Dermatology.
‘Compelling Evidence’
“This well-executed study by Heo et al provides compelling evidence to support an association between COVID-19 infection and the development of subsequent autoimmune and autoinflammatory skin diseases,” wrote authors led by Lisa M. Arkin, MD, of the Department of Dermatology, University of Wisconsin School of Medicine and Public Health in Madison, in an accompanying editorial.
Using databases from Korea’s National Health Insurance Service and the Korea Disease Control and Prevention Agency, investigators led by Yeon-Woo Heo, MD, a dermatology resident at Yonsei University Wonju College of Medicine, Wonju, Republic of Korea, compared 3.1 million people who had COVID-19 with 3.8 million controls, all with at least 180 days’ follow-up through December 31, 2022.
At a mean follow-up of 287 days in both cohorts, authors found significantly elevated risks for AA and vitiligo (adjusted hazard ratio [aHR], 1.11 for both), AT (aHR, 1.24), Behçet disease (aHR, 1.45), and BP (aHR, 1.62) in the post–COVID-19 cohort. The infection also raised the risk for other conditions such as systemic lupus erythematosus (aHR, 1.14) and Crohn’s disease (aHR, 1.35).
In subgroup analyses, demographic factors were associated with diverse effects: COVID-19 infection was associated with significantly higher odds of developing AA (for both men and women), vitiligo (men), Behçet disease (men and women), Crohn’s disease (men), ulcerative colitis (men), rheumatoid arthritis (men and women), systemic lupus erythematosus (men), ankylosing spondylitis (men), AT (women), and BP (women) than controls.
Those aged under 40 years were more likely to develop AA, primary cicatricial alopecia, Behçet disease, and ulcerative colitis, while those aged 40 years or older were more likely to develop AA, AT, vitiligo, Behçet disease, Crohn’s disease, rheumatoid arthritis, systemic lupus erythematosus, Sjögren’s syndrome, ankylosing spondylitis, and BP.
Additionally, severe COVID-19 requiring intensive care unit admission was associated with a significantly increased risk for autoimmune diseases, including AA, psoriasis, BP, and sarcoidosis. By timeframe, risks for AA, AT, and psoriasis were significantly higher during the initial Delta-dominant period.
Vaccination Effect
Moreover, vaccinated individuals were less likely to develop AA, AT, psoriasis, Behçet disease, and various nondermatologic conditions than were those who were unvaccinated. This finding, wrote Heo and colleagues, “may provide evidence to support the hypothesis that COVID-19 vaccines can help prevent autoimmune diseases.”
“That’s the part we all need to take into our offices tomorrow,” said Brett King, MD, PhD, a Fairfield, Connecticut–based dermatologist in private practice. He was not involved with the study but was asked to comment.
Overall, King said, the study carries two main messages. “The first is that COVID-19 infection increases the likelihood of developing an autoimmune or autoinflammatory disease in a large population.” The second and very important message is that being vaccinated against COVID-19 provides protection against developing an autoimmune or autoinflammatory disease.
“My concern is that the popular media highlights the first part,” said King, “and everybody who develops alopecia areata, vitiligo, or sarcoidosis blames COVID-19. That’s not what this work says.”
The foregoing distinction is especially important during the fall and winter, he added, when people getting influenza vaccines are routinely offered COVID-19 vaccines. “Many patients have said, ‘I got the COVID vaccine and developed alopecia areata 6 months later.’ Nearly everybody who has developed a new or worsening health condition in the last almost 5 years has had the perfect fall guy — the COVID vaccine or infection.”
With virtually all patients asking if they should get an updated COVID-19 vaccine or booster, he added, many report having heard that such vaccines cause AA, vitiligo, or other diseases. “To anchor these conversations in real data and not just anecdotes from a blog or Facebook is very useful,” said King, “and now we have very good data saying that the COVID vaccine is protective against these disorders.”
George Han, MD, PhD, associate professor of dermatology at the Donald and Barbara Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York, applauds investigators’ use of a large, robust database but suggests interpreting results cautiously. He was not involved with the study but was asked to comment.
“You could do a large, well-done study,” Han said, “but it could still not necessarily be generalizable. These autoimmune conditions they’re looking at have clear ethnic and racial biases.” Heo and colleagues acknowledged shortcomings including their study population’s monomorphic nature.
Additional issues that limit the study’s impact, said Han, include the difficulty of conceptualizing a 10%-20% increase in conditions that at baseline are rare. And many of the findings reflected natural patterns, he said. For instance, BP more commonly affects older people, COVID-19 notwithstanding.
Han said that for him, the study’s main value going forward is helping to explain a rash of worsening inflammatory skin disease that many dermatologists saw early in the pandemic. “We would regularly see patients who were well controlled with, for example, psoriasis or eczema. But after COVID-19 infection or a vaccine (usually mRNA-type), in some cases they would come in flaring badly.” This happened at least a dozen times during the first year of post-shutdown appointments, he said.
“We’ve seen patients who have flared multiple times — they get the booster, then flare again,” Han added. Similar patterns occurred with pyoderma gangrenosum and other inflammatory skin diseases, he said.
Given the modest effect sizes of the associations reported in the Korean study, Arkin and colleagues wrote in their JAMA Dermatology editorial that surveillance for autoimmune disease is probably not warranted without new examination findings or symptoms. “For certain,” King said, “we should not go hunting for things that aren’t obviously there.”
Rather, Arkin and colleagues wrote, the higher autoimmunity rates seen among the unvaccinated, as well as during the Delta phase (when patients were sicker and hospitalizations were more likely) and in patients requiring intensive care, suggest that “interventions that reduce disease severity could also potentially reduce long-term risk of subsequent autoimmune sequelae.”
Future research addressing whether people with preexisting autoimmune conditions are at greater risk for flares or developing new autoimmune diseases following COVID-19 infection “would help to frame an evidence-based approach for patients with autoimmune disorders who develop COVID-19 infection, including the role for antiviral treatments,” they added.
The study was supported by grants from the Research Program of the Korea Medical Institute, the Korea Health Industry Development Institute, and the National Research Foundation of Korea. Han and King reported no relevant financial relationships. Arkin disclosed receiving research grants to her institution from Amgen and Eli Lilly, personal fees from Sanofi/Regeneron for consulting, and personal consulting fees from Merck outside the submitted work. Another author reported personal consulting fees from Dexcel Pharma and Honeydew outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
FROM JAMA DERMATOLOGY
What’s Eating You? Hookworm and Cutaneous Larva Migrans
What’s Eating You? Hookworm and
Cutaneous Larva Migrans
It is estimated that the prevalence of human hookworm infection is approximately 450 million individuals worldwide, representing a substantial global disease burden.1 The annual global public health burden ranges from approximately 2 million to 4 million disability-adjusted life-years and $10 billion to $140 billion in hookwormrelated costs.2 In this article, we discuss the lifecycle, transmission, and disease burden of cutaneous larva migrans (CLM) as well as prevention and treatment strategies.
Background
The Ancylostomatidae nematode family comprises at least 68 known species of hookworm that infect more than 110 different species of mammals.3 Many of these parasites are able to infect more than 1 primary host species, but from a disease perspective they can be classified as either anthropophilic, with humans as the intended host, or zoonotic, with humans as an incidental host. It is important to make this distinction because, though the lifecycles and biology of hookworm species generally are similar, the manifestations of incidental human infection from zoonotic hookworms are different from those of anthropophilic hookworms. Of the anthropophilic species, Necator americanus and Ancylostoma duodenale predominate. In the instance of zoonotic hookworm, dog-infecting A caninum and cat- and doginfecting A braziliense and Uncinaria stenocephala are common causes of incidental human disease.3
The life cycle of Ancylostomatidae organisms is astounding. Through millions of years of co-evolution with mammals,4 these parasitic worms have developed perhaps one of the most circuitous paths to propagate themselves in the natural world. Hookworms start their arduous journey as eggs deposited in soil, sand, and ground vegetation from the feces of infected animals.5 Approximately 1 day after the eggs are deposited, they hatch and begin the larval stage, during which they become infective 1 to 5 weeks later. At this point, the larvae become sensitive to their environment, responding to rising temperatures, increasing carbon dioxide levels, and vibrations in the soil—all of which suggest the presence of a potential host and contribute to a concordant increase in undulatory movement of the larvae.5,6 Here, the most vulnerable tissues include the uncovered soles, palms, and buttocks of host mammals that come into contact with contaminated soil. In an undulating fashion and guided by temperature cues, the larvae locate the skin of the host and utilize a mixture of enzymes including hyaluronidases, metalloprotease, and other proteases to penetrate the epidermis.7 Anthropophilic hookworms such as N americanus and A duodenale will enter the circulatory system; from there, the hookworms migrate through the right-sided cardiopulmonary circuit and eventually ascend into the pulmonary vasculature.8 They then penetrate the lung capillary beds and parenchyma to reach the alveoli, ascend the respiratory tree, and, with the help of the mucociliary escalator, reach the esophagus, where they are swallowed by the host. In the gastrointestinal tract, adult hookworms consume host blood, mate, and lay eggs over a period of approximately 1 to 3 years if left untreated.9 Eggs are laid into the lower gastrointestinal tract, and the journey begins again in feces contacting ground or soil.
Geographic Distribution
Hookworms are found in almost all regions of the world, with species-specific distributions that highlight tropical and subtropical regions. Necator americanus and A duodenale are the most common hookworm species, with the former found predominantly in Southeast Asia and Latin America and the latter in Asia-Pacific regions.10 The highest prevalence of hookworms is in Southeast Asia followed by Sub-Saharan Africa, and the unique climate and soil composition of a region help determine the best environments for specific species of hookworm to thrive.11 In addition, socioeconomics and social determinants of health play a big role in the spread of hookworms, as hygiene practices (eg, wearing clean shoes and clothing, bathing), infrastructure (eg, clean water and streets), and anthelmintic campaigns help reduce transmission.12 Soil-transmitted helminths were once endemic to the southeastern United States, with some reports of approximately 40% of individuals infected in the south in the early 1900s.13 Anthelmintic campaigns such as water, sanitation, and hygiene programs as well as deworming of humans and livestock have proven effective in reducing the prevalence of helminth disease in industrialized nations.13,14 However, zoonotic infections remain a problem in these regions, and in some parts of the United States more than 40% of sampled cats and dogs harbored species such as A braziliense.15
Clinical Manifestation
Initial hookworm infection often goes unnoticed because symptoms can range in severity, but it is characterized by transient ground itch—a local pruritic, erythematous, and papular eruption that develops in response to epidermal penetration.16 Because the larvae must traverse the host from skin to target organs for reproduction over several weeks, iron-deficiency anemia will manifest much later than signs of the initial penetration. In the case of incidental infection from zoonotic Ancylostomatidae organisms, the misguided larvae result in CLM, an often intensely pruritic skin condition that will self-resolve in 2 to 8 weeks with eventual death of the larvae.5
Diagnosis and Pathology of Disease
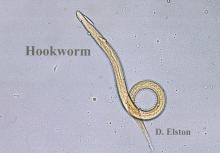
Zoonotic Hookworm—The major presenting sign of zoonotic hookworm infection is CLM. The diagnosis of CLM usually is made clinically, as the larvae themselves are 0.5 mm thick to 10 mm long (Figure 1) and usually extend several centimeters beyond the dermal lesion, with dermoscopy having limited utility.17 Patients may begin to experience itching as little as 1 hour after hookworm penetration of the skin.18 Once in contact with the skin, the hookworms’ hyaluronidases and proteases are capable of breaking through the epidermis, but zoonotic hookworms typically are unable to penetrate the basal layer of the human epidermis and remain entombed between the stratum granulosum and stratum corneum. With the exception of rare cases of direct or indirect pulmonary involvement resulting in Löffler syndrome,19 the larvae will die within weeks to months, and symptoms will subsequently resolve.
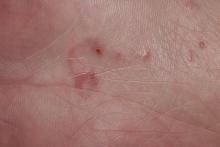
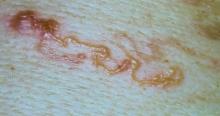
Although the infection generally is self-limiting, the dermatologic manifestations of CLM can be severe and warrant intervention. The lesions start as small reddish papules at the site of penetration (Figure 2), then the hallmark elevated, migrating, serpiginous, urticarial rash develops (Figure 3). Cutaneous larva migrans generally manifests unilaterally and is both erythematous and intensely pruritic. As the larvae migrate, they leave behind 1- to 5-cm tunneled creeping eruptions in their wake. The lesions, which can manifest with pain or be painless, may develop eczematous, bullous, follicular, or impetiginized appearances.20 Atypical manifestations include folliculitis and urticarial plaques.17
cutaneous larval migrans on the palm.
Anthropophilic Hookworm—The lifecycles of N americanus and A duodenale are completed in human infection. Dermatologic manifestations are transient with the development of ground itch at the site of epidermal penetration. The hookworms employ collagenases that allow penetration of the basal layer of the skin, and eosinophilia develops as the parasites travel from the skin to the small intestine. Once attached to the gastrointestinal lumen, blood meals and proteolytic enzymes result in iron-deficiency anemia in the host and may lead to weakness, fatigue, and low birth weights in pregnant patients. With prolonged infection or heavy parasitic burden, patients can develop hypoproteinemia, anasarca, and yellowing of the skin known as chlorosis.11 A clinical diagnosis can be made by examining patient stool samples for eggs, and definitive characterization can be made using molecular tools such as polymerase chain reaction.21,22
Common to hookworm infections is the immune reaction, which promotes inflammation with localized eosinophilia and mastocytosis.11 In a clinical biopsy specimen of gut—usually obtained through esophagogastroduodenoscopy— T-helper (Th) 2–type immune (IL-4, IL-5, IL-9 and IL-13), regulatory Th10 (IL-10 and transcription growth factor β), and some evidence of Th1 (interferon gamma and IL-2) cytokines are present, but little evidence of Th17-type immune response was found.23 It is believed that in zoonotic infections, antiparasitic IgE from basophils are somewhat successful at trapping the helminths in the epidermis, but in the anthropophilic species, IgE and Th2 responses are ineffective at clearing the parasite from the gut, and the defeated immune system transitions to a host-tolerance approach of limiting infection.11 It is now believed that this natural armistice can be manipulated into a potential therapy against autoimmune and inflammatory conditions. Intentional infection with zoonotic whipworm or hookworm has been proposed as a mechanism of switching Th1 and Th2 responses to host-tolerant mechanisms in conditions such as Crohn disease and celiac disease,24 and it has even been hypothesized that prior hookworm infection may reduce the chance of developing allergic conditions such as eczema.25
Treatment and Prevention
The World Health Organization and Centers for Disease Control and Prevention recommend a single oral dose of 400 mg albendazole for adults or 10 to 15 mg/kg in children for CLM. A single dose of ivermectin at 12 mg in adults or 150 μg/kg in children can be used as an alternative where albendazole is not available.11 Topical applications of thiabendazole 10% to 15% under occlusion or 3 times daily for 15 days without occlusion also can manage CLM, and pruritus can be treated with topical corticosteroids for symptomatic relief. Oral albendazole 400 mg twice daily or mebendazole 100 mg twice daily for 3 days or a single 500-mg dose, as well as 11 mg/kg (up to a maximum of 1 g) oral pyrantel pamoate once daily for 3 days can be used to treat intestinal hookworm infection, though it should be avoided in pregnancy. Iron deficiency should be managed with supplementation.11
Prevention of hookworm infection is focused around 2 broad public health efforts: mass drug administration programs and the water, sanitation, and hygiene program. In mass drug administration, treatments such as benzimidazoles are given in mass to communities affected by endemic hookworm as a single dose to reduce the burden of disease. Together, these strategies effectively eliminated hookworms in many developed nations, but areas of resurgence are beginning to surface worldwide. With changes in climate, emerging drug resistance, and socioeconomic disparities, particularly affecting the southeast, a resurgence of hookworm has occurred in the United States.26 One recent study demonstrated that almost one-third (19/55) of children sampled in an impoverished area of rural Alabama had hookworm eggs in their stool.27 Furthermore, pets serve not only as zoonotic reservoirs for CLM recurrence but also as vehicles for the evolution of drug-resistant strains, leading some to call for a ban of animals from beaches and playgrounds as well as tightly controlled veterinary programs.5,28 Ubiquitous benzimidazole use in livestock has led to bendazole-resistant strains, and it is likely that with continued and poorly adherent drug use, more zoonotic and anthropophilic drug-resistant strains of hookworm will emerge.29,30
Conclusion
The burden of hookworm infection and CLM is substantial in parts of the United States. Dermatologists play a critical role in the recognition and management of hookworm infection for both treatment of affected patients and the subsequent prevention of its spread. As drug-resistant strains evolve, clinicians, public health officials, and scientists need to continue to work together to prevent and treat hookworm infection.
- Vos T, Abajobir AA, Abate KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1211-1259.
- Bartsch SM, Hotez PJ, Asti L, et al. The global economic and health burden of human hookworm infection. PLoS Negl Trop Dis. 2016;10:E0004922.
- Seguel M, Gottdenker N. The diversity and impact of hookworm infections in wildlife. Int J Parasitol Parasites Wildl. 2017;6:177-194.
- Adams BJ, Peat SM, Dillman AR. Phylogeny and evolution. In: Entomopathogenic Nematodes: Systematics, Phylogeny and Bacterial Symbionts. Brill; 2010:693-733.
