User login
For MD-IQ use only
An Epidemiologist’s Guide to Debunking Nutritional Research
You’re invited to a dinner party but you struggle to make small talk. Do not worry; that will invariably crop up over cocktails. Because all journalism has been reduced to listicles, here are four ways to seem clever at dinner parties.
1. The Predinner Cocktails: A Lesson in Reverse Causation
Wine connoisseurs sniff, swirl, and gently swish the wine in their mouths before spitting out and cleansing their palates to better appreciate the subtlety of each vintage. If you’re not an oenophile, no matter. Whenever somebody claims that moderate amounts of alcohol are good for your heart, this is your moment to pounce. Interject yourself in the conversation and tell everybody about reverse causation.
Reverse causation, also known as protopathic bias, involves misinterpreting the directionality of an association. You assume that X leads to Y, when in fact Y leads to X. Temporal paradoxes are useful plot devices in science fiction movies, but they have no place in medical research. In our bland world, cause must precede effect. As such, smoking leads to lung cancer; lung cancer doesn’t make you smoke more.
But with alcohol, directionality is less obvious. Many studies of alcohol and cardiovascular disease have demonstrated a U-shaped association, with risk being lowest among those who drink moderate amounts of alcohol (usually one to two drinks per day) and higher in those who drink more and also those who drink very little.
But one must ask why some people drink little or no alcohol. There is an important difference between former drinkers and never drinkers. Former drinkers cut back for a reason. More likely than not, the reason for this newfound sobriety was medical. A new cancer diagnosis, the emergence of atrial fibrillation, the development of diabetes, or rising blood pressure are all good reasons to reduce or eliminate alcohol. A cross-sectional study will fail to capture that alcohol consumption changes over time — people who now don’t drink may have imbibed alcohol in years past. It was not abstinence that led to an increased risk for heart disease; it was the increased risk for heart disease that led to abstinence.
You see the same phenomenon with the so-called obesity paradox. The idea that being a little overweight is good for you may appeal when you no longer fit into last year’s pants. But people who are underweight are so for a reason. Malnutrition, cachexia from cancer, or some other cause is almost certainly driving up the risk at the left-hand side of the U-shaped curve that makes the middle part seem better than it actually is.
Food consumption changes over time. A cross-sectional survey at one point in time cannot accurately capture past habits and distant exposures, especially for diseases such as heart disease and cancer that develop slowly over time. Studies on alcohol that try to overcome these shortcomings by eliminating former drinkers, or by using Mendelian randomization to better account for past exposure, do not show a cardiovascular benefit for moderate red wine drinking.
2. The Hors D’oeuvres — The Importance of RCTs
Now that you have made yourself the center of attention, it is time to cement your newfound reputation as a font of scientific knowledge. Most self-respecting hosts will serve smoked salmon as an amuse-bouche before the main meal. When someone mentions the health benefits of fish oils, you should take the opportunity to teach them about confounding.
Fish, especially cold-water fish from northern climates, have relatively high amounts of omega-3 fatty acids. Despite the plethora of observational studies suggesting a cardiovascular benefit, it’s now relatively clear that fish oil or omega-3 supplements have no medical benefit.
This will probably come as a shock to the worried well, but many studies, including VITAL and ASCEND, have demonstrated no cardiovascular or cancer benefit to supplementation with omega-3s. The reason is straightforward and explains why hormone replacement therapy, vitamin D, and myriad purported game-changers never panned out. Confounding is hard to overcome in observational research.
Prior to the publication of the Women’s Health Initiative (WHI) Study, hormone replacement therapy was routinely prescribed to postmenopausal women because numerous observational studies suggested a cardiovascular benefit. But with the publication of the WHI study, it became clear that much of that “benefit” was due to confounding. The women choosing to take hormones were more health conscious at baseline and healthier overall.
A similar phenomenon occurred during COVID. Patients with low serum vitamin D levels had worse outcomes, prompting many to suggest vitamin D supplementation as a possible treatment. Trials did not support the intervention because we’d overlooked the obvious. People with vitamin D deficiency have underlying health problems that contribute to the vitamin D deficiency. They are probably older, frailer, possibly with a poorer diet. No amount of statistical adjustment can account for all those differences, and some degree of residual confounding will always persist.
The only way to overcome confounding is with randomization. When patients are randomly assigned to one group or another, their baseline differences largely balance out if the randomization was performed properly and the groups were large enough. There is a role for observational research, such as in situations where ethics, cost, and practicality do not allow for a randomized controlled trial. But randomized controlled trials have largely put to rest the purported health benefits of over-the-counter fish oils, omega-3s, and vitamin D.
3. The Main Course — Absolute vs Relative Risk
When you get to the main course, all eyes will now be on you. You will almost certainly be called upon to pronounce on the harms or benefits of red meat consumption. Begin by regaling your guests with a little trivia. Ask them if they know the definition of red meat and white meat. When someone says pork is white meat, you can reveal that “pork, the other white meat,” was a marketing slogan with no scientific underpinning. Now that everyone is lulled into a stupefied silence, tell them that red meat comes from mammals and white meat comes from birds. As they process this revelation, you can now launch into the deeply mathematical concept of absolute vs relative risk.
Many etiquette books will caution against bringing up math at a dinner party. These books are wrong. Everyone finds math interesting if they are primed properly. For example, you can point to a study claiming that berries reduce cardiovascular risk in women. Even if true — and there is reason to be cautious, given the observational nature of the research — we need to understand what the authors meant by a 32% risk reduction. (Side note: It was a reduction in hazard, with a hazard ratio of 0.68 (95% CI, 0.49-0.96), but we won’t dwell on the difference between hazard ratios and risk ratios right now.)
This relative risk reduction has to be interpreted carefully. The authors divided the population into quintiles based on their consumption of anthocyanins (the antioxidant in blueberries and strawberries) and compared the bottom fifth (average consumption, 2.5 mg/d) with the top fifth (average consumption, 25 mg/d). The bottom quintile had 126 myocardial infarctions (MIs) over 324,793 patient-years compared with 59 MIs over 332,143 patient-years. Some quick math shows an approximate reduction from 39 to 18 MIs per 100,000 patient-years. Or to put it another way, you must get 4762 women to increase their berry consumption 10-fold for 1 year to prevent one heart attack. Feel free to show people how you calculated this number. They will be impressed by your head for numbers. It is nothing more than 39 minus 18, divided by 100,000, to get the absolute risk reduction. Take the reciprocal of this (ie, 1 divided by this number) to get the number needed to treat.
Describing risks in absolute terms or using number needed to treat (or harm) can help conceptualize statistics that are sometimes hard to wrap your head around.
4. Dessert — Funding
By the time the coffee is served, everyone will be hanging on to your every word. This is as it should be, and you should not be afraid of your newfound power and influence.
Dessert will probably involve some form of chocolate, possibly in cake format. (Anyone who serves fruit as dessert is not someone you should associate with.) Take the opportunity to tell your follow diners that chocolate is not actually good for you and will not boost brain performance.
The health benefits of chocolate are often repeated but rarely scrutinized. In fact, much of the scientific research purporting to show that chocolate is good for you did not actually study chocolate. It usually involved a cocoa bean extract because the chocolate manufacturing process destroys the supposedly health-promoting antioxidants in the cocoa bean. It is true that dark chocolate has more antioxidants than milk chocolate, and that the addition of milk to chocolate further inactivates the potentially healthy antioxidants. But the amount of sugar and fat that has to be added to chocolate to make it palatable precludes any serious consideration about health benefits. Dark chocolate may have less fat and sugar than milk chocolate, but it still has a lot.
But even the cocoa bean extract doesn’t seem to do much for your heart or your brain. The long-awaited COSMOS study was published with surprisingly little fanfare. The largest randomized controlled trial of chocolate (or rather cocoa bean extract) was supposed to settle the issue definitively.
COSMOS showed no cardiovascular or neurocognitive benefit to the cocoa bean extract. But the health halo of chocolate continues to be bolstered by many studies funded by chocolate manufacturers.
We are appropriately critical of the pharmaceutical industry’s involvement in drug research. However, we should not forget that any private entity is prone to the same self-interest regardless of its product’s tastiness. How many of you knew that there was an avocado lobby funding research? No matter how many industry-funded observational studies using surrogate endpoints are out there telling you that chocolate is healthy, a randomized trial with hard clinical endpoints such as COSMOS should generally win the day.
The Final Goodbyes — Summarizing Your Case
As the party slowly winds down and everyone is saddened that you will soon take your leave, synthesize everything you have taught them over the evening. Like movies, not all studies are good. Some are just bad. They can be prone to reverse causation or confounding, and they may report relative risks when absolute risks would be more telling. Reading research studies critically is essential for separating the wheat from the chaff. With the knowledge you have now imparted to your friends, they will be much better consumers of medical news, especially when it comes to food.
And they will no doubt thank you for it by never inviting you to another dinner party!
Labos, a cardiologist at Hôpital, Notre-Dame, Montreal, Quebec, Canada, has disclosed no relevant financial relationships. He has a degree in epidemiology.
A version of this article appeared on Medscape.com.
You’re invited to a dinner party but you struggle to make small talk. Do not worry; that will invariably crop up over cocktails. Because all journalism has been reduced to listicles, here are four ways to seem clever at dinner parties.
1. The Predinner Cocktails: A Lesson in Reverse Causation
Wine connoisseurs sniff, swirl, and gently swish the wine in their mouths before spitting out and cleansing their palates to better appreciate the subtlety of each vintage. If you’re not an oenophile, no matter. Whenever somebody claims that moderate amounts of alcohol are good for your heart, this is your moment to pounce. Interject yourself in the conversation and tell everybody about reverse causation.
Reverse causation, also known as protopathic bias, involves misinterpreting the directionality of an association. You assume that X leads to Y, when in fact Y leads to X. Temporal paradoxes are useful plot devices in science fiction movies, but they have no place in medical research. In our bland world, cause must precede effect. As such, smoking leads to lung cancer; lung cancer doesn’t make you smoke more.
But with alcohol, directionality is less obvious. Many studies of alcohol and cardiovascular disease have demonstrated a U-shaped association, with risk being lowest among those who drink moderate amounts of alcohol (usually one to two drinks per day) and higher in those who drink more and also those who drink very little.
But one must ask why some people drink little or no alcohol. There is an important difference between former drinkers and never drinkers. Former drinkers cut back for a reason. More likely than not, the reason for this newfound sobriety was medical. A new cancer diagnosis, the emergence of atrial fibrillation, the development of diabetes, or rising blood pressure are all good reasons to reduce or eliminate alcohol. A cross-sectional study will fail to capture that alcohol consumption changes over time — people who now don’t drink may have imbibed alcohol in years past. It was not abstinence that led to an increased risk for heart disease; it was the increased risk for heart disease that led to abstinence.
You see the same phenomenon with the so-called obesity paradox. The idea that being a little overweight is good for you may appeal when you no longer fit into last year’s pants. But people who are underweight are so for a reason. Malnutrition, cachexia from cancer, or some other cause is almost certainly driving up the risk at the left-hand side of the U-shaped curve that makes the middle part seem better than it actually is.
Food consumption changes over time. A cross-sectional survey at one point in time cannot accurately capture past habits and distant exposures, especially for diseases such as heart disease and cancer that develop slowly over time. Studies on alcohol that try to overcome these shortcomings by eliminating former drinkers, or by using Mendelian randomization to better account for past exposure, do not show a cardiovascular benefit for moderate red wine drinking.
2. The Hors D’oeuvres — The Importance of RCTs
Now that you have made yourself the center of attention, it is time to cement your newfound reputation as a font of scientific knowledge. Most self-respecting hosts will serve smoked salmon as an amuse-bouche before the main meal. When someone mentions the health benefits of fish oils, you should take the opportunity to teach them about confounding.
Fish, especially cold-water fish from northern climates, have relatively high amounts of omega-3 fatty acids. Despite the plethora of observational studies suggesting a cardiovascular benefit, it’s now relatively clear that fish oil or omega-3 supplements have no medical benefit.
This will probably come as a shock to the worried well, but many studies, including VITAL and ASCEND, have demonstrated no cardiovascular or cancer benefit to supplementation with omega-3s. The reason is straightforward and explains why hormone replacement therapy, vitamin D, and myriad purported game-changers never panned out. Confounding is hard to overcome in observational research.
Prior to the publication of the Women’s Health Initiative (WHI) Study, hormone replacement therapy was routinely prescribed to postmenopausal women because numerous observational studies suggested a cardiovascular benefit. But with the publication of the WHI study, it became clear that much of that “benefit” was due to confounding. The women choosing to take hormones were more health conscious at baseline and healthier overall.
A similar phenomenon occurred during COVID. Patients with low serum vitamin D levels had worse outcomes, prompting many to suggest vitamin D supplementation as a possible treatment. Trials did not support the intervention because we’d overlooked the obvious. People with vitamin D deficiency have underlying health problems that contribute to the vitamin D deficiency. They are probably older, frailer, possibly with a poorer diet. No amount of statistical adjustment can account for all those differences, and some degree of residual confounding will always persist.
The only way to overcome confounding is with randomization. When patients are randomly assigned to one group or another, their baseline differences largely balance out if the randomization was performed properly and the groups were large enough. There is a role for observational research, such as in situations where ethics, cost, and practicality do not allow for a randomized controlled trial. But randomized controlled trials have largely put to rest the purported health benefits of over-the-counter fish oils, omega-3s, and vitamin D.
3. The Main Course — Absolute vs Relative Risk
When you get to the main course, all eyes will now be on you. You will almost certainly be called upon to pronounce on the harms or benefits of red meat consumption. Begin by regaling your guests with a little trivia. Ask them if they know the definition of red meat and white meat. When someone says pork is white meat, you can reveal that “pork, the other white meat,” was a marketing slogan with no scientific underpinning. Now that everyone is lulled into a stupefied silence, tell them that red meat comes from mammals and white meat comes from birds. As they process this revelation, you can now launch into the deeply mathematical concept of absolute vs relative risk.
Many etiquette books will caution against bringing up math at a dinner party. These books are wrong. Everyone finds math interesting if they are primed properly. For example, you can point to a study claiming that berries reduce cardiovascular risk in women. Even if true — and there is reason to be cautious, given the observational nature of the research — we need to understand what the authors meant by a 32% risk reduction. (Side note: It was a reduction in hazard, with a hazard ratio of 0.68 (95% CI, 0.49-0.96), but we won’t dwell on the difference between hazard ratios and risk ratios right now.)
This relative risk reduction has to be interpreted carefully. The authors divided the population into quintiles based on their consumption of anthocyanins (the antioxidant in blueberries and strawberries) and compared the bottom fifth (average consumption, 2.5 mg/d) with the top fifth (average consumption, 25 mg/d). The bottom quintile had 126 myocardial infarctions (MIs) over 324,793 patient-years compared with 59 MIs over 332,143 patient-years. Some quick math shows an approximate reduction from 39 to 18 MIs per 100,000 patient-years. Or to put it another way, you must get 4762 women to increase their berry consumption 10-fold for 1 year to prevent one heart attack. Feel free to show people how you calculated this number. They will be impressed by your head for numbers. It is nothing more than 39 minus 18, divided by 100,000, to get the absolute risk reduction. Take the reciprocal of this (ie, 1 divided by this number) to get the number needed to treat.
Describing risks in absolute terms or using number needed to treat (or harm) can help conceptualize statistics that are sometimes hard to wrap your head around.
4. Dessert — Funding
By the time the coffee is served, everyone will be hanging on to your every word. This is as it should be, and you should not be afraid of your newfound power and influence.
Dessert will probably involve some form of chocolate, possibly in cake format. (Anyone who serves fruit as dessert is not someone you should associate with.) Take the opportunity to tell your follow diners that chocolate is not actually good for you and will not boost brain performance.
The health benefits of chocolate are often repeated but rarely scrutinized. In fact, much of the scientific research purporting to show that chocolate is good for you did not actually study chocolate. It usually involved a cocoa bean extract because the chocolate manufacturing process destroys the supposedly health-promoting antioxidants in the cocoa bean. It is true that dark chocolate has more antioxidants than milk chocolate, and that the addition of milk to chocolate further inactivates the potentially healthy antioxidants. But the amount of sugar and fat that has to be added to chocolate to make it palatable precludes any serious consideration about health benefits. Dark chocolate may have less fat and sugar than milk chocolate, but it still has a lot.
But even the cocoa bean extract doesn’t seem to do much for your heart or your brain. The long-awaited COSMOS study was published with surprisingly little fanfare. The largest randomized controlled trial of chocolate (or rather cocoa bean extract) was supposed to settle the issue definitively.
COSMOS showed no cardiovascular or neurocognitive benefit to the cocoa bean extract. But the health halo of chocolate continues to be bolstered by many studies funded by chocolate manufacturers.
We are appropriately critical of the pharmaceutical industry’s involvement in drug research. However, we should not forget that any private entity is prone to the same self-interest regardless of its product’s tastiness. How many of you knew that there was an avocado lobby funding research? No matter how many industry-funded observational studies using surrogate endpoints are out there telling you that chocolate is healthy, a randomized trial with hard clinical endpoints such as COSMOS should generally win the day.
