User login
Could Targeting ‘Zombie Cells’ Extend a Healthy Lifespan?
What if a drug could help you live a longer, healthier life?
Scientists at the University of Connecticut are working on it. In a new study in Cell Metabolism, researchers described how to target specific cells to extend the lifespan and improve the health of mice late in life.
The study builds on a growing body of research, mostly in animals, testing interventions to slow aging and prolong health span, the length of time that one is not just alive but also healthy.
“Aging is the most important risk factor for every disease that we deal with in adult human beings,” said cardiologist Douglas Vaughan, MD, director of the Potocsnak Longevity Institute at Northwestern University’s Feinberg School of Medicine, Chicago. (Dr. Vaughan was not involved in the new study.) “So the big hypothesis is: If we could slow down aging just a little bit, we can push back the onset of disease.”
Senescent cells — or “zombie cells” — secrete harmful substances that disrupt tissue functioning. They’ve been linked to chronic inflammation, tissue damage, and the development of age-related diseases.
Senescence can be characterized by the accumulation of cells with high levels of specific markers like p21, or p21high cells. Almost any cell can become a p21high cell, and they accumulate with age, said Ming Xu, PhD, a professor at the UConn Center on Aging, UConn Health, Farmington, Connecticut, who led the study.
By targeting and eliminating p21high senescent cells, Dr. Xu hopes to develop novel therapies that might help people live longer and enjoy more years in good health.
Such a treatment could be ready for human trials in 2-5 years, Dr. Xu said.
What the Researchers Did
Xu and colleagues used genetic engineering to eliminate p21high cells in mice, introducing into their genome something they describe as an inducible “suicide gene.” Giving the mice a certain drug (a low dose of tamoxifen) activated the suicide gene in all p21high cells, causing them to die. Administering this treatment once a month, from age 20 months (older age) until the end of life, significantly extended the rodents’ lifespan, reduced inflammation, and decreased gene activity linked to aging.
Treated mice lived, on average, for 33 months — 3 months longer than the untreated mice. The oldest treated mouse lived to 43 months — roughly 130 in human years.
But the treated mice didn’t just live longer; they were also healthier. In humans, walking speed and grip strength can be clues of overall health and vitality. The old, treated mice were able to walk faster and grip objects with greater strength than untreated mice of the same age.
Dr. Xu’s lab is now testing drugs that target p21high cells in hopes of finding one that would work in humans. Leveraging immunotherapy technology to target these cells could be another option, Dr. Xu said.
The team also plans to test whether eliminating p21high cells could prevent or alleviate diabetes or Alzheimer’s disease.
Challenges and Criticisms
The research provides “important evidence that targeting senescence and the molecular components of that pathway might provide some benefit in the long term,” Dr. Vaughan said.
But killing senescent cells could come with downsides.
“Senescence protects us from hyperproliferative responses,” potentially blocking cells from becoming malignant, Dr. Vaughan said. “There’s this effect on aging that is desirable, but at the same time, you may enhance your risk of cancer or malignancy or excessive proliferation in some cells.”
And of course, we don’t necessarily need drugs to prolong healthy life, Dr. Vaughan pointed out.
For many people, a long healthy life is already within reach. Humans live longer on average than they used to, and simple lifestyle choices — nourishing your body well, staying active, and maintaining a healthy weight — can increase one’s chances of good health.
The most consistently demonstrated intervention for extending lifespan “in almost every animal species is caloric restriction,” Dr. Vaughan said. (Dr. Xu’s team is also investigating whether fasting and exercise can lead to a decrease in p21high cells.)
As for brain health, Dr. Vaughan and colleagues at Northwestern are studying “super agers,” people who are cognitively intact into their 90s.
“The one single thing that they found that contributes to that process, and contributes to that success, is really a social network and human bonds and interaction,” Dr. Vaughan said.
A version of this article appeared on Medscape.com.
What if a drug could help you live a longer, healthier life?
Scientists at the University of Connecticut are working on it. In a new study in Cell Metabolism, researchers described how to target specific cells to extend the lifespan and improve the health of mice late in life.
The study builds on a growing body of research, mostly in animals, testing interventions to slow aging and prolong health span, the length of time that one is not just alive but also healthy.
“Aging is the most important risk factor for every disease that we deal with in adult human beings,” said cardiologist Douglas Vaughan, MD, director of the Potocsnak Longevity Institute at Northwestern University’s Feinberg School of Medicine, Chicago. (Dr. Vaughan was not involved in the new study.) “So the big hypothesis is: If we could slow down aging just a little bit, we can push back the onset of disease.”
Senescent cells — or “zombie cells” — secrete harmful substances that disrupt tissue functioning. They’ve been linked to chronic inflammation, tissue damage, and the development of age-related diseases.
Senescence can be characterized by the accumulation of cells with high levels of specific markers like p21, or p21high cells. Almost any cell can become a p21high cell, and they accumulate with age, said Ming Xu, PhD, a professor at the UConn Center on Aging, UConn Health, Farmington, Connecticut, who led the study.
By targeting and eliminating p21high senescent cells, Dr. Xu hopes to develop novel therapies that might help people live longer and enjoy more years in good health.
Such a treatment could be ready for human trials in 2-5 years, Dr. Xu said.
What the Researchers Did
Xu and colleagues used genetic engineering to eliminate p21high cells in mice, introducing into their genome something they describe as an inducible “suicide gene.” Giving the mice a certain drug (a low dose of tamoxifen) activated the suicide gene in all p21high cells, causing them to die. Administering this treatment once a month, from age 20 months (older age) until the end of life, significantly extended the rodents’ lifespan, reduced inflammation, and decreased gene activity linked to aging.
Treated mice lived, on average, for 33 months — 3 months longer than the untreated mice. The oldest treated mouse lived to 43 months — roughly 130 in human years.
But the treated mice didn’t just live longer; they were also healthier. In humans, walking speed and grip strength can be clues of overall health and vitality. The old, treated mice were able to walk faster and grip objects with greater strength than untreated mice of the same age.
Dr. Xu’s lab is now testing drugs that target p21high cells in hopes of finding one that would work in humans. Leveraging immunotherapy technology to target these cells could be another option, Dr. Xu said.
The team also plans to test whether eliminating p21high cells could prevent or alleviate diabetes or Alzheimer’s disease.
Challenges and Criticisms
The research provides “important evidence that targeting senescence and the molecular components of that pathway might provide some benefit in the long term,” Dr. Vaughan said.
But killing senescent cells could come with downsides.
“Senescence protects us from hyperproliferative responses,” potentially blocking cells from becoming malignant, Dr. Vaughan said. “There’s this effect on aging that is desirable, but at the same time, you may enhance your risk of cancer or malignancy or excessive proliferation in some cells.”
And of course, we don’t necessarily need drugs to prolong healthy life, Dr. Vaughan pointed out.
For many people, a long healthy life is already within reach. Humans live longer on average than they used to, and simple lifestyle choices — nourishing your body well, staying active, and maintaining a healthy weight — can increase one’s chances of good health.
The most consistently demonstrated intervention for extending lifespan “in almost every animal species is caloric restriction,” Dr. Vaughan said. (Dr. Xu’s team is also investigating whether fasting and exercise can lead to a decrease in p21high cells.)
As for brain health, Dr. Vaughan and colleagues at Northwestern are studying “super agers,” people who are cognitively intact into their 90s.
“The one single thing that they found that contributes to that process, and contributes to that success, is really a social network and human bonds and interaction,” Dr. Vaughan said.
A version of this article appeared on Medscape.com.
What if a drug could help you live a longer, healthier life?
Scientists at the University of Connecticut are working on it. In a new study in Cell Metabolism, researchers described how to target specific cells to extend the lifespan and improve the health of mice late in life.
The study builds on a growing body of research, mostly in animals, testing interventions to slow aging and prolong health span, the length of time that one is not just alive but also healthy.
“Aging is the most important risk factor for every disease that we deal with in adult human beings,” said cardiologist Douglas Vaughan, MD, director of the Potocsnak Longevity Institute at Northwestern University’s Feinberg School of Medicine, Chicago. (Dr. Vaughan was not involved in the new study.) “So the big hypothesis is: If we could slow down aging just a little bit, we can push back the onset of disease.”
Senescent cells — or “zombie cells” — secrete harmful substances that disrupt tissue functioning. They’ve been linked to chronic inflammation, tissue damage, and the development of age-related diseases.
Senescence can be characterized by the accumulation of cells with high levels of specific markers like p21, or p21high cells. Almost any cell can become a p21high cell, and they accumulate with age, said Ming Xu, PhD, a professor at the UConn Center on Aging, UConn Health, Farmington, Connecticut, who led the study.
By targeting and eliminating p21high senescent cells, Dr. Xu hopes to develop novel therapies that might help people live longer and enjoy more years in good health.
Such a treatment could be ready for human trials in 2-5 years, Dr. Xu said.
What the Researchers Did
Xu and colleagues used genetic engineering to eliminate p21high cells in mice, introducing into their genome something they describe as an inducible “suicide gene.” Giving the mice a certain drug (a low dose of tamoxifen) activated the suicide gene in all p21high cells, causing them to die. Administering this treatment once a month, from age 20 months (older age) until the end of life, significantly extended the rodents’ lifespan, reduced inflammation, and decreased gene activity linked to aging.
Treated mice lived, on average, for 33 months — 3 months longer than the untreated mice. The oldest treated mouse lived to 43 months — roughly 130 in human years.
But the treated mice didn’t just live longer; they were also healthier. In humans, walking speed and grip strength can be clues of overall health and vitality. The old, treated mice were able to walk faster and grip objects with greater strength than untreated mice of the same age.
Dr. Xu’s lab is now testing drugs that target p21high cells in hopes of finding one that would work in humans. Leveraging immunotherapy technology to target these cells could be another option, Dr. Xu said.
The team also plans to test whether eliminating p21high cells could prevent or alleviate diabetes or Alzheimer’s disease.
Challenges and Criticisms
The research provides “important evidence that targeting senescence and the molecular components of that pathway might provide some benefit in the long term,” Dr. Vaughan said.
But killing senescent cells could come with downsides.
“Senescence protects us from hyperproliferative responses,” potentially blocking cells from becoming malignant, Dr. Vaughan said. “There’s this effect on aging that is desirable, but at the same time, you may enhance your risk of cancer or malignancy or excessive proliferation in some cells.”
And of course, we don’t necessarily need drugs to prolong healthy life, Dr. Vaughan pointed out.
For many people, a long healthy life is already within reach. Humans live longer on average than they used to, and simple lifestyle choices — nourishing your body well, staying active, and maintaining a healthy weight — can increase one’s chances of good health.
The most consistently demonstrated intervention for extending lifespan “in almost every animal species is caloric restriction,” Dr. Vaughan said. (Dr. Xu’s team is also investigating whether fasting and exercise can lead to a decrease in p21high cells.)
As for brain health, Dr. Vaughan and colleagues at Northwestern are studying “super agers,” people who are cognitively intact into their 90s.
“The one single thing that they found that contributes to that process, and contributes to that success, is really a social network and human bonds and interaction,” Dr. Vaughan said.
A version of this article appeared on Medscape.com.
What You Need to Know About Oropouche Virus Disease
The European Centre for Disease Prevention and Control (ECDC) has issued a warning to travelers in areas in South and Central America and the Caribbean affected by a current outbreak of Oropouche virus (OROV) disease. The ECDC said that there had been more than 8000 cases reported in these areas since January, with 19 imported cases reported in Europe for the first time in June and July. Of these, 12 were in Spain, five were in Italy, and two were in Germany.
The ECDC’s Threat Assessment Brief of Aug. 9 said that one of those affected had traveled to Brazil and the other 18 to Cuba; however, outbreaks have also been reported this year in Bolivia, Colombia, and Peru. Though the overall risk for infection to European travelers to OROV-epidemic countries was assessed as moderate, it was higher in the more affected municipalities of the northern states of Brazil and/or the Amazon region, and/or if personal protection measures are not taken.
An editorial published Aug. 8 in The Lancet Infectious Diseases described OROV as a “mysterious threat,” which there is limited knowledge about despite some half a million cases recorded since it was first detected in Trinidad and Tobago in 1955.
OROV is transmitted primarily through bites from infected midges (Culicoides paraensis). However, some mosquitoes species can also spread the virus, which causes symptoms very similar to other arbovirus diseases from the same regions, such as dengue, chikungunya, and Zika virus infection.
