User login
History of Sexual Abuse May Affect Risk for Chronic Migraine
VALENCIA, SPAIN—A history of childhood sexual abuse is nearly fourfold more common among patients with chronic migraine than in those with episodic migraine, according to research presented at the International Headache Congress. This association raises the possibility that prior sexual abuse is a contributing factor in the transformation from episodic migraine to chronic migraine, said Brad Torphy, MD, of Diamond Headache Clinic in Chicago.
“The clinical implication of these findings, and what I’m stressing, is the importance of intervention—such as psychological counseling—in episodic migraine patients who have a history of abuse,” he said in an interview. “The other key point is that patients who have episodic migraine may warrant more aggressive therapy, including going on preventive medications sooner, if they have a history of sexual abuse, because they’re probably at risk for progression to chronic migraine.”
Dr. Torphy presented a retrospective study of all new patients seen at the Diamond Headache Clinic during the final four months of last year. The new-patient questionnaire includes a section about past sexual abuse. Such a history was reported by six (4.4%) of the 135 patients with episodic migraine, compared with 30 (15.5%) of the 194 patients with chronic migraine.
Based upon his experience in the clinic, Dr. Torphy suspects that the true proportion of patients with a positive history for sexual abuse is considerably higher than the rates the new-patient questionnaire would suggest.
“A lot of factors would lead to that being a very low number,” Dr. Torphy continued. “It’s the patient’s first visit, and it’s a paper questionnaire, so patients may not be comfortable checking that box when they don’t know who’s going to see the results. I’ve had cases where patients shared with me only after two or three visits that, yes, I do have that history. I think it’s underreported across the board.”
In his review of the literature, he found that other investigators have tended either to lump together all kinds of abuse—physical, emotional, and sexual—in analyzing an association with migraine, or if they looked at sexual abuse in particular, it was in association with all types of chronic pain, not specifically migraine.
—Bruce Jancin
VALENCIA, SPAIN—A history of childhood sexual abuse is nearly fourfold more common among patients with chronic migraine than in those with episodic migraine, according to research presented at the International Headache Congress. This association raises the possibility that prior sexual abuse is a contributing factor in the transformation from episodic migraine to chronic migraine, said Brad Torphy, MD, of Diamond Headache Clinic in Chicago.
“The clinical implication of these findings, and what I’m stressing, is the importance of intervention—such as psychological counseling—in episodic migraine patients who have a history of abuse,” he said in an interview. “The other key point is that patients who have episodic migraine may warrant more aggressive therapy, including going on preventive medications sooner, if they have a history of sexual abuse, because they’re probably at risk for progression to chronic migraine.”
Dr. Torphy presented a retrospective study of all new patients seen at the Diamond Headache Clinic during the final four months of last year. The new-patient questionnaire includes a section about past sexual abuse. Such a history was reported by six (4.4%) of the 135 patients with episodic migraine, compared with 30 (15.5%) of the 194 patients with chronic migraine.
Based upon his experience in the clinic, Dr. Torphy suspects that the true proportion of patients with a positive history for sexual abuse is considerably higher than the rates the new-patient questionnaire would suggest.
“A lot of factors would lead to that being a very low number,” Dr. Torphy continued. “It’s the patient’s first visit, and it’s a paper questionnaire, so patients may not be comfortable checking that box when they don’t know who’s going to see the results. I’ve had cases where patients shared with me only after two or three visits that, yes, I do have that history. I think it’s underreported across the board.”
In his review of the literature, he found that other investigators have tended either to lump together all kinds of abuse—physical, emotional, and sexual—in analyzing an association with migraine, or if they looked at sexual abuse in particular, it was in association with all types of chronic pain, not specifically migraine.
—Bruce Jancin
VALENCIA, SPAIN—A history of childhood sexual abuse is nearly fourfold more common among patients with chronic migraine than in those with episodic migraine, according to research presented at the International Headache Congress. This association raises the possibility that prior sexual abuse is a contributing factor in the transformation from episodic migraine to chronic migraine, said Brad Torphy, MD, of Diamond Headache Clinic in Chicago.
“The clinical implication of these findings, and what I’m stressing, is the importance of intervention—such as psychological counseling—in episodic migraine patients who have a history of abuse,” he said in an interview. “The other key point is that patients who have episodic migraine may warrant more aggressive therapy, including going on preventive medications sooner, if they have a history of sexual abuse, because they’re probably at risk for progression to chronic migraine.”
Dr. Torphy presented a retrospective study of all new patients seen at the Diamond Headache Clinic during the final four months of last year. The new-patient questionnaire includes a section about past sexual abuse. Such a history was reported by six (4.4%) of the 135 patients with episodic migraine, compared with 30 (15.5%) of the 194 patients with chronic migraine.
Based upon his experience in the clinic, Dr. Torphy suspects that the true proportion of patients with a positive history for sexual abuse is considerably higher than the rates the new-patient questionnaire would suggest.
“A lot of factors would lead to that being a very low number,” Dr. Torphy continued. “It’s the patient’s first visit, and it’s a paper questionnaire, so patients may not be comfortable checking that box when they don’t know who’s going to see the results. I’ve had cases where patients shared with me only after two or three visits that, yes, I do have that history. I think it’s underreported across the board.”
In his review of the literature, he found that other investigators have tended either to lump together all kinds of abuse—physical, emotional, and sexual—in analyzing an association with migraine, or if they looked at sexual abuse in particular, it was in association with all types of chronic pain, not specifically migraine.
—Bruce Jancin
A New Biomarker for Episodic Migraine in Women?
Sphingolipid metabolism is altered in women with episodic migraine, according to a study published online ahead of print September 9 in Neurology. According to the study authors, serum sphingolipid panels may have the potential to differentiate episodic migraine presence and absence. “While more research is needed to confirm these initial findings, the possibility of discovering a new biomarker for migraine is exciting,” said lead author B. Lee Peterlin, DO, Director of Johns Hopkins Headache Research and Associate Professor of Neurology at Johns Hopkins University School of Medicine in Baltimore.
Case–Control Study
In this study, 88 female participants, 52 with episodic migraine and 36 without, were evaluated on the basis of demographic and health-related criteria, including marital status, BMI, and neuronal functioning. The patients also submitted blood samples, which were tested for lipid concentrations among other things.
The findings suggest that migraineurs had a decreased de novo synthesis of ceramides, which, paired with an independent downstream increase in the conversion of ceramide metabolic products, resulted in a major deficit. Women with migraines had an average concentration of ceramide levels of 6,000 ng/mL, a 43% decrease when compared with controls, who had an average of 10,500 ng/mL of ceramide in their blood.
There was also a marked difference in sphingolipid concentrations in those with migraines, with some species increasing while others were decreased. Dr. Peterlin and colleagues used the difference in blood lipid levels to create a list of 10 sphingolipids thought to classify episodic migraine. In a later test, this constructed biomarker evaluative list was 100% effective in identifying women with and without episodic migraine for a small group of eight migraineurs and six controls.
“This study is an important contribution to our understanding of the pathophysiology of migraine and may have vast practical, clinical, and therapeutic implications if it is supported by further studies,” said Karl Ekbom, PhD, a neurologist at the Karolinska Institutet in Stockholm, in an accompanying commentary.
The study suggests that the presence of migraines is a neurologic disorder of sphingolipid dsymetabolism. With the positive identification of these biomarkers comes the potential of targeted drug therapies directed against the specific sphingolipid pathways involved.
—Adaeze Stephanie Onyechi
Suggested Reading
Peterlin BL, Mielke MM, Dickens AM, et al. Interictal, circulating sphingolipids in women with episodic migraine. Neurology. 2015 Sept 9 [Epub ahead of print].
Ekbom K. Comment: Altered sphingolipid metabolism—a marker for episodic migraine? Neurology. 2015 Sept 9 [Epub ahead of print].
Sphingolipid metabolism is altered in women with episodic migraine, according to a study published online ahead of print September 9 in Neurology. According to the study authors, serum sphingolipid panels may have the potential to differentiate episodic migraine presence and absence. “While more research is needed to confirm these initial findings, the possibility of discovering a new biomarker for migraine is exciting,” said lead author B. Lee Peterlin, DO, Director of Johns Hopkins Headache Research and Associate Professor of Neurology at Johns Hopkins University School of Medicine in Baltimore.
Case–Control Study
In this study, 88 female participants, 52 with episodic migraine and 36 without, were evaluated on the basis of demographic and health-related criteria, including marital status, BMI, and neuronal functioning. The patients also submitted blood samples, which were tested for lipid concentrations among other things.
The findings suggest that migraineurs had a decreased de novo synthesis of ceramides, which, paired with an independent downstream increase in the conversion of ceramide metabolic products, resulted in a major deficit. Women with migraines had an average concentration of ceramide levels of 6,000 ng/mL, a 43% decrease when compared with controls, who had an average of 10,500 ng/mL of ceramide in their blood.
There was also a marked difference in sphingolipid concentrations in those with migraines, with some species increasing while others were decreased. Dr. Peterlin and colleagues used the difference in blood lipid levels to create a list of 10 sphingolipids thought to classify episodic migraine. In a later test, this constructed biomarker evaluative list was 100% effective in identifying women with and without episodic migraine for a small group of eight migraineurs and six controls.
“This study is an important contribution to our understanding of the pathophysiology of migraine and may have vast practical, clinical, and therapeutic implications if it is supported by further studies,” said Karl Ekbom, PhD, a neurologist at the Karolinska Institutet in Stockholm, in an accompanying commentary.
The study suggests that the presence of migraines is a neurologic disorder of sphingolipid dsymetabolism. With the positive identification of these biomarkers comes the potential of targeted drug therapies directed against the specific sphingolipid pathways involved.
—Adaeze Stephanie Onyechi
Sphingolipid metabolism is altered in women with episodic migraine, according to a study published online ahead of print September 9 in Neurology. According to the study authors, serum sphingolipid panels may have the potential to differentiate episodic migraine presence and absence. “While more research is needed to confirm these initial findings, the possibility of discovering a new biomarker for migraine is exciting,” said lead author B. Lee Peterlin, DO, Director of Johns Hopkins Headache Research and Associate Professor of Neurology at Johns Hopkins University School of Medicine in Baltimore.
Case–Control Study
In this study, 88 female participants, 52 with episodic migraine and 36 without, were evaluated on the basis of demographic and health-related criteria, including marital status, BMI, and neuronal functioning. The patients also submitted blood samples, which were tested for lipid concentrations among other things.
The findings suggest that migraineurs had a decreased de novo synthesis of ceramides, which, paired with an independent downstream increase in the conversion of ceramide metabolic products, resulted in a major deficit. Women with migraines had an average concentration of ceramide levels of 6,000 ng/mL, a 43% decrease when compared with controls, who had an average of 10,500 ng/mL of ceramide in their blood.
There was also a marked difference in sphingolipid concentrations in those with migraines, with some species increasing while others were decreased. Dr. Peterlin and colleagues used the difference in blood lipid levels to create a list of 10 sphingolipids thought to classify episodic migraine. In a later test, this constructed biomarker evaluative list was 100% effective in identifying women with and without episodic migraine for a small group of eight migraineurs and six controls.
“This study is an important contribution to our understanding of the pathophysiology of migraine and may have vast practical, clinical, and therapeutic implications if it is supported by further studies,” said Karl Ekbom, PhD, a neurologist at the Karolinska Institutet in Stockholm, in an accompanying commentary.
The study suggests that the presence of migraines is a neurologic disorder of sphingolipid dsymetabolism. With the positive identification of these biomarkers comes the potential of targeted drug therapies directed against the specific sphingolipid pathways involved.
—Adaeze Stephanie Onyechi
Suggested Reading
Peterlin BL, Mielke MM, Dickens AM, et al. Interictal, circulating sphingolipids in women with episodic migraine. Neurology. 2015 Sept 9 [Epub ahead of print].
Ekbom K. Comment: Altered sphingolipid metabolism—a marker for episodic migraine? Neurology. 2015 Sept 9 [Epub ahead of print].
Suggested Reading
Peterlin BL, Mielke MM, Dickens AM, et al. Interictal, circulating sphingolipids in women with episodic migraine. Neurology. 2015 Sept 9 [Epub ahead of print].
Ekbom K. Comment: Altered sphingolipid metabolism—a marker for episodic migraine? Neurology. 2015 Sept 9 [Epub ahead of print].
Sacubitril-valsartan and the evolution of heart failure care
Three decades ago, the only drugs we had for treating chronic heart failure were digitalis and loop diuretics. The mortality rate was very high, and heart transplantation was a newly developing treatment that could help only a very few patients.
The early 80s heralded new hope for patients with heart failure, with the introduction of angiotensin-converting enzyme (ACE) inhibitors1–5 and, later, beta-blockers. Beta-blockers were considered contraindicated in heart failure until new trials provided evidence of dramatic benefit such as better quality of life and longer survival.6–8 ACE inhibitors, along with beta-blockers, quickly became the standard of care for all patients with systolic heart failure.
The implantable cardioverter-defibrillator (ICD) required numerous clinical trials in ischemic and nonischemic cardiomyopathy to define its role.9,10 Cardiac resynchronization therapy did not arrive until 15 years ago and is now indicated in a specific niche of patients with left bundle branch block.11,12 Mineralocorticoid antagonists required three pivotal clinical trials before their important role in the treatment of systolic heart failure was defined.13–16
And in the current decade, the roles of ACE inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, mineralocorticoid antagonists, ICDs, and cardiac resynchronization therapy have been further defined, as reflected in the latest guidelines for the treatment of systolic heart failure.17
Guideline-directed medical therapy for systolic heart failure with the agents and devices mentioned above improves quality of life and extends survival. It was therefore hard to imagine that any new additive therapy could offer significant incremental improvement. However, more than 5 years ago, in an ambitious effort, the largest global clinical trial ever performed in chronic heart failure was launched with a novel agent.18
THE PARADIGM-HF TRIAL
In this issue of the Journal, Sabe et al19 describe the results of the Prospective Comparison of ARNI With ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial of the novel combination drug sacubitril-valsartan, designated LCZ696 during its development and now available as Entresto.20
The mean age of the 8,442 patients in PARADIGM-HF was 64, and 78% were men. Despite guideline-directed medical therapy (93% of the patients were receiving a beta-blocker, and 60% were receiving a mineralocorticoid receptor antagonist), patients had persistent symptoms and signs of heart failure, diminished health-related quality of life, reduced ejection fraction (mean 29%), and elevated n-terminal pro-B-type natriuretic peptide levels (median 1,608 pg/mL, interquartile range 886–3,221).
The investigators reported a remarkable 20% reduction in the primary outcome of death from cardiovascular causes or hospitalization for heart failure in the patients who received sacubitril-valsartan compared with enalapril.20
Sacubitril-valsartan was reviewed under a US Food and Drug Administration (FDA) program that provides expedited review of drugs that are intended to treat a serious disease or condition and that may provide a significant improvement over available therapy. It was also granted a fast-track designation, which supports FDA efforts to facilitate the development and expedite the review of drugs to treat serious and life-threatening conditions and fill an unmet medical need. The FDA approved sacubitril-valsartan on July 7, 2015, for use in place of an ACE inhibitor or ARB in patients with New York Heart Association class II, III, or IV heart failure with reduced ejection fraction.21
WHAT WE STILL NEED TO KNOW
The results of PARADIGM-HF are generalizable, and sacubitril-valsartan was well tolerated in patients whose blood pressure was acceptable and who were able to tolerate ACE inhibitors in target doses. More than 90% of patients were receiving a beta-blocker. The dosing of enalapril (target 10 mg twice a day) is the guideline-directed target dose, and ACE inhibition is considered the gold standard for heart failure with reduced ejection fraction. Sacubitril-valsartan vs enalapril was a very appropriate comparison.
Far fewer PARADIGM-HF patients outside the United States had an ICD than those in the United States, which is a common finding in global clinical trials. However, Desai et al reported that sacubitril-valsartan reduced rates of cardiovascular mortality both from worsening heart failure and from sudden cardiac death, independent of whether the patient had an ICD.22
Sacubitril-valsartan is taken twice a day, but most heart failure patients already take medications at several times during the day, so this should not pose a problem.
More information is needed on the use of this new drug in patients with New York Heart Association class IV symptoms, as only 60 patients with class IV symptoms were included in the PARADIGM-HF trial. Also, the efficacy of the drug in patients unable to tolerate a full dose will need to be analyzed.
PARADIGM-HF was conducted in stable, nonhospitalized patients with chronic heart failure; the use of the drug in new-onset heart failure and its initiation in hospitalized patients will require further study. In addition, the PARAGON-HF trial23 will examine the efficacy of sacubitril-valsartan in patients with heart failure and an ejection fraction of 45% or higher.
Sacubitril-valsartan ushers in a new era in heart failure treatment for patients with reduced ejection fraction and will certainly prompt quick revision of heart failure guidelines.
- Captopril Multicenter Research Group. A placebo-controlled trial of captopril in refractory chronic congestive heart failure. J Am Coll Cardiol 1983; 2:755–763.
- Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987; 316:1429–1435.
- The SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fraction and congestive heart failure. N Engl J Med 1991; 325:293–302.
- Cohn JN, Johnson G, Ziesche S, et al. A comparison of enalapril with hydralazine-isosorbide dinitrate in the treatment of chronic congestive heart failure. N Engl J Med 1991; 325:303–310.
- Pfeffer MA, Braunwald E, Moyé LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 1992; 327:669–677.
- Packer M, Coats AJ, Fowler MB, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med 2001; 344:1651–1658.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999; 353:2001–2007.
- Brophy JM, Joseph L, Rouleau JL. Beta-blockers in congestive heart failure. A Bayesian meta-analysis. Ann Intern Med 2001; 134:550–560.
- Buxton AE, Lee KL, Fisher JD, et al. A randomized study of the prevention of sudden death in patients with coronary artery disease. Multicenter Unsustained Tachycardia Trial Investigators. N Engl J Med 1999; 341:1882–1890.
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med 2002; 346:877–883.
- Abraham WT, Fisher WG, Smith AL, et al; MIRACLE Study Group. Multicenter InSync Randomized Clinical Evaluation. Cardiac resynchronization in chronic heart failure. N Engl J Med 2002; 346:1845–1853.
- McAlister FA, Ezekowitz J, Hooton N, et al. Cardiac resynchronization therapy for patients with left ventricular systolic dysfunction: a systematic review. JAMA 2007; 297:2502–2514.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:709–717.
- Pitt B, Remme W, Zannad F, et al; Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 2003; 348:1309–1321.
- Pitt B, White H, Nicolau J, et al; EPHESUS Investigators. Eplerenone reduces mortality 30 days after randomization following acute myocardial infarction in patients with left ventricular systolic dysfunction and heart failure. J Am Coll Cardiol 2005; 46:425–431.
- Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011; 364:11–21.
- Yancy CW, Jessup M, Bozkurt B, et al; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 62:e147–e239.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Committees and Investigators. Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Fail 2013; 15:1062–1073.
- Sabe IA, Jacob MS, Taylor DO. A new class of drugs for systolic heart failure: The PARADIGM-HF study. Cleve Clin J Med 2015; 82:693–701.
- McMurray JJ, Packer M, Desai AS, Gong J, et al; PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014; 371:993–1004.
- US Food and Drug Administration. FDA approves new drug to treat heart failure. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm453845.htm. Accessed September 2, 2015.
- Desai AS, McMurray JJ, Packer M, et al. Effect of the angiotensin-receptor-neprilysin inhibitor LCZ696 compared with enalapril on mode of death in heart failure patients. Eur Heart J 2015; 36:1990–1997.
- ClinicalTrials.gov. Efficacy and Safety of LCZ696 Compared to Valsartan, on Morbidity and Mortality in Heart Failure Patients With Preserved Ejection Fraction (PARAGON-HF). https://clinicaltrials.gov/ct2/show/NCT01920711. Accessed September 2, 2015.
Three decades ago, the only drugs we had for treating chronic heart failure were digitalis and loop diuretics. The mortality rate was very high, and heart transplantation was a newly developing treatment that could help only a very few patients.
The early 80s heralded new hope for patients with heart failure, with the introduction of angiotensin-converting enzyme (ACE) inhibitors1–5 and, later, beta-blockers. Beta-blockers were considered contraindicated in heart failure until new trials provided evidence of dramatic benefit such as better quality of life and longer survival.6–8 ACE inhibitors, along with beta-blockers, quickly became the standard of care for all patients with systolic heart failure.
The implantable cardioverter-defibrillator (ICD) required numerous clinical trials in ischemic and nonischemic cardiomyopathy to define its role.9,10 Cardiac resynchronization therapy did not arrive until 15 years ago and is now indicated in a specific niche of patients with left bundle branch block.11,12 Mineralocorticoid antagonists required three pivotal clinical trials before their important role in the treatment of systolic heart failure was defined.13–16
And in the current decade, the roles of ACE inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, mineralocorticoid antagonists, ICDs, and cardiac resynchronization therapy have been further defined, as reflected in the latest guidelines for the treatment of systolic heart failure.17
Guideline-directed medical therapy for systolic heart failure with the agents and devices mentioned above improves quality of life and extends survival. It was therefore hard to imagine that any new additive therapy could offer significant incremental improvement. However, more than 5 years ago, in an ambitious effort, the largest global clinical trial ever performed in chronic heart failure was launched with a novel agent.18
THE PARADIGM-HF TRIAL
In this issue of the Journal, Sabe et al19 describe the results of the Prospective Comparison of ARNI With ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial of the novel combination drug sacubitril-valsartan, designated LCZ696 during its development and now available as Entresto.20
The mean age of the 8,442 patients in PARADIGM-HF was 64, and 78% were men. Despite guideline-directed medical therapy (93% of the patients were receiving a beta-blocker, and 60% were receiving a mineralocorticoid receptor antagonist), patients had persistent symptoms and signs of heart failure, diminished health-related quality of life, reduced ejection fraction (mean 29%), and elevated n-terminal pro-B-type natriuretic peptide levels (median 1,608 pg/mL, interquartile range 886–3,221).
The investigators reported a remarkable 20% reduction in the primary outcome of death from cardiovascular causes or hospitalization for heart failure in the patients who received sacubitril-valsartan compared with enalapril.20
Sacubitril-valsartan was reviewed under a US Food and Drug Administration (FDA) program that provides expedited review of drugs that are intended to treat a serious disease or condition and that may provide a significant improvement over available therapy. It was also granted a fast-track designation, which supports FDA efforts to facilitate the development and expedite the review of drugs to treat serious and life-threatening conditions and fill an unmet medical need. The FDA approved sacubitril-valsartan on July 7, 2015, for use in place of an ACE inhibitor or ARB in patients with New York Heart Association class II, III, or IV heart failure with reduced ejection fraction.21
WHAT WE STILL NEED TO KNOW
The results of PARADIGM-HF are generalizable, and sacubitril-valsartan was well tolerated in patients whose blood pressure was acceptable and who were able to tolerate ACE inhibitors in target doses. More than 90% of patients were receiving a beta-blocker. The dosing of enalapril (target 10 mg twice a day) is the guideline-directed target dose, and ACE inhibition is considered the gold standard for heart failure with reduced ejection fraction. Sacubitril-valsartan vs enalapril was a very appropriate comparison.
Far fewer PARADIGM-HF patients outside the United States had an ICD than those in the United States, which is a common finding in global clinical trials. However, Desai et al reported that sacubitril-valsartan reduced rates of cardiovascular mortality both from worsening heart failure and from sudden cardiac death, independent of whether the patient had an ICD.22
Sacubitril-valsartan is taken twice a day, but most heart failure patients already take medications at several times during the day, so this should not pose a problem.
More information is needed on the use of this new drug in patients with New York Heart Association class IV symptoms, as only 60 patients with class IV symptoms were included in the PARADIGM-HF trial. Also, the efficacy of the drug in patients unable to tolerate a full dose will need to be analyzed.
PARADIGM-HF was conducted in stable, nonhospitalized patients with chronic heart failure; the use of the drug in new-onset heart failure and its initiation in hospitalized patients will require further study. In addition, the PARAGON-HF trial23 will examine the efficacy of sacubitril-valsartan in patients with heart failure and an ejection fraction of 45% or higher.
Sacubitril-valsartan ushers in a new era in heart failure treatment for patients with reduced ejection fraction and will certainly prompt quick revision of heart failure guidelines.
Three decades ago, the only drugs we had for treating chronic heart failure were digitalis and loop diuretics. The mortality rate was very high, and heart transplantation was a newly developing treatment that could help only a very few patients.
The early 80s heralded new hope for patients with heart failure, with the introduction of angiotensin-converting enzyme (ACE) inhibitors1–5 and, later, beta-blockers. Beta-blockers were considered contraindicated in heart failure until new trials provided evidence of dramatic benefit such as better quality of life and longer survival.6–8 ACE inhibitors, along with beta-blockers, quickly became the standard of care for all patients with systolic heart failure.
The implantable cardioverter-defibrillator (ICD) required numerous clinical trials in ischemic and nonischemic cardiomyopathy to define its role.9,10 Cardiac resynchronization therapy did not arrive until 15 years ago and is now indicated in a specific niche of patients with left bundle branch block.11,12 Mineralocorticoid antagonists required three pivotal clinical trials before their important role in the treatment of systolic heart failure was defined.13–16
And in the current decade, the roles of ACE inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, mineralocorticoid antagonists, ICDs, and cardiac resynchronization therapy have been further defined, as reflected in the latest guidelines for the treatment of systolic heart failure.17
Guideline-directed medical therapy for systolic heart failure with the agents and devices mentioned above improves quality of life and extends survival. It was therefore hard to imagine that any new additive therapy could offer significant incremental improvement. However, more than 5 years ago, in an ambitious effort, the largest global clinical trial ever performed in chronic heart failure was launched with a novel agent.18
THE PARADIGM-HF TRIAL
In this issue of the Journal, Sabe et al19 describe the results of the Prospective Comparison of ARNI With ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial of the novel combination drug sacubitril-valsartan, designated LCZ696 during its development and now available as Entresto.20
The mean age of the 8,442 patients in PARADIGM-HF was 64, and 78% were men. Despite guideline-directed medical therapy (93% of the patients were receiving a beta-blocker, and 60% were receiving a mineralocorticoid receptor antagonist), patients had persistent symptoms and signs of heart failure, diminished health-related quality of life, reduced ejection fraction (mean 29%), and elevated n-terminal pro-B-type natriuretic peptide levels (median 1,608 pg/mL, interquartile range 886–3,221).
The investigators reported a remarkable 20% reduction in the primary outcome of death from cardiovascular causes or hospitalization for heart failure in the patients who received sacubitril-valsartan compared with enalapril.20
Sacubitril-valsartan was reviewed under a US Food and Drug Administration (FDA) program that provides expedited review of drugs that are intended to treat a serious disease or condition and that may provide a significant improvement over available therapy. It was also granted a fast-track designation, which supports FDA efforts to facilitate the development and expedite the review of drugs to treat serious and life-threatening conditions and fill an unmet medical need. The FDA approved sacubitril-valsartan on July 7, 2015, for use in place of an ACE inhibitor or ARB in patients with New York Heart Association class II, III, or IV heart failure with reduced ejection fraction.21
WHAT WE STILL NEED TO KNOW
The results of PARADIGM-HF are generalizable, and sacubitril-valsartan was well tolerated in patients whose blood pressure was acceptable and who were able to tolerate ACE inhibitors in target doses. More than 90% of patients were receiving a beta-blocker. The dosing of enalapril (target 10 mg twice a day) is the guideline-directed target dose, and ACE inhibition is considered the gold standard for heart failure with reduced ejection fraction. Sacubitril-valsartan vs enalapril was a very appropriate comparison.
Far fewer PARADIGM-HF patients outside the United States had an ICD than those in the United States, which is a common finding in global clinical trials. However, Desai et al reported that sacubitril-valsartan reduced rates of cardiovascular mortality both from worsening heart failure and from sudden cardiac death, independent of whether the patient had an ICD.22
Sacubitril-valsartan is taken twice a day, but most heart failure patients already take medications at several times during the day, so this should not pose a problem.
More information is needed on the use of this new drug in patients with New York Heart Association class IV symptoms, as only 60 patients with class IV symptoms were included in the PARADIGM-HF trial. Also, the efficacy of the drug in patients unable to tolerate a full dose will need to be analyzed.
PARADIGM-HF was conducted in stable, nonhospitalized patients with chronic heart failure; the use of the drug in new-onset heart failure and its initiation in hospitalized patients will require further study. In addition, the PARAGON-HF trial23 will examine the efficacy of sacubitril-valsartan in patients with heart failure and an ejection fraction of 45% or higher.
Sacubitril-valsartan ushers in a new era in heart failure treatment for patients with reduced ejection fraction and will certainly prompt quick revision of heart failure guidelines.
- Captopril Multicenter Research Group. A placebo-controlled trial of captopril in refractory chronic congestive heart failure. J Am Coll Cardiol 1983; 2:755–763.
- Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987; 316:1429–1435.
- The SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fraction and congestive heart failure. N Engl J Med 1991; 325:293–302.
- Cohn JN, Johnson G, Ziesche S, et al. A comparison of enalapril with hydralazine-isosorbide dinitrate in the treatment of chronic congestive heart failure. N Engl J Med 1991; 325:303–310.
- Pfeffer MA, Braunwald E, Moyé LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 1992; 327:669–677.
- Packer M, Coats AJ, Fowler MB, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med 2001; 344:1651–1658.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999; 353:2001–2007.
- Brophy JM, Joseph L, Rouleau JL. Beta-blockers in congestive heart failure. A Bayesian meta-analysis. Ann Intern Med 2001; 134:550–560.
- Buxton AE, Lee KL, Fisher JD, et al. A randomized study of the prevention of sudden death in patients with coronary artery disease. Multicenter Unsustained Tachycardia Trial Investigators. N Engl J Med 1999; 341:1882–1890.
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med 2002; 346:877–883.
- Abraham WT, Fisher WG, Smith AL, et al; MIRACLE Study Group. Multicenter InSync Randomized Clinical Evaluation. Cardiac resynchronization in chronic heart failure. N Engl J Med 2002; 346:1845–1853.
- McAlister FA, Ezekowitz J, Hooton N, et al. Cardiac resynchronization therapy for patients with left ventricular systolic dysfunction: a systematic review. JAMA 2007; 297:2502–2514.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:709–717.
- Pitt B, Remme W, Zannad F, et al; Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 2003; 348:1309–1321.
- Pitt B, White H, Nicolau J, et al; EPHESUS Investigators. Eplerenone reduces mortality 30 days after randomization following acute myocardial infarction in patients with left ventricular systolic dysfunction and heart failure. J Am Coll Cardiol 2005; 46:425–431.
- Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011; 364:11–21.
- Yancy CW, Jessup M, Bozkurt B, et al; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 62:e147–e239.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Committees and Investigators. Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Fail 2013; 15:1062–1073.
- Sabe IA, Jacob MS, Taylor DO. A new class of drugs for systolic heart failure: The PARADIGM-HF study. Cleve Clin J Med 2015; 82:693–701.
- McMurray JJ, Packer M, Desai AS, Gong J, et al; PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014; 371:993–1004.
- US Food and Drug Administration. FDA approves new drug to treat heart failure. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm453845.htm. Accessed September 2, 2015.
- Desai AS, McMurray JJ, Packer M, et al. Effect of the angiotensin-receptor-neprilysin inhibitor LCZ696 compared with enalapril on mode of death in heart failure patients. Eur Heart J 2015; 36:1990–1997.
- ClinicalTrials.gov. Efficacy and Safety of LCZ696 Compared to Valsartan, on Morbidity and Mortality in Heart Failure Patients With Preserved Ejection Fraction (PARAGON-HF). https://clinicaltrials.gov/ct2/show/NCT01920711. Accessed September 2, 2015.
- Captopril Multicenter Research Group. A placebo-controlled trial of captopril in refractory chronic congestive heart failure. J Am Coll Cardiol 1983; 2:755–763.
- Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987; 316:1429–1435.
- The SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fraction and congestive heart failure. N Engl J Med 1991; 325:293–302.
- Cohn JN, Johnson G, Ziesche S, et al. A comparison of enalapril with hydralazine-isosorbide dinitrate in the treatment of chronic congestive heart failure. N Engl J Med 1991; 325:303–310.
- Pfeffer MA, Braunwald E, Moyé LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 1992; 327:669–677.
- Packer M, Coats AJ, Fowler MB, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med 2001; 344:1651–1658.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999; 353:2001–2007.
- Brophy JM, Joseph L, Rouleau JL. Beta-blockers in congestive heart failure. A Bayesian meta-analysis. Ann Intern Med 2001; 134:550–560.
- Buxton AE, Lee KL, Fisher JD, et al. A randomized study of the prevention of sudden death in patients with coronary artery disease. Multicenter Unsustained Tachycardia Trial Investigators. N Engl J Med 1999; 341:1882–1890.
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med 2002; 346:877–883.
- Abraham WT, Fisher WG, Smith AL, et al; MIRACLE Study Group. Multicenter InSync Randomized Clinical Evaluation. Cardiac resynchronization in chronic heart failure. N Engl J Med 2002; 346:1845–1853.
- McAlister FA, Ezekowitz J, Hooton N, et al. Cardiac resynchronization therapy for patients with left ventricular systolic dysfunction: a systematic review. JAMA 2007; 297:2502–2514.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:709–717.
- Pitt B, Remme W, Zannad F, et al; Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 2003; 348:1309–1321.
- Pitt B, White H, Nicolau J, et al; EPHESUS Investigators. Eplerenone reduces mortality 30 days after randomization following acute myocardial infarction in patients with left ventricular systolic dysfunction and heart failure. J Am Coll Cardiol 2005; 46:425–431.
- Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011; 364:11–21.
- Yancy CW, Jessup M, Bozkurt B, et al; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 62:e147–e239.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Committees and Investigators. Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Fail 2013; 15:1062–1073.
- Sabe IA, Jacob MS, Taylor DO. A new class of drugs for systolic heart failure: The PARADIGM-HF study. Cleve Clin J Med 2015; 82:693–701.
- McMurray JJ, Packer M, Desai AS, Gong J, et al; PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014; 371:993–1004.
- US Food and Drug Administration. FDA approves new drug to treat heart failure. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm453845.htm. Accessed September 2, 2015.
- Desai AS, McMurray JJ, Packer M, et al. Effect of the angiotensin-receptor-neprilysin inhibitor LCZ696 compared with enalapril on mode of death in heart failure patients. Eur Heart J 2015; 36:1990–1997.
- ClinicalTrials.gov. Efficacy and Safety of LCZ696 Compared to Valsartan, on Morbidity and Mortality in Heart Failure Patients With Preserved Ejection Fraction (PARAGON-HF). https://clinicaltrials.gov/ct2/show/NCT01920711. Accessed September 2, 2015.
A new class of drugs for systolic heart failure: The PARADIGM-HF study
In a large phase trial, a combination drug that contains the angiotensin II receptor blocker (ARB) valsartan and the neprilysin inhibitor sacubitril was found to be superior to the angiotensin-converting enzyme (ACE) inhibitor enalapril in terms of important end points, including death and hospitalization for heart failure, in patients with heart failure with reduced ejection fraction.1
Recently approved by the US Food and Drug Administration, this combination drug, marketed under the brand name Entresto, represents a new drug class, angiotensin receptor-neprilysin inhibitors, or ARNIs.
This article is an overview of the Prospective Comparison of ARNI With ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial1 and the implications it may have on the care of patients with chronic heart failure.
NEED FOR NEW HEART FAILURE DRUGS
Heart failure is a major public health problem, and the care of patients with heart failure is challenging.
Almost 6 million US adults have heart failure, and the prevalence is projected to increase in the next few decades as the population continues to age.2 Furthermore, the total healthcare cost for heart failure patients was almost $31 billion in 2012 and is projected to rise to $70 billion by 2030.2
The care of patients with severely decompensated heart failure has changed dramatically in the last few decades with advances in heart transplantation and mechanical support devices. But day-to-day management of patients with chronic mildly to moderately symptomatic heart failure continues to pose a clinical challenge.
The drugs currently available for these patients include beta-blockers, ACE inhibitors, ARBs, aldosterone antagonists, digoxin, diuretics, and vasodilators. But even with these drugs, the death and readmission rates of patients with heart failure with reduced ejection fraction remain high. More than 50% of patients with heart failure die within 5 years of diagnosis,3 and 25% of patients hospitalized with heart failure are readmitted within 30 days of discharge.2 Furthermore, death rates are higher in those patients who have a history of heart failure hospitalization.4
Although heart failure with preserved ejection fraction encompasses an important group of heart failure patients with high morbidity, the focus of this article will be on patients with heart failure with reduced ejection fraction.
Available drugs to date
The cornerstone drugs that lower the odds of death in patients with heart failure with reduced ejection fraction are ACE inhibitors, ARBs, beta-blockers, and mineralocorticoid antagonists.
ACE inhibitors were the first class of drugs shown to reduce the death rate in patients with heart failure with reduced ejection fraction. The landmark CONSENSUS trial,5 published in 1987, found that the death rate in patients who received enalapril was 27% lower than in those receiving placebo, an effect driven entirely by a reduction in progressive heart failure. Similarly, the SOLVD trial,6 published in 1991, showed a 26% reduction in heart failure hospitalization and a 16% lower rate of death with enalapril compared with placebo, an effect driven predominantly by a decrease in the progression of heart failure.
ARBs have also been shown to decrease the rate of death, although not by as much as ACE inhibitors. In the CHARM trial,7 compared with placebo, candesartan significantly decreased the risk of death from any cause, of death from cardiovascular causes, and of hospitalization related to heart failure.7
Beta-blockers. The MERIT-HF trial,8 published in 1999, was stopped early because fewer patients were dying in the group receiving metoprolol succinate than in the group receiving placebo (relative risk 0.66). Similarly, in 2001, the COPERNICUS trial9 reported a 34% reduction in deaths in patients receiving carvedilol in addition to an ACE inhibitor compared with those receiving an ACE inhibitor alone.
Mineralocorticoid receptor antagonists were found to be beneficial when added to standard therapy for chronic symptomatic heart failure in the RALES10 and EMPHASIS-HF11 trials.
Vasodilators (specifically, the combination of isosorbide dinitrate and hydralazine) were found to have benefit in terms of mortality when added to standard therapy in African American patients in the A-HeEFT trial.12
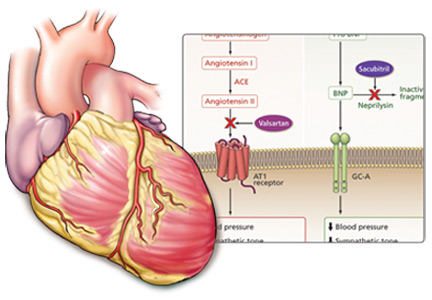
WHY INHIBIT BOTH ANGIOTENSIN AND NEPRILYSIN?
The renin-angiotensin-aldosterone system is a major focus in treating heart failure, as overactivity of this system plays a key role in the pathophysiology of this disease. Therefore, essential drugs for heart failure patients include those that inhibit overactivity of this system such as ACE inhibitors, ARBs, and aldosterone antagonists.
The natriuretic peptide system is another important pathway that can be targeted in patients with heart failure. Natriuretic peptides are key molecules that counteract heart failure, as they contribute to diuresis and vasodilation and protect against vascular remodeling.13 An increased understanding of the importance of this system in slowing the progression of heart failure has motivated evaluation of drugs such as nesiritide in patients with symptomatic heart failure. However, these drugs can cause hypotension and have limited bioavailability.14
Neprilysin is an endopeptidase—an endogenous enzyme that degrades vasoactive peptides such as bradykinin and natriuretic peptides.14 Drugs that inhibit neprilysin increase the levels of these peptides and thus counteract neurohormonal stimuli that lead to cardiac remodeling, sodium retention, and vasoconstriction.15
However, neprilysin also hydrolyzes angiotensin I to angiotensin (1–7), an inhibitor of angiotensin II. Thus, inhibition of neprilysin alone could lead to increased activity of angiotensin II and so have an overall neutral effect. To be beneficial, neprilysin inhibition needs to be combined with renin-angiotensin system inhibition. Furthermore, the benefit of renin-angiotensin-aldosterone system blockade may be amplified by up-regulation of the endogenous natriuretic peptide system.15
Omapatrilat, the most studied combination neprilysin inhibitor and ACE inhibitor, improved cardiac function and decreased cardiac mass in animal experiments.15 In addition, this drug showed promise in terms of blood pressure, heart failure readmissions, death, and preservation of renal function when compared with ACE inhibitors in patients with heart failure.15–17 But in clinical trials this drug posed a greater risk of hypotension, dizziness, and, its major shortcoming, an unacceptably high incidence of angioedema compared with ACE inhibitors.15,16,18 This higher risk of angioedema is thought to be from inhibition of three enzymes that break down bradykinin: ACE, neprilysin, and aminopeptidase P.19
ARNIs contain an angiotensin receptor blocker rather than an ACE inhibitor, and thus in theory they may be associated with a lower risk of angioedema.19 Sacubitril-valsartan, the first drug of this class, contains its two constitutive drugs in a one-to-one molecular ratio (Figure 1).
