User login
Pediatric Hospitalization Epidemiology
Improvement in the quality of hospital care in the United States is a national priority, both to advance patient safety and because our expenditures exceed any other nation's, but our health outcomes lag behind.[1, 2] Healthcare spending for children is growing at a faster rate than any other age group, with hospital care accounting for more than 40% of pediatric healthcare expenditures.[3] Inpatient healthcare comprises a greater proportion of healthcare costs for children than for adults, yet we have limited knowledge about where this care is provided.[4]
There is substantial variability in the settings in which children are hospitalized. Children may be hospitalized in freestanding children's hospitals, where all services are designed for children and which operate independently of adult‐focused institutions. They may also be hospitalized in general hospitals where care may be provided in a general inpatient bed, on a dedicated pediatric ward, or in a children's hospital nested within a hospital, which may have specialized nursing and physician care but often shares other resources such as laboratory and radiology with the primarily adult‐focused institution. Medical students and residents may be trained in all of these settings. We know little about how these hospital types differ with respect to patient populations, disease volumes, and resource utilization, and this knowledge is important to inform clinical programs, implementation research, and quality improvement (QI) priorities. To this end, we aimed to describe the volume and characteristics of pediatric hospitalizations at acute care general hospitals and freestanding children's hospitals in the United States.
METHODS
Study Design and Eligibility
The data source for this analysis was the Healthcare Cost and Utilization Project's (HCUP) 2012 Kids' Inpatient Database (KID). We conducted a cross‐sectional study of hospitalizations in children and adolescents less than 18 years of age, excluding in‐hospital births and hospitalizations for pregnancy and delivery (identified using All Patient Refined‐Diagnostic Related Groups [APR‐DRGs]).[5] Neonatal hospitalizations not representing in‐hospital births but resulting from transfers or new admissions were retained. Because the dataset does not contain identifiable information, the institutional review board at Baystate Medical Center determined that our study did not constitute human subjects research.
The KID is released every 3 years and is the only publicly available, nationally representative database developed to study pediatric hospitalizations, including an 80% sample of noninborn pediatric discharges from all community, nonrehabilitation hospitals from 44 participating states.[6] Short‐term rehabilitation hospitals, long‐term nonacute care hospitals, psychiatric hospitals, and alcoholism/chemical dependency treatment facilities are excluded. The KID contains information on all patients, regardless of payer, and provides discharge weights to calculate national estimates.[6] It contains both hospital‐level and patient‐level variables, including demographic characteristics, charges, and other clinical and resource use data available from discharge abstracts. Beginning in 2012, freestanding children's hospitals (FCHs) are assigned to a separate stratum in the KID, with data from the Children's Hospital Association used by HCUP to verify the American Hospital Association's (AHA) list of FCHs.[6] Hospitals that are not FCHs were categorized as general hospitals (GHs). We were interested in examining patterns of care at acute care hospitals and not specialty hospitals; unlike previous years, the KID 2012 does not include a specialty hospital identifier.[6] Therefore, as a proxy for specialty hospital status, we excluded hospitals that had 2% hospitalizations for 12 common medical conditions (pneumonia, asthma, bronchiolitis, cellulitis, dehydration, urinary tract infection, neonatal hyperbilirubinemia, fever, upper respiratory infection, infectious gastroenteritis, unspecified viral infection, and croup). These medical conditions were the 12 most common reasons for medical hospitalizations identified using Keren's pediatric diagnosis code grouper,[7] excluding chronic diseases, and represented 26.2% of all pediatric hospitalizations. This 2% threshold was developed empirically, based on visual analysis of the distribution of cases across hospitals and was limited to hospitals with total pediatric volumes >25/year, allowing for stable case‐mix estimates.
Descriptor Variables
Hospital level characteristics included US Census region; teaching status classified in the KID based on results of the AHA Annual Survey; urban/rural location; hospital ownership, classified as public, private nonprofit and private investor‐owned; and total volume of pediatric hospitalizations, in deciles.[6] At the patient level, we examined age, gender, race/ethnicity, expected primary payer, and median household income (in quartiles) for patient's zip code. Medical complexity was categorized as (1) nonchronic disease, (2) complex chronic disease, or (3) noncomplex chronic disease, using the previously validated Pediatric Medical Complexity Algorithm (PMCA) based on International Classification of Diseases, 9th Revision, Clinical Modification (ICD‐9‐CM) codes.[8] Disease severity was classified based on APR‐DRG severity of illness coding, which classifies illnesses severity as minor, moderate, major, or extreme.[9]
We examined the following characteristics of the hospitalizations: (1) length of hospital stay (LOS) measured in calendar days; (2) high‐turnover hospitalization defined as LOS less than 2 days[10, 11, 12]; (3) long LOS, defined as greater than 4 days, equivalent to LOS greater than the 75th percentile; (4) neonatal versus non‐neonatal hospitalization, identified using APR‐DRGs; (5) admission type categorized as elective and nonelective; (6) admission source, categorized as transfer from another acute care hospital, admission from the emergency department, or direct admission; (7) discharge status, categorized as routine discharge, transfer to another hospital or healthcare facility, and discharge against medical advice; and (8) total hospital costs, calculated by applying the cost‐to‐charge ratios available in the KID to total hospital charges.
Reasons for hospitalization were categorized using the pediatric diagnosis code grouper by Keren, which uses ICD‐9‐CM codes to group common and costly principal diagnoses into distinct conditions (eg, pneumonia, idiopathic scoliosis), excluding children who have ICD‐9‐CM principal procedure codes unlikely related to their principal diagnosis (for example, appendectomy for a child with a principal diagnosis of pneumonia).[7] This pediatric grouper classifies diagnoses as medical, surgical, or medical‐surgical based on whether 20% (medical), >80% (surgical) or between 20% and 80% (medical‐surgical) of encounters for the condition had an ICD‐9‐CM principal procedure code for a surgery related to that condition. We further characterized medical hospitalizations as either medical or mental health hospitalizations.
Statistical Analysis
We categorized each discharge record as a hospitalization at a GH or an FCH. We then calculated patient‐level summary statistics, applying weights to calculate national estimates with an associated standard deviation (SD). We assessed differences in characteristics of hospitalizations at GHs and FCHs using Rao‐Scott 2 tests for categorical variables and Wald F tests for continuous variables.[6] We identified the most common reasons for hospitalization, including those responsible for at least 2% of all medical or surgical hospitalizations and at least 0.5% of medical hospitalizations for mental health diagnoses, given the lower prevalence of these conditions and our desire to include mental health diagnoses in our analysis. For these common conditions, we calculated the proportion of condition‐specific hospitalizations and aggregate hospital costs at GHs and FCHs. We also determined the number of hospitalizations at each hospital and calculated the median and interquartile range for the number of hospitalizations for each of these conditions according to hospital type, assessing for differences using Kruskal‐Wallis tests. Finally, we identified the most common and costly conditions at GHs and FCHs by ranking frequency and aggregate costs for each condition according to hospital type, limited to the 20 most costly and/or prevalent pediatric diagnoses. Because we used a novel method to identify specialty hospitals in this dataset, we repeated these analyses using all hospitals classified as a GH and FCH as a sensitivity analysis.
RESULTS
Overall, 3866 hospitals were categorized as a GH, whereas 70 hospitals were categorized as FCHs. Following exclusion of specialty hospitals, 3758 GHs and 50 FCHs were retained in this study. The geographic distribution of hospitals was similar, but although GHs included those in both urban and rural regions, all FCHs were located in urban regions (Table 1).
| General Hospitals, n = 3,758 | Children's Hospitals, n = 50 | ||||
|---|---|---|---|---|---|
| Hospital characteristics | n | % | n | % | P Value |
| |||||
| Geographic region | |||||
| Northeast | 458 | 12.2 | 4 | 8.0 | 0.50 |
| Midwest | 1,209 | 32.2 | 15 | 30.0 | |
| South | 1,335 | 35.6 | 17 | 34.0 | |
| West | 753 | 20.1 | 14 | 28.0 | |
| Location and teaching status | |||||
| Rural | 1,524 | 40.6 | 0 | 0 | 0.0001 |
| Urban nonteaching | 1,506 | 40.1 | 7 | 14.0 | |
| Urban teaching | 725 | 19.3 | 43 | 86.0 | |
| Hospital ownership | |||||
| Government, nonfederal | 741 | 19.7 | 0 | 0 | 0.0001 |
| Private, nonprofit | 2,364 | 63.0 | 48 | 96.0 | |
| Private, investor‐owned | 650 | 17.3 | 2 | 4.0 | |
| Volume of pediatric hospitalizations (deciles) | |||||
| 185 hospitalizations/year (8th decile) | 2,664 | 71.0 | 0 | 0 | 0.0001 |
| 186375 hospitalizations/year (8th decile) | 378 | 10.1 | 2 | 4.0 | |
| 376996 hospitalizations/year (9th decile) | 380 | 10.1 | 1 | 2.0 | |
| >986 hospitalizations/year (10th decile) | 333 | 8.9 | 47 | 94.0 | |
| Volume of pediatric hospitalizations, median [IQR] | 56 | [14240] | 12,001 | [5,83815,448] | 0.0001 |
A total of 1,407,822 (SD 50,456) hospitalizations occurred at GHs, representing 71.7% of pediatric hospitalizations, whereas 554,458 (SD 45,046) hospitalizations occurred at FCHs. Hospitalizations at GHs accounted for 63.6% of days in hospital and 50.0% of pediatric inpatient healthcare costs. Eighty percent of the GHs had total pediatric patient volumes of less than 375 hospitalizations yearly; 11.1% of pediatric hospitalizations occurred at these lower‐volume centers. At FCHs, the median volume of pediatric hospitalizations was 12,001 (interquartile range [IQR]: 583815,448). A total of 36 GHs had pediatric hospitalization volumes in this IQR.
The median age for pediatric patients was slightly higher at GHs, whereas gender, race/ethnicity, primary payer, and median household income for zip code did not differ significantly between hospital types (Table 2). Medical complexity differed between hospital types: children with complex chronic diseases represented 20.2% of hospitalizations at GHs and 35.6% of hospitalizations at FCHs. Severity of illness differed between hospital types, with fewer hospitalizations categorized at the highest level of severity at GHs than FCHs. There were no significant differences between hospital types with respect to the proportion of hospitalizations categorized as neonatal hospitalizations or as elective hospitalizations. The median LOS was shorter at GHs than FCHs. Approximately 1 in 5 children hospitalized at GHs had LOS greater than 4 days, whereas almost 30% of children hospitalized at FCHs had LOS of this duration.
| Patient Characteristics |
General Hospitals,1,407,822 (50,456), 71.7% |
Children's Hospitals,554,458 (45,046), 28.3% |
P Value | ||||
|---|---|---|---|---|---|---|---|
| n | (SD Weighted Frequency) | (%) | n | (SD Weighted Frequency) | % | ||
| |||||||
| Age, y, median [IQR] | 3.6 [011.7] | 3.4 [010.8] | 0.001 | ||||
| Gender (% female) | 644,250 | (23,089) | 45.8 | 254,505 | (20,688) | 45.9 | 0.50 |
| Race* | |||||||
| White | 668,876 | (27,741) | 47.5 | 233,930 | (26,349) | 42.2 | 0.05 |
| Black | 231,586 | (12,890) | 16.5 | 80,568 | (11,739) | 14.5 | |
| Hispanic | 279,021 | (16,843) | 19.8 | 12,1425 | (21,183) | 21.9 | |
| Other | 133,062 | (8,572) | 9.5 | 41,190 | (6,394) | 7.4 | |
| Insurance status | |||||||
| Public | 740,033 | (28,675) | 52.6 | 284,795 | (25,324) | 51.4 | 0.90 |
| Private | 563,562 | (21,930) | 40.0 | 224,042 | (21,613) | 40.4 | |
| Uninsured | 37,265 | (1,445) | 2.7 | 16,355 | (3,804) | 3.0 | |
| No charge/other/unknown | 66,962 | (5,807) | 4.8 | 29,266 | (6,789) | 5.3 | |
| Median household income for zip code, quartiles | |||||||
| $38,999 | 457,139 | (19,725) | 33.3 | 164,831 | (17,016) | 30.1 | 0.07 |
| $39,000$47,999 | 347,229 | (14,104) | 25.3 | 125,105 | (10,712) | 22.9 | |
| $48,000$62,999 | 304,795 | (13,427) | 22.2 | 134,915 | (13,999) | 24.7 | |
| >$63,000 | 263,171 | (15,418) | 19.2 | 122,164 | (16,279) | 22.3 | |
| Medical complexity | |||||||
| Nonchronic disease | 717,009 | (21,807) | 50.9 | 211,089 | (17,023) | 38.1 | 0.001 |
| Noncomplex chronic disease | 406,070 | (14,951) | 28.8 | 146,077 | (12,442) | 26.4 | |
| Complex chronic disease | 284,742 | (17,111) | 20.2 | 197,292 | (18,236) | 35.6 | |
| APR‐DRG severity of illness | |||||||
| 1 (lowest severity) | 730,134 | (23,162) | 51.9 | 217,202 | (18,433) | 39.2 | 0.001 |
| 2 | 486,748 | (18,395) | 34.6 | 202,931 | (16,864) | 36.6 | |
| 3 | 146,921 | (8,432) | 10.4 | 100,566 | (9,041) | 18.1 | |
| 4 (highest severity) | 41,749 | (3,002) | 3.0 | 33,340 | (3,199) | 6.0 | |
| Hospitalization characteristics | |||||||
| Neonatal hospitalization | 98,512 | (3,336) | 7.0 | 39,584 | (4,274) | 7.1 | 0.84 |
| Admission type | |||||||
| Elective | 255,774 | (12,285) | 18.3 | 109,854 | (13,061) | 19.8 | 0.05 |
| Length of stay, d, (median [IQR]) | 1.8 (0.01) [0.8‐3.6] | 2.2 (0.06) [1.1‐4.7] | 0.001 | ||||
| High turnover hospitalizations | 416,790 | (14,995) | 29.6 | 130,441 | (12,405) | 23.5 | 0.001 |
| Length of stay >4 days | 298,315 | (14,421) | 21.2 | 161,804 | (14,354) | 29.2 | 0.001 |
| Admission source | |||||||
| Transfer from another acute care hospital | 154,058 | (10,067) | 10.9 | 82,118 | (8,952) | 14.8 | 0.05 |
| Direct admission | 550,123 | (21,954) | 39.1 | 211,117 | (20,203) | 38.1 | |
| Admission from ED | 703,641 | (26,155) | 50.0 | 261,223 | (28,708) | 47.1 | |
| Discharge status | |||||||
| Routine | 1,296,638 | (46,012) | 92.1 | 519,785 | (42,613) | 93.8 | 0.01 |
| Transfer to another hospital or healthcare facility | 56,115 | (1,922) | 4.0 | 13,035 | (1,437) | 2.4 | |
| Discharge against medical advice | 2,792 | (181) | 0.2 | 382 | (70) | 0.1 | |
| Other | 52,276 | (4,223) | 3.7 | 21,256 | (4,501) | 3.8 | |
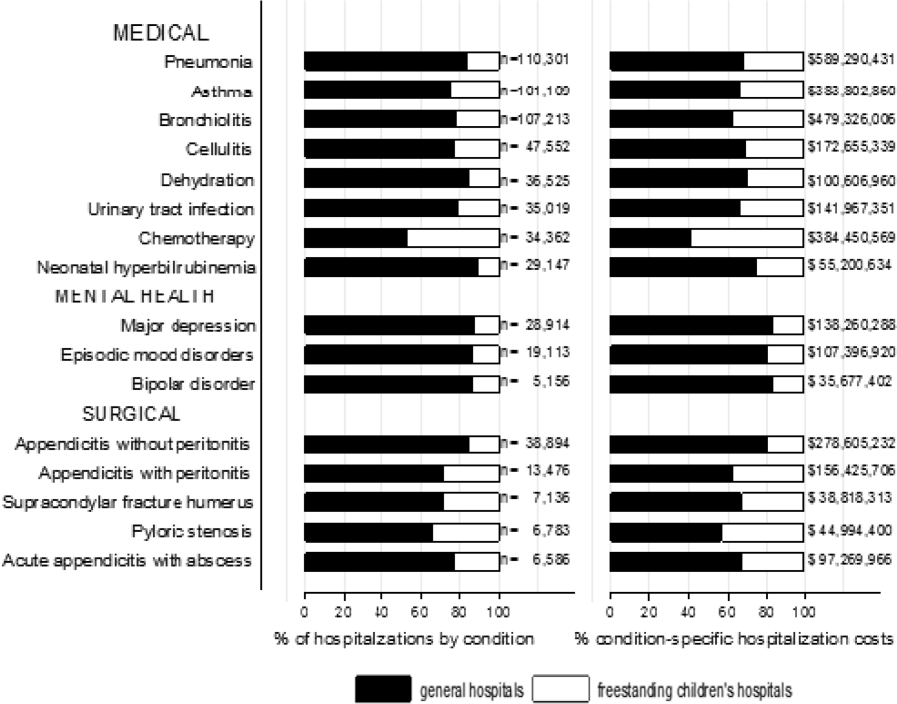
The most common pediatric medical, mental health, and surgical conditions are shown in Figure 1, together representing 32% of pediatric hospitalizations during the study period. For these medical conditions, 77.9% of hospitalizations occurred at GHs, ranging from 52.6% of chemotherapy hospitalizations to 89.0% of hospitalizations for neonatal hyperbilirubinemia. Sixty‐two percent of total hospital costs for these conditions were incurred at GHs. For the common mental health hospitalizations, 86% of hospitalizations occurred at GHs. The majority of hospitalizations and aggregate hospital costs for common surgical conditions also occurred at GHs.
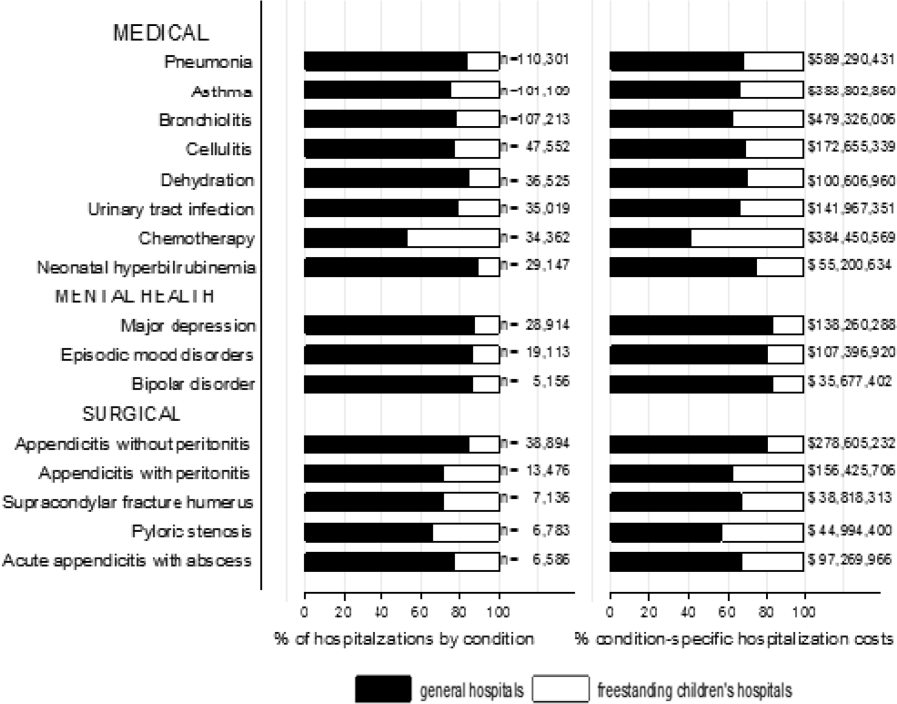
Whereas pneumonia, asthma, and bronchiolitis were the most common reasons for hospitalization at both GHs and FCHs, the most costly conditions differed (see Supporting Table 1 in the online version of this article). At GHs, these respiratory diseases were responsible for the highest condition‐specific total hospital costs. At FCHs, the highest aggregate costs were due to respiratory distress syndrome and chemotherapy. Congenital heart diseases, including hypoplastic left heart syndrome, transposition of the great vessels, tetralogy of Fallot, endocardial cushion defects, coarctation of the aorta and ventricular septal defects accounted for 6 of the 20 most costly conditions at FCHs.