Most cases are mild, but meningitis and encephalitis can occur as well as possible fetal death and deformities after infection in pregnancy. Last month, the first fatal cases were reported in two young Brazilian women who, concerningly, had no comorbidities.
This news organization asked Jan Felix Drexler, MD, of the Institute of Virology at Charité – Universitätsmedizin in Berlin, Germany, who has studied the emergence of Oropouche fever in Latin America, what clinicians should know about OROV disease.
What are the main symptoms of OROV disease for which clinicians should be alert?
The main symptoms are not different from other arboviral infections, ie, fever, maybe joint and muscle pain, maybe rash. The problem is that we do not know how often severe disease may occur because we do not know whether the severe cases that have been postulated, including death in apparently healthy people and congenital infection, are due to increased testing; an altered virus; or an altered, more intense circulation (so that many more infections simply lead to rare severe cases appearing). Be alert and ask for testing in your patients.
What is the differential diagnosis if a recent traveler to affected regions presents with symptoms? Are there any clues to suggest whether the disease is Oropouche as opposed to Zika, etc.?
The main message is: Do not assume a particular infection based on clinical symptoms. If your patient is returning from or living in an endemic area, consider OROV disease in the differential diagnosis.
What personal protective measures should clinicians advise travelers in affected areas to take? Do these differ from normal mosquito precautions?
Repellents are extremely important as usual. However, there are differences. Mosquito nets’ hole sizes need to be smaller than those used against the vectors of malaria or dengue; in other words, they need to have a higher mesh. The problem is that nets with high mesh are complicated in very hot and humid conditions because they also limit ventilation. Travelers should discuss with local suppliers about the best trade-off.
The risk for midge bites is likely highest at dawn and dusk in still and humid conditions. So on the one hand, one could recommend avoiding those areas and being outside during those times of the day. On the other hand, specific recommendations cannot be made robustly because we cannot exclude other invertebrate vectors at current knowledge. Some studies have implicated that mosquitoes may also transmit the virus. If that holds true, then we are back to reducing any bite.
Should pregnant women be advised to avoid travel to affected regions?
Not immediately, but caution must be taken. We simply do not have sufficient data to gauge the risk for potential congenital infection. Much more epidemiologic data and controlled infection experiments will be required to make evidence-based recommendations.
All the cases reported in Europe so far were imported from Cuba and Brazil. Is there any risk for local transmission, eg, via midges/mosquitoes that might hitch a ride on an aircraft, as in cases of airport malaria?
Not immediately, but it cannot be excluded. We know very little about the infection intensity in the vectors. Controlled infection experiments, including robustness of vectors against commonly used insecticides in airplanes, need to be done.
What is the risk for an animal reservoir emerging in Europe?
We do not know, but there is also no reason for ringing the alarm bells. Controlled infection experiments and surveillance will be required.
Is treatment purely supportive or are there any specific agents worth trying in case of severe symptoms/neurologic involvement?
No specific treatment can be recommended as is. However, severe dengue illustrates the relevance of supportive treatment, which is hugely effective in reducing mortality.
The Lancet paper states: “Several laboratory tests have been developed but robust commercial tests are hardly available.” How likely is it that laboratories in Europe will have the capability to test for the Oropouche organism?
European laboratory networks have already taken action, and testing is now available at least in the major and reference laboratories. If a clinician asks for OROV testing, they will probably get a robust answer in a reasonable timespan. Of course, that can be improved once we have more cases and more laboratories will be equipped for testing.
Is there anything else you think clinicians should be aware of?
The most important is to think beyond the textbooks we know from medical school. Things change rapidly in a connected world under altered climate conditions.
Dr. Drexler has no conflicts of interest to declare.
A version of this article first appeared on Medscape.com.
The European Centre for Disease Prevention and Control (ECDC) has issued a warning to travelers in areas in South and Central America and the Caribbean affected by a current outbreak of Oropouche virus (OROV) disease. The ECDC said that there had been more than 8000 cases reported in these areas since January, with 19 imported cases reported in Europe for the first time in June and July. Of these, 12 were in Spain, five were in Italy, and two were in Germany.
The ECDC’s Threat Assessment Brief of Aug. 9 said that one of those affected had traveled to Brazil and the other 18 to Cuba; however, outbreaks have also been reported this year in Bolivia, Colombia, and Peru. Though the overall risk for infection to European travelers to OROV-epidemic countries was assessed as moderate, it was higher in the more affected municipalities of the northern states of Brazil and/or the Amazon region, and/or if personal protection measures are not taken.
An editorial published Aug. 8 in The Lancet Infectious Diseases described OROV as a “mysterious threat,” which there is limited knowledge about despite some half a million cases recorded since it was first detected in Trinidad and Tobago in 1955.
OROV is transmitted primarily through bites from infected midges (Culicoides paraensis). However, some mosquitoes species can also spread the virus, which causes symptoms very similar to other arbovirus diseases from the same regions, such as dengue, chikungunya, and Zika virus infection.
Most cases are mild, but meningitis and encephalitis can occur as well as possible fetal death and deformities after infection in pregnancy. Last month, the first fatal cases were reported in two young Brazilian women who, concerningly, had no comorbidities.
This news organization asked Jan Felix Drexler, MD, of the Institute of Virology at Charité – Universitätsmedizin in Berlin, Germany, who has studied the emergence of Oropouche fever in Latin America, what clinicians should know about OROV disease.
What are the main symptoms of OROV disease for which clinicians should be alert?
The main symptoms are not different from other arboviral infections, ie, fever, maybe joint and muscle pain, maybe rash. The problem is that we do not know how often severe disease may occur because we do not know whether the severe cases that have been postulated, including death in apparently healthy people and congenital infection, are due to increased testing; an altered virus; or an altered, more intense circulation (so that many more infections simply lead to rare severe cases appearing). Be alert and ask for testing in your patients.
What is the differential diagnosis if a recent traveler to affected regions presents with symptoms? Are there any clues to suggest whether the disease is Oropouche as opposed to Zika, etc.?
The main message is: Do not assume a particular infection based on clinical symptoms. If your patient is returning from or living in an endemic area, consider OROV disease in the differential diagnosis.
What personal protective measures should clinicians advise travelers in affected areas to take? Do these differ from normal mosquito precautions?
Repellents are extremely important as usual. However, there are differences. Mosquito nets’ hole sizes need to be smaller than those used against the vectors of malaria or dengue; in other words, they need to have a higher mesh. The problem is that nets with high mesh are complicated in very hot and humid conditions because they also limit ventilation. Travelers should discuss with local suppliers about the best trade-off.
The risk for midge bites is likely highest at dawn and dusk in still and humid conditions. So on the one hand, one could recommend avoiding those areas and being outside during those times of the day. On the other hand, specific recommendations cannot be made robustly because we cannot exclude other invertebrate vectors at current knowledge. Some studies have implicated that mosquitoes may also transmit the virus. If that holds true, then we are back to reducing any bite.
Should pregnant women be advised to avoid travel to affected regions?
Not immediately, but caution must be taken. We simply do not have sufficient data to gauge the risk for potential congenital infection. Much more epidemiologic data and controlled infection experiments will be required to make evidence-based recommendations.
All the cases reported in Europe so far were imported from Cuba and Brazil. Is there any risk for local transmission, eg, via midges/mosquitoes that might hitch a ride on an aircraft, as in cases of airport malaria?
Not immediately, but it cannot be excluded. We know very little about the infection intensity in the vectors. Controlled infection experiments, including robustness of vectors against commonly used insecticides in airplanes, need to be done.
What is the risk for an animal reservoir emerging in Europe?
We do not know, but there is also no reason for ringing the alarm bells. Controlled infection experiments and surveillance will be required.
Is treatment purely supportive or are there any specific agents worth trying in case of severe symptoms/neurologic involvement?
No specific treatment can be recommended as is. However, severe dengue illustrates the relevance of supportive treatment, which is hugely effective in reducing mortality.
The Lancet paper states: “Several laboratory tests have been developed but robust commercial tests are hardly available.” How likely is it that laboratories in Europe will have the capability to test for the Oropouche organism?
European laboratory networks have already taken action, and testing is now available at least in the major and reference laboratories. If a clinician asks for OROV testing, they will probably get a robust answer in a reasonable timespan. Of course, that can be improved once we have more cases and more laboratories will be equipped for testing.
Is there anything else you think clinicians should be aware of?
The most important is to think beyond the textbooks we know from medical school. Things change rapidly in a connected world under altered climate conditions.
Dr. Drexler has no conflicts of interest to declare.
A version of this article first appeared on Medscape.com.
The European Centre for Disease Prevention and Control (ECDC) has issued a warning to travelers in areas in South and Central America and the Caribbean affected by a current outbreak of Oropouche virus (OROV) disease. The ECDC said that there had been more than 8000 cases reported in these areas since January, with 19 imported cases reported in Europe for the first time in June and July. Of these, 12 were in Spain, five were in Italy, and two were in Germany.
The ECDC’s Threat Assessment Brief of Aug. 9 said that one of those affected had traveled to Brazil and the other 18 to Cuba; however, outbreaks have also been reported this year in Bolivia, Colombia, and Peru. Though the overall risk for infection to European travelers to OROV-epidemic countries was assessed as moderate, it was higher in the more affected municipalities of the northern states of Brazil and/or the Amazon region, and/or if personal protection measures are not taken.
An editorial published Aug. 8 in The Lancet Infectious Diseases described OROV as a “mysterious threat,” which there is limited knowledge about despite some half a million cases recorded since it was first detected in Trinidad and Tobago in 1955.
OROV is transmitted primarily through bites from infected midges (Culicoides paraensis). However, some mosquitoes species can also spread the virus, which causes symptoms very similar to other arbovirus diseases from the same regions, such as dengue, chikungunya, and Zika virus infection.
Most cases are mild, but meningitis and encephalitis can occur as well as possible fetal death and deformities after infection in pregnancy. Last month, the first fatal cases were reported in two young Brazilian women who, concerningly, had no comorbidities.
This news organization asked Jan Felix Drexler, MD, of the Institute of Virology at Charité – Universitätsmedizin in Berlin, Germany, who has studied the emergence of Oropouche fever in Latin America, what clinicians should know about OROV disease.
What are the main symptoms of OROV disease for which clinicians should be alert?
The main symptoms are not different from other arboviral infections, ie, fever, maybe joint and muscle pain, maybe rash. The problem is that we do not know how often severe disease may occur because we do not know whether the severe cases that have been postulated, including death in apparently healthy people and congenital infection, are due to increased testing; an altered virus; or an altered, more intense circulation (so that many more infections simply lead to rare severe cases appearing). Be alert and ask for testing in your patients.
What is the differential diagnosis if a recent traveler to affected regions presents with symptoms? Are there any clues to suggest whether the disease is Oropouche as opposed to Zika, etc.?
The main message is: Do not assume a particular infection based on clinical symptoms. If your patient is returning from or living in an endemic area, consider OROV disease in the differential diagnosis.
What personal protective measures should clinicians advise travelers in affected areas to take? Do these differ from normal mosquito precautions?
Repellents are extremely important as usual. However, there are differences. Mosquito nets’ hole sizes need to be smaller than those used against the vectors of malaria or dengue; in other words, they need to have a higher mesh. The problem is that nets with high mesh are complicated in very hot and humid conditions because they also limit ventilation. Travelers should discuss with local suppliers about the best trade-off.
The risk for midge bites is likely highest at dawn and dusk in still and humid conditions. So on the one hand, one could recommend avoiding those areas and being outside during those times of the day. On the other hand, specific recommendations cannot be made robustly because we cannot exclude other invertebrate vectors at current knowledge. Some studies have implicated that mosquitoes may also transmit the virus. If that holds true, then we are back to reducing any bite.
Should pregnant women be advised to avoid travel to affected regions?
Not immediately, but caution must be taken. We simply do not have sufficient data to gauge the risk for potential congenital infection. Much more epidemiologic data and controlled infection experiments will be required to make evidence-based recommendations.
All the cases reported in Europe so far were imported from Cuba and Brazil. Is there any risk for local transmission, eg, via midges/mosquitoes that might hitch a ride on an aircraft, as in cases of airport malaria?
Not immediately, but it cannot be excluded. We know very little about the infection intensity in the vectors. Controlled infection experiments, including robustness of vectors against commonly used insecticides in airplanes, need to be done.
What is the risk for an animal reservoir emerging in Europe?
