User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Physician group staffing down, expenses up, new reports show
Physician groups saw staff-to-physician ratios decline even as their workforce expenses rose between 2019 and 2021, according to recent reports from the American Medical Group Association (AMGA) and the Medical Group Management Association (MGMA).
As patients started to return to doctors’ offices as the pandemic eased in 2021, physician groups found it increasingly difficult to recruit and retain lower-level clinicians, including medical assistants and LPNs, officials from both associations told this news organization. Many clinics had to raise their pay scales to be competitive with employers in other fields, and some had to hire higher-priced RNs to keep their practices running.
The AMGA report was based largely on data from groups of over 500 physicians, mostly affiliated with health systems. According to a news release accompanying the report, the ratio between full-time equivalent (FTE) clinic staff and health care professionals in direct patient care dropped by 11.3% between 2019 and 2021. The ratio of medical assistants (MAs) to clinicians declined by a greater percentage.
In the MGMA report, which represented about 4,000 practices ranging from very small (two doctors) to very large groups, total support staff per FTE primary-care physician dropped by 18% from 2019 to 2021 in independent groups and by 13% in hospital-affiliated groups. The ratios decreased by smaller amounts in surgical practices.
In contrast, nonsurgical specialty groups under both types of ownership saw their staffing ratios rise slightly.
Although it’s unclear why medical specialties increased their staff while other types of specialties lost employees, Ron Holder, MHA, chief operating officer of MGMA, said that some specialists may have opened more ancillary facilities and hired new employees to recoup revenue lost during the pandemic.
Expenses rise sharply
The AMGA report found that staffing expenses for the surveyed groups increased by 15% between 2019 and 2021.
“We saw a decrease in staff and an increase in expenses during that time period, and there are a few reasons for that,” Rose Wagner, RN, chief operating officer of AMGA, said. “Groups increased salaries to maintain staff. We also saw lower-paid staff find other jobs outside of health care. For example, medical assistants and receptionists could find jobs outside of health care that paid more. [Open positions] got back-filled with other higher paid staff, such as RNs, doing lower skilled jobs.”
Mr. Holder added that rising wages in other sectors made leaving physician groups more attractive for employees.
“Three years ago, there weren’t many positions in a medical practice where you were competing with Chick-fil-A or Taco Bell,” he said. In Denver, where Mr. Holder is based, “every restaurant in town is now advertising $17-$19 [hourly] starting pay just to do fast food. That causes practices to either lose employees or pay more for the employees they have. So that raises per-employee expense significantly,” he said.
Mr. Holder noted that inflation also has driven up wages as employees demand higher pay to keep up with the cost of living.
Unusual exodus of employees
Fred Horton, MHA, president of AMGA Consulting, said he has never seen so many people leaving health care for other occupations.
Some exits resulted from practices laying people off early in the pandemic, but most staff members who left practices were seeking higher pay, he said. In addition, Ms. Wagner noted, some staff members didn’t want to be exposed to COVID at work.
“There was an exodus from health care that was different from what we’d experienced in the past,” Mr. Horton added. “It’s still extremely challenging to get up to the staffing levels that are appropriate.”
Mr. Holder, however, said that the situation is slowly improving. “Health care is fairly recession-proof, because people need it. So when you see companies in other industries closing shop or reducing their head count, that actually helps health care recruiting in some jobs. And people are coming back to the workplace who previously were worried about COVID or didn’t want to get the vaccine.”
Paying more for nurses
In 2021, groups adopted a variety of tactics to adapt to the pandemic and respond to patient demand, the AMGA survey shows. Forty percent of system-affiliated groups and 18% of independent practices changed registered nurses’ responsibilities, in many cases having them do the work of medical assistants who were in short supply.
Some practices hired RNs, who have historically been utilized less by primary care than by surgical specialties, Mr. Holder noted. Other clinics paid temp agencies to supply nurses at a steep cost.
“When you’re short staffed, you end up paying more overtime, you end up paying temporary agencies at higher dollars, and you hire higher skilled people to do lower-skilled work,” Ms. Wagner said.
Meanwhile, many physician groups tried to cope with the physician shortage by bringing on more advanced practice clinicians (APCs), including nurse practitioners (NPs) and physician assistants (PAs). Seventy percent of the AMGA groups used this strategy, the report revealed.
“The use of APCs has been steadily increasing as groups try to adopt a lower-cost care model in the midst of a nationwide physician shortage,” Ms. Wagner said in the press release.
Changes in patient care
About half of the groups in the AMGA survey said they changed their staff structure to allow APCs to carry their own patient panels. Although most of these clinicians were probably under physician supervision, nearly half of the states now allow NPs to practice autonomously.
Mr. Horton cautioned that APCs can’t fully substitute for physicians and require the same support staff that doctors do if they have their own panels. In primary care groups, Mr. Holder noted, the average salary of an APC “is continuing to rise, and there isn’t a huge difference between what they and doctors make.”
Nevertheless, he added, “there are more NPs and PAs being added to the marketplace all the time, whereas [physician] residency programs aren’t really growing. There are caps on the number of residency positions, and some physicians are retiring. So the clock is ticking to the point where someday doctors will be grossly outnumbered by NPs.”
A version of this article first appeared on Medscape.com.
Physician groups saw staff-to-physician ratios decline even as their workforce expenses rose between 2019 and 2021, according to recent reports from the American Medical Group Association (AMGA) and the Medical Group Management Association (MGMA).
As patients started to return to doctors’ offices as the pandemic eased in 2021, physician groups found it increasingly difficult to recruit and retain lower-level clinicians, including medical assistants and LPNs, officials from both associations told this news organization. Many clinics had to raise their pay scales to be competitive with employers in other fields, and some had to hire higher-priced RNs to keep their practices running.
The AMGA report was based largely on data from groups of over 500 physicians, mostly affiliated with health systems. According to a news release accompanying the report, the ratio between full-time equivalent (FTE) clinic staff and health care professionals in direct patient care dropped by 11.3% between 2019 and 2021. The ratio of medical assistants (MAs) to clinicians declined by a greater percentage.
In the MGMA report, which represented about 4,000 practices ranging from very small (two doctors) to very large groups, total support staff per FTE primary-care physician dropped by 18% from 2019 to 2021 in independent groups and by 13% in hospital-affiliated groups. The ratios decreased by smaller amounts in surgical practices.
In contrast, nonsurgical specialty groups under both types of ownership saw their staffing ratios rise slightly.
Although it’s unclear why medical specialties increased their staff while other types of specialties lost employees, Ron Holder, MHA, chief operating officer of MGMA, said that some specialists may have opened more ancillary facilities and hired new employees to recoup revenue lost during the pandemic.
Expenses rise sharply
The AMGA report found that staffing expenses for the surveyed groups increased by 15% between 2019 and 2021.
“We saw a decrease in staff and an increase in expenses during that time period, and there are a few reasons for that,” Rose Wagner, RN, chief operating officer of AMGA, said. “Groups increased salaries to maintain staff. We also saw lower-paid staff find other jobs outside of health care. For example, medical assistants and receptionists could find jobs outside of health care that paid more. [Open positions] got back-filled with other higher paid staff, such as RNs, doing lower skilled jobs.”
Mr. Holder added that rising wages in other sectors made leaving physician groups more attractive for employees.
“Three years ago, there weren’t many positions in a medical practice where you were competing with Chick-fil-A or Taco Bell,” he said. In Denver, where Mr. Holder is based, “every restaurant in town is now advertising $17-$19 [hourly] starting pay just to do fast food. That causes practices to either lose employees or pay more for the employees they have. So that raises per-employee expense significantly,” he said.
Mr. Holder noted that inflation also has driven up wages as employees demand higher pay to keep up with the cost of living.
Unusual exodus of employees
Fred Horton, MHA, president of AMGA Consulting, said he has never seen so many people leaving health care for other occupations.
Some exits resulted from practices laying people off early in the pandemic, but most staff members who left practices were seeking higher pay, he said. In addition, Ms. Wagner noted, some staff members didn’t want to be exposed to COVID at work.
“There was an exodus from health care that was different from what we’d experienced in the past,” Mr. Horton added. “It’s still extremely challenging to get up to the staffing levels that are appropriate.”
Mr. Holder, however, said that the situation is slowly improving. “Health care is fairly recession-proof, because people need it. So when you see companies in other industries closing shop or reducing their head count, that actually helps health care recruiting in some jobs. And people are coming back to the workplace who previously were worried about COVID or didn’t want to get the vaccine.”
Paying more for nurses
In 2021, groups adopted a variety of tactics to adapt to the pandemic and respond to patient demand, the AMGA survey shows. Forty percent of system-affiliated groups and 18% of independent practices changed registered nurses’ responsibilities, in many cases having them do the work of medical assistants who were in short supply.
Some practices hired RNs, who have historically been utilized less by primary care than by surgical specialties, Mr. Holder noted. Other clinics paid temp agencies to supply nurses at a steep cost.
“When you’re short staffed, you end up paying more overtime, you end up paying temporary agencies at higher dollars, and you hire higher skilled people to do lower-skilled work,” Ms. Wagner said.
Meanwhile, many physician groups tried to cope with the physician shortage by bringing on more advanced practice clinicians (APCs), including nurse practitioners (NPs) and physician assistants (PAs). Seventy percent of the AMGA groups used this strategy, the report revealed.
“The use of APCs has been steadily increasing as groups try to adopt a lower-cost care model in the midst of a nationwide physician shortage,” Ms. Wagner said in the press release.
Changes in patient care
About half of the groups in the AMGA survey said they changed their staff structure to allow APCs to carry their own patient panels. Although most of these clinicians were probably under physician supervision, nearly half of the states now allow NPs to practice autonomously.
Mr. Horton cautioned that APCs can’t fully substitute for physicians and require the same support staff that doctors do if they have their own panels. In primary care groups, Mr. Holder noted, the average salary of an APC “is continuing to rise, and there isn’t a huge difference between what they and doctors make.”
Nevertheless, he added, “there are more NPs and PAs being added to the marketplace all the time, whereas [physician] residency programs aren’t really growing. There are caps on the number of residency positions, and some physicians are retiring. So the clock is ticking to the point where someday doctors will be grossly outnumbered by NPs.”
A version of this article first appeared on Medscape.com.
Physician groups saw staff-to-physician ratios decline even as their workforce expenses rose between 2019 and 2021, according to recent reports from the American Medical Group Association (AMGA) and the Medical Group Management Association (MGMA).
As patients started to return to doctors’ offices as the pandemic eased in 2021, physician groups found it increasingly difficult to recruit and retain lower-level clinicians, including medical assistants and LPNs, officials from both associations told this news organization. Many clinics had to raise their pay scales to be competitive with employers in other fields, and some had to hire higher-priced RNs to keep their practices running.
The AMGA report was based largely on data from groups of over 500 physicians, mostly affiliated with health systems. According to a news release accompanying the report, the ratio between full-time equivalent (FTE) clinic staff and health care professionals in direct patient care dropped by 11.3% between 2019 and 2021. The ratio of medical assistants (MAs) to clinicians declined by a greater percentage.
In the MGMA report, which represented about 4,000 practices ranging from very small (two doctors) to very large groups, total support staff per FTE primary-care physician dropped by 18% from 2019 to 2021 in independent groups and by 13% in hospital-affiliated groups. The ratios decreased by smaller amounts in surgical practices.
In contrast, nonsurgical specialty groups under both types of ownership saw their staffing ratios rise slightly.
Although it’s unclear why medical specialties increased their staff while other types of specialties lost employees, Ron Holder, MHA, chief operating officer of MGMA, said that some specialists may have opened more ancillary facilities and hired new employees to recoup revenue lost during the pandemic.
Expenses rise sharply
The AMGA report found that staffing expenses for the surveyed groups increased by 15% between 2019 and 2021.
“We saw a decrease in staff and an increase in expenses during that time period, and there are a few reasons for that,” Rose Wagner, RN, chief operating officer of AMGA, said. “Groups increased salaries to maintain staff. We also saw lower-paid staff find other jobs outside of health care. For example, medical assistants and receptionists could find jobs outside of health care that paid more. [Open positions] got back-filled with other higher paid staff, such as RNs, doing lower skilled jobs.”
Mr. Holder added that rising wages in other sectors made leaving physician groups more attractive for employees.
“Three years ago, there weren’t many positions in a medical practice where you were competing with Chick-fil-A or Taco Bell,” he said. In Denver, where Mr. Holder is based, “every restaurant in town is now advertising $17-$19 [hourly] starting pay just to do fast food. That causes practices to either lose employees or pay more for the employees they have. So that raises per-employee expense significantly,” he said.
Mr. Holder noted that inflation also has driven up wages as employees demand higher pay to keep up with the cost of living.
Unusual exodus of employees
Fred Horton, MHA, president of AMGA Consulting, said he has never seen so many people leaving health care for other occupations.
Some exits resulted from practices laying people off early in the pandemic, but most staff members who left practices were seeking higher pay, he said. In addition, Ms. Wagner noted, some staff members didn’t want to be exposed to COVID at work.
“There was an exodus from health care that was different from what we’d experienced in the past,” Mr. Horton added. “It’s still extremely challenging to get up to the staffing levels that are appropriate.”
Mr. Holder, however, said that the situation is slowly improving. “Health care is fairly recession-proof, because people need it. So when you see companies in other industries closing shop or reducing their head count, that actually helps health care recruiting in some jobs. And people are coming back to the workplace who previously were worried about COVID or didn’t want to get the vaccine.”
Paying more for nurses
In 2021, groups adopted a variety of tactics to adapt to the pandemic and respond to patient demand, the AMGA survey shows. Forty percent of system-affiliated groups and 18% of independent practices changed registered nurses’ responsibilities, in many cases having them do the work of medical assistants who were in short supply.
Some practices hired RNs, who have historically been utilized less by primary care than by surgical specialties, Mr. Holder noted. Other clinics paid temp agencies to supply nurses at a steep cost.
“When you’re short staffed, you end up paying more overtime, you end up paying temporary agencies at higher dollars, and you hire higher skilled people to do lower-skilled work,” Ms. Wagner said.
Meanwhile, many physician groups tried to cope with the physician shortage by bringing on more advanced practice clinicians (APCs), including nurse practitioners (NPs) and physician assistants (PAs). Seventy percent of the AMGA groups used this strategy, the report revealed.
“The use of APCs has been steadily increasing as groups try to adopt a lower-cost care model in the midst of a nationwide physician shortage,” Ms. Wagner said in the press release.
Changes in patient care
About half of the groups in the AMGA survey said they changed their staff structure to allow APCs to carry their own patient panels. Although most of these clinicians were probably under physician supervision, nearly half of the states now allow NPs to practice autonomously.
Mr. Horton cautioned that APCs can’t fully substitute for physicians and require the same support staff that doctors do if they have their own panels. In primary care groups, Mr. Holder noted, the average salary of an APC “is continuing to rise, and there isn’t a huge difference between what they and doctors make.”
Nevertheless, he added, “there are more NPs and PAs being added to the marketplace all the time, whereas [physician] residency programs aren’t really growing. There are caps on the number of residency positions, and some physicians are retiring. So the clock is ticking to the point where someday doctors will be grossly outnumbered by NPs.”
A version of this article first appeared on Medscape.com.
Ticking Time: Spreading Awareness About African Tick-Bite Fever
To the Editor:
One of the more common tick-borne infections seen in travelers returning from sub-Saharan Africa is caused by Rickettsia africae, which is the etiologic agent of African tick-bite fever (ATBF), the most common tick-borne bacterial zoonosis.1 There are 2 known tick vectors of disease: Amblyomma variegatum, found in sub-Saharan Africa and the West Indies, and Amblyomma hebraeum, found specifically in southern Africa.2,3
Unlike other disease-carrying ticks that passively wait on vegetation to be picked up by a host, A hebraeum uniquely attract other nearby ticks to the host. Studies have shown that male ticks feeding on a nonhuman host (usually cattle) can emit an aggression-attachment pheromone that attracts other ticks to the host. The presence of the pheromone allows unfed ticks to actively discriminate between hosts on which these parasites have fed successfully (ie, suitable hosts) and those on which they have not.4
The aggressive hunting nature of A hebraeum explains the clinical presentation of multiple eschars and why large groups of exposed travelers—such as soldiers, leisure safari tourists, game hunters, and foreign-aid workers—are affected.2
Another southern African spotted fever group, Rickettsia conorii is the causative agent of Mediterranean spotted fever (MSF). Ticks carrying R conorii exhibit a much less aggressive hunting stylethan A hebraeum; consequently, infected patients present with a single eschar site.5
Rickettsia africae is estimated to have very high prevalence (95.2%) in Amblyomma ticks and a fairly high prevalence (approximately 4.0% to 8.6%) in travelers coming from rural southern Africa,6,7 with an incubation period of 5 to 10 days after inoculation by an infected tick.8 Signs include fever, a generalized maculopapular or papulovesicular rash, and regional lymphadenopathy; symptoms include fatigue, headache, and myalgia.
The inoculation eschar—single or multiple—commonly presents on the legs and is accompanied by tender lymphadenopathy of draining nodes1,8 More severe findings, such as myocarditis and subacute neuropathy, have been reported in elderly patients.9
A 77-year-old man presented with a pruritic maculopapular and papulovesicular rash distributed over the upper and lower extremities of 3 weeks’ duration. The patient reported having been on a 12-day mission trip to Limpopo, South Africa, where he was constructing and installing safe toilets to replace dangerous toilet pits. He believed he had been bitten by 2 ticks, after which he noted a dark purple and black patch on the left lower leg by the third day of the trip. He developed a sudden persistent pruritic rash, first on the lower extremities and then spreading to the upper extremities. The patient was seen by an American physician in South Africa who gave him a 7-day course of oral doxycycline monohydrate 100 mg twice daily. He then returned to the United States.