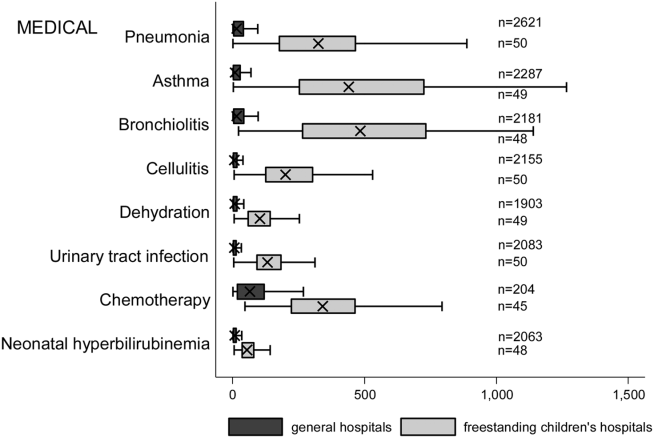
Figure 2 illustrates the volume of hospitalizations, per hospital, at GHs and FCHs for the most common medical hospitalizations. The median number of hospitalizations, per hospital, was consistently significantly lower at GHs than at FCHs (all P values 0.001). Similar results for surgical and mental health hospitalizations are shown as Supporting Figures 1 and 2 in the online version of this article. In our sensitivity analyses that included all hospitals classified as GH and FCH, all results were essentially unchanged.
Recognizing the wide range of pediatric volumes at GHs (Table 1) and our inability to differentiate children's hospitals nested within GHs from GHs with pediatric beds, we examined differences in patient and hospitalization characteristics at GHs with volumes 5838 hospitalizations (the 25th percentile for FCH volume) and GHs with pediatric volumes 5838/year (see Supporting Table 2 in the online version of this article). We also compared patient and hospitalization characteristics at FCHs and the higher‐volume GHs. A total of 36 GHs had pediatric volumes 5838, with hospitalizations at these sites together accounting for 15.4% of all pediatric hospitalizations. Characteristics of patients hospitalized at these higher‐volume GHs were similar to patients hospitalized at FCHs, but they had significantly lower disease severity, fewer neonatal hospitalizations, shorter LOS, and more high‐turnover hospitalizations than patients hospitalized at FCHs. We also observed several differences between children hospitalized at higher‐ and lower‐volume GHs (see Supporting Table 2 in the online version of this article). Children hospitalized at the lower‐volume GHs were more likely to have public health insurance and less likely to have complex chronic diseases, although overall, 39.0% of all hospitalizations for children with complex chronic diseases occurred at these lower‐volume GHs. Compared to children hospitalized at higher‐volume GHs, children hospitalized at the lower‐volume hospitals had significantly lower disease severity, shorter LOS, more direct admissions, and a greater proportion of routine discharges.
DISCUSSION
Of the 2 million pediatric hospitalizations in the United States in 2012, more than 70% occurred at GHs. We observed considerable heterogeneity in pediatric volumes across GHs, with 11% of pediatric hospitalizations occurring at hospitals with pediatric volumes of 375 hospitalizations annually, whereas 15% of pediatric hospitalizations occurred at GHs with volumes similar to those observed at FCHs. The remaining pediatric hospitalizations at GHs occurred at centers with intermediate volumes. The most common reasons for hospitalization were similar at GHs and FCHs, but the most costly conditions differed substantially. These findings have important implications for pediatric clinical care programs, research, and QI efforts.
Our finding that more than 70% of pediatric hospitalizations occurred at GHs speaks to the importance of quality measurement at these hospitals, whereas low per‐hospital pediatric volumes at the majority of GHs makes such measurement particularly challenging. Several previous studies have illustrated that volumes of pediatric hospitalizations are too small to detect meaningful differences in quality between hospitals using established condition‐specific metrics.[13, 14, 15] Our finding that more than 10% of pediatric hospitalizations occurred at GHs with pediatric volumes 375 year supports previous research suggesting that cross‐cutting, all‐condition quality metrics, composite measures, and/or multihospital reporting networks may be needed to enable quality measurement at these sites. In addition, the heterogeneity in patient volumes and characteristics across GHs raise questions about the applicability of quality metrics developed and validated at FCHs to the many GH settings. Field‐testing quality measures to ensure their validity at diverse GHs, particularly those with patient volumes and infrastructure different from FCHs, will be important to meaningful pediatric quality measurement.
Our results illustrating differences in the most common and costly conditions at GHs and FCHs have further implications for prioritization and implementation of research and QI efforts. Implementation research and QI efforts focused on cardiac and neurosurgical procedures, as well as neonatal intensive care, may have considerable impact on cost and quality at FCHs. At GHs, research and QI efforts focused on common conditions are needed to increase our knowledge of contextually relevant barriers to and facilitators of high‐quality pediatric care. This, however, can be made more difficult by small sample sizes, limited resources, and infrastructure, and competing priorities in adult‐focused GH settings.[16, 17, 18] Multihospital learning collaboratives and partnerships between FCHs and GHs can begin to address these challenges, but their success is contingent upon national advocacy and funding to support pediatric research and quality measures at GHs.
One of the most notable differences in the characteristics of pediatric hospitalizations at GHs and FCHs was the proportion of hospitalizations attributable to children with medical complexity (CMC); more than one‐third of hospitalizations at FCHs were for CMC compared to 1 in 5 at GHs. These findings align with the results of several previous studies describing the substantial resource utilization attributed to CMC, and with growing research, innovation, and quality metrics focused on improving both inpatient and outpatient care for these vulnerable children.[19, 20, 21, 22] Structured complex care programs, developed to improve care coordination and healthcare quality for CMC, are common at FCHs, and have been associated with decreased resource utilization and improved outcomes.[23, 24, 25] Notably, however, more than half of all hospitalizations for CMC, exceeding 250,000 annually, occurred at GHs, and almost 40% of hospitalizations for CMC occurred at the lower‐volume GHs. These findings speak to the importance of translating effective and innovative programs of care for CMC to GHs as resources allow, accompanied by robust evaluations of their effectiveness. Lower patient volume at most GHs, however, may be a barrier to dedicated CMC programs. As a result, decentralized community‐based programs of care for CMC, linking primary care programs with regional and tertiary care hospitals, warrant further consideration.[26, 27, 28]
This analysis should be interpreted in light of several limitations. First, we were unable to distinguish between GHs with scant pediatric‐specific resources from those with a large volume of dedicated pediatric resources, such as children's hospitals nested within GHs. We did identify 36 GHs with pediatric volumes similar to those observed at FCHs (see Supporting Table 2 in the online version of this article); patient and hospitalization characteristics at these higher‐volume GHs were similar in many ways to children hospitalized at FCHs. Several of these higher‐volume GHs may have considerable resources dedicated to the care of children, including subspecialty care, and may represent children's hospitals nested within GHs. Because nested children's hospitals are included in the GH categorization, our results may have underestimated the proportion of children cared for at children's hospitals. Further work is needed to identify the health systems challenges and opportunities that may be unique to these institutions. Second, because the 2012 KID does not include a specialty hospital indicator, we developed a proxy method for identifying these hospitals, which may have resulted in some misclassification. We are reassured that the results of our analyses did not change substantively when we included all hospitals. Similarly, although we are reassured that the number of hospitals classified in our analysis as acute care FCHs aligns, approximately, with the number of hospitals classified as such by the Children's Hospital Association, we were unable to assess the validity of this variable within the KID. Third, the KID does not link records at the patient level, so we are unable to report the number of unique children included in this analysis. In addition, the KID includes only inpatient stays with exclusion of observation status stays; potential differences between GH and FCH in the use of observation status could have biased our findings. Fifth, we used the PMCA to identify CMC; although this algorithm has been shown to have excellent sensitivity in identifying children with chronic diseases, using up to 3 years of Medicaid claims data, the sensitivity using the KID, where only 1 inpatient stay is available for assessment, is unknown.[8, 29] Similarly, use of Keren's pediatric diagnosis grouper to classify reasons for hospitalization may have resulted in misclassification, though there are few other nonproprietary pediatric‐specific diagnostic groupers available.
In 2012, more than 70% of pediatric hospitalizations occurred at GHs in the United States. The considerably higher pediatric volumes at FCHs makes these institutions well suited for research, innovation, and the development and application of disease‐specific QI initiatives. Recognizing that the majority of pediatric hospitalizations occurred at GHs, there is a clear need for implementation research, program development, and quality metrics that align with the characteristics of hospitalizations at these centers. National support for research and quality improvement that reflects the diverse hospital settings where children receive their hospital care is critical to further our nation's goal of improving hospital quality for children.
Disclosures
Dr. Leyenaar was supported by grant number K08HS024133 from the Agency for Healthcare Research and Quality. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality. The authors have no conflicts of interest relevant to this article to disclose.
- , , , . Mirror, Mirror on the wall: how the performance of the US health care system compares internationally. The Commonwealth Fund. Available at: http://www.commonwealthfund.org/publications/fund‐reports/2014/jun/mirror‐mirror. Published June 16, 2014. Accessed August 26, 2015.
- , , , , , . Higher cost, but poorer outcomes: the US health disadvantage and implications for pediatrics. Pediatrics. 2015;135(6):961–964.
- , , , , . US health spending trends by age and gender: selected years 2002–10. Health Aff (Millwood). 2014;33(5):815–822.
- , , . Costs for hospital stays in the United States, 2012. Healthcare Cost and Utilization Project 181. Available at: http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb181‐Hospital‐Costs‐United‐States‐2012.pdf. Published October 2014. Accessed September 2015.
- , , , et al. All Patient Refined Diagnosis Related Groups: Methodology Overview. 3M Health Information Systems. Available at: https://www.hcup‐us.ahrq.gov/db/nation/nis/APR‐DRGsV20MethodologyOverviewandBibliography.pdf. Accessed February 8, 2016.
- Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project. Introduction to the HCUP Kids' Inpatient Database (KID) 2012. Available at: https://www.hcup‐us.ahrq.gov/db/nation/kid/kid_2012_introduction.jsp. Published Issued July 2014. Accessed February 8, 2016.
- . Prioritization of comparative effectiveness research topics in hospital pediatrics. Arch Pediatr Adolesc Med. 2012;166(12):1155.
- , , , et al. Pediatric medical complexity algorithm: a new method to stratify children by medical complexity. Pediatrics. 2014;133(6):e1647–e1654.
- , , , et al. 3M APR DRG Classification System. 3M Health Information Systems. Available at: http://www.hcup‐us.ahrq.gov/db/nation/nis/v261_aprdrg_meth_ovrview.pdf. Accessed August 7, 2015.
- , , , , , . Trends in high‐turnover stays among children hospitalized in the United States, 1993–2003. Pediatrics. 2009;123(3):996–1002.
- , , , . High turnover stays for pediatric asthma in the United States. Med Care. 2010;48(9):827–833.
- , , , , . Variation and outcomes associated with direct admission among children with pneumonia in the United States. JAMA Pediatr. 2014;168(9):829–836.
- , , , et al. Recognizing differences in hospital quality performance for pediatric inpatient care. Pediatrics. 2015;136(2):251–262.
- , , . Small numbers limit the use of the inpatient pediatric quality indicators for hospital comparison. Acad Pediatr. 2010;10(4):266–273.
- , , , et al. Statistical uncertainty of mortality rates and rankings for children's hospitals. Pediatrics. 2011;128(4):e966–e972.
- , , , , . Determinants of career satisfaction among pediatric hospitalists: a qualitative exploration. Acad Pediatr. 2014;14(4):361–368.
- , , , et al. Quality improvement research in pediatric hospital medicine and the role of the Pediatric Research in Inpatient Settings (PRIS) network. Acad Pediatr. 2013;13(6 suppl):S54–S60.
- . Roles for children's hospitals in pediatric collaborative improvement networks. Pediatrics. 2013;131(suppl 4):S215–S218.
- , , , et al. Children with medical complexity: an emerging population for clinical and research initiatives. Pediatrics. 2011;127(3):529–538.
- , , , et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647–655.
- , , , , , . Patterns and costs of health care use of children with medical complexity. Pediatrics. 2012;130(6):e1463–e1470.
- , , , , , . Hospital utilization and characteristics of patients experiencing recurrent readmissions within children's hospitals. JAMA. 2011;305(7):682–690.
- , , , et al. Characteristics of hospitalizations for patients who use a structured clinical care program for children with medical complexity. J Pediatr. 2011;159(2):284–290.
- , , , . Hospital‐based comprehensive care programs for children with special health care needs: a systematic review. Arch Pediatr Adolesc Med. 2011;165(6):554–561.
- , , , , , . A tertiary care–primary care partnership model for medically complex and fragile children and youth with special health care needs. Arch Pediatr Adolesc Med. 2007;161(10):937–944.
- , , , et al. Integrated complex care coordination for children with medical complexity: a mixed‐methods evaluation of tertiary care‐community collaboration. BMC Health Serv Res. 2012;12:366.
- , , , . Medical transport of children with complex chronic conditions. Emerg Med Int. 2012;2012:837020.
- , , . Comanagement of medically complex children by subspecialists, generalists, and care coordinators. Pediatrics. 2014;134(2):203–205.
- , , , , . Ways to identify children with medical complexity and the importance of why. J Pediatr. 2015;167(2):229–237.
Improvement in the quality of hospital care in the United States is a national priority, both to advance patient safety and because our expenditures exceed any other nation's, but our health outcomes lag behind.[1, 2] Healthcare spending for children is growing at a faster rate than any other age group, with hospital care accounting for more than 40% of pediatric healthcare expenditures.[3] Inpatient healthcare comprises a greater proportion of healthcare costs for children than for adults, yet we have limited knowledge about where this care is provided.[4]
There is substantial variability in the settings in which children are hospitalized. Children may be hospitalized in freestanding children's hospitals, where all services are designed for children and which operate independently of adult‐focused institutions. They may also be hospitalized in general hospitals where care may be provided in a general inpatient bed, on a dedicated pediatric ward, or in a children's hospital nested within a hospital, which may have specialized nursing and physician care but often shares other resources such as laboratory and radiology with the primarily adult‐focused institution. Medical students and residents may be trained in all of these settings. We know little about how these hospital types differ with respect to patient populations, disease volumes, and resource utilization, and this knowledge is important to inform clinical programs, implementation research, and quality improvement (QI) priorities. To this end, we aimed to describe the volume and characteristics of pediatric hospitalizations at acute care general hospitals and freestanding children's hospitals in the United States.
METHODS
Study Design and Eligibility
The data source for this analysis was the Healthcare Cost and Utilization Project's (HCUP) 2012 Kids' Inpatient Database (KID). We conducted a cross‐sectional study of hospitalizations in children and adolescents less than 18 years of age, excluding in‐hospital births and hospitalizations for pregnancy and delivery (identified using All Patient Refined‐Diagnostic Related Groups [APR‐DRGs]).[5] Neonatal hospitalizations not representing in‐hospital births but resulting from transfers or new admissions were retained. Because the dataset does not contain identifiable information, the institutional review board at Baystate Medical Center determined that our study did not constitute human subjects research.
The KID is released every 3 years and is the only publicly available, nationally representative database developed to study pediatric hospitalizations, including an 80% sample of noninborn pediatric discharges from all community, nonrehabilitation hospitals from 44 participating states.[6] Short‐term rehabilitation hospitals, long‐term nonacute care hospitals, psychiatric hospitals, and alcoholism/chemical dependency treatment facilities are excluded. The KID contains information on all patients, regardless of payer, and provides discharge weights to calculate national estimates.[6] It contains both hospital‐level and patient‐level variables, including demographic characteristics, charges, and other clinical and resource use data available from discharge abstracts. Beginning in 2012, freestanding children's hospitals (FCHs) are assigned to a separate stratum in the KID, with data from the Children's Hospital Association used by HCUP to verify the American Hospital Association's (AHA) list of FCHs.[6] Hospitals that are not FCHs were categorized as general hospitals (GHs). We were interested in examining patterns of care at acute care hospitals and not specialty hospitals; unlike previous years, the KID 2012 does not include a specialty hospital identifier.[6] Therefore, as a proxy for specialty hospital status, we excluded hospitals that had 2% hospitalizations for 12 common medical conditions (pneumonia, asthma, bronchiolitis, cellulitis, dehydration, urinary tract infection, neonatal hyperbilirubinemia, fever, upper respiratory infection, infectious gastroenteritis, unspecified viral infection, and croup). These medical conditions were the 12 most common reasons for medical hospitalizations identified using Keren's pediatric diagnosis code grouper,[7] excluding chronic diseases, and represented 26.2% of all pediatric hospitalizations. This 2% threshold was developed empirically, based on visual analysis of the distribution of cases across hospitals and was limited to hospitals with total pediatric volumes >25/year, allowing for stable case‐mix estimates.
Descriptor Variables
Hospital level characteristics included US Census region; teaching status classified in the KID based on results of the AHA Annual Survey; urban/rural location; hospital ownership, classified as public, private nonprofit and private investor‐owned; and total volume of pediatric hospitalizations, in deciles.[6] At the patient level, we examined age, gender, race/ethnicity, expected primary payer, and median household income (in quartiles) for patient's zip code. Medical complexity was categorized as (1) nonchronic disease, (2) complex chronic disease, or (3) noncomplex chronic disease, using the previously validated Pediatric Medical Complexity Algorithm (PMCA) based on International Classification of Diseases, 9th Revision, Clinical Modification (ICD‐9‐CM) codes.[8] Disease severity was classified based on APR‐DRG severity of illness coding, which classifies illnesses severity as minor, moderate, major, or extreme.[9]
We examined the following characteristics of the hospitalizations: (1) length of hospital stay (LOS) measured in calendar days; (2) high‐turnover hospitalization defined as LOS less than 2 days[10, 11, 12]; (3) long LOS, defined as greater than 4 days, equivalent to LOS greater than the 75th percentile; (4) neonatal versus non‐neonatal hospitalization, identified using APR‐DRGs; (5) admission type categorized as elective and nonelective; (6) admission source, categorized as transfer from another acute care hospital, admission from the emergency department, or direct admission; (7) discharge status, categorized as routine discharge, transfer to another hospital or healthcare facility, and discharge against medical advice; and (8) total hospital costs, calculated by applying the cost‐to‐charge ratios available in the KID to total hospital charges.
Reasons for hospitalization were categorized using the pediatric diagnosis code grouper by Keren, which uses ICD‐9‐CM codes to group common and costly principal diagnoses into distinct conditions (eg, pneumonia, idiopathic scoliosis), excluding children who have ICD‐9‐CM principal procedure codes unlikely related to their principal diagnosis (for example, appendectomy for a child with a principal diagnosis of pneumonia).[7] This pediatric grouper classifies diagnoses as medical, surgical, or medical‐surgical based on whether 20% (medical), >80% (surgical) or between 20% and 80% (medical‐surgical) of encounters for the condition had an ICD‐9‐CM principal procedure code for a surgery related to that condition. We further characterized medical hospitalizations as either medical or mental health hospitalizations.