The Final Goodbyes — Summarizing Your Case
As the party slowly winds down and everyone is saddened that you will soon take your leave, synthesize everything you have taught them over the evening. Like movies, not all studies are good. Some are just bad. They can be prone to reverse causation or confounding, and they may report relative risks when absolute risks would be more telling. Reading research studies critically is essential for separating the wheat from the chaff. With the knowledge you have now imparted to your friends, they will be much better consumers of medical news, especially when it comes to food.
And they will no doubt thank you for it by never inviting you to another dinner party!
Labos, a cardiologist at Hôpital, Notre-Dame, Montreal, Quebec, Canada, has disclosed no relevant financial relationships. He has a degree in epidemiology.
A version of this article appeared on Medscape.com.
You’re invited to a dinner party but you struggle to make small talk. Do not worry; that will invariably crop up over cocktails. Because all journalism has been reduced to listicles, here are four ways to seem clever at dinner parties.
1. The Predinner Cocktails: A Lesson in Reverse Causation
Wine connoisseurs sniff, swirl, and gently swish the wine in their mouths before spitting out and cleansing their palates to better appreciate the subtlety of each vintage. If you’re not an oenophile, no matter. Whenever somebody claims that moderate amounts of alcohol are good for your heart, this is your moment to pounce. Interject yourself in the conversation and tell everybody about reverse causation.
Reverse causation, also known as protopathic bias, involves misinterpreting the directionality of an association. You assume that X leads to Y, when in fact Y leads to X. Temporal paradoxes are useful plot devices in science fiction movies, but they have no place in medical research. In our bland world, cause must precede effect. As such, smoking leads to lung cancer; lung cancer doesn’t make you smoke more.
But with alcohol, directionality is less obvious. Many studies of alcohol and cardiovascular disease have demonstrated a U-shaped association, with risk being lowest among those who drink moderate amounts of alcohol (usually one to two drinks per day) and higher in those who drink more and also those who drink very little.
But one must ask why some people drink little or no alcohol. There is an important difference between former drinkers and never drinkers. Former drinkers cut back for a reason. More likely than not, the reason for this newfound sobriety was medical. A new cancer diagnosis, the emergence of atrial fibrillation, the development of diabetes, or rising blood pressure are all good reasons to reduce or eliminate alcohol. A cross-sectional study will fail to capture that alcohol consumption changes over time — people who now don’t drink may have imbibed alcohol in years past. It was not abstinence that led to an increased risk for heart disease; it was the increased risk for heart disease that led to abstinence.
You see the same phenomenon with the so-called obesity paradox. The idea that being a little overweight is good for you may appeal when you no longer fit into last year’s pants. But people who are underweight are so for a reason. Malnutrition, cachexia from cancer, or some other cause is almost certainly driving up the risk at the left-hand side of the U-shaped curve that makes the middle part seem better than it actually is.
Food consumption changes over time. A cross-sectional survey at one point in time cannot accurately capture past habits and distant exposures, especially for diseases such as heart disease and cancer that develop slowly over time. Studies on alcohol that try to overcome these shortcomings by eliminating former drinkers, or by using Mendelian randomization to better account for past exposure, do not show a cardiovascular benefit for moderate red wine drinking.
2. The Hors D’oeuvres — The Importance of RCTs
Now that you have made yourself the center of attention, it is time to cement your newfound reputation as a font of scientific knowledge. Most self-respecting hosts will serve smoked salmon as an amuse-bouche before the main meal. When someone mentions the health benefits of fish oils, you should take the opportunity to teach them about confounding.
Fish, especially cold-water fish from northern climates, have relatively high amounts of omega-3 fatty acids. Despite the plethora of observational studies suggesting a cardiovascular benefit, it’s now relatively clear that fish oil or omega-3 supplements have no medical benefit.
This will probably come as a shock to the worried well, but many studies, including VITAL and ASCEND, have demonstrated no cardiovascular or cancer benefit to supplementation with omega-3s. The reason is straightforward and explains why hormone replacement therapy, vitamin D, and myriad purported game-changers never panned out. Confounding is hard to overcome in observational research.
Prior to the publication of the Women’s Health Initiative (WHI) Study, hormone replacement therapy was routinely prescribed to postmenopausal women because numerous observational studies suggested a cardiovascular benefit. But with the publication of the WHI study, it became clear that much of that “benefit” was due to confounding. The women choosing to take hormones were more health conscious at baseline and healthier overall.
A similar phenomenon occurred during COVID. Patients with low serum vitamin D levels had worse outcomes, prompting many to suggest vitamin D supplementation as a possible treatment. Trials did not support the intervention because we’d overlooked the obvious. People with vitamin D deficiency have underlying health problems that contribute to the vitamin D deficiency. They are probably older, frailer, possibly with a poorer diet. No amount of statistical adjustment can account for all those differences, and some degree of residual confounding will always persist.
The only way to overcome confounding is with randomization. When patients are randomly assigned to one group or another, their baseline differences largely balance out if the randomization was performed properly and the groups were large enough. There is a role for observational research, such as in situations where ethics, cost, and practicality do not allow for a randomized controlled trial. But randomized controlled trials have largely put to rest the purported health benefits of over-the-counter fish oils, omega-3s, and vitamin D.
3. The Main Course — Absolute vs Relative Risk
When you get to the main course, all eyes will now be on you. You will almost certainly be called upon to pronounce on the harms or benefits of red meat consumption. Begin by regaling your guests with a little trivia. Ask them if they know the definition of red meat and white meat. When someone says pork is white meat, you can reveal that “pork, the other white meat,” was a marketing slogan with no scientific underpinning. Now that everyone is lulled into a stupefied silence, tell them that red meat comes from mammals and white meat comes from birds. As they process this revelation, you can now launch into the deeply mathematical concept of absolute vs relative risk.
Many etiquette books will caution against bringing up math at a dinner party. These books are wrong. Everyone finds math interesting if they are primed properly. For example, you can point to a study claiming that berries reduce cardiovascular risk in women. Even if true — and there is reason to be cautious, given the observational nature of the research — we need to understand what the authors meant by a 32% risk reduction. (Side note: It was a reduction in hazard, with a hazard ratio of 0.68 (95% CI, 0.49-0.96), but we won’t dwell on the difference between hazard ratios and risk ratios right now.)
This relative risk reduction has to be interpreted carefully. The authors divided the population into quintiles based on their consumption of anthocyanins (the antioxidant in blueberries and strawberries) and compared the bottom fifth (average consumption, 2.5 mg/d) with the top fifth (average consumption, 25 mg/d). The bottom quintile had 126 myocardial infarctions (MIs) over 324,793 patient-years compared with 59 MIs over 332,143 patient-years. Some quick math shows an approximate reduction from 39 to 18 MIs per 100,000 patient-years. Or to put it another way, you must get 4762 women to increase their berry consumption 10-fold for 1 year to prevent one heart attack. Feel free to show people how you calculated this number. They will be impressed by your head for numbers. It is nothing more than 39 minus 18, divided by 100,000, to get the absolute risk reduction. Take the reciprocal of this (ie, 1 divided by this number) to get the number needed to treat.
Describing risks in absolute terms or using number needed to treat (or harm) can help conceptualize statistics that are sometimes hard to wrap your head around.
4. Dessert — Funding
By the time the coffee is served, everyone will be hanging on to your every word. This is as it should be, and you should not be afraid of your newfound power and influence.
Dessert will probably involve some form of chocolate, possibly in cake format. (Anyone who serves fruit as dessert is not someone you should associate with.) Take the opportunity to tell your follow diners that chocolate is not actually good for you and will not boost brain performance.
The health benefits of chocolate are often repeated but rarely scrutinized. In fact, much of the scientific research purporting to show that chocolate is good for you did not actually study chocolate. It usually involved a cocoa bean extract because the chocolate manufacturing process destroys the supposedly health-promoting antioxidants in the cocoa bean. It is true that dark chocolate has more antioxidants than milk chocolate, and that the addition of milk to chocolate further inactivates the potentially healthy antioxidants. But the amount of sugar and fat that has to be added to chocolate to make it palatable precludes any serious consideration about health benefits. Dark chocolate may have less fat and sugar than milk chocolate, but it still has a lot.
But even the cocoa bean extract doesn’t seem to do much for your heart or your brain. The long-awaited COSMOS study was published with surprisingly little fanfare. The largest randomized controlled trial of chocolate (or rather cocoa bean extract) was supposed to settle the issue definitively.
COSMOS showed no cardiovascular or neurocognitive benefit to the cocoa bean extract. But the health halo of chocolate continues to be bolstered by many studies funded by chocolate manufacturers.
We are appropriately critical of the pharmaceutical industry’s involvement in drug research. However, we should not forget that any private entity is prone to the same self-interest regardless of its product’s tastiness. How many of you knew that there was an avocado lobby funding research? No matter how many industry-funded observational studies using surrogate endpoints are out there telling you that chocolate is healthy, a randomized trial with hard clinical endpoints such as COSMOS should generally win the day.
The Final Goodbyes — Summarizing Your Case
As the party slowly winds down and everyone is saddened that you will soon take your leave, synthesize everything you have taught them over the evening. Like movies, not all studies are good. Some are just bad. They can be prone to reverse causation or confounding, and they may report relative risks when absolute risks would be more telling. Reading research studies critically is essential for separating the wheat from the chaff. With the knowledge you have now imparted to your friends, they will be much better consumers of medical news, especially when it comes to food.
And they will no doubt thank you for it by never inviting you to another dinner party!
Labos, a cardiologist at Hôpital, Notre-Dame, Montreal, Quebec, Canada, has disclosed no relevant financial relationships. He has a degree in epidemiology.
A version of this article appeared on Medscape.com.
Transitioning from Employment in Academia to Private Practice
After more than 10 years of serving in a large academic medical center in Chicago, Illinois, that was part of a national health care system, the decision to transition into private practice wasn’t one I made lightly.
Having built a rewarding career and spent over a quarter of my life in an academic medical center and a national health system, the move to starting an independent practice from scratch was both exciting and daunting. The notion of leaving behind the structure, resources, and safety of the large health system was unsettling. However, as the landscape of health care continues to evolve, with worsening large structural problems within the U.S. health care system, I realized that starting an independent gastroenterology practice — focused on trying to fix some of these large-scale problems from the start — would not only align with my professional goals but also provide the personal satisfaction I had failed to find.
As I reflect on my journey, there are a few key lessons I learned from making this leap — lessons that helped me transition from a highly structured employed physician environment to leading a thriving independent practice focused on redesigning gastroenterology care from scratch.
Lesson 1: Autonomy Opens the Door to Innovation
One of the primary reasons I left the employed physician setting was to gain greater control over my clinical practice and decision-making processes.
In a national health care system, the goal of standardization often dictates not only clinical care, but many “back end” aspects of the entire health care experience. We often see the things that are more visible, such as what supplies/equipment you use, how your patient appointments are scheduled, how many support staff members are assigned to help your practice, what electronic health record system you use, and how shared resources (like GI lab block time or anesthesia teams) are allocated.
However, this also impacts things we don’t usually see, such as what fees are billed for care you are providing (like facility fees), communication systems that your patients need to navigate for help, human resource systems you use, and retirement/health benefits you and your other team members receive.
Standardization has two adverse consequences: 1) it does not allow for personalization and as a result, 2) it suppresses innovation. Standard protocols can streamline processes, but they sometimes fail to account for the nuanced differences between patients, such as genetic factors, unique medical histories, or responses/failures to prior treatments. This rigidity can stifle innovation, as physicians are often bound by guidelines that may not reflect the latest advancements or allow for creative, individualized approaches to care. In the long term, an overemphasis on standardization risks turning health care into a one-size-fits-all model, undermining the potential for breakthroughs.
The transition was challenging at first, as we needed to engage our entire new practice with a different mindset now that many of us had autonomy for the first time. Instead of everyone just practicing health care the way they had done before, we took a page from Elon Musk and challenged every member of the team to ask three questions about everything they do on a daily basis:
- Is what I am doing helping a patient get healthy? (Question every requirement)
- If not, do I still need to do this? (Delete any part of the process you can)
- If so, how can I make this easier, faster, or automated? (Simplify and optimize, accelerate cycle time, and automate)
The freedom to innovate is a hallmark of independent practice. Embracing innovation in every aspect of the practice has been the most critical lesson of this journey.
Lesson 2: Financial Stewardship is Critical for Sustainability
Running an independent practice is not just about medicine — it’s also about managing a business.
This was a stark shift from the large academic health systems, where financial decisions were handled by the “administration.” In my new role as a business owner, understanding the financial aspects of health care was crucial for success. The cost of what patients pay for health care in the United States (either directly in deductibles and coinsurance or indirectly through insurance premiums) is unsustainably high. However, inflation continues to cause substantial increases in almost all the costs of delivering care: medical supplies, salaries, benefits, IT costs, etc. It was critical to develop a financial plan that accounted for these two macro-economic trends, and ideally helped solve for both. In our case, delivering high quality care with a lower cost to patients and payers.
We started by reevaluating our relationship with payers. Whereas being part of a large academic health system, we are often taught to look at payers as the adversary; as an independent practice looking to redesign the health care experience, it was critical for us to look to the payers as a partner in this journey. Understanding payer expectations and structuring contracts that aligned with shared goals of reducing total health care costs for patients was one of the foundations of our financial plan.
Offering office-based endoscopy was one innovation we implemented to significantly impact both patient affordability and practice revenue. By performing procedures like colonoscopies and upper endoscopies in an office setting rather than a hospital or ambulatory surgery center, we eliminated facility fees, which are often a significant part of the total cost of care. This directly lowers out-of-pocket expenses for patients and reduces the overall financial burden on insurance companies. At the same time, it allows the practice to capture more of the revenue from these procedures, without the overhead costs associated with larger facilities. This model creates a win-win situation: patients save money while receiving the same quality of care, and the practice experiences an increase in profitability and autonomy in managing its services.
Lesson 3: Collaborative Care and Multidisciplinary Teams Can Exist Anywhere
One aspect I deeply valued in academia was the collaborative environment — having specialists across disciplines work together on challenging cases. In private practice, I was concerned that I would lose this collegial atmosphere. However, I quickly learned that building a robust network of multidisciplinary collaborators was achievable in independent practice, just like it was in a large health system.
In our practice, we established close relationships with primary care physicians, surgeons, advanced practice providers, dietitians, behavioral health specialists, and others. These partnerships were not just referral networks but integrated care teams where communication and shared decision-making were prioritized. By fostering collaboration, we could offer patients comprehensive care that addressed their physical, psychological, and nutritional needs.
For example, managing patients with chronic conditions like inflammatory bowel disease, cirrhosis, or obesity requires more than just prescribing medications. It involves regular monitoring, dietary adjustments, psychological support, and in some cases, surgical intervention. In an academic setting, coordinating this level of care can be cumbersome due to institutional barriers and siloed departments. In our practice, some of these relationships are achieved through partnerships with other like-minded practices. In other situations, team members of other disciplines are employed directly by our practice. Being in an independent practice allowed us the flexibility to prioritize working with the right team members first, and then structuring the relationship model second.
Lesson 4: Technology Is a Vital Tool in Redesigning Health Care
When I worked in a large academic health system, technology was often seen as an administrative burden rather than a clinical asset. Electronic health records (EHR) and a lot of the other IT systems that health care workers and patients interacted with on a regular basis were viewed as a barrier to care or a cause of time burdens instead of as tools to make health care easier. As we built our new practice from scratch, it was critical that we had an IT infrastructure that aligned with our core goals: simplify and automate the health care experience for everyone.
For our practice, we didn’t try to re-invent the wheel. Instead we copied from other industries who had already figured out a great solution for a problem we had. We wanted our patients to have a great customer service experience when interacting with our practice for scheduling, questions, refills, etc. So we implemented a unified communication system that some Fortune 100 companies, with perennial high scores for customer service, used. We wanted a great human resource system that would streamline the administrative time it would take to handle all HR needs for our practice. So we implemented an HR information system that had the best ratings for automation and integration with other business systems. At every point in the process, we reminded ourselves to focus on simplification and automation for every user of the system.
Conclusion: A Rewarding Transition
The lessons I’ve learned along the way — embracing autonomy, understanding financial stewardship, fostering collaboration, and leveraging technology — have helped me work toward a better total health care experience for the community.
This journey has also been deeply fulfilling on a personal level. It has allowed me to build stronger relationships with my patients, focus on long-term health outcomes, and create a practice where innovation and quality truly matter. While the challenges of running a private practice are real, the rewards — both for me and my patients — are immeasurable. If I had to do it all over again, I wouldn’t hesitate for a moment. If anything, I should have done it earlier.
Dr. Gupta is Managing Partner at Midwest Digestive Health & Nutrition, in Des Plaines, Illinois. He has reported no conflicts of interest in relation to this article.
After more than 10 years of serving in a large academic medical center in Chicago, Illinois, that was part of a national health care system, the decision to transition into private practice wasn’t one I made lightly.
Having built a rewarding career and spent over a quarter of my life in an academic medical center and a national health system, the move to starting an independent practice from scratch was both exciting and daunting. The notion of leaving behind the structure, resources, and safety of the large health system was unsettling. However, as the landscape of health care continues to evolve, with worsening large structural problems within the U.S. health care system, I realized that starting an independent gastroenterology practice — focused on trying to fix some of these large-scale problems from the start — would not only align with my professional goals but also provide the personal satisfaction I had failed to find.