PARADIGM-HF investigated the benefit of this drug in patients with systolic heart failure.1
STUDY DESIGN AND OBJECTIVES
PARADIGM-HF was a double-blind, randomized controlled trial comparing sacubitril-valsartan and enalapril in patients with chronic systolic heart failure. As such, it was the first trial in recent years to study a new drug in comparison with a well-established heart failure drug rather than as an add-on strategy.1
Inclusion criteria
To be included in the PARADIGM-HF trial, patients had to have:
- A left ventricular ejection fraction of 40% or less (later changed to ≤ 35%)
- New York Heart Association class II, III, or IV symptoms
- A B-type natriuretic peptide (BNP) level of at least 150 pg/mL or an N-terminal proBNP (NT-proBNP) level of at least 600 pg/mL; for patients hospitalized for heart failure within the previous 12 months, the cut points were lower (BNP ≥ 100 pg/mL or NT-proBNP ≥ 400 pg/mL).
End points
The primary end point was the composite of cardiovascular death or first hospitalization for heart failure. Other outcomes assessed were time to death from any cause, the change from baseline in the Kansas City Cardiomyopathy Questionnaire (KCCQ) score at 8 months, time to new-onset atrial fibrillation, and the time to decline in renal function (defined as end-stage renal disease or a decrease in estimated glomerular filtration rate of at least 50% from randomization). All end points were blindly adjudicated by a clinical end points committee.
Two run-in periods
The study enrolled 10,521 patients from 1,043 centers in 47 countries, who entered the initial run-in period consisting of 2 weeks of treatment with enalapril at the study dosage (10 mg twice daily) in order to ensure no unacceptable side effects. At this point, 1,102 patients exited the study, leaving 9,419 who entered the second run-in period.
The second run-in period consisted of 4 weeks of treatment with sacubitril-valsartan, initially at half the study regimen (100 mg twice daily) and eventually at the full study dosage (200 mg twice daily). During the second run-in period, 977 participants left the study, leaving a total of 8,442 patients who underwent randomization. Forty-three patients were then excluded (6 because of invalid randomization and 37 because of four sites that closed because of major violations of good clinical practice).
Of those randomized, 4,187 patients were assigned to the sacubitril-valsartan treatment group and 4,212 were assigned to the enalapril group. The investigators used an intention-to-treat analysis for this study.
Most patients had NYHA class II symptoms
The randomized patients had a mean age of 64 years, 75% were men, 66% were white, and 58% were from Europe (only 7% were from North America). The mean left ventricular ejection fraction was about 30%, and 60% of the study participants had an ischemic cause for their cardiomyopathy. Although one of the inclusion criteria was New York Heart Association class II, III, or IV symptoms, about 5% of the patients had class I symptoms. Seventy percent had class II symptoms, 24% had class III, and less than 1% had class IV symptoms.
At the time of randomization, 78% of the patients were taking an ACE inhibitor and 93% were taking a beta-blocker, but only a little more than half were taking a mineralocorticoid antagonist and only about 15% had an implantable cardioverter-defibrillator.
STUDY OUTCOMES
This study was designed to detect a 15% lower risk of cardiovascular death in the sacubitril-valsartan group. It was projected to continue for at least 34 months but was stopped early because of an overwhelming benefit of the new drug at a median follow-up of 27 months.
Major findings
The primary composite outcome (cardiovascular death or first hospitalization for heart failure)1 occurred in 21.8% of the patients in the sacubitril-valsartan group vs 26.5% of patients in the enalapril group (hazard ratio [HR] 0.80, 95% confidence interval [CI] 0.73–0.87, P < .001). The number of patients who needed to be treated to prevent one occurrence of the primary composite outcome (100/absolute risk reduction) was only 21. The benefit was strong and consistent across both of the individual components of the composite outcome:
- Cardiovascular death 13.3% vs 16.5%, HR 0.80 (95% CI 0.71–0.89), P < .001
- First hospitalization for worsening heart failure 12.8% vs 15.6%, HR 0.79 (95% CI 0.71–0.89), P < .001.
Secondary outcomes. The sacubitril-valsartan group had a significantly lower rate of death from any cause (17.0% vs 19.8%, HR 0.84, 95% CI 0.76–0.93, P < .001) and a lower mean decrease in KCCQ clinical summary scores at 8 months (2.99 points vs 4.63 points, mean difference 1.64, 95% CI 0.63–2.65, P = .001). The KCCQ score measures subjective symptoms and physical limitations caused by heart failure; possible scores range from 0 to 100, with a higher score indicating better functional status. Notably, sacubitril-valsartan did not increase the KCCQ score in these patients; rather, sacubitril-valsartan recipients had a lower decrease in their scores than those in the enalapril group.
The incidence of new-onset atrial fibrillation was the same in both groups (3.1% and 3.1%).1 A decline in renal function (defined as end-stage renal disease, a decrease of 50% or more in estimated glomerular filtration rate from the value at randomization or a decrease in the estimated glomerular filtration rate of more than 30 mL/min/1.73 m2 to less than 60 mL/min/1.73 m2) occurred in 2.2% of the valsartan-sacubitril group and 2.6% of the enalapril group (P = .28).
The effects of the study drug on the primary composite outcome and on death from a cardiovascular cause were similar in all prespecified subgroups except for NYHA class: the reduction in the risk of the composite outcome was lower in sacubitril-valsartan recipients with NYHA I or II symptoms than in those with NYHA III or IV symptoms (P for interaction .03). However, there were no differences in the other prespecified subgroups, defined by age, sex, race, region, estimated glomerular filtration rate, diabetes, systolic blood pressure, ejection fraction, atrial fibrillation, NT-proBNP, hypertension, previous use of an ACE inhibitor, previous use of an aldosterone antagonist, previous heart failure hospitalization, and time since diagnosis of heart failure.
SAFETY: ANGIOEDEMA, HYPOTENSION, AND RENAL DYSFUNCTION
Angioedema
As noted above, the combination of neprilysin inhibitors and ACE inhibitors has been associated with an increased risk of angioedema. That was an important consideration before starting this study, which used a combination of a neprilysin inhibitor and an ARB in an attempt to avoid this serious side effect.
As it happened, there was no increased risk of significant angioedema with sacubitril-valsartan use compared with enalapril. Rates were similar to those in other studies, which showed a less than 1% risk of angioedema caused by ACE inhibitors.20,21 Only 19 patients (0.5%) in the sacubitril-valsartan group and 10 patients (0.2%) in the enalapril group experienced any angioedema. Of these, just three patients in the sacubitril-valsartan group and one patient in the enalapril group experienced angioedema that required hospitalization (P = .31). None of these patients had airway compromise due to angioedema.
Hypotension, cough, renal dysfunction, hyperkalemia
Other safety issues that were assessed included hypotension, worsening renal function, increase in potassium levels, and cough. Patients in the sacubitril-valsartan group were more likely to have symptomatic hypotension than patients in the enalapril group (14.0% vs 9.2%, P < .001); however, the authors noted that this was a rare cause of drug discontinuation.
Patients in the sacubitril-valsartan group were less likely to develop cough (11.3% vs 14.3%, P < .001), a serum creatinine level of 2.5 mg/dL or more (3.3% vs 4.5%, P = .007), or a serum potassium level of more than 6.0 mmol/L (11.3% vs 14.3%, P = .007).1
During the two run-in periods combined, 12% of the patients in the study withdrew because of adverse events, including cough, renal dysfunction, hyperkalemia, and symptomatic hypotension. During the enalapril run-in period, 591 patients (5.6%) withdrew due to adverse events, and 547 patients (5.8%) withdrew due to these events during the sacubitril-valsartan run-in period. After adjusting for the shorter time on enalapril, there was a higher rate of withdrawal because of adverse events from enalapril than from sacubitril-valsartan.
LOWER RISK OF CLINICAL PROGRESSION
In a separate paper,22 the PARADIGM-HF investigators reported that, among the survivors in the study, those who received sacubitril-valsartan fared better in terms of a number of markers of progression of heart failure, with lower rates of:
- Intensification of medical treatment for heart failure
- Emergency department visits for worsening heart failure
- Hospitalization for worsening heart failure
- Need for intensive care
- Need for intravenous inotropic agents
- Need for cardiac devices or heart transplants
- Worsening symptom scores
- Elevation of biomarkers of myocardial injury.
QUESTIONS AND CONCERNS
Although this study, which was funded by the manufacturer, showed consistent benefit for sacubitril-valsartan over enalapril, questions remain.
Are the findings generalizable?
Despite the study’s rigorous run-in period, 12% of patients dropped out because of adverse events, and thus the patients who completed the study may not be representative of the general population of heart failure patients. The authors included this double-level wash-out to ensure patient tolerance of both drugs. But in everyday practice, a significant number of patients may be unable to tolerate one of these drugs.
Moreover, after adjusting for the difference in the run-in periods, patients actually withdrew more often during the enalapril run-in period than during the sacubitril-valsartan run-in period. However, there may be overlap in tolerability in these two drugs, which both affect the renin-angiotensin-aldosterone system. Thus, the enalapril run-in period may have contributed to the lower tolerability of this drug compared with sacubitril-valsartan.
Were patients receiving the best possible therapy?
Another important point when considering how we treat heart failure patients in the United States is how few patients in this study were using cardiac implantable electronic devices. Only 15% of the patients in this study had an implantable cardioverter-defibrillator despite a mean left ventricular ejection fraction less than 30%. This likely reflects differences in practice internationally; however, based on American College of Cardiology, American Heart Association, and Heart Rhythm Society guidelines, these patients would have a class I indication for an implantable cardioverter-defibrillator for primary prevention of sudden cardiac death.23
Therefore, based on these recommendations, the patients in this study were not necessarily on optimal medical and device therapy and furthermore may not be representative of heart failure patients in the United States.
Was enalapril 10 mg twice a day a fair comparison?
Another concern about the results of this study relates to the dosages used in the two treatment groups. The sacubitril-valsartan formulation included full-dose valsartan, whereas the ACE inhibitor group received enalapril at less than a full dose.
Although the authors explained that the dose of enalapril chosen for the study was based on the one used in previous studies that showed survival benefit, this raises the question of whether the significant difference in outcomes in this trial was due to a greater inhibition of the renin-angiotensin-aldosterone system related to a higher dose of drug in the sacubitril-valsartan group.
What about black patients taking hydralazine-isosorbide?
Only about 5% of patients in the PARADIGM-HF trial were black. Based on the A-HeFT study results, black patients can be prescribed an ACE inhibitor as well as hydralazine and isosorbide dinitrate as tolerated to decrease the risk of death. Does sacubitril-valsartan offer benefit to these patients compared with a regimen of an ACE inhibitor, hydralazine, and isosorbide dinitrate?
Another concern is that the incidence of angioedema observed with ACE inhibitors and omapatrilat is higher in black patients.15,21 Thus, it would be prudent to investigate whether the risk of angioedema with sacubitril-valsartan would be higher if more black patients are studied.
IMPLICATIONS AND CONSIDERATIONS
In this study, sacubitril-valsartan showed impressive and consistent results, with an almost 20% decrease in the composite end point of heart failure hospitalization or cardiovascular death and a similar decrease in the composite outcomes with a very low number needed to treat (21 patients). It did not show a decrease in the incidence of new-onset atrial fibrillation; however, only 80 cases of atrial fibrillation were reported, so there may have been a lack of statistical power to detect a difference.
To avoid angioedema, wait at least 36 hours after stopping an ACE inhibitor. Sacubitril-valsartan was not associated with an increased risk of severe angioedema, and no patients experienced life-threatening angioedema. In the trial, the sacubitril-valsartan run-in period was started at least 24 hours after enalapril was stopped, and thus the authors recommended at least a 1-day washout period after discontinuing an ACE inhibitor to avoid angioedema in patients starting sacubitril-valsartan.
Hypotension is a concern. Although there was actually a decreased risk of renal dysfunction, hyperkalemia, and cough compared with enalapril, there was a significantly increased rate of symptomatic hypotension in the sacubitril-valsartan group, which raises the question of patient tolerance and physician comfort when prescribing and titrating this drug in clinical practice. This side effect will be an important consideration when attempting to titrate the drug to target doses.
Start treatment early. This trial largely consisted of patients with NYHA class II or III symptoms, with about 70% of patients with class II symptoms. Since this drug showed benefit in patients with mildly to moderately symptomatic heart failure, clinicians who are considering prescribing this drug should not wait until the patient is closer to end-stage disease. Patients with mildly symptomatic heart failure may be followed by a general cardiologist, internist, or both, and thus it is important to emphasize to the entire medical community the need to start this medication early on.
How much will it cost? Cost is a concern that could heavily weigh on the decision to prescribe this drug. Generic ACE inhibitors are relatively inexpensive, and it may difficult to switch from an affordable generic drug to a new drug that is likely to be much more expensive. Arguably, this drug may be cost-effective in the long run owing to a large decrease in heart failure readmissions. We await further analyses to evaluate this issue.
Will patients take a twice-a-day drug as prescribed? Most patients who are prescribed an ACE inhibitor take it just once a day, and switching from a daily to a twice-daily drug may present a challenge for some.
What about other outcomes? Based on this study, it is unclear what effect sacubitril-valsartan has on the incidence of fatal arrhythmias, sudden cardiac death, and pump failure. Furthermore, the effect on quality of life is still uncertain. Quality of life is an integral component in the evaluation of heart failure patients, and in this study the changes in KCCQ scores were not impressive. We hope to see further evaluations of this drug’s impact on quality of life of patients with heart failure. Furthermore, it would be interesting to study if this drug has any long-term effects on the need for advanced therapies such as left ventricular assist devices and orthotopic heart transplant.
What about patients with heart failure with preserved ejection fraction? This study included only patients with heart failure with reduced ejection fraction. However PARAMOUNT, a phase 2 study that evaluated the benefit of sacubitril-valsartan in patients with heart failure with preserved ejection fraction, has shown encouraging results.24 We look forward to further investigation of this agent in patients with heart failure with preserved ejection fraction.
Sacubitril-valsartan, the first ARNI to be studied in humans, has a dual action in that it enhances the activity of the natriuretic peptide system and inhibits that of the renin-angiotensin-aldosterone system. It is the first drug in over a decade to show mortality benefit in patients with chronic systolic heart failure when compared with an already well-established heart failure medication. It appears to decrease rates of mortality and heart failure hospitalization without increasing the risk of severe angioedema in patients with mild or moderate chronic systolic heart failure. Symptomatic hypotension and high cost may pose the largest barriers to the use of this new drug. And we have yet to see how the clinical community and patients with heart failure will respond to it.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014; 371:993–1004.
- Dunlay SM, Pereira NL, Kushwaha SS. Contemporary strategies in the diagnosis and management of heart failure. Mayo Clin Proc 2014; 89:662–676.
- Go AS, Mozaffarian D, Roger VL, et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2013 update: a report from the American Heart Association. Circulation 2013; 127:e6–e245.
- Bello NA, Claggett B, Desai AS, et al. Influence of previous heart failure hospitalization on cardiovascular events in patients with reduced and preserved ejection fraction. Circ Heart Fail 2014; 7:590–595.
- Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987; 316:1429–1435.
- Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med 1991; 325:293–302.
- Pfeffer MA, Swedberg K, Granger CB, et al; CHARM Investigators and Committees. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme. Lancet 2003; 362:759–766.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999; 353:2001–2007.
- Packer M, Coats AJ, Fowler MB, et al; Carvedilol Prospective Randomized Cumulative Survival Study Group. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med 2001; 344:1651–1658.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:709–717.
- Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011; 364:11–21.
- Taylor AL, Ziesche S, Yancy C, et al; African-American Heart Failure Trial Investigators. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med 2004; 351:2049–2057.
- Schreiner GF, Protter AA. B-type natriuretic peptide for the treatment of congestive heart failure. Curr Opin Pharmacol 2002; 2:142–147.
- von Lueder TG, Sangaralingham SJ, Wang BH, et al. Renin-angiotensin blockade combined with natriuretic peptide system augmentation: novel therapeutic concepts to combat heart failure. Circ Heart Fail 2013; 6:594–605.
- Abassi Z, Karram T, Ellaham S, Winaver J, Hoffman A. Implications of the natriuretic peptide system in the pathogenesis of heart failure: diagnostic and therapeutic importance. Pharmacol Ther 2004; 102:223–241.
- Packer M, Califf RM, Konstam MA, et al. Comparison of omapatrilat and enalapril in patients with chronic heart failure: the Omapatrilat Versus Enalapril Randomized Trial of Utility in Reducing Events (OVERTURE). Circulation 2002; 106:920–926.
- Rouleau JL, Pfeffer MA, Stewart DJ, et al. Comparison of vasopeptidase inhibitor, omapatrilat, and lisinopril on exercise tolerance and morbidity in patients with heart failure: IMPRESS randomised trial. Lancet 2000; 356:615–620.
- Kostis JB, Packer M, Black HR, Schmieder R, Henry D, Levy E. Omapatrilat and enalapril in patients with hypertension: the Omapatrilat Cardiovascular Treatment vs. Enalapril (OCTAVE) trial. Am J Hypertens 2004; 17:103–111.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Committees and Investigators. Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Fail 2013; 15:1062–1073.
- Toh S, Reichman ME, Houstoun M, et al. Comparative risk for angioedema associated with the use of drugs that target the renin-angiotensin-aldosterone system. Arch Intern Med 2012; 172:1582–1589.
- Kostis JB, Kim HJ, Rusnak J, et al. Incidence and characteristics of angioedema associated with enalapril. Arch Intern Med 2005; 165:1637–1642.
- Packer M, McMurray JJV, Desai AS, et al; on behalf of the PARADIGM-HF Investigators and Coordinators. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation 2015; 131:54–61.
- Epstein AE, Dimarco JP, Ellenbogen KA, et al; American College of Cardiology/American Heart Association Task Force on Practice; American Association for Thoracic Surgery; Society of Thoracic Surgeons. ACC/AHA/HRS 2008 guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: executive summary. Heart Rhythm 2008; 5:934–955.
- Solomon SD, Zile M, Pieske B, et al; Prospective comparison of ARNI with ARB on Management Of Heart Failure with Preserved Ejection Fraction (PARAMOUNT) Investigators. The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. Lancet 2012; 380:1387–1395.
In a large phase trial, a combination drug that contains the angiotensin II receptor blocker (ARB) valsartan and the neprilysin inhibitor sacubitril was found to be superior to the angiotensin-converting enzyme (ACE) inhibitor enalapril in terms of important end points, including death and hospitalization for heart failure, in patients with heart failure with reduced ejection fraction.1
Recently approved by the US Food and Drug Administration, this combination drug, marketed under the brand name Entresto, represents a new drug class, angiotensin receptor-neprilysin inhibitors, or ARNIs.
This article is an overview of the Prospective Comparison of ARNI With ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial1 and the implications it may have on the care of patients with chronic heart failure.
NEED FOR NEW HEART FAILURE DRUGS
Heart failure is a major public health problem, and the care of patients with heart failure is challenging.
Almost 6 million US adults have heart failure, and the prevalence is projected to increase in the next few decades as the population continues to age.2 Furthermore, the total healthcare cost for heart failure patients was almost $31 billion in 2012 and is projected to rise to $70 billion by 2030.2
The care of patients with severely decompensated heart failure has changed dramatically in the last few decades with advances in heart transplantation and mechanical support devices. But day-to-day management of patients with chronic mildly to moderately symptomatic heart failure continues to pose a clinical challenge.
The drugs currently available for these patients include beta-blockers, ACE inhibitors, ARBs, aldosterone antagonists, digoxin, diuretics, and vasodilators. But even with these drugs, the death and readmission rates of patients with heart failure with reduced ejection fraction remain high. More than 50% of patients with heart failure die within 5 years of diagnosis,3 and 25% of patients hospitalized with heart failure are readmitted within 30 days of discharge.2 Furthermore, death rates are higher in those patients who have a history of heart failure hospitalization.4
Although heart failure with preserved ejection fraction encompasses an important group of heart failure patients with high morbidity, the focus of this article will be on patients with heart failure with reduced ejection fraction.
Available drugs to date
The cornerstone drugs that lower the odds of death in patients with heart failure with reduced ejection fraction are ACE inhibitors, ARBs, beta-blockers, and mineralocorticoid antagonists.
ACE inhibitors were the first class of drugs shown to reduce the death rate in patients with heart failure with reduced ejection fraction. The landmark CONSENSUS trial,5 published in 1987, found that the death rate in patients who received enalapril was 27% lower than in those receiving placebo, an effect driven entirely by a reduction in progressive heart failure. Similarly, the SOLVD trial,6 published in 1991, showed a 26% reduction in heart failure hospitalization and a 16% lower rate of death with enalapril compared with placebo, an effect driven predominantly by a decrease in the progression of heart failure.
ARBs have also been shown to decrease the rate of death, although not by as much as ACE inhibitors. In the CHARM trial,7 compared with placebo, candesartan significantly decreased the risk of death from any cause, of death from cardiovascular causes, and of hospitalization related to heart failure.7
Beta-blockers. The MERIT-HF trial,8 published in 1999, was stopped early because fewer patients were dying in the group receiving metoprolol succinate than in the group receiving placebo (relative risk 0.66). Similarly, in 2001, the COPERNICUS trial9 reported a 34% reduction in deaths in patients receiving carvedilol in addition to an ACE inhibitor compared with those receiving an ACE inhibitor alone.
Mineralocorticoid receptor antagonists were found to be beneficial when added to standard therapy for chronic symptomatic heart failure in the RALES10 and EMPHASIS-HF11 trials.
Vasodilators (specifically, the combination of isosorbide dinitrate and hydralazine) were found to have benefit in terms of mortality when added to standard therapy in African American patients in the A-HeEFT trial.12
WHY INHIBIT BOTH ANGIOTENSIN AND NEPRILYSIN?
The renin-angiotensin-aldosterone system is a major focus in treating heart failure, as overactivity of this system plays a key role in the pathophysiology of this disease. Therefore, essential drugs for heart failure patients include those that inhibit overactivity of this system such as ACE inhibitors, ARBs, and aldosterone antagonists.
The natriuretic peptide system is another important pathway that can be targeted in patients with heart failure. Natriuretic peptides are key molecules that counteract heart failure, as they contribute to diuresis and vasodilation and protect against vascular remodeling.13 An increased understanding of the importance of this system in slowing the progression of heart failure has motivated evaluation of drugs such as nesiritide in patients with symptomatic heart failure. However, these drugs can cause hypotension and have limited bioavailability.14
Neprilysin is an endopeptidase—an endogenous enzyme that degrades vasoactive peptides such as bradykinin and natriuretic peptides.14 Drugs that inhibit neprilysin increase the levels of these peptides and thus counteract neurohormonal stimuli that lead to cardiac remodeling, sodium retention, and vasoconstriction.15
However, neprilysin also hydrolyzes angiotensin I to angiotensin (1–7), an inhibitor of angiotensin II. Thus, inhibition of neprilysin alone could lead to increased activity of angiotensin II and so have an overall neutral effect. To be beneficial, neprilysin inhibition needs to be combined with renin-angiotensin system inhibition. Furthermore, the benefit of renin-angiotensin-aldosterone system blockade may be amplified by up-regulation of the endogenous natriuretic peptide system.15
Omapatrilat, the most studied combination neprilysin inhibitor and ACE inhibitor, improved cardiac function and decreased cardiac mass in animal experiments.15 In addition, this drug showed promise in terms of blood pressure, heart failure readmissions, death, and preservation of renal function when compared with ACE inhibitors in patients with heart failure.15–17 But in clinical trials this drug posed a greater risk of hypotension, dizziness, and, its major shortcoming, an unacceptably high incidence of angioedema compared with ACE inhibitors.15,16,18 This higher risk of angioedema is thought to be from inhibition of three enzymes that break down bradykinin: ACE, neprilysin, and aminopeptidase P.19
ARNIs contain an angiotensin receptor blocker rather than an ACE inhibitor, and thus in theory they may be associated with a lower risk of angioedema.19 Sacubitril-valsartan, the first drug of this class, contains its two constitutive drugs in a one-to-one molecular ratio (Figure 1).
PARADIGM-HF investigated the benefit of this drug in patients with systolic heart failure.1
STUDY DESIGN AND OBJECTIVES
PARADIGM-HF was a double-blind, randomized controlled trial comparing sacubitril-valsartan and enalapril in patients with chronic systolic heart failure. As such, it was the first trial in recent years to study a new drug in comparison with a well-established heart failure drug rather than as an add-on strategy.1
Inclusion criteria
To be included in the PARADIGM-HF trial, patients had to have:
- A left ventricular ejection fraction of 40% or less (later changed to ≤ 35%)
- New York Heart Association class II, III, or IV symptoms
- A B-type natriuretic peptide (BNP) level of at least 150 pg/mL or an N-terminal proBNP (NT-proBNP) level of at least 600 pg/mL; for patients hospitalized for heart failure within the previous 12 months, the cut points were lower (BNP ≥ 100 pg/mL or NT-proBNP ≥ 400 pg/mL).
End points
The primary end point was the composite of cardiovascular death or first hospitalization for heart failure. Other outcomes assessed were time to death from any cause, the change from baseline in the Kansas City Cardiomyopathy Questionnaire (KCCQ) score at 8 months, time to new-onset atrial fibrillation, and the time to decline in renal function (defined as end-stage renal disease or a decrease in estimated glomerular filtration rate of at least 50% from randomization). All end points were blindly adjudicated by a clinical end points committee.
Two run-in periods
The study enrolled 10,521 patients from 1,043 centers in 47 countries, who entered the initial run-in period consisting of 2 weeks of treatment with enalapril at the study dosage (10 mg twice daily) in order to ensure no unacceptable side effects. At this point, 1,102 patients exited the study, leaving 9,419 who entered the second run-in period.
The second run-in period consisted of 4 weeks of treatment with sacubitril-valsartan, initially at half the study regimen (100 mg twice daily) and eventually at the full study dosage (200 mg twice daily). During the second run-in period, 977 participants left the study, leaving a total of 8,442 patients who underwent randomization. Forty-three patients were then excluded (6 because of invalid randomization and 37 because of four sites that closed because of major violations of good clinical practice).
Of those randomized, 4,187 patients were assigned to the sacubitril-valsartan treatment group and 4,212 were assigned to the enalapril group. The investigators used an intention-to-treat analysis for this study.
Most patients had NYHA class II symptoms
The randomized patients had a mean age of 64 years, 75% were men, 66% were white, and 58% were from Europe (only 7% were from North America). The mean left ventricular ejection fraction was about 30%, and 60% of the study participants had an ischemic cause for their cardiomyopathy. Although one of the inclusion criteria was New York Heart Association class II, III, or IV symptoms, about 5% of the patients had class I symptoms. Seventy percent had class II symptoms, 24% had class III, and less than 1% had class IV symptoms.
At the time of randomization, 78% of the patients were taking an ACE inhibitor and 93% were taking a beta-blocker, but only a little more than half were taking a mineralocorticoid antagonist and only about 15% had an implantable cardioverter-defibrillator.
STUDY OUTCOMES
This study was designed to detect a 15% lower risk of cardiovascular death in the sacubitril-valsartan group. It was projected to continue for at least 34 months but was stopped early because of an overwhelming benefit of the new drug at a median follow-up of 27 months.
Major findings
The primary composite outcome (cardiovascular death or first hospitalization for heart failure)1 occurred in 21.8% of the patients in the sacubitril-valsartan group vs 26.5% of patients in the enalapril group (hazard ratio [HR] 0.80, 95% confidence interval [CI] 0.73–0.87, P < .001). The number of patients who needed to be treated to prevent one occurrence of the primary composite outcome (100/absolute risk reduction) was only 21. The benefit was strong and consistent across both of the individual components of the composite outcome:
- Cardiovascular death 13.3% vs 16.5%, HR 0.80 (95% CI 0.71–0.89), P < .001
- First hospitalization for worsening heart failure 12.8% vs 15.6%, HR 0.79 (95% CI 0.71–0.89), P < .001.
Secondary outcomes. The sacubitril-valsartan group had a significantly lower rate of death from any cause (17.0% vs 19.8%, HR 0.84, 95% CI 0.76–0.93, P < .001) and a lower mean decrease in KCCQ clinical summary scores at 8 months (2.99 points vs 4.63 points, mean difference 1.64, 95% CI 0.63–2.65, P = .001). The KCCQ score measures subjective symptoms and physical limitations caused by heart failure; possible scores range from 0 to 100, with a higher score indicating better functional status. Notably, sacubitril-valsartan did not increase the KCCQ score in these patients; rather, sacubitril-valsartan recipients had a lower decrease in their scores than those in the enalapril group.
The incidence of new-onset atrial fibrillation was the same in both groups (3.1% and 3.1%).1 A decline in renal function (defined as end-stage renal disease, a decrease of 50% or more in estimated glomerular filtration rate from the value at randomization or a decrease in the estimated glomerular filtration rate of more than 30 mL/min/1.73 m2 to less than 60 mL/min/1.73 m2) occurred in 2.2% of the valsartan-sacubitril group and 2.6% of the enalapril group (P = .28).
The effects of the study drug on the primary composite outcome and on death from a cardiovascular cause were similar in all prespecified subgroups except for NYHA class: the reduction in the risk of the composite outcome was lower in sacubitril-valsartan recipients with NYHA I or II symptoms than in those with NYHA III or IV symptoms (P for interaction .03). However, there were no differences in the other prespecified subgroups, defined by age, sex, race, region, estimated glomerular filtration rate, diabetes, systolic blood pressure, ejection fraction, atrial fibrillation, NT-proBNP, hypertension, previous use of an ACE inhibitor, previous use of an aldosterone antagonist, previous heart failure hospitalization, and time since diagnosis of heart failure.
SAFETY: ANGIOEDEMA, HYPOTENSION, AND RENAL DYSFUNCTION
Angioedema
As noted above, the combination of neprilysin inhibitors and ACE inhibitors has been associated with an increased risk of angioedema. That was an important consideration before starting this study, which used a combination of a neprilysin inhibitor and an ARB in an attempt to avoid this serious side effect.
As it happened, there was no increased risk of significant angioedema with sacubitril-valsartan use compared with enalapril. Rates were similar to those in other studies, which showed a less than 1% risk of angioedema caused by ACE inhibitors.20,21 Only 19 patients (0.5%) in the sacubitril-valsartan group and 10 patients (0.2%) in the enalapril group experienced any angioedema. Of these, just three patients in the sacubitril-valsartan group and one patient in the enalapril group experienced angioedema that required hospitalization (P = .31). None of these patients had airway compromise due to angioedema.
Hypotension, cough, renal dysfunction, hyperkalemia
Other safety issues that were assessed included hypotension, worsening renal function, increase in potassium levels, and cough. Patients in the sacubitril-valsartan group were more likely to have symptomatic hypotension than patients in the enalapril group (14.0% vs 9.2%, P < .001); however, the authors noted that this was a rare cause of drug discontinuation.
Patients in the sacubitril-valsartan group were less likely to develop cough (11.3% vs 14.3%, P < .001), a serum creatinine level of 2.5 mg/dL or more (3.3% vs 4.5%, P = .007), or a serum potassium level of more than 6.0 mmol/L (11.3% vs 14.3%, P = .007).1
During the two run-in periods combined, 12% of the patients in the study withdrew because of adverse events, including cough, renal dysfunction, hyperkalemia, and symptomatic hypotension. During the enalapril run-in period, 591 patients (5.6%) withdrew due to adverse events, and 547 patients (5.8%) withdrew due to these events during the sacubitril-valsartan run-in period. After adjusting for the shorter time on enalapril, there was a higher rate of withdrawal because of adverse events from enalapril than from sacubitril-valsartan.
LOWER RISK OF CLINICAL PROGRESSION
In a separate paper,22 the PARADIGM-HF investigators reported that, among the survivors in the study, those who received sacubitril-valsartan fared better in terms of a number of markers of progression of heart failure, with lower rates of:
- Intensification of medical treatment for heart failure
- Emergency department visits for worsening heart failure
- Hospitalization for worsening heart failure
- Need for intensive care
- Need for intravenous inotropic agents
- Need for cardiac devices or heart transplants
- Worsening symptom scores
- Elevation of biomarkers of myocardial injury.
QUESTIONS AND CONCERNS
Although this study, which was funded by the manufacturer, showed consistent benefit for sacubitril-valsartan over enalapril, questions remain.
Are the findings generalizable?
Despite the study’s rigorous run-in period, 12% of patients dropped out because of adverse events, and thus the patients who completed the study may not be representative of the general population of heart failure patients. The authors included this double-level wash-out to ensure patient tolerance of both drugs. But in everyday practice, a significant number of patients may be unable to tolerate one of these drugs.
Moreover, after adjusting for the difference in the run-in periods, patients actually withdrew more often during the enalapril run-in period than during the sacubitril-valsartan run-in period. However, there may be overlap in tolerability in these two drugs, which both affect the renin-angiotensin-aldosterone system. Thus, the enalapril run-in period may have contributed to the lower tolerability of this drug compared with sacubitril-valsartan.
Were patients receiving the best possible therapy?
Another important point when considering how we treat heart failure patients in the United States is how few patients in this study were using cardiac implantable electronic devices. Only 15% of the patients in this study had an implantable cardioverter-defibrillator despite a mean left ventricular ejection fraction less than 30%. This likely reflects differences in practice internationally; however, based on American College of Cardiology, American Heart Association, and Heart Rhythm Society guidelines, these patients would have a class I indication for an implantable cardioverter-defibrillator for primary prevention of sudden cardiac death.23
Therefore, based on these recommendations, the patients in this study were not necessarily on optimal medical and device therapy and furthermore may not be representative of heart failure patients in the United States.
Was enalapril 10 mg twice a day a fair comparison?
Another concern about the results of this study relates to the dosages used in the two treatment groups. The sacubitril-valsartan formulation included full-dose valsartan, whereas the ACE inhibitor group received enalapril at less than a full dose.
Although the authors explained that the dose of enalapril chosen for the study was based on the one used in previous studies that showed survival benefit, this raises the question of whether the significant difference in outcomes in this trial was due to a greater inhibition of the renin-angiotensin-aldosterone system related to a higher dose of drug in the sacubitril-valsartan group.
What about black patients taking hydralazine-isosorbide?
Only about 5% of patients in the PARADIGM-HF trial were black. Based on the A-HeFT study results, black patients can be prescribed an ACE inhibitor as well as hydralazine and isosorbide dinitrate as tolerated to decrease the risk of death. Does sacubitril-valsartan offer benefit to these patients compared with a regimen of an ACE inhibitor, hydralazine, and isosorbide dinitrate?
Another concern is that the incidence of angioedema observed with ACE inhibitors and omapatrilat is higher in black patients.15,21 Thus, it would be prudent to investigate whether the risk of angioedema with sacubitril-valsartan would be higher if more black patients are studied.
IMPLICATIONS AND CONSIDERATIONS
In this study, sacubitril-valsartan showed impressive and consistent results, with an almost 20% decrease in the composite end point of heart failure hospitalization or cardiovascular death and a similar decrease in the composite outcomes with a very low number needed to treat (21 patients). It did not show a decrease in the incidence of new-onset atrial fibrillation; however, only 80 cases of atrial fibrillation were reported, so there may have been a lack of statistical power to detect a difference.
To avoid angioedema, wait at least 36 hours after stopping an ACE inhibitor. Sacubitril-valsartan was not associated with an increased risk of severe angioedema, and no patients experienced life-threatening angioedema. In the trial, the sacubitril-valsartan run-in period was started at least 24 hours after enalapril was stopped, and thus the authors recommended at least a 1-day washout period after discontinuing an ACE inhibitor to avoid angioedema in patients starting sacubitril-valsartan.
Hypotension is a concern. Although there was actually a decreased risk of renal dysfunction, hyperkalemia, and cough compared with enalapril, there was a significantly increased rate of symptomatic hypotension in the sacubitril-valsartan group, which raises the question of patient tolerance and physician comfort when prescribing and titrating this drug in clinical practice. This side effect will be an important consideration when attempting to titrate the drug to target doses.
Start treatment early. This trial largely consisted of patients with NYHA class II or III symptoms, with about 70% of patients with class II symptoms. Since this drug showed benefit in patients with mildly to moderately symptomatic heart failure, clinicians who are considering prescribing this drug should not wait until the patient is closer to end-stage disease. Patients with mildly symptomatic heart failure may be followed by a general cardiologist, internist, or both, and thus it is important to emphasize to the entire medical community the need to start this medication early on.
How much will it cost? Cost is a concern that could heavily weigh on the decision to prescribe this drug. Generic ACE inhibitors are relatively inexpensive, and it may difficult to switch from an affordable generic drug to a new drug that is likely to be much more expensive. Arguably, this drug may be cost-effective in the long run owing to a large decrease in heart failure readmissions. We await further analyses to evaluate this issue.
Will patients take a twice-a-day drug as prescribed? Most patients who are prescribed an ACE inhibitor take it just once a day, and switching from a daily to a twice-daily drug may present a challenge for some.
What about other outcomes? Based on this study, it is unclear what effect sacubitril-valsartan has on the incidence of fatal arrhythmias, sudden cardiac death, and pump failure. Furthermore, the effect on quality of life is still uncertain. Quality of life is an integral component in the evaluation of heart failure patients, and in this study the changes in KCCQ scores were not impressive. We hope to see further evaluations of this drug’s impact on quality of life of patients with heart failure. Furthermore, it would be interesting to study if this drug has any long-term effects on the need for advanced therapies such as left ventricular assist devices and orthotopic heart transplant.
What about patients with heart failure with preserved ejection fraction? This study included only patients with heart failure with reduced ejection fraction. However PARAMOUNT, a phase 2 study that evaluated the benefit of sacubitril-valsartan in patients with heart failure with preserved ejection fraction, has shown encouraging results.24 We look forward to further investigation of this agent in patients with heart failure with preserved ejection fraction.
Sacubitril-valsartan, the first ARNI to be studied in humans, has a dual action in that it enhances the activity of the natriuretic peptide system and inhibits that of the renin-angiotensin-aldosterone system. It is the first drug in over a decade to show mortality benefit in patients with chronic systolic heart failure when compared with an already well-established heart failure medication. It appears to decrease rates of mortality and heart failure hospitalization without increasing the risk of severe angioedema in patients with mild or moderate chronic systolic heart failure. Symptomatic hypotension and high cost may pose the largest barriers to the use of this new drug. And we have yet to see how the clinical community and patients with heart failure will respond to it.
In a large phase trial, a combination drug that contains the angiotensin II receptor blocker (ARB) valsartan and the neprilysin inhibitor sacubitril was found to be superior to the angiotensin-converting enzyme (ACE) inhibitor enalapril in terms of important end points, including death and hospitalization for heart failure, in patients with heart failure with reduced ejection fraction.1
Recently approved by the US Food and Drug Administration, this combination drug, marketed under the brand name Entresto, represents a new drug class, angiotensin receptor-neprilysin inhibitors, or ARNIs.
This article is an overview of the Prospective Comparison of ARNI With ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial1 and the implications it may have on the care of patients with chronic heart failure.
NEED FOR NEW HEART FAILURE DRUGS
Heart failure is a major public health problem, and the care of patients with heart failure is challenging.
Almost 6 million US adults have heart failure, and the prevalence is projected to increase in the next few decades as the population continues to age.2 Furthermore, the total healthcare cost for heart failure patients was almost $31 billion in 2012 and is projected to rise to $70 billion by 2030.2
The care of patients with severely decompensated heart failure has changed dramatically in the last few decades with advances in heart transplantation and mechanical support devices. But day-to-day management of patients with chronic mildly to moderately symptomatic heart failure continues to pose a clinical challenge.
The drugs currently available for these patients include beta-blockers, ACE inhibitors, ARBs, aldosterone antagonists, digoxin, diuretics, and vasodilators. But even with these drugs, the death and readmission rates of patients with heart failure with reduced ejection fraction remain high. More than 50% of patients with heart failure die within 5 years of diagnosis,3 and 25% of patients hospitalized with heart failure are readmitted within 30 days of discharge.2 Furthermore, death rates are higher in those patients who have a history of heart failure hospitalization.4
Although heart failure with preserved ejection fraction encompasses an important group of heart failure patients with high morbidity, the focus of this article will be on patients with heart failure with reduced ejection fraction.