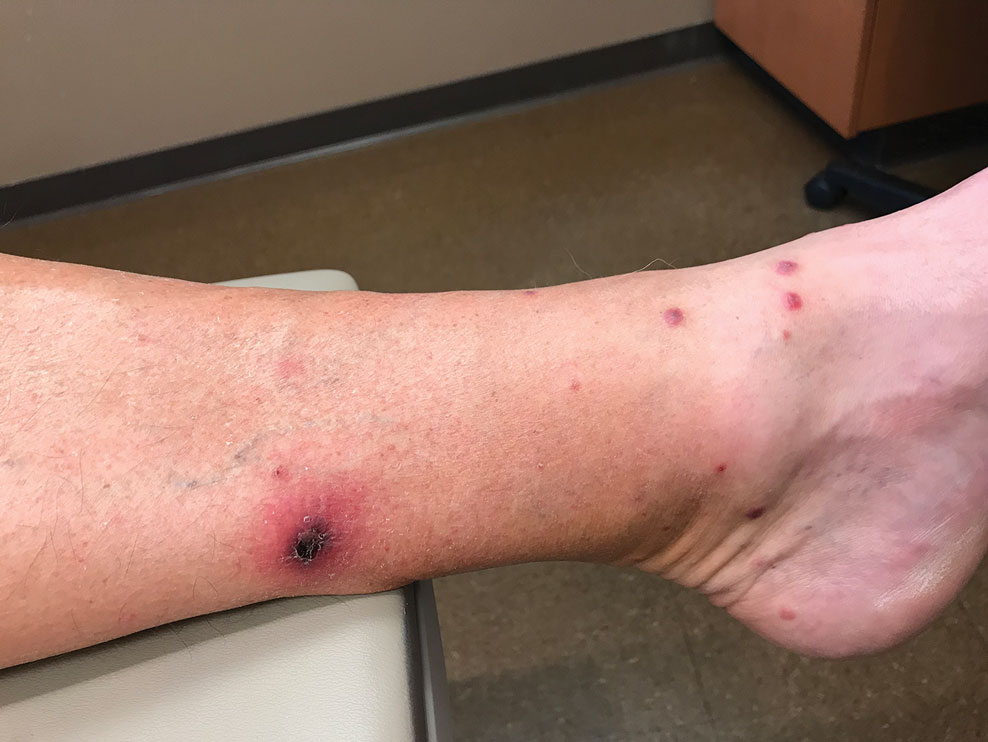
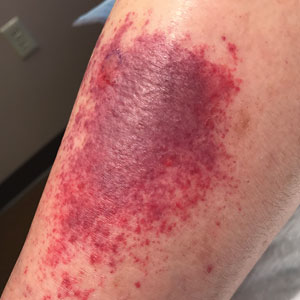
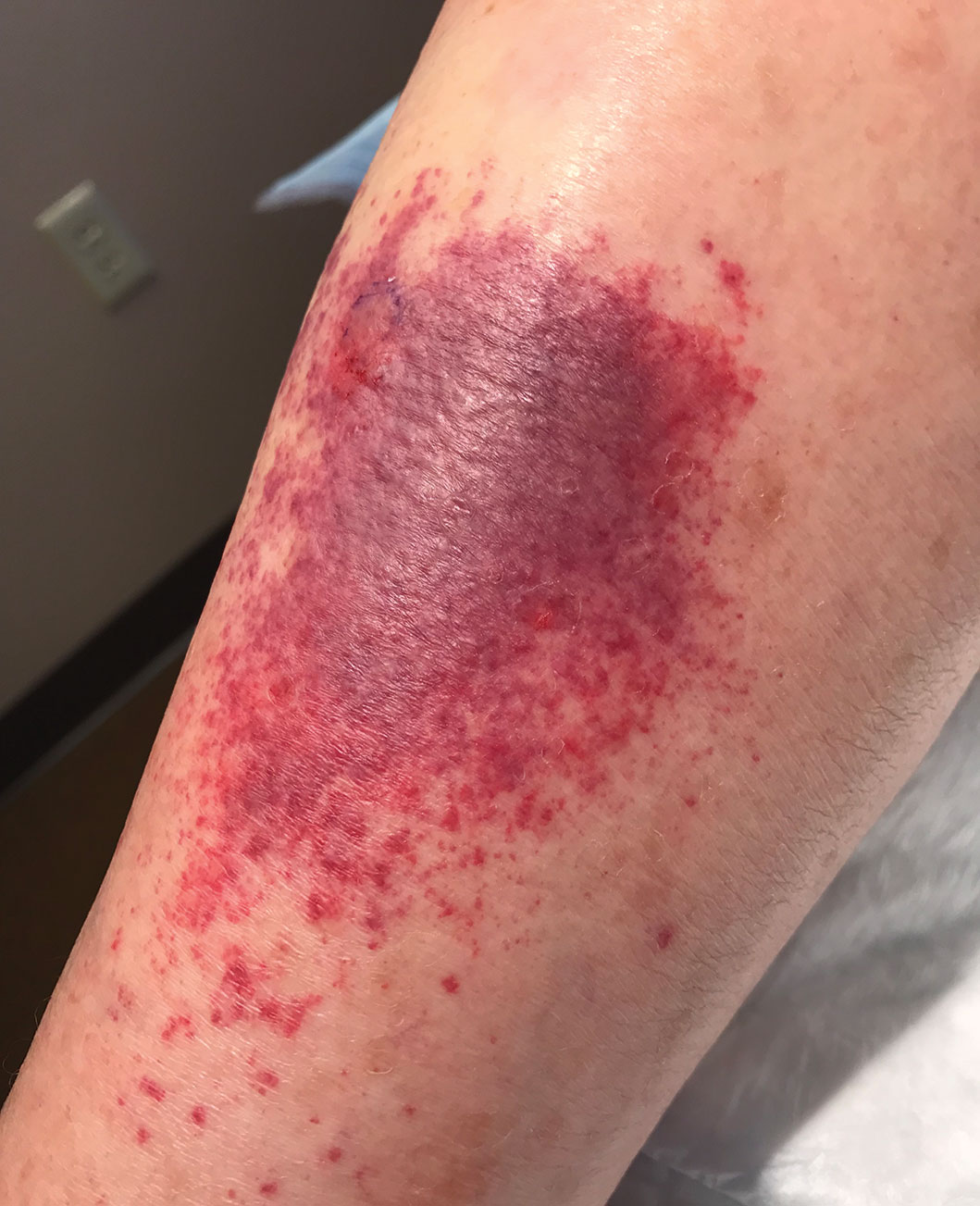
Sixteen days after being bitten by the ticks, the patient was examined in our dermatology office. Physical examination revealed an erythematous plaque with a central eschar over the medial aspect of the left leg (Figure 1) and multiple 3- to 6-mm, erythematous, dome-shaped papules scattered over the dorsal aspects of the feet and ankles (Figure 2). The examination was otherwise normal. Blood was drawn the same day for laboratory analysis; no abnormalities of platelets, red blood cells, or white blood cells were found. Results of a chemistry panel and liver enzyme tests were within reference range.
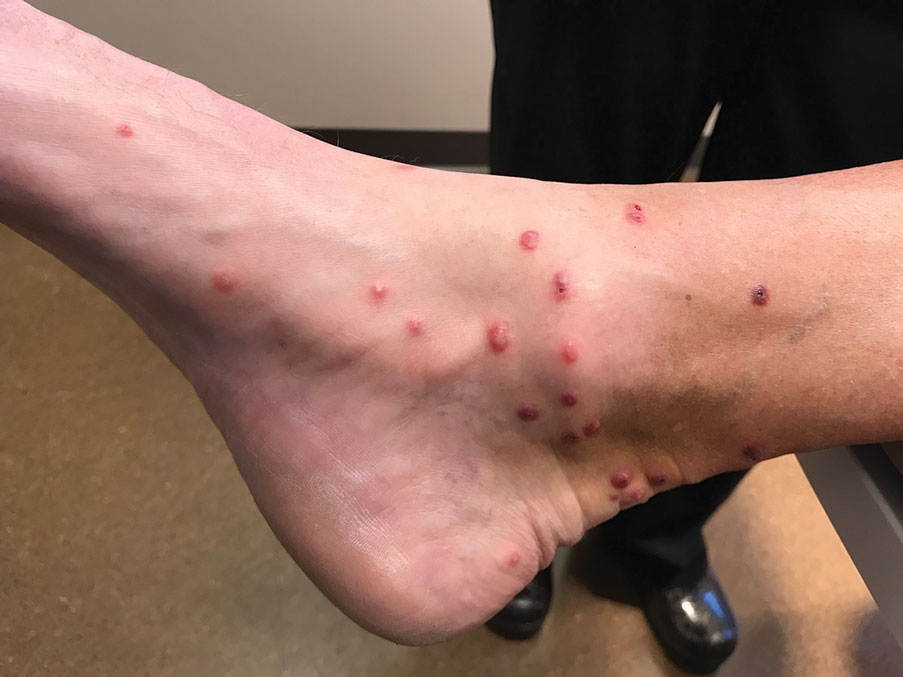
Skin biopsies were taken to elucidate the underlying pathology. Although an arthropod assault was suspected, there also was concern for deep vessel vasculitis because of the presence of considerable petechiae and purpura (Figure 3). Histologically, leukocytoclasia was seen in deep dermal blood vessels. A mild eosinophilic spongiosis with a mixed dermal infiltrate was identified—strengthening our suspicion of an arthropod assault. Bacterial cultures for aerobes and anaerobes using material taken from the right shin showed no growth.
Ten days after the initial biopsies, serum specimens were drawn and swabs of eschar were collected and sent to the Centers for Disease Control and Prevention for further testing. Serum was tested by immunofluorescence assay (IFA) for spotted fever group IgG to detect Rocky Mountain spotted fever and ATBF antibodies; both tests were negative. Swab material from eschar was tested again by IFA for spotted fever group IgG (Rocky Mountain spotted fever) and antibodies to ATBF and with bacterial culture isolation and nucleic acid amplification; the culture and amplification came back positive for R africae.
Because the specialized tests confirmed infection with R africae, the patient was given triamcinolone cream 0.1% to apply twice daily to the pruritic lesions for as long as 4 weeks; an additional 14-day course of oral doxycycline monohydrate (100 mg twice daily) was given. At follow-up, the lesions had fully resolved without evident scarring.
Various diagnostic techniques can detect R africae. Bacterial culture and the polymerase chain reaction are specific and therefore diagnostic. In addition, the diagnosis of rickettsiosis can be made with serology testing, in which disease-specific antibodies are detected by indirect IFA using disease-specific antigens.
Antigens from R rickettsii (the agent of Rocky Mountain spotted fever), R conorii (Mediterranean spotted fever), and R africae (ATBF) are commercially available for making the diagnosis of rickettsiosis. However, antigens from R conorii exhibit cross-reactivity with R africae, which can confound the diagnosis.1,10 Serologic IFA tests have been shown to be less sensitive, especially when performed after antibacterial treatment has started.
In a study, 17 of 65 (26%) ATBF-confirmed patients were seronegative (acute and convalescent-phase sera) against R africae; 14 had received doxycycline during the first week of clinical signs. The current hypothesis is that R africae is highly sensitive to doxycycline and that early exposure to the drug prevented development of detectable titers of reactive antibodies, thus producing a negative serology test.11
Furthermore, it has been shown that seroconversion of IgG and IgM antibodies in R africae–infected sera is delayed compared to what is observed with R conorii–infected sera. Typically, seroconversion of R conorii–infected sera can be detected within 2 weeks; seroconversion in R africae–infected sera can take 4 weeks or longer.11
Our patient had a confirmed case of ATBF secondary to R africae infection, which was evident from tissue culture isolation and polymerase chain reaction analysis of swab material obtained from eschar sites, both of which yielded a positive result for R africae. The traveler’s negative serologic status might be due to his early exposure to doxycycline or to the 4-week delay in R africae seroconversion; his serum was collected only 3 weeks after the tick bites.
Clinical signs also aid in making the diagnosis of ATBF and distinguishing R conorii from R africae infection. Because of the aggressive hunting nature of the tick carrying R africae, they are associated with multiple eschars and tend to affect groups of multiple people, especially in rural areas.4,5 In contrast, ticks carrying R conorii yield a single eschar due to their passive style of infecting a host and because they favor a single host within urban areas.5 Both infections exhibit a maculopapular or papulovesicular rash and are accompanied by fatigue, headache, and myalgia, though ATBF tends to present with a milder rash than MSF.
Infection with either R conorii or R africae responds to tetracyclines, quinolones, and macrolides.10,12
African tick-bite fever is becoming more common, which should encourage clinicians to become familiar with the disease. Less than 2 decades ago, ATBF virtually was unknown outside of Zimbabwe, Botswana, Tanzania, Zambia, and Kenya, where it is endemic. However, after the abolition of apartheid in the 1990s, international tourism in southern Africa increased 6-fold.13 African tick-bite fever is now one of the most common rickettsial infections in Africa.7 In addition to diagnosing ATBF and managing infected patients, clinicians can help prevent ATBF in individuals who travel to endemic areas by recommending commercial topical insect repellents containing at least 19.5% N,N-diethyl-meta-toluamide (DEET).14
- Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657-702. doi:10.1128/CMR.00032-13
- Jensenius M, Fournier P-E, Vene S, et al; Norwegian African Tick Bite Fever Study Group. African tick bite fever in travelers to rural sub-equatorial Africa. Clin Infect Dis. 2003;36:1411-1417. doi:10.1086/375083
- Parola P, Jourdan J, Raoult D. Tick-borne infection caused by Rickettsia africae in the West Indies. N Engl J Med. 1998;338:1391-1392. doi:10.1056/NEJM199805073381918
- Norval RA, Andrew HR, Yunker CE. Pheromone-mediation of host-selection in bont ticks (Amblyomma hebraeum Koch). Science. 1989;243:364-365. doi:10.1126/science.2911745
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928. doi:10.1086/319347
- Kelly PJ, Mason PR. Transmission of a spotted fever group rickettsia by Amblyomma hebraeum (Acari: Ixodidae). J Med Entomol. 1991;28:598-600. doi:10.1093/jmedent/28.5.598
- Maina AN, Jiang J, Omulo SA, et al. High prevalence of Rickettsia africae variants in Amblyomma variegatum ticks from domestic mammals in rural Western Kenya: implications for human health. Vector Borne Zoonotic Dis. 2014;14:693-702. doi:10.1089/vbz.2014.1578
- Jensenius M, Fournier P-E, Kelly P, et al. African tick bite fever. Lancet Infect Dis. 2003;3:557-564. doi:10.1016/s1473-3099(03)00739-4
- Roch N, Epaulard O, Pelloux I, et al. African tick bite fever in elderly patients: 8 cases in French tourists returning from South Africa. Clin Infect Dis. 2008;47:E28-E35. doi:10.1086/589868
- Palau L, Pankey GA. Mediterranean spotted fever in travelers from the United States. J Travel Med. 1997;4:179-182. doi:10.1111/j.1708-8305.1997.tb00816.x
- Fournier P-E, Jensenius M, Laferl H, et al. Kinetics of antibody responses in Rickettsia africae and Rickettsia conorii infections. Clin Diagn Lab Immunol. 2002;9:324-328. doi:10.1128/cdli.9.2.324-328.2002
- Brouqui P, Bacellar F, Baranton G, et al; ; . Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10:1108-1132. doi:10.1111/j.1469-0691.2004.01019.x
- Rolain JM, Jensenius M, Raoult D. Rickettsial infections—a threat to travelers? Curr Opin Infect Dis. 2004;17:433-437. doi:10.1097/00001432-200410000-00008
- Jensenius M, Pretorius AM, Clarke F, et al. Repellent efficacy of four commercial DEET lotions against Amblyomma hebraeum (Acari: Ixodidae), the principal vector of Rickettsia africae in southern Africa. Trans R Soc Trop Med Hyg. 2005;99:708-711. doi:10.1016/j.trstmh.2005.01.006
To the Editor:
One of the more common tick-borne infections seen in travelers returning from sub-Saharan Africa is caused by Rickettsia africae, which is the etiologic agent of African tick-bite fever (ATBF), the most common tick-borne bacterial zoonosis.1 There are 2 known tick vectors of disease: Amblyomma variegatum, found in sub-Saharan Africa and the West Indies, and Amblyomma hebraeum, found specifically in southern Africa.2,3
Unlike other disease-carrying ticks that passively wait on vegetation to be picked up by a host, A hebraeum uniquely attract other nearby ticks to the host. Studies have shown that male ticks feeding on a nonhuman host (usually cattle) can emit an aggression-attachment pheromone that attracts other ticks to the host. The presence of the pheromone allows unfed ticks to actively discriminate between hosts on which these parasites have fed successfully (ie, suitable hosts) and those on which they have not.4
The aggressive hunting nature of A hebraeum explains the clinical presentation of multiple eschars and why large groups of exposed travelers—such as soldiers, leisure safari tourists, game hunters, and foreign-aid workers—are affected.2
Another southern African spotted fever group, Rickettsia conorii is the causative agent of Mediterranean spotted fever (MSF). Ticks carrying R conorii exhibit a much less aggressive hunting stylethan A hebraeum; consequently, infected patients present with a single eschar site.5
Rickettsia africae is estimated to have very high prevalence (95.2%) in Amblyomma ticks and a fairly high prevalence (approximately 4.0% to 8.6%) in travelers coming from rural southern Africa,6,7 with an incubation period of 5 to 10 days after inoculation by an infected tick.8 Signs include fever, a generalized maculopapular or papulovesicular rash, and regional lymphadenopathy; symptoms include fatigue, headache, and myalgia.
The inoculation eschar—single or multiple—commonly presents on the legs and is accompanied by tender lymphadenopathy of draining nodes1,8 More severe findings, such as myocarditis and subacute neuropathy, have been reported in elderly patients.9
A 77-year-old man presented with a pruritic maculopapular and papulovesicular rash distributed over the upper and lower extremities of 3 weeks’ duration. The patient reported having been on a 12-day mission trip to Limpopo, South Africa, where he was constructing and installing safe toilets to replace dangerous toilet pits. He believed he had been bitten by 2 ticks, after which he noted a dark purple and black patch on the left lower leg by the third day of the trip. He developed a sudden persistent pruritic rash, first on the lower extremities and then spreading to the upper extremities. The patient was seen by an American physician in South Africa who gave him a 7-day course of oral doxycycline monohydrate 100 mg twice daily. He then returned to the United States.

Sixteen days after being bitten by the ticks, the patient was examined in our dermatology office. Physical examination revealed an erythematous plaque with a central eschar over the medial aspect of the left leg (Figure 1) and multiple 3- to 6-mm, erythematous, dome-shaped papules scattered over the dorsal aspects of the feet and ankles (Figure 2). The examination was otherwise normal. Blood was drawn the same day for laboratory analysis; no abnormalities of platelets, red blood cells, or white blood cells were found. Results of a chemistry panel and liver enzyme tests were within reference range.

Skin biopsies were taken to elucidate the underlying pathology. Although an arthropod assault was suspected, there also was concern for deep vessel vasculitis because of the presence of considerable petechiae and purpura (Figure 3). Histologically, leukocytoclasia was seen in deep dermal blood vessels. A mild eosinophilic spongiosis with a mixed dermal infiltrate was identified—strengthening our suspicion of an arthropod assault. Bacterial cultures for aerobes and anaerobes using material taken from the right shin showed no growth.

Ten days after the initial biopsies, serum specimens were drawn and swabs of eschar were collected and sent to the Centers for Disease Control and Prevention for further testing. Serum was tested by immunofluorescence assay (IFA) for spotted fever group IgG to detect Rocky Mountain spotted fever and ATBF antibodies; both tests were negative. Swab material from eschar was tested again by IFA for spotted fever group IgG (Rocky Mountain spotted fever) and antibodies to ATBF and with bacterial culture isolation and nucleic acid amplification; the culture and amplification came back positive for R africae.
Because the specialized tests confirmed infection with R africae, the patient was given triamcinolone cream 0.1% to apply twice daily to the pruritic lesions for as long as 4 weeks; an additional 14-day course of oral doxycycline monohydrate (100 mg twice daily) was given. At follow-up, the lesions had fully resolved without evident scarring.
Various diagnostic techniques can detect R africae. Bacterial culture and the polymerase chain reaction are specific and therefore diagnostic. In addition, the diagnosis of rickettsiosis can be made with serology testing, in which disease-specific antibodies are detected by indirect IFA using disease-specific antigens.
Antigens from R rickettsii (the agent of Rocky Mountain spotted fever), R conorii (Mediterranean spotted fever), and R africae (ATBF) are commercially available for making the diagnosis of rickettsiosis. However, antigens from R conorii exhibit cross-reactivity with R africae, which can confound the diagnosis.1,10 Serologic IFA tests have been shown to be less sensitive, especially when performed after antibacterial treatment has started.
In a study, 17 of 65 (26%) ATBF-confirmed patients were seronegative (acute and convalescent-phase sera) against R africae; 14 had received doxycycline during the first week of clinical signs. The current hypothesis is that R africae is highly sensitive to doxycycline and that early exposure to the drug prevented development of detectable titers of reactive antibodies, thus producing a negative serology test.11
Furthermore, it has been shown that seroconversion of IgG and IgM antibodies in R africae–infected sera is delayed compared to what is observed with R conorii–infected sera. Typically, seroconversion of R conorii–infected sera can be detected within 2 weeks; seroconversion in R africae–infected sera can take 4 weeks or longer.11
Our patient had a confirmed case of ATBF secondary to R africae infection, which was evident from tissue culture isolation and polymerase chain reaction analysis of swab material obtained from eschar sites, both of which yielded a positive result for R africae. The traveler’s negative serologic status might be due to his early exposure to doxycycline or to the 4-week delay in R africae seroconversion; his serum was collected only 3 weeks after the tick bites.
Clinical signs also aid in making the diagnosis of ATBF and distinguishing R conorii from R africae infection. Because of the aggressive hunting nature of the tick carrying R africae, they are associated with multiple eschars and tend to affect groups of multiple people, especially in rural areas.4,5 In contrast, ticks carrying R conorii yield a single eschar due to their passive style of infecting a host and because they favor a single host within urban areas.5 Both infections exhibit a maculopapular or papulovesicular rash and are accompanied by fatigue, headache, and myalgia, though ATBF tends to present with a milder rash than MSF.