We do not know, but there is also no reason for ringing the alarm bells. Controlled infection experiments and surveillance will be required.
Is treatment purely supportive or are there any specific agents worth trying in case of severe symptoms/neurologic involvement?
No specific treatment can be recommended as is. However, severe dengue illustrates the relevance of supportive treatment, which is hugely effective in reducing mortality.
The Lancet paper states: “Several laboratory tests have been developed but robust commercial tests are hardly available.” How likely is it that laboratories in Europe will have the capability to test for the Oropouche organism?
European laboratory networks have already taken action, and testing is now available at least in the major and reference laboratories. If a clinician asks for OROV testing, they will probably get a robust answer in a reasonable timespan. Of course, that can be improved once we have more cases and more laboratories will be equipped for testing.
Is there anything else you think clinicians should be aware of?
The most important is to think beyond the textbooks we know from medical school. Things change rapidly in a connected world under altered climate conditions.
Dr. Drexler has no conflicts of interest to declare.
A version of this article first appeared on Medscape.com.
Top-Down Treatment Appears Better for Patients With Crohn’s Disease
according to a recent study.
Top-down treatment achieved substantially better outcomes at one year after diagnosis than step-up treatment, with nearly 80% of those receiving top-down therapy having both symptoms and inflammatory markers controlled, as compared with only 15% of those receiving accelerated step-up therapy.
“Up until now, the view has been: ‘Why would you use a more expensive treatment strategy and potentially overtreat people if there’s a chance they might do fine anyway?’ ” asked senior author Miles Parkes, MBBS, professor of translational gastroenterology at the University of Cambridge in England and director of the National Institute for Health and Care Research (NIHR) Cambridge Biomedical Research Centre.
“As we’ve shown, and as previous studies have demonstrated, there’s actually a pretty high risk that an individual with Crohn’s disease will experience disease flares and complications even in the first year after diagnosis,” he said. “We now know we can prevent the majority of adverse outcomes, including need for urgent surgery, by providing a treatment strategy that is safe and becoming increasingly affordable.”
The study was published in The Lancet Gastroenterology & Hepatology.
Comparing Treatments
Dr. Parkes and colleagues conducted a multicenter, open-label, biomarker-stratified randomized controlled trial among adults with newly diagnosed active Crohn’s disease. Participants were tested for a prognostic biomarker derived from T-cell transcriptional signatures and randomly assigned to a top-down or accelerated step-up treatment based on biomarker subgroup, endoscopic inflammation (mild, moderate, or severe), and extent (colonic or other).
The primary endpoint was sustained steroid-free and surgery-free remission after completing a steroid induction (maximum 8-week course) to week 48. Remission was defined by a composite of symptoms and inflammatory markers at all visits, with a Harvey-Bradshaw Index (HBI) score of less than 5 or resolved inflammatory markers or both, while a flare was defined as active symptoms (HBI ≥ 5) and raised inflammatory markers.
Across 40 UK hospitals, 386 patients (mean age, 33.6 years; 54% male) were randomized, with 193 receiving a top-down therapy of combination intravenous infliximab plus immunomodulator (azathioprine, low-dose mercaptopurine with allopurinol, or methotrexate) and 193 receiving an accelerated step-up therapy of an immunomodulator and then infliximab if further flares occurred after the steroid course. In the step-up group, 85% required escalation to an immunomodulator, and 41% required infliximab by week 48.
Overall, sustained steroid-free and surgery-free remission was significantly more frequent in the top-down group than in the accelerated step-up group (among 149 of 189 patients vs 29 of 190 patients), at 79% vs 15%, marking an absolute difference of 64 percentage points.
Top-down treatment also showed greater efficacy in achieving endoscopic remission (67% vs 44%), improved quality of life, lower need for steroids, and reduced number of flares requiring treatment escalation.
In addition, there were fewer adverse events (168 vs 315) and fewer serious adverse events (15 vs 42) in the top-down group than in the step-up group. There were also fewer complications that required urgent abdominal surgery, with one in the top-down group for gallstone ileus and nine in the step-up group requiring intestinal resection for structuring or fistulating complications.
However, the biomarker showed no clinical utility, and none of the baseline measurements predicted which patients were at risk of adverse outcomes with the step-up approach, Dr. Parkes said.
“The key message is that Crohn’s is unpredictable, hence you are better off treating everyone who has significant disease at diagnosis with combo therapy (anti-TNF [tumor necrosis factor] plus immunomodulator) rather than ‘wait and see,’ as bad things happen to people with uncontrolled inflammation during that ‘wait and see’ stage,” he said.
Additional Considerations
In the PROFILE trial, the need for a prognostic biomarker was based on the lack of an effective, safe, and affordable treatment strategy for newly diagnosed patients, the study authors wrote, but effective top-down management could reduce the need for a biomarker.
“In one sense, this is a negative study as the blood-based CD8+ T-cell transcriptomic biomarker that was being studied was not predictive of outcomes at all. But PROFILE makes it very clear that early effective therapy leads to better outcomes than accelerated step-up therapy,” said Neeraj Narula, MD, associate professor of medicine at McMaster University, Hamilton, Ontario, Canada, and staff gastroenterologist focused on inflammatory bowel disease at Hamilton Health Sciences.
Dr. Narula, who wasn’t involved with this study, has researched the comparative effectiveness of biologics for endoscopic healing of the ileum and colon in Crohn’s disease. He and colleagues found that anti-TNF biologics were effective in achieving 1-year endoscopic healing in moderate to severe Crohn’s disease.
“These findings likely aren’t specific to infliximab/azathioprine, and I suspect similar outcomes would be shown for other advanced therapies used early in the course of disease,” he said. “There does remain a concern that using this strategy for all patients may lead to overtreatment of some, but perhaps any harm done by overtreatment of a minority may be offset by the harm resulting from undertreatment of the majority. It’s hard to say for sure, but it certainly gives us some food for thought.”
The study was funded by Wellcome and PredictImmune and jointly sponsored by the University of Cambridge and Cambridge University Hospitals NHS Foundation Trust. Dr. Parkes and several authors declared fees and grants from numerous companies outside of this study. Dr. Narula reported no relevant disclosures.
according to a recent study.
Top-down treatment achieved substantially better outcomes at one year after diagnosis than step-up treatment, with nearly 80% of those receiving top-down therapy having both symptoms and inflammatory markers controlled, as compared with only 15% of those receiving accelerated step-up therapy.
“Up until now, the view has been: ‘Why would you use a more expensive treatment strategy and potentially overtreat people if there’s a chance they might do fine anyway?’ ” asked senior author Miles Parkes, MBBS, professor of translational gastroenterology at the University of Cambridge in England and director of the National Institute for Health and Care Research (NIHR) Cambridge Biomedical Research Centre.
“As we’ve shown, and as previous studies have demonstrated, there’s actually a pretty high risk that an individual with Crohn’s disease will experience disease flares and complications even in the first year after diagnosis,” he said. “We now know we can prevent the majority of adverse outcomes, including need for urgent surgery, by providing a treatment strategy that is safe and becoming increasingly affordable.”
The study was published in The Lancet Gastroenterology & Hepatology.
Comparing Treatments
Dr. Parkes and colleagues conducted a multicenter, open-label, biomarker-stratified randomized controlled trial among adults with newly diagnosed active Crohn’s disease. Participants were tested for a prognostic biomarker derived from T-cell transcriptional signatures and randomly assigned to a top-down or accelerated step-up treatment based on biomarker subgroup, endoscopic inflammation (mild, moderate, or severe), and extent (colonic or other).
The primary endpoint was sustained steroid-free and surgery-free remission after completing a steroid induction (maximum 8-week course) to week 48. Remission was defined by a composite of symptoms and inflammatory markers at all visits, with a Harvey-Bradshaw Index (HBI) score of less than 5 or resolved inflammatory markers or both, while a flare was defined as active symptoms (HBI ≥ 5) and raised inflammatory markers.
Across 40 UK hospitals, 386 patients (mean age, 33.6 years; 54% male) were randomized, with 193 receiving a top-down therapy of combination intravenous infliximab plus immunomodulator (azathioprine, low-dose mercaptopurine with allopurinol, or methotrexate) and 193 receiving an accelerated step-up therapy of an immunomodulator and then infliximab if further flares occurred after the steroid course. In the step-up group, 85% required escalation to an immunomodulator, and 41% required infliximab by week 48.
Overall, sustained steroid-free and surgery-free remission was significantly more frequent in the top-down group than in the accelerated step-up group (among 149 of 189 patients vs 29 of 190 patients), at 79% vs 15%, marking an absolute difference of 64 percentage points.
Top-down treatment also showed greater efficacy in achieving endoscopic remission (67% vs 44%), improved quality of life, lower need for steroids, and reduced number of flares requiring treatment escalation.
In addition, there were fewer adverse events (168 vs 315) and fewer serious adverse events (15 vs 42) in the top-down group than in the step-up group. There were also fewer complications that required urgent abdominal surgery, with one in the top-down group for gallstone ileus and nine in the step-up group requiring intestinal resection for structuring or fistulating complications.
However, the biomarker showed no clinical utility, and none of the baseline measurements predicted which patients were at risk of adverse outcomes with the step-up approach, Dr. Parkes said.
“The key message is that Crohn’s is unpredictable, hence you are better off treating everyone who has significant disease at diagnosis with combo therapy (anti-TNF [tumor necrosis factor] plus immunomodulator) rather than ‘wait and see,’ as bad things happen to people with uncontrolled inflammation during that ‘wait and see’ stage,” he said.
Additional Considerations
In the PROFILE trial, the need for a prognostic biomarker was based on the lack of an effective, safe, and affordable treatment strategy for newly diagnosed patients, the study authors wrote, but effective top-down management could reduce the need for a biomarker.
“In one sense, this is a negative study as the blood-based CD8+ T-cell transcriptomic biomarker that was being studied was not predictive of outcomes at all. But PROFILE makes it very clear that early effective therapy leads to better outcomes than accelerated step-up therapy,” said Neeraj Narula, MD, associate professor of medicine at McMaster University, Hamilton, Ontario, Canada, and staff gastroenterologist focused on inflammatory bowel disease at Hamilton Health Sciences.
Dr. Narula, who wasn’t involved with this study, has researched the comparative effectiveness of biologics for endoscopic healing of the ileum and colon in Crohn’s disease. He and colleagues found that anti-TNF biologics were effective in achieving 1-year endoscopic healing in moderate to severe Crohn’s disease.
“These findings likely aren’t specific to infliximab/azathioprine, and I suspect similar outcomes would be shown for other advanced therapies used early in the course of disease,” he said. “There does remain a concern that using this strategy for all patients may lead to overtreatment of some, but perhaps any harm done by overtreatment of a minority may be offset by the harm resulting from undertreatment of the majority. It’s hard to say for sure, but it certainly gives us some food for thought.”
The study was funded by Wellcome and PredictImmune and jointly sponsored by the University of Cambridge and Cambridge University Hospitals NHS Foundation Trust. Dr. Parkes and several authors declared fees and grants from numerous companies outside of this study. Dr. Narula reported no relevant disclosures.
according to a recent study.
Top-down treatment achieved substantially better outcomes at one year after diagnosis than step-up treatment, with nearly 80% of those receiving top-down therapy having both symptoms and inflammatory markers controlled, as compared with only 15% of those receiving accelerated step-up therapy.
“Up until now, the view has been: ‘Why would you use a more expensive treatment strategy and potentially overtreat people if there’s a chance they might do fine anyway?’ ” asked senior author Miles Parkes, MBBS, professor of translational gastroenterology at the University of Cambridge in England and director of the National Institute for Health and Care Research (NIHR) Cambridge Biomedical Research Centre.
“As we’ve shown, and as previous studies have demonstrated, there’s actually a pretty high risk that an individual with Crohn’s disease will experience disease flares and complications even in the first year after diagnosis,” he said. “We now know we can prevent the majority of adverse outcomes, including need for urgent surgery, by providing a treatment strategy that is safe and becoming increasingly affordable.”
The study was published in The Lancet Gastroenterology & Hepatology.
Comparing Treatments
Dr. Parkes and colleagues conducted a multicenter, open-label, biomarker-stratified randomized controlled trial among adults with newly diagnosed active Crohn’s disease. Participants were tested for a prognostic biomarker derived from T-cell transcriptional signatures and randomly assigned to a top-down or accelerated step-up treatment based on biomarker subgroup, endoscopic inflammation (mild, moderate, or severe), and extent (colonic or other).