Available drugs to date
The cornerstone drugs that lower the odds of death in patients with heart failure with reduced ejection fraction are ACE inhibitors, ARBs, beta-blockers, and mineralocorticoid antagonists.
ACE inhibitors were the first class of drugs shown to reduce the death rate in patients with heart failure with reduced ejection fraction. The landmark CONSENSUS trial,5 published in 1987, found that the death rate in patients who received enalapril was 27% lower than in those receiving placebo, an effect driven entirely by a reduction in progressive heart failure. Similarly, the SOLVD trial,6 published in 1991, showed a 26% reduction in heart failure hospitalization and a 16% lower rate of death with enalapril compared with placebo, an effect driven predominantly by a decrease in the progression of heart failure.
ARBs have also been shown to decrease the rate of death, although not by as much as ACE inhibitors. In the CHARM trial,7 compared with placebo, candesartan significantly decreased the risk of death from any cause, of death from cardiovascular causes, and of hospitalization related to heart failure.7
Beta-blockers. The MERIT-HF trial,8 published in 1999, was stopped early because fewer patients were dying in the group receiving metoprolol succinate than in the group receiving placebo (relative risk 0.66). Similarly, in 2001, the COPERNICUS trial9 reported a 34% reduction in deaths in patients receiving carvedilol in addition to an ACE inhibitor compared with those receiving an ACE inhibitor alone.
Mineralocorticoid receptor antagonists were found to be beneficial when added to standard therapy for chronic symptomatic heart failure in the RALES10 and EMPHASIS-HF11 trials.
Vasodilators (specifically, the combination of isosorbide dinitrate and hydralazine) were found to have benefit in terms of mortality when added to standard therapy in African American patients in the A-HeEFT trial.12
WHY INHIBIT BOTH ANGIOTENSIN AND NEPRILYSIN?
The renin-angiotensin-aldosterone system is a major focus in treating heart failure, as overactivity of this system plays a key role in the pathophysiology of this disease. Therefore, essential drugs for heart failure patients include those that inhibit overactivity of this system such as ACE inhibitors, ARBs, and aldosterone antagonists.
The natriuretic peptide system is another important pathway that can be targeted in patients with heart failure. Natriuretic peptides are key molecules that counteract heart failure, as they contribute to diuresis and vasodilation and protect against vascular remodeling.13 An increased understanding of the importance of this system in slowing the progression of heart failure has motivated evaluation of drugs such as nesiritide in patients with symptomatic heart failure. However, these drugs can cause hypotension and have limited bioavailability.14
Neprilysin is an endopeptidase—an endogenous enzyme that degrades vasoactive peptides such as bradykinin and natriuretic peptides.14 Drugs that inhibit neprilysin increase the levels of these peptides and thus counteract neurohormonal stimuli that lead to cardiac remodeling, sodium retention, and vasoconstriction.15
However, neprilysin also hydrolyzes angiotensin I to angiotensin (1–7), an inhibitor of angiotensin II. Thus, inhibition of neprilysin alone could lead to increased activity of angiotensin II and so have an overall neutral effect. To be beneficial, neprilysin inhibition needs to be combined with renin-angiotensin system inhibition. Furthermore, the benefit of renin-angiotensin-aldosterone system blockade may be amplified by up-regulation of the endogenous natriuretic peptide system.15
Omapatrilat, the most studied combination neprilysin inhibitor and ACE inhibitor, improved cardiac function and decreased cardiac mass in animal experiments.15 In addition, this drug showed promise in terms of blood pressure, heart failure readmissions, death, and preservation of renal function when compared with ACE inhibitors in patients with heart failure.15–17 But in clinical trials this drug posed a greater risk of hypotension, dizziness, and, its major shortcoming, an unacceptably high incidence of angioedema compared with ACE inhibitors.15,16,18 This higher risk of angioedema is thought to be from inhibition of three enzymes that break down bradykinin: ACE, neprilysin, and aminopeptidase P.19
ARNIs contain an angiotensin receptor blocker rather than an ACE inhibitor, and thus in theory they may be associated with a lower risk of angioedema.19 Sacubitril-valsartan, the first drug of this class, contains its two constitutive drugs in a one-to-one molecular ratio (Figure 1).
PARADIGM-HF investigated the benefit of this drug in patients with systolic heart failure.1
STUDY DESIGN AND OBJECTIVES
PARADIGM-HF was a double-blind, randomized controlled trial comparing sacubitril-valsartan and enalapril in patients with chronic systolic heart failure. As such, it was the first trial in recent years to study a new drug in comparison with a well-established heart failure drug rather than as an add-on strategy.1
Inclusion criteria
To be included in the PARADIGM-HF trial, patients had to have:
- A left ventricular ejection fraction of 40% or less (later changed to ≤ 35%)
- New York Heart Association class II, III, or IV symptoms
- A B-type natriuretic peptide (BNP) level of at least 150 pg/mL or an N-terminal proBNP (NT-proBNP) level of at least 600 pg/mL; for patients hospitalized for heart failure within the previous 12 months, the cut points were lower (BNP ≥ 100 pg/mL or NT-proBNP ≥ 400 pg/mL).
End points
The primary end point was the composite of cardiovascular death or first hospitalization for heart failure. Other outcomes assessed were time to death from any cause, the change from baseline in the Kansas City Cardiomyopathy Questionnaire (KCCQ) score at 8 months, time to new-onset atrial fibrillation, and the time to decline in renal function (defined as end-stage renal disease or a decrease in estimated glomerular filtration rate of at least 50% from randomization). All end points were blindly adjudicated by a clinical end points committee.
Two run-in periods
The study enrolled 10,521 patients from 1,043 centers in 47 countries, who entered the initial run-in period consisting of 2 weeks of treatment with enalapril at the study dosage (10 mg twice daily) in order to ensure no unacceptable side effects. At this point, 1,102 patients exited the study, leaving 9,419 who entered the second run-in period.
The second run-in period consisted of 4 weeks of treatment with sacubitril-valsartan, initially at half the study regimen (100 mg twice daily) and eventually at the full study dosage (200 mg twice daily). During the second run-in period, 977 participants left the study, leaving a total of 8,442 patients who underwent randomization. Forty-three patients were then excluded (6 because of invalid randomization and 37 because of four sites that closed because of major violations of good clinical practice).
Of those randomized, 4,187 patients were assigned to the sacubitril-valsartan treatment group and 4,212 were assigned to the enalapril group. The investigators used an intention-to-treat analysis for this study.
Most patients had NYHA class II symptoms
The randomized patients had a mean age of 64 years, 75% were men, 66% were white, and 58% were from Europe (only 7% were from North America). The mean left ventricular ejection fraction was about 30%, and 60% of the study participants had an ischemic cause for their cardiomyopathy. Although one of the inclusion criteria was New York Heart Association class II, III, or IV symptoms, about 5% of the patients had class I symptoms. Seventy percent had class II symptoms, 24% had class III, and less than 1% had class IV symptoms.
At the time of randomization, 78% of the patients were taking an ACE inhibitor and 93% were taking a beta-blocker, but only a little more than half were taking a mineralocorticoid antagonist and only about 15% had an implantable cardioverter-defibrillator.
STUDY OUTCOMES
This study was designed to detect a 15% lower risk of cardiovascular death in the sacubitril-valsartan group. It was projected to continue for at least 34 months but was stopped early because of an overwhelming benefit of the new drug at a median follow-up of 27 months.
Major findings
The primary composite outcome (cardiovascular death or first hospitalization for heart failure)1 occurred in 21.8% of the patients in the sacubitril-valsartan group vs 26.5% of patients in the enalapril group (hazard ratio [HR] 0.80, 95% confidence interval [CI] 0.73–0.87, P < .001). The number of patients who needed to be treated to prevent one occurrence of the primary composite outcome (100/absolute risk reduction) was only 21. The benefit was strong and consistent across both of the individual components of the composite outcome:
- Cardiovascular death 13.3% vs 16.5%, HR 0.80 (95% CI 0.71–0.89), P < .001
- First hospitalization for worsening heart failure 12.8% vs 15.6%, HR 0.79 (95% CI 0.71–0.89), P < .001.
Secondary outcomes. The sacubitril-valsartan group had a significantly lower rate of death from any cause (17.0% vs 19.8%, HR 0.84, 95% CI 0.76–0.93, P < .001) and a lower mean decrease in KCCQ clinical summary scores at 8 months (2.99 points vs 4.63 points, mean difference 1.64, 95% CI 0.63–2.65, P = .001). The KCCQ score measures subjective symptoms and physical limitations caused by heart failure; possible scores range from 0 to 100, with a higher score indicating better functional status. Notably, sacubitril-valsartan did not increase the KCCQ score in these patients; rather, sacubitril-valsartan recipients had a lower decrease in their scores than those in the enalapril group.
The incidence of new-onset atrial fibrillation was the same in both groups (3.1% and 3.1%).1 A decline in renal function (defined as end-stage renal disease, a decrease of 50% or more in estimated glomerular filtration rate from the value at randomization or a decrease in the estimated glomerular filtration rate of more than 30 mL/min/1.73 m2 to less than 60 mL/min/1.73 m2) occurred in 2.2% of the valsartan-sacubitril group and 2.6% of the enalapril group (P = .28).
The effects of the study drug on the primary composite outcome and on death from a cardiovascular cause were similar in all prespecified subgroups except for NYHA class: the reduction in the risk of the composite outcome was lower in sacubitril-valsartan recipients with NYHA I or II symptoms than in those with NYHA III or IV symptoms (P for interaction .03). However, there were no differences in the other prespecified subgroups, defined by age, sex, race, region, estimated glomerular filtration rate, diabetes, systolic blood pressure, ejection fraction, atrial fibrillation, NT-proBNP, hypertension, previous use of an ACE inhibitor, previous use of an aldosterone antagonist, previous heart failure hospitalization, and time since diagnosis of heart failure.
SAFETY: ANGIOEDEMA, HYPOTENSION, AND RENAL DYSFUNCTION
Angioedema
As noted above, the combination of neprilysin inhibitors and ACE inhibitors has been associated with an increased risk of angioedema. That was an important consideration before starting this study, which used a combination of a neprilysin inhibitor and an ARB in an attempt to avoid this serious side effect.
As it happened, there was no increased risk of significant angioedema with sacubitril-valsartan use compared with enalapril. Rates were similar to those in other studies, which showed a less than 1% risk of angioedema caused by ACE inhibitors.20,21 Only 19 patients (0.5%) in the sacubitril-valsartan group and 10 patients (0.2%) in the enalapril group experienced any angioedema. Of these, just three patients in the sacubitril-valsartan group and one patient in the enalapril group experienced angioedema that required hospitalization (P = .31). None of these patients had airway compromise due to angioedema.
Hypotension, cough, renal dysfunction, hyperkalemia
Other safety issues that were assessed included hypotension, worsening renal function, increase in potassium levels, and cough. Patients in the sacubitril-valsartan group were more likely to have symptomatic hypotension than patients in the enalapril group (14.0% vs 9.2%, P < .001); however, the authors noted that this was a rare cause of drug discontinuation.
Patients in the sacubitril-valsartan group were less likely to develop cough (11.3% vs 14.3%, P < .001), a serum creatinine level of 2.5 mg/dL or more (3.3% vs 4.5%, P = .007), or a serum potassium level of more than 6.0 mmol/L (11.3% vs 14.3%, P = .007).1
During the two run-in periods combined, 12% of the patients in the study withdrew because of adverse events, including cough, renal dysfunction, hyperkalemia, and symptomatic hypotension. During the enalapril run-in period, 591 patients (5.6%) withdrew due to adverse events, and 547 patients (5.8%) withdrew due to these events during the sacubitril-valsartan run-in period. After adjusting for the shorter time on enalapril, there was a higher rate of withdrawal because of adverse events from enalapril than from sacubitril-valsartan.
LOWER RISK OF CLINICAL PROGRESSION
In a separate paper,22 the PARADIGM-HF investigators reported that, among the survivors in the study, those who received sacubitril-valsartan fared better in terms of a number of markers of progression of heart failure, with lower rates of:
- Intensification of medical treatment for heart failure
- Emergency department visits for worsening heart failure
- Hospitalization for worsening heart failure
- Need for intensive care
- Need for intravenous inotropic agents
- Need for cardiac devices or heart transplants
- Worsening symptom scores
- Elevation of biomarkers of myocardial injury.
QUESTIONS AND CONCERNS
Although this study, which was funded by the manufacturer, showed consistent benefit for sacubitril-valsartan over enalapril, questions remain.
Are the findings generalizable?
Despite the study’s rigorous run-in period, 12% of patients dropped out because of adverse events, and thus the patients who completed the study may not be representative of the general population of heart failure patients. The authors included this double-level wash-out to ensure patient tolerance of both drugs. But in everyday practice, a significant number of patients may be unable to tolerate one of these drugs.
Moreover, after adjusting for the difference in the run-in periods, patients actually withdrew more often during the enalapril run-in period than during the sacubitril-valsartan run-in period. However, there may be overlap in tolerability in these two drugs, which both affect the renin-angiotensin-aldosterone system. Thus, the enalapril run-in period may have contributed to the lower tolerability of this drug compared with sacubitril-valsartan.
Were patients receiving the best possible therapy?
Another important point when considering how we treat heart failure patients in the United States is how few patients in this study were using cardiac implantable electronic devices. Only 15% of the patients in this study had an implantable cardioverter-defibrillator despite a mean left ventricular ejection fraction less than 30%. This likely reflects differences in practice internationally; however, based on American College of Cardiology, American Heart Association, and Heart Rhythm Society guidelines, these patients would have a class I indication for an implantable cardioverter-defibrillator for primary prevention of sudden cardiac death.23
Therefore, based on these recommendations, the patients in this study were not necessarily on optimal medical and device therapy and furthermore may not be representative of heart failure patients in the United States.
Was enalapril 10 mg twice a day a fair comparison?
Another concern about the results of this study relates to the dosages used in the two treatment groups. The sacubitril-valsartan formulation included full-dose valsartan, whereas the ACE inhibitor group received enalapril at less than a full dose.
Although the authors explained that the dose of enalapril chosen for the study was based on the one used in previous studies that showed survival benefit, this raises the question of whether the significant difference in outcomes in this trial was due to a greater inhibition of the renin-angiotensin-aldosterone system related to a higher dose of drug in the sacubitril-valsartan group.
What about black patients taking hydralazine-isosorbide?
Only about 5% of patients in the PARADIGM-HF trial were black. Based on the A-HeFT study results, black patients can be prescribed an ACE inhibitor as well as hydralazine and isosorbide dinitrate as tolerated to decrease the risk of death. Does sacubitril-valsartan offer benefit to these patients compared with a regimen of an ACE inhibitor, hydralazine, and isosorbide dinitrate?
Another concern is that the incidence of angioedema observed with ACE inhibitors and omapatrilat is higher in black patients.15,21 Thus, it would be prudent to investigate whether the risk of angioedema with sacubitril-valsartan would be higher if more black patients are studied.
IMPLICATIONS AND CONSIDERATIONS
In this study, sacubitril-valsartan showed impressive and consistent results, with an almost 20% decrease in the composite end point of heart failure hospitalization or cardiovascular death and a similar decrease in the composite outcomes with a very low number needed to treat (21 patients). It did not show a decrease in the incidence of new-onset atrial fibrillation; however, only 80 cases of atrial fibrillation were reported, so there may have been a lack of statistical power to detect a difference.
To avoid angioedema, wait at least 36 hours after stopping an ACE inhibitor. Sacubitril-valsartan was not associated with an increased risk of severe angioedema, and no patients experienced life-threatening angioedema. In the trial, the sacubitril-valsartan run-in period was started at least 24 hours after enalapril was stopped, and thus the authors recommended at least a 1-day washout period after discontinuing an ACE inhibitor to avoid angioedema in patients starting sacubitril-valsartan.
Hypotension is a concern. Although there was actually a decreased risk of renal dysfunction, hyperkalemia, and cough compared with enalapril, there was a significantly increased rate of symptomatic hypotension in the sacubitril-valsartan group, which raises the question of patient tolerance and physician comfort when prescribing and titrating this drug in clinical practice. This side effect will be an important consideration when attempting to titrate the drug to target doses.
Start treatment early. This trial largely consisted of patients with NYHA class II or III symptoms, with about 70% of patients with class II symptoms. Since this drug showed benefit in patients with mildly to moderately symptomatic heart failure, clinicians who are considering prescribing this drug should not wait until the patient is closer to end-stage disease. Patients with mildly symptomatic heart failure may be followed by a general cardiologist, internist, or both, and thus it is important to emphasize to the entire medical community the need to start this medication early on.
How much will it cost? Cost is a concern that could heavily weigh on the decision to prescribe this drug. Generic ACE inhibitors are relatively inexpensive, and it may difficult to switch from an affordable generic drug to a new drug that is likely to be much more expensive. Arguably, this drug may be cost-effective in the long run owing to a large decrease in heart failure readmissions. We await further analyses to evaluate this issue.
Will patients take a twice-a-day drug as prescribed? Most patients who are prescribed an ACE inhibitor take it just once a day, and switching from a daily to a twice-daily drug may present a challenge for some.
What about other outcomes? Based on this study, it is unclear what effect sacubitril-valsartan has on the incidence of fatal arrhythmias, sudden cardiac death, and pump failure. Furthermore, the effect on quality of life is still uncertain. Quality of life is an integral component in the evaluation of heart failure patients, and in this study the changes in KCCQ scores were not impressive. We hope to see further evaluations of this drug’s impact on quality of life of patients with heart failure. Furthermore, it would be interesting to study if this drug has any long-term effects on the need for advanced therapies such as left ventricular assist devices and orthotopic heart transplant.
What about patients with heart failure with preserved ejection fraction? This study included only patients with heart failure with reduced ejection fraction. However PARAMOUNT, a phase 2 study that evaluated the benefit of sacubitril-valsartan in patients with heart failure with preserved ejection fraction, has shown encouraging results.24 We look forward to further investigation of this agent in patients with heart failure with preserved ejection fraction.
Sacubitril-valsartan, the first ARNI to be studied in humans, has a dual action in that it enhances the activity of the natriuretic peptide system and inhibits that of the renin-angiotensin-aldosterone system. It is the first drug in over a decade to show mortality benefit in patients with chronic systolic heart failure when compared with an already well-established heart failure medication. It appears to decrease rates of mortality and heart failure hospitalization without increasing the risk of severe angioedema in patients with mild or moderate chronic systolic heart failure. Symptomatic hypotension and high cost may pose the largest barriers to the use of this new drug. And we have yet to see how the clinical community and patients with heart failure will respond to it.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014; 371:993–1004.
- Dunlay SM, Pereira NL, Kushwaha SS. Contemporary strategies in the diagnosis and management of heart failure. Mayo Clin Proc 2014; 89:662–676.
- Go AS, Mozaffarian D, Roger VL, et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2013 update: a report from the American Heart Association. Circulation 2013; 127:e6–e245.
- Bello NA, Claggett B, Desai AS, et al. Influence of previous heart failure hospitalization on cardiovascular events in patients with reduced and preserved ejection fraction. Circ Heart Fail 2014; 7:590–595.
- Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987; 316:1429–1435.
- Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med 1991; 325:293–302.
- Pfeffer MA, Swedberg K, Granger CB, et al; CHARM Investigators and Committees. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme. Lancet 2003; 362:759–766.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999; 353:2001–2007.
- Packer M, Coats AJ, Fowler MB, et al; Carvedilol Prospective Randomized Cumulative Survival Study Group. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med 2001; 344:1651–1658.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:709–717.
- Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011; 364:11–21.
- Taylor AL, Ziesche S, Yancy C, et al; African-American Heart Failure Trial Investigators. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med 2004; 351:2049–2057.
- Schreiner GF, Protter AA. B-type natriuretic peptide for the treatment of congestive heart failure. Curr Opin Pharmacol 2002; 2:142–147.
- von Lueder TG, Sangaralingham SJ, Wang BH, et al. Renin-angiotensin blockade combined with natriuretic peptide system augmentation: novel therapeutic concepts to combat heart failure. Circ Heart Fail 2013; 6:594–605.
- Abassi Z, Karram T, Ellaham S, Winaver J, Hoffman A. Implications of the natriuretic peptide system in the pathogenesis of heart failure: diagnostic and therapeutic importance. Pharmacol Ther 2004; 102:223–241.
- Packer M, Califf RM, Konstam MA, et al. Comparison of omapatrilat and enalapril in patients with chronic heart failure: the Omapatrilat Versus Enalapril Randomized Trial of Utility in Reducing Events (OVERTURE). Circulation 2002; 106:920–926.
- Rouleau JL, Pfeffer MA, Stewart DJ, et al. Comparison of vasopeptidase inhibitor, omapatrilat, and lisinopril on exercise tolerance and morbidity in patients with heart failure: IMPRESS randomised trial. Lancet 2000; 356:615–620.
- Kostis JB, Packer M, Black HR, Schmieder R, Henry D, Levy E. Omapatrilat and enalapril in patients with hypertension: the Omapatrilat Cardiovascular Treatment vs. Enalapril (OCTAVE) trial. Am J Hypertens 2004; 17:103–111.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Committees and Investigators. Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Fail 2013; 15:1062–1073.
- Toh S, Reichman ME, Houstoun M, et al. Comparative risk for angioedema associated with the use of drugs that target the renin-angiotensin-aldosterone system. Arch Intern Med 2012; 172:1582–1589.
- Kostis JB, Kim HJ, Rusnak J, et al. Incidence and characteristics of angioedema associated with enalapril. Arch Intern Med 2005; 165:1637–1642.
- Packer M, McMurray JJV, Desai AS, et al; on behalf of the PARADIGM-HF Investigators and Coordinators. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation 2015; 131:54–61.
- Epstein AE, Dimarco JP, Ellenbogen KA, et al; American College of Cardiology/American Heart Association Task Force on Practice; American Association for Thoracic Surgery; Society of Thoracic Surgeons. ACC/AHA/HRS 2008 guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: executive summary. Heart Rhythm 2008; 5:934–955.
- Solomon SD, Zile M, Pieske B, et al; Prospective comparison of ARNI with ARB on Management Of Heart Failure with Preserved Ejection Fraction (PARAMOUNT) Investigators. The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. Lancet 2012; 380:1387–1395.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014; 371:993–1004.
- Dunlay SM, Pereira NL, Kushwaha SS. Contemporary strategies in the diagnosis and management of heart failure. Mayo Clin Proc 2014; 89:662–676.
- Go AS, Mozaffarian D, Roger VL, et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2013 update: a report from the American Heart Association. Circulation 2013; 127:e6–e245.
- Bello NA, Claggett B, Desai AS, et al. Influence of previous heart failure hospitalization on cardiovascular events in patients with reduced and preserved ejection fraction. Circ Heart Fail 2014; 7:590–595.
- Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987; 316:1429–1435.
- Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med 1991; 325:293–302.
- Pfeffer MA, Swedberg K, Granger CB, et al; CHARM Investigators and Committees. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme. Lancet 2003; 362:759–766.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999; 353:2001–2007.
- Packer M, Coats AJ, Fowler MB, et al; Carvedilol Prospective Randomized Cumulative Survival Study Group. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med 2001; 344:1651–1658.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999; 341:709–717.
- Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011; 364:11–21.
- Taylor AL, Ziesche S, Yancy C, et al; African-American Heart Failure Trial Investigators. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med 2004; 351:2049–2057.
- Schreiner GF, Protter AA. B-type natriuretic peptide for the treatment of congestive heart failure. Curr Opin Pharmacol 2002; 2:142–147.
- von Lueder TG, Sangaralingham SJ, Wang BH, et al. Renin-angiotensin blockade combined with natriuretic peptide system augmentation: novel therapeutic concepts to combat heart failure. Circ Heart Fail 2013; 6:594–605.
- Abassi Z, Karram T, Ellaham S, Winaver J, Hoffman A. Implications of the natriuretic peptide system in the pathogenesis of heart failure: diagnostic and therapeutic importance. Pharmacol Ther 2004; 102:223–241.
- Packer M, Califf RM, Konstam MA, et al. Comparison of omapatrilat and enalapril in patients with chronic heart failure: the Omapatrilat Versus Enalapril Randomized Trial of Utility in Reducing Events (OVERTURE). Circulation 2002; 106:920–926.
- Rouleau JL, Pfeffer MA, Stewart DJ, et al. Comparison of vasopeptidase inhibitor, omapatrilat, and lisinopril on exercise tolerance and morbidity in patients with heart failure: IMPRESS randomised trial. Lancet 2000; 356:615–620.
- Kostis JB, Packer M, Black HR, Schmieder R, Henry D, Levy E. Omapatrilat and enalapril in patients with hypertension: the Omapatrilat Cardiovascular Treatment vs. Enalapril (OCTAVE) trial. Am J Hypertens 2004; 17:103–111.
- McMurray JJ, Packer M, Desai AS, et al; PARADIGM-HF Committees and Investigators. Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Fail 2013; 15:1062–1073.
- Toh S, Reichman ME, Houstoun M, et al. Comparative risk for angioedema associated with the use of drugs that target the renin-angiotensin-aldosterone system. Arch Intern Med 2012; 172:1582–1589.
- Kostis JB, Kim HJ, Rusnak J, et al. Incidence and characteristics of angioedema associated with enalapril. Arch Intern Med 2005; 165:1637–1642.
- Packer M, McMurray JJV, Desai AS, et al; on behalf of the PARADIGM-HF Investigators and Coordinators. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation 2015; 131:54–61.
- Epstein AE, Dimarco JP, Ellenbogen KA, et al; American College of Cardiology/American Heart Association Task Force on Practice; American Association for Thoracic Surgery; Society of Thoracic Surgeons. ACC/AHA/HRS 2008 guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: executive summary. Heart Rhythm 2008; 5:934–955.
- Solomon SD, Zile M, Pieske B, et al; Prospective comparison of ARNI with ARB on Management Of Heart Failure with Preserved Ejection Fraction (PARAMOUNT) Investigators. The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. Lancet 2012; 380:1387–1395.
KEY POINTS
- Neprilysin is an endogenous enzyme that degrades vasoactive peptides such as bradykinin and natriuretic peptides. Inhibition of neprilysin raises the levels of these peptides, leading to less cardiac remodeling, less sodium retention, and less vasoconstriction.
- Neprilysin inhibition must be combined with inhibition of the renin-angiotensin-aldosterone system, optimally with an angiotensin II receptor blocker.
- PARADIGM-HF showed a 20% reduction in the primary outcome of death from cardiovascular causes or hospitalization for heart failure with sacubitril-valsartan 200 mg twice daily vs enalapril 10 mg twice daily at a median follow-up of 27 months.
- The ultimate role of combined neprilysin and angiotensin receptor inhibitors remains to be determined.
Genitourinary manifestations of sickle cell disease
Sickle cell disease is a common genetic disorder in the United States that disproportionately affects people of African ancestry. The characteristic sickling of red blood cells under conditions of reduced oxygen tension leads to intravascular hemolysis and vaso-occlusive events, which in turn cause tissue ischemia-reperfusion injury affecting multiple organs, including the genitourinary system.1–3
In this paper, we review the genitourinary effects of sickle cell disease, focusing on sickle cell nephropathy, priapism, and renal medullary carcinoma.
THE WIDE-RANGING EFFECTS OF SICKLE CELL DISEASE
In the United States, sickle cell disease affects 1 of every 500 blacks and 1 of every 36,000 Hispanics.1 The term describes hemoglobinopathies associated with sickling of red blood cells.
Sickling of red blood cells results from a single base-pair change in the beta-globin gene from glutamic acid to valine at position 6, causing abnormal hemoglobin (hemoglobin S), which polymerizes under conditions of reduced oxygen tension and alters the biconcave disk shape into a rigid, irregular, unstable cell. The sickle-shaped cells are prone to intravascular hemolysis,2 causing intermittent vaso-occlusive events that result in tissue ischemia-reperfusion injury. Genitourinary problems include impaired ability to concentrate urine, hematuria, renal medullary carcinoma, and increased frequency of urinary tract infection.
SICKLE CELL NEPHROPATHY
The kidney is one of the most frequently affected organs in sickle cell disease. Renal manifestations begin to appear in early childhood, with impaired medullary concentrating ability and ischemic damage to the tubular cells caused by sickling within the vasa recta renis precipitated by the acidic, hypoxic, and hypertonic environment in the renal medulla.
As in early diabetic nephropathy, renal blood flow is enhanced and the glomerular filtration rate (GFR) is increased. Increased cardiac output as a result of anemia, localized release of prostaglandins, and a hypoxia-induced increase in nitric oxide synthesis all play a role in the increase in GFR.4,5
Oxidative stress, an increase in markers of inflammation, and local activation of the renin-angiotensin system contribute to renal damage in sickle cell disease.5–7 The resulting hyperfiltration injury leads to microalbuminuria, which occurs in 20% to 40% of children with sickle cell anemia8,9 and in as many as 60% of adults.
The glomerular lesions associated with sickle cell disease vary from glomerulopathy in the early stages to secondary focal segmental glomerulosclerosis, membranoproliferative glomerulonephritis, and glomerular thrombotic microangiopathy.10
Clinical presentations and workup
Clinical presentations are not limited to glomerular disease but include hyperchloremic metabolic acidosis and hyperkalemia resulting from defects in potassium secretion and renal acidification.
Hyperphosphatemia—a result of increased reabsorption of phosphorus, increased secretion of uric acid, and increased creatinine clearance—is seen in patients with sickle cell disease.11,12 About 10% of patients can develop an acute kidney injury as a result of volume depletion, rhabdomyolysis, renal vein thrombosis, papillary necrosis, and urinary tract obstruction secondary to blood clots.11,13
Up to 30% of adult patients with sickle cell disease develop chronic kidney disease. Predictors include severe anemia, hypertension, proteinuria, nephrotic syndrome, and microscopic hematuria.14 From 4% to 12% of patients go on to develop end-stage renal disease, but with a 1-year mortality rate three times higher than in patients without sickle cell disease.15
In general, patients with sickle cell anemia have blood pressures below those of age- and sex-matched individuals, but elevated blood pressure and low GFR are not uncommon in affected children. In a cohort of 48 children ages 3 to 18, 8.3% had an estimated GFR less than 90 mL/min/1.73 m2, and 16.7% had elevated blood pressure (prehypertension and hypertension).16
In patients with sickle cell disease, evaluation of proteinuria, hematuria, hypertension, and renal failure should take into consideration the unique renal physiologic and pathologic processes involved. Recent evidence17,18 suggests that the Chronic Kidney Disease Epidemiology Collaboration equation provides a better estimate of GFR than the Modification of Diet in Renal Disease and Cockcroft-Gault equations, although all three creatinine-based methods overestimate GFR in patients with sickle cell disease when compared with GFR measured with technetium-99m-labeled diethylenetriamine penta-acetic acid renal scanning.
Treatment options
Treatment of sickle cell nephropathy includes adequate fluid intake (given the loss of concentrating ability), adequate blood pressure control, use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers (ARBs) in patients who have microalbuminuria or proteinuria (or both)9,11,19 and hydroxyurea. Treatment with enalapril has been shown to decrease proteinuria in patients with sickle cell nephropathy.9 In a cohort of children with sickle cell disease, four of nine patients treated with an ACE inhibitor developed hyperkalemia, leading to discontinuation of the drug in three patients.9
ACE inhibitors and ARBs must be used cautiously in these patients because they have defects in potassium secretion. Hydroxyurea has also been shown to decrease hyperfiltration and microalbuminuria in recent studies,20,21 and this could protect against the development of overt nephropathy.
Higher mortality rates have been reported in patients with sickle cell disease who developed end-stage renal disease than in patients with end-stage renal disease without sickle cell disease. Sickle cell disease also increases the risk of pulmonary hypertension and the vaso-occlusive complication known as acute chest syndrome, contributing to increased mortality rates. Of note, in a study that looked at the association between mortality rates and pre-end-stage care of renal disease using data from the Centers for Medicare and Medicaid Services, patients with sickle cell disease who had had predialysis nephrology care had lower mortality rates.15
Treatments for end-stage renal disease are also effective in patients with sickle cell disease and include hemodialysis, peritoneal dialysis, and renal transplantation. Data from the Organ Procurement and Transplantation Network and the United Network for Organ Sharing show that from 2000 to 2011, African American kidney recipients with sickle cell disease had better survival rates than patients who had undergone transplantation from 1988 to 1999, although rates of long-term survival and graft survival were lower than in transplant recipients with other diagnoses.22
It is important to note that complications as a result of vaso-occlusive events and thrombosis can lead to graft loss; therefore, sickle cell crisis after transplantation requires careful management.
Take-home messages
- Loss of urine-concentrating ability and hyperfiltration are the earliest pathologic changes in sickle cell disease.
- Microalbuminuria as seen in diabetic nephropathy is the earliest manifestation of sickle cell nephropathy, and the prevalence increases as these patients get older and live longer.
- ACE inhibitors or ARBs should be used with caution, given the heightened risk of hyperkalemia in sickle cell disease.
- Recent results with hydroxyurea in decreasing hyperfiltration and microalbuminuria are encouraging.
- Early referral for predialysis nephrologic care is needed in sickle cell patients with chronic kidney disease.
PRIAPISM IN SICKLE CELL DISEASE
Priapism was formerly defined as a full, painful erection lasting more than 4 hours and unrelated to sexual stimulation or orgasm. But priapism is now recognized as two separate disorders—ischemic (veno-occlusive, low-flow) priapism and nonischemic (arterial, high-flow) priapism. The new definition includes both disorders: ie, a full or partial erection lasting more than 4 hours and unrelated to sexual stimulation or orgasm.
Ischemic priapism
Hematologic disorders are major contributors to ischemic priapism and include sickle cell disease, multiple myeloma, fat emboli (hyperalimentation),23 glucose-6-phosphate dehydrogenase deficiency, and hemoglobin Olmsted variant.24
Ischemic priapism is often seen in sickle cell disease and is considered an emergency. It is characterized by an abnormally rigid erection not involving the glans penis. Entrapment of blood in the corpora cavernosa leads to hypoxia, hypercarbia, and acidosis, which in turn leads to a painful compartment syndrome that, if untreated, results in smooth muscle necrosis and subsequent fibrosis. The results are a smaller penis and erectile dysfunction that is unresponsive to any treatment other than implantation of a penile prosthesis. However, scarring of the corpora cavernosa can make this procedure exceedingly difficult, requiring advanced techniques such as corporeal excavation.25
Men with a subtype of ischemic priapism called “stuttering” priapism26 suffer recurrent prolonged erections during sleep. The patient awakens with a painful erection that usually subsides, but sometimes only after several hours. Patients with this disorder suffer from sleep deprivation. Stuttering priapism may lead to full-blown ischemic priapism that does not resolve without intervention.
Nonischemic priapism
In nonischemic priapism, the corpora are engorged but not rigid. The condition results from unregulated arterial inflow and thus is not painful and does not result in damage to the corporeal smooth muscle.
Most cases of nonischemic priapism follow blunt perineal trauma or trauma associated with needle insertion into the corpora. This form of priapism is not associated with sickle cell disease. Because tissue damage does not occur, nonischemic or arterial priapism is not considered an emergency.
Treatment guidelines
Differentiating ischemic from nonischemic priapism is usually straightforward, based on the history, physical examination, corporeal blood gases, and duplex ultrasonography.27
Ischemic priapism is an emergency. After needle aspiration of blood from the corpora cavernosa, phenylephrine is diluted with normal saline to a concentration of 100 to 500 µg/mL and is injected in 1-mL amounts repeatedly at 3- to 5-minute intervals until the erection subsides or until a 1-hour time limit is reached. Blood pressure and pulse are monitored during these injections. If aspiration and phenylephrine irrigation fail, surgical shunting is performed.27
Measures to treat sickle cell disease (hydration, oxygen, exchange transfusions) may be employed simultaneously but should never delay aspiration and phenylephrine injections.25
As nonischemic priapism is not considered an emergency, management begins with observation. Patients eventually become dissatisfied with their constant partial erection, and they then present for treatment. Most cases resolve after selective catheterization of the internal pudendal artery and embolization of the fistula with absorbable material. If this fails, surgical exploration with ligation of the vessels leading to the fistula is indicated.
Prevalence in sickle cell trait vs sickle cell disease
Ischemic priapism is uncommon in men with sickle cell trait, but prevalence rates in men with sickle cell disease are as high as 42%.28 In a study of 130 men with sickle cell disease, 35% had a history of prolonged ischemic priapism, 72% had a history of stuttering priapism, and 75% of men with stuttering priapism had their first episode before age 20.29
Rates of erectile dysfunction increase with the duration of ischemic episodes and range from 20% to 90%.28,30 In childhood, sickle cell disease accounts for 63% of the cases of ischemic priapism, and in adults it accounts for 23% of cases.31
Take-home messages
- Sickle cell disease accounts for two-thirds of cases of ischemic priapism in children, and one-fourth of adult cases.
- Ischemic priapism is a medical emergency.
- Treatment with aspiration and phenylephrine injections should begin immediately and should not await treatment measures for sickle cell disease (hydration, oxygen, exchange transfusions).
OTHER UROLOGIC COMPLICATIONS OF SICKLE CELL DISEASE
Other urologic complications of sickle cell trait and sickle cell disease include microscopic hematuria, gross hematuria, and renal colic. A formal evaluation of any patient with persistent microscopic hematuria or gross hematuria should consist of urinalysis, computed tomography, and cystoscopy. This approach assesses the upper and lower genitourinary system for treatable causes. Renal ultrasonography can be used instead of computed tomography but tends to provide less information.
Special considerations
In patients with sickle cell trait and sickle cell disease, chronic hypoxia and subsequent sickling of erythrocytes in the renal medulla can lead to papillary hypertrophy and papillary necrosis. In papillary hypertrophy, friable blood vessels can rupture, resulting in microscopic and gross hematuria. In papillary necrosis, the papilla can slough off and become lodged in the ureter.
Nevertheless, hematuria and renal colic in patients with sickle cell disease or trait are most often attributable to common causes such as infection and stones. A finding of hydronephrosis in the absence of a stone, however, suggests obstruction due to a clot or a sloughed papilla. Ureteroscopy, fulguration, and ureteral stent placement can stop the bleeding and alleviate obstruction in these cases.
Renal medullary carcinoma
Another important reason to order imaging in patients with sickle cell disease or trait who present with urologic symptoms is to rule out renal medullary carcinoma, a rare but aggressive cancer that arises from the collecting duct epithelium. This cancer is twice as likely to occur in males than in females; it has been reported in patients ranging in age from 10 to 40, with a median age at presentation of 26.32
When patients present with symptomatic renal medullary cancer, in most cases the cancer has already metastasized.
On computed tomography, the tumor tends to occupy a central location in the kidney and appears to infiltrate and replace adjacent kidney tissue. Retroperitoneal lymphadenopathy and metastasis are common.
Treatment typically entails radical nephrectomy, chemotherapy, and in some circumstances, radiotherapy. Case reports have shown promising tumor responses to carboplatin and paclitaxel regimens.33,34 Also, a low threshold for imaging in patients with sickle cell disease and trait may increase the odds of early detection of this aggressive cancer.
- Centers for Disease Control and Prevention (CDC). Sickle cell disease (SCD). Data and statistics. www.cdc.gov/ncbddd/sicklecell/data.html. Accessed August 18, 2015.
- Paulin L, Itano HA, Singer SJ, Wells IC. Sickle cell anemia, a molecular disease. Science 1949; 110:543–548.
- Powars DR, Chan LS, Hiti A, Ramicone E, Johnson C. Outcome of sickle cell anemia: a 4-decade observational study of 1056 patients. Medicine (Baltimore) 2005; 84:363–376.
- Haymann JP, Stankovic K, Levy P, et al. Glomerular hyperfiltration in adult sickle cell anemia: a frequent hemolysis associated feature. Clin J Am Soc Nephrol 2010; 5:756–761.
- da Silva GB Jr, Libório AB, Daher Ede F. New insights on pathophysiology, clinical manifestations, diagnosis, and treatment of sickle cell nephropathy. Ann Hematol 2011; 90:1371–1379.
- Emokpae MA, Uadia PO, Gadzama AA. Correlation of oxidative stress and inflammatory markers with the severity of sickle cell nephropathy. Ann Afr Med 2010; 9:141–146.
- Chirico EN, Pialoux V. Role of oxidative stress in the pathogenesis of sickle cell disease. IUBMB Life 2012; 64:72–80.