- Heukelbach J, Feldmeier H. Epidemiological and clinical characteristics of hookworm-related cutaneous larva migrans. Lancet Infect Dis. 2008;8:302-309.
- Haas W, Haberl B, Idris I, et al. Infective larvae of the human hookworms Necator americanus and Ancylostoma duodenale differ in their orientation behaviour when crawling on surfaces. Parasitol Res. 2005;95:25-29.
- Hotez P, Narasimhan S, Haggerty J, et al. Hyaluronidase from infective Ancylostoma hookworm larvae and its possible function as a virulence factor in tissue invasion and in cutaneous larva migrans. Infect Immun. 1992;60:1018-1023.
- Brooker S, Bethony J, Hotez PJ. Human hookworm infection in the 21st century. Adv Parasitol. 2004;58:197-288.
- Hoagland K, Schad G. Necator americanus and Ancylostoma duodenale: life history parameters and epidemiological implications of two sympatric hookworms of humans. Exp Parasitol. 1978;44:36-49.
- Clements ACA, Alene KA. Global distribution of human hookworm species and differences in their morbidity effects: a systematic review. Lancet Microbe. 2022;3:E72-E79.
- Loukas A, Hotez PJ, Diemert D, et al. Hookworm infection. Nat Rev Dis Primers. 2016;2:1-18.
- Gazzinelli A, Correa-Oliveira R, Yang GJ, et al. A research agenda for helminth diseases of humans: social ecology, environmental determinants, and health systems. PLoS Negl Trop Dis. 2012;6:E1603.
- Starr MC, Montgomery SP. Soil-transmitted helminthiasis in the United States: a systematic review—1940-2010. Am J Trop Med Hyg. 2011;85:680-684.
- Strunz EC, Addiss DG, Stocks ME, et al. Water, sanitation, hygiene, and soil-transmitted helminth infection: a systematic review and metaanalysis. PLoS Med. 2014;11:E1001620.
- Liotta JL, Youn H, Aksel S, et al. Prevalence of Ancylostoma braziliense in dogs from Alachua and Marion Counties, Florida, United States. J Parasitol. 2012;98:1039-1040.
- Hotez PJ, Brooker S, Bethony JM, et al. Hookworm infection. N Engl J Med. 2004;351:799-807.
- Prickett KA, Ferringer TC. What’s eating you? cutaneous larva migrans. Cutis. 2015;95:126-128.
- Feldmeier H, Schuster A. Mini review: hookworm-related cutaneous larva migrans. Eur J Clin Microbiol Infect Dis. 2012;31:915-918.
- Tan SK, Liu TT. Cutaneous larva migrans complicated by Löffler syndrome. Arch Dermatol. 2010;146:210-212.
- Eksomtramage T, Aiempanakit K. Bullous and pustular cutaneous larva migrans: two case reports and a literature review. IDCases. 2018;12:130-132.
- Utzinger J, Rinaldi L, Lohourignon LK, et al. FLOTAC: a new sensitive technique for the diagnosis of hookworm infections in humans. Trans R Soc Trop Med Hyg. 2008;102:84-90.
- Chidambaram M, Parija SC, Toi PC, et al. Evaluation of the utility of conventional polymerase chain reaction for detection and species differentiation in human hookworm infections. Trop Parasitol. 2017;7:111-116.
- Gaze S, McSorley HJ, Daveson J, et al. Characterising the mucosal and systemic immune responses to experimental human hookworm infection. PLoS Pathog. 2012;8:E1002520.
- Croese J, O’Neil J, Masson J, et al. A proof of concept study establishing Necator americanus in Crohn’s patients and reservoir donors. Gut. 2006;55:136-137.
- Mpairwe H, Amoah AS. Parasites and allergy: observations from Africa. Parasite Immunol. 2019;41:E12589.
- Albonico M, Savioli L. Hookworm: a neglected resurgent infection. Editorial. BMJ. 2017;359:j4813.
- McKenna ML, McAtee S, Bryan PE, et al. Human intestinal parasite burden and poor sanitation in rural Alabama. Am J Trop Med Hyg. 2017;97:1623-1628.
- Traversa D. Pet roundworms and hookworms: a continuing need for global worming. Parasit Vectors. 2012;5:1-19.
- Geerts S, Gryseels B. Drug resistance in human helminths: current situation and lessons from livestock. Clin Microbiol Rev. 2000;13:207-222.
- Jimenez Castro PD, Howell SB, Schaefer JJ, et al. Multiple drug resistance in the canine hookworm Ancylostoma caninum: an emerging threat? Parasit Vectors. 2019;12:1-15.
It is estimated that the prevalence of human hookworm infection is approximately 450 million individuals worldwide, representing a substantial global disease burden.1 The annual global public health burden ranges from approximately 2 million to 4 million disability-adjusted life-years and $10 billion to $140 billion in hookwormrelated costs.2 In this article, we discuss the lifecycle, transmission, and disease burden of cutaneous larva migrans (CLM) as well as prevention and treatment strategies.
Background
The Ancylostomatidae nematode family comprises at least 68 known species of hookworm that infect more than 110 different species of mammals.3 Many of these parasites are able to infect more than 1 primary host species, but from a disease perspective they can be classified as either anthropophilic, with humans as the intended host, or zoonotic, with humans as an incidental host. It is important to make this distinction because, though the lifecycles and biology of hookworm species generally are similar, the manifestations of incidental human infection from zoonotic hookworms are different from those of anthropophilic hookworms. Of the anthropophilic species, Necator americanus and Ancylostoma duodenale predominate. In the instance of zoonotic hookworm, dog-infecting A caninum and cat- and doginfecting A braziliense and Uncinaria stenocephala are common causes of incidental human disease.3
The life cycle of Ancylostomatidae organisms is astounding. Through millions of years of co-evolution with mammals,4 these parasitic worms have developed perhaps one of the most circuitous paths to propagate themselves in the natural world. Hookworms start their arduous journey as eggs deposited in soil, sand, and ground vegetation from the feces of infected animals.5 Approximately 1 day after the eggs are deposited, they hatch and begin the larval stage, during which they become infective 1 to 5 weeks later. At this point, the larvae become sensitive to their environment, responding to rising temperatures, increasing carbon dioxide levels, and vibrations in the soil—all of which suggest the presence of a potential host and contribute to a concordant increase in undulatory movement of the larvae.5,6 Here, the most vulnerable tissues include the uncovered soles, palms, and buttocks of host mammals that come into contact with contaminated soil. In an undulating fashion and guided by temperature cues, the larvae locate the skin of the host and utilize a mixture of enzymes including hyaluronidases, metalloprotease, and other proteases to penetrate the epidermis.7 Anthropophilic hookworms such as N americanus and A duodenale will enter the circulatory system; from there, the hookworms migrate through the right-sided cardiopulmonary circuit and eventually ascend into the pulmonary vasculature.8 They then penetrate the lung capillary beds and parenchyma to reach the alveoli, ascend the respiratory tree, and, with the help of the mucociliary escalator, reach the esophagus, where they are swallowed by the host. In the gastrointestinal tract, adult hookworms consume host blood, mate, and lay eggs over a period of approximately 1 to 3 years if left untreated.9 Eggs are laid into the lower gastrointestinal tract, and the journey begins again in feces contacting ground or soil.
Geographic Distribution
Hookworms are found in almost all regions of the world, with species-specific distributions that highlight tropical and subtropical regions. Necator americanus and A duodenale are the most common hookworm species, with the former found predominantly in Southeast Asia and Latin America and the latter in Asia-Pacific regions.10 The highest prevalence of hookworms is in Southeast Asia followed by Sub-Saharan Africa, and the unique climate and soil composition of a region help determine the best environments for specific species of hookworm to thrive.11 In addition, socioeconomics and social determinants of health play a big role in the spread of hookworms, as hygiene practices (eg, wearing clean shoes and clothing, bathing), infrastructure (eg, clean water and streets), and anthelmintic campaigns help reduce transmission.12 Soil-transmitted helminths were once endemic to the southeastern United States, with some reports of approximately 40% of individuals infected in the south in the early 1900s.13 Anthelmintic campaigns such as water, sanitation, and hygiene programs as well as deworming of humans and livestock have proven effective in reducing the prevalence of helminth disease in industrialized nations.13,14 However, zoonotic infections remain a problem in these regions, and in some parts of the United States more than 40% of sampled cats and dogs harbored species such as A braziliense.15
Clinical Manifestation
Initial hookworm infection often goes unnoticed because symptoms can range in severity, but it is characterized by transient ground itch—a local pruritic, erythematous, and papular eruption that develops in response to epidermal penetration.16 Because the larvae must traverse the host from skin to target organs for reproduction over several weeks, iron-deficiency anemia will manifest much later than signs of the initial penetration. In the case of incidental infection from zoonotic Ancylostomatidae organisms, the misguided larvae result in CLM, an often intensely pruritic skin condition that will self-resolve in 2 to 8 weeks with eventual death of the larvae.5
Diagnosis and Pathology of Disease
Zoonotic Hookworm—The major presenting sign of zoonotic hookworm infection is CLM. The diagnosis of CLM usually is made clinically, as the larvae themselves are 0.5 mm thick to 10 mm long (Figure 1) and usually extend several centimeters beyond the dermal lesion, with dermoscopy having limited utility.17 Patients may begin to experience itching as little as 1 hour after hookworm penetration of the skin.18 Once in contact with the skin, the hookworms’ hyaluronidases and proteases are capable of breaking through the epidermis, but zoonotic hookworms typically are unable to penetrate the basal layer of the human epidermis and remain entombed between the stratum granulosum and stratum corneum. With the exception of rare cases of direct or indirect pulmonary involvement resulting in Löffler syndrome,19 the larvae will die within weeks to months, and symptoms will subsequently resolve.
Although the infection generally is self-limiting, the dermatologic manifestations of CLM can be severe and warrant intervention. The lesions start as small reddish papules at the site of penetration (Figure 2), then the hallmark elevated, migrating, serpiginous, urticarial rash develops (Figure 3). Cutaneous larva migrans generally manifests unilaterally and is both erythematous and intensely pruritic. As the larvae migrate, they leave behind 1- to 5-cm tunneled creeping eruptions in their wake. The lesions, which can manifest with pain or be painless, may develop eczematous, bullous, follicular, or impetiginized appearances.20 Atypical manifestations include folliculitis and urticarial plaques.17
cutaneous larval migrans on the palm.
Anthropophilic Hookworm—The lifecycles of N americanus and A duodenale are completed in human infection. Dermatologic manifestations are transient with the development of ground itch at the site of epidermal penetration. The hookworms employ collagenases that allow penetration of the basal layer of the skin, and eosinophilia develops as the parasites travel from the skin to the small intestine. Once attached to the gastrointestinal lumen, blood meals and proteolytic enzymes result in iron-deficiency anemia in the host and may lead to weakness, fatigue, and low birth weights in pregnant patients. With prolonged infection or heavy parasitic burden, patients can develop hypoproteinemia, anasarca, and yellowing of the skin known as chlorosis.11 A clinical diagnosis can be made by examining patient stool samples for eggs, and definitive characterization can be made using molecular tools such as polymerase chain reaction.21,22
Common to hookworm infections is the immune reaction, which promotes inflammation with localized eosinophilia and mastocytosis.11 In a clinical biopsy specimen of gut—usually obtained through esophagogastroduodenoscopy— T-helper (Th) 2–type immune (IL-4, IL-5, IL-9 and IL-13), regulatory Th10 (IL-10 and transcription growth factor β), and some evidence of Th1 (interferon gamma and IL-2) cytokines are present, but little evidence of Th17-type immune response was found.23 It is believed that in zoonotic infections, antiparasitic IgE from basophils are somewhat successful at trapping the helminths in the epidermis, but in the anthropophilic species, IgE and Th2 responses are ineffective at clearing the parasite from the gut, and the defeated immune system transitions to a host-tolerance approach of limiting infection.11 It is now believed that this natural armistice can be manipulated into a potential therapy against autoimmune and inflammatory conditions. Intentional infection with zoonotic whipworm or hookworm has been proposed as a mechanism of switching Th1 and Th2 responses to host-tolerant mechanisms in conditions such as Crohn disease and celiac disease,24 and it has even been hypothesized that prior hookworm infection may reduce the chance of developing allergic conditions such as eczema.25
Treatment and Prevention
The World Health Organization and Centers for Disease Control and Prevention recommend a single oral dose of 400 mg albendazole for adults or 10 to 15 mg/kg in children for CLM. A single dose of ivermectin at 12 mg in adults or 150 μg/kg in children can be used as an alternative where albendazole is not available.11 Topical applications of thiabendazole 10% to 15% under occlusion or 3 times daily for 15 days without occlusion also can manage CLM, and pruritus can be treated with topical corticosteroids for symptomatic relief. Oral albendazole 400 mg twice daily or mebendazole 100 mg twice daily for 3 days or a single 500-mg dose, as well as 11 mg/kg (up to a maximum of 1 g) oral pyrantel pamoate once daily for 3 days can be used to treat intestinal hookworm infection, though it should be avoided in pregnancy. Iron deficiency should be managed with supplementation.11
Prevention of hookworm infection is focused around 2 broad public health efforts: mass drug administration programs and the water, sanitation, and hygiene program. In mass drug administration, treatments such as benzimidazoles are given in mass to communities affected by endemic hookworm as a single dose to reduce the burden of disease. Together, these strategies effectively eliminated hookworms in many developed nations, but areas of resurgence are beginning to surface worldwide. With changes in climate, emerging drug resistance, and socioeconomic disparities, particularly affecting the southeast, a resurgence of hookworm has occurred in the United States.26 One recent study demonstrated that almost one-third (19/55) of children sampled in an impoverished area of rural Alabama had hookworm eggs in their stool.27 Furthermore, pets serve not only as zoonotic reservoirs for CLM recurrence but also as vehicles for the evolution of drug-resistant strains, leading some to call for a ban of animals from beaches and playgrounds as well as tightly controlled veterinary programs.5,28 Ubiquitous benzimidazole use in livestock has led to bendazole-resistant strains, and it is likely that with continued and poorly adherent drug use, more zoonotic and anthropophilic drug-resistant strains of hookworm will emerge.29,30
Conclusion
The burden of hookworm infection and CLM is substantial in parts of the United States. Dermatologists play a critical role in the recognition and management of hookworm infection for both treatment of affected patients and the subsequent prevention of its spread. As drug-resistant strains evolve, clinicians, public health officials, and scientists need to continue to work together to prevent and treat hookworm infection.
It is estimated that the prevalence of human hookworm infection is approximately 450 million individuals worldwide, representing a substantial global disease burden.1 The annual global public health burden ranges from approximately 2 million to 4 million disability-adjusted life-years and $10 billion to $140 billion in hookwormrelated costs.2 In this article, we discuss the lifecycle, transmission, and disease burden of cutaneous larva migrans (CLM) as well as prevention and treatment strategies.
Background
The Ancylostomatidae nematode family comprises at least 68 known species of hookworm that infect more than 110 different species of mammals.3 Many of these parasites are able to infect more than 1 primary host species, but from a disease perspective they can be classified as either anthropophilic, with humans as the intended host, or zoonotic, with humans as an incidental host. It is important to make this distinction because, though the lifecycles and biology of hookworm species generally are similar, the manifestations of incidental human infection from zoonotic hookworms are different from those of anthropophilic hookworms. Of the anthropophilic species, Necator americanus and Ancylostoma duodenale predominate. In the instance of zoonotic hookworm, dog-infecting A caninum and cat- and doginfecting A braziliense and Uncinaria stenocephala are common causes of incidental human disease.3
The life cycle of Ancylostomatidae organisms is astounding. Through millions of years of co-evolution with mammals,4 these parasitic worms have developed perhaps one of the most circuitous paths to propagate themselves in the natural world. Hookworms start their arduous journey as eggs deposited in soil, sand, and ground vegetation from the feces of infected animals.5 Approximately 1 day after the eggs are deposited, they hatch and begin the larval stage, during which they become infective 1 to 5 weeks later. At this point, the larvae become sensitive to their environment, responding to rising temperatures, increasing carbon dioxide levels, and vibrations in the soil—all of which suggest the presence of a potential host and contribute to a concordant increase in undulatory movement of the larvae.5,6 Here, the most vulnerable tissues include the uncovered soles, palms, and buttocks of host mammals that come into contact with contaminated soil. In an undulating fashion and guided by temperature cues, the larvae locate the skin of the host and utilize a mixture of enzymes including hyaluronidases, metalloprotease, and other proteases to penetrate the epidermis.7 Anthropophilic hookworms such as N americanus and A duodenale will enter the circulatory system; from there, the hookworms migrate through the right-sided cardiopulmonary circuit and eventually ascend into the pulmonary vasculature.8 They then penetrate the lung capillary beds and parenchyma to reach the alveoli, ascend the respiratory tree, and, with the help of the mucociliary escalator, reach the esophagus, where they are swallowed by the host. In the gastrointestinal tract, adult hookworms consume host blood, mate, and lay eggs over a period of approximately 1 to 3 years if left untreated.9 Eggs are laid into the lower gastrointestinal tract, and the journey begins again in feces contacting ground or soil.
