User login
Smartphones for children with type 1 diabetes: Cause for concern?
My young patient with type 1 diabetes (T1D) had her cell phone out to provide a share code for her Dexcom clarity app as she was checking into her visit. As my nurse was recording the code, the patient asked him, “Hey, can you add me on Snapchat?”
Her father scrolled through his own Facebook feed in the chair next to her, showing no concern that his daughter was looking to connect with an adult on a social media platform. Meanwhile, we were all grateful that the little girl, who had had a seizure due to hypoglycemia in her preschool and pre–continuous glucose monitoring (CGM) years, had access to the tools harnessed within the sparkly encased phone she held in her small hands. But did anyone in the room fully understand the potential dangers?
We are living in an exhilarating era of diabetes technology, a treatment environment that I couldn’t have dreamed of during my pediatric endocrinology fellowship. T1D is a volatile condition that changes day to day, especially in growing children. A short decade ago, the best CGM available was a bulky device on loan to patients for 3 days at a time. Information was later downloaded in-office to get a better idea of general glucose trends, if insurance would approve its use at all.
Now, we have a variety of very wearable and accurate disposable CGMs accessible to most patients. Every major insulin pump has available closed-loop capabilities. Some patients can dose from apps on their cell phones rather than juggle another device or draw attention to an insulin pump at the cafeteria table.
These developments have been game changers for children and teenagers with diabetes and for their families. When wondering whether an athlete’s dazed appearance on a soccer field was due to hypoglycemia, a parent no longer must demand that a coach pull the player – a quick glance at a smartphone app can verify the blood glucose and change rate. Children can use programs and search engines to quickly verify carbohydrate counts. Life360 and other tracking programs have increased parental feelings of security, especially with young drivers living with a chronic medical condition.
The inevitable outcome of this available technology is that children living with T1D are given cell phones far earlier than are their siblings or peers owing to “necessity.” Parents understandably want a means to stay in close contact with their children in case of a medical emergency. As a physician and mother of young children, I am thankful for the technology that keeps my patients safer and that allows them to fully participate in everything from sports to travel to an uninterrupted night’s sleep.
Smartphone presence in classrooms empowers teachers, students, parents, and school nurses to be aware of glycemic trends and prevent hypoglycemic emergencies. Smartphones have also shown to be a major distraction in that setting, causing many schools to ban their use entirely. Video apps such as YouTube and TikTok can provide a wealth of support and medical information but may also open the door to misinformation and dangerous social contagion, particularly surrounding disordered eating. Informative podcasts such as The Juicebox Podcast and online forums provide incredible support for families, but the constant siren call of a phone in their pockets leads to distracted parents constantly tending to other conversations or responding to ever more demanding employers rather than focusing on face-to-face education sessions.
The Surgeon General recently released a report concerning social media use in children. This eye-opening report revealed that one-third of children admitted to using their cell phones “almost constantly.” Social media use is associated with higher rates of anxiety and depression, especially in teen girls. This is particularly concerning for children with T1D, who are more likely to suffer from these conditions.
Beyond mental health concerns, especially to developing brains, unfettered Internet use increases the risk that children are exposed to predators and harmful content. The online safety monitoring platform Bark shared data from its 2021 surveillance. Bark found that 72% of tweens and 85% of teens were involved in an online bullying situation. Sixty-nine percent of tweens and 91% of teens encountered nudity or sexual content. Ten percent of tweens and 21% of teens encountered predatory behavior.
These alarming finds mirror the prevalence suggested by conversations in my office. I hear reports of my patients sneaking out at night to meet adults they met through social media, having suicidal ideation and attempts after Internet bullying, and sharing earnest belief in bizarre conspiracy theories gleaned from online forums that lead to dangerous health care practices.
Furthermore, time is a finite resource. Teens who are spending an average of 3.5 hours daily on their devices are running out of time to play, study, and grow extracurricular interests. My friend who coaches high school baseball lamented recently the poor athleticism in his recent teams. He theorized that his players had spent their summers on tablets rather than playing catch or climbing trees. The resulting declines in exercise in young people only serve to worsen the childhood obesity epidemic.
What is a concerned parent to do? First, all phones have controls that allow parents to choose which apps are allowed and which are blocked. Caregivers must understand how various social media platforms work. Installing programs such as Bark provides an additional layer of monitoring, though these are no substitute for parental vigilance. Importantly, parents should talk to their children about their concerns regarding social media.
Sadly, I have often noticed that caregivers pity the extra hardships their children endure as the result of T1D and other chronic diseases. Being lax with rules to attempt to compensate for other suffering is far too tempting. The goal is for children and teens living with T1D to have a full and normal childhood, and unrestricted smartphone access and early social media use should not be the goal for any child. For every family, a media use plan is a smart approach. The American Academy of Pediatrics suggests several commonsense steps to use technology wisely, and parents often must address their own relationships with their devices to model healthy engagement.
As health care professionals, we owe it to our patients to discuss the ups and downs of technology with our patients. We can’t ostrich our way through this. We can point our patients and families to supportive groups such as Osprey (Old School Parents Raising Engaged Youth), founded by Ben and Erin Napier from the HGTV show Home Town along with my college friends Taylor and Dr. Catherine Sledge. Wait Until 8th provides information and motivation for parents to make wise choices regarding phone use for their children. The documentary Childhood 2.0 is another compelling resource developed by pediatric emergency physician Dr. Free Hess and her team that summarizes many of these concerns.
In another decade, many of these dangers will be far clearer. As ubiquitous as smartphone misuse is in our society, I remain hopeful that our society will change its behaviors. Just because “everyone else” allows an unhealthy relationship with technology doesn’t mean that we should for our children.
When I was a child, smoking was glamorized in movies and restaurants had dedicated smoking sections. After strong public policy efforts, many geared toward children, smoking is now almost unthinkable. My 8-year-old asked me lately whether a lady smoking a cigarette in the car next to us would have to go to jail. I chose a career in pediatrics because I am an optimist at my very core. We can’t ignore the dangers associated with the wide door opened by mobile devices. We can celebrate the benefits while clearly facing the pitfalls.
Dr. Lilley is director of the pediatric diabetes and lipid program at the Mississippi Center for Advanced Medicine, Madison. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
My young patient with type 1 diabetes (T1D) had her cell phone out to provide a share code for her Dexcom clarity app as she was checking into her visit. As my nurse was recording the code, the patient asked him, “Hey, can you add me on Snapchat?”
Her father scrolled through his own Facebook feed in the chair next to her, showing no concern that his daughter was looking to connect with an adult on a social media platform. Meanwhile, we were all grateful that the little girl, who had had a seizure due to hypoglycemia in her preschool and pre–continuous glucose monitoring (CGM) years, had access to the tools harnessed within the sparkly encased phone she held in her small hands. But did anyone in the room fully understand the potential dangers?
We are living in an exhilarating era of diabetes technology, a treatment environment that I couldn’t have dreamed of during my pediatric endocrinology fellowship. T1D is a volatile condition that changes day to day, especially in growing children. A short decade ago, the best CGM available was a bulky device on loan to patients for 3 days at a time. Information was later downloaded in-office to get a better idea of general glucose trends, if insurance would approve its use at all.
Now, we have a variety of very wearable and accurate disposable CGMs accessible to most patients. Every major insulin pump has available closed-loop capabilities. Some patients can dose from apps on their cell phones rather than juggle another device or draw attention to an insulin pump at the cafeteria table.
These developments have been game changers for children and teenagers with diabetes and for their families. When wondering whether an athlete’s dazed appearance on a soccer field was due to hypoglycemia, a parent no longer must demand that a coach pull the player – a quick glance at a smartphone app can verify the blood glucose and change rate. Children can use programs and search engines to quickly verify carbohydrate counts. Life360 and other tracking programs have increased parental feelings of security, especially with young drivers living with a chronic medical condition.
The inevitable outcome of this available technology is that children living with T1D are given cell phones far earlier than are their siblings or peers owing to “necessity.” Parents understandably want a means to stay in close contact with their children in case of a medical emergency. As a physician and mother of young children, I am thankful for the technology that keeps my patients safer and that allows them to fully participate in everything from sports to travel to an uninterrupted night’s sleep.
Smartphone presence in classrooms empowers teachers, students, parents, and school nurses to be aware of glycemic trends and prevent hypoglycemic emergencies. Smartphones have also shown to be a major distraction in that setting, causing many schools to ban their use entirely. Video apps such as YouTube and TikTok can provide a wealth of support and medical information but may also open the door to misinformation and dangerous social contagion, particularly surrounding disordered eating. Informative podcasts such as The Juicebox Podcast and online forums provide incredible support for families, but the constant siren call of a phone in their pockets leads to distracted parents constantly tending to other conversations or responding to ever more demanding employers rather than focusing on face-to-face education sessions.
The Surgeon General recently released a report concerning social media use in children. This eye-opening report revealed that one-third of children admitted to using their cell phones “almost constantly.” Social media use is associated with higher rates of anxiety and depression, especially in teen girls. This is particularly concerning for children with T1D, who are more likely to suffer from these conditions.
Beyond mental health concerns, especially to developing brains, unfettered Internet use increases the risk that children are exposed to predators and harmful content. The online safety monitoring platform Bark shared data from its 2021 surveillance. Bark found that 72% of tweens and 85% of teens were involved in an online bullying situation. Sixty-nine percent of tweens and 91% of teens encountered nudity or sexual content. Ten percent of tweens and 21% of teens encountered predatory behavior.
These alarming finds mirror the prevalence suggested by conversations in my office. I hear reports of my patients sneaking out at night to meet adults they met through social media, having suicidal ideation and attempts after Internet bullying, and sharing earnest belief in bizarre conspiracy theories gleaned from online forums that lead to dangerous health care practices.
Furthermore, time is a finite resource. Teens who are spending an average of 3.5 hours daily on their devices are running out of time to play, study, and grow extracurricular interests. My friend who coaches high school baseball lamented recently the poor athleticism in his recent teams. He theorized that his players had spent their summers on tablets rather than playing catch or climbing trees. The resulting declines in exercise in young people only serve to worsen the childhood obesity epidemic.
What is a concerned parent to do? First, all phones have controls that allow parents to choose which apps are allowed and which are blocked. Caregivers must understand how various social media platforms work. Installing programs such as Bark provides an additional layer of monitoring, though these are no substitute for parental vigilance. Importantly, parents should talk to their children about their concerns regarding social media.
Sadly, I have often noticed that caregivers pity the extra hardships their children endure as the result of T1D and other chronic diseases. Being lax with rules to attempt to compensate for other suffering is far too tempting. The goal is for children and teens living with T1D to have a full and normal childhood, and unrestricted smartphone access and early social media use should not be the goal for any child. For every family, a media use plan is a smart approach. The American Academy of Pediatrics suggests several commonsense steps to use technology wisely, and parents often must address their own relationships with their devices to model healthy engagement.
As health care professionals, we owe it to our patients to discuss the ups and downs of technology with our patients. We can’t ostrich our way through this. We can point our patients and families to supportive groups such as Osprey (Old School Parents Raising Engaged Youth), founded by Ben and Erin Napier from the HGTV show Home Town along with my college friends Taylor and Dr. Catherine Sledge. Wait Until 8th provides information and motivation for parents to make wise choices regarding phone use for their children. The documentary Childhood 2.0 is another compelling resource developed by pediatric emergency physician Dr. Free Hess and her team that summarizes many of these concerns.
In another decade, many of these dangers will be far clearer. As ubiquitous as smartphone misuse is in our society, I remain hopeful that our society will change its behaviors. Just because “everyone else” allows an unhealthy relationship with technology doesn’t mean that we should for our children.
When I was a child, smoking was glamorized in movies and restaurants had dedicated smoking sections. After strong public policy efforts, many geared toward children, smoking is now almost unthinkable. My 8-year-old asked me lately whether a lady smoking a cigarette in the car next to us would have to go to jail. I chose a career in pediatrics because I am an optimist at my very core. We can’t ignore the dangers associated with the wide door opened by mobile devices. We can celebrate the benefits while clearly facing the pitfalls.
Dr. Lilley is director of the pediatric diabetes and lipid program at the Mississippi Center for Advanced Medicine, Madison. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
My young patient with type 1 diabetes (T1D) had her cell phone out to provide a share code for her Dexcom clarity app as she was checking into her visit. As my nurse was recording the code, the patient asked him, “Hey, can you add me on Snapchat?”
Her father scrolled through his own Facebook feed in the chair next to her, showing no concern that his daughter was looking to connect with an adult on a social media platform. Meanwhile, we were all grateful that the little girl, who had had a seizure due to hypoglycemia in her preschool and pre–continuous glucose monitoring (CGM) years, had access to the tools harnessed within the sparkly encased phone she held in her small hands. But did anyone in the room fully understand the potential dangers?
We are living in an exhilarating era of diabetes technology, a treatment environment that I couldn’t have dreamed of during my pediatric endocrinology fellowship. T1D is a volatile condition that changes day to day, especially in growing children. A short decade ago, the best CGM available was a bulky device on loan to patients for 3 days at a time. Information was later downloaded in-office to get a better idea of general glucose trends, if insurance would approve its use at all.
Now, we have a variety of very wearable and accurate disposable CGMs accessible to most patients. Every major insulin pump has available closed-loop capabilities. Some patients can dose from apps on their cell phones rather than juggle another device or draw attention to an insulin pump at the cafeteria table.
These developments have been game changers for children and teenagers with diabetes and for their families. When wondering whether an athlete’s dazed appearance on a soccer field was due to hypoglycemia, a parent no longer must demand that a coach pull the player – a quick glance at a smartphone app can verify the blood glucose and change rate. Children can use programs and search engines to quickly verify carbohydrate counts. Life360 and other tracking programs have increased parental feelings of security, especially with young drivers living with a chronic medical condition.
The inevitable outcome of this available technology is that children living with T1D are given cell phones far earlier than are their siblings or peers owing to “necessity.” Parents understandably want a means to stay in close contact with their children in case of a medical emergency. As a physician and mother of young children, I am thankful for the technology that keeps my patients safer and that allows them to fully participate in everything from sports to travel to an uninterrupted night’s sleep.
Smartphone presence in classrooms empowers teachers, students, parents, and school nurses to be aware of glycemic trends and prevent hypoglycemic emergencies. Smartphones have also shown to be a major distraction in that setting, causing many schools to ban their use entirely. Video apps such as YouTube and TikTok can provide a wealth of support and medical information but may also open the door to misinformation and dangerous social contagion, particularly surrounding disordered eating. Informative podcasts such as The Juicebox Podcast and online forums provide incredible support for families, but the constant siren call of a phone in their pockets leads to distracted parents constantly tending to other conversations or responding to ever more demanding employers rather than focusing on face-to-face education sessions.
The Surgeon General recently released a report concerning social media use in children. This eye-opening report revealed that one-third of children admitted to using their cell phones “almost constantly.” Social media use is associated with higher rates of anxiety and depression, especially in teen girls. This is particularly concerning for children with T1D, who are more likely to suffer from these conditions.
Beyond mental health concerns, especially to developing brains, unfettered Internet use increases the risk that children are exposed to predators and harmful content. The online safety monitoring platform Bark shared data from its 2021 surveillance. Bark found that 72% of tweens and 85% of teens were involved in an online bullying situation. Sixty-nine percent of tweens and 91% of teens encountered nudity or sexual content. Ten percent of tweens and 21% of teens encountered predatory behavior.
These alarming finds mirror the prevalence suggested by conversations in my office. I hear reports of my patients sneaking out at night to meet adults they met through social media, having suicidal ideation and attempts after Internet bullying, and sharing earnest belief in bizarre conspiracy theories gleaned from online forums that lead to dangerous health care practices.