Infection with either R conorii or R africae responds to tetracyclines, quinolones, and macrolides.10,12
African tick-bite fever is becoming more common, which should encourage clinicians to become familiar with the disease. Less than 2 decades ago, ATBF virtually was unknown outside of Zimbabwe, Botswana, Tanzania, Zambia, and Kenya, where it is endemic. However, after the abolition of apartheid in the 1990s, international tourism in southern Africa increased 6-fold.13 African tick-bite fever is now one of the most common rickettsial infections in Africa.7 In addition to diagnosing ATBF and managing infected patients, clinicians can help prevent ATBF in individuals who travel to endemic areas by recommending commercial topical insect repellents containing at least 19.5% N,N-diethyl-meta-toluamide (DEET).14
To the Editor:
One of the more common tick-borne infections seen in travelers returning from sub-Saharan Africa is caused by Rickettsia africae, which is the etiologic agent of African tick-bite fever (ATBF), the most common tick-borne bacterial zoonosis.1 There are 2 known tick vectors of disease: Amblyomma variegatum, found in sub-Saharan Africa and the West Indies, and Amblyomma hebraeum, found specifically in southern Africa.2,3
Unlike other disease-carrying ticks that passively wait on vegetation to be picked up by a host, A hebraeum uniquely attract other nearby ticks to the host. Studies have shown that male ticks feeding on a nonhuman host (usually cattle) can emit an aggression-attachment pheromone that attracts other ticks to the host. The presence of the pheromone allows unfed ticks to actively discriminate between hosts on which these parasites have fed successfully (ie, suitable hosts) and those on which they have not.4
The aggressive hunting nature of A hebraeum explains the clinical presentation of multiple eschars and why large groups of exposed travelers—such as soldiers, leisure safari tourists, game hunters, and foreign-aid workers—are affected.2
Another southern African spotted fever group, Rickettsia conorii is the causative agent of Mediterranean spotted fever (MSF). Ticks carrying R conorii exhibit a much less aggressive hunting stylethan A hebraeum; consequently, infected patients present with a single eschar site.5
Rickettsia africae is estimated to have very high prevalence (95.2%) in Amblyomma ticks and a fairly high prevalence (approximately 4.0% to 8.6%) in travelers coming from rural southern Africa,6,7 with an incubation period of 5 to 10 days after inoculation by an infected tick.8 Signs include fever, a generalized maculopapular or papulovesicular rash, and regional lymphadenopathy; symptoms include fatigue, headache, and myalgia.
The inoculation eschar—single or multiple—commonly presents on the legs and is accompanied by tender lymphadenopathy of draining nodes1,8 More severe findings, such as myocarditis and subacute neuropathy, have been reported in elderly patients.9
A 77-year-old man presented with a pruritic maculopapular and papulovesicular rash distributed over the upper and lower extremities of 3 weeks’ duration. The patient reported having been on a 12-day mission trip to Limpopo, South Africa, where he was constructing and installing safe toilets to replace dangerous toilet pits. He believed he had been bitten by 2 ticks, after which he noted a dark purple and black patch on the left lower leg by the third day of the trip. He developed a sudden persistent pruritic rash, first on the lower extremities and then spreading to the upper extremities. The patient was seen by an American physician in South Africa who gave him a 7-day course of oral doxycycline monohydrate 100 mg twice daily. He then returned to the United States.

Sixteen days after being bitten by the ticks, the patient was examined in our dermatology office. Physical examination revealed an erythematous plaque with a central eschar over the medial aspect of the left leg (Figure 1) and multiple 3- to 6-mm, erythematous, dome-shaped papules scattered over the dorsal aspects of the feet and ankles (Figure 2). The examination was otherwise normal. Blood was drawn the same day for laboratory analysis; no abnormalities of platelets, red blood cells, or white blood cells were found. Results of a chemistry panel and liver enzyme tests were within reference range.

Skin biopsies were taken to elucidate the underlying pathology. Although an arthropod assault was suspected, there also was concern for deep vessel vasculitis because of the presence of considerable petechiae and purpura (Figure 3). Histologically, leukocytoclasia was seen in deep dermal blood vessels. A mild eosinophilic spongiosis with a mixed dermal infiltrate was identified—strengthening our suspicion of an arthropod assault. Bacterial cultures for aerobes and anaerobes using material taken from the right shin showed no growth.

Ten days after the initial biopsies, serum specimens were drawn and swabs of eschar were collected and sent to the Centers for Disease Control and Prevention for further testing. Serum was tested by immunofluorescence assay (IFA) for spotted fever group IgG to detect Rocky Mountain spotted fever and ATBF antibodies; both tests were negative. Swab material from eschar was tested again by IFA for spotted fever group IgG (Rocky Mountain spotted fever) and antibodies to ATBF and with bacterial culture isolation and nucleic acid amplification; the culture and amplification came back positive for R africae.
Because the specialized tests confirmed infection with R africae, the patient was given triamcinolone cream 0.1% to apply twice daily to the pruritic lesions for as long as 4 weeks; an additional 14-day course of oral doxycycline monohydrate (100 mg twice daily) was given. At follow-up, the lesions had fully resolved without evident scarring.
Various diagnostic techniques can detect R africae. Bacterial culture and the polymerase chain reaction are specific and therefore diagnostic. In addition, the diagnosis of rickettsiosis can be made with serology testing, in which disease-specific antibodies are detected by indirect IFA using disease-specific antigens.
Antigens from R rickettsii (the agent of Rocky Mountain spotted fever), R conorii (Mediterranean spotted fever), and R africae (ATBF) are commercially available for making the diagnosis of rickettsiosis. However, antigens from R conorii exhibit cross-reactivity with R africae, which can confound the diagnosis.1,10 Serologic IFA tests have been shown to be less sensitive, especially when performed after antibacterial treatment has started.
In a study, 17 of 65 (26%) ATBF-confirmed patients were seronegative (acute and convalescent-phase sera) against R africae; 14 had received doxycycline during the first week of clinical signs. The current hypothesis is that R africae is highly sensitive to doxycycline and that early exposure to the drug prevented development of detectable titers of reactive antibodies, thus producing a negative serology test.11
Furthermore, it has been shown that seroconversion of IgG and IgM antibodies in R africae–infected sera is delayed compared to what is observed with R conorii–infected sera. Typically, seroconversion of R conorii–infected sera can be detected within 2 weeks; seroconversion in R africae–infected sera can take 4 weeks or longer.11
Our patient had a confirmed case of ATBF secondary to R africae infection, which was evident from tissue culture isolation and polymerase chain reaction analysis of swab material obtained from eschar sites, both of which yielded a positive result for R africae. The traveler’s negative serologic status might be due to his early exposure to doxycycline or to the 4-week delay in R africae seroconversion; his serum was collected only 3 weeks after the tick bites.
Clinical signs also aid in making the diagnosis of ATBF and distinguishing R conorii from R africae infection. Because of the aggressive hunting nature of the tick carrying R africae, they are associated with multiple eschars and tend to affect groups of multiple people, especially in rural areas.4,5 In contrast, ticks carrying R conorii yield a single eschar due to their passive style of infecting a host and because they favor a single host within urban areas.5 Both infections exhibit a maculopapular or papulovesicular rash and are accompanied by fatigue, headache, and myalgia, though ATBF tends to present with a milder rash than MSF.
Infection with either R conorii or R africae responds to tetracyclines, quinolones, and macrolides.10,12
African tick-bite fever is becoming more common, which should encourage clinicians to become familiar with the disease. Less than 2 decades ago, ATBF virtually was unknown outside of Zimbabwe, Botswana, Tanzania, Zambia, and Kenya, where it is endemic. However, after the abolition of apartheid in the 1990s, international tourism in southern Africa increased 6-fold.13 African tick-bite fever is now one of the most common rickettsial infections in Africa.7 In addition to diagnosing ATBF and managing infected patients, clinicians can help prevent ATBF in individuals who travel to endemic areas by recommending commercial topical insect repellents containing at least 19.5% N,N-diethyl-meta-toluamide (DEET).14
- Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657-702. doi:10.1128/CMR.00032-13
- Jensenius M, Fournier P-E, Vene S, et al; Norwegian African Tick Bite Fever Study Group. African tick bite fever in travelers to rural sub-equatorial Africa. Clin Infect Dis. 2003;36:1411-1417. doi:10.1086/375083
- Parola P, Jourdan J, Raoult D. Tick-borne infection caused by Rickettsia africae in the West Indies. N Engl J Med. 1998;338:1391-1392. doi:10.1056/NEJM199805073381918
- Norval RA, Andrew HR, Yunker CE. Pheromone-mediation of host-selection in bont ticks (Amblyomma hebraeum Koch). Science. 1989;243:364-365. doi:10.1126/science.2911745
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928. doi:10.1086/319347
- Kelly PJ, Mason PR. Transmission of a spotted fever group rickettsia by Amblyomma hebraeum (Acari: Ixodidae). J Med Entomol. 1991;28:598-600. doi:10.1093/jmedent/28.5.598
- Maina AN, Jiang J, Omulo SA, et al. High prevalence of Rickettsia africae variants in Amblyomma variegatum ticks from domestic mammals in rural Western Kenya: implications for human health. Vector Borne Zoonotic Dis. 2014;14:693-702. doi:10.1089/vbz.2014.1578
- Jensenius M, Fournier P-E, Kelly P, et al. African tick bite fever. Lancet Infect Dis. 2003;3:557-564. doi:10.1016/s1473-3099(03)00739-4
- Roch N, Epaulard O, Pelloux I, et al. African tick bite fever in elderly patients: 8 cases in French tourists returning from South Africa. Clin Infect Dis. 2008;47:E28-E35. doi:10.1086/589868
- Palau L, Pankey GA. Mediterranean spotted fever in travelers from the United States. J Travel Med. 1997;4:179-182. doi:10.1111/j.1708-8305.1997.tb00816.x
- Fournier P-E, Jensenius M, Laferl H, et al. Kinetics of antibody responses in Rickettsia africae and Rickettsia conorii infections. Clin Diagn Lab Immunol. 2002;9:324-328. doi:10.1128/cdli.9.2.324-328.2002
- Brouqui P, Bacellar F, Baranton G, et al; ; . Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10:1108-1132. doi:10.1111/j.1469-0691.2004.01019.x
- Rolain JM, Jensenius M, Raoult D. Rickettsial infections—a threat to travelers? Curr Opin Infect Dis. 2004;17:433-437. doi:10.1097/00001432-200410000-00008
- Jensenius M, Pretorius AM, Clarke F, et al. Repellent efficacy of four commercial DEET lotions against Amblyomma hebraeum (Acari: Ixodidae), the principal vector of Rickettsia africae in southern Africa. Trans R Soc Trop Med Hyg. 2005;99:708-711. doi:10.1016/j.trstmh.2005.01.006
- Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657-702. doi:10.1128/CMR.00032-13
- Jensenius M, Fournier P-E, Vene S, et al; Norwegian African Tick Bite Fever Study Group. African tick bite fever in travelers to rural sub-equatorial Africa. Clin Infect Dis. 2003;36:1411-1417. doi:10.1086/375083
- Parola P, Jourdan J, Raoult D. Tick-borne infection caused by Rickettsia africae in the West Indies. N Engl J Med. 1998;338:1391-1392. doi:10.1056/NEJM199805073381918
- Norval RA, Andrew HR, Yunker CE. Pheromone-mediation of host-selection in bont ticks (Amblyomma hebraeum Koch). Science. 1989;243:364-365. doi:10.1126/science.2911745
- Parola P, Raoult D. Ticks and tickborne bacterial diseases in humans: an emerging infectious threat. Clin Infect Dis. 2001;32:897-928. doi:10.1086/319347
- Kelly PJ, Mason PR. Transmission of a spotted fever group rickettsia by Amblyomma hebraeum (Acari: Ixodidae). J Med Entomol. 1991;28:598-600. doi:10.1093/jmedent/28.5.598
- Maina AN, Jiang J, Omulo SA, et al. High prevalence of Rickettsia africae variants in Amblyomma variegatum ticks from domestic mammals in rural Western Kenya: implications for human health. Vector Borne Zoonotic Dis. 2014;14:693-702. doi:10.1089/vbz.2014.1578
- Jensenius M, Fournier P-E, Kelly P, et al. African tick bite fever. Lancet Infect Dis. 2003;3:557-564. doi:10.1016/s1473-3099(03)00739-4
- Roch N, Epaulard O, Pelloux I, et al. African tick bite fever in elderly patients: 8 cases in French tourists returning from South Africa. Clin Infect Dis. 2008;47:E28-E35. doi:10.1086/589868
- Palau L, Pankey GA. Mediterranean spotted fever in travelers from the United States. J Travel Med. 1997;4:179-182. doi:10.1111/j.1708-8305.1997.tb00816.x
- Fournier P-E, Jensenius M, Laferl H, et al. Kinetics of antibody responses in Rickettsia africae and Rickettsia conorii infections. Clin Diagn Lab Immunol. 2002;9:324-328. doi:10.1128/cdli.9.2.324-328.2002
- Brouqui P, Bacellar F, Baranton G, et al; ; . Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10:1108-1132. doi:10.1111/j.1469-0691.2004.01019.x
- Rolain JM, Jensenius M, Raoult D. Rickettsial infections—a threat to travelers? Curr Opin Infect Dis. 2004;17:433-437. doi:10.1097/00001432-200410000-00008
- Jensenius M, Pretorius AM, Clarke F, et al. Repellent efficacy of four commercial DEET lotions against Amblyomma hebraeum (Acari: Ixodidae), the principal vector of Rickettsia africae in southern Africa. Trans R Soc Trop Med Hyg. 2005;99:708-711. doi:10.1016/j.trstmh.2005.01.006
PRACTICE POINTS
- African tick-bite fever (ATBF) is one of the more common tick-borne bacterial zoonoses and should be considered in patients presenting with multiple eschar sites who have had exposure to rural areas of southern Africa in the preceding 2 weeks.
- Ticks carrying Rickettsia africae are unique, given their ability to actively be attracted to and hunt their nonhuman hosts via an aggression-attachment pheromone.
- Laboratory diagnosis of ATBF can be challenging due to the high cross-reactivity of Helvetica Neue LT StdR africae with Helvetica Neue LT StdRickettsia conorii in serologic testing and the delay in seroconversion in Helvetica Neue LT StdR africae infection.
Doctors and their families tend to ignore medical guidelines
according to a study by economic professors from the Massachusetts Institute of Technology, Cambridge; Stanford (Calif.) University; and the George Gund Professor of Economics and Business Administration at Harvard University, Boston.
What to know
- Doctors’ medical knowledge may influence them and their families to often ignore medical advice while the rest of the population adheres to general medication guidelines.
- Of the 63 guidelines used in the study, doctors and their families followed the standards less than a third of the time.
- The difference in adherence to guidelines between experts and nonexperts is largest with respect to antibiotics, in which doctors and their families are 5.2 percentage points less in compliance than everyone else.
- Doctors could be more likely to prescribe broader-spectrum antibiotics for themselves and their families, whereas most patients receive more narrow-spectrum antibiotics.
- Many members of the general public don’t understand medical guidelines, finding them too complex to follow, and many people don’t trust their doctors.
This is a summary of the article, “A Taste of Their Own Medicine: Guideline Adherence and Access to Expertise,” published in the American Economic Review: Insights on December 13, 2022. The full article can be found on aeaweb.org.
A version of this article first appeared on Medscape.com.
according to a study by economic professors from the Massachusetts Institute of Technology, Cambridge; Stanford (Calif.) University; and the George Gund Professor of Economics and Business Administration at Harvard University, Boston.
What to know
- Doctors’ medical knowledge may influence them and their families to often ignore medical advice while the rest of the population adheres to general medication guidelines.
- Of the 63 guidelines used in the study, doctors and their families followed the standards less than a third of the time.
- The difference in adherence to guidelines between experts and nonexperts is largest with respect to antibiotics, in which doctors and their families are 5.2 percentage points less in compliance than everyone else.
- Doctors could be more likely to prescribe broader-spectrum antibiotics for themselves and their families, whereas most patients receive more narrow-spectrum antibiotics.
- Many members of the general public don’t understand medical guidelines, finding them too complex to follow, and many people don’t trust their doctors.
This is a summary of the article, “A Taste of Their Own Medicine: Guideline Adherence and Access to Expertise,” published in the American Economic Review: Insights on December 13, 2022. The full article can be found on aeaweb.org.
A version of this article first appeared on Medscape.com.
according to a study by economic professors from the Massachusetts Institute of Technology, Cambridge; Stanford (Calif.) University; and the George Gund Professor of Economics and Business Administration at Harvard University, Boston.
What to know
- Doctors’ medical knowledge may influence them and their families to often ignore medical advice while the rest of the population adheres to general medication guidelines.
- Of the 63 guidelines used in the study, doctors and their families followed the standards less than a third of the time.
- The difference in adherence to guidelines between experts and nonexperts is largest with respect to antibiotics, in which doctors and their families are 5.2 percentage points less in compliance than everyone else.
- Doctors could be more likely to prescribe broader-spectrum antibiotics for themselves and their families, whereas most patients receive more narrow-spectrum antibiotics.
- Many members of the general public don’t understand medical guidelines, finding them too complex to follow, and many people don’t trust their doctors.
This is a summary of the article, “A Taste of Their Own Medicine: Guideline Adherence and Access to Expertise,” published in the American Economic Review: Insights on December 13, 2022. The full article can be found on aeaweb.org.
A version of this article first appeared on Medscape.com.
Docs with one paid malpractice claim are four times more likely to have another
In this retrospective case-control study, law and public health researchers from Georgetown University, the National Opinion Research Center, the University of Colorado, and Northwestern University analyzed paid malpractice claims for all licensed U.S. physicians.
The findings suggest that a single malpractice claim may not be a random stroke of bad luck but instead holds some predictive power into the risk for future paid claims.
“A four times increase in risk is huge, particularly since we observe a similar increase in both high-risk and lower-risk specialties,” David Hyman, JD, MD, professor of health law and policy at Georgetown University, Washington, and lead researcher on the study, told this news organization. “There are surely some false positives, but there must be lots of actual negligence too, or we would not see these results.”
For the 881,876 physicians analyzed, researchers looked at malpractice claims paid during two 5-year periods: 2009-2013 and 2014-2018. Nearly 96% of physicians had no paid malpractice claims between 2009 and 2013; 3% had one, and less than 1% had multiple claims. The proportion of physicians with paid claims between 2014 and 2018 was similar.
Compared with physicians with no 2009-2013 claims, a physician with just one paid claim in that time period had a 3.7 times higher risk for a future paid claim. Physicians with two paid claims were nearly 7 times more likely to have a future paid claim, and those with three or more paid claims were more than 11 times more likely to have one.
Approximately 3% of physicians with no paid claims between 2009 and 2013 had a future paid claim, growing to 12.4% of those with one paid claim during that time.
The study’s findings may have implications for medical licensing boards and hospitals granting staff privileges.
“After some number of paid claims, there should be an official response” from these entities, such as a hands-on assessment of technical skills or assignment of a peer mentor, said Dr. Hyman, who is also coauthor of a book titled “Medical Malpractice Litigation: How It Works, Why Tort Reform Hasn’t Helped.” A graduated set of interventions, whether voluntary or mandatory, can reduce future claim risk and patient harm, Dr. Hyman added.
Interventions may include error avoidance and post-error communication training, counseling to improve bedside skills, and encouragement to move into nonclinical practice. Either way, Dr. Hyman says a nuanced intervention strategy would be a welcome shift away from the current “all or nothing approach” that too often ends in the revocation of a physician’s medical license.
Although there are strategies to proactively identify physicians with excess risk for malpractice claims and implement preventive measures – like Vanderbilt University’s Patient Advocacy Reporting System, for example – most hospitals and physician groups fail to initiate even informal interventions after a malpractice settlement or verdict, which is a missed opportunity, Dr. Hyman said.
A version of this article first appeared on Medscape.com.
In this retrospective case-control study, law and public health researchers from Georgetown University, the National Opinion Research Center, the University of Colorado, and Northwestern University analyzed paid malpractice claims for all licensed U.S. physicians.
The findings suggest that a single malpractice claim may not be a random stroke of bad luck but instead holds some predictive power into the risk for future paid claims.
“A four times increase in risk is huge, particularly since we observe a similar increase in both high-risk and lower-risk specialties,” David Hyman, JD, MD, professor of health law and policy at Georgetown University, Washington, and lead researcher on the study, told this news organization. “There are surely some false positives, but there must be lots of actual negligence too, or we would not see these results.”
For the 881,876 physicians analyzed, researchers looked at malpractice claims paid during two 5-year periods: 2009-2013 and 2014-2018. Nearly 96% of physicians had no paid malpractice claims between 2009 and 2013; 3% had one, and less than 1% had multiple claims. The proportion of physicians with paid claims between 2014 and 2018 was similar.
Compared with physicians with no 2009-2013 claims, a physician with just one paid claim in that time period had a 3.7 times higher risk for a future paid claim. Physicians with two paid claims were nearly 7 times more likely to have a future paid claim, and those with three or more paid claims were more than 11 times more likely to have one.
Approximately 3% of physicians with no paid claims between 2009 and 2013 had a future paid claim, growing to 12.4% of those with one paid claim during that time.
The study’s findings may have implications for medical licensing boards and hospitals granting staff privileges.