Geographic Distribution
Hookworms are found in almost all regions of the world, with species-specific distributions that highlight tropical and subtropical regions. Necator americanus and A duodenale are the most common hookworm species, with the former found predominantly in Southeast Asia and Latin America and the latter in Asia-Pacific regions.10 The highest prevalence of hookworms is in Southeast Asia followed by Sub-Saharan Africa, and the unique climate and soil composition of a region help determine the best environments for specific species of hookworm to thrive.11 In addition, socioeconomics and social determinants of health play a big role in the spread of hookworms, as hygiene practices (eg, wearing clean shoes and clothing, bathing), infrastructure (eg, clean water and streets), and anthelmintic campaigns help reduce transmission.12 Soil-transmitted helminths were once endemic to the southeastern United States, with some reports of approximately 40% of individuals infected in the south in the early 1900s.13 Anthelmintic campaigns such as water, sanitation, and hygiene programs as well as deworming of humans and livestock have proven effective in reducing the prevalence of helminth disease in industrialized nations.13,14 However, zoonotic infections remain a problem in these regions, and in some parts of the United States more than 40% of sampled cats and dogs harbored species such as A braziliense.15
Clinical Manifestation
Initial hookworm infection often goes unnoticed because symptoms can range in severity, but it is characterized by transient ground itch—a local pruritic, erythematous, and papular eruption that develops in response to epidermal penetration.16 Because the larvae must traverse the host from skin to target organs for reproduction over several weeks, iron-deficiency anemia will manifest much later than signs of the initial penetration. In the case of incidental infection from zoonotic Ancylostomatidae organisms, the misguided larvae result in CLM, an often intensely pruritic skin condition that will self-resolve in 2 to 8 weeks with eventual death of the larvae.5
Diagnosis and Pathology of Disease
Zoonotic Hookworm—The major presenting sign of zoonotic hookworm infection is CLM. The diagnosis of CLM usually is made clinically, as the larvae themselves are 0.5 mm thick to 10 mm long (Figure 1) and usually extend several centimeters beyond the dermal lesion, with dermoscopy having limited utility.17 Patients may begin to experience itching as little as 1 hour after hookworm penetration of the skin.18 Once in contact with the skin, the hookworms’ hyaluronidases and proteases are capable of breaking through the epidermis, but zoonotic hookworms typically are unable to penetrate the basal layer of the human epidermis and remain entombed between the stratum granulosum and stratum corneum. With the exception of rare cases of direct or indirect pulmonary involvement resulting in Löffler syndrome,19 the larvae will die within weeks to months, and symptoms will subsequently resolve.
Although the infection generally is self-limiting, the dermatologic manifestations of CLM can be severe and warrant intervention. The lesions start as small reddish papules at the site of penetration (Figure 2), then the hallmark elevated, migrating, serpiginous, urticarial rash develops (Figure 3). Cutaneous larva migrans generally manifests unilaterally and is both erythematous and intensely pruritic. As the larvae migrate, they leave behind 1- to 5-cm tunneled creeping eruptions in their wake. The lesions, which can manifest with pain or be painless, may develop eczematous, bullous, follicular, or impetiginized appearances.20 Atypical manifestations include folliculitis and urticarial plaques.17
cutaneous larval migrans on the palm.
Anthropophilic Hookworm—The lifecycles of N americanus and A duodenale are completed in human infection. Dermatologic manifestations are transient with the development of ground itch at the site of epidermal penetration. The hookworms employ collagenases that allow penetration of the basal layer of the skin, and eosinophilia develops as the parasites travel from the skin to the small intestine. Once attached to the gastrointestinal lumen, blood meals and proteolytic enzymes result in iron-deficiency anemia in the host and may lead to weakness, fatigue, and low birth weights in pregnant patients. With prolonged infection or heavy parasitic burden, patients can develop hypoproteinemia, anasarca, and yellowing of the skin known as chlorosis.11 A clinical diagnosis can be made by examining patient stool samples for eggs, and definitive characterization can be made using molecular tools such as polymerase chain reaction.21,22
Common to hookworm infections is the immune reaction, which promotes inflammation with localized eosinophilia and mastocytosis.11 In a clinical biopsy specimen of gut—usually obtained through esophagogastroduodenoscopy— T-helper (Th) 2–type immune (IL-4, IL-5, IL-9 and IL-13), regulatory Th10 (IL-10 and transcription growth factor β), and some evidence of Th1 (interferon gamma and IL-2) cytokines are present, but little evidence of Th17-type immune response was found.23 It is believed that in zoonotic infections, antiparasitic IgE from basophils are somewhat successful at trapping the helminths in the epidermis, but in the anthropophilic species, IgE and Th2 responses are ineffective at clearing the parasite from the gut, and the defeated immune system transitions to a host-tolerance approach of limiting infection.11 It is now believed that this natural armistice can be manipulated into a potential therapy against autoimmune and inflammatory conditions. Intentional infection with zoonotic whipworm or hookworm has been proposed as a mechanism of switching Th1 and Th2 responses to host-tolerant mechanisms in conditions such as Crohn disease and celiac disease,24 and it has even been hypothesized that prior hookworm infection may reduce the chance of developing allergic conditions such as eczema.25
Treatment and Prevention
The World Health Organization and Centers for Disease Control and Prevention recommend a single oral dose of 400 mg albendazole for adults or 10 to 15 mg/kg in children for CLM. A single dose of ivermectin at 12 mg in adults or 150 μg/kg in children can be used as an alternative where albendazole is not available.11 Topical applications of thiabendazole 10% to 15% under occlusion or 3 times daily for 15 days without occlusion also can manage CLM, and pruritus can be treated with topical corticosteroids for symptomatic relief. Oral albendazole 400 mg twice daily or mebendazole 100 mg twice daily for 3 days or a single 500-mg dose, as well as 11 mg/kg (up to a maximum of 1 g) oral pyrantel pamoate once daily for 3 days can be used to treat intestinal hookworm infection, though it should be avoided in pregnancy. Iron deficiency should be managed with supplementation.11
Prevention of hookworm infection is focused around 2 broad public health efforts: mass drug administration programs and the water, sanitation, and hygiene program. In mass drug administration, treatments such as benzimidazoles are given in mass to communities affected by endemic hookworm as a single dose to reduce the burden of disease. Together, these strategies effectively eliminated hookworms in many developed nations, but areas of resurgence are beginning to surface worldwide. With changes in climate, emerging drug resistance, and socioeconomic disparities, particularly affecting the southeast, a resurgence of hookworm has occurred in the United States.26 One recent study demonstrated that almost one-third (19/55) of children sampled in an impoverished area of rural Alabama had hookworm eggs in their stool.27 Furthermore, pets serve not only as zoonotic reservoirs for CLM recurrence but also as vehicles for the evolution of drug-resistant strains, leading some to call for a ban of animals from beaches and playgrounds as well as tightly controlled veterinary programs.5,28 Ubiquitous benzimidazole use in livestock has led to bendazole-resistant strains, and it is likely that with continued and poorly adherent drug use, more zoonotic and anthropophilic drug-resistant strains of hookworm will emerge.29,30
Conclusion
The burden of hookworm infection and CLM is substantial in parts of the United States. Dermatologists play a critical role in the recognition and management of hookworm infection for both treatment of affected patients and the subsequent prevention of its spread. As drug-resistant strains evolve, clinicians, public health officials, and scientists need to continue to work together to prevent and treat hookworm infection.
- Vos T, Abajobir AA, Abate KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1211-1259.
- Bartsch SM, Hotez PJ, Asti L, et al. The global economic and health burden of human hookworm infection. PLoS Negl Trop Dis. 2016;10:E0004922.
- Seguel M, Gottdenker N. The diversity and impact of hookworm infections in wildlife. Int J Parasitol Parasites Wildl. 2017;6:177-194.
- Adams BJ, Peat SM, Dillman AR. Phylogeny and evolution. In: Entomopathogenic Nematodes: Systematics, Phylogeny and Bacterial Symbionts. Brill; 2010:693-733.
- Heukelbach J, Feldmeier H. Epidemiological and clinical characteristics of hookworm-related cutaneous larva migrans. Lancet Infect Dis. 2008;8:302-309.
- Haas W, Haberl B, Idris I, et al. Infective larvae of the human hookworms Necator americanus and Ancylostoma duodenale differ in their orientation behaviour when crawling on surfaces. Parasitol Res. 2005;95:25-29.
- Hotez P, Narasimhan S, Haggerty J, et al. Hyaluronidase from infective Ancylostoma hookworm larvae and its possible function as a virulence factor in tissue invasion and in cutaneous larva migrans. Infect Immun. 1992;60:1018-1023.
- Brooker S, Bethony J, Hotez PJ. Human hookworm infection in the 21st century. Adv Parasitol. 2004;58:197-288.
- Hoagland K, Schad G. Necator americanus and Ancylostoma duodenale: life history parameters and epidemiological implications of two sympatric hookworms of humans. Exp Parasitol. 1978;44:36-49.
- Clements ACA, Alene KA. Global distribution of human hookworm species and differences in their morbidity effects: a systematic review. Lancet Microbe. 2022;3:E72-E79.
- Loukas A, Hotez PJ, Diemert D, et al. Hookworm infection. Nat Rev Dis Primers. 2016;2:1-18.
- Gazzinelli A, Correa-Oliveira R, Yang GJ, et al. A research agenda for helminth diseases of humans: social ecology, environmental determinants, and health systems. PLoS Negl Trop Dis. 2012;6:E1603.
- Starr MC, Montgomery SP. Soil-transmitted helminthiasis in the United States: a systematic review—1940-2010. Am J Trop Med Hyg. 2011;85:680-684.
- Strunz EC, Addiss DG, Stocks ME, et al. Water, sanitation, hygiene, and soil-transmitted helminth infection: a systematic review and metaanalysis. PLoS Med. 2014;11:E1001620.
- Liotta JL, Youn H, Aksel S, et al. Prevalence of Ancylostoma braziliense in dogs from Alachua and Marion Counties, Florida, United States. J Parasitol. 2012;98:1039-1040.
- Hotez PJ, Brooker S, Bethony JM, et al. Hookworm infection. N Engl J Med. 2004;351:799-807.
- Prickett KA, Ferringer TC. What’s eating you? cutaneous larva migrans. Cutis. 2015;95:126-128.
- Feldmeier H, Schuster A. Mini review: hookworm-related cutaneous larva migrans. Eur J Clin Microbiol Infect Dis. 2012;31:915-918.
- Tan SK, Liu TT. Cutaneous larva migrans complicated by Löffler syndrome. Arch Dermatol. 2010;146:210-212.
- Eksomtramage T, Aiempanakit K. Bullous and pustular cutaneous larva migrans: two case reports and a literature review. IDCases. 2018;12:130-132.
- Utzinger J, Rinaldi L, Lohourignon LK, et al. FLOTAC: a new sensitive technique for the diagnosis of hookworm infections in humans. Trans R Soc Trop Med Hyg. 2008;102:84-90.
- Chidambaram M, Parija SC, Toi PC, et al. Evaluation of the utility of conventional polymerase chain reaction for detection and species differentiation in human hookworm infections. Trop Parasitol. 2017;7:111-116.
- Gaze S, McSorley HJ, Daveson J, et al. Characterising the mucosal and systemic immune responses to experimental human hookworm infection. PLoS Pathog. 2012;8:E1002520.
- Croese J, O’Neil J, Masson J, et al. A proof of concept study establishing Necator americanus in Crohn’s patients and reservoir donors. Gut. 2006;55:136-137.
- Mpairwe H, Amoah AS. Parasites and allergy: observations from Africa. Parasite Immunol. 2019;41:E12589.
- Albonico M, Savioli L. Hookworm: a neglected resurgent infection. Editorial. BMJ. 2017;359:j4813.
- McKenna ML, McAtee S, Bryan PE, et al. Human intestinal parasite burden and poor sanitation in rural Alabama. Am J Trop Med Hyg. 2017;97:1623-1628.
- Traversa D. Pet roundworms and hookworms: a continuing need for global worming. Parasit Vectors. 2012;5:1-19.
- Geerts S, Gryseels B. Drug resistance in human helminths: current situation and lessons from livestock. Clin Microbiol Rev. 2000;13:207-222.
- Jimenez Castro PD, Howell SB, Schaefer JJ, et al. Multiple drug resistance in the canine hookworm Ancylostoma caninum: an emerging threat? Parasit Vectors. 2019;12:1-15.
- Vos T, Abajobir AA, Abate KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1211-1259.
- Bartsch SM, Hotez PJ, Asti L, et al. The global economic and health burden of human hookworm infection. PLoS Negl Trop Dis. 2016;10:E0004922.
- Seguel M, Gottdenker N. The diversity and impact of hookworm infections in wildlife. Int J Parasitol Parasites Wildl. 2017;6:177-194.
- Adams BJ, Peat SM, Dillman AR. Phylogeny and evolution. In: Entomopathogenic Nematodes: Systematics, Phylogeny and Bacterial Symbionts. Brill; 2010:693-733.
- Heukelbach J, Feldmeier H. Epidemiological and clinical characteristics of hookworm-related cutaneous larva migrans. Lancet Infect Dis. 2008;8:302-309.
- Haas W, Haberl B, Idris I, et al. Infective larvae of the human hookworms Necator americanus and Ancylostoma duodenale differ in their orientation behaviour when crawling on surfaces. Parasitol Res. 2005;95:25-29.
- Hotez P, Narasimhan S, Haggerty J, et al. Hyaluronidase from infective Ancylostoma hookworm larvae and its possible function as a virulence factor in tissue invasion and in cutaneous larva migrans. Infect Immun. 1992;60:1018-1023.
- Brooker S, Bethony J, Hotez PJ. Human hookworm infection in the 21st century. Adv Parasitol. 2004;58:197-288.
- Hoagland K, Schad G. Necator americanus and Ancylostoma duodenale: life history parameters and epidemiological implications of two sympatric hookworms of humans. Exp Parasitol. 1978;44:36-49.
- Clements ACA, Alene KA. Global distribution of human hookworm species and differences in their morbidity effects: a systematic review. Lancet Microbe. 2022;3:E72-E79.
- Loukas A, Hotez PJ, Diemert D, et al. Hookworm infection. Nat Rev Dis Primers. 2016;2:1-18.
- Gazzinelli A, Correa-Oliveira R, Yang GJ, et al. A research agenda for helminth diseases of humans: social ecology, environmental determinants, and health systems. PLoS Negl Trop Dis. 2012;6:E1603.
- Starr MC, Montgomery SP. Soil-transmitted helminthiasis in the United States: a systematic review—1940-2010. Am J Trop Med Hyg. 2011;85:680-684.
- Strunz EC, Addiss DG, Stocks ME, et al. Water, sanitation, hygiene, and soil-transmitted helminth infection: a systematic review and metaanalysis. PLoS Med. 2014;11:E1001620.
- Liotta JL, Youn H, Aksel S, et al. Prevalence of Ancylostoma braziliense in dogs from Alachua and Marion Counties, Florida, United States. J Parasitol. 2012;98:1039-1040.
- Hotez PJ, Brooker S, Bethony JM, et al. Hookworm infection. N Engl J Med. 2004;351:799-807.
- Prickett KA, Ferringer TC. What’s eating you? cutaneous larva migrans. Cutis. 2015;95:126-128.
- Feldmeier H, Schuster A. Mini review: hookworm-related cutaneous larva migrans. Eur J Clin Microbiol Infect Dis. 2012;31:915-918.
- Tan SK, Liu TT. Cutaneous larva migrans complicated by Löffler syndrome. Arch Dermatol. 2010;146:210-212.
- Eksomtramage T, Aiempanakit K. Bullous and pustular cutaneous larva migrans: two case reports and a literature review. IDCases. 2018;12:130-132.
- Utzinger J, Rinaldi L, Lohourignon LK, et al. FLOTAC: a new sensitive technique for the diagnosis of hookworm infections in humans. Trans R Soc Trop Med Hyg. 2008;102:84-90.
- Chidambaram M, Parija SC, Toi PC, et al. Evaluation of the utility of conventional polymerase chain reaction for detection and species differentiation in human hookworm infections. Trop Parasitol. 2017;7:111-116.
- Gaze S, McSorley HJ, Daveson J, et al. Characterising the mucosal and systemic immune responses to experimental human hookworm infection. PLoS Pathog. 2012;8:E1002520.
- Croese J, O’Neil J, Masson J, et al. A proof of concept study establishing Necator americanus in Crohn’s patients and reservoir donors. Gut. 2006;55:136-137.
- Mpairwe H, Amoah AS. Parasites and allergy: observations from Africa. Parasite Immunol. 2019;41:E12589.
- Albonico M, Savioli L. Hookworm: a neglected resurgent infection. Editorial. BMJ. 2017;359:j4813.
- McKenna ML, McAtee S, Bryan PE, et al. Human intestinal parasite burden and poor sanitation in rural Alabama. Am J Trop Med Hyg. 2017;97:1623-1628.
- Traversa D. Pet roundworms and hookworms: a continuing need for global worming. Parasit Vectors. 2012;5:1-19.
- Geerts S, Gryseels B. Drug resistance in human helminths: current situation and lessons from livestock. Clin Microbiol Rev. 2000;13:207-222.
- Jimenez Castro PD, Howell SB, Schaefer JJ, et al. Multiple drug resistance in the canine hookworm Ancylostoma caninum: an emerging threat? Parasit Vectors. 2019;12:1-15.
What’s Eating You? Hookworm and
Cutaneous Larva Migrans
What’s Eating You? Hookworm and
Cutaneous Larva Migrans
PRACTICE POINTS
- Anthropophilic hookworm infection should be considered with evidence of either transient ground itch or iron-deficient anemia in individuals who go barefoot, permitting ground-to-skin transmission.