Furthermore, time is a finite resource. Teens who are spending an average of 3.5 hours daily on their devices are running out of time to play, study, and grow extracurricular interests. My friend who coaches high school baseball lamented recently the poor athleticism in his recent teams. He theorized that his players had spent their summers on tablets rather than playing catch or climbing trees. The resulting declines in exercise in young people only serve to worsen the childhood obesity epidemic.
What is a concerned parent to do? First, all phones have controls that allow parents to choose which apps are allowed and which are blocked. Caregivers must understand how various social media platforms work. Installing programs such as Bark provides an additional layer of monitoring, though these are no substitute for parental vigilance. Importantly, parents should talk to their children about their concerns regarding social media.
Sadly, I have often noticed that caregivers pity the extra hardships their children endure as the result of T1D and other chronic diseases. Being lax with rules to attempt to compensate for other suffering is far too tempting. The goal is for children and teens living with T1D to have a full and normal childhood, and unrestricted smartphone access and early social media use should not be the goal for any child. For every family, a media use plan is a smart approach. The American Academy of Pediatrics suggests several commonsense steps to use technology wisely, and parents often must address their own relationships with their devices to model healthy engagement.
As health care professionals, we owe it to our patients to discuss the ups and downs of technology with our patients. We can’t ostrich our way through this. We can point our patients and families to supportive groups such as Osprey (Old School Parents Raising Engaged Youth), founded by Ben and Erin Napier from the HGTV show Home Town along with my college friends Taylor and Dr. Catherine Sledge. Wait Until 8th provides information and motivation for parents to make wise choices regarding phone use for their children. The documentary Childhood 2.0 is another compelling resource developed by pediatric emergency physician Dr. Free Hess and her team that summarizes many of these concerns.
In another decade, many of these dangers will be far clearer. As ubiquitous as smartphone misuse is in our society, I remain hopeful that our society will change its behaviors. Just because “everyone else” allows an unhealthy relationship with technology doesn’t mean that we should for our children.
When I was a child, smoking was glamorized in movies and restaurants had dedicated smoking sections. After strong public policy efforts, many geared toward children, smoking is now almost unthinkable. My 8-year-old asked me lately whether a lady smoking a cigarette in the car next to us would have to go to jail. I chose a career in pediatrics because I am an optimist at my very core. We can’t ignore the dangers associated with the wide door opened by mobile devices. We can celebrate the benefits while clearly facing the pitfalls.
Dr. Lilley is director of the pediatric diabetes and lipid program at the Mississippi Center for Advanced Medicine, Madison. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
CHMP recommends marketing of biologic for atopic dermatitis
The who are candidates for systemic therapy.
Lebrikizumab is an investigational, monoclonal antibody that binds to cytokine interleukin (IL)-13, which has been implicated in driving the type-2 inflammatory loop in the skin, leading to skin barrier dysfunction, itch, skin thickening, and infection. The biologic is being developed by Almirall and is designed to be administered once per month. Lebrikizumab is not yet available in the United States.
According to a press release from Almirall, the CHMP opinion was based on three pivotal phase 3 studies that showed long-term response in skin clearance and itch control. ADvocate 1 and ADvocate 2 evaluated lebrikizumab as monotherapy, while ADhere assessed lebrikizumab in combination with topical corticosteroids (TCS) in adult and adolescent patients with moderate to severe AD. At week 16, more than 50% of patients with moderate to severe AD experienced at least a 75% reduction in disease severity (EASI-75) when receiving lebrikizumab monotherapy in the ADvocate studies and nearly 70% of patients receiving lebrikizumab combined with standard-of-care TCS achieved EASI-75 in the ADhere trial.
Most adverse events across the studies were mild or moderate. The most common reactions were conjunctivitis, injection site reactions, allergic conjunctivitis, and dry eye.
The who are candidates for systemic therapy.
Lebrikizumab is an investigational, monoclonal antibody that binds to cytokine interleukin (IL)-13, which has been implicated in driving the type-2 inflammatory loop in the skin, leading to skin barrier dysfunction, itch, skin thickening, and infection. The biologic is being developed by Almirall and is designed to be administered once per month. Lebrikizumab is not yet available in the United States.
According to a press release from Almirall, the CHMP opinion was based on three pivotal phase 3 studies that showed long-term response in skin clearance and itch control. ADvocate 1 and ADvocate 2 evaluated lebrikizumab as monotherapy, while ADhere assessed lebrikizumab in combination with topical corticosteroids (TCS) in adult and adolescent patients with moderate to severe AD. At week 16, more than 50% of patients with moderate to severe AD experienced at least a 75% reduction in disease severity (EASI-75) when receiving lebrikizumab monotherapy in the ADvocate studies and nearly 70% of patients receiving lebrikizumab combined with standard-of-care TCS achieved EASI-75 in the ADhere trial.
Most adverse events across the studies were mild or moderate. The most common reactions were conjunctivitis, injection site reactions, allergic conjunctivitis, and dry eye.
The who are candidates for systemic therapy.
Lebrikizumab is an investigational, monoclonal antibody that binds to cytokine interleukin (IL)-13, which has been implicated in driving the type-2 inflammatory loop in the skin, leading to skin barrier dysfunction, itch, skin thickening, and infection. The biologic is being developed by Almirall and is designed to be administered once per month. Lebrikizumab is not yet available in the United States.
According to a press release from Almirall, the CHMP opinion was based on three pivotal phase 3 studies that showed long-term response in skin clearance and itch control. ADvocate 1 and ADvocate 2 evaluated lebrikizumab as monotherapy, while ADhere assessed lebrikizumab in combination with topical corticosteroids (TCS) in adult and adolescent patients with moderate to severe AD. At week 16, more than 50% of patients with moderate to severe AD experienced at least a 75% reduction in disease severity (EASI-75) when receiving lebrikizumab monotherapy in the ADvocate studies and nearly 70% of patients receiving lebrikizumab combined with standard-of-care TCS achieved EASI-75 in the ADhere trial.
Most adverse events across the studies were mild or moderate. The most common reactions were conjunctivitis, injection site reactions, allergic conjunctivitis, and dry eye.
It’s back to school for asthma, too
The years go by, and nothing much changes: The first 2 weeks of the new school year have brought with them a rise in emergency department (ED) admissions for asthma in patients under age 15 years. A more relaxed approach to maintenance therapy for the condition over the summer holidays, exposure to allergens at school, and the surge in viral respiratory infections that accompanies the return to group settings explain this trend, which can be foreseen.
According to Public Health France, which has just relaunched its epidemiological monitoring, these cases reach their peak around 2 weeks after the start of the new term.
In its first weekly review on Aug. 22, 2023, the authority reported a slight uptick in cases in its Indian Ocean overseas departments, and the calm before the storm in mainland France.
Last year, between weeks 35 and 36, the increases were 82% for SOS Médecins (the French home doctor visit service), 169% for EDs, and 33% for hospital admissions.
These data are similar to the figures obtained over the past 3 years. The authors of this monitoring, using the SurSaUD system, France’s program for monitoring emergency cases and deaths, attribute these increases to the surge in viral respiratory infections seen after the return to group settings after the school summer holidays.
Indeed, viral-induced exacerbations are mostly caused by rhinoviruses, which circulate throughout the year, but more so during the autumn and winter months. These are probably the main culprits behind the epidemics seen once schools have reopened. Yet relaxation of maintenance asthma treatment (inhaled corticosteroids alone or in combination with long-acting bronchodilators) during the summer holidays also plays a significant role in this yearly recurrence.
Compliance ends with school
Flore Amat, MD, PhD, pediatric respiratory and allergy specialist and coordinating doctor at the Zephyr asthma clinic (Robert-Debré Hospital, Paris Public Hospitals) acknowledged, “The summer holidays are often a time when compliance with maintenance therapy is relaxed.” Aware of this fact, doctors prefer to strike a deal with their young patients. “For some of our young and teenage asthma patients, we support their relaxed approach to medication during the summer holidays,” she admitted. “In July and August, there are fewer viruses circulating, and the weather is often dry, which limits the risk of an asthma attack, meaning we can ease off the maintenance therapy, or even stop taking it altogether. We tell parents and children to start taking them again 2 weeks before school starts; 2 weeks being the minimum time needed for inhaled corticosteroids to start taking effect again.” Unsurprisingly, some forget to do so or simply don’t.
Two other things contribute to the rise in asthma attacks in children in early September. The first relates to exposure to allergens, especially dust mites. “Ninety percent of asthmatic children are allergic,” said Frédéric le Guillou, MD, respiratory medicine specialist and chair of the French Society for Respiratory Health, an organization aimed at patients and health care professionals. “Don’t forget that asthma is the leading chronic condition in childhood, with a prevalence estimated at between 8% and 10% of children and adolescents. So, we’re talking about considerable numbers of children being affected.”
Although dust mites are a year-round problem, their peak period of reproduction mainly occurs during the wetter months (March to April and September to November). This means that there is a risk of relapse in asthmatic children who are allergic to dust mites when school starts again after the summer holidays. “In such children, any signs of unmanageable allergic rhinitis should be examined,” said Dr. Amat, “these signs being permanent nasal congestion, runny nose, et cetera.”
Finally, we can also add “the stress and anxiety generated by the school setting and settling back into a routine” to the list of likely explanations for this peak in asthma attacks, Dr. Amat concluded.
Check-up time
Children and teenagers with asthma should have a check-up with their respiratory medicine specialists at the start of the new term to confirm that their condition is under control and to determine whether any changes need to be made to their maintenance therapy. “Looking back at previous Septembers and winters is informative in adapting a patient’s treatment plan,” said Dr. Amat. “If maintenance therapy has been stopped during the summer, take the opportunity to represcribe it or modify it if, for example, the dose of inhaled corticosteroids has not been enough to prevent attacks in years gone by. Adequate control of symptoms over the summer months suggests that treatment should be bolstered with preventive therapy to cope with the autumn and winter months. Finally, the factors aggravating poor management of asthma should be dealt with, such as intranasal antihistamines and corticosteroids in allergic rhinitis, specific immunotherapy in patients with controlled asthma but with significant allergy symptoms.”
The start-of-term visit to the doctor’s office is also the perfect opportunity to carry out respiratory function testing (RFT), if this has not been done for over a year in patients whose asthma is well managed. “RFT is indicated in the 3 months following any changes to maintenance therapy, every 3 to 6 months in patients with poorly controlled asthma, and after stopping maintenance therapy or when considering stopping treatment permanently or for an extended period of time,” noted Dr. Amat.
The distinction between difficult asthma (suboptimal treatment plan, poor compliance, persisting allergen exposure, etc.) and severe asthma may be made during this back-to-school asthma review. In specialist clinics, children with severe asthma (not controlled by combined treatment with maximum-dose corticosteroids and maximum-dose bronchodilators) may, like adults, benefit from some biotherapies.
Commentary from Madiha Ellaffi, MD, respiratory medicine specialist
When children experience relatively calm summers without seasonal summer allergies to certain pollens or molds (such as Alternaria, some grasses, etc.) that require maintenance therapy to be continued, we know full well that compliance is often left up to the child. What would be better would be striking a “deal” with these young people: Maintenance treatment can be stopped over the summer, providing that their usual dose is quite low or their asthma is considered mild to moderate, but it must be restarted before going back to school in September. An action plan should be discussed in the event of an asthma attack, and treatment bolstered to overcome this hurdle, should it occur, such as double inhaled corticosteroid doses, etc. Indeed, this period is conducive to asthma exacerbations due to stress, the return of students to confined classrooms, pollutants released by the deep cleaning of school buildings that occurs at the start of term (particularly the release of volatile organic compounds that irritate the airways), and the lack of ventilation in classrooms, which is conducive to the spread of viruses that can cause worsening asthma symptoms. I’d also like to remind parents of the importance of detecting early symptoms (such as wheezing, cough, bronchitis, itchy throat and nose, etc.) in warding off asthma attacks or severe symptoms. I insist on basic measures, such as nasal irrigation, treating allergic rhinitis, which can exacerbate asthma, and ensuring good habits at home to prevent dust mites and mold, such as vacuuming, airing houses, etc. It is sensible to assess the risk of asthma attacks at the start of term according to the child’s allergy profile and their previous history, like starting treatment for allergic rhinitis if not already being taken.
This article was translated from Medscape’s French edition. A version of this article appeared on Medscape.com.
The years go by, and nothing much changes: The first 2 weeks of the new school year have brought with them a rise in emergency department (ED) admissions for asthma in patients under age 15 years. A more relaxed approach to maintenance therapy for the condition over the summer holidays, exposure to allergens at school, and the surge in viral respiratory infections that accompanies the return to group settings explain this trend, which can be foreseen.
According to Public Health France, which has just relaunched its epidemiological monitoring, these cases reach their peak around 2 weeks after the start of the new term.
In its first weekly review on Aug. 22, 2023, the authority reported a slight uptick in cases in its Indian Ocean overseas departments, and the calm before the storm in mainland France.
Last year, between weeks 35 and 36, the increases were 82% for SOS Médecins (the French home doctor visit service), 169% for EDs, and 33% for hospital admissions.
These data are similar to the figures obtained over the past 3 years. The authors of this monitoring, using the SurSaUD system, France’s program for monitoring emergency cases and deaths, attribute these increases to the surge in viral respiratory infections seen after the return to group settings after the school summer holidays.
Indeed, viral-induced exacerbations are mostly caused by rhinoviruses, which circulate throughout the year, but more so during the autumn and winter months. These are probably the main culprits behind the epidemics seen once schools have reopened. Yet relaxation of maintenance asthma treatment (inhaled corticosteroids alone or in combination with long-acting bronchodilators) during the summer holidays also plays a significant role in this yearly recurrence.
Compliance ends with school
Flore Amat, MD, PhD, pediatric respiratory and allergy specialist and coordinating doctor at the Zephyr asthma clinic (Robert-Debré Hospital, Paris Public Hospitals) acknowledged, “The summer holidays are often a time when compliance with maintenance therapy is relaxed.” Aware of this fact, doctors prefer to strike a deal with their young patients. “For some of our young and teenage asthma patients, we support their relaxed approach to medication during the summer holidays,” she admitted. “In July and August, there are fewer viruses circulating, and the weather is often dry, which limits the risk of an asthma attack, meaning we can ease off the maintenance therapy, or even stop taking it altogether. We tell parents and children to start taking them again 2 weeks before school starts; 2 weeks being the minimum time needed for inhaled corticosteroids to start taking effect again.” Unsurprisingly, some forget to do so or simply don’t.
Two other things contribute to the rise in asthma attacks in children in early September. The first relates to exposure to allergens, especially dust mites. “Ninety percent of asthmatic children are allergic,” said Frédéric le Guillou, MD, respiratory medicine specialist and chair of the French Society for Respiratory Health, an organization aimed at patients and health care professionals. “Don’t forget that asthma is the leading chronic condition in childhood, with a prevalence estimated at between 8% and 10% of children and adolescents. So, we’re talking about considerable numbers of children being affected.”
Although dust mites are a year-round problem, their peak period of reproduction mainly occurs during the wetter months (March to April and September to November). This means that there is a risk of relapse in asthmatic children who are allergic to dust mites when school starts again after the summer holidays. “In such children, any signs of unmanageable allergic rhinitis should be examined,” said Dr. Amat, “these signs being permanent nasal congestion, runny nose, et cetera.”