The primary endpoint was sustained steroid-free and surgery-free remission after completing a steroid induction (maximum 8-week course) to week 48. Remission was defined by a composite of symptoms and inflammatory markers at all visits, with a Harvey-Bradshaw Index (HBI) score of less than 5 or resolved inflammatory markers or both, while a flare was defined as active symptoms (HBI ≥ 5) and raised inflammatory markers.
Across 40 UK hospitals, 386 patients (mean age, 33.6 years; 54% male) were randomized, with 193 receiving a top-down therapy of combination intravenous infliximab plus immunomodulator (azathioprine, low-dose mercaptopurine with allopurinol, or methotrexate) and 193 receiving an accelerated step-up therapy of an immunomodulator and then infliximab if further flares occurred after the steroid course. In the step-up group, 85% required escalation to an immunomodulator, and 41% required infliximab by week 48.
Overall, sustained steroid-free and surgery-free remission was significantly more frequent in the top-down group than in the accelerated step-up group (among 149 of 189 patients vs 29 of 190 patients), at 79% vs 15%, marking an absolute difference of 64 percentage points.
Top-down treatment also showed greater efficacy in achieving endoscopic remission (67% vs 44%), improved quality of life, lower need for steroids, and reduced number of flares requiring treatment escalation.
In addition, there were fewer adverse events (168 vs 315) and fewer serious adverse events (15 vs 42) in the top-down group than in the step-up group. There were also fewer complications that required urgent abdominal surgery, with one in the top-down group for gallstone ileus and nine in the step-up group requiring intestinal resection for structuring or fistulating complications.
However, the biomarker showed no clinical utility, and none of the baseline measurements predicted which patients were at risk of adverse outcomes with the step-up approach, Dr. Parkes said.
“The key message is that Crohn’s is unpredictable, hence you are better off treating everyone who has significant disease at diagnosis with combo therapy (anti-TNF [tumor necrosis factor] plus immunomodulator) rather than ‘wait and see,’ as bad things happen to people with uncontrolled inflammation during that ‘wait and see’ stage,” he said.
Additional Considerations
In the PROFILE trial, the need for a prognostic biomarker was based on the lack of an effective, safe, and affordable treatment strategy for newly diagnosed patients, the study authors wrote, but effective top-down management could reduce the need for a biomarker.
“In one sense, this is a negative study as the blood-based CD8+ T-cell transcriptomic biomarker that was being studied was not predictive of outcomes at all. But PROFILE makes it very clear that early effective therapy leads to better outcomes than accelerated step-up therapy,” said Neeraj Narula, MD, associate professor of medicine at McMaster University, Hamilton, Ontario, Canada, and staff gastroenterologist focused on inflammatory bowel disease at Hamilton Health Sciences.
Dr. Narula, who wasn’t involved with this study, has researched the comparative effectiveness of biologics for endoscopic healing of the ileum and colon in Crohn’s disease. He and colleagues found that anti-TNF biologics were effective in achieving 1-year endoscopic healing in moderate to severe Crohn’s disease.
“These findings likely aren’t specific to infliximab/azathioprine, and I suspect similar outcomes would be shown for other advanced therapies used early in the course of disease,” he said. “There does remain a concern that using this strategy for all patients may lead to overtreatment of some, but perhaps any harm done by overtreatment of a minority may be offset by the harm resulting from undertreatment of the majority. It’s hard to say for sure, but it certainly gives us some food for thought.”
The study was funded by Wellcome and PredictImmune and jointly sponsored by the University of Cambridge and Cambridge University Hospitals NHS Foundation Trust. Dr. Parkes and several authors declared fees and grants from numerous companies outside of this study. Dr. Narula reported no relevant disclosures.
FROM THE LANCET GASTROENTEROLOGY & HEPATOLOGY
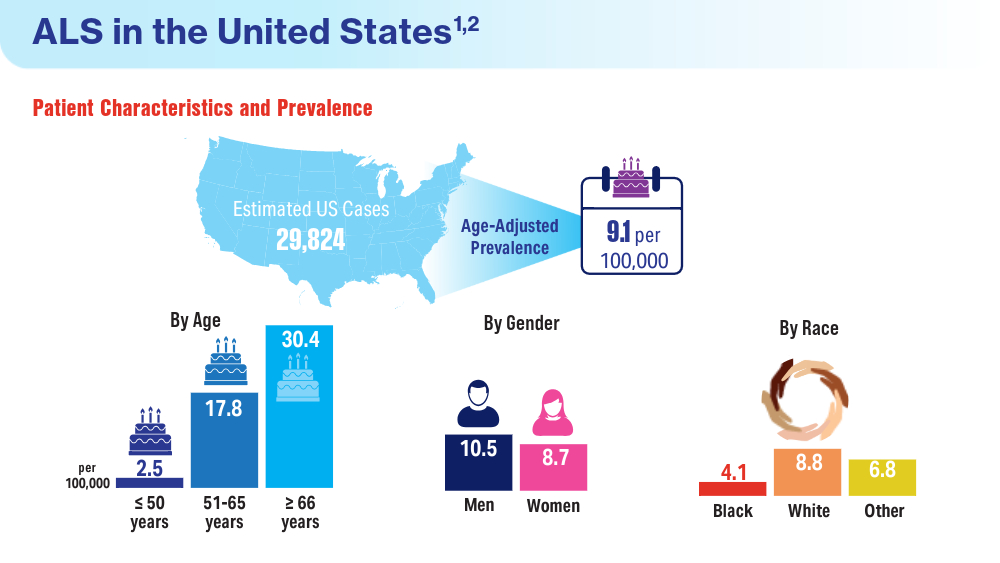
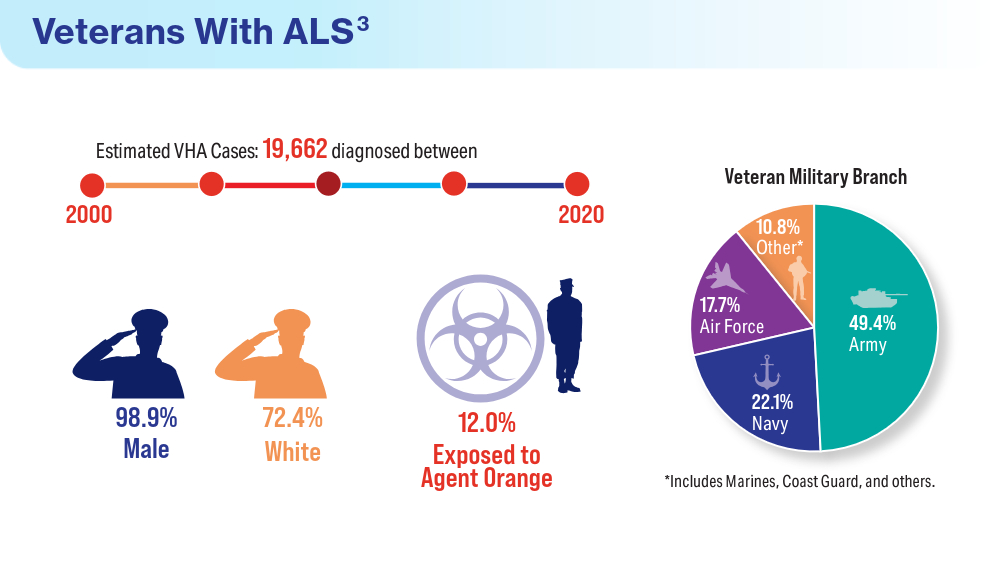
Federal Health Care Data Trends 2024
Federal Health Care Data Trends is a special supplement to Federal Practitioner, showcasing the latest research in health care for veterans and active-duty military members via compelling infographics. Click below to view highlights from the issue:
Federal Health Care Data Trends is a special supplement to Federal Practitioner, showcasing the latest research in health care for veterans and active-duty military members via compelling infographics. Click below to view highlights from the issue:
Federal Health Care Data Trends is a special supplement to Federal Practitioner, showcasing the latest research in health care for veterans and active-duty military members via compelling infographics. Click below to view highlights from the issue:
Could Mobile Tech Help to Minimize COPD Exacerbations?
Could mobile technology help patients with chronic obstructive pulmonary disease (COPD) who may not seek care until they experience an exacerbation?
Self-management interventions for COPD can potentially improve quality of life and reduce hospitalizations, wrote Robert Wu, MD, associate professor in the Department of Medicine at the University of Toronto, and colleagues. However, data on the use of devices and apps to manage COPD by providing reminders for self-care, predicting early exacerbations, and facilitating communication with healthcare providers are limited, they said.
In a study published in COPD: Journal of Chronic Obstructive Pulmonary Disease, the researchers reported details from interviews with 26 adult patients with COPD who used a wearable device and app for 6 months to help manage their condition. The interviews were part of a larger cohort study.
“The motivation for this study was to understand the patient perspective on using wearables to help support their chronic lung condition,” Dr. Wu said in an interview. “People with COPD can be at high risk of being admitted to hospital, so it is important to see if innovative technology like wearables or remote monitoring can help them,” he said.
Individuals with COPD tend to be older and less technologically adept, and they may be less willing to adopt new technology, he added. “We wanted to understand what would make people use a self-management app,” he said.
On enrollment in the study, patients received a smartwatch and a smartphone with a preinstalled app for COPD management. The app included daily reminders to take medication, perform guided breathing sessions, check blood oxygen on the smartwatch or an oximeter, and complete a symptom questionnaire. The app also allowed participants to record when they exercised and provided feedback on heart rate and daily activity, including passive step counts. Participants earned stars for meeting daily exercise goals of active minutes and total steps.
Participants received training in the use of the app from members of the research team and completed semi-structured interviews after using the items for 6 months.
The researchers divided their findings into four main themes: information, support and reassurance; barriers to adoption; impact on communication with healthcare providers; and opportunities for improvement.
Overall, most patients reported that the feedback they received through the app was useful. In particular, participants reported that the app and smartwatch provided reassurance and feedback about stable vitals during exercise, which encouraged some to adhere to regular exercise routines. Approximately two thirds (65%) said that the daily exercise reminders were motivational. In addition, 20% reported that they interpreted vital data, including heart rate, as a signal to slow down.
Participants rated medication reminders and the option to create an action plan for COPD management as the least useful features; 69% said that they already had medication reminders in place.
A total of four patients experienced technical difficulties with the app that kept it from impacting their disease management. Some of the suggestions from participants for improvement included adding information about food intake, weight, blood pressure, and temperature to the health information being tracked, as well as restoring the oxygen saturation measure, which had been disabled because of accuracy concerns. Barriers to use of the device and app included the bulkiness of the device as well as the reported technical malfunctions.
The findings were limited by several factors, including the small sample size and likely focus on early adopters of technology, which may not represent most patients with COPD, the researchers noted. Other limitations included the recruitment of most patients after the start of the COVID-19 pandemic, which may have affected their experience and also limited the assessment of the app on communication with healthcare providers, the researchers noted. The study also did not address financial or social barriers.
However, the results suggest that patients with COPD identified the potential value of wearable devices for disease management and that improved technology could promote patient empowerment and lifestyle changes, the researchers concluded.
Technology Can Augment Care and Connections
“As clinicians and researchers, we have ideas about what patients would want, but it is always better to get their feedback of what they really want and what they would use,” Dr. Wu told this news organization. “We thought older adults with COPD would be less likely to engage with the technology. We found that many wanted to have their data to help make connections with their condition, and some purchased smartwatches after the study to make these connections,” he said.
The takeaway message from the current study is that people with COPD may benefit from self-management apps, but they would like to use them in collaboration with their healthcare team, said Dr. Wu. “Clinicians may see more of their patients bringing in data from wearables and apps,” he noted.
Concerns persist that using technology to help support people with COPD could increase the “digital divide” and that those with lower digital literacy, financial insecurity, or English as a second language could be left behind, and it is important to remain attentive to equity in pursuing the use of devices and apps, Dr. Wu told this news organization.
Looking ahead, research involving self-management, remote monitoring, and wearable devices has focused on other conditions such as heart failure and diabetes, and more work is needed to examine how these technologies can improve care for patients with COPD, said Dr. Wu. “We see this study as one important step — to understand what will motivate people to use self-management apps and wearables,” he said.
“Acute exacerbations of COPD are very important events that can alter quality of life, lung function, and even mortality in COPD,” said Nathaniel Marchetti, DO, medical director of the Respiratory Intensive Care Unit at Temple University Hospital, Philadelphia, in an interview.