- Zoonotic hookworm infection manifests as cutaneous larva migrans, an elevated serpiginous rash that, while usually self-resolving, can be intensely pruritic and should be treated accordingly.
- Considered a neglected tropical disease, hookworm infection still represents an enormous global disease burden. In addition to ongoing afflicted regions, hookworms are making a resurgence in developed nations, and drug-resistant strains have evolved.
Holding RA, SpA Drugs Did Not Improve Antibody Response to COVID Vaccine
WASHINGTON — There is no benefit to interrupting treatment with many of the available targeted synthetic or biologic disease-modifying antirheumatic drugs for rheumatoid arthritis (RA) or spondyloarthritis (SpA) at the time of a repeat COVID-19 vaccine dose, new research found.
In the multicenter, randomized controlled COVID Vaccine Response (COVER) trial of 577 patients with RA or SpA taking either abatacept, Janus kinase (JAK) inhibitors, interleukin (IL)–17 inhibitors, or tumor necrosis factor (TNF) inhibitors, holding those drugs for 2 weeks at the time of COVID-19 vaccination supplemental doses didn’t improve antibody response to the vaccine but did lead to disease flares. Most participants had significant antibody responses to the vaccine, regardless of whether their medication had been held or continued, Jeffrey R. Curtis, MD, the Harbert-Ball Professor of Medicine, Epidemiology, and Computer Science at the University of Alabama at Birmingham, reported at the annual meeting of the American College of Rheumatology (ACR).
Guidelines issued by ACR in 2023 recommended holding abatacept for the COVID vaccine but said that “the task force failed to reach consensus” on whether or not to temporarily interrupt the other medications following primary vaccination or supplemental/booster dosing.
Curtis, who was an author on those guidelines, said in an interview, “to date, we haven’t known whether it might be a good idea to hold certain drugs at the time patients receive their next dose of the COVID vaccine. ... That’s because without direct evidence, you have people trading opinions based on extrapolated data.”
The inability to measure cell-mediated immunity and only humoral (ie, antibody-based) immunity is a limitation in COVER. “Nevertheless, based on what we know now, it isn’t advisable to hold any of the four drug classes that we studied at the time patients receive their next COVID vaccine dose. This finding is in contrast to data from a different trial showing that holding methotrexate for 2 weeks does appear to help in response to COVID-19 vaccination, as well as influenza vaccine,” Curtis said.
Asked to comment, session moderator Elena Myasoedova, MD, PhD, consultant rheumatologist and director of the Inflammatory Arthritis Clinic at the Mayo Clinic, Rochester, Minnesota, said in an interview: “This has been an area of clinical uncertainty. It raises a lot of questions from patients and from physicians alike as to whether or not to hold the medication because the implications are flares, and that’s impactful for patients. Patients care about their RA status and how it is controlled, and if there is no difference, then there is no reason to change the medication regimen.”
To Hold or Not to Hold: COVER Shows It Makes Little Difference to Vaccine Response
In COVER, 128 patients were taking abatacept, 96 IL-17 inhibitors, 237 JAK inhibitors, and 116 TNF inhibitors. The study was conducted within 30 sites of the Excellence Network in Rheumatology, a rheumatology practice–based research network launched in 2021. Participants were identified and enrolled at clinic visits immediately prior to receiving their COVID-19 boosters (in routine settings).
All had previously received two or more doses of the mRNA vaccines made by Pfizer or Moderna. Blood was drawn, and they were randomized 1:1 to either continue or hold their disease medication for 2 weeks following the booster. Blood was collected again at 6 weeks post vaccine.
Anti–receptor-binding domain (RBD) IgG antibody titers increased significantly in all drug categories across both study arms, with no differences between the hold vs continue medication groups, even after adjustments for age, sex, body mass index, methotrexate use, steroid use, and time from booster to measurement. All groups also showed increases in geometric mean fold rise of more than 3%.
Subgroup analyses showed no major differences between antibody responses in the hold vs continue groups. The anti-RBD IgG response was lower for abatacept and JAK inhibitors than for the other two drugs, but there was still no significant benefit to holding them for 2 weeks post vaccination.
Holding Drugs Leads to Disease Flares
On the flip side, there were significant differences between the two groups in their responses to the question: “Did you experience any flare or worsening of your autoimmune disease following your recent COVID-19 booster dose?” Overall, 27% of the hold group responded that they had, compared with just 13% of the continue group (P < .05). This difference was greatest in the JAK inhibitor group (33% vs 9%; P < .05).
Among those reporting flares or worsening disease, both the severity and the duration of the flares were about the same. “Interestingly, the duration is beyond a week for the majority of patients. The reason that’s important is, any symptoms that are so-called flare might simply be reactogenicity symptoms, and that might be confused for flare or disease worsening, but you see that a majority of patients actually have those symptoms extending beyond the week. Most of them are worsening in arthritis, as you might expect,” Curtis said in his presentation.
Asked what they did about the flare, only a minority of patients reported contacting a healthcare provider. In all, 68% of the hold group and 78% of the continue group took no action. That’s good in the sense that most of the flares weren’t severe, but it has implications for research, Curtis pointed out.
“A lot of times in the vaccine literature, people do retrospective chart review by looking to see what the doctor said as to whether the patient had a flare. And what this would tell you is patients may be reporting a lot of flares that their doctor doesn’t know anything about. So if you really want to know whether people are having a flare, even a mild flare, you really have to collect prospective data.”
COVID is Not the Last Pandemic
“These results are reassuring, although I think we need a bit more data on abatacept,” Myasoedova said, adding, “I was also interested in the outcomes, such as severe infections, that actually happened to these patients. What we see in the labs in their immune response is one thing, but then also important is what actually evolves in terms of the outcomes, especially with abatacept.”
Overall, she said, “I think it’s reassuring and definitely informs clinical practice going forward. But then probably we’ll learn more. What we’re hearing is COVID is not the last pandemic.”
The COVER trial receives support from AbbVie, BMS, Eli Lilly, Novartis, and Pfizer. Curtis has received research grants and consulting fees from AbbVie, Amgen, BMS, GSK, Eli Lilly, Novartis, Pfizer, Sanofi, and UCB. Myasoedova has no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — There is no benefit to interrupting treatment with many of the available targeted synthetic or biologic disease-modifying antirheumatic drugs for rheumatoid arthritis (RA) or spondyloarthritis (SpA) at the time of a repeat COVID-19 vaccine dose, new research found.
In the multicenter, randomized controlled COVID Vaccine Response (COVER) trial of 577 patients with RA or SpA taking either abatacept, Janus kinase (JAK) inhibitors, interleukin (IL)–17 inhibitors, or tumor necrosis factor (TNF) inhibitors, holding those drugs for 2 weeks at the time of COVID-19 vaccination supplemental doses didn’t improve antibody response to the vaccine but did lead to disease flares. Most participants had significant antibody responses to the vaccine, regardless of whether their medication had been held or continued, Jeffrey R. Curtis, MD, the Harbert-Ball Professor of Medicine, Epidemiology, and Computer Science at the University of Alabama at Birmingham, reported at the annual meeting of the American College of Rheumatology (ACR).
Guidelines issued by ACR in 2023 recommended holding abatacept for the COVID vaccine but said that “the task force failed to reach consensus” on whether or not to temporarily interrupt the other medications following primary vaccination or supplemental/booster dosing.
Curtis, who was an author on those guidelines, said in an interview, “to date, we haven’t known whether it might be a good idea to hold certain drugs at the time patients receive their next dose of the COVID vaccine. ... That’s because without direct evidence, you have people trading opinions based on extrapolated data.”
The inability to measure cell-mediated immunity and only humoral (ie, antibody-based) immunity is a limitation in COVER. “Nevertheless, based on what we know now, it isn’t advisable to hold any of the four drug classes that we studied at the time patients receive their next COVID vaccine dose. This finding is in contrast to data from a different trial showing that holding methotrexate for 2 weeks does appear to help in response to COVID-19 vaccination, as well as influenza vaccine,” Curtis said.
Asked to comment, session moderator Elena Myasoedova, MD, PhD, consultant rheumatologist and director of the Inflammatory Arthritis Clinic at the Mayo Clinic, Rochester, Minnesota, said in an interview: “This has been an area of clinical uncertainty. It raises a lot of questions from patients and from physicians alike as to whether or not to hold the medication because the implications are flares, and that’s impactful for patients. Patients care about their RA status and how it is controlled, and if there is no difference, then there is no reason to change the medication regimen.”
To Hold or Not to Hold: COVER Shows It Makes Little Difference to Vaccine Response
In COVER, 128 patients were taking abatacept, 96 IL-17 inhibitors, 237 JAK inhibitors, and 116 TNF inhibitors. The study was conducted within 30 sites of the Excellence Network in Rheumatology, a rheumatology practice–based research network launched in 2021. Participants were identified and enrolled at clinic visits immediately prior to receiving their COVID-19 boosters (in routine settings).
All had previously received two or more doses of the mRNA vaccines made by Pfizer or Moderna. Blood was drawn, and they were randomized 1:1 to either continue or hold their disease medication for 2 weeks following the booster. Blood was collected again at 6 weeks post vaccine.
Anti–receptor-binding domain (RBD) IgG antibody titers increased significantly in all drug categories across both study arms, with no differences between the hold vs continue medication groups, even after adjustments for age, sex, body mass index, methotrexate use, steroid use, and time from booster to measurement. All groups also showed increases in geometric mean fold rise of more than 3%.
Subgroup analyses showed no major differences between antibody responses in the hold vs continue groups. The anti-RBD IgG response was lower for abatacept and JAK inhibitors than for the other two drugs, but there was still no significant benefit to holding them for 2 weeks post vaccination.
Holding Drugs Leads to Disease Flares
On the flip side, there were significant differences between the two groups in their responses to the question: “Did you experience any flare or worsening of your autoimmune disease following your recent COVID-19 booster dose?” Overall, 27% of the hold group responded that they had, compared with just 13% of the continue group (P < .05). This difference was greatest in the JAK inhibitor group (33% vs 9%; P < .05).
Among those reporting flares or worsening disease, both the severity and the duration of the flares were about the same. “Interestingly, the duration is beyond a week for the majority of patients. The reason that’s important is, any symptoms that are so-called flare might simply be reactogenicity symptoms, and that might be confused for flare or disease worsening, but you see that a majority of patients actually have those symptoms extending beyond the week. Most of them are worsening in arthritis, as you might expect,” Curtis said in his presentation.
Asked what they did about the flare, only a minority of patients reported contacting a healthcare provider. In all, 68% of the hold group and 78% of the continue group took no action. That’s good in the sense that most of the flares weren’t severe, but it has implications for research, Curtis pointed out.
“A lot of times in the vaccine literature, people do retrospective chart review by looking to see what the doctor said as to whether the patient had a flare. And what this would tell you is patients may be reporting a lot of flares that their doctor doesn’t know anything about. So if you really want to know whether people are having a flare, even a mild flare, you really have to collect prospective data.”
COVID is Not the Last Pandemic
“These results are reassuring, although I think we need a bit more data on abatacept,” Myasoedova said, adding, “I was also interested in the outcomes, such as severe infections, that actually happened to these patients. What we see in the labs in their immune response is one thing, but then also important is what actually evolves in terms of the outcomes, especially with abatacept.”
Overall, she said, “I think it’s reassuring and definitely informs clinical practice going forward. But then probably we’ll learn more. What we’re hearing is COVID is not the last pandemic.”
The COVER trial receives support from AbbVie, BMS, Eli Lilly, Novartis, and Pfizer. Curtis has received research grants and consulting fees from AbbVie, Amgen, BMS, GSK, Eli Lilly, Novartis, Pfizer, Sanofi, and UCB. Myasoedova has no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — There is no benefit to interrupting treatment with many of the available targeted synthetic or biologic disease-modifying antirheumatic drugs for rheumatoid arthritis (RA) or spondyloarthritis (SpA) at the time of a repeat COVID-19 vaccine dose, new research found.
In the multicenter, randomized controlled COVID Vaccine Response (COVER) trial of 577 patients with RA or SpA taking either abatacept, Janus kinase (JAK) inhibitors, interleukin (IL)–17 inhibitors, or tumor necrosis factor (TNF) inhibitors, holding those drugs for 2 weeks at the time of COVID-19 vaccination supplemental doses didn’t improve antibody response to the vaccine but did lead to disease flares. Most participants had significant antibody responses to the vaccine, regardless of whether their medication had been held or continued, Jeffrey R. Curtis, MD, the Harbert-Ball Professor of Medicine, Epidemiology, and Computer Science at the University of Alabama at Birmingham, reported at the annual meeting of the American College of Rheumatology (ACR).
Guidelines issued by ACR in 2023 recommended holding abatacept for the COVID vaccine but said that “the task force failed to reach consensus” on whether or not to temporarily interrupt the other medications following primary vaccination or supplemental/booster dosing.
Curtis, who was an author on those guidelines, said in an interview, “to date, we haven’t known whether it might be a good idea to hold certain drugs at the time patients receive their next dose of the COVID vaccine. ... That’s because without direct evidence, you have people trading opinions based on extrapolated data.”
The inability to measure cell-mediated immunity and only humoral (ie, antibody-based) immunity is a limitation in COVER. “Nevertheless, based on what we know now, it isn’t advisable to hold any of the four drug classes that we studied at the time patients receive their next COVID vaccine dose. This finding is in contrast to data from a different trial showing that holding methotrexate for 2 weeks does appear to help in response to COVID-19 vaccination, as well as influenza vaccine,” Curtis said.
Asked to comment, session moderator Elena Myasoedova, MD, PhD, consultant rheumatologist and director of the Inflammatory Arthritis Clinic at the Mayo Clinic, Rochester, Minnesota, said in an interview: “This has been an area of clinical uncertainty. It raises a lot of questions from patients and from physicians alike as to whether or not to hold the medication because the implications are flares, and that’s impactful for patients. Patients care about their RA status and how it is controlled, and if there is no difference, then there is no reason to change the medication regimen.”
To Hold or Not to Hold: COVER Shows It Makes Little Difference to Vaccine Response
In COVER, 128 patients were taking abatacept, 96 IL-17 inhibitors, 237 JAK inhibitors, and 116 TNF inhibitors. The study was conducted within 30 sites of the Excellence Network in Rheumatology, a rheumatology practice–based research network launched in 2021. Participants were identified and enrolled at clinic visits immediately prior to receiving their COVID-19 boosters (in routine settings).
All had previously received two or more doses of the mRNA vaccines made by Pfizer or Moderna. Blood was drawn, and they were randomized 1:1 to either continue or hold their disease medication for 2 weeks following the booster. Blood was collected again at 6 weeks post vaccine.
Anti–receptor-binding domain (RBD) IgG antibody titers increased significantly in all drug categories across both study arms, with no differences between the hold vs continue medication groups, even after adjustments for age, sex, body mass index, methotrexate use, steroid use, and time from booster to measurement. All groups also showed increases in geometric mean fold rise of more than 3%.
Subgroup analyses showed no major differences between antibody responses in the hold vs continue groups. The anti-RBD IgG response was lower for abatacept and JAK inhibitors than for the other two drugs, but there was still no significant benefit to holding them for 2 weeks post vaccination.
Holding Drugs Leads to Disease Flares
On the flip side, there were significant differences between the two groups in their responses to the question: “Did you experience any flare or worsening of your autoimmune disease following your recent COVID-19 booster dose?” Overall, 27% of the hold group responded that they had, compared with just 13% of the continue group (P < .05). This difference was greatest in the JAK inhibitor group (33% vs 9%; P < .05).
Among those reporting flares or worsening disease, both the severity and the duration of the flares were about the same. “Interestingly, the duration is beyond a week for the majority of patients. The reason that’s important is, any symptoms that are so-called flare might simply be reactogenicity symptoms, and that might be confused for flare or disease worsening, but you see that a majority of patients actually have those symptoms extending beyond the week. Most of them are worsening in arthritis, as you might expect,” Curtis said in his presentation.
Asked what they did about the flare, only a minority of patients reported contacting a healthcare provider. In all, 68% of the hold group and 78% of the continue group took no action. That’s good in the sense that most of the flares weren’t severe, but it has implications for research, Curtis pointed out.
“A lot of times in the vaccine literature, people do retrospective chart review by looking to see what the doctor said as to whether the patient had a flare. And what this would tell you is patients may be reporting a lot of flares that their doctor doesn’t know anything about. So if you really want to know whether people are having a flare, even a mild flare, you really have to collect prospective data.”
COVID is Not the Last Pandemic
“These results are reassuring, although I think we need a bit more data on abatacept,” Myasoedova said, adding, “I was also interested in the outcomes, such as severe infections, that actually happened to these patients. What we see in the labs in their immune response is one thing, but then also important is what actually evolves in terms of the outcomes, especially with abatacept.”
Overall, she said, “I think it’s reassuring and definitely informs clinical practice going forward. But then probably we’ll learn more. What we’re hearing is COVID is not the last pandemic.”
The COVER trial receives support from AbbVie, BMS, Eli Lilly, Novartis, and Pfizer. Curtis has received research grants and consulting fees from AbbVie, Amgen, BMS, GSK, Eli Lilly, Novartis, Pfizer, Sanofi, and UCB. Myasoedova has no disclosures.
A version of this article first appeared on Medscape.com.
FROM ACR 2024
Ob.Gyn. Says Collaboration with Dermatologists Essential for Managing Vulvar Dermatoses
— and she believes collaboration with dermatologists is essential, especially for complex cases in what she calls a neglected, data-poor area of medicine.