Finally, we can also add “the stress and anxiety generated by the school setting and settling back into a routine” to the list of likely explanations for this peak in asthma attacks, Dr. Amat concluded.
Check-up time
Children and teenagers with asthma should have a check-up with their respiratory medicine specialists at the start of the new term to confirm that their condition is under control and to determine whether any changes need to be made to their maintenance therapy. “Looking back at previous Septembers and winters is informative in adapting a patient’s treatment plan,” said Dr. Amat. “If maintenance therapy has been stopped during the summer, take the opportunity to represcribe it or modify it if, for example, the dose of inhaled corticosteroids has not been enough to prevent attacks in years gone by. Adequate control of symptoms over the summer months suggests that treatment should be bolstered with preventive therapy to cope with the autumn and winter months. Finally, the factors aggravating poor management of asthma should be dealt with, such as intranasal antihistamines and corticosteroids in allergic rhinitis, specific immunotherapy in patients with controlled asthma but with significant allergy symptoms.”
The start-of-term visit to the doctor’s office is also the perfect opportunity to carry out respiratory function testing (RFT), if this has not been done for over a year in patients whose asthma is well managed. “RFT is indicated in the 3 months following any changes to maintenance therapy, every 3 to 6 months in patients with poorly controlled asthma, and after stopping maintenance therapy or when considering stopping treatment permanently or for an extended period of time,” noted Dr. Amat.
The distinction between difficult asthma (suboptimal treatment plan, poor compliance, persisting allergen exposure, etc.) and severe asthma may be made during this back-to-school asthma review. In specialist clinics, children with severe asthma (not controlled by combined treatment with maximum-dose corticosteroids and maximum-dose bronchodilators) may, like adults, benefit from some biotherapies.
Commentary from Madiha Ellaffi, MD, respiratory medicine specialist
When children experience relatively calm summers without seasonal summer allergies to certain pollens or molds (such as Alternaria, some grasses, etc.) that require maintenance therapy to be continued, we know full well that compliance is often left up to the child. What would be better would be striking a “deal” with these young people: Maintenance treatment can be stopped over the summer, providing that their usual dose is quite low or their asthma is considered mild to moderate, but it must be restarted before going back to school in September. An action plan should be discussed in the event of an asthma attack, and treatment bolstered to overcome this hurdle, should it occur, such as double inhaled corticosteroid doses, etc. Indeed, this period is conducive to asthma exacerbations due to stress, the return of students to confined classrooms, pollutants released by the deep cleaning of school buildings that occurs at the start of term (particularly the release of volatile organic compounds that irritate the airways), and the lack of ventilation in classrooms, which is conducive to the spread of viruses that can cause worsening asthma symptoms. I’d also like to remind parents of the importance of detecting early symptoms (such as wheezing, cough, bronchitis, itchy throat and nose, etc.) in warding off asthma attacks or severe symptoms. I insist on basic measures, such as nasal irrigation, treating allergic rhinitis, which can exacerbate asthma, and ensuring good habits at home to prevent dust mites and mold, such as vacuuming, airing houses, etc. It is sensible to assess the risk of asthma attacks at the start of term according to the child’s allergy profile and their previous history, like starting treatment for allergic rhinitis if not already being taken.
This article was translated from Medscape’s French edition. A version of this article appeared on Medscape.com.
The years go by, and nothing much changes: The first 2 weeks of the new school year have brought with them a rise in emergency department (ED) admissions for asthma in patients under age 15 years. A more relaxed approach to maintenance therapy for the condition over the summer holidays, exposure to allergens at school, and the surge in viral respiratory infections that accompanies the return to group settings explain this trend, which can be foreseen.
According to Public Health France, which has just relaunched its epidemiological monitoring, these cases reach their peak around 2 weeks after the start of the new term.
In its first weekly review on Aug. 22, 2023, the authority reported a slight uptick in cases in its Indian Ocean overseas departments, and the calm before the storm in mainland France.
Last year, between weeks 35 and 36, the increases were 82% for SOS Médecins (the French home doctor visit service), 169% for EDs, and 33% for hospital admissions.
These data are similar to the figures obtained over the past 3 years. The authors of this monitoring, using the SurSaUD system, France’s program for monitoring emergency cases and deaths, attribute these increases to the surge in viral respiratory infections seen after the return to group settings after the school summer holidays.
Indeed, viral-induced exacerbations are mostly caused by rhinoviruses, which circulate throughout the year, but more so during the autumn and winter months. These are probably the main culprits behind the epidemics seen once schools have reopened. Yet relaxation of maintenance asthma treatment (inhaled corticosteroids alone or in combination with long-acting bronchodilators) during the summer holidays also plays a significant role in this yearly recurrence.
Compliance ends with school
Flore Amat, MD, PhD, pediatric respiratory and allergy specialist and coordinating doctor at the Zephyr asthma clinic (Robert-Debré Hospital, Paris Public Hospitals) acknowledged, “The summer holidays are often a time when compliance with maintenance therapy is relaxed.” Aware of this fact, doctors prefer to strike a deal with their young patients. “For some of our young and teenage asthma patients, we support their relaxed approach to medication during the summer holidays,” she admitted. “In July and August, there are fewer viruses circulating, and the weather is often dry, which limits the risk of an asthma attack, meaning we can ease off the maintenance therapy, or even stop taking it altogether. We tell parents and children to start taking them again 2 weeks before school starts; 2 weeks being the minimum time needed for inhaled corticosteroids to start taking effect again.” Unsurprisingly, some forget to do so or simply don’t.
Two other things contribute to the rise in asthma attacks in children in early September. The first relates to exposure to allergens, especially dust mites. “Ninety percent of asthmatic children are allergic,” said Frédéric le Guillou, MD, respiratory medicine specialist and chair of the French Society for Respiratory Health, an organization aimed at patients and health care professionals. “Don’t forget that asthma is the leading chronic condition in childhood, with a prevalence estimated at between 8% and 10% of children and adolescents. So, we’re talking about considerable numbers of children being affected.”
Although dust mites are a year-round problem, their peak period of reproduction mainly occurs during the wetter months (March to April and September to November). This means that there is a risk of relapse in asthmatic children who are allergic to dust mites when school starts again after the summer holidays. “In such children, any signs of unmanageable allergic rhinitis should be examined,” said Dr. Amat, “these signs being permanent nasal congestion, runny nose, et cetera.”
Finally, we can also add “the stress and anxiety generated by the school setting and settling back into a routine” to the list of likely explanations for this peak in asthma attacks, Dr. Amat concluded.
Check-up time
Children and teenagers with asthma should have a check-up with their respiratory medicine specialists at the start of the new term to confirm that their condition is under control and to determine whether any changes need to be made to their maintenance therapy. “Looking back at previous Septembers and winters is informative in adapting a patient’s treatment plan,” said Dr. Amat. “If maintenance therapy has been stopped during the summer, take the opportunity to represcribe it or modify it if, for example, the dose of inhaled corticosteroids has not been enough to prevent attacks in years gone by. Adequate control of symptoms over the summer months suggests that treatment should be bolstered with preventive therapy to cope with the autumn and winter months. Finally, the factors aggravating poor management of asthma should be dealt with, such as intranasal antihistamines and corticosteroids in allergic rhinitis, specific immunotherapy in patients with controlled asthma but with significant allergy symptoms.”
The start-of-term visit to the doctor’s office is also the perfect opportunity to carry out respiratory function testing (RFT), if this has not been done for over a year in patients whose asthma is well managed. “RFT is indicated in the 3 months following any changes to maintenance therapy, every 3 to 6 months in patients with poorly controlled asthma, and after stopping maintenance therapy or when considering stopping treatment permanently or for an extended period of time,” noted Dr. Amat.
The distinction between difficult asthma (suboptimal treatment plan, poor compliance, persisting allergen exposure, etc.) and severe asthma may be made during this back-to-school asthma review. In specialist clinics, children with severe asthma (not controlled by combined treatment with maximum-dose corticosteroids and maximum-dose bronchodilators) may, like adults, benefit from some biotherapies.
Commentary from Madiha Ellaffi, MD, respiratory medicine specialist
When children experience relatively calm summers without seasonal summer allergies to certain pollens or molds (such as Alternaria, some grasses, etc.) that require maintenance therapy to be continued, we know full well that compliance is often left up to the child. What would be better would be striking a “deal” with these young people: Maintenance treatment can be stopped over the summer, providing that their usual dose is quite low or their asthma is considered mild to moderate, but it must be restarted before going back to school in September. An action plan should be discussed in the event of an asthma attack, and treatment bolstered to overcome this hurdle, should it occur, such as double inhaled corticosteroid doses, etc. Indeed, this period is conducive to asthma exacerbations due to stress, the return of students to confined classrooms, pollutants released by the deep cleaning of school buildings that occurs at the start of term (particularly the release of volatile organic compounds that irritate the airways), and the lack of ventilation in classrooms, which is conducive to the spread of viruses that can cause worsening asthma symptoms. I’d also like to remind parents of the importance of detecting early symptoms (such as wheezing, cough, bronchitis, itchy throat and nose, etc.) in warding off asthma attacks or severe symptoms. I insist on basic measures, such as nasal irrigation, treating allergic rhinitis, which can exacerbate asthma, and ensuring good habits at home to prevent dust mites and mold, such as vacuuming, airing houses, etc. It is sensible to assess the risk of asthma attacks at the start of term according to the child’s allergy profile and their previous history, like starting treatment for allergic rhinitis if not already being taken.
This article was translated from Medscape’s French edition. A version of this article appeared on Medscape.com.
Minimally invasive surfactant shows some benefit in infants’ first 2 years
Results of the OPTIMIST follow-up study were published online in JAMA.
Researchers, led by Peter A. Dargaville, MD, department of paediatrics, Royal Hobart (Australia) Hospital, found that MIST, which involves administering surfactant via a thin catheter, compared with sham treatment, did not reduce the incidence of death or neurodevelopmental disability (NDD) by 2 years of age.
However, infants who received MIST had lower rates of poor respiratory outcomes during those first 2 years of life.
Study spanned 11 countries
The study was conducted in 33 tertiary neonatal intensive care units (NICUs) in 11 countries, including Australia, Canada, Israel, New Zealand, Qatar, Singapore, Slovenia, the Netherlands, Turkey, the United Kingdom, and the United States.
It included 486 infants 25-28 weeks old supported with CPAP; 453 had follow-up data available and data on the key secondary outcome were available for 434 infants.
The sham treatment consisted of only transient repositioning without airway instruments. Treating clinicians, outcome assessors, and parents were blinded to group status.
No significant difference in deaths, NDD
Death or NDD occurred in 36.3% of the patients in the MIST group and 36.1% in the control group (risk difference, 0%; 95% confidence interval, −7.6% to 7.7%; relative risk, 1.0; 95% confidence interval, 0.81-1.24).
Secondary respiratory outcomes were better in the MIST group:
- Hospitalization with respiratory illness occurred in 25.1% in the MIST group versus 38.2% in the control group (RR, 0.66; 95% CI, 0.54-0.81).
- Parent-reported wheezing or breathing difficulty occurred in 40.6% in the MIST group versus 53.6% in controls (RR, 0.76; 95% CI, 0.63-0.90).
- Asthma diagnosed by a physician was reported in 4.4% and 11.9% of MIST and control-group infants, respectively.
- Reported use of inhaled relievers (beta2 agonists) was 23.9% in the MIST group versus 38.7% in controls.
The previous study of early outcomes of deaths or bronchopulmonary dysplasia (BPD; chronic lung injury in preterm infants) was published by the same group of researchers in 2021.
Important benefit for respiratory health
Suhas G. Kallapur, MD, chief of the divisions of neonatology and developmental biology at University of California, Los Angeles, who was not part of either study, said: “This is one of the largest studies to date examining whether the MIST procedure for surfactant is beneficial in preterm babies born at 25-28 weeks’ gestation.”
Overall, when considering the 2021 and 2023 studies together, it appears that the MIST therapy has important benefits for respiratory health during a NICU stay and in early infancy, even though the primary outcome of death or NDD was not different between the treatment and control groups, Dr. Kallapur said.
“The slight (nonsignificant) increase in deaths in the MIST group was confined to the more immature babies – 25-26 weeks’ gestation at birth,” he pointed out. “In the bigger and more mature babies – 27-28 week gestation infants – the benefits of MIST therapy occurred without any increase in mortality, suggesting that this group of babies may be the group that stands to benefit most from this therapy.”
Dr. Kallapur said new data in the developmental origins of health and disease “now show that the trajectory of respiratory health in infancy is an important determinant of respiratory health into adulthood and older age.”
Therefore, the finding of benefit to respiratory health is particularly important, he said.
He noted that MIST or similar therapy is already in use in many NICUs throughout the world and that those already using it will likely feel vindicated by this study.
“Neonatologists who were on the sidelines will likely see these results – especially childhood respiratory outcomes – as a reason to initiate this procedure in all but the most immature preterm infants,” Dr. Kallapur says.
Dr. Dargaville reports personal fees from AbbVie and Chiesi Farmaceutici and provision of surfactant at reduced cost and support for conference travel from Chiesi Farmaceutici during the conduct of the study; in addition, Dr. Dargaville has been issued a patent for a catheter design. One coauthor reports grants from Chiesi Farmaceutici during the conduct of the study. Another coauthor reports serving as chief investigator for OPTI-SURF, an observational study on United Kingdom neonatal surfactant use in respiratory distress syndrome funded by Chiesi UK outside the submitted work. A third coauthor reports personal fees from Chiesi Farmaceutici outside the submitted work. This study was funded by grants from the Royal Hobart Hospital Research Foundation and the Australian National Health and Medical Research Council. Exogenous surfactant was provided at reduced cost by Chiesi Farmaceutici. Dr. Kallapur has no relevant financial relationships.
Results of the OPTIMIST follow-up study were published online in JAMA.
Researchers, led by Peter A. Dargaville, MD, department of paediatrics, Royal Hobart (Australia) Hospital, found that MIST, which involves administering surfactant via a thin catheter, compared with sham treatment, did not reduce the incidence of death or neurodevelopmental disability (NDD) by 2 years of age.
However, infants who received MIST had lower rates of poor respiratory outcomes during those first 2 years of life.
Study spanned 11 countries
The study was conducted in 33 tertiary neonatal intensive care units (NICUs) in 11 countries, including Australia, Canada, Israel, New Zealand, Qatar, Singapore, Slovenia, the Netherlands, Turkey, the United Kingdom, and the United States.
It included 486 infants 25-28 weeks old supported with CPAP; 453 had follow-up data available and data on the key secondary outcome were available for 434 infants.
The sham treatment consisted of only transient repositioning without airway instruments. Treating clinicians, outcome assessors, and parents were blinded to group status.
No significant difference in deaths, NDD
Death or NDD occurred in 36.3% of the patients in the MIST group and 36.1% in the control group (risk difference, 0%; 95% confidence interval, −7.6% to 7.7%; relative risk, 1.0; 95% confidence interval, 0.81-1.24).
Secondary respiratory outcomes were better in the MIST group:
- Hospitalization with respiratory illness occurred in 25.1% in the MIST group versus 38.2% in the control group (RR, 0.66; 95% CI, 0.54-0.81).
- Parent-reported wheezing or breathing difficulty occurred in 40.6% in the MIST group versus 53.6% in controls (RR, 0.76; 95% CI, 0.63-0.90).
- Asthma diagnosed by a physician was reported in 4.4% and 11.9% of MIST and control-group infants, respectively.
- Reported use of inhaled relievers (beta2 agonists) was 23.9% in the MIST group versus 38.7% in controls.