“After some number of paid claims, there should be an official response” from these entities, such as a hands-on assessment of technical skills or assignment of a peer mentor, said Dr. Hyman, who is also coauthor of a book titled “Medical Malpractice Litigation: How It Works, Why Tort Reform Hasn’t Helped.” A graduated set of interventions, whether voluntary or mandatory, can reduce future claim risk and patient harm, Dr. Hyman added.
Interventions may include error avoidance and post-error communication training, counseling to improve bedside skills, and encouragement to move into nonclinical practice. Either way, Dr. Hyman says a nuanced intervention strategy would be a welcome shift away from the current “all or nothing approach” that too often ends in the revocation of a physician’s medical license.
Although there are strategies to proactively identify physicians with excess risk for malpractice claims and implement preventive measures – like Vanderbilt University’s Patient Advocacy Reporting System, for example – most hospitals and physician groups fail to initiate even informal interventions after a malpractice settlement or verdict, which is a missed opportunity, Dr. Hyman said.
A version of this article first appeared on Medscape.com.
In this retrospective case-control study, law and public health researchers from Georgetown University, the National Opinion Research Center, the University of Colorado, and Northwestern University analyzed paid malpractice claims for all licensed U.S. physicians.
The findings suggest that a single malpractice claim may not be a random stroke of bad luck but instead holds some predictive power into the risk for future paid claims.
“A four times increase in risk is huge, particularly since we observe a similar increase in both high-risk and lower-risk specialties,” David Hyman, JD, MD, professor of health law and policy at Georgetown University, Washington, and lead researcher on the study, told this news organization. “There are surely some false positives, but there must be lots of actual negligence too, or we would not see these results.”
For the 881,876 physicians analyzed, researchers looked at malpractice claims paid during two 5-year periods: 2009-2013 and 2014-2018. Nearly 96% of physicians had no paid malpractice claims between 2009 and 2013; 3% had one, and less than 1% had multiple claims. The proportion of physicians with paid claims between 2014 and 2018 was similar.
Compared with physicians with no 2009-2013 claims, a physician with just one paid claim in that time period had a 3.7 times higher risk for a future paid claim. Physicians with two paid claims were nearly 7 times more likely to have a future paid claim, and those with three or more paid claims were more than 11 times more likely to have one.
Approximately 3% of physicians with no paid claims between 2009 and 2013 had a future paid claim, growing to 12.4% of those with one paid claim during that time.
The study’s findings may have implications for medical licensing boards and hospitals granting staff privileges.
“After some number of paid claims, there should be an official response” from these entities, such as a hands-on assessment of technical skills or assignment of a peer mentor, said Dr. Hyman, who is also coauthor of a book titled “Medical Malpractice Litigation: How It Works, Why Tort Reform Hasn’t Helped.” A graduated set of interventions, whether voluntary or mandatory, can reduce future claim risk and patient harm, Dr. Hyman added.
Interventions may include error avoidance and post-error communication training, counseling to improve bedside skills, and encouragement to move into nonclinical practice. Either way, Dr. Hyman says a nuanced intervention strategy would be a welcome shift away from the current “all or nothing approach” that too often ends in the revocation of a physician’s medical license.
Although there are strategies to proactively identify physicians with excess risk for malpractice claims and implement preventive measures – like Vanderbilt University’s Patient Advocacy Reporting System, for example – most hospitals and physician groups fail to initiate even informal interventions after a malpractice settlement or verdict, which is a missed opportunity, Dr. Hyman said.
A version of this article first appeared on Medscape.com.
FROM JAMA
Autoantibodies signal reduced cancer risk in dermatomyositis
Adults with the inflammatory autoimmune myopathy dermatomyositis are at increased for concurrent cancers, but new research suggests that certain autoantibodies in patients with a specific dermatomyositis subtype may actually protect against cancer.
A study of cohorts of patients with dermatomyositis, other rheumatic diseases, and those without disease showed that among patients with dermatomyositis positive for antitranscriptional intermediary factor 1 (anti–TIF1-gamma) autoantibodies – a disease subtype associated with increased cancer risk – the presence of autoantibodies directed against cell division cycle and apoptosis regulator 1 (CCAR1) was associated with reduced cancer risk “to a level comparable to that seen in the general population,” Christopher A. Mecoli, MD, MHS, of Johns Hopkins University, Baltimore, and colleagues reported.
“Our prior data suggest that there are autoantigens that, when targeted simultaneously with CCAR1, provide additional cancer protection. Although these autoantigens are less frequently targeted, it is likely that additional, more prevalent ‘autoantigen hubs’ remain undiscovered,” they wrote in Arthritis & Rheumatology.
Identification of other autoantibodies both in the anti–TIF1-gamma–positive and other dermatomyositis subgroups may help with cancer risk stratification in patients with the disease and may ultimately improve cancer screening for adults with dermatomyositis, the investigators said.
Toward precision medicine
“I think this is a step toward precision medicine in patients with rheumatic disease, specifically myositis,” Dr. Mecoli said in an interview.
The study supports earlier work showing that dermatomyositis and related myopathies are heterogeneous, he said, noting that, “if you put 10 myositis patients in the same room, you wouldn’t get that they all have the same disease because they can look so different from one another.”
The association of dermatomyositis with concurrent cancers has been known for decades, but in recent years his team and other investigators have noted that the association holds true for only some patients with dermatomyositis, most notably those patients positive for anti–TIF1-gamma autoantibodies.
“And then, of course, once you really start studying just one gamma-positive dermatomyositis patient, you realize that even among that group it is heterogeneous in terms of their cancer risk, and that was the main focus of this study: to reconcile this clinical observation that I had a lot of patients with TIF1-gamma dermatomyositis who never get diagnosed with cancer,” Dr. Mecoli said.
Study details
Dr. Dr. Mecoli and colleagues previously reported that immune responses to CCAR1 and other autoantigens seen in patients with dermatomyositis were associated with lower probability of cancer occurrence.
In the current study, they focused on the disease specificity, clinical phenotype, and cancer risk for patients with dermatomyositis and anti-CCAR1 autoantibodies.
They looked at all patients aged 18 or older with a probable or definite finding of dermatomyositis, according to 2017 American College of Rheumatology/European Alliance of Associations for Rheumatology Idiopathic Inflammatory Myopathy criteria, who were seen at Stanford (Calif.) University Medical Center from August 2004 to April 2020 (101 patients), or the Johns Hopkins Myositis Center (141 patients) from January 2007 to December 2020.
Controls included 44 patients evaluated at the Johns Hopkins Myositis Center with immune-mediated necrotizing myopathy, 186 patients with anti–TIF1-gamma–negative dermatomyositis (defined as an enzyme-linked immunosorbent assay readout of less than seven units) evaluated at either Stanford or Johns Hopkins, 44 patients with inclusion body myositis evaluated at Johns Hopkins, and 46 patients with systemic lupus erythematosus from the Hopkins Lupus Cohort. The investigators also assayed serum from 32 healthy individuals.
They found that patients with anti–TIF1-gamma–positive dermatomyositis were significantly more likely than those with anti–TIF1-gamma–negative dermatomyositis to have anti-CCAR1 autoantibodies (32% vs. 8%; P < .001). Additionally, they noted that the anti-CCAR1 autoantibodies were not seen in serum from healthy controls and were found at only very low frequencies among patients with other rheumatic diseases.
When they looked at the incidence of cancer from the time of dermatomyositis onset (defined as the first patient-reported symptoms of rash, weakness, myalgia, or dyspnea) they found that the standardized incidence ratio in anti–TIF1-gamma–positive patients in both the Stanford and Hopkins cohorts was higher than expected, with SIRs of 3.49 and 4.54, respectively (P < .001 for each comparison).
However, among those patients who were both anti–TIF1-gamma positive and anti-CCAR1 positive, the SIRs were 1.78 in the Stanford cohort and 1.61 in the Hopkins cohort, and neither SIR was significantly higher than that of the general population.
Risk prediction
Their findings suggest that autoantibody profiles might be used for cancer risk stratification in patients with anti–TIF1-gamma–positive dermatomyositis, Dr. Mecoli said.
“Are we overscreening? What is the cost in terms of patient anxiety, in terms of radiation, and in terms of false positive results?” he asked. “If I had a patient in front of me with anti–TIF1-gamma dermatomyositis, I would probably manage them differently if I knew that they were CCAR1 positive, because the presence of that additional autoantibody attenuates their cancer risk relative to the general population.”
In an editorial accompanying the study, Manabu Fujimoto, MD, of the department of dermatology at Osaka (Japan) University, commented that it “is of clinical importance in that combination of autoantibodies can predict cancer risk with more accuracy. At the same time, this study will give an insight into the pathomechanisms of how antitumor activity may shape autoimmunity in dermatomyositis.”
It will be “intriguing” to discover whether anti-CCAR1 autoantibodies act only against tumors or might also have an impact on dermatomyositis itself, Dr. Fujimoto said.
The research was supported by grants from the National Institutes of Health; Huayi and Siuling Zhang Discovery Fund; Peter Buck, MD; and the Donald B. and Dorothy L. Stabler Foundation. The authors and Dr. Fujimoto reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adults with the inflammatory autoimmune myopathy dermatomyositis are at increased for concurrent cancers, but new research suggests that certain autoantibodies in patients with a specific dermatomyositis subtype may actually protect against cancer.
A study of cohorts of patients with dermatomyositis, other rheumatic diseases, and those without disease showed that among patients with dermatomyositis positive for antitranscriptional intermediary factor 1 (anti–TIF1-gamma) autoantibodies – a disease subtype associated with increased cancer risk – the presence of autoantibodies directed against cell division cycle and apoptosis regulator 1 (CCAR1) was associated with reduced cancer risk “to a level comparable to that seen in the general population,” Christopher A. Mecoli, MD, MHS, of Johns Hopkins University, Baltimore, and colleagues reported.
“Our prior data suggest that there are autoantigens that, when targeted simultaneously with CCAR1, provide additional cancer protection. Although these autoantigens are less frequently targeted, it is likely that additional, more prevalent ‘autoantigen hubs’ remain undiscovered,” they wrote in Arthritis & Rheumatology.
Identification of other autoantibodies both in the anti–TIF1-gamma–positive and other dermatomyositis subgroups may help with cancer risk stratification in patients with the disease and may ultimately improve cancer screening for adults with dermatomyositis, the investigators said.
Toward precision medicine
“I think this is a step toward precision medicine in patients with rheumatic disease, specifically myositis,” Dr. Mecoli said in an interview.
The study supports earlier work showing that dermatomyositis and related myopathies are heterogeneous, he said, noting that, “if you put 10 myositis patients in the same room, you wouldn’t get that they all have the same disease because they can look so different from one another.”
The association of dermatomyositis with concurrent cancers has been known for decades, but in recent years his team and other investigators have noted that the association holds true for only some patients with dermatomyositis, most notably those patients positive for anti–TIF1-gamma autoantibodies.
“And then, of course, once you really start studying just one gamma-positive dermatomyositis patient, you realize that even among that group it is heterogeneous in terms of their cancer risk, and that was the main focus of this study: to reconcile this clinical observation that I had a lot of patients with TIF1-gamma dermatomyositis who never get diagnosed with cancer,” Dr. Mecoli said.
Study details
Dr. Dr. Mecoli and colleagues previously reported that immune responses to CCAR1 and other autoantigens seen in patients with dermatomyositis were associated with lower probability of cancer occurrence.
In the current study, they focused on the disease specificity, clinical phenotype, and cancer risk for patients with dermatomyositis and anti-CCAR1 autoantibodies.
They looked at all patients aged 18 or older with a probable or definite finding of dermatomyositis, according to 2017 American College of Rheumatology/European Alliance of Associations for Rheumatology Idiopathic Inflammatory Myopathy criteria, who were seen at Stanford (Calif.) University Medical Center from August 2004 to April 2020 (101 patients), or the Johns Hopkins Myositis Center (141 patients) from January 2007 to December 2020.
Controls included 44 patients evaluated at the Johns Hopkins Myositis Center with immune-mediated necrotizing myopathy, 186 patients with anti–TIF1-gamma–negative dermatomyositis (defined as an enzyme-linked immunosorbent assay readout of less than seven units) evaluated at either Stanford or Johns Hopkins, 44 patients with inclusion body myositis evaluated at Johns Hopkins, and 46 patients with systemic lupus erythematosus from the Hopkins Lupus Cohort. The investigators also assayed serum from 32 healthy individuals.
They found that patients with anti–TIF1-gamma–positive dermatomyositis were significantly more likely than those with anti–TIF1-gamma–negative dermatomyositis to have anti-CCAR1 autoantibodies (32% vs. 8%; P < .001). Additionally, they noted that the anti-CCAR1 autoantibodies were not seen in serum from healthy controls and were found at only very low frequencies among patients with other rheumatic diseases.
When they looked at the incidence of cancer from the time of dermatomyositis onset (defined as the first patient-reported symptoms of rash, weakness, myalgia, or dyspnea) they found that the standardized incidence ratio in anti–TIF1-gamma–positive patients in both the Stanford and Hopkins cohorts was higher than expected, with SIRs of 3.49 and 4.54, respectively (P < .001 for each comparison).
However, among those patients who were both anti–TIF1-gamma positive and anti-CCAR1 positive, the SIRs were 1.78 in the Stanford cohort and 1.61 in the Hopkins cohort, and neither SIR was significantly higher than that of the general population.
Risk prediction
Their findings suggest that autoantibody profiles might be used for cancer risk stratification in patients with anti–TIF1-gamma–positive dermatomyositis, Dr. Mecoli said.
“Are we overscreening? What is the cost in terms of patient anxiety, in terms of radiation, and in terms of false positive results?” he asked. “If I had a patient in front of me with anti–TIF1-gamma dermatomyositis, I would probably manage them differently if I knew that they were CCAR1 positive, because the presence of that additional autoantibody attenuates their cancer risk relative to the general population.”
In an editorial accompanying the study, Manabu Fujimoto, MD, of the department of dermatology at Osaka (Japan) University, commented that it “is of clinical importance in that combination of autoantibodies can predict cancer risk with more accuracy. At the same time, this study will give an insight into the pathomechanisms of how antitumor activity may shape autoimmunity in dermatomyositis.”
It will be “intriguing” to discover whether anti-CCAR1 autoantibodies act only against tumors or might also have an impact on dermatomyositis itself, Dr. Fujimoto said.
The research was supported by grants from the National Institutes of Health; Huayi and Siuling Zhang Discovery Fund; Peter Buck, MD; and the Donald B. and Dorothy L. Stabler Foundation. The authors and Dr. Fujimoto reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adults with the inflammatory autoimmune myopathy dermatomyositis are at increased for concurrent cancers, but new research suggests that certain autoantibodies in patients with a specific dermatomyositis subtype may actually protect against cancer.
A study of cohorts of patients with dermatomyositis, other rheumatic diseases, and those without disease showed that among patients with dermatomyositis positive for antitranscriptional intermediary factor 1 (anti–TIF1-gamma) autoantibodies – a disease subtype associated with increased cancer risk – the presence of autoantibodies directed against cell division cycle and apoptosis regulator 1 (CCAR1) was associated with reduced cancer risk “to a level comparable to that seen in the general population,” Christopher A. Mecoli, MD, MHS, of Johns Hopkins University, Baltimore, and colleagues reported.
“Our prior data suggest that there are autoantigens that, when targeted simultaneously with CCAR1, provide additional cancer protection. Although these autoantigens are less frequently targeted, it is likely that additional, more prevalent ‘autoantigen hubs’ remain undiscovered,” they wrote in Arthritis & Rheumatology.
Identification of other autoantibodies both in the anti–TIF1-gamma–positive and other dermatomyositis subgroups may help with cancer risk stratification in patients with the disease and may ultimately improve cancer screening for adults with dermatomyositis, the investigators said.
Toward precision medicine
“I think this is a step toward precision medicine in patients with rheumatic disease, specifically myositis,” Dr. Mecoli said in an interview.
The study supports earlier work showing that dermatomyositis and related myopathies are heterogeneous, he said, noting that, “if you put 10 myositis patients in the same room, you wouldn’t get that they all have the same disease because they can look so different from one another.”
The association of dermatomyositis with concurrent cancers has been known for decades, but in recent years his team and other investigators have noted that the association holds true for only some patients with dermatomyositis, most notably those patients positive for anti–TIF1-gamma autoantibodies.
“And then, of course, once you really start studying just one gamma-positive dermatomyositis patient, you realize that even among that group it is heterogeneous in terms of their cancer risk, and that was the main focus of this study: to reconcile this clinical observation that I had a lot of patients with TIF1-gamma dermatomyositis who never get diagnosed with cancer,” Dr. Mecoli said.
Study details
Dr. Dr. Mecoli and colleagues previously reported that immune responses to CCAR1 and other autoantigens seen in patients with dermatomyositis were associated with lower probability of cancer occurrence.
In the current study, they focused on the disease specificity, clinical phenotype, and cancer risk for patients with dermatomyositis and anti-CCAR1 autoantibodies.
They looked at all patients aged 18 or older with a probable or definite finding of dermatomyositis, according to 2017 American College of Rheumatology/European Alliance of Associations for Rheumatology Idiopathic Inflammatory Myopathy criteria, who were seen at Stanford (Calif.) University Medical Center from August 2004 to April 2020 (101 patients), or the Johns Hopkins Myositis Center (141 patients) from January 2007 to December 2020.
Controls included 44 patients evaluated at the Johns Hopkins Myositis Center with immune-mediated necrotizing myopathy, 186 patients with anti–TIF1-gamma–negative dermatomyositis (defined as an enzyme-linked immunosorbent assay readout of less than seven units) evaluated at either Stanford or Johns Hopkins, 44 patients with inclusion body myositis evaluated at Johns Hopkins, and 46 patients with systemic lupus erythematosus from the Hopkins Lupus Cohort. The investigators also assayed serum from 32 healthy individuals.
They found that patients with anti–TIF1-gamma–positive dermatomyositis were significantly more likely than those with anti–TIF1-gamma–negative dermatomyositis to have anti-CCAR1 autoantibodies (32% vs. 8%; P < .001). Additionally, they noted that the anti-CCAR1 autoantibodies were not seen in serum from healthy controls and were found at only very low frequencies among patients with other rheumatic diseases.
When they looked at the incidence of cancer from the time of dermatomyositis onset (defined as the first patient-reported symptoms of rash, weakness, myalgia, or dyspnea) they found that the standardized incidence ratio in anti–TIF1-gamma–positive patients in both the Stanford and Hopkins cohorts was higher than expected, with SIRs of 3.49 and 4.54, respectively (P < .001 for each comparison).
However, among those patients who were both anti–TIF1-gamma positive and anti-CCAR1 positive, the SIRs were 1.78 in the Stanford cohort and 1.61 in the Hopkins cohort, and neither SIR was significantly higher than that of the general population.
Risk prediction
Their findings suggest that autoantibody profiles might be used for cancer risk stratification in patients with anti–TIF1-gamma–positive dermatomyositis, Dr. Mecoli said.
“Are we overscreening? What is the cost in terms of patient anxiety, in terms of radiation, and in terms of false positive results?” he asked. “If I had a patient in front of me with anti–TIF1-gamma dermatomyositis, I would probably manage them differently if I knew that they were CCAR1 positive, because the presence of that additional autoantibody attenuates their cancer risk relative to the general population.”
In an editorial accompanying the study, Manabu Fujimoto, MD, of the department of dermatology at Osaka (Japan) University, commented that it “is of clinical importance in that combination of autoantibodies can predict cancer risk with more accuracy. At the same time, this study will give an insight into the pathomechanisms of how antitumor activity may shape autoimmunity in dermatomyositis.”
It will be “intriguing” to discover whether anti-CCAR1 autoantibodies act only against tumors or might also have an impact on dermatomyositis itself, Dr. Fujimoto said.
The research was supported by grants from the National Institutes of Health; Huayi and Siuling Zhang Discovery Fund; Peter Buck, MD; and the Donald B. and Dorothy L. Stabler Foundation. The authors and Dr. Fujimoto reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ARTHRITIS & RHEUMATOLOGY
Treating rosacea: Combination therapy, benzoyl peroxide, and the ‘STOP’ mnemonic
HONOLULU – More often than not, patients with rosacea require a combination of treatments to optimize the management of the disease, according to Julie C. Harper, MD.
“We’ve been more comfortable with the idea of combination therapy for acne than we have been for rosacea,” Dr. Harper, who practices in Birmingham, Ala., said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “If patients are doing great on one treatment, then don’t change it. But if there’s room for improvement, think about combinations.”
