User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Everyone wins when losers get paid
Bribery really is the solution to all of life’s problems
Breaking news: The United States has a bit of an obesity epidemic. Okay, maybe not so breaking news. But it’s a problem we’ve been struggling with for a very long time. Part of the issue is that there really is no secret to weight loss. Pretty much anything can work if you’re committed. The millions of diets floating around are testament to this idea.
The problem of losing weight is amplified if you don’t rake in the big bucks. Lower-income individuals often can’t afford healthy superfoods, and they’re often too busy to spend time at classes, exercising, or following programs. A group of researchers at New York University has offered up an alternate solution to encourage weight loss in low-income people: Pay them.
Specifically, pay them for losing weight. A reward, if you will. The researchers recruited several hundred lower-income people and split them into three groups. All participants received a free 1-year membership to a gym and weight-loss program, as well as food journals and fitness devices, but one group received payment (on average, about $300 overall) for attending meetings, exercising a certain amount every week, or weighing themselves twice a week. About 40% of people in this group lost 5% of their body weight after 6 months, twice as many as in the group that did not receive payment for performing these tasks.
The big winners, however, were those in the third group. They also received the free stuff, but the researchers offered them a more simple and direct bribe: Lose 5% of your weight over 6 months and we’ll pay you. The reward? About $450 on average, and it worked very well, with half this group losing the weight after 6 months. That said, after a year something like a fifth of this group put the weight back on, bringing them in line with the group that was paid to perform tasks. Still, both groups outperformed the control group, which received no money.
The takeaway from this research is pretty obvious. Pay people a fair price to do something, and they’ll do it. This is a lesson that has absolutely no relevance in the modern world. Nope, none whatsoever. We all receive completely fair wages. We all have plenty of money to pay for things. Everything is fine.
More green space, less medicine
Have you heard of the 3-30-300 rule? Proposed by urban forester Cecil Konijnendijk, it’s become the rule of thumb for urban planners and other foresters into getting more green space in populated areas. A recent study has found that people who lived within this 3-30-300 rule had better mental health and less medication use.
If you’re not an urban forester, however, you may not know what the 3-30-300 rule is. But it’s pretty simple, people should be able to see at least three trees from their home, have 30% tree canopy in their neighborhood, and have 300 Spartans to defend against the Persian army.
We may have made that last one up. It’s actually have a green space or park within 300 meters of your home.
In the new study, only 4.7% of people surveyed lived in an area that followed all three rules. About 62% of the surveyed lived with a green space at least 300 meters away, 43% had at least three trees within 15 meters from their home, and a rather pitiful 9% had adequate tree canopy coverage in their neighborhood.
Greater adherence to the 3-30-300 rule was associated with fewer visits to the psychologist, with 8.3% of the participants reporting a psychologist visit in the last year. The data come from a sample of a little over 3,000 Barcelona residents aged 15-97 who were randomly selected to participate in the Barcelona Public Health Agency Survey.
“There is an urgent need to provide citizens with more green space,” said Mark Nieuwenhuijsen, lead author of the study. “We may need to tear out asphalt and plant more trees, which would not only improve health, but also reduce heat island effects and contribute to carbon capture.”
The main goal and message is that more green space is good for everyone. So if you’re feeling a little overwhelmed, take a breather and sit somewhere green. Or call those 300 Spartans and get them to start knocking some buildings down.
Said the toilet to the engineer: Do you hear what I hear?
A mythical hero’s journey took Dorothy along the yellow brick road to find the Wizard of Oz. Huckleberry Finn used a raft to float down the Mississippi River. Luke Skywalker did most of his traveling between planets. For the rest of us, the journey may be just a bit shorter.
Also a bit less heroic. Unless, of course, you’re prepping for a colonoscopy. Yup, we’re headed to the toilet, but not just any toilet. This toilet was the subject of a presentation at the annual meeting of the Acoustical Society of America, titled “The feces thesis: Using machine learning to detect diarrhea,” and that presentation was the hero’s journey of Maia Gatlin, PhD, a research engineer at the Georgia Institute of Technology.
She and her team attached a noninvasive microphone sensor to a toilet, and now they can identify bowel diseases without collecting any identifiable information.
The audio sample of an excretion event is “transformed into a spectrogram, which essentially captures the sound in an image. Different events produce different features in the audio and the spectrogram. For example, urination creates a consistent tone, while defecation may have a singular tone. In contrast, diarrhea is more random,” they explained in the written statement.