The previous study of early outcomes of deaths or bronchopulmonary dysplasia (BPD; chronic lung injury in preterm infants) was published by the same group of researchers in 2021.
Important benefit for respiratory health
Suhas G. Kallapur, MD, chief of the divisions of neonatology and developmental biology at University of California, Los Angeles, who was not part of either study, said: “This is one of the largest studies to date examining whether the MIST procedure for surfactant is beneficial in preterm babies born at 25-28 weeks’ gestation.”
Overall, when considering the 2021 and 2023 studies together, it appears that the MIST therapy has important benefits for respiratory health during a NICU stay and in early infancy, even though the primary outcome of death or NDD was not different between the treatment and control groups, Dr. Kallapur said.
“The slight (nonsignificant) increase in deaths in the MIST group was confined to the more immature babies – 25-26 weeks’ gestation at birth,” he pointed out. “In the bigger and more mature babies – 27-28 week gestation infants – the benefits of MIST therapy occurred without any increase in mortality, suggesting that this group of babies may be the group that stands to benefit most from this therapy.”
Dr. Kallapur said new data in the developmental origins of health and disease “now show that the trajectory of respiratory health in infancy is an important determinant of respiratory health into adulthood and older age.”
Therefore, the finding of benefit to respiratory health is particularly important, he said.
He noted that MIST or similar therapy is already in use in many NICUs throughout the world and that those already using it will likely feel vindicated by this study.
“Neonatologists who were on the sidelines will likely see these results – especially childhood respiratory outcomes – as a reason to initiate this procedure in all but the most immature preterm infants,” Dr. Kallapur says.
Dr. Dargaville reports personal fees from AbbVie and Chiesi Farmaceutici and provision of surfactant at reduced cost and support for conference travel from Chiesi Farmaceutici during the conduct of the study; in addition, Dr. Dargaville has been issued a patent for a catheter design. One coauthor reports grants from Chiesi Farmaceutici during the conduct of the study. Another coauthor reports serving as chief investigator for OPTI-SURF, an observational study on United Kingdom neonatal surfactant use in respiratory distress syndrome funded by Chiesi UK outside the submitted work. A third coauthor reports personal fees from Chiesi Farmaceutici outside the submitted work. This study was funded by grants from the Royal Hobart Hospital Research Foundation and the Australian National Health and Medical Research Council. Exogenous surfactant was provided at reduced cost by Chiesi Farmaceutici. Dr. Kallapur has no relevant financial relationships.
Results of the OPTIMIST follow-up study were published online in JAMA.
Researchers, led by Peter A. Dargaville, MD, department of paediatrics, Royal Hobart (Australia) Hospital, found that MIST, which involves administering surfactant via a thin catheter, compared with sham treatment, did not reduce the incidence of death or neurodevelopmental disability (NDD) by 2 years of age.
However, infants who received MIST had lower rates of poor respiratory outcomes during those first 2 years of life.
Study spanned 11 countries
The study was conducted in 33 tertiary neonatal intensive care units (NICUs) in 11 countries, including Australia, Canada, Israel, New Zealand, Qatar, Singapore, Slovenia, the Netherlands, Turkey, the United Kingdom, and the United States.
It included 486 infants 25-28 weeks old supported with CPAP; 453 had follow-up data available and data on the key secondary outcome were available for 434 infants.
The sham treatment consisted of only transient repositioning without airway instruments. Treating clinicians, outcome assessors, and parents were blinded to group status.
No significant difference in deaths, NDD
Death or NDD occurred in 36.3% of the patients in the MIST group and 36.1% in the control group (risk difference, 0%; 95% confidence interval, −7.6% to 7.7%; relative risk, 1.0; 95% confidence interval, 0.81-1.24).
Secondary respiratory outcomes were better in the MIST group:
- Hospitalization with respiratory illness occurred in 25.1% in the MIST group versus 38.2% in the control group (RR, 0.66; 95% CI, 0.54-0.81).
- Parent-reported wheezing or breathing difficulty occurred in 40.6% in the MIST group versus 53.6% in controls (RR, 0.76; 95% CI, 0.63-0.90).
- Asthma diagnosed by a physician was reported in 4.4% and 11.9% of MIST and control-group infants, respectively.
- Reported use of inhaled relievers (beta2 agonists) was 23.9% in the MIST group versus 38.7% in controls.
The previous study of early outcomes of deaths or bronchopulmonary dysplasia (BPD; chronic lung injury in preterm infants) was published by the same group of researchers in 2021.
Important benefit for respiratory health
Suhas G. Kallapur, MD, chief of the divisions of neonatology and developmental biology at University of California, Los Angeles, who was not part of either study, said: “This is one of the largest studies to date examining whether the MIST procedure for surfactant is beneficial in preterm babies born at 25-28 weeks’ gestation.”
Overall, when considering the 2021 and 2023 studies together, it appears that the MIST therapy has important benefits for respiratory health during a NICU stay and in early infancy, even though the primary outcome of death or NDD was not different between the treatment and control groups, Dr. Kallapur said.
“The slight (nonsignificant) increase in deaths in the MIST group was confined to the more immature babies – 25-26 weeks’ gestation at birth,” he pointed out. “In the bigger and more mature babies – 27-28 week gestation infants – the benefits of MIST therapy occurred without any increase in mortality, suggesting that this group of babies may be the group that stands to benefit most from this therapy.”
Dr. Kallapur said new data in the developmental origins of health and disease “now show that the trajectory of respiratory health in infancy is an important determinant of respiratory health into adulthood and older age.”
Therefore, the finding of benefit to respiratory health is particularly important, he said.
He noted that MIST or similar therapy is already in use in many NICUs throughout the world and that those already using it will likely feel vindicated by this study.
“Neonatologists who were on the sidelines will likely see these results – especially childhood respiratory outcomes – as a reason to initiate this procedure in all but the most immature preterm infants,” Dr. Kallapur says.
Dr. Dargaville reports personal fees from AbbVie and Chiesi Farmaceutici and provision of surfactant at reduced cost and support for conference travel from Chiesi Farmaceutici during the conduct of the study; in addition, Dr. Dargaville has been issued a patent for a catheter design. One coauthor reports grants from Chiesi Farmaceutici during the conduct of the study. Another coauthor reports serving as chief investigator for OPTI-SURF, an observational study on United Kingdom neonatal surfactant use in respiratory distress syndrome funded by Chiesi UK outside the submitted work. A third coauthor reports personal fees from Chiesi Farmaceutici outside the submitted work. This study was funded by grants from the Royal Hobart Hospital Research Foundation and the Australian National Health and Medical Research Council. Exogenous surfactant was provided at reduced cost by Chiesi Farmaceutici. Dr. Kallapur has no relevant financial relationships.
FROM JAMA
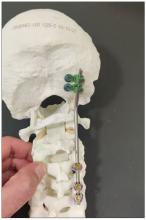
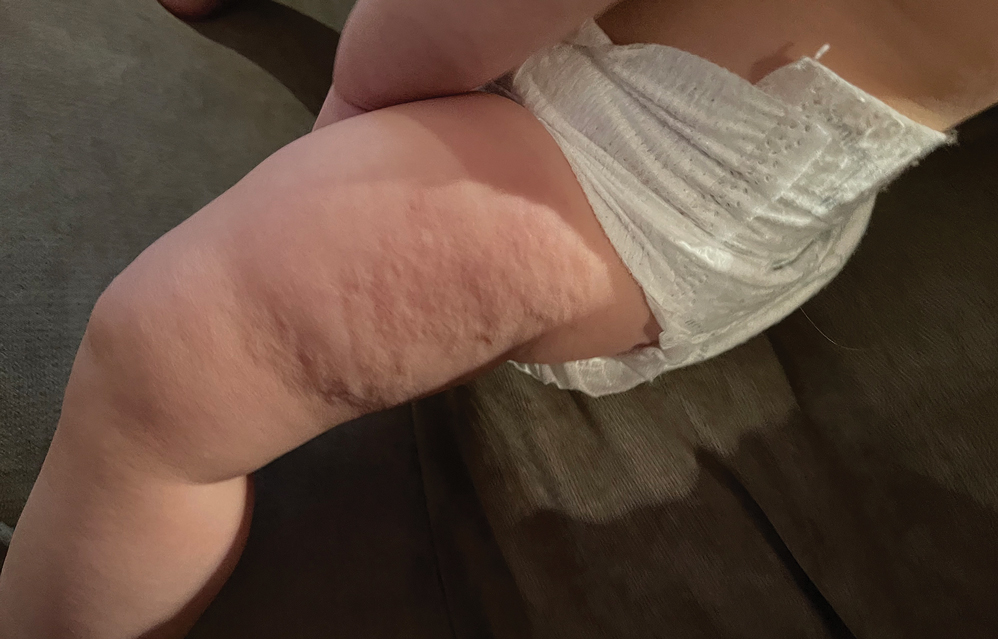
Transient Skin Rippling in an Infant
The Diagnosis: Infantile Transient Smooth Muscle Contraction of the Skin
A diagnosis of infantile transient smooth muscle contraction of the skin (ITSMC) was made based on our patient’s clinical presentation and eliminating the diagnoses in the differential. No treatment ultimately was indicated, as episodes became less frequent over time.
The term infantile transient smooth muscle contraction of the skin was first proposed in 2013 by Torrelo et al,1 who described 9 newborns with episodic skin rippling occasionally associated with exposure to cold or friction. The authors postulated that ITSMC was the result of a transient contraction of the arrector pili smooth muscle fibers of the skin, secondary to autonomic immaturity, primitive reflexes, or smooth muscle hypersensitivity.1 Since this first description, ITSMC has remained a rarely reported and poorly understood phenomenon with rare identified cases in the literature.2,3 Clinical history and examination of infants with intermittent transient skin rippling help to distinguish ITSMC from other diagnoses without the need for biopsy, which is particularly undesirable in the pediatric population.
Congenital smooth muscle hamartoma is a benign proliferation of mature smooth muscle that also can arise from the arrector pili muscles.4 In contrast to ITSMC, a hamartoma does not clear; rather, it persists and grows proportionally with the child and is associated with overlying hyperpigmentation and hypertrichosis. The transient nature of ITSMC may be worrisome for mastocytoma; however, this condition presents as erythematous, yellow, red, or brown macules, papules, plaques, or nodules with a positive Darier sign.5 Although the differential diagnosis includes the shagreen patch characteristic of tuberous sclerosis, this irregular plaque typically is located on the lower back with overlying peau d’orange skin changes, and our patient lacked other features indicative of this condition.6 Becker nevus also remains a consideration in patients with rippled skin, but this entity typically becomes more notable at puberty and is associated with hyperpigmentation and hypertrichosis and is a type of smooth muscle hamartoma.4
Our case highlighted the unusual presentation of ITSMC, a condition that can easily go unrecognized, leading to unnecessary referrals and concern. Familiarity with this benign diagnosis is essential to inform prognosis and guide management.
- Torrelo A, Moreno S, Castro C, et al. Infantile transient smooth muscle contraction of the skin. J Am Acad Dermatol. 2013;69:498-500. doi:10.1016/j.jaad.2013.04.029
- Theodosiou G, Belfrage E, Berggård K, et al. Infantile transient smooth muscle contraction of the skin: a case report and literature review. Eur J Dermatol. 2021;31:260-261. doi:10.1684/ejd.2021.3996
- Topham C, Deacon DC, Bowen A, et al. More than goosebumps: a case of marked skin dimpling in an infant. Pediatr Dermatol. 2019;36:E71-E72. doi:10.1111/pde.13791
- Raboudi A, Litaiem N. Congenital smooth muscle hamartoma. StatPearls. StatPearls Publishing; 2022.
- Leung AKC, Lam JM, Leong KF. Childhood solitary cutaneous mastocytoma: clinical manifestations, diagnosis, evaluation, and management. Curr Pediatr Rev. 2019;15:42-46. doi:10.2174/1573396315666 181120163952
- Bongiorno MA, Nathan N, Oyerinde O, et al. Clinical characteristics of connective tissue nevi in tuberous sclerosis complex with special emphasis on shagreen patches. JAMA Dermatol. 2017;153:660-665. doi:10.1001/jamadermatol.2017.0298
The Diagnosis: Infantile Transient Smooth Muscle Contraction of the Skin
A diagnosis of infantile transient smooth muscle contraction of the skin (ITSMC) was made based on our patient’s clinical presentation and eliminating the diagnoses in the differential. No treatment ultimately was indicated, as episodes became less frequent over time.
The term infantile transient smooth muscle contraction of the skin was first proposed in 2013 by Torrelo et al,1 who described 9 newborns with episodic skin rippling occasionally associated with exposure to cold or friction. The authors postulated that ITSMC was the result of a transient contraction of the arrector pili smooth muscle fibers of the skin, secondary to autonomic immaturity, primitive reflexes, or smooth muscle hypersensitivity.1 Since this first description, ITSMC has remained a rarely reported and poorly understood phenomenon with rare identified cases in the literature.2,3 Clinical history and examination of infants with intermittent transient skin rippling help to distinguish ITSMC from other diagnoses without the need for biopsy, which is particularly undesirable in the pediatric population.
Congenital smooth muscle hamartoma is a benign proliferation of mature smooth muscle that also can arise from the arrector pili muscles.4 In contrast to ITSMC, a hamartoma does not clear; rather, it persists and grows proportionally with the child and is associated with overlying hyperpigmentation and hypertrichosis. The transient nature of ITSMC may be worrisome for mastocytoma; however, this condition presents as erythematous, yellow, red, or brown macules, papules, plaques, or nodules with a positive Darier sign.5 Although the differential diagnosis includes the shagreen patch characteristic of tuberous sclerosis, this irregular plaque typically is located on the lower back with overlying peau d’orange skin changes, and our patient lacked other features indicative of this condition.6 Becker nevus also remains a consideration in patients with rippled skin, but this entity typically becomes more notable at puberty and is associated with hyperpigmentation and hypertrichosis and is a type of smooth muscle hamartoma.4
Our case highlighted the unusual presentation of ITSMC, a condition that can easily go unrecognized, leading to unnecessary referrals and concern. Familiarity with this benign diagnosis is essential to inform prognosis and guide management.
The Diagnosis: Infantile Transient Smooth Muscle Contraction of the Skin
A diagnosis of infantile transient smooth muscle contraction of the skin (ITSMC) was made based on our patient’s clinical presentation and eliminating the diagnoses in the differential. No treatment ultimately was indicated, as episodes became less frequent over time.
The term infantile transient smooth muscle contraction of the skin was first proposed in 2013 by Torrelo et al,1 who described 9 newborns with episodic skin rippling occasionally associated with exposure to cold or friction. The authors postulated that ITSMC was the result of a transient contraction of the arrector pili smooth muscle fibers of the skin, secondary to autonomic immaturity, primitive reflexes, or smooth muscle hypersensitivity.1 Since this first description, ITSMC has remained a rarely reported and poorly understood phenomenon with rare identified cases in the literature.2,3 Clinical history and examination of infants with intermittent transient skin rippling help to distinguish ITSMC from other diagnoses without the need for biopsy, which is particularly undesirable in the pediatric population.