Treatment options for papules and pustules include ivermectin, metronidazole, azelaic acid, sodium sulfacetamide/sulfur, modified release doxycycline, minocycline foam, and microencapsulated benzoyl peroxide, 5%. Options for persistent background erythema include brimonidine and oxymetazoline, as well as device-based treatments, which include the pulsed dye laser, the KTP laser, intense pulsed light, and electrosurgery.
Dr. Harper said that she has been especially surprised by the effectiveness of one of these options, microencapsulated benzoyl peroxide cream, 5% (Epsolay), which is approved by the Food and Drug Administration for treating inflammatory lesions of rosacea in adults. In two identical, phase 3 randomized clinical trials of patients with inflammatory rosacea lesions, those treated with microencapsulated benzoyl peroxide achieved a 68.8% reduction in inflammatory lesions at 12 weeks (including 42.5% at week 2), compared with 38%-46% of those on the vehicle, according to the April 2022 announcement of the approval from the manufacturers, Sol-Gel Technologies and Galderma.
“A common drug is playing a key role,” Dr. Harper said. “What’s the mechanism of action? I have no idea. I wonder if there may be a bacterial pathogen after all,” possibly Staphylococcus epidermidis, she added. However, she noted, “it does appear that benzoyl peroxide has an impact on Demodex, so maybe that’s the primary way it’s working.”
In her opinion, a key standout from the clinical trial data is the drug’s rapid onset of action, with a 42.5% reduction of lesions at week 2. “What makes this different is that the 5% microencapsulated benzoyl peroxide cream is wrapped up in a silica shell,” said Dr. Harper, a past president of the American Acne and Rosacea Society. “The silica shell kind of acts like a speed bump that slows the release of drug onto the skin. We think that’s what may be giving us this better tolerability.”
In an interview at the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that prior to the approval of Epsolay, benzoyl peroxide was never considered a first-line treatment for rosacea. “The problem is, the conventional formulation is irritating to the skin,” said Dr. Stein Gold, who was involved in clinical trials of Epsolay.
“The benzoyl peroxide encapsulated in the silica shell allows for a slow and steady delivery of medication to the skin in a very controlled manner. It is exceptionally good at getting rosacea under control. In the clinical trials, when we looked at the baseline irritation of the skin and followed those patients when they used the benzoyl 5% microencapsulated benzoyl peroxide cream, the irritation improved.”
‘STOP’ mnemonic
When treating her patients with rosacea, Dr. Harper incorporates the mnemonic “STOP” to these patient visits:
S: Identify signs and symptoms of rosacea.
T: Discuss triggers. “We cannot make this disease triggerless, so when you’re talking to your patients, you need to find out what’s triggering their rosacea,” she said.
O: Agree on a treatment outcome. “What is it that’s important to the patient?” she said. “They may tell you, ‘I want to be able to not be so red,’ or ‘I want to get rid of the bumps,’ or ‘I want my eyes to not feel so dry.’ ”
P: Develop a plan that helps achieve that desired outcome with patients.
Dr. Harper disclosed ties with Almirall, Cassiopeia, Cutera, Galderma, EPI, L’Oréal, Ortho Dermatologics, Sol Gel, Sun Pharmaceutical Industries, and Vyne.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries.
Medscape and this news organization are owned by the same parent company.
HONOLULU – More often than not, patients with rosacea require a combination of treatments to optimize the management of the disease, according to Julie C. Harper, MD.
“We’ve been more comfortable with the idea of combination therapy for acne than we have been for rosacea,” Dr. Harper, who practices in Birmingham, Ala., said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “If patients are doing great on one treatment, then don’t change it. But if there’s room for improvement, think about combinations.”
Treatment options for papules and pustules include ivermectin, metronidazole, azelaic acid, sodium sulfacetamide/sulfur, modified release doxycycline, minocycline foam, and microencapsulated benzoyl peroxide, 5%. Options for persistent background erythema include brimonidine and oxymetazoline, as well as device-based treatments, which include the pulsed dye laser, the KTP laser, intense pulsed light, and electrosurgery.
Dr. Harper said that she has been especially surprised by the effectiveness of one of these options, microencapsulated benzoyl peroxide cream, 5% (Epsolay), which is approved by the Food and Drug Administration for treating inflammatory lesions of rosacea in adults. In two identical, phase 3 randomized clinical trials of patients with inflammatory rosacea lesions, those treated with microencapsulated benzoyl peroxide achieved a 68.8% reduction in inflammatory lesions at 12 weeks (including 42.5% at week 2), compared with 38%-46% of those on the vehicle, according to the April 2022 announcement of the approval from the manufacturers, Sol-Gel Technologies and Galderma.
“A common drug is playing a key role,” Dr. Harper said. “What’s the mechanism of action? I have no idea. I wonder if there may be a bacterial pathogen after all,” possibly Staphylococcus epidermidis, she added. However, she noted, “it does appear that benzoyl peroxide has an impact on Demodex, so maybe that’s the primary way it’s working.”
In her opinion, a key standout from the clinical trial data is the drug’s rapid onset of action, with a 42.5% reduction of lesions at week 2. “What makes this different is that the 5% microencapsulated benzoyl peroxide cream is wrapped up in a silica shell,” said Dr. Harper, a past president of the American Acne and Rosacea Society. “The silica shell kind of acts like a speed bump that slows the release of drug onto the skin. We think that’s what may be giving us this better tolerability.”
In an interview at the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that prior to the approval of Epsolay, benzoyl peroxide was never considered a first-line treatment for rosacea. “The problem is, the conventional formulation is irritating to the skin,” said Dr. Stein Gold, who was involved in clinical trials of Epsolay.
“The benzoyl peroxide encapsulated in the silica shell allows for a slow and steady delivery of medication to the skin in a very controlled manner. It is exceptionally good at getting rosacea under control. In the clinical trials, when we looked at the baseline irritation of the skin and followed those patients when they used the benzoyl 5% microencapsulated benzoyl peroxide cream, the irritation improved.”
‘STOP’ mnemonic
When treating her patients with rosacea, Dr. Harper incorporates the mnemonic “STOP” to these patient visits:
S: Identify signs and symptoms of rosacea.
T: Discuss triggers. “We cannot make this disease triggerless, so when you’re talking to your patients, you need to find out what’s triggering their rosacea,” she said.
O: Agree on a treatment outcome. “What is it that’s important to the patient?” she said. “They may tell you, ‘I want to be able to not be so red,’ or ‘I want to get rid of the bumps,’ or ‘I want my eyes to not feel so dry.’ ”
P: Develop a plan that helps achieve that desired outcome with patients.
Dr. Harper disclosed ties with Almirall, Cassiopeia, Cutera, Galderma, EPI, L’Oréal, Ortho Dermatologics, Sol Gel, Sun Pharmaceutical Industries, and Vyne.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries.
Medscape and this news organization are owned by the same parent company.
HONOLULU – More often than not, patients with rosacea require a combination of treatments to optimize the management of the disease, according to Julie C. Harper, MD.
“We’ve been more comfortable with the idea of combination therapy for acne than we have been for rosacea,” Dr. Harper, who practices in Birmingham, Ala., said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “If patients are doing great on one treatment, then don’t change it. But if there’s room for improvement, think about combinations.”
Treatment options for papules and pustules include ivermectin, metronidazole, azelaic acid, sodium sulfacetamide/sulfur, modified release doxycycline, minocycline foam, and microencapsulated benzoyl peroxide, 5%. Options for persistent background erythema include brimonidine and oxymetazoline, as well as device-based treatments, which include the pulsed dye laser, the KTP laser, intense pulsed light, and electrosurgery.
Dr. Harper said that she has been especially surprised by the effectiveness of one of these options, microencapsulated benzoyl peroxide cream, 5% (Epsolay), which is approved by the Food and Drug Administration for treating inflammatory lesions of rosacea in adults. In two identical, phase 3 randomized clinical trials of patients with inflammatory rosacea lesions, those treated with microencapsulated benzoyl peroxide achieved a 68.8% reduction in inflammatory lesions at 12 weeks (including 42.5% at week 2), compared with 38%-46% of those on the vehicle, according to the April 2022 announcement of the approval from the manufacturers, Sol-Gel Technologies and Galderma.
“A common drug is playing a key role,” Dr. Harper said. “What’s the mechanism of action? I have no idea. I wonder if there may be a bacterial pathogen after all,” possibly Staphylococcus epidermidis, she added. However, she noted, “it does appear that benzoyl peroxide has an impact on Demodex, so maybe that’s the primary way it’s working.”
In her opinion, a key standout from the clinical trial data is the drug’s rapid onset of action, with a 42.5% reduction of lesions at week 2. “What makes this different is that the 5% microencapsulated benzoyl peroxide cream is wrapped up in a silica shell,” said Dr. Harper, a past president of the American Acne and Rosacea Society. “The silica shell kind of acts like a speed bump that slows the release of drug onto the skin. We think that’s what may be giving us this better tolerability.”
In an interview at the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that prior to the approval of Epsolay, benzoyl peroxide was never considered a first-line treatment for rosacea. “The problem is, the conventional formulation is irritating to the skin,” said Dr. Stein Gold, who was involved in clinical trials of Epsolay.
“The benzoyl peroxide encapsulated in the silica shell allows for a slow and steady delivery of medication to the skin in a very controlled manner. It is exceptionally good at getting rosacea under control. In the clinical trials, when we looked at the baseline irritation of the skin and followed those patients when they used the benzoyl 5% microencapsulated benzoyl peroxide cream, the irritation improved.”
‘STOP’ mnemonic
When treating her patients with rosacea, Dr. Harper incorporates the mnemonic “STOP” to these patient visits:
S: Identify signs and symptoms of rosacea.
T: Discuss triggers. “We cannot make this disease triggerless, so when you’re talking to your patients, you need to find out what’s triggering their rosacea,” she said.
O: Agree on a treatment outcome. “What is it that’s important to the patient?” she said. “They may tell you, ‘I want to be able to not be so red,’ or ‘I want to get rid of the bumps,’ or ‘I want my eyes to not feel so dry.’ ”
P: Develop a plan that helps achieve that desired outcome with patients.
Dr. Harper disclosed ties with Almirall, Cassiopeia, Cutera, Galderma, EPI, L’Oréal, Ortho Dermatologics, Sol Gel, Sun Pharmaceutical Industries, and Vyne.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries.
Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPE LIVE! HAWAII DERMATOLOGY SEMINAR
Zika virus still calls for preparedness and vaccine development
Warming U.S. temperatures, the resumption of travel, and new knowledge about Zika’s long-term effects on children signal that Zika prevention and vaccine development should be on public health officials’, doctors’, and communities’ radar, even when community infection is not occurring.
“Although we haven’t seen confirmed Zika virus circulation in the continental United States or its territories for several years, it’s still something that we are closely monitoring, particularly as we move into the summer months,” Erin Staples, MD, PhD, medical epidemiologist at the Arboviral Diseases Branch of the Centers for Disease Control and Prevention in Fort Collins, Colo., told this news organization.
“This is because cases are still being reported in other countries, particularly in South America. Travel to these places is increasing following the pandemic, leaving more potential for individuals who might have acquired the infection to come back and restart community transmission.”
How Zika might reemerge
The Aedes aegypti mosquito is the vector by which Zika spreads, and “during the COVID pandemic, these mosquitoes moved further north in the United States, into southern California, and were identified as far north as Washington, D.C.,” said Neil Silverman, MD, professor of clinical obstetrics and gynecology and director of the Infections in Pregnancy Program at UCLA Medical Center in Los Angeles.
“On a population level, Americans have essentially no immunity to Zika from prior infection, and there is no vaccine yet approved. If individuals infected with Zika came into a U.S. region where the Aedes aegypti mosquito was present, that population could be very susceptible to infection spread and even another outbreak. This would be a confluence of bad circumstances, but that’s exactly what infectious disease specialists continue to be watchful about, especially because Zika is so dangerous for fetuses,” said Dr. Silverman.
How the public can prepare
The CDC recommends that pregnant women or women who plan to become pregnant avoid traveling to regions where there are currently outbreaks of Zika, but this is not the only way that individuals can protect themselves.
“The message we want to deliver to people is that in the United States, people are at risk for several mosquito-borne diseases every summer beyond just Zika,” Dr. Staples said. “It’s really important that people are instructed to make a habit of wearing EPA [U.S. Environmental Protection Agency)–registered insect repellents when they go outside. Right now, that is the single best tool that we have to prevent mosquito-borne diseases in the U.S.
“From a community standpoint, there are several emerging mosquito control methods that are being evaluated right now, such as genetic modification and irradiation of mosquitoes. These methods are aimed at producing sterile mosquitoes that are released into the wild to mate with the local mosquito population, which will render them infertile. This leads, over time, to suppression of the overall Aedes aegypti mosquito population – the main vector of Zika transmission,” said Dr. Staples.
Monica Gandhi, MD, MPH, professor of medicine and associate chief of the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco, encourages her patients to wear mosquito repellent but cautioned that “there’s no antiviral that you can take for Zika. Until we have a vaccine, the key to controlling/preventing Zika is controlling the mosquitoes that spread the virus.”
Vaccines
The National Institute of Allergy and Infectious Diseases (NIAID) is currently investigating a variety of Zika vaccines, including a DNA-based vaccine, (phase 2), a purified inactivated virus vaccine (phase 1), live attenuated vaccines (phase 2), and mRNA vaccines (phase 2).
“I’m most excited about mRNA vaccines because they help patients produce a lot of proteins. The protein from a typical protein-adjuvant vaccine will break down, and patients can only raise an immune response to whatever proteins are left. On the other hand, mRNA vaccines provide the body [with] a recipe to make the protein from the pathogen in high amounts, so that a strong immune response can be raised for protection,” noted Dr. Gandhi.
Moderna’s mRNA-1893 vaccine was recently studied in a randomized, observer-blind, controlled, phase 1 trial among 120 adults in the United States and Puerto Rico, the results of which were published online in The Lancet. “The vaccine was found to be generally well tolerated with no serious adverse events considered related to vaccine. Furthermore, the vaccine was able to generate a potent immune response that was capable of neutralizing the virus in vitro,” said Brett Leav, MD, executive director of clinical development for public health vaccines at Moderna.
“Our mRNA platform technology ... can be very helpful against emerging pandemic threats, as we saw in response to COVID-19. What is unique in our approach is that if the genetic sequence of the virus is known, we can quickly generate vaccines to test for their capability to generate a functional immune response. In the case of the mRNA-1893 trial, the vaccine was developed with antigens that were present in the strain of virus circulating in 2016, but we could easily match whatever strain reemerges,” said Dr. Leav.
A phase 2 trial to confirm the dose of mRNA-1893 in a larger study population is underway.
Although it’s been demonstrated that Moderna’s mRNA vaccine is safe and effective, moving from a phase 2 to a phase 3 study presents a challenge, given the fact that currently, the disease burden from Zika is low. If an outbreak were to occur in the future, these mRNA vaccines could potentially be given emergency approval, as occurred during the COVID pandemic, according to Dr. Silverman.
If approved, provisionally or through a traditional route, the vaccine would “accelerate the ability to tamp down any further outbreaks, because vaccine-based immunity could be made available to a large portion of the population who were pregnant or planning a pregnancy, not just in the U.S. but also in these endemic areas,” said Dr. Silverman.
Takeaways from the last Zika outbreak
Practical steps such as mosquito eradication and development of vaccines are not the only takeaway from the recent Zika epidemics inside and outside the United States. A clearer picture of the short- and long-term stakes of the disease has emerged.
According to the CDC, most people who become infected with Zika experience only mild symptoms, such as fever, rash, headache, and muscle pain, but babies conceived by mothers infected with Zika are at risk for stillbirth, miscarriage, and microcephaly and other brain defects.
Although a pregnant woman who tests positive for Zika is in a very high-risk situation, “data show that only about 30% of mothers with Zika have a baby with birth defects. If a pregnant woman contracted Zika, what would happen is we would just do very close screening by ultrasound of the fetus. If microcephaly in utero or fetal brain defects were observed, then a mother would be counseled on her options,” said Dr. Gandhi.
Dr. Silverman noted that “new data on children who were exposed in utero and had normal exams, including head measurements when they were born, have raised concerns. In recently published long-term follow-up studies, even when children born to mothers infected with Zika during pregnancy had normal head growth at least 3 years after birth, they were still at risk for neurodevelopmental delay and behavioral disorders, including impact on coordination and executive function.
“This is another good reason to keep the potential risks of Zika active in the public’s consciousness and in public health planning.”
Dr. Silverman, Dr. Gandhi, and Dr. Staples have disclosed no relevant financial relationships. Dr. Leav is an employee of Moderna and owns stock in the company.
A version of this article originally appeared on Medscape.com.
Warming U.S. temperatures, the resumption of travel, and new knowledge about Zika’s long-term effects on children signal that Zika prevention and vaccine development should be on public health officials’, doctors’, and communities’ radar, even when community infection is not occurring.
“Although we haven’t seen confirmed Zika virus circulation in the continental United States or its territories for several years, it’s still something that we are closely monitoring, particularly as we move into the summer months,” Erin Staples, MD, PhD, medical epidemiologist at the Arboviral Diseases Branch of the Centers for Disease Control and Prevention in Fort Collins, Colo., told this news organization.
“This is because cases are still being reported in other countries, particularly in South America. Travel to these places is increasing following the pandemic, leaving more potential for individuals who might have acquired the infection to come back and restart community transmission.”
How Zika might reemerge
The Aedes aegypti mosquito is the vector by which Zika spreads, and “during the COVID pandemic, these mosquitoes moved further north in the United States, into southern California, and were identified as far north as Washington, D.C.,” said Neil Silverman, MD, professor of clinical obstetrics and gynecology and director of the Infections in Pregnancy Program at UCLA Medical Center in Los Angeles.
“On a population level, Americans have essentially no immunity to Zika from prior infection, and there is no vaccine yet approved. If individuals infected with Zika came into a U.S. region where the Aedes aegypti mosquito was present, that population could be very susceptible to infection spread and even another outbreak. This would be a confluence of bad circumstances, but that’s exactly what infectious disease specialists continue to be watchful about, especially because Zika is so dangerous for fetuses,” said Dr. Silverman.
How the public can prepare
The CDC recommends that pregnant women or women who plan to become pregnant avoid traveling to regions where there are currently outbreaks of Zika, but this is not the only way that individuals can protect themselves.
“The message we want to deliver to people is that in the United States, people are at risk for several mosquito-borne diseases every summer beyond just Zika,” Dr. Staples said. “It’s really important that people are instructed to make a habit of wearing EPA [U.S. Environmental Protection Agency)–registered insect repellents when they go outside. Right now, that is the single best tool that we have to prevent mosquito-borne diseases in the U.S.
“From a community standpoint, there are several emerging mosquito control methods that are being evaluated right now, such as genetic modification and irradiation of mosquitoes. These methods are aimed at producing sterile mosquitoes that are released into the wild to mate with the local mosquito population, which will render them infertile. This leads, over time, to suppression of the overall Aedes aegypti mosquito population – the main vector of Zika transmission,” said Dr. Staples.
Monica Gandhi, MD, MPH, professor of medicine and associate chief of the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco, encourages her patients to wear mosquito repellent but cautioned that “there’s no antiviral that you can take for Zika. Until we have a vaccine, the key to controlling/preventing Zika is controlling the mosquitoes that spread the virus.”
Vaccines
The National Institute of Allergy and Infectious Diseases (NIAID) is currently investigating a variety of Zika vaccines, including a DNA-based vaccine, (phase 2), a purified inactivated virus vaccine (phase 1), live attenuated vaccines (phase 2), and mRNA vaccines (phase 2).
“I’m most excited about mRNA vaccines because they help patients produce a lot of proteins. The protein from a typical protein-adjuvant vaccine will break down, and patients can only raise an immune response to whatever proteins are left. On the other hand, mRNA vaccines provide the body [with] a recipe to make the protein from the pathogen in high amounts, so that a strong immune response can be raised for protection,” noted Dr. Gandhi.
Moderna’s mRNA-1893 vaccine was recently studied in a randomized, observer-blind, controlled, phase 1 trial among 120 adults in the United States and Puerto Rico, the results of which were published online in The Lancet. “The vaccine was found to be generally well tolerated with no serious adverse events considered related to vaccine. Furthermore, the vaccine was able to generate a potent immune response that was capable of neutralizing the virus in vitro,” said Brett Leav, MD, executive director of clinical development for public health vaccines at Moderna.