She also recommends that dermatologists have a good understanding of the vestibule, “one of the most important structures in vulvar medicine,” and that they become equipped to recognize generalized and localized causes of vulvar pain and/or itch.
“The problem is, we don’t talk about [vulvovaginal pain and itch] ... it’s taboo and we’re not taught about it in medical school,” Cigna, assistant professor of obstetrics and gynecology at The George Washington University (GWU), Washington, DC, said in a grand rounds lecture held recently at the GWU School of Medicine and Health Sciences Department of Dermatology.
“There are dermatologists who don’t have much training in vulvar dermatology, and a lot of gyns don’t get as much training” as they should, she said in an interview after the lecture. “So who’s looking at people’s vulvar skin and figuring out what’s going on and giving them effective treatments and evidence-based education?”
Cigna and dermatologist Emily Murphy, MD, will be co-directors of a joint ob.gyn-dermatology Vulvar Dermatology Clinic at GWU that will be launched in 2025, with monthly clinics for particularly challenging cases where the etiology is unclear or treatment is ineffective. “We want to collaborate in a more systematic way and put our heads together and think creatively about what will improve patient care,” Cigna said in the interview.
Dermatologists have valuable expertise in the immunology and genetic factors involved in skin disorders, Cigna said. Moreover, Murphy, assistant professor of dermatology and director of the Vulvar Health Program at GWU, said in an interview, dermatologists “are comfortable in going to off-label systemic medications that ob.gyns may not use that often” and bring to the table expertise in various types of procedures.
Murphy recently trained with Melissa Mauskar, MD, associate of dermatology and obstetrics and gynecology at the University of Texas Southwestern, Dallas, and founder and director of the Gynecologic Dermatology Clinic there. “It’s so important for dermatologists to be involved. It just takes some extra training that residents aren’t getting right now,” said Murphy, a member of the newly formed Vulvar Dermatoses Research Consortium.
In her grand rounds lecture, Cigna offered pearls to dermatologists for approaching a history and exam and covered highlights of the diagnosis and treatment of various problems, from vulvar Candida infections and lichen simplex chronicus to vulvar lichen sclerosus (LS), vulvar lichen planus (LP), vulvar Crohn’s disease, pudendal neuralgia, and pelvic floor muscle spasm, as well as the role of mast cell proliferation in vulvar issues.
Approaching the History and Exam
A comprehensive history covers the start, duration, and location of pain and/or itching as well as a detailed timeline (such as timing of potential causes, including injuries or births) and symptoms (such as burning, cutting, aching, and stinging). The question of whether pain “is on the outside, at the entrance, or deeper inside” is “crucial, especially for those in dermatology,” Cigna emphasized.
“And if you’re seeing a patient for a vulvar condition, please ask them about sex. Ask, is this affecting your sexual or intimate life with your partner because this can also give you a clue about what’s going on and how you can help them,” she told the audience of dermatologists.
Queries about trauma history (physical and emotional/verbal), competitive sports (such as daily cycling, equestrian, and heavy weight lifting), endometriosis/gynecologic surgery, connective tissue disorders (such as Ehler-Danlos syndrome), and irritable bowel syndrome are all potentially important to consider. It is important to ask about anxiety, depression, and obsessive-compulsive disorder, which do not cause — but are highly associated with — vulvar dermatoses, she said.
A surprisingly large number of people with vulvovaginal issues are being diagnosed with Ehler-Danlos syndrome, so “I’m always asking, are you hypermobile because this might be affecting the musculoskeletal system, which might be affecting the pelvis,” Cigna said. “Anything that affects the pelvis can affect the vulva as well.”
The pelvic examination should be “offered” rather than assumed to be part of the exam, as part of a trauma-informed approach that is crucial for earning trust, she advised. “Just saying, ‘we’re going to talk, and then I can offer you an exam if you like’…patients like it. It helps them feel safer and more open.”
Many diagnoses are differentiated by eliciting pain on the anterior vs the posterior half of the vulvar vestibule — the part of the vulva that lies between the labia minora and is composed of nonkeratinized tissue with embryonic origins in the endoderm. “If you touch on the keratinized skin (of the vulva) and they don’t have pain, but on the vestibule they do have pain, and there is no pain inside the vagina, this suggests there is a vestibular problem,” said Cigna.
Pain/tenderness isolated to the posterior half of the vestibule suggests a muscular cause, and pain in both the posterior and anterior parts of the vestibule suggests a cause that is more systemic or diffuse, which could be a result of a hormonal issue such as one related to oral contraceptives or decreased testosterone, or a nerve-related process.
Cigna uses gentle swipes of a Q-tip moistened with water or gel to examine the vulva rather than a poke or touch, with the exception being the posterior vestibule, which overlies muscle insertion sites. “Make sure to get a baseline in remote areas such as the inner thigh, and always distinguish between ‘scratchy/sensitive’ sensations and pain,” she said, noting the value of having the patient hold a mirror on her inner thigh.
Causes of Vulvar Itch: Infectious and Noninfectious
With vulvar candidiasis, a common infectious cause of vulvar itch, “you have to ask if they’re also itching on the inside because if you treat them with a topical and you don’t treat the vaginal yeast infection that may be co-occurring, they’ll keep reseeding their vulvar skin,” Cigna said, “and it will never be fully treated.”
Candida albicans is the most common cause of vulvar or vulvovaginal candidiasis, and resistance to antifungals has been rising. Non-albicans Candida “tends to have even higher resistance rates,” she said. Ordering a sensitivity panel along with the culture is helpful, but “comprehensive vaginal biome” panels are generally not useful. “It’s hard to correlate the information clinically,” she said, “and there’s not always a lot of information about susceptibilities, which is what I really like to know.”
Cigna’s treatments for vaginal infections include miconazole, terconazole, and fluconazole (and occasionally, itraconazole or voriconazole — a “decision we don’t take lightly”). Vulvar treatments include nystatin ointment, clotrimazole cream, and miconazole cream. Often, optimal treatment involves addressing “both inside and out,” she said, noting the importance of also killing yeast in undergarment fabric.
“In my experience, Diflucan [oral fluconazole] doesn’t treat persistent vulvar cutaneous skin yeast well, so while I might try Diflucan, I typically use something topical as well,” she said. “And with vaginal yeast, we do use boric acid from time to time, especially for non-albicans species because it tends to be a little more effective.”
Noninfectious causes of vulvar itch include allergic, neuropathic, and muscular causes, as well as autoimmune dermatoses and mast cell activation syndrome. Well known in dermatology are acute contact dermatitis and lichen simplex chronicus — both characterized by induration, thickening, and a “puffy” erythematous appearance, and worsening of pruritus at night. What may be less appreciated is the long list of implicated allergens , including Always menstrual pads made of a plastic-containing “dry weave” material, Cigna said. There are at least several cotton-only, low-preservative feminine products available on the market, she noted.
Common Autoimmune Vulvar Dermatoses: LS and LP
Vulvar LS has traditionally been thought to affect mainly prepubertal and postmenopausal women, but the autoimmune condition is now known to affect more reproductive-age people with vulvas than previously appreciated, Cigna said.
And notably, in an observational web-based study of premenopausal women (aged 18-50 years) with biopsy-confirmed vulvar LS, the leading symptom was not itch but dyspareunia and tearing with intercourse. This means “we’re missing people,” said Cigna, an author of the study. “We think the reason we’re not seeing itch as commonly in this population is that itch is likely mediated by the low estrogen state of pre- and postmenopausal people.”
Vulvar LS also occurs in pregnancy, with symptoms that are either stable or decrease during pregnancy and increase in the postpartum period, as demonstrated in a recently published online survey.
Patients with vulvar LS can present with hypopigmentation, lichenification, and scarring and architectural changes, the latter of which can involve clitoral phimosis, labial resorption, and narrowing of the introitus. (The vaginal mucosa is unaffected.) The presentation can be subtle, especially in premenopausal women, and differentiation between LS, vitiligo, and yeast is sometimes necessary.
A timely biopsy-driven definitive diagnosis is important because vulvar LS increases the risk for cancer if it’s not adequately treated and because long-term steroid use can affect the accuracy of pathology reports. “We really care about keeping this disease in remission as much as possible,” Cigna said. Experts in the field recommend long-term maintenance therapy with a mid-ultra-potent steroid one to three times/week or an alternative. “I’ve just started using ruxolitinib cream, a Janus kinase (JAK) inhibitor, and tacrolimus, a calcineurin inhibitor,” she said.
With vulvar LP, based on current evidence, the risk for malignant transformation is low, but “it crosses into the vagina and can cause vaginal adhesions, so if you’re diagnosing someone with lichen planus, you need to make sure you’re talking with them about dilators, and if you’re not comfortable, send them to [gyn],” she said.
The use of vulvoscopy is important for one’s ability to see the fine Wickham’s striae that often characterize vulvar LP, she noted. Medical treatments for vulvar LP include topical calcineurin inhibitors, high-potency steroids, and JAK inhibitors.
Surgical treatment of vulvar granuloma fissuratum caused by vulvar LS is possible (when the patient is in complete remission, to prevent koebnerization), with daily post-op application of clobetasol and retraction of tissues, noted Cigna, the author of a study on vulvar lysis of adhesions.
With both LS and LP, Cigna said, “don’t forget (consideration of) hormones” as an adjunctive treatment, especially in postmenopausal women. “Patients in a low hormone state will have more flares.”
Vulvar Crohn’s
“We all have to know how to look for this,” Cigna said. “Unilateral or asymmetric swelling is classic, but don’t rule out the diagnosis if you see symmetric swelling.” Patients also typically have linear “knife-like” fissures or ulcerations, the vulva “is very indurated,” and “swelling is so intense, the patients are miserable,” she said.
Vulvar Crohn’s disease may precede intestinal disease in 20%-30% of patients, so referral to a gastroenterologist — and ideally subsequent collaboration — is important, as vulvar manifestations are treated with systemic medications typical for Crohn’s.
A biopsy is required for diagnosis, and the pathologist should be advised to look for lichenified squamous mucosa with the Touton giant cell reaction. “Vulvar Crohn’s is a rare enough disorder that if you don’t have an experienced or informed pathologist looking at your specimen, they may miss it because they won’t be looking for it,” Cigna added in the interview. “You should be really clear about what you’re looking for.”
Neuropathic Itch, Pelvic Floor Muscle Spasm
Patients with pudendal neuralgia — caused by an injured, entrapped, or irritated pudendal nerve (originating from S2-S4) — typically present with chronic vulvar and pelvic pain that is often unprovoked and worsens with sitting. Itching upon touch is often another symptom, and some patients describe a foreign body sensation. The cause is often trauma (such as an accident or childbirth-related) as opposed to myofascial irritation, Cigna explained in her lecture.
“Your exam will be largely normal, with no skin findings, so patients will get sent away if you don’t know to look for pudendal neuralgia by pressing on the pudendal nerve or doing (or referring for) a diagnostic nerve block,” Cigna added in the interview.
Persistent genital arousal disorder (PGAD) is “more global” in that it can also originate not only from the pudendal nerve but also from nerve roots higher in the spine or even from the brain. “People feel a sense of arousal, but some describe it as an itch,” Cigna said in her lecture, referring to a 2021 consensus document on PGAD/genito-pelvic dysesthesia by the International Society for the Study of Women’s Sexual Health as a valuable resource for understanding and managing the condition.
Diagnosis and treatment usually start with a pudendal nerve block with a combination of steroid and anesthetic. If this does not relieve arousal/itching, the next step may be an MRI to look higher in the spine.
Pelvic Floor Muscle Spasm
Vulvar pain, skin itching, and irritation can be symptoms of pelvic floor muscle spasm. “Oftentimes people come to me and say, ‘I have a dermatologic problem,’” Cigna said. “The skin may look red and erythematous, but it’s probably more likely a muscle problem when you’re not finding anything, and no amount of steroid will help the itch go away when the problem lies underneath.”
Co-occurring symptoms can include vaginal dryness, clitoral pain, urethral discomfort, bladder pain/irritation, increased urgency, constipation, and anal fissures. The first-line treatment approach is pelvic floor therapy.
“Pelvic floor therapy is not just for incontinence. It’s also for pain and discomfort from muscles,” she said, noting that most patients with vulvar disorders are referred for pelvic floor therapy. “Almost all of them end up having pelvic floor dysfunction because the pelvic floor muscles spasm whenever there’s pain or inflammation.”
A Cautionary Word on Vulvodynia, and a Mast Cell Paradigm to Explore
Vulvodynia is defined as persistent pain of at least 3 months’ duration with no clear cause. “These are the patients with no skin findings,” Cigna said. But in most cases, she said, careful investigation identifies causes that are musculoskeletal, hormonal, or nerve-related.
“It’s a term that’s thrown around a lot — it’s kind of a catchall. Yet it should be a small minority of patients who truly have a diagnosis of vulvodynia,” she said.
In the early stages of investigation is the idea that mast cell proliferation and mast cell activation may play a role in some cases of vulvar and vestibular pain and itching. “We see that some patients with vulvodynia and vestibulodynia have mast cells that are increased in number in the epithelium and beneath the epithelium, and nerve staining shows an increased number of nerve endings traveling into the epithelium,” Cigna said.
“We do diagnose some people clinically” based on urticaria and other symptoms suggestive of mast cell proliferation/activation (such as flushing, abdominal cramping, diarrhea, hypotensive syncope or near syncope, and tachycardia), and “then we send them to the allergist for testing,” Cigna said.
Cigna and Murphy have no relevant financial disclosures.
A version of this article appeared on Medscape.com.
— and she believes collaboration with dermatologists is essential, especially for complex cases in what she calls a neglected, data-poor area of medicine.
She also recommends that dermatologists have a good understanding of the vestibule, “one of the most important structures in vulvar medicine,” and that they become equipped to recognize generalized and localized causes of vulvar pain and/or itch.
“The problem is, we don’t talk about [vulvovaginal pain and itch] ... it’s taboo and we’re not taught about it in medical school,” Cigna, assistant professor of obstetrics and gynecology at The George Washington University (GWU), Washington, DC, said in a grand rounds lecture held recently at the GWU School of Medicine and Health Sciences Department of Dermatology.
“There are dermatologists who don’t have much training in vulvar dermatology, and a lot of gyns don’t get as much training” as they should, she said in an interview after the lecture. “So who’s looking at people’s vulvar skin and figuring out what’s going on and giving them effective treatments and evidence-based education?”
Cigna and dermatologist Emily Murphy, MD, will be co-directors of a joint ob.gyn-dermatology Vulvar Dermatology Clinic at GWU that will be launched in 2025, with monthly clinics for particularly challenging cases where the etiology is unclear or treatment is ineffective. “We want to collaborate in a more systematic way and put our heads together and think creatively about what will improve patient care,” Cigna said in the interview.
Dermatologists have valuable expertise in the immunology and genetic factors involved in skin disorders, Cigna said. Moreover, Murphy, assistant professor of dermatology and director of the Vulvar Health Program at GWU, said in an interview, dermatologists “are comfortable in going to off-label systemic medications that ob.gyns may not use that often” and bring to the table expertise in various types of procedures.
Murphy recently trained with Melissa Mauskar, MD, associate of dermatology and obstetrics and gynecology at the University of Texas Southwestern, Dallas, and founder and director of the Gynecologic Dermatology Clinic there. “It’s so important for dermatologists to be involved. It just takes some extra training that residents aren’t getting right now,” said Murphy, a member of the newly formed Vulvar Dermatoses Research Consortium.
In her grand rounds lecture, Cigna offered pearls to dermatologists for approaching a history and exam and covered highlights of the diagnosis and treatment of various problems, from vulvar Candida infections and lichen simplex chronicus to vulvar lichen sclerosus (LS), vulvar lichen planus (LP), vulvar Crohn’s disease, pudendal neuralgia, and pelvic floor muscle spasm, as well as the role of mast cell proliferation in vulvar issues.
Approaching the History and Exam
A comprehensive history covers the start, duration, and location of pain and/or itching as well as a detailed timeline (such as timing of potential causes, including injuries or births) and symptoms (such as burning, cutting, aching, and stinging). The question of whether pain “is on the outside, at the entrance, or deeper inside” is “crucial, especially for those in dermatology,” Cigna emphasized.
“And if you’re seeing a patient for a vulvar condition, please ask them about sex. Ask, is this affecting your sexual or intimate life with your partner because this can also give you a clue about what’s going on and how you can help them,” she told the audience of dermatologists.
Queries about trauma history (physical and emotional/verbal), competitive sports (such as daily cycling, equestrian, and heavy weight lifting), endometriosis/gynecologic surgery, connective tissue disorders (such as Ehler-Danlos syndrome), and irritable bowel syndrome are all potentially important to consider. It is important to ask about anxiety, depression, and obsessive-compulsive disorder, which do not cause — but are highly associated with — vulvar dermatoses, she said.
A surprisingly large number of people with vulvovaginal issues are being diagnosed with Ehler-Danlos syndrome, so “I’m always asking, are you hypermobile because this might be affecting the musculoskeletal system, which might be affecting the pelvis,” Cigna said. “Anything that affects the pelvis can affect the vulva as well.”
The pelvic examination should be “offered” rather than assumed to be part of the exam, as part of a trauma-informed approach that is crucial for earning trust, she advised. “Just saying, ‘we’re going to talk, and then I can offer you an exam if you like’…patients like it. It helps them feel safer and more open.”