- Datta V, Ayengar JR, Karpate S, Chaturvedi P. Microalbuminuria as a predictor of early glomerular injury in children with sickle cell disease. Indian J Pediatr 2003; 70:307–309.
- Falk RJ, Scheinman J, Phillips G, Orringer E, Johnson A, Jennette JC. Prevalence and pathologic features of sickle cell nephropathy and response to inhibition of angiotensin-converting enzyme. N Engl J Med 1992; 326:910–915.
- Maigne G, Ferlicot S, Galacteros F, et al. Glomerular lesions in patients with sickle cell disease. Medicine (Baltimore) 2010; 89:18–27.
- Sharpe CC, Thein SL. Sickle cell nephropathy—a practical approach. Br J Haematol 2011; 155:287–297.
- Batlle D, Itsarayoungyuen K, Arruda JA, Kurtzman NA. Hyperkalemic hyperchloremic metabolic acidosis in sickle cell hemoglobinopathies. Am J Med 1982; 72:188–192.
- Sklar AH, Perez JC, Harp RJ, Caruana RJ. Acute renal failure in sickle cell anemia. Int J Artif Organs 1990; 13:347–351.
- Powars DR, Elliott-Mills DD, Chan L, et al. Chronic renal failure in sickle cell disease: risk factors, clinical course, and mortality. Ann Intern Med 1991; 115:614–620.
- McClellan AC, Luthi JC, Lynch JR, et al. High one year mortality in adults with sickle cell disease and end-stage renal disease. Br J Haematol 2012; 159:360–367.
- Bodas P, Huang A, O Riordan MA, Sedor JR, Dell KM. The prevalence of hypertension and abnormal kidney function in children with sickle cell disease—a cross sectional review. BMC Nephrol 2013; 14:237.
- Asnani MR, Lynch O, Reid ME. Determining glomerular filtration rate in homozygous sickle cell disease: utility of serum creatinine based estimating equations. PLoS One 2013; 8:e69922.
- Arlet JB, Ribeil JA, Chatellier G, et al. Determination of the best method to estimate glomerular filtration rate from serum creatinine in adult patients with sickle cell disease: a prospective observational cohort study. BMC Nephrol 2012; 13:83.
- McKie KT, Hanevold CD, Hernandez C, Waller JL, Ortiz L, McKie KM. Prevalence, prevention, and treatment of microalbuminuria and proteinuria in children with sickle cell disease. J Pediatr Hematol Oncol 2007; 29:140–144.
- Laurin LP, Nachman PH, Desai PC, Ataga KI, Derebail VK. Hydroxyurea is associated with lower prevalence of albuminuria in adults with sickle cell disease. Nephrol Dial Transplant 2014; 29:1211–1218.
- Aygun B, Mortier NA, Smeltzer MP, Shulkin BL, Hankins JS, Ware RE. Hydroxyurea treatment decreases glomerular hyperfiltration in children with sickle cell anemia. Am J Hematol 2013; 88:116–119.
- Huang E, Parke C, Mehrnia A, et al. Improved survival among sickle cell kidney transplant recipients in the recent era. Nephrol Dial Transplant 2013; 28:1039–1046.
- Klein EA, Montague DK, Steiger E. Priapism associated with the use of intravenous fat emulsion: case reports and postulated pathogenesis. J Urol May 1985; 133:857–859.
- Thuret I, Bardakdjian J, Badens C, et al. Priapism following splenectomy in an unstable hemoglobin: hemoglobin Olmsted beta 141 (H19) Leu-->Arg. Am J Hematol 1996; 51:133–136.
- Montague DK, Angermeier KW. Corporeal excavation: new technique for penile prosthesis implantation in men with severe corporeal fibrosis. Urology 2006; 67:1072–1075.
- Levey HR, Kutlu O, Bivalacqua TJ. Medical management of ischemic stuttering priapism: a contemporary review of the literature. Asian J Androl 2012; 14:156–163.
- Montague DK, Jarow J, Broderick GA, et al; Members of the Erectile Dysfunction Guideline Update Panel; American Urological Association. American Urological Association guideline on the management of priapism. J Urol 2003; 170:1318–1324.
- Emond AM, Holman R, Hayes RJ, Serjeant GR. Priapism and impotence in homozygous sickle cell disease. Arch Intern Med 1980; 140:1434–1437.
- Adeyoju AB, Olujohungbe AB, Morris J, et al. Priapism in sickle-cell disease; incidence, risk factors and complications—an international multicentre study. BJU Int 2002; 90:898–902.
- Pryor J, Akkus E, Alter G, et al. Priapism. J Sex Med 2004; 1:116–120.
- Nelson JH, 3rd, Winter CC. Priapism: evolution of management in 48 patients in a 22-year series. J Urol 1977; 117:455–458.
- Liu Q, Galli S, Srinivasan R, Linehan WM, Tsokos M, Merino MJ. Renal medullary carcinoma: molecular, immunohistochemistry, and morphologic correlation. Am J Surg Pathol 2013; 37:368–374.
- Gangireddy VG, Liles GB, Sostre GD, Coleman T. Response of metastatic renal medullary carcinoma to carboplatinum and Paclitaxel chemotherapy. Clin Genitourin Cancer 2012; 10:134–139.
- Walsh AM, Fiveash JB, Reddy AT, Friedman GK. Response to radiation in renal medullary carcinoma. Rare Tumors 2011; 3:e32.
Sickle cell disease is a common genetic disorder in the United States that disproportionately affects people of African ancestry. The characteristic sickling of red blood cells under conditions of reduced oxygen tension leads to intravascular hemolysis and vaso-occlusive events, which in turn cause tissue ischemia-reperfusion injury affecting multiple organs, including the genitourinary system.1–3
In this paper, we review the genitourinary effects of sickle cell disease, focusing on sickle cell nephropathy, priapism, and renal medullary carcinoma.
THE WIDE-RANGING EFFECTS OF SICKLE CELL DISEASE
In the United States, sickle cell disease affects 1 of every 500 blacks and 1 of every 36,000 Hispanics.1 The term describes hemoglobinopathies associated with sickling of red blood cells.
Sickling of red blood cells results from a single base-pair change in the beta-globin gene from glutamic acid to valine at position 6, causing abnormal hemoglobin (hemoglobin S), which polymerizes under conditions of reduced oxygen tension and alters the biconcave disk shape into a rigid, irregular, unstable cell. The sickle-shaped cells are prone to intravascular hemolysis,2 causing intermittent vaso-occlusive events that result in tissue ischemia-reperfusion injury. Genitourinary problems include impaired ability to concentrate urine, hematuria, renal medullary carcinoma, and increased frequency of urinary tract infection.
SICKLE CELL NEPHROPATHY
The kidney is one of the most frequently affected organs in sickle cell disease. Renal manifestations begin to appear in early childhood, with impaired medullary concentrating ability and ischemic damage to the tubular cells caused by sickling within the vasa recta renis precipitated by the acidic, hypoxic, and hypertonic environment in the renal medulla.
As in early diabetic nephropathy, renal blood flow is enhanced and the glomerular filtration rate (GFR) is increased. Increased cardiac output as a result of anemia, localized release of prostaglandins, and a hypoxia-induced increase in nitric oxide synthesis all play a role in the increase in GFR.4,5
Oxidative stress, an increase in markers of inflammation, and local activation of the renin-angiotensin system contribute to renal damage in sickle cell disease.5–7 The resulting hyperfiltration injury leads to microalbuminuria, which occurs in 20% to 40% of children with sickle cell anemia8,9 and in as many as 60% of adults.
The glomerular lesions associated with sickle cell disease vary from glomerulopathy in the early stages to secondary focal segmental glomerulosclerosis, membranoproliferative glomerulonephritis, and glomerular thrombotic microangiopathy.10
Clinical presentations and workup
Clinical presentations are not limited to glomerular disease but include hyperchloremic metabolic acidosis and hyperkalemia resulting from defects in potassium secretion and renal acidification.
Hyperphosphatemia—a result of increased reabsorption of phosphorus, increased secretion of uric acid, and increased creatinine clearance—is seen in patients with sickle cell disease.11,12 About 10% of patients can develop an acute kidney injury as a result of volume depletion, rhabdomyolysis, renal vein thrombosis, papillary necrosis, and urinary tract obstruction secondary to blood clots.11,13
Up to 30% of adult patients with sickle cell disease develop chronic kidney disease. Predictors include severe anemia, hypertension, proteinuria, nephrotic syndrome, and microscopic hematuria.14 From 4% to 12% of patients go on to develop end-stage renal disease, but with a 1-year mortality rate three times higher than in patients without sickle cell disease.15
In general, patients with sickle cell anemia have blood pressures below those of age- and sex-matched individuals, but elevated blood pressure and low GFR are not uncommon in affected children. In a cohort of 48 children ages 3 to 18, 8.3% had an estimated GFR less than 90 mL/min/1.73 m2, and 16.7% had elevated blood pressure (prehypertension and hypertension).16
In patients with sickle cell disease, evaluation of proteinuria, hematuria, hypertension, and renal failure should take into consideration the unique renal physiologic and pathologic processes involved. Recent evidence17,18 suggests that the Chronic Kidney Disease Epidemiology Collaboration equation provides a better estimate of GFR than the Modification of Diet in Renal Disease and Cockcroft-Gault equations, although all three creatinine-based methods overestimate GFR in patients with sickle cell disease when compared with GFR measured with technetium-99m-labeled diethylenetriamine penta-acetic acid renal scanning.
Treatment options
Treatment of sickle cell nephropathy includes adequate fluid intake (given the loss of concentrating ability), adequate blood pressure control, use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers (ARBs) in patients who have microalbuminuria or proteinuria (or both)9,11,19 and hydroxyurea. Treatment with enalapril has been shown to decrease proteinuria in patients with sickle cell nephropathy.9 In a cohort of children with sickle cell disease, four of nine patients treated with an ACE inhibitor developed hyperkalemia, leading to discontinuation of the drug in three patients.9
ACE inhibitors and ARBs must be used cautiously in these patients because they have defects in potassium secretion. Hydroxyurea has also been shown to decrease hyperfiltration and microalbuminuria in recent studies,20,21 and this could protect against the development of overt nephropathy.
Higher mortality rates have been reported in patients with sickle cell disease who developed end-stage renal disease than in patients with end-stage renal disease without sickle cell disease. Sickle cell disease also increases the risk of pulmonary hypertension and the vaso-occlusive complication known as acute chest syndrome, contributing to increased mortality rates. Of note, in a study that looked at the association between mortality rates and pre-end-stage care of renal disease using data from the Centers for Medicare and Medicaid Services, patients with sickle cell disease who had had predialysis nephrology care had lower mortality rates.15
Treatments for end-stage renal disease are also effective in patients with sickle cell disease and include hemodialysis, peritoneal dialysis, and renal transplantation. Data from the Organ Procurement and Transplantation Network and the United Network for Organ Sharing show that from 2000 to 2011, African American kidney recipients with sickle cell disease had better survival rates than patients who had undergone transplantation from 1988 to 1999, although rates of long-term survival and graft survival were lower than in transplant recipients with other diagnoses.22
It is important to note that complications as a result of vaso-occlusive events and thrombosis can lead to graft loss; therefore, sickle cell crisis after transplantation requires careful management.
Take-home messages
- Loss of urine-concentrating ability and hyperfiltration are the earliest pathologic changes in sickle cell disease.
- Microalbuminuria as seen in diabetic nephropathy is the earliest manifestation of sickle cell nephropathy, and the prevalence increases as these patients get older and live longer.
- ACE inhibitors or ARBs should be used with caution, given the heightened risk of hyperkalemia in sickle cell disease.
- Recent results with hydroxyurea in decreasing hyperfiltration and microalbuminuria are encouraging.
- Early referral for predialysis nephrologic care is needed in sickle cell patients with chronic kidney disease.
PRIAPISM IN SICKLE CELL DISEASE
Priapism was formerly defined as a full, painful erection lasting more than 4 hours and unrelated to sexual stimulation or orgasm. But priapism is now recognized as two separate disorders—ischemic (veno-occlusive, low-flow) priapism and nonischemic (arterial, high-flow) priapism. The new definition includes both disorders: ie, a full or partial erection lasting more than 4 hours and unrelated to sexual stimulation or orgasm.
Ischemic priapism
Hematologic disorders are major contributors to ischemic priapism and include sickle cell disease, multiple myeloma, fat emboli (hyperalimentation),23 glucose-6-phosphate dehydrogenase deficiency, and hemoglobin Olmsted variant.24
Ischemic priapism is often seen in sickle cell disease and is considered an emergency. It is characterized by an abnormally rigid erection not involving the glans penis. Entrapment of blood in the corpora cavernosa leads to hypoxia, hypercarbia, and acidosis, which in turn leads to a painful compartment syndrome that, if untreated, results in smooth muscle necrosis and subsequent fibrosis. The results are a smaller penis and erectile dysfunction that is unresponsive to any treatment other than implantation of a penile prosthesis. However, scarring of the corpora cavernosa can make this procedure exceedingly difficult, requiring advanced techniques such as corporeal excavation.25
Men with a subtype of ischemic priapism called “stuttering” priapism26 suffer recurrent prolonged erections during sleep. The patient awakens with a painful erection that usually subsides, but sometimes only after several hours. Patients with this disorder suffer from sleep deprivation. Stuttering priapism may lead to full-blown ischemic priapism that does not resolve without intervention.
Nonischemic priapism
In nonischemic priapism, the corpora are engorged but not rigid. The condition results from unregulated arterial inflow and thus is not painful and does not result in damage to the corporeal smooth muscle.
Most cases of nonischemic priapism follow blunt perineal trauma or trauma associated with needle insertion into the corpora. This form of priapism is not associated with sickle cell disease. Because tissue damage does not occur, nonischemic or arterial priapism is not considered an emergency.
Treatment guidelines
Differentiating ischemic from nonischemic priapism is usually straightforward, based on the history, physical examination, corporeal blood gases, and duplex ultrasonography.27
Ischemic priapism is an emergency. After needle aspiration of blood from the corpora cavernosa, phenylephrine is diluted with normal saline to a concentration of 100 to 500 µg/mL and is injected in 1-mL amounts repeatedly at 3- to 5-minute intervals until the erection subsides or until a 1-hour time limit is reached. Blood pressure and pulse are monitored during these injections. If aspiration and phenylephrine irrigation fail, surgical shunting is performed.27
Measures to treat sickle cell disease (hydration, oxygen, exchange transfusions) may be employed simultaneously but should never delay aspiration and phenylephrine injections.25
As nonischemic priapism is not considered an emergency, management begins with observation. Patients eventually become dissatisfied with their constant partial erection, and they then present for treatment. Most cases resolve after selective catheterization of the internal pudendal artery and embolization of the fistula with absorbable material. If this fails, surgical exploration with ligation of the vessels leading to the fistula is indicated.
Prevalence in sickle cell trait vs sickle cell disease
Ischemic priapism is uncommon in men with sickle cell trait, but prevalence rates in men with sickle cell disease are as high as 42%.28 In a study of 130 men with sickle cell disease, 35% had a history of prolonged ischemic priapism, 72% had a history of stuttering priapism, and 75% of men with stuttering priapism had their first episode before age 20.29
Rates of erectile dysfunction increase with the duration of ischemic episodes and range from 20% to 90%.28,30 In childhood, sickle cell disease accounts for 63% of the cases of ischemic priapism, and in adults it accounts for 23% of cases.31
Take-home messages
- Sickle cell disease accounts for two-thirds of cases of ischemic priapism in children, and one-fourth of adult cases.
- Ischemic priapism is a medical emergency.
- Treatment with aspiration and phenylephrine injections should begin immediately and should not await treatment measures for sickle cell disease (hydration, oxygen, exchange transfusions).
OTHER UROLOGIC COMPLICATIONS OF SICKLE CELL DISEASE
Other urologic complications of sickle cell trait and sickle cell disease include microscopic hematuria, gross hematuria, and renal colic. A formal evaluation of any patient with persistent microscopic hematuria or gross hematuria should consist of urinalysis, computed tomography, and cystoscopy. This approach assesses the upper and lower genitourinary system for treatable causes. Renal ultrasonography can be used instead of computed tomography but tends to provide less information.
Special considerations
In patients with sickle cell trait and sickle cell disease, chronic hypoxia and subsequent sickling of erythrocytes in the renal medulla can lead to papillary hypertrophy and papillary necrosis. In papillary hypertrophy, friable blood vessels can rupture, resulting in microscopic and gross hematuria. In papillary necrosis, the papilla can slough off and become lodged in the ureter.
Nevertheless, hematuria and renal colic in patients with sickle cell disease or trait are most often attributable to common causes such as infection and stones. A finding of hydronephrosis in the absence of a stone, however, suggests obstruction due to a clot or a sloughed papilla. Ureteroscopy, fulguration, and ureteral stent placement can stop the bleeding and alleviate obstruction in these cases.
Renal medullary carcinoma
Another important reason to order imaging in patients with sickle cell disease or trait who present with urologic symptoms is to rule out renal medullary carcinoma, a rare but aggressive cancer that arises from the collecting duct epithelium. This cancer is twice as likely to occur in males than in females; it has been reported in patients ranging in age from 10 to 40, with a median age at presentation of 26.32
When patients present with symptomatic renal medullary cancer, in most cases the cancer has already metastasized.
On computed tomography, the tumor tends to occupy a central location in the kidney and appears to infiltrate and replace adjacent kidney tissue. Retroperitoneal lymphadenopathy and metastasis are common.
Treatment typically entails radical nephrectomy, chemotherapy, and in some circumstances, radiotherapy. Case reports have shown promising tumor responses to carboplatin and paclitaxel regimens.33,34 Also, a low threshold for imaging in patients with sickle cell disease and trait may increase the odds of early detection of this aggressive cancer.
Sickle cell disease is a common genetic disorder in the United States that disproportionately affects people of African ancestry. The characteristic sickling of red blood cells under conditions of reduced oxygen tension leads to intravascular hemolysis and vaso-occlusive events, which in turn cause tissue ischemia-reperfusion injury affecting multiple organs, including the genitourinary system.1–3
In this paper, we review the genitourinary effects of sickle cell disease, focusing on sickle cell nephropathy, priapism, and renal medullary carcinoma.
THE WIDE-RANGING EFFECTS OF SICKLE CELL DISEASE
In the United States, sickle cell disease affects 1 of every 500 blacks and 1 of every 36,000 Hispanics.1 The term describes hemoglobinopathies associated with sickling of red blood cells.
Sickling of red blood cells results from a single base-pair change in the beta-globin gene from glutamic acid to valine at position 6, causing abnormal hemoglobin (hemoglobin S), which polymerizes under conditions of reduced oxygen tension and alters the biconcave disk shape into a rigid, irregular, unstable cell. The sickle-shaped cells are prone to intravascular hemolysis,2 causing intermittent vaso-occlusive events that result in tissue ischemia-reperfusion injury. Genitourinary problems include impaired ability to concentrate urine, hematuria, renal medullary carcinoma, and increased frequency of urinary tract infection.
SICKLE CELL NEPHROPATHY
The kidney is one of the most frequently affected organs in sickle cell disease. Renal manifestations begin to appear in early childhood, with impaired medullary concentrating ability and ischemic damage to the tubular cells caused by sickling within the vasa recta renis precipitated by the acidic, hypoxic, and hypertonic environment in the renal medulla.
As in early diabetic nephropathy, renal blood flow is enhanced and the glomerular filtration rate (GFR) is increased. Increased cardiac output as a result of anemia, localized release of prostaglandins, and a hypoxia-induced increase in nitric oxide synthesis all play a role in the increase in GFR.4,5
Oxidative stress, an increase in markers of inflammation, and local activation of the renin-angiotensin system contribute to renal damage in sickle cell disease.5–7 The resulting hyperfiltration injury leads to microalbuminuria, which occurs in 20% to 40% of children with sickle cell anemia8,9 and in as many as 60% of adults.
The glomerular lesions associated with sickle cell disease vary from glomerulopathy in the early stages to secondary focal segmental glomerulosclerosis, membranoproliferative glomerulonephritis, and glomerular thrombotic microangiopathy.10
Clinical presentations and workup
Clinical presentations are not limited to glomerular disease but include hyperchloremic metabolic acidosis and hyperkalemia resulting from defects in potassium secretion and renal acidification.
Hyperphosphatemia—a result of increased reabsorption of phosphorus, increased secretion of uric acid, and increased creatinine clearance—is seen in patients with sickle cell disease.11,12 About 10% of patients can develop an acute kidney injury as a result of volume depletion, rhabdomyolysis, renal vein thrombosis, papillary necrosis, and urinary tract obstruction secondary to blood clots.11,13
Up to 30% of adult patients with sickle cell disease develop chronic kidney disease. Predictors include severe anemia, hypertension, proteinuria, nephrotic syndrome, and microscopic hematuria.14 From 4% to 12% of patients go on to develop end-stage renal disease, but with a 1-year mortality rate three times higher than in patients without sickle cell disease.15
In general, patients with sickle cell anemia have blood pressures below those of age- and sex-matched individuals, but elevated blood pressure and low GFR are not uncommon in affected children. In a cohort of 48 children ages 3 to 18, 8.3% had an estimated GFR less than 90 mL/min/1.73 m2, and 16.7% had elevated blood pressure (prehypertension and hypertension).16
In patients with sickle cell disease, evaluation of proteinuria, hematuria, hypertension, and renal failure should take into consideration the unique renal physiologic and pathologic processes involved. Recent evidence17,18 suggests that the Chronic Kidney Disease Epidemiology Collaboration equation provides a better estimate of GFR than the Modification of Diet in Renal Disease and Cockcroft-Gault equations, although all three creatinine-based methods overestimate GFR in patients with sickle cell disease when compared with GFR measured with technetium-99m-labeled diethylenetriamine penta-acetic acid renal scanning.
Treatment options
Treatment of sickle cell nephropathy includes adequate fluid intake (given the loss of concentrating ability), adequate blood pressure control, use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers (ARBs) in patients who have microalbuminuria or proteinuria (or both)9,11,19 and hydroxyurea. Treatment with enalapril has been shown to decrease proteinuria in patients with sickle cell nephropathy.9 In a cohort of children with sickle cell disease, four of nine patients treated with an ACE inhibitor developed hyperkalemia, leading to discontinuation of the drug in three patients.9
ACE inhibitors and ARBs must be used cautiously in these patients because they have defects in potassium secretion. Hydroxyurea has also been shown to decrease hyperfiltration and microalbuminuria in recent studies,20,21 and this could protect against the development of overt nephropathy.
Higher mortality rates have been reported in patients with sickle cell disease who developed end-stage renal disease than in patients with end-stage renal disease without sickle cell disease. Sickle cell disease also increases the risk of pulmonary hypertension and the vaso-occlusive complication known as acute chest syndrome, contributing to increased mortality rates. Of note, in a study that looked at the association between mortality rates and pre-end-stage care of renal disease using data from the Centers for Medicare and Medicaid Services, patients with sickle cell disease who had had predialysis nephrology care had lower mortality rates.15
Treatments for end-stage renal disease are also effective in patients with sickle cell disease and include hemodialysis, peritoneal dialysis, and renal transplantation. Data from the Organ Procurement and Transplantation Network and the United Network for Organ Sharing show that from 2000 to 2011, African American kidney recipients with sickle cell disease had better survival rates than patients who had undergone transplantation from 1988 to 1999, although rates of long-term survival and graft survival were lower than in transplant recipients with other diagnoses.22
It is important to note that complications as a result of vaso-occlusive events and thrombosis can lead to graft loss; therefore, sickle cell crisis after transplantation requires careful management.
Take-home messages
- Loss of urine-concentrating ability and hyperfiltration are the earliest pathologic changes in sickle cell disease.
- Microalbuminuria as seen in diabetic nephropathy is the earliest manifestation of sickle cell nephropathy, and the prevalence increases as these patients get older and live longer.
- ACE inhibitors or ARBs should be used with caution, given the heightened risk of hyperkalemia in sickle cell disease.
- Recent results with hydroxyurea in decreasing hyperfiltration and microalbuminuria are encouraging.
- Early referral for predialysis nephrologic care is needed in sickle cell patients with chronic kidney disease.
PRIAPISM IN SICKLE CELL DISEASE
Priapism was formerly defined as a full, painful erection lasting more than 4 hours and unrelated to sexual stimulation or orgasm. But priapism is now recognized as two separate disorders—ischemic (veno-occlusive, low-flow) priapism and nonischemic (arterial, high-flow) priapism. The new definition includes both disorders: ie, a full or partial erection lasting more than 4 hours and unrelated to sexual stimulation or orgasm.
Ischemic priapism
Hematologic disorders are major contributors to ischemic priapism and include sickle cell disease, multiple myeloma, fat emboli (hyperalimentation),23 glucose-6-phosphate dehydrogenase deficiency, and hemoglobin Olmsted variant.24
Ischemic priapism is often seen in sickle cell disease and is considered an emergency. It is characterized by an abnormally rigid erection not involving the glans penis. Entrapment of blood in the corpora cavernosa leads to hypoxia, hypercarbia, and acidosis, which in turn leads to a painful compartment syndrome that, if untreated, results in smooth muscle necrosis and subsequent fibrosis. The results are a smaller penis and erectile dysfunction that is unresponsive to any treatment other than implantation of a penile prosthesis. However, scarring of the corpora cavernosa can make this procedure exceedingly difficult, requiring advanced techniques such as corporeal excavation.25
Men with a subtype of ischemic priapism called “stuttering” priapism26 suffer recurrent prolonged erections during sleep. The patient awakens with a painful erection that usually subsides, but sometimes only after several hours. Patients with this disorder suffer from sleep deprivation. Stuttering priapism may lead to full-blown ischemic priapism that does not resolve without intervention.
Nonischemic priapism
In nonischemic priapism, the corpora are engorged but not rigid. The condition results from unregulated arterial inflow and thus is not painful and does not result in damage to the corporeal smooth muscle.
Most cases of nonischemic priapism follow blunt perineal trauma or trauma associated with needle insertion into the corpora. This form of priapism is not associated with sickle cell disease. Because tissue damage does not occur, nonischemic or arterial priapism is not considered an emergency.
Treatment guidelines
Differentiating ischemic from nonischemic priapism is usually straightforward, based on the history, physical examination, corporeal blood gases, and duplex ultrasonography.27
Ischemic priapism is an emergency. After needle aspiration of blood from the corpora cavernosa, phenylephrine is diluted with normal saline to a concentration of 100 to 500 µg/mL and is injected in 1-mL amounts repeatedly at 3- to 5-minute intervals until the erection subsides or until a 1-hour time limit is reached. Blood pressure and pulse are monitored during these injections. If aspiration and phenylephrine irrigation fail, surgical shunting is performed.27
Measures to treat sickle cell disease (hydration, oxygen, exchange transfusions) may be employed simultaneously but should never delay aspiration and phenylephrine injections.25
As nonischemic priapism is not considered an emergency, management begins with observation. Patients eventually become dissatisfied with their constant partial erection, and they then present for treatment. Most cases resolve after selective catheterization of the internal pudendal artery and embolization of the fistula with absorbable material. If this fails, surgical exploration with ligation of the vessels leading to the fistula is indicated.
Prevalence in sickle cell trait vs sickle cell disease
Ischemic priapism is uncommon in men with sickle cell trait, but prevalence rates in men with sickle cell disease are as high as 42%.28 In a study of 130 men with sickle cell disease, 35% had a history of prolonged ischemic priapism, 72% had a history of stuttering priapism, and 75% of men with stuttering priapism had their first episode before age 20.29
Rates of erectile dysfunction increase with the duration of ischemic episodes and range from 20% to 90%.28,30 In childhood, sickle cell disease accounts for 63% of the cases of ischemic priapism, and in adults it accounts for 23% of cases.31
Take-home messages
- Sickle cell disease accounts for two-thirds of cases of ischemic priapism in children, and one-fourth of adult cases.
- Ischemic priapism is a medical emergency.
- Treatment with aspiration and phenylephrine injections should begin immediately and should not await treatment measures for sickle cell disease (hydration, oxygen, exchange transfusions).
OTHER UROLOGIC COMPLICATIONS OF SICKLE CELL DISEASE
Other urologic complications of sickle cell trait and sickle cell disease include microscopic hematuria, gross hematuria, and renal colic. A formal evaluation of any patient with persistent microscopic hematuria or gross hematuria should consist of urinalysis, computed tomography, and cystoscopy. This approach assesses the upper and lower genitourinary system for treatable causes. Renal ultrasonography can be used instead of computed tomography but tends to provide less information.
Special considerations
In patients with sickle cell trait and sickle cell disease, chronic hypoxia and subsequent sickling of erythrocytes in the renal medulla can lead to papillary hypertrophy and papillary necrosis. In papillary hypertrophy, friable blood vessels can rupture, resulting in microscopic and gross hematuria. In papillary necrosis, the papilla can slough off and become lodged in the ureter.
Nevertheless, hematuria and renal colic in patients with sickle cell disease or trait are most often attributable to common causes such as infection and stones. A finding of hydronephrosis in the absence of a stone, however, suggests obstruction due to a clot or a sloughed papilla. Ureteroscopy, fulguration, and ureteral stent placement can stop the bleeding and alleviate obstruction in these cases.
Renal medullary carcinoma
Another important reason to order imaging in patients with sickle cell disease or trait who present with urologic symptoms is to rule out renal medullary carcinoma, a rare but aggressive cancer that arises from the collecting duct epithelium. This cancer is twice as likely to occur in males than in females; it has been reported in patients ranging in age from 10 to 40, with a median age at presentation of 26.32
When patients present with symptomatic renal medullary cancer, in most cases the cancer has already metastasized.
On computed tomography, the tumor tends to occupy a central location in the kidney and appears to infiltrate and replace adjacent kidney tissue. Retroperitoneal lymphadenopathy and metastasis are common.
Treatment typically entails radical nephrectomy, chemotherapy, and in some circumstances, radiotherapy. Case reports have shown promising tumor responses to carboplatin and paclitaxel regimens.33,34 Also, a low threshold for imaging in patients with sickle cell disease and trait may increase the odds of early detection of this aggressive cancer.
- Centers for Disease Control and Prevention (CDC). Sickle cell disease (SCD). Data and statistics. www.cdc.gov/ncbddd/sicklecell/data.html. Accessed August 18, 2015.
- Paulin L, Itano HA, Singer SJ, Wells IC. Sickle cell anemia, a molecular disease. Science 1949; 110:543–548.
- Powars DR, Chan LS, Hiti A, Ramicone E, Johnson C. Outcome of sickle cell anemia: a 4-decade observational study of 1056 patients. Medicine (Baltimore) 2005; 84:363–376.
- Haymann JP, Stankovic K, Levy P, et al. Glomerular hyperfiltration in adult sickle cell anemia: a frequent hemolysis associated feature. Clin J Am Soc Nephrol 2010; 5:756–761.
- da Silva GB Jr, Libório AB, Daher Ede F. New insights on pathophysiology, clinical manifestations, diagnosis, and treatment of sickle cell nephropathy. Ann Hematol 2011; 90:1371–1379.
- Emokpae MA, Uadia PO, Gadzama AA. Correlation of oxidative stress and inflammatory markers with the severity of sickle cell nephropathy. Ann Afr Med 2010; 9:141–146.
- Chirico EN, Pialoux V. Role of oxidative stress in the pathogenesis of sickle cell disease. IUBMB Life 2012; 64:72–80.
- Datta V, Ayengar JR, Karpate S, Chaturvedi P. Microalbuminuria as a predictor of early glomerular injury in children with sickle cell disease. Indian J Pediatr 2003; 70:307–309.
- Falk RJ, Scheinman J, Phillips G, Orringer E, Johnson A, Jennette JC. Prevalence and pathologic features of sickle cell nephropathy and response to inhibition of angiotensin-converting enzyme. N Engl J Med 1992; 326:910–915.
- Maigne G, Ferlicot S, Galacteros F, et al. Glomerular lesions in patients with sickle cell disease. Medicine (Baltimore) 2010; 89:18–27.
- Sharpe CC, Thein SL. Sickle cell nephropathy—a practical approach. Br J Haematol 2011; 155:287–297.
- Batlle D, Itsarayoungyuen K, Arruda JA, Kurtzman NA. Hyperkalemic hyperchloremic metabolic acidosis in sickle cell hemoglobinopathies. Am J Med 1982; 72:188–192.
- Sklar AH, Perez JC, Harp RJ, Caruana RJ. Acute renal failure in sickle cell anemia. Int J Artif Organs 1990; 13:347–351.
- Powars DR, Elliott-Mills DD, Chan L, et al. Chronic renal failure in sickle cell disease: risk factors, clinical course, and mortality. Ann Intern Med 1991; 115:614–620.
- McClellan AC, Luthi JC, Lynch JR, et al. High one year mortality in adults with sickle cell disease and end-stage renal disease. Br J Haematol 2012; 159:360–367.
- Bodas P, Huang A, O Riordan MA, Sedor JR, Dell KM. The prevalence of hypertension and abnormal kidney function in children with sickle cell disease—a cross sectional review. BMC Nephrol 2013; 14:237.
- Asnani MR, Lynch O, Reid ME. Determining glomerular filtration rate in homozygous sickle cell disease: utility of serum creatinine based estimating equations. PLoS One 2013; 8:e69922.
- Arlet JB, Ribeil JA, Chatellier G, et al. Determination of the best method to estimate glomerular filtration rate from serum creatinine in adult patients with sickle cell disease: a prospective observational cohort study. BMC Nephrol 2012; 13:83.
- McKie KT, Hanevold CD, Hernandez C, Waller JL, Ortiz L, McKie KM. Prevalence, prevention, and treatment of microalbuminuria and proteinuria in children with sickle cell disease. J Pediatr Hematol Oncol 2007; 29:140–144.
- Laurin LP, Nachman PH, Desai PC, Ataga KI, Derebail VK. Hydroxyurea is associated with lower prevalence of albuminuria in adults with sickle cell disease. Nephrol Dial Transplant 2014; 29:1211–1218.
- Aygun B, Mortier NA, Smeltzer MP, Shulkin BL, Hankins JS, Ware RE. Hydroxyurea treatment decreases glomerular hyperfiltration in children with sickle cell anemia. Am J Hematol 2013; 88:116–119.
- Huang E, Parke C, Mehrnia A, et al. Improved survival among sickle cell kidney transplant recipients in the recent era. Nephrol Dial Transplant 2013; 28:1039–1046.
- Klein EA, Montague DK, Steiger E. Priapism associated with the use of intravenous fat emulsion: case reports and postulated pathogenesis. J Urol May 1985; 133:857–859.
- Thuret I, Bardakdjian J, Badens C, et al. Priapism following splenectomy in an unstable hemoglobin: hemoglobin Olmsted beta 141 (H19) Leu-->Arg. Am J Hematol 1996; 51:133–136.
- Montague DK, Angermeier KW. Corporeal excavation: new technique for penile prosthesis implantation in men with severe corporeal fibrosis. Urology 2006; 67:1072–1075.
- Levey HR, Kutlu O, Bivalacqua TJ. Medical management of ischemic stuttering priapism: a contemporary review of the literature. Asian J Androl 2012; 14:156–163.
- Montague DK, Jarow J, Broderick GA, et al; Members of the Erectile Dysfunction Guideline Update Panel; American Urological Association. American Urological Association guideline on the management of priapism. J Urol 2003; 170:1318–1324.
- Emond AM, Holman R, Hayes RJ, Serjeant GR. Priapism and impotence in homozygous sickle cell disease. Arch Intern Med 1980; 140:1434–1437.
- Adeyoju AB, Olujohungbe AB, Morris J, et al. Priapism in sickle-cell disease; incidence, risk factors and complications—an international multicentre study. BJU Int 2002; 90:898–902.
- Pryor J, Akkus E, Alter G, et al. Priapism. J Sex Med 2004; 1:116–120.
- Nelson JH, 3rd, Winter CC. Priapism: evolution of management in 48 patients in a 22-year series. J Urol 1977; 117:455–458.
- Liu Q, Galli S, Srinivasan R, Linehan WM, Tsokos M, Merino MJ. Renal medullary carcinoma: molecular, immunohistochemistry, and morphologic correlation. Am J Surg Pathol 2013; 37:368–374.
- Gangireddy VG, Liles GB, Sostre GD, Coleman T. Response of metastatic renal medullary carcinoma to carboplatinum and Paclitaxel chemotherapy. Clin Genitourin Cancer 2012; 10:134–139.
- Walsh AM, Fiveash JB, Reddy AT, Friedman GK. Response to radiation in renal medullary carcinoma. Rare Tumors 2011; 3:e32.
- Centers for Disease Control and Prevention (CDC). Sickle cell disease (SCD). Data and statistics. www.cdc.gov/ncbddd/sicklecell/data.html. Accessed August 18, 2015.
- Paulin L, Itano HA, Singer SJ, Wells IC. Sickle cell anemia, a molecular disease. Science 1949; 110:543–548.
- Powars DR, Chan LS, Hiti A, Ramicone E, Johnson C. Outcome of sickle cell anemia: a 4-decade observational study of 1056 patients. Medicine (Baltimore) 2005; 84:363–376.
- Haymann JP, Stankovic K, Levy P, et al. Glomerular hyperfiltration in adult sickle cell anemia: a frequent hemolysis associated feature. Clin J Am Soc Nephrol 2010; 5:756–761.
- da Silva GB Jr, Libório AB, Daher Ede F. New insights on pathophysiology, clinical manifestations, diagnosis, and treatment of sickle cell nephropathy. Ann Hematol 2011; 90:1371–1379.
- Emokpae MA, Uadia PO, Gadzama AA. Correlation of oxidative stress and inflammatory markers with the severity of sickle cell nephropathy. Ann Afr Med 2010; 9:141–146.
- Chirico EN, Pialoux V. Role of oxidative stress in the pathogenesis of sickle cell disease. IUBMB Life 2012; 64:72–80.
- Datta V, Ayengar JR, Karpate S, Chaturvedi P. Microalbuminuria as a predictor of early glomerular injury in children with sickle cell disease. Indian J Pediatr 2003; 70:307–309.
- Falk RJ, Scheinman J, Phillips G, Orringer E, Johnson A, Jennette JC. Prevalence and pathologic features of sickle cell nephropathy and response to inhibition of angiotensin-converting enzyme. N Engl J Med 1992; 326:910–915.
- Maigne G, Ferlicot S, Galacteros F, et al. Glomerular lesions in patients with sickle cell disease. Medicine (Baltimore) 2010; 89:18–27.
- Sharpe CC, Thein SL. Sickle cell nephropathy—a practical approach. Br J Haematol 2011; 155:287–297.
- Batlle D, Itsarayoungyuen K, Arruda JA, Kurtzman NA. Hyperkalemic hyperchloremic metabolic acidosis in sickle cell hemoglobinopathies. Am J Med 1982; 72:188–192.
- Sklar AH, Perez JC, Harp RJ, Caruana RJ. Acute renal failure in sickle cell anemia. Int J Artif Organs 1990; 13:347–351.
- Powars DR, Elliott-Mills DD, Chan L, et al. Chronic renal failure in sickle cell disease: risk factors, clinical course, and mortality. Ann Intern Med 1991; 115:614–620.
- McClellan AC, Luthi JC, Lynch JR, et al. High one year mortality in adults with sickle cell disease and end-stage renal disease. Br J Haematol 2012; 159:360–367.
- Bodas P, Huang A, O Riordan MA, Sedor JR, Dell KM. The prevalence of hypertension and abnormal kidney function in children with sickle cell disease—a cross sectional review. BMC Nephrol 2013; 14:237.
- Asnani MR, Lynch O, Reid ME. Determining glomerular filtration rate in homozygous sickle cell disease: utility of serum creatinine based estimating equations. PLoS One 2013; 8:e69922.
- Arlet JB, Ribeil JA, Chatellier G, et al. Determination of the best method to estimate glomerular filtration rate from serum creatinine in adult patients with sickle cell disease: a prospective observational cohort study. BMC Nephrol 2012; 13:83.
- McKie KT, Hanevold CD, Hernandez C, Waller JL, Ortiz L, McKie KM. Prevalence, prevention, and treatment of microalbuminuria and proteinuria in children with sickle cell disease. J Pediatr Hematol Oncol 2007; 29:140–144.
- Laurin LP, Nachman PH, Desai PC, Ataga KI, Derebail VK. Hydroxyurea is associated with lower prevalence of albuminuria in adults with sickle cell disease. Nephrol Dial Transplant 2014; 29:1211–1218.