“Our mRNA platform technology ... can be very helpful against emerging pandemic threats, as we saw in response to COVID-19. What is unique in our approach is that if the genetic sequence of the virus is known, we can quickly generate vaccines to test for their capability to generate a functional immune response. In the case of the mRNA-1893 trial, the vaccine was developed with antigens that were present in the strain of virus circulating in 2016, but we could easily match whatever strain reemerges,” said Dr. Leav.
A phase 2 trial to confirm the dose of mRNA-1893 in a larger study population is underway.
Although it’s been demonstrated that Moderna’s mRNA vaccine is safe and effective, moving from a phase 2 to a phase 3 study presents a challenge, given the fact that currently, the disease burden from Zika is low. If an outbreak were to occur in the future, these mRNA vaccines could potentially be given emergency approval, as occurred during the COVID pandemic, according to Dr. Silverman.
If approved, provisionally or through a traditional route, the vaccine would “accelerate the ability to tamp down any further outbreaks, because vaccine-based immunity could be made available to a large portion of the population who were pregnant or planning a pregnancy, not just in the U.S. but also in these endemic areas,” said Dr. Silverman.
Takeaways from the last Zika outbreak
Practical steps such as mosquito eradication and development of vaccines are not the only takeaway from the recent Zika epidemics inside and outside the United States. A clearer picture of the short- and long-term stakes of the disease has emerged.
According to the CDC, most people who become infected with Zika experience only mild symptoms, such as fever, rash, headache, and muscle pain, but babies conceived by mothers infected with Zika are at risk for stillbirth, miscarriage, and microcephaly and other brain defects.
Although a pregnant woman who tests positive for Zika is in a very high-risk situation, “data show that only about 30% of mothers with Zika have a baby with birth defects. If a pregnant woman contracted Zika, what would happen is we would just do very close screening by ultrasound of the fetus. If microcephaly in utero or fetal brain defects were observed, then a mother would be counseled on her options,” said Dr. Gandhi.
Dr. Silverman noted that “new data on children who were exposed in utero and had normal exams, including head measurements when they were born, have raised concerns. In recently published long-term follow-up studies, even when children born to mothers infected with Zika during pregnancy had normal head growth at least 3 years after birth, they were still at risk for neurodevelopmental delay and behavioral disorders, including impact on coordination and executive function.
“This is another good reason to keep the potential risks of Zika active in the public’s consciousness and in public health planning.”
Dr. Silverman, Dr. Gandhi, and Dr. Staples have disclosed no relevant financial relationships. Dr. Leav is an employee of Moderna and owns stock in the company.
A version of this article originally appeared on Medscape.com.
Warming U.S. temperatures, the resumption of travel, and new knowledge about Zika’s long-term effects on children signal that Zika prevention and vaccine development should be on public health officials’, doctors’, and communities’ radar, even when community infection is not occurring.
“Although we haven’t seen confirmed Zika virus circulation in the continental United States or its territories for several years, it’s still something that we are closely monitoring, particularly as we move into the summer months,” Erin Staples, MD, PhD, medical epidemiologist at the Arboviral Diseases Branch of the Centers for Disease Control and Prevention in Fort Collins, Colo., told this news organization.
“This is because cases are still being reported in other countries, particularly in South America. Travel to these places is increasing following the pandemic, leaving more potential for individuals who might have acquired the infection to come back and restart community transmission.”
How Zika might reemerge
The Aedes aegypti mosquito is the vector by which Zika spreads, and “during the COVID pandemic, these mosquitoes moved further north in the United States, into southern California, and were identified as far north as Washington, D.C.,” said Neil Silverman, MD, professor of clinical obstetrics and gynecology and director of the Infections in Pregnancy Program at UCLA Medical Center in Los Angeles.
“On a population level, Americans have essentially no immunity to Zika from prior infection, and there is no vaccine yet approved. If individuals infected with Zika came into a U.S. region where the Aedes aegypti mosquito was present, that population could be very susceptible to infection spread and even another outbreak. This would be a confluence of bad circumstances, but that’s exactly what infectious disease specialists continue to be watchful about, especially because Zika is so dangerous for fetuses,” said Dr. Silverman.
How the public can prepare
The CDC recommends that pregnant women or women who plan to become pregnant avoid traveling to regions where there are currently outbreaks of Zika, but this is not the only way that individuals can protect themselves.
“The message we want to deliver to people is that in the United States, people are at risk for several mosquito-borne diseases every summer beyond just Zika,” Dr. Staples said. “It’s really important that people are instructed to make a habit of wearing EPA [U.S. Environmental Protection Agency)–registered insect repellents when they go outside. Right now, that is the single best tool that we have to prevent mosquito-borne diseases in the U.S.
“From a community standpoint, there are several emerging mosquito control methods that are being evaluated right now, such as genetic modification and irradiation of mosquitoes. These methods are aimed at producing sterile mosquitoes that are released into the wild to mate with the local mosquito population, which will render them infertile. This leads, over time, to suppression of the overall Aedes aegypti mosquito population – the main vector of Zika transmission,” said Dr. Staples.
Monica Gandhi, MD, MPH, professor of medicine and associate chief of the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco, encourages her patients to wear mosquito repellent but cautioned that “there’s no antiviral that you can take for Zika. Until we have a vaccine, the key to controlling/preventing Zika is controlling the mosquitoes that spread the virus.”
Vaccines
The National Institute of Allergy and Infectious Diseases (NIAID) is currently investigating a variety of Zika vaccines, including a DNA-based vaccine, (phase 2), a purified inactivated virus vaccine (phase 1), live attenuated vaccines (phase 2), and mRNA vaccines (phase 2).
“I’m most excited about mRNA vaccines because they help patients produce a lot of proteins. The protein from a typical protein-adjuvant vaccine will break down, and patients can only raise an immune response to whatever proteins are left. On the other hand, mRNA vaccines provide the body [with] a recipe to make the protein from the pathogen in high amounts, so that a strong immune response can be raised for protection,” noted Dr. Gandhi.
Moderna’s mRNA-1893 vaccine was recently studied in a randomized, observer-blind, controlled, phase 1 trial among 120 adults in the United States and Puerto Rico, the results of which were published online in The Lancet. “The vaccine was found to be generally well tolerated with no serious adverse events considered related to vaccine. Furthermore, the vaccine was able to generate a potent immune response that was capable of neutralizing the virus in vitro,” said Brett Leav, MD, executive director of clinical development for public health vaccines at Moderna.
“Our mRNA platform technology ... can be very helpful against emerging pandemic threats, as we saw in response to COVID-19. What is unique in our approach is that if the genetic sequence of the virus is known, we can quickly generate vaccines to test for their capability to generate a functional immune response. In the case of the mRNA-1893 trial, the vaccine was developed with antigens that were present in the strain of virus circulating in 2016, but we could easily match whatever strain reemerges,” said Dr. Leav.
A phase 2 trial to confirm the dose of mRNA-1893 in a larger study population is underway.
Although it’s been demonstrated that Moderna’s mRNA vaccine is safe and effective, moving from a phase 2 to a phase 3 study presents a challenge, given the fact that currently, the disease burden from Zika is low. If an outbreak were to occur in the future, these mRNA vaccines could potentially be given emergency approval, as occurred during the COVID pandemic, according to Dr. Silverman.
If approved, provisionally or through a traditional route, the vaccine would “accelerate the ability to tamp down any further outbreaks, because vaccine-based immunity could be made available to a large portion of the population who were pregnant or planning a pregnancy, not just in the U.S. but also in these endemic areas,” said Dr. Silverman.
Takeaways from the last Zika outbreak
Practical steps such as mosquito eradication and development of vaccines are not the only takeaway from the recent Zika epidemics inside and outside the United States. A clearer picture of the short- and long-term stakes of the disease has emerged.
According to the CDC, most people who become infected with Zika experience only mild symptoms, such as fever, rash, headache, and muscle pain, but babies conceived by mothers infected with Zika are at risk for stillbirth, miscarriage, and microcephaly and other brain defects.
Although a pregnant woman who tests positive for Zika is in a very high-risk situation, “data show that only about 30% of mothers with Zika have a baby with birth defects. If a pregnant woman contracted Zika, what would happen is we would just do very close screening by ultrasound of the fetus. If microcephaly in utero or fetal brain defects were observed, then a mother would be counseled on her options,” said Dr. Gandhi.
Dr. Silverman noted that “new data on children who were exposed in utero and had normal exams, including head measurements when they were born, have raised concerns. In recently published long-term follow-up studies, even when children born to mothers infected with Zika during pregnancy had normal head growth at least 3 years after birth, they were still at risk for neurodevelopmental delay and behavioral disorders, including impact on coordination and executive function.
“This is another good reason to keep the potential risks of Zika active in the public’s consciousness and in public health planning.”
Dr. Silverman, Dr. Gandhi, and Dr. Staples have disclosed no relevant financial relationships. Dr. Leav is an employee of Moderna and owns stock in the company.
A version of this article originally appeared on Medscape.com.
Psychosocial Burden of Hidradenitis Suppurativa
Hidradenitis suppurativa (HS) is a chronic inflammatory skin disease that typically presents as inflamed nodules, abscesses, and sinus tracts in the apocrine gland–bearing regions.
Chronic pain, fatigue, and social isolation put patients with HS at risk for psychiatric comorbidities, with the prevalence of depression estimated to be as high as 43%. Work productivity and sexual relationships may also be affected.
Early treatment of the condition, as well as screening for depression, may help alleviate the psychosocial burden of HS.
In this ReCAP, Dr Jennifer Hsiao from Keck School of Medicine, University of Southern California, Los Angeles, outlines mental health screening tools that dermatologists can use to help recognize at-risk patients and potentially refer them to mental health experts.
--
Jennifer Hsiao, MD, Associate Professor, Department of Dermatology, Keck School of Medicine, University of Southern California, Los Angeles, California
Jennifer Hsiao, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Boehringer Ingelheim; Novartis; UCB
Serve(d) as a speaker or a member of a speakers bureau for: AbbVie
Hidradenitis suppurativa (HS) is a chronic inflammatory skin disease that typically presents as inflamed nodules, abscesses, and sinus tracts in the apocrine gland–bearing regions.
Chronic pain, fatigue, and social isolation put patients with HS at risk for psychiatric comorbidities, with the prevalence of depression estimated to be as high as 43%. Work productivity and sexual relationships may also be affected.
Early treatment of the condition, as well as screening for depression, may help alleviate the psychosocial burden of HS.
In this ReCAP, Dr Jennifer Hsiao from Keck School of Medicine, University of Southern California, Los Angeles, outlines mental health screening tools that dermatologists can use to help recognize at-risk patients and potentially refer them to mental health experts.
--
Jennifer Hsiao, MD, Associate Professor, Department of Dermatology, Keck School of Medicine, University of Southern California, Los Angeles, California
Jennifer Hsiao, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Boehringer Ingelheim; Novartis; UCB
Serve(d) as a speaker or a member of a speakers bureau for: AbbVie
Hidradenitis suppurativa (HS) is a chronic inflammatory skin disease that typically presents as inflamed nodules, abscesses, and sinus tracts in the apocrine gland–bearing regions.
Chronic pain, fatigue, and social isolation put patients with HS at risk for psychiatric comorbidities, with the prevalence of depression estimated to be as high as 43%. Work productivity and sexual relationships may also be affected.
Early treatment of the condition, as well as screening for depression, may help alleviate the psychosocial burden of HS.
In this ReCAP, Dr Jennifer Hsiao from Keck School of Medicine, University of Southern California, Los Angeles, outlines mental health screening tools that dermatologists can use to help recognize at-risk patients and potentially refer them to mental health experts.
--
Jennifer Hsiao, MD, Associate Professor, Department of Dermatology, Keck School of Medicine, University of Southern California, Los Angeles, California
Jennifer Hsiao, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Boehringer Ingelheim; Novartis; UCB
Serve(d) as a speaker or a member of a speakers bureau for: AbbVie

New influx of Humira biosimilars may not drive immediate change
Gastroenterologists in 2023 will have more tools in their arsenal to treat patients with Crohn’s disease or ulcerative colitis. As many as 8-10 adalimumab biosimilars are anticipated to come on the market this year, giving mainstay drug Humira some vigorous competition.
Three scenarios will drive adalimumab biosimilar initiation: Insurance preference for the initial treatment of a newly diagnosed condition, a change in a patient’s insurance plan, or an insurance-mandated switch, said Edward C. Oldfield IV, MD, assistant professor at Eastern Virginia Medical School’s division of gastroenterology in Norfolk.
“Outside of these scenarios, I would encourage patients to remain on their current biologic so long as cost and accessibility remain stable,” said Dr. Oldfield.
Many factors will contribute to the success of biosimilars. Will physicians be prescribing them? How are biosimilars placed on formularies and will they be given preferred status? How will manufacturers price their biosimilars? “We have to wait and see to get the answers to these questions,” said Steven Newmark, JD, MPA, chief legal officer and director of policy, Global Healthy Living Foundation/CreakyJoints, a nonprofit advocacy organization based in New York.
Prescribing biosimilars is no different than prescribing originator biologics, so providers should know how to use them, said Mr. Newmark. “Most important will be the availability of patient-friendly resources that providers can share with their patients to provide education about and confidence in using biosimilars,” he added.
Overall, biosimilars are a good thing, said Dr. Oldfield. “In the long run they should bring down costs and increase access to medications for our patients.”
Others are skeptical that the adalimumab biosimilars will save patients much money.
Biosimilar laws were created to lower costs. However, if a patient with insurance pays only $5 a month out of pocket for Humira – a drug that normally costs $7,000 without coverage – it’s unlikely they would want to switch unless there’s comparable savings from the biosimilar, said Stephen B. Hanauer, MD, medical director of the Digestive Health Center and professor of medicine at Northwestern Medicine, Northwestern University, Evanston, Ill.
Like generics, Humira biosimilars may face some initial backlash, said Dr. Hanauer.
2023 broadens scope of adalimumab treatments
The American Gastroenterological Association describes a biosimilar as something that’s “highly similar to, but not an exact copy of, a biologic reference product already approved” by the Food and Drug Administration. Congress under the 2010 Affordable Care Act created a special, abbreviated pathway to approval for biosimilars.
AbbVie’s Humira, the global revenue for which exceeded $20 billion in 2021, has long dominated the U.S. market on injectable treatments for autoimmune diseases. The popular drug faces some competition in 2023, however, following a series of legal settlements that allowed AbbVie competitors to release their own adalimumab biosimilars.
“So far, we haven’t seen biosimilars live up to their potential in the U.S. in the inflammatory space,” said Mr. Newmark. This may change, however. Previously, biosimilars have required infusion, which demanded more time, commitment, and travel from patients. “The new set of forthcoming Humira biosimilars are injectables, an administration method preferred by patients,” he said.
The FDA will approve a biosimilar if it determines that the biological product is highly similar to the reference product, and that there are no clinically meaningful differences between the biological and reference product in terms of the safety, purity, and potency of the product.
The agency to date has approved 8 adalimumab biosimilars. These include: Idacio (adalimumab-aacf, Fresenius Kabi); Amjevita (adalimumab-atto, Amgen); Hadlima (adalimumab-bwwd, Organon); Cyltezo (adalimumab-adbm, Boehringer Ingelheim); Yusimry (adalimumab-aqvh from Coherus BioSciences); Hulio (adalimumab-fkjp; Mylan/Fujifilm Kyowa Kirin Biologics); Hyrimoz (adalimumab-adaz, Sandoz), and Abrilada (adalimumab-afzb, Pfizer).
“While FDA doesn’t formally track when products come to market, we know based on published reports that application holders for many of the currently FDA-approved biosimilars plan to market this year, starting with Amjevita being the first adalimumab biosimilar launched” in January, said Sarah Yim, MD, director of the Office of Therapeutic Biologics and Biosimilars at the agency.
At press time, two other companies (Celltrion and Alvotech/Teva) were awaiting FDA approval for their adalimumab biosimilar drugs.
Among the eight approved drugs, Cyltezo is the only one that has a designation for interchangeability with Humira.
An interchangeable biosimilar may be substituted at the pharmacy without the intervention of the prescriber – much like generics are substituted, depending on state laws, said Dr. Yim. “However, in terms of safety and effectiveness, FDA’s standards for approval mean that biosimilar or interchangeable biosimilar products can be used in place of the reference product they were compared to.”
FDA-approved biosimilars undergo a rigorous evaluation for safety, effectiveness, and quality for their approved conditions of use, she continued. “Therefore, patients and health care providers can rely on a biosimilar to be as safe and effective for its approved uses as the original biological product.”
Remicade as a yard stick
Gastroenterologists dealt with this situation once before, when Remicade (infliximab) biosimilars came on the market in 2016, noted Miguel Regueiro, MD, chair of the Digestive Disease and Surgery Institute at the Cleveland Clinic.
Remicade and Humira are both tumor necrosis factor inhibitors with the same mechanism of action and many of the same indications. “We already had that experience with Remicade and biosimilar switch 2 or 3 years ago. Now we’re talking about Humira,” said Dr. Regueiro.
Most GI doctors have prescribed one of the more common infliximab biosimilars (Inflectra or Renflexis), noted Dr. Oldfield.
Cardinal Health, which recently surveyed 300 gastroenterologists, rheumatologists, and dermatologists about adalimumab biosimilars, found that gastroenterologists had the highest comfort level in prescribing them. Their top concern, however, was changing a patient from adalimumab to an adalimumab biosimilar.
For most patients, Dr. Oldfield sees the Humira reference biologic and biosimilar as equivalent.
However, he said he would change a patient’s drug only if there were a good reason or if his hand was forced by insurance. He would not make the change for a patient who recently began induction with the reference biologic or a patient with highly active clinical disease.
“While there is limited data to support this, I would also have some qualms about changing a patient from reference biologic to a biosimilar if they previously had immune-mediated pharmacokinetic failure due to antibody development with a biologic and were currently doing well on their new biologic,” he said.
Those with a new ulcerative colitis or Crohn’s diagnosis who are initiating a biologic for the first time might consider a biosimilar. If a patient is transitioning from a reference biologic to a biosimilar, “I would want to make that change during a time of stable remission and with the recognition that the switch is not a temporary switch, but a long-term switch,” he continued.
A paper that reviewed 23 observational studies of adalimumab and other biosimilars found that switching biosimilars was safe and effective. But if possible, patients should minimize the number of switches until more robust long-term data are available, added Dr. Oldfield.
If a patient is apprehensive about switching to a new therapy, “one may need to be cognizant of the ‘nocebo’ effect in which there is an unexplained or unfavorable therapeutic effect after switching,” he said.
Other gastroenterologists voiced similar reservations about switching. “I won’t use an adalimumab biosimilar unless the patient requests it, the insurance requires it, or there is a cost advantage for the patient such that they prefer it,” said Doug Wolf, MD, an Atlanta gastroenterologist.
“There is no medical treatment advantage to a biosimilar, especially if switching from Humira,” added Dr. Wolf.
Insurance will guide treatment
Once a drug is approved for use by the FDA, that drug will be available in all 50 states. “Different private insurance formularies, as well as state Medicaid formularies, might affect the actual ability of patients to receive such drugs,” said Mr. Newmark.
Patients should consult with their providers and insurance companies to see what therapies are available, he advised.
Dr. Hanauer anticipates some headaches arising for patients and doctors alike when negotiating for a specific drug.
Cyltezo may be the only biosimilar interchangeable with Humira, but the third-party pharmacy benefit manager (PBM) could negotiate for one of the noninterchangeable ones. “On a yearly basis they could switch their preference,” said Dr. Hanauer.
In the Cardinal Health survey, more than 60% of respondents said they would feel comfortable prescribing an adalimumab biosimilar only with an interchangeability designation.
A PBM may offer a patient Cyltezo if it’s cheaper than Humira. If the patient insists on staying on Humira, then they’ll have to pay more for that drug on their payer’s formulary, said Dr. Hanauer. In a worst-case scenario, a physician may have to appeal on a patient’s behalf to get Humira if the insurer offers only the biosimilar.
Taking that step to appeal is a major hassle for the physician, and leads to extra back door costs as well, said Dr. Hanauer.
Humira manufacturer AbbVie, in turn, may offer discounts and rebates to the PBMs to put Humira on their formulary. “That’s the AbbVie negotiating power. It’s not that the cost is going to be that much different. It’s going to be that there are rebates and discounts that are going to make the cost different,” he added.
As a community physician, Dr. Oldfield has specific concerns about accessibility.
The ever-increasing burden of insurance documentation and prior authorization means it can take weeks or months to get these medications approved. “The addition of new biosimilars is a welcome entrance if it can get patients the medications they need when they need it,” he said.