They used a machine learning algorithm to classify each spectrogram based on its features. “The algorithm’s performance was tested against data with and without background noises to make sure it was learning the right sound features, regardless of the sensor’s environment,” Dr. Gatlin and associates wrote.
Their goal is to use the toilet sensor in areas where cholera is common to prevent the spread of disease. After that, who knows? “Perhaps someday, our algorithm can be used with existing in-home smart devices to monitor one’s own bowel movements and health!” she suggested.
That would be a heroic toilet indeed.
Bribery really is the solution to all of life’s problems
Breaking news: The United States has a bit of an obesity epidemic. Okay, maybe not so breaking news. But it’s a problem we’ve been struggling with for a very long time. Part of the issue is that there really is no secret to weight loss. Pretty much anything can work if you’re committed. The millions of diets floating around are testament to this idea.
The problem of losing weight is amplified if you don’t rake in the big bucks. Lower-income individuals often can’t afford healthy superfoods, and they’re often too busy to spend time at classes, exercising, or following programs. A group of researchers at New York University has offered up an alternate solution to encourage weight loss in low-income people: Pay them.
Specifically, pay them for losing weight. A reward, if you will. The researchers recruited several hundred lower-income people and split them into three groups. All participants received a free 1-year membership to a gym and weight-loss program, as well as food journals and fitness devices, but one group received payment (on average, about $300 overall) for attending meetings, exercising a certain amount every week, or weighing themselves twice a week. About 40% of people in this group lost 5% of their body weight after 6 months, twice as many as in the group that did not receive payment for performing these tasks.
The big winners, however, were those in the third group. They also received the free stuff, but the researchers offered them a more simple and direct bribe: Lose 5% of your weight over 6 months and we’ll pay you. The reward? About $450 on average, and it worked very well, with half this group losing the weight after 6 months. That said, after a year something like a fifth of this group put the weight back on, bringing them in line with the group that was paid to perform tasks. Still, both groups outperformed the control group, which received no money.
The takeaway from this research is pretty obvious. Pay people a fair price to do something, and they’ll do it. This is a lesson that has absolutely no relevance in the modern world. Nope, none whatsoever. We all receive completely fair wages. We all have plenty of money to pay for things. Everything is fine.
More green space, less medicine
Have you heard of the 3-30-300 rule? Proposed by urban forester Cecil Konijnendijk, it’s become the rule of thumb for urban planners and other foresters into getting more green space in populated areas. A recent study has found that people who lived within this 3-30-300 rule had better mental health and less medication use.
If you’re not an urban forester, however, you may not know what the 3-30-300 rule is. But it’s pretty simple, people should be able to see at least three trees from their home, have 30% tree canopy in their neighborhood, and have 300 Spartans to defend against the Persian army.
We may have made that last one up. It’s actually have a green space or park within 300 meters of your home.
In the new study, only 4.7% of people surveyed lived in an area that followed all three rules. About 62% of the surveyed lived with a green space at least 300 meters away, 43% had at least three trees within 15 meters from their home, and a rather pitiful 9% had adequate tree canopy coverage in their neighborhood.
Greater adherence to the 3-30-300 rule was associated with fewer visits to the psychologist, with 8.3% of the participants reporting a psychologist visit in the last year. The data come from a sample of a little over 3,000 Barcelona residents aged 15-97 who were randomly selected to participate in the Barcelona Public Health Agency Survey.
“There is an urgent need to provide citizens with more green space,” said Mark Nieuwenhuijsen, lead author of the study. “We may need to tear out asphalt and plant more trees, which would not only improve health, but also reduce heat island effects and contribute to carbon capture.”
The main goal and message is that more green space is good for everyone. So if you’re feeling a little overwhelmed, take a breather and sit somewhere green. Or call those 300 Spartans and get them to start knocking some buildings down.
Said the toilet to the engineer: Do you hear what I hear?
A mythical hero’s journey took Dorothy along the yellow brick road to find the Wizard of Oz. Huckleberry Finn used a raft to float down the Mississippi River. Luke Skywalker did most of his traveling between planets. For the rest of us, the journey may be just a bit shorter.
Also a bit less heroic. Unless, of course, you’re prepping for a colonoscopy. Yup, we’re headed to the toilet, but not just any toilet. This toilet was the subject of a presentation at the annual meeting of the Acoustical Society of America, titled “The feces thesis: Using machine learning to detect diarrhea,” and that presentation was the hero’s journey of Maia Gatlin, PhD, a research engineer at the Georgia Institute of Technology.