As I reflect on my journey, there are a few key lessons I learned from making this leap — lessons that helped me transition from a highly structured employed physician environment to leading a thriving independent practice focused on redesigning gastroenterology care from scratch.
Lesson 1: Autonomy Opens the Door to Innovation
One of the primary reasons I left the employed physician setting was to gain greater control over my clinical practice and decision-making processes.
In a national health care system, the goal of standardization often dictates not only clinical care, but many “back end” aspects of the entire health care experience. We often see the things that are more visible, such as what supplies/equipment you use, how your patient appointments are scheduled, how many support staff members are assigned to help your practice, what electronic health record system you use, and how shared resources (like GI lab block time or anesthesia teams) are allocated.
However, this also impacts things we don’t usually see, such as what fees are billed for care you are providing (like facility fees), communication systems that your patients need to navigate for help, human resource systems you use, and retirement/health benefits you and your other team members receive.
Standardization has two adverse consequences: 1) it does not allow for personalization and as a result, 2) it suppresses innovation. Standard protocols can streamline processes, but they sometimes fail to account for the nuanced differences between patients, such as genetic factors, unique medical histories, or responses/failures to prior treatments. This rigidity can stifle innovation, as physicians are often bound by guidelines that may not reflect the latest advancements or allow for creative, individualized approaches to care. In the long term, an overemphasis on standardization risks turning health care into a one-size-fits-all model, undermining the potential for breakthroughs.
The transition was challenging at first, as we needed to engage our entire new practice with a different mindset now that many of us had autonomy for the first time. Instead of everyone just practicing health care the way they had done before, we took a page from Elon Musk and challenged every member of the team to ask three questions about everything they do on a daily basis:
- Is what I am doing helping a patient get healthy? (Question every requirement)
- If not, do I still need to do this? (Delete any part of the process you can)
- If so, how can I make this easier, faster, or automated? (Simplify and optimize, accelerate cycle time, and automate)
The freedom to innovate is a hallmark of independent practice. Embracing innovation in every aspect of the practice has been the most critical lesson of this journey.
Lesson 2: Financial Stewardship is Critical for Sustainability
Running an independent practice is not just about medicine — it’s also about managing a business.
This was a stark shift from the large academic health systems, where financial decisions were handled by the “administration.” In my new role as a business owner, understanding the financial aspects of health care was crucial for success. The cost of what patients pay for health care in the United States (either directly in deductibles and coinsurance or indirectly through insurance premiums) is unsustainably high. However, inflation continues to cause substantial increases in almost all the costs of delivering care: medical supplies, salaries, benefits, IT costs, etc. It was critical to develop a financial plan that accounted for these two macro-economic trends, and ideally helped solve for both. In our case, delivering high quality care with a lower cost to patients and payers.
We started by reevaluating our relationship with payers. Whereas being part of a large academic health system, we are often taught to look at payers as the adversary; as an independent practice looking to redesign the health care experience, it was critical for us to look to the payers as a partner in this journey. Understanding payer expectations and structuring contracts that aligned with shared goals of reducing total health care costs for patients was one of the foundations of our financial plan.
Offering office-based endoscopy was one innovation we implemented to significantly impact both patient affordability and practice revenue. By performing procedures like colonoscopies and upper endoscopies in an office setting rather than a hospital or ambulatory surgery center, we eliminated facility fees, which are often a significant part of the total cost of care. This directly lowers out-of-pocket expenses for patients and reduces the overall financial burden on insurance companies. At the same time, it allows the practice to capture more of the revenue from these procedures, without the overhead costs associated with larger facilities. This model creates a win-win situation: patients save money while receiving the same quality of care, and the practice experiences an increase in profitability and autonomy in managing its services.
Lesson 3: Collaborative Care and Multidisciplinary Teams Can Exist Anywhere
One aspect I deeply valued in academia was the collaborative environment — having specialists across disciplines work together on challenging cases. In private practice, I was concerned that I would lose this collegial atmosphere. However, I quickly learned that building a robust network of multidisciplinary collaborators was achievable in independent practice, just like it was in a large health system.
In our practice, we established close relationships with primary care physicians, surgeons, advanced practice providers, dietitians, behavioral health specialists, and others. These partnerships were not just referral networks but integrated care teams where communication and shared decision-making were prioritized. By fostering collaboration, we could offer patients comprehensive care that addressed their physical, psychological, and nutritional needs.
For example, managing patients with chronic conditions like inflammatory bowel disease, cirrhosis, or obesity requires more than just prescribing medications. It involves regular monitoring, dietary adjustments, psychological support, and in some cases, surgical intervention. In an academic setting, coordinating this level of care can be cumbersome due to institutional barriers and siloed departments. In our practice, some of these relationships are achieved through partnerships with other like-minded practices. In other situations, team members of other disciplines are employed directly by our practice. Being in an independent practice allowed us the flexibility to prioritize working with the right team members first, and then structuring the relationship model second.
Lesson 4: Technology Is a Vital Tool in Redesigning Health Care
When I worked in a large academic health system, technology was often seen as an administrative burden rather than a clinical asset. Electronic health records (EHR) and a lot of the other IT systems that health care workers and patients interacted with on a regular basis were viewed as a barrier to care or a cause of time burdens instead of as tools to make health care easier. As we built our new practice from scratch, it was critical that we had an IT infrastructure that aligned with our core goals: simplify and automate the health care experience for everyone.
For our practice, we didn’t try to re-invent the wheel. Instead we copied from other industries who had already figured out a great solution for a problem we had. We wanted our patients to have a great customer service experience when interacting with our practice for scheduling, questions, refills, etc. So we implemented a unified communication system that some Fortune 100 companies, with perennial high scores for customer service, used. We wanted a great human resource system that would streamline the administrative time it would take to handle all HR needs for our practice. So we implemented an HR information system that had the best ratings for automation and integration with other business systems. At every point in the process, we reminded ourselves to focus on simplification and automation for every user of the system.
Conclusion: A Rewarding Transition
The lessons I’ve learned along the way — embracing autonomy, understanding financial stewardship, fostering collaboration, and leveraging technology — have helped me work toward a better total health care experience for the community.
This journey has also been deeply fulfilling on a personal level. It has allowed me to build stronger relationships with my patients, focus on long-term health outcomes, and create a practice where innovation and quality truly matter. While the challenges of running a private practice are real, the rewards — both for me and my patients — are immeasurable. If I had to do it all over again, I wouldn’t hesitate for a moment. If anything, I should have done it earlier.
Dr. Gupta is Managing Partner at Midwest Digestive Health & Nutrition, in Des Plaines, Illinois. He has reported no conflicts of interest in relation to this article.
After more than 10 years of serving in a large academic medical center in Chicago, Illinois, that was part of a national health care system, the decision to transition into private practice wasn’t one I made lightly.
Having built a rewarding career and spent over a quarter of my life in an academic medical center and a national health system, the move to starting an independent practice from scratch was both exciting and daunting. The notion of leaving behind the structure, resources, and safety of the large health system was unsettling. However, as the landscape of health care continues to evolve, with worsening large structural problems within the U.S. health care system, I realized that starting an independent gastroenterology practice — focused on trying to fix some of these large-scale problems from the start — would not only align with my professional goals but also provide the personal satisfaction I had failed to find.
As I reflect on my journey, there are a few key lessons I learned from making this leap — lessons that helped me transition from a highly structured employed physician environment to leading a thriving independent practice focused on redesigning gastroenterology care from scratch.
Lesson 1: Autonomy Opens the Door to Innovation
One of the primary reasons I left the employed physician setting was to gain greater control over my clinical practice and decision-making processes.
In a national health care system, the goal of standardization often dictates not only clinical care, but many “back end” aspects of the entire health care experience. We often see the things that are more visible, such as what supplies/equipment you use, how your patient appointments are scheduled, how many support staff members are assigned to help your practice, what electronic health record system you use, and how shared resources (like GI lab block time or anesthesia teams) are allocated.
However, this also impacts things we don’t usually see, such as what fees are billed for care you are providing (like facility fees), communication systems that your patients need to navigate for help, human resource systems you use, and retirement/health benefits you and your other team members receive.
Standardization has two adverse consequences: 1) it does not allow for personalization and as a result, 2) it suppresses innovation. Standard protocols can streamline processes, but they sometimes fail to account for the nuanced differences between patients, such as genetic factors, unique medical histories, or responses/failures to prior treatments. This rigidity can stifle innovation, as physicians are often bound by guidelines that may not reflect the latest advancements or allow for creative, individualized approaches to care. In the long term, an overemphasis on standardization risks turning health care into a one-size-fits-all model, undermining the potential for breakthroughs.
The transition was challenging at first, as we needed to engage our entire new practice with a different mindset now that many of us had autonomy for the first time. Instead of everyone just practicing health care the way they had done before, we took a page from Elon Musk and challenged every member of the team to ask three questions about everything they do on a daily basis:
- Is what I am doing helping a patient get healthy? (Question every requirement)
- If not, do I still need to do this? (Delete any part of the process you can)
- If so, how can I make this easier, faster, or automated? (Simplify and optimize, accelerate cycle time, and automate)
The freedom to innovate is a hallmark of independent practice. Embracing innovation in every aspect of the practice has been the most critical lesson of this journey.
Lesson 2: Financial Stewardship is Critical for Sustainability
Running an independent practice is not just about medicine — it’s also about managing a business.
This was a stark shift from the large academic health systems, where financial decisions were handled by the “administration.” In my new role as a business owner, understanding the financial aspects of health care was crucial for success. The cost of what patients pay for health care in the United States (either directly in deductibles and coinsurance or indirectly through insurance premiums) is unsustainably high. However, inflation continues to cause substantial increases in almost all the costs of delivering care: medical supplies, salaries, benefits, IT costs, etc. It was critical to develop a financial plan that accounted for these two macro-economic trends, and ideally helped solve for both. In our case, delivering high quality care with a lower cost to patients and payers.
We started by reevaluating our relationship with payers. Whereas being part of a large academic health system, we are often taught to look at payers as the adversary; as an independent practice looking to redesign the health care experience, it was critical for us to look to the payers as a partner in this journey. Understanding payer expectations and structuring contracts that aligned with shared goals of reducing total health care costs for patients was one of the foundations of our financial plan.
Offering office-based endoscopy was one innovation we implemented to significantly impact both patient affordability and practice revenue. By performing procedures like colonoscopies and upper endoscopies in an office setting rather than a hospital or ambulatory surgery center, we eliminated facility fees, which are often a significant part of the total cost of care. This directly lowers out-of-pocket expenses for patients and reduces the overall financial burden on insurance companies. At the same time, it allows the practice to capture more of the revenue from these procedures, without the overhead costs associated with larger facilities. This model creates a win-win situation: patients save money while receiving the same quality of care, and the practice experiences an increase in profitability and autonomy in managing its services.
Lesson 3: Collaborative Care and Multidisciplinary Teams Can Exist Anywhere
One aspect I deeply valued in academia was the collaborative environment — having specialists across disciplines work together on challenging cases. In private practice, I was concerned that I would lose this collegial atmosphere. However, I quickly learned that building a robust network of multidisciplinary collaborators was achievable in independent practice, just like it was in a large health system.
In our practice, we established close relationships with primary care physicians, surgeons, advanced practice providers, dietitians, behavioral health specialists, and others. These partnerships were not just referral networks but integrated care teams where communication and shared decision-making were prioritized. By fostering collaboration, we could offer patients comprehensive care that addressed their physical, psychological, and nutritional needs.
For example, managing patients with chronic conditions like inflammatory bowel disease, cirrhosis, or obesity requires more than just prescribing medications. It involves regular monitoring, dietary adjustments, psychological support, and in some cases, surgical intervention. In an academic setting, coordinating this level of care can be cumbersome due to institutional barriers and siloed departments. In our practice, some of these relationships are achieved through partnerships with other like-minded practices. In other situations, team members of other disciplines are employed directly by our practice. Being in an independent practice allowed us the flexibility to prioritize working with the right team members first, and then structuring the relationship model second.
Lesson 4: Technology Is a Vital Tool in Redesigning Health Care
When I worked in a large academic health system, technology was often seen as an administrative burden rather than a clinical asset. Electronic health records (EHR) and a lot of the other IT systems that health care workers and patients interacted with on a regular basis were viewed as a barrier to care or a cause of time burdens instead of as tools to make health care easier. As we built our new practice from scratch, it was critical that we had an IT infrastructure that aligned with our core goals: simplify and automate the health care experience for everyone.
For our practice, we didn’t try to re-invent the wheel. Instead we copied from other industries who had already figured out a great solution for a problem we had. We wanted our patients to have a great customer service experience when interacting with our practice for scheduling, questions, refills, etc. So we implemented a unified communication system that some Fortune 100 companies, with perennial high scores for customer service, used. We wanted a great human resource system that would streamline the administrative time it would take to handle all HR needs for our practice. So we implemented an HR information system that had the best ratings for automation and integration with other business systems. At every point in the process, we reminded ourselves to focus on simplification and automation for every user of the system.
Conclusion: A Rewarding Transition
The lessons I’ve learned along the way — embracing autonomy, understanding financial stewardship, fostering collaboration, and leveraging technology — have helped me work toward a better total health care experience for the community.
This journey has also been deeply fulfilling on a personal level. It has allowed me to build stronger relationships with my patients, focus on long-term health outcomes, and create a practice where innovation and quality truly matter. While the challenges of running a private practice are real, the rewards — both for me and my patients — are immeasurable. If I had to do it all over again, I wouldn’t hesitate for a moment. If anything, I should have done it earlier.
Dr. Gupta is Managing Partner at Midwest Digestive Health & Nutrition, in Des Plaines, Illinois. He has reported no conflicts of interest in relation to this article.
A Child’s Picky Eating: Normal Phase or Health Concern?
PARIS — “My child is a poor eater” is a complaint frequently heard during medical consultations. Such concerns are often unjustified but a source of much parental frustration.
Marc Bellaïche, MD, a pediatrician at Robert-Debré Hospital in Paris, addressed this issue at France’s annual general medicine conference (JNMG 2024). His presentation focused on distinguishing between parental perception, typical childhood behaviors, and feeding issues that require intervention.
In assessing parental worries, tools such as The Montreal Children’s Hospital Feeding Scale for children aged 6 months to 6 years and the Baby Eating Behavior Questionnaire for those under 6 months can help identify and monitor feeding issues. Observing the child eat, when possible, is also valuable.
Key Phases and Development
Bellaïche focused on children under 6 years, as they frequently experience feeding challenges during critical development phases, such as weaning or when the child is able to sit up.
A phase of neophilia (interest in new foods) typically occurs before 12 months, followed by a phase of neophobia (fear of new foods) between ages 1 and 3 years. This neophobia is a normal part of neuropsychological, sensory, and taste development and can persist if a key developmental moment is marked by a choking incident, mealtime stress, or forced feeding. “Challenges differ between a difficult 3-year-old and a 6- or 7-year-old who still refuses new foods,” he explained.
Parental Pressure and Nutritional Balance
Nutritional balance is essential, but “parental pressure is often too high.” Parents worry because they see food as a “nutraceutical.” Bellaïche recommended defusing anxiety by keeping mealtimes calm, allowing the child to eat at their pace, avoiding force-feeding, keeping meals brief, and avoiding snacks. While “it’s important to stay vigilant — as it’s incorrect to assume a child won’t let themselves starve — most cases can be managed in general practice through parental guidance, empathy, and a positive approach.”
Monitoring growth and weight curves is crucial, with the Kanawati index (ratio of arm circumference to head circumference) being a reliable indicator for specialist referral if < 0.31. A varied diet is important for nutritional balance; when this isn’t achieved, continued consumption of toddler formula after age 3 can prevent iron and calcium deficiencies.
When eating difficulties are documented, healthcare providers should investigate for underlying organic, digestive, or extra-digestive diseases (neurologic, cardiac, renal, etc.). “It’s best not to hastily diagnose cow’s milk protein allergy,” Bellaïche advised, as cases are relatively rare and unnecessarily eliminating milk can complicate a child’s relationship with food. Similarly, gastroesophageal reflux disease should be objectively diagnosed to avoid unnecessary proton pump inhibitor treatment and associated side effects.
For children with low birth weight, mild congenital heart disease, or suggestive dysmorphology, consider evaluating for a genetic syndrome.
Avoidant/Restrictive Food Intake Disorder (ARFID)
ARFID is marked by a lack of interest in food and avoidance due to sensory characteristics. Often observed in anxious children, ARFID is diagnosed in approximately 20% of children with autism spectrum disorder, where food selectivity is prevalent. This condition can hinder a child’s development and may necessitate nutritional supplementation.
Case Profiles in Eating Issues
Bellaïche outlined three typical cases among children considered “picky eaters”:
- The small eater: Often near the lower growth curve limits, this child “grazes and doesn’t sit still.” These children are usually active and have a family history of similar eating habits. Parents should encourage psychomotor activities, discourage snacks outside of mealtimes, and consider fun family picnics on the floor, offering a mezze-style variety of foods.
- The child with a history of trauma: Children with trauma (from intubation, nasogastric tubes, severe vomiting, forced feeding, or choking) may develop aversions requiring behavioral intervention.
