User login
What's your diagnosis?
The diagnosis
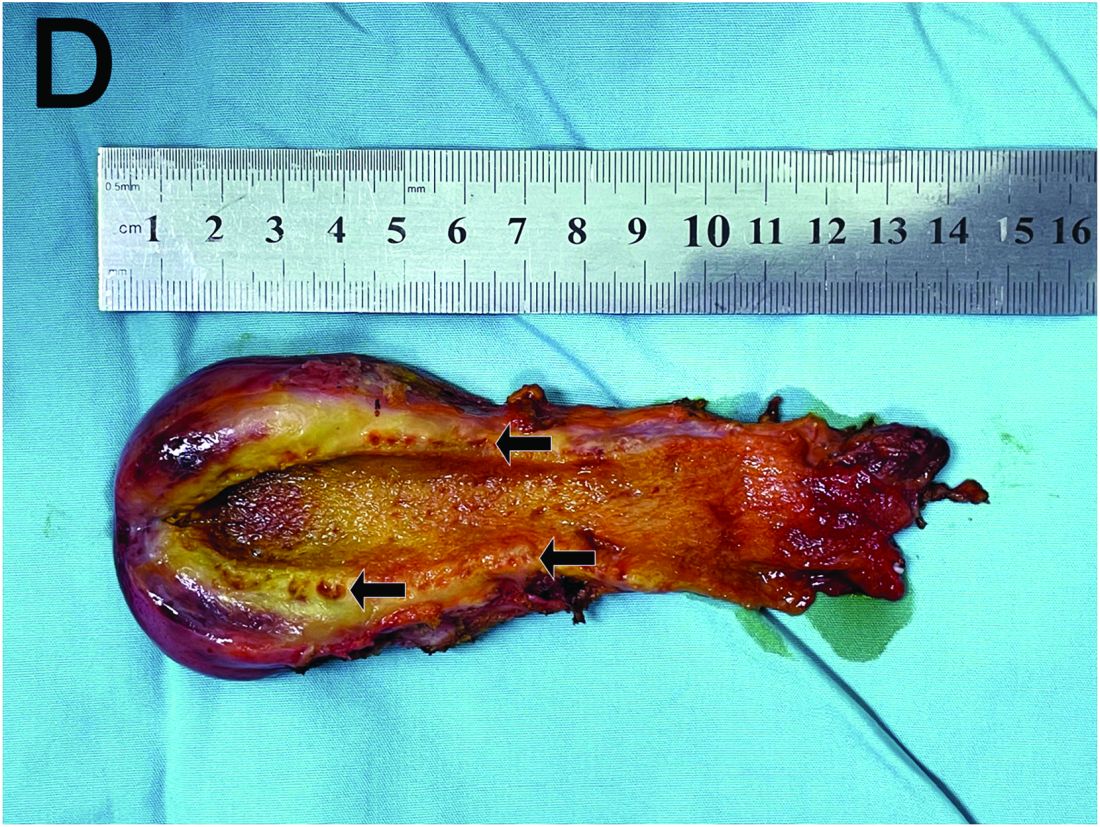
Based on the clinical and imaging findings, a diagnosis of gallbladder adenomyomatosis was made. GA is a benign and usually asymptomatic condition that occurs mainly beyond the age of 50-60 years and is very rare in childhood.1 Symptomatic gallbladder adenomyomatosis indicates cholecystectomy, considering the presence of inflammation or gallbladder stones.2 Therefore, a laparoscopic cholecystectomy was performed on our patient. Rokitansky-Aschoff sinuses were seen in the entire thickened gallbladder wall on gross pathologic examination (Figure D). Histopathologic examination confirmed the diagnosis of GA with cholecystitis. The patient was eventually diagnosed with diffuse GA. She was successfully discharged from the hospital 4 days after surgery, and 3 months of follow-up were uneventful.
References
Eroglu N et al. Diffuse adenomyomatosis of the gallbladder in a child. J Pediatr Hematol Oncol. 2016;38:e307-9.
Bonatti M. et al. Gallbladder adenomyomatosis: imaging findings, tricks and pitfalls. Insights Imaging. 2017;8:243-53.
Hammad AY et al. A literature review of radiological findings to guide the diagnosis of gallbladder adenomyomatosis. HPB (Oxford). 2016;18:129-35.
The diagnosis
Based on the clinical and imaging findings, a diagnosis of gallbladder adenomyomatosis was made. GA is a benign and usually asymptomatic condition that occurs mainly beyond the age of 50-60 years and is very rare in childhood.1 Symptomatic gallbladder adenomyomatosis indicates cholecystectomy, considering the presence of inflammation or gallbladder stones.2 Therefore, a laparoscopic cholecystectomy was performed on our patient. Rokitansky-Aschoff sinuses were seen in the entire thickened gallbladder wall on gross pathologic examination (Figure D). Histopathologic examination confirmed the diagnosis of GA with cholecystitis. The patient was eventually diagnosed with diffuse GA. She was successfully discharged from the hospital 4 days after surgery, and 3 months of follow-up were uneventful.

References
Eroglu N et al. Diffuse adenomyomatosis of the gallbladder in a child. J Pediatr Hematol Oncol. 2016;38:e307-9.
Bonatti M. et al. Gallbladder adenomyomatosis: imaging findings, tricks and pitfalls. Insights Imaging. 2017;8:243-53.
Hammad AY et al. A literature review of radiological findings to guide the diagnosis of gallbladder adenomyomatosis. HPB (Oxford). 2016;18:129-35.
The diagnosis
Based on the clinical and imaging findings, a diagnosis of gallbladder adenomyomatosis was made. GA is a benign and usually asymptomatic condition that occurs mainly beyond the age of 50-60 years and is very rare in childhood.1 Symptomatic gallbladder adenomyomatosis indicates cholecystectomy, considering the presence of inflammation or gallbladder stones.2 Therefore, a laparoscopic cholecystectomy was performed on our patient. Rokitansky-Aschoff sinuses were seen in the entire thickened gallbladder wall on gross pathologic examination (Figure D). Histopathologic examination confirmed the diagnosis of GA with cholecystitis. The patient was eventually diagnosed with diffuse GA. She was successfully discharged from the hospital 4 days after surgery, and 3 months of follow-up were uneventful.

References
Eroglu N et al. Diffuse adenomyomatosis of the gallbladder in a child. J Pediatr Hematol Oncol. 2016;38:e307-9.
Bonatti M. et al. Gallbladder adenomyomatosis: imaging findings, tricks and pitfalls. Insights Imaging. 2017;8:243-53.
Hammad AY et al. A literature review of radiological findings to guide the diagnosis of gallbladder adenomyomatosis. HPB (Oxford). 2016;18:129-35.
A 15-year-old girl presented with an 18-month history of intermittent right upper quadrant pain that appeared after meals and was relieved after rest. She denied any nausea, vomiting, chills, diarrhea, or constipation. The patient reported no trauma. At admission, physical examination showed tenderness in the right upper abdomen without rebound or guarding. Murphy's sign was also present. The laboratory tests were unremarkable.
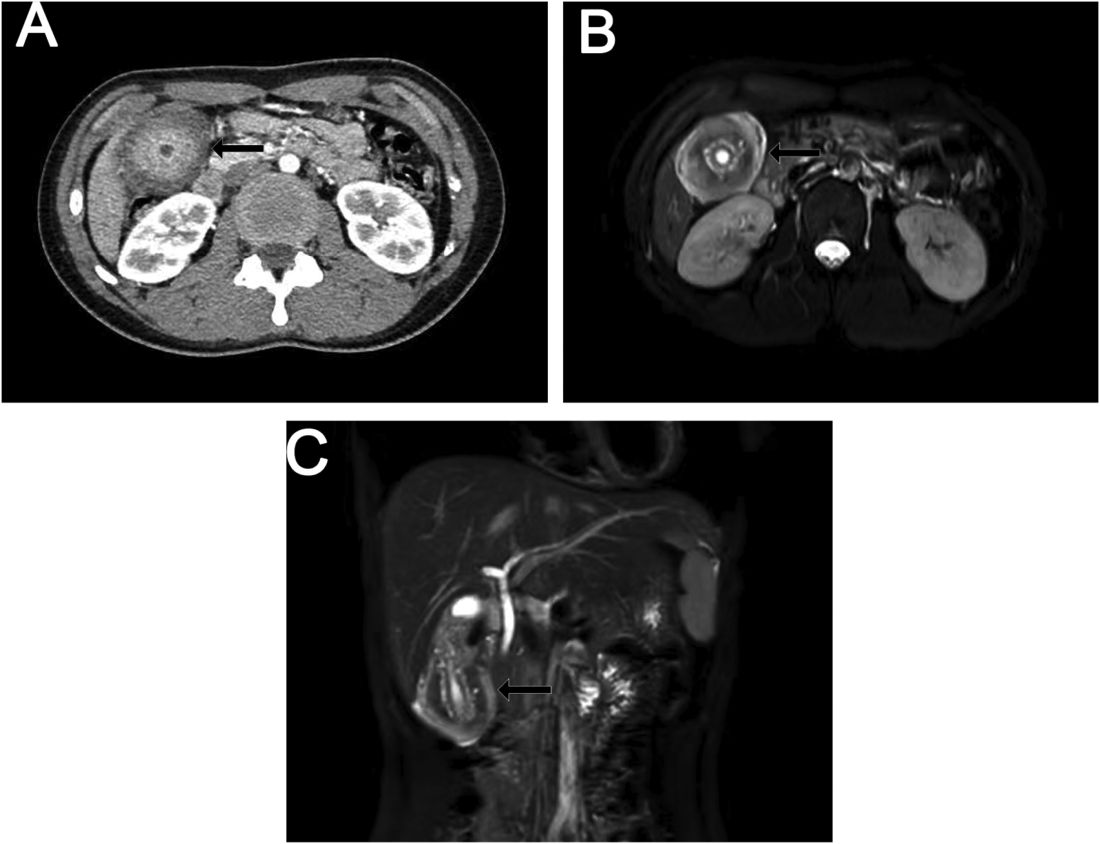
Ultrasound examination indicated gallbladder wall thickening. Furthermore, a contrast-enhanced computed tomographic scan showed marked gallbladder wall thickening with an annular unenhanced proliferative muscularis layer surrounding enhanced proliferative mucosal epithelium (Figure A), and magnetic resonance imaging showed multiple cyst-like spaces in the gallbladder wall (Figures B and C).
What is the diagnosis, and how should it be managed?
Previously published in Gastroenterology
Celiac disease: Update on diagnosis and monitoring
Celiac disease is a small bowel disorder. Specific antibodies along with a duodenal biopsy allow a secure diagnosis of celiac disease. Case detection rates have improved but many patients remain undiagnosed.
The only treatment available at present is a gluten-free diet (GFD). Most patients respond clinically to a GFD but histologic recovery is not always complete and may result in clinical consequences.
The anti-tissue transglutaminase IgA test (tTg-IgA) is the best initial serology test. A total IgA level appropriate for age is required to interpret a negative result. In patients with IgA deficiency, the deamidated gliadin peptide (DGP) antibodies, and/or tTg-IgA, may be helpful for diagnosis along with a duodenal biopsy.
First-degree female relatives with homozygous DQ2 positivity are at highest risk.
Both serology and duodenal biopsy have pitfalls in the diagnosis of celiac disease. In children, the diagnosis is secure with a tTg-IgA rate of at least 10 times the upper limit of normal (≥10×ULN) with positive endomysial antibodies (EMA).
There is less data on the correlation of tTg-IgA ≥10×ULN positive with villous atrophy in adults. All others require biopsy for diagnosis.
Considerations to forgo biopsy in adults include: tTg-IgA of ≥10×ULN positive, serology positive test in patients following GFD, or otherwise unable to undergo endoscopy with duodenal biopsy, or shared decision-making. Celiac disease recovery is assessed by clinical response to a GFD and antibody conversion to negative, which does not always correlate with histology.
Clinical consequences of persistent villous atrophy include increased risks for lymphoproliferative malignancy, hip fracture, and refractory celiac disease.
Dr. Semrad is director of the small bowel disease and nutrition program at the University of Chicago Medicine where she is a professor of medicine. She disclosed no conflicts of interest.
References
Rubio-Tapia et al. Am J Gastroenterol. 2023;118:59-76.
Husby S et al. J Pediatr Gastroenterol Nutr. 2020;70:141-57.
Celiac disease is a small bowel disorder. Specific antibodies along with a duodenal biopsy allow a secure diagnosis of celiac disease. Case detection rates have improved but many patients remain undiagnosed.
The only treatment available at present is a gluten-free diet (GFD). Most patients respond clinically to a GFD but histologic recovery is not always complete and may result in clinical consequences.
The anti-tissue transglutaminase IgA test (tTg-IgA) is the best initial serology test. A total IgA level appropriate for age is required to interpret a negative result. In patients with IgA deficiency, the deamidated gliadin peptide (DGP) antibodies, and/or tTg-IgA, may be helpful for diagnosis along with a duodenal biopsy.
First-degree female relatives with homozygous DQ2 positivity are at highest risk.
Both serology and duodenal biopsy have pitfalls in the diagnosis of celiac disease. In children, the diagnosis is secure with a tTg-IgA rate of at least 10 times the upper limit of normal (≥10×ULN) with positive endomysial antibodies (EMA).
There is less data on the correlation of tTg-IgA ≥10×ULN positive with villous atrophy in adults. All others require biopsy for diagnosis.
Considerations to forgo biopsy in adults include: tTg-IgA of ≥10×ULN positive, serology positive test in patients following GFD, or otherwise unable to undergo endoscopy with duodenal biopsy, or shared decision-making. Celiac disease recovery is assessed by clinical response to a GFD and antibody conversion to negative, which does not always correlate with histology.
Clinical consequences of persistent villous atrophy include increased risks for lymphoproliferative malignancy, hip fracture, and refractory celiac disease.
Dr. Semrad is director of the small bowel disease and nutrition program at the University of Chicago Medicine where she is a professor of medicine. She disclosed no conflicts of interest.
References
Rubio-Tapia et al. Am J Gastroenterol. 2023;118:59-76.
Husby S et al. J Pediatr Gastroenterol Nutr. 2020;70:141-57.
Celiac disease is a small bowel disorder. Specific antibodies along with a duodenal biopsy allow a secure diagnosis of celiac disease. Case detection rates have improved but many patients remain undiagnosed.
The only treatment available at present is a gluten-free diet (GFD). Most patients respond clinically to a GFD but histologic recovery is not always complete and may result in clinical consequences.
The anti-tissue transglutaminase IgA test (tTg-IgA) is the best initial serology test. A total IgA level appropriate for age is required to interpret a negative result. In patients with IgA deficiency, the deamidated gliadin peptide (DGP) antibodies, and/or tTg-IgA, may be helpful for diagnosis along with a duodenal biopsy.
First-degree female relatives with homozygous DQ2 positivity are at highest risk.
Both serology and duodenal biopsy have pitfalls in the diagnosis of celiac disease. In children, the diagnosis is secure with a tTg-IgA rate of at least 10 times the upper limit of normal (≥10×ULN) with positive endomysial antibodies (EMA).