Many diagnoses are differentiated by eliciting pain on the anterior vs the posterior half of the vulvar vestibule — the part of the vulva that lies between the labia minora and is composed of nonkeratinized tissue with embryonic origins in the endoderm. “If you touch on the keratinized skin (of the vulva) and they don’t have pain, but on the vestibule they do have pain, and there is no pain inside the vagina, this suggests there is a vestibular problem,” said Cigna.
Pain/tenderness isolated to the posterior half of the vestibule suggests a muscular cause, and pain in both the posterior and anterior parts of the vestibule suggests a cause that is more systemic or diffuse, which could be a result of a hormonal issue such as one related to oral contraceptives or decreased testosterone, or a nerve-related process.
Cigna uses gentle swipes of a Q-tip moistened with water or gel to examine the vulva rather than a poke or touch, with the exception being the posterior vestibule, which overlies muscle insertion sites. “Make sure to get a baseline in remote areas such as the inner thigh, and always distinguish between ‘scratchy/sensitive’ sensations and pain,” she said, noting the value of having the patient hold a mirror on her inner thigh.
Causes of Vulvar Itch: Infectious and Noninfectious
With vulvar candidiasis, a common infectious cause of vulvar itch, “you have to ask if they’re also itching on the inside because if you treat them with a topical and you don’t treat the vaginal yeast infection that may be co-occurring, they’ll keep reseeding their vulvar skin,” Cigna said, “and it will never be fully treated.”
Candida albicans is the most common cause of vulvar or vulvovaginal candidiasis, and resistance to antifungals has been rising. Non-albicans Candida “tends to have even higher resistance rates,” she said. Ordering a sensitivity panel along with the culture is helpful, but “comprehensive vaginal biome” panels are generally not useful. “It’s hard to correlate the information clinically,” she said, “and there’s not always a lot of information about susceptibilities, which is what I really like to know.”
Cigna’s treatments for vaginal infections include miconazole, terconazole, and fluconazole (and occasionally, itraconazole or voriconazole — a “decision we don’t take lightly”). Vulvar treatments include nystatin ointment, clotrimazole cream, and miconazole cream. Often, optimal treatment involves addressing “both inside and out,” she said, noting the importance of also killing yeast in undergarment fabric.
“In my experience, Diflucan [oral fluconazole] doesn’t treat persistent vulvar cutaneous skin yeast well, so while I might try Diflucan, I typically use something topical as well,” she said. “And with vaginal yeast, we do use boric acid from time to time, especially for non-albicans species because it tends to be a little more effective.”
Noninfectious causes of vulvar itch include allergic, neuropathic, and muscular causes, as well as autoimmune dermatoses and mast cell activation syndrome. Well known in dermatology are acute contact dermatitis and lichen simplex chronicus — both characterized by induration, thickening, and a “puffy” erythematous appearance, and worsening of pruritus at night. What may be less appreciated is the long list of implicated allergens , including Always menstrual pads made of a plastic-containing “dry weave” material, Cigna said. There are at least several cotton-only, low-preservative feminine products available on the market, she noted.
Common Autoimmune Vulvar Dermatoses: LS and LP
Vulvar LS has traditionally been thought to affect mainly prepubertal and postmenopausal women, but the autoimmune condition is now known to affect more reproductive-age people with vulvas than previously appreciated, Cigna said.
And notably, in an observational web-based study of premenopausal women (aged 18-50 years) with biopsy-confirmed vulvar LS, the leading symptom was not itch but dyspareunia and tearing with intercourse. This means “we’re missing people,” said Cigna, an author of the study. “We think the reason we’re not seeing itch as commonly in this population is that itch is likely mediated by the low estrogen state of pre- and postmenopausal people.”
Vulvar LS also occurs in pregnancy, with symptoms that are either stable or decrease during pregnancy and increase in the postpartum period, as demonstrated in a recently published online survey.
Patients with vulvar LS can present with hypopigmentation, lichenification, and scarring and architectural changes, the latter of which can involve clitoral phimosis, labial resorption, and narrowing of the introitus. (The vaginal mucosa is unaffected.) The presentation can be subtle, especially in premenopausal women, and differentiation between LS, vitiligo, and yeast is sometimes necessary.
A timely biopsy-driven definitive diagnosis is important because vulvar LS increases the risk for cancer if it’s not adequately treated and because long-term steroid use can affect the accuracy of pathology reports. “We really care about keeping this disease in remission as much as possible,” Cigna said. Experts in the field recommend long-term maintenance therapy with a mid-ultra-potent steroid one to three times/week or an alternative. “I’ve just started using ruxolitinib cream, a Janus kinase (JAK) inhibitor, and tacrolimus, a calcineurin inhibitor,” she said.
With vulvar LP, based on current evidence, the risk for malignant transformation is low, but “it crosses into the vagina and can cause vaginal adhesions, so if you’re diagnosing someone with lichen planus, you need to make sure you’re talking with them about dilators, and if you’re not comfortable, send them to [gyn],” she said.
The use of vulvoscopy is important for one’s ability to see the fine Wickham’s striae that often characterize vulvar LP, she noted. Medical treatments for vulvar LP include topical calcineurin inhibitors, high-potency steroids, and JAK inhibitors.
Surgical treatment of vulvar granuloma fissuratum caused by vulvar LS is possible (when the patient is in complete remission, to prevent koebnerization), with daily post-op application of clobetasol and retraction of tissues, noted Cigna, the author of a study on vulvar lysis of adhesions.
With both LS and LP, Cigna said, “don’t forget (consideration of) hormones” as an adjunctive treatment, especially in postmenopausal women. “Patients in a low hormone state will have more flares.”
Vulvar Crohn’s
“We all have to know how to look for this,” Cigna said. “Unilateral or asymmetric swelling is classic, but don’t rule out the diagnosis if you see symmetric swelling.” Patients also typically have linear “knife-like” fissures or ulcerations, the vulva “is very indurated,” and “swelling is so intense, the patients are miserable,” she said.
Vulvar Crohn’s disease may precede intestinal disease in 20%-30% of patients, so referral to a gastroenterologist — and ideally subsequent collaboration — is important, as vulvar manifestations are treated with systemic medications typical for Crohn’s.
A biopsy is required for diagnosis, and the pathologist should be advised to look for lichenified squamous mucosa with the Touton giant cell reaction. “Vulvar Crohn’s is a rare enough disorder that if you don’t have an experienced or informed pathologist looking at your specimen, they may miss it because they won’t be looking for it,” Cigna added in the interview. “You should be really clear about what you’re looking for.”
Neuropathic Itch, Pelvic Floor Muscle Spasm
Patients with pudendal neuralgia — caused by an injured, entrapped, or irritated pudendal nerve (originating from S2-S4) — typically present with chronic vulvar and pelvic pain that is often unprovoked and worsens with sitting. Itching upon touch is often another symptom, and some patients describe a foreign body sensation. The cause is often trauma (such as an accident or childbirth-related) as opposed to myofascial irritation, Cigna explained in her lecture.
“Your exam will be largely normal, with no skin findings, so patients will get sent away if you don’t know to look for pudendal neuralgia by pressing on the pudendal nerve or doing (or referring for) a diagnostic nerve block,” Cigna added in the interview.
Persistent genital arousal disorder (PGAD) is “more global” in that it can also originate not only from the pudendal nerve but also from nerve roots higher in the spine or even from the brain. “People feel a sense of arousal, but some describe it as an itch,” Cigna said in her lecture, referring to a 2021 consensus document on PGAD/genito-pelvic dysesthesia by the International Society for the Study of Women’s Sexual Health as a valuable resource for understanding and managing the condition.
Diagnosis and treatment usually start with a pudendal nerve block with a combination of steroid and anesthetic. If this does not relieve arousal/itching, the next step may be an MRI to look higher in the spine.
Pelvic Floor Muscle Spasm
Vulvar pain, skin itching, and irritation can be symptoms of pelvic floor muscle spasm. “Oftentimes people come to me and say, ‘I have a dermatologic problem,’” Cigna said. “The skin may look red and erythematous, but it’s probably more likely a muscle problem when you’re not finding anything, and no amount of steroid will help the itch go away when the problem lies underneath.”
Co-occurring symptoms can include vaginal dryness, clitoral pain, urethral discomfort, bladder pain/irritation, increased urgency, constipation, and anal fissures. The first-line treatment approach is pelvic floor therapy.
“Pelvic floor therapy is not just for incontinence. It’s also for pain and discomfort from muscles,” she said, noting that most patients with vulvar disorders are referred for pelvic floor therapy. “Almost all of them end up having pelvic floor dysfunction because the pelvic floor muscles spasm whenever there’s pain or inflammation.”
A Cautionary Word on Vulvodynia, and a Mast Cell Paradigm to Explore
Vulvodynia is defined as persistent pain of at least 3 months’ duration with no clear cause. “These are the patients with no skin findings,” Cigna said. But in most cases, she said, careful investigation identifies causes that are musculoskeletal, hormonal, or nerve-related.
“It’s a term that’s thrown around a lot — it’s kind of a catchall. Yet it should be a small minority of patients who truly have a diagnosis of vulvodynia,” she said.
In the early stages of investigation is the idea that mast cell proliferation and mast cell activation may play a role in some cases of vulvar and vestibular pain and itching. “We see that some patients with vulvodynia and vestibulodynia have mast cells that are increased in number in the epithelium and beneath the epithelium, and nerve staining shows an increased number of nerve endings traveling into the epithelium,” Cigna said.
“We do diagnose some people clinically” based on urticaria and other symptoms suggestive of mast cell proliferation/activation (such as flushing, abdominal cramping, diarrhea, hypotensive syncope or near syncope, and tachycardia), and “then we send them to the allergist for testing,” Cigna said.
Cigna and Murphy have no relevant financial disclosures.
A version of this article appeared on Medscape.com.
— and she believes collaboration with dermatologists is essential, especially for complex cases in what she calls a neglected, data-poor area of medicine.
She also recommends that dermatologists have a good understanding of the vestibule, “one of the most important structures in vulvar medicine,” and that they become equipped to recognize generalized and localized causes of vulvar pain and/or itch.
“The problem is, we don’t talk about [vulvovaginal pain and itch] ... it’s taboo and we’re not taught about it in medical school,” Cigna, assistant professor of obstetrics and gynecology at The George Washington University (GWU), Washington, DC, said in a grand rounds lecture held recently at the GWU School of Medicine and Health Sciences Department of Dermatology.
“There are dermatologists who don’t have much training in vulvar dermatology, and a lot of gyns don’t get as much training” as they should, she said in an interview after the lecture. “So who’s looking at people’s vulvar skin and figuring out what’s going on and giving them effective treatments and evidence-based education?”
Cigna and dermatologist Emily Murphy, MD, will be co-directors of a joint ob.gyn-dermatology Vulvar Dermatology Clinic at GWU that will be launched in 2025, with monthly clinics for particularly challenging cases where the etiology is unclear or treatment is ineffective. “We want to collaborate in a more systematic way and put our heads together and think creatively about what will improve patient care,” Cigna said in the interview.
Dermatologists have valuable expertise in the immunology and genetic factors involved in skin disorders, Cigna said. Moreover, Murphy, assistant professor of dermatology and director of the Vulvar Health Program at GWU, said in an interview, dermatologists “are comfortable in going to off-label systemic medications that ob.gyns may not use that often” and bring to the table expertise in various types of procedures.
Murphy recently trained with Melissa Mauskar, MD, associate of dermatology and obstetrics and gynecology at the University of Texas Southwestern, Dallas, and founder and director of the Gynecologic Dermatology Clinic there. “It’s so important for dermatologists to be involved. It just takes some extra training that residents aren’t getting right now,” said Murphy, a member of the newly formed Vulvar Dermatoses Research Consortium.
In her grand rounds lecture, Cigna offered pearls to dermatologists for approaching a history and exam and covered highlights of the diagnosis and treatment of various problems, from vulvar Candida infections and lichen simplex chronicus to vulvar lichen sclerosus (LS), vulvar lichen planus (LP), vulvar Crohn’s disease, pudendal neuralgia, and pelvic floor muscle spasm, as well as the role of mast cell proliferation in vulvar issues.
Approaching the History and Exam
A comprehensive history covers the start, duration, and location of pain and/or itching as well as a detailed timeline (such as timing of potential causes, including injuries or births) and symptoms (such as burning, cutting, aching, and stinging). The question of whether pain “is on the outside, at the entrance, or deeper inside” is “crucial, especially for those in dermatology,” Cigna emphasized.
“And if you’re seeing a patient for a vulvar condition, please ask them about sex. Ask, is this affecting your sexual or intimate life with your partner because this can also give you a clue about what’s going on and how you can help them,” she told the audience of dermatologists.
Queries about trauma history (physical and emotional/verbal), competitive sports (such as daily cycling, equestrian, and heavy weight lifting), endometriosis/gynecologic surgery, connective tissue disorders (such as Ehler-Danlos syndrome), and irritable bowel syndrome are all potentially important to consider. It is important to ask about anxiety, depression, and obsessive-compulsive disorder, which do not cause — but are highly associated with — vulvar dermatoses, she said.
A surprisingly large number of people with vulvovaginal issues are being diagnosed with Ehler-Danlos syndrome, so “I’m always asking, are you hypermobile because this might be affecting the musculoskeletal system, which might be affecting the pelvis,” Cigna said. “Anything that affects the pelvis can affect the vulva as well.”
The pelvic examination should be “offered” rather than assumed to be part of the exam, as part of a trauma-informed approach that is crucial for earning trust, she advised. “Just saying, ‘we’re going to talk, and then I can offer you an exam if you like’…patients like it. It helps them feel safer and more open.”
Many diagnoses are differentiated by eliciting pain on the anterior vs the posterior half of the vulvar vestibule — the part of the vulva that lies between the labia minora and is composed of nonkeratinized tissue with embryonic origins in the endoderm. “If you touch on the keratinized skin (of the vulva) and they don’t have pain, but on the vestibule they do have pain, and there is no pain inside the vagina, this suggests there is a vestibular problem,” said Cigna.
Pain/tenderness isolated to the posterior half of the vestibule suggests a muscular cause, and pain in both the posterior and anterior parts of the vestibule suggests a cause that is more systemic or diffuse, which could be a result of a hormonal issue such as one related to oral contraceptives or decreased testosterone, or a nerve-related process.
Cigna uses gentle swipes of a Q-tip moistened with water or gel to examine the vulva rather than a poke or touch, with the exception being the posterior vestibule, which overlies muscle insertion sites. “Make sure to get a baseline in remote areas such as the inner thigh, and always distinguish between ‘scratchy/sensitive’ sensations and pain,” she said, noting the value of having the patient hold a mirror on her inner thigh.
Causes of Vulvar Itch: Infectious and Noninfectious
With vulvar candidiasis, a common infectious cause of vulvar itch, “you have to ask if they’re also itching on the inside because if you treat them with a topical and you don’t treat the vaginal yeast infection that may be co-occurring, they’ll keep reseeding their vulvar skin,” Cigna said, “and it will never be fully treated.”
Candida albicans is the most common cause of vulvar or vulvovaginal candidiasis, and resistance to antifungals has been rising. Non-albicans Candida “tends to have even higher resistance rates,” she said. Ordering a sensitivity panel along with the culture is helpful, but “comprehensive vaginal biome” panels are generally not useful. “It’s hard to correlate the information clinically,” she said, “and there’s not always a lot of information about susceptibilities, which is what I really like to know.”
Cigna’s treatments for vaginal infections include miconazole, terconazole, and fluconazole (and occasionally, itraconazole or voriconazole — a “decision we don’t take lightly”). Vulvar treatments include nystatin ointment, clotrimazole cream, and miconazole cream. Often, optimal treatment involves addressing “both inside and out,” she said, noting the importance of also killing yeast in undergarment fabric.
“In my experience, Diflucan [oral fluconazole] doesn’t treat persistent vulvar cutaneous skin yeast well, so while I might try Diflucan, I typically use something topical as well,” she said. “And with vaginal yeast, we do use boric acid from time to time, especially for non-albicans species because it tends to be a little more effective.”
Noninfectious causes of vulvar itch include allergic, neuropathic, and muscular causes, as well as autoimmune dermatoses and mast cell activation syndrome. Well known in dermatology are acute contact dermatitis and lichen simplex chronicus — both characterized by induration, thickening, and a “puffy” erythematous appearance, and worsening of pruritus at night. What may be less appreciated is the long list of implicated allergens , including Always menstrual pads made of a plastic-containing “dry weave” material, Cigna said. There are at least several cotton-only, low-preservative feminine products available on the market, she noted.
Common Autoimmune Vulvar Dermatoses: LS and LP
Vulvar LS has traditionally been thought to affect mainly prepubertal and postmenopausal women, but the autoimmune condition is now known to affect more reproductive-age people with vulvas than previously appreciated, Cigna said.
And notably, in an observational web-based study of premenopausal women (aged 18-50 years) with biopsy-confirmed vulvar LS, the leading symptom was not itch but dyspareunia and tearing with intercourse. This means “we’re missing people,” said Cigna, an author of the study. “We think the reason we’re not seeing itch as commonly in this population is that itch is likely mediated by the low estrogen state of pre- and postmenopausal people.”
Vulvar LS also occurs in pregnancy, with symptoms that are either stable or decrease during pregnancy and increase in the postpartum period, as demonstrated in a recently published online survey.