“Many of these exacerbations are not recognized by clinicians or even patients until they present late and end up in an urgent office visit with a physician or in the emergency room [ER], so addressing exacerbations earlier has the potential to avoid ER visits or hospitalizations,” he said.
The study identified areas for further research, Dr. Marchetti said. “More information would be needed to determine if the use of an app to monitor heart rate, symptoms, and oxygen saturation could alter important outcomes in COPD such as exacerbations,” he noted.
As for limitations, “no one wants to carry two smartphones,” said Dr. Marchetti. “Future devices need to be easy to use and available on the patient’s own phone,” he said. Patients should be able to choose a smartwatch or possibly a bracelet that can be synced to a smartphone, he added. The current study also failed to address what would be done with collected data, such as link them to health professionals who would offer treatment when needed, he said.
Overall, the data from the current study suggest that patients with COPD would like some device that monitors symptoms and vital signs and offers suggestions/incentives to exercise and take medications, Dr. Marchetti told this news organization. “A larger study will be needed that compares how such a device could improve outcomes of COPD; outcomes could include admissions/ER visits, exercise performance, or compliance with medication,” he said. In addition, clinical algorithms for the identification and treatment of acute exacerbations of COPD would be needed, Dr. Marchetti noted. These algorithms would determine whether treatment decisions would be initiated by a clinical team of health professionals or whether clinicians would provide medications that the patients would then decide to take based on data collected on the app, using the investigator-provided algorithms, he said.
The study was supported in part by Samsung Research America (SRA) and was initiated by Dr. Wu with input from SRA, but the company had no role in the methods or results. The study also was supported by grants from the National Natural Science Foundation of China.
Dr. Marchetti had no relevant financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Could mobile technology help patients with chronic obstructive pulmonary disease (COPD) who may not seek care until they experience an exacerbation?
Self-management interventions for COPD can potentially improve quality of life and reduce hospitalizations, wrote Robert Wu, MD, associate professor in the Department of Medicine at the University of Toronto, and colleagues. However, data on the use of devices and apps to manage COPD by providing reminders for self-care, predicting early exacerbations, and facilitating communication with healthcare providers are limited, they said.
In a study published in COPD: Journal of Chronic Obstructive Pulmonary Disease, the researchers reported details from interviews with 26 adult patients with COPD who used a wearable device and app for 6 months to help manage their condition. The interviews were part of a larger cohort study.
“The motivation for this study was to understand the patient perspective on using wearables to help support their chronic lung condition,” Dr. Wu said in an interview. “People with COPD can be at high risk of being admitted to hospital, so it is important to see if innovative technology like wearables or remote monitoring can help them,” he said.
Individuals with COPD tend to be older and less technologically adept, and they may be less willing to adopt new technology, he added. “We wanted to understand what would make people use a self-management app,” he said.
On enrollment in the study, patients received a smartwatch and a smartphone with a preinstalled app for COPD management. The app included daily reminders to take medication, perform guided breathing sessions, check blood oxygen on the smartwatch or an oximeter, and complete a symptom questionnaire. The app also allowed participants to record when they exercised and provided feedback on heart rate and daily activity, including passive step counts. Participants earned stars for meeting daily exercise goals of active minutes and total steps.
Participants received training in the use of the app from members of the research team and completed semi-structured interviews after using the items for 6 months.
The researchers divided their findings into four main themes: information, support and reassurance; barriers to adoption; impact on communication with healthcare providers; and opportunities for improvement.
Overall, most patients reported that the feedback they received through the app was useful. In particular, participants reported that the app and smartwatch provided reassurance and feedback about stable vitals during exercise, which encouraged some to adhere to regular exercise routines. Approximately two thirds (65%) said that the daily exercise reminders were motivational. In addition, 20% reported that they interpreted vital data, including heart rate, as a signal to slow down.
Participants rated medication reminders and the option to create an action plan for COPD management as the least useful features; 69% said that they already had medication reminders in place.
A total of four patients experienced technical difficulties with the app that kept it from impacting their disease management. Some of the suggestions from participants for improvement included adding information about food intake, weight, blood pressure, and temperature to the health information being tracked, as well as restoring the oxygen saturation measure, which had been disabled because of accuracy concerns. Barriers to use of the device and app included the bulkiness of the device as well as the reported technical malfunctions.
The findings were limited by several factors, including the small sample size and likely focus on early adopters of technology, which may not represent most patients with COPD, the researchers noted. Other limitations included the recruitment of most patients after the start of the COVID-19 pandemic, which may have affected their experience and also limited the assessment of the app on communication with healthcare providers, the researchers noted. The study also did not address financial or social barriers.
However, the results suggest that patients with COPD identified the potential value of wearable devices for disease management and that improved technology could promote patient empowerment and lifestyle changes, the researchers concluded.
Technology Can Augment Care and Connections
“As clinicians and researchers, we have ideas about what patients would want, but it is always better to get their feedback of what they really want and what they would use,” Dr. Wu told this news organization. “We thought older adults with COPD would be less likely to engage with the technology. We found that many wanted to have their data to help make connections with their condition, and some purchased smartwatches after the study to make these connections,” he said.
The takeaway message from the current study is that people with COPD may benefit from self-management apps, but they would like to use them in collaboration with their healthcare team, said Dr. Wu. “Clinicians may see more of their patients bringing in data from wearables and apps,” he noted.
Concerns persist that using technology to help support people with COPD could increase the “digital divide” and that those with lower digital literacy, financial insecurity, or English as a second language could be left behind, and it is important to remain attentive to equity in pursuing the use of devices and apps, Dr. Wu told this news organization.
Looking ahead, research involving self-management, remote monitoring, and wearable devices has focused on other conditions such as heart failure and diabetes, and more work is needed to examine how these technologies can improve care for patients with COPD, said Dr. Wu. “We see this study as one important step — to understand what will motivate people to use self-management apps and wearables,” he said.
“Acute exacerbations of COPD are very important events that can alter quality of life, lung function, and even mortality in COPD,” said Nathaniel Marchetti, DO, medical director of the Respiratory Intensive Care Unit at Temple University Hospital, Philadelphia, in an interview.
“Many of these exacerbations are not recognized by clinicians or even patients until they present late and end up in an urgent office visit with a physician or in the emergency room [ER], so addressing exacerbations earlier has the potential to avoid ER visits or hospitalizations,” he said.
The study identified areas for further research, Dr. Marchetti said. “More information would be needed to determine if the use of an app to monitor heart rate, symptoms, and oxygen saturation could alter important outcomes in COPD such as exacerbations,” he noted.
As for limitations, “no one wants to carry two smartphones,” said Dr. Marchetti. “Future devices need to be easy to use and available on the patient’s own phone,” he said. Patients should be able to choose a smartwatch or possibly a bracelet that can be synced to a smartphone, he added. The current study also failed to address what would be done with collected data, such as link them to health professionals who would offer treatment when needed, he said.
Overall, the data from the current study suggest that patients with COPD would like some device that monitors symptoms and vital signs and offers suggestions/incentives to exercise and take medications, Dr. Marchetti told this news organization. “A larger study will be needed that compares how such a device could improve outcomes of COPD; outcomes could include admissions/ER visits, exercise performance, or compliance with medication,” he said. In addition, clinical algorithms for the identification and treatment of acute exacerbations of COPD would be needed, Dr. Marchetti noted. These algorithms would determine whether treatment decisions would be initiated by a clinical team of health professionals or whether clinicians would provide medications that the patients would then decide to take based on data collected on the app, using the investigator-provided algorithms, he said.
The study was supported in part by Samsung Research America (SRA) and was initiated by Dr. Wu with input from SRA, but the company had no role in the methods or results. The study also was supported by grants from the National Natural Science Foundation of China.
Dr. Marchetti had no relevant financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Could mobile technology help patients with chronic obstructive pulmonary disease (COPD) who may not seek care until they experience an exacerbation?
Self-management interventions for COPD can potentially improve quality of life and reduce hospitalizations, wrote Robert Wu, MD, associate professor in the Department of Medicine at the University of Toronto, and colleagues. However, data on the use of devices and apps to manage COPD by providing reminders for self-care, predicting early exacerbations, and facilitating communication with healthcare providers are limited, they said.
In a study published in COPD: Journal of Chronic Obstructive Pulmonary Disease, the researchers reported details from interviews with 26 adult patients with COPD who used a wearable device and app for 6 months to help manage their condition. The interviews were part of a larger cohort study.
“The motivation for this study was to understand the patient perspective on using wearables to help support their chronic lung condition,” Dr. Wu said in an interview. “People with COPD can be at high risk of being admitted to hospital, so it is important to see if innovative technology like wearables or remote monitoring can help them,” he said.
Individuals with COPD tend to be older and less technologically adept, and they may be less willing to adopt new technology, he added. “We wanted to understand what would make people use a self-management app,” he said.
On enrollment in the study, patients received a smartwatch and a smartphone with a preinstalled app for COPD management. The app included daily reminders to take medication, perform guided breathing sessions, check blood oxygen on the smartwatch or an oximeter, and complete a symptom questionnaire. The app also allowed participants to record when they exercised and provided feedback on heart rate and daily activity, including passive step counts. Participants earned stars for meeting daily exercise goals of active minutes and total steps.
Participants received training in the use of the app from members of the research team and completed semi-structured interviews after using the items for 6 months.
The researchers divided their findings into four main themes: information, support and reassurance; barriers to adoption; impact on communication with healthcare providers; and opportunities for improvement.
Overall, most patients reported that the feedback they received through the app was useful. In particular, participants reported that the app and smartwatch provided reassurance and feedback about stable vitals during exercise, which encouraged some to adhere to regular exercise routines. Approximately two thirds (65%) said that the daily exercise reminders were motivational. In addition, 20% reported that they interpreted vital data, including heart rate, as a signal to slow down.
Participants rated medication reminders and the option to create an action plan for COPD management as the least useful features; 69% said that they already had medication reminders in place.
A total of four patients experienced technical difficulties with the app that kept it from impacting their disease management. Some of the suggestions from participants for improvement included adding information about food intake, weight, blood pressure, and temperature to the health information being tracked, as well as restoring the oxygen saturation measure, which had been disabled because of accuracy concerns. Barriers to use of the device and app included the bulkiness of the device as well as the reported technical malfunctions.
The findings were limited by several factors, including the small sample size and likely focus on early adopters of technology, which may not represent most patients with COPD, the researchers noted. Other limitations included the recruitment of most patients after the start of the COVID-19 pandemic, which may have affected their experience and also limited the assessment of the app on communication with healthcare providers, the researchers noted. The study also did not address financial or social barriers.
However, the results suggest that patients with COPD identified the potential value of wearable devices for disease management and that improved technology could promote patient empowerment and lifestyle changes, the researchers concluded.
Technology Can Augment Care and Connections
“As clinicians and researchers, we have ideas about what patients would want, but it is always better to get their feedback of what they really want and what they would use,” Dr. Wu told this news organization. “We thought older adults with COPD would be less likely to engage with the technology. We found that many wanted to have their data to help make connections with their condition, and some purchased smartwatches after the study to make these connections,” he said.
The takeaway message from the current study is that people with COPD may benefit from self-management apps, but they would like to use them in collaboration with their healthcare team, said Dr. Wu. “Clinicians may see more of their patients bringing in data from wearables and apps,” he noted.
Concerns persist that using technology to help support people with COPD could increase the “digital divide” and that those with lower digital literacy, financial insecurity, or English as a second language could be left behind, and it is important to remain attentive to equity in pursuing the use of devices and apps, Dr. Wu told this news organization.
Looking ahead, research involving self-management, remote monitoring, and wearable devices has focused on other conditions such as heart failure and diabetes, and more work is needed to examine how these technologies can improve care for patients with COPD, said Dr. Wu. “We see this study as one important step — to understand what will motivate people to use self-management apps and wearables,” he said.
“Acute exacerbations of COPD are very important events that can alter quality of life, lung function, and even mortality in COPD,” said Nathaniel Marchetti, DO, medical director of the Respiratory Intensive Care Unit at Temple University Hospital, Philadelphia, in an interview.
“Many of these exacerbations are not recognized by clinicians or even patients until they present late and end up in an urgent office visit with a physician or in the emergency room [ER], so addressing exacerbations earlier has the potential to avoid ER visits or hospitalizations,” he said.