There is less data on the correlation of tTg-IgA ≥10×ULN positive with villous atrophy in adults. All others require biopsy for diagnosis.
Considerations to forgo biopsy in adults include: tTg-IgA of ≥10×ULN positive, serology positive test in patients following GFD, or otherwise unable to undergo endoscopy with duodenal biopsy, or shared decision-making. Celiac disease recovery is assessed by clinical response to a GFD and antibody conversion to negative, which does not always correlate with histology.
Clinical consequences of persistent villous atrophy include increased risks for lymphoproliferative malignancy, hip fracture, and refractory celiac disease.
Dr. Semrad is director of the small bowel disease and nutrition program at the University of Chicago Medicine where she is a professor of medicine. She disclosed no conflicts of interest.
References
Rubio-Tapia et al. Am J Gastroenterol. 2023;118:59-76.
Husby S et al. J Pediatr Gastroenterol Nutr. 2020;70:141-57.
Advances in pancreaticobiliary disease interventions: More options and better outcomes
Highlights of advances in pancreaticobiliary disease interventions were reviewed at this year’s Digestive Disease Week (DDW) as part of the American Gastroenterological Association (AGA) postgraduate course.
Over the last several decades, the endoscopic treatment of pancreaticobiliary disease has advanced exponentially. Evidence-based advances are changing the landscape of pancreaticobiliary disease management.
While endoscopic retrograde cholangiopancreatography (ERCP) with transpapillary stent placement is first-line for the treatment of biliary obstruction, endoscopic ultrasound (EUS)-guided biliary drainage has emerged as an effective alternative in cases of failed ERCP. These procedures can be performed via a transhepatic approach (hepaticogastrostomy) from the proximal stomach, an extrahepatic approach (choledochoduodenostomy) from the duodenum, or via the gallbladder. Numerous studies have proved the safety and efficacy of these interventions in malignant biliary obstruction. A recent systematic meta-analysis pooled all of these approaches and concluded that EUS-guided biliary drainage is also reasonable to offer in benign disease when ERCP has failed or is not technically possible.
EUS-guided gallbladder drainage is similarly emerging as an alternative approach for management of acute cholecystitis. This is a reasonable option in patients with acute cholecystitis who are poor surgical candidates, have no evidence of gallbladder perforation, and will tolerate sedation. Moreover, this approach may be preferred over ERCP with cystic duct stent placement in the setting of a large stone burden, gastric outlet obstruction, or when an indwelling metal biliary stent occludes the cystic duct. Multidisciplinary discussion with surgical and interventional radiology services is essential, especially given this technique may preclude future cholecystectomy.
Indeterminate biliary strictures historically pose a major diagnostic challenge, and current approaches in the evaluation of such strictures lack diagnostic sensitivity. ERCP with concurrent brushing of the bile duct for cytology remains the most commonly used method of acquiring tissue. However, the sensitivity of diagnosis on brush cytology remains frustratingly low. Recent compelling evidence for increasing the number of brush passes to 30 in an indeterminate stricture improves diagnostic sensitivity and is a simple, safe, and low-cost intervention. This approach may ultimately decrease the number of patients requiring surgical intervention, which is particularly important when up to one-fifth of suspected biliary malignancies are found to be benign after surgical resection.
Not only have studies addressed increasing the diagnostic yield of stricture evaluation, but the treatment of biliary strictures has also evolved. Various stents are available, and different practice patterns have emerged for management of this entity. In an updated meta-analysis of randomized controlled trials evaluating multiple plastic stents versus a single covered metal stent for benign biliary strictures, no difference was found in stricture resolution, stricture recurrence, stent migration or adverse events. However, those patients treated with covered metal stents required fewer sessions of ERCP for stricture resolution. Moreover, no difference in stricture resolution was seen in subgroup analysis between anastomotic strictures, chronic pancreatitis, or bile duct injury. Despite higher cost of the stent itself, covered metal stents may ultimately lead to an overall decrease in health care expenditure.
The above examples are only a small subset of the progress that has been made in endoscopic management of pancreaticobiliary disease. The armamentarium of tools and techniques will continue to evolve to help us provide better minimally invasive care for our patients.
Dr. Schulman is associate professor in the division of gastroenterology and hepatology and the department of surgery at the University of Michigan. She is the incoming chief of endoscopy and the director of bariatric endoscopy. She disclosed consultancy work with Apollo Endosurgery, Boston Scientific, Olympus and MicroTech. She also disclosed research and grant support from GI Dynamics and Fractyl.
Highlights of advances in pancreaticobiliary disease interventions were reviewed at this year’s Digestive Disease Week (DDW) as part of the American Gastroenterological Association (AGA) postgraduate course.
Over the last several decades, the endoscopic treatment of pancreaticobiliary disease has advanced exponentially. Evidence-based advances are changing the landscape of pancreaticobiliary disease management.
While endoscopic retrograde cholangiopancreatography (ERCP) with transpapillary stent placement is first-line for the treatment of biliary obstruction, endoscopic ultrasound (EUS)-guided biliary drainage has emerged as an effective alternative in cases of failed ERCP. These procedures can be performed via a transhepatic approach (hepaticogastrostomy) from the proximal stomach, an extrahepatic approach (choledochoduodenostomy) from the duodenum, or via the gallbladder. Numerous studies have proved the safety and efficacy of these interventions in malignant biliary obstruction. A recent systematic meta-analysis pooled all of these approaches and concluded that EUS-guided biliary drainage is also reasonable to offer in benign disease when ERCP has failed or is not technically possible.
EUS-guided gallbladder drainage is similarly emerging as an alternative approach for management of acute cholecystitis. This is a reasonable option in patients with acute cholecystitis who are poor surgical candidates, have no evidence of gallbladder perforation, and will tolerate sedation. Moreover, this approach may be preferred over ERCP with cystic duct stent placement in the setting of a large stone burden, gastric outlet obstruction, or when an indwelling metal biliary stent occludes the cystic duct. Multidisciplinary discussion with surgical and interventional radiology services is essential, especially given this technique may preclude future cholecystectomy.
Indeterminate biliary strictures historically pose a major diagnostic challenge, and current approaches in the evaluation of such strictures lack diagnostic sensitivity. ERCP with concurrent brushing of the bile duct for cytology remains the most commonly used method of acquiring tissue. However, the sensitivity of diagnosis on brush cytology remains frustratingly low. Recent compelling evidence for increasing the number of brush passes to 30 in an indeterminate stricture improves diagnostic sensitivity and is a simple, safe, and low-cost intervention. This approach may ultimately decrease the number of patients requiring surgical intervention, which is particularly important when up to one-fifth of suspected biliary malignancies are found to be benign after surgical resection.
Not only have studies addressed increasing the diagnostic yield of stricture evaluation, but the treatment of biliary strictures has also evolved. Various stents are available, and different practice patterns have emerged for management of this entity. In an updated meta-analysis of randomized controlled trials evaluating multiple plastic stents versus a single covered metal stent for benign biliary strictures, no difference was found in stricture resolution, stricture recurrence, stent migration or adverse events. However, those patients treated with covered metal stents required fewer sessions of ERCP for stricture resolution. Moreover, no difference in stricture resolution was seen in subgroup analysis between anastomotic strictures, chronic pancreatitis, or bile duct injury. Despite higher cost of the stent itself, covered metal stents may ultimately lead to an overall decrease in health care expenditure.
The above examples are only a small subset of the progress that has been made in endoscopic management of pancreaticobiliary disease. The armamentarium of tools and techniques will continue to evolve to help us provide better minimally invasive care for our patients.
Dr. Schulman is associate professor in the division of gastroenterology and hepatology and the department of surgery at the University of Michigan. She is the incoming chief of endoscopy and the director of bariatric endoscopy. She disclosed consultancy work with Apollo Endosurgery, Boston Scientific, Olympus and MicroTech. She also disclosed research and grant support from GI Dynamics and Fractyl.
Highlights of advances in pancreaticobiliary disease interventions were reviewed at this year’s Digestive Disease Week (DDW) as part of the American Gastroenterological Association (AGA) postgraduate course.
Over the last several decades, the endoscopic treatment of pancreaticobiliary disease has advanced exponentially. Evidence-based advances are changing the landscape of pancreaticobiliary disease management.
While endoscopic retrograde cholangiopancreatography (ERCP) with transpapillary stent placement is first-line for the treatment of biliary obstruction, endoscopic ultrasound (EUS)-guided biliary drainage has emerged as an effective alternative in cases of failed ERCP. These procedures can be performed via a transhepatic approach (hepaticogastrostomy) from the proximal stomach, an extrahepatic approach (choledochoduodenostomy) from the duodenum, or via the gallbladder. Numerous studies have proved the safety and efficacy of these interventions in malignant biliary obstruction. A recent systematic meta-analysis pooled all of these approaches and concluded that EUS-guided biliary drainage is also reasonable to offer in benign disease when ERCP has failed or is not technically possible.
EUS-guided gallbladder drainage is similarly emerging as an alternative approach for management of acute cholecystitis. This is a reasonable option in patients with acute cholecystitis who are poor surgical candidates, have no evidence of gallbladder perforation, and will tolerate sedation. Moreover, this approach may be preferred over ERCP with cystic duct stent placement in the setting of a large stone burden, gastric outlet obstruction, or when an indwelling metal biliary stent occludes the cystic duct. Multidisciplinary discussion with surgical and interventional radiology services is essential, especially given this technique may preclude future cholecystectomy.
Indeterminate biliary strictures historically pose a major diagnostic challenge, and current approaches in the evaluation of such strictures lack diagnostic sensitivity. ERCP with concurrent brushing of the bile duct for cytology remains the most commonly used method of acquiring tissue. However, the sensitivity of diagnosis on brush cytology remains frustratingly low. Recent compelling evidence for increasing the number of brush passes to 30 in an indeterminate stricture improves diagnostic sensitivity and is a simple, safe, and low-cost intervention. This approach may ultimately decrease the number of patients requiring surgical intervention, which is particularly important when up to one-fifth of suspected biliary malignancies are found to be benign after surgical resection.
Not only have studies addressed increasing the diagnostic yield of stricture evaluation, but the treatment of biliary strictures has also evolved. Various stents are available, and different practice patterns have emerged for management of this entity. In an updated meta-analysis of randomized controlled trials evaluating multiple plastic stents versus a single covered metal stent for benign biliary strictures, no difference was found in stricture resolution, stricture recurrence, stent migration or adverse events. However, those patients treated with covered metal stents required fewer sessions of ERCP for stricture resolution. Moreover, no difference in stricture resolution was seen in subgroup analysis between anastomotic strictures, chronic pancreatitis, or bile duct injury. Despite higher cost of the stent itself, covered metal stents may ultimately lead to an overall decrease in health care expenditure.
The above examples are only a small subset of the progress that has been made in endoscopic management of pancreaticobiliary disease. The armamentarium of tools and techniques will continue to evolve to help us provide better minimally invasive care for our patients.
Dr. Schulman is associate professor in the division of gastroenterology and hepatology and the department of surgery at the University of Michigan. She is the incoming chief of endoscopy and the director of bariatric endoscopy. She disclosed consultancy work with Apollo Endosurgery, Boston Scientific, Olympus and MicroTech. She also disclosed research and grant support from GI Dynamics and Fractyl.
Conflicting blood pressure targets: Déjà vu all over again
Stop me if you’ve heard this before. There’s a controversy over blood pressure targets. Some argue for 140/90 mm Hg, others for 130/80 mm Hg, and some super ambitious folks think that we should aim for 120/80 mm Hg. If this sounds familiar, it should. We did it in 2017. It’s unclear what, if anything, we learned from the experience. On the upside, it’s not as bad as it was 100 years ago.
When high blood pressure was a ‘good’ thing
Back then, many believed that you needed higher blood pressure as you got older to push the blood through your progressively stiffened and hardened arteries. Hence the name “essential” hypertension. The concern was that lowering blood pressure would hypoperfuse your organs and be dangerous. In the 1930s, John Hay told an audience at a British Medical Association lecture: “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.”
The 1900s were a simpler time when people had fatal strokes in their 50s, and their families were consoled by the knowledge that they had lived a good life.
If our thinking around blood pressure had evolved slightly faster, perhaps President Roosevelt wouldn’t have died of a stroke during World War II as his doctors watched his systolic blood pressure climb above 200 mm Hg and suggested massages and barbiturates to take the edge off.
The current controversy
Not that long ago, 180 mm Hg was considered mild hypertension. Now, we are arguing about a systolic blood pressure of 140 versus 130 mm Hg.
The American Academy of Family Physicians takes the view that 140/90 mm Hg is good enough for most people. Their most recent clinical practice guideline, based primarily on two 2020 Cochrane Reviews of blood pressure targets in patients with and without cardiovascular disease, did not find any mortality benefit for a lower blood pressure threshold.
This puts the AAFP guideline in conflict with the 2017 guideline issued jointly by the American College of Cardiology, American Heart Association, and nine other groups, which recommended a target of 130/80 mm Hg for pretty much everyone. Though they say greater than 140/90 mm Hg should be the threshold for low-risk patients or for starting therapy post stroke, we often forget those nuances. The main point of contention is that the AAFP guideline was looking for a mortality benefit, whereas the ACC/AHA/everyone else guideline was looking at preventing cardiovascular events. The latter guideline was driven mainly by the results of the SPRINT trial. ACC/AHA argue for more aggressive targets to prevent the things that cardiologists care about, namely heart attacks.
The AAFP guideline conceded that more aggressive control will result in fewer myocardial infarctions but warn that it comes with more adverse events. Treating 1,000 patients to this lower target would theoretically prevent four MIs, possibly prevent three strokes, but result in 30 adverse events.
In the end, what we are seeing here is not so much a debate over the evidence as a debate over priorities. Interventions that don’t improve mortality can be questioned in terms of their cost effectiveness. But you probably don’t want to have a heart attack (even a nonfatal one). And you certainly don’t want to have a stroke. However, lower blood pressure targets inevitably require more medications. Notwithstanding the economic costs, the dangers of polypharmacy, medication interactions, side effects, and syncope leading to falls cannot be ignored. Falls are not benign adverse events, especially in older adults.
The counter argument is that physicians are human and often let things slide. Set the target at 140/90 mm Hg, and many physicians won’t jump on a systolic blood pressure of 144 mm Hg. Set the target at 130 mm Hg, and maybe they’ll be more likely to react. There’s a fine line between permissiveness and complacency.
If you zoom out and look at the multitude of blood pressure guidelines, you start to notice an important fact. There is not much daylight between them. There are subtle differences in what constitutes high risk and different definitions of older (older should be defined as 10 years older than the reader’s current age). But otherwise, the blood pressure targets are not that different.
Does that final 10 mm Hg really matter when barriers to care mean that tens of millions in the United States are unaware they have hypertension? Even among those diagnosed, many are either untreated or inadequately treated.
With this context, perhaps the most insightful thing that can be said about the blood pressure guideline controversy is that it’s not all that controversial. We can likely all agree that we need to be better at treating hypertension and that creative solutions to reach underserved communities are necessary.
Arguing about 140/90 mm Hg or 130/80 mm Hg is less important than acknowledging that we should be aggressive in screening for and treating hypertension. We should acknowledge that beyond a certain point any cardiovascular benefit comes at the cost of hypotension and side effects. That tipping point will be different for different groups, and probably at a higher set point in older patients.