Patients with vulvar LS can present with hypopigmentation, lichenification, and scarring and architectural changes, the latter of which can involve clitoral phimosis, labial resorption, and narrowing of the introitus. (The vaginal mucosa is unaffected.) The presentation can be subtle, especially in premenopausal women, and differentiation between LS, vitiligo, and yeast is sometimes necessary.
A timely biopsy-driven definitive diagnosis is important because vulvar LS increases the risk for cancer if it’s not adequately treated and because long-term steroid use can affect the accuracy of pathology reports. “We really care about keeping this disease in remission as much as possible,” Cigna said. Experts in the field recommend long-term maintenance therapy with a mid-ultra-potent steroid one to three times/week or an alternative. “I’ve just started using ruxolitinib cream, a Janus kinase (JAK) inhibitor, and tacrolimus, a calcineurin inhibitor,” she said.
With vulvar LP, based on current evidence, the risk for malignant transformation is low, but “it crosses into the vagina and can cause vaginal adhesions, so if you’re diagnosing someone with lichen planus, you need to make sure you’re talking with them about dilators, and if you’re not comfortable, send them to [gyn],” she said.
The use of vulvoscopy is important for one’s ability to see the fine Wickham’s striae that often characterize vulvar LP, she noted. Medical treatments for vulvar LP include topical calcineurin inhibitors, high-potency steroids, and JAK inhibitors.
Surgical treatment of vulvar granuloma fissuratum caused by vulvar LS is possible (when the patient is in complete remission, to prevent koebnerization), with daily post-op application of clobetasol and retraction of tissues, noted Cigna, the author of a study on vulvar lysis of adhesions.
With both LS and LP, Cigna said, “don’t forget (consideration of) hormones” as an adjunctive treatment, especially in postmenopausal women. “Patients in a low hormone state will have more flares.”
Vulvar Crohn’s
“We all have to know how to look for this,” Cigna said. “Unilateral or asymmetric swelling is classic, but don’t rule out the diagnosis if you see symmetric swelling.” Patients also typically have linear “knife-like” fissures or ulcerations, the vulva “is very indurated,” and “swelling is so intense, the patients are miserable,” she said.
Vulvar Crohn’s disease may precede intestinal disease in 20%-30% of patients, so referral to a gastroenterologist — and ideally subsequent collaboration — is important, as vulvar manifestations are treated with systemic medications typical for Crohn’s.
A biopsy is required for diagnosis, and the pathologist should be advised to look for lichenified squamous mucosa with the Touton giant cell reaction. “Vulvar Crohn’s is a rare enough disorder that if you don’t have an experienced or informed pathologist looking at your specimen, they may miss it because they won’t be looking for it,” Cigna added in the interview. “You should be really clear about what you’re looking for.”
Neuropathic Itch, Pelvic Floor Muscle Spasm
Patients with pudendal neuralgia — caused by an injured, entrapped, or irritated pudendal nerve (originating from S2-S4) — typically present with chronic vulvar and pelvic pain that is often unprovoked and worsens with sitting. Itching upon touch is often another symptom, and some patients describe a foreign body sensation. The cause is often trauma (such as an accident or childbirth-related) as opposed to myofascial irritation, Cigna explained in her lecture.
“Your exam will be largely normal, with no skin findings, so patients will get sent away if you don’t know to look for pudendal neuralgia by pressing on the pudendal nerve or doing (or referring for) a diagnostic nerve block,” Cigna added in the interview.
Persistent genital arousal disorder (PGAD) is “more global” in that it can also originate not only from the pudendal nerve but also from nerve roots higher in the spine or even from the brain. “People feel a sense of arousal, but some describe it as an itch,” Cigna said in her lecture, referring to a 2021 consensus document on PGAD/genito-pelvic dysesthesia by the International Society for the Study of Women’s Sexual Health as a valuable resource for understanding and managing the condition.
Diagnosis and treatment usually start with a pudendal nerve block with a combination of steroid and anesthetic. If this does not relieve arousal/itching, the next step may be an MRI to look higher in the spine.
Pelvic Floor Muscle Spasm
Vulvar pain, skin itching, and irritation can be symptoms of pelvic floor muscle spasm. “Oftentimes people come to me and say, ‘I have a dermatologic problem,’” Cigna said. “The skin may look red and erythematous, but it’s probably more likely a muscle problem when you’re not finding anything, and no amount of steroid will help the itch go away when the problem lies underneath.”
Co-occurring symptoms can include vaginal dryness, clitoral pain, urethral discomfort, bladder pain/irritation, increased urgency, constipation, and anal fissures. The first-line treatment approach is pelvic floor therapy.
“Pelvic floor therapy is not just for incontinence. It’s also for pain and discomfort from muscles,” she said, noting that most patients with vulvar disorders are referred for pelvic floor therapy. “Almost all of them end up having pelvic floor dysfunction because the pelvic floor muscles spasm whenever there’s pain or inflammation.”
A Cautionary Word on Vulvodynia, and a Mast Cell Paradigm to Explore
Vulvodynia is defined as persistent pain of at least 3 months’ duration with no clear cause. “These are the patients with no skin findings,” Cigna said. But in most cases, she said, careful investigation identifies causes that are musculoskeletal, hormonal, or nerve-related.
“It’s a term that’s thrown around a lot — it’s kind of a catchall. Yet it should be a small minority of patients who truly have a diagnosis of vulvodynia,” she said.
In the early stages of investigation is the idea that mast cell proliferation and mast cell activation may play a role in some cases of vulvar and vestibular pain and itching. “We see that some patients with vulvodynia and vestibulodynia have mast cells that are increased in number in the epithelium and beneath the epithelium, and nerve staining shows an increased number of nerve endings traveling into the epithelium,” Cigna said.
“We do diagnose some people clinically” based on urticaria and other symptoms suggestive of mast cell proliferation/activation (such as flushing, abdominal cramping, diarrhea, hypotensive syncope or near syncope, and tachycardia), and “then we send them to the allergist for testing,” Cigna said.
Cigna and Murphy have no relevant financial disclosures.
A version of this article appeared on Medscape.com.
New Strategy Led to Modest Decline in Antibiotic Misuse
TOPLINE:
particularly in general practice.
METHODOLOGY:
- Researchers conducted this study to assess the impact of an intervention on antibiotic prescribing and dispensing for common infections.
- Healthcare professionals from general practice, out-of-hours services, nursing homes, and community pharmacies in France, Greece, Lithuania, Poland, and Spain registered their interactions with patients related to antibiotic prescribing and dispensing both prior to and following the intervention.
- Overall, 407 healthcare professionals participated in the first registration, of whom 345 undertook the intervention and participated in the second registration; they documented 10,744 infections during the initial registration and 10,132 cases during the second period.
- The 5-hour intervention included evaluating and discussing feedback on the outcomes of the initial registration, improving communication skills, and offering communication tools.
- The impact of this intervention was calculated from potential unnecessary antibiotic prescriptions, non–first-line antibiotic choices, and percentage of good and wrong safety advice given for each prescription.
TAKEAWAY:
- General practice clinicians showed a significant overall reduction in unnecessary antibiotic prescriptions from 72.2% during the first registration to 65.2% after the intervention (P < .001), with variations across countries ranging from a 19.9% reduction in Lithuania to a 1.3% increase in Greece.
- Out-of-hours services showed a minimal change in unnecessary antibiotic prescribing from 52.5% to 52.1%, whereas nursing homes showed a slight increase from 56.1% to 58.6%.
- Community pharmacies showed significant improvements, with the provision of correct advice increasing by 17% (P < .001) and safety checks improving from 47% to 55.3% in 1 year (P < .001).
- However, the choice of non–first-line antibiotics significantly increased by 29.2% in the second registration period (P < .001).
IN PRACTICE:
“These findings highlight the need for alternative and tailored approaches in antimicrobial stewardship programs in long-term care facilities, with a greater focus on nurses. This includes implementing hygiene measures and empowering nurses to improve the diagnosis of suspected infections, such as urinary tract infections, while debunking prevalent myths and providing clear-cut information for better management of these common infections,” the authors wrote.
SOURCE:
The study was led by Ana García-Sangenís, of Fundació Institut Universitari per a la Recerca a l’Atenció Primària de Salut Jordi Gol i Gurina, Barcelona, Spain, and was published online on November 12, 2024, in Family Practice.
LIMITATIONS:
The study lacked a control group, which limited the ability to attribute changes solely to the intervention. The voluntary participation of healthcare professionals might have introduced selection bias, as participants might have had a greater interest in quality improvement programs than the general population of healthcare providers. Clinical outcomes were not evaluated, which may have created ambiguity regarding whether complication rates or clinical failures varied between the groups.
DISCLOSURES:
This study received funding from the European Union’s Third Health Programme. One author reported receiving fees from pharmaceutical companies and acting as a member of the board of Steno Diabetes Center, Odense, Denmark.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
particularly in general practice.
METHODOLOGY:
- Researchers conducted this study to assess the impact of an intervention on antibiotic prescribing and dispensing for common infections.
- Healthcare professionals from general practice, out-of-hours services, nursing homes, and community pharmacies in France, Greece, Lithuania, Poland, and Spain registered their interactions with patients related to antibiotic prescribing and dispensing both prior to and following the intervention.
- Overall, 407 healthcare professionals participated in the first registration, of whom 345 undertook the intervention and participated in the second registration; they documented 10,744 infections during the initial registration and 10,132 cases during the second period.
- The 5-hour intervention included evaluating and discussing feedback on the outcomes of the initial registration, improving communication skills, and offering communication tools.
- The impact of this intervention was calculated from potential unnecessary antibiotic prescriptions, non–first-line antibiotic choices, and percentage of good and wrong safety advice given for each prescription.
TAKEAWAY:
- General practice clinicians showed a significant overall reduction in unnecessary antibiotic prescriptions from 72.2% during the first registration to 65.2% after the intervention (P < .001), with variations across countries ranging from a 19.9% reduction in Lithuania to a 1.3% increase in Greece.
- Out-of-hours services showed a minimal change in unnecessary antibiotic prescribing from 52.5% to 52.1%, whereas nursing homes showed a slight increase from 56.1% to 58.6%.
- Community pharmacies showed significant improvements, with the provision of correct advice increasing by 17% (P < .001) and safety checks improving from 47% to 55.3% in 1 year (P < .001).
- However, the choice of non–first-line antibiotics significantly increased by 29.2% in the second registration period (P < .001).
IN PRACTICE:
“These findings highlight the need for alternative and tailored approaches in antimicrobial stewardship programs in long-term care facilities, with a greater focus on nurses. This includes implementing hygiene measures and empowering nurses to improve the diagnosis of suspected infections, such as urinary tract infections, while debunking prevalent myths and providing clear-cut information for better management of these common infections,” the authors wrote.
SOURCE:
The study was led by Ana García-Sangenís, of Fundació Institut Universitari per a la Recerca a l’Atenció Primària de Salut Jordi Gol i Gurina, Barcelona, Spain, and was published online on November 12, 2024, in Family Practice.
LIMITATIONS:
The study lacked a control group, which limited the ability to attribute changes solely to the intervention. The voluntary participation of healthcare professionals might have introduced selection bias, as participants might have had a greater interest in quality improvement programs than the general population of healthcare providers. Clinical outcomes were not evaluated, which may have created ambiguity regarding whether complication rates or clinical failures varied between the groups.
DISCLOSURES:
This study received funding from the European Union’s Third Health Programme. One author reported receiving fees from pharmaceutical companies and acting as a member of the board of Steno Diabetes Center, Odense, Denmark.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
particularly in general practice.
METHODOLOGY:
- Researchers conducted this study to assess the impact of an intervention on antibiotic prescribing and dispensing for common infections.
- Healthcare professionals from general practice, out-of-hours services, nursing homes, and community pharmacies in France, Greece, Lithuania, Poland, and Spain registered their interactions with patients related to antibiotic prescribing and dispensing both prior to and following the intervention.
- Overall, 407 healthcare professionals participated in the first registration, of whom 345 undertook the intervention and participated in the second registration; they documented 10,744 infections during the initial registration and 10,132 cases during the second period.
- The 5-hour intervention included evaluating and discussing feedback on the outcomes of the initial registration, improving communication skills, and offering communication tools.
- The impact of this intervention was calculated from potential unnecessary antibiotic prescriptions, non–first-line antibiotic choices, and percentage of good and wrong safety advice given for each prescription.
TAKEAWAY:
- General practice clinicians showed a significant overall reduction in unnecessary antibiotic prescriptions from 72.2% during the first registration to 65.2% after the intervention (P < .001), with variations across countries ranging from a 19.9% reduction in Lithuania to a 1.3% increase in Greece.
- Out-of-hours services showed a minimal change in unnecessary antibiotic prescribing from 52.5% to 52.1%, whereas nursing homes showed a slight increase from 56.1% to 58.6%.
- Community pharmacies showed significant improvements, with the provision of correct advice increasing by 17% (P < .001) and safety checks improving from 47% to 55.3% in 1 year (P < .001).
- However, the choice of non–first-line antibiotics significantly increased by 29.2% in the second registration period (P < .001).
IN PRACTICE:
“These findings highlight the need for alternative and tailored approaches in antimicrobial stewardship programs in long-term care facilities, with a greater focus on nurses. This includes implementing hygiene measures and empowering nurses to improve the diagnosis of suspected infections, such as urinary tract infections, while debunking prevalent myths and providing clear-cut information for better management of these common infections,” the authors wrote.
SOURCE:
The study was led by Ana García-Sangenís, of Fundació Institut Universitari per a la Recerca a l’Atenció Primària de Salut Jordi Gol i Gurina, Barcelona, Spain, and was published online on November 12, 2024, in Family Practice.
LIMITATIONS:
The study lacked a control group, which limited the ability to attribute changes solely to the intervention. The voluntary participation of healthcare professionals might have introduced selection bias, as participants might have had a greater interest in quality improvement programs than the general population of healthcare providers. Clinical outcomes were not evaluated, which may have created ambiguity regarding whether complication rates or clinical failures varied between the groups.
DISCLOSURES:
This study received funding from the European Union’s Third Health Programme. One author reported receiving fees from pharmaceutical companies and acting as a member of the board of Steno Diabetes Center, Odense, Denmark.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Expert Reviews Options for Revitalizing Dystrophic Nails
LAS VEGAS —
“With the fingernails, we don’t often see onychomycosis, but with toenails, we certainly do,” Tracey C. Vlahovic, DPM, a professor at the Samuel Merritt University College of Podiatric Medicine, Oakland, California, said at the Society of Dermatology Physician Associates (SDPA) 22nd Annual Fall Dermatology Conference. “But toenails are subject to a lot of forces beyond just fungal [infections]. We have the wear and tear of wearing shoes, gait, and other physical activity.”
For example, she continued, some runners develop second-toenail dystrophy “because there’s constant repetitive trauma to the toenail, and [poorly fitting] shoes can contribute to that. Biomechanical issues are a unique consideration when you’re dealing with toenail issues.”
Vlahovic highlighted several options that can help improve the appearance of dystrophic nails as they recover or grow back:
Urea nail preparations: To temporarily soften the nail.
Genadur (hydroxypropyl chitosan): This product “is used mainly for psoriatic nails, but I use it for all different kinds of nail dystrophy,” she said.
DermaNail (acetyl mandelic acid solution): This can be used for brittle nails and fingernails. Vlahovic said she recommends it be used on toenails “in addition to the onychomycosis and other nail dystrophy treatments that I’m doing because it really helps to hydrate the nail unit.”
Kerasal Fungal Nail Renewal (ingredients include propylene glycol, urea, glycerin, and lactic acid): This product is used “for smoothing out the appearance of the nail,” she said.
KeryFlex: Applied in an office setting, this resin-based product restores the appearance of an individual’s natural nails. “It comes in two colors [and] absorbs the shock of what is going on mechanically with the feet,” Vlahovic said. “So, if I’m treating a ballet dancer performing en pointe, or a soccer player, it’s something I can use to protect the nail, but also to make it cosmetically more acceptable.”
NECPro: A nail reconstruction method that involves the use of a composite used mainly by podiatrists, it “helps you not only create a barrier, but to create a natural-looking color that matches your own nail color,” she said.
In Vlahovic’s experience, KeryFlex and NECPro last 6-8 weeks. “You can use nail polish on top of them if you’d like, but they’re basically cosmetic barriers to protect the nail unit,” she said.
Vlahovic has disclosed being a consultant and investigator for Ortho Dermatologics and Sagis Diagnostics.
A version of this article appeared on Medscape.com.
LAS VEGAS —
“With the fingernails, we don’t often see onychomycosis, but with toenails, we certainly do,” Tracey C. Vlahovic, DPM, a professor at the Samuel Merritt University College of Podiatric Medicine, Oakland, California, said at the Society of Dermatology Physician Associates (SDPA) 22nd Annual Fall Dermatology Conference. “But toenails are subject to a lot of forces beyond just fungal [infections]. We have the wear and tear of wearing shoes, gait, and other physical activity.”
For example, she continued, some runners develop second-toenail dystrophy “because there’s constant repetitive trauma to the toenail, and [poorly fitting] shoes can contribute to that. Biomechanical issues are a unique consideration when you’re dealing with toenail issues.”
Vlahovic highlighted several options that can help improve the appearance of dystrophic nails as they recover or grow back:
Urea nail preparations: To temporarily soften the nail.
Genadur (hydroxypropyl chitosan): This product “is used mainly for psoriatic nails, but I use it for all different kinds of nail dystrophy,” she said.