- The child with high sensory sensitivity: This child dislikes getting the hands dirty, avoids mouthing objects, or resists certain textures, such as grass and sand. Gradual behavioral approaches with sensory play and visually appealing new foods can be beneficial. Guided self-led food exploration (baby-led weaning) may also help, though dairy intake is often needed to prevent deficiencies during this stage.
Finally, gastroesophageal reflux disease or constipation can contribute to appetite loss. Studies have shown that treating these issues can improve appetite in small eaters.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
PARIS — “My child is a poor eater” is a complaint frequently heard during medical consultations. Such concerns are often unjustified but a source of much parental frustration.
Marc Bellaïche, MD, a pediatrician at Robert-Debré Hospital in Paris, addressed this issue at France’s annual general medicine conference (JNMG 2024). His presentation focused on distinguishing between parental perception, typical childhood behaviors, and feeding issues that require intervention.
In assessing parental worries, tools such as The Montreal Children’s Hospital Feeding Scale for children aged 6 months to 6 years and the Baby Eating Behavior Questionnaire for those under 6 months can help identify and monitor feeding issues. Observing the child eat, when possible, is also valuable.
Key Phases and Development
Bellaïche focused on children under 6 years, as they frequently experience feeding challenges during critical development phases, such as weaning or when the child is able to sit up.
A phase of neophilia (interest in new foods) typically occurs before 12 months, followed by a phase of neophobia (fear of new foods) between ages 1 and 3 years. This neophobia is a normal part of neuropsychological, sensory, and taste development and can persist if a key developmental moment is marked by a choking incident, mealtime stress, or forced feeding. “Challenges differ between a difficult 3-year-old and a 6- or 7-year-old who still refuses new foods,” he explained.
Parental Pressure and Nutritional Balance
Nutritional balance is essential, but “parental pressure is often too high.” Parents worry because they see food as a “nutraceutical.” Bellaïche recommended defusing anxiety by keeping mealtimes calm, allowing the child to eat at their pace, avoiding force-feeding, keeping meals brief, and avoiding snacks. While “it’s important to stay vigilant — as it’s incorrect to assume a child won’t let themselves starve — most cases can be managed in general practice through parental guidance, empathy, and a positive approach.”
Monitoring growth and weight curves is crucial, with the Kanawati index (ratio of arm circumference to head circumference) being a reliable indicator for specialist referral if < 0.31. A varied diet is important for nutritional balance; when this isn’t achieved, continued consumption of toddler formula after age 3 can prevent iron and calcium deficiencies.
When eating difficulties are documented, healthcare providers should investigate for underlying organic, digestive, or extra-digestive diseases (neurologic, cardiac, renal, etc.). “It’s best not to hastily diagnose cow’s milk protein allergy,” Bellaïche advised, as cases are relatively rare and unnecessarily eliminating milk can complicate a child’s relationship with food. Similarly, gastroesophageal reflux disease should be objectively diagnosed to avoid unnecessary proton pump inhibitor treatment and associated side effects.
For children with low birth weight, mild congenital heart disease, or suggestive dysmorphology, consider evaluating for a genetic syndrome.
Avoidant/Restrictive Food Intake Disorder (ARFID)
ARFID is marked by a lack of interest in food and avoidance due to sensory characteristics. Often observed in anxious children, ARFID is diagnosed in approximately 20% of children with autism spectrum disorder, where food selectivity is prevalent. This condition can hinder a child’s development and may necessitate nutritional supplementation.
Case Profiles in Eating Issues
Bellaïche outlined three typical cases among children considered “picky eaters”:
- The small eater: Often near the lower growth curve limits, this child “grazes and doesn’t sit still.” These children are usually active and have a family history of similar eating habits. Parents should encourage psychomotor activities, discourage snacks outside of mealtimes, and consider fun family picnics on the floor, offering a mezze-style variety of foods.
- The child with a history of trauma: Children with trauma (from intubation, nasogastric tubes, severe vomiting, forced feeding, or choking) may develop aversions requiring behavioral intervention.
- The child with high sensory sensitivity: This child dislikes getting the hands dirty, avoids mouthing objects, or resists certain textures, such as grass and sand. Gradual behavioral approaches with sensory play and visually appealing new foods can be beneficial. Guided self-led food exploration (baby-led weaning) may also help, though dairy intake is often needed to prevent deficiencies during this stage.
Finally, gastroesophageal reflux disease or constipation can contribute to appetite loss. Studies have shown that treating these issues can improve appetite in small eaters.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
PARIS — “My child is a poor eater” is a complaint frequently heard during medical consultations. Such concerns are often unjustified but a source of much parental frustration.
Marc Bellaïche, MD, a pediatrician at Robert-Debré Hospital in Paris, addressed this issue at France’s annual general medicine conference (JNMG 2024). His presentation focused on distinguishing between parental perception, typical childhood behaviors, and feeding issues that require intervention.
In assessing parental worries, tools such as The Montreal Children’s Hospital Feeding Scale for children aged 6 months to 6 years and the Baby Eating Behavior Questionnaire for those under 6 months can help identify and monitor feeding issues. Observing the child eat, when possible, is also valuable.
Key Phases and Development
Bellaïche focused on children under 6 years, as they frequently experience feeding challenges during critical development phases, such as weaning or when the child is able to sit up.
A phase of neophilia (interest in new foods) typically occurs before 12 months, followed by a phase of neophobia (fear of new foods) between ages 1 and 3 years. This neophobia is a normal part of neuropsychological, sensory, and taste development and can persist if a key developmental moment is marked by a choking incident, mealtime stress, or forced feeding. “Challenges differ between a difficult 3-year-old and a 6- or 7-year-old who still refuses new foods,” he explained.
Parental Pressure and Nutritional Balance
Nutritional balance is essential, but “parental pressure is often too high.” Parents worry because they see food as a “nutraceutical.” Bellaïche recommended defusing anxiety by keeping mealtimes calm, allowing the child to eat at their pace, avoiding force-feeding, keeping meals brief, and avoiding snacks. While “it’s important to stay vigilant — as it’s incorrect to assume a child won’t let themselves starve — most cases can be managed in general practice through parental guidance, empathy, and a positive approach.”
Monitoring growth and weight curves is crucial, with the Kanawati index (ratio of arm circumference to head circumference) being a reliable indicator for specialist referral if < 0.31. A varied diet is important for nutritional balance; when this isn’t achieved, continued consumption of toddler formula after age 3 can prevent iron and calcium deficiencies.
When eating difficulties are documented, healthcare providers should investigate for underlying organic, digestive, or extra-digestive diseases (neurologic, cardiac, renal, etc.). “It’s best not to hastily diagnose cow’s milk protein allergy,” Bellaïche advised, as cases are relatively rare and unnecessarily eliminating milk can complicate a child’s relationship with food. Similarly, gastroesophageal reflux disease should be objectively diagnosed to avoid unnecessary proton pump inhibitor treatment and associated side effects.
For children with low birth weight, mild congenital heart disease, or suggestive dysmorphology, consider evaluating for a genetic syndrome.
Avoidant/Restrictive Food Intake Disorder (ARFID)
ARFID is marked by a lack of interest in food and avoidance due to sensory characteristics. Often observed in anxious children, ARFID is diagnosed in approximately 20% of children with autism spectrum disorder, where food selectivity is prevalent. This condition can hinder a child’s development and may necessitate nutritional supplementation.
Case Profiles in Eating Issues
Bellaïche outlined three typical cases among children considered “picky eaters”:
- The small eater: Often near the lower growth curve limits, this child “grazes and doesn’t sit still.” These children are usually active and have a family history of similar eating habits. Parents should encourage psychomotor activities, discourage snacks outside of mealtimes, and consider fun family picnics on the floor, offering a mezze-style variety of foods.
- The child with a history of trauma: Children with trauma (from intubation, nasogastric tubes, severe vomiting, forced feeding, or choking) may develop aversions requiring behavioral intervention.
- The child with high sensory sensitivity: This child dislikes getting the hands dirty, avoids mouthing objects, or resists certain textures, such as grass and sand. Gradual behavioral approaches with sensory play and visually appealing new foods can be beneficial. Guided self-led food exploration (baby-led weaning) may also help, though dairy intake is often needed to prevent deficiencies during this stage.
Finally, gastroesophageal reflux disease or constipation can contribute to appetite loss. Studies have shown that treating these issues can improve appetite in small eaters.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
FROM JNMG 2024
Building an AI Army of Digital Twins to Fight Cancer
A patient has cancer. It’s decision time.
Clinician and patient alike face, really, the ultimate challenge when making those decisions. They have to consider the patient’s individual circumstances, available treatment options, potential side effects, relevant clinical data such as the patient’s genetic profile and cancer specifics, and more.
“That’s a lot of information to hold,” said Uzma Asghar, PhD, MRCP, a British consultant medical oncologist at The Royal Marsden Hospital and a chief scientific officer at Concr LTD.
What if there were a way to test — quickly and accurately — all the potential paths forward?
That’s the goal of digital twins.
“What the [digital twin] model can do for the clinician is to hold all that information and process it really quickly, within a couple of minutes,” Asghar noted.
A digital twin is more than just a computer model or simulation because it copies a real-world person and relies on real-world data. Some digital twin programs also integrate new information as it becomes available. This technology holds promise for personalized medicine, drug discovery, developing screening strategies, and better understanding diseases.
How to Deliver a Twin
To create a digital twin, experts develop a computer model with data to hone its expertise in an area of medicine, such as cancer types and treatments. Then “you train the model on information it’s seen, and then introduce a patient and patient’s information,” said Asghar.
Asghar is currently working with colleagues to develop digital twins that could eventually help solve the aforementioned cancer scenario — a doctor and patient decide the best course of cancer treatment. But their applications are manifold, particularly in clinical research.
Digital twins often include a machine learning component, which would fall under the umbrella term of AI, said Asghar, but it’s not like ChatGPT or other generative AI modules many people are now familiar with.
“The difference here is the model is not there to replace the clinician or to replace clinical trials,” Asghar noted. Instead, digital twins help make decisions faster in a way that can be more affordable.
Digital Twins to Predict Cancer Outcomes
Asghar is currently involved in UK clinical trials enrolling patients with cancer to test the accuracy of digital twin programs.
At this point, these studies do not yet use digital twins to guide the course of treatment, which is something they hope to do eventually. For now, they are still at the validation phase — the digital twin program makes predictions about the treatments and then the researchers later evaluate how accurate the predictions turned out to be based on real information from the enrolled patients.
Their current model gives predictions for RECIST (response evaluation criteria in solid tumor), treatment response, and survival. In addition to collecting data from ongoing clinical trials, they’ve used retrospective data, such as from the Cancer Tumor Atlas, to test the model.
“We’ve clinically validated it now in over 9000 patients,” said Asghar, who noted that they are constantly testing it on new patients. Their data include 30 chemotherapies and 23 cancer types, but they are focusing on four: Triple-negative breast cancer, cancer of unknown primary, pancreatic cancer, and colorectal cancer.
“The reason for choosing those four cancer types is that they are aggressive, their response to chemotherapy isn’t as great, and the outcome for those patient populations, there’s significant room for improvement,” Asghar explained.
Currently, Asghar said, the model is around 80%-90% correct in predicting what the actual clinical outcomes turn out to be.
The final stage of their work, before it becomes widely available to clinicians, will be to integrate it into a clinical trial in which some clinicians use the model to make decisions about treatment vs some who don’t use the model. By studying patient outcomes in both groups, they will be able to determine the value of the digital twin program they created.
What Else Can a Twin Do? A Lot
While a model that helps clinicians make decisions about cancer treatments may be among the first digital twin programs that become widely available, there are many other kinds of digital twins in the works.
For example, a digital twin could be used as a benchmark for a patient to determine how their cancer might have progressed without treatment. Say a patient’s tumor grew during treatment, it might seem like the treatment failed, but a digital twin might show that if left untreated, the tumor would have grown five times as fast, said Paul Macklin, PhD, professor in the Department of Intelligent Systems Engineering at Indiana University Bloomington.
Alternatively, if the virtual patient’s tumor is around the same size as the real patient’s tumor, “that means that treatment has lost its efficacy. It’s time to do something new,” said Macklin. And a digital twin could help with not only choosing a therapy but also choosing a dosing schedule, he noted.
The models can also be updated as new treatments come out, which could help clinicians virtually explore how they might affect a patient before having that patient switch treatments.
Digital twins could also assist in decision-making based on a patient’s priorities and real-life circumstances. “Maybe your priority is not necessarily to shrink this [tumor] at all costs ... maybe your priority is some mix of that and also quality of life,” Macklin said, referring to potential side effects. Or if someone lives 3 hours from the nearest cancer center, a digital twin could help determine whether less frequent treatments could still be effective.
And while much of the activity around digital twins in biomedical research has been focused on cancer, Asghar said the technology has the potential to be applied to other diseases as well. A digital twin for cardiovascular disease could help doctors choose the best treatment. It could also integrate new information from a smartwatch or glucose monitor to make better predictions and help doctors adjust the treatment plan.
Faster, More Effective Research With Twins
Because digital twin programs can quickly analyze large datasets, they can also make real-world studies more effective and efficient.
Though digital twins would not fully replace real clinical trials, they could help run through preliminary scenarios before starting a full clinical trial, which would “save everybody some money, time and pain and risk,” said Macklin.
It’s also possible to use digital twins to design better screening strategies for early cancer detection and monitoring, said Ioannis Zervantonakis, PhD, a bioengineering professor at the University of Pittsburgh.
Zervantonakis is tapping digital twin technology for research that homes in on understanding tumors. In this case, the digital twin is a virtual representation of a real tumor, complete with its complex network of cells and the surrounding tissue.
Zervantonakis’ lab is using the technology to study cell-cell interactions in the tumor microenvironment, with a focus on human epidermal growth factor receptor 2–targeted therapy resistance in breast cancer. The digital twin they developed will simulate tumor growth, predict drug response, analyze cellular interactions, and optimize treatment strategies.
The Long Push Forward
One big hurdle to making digital twins more widely available is that regulation for the technology is still in progress.
“We’re developing the technology, and what’s also happening is the regulatory framework is being developed in parallel. So we’re almost developing things blindly on the basis that we think this is what the regulators would want,” explained Asghar.
“It’s really important that these technologies are regulated properly, just like drugs, and that’s what we’re pushing and advocating for,” said Asghar, noting that people need to know that like drugs, a digital twin has strengths and limitations.
And while a digital twin can be a cost-saving approach in the long run, it does require funding to get a program built, and finding funds can be difficult because not everyone knows about the technology. More funding means more trials.
With more data, Asghar is hopeful that within a few years, a digital twin model could be available for clinicians to use to help inform treatment decisions. This could lead to more effective treatments and, ultimately, better patient outcomes.
A version of this article appeared on Medscape.com.
A patient has cancer. It’s decision time.
Clinician and patient alike face, really, the ultimate challenge when making those decisions. They have to consider the patient’s individual circumstances, available treatment options, potential side effects, relevant clinical data such as the patient’s genetic profile and cancer specifics, and more.
“That’s a lot of information to hold,” said Uzma Asghar, PhD, MRCP, a British consultant medical oncologist at The Royal Marsden Hospital and a chief scientific officer at Concr LTD.
What if there were a way to test — quickly and accurately — all the potential paths forward?
That’s the goal of digital twins.
“What the [digital twin] model can do for the clinician is to hold all that information and process it really quickly, within a couple of minutes,” Asghar noted.
A digital twin is more than just a computer model or simulation because it copies a real-world person and relies on real-world data. Some digital twin programs also integrate new information as it becomes available. This technology holds promise for personalized medicine, drug discovery, developing screening strategies, and better understanding diseases.
How to Deliver a Twin
To create a digital twin, experts develop a computer model with data to hone its expertise in an area of medicine, such as cancer types and treatments. Then “you train the model on information it’s seen, and then introduce a patient and patient’s information,” said Asghar.
Asghar is currently working with colleagues to develop digital twins that could eventually help solve the aforementioned cancer scenario — a doctor and patient decide the best course of cancer treatment. But their applications are manifold, particularly in clinical research.
Digital twins often include a machine learning component, which would fall under the umbrella term of AI, said Asghar, but it’s not like ChatGPT or other generative AI modules many people are now familiar with.
“The difference here is the model is not there to replace the clinician or to replace clinical trials,” Asghar noted. Instead, digital twins help make decisions faster in a way that can be more affordable.
Digital Twins to Predict Cancer Outcomes
Asghar is currently involved in UK clinical trials enrolling patients with cancer to test the accuracy of digital twin programs.
At this point, these studies do not yet use digital twins to guide the course of treatment, which is something they hope to do eventually. For now, they are still at the validation phase — the digital twin program makes predictions about the treatments and then the researchers later evaluate how accurate the predictions turned out to be based on real information from the enrolled patients.
Their current model gives predictions for RECIST (response evaluation criteria in solid tumor), treatment response, and survival. In addition to collecting data from ongoing clinical trials, they’ve used retrospective data, such as from the Cancer Tumor Atlas, to test the model.
“We’ve clinically validated it now in over 9000 patients,” said Asghar, who noted that they are constantly testing it on new patients. Their data include 30 chemotherapies and 23 cancer types, but they are focusing on four: Triple-negative breast cancer, cancer of unknown primary, pancreatic cancer, and colorectal cancer.