Individualizing care isn’t difficult. We do it all the time. We just shouldn’t be letting people walk around with untreated hypertension. It’s not the 1900s anymore.
Dr. Labos is a cardiologist at Hôpital Notre-Dame, Montreal. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Stop me if you’ve heard this before. There’s a controversy over blood pressure targets. Some argue for 140/90 mm Hg, others for 130/80 mm Hg, and some super ambitious folks think that we should aim for 120/80 mm Hg. If this sounds familiar, it should. We did it in 2017. It’s unclear what, if anything, we learned from the experience. On the upside, it’s not as bad as it was 100 years ago.
When high blood pressure was a ‘good’ thing
Back then, many believed that you needed higher blood pressure as you got older to push the blood through your progressively stiffened and hardened arteries. Hence the name “essential” hypertension. The concern was that lowering blood pressure would hypoperfuse your organs and be dangerous. In the 1930s, John Hay told an audience at a British Medical Association lecture: “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.”
The 1900s were a simpler time when people had fatal strokes in their 50s, and their families were consoled by the knowledge that they had lived a good life.
If our thinking around blood pressure had evolved slightly faster, perhaps President Roosevelt wouldn’t have died of a stroke during World War II as his doctors watched his systolic blood pressure climb above 200 mm Hg and suggested massages and barbiturates to take the edge off.
The current controversy
Not that long ago, 180 mm Hg was considered mild hypertension. Now, we are arguing about a systolic blood pressure of 140 versus 130 mm Hg.
The American Academy of Family Physicians takes the view that 140/90 mm Hg is good enough for most people. Their most recent clinical practice guideline, based primarily on two 2020 Cochrane Reviews of blood pressure targets in patients with and without cardiovascular disease, did not find any mortality benefit for a lower blood pressure threshold.
This puts the AAFP guideline in conflict with the 2017 guideline issued jointly by the American College of Cardiology, American Heart Association, and nine other groups, which recommended a target of 130/80 mm Hg for pretty much everyone. Though they say greater than 140/90 mm Hg should be the threshold for low-risk patients or for starting therapy post stroke, we often forget those nuances. The main point of contention is that the AAFP guideline was looking for a mortality benefit, whereas the ACC/AHA/everyone else guideline was looking at preventing cardiovascular events. The latter guideline was driven mainly by the results of the SPRINT trial. ACC/AHA argue for more aggressive targets to prevent the things that cardiologists care about, namely heart attacks.
The AAFP guideline conceded that more aggressive control will result in fewer myocardial infarctions but warn that it comes with more adverse events. Treating 1,000 patients to this lower target would theoretically prevent four MIs, possibly prevent three strokes, but result in 30 adverse events.
In the end, what we are seeing here is not so much a debate over the evidence as a debate over priorities. Interventions that don’t improve mortality can be questioned in terms of their cost effectiveness. But you probably don’t want to have a heart attack (even a nonfatal one). And you certainly don’t want to have a stroke. However, lower blood pressure targets inevitably require more medications. Notwithstanding the economic costs, the dangers of polypharmacy, medication interactions, side effects, and syncope leading to falls cannot be ignored. Falls are not benign adverse events, especially in older adults.
The counter argument is that physicians are human and often let things slide. Set the target at 140/90 mm Hg, and many physicians won’t jump on a systolic blood pressure of 144 mm Hg. Set the target at 130 mm Hg, and maybe they’ll be more likely to react. There’s a fine line between permissiveness and complacency.
If you zoom out and look at the multitude of blood pressure guidelines, you start to notice an important fact. There is not much daylight between them. There are subtle differences in what constitutes high risk and different definitions of older (older should be defined as 10 years older than the reader’s current age). But otherwise, the blood pressure targets are not that different.
Does that final 10 mm Hg really matter when barriers to care mean that tens of millions in the United States are unaware they have hypertension? Even among those diagnosed, many are either untreated or inadequately treated.
With this context, perhaps the most insightful thing that can be said about the blood pressure guideline controversy is that it’s not all that controversial. We can likely all agree that we need to be better at treating hypertension and that creative solutions to reach underserved communities are necessary.
Arguing about 140/90 mm Hg or 130/80 mm Hg is less important than acknowledging that we should be aggressive in screening for and treating hypertension. We should acknowledge that beyond a certain point any cardiovascular benefit comes at the cost of hypotension and side effects. That tipping point will be different for different groups, and probably at a higher set point in older patients.
Individualizing care isn’t difficult. We do it all the time. We just shouldn’t be letting people walk around with untreated hypertension. It’s not the 1900s anymore.
Dr. Labos is a cardiologist at Hôpital Notre-Dame, Montreal. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Stop me if you’ve heard this before. There’s a controversy over blood pressure targets. Some argue for 140/90 mm Hg, others for 130/80 mm Hg, and some super ambitious folks think that we should aim for 120/80 mm Hg. If this sounds familiar, it should. We did it in 2017. It’s unclear what, if anything, we learned from the experience. On the upside, it’s not as bad as it was 100 years ago.
When high blood pressure was a ‘good’ thing
Back then, many believed that you needed higher blood pressure as you got older to push the blood through your progressively stiffened and hardened arteries. Hence the name “essential” hypertension. The concern was that lowering blood pressure would hypoperfuse your organs and be dangerous. In the 1930s, John Hay told an audience at a British Medical Association lecture: “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.”
The 1900s were a simpler time when people had fatal strokes in their 50s, and their families were consoled by the knowledge that they had lived a good life.
If our thinking around blood pressure had evolved slightly faster, perhaps President Roosevelt wouldn’t have died of a stroke during World War II as his doctors watched his systolic blood pressure climb above 200 mm Hg and suggested massages and barbiturates to take the edge off.
The current controversy
Not that long ago, 180 mm Hg was considered mild hypertension. Now, we are arguing about a systolic blood pressure of 140 versus 130 mm Hg.
The American Academy of Family Physicians takes the view that 140/90 mm Hg is good enough for most people. Their most recent clinical practice guideline, based primarily on two 2020 Cochrane Reviews of blood pressure targets in patients with and without cardiovascular disease, did not find any mortality benefit for a lower blood pressure threshold.
This puts the AAFP guideline in conflict with the 2017 guideline issued jointly by the American College of Cardiology, American Heart Association, and nine other groups, which recommended a target of 130/80 mm Hg for pretty much everyone. Though they say greater than 140/90 mm Hg should be the threshold for low-risk patients or for starting therapy post stroke, we often forget those nuances. The main point of contention is that the AAFP guideline was looking for a mortality benefit, whereas the ACC/AHA/everyone else guideline was looking at preventing cardiovascular events. The latter guideline was driven mainly by the results of the SPRINT trial. ACC/AHA argue for more aggressive targets to prevent the things that cardiologists care about, namely heart attacks.
The AAFP guideline conceded that more aggressive control will result in fewer myocardial infarctions but warn that it comes with more adverse events. Treating 1,000 patients to this lower target would theoretically prevent four MIs, possibly prevent three strokes, but result in 30 adverse events.
In the end, what we are seeing here is not so much a debate over the evidence as a debate over priorities. Interventions that don’t improve mortality can be questioned in terms of their cost effectiveness. But you probably don’t want to have a heart attack (even a nonfatal one). And you certainly don’t want to have a stroke. However, lower blood pressure targets inevitably require more medications. Notwithstanding the economic costs, the dangers of polypharmacy, medication interactions, side effects, and syncope leading to falls cannot be ignored. Falls are not benign adverse events, especially in older adults.
The counter argument is that physicians are human and often let things slide. Set the target at 140/90 mm Hg, and many physicians won’t jump on a systolic blood pressure of 144 mm Hg. Set the target at 130 mm Hg, and maybe they’ll be more likely to react. There’s a fine line between permissiveness and complacency.
If you zoom out and look at the multitude of blood pressure guidelines, you start to notice an important fact. There is not much daylight between them. There are subtle differences in what constitutes high risk and different definitions of older (older should be defined as 10 years older than the reader’s current age). But otherwise, the blood pressure targets are not that different.
Does that final 10 mm Hg really matter when barriers to care mean that tens of millions in the United States are unaware they have hypertension? Even among those diagnosed, many are either untreated or inadequately treated.
With this context, perhaps the most insightful thing that can be said about the blood pressure guideline controversy is that it’s not all that controversial. We can likely all agree that we need to be better at treating hypertension and that creative solutions to reach underserved communities are necessary.
Arguing about 140/90 mm Hg or 130/80 mm Hg is less important than acknowledging that we should be aggressive in screening for and treating hypertension. We should acknowledge that beyond a certain point any cardiovascular benefit comes at the cost of hypotension and side effects. That tipping point will be different for different groups, and probably at a higher set point in older patients.
Individualizing care isn’t difficult. We do it all the time. We just shouldn’t be letting people walk around with untreated hypertension. It’s not the 1900s anymore.
Dr. Labos is a cardiologist at Hôpital Notre-Dame, Montreal. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Antidrug antibody effects compared across RA biologics
TOPLINE:
In patients with rheumatoid arthritis, the presence of antidrug antibodies was associated with a diminished response to biologic disease-modifying antirheumatic drugs in a prospective cohort study.
METHODOLOGY:
- Researchers prospectively analyzed data from 230 patients (mean age, 54.3 years; 77.0% women) with RA diagnosis recruited from March 3, 2014, to June 21, 2016.
- All were initiating new treatment with an anti–tumor necrosis factor (TNF) monoclonal antibody (mAb; either infliximab or adalimumab), etanercept, tocilizumab, or rituximab, according to the choice of the treating physician.
- The primary outcome was the association of antidrug antibody positivity with European Alliance of Associations for Rheumatology (EULAR) response to treatment at month 12, assessed through univariate logistic regression.
TAKEAWAY:
- At month 12, antidrug antibody positivity was 38.2% in patients who were treated with anti-TNF mAbs, 6.1% with etanercept, 50.0% with rituximab, and 20.0% with tocilizumab.
- There was an inverse association between antidrug antibody positivity directed against all biologic drugs and EULAR response at month 12 (odds ratio, 0.19; 95% confidence interval, 0.09-0.38; P < .001).
- In the multivariable analysis, antidrug antibodies, body mass index, and rheumatoid factor were independently and inversely associated with response to treatment.
- There was a significantly higher drug concentration of anti-TNF mAbs in patients with antidrug antibody–negative vs. antidrug antibody–positive status (mean difference, –9.6 mg/L; 95% CI, –12.4 to –6.9; P < .001).
IN PRACTICE:
Findings of this study suggest that antidrug antibodies are associated with nonresponse to biologic drugs and can be monitored in the management of patients with RA, particularly nonresponders.
SOURCE:
Samuel Bitouin, MD, PhD, of the rheumatology department at Paris-Saclay University, and coauthors in the ABIRISK (Anti-Biopharmaceutical Immunization: Prediction and Analysis of Clinical Relevance to Minimize the Risk) consortium reported the study in JAMA Network Open. The work was funded by a grant from the European Union Innovative Medicines Initiative.
LIMITATIONS:
Though the study demonstrated an association when all biologic drugs were analyzed together, it was not powered to demonstrate an association for each drug class.
DISCLOSURES:
Many authors reported financial relationships with pharmaceutical companies.
A version of this article first appeared on Medscape.com.
TOPLINE:
In patients with rheumatoid arthritis, the presence of antidrug antibodies was associated with a diminished response to biologic disease-modifying antirheumatic drugs in a prospective cohort study.
METHODOLOGY:
- Researchers prospectively analyzed data from 230 patients (mean age, 54.3 years; 77.0% women) with RA diagnosis recruited from March 3, 2014, to June 21, 2016.
- All were initiating new treatment with an anti–tumor necrosis factor (TNF) monoclonal antibody (mAb; either infliximab or adalimumab), etanercept, tocilizumab, or rituximab, according to the choice of the treating physician.
- The primary outcome was the association of antidrug antibody positivity with European Alliance of Associations for Rheumatology (EULAR) response to treatment at month 12, assessed through univariate logistic regression.
TAKEAWAY:
- At month 12, antidrug antibody positivity was 38.2% in patients who were treated with anti-TNF mAbs, 6.1% with etanercept, 50.0% with rituximab, and 20.0% with tocilizumab.
- There was an inverse association between antidrug antibody positivity directed against all biologic drugs and EULAR response at month 12 (odds ratio, 0.19; 95% confidence interval, 0.09-0.38; P < .001).
- In the multivariable analysis, antidrug antibodies, body mass index, and rheumatoid factor were independently and inversely associated with response to treatment.
- There was a significantly higher drug concentration of anti-TNF mAbs in patients with antidrug antibody–negative vs. antidrug antibody–positive status (mean difference, –9.6 mg/L; 95% CI, –12.4 to –6.9; P < .001).
IN PRACTICE:
Findings of this study suggest that antidrug antibodies are associated with nonresponse to biologic drugs and can be monitored in the management of patients with RA, particularly nonresponders.
SOURCE:
Samuel Bitouin, MD, PhD, of the rheumatology department at Paris-Saclay University, and coauthors in the ABIRISK (Anti-Biopharmaceutical Immunization: Prediction and Analysis of Clinical Relevance to Minimize the Risk) consortium reported the study in JAMA Network Open. The work was funded by a grant from the European Union Innovative Medicines Initiative.
LIMITATIONS:
Though the study demonstrated an association when all biologic drugs were analyzed together, it was not powered to demonstrate an association for each drug class.
DISCLOSURES:
Many authors reported financial relationships with pharmaceutical companies.
A version of this article first appeared on Medscape.com.
TOPLINE:
In patients with rheumatoid arthritis, the presence of antidrug antibodies was associated with a diminished response to biologic disease-modifying antirheumatic drugs in a prospective cohort study.
METHODOLOGY:
- Researchers prospectively analyzed data from 230 patients (mean age, 54.3 years; 77.0% women) with RA diagnosis recruited from March 3, 2014, to June 21, 2016.
- All were initiating new treatment with an anti–tumor necrosis factor (TNF) monoclonal antibody (mAb; either infliximab or adalimumab), etanercept, tocilizumab, or rituximab, according to the choice of the treating physician.
- The primary outcome was the association of antidrug antibody positivity with European Alliance of Associations for Rheumatology (EULAR) response to treatment at month 12, assessed through univariate logistic regression.
TAKEAWAY:
- At month 12, antidrug antibody positivity was 38.2% in patients who were treated with anti-TNF mAbs, 6.1% with etanercept, 50.0% with rituximab, and 20.0% with tocilizumab.
- There was an inverse association between antidrug antibody positivity directed against all biologic drugs and EULAR response at month 12 (odds ratio, 0.19; 95% confidence interval, 0.09-0.38; P < .001).
- In the multivariable analysis, antidrug antibodies, body mass index, and rheumatoid factor were independently and inversely associated with response to treatment.
- There was a significantly higher drug concentration of anti-TNF mAbs in patients with antidrug antibody–negative vs. antidrug antibody–positive status (mean difference, –9.6 mg/L; 95% CI, –12.4 to –6.9; P < .001).