- Aygun B, Mortier NA, Smeltzer MP, Shulkin BL, Hankins JS, Ware RE. Hydroxyurea treatment decreases glomerular hyperfiltration in children with sickle cell anemia. Am J Hematol 2013; 88:116–119.
- Huang E, Parke C, Mehrnia A, et al. Improved survival among sickle cell kidney transplant recipients in the recent era. Nephrol Dial Transplant 2013; 28:1039–1046.
- Klein EA, Montague DK, Steiger E. Priapism associated with the use of intravenous fat emulsion: case reports and postulated pathogenesis. J Urol May 1985; 133:857–859.
- Thuret I, Bardakdjian J, Badens C, et al. Priapism following splenectomy in an unstable hemoglobin: hemoglobin Olmsted beta 141 (H19) Leu-->Arg. Am J Hematol 1996; 51:133–136.
- Montague DK, Angermeier KW. Corporeal excavation: new technique for penile prosthesis implantation in men with severe corporeal fibrosis. Urology 2006; 67:1072–1075.
- Levey HR, Kutlu O, Bivalacqua TJ. Medical management of ischemic stuttering priapism: a contemporary review of the literature. Asian J Androl 2012; 14:156–163.
- Montague DK, Jarow J, Broderick GA, et al; Members of the Erectile Dysfunction Guideline Update Panel; American Urological Association. American Urological Association guideline on the management of priapism. J Urol 2003; 170:1318–1324.
- Emond AM, Holman R, Hayes RJ, Serjeant GR. Priapism and impotence in homozygous sickle cell disease. Arch Intern Med 1980; 140:1434–1437.
- Adeyoju AB, Olujohungbe AB, Morris J, et al. Priapism in sickle-cell disease; incidence, risk factors and complications—an international multicentre study. BJU Int 2002; 90:898–902.
- Pryor J, Akkus E, Alter G, et al. Priapism. J Sex Med 2004; 1:116–120.
- Nelson JH, 3rd, Winter CC. Priapism: evolution of management in 48 patients in a 22-year series. J Urol 1977; 117:455–458.
- Liu Q, Galli S, Srinivasan R, Linehan WM, Tsokos M, Merino MJ. Renal medullary carcinoma: molecular, immunohistochemistry, and morphologic correlation. Am J Surg Pathol 2013; 37:368–374.
- Gangireddy VG, Liles GB, Sostre GD, Coleman T. Response of metastatic renal medullary carcinoma to carboplatinum and Paclitaxel chemotherapy. Clin Genitourin Cancer 2012; 10:134–139.
- Walsh AM, Fiveash JB, Reddy AT, Friedman GK. Response to radiation in renal medullary carcinoma. Rare Tumors 2011; 3:e32.
KEY POINTS
- Microalbuminuria as seen in diabetic nephropathy is the earliest manifestation of sickle cell nephropathy, and the prevalence increases as these patients get older and live longer.
- Ischemic priapism is a medical emergency. Treatment with aspiration and phenylephrine injections should begin immediately and should not await treatment measures for sickle cell disease.
- In patients with sickle cell trait and sickle cell disease, chronic hypoxia and subsequent sickling of erythrocytes in the renal medulla can lead to papillary hypertrophy and papillary necrosis.
Do healthy patients need routine laboratory testing before elective noncardiac surgery?
A 63-year-old physician is referred for preoperative evaluation before arthroscopic repair of a torn medial meniscus. Her exercise tolerance was excellent before the knee injury, including running without cardiopulmonary symptoms. She is otherwise healthy except for hypertension that is well controlled on amlodipine. She has no known history of liver or kidney disease, bleeding disorder, recent illness, or complications with anesthesia. She inquires as to whether “routine blood testing” is needed before the procedure.
What laboratory studies, if any, should be ordered?
UNLIKELY TO BE OF BENEFIT
Preoperative laboratory testing is not necessary in this otherwise healthy, asymptomatic patient. In the absence of clinical indications, routine testing before elective, low-risk procedures often increases both the cost of care and the potential anxiety caused by abnormal results that provide no substantial benefit to the patient or the clinician.
Preoperative diagnostic tests should be ordered only to identify and optimize disorders that alter the likelihood of perioperative and postoperative adverse outcomes and to establish a baseline assessment. Yet clinicians often perceive that laboratory testing is required by their organization or by other providers.
A comprehensive history and physical examination are the cornerstones of the effective preoperative evaluation. Preferably, the history and examination should guide further testing rather than ordering a battery of standard tests for all patients. However, selective preoperative laboratory testing may be useful in certain situations, such as in patients undergoing high-risk procedures and those with known underlying conditions or factors that may affect operative management (Table 1).
Unfortunately, high-quality evidence for this selective approach is lacking. According to one observational study,1 when laboratory testing is appropriate, it is reasonable to use test results already obtained and normal within the preceding 4 months unless the patient has had an interim change in health status.
Definitions of risk stratification (eg, urgency of surgical procedure, graded risk according to type of operation) and tools such as the Revised Cardiac Risk Index can be found in the 2014 American College of Cardiology/American Heart Association guidelines2 and may be useful to distinguish healthy patients from those with significant comorbidities, as well as to distinguish low-risk, elective procedures from those that impart higher risk.
Professional societies and guidelines in many countries have criticized the habitual practice of extensive, nonselective laboratory testing.3–6 Yet despite lack of evidence of benefit, routine preoperative testing is still often done. At an estimated cost of more than $18 billion in the United States annually,7 preoperative testing deserves attention, especially in this time of ballooning healthcare costs and increased focus on effective and efficient care.
EVIDENCE AND GUIDELINES
Numerous studies have established that routine laboratory testing rarely changes the preoperative management of the patient or improves surgical outcomes. Narr et al8 found that 160 (4%) of 3,782 patients who underwent ambulatory surgery had abnormal test results, and only 10 required treatment. In this study, there was no association between abnormal test results and perioperative management or postoperative adverse events.
In a systematic review, Smetana and Macpherson9 noted that the incidence of laboratory test abnormalities that led to a change in management ranged from 0.1% to 2.6%. Notably, clinicians ignore 30% to 60% of abnormal preoperative laboratory results, a practice that may create additional medicolegal risk.7
Little evidence exists that helps in the development of guidelines for preoperative laboratory testing. Most guidelines are based on expert opinion, case series, and consensus. As an example of the heterogeneity this creates, the American Society of Anesthesiologists, the Ontario Preoperative Testing Group, and the Canadian Anesthesiologists’ Society provide different recommended indications for preoperative laboratory testing in patients with “advanced age” but do not define a clear minimum age for this cohort.10
However, one area that does have substantial data is cataract surgery. Patients in their usual state of health who are to undergo this procedure do not require preoperative testing, a claim supported by high-quality evidence including a 2012 Cochrane systematic review.11
Munro et al5 performed a systematic review of the evidence behind preoperative laboratory testing, concluding that the power of preoperative tests to predict adverse postoperative outcomes in asymptomatic patients is either weak or nonexistent. The National Institute for Health and Clinical Excellence guidelines of 2003,6 the Practice Advisory for Preanesthesia Evaluation of the American Society of Anesthesiologists of 2012,12 the Institute for Clinical Systems Improvement guideline of 2012,13 and a systematic review conducted by Johansson et al14 found no evidence from high-quality studies to support the claim that routine preoperative testing is beneficial in healthy adults undergoing noncardiac surgery, but that certain patient populations may benefit from selective testing.
A randomized controlled trial evaluated the elimination of preoperative testing in patients undergoing low-risk ambulatory surgery and found no difference in perioperative adverse events in the control and intervention arms.15 Similar studies achieved the same results.
The Choosing Wisely campaign
The American Board of Internal Medicine Foundation has partnered with medical specialty societies to create lists of common practice patterns that should be questioned and possibly discontinued. These lists are collectively called the Choosing Wisely campaign (www.choosingwisely.org). Avoiding routine preoperative laboratory testing in patients undergoing low-risk surgery without clinical indications can be found in the lists for the American Society of Anesthesiologists, the American Society for Clinical Pathology, and the Society of General Internal Medicine.
THE POSSIBLE HARMS OF TESTING
The prevalence of unrecognized disease that influences the risk of surgery in healthy patients is low, and thus the predictive value of abnormal test values in these patients is low. This leads to substantial false-positivity, which is of uncertain clinical significance and which may in turn cause a cascade of further testing. Not surprisingly, the probability of an abnormal test result increases dramatically with the number of tests ordered, a fact that magnifies the problem of false-positive results.
The costs and harms associated with testing are both direct and indirect. Direct effects include increased healthcare costs of further testing or potentially unnecessary treatment as well as risk associated with additional testing, though these are not common, as there is a low (< 3%) incidence of a change in preoperative management based on an abnormal test result. Likewise, normal results do not appear to substantially reduce the likelihood of postoperative complications.9
Indirect effects, which are particularly challenging to measure, may include time lost from employment to pursue further evaluation and anxiety surrounding abnormal results.
THE CLINICAL BOTTOM LINE
Based on over 2 decades of data, our 63-year-old patient should not undergo “routine” preoperative laboratory testing before her upcoming elective, low-risk, noncardiac procedure. Her hypertension is well controlled, and she is taking no medications that may lead to clinically significant metabolic derangements or significant changes in surgical outcome. There are no convincing clinical indications for further laboratory investigation. Further, the results are unlikely to affect the preoperative management and rate of adverse events; the direct and indirect costs may be substantial; and there is a small but tangible risk of harm.
Given the myriad factors that influence unnecessary preoperative testing, a focus on systems-level solutions is paramount. Key steps may include creation and adoption of clear and consistent guidelines, development of clinical care pathways, physician education and modification of practice, interdisciplinary communication and information sharing, economic analysis, and outcomes assessment.
- Macpherson DS, Snow R, Lofgren RP. Preoperative screening: value of previous tests. Ann Intern Med 1990; 113:969–973.
- Fleisher LA, Fleischmann KE, Auerbach AD, et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery. Circulation 2014; 130:e278–e333.
- Schein OD, Katz J, Bass EB, et al. The value of routine preoperative medical testing before cataract surgery. Study of medical testing for cataract surgery. N Engl J Med 2000; 342:168–175.
- The Swedish Council on Technology Assessment in Health Care (SBU). Preoperative routines. Stockholm, 1989.
- Munro J, Booth A, Nicholl J. Routine preoperative testing: a systematic review of the evidence. Health Technol Assess 1997; 1:1–62.
- National Institute for Health and Clinical Excellence (NICE). Preoperative tests: The use of routine preoperative tests for elective surgery. London: National Collaborating Centre for Acute Care, 2003.
- Roizen MF. More preoperative assessment by physicians and less by laboratory tests. N Engl J Med 2000; 342:204–205.
- Narr BJ, Hansen TR, Warner MA. Preoperative laboratory screening in healthy Mayo patients: cost-effective elimination of tests and unchanged outcomes. Mayo Clin Proc 1991; 66:155–159.
- Smetana GW, Macpherson DS. The case against routine preoperative laboratory testing. Med Clin North Am 2003; 87:7–40.
- Benarroch-Gampel J, Sheffield KM, Duncan CB, et al. Preoperative laboratory testing in patients undergoing elective, low-risk ambulatory surgery. Ann Surg 2012; 256:518–528.
- Keay L, Lindsley K, Tielsch J, Katz J, Schein O. Routine preoperative medical testing for cataract surgery. Cochrane Database Syst Rev 2012; 3:CD007293.
- Committee on Standards and Practice Parameters; Apfelbaum JL, Connis RT, Nickinovich DG, et al. Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology 2012; 116:522–538.
- Institute for Clinical Systems Improvement (ICSI). Health care guideline: preoperative evaluation. 10th ed. Bloomington, MN: Institute for Clinical Systems Improvement; 2012.
- Johansson T, Fritsch G, Flamm M, et al. Effectiveness of non-cardiac preoperative testing in non-cardiac elective surgery: a systematic review. Br J Anaesth 2013; 110:926–939.
- Chung F, Yuan H, Yin L, Vairavanathan S, Wong DT. Elimination of preoperative testing in ambulatory surgery. Anesth Analg 2009; 108:467–475.
A 63-year-old physician is referred for preoperative evaluation before arthroscopic repair of a torn medial meniscus. Her exercise tolerance was excellent before the knee injury, including running without cardiopulmonary symptoms. She is otherwise healthy except for hypertension that is well controlled on amlodipine. She has no known history of liver or kidney disease, bleeding disorder, recent illness, or complications with anesthesia. She inquires as to whether “routine blood testing” is needed before the procedure.
What laboratory studies, if any, should be ordered?
UNLIKELY TO BE OF BENEFIT
Preoperative laboratory testing is not necessary in this otherwise healthy, asymptomatic patient. In the absence of clinical indications, routine testing before elective, low-risk procedures often increases both the cost of care and the potential anxiety caused by abnormal results that provide no substantial benefit to the patient or the clinician.
Preoperative diagnostic tests should be ordered only to identify and optimize disorders that alter the likelihood of perioperative and postoperative adverse outcomes and to establish a baseline assessment. Yet clinicians often perceive that laboratory testing is required by their organization or by other providers.
A comprehensive history and physical examination are the cornerstones of the effective preoperative evaluation. Preferably, the history and examination should guide further testing rather than ordering a battery of standard tests for all patients. However, selective preoperative laboratory testing may be useful in certain situations, such as in patients undergoing high-risk procedures and those with known underlying conditions or factors that may affect operative management (Table 1).
Unfortunately, high-quality evidence for this selective approach is lacking. According to one observational study,1 when laboratory testing is appropriate, it is reasonable to use test results already obtained and normal within the preceding 4 months unless the patient has had an interim change in health status.
Definitions of risk stratification (eg, urgency of surgical procedure, graded risk according to type of operation) and tools such as the Revised Cardiac Risk Index can be found in the 2014 American College of Cardiology/American Heart Association guidelines2 and may be useful to distinguish healthy patients from those with significant comorbidities, as well as to distinguish low-risk, elective procedures from those that impart higher risk.
Professional societies and guidelines in many countries have criticized the habitual practice of extensive, nonselective laboratory testing.3–6 Yet despite lack of evidence of benefit, routine preoperative testing is still often done. At an estimated cost of more than $18 billion in the United States annually,7 preoperative testing deserves attention, especially in this time of ballooning healthcare costs and increased focus on effective and efficient care.
EVIDENCE AND GUIDELINES
Numerous studies have established that routine laboratory testing rarely changes the preoperative management of the patient or improves surgical outcomes. Narr et al8 found that 160 (4%) of 3,782 patients who underwent ambulatory surgery had abnormal test results, and only 10 required treatment. In this study, there was no association between abnormal test results and perioperative management or postoperative adverse events.
In a systematic review, Smetana and Macpherson9 noted that the incidence of laboratory test abnormalities that led to a change in management ranged from 0.1% to 2.6%. Notably, clinicians ignore 30% to 60% of abnormal preoperative laboratory results, a practice that may create additional medicolegal risk.7
Little evidence exists that helps in the development of guidelines for preoperative laboratory testing. Most guidelines are based on expert opinion, case series, and consensus. As an example of the heterogeneity this creates, the American Society of Anesthesiologists, the Ontario Preoperative Testing Group, and the Canadian Anesthesiologists’ Society provide different recommended indications for preoperative laboratory testing in patients with “advanced age” but do not define a clear minimum age for this cohort.10
However, one area that does have substantial data is cataract surgery. Patients in their usual state of health who are to undergo this procedure do not require preoperative testing, a claim supported by high-quality evidence including a 2012 Cochrane systematic review.11
Munro et al5 performed a systematic review of the evidence behind preoperative laboratory testing, concluding that the power of preoperative tests to predict adverse postoperative outcomes in asymptomatic patients is either weak or nonexistent. The National Institute for Health and Clinical Excellence guidelines of 2003,6 the Practice Advisory for Preanesthesia Evaluation of the American Society of Anesthesiologists of 2012,12 the Institute for Clinical Systems Improvement guideline of 2012,13 and a systematic review conducted by Johansson et al14 found no evidence from high-quality studies to support the claim that routine preoperative testing is beneficial in healthy adults undergoing noncardiac surgery, but that certain patient populations may benefit from selective testing.
A randomized controlled trial evaluated the elimination of preoperative testing in patients undergoing low-risk ambulatory surgery and found no difference in perioperative adverse events in the control and intervention arms.15 Similar studies achieved the same results.
The Choosing Wisely campaign
The American Board of Internal Medicine Foundation has partnered with medical specialty societies to create lists of common practice patterns that should be questioned and possibly discontinued. These lists are collectively called the Choosing Wisely campaign (www.choosingwisely.org). Avoiding routine preoperative laboratory testing in patients undergoing low-risk surgery without clinical indications can be found in the lists for the American Society of Anesthesiologists, the American Society for Clinical Pathology, and the Society of General Internal Medicine.
THE POSSIBLE HARMS OF TESTING
The prevalence of unrecognized disease that influences the risk of surgery in healthy patients is low, and thus the predictive value of abnormal test values in these patients is low. This leads to substantial false-positivity, which is of uncertain clinical significance and which may in turn cause a cascade of further testing. Not surprisingly, the probability of an abnormal test result increases dramatically with the number of tests ordered, a fact that magnifies the problem of false-positive results.
The costs and harms associated with testing are both direct and indirect. Direct effects include increased healthcare costs of further testing or potentially unnecessary treatment as well as risk associated with additional testing, though these are not common, as there is a low (< 3%) incidence of a change in preoperative management based on an abnormal test result. Likewise, normal results do not appear to substantially reduce the likelihood of postoperative complications.9
Indirect effects, which are particularly challenging to measure, may include time lost from employment to pursue further evaluation and anxiety surrounding abnormal results.
THE CLINICAL BOTTOM LINE
Based on over 2 decades of data, our 63-year-old patient should not undergo “routine” preoperative laboratory testing before her upcoming elective, low-risk, noncardiac procedure. Her hypertension is well controlled, and she is taking no medications that may lead to clinically significant metabolic derangements or significant changes in surgical outcome. There are no convincing clinical indications for further laboratory investigation. Further, the results are unlikely to affect the preoperative management and rate of adverse events; the direct and indirect costs may be substantial; and there is a small but tangible risk of harm.
Given the myriad factors that influence unnecessary preoperative testing, a focus on systems-level solutions is paramount. Key steps may include creation and adoption of clear and consistent guidelines, development of clinical care pathways, physician education and modification of practice, interdisciplinary communication and information sharing, economic analysis, and outcomes assessment.
A 63-year-old physician is referred for preoperative evaluation before arthroscopic repair of a torn medial meniscus. Her exercise tolerance was excellent before the knee injury, including running without cardiopulmonary symptoms. She is otherwise healthy except for hypertension that is well controlled on amlodipine. She has no known history of liver or kidney disease, bleeding disorder, recent illness, or complications with anesthesia. She inquires as to whether “routine blood testing” is needed before the procedure.
What laboratory studies, if any, should be ordered?
UNLIKELY TO BE OF BENEFIT
Preoperative laboratory testing is not necessary in this otherwise healthy, asymptomatic patient. In the absence of clinical indications, routine testing before elective, low-risk procedures often increases both the cost of care and the potential anxiety caused by abnormal results that provide no substantial benefit to the patient or the clinician.
Preoperative diagnostic tests should be ordered only to identify and optimize disorders that alter the likelihood of perioperative and postoperative adverse outcomes and to establish a baseline assessment. Yet clinicians often perceive that laboratory testing is required by their organization or by other providers.
A comprehensive history and physical examination are the cornerstones of the effective preoperative evaluation. Preferably, the history and examination should guide further testing rather than ordering a battery of standard tests for all patients. However, selective preoperative laboratory testing may be useful in certain situations, such as in patients undergoing high-risk procedures and those with known underlying conditions or factors that may affect operative management (Table 1).
Unfortunately, high-quality evidence for this selective approach is lacking. According to one observational study,1 when laboratory testing is appropriate, it is reasonable to use test results already obtained and normal within the preceding 4 months unless the patient has had an interim change in health status.
Definitions of risk stratification (eg, urgency of surgical procedure, graded risk according to type of operation) and tools such as the Revised Cardiac Risk Index can be found in the 2014 American College of Cardiology/American Heart Association guidelines2 and may be useful to distinguish healthy patients from those with significant comorbidities, as well as to distinguish low-risk, elective procedures from those that impart higher risk.
Professional societies and guidelines in many countries have criticized the habitual practice of extensive, nonselective laboratory testing.3–6 Yet despite lack of evidence of benefit, routine preoperative testing is still often done. At an estimated cost of more than $18 billion in the United States annually,7 preoperative testing deserves attention, especially in this time of ballooning healthcare costs and increased focus on effective and efficient care.
EVIDENCE AND GUIDELINES
Numerous studies have established that routine laboratory testing rarely changes the preoperative management of the patient or improves surgical outcomes. Narr et al8 found that 160 (4%) of 3,782 patients who underwent ambulatory surgery had abnormal test results, and only 10 required treatment. In this study, there was no association between abnormal test results and perioperative management or postoperative adverse events.
In a systematic review, Smetana and Macpherson9 noted that the incidence of laboratory test abnormalities that led to a change in management ranged from 0.1% to 2.6%. Notably, clinicians ignore 30% to 60% of abnormal preoperative laboratory results, a practice that may create additional medicolegal risk.7
Little evidence exists that helps in the development of guidelines for preoperative laboratory testing. Most guidelines are based on expert opinion, case series, and consensus. As an example of the heterogeneity this creates, the American Society of Anesthesiologists, the Ontario Preoperative Testing Group, and the Canadian Anesthesiologists’ Society provide different recommended indications for preoperative laboratory testing in patients with “advanced age” but do not define a clear minimum age for this cohort.10
However, one area that does have substantial data is cataract surgery. Patients in their usual state of health who are to undergo this procedure do not require preoperative testing, a claim supported by high-quality evidence including a 2012 Cochrane systematic review.11
Munro et al5 performed a systematic review of the evidence behind preoperative laboratory testing, concluding that the power of preoperative tests to predict adverse postoperative outcomes in asymptomatic patients is either weak or nonexistent. The National Institute for Health and Clinical Excellence guidelines of 2003,6 the Practice Advisory for Preanesthesia Evaluation of the American Society of Anesthesiologists of 2012,12 the Institute for Clinical Systems Improvement guideline of 2012,13 and a systematic review conducted by Johansson et al14 found no evidence from high-quality studies to support the claim that routine preoperative testing is beneficial in healthy adults undergoing noncardiac surgery, but that certain patient populations may benefit from selective testing.
A randomized controlled trial evaluated the elimination of preoperative testing in patients undergoing low-risk ambulatory surgery and found no difference in perioperative adverse events in the control and intervention arms.15 Similar studies achieved the same results.
The Choosing Wisely campaign
The American Board of Internal Medicine Foundation has partnered with medical specialty societies to create lists of common practice patterns that should be questioned and possibly discontinued. These lists are collectively called the Choosing Wisely campaign (www.choosingwisely.org). Avoiding routine preoperative laboratory testing in patients undergoing low-risk surgery without clinical indications can be found in the lists for the American Society of Anesthesiologists, the American Society for Clinical Pathology, and the Society of General Internal Medicine.
THE POSSIBLE HARMS OF TESTING
The prevalence of unrecognized disease that influences the risk of surgery in healthy patients is low, and thus the predictive value of abnormal test values in these patients is low. This leads to substantial false-positivity, which is of uncertain clinical significance and which may in turn cause a cascade of further testing. Not surprisingly, the probability of an abnormal test result increases dramatically with the number of tests ordered, a fact that magnifies the problem of false-positive results.
The costs and harms associated with testing are both direct and indirect. Direct effects include increased healthcare costs of further testing or potentially unnecessary treatment as well as risk associated with additional testing, though these are not common, as there is a low (< 3%) incidence of a change in preoperative management based on an abnormal test result. Likewise, normal results do not appear to substantially reduce the likelihood of postoperative complications.9
Indirect effects, which are particularly challenging to measure, may include time lost from employment to pursue further evaluation and anxiety surrounding abnormal results.
THE CLINICAL BOTTOM LINE
Based on over 2 decades of data, our 63-year-old patient should not undergo “routine” preoperative laboratory testing before her upcoming elective, low-risk, noncardiac procedure. Her hypertension is well controlled, and she is taking no medications that may lead to clinically significant metabolic derangements or significant changes in surgical outcome. There are no convincing clinical indications for further laboratory investigation. Further, the results are unlikely to affect the preoperative management and rate of adverse events; the direct and indirect costs may be substantial; and there is a small but tangible risk of harm.
Given the myriad factors that influence unnecessary preoperative testing, a focus on systems-level solutions is paramount. Key steps may include creation and adoption of clear and consistent guidelines, development of clinical care pathways, physician education and modification of practice, interdisciplinary communication and information sharing, economic analysis, and outcomes assessment.
- Macpherson DS, Snow R, Lofgren RP. Preoperative screening: value of previous tests. Ann Intern Med 1990; 113:969–973.
- Fleisher LA, Fleischmann KE, Auerbach AD, et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery. Circulation 2014; 130:e278–e333.
- Schein OD, Katz J, Bass EB, et al. The value of routine preoperative medical testing before cataract surgery. Study of medical testing for cataract surgery. N Engl J Med 2000; 342:168–175.
- The Swedish Council on Technology Assessment in Health Care (SBU). Preoperative routines. Stockholm, 1989.
- Munro J, Booth A, Nicholl J. Routine preoperative testing: a systematic review of the evidence. Health Technol Assess 1997; 1:1–62.
- National Institute for Health and Clinical Excellence (NICE). Preoperative tests: The use of routine preoperative tests for elective surgery. London: National Collaborating Centre for Acute Care, 2003.
- Roizen MF. More preoperative assessment by physicians and less by laboratory tests. N Engl J Med 2000; 342:204–205.
- Narr BJ, Hansen TR, Warner MA. Preoperative laboratory screening in healthy Mayo patients: cost-effective elimination of tests and unchanged outcomes. Mayo Clin Proc 1991; 66:155–159.
- Smetana GW, Macpherson DS. The case against routine preoperative laboratory testing. Med Clin North Am 2003; 87:7–40.
- Benarroch-Gampel J, Sheffield KM, Duncan CB, et al. Preoperative laboratory testing in patients undergoing elective, low-risk ambulatory surgery. Ann Surg 2012; 256:518–528.
- Keay L, Lindsley K, Tielsch J, Katz J, Schein O. Routine preoperative medical testing for cataract surgery. Cochrane Database Syst Rev 2012; 3:CD007293.
- Committee on Standards and Practice Parameters; Apfelbaum JL, Connis RT, Nickinovich DG, et al. Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology 2012; 116:522–538.
- Institute for Clinical Systems Improvement (ICSI). Health care guideline: preoperative evaluation. 10th ed. Bloomington, MN: Institute for Clinical Systems Improvement; 2012.
- Johansson T, Fritsch G, Flamm M, et al. Effectiveness of non-cardiac preoperative testing in non-cardiac elective surgery: a systematic review. Br J Anaesth 2013; 110:926–939.
- Chung F, Yuan H, Yin L, Vairavanathan S, Wong DT. Elimination of preoperative testing in ambulatory surgery. Anesth Analg 2009; 108:467–475.
- Macpherson DS, Snow R, Lofgren RP. Preoperative screening: value of previous tests. Ann Intern Med 1990; 113:969–973.
- Fleisher LA, Fleischmann KE, Auerbach AD, et al. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery. Circulation 2014; 130:e278–e333.
- Schein OD, Katz J, Bass EB, et al. The value of routine preoperative medical testing before cataract surgery. Study of medical testing for cataract surgery. N Engl J Med 2000; 342:168–175.
- The Swedish Council on Technology Assessment in Health Care (SBU). Preoperative routines. Stockholm, 1989.
- Munro J, Booth A, Nicholl J. Routine preoperative testing: a systematic review of the evidence. Health Technol Assess 1997; 1:1–62.
- National Institute for Health and Clinical Excellence (NICE). Preoperative tests: The use of routine preoperative tests for elective surgery. London: National Collaborating Centre for Acute Care, 2003.
- Roizen MF. More preoperative assessment by physicians and less by laboratory tests. N Engl J Med 2000; 342:204–205.
- Narr BJ, Hansen TR, Warner MA. Preoperative laboratory screening in healthy Mayo patients: cost-effective elimination of tests and unchanged outcomes. Mayo Clin Proc 1991; 66:155–159.
- Smetana GW, Macpherson DS. The case against routine preoperative laboratory testing. Med Clin North Am 2003; 87:7–40.
- Benarroch-Gampel J, Sheffield KM, Duncan CB, et al. Preoperative laboratory testing in patients undergoing elective, low-risk ambulatory surgery. Ann Surg 2012; 256:518–528.
- Keay L, Lindsley K, Tielsch J, Katz J, Schein O. Routine preoperative medical testing for cataract surgery. Cochrane Database Syst Rev 2012; 3:CD007293.
- Committee on Standards and Practice Parameters; Apfelbaum JL, Connis RT, Nickinovich DG, et al. Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology 2012; 116:522–538.
- Institute for Clinical Systems Improvement (ICSI). Health care guideline: preoperative evaluation. 10th ed. Bloomington, MN: Institute for Clinical Systems Improvement; 2012.
- Johansson T, Fritsch G, Flamm M, et al. Effectiveness of non-cardiac preoperative testing in non-cardiac elective surgery: a systematic review. Br J Anaesth 2013; 110:926–939.
- Chung F, Yuan H, Yin L, Vairavanathan S, Wong DT. Elimination of preoperative testing in ambulatory surgery. Anesth Analg 2009; 108:467–475.
Why do clinicians continue to order ‘routine preoperative tests’ despite the evidence?
Guidelines and practice advisories issued by several medical societies, including the American Society of Anesthesiologists,1 American Heart Association (AHA) and American College of Cardiology (ACC),2 and Society of General Internal Medicine,3 advise against routine preoperative testing for patients undergoing low-risk surgical procedures. Such testing often includes routine blood chemistry, complete blood cell counts, measures of the clotting system, and cardiac stress testing.
In this issue of the Cleveland Clinic Journal of Medicine, Dr. Nathan Houchens reviews the evidence against these measures.4
Despite a substantial body of evidence going back more than 2 decades that includes prospective randomized controlled trials,5–10 physicians continue to order unnecessary, ineffective, and costly tests in the perioperative period.11 The process of abandoning current medical practice—a phenomenon known as medical reversal12—often takes years,13 because it is more difficult to convince physicians to discontinue a current behavior than to implement a new one.14 The study of what makes physicians accept new therapies and abandon old ones began more than half a century ago.15
More recently, Cabana et al16 created a framework to understand why physicians do not follow clinical practice guidelines. Among the reasons are lack of familiarity or agreement with the contents of the guideline, lack of outcome expectancy, inertia of previous practice, and external barriers to implementation.
The rapid proliferation of guidelines in the past 20 years has led to numerous conflicting recommendations, many of which are based primarily on expert opinion.17 Guidelines based solely on randomized trials have also come under fire.18,19
In the case of preoperative testing, the recommendations are generally evidence-based and consistent. Why then do physicians appear to disregard the evidence? We propose several reasons why they might do so.
SOME PHYSICIANS ARE UNFAMILIAR WITH THE EVIDENCE
The complexity of the evidence summarized in guidelines has increased exponentially in the last decade, but physician time to assess the evidence has not increased. For example, the number of references in the executive summary of the ACC/AHA perioperative guidelines increased from 96 in 2002 to 252 in 2014. Most of the recommendations are backed by substantial amounts of high-quality evidence. For example, there are 17 prospective and 13 retrospective studies demonstrating that routine testing with the prothrombin time and the partial thromboplastin time is not helpful in asymptomatic patients.20
Although compliance with medical evidence varies among specialties,21 most physicians do not have time to keep up with the ever-increasing amount of information. Specifically in the area of cardiac risk assessment, there has been a rapid proliferation of tests that can be used to assess cardiac risk.22–28 In a Harris Interactive survey from 2008, physicians reported not applying medical evidence routinely. One-third believed they would do it more if they had the time.29 Without information technology support to provide medical information at the point of care,30 especially in small practices, using evidence may not be practical. Simply making the information available online and not promoting it actively does not improve utilization.31
As a consequence, physicians continue to order unnecessary tests, even though they may not feel confident interpreting the results.32
PHYSICIANS MAY NOT BELIEVE THE EVIDENCE
A lack of transparency in evidence-based guidelines and, sometimes, a lack of flexibility and relevance to clinical practice are important barriers to physicians’ acceptance of and adherence to evidence-based clinical practice guidelines.30
Even experts who write guidelines may not be swayed by the evidence. For example, a randomized prospective trial of almost 6,000 patients reported that coronary artery revascularization before elective major vascular surgery does not affect long-term mortality rates.33 Based on this study, the 2014 ACC/AHA guidelines2 advised against revascularization before noncardiac surgery exclusively to reduce perioperative cardiac events. Yet the same guidelines do recommend assessing for myocardial ischemia in patients with elevated risk and poor or unknown functional capacity, using a pharmacologic stress test. Based on the extent of the stress test abnormalities, coronary angiography and revascularization are then suggested for patients willing to undergo coronary artery bypass grafting (CABG) or percutaneous coronary intervention.2
The 2014 European Society of Cardiology and European Society of Anaesthesiology guidelines directly recommend revascularization before high-risk surgery, depending on the extent of a stress-induced perfusion defect.34 This recommendation relies on data from the Coronary Artery Surgery Study registry, which included almost 25,000 patients who underwent coronary angiography from 1975 through 1979. At a mean follow-up of 4.1 years, 1,961 patients underwent high-risk surgery. In this observational cohort, patients who underwent CABG had a lower risk of death and myocardial infarction after surgery.35 The reliance of medical societies34 on data that are more than 30 years old—when operative mortality rates and the treatment of coronary artery disease have changed substantially in the interim and despite the fact that this study did not test whether preoperative revascularization can reduce postoperative mortality—reflects a certain resistance to accept the results of the more recent and relevant randomized trial.33
Other physicians may also prefer to rely on selective data or to simply defer to guidelines that support their beliefs. Some physicians find that evidence-based guidelines are impractical and rigid and reduce their autonomy.36 For many physicians, trials that use surrogate end points and short-term outcomes are not sufficiently compelling to make them abandon current practice.37 Finally, when members of the guideline committees have financial associations with the pharmaceutical industry, or when corporations interested in the outcomes provide financial support for a trial’s development, the likelihood of a recommendation being trusted and used by physicians is drastically reduced.38
PRACTICING DEFENSIVELY
Even if physicians are familiar with the evidence and believe it, they may choose not to act on it. One reason is fear of litigation.
In court, attorneys can use guidelines as well as articles from medical journals as both exculpatory and inculpatory evidence. But they more frequently rely on the standard of care, or what most physicians would do under similar circumstances. If a patient has a bad outcome, such as a perioperative myocardial infarction or life-threatening bleeding, the defendant may assert that testing was unwarranted because guidelines do not recommend it or because the probability of such an outcome was low. However, because the outcome occurred, the jury may not believe that the probability was low enough not to consider, especially if expert witnesses testify that the standard of care would be to order the test.
In areas of controversy, physicians generally believe that erring on the side of more testing is more defensible in court.39 Indeed, following established practice traditions, learned during residency,11,40 may absolve physicians in negligence claims if the way medical care was delivered is supported by recognized and respected physicians.41
As a consequence, physicians prefer to practice the same way their peers do rather than follow the evidence. Unfortunately, the more procedures physicians perform for low-risk patients, the more likely these tests will become accepted as the legal standard of care.42 In this vicious circle, the new standard of care can increase the risk of litigation for others.43 Although unnecessary testing that leads to harmful invasive tests or procedures can also result in malpractice litigation, physicians may not consider this possibility.
FINANCIAL INCENTIVES
The threat of malpractice litigation provides a negative financial incentive to keep performing unnecessary tests, but there are a number of positive incentives as well.
First, physicians often feel compelled to order tests when they believe that physicians referring the patients want the tests done, or when they fear that not completing the tests could delay or cancel the scheduled surgery.40 Refusing to order the test could result in a loss of future referrals. In contrast, ordering tests allows them to meet expectations, preserve trust, and appear more valuable to referring physicians and their patients.
Insurance companies are complicit in these practices. Paying for unnecessary tests can create direct financial incentives for physicians or institutions that own on-site laboratories or diagnostic imaging equipment. Evidence shows that under those circumstances physicians do order more tests. Self-referral and referral to facilities where physicians have a financial interest is associated with increased healthcare costs.44 In addition to direct revenues for the tests performed, physicians may also bill for test interpretation, follow-up visits, and additional procedures generated from test results.
This may be one explanation why the ordering of cardiac tests (stress testing, echocardiography, vascular ultrasonography) by US physicians varies widely from state to state.45
RECOMMENDATIONS TO REDUCE INAPPROPRIATE TESTING
To counter these influences, we propose a multifaceted intervention that includes the following:
- Establish preoperative clinics staffed by experts. Despite the large volume of potentially relevant evidence, the number of articles directly supporting or refuting preoperative laboratory testing is small enough that physicians who routinely engage in preoperative assessment should easily master the evidence.
- Identify local leaders who can convince colleagues of the evidence. Distribute evidence summaries or guidelines with references to major articles that support each recommendation.
- Work with clinical practice committees to establish new standards of care within the hospital. Establish hospital care paths to dictate and support local standards of care. Measure individual physician performance and offer feedback with the goal of reducing utilization.
- National societies should recommend that insurance companies remove inappropriate financial incentives. If companies deny payment for inappropriate testing, physicians will stop ordering it. Even requirements for preauthorization of tests should reduce utilization. The Choosing Wisely campaign (www.choosingwisely.org) would be a good place to start.
- Committee on Standards and Practice Parameters, Apfelbaum JL, Connis RT, Nickinovich DG, et al. Practice advisory for preanesthesia evaluation. An updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology 2012; 116:522–538.
- Fleisher LA, Fleischmann KE, Auerbach AD, et al; American College of Cardiology and American Heart Association. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol 2014; 64:e77–e137.
- Society of General Internal Medicine. Don’t perform routine pre-operative testing before low-risk surgical procedures. Choosing Wisely. An initiative of the ABIM Foundation. September 12, 2013. www.choosingwisely.org/clinician-lists/society-general-internal-medicine-routine-preoperative-testing-before-low-risk-surgery/. Accessed August 31, 2015.
- Houchens N. Should healthy patients undergoing low-risk, elective, noncardiac surgery undergo routine preoperative laboratory testing? Cleve Clin J Med 2015; 82:664–666.
- Rohrer MJ, Michelotti MC, Nahrwold DL. A prospective evaluation of the efficacy of preoperative coagulation testing. Ann Surg 1988; 208:554–557.
- Eagle KA, Coley CM, Newell JB, et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med 1989; 110:859–866.
- Mangano DT, London MJ, Tubau JF, et al. Dipyridamole thallium-201 scintigraphy as a preoperative screening test. A reexamination of its predictive potential. Study of Perioperative Ischemia Research Group. Circulation 1991; 84:493–502.
- Stratmann HG, Younis LT, Wittry MD, Amato M, Mark AL, Miller DD. Dipyridamole technetium 99m sestamibi myocardial tomography for preoperative cardiac risk stratification before major or minor nonvascular surgery. Am Heart J 1996; 132:536–541.
- Schein OD, Katz J, Bass EB, et al. The value of routine preoperative medical testing before cataract surgery. Study of Medical Testing for Cataract Surgery. N Engl J Med 2000; 342:168–175.
- Hashimoto J, Nakahara T, Bai J, Kitamura N, Kasamatsu T, Kubo A. Preoperative risk stratification with myocardial perfusion imaging in intermediate and low-risk non-cardiac surgery. Circ J 2007; 71:1395–1400.
- Smetana GW. The conundrum of unnecessary preoperative testing. JAMA Intern Med 2015; 175:1359–1361.
- Prasad V, Cifu A. Medical reversal: why we must raise the bar before adopting new technologies. Yale J Biol Med 2011; 84:471–478.
- Tatsioni A, Bonitsis NG, Ioannidis JP. Persistence of contradicted claims in the literature. JAMA 2007; 298:2517–2526.
- Moscucci M. Medical reversal, clinical trials, and the “late” open artery hypothesis in acute myocardial infarction. Arch Intern Med 2011; 171:1643–1644.
- Coleman J, Menzel H, Katz E. Social processes in physicians’ adoption of a new drug. J Chronic Dis 1959; 9:1–19.
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA 1999; 282:1458–1465.
- Tricoci P, Allen JM, Kramer JM, Califf RM, Smith SC Jr. Scientific evidence underlying the ACC/AHA clinical practice guidelines. JAMA 2009; 301:831–841.