Congenital smooth muscle hamartoma is a benign proliferation of mature smooth muscle that also can arise from the arrector pili muscles.4 In contrast to ITSMC, a hamartoma does not clear; rather, it persists and grows proportionally with the child and is associated with overlying hyperpigmentation and hypertrichosis. The transient nature of ITSMC may be worrisome for mastocytoma; however, this condition presents as erythematous, yellow, red, or brown macules, papules, plaques, or nodules with a positive Darier sign.5 Although the differential diagnosis includes the shagreen patch characteristic of tuberous sclerosis, this irregular plaque typically is located on the lower back with overlying peau d’orange skin changes, and our patient lacked other features indicative of this condition.6 Becker nevus also remains a consideration in patients with rippled skin, but this entity typically becomes more notable at puberty and is associated with hyperpigmentation and hypertrichosis and is a type of smooth muscle hamartoma.4
Our case highlighted the unusual presentation of ITSMC, a condition that can easily go unrecognized, leading to unnecessary referrals and concern. Familiarity with this benign diagnosis is essential to inform prognosis and guide management.
- Torrelo A, Moreno S, Castro C, et al. Infantile transient smooth muscle contraction of the skin. J Am Acad Dermatol. 2013;69:498-500. doi:10.1016/j.jaad.2013.04.029
- Theodosiou G, Belfrage E, Berggård K, et al. Infantile transient smooth muscle contraction of the skin: a case report and literature review. Eur J Dermatol. 2021;31:260-261. doi:10.1684/ejd.2021.3996
- Topham C, Deacon DC, Bowen A, et al. More than goosebumps: a case of marked skin dimpling in an infant. Pediatr Dermatol. 2019;36:E71-E72. doi:10.1111/pde.13791
- Raboudi A, Litaiem N. Congenital smooth muscle hamartoma. StatPearls. StatPearls Publishing; 2022.
- Leung AKC, Lam JM, Leong KF. Childhood solitary cutaneous mastocytoma: clinical manifestations, diagnosis, evaluation, and management. Curr Pediatr Rev. 2019;15:42-46. doi:10.2174/1573396315666 181120163952
- Bongiorno MA, Nathan N, Oyerinde O, et al. Clinical characteristics of connective tissue nevi in tuberous sclerosis complex with special emphasis on shagreen patches. JAMA Dermatol. 2017;153:660-665. doi:10.1001/jamadermatol.2017.0298
- Torrelo A, Moreno S, Castro C, et al. Infantile transient smooth muscle contraction of the skin. J Am Acad Dermatol. 2013;69:498-500. doi:10.1016/j.jaad.2013.04.029
- Theodosiou G, Belfrage E, Berggård K, et al. Infantile transient smooth muscle contraction of the skin: a case report and literature review. Eur J Dermatol. 2021;31:260-261. doi:10.1684/ejd.2021.3996
- Topham C, Deacon DC, Bowen A, et al. More than goosebumps: a case of marked skin dimpling in an infant. Pediatr Dermatol. 2019;36:E71-E72. doi:10.1111/pde.13791
- Raboudi A, Litaiem N. Congenital smooth muscle hamartoma. StatPearls. StatPearls Publishing; 2022.
- Leung AKC, Lam JM, Leong KF. Childhood solitary cutaneous mastocytoma: clinical manifestations, diagnosis, evaluation, and management. Curr Pediatr Rev. 2019;15:42-46. doi:10.2174/1573396315666 181120163952
- Bongiorno MA, Nathan N, Oyerinde O, et al. Clinical characteristics of connective tissue nevi in tuberous sclerosis complex with special emphasis on shagreen patches. JAMA Dermatol. 2017;153:660-665. doi:10.1001/jamadermatol.2017.0298
A healthy, full-term, 5-month-old infant boy presented to dermatology for evaluation of an intermittent, asymptomatic, rippled skin texture of the left thigh that resolved completely between flares. The parents noted fewer than 10 intermittent flares prior to the initial presentation at 5 months. Physical examination of the patient’s skin revealed no epidermal abnormalities, dermatographism, or subcutaneous nodules, and there was no positive Darier sign. A subsequent flare at 9 months of age occurred concurrently with fevers up to 39.4 °C (103 °F), and a corresponding photograph (quiz image) provided by the parents due to the intermittent and transient nature of the condition demonstrated an ill-defined, raised, rippled plaque on the left lateral thigh.
Company submits supplemental NDA for topical atopic dermatitis treatment
in adults and children aged 6 years and older.
Roflumilast cream 0.3% (Zoryve) is currently approved by the FDA for the topical treatment of plaque psoriasis, including intertriginous areas, in patients 12 years of age and older. Submission of the sNDA is based on positive results from the Interventional Trial Evaluating Roflumilast Cream for the Treatment of Atopic Dermatitis (INTEGUMENT-1 and INTEGUMENT-2) trials; two identical Phase 3, vehicle-controlled trials in which roflumilast cream 0.15% or vehicle was applied once daily for 4 weeks to individuals 6 years of age and older with mild to moderate AD involving at least 3% body surface area. Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor.
According to a press release from Arcutis, both studies met the primary endpoint of IGA Success, which was defined as a validated Investigator Global Assessment – Atopic Dermatitis (vIGA-AD) score of ‘clear’ or ‘almost clear’ plus a 2-grade improvement from baseline at week 4. In INTEGUMENT-1 this endpoint was achieved by 32.0% of subjects in the roflumilast cream group vs. 15.2% of those in the vehicle group (P < .0001). In INTEGUMENT-2, this endpoint was achieved by 28.9% of subjects in the roflumilast cream group vs. 12.0% of those in the vehicle group (P < .0001). The most common adverse reactions based on data from the combined trials were headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
in adults and children aged 6 years and older.
Roflumilast cream 0.3% (Zoryve) is currently approved by the FDA for the topical treatment of plaque psoriasis, including intertriginous areas, in patients 12 years of age and older. Submission of the sNDA is based on positive results from the Interventional Trial Evaluating Roflumilast Cream for the Treatment of Atopic Dermatitis (INTEGUMENT-1 and INTEGUMENT-2) trials; two identical Phase 3, vehicle-controlled trials in which roflumilast cream 0.15% or vehicle was applied once daily for 4 weeks to individuals 6 years of age and older with mild to moderate AD involving at least 3% body surface area. Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor.
According to a press release from Arcutis, both studies met the primary endpoint of IGA Success, which was defined as a validated Investigator Global Assessment – Atopic Dermatitis (vIGA-AD) score of ‘clear’ or ‘almost clear’ plus a 2-grade improvement from baseline at week 4. In INTEGUMENT-1 this endpoint was achieved by 32.0% of subjects in the roflumilast cream group vs. 15.2% of those in the vehicle group (P < .0001). In INTEGUMENT-2, this endpoint was achieved by 28.9% of subjects in the roflumilast cream group vs. 12.0% of those in the vehicle group (P < .0001). The most common adverse reactions based on data from the combined trials were headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
in adults and children aged 6 years and older.
Roflumilast cream 0.3% (Zoryve) is currently approved by the FDA for the topical treatment of plaque psoriasis, including intertriginous areas, in patients 12 years of age and older. Submission of the sNDA is based on positive results from the Interventional Trial Evaluating Roflumilast Cream for the Treatment of Atopic Dermatitis (INTEGUMENT-1 and INTEGUMENT-2) trials; two identical Phase 3, vehicle-controlled trials in which roflumilast cream 0.15% or vehicle was applied once daily for 4 weeks to individuals 6 years of age and older with mild to moderate AD involving at least 3% body surface area. Roflumilast is a phosphodiesterase-4 (PDE-4) inhibitor.
According to a press release from Arcutis, both studies met the primary endpoint of IGA Success, which was defined as a validated Investigator Global Assessment – Atopic Dermatitis (vIGA-AD) score of ‘clear’ or ‘almost clear’ plus a 2-grade improvement from baseline at week 4. In INTEGUMENT-1 this endpoint was achieved by 32.0% of subjects in the roflumilast cream group vs. 15.2% of those in the vehicle group (P < .0001). In INTEGUMENT-2, this endpoint was achieved by 28.9% of subjects in the roflumilast cream group vs. 12.0% of those in the vehicle group (P < .0001). The most common adverse reactions based on data from the combined trials were headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
RSV season has started, and this year could be different
The Centers for Disease Control and Prevention issued a national alert to health officials Sept. 5, urging them to offer new medicines that can prevent severe cases of the respiratory virus in very young children and in older people. Those two groups are at the highest risk of potentially deadly complications from RSV.
Typically, the CDC considers the start of RSV season to occur when the rate of positive tests for the virus goes above 3% for 2 consecutive weeks. In Florida, the rate has been around 5% in recent weeks, and in Georgia, there has been an increase in RSV-related hospitalizations. Most of the hospitalizations in Georgia have been among infants less than a year old.
“Historically, such regional increases have predicted the beginning of RSV season nationally, with increased RSV activity spreading north and west over the following 2-3 months,” the CDC said.
Most children have been infected with RSV by the time they are 2 years old. Historically, up to 80,000 children under 5 years old are hospitalized annually because of the virus, and between 100 and 300 die from complications each year.
Those figures could be drastically different this year because new preventive treatments are available.
The CDC recommends that all children under 8 months old receive the newly approved monoclonal antibody treatment nirsevimab (Beyfortus). Children up to 19 months old at high risk of severe complications from RSV are also eligible for the single-dose shot. In clinical trials, the treatment was 80% effective at preventing RSV infections from becoming so severe that children had to be hospitalized. The protection lasted about 5 months.
Older people are also at a heightened risk of severe illness from RSV, and two new vaccines are available this season. The vaccines are called Arexvy and Abrysvo, and the single-dose shots are approved for people ages 60 years and older. They are more than 80% effective at making severe lower respiratory complications less likely.
Last year’s RSV season started during the summer and peaked in October and November, which was earlier than usual. There’s no indication yet of when RSV season may peak this year. Last year and throughout the pandemic, RSV held its historical pattern of starting in Florida.
A version of this article appeared on WebMD.com.
The Centers for Disease Control and Prevention issued a national alert to health officials Sept. 5, urging them to offer new medicines that can prevent severe cases of the respiratory virus in very young children and in older people. Those two groups are at the highest risk of potentially deadly complications from RSV.
Typically, the CDC considers the start of RSV season to occur when the rate of positive tests for the virus goes above 3% for 2 consecutive weeks. In Florida, the rate has been around 5% in recent weeks, and in Georgia, there has been an increase in RSV-related hospitalizations. Most of the hospitalizations in Georgia have been among infants less than a year old.
“Historically, such regional increases have predicted the beginning of RSV season nationally, with increased RSV activity spreading north and west over the following 2-3 months,” the CDC said.
Most children have been infected with RSV by the time they are 2 years old. Historically, up to 80,000 children under 5 years old are hospitalized annually because of the virus, and between 100 and 300 die from complications each year.
Those figures could be drastically different this year because new preventive treatments are available.
The CDC recommends that all children under 8 months old receive the newly approved monoclonal antibody treatment nirsevimab (Beyfortus). Children up to 19 months old at high risk of severe complications from RSV are also eligible for the single-dose shot. In clinical trials, the treatment was 80% effective at preventing RSV infections from becoming so severe that children had to be hospitalized. The protection lasted about 5 months.
Older people are also at a heightened risk of severe illness from RSV, and two new vaccines are available this season. The vaccines are called Arexvy and Abrysvo, and the single-dose shots are approved for people ages 60 years and older. They are more than 80% effective at making severe lower respiratory complications less likely.
Last year’s RSV season started during the summer and peaked in October and November, which was earlier than usual. There’s no indication yet of when RSV season may peak this year. Last year and throughout the pandemic, RSV held its historical pattern of starting in Florida.
A version of this article appeared on WebMD.com.
The Centers for Disease Control and Prevention issued a national alert to health officials Sept. 5, urging them to offer new medicines that can prevent severe cases of the respiratory virus in very young children and in older people. Those two groups are at the highest risk of potentially deadly complications from RSV.
Typically, the CDC considers the start of RSV season to occur when the rate of positive tests for the virus goes above 3% for 2 consecutive weeks. In Florida, the rate has been around 5% in recent weeks, and in Georgia, there has been an increase in RSV-related hospitalizations. Most of the hospitalizations in Georgia have been among infants less than a year old.
“Historically, such regional increases have predicted the beginning of RSV season nationally, with increased RSV activity spreading north and west over the following 2-3 months,” the CDC said.
Most children have been infected with RSV by the time they are 2 years old. Historically, up to 80,000 children under 5 years old are hospitalized annually because of the virus, and between 100 and 300 die from complications each year.
Those figures could be drastically different this year because new preventive treatments are available.
The CDC recommends that all children under 8 months old receive the newly approved monoclonal antibody treatment nirsevimab (Beyfortus). Children up to 19 months old at high risk of severe complications from RSV are also eligible for the single-dose shot. In clinical trials, the treatment was 80% effective at preventing RSV infections from becoming so severe that children had to be hospitalized. The protection lasted about 5 months.
Older people are also at a heightened risk of severe illness from RSV, and two new vaccines are available this season. The vaccines are called Arexvy and Abrysvo, and the single-dose shots are approved for people ages 60 years and older. They are more than 80% effective at making severe lower respiratory complications less likely.
Last year’s RSV season started during the summer and peaked in October and November, which was earlier than usual. There’s no indication yet of when RSV season may peak this year. Last year and throughout the pandemic, RSV held its historical pattern of starting in Florida.
A version of this article appeared on WebMD.com.
3D-printed meds customize the exact dose for sick children
Convincing kids to take their medicine could become much easier. Researchers at Texas A&M University are developing a new method of pharmaceutical 3D printing with pediatric patients in mind.
They hope to print precisely dosed tablets in child-friendly shapes and flavors. While the effort is focused on two drugs for pediatric AIDS, the process could be used to print other medicines, including for adults.
Researchers from Britain, Australia, and the University of Texas at Austin are also in the early stages of 3D-printed medication projects. It’s a promising venture in the broader pursuit of “personalized medicine,” tailoring treatments to each patient’s unique needs.
Drug mass production fails to address pediatric patients, who often need different dosages and combinations of medicines as they grow. As a result, adult tablets are often crushed and dissolved in liquid – known as compounding – and given to children. But this can harm drug quality and make doses less precise.
“Suppose the child needs 3.4 milligrams and only a 10-milligram tablet is available. Once you manipulate the dosage from solid to liquid, how do you ensure that it has the same amount of drug in it?” said co-principal investigator Mansoor Khan, PhD, a professor of pharmaceutical sciences at Texas A&M.
Most pharmacies lack the equipment to test compounded drug quality, he said. And liquified drugs taste bad because the pill coating has been ground away.
“Flavor is a big issue,” said Olive Eckstein, MD, an assistant professor of pediatric hematology-oncology at Texas Children’s Hospital and Baylor College of Medicine, who is not involved in the research. “Hospitals will sometimes delay discharging pediatric patients because they can’t take their meds orally and have to get an IV formulation.”
Updating pharmaceutical 3D printing
The FDA approved a 3D-printed drug in 2015, but since then, progress has stalled, largely because the method relied on solvents to bind drug particles together. Over time, solvents can compromise shelf life, according to co-principal investigator Mathew Kuttolamadom, PhD, an associate professor of engineering at Texas A&M.
The Texas A&M team is using a different method, without solvents. First, they create a powder mixture of the drug, a biocompatible polymer (such as lactose), and a sheen, a pigment that colors the tablet and allows heat to be absorbed. Flavoring can also be added. Next, the mixture is heated in the printer chamber.
“The polymer should melt just enough. That gives the tablet structural strength. But it should not melt too much, whereby the drug can start dissolving into the polymer,” Dr. Kuttolamadom said.
The tablets are finished with precise applications of laser heat. Using computer-aided design software, the researchers can create tablets in almost any shape, such as “stars or teddy bears,” he said.
After much trial and error, the researchers have printed tablets that won’t break apart or become soggy.