Statistical Analysis
We categorized each discharge record as a hospitalization at a GH or an FCH. We then calculated patient‐level summary statistics, applying weights to calculate national estimates with an associated standard deviation (SD). We assessed differences in characteristics of hospitalizations at GHs and FCHs using Rao‐Scott 2 tests for categorical variables and Wald F tests for continuous variables.[6] We identified the most common reasons for hospitalization, including those responsible for at least 2% of all medical or surgical hospitalizations and at least 0.5% of medical hospitalizations for mental health diagnoses, given the lower prevalence of these conditions and our desire to include mental health diagnoses in our analysis. For these common conditions, we calculated the proportion of condition‐specific hospitalizations and aggregate hospital costs at GHs and FCHs. We also determined the number of hospitalizations at each hospital and calculated the median and interquartile range for the number of hospitalizations for each of these conditions according to hospital type, assessing for differences using Kruskal‐Wallis tests. Finally, we identified the most common and costly conditions at GHs and FCHs by ranking frequency and aggregate costs for each condition according to hospital type, limited to the 20 most costly and/or prevalent pediatric diagnoses. Because we used a novel method to identify specialty hospitals in this dataset, we repeated these analyses using all hospitals classified as a GH and FCH as a sensitivity analysis.
RESULTS
Overall, 3866 hospitals were categorized as a GH, whereas 70 hospitals were categorized as FCHs. Following exclusion of specialty hospitals, 3758 GHs and 50 FCHs were retained in this study. The geographic distribution of hospitals was similar, but although GHs included those in both urban and rural regions, all FCHs were located in urban regions (Table 1).
| General Hospitals, n = 3,758 | Children's Hospitals, n = 50 | ||||
|---|---|---|---|---|---|
| Hospital characteristics | n | % | n | % | P Value |
| |||||
| Geographic region | |||||
| Northeast | 458 | 12.2 | 4 | 8.0 | 0.50 |
| Midwest | 1,209 | 32.2 | 15 | 30.0 | |
| South | 1,335 | 35.6 | 17 | 34.0 | |
| West | 753 | 20.1 | 14 | 28.0 | |
| Location and teaching status | |||||
| Rural | 1,524 | 40.6 | 0 | 0 | 0.0001 |
| Urban nonteaching | 1,506 | 40.1 | 7 | 14.0 | |
| Urban teaching | 725 | 19.3 | 43 | 86.0 | |
| Hospital ownership | |||||
| Government, nonfederal | 741 | 19.7 | 0 | 0 | 0.0001 |
| Private, nonprofit | 2,364 | 63.0 | 48 | 96.0 | |
| Private, investor‐owned | 650 | 17.3 | 2 | 4.0 | |
| Volume of pediatric hospitalizations (deciles) | |||||
| 185 hospitalizations/year (8th decile) | 2,664 | 71.0 | 0 | 0 | 0.0001 |
| 186375 hospitalizations/year (8th decile) | 378 | 10.1 | 2 | 4.0 | |
| 376996 hospitalizations/year (9th decile) | 380 | 10.1 | 1 | 2.0 | |
| >986 hospitalizations/year (10th decile) | 333 | 8.9 | 47 | 94.0 | |
| Volume of pediatric hospitalizations, median [IQR] | 56 | [14240] | 12,001 | [5,83815,448] | 0.0001 |
A total of 1,407,822 (SD 50,456) hospitalizations occurred at GHs, representing 71.7% of pediatric hospitalizations, whereas 554,458 (SD 45,046) hospitalizations occurred at FCHs. Hospitalizations at GHs accounted for 63.6% of days in hospital and 50.0% of pediatric inpatient healthcare costs. Eighty percent of the GHs had total pediatric patient volumes of less than 375 hospitalizations yearly; 11.1% of pediatric hospitalizations occurred at these lower‐volume centers. At FCHs, the median volume of pediatric hospitalizations was 12,001 (interquartile range [IQR]: 583815,448). A total of 36 GHs had pediatric hospitalization volumes in this IQR.
The median age for pediatric patients was slightly higher at GHs, whereas gender, race/ethnicity, primary payer, and median household income for zip code did not differ significantly between hospital types (Table 2). Medical complexity differed between hospital types: children with complex chronic diseases represented 20.2% of hospitalizations at GHs and 35.6% of hospitalizations at FCHs. Severity of illness differed between hospital types, with fewer hospitalizations categorized at the highest level of severity at GHs than FCHs. There were no significant differences between hospital types with respect to the proportion of hospitalizations categorized as neonatal hospitalizations or as elective hospitalizations. The median LOS was shorter at GHs than FCHs. Approximately 1 in 5 children hospitalized at GHs had LOS greater than 4 days, whereas almost 30% of children hospitalized at FCHs had LOS of this duration.
| Patient Characteristics |
General Hospitals,1,407,822 (50,456), 71.7% |
Children's Hospitals,554,458 (45,046), 28.3% |
P Value | ||||
|---|---|---|---|---|---|---|---|
| n | (SD Weighted Frequency) | (%) | n | (SD Weighted Frequency) | % | ||
| |||||||
| Age, y, median [IQR] | 3.6 [011.7] | 3.4 [010.8] | 0.001 | ||||
| Gender (% female) | 644,250 | (23,089) | 45.8 | 254,505 | (20,688) | 45.9 | 0.50 |
| Race* | |||||||
| White | 668,876 | (27,741) | 47.5 | 233,930 | (26,349) | 42.2 | 0.05 |
| Black | 231,586 | (12,890) | 16.5 | 80,568 | (11,739) | 14.5 | |
| Hispanic | 279,021 | (16,843) | 19.8 | 12,1425 | (21,183) | 21.9 | |
| Other | 133,062 | (8,572) | 9.5 | 41,190 | (6,394) | 7.4 | |
| Insurance status | |||||||
| Public | 740,033 | (28,675) | 52.6 | 284,795 | (25,324) | 51.4 | 0.90 |
| Private | 563,562 | (21,930) | 40.0 | 224,042 | (21,613) | 40.4 | |
| Uninsured | 37,265 | (1,445) | 2.7 | 16,355 | (3,804) | 3.0 | |
| No charge/other/unknown | 66,962 | (5,807) | 4.8 | 29,266 | (6,789) | 5.3 | |
| Median household income for zip code, quartiles | |||||||
| $38,999 | 457,139 | (19,725) | 33.3 | 164,831 | (17,016) | 30.1 | 0.07 |
| $39,000$47,999 | 347,229 | (14,104) | 25.3 | 125,105 | (10,712) | 22.9 | |
| $48,000$62,999 | 304,795 | (13,427) | 22.2 | 134,915 | (13,999) | 24.7 | |
| >$63,000 | 263,171 | (15,418) | 19.2 | 122,164 | (16,279) | 22.3 | |
| Medical complexity | |||||||
| Nonchronic disease | 717,009 | (21,807) | 50.9 | 211,089 | (17,023) | 38.1 | 0.001 |
| Noncomplex chronic disease | 406,070 | (14,951) | 28.8 | 146,077 | (12,442) | 26.4 | |
| Complex chronic disease | 284,742 | (17,111) | 20.2 | 197,292 | (18,236) | 35.6 | |
| APR‐DRG severity of illness | |||||||
| 1 (lowest severity) | 730,134 | (23,162) | 51.9 | 217,202 | (18,433) | 39.2 | 0.001 |
| 2 | 486,748 | (18,395) | 34.6 | 202,931 | (16,864) | 36.6 | |
| 3 | 146,921 | (8,432) | 10.4 | 100,566 | (9,041) | 18.1 | |
| 4 (highest severity) | 41,749 | (3,002) | 3.0 | 33,340 | (3,199) | 6.0 | |
| Hospitalization characteristics | |||||||
| Neonatal hospitalization | 98,512 | (3,336) | 7.0 | 39,584 | (4,274) | 7.1 | 0.84 |
| Admission type | |||||||
| Elective | 255,774 | (12,285) | 18.3 | 109,854 | (13,061) | 19.8 | 0.05 |
| Length of stay, d, (median [IQR]) | 1.8 (0.01) [0.8‐3.6] | 2.2 (0.06) [1.1‐4.7] | 0.001 | ||||
| High turnover hospitalizations | 416,790 | (14,995) | 29.6 | 130,441 | (12,405) | 23.5 | 0.001 |
| Length of stay >4 days | 298,315 | (14,421) | 21.2 | 161,804 | (14,354) | 29.2 | 0.001 |
| Admission source | |||||||
| Transfer from another acute care hospital | 154,058 | (10,067) | 10.9 | 82,118 | (8,952) | 14.8 | 0.05 |
| Direct admission | 550,123 | (21,954) | 39.1 | 211,117 | (20,203) | 38.1 | |
| Admission from ED | 703,641 | (26,155) | 50.0 | 261,223 | (28,708) | 47.1 | |
| Discharge status | |||||||
| Routine | 1,296,638 | (46,012) | 92.1 | 519,785 | (42,613) | 93.8 | 0.01 |
| Transfer to another hospital or healthcare facility | 56,115 | (1,922) | 4.0 | 13,035 | (1,437) | 2.4 | |
| Discharge against medical advice | 2,792 | (181) | 0.2 | 382 | (70) | 0.1 | |
| Other | 52,276 | (4,223) | 3.7 | 21,256 | (4,501) | 3.8 | |
The most common pediatric medical, mental health, and surgical conditions are shown in Figure 1, together representing 32% of pediatric hospitalizations during the study period. For these medical conditions, 77.9% of hospitalizations occurred at GHs, ranging from 52.6% of chemotherapy hospitalizations to 89.0% of hospitalizations for neonatal hyperbilirubinemia. Sixty‐two percent of total hospital costs for these conditions were incurred at GHs. For the common mental health hospitalizations, 86% of hospitalizations occurred at GHs. The majority of hospitalizations and aggregate hospital costs for common surgical conditions also occurred at GHs.

Whereas pneumonia, asthma, and bronchiolitis were the most common reasons for hospitalization at both GHs and FCHs, the most costly conditions differed (see Supporting Table 1 in the online version of this article). At GHs, these respiratory diseases were responsible for the highest condition‐specific total hospital costs. At FCHs, the highest aggregate costs were due to respiratory distress syndrome and chemotherapy. Congenital heart diseases, including hypoplastic left heart syndrome, transposition of the great vessels, tetralogy of Fallot, endocardial cushion defects, coarctation of the aorta and ventricular septal defects accounted for 6 of the 20 most costly conditions at FCHs.
Figure 2 illustrates the volume of hospitalizations, per hospital, at GHs and FCHs for the most common medical hospitalizations. The median number of hospitalizations, per hospital, was consistently significantly lower at GHs than at FCHs (all P values 0.001). Similar results for surgical and mental health hospitalizations are shown as Supporting Figures 1 and 2 in the online version of this article. In our sensitivity analyses that included all hospitals classified as GH and FCH, all results were essentially unchanged.

Recognizing the wide range of pediatric volumes at GHs (Table 1) and our inability to differentiate children's hospitals nested within GHs from GHs with pediatric beds, we examined differences in patient and hospitalization characteristics at GHs with volumes 5838 hospitalizations (the 25th percentile for FCH volume) and GHs with pediatric volumes 5838/year (see Supporting Table 2 in the online version of this article). We also compared patient and hospitalization characteristics at FCHs and the higher‐volume GHs. A total of 36 GHs had pediatric volumes 5838, with hospitalizations at these sites together accounting for 15.4% of all pediatric hospitalizations. Characteristics of patients hospitalized at these higher‐volume GHs were similar to patients hospitalized at FCHs, but they had significantly lower disease severity, fewer neonatal hospitalizations, shorter LOS, and more high‐turnover hospitalizations than patients hospitalized at FCHs. We also observed several differences between children hospitalized at higher‐ and lower‐volume GHs (see Supporting Table 2 in the online version of this article). Children hospitalized at the lower‐volume GHs were more likely to have public health insurance and less likely to have complex chronic diseases, although overall, 39.0% of all hospitalizations for children with complex chronic diseases occurred at these lower‐volume GHs. Compared to children hospitalized at higher‐volume GHs, children hospitalized at the lower‐volume hospitals had significantly lower disease severity, shorter LOS, more direct admissions, and a greater proportion of routine discharges.
DISCUSSION
Of the 2 million pediatric hospitalizations in the United States in 2012, more than 70% occurred at GHs. We observed considerable heterogeneity in pediatric volumes across GHs, with 11% of pediatric hospitalizations occurring at hospitals with pediatric volumes of 375 hospitalizations annually, whereas 15% of pediatric hospitalizations occurred at GHs with volumes similar to those observed at FCHs. The remaining pediatric hospitalizations at GHs occurred at centers with intermediate volumes. The most common reasons for hospitalization were similar at GHs and FCHs, but the most costly conditions differed substantially. These findings have important implications for pediatric clinical care programs, research, and QI efforts.
Our finding that more than 70% of pediatric hospitalizations occurred at GHs speaks to the importance of quality measurement at these hospitals, whereas low per‐hospital pediatric volumes at the majority of GHs makes such measurement particularly challenging. Several previous studies have illustrated that volumes of pediatric hospitalizations are too small to detect meaningful differences in quality between hospitals using established condition‐specific metrics.[13, 14, 15] Our finding that more than 10% of pediatric hospitalizations occurred at GHs with pediatric volumes 375 year supports previous research suggesting that cross‐cutting, all‐condition quality metrics, composite measures, and/or multihospital reporting networks may be needed to enable quality measurement at these sites. In addition, the heterogeneity in patient volumes and characteristics across GHs raise questions about the applicability of quality metrics developed and validated at FCHs to the many GH settings. Field‐testing quality measures to ensure their validity at diverse GHs, particularly those with patient volumes and infrastructure different from FCHs, will be important to meaningful pediatric quality measurement.
Our results illustrating differences in the most common and costly conditions at GHs and FCHs have further implications for prioritization and implementation of research and QI efforts. Implementation research and QI efforts focused on cardiac and neurosurgical procedures, as well as neonatal intensive care, may have considerable impact on cost and quality at FCHs. At GHs, research and QI efforts focused on common conditions are needed to increase our knowledge of contextually relevant barriers to and facilitators of high‐quality pediatric care. This, however, can be made more difficult by small sample sizes, limited resources, and infrastructure, and competing priorities in adult‐focused GH settings.[16, 17, 18] Multihospital learning collaboratives and partnerships between FCHs and GHs can begin to address these challenges, but their success is contingent upon national advocacy and funding to support pediatric research and quality measures at GHs.
One of the most notable differences in the characteristics of pediatric hospitalizations at GHs and FCHs was the proportion of hospitalizations attributable to children with medical complexity (CMC); more than one‐third of hospitalizations at FCHs were for CMC compared to 1 in 5 at GHs. These findings align with the results of several previous studies describing the substantial resource utilization attributed to CMC, and with growing research, innovation, and quality metrics focused on improving both inpatient and outpatient care for these vulnerable children.[19, 20, 21, 22] Structured complex care programs, developed to improve care coordination and healthcare quality for CMC, are common at FCHs, and have been associated with decreased resource utilization and improved outcomes.[23, 24, 25] Notably, however, more than half of all hospitalizations for CMC, exceeding 250,000 annually, occurred at GHs, and almost 40% of hospitalizations for CMC occurred at the lower‐volume GHs. These findings speak to the importance of translating effective and innovative programs of care for CMC to GHs as resources allow, accompanied by robust evaluations of their effectiveness. Lower patient volume at most GHs, however, may be a barrier to dedicated CMC programs. As a result, decentralized community‐based programs of care for CMC, linking primary care programs with regional and tertiary care hospitals, warrant further consideration.[26, 27, 28]
This analysis should be interpreted in light of several limitations. First, we were unable to distinguish between GHs with scant pediatric‐specific resources from those with a large volume of dedicated pediatric resources, such as children's hospitals nested within GHs. We did identify 36 GHs with pediatric volumes similar to those observed at FCHs (see Supporting Table 2 in the online version of this article); patient and hospitalization characteristics at these higher‐volume GHs were similar in many ways to children hospitalized at FCHs. Several of these higher‐volume GHs may have considerable resources dedicated to the care of children, including subspecialty care, and may represent children's hospitals nested within GHs. Because nested children's hospitals are included in the GH categorization, our results may have underestimated the proportion of children cared for at children's hospitals. Further work is needed to identify the health systems challenges and opportunities that may be unique to these institutions. Second, because the 2012 KID does not include a specialty hospital indicator, we developed a proxy method for identifying these hospitals, which may have resulted in some misclassification. We are reassured that the results of our analyses did not change substantively when we included all hospitals. Similarly, although we are reassured that the number of hospitals classified in our analysis as acute care FCHs aligns, approximately, with the number of hospitals classified as such by the Children's Hospital Association, we were unable to assess the validity of this variable within the KID. Third, the KID does not link records at the patient level, so we are unable to report the number of unique children included in this analysis. In addition, the KID includes only inpatient stays with exclusion of observation status stays; potential differences between GH and FCH in the use of observation status could have biased our findings. Fifth, we used the PMCA to identify CMC; although this algorithm has been shown to have excellent sensitivity in identifying children with chronic diseases, using up to 3 years of Medicaid claims data, the sensitivity using the KID, where only 1 inpatient stay is available for assessment, is unknown.[8, 29] Similarly, use of Keren's pediatric diagnosis grouper to classify reasons for hospitalization may have resulted in misclassification, though there are few other nonproprietary pediatric‐specific diagnostic groupers available.
In 2012, more than 70% of pediatric hospitalizations occurred at GHs in the United States. The considerably higher pediatric volumes at FCHs makes these institutions well suited for research, innovation, and the development and application of disease‐specific QI initiatives. Recognizing that the majority of pediatric hospitalizations occurred at GHs, there is a clear need for implementation research, program development, and quality metrics that align with the characteristics of hospitalizations at these centers. National support for research and quality improvement that reflects the diverse hospital settings where children receive their hospital care is critical to further our nation's goal of improving hospital quality for children.
Disclosures
Dr. Leyenaar was supported by grant number K08HS024133 from the Agency for Healthcare Research and Quality. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality. The authors have no conflicts of interest relevant to this article to disclose.
Improvement in the quality of hospital care in the United States is a national priority, both to advance patient safety and because our expenditures exceed any other nation's, but our health outcomes lag behind.[1, 2] Healthcare spending for children is growing at a faster rate than any other age group, with hospital care accounting for more than 40% of pediatric healthcare expenditures.[3] Inpatient healthcare comprises a greater proportion of healthcare costs for children than for adults, yet we have limited knowledge about where this care is provided.[4]
There is substantial variability in the settings in which children are hospitalized. Children may be hospitalized in freestanding children's hospitals, where all services are designed for children and which operate independently of adult‐focused institutions. They may also be hospitalized in general hospitals where care may be provided in a general inpatient bed, on a dedicated pediatric ward, or in a children's hospital nested within a hospital, which may have specialized nursing and physician care but often shares other resources such as laboratory and radiology with the primarily adult‐focused institution. Medical students and residents may be trained in all of these settings. We know little about how these hospital types differ with respect to patient populations, disease volumes, and resource utilization, and this knowledge is important to inform clinical programs, implementation research, and quality improvement (QI) priorities. To this end, we aimed to describe the volume and characteristics of pediatric hospitalizations at acute care general hospitals and freestanding children's hospitals in the United States.