IN PRACTICE:
Findings of this study suggest that antidrug antibodies are associated with nonresponse to biologic drugs and can be monitored in the management of patients with RA, particularly nonresponders.
SOURCE:
Samuel Bitouin, MD, PhD, of the rheumatology department at Paris-Saclay University, and coauthors in the ABIRISK (Anti-Biopharmaceutical Immunization: Prediction and Analysis of Clinical Relevance to Minimize the Risk) consortium reported the study in JAMA Network Open. The work was funded by a grant from the European Union Innovative Medicines Initiative.
LIMITATIONS:
Though the study demonstrated an association when all biologic drugs were analyzed together, it was not powered to demonstrate an association for each drug class.
DISCLOSURES:
Many authors reported financial relationships with pharmaceutical companies.
A version of this article first appeared on Medscape.com.
Can berberine live up to the claim that it’s ‘nature’s Ozempic’?
Berberine, a plant-derived compound historically used in traditional Chinese medicine, is experiencing increased popularity thanks to social media, especially TikTok, where the hashtag #berberine has more than 75 million views at the time of this writing.
Off-the-shelf berberine comes as a yellow-orange powder usually encased in a capsule or mixed into tablet form. It’s extracted from the roots, stems, and leaves of various plants, including goldenseal and barberry.
Its use is additionally promoted for insulin resistance, polycystic ovary syndrome, and even cancer, but medical experts are warning potential users that it lacks robust evidence to support its use.
“There’s not that much data on it,” says Reshmi Srinath, MD, director of the Mount Sinai weight and metabolism management program, New York. “It’s sort of shocking now that it’s popped up into the media, to be frank.”
In response to berberine’s online popularity, the National Center for Complementary and Integrative Health issued a warning, stating that “there isn’t enough rigorous scientific evidence to determine whether it is effective.”
Overstated claims, lack of scientific research?
Other endocrinologists and weight management experts agree. “The claims are pretty overstated when it comes to the impact on weight loss, based on the evidence in the literature that’s currently available,” says Jaime Almandoz, MD, medical director of the UT Southwestern Medical Center, Dallas, weight wellness program.
A review of 12 randomized controlled trials evaluating berberine’s effects on obesity concluded that the treatment moderately decreased body weight. The trials included were conducted over only a few months and had small numbers of participants, and weight loss was not the primary outcome measure.
“There are few randomized controlled trials,” says Ivania Rizo, MD, an endocrinologist at Boston University. “It appears that they all have some low quality of methods which essentially can lead to an increased risk of bias.”
Another review, of 35 studies – most of them on animals and human cells and similarly underpowered – concluded that berberine showed promise for reducing blood glucose. A separate study found that berberine treatment actually increased the body weight and appetite of rats.
How exactly berberine elicits these effects is not entirely clear. Several studies point to its activation of AMP-activated protein kinase, which improves glucose tolerance in rats, as the mechanism for weight loss. Metformin, a drug used to improve glycemic control in people with type 2 diabetes, works in a similar way. Other researchers have hypothesized a link between berberine and the gut microbiome to explain its effect on type 2 diabetes and weight loss, though the clinical data to substantiate this link are shaky.
“I caution my patients about dietary supplements for weight management because we do not have high-quality data demonstrating efficacy,” Katherine Saunders, MD, DABOM, an obesity expert and cofounder of Intellihealth, a platform for obesity management, said in an email.
Experimenting with berberine
Despite the lack of substantial evidence supporting berberine’s use for weight management and obesity, interest in the supplement seems to be increasing. One reason could be that lifestyle interventions aren’t sufficient for most people with obesity to lose a significant amount of weight, with many requiring medical intervention, according to Dr. Saunders.
But access to treatment providers is limited. “As a result, it is not uncommon for individuals with obesity to experiment with dietary supplements like berberine,” she observed.
Dr. Srinath, the Mount Sinai doctor, says many patients have asked for her thoughts on berberine as a weight loss supplement. “I say, you know, it’s something you’re welcome to try, but we don’t have enough data at this time to recommend it.”
The hype surrounding the supplement isn’t all that surprising. About 42% of adults in the United States have obesity, according to 2019-2023 National Health and Nutrition Examination Survey data, pointing to a serious need for accessible drugs to address the condition. Berberine is available over the counter and is far cheaper than most of the newer U.S. Food and Drug Administration–approved drugs for weight loss.
Wegovy, semaglutide approved to treat obesity, can cost as much as $1,300 per package; and Ozempic, semaglutide approved to treat type 2 diabetes, can cost more than $1,000 per month. “That’s a very steep price to pay,” says Dr. Srinath.
Many insurance companies won’t cover the drugs, curbing access to Americans who need them, says Dr. Almandoz. Federally sponsored programs such as Medicare and Medicaid also don’t cover the drugs, which are approved for obesity and weight management. “That’s been a huge hole in our health care system,” says Dr. Srinath. “That’s sort of what’s been driving interest in supplements and things like that.”
Among adults trying to lose weight, only about 3% said they took prescription medication for weight loss, according to a report from the U.S. Government Accountability Office. This report includes 2013-2016 data, predating Wegovy’s approval for chronic weight management.
“These classes are notorious for being quite pricey and not well covered by insurance,” says Dr. Almandoz. “It’s easy to see why someone would promote something that someone may have more access to.”
Comparing Ozempic or Wegovy with berberine can be misleading. Those drugs work by mimicking the effect of the hormone GLP-1 to help reduce appetite.
A clinical trial assessing the efficacy of semaglutide found that adults with obesity who took the drug for 68 weeks lost approximately 15% of their body weight in combination with lifestyle changes. The FDA approval was based on this trial and three others that showed similarly substantial reductions in weight.
The trials also document the many side effects of taking the drugs, primarily gastrointestinal in nature. The short- and long-term effects of berberine, on the other hand, are less clear. Some of the clinical trials reported diarrhea and stomach upset as the most common adverse effects.
Its perception as a naturally derived option for weight loss, though, might encourage people to overlook the potential interactions that berberine could have with other drugs, according to Dr. Almandoz.
He says clinicians considering natural products or nutraceuticals for patients should check for potential side effects and find reliable database sources to determine any potential medication interactions for patients. But the unregulated nature of berberine makes this challenging, Dr. Almandoz adds.
The dosage, formulations, and quality of berberine vary in each study and each product because supplements don’t need to pass through the checks and balances of the FDA to land on shelves.
The lack of regulation could incentivize some companies to add stimulants to enhance any weight loss effect that the supplement may have. Those additives might interact with other health conditions or cause side effects like anxiety, says Dr. Almandoz.
Berberine should also not be taken during pregnancy or while breastfeeding, and it is unsafe for young children; in newborns and children, the supplement can cause higher levels of bilirubin in the blood, worsening any jaundice at birth and posing a greater risk for kernicterus.
Dr. Rizo urges patients, before they ask for berberine, to first ask for safe and effective interventions they can access. “I don’t want to have people not use effective interventions that are currently available to them, and instead use something that needs to be better studied and needs to be better regulated,” she says.
While the “nature’s Ozempic” catchphrase could be drawing in potential users with its dubious comparison, berberine’s escalating popularity might also be a symptom of people seeking a quick fix, the experts worry.
“That’s my fear,” says Dr. Srinath. “ ‘Let me get this medicine, let me lose the weight fast,’ but at the end of the day, weight management is a long-term journey. It takes time, it takes effort, it is not easy, and there is no quick fix.”
This is another concern for doctors; for people who’ve struggled with losing weight for years, not seeing results from berberine could feel like another failure.
“It will give them another opportunity to feel like they are being unsuccessful or that they are failing at weight loss again,” says Dr. Almandoz. “It feeds into the hopelessness that many people with obesity have around their weight management.”
A version of this article first appeared on Medscape.com.
Berberine, a plant-derived compound historically used in traditional Chinese medicine, is experiencing increased popularity thanks to social media, especially TikTok, where the hashtag #berberine has more than 75 million views at the time of this writing.
Off-the-shelf berberine comes as a yellow-orange powder usually encased in a capsule or mixed into tablet form. It’s extracted from the roots, stems, and leaves of various plants, including goldenseal and barberry.
Its use is additionally promoted for insulin resistance, polycystic ovary syndrome, and even cancer, but medical experts are warning potential users that it lacks robust evidence to support its use.
“There’s not that much data on it,” says Reshmi Srinath, MD, director of the Mount Sinai weight and metabolism management program, New York. “It’s sort of shocking now that it’s popped up into the media, to be frank.”
In response to berberine’s online popularity, the National Center for Complementary and Integrative Health issued a warning, stating that “there isn’t enough rigorous scientific evidence to determine whether it is effective.”
Overstated claims, lack of scientific research?
Other endocrinologists and weight management experts agree. “The claims are pretty overstated when it comes to the impact on weight loss, based on the evidence in the literature that’s currently available,” says Jaime Almandoz, MD, medical director of the UT Southwestern Medical Center, Dallas, weight wellness program.
A review of 12 randomized controlled trials evaluating berberine’s effects on obesity concluded that the treatment moderately decreased body weight. The trials included were conducted over only a few months and had small numbers of participants, and weight loss was not the primary outcome measure.
“There are few randomized controlled trials,” says Ivania Rizo, MD, an endocrinologist at Boston University. “It appears that they all have some low quality of methods which essentially can lead to an increased risk of bias.”
Another review, of 35 studies – most of them on animals and human cells and similarly underpowered – concluded that berberine showed promise for reducing blood glucose. A separate study found that berberine treatment actually increased the body weight and appetite of rats.
How exactly berberine elicits these effects is not entirely clear. Several studies point to its activation of AMP-activated protein kinase, which improves glucose tolerance in rats, as the mechanism for weight loss. Metformin, a drug used to improve glycemic control in people with type 2 diabetes, works in a similar way. Other researchers have hypothesized a link between berberine and the gut microbiome to explain its effect on type 2 diabetes and weight loss, though the clinical data to substantiate this link are shaky.
“I caution my patients about dietary supplements for weight management because we do not have high-quality data demonstrating efficacy,” Katherine Saunders, MD, DABOM, an obesity expert and cofounder of Intellihealth, a platform for obesity management, said in an email.
Experimenting with berberine
Despite the lack of substantial evidence supporting berberine’s use for weight management and obesity, interest in the supplement seems to be increasing. One reason could be that lifestyle interventions aren’t sufficient for most people with obesity to lose a significant amount of weight, with many requiring medical intervention, according to Dr. Saunders.
But access to treatment providers is limited. “As a result, it is not uncommon for individuals with obesity to experiment with dietary supplements like berberine,” she observed.
Dr. Srinath, the Mount Sinai doctor, says many patients have asked for her thoughts on berberine as a weight loss supplement. “I say, you know, it’s something you’re welcome to try, but we don’t have enough data at this time to recommend it.”
The hype surrounding the supplement isn’t all that surprising. About 42% of adults in the United States have obesity, according to 2019-2023 National Health and Nutrition Examination Survey data, pointing to a serious need for accessible drugs to address the condition. Berberine is available over the counter and is far cheaper than most of the newer U.S. Food and Drug Administration–approved drugs for weight loss.
Wegovy, semaglutide approved to treat obesity, can cost as much as $1,300 per package; and Ozempic, semaglutide approved to treat type 2 diabetes, can cost more than $1,000 per month. “That’s a very steep price to pay,” says Dr. Srinath.
Many insurance companies won’t cover the drugs, curbing access to Americans who need them, says Dr. Almandoz. Federally sponsored programs such as Medicare and Medicaid also don’t cover the drugs, which are approved for obesity and weight management. “That’s been a huge hole in our health care system,” says Dr. Srinath. “That’s sort of what’s been driving interest in supplements and things like that.”
Among adults trying to lose weight, only about 3% said they took prescription medication for weight loss, according to a report from the U.S. Government Accountability Office. This report includes 2013-2016 data, predating Wegovy’s approval for chronic weight management.
“These classes are notorious for being quite pricey and not well covered by insurance,” says Dr. Almandoz. “It’s easy to see why someone would promote something that someone may have more access to.”
Comparing Ozempic or Wegovy with berberine can be misleading. Those drugs work by mimicking the effect of the hormone GLP-1 to help reduce appetite.
A clinical trial assessing the efficacy of semaglutide found that adults with obesity who took the drug for 68 weeks lost approximately 15% of their body weight in combination with lifestyle changes. The FDA approval was based on this trial and three others that showed similarly substantial reductions in weight.
The trials also document the many side effects of taking the drugs, primarily gastrointestinal in nature. The short- and long-term effects of berberine, on the other hand, are less clear. Some of the clinical trials reported diarrhea and stomach upset as the most common adverse effects.
Its perception as a naturally derived option for weight loss, though, might encourage people to overlook the potential interactions that berberine could have with other drugs, according to Dr. Almandoz.
He says clinicians considering natural products or nutraceuticals for patients should check for potential side effects and find reliable database sources to determine any potential medication interactions for patients. But the unregulated nature of berberine makes this challenging, Dr. Almandoz adds.
The dosage, formulations, and quality of berberine vary in each study and each product because supplements don’t need to pass through the checks and balances of the FDA to land on shelves.
The lack of regulation could incentivize some companies to add stimulants to enhance any weight loss effect that the supplement may have. Those additives might interact with other health conditions or cause side effects like anxiety, says Dr. Almandoz.
Berberine should also not be taken during pregnancy or while breastfeeding, and it is unsafe for young children; in newborns and children, the supplement can cause higher levels of bilirubin in the blood, worsening any jaundice at birth and posing a greater risk for kernicterus.
Dr. Rizo urges patients, before they ask for berberine, to first ask for safe and effective interventions they can access. “I don’t want to have people not use effective interventions that are currently available to them, and instead use something that needs to be better studied and needs to be better regulated,” she says.
While the “nature’s Ozempic” catchphrase could be drawing in potential users with its dubious comparison, berberine’s escalating popularity might also be a symptom of people seeking a quick fix, the experts worry.
“That’s my fear,” says Dr. Srinath. “ ‘Let me get this medicine, let me lose the weight fast,’ but at the end of the day, weight management is a long-term journey. It takes time, it takes effort, it is not easy, and there is no quick fix.”
This is another concern for doctors; for people who’ve struggled with losing weight for years, not seeing results from berberine could feel like another failure.
“It will give them another opportunity to feel like they are being unsuccessful or that they are failing at weight loss again,” says Dr. Almandoz. “It feeds into the hopelessness that many people with obesity have around their weight management.”
A version of this article first appeared on Medscape.com.
Berberine, a plant-derived compound historically used in traditional Chinese medicine, is experiencing increased popularity thanks to social media, especially TikTok, where the hashtag #berberine has more than 75 million views at the time of this writing.
Off-the-shelf berberine comes as a yellow-orange powder usually encased in a capsule or mixed into tablet form. It’s extracted from the roots, stems, and leaves of various plants, including goldenseal and barberry.
Its use is additionally promoted for insulin resistance, polycystic ovary syndrome, and even cancer, but medical experts are warning potential users that it lacks robust evidence to support its use.
“There’s not that much data on it,” says Reshmi Srinath, MD, director of the Mount Sinai weight and metabolism management program, New York. “It’s sort of shocking now that it’s popped up into the media, to be frank.”