She and her team attached a noninvasive microphone sensor to a toilet, and now they can identify bowel diseases without collecting any identifiable information.
The audio sample of an excretion event is “transformed into a spectrogram, which essentially captures the sound in an image. Different events produce different features in the audio and the spectrogram. For example, urination creates a consistent tone, while defecation may have a singular tone. In contrast, diarrhea is more random,” they explained in the written statement.
They used a machine learning algorithm to classify each spectrogram based on its features. “The algorithm’s performance was tested against data with and without background noises to make sure it was learning the right sound features, regardless of the sensor’s environment,” Dr. Gatlin and associates wrote.
Their goal is to use the toilet sensor in areas where cholera is common to prevent the spread of disease. After that, who knows? “Perhaps someday, our algorithm can be used with existing in-home smart devices to monitor one’s own bowel movements and health!” she suggested.
That would be a heroic toilet indeed.
Bribery really is the solution to all of life’s problems
Breaking news: The United States has a bit of an obesity epidemic. Okay, maybe not so breaking news. But it’s a problem we’ve been struggling with for a very long time. Part of the issue is that there really is no secret to weight loss. Pretty much anything can work if you’re committed. The millions of diets floating around are testament to this idea.
The problem of losing weight is amplified if you don’t rake in the big bucks. Lower-income individuals often can’t afford healthy superfoods, and they’re often too busy to spend time at classes, exercising, or following programs. A group of researchers at New York University has offered up an alternate solution to encourage weight loss in low-income people: Pay them.
Specifically, pay them for losing weight. A reward, if you will. The researchers recruited several hundred lower-income people and split them into three groups. All participants received a free 1-year membership to a gym and weight-loss program, as well as food journals and fitness devices, but one group received payment (on average, about $300 overall) for attending meetings, exercising a certain amount every week, or weighing themselves twice a week. About 40% of people in this group lost 5% of their body weight after 6 months, twice as many as in the group that did not receive payment for performing these tasks.
The big winners, however, were those in the third group. They also received the free stuff, but the researchers offered them a more simple and direct bribe: Lose 5% of your weight over 6 months and we’ll pay you. The reward? About $450 on average, and it worked very well, with half this group losing the weight after 6 months. That said, after a year something like a fifth of this group put the weight back on, bringing them in line with the group that was paid to perform tasks. Still, both groups outperformed the control group, which received no money.
The takeaway from this research is pretty obvious. Pay people a fair price to do something, and they’ll do it. This is a lesson that has absolutely no relevance in the modern world. Nope, none whatsoever. We all receive completely fair wages. We all have plenty of money to pay for things. Everything is fine.
More green space, less medicine
Have you heard of the 3-30-300 rule? Proposed by urban forester Cecil Konijnendijk, it’s become the rule of thumb for urban planners and other foresters into getting more green space in populated areas. A recent study has found that people who lived within this 3-30-300 rule had better mental health and less medication use.
If you’re not an urban forester, however, you may not know what the 3-30-300 rule is. But it’s pretty simple, people should be able to see at least three trees from their home, have 30% tree canopy in their neighborhood, and have 300 Spartans to defend against the Persian army.
We may have made that last one up. It’s actually have a green space or park within 300 meters of your home.
In the new study, only 4.7% of people surveyed lived in an area that followed all three rules. About 62% of the surveyed lived with a green space at least 300 meters away, 43% had at least three trees within 15 meters from their home, and a rather pitiful 9% had adequate tree canopy coverage in their neighborhood.
Greater adherence to the 3-30-300 rule was associated with fewer visits to the psychologist, with 8.3% of the participants reporting a psychologist visit in the last year. The data come from a sample of a little over 3,000 Barcelona residents aged 15-97 who were randomly selected to participate in the Barcelona Public Health Agency Survey.
“There is an urgent need to provide citizens with more green space,” said Mark Nieuwenhuijsen, lead author of the study. “We may need to tear out asphalt and plant more trees, which would not only improve health, but also reduce heat island effects and contribute to carbon capture.”
The main goal and message is that more green space is good for everyone. So if you’re feeling a little overwhelmed, take a breather and sit somewhere green. Or call those 300 Spartans and get them to start knocking some buildings down.
Said the toilet to the engineer: Do you hear what I hear?