“The reason for choosing those four cancer types is that they are aggressive, their response to chemotherapy isn’t as great, and the outcome for those patient populations, there’s significant room for improvement,” Asghar explained.
Currently, Asghar said, the model is around 80%-90% correct in predicting what the actual clinical outcomes turn out to be.
The final stage of their work, before it becomes widely available to clinicians, will be to integrate it into a clinical trial in which some clinicians use the model to make decisions about treatment vs some who don’t use the model. By studying patient outcomes in both groups, they will be able to determine the value of the digital twin program they created.
What Else Can a Twin Do? A Lot
While a model that helps clinicians make decisions about cancer treatments may be among the first digital twin programs that become widely available, there are many other kinds of digital twins in the works.
For example, a digital twin could be used as a benchmark for a patient to determine how their cancer might have progressed without treatment. Say a patient’s tumor grew during treatment, it might seem like the treatment failed, but a digital twin might show that if left untreated, the tumor would have grown five times as fast, said Paul Macklin, PhD, professor in the Department of Intelligent Systems Engineering at Indiana University Bloomington.
Alternatively, if the virtual patient’s tumor is around the same size as the real patient’s tumor, “that means that treatment has lost its efficacy. It’s time to do something new,” said Macklin. And a digital twin could help with not only choosing a therapy but also choosing a dosing schedule, he noted.
The models can also be updated as new treatments come out, which could help clinicians virtually explore how they might affect a patient before having that patient switch treatments.
Digital twins could also assist in decision-making based on a patient’s priorities and real-life circumstances. “Maybe your priority is not necessarily to shrink this [tumor] at all costs ... maybe your priority is some mix of that and also quality of life,” Macklin said, referring to potential side effects. Or if someone lives 3 hours from the nearest cancer center, a digital twin could help determine whether less frequent treatments could still be effective.
And while much of the activity around digital twins in biomedical research has been focused on cancer, Asghar said the technology has the potential to be applied to other diseases as well. A digital twin for cardiovascular disease could help doctors choose the best treatment. It could also integrate new information from a smartwatch or glucose monitor to make better predictions and help doctors adjust the treatment plan.
Faster, More Effective Research With Twins
Because digital twin programs can quickly analyze large datasets, they can also make real-world studies more effective and efficient.
Though digital twins would not fully replace real clinical trials, they could help run through preliminary scenarios before starting a full clinical trial, which would “save everybody some money, time and pain and risk,” said Macklin.
It’s also possible to use digital twins to design better screening strategies for early cancer detection and monitoring, said Ioannis Zervantonakis, PhD, a bioengineering professor at the University of Pittsburgh.
Zervantonakis is tapping digital twin technology for research that homes in on understanding tumors. In this case, the digital twin is a virtual representation of a real tumor, complete with its complex network of cells and the surrounding tissue.
Zervantonakis’ lab is using the technology to study cell-cell interactions in the tumor microenvironment, with a focus on human epidermal growth factor receptor 2–targeted therapy resistance in breast cancer. The digital twin they developed will simulate tumor growth, predict drug response, analyze cellular interactions, and optimize treatment strategies.
The Long Push Forward
One big hurdle to making digital twins more widely available is that regulation for the technology is still in progress.
“We’re developing the technology, and what’s also happening is the regulatory framework is being developed in parallel. So we’re almost developing things blindly on the basis that we think this is what the regulators would want,” explained Asghar.
“It’s really important that these technologies are regulated properly, just like drugs, and that’s what we’re pushing and advocating for,” said Asghar, noting that people need to know that like drugs, a digital twin has strengths and limitations.
And while a digital twin can be a cost-saving approach in the long run, it does require funding to get a program built, and finding funds can be difficult because not everyone knows about the technology. More funding means more trials.
With more data, Asghar is hopeful that within a few years, a digital twin model could be available for clinicians to use to help inform treatment decisions. This could lead to more effective treatments and, ultimately, better patient outcomes.
A version of this article appeared on Medscape.com.
A patient has cancer. It’s decision time.
Clinician and patient alike face, really, the ultimate challenge when making those decisions. They have to consider the patient’s individual circumstances, available treatment options, potential side effects, relevant clinical data such as the patient’s genetic profile and cancer specifics, and more.
“That’s a lot of information to hold,” said Uzma Asghar, PhD, MRCP, a British consultant medical oncologist at The Royal Marsden Hospital and a chief scientific officer at Concr LTD.
What if there were a way to test — quickly and accurately — all the potential paths forward?
That’s the goal of digital twins.
“What the [digital twin] model can do for the clinician is to hold all that information and process it really quickly, within a couple of minutes,” Asghar noted.
A digital twin is more than just a computer model or simulation because it copies a real-world person and relies on real-world data. Some digital twin programs also integrate new information as it becomes available. This technology holds promise for personalized medicine, drug discovery, developing screening strategies, and better understanding diseases.
How to Deliver a Twin
To create a digital twin, experts develop a computer model with data to hone its expertise in an area of medicine, such as cancer types and treatments. Then “you train the model on information it’s seen, and then introduce a patient and patient’s information,” said Asghar.
Asghar is currently working with colleagues to develop digital twins that could eventually help solve the aforementioned cancer scenario — a doctor and patient decide the best course of cancer treatment. But their applications are manifold, particularly in clinical research.
Digital twins often include a machine learning component, which would fall under the umbrella term of AI, said Asghar, but it’s not like ChatGPT or other generative AI modules many people are now familiar with.
“The difference here is the model is not there to replace the clinician or to replace clinical trials,” Asghar noted. Instead, digital twins help make decisions faster in a way that can be more affordable.
Digital Twins to Predict Cancer Outcomes
Asghar is currently involved in UK clinical trials enrolling patients with cancer to test the accuracy of digital twin programs.
At this point, these studies do not yet use digital twins to guide the course of treatment, which is something they hope to do eventually. For now, they are still at the validation phase — the digital twin program makes predictions about the treatments and then the researchers later evaluate how accurate the predictions turned out to be based on real information from the enrolled patients.
Their current model gives predictions for RECIST (response evaluation criteria in solid tumor), treatment response, and survival. In addition to collecting data from ongoing clinical trials, they’ve used retrospective data, such as from the Cancer Tumor Atlas, to test the model.
“We’ve clinically validated it now in over 9000 patients,” said Asghar, who noted that they are constantly testing it on new patients. Their data include 30 chemotherapies and 23 cancer types, but they are focusing on four: Triple-negative breast cancer, cancer of unknown primary, pancreatic cancer, and colorectal cancer.
“The reason for choosing those four cancer types is that they are aggressive, their response to chemotherapy isn’t as great, and the outcome for those patient populations, there’s significant room for improvement,” Asghar explained.
Currently, Asghar said, the model is around 80%-90% correct in predicting what the actual clinical outcomes turn out to be.
The final stage of their work, before it becomes widely available to clinicians, will be to integrate it into a clinical trial in which some clinicians use the model to make decisions about treatment vs some who don’t use the model. By studying patient outcomes in both groups, they will be able to determine the value of the digital twin program they created.
What Else Can a Twin Do? A Lot
While a model that helps clinicians make decisions about cancer treatments may be among the first digital twin programs that become widely available, there are many other kinds of digital twins in the works.
For example, a digital twin could be used as a benchmark for a patient to determine how their cancer might have progressed without treatment. Say a patient’s tumor grew during treatment, it might seem like the treatment failed, but a digital twin might show that if left untreated, the tumor would have grown five times as fast, said Paul Macklin, PhD, professor in the Department of Intelligent Systems Engineering at Indiana University Bloomington.
Alternatively, if the virtual patient’s tumor is around the same size as the real patient’s tumor, “that means that treatment has lost its efficacy. It’s time to do something new,” said Macklin. And a digital twin could help with not only choosing a therapy but also choosing a dosing schedule, he noted.
The models can also be updated as new treatments come out, which could help clinicians virtually explore how they might affect a patient before having that patient switch treatments.
Digital twins could also assist in decision-making based on a patient’s priorities and real-life circumstances. “Maybe your priority is not necessarily to shrink this [tumor] at all costs ... maybe your priority is some mix of that and also quality of life,” Macklin said, referring to potential side effects. Or if someone lives 3 hours from the nearest cancer center, a digital twin could help determine whether less frequent treatments could still be effective.
And while much of the activity around digital twins in biomedical research has been focused on cancer, Asghar said the technology has the potential to be applied to other diseases as well. A digital twin for cardiovascular disease could help doctors choose the best treatment. It could also integrate new information from a smartwatch or glucose monitor to make better predictions and help doctors adjust the treatment plan.
Faster, More Effective Research With Twins
Because digital twin programs can quickly analyze large datasets, they can also make real-world studies more effective and efficient.
Though digital twins would not fully replace real clinical trials, they could help run through preliminary scenarios before starting a full clinical trial, which would “save everybody some money, time and pain and risk,” said Macklin.
It’s also possible to use digital twins to design better screening strategies for early cancer detection and monitoring, said Ioannis Zervantonakis, PhD, a bioengineering professor at the University of Pittsburgh.
Zervantonakis is tapping digital twin technology for research that homes in on understanding tumors. In this case, the digital twin is a virtual representation of a real tumor, complete with its complex network of cells and the surrounding tissue.
Zervantonakis’ lab is using the technology to study cell-cell interactions in the tumor microenvironment, with a focus on human epidermal growth factor receptor 2–targeted therapy resistance in breast cancer. The digital twin they developed will simulate tumor growth, predict drug response, analyze cellular interactions, and optimize treatment strategies.
The Long Push Forward
One big hurdle to making digital twins more widely available is that regulation for the technology is still in progress.
“We’re developing the technology, and what’s also happening is the regulatory framework is being developed in parallel. So we’re almost developing things blindly on the basis that we think this is what the regulators would want,” explained Asghar.
“It’s really important that these technologies are regulated properly, just like drugs, and that’s what we’re pushing and advocating for,” said Asghar, noting that people need to know that like drugs, a digital twin has strengths and limitations.
And while a digital twin can be a cost-saving approach in the long run, it does require funding to get a program built, and finding funds can be difficult because not everyone knows about the technology. More funding means more trials.
With more data, Asghar is hopeful that within a few years, a digital twin model could be available for clinicians to use to help inform treatment decisions. This could lead to more effective treatments and, ultimately, better patient outcomes.
A version of this article appeared on Medscape.com.
Smokeless Tobacco, Areca Nut Chewing Behind 1 in 3 Oral Cancers: IARC Report
“Smokeless tobacco and areca nut products are available to consumers in many different forms across the world, but consuming smokeless tobacco and areca nut is linked to multiple diseases, including oral cancer,” Harriet Rumgay, PhD, a scientist in the Cancer Surveillance Branch at IARC and first author of the study in Lancet Oncology, said in a news release.
Worldwide, about 300 million people use smokeless tobacco and 600 million people use areca (also called betel) nut, one of the most popular psychoactive substances in the world after nicotine, alcohol, and caffeine. Smokeless tobacco products are consumed without burning and can be chewed, sucked, inhaled, applied locally, or ingested. Areca nut is the seed of the areca palm and can be consumed in various forms.
“Our estimates highlight the burden these products pose on health care and the importance of prevention strategies to reduce consumption of smokeless tobacco and areca nut,” Rumgay said.
According to the new report, in 2022, an estimated 120,200 of the 389,800 (30.8%) global cases of oral cancer were attributable to these products.
More than three quarters (77%) of attributable cases were among men and about one quarter (23%) among women.
The vast majority (96%) of all oral cancer cases caused by smokeless tobacco and areca nut use occurred in low- and middle-income countries.
Regions with the highest burden of oral cancers from these products were Southcentral Asia — with 105,500 of 120,200 cases (nearly 88%), including 83,400 in India, 9700 in Bangladesh, 8900 in Pakistan, and 1300 in Sri Lanka — followed by Southeastern Asia with a total of 3900 cases (1600 in Myanmar, 990 in Indonesia, and 785 in Thailand) and East Asia with 3300 cases (3200 in China).
Limitations and Action Points
The authors noted a limitation of the analysis is not accounting for the potential synergistic effects of combined use of smokeless tobacco or areca nut products with other risk factors for oral cancer, such as smoking tobacco or drinking alcohol.
The researchers explained that combined consumption of smokeless tobacco or areca nut, smoked tobacco, and alcohol has a “multiplicative effect” on oral cancer risk, with reported odds ratios increasing from 2.7 for smokeless tobacco only, 7.0 for smoked tobacco only, and 1.6 for alcohol only to 16.2 for all three exposures (vs no use).
However, the proportion of people who chewed tobacco and also smoked in countries with high smokeless tobacco or areca nut use was small. In India, for example, 6% of men and 0.5% of women in 2016-2017 were dual users of both smoked and smokeless tobacco, compared with 23% of men and 12% of women who only used smokeless tobacco.
Overall, curbing or preventing smokeless tobacco and areca nut use could help avoid many instances of oral cancer.
Despite “encouraging trends” in control of tobacco smoking in many regions of the world over the past two decades, progress in reducing the prevalence of smokeless tobacco consumption has stalled in many countries that are major consumers, the authors said.
Compounding the problem, areca nut does not fall within the WHO framework of tobacco control and there are very few areca nut control policies worldwide.
Smokeless tobacco control must be “prioritized” and a framework on areca nut control should be developed with guidelines to incorporate areca nut prevention into cancer control programs, the authors concluded.
Funding for the study was provided by the French National Cancer Institute. The authors had no relevant disclosures.
A version of this article first appeared on Medscape.com.
“Smokeless tobacco and areca nut products are available to consumers in many different forms across the world, but consuming smokeless tobacco and areca nut is linked to multiple diseases, including oral cancer,” Harriet Rumgay, PhD, a scientist in the Cancer Surveillance Branch at IARC and first author of the study in Lancet Oncology, said in a news release.
Worldwide, about 300 million people use smokeless tobacco and 600 million people use areca (also called betel) nut, one of the most popular psychoactive substances in the world after nicotine, alcohol, and caffeine. Smokeless tobacco products are consumed without burning and can be chewed, sucked, inhaled, applied locally, or ingested. Areca nut is the seed of the areca palm and can be consumed in various forms.
“Our estimates highlight the burden these products pose on health care and the importance of prevention strategies to reduce consumption of smokeless tobacco and areca nut,” Rumgay said.
According to the new report, in 2022, an estimated 120,200 of the 389,800 (30.8%) global cases of oral cancer were attributable to these products.
More than three quarters (77%) of attributable cases were among men and about one quarter (23%) among women.
The vast majority (96%) of all oral cancer cases caused by smokeless tobacco and areca nut use occurred in low- and middle-income countries.
Regions with the highest burden of oral cancers from these products were Southcentral Asia — with 105,500 of 120,200 cases (nearly 88%), including 83,400 in India, 9700 in Bangladesh, 8900 in Pakistan, and 1300 in Sri Lanka — followed by Southeastern Asia with a total of 3900 cases (1600 in Myanmar, 990 in Indonesia, and 785 in Thailand) and East Asia with 3300 cases (3200 in China).
Limitations and Action Points
The authors noted a limitation of the analysis is not accounting for the potential synergistic effects of combined use of smokeless tobacco or areca nut products with other risk factors for oral cancer, such as smoking tobacco or drinking alcohol.
The researchers explained that combined consumption of smokeless tobacco or areca nut, smoked tobacco, and alcohol has a “multiplicative effect” on oral cancer risk, with reported odds ratios increasing from 2.7 for smokeless tobacco only, 7.0 for smoked tobacco only, and 1.6 for alcohol only to 16.2 for all three exposures (vs no use).
However, the proportion of people who chewed tobacco and also smoked in countries with high smokeless tobacco or areca nut use was small. In India, for example, 6% of men and 0.5% of women in 2016-2017 were dual users of both smoked and smokeless tobacco, compared with 23% of men and 12% of women who only used smokeless tobacco.
Overall, curbing or preventing smokeless tobacco and areca nut use could help avoid many instances of oral cancer.
Despite “encouraging trends” in control of tobacco smoking in many regions of the world over the past two decades, progress in reducing the prevalence of smokeless tobacco consumption has stalled in many countries that are major consumers, the authors said.
Compounding the problem, areca nut does not fall within the WHO framework of tobacco control and there are very few areca nut control policies worldwide.
Smokeless tobacco control must be “prioritized” and a framework on areca nut control should be developed with guidelines to incorporate areca nut prevention into cancer control programs, the authors concluded.
Funding for the study was provided by the French National Cancer Institute. The authors had no relevant disclosures.
A version of this article first appeared on Medscape.com.
“Smokeless tobacco and areca nut products are available to consumers in many different forms across the world, but consuming smokeless tobacco and areca nut is linked to multiple diseases, including oral cancer,” Harriet Rumgay, PhD, a scientist in the Cancer Surveillance Branch at IARC and first author of the study in Lancet Oncology, said in a news release.
Worldwide, about 300 million people use smokeless tobacco and 600 million people use areca (also called betel) nut, one of the most popular psychoactive substances in the world after nicotine, alcohol, and caffeine. Smokeless tobacco products are consumed without burning and can be chewed, sucked, inhaled, applied locally, or ingested. Areca nut is the seed of the areca palm and can be consumed in various forms.
“Our estimates highlight the burden these products pose on health care and the importance of prevention strategies to reduce consumption of smokeless tobacco and areca nut,” Rumgay said.