The study identified areas for further research, Dr. Marchetti said. “More information would be needed to determine if the use of an app to monitor heart rate, symptoms, and oxygen saturation could alter important outcomes in COPD such as exacerbations,” he noted.
As for limitations, “no one wants to carry two smartphones,” said Dr. Marchetti. “Future devices need to be easy to use and available on the patient’s own phone,” he said. Patients should be able to choose a smartwatch or possibly a bracelet that can be synced to a smartphone, he added. The current study also failed to address what would be done with collected data, such as link them to health professionals who would offer treatment when needed, he said.
Overall, the data from the current study suggest that patients with COPD would like some device that monitors symptoms and vital signs and offers suggestions/incentives to exercise and take medications, Dr. Marchetti told this news organization. “A larger study will be needed that compares how such a device could improve outcomes of COPD; outcomes could include admissions/ER visits, exercise performance, or compliance with medication,” he said. In addition, clinical algorithms for the identification and treatment of acute exacerbations of COPD would be needed, Dr. Marchetti noted. These algorithms would determine whether treatment decisions would be initiated by a clinical team of health professionals or whether clinicians would provide medications that the patients would then decide to take based on data collected on the app, using the investigator-provided algorithms, he said.
The study was supported in part by Samsung Research America (SRA) and was initiated by Dr. Wu with input from SRA, but the company had no role in the methods or results. The study also was supported by grants from the National Natural Science Foundation of China.
Dr. Marchetti had no relevant financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Ultraprocessed Foods Upped Risk for Diabetic Complications
TOPLINE:
In patients with type 2 diabetes (T2D), eating more ultraprocessed food (UPF) increased the overall risk for microvascular complications and for diabetic kidney disease in particular. The risk was partly mediated by biomarkers related to body weight, lipid metabolism, and inflammation.
METHODOLOGY:
- Researchers evaluated the association between the intake of UPF and the risk for diabetic microvascular complications in a prospective cohort of 5685 participants with T2D (mean age, 59.7 years; 63.8% men) from the UK Biobank.
- Dietary information of participants was collected with a web-based 24-hour dietary recall tool that recorded the frequency of consumption of 206 foods and 32 beverages.
- Researchers found five patterns that accounted for one third of UPF intake variation by estimated weight (not calories): Bread and spreads; cereal with liquids; high dairy and low cured meat; sugary beverages and snacks; and mixed beverages and savory snacks.
- The outcomes included the risk for overall microvascular complications; for diabetic retinopathy, diabetic neuropathy, and diabetic kidney disease; and for biomarkers related to microvascular complications.
TAKEAWAY:
- During a median follow-up duration of 12.7 years, 1243 composite microvascular complications were reported, including 599 diabetic retinopathy, 237 diabetic neuropathy, and 662 diabetic kidney disease events.
- Each 10% increase in the proportion of UPF consumption increased the risk for composite microvascular complications by 8% (hazard ratio [HR], 1.08; 95% CI, 1.03-1.13) and diabetic kidney disease by 13% (HR 1.13; 95% CI, 1.06-1.20). No significant UPF intake association was found with diabetic retinopathy or diabetic neuropathy.
- In the biomarker mediation analysis, body mass index, triglycerides, and C-reactive protein collectively explained 22% (P < .001) and 15.8% (P < .001) of the associations of UPF consumption with composite microvascular complications and diabetic kidney disease, respectively.
- The food pattern rich in sugary beverages and snacks increased the risk for diabetic kidney disease, whereas the pattern rich in mixed beverages and savory snacks increased the risk for composite microvascular complications and diabetic retinopathy.
IN PRACTICE:
“In view of microvascular complications, our findings further support adhering to the recommendations outlined in the American Diabetes Association’s 2022 guidelines, which advocate for the preference of whole foods over highly processed ones,” the authors wrote.
SOURCE:
The study was led by Yue Li, MBBS, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. It was published online in The American Journal of Clinical Nutrition.
LIMITATIONS:
The dietary recall used in the UK Biobank was not specifically designed to collect dietary data according to the Nova food categories used in the study, which may have led to misclassifications. Data on usual dietary intake may not have been captured accurately, as all participants did not provide multiple dietary recalls. Individuals with T2D who completed dietary assessments were more likely to have a higher socioeconomic status and healthier lifestyle than those not filling the assessment, which could have resulted in an underrepresentation of high UPF consumers.
DISCLOSURES:
Some authors received funding from the National Natural Science Foundation of China, the National Key Research and Development Program of China, and other government sources. None of the authors declared any competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In patients with type 2 diabetes (T2D), eating more ultraprocessed food (UPF) increased the overall risk for microvascular complications and for diabetic kidney disease in particular. The risk was partly mediated by biomarkers related to body weight, lipid metabolism, and inflammation.
METHODOLOGY:
- Researchers evaluated the association between the intake of UPF and the risk for diabetic microvascular complications in a prospective cohort of 5685 participants with T2D (mean age, 59.7 years; 63.8% men) from the UK Biobank.
- Dietary information of participants was collected with a web-based 24-hour dietary recall tool that recorded the frequency of consumption of 206 foods and 32 beverages.
- Researchers found five patterns that accounted for one third of UPF intake variation by estimated weight (not calories): Bread and spreads; cereal with liquids; high dairy and low cured meat; sugary beverages and snacks; and mixed beverages and savory snacks.
- The outcomes included the risk for overall microvascular complications; for diabetic retinopathy, diabetic neuropathy, and diabetic kidney disease; and for biomarkers related to microvascular complications.
TAKEAWAY:
- During a median follow-up duration of 12.7 years, 1243 composite microvascular complications were reported, including 599 diabetic retinopathy, 237 diabetic neuropathy, and 662 diabetic kidney disease events.
- Each 10% increase in the proportion of UPF consumption increased the risk for composite microvascular complications by 8% (hazard ratio [HR], 1.08; 95% CI, 1.03-1.13) and diabetic kidney disease by 13% (HR 1.13; 95% CI, 1.06-1.20). No significant UPF intake association was found with diabetic retinopathy or diabetic neuropathy.
- In the biomarker mediation analysis, body mass index, triglycerides, and C-reactive protein collectively explained 22% (P < .001) and 15.8% (P < .001) of the associations of UPF consumption with composite microvascular complications and diabetic kidney disease, respectively.
- The food pattern rich in sugary beverages and snacks increased the risk for diabetic kidney disease, whereas the pattern rich in mixed beverages and savory snacks increased the risk for composite microvascular complications and diabetic retinopathy.
IN PRACTICE:
“In view of microvascular complications, our findings further support adhering to the recommendations outlined in the American Diabetes Association’s 2022 guidelines, which advocate for the preference of whole foods over highly processed ones,” the authors wrote.
SOURCE:
The study was led by Yue Li, MBBS, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. It was published online in The American Journal of Clinical Nutrition.
LIMITATIONS:
The dietary recall used in the UK Biobank was not specifically designed to collect dietary data according to the Nova food categories used in the study, which may have led to misclassifications. Data on usual dietary intake may not have been captured accurately, as all participants did not provide multiple dietary recalls. Individuals with T2D who completed dietary assessments were more likely to have a higher socioeconomic status and healthier lifestyle than those not filling the assessment, which could have resulted in an underrepresentation of high UPF consumers.
DISCLOSURES:
Some authors received funding from the National Natural Science Foundation of China, the National Key Research and Development Program of China, and other government sources. None of the authors declared any competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In patients with type 2 diabetes (T2D), eating more ultraprocessed food (UPF) increased the overall risk for microvascular complications and for diabetic kidney disease in particular. The risk was partly mediated by biomarkers related to body weight, lipid metabolism, and inflammation.
METHODOLOGY:
- Researchers evaluated the association between the intake of UPF and the risk for diabetic microvascular complications in a prospective cohort of 5685 participants with T2D (mean age, 59.7 years; 63.8% men) from the UK Biobank.
- Dietary information of participants was collected with a web-based 24-hour dietary recall tool that recorded the frequency of consumption of 206 foods and 32 beverages.
- Researchers found five patterns that accounted for one third of UPF intake variation by estimated weight (not calories): Bread and spreads; cereal with liquids; high dairy and low cured meat; sugary beverages and snacks; and mixed beverages and savory snacks.
- The outcomes included the risk for overall microvascular complications; for diabetic retinopathy, diabetic neuropathy, and diabetic kidney disease; and for biomarkers related to microvascular complications.
TAKEAWAY:
- During a median follow-up duration of 12.7 years, 1243 composite microvascular complications were reported, including 599 diabetic retinopathy, 237 diabetic neuropathy, and 662 diabetic kidney disease events.
- Each 10% increase in the proportion of UPF consumption increased the risk for composite microvascular complications by 8% (hazard ratio [HR], 1.08; 95% CI, 1.03-1.13) and diabetic kidney disease by 13% (HR 1.13; 95% CI, 1.06-1.20). No significant UPF intake association was found with diabetic retinopathy or diabetic neuropathy.
- In the biomarker mediation analysis, body mass index, triglycerides, and C-reactive protein collectively explained 22% (P < .001) and 15.8% (P < .001) of the associations of UPF consumption with composite microvascular complications and diabetic kidney disease, respectively.
- The food pattern rich in sugary beverages and snacks increased the risk for diabetic kidney disease, whereas the pattern rich in mixed beverages and savory snacks increased the risk for composite microvascular complications and diabetic retinopathy.
IN PRACTICE:
“In view of microvascular complications, our findings further support adhering to the recommendations outlined in the American Diabetes Association’s 2022 guidelines, which advocate for the preference of whole foods over highly processed ones,” the authors wrote.
SOURCE:
The study was led by Yue Li, MBBS, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. It was published online in The American Journal of Clinical Nutrition.
LIMITATIONS:
The dietary recall used in the UK Biobank was not specifically designed to collect dietary data according to the Nova food categories used in the study, which may have led to misclassifications. Data on usual dietary intake may not have been captured accurately, as all participants did not provide multiple dietary recalls. Individuals with T2D who completed dietary assessments were more likely to have a higher socioeconomic status and healthier lifestyle than those not filling the assessment, which could have resulted in an underrepresentation of high UPF consumers.
DISCLOSURES:
Some authors received funding from the National Natural Science Foundation of China, the National Key Research and Development Program of China, and other government sources. None of the authors declared any competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
What Would ‘Project 2025’ Mean for Health and Healthcare?