A mythical hero’s journey took Dorothy along the yellow brick road to find the Wizard of Oz. Huckleberry Finn used a raft to float down the Mississippi River. Luke Skywalker did most of his traveling between planets. For the rest of us, the journey may be just a bit shorter.
Also a bit less heroic. Unless, of course, you’re prepping for a colonoscopy. Yup, we’re headed to the toilet, but not just any toilet. This toilet was the subject of a presentation at the annual meeting of the Acoustical Society of America, titled “The feces thesis: Using machine learning to detect diarrhea,” and that presentation was the hero’s journey of Maia Gatlin, PhD, a research engineer at the Georgia Institute of Technology.
She and her team attached a noninvasive microphone sensor to a toilet, and now they can identify bowel diseases without collecting any identifiable information.
The audio sample of an excretion event is “transformed into a spectrogram, which essentially captures the sound in an image. Different events produce different features in the audio and the spectrogram. For example, urination creates a consistent tone, while defecation may have a singular tone. In contrast, diarrhea is more random,” they explained in the written statement.
They used a machine learning algorithm to classify each spectrogram based on its features. “The algorithm’s performance was tested against data with and without background noises to make sure it was learning the right sound features, regardless of the sensor’s environment,” Dr. Gatlin and associates wrote.
Their goal is to use the toilet sensor in areas where cholera is common to prevent the spread of disease. After that, who knows? “Perhaps someday, our algorithm can be used with existing in-home smart devices to monitor one’s own bowel movements and health!” she suggested.
That would be a heroic toilet indeed.
‘Slugging’: A TikTok skin trend that has some merit
They’ve been around for a while and show no signs of going away: videos on TikTok of people, often teens, slathering their face with petroleum jelly and claiming that it’s transformed their skin, cured their acne, or given them an amazing “glow up.”
TikTok videos mentioning petrolatum increased by 46% and Instagram videos by 93% from 2021 to 2022, reported Gabriel Santos Malave, BA, of the Icahn School of Medicine at Mount Sinai, New York, and William D. James, MD, professor of dermatology, University of Pennsylvania, Philadelphia, in a review of petroleum jelly’s uses recently published in Cutis.
The authors said that after application of a moisturizer.
In a typical demonstration, a dermatologist in the United Kingdom showed how she incorporates slugging into her routine in a TikTok video that’s had more than 1 million views.
Unlike many TikTok trends, slugging may not be entirely bad, say dermatologists.
“I think it’s a great way to keep your skin protected and moisturized, especially in those dry, cold winter months,” said dermatologist Mamina Turegano, MD, in a video posted in February 2022. That TikTok video has had more than 6 million views.
Dr. Turegano, who is in private practice in the New Orleans suburb of Metairie, La., told this news organization that she decided to post about slugging after she’d noticed that the topic was trending. Also, she had tried the technique herself when she was a resident in Washington more than a decade ago.
At the time, Dr. Turegano said that she was aware that “putting petroleum jelly on your face was not a normal thing.” But, given its history of being used in dermatology, she gave it a try and found that it worked well for her dry skin, she said.
Dr. Turegano is one among many dermatologists who have joined TikTok to dispel myths, educate, and inform. It’s important for them to be there “to engage and empower the public to become a better consumer of information out there and take ownership of their skin health,” said Jean McGee, MD, PhD, a dermatologist at Beth Israel Deaconess Medical Center, Boston, and assistant professor of dermatology at Harvard Medical School, also in Boston.
Dr. McGee and colleagues studied TikTok content on slugging and found that by far, videos that were created by health care providers were more educational. Dermatologists who posted were more likely to discuss the risks and benefits, whereas so-called “influencers” rarely posted on the risks, according to the study, published in Clinics in Dermatology.
Slugging is generally safe and effective for those who have a compromised skin barrier or “for those who have sensitive skin and can’t tolerate other products but need some form of moisturization,” said Dr. Turegano.
“Its oil-based nature allows it to seal water in the skin by creating a hydrophobic barrier that decreases transepidermal water loss (TEWL),” write Mr. Malave and Dr. James in Cutis. They note that petrolatum reduces TEWL by 98%, compared with only 20% to 30% for other oil-based moisturizers.
Dermatologists have often recommended a “seal and trap” regimen for dry skin or eczema. It involves a short, lukewarm shower, followed by immediately moisturizing with a petrolatum-based ointment, said Dr. McGee.
This could be safe for the face, but “other variables need to be considered,” including use of other topical medications and other skin care practices, she added.