In response to berberine’s online popularity, the National Center for Complementary and Integrative Health issued a warning, stating that “there isn’t enough rigorous scientific evidence to determine whether it is effective.”
Overstated claims, lack of scientific research?
Other endocrinologists and weight management experts agree. “The claims are pretty overstated when it comes to the impact on weight loss, based on the evidence in the literature that’s currently available,” says Jaime Almandoz, MD, medical director of the UT Southwestern Medical Center, Dallas, weight wellness program.
A review of 12 randomized controlled trials evaluating berberine’s effects on obesity concluded that the treatment moderately decreased body weight. The trials included were conducted over only a few months and had small numbers of participants, and weight loss was not the primary outcome measure.
“There are few randomized controlled trials,” says Ivania Rizo, MD, an endocrinologist at Boston University. “It appears that they all have some low quality of methods which essentially can lead to an increased risk of bias.”
Another review, of 35 studies – most of them on animals and human cells and similarly underpowered – concluded that berberine showed promise for reducing blood glucose. A separate study found that berberine treatment actually increased the body weight and appetite of rats.
How exactly berberine elicits these effects is not entirely clear. Several studies point to its activation of AMP-activated protein kinase, which improves glucose tolerance in rats, as the mechanism for weight loss. Metformin, a drug used to improve glycemic control in people with type 2 diabetes, works in a similar way. Other researchers have hypothesized a link between berberine and the gut microbiome to explain its effect on type 2 diabetes and weight loss, though the clinical data to substantiate this link are shaky.
“I caution my patients about dietary supplements for weight management because we do not have high-quality data demonstrating efficacy,” Katherine Saunders, MD, DABOM, an obesity expert and cofounder of Intellihealth, a platform for obesity management, said in an email.
Experimenting with berberine
Despite the lack of substantial evidence supporting berberine’s use for weight management and obesity, interest in the supplement seems to be increasing. One reason could be that lifestyle interventions aren’t sufficient for most people with obesity to lose a significant amount of weight, with many requiring medical intervention, according to Dr. Saunders.
But access to treatment providers is limited. “As a result, it is not uncommon for individuals with obesity to experiment with dietary supplements like berberine,” she observed.
Dr. Srinath, the Mount Sinai doctor, says many patients have asked for her thoughts on berberine as a weight loss supplement. “I say, you know, it’s something you’re welcome to try, but we don’t have enough data at this time to recommend it.”
The hype surrounding the supplement isn’t all that surprising. About 42% of adults in the United States have obesity, according to 2019-2023 National Health and Nutrition Examination Survey data, pointing to a serious need for accessible drugs to address the condition. Berberine is available over the counter and is far cheaper than most of the newer U.S. Food and Drug Administration–approved drugs for weight loss.
Wegovy, semaglutide approved to treat obesity, can cost as much as $1,300 per package; and Ozempic, semaglutide approved to treat type 2 diabetes, can cost more than $1,000 per month. “That’s a very steep price to pay,” says Dr. Srinath.
Many insurance companies won’t cover the drugs, curbing access to Americans who need them, says Dr. Almandoz. Federally sponsored programs such as Medicare and Medicaid also don’t cover the drugs, which are approved for obesity and weight management. “That’s been a huge hole in our health care system,” says Dr. Srinath. “That’s sort of what’s been driving interest in supplements and things like that.”
Among adults trying to lose weight, only about 3% said they took prescription medication for weight loss, according to a report from the U.S. Government Accountability Office. This report includes 2013-2016 data, predating Wegovy’s approval for chronic weight management.
“These classes are notorious for being quite pricey and not well covered by insurance,” says Dr. Almandoz. “It’s easy to see why someone would promote something that someone may have more access to.”
Comparing Ozempic or Wegovy with berberine can be misleading. Those drugs work by mimicking the effect of the hormone GLP-1 to help reduce appetite.
A clinical trial assessing the efficacy of semaglutide found that adults with obesity who took the drug for 68 weeks lost approximately 15% of their body weight in combination with lifestyle changes. The FDA approval was based on this trial and three others that showed similarly substantial reductions in weight.
The trials also document the many side effects of taking the drugs, primarily gastrointestinal in nature. The short- and long-term effects of berberine, on the other hand, are less clear. Some of the clinical trials reported diarrhea and stomach upset as the most common adverse effects.
Its perception as a naturally derived option for weight loss, though, might encourage people to overlook the potential interactions that berberine could have with other drugs, according to Dr. Almandoz.
He says clinicians considering natural products or nutraceuticals for patients should check for potential side effects and find reliable database sources to determine any potential medication interactions for patients. But the unregulated nature of berberine makes this challenging, Dr. Almandoz adds.
The dosage, formulations, and quality of berberine vary in each study and each product because supplements don’t need to pass through the checks and balances of the FDA to land on shelves.
The lack of regulation could incentivize some companies to add stimulants to enhance any weight loss effect that the supplement may have. Those additives might interact with other health conditions or cause side effects like anxiety, says Dr. Almandoz.
Berberine should also not be taken during pregnancy or while breastfeeding, and it is unsafe for young children; in newborns and children, the supplement can cause higher levels of bilirubin in the blood, worsening any jaundice at birth and posing a greater risk for kernicterus.
Dr. Rizo urges patients, before they ask for berberine, to first ask for safe and effective interventions they can access. “I don’t want to have people not use effective interventions that are currently available to them, and instead use something that needs to be better studied and needs to be better regulated,” she says.
While the “nature’s Ozempic” catchphrase could be drawing in potential users with its dubious comparison, berberine’s escalating popularity might also be a symptom of people seeking a quick fix, the experts worry.
“That’s my fear,” says Dr. Srinath. “ ‘Let me get this medicine, let me lose the weight fast,’ but at the end of the day, weight management is a long-term journey. It takes time, it takes effort, it is not easy, and there is no quick fix.”
This is another concern for doctors; for people who’ve struggled with losing weight for years, not seeing results from berberine could feel like another failure.
“It will give them another opportunity to feel like they are being unsuccessful or that they are failing at weight loss again,” says Dr. Almandoz. “It feeds into the hopelessness that many people with obesity have around their weight management.”
A version of this article first appeared on Medscape.com.
New German guidelines change the paradigm for asthma
Asthma has long been associated with the use of inhalers to control symptoms. The new S2K guideline on the management of asthma, compiled by experts and published in March 2023, aims to change this. “For decades, we have known about medication that can be used to put asthma into remission. The patient can go out or travel on vacation without an inhaler. This is possible. This is a symptom-prevention approach,” said the guideline coordinator Marek Lommatzsch, MD, PhD, head senior physician of the pulmonology department at the University Medicine Rostock, Germany, in an interview.
The guideline was created by the German Respiratory Society, and a further 11 professional societies from Germany and Austria were involved in the update. The authors comprehensively revised the guideline from 2017, and the evidence-based national disease management guideline (NVL) for general asthma care from 2020 was amended.
Erika von Mutius, MD, PhD, pediatrician and professor of pediatric allergology and pulmonology at the Dr. Von Hauner Children’s Hospital, Munich, and director of the Institute of Asthma and Allergy Prevention at Helmholtz Munich, was not directly involved in the guideline. She said,
Anti-inflammatory therapy
The significance of anti-inflammatory therapy was stressed in the NVL from 2020. The new guideline holds that anti-inflammatory therapy should be considered the primary therapeutic option. “We are making a U-turn: only treat the respiratory inflammation. Salbutamol should still only be given in exceptional cases as required,” according to Dr. Lommatzsch.
In the guideline, asthma therapy is described using an updated step-by-step plan. Inhaled glucocorticoids (ICS) represent the most important pillar of therapy. ICS can be used as permanent therapy or as as-needed therapy in fixed combination with formoterol, which rapidly dilates the airways.
Allergen immunotherapy, also known as hyposensitization, and biologics are also effective anti-inflammatory treatments, Dr. Lommatzsch added. “We must ensure that these anti-inflammatory medicines are also used effectively. Mild to moderate forms of asthma can be treated easily by a primary care physician,” he said. Basic diagnostics in the form of a blood sample are required. A somewhat more comprehensive medical history is also needed. “It takes a little more time and involves more than just taking the inhaler out of the cupboard.”
The situation regarding children, however, is a little different with regard to anti-inflammatory therapy, Dr. Von Mutius explained. “Childhood asthma has many forms, and confirming the diagnosis is not always straightforward, especially in infancy. If needed, salbutamol can be prescribed. However, the anti-inflammatory medication should usually also be administered.”
She emphasized that the guideline has been designed in a sophisticated way that offers the option of “using medical experience to see what is suitable for this family or better for this patient. This is still always subject to medical judgment and responsibility. I find this really successful.”
Diagnostics using biomarkers
The previous guideline concentrated on measuring lung function as a way of diagnosing asthmatic illness. Three biomarkers were brought to the fore:
- Eosinophils in the blood.
- IgE levels.
- The FeNO test (proportion of nitrogen monoxide in exhaled air).
Slightly amended, the guideline now states that the FeNO test is implemented as “an integral component of specialist diagnosis.”
The test measures the nitrogen monoxide content of exhaled air as an indicator of inflammation in the airways. However, this test must often be paid for by the patient. “In this respect, we want to give a nudge in the direction of the political decision-makers,” emphasized Dr. Lommatzsch.
Dr. Von Mutius added that use of the FeNO test has not been established in many practices and outpatient clinics. The inflammatory marker is also subject to fluctuations. “This is an update to the guideline where we must wait to see the political response.”
Which biologic?
Despite treatment with the established therapies, the symptoms of asthma can persist in some people with severe forms of the condition. Biologics are highly effective for these patients and are preferable in the last stage of therapy to long-term therapy with oral steroids, which have numerous side effects. The current guideline provides an overview diagram to help decide which biologic is suitable for which patient.
“There are six biologics that can be used to treat severe asthma. Officially, almost any biologic can be taken into consideration for a patient, since the approvals overlap. Nevertheless, we know that certain patients benefit hugely from certain biologics. A targeted choice should therefore be made,” explained Dr. Lommatzsch.
Biologics were mentioned in the 2020 NVL but not to the great extent that they are in the latest version. “For the first time, we have created an overview diagram for the individual choice of biologic. With it, we have now set a standard,” said Dr. Lommatzsch.
Therapy with biologics has brought about rapid progress for adults. Dr. Von Mutius anticipates challenges in approving such therapeutics for pediatric treatment. “As is often the case, these therapies are not approved for young children. Meanwhile, dupilumab is approved for children aged 6 months and older; unfortunately, the indication for this is actually atopic dermatitis,” she explains.
When using this therapy for pediatric patients, it is therefore important to explain the options to parents and to inform them of side effects. Severe forms of asthma are rare in children; they are uncommon in adults but are more prevalent than in children.
Children and adolescents
One new chapter in the guideline describes giving medical advice to adolescents choosing a career. A table has been compiled that contains information regarding jobs and their respective allergy and asthma risk. The table is designed to be displayed in a medical practice.
Another chapter characterizes the interrelation between asthma and mental health. It differentiates between psychiatric comorbidities for which the patient requires professional help and the stress caused by the asthmatic illness itself. Many patients do not have a mental illness but do suffer under the everyday strain of having asthma, said Dr. Lommatzsch. Therefore, it is important to educate patients and their relatives on how to make a strength out of this supposed weakness – the asthmatic illness. “We have established a procedure for this and have summarized its key points in the guideline,” said Dr. Lommatzsch.
Other updates to the guideline cover asthma in different contexts, such as in pregnant women. The updates address adrenal insufficiency as a side effect of the use of steroids over many years. In addition, the guideline contains a chapter on digital apps that can help with diagnostics and medical history.
Dr. Lommatzsch highlighted a new tool. “By using 15 key points summarized in a table, the guideline displays the essential differences between COPD [chronic obstructive pulmonary disease] and asthma in terms of the symptoms and the findings. It is the most modern table available in Germany that differentiates between the two diseases.”
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
Asthma has long been associated with the use of inhalers to control symptoms. The new S2K guideline on the management of asthma, compiled by experts and published in March 2023, aims to change this. “For decades, we have known about medication that can be used to put asthma into remission. The patient can go out or travel on vacation without an inhaler. This is possible. This is a symptom-prevention approach,” said the guideline coordinator Marek Lommatzsch, MD, PhD, head senior physician of the pulmonology department at the University Medicine Rostock, Germany, in an interview.
The guideline was created by the German Respiratory Society, and a further 11 professional societies from Germany and Austria were involved in the update. The authors comprehensively revised the guideline from 2017, and the evidence-based national disease management guideline (NVL) for general asthma care from 2020 was amended.
Erika von Mutius, MD, PhD, pediatrician and professor of pediatric allergology and pulmonology at the Dr. Von Hauner Children’s Hospital, Munich, and director of the Institute of Asthma and Allergy Prevention at Helmholtz Munich, was not directly involved in the guideline. She said,
Anti-inflammatory therapy
The significance of anti-inflammatory therapy was stressed in the NVL from 2020. The new guideline holds that anti-inflammatory therapy should be considered the primary therapeutic option. “We are making a U-turn: only treat the respiratory inflammation. Salbutamol should still only be given in exceptional cases as required,” according to Dr. Lommatzsch.
In the guideline, asthma therapy is described using an updated step-by-step plan. Inhaled glucocorticoids (ICS) represent the most important pillar of therapy. ICS can be used as permanent therapy or as as-needed therapy in fixed combination with formoterol, which rapidly dilates the airways.
Allergen immunotherapy, also known as hyposensitization, and biologics are also effective anti-inflammatory treatments, Dr. Lommatzsch added. “We must ensure that these anti-inflammatory medicines are also used effectively. Mild to moderate forms of asthma can be treated easily by a primary care physician,” he said. Basic diagnostics in the form of a blood sample are required. A somewhat more comprehensive medical history is also needed. “It takes a little more time and involves more than just taking the inhaler out of the cupboard.”
The situation regarding children, however, is a little different with regard to anti-inflammatory therapy, Dr. Von Mutius explained. “Childhood asthma has many forms, and confirming the diagnosis is not always straightforward, especially in infancy. If needed, salbutamol can be prescribed. However, the anti-inflammatory medication should usually also be administered.”
She emphasized that the guideline has been designed in a sophisticated way that offers the option of “using medical experience to see what is suitable for this family or better for this patient. This is still always subject to medical judgment and responsibility. I find this really successful.”
Diagnostics using biomarkers
The previous guideline concentrated on measuring lung function as a way of diagnosing asthmatic illness. Three biomarkers were brought to the fore:
- Eosinophils in the blood.
- IgE levels.
- The FeNO test (proportion of nitrogen monoxide in exhaled air).
Slightly amended, the guideline now states that the FeNO test is implemented as “an integral component of specialist diagnosis.”
The test measures the nitrogen monoxide content of exhaled air as an indicator of inflammation in the airways. However, this test must often be paid for by the patient. “In this respect, we want to give a nudge in the direction of the political decision-makers,” emphasized Dr. Lommatzsch.
Dr. Von Mutius added that use of the FeNO test has not been established in many practices and outpatient clinics. The inflammatory marker is also subject to fluctuations. “This is an update to the guideline where we must wait to see the political response.”
Which biologic?