According to the new report, in 2022, an estimated 120,200 of the 389,800 (30.8%) global cases of oral cancer were attributable to these products.
More than three quarters (77%) of attributable cases were among men and about one quarter (23%) among women.
The vast majority (96%) of all oral cancer cases caused by smokeless tobacco and areca nut use occurred in low- and middle-income countries.
Regions with the highest burden of oral cancers from these products were Southcentral Asia — with 105,500 of 120,200 cases (nearly 88%), including 83,400 in India, 9700 in Bangladesh, 8900 in Pakistan, and 1300 in Sri Lanka — followed by Southeastern Asia with a total of 3900 cases (1600 in Myanmar, 990 in Indonesia, and 785 in Thailand) and East Asia with 3300 cases (3200 in China).
Limitations and Action Points
The authors noted a limitation of the analysis is not accounting for the potential synergistic effects of combined use of smokeless tobacco or areca nut products with other risk factors for oral cancer, such as smoking tobacco or drinking alcohol.
The researchers explained that combined consumption of smokeless tobacco or areca nut, smoked tobacco, and alcohol has a “multiplicative effect” on oral cancer risk, with reported odds ratios increasing from 2.7 for smokeless tobacco only, 7.0 for smoked tobacco only, and 1.6 for alcohol only to 16.2 for all three exposures (vs no use).
However, the proportion of people who chewed tobacco and also smoked in countries with high smokeless tobacco or areca nut use was small. In India, for example, 6% of men and 0.5% of women in 2016-2017 were dual users of both smoked and smokeless tobacco, compared with 23% of men and 12% of women who only used smokeless tobacco.
Overall, curbing or preventing smokeless tobacco and areca nut use could help avoid many instances of oral cancer.
Despite “encouraging trends” in control of tobacco smoking in many regions of the world over the past two decades, progress in reducing the prevalence of smokeless tobacco consumption has stalled in many countries that are major consumers, the authors said.
Compounding the problem, areca nut does not fall within the WHO framework of tobacco control and there are very few areca nut control policies worldwide.
Smokeless tobacco control must be “prioritized” and a framework on areca nut control should be developed with guidelines to incorporate areca nut prevention into cancer control programs, the authors concluded.
Funding for the study was provided by the French National Cancer Institute. The authors had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM THE LANCET ONCOLOGY
Study Finds No Significant Effect of Low-Dose Oral Minoxidil on BP
TOPLINE:
but is associated with a slight increase in heart rate and a 5% incidence of hypotensive symptoms.
METHODOLOGY:
- Researchers conducted a systematic review and meta-analysis of 16 studies, which involved 2387 patients with alopecia (60.7% women) who received minoxidil, a vasodilator originally developed as an antihypertensive, at doses of 5 mg or less per day.
- Outcomes included changes in mean arterial pressure, systolic BP, diastolic BP, and heart rate.
- Mean differences were calculated between pretreatment and posttreatment values.
TAKEAWAY:
- Hypotensive symptoms were reported in 5% patients, with no significant hypotensive episodes. About 1.8% patients experienced lightheadedness or syncope, 1.2% experienced dizziness, 0.9% had tachycardia, and 0.8% had palpitations.
- LDOM did not significantly alter systolic BP (mean difference, –0.13; 95% CI, –2.67 to 2.41), diastolic BP (mean difference, –1.25; 95% CI, –3.21 to 0.71), and mean arterial pressure (mean difference, –1.92; 95% CI, –4.00 to 0.17).
- LDOM led to a significant increase in heart rate (mean difference, 2.67 beats/min; 95% CI, 0.34-5.01), a difference the authors wrote would “likely not be clinically significant for most patients.”
- Hypertrichosis was the most common side effect (59.6%) and reason for stopping treatment (accounting for nearly 35% of discontinuations).
IN PRACTICE:
“LDOM appears to be a safe treatment for alopecia with no significant impact on blood pressure,” the authors wrote, noting that the study “addresses gaps in clinical knowledge involving LDOM.” Based on their results, they recommended that BP and heart rate “do not need to be closely monitored in patients without prior cardiovascular risk history.”
SOURCE:
The study was led by Matthew Chen, BS, Stony Brook Dermatology in New York. It was published online in The Journal of the American Academy of Dermatology.
LIMITATIONS:
The studies included had small sample sizes and retrospective designs, which may limit the reliability of the findings. Additional limitations include the absence of control groups, a potential recall bias in adverse effect reporting, and variability in dosing regimens and BP monitoring.
DISCLOSURES:
The authors reported no external funding or conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
but is associated with a slight increase in heart rate and a 5% incidence of hypotensive symptoms.
METHODOLOGY:
- Researchers conducted a systematic review and meta-analysis of 16 studies, which involved 2387 patients with alopecia (60.7% women) who received minoxidil, a vasodilator originally developed as an antihypertensive, at doses of 5 mg or less per day.
- Outcomes included changes in mean arterial pressure, systolic BP, diastolic BP, and heart rate.
- Mean differences were calculated between pretreatment and posttreatment values.
TAKEAWAY:
- Hypotensive symptoms were reported in 5% patients, with no significant hypotensive episodes. About 1.8% patients experienced lightheadedness or syncope, 1.2% experienced dizziness, 0.9% had tachycardia, and 0.8% had palpitations.
- LDOM did not significantly alter systolic BP (mean difference, –0.13; 95% CI, –2.67 to 2.41), diastolic BP (mean difference, –1.25; 95% CI, –3.21 to 0.71), and mean arterial pressure (mean difference, –1.92; 95% CI, –4.00 to 0.17).
- LDOM led to a significant increase in heart rate (mean difference, 2.67 beats/min; 95% CI, 0.34-5.01), a difference the authors wrote would “likely not be clinically significant for most patients.”
- Hypertrichosis was the most common side effect (59.6%) and reason for stopping treatment (accounting for nearly 35% of discontinuations).
IN PRACTICE:
“LDOM appears to be a safe treatment for alopecia with no significant impact on blood pressure,” the authors wrote, noting that the study “addresses gaps in clinical knowledge involving LDOM.” Based on their results, they recommended that BP and heart rate “do not need to be closely monitored in patients without prior cardiovascular risk history.”
SOURCE:
The study was led by Matthew Chen, BS, Stony Brook Dermatology in New York. It was published online in The Journal of the American Academy of Dermatology.
LIMITATIONS:
The studies included had small sample sizes and retrospective designs, which may limit the reliability of the findings. Additional limitations include the absence of control groups, a potential recall bias in adverse effect reporting, and variability in dosing regimens and BP monitoring.
DISCLOSURES:
The authors reported no external funding or conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
but is associated with a slight increase in heart rate and a 5% incidence of hypotensive symptoms.
METHODOLOGY:
- Researchers conducted a systematic review and meta-analysis of 16 studies, which involved 2387 patients with alopecia (60.7% women) who received minoxidil, a vasodilator originally developed as an antihypertensive, at doses of 5 mg or less per day.
- Outcomes included changes in mean arterial pressure, systolic BP, diastolic BP, and heart rate.
- Mean differences were calculated between pretreatment and posttreatment values.
TAKEAWAY:
- Hypotensive symptoms were reported in 5% patients, with no significant hypotensive episodes. About 1.8% patients experienced lightheadedness or syncope, 1.2% experienced dizziness, 0.9% had tachycardia, and 0.8% had palpitations.
- LDOM did not significantly alter systolic BP (mean difference, –0.13; 95% CI, –2.67 to 2.41), diastolic BP (mean difference, –1.25; 95% CI, –3.21 to 0.71), and mean arterial pressure (mean difference, –1.92; 95% CI, –4.00 to 0.17).
- LDOM led to a significant increase in heart rate (mean difference, 2.67 beats/min; 95% CI, 0.34-5.01), a difference the authors wrote would “likely not be clinically significant for most patients.”
- Hypertrichosis was the most common side effect (59.6%) and reason for stopping treatment (accounting for nearly 35% of discontinuations).
IN PRACTICE:
“LDOM appears to be a safe treatment for alopecia with no significant impact on blood pressure,” the authors wrote, noting that the study “addresses gaps in clinical knowledge involving LDOM.” Based on their results, they recommended that BP and heart rate “do not need to be closely monitored in patients without prior cardiovascular risk history.”
SOURCE:
The study was led by Matthew Chen, BS, Stony Brook Dermatology in New York. It was published online in The Journal of the American Academy of Dermatology.
LIMITATIONS:
The studies included had small sample sizes and retrospective designs, which may limit the reliability of the findings. Additional limitations include the absence of control groups, a potential recall bias in adverse effect reporting, and variability in dosing regimens and BP monitoring.
DISCLOSURES:
The authors reported no external funding or conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Physicians as Advocates
As physicians, we hold positions of power and privilege in society. With that power and privilege comes a sacred responsibility to serve as healthcare advocates, for our individual patients as well as for our larger communities in helping them navigate the complexities of the healthcare system and recognize and address barriers to care.
In addition to delivering high-quality, patient-centered care in our clinics and endoscopy units, it is imperative that gastroenterologists use our individual and collective voice in promoting policy reforms that improve healthcare access, affordability, and equity, and help the healthcare system work better for our patients.
Particularly in the wake of Election Day, it is critical to realize the power of physician advocacy in informing rational policymaking relating to healthcare. In furtherance of this goal, AGA held its largest ever Advocacy Day this Fall, bringing together more than 100 members and patient advocates from 28 states to educate members of Congress and their staff about policies affecting GI patient care, including prior authorization, step therapy, federal research funding, and Medicare reimbursement. As a new slate of elected officials prepares to take office in January, I hope you will join me in considering how best to use your voice as a physician to promote systemic change, whether through participating in a future AGA Congressional Advocacy Day, or by meeting independently with your local, state, or federal elected officials to discuss the issues critical to the well-being of your patients and communities. Now more than ever, physician advocacy is critical to achieving a high-performing healthcare system.
In GIHN’s December issue, In our Member Spotlight column, Dr. Jeffrey Lee (Kaiser Permanente N. California) updates us on his innovative research on colorectal cancer prevention. We hope you have a wonderful holiday season with your friends and family and look forward to seeing you again in 2025!
Megan A. Adams, MD, JD, MSc
Editor in Chief
As physicians, we hold positions of power and privilege in society. With that power and privilege comes a sacred responsibility to serve as healthcare advocates, for our individual patients as well as for our larger communities in helping them navigate the complexities of the healthcare system and recognize and address barriers to care.
In addition to delivering high-quality, patient-centered care in our clinics and endoscopy units, it is imperative that gastroenterologists use our individual and collective voice in promoting policy reforms that improve healthcare access, affordability, and equity, and help the healthcare system work better for our patients.
Particularly in the wake of Election Day, it is critical to realize the power of physician advocacy in informing rational policymaking relating to healthcare. In furtherance of this goal, AGA held its largest ever Advocacy Day this Fall, bringing together more than 100 members and patient advocates from 28 states to educate members of Congress and their staff about policies affecting GI patient care, including prior authorization, step therapy, federal research funding, and Medicare reimbursement. As a new slate of elected officials prepares to take office in January, I hope you will join me in considering how best to use your voice as a physician to promote systemic change, whether through participating in a future AGA Congressional Advocacy Day, or by meeting independently with your local, state, or federal elected officials to discuss the issues critical to the well-being of your patients and communities. Now more than ever, physician advocacy is critical to achieving a high-performing healthcare system.
In GIHN’s December issue, In our Member Spotlight column, Dr. Jeffrey Lee (Kaiser Permanente N. California) updates us on his innovative research on colorectal cancer prevention. We hope you have a wonderful holiday season with your friends and family and look forward to seeing you again in 2025!
Megan A. Adams, MD, JD, MSc
Editor in Chief
As physicians, we hold positions of power and privilege in society. With that power and privilege comes a sacred responsibility to serve as healthcare advocates, for our individual patients as well as for our larger communities in helping them navigate the complexities of the healthcare system and recognize and address barriers to care.
In addition to delivering high-quality, patient-centered care in our clinics and endoscopy units, it is imperative that gastroenterologists use our individual and collective voice in promoting policy reforms that improve healthcare access, affordability, and equity, and help the healthcare system work better for our patients.
Particularly in the wake of Election Day, it is critical to realize the power of physician advocacy in informing rational policymaking relating to healthcare. In furtherance of this goal, AGA held its largest ever Advocacy Day this Fall, bringing together more than 100 members and patient advocates from 28 states to educate members of Congress and their staff about policies affecting GI patient care, including prior authorization, step therapy, federal research funding, and Medicare reimbursement. As a new slate of elected officials prepares to take office in January, I hope you will join me in considering how best to use your voice as a physician to promote systemic change, whether through participating in a future AGA Congressional Advocacy Day, or by meeting independently with your local, state, or federal elected officials to discuss the issues critical to the well-being of your patients and communities. Now more than ever, physician advocacy is critical to achieving a high-performing healthcare system.
In GIHN’s December issue, In our Member Spotlight column, Dr. Jeffrey Lee (Kaiser Permanente N. California) updates us on his innovative research on colorectal cancer prevention. We hope you have a wonderful holiday season with your friends and family and look forward to seeing you again in 2025!
Megan A. Adams, MD, JD, MSc
Editor in Chief
Many Patients With Cancer Visit EDs Before Diagnosis
Researchers examined Institute for Clinical Evaluative Sciences (ICES) data that had been gathered from January 1, 2014, to December 31, 2021. The study focused on patients aged 18 years or older with confirmed primary cancer diagnoses.
Factors associated with an increased likelihood of an ED visit ahead of diagnosis included having certain cancers, living in rural areas, and having less access to primary care, according to study author Keerat Grewal, MD, an emergency physician and clinician scientist at the Schwartz/Reisman Emergency Medicine Institute at Sinai Health in Toronto, Ontario, Canada, and coauthors.
“The ED is a distressing environment for patients to receive a possible cancer diagnosis,” the authors wrote. “Moreover, it is frequently ill equipped to provide ongoing continuity of care, which can lead patients down a poorly defined diagnostic pathway before receiving a confirmed diagnosis based on tissue and a subsequent treatment plan.”
The findings were published online on November 4 in CMAJ).
Neurologic Cancers Prominent
In an interview, Grewal said in an interview that the study reflects her desire as an emergency room physician to understand why so many patients with cancer get the initial reports about their disease from clinicians whom they often have just met for the first time.
Among patients with an ED visit before cancer diagnosis, 51.4% were admitted to hospital from the most recent visit.
Compared with patients with a family physician on whom they could rely for routine care, those who had no outpatient visits (odds ratio [OR], 2.09) or fewer than three outpatient visits (OR, 1.41) in the 6-30 months before cancer diagnosis were more likely to have an ED visit before their cancer diagnosis.
Other factors associated with increased odds of ED use before cancer diagnosis included rurality (OR, 1.15), residence in northern Ontario (northeast region: OR, 1.14 and northwest region: OR, 1.27 vs Toronto region), and living in the most marginalized areas (material resource deprivation: OR, 1.37 and housing stability: OR, 1.09 vs least marginalized area).
The researchers also found that patients with certain cancers were more likely to have sought care in the ED. They compared these cancers with breast cancer, which is often detected through screening.
“Patients with neurologic cancers had extremely high odds of ED use before cancer diagnosis,” the authors wrote. “This is likely because of the emergent nature of presentation, with acute neurologic symptoms such as weakness, confusion, or seizures, which require urgent assessment.” On the other hand, pancreatic, liver, or thoracic cancer can trigger nonspecific symptoms that may be ignored until they reach a crisis level that prompts an ED visit.
The limitations of the study included its inability to identify cancer-related ED visits and its narrow focus on patients in Ontario, according to the researchers. But the use of the ICES databases also allowed researchers access to a broader pool of data than are available in many other cases.
The findings in the new paper echo those of previous research, the authors noted. Research in the United Kingdom found that 24%-31% of cancer diagnoses involved the ED. In addition, a study of people enrolled in the US Medicare program, which serves patients aged 65 years or older, found that 23% were seen in the ED in the 30 days before diagnosis.
‘Unpacking the Data’
The current findings also are consistent with those of an International Cancer Benchmarking Partnership study that was published in 2022 in The Lancet Oncology, said Erika Nicholson, MHS, vice president of cancer systems and innovation at the Canadian Partnership Against Cancer. The latter study analyzed cancer registration and linked hospital admissions data from 14 jurisdictions in Australia, Canada, Denmark, New Zealand, Norway, and the United Kingdom.
“We see similar trends in terms of people visiting EDs and being diagnosed through EDs internationally,” Nicholson said. “We’re working with partners to put in place different strategies to address the challenges” that this phenomenon presents in terms of improving screening and follow-up care.
“Cancer is not one disease, but many diseases,” she said. “They present differently. We’re focused on really unpacking the data and understanding them.”
All this research highlights the need for more services and personnel to address cancer, including people who are trained to help patients cope after getting concerning news through emergency care, she said.
“That means having a system that fully supports you and helps you navigate through that diagnostic process,” Nicholson said. Addressing the added challenges for patients who don’t have secure housing is a special need, she added.
This study was supported by the Canadian Institutes of Health Research (CIHR). Grewal reported receiving grants from CIHR and the Canadian Association of Emergency Physicians. Nicholson reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Researchers examined Institute for Clinical Evaluative Sciences (ICES) data that had been gathered from January 1, 2014, to December 31, 2021. The study focused on patients aged 18 years or older with confirmed primary cancer diagnoses.