- Moher D, Hopewell S, Schulz KF, et al; CONSORT. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. Int J Surg 2012; 10:28–55.
- Gattinoni L, Giomarelli P. Acquiring knowledge in intensive care: merits and pitfalls of randomized controlled trials. Intensive Care Med 2015; 41:1460–1464.
- Levy JH, Szlam F, Wolberg AS, Winkler A. Clinical use of the activated partial thromboplastin time and prothrombin time for screening: a review of the literature and current guidelines for testing. Clin Lab Med 2014; 34:453–477.
- Dale W, Hemmerich J, Moliski E, Schwarze ML, Tung A. Effect of specialty and recent experience on perioperative decision-making for abdominal aortic aneurysm repair. J Am Geriatr Soc 2012; 60:1889–1894.
- Underwood SR, Anagnostopoulos C, Cerqueira M, et al; British Cardiac Society, British Nuclear Cardiology Society, British Nuclear Medicine Society, Royal College of Physicians of London, Royal College of Physicians of London. Myocardial perfusion scintigraphy: the evidence. Eur J Nucl Med Mol Imaging 2004; 31:261–291.
- Das MK, Pellikka PA, Mahoney DW, et al. Assessment of cardiac risk before nonvascular surgery: dobutamine stress echocardiography in 530 patients. J Am Coll Cardiol 2000; 35:1647–1653.
- Meijboom WB, Mollet NR, Van Mieghem CA, et al. Pre-operative computed tomography coronary angiography to detect significant coronary artery disease in patients referred for cardiac valve surgery. J Am Coll Cardiol 2006; 48:1658–1665.
- Russo V, Gostoli V, Lovato L, et al. Clinical value of multidetector CT coronary angiography as a preoperative screening test before non-coronary cardiac surgery. Heart 2007; 93:1591–1598.
- Schuetz GM, Zacharopoulou NM, Schlattmann P, Dewey M. Meta-analysis: noninvasive coronary angiography using computed tomography versus magnetic resonance imaging. Ann Intern Med 2010; 152:167–177.
- Bluemke DA, Achenbach S, Budoff M, et al. Noninvasive coronary artery imaging: magnetic resonance angiography and multidetector computed tomography angiography: a scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention of the Council on Cardiovascular Radiology and Intervention, and the Councils on Clinical Cardiology and Cardiovascular Disease in the Young. Circulation 2008; 118:586–606.
- Nagel E, Lehmkuhl HB, Bocksch W, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation 1999; 99:763–770.
- Taylor H. Physicians’ use of clinical guidelines—and how to increase it. Healthcare News 2008; 8:32–55. www.harrisinteractive.com/vault/HI_HealthCareNews2008Vol8_Iss04.pdf. Accessed August 31, 2015.
- Kenefick H, Lee J, Fleishman V. Improving physician adherence to clinical practice guidelines. Barriers and stragies for change. New England Healthcare Institute, February 2008. www.nehi.net/writable/publication_files/file/cpg_report_final.pdf. Accessed August 31, 2015.
- Williams J, Cheung WY, Price DE, et al. Clinical guidelines online: do they improve compliance? Postgrad Med J 2004; 80:415–419.
- Wians F. Clinical laboratory tests: which, why, and what do the results mean? Lab Medicine 2009; 40:105–113.
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004; 351:2795–2804.
- Kristensen SD, Knuuti J, Saraste A, et al; Authors/Task Force Members. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Eur Heart J 2014; 35:2383–2431.
- Eagle KA, Rihal CS, Mickel MC, Holmes DR, Foster ED, Gersh BJ. Cardiac risk of noncardiac surgery: influence of coronary disease and type of surgery in 3368 operations. CASS Investigators and University of Michigan Heart Care Program. Coronary Artery Surgery Study. Circulation 1997; 96:1882–1887.
- Farquhar CM, Kofa EW, Slutsky JR. Clinicians’ attitudes to clinical practice guidelines: a systematic review. Med J Aust 2002; 177:502–506.
- Prasad V, Cifu A, Ioannidis JP. Reversals of established medical practices: evidence to abandon ship. JAMA 2012; 307:37–38.
- Steinbrook R. Guidance for guidelines. N Engl J Med 2007; 356:331–333.
- Sirovich BE, Woloshin S, Schwartz LM. Too little? Too much? Primary care physicians’ views on US health care: a brief report. Arch Intern Med 2011; 171:1582–1585.
- Brown SR, Brown J. Why do physicians order unnecessary preoperative tests? A qualitative study. Fam Med 2011; 43:338–343.
- LeCraw LL. Use of clinical practice guidelines in medical malpractice litigation. J Oncol Pract 2007; 3:254.
- Studdert DM, Mello MM, Sage WM, et al. Defensive medicine among high-risk specialist physicians in a volatile malpractice environment. JAMA 2005; 293:2609–2617.
- Budetti PP. Tort reform and the patient safety movement: seeking common ground. JAMA 2005; 293:2660–2662.
- Bishop TF, Federman AD, Ross JS. Laboratory test ordering at physician offices with and without on-site laboratories. J Gen Intern Med 2010; 25:1057–1063.
- Rosenthal E. Medical costs rise as retirees winter in Florida. The New York Times, Jan 31, 2015. http://nyti.ms/1vmjfa5. Accessed August 31, 2015.
Guidelines and practice advisories issued by several medical societies, including the American Society of Anesthesiologists,1 American Heart Association (AHA) and American College of Cardiology (ACC),2 and Society of General Internal Medicine,3 advise against routine preoperative testing for patients undergoing low-risk surgical procedures. Such testing often includes routine blood chemistry, complete blood cell counts, measures of the clotting system, and cardiac stress testing.
In this issue of the Cleveland Clinic Journal of Medicine, Dr. Nathan Houchens reviews the evidence against these measures.4
Despite a substantial body of evidence going back more than 2 decades that includes prospective randomized controlled trials,5–10 physicians continue to order unnecessary, ineffective, and costly tests in the perioperative period.11 The process of abandoning current medical practice—a phenomenon known as medical reversal12—often takes years,13 because it is more difficult to convince physicians to discontinue a current behavior than to implement a new one.14 The study of what makes physicians accept new therapies and abandon old ones began more than half a century ago.15
More recently, Cabana et al16 created a framework to understand why physicians do not follow clinical practice guidelines. Among the reasons are lack of familiarity or agreement with the contents of the guideline, lack of outcome expectancy, inertia of previous practice, and external barriers to implementation.
The rapid proliferation of guidelines in the past 20 years has led to numerous conflicting recommendations, many of which are based primarily on expert opinion.17 Guidelines based solely on randomized trials have also come under fire.18,19
In the case of preoperative testing, the recommendations are generally evidence-based and consistent. Why then do physicians appear to disregard the evidence? We propose several reasons why they might do so.
SOME PHYSICIANS ARE UNFAMILIAR WITH THE EVIDENCE
The complexity of the evidence summarized in guidelines has increased exponentially in the last decade, but physician time to assess the evidence has not increased. For example, the number of references in the executive summary of the ACC/AHA perioperative guidelines increased from 96 in 2002 to 252 in 2014. Most of the recommendations are backed by substantial amounts of high-quality evidence. For example, there are 17 prospective and 13 retrospective studies demonstrating that routine testing with the prothrombin time and the partial thromboplastin time is not helpful in asymptomatic patients.20
Although compliance with medical evidence varies among specialties,21 most physicians do not have time to keep up with the ever-increasing amount of information. Specifically in the area of cardiac risk assessment, there has been a rapid proliferation of tests that can be used to assess cardiac risk.22–28 In a Harris Interactive survey from 2008, physicians reported not applying medical evidence routinely. One-third believed they would do it more if they had the time.29 Without information technology support to provide medical information at the point of care,30 especially in small practices, using evidence may not be practical. Simply making the information available online and not promoting it actively does not improve utilization.31
As a consequence, physicians continue to order unnecessary tests, even though they may not feel confident interpreting the results.32
PHYSICIANS MAY NOT BELIEVE THE EVIDENCE
A lack of transparency in evidence-based guidelines and, sometimes, a lack of flexibility and relevance to clinical practice are important barriers to physicians’ acceptance of and adherence to evidence-based clinical practice guidelines.30
Even experts who write guidelines may not be swayed by the evidence. For example, a randomized prospective trial of almost 6,000 patients reported that coronary artery revascularization before elective major vascular surgery does not affect long-term mortality rates.33 Based on this study, the 2014 ACC/AHA guidelines2 advised against revascularization before noncardiac surgery exclusively to reduce perioperative cardiac events. Yet the same guidelines do recommend assessing for myocardial ischemia in patients with elevated risk and poor or unknown functional capacity, using a pharmacologic stress test. Based on the extent of the stress test abnormalities, coronary angiography and revascularization are then suggested for patients willing to undergo coronary artery bypass grafting (CABG) or percutaneous coronary intervention.2
The 2014 European Society of Cardiology and European Society of Anaesthesiology guidelines directly recommend revascularization before high-risk surgery, depending on the extent of a stress-induced perfusion defect.34 This recommendation relies on data from the Coronary Artery Surgery Study registry, which included almost 25,000 patients who underwent coronary angiography from 1975 through 1979. At a mean follow-up of 4.1 years, 1,961 patients underwent high-risk surgery. In this observational cohort, patients who underwent CABG had a lower risk of death and myocardial infarction after surgery.35 The reliance of medical societies34 on data that are more than 30 years old—when operative mortality rates and the treatment of coronary artery disease have changed substantially in the interim and despite the fact that this study did not test whether preoperative revascularization can reduce postoperative mortality—reflects a certain resistance to accept the results of the more recent and relevant randomized trial.33
Other physicians may also prefer to rely on selective data or to simply defer to guidelines that support their beliefs. Some physicians find that evidence-based guidelines are impractical and rigid and reduce their autonomy.36 For many physicians, trials that use surrogate end points and short-term outcomes are not sufficiently compelling to make them abandon current practice.37 Finally, when members of the guideline committees have financial associations with the pharmaceutical industry, or when corporations interested in the outcomes provide financial support for a trial’s development, the likelihood of a recommendation being trusted and used by physicians is drastically reduced.38
PRACTICING DEFENSIVELY
Even if physicians are familiar with the evidence and believe it, they may choose not to act on it. One reason is fear of litigation.
In court, attorneys can use guidelines as well as articles from medical journals as both exculpatory and inculpatory evidence. But they more frequently rely on the standard of care, or what most physicians would do under similar circumstances. If a patient has a bad outcome, such as a perioperative myocardial infarction or life-threatening bleeding, the defendant may assert that testing was unwarranted because guidelines do not recommend it or because the probability of such an outcome was low. However, because the outcome occurred, the jury may not believe that the probability was low enough not to consider, especially if expert witnesses testify that the standard of care would be to order the test.
In areas of controversy, physicians generally believe that erring on the side of more testing is more defensible in court.39 Indeed, following established practice traditions, learned during residency,11,40 may absolve physicians in negligence claims if the way medical care was delivered is supported by recognized and respected physicians.41
As a consequence, physicians prefer to practice the same way their peers do rather than follow the evidence. Unfortunately, the more procedures physicians perform for low-risk patients, the more likely these tests will become accepted as the legal standard of care.42 In this vicious circle, the new standard of care can increase the risk of litigation for others.43 Although unnecessary testing that leads to harmful invasive tests or procedures can also result in malpractice litigation, physicians may not consider this possibility.
FINANCIAL INCENTIVES
The threat of malpractice litigation provides a negative financial incentive to keep performing unnecessary tests, but there are a number of positive incentives as well.
First, physicians often feel compelled to order tests when they believe that physicians referring the patients want the tests done, or when they fear that not completing the tests could delay or cancel the scheduled surgery.40 Refusing to order the test could result in a loss of future referrals. In contrast, ordering tests allows them to meet expectations, preserve trust, and appear more valuable to referring physicians and their patients.
Insurance companies are complicit in these practices. Paying for unnecessary tests can create direct financial incentives for physicians or institutions that own on-site laboratories or diagnostic imaging equipment. Evidence shows that under those circumstances physicians do order more tests. Self-referral and referral to facilities where physicians have a financial interest is associated with increased healthcare costs.44 In addition to direct revenues for the tests performed, physicians may also bill for test interpretation, follow-up visits, and additional procedures generated from test results.
This may be one explanation why the ordering of cardiac tests (stress testing, echocardiography, vascular ultrasonography) by US physicians varies widely from state to state.45
RECOMMENDATIONS TO REDUCE INAPPROPRIATE TESTING
To counter these influences, we propose a multifaceted intervention that includes the following:
- Establish preoperative clinics staffed by experts. Despite the large volume of potentially relevant evidence, the number of articles directly supporting or refuting preoperative laboratory testing is small enough that physicians who routinely engage in preoperative assessment should easily master the evidence.
- Identify local leaders who can convince colleagues of the evidence. Distribute evidence summaries or guidelines with references to major articles that support each recommendation.
- Work with clinical practice committees to establish new standards of care within the hospital. Establish hospital care paths to dictate and support local standards of care. Measure individual physician performance and offer feedback with the goal of reducing utilization.
- National societies should recommend that insurance companies remove inappropriate financial incentives. If companies deny payment for inappropriate testing, physicians will stop ordering it. Even requirements for preauthorization of tests should reduce utilization. The Choosing Wisely campaign (www.choosingwisely.org) would be a good place to start.
Guidelines and practice advisories issued by several medical societies, including the American Society of Anesthesiologists,1 American Heart Association (AHA) and American College of Cardiology (ACC),2 and Society of General Internal Medicine,3 advise against routine preoperative testing for patients undergoing low-risk surgical procedures. Such testing often includes routine blood chemistry, complete blood cell counts, measures of the clotting system, and cardiac stress testing.
In this issue of the Cleveland Clinic Journal of Medicine, Dr. Nathan Houchens reviews the evidence against these measures.4
Despite a substantial body of evidence going back more than 2 decades that includes prospective randomized controlled trials,5–10 physicians continue to order unnecessary, ineffective, and costly tests in the perioperative period.11 The process of abandoning current medical practice—a phenomenon known as medical reversal12—often takes years,13 because it is more difficult to convince physicians to discontinue a current behavior than to implement a new one.14 The study of what makes physicians accept new therapies and abandon old ones began more than half a century ago.15
More recently, Cabana et al16 created a framework to understand why physicians do not follow clinical practice guidelines. Among the reasons are lack of familiarity or agreement with the contents of the guideline, lack of outcome expectancy, inertia of previous practice, and external barriers to implementation.
The rapid proliferation of guidelines in the past 20 years has led to numerous conflicting recommendations, many of which are based primarily on expert opinion.17 Guidelines based solely on randomized trials have also come under fire.18,19
In the case of preoperative testing, the recommendations are generally evidence-based and consistent. Why then do physicians appear to disregard the evidence? We propose several reasons why they might do so.
SOME PHYSICIANS ARE UNFAMILIAR WITH THE EVIDENCE
The complexity of the evidence summarized in guidelines has increased exponentially in the last decade, but physician time to assess the evidence has not increased. For example, the number of references in the executive summary of the ACC/AHA perioperative guidelines increased from 96 in 2002 to 252 in 2014. Most of the recommendations are backed by substantial amounts of high-quality evidence. For example, there are 17 prospective and 13 retrospective studies demonstrating that routine testing with the prothrombin time and the partial thromboplastin time is not helpful in asymptomatic patients.20
Although compliance with medical evidence varies among specialties,21 most physicians do not have time to keep up with the ever-increasing amount of information. Specifically in the area of cardiac risk assessment, there has been a rapid proliferation of tests that can be used to assess cardiac risk.22–28 In a Harris Interactive survey from 2008, physicians reported not applying medical evidence routinely. One-third believed they would do it more if they had the time.29 Without information technology support to provide medical information at the point of care,30 especially in small practices, using evidence may not be practical. Simply making the information available online and not promoting it actively does not improve utilization.31
As a consequence, physicians continue to order unnecessary tests, even though they may not feel confident interpreting the results.32
PHYSICIANS MAY NOT BELIEVE THE EVIDENCE
A lack of transparency in evidence-based guidelines and, sometimes, a lack of flexibility and relevance to clinical practice are important barriers to physicians’ acceptance of and adherence to evidence-based clinical practice guidelines.30
Even experts who write guidelines may not be swayed by the evidence. For example, a randomized prospective trial of almost 6,000 patients reported that coronary artery revascularization before elective major vascular surgery does not affect long-term mortality rates.33 Based on this study, the 2014 ACC/AHA guidelines2 advised against revascularization before noncardiac surgery exclusively to reduce perioperative cardiac events. Yet the same guidelines do recommend assessing for myocardial ischemia in patients with elevated risk and poor or unknown functional capacity, using a pharmacologic stress test. Based on the extent of the stress test abnormalities, coronary angiography and revascularization are then suggested for patients willing to undergo coronary artery bypass grafting (CABG) or percutaneous coronary intervention.2
The 2014 European Society of Cardiology and European Society of Anaesthesiology guidelines directly recommend revascularization before high-risk surgery, depending on the extent of a stress-induced perfusion defect.34 This recommendation relies on data from the Coronary Artery Surgery Study registry, which included almost 25,000 patients who underwent coronary angiography from 1975 through 1979. At a mean follow-up of 4.1 years, 1,961 patients underwent high-risk surgery. In this observational cohort, patients who underwent CABG had a lower risk of death and myocardial infarction after surgery.35 The reliance of medical societies34 on data that are more than 30 years old—when operative mortality rates and the treatment of coronary artery disease have changed substantially in the interim and despite the fact that this study did not test whether preoperative revascularization can reduce postoperative mortality—reflects a certain resistance to accept the results of the more recent and relevant randomized trial.33
Other physicians may also prefer to rely on selective data or to simply defer to guidelines that support their beliefs. Some physicians find that evidence-based guidelines are impractical and rigid and reduce their autonomy.36 For many physicians, trials that use surrogate end points and short-term outcomes are not sufficiently compelling to make them abandon current practice.37 Finally, when members of the guideline committees have financial associations with the pharmaceutical industry, or when corporations interested in the outcomes provide financial support for a trial’s development, the likelihood of a recommendation being trusted and used by physicians is drastically reduced.38
PRACTICING DEFENSIVELY
Even if physicians are familiar with the evidence and believe it, they may choose not to act on it. One reason is fear of litigation.
In court, attorneys can use guidelines as well as articles from medical journals as both exculpatory and inculpatory evidence. But they more frequently rely on the standard of care, or what most physicians would do under similar circumstances. If a patient has a bad outcome, such as a perioperative myocardial infarction or life-threatening bleeding, the defendant may assert that testing was unwarranted because guidelines do not recommend it or because the probability of such an outcome was low. However, because the outcome occurred, the jury may not believe that the probability was low enough not to consider, especially if expert witnesses testify that the standard of care would be to order the test.
In areas of controversy, physicians generally believe that erring on the side of more testing is more defensible in court.39 Indeed, following established practice traditions, learned during residency,11,40 may absolve physicians in negligence claims if the way medical care was delivered is supported by recognized and respected physicians.41
As a consequence, physicians prefer to practice the same way their peers do rather than follow the evidence. Unfortunately, the more procedures physicians perform for low-risk patients, the more likely these tests will become accepted as the legal standard of care.42 In this vicious circle, the new standard of care can increase the risk of litigation for others.43 Although unnecessary testing that leads to harmful invasive tests or procedures can also result in malpractice litigation, physicians may not consider this possibility.
FINANCIAL INCENTIVES
The threat of malpractice litigation provides a negative financial incentive to keep performing unnecessary tests, but there are a number of positive incentives as well.
First, physicians often feel compelled to order tests when they believe that physicians referring the patients want the tests done, or when they fear that not completing the tests could delay or cancel the scheduled surgery.40 Refusing to order the test could result in a loss of future referrals. In contrast, ordering tests allows them to meet expectations, preserve trust, and appear more valuable to referring physicians and their patients.
Insurance companies are complicit in these practices. Paying for unnecessary tests can create direct financial incentives for physicians or institutions that own on-site laboratories or diagnostic imaging equipment. Evidence shows that under those circumstances physicians do order more tests. Self-referral and referral to facilities where physicians have a financial interest is associated with increased healthcare costs.44 In addition to direct revenues for the tests performed, physicians may also bill for test interpretation, follow-up visits, and additional procedures generated from test results.
This may be one explanation why the ordering of cardiac tests (stress testing, echocardiography, vascular ultrasonography) by US physicians varies widely from state to state.45
RECOMMENDATIONS TO REDUCE INAPPROPRIATE TESTING
To counter these influences, we propose a multifaceted intervention that includes the following:
- Establish preoperative clinics staffed by experts. Despite the large volume of potentially relevant evidence, the number of articles directly supporting or refuting preoperative laboratory testing is small enough that physicians who routinely engage in preoperative assessment should easily master the evidence.
- Identify local leaders who can convince colleagues of the evidence. Distribute evidence summaries or guidelines with references to major articles that support each recommendation.
- Work with clinical practice committees to establish new standards of care within the hospital. Establish hospital care paths to dictate and support local standards of care. Measure individual physician performance and offer feedback with the goal of reducing utilization.
- National societies should recommend that insurance companies remove inappropriate financial incentives. If companies deny payment for inappropriate testing, physicians will stop ordering it. Even requirements for preauthorization of tests should reduce utilization. The Choosing Wisely campaign (www.choosingwisely.org) would be a good place to start.
- Committee on Standards and Practice Parameters, Apfelbaum JL, Connis RT, Nickinovich DG, et al. Practice advisory for preanesthesia evaluation. An updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology 2012; 116:522–538.
- Fleisher LA, Fleischmann KE, Auerbach AD, et al; American College of Cardiology and American Heart Association. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol 2014; 64:e77–e137.
- Society of General Internal Medicine. Don’t perform routine pre-operative testing before low-risk surgical procedures. Choosing Wisely. An initiative of the ABIM Foundation. September 12, 2013. www.choosingwisely.org/clinician-lists/society-general-internal-medicine-routine-preoperative-testing-before-low-risk-surgery/. Accessed August 31, 2015.
- Houchens N. Should healthy patients undergoing low-risk, elective, noncardiac surgery undergo routine preoperative laboratory testing? Cleve Clin J Med 2015; 82:664–666.
- Rohrer MJ, Michelotti MC, Nahrwold DL. A prospective evaluation of the efficacy of preoperative coagulation testing. Ann Surg 1988; 208:554–557.
- Eagle KA, Coley CM, Newell JB, et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med 1989; 110:859–866.
- Mangano DT, London MJ, Tubau JF, et al. Dipyridamole thallium-201 scintigraphy as a preoperative screening test. A reexamination of its predictive potential. Study of Perioperative Ischemia Research Group. Circulation 1991; 84:493–502.
- Stratmann HG, Younis LT, Wittry MD, Amato M, Mark AL, Miller DD. Dipyridamole technetium 99m sestamibi myocardial tomography for preoperative cardiac risk stratification before major or minor nonvascular surgery. Am Heart J 1996; 132:536–541.
- Schein OD, Katz J, Bass EB, et al. The value of routine preoperative medical testing before cataract surgery. Study of Medical Testing for Cataract Surgery. N Engl J Med 2000; 342:168–175.
- Hashimoto J, Nakahara T, Bai J, Kitamura N, Kasamatsu T, Kubo A. Preoperative risk stratification with myocardial perfusion imaging in intermediate and low-risk non-cardiac surgery. Circ J 2007; 71:1395–1400.
- Smetana GW. The conundrum of unnecessary preoperative testing. JAMA Intern Med 2015; 175:1359–1361.
- Prasad V, Cifu A. Medical reversal: why we must raise the bar before adopting new technologies. Yale J Biol Med 2011; 84:471–478.
- Tatsioni A, Bonitsis NG, Ioannidis JP. Persistence of contradicted claims in the literature. JAMA 2007; 298:2517–2526.
- Moscucci M. Medical reversal, clinical trials, and the “late” open artery hypothesis in acute myocardial infarction. Arch Intern Med 2011; 171:1643–1644.
- Coleman J, Menzel H, Katz E. Social processes in physicians’ adoption of a new drug. J Chronic Dis 1959; 9:1–19.
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA 1999; 282:1458–1465.
- Tricoci P, Allen JM, Kramer JM, Califf RM, Smith SC Jr. Scientific evidence underlying the ACC/AHA clinical practice guidelines. JAMA 2009; 301:831–841.
- Moher D, Hopewell S, Schulz KF, et al; CONSORT. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. Int J Surg 2012; 10:28–55.
- Gattinoni L, Giomarelli P. Acquiring knowledge in intensive care: merits and pitfalls of randomized controlled trials. Intensive Care Med 2015; 41:1460–1464.
- Levy JH, Szlam F, Wolberg AS, Winkler A. Clinical use of the activated partial thromboplastin time and prothrombin time for screening: a review of the literature and current guidelines for testing. Clin Lab Med 2014; 34:453–477.
- Dale W, Hemmerich J, Moliski E, Schwarze ML, Tung A. Effect of specialty and recent experience on perioperative decision-making for abdominal aortic aneurysm repair. J Am Geriatr Soc 2012; 60:1889–1894.
- Underwood SR, Anagnostopoulos C, Cerqueira M, et al; British Cardiac Society, British Nuclear Cardiology Society, British Nuclear Medicine Society, Royal College of Physicians of London, Royal College of Physicians of London. Myocardial perfusion scintigraphy: the evidence. Eur J Nucl Med Mol Imaging 2004; 31:261–291.
- Das MK, Pellikka PA, Mahoney DW, et al. Assessment of cardiac risk before nonvascular surgery: dobutamine stress echocardiography in 530 patients. J Am Coll Cardiol 2000; 35:1647–1653.
- Meijboom WB, Mollet NR, Van Mieghem CA, et al. Pre-operative computed tomography coronary angiography to detect significant coronary artery disease in patients referred for cardiac valve surgery. J Am Coll Cardiol 2006; 48:1658–1665.
- Russo V, Gostoli V, Lovato L, et al. Clinical value of multidetector CT coronary angiography as a preoperative screening test before non-coronary cardiac surgery. Heart 2007; 93:1591–1598.
- Schuetz GM, Zacharopoulou NM, Schlattmann P, Dewey M. Meta-analysis: noninvasive coronary angiography using computed tomography versus magnetic resonance imaging. Ann Intern Med 2010; 152:167–177.
- Bluemke DA, Achenbach S, Budoff M, et al. Noninvasive coronary artery imaging: magnetic resonance angiography and multidetector computed tomography angiography: a scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention of the Council on Cardiovascular Radiology and Intervention, and the Councils on Clinical Cardiology and Cardiovascular Disease in the Young. Circulation 2008; 118:586–606.
- Nagel E, Lehmkuhl HB, Bocksch W, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation 1999; 99:763–770.
- Taylor H. Physicians’ use of clinical guidelines—and how to increase it. Healthcare News 2008; 8:32–55. www.harrisinteractive.com/vault/HI_HealthCareNews2008Vol8_Iss04.pdf. Accessed August 31, 2015.
- Kenefick H, Lee J, Fleishman V. Improving physician adherence to clinical practice guidelines. Barriers and stragies for change. New England Healthcare Institute, February 2008. www.nehi.net/writable/publication_files/file/cpg_report_final.pdf. Accessed August 31, 2015.
- Williams J, Cheung WY, Price DE, et al. Clinical guidelines online: do they improve compliance? Postgrad Med J 2004; 80:415–419.
- Wians F. Clinical laboratory tests: which, why, and what do the results mean? Lab Medicine 2009; 40:105–113.
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004; 351:2795–2804.
- Kristensen SD, Knuuti J, Saraste A, et al; Authors/Task Force Members. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Eur Heart J 2014; 35:2383–2431.
- Eagle KA, Rihal CS, Mickel MC, Holmes DR, Foster ED, Gersh BJ. Cardiac risk of noncardiac surgery: influence of coronary disease and type of surgery in 3368 operations. CASS Investigators and University of Michigan Heart Care Program. Coronary Artery Surgery Study. Circulation 1997; 96:1882–1887.
- Farquhar CM, Kofa EW, Slutsky JR. Clinicians’ attitudes to clinical practice guidelines: a systematic review. Med J Aust 2002; 177:502–506.
- Prasad V, Cifu A, Ioannidis JP. Reversals of established medical practices: evidence to abandon ship. JAMA 2012; 307:37–38.
- Steinbrook R. Guidance for guidelines. N Engl J Med 2007; 356:331–333.
- Sirovich BE, Woloshin S, Schwartz LM. Too little? Too much? Primary care physicians’ views on US health care: a brief report. Arch Intern Med 2011; 171:1582–1585.
- Brown SR, Brown J. Why do physicians order unnecessary preoperative tests? A qualitative study. Fam Med 2011; 43:338–343.
- LeCraw LL. Use of clinical practice guidelines in medical malpractice litigation. J Oncol Pract 2007; 3:254.
- Studdert DM, Mello MM, Sage WM, et al. Defensive medicine among high-risk specialist physicians in a volatile malpractice environment. JAMA 2005; 293:2609–2617.
- Budetti PP. Tort reform and the patient safety movement: seeking common ground. JAMA 2005; 293:2660–2662.
- Bishop TF, Federman AD, Ross JS. Laboratory test ordering at physician offices with and without on-site laboratories. J Gen Intern Med 2010; 25:1057–1063.
- Rosenthal E. Medical costs rise as retirees winter in Florida. The New York Times, Jan 31, 2015. http://nyti.ms/1vmjfa5. Accessed August 31, 2015.
- Committee on Standards and Practice Parameters, Apfelbaum JL, Connis RT, Nickinovich DG, et al. Practice advisory for preanesthesia evaluation. An updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology 2012; 116:522–538.
- Fleisher LA, Fleischmann KE, Auerbach AD, et al; American College of Cardiology and American Heart Association. 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol 2014; 64:e77–e137.
- Society of General Internal Medicine. Don’t perform routine pre-operative testing before low-risk surgical procedures. Choosing Wisely. An initiative of the ABIM Foundation. September 12, 2013. www.choosingwisely.org/clinician-lists/society-general-internal-medicine-routine-preoperative-testing-before-low-risk-surgery/. Accessed August 31, 2015.
- Houchens N. Should healthy patients undergoing low-risk, elective, noncardiac surgery undergo routine preoperative laboratory testing? Cleve Clin J Med 2015; 82:664–666.
- Rohrer MJ, Michelotti MC, Nahrwold DL. A prospective evaluation of the efficacy of preoperative coagulation testing. Ann Surg 1988; 208:554–557.
- Eagle KA, Coley CM, Newell JB, et al. Combining clinical and thallium data optimizes preoperative assessment of cardiac risk before major vascular surgery. Ann Intern Med 1989; 110:859–866.
- Mangano DT, London MJ, Tubau JF, et al. Dipyridamole thallium-201 scintigraphy as a preoperative screening test. A reexamination of its predictive potential. Study of Perioperative Ischemia Research Group. Circulation 1991; 84:493–502.
- Stratmann HG, Younis LT, Wittry MD, Amato M, Mark AL, Miller DD. Dipyridamole technetium 99m sestamibi myocardial tomography for preoperative cardiac risk stratification before major or minor nonvascular surgery. Am Heart J 1996; 132:536–541.
- Schein OD, Katz J, Bass EB, et al. The value of routine preoperative medical testing before cataract surgery. Study of Medical Testing for Cataract Surgery. N Engl J Med 2000; 342:168–175.
- Hashimoto J, Nakahara T, Bai J, Kitamura N, Kasamatsu T, Kubo A. Preoperative risk stratification with myocardial perfusion imaging in intermediate and low-risk non-cardiac surgery. Circ J 2007; 71:1395–1400.
- Smetana GW. The conundrum of unnecessary preoperative testing. JAMA Intern Med 2015; 175:1359–1361.
- Prasad V, Cifu A. Medical reversal: why we must raise the bar before adopting new technologies. Yale J Biol Med 2011; 84:471–478.
- Tatsioni A, Bonitsis NG, Ioannidis JP. Persistence of contradicted claims in the literature. JAMA 2007; 298:2517–2526.
- Moscucci M. Medical reversal, clinical trials, and the “late” open artery hypothesis in acute myocardial infarction. Arch Intern Med 2011; 171:1643–1644.
- Coleman J, Menzel H, Katz E. Social processes in physicians’ adoption of a new drug. J Chronic Dis 1959; 9:1–19.
- Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA 1999; 282:1458–1465.
- Tricoci P, Allen JM, Kramer JM, Califf RM, Smith SC Jr. Scientific evidence underlying the ACC/AHA clinical practice guidelines. JAMA 2009; 301:831–841.
- Moher D, Hopewell S, Schulz KF, et al; CONSORT. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. Int J Surg 2012; 10:28–55.
- Gattinoni L, Giomarelli P. Acquiring knowledge in intensive care: merits and pitfalls of randomized controlled trials. Intensive Care Med 2015; 41:1460–1464.
- Levy JH, Szlam F, Wolberg AS, Winkler A. Clinical use of the activated partial thromboplastin time and prothrombin time for screening: a review of the literature and current guidelines for testing. Clin Lab Med 2014; 34:453–477.
- Dale W, Hemmerich J, Moliski E, Schwarze ML, Tung A. Effect of specialty and recent experience on perioperative decision-making for abdominal aortic aneurysm repair. J Am Geriatr Soc 2012; 60:1889–1894.
- Underwood SR, Anagnostopoulos C, Cerqueira M, et al; British Cardiac Society, British Nuclear Cardiology Society, British Nuclear Medicine Society, Royal College of Physicians of London, Royal College of Physicians of London. Myocardial perfusion scintigraphy: the evidence. Eur J Nucl Med Mol Imaging 2004; 31:261–291.
- Das MK, Pellikka PA, Mahoney DW, et al. Assessment of cardiac risk before nonvascular surgery: dobutamine stress echocardiography in 530 patients. J Am Coll Cardiol 2000; 35:1647–1653.
- Meijboom WB, Mollet NR, Van Mieghem CA, et al. Pre-operative computed tomography coronary angiography to detect significant coronary artery disease in patients referred for cardiac valve surgery. J Am Coll Cardiol 2006; 48:1658–1665.
- Russo V, Gostoli V, Lovato L, et al. Clinical value of multidetector CT coronary angiography as a preoperative screening test before non-coronary cardiac surgery. Heart 2007; 93:1591–1598.
- Schuetz GM, Zacharopoulou NM, Schlattmann P, Dewey M. Meta-analysis: noninvasive coronary angiography using computed tomography versus magnetic resonance imaging. Ann Intern Med 2010; 152:167–177.
- Bluemke DA, Achenbach S, Budoff M, et al. Noninvasive coronary artery imaging: magnetic resonance angiography and multidetector computed tomography angiography: a scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention of the Council on Cardiovascular Radiology and Intervention, and the Councils on Clinical Cardiology and Cardiovascular Disease in the Young. Circulation 2008; 118:586–606.
- Nagel E, Lehmkuhl HB, Bocksch W, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation 1999; 99:763–770.
- Taylor H. Physicians’ use of clinical guidelines—and how to increase it. Healthcare News 2008; 8:32–55. www.harrisinteractive.com/vault/HI_HealthCareNews2008Vol8_Iss04.pdf. Accessed August 31, 2015.
- Kenefick H, Lee J, Fleishman V. Improving physician adherence to clinical practice guidelines. Barriers and stragies for change. New England Healthcare Institute, February 2008. www.nehi.net/writable/publication_files/file/cpg_report_final.pdf. Accessed August 31, 2015.
- Williams J, Cheung WY, Price DE, et al. Clinical guidelines online: do they improve compliance? Postgrad Med J 2004; 80:415–419.
- Wians F. Clinical laboratory tests: which, why, and what do the results mean? Lab Medicine 2009; 40:105–113.
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004; 351:2795–2804.
- Kristensen SD, Knuuti J, Saraste A, et al; Authors/Task Force Members. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Eur Heart J 2014; 35:2383–2431.
- Eagle KA, Rihal CS, Mickel MC, Holmes DR, Foster ED, Gersh BJ. Cardiac risk of noncardiac surgery: influence of coronary disease and type of surgery in 3368 operations. CASS Investigators and University of Michigan Heart Care Program. Coronary Artery Surgery Study. Circulation 1997; 96:1882–1887.
- Farquhar CM, Kofa EW, Slutsky JR. Clinicians’ attitudes to clinical practice guidelines: a systematic review. Med J Aust 2002; 177:502–506.
- Prasad V, Cifu A, Ioannidis JP. Reversals of established medical practices: evidence to abandon ship. JAMA 2012; 307:37–38.
- Steinbrook R. Guidance for guidelines. N Engl J Med 2007; 356:331–333.
- Sirovich BE, Woloshin S, Schwartz LM. Too little? Too much? Primary care physicians’ views on US health care: a brief report. Arch Intern Med 2011; 171:1582–1585.
- Brown SR, Brown J. Why do physicians order unnecessary preoperative tests? A qualitative study. Fam Med 2011; 43:338–343.
- LeCraw LL. Use of clinical practice guidelines in medical malpractice litigation. J Oncol Pract 2007; 3:254.
- Studdert DM, Mello MM, Sage WM, et al. Defensive medicine among high-risk specialist physicians in a volatile malpractice environment. JAMA 2005; 293:2609–2617.
- Budetti PP. Tort reform and the patient safety movement: seeking common ground. JAMA 2005; 293:2660–2662.
- Bishop TF, Federman AD, Ross JS. Laboratory test ordering at physician offices with and without on-site laboratories. J Gen Intern Med 2010; 25:1057–1063.
- Rosenthal E. Medical costs rise as retirees winter in Florida. The New York Times, Jan 31, 2015. http://nyti.ms/1vmjfa5. Accessed August 31, 2015.
A 56-year-old with diarrhea and weakness
A 56-year-old man presents to the emergency department with nausea, weakness, and exertional dyspnea, which have been going on for 1 week. He is sent by his primary care physician after being noted to be hypotensive with a weak, thready pulse.
He has had diarrhea with intermittent abdominal pain over the past year, with 10 stools daily, including 3 or 4 at night. The stools are described as large, nonbloody, sticky, greasy, and occasionally watery. Stools are fewer when he curtails his food intake. The diarrhea is associated with occasional diffuse abdominal pain he describes as a burning sensation. He has no incontinence or tenesmus. He reports that he has unintentionally lost 137 lb (62 kg) over the past year. He has not taken over-the-counter antidiarrheal agents.
CHRONIC DIARRHEA
1. Chronic diarrhea is defined as lasting for at least how long?
- 1 week
- 2 weeks
- 3 weeks
- 4 weeks
Chronic diarrhea is defined as looser stools for more than 4 weeks,1 a period that allows most cases of acute, self-limited, infectious diarrhea to resolve.
Because individuals perceive diarrhea differently, reported prevalence rates of chronic diarrhea vary.2 Based on the definition of having excessive stool frequency, the prevalence in the United States is about 5%.1
In developing countries, the most common cause of chronic diarrhea is infection. In developed nations, irritable bowel syndrome, inflammatory bowel disease, malabsorption syndrome, and chronic infection predominate.1
Once chronicity is established, diarrhea should be characterized as inflammatory, fatty, or watery (Table 1).3
CASE CONTINUED: HISTORY OF HYPERTENSION, DIABETES
Our patient reports that he has never traveled outside the United States. He has a history of hypertension and type 2 diabetes mellitus that is controlled on oral agents. He has had surgery for a radial fracture and for reconstruction of his knees. He uses no tobacco, alcohol, or illicit drugs and works as a train engineer. He has no pets. He knows of no family history of inflammatory bowel disease or chronic diarrhea.
Comment. Patients with diabetes are at increased risk of gastrointestinal problems, with severity increasing with poorer control.4 Although our patient’s diabetes puts him at risk of diabetic autonomic neuropathy, his blood glucose control has been consistently good since his diagnosis, and his last measured hemoglobin A1c was 7.3% (reference range 4%–7%). His description of greasy stools in conjunction with his marked weight loss puts fatty diarrhea higher on the differential diagnosis.
DRUG-INDUCED DIARRHEA
His medications include glimepiride 1 mg twice daily, lisinopril 10 mg daily, metformin 500 mg twice daily, omeprazole 40 mg daily, and naproxen 220 mg daily. He has been taking metformin for at least 2 years. He is allergic to pentobarbital.
2. Which of his medications is least likely to be associated with his diarrhea?
- Lisinopril
- Metformin
- Glimepiride
- Naproxen
More than 700 drugs are known to cause diarrhea, often through the interplay of simultaneous mechanisms.5 The diagnosis of drug-induced diarrhea requires taking a careful medication history and establishing a temporal relationship between the drug and the diarrheal symptoms. Treatment consists of withdrawing the offending agent.