When it comes to prescribing biologics, many physicians rely on ancillary staff for assistance. It’s a team effort to sift through all the paperwork, observed Dr. Oldfield.
“While many community GI practices have specialized staff to deal with prior authorizations, they are still a far cry from the IBD [inflammatory bowel disease] academic centers where there are often pharmacists, nursing specialists, and home-monitoring programs to check in on patients,” he explained.
Landscape on cost is uncertain
At present, little is known about the cost of the biosimilars and impact on future drug pricing, said Dr. Oldfield.
At least for Medicare, Humira biosimilars will be considered Medicare Part D drugs if used for a medically accepted indication, said a spokesperson for the Centers for Medicare and Medicaid Services.
Part D sponsors (pharmacy and therapeutic committees) “will make the determination as to whether Amjevita and other products will be added to their formularies,” said the spokesperson.
Patients never saw a significant cost savings with Remicade biosimilars. “I imagine the same would be true with biosimilars for Humira,” said Dr. Regueiro. Patients may see greater access to these drugs, however, because the insurance plan or the pharmacy plan will make them more readily available, he added.
The hope is that, as biosimilars are introduced, the price of the originator biologic will go down, said Mr. Newmark. “Therefore, we can expect Humira to be offered at a lower price as it faces competition. Where it will sit in comparison to the forthcoming biosimilars will depend on how much biosimilar companies drop their price and how much pressure will be on PBMs and insurers to cover the lowest list price drug,” he said.
AbbVie did not respond to several requests for comment.
Charitable patient assistance programs for biosimilars or biologics can help offset the price of copayments, Mr. Newmark offered.
Ideally, insurers will offer designated biosimilars at a reduced or even no out-of-pocket expense on their formularies. This should lead to a decreased administrative burden for approval with streamlined (or even removal) of prior authorizations for certain medications, said Dr. Oldfield.
Without insurance or medication assistance programs, the cost of biosimilars is prohibitively expensive, he added.
“Biosimilars have higher research, development, and manufacturing costs than what people conventionally think of [for] a generic medication.”
Educating, advising patients
Dr. Oldfield advised that gastroenterologists refer to biologics by the generic name rather than branded name when initiating therapy unless there is a very specific reason not to. “This approach should make the process more streamlined and less subjected to quick denials for brand-only requests as biosimilars start to assume a larger market share,” he said.
Uptake of the Humira biosimilars also will depend on proper education of physicians and patients and their comfort level with the biosimilars, said Dr. Regueiro. Cleveland Clinic uses a team approach to educate on this topic, relying on pharmacists, clinicians, and nurses to explain that there’s no real difference between the reference drug and its biosimilars, based on efficacy and safety data.
Physicians can also direct patients to patient-friendly resources, said Mr. Newmark. “By starting the conversation early, it ensures that when/if the time comes that your patient is switched to or chooses a biosimilar they will feel more confident because they have the knowledge to make decisions about their care.”
The Global Healthy Living Foundation’s podcast, Breaking Down Biosimilars , is a free resource for patients, he added.
It’s important that doctors also understand these products so they can explain to their patients what to expect, said the FDA’s Dr. Yim. The FDA provides educational materials on its website, including a comprehensive curriculum toolkit.
Dr. Hanauer has served as a consultant for AbbVie, Amgen, American College of Gastroenterology, GlaxoSmithKline, American Gastroenterological Association, Pfizer, and a host of other companies . Dr. Regueiro has served on advisory boards and as a consultant for Abbvie, Janssen, UCB, Takeda, Pfizer, BMS, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, TARGET Pharma Solutions,Trellis, and Boehringer Ingelheim Pharmaceuticals. Dr. Wolf, Dr. Yim, Dr. Oldfield, and Mr. Newmark have no financial conflicts of interest.
Gastroenterologists in 2023 will have more tools in their arsenal to treat patients with Crohn’s disease or ulcerative colitis. As many as 8-10 adalimumab biosimilars are anticipated to come on the market this year, giving mainstay drug Humira some vigorous competition.
Three scenarios will drive adalimumab biosimilar initiation: Insurance preference for the initial treatment of a newly diagnosed condition, a change in a patient’s insurance plan, or an insurance-mandated switch, said Edward C. Oldfield IV, MD, assistant professor at Eastern Virginia Medical School’s division of gastroenterology in Norfolk.
“Outside of these scenarios, I would encourage patients to remain on their current biologic so long as cost and accessibility remain stable,” said Dr. Oldfield.
Many factors will contribute to the success of biosimilars. Will physicians be prescribing them? How are biosimilars placed on formularies and will they be given preferred status? How will manufacturers price their biosimilars? “We have to wait and see to get the answers to these questions,” said Steven Newmark, JD, MPA, chief legal officer and director of policy, Global Healthy Living Foundation/CreakyJoints, a nonprofit advocacy organization based in New York.
Prescribing biosimilars is no different than prescribing originator biologics, so providers should know how to use them, said Mr. Newmark. “Most important will be the availability of patient-friendly resources that providers can share with their patients to provide education about and confidence in using biosimilars,” he added.
Overall, biosimilars are a good thing, said Dr. Oldfield. “In the long run they should bring down costs and increase access to medications for our patients.”
Others are skeptical that the adalimumab biosimilars will save patients much money.
Biosimilar laws were created to lower costs. However, if a patient with insurance pays only $5 a month out of pocket for Humira – a drug that normally costs $7,000 without coverage – it’s unlikely they would want to switch unless there’s comparable savings from the biosimilar, said Stephen B. Hanauer, MD, medical director of the Digestive Health Center and professor of medicine at Northwestern Medicine, Northwestern University, Evanston, Ill.
Like generics, Humira biosimilars may face some initial backlash, said Dr. Hanauer.
2023 broadens scope of adalimumab treatments
The American Gastroenterological Association describes a biosimilar as something that’s “highly similar to, but not an exact copy of, a biologic reference product already approved” by the Food and Drug Administration. Congress under the 2010 Affordable Care Act created a special, abbreviated pathway to approval for biosimilars.
AbbVie’s Humira, the global revenue for which exceeded $20 billion in 2021, has long dominated the U.S. market on injectable treatments for autoimmune diseases. The popular drug faces some competition in 2023, however, following a series of legal settlements that allowed AbbVie competitors to release their own adalimumab biosimilars.
“So far, we haven’t seen biosimilars live up to their potential in the U.S. in the inflammatory space,” said Mr. Newmark. This may change, however. Previously, biosimilars have required infusion, which demanded more time, commitment, and travel from patients. “The new set of forthcoming Humira biosimilars are injectables, an administration method preferred by patients,” he said.
The FDA will approve a biosimilar if it determines that the biological product is highly similar to the reference product, and that there are no clinically meaningful differences between the biological and reference product in terms of the safety, purity, and potency of the product.
The agency to date has approved 8 adalimumab biosimilars. These include: Idacio (adalimumab-aacf, Fresenius Kabi); Amjevita (adalimumab-atto, Amgen); Hadlima (adalimumab-bwwd, Organon); Cyltezo (adalimumab-adbm, Boehringer Ingelheim); Yusimry (adalimumab-aqvh from Coherus BioSciences); Hulio (adalimumab-fkjp; Mylan/Fujifilm Kyowa Kirin Biologics); Hyrimoz (adalimumab-adaz, Sandoz), and Abrilada (adalimumab-afzb, Pfizer).
“While FDA doesn’t formally track when products come to market, we know based on published reports that application holders for many of the currently FDA-approved biosimilars plan to market this year, starting with Amjevita being the first adalimumab biosimilar launched” in January, said Sarah Yim, MD, director of the Office of Therapeutic Biologics and Biosimilars at the agency.
At press time, two other companies (Celltrion and Alvotech/Teva) were awaiting FDA approval for their adalimumab biosimilar drugs.
Among the eight approved drugs, Cyltezo is the only one that has a designation for interchangeability with Humira.
An interchangeable biosimilar may be substituted at the pharmacy without the intervention of the prescriber – much like generics are substituted, depending on state laws, said Dr. Yim. “However, in terms of safety and effectiveness, FDA’s standards for approval mean that biosimilar or interchangeable biosimilar products can be used in place of the reference product they were compared to.”
FDA-approved biosimilars undergo a rigorous evaluation for safety, effectiveness, and quality for their approved conditions of use, she continued. “Therefore, patients and health care providers can rely on a biosimilar to be as safe and effective for its approved uses as the original biological product.”
Remicade as a yard stick
Gastroenterologists dealt with this situation once before, when Remicade (infliximab) biosimilars came on the market in 2016, noted Miguel Regueiro, MD, chair of the Digestive Disease and Surgery Institute at the Cleveland Clinic.
Remicade and Humira are both tumor necrosis factor inhibitors with the same mechanism of action and many of the same indications. “We already had that experience with Remicade and biosimilar switch 2 or 3 years ago. Now we’re talking about Humira,” said Dr. Regueiro.
Most GI doctors have prescribed one of the more common infliximab biosimilars (Inflectra or Renflexis), noted Dr. Oldfield.
Cardinal Health, which recently surveyed 300 gastroenterologists, rheumatologists, and dermatologists about adalimumab biosimilars, found that gastroenterologists had the highest comfort level in prescribing them. Their top concern, however, was changing a patient from adalimumab to an adalimumab biosimilar.
For most patients, Dr. Oldfield sees the Humira reference biologic and biosimilar as equivalent.
However, he said he would change a patient’s drug only if there were a good reason or if his hand was forced by insurance. He would not make the change for a patient who recently began induction with the reference biologic or a patient with highly active clinical disease.
“While there is limited data to support this, I would also have some qualms about changing a patient from reference biologic to a biosimilar if they previously had immune-mediated pharmacokinetic failure due to antibody development with a biologic and were currently doing well on their new biologic,” he said.
Those with a new ulcerative colitis or Crohn’s diagnosis who are initiating a biologic for the first time might consider a biosimilar. If a patient is transitioning from a reference biologic to a biosimilar, “I would want to make that change during a time of stable remission and with the recognition that the switch is not a temporary switch, but a long-term switch,” he continued.
A paper that reviewed 23 observational studies of adalimumab and other biosimilars found that switching biosimilars was safe and effective. But if possible, patients should minimize the number of switches until more robust long-term data are available, added Dr. Oldfield.
If a patient is apprehensive about switching to a new therapy, “one may need to be cognizant of the ‘nocebo’ effect in which there is an unexplained or unfavorable therapeutic effect after switching,” he said.
Other gastroenterologists voiced similar reservations about switching. “I won’t use an adalimumab biosimilar unless the patient requests it, the insurance requires it, or there is a cost advantage for the patient such that they prefer it,” said Doug Wolf, MD, an Atlanta gastroenterologist.
“There is no medical treatment advantage to a biosimilar, especially if switching from Humira,” added Dr. Wolf.
Insurance will guide treatment
Once a drug is approved for use by the FDA, that drug will be available in all 50 states. “Different private insurance formularies, as well as state Medicaid formularies, might affect the actual ability of patients to receive such drugs,” said Mr. Newmark.
Patients should consult with their providers and insurance companies to see what therapies are available, he advised.
Dr. Hanauer anticipates some headaches arising for patients and doctors alike when negotiating for a specific drug.
Cyltezo may be the only biosimilar interchangeable with Humira, but the third-party pharmacy benefit manager (PBM) could negotiate for one of the noninterchangeable ones. “On a yearly basis they could switch their preference,” said Dr. Hanauer.
In the Cardinal Health survey, more than 60% of respondents said they would feel comfortable prescribing an adalimumab biosimilar only with an interchangeability designation.
A PBM may offer a patient Cyltezo if it’s cheaper than Humira. If the patient insists on staying on Humira, then they’ll have to pay more for that drug on their payer’s formulary, said Dr. Hanauer. In a worst-case scenario, a physician may have to appeal on a patient’s behalf to get Humira if the insurer offers only the biosimilar.
Taking that step to appeal is a major hassle for the physician, and leads to extra back door costs as well, said Dr. Hanauer.
Humira manufacturer AbbVie, in turn, may offer discounts and rebates to the PBMs to put Humira on their formulary. “That’s the AbbVie negotiating power. It’s not that the cost is going to be that much different. It’s going to be that there are rebates and discounts that are going to make the cost different,” he added.
As a community physician, Dr. Oldfield has specific concerns about accessibility.
The ever-increasing burden of insurance documentation and prior authorization means it can take weeks or months to get these medications approved. “The addition of new biosimilars is a welcome entrance if it can get patients the medications they need when they need it,” he said.
When it comes to prescribing biologics, many physicians rely on ancillary staff for assistance. It’s a team effort to sift through all the paperwork, observed Dr. Oldfield.
“While many community GI practices have specialized staff to deal with prior authorizations, they are still a far cry from the IBD [inflammatory bowel disease] academic centers where there are often pharmacists, nursing specialists, and home-monitoring programs to check in on patients,” he explained.
Landscape on cost is uncertain
At present, little is known about the cost of the biosimilars and impact on future drug pricing, said Dr. Oldfield.
At least for Medicare, Humira biosimilars will be considered Medicare Part D drugs if used for a medically accepted indication, said a spokesperson for the Centers for Medicare and Medicaid Services.
Part D sponsors (pharmacy and therapeutic committees) “will make the determination as to whether Amjevita and other products will be added to their formularies,” said the spokesperson.
Patients never saw a significant cost savings with Remicade biosimilars. “I imagine the same would be true with biosimilars for Humira,” said Dr. Regueiro. Patients may see greater access to these drugs, however, because the insurance plan or the pharmacy plan will make them more readily available, he added.
The hope is that, as biosimilars are introduced, the price of the originator biologic will go down, said Mr. Newmark. “Therefore, we can expect Humira to be offered at a lower price as it faces competition. Where it will sit in comparison to the forthcoming biosimilars will depend on how much biosimilar companies drop their price and how much pressure will be on PBMs and insurers to cover the lowest list price drug,” he said.
AbbVie did not respond to several requests for comment.
Charitable patient assistance programs for biosimilars or biologics can help offset the price of copayments, Mr. Newmark offered.
Ideally, insurers will offer designated biosimilars at a reduced or even no out-of-pocket expense on their formularies. This should lead to a decreased administrative burden for approval with streamlined (or even removal) of prior authorizations for certain medications, said Dr. Oldfield.
Without insurance or medication assistance programs, the cost of biosimilars is prohibitively expensive, he added.
“Biosimilars have higher research, development, and manufacturing costs than what people conventionally think of [for] a generic medication.”
Educating, advising patients
Dr. Oldfield advised that gastroenterologists refer to biologics by the generic name rather than branded name when initiating therapy unless there is a very specific reason not to. “This approach should make the process more streamlined and less subjected to quick denials for brand-only requests as biosimilars start to assume a larger market share,” he said.
Uptake of the Humira biosimilars also will depend on proper education of physicians and patients and their comfort level with the biosimilars, said Dr. Regueiro. Cleveland Clinic uses a team approach to educate on this topic, relying on pharmacists, clinicians, and nurses to explain that there’s no real difference between the reference drug and its biosimilars, based on efficacy and safety data.
Physicians can also direct patients to patient-friendly resources, said Mr. Newmark. “By starting the conversation early, it ensures that when/if the time comes that your patient is switched to or chooses a biosimilar they will feel more confident because they have the knowledge to make decisions about their care.”
The Global Healthy Living Foundation’s podcast, Breaking Down Biosimilars , is a free resource for patients, he added.
It’s important that doctors also understand these products so they can explain to their patients what to expect, said the FDA’s Dr. Yim. The FDA provides educational materials on its website, including a comprehensive curriculum toolkit.
Dr. Hanauer has served as a consultant for AbbVie, Amgen, American College of Gastroenterology, GlaxoSmithKline, American Gastroenterological Association, Pfizer, and a host of other companies . Dr. Regueiro has served on advisory boards and as a consultant for Abbvie, Janssen, UCB, Takeda, Pfizer, BMS, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, TARGET Pharma Solutions,Trellis, and Boehringer Ingelheim Pharmaceuticals. Dr. Wolf, Dr. Yim, Dr. Oldfield, and Mr. Newmark have no financial conflicts of interest.
Gastroenterologists in 2023 will have more tools in their arsenal to treat patients with Crohn’s disease or ulcerative colitis. As many as 8-10 adalimumab biosimilars are anticipated to come on the market this year, giving mainstay drug Humira some vigorous competition.
Three scenarios will drive adalimumab biosimilar initiation: Insurance preference for the initial treatment of a newly diagnosed condition, a change in a patient’s insurance plan, or an insurance-mandated switch, said Edward C. Oldfield IV, MD, assistant professor at Eastern Virginia Medical School’s division of gastroenterology in Norfolk.
“Outside of these scenarios, I would encourage patients to remain on their current biologic so long as cost and accessibility remain stable,” said Dr. Oldfield.
Many factors will contribute to the success of biosimilars. Will physicians be prescribing them? How are biosimilars placed on formularies and will they be given preferred status? How will manufacturers price their biosimilars? “We have to wait and see to get the answers to these questions,” said Steven Newmark, JD, MPA, chief legal officer and director of policy, Global Healthy Living Foundation/CreakyJoints, a nonprofit advocacy organization based in New York.
Prescribing biosimilars is no different than prescribing originator biologics, so providers should know how to use them, said Mr. Newmark. “Most important will be the availability of patient-friendly resources that providers can share with their patients to provide education about and confidence in using biosimilars,” he added.
Overall, biosimilars are a good thing, said Dr. Oldfield. “In the long run they should bring down costs and increase access to medications for our patients.”
Others are skeptical that the adalimumab biosimilars will save patients much money.
Biosimilar laws were created to lower costs. However, if a patient with insurance pays only $5 a month out of pocket for Humira – a drug that normally costs $7,000 without coverage – it’s unlikely they would want to switch unless there’s comparable savings from the biosimilar, said Stephen B. Hanauer, MD, medical director of the Digestive Health Center and professor of medicine at Northwestern Medicine, Northwestern University, Evanston, Ill.
Like generics, Humira biosimilars may face some initial backlash, said Dr. Hanauer.
2023 broadens scope of adalimumab treatments
The American Gastroenterological Association describes a biosimilar as something that’s “highly similar to, but not an exact copy of, a biologic reference product already approved” by the Food and Drug Administration. Congress under the 2010 Affordable Care Act created a special, abbreviated pathway to approval for biosimilars.
AbbVie’s Humira, the global revenue for which exceeded $20 billion in 2021, has long dominated the U.S. market on injectable treatments for autoimmune diseases. The popular drug faces some competition in 2023, however, following a series of legal settlements that allowed AbbVie competitors to release their own adalimumab biosimilars.
“So far, we haven’t seen biosimilars live up to their potential in the U.S. in the inflammatory space,” said Mr. Newmark. This may change, however. Previously, biosimilars have required infusion, which demanded more time, commitment, and travel from patients. “The new set of forthcoming Humira biosimilars are injectables, an administration method preferred by patients,” he said.
The FDA will approve a biosimilar if it determines that the biological product is highly similar to the reference product, and that there are no clinically meaningful differences between the biological and reference product in terms of the safety, purity, and potency of the product.
The agency to date has approved 8 adalimumab biosimilars. These include: Idacio (adalimumab-aacf, Fresenius Kabi); Amjevita (adalimumab-atto, Amgen); Hadlima (adalimumab-bwwd, Organon); Cyltezo (adalimumab-adbm, Boehringer Ingelheim); Yusimry (adalimumab-aqvh from Coherus BioSciences); Hulio (adalimumab-fkjp; Mylan/Fujifilm Kyowa Kirin Biologics); Hyrimoz (adalimumab-adaz, Sandoz), and Abrilada (adalimumab-afzb, Pfizer).
“While FDA doesn’t formally track when products come to market, we know based on published reports that application holders for many of the currently FDA-approved biosimilars plan to market this year, starting with Amjevita being the first adalimumab biosimilar launched” in January, said Sarah Yim, MD, director of the Office of Therapeutic Biologics and Biosimilars at the agency.
At press time, two other companies (Celltrion and Alvotech/Teva) were awaiting FDA approval for their adalimumab biosimilar drugs.
Among the eight approved drugs, Cyltezo is the only one that has a designation for interchangeability with Humira.
An interchangeable biosimilar may be substituted at the pharmacy without the intervention of the prescriber – much like generics are substituted, depending on state laws, said Dr. Yim. “However, in terms of safety and effectiveness, FDA’s standards for approval mean that biosimilar or interchangeable biosimilar products can be used in place of the reference product they were compared to.”