Factors associated with an increased likelihood of an ED visit ahead of diagnosis included having certain cancers, living in rural areas, and having less access to primary care, according to study author Keerat Grewal, MD, an emergency physician and clinician scientist at the Schwartz/Reisman Emergency Medicine Institute at Sinai Health in Toronto, Ontario, Canada, and coauthors.
“The ED is a distressing environment for patients to receive a possible cancer diagnosis,” the authors wrote. “Moreover, it is frequently ill equipped to provide ongoing continuity of care, which can lead patients down a poorly defined diagnostic pathway before receiving a confirmed diagnosis based on tissue and a subsequent treatment plan.”
The findings were published online on November 4 in CMAJ).
Neurologic Cancers Prominent
In an interview, Grewal said in an interview that the study reflects her desire as an emergency room physician to understand why so many patients with cancer get the initial reports about their disease from clinicians whom they often have just met for the first time.
Among patients with an ED visit before cancer diagnosis, 51.4% were admitted to hospital from the most recent visit.
Compared with patients with a family physician on whom they could rely for routine care, those who had no outpatient visits (odds ratio [OR], 2.09) or fewer than three outpatient visits (OR, 1.41) in the 6-30 months before cancer diagnosis were more likely to have an ED visit before their cancer diagnosis.
Other factors associated with increased odds of ED use before cancer diagnosis included rurality (OR, 1.15), residence in northern Ontario (northeast region: OR, 1.14 and northwest region: OR, 1.27 vs Toronto region), and living in the most marginalized areas (material resource deprivation: OR, 1.37 and housing stability: OR, 1.09 vs least marginalized area).
The researchers also found that patients with certain cancers were more likely to have sought care in the ED. They compared these cancers with breast cancer, which is often detected through screening.
“Patients with neurologic cancers had extremely high odds of ED use before cancer diagnosis,” the authors wrote. “This is likely because of the emergent nature of presentation, with acute neurologic symptoms such as weakness, confusion, or seizures, which require urgent assessment.” On the other hand, pancreatic, liver, or thoracic cancer can trigger nonspecific symptoms that may be ignored until they reach a crisis level that prompts an ED visit.
The limitations of the study included its inability to identify cancer-related ED visits and its narrow focus on patients in Ontario, according to the researchers. But the use of the ICES databases also allowed researchers access to a broader pool of data than are available in many other cases.
The findings in the new paper echo those of previous research, the authors noted. Research in the United Kingdom found that 24%-31% of cancer diagnoses involved the ED. In addition, a study of people enrolled in the US Medicare program, which serves patients aged 65 years or older, found that 23% were seen in the ED in the 30 days before diagnosis.
‘Unpacking the Data’
The current findings also are consistent with those of an International Cancer Benchmarking Partnership study that was published in 2022 in The Lancet Oncology, said Erika Nicholson, MHS, vice president of cancer systems and innovation at the Canadian Partnership Against Cancer. The latter study analyzed cancer registration and linked hospital admissions data from 14 jurisdictions in Australia, Canada, Denmark, New Zealand, Norway, and the United Kingdom.
“We see similar trends in terms of people visiting EDs and being diagnosed through EDs internationally,” Nicholson said. “We’re working with partners to put in place different strategies to address the challenges” that this phenomenon presents in terms of improving screening and follow-up care.
“Cancer is not one disease, but many diseases,” she said. “They present differently. We’re focused on really unpacking the data and understanding them.”
All this research highlights the need for more services and personnel to address cancer, including people who are trained to help patients cope after getting concerning news through emergency care, she said.
“That means having a system that fully supports you and helps you navigate through that diagnostic process,” Nicholson said. Addressing the added challenges for patients who don’t have secure housing is a special need, she added.
This study was supported by the Canadian Institutes of Health Research (CIHR). Grewal reported receiving grants from CIHR and the Canadian Association of Emergency Physicians. Nicholson reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Researchers examined Institute for Clinical Evaluative Sciences (ICES) data that had been gathered from January 1, 2014, to December 31, 2021. The study focused on patients aged 18 years or older with confirmed primary cancer diagnoses.
Factors associated with an increased likelihood of an ED visit ahead of diagnosis included having certain cancers, living in rural areas, and having less access to primary care, according to study author Keerat Grewal, MD, an emergency physician and clinician scientist at the Schwartz/Reisman Emergency Medicine Institute at Sinai Health in Toronto, Ontario, Canada, and coauthors.
“The ED is a distressing environment for patients to receive a possible cancer diagnosis,” the authors wrote. “Moreover, it is frequently ill equipped to provide ongoing continuity of care, which can lead patients down a poorly defined diagnostic pathway before receiving a confirmed diagnosis based on tissue and a subsequent treatment plan.”
The findings were published online on November 4 in CMAJ).
Neurologic Cancers Prominent
In an interview, Grewal said in an interview that the study reflects her desire as an emergency room physician to understand why so many patients with cancer get the initial reports about their disease from clinicians whom they often have just met for the first time.
Among patients with an ED visit before cancer diagnosis, 51.4% were admitted to hospital from the most recent visit.
Compared with patients with a family physician on whom they could rely for routine care, those who had no outpatient visits (odds ratio [OR], 2.09) or fewer than three outpatient visits (OR, 1.41) in the 6-30 months before cancer diagnosis were more likely to have an ED visit before their cancer diagnosis.
Other factors associated with increased odds of ED use before cancer diagnosis included rurality (OR, 1.15), residence in northern Ontario (northeast region: OR, 1.14 and northwest region: OR, 1.27 vs Toronto region), and living in the most marginalized areas (material resource deprivation: OR, 1.37 and housing stability: OR, 1.09 vs least marginalized area).
The researchers also found that patients with certain cancers were more likely to have sought care in the ED. They compared these cancers with breast cancer, which is often detected through screening.
“Patients with neurologic cancers had extremely high odds of ED use before cancer diagnosis,” the authors wrote. “This is likely because of the emergent nature of presentation, with acute neurologic symptoms such as weakness, confusion, or seizures, which require urgent assessment.” On the other hand, pancreatic, liver, or thoracic cancer can trigger nonspecific symptoms that may be ignored until they reach a crisis level that prompts an ED visit.
The limitations of the study included its inability to identify cancer-related ED visits and its narrow focus on patients in Ontario, according to the researchers. But the use of the ICES databases also allowed researchers access to a broader pool of data than are available in many other cases.
The findings in the new paper echo those of previous research, the authors noted. Research in the United Kingdom found that 24%-31% of cancer diagnoses involved the ED. In addition, a study of people enrolled in the US Medicare program, which serves patients aged 65 years or older, found that 23% were seen in the ED in the 30 days before diagnosis.
‘Unpacking the Data’
The current findings also are consistent with those of an International Cancer Benchmarking Partnership study that was published in 2022 in The Lancet Oncology, said Erika Nicholson, MHS, vice president of cancer systems and innovation at the Canadian Partnership Against Cancer. The latter study analyzed cancer registration and linked hospital admissions data from 14 jurisdictions in Australia, Canada, Denmark, New Zealand, Norway, and the United Kingdom.
“We see similar trends in terms of people visiting EDs and being diagnosed through EDs internationally,” Nicholson said. “We’re working with partners to put in place different strategies to address the challenges” that this phenomenon presents in terms of improving screening and follow-up care.
“Cancer is not one disease, but many diseases,” she said. “They present differently. We’re focused on really unpacking the data and understanding them.”
All this research highlights the need for more services and personnel to address cancer, including people who are trained to help patients cope after getting concerning news through emergency care, she said.
“That means having a system that fully supports you and helps you navigate through that diagnostic process,” Nicholson said. Addressing the added challenges for patients who don’t have secure housing is a special need, she added.
This study was supported by the Canadian Institutes of Health Research (CIHR). Grewal reported receiving grants from CIHR and the Canadian Association of Emergency Physicians. Nicholson reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM CMAJ
Plasma Omega-6 and Omega-3 Fatty Acids Inversely Associated With Cancer
TOPLINE:
Higher plasma levels of omega-6 and omega-3 fatty acids are associated with a lower incidence of cancer. However, omega-3 fatty acids are linked to an increased risk for prostate cancer, specifically.
METHODOLOGY:
- Researchers looked for associations of plasma omega-3 and omega-6 polyunsaturated fatty acids (PUFAs) with the incidence of cancer overall and 19 site-specific cancers in the large population-based prospective UK Biobank cohort.
- They included 253,138 participants aged 37-73 years who were followed for an average of 12.9 years, with 29,838 diagnosed with cancer.
- Plasma levels of omega-3 and omega-6 fatty acids were measured using nuclear magnetic resonance and expressed as percentages of total fatty acids.
- Participants with cancer diagnoses at baseline, those who withdrew from the study, and those with missing data on plasma PUFAs were excluded.
- The study adjusted for multiple covariates, including age, sex, ethnicity, socioeconomic status, lifestyle behaviors, and family history of diseases.
TAKEAWAY:
- Higher plasma levels of omega-6 and omega-3 fatty acids were associated with a 2% and 1% reduction in overall cancer risk per SD increase, respectively (P = .001 and P = .03).
- Omega-6 fatty acids were inversely associated with 14 site-specific cancers, whereas omega-3 fatty acids were inversely associated with five site-specific cancers.
- Prostate cancer was positively associated with omega-3 fatty acids, with a 3% increased risk per SD increase (P = .049).
- A higher omega-6/omega-3 ratio was associated with an increased risk for overall cancer, and three site-specific cancers showed positive associations with the ratio. “Each standard deviation increase, corresponding to a 13.13 increase in the omega ratio, was associated with a 2% increase in the risk of rectum cancer,” for example, the authors wrote.
IN PRACTICE:
“Overall, our findings provide support for possible small net protective roles of omega-3 and omega-6 PUFAs in the development of new cancer incidence. Our study also suggests that the usage of circulating blood biomarkers captures different aspects of dietary intake, reduces measurement errors, and thus enhances statistical power. The differential effects of omega-6% and omega-3% in age and sex subgroups warrant future investigation,” wrote the authors of the study.
SOURCE:
The study was led by Yuchen Zhang of the University of Georgia in Athens, Georgia. It was published online in the International Journal of Cancer.
LIMITATIONS:
The study’s potential for selective bias persists due to the participant sample skewing heavily toward European ancestry and White ethnicity. The number of events was small for some specific cancer sites, which may have limited the statistical power. The study focused on total omega-3 and omega-6 PUFAs, with only two individual fatty acids measured. Future studies are needed to examine the roles of other individual PUFAs and specific genetic variants.
DISCLOSURES:
This study was supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Higher plasma levels of omega-6 and omega-3 fatty acids are associated with a lower incidence of cancer. However, omega-3 fatty acids are linked to an increased risk for prostate cancer, specifically.
METHODOLOGY:
- Researchers looked for associations of plasma omega-3 and omega-6 polyunsaturated fatty acids (PUFAs) with the incidence of cancer overall and 19 site-specific cancers in the large population-based prospective UK Biobank cohort.
- They included 253,138 participants aged 37-73 years who were followed for an average of 12.9 years, with 29,838 diagnosed with cancer.
- Plasma levels of omega-3 and omega-6 fatty acids were measured using nuclear magnetic resonance and expressed as percentages of total fatty acids.
- Participants with cancer diagnoses at baseline, those who withdrew from the study, and those with missing data on plasma PUFAs were excluded.
- The study adjusted for multiple covariates, including age, sex, ethnicity, socioeconomic status, lifestyle behaviors, and family history of diseases.
TAKEAWAY:
- Higher plasma levels of omega-6 and omega-3 fatty acids were associated with a 2% and 1% reduction in overall cancer risk per SD increase, respectively (P = .001 and P = .03).
- Omega-6 fatty acids were inversely associated with 14 site-specific cancers, whereas omega-3 fatty acids were inversely associated with five site-specific cancers.
- Prostate cancer was positively associated with omega-3 fatty acids, with a 3% increased risk per SD increase (P = .049).
- A higher omega-6/omega-3 ratio was associated with an increased risk for overall cancer, and three site-specific cancers showed positive associations with the ratio. “Each standard deviation increase, corresponding to a 13.13 increase in the omega ratio, was associated with a 2% increase in the risk of rectum cancer,” for example, the authors wrote.
IN PRACTICE:
“Overall, our findings provide support for possible small net protective roles of omega-3 and omega-6 PUFAs in the development of new cancer incidence. Our study also suggests that the usage of circulating blood biomarkers captures different aspects of dietary intake, reduces measurement errors, and thus enhances statistical power. The differential effects of omega-6% and omega-3% in age and sex subgroups warrant future investigation,” wrote the authors of the study.
SOURCE:
The study was led by Yuchen Zhang of the University of Georgia in Athens, Georgia. It was published online in the International Journal of Cancer.
LIMITATIONS:
The study’s potential for selective bias persists due to the participant sample skewing heavily toward European ancestry and White ethnicity. The number of events was small for some specific cancer sites, which may have limited the statistical power. The study focused on total omega-3 and omega-6 PUFAs, with only two individual fatty acids measured. Future studies are needed to examine the roles of other individual PUFAs and specific genetic variants.
DISCLOSURES:
This study was supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Higher plasma levels of omega-6 and omega-3 fatty acids are associated with a lower incidence of cancer. However, omega-3 fatty acids are linked to an increased risk for prostate cancer, specifically.
METHODOLOGY:
- Researchers looked for associations of plasma omega-3 and omega-6 polyunsaturated fatty acids (PUFAs) with the incidence of cancer overall and 19 site-specific cancers in the large population-based prospective UK Biobank cohort.
- They included 253,138 participants aged 37-73 years who were followed for an average of 12.9 years, with 29,838 diagnosed with cancer.
- Plasma levels of omega-3 and omega-6 fatty acids were measured using nuclear magnetic resonance and expressed as percentages of total fatty acids.
- Participants with cancer diagnoses at baseline, those who withdrew from the study, and those with missing data on plasma PUFAs were excluded.
- The study adjusted for multiple covariates, including age, sex, ethnicity, socioeconomic status, lifestyle behaviors, and family history of diseases.
TAKEAWAY:
- Higher plasma levels of omega-6 and omega-3 fatty acids were associated with a 2% and 1% reduction in overall cancer risk per SD increase, respectively (P = .001 and P = .03).
- Omega-6 fatty acids were inversely associated with 14 site-specific cancers, whereas omega-3 fatty acids were inversely associated with five site-specific cancers.
- Prostate cancer was positively associated with omega-3 fatty acids, with a 3% increased risk per SD increase (P = .049).
- A higher omega-6/omega-3 ratio was associated with an increased risk for overall cancer, and three site-specific cancers showed positive associations with the ratio. “Each standard deviation increase, corresponding to a 13.13 increase in the omega ratio, was associated with a 2% increase in the risk of rectum cancer,” for example, the authors wrote.
IN PRACTICE:
“Overall, our findings provide support for possible small net protective roles of omega-3 and omega-6 PUFAs in the development of new cancer incidence. Our study also suggests that the usage of circulating blood biomarkers captures different aspects of dietary intake, reduces measurement errors, and thus enhances statistical power. The differential effects of omega-6% and omega-3% in age and sex subgroups warrant future investigation,” wrote the authors of the study.
SOURCE:
The study was led by Yuchen Zhang of the University of Georgia in Athens, Georgia. It was published online in the International Journal of Cancer.
LIMITATIONS:
The study’s potential for selective bias persists due to the participant sample skewing heavily toward European ancestry and White ethnicity. The number of events was small for some specific cancer sites, which may have limited the statistical power. The study focused on total omega-3 and omega-6 PUFAs, with only two individual fatty acids measured. Future studies are needed to examine the roles of other individual PUFAs and specific genetic variants.
DISCLOSURES:
This study was supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Pinto Bean Pressure Wraps: A Novel Approach to Treating Digital Warts
Practice Gap
Verruca vulgaris is a common dermatologic challenge due to its high prevalence and tendency to recur following routinely employed destructive modalities (eg, cryotherapy, electrosurgery), which can incur a considerable amount of pain and some risk for scarring.1,2 Other treatment methods for warts such as topical salicylic acid preparations, topical immunotherapy, or intralesional allergen injections often require multiple treatment sessions.3,4 Furthermore, the financial burden of traditional wart treatment can be substantial.4 Better techniques are needed to improve the clinician’s approach to treating warts. We describe a home-based technique to treat common digital warts using pinto bean pressure wraps to induce ischemic changes in wart tissue with similar response rates to commonly used modalities.
Technique
Our technique utilizes a small, hard, convex object that is applied directly over the digital wart. A simple self-adhesive wrap is used to cover the object and maintain constant pressure on the wart overnight. We typically use a dried pinto bean (a variety of the common bean Phaseolus vulgaris) acquired from a local grocery store due to its ideal size, hard surface, and convex shape (Figure 1). The bean is taped in place directly overlying the wart and covered with a self-adhesive wrap overnight. The wrap is removed in the morning, and often no further treatment is needed. The ischemic wart tissue is allowed to slough spontaneously over 1 to 2 weeks. No wound care or dressing is necessary (Figure 2). Larger warts may require application of the pressure wraps for 2 to 3 additional nights. While most warts resolve with this technique, we have observed a recurrence rate similar to that for cryotherapy. Patients are advised that any recurrent warts can be re-treated monthly, if needed, until resolution.