Nonsteroidal anti-inflammatory drugs (eg, naproxen) are associated with collagenous colitis that occurs mostly after long-term use (> 6 months). Metformin-induced diarrhea is related to fat malabsorption. Olmesartan, an angiotensin II receptor antagonist, has been associated with severe sprue-like enteropathy. On the other hand, the incidence of diarrhea with lisinopril is similar to that with placebo.7
CASE CONTINUED: EXAMINATION AND LABORATORY VALUES
The patient’s primary care physician had recently referred him to a gastroenterologist, and 4 days before presenting to the emergency department he had undergone abdominal and pelvic computed tomography (CT) with iodinated contrast, which had showed hepatic steatosis and pancreatic atrophy.
On examination now, the patient’s temperature is 97.5°F (36.4°C), heart rate 90 beats per minute, respirations 18 breaths per minute, oxygen saturation 99% on room air, and blood pressure 85/55 mm Hg. His body mass index is 32.5 kg/m2. His oral mucosa is dry. The rest of the examination is normal. No rash or ulcers are noted.
His laboratory values (Table 2) are notable for sodium 130 mmol/L, potassium 2.2 mmol/L, bicarbonate 9 mmol/L, blood urea nitrogen 32 mg/dL, creatinine 4.18 mg/dL, and international normalized ratio 5.4. Arterial blood gases drawn on admission reveal pH 7.32 and pCO2 19 mm Hg.
ACID-BASE DISTURBANCES
3. The patient’s acidosis is most likely related to which of the following?
- Sepsis
- Diarrhea
- Metformin
- Acute kidney injury
It is most likely related to diarrhea. The patient has a non-anion-gap metabolic acidosis. (The anion gap can be calculated by subtracting the sum of the serum bicarbonate and chloride values from the sodium—here, 130 – [112 + 9] = 9—and most textbooks list the reference range as 10–12 mmol/L.) Non-anion-gap metabolic acidosis results from excessive loss of bicarbonate or impaired ability of the kidney to excrete acid. Bicarbonate losses can occur in diarrhea or in ureteral diversion to the colon. Impairment in urinary acidification can occur in renal tubular acidosis.
To determine the cause of non-anion-gap acidosis, calculating the urine anion gap can be useful (Table 3), as it reflects the ability of the kidneys to excrete acid and is an indirect measure of ammonium excretion. Our patient’s urine anion gap is –45 mmol/L ([62 + 8] – 115), which supports diarrhea as the cause of his non-anion-gap acidosis. Sepsis, metformin use, or acute kidney injury would result in an anion-gap acidosis.
To manage acid-base disturbances, it is important to first determine whether there is a single primary disturbance with compensation or a mixed disorder. In the case of metabolic acidosis, for every 1-mmol/L decrease in bicarbonate, there should be a corresponding 1.3-mm Hg decrease in pCO2. Our patient’s laboratory data show that he had a pure non-anion-gap metabolic acidosis.8 His sensation of dyspnea was likely related to respiratory compensation as evidenced by an appropriately low pCO2.
CASE CONTINUED: HIS LABORATORY VALUES IMPROVE
The patient is admitted to the hospital for fluid resuscitation with normal saline and potassium and magnesium replacement.
Renal ultrasonography reveals normal-appearing kidneys without obstruction. The calculated fractional excretion of sodium is 3.4%. Urine microscopy reveals two to five hyaline casts per low-power field. His urine output remains adequate, and 3 days after hospitalization, his renal function starts to improve, as reflected in falling serum creatinine and blood urea nitrogen levels: his creatinine level has declined to 1.91 mg/dL and his blood urea nitrogen level has declined to 24 mg/dL. His acute kidney injury is attributed to intravenous contrast given for computed tomography, as well as to volume depletion and hypotension.
Stool studies for ova, parasites, and Clostridium difficile are negative. Fecal calprotectin and lactoferrin are useful noninvasive markers of intestinal inflammation but were not checked in this case.
Loperamide, taken as needed, is started for his diarrhea, along with empiric pancreatic enzyme replacement. After 3 days of treatment with oral vitamin K 10 mg, his international normalized ratio comes down to 1.4, from his admission value of 5.4. Given the clinical concern for fat malabsorption, vitamin D levels are also checked: his 25-hydroxyvitamin D level is less than 10 ng/mL (lower limit of normal 20). His fecal neutral fats are reported as normal, but split fats are increased.
STOOL FAT STUDIES
4. What does increased fecal split fats but normal fecal neutral fats imply?
- Pancreatic insufficiency
- Intestinal malabsorption
- Does not differentiate between the two
The finding does not differentiate between pancreatic insufficiency and intestinal malabsorption. The two-step Sudan stain has been used to differentiate maldigestion (eg, caused by pancreatic insufficiency) from malabsorption. Although patients with impaired digestion were once thought to excrete excessive amounts of intact triglyceride whereas those with malabsorption excrete more of the lipolytic or “split” product, the Sudan stain does not differentiate between the two.10 Stool fecal-elastase 1 testing correlates well with pancreatic exocrine function but was not performed in our patient.11
CASE CONTINUED: CELIAC DISEASE IS DIAGNOSED
Given the description of his stools, unintentional weight loss, and improvement of stool frequency with fasting, serologic testing for celiac disease is performed (Table 4). The patient undergoes esophagogastroduodenoscopy, which shows mild duodenitis. Small-bowel biopsy reveals blunted villous architecture and increased mixed inflammatory cells of the epithelium and lamina propria, suggestive of celiac disease.
The patient is diagnosed with celiac disease and is counseled to follow a gluten-free diet. He is discharged home and scheduled to follow up with a gastroenterologist and nephrologist. His liver function test abnormalities are attributed to a combination of nonalcoholic steatohepatitis and celiac disease.
CELIAC DISEASE AND MALABSORPTION
Celiac disease is an immune-mediated disorder that causes mucosal injury to the small intestine, leading to malabsorption. It is triggered by gluten intake in genetically susceptible individuals. The HLA-DQ2 haplotype is expressed in nearly 90% of patients with the disease.
The worldwide prevalence of celiac disease is about 0.6% to 1%. Those with an affected first-degree relative, type 1 diabetes, Hashimoto thyroiditis, an autoimmune disease, Down syndrome, Turner syndrome, or IgA deficiency have an increased risk.
Celiac disease presents with chronic diarrhea, weight loss, and abdominal distention and pain. Sequelae of nutrient malabsorption such as iron-deficiency anemia, short stature, and osteopenia may be evident. Liver function may also be impaired. Dermatitis herpetiformis and gluten ataxia are rarer presentations of celiac disease.12
In the absence of immunoglobulin (Ig) A deficiency, measurement of serum IgA anti-tissue transglutaminase antibodies is recommended for initial testing. IgG antitissue transglutaminase antibodies can be measured in those with IgA deficiency.12
Duodenal biopsies to confirm the diagnosis are recommended in adults unless they have previously had biopsy-proven dermatitis herpetiformis.
Gluten-free diet
The treatment for celiac disease is avoidance of gluten. Patients who consult with a nutritionist and participate in an advocacy group are more likely to adhere to a gluten-free diet, and the physician should strongly encourage and facilitate these activities.13
Untreated disease can lead to osteoporosis, impaired splenic function with increased risk of infection with encapsulated organisms, infertility or recurrent abortion, ulcerative jejunoileitis, and lymphoma.12 Patients should be monitored annually for adherence to the gluten-free diet and for the development of any associated condition. Despite adherence to a gluten-free diet, calcium absorption and bone mineral density are lower in patients with celiac disease than in controls.14 Careful monitoring of fracture risk and adequate calcium and vitamin D replacement are also important.
Our patient undergoes dual-emission x-ray absorptiometry after discharge, with results consistent with osteopenia. His T scores range from –0.2 at the right hip to –1.1 in the left femoral neck.
Recurrence or persistently abnormal levels of IgA anti-tissue transglutaminase antibodies usually indicates poor dietary compliance.12
5. In patients whose symptoms do not improve on gluten restriction, there should be concern for which of the following?
- Lymphoma
- Nonadherence to gluten restriction
- Microscopic colitis
- All of the above
The answer is all of the above. Up to 30% of patients have persistent symptoms on a gluten-free diet. Persistent exposure to gluten is the most common reason for lack of clinical improvement. In addition, bacterial overgrowth of the small bowel, lactose intolerance, pancreatic insufficiency, and microscopic colitis may coexist with celiac disease and may contribute to ongoing symptoms. In a small subset of patients with persistent villous atrophy and symptoms despite strict adherence to a gluten-free diet for 12 months, the disease is deemed “refractory.” Refractory celiac disease can be a precursor to enteropathy-associated T-cell lymphoma.13
CASE CONCLUDED
On telephone follow-up 3 weeks after discharge, the patient reports complete resolution of diarrhea and stabilization of his weight. He reports strict adherence to a gluten-free diet and feels he is coping well.
Diagnoses
- Presenting weakness secondary to dehydration and hypokalemia
- Dyspnea secondary to respiratory compensation for metabolic acidosis
- Non-anion-gap metabolic acidosis secondary to diarrhea
- Acute kidney injury secondary to iodinated contrast, volume depletion, hypotension
- Chronic diarrhea secondary to celiac disease
- Coagulopathy secondary to fat malabsorption secondary to celiac disease.
- Fine KD, Schiller LR. AGA technical review on the evaluation and management of chronic diarrhea. Gastroenterology 1999; 116:1464–1486.
- Talley NJ, Weaver AL, Zinsmeister AR, Melton LJ 3rd. Self-reported diarrhea: what does it mean? Am J Gastroenterol 1994; 89:1160–1164.
- Sweetser S. Evaluating the patient with diarrhea: a case-based approach. Mayo Clin Proc 2012; 87:596–602.
- Bytzer P, Talley NJ, Leemon M, Young LJ, Jones MP, Horowitz M. Prevalence of gastrointestinal symptoms associated with diabetes mellitus: a population-based survey of 15,000 adults. Arch Intern Med 2001; 161:1989–1996.
- Chassany O, Michaux A, Bergmann JF. Drug-induced diarrhoea. Drug Saf 2000; 22:53–72.
- Rubio-Tapia A, Herman ML, Ludvigsson JF, et al. Severe spruelike enteropathy associated with olmesartan. Mayo Clin Proc 2012; 87:732–738.
- Zestril (lisinopril) tablets. www.accessdata.fda.gov/drugsatfda_docs/label/2012/019777s062lbl.pdf. Accessed September 8, 2015.
- Whittier WL, Rutecki GW. Primer on clinical acid-base problem solving. Dis Mon 2004; 50:122–162.
- Langhorst J, Elsenbruch S, Koelzer J, Rueffer A, Michalsen A, Dobos GJ. Noninvasive markers in the assessment of intestinal inflammation in inflammatory bowel diseases: performance of fecal lactoferrin, calprotectin, and PMN-elastase, CRP, and clinical indices. Am J Gastroenterol 2008; 103:162–169.
- Khouri MR, Ng SN, Huang G, Shiau YF. Fecal triglyceride excretion is not excessive in pancreatic insufficiency. Gastroenterology 1989; 96:848–852.
- Dominici R, Franzini C. Fecal elastase-1 as a test for pancreatic function: a review. Clin Chem Lab Med 2002; 40:325–332.
- Fasano A, Catassi C. Celiac disease. New Engl J Med 2012; 367:2419–2426.
- Mooney PD, Hadjivassiliou M, Sanders DS. Coeliac disease. BMJ 2014; 348:g1561–g1561.
- Pazianas M, Butcher GP, Subhani JM, et al. Calcium absorption and bone mineral density in celiacs after long term treatment with gluten-free diet and adequate calcium intake. Osteoporos Int 2005; 16:56–63.
A 56-year-old man presents to the emergency department with nausea, weakness, and exertional dyspnea, which have been going on for 1 week. He is sent by his primary care physician after being noted to be hypotensive with a weak, thready pulse.
He has had diarrhea with intermittent abdominal pain over the past year, with 10 stools daily, including 3 or 4 at night. The stools are described as large, nonbloody, sticky, greasy, and occasionally watery. Stools are fewer when he curtails his food intake. The diarrhea is associated with occasional diffuse abdominal pain he describes as a burning sensation. He has no incontinence or tenesmus. He reports that he has unintentionally lost 137 lb (62 kg) over the past year. He has not taken over-the-counter antidiarrheal agents.
CHRONIC DIARRHEA
1. Chronic diarrhea is defined as lasting for at least how long?
- 1 week
- 2 weeks
- 3 weeks
- 4 weeks
Chronic diarrhea is defined as looser stools for more than 4 weeks,1 a period that allows most cases of acute, self-limited, infectious diarrhea to resolve.
Because individuals perceive diarrhea differently, reported prevalence rates of chronic diarrhea vary.2 Based on the definition of having excessive stool frequency, the prevalence in the United States is about 5%.1
In developing countries, the most common cause of chronic diarrhea is infection. In developed nations, irritable bowel syndrome, inflammatory bowel disease, malabsorption syndrome, and chronic infection predominate.1
Once chronicity is established, diarrhea should be characterized as inflammatory, fatty, or watery (Table 1).3
CASE CONTINUED: HISTORY OF HYPERTENSION, DIABETES
Our patient reports that he has never traveled outside the United States. He has a history of hypertension and type 2 diabetes mellitus that is controlled on oral agents. He has had surgery for a radial fracture and for reconstruction of his knees. He uses no tobacco, alcohol, or illicit drugs and works as a train engineer. He has no pets. He knows of no family history of inflammatory bowel disease or chronic diarrhea.
Comment. Patients with diabetes are at increased risk of gastrointestinal problems, with severity increasing with poorer control.4 Although our patient’s diabetes puts him at risk of diabetic autonomic neuropathy, his blood glucose control has been consistently good since his diagnosis, and his last measured hemoglobin A1c was 7.3% (reference range 4%–7%). His description of greasy stools in conjunction with his marked weight loss puts fatty diarrhea higher on the differential diagnosis.
DRUG-INDUCED DIARRHEA
His medications include glimepiride 1 mg twice daily, lisinopril 10 mg daily, metformin 500 mg twice daily, omeprazole 40 mg daily, and naproxen 220 mg daily. He has been taking metformin for at least 2 years. He is allergic to pentobarbital.
2. Which of his medications is least likely to be associated with his diarrhea?
- Lisinopril
- Metformin
- Glimepiride
- Naproxen
More than 700 drugs are known to cause diarrhea, often through the interplay of simultaneous mechanisms.5 The diagnosis of drug-induced diarrhea requires taking a careful medication history and establishing a temporal relationship between the drug and the diarrheal symptoms. Treatment consists of withdrawing the offending agent.
Nonsteroidal anti-inflammatory drugs (eg, naproxen) are associated with collagenous colitis that occurs mostly after long-term use (> 6 months). Metformin-induced diarrhea is related to fat malabsorption. Olmesartan, an angiotensin II receptor antagonist, has been associated with severe sprue-like enteropathy. On the other hand, the incidence of diarrhea with lisinopril is similar to that with placebo.7
CASE CONTINUED: EXAMINATION AND LABORATORY VALUES
The patient’s primary care physician had recently referred him to a gastroenterologist, and 4 days before presenting to the emergency department he had undergone abdominal and pelvic computed tomography (CT) with iodinated contrast, which had showed hepatic steatosis and pancreatic atrophy.
On examination now, the patient’s temperature is 97.5°F (36.4°C), heart rate 90 beats per minute, respirations 18 breaths per minute, oxygen saturation 99% on room air, and blood pressure 85/55 mm Hg. His body mass index is 32.5 kg/m2. His oral mucosa is dry. The rest of the examination is normal. No rash or ulcers are noted.
His laboratory values (Table 2) are notable for sodium 130 mmol/L, potassium 2.2 mmol/L, bicarbonate 9 mmol/L, blood urea nitrogen 32 mg/dL, creatinine 4.18 mg/dL, and international normalized ratio 5.4. Arterial blood gases drawn on admission reveal pH 7.32 and pCO2 19 mm Hg.
ACID-BASE DISTURBANCES
3. The patient’s acidosis is most likely related to which of the following?
- Sepsis
- Diarrhea
- Metformin
- Acute kidney injury
It is most likely related to diarrhea. The patient has a non-anion-gap metabolic acidosis. (The anion gap can be calculated by subtracting the sum of the serum bicarbonate and chloride values from the sodium—here, 130 – [112 + 9] = 9—and most textbooks list the reference range as 10–12 mmol/L.) Non-anion-gap metabolic acidosis results from excessive loss of bicarbonate or impaired ability of the kidney to excrete acid. Bicarbonate losses can occur in diarrhea or in ureteral diversion to the colon. Impairment in urinary acidification can occur in renal tubular acidosis.
To determine the cause of non-anion-gap acidosis, calculating the urine anion gap can be useful (Table 3), as it reflects the ability of the kidneys to excrete acid and is an indirect measure of ammonium excretion. Our patient’s urine anion gap is –45 mmol/L ([62 + 8] – 115), which supports diarrhea as the cause of his non-anion-gap acidosis. Sepsis, metformin use, or acute kidney injury would result in an anion-gap acidosis.
To manage acid-base disturbances, it is important to first determine whether there is a single primary disturbance with compensation or a mixed disorder. In the case of metabolic acidosis, for every 1-mmol/L decrease in bicarbonate, there should be a corresponding 1.3-mm Hg decrease in pCO2. Our patient’s laboratory data show that he had a pure non-anion-gap metabolic acidosis.8 His sensation of dyspnea was likely related to respiratory compensation as evidenced by an appropriately low pCO2.
CASE CONTINUED: HIS LABORATORY VALUES IMPROVE
The patient is admitted to the hospital for fluid resuscitation with normal saline and potassium and magnesium replacement.
Renal ultrasonography reveals normal-appearing kidneys without obstruction. The calculated fractional excretion of sodium is 3.4%. Urine microscopy reveals two to five hyaline casts per low-power field. His urine output remains adequate, and 3 days after hospitalization, his renal function starts to improve, as reflected in falling serum creatinine and blood urea nitrogen levels: his creatinine level has declined to 1.91 mg/dL and his blood urea nitrogen level has declined to 24 mg/dL. His acute kidney injury is attributed to intravenous contrast given for computed tomography, as well as to volume depletion and hypotension.
Stool studies for ova, parasites, and Clostridium difficile are negative. Fecal calprotectin and lactoferrin are useful noninvasive markers of intestinal inflammation but were not checked in this case.
Loperamide, taken as needed, is started for his diarrhea, along with empiric pancreatic enzyme replacement. After 3 days of treatment with oral vitamin K 10 mg, his international normalized ratio comes down to 1.4, from his admission value of 5.4. Given the clinical concern for fat malabsorption, vitamin D levels are also checked: his 25-hydroxyvitamin D level is less than 10 ng/mL (lower limit of normal 20). His fecal neutral fats are reported as normal, but split fats are increased.
STOOL FAT STUDIES
4. What does increased fecal split fats but normal fecal neutral fats imply?
- Pancreatic insufficiency
- Intestinal malabsorption
- Does not differentiate between the two
The finding does not differentiate between pancreatic insufficiency and intestinal malabsorption. The two-step Sudan stain has been used to differentiate maldigestion (eg, caused by pancreatic insufficiency) from malabsorption. Although patients with impaired digestion were once thought to excrete excessive amounts of intact triglyceride whereas those with malabsorption excrete more of the lipolytic or “split” product, the Sudan stain does not differentiate between the two.10 Stool fecal-elastase 1 testing correlates well with pancreatic exocrine function but was not performed in our patient.11
CASE CONTINUED: CELIAC DISEASE IS DIAGNOSED
Given the description of his stools, unintentional weight loss, and improvement of stool frequency with fasting, serologic testing for celiac disease is performed (Table 4). The patient undergoes esophagogastroduodenoscopy, which shows mild duodenitis. Small-bowel biopsy reveals blunted villous architecture and increased mixed inflammatory cells of the epithelium and lamina propria, suggestive of celiac disease.
The patient is diagnosed with celiac disease and is counseled to follow a gluten-free diet. He is discharged home and scheduled to follow up with a gastroenterologist and nephrologist. His liver function test abnormalities are attributed to a combination of nonalcoholic steatohepatitis and celiac disease.
CELIAC DISEASE AND MALABSORPTION
Celiac disease is an immune-mediated disorder that causes mucosal injury to the small intestine, leading to malabsorption. It is triggered by gluten intake in genetically susceptible individuals. The HLA-DQ2 haplotype is expressed in nearly 90% of patients with the disease.
The worldwide prevalence of celiac disease is about 0.6% to 1%. Those with an affected first-degree relative, type 1 diabetes, Hashimoto thyroiditis, an autoimmune disease, Down syndrome, Turner syndrome, or IgA deficiency have an increased risk.
Celiac disease presents with chronic diarrhea, weight loss, and abdominal distention and pain. Sequelae of nutrient malabsorption such as iron-deficiency anemia, short stature, and osteopenia may be evident. Liver function may also be impaired. Dermatitis herpetiformis and gluten ataxia are rarer presentations of celiac disease.12
In the absence of immunoglobulin (Ig) A deficiency, measurement of serum IgA anti-tissue transglutaminase antibodies is recommended for initial testing. IgG antitissue transglutaminase antibodies can be measured in those with IgA deficiency.12
Duodenal biopsies to confirm the diagnosis are recommended in adults unless they have previously had biopsy-proven dermatitis herpetiformis.
Gluten-free diet
The treatment for celiac disease is avoidance of gluten. Patients who consult with a nutritionist and participate in an advocacy group are more likely to adhere to a gluten-free diet, and the physician should strongly encourage and facilitate these activities.13
Untreated disease can lead to osteoporosis, impaired splenic function with increased risk of infection with encapsulated organisms, infertility or recurrent abortion, ulcerative jejunoileitis, and lymphoma.12 Patients should be monitored annually for adherence to the gluten-free diet and for the development of any associated condition. Despite adherence to a gluten-free diet, calcium absorption and bone mineral density are lower in patients with celiac disease than in controls.14 Careful monitoring of fracture risk and adequate calcium and vitamin D replacement are also important.
Our patient undergoes dual-emission x-ray absorptiometry after discharge, with results consistent with osteopenia. His T scores range from –0.2 at the right hip to –1.1 in the left femoral neck.
Recurrence or persistently abnormal levels of IgA anti-tissue transglutaminase antibodies usually indicates poor dietary compliance.12
5. In patients whose symptoms do not improve on gluten restriction, there should be concern for which of the following?
- Lymphoma
- Nonadherence to gluten restriction
- Microscopic colitis
- All of the above
The answer is all of the above. Up to 30% of patients have persistent symptoms on a gluten-free diet. Persistent exposure to gluten is the most common reason for lack of clinical improvement. In addition, bacterial overgrowth of the small bowel, lactose intolerance, pancreatic insufficiency, and microscopic colitis may coexist with celiac disease and may contribute to ongoing symptoms. In a small subset of patients with persistent villous atrophy and symptoms despite strict adherence to a gluten-free diet for 12 months, the disease is deemed “refractory.” Refractory celiac disease can be a precursor to enteropathy-associated T-cell lymphoma.13
CASE CONCLUDED
On telephone follow-up 3 weeks after discharge, the patient reports complete resolution of diarrhea and stabilization of his weight. He reports strict adherence to a gluten-free diet and feels he is coping well.
Diagnoses
- Presenting weakness secondary to dehydration and hypokalemia
- Dyspnea secondary to respiratory compensation for metabolic acidosis
- Non-anion-gap metabolic acidosis secondary to diarrhea
- Acute kidney injury secondary to iodinated contrast, volume depletion, hypotension
- Chronic diarrhea secondary to celiac disease
- Coagulopathy secondary to fat malabsorption secondary to celiac disease.
A 56-year-old man presents to the emergency department with nausea, weakness, and exertional dyspnea, which have been going on for 1 week. He is sent by his primary care physician after being noted to be hypotensive with a weak, thready pulse.
He has had diarrhea with intermittent abdominal pain over the past year, with 10 stools daily, including 3 or 4 at night. The stools are described as large, nonbloody, sticky, greasy, and occasionally watery. Stools are fewer when he curtails his food intake. The diarrhea is associated with occasional diffuse abdominal pain he describes as a burning sensation. He has no incontinence or tenesmus. He reports that he has unintentionally lost 137 lb (62 kg) over the past year. He has not taken over-the-counter antidiarrheal agents.
CHRONIC DIARRHEA
1. Chronic diarrhea is defined as lasting for at least how long?
- 1 week
- 2 weeks
- 3 weeks
- 4 weeks
Chronic diarrhea is defined as looser stools for more than 4 weeks,1 a period that allows most cases of acute, self-limited, infectious diarrhea to resolve.
Because individuals perceive diarrhea differently, reported prevalence rates of chronic diarrhea vary.2 Based on the definition of having excessive stool frequency, the prevalence in the United States is about 5%.1
In developing countries, the most common cause of chronic diarrhea is infection. In developed nations, irritable bowel syndrome, inflammatory bowel disease, malabsorption syndrome, and chronic infection predominate.1
Once chronicity is established, diarrhea should be characterized as inflammatory, fatty, or watery (Table 1).3
CASE CONTINUED: HISTORY OF HYPERTENSION, DIABETES
Our patient reports that he has never traveled outside the United States. He has a history of hypertension and type 2 diabetes mellitus that is controlled on oral agents. He has had surgery for a radial fracture and for reconstruction of his knees. He uses no tobacco, alcohol, or illicit drugs and works as a train engineer. He has no pets. He knows of no family history of inflammatory bowel disease or chronic diarrhea.
Comment. Patients with diabetes are at increased risk of gastrointestinal problems, with severity increasing with poorer control.4 Although our patient’s diabetes puts him at risk of diabetic autonomic neuropathy, his blood glucose control has been consistently good since his diagnosis, and his last measured hemoglobin A1c was 7.3% (reference range 4%–7%). His description of greasy stools in conjunction with his marked weight loss puts fatty diarrhea higher on the differential diagnosis.
DRUG-INDUCED DIARRHEA
His medications include glimepiride 1 mg twice daily, lisinopril 10 mg daily, metformin 500 mg twice daily, omeprazole 40 mg daily, and naproxen 220 mg daily. He has been taking metformin for at least 2 years. He is allergic to pentobarbital.
2. Which of his medications is least likely to be associated with his diarrhea?
- Lisinopril
- Metformin
- Glimepiride
- Naproxen
More than 700 drugs are known to cause diarrhea, often through the interplay of simultaneous mechanisms.5 The diagnosis of drug-induced diarrhea requires taking a careful medication history and establishing a temporal relationship between the drug and the diarrheal symptoms. Treatment consists of withdrawing the offending agent.
Nonsteroidal anti-inflammatory drugs (eg, naproxen) are associated with collagenous colitis that occurs mostly after long-term use (> 6 months). Metformin-induced diarrhea is related to fat malabsorption. Olmesartan, an angiotensin II receptor antagonist, has been associated with severe sprue-like enteropathy. On the other hand, the incidence of diarrhea with lisinopril is similar to that with placebo.7
CASE CONTINUED: EXAMINATION AND LABORATORY VALUES
The patient’s primary care physician had recently referred him to a gastroenterologist, and 4 days before presenting to the emergency department he had undergone abdominal and pelvic computed tomography (CT) with iodinated contrast, which had showed hepatic steatosis and pancreatic atrophy.
On examination now, the patient’s temperature is 97.5°F (36.4°C), heart rate 90 beats per minute, respirations 18 breaths per minute, oxygen saturation 99% on room air, and blood pressure 85/55 mm Hg. His body mass index is 32.5 kg/m2. His oral mucosa is dry. The rest of the examination is normal. No rash or ulcers are noted.
His laboratory values (Table 2) are notable for sodium 130 mmol/L, potassium 2.2 mmol/L, bicarbonate 9 mmol/L, blood urea nitrogen 32 mg/dL, creatinine 4.18 mg/dL, and international normalized ratio 5.4. Arterial blood gases drawn on admission reveal pH 7.32 and pCO2 19 mm Hg.
ACID-BASE DISTURBANCES
3. The patient’s acidosis is most likely related to which of the following?
- Sepsis
- Diarrhea
- Metformin
- Acute kidney injury
It is most likely related to diarrhea. The patient has a non-anion-gap metabolic acidosis. (The anion gap can be calculated by subtracting the sum of the serum bicarbonate and chloride values from the sodium—here, 130 – [112 + 9] = 9—and most textbooks list the reference range as 10–12 mmol/L.) Non-anion-gap metabolic acidosis results from excessive loss of bicarbonate or impaired ability of the kidney to excrete acid. Bicarbonate losses can occur in diarrhea or in ureteral diversion to the colon. Impairment in urinary acidification can occur in renal tubular acidosis.
To determine the cause of non-anion-gap acidosis, calculating the urine anion gap can be useful (Table 3), as it reflects the ability of the kidneys to excrete acid and is an indirect measure of ammonium excretion. Our patient’s urine anion gap is –45 mmol/L ([62 + 8] – 115), which supports diarrhea as the cause of his non-anion-gap acidosis. Sepsis, metformin use, or acute kidney injury would result in an anion-gap acidosis.
To manage acid-base disturbances, it is important to first determine whether there is a single primary disturbance with compensation or a mixed disorder. In the case of metabolic acidosis, for every 1-mmol/L decrease in bicarbonate, there should be a corresponding 1.3-mm Hg decrease in pCO2. Our patient’s laboratory data show that he had a pure non-anion-gap metabolic acidosis.8 His sensation of dyspnea was likely related to respiratory compensation as evidenced by an appropriately low pCO2.
CASE CONTINUED: HIS LABORATORY VALUES IMPROVE
The patient is admitted to the hospital for fluid resuscitation with normal saline and potassium and magnesium replacement.
Renal ultrasonography reveals normal-appearing kidneys without obstruction. The calculated fractional excretion of sodium is 3.4%. Urine microscopy reveals two to five hyaline casts per low-power field. His urine output remains adequate, and 3 days after hospitalization, his renal function starts to improve, as reflected in falling serum creatinine and blood urea nitrogen levels: his creatinine level has declined to 1.91 mg/dL and his blood urea nitrogen level has declined to 24 mg/dL. His acute kidney injury is attributed to intravenous contrast given for computed tomography, as well as to volume depletion and hypotension.
Stool studies for ova, parasites, and Clostridium difficile are negative. Fecal calprotectin and lactoferrin are useful noninvasive markers of intestinal inflammation but were not checked in this case.
Loperamide, taken as needed, is started for his diarrhea, along with empiric pancreatic enzyme replacement. After 3 days of treatment with oral vitamin K 10 mg, his international normalized ratio comes down to 1.4, from his admission value of 5.4. Given the clinical concern for fat malabsorption, vitamin D levels are also checked: his 25-hydroxyvitamin D level is less than 10 ng/mL (lower limit of normal 20). His fecal neutral fats are reported as normal, but split fats are increased.
STOOL FAT STUDIES
4. What does increased fecal split fats but normal fecal neutral fats imply?
- Pancreatic insufficiency
- Intestinal malabsorption
- Does not differentiate between the two
The finding does not differentiate between pancreatic insufficiency and intestinal malabsorption. The two-step Sudan stain has been used to differentiate maldigestion (eg, caused by pancreatic insufficiency) from malabsorption. Although patients with impaired digestion were once thought to excrete excessive amounts of intact triglyceride whereas those with malabsorption excrete more of the lipolytic or “split” product, the Sudan stain does not differentiate between the two.10 Stool fecal-elastase 1 testing correlates well with pancreatic exocrine function but was not performed in our patient.11
CASE CONTINUED: CELIAC DISEASE IS DIAGNOSED
Given the description of his stools, unintentional weight loss, and improvement of stool frequency with fasting, serologic testing for celiac disease is performed (Table 4). The patient undergoes esophagogastroduodenoscopy, which shows mild duodenitis. Small-bowel biopsy reveals blunted villous architecture and increased mixed inflammatory cells of the epithelium and lamina propria, suggestive of celiac disease.
The patient is diagnosed with celiac disease and is counseled to follow a gluten-free diet. He is discharged home and scheduled to follow up with a gastroenterologist and nephrologist. His liver function test abnormalities are attributed to a combination of nonalcoholic steatohepatitis and celiac disease.
CELIAC DISEASE AND MALABSORPTION
Celiac disease is an immune-mediated disorder that causes mucosal injury to the small intestine, leading to malabsorption. It is triggered by gluten intake in genetically susceptible individuals. The HLA-DQ2 haplotype is expressed in nearly 90% of patients with the disease.
The worldwide prevalence of celiac disease is about 0.6% to 1%. Those with an affected first-degree relative, type 1 diabetes, Hashimoto thyroiditis, an autoimmune disease, Down syndrome, Turner syndrome, or IgA deficiency have an increased risk.
Celiac disease presents with chronic diarrhea, weight loss, and abdominal distention and pain. Sequelae of nutrient malabsorption such as iron-deficiency anemia, short stature, and osteopenia may be evident. Liver function may also be impaired. Dermatitis herpetiformis and gluten ataxia are rarer presentations of celiac disease.12
In the absence of immunoglobulin (Ig) A deficiency, measurement of serum IgA anti-tissue transglutaminase antibodies is recommended for initial testing. IgG antitissue transglutaminase antibodies can be measured in those with IgA deficiency.12
Duodenal biopsies to confirm the diagnosis are recommended in adults unless they have previously had biopsy-proven dermatitis herpetiformis.
Gluten-free diet
The treatment for celiac disease is avoidance of gluten. Patients who consult with a nutritionist and participate in an advocacy group are more likely to adhere to a gluten-free diet, and the physician should strongly encourage and facilitate these activities.13
Untreated disease can lead to osteoporosis, impaired splenic function with increased risk of infection with encapsulated organisms, infertility or recurrent abortion, ulcerative jejunoileitis, and lymphoma.12 Patients should be monitored annually for adherence to the gluten-free diet and for the development of any associated condition. Despite adherence to a gluten-free diet, calcium absorption and bone mineral density are lower in patients with celiac disease than in controls.14 Careful monitoring of fracture risk and adequate calcium and vitamin D replacement are also important.
Our patient undergoes dual-emission x-ray absorptiometry after discharge, with results consistent with osteopenia. His T scores range from –0.2 at the right hip to –1.1 in the left femoral neck.
Recurrence or persistently abnormal levels of IgA anti-tissue transglutaminase antibodies usually indicates poor dietary compliance.12
5. In patients whose symptoms do not improve on gluten restriction, there should be concern for which of the following?
- Lymphoma
- Nonadherence to gluten restriction
- Microscopic colitis
- All of the above
The answer is all of the above. Up to 30% of patients have persistent symptoms on a gluten-free diet. Persistent exposure to gluten is the most common reason for lack of clinical improvement. In addition, bacterial overgrowth of the small bowel, lactose intolerance, pancreatic insufficiency, and microscopic colitis may coexist with celiac disease and may contribute to ongoing symptoms. In a small subset of patients with persistent villous atrophy and symptoms despite strict adherence to a gluten-free diet for 12 months, the disease is deemed “refractory.” Refractory celiac disease can be a precursor to enteropathy-associated T-cell lymphoma.13
CASE CONCLUDED
On telephone follow-up 3 weeks after discharge, the patient reports complete resolution of diarrhea and stabilization of his weight. He reports strict adherence to a gluten-free diet and feels he is coping well.
Diagnoses
- Presenting weakness secondary to dehydration and hypokalemia
- Dyspnea secondary to respiratory compensation for metabolic acidosis
- Non-anion-gap metabolic acidosis secondary to diarrhea
- Acute kidney injury secondary to iodinated contrast, volume depletion, hypotension
- Chronic diarrhea secondary to celiac disease
- Coagulopathy secondary to fat malabsorption secondary to celiac disease.
- Fine KD, Schiller LR. AGA technical review on the evaluation and management of chronic diarrhea. Gastroenterology 1999; 116:1464–1486.
- Talley NJ, Weaver AL, Zinsmeister AR, Melton LJ 3rd. Self-reported diarrhea: what does it mean? Am J Gastroenterol 1994; 89:1160–1164.
- Sweetser S. Evaluating the patient with diarrhea: a case-based approach. Mayo Clin Proc 2012; 87:596–602.
- Bytzer P, Talley NJ, Leemon M, Young LJ, Jones MP, Horowitz M. Prevalence of gastrointestinal symptoms associated with diabetes mellitus: a population-based survey of 15,000 adults. Arch Intern Med 2001; 161:1989–1996.
- Chassany O, Michaux A, Bergmann JF. Drug-induced diarrhoea. Drug Saf 2000; 22:53–72.
- Rubio-Tapia A, Herman ML, Ludvigsson JF, et al. Severe spruelike enteropathy associated with olmesartan. Mayo Clin Proc 2012; 87:732–738.
- Zestril (lisinopril) tablets. www.accessdata.fda.gov/drugsatfda_docs/label/2012/019777s062lbl.pdf. Accessed September 8, 2015.
- Whittier WL, Rutecki GW. Primer on clinical acid-base problem solving. Dis Mon 2004; 50:122–162.
- Langhorst J, Elsenbruch S, Koelzer J, Rueffer A, Michalsen A, Dobos GJ. Noninvasive markers in the assessment of intestinal inflammation in inflammatory bowel diseases: performance of fecal lactoferrin, calprotectin, and PMN-elastase, CRP, and clinical indices. Am J Gastroenterol 2008; 103:162–169.
- Khouri MR, Ng SN, Huang G, Shiau YF. Fecal triglyceride excretion is not excessive in pancreatic insufficiency. Gastroenterology 1989; 96:848–852.
- Dominici R, Franzini C. Fecal elastase-1 as a test for pancreatic function: a review. Clin Chem Lab Med 2002; 40:325–332.
- Fasano A, Catassi C. Celiac disease. New Engl J Med 2012; 367:2419–2426.
- Mooney PD, Hadjivassiliou M, Sanders DS. Coeliac disease. BMJ 2014; 348:g1561–g1561.
- Pazianas M, Butcher GP, Subhani JM, et al. Calcium absorption and bone mineral density in celiacs after long term treatment with gluten-free diet and adequate calcium intake. Osteoporos Int 2005; 16:56–63.
- Fine KD, Schiller LR. AGA technical review on the evaluation and management of chronic diarrhea. Gastroenterology 1999; 116:1464–1486.
- Talley NJ, Weaver AL, Zinsmeister AR, Melton LJ 3rd. Self-reported diarrhea: what does it mean? Am J Gastroenterol 1994; 89:1160–1164.
- Sweetser S. Evaluating the patient with diarrhea: a case-based approach. Mayo Clin Proc 2012; 87:596–602.
- Bytzer P, Talley NJ, Leemon M, Young LJ, Jones MP, Horowitz M. Prevalence of gastrointestinal symptoms associated with diabetes mellitus: a population-based survey of 15,000 adults. Arch Intern Med 2001; 161:1989–1996.
- Chassany O, Michaux A, Bergmann JF. Drug-induced diarrhoea. Drug Saf 2000; 22:53–72.
- Rubio-Tapia A, Herman ML, Ludvigsson JF, et al. Severe spruelike enteropathy associated with olmesartan. Mayo Clin Proc 2012; 87:732–738.
- Zestril (lisinopril) tablets. www.accessdata.fda.gov/drugsatfda_docs/label/2012/019777s062lbl.pdf. Accessed September 8, 2015.
- Whittier WL, Rutecki GW. Primer on clinical acid-base problem solving. Dis Mon 2004; 50:122–162.
- Langhorst J, Elsenbruch S, Koelzer J, Rueffer A, Michalsen A, Dobos GJ. Noninvasive markers in the assessment of intestinal inflammation in inflammatory bowel diseases: performance of fecal lactoferrin, calprotectin, and PMN-elastase, CRP, and clinical indices. Am J Gastroenterol 2008; 103:162–169.
- Khouri MR, Ng SN, Huang G, Shiau YF. Fecal triglyceride excretion is not excessive in pancreatic insufficiency. Gastroenterology 1989; 96:848–852.
- Dominici R, Franzini C. Fecal elastase-1 as a test for pancreatic function: a review. Clin Chem Lab Med 2002; 40:325–332.
- Fasano A, Catassi C. Celiac disease. New Engl J Med 2012; 367:2419–2426.
- Mooney PD, Hadjivassiliou M, Sanders DS. Coeliac disease. BMJ 2014; 348:g1561–g1561.
- Pazianas M, Butcher GP, Subhani JM, et al. Calcium absorption and bone mineral density in celiacs after long term treatment with gluten-free diet and adequate calcium intake. Osteoporos Int 2005; 16:56–63.