METHODS
Study Design and Eligibility
The data source for this analysis was the Healthcare Cost and Utilization Project's (HCUP) 2012 Kids' Inpatient Database (KID). We conducted a cross‐sectional study of hospitalizations in children and adolescents less than 18 years of age, excluding in‐hospital births and hospitalizations for pregnancy and delivery (identified using All Patient Refined‐Diagnostic Related Groups [APR‐DRGs]).[5] Neonatal hospitalizations not representing in‐hospital births but resulting from transfers or new admissions were retained. Because the dataset does not contain identifiable information, the institutional review board at Baystate Medical Center determined that our study did not constitute human subjects research.
The KID is released every 3 years and is the only publicly available, nationally representative database developed to study pediatric hospitalizations, including an 80% sample of noninborn pediatric discharges from all community, nonrehabilitation hospitals from 44 participating states.[6] Short‐term rehabilitation hospitals, long‐term nonacute care hospitals, psychiatric hospitals, and alcoholism/chemical dependency treatment facilities are excluded. The KID contains information on all patients, regardless of payer, and provides discharge weights to calculate national estimates.[6] It contains both hospital‐level and patient‐level variables, including demographic characteristics, charges, and other clinical and resource use data available from discharge abstracts. Beginning in 2012, freestanding children's hospitals (FCHs) are assigned to a separate stratum in the KID, with data from the Children's Hospital Association used by HCUP to verify the American Hospital Association's (AHA) list of FCHs.[6] Hospitals that are not FCHs were categorized as general hospitals (GHs). We were interested in examining patterns of care at acute care hospitals and not specialty hospitals; unlike previous years, the KID 2012 does not include a specialty hospital identifier.[6] Therefore, as a proxy for specialty hospital status, we excluded hospitals that had 2% hospitalizations for 12 common medical conditions (pneumonia, asthma, bronchiolitis, cellulitis, dehydration, urinary tract infection, neonatal hyperbilirubinemia, fever, upper respiratory infection, infectious gastroenteritis, unspecified viral infection, and croup). These medical conditions were the 12 most common reasons for medical hospitalizations identified using Keren's pediatric diagnosis code grouper,[7] excluding chronic diseases, and represented 26.2% of all pediatric hospitalizations. This 2% threshold was developed empirically, based on visual analysis of the distribution of cases across hospitals and was limited to hospitals with total pediatric volumes >25/year, allowing for stable case‐mix estimates.
Descriptor Variables
Hospital level characteristics included US Census region; teaching status classified in the KID based on results of the AHA Annual Survey; urban/rural location; hospital ownership, classified as public, private nonprofit and private investor‐owned; and total volume of pediatric hospitalizations, in deciles.[6] At the patient level, we examined age, gender, race/ethnicity, expected primary payer, and median household income (in quartiles) for patient's zip code. Medical complexity was categorized as (1) nonchronic disease, (2) complex chronic disease, or (3) noncomplex chronic disease, using the previously validated Pediatric Medical Complexity Algorithm (PMCA) based on International Classification of Diseases, 9th Revision, Clinical Modification (ICD‐9‐CM) codes.[8] Disease severity was classified based on APR‐DRG severity of illness coding, which classifies illnesses severity as minor, moderate, major, or extreme.[9]
We examined the following characteristics of the hospitalizations: (1) length of hospital stay (LOS) measured in calendar days; (2) high‐turnover hospitalization defined as LOS less than 2 days[10, 11, 12]; (3) long LOS, defined as greater than 4 days, equivalent to LOS greater than the 75th percentile; (4) neonatal versus non‐neonatal hospitalization, identified using APR‐DRGs; (5) admission type categorized as elective and nonelective; (6) admission source, categorized as transfer from another acute care hospital, admission from the emergency department, or direct admission; (7) discharge status, categorized as routine discharge, transfer to another hospital or healthcare facility, and discharge against medical advice; and (8) total hospital costs, calculated by applying the cost‐to‐charge ratios available in the KID to total hospital charges.
Reasons for hospitalization were categorized using the pediatric diagnosis code grouper by Keren, which uses ICD‐9‐CM codes to group common and costly principal diagnoses into distinct conditions (eg, pneumonia, idiopathic scoliosis), excluding children who have ICD‐9‐CM principal procedure codes unlikely related to their principal diagnosis (for example, appendectomy for a child with a principal diagnosis of pneumonia).[7] This pediatric grouper classifies diagnoses as medical, surgical, or medical‐surgical based on whether 20% (medical), >80% (surgical) or between 20% and 80% (medical‐surgical) of encounters for the condition had an ICD‐9‐CM principal procedure code for a surgery related to that condition. We further characterized medical hospitalizations as either medical or mental health hospitalizations.
Statistical Analysis
We categorized each discharge record as a hospitalization at a GH or an FCH. We then calculated patient‐level summary statistics, applying weights to calculate national estimates with an associated standard deviation (SD). We assessed differences in characteristics of hospitalizations at GHs and FCHs using Rao‐Scott 2 tests for categorical variables and Wald F tests for continuous variables.[6] We identified the most common reasons for hospitalization, including those responsible for at least 2% of all medical or surgical hospitalizations and at least 0.5% of medical hospitalizations for mental health diagnoses, given the lower prevalence of these conditions and our desire to include mental health diagnoses in our analysis. For these common conditions, we calculated the proportion of condition‐specific hospitalizations and aggregate hospital costs at GHs and FCHs. We also determined the number of hospitalizations at each hospital and calculated the median and interquartile range for the number of hospitalizations for each of these conditions according to hospital type, assessing for differences using Kruskal‐Wallis tests. Finally, we identified the most common and costly conditions at GHs and FCHs by ranking frequency and aggregate costs for each condition according to hospital type, limited to the 20 most costly and/or prevalent pediatric diagnoses. Because we used a novel method to identify specialty hospitals in this dataset, we repeated these analyses using all hospitals classified as a GH and FCH as a sensitivity analysis.
RESULTS
Overall, 3866 hospitals were categorized as a GH, whereas 70 hospitals were categorized as FCHs. Following exclusion of specialty hospitals, 3758 GHs and 50 FCHs were retained in this study. The geographic distribution of hospitals was similar, but although GHs included those in both urban and rural regions, all FCHs were located in urban regions (Table 1).
| General Hospitals, n = 3,758 | Children's Hospitals, n = 50 | ||||
|---|---|---|---|---|---|
| Hospital characteristics | n | % | n | % | P Value |
| |||||
| Geographic region | |||||
| Northeast | 458 | 12.2 | 4 | 8.0 | 0.50 |
| Midwest | 1,209 | 32.2 | 15 | 30.0 | |
| South | 1,335 | 35.6 | 17 | 34.0 | |
| West | 753 | 20.1 | 14 | 28.0 | |
| Location and teaching status | |||||
| Rural | 1,524 | 40.6 | 0 | 0 | 0.0001 |
| Urban nonteaching | 1,506 | 40.1 | 7 | 14.0 | |
| Urban teaching | 725 | 19.3 | 43 | 86.0 | |
| Hospital ownership | |||||
| Government, nonfederal | 741 | 19.7 | 0 | 0 | 0.0001 |
| Private, nonprofit | 2,364 | 63.0 | 48 | 96.0 | |
| Private, investor‐owned | 650 | 17.3 | 2 | 4.0 | |
| Volume of pediatric hospitalizations (deciles) | |||||
| 185 hospitalizations/year (8th decile) | 2,664 | 71.0 | 0 | 0 | 0.0001 |
| 186375 hospitalizations/year (8th decile) | 378 | 10.1 | 2 | 4.0 | |
| 376996 hospitalizations/year (9th decile) | 380 | 10.1 | 1 | 2.0 | |
| >986 hospitalizations/year (10th decile) | 333 | 8.9 | 47 | 94.0 | |
| Volume of pediatric hospitalizations, median [IQR] | 56 | [14240] | 12,001 | [5,83815,448] | 0.0001 |
A total of 1,407,822 (SD 50,456) hospitalizations occurred at GHs, representing 71.7% of pediatric hospitalizations, whereas 554,458 (SD 45,046) hospitalizations occurred at FCHs. Hospitalizations at GHs accounted for 63.6% of days in hospital and 50.0% of pediatric inpatient healthcare costs. Eighty percent of the GHs had total pediatric patient volumes of less than 375 hospitalizations yearly; 11.1% of pediatric hospitalizations occurred at these lower‐volume centers. At FCHs, the median volume of pediatric hospitalizations was 12,001 (interquartile range [IQR]: 583815,448). A total of 36 GHs had pediatric hospitalization volumes in this IQR.
The median age for pediatric patients was slightly higher at GHs, whereas gender, race/ethnicity, primary payer, and median household income for zip code did not differ significantly between hospital types (Table 2). Medical complexity differed between hospital types: children with complex chronic diseases represented 20.2% of hospitalizations at GHs and 35.6% of hospitalizations at FCHs. Severity of illness differed between hospital types, with fewer hospitalizations categorized at the highest level of severity at GHs than FCHs. There were no significant differences between hospital types with respect to the proportion of hospitalizations categorized as neonatal hospitalizations or as elective hospitalizations. The median LOS was shorter at GHs than FCHs. Approximately 1 in 5 children hospitalized at GHs had LOS greater than 4 days, whereas almost 30% of children hospitalized at FCHs had LOS of this duration.
| Patient Characteristics |
General Hospitals,1,407,822 (50,456), 71.7% |
Children's Hospitals,554,458 (45,046), 28.3% |
P Value | ||||
|---|---|---|---|---|---|---|---|
| n | (SD Weighted Frequency) | (%) | n | (SD Weighted Frequency) | % | ||
| |||||||
| Age, y, median [IQR] | 3.6 [011.7] | 3.4 [010.8] | 0.001 | ||||
| Gender (% female) | 644,250 | (23,089) | 45.8 | 254,505 | (20,688) | 45.9 | 0.50 |
| Race* | |||||||
| White | 668,876 | (27,741) | 47.5 | 233,930 | (26,349) | 42.2 | 0.05 |
| Black | 231,586 | (12,890) | 16.5 | 80,568 | (11,739) | 14.5 | |
| Hispanic | 279,021 | (16,843) | 19.8 | 12,1425 | (21,183) | 21.9 | |
| Other | 133,062 | (8,572) | 9.5 | 41,190 | (6,394) | 7.4 | |
| Insurance status | |||||||
| Public | 740,033 | (28,675) | 52.6 | 284,795 | (25,324) | 51.4 | 0.90 |
| Private | 563,562 | (21,930) | 40.0 | 224,042 | (21,613) | 40.4 | |
| Uninsured | 37,265 | (1,445) | 2.7 | 16,355 | (3,804) | 3.0 | |
| No charge/other/unknown | 66,962 | (5,807) | 4.8 | 29,266 | (6,789) | 5.3 | |
| Median household income for zip code, quartiles | |||||||
| $38,999 | 457,139 | (19,725) | 33.3 | 164,831 | (17,016) | 30.1 | 0.07 |
| $39,000$47,999 | 347,229 | (14,104) | 25.3 | 125,105 | (10,712) | 22.9 | |
| $48,000$62,999 | 304,795 | (13,427) | 22.2 | 134,915 | (13,999) | 24.7 | |
| >$63,000 | 263,171 | (15,418) | 19.2 | 122,164 | (16,279) | 22.3 | |
| Medical complexity | |||||||
| Nonchronic disease | 717,009 | (21,807) | 50.9 | 211,089 | (17,023) | 38.1 | 0.001 |
| Noncomplex chronic disease | 406,070 | (14,951) | 28.8 | 146,077 | (12,442) | 26.4 | |
| Complex chronic disease | 284,742 | (17,111) | 20.2 | 197,292 | (18,236) | 35.6 | |
| APR‐DRG severity of illness | |||||||
| 1 (lowest severity) | 730,134 | (23,162) | 51.9 | 217,202 | (18,433) | 39.2 | 0.001 |
| 2 | 486,748 | (18,395) | 34.6 | 202,931 | (16,864) | 36.6 | |
| 3 | 146,921 | (8,432) | 10.4 | 100,566 | (9,041) | 18.1 | |
| 4 (highest severity) | 41,749 | (3,002) | 3.0 | 33,340 | (3,199) | 6.0 | |
| Hospitalization characteristics | |||||||
| Neonatal hospitalization | 98,512 | (3,336) | 7.0 | 39,584 | (4,274) | 7.1 | 0.84 |
| Admission type | |||||||
| Elective | 255,774 | (12,285) | 18.3 | 109,854 | (13,061) | 19.8 | 0.05 |
| Length of stay, d, (median [IQR]) | 1.8 (0.01) [0.8‐3.6] | 2.2 (0.06) [1.1‐4.7] | 0.001 | ||||
| High turnover hospitalizations | 416,790 | (14,995) | 29.6 | 130,441 | (12,405) | 23.5 | 0.001 |
| Length of stay >4 days | 298,315 | (14,421) | 21.2 | 161,804 | (14,354) | 29.2 | 0.001 |
| Admission source | |||||||
| Transfer from another acute care hospital | 154,058 | (10,067) | 10.9 | 82,118 | (8,952) | 14.8 | 0.05 |
| Direct admission | 550,123 | (21,954) | 39.1 | 211,117 | (20,203) | 38.1 | |
| Admission from ED | 703,641 | (26,155) | 50.0 | 261,223 | (28,708) | 47.1 | |
| Discharge status | |||||||
| Routine | 1,296,638 | (46,012) | 92.1 | 519,785 | (42,613) | 93.8 | 0.01 |
| Transfer to another hospital or healthcare facility | 56,115 | (1,922) | 4.0 | 13,035 | (1,437) | 2.4 | |
| Discharge against medical advice | 2,792 | (181) | 0.2 | 382 | (70) | 0.1 | |
| Other | 52,276 | (4,223) | 3.7 | 21,256 | (4,501) | 3.8 | |
The most common pediatric medical, mental health, and surgical conditions are shown in Figure 1, together representing 32% of pediatric hospitalizations during the study period. For these medical conditions, 77.9% of hospitalizations occurred at GHs, ranging from 52.6% of chemotherapy hospitalizations to 89.0% of hospitalizations for neonatal hyperbilirubinemia. Sixty‐two percent of total hospital costs for these conditions were incurred at GHs. For the common mental health hospitalizations, 86% of hospitalizations occurred at GHs. The majority of hospitalizations and aggregate hospital costs for common surgical conditions also occurred at GHs.

Whereas pneumonia, asthma, and bronchiolitis were the most common reasons for hospitalization at both GHs and FCHs, the most costly conditions differed (see Supporting Table 1 in the online version of this article). At GHs, these respiratory diseases were responsible for the highest condition‐specific total hospital costs. At FCHs, the highest aggregate costs were due to respiratory distress syndrome and chemotherapy. Congenital heart diseases, including hypoplastic left heart syndrome, transposition of the great vessels, tetralogy of Fallot, endocardial cushion defects, coarctation of the aorta and ventricular septal defects accounted for 6 of the 20 most costly conditions at FCHs.
Figure 2 illustrates the volume of hospitalizations, per hospital, at GHs and FCHs for the most common medical hospitalizations. The median number of hospitalizations, per hospital, was consistently significantly lower at GHs than at FCHs (all P values 0.001). Similar results for surgical and mental health hospitalizations are shown as Supporting Figures 1 and 2 in the online version of this article. In our sensitivity analyses that included all hospitals classified as GH and FCH, all results were essentially unchanged.

Recognizing the wide range of pediatric volumes at GHs (Table 1) and our inability to differentiate children's hospitals nested within GHs from GHs with pediatric beds, we examined differences in patient and hospitalization characteristics at GHs with volumes 5838 hospitalizations (the 25th percentile for FCH volume) and GHs with pediatric volumes 5838/year (see Supporting Table 2 in the online version of this article). We also compared patient and hospitalization characteristics at FCHs and the higher‐volume GHs. A total of 36 GHs had pediatric volumes 5838, with hospitalizations at these sites together accounting for 15.4% of all pediatric hospitalizations. Characteristics of patients hospitalized at these higher‐volume GHs were similar to patients hospitalized at FCHs, but they had significantly lower disease severity, fewer neonatal hospitalizations, shorter LOS, and more high‐turnover hospitalizations than patients hospitalized at FCHs. We also observed several differences between children hospitalized at higher‐ and lower‐volume GHs (see Supporting Table 2 in the online version of this article). Children hospitalized at the lower‐volume GHs were more likely to have public health insurance and less likely to have complex chronic diseases, although overall, 39.0% of all hospitalizations for children with complex chronic diseases occurred at these lower‐volume GHs. Compared to children hospitalized at higher‐volume GHs, children hospitalized at the lower‐volume hospitals had significantly lower disease severity, shorter LOS, more direct admissions, and a greater proportion of routine discharges.
DISCUSSION
Of the 2 million pediatric hospitalizations in the United States in 2012, more than 70% occurred at GHs. We observed considerable heterogeneity in pediatric volumes across GHs, with 11% of pediatric hospitalizations occurring at hospitals with pediatric volumes of 375 hospitalizations annually, whereas 15% of pediatric hospitalizations occurred at GHs with volumes similar to those observed at FCHs. The remaining pediatric hospitalizations at GHs occurred at centers with intermediate volumes. The most common reasons for hospitalization were similar at GHs and FCHs, but the most costly conditions differed substantially. These findings have important implications for pediatric clinical care programs, research, and QI efforts.
Our finding that more than 70% of pediatric hospitalizations occurred at GHs speaks to the importance of quality measurement at these hospitals, whereas low per‐hospital pediatric volumes at the majority of GHs makes such measurement particularly challenging. Several previous studies have illustrated that volumes of pediatric hospitalizations are too small to detect meaningful differences in quality between hospitals using established condition‐specific metrics.[13, 14, 15] Our finding that more than 10% of pediatric hospitalizations occurred at GHs with pediatric volumes 375 year supports previous research suggesting that cross‐cutting, all‐condition quality metrics, composite measures, and/or multihospital reporting networks may be needed to enable quality measurement at these sites. In addition, the heterogeneity in patient volumes and characteristics across GHs raise questions about the applicability of quality metrics developed and validated at FCHs to the many GH settings. Field‐testing quality measures to ensure their validity at diverse GHs, particularly those with patient volumes and infrastructure different from FCHs, will be important to meaningful pediatric quality measurement.
Our results illustrating differences in the most common and costly conditions at GHs and FCHs have further implications for prioritization and implementation of research and QI efforts. Implementation research and QI efforts focused on cardiac and neurosurgical procedures, as well as neonatal intensive care, may have considerable impact on cost and quality at FCHs. At GHs, research and QI efforts focused on common conditions are needed to increase our knowledge of contextually relevant barriers to and facilitators of high‐quality pediatric care. This, however, can be made more difficult by small sample sizes, limited resources, and infrastructure, and competing priorities in adult‐focused GH settings.[16, 17, 18] Multihospital learning collaboratives and partnerships between FCHs and GHs can begin to address these challenges, but their success is contingent upon national advocacy and funding to support pediatric research and quality measures at GHs.