FDA-approved biosimilars undergo a rigorous evaluation for safety, effectiveness, and quality for their approved conditions of use, she continued. “Therefore, patients and health care providers can rely on a biosimilar to be as safe and effective for its approved uses as the original biological product.”
Remicade as a yard stick
Gastroenterologists dealt with this situation once before, when Remicade (infliximab) biosimilars came on the market in 2016, noted Miguel Regueiro, MD, chair of the Digestive Disease and Surgery Institute at the Cleveland Clinic.
Remicade and Humira are both tumor necrosis factor inhibitors with the same mechanism of action and many of the same indications. “We already had that experience with Remicade and biosimilar switch 2 or 3 years ago. Now we’re talking about Humira,” said Dr. Regueiro.
Most GI doctors have prescribed one of the more common infliximab biosimilars (Inflectra or Renflexis), noted Dr. Oldfield.
Cardinal Health, which recently surveyed 300 gastroenterologists, rheumatologists, and dermatologists about adalimumab biosimilars, found that gastroenterologists had the highest comfort level in prescribing them. Their top concern, however, was changing a patient from adalimumab to an adalimumab biosimilar.
For most patients, Dr. Oldfield sees the Humira reference biologic and biosimilar as equivalent.
However, he said he would change a patient’s drug only if there were a good reason or if his hand was forced by insurance. He would not make the change for a patient who recently began induction with the reference biologic or a patient with highly active clinical disease.
“While there is limited data to support this, I would also have some qualms about changing a patient from reference biologic to a biosimilar if they previously had immune-mediated pharmacokinetic failure due to antibody development with a biologic and were currently doing well on their new biologic,” he said.
Those with a new ulcerative colitis or Crohn’s diagnosis who are initiating a biologic for the first time might consider a biosimilar. If a patient is transitioning from a reference biologic to a biosimilar, “I would want to make that change during a time of stable remission and with the recognition that the switch is not a temporary switch, but a long-term switch,” he continued.
A paper that reviewed 23 observational studies of adalimumab and other biosimilars found that switching biosimilars was safe and effective. But if possible, patients should minimize the number of switches until more robust long-term data are available, added Dr. Oldfield.
If a patient is apprehensive about switching to a new therapy, “one may need to be cognizant of the ‘nocebo’ effect in which there is an unexplained or unfavorable therapeutic effect after switching,” he said.
Other gastroenterologists voiced similar reservations about switching. “I won’t use an adalimumab biosimilar unless the patient requests it, the insurance requires it, or there is a cost advantage for the patient such that they prefer it,” said Doug Wolf, MD, an Atlanta gastroenterologist.
“There is no medical treatment advantage to a biosimilar, especially if switching from Humira,” added Dr. Wolf.
Insurance will guide treatment
Once a drug is approved for use by the FDA, that drug will be available in all 50 states. “Different private insurance formularies, as well as state Medicaid formularies, might affect the actual ability of patients to receive such drugs,” said Mr. Newmark.
Patients should consult with their providers and insurance companies to see what therapies are available, he advised.
Dr. Hanauer anticipates some headaches arising for patients and doctors alike when negotiating for a specific drug.
Cyltezo may be the only biosimilar interchangeable with Humira, but the third-party pharmacy benefit manager (PBM) could negotiate for one of the noninterchangeable ones. “On a yearly basis they could switch their preference,” said Dr. Hanauer.
In the Cardinal Health survey, more than 60% of respondents said they would feel comfortable prescribing an adalimumab biosimilar only with an interchangeability designation.
A PBM may offer a patient Cyltezo if it’s cheaper than Humira. If the patient insists on staying on Humira, then they’ll have to pay more for that drug on their payer’s formulary, said Dr. Hanauer. In a worst-case scenario, a physician may have to appeal on a patient’s behalf to get Humira if the insurer offers only the biosimilar.
Taking that step to appeal is a major hassle for the physician, and leads to extra back door costs as well, said Dr. Hanauer.
Humira manufacturer AbbVie, in turn, may offer discounts and rebates to the PBMs to put Humira on their formulary. “That’s the AbbVie negotiating power. It’s not that the cost is going to be that much different. It’s going to be that there are rebates and discounts that are going to make the cost different,” he added.
As a community physician, Dr. Oldfield has specific concerns about accessibility.
The ever-increasing burden of insurance documentation and prior authorization means it can take weeks or months to get these medications approved. “The addition of new biosimilars is a welcome entrance if it can get patients the medications they need when they need it,” he said.
When it comes to prescribing biologics, many physicians rely on ancillary staff for assistance. It’s a team effort to sift through all the paperwork, observed Dr. Oldfield.
“While many community GI practices have specialized staff to deal with prior authorizations, they are still a far cry from the IBD [inflammatory bowel disease] academic centers where there are often pharmacists, nursing specialists, and home-monitoring programs to check in on patients,” he explained.
Landscape on cost is uncertain
At present, little is known about the cost of the biosimilars and impact on future drug pricing, said Dr. Oldfield.
At least for Medicare, Humira biosimilars will be considered Medicare Part D drugs if used for a medically accepted indication, said a spokesperson for the Centers for Medicare and Medicaid Services.
Part D sponsors (pharmacy and therapeutic committees) “will make the determination as to whether Amjevita and other products will be added to their formularies,” said the spokesperson.
Patients never saw a significant cost savings with Remicade biosimilars. “I imagine the same would be true with biosimilars for Humira,” said Dr. Regueiro. Patients may see greater access to these drugs, however, because the insurance plan or the pharmacy plan will make them more readily available, he added.
The hope is that, as biosimilars are introduced, the price of the originator biologic will go down, said Mr. Newmark. “Therefore, we can expect Humira to be offered at a lower price as it faces competition. Where it will sit in comparison to the forthcoming biosimilars will depend on how much biosimilar companies drop their price and how much pressure will be on PBMs and insurers to cover the lowest list price drug,” he said.
AbbVie did not respond to several requests for comment.
Charitable patient assistance programs for biosimilars or biologics can help offset the price of copayments, Mr. Newmark offered.
Ideally, insurers will offer designated biosimilars at a reduced or even no out-of-pocket expense on their formularies. This should lead to a decreased administrative burden for approval with streamlined (or even removal) of prior authorizations for certain medications, said Dr. Oldfield.
Without insurance or medication assistance programs, the cost of biosimilars is prohibitively expensive, he added.
“Biosimilars have higher research, development, and manufacturing costs than what people conventionally think of [for] a generic medication.”
Educating, advising patients
Dr. Oldfield advised that gastroenterologists refer to biologics by the generic name rather than branded name when initiating therapy unless there is a very specific reason not to. “This approach should make the process more streamlined and less subjected to quick denials for brand-only requests as biosimilars start to assume a larger market share,” he said.
Uptake of the Humira biosimilars also will depend on proper education of physicians and patients and their comfort level with the biosimilars, said Dr. Regueiro. Cleveland Clinic uses a team approach to educate on this topic, relying on pharmacists, clinicians, and nurses to explain that there’s no real difference between the reference drug and its biosimilars, based on efficacy and safety data.
Physicians can also direct patients to patient-friendly resources, said Mr. Newmark. “By starting the conversation early, it ensures that when/if the time comes that your patient is switched to or chooses a biosimilar they will feel more confident because they have the knowledge to make decisions about their care.”
The Global Healthy Living Foundation’s podcast, Breaking Down Biosimilars , is a free resource for patients, he added.
It’s important that doctors also understand these products so they can explain to their patients what to expect, said the FDA’s Dr. Yim. The FDA provides educational materials on its website, including a comprehensive curriculum toolkit.
Dr. Hanauer has served as a consultant for AbbVie, Amgen, American College of Gastroenterology, GlaxoSmithKline, American Gastroenterological Association, Pfizer, and a host of other companies . Dr. Regueiro has served on advisory boards and as a consultant for Abbvie, Janssen, UCB, Takeda, Pfizer, BMS, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, TARGET Pharma Solutions,Trellis, and Boehringer Ingelheim Pharmaceuticals. Dr. Wolf, Dr. Yim, Dr. Oldfield, and Mr. Newmark have no financial conflicts of interest.
What does the future of psoriasis treatment look like?
HONOLULU – During office visits with Andrew Blauvelt, MD, MBA, many patients well controlled on biologic therapy for their moderate to severe psoriasis often ask him when their scheduled injections can stop.
The most common question he hears is, “ ‘Why do I have to keep doing this? I’ve been clear for 2 or 3 years,’ ” Dr. Blauvelt, president of Oregon Medical Research Center, Portland, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “We have terrific drugs for psoriasis, but how can we do better?”
Development of oral biologics. At least two companies are developing a peptide-type small molecule that blocks interleukin (IL)-17 or IL-23 signaling, but would be given as a pill, he said. Another concept in the works is a robotic pill for drug delivery. The pill, which is being developed by Rani Therapeutics, protects the biotherapeutic drug payload from digestion in the GI tract and auto-injects it into the wall of the small intestine, according to a report of two studies that demonstrated the safety and tolerability of the robotic pill in healthy humans.
In an animal study, the same researchers showed that delivering monoclonal antibodies with the robotic pill achieved bioavailability on par with that obtained by standard subcutaneous injections.
Identifying “super responders” who require less frequent dosing of medication. “There’s data to suggest that we can kind of back off treatment in these patients,” Dr. Blauvelt said.
Hitting treatment hard and early. “There’s a concept in medicine of hitting disease hard and hitting it early, before the disease can establish itself and cause damage,” he said.
Targeting tissue resident memory T cells. In psoriasis, the idea is that if you treat earlier, when patients are just diagnosed, “perhaps you might be able to decrease resident memory T cells that set up shop in the skin and are responsible for disease recurrences,” Dr. Blauvelt said. “Research has shown that IL-23 blockers decrease tissue resident memory T cells, and IL-17 blockers don’t. This could explain why we see long remissions in this class of drug because we’re getting at these resident memory T cells and knocking them down,” he explained. “Our hypothesis is that hitting hard and early in the treatment course with high-dose IL-23 blockade may be an effective strategy to induce long-term remissions and possible cure, what we call ‘knock-out therapy.’ ”
In a pilot study of 20 patients, Dr. Blauvelt and colleagues are evaluating whether higher initial doses of the IL-23 antagonist risankizumab (300 mg and 600 mg, 2 times and 4 times the standard initial doses for plaque psoriasis) can more effectively target resident memory T cells. “This involves dosing at weeks 0, 4, and 16, then stopping and measuring resident T cells in the tissue to see how long we can induce psoriasis remissions,” Dr. Blauvelt said.
“I have no data to share, but I think we have the potential for unprecedented PASI-100 numbers with no added safety concerns, and the potential to break away from established regular dosing patterns,” such as the possibility of yearly dosing, the possibility of long-term remissions, and the possibility of cure in some patients, he noted.
Inducing tolerance. This refers to efforts aimed at increasing regulatory T cells, which are natural T cells that calm inflammation. He described it as “revving up our natural anti-inflammatory T cells to help balance the immune system.”
Gene editing. This involves using CRISPR gene editing technology to cut genes as a way to cure disease. “What if we cut the IL-23 receptor?” Dr. Blauvelt asked. “You would get rid of that whole signaling pathway. Would the patient be fine?”
In an interview a the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that Dr. Blauvelt “has a very exciting view” of the future of psoriasis treatments. “I think that some of it will come true; we’ll have to see which,” Dr. Stein Gold said. “The idea that we might be able to change the trajectory of disease by being aggressive upfront, and possibly modify the course, is exciting. That would be a wonderful new treatment approach.”
Dr. Blauvelt disclosed ties with AbbVie, Abcentra, Affibody, Aligos, Almirall, Alumis, Amgen, AnaptysBio, Arcutis, Arena, ASLAN Pharma, Athenex, Bluefin, Boehringer Ingelheim, Bristol Myers Squibb, Cara, Dermavant, EcoR1, Escient, Evelo, Evommune, Forte, Galderma, Highlightll, Incyte, Innovent Bio, Janssen, Landos, Leo, Lilly, Merck, Novartis, Pfizer, Rapt, Regeneron, Sanofi-Genzyme, Spherix, Sun Pharmaceuticals Industries, TLL Pharmaceutical, TrialSpark, UCB Pharma, Vibliome, and Xencor.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries, Ltd.
Medscape and this news organization are owned by the same parent company.
HONOLULU – During office visits with Andrew Blauvelt, MD, MBA, many patients well controlled on biologic therapy for their moderate to severe psoriasis often ask him when their scheduled injections can stop.
The most common question he hears is, “ ‘Why do I have to keep doing this? I’ve been clear for 2 or 3 years,’ ” Dr. Blauvelt, president of Oregon Medical Research Center, Portland, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “We have terrific drugs for psoriasis, but how can we do better?”
Development of oral biologics. At least two companies are developing a peptide-type small molecule that blocks interleukin (IL)-17 or IL-23 signaling, but would be given as a pill, he said. Another concept in the works is a robotic pill for drug delivery. The pill, which is being developed by Rani Therapeutics, protects the biotherapeutic drug payload from digestion in the GI tract and auto-injects it into the wall of the small intestine, according to a report of two studies that demonstrated the safety and tolerability of the robotic pill in healthy humans.
In an animal study, the same researchers showed that delivering monoclonal antibodies with the robotic pill achieved bioavailability on par with that obtained by standard subcutaneous injections.
Identifying “super responders” who require less frequent dosing of medication. “There’s data to suggest that we can kind of back off treatment in these patients,” Dr. Blauvelt said.
Hitting treatment hard and early. “There’s a concept in medicine of hitting disease hard and hitting it early, before the disease can establish itself and cause damage,” he said.
Targeting tissue resident memory T cells. In psoriasis, the idea is that if you treat earlier, when patients are just diagnosed, “perhaps you might be able to decrease resident memory T cells that set up shop in the skin and are responsible for disease recurrences,” Dr. Blauvelt said. “Research has shown that IL-23 blockers decrease tissue resident memory T cells, and IL-17 blockers don’t. This could explain why we see long remissions in this class of drug because we’re getting at these resident memory T cells and knocking them down,” he explained. “Our hypothesis is that hitting hard and early in the treatment course with high-dose IL-23 blockade may be an effective strategy to induce long-term remissions and possible cure, what we call ‘knock-out therapy.’ ”
In a pilot study of 20 patients, Dr. Blauvelt and colleagues are evaluating whether higher initial doses of the IL-23 antagonist risankizumab (300 mg and 600 mg, 2 times and 4 times the standard initial doses for plaque psoriasis) can more effectively target resident memory T cells. “This involves dosing at weeks 0, 4, and 16, then stopping and measuring resident T cells in the tissue to see how long we can induce psoriasis remissions,” Dr. Blauvelt said.
“I have no data to share, but I think we have the potential for unprecedented PASI-100 numbers with no added safety concerns, and the potential to break away from established regular dosing patterns,” such as the possibility of yearly dosing, the possibility of long-term remissions, and the possibility of cure in some patients, he noted.
Inducing tolerance. This refers to efforts aimed at increasing regulatory T cells, which are natural T cells that calm inflammation. He described it as “revving up our natural anti-inflammatory T cells to help balance the immune system.”
Gene editing. This involves using CRISPR gene editing technology to cut genes as a way to cure disease. “What if we cut the IL-23 receptor?” Dr. Blauvelt asked. “You would get rid of that whole signaling pathway. Would the patient be fine?”
In an interview a the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that Dr. Blauvelt “has a very exciting view” of the future of psoriasis treatments. “I think that some of it will come true; we’ll have to see which,” Dr. Stein Gold said. “The idea that we might be able to change the trajectory of disease by being aggressive upfront, and possibly modify the course, is exciting. That would be a wonderful new treatment approach.”
Dr. Blauvelt disclosed ties with AbbVie, Abcentra, Affibody, Aligos, Almirall, Alumis, Amgen, AnaptysBio, Arcutis, Arena, ASLAN Pharma, Athenex, Bluefin, Boehringer Ingelheim, Bristol Myers Squibb, Cara, Dermavant, EcoR1, Escient, Evelo, Evommune, Forte, Galderma, Highlightll, Incyte, Innovent Bio, Janssen, Landos, Leo, Lilly, Merck, Novartis, Pfizer, Rapt, Regeneron, Sanofi-Genzyme, Spherix, Sun Pharmaceuticals Industries, TLL Pharmaceutical, TrialSpark, UCB Pharma, Vibliome, and Xencor.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries, Ltd.
Medscape and this news organization are owned by the same parent company.
HONOLULU – During office visits with Andrew Blauvelt, MD, MBA, many patients well controlled on biologic therapy for their moderate to severe psoriasis often ask him when their scheduled injections can stop.
The most common question he hears is, “ ‘Why do I have to keep doing this? I’ve been clear for 2 or 3 years,’ ” Dr. Blauvelt, president of Oregon Medical Research Center, Portland, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “We have terrific drugs for psoriasis, but how can we do better?”
Development of oral biologics. At least two companies are developing a peptide-type small molecule that blocks interleukin (IL)-17 or IL-23 signaling, but would be given as a pill, he said. Another concept in the works is a robotic pill for drug delivery. The pill, which is being developed by Rani Therapeutics, protects the biotherapeutic drug payload from digestion in the GI tract and auto-injects it into the wall of the small intestine, according to a report of two studies that demonstrated the safety and tolerability of the robotic pill in healthy humans.
In an animal study, the same researchers showed that delivering monoclonal antibodies with the robotic pill achieved bioavailability on par with that obtained by standard subcutaneous injections.
Identifying “super responders” who require less frequent dosing of medication. “There’s data to suggest that we can kind of back off treatment in these patients,” Dr. Blauvelt said.
Hitting treatment hard and early. “There’s a concept in medicine of hitting disease hard and hitting it early, before the disease can establish itself and cause damage,” he said.
Targeting tissue resident memory T cells. In psoriasis, the idea is that if you treat earlier, when patients are just diagnosed, “perhaps you might be able to decrease resident memory T cells that set up shop in the skin and are responsible for disease recurrences,” Dr. Blauvelt said. “Research has shown that IL-23 blockers decrease tissue resident memory T cells, and IL-17 blockers don’t. This could explain why we see long remissions in this class of drug because we’re getting at these resident memory T cells and knocking them down,” he explained. “Our hypothesis is that hitting hard and early in the treatment course with high-dose IL-23 blockade may be an effective strategy to induce long-term remissions and possible cure, what we call ‘knock-out therapy.’ ”
In a pilot study of 20 patients, Dr. Blauvelt and colleagues are evaluating whether higher initial doses of the IL-23 antagonist risankizumab (300 mg and 600 mg, 2 times and 4 times the standard initial doses for plaque psoriasis) can more effectively target resident memory T cells. “This involves dosing at weeks 0, 4, and 16, then stopping and measuring resident T cells in the tissue to see how long we can induce psoriasis remissions,” Dr. Blauvelt said.
“I have no data to share, but I think we have the potential for unprecedented PASI-100 numbers with no added safety concerns, and the potential to break away from established regular dosing patterns,” such as the possibility of yearly dosing, the possibility of long-term remissions, and the possibility of cure in some patients, he noted.
Inducing tolerance. This refers to efforts aimed at increasing regulatory T cells, which are natural T cells that calm inflammation. He described it as “revving up our natural anti-inflammatory T cells to help balance the immune system.”
Gene editing. This involves using CRISPR gene editing technology to cut genes as a way to cure disease. “What if we cut the IL-23 receptor?” Dr. Blauvelt asked. “You would get rid of that whole signaling pathway. Would the patient be fine?”
In an interview a the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that Dr. Blauvelt “has a very exciting view” of the future of psoriasis treatments. “I think that some of it will come true; we’ll have to see which,” Dr. Stein Gold said. “The idea that we might be able to change the trajectory of disease by being aggressive upfront, and possibly modify the course, is exciting. That would be a wonderful new treatment approach.”
Dr. Blauvelt disclosed ties with AbbVie, Abcentra, Affibody, Aligos, Almirall, Alumis, Amgen, AnaptysBio, Arcutis, Arena, ASLAN Pharma, Athenex, Bluefin, Boehringer Ingelheim, Bristol Myers Squibb, Cara, Dermavant, EcoR1, Escient, Evelo, Evommune, Forte, Galderma, Highlightll, Incyte, Innovent Bio, Janssen, Landos, Leo, Lilly, Merck, Novartis, Pfizer, Rapt, Regeneron, Sanofi-Genzyme, Spherix, Sun Pharmaceuticals Industries, TLL Pharmaceutical, TrialSpark, UCB Pharma, Vibliome, and Xencor.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries, Ltd.
Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPE LIVE! HAWAII DERMATOLOGY SEMINAR