Now they are testing how different laser scan speeds affect the structure of the tablet, which in turn affects the rate at which drugs dissolve. Slowing down the laser imparts more energy, strengthening the tablet structure and making drugs dissolve slower, for a longer release inside the body.
The researchers hope to develop machine learning models to test different laser speed combinations. Eventually, they could create tablets that combine drugs with different dissolve rates.
“The outside could be a rapid release, and the inside could be an extended release or a sustained release, or even a completely different drug,” Dr. Kuttolamadom said.
Older patients who take many daily medications could benefit from the technology. “Personalized tablets could be printed at your local pharmacy,” he said, “even before you leave your doctor’s office.”
A version of this article first appeared on WebMD.com.
Convincing kids to take their medicine could become much easier. Researchers at Texas A&M University are developing a new method of pharmaceutical 3D printing with pediatric patients in mind.
They hope to print precisely dosed tablets in child-friendly shapes and flavors. While the effort is focused on two drugs for pediatric AIDS, the process could be used to print other medicines, including for adults.
Researchers from Britain, Australia, and the University of Texas at Austin are also in the early stages of 3D-printed medication projects. It’s a promising venture in the broader pursuit of “personalized medicine,” tailoring treatments to each patient’s unique needs.
Drug mass production fails to address pediatric patients, who often need different dosages and combinations of medicines as they grow. As a result, adult tablets are often crushed and dissolved in liquid – known as compounding – and given to children. But this can harm drug quality and make doses less precise.
“Suppose the child needs 3.4 milligrams and only a 10-milligram tablet is available. Once you manipulate the dosage from solid to liquid, how do you ensure that it has the same amount of drug in it?” said co-principal investigator Mansoor Khan, PhD, a professor of pharmaceutical sciences at Texas A&M.
Most pharmacies lack the equipment to test compounded drug quality, he said. And liquified drugs taste bad because the pill coating has been ground away.
“Flavor is a big issue,” said Olive Eckstein, MD, an assistant professor of pediatric hematology-oncology at Texas Children’s Hospital and Baylor College of Medicine, who is not involved in the research. “Hospitals will sometimes delay discharging pediatric patients because they can’t take their meds orally and have to get an IV formulation.”
Updating pharmaceutical 3D printing
The FDA approved a 3D-printed drug in 2015, but since then, progress has stalled, largely because the method relied on solvents to bind drug particles together. Over time, solvents can compromise shelf life, according to co-principal investigator Mathew Kuttolamadom, PhD, an associate professor of engineering at Texas A&M.
The Texas A&M team is using a different method, without solvents. First, they create a powder mixture of the drug, a biocompatible polymer (such as lactose), and a sheen, a pigment that colors the tablet and allows heat to be absorbed. Flavoring can also be added. Next, the mixture is heated in the printer chamber.
“The polymer should melt just enough. That gives the tablet structural strength. But it should not melt too much, whereby the drug can start dissolving into the polymer,” Dr. Kuttolamadom said.
The tablets are finished with precise applications of laser heat. Using computer-aided design software, the researchers can create tablets in almost any shape, such as “stars or teddy bears,” he said.
After much trial and error, the researchers have printed tablets that won’t break apart or become soggy.
Now they are testing how different laser scan speeds affect the structure of the tablet, which in turn affects the rate at which drugs dissolve. Slowing down the laser imparts more energy, strengthening the tablet structure and making drugs dissolve slower, for a longer release inside the body.
The researchers hope to develop machine learning models to test different laser speed combinations. Eventually, they could create tablets that combine drugs with different dissolve rates.
“The outside could be a rapid release, and the inside could be an extended release or a sustained release, or even a completely different drug,” Dr. Kuttolamadom said.
Older patients who take many daily medications could benefit from the technology. “Personalized tablets could be printed at your local pharmacy,” he said, “even before you leave your doctor’s office.”
A version of this article first appeared on WebMD.com.
Convincing kids to take their medicine could become much easier. Researchers at Texas A&M University are developing a new method of pharmaceutical 3D printing with pediatric patients in mind.
They hope to print precisely dosed tablets in child-friendly shapes and flavors. While the effort is focused on two drugs for pediatric AIDS, the process could be used to print other medicines, including for adults.
Researchers from Britain, Australia, and the University of Texas at Austin are also in the early stages of 3D-printed medication projects. It’s a promising venture in the broader pursuit of “personalized medicine,” tailoring treatments to each patient’s unique needs.
Drug mass production fails to address pediatric patients, who often need different dosages and combinations of medicines as they grow. As a result, adult tablets are often crushed and dissolved in liquid – known as compounding – and given to children. But this can harm drug quality and make doses less precise.
“Suppose the child needs 3.4 milligrams and only a 10-milligram tablet is available. Once you manipulate the dosage from solid to liquid, how do you ensure that it has the same amount of drug in it?” said co-principal investigator Mansoor Khan, PhD, a professor of pharmaceutical sciences at Texas A&M.
Most pharmacies lack the equipment to test compounded drug quality, he said. And liquified drugs taste bad because the pill coating has been ground away.
“Flavor is a big issue,” said Olive Eckstein, MD, an assistant professor of pediatric hematology-oncology at Texas Children’s Hospital and Baylor College of Medicine, who is not involved in the research. “Hospitals will sometimes delay discharging pediatric patients because they can’t take their meds orally and have to get an IV formulation.”
Updating pharmaceutical 3D printing
The FDA approved a 3D-printed drug in 2015, but since then, progress has stalled, largely because the method relied on solvents to bind drug particles together. Over time, solvents can compromise shelf life, according to co-principal investigator Mathew Kuttolamadom, PhD, an associate professor of engineering at Texas A&M.
The Texas A&M team is using a different method, without solvents. First, they create a powder mixture of the drug, a biocompatible polymer (such as lactose), and a sheen, a pigment that colors the tablet and allows heat to be absorbed. Flavoring can also be added. Next, the mixture is heated in the printer chamber.
“The polymer should melt just enough. That gives the tablet structural strength. But it should not melt too much, whereby the drug can start dissolving into the polymer,” Dr. Kuttolamadom said.
The tablets are finished with precise applications of laser heat. Using computer-aided design software, the researchers can create tablets in almost any shape, such as “stars or teddy bears,” he said.
After much trial and error, the researchers have printed tablets that won’t break apart or become soggy.
Now they are testing how different laser scan speeds affect the structure of the tablet, which in turn affects the rate at which drugs dissolve. Slowing down the laser imparts more energy, strengthening the tablet structure and making drugs dissolve slower, for a longer release inside the body.
The researchers hope to develop machine learning models to test different laser speed combinations. Eventually, they could create tablets that combine drugs with different dissolve rates.
“The outside could be a rapid release, and the inside could be an extended release or a sustained release, or even a completely different drug,” Dr. Kuttolamadom said.
Older patients who take many daily medications could benefit from the technology. “Personalized tablets could be printed at your local pharmacy,” he said, “even before you leave your doctor’s office.”
A version of this article first appeared on WebMD.com.
ESC issues first comprehensive cardiomyopathy guidelines
The European Society of Cardiology has released new guidelines for cardiomyopathies, their first major comprehensive international guidelines to address diagnosis and treatment of the broad causes of heart muscle dysfunction.
The document was released in conjunction with the annual congress of the European Society of Cardiology and is also available online in the European Heart Journal.
“We have considered cardiomyopathies across the life course from pediatric to adult,” explained Elena Arbelo, MD, PhD, coordinator of the cardiac genetic diseases and sudden arrhythmic death unit, Hospital Clinic de Barcelona. Dr. Arbelo is first author and one of two chairpersons of the ESC task force that brought the guidelines forward.
Not an update, Dr. Arbelo said.
Guidelines organize cardiomyopathy phenotypes
Cardiomyopathy can present at any age. It can have multiple complex etiologies, including genetic predisposition, heart muscle injury caused by disease, or a mix of participating factors. The ESC task force employed several strategies in taking a comprehensive approach to the condition, said Juan Kaski, MD, PhD, professor of pediatric inherited cardiovascular medicine at the University College of London.
“From my point of view, the key innovations include a diagnostic workup that starts with a detailed phenotypic description, including the new phenotype of nondilated left ventricular cardiomyopathy, that then triggers a multiparametric, systematic evaluation,” said Dr. Kaski, cochair of the task force.
As explained in the introduction to the guideline and reiterated by both Dr. Arbelo and Dr. Kaski, the guidelines have been organized around the patient pathway, meaning that focus should be placed on recognizing the presenting phenotype as a critical first step in discerning the underlying etiology and its treatments.
“Central to this approach is not only the individual patient but also the family as a whole,” Dr. Arbelo said. “Clinical findings in relatives are essential for understanding what happens to the patient and vice versa.”
Genetic testing in children described
The new guidelines include specific recommendations about genetic testing of children. They also emphasize the value of cardiovascular magnetic resonance (CMR) imaging in the “diagnosis, screening, monitoring, and prognostication” for patients of all ages, according to Dr. Kaski.
“CMR is recommended at the initial evaluation for every patient with cardiomyopathy,” Dr. Arbelo said. It should be “considered” during follow-up and for many other applications, including the evaluation of “genotype-positive but phenotype-negative relatives.”
Etiologic prediction models have been incorporated into the guidelines, including genotyping for dilated cardiomyopathies and nondilated left ventricular cardiomyopathy, said both Dr. Arbelo and Dr. Kaski, interviewed separately. They both indicated that the task force did their best to make the guidelines user friendly.
Each of the recommendations in the guidelines is provided with an evidence-based classification. In order, these are class I (recommended), class IIa (should be considered), class IIb (may be considered), and class III (not recommended).
Many symptoms are cardiomyopathy related
Dr. Kaski and Dr. Arbelo both emphasized that the guidelines draw attention to the relationship of cardiomyopathy to common cardiovascular conditions, such as heart failure, arrhythmia, and chest pain. Dr. Kaski pointed out that these are the types of problems commonly encountered by general cardiologists and well as primary care physicians.
In 2014, the ESC published guidelines specific to HCM. The new broader guidelines do not overlook this subtype. According to Dr. Kaski, there have been several innovations in HCM since the previous guidelines, such as when to consider cardiac myosin inhibitors for symptomatic left ventricular outflow tract obstruction.
The ESC guidelines place an emphasis on a “coordinated, systematic, and individualized” care pathway based on a multidisciplinary approach, according to Dr. Arbelo. Although the composition of the interdisciplinary team depends on the individual case, the guidelines recognize a key role for general cardiologists in managing the majority of patients. Suggestions of when to refer challenging cases to expert centers are outlined.
32 key messages derived from guidelines
The guidelines include almost 90 pages of recommendations. The task force isolated 32 key messages from 13 sections ranging from descriptions of how the patient pathway is defined to what types of physical activity should be considered for different forms of cardiomyopathy. There is also a section devoted to important gaps in evidence and areas in which there is the most need for further studies.
The guidelines end with a comprehensive list of “what to do” and “what not to do” in the diagnosis and care of cardiomyopathy. These include most of the class I recommendations and summarize some important class III cautions.
“Most of the recommendations in the guideline are new,” the authors wrote in the introduction. Although they acknowledged that they did not attempt to provide detailed recommendations for every cardiomyopathy phenotype, they endeavored to cover general evaluation and management issues supported by relevant evidence.
Dr. Arbelo and Dr. Kaski disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The European Society of Cardiology has released new guidelines for cardiomyopathies, their first major comprehensive international guidelines to address diagnosis and treatment of the broad causes of heart muscle dysfunction.
The document was released in conjunction with the annual congress of the European Society of Cardiology and is also available online in the European Heart Journal.
“We have considered cardiomyopathies across the life course from pediatric to adult,” explained Elena Arbelo, MD, PhD, coordinator of the cardiac genetic diseases and sudden arrhythmic death unit, Hospital Clinic de Barcelona. Dr. Arbelo is first author and one of two chairpersons of the ESC task force that brought the guidelines forward.
Not an update, Dr. Arbelo said.
Guidelines organize cardiomyopathy phenotypes
Cardiomyopathy can present at any age. It can have multiple complex etiologies, including genetic predisposition, heart muscle injury caused by disease, or a mix of participating factors. The ESC task force employed several strategies in taking a comprehensive approach to the condition, said Juan Kaski, MD, PhD, professor of pediatric inherited cardiovascular medicine at the University College of London.
“From my point of view, the key innovations include a diagnostic workup that starts with a detailed phenotypic description, including the new phenotype of nondilated left ventricular cardiomyopathy, that then triggers a multiparametric, systematic evaluation,” said Dr. Kaski, cochair of the task force.
As explained in the introduction to the guideline and reiterated by both Dr. Arbelo and Dr. Kaski, the guidelines have been organized around the patient pathway, meaning that focus should be placed on recognizing the presenting phenotype as a critical first step in discerning the underlying etiology and its treatments.
“Central to this approach is not only the individual patient but also the family as a whole,” Dr. Arbelo said. “Clinical findings in relatives are essential for understanding what happens to the patient and vice versa.”
Genetic testing in children described
The new guidelines include specific recommendations about genetic testing of children. They also emphasize the value of cardiovascular magnetic resonance (CMR) imaging in the “diagnosis, screening, monitoring, and prognostication” for patients of all ages, according to Dr. Kaski.
“CMR is recommended at the initial evaluation for every patient with cardiomyopathy,” Dr. Arbelo said. It should be “considered” during follow-up and for many other applications, including the evaluation of “genotype-positive but phenotype-negative relatives.”
Etiologic prediction models have been incorporated into the guidelines, including genotyping for dilated cardiomyopathies and nondilated left ventricular cardiomyopathy, said both Dr. Arbelo and Dr. Kaski, interviewed separately. They both indicated that the task force did their best to make the guidelines user friendly.
Each of the recommendations in the guidelines is provided with an evidence-based classification. In order, these are class I (recommended), class IIa (should be considered), class IIb (may be considered), and class III (not recommended).
Many symptoms are cardiomyopathy related
Dr. Kaski and Dr. Arbelo both emphasized that the guidelines draw attention to the relationship of cardiomyopathy to common cardiovascular conditions, such as heart failure, arrhythmia, and chest pain. Dr. Kaski pointed out that these are the types of problems commonly encountered by general cardiologists and well as primary care physicians.
In 2014, the ESC published guidelines specific to HCM. The new broader guidelines do not overlook this subtype. According to Dr. Kaski, there have been several innovations in HCM since the previous guidelines, such as when to consider cardiac myosin inhibitors for symptomatic left ventricular outflow tract obstruction.
The ESC guidelines place an emphasis on a “coordinated, systematic, and individualized” care pathway based on a multidisciplinary approach, according to Dr. Arbelo. Although the composition of the interdisciplinary team depends on the individual case, the guidelines recognize a key role for general cardiologists in managing the majority of patients. Suggestions of when to refer challenging cases to expert centers are outlined.
32 key messages derived from guidelines
The guidelines include almost 90 pages of recommendations. The task force isolated 32 key messages from 13 sections ranging from descriptions of how the patient pathway is defined to what types of physical activity should be considered for different forms of cardiomyopathy. There is also a section devoted to important gaps in evidence and areas in which there is the most need for further studies.
The guidelines end with a comprehensive list of “what to do” and “what not to do” in the diagnosis and care of cardiomyopathy. These include most of the class I recommendations and summarize some important class III cautions.
“Most of the recommendations in the guideline are new,” the authors wrote in the introduction. Although they acknowledged that they did not attempt to provide detailed recommendations for every cardiomyopathy phenotype, they endeavored to cover general evaluation and management issues supported by relevant evidence.