The concept of double-layering a moisturizer and an occlusive agent can be beneficial but more typically for the hands and feet, where the skin can be severely dry and cracked, said Adam Friedman, MD, professor and chair of dermatology, George Washington University, Washington. “I would not recommend that on the face,” Dr. Friedman told this news organization.
He and other dermatologists warned about the potential for slugging – given petroleum jelly’s occlusive nature – to enhance the action of any topical steroid, retinol, or exfoliating agent.
Muneeb Shah, MD, who practices in Mooresville, N.C., is one of the most popular dermatologists on TikTok, with more than 17 million followers. He also warned in a February 2022 video about potential downsides. “Be careful after using retinol or exfoliating acids because it may actually irritate your skin more,” he says in the video.
“Slugging is awesome for some people but not for others, and not for every night,” said Whitney Bowe, MD, on a TikTok video she posted in July. She recommended it for eczema or really dry skin. Dr. Bowe, who practices with Advanced Dermatology in New York, advised those with acne-prone skin to “skip this trend.”
On a web page aimed at the general public, the American Academy of Dermatology similarly cautioned, “Avoid putting petroleum jelly on your face if you are acne-prone, as this may cause breakouts in some people.”
Acne cure or pore clogger?
And yet, plenty of TikTok users claim that it has improved their acne.
One such user posted a before and after video purporting to show that slugging had almost completely eliminated her acne and prior scarring. Not surprisingly, it has been viewed some 9 million times and got 1.5 million “likes.”
Dr. Friedman notes that it’s theoretically possible – but not likely – that acne could improve by slugging, given that acne basically is a disease of barrier disruption. “The idea here is you have disrupted skin barrier throughout the face regardless of whether you have a pimple in that spot or not, so you need to repair it,” he said. “That’s where I think slugging is somewhat on the right track, because by putting an occlusive agent on the skin, you are restoring the barrier element,” he said.
However, applying a thick, greasy ointment on the face could block pores and cause a backup of sebum and dead skin cells, and it could trap bacteria, he said. “Skin barrier protection and repair is central to acne management, but you need to do it in a safe way,” he said. He noted that that means applying an oil-free moisturizer to damp skin.
Dr. Turegano said she has seen slugging improve acne, but it’s hard to say which people with acne-prone skin would be the best candidates. Those who have used harsh products to treat acne and subsequently experienced worsening acne could potentially benefit, she said.
Even so, she said, “I’d be very cautious in anyone with acne.”
Dr. Friedman, Dr. McGee, and Dr. Turegano reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
They’ve been around for a while and show no signs of going away: videos on TikTok of people, often teens, slathering their face with petroleum jelly and claiming that it’s transformed their skin, cured their acne, or given them an amazing “glow up.”
TikTok videos mentioning petrolatum increased by 46% and Instagram videos by 93% from 2021 to 2022, reported Gabriel Santos Malave, BA, of the Icahn School of Medicine at Mount Sinai, New York, and William D. James, MD, professor of dermatology, University of Pennsylvania, Philadelphia, in a review of petroleum jelly’s uses recently published in Cutis.
The authors said that after application of a moisturizer.
In a typical demonstration, a dermatologist in the United Kingdom showed how she incorporates slugging into her routine in a TikTok video that’s had more than 1 million views.
Unlike many TikTok trends, slugging may not be entirely bad, say dermatologists.
“I think it’s a great way to keep your skin protected and moisturized, especially in those dry, cold winter months,” said dermatologist Mamina Turegano, MD, in a video posted in February 2022. That TikTok video has had more than 6 million views.
Dr. Turegano, who is in private practice in the New Orleans suburb of Metairie, La., told this news organization that she decided to post about slugging after she’d noticed that the topic was trending. Also, she had tried the technique herself when she was a resident in Washington more than a decade ago.
At the time, Dr. Turegano said that she was aware that “putting petroleum jelly on your face was not a normal thing.” But, given its history of being used in dermatology, she gave it a try and found that it worked well for her dry skin, she said.
Dr. Turegano is one among many dermatologists who have joined TikTok to dispel myths, educate, and inform. It’s important for them to be there “to engage and empower the public to become a better consumer of information out there and take ownership of their skin health,” said Jean McGee, MD, PhD, a dermatologist at Beth Israel Deaconess Medical Center, Boston, and assistant professor of dermatology at Harvard Medical School, also in Boston.