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Data Trends 2024: Amyotrophic Lateral Sclerosis (ALS)
- Mehta P, Raymond J, Zhang Y, et al. Prevalence of amyotrophic lateral sclerosis in the United States, 2018. Amyotroph Lateral Scler Frontotemporal Degener. Published online August 21, 2023. doi:10.1080/21678421.2023.2245858
- What is amyotrophic lateral sclerosis (ALS)? Centers for Disease Control and Prevention. Updated May 13, 2022. Accessed April 15, 2024. https://www.cdc.gov/als/WhatisAmyotrophiclateralsclerosis.html
- Reimer RJ, Goncalves A, Soper B, et al. An electronic health record cohort of veterans with amyotrophic lateral sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. Published online August 9, 2023. doi:10.1080/21678421.2023.2239300
- Kudritzki V, Howard IM. Telehealth-based exercise in amyotrophic lateral sclerosis. Front Neurol. 2023;14:1238916. doi:10.3389/fneur.2023.1238916
- Colvin LE, Foster ZW, Stein TD, et al. Utility of the ALSFRS-R for predicting ALS and comorbid disease neuropathology: the Veterans Affairs Biorepository Brain Bank. Muscle Nerve. 2022;66(2):167-174. doi:10.1002/mus.27635
- Rabadi MH, Russell KC, Xu C. Predictors of mortality in veterans with amyotrophic lateral sclerosis: respiratory status and speech disorder at presentation. Med Sci Monit. 2024;30:e943288. doi:10.12659/MSM.943288
- Mehta P, Raymond J, Zhang Y, et al. Prevalence of amyotrophic lateral sclerosis in the United States, 2018. Amyotroph Lateral Scler Frontotemporal Degener. Published online August 21, 2023. doi:10.1080/21678421.2023.2245858
- What is amyotrophic lateral sclerosis (ALS)? Centers for Disease Control and Prevention. Updated May 13, 2022. Accessed April 15, 2024. https://www.cdc.gov/als/WhatisAmyotrophiclateralsclerosis.html
- Reimer RJ, Goncalves A, Soper B, et al. An electronic health record cohort of veterans with amyotrophic lateral sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. Published online August 9, 2023. doi:10.1080/21678421.2023.2239300
- Kudritzki V, Howard IM. Telehealth-based exercise in amyotrophic lateral sclerosis. Front Neurol. 2023;14:1238916. doi:10.3389/fneur.2023.1238916
- Colvin LE, Foster ZW, Stein TD, et al. Utility of the ALSFRS-R for predicting ALS and comorbid disease neuropathology: the Veterans Affairs Biorepository Brain Bank. Muscle Nerve. 2022;66(2):167-174. doi:10.1002/mus.27635
- Rabadi MH, Russell KC, Xu C. Predictors of mortality in veterans with amyotrophic lateral sclerosis: respiratory status and speech disorder at presentation. Med Sci Monit. 2024;30:e943288. doi:10.12659/MSM.943288
- Mehta P, Raymond J, Zhang Y, et al. Prevalence of amyotrophic lateral sclerosis in the United States, 2018. Amyotroph Lateral Scler Frontotemporal Degener. Published online August 21, 2023. doi:10.1080/21678421.2023.2245858
- What is amyotrophic lateral sclerosis (ALS)? Centers for Disease Control and Prevention. Updated May 13, 2022. Accessed April 15, 2024. https://www.cdc.gov/als/WhatisAmyotrophiclateralsclerosis.html
- Reimer RJ, Goncalves A, Soper B, et al. An electronic health record cohort of veterans with amyotrophic lateral sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. Published online August 9, 2023. doi:10.1080/21678421.2023.2239300
- Kudritzki V, Howard IM. Telehealth-based exercise in amyotrophic lateral sclerosis. Front Neurol. 2023;14:1238916. doi:10.3389/fneur.2023.1238916
- Colvin LE, Foster ZW, Stein TD, et al. Utility of the ALSFRS-R for predicting ALS and comorbid disease neuropathology: the Veterans Affairs Biorepository Brain Bank. Muscle Nerve. 2022;66(2):167-174. doi:10.1002/mus.27635
- Rabadi MH, Russell KC, Xu C. Predictors of mortality in veterans with amyotrophic lateral sclerosis: respiratory status and speech disorder at presentation. Med Sci Monit. 2024;30:e943288. doi:10.12659/MSM.943288
‘Blood Everywhere’: Nurses Control In-Flight Crisis
My husband Scott and I were flying back to Washington state with our two kids, who were about 1 and 4. We had been in Florida for a family vacation, and we were near the end of the flight, with both kids passed out on top of me.
Suddenly, there was some scuffling and a lot of movement from the flight attendants. The announcement came: “Are there any healthcare providers on board?” My husband and I are both nurses. We looked at each other, and we looked at our sleeping kids. Should we say anything?
One of the stewardesses walked by looking very flustered. My husband was in the aisle seat, so he leaned out and told her that we were nurses. Her eyes got all big, and she said: “Oh yeah, come on up.”
She was looking at both of us. I said, “I think he’s got it.” I assumed it wasn’t that big of a deal. Plus — kids sleeping on top of me.
Scott went up to the front of the plane. But a few minutes later, the stewardess came back and said: “You need to help.” I was holding my 1-year-old son, so I handed her my kid. She sat down with him, and I boogied up to the front of the plane.
I got to the first-class stewards’ area where the restrooms are and the cabinets with all the food and drinks.
When I saw the bleeding, my first reaction was we need to apply pressure. I asked for a towel. There were no towels. A blanket? Anything to help absorb the blood? Nope. They had nothing. I was given a pair of gloves that were much too big and a fistful of cocktail napkins.
It was such a small space there wasn’t any way to be next to the man. So, I kind of squatted over the top of him to reach behind his head. I got a stack of napkins on there and held pressure as hard as I could with the tips of my fingers on one hand.
I’m a postanesthesia care unit nurse, so my next thought was to check his pupils and make sure he had a good airway by doing a jaw thrust and a chin lift. I noticed there was blood in his mouth. His breathing was in short gusts. I was trying to do all that with my free hand without crushing him with my body.
Scott had made some ice packs, so I applied those as well, which helped to constrict the bleeding. Then he checked the plane’s medical kit to try to get an intravenous (IV) started. It wasn’t easy. The IV start kit was very different from what you would normally use. And at the same time, the plane had started to descend for landing, so we were on an angle. But he tried.
We asked about what had happened. The steward team said the man had fallen and hit his head on one of the stainless steel cabinets. He seemed to be in his 70s or 80s, a tall, solid guy.
His wife was sitting nearby — pretty calm and stoic given the circumstances. We asked her about his medical history, trying to get a feel for why he might be unconscious. He was still totally out. She told us he had diabetes. He was on a blood pressure medication and also a blood thinner.
The plane kept going down. I was in a really awkward position, squatting and holding myself up against the cabinets. I just kept talking to the man, trying to get him to wake up. “Can you hear me? Everything’s okay. You hit your head.”
Someone brought us an oxygen tank. I looked for the mask. And realized it wasn’t a mask. It was a plastic bag. I set it on the patient’s face, and it felt like I was suffocating him. So, I tried to do it blowby to just increase the oxygen in the air near his face.
At one point, his breathing was agonal for a few minutes, which really concerned me. My fear was that he was going to stop breathing. I rubbed his chest and kind of said: “Hey, let’s not do that!”
I would have felt a lot better about resuscitating him with an actual oxygen mask rather than a plastic bag.
The amount of blood definitely looked alarming. I couldn’t tell how much he was actively bleeding. But it was a lot. He wasn’t turning gray though, so that was a good sign.
Finally, he started coming to and opening his eyes. I introduced myself and asked him: “Do you know where you are? Do you know what’s going on?” Trying to see if he was oriented at all.
Eventually, he was able to talk to me, so I kept asking questions: “Are you guys on vacation? Where are you headed? Where are you staying?”
He told me they were going to visit his granddaughter, and he was able to talk about that. He didn’t try to get up, which I was glad about, because that would’ve been really challenging to navigate.
I could tell he was embarrassed about what had happened. I’ve helped a lot of older gentlemen after falling down, and their egos are often bruised. They don’t want to be in a position of needing help.
Finally, the plane landed. There was blood absolutely everywhere. The ice packs had melted, and the water had mixed with the pool of blood. It was such a mess.
The pilots had called the airport ahead to let them know we needed medical services. So, the first responding team came on right away. They stabilized the man with a board, put the neck brace on him, and did all the stuff you do for a patient after a fall.
I gave them a report — that’s just my style. But it didn’t seem like they needed a lot of information at that point.
I was finally able to talk to the man’s wife who was clearly terrified. I gave her a hug and told her he would be all right. She thanked us.
The emergency team didn’t seem to have anything to help staunch the bleeding either because the rolling gurney left puddles of blood all down the gangway, causing a significant biohazard problem.
They let one person leave who had a connecting flight, but everyone else had to get off from the rear of the plane and walk across the tarmac.
When we finally got back to our seats, the stewardess was still sitting with our kids. They were both totally chill, watching some show, apparently very well behaved. Our daughter asked us what was going on, and I said: “Oh, somebody got hurt at the front of the plane.” She’s so used to hearing that we work with sick people that it didn’t faze her at all.
As we left, we got a lot of thank-yous from people who had been sitting up front and saw what happened.
When we got home, there was still blood on my shoes. I remember looking at them and thinking: Disinfect or throw away? I disinfected them. They were still a good pair of shoes.
A few days later, we got an email from the airline with a voucher, expressing their gratitude for our help. That was nice and unexpected.
I responded with a suggestion: How about having some protocols for medical events on airplanes? Pilots go through checklists for almost everything they do. Why wouldn’t they have something like that for medical responses?
I also asked how the man and his wife were doing. But they couldn’t disclose that information.
It was certainly strange being out of my element, helping a patient in that tiny little space; I’m used to working in a recovery room where you have literally everything you need within arm’s reach — the Ambu bag, suction, and bandages. And with airway management, there’s usually more than one person in the room to assist. If there’s a problem, a whole bunch of people show up around the bed so fast.
I’m definitely thinking about field medicine a lot more. Wondering what I would do in certain situations. While debriefing with my mom (an advanced registered nurse practitioner), she pointed out that we should have asked passengers for sanitary pads or diapers to stabilize the bleeding instead of the cocktail napkins. Brilliant idea! I didn’t think of it in the moment. But I’m keeping that little tip tucked in my back pocket for any future bleeding-in-the-wild scenarios.
Audra Podruzny, MSN, RN, CPAN, lives in Washington state and is currently attending the Washington State University Doctor of Nursing Practice Family Nurse Practitioner program.
A version of this article first appeared on Medscape.com.
My husband Scott and I were flying back to Washington state with our two kids, who were about 1 and 4. We had been in Florida for a family vacation, and we were near the end of the flight, with both kids passed out on top of me.
Suddenly, there was some scuffling and a lot of movement from the flight attendants. The announcement came: “Are there any healthcare providers on board?” My husband and I are both nurses. We looked at each other, and we looked at our sleeping kids. Should we say anything?
One of the stewardesses walked by looking very flustered. My husband was in the aisle seat, so he leaned out and told her that we were nurses. Her eyes got all big, and she said: “Oh yeah, come on up.”
She was looking at both of us. I said, “I think he’s got it.” I assumed it wasn’t that big of a deal. Plus — kids sleeping on top of me.
Scott went up to the front of the plane. But a few minutes later, the stewardess came back and said: “You need to help.” I was holding my 1-year-old son, so I handed her my kid. She sat down with him, and I boogied up to the front of the plane.
I got to the first-class stewards’ area where the restrooms are and the cabinets with all the food and drinks.
When I saw the bleeding, my first reaction was we need to apply pressure. I asked for a towel. There were no towels. A blanket? Anything to help absorb the blood? Nope. They had nothing. I was given a pair of gloves that were much too big and a fistful of cocktail napkins.
It was such a small space there wasn’t any way to be next to the man. So, I kind of squatted over the top of him to reach behind his head. I got a stack of napkins on there and held pressure as hard as I could with the tips of my fingers on one hand.
I’m a postanesthesia care unit nurse, so my next thought was to check his pupils and make sure he had a good airway by doing a jaw thrust and a chin lift. I noticed there was blood in his mouth. His breathing was in short gusts. I was trying to do all that with my free hand without crushing him with my body.
Scott had made some ice packs, so I applied those as well, which helped to constrict the bleeding. Then he checked the plane’s medical kit to try to get an intravenous (IV) started. It wasn’t easy. The IV start kit was very different from what you would normally use. And at the same time, the plane had started to descend for landing, so we were on an angle. But he tried.
We asked about what had happened. The steward team said the man had fallen and hit his head on one of the stainless steel cabinets. He seemed to be in his 70s or 80s, a tall, solid guy.
His wife was sitting nearby — pretty calm and stoic given the circumstances. We asked her about his medical history, trying to get a feel for why he might be unconscious. He was still totally out. She told us he had diabetes. He was on a blood pressure medication and also a blood thinner.
The plane kept going down. I was in a really awkward position, squatting and holding myself up against the cabinets. I just kept talking to the man, trying to get him to wake up. “Can you hear me? Everything’s okay. You hit your head.”
Someone brought us an oxygen tank. I looked for the mask. And realized it wasn’t a mask. It was a plastic bag. I set it on the patient’s face, and it felt like I was suffocating him. So, I tried to do it blowby to just increase the oxygen in the air near his face.
At one point, his breathing was agonal for a few minutes, which really concerned me. My fear was that he was going to stop breathing. I rubbed his chest and kind of said: “Hey, let’s not do that!”
I would have felt a lot better about resuscitating him with an actual oxygen mask rather than a plastic bag.
The amount of blood definitely looked alarming. I couldn’t tell how much he was actively bleeding. But it was a lot. He wasn’t turning gray though, so that was a good sign.
Finally, he started coming to and opening his eyes. I introduced myself and asked him: “Do you know where you are? Do you know what’s going on?” Trying to see if he was oriented at all.
Eventually, he was able to talk to me, so I kept asking questions: “Are you guys on vacation? Where are you headed? Where are you staying?”
He told me they were going to visit his granddaughter, and he was able to talk about that. He didn’t try to get up, which I was glad about, because that would’ve been really challenging to navigate.
I could tell he was embarrassed about what had happened. I’ve helped a lot of older gentlemen after falling down, and their egos are often bruised. They don’t want to be in a position of needing help.