One of the most notable differences in the characteristics of pediatric hospitalizations at GHs and FCHs was the proportion of hospitalizations attributable to children with medical complexity (CMC); more than one‐third of hospitalizations at FCHs were for CMC compared to 1 in 5 at GHs. These findings align with the results of several previous studies describing the substantial resource utilization attributed to CMC, and with growing research, innovation, and quality metrics focused on improving both inpatient and outpatient care for these vulnerable children.[19, 20, 21, 22] Structured complex care programs, developed to improve care coordination and healthcare quality for CMC, are common at FCHs, and have been associated with decreased resource utilization and improved outcomes.[23, 24, 25] Notably, however, more than half of all hospitalizations for CMC, exceeding 250,000 annually, occurred at GHs, and almost 40% of hospitalizations for CMC occurred at the lower‐volume GHs. These findings speak to the importance of translating effective and innovative programs of care for CMC to GHs as resources allow, accompanied by robust evaluations of their effectiveness. Lower patient volume at most GHs, however, may be a barrier to dedicated CMC programs. As a result, decentralized community‐based programs of care for CMC, linking primary care programs with regional and tertiary care hospitals, warrant further consideration.[26, 27, 28]
This analysis should be interpreted in light of several limitations. First, we were unable to distinguish between GHs with scant pediatric‐specific resources from those with a large volume of dedicated pediatric resources, such as children's hospitals nested within GHs. We did identify 36 GHs with pediatric volumes similar to those observed at FCHs (see Supporting Table 2 in the online version of this article); patient and hospitalization characteristics at these higher‐volume GHs were similar in many ways to children hospitalized at FCHs. Several of these higher‐volume GHs may have considerable resources dedicated to the care of children, including subspecialty care, and may represent children's hospitals nested within GHs. Because nested children's hospitals are included in the GH categorization, our results may have underestimated the proportion of children cared for at children's hospitals. Further work is needed to identify the health systems challenges and opportunities that may be unique to these institutions. Second, because the 2012 KID does not include a specialty hospital indicator, we developed a proxy method for identifying these hospitals, which may have resulted in some misclassification. We are reassured that the results of our analyses did not change substantively when we included all hospitals. Similarly, although we are reassured that the number of hospitals classified in our analysis as acute care FCHs aligns, approximately, with the number of hospitals classified as such by the Children's Hospital Association, we were unable to assess the validity of this variable within the KID. Third, the KID does not link records at the patient level, so we are unable to report the number of unique children included in this analysis. In addition, the KID includes only inpatient stays with exclusion of observation status stays; potential differences between GH and FCH in the use of observation status could have biased our findings. Fifth, we used the PMCA to identify CMC; although this algorithm has been shown to have excellent sensitivity in identifying children with chronic diseases, using up to 3 years of Medicaid claims data, the sensitivity using the KID, where only 1 inpatient stay is available for assessment, is unknown.[8, 29] Similarly, use of Keren's pediatric diagnosis grouper to classify reasons for hospitalization may have resulted in misclassification, though there are few other nonproprietary pediatric‐specific diagnostic groupers available.
In 2012, more than 70% of pediatric hospitalizations occurred at GHs in the United States. The considerably higher pediatric volumes at FCHs makes these institutions well suited for research, innovation, and the development and application of disease‐specific QI initiatives. Recognizing that the majority of pediatric hospitalizations occurred at GHs, there is a clear need for implementation research, program development, and quality metrics that align with the characteristics of hospitalizations at these centers. National support for research and quality improvement that reflects the diverse hospital settings where children receive their hospital care is critical to further our nation's goal of improving hospital quality for children.
Disclosures
Dr. Leyenaar was supported by grant number K08HS024133 from the Agency for Healthcare Research and Quality. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality. The authors have no conflicts of interest relevant to this article to disclose.
- , , , . Mirror, Mirror on the wall: how the performance of the US health care system compares internationally. The Commonwealth Fund. Available at: http://www.commonwealthfund.org/publications/fund‐reports/2014/jun/mirror‐mirror. Published June 16, 2014. Accessed August 26, 2015.
- , , , , , . Higher cost, but poorer outcomes: the US health disadvantage and implications for pediatrics. Pediatrics. 2015;135(6):961–964.
- , , , , . US health spending trends by age and gender: selected years 2002–10. Health Aff (Millwood). 2014;33(5):815–822.
- , , . Costs for hospital stays in the United States, 2012. Healthcare Cost and Utilization Project 181. Available at: http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb181‐Hospital‐Costs‐United‐States‐2012.pdf. Published October 2014. Accessed September 2015.
- , , , et al. All Patient Refined Diagnosis Related Groups: Methodology Overview. 3M Health Information Systems. Available at: https://www.hcup‐us.ahrq.gov/db/nation/nis/APR‐DRGsV20MethodologyOverviewandBibliography.pdf. Accessed February 8, 2016.
- Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project. Introduction to the HCUP Kids' Inpatient Database (KID) 2012. Available at: https://www.hcup‐us.ahrq.gov/db/nation/kid/kid_2012_introduction.jsp. Published Issued July 2014. Accessed February 8, 2016.
- . Prioritization of comparative effectiveness research topics in hospital pediatrics. Arch Pediatr Adolesc Med. 2012;166(12):1155.
- , , , et al. Pediatric medical complexity algorithm: a new method to stratify children by medical complexity. Pediatrics. 2014;133(6):e1647–e1654.
- , , , et al. 3M APR DRG Classification System. 3M Health Information Systems. Available at: http://www.hcup‐us.ahrq.gov/db/nation/nis/v261_aprdrg_meth_ovrview.pdf. Accessed August 7, 2015.
- , , , , , . Trends in high‐turnover stays among children hospitalized in the United States, 1993–2003. Pediatrics. 2009;123(3):996–1002.
- , , , . High turnover stays for pediatric asthma in the United States. Med Care. 2010;48(9):827–833.
- , , , , . Variation and outcomes associated with direct admission among children with pneumonia in the United States. JAMA Pediatr. 2014;168(9):829–836.
- , , , et al. Recognizing differences in hospital quality performance for pediatric inpatient care. Pediatrics. 2015;136(2):251–262.
- , , . Small numbers limit the use of the inpatient pediatric quality indicators for hospital comparison. Acad Pediatr. 2010;10(4):266–273.
- , , , et al. Statistical uncertainty of mortality rates and rankings for children's hospitals. Pediatrics. 2011;128(4):e966–e972.
- , , , , . Determinants of career satisfaction among pediatric hospitalists: a qualitative exploration. Acad Pediatr. 2014;14(4):361–368.
- , , , et al. Quality improvement research in pediatric hospital medicine and the role of the Pediatric Research in Inpatient Settings (PRIS) network. Acad Pediatr. 2013;13(6 suppl):S54–S60.
- . Roles for children's hospitals in pediatric collaborative improvement networks. Pediatrics. 2013;131(suppl 4):S215–S218.
- , , , et al. Children with medical complexity: an emerging population for clinical and research initiatives. Pediatrics. 2011;127(3):529–538.
- , , , et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647–655.
- , , , , , . Patterns and costs of health care use of children with medical complexity. Pediatrics. 2012;130(6):e1463–e1470.
- , , , , , . Hospital utilization and characteristics of patients experiencing recurrent readmissions within children's hospitals. JAMA. 2011;305(7):682–690.
- , , , et al. Characteristics of hospitalizations for patients who use a structured clinical care program for children with medical complexity. J Pediatr. 2011;159(2):284–290.
- , , , . Hospital‐based comprehensive care programs for children with special health care needs: a systematic review. Arch Pediatr Adolesc Med. 2011;165(6):554–561.
- , , , , , . A tertiary care–primary care partnership model for medically complex and fragile children and youth with special health care needs. Arch Pediatr Adolesc Med. 2007;161(10):937–944.
- , , , et al. Integrated complex care coordination for children with medical complexity: a mixed‐methods evaluation of tertiary care‐community collaboration. BMC Health Serv Res. 2012;12:366.
- , , , . Medical transport of children with complex chronic conditions. Emerg Med Int. 2012;2012:837020.
- , , . Comanagement of medically complex children by subspecialists, generalists, and care coordinators. Pediatrics. 2014;134(2):203–205.
- , , , , . Ways to identify children with medical complexity and the importance of why. J Pediatr. 2015;167(2):229–237.
- , , , . Mirror, Mirror on the wall: how the performance of the US health care system compares internationally. The Commonwealth Fund. Available at: http://www.commonwealthfund.org/publications/fund‐reports/2014/jun/mirror‐mirror. Published June 16, 2014. Accessed August 26, 2015.
- , , , , , . Higher cost, but poorer outcomes: the US health disadvantage and implications for pediatrics. Pediatrics. 2015;135(6):961–964.
- , , , , . US health spending trends by age and gender: selected years 2002–10. Health Aff (Millwood). 2014;33(5):815–822.
- , , . Costs for hospital stays in the United States, 2012. Healthcare Cost and Utilization Project 181. Available at: http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb181‐Hospital‐Costs‐United‐States‐2012.pdf. Published October 2014. Accessed September 2015.
- , , , et al. All Patient Refined Diagnosis Related Groups: Methodology Overview. 3M Health Information Systems. Available at: https://www.hcup‐us.ahrq.gov/db/nation/nis/APR‐DRGsV20MethodologyOverviewandBibliography.pdf. Accessed February 8, 2016.
- Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project. Introduction to the HCUP Kids' Inpatient Database (KID) 2012. Available at: https://www.hcup‐us.ahrq.gov/db/nation/kid/kid_2012_introduction.jsp. Published Issued July 2014. Accessed February 8, 2016.
- . Prioritization of comparative effectiveness research topics in hospital pediatrics. Arch Pediatr Adolesc Med. 2012;166(12):1155.
- , , , et al. Pediatric medical complexity algorithm: a new method to stratify children by medical complexity. Pediatrics. 2014;133(6):e1647–e1654.
- , , , et al. 3M APR DRG Classification System. 3M Health Information Systems. Available at: http://www.hcup‐us.ahrq.gov/db/nation/nis/v261_aprdrg_meth_ovrview.pdf. Accessed August 7, 2015.
- , , , , , . Trends in high‐turnover stays among children hospitalized in the United States, 1993–2003. Pediatrics. 2009;123(3):996–1002.
- , , , . High turnover stays for pediatric asthma in the United States. Med Care. 2010;48(9):827–833.
- , , , , . Variation and outcomes associated with direct admission among children with pneumonia in the United States. JAMA Pediatr. 2014;168(9):829–836.
- , , , et al. Recognizing differences in hospital quality performance for pediatric inpatient care. Pediatrics. 2015;136(2):251–262.
- , , . Small numbers limit the use of the inpatient pediatric quality indicators for hospital comparison. Acad Pediatr. 2010;10(4):266–273.
- , , , et al. Statistical uncertainty of mortality rates and rankings for children's hospitals. Pediatrics. 2011;128(4):e966–e972.
- , , , , . Determinants of career satisfaction among pediatric hospitalists: a qualitative exploration. Acad Pediatr. 2014;14(4):361–368.
- , , , et al. Quality improvement research in pediatric hospital medicine and the role of the Pediatric Research in Inpatient Settings (PRIS) network. Acad Pediatr. 2013;13(6 suppl):S54–S60.
- . Roles for children's hospitals in pediatric collaborative improvement networks. Pediatrics. 2013;131(suppl 4):S215–S218.
- , , , et al. Children with medical complexity: an emerging population for clinical and research initiatives. Pediatrics. 2011;127(3):529–538.
- , , , et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647–655.
- , , , , , . Patterns and costs of health care use of children with medical complexity. Pediatrics. 2012;130(6):e1463–e1470.
- , , , , , . Hospital utilization and characteristics of patients experiencing recurrent readmissions within children's hospitals. JAMA. 2011;305(7):682–690.
- , , , et al. Characteristics of hospitalizations for patients who use a structured clinical care program for children with medical complexity. J Pediatr. 2011;159(2):284–290.
- , , , . Hospital‐based comprehensive care programs for children with special health care needs: a systematic review. Arch Pediatr Adolesc Med. 2011;165(6):554–561.
- , , , , , . A tertiary care–primary care partnership model for medically complex and fragile children and youth with special health care needs. Arch Pediatr Adolesc Med. 2007;161(10):937–944.
- , , , et al. Integrated complex care coordination for children with medical complexity: a mixed‐methods evaluation of tertiary care‐community collaboration. BMC Health Serv Res. 2012;12:366.
- , , , . Medical transport of children with complex chronic conditions. Emerg Med Int. 2012;2012:837020.
- , , . Comanagement of medically complex children by subspecialists, generalists, and care coordinators. Pediatrics. 2014;134(2):203–205.
- , , , , . Ways to identify children with medical complexity and the importance of why. J Pediatr. 2015;167(2):229–237.
LETTER: Point-of-Care Ultrasound: The (Sound) Wave of the Future for Hospitalists
Small devices carried in pockets during rounds can enable hospitalists to make quick decisions at the bedside, enhance and teach physical exam skills, streamline patient flow through the hospital, and potentially avoid the cost and risk of exposure to radiation. Point-of-care (POC) ultrasound enhances both patient satisfaction and the clinician’s professional satisfaction. Hospital medicine will be the next field to rapidly assimilate its use.
POC, or “bedside,” ultrasound has been used by ob-gyns, vascular access, and procedural teams for quite some time. Of late, emergency medicine and critical care physicians have adopted its use. It offers the advantage of gaining immediate information regarding the patient through dynamic imaging and the ability to integrate that information into the clinical picture. This enables providers to make decisions about patient care in real time.
With the advent of affordable handheld devices with quality images, rounding with these devices has become practical for hospitalists. Hospitalists should rapidly embrace this skill set. POC ultrasound can be very useful to quickly improve patient diagnosis, patient satisfaction, patient safety, length of stay, and provider satisfaction.
For example, in patients complaining of dyspnea, for which there is not a clear diagnosis of COPD, congestive heart failure, pulmonary embolism, or pneumonia, a focused cardiac ultrasound can rapidly differentiate between right ventricular dysfunction, left ventricular dysfunction, pericardial effusion, or a hyperdynamic heart. Lung ultrasound with diffuse or focal “B lines,” focal consolidation, and/or pleural effusion can assist in differentiating the cause as well.
POC ultrasound also is a teaching tool that can enhance exam skills. Hospitalists can confirm exam findings and teach as they palpate the liver or percuss the chest. Performing a procedure such as paracentesis or a central line with ultrasound guidance is now considered standard of care in some centers. The literature shows ultrasound guidance is safer even when compared to clinicians skilled in landmark techniques. In addition, many hospitalists and/or trainees will work in areas where 24-7 echo, interventional radiologists, and ultrasound techs are not available. Hospitalists need to know how to use POC ultrasound to serve patients well.
POC ultrasound can also be used in daily care. For heart failure patients, watching the B lines (pulmonary edema), pleural effusions, and inferior vena cava size can avoid over- or under-diuresis and reduce length of stay and cost. The same can be said for patients with percutaneous catheters to ensure proper drainage of the pockets of fluid in the chest or abdomen.
It is important to know the limitations of POC ultrasound. It is best used to answer binary questions (e.g., pericardial effusion present or not). It is a skill to be acquired and honed, and it requires specialized training. There are many one- to two-day courses as well simulators and other means. The basics of image acquisition and interpretation can be found online, and much of it is free. Manufacturers often are willing to provide machines to practice with.
Many patients enjoy seeing the images and having a better understanding of their disease process, which leads to improved patient satisfaction. Overall, there are many benefits for hospitalists.
Gordon Johnson, MD, hospitalist and president, Oregon/Southwest Washington SHM Chapter
Small devices carried in pockets during rounds can enable hospitalists to make quick decisions at the bedside, enhance and teach physical exam skills, streamline patient flow through the hospital, and potentially avoid the cost and risk of exposure to radiation. Point-of-care (POC) ultrasound enhances both patient satisfaction and the clinician’s professional satisfaction. Hospital medicine will be the next field to rapidly assimilate its use.
POC, or “bedside,” ultrasound has been used by ob-gyns, vascular access, and procedural teams for quite some time. Of late, emergency medicine and critical care physicians have adopted its use. It offers the advantage of gaining immediate information regarding the patient through dynamic imaging and the ability to integrate that information into the clinical picture. This enables providers to make decisions about patient care in real time.
With the advent of affordable handheld devices with quality images, rounding with these devices has become practical for hospitalists. Hospitalists should rapidly embrace this skill set. POC ultrasound can be very useful to quickly improve patient diagnosis, patient satisfaction, patient safety, length of stay, and provider satisfaction.
For example, in patients complaining of dyspnea, for which there is not a clear diagnosis of COPD, congestive heart failure, pulmonary embolism, or pneumonia, a focused cardiac ultrasound can rapidly differentiate between right ventricular dysfunction, left ventricular dysfunction, pericardial effusion, or a hyperdynamic heart. Lung ultrasound with diffuse or focal “B lines,” focal consolidation, and/or pleural effusion can assist in differentiating the cause as well.
POC ultrasound also is a teaching tool that can enhance exam skills. Hospitalists can confirm exam findings and teach as they palpate the liver or percuss the chest. Performing a procedure such as paracentesis or a central line with ultrasound guidance is now considered standard of care in some centers. The literature shows ultrasound guidance is safer even when compared to clinicians skilled in landmark techniques. In addition, many hospitalists and/or trainees will work in areas where 24-7 echo, interventional radiologists, and ultrasound techs are not available. Hospitalists need to know how to use POC ultrasound to serve patients well.
POC ultrasound can also be used in daily care. For heart failure patients, watching the B lines (pulmonary edema), pleural effusions, and inferior vena cava size can avoid over- or under-diuresis and reduce length of stay and cost. The same can be said for patients with percutaneous catheters to ensure proper drainage of the pockets of fluid in the chest or abdomen.
It is important to know the limitations of POC ultrasound. It is best used to answer binary questions (e.g., pericardial effusion present or not). It is a skill to be acquired and honed, and it requires specialized training. There are many one- to two-day courses as well simulators and other means. The basics of image acquisition and interpretation can be found online, and much of it is free. Manufacturers often are willing to provide machines to practice with.
Many patients enjoy seeing the images and having a better understanding of their disease process, which leads to improved patient satisfaction. Overall, there are many benefits for hospitalists.
Gordon Johnson, MD, hospitalist and president, Oregon/Southwest Washington SHM Chapter
Small devices carried in pockets during rounds can enable hospitalists to make quick decisions at the bedside, enhance and teach physical exam skills, streamline patient flow through the hospital, and potentially avoid the cost and risk of exposure to radiation. Point-of-care (POC) ultrasound enhances both patient satisfaction and the clinician’s professional satisfaction. Hospital medicine will be the next field to rapidly assimilate its use.
POC, or “bedside,” ultrasound has been used by ob-gyns, vascular access, and procedural teams for quite some time. Of late, emergency medicine and critical care physicians have adopted its use. It offers the advantage of gaining immediate information regarding the patient through dynamic imaging and the ability to integrate that information into the clinical picture. This enables providers to make decisions about patient care in real time.
With the advent of affordable handheld devices with quality images, rounding with these devices has become practical for hospitalists. Hospitalists should rapidly embrace this skill set. POC ultrasound can be very useful to quickly improve patient diagnosis, patient satisfaction, patient safety, length of stay, and provider satisfaction.
For example, in patients complaining of dyspnea, for which there is not a clear diagnosis of COPD, congestive heart failure, pulmonary embolism, or pneumonia, a focused cardiac ultrasound can rapidly differentiate between right ventricular dysfunction, left ventricular dysfunction, pericardial effusion, or a hyperdynamic heart. Lung ultrasound with diffuse or focal “B lines,” focal consolidation, and/or pleural effusion can assist in differentiating the cause as well.
POC ultrasound also is a teaching tool that can enhance exam skills. Hospitalists can confirm exam findings and teach as they palpate the liver or percuss the chest. Performing a procedure such as paracentesis or a central line with ultrasound guidance is now considered standard of care in some centers. The literature shows ultrasound guidance is safer even when compared to clinicians skilled in landmark techniques. In addition, many hospitalists and/or trainees will work in areas where 24-7 echo, interventional radiologists, and ultrasound techs are not available. Hospitalists need to know how to use POC ultrasound to serve patients well.