Dr. McGee and colleagues studied TikTok content on slugging and found that by far, videos that were created by health care providers were more educational. Dermatologists who posted were more likely to discuss the risks and benefits, whereas so-called “influencers” rarely posted on the risks, according to the study, published in Clinics in Dermatology.
Slugging is generally safe and effective for those who have a compromised skin barrier or “for those who have sensitive skin and can’t tolerate other products but need some form of moisturization,” said Dr. Turegano.
“Its oil-based nature allows it to seal water in the skin by creating a hydrophobic barrier that decreases transepidermal water loss (TEWL),” write Mr. Malave and Dr. James in Cutis. They note that petrolatum reduces TEWL by 98%, compared with only 20% to 30% for other oil-based moisturizers.
Dermatologists have often recommended a “seal and trap” regimen for dry skin or eczema. It involves a short, lukewarm shower, followed by immediately moisturizing with a petrolatum-based ointment, said Dr. McGee.
This could be safe for the face, but “other variables need to be considered,” including use of other topical medications and other skin care practices, she added.
The concept of double-layering a moisturizer and an occlusive agent can be beneficial but more typically for the hands and feet, where the skin can be severely dry and cracked, said Adam Friedman, MD, professor and chair of dermatology, George Washington University, Washington. “I would not recommend that on the face,” Dr. Friedman told this news organization.
He and other dermatologists warned about the potential for slugging – given petroleum jelly’s occlusive nature – to enhance the action of any topical steroid, retinol, or exfoliating agent.
Muneeb Shah, MD, who practices in Mooresville, N.C., is one of the most popular dermatologists on TikTok, with more than 17 million followers. He also warned in a February 2022 video about potential downsides. “Be careful after using retinol or exfoliating acids because it may actually irritate your skin more,” he says in the video.
“Slugging is awesome for some people but not for others, and not for every night,” said Whitney Bowe, MD, on a TikTok video she posted in July. She recommended it for eczema or really dry skin. Dr. Bowe, who practices with Advanced Dermatology in New York, advised those with acne-prone skin to “skip this trend.”
On a web page aimed at the general public, the American Academy of Dermatology similarly cautioned, “Avoid putting petroleum jelly on your face if you are acne-prone, as this may cause breakouts in some people.”
Acne cure or pore clogger?
And yet, plenty of TikTok users claim that it has improved their acne.
One such user posted a before and after video purporting to show that slugging had almost completely eliminated her acne and prior scarring. Not surprisingly, it has been viewed some 9 million times and got 1.5 million “likes.”
Dr. Friedman notes that it’s theoretically possible – but not likely – that acne could improve by slugging, given that acne basically is a disease of barrier disruption. “The idea here is you have disrupted skin barrier throughout the face regardless of whether you have a pimple in that spot or not, so you need to repair it,” he said. “That’s where I think slugging is somewhat on the right track, because by putting an occlusive agent on the skin, you are restoring the barrier element,” he said.
However, applying a thick, greasy ointment on the face could block pores and cause a backup of sebum and dead skin cells, and it could trap bacteria, he said. “Skin barrier protection and repair is central to acne management, but you need to do it in a safe way,” he said. He noted that that means applying an oil-free moisturizer to damp skin.
Dr. Turegano said she has seen slugging improve acne, but it’s hard to say which people with acne-prone skin would be the best candidates. Those who have used harsh products to treat acne and subsequently experienced worsening acne could potentially benefit, she said.
Even so, she said, “I’d be very cautious in anyone with acne.”
Dr. Friedman, Dr. McGee, and Dr. Turegano reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
They’ve been around for a while and show no signs of going away: videos on TikTok of people, often teens, slathering their face with petroleum jelly and claiming that it’s transformed their skin, cured their acne, or given them an amazing “glow up.”
TikTok videos mentioning petrolatum increased by 46% and Instagram videos by 93% from 2021 to 2022, reported Gabriel Santos Malave, BA, of the Icahn School of Medicine at Mount Sinai, New York, and William D. James, MD, professor of dermatology, University of Pennsylvania, Philadelphia, in a review of petroleum jelly’s uses recently published in Cutis.
The authors said that after application of a moisturizer.
In a typical demonstration, a dermatologist in the United Kingdom showed how she incorporates slugging into her routine in a TikTok video that’s had more than 1 million views.
Unlike many TikTok trends, slugging may not be entirely bad, say dermatologists.