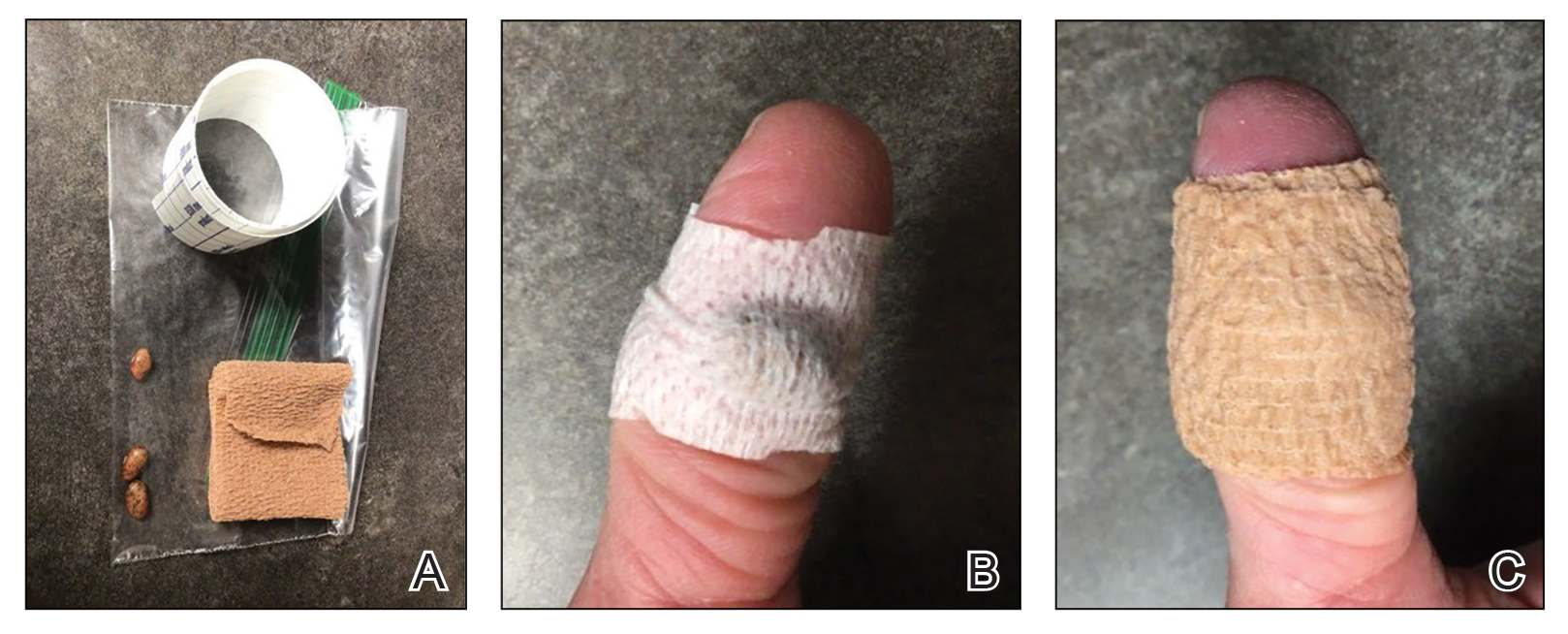
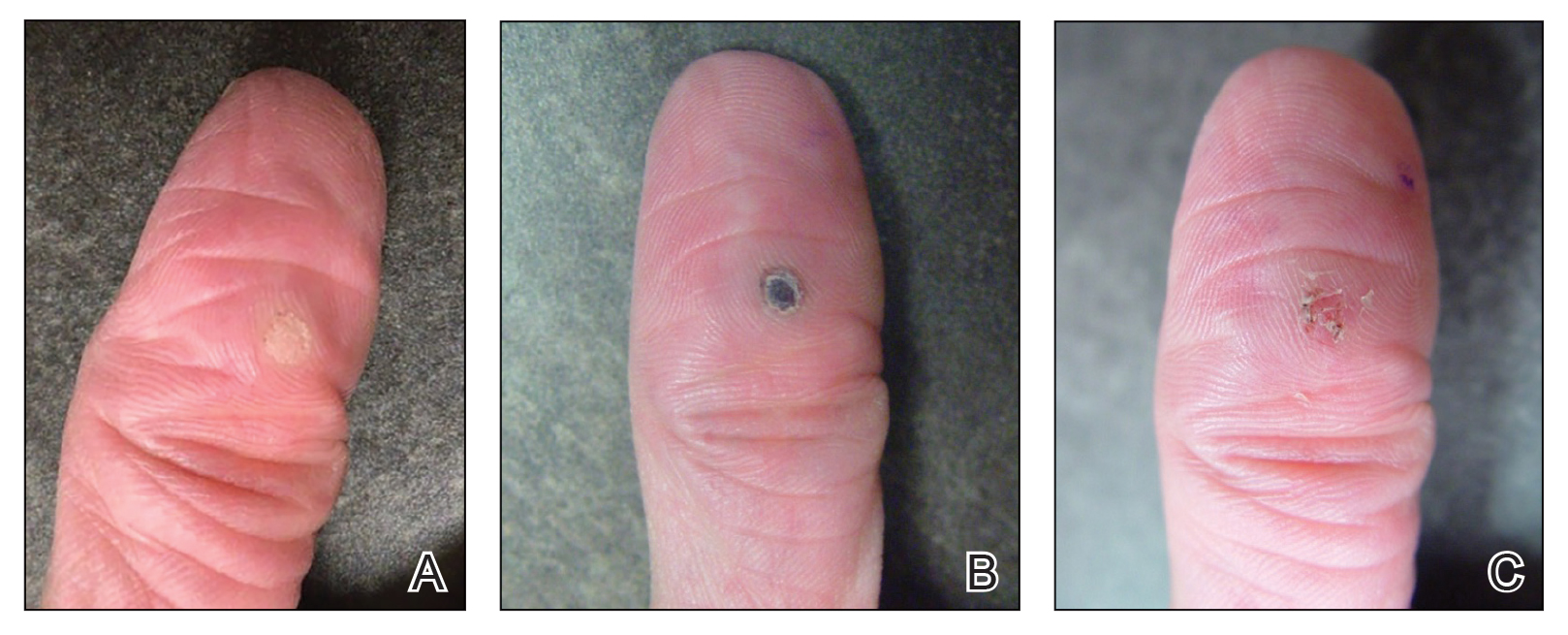
What to Use and How to Prepare—Any small, hard, convex object can be used for the pressure wrap; we also have used appropriately sized and shaped plastic shirt buttons with similar results. Home kits can be assembled in advance and provided to patients at their initial visit along with appropriate instructions (Figure 1A).
Effects on the Skin and Distal Digit—Application of pressure wraps does not harm normal skin; however, care should be taken when the self-adherent wrap is applied so as not to induce ischemia of the distal digit. The wrap should be applied using gentle pressure with patients experiencing minimal discomfort from the overnight application.
Indications—This pressure wrap technique can be employed on most digital warts, including periungual warts, which can be difficult to treat by other means. However, in our experience this technique is not effective for nondigital warts, likely due to the inability to maintain adequate pressure with the overlying dressing. Patients at risk for compromised digital perfusion, such as those with Raynaud phenomenon or systemic sclerosis, should not be treated with pressure wraps due to possible digital ischemia.
Precautions—Patients should be advised that the pinto bean should only be used if dry and should not be ingested. The bean can be a choking hazard for small children, therefore appropriate precautions should be used. Allergic contact dermatitis to the materials used in this technique is possible, but we have never observed this. The pinto bean can be reused for future application as long as it remains dry and provides a hard convex surface.
Practice Implications
The probable mechanism of the ischemic changes to the wart tissue likely is the occlusion of tortuous blood vessels in the dermal papillae, which are intrinsic to wart tissue and absent in normal skin.1 This pressure-induced ischemic injury allows for selective destruction of the wart tissue with sparing of the normal skin. Our technique is fairly novel, although at least one report in the literature has described the use of a mechanical device to induce ischemic changes in skin tags.5
The use of pinto bean pressure wraps to induce ischemic change in digital warts provides a low-risk and nearly pain-free alternative to more expensive and invasive treatment methods. Moreover, this technique allows for a low-cost home-based therapy that can be repeated easily for other digital sites or if recurrence is noted.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20:145-154.
- Lipke M. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi:10.3121/cmr.4.4.273
- Muse M, Stiff K, Glines K, et al. A review of intralesional wart therapy. Dermatol Online J. 2020;26:2. doi:10.5070/D3263048027
- Berna R, Margolis D, Barbieri J. Annual health care utilization and costs for treatment of cutaneous and anogenital warts among a commercially insured population in the US, 2017-2019. JAMA Dermatol. 2022;158:695-697. doi:10.1001/jamadermatol.2022.0964
- Fredriksson C, Ilias M, Anderson C. New mechanical device for effective removal of skin tags in routine health care. Dermatol Online J. 2009;15:9. doi:10.5070/D37tj2800k
Practice Gap
Verruca vulgaris is a common dermatologic challenge due to its high prevalence and tendency to recur following routinely employed destructive modalities (eg, cryotherapy, electrosurgery), which can incur a considerable amount of pain and some risk for scarring.1,2 Other treatment methods for warts such as topical salicylic acid preparations, topical immunotherapy, or intralesional allergen injections often require multiple treatment sessions.3,4 Furthermore, the financial burden of traditional wart treatment can be substantial.4 Better techniques are needed to improve the clinician’s approach to treating warts. We describe a home-based technique to treat common digital warts using pinto bean pressure wraps to induce ischemic changes in wart tissue with similar response rates to commonly used modalities.
Technique
Our technique utilizes a small, hard, convex object that is applied directly over the digital wart. A simple self-adhesive wrap is used to cover the object and maintain constant pressure on the wart overnight. We typically use a dried pinto bean (a variety of the common bean Phaseolus vulgaris) acquired from a local grocery store due to its ideal size, hard surface, and convex shape (Figure 1). The bean is taped in place directly overlying the wart and covered with a self-adhesive wrap overnight. The wrap is removed in the morning, and often no further treatment is needed. The ischemic wart tissue is allowed to slough spontaneously over 1 to 2 weeks. No wound care or dressing is necessary (Figure 2). Larger warts may require application of the pressure wraps for 2 to 3 additional nights. While most warts resolve with this technique, we have observed a recurrence rate similar to that for cryotherapy. Patients are advised that any recurrent warts can be re-treated monthly, if needed, until resolution.


What to Use and How to Prepare—Any small, hard, convex object can be used for the pressure wrap; we also have used appropriately sized and shaped plastic shirt buttons with similar results. Home kits can be assembled in advance and provided to patients at their initial visit along with appropriate instructions (Figure 1A).
Effects on the Skin and Distal Digit—Application of pressure wraps does not harm normal skin; however, care should be taken when the self-adherent wrap is applied so as not to induce ischemia of the distal digit. The wrap should be applied using gentle pressure with patients experiencing minimal discomfort from the overnight application.
Indications—This pressure wrap technique can be employed on most digital warts, including periungual warts, which can be difficult to treat by other means. However, in our experience this technique is not effective for nondigital warts, likely due to the inability to maintain adequate pressure with the overlying dressing. Patients at risk for compromised digital perfusion, such as those with Raynaud phenomenon or systemic sclerosis, should not be treated with pressure wraps due to possible digital ischemia.
Precautions—Patients should be advised that the pinto bean should only be used if dry and should not be ingested. The bean can be a choking hazard for small children, therefore appropriate precautions should be used. Allergic contact dermatitis to the materials used in this technique is possible, but we have never observed this. The pinto bean can be reused for future application as long as it remains dry and provides a hard convex surface.
Practice Implications
The probable mechanism of the ischemic changes to the wart tissue likely is the occlusion of tortuous blood vessels in the dermal papillae, which are intrinsic to wart tissue and absent in normal skin.1 This pressure-induced ischemic injury allows for selective destruction of the wart tissue with sparing of the normal skin. Our technique is fairly novel, although at least one report in the literature has described the use of a mechanical device to induce ischemic changes in skin tags.5
The use of pinto bean pressure wraps to induce ischemic change in digital warts provides a low-risk and nearly pain-free alternative to more expensive and invasive treatment methods. Moreover, this technique allows for a low-cost home-based therapy that can be repeated easily for other digital sites or if recurrence is noted.
Practice Gap
Verruca vulgaris is a common dermatologic challenge due to its high prevalence and tendency to recur following routinely employed destructive modalities (eg, cryotherapy, electrosurgery), which can incur a considerable amount of pain and some risk for scarring.1,2 Other treatment methods for warts such as topical salicylic acid preparations, topical immunotherapy, or intralesional allergen injections often require multiple treatment sessions.3,4 Furthermore, the financial burden of traditional wart treatment can be substantial.4 Better techniques are needed to improve the clinician’s approach to treating warts. We describe a home-based technique to treat common digital warts using pinto bean pressure wraps to induce ischemic changes in wart tissue with similar response rates to commonly used modalities.
Technique
Our technique utilizes a small, hard, convex object that is applied directly over the digital wart. A simple self-adhesive wrap is used to cover the object and maintain constant pressure on the wart overnight. We typically use a dried pinto bean (a variety of the common bean Phaseolus vulgaris) acquired from a local grocery store due to its ideal size, hard surface, and convex shape (Figure 1). The bean is taped in place directly overlying the wart and covered with a self-adhesive wrap overnight. The wrap is removed in the morning, and often no further treatment is needed. The ischemic wart tissue is allowed to slough spontaneously over 1 to 2 weeks. No wound care or dressing is necessary (Figure 2). Larger warts may require application of the pressure wraps for 2 to 3 additional nights. While most warts resolve with this technique, we have observed a recurrence rate similar to that for cryotherapy. Patients are advised that any recurrent warts can be re-treated monthly, if needed, until resolution.


What to Use and How to Prepare—Any small, hard, convex object can be used for the pressure wrap; we also have used appropriately sized and shaped plastic shirt buttons with similar results. Home kits can be assembled in advance and provided to patients at their initial visit along with appropriate instructions (Figure 1A).
Effects on the Skin and Distal Digit—Application of pressure wraps does not harm normal skin; however, care should be taken when the self-adherent wrap is applied so as not to induce ischemia of the distal digit. The wrap should be applied using gentle pressure with patients experiencing minimal discomfort from the overnight application.
Indications—This pressure wrap technique can be employed on most digital warts, including periungual warts, which can be difficult to treat by other means. However, in our experience this technique is not effective for nondigital warts, likely due to the inability to maintain adequate pressure with the overlying dressing. Patients at risk for compromised digital perfusion, such as those with Raynaud phenomenon or systemic sclerosis, should not be treated with pressure wraps due to possible digital ischemia.
Precautions—Patients should be advised that the pinto bean should only be used if dry and should not be ingested. The bean can be a choking hazard for small children, therefore appropriate precautions should be used. Allergic contact dermatitis to the materials used in this technique is possible, but we have never observed this. The pinto bean can be reused for future application as long as it remains dry and provides a hard convex surface.
Practice Implications
The probable mechanism of the ischemic changes to the wart tissue likely is the occlusion of tortuous blood vessels in the dermal papillae, which are intrinsic to wart tissue and absent in normal skin.1 This pressure-induced ischemic injury allows for selective destruction of the wart tissue with sparing of the normal skin. Our technique is fairly novel, although at least one report in the literature has described the use of a mechanical device to induce ischemic changes in skin tags.5
The use of pinto bean pressure wraps to induce ischemic change in digital warts provides a low-risk and nearly pain-free alternative to more expensive and invasive treatment methods. Moreover, this technique allows for a low-cost home-based therapy that can be repeated easily for other digital sites or if recurrence is noted.
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20:145-154.
- Lipke M. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi:10.3121/cmr.4.4.273
- Muse M, Stiff K, Glines K, et al. A review of intralesional wart therapy. Dermatol Online J. 2020;26:2. doi:10.5070/D3263048027
- Berna R, Margolis D, Barbieri J. Annual health care utilization and costs for treatment of cutaneous and anogenital warts among a commercially insured population in the US, 2017-2019. JAMA Dermatol. 2022;158:695-697. doi:10.1001/jamadermatol.2022.0964
- Fredriksson C, Ilias M, Anderson C. New mechanical device for effective removal of skin tags in routine health care. Dermatol Online J. 2009;15:9. doi:10.5070/D37tj2800k
- Cardoso J, Calonje E. Cutaneous manifestations of human papillomaviruses: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20:145-154.
- Lipke M. An armamentarium of wart treatments. Clin Med Res. 2006;4:273-293. doi:10.3121/cmr.4.4.273
- Muse M, Stiff K, Glines K, et al. A review of intralesional wart therapy. Dermatol Online J. 2020;26:2. doi:10.5070/D3263048027
- Berna R, Margolis D, Barbieri J. Annual health care utilization and costs for treatment of cutaneous and anogenital warts among a commercially insured population in the US, 2017-2019. JAMA Dermatol. 2022;158:695-697. doi:10.1001/jamadermatol.2022.0964
- Fredriksson C, Ilias M, Anderson C. New mechanical device for effective removal of skin tags in routine health care. Dermatol Online J. 2009;15:9. doi:10.5070/D37tj2800k