POC ultrasound can also be used in daily care. For heart failure patients, watching the B lines (pulmonary edema), pleural effusions, and inferior vena cava size can avoid over- or under-diuresis and reduce length of stay and cost. The same can be said for patients with percutaneous catheters to ensure proper drainage of the pockets of fluid in the chest or abdomen.
It is important to know the limitations of POC ultrasound. It is best used to answer binary questions (e.g., pericardial effusion present or not). It is a skill to be acquired and honed, and it requires specialized training. There are many one- to two-day courses as well simulators and other means. The basics of image acquisition and interpretation can be found online, and much of it is free. Manufacturers often are willing to provide machines to practice with.
Many patients enjoy seeing the images and having a better understanding of their disease process, which leads to improved patient satisfaction. Overall, there are many benefits for hospitalists.
Gordon Johnson, MD, hospitalist and president, Oregon/Southwest Washington SHM Chapter
Efficacy of malaria vaccine declines over time

Photo by Caitlin Kleiboer
Results from a phase 2 study of the malaria vaccine RTS,S (also known as RTS,S/AS01 or Mosquirix) suggest its efficacy decreases over time, and this decline is fastest in children living in areas with higher-than-average rates of malaria.
Researchers say the results suggest the benefits of the vaccine are likely to vary across different populations and highlight the need for more research to
determine the most effective way of using RTS,S, which last year became the first malaria vaccine to receive a green light from the European Medicines Agency.
“We found that 3-dose vaccination with RTS,S was initially protective, but this was offset by a rebound in later years among children exposed to higher-than-average levels of malaria-carrying mosquitoes,” said Philip Bejon, PhD, of the Kenya Medical Research Institute–Wellcome Trust Programme in Kilifi, Kenya.
Dr Bejon and his colleagues reported these results in NEJM.
The researchers followed 447 children who had received 3 doses of either RTS,S or a rabies (control) vaccine when they were 5 months to 17 months old.
After 7 years, there were 312 children still involved in the study. During the first year, the risk of getting malaria in the vaccinated children was 35.9% less than in the control group. After 7 years, this protection fell to 3.6%.
And in children exposed to higher-than-average rates of malaria, there were slightly more cases of malaria in the vaccinated group than the control group—1002 and 992 cases, respectively—5 years after vaccination.
This “rebound” effect, which has been seen in previous studies, is thought to occur because children initially protected by the vaccine develop their natural immunity against malaria more slowly than unvaccinated children.
Results from a phase 3 study showed that 3 doses of RTS,S reduced the risk of malaria in young children by 28% over 4 years, but this improved to 36% when children were given a fourth dose 18 months after the first dose. Longer-term follow up of these children is ongoing.
“Overall, our study shows that RTS,S can benefit children but suggests that a fourth dose may be important for sustaining this protection over the long term and to protect against a potential rebound,” said Ally Olotu, PhD, of the Kenya Medical Research Institute–Wellcome Trust Programme.
“Results from 3 sites involved in the original phase 3 study that are continuing follow up, and the WHO’s planned pilot program, will tell us more about the vaccine’s efficacy in different settings and help determine which populations would benefit most from receiving it as part of a wider vaccination strategy.” ![]()

Photo by Caitlin Kleiboer
Results from a phase 2 study of the malaria vaccine RTS,S (also known as RTS,S/AS01 or Mosquirix) suggest its efficacy decreases over time, and this decline is fastest in children living in areas with higher-than-average rates of malaria.
Researchers say the results suggest the benefits of the vaccine are likely to vary across different populations and highlight the need for more research to
determine the most effective way of using RTS,S, which last year became the first malaria vaccine to receive a green light from the European Medicines Agency.
“We found that 3-dose vaccination with RTS,S was initially protective, but this was offset by a rebound in later years among children exposed to higher-than-average levels of malaria-carrying mosquitoes,” said Philip Bejon, PhD, of the Kenya Medical Research Institute–Wellcome Trust Programme in Kilifi, Kenya.
Dr Bejon and his colleagues reported these results in NEJM.
The researchers followed 447 children who had received 3 doses of either RTS,S or a rabies (control) vaccine when they were 5 months to 17 months old.
After 7 years, there were 312 children still involved in the study. During the first year, the risk of getting malaria in the vaccinated children was 35.9% less than in the control group. After 7 years, this protection fell to 3.6%.
And in children exposed to higher-than-average rates of malaria, there were slightly more cases of malaria in the vaccinated group than the control group—1002 and 992 cases, respectively—5 years after vaccination.
This “rebound” effect, which has been seen in previous studies, is thought to occur because children initially protected by the vaccine develop their natural immunity against malaria more slowly than unvaccinated children.
Results from a phase 3 study showed that 3 doses of RTS,S reduced the risk of malaria in young children by 28% over 4 years, but this improved to 36% when children were given a fourth dose 18 months after the first dose. Longer-term follow up of these children is ongoing.
“Overall, our study shows that RTS,S can benefit children but suggests that a fourth dose may be important for sustaining this protection over the long term and to protect against a potential rebound,” said Ally Olotu, PhD, of the Kenya Medical Research Institute–Wellcome Trust Programme.
“Results from 3 sites involved in the original phase 3 study that are continuing follow up, and the WHO’s planned pilot program, will tell us more about the vaccine’s efficacy in different settings and help determine which populations would benefit most from receiving it as part of a wider vaccination strategy.” ![]()

Photo by Caitlin Kleiboer
Results from a phase 2 study of the malaria vaccine RTS,S (also known as RTS,S/AS01 or Mosquirix) suggest its efficacy decreases over time, and this decline is fastest in children living in areas with higher-than-average rates of malaria.
Researchers say the results suggest the benefits of the vaccine are likely to vary across different populations and highlight the need for more research to
determine the most effective way of using RTS,S, which last year became the first malaria vaccine to receive a green light from the European Medicines Agency.
“We found that 3-dose vaccination with RTS,S was initially protective, but this was offset by a rebound in later years among children exposed to higher-than-average levels of malaria-carrying mosquitoes,” said Philip Bejon, PhD, of the Kenya Medical Research Institute–Wellcome Trust Programme in Kilifi, Kenya.
Dr Bejon and his colleagues reported these results in NEJM.
The researchers followed 447 children who had received 3 doses of either RTS,S or a rabies (control) vaccine when they were 5 months to 17 months old.
After 7 years, there were 312 children still involved in the study. During the first year, the risk of getting malaria in the vaccinated children was 35.9% less than in the control group. After 7 years, this protection fell to 3.6%.
And in children exposed to higher-than-average rates of malaria, there were slightly more cases of malaria in the vaccinated group than the control group—1002 and 992 cases, respectively—5 years after vaccination.
This “rebound” effect, which has been seen in previous studies, is thought to occur because children initially protected by the vaccine develop their natural immunity against malaria more slowly than unvaccinated children.
Results from a phase 3 study showed that 3 doses of RTS,S reduced the risk of malaria in young children by 28% over 4 years, but this improved to 36% when children were given a fourth dose 18 months after the first dose. Longer-term follow up of these children is ongoing.
“Overall, our study shows that RTS,S can benefit children but suggests that a fourth dose may be important for sustaining this protection over the long term and to protect against a potential rebound,” said Ally Olotu, PhD, of the Kenya Medical Research Institute–Wellcome Trust Programme.
“Results from 3 sites involved in the original phase 3 study that are continuing follow up, and the WHO’s planned pilot program, will tell us more about the vaccine’s efficacy in different settings and help determine which populations would benefit most from receiving it as part of a wider vaccination strategy.” ![]()
MACRA Rule Offers Little Clarity for Hospitalists
Last year, Congress put an end to the Sustainable Growth Rate (SGR), which had become a yearly battle fought on behalf of and by physicians to prevent significant last-minute cuts to Medicare reimbursement. Many hoped its replacement would provide more stability and certainty.
However, that replacement, the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA), has been anything but clear. On April 27, 2016, the Centers for Medicare & Medicaid Services (CMS) issued a Notice of Proposed Rulemaking in what it called a “first step” in implementing MACRA. CMS accepted feedback and input on the proposed rule through June 27, 2016.
The Society of Hospital Medicine worked to provide comment on what it sees as the biggest concerns of hospitalists.
For example, it remains unclear what quality markers CMS will use to evaluate hospitalists under MACRA, says Rush University Medical Center’s Suparna Dutta, MD, MPH, a hospitalist, assistant professor of medicine, and member of the SHM Public Policy Committee (PPC). “The biggest piece is, what will be used universally for all hospitalists and attributed to the work that we do?”
MACRA represents “a milestone” in efforts to “advance a healthcare system that rewards better care, smarter spending, and healthier people,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in a statement issued the day the proposed rule was announced.
What it is designed to do, says Ron Greeno, MD, MHM, president-elect of SHM, PPC chair, and senior advisor for medical affairs at TeamHealth, is push physicians to move toward alternative payment models.
To achieve this, MACRA creates a framework called the Quality Payment Program, which offers physicians two paths for value-over-volume-based payments: MIPS, for Merit-Based Incentive Payment System, and APMs, for Advanced Alternative Payment Models. The benchmark period for both pathways begins Jan. 1, 2017, and MACRA reimbursement would begin Jan. 1, 2019.
Under MIPS, current quality measurement programs are streamlined into a single payment adjustment, including the Physician Value-Based Modifier, the Electronic Health Record (EHR) Incentive Program and the Physician Quality Reporting System (PQRS).
Physicians will not assume risk on the MIPS pathway, but payment adjustments will be based on their MIPS score, which grows each year through 2022 and ranges that year from +9% to -9%. It will be budget neutral: The top half of scorers will see increases in payments, while the bottom half will see cuts. Additional adjustments will be given to top performers through 2024.
However, as Dr. Dutta and fellow PPC member Lauren Doctoroff, MD, FHM, a hospitalist at Beth Israel Deaconess Medical Center and instructor at Harvard Medical School, wrote for The Hospitalist in March 2016, it is not yet clear how MIPS scores will be calculated for hospitalists.
“The problem is that there is not a typical hospitalist in terms of the work that we do,” Dr. Dutta says. “It depends on the hospital and the types of responsibilities the hospitalists have and the types of patients they care for.”
CMS says 50% of the MIPS score will come from six reported measures that reflect different specialties and practices; 25% will come from technology use, with a focus on interoperability and information exchange; 15% will come from clinical improvement practices, like care coordination; and 10% will be based on cost, chosen from among 40 episode-specific measures.
The new hospitalist billing code, which has not yet been implemented, should be a tremendous help under MACRA, Dr. Dutta says. “As CMS plans on using peer-comparison groups for quality and cost measures, it is really important that we now have a specialty billing code for hospitalists, which should ensure we have a fair and valid comparison pool for any metrics we are measured on for MIPS.”
The second path may be much harder for hospitalists to achieve since it requires that physicians share in risk and reward and participate in alternative payment models like Next Generation ACO or the Comprehensive Primary Care Plus model.
Most hospitalists will not be candidates for taking on risks under APM since physicians need to achieve a threshold for taking on more than nominal financial risk, Dr. Dutta says, noting SHM’s efforts to better understand the implications.
“It depends on the the percentage of patients you’re seeing in an APM, and you might hit your threshold if your market has a lot of Medicare ACOs or risk-sharing, but it’s not something hospitalists can consistently plan on,” Dr. Dutta says.
Most hospitalists have little control over whether their facility participates in an APM, Dr. Dutta says, but allowing the APM to which a patient belongs count toward the care provided by hospitalists—though a patient may align with several APMs—may help reach these thresholds.
Feedback from SHM to CMS also included asking to allow the Bundled Payments for Care Improvement Initiative (BPCI) to qualify for APM and seeking clarification into whether hospitalists can tap into cost and quality metrics hospitals are already reporting to CMS.
“Hospitals are collecting a certain amount of data because they have to for Medicare, and that might be a good indicator of what hospitalists are doing,” Dr. Dutta says. This includes services like DVT prophylaxis after surgery in hospitals where hospitalists provide a majority of post-operative care or safety measures like CLABSI (central line–associated bloodstream infection) rates.
To stay up to date with MACRA, visit SHM’s MACRA website and follow @SHMadvocacy on Twitter. TH
Corrected version July 13, 2016.
Kelly April Tyrrell is a freelance writer in Madison, Wis.
Last year, Congress put an end to the Sustainable Growth Rate (SGR), which had become a yearly battle fought on behalf of and by physicians to prevent significant last-minute cuts to Medicare reimbursement. Many hoped its replacement would provide more stability and certainty.
However, that replacement, the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA), has been anything but clear. On April 27, 2016, the Centers for Medicare & Medicaid Services (CMS) issued a Notice of Proposed Rulemaking in what it called a “first step” in implementing MACRA. CMS accepted feedback and input on the proposed rule through June 27, 2016.
The Society of Hospital Medicine worked to provide comment on what it sees as the biggest concerns of hospitalists.
For example, it remains unclear what quality markers CMS will use to evaluate hospitalists under MACRA, says Rush University Medical Center’s Suparna Dutta, MD, MPH, a hospitalist, assistant professor of medicine, and member of the SHM Public Policy Committee (PPC). “The biggest piece is, what will be used universally for all hospitalists and attributed to the work that we do?”
MACRA represents “a milestone” in efforts to “advance a healthcare system that rewards better care, smarter spending, and healthier people,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in a statement issued the day the proposed rule was announced.
What it is designed to do, says Ron Greeno, MD, MHM, president-elect of SHM, PPC chair, and senior advisor for medical affairs at TeamHealth, is push physicians to move toward alternative payment models.
To achieve this, MACRA creates a framework called the Quality Payment Program, which offers physicians two paths for value-over-volume-based payments: MIPS, for Merit-Based Incentive Payment System, and APMs, for Advanced Alternative Payment Models. The benchmark period for both pathways begins Jan. 1, 2017, and MACRA reimbursement would begin Jan. 1, 2019.
Under MIPS, current quality measurement programs are streamlined into a single payment adjustment, including the Physician Value-Based Modifier, the Electronic Health Record (EHR) Incentive Program and the Physician Quality Reporting System (PQRS).
Physicians will not assume risk on the MIPS pathway, but payment adjustments will be based on their MIPS score, which grows each year through 2022 and ranges that year from +9% to -9%. It will be budget neutral: The top half of scorers will see increases in payments, while the bottom half will see cuts. Additional adjustments will be given to top performers through 2024.
However, as Dr. Dutta and fellow PPC member Lauren Doctoroff, MD, FHM, a hospitalist at Beth Israel Deaconess Medical Center and instructor at Harvard Medical School, wrote for The Hospitalist in March 2016, it is not yet clear how MIPS scores will be calculated for hospitalists.
“The problem is that there is not a typical hospitalist in terms of the work that we do,” Dr. Dutta says. “It depends on the hospital and the types of responsibilities the hospitalists have and the types of patients they care for.”
CMS says 50% of the MIPS score will come from six reported measures that reflect different specialties and practices; 25% will come from technology use, with a focus on interoperability and information exchange; 15% will come from clinical improvement practices, like care coordination; and 10% will be based on cost, chosen from among 40 episode-specific measures.
The new hospitalist billing code, which has not yet been implemented, should be a tremendous help under MACRA, Dr. Dutta says. “As CMS plans on using peer-comparison groups for quality and cost measures, it is really important that we now have a specialty billing code for hospitalists, which should ensure we have a fair and valid comparison pool for any metrics we are measured on for MIPS.”
The second path may be much harder for hospitalists to achieve since it requires that physicians share in risk and reward and participate in alternative payment models like Next Generation ACO or the Comprehensive Primary Care Plus model.
Most hospitalists will not be candidates for taking on risks under APM since physicians need to achieve a threshold for taking on more than nominal financial risk, Dr. Dutta says, noting SHM’s efforts to better understand the implications.
“It depends on the the percentage of patients you’re seeing in an APM, and you might hit your threshold if your market has a lot of Medicare ACOs or risk-sharing, but it’s not something hospitalists can consistently plan on,” Dr. Dutta says.
Most hospitalists have little control over whether their facility participates in an APM, Dr. Dutta says, but allowing the APM to which a patient belongs count toward the care provided by hospitalists—though a patient may align with several APMs—may help reach these thresholds.
Feedback from SHM to CMS also included asking to allow the Bundled Payments for Care Improvement Initiative (BPCI) to qualify for APM and seeking clarification into whether hospitalists can tap into cost and quality metrics hospitals are already reporting to CMS.
“Hospitals are collecting a certain amount of data because they have to for Medicare, and that might be a good indicator of what hospitalists are doing,” Dr. Dutta says. This includes services like DVT prophylaxis after surgery in hospitals where hospitalists provide a majority of post-operative care or safety measures like CLABSI (central line–associated bloodstream infection) rates.
To stay up to date with MACRA, visit SHM’s MACRA website and follow @SHMadvocacy on Twitter. TH
Corrected version July 13, 2016.
Kelly April Tyrrell is a freelance writer in Madison, Wis.
Last year, Congress put an end to the Sustainable Growth Rate (SGR), which had become a yearly battle fought on behalf of and by physicians to prevent significant last-minute cuts to Medicare reimbursement. Many hoped its replacement would provide more stability and certainty.
However, that replacement, the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA), has been anything but clear. On April 27, 2016, the Centers for Medicare & Medicaid Services (CMS) issued a Notice of Proposed Rulemaking in what it called a “first step” in implementing MACRA. CMS accepted feedback and input on the proposed rule through June 27, 2016.
The Society of Hospital Medicine worked to provide comment on what it sees as the biggest concerns of hospitalists.
For example, it remains unclear what quality markers CMS will use to evaluate hospitalists under MACRA, says Rush University Medical Center’s Suparna Dutta, MD, MPH, a hospitalist, assistant professor of medicine, and member of the SHM Public Policy Committee (PPC). “The biggest piece is, what will be used universally for all hospitalists and attributed to the work that we do?”
MACRA represents “a milestone” in efforts to “advance a healthcare system that rewards better care, smarter spending, and healthier people,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in a statement issued the day the proposed rule was announced.
What it is designed to do, says Ron Greeno, MD, MHM, president-elect of SHM, PPC chair, and senior advisor for medical affairs at TeamHealth, is push physicians to move toward alternative payment models.
To achieve this, MACRA creates a framework called the Quality Payment Program, which offers physicians two paths for value-over-volume-based payments: MIPS, for Merit-Based Incentive Payment System, and APMs, for Advanced Alternative Payment Models. The benchmark period for both pathways begins Jan. 1, 2017, and MACRA reimbursement would begin Jan. 1, 2019.
Under MIPS, current quality measurement programs are streamlined into a single payment adjustment, including the Physician Value-Based Modifier, the Electronic Health Record (EHR) Incentive Program and the Physician Quality Reporting System (PQRS).
Physicians will not assume risk on the MIPS pathway, but payment adjustments will be based on their MIPS score, which grows each year through 2022 and ranges that year from +9% to -9%. It will be budget neutral: The top half of scorers will see increases in payments, while the bottom half will see cuts. Additional adjustments will be given to top performers through 2024.
However, as Dr. Dutta and fellow PPC member Lauren Doctoroff, MD, FHM, a hospitalist at Beth Israel Deaconess Medical Center and instructor at Harvard Medical School, wrote for The Hospitalist in March 2016, it is not yet clear how MIPS scores will be calculated for hospitalists.
“The problem is that there is not a typical hospitalist in terms of the work that we do,” Dr. Dutta says. “It depends on the hospital and the types of responsibilities the hospitalists have and the types of patients they care for.”