DermaNail (acetyl mandelic acid solution): This can be used for brittle nails and fingernails. Vlahovic said she recommends it be used on toenails “in addition to the onychomycosis and other nail dystrophy treatments that I’m doing because it really helps to hydrate the nail unit.”
Kerasal Fungal Nail Renewal (ingredients include propylene glycol, urea, glycerin, and lactic acid): This product is used “for smoothing out the appearance of the nail,” she said.
KeryFlex: Applied in an office setting, this resin-based product restores the appearance of an individual’s natural nails. “It comes in two colors [and] absorbs the shock of what is going on mechanically with the feet,” Vlahovic said. “So, if I’m treating a ballet dancer performing en pointe, or a soccer player, it’s something I can use to protect the nail, but also to make it cosmetically more acceptable.”
NECPro: A nail reconstruction method that involves the use of a composite used mainly by podiatrists, it “helps you not only create a barrier, but to create a natural-looking color that matches your own nail color,” she said.
In Vlahovic’s experience, KeryFlex and NECPro last 6-8 weeks. “You can use nail polish on top of them if you’d like, but they’re basically cosmetic barriers to protect the nail unit,” she said.
Vlahovic has disclosed being a consultant and investigator for Ortho Dermatologics and Sagis Diagnostics.
A version of this article appeared on Medscape.com.
LAS VEGAS —
“With the fingernails, we don’t often see onychomycosis, but with toenails, we certainly do,” Tracey C. Vlahovic, DPM, a professor at the Samuel Merritt University College of Podiatric Medicine, Oakland, California, said at the Society of Dermatology Physician Associates (SDPA) 22nd Annual Fall Dermatology Conference. “But toenails are subject to a lot of forces beyond just fungal [infections]. We have the wear and tear of wearing shoes, gait, and other physical activity.”
For example, she continued, some runners develop second-toenail dystrophy “because there’s constant repetitive trauma to the toenail, and [poorly fitting] shoes can contribute to that. Biomechanical issues are a unique consideration when you’re dealing with toenail issues.”
Vlahovic highlighted several options that can help improve the appearance of dystrophic nails as they recover or grow back:
Urea nail preparations: To temporarily soften the nail.
Genadur (hydroxypropyl chitosan): This product “is used mainly for psoriatic nails, but I use it for all different kinds of nail dystrophy,” she said.
DermaNail (acetyl mandelic acid solution): This can be used for brittle nails and fingernails. Vlahovic said she recommends it be used on toenails “in addition to the onychomycosis and other nail dystrophy treatments that I’m doing because it really helps to hydrate the nail unit.”
Kerasal Fungal Nail Renewal (ingredients include propylene glycol, urea, glycerin, and lactic acid): This product is used “for smoothing out the appearance of the nail,” she said.
KeryFlex: Applied in an office setting, this resin-based product restores the appearance of an individual’s natural nails. “It comes in two colors [and] absorbs the shock of what is going on mechanically with the feet,” Vlahovic said. “So, if I’m treating a ballet dancer performing en pointe, or a soccer player, it’s something I can use to protect the nail, but also to make it cosmetically more acceptable.”
NECPro: A nail reconstruction method that involves the use of a composite used mainly by podiatrists, it “helps you not only create a barrier, but to create a natural-looking color that matches your own nail color,” she said.
In Vlahovic’s experience, KeryFlex and NECPro last 6-8 weeks. “You can use nail polish on top of them if you’d like, but they’re basically cosmetic barriers to protect the nail unit,” she said.
Vlahovic has disclosed being a consultant and investigator for Ortho Dermatologics and Sagis Diagnostics.
A version of this article appeared on Medscape.com.
FROM SDPA 24
Does Antibiotic Use During Influenza Infection Worsen Lung Immunity?
TOPLINE:
Antibiotic use during influenza infection increases lung eosinophils, impairing immunity against secondary bacterial pneumonia. This study highlights the detrimental effects of antibiotics on lung health during viral infections.
METHODOLOGY:
- Researchers conducted a murine model study to evaluate the impact of antibiotic use during influenza infection on lung immunity. Mice were treated with a broad-spectrum antibiotic cocktail (vancomycin, neomycin, ampicillin, and metronidazole) starting 7 days before influenza infection.
- The study included intranasal infection with influenza virus followed by a secondary challenge with methicillin-resistant Staphylococcus aureus (MRSA).
- Finally, in sub-study, a total of three cohorts of hospitalized patients were evaluated to correlate eosinophil levels with antibiotic use, systemic inflammation, and outcomes.
TAKEAWAY:
- Antibiotic use during influenza infection impairs lung immunity, leading to increased lung eosinophils and reduced macrophage function.
- The study found that antibiotic treatment during influenza infection caused fungal dysbiosis, driving lung eosinophilia and impairing MRSA clearance.
- The detrimental effects of antibiotics on lung immunity were specific to the two-hit model of influenza followed by MRSA infection in mice.
- In hospitalized patients, eosinophil levels positively correlated with antibiotic use, systemic inflammation, and worsened outcomes.
IN PRACTICE:
“Our study highlights the pernicious effects of antibiotic use during viral infections and defines a mechanism whereby antibiotics perturb the gut mycobiome and result in lung eosinophilia. In turn, lung eosinophils, via release of MBP-1, suppress alveolar macrophage clearance of bacteria,” the authors of the study wrote.
SOURCE:
This study was led by Marilia Sanches Santos Rizzo Zuttion, Cedars-Sinai Medical Center in Los Angeles. It was published online in The Journal of Clinical Investigation.
LIMITATIONS:
This study’s limitations included the use of a murine model, which may not fully replicate human immune responses. Additionally, the study focused on a specific antibiotic cocktail, and results may vary with different antibiotics. The findings were also specific to the two-hit model of influenza followed by MRSA infection, limiting generalizability to other infections.
DISCLOSURES:
This study was supported by grants from the National Institutes of Health. Marilia Sanches Santos Rizzo Zuttion received research funding from Pfizer Inc. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Antibiotic use during influenza infection increases lung eosinophils, impairing immunity against secondary bacterial pneumonia. This study highlights the detrimental effects of antibiotics on lung health during viral infections.
METHODOLOGY:
- Researchers conducted a murine model study to evaluate the impact of antibiotic use during influenza infection on lung immunity. Mice were treated with a broad-spectrum antibiotic cocktail (vancomycin, neomycin, ampicillin, and metronidazole) starting 7 days before influenza infection.
- The study included intranasal infection with influenza virus followed by a secondary challenge with methicillin-resistant Staphylococcus aureus (MRSA).
- Finally, in sub-study, a total of three cohorts of hospitalized patients were evaluated to correlate eosinophil levels with antibiotic use, systemic inflammation, and outcomes.
TAKEAWAY:
- Antibiotic use during influenza infection impairs lung immunity, leading to increased lung eosinophils and reduced macrophage function.
- The study found that antibiotic treatment during influenza infection caused fungal dysbiosis, driving lung eosinophilia and impairing MRSA clearance.
- The detrimental effects of antibiotics on lung immunity were specific to the two-hit model of influenza followed by MRSA infection in mice.
- In hospitalized patients, eosinophil levels positively correlated with antibiotic use, systemic inflammation, and worsened outcomes.
IN PRACTICE:
“Our study highlights the pernicious effects of antibiotic use during viral infections and defines a mechanism whereby antibiotics perturb the gut mycobiome and result in lung eosinophilia. In turn, lung eosinophils, via release of MBP-1, suppress alveolar macrophage clearance of bacteria,” the authors of the study wrote.
SOURCE:
This study was led by Marilia Sanches Santos Rizzo Zuttion, Cedars-Sinai Medical Center in Los Angeles. It was published online in The Journal of Clinical Investigation.
LIMITATIONS:
This study’s limitations included the use of a murine model, which may not fully replicate human immune responses. Additionally, the study focused on a specific antibiotic cocktail, and results may vary with different antibiotics. The findings were also specific to the two-hit model of influenza followed by MRSA infection, limiting generalizability to other infections.
DISCLOSURES:
This study was supported by grants from the National Institutes of Health. Marilia Sanches Santos Rizzo Zuttion received research funding from Pfizer Inc. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Antibiotic use during influenza infection increases lung eosinophils, impairing immunity against secondary bacterial pneumonia. This study highlights the detrimental effects of antibiotics on lung health during viral infections.
METHODOLOGY:
- Researchers conducted a murine model study to evaluate the impact of antibiotic use during influenza infection on lung immunity. Mice were treated with a broad-spectrum antibiotic cocktail (vancomycin, neomycin, ampicillin, and metronidazole) starting 7 days before influenza infection.
- The study included intranasal infection with influenza virus followed by a secondary challenge with methicillin-resistant Staphylococcus aureus (MRSA).
- Finally, in sub-study, a total of three cohorts of hospitalized patients were evaluated to correlate eosinophil levels with antibiotic use, systemic inflammation, and outcomes.
TAKEAWAY:
- Antibiotic use during influenza infection impairs lung immunity, leading to increased lung eosinophils and reduced macrophage function.
- The study found that antibiotic treatment during influenza infection caused fungal dysbiosis, driving lung eosinophilia and impairing MRSA clearance.
- The detrimental effects of antibiotics on lung immunity were specific to the two-hit model of influenza followed by MRSA infection in mice.
- In hospitalized patients, eosinophil levels positively correlated with antibiotic use, systemic inflammation, and worsened outcomes.
IN PRACTICE:
“Our study highlights the pernicious effects of antibiotic use during viral infections and defines a mechanism whereby antibiotics perturb the gut mycobiome and result in lung eosinophilia. In turn, lung eosinophils, via release of MBP-1, suppress alveolar macrophage clearance of bacteria,” the authors of the study wrote.
SOURCE:
This study was led by Marilia Sanches Santos Rizzo Zuttion, Cedars-Sinai Medical Center in Los Angeles. It was published online in The Journal of Clinical Investigation.
LIMITATIONS:
This study’s limitations included the use of a murine model, which may not fully replicate human immune responses. Additionally, the study focused on a specific antibiotic cocktail, and results may vary with different antibiotics. The findings were also specific to the two-hit model of influenza followed by MRSA infection, limiting generalizability to other infections.
DISCLOSURES:
This study was supported by grants from the National Institutes of Health. Marilia Sanches Santos Rizzo Zuttion received research funding from Pfizer Inc. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Periodontitis Management: GPs Should Play a Role
Periodontitis is a chronic inflammatory disease that triggers a local immuno-inflammatory response, potentially leading to periodontal tissue destruction and tooth loss. Affecting 1.1 billion people worldwide, periodontitis is recognized as a significant public health issue. It is also linked to a number of other conditions, such as diabetes, cardiovascular disease, and respiratory disorders. The European Federation of Periodontology recently published a consensus report recommending that the optimal management of periodontitis should involve a collaboration between general practitioners (GPs) and oral health professionals.
Diabetes and Periodontitis
A bidirectional association exists between diabetes and periodontitis. Hyperglycemia accelerates periodontitis progression by promoting inflammation and hindering the healing process, while periodontitis is associated with higher hemoglobin A1c levels in patients with diabetes and an increased risk for diabetes development in others. Intervention studies have demonstrated the positive effect of glycemic control on periodontitis and vice versa, with periodontal treatment improving A1c levels.
GPs can raise awareness of the links between these conditions as well as emphasize the benefits of addressing both metabolic and periodontal abnormalities. They should refer patients with diabetes to oral health specialists and look for signs of periodontitis, such as bleeding gums and loose teeth, in patients with diabetes and those with prediabetes.
Cardiovascular Diseases and Periodontitis
Cardiovascular diseases and periodontitis are linked by their epidemiological associations and common biologic mechanisms. This connection can be explained by some of their shared risk factors, such as smoking and systemic inflammatory pathways. Although no intervention studies have shown a direct reduction in cardiovascular risk from periodontal care, two studies have demonstrated improvements in surrogate markers such as blood pressure and arterial stiffness. GPs should inquire about symptoms of periodontitis in cardiovascular patients and, if necessary, refer them to oral health specialists. Periodontal treatments, whether surgical or nonsurgical, pose no risk for patients receiving well-managed secondary preventive treatments.
Respiratory Diseases and Periodontitis
The primary evidence linking periodontitis with chronic respiratory diseases concerns chronic obstructive pulmonary disease (COPD). Individuals with periodontitis have a 33% higher risk of developing COPD, and patients with COPD and periodontitis may experience a greater decline in lung function. An established association also exists between periodontitis and obstructive sleep apnea, although the data remain inconclusive regarding a link with asthma. GPs should encourage patients with COPD to quit smoking, as it benefits both respiratory and oral health.
Finally, based on meta-analyses of COVID-19, experts note significant associations between periodontitis and the need for assisted ventilation or the risk for death during a COVID-19 infection.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Periodontitis is a chronic inflammatory disease that triggers a local immuno-inflammatory response, potentially leading to periodontal tissue destruction and tooth loss. Affecting 1.1 billion people worldwide, periodontitis is recognized as a significant public health issue. It is also linked to a number of other conditions, such as diabetes, cardiovascular disease, and respiratory disorders. The European Federation of Periodontology recently published a consensus report recommending that the optimal management of periodontitis should involve a collaboration between general practitioners (GPs) and oral health professionals.
Diabetes and Periodontitis
A bidirectional association exists between diabetes and periodontitis. Hyperglycemia accelerates periodontitis progression by promoting inflammation and hindering the healing process, while periodontitis is associated with higher hemoglobin A1c levels in patients with diabetes and an increased risk for diabetes development in others. Intervention studies have demonstrated the positive effect of glycemic control on periodontitis and vice versa, with periodontal treatment improving A1c levels.
GPs can raise awareness of the links between these conditions as well as emphasize the benefits of addressing both metabolic and periodontal abnormalities. They should refer patients with diabetes to oral health specialists and look for signs of periodontitis, such as bleeding gums and loose teeth, in patients with diabetes and those with prediabetes.
Cardiovascular Diseases and Periodontitis
Cardiovascular diseases and periodontitis are linked by their epidemiological associations and common biologic mechanisms. This connection can be explained by some of their shared risk factors, such as smoking and systemic inflammatory pathways. Although no intervention studies have shown a direct reduction in cardiovascular risk from periodontal care, two studies have demonstrated improvements in surrogate markers such as blood pressure and arterial stiffness. GPs should inquire about symptoms of periodontitis in cardiovascular patients and, if necessary, refer them to oral health specialists. Periodontal treatments, whether surgical or nonsurgical, pose no risk for patients receiving well-managed secondary preventive treatments.
Respiratory Diseases and Periodontitis
The primary evidence linking periodontitis with chronic respiratory diseases concerns chronic obstructive pulmonary disease (COPD). Individuals with periodontitis have a 33% higher risk of developing COPD, and patients with COPD and periodontitis may experience a greater decline in lung function. An established association also exists between periodontitis and obstructive sleep apnea, although the data remain inconclusive regarding a link with asthma. GPs should encourage patients with COPD to quit smoking, as it benefits both respiratory and oral health.
Finally, based on meta-analyses of COVID-19, experts note significant associations between periodontitis and the need for assisted ventilation or the risk for death during a COVID-19 infection.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Periodontitis is a chronic inflammatory disease that triggers a local immuno-inflammatory response, potentially leading to periodontal tissue destruction and tooth loss. Affecting 1.1 billion people worldwide, periodontitis is recognized as a significant public health issue. It is also linked to a number of other conditions, such as diabetes, cardiovascular disease, and respiratory disorders. The European Federation of Periodontology recently published a consensus report recommending that the optimal management of periodontitis should involve a collaboration between general practitioners (GPs) and oral health professionals.
Diabetes and Periodontitis
A bidirectional association exists between diabetes and periodontitis. Hyperglycemia accelerates periodontitis progression by promoting inflammation and hindering the healing process, while periodontitis is associated with higher hemoglobin A1c levels in patients with diabetes and an increased risk for diabetes development in others. Intervention studies have demonstrated the positive effect of glycemic control on periodontitis and vice versa, with periodontal treatment improving A1c levels.
GPs can raise awareness of the links between these conditions as well as emphasize the benefits of addressing both metabolic and periodontal abnormalities. They should refer patients with diabetes to oral health specialists and look for signs of periodontitis, such as bleeding gums and loose teeth, in patients with diabetes and those with prediabetes.
Cardiovascular Diseases and Periodontitis
Cardiovascular diseases and periodontitis are linked by their epidemiological associations and common biologic mechanisms. This connection can be explained by some of their shared risk factors, such as smoking and systemic inflammatory pathways. Although no intervention studies have shown a direct reduction in cardiovascular risk from periodontal care, two studies have demonstrated improvements in surrogate markers such as blood pressure and arterial stiffness. GPs should inquire about symptoms of periodontitis in cardiovascular patients and, if necessary, refer them to oral health specialists. Periodontal treatments, whether surgical or nonsurgical, pose no risk for patients receiving well-managed secondary preventive treatments.
Respiratory Diseases and Periodontitis
The primary evidence linking periodontitis with chronic respiratory diseases concerns chronic obstructive pulmonary disease (COPD). Individuals with periodontitis have a 33% higher risk of developing COPD, and patients with COPD and periodontitis may experience a greater decline in lung function. An established association also exists between periodontitis and obstructive sleep apnea, although the data remain inconclusive regarding a link with asthma. GPs should encourage patients with COPD to quit smoking, as it benefits both respiratory and oral health.
Finally, based on meta-analyses of COVID-19, experts note significant associations between periodontitis and the need for assisted ventilation or the risk for death during a COVID-19 infection.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.