Dr. Arbelo and Dr. Kaski disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The European Society of Cardiology has released new guidelines for cardiomyopathies, their first major comprehensive international guidelines to address diagnosis and treatment of the broad causes of heart muscle dysfunction.
The document was released in conjunction with the annual congress of the European Society of Cardiology and is also available online in the European Heart Journal.
“We have considered cardiomyopathies across the life course from pediatric to adult,” explained Elena Arbelo, MD, PhD, coordinator of the cardiac genetic diseases and sudden arrhythmic death unit, Hospital Clinic de Barcelona. Dr. Arbelo is first author and one of two chairpersons of the ESC task force that brought the guidelines forward.
Not an update, Dr. Arbelo said.
Guidelines organize cardiomyopathy phenotypes
Cardiomyopathy can present at any age. It can have multiple complex etiologies, including genetic predisposition, heart muscle injury caused by disease, or a mix of participating factors. The ESC task force employed several strategies in taking a comprehensive approach to the condition, said Juan Kaski, MD, PhD, professor of pediatric inherited cardiovascular medicine at the University College of London.
“From my point of view, the key innovations include a diagnostic workup that starts with a detailed phenotypic description, including the new phenotype of nondilated left ventricular cardiomyopathy, that then triggers a multiparametric, systematic evaluation,” said Dr. Kaski, cochair of the task force.
As explained in the introduction to the guideline and reiterated by both Dr. Arbelo and Dr. Kaski, the guidelines have been organized around the patient pathway, meaning that focus should be placed on recognizing the presenting phenotype as a critical first step in discerning the underlying etiology and its treatments.
“Central to this approach is not only the individual patient but also the family as a whole,” Dr. Arbelo said. “Clinical findings in relatives are essential for understanding what happens to the patient and vice versa.”
Genetic testing in children described
The new guidelines include specific recommendations about genetic testing of children. They also emphasize the value of cardiovascular magnetic resonance (CMR) imaging in the “diagnosis, screening, monitoring, and prognostication” for patients of all ages, according to Dr. Kaski.
“CMR is recommended at the initial evaluation for every patient with cardiomyopathy,” Dr. Arbelo said. It should be “considered” during follow-up and for many other applications, including the evaluation of “genotype-positive but phenotype-negative relatives.”
Etiologic prediction models have been incorporated into the guidelines, including genotyping for dilated cardiomyopathies and nondilated left ventricular cardiomyopathy, said both Dr. Arbelo and Dr. Kaski, interviewed separately. They both indicated that the task force did their best to make the guidelines user friendly.
Each of the recommendations in the guidelines is provided with an evidence-based classification. In order, these are class I (recommended), class IIa (should be considered), class IIb (may be considered), and class III (not recommended).
Many symptoms are cardiomyopathy related
Dr. Kaski and Dr. Arbelo both emphasized that the guidelines draw attention to the relationship of cardiomyopathy to common cardiovascular conditions, such as heart failure, arrhythmia, and chest pain. Dr. Kaski pointed out that these are the types of problems commonly encountered by general cardiologists and well as primary care physicians.
In 2014, the ESC published guidelines specific to HCM. The new broader guidelines do not overlook this subtype. According to Dr. Kaski, there have been several innovations in HCM since the previous guidelines, such as when to consider cardiac myosin inhibitors for symptomatic left ventricular outflow tract obstruction.
The ESC guidelines place an emphasis on a “coordinated, systematic, and individualized” care pathway based on a multidisciplinary approach, according to Dr. Arbelo. Although the composition of the interdisciplinary team depends on the individual case, the guidelines recognize a key role for general cardiologists in managing the majority of patients. Suggestions of when to refer challenging cases to expert centers are outlined.
32 key messages derived from guidelines
The guidelines include almost 90 pages of recommendations. The task force isolated 32 key messages from 13 sections ranging from descriptions of how the patient pathway is defined to what types of physical activity should be considered for different forms of cardiomyopathy. There is also a section devoted to important gaps in evidence and areas in which there is the most need for further studies.
The guidelines end with a comprehensive list of “what to do” and “what not to do” in the diagnosis and care of cardiomyopathy. These include most of the class I recommendations and summarize some important class III cautions.
“Most of the recommendations in the guideline are new,” the authors wrote in the introduction. Although they acknowledged that they did not attempt to provide detailed recommendations for every cardiomyopathy phenotype, they endeavored to cover general evaluation and management issues supported by relevant evidence.
Dr. Arbelo and Dr. Kaski disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE ESC CONGRESS 2023
‘Decapitated’ boy saved by surgery team
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: I am joined today by Dr. Ohad Einav. He’s a staff surgeon in orthopedics at Hadassah Medical Center in Jerusalem. He’s with me to talk about an absolutely incredible surgical case, something that is terrifying to most non–orthopedic surgeons and I imagine is fairly scary for spine surgeons like him as well. But what we don’t have is information about how this works from a medical perspective. So, first of all, Dr. Einav, thank you for taking time to speak with me today.
Ohad Einav, MD: Thank you for having me.
Dr. Wilson: Can you tell us about Suleiman Hassan and what happened to him before he came into your care?
Dr. Einav: Hassan is a 12-year-old child who was riding his bicycle on the West Bank, about 40 minutes from here. Unfortunately, he was involved in a motor vehicle accident and he suffered injuries to his abdomen and cervical spine. He was transported to our service by helicopter from the scene of the accident.
Dr. Wilson: “Injury to the cervical spine” might be something of an understatement. He had what’s called atlanto-occipital dislocation, colloquially often referred to as internal decapitation. Can you tell us what that means? It sounds terrifying.
Dr. Einav: It’s an injury to the ligaments between the occiput and the upper cervical spine, with or without bony fracture. The atlanto-occipital joint is formed by the superior articular facet of the atlas and the occipital condyle, stabilized by an articular capsule between the head and neck, and is supported by various ligaments around it that stabilize the joint and allow joint movements, including flexion, extension, and some rotation in the lower levels.
Dr. Wilson: This joint has several degrees of freedom, which means it needs a lot of support. With this type of injury, where essentially you have severing of the ligaments, is it usually survivable? How dangerous is this?
Dr. Einav: The mortality rate is 50%-60%, depending on the primary impact, the injury, transportation later on, and then the surgery and surgical management.
Dr. Wilson: Tell us a bit about this patient’s status when he came to your medical center. I assume he was in bad shape.
Dr. Einav: Hassan arrived at our medical center with a Glasgow Coma Scale score of 15. He was fully conscious. He was hemodynamically stable except for a bad laceration on his abdomen. He had a Philadelphia collar around his neck. He was transported by chopper because the paramedics suspected that he had a cervical spine injury and decided to bring him to a Level 1 trauma center.
He was monitored and we treated him according to the ATLS [advanced trauma life support] protocol. He didn’t have any gross sensory deficits, but he was a little confused about the whole situation and the accident. Therefore, we could do a general examination but we couldn’t rely on that regarding any sensory deficit that he may or may not have. We decided as a team that it would be better to slow down and control the situation. We decided not to operate on him immediately. We basically stabilized him and made sure that he didn’t have any traumatic internal organ damage. Later on we took him to the OR and performed surgery.
Dr. Wilson: It’s amazing that he had intact motor function, considering the extent of his injury. The spinal cord was spared somewhat during the injury. There must have been a moment when you realized that this kid, who was conscious and could move all four extremities, had a very severe neck injury. Was that due to a CT scan or physical exam? And what was your feeling when you saw that he had atlanto-occipital dislocation?
Dr. Einav: As a surgeon, you have a gut feeling in regard to the general examination of the patient. But I never rely on gut feelings. On the CT, I understood exactly what he had, what we needed to do, and the time frame.
Dr. Wilson: You’ve done these types of surgeries before, right? Obviously, no one has done a lot of them because this isn’t very common. But you knew what to do. Did you have a plan? Where does your experience come into play in a situation like this?
Dr. Einav: I graduated from the spine program of Toronto University, where I did a fellowship in trauma of the spine and complex spine surgery. I had very good teachers, and during my fellowship I treated a few cases in older patients that were similar but not the same. Therefore, I knew exactly what needed to be done.
Dr. Wilson: For those of us who aren’t surgeons, take us into the OR with you. This is obviously an incredibly delicate procedure. You are high up in the spinal cord at the base of the brain. The slightest mistake could have devastating consequences. What are the key elements of this procedure? What can go wrong here? What is the number-one thing you have to look out for when you’re trying to fix an internal decapitation?
Dr. Einav: The key element in surgeries of the cervical spine – trauma and complex spine surgery – is planning. I never go to the OR without knowing what I’m going to do. I have a few plans – plan A, plan B, plan C – in case something fails. So, I definitely know what the next step will be. I always think about the surgery a few hours before, if I have time to prepare.
The second thing that is very important is teamwork. The team needs to be coordinated. Everybody needs to know what their job is. With these types of injuries, it’s not the time for rookies. If you are new, please stand back and let the more experienced people do that job. I’m talking about surgeons, nurses, anesthesiologists – everyone.
Another important thing in planning is choosing the right hardware. For example, in this case we had a problem because most of the hardware is designed for adults, and we had to improvise because there isn’t a lot of hardware on the market for the pediatric population. The adult plates and screws are too big, so we had to improvise.
Dr. Wilson: Tell us more about that. How do you improvise spinal hardware for a 12-year-old?
Dr. Einav: In this case, I chose to use hardware from one of the companies that works with us.
You can see in this model the area of the injury, and the area that we worked on. To perform the surgery, I had to use some plates and rods from a different company. This company’s (NuVasive) hardware has a small attachment to the skull, which was helpful for affixing the skull to the cervical spine, instead of using a big plate that would sit at the base of the skull and would not be very good for him. Most of the hardware is made for adults and not for kids.
Dr. Wilson: Will that hardware preserve the motor function of his neck? Will he be able to turn his head and extend and flex it?
Dr. Einav: The injury leads to instability and destruction of both articulations between the head and neck. Therefore, those articulations won’t be able to function the same way in the future. There is a decrease of something like 50% of the flexion and extension of Hassan’s cervical spine. Therefore, I decided that in this case there would be no chance of saving Hassan’s motor function unless we performed a fusion between the head and the neck, and therefore I decided that this would be the best procedure with the best survival rate. So, in the future, he will have some diminished flexion, extension, and rotation of his head.
Dr. Wilson: How long did his surgery take?
Dr. Einav: To be honest, I don’t remember. But I can tell you that it took us time. It was very challenging to coordinate with everyone. The most problematic part of the surgery to perform is what we call “flip-over.”
The anesthesiologist intubated the patient when he was supine, and later on, we flipped him prone to operate on the spine. This maneuver can actually lead to injury by itself, and injury at this level is fatal. So, we took our time and got Hassan into the OR. The anesthesiologist did a great job with the GlideScope – inserting the endotracheal tube. Later on, we neuromonitored him. Basically, we connected Hassan’s peripheral nerves to a computer and monitored his motor function. Gently we flipped him over, and after that we saw a little change in his motor function, so we had to modify his position so we could preserve his motor function. We then started the procedure, which took a few hours. I don’t know exactly how many.
Dr. Wilson: That just speaks to how delicate this is for everything from the intubation, where typically you’re manipulating the head, to the repositioning. Clearly this requires a lot of teamwork.
What happened after the operation? How is he doing?
Dr. Einav: After the operation, Hassan had a great recovery. He’s doing well. He doesn’t have any motor or sensory deficits. He’s able to ambulate without any aid. He had no signs of infection, which can happen after a car accident, neither from his abdominal wound nor from the occipital cervical surgery. He feels well. We saw him in the clinic. We removed his collar. We monitored him at the clinic. He looked amazing.
Dr. Wilson: That’s incredible. Are there long-term risks for him that you need to be looking out for?
Dr. Einav: Yes, and that’s the reason that we are monitoring him post surgery. While he was in the hospital, we monitored his motor and sensory functions, as well as his wound healing. Later on, in the clinic, for a few weeks after surgery we monitored for any failure of the hardware and bone graft. We check for healing of the bone graft and bone substitutes we put in to heal those bones.
Dr. Wilson: He will grow, right? He’s only 12, so he still has some years of growth in him. Is he going to need more surgery or any kind of hardware upgrade?
Dr. Einav: I hope not. In my surgeries, I never rely on the hardware for long durations. If I decide to do, for example, fusion, I rely on the hardware for a certain amount of time. And then I plan that the biology will do the work. If I plan for fusion, I put bone grafts in the preferred area for a fusion. Then if the hardware fails, I wouldn’t need to take out the hardware, and there would be no change in the condition of the patient.
Dr. Wilson: What an incredible story. It’s clear that you and your team kept your cool despite a very high-acuity situation with a ton of risk. What a tremendous outcome that this boy is not only alive but fully functional. So, congratulations to you and your team. That was very strong work.
Dr. Einav: Thank you very much. I would like to thank our team. We have to remember that the surgeon is not standing alone in the war. Hassan’s story is a success story of a very big group of people from various backgrounds and religions. They work day and night to help people and save lives. To the paramedics, the physiologists, the traumatologists, the pediatricians, the nurses, the physiotherapists, and obviously the surgeons, a big thank you. His story is our success story.
Dr. Wilson: It’s inspiring to see so many people come together to do what we all are here for, which is to fight against suffering, disease, and death. Thank you for keeping up that fight. And thank you for joining me here.
Dr. Einav: Thank you very much.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: I am joined today by Dr. Ohad Einav. He’s a staff surgeon in orthopedics at Hadassah Medical Center in Jerusalem. He’s with me to talk about an absolutely incredible surgical case, something that is terrifying to most non–orthopedic surgeons and I imagine is fairly scary for spine surgeons like him as well. But what we don’t have is information about how this works from a medical perspective. So, first of all, Dr. Einav, thank you for taking time to speak with me today.
Ohad Einav, MD: Thank you for having me.
Dr. Wilson: Can you tell us about Suleiman Hassan and what happened to him before he came into your care?
Dr. Einav: Hassan is a 12-year-old child who was riding his bicycle on the West Bank, about 40 minutes from here. Unfortunately, he was involved in a motor vehicle accident and he suffered injuries to his abdomen and cervical spine. He was transported to our service by helicopter from the scene of the accident.
Dr. Wilson: “Injury to the cervical spine” might be something of an understatement. He had what’s called atlanto-occipital dislocation, colloquially often referred to as internal decapitation. Can you tell us what that means? It sounds terrifying.
Dr. Einav: It’s an injury to the ligaments between the occiput and the upper cervical spine, with or without bony fracture. The atlanto-occipital joint is formed by the superior articular facet of the atlas and the occipital condyle, stabilized by an articular capsule between the head and neck, and is supported by various ligaments around it that stabilize the joint and allow joint movements, including flexion, extension, and some rotation in the lower levels.
Dr. Wilson: This joint has several degrees of freedom, which means it needs a lot of support. With this type of injury, where essentially you have severing of the ligaments, is it usually survivable? How dangerous is this?
Dr. Einav: The mortality rate is 50%-60%, depending on the primary impact, the injury, transportation later on, and then the surgery and surgical management.
Dr. Wilson: Tell us a bit about this patient’s status when he came to your medical center. I assume he was in bad shape.
Dr. Einav: Hassan arrived at our medical center with a Glasgow Coma Scale score of 15. He was fully conscious. He was hemodynamically stable except for a bad laceration on his abdomen. He had a Philadelphia collar around his neck. He was transported by chopper because the paramedics suspected that he had a cervical spine injury and decided to bring him to a Level 1 trauma center.