CMS says 50% of the MIPS score will come from six reported measures that reflect different specialties and practices; 25% will come from technology use, with a focus on interoperability and information exchange; 15% will come from clinical improvement practices, like care coordination; and 10% will be based on cost, chosen from among 40 episode-specific measures.
The new hospitalist billing code, which has not yet been implemented, should be a tremendous help under MACRA, Dr. Dutta says. “As CMS plans on using peer-comparison groups for quality and cost measures, it is really important that we now have a specialty billing code for hospitalists, which should ensure we have a fair and valid comparison pool for any metrics we are measured on for MIPS.”
The second path may be much harder for hospitalists to achieve since it requires that physicians share in risk and reward and participate in alternative payment models like Next Generation ACO or the Comprehensive Primary Care Plus model.
Most hospitalists will not be candidates for taking on risks under APM since physicians need to achieve a threshold for taking on more than nominal financial risk, Dr. Dutta says, noting SHM’s efforts to better understand the implications.
“It depends on the the percentage of patients you’re seeing in an APM, and you might hit your threshold if your market has a lot of Medicare ACOs or risk-sharing, but it’s not something hospitalists can consistently plan on,” Dr. Dutta says.
Most hospitalists have little control over whether their facility participates in an APM, Dr. Dutta says, but allowing the APM to which a patient belongs count toward the care provided by hospitalists—though a patient may align with several APMs—may help reach these thresholds.
Feedback from SHM to CMS also included asking to allow the Bundled Payments for Care Improvement Initiative (BPCI) to qualify for APM and seeking clarification into whether hospitalists can tap into cost and quality metrics hospitals are already reporting to CMS.
“Hospitals are collecting a certain amount of data because they have to for Medicare, and that might be a good indicator of what hospitalists are doing,” Dr. Dutta says. This includes services like DVT prophylaxis after surgery in hospitals where hospitalists provide a majority of post-operative care or safety measures like CLABSI (central line–associated bloodstream infection) rates.
To stay up to date with MACRA, visit SHM’s MACRA website and follow @SHMadvocacy on Twitter. TH
Corrected version July 13, 2016.
Kelly April Tyrrell is a freelance writer in Madison, Wis.
Telomere length linked to neutrophil recovery in AML

Image by Volker Brinkmann
Researchers say they have discovered a way to predict which children with acute myeloid leukemia (AML) are at the highest risk of delayed neutrophil recovery.
The team examined the role of telomeres in neutrophil recovery and found that the length of a patient’s telomeres can indicate the rate of recovery following chemotherapy.
The group reported their findings in the Journal of Clinical Oncology.
“We were interested in telomere length as a marker of blood count recovery because defects in telomere maintenance are known risks for bone marrow failure and aplastic anemia,” said study author Maria Monica Gramatges, MD, PhD, of Baylor College of Medicine in Houston, Texas.
“We know that up to 15% to 20% of children can take 2 months or longer to recover their blood counts after a course of AML chemotherapy. Our goal was to understand if these children had an underlying genetic predisposition associated with an impaired capacity for recovery.”
Dr Gramatges and her colleagues hypothesized that short telomere length could be associated with a delay in neutrophil recovery.
So they obtained bone marrow samples from AML patients who recovered as expected (within 30 days) after each chemotherapy course (n=62), and from AML patients who experienced significant delays in recovery after chemotherapy (n=53).
The team then measured telomere length on each subject and categorized the group by quartile, from shortest to longest.
Subjects in the quartile with the shortest telomere lengths took the longest to recover, especially during the last 2 courses of chemotherapy. In an adjusted analysis, lower telomere content was significantly associated with prolonged neutropenia after the fourth (P=0.002) and fifth courses of chemotherapy (P=0.009).
The researchers said these results support the hypothesis that telomeres are an indicator of capacity for neutrophil recovery following chemotherapy.
Dr Gramatges hopes the results of this study will be helpful in further understanding which children are at a higher risk for prolonged myelosuppression and how to target those children with modified treatments, improved supportive care, and closer monitoring in order to prevent potential complications such as severe infections.
“A significant proportion of children with AML suffer from treatment-related toxicities, with some succumbing to complications of the therapies we give, rather than from the actual cancer itself,” Dr Gramatges said.
“We hope this research will help us identify those who are at a higher risk for delayed recovery and use this knowledge to reduce the morbidity and mortality associated with AML treatment.” ![]()

Image by Volker Brinkmann
Researchers say they have discovered a way to predict which children with acute myeloid leukemia (AML) are at the highest risk of delayed neutrophil recovery.
The team examined the role of telomeres in neutrophil recovery and found that the length of a patient’s telomeres can indicate the rate of recovery following chemotherapy.
The group reported their findings in the Journal of Clinical Oncology.
“We were interested in telomere length as a marker of blood count recovery because defects in telomere maintenance are known risks for bone marrow failure and aplastic anemia,” said study author Maria Monica Gramatges, MD, PhD, of Baylor College of Medicine in Houston, Texas.
“We know that up to 15% to 20% of children can take 2 months or longer to recover their blood counts after a course of AML chemotherapy. Our goal was to understand if these children had an underlying genetic predisposition associated with an impaired capacity for recovery.”
Dr Gramatges and her colleagues hypothesized that short telomere length could be associated with a delay in neutrophil recovery.
So they obtained bone marrow samples from AML patients who recovered as expected (within 30 days) after each chemotherapy course (n=62), and from AML patients who experienced significant delays in recovery after chemotherapy (n=53).
The team then measured telomere length on each subject and categorized the group by quartile, from shortest to longest.
Subjects in the quartile with the shortest telomere lengths took the longest to recover, especially during the last 2 courses of chemotherapy. In an adjusted analysis, lower telomere content was significantly associated with prolonged neutropenia after the fourth (P=0.002) and fifth courses of chemotherapy (P=0.009).
The researchers said these results support the hypothesis that telomeres are an indicator of capacity for neutrophil recovery following chemotherapy.
Dr Gramatges hopes the results of this study will be helpful in further understanding which children are at a higher risk for prolonged myelosuppression and how to target those children with modified treatments, improved supportive care, and closer monitoring in order to prevent potential complications such as severe infections.
“A significant proportion of children with AML suffer from treatment-related toxicities, with some succumbing to complications of the therapies we give, rather than from the actual cancer itself,” Dr Gramatges said.
“We hope this research will help us identify those who are at a higher risk for delayed recovery and use this knowledge to reduce the morbidity and mortality associated with AML treatment.” ![]()

Image by Volker Brinkmann
Researchers say they have discovered a way to predict which children with acute myeloid leukemia (AML) are at the highest risk of delayed neutrophil recovery.
The team examined the role of telomeres in neutrophil recovery and found that the length of a patient’s telomeres can indicate the rate of recovery following chemotherapy.
The group reported their findings in the Journal of Clinical Oncology.
“We were interested in telomere length as a marker of blood count recovery because defects in telomere maintenance are known risks for bone marrow failure and aplastic anemia,” said study author Maria Monica Gramatges, MD, PhD, of Baylor College of Medicine in Houston, Texas.
“We know that up to 15% to 20% of children can take 2 months or longer to recover their blood counts after a course of AML chemotherapy. Our goal was to understand if these children had an underlying genetic predisposition associated with an impaired capacity for recovery.”
Dr Gramatges and her colleagues hypothesized that short telomere length could be associated with a delay in neutrophil recovery.
So they obtained bone marrow samples from AML patients who recovered as expected (within 30 days) after each chemotherapy course (n=62), and from AML patients who experienced significant delays in recovery after chemotherapy (n=53).
The team then measured telomere length on each subject and categorized the group by quartile, from shortest to longest.
Subjects in the quartile with the shortest telomere lengths took the longest to recover, especially during the last 2 courses of chemotherapy. In an adjusted analysis, lower telomere content was significantly associated with prolonged neutropenia after the fourth (P=0.002) and fifth courses of chemotherapy (P=0.009).
The researchers said these results support the hypothesis that telomeres are an indicator of capacity for neutrophil recovery following chemotherapy.
Dr Gramatges hopes the results of this study will be helpful in further understanding which children are at a higher risk for prolonged myelosuppression and how to target those children with modified treatments, improved supportive care, and closer monitoring in order to prevent potential complications such as severe infections.
“A significant proportion of children with AML suffer from treatment-related toxicities, with some succumbing to complications of the therapies we give, rather than from the actual cancer itself,” Dr Gramatges said.
“We hope this research will help us identify those who are at a higher risk for delayed recovery and use this knowledge to reduce the morbidity and mortality associated with AML treatment.” ![]()
Hidradenitis Suppurativa Video Roundtable
This 4-part video series is moderated by Jeffrey M. Weinberg, MD, and features discussion among dermatologists on this chronic inflammatory condition that has a significant impact on the quality of life of patients. Moderated by Jeffrey M. Weinberg, MD, this series covers pathogenesis, comorbidities, diagnosis, treatment, and patient education.
This video roundtable was produced by the Custom Programs division of Frontline Medical Communications. The editorial staff of Dermatology News was not involved in developing the video roundtable.
Participants include:
- Robert G. Micheletti, MD
- George Han, MD, PhD
- Mary Ruth Buchness, MD
Disclosure: The faculty received modest honoraria from Frontline Medical Communications for their time participating in this roundtable, and maintained complete editorial control over all content presented.
Dr. Weinberg discloses that he has received honoraria from AbbVie Inc.
Dr. Buchness discloses that she is on the speakers’ bureau for AbbVie Inc.
Dr. Micheletti and Dr. Han have nothing to disclose.
This 4-part video series is moderated by Jeffrey M. Weinberg, MD, and features discussion among dermatologists on this chronic inflammatory condition that has a significant impact on the quality of life of patients. Moderated by Jeffrey M. Weinberg, MD, this series covers pathogenesis, comorbidities, diagnosis, treatment, and patient education.
This video roundtable was produced by the Custom Programs division of Frontline Medical Communications. The editorial staff of Dermatology News was not involved in developing the video roundtable.
Participants include:
- Robert G. Micheletti, MD
- George Han, MD, PhD
- Mary Ruth Buchness, MD
Disclosure: The faculty received modest honoraria from Frontline Medical Communications for their time participating in this roundtable, and maintained complete editorial control over all content presented.
Dr. Weinberg discloses that he has received honoraria from AbbVie Inc.
Dr. Buchness discloses that she is on the speakers’ bureau for AbbVie Inc.
Dr. Micheletti and Dr. Han have nothing to disclose.
This 4-part video series is moderated by Jeffrey M. Weinberg, MD, and features discussion among dermatologists on this chronic inflammatory condition that has a significant impact on the quality of life of patients. Moderated by Jeffrey M. Weinberg, MD, this series covers pathogenesis, comorbidities, diagnosis, treatment, and patient education.
This video roundtable was produced by the Custom Programs division of Frontline Medical Communications. The editorial staff of Dermatology News was not involved in developing the video roundtable.
Participants include:
- Robert G. Micheletti, MD
- George Han, MD, PhD
- Mary Ruth Buchness, MD
Disclosure: The faculty received modest honoraria from Frontline Medical Communications for their time participating in this roundtable, and maintained complete editorial control over all content presented.
Dr. Weinberg discloses that he has received honoraria from AbbVie Inc.
Dr. Buchness discloses that she is on the speakers’ bureau for AbbVie Inc.
Dr. Micheletti and Dr. Han have nothing to disclose.
Supreme Court will not hear pharmacy religious liberty case
The U.S. Supreme Court has refused to decide whether pharmacists with strongly held religious beliefs can be forced to dispense emergency contraception to patients.
Justices did not explain their June 28 denial of Stormans, Inc. vs. Wiesman, but the decision was made over the objection of Chief Justice John G. Roberts Jr., Associate Justice Samuel Alito Jr., and Associate Justice Clarence Thomas. In his dissent, Associate Justice Alito wrote the court should have heard the case to ensure that novel and concededly “unnecessary burden on religious objectors” does not trample fundamental rights.
“If this is a sign of how religious liberty claims will be treated in the years ahead, those who value religious freedom have cause for great concern,” he wrote in his dissent.
At issue in the case is a 2007 rule by Washington state that a family-owned pharmacy in Olympia must provide Plan B contraception to patients. The “delivery rule” creates “a duty for pharmacists to deliver lawfully prescribed drugs or devices in a timely manner and does not allow for conscience-based decisions not to dispense the drug. The Stormans family, who own Ralph’s Thriftway, sued the state over the regulation, alleging violations of the free exercise, equal protection, and due process clauses of the Constitution. The business owners equate emergency contraception to abortion, and they argue that dispensing the medication violates their religious beliefs.
The 9th U.S. Circuit Court of Appeals sided with the state, calling the regulations “neutral and generally applicable.” The plaintiffs appealed to the Supreme Court. The denial by the high court allows the 9th Circuit decision to stand.
Nearly 20 court briefs were issued to the Supreme Court in the case, including briefs by the American Association of Pro-Life Obstetricians and Gynecologists and 4,609 individual health care professionals in support of the pharmacy.
“By effectively prohibiting exemptions for religious reasons, the state of Washington’s regulations depart radically from widely established norms within the health care industry protecting the individual conscience rights of health care professionals,” the health care professionals wrote in their brief. “Such norms favoring the freedom of conscience are particularly well established where, as here, the practitioner’s right to decline care applies to particular treatments, not to individual patients or classes of persons; and where, as here, declining treatment for reasons of religious conscience has no practical impact on quality or availability of care.”
On Twitter @legal_med
The U.S. Supreme Court has refused to decide whether pharmacists with strongly held religious beliefs can be forced to dispense emergency contraception to patients.
Justices did not explain their June 28 denial of Stormans, Inc. vs. Wiesman, but the decision was made over the objection of Chief Justice John G. Roberts Jr., Associate Justice Samuel Alito Jr., and Associate Justice Clarence Thomas. In his dissent, Associate Justice Alito wrote the court should have heard the case to ensure that novel and concededly “unnecessary burden on religious objectors” does not trample fundamental rights.
“If this is a sign of how religious liberty claims will be treated in the years ahead, those who value religious freedom have cause for great concern,” he wrote in his dissent.
At issue in the case is a 2007 rule by Washington state that a family-owned pharmacy in Olympia must provide Plan B contraception to patients. The “delivery rule” creates “a duty for pharmacists to deliver lawfully prescribed drugs or devices in a timely manner and does not allow for conscience-based decisions not to dispense the drug. The Stormans family, who own Ralph’s Thriftway, sued the state over the regulation, alleging violations of the free exercise, equal protection, and due process clauses of the Constitution. The business owners equate emergency contraception to abortion, and they argue that dispensing the medication violates their religious beliefs.
The 9th U.S. Circuit Court of Appeals sided with the state, calling the regulations “neutral and generally applicable.” The plaintiffs appealed to the Supreme Court. The denial by the high court allows the 9th Circuit decision to stand.
Nearly 20 court briefs were issued to the Supreme Court in the case, including briefs by the American Association of Pro-Life Obstetricians and Gynecologists and 4,609 individual health care professionals in support of the pharmacy.
“By effectively prohibiting exemptions for religious reasons, the state of Washington’s regulations depart radically from widely established norms within the health care industry protecting the individual conscience rights of health care professionals,” the health care professionals wrote in their brief. “Such norms favoring the freedom of conscience are particularly well established where, as here, the practitioner’s right to decline care applies to particular treatments, not to individual patients or classes of persons; and where, as here, declining treatment for reasons of religious conscience has no practical impact on quality or availability of care.”
On Twitter @legal_med
The U.S. Supreme Court has refused to decide whether pharmacists with strongly held religious beliefs can be forced to dispense emergency contraception to patients.
Justices did not explain their June 28 denial of Stormans, Inc. vs. Wiesman, but the decision was made over the objection of Chief Justice John G. Roberts Jr., Associate Justice Samuel Alito Jr., and Associate Justice Clarence Thomas. In his dissent, Associate Justice Alito wrote the court should have heard the case to ensure that novel and concededly “unnecessary burden on religious objectors” does not trample fundamental rights.
“If this is a sign of how religious liberty claims will be treated in the years ahead, those who value religious freedom have cause for great concern,” he wrote in his dissent.
At issue in the case is a 2007 rule by Washington state that a family-owned pharmacy in Olympia must provide Plan B contraception to patients. The “delivery rule” creates “a duty for pharmacists to deliver lawfully prescribed drugs or devices in a timely manner and does not allow for conscience-based decisions not to dispense the drug. The Stormans family, who own Ralph’s Thriftway, sued the state over the regulation, alleging violations of the free exercise, equal protection, and due process clauses of the Constitution. The business owners equate emergency contraception to abortion, and they argue that dispensing the medication violates their religious beliefs.
The 9th U.S. Circuit Court of Appeals sided with the state, calling the regulations “neutral and generally applicable.” The plaintiffs appealed to the Supreme Court. The denial by the high court allows the 9th Circuit decision to stand.
Nearly 20 court briefs were issued to the Supreme Court in the case, including briefs by the American Association of Pro-Life Obstetricians and Gynecologists and 4,609 individual health care professionals in support of the pharmacy.
“By effectively prohibiting exemptions for religious reasons, the state of Washington’s regulations depart radically from widely established norms within the health care industry protecting the individual conscience rights of health care professionals,” the health care professionals wrote in their brief. “Such norms favoring the freedom of conscience are particularly well established where, as here, the practitioner’s right to decline care applies to particular treatments, not to individual patients or classes of persons; and where, as here, declining treatment for reasons of religious conscience has no practical impact on quality or availability of care.”
On Twitter @legal_med
Republicans Propose "A Better Way" to Regulate Healthcare
WASHINGTON - U.S. House of Representatives Speaker Paul Ryan unveiled a Republican healthcare agenda on Wednesday that would repeal Obamacare but keep some of its more popular provisions.
The proposal is part of Ryan's blueprint, titled "A Better Way," which offers a Republican alternative to the Democratic Party on policy issues ahead of the Nov. 8 election.
Earlier this month, Ryan, the country's highest-ranking elected Republican, released initiatives on national security and combating poverty. Proposals on regulation, tax reform and constitutional authority are expected in the coming weeks.
Republicans have challenged President Barack Obama's signature healthcare law, the Affordable Care Act, since it was enacted in 2010 after a bitter fight in Congress.
"Obamacare has limited choices for patients, driven up costs for consumers, and buried employers and health care providers under thousands of new regulations," a draft of the Ryan plan said. "This law cannot be fixed."
But Ryan's proposal would keep some popular aspects of the law, including not allowing people with pre-existing conditions to be denied coverage and permitting children to stay on their parents' coverage until age 26.
The Obama administration says some 20 million Americans have become insured as a result of the Affordable Care Act.
The Ryan plan recycles long-held Republican proposals like allowing consumers to buy health insurance across state lines, expanding the use of health savings accounts and giving states block grants to run the Medicaid program for the poor.
For people who do not get insurance through their jobs, the Republican plan would establish a refundable tax credit. Obamacare, by contrast, provides subsidies to some lower-income people to buy insurance if they do not qualify for Medicaid.
The Republican proposal would gradually increase the Medicare eligibility age, which currently is 65, to match that of the Social Security pension plan, which is 67 for people born in 1960 or later.
Like Obamacare's so-called Cadillac tax on expensive healthcare plans offered by employers, the Republican proposal would cap the tax deductibility of employer-based plans.
The Republican plan includes medical liability reform that would put a cap on non-economic damages awarded in lawsuits, a measure aimed at cutting overall healthcare costs.
Under Obamacare, many states expanded the number of people eligible for Medicaid. The Republican plan would allow states that decided to expand Medicaid before this year to keep the expansion, while preventing any new states from doing so.
WASHINGTON - U.S. House of Representatives Speaker Paul Ryan unveiled a Republican healthcare agenda on Wednesday that would repeal Obamacare but keep some of its more popular provisions.