“I think it’s a great way to keep your skin protected and moisturized, especially in those dry, cold winter months,” said dermatologist Mamina Turegano, MD, in a video posted in February 2022. That TikTok video has had more than 6 million views.
Dr. Turegano, who is in private practice in the New Orleans suburb of Metairie, La., told this news organization that she decided to post about slugging after she’d noticed that the topic was trending. Also, she had tried the technique herself when she was a resident in Washington more than a decade ago.
At the time, Dr. Turegano said that she was aware that “putting petroleum jelly on your face was not a normal thing.” But, given its history of being used in dermatology, she gave it a try and found that it worked well for her dry skin, she said.
Dr. Turegano is one among many dermatologists who have joined TikTok to dispel myths, educate, and inform. It’s important for them to be there “to engage and empower the public to become a better consumer of information out there and take ownership of their skin health,” said Jean McGee, MD, PhD, a dermatologist at Beth Israel Deaconess Medical Center, Boston, and assistant professor of dermatology at Harvard Medical School, also in Boston.
Dr. McGee and colleagues studied TikTok content on slugging and found that by far, videos that were created by health care providers were more educational. Dermatologists who posted were more likely to discuss the risks and benefits, whereas so-called “influencers” rarely posted on the risks, according to the study, published in Clinics in Dermatology.
Slugging is generally safe and effective for those who have a compromised skin barrier or “for those who have sensitive skin and can’t tolerate other products but need some form of moisturization,” said Dr. Turegano.
“Its oil-based nature allows it to seal water in the skin by creating a hydrophobic barrier that decreases transepidermal water loss (TEWL),” write Mr. Malave and Dr. James in Cutis. They note that petrolatum reduces TEWL by 98%, compared with only 20% to 30% for other oil-based moisturizers.
Dermatologists have often recommended a “seal and trap” regimen for dry skin or eczema. It involves a short, lukewarm shower, followed by immediately moisturizing with a petrolatum-based ointment, said Dr. McGee.
This could be safe for the face, but “other variables need to be considered,” including use of other topical medications and other skin care practices, she added.
The concept of double-layering a moisturizer and an occlusive agent can be beneficial but more typically for the hands and feet, where the skin can be severely dry and cracked, said Adam Friedman, MD, professor and chair of dermatology, George Washington University, Washington. “I would not recommend that on the face,” Dr. Friedman told this news organization.
He and other dermatologists warned about the potential for slugging – given petroleum jelly’s occlusive nature – to enhance the action of any topical steroid, retinol, or exfoliating agent.
Muneeb Shah, MD, who practices in Mooresville, N.C., is one of the most popular dermatologists on TikTok, with more than 17 million followers. He also warned in a February 2022 video about potential downsides. “Be careful after using retinol or exfoliating acids because it may actually irritate your skin more,” he says in the video.
“Slugging is awesome for some people but not for others, and not for every night,” said Whitney Bowe, MD, on a TikTok video she posted in July. She recommended it for eczema or really dry skin. Dr. Bowe, who practices with Advanced Dermatology in New York, advised those with acne-prone skin to “skip this trend.”
On a web page aimed at the general public, the American Academy of Dermatology similarly cautioned, “Avoid putting petroleum jelly on your face if you are acne-prone, as this may cause breakouts in some people.”
Acne cure or pore clogger?
And yet, plenty of TikTok users claim that it has improved their acne.
One such user posted a before and after video purporting to show that slugging had almost completely eliminated her acne and prior scarring. Not surprisingly, it has been viewed some 9 million times and got 1.5 million “likes.”
Dr. Friedman notes that it’s theoretically possible – but not likely – that acne could improve by slugging, given that acne basically is a disease of barrier disruption. “The idea here is you have disrupted skin barrier throughout the face regardless of whether you have a pimple in that spot or not, so you need to repair it,” he said. “That’s where I think slugging is somewhat on the right track, because by putting an occlusive agent on the skin, you are restoring the barrier element,” he said.
However, applying a thick, greasy ointment on the face could block pores and cause a backup of sebum and dead skin cells, and it could trap bacteria, he said. “Skin barrier protection and repair is central to acne management, but you need to do it in a safe way,” he said. He noted that that means applying an oil-free moisturizer to damp skin.
Dr. Turegano said she has seen slugging improve acne, but it’s hard to say which people with acne-prone skin would be the best candidates. Those who have used harsh products to treat acne and subsequently experienced worsening acne could potentially benefit, she said.
Even so, she said, “I’d be very cautious in anyone with acne.”