He was monitored and we treated him according to the ATLS [advanced trauma life support] protocol. He didn’t have any gross sensory deficits, but he was a little confused about the whole situation and the accident. Therefore, we could do a general examination but we couldn’t rely on that regarding any sensory deficit that he may or may not have. We decided as a team that it would be better to slow down and control the situation. We decided not to operate on him immediately. We basically stabilized him and made sure that he didn’t have any traumatic internal organ damage. Later on we took him to the OR and performed surgery.
Dr. Wilson: It’s amazing that he had intact motor function, considering the extent of his injury. The spinal cord was spared somewhat during the injury. There must have been a moment when you realized that this kid, who was conscious and could move all four extremities, had a very severe neck injury. Was that due to a CT scan or physical exam? And what was your feeling when you saw that he had atlanto-occipital dislocation?
Dr. Einav: As a surgeon, you have a gut feeling in regard to the general examination of the patient. But I never rely on gut feelings. On the CT, I understood exactly what he had, what we needed to do, and the time frame.
Dr. Wilson: You’ve done these types of surgeries before, right? Obviously, no one has done a lot of them because this isn’t very common. But you knew what to do. Did you have a plan? Where does your experience come into play in a situation like this?
Dr. Einav: I graduated from the spine program of Toronto University, where I did a fellowship in trauma of the spine and complex spine surgery. I had very good teachers, and during my fellowship I treated a few cases in older patients that were similar but not the same. Therefore, I knew exactly what needed to be done.
Dr. Wilson: For those of us who aren’t surgeons, take us into the OR with you. This is obviously an incredibly delicate procedure. You are high up in the spinal cord at the base of the brain. The slightest mistake could have devastating consequences. What are the key elements of this procedure? What can go wrong here? What is the number-one thing you have to look out for when you’re trying to fix an internal decapitation?
Dr. Einav: The key element in surgeries of the cervical spine – trauma and complex spine surgery – is planning. I never go to the OR without knowing what I’m going to do. I have a few plans – plan A, plan B, plan C – in case something fails. So, I definitely know what the next step will be. I always think about the surgery a few hours before, if I have time to prepare.
The second thing that is very important is teamwork. The team needs to be coordinated. Everybody needs to know what their job is. With these types of injuries, it’s not the time for rookies. If you are new, please stand back and let the more experienced people do that job. I’m talking about surgeons, nurses, anesthesiologists – everyone.
Another important thing in planning is choosing the right hardware. For example, in this case we had a problem because most of the hardware is designed for adults, and we had to improvise because there isn’t a lot of hardware on the market for the pediatric population. The adult plates and screws are too big, so we had to improvise.
Dr. Wilson: Tell us more about that. How do you improvise spinal hardware for a 12-year-old?
Dr. Einav: In this case, I chose to use hardware from one of the companies that works with us.
You can see in this model the area of the injury, and the area that we worked on. To perform the surgery, I had to use some plates and rods from a different company. This company’s (NuVasive) hardware has a small attachment to the skull, which was helpful for affixing the skull to the cervical spine, instead of using a big plate that would sit at the base of the skull and would not be very good for him. Most of the hardware is made for adults and not for kids.
Dr. Wilson: Will that hardware preserve the motor function of his neck? Will he be able to turn his head and extend and flex it?
Dr. Einav: The injury leads to instability and destruction of both articulations between the head and neck. Therefore, those articulations won’t be able to function the same way in the future. There is a decrease of something like 50% of the flexion and extension of Hassan’s cervical spine. Therefore, I decided that in this case there would be no chance of saving Hassan’s motor function unless we performed a fusion between the head and the neck, and therefore I decided that this would be the best procedure with the best survival rate. So, in the future, he will have some diminished flexion, extension, and rotation of his head.
Dr. Wilson: How long did his surgery take?
Dr. Einav: To be honest, I don’t remember. But I can tell you that it took us time. It was very challenging to coordinate with everyone. The most problematic part of the surgery to perform is what we call “flip-over.”
The anesthesiologist intubated the patient when he was supine, and later on, we flipped him prone to operate on the spine. This maneuver can actually lead to injury by itself, and injury at this level is fatal. So, we took our time and got Hassan into the OR. The anesthesiologist did a great job with the GlideScope – inserting the endotracheal tube. Later on, we neuromonitored him. Basically, we connected Hassan’s peripheral nerves to a computer and monitored his motor function. Gently we flipped him over, and after that we saw a little change in his motor function, so we had to modify his position so we could preserve his motor function. We then started the procedure, which took a few hours. I don’t know exactly how many.
Dr. Wilson: That just speaks to how delicate this is for everything from the intubation, where typically you’re manipulating the head, to the repositioning. Clearly this requires a lot of teamwork.
What happened after the operation? How is he doing?
Dr. Einav: After the operation, Hassan had a great recovery. He’s doing well. He doesn’t have any motor or sensory deficits. He’s able to ambulate without any aid. He had no signs of infection, which can happen after a car accident, neither from his abdominal wound nor from the occipital cervical surgery. He feels well. We saw him in the clinic. We removed his collar. We monitored him at the clinic. He looked amazing.
Dr. Wilson: That’s incredible. Are there long-term risks for him that you need to be looking out for?
Dr. Einav: Yes, and that’s the reason that we are monitoring him post surgery. While he was in the hospital, we monitored his motor and sensory functions, as well as his wound healing. Later on, in the clinic, for a few weeks after surgery we monitored for any failure of the hardware and bone graft. We check for healing of the bone graft and bone substitutes we put in to heal those bones.
Dr. Wilson: He will grow, right? He’s only 12, so he still has some years of growth in him. Is he going to need more surgery or any kind of hardware upgrade?
Dr. Einav: I hope not. In my surgeries, I never rely on the hardware for long durations. If I decide to do, for example, fusion, I rely on the hardware for a certain amount of time. And then I plan that the biology will do the work. If I plan for fusion, I put bone grafts in the preferred area for a fusion. Then if the hardware fails, I wouldn’t need to take out the hardware, and there would be no change in the condition of the patient.
Dr. Wilson: What an incredible story. It’s clear that you and your team kept your cool despite a very high-acuity situation with a ton of risk. What a tremendous outcome that this boy is not only alive but fully functional. So, congratulations to you and your team. That was very strong work.
Dr. Einav: Thank you very much. I would like to thank our team. We have to remember that the surgeon is not standing alone in the war. Hassan’s story is a success story of a very big group of people from various backgrounds and religions. They work day and night to help people and save lives. To the paramedics, the physiologists, the traumatologists, the pediatricians, the nurses, the physiotherapists, and obviously the surgeons, a big thank you. His story is our success story.
Dr. Wilson: It’s inspiring to see so many people come together to do what we all are here for, which is to fight against suffering, disease, and death. Thank you for keeping up that fight. And thank you for joining me here.
Dr. Einav: Thank you very much.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: I am joined today by Dr. Ohad Einav. He’s a staff surgeon in orthopedics at Hadassah Medical Center in Jerusalem. He’s with me to talk about an absolutely incredible surgical case, something that is terrifying to most non–orthopedic surgeons and I imagine is fairly scary for spine surgeons like him as well. But what we don’t have is information about how this works from a medical perspective. So, first of all, Dr. Einav, thank you for taking time to speak with me today.
Ohad Einav, MD: Thank you for having me.
Dr. Wilson: Can you tell us about Suleiman Hassan and what happened to him before he came into your care?
Dr. Einav: Hassan is a 12-year-old child who was riding his bicycle on the West Bank, about 40 minutes from here. Unfortunately, he was involved in a motor vehicle accident and he suffered injuries to his abdomen and cervical spine. He was transported to our service by helicopter from the scene of the accident.
Dr. Wilson: “Injury to the cervical spine” might be something of an understatement. He had what’s called atlanto-occipital dislocation, colloquially often referred to as internal decapitation. Can you tell us what that means? It sounds terrifying.
Dr. Einav: It’s an injury to the ligaments between the occiput and the upper cervical spine, with or without bony fracture. The atlanto-occipital joint is formed by the superior articular facet of the atlas and the occipital condyle, stabilized by an articular capsule between the head and neck, and is supported by various ligaments around it that stabilize the joint and allow joint movements, including flexion, extension, and some rotation in the lower levels.
Dr. Wilson: This joint has several degrees of freedom, which means it needs a lot of support. With this type of injury, where essentially you have severing of the ligaments, is it usually survivable? How dangerous is this?
Dr. Einav: The mortality rate is 50%-60%, depending on the primary impact, the injury, transportation later on, and then the surgery and surgical management.
Dr. Wilson: Tell us a bit about this patient’s status when he came to your medical center. I assume he was in bad shape.
Dr. Einav: Hassan arrived at our medical center with a Glasgow Coma Scale score of 15. He was fully conscious. He was hemodynamically stable except for a bad laceration on his abdomen. He had a Philadelphia collar around his neck. He was transported by chopper because the paramedics suspected that he had a cervical spine injury and decided to bring him to a Level 1 trauma center.
He was monitored and we treated him according to the ATLS [advanced trauma life support] protocol. He didn’t have any gross sensory deficits, but he was a little confused about the whole situation and the accident. Therefore, we could do a general examination but we couldn’t rely on that regarding any sensory deficit that he may or may not have. We decided as a team that it would be better to slow down and control the situation. We decided not to operate on him immediately. We basically stabilized him and made sure that he didn’t have any traumatic internal organ damage. Later on we took him to the OR and performed surgery.
Dr. Wilson: It’s amazing that he had intact motor function, considering the extent of his injury. The spinal cord was spared somewhat during the injury. There must have been a moment when you realized that this kid, who was conscious and could move all four extremities, had a very severe neck injury. Was that due to a CT scan or physical exam? And what was your feeling when you saw that he had atlanto-occipital dislocation?
Dr. Einav: As a surgeon, you have a gut feeling in regard to the general examination of the patient. But I never rely on gut feelings. On the CT, I understood exactly what he had, what we needed to do, and the time frame.
Dr. Wilson: You’ve done these types of surgeries before, right? Obviously, no one has done a lot of them because this isn’t very common. But you knew what to do. Did you have a plan? Where does your experience come into play in a situation like this?
Dr. Einav: I graduated from the spine program of Toronto University, where I did a fellowship in trauma of the spine and complex spine surgery. I had very good teachers, and during my fellowship I treated a few cases in older patients that were similar but not the same. Therefore, I knew exactly what needed to be done.
Dr. Wilson: For those of us who aren’t surgeons, take us into the OR with you. This is obviously an incredibly delicate procedure. You are high up in the spinal cord at the base of the brain. The slightest mistake could have devastating consequences. What are the key elements of this procedure? What can go wrong here? What is the number-one thing you have to look out for when you’re trying to fix an internal decapitation?
Dr. Einav: The key element in surgeries of the cervical spine – trauma and complex spine surgery – is planning. I never go to the OR without knowing what I’m going to do. I have a few plans – plan A, plan B, plan C – in case something fails. So, I definitely know what the next step will be. I always think about the surgery a few hours before, if I have time to prepare.
The second thing that is very important is teamwork. The team needs to be coordinated. Everybody needs to know what their job is. With these types of injuries, it’s not the time for rookies. If you are new, please stand back and let the more experienced people do that job. I’m talking about surgeons, nurses, anesthesiologists – everyone.
Another important thing in planning is choosing the right hardware. For example, in this case we had a problem because most of the hardware is designed for adults, and we had to improvise because there isn’t a lot of hardware on the market for the pediatric population. The adult plates and screws are too big, so we had to improvise.
Dr. Wilson: Tell us more about that. How do you improvise spinal hardware for a 12-year-old?
Dr. Einav: In this case, I chose to use hardware from one of the companies that works with us.
You can see in this model the area of the injury, and the area that we worked on. To perform the surgery, I had to use some plates and rods from a different company. This company’s (NuVasive) hardware has a small attachment to the skull, which was helpful for affixing the skull to the cervical spine, instead of using a big plate that would sit at the base of the skull and would not be very good for him. Most of the hardware is made for adults and not for kids.
Dr. Wilson: Will that hardware preserve the motor function of his neck? Will he be able to turn his head and extend and flex it?
Dr. Einav: The injury leads to instability and destruction of both articulations between the head and neck. Therefore, those articulations won’t be able to function the same way in the future. There is a decrease of something like 50% of the flexion and extension of Hassan’s cervical spine. Therefore, I decided that in this case there would be no chance of saving Hassan’s motor function unless we performed a fusion between the head and the neck, and therefore I decided that this would be the best procedure with the best survival rate. So, in the future, he will have some diminished flexion, extension, and rotation of his head.
Dr. Wilson: How long did his surgery take?
Dr. Einav: To be honest, I don’t remember. But I can tell you that it took us time. It was very challenging to coordinate with everyone. The most problematic part of the surgery to perform is what we call “flip-over.”
The anesthesiologist intubated the patient when he was supine, and later on, we flipped him prone to operate on the spine. This maneuver can actually lead to injury by itself, and injury at this level is fatal. So, we took our time and got Hassan into the OR. The anesthesiologist did a great job with the GlideScope – inserting the endotracheal tube. Later on, we neuromonitored him. Basically, we connected Hassan’s peripheral nerves to a computer and monitored his motor function. Gently we flipped him over, and after that we saw a little change in his motor function, so we had to modify his position so we could preserve his motor function. We then started the procedure, which took a few hours. I don’t know exactly how many.
Dr. Wilson: That just speaks to how delicate this is for everything from the intubation, where typically you’re manipulating the head, to the repositioning. Clearly this requires a lot of teamwork.
What happened after the operation? How is he doing?
Dr. Einav: After the operation, Hassan had a great recovery. He’s doing well. He doesn’t have any motor or sensory deficits. He’s able to ambulate without any aid. He had no signs of infection, which can happen after a car accident, neither from his abdominal wound nor from the occipital cervical surgery. He feels well. We saw him in the clinic. We removed his collar. We monitored him at the clinic. He looked amazing.
Dr. Wilson: That’s incredible. Are there long-term risks for him that you need to be looking out for?
Dr. Einav: Yes, and that’s the reason that we are monitoring him post surgery. While he was in the hospital, we monitored his motor and sensory functions, as well as his wound healing. Later on, in the clinic, for a few weeks after surgery we monitored for any failure of the hardware and bone graft. We check for healing of the bone graft and bone substitutes we put in to heal those bones.
Dr. Wilson: He will grow, right? He’s only 12, so he still has some years of growth in him. Is he going to need more surgery or any kind of hardware upgrade?
Dr. Einav: I hope not. In my surgeries, I never rely on the hardware for long durations. If I decide to do, for example, fusion, I rely on the hardware for a certain amount of time. And then I plan that the biology will do the work. If I plan for fusion, I put bone grafts in the preferred area for a fusion. Then if the hardware fails, I wouldn’t need to take out the hardware, and there would be no change in the condition of the patient.
Dr. Wilson: What an incredible story. It’s clear that you and your team kept your cool despite a very high-acuity situation with a ton of risk. What a tremendous outcome that this boy is not only alive but fully functional. So, congratulations to you and your team. That was very strong work.
Dr. Einav: Thank you very much. I would like to thank our team. We have to remember that the surgeon is not standing alone in the war. Hassan’s story is a success story of a very big group of people from various backgrounds and religions. They work day and night to help people and save lives. To the paramedics, the physiologists, the traumatologists, the pediatricians, the nurses, the physiotherapists, and obviously the surgeons, a big thank you. His story is our success story.
Dr. Wilson: It’s inspiring to see so many people come together to do what we all are here for, which is to fight against suffering, disease, and death. Thank you for keeping up that fight. And thank you for joining me here.
Dr. Einav: Thank you very much.
A version of this article first appeared on Medscape.com.