Finally, the plane landed. There was blood absolutely everywhere. The ice packs had melted, and the water had mixed with the pool of blood. It was such a mess.
The pilots had called the airport ahead to let them know we needed medical services. So, the first responding team came on right away. They stabilized the man with a board, put the neck brace on him, and did all the stuff you do for a patient after a fall.
I gave them a report — that’s just my style. But it didn’t seem like they needed a lot of information at that point.
I was finally able to talk to the man’s wife who was clearly terrified. I gave her a hug and told her he would be all right. She thanked us.
The emergency team didn’t seem to have anything to help staunch the bleeding either because the rolling gurney left puddles of blood all down the gangway, causing a significant biohazard problem.
They let one person leave who had a connecting flight, but everyone else had to get off from the rear of the plane and walk across the tarmac.
When we finally got back to our seats, the stewardess was still sitting with our kids. They were both totally chill, watching some show, apparently very well behaved. Our daughter asked us what was going on, and I said: “Oh, somebody got hurt at the front of the plane.” She’s so used to hearing that we work with sick people that it didn’t faze her at all.
As we left, we got a lot of thank-yous from people who had been sitting up front and saw what happened.
When we got home, there was still blood on my shoes. I remember looking at them and thinking: Disinfect or throw away? I disinfected them. They were still a good pair of shoes.
A few days later, we got an email from the airline with a voucher, expressing their gratitude for our help. That was nice and unexpected.
I responded with a suggestion: How about having some protocols for medical events on airplanes? Pilots go through checklists for almost everything they do. Why wouldn’t they have something like that for medical responses?
I also asked how the man and his wife were doing. But they couldn’t disclose that information.
It was certainly strange being out of my element, helping a patient in that tiny little space; I’m used to working in a recovery room where you have literally everything you need within arm’s reach — the Ambu bag, suction, and bandages. And with airway management, there’s usually more than one person in the room to assist. If there’s a problem, a whole bunch of people show up around the bed so fast.
I’m definitely thinking about field medicine a lot more. Wondering what I would do in certain situations. While debriefing with my mom (an advanced registered nurse practitioner), she pointed out that we should have asked passengers for sanitary pads or diapers to stabilize the bleeding instead of the cocktail napkins. Brilliant idea! I didn’t think of it in the moment. But I’m keeping that little tip tucked in my back pocket for any future bleeding-in-the-wild scenarios.
Audra Podruzny, MSN, RN, CPAN, lives in Washington state and is currently attending the Washington State University Doctor of Nursing Practice Family Nurse Practitioner program.
A version of this article first appeared on Medscape.com.
My husband Scott and I were flying back to Washington state with our two kids, who were about 1 and 4. We had been in Florida for a family vacation, and we were near the end of the flight, with both kids passed out on top of me.
Suddenly, there was some scuffling and a lot of movement from the flight attendants. The announcement came: “Are there any healthcare providers on board?” My husband and I are both nurses. We looked at each other, and we looked at our sleeping kids. Should we say anything?
One of the stewardesses walked by looking very flustered. My husband was in the aisle seat, so he leaned out and told her that we were nurses. Her eyes got all big, and she said: “Oh yeah, come on up.”
She was looking at both of us. I said, “I think he’s got it.” I assumed it wasn’t that big of a deal. Plus — kids sleeping on top of me.
Scott went up to the front of the plane. But a few minutes later, the stewardess came back and said: “You need to help.” I was holding my 1-year-old son, so I handed her my kid. She sat down with him, and I boogied up to the front of the plane.
I got to the first-class stewards’ area where the restrooms are and the cabinets with all the food and drinks.
When I saw the bleeding, my first reaction was we need to apply pressure. I asked for a towel. There were no towels. A blanket? Anything to help absorb the blood? Nope. They had nothing. I was given a pair of gloves that were much too big and a fistful of cocktail napkins.
It was such a small space there wasn’t any way to be next to the man. So, I kind of squatted over the top of him to reach behind his head. I got a stack of napkins on there and held pressure as hard as I could with the tips of my fingers on one hand.
I’m a postanesthesia care unit nurse, so my next thought was to check his pupils and make sure he had a good airway by doing a jaw thrust and a chin lift. I noticed there was blood in his mouth. His breathing was in short gusts. I was trying to do all that with my free hand without crushing him with my body.
Scott had made some ice packs, so I applied those as well, which helped to constrict the bleeding. Then he checked the plane’s medical kit to try to get an intravenous (IV) started. It wasn’t easy. The IV start kit was very different from what you would normally use. And at the same time, the plane had started to descend for landing, so we were on an angle. But he tried.
We asked about what had happened. The steward team said the man had fallen and hit his head on one of the stainless steel cabinets. He seemed to be in his 70s or 80s, a tall, solid guy.
His wife was sitting nearby — pretty calm and stoic given the circumstances. We asked her about his medical history, trying to get a feel for why he might be unconscious. He was still totally out. She told us he had diabetes. He was on a blood pressure medication and also a blood thinner.
The plane kept going down. I was in a really awkward position, squatting and holding myself up against the cabinets. I just kept talking to the man, trying to get him to wake up. “Can you hear me? Everything’s okay. You hit your head.”
Someone brought us an oxygen tank. I looked for the mask. And realized it wasn’t a mask. It was a plastic bag. I set it on the patient’s face, and it felt like I was suffocating him. So, I tried to do it blowby to just increase the oxygen in the air near his face.
At one point, his breathing was agonal for a few minutes, which really concerned me. My fear was that he was going to stop breathing. I rubbed his chest and kind of said: “Hey, let’s not do that!”
I would have felt a lot better about resuscitating him with an actual oxygen mask rather than a plastic bag.
The amount of blood definitely looked alarming. I couldn’t tell how much he was actively bleeding. But it was a lot. He wasn’t turning gray though, so that was a good sign.
Finally, he started coming to and opening his eyes. I introduced myself and asked him: “Do you know where you are? Do you know what’s going on?” Trying to see if he was oriented at all.
Eventually, he was able to talk to me, so I kept asking questions: “Are you guys on vacation? Where are you headed? Where are you staying?”
He told me they were going to visit his granddaughter, and he was able to talk about that. He didn’t try to get up, which I was glad about, because that would’ve been really challenging to navigate.
I could tell he was embarrassed about what had happened. I’ve helped a lot of older gentlemen after falling down, and their egos are often bruised. They don’t want to be in a position of needing help.
Finally, the plane landed. There was blood absolutely everywhere. The ice packs had melted, and the water had mixed with the pool of blood. It was such a mess.
The pilots had called the airport ahead to let them know we needed medical services. So, the first responding team came on right away. They stabilized the man with a board, put the neck brace on him, and did all the stuff you do for a patient after a fall.
I gave them a report — that’s just my style. But it didn’t seem like they needed a lot of information at that point.
I was finally able to talk to the man’s wife who was clearly terrified. I gave her a hug and told her he would be all right. She thanked us.
The emergency team didn’t seem to have anything to help staunch the bleeding either because the rolling gurney left puddles of blood all down the gangway, causing a significant biohazard problem.
They let one person leave who had a connecting flight, but everyone else had to get off from the rear of the plane and walk across the tarmac.
When we finally got back to our seats, the stewardess was still sitting with our kids. They were both totally chill, watching some show, apparently very well behaved. Our daughter asked us what was going on, and I said: “Oh, somebody got hurt at the front of the plane.” She’s so used to hearing that we work with sick people that it didn’t faze her at all.
As we left, we got a lot of thank-yous from people who had been sitting up front and saw what happened.
When we got home, there was still blood on my shoes. I remember looking at them and thinking: Disinfect or throw away? I disinfected them. They were still a good pair of shoes.
A few days later, we got an email from the airline with a voucher, expressing their gratitude for our help. That was nice and unexpected.
I responded with a suggestion: How about having some protocols for medical events on airplanes? Pilots go through checklists for almost everything they do. Why wouldn’t they have something like that for medical responses?
I also asked how the man and his wife were doing. But they couldn’t disclose that information.
It was certainly strange being out of my element, helping a patient in that tiny little space; I’m used to working in a recovery room where you have literally everything you need within arm’s reach — the Ambu bag, suction, and bandages. And with airway management, there’s usually more than one person in the room to assist. If there’s a problem, a whole bunch of people show up around the bed so fast.
I’m definitely thinking about field medicine a lot more. Wondering what I would do in certain situations. While debriefing with my mom (an advanced registered nurse practitioner), she pointed out that we should have asked passengers for sanitary pads or diapers to stabilize the bleeding instead of the cocktail napkins. Brilliant idea! I didn’t think of it in the moment. But I’m keeping that little tip tucked in my back pocket for any future bleeding-in-the-wild scenarios.
Audra Podruzny, MSN, RN, CPAN, lives in Washington state and is currently attending the Washington State University Doctor of Nursing Practice Family Nurse Practitioner program.
A version of this article first appeared on Medscape.com.
Data Trends 2024: Arthritis
- US Department of Veterans Affairs. Overview of VA research on arthritis. Accessed March 25, 2024. https://www.research.va.gov/topics/arthritis.cfm
- Fallon EA, Boring MA, Foster AL, Stowe EW, Lites TD, Allen KD. Arthritis prevalence among veterans — United States, 2017–2021. MMWR Recomm Reports. 2023;72(45):1209-1216. doi:10.15585/mmwr.mm7245a1
- Huffman KF, Ambrose KR, Nelson AE, Allen KD, Golightly YM, Callahan LF. The critical role of physical activity and weight management in knee and hip osteoarthritis: a narrative review. J Rheumatol. 2023;51(3):224-233. doi:10.3899/ jrheum.2023-0819
- Lo GH. Successfully treating patients with osteoarthritis: how encouragement of physical activity can generate the best outcomes. A physician’s perspective. J Rheumatol. 2023:jrheum.2023-0899. doi:10.3899/jrheum.2023-0899
- Overton C, Nelson AE, Neogi T. Osteoarthritis treatment guidelines from six professional societies: similarities and differences. Rheum Dis Clin North Am. 2022;48(3):637-657. doi:10.1016/j.rdc.2022.03.009
- US Department of Veterans Affairs. Overview of VA research on arthritis. Accessed March 25, 2024. https://www.research.va.gov/topics/arthritis.cfm
- Fallon EA, Boring MA, Foster AL, Stowe EW, Lites TD, Allen KD. Arthritis prevalence among veterans — United States, 2017–2021. MMWR Recomm Reports. 2023;72(45):1209-1216. doi:10.15585/mmwr.mm7245a1
- Huffman KF, Ambrose KR, Nelson AE, Allen KD, Golightly YM, Callahan LF. The critical role of physical activity and weight management in knee and hip osteoarthritis: a narrative review. J Rheumatol. 2023;51(3):224-233. doi:10.3899/ jrheum.2023-0819
- Lo GH. Successfully treating patients with osteoarthritis: how encouragement of physical activity can generate the best outcomes. A physician’s perspective. J Rheumatol. 2023:jrheum.2023-0899. doi:10.3899/jrheum.2023-0899
- Overton C, Nelson AE, Neogi T. Osteoarthritis treatment guidelines from six professional societies: similarities and differences. Rheum Dis Clin North Am. 2022;48(3):637-657. doi:10.1016/j.rdc.2022.03.009
- US Department of Veterans Affairs. Overview of VA research on arthritis. Accessed March 25, 2024. https://www.research.va.gov/topics/arthritis.cfm
- Fallon EA, Boring MA, Foster AL, Stowe EW, Lites TD, Allen KD. Arthritis prevalence among veterans — United States, 2017–2021. MMWR Recomm Reports. 2023;72(45):1209-1216. doi:10.15585/mmwr.mm7245a1
- Huffman KF, Ambrose KR, Nelson AE, Allen KD, Golightly YM, Callahan LF. The critical role of physical activity and weight management in knee and hip osteoarthritis: a narrative review. J Rheumatol. 2023;51(3):224-233. doi:10.3899/ jrheum.2023-0819
- Lo GH. Successfully treating patients with osteoarthritis: how encouragement of physical activity can generate the best outcomes. A physician’s perspective. J Rheumatol. 2023:jrheum.2023-0899. doi:10.3899/jrheum.2023-0899
- Overton C, Nelson AE, Neogi T. Osteoarthritis treatment guidelines from six professional societies: similarities and differences. Rheum Dis Clin North Am. 2022;48(3):637-657. doi:10.1016/j.rdc.2022.03.009