Are breast and pelvic exams necessary when prescribing hormonal contraception?
No. According to 2013 guidelines of the US Centers for Disease Control and Prevention (CDC),1 there is little evidence of benefit for many of the tests commonly mandated by healthcare providers before prescribing hormonal contraception (pill, ring, patch). These tests include breast and pelvic examinations, screening for cervical and sexually transmitted infections, laboratory testing, and mammography.
Only a medical history and blood pressure measurement are needed before prescribing estrogen-containing contraceptives. Patients who have elevated blood pressure but have not been previously diagnosed with hypertension should be preferentially offered other forms of contraception to avoid an additional risk of stroke or myocardial infarction, such as progestin-only products and intrauterine devices (IUDs). Women with blood pressures between 140/90 and 160/100 mm Hg may use estrogen-containing contraceptives only if other options are not appropriate. The CDC guidelines further state that if a patient is unable to come to the office for blood pressure assessment, then a community reading reported by the patient may be used to guide decision-making.
IS A PELVIC EXAMINATION NEEDED?
A pelvic examination (cervical inspection and bimanual examination) will not affect decisions related to prescribing contraceptives, except when prescribing female barrier methods (diaphragm, cervical cap) or IUDs.
Based on a systematic review of the literature between 1946 and 2014, the American College of Physicians now recommends against a screening pelvic examination in asymptomatic, nonpregnant, adult women when a Papanicolaou test is not otherwise indicated.2
The American College of Obstetricians and Gynecologists (ACOG) acknowledges that no current scientific evidence supports or refutes the need for an annual pelvic examination for an asymptomatic, low-risk patient. But ACOG supports pelvic examinations as a way to establish open communication with patients about sexual health and reproduction.3 ACOG also recommends an annual health visit for all women. Whether or not a pelvic examination is performed, women should be counseled annually about birth control and offered contraception.
Patients should also be encouraged to keep their preventive care up-to-date, including cervical cancer screening with a Papanicolaou test or a human papillomavirus test (or both) at appropriate intervals, especially if the patient has cervical abnormalities requiring follow-up. However, falling behind on preventive care should not be a barrier to obtaining contraception.
IMPROVING ADHERENCE, DECREASING UNINTENDED PREGNANCY
One goal of the CDC’s 2013 guidelines was to remove unnecessary barriers to women’s access to contraceptives. In the United States, half of all pregnancies are unintended, and almost half of unintended pregnancies lead to abortion.4 Only half of women who have had an abortion used any contraceptive method within the last month.5 This suggests high levels of unprotected and underprotected sex.
For most patients, several national societies now recommend long-acting reversible contraceptive (LARC) methods, which include IUDs and progestin-only arm implants, because they have lower failure rates in a real-world setting.1,6,7 LARC methods offer the advantage of the patient’s not having to remember to take, apply, or insert the contraceptive (ie, they are worry-free), and of not having to rely on a yearly appointment for refills.
The Contraceptive CHOICE Project8 was a large prospective cohort study that assessed the impact of offering contraception free of charge in St. Louis, Missouri. Most of the 9,256 women who participated selected a LARC method.8 Those taking combined hormonal contraceptives (ie, birth control pill, patch, or ring) had a higher contraceptive failure rate than those using LARC methods (4.55 vs 0.27 per 100 participant-years; hazard ratio after adjustment for age, education, and unintended pregnancy history, 21.8; 95% confidence interval 13.7–34.9). The rate of unintended pregnancy in those under age 21 using combined hormonal contraceptives was almost twice as high as in older participants. Subsequent analyses showed that the abortion rates in the St. Louis region decreased to less than a quarter of the national average after the start of this project.9
Given that the failure rate with combined hormonal contraceptives averages 9% per year,1 it is of the utmost importance that providers not limit access to patients’ prescriptions by requesting unnecessary visits and tests. If oral contraception is selected, women who are dispensed a full year’s supply of pill packs are more likely to continue with their contraceptive in the long term.10
THE PATIENT WITH A COMPLEX MEDICAL HISTORY
Limiting a woman’s contraceptive choices can increase her odds of experiencing an unintended pregnancy, which is associated with a far greater risk of adverse events than any contraceptive.11 Thus, the CDC developed separate guidelines in 2010 to help determine all available options for the patient with medical comorbidities and with a concerning family history (ie, breast cancer, venous thromboembolism).12 It can be helpful to consult the 2010 CDC medical eligibility criteria before offering contraception to these patients. Compared with the 2013 guidelines, which provide practical advice on how to use each contraceptive, the 2010 guidelines give guidance on when it is appropriate to prescribe each contraceptive—eg, which contraceptives are preferred based on a patient’s risk factors, medical history, and medication use. In addition to a two-page color summary chart of the 2010 medical eligibility criteria on the CDC website (https://www.cdc.gov/reproductivehealth/unintendedpregnancy/pdf/legal_summary-chart_english_final_tag508.pdf), a free mobile app is also available to guide decision-making.13
Pregnancy should be ruled out before initiating any contraceptive. This can be done through a detailed history. The six-item checklist in Table 1 has a 99.8% negative predictive value, so healthcare providers may be confident that a woman is not pregnant if pregnancy is excluded based on this history.14 A pregnancy test is needed in those who test positive on the checklist if they wish to start a LARC method such as an IUD or a progestin-only arm implant. However, because the test has a high false-positive rate, initiation of shorter-acting methods such as combined hormonal contraceptives should not be delayed on the basis of a positive checklist screen alone.1
Emergency contraception taken orally should be offered without an office visit, as its short duration of use allows women with traditional contraindications to hormonal contraceptives to safely use this birth control method.1,12 Because all emergency contraceptives must be used within 5 days of intercourse (the earlier the better), unnecessary office visits delay access and effectiveness.
Although a levonorgestrel-based emergency contraceptive is available over the counter, ulipristal acetate is more effective, especially in women who are overweight.15 A copper IUD placed within 5 days of intercourse is the most effective form of emergency contraception15 but requires an office visit. This method is an option for most women but should be strongly considered for women at highest risk of pregnancy (previous unintended pregnancy, intercourse at midcycle, obesity).
In summary, most women may safely begin their hormonal contraceptive with a detailed medical history alone, without additional office visits, examinations, or screening tests.
- Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control and Prevention (CDC). US selected practice recommendations for contraceptive use, 2013: adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm Rep 2013; 62:1–60.
- Qaseem A, Humphrey LL, Harris R, et al; Clinical Guidelines Committee of the American College of Physicians. Screening pelvic examination in adult women: a clinical practice guideline from the American College of Physicians. Ann Intern Med 2014; 161:67–72.
- American Congress of Obstetricians and Gynecologists. ACOG practice advisory on annual pelvic examination recommendations; 2014. www.acog.org/About-ACOG/News-Room/Practice-Advisories/ACOG-Practice-Advisory-on-Annual-Pelvic-Examination-Recommendations. Accessed September 8, 2015.
- Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception 2011; 84:478–485.
- Jones RK, Darroch JE, Henshaw SK. Contraceptive use among US women having abortions in 2000-2001. Perspect Sex Reprod Health 2002; 34:294–303.
- Committee on Health Care for Underserved Women. Committee opinion no. 615: access to contraception. Obstet Gynecol 2015; 125:250–255.
- Committee on Adolescent Health Care. Committee opinion no. 598: the initial reproductive health visit. Obstet Gynecol 2014; 123:1143–1147.
- Winner B, Peipert JF, Zhao Q, et al. Effectiveness of long-acting reversible contraception. N Engl J Med 2012; 366:1998–2007.
- Secura GM, Madden T, McNicholas C, et al. Provision of no-cost, long-acting contraception and teenage pregnancy. N Engl J Med 2014; 371:1316–1323.
- Committee on Gynecologic Practice, American College of Obstetricians and Gynecologists. Over-the-counter access to oral contraceptives. Committee opinion no 544. Obstet Gynecol 2012; 120:1527–1531.
- Committee on Gynecologic Practice. ACOG committee opinion number 540: risk of venous thromboembolism among users of drospirenone-containing oral contraceptive pills. Obstet Gynecol 2012; 120:1239–1242.
- Centers for Disease Control and Prevention (CDC). US medical eligibility criteria for contraceptive use, 2010. MMWR Recomm Rep 2010; 59:1–86.
- Centers for Disease Control and Prevention (CDC). United States medical eligibility criteria (US MEC) for contraceptive use, 2010. www.cdc.gov/reproductivehealth/unintendedpregnancy/usmec.htm. Accessed September 8, 2015.
- Min J, Buckel C, Secura GM, Peipert JF, Madden T. Performance of a checklist to exclude pregnancy at the time of contraceptive initiation among women with a negative urine pregnancy test. Contraception 2015; 91:80–84.
- Batur P. Emergency contraception: separating fact from fiction. Cleve Clin J Med 2012; 79:771–776.
No. According to 2013 guidelines of the US Centers for Disease Control and Prevention (CDC),1 there is little evidence of benefit for many of the tests commonly mandated by healthcare providers before prescribing hormonal contraception (pill, ring, patch). These tests include breast and pelvic examinations, screening for cervical and sexually transmitted infections, laboratory testing, and mammography.
Only a medical history and blood pressure measurement are needed before prescribing estrogen-containing contraceptives. Patients who have elevated blood pressure but have not been previously diagnosed with hypertension should be preferentially offered other forms of contraception to avoid an additional risk of stroke or myocardial infarction, such as progestin-only products and intrauterine devices (IUDs). Women with blood pressures between 140/90 and 160/100 mm Hg may use estrogen-containing contraceptives only if other options are not appropriate. The CDC guidelines further state that if a patient is unable to come to the office for blood pressure assessment, then a community reading reported by the patient may be used to guide decision-making.
IS A PELVIC EXAMINATION NEEDED?
A pelvic examination (cervical inspection and bimanual examination) will not affect decisions related to prescribing contraceptives, except when prescribing female barrier methods (diaphragm, cervical cap) or IUDs.
Based on a systematic review of the literature between 1946 and 2014, the American College of Physicians now recommends against a screening pelvic examination in asymptomatic, nonpregnant, adult women when a Papanicolaou test is not otherwise indicated.2
The American College of Obstetricians and Gynecologists (ACOG) acknowledges that no current scientific evidence supports or refutes the need for an annual pelvic examination for an asymptomatic, low-risk patient. But ACOG supports pelvic examinations as a way to establish open communication with patients about sexual health and reproduction.3 ACOG also recommends an annual health visit for all women. Whether or not a pelvic examination is performed, women should be counseled annually about birth control and offered contraception.
Patients should also be encouraged to keep their preventive care up-to-date, including cervical cancer screening with a Papanicolaou test or a human papillomavirus test (or both) at appropriate intervals, especially if the patient has cervical abnormalities requiring follow-up. However, falling behind on preventive care should not be a barrier to obtaining contraception.
IMPROVING ADHERENCE, DECREASING UNINTENDED PREGNANCY
One goal of the CDC’s 2013 guidelines was to remove unnecessary barriers to women’s access to contraceptives. In the United States, half of all pregnancies are unintended, and almost half of unintended pregnancies lead to abortion.4 Only half of women who have had an abortion used any contraceptive method within the last month.5 This suggests high levels of unprotected and underprotected sex.
For most patients, several national societies now recommend long-acting reversible contraceptive (LARC) methods, which include IUDs and progestin-only arm implants, because they have lower failure rates in a real-world setting.1,6,7 LARC methods offer the advantage of the patient’s not having to remember to take, apply, or insert the contraceptive (ie, they are worry-free), and of not having to rely on a yearly appointment for refills.
The Contraceptive CHOICE Project8 was a large prospective cohort study that assessed the impact of offering contraception free of charge in St. Louis, Missouri. Most of the 9,256 women who participated selected a LARC method.8 Those taking combined hormonal contraceptives (ie, birth control pill, patch, or ring) had a higher contraceptive failure rate than those using LARC methods (4.55 vs 0.27 per 100 participant-years; hazard ratio after adjustment for age, education, and unintended pregnancy history, 21.8; 95% confidence interval 13.7–34.9). The rate of unintended pregnancy in those under age 21 using combined hormonal contraceptives was almost twice as high as in older participants. Subsequent analyses showed that the abortion rates in the St. Louis region decreased to less than a quarter of the national average after the start of this project.9
Given that the failure rate with combined hormonal contraceptives averages 9% per year,1 it is of the utmost importance that providers not limit access to patients’ prescriptions by requesting unnecessary visits and tests. If oral contraception is selected, women who are dispensed a full year’s supply of pill packs are more likely to continue with their contraceptive in the long term.10
THE PATIENT WITH A COMPLEX MEDICAL HISTORY
Limiting a woman’s contraceptive choices can increase her odds of experiencing an unintended pregnancy, which is associated with a far greater risk of adverse events than any contraceptive.11 Thus, the CDC developed separate guidelines in 2010 to help determine all available options for the patient with medical comorbidities and with a concerning family history (ie, breast cancer, venous thromboembolism).12 It can be helpful to consult the 2010 CDC medical eligibility criteria before offering contraception to these patients. Compared with the 2013 guidelines, which provide practical advice on how to use each contraceptive, the 2010 guidelines give guidance on when it is appropriate to prescribe each contraceptive—eg, which contraceptives are preferred based on a patient’s risk factors, medical history, and medication use. In addition to a two-page color summary chart of the 2010 medical eligibility criteria on the CDC website (https://www.cdc.gov/reproductivehealth/unintendedpregnancy/pdf/legal_summary-chart_english_final_tag508.pdf), a free mobile app is also available to guide decision-making.13
Pregnancy should be ruled out before initiating any contraceptive. This can be done through a detailed history. The six-item checklist in Table 1 has a 99.8% negative predictive value, so healthcare providers may be confident that a woman is not pregnant if pregnancy is excluded based on this history.14 A pregnancy test is needed in those who test positive on the checklist if they wish to start a LARC method such as an IUD or a progestin-only arm implant. However, because the test has a high false-positive rate, initiation of shorter-acting methods such as combined hormonal contraceptives should not be delayed on the basis of a positive checklist screen alone.1
Emergency contraception taken orally should be offered without an office visit, as its short duration of use allows women with traditional contraindications to hormonal contraceptives to safely use this birth control method.1,12 Because all emergency contraceptives must be used within 5 days of intercourse (the earlier the better), unnecessary office visits delay access and effectiveness.
Although a levonorgestrel-based emergency contraceptive is available over the counter, ulipristal acetate is more effective, especially in women who are overweight.15 A copper IUD placed within 5 days of intercourse is the most effective form of emergency contraception15 but requires an office visit. This method is an option for most women but should be strongly considered for women at highest risk of pregnancy (previous unintended pregnancy, intercourse at midcycle, obesity).
In summary, most women may safely begin their hormonal contraceptive with a detailed medical history alone, without additional office visits, examinations, or screening tests.
No. According to 2013 guidelines of the US Centers for Disease Control and Prevention (CDC),1 there is little evidence of benefit for many of the tests commonly mandated by healthcare providers before prescribing hormonal contraception (pill, ring, patch). These tests include breast and pelvic examinations, screening for cervical and sexually transmitted infections, laboratory testing, and mammography.
Only a medical history and blood pressure measurement are needed before prescribing estrogen-containing contraceptives. Patients who have elevated blood pressure but have not been previously diagnosed with hypertension should be preferentially offered other forms of contraception to avoid an additional risk of stroke or myocardial infarction, such as progestin-only products and intrauterine devices (IUDs). Women with blood pressures between 140/90 and 160/100 mm Hg may use estrogen-containing contraceptives only if other options are not appropriate. The CDC guidelines further state that if a patient is unable to come to the office for blood pressure assessment, then a community reading reported by the patient may be used to guide decision-making.
IS A PELVIC EXAMINATION NEEDED?
A pelvic examination (cervical inspection and bimanual examination) will not affect decisions related to prescribing contraceptives, except when prescribing female barrier methods (diaphragm, cervical cap) or IUDs.
Based on a systematic review of the literature between 1946 and 2014, the American College of Physicians now recommends against a screening pelvic examination in asymptomatic, nonpregnant, adult women when a Papanicolaou test is not otherwise indicated.2
The American College of Obstetricians and Gynecologists (ACOG) acknowledges that no current scientific evidence supports or refutes the need for an annual pelvic examination for an asymptomatic, low-risk patient. But ACOG supports pelvic examinations as a way to establish open communication with patients about sexual health and reproduction.3 ACOG also recommends an annual health visit for all women. Whether or not a pelvic examination is performed, women should be counseled annually about birth control and offered contraception.
Patients should also be encouraged to keep their preventive care up-to-date, including cervical cancer screening with a Papanicolaou test or a human papillomavirus test (or both) at appropriate intervals, especially if the patient has cervical abnormalities requiring follow-up. However, falling behind on preventive care should not be a barrier to obtaining contraception.
IMPROVING ADHERENCE, DECREASING UNINTENDED PREGNANCY
One goal of the CDC’s 2013 guidelines was to remove unnecessary barriers to women’s access to contraceptives. In the United States, half of all pregnancies are unintended, and almost half of unintended pregnancies lead to abortion.4 Only half of women who have had an abortion used any contraceptive method within the last month.5 This suggests high levels of unprotected and underprotected sex.
For most patients, several national societies now recommend long-acting reversible contraceptive (LARC) methods, which include IUDs and progestin-only arm implants, because they have lower failure rates in a real-world setting.1,6,7 LARC methods offer the advantage of the patient’s not having to remember to take, apply, or insert the contraceptive (ie, they are worry-free), and of not having to rely on a yearly appointment for refills.
The Contraceptive CHOICE Project8 was a large prospective cohort study that assessed the impact of offering contraception free of charge in St. Louis, Missouri. Most of the 9,256 women who participated selected a LARC method.8 Those taking combined hormonal contraceptives (ie, birth control pill, patch, or ring) had a higher contraceptive failure rate than those using LARC methods (4.55 vs 0.27 per 100 participant-years; hazard ratio after adjustment for age, education, and unintended pregnancy history, 21.8; 95% confidence interval 13.7–34.9). The rate of unintended pregnancy in those under age 21 using combined hormonal contraceptives was almost twice as high as in older participants. Subsequent analyses showed that the abortion rates in the St. Louis region decreased to less than a quarter of the national average after the start of this project.9
Given that the failure rate with combined hormonal contraceptives averages 9% per year,1 it is of the utmost importance that providers not limit access to patients’ prescriptions by requesting unnecessary visits and tests. If oral contraception is selected, women who are dispensed a full year’s supply of pill packs are more likely to continue with their contraceptive in the long term.10
THE PATIENT WITH A COMPLEX MEDICAL HISTORY
Limiting a woman’s contraceptive choices can increase her odds of experiencing an unintended pregnancy, which is associated with a far greater risk of adverse events than any contraceptive.11 Thus, the CDC developed separate guidelines in 2010 to help determine all available options for the patient with medical comorbidities and with a concerning family history (ie, breast cancer, venous thromboembolism).12 It can be helpful to consult the 2010 CDC medical eligibility criteria before offering contraception to these patients. Compared with the 2013 guidelines, which provide practical advice on how to use each contraceptive, the 2010 guidelines give guidance on when it is appropriate to prescribe each contraceptive—eg, which contraceptives are preferred based on a patient’s risk factors, medical history, and medication use. In addition to a two-page color summary chart of the 2010 medical eligibility criteria on the CDC website (https://www.cdc.gov/reproductivehealth/unintendedpregnancy/pdf/legal_summary-chart_english_final_tag508.pdf), a free mobile app is also available to guide decision-making.13
Pregnancy should be ruled out before initiating any contraceptive. This can be done through a detailed history. The six-item checklist in Table 1 has a 99.8% negative predictive value, so healthcare providers may be confident that a woman is not pregnant if pregnancy is excluded based on this history.14 A pregnancy test is needed in those who test positive on the checklist if they wish to start a LARC method such as an IUD or a progestin-only arm implant. However, because the test has a high false-positive rate, initiation of shorter-acting methods such as combined hormonal contraceptives should not be delayed on the basis of a positive checklist screen alone.1
Emergency contraception taken orally should be offered without an office visit, as its short duration of use allows women with traditional contraindications to hormonal contraceptives to safely use this birth control method.1,12 Because all emergency contraceptives must be used within 5 days of intercourse (the earlier the better), unnecessary office visits delay access and effectiveness.
Although a levonorgestrel-based emergency contraceptive is available over the counter, ulipristal acetate is more effective, especially in women who are overweight.15 A copper IUD placed within 5 days of intercourse is the most effective form of emergency contraception15 but requires an office visit. This method is an option for most women but should be strongly considered for women at highest risk of pregnancy (previous unintended pregnancy, intercourse at midcycle, obesity).
In summary, most women may safely begin their hormonal contraceptive with a detailed medical history alone, without additional office visits, examinations, or screening tests.
- Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control and Prevention (CDC). US selected practice recommendations for contraceptive use, 2013: adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm Rep 2013; 62:1–60.
- Qaseem A, Humphrey LL, Harris R, et al; Clinical Guidelines Committee of the American College of Physicians. Screening pelvic examination in adult women: a clinical practice guideline from the American College of Physicians. Ann Intern Med 2014; 161:67–72.
- American Congress of Obstetricians and Gynecologists. ACOG practice advisory on annual pelvic examination recommendations; 2014. www.acog.org/About-ACOG/News-Room/Practice-Advisories/ACOG-Practice-Advisory-on-Annual-Pelvic-Examination-Recommendations. Accessed September 8, 2015.
- Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception 2011; 84:478–485.
- Jones RK, Darroch JE, Henshaw SK. Contraceptive use among US women having abortions in 2000-2001. Perspect Sex Reprod Health 2002; 34:294–303.
- Committee on Health Care for Underserved Women. Committee opinion no. 615: access to contraception. Obstet Gynecol 2015; 125:250–255.
- Committee on Adolescent Health Care. Committee opinion no. 598: the initial reproductive health visit. Obstet Gynecol 2014; 123:1143–1147.
- Winner B, Peipert JF, Zhao Q, et al. Effectiveness of long-acting reversible contraception. N Engl J Med 2012; 366:1998–2007.
- Secura GM, Madden T, McNicholas C, et al. Provision of no-cost, long-acting contraception and teenage pregnancy. N Engl J Med 2014; 371:1316–1323.
- Committee on Gynecologic Practice, American College of Obstetricians and Gynecologists. Over-the-counter access to oral contraceptives. Committee opinion no 544. Obstet Gynecol 2012; 120:1527–1531.
- Committee on Gynecologic Practice. ACOG committee opinion number 540: risk of venous thromboembolism among users of drospirenone-containing oral contraceptive pills. Obstet Gynecol 2012; 120:1239–1242.
- Centers for Disease Control and Prevention (CDC). US medical eligibility criteria for contraceptive use, 2010. MMWR Recomm Rep 2010; 59:1–86.
- Centers for Disease Control and Prevention (CDC). United States medical eligibility criteria (US MEC) for contraceptive use, 2010. www.cdc.gov/reproductivehealth/unintendedpregnancy/usmec.htm. Accessed September 8, 2015.
- Min J, Buckel C, Secura GM, Peipert JF, Madden T. Performance of a checklist to exclude pregnancy at the time of contraceptive initiation among women with a negative urine pregnancy test. Contraception 2015; 91:80–84.
- Batur P. Emergency contraception: separating fact from fiction. Cleve Clin J Med 2012; 79:771–776.
- Division of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control and Prevention (CDC). US selected practice recommendations for contraceptive use, 2013: adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm Rep 2013; 62:1–60.
- Qaseem A, Humphrey LL, Harris R, et al; Clinical Guidelines Committee of the American College of Physicians. Screening pelvic examination in adult women: a clinical practice guideline from the American College of Physicians. Ann Intern Med 2014; 161:67–72.
- American Congress of Obstetricians and Gynecologists. ACOG practice advisory on annual pelvic examination recommendations; 2014. www.acog.org/About-ACOG/News-Room/Practice-Advisories/ACOG-Practice-Advisory-on-Annual-Pelvic-Examination-Recommendations. Accessed September 8, 2015.
- Finer LB, Zolna MR. Unintended pregnancy in the United States: incidence and disparities, 2006. Contraception 2011; 84:478–485.
- Jones RK, Darroch JE, Henshaw SK. Contraceptive use among US women having abortions in 2000-2001. Perspect Sex Reprod Health 2002; 34:294–303.
- Committee on Health Care for Underserved Women. Committee opinion no. 615: access to contraception. Obstet Gynecol 2015; 125:250–255.
- Committee on Adolescent Health Care. Committee opinion no. 598: the initial reproductive health visit. Obstet Gynecol 2014; 123:1143–1147.
- Winner B, Peipert JF, Zhao Q, et al. Effectiveness of long-acting reversible contraception. N Engl J Med 2012; 366:1998–2007.
- Secura GM, Madden T, McNicholas C, et al. Provision of no-cost, long-acting contraception and teenage pregnancy. N Engl J Med 2014; 371:1316–1323.
- Committee on Gynecologic Practice, American College of Obstetricians and Gynecologists. Over-the-counter access to oral contraceptives. Committee opinion no 544. Obstet Gynecol 2012; 120:1527–1531.
- Committee on Gynecologic Practice. ACOG committee opinion number 540: risk of venous thromboembolism among users of drospirenone-containing oral contraceptive pills. Obstet Gynecol 2012; 120:1239–1242.
- Centers for Disease Control and Prevention (CDC). US medical eligibility criteria for contraceptive use, 2010. MMWR Recomm Rep 2010; 59:1–86.
- Centers for Disease Control and Prevention (CDC). United States medical eligibility criteria (US MEC) for contraceptive use, 2010. www.cdc.gov/reproductivehealth/unintendedpregnancy/usmec.htm. Accessed September 8, 2015.
- Min J, Buckel C, Secura GM, Peipert JF, Madden T. Performance of a checklist to exclude pregnancy at the time of contraceptive initiation among women with a negative urine pregnancy test. Contraception 2015; 91:80–84.
- Batur P. Emergency contraception: separating fact from fiction. Cleve Clin J Med 2012; 79:771–776.
Upper-limb deep vein thrombosis in Paget-Schroetter syndrome
A 43-year-old man with no medical history presented with pain and swelling in his left arm for 2 weeks. He was a regular weight lifter, and his exercise routine included repetitive hyperextension and hyperabduction of his arms while lifting heavy weights.
He had no history of recent trauma or venous cannulation of the left arm. His family history was negative for thrombophilic disorders. Physical examination revealed a swollen and erythematous left arm and visible venous collaterals at the neck, shoulder, and chest. There was no evidence of arterial insufficiency.
Duplex ultrasonography confirmed thrombosis of the left brachial, axillary, and subclavian veins. Further evaluation with computed tomography showed no intrathoracic mass but revealed several subsegmental pulmonary thrombi in the right lung. A screen for thrombophilia was negative. Venography confirmed complete thrombotic occlusion of the subclavian, axillary, and brachial veins (Figure 1).
Catheter-directed thrombolysis with tissue plasminogen activator resulted in complete resolution of the thrombosis, but venography after 3 days of thrombolysis showed 50% residual stenosis of the left subclavian vein where it passes under the first rib (Figure 2). The redness and swelling had markedly improved 2 days after thrombolytic therapy. He was discharged home on rivaroxaban 20 mg daily.
Follow-up venography 2 months later (Figure 3), with the patient performing hyperabduction of the arms, showed a patent subclavian vein with no thrombosis, but dynamic compression and occlusion of the subclavian vein where it passes the first rib. Magnetic resonance imaging (MRI) of the neck showed no cervical (ie, extra) rib and no soft-tissue abnormalities of the scalene triangle.
Following this, the patient underwent resection of the left first rib for decompression of the venous thoracic outlet, which resulted in resolution of his symptoms. He remained asymptomatic at 6-month follow-up.
PAGET-SCHROETTER SYNDROME
Paget-Schroetter syndrome, also referred to as effort-induced or effort thrombosis, is thrombosis of the axillary or subclavian vein associated with strenuous and repetitive activity of the arms. Anatomic abnormalities at the thoracic outlet—cervical rib, congenital bands, hypertrophy of scalene tendons, abnormal insertion of the costoclavicular ligament—and repetitive trauma to the endothelium of the subclavian vein are key factors in its initiation and progression.
The condition is seen primarily in young people who participate in strenuous activities such as rowing, weight lifting, and baseball pitching. It is estimated to be the cause of 40% of cases of primary upper-extremity deep vein thrombosis in the absence of an obvious risk factor or trigger such as a central venous catheter, pacemaker, port, or occult malignancy.1
A provocative test such as the Adson test or hyperabduction test during MRI or venography helps confirm thoracic outlet obstruction by demonstrating dynamic obstruction.2
TREATMENT CONSIDERATIONS
There are no universal guidelines for the treatment of Paget-Schroetter syndrome. However, the available data3–5 suggest a multimodal approach that involves early catheter-directed thrombolysis and subsequent surgical decompression of the thoracic outlet. This can restore venous patency and reduce the risk of long-term complications such as rethrombosis and postthrombotic syndrome.3–5
Surgical treatment includes resection of the first rib and division of the scalene muscles and the costoclavicular ligament. MRI with provocative testing helps guide the surgical approach. Anticoagulation therapy alone—ie, without thrombolysis and surgical decompression—is inadequate as it leads to recurrence of thrombosis and residual symptoms.6
Paget-Schroetter syndrome should not be managed the same as lower-extremity deep vein thrombosis because the cause and the exacerbating factors are different.
Unanswered questions
Because we have no data from randomized controlled trials, questions about management remain. What should be the duration of anticoagulation, especially in the absence of coexisting thrombophilia? Is thrombophilia screening useful? What is the optimal timing for starting thrombolytic therapy?
A careful history and heightened suspicion are required to make this diagnosis. If undiagnosed, it carries a risk of significant long-term morbidity and death. Dynamic obstruction during venography, in addition to MRI, can help identify an anatomic obstruction.
- Bernardi E, Pesavento R, Prandoni P. Upper extremity deep venous thrombosis. Semin Thromb Hemost 2006; 32:729–736.
- Demirbag D, Unlu E, Ozdemir F, et al. The relationship between magnetic resonance imaging findings and postural maneuver and physical examination tests in patients with thoracic outlet syndrome: results of a double-blind, controlled study. Arch Phys Med Rehabil 2007; 88:844–851.
- Alla VM, Natarajan N, Kaushik M, Warrier R, Nair CK. Paget-Schroetter syndrome: review of pathogenesis and treatment of effort thrombosis. West J Emerg Med 2010; 11:358–362.
- Molina JE, Hunter DW, Dietz CA. Paget-Schroetter syndrome treated with thrombolytics and immediate surgery. J Vasc Surg 2007; 45:328–334.
- Thompson RW. Comprehensive management of subclavian vein effort thrombosis. Semin Intervent Radiol 2012; 29:44–51.
- AbuRahma AF, Robinson PA. Effort subclavian vein thrombosis: evolution of management. J Endovasc Ther 2000; 7:302–308.
A 43-year-old man with no medical history presented with pain and swelling in his left arm for 2 weeks. He was a regular weight lifter, and his exercise routine included repetitive hyperextension and hyperabduction of his arms while lifting heavy weights.
He had no history of recent trauma or venous cannulation of the left arm. His family history was negative for thrombophilic disorders. Physical examination revealed a swollen and erythematous left arm and visible venous collaterals at the neck, shoulder, and chest. There was no evidence of arterial insufficiency.
Duplex ultrasonography confirmed thrombosis of the left brachial, axillary, and subclavian veins. Further evaluation with computed tomography showed no intrathoracic mass but revealed several subsegmental pulmonary thrombi in the right lung. A screen for thrombophilia was negative. Venography confirmed complete thrombotic occlusion of the subclavian, axillary, and brachial veins (Figure 1).
Catheter-directed thrombolysis with tissue plasminogen activator resulted in complete resolution of the thrombosis, but venography after 3 days of thrombolysis showed 50% residual stenosis of the left subclavian vein where it passes under the first rib (Figure 2). The redness and swelling had markedly improved 2 days after thrombolytic therapy. He was discharged home on rivaroxaban 20 mg daily.
Follow-up venography 2 months later (Figure 3), with the patient performing hyperabduction of the arms, showed a patent subclavian vein with no thrombosis, but dynamic compression and occlusion of the subclavian vein where it passes the first rib. Magnetic resonance imaging (MRI) of the neck showed no cervical (ie, extra) rib and no soft-tissue abnormalities of the scalene triangle.
Following this, the patient underwent resection of the left first rib for decompression of the venous thoracic outlet, which resulted in resolution of his symptoms. He remained asymptomatic at 6-month follow-up.
PAGET-SCHROETTER SYNDROME
Paget-Schroetter syndrome, also referred to as effort-induced or effort thrombosis, is thrombosis of the axillary or subclavian vein associated with strenuous and repetitive activity of the arms. Anatomic abnormalities at the thoracic outlet—cervical rib, congenital bands, hypertrophy of scalene tendons, abnormal insertion of the costoclavicular ligament—and repetitive trauma to the endothelium of the subclavian vein are key factors in its initiation and progression.
The condition is seen primarily in young people who participate in strenuous activities such as rowing, weight lifting, and baseball pitching. It is estimated to be the cause of 40% of cases of primary upper-extremity deep vein thrombosis in the absence of an obvious risk factor or trigger such as a central venous catheter, pacemaker, port, or occult malignancy.1
A provocative test such as the Adson test or hyperabduction test during MRI or venography helps confirm thoracic outlet obstruction by demonstrating dynamic obstruction.2
TREATMENT CONSIDERATIONS
There are no universal guidelines for the treatment of Paget-Schroetter syndrome. However, the available data3–5 suggest a multimodal approach that involves early catheter-directed thrombolysis and subsequent surgical decompression of the thoracic outlet. This can restore venous patency and reduce the risk of long-term complications such as rethrombosis and postthrombotic syndrome.3–5
Surgical treatment includes resection of the first rib and division of the scalene muscles and the costoclavicular ligament. MRI with provocative testing helps guide the surgical approach. Anticoagulation therapy alone—ie, without thrombolysis and surgical decompression—is inadequate as it leads to recurrence of thrombosis and residual symptoms.6
Paget-Schroetter syndrome should not be managed the same as lower-extremity deep vein thrombosis because the cause and the exacerbating factors are different.
Unanswered questions
Because we have no data from randomized controlled trials, questions about management remain. What should be the duration of anticoagulation, especially in the absence of coexisting thrombophilia? Is thrombophilia screening useful? What is the optimal timing for starting thrombolytic therapy?
A careful history and heightened suspicion are required to make this diagnosis. If undiagnosed, it carries a risk of significant long-term morbidity and death. Dynamic obstruction during venography, in addition to MRI, can help identify an anatomic obstruction.
A 43-year-old man with no medical history presented with pain and swelling in his left arm for 2 weeks. He was a regular weight lifter, and his exercise routine included repetitive hyperextension and hyperabduction of his arms while lifting heavy weights.
He had no history of recent trauma or venous cannulation of the left arm. His family history was negative for thrombophilic disorders. Physical examination revealed a swollen and erythematous left arm and visible venous collaterals at the neck, shoulder, and chest. There was no evidence of arterial insufficiency.
Duplex ultrasonography confirmed thrombosis of the left brachial, axillary, and subclavian veins. Further evaluation with computed tomography showed no intrathoracic mass but revealed several subsegmental pulmonary thrombi in the right lung. A screen for thrombophilia was negative. Venography confirmed complete thrombotic occlusion of the subclavian, axillary, and brachial veins (Figure 1).
Catheter-directed thrombolysis with tissue plasminogen activator resulted in complete resolution of the thrombosis, but venography after 3 days of thrombolysis showed 50% residual stenosis of the left subclavian vein where it passes under the first rib (Figure 2). The redness and swelling had markedly improved 2 days after thrombolytic therapy. He was discharged home on rivaroxaban 20 mg daily.
Follow-up venography 2 months later (Figure 3), with the patient performing hyperabduction of the arms, showed a patent subclavian vein with no thrombosis, but dynamic compression and occlusion of the subclavian vein where it passes the first rib. Magnetic resonance imaging (MRI) of the neck showed no cervical (ie, extra) rib and no soft-tissue abnormalities of the scalene triangle.
Following this, the patient underwent resection of the left first rib for decompression of the venous thoracic outlet, which resulted in resolution of his symptoms. He remained asymptomatic at 6-month follow-up.
PAGET-SCHROETTER SYNDROME
Paget-Schroetter syndrome, also referred to as effort-induced or effort thrombosis, is thrombosis of the axillary or subclavian vein associated with strenuous and repetitive activity of the arms. Anatomic abnormalities at the thoracic outlet—cervical rib, congenital bands, hypertrophy of scalene tendons, abnormal insertion of the costoclavicular ligament—and repetitive trauma to the endothelium of the subclavian vein are key factors in its initiation and progression.
The condition is seen primarily in young people who participate in strenuous activities such as rowing, weight lifting, and baseball pitching. It is estimated to be the cause of 40% of cases of primary upper-extremity deep vein thrombosis in the absence of an obvious risk factor or trigger such as a central venous catheter, pacemaker, port, or occult malignancy.1
A provocative test such as the Adson test or hyperabduction test during MRI or venography helps confirm thoracic outlet obstruction by demonstrating dynamic obstruction.2
TREATMENT CONSIDERATIONS
There are no universal guidelines for the treatment of Paget-Schroetter syndrome. However, the available data3–5 suggest a multimodal approach that involves early catheter-directed thrombolysis and subsequent surgical decompression of the thoracic outlet. This can restore venous patency and reduce the risk of long-term complications such as rethrombosis and postthrombotic syndrome.3–5
Surgical treatment includes resection of the first rib and division of the scalene muscles and the costoclavicular ligament. MRI with provocative testing helps guide the surgical approach. Anticoagulation therapy alone—ie, without thrombolysis and surgical decompression—is inadequate as it leads to recurrence of thrombosis and residual symptoms.6
Paget-Schroetter syndrome should not be managed the same as lower-extremity deep vein thrombosis because the cause and the exacerbating factors are different.
Unanswered questions
Because we have no data from randomized controlled trials, questions about management remain. What should be the duration of anticoagulation, especially in the absence of coexisting thrombophilia? Is thrombophilia screening useful? What is the optimal timing for starting thrombolytic therapy?
A careful history and heightened suspicion are required to make this diagnosis. If undiagnosed, it carries a risk of significant long-term morbidity and death. Dynamic obstruction during venography, in addition to MRI, can help identify an anatomic obstruction.
- Bernardi E, Pesavento R, Prandoni P. Upper extremity deep venous thrombosis. Semin Thromb Hemost 2006; 32:729–736.
- Demirbag D, Unlu E, Ozdemir F, et al. The relationship between magnetic resonance imaging findings and postural maneuver and physical examination tests in patients with thoracic outlet syndrome: results of a double-blind, controlled study. Arch Phys Med Rehabil 2007; 88:844–851.
- Alla VM, Natarajan N, Kaushik M, Warrier R, Nair CK. Paget-Schroetter syndrome: review of pathogenesis and treatment of effort thrombosis. West J Emerg Med 2010; 11:358–362.
- Molina JE, Hunter DW, Dietz CA. Paget-Schroetter syndrome treated with thrombolytics and immediate surgery. J Vasc Surg 2007; 45:328–334.
- Thompson RW. Comprehensive management of subclavian vein effort thrombosis. Semin Intervent Radiol 2012; 29:44–51.
- AbuRahma AF, Robinson PA. Effort subclavian vein thrombosis: evolution of management. J Endovasc Ther 2000; 7:302–308.
- Bernardi E, Pesavento R, Prandoni P. Upper extremity deep venous thrombosis. Semin Thromb Hemost 2006; 32:729–736.
- Demirbag D, Unlu E, Ozdemir F, et al. The relationship between magnetic resonance imaging findings and postural maneuver and physical examination tests in patients with thoracic outlet syndrome: results of a double-blind, controlled study. Arch Phys Med Rehabil 2007; 88:844–851.
- Alla VM, Natarajan N, Kaushik M, Warrier R, Nair CK. Paget-Schroetter syndrome: review of pathogenesis and treatment of effort thrombosis. West J Emerg Med 2010; 11:358–362.
- Molina JE, Hunter DW, Dietz CA. Paget-Schroetter syndrome treated with thrombolytics and immediate surgery. J Vasc Surg 2007; 45:328–334.
- Thompson RW. Comprehensive management of subclavian vein effort thrombosis. Semin Intervent Radiol 2012; 29:44–51.
- AbuRahma AF, Robinson PA. Effort subclavian vein thrombosis: evolution of management. J Endovasc Ther 2000; 7:302–308.