Despite treatment with the established therapies, the symptoms of asthma can persist in some people with severe forms of the condition. Biologics are highly effective for these patients and are preferable in the last stage of therapy to long-term therapy with oral steroids, which have numerous side effects. The current guideline provides an overview diagram to help decide which biologic is suitable for which patient.
“There are six biologics that can be used to treat severe asthma. Officially, almost any biologic can be taken into consideration for a patient, since the approvals overlap. Nevertheless, we know that certain patients benefit hugely from certain biologics. A targeted choice should therefore be made,” explained Dr. Lommatzsch.
Biologics were mentioned in the 2020 NVL but not to the great extent that they are in the latest version. “For the first time, we have created an overview diagram for the individual choice of biologic. With it, we have now set a standard,” said Dr. Lommatzsch.
Therapy with biologics has brought about rapid progress for adults. Dr. Von Mutius anticipates challenges in approving such therapeutics for pediatric treatment. “As is often the case, these therapies are not approved for young children. Meanwhile, dupilumab is approved for children aged 6 months and older; unfortunately, the indication for this is actually atopic dermatitis,” she explains.
When using this therapy for pediatric patients, it is therefore important to explain the options to parents and to inform them of side effects. Severe forms of asthma are rare in children; they are uncommon in adults but are more prevalent than in children.
Children and adolescents
One new chapter in the guideline describes giving medical advice to adolescents choosing a career. A table has been compiled that contains information regarding jobs and their respective allergy and asthma risk. The table is designed to be displayed in a medical practice.
Another chapter characterizes the interrelation between asthma and mental health. It differentiates between psychiatric comorbidities for which the patient requires professional help and the stress caused by the asthmatic illness itself. Many patients do not have a mental illness but do suffer under the everyday strain of having asthma, said Dr. Lommatzsch. Therefore, it is important to educate patients and their relatives on how to make a strength out of this supposed weakness – the asthmatic illness. “We have established a procedure for this and have summarized its key points in the guideline,” said Dr. Lommatzsch.
Other updates to the guideline cover asthma in different contexts, such as in pregnant women. The updates address adrenal insufficiency as a side effect of the use of steroids over many years. In addition, the guideline contains a chapter on digital apps that can help with diagnostics and medical history.
Dr. Lommatzsch highlighted a new tool. “By using 15 key points summarized in a table, the guideline displays the essential differences between COPD [chronic obstructive pulmonary disease] and asthma in terms of the symptoms and the findings. It is the most modern table available in Germany that differentiates between the two diseases.”
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
Asthma has long been associated with the use of inhalers to control symptoms. The new S2K guideline on the management of asthma, compiled by experts and published in March 2023, aims to change this. “For decades, we have known about medication that can be used to put asthma into remission. The patient can go out or travel on vacation without an inhaler. This is possible. This is a symptom-prevention approach,” said the guideline coordinator Marek Lommatzsch, MD, PhD, head senior physician of the pulmonology department at the University Medicine Rostock, Germany, in an interview.
The guideline was created by the German Respiratory Society, and a further 11 professional societies from Germany and Austria were involved in the update. The authors comprehensively revised the guideline from 2017, and the evidence-based national disease management guideline (NVL) for general asthma care from 2020 was amended.
Erika von Mutius, MD, PhD, pediatrician and professor of pediatric allergology and pulmonology at the Dr. Von Hauner Children’s Hospital, Munich, and director of the Institute of Asthma and Allergy Prevention at Helmholtz Munich, was not directly involved in the guideline. She said,
Anti-inflammatory therapy
The significance of anti-inflammatory therapy was stressed in the NVL from 2020. The new guideline holds that anti-inflammatory therapy should be considered the primary therapeutic option. “We are making a U-turn: only treat the respiratory inflammation. Salbutamol should still only be given in exceptional cases as required,” according to Dr. Lommatzsch.
In the guideline, asthma therapy is described using an updated step-by-step plan. Inhaled glucocorticoids (ICS) represent the most important pillar of therapy. ICS can be used as permanent therapy or as as-needed therapy in fixed combination with formoterol, which rapidly dilates the airways.
Allergen immunotherapy, also known as hyposensitization, and biologics are also effective anti-inflammatory treatments, Dr. Lommatzsch added. “We must ensure that these anti-inflammatory medicines are also used effectively. Mild to moderate forms of asthma can be treated easily by a primary care physician,” he said. Basic diagnostics in the form of a blood sample are required. A somewhat more comprehensive medical history is also needed. “It takes a little more time and involves more than just taking the inhaler out of the cupboard.”
The situation regarding children, however, is a little different with regard to anti-inflammatory therapy, Dr. Von Mutius explained. “Childhood asthma has many forms, and confirming the diagnosis is not always straightforward, especially in infancy. If needed, salbutamol can be prescribed. However, the anti-inflammatory medication should usually also be administered.”
She emphasized that the guideline has been designed in a sophisticated way that offers the option of “using medical experience to see what is suitable for this family or better for this patient. This is still always subject to medical judgment and responsibility. I find this really successful.”
Diagnostics using biomarkers
The previous guideline concentrated on measuring lung function as a way of diagnosing asthmatic illness. Three biomarkers were brought to the fore:
- Eosinophils in the blood.
- IgE levels.
- The FeNO test (proportion of nitrogen monoxide in exhaled air).
Slightly amended, the guideline now states that the FeNO test is implemented as “an integral component of specialist diagnosis.”
The test measures the nitrogen monoxide content of exhaled air as an indicator of inflammation in the airways. However, this test must often be paid for by the patient. “In this respect, we want to give a nudge in the direction of the political decision-makers,” emphasized Dr. Lommatzsch.
Dr. Von Mutius added that use of the FeNO test has not been established in many practices and outpatient clinics. The inflammatory marker is also subject to fluctuations. “This is an update to the guideline where we must wait to see the political response.”
Which biologic?
Despite treatment with the established therapies, the symptoms of asthma can persist in some people with severe forms of the condition. Biologics are highly effective for these patients and are preferable in the last stage of therapy to long-term therapy with oral steroids, which have numerous side effects. The current guideline provides an overview diagram to help decide which biologic is suitable for which patient.
“There are six biologics that can be used to treat severe asthma. Officially, almost any biologic can be taken into consideration for a patient, since the approvals overlap. Nevertheless, we know that certain patients benefit hugely from certain biologics. A targeted choice should therefore be made,” explained Dr. Lommatzsch.
Biologics were mentioned in the 2020 NVL but not to the great extent that they are in the latest version. “For the first time, we have created an overview diagram for the individual choice of biologic. With it, we have now set a standard,” said Dr. Lommatzsch.
Therapy with biologics has brought about rapid progress for adults. Dr. Von Mutius anticipates challenges in approving such therapeutics for pediatric treatment. “As is often the case, these therapies are not approved for young children. Meanwhile, dupilumab is approved for children aged 6 months and older; unfortunately, the indication for this is actually atopic dermatitis,” she explains.
When using this therapy for pediatric patients, it is therefore important to explain the options to parents and to inform them of side effects. Severe forms of asthma are rare in children; they are uncommon in adults but are more prevalent than in children.
Children and adolescents
One new chapter in the guideline describes giving medical advice to adolescents choosing a career. A table has been compiled that contains information regarding jobs and their respective allergy and asthma risk. The table is designed to be displayed in a medical practice.
Another chapter characterizes the interrelation between asthma and mental health. It differentiates between psychiatric comorbidities for which the patient requires professional help and the stress caused by the asthmatic illness itself. Many patients do not have a mental illness but do suffer under the everyday strain of having asthma, said Dr. Lommatzsch. Therefore, it is important to educate patients and their relatives on how to make a strength out of this supposed weakness – the asthmatic illness. “We have established a procedure for this and have summarized its key points in the guideline,” said Dr. Lommatzsch.
Other updates to the guideline cover asthma in different contexts, such as in pregnant women. The updates address adrenal insufficiency as a side effect of the use of steroids over many years. In addition, the guideline contains a chapter on digital apps that can help with diagnostics and medical history.
Dr. Lommatzsch highlighted a new tool. “By using 15 key points summarized in a table, the guideline displays the essential differences between COPD [chronic obstructive pulmonary disease] and asthma in terms of the symptoms and the findings. It is the most modern table available in Germany that differentiates between the two diseases.”
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
FDA approves first over-the-counter birth control pill
The Food and Drug Administration’s approval today of the first birth control pill for women to be available without a prescription is being hailed by many as a long-needed development, but there remain questions to be resolved, including how much the drug will cost and how it will be used.
The drug, Opill, is expected to be available early next year, and its maker has yet to reveal a retail price. It is the same birth control pill that has been available by prescription for 50 years. But for the first time, women will be able to buy the contraception at a local pharmacy, other retail locations, or online without having to see a doctor first.
Likely to drive debate
Contraception in the United States is not without controversy. The FDA’s approval spurred reactions both for and against making hormonal birth control for women available without a prescription.
“It’s an exciting time, especially right now when reproductive rights are being curtailed in a lot of states. Giving people an additional option for contraception will change people’s lives,” said Beverly Gray, MD, division director of Women’s Community and Population Health at Duke University Medical Center in Durham, N.C.
“It’s a huge win for patients who need better access to contraception,” said Dr. Gray, who is also a spokesperson for the American College of Obstetricians and Gynecologists.
Women who want hormonal birth control but live in areas without convenient access to a doctor, women who cannot easily take time off of work to see a doctor and get a prescription filled, and women without insurance are examples of people who will benefit, she said.
The Catholic Medical Association, in contrast, expressed “deep concern and disappointment” after an FDA advisory committee’s unanimous vote on May 11 recommending the drug be available over the counter. In a statement after the vote, the group cited “extensive medical studies demonstrating the risks and adverse effects of hormonal contraceptives,” adding that “the social impact of [full approval] would be dramatic.”
But doctors largely disagreed.
“It is definitely a huge win for reproductive autonomy. I’m glad that the FDA is prioritizing patient safety and well-being over politics,” said Catherine Cansino, MD, MPH, an ob.gyn. and clinical professor in the University of California Davis department of obstetrics and gynecology. She said the FDA approved the over-the-counter version because the medication is safe.
While opponents like the Catholic Medical Association cite safety concerns and believe doctors should screen all women before prescribing hormonal contraception, Dr. Gray disagreed. “There’s a lot of evidence that patients can figure out if a progestin-only pill is right for them and safe for them. Medical professionals don’t have to be the gatekeepers for contraception,” she said.
Pricing unknown
Whether insurance companies will pay for Opill now that it will be available without a prescription remains unknown. For some medications, paying a copay through insurance can be less expensive than buying at a retail price.
“Although pricing issues will be relevant, the FDA’s decision will enhance women’s access to hormonal birth control,” said Andrew M. Kaunitz, MD, a professor and associate chairman in the department of obstetrics and gynecology at the University of Florida College of Medicine in Jacksonville.
The drugmaker, Perrigo, based in Ireland, has not yet announced how much the pill will cost. The price tag could affect how widely available this form of birth control is. The drug has been shown to be as much as 93% effective for pregnancy prevention. Perrigo says it plans to make the pill available at low or no cost to some women.
Caveats to consider
There are some women for whom hormonal contraceptives have always carried greater risks. For example, women who have breast cancer or a history of breast cancer should not use hormonal contraceptives, the FDA said in a news release announcing the approval. Women with other types of cancer should check with their doctors first, the agency noted.
Women who smoke, who take some medications to lower blood pressure, or who have migraines should also take caution, Dr. Cansino said. “People with migraines may not be suitable for over-the-counter oral contraceptives. But a simple screening through a provider can identify whether you are truly eligible or not.”
Irregular bleeding, headaches, dizziness, nausea, increased appetite, belly pain, cramps, or bloating are the most common side effects of Opill, the FDA said.
The Opill is a progestin-only birth control pill. Similar pills have been available in the United Kingdiom for about 2 years, often referred to as “mini pills” because they contain a single hormone. In contrast, prescription birth control pills in the United States and elsewhere contain more than one hormone, estrogen and progestin, to prevent pregnancy.
Prescription pill packs for combination contraception often feature a week of placebo pills without an active ingredient. While skipping a placebo pill might not make a difference in pregnancy prevention, Opill is different. Every pill in the packet will contain medication, Gray said. “So it’s important to take the pill the same time every day for it to be most effective.”
Even though this may mean one less visit to your doctor, Dr. Kaunitz hopes women will stay up to date on their other medical checkups. “One of our challenges as providers of care to women will be to encourage them to continue to receive important services, including cancer screening and vaccinations, even while they can initiate and continue hormonal contraception without contact with a provider.”
Just the beginning?
The American Medical Association hopes this approval signals more to come.
“While we applaud this move, the AMA continues to urge the FDA and HHS to consider a variety of oral contraceptive options for over-the-counter use,” the association, which has more than 250,000 doctor members, said in a statement. “It is important patients have options when choosing which type of birth control works best for them,”
The American College of Obstetricians and Gynecologists said the FDA’s decision will help many women. “We are glad that more patients will now be empowered to choose when and where they obtain a safe method of contraception without having to wait for a medical appointment or for a prescription to be filled,” Verda J. Hicks, MD, the group’s president, and Christopher M. Zahn, MD, interim chief executive officer, said in a statement.
“Allowing individuals to access birth control at their local pharmacy or drug store will eliminate some barriers,” they said.
A version of this article first appeared on WebMD.com.
This article was updated 7/13/23.
The Food and Drug Administration’s approval today of the first birth control pill for women to be available without a prescription is being hailed by many as a long-needed development, but there remain questions to be resolved, including how much the drug will cost and how it will be used.
The drug, Opill, is expected to be available early next year, and its maker has yet to reveal a retail price. It is the same birth control pill that has been available by prescription for 50 years. But for the first time, women will be able to buy the contraception at a local pharmacy, other retail locations, or online without having to see a doctor first.
Likely to drive debate
Contraception in the United States is not without controversy. The FDA’s approval spurred reactions both for and against making hormonal birth control for women available without a prescription.
“It’s an exciting time, especially right now when reproductive rights are being curtailed in a lot of states. Giving people an additional option for contraception will change people’s lives,” said Beverly Gray, MD, division director of Women’s Community and Population Health at Duke University Medical Center in Durham, N.C.
“It’s a huge win for patients who need better access to contraception,” said Dr. Gray, who is also a spokesperson for the American College of Obstetricians and Gynecologists.
Women who want hormonal birth control but live in areas without convenient access to a doctor, women who cannot easily take time off of work to see a doctor and get a prescription filled, and women without insurance are examples of people who will benefit, she said.
The Catholic Medical Association, in contrast, expressed “deep concern and disappointment” after an FDA advisory committee’s unanimous vote on May 11 recommending the drug be available over the counter. In a statement after the vote, the group cited “extensive medical studies demonstrating the risks and adverse effects of hormonal contraceptives,” adding that “the social impact of [full approval] would be dramatic.”
But doctors largely disagreed.
“It is definitely a huge win for reproductive autonomy. I’m glad that the FDA is prioritizing patient safety and well-being over politics,” said Catherine Cansino, MD, MPH, an ob.gyn. and clinical professor in the University of California Davis department of obstetrics and gynecology. She said the FDA approved the over-the-counter version because the medication is safe.