The proposal is part of Ryan's blueprint, titled "A Better Way," which offers a Republican alternative to the Democratic Party on policy issues ahead of the Nov. 8 election.
Earlier this month, Ryan, the country's highest-ranking elected Republican, released initiatives on national security and combating poverty. Proposals on regulation, tax reform and constitutional authority are expected in the coming weeks.
Republicans have challenged President Barack Obama's signature healthcare law, the Affordable Care Act, since it was enacted in 2010 after a bitter fight in Congress.
"Obamacare has limited choices for patients, driven up costs for consumers, and buried employers and health care providers under thousands of new regulations," a draft of the Ryan plan said. "This law cannot be fixed."
But Ryan's proposal would keep some popular aspects of the law, including not allowing people with pre-existing conditions to be denied coverage and permitting children to stay on their parents' coverage until age 26.
The Obama administration says some 20 million Americans have become insured as a result of the Affordable Care Act.
The Ryan plan recycles long-held Republican proposals like allowing consumers to buy health insurance across state lines, expanding the use of health savings accounts and giving states block grants to run the Medicaid program for the poor.
For people who do not get insurance through their jobs, the Republican plan would establish a refundable tax credit. Obamacare, by contrast, provides subsidies to some lower-income people to buy insurance if they do not qualify for Medicaid.
The Republican proposal would gradually increase the Medicare eligibility age, which currently is 65, to match that of the Social Security pension plan, which is 67 for people born in 1960 or later.
Like Obamacare's so-called Cadillac tax on expensive healthcare plans offered by employers, the Republican proposal would cap the tax deductibility of employer-based plans.
The Republican plan includes medical liability reform that would put a cap on non-economic damages awarded in lawsuits, a measure aimed at cutting overall healthcare costs.
Under Obamacare, many states expanded the number of people eligible for Medicaid. The Republican plan would allow states that decided to expand Medicaid before this year to keep the expansion, while preventing any new states from doing so.
WASHINGTON - U.S. House of Representatives Speaker Paul Ryan unveiled a Republican healthcare agenda on Wednesday that would repeal Obamacare but keep some of its more popular provisions.
The proposal is part of Ryan's blueprint, titled "A Better Way," which offers a Republican alternative to the Democratic Party on policy issues ahead of the Nov. 8 election.
Earlier this month, Ryan, the country's highest-ranking elected Republican, released initiatives on national security and combating poverty. Proposals on regulation, tax reform and constitutional authority are expected in the coming weeks.
Republicans have challenged President Barack Obama's signature healthcare law, the Affordable Care Act, since it was enacted in 2010 after a bitter fight in Congress.
"Obamacare has limited choices for patients, driven up costs for consumers, and buried employers and health care providers under thousands of new regulations," a draft of the Ryan plan said. "This law cannot be fixed."
But Ryan's proposal would keep some popular aspects of the law, including not allowing people with pre-existing conditions to be denied coverage and permitting children to stay on their parents' coverage until age 26.
The Obama administration says some 20 million Americans have become insured as a result of the Affordable Care Act.
The Ryan plan recycles long-held Republican proposals like allowing consumers to buy health insurance across state lines, expanding the use of health savings accounts and giving states block grants to run the Medicaid program for the poor.
For people who do not get insurance through their jobs, the Republican plan would establish a refundable tax credit. Obamacare, by contrast, provides subsidies to some lower-income people to buy insurance if they do not qualify for Medicaid.
The Republican proposal would gradually increase the Medicare eligibility age, which currently is 65, to match that of the Social Security pension plan, which is 67 for people born in 1960 or later.
Like Obamacare's so-called Cadillac tax on expensive healthcare plans offered by employers, the Republican proposal would cap the tax deductibility of employer-based plans.
The Republican plan includes medical liability reform that would put a cap on non-economic damages awarded in lawsuits, a measure aimed at cutting overall healthcare costs.
Under Obamacare, many states expanded the number of people eligible for Medicaid. The Republican plan would allow states that decided to expand Medicaid before this year to keep the expansion, while preventing any new states from doing so.
Cisplatin-based chemo may be linked to hearing loss
In male patients with adult-onset germ cell tumors, cisplatin-based chemotherapy may be associated with hearing loss, according to the results of the large, multicenter Platinum Study.
For every 100-mg/m2 increase in cumulative cisplatin dose, a 3.2-dB decline in overall hearing threshold occurred, Robert Frisina, PhD, of the University of South Florida, Tampa, and his associates reported (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2016.66.8822).
A total of 488 men with adult-onset germ cell tumors who were treated with cisplatin-based chemotherapy were consented into this study, and completed questionnaires concerning neurotoxic symptoms, lifestyle habits, and medication use. Each patient underwent bone-conduction and speech-conducting threshold testing. Pure-tone air conduction thresholds were obtained bilaterally at speech frequency range (0.25 to 12 kHz). Classification of hearing loss and assessment of severity followed standardized criteria as defined by the American Speech-Language-Hearing Association. Median age at cancer diagnosis was 31 years; the median interval between chemotherapy and audiometric testing was 4.25 years. Median cumulative cisplatin dose was 400 mg/m2. Increasing cumulative cisplatin dose was associated with increasing (worse) hearing thresholds at 4 kHz (P = .021), 6 kHz (P = .0017), 8 kHz (P less than .001), 10 kHz (P less than .001), and 12 kHz (P = .0013) after correcting for age.
Cumulative cisplatin doses above 300 mg/m2 were associated with more severe hearing loss, compared with doses less than 300 mg/m2 (odds ratio, 1.59; 95% confidence interval, 1.14-2.21; P = .0066).
Conductive hearing loss in the middle ear was not associated with drug exposure dosage levels. Hypertension was identified as a risk factor for hearing loss as impaired overall hearing threshold was significantly associated with hypertension when correcting for age and cisplatin dose (n = 60, P = .0066).
“Because alterations in the highly successful [germ cell tumor] regimens are unlikely, our results point to the importance of ongoing research aimed at the identification of genetic variants associated with cisplatin-related ototoxicity,” investigators wrote. They also suggested that cancer patients treated with cisplatin should be careful to avoid noise exposure, ototoxic drugs, and other factors that could further increase damage.
This study was funded by the National Cancer Institute. Dr. Frisina reported holding patents related to hearing loss products. Six other investigators reported serving in advisory roles, receiving financial compensation or honoraria from multiple pharmaceutical and biomedical companies.
On Twitter @jessnicolecraig
In male patients with adult-onset germ cell tumors, cisplatin-based chemotherapy may be associated with hearing loss, according to the results of the large, multicenter Platinum Study.
For every 100-mg/m2 increase in cumulative cisplatin dose, a 3.2-dB decline in overall hearing threshold occurred, Robert Frisina, PhD, of the University of South Florida, Tampa, and his associates reported (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2016.66.8822).
A total of 488 men with adult-onset germ cell tumors who were treated with cisplatin-based chemotherapy were consented into this study, and completed questionnaires concerning neurotoxic symptoms, lifestyle habits, and medication use. Each patient underwent bone-conduction and speech-conducting threshold testing. Pure-tone air conduction thresholds were obtained bilaterally at speech frequency range (0.25 to 12 kHz). Classification of hearing loss and assessment of severity followed standardized criteria as defined by the American Speech-Language-Hearing Association. Median age at cancer diagnosis was 31 years; the median interval between chemotherapy and audiometric testing was 4.25 years. Median cumulative cisplatin dose was 400 mg/m2. Increasing cumulative cisplatin dose was associated with increasing (worse) hearing thresholds at 4 kHz (P = .021), 6 kHz (P = .0017), 8 kHz (P less than .001), 10 kHz (P less than .001), and 12 kHz (P = .0013) after correcting for age.
Cumulative cisplatin doses above 300 mg/m2 were associated with more severe hearing loss, compared with doses less than 300 mg/m2 (odds ratio, 1.59; 95% confidence interval, 1.14-2.21; P = .0066).
Conductive hearing loss in the middle ear was not associated with drug exposure dosage levels. Hypertension was identified as a risk factor for hearing loss as impaired overall hearing threshold was significantly associated with hypertension when correcting for age and cisplatin dose (n = 60, P = .0066).
“Because alterations in the highly successful [germ cell tumor] regimens are unlikely, our results point to the importance of ongoing research aimed at the identification of genetic variants associated with cisplatin-related ototoxicity,” investigators wrote. They also suggested that cancer patients treated with cisplatin should be careful to avoid noise exposure, ototoxic drugs, and other factors that could further increase damage.
This study was funded by the National Cancer Institute. Dr. Frisina reported holding patents related to hearing loss products. Six other investigators reported serving in advisory roles, receiving financial compensation or honoraria from multiple pharmaceutical and biomedical companies.
On Twitter @jessnicolecraig
In male patients with adult-onset germ cell tumors, cisplatin-based chemotherapy may be associated with hearing loss, according to the results of the large, multicenter Platinum Study.
For every 100-mg/m2 increase in cumulative cisplatin dose, a 3.2-dB decline in overall hearing threshold occurred, Robert Frisina, PhD, of the University of South Florida, Tampa, and his associates reported (J Clin Oncol. 2016 Jun. doi: 10.1200/JCO.2016.66.8822).
A total of 488 men with adult-onset germ cell tumors who were treated with cisplatin-based chemotherapy were consented into this study, and completed questionnaires concerning neurotoxic symptoms, lifestyle habits, and medication use. Each patient underwent bone-conduction and speech-conducting threshold testing. Pure-tone air conduction thresholds were obtained bilaterally at speech frequency range (0.25 to 12 kHz). Classification of hearing loss and assessment of severity followed standardized criteria as defined by the American Speech-Language-Hearing Association. Median age at cancer diagnosis was 31 years; the median interval between chemotherapy and audiometric testing was 4.25 years. Median cumulative cisplatin dose was 400 mg/m2. Increasing cumulative cisplatin dose was associated with increasing (worse) hearing thresholds at 4 kHz (P = .021), 6 kHz (P = .0017), 8 kHz (P less than .001), 10 kHz (P less than .001), and 12 kHz (P = .0013) after correcting for age.
Cumulative cisplatin doses above 300 mg/m2 were associated with more severe hearing loss, compared with doses less than 300 mg/m2 (odds ratio, 1.59; 95% confidence interval, 1.14-2.21; P = .0066).
Conductive hearing loss in the middle ear was not associated with drug exposure dosage levels. Hypertension was identified as a risk factor for hearing loss as impaired overall hearing threshold was significantly associated with hypertension when correcting for age and cisplatin dose (n = 60, P = .0066).
“Because alterations in the highly successful [germ cell tumor] regimens are unlikely, our results point to the importance of ongoing research aimed at the identification of genetic variants associated with cisplatin-related ototoxicity,” investigators wrote. They also suggested that cancer patients treated with cisplatin should be careful to avoid noise exposure, ototoxic drugs, and other factors that could further increase damage.
This study was funded by the National Cancer Institute. Dr. Frisina reported holding patents related to hearing loss products. Six other investigators reported serving in advisory roles, receiving financial compensation or honoraria from multiple pharmaceutical and biomedical companies.
On Twitter @jessnicolecraig
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: In male patients with adult-onset germ cell tumors, cisplatin-based chemotherapy may be associated with hearing loss.
Major finding: For every 100-mg/m2 increase in cumulative cisplatin dose, a 3.2-dB decline in overall hearing threshold occurred.
Data source: A multicenter study of 488 men with adult-onset germ cell tumors.
Disclosures: This study was funded by the National Cancer Institute. Dr. Frisina reported holding patents related to hearing loss. Six other investigators reported serving in advisory roles, receiving financial compensation or honoraria from multiple pharmaceutical and biomedical companies.
Sticks and stones and words
“Sticks and stones may break my bones, but words will never hurt me.” This mantra was the retort of choice for thousands of apparently resilient premillennial children. But you and I, and just about everyone else, know that words can be very hurtful. A recent article in the journal Eating and Weight Disorders entitled, “ ‘Don’t eat so much’: How parent comments relate to female weight satisfaction” (Eat Weight Disord. 2016 Jun 6. [Epub ahead of print]) reminds us that the pain can last forever.
In a retrospective study of 501 young women aged 20-35 years, the investigators asked whether the women could recall their parents making any comments about their weight when they were young children. What the authors discovered was that even among young women who were of normal weight, those who could recall their parents making a comment about their weight were more dissatisfied with their body weight than the young women who could not recall such a comment. However, if the comment had been about eating habits and not weight, then there was no significant association with weight dissatisfaction.
Before we rush out to send all of the parents of weight-dissatisfied young women on a guilt trip, let’s remember that this was a retrospective study. Let’s consider the not unlikely explanation that there may be something built into the psyche of weight-dissatisfied young women that sharpens their memory for negative comments from friends and family.
Regardless of how we interpret the findings from this study, it is probably safe to say that telling a young girl that she is overweight doesn’t help and should be avoided. This is just another example of how poorly chosen words can be hurtful. But it is also an example of how words alone are seldom shapers of positive behaviors. You can’t talk a picky eater into eating spinach anymore than you can talk the child in the middle of a tantrum into settling down. Good manners are best learned by modeling the behavior of respected adults and not by being subjected to a series of parental lectures. Telling a child she is overweight won’t solve the problem.
So what is the parent of an obese child to do? Unfortunately, many parents of obese children don’t perceive their child as being significantly overweight. But let’s assume we have cleared that hurdle of denial. If telling the child she is overweight is the wrong thing to do, then her parents are forced into using strategies that are subliminal, applied slowly and patiently – silently.
These strategies could include gradually decreasing the child’s screen time, hoping that it will be replaced by calorie-burning activities; changing the food available for all the inhabitants of the home to increase the likelihood that healthier choices will dominate; and decreasing serving sizes. It is critical that these changes are done so slowly that they go unnoticed by the child. If the child questions the changes, then the response should be that they are being done to help the entire family to be healthier, and that they are not being targeted at any one individual. Of course, the big problem is getting the rest of the family to buy into the changes so that the overweight child doesn’t become a scapegoat.
Shielding the overweight child from the blame game is much easier if the parents have been careful to avoid labeling from the moment they realized or accepted that the child had a weight problem. Here is where we pediatricians can play a critical role in our choice of words, and the setting in which we discuss the child’s weight with the parents. We must point out to the parents that their words can create a hurt that may not ever go away.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
“Sticks and stones may break my bones, but words will never hurt me.” This mantra was the retort of choice for thousands of apparently resilient premillennial children. But you and I, and just about everyone else, know that words can be very hurtful. A recent article in the journal Eating and Weight Disorders entitled, “ ‘Don’t eat so much’: How parent comments relate to female weight satisfaction” (Eat Weight Disord. 2016 Jun 6. [Epub ahead of print]) reminds us that the pain can last forever.
In a retrospective study of 501 young women aged 20-35 years, the investigators asked whether the women could recall their parents making any comments about their weight when they were young children. What the authors discovered was that even among young women who were of normal weight, those who could recall their parents making a comment about their weight were more dissatisfied with their body weight than the young women who could not recall such a comment. However, if the comment had been about eating habits and not weight, then there was no significant association with weight dissatisfaction.
Before we rush out to send all of the parents of weight-dissatisfied young women on a guilt trip, let’s remember that this was a retrospective study. Let’s consider the not unlikely explanation that there may be something built into the psyche of weight-dissatisfied young women that sharpens their memory for negative comments from friends and family.
Regardless of how we interpret the findings from this study, it is probably safe to say that telling a young girl that she is overweight doesn’t help and should be avoided. This is just another example of how poorly chosen words can be hurtful. But it is also an example of how words alone are seldom shapers of positive behaviors. You can’t talk a picky eater into eating spinach anymore than you can talk the child in the middle of a tantrum into settling down. Good manners are best learned by modeling the behavior of respected adults and not by being subjected to a series of parental lectures. Telling a child she is overweight won’t solve the problem.
So what is the parent of an obese child to do? Unfortunately, many parents of obese children don’t perceive their child as being significantly overweight. But let’s assume we have cleared that hurdle of denial. If telling the child she is overweight is the wrong thing to do, then her parents are forced into using strategies that are subliminal, applied slowly and patiently – silently.
These strategies could include gradually decreasing the child’s screen time, hoping that it will be replaced by calorie-burning activities; changing the food available for all the inhabitants of the home to increase the likelihood that healthier choices will dominate; and decreasing serving sizes. It is critical that these changes are done so slowly that they go unnoticed by the child. If the child questions the changes, then the response should be that they are being done to help the entire family to be healthier, and that they are not being targeted at any one individual. Of course, the big problem is getting the rest of the family to buy into the changes so that the overweight child doesn’t become a scapegoat.
Shielding the overweight child from the blame game is much easier if the parents have been careful to avoid labeling from the moment they realized or accepted that the child had a weight problem. Here is where we pediatricians can play a critical role in our choice of words, and the setting in which we discuss the child’s weight with the parents. We must point out to the parents that their words can create a hurt that may not ever go away.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
“Sticks and stones may break my bones, but words will never hurt me.” This mantra was the retort of choice for thousands of apparently resilient premillennial children. But you and I, and just about everyone else, know that words can be very hurtful. A recent article in the journal Eating and Weight Disorders entitled, “ ‘Don’t eat so much’: How parent comments relate to female weight satisfaction” (Eat Weight Disord. 2016 Jun 6. [Epub ahead of print]) reminds us that the pain can last forever.
In a retrospective study of 501 young women aged 20-35 years, the investigators asked whether the women could recall their parents making any comments about their weight when they were young children. What the authors discovered was that even among young women who were of normal weight, those who could recall their parents making a comment about their weight were more dissatisfied with their body weight than the young women who could not recall such a comment. However, if the comment had been about eating habits and not weight, then there was no significant association with weight dissatisfaction.
Before we rush out to send all of the parents of weight-dissatisfied young women on a guilt trip, let’s remember that this was a retrospective study. Let’s consider the not unlikely explanation that there may be something built into the psyche of weight-dissatisfied young women that sharpens their memory for negative comments from friends and family.
Regardless of how we interpret the findings from this study, it is probably safe to say that telling a young girl that she is overweight doesn’t help and should be avoided. This is just another example of how poorly chosen words can be hurtful. But it is also an example of how words alone are seldom shapers of positive behaviors. You can’t talk a picky eater into eating spinach anymore than you can talk the child in the middle of a tantrum into settling down. Good manners are best learned by modeling the behavior of respected adults and not by being subjected to a series of parental lectures. Telling a child she is overweight won’t solve the problem.
So what is the parent of an obese child to do? Unfortunately, many parents of obese children don’t perceive their child as being significantly overweight. But let’s assume we have cleared that hurdle of denial. If telling the child she is overweight is the wrong thing to do, then her parents are forced into using strategies that are subliminal, applied slowly and patiently – silently.
These strategies could include gradually decreasing the child’s screen time, hoping that it will be replaced by calorie-burning activities; changing the food available for all the inhabitants of the home to increase the likelihood that healthier choices will dominate; and decreasing serving sizes. It is critical that these changes are done so slowly that they go unnoticed by the child. If the child questions the changes, then the response should be that they are being done to help the entire family to be healthier, and that they are not being targeted at any one individual. Of course, the big problem is getting the rest of the family to buy into the changes so that the overweight child doesn’t become a scapegoat.
Shielding the overweight child from the blame game is much easier if the parents have been careful to avoid labeling from the moment they realized or accepted that the child had a weight problem. Here is where we pediatricians can play a critical role in our choice of words, and the setting in which we discuss the child’s weight with the parents. We must point out to the parents that their words can create a hurt that may not ever go away.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”