While opponents like the Catholic Medical Association cite safety concerns and believe doctors should screen all women before prescribing hormonal contraception, Dr. Gray disagreed. “There’s a lot of evidence that patients can figure out if a progestin-only pill is right for them and safe for them. Medical professionals don’t have to be the gatekeepers for contraception,” she said.
Pricing unknown
Whether insurance companies will pay for Opill now that it will be available without a prescription remains unknown. For some medications, paying a copay through insurance can be less expensive than buying at a retail price.
“Although pricing issues will be relevant, the FDA’s decision will enhance women’s access to hormonal birth control,” said Andrew M. Kaunitz, MD, a professor and associate chairman in the department of obstetrics and gynecology at the University of Florida College of Medicine in Jacksonville.
The drugmaker, Perrigo, based in Ireland, has not yet announced how much the pill will cost. The price tag could affect how widely available this form of birth control is. The drug has been shown to be as much as 93% effective for pregnancy prevention. Perrigo says it plans to make the pill available at low or no cost to some women.
Caveats to consider
There are some women for whom hormonal contraceptives have always carried greater risks. For example, women who have breast cancer or a history of breast cancer should not use hormonal contraceptives, the FDA said in a news release announcing the approval. Women with other types of cancer should check with their doctors first, the agency noted.
Women who smoke, who take some medications to lower blood pressure, or who have migraines should also take caution, Dr. Cansino said. “People with migraines may not be suitable for over-the-counter oral contraceptives. But a simple screening through a provider can identify whether you are truly eligible or not.”
Irregular bleeding, headaches, dizziness, nausea, increased appetite, belly pain, cramps, or bloating are the most common side effects of Opill, the FDA said.
The Opill is a progestin-only birth control pill. Similar pills have been available in the United Kingdiom for about 2 years, often referred to as “mini pills” because they contain a single hormone. In contrast, prescription birth control pills in the United States and elsewhere contain more than one hormone, estrogen and progestin, to prevent pregnancy.
Prescription pill packs for combination contraception often feature a week of placebo pills without an active ingredient. While skipping a placebo pill might not make a difference in pregnancy prevention, Opill is different. Every pill in the packet will contain medication, Gray said. “So it’s important to take the pill the same time every day for it to be most effective.”
Even though this may mean one less visit to your doctor, Dr. Kaunitz hopes women will stay up to date on their other medical checkups. “One of our challenges as providers of care to women will be to encourage them to continue to receive important services, including cancer screening and vaccinations, even while they can initiate and continue hormonal contraception without contact with a provider.”
Just the beginning?
The American Medical Association hopes this approval signals more to come.
“While we applaud this move, the AMA continues to urge the FDA and HHS to consider a variety of oral contraceptive options for over-the-counter use,” the association, which has more than 250,000 doctor members, said in a statement. “It is important patients have options when choosing which type of birth control works best for them,”
The American College of Obstetricians and Gynecologists said the FDA’s decision will help many women. “We are glad that more patients will now be empowered to choose when and where they obtain a safe method of contraception without having to wait for a medical appointment or for a prescription to be filled,” Verda J. Hicks, MD, the group’s president, and Christopher M. Zahn, MD, interim chief executive officer, said in a statement.
“Allowing individuals to access birth control at their local pharmacy or drug store will eliminate some barriers,” they said.
A version of this article first appeared on WebMD.com.
This article was updated 7/13/23.
The Food and Drug Administration’s approval today of the first birth control pill for women to be available without a prescription is being hailed by many as a long-needed development, but there remain questions to be resolved, including how much the drug will cost and how it will be used.
The drug, Opill, is expected to be available early next year, and its maker has yet to reveal a retail price. It is the same birth control pill that has been available by prescription for 50 years. But for the first time, women will be able to buy the contraception at a local pharmacy, other retail locations, or online without having to see a doctor first.
Likely to drive debate
Contraception in the United States is not without controversy. The FDA’s approval spurred reactions both for and against making hormonal birth control for women available without a prescription.
“It’s an exciting time, especially right now when reproductive rights are being curtailed in a lot of states. Giving people an additional option for contraception will change people’s lives,” said Beverly Gray, MD, division director of Women’s Community and Population Health at Duke University Medical Center in Durham, N.C.
“It’s a huge win for patients who need better access to contraception,” said Dr. Gray, who is also a spokesperson for the American College of Obstetricians and Gynecologists.
Women who want hormonal birth control but live in areas without convenient access to a doctor, women who cannot easily take time off of work to see a doctor and get a prescription filled, and women without insurance are examples of people who will benefit, she said.
The Catholic Medical Association, in contrast, expressed “deep concern and disappointment” after an FDA advisory committee’s unanimous vote on May 11 recommending the drug be available over the counter. In a statement after the vote, the group cited “extensive medical studies demonstrating the risks and adverse effects of hormonal contraceptives,” adding that “the social impact of [full approval] would be dramatic.”
But doctors largely disagreed.
“It is definitely a huge win for reproductive autonomy. I’m glad that the FDA is prioritizing patient safety and well-being over politics,” said Catherine Cansino, MD, MPH, an ob.gyn. and clinical professor in the University of California Davis department of obstetrics and gynecology. She said the FDA approved the over-the-counter version because the medication is safe.
While opponents like the Catholic Medical Association cite safety concerns and believe doctors should screen all women before prescribing hormonal contraception, Dr. Gray disagreed. “There’s a lot of evidence that patients can figure out if a progestin-only pill is right for them and safe for them. Medical professionals don’t have to be the gatekeepers for contraception,” she said.
Pricing unknown
Whether insurance companies will pay for Opill now that it will be available without a prescription remains unknown. For some medications, paying a copay through insurance can be less expensive than buying at a retail price.
“Although pricing issues will be relevant, the FDA’s decision will enhance women’s access to hormonal birth control,” said Andrew M. Kaunitz, MD, a professor and associate chairman in the department of obstetrics and gynecology at the University of Florida College of Medicine in Jacksonville.
The drugmaker, Perrigo, based in Ireland, has not yet announced how much the pill will cost. The price tag could affect how widely available this form of birth control is. The drug has been shown to be as much as 93% effective for pregnancy prevention. Perrigo says it plans to make the pill available at low or no cost to some women.
Caveats to consider
There are some women for whom hormonal contraceptives have always carried greater risks. For example, women who have breast cancer or a history of breast cancer should not use hormonal contraceptives, the FDA said in a news release announcing the approval. Women with other types of cancer should check with their doctors first, the agency noted.
Women who smoke, who take some medications to lower blood pressure, or who have migraines should also take caution, Dr. Cansino said. “People with migraines may not be suitable for over-the-counter oral contraceptives. But a simple screening through a provider can identify whether you are truly eligible or not.”
Irregular bleeding, headaches, dizziness, nausea, increased appetite, belly pain, cramps, or bloating are the most common side effects of Opill, the FDA said.
The Opill is a progestin-only birth control pill. Similar pills have been available in the United Kingdiom for about 2 years, often referred to as “mini pills” because they contain a single hormone. In contrast, prescription birth control pills in the United States and elsewhere contain more than one hormone, estrogen and progestin, to prevent pregnancy.
Prescription pill packs for combination contraception often feature a week of placebo pills without an active ingredient. While skipping a placebo pill might not make a difference in pregnancy prevention, Opill is different. Every pill in the packet will contain medication, Gray said. “So it’s important to take the pill the same time every day for it to be most effective.”
Even though this may mean one less visit to your doctor, Dr. Kaunitz hopes women will stay up to date on their other medical checkups. “One of our challenges as providers of care to women will be to encourage them to continue to receive important services, including cancer screening and vaccinations, even while they can initiate and continue hormonal contraception without contact with a provider.”
Just the beginning?
The American Medical Association hopes this approval signals more to come.
“While we applaud this move, the AMA continues to urge the FDA and HHS to consider a variety of oral contraceptive options for over-the-counter use,” the association, which has more than 250,000 doctor members, said in a statement. “It is important patients have options when choosing which type of birth control works best for them,”
The American College of Obstetricians and Gynecologists said the FDA’s decision will help many women. “We are glad that more patients will now be empowered to choose when and where they obtain a safe method of contraception without having to wait for a medical appointment or for a prescription to be filled,” Verda J. Hicks, MD, the group’s president, and Christopher M. Zahn, MD, interim chief executive officer, said in a statement.
“Allowing individuals to access birth control at their local pharmacy or drug store will eliminate some barriers,” they said.
A version of this article first appeared on WebMD.com.
This article was updated 7/13/23.
How psychedelics can heal a broken mind
As children learn to walk and talk, their brains are remarkably open to new information. They gather knowledge from parents, their environment, and trial and error. Teenagers do too, as they adopt the emotional and intellectual skills needed to become adults.
In adulthood, however, our minds become relatively locked, closed to new information. This saves energy and lets us navigate the world more efficiently. But that also makes it harder to adapt, learn a new language or skill, or recover from psychological or physical trauma. For those who’ve dealt with abuse, abandonment, or physical violence, that lockdown can lead to a lifetime of suffering, substance abuse, and other maladaptive behaviors.
The study, published in Nature, reflects a renaissance of using and researching psychedelics to treat a range of mental health conditions.
Scientists at Johns Hopkins University in Baltimore were investigating the drugs’ effects on “critical periods” for social learning, times when the brain is more open to new information that diminish as we age. Success in mice suggests that psychedelics can start a fresh period of learning.
If the finding bears out in future studies, the therapeutic horizon for psychedelics could expand to other opportunities to retrain the brain, including recovery from a stroke, traumatic brain injury, and even hearing loss and paralysis.
The stakes are big, and the future is promising, said lead researcher Gul Dolen, MD, PhD, an associate professor of neuroscience at Johns Hopkins University. Psychedelics “could be the key that unlocks the brain and helps people after one dose, rather than subjecting them to a lifetime of drugs.”
The psychedelic advantage
Dr. Dolen, who launched her career in addiction studies, has long been fascinated by critical periods and their influence on adult behavior.
“There have been three Nobel Prizes awarded for work on critical periods,” she said. One study in mice, for instance, identified 15 periods of social learning that define their behaviors for a lifetime.
Prior research has found that MDMA (commonly known as ecstasy) can help soldiers reconsider traumatic events on the battlefield, learn from them, and move on. That phenomenon had all the earmarks of a critical period for social learning. Perhaps, Dr. Dolen said, psychedelics could open a critical period in a soldier’s life – or a drug-addicted person’s or rape survivor’s – and give them tools to process their trauma.
In the placebo-controlled experiment, she and her team gave mice psychedelic drugs and a behavioral test to gauge the rodents’ ability to learn from their environment.
“All of the psychedelics opened the critical period of social learning for varying lengths of time,” said Dr. Dolen.
Ketamine achieved that reopening for 2 days, while the other drugs – ibogaine, LSD, MDMA, and psilocybin – opened critical periods of between 2 and 4 weeks, long after the drugs’ acute effects had worn off.
In humans, Dr. Dolen stressed, opening a critical period would be a sensitive process.
“You wouldn’t achieve these results if you dropped ecstasy and attended a rave,” she said. “The key seems to be to establish an intention for the therapy: Discuss what you hope to get from the experience, be guided through it, and process it with the therapist after the fact.”
“You need to be careful with a patient once they’re off the psychedelic,” she said, “because they’re in a state of openness and vulnerability similar to a child.”
The push for psychedelic therapy
Another psychedelics researcher, Matthew Lowe, PhD, sees promise in the Johns Hopkins study. The drugs “place the brain in a more malleable and flexible state,” said Dr. Lowe, the executive director and chief science officer for Unlimited Sciences, a psychedelics research nonprofit.
He expects that psychedelics may help people break out of negative behavior patterns.
“These findings show significant promise for treating a wide range of neuropsychiatric diseases, including depression, PTSD, and addiction,” he said.
Dr. Dolen said using psychedelics in critical-period therapy “opens up all sorts of possibilities for the rest of the brain.” Future research may also lead to treatments for deafness, physical disabilities, and drug and alcohol addiction. She is currently raising funds for a clinical trial to see if psychedelics can improve motor impairment after a stroke.
“Growing legislative openness” to the use of psychedelics could open the door for millions to benefit from mental health treatment “through clinical trials and legal therapeutic pathways as they open up,” said Benjamin Lightburn, CEO and cofounder of Filament Health, a company based in British Columbia that provides naturally derived psilocybin for clinical trials.
Several states have made moves toward decriminalization or permitting the drugs’ use under medical supervision. In a scientific paper, Washington University researchers, using an analytic model based on marijuana legalization, projected that most states will legalize psychedelics in the next 10-15 years. And on July 1, Australia became the first country to allow psilocybin and MDMA to be prescribed by doctors to treat psychiatric conditions. The U.S. could potentially approve MDMA for therapy later in 2023.
A version of this article first appeared on WebMD.com.
As children learn to walk and talk, their brains are remarkably open to new information. They gather knowledge from parents, their environment, and trial and error. Teenagers do too, as they adopt the emotional and intellectual skills needed to become adults.
In adulthood, however, our minds become relatively locked, closed to new information. This saves energy and lets us navigate the world more efficiently. But that also makes it harder to adapt, learn a new language or skill, or recover from psychological or physical trauma. For those who’ve dealt with abuse, abandonment, or physical violence, that lockdown can lead to a lifetime of suffering, substance abuse, and other maladaptive behaviors.
The study, published in Nature, reflects a renaissance of using and researching psychedelics to treat a range of mental health conditions.
Scientists at Johns Hopkins University in Baltimore were investigating the drugs’ effects on “critical periods” for social learning, times when the brain is more open to new information that diminish as we age. Success in mice suggests that psychedelics can start a fresh period of learning.
If the finding bears out in future studies, the therapeutic horizon for psychedelics could expand to other opportunities to retrain the brain, including recovery from a stroke, traumatic brain injury, and even hearing loss and paralysis.
The stakes are big, and the future is promising, said lead researcher Gul Dolen, MD, PhD, an associate professor of neuroscience at Johns Hopkins University. Psychedelics “could be the key that unlocks the brain and helps people after one dose, rather than subjecting them to a lifetime of drugs.”
The psychedelic advantage
Dr. Dolen, who launched her career in addiction studies, has long been fascinated by critical periods and their influence on adult behavior.
“There have been three Nobel Prizes awarded for work on critical periods,” she said. One study in mice, for instance, identified 15 periods of social learning that define their behaviors for a lifetime.
Prior research has found that MDMA (commonly known as ecstasy) can help soldiers reconsider traumatic events on the battlefield, learn from them, and move on. That phenomenon had all the earmarks of a critical period for social learning. Perhaps, Dr. Dolen said, psychedelics could open a critical period in a soldier’s life – or a drug-addicted person’s or rape survivor’s – and give them tools to process their trauma.
In the placebo-controlled experiment, she and her team gave mice psychedelic drugs and a behavioral test to gauge the rodents’ ability to learn from their environment.
“All of the psychedelics opened the critical period of social learning for varying lengths of time,” said Dr. Dolen.
Ketamine achieved that reopening for 2 days, while the other drugs – ibogaine, LSD, MDMA, and psilocybin – opened critical periods of between 2 and 4 weeks, long after the drugs’ acute effects had worn off.
In humans, Dr. Dolen stressed, opening a critical period would be a sensitive process.
“You wouldn’t achieve these results if you dropped ecstasy and attended a rave,” she said. “The key seems to be to establish an intention for the therapy: Discuss what you hope to get from the experience, be guided through it, and process it with the therapist after the fact.”
“You need to be careful with a patient once they’re off the psychedelic,” she said, “because they’re in a state of openness and vulnerability similar to a child.”
The push for psychedelic therapy
Another psychedelics researcher, Matthew Lowe, PhD, sees promise in the Johns Hopkins study. The drugs “place the brain in a more malleable and flexible state,” said Dr. Lowe, the executive director and chief science officer for Unlimited Sciences, a psychedelics research nonprofit.
He expects that psychedelics may help people break out of negative behavior patterns.
“These findings show significant promise for treating a wide range of neuropsychiatric diseases, including depression, PTSD, and addiction,” he said.
Dr. Dolen said using psychedelics in critical-period therapy “opens up all sorts of possibilities for the rest of the brain.” Future research may also lead to treatments for deafness, physical disabilities, and drug and alcohol addiction. She is currently raising funds for a clinical trial to see if psychedelics can improve motor impairment after a stroke.
“Growing legislative openness” to the use of psychedelics could open the door for millions to benefit from mental health treatment “through clinical trials and legal therapeutic pathways as they open up,” said Benjamin Lightburn, CEO and cofounder of Filament Health, a company based in British Columbia that provides naturally derived psilocybin for clinical trials.
Several states have made moves toward decriminalization or permitting the drugs’ use under medical supervision. In a scientific paper, Washington University researchers, using an analytic model based on marijuana legalization, projected that most states will legalize psychedelics in the next 10-15 years. And on July 1, Australia became the first country to allow psilocybin and MDMA to be prescribed by doctors to treat psychiatric conditions. The U.S. could potentially approve MDMA for therapy later in 2023.
A version of this article first appeared on WebMD.com.
As children learn to walk and talk, their brains are remarkably open to new information. They gather knowledge from parents, their environment, and trial and error. Teenagers do too, as they adopt the emotional and intellectual skills needed to become adults.
In adulthood, however, our minds become relatively locked, closed to new information. This saves energy and lets us navigate the world more efficiently. But that also makes it harder to adapt, learn a new language or skill, or recover from psychological or physical trauma. For those who’ve dealt with abuse, abandonment, or physical violence, that lockdown can lead to a lifetime of suffering, substance abuse, and other maladaptive behaviors.
The study, published in Nature, reflects a renaissance of using and researching psychedelics to treat a range of mental health conditions.
Scientists at Johns Hopkins University in Baltimore were investigating the drugs’ effects on “critical periods” for social learning, times when the brain is more open to new information that diminish as we age. Success in mice suggests that psychedelics can start a fresh period of learning.
If the finding bears out in future studies, the therapeutic horizon for psychedelics could expand to other opportunities to retrain the brain, including recovery from a stroke, traumatic brain injury, and even hearing loss and paralysis.
The stakes are big, and the future is promising, said lead researcher Gul Dolen, MD, PhD, an associate professor of neuroscience at Johns Hopkins University. Psychedelics “could be the key that unlocks the brain and helps people after one dose, rather than subjecting them to a lifetime of drugs.”
The psychedelic advantage
Dr. Dolen, who launched her career in addiction studies, has long been fascinated by critical periods and their influence on adult behavior.
“There have been three Nobel Prizes awarded for work on critical periods,” she said. One study in mice, for instance, identified 15 periods of social learning that define their behaviors for a lifetime.
Prior research has found that MDMA (commonly known as ecstasy) can help soldiers reconsider traumatic events on the battlefield, learn from them, and move on. That phenomenon had all the earmarks of a critical period for social learning. Perhaps, Dr. Dolen said, psychedelics could open a critical period in a soldier’s life – or a drug-addicted person’s or rape survivor’s – and give them tools to process their trauma.
In the placebo-controlled experiment, she and her team gave mice psychedelic drugs and a behavioral test to gauge the rodents’ ability to learn from their environment.
“All of the psychedelics opened the critical period of social learning for varying lengths of time,” said Dr. Dolen.
Ketamine achieved that reopening for 2 days, while the other drugs – ibogaine, LSD, MDMA, and psilocybin – opened critical periods of between 2 and 4 weeks, long after the drugs’ acute effects had worn off.
In humans, Dr. Dolen stressed, opening a critical period would be a sensitive process.
“You wouldn’t achieve these results if you dropped ecstasy and attended a rave,” she said. “The key seems to be to establish an intention for the therapy: Discuss what you hope to get from the experience, be guided through it, and process it with the therapist after the fact.”
“You need to be careful with a patient once they’re off the psychedelic,” she said, “because they’re in a state of openness and vulnerability similar to a child.”
The push for psychedelic therapy
Another psychedelics researcher, Matthew Lowe, PhD, sees promise in the Johns Hopkins study. The drugs “place the brain in a more malleable and flexible state,” said Dr. Lowe, the executive director and chief science officer for Unlimited Sciences, a psychedelics research nonprofit.
He expects that psychedelics may help people break out of negative behavior patterns.
“These findings show significant promise for treating a wide range of neuropsychiatric diseases, including depression, PTSD, and addiction,” he said.
Dr. Dolen said using psychedelics in critical-period therapy “opens up all sorts of possibilities for the rest of the brain.” Future research may also lead to treatments for deafness, physical disabilities, and drug and alcohol addiction. She is currently raising funds for a clinical trial to see if psychedelics can improve motor impairment after a stroke.
“Growing legislative openness” to the use of psychedelics could open the door for millions to benefit from mental health treatment “through clinical trials and legal therapeutic pathways as they open up,” said Benjamin Lightburn, CEO and cofounder of Filament Health, a company based in British Columbia that provides naturally derived psilocybin for clinical trials.
Several states have made moves toward decriminalization or permitting the drugs’ use under medical supervision. In a scientific paper, Washington University researchers, using an analytic model based on marijuana legalization, projected that most states will legalize psychedelics in the next 10-15 years. And on July 1, Australia became the first country to allow psilocybin and MDMA to be prescribed by doctors to treat psychiatric conditions. The U.S. could potentially approve MDMA for therapy later in 2023.
A version of this article first appeared on WebMD.com.
FROM NATURE
Eosinophilic esophagitis: A year in review
At the AGA postgraduate course in May, we highlighted recent noteworthy randomized controlled trials (RCT) using eosinophil-targeting biologic therapy, esophageal-optimized corticosteroid preparations, and dietary elimination in EoE.
Dupilumab, a monoclonal antibody that blocks interleukin-4 and IL-13 signaling, was tested in a phase 3 trial for adults and adolescents with EoE.1 In this double-blind, randomized, placebo-controlled trial, the efficacy of subcutaneous dupilumab 300 mg weekly or every other week was compared against placebo. Stringent histologic remission (≤ 6 eosinophils/high power field) occurred in approximately 60% who received dupilumab (either dose) versus 5% in placebo. However, significant symptom improvement was seen only with 300 g weekly dupilumab.
On the topical corticosteroid front, the results of two RCTs using fluticasone orally disintegrating tablet (APT-1011) and budesonide oral suspension (BOS) were published. In the APT-1011 phase 2b trial, patients were randomized to receive 1.5 mg or 3 mg daily or b.i.d. versus placebo for 12 weeks.2 High histologic response rates and improvement in dysphagia frequency were seen with all ≥ 3-mg daily-dose APT-1011, compared with placebo. However, adverse events (that is, candidiasis) were highest among those on 3 mg b.i.d. Thus, 3 mg daily APT-1011 was thought to offer the most favorable risk-benefit profile. In the BOS phase 3 trial, patients were randomized 2:1 to received BOS 2 mg b.i.d. or placebo for 12 weeks.3 BOS was superior to placebo in histologic, symptomatic, and endoscopic outcomes.
Diet remains the only therapy targeting the cause of EoE and offers a potential drug-free remission. In the randomized, open label trial of 1- versus 6-food elimination diet, adult patients were allocated 1:1 to 1FED (animal milk) or 6FED (animal milk, wheat, egg, soy, fish/shellfish, and peanuts/tree nuts) for 6 weeks.4 No significant difference in partial or stringent remission was found between the two groups. Step-up therapy resulted in an additional 43% histologic response in those who underwent 6FED after failing 1FED and 82% histologic response in those who received swallowed fluticasone 880 mcg b.i.d after failing 6FED. Hence, eliminating animal milk alone in a step-up treatment approach is reasonable.
We have witnessed major progress to expand EoE treatment options in the last year. Long-term efficacy and side-effect data, as well as studies comparing between therapies are needed to improve shared decision-making and strategies to implement tailored care in EoE.
Dr. Chen is with the division of gastroenterology and hepatology, department of internal medicine at the University of Michigan, Ann Arbor. She disclosed consultancy work with Phathom Pharmaceuticals.
References
1. Dellon ES et al. N Engl J Med. 2022;387(25):2317-30.
2. Dellon ES et al. Clin Gastroenterol Hepatol. 2022;20(11):2485-94e15.
3. Hirano I et al. Budesonide. Clin Gastroenterol Hepatol. 2022;20(3):525-34e10.
4. Kliewer KL et al. Lancet Gastroenterol Hepatol. 2023;8(5):408-21.
At the AGA postgraduate course in May, we highlighted recent noteworthy randomized controlled trials (RCT) using eosinophil-targeting biologic therapy, esophageal-optimized corticosteroid preparations, and dietary elimination in EoE.
Dupilumab, a monoclonal antibody that blocks interleukin-4 and IL-13 signaling, was tested in a phase 3 trial for adults and adolescents with EoE.1 In this double-blind, randomized, placebo-controlled trial, the efficacy of subcutaneous dupilumab 300 mg weekly or every other week was compared against placebo. Stringent histologic remission (≤ 6 eosinophils/high power field) occurred in approximately 60% who received dupilumab (either dose) versus 5% in placebo. However, significant symptom improvement was seen only with 300 g weekly dupilumab.
On the topical corticosteroid front, the results of two RCTs using fluticasone orally disintegrating tablet (APT-1011) and budesonide oral suspension (BOS) were published. In the APT-1011 phase 2b trial, patients were randomized to receive 1.5 mg or 3 mg daily or b.i.d. versus placebo for 12 weeks.2 High histologic response rates and improvement in dysphagia frequency were seen with all ≥ 3-mg daily-dose APT-1011, compared with placebo. However, adverse events (that is, candidiasis) were highest among those on 3 mg b.i.d. Thus, 3 mg daily APT-1011 was thought to offer the most favorable risk-benefit profile. In the BOS phase 3 trial, patients were randomized 2:1 to received BOS 2 mg b.i.d. or placebo for 12 weeks.3 BOS was superior to placebo in histologic, symptomatic, and endoscopic outcomes.
Diet remains the only therapy targeting the cause of EoE and offers a potential drug-free remission. In the randomized, open label trial of 1- versus 6-food elimination diet, adult patients were allocated 1:1 to 1FED (animal milk) or 6FED (animal milk, wheat, egg, soy, fish/shellfish, and peanuts/tree nuts) for 6 weeks.4 No significant difference in partial or stringent remission was found between the two groups. Step-up therapy resulted in an additional 43% histologic response in those who underwent 6FED after failing 1FED and 82% histologic response in those who received swallowed fluticasone 880 mcg b.i.d after failing 6FED. Hence, eliminating animal milk alone in a step-up treatment approach is reasonable.
We have witnessed major progress to expand EoE treatment options in the last year. Long-term efficacy and side-effect data, as well as studies comparing between therapies are needed to improve shared decision-making and strategies to implement tailored care in EoE.
Dr. Chen is with the division of gastroenterology and hepatology, department of internal medicine at the University of Michigan, Ann Arbor. She disclosed consultancy work with Phathom Pharmaceuticals.
References
1. Dellon ES et al. N Engl J Med. 2022;387(25):2317-30.
2. Dellon ES et al. Clin Gastroenterol Hepatol. 2022;20(11):2485-94e15.
3. Hirano I et al. Budesonide. Clin Gastroenterol Hepatol. 2022;20(3):525-34e10.
4. Kliewer KL et al. Lancet Gastroenterol Hepatol. 2023;8(5):408-21.
At the AGA postgraduate course in May, we highlighted recent noteworthy randomized controlled trials (RCT) using eosinophil-targeting biologic therapy, esophageal-optimized corticosteroid preparations, and dietary elimination in EoE.
Dupilumab, a monoclonal antibody that blocks interleukin-4 and IL-13 signaling, was tested in a phase 3 trial for adults and adolescents with EoE.1 In this double-blind, randomized, placebo-controlled trial, the efficacy of subcutaneous dupilumab 300 mg weekly or every other week was compared against placebo. Stringent histologic remission (≤ 6 eosinophils/high power field) occurred in approximately 60% who received dupilumab (either dose) versus 5% in placebo. However, significant symptom improvement was seen only with 300 g weekly dupilumab.
On the topical corticosteroid front, the results of two RCTs using fluticasone orally disintegrating tablet (APT-1011) and budesonide oral suspension (BOS) were published. In the APT-1011 phase 2b trial, patients were randomized to receive 1.5 mg or 3 mg daily or b.i.d. versus placebo for 12 weeks.2 High histologic response rates and improvement in dysphagia frequency were seen with all ≥ 3-mg daily-dose APT-1011, compared with placebo. However, adverse events (that is, candidiasis) were highest among those on 3 mg b.i.d. Thus, 3 mg daily APT-1011 was thought to offer the most favorable risk-benefit profile. In the BOS phase 3 trial, patients were randomized 2:1 to received BOS 2 mg b.i.d. or placebo for 12 weeks.3 BOS was superior to placebo in histologic, symptomatic, and endoscopic outcomes.
Diet remains the only therapy targeting the cause of EoE and offers a potential drug-free remission. In the randomized, open label trial of 1- versus 6-food elimination diet, adult patients were allocated 1:1 to 1FED (animal milk) or 6FED (animal milk, wheat, egg, soy, fish/shellfish, and peanuts/tree nuts) for 6 weeks.4 No significant difference in partial or stringent remission was found between the two groups. Step-up therapy resulted in an additional 43% histologic response in those who underwent 6FED after failing 1FED and 82% histologic response in those who received swallowed fluticasone 880 mcg b.i.d after failing 6FED. Hence, eliminating animal milk alone in a step-up treatment approach is reasonable.
We have witnessed major progress to expand EoE treatment options in the last year. Long-term efficacy and side-effect data, as well as studies comparing between therapies are needed to improve shared decision-making and strategies to implement tailored care in EoE.
Dr. Chen is with the division of gastroenterology and hepatology, department of internal medicine at the University of Michigan, Ann Arbor. She disclosed consultancy work with Phathom Pharmaceuticals.
References
1. Dellon ES et al. N Engl J Med. 2022;387(25):2317-30.
2. Dellon ES et al. Clin Gastroenterol Hepatol. 2022;20(11):2485-94e15.
3. Hirano I et al. Budesonide. Clin Gastroenterol Hepatol. 2022;20(3):525-34e10.
4. Kliewer KL et al. Lancet Gastroenterol Hepatol. 2023;8(5):408-21.