Dr. Friedman, Dr. McGee, and Dr. Turegano reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
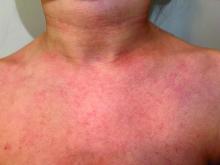
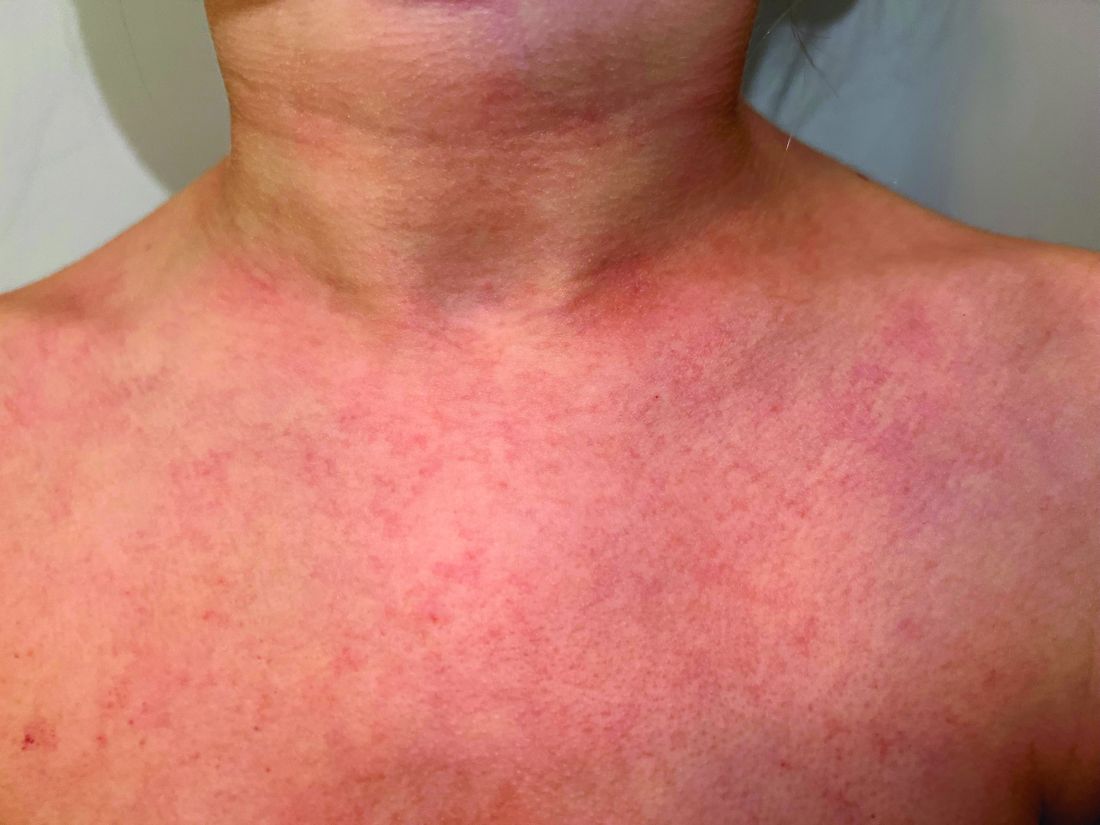
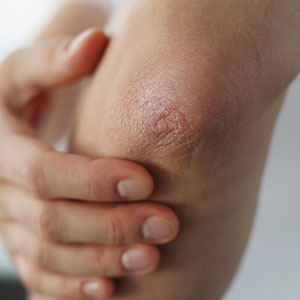
A 9-year old female presented with 1 day of fever, fatigue, and sore throat
This condition typically presents in the setting of Streptococcus pyogenes pharyngitis, or strep throat, and is spread via mucosal transfer in close proximity such as classrooms and nurseries. The dermatologic symptoms are a result of the endotoxin produced by S. pyogenes, which is part of the group A Strep bacteria. Clinically, the presentation can be differentiated from an allergic eruption by its relation to acute pharyngitis, insidious onset, and lack of confluence of the lesions. Diagnosis is supported by a throat culture and rapid strep test, although a rapid test lacks reliability in older patients who are less commonly affected and likely to be carriers. First-line treatment is penicillin or amoxicillin, but first-generation cephalosporins, clindamycin, or erythromycin are sufficient if the patient is allergic to penicillins. Prognosis worsens as time between onset and treatment increases, but is overall excellent now with the introduction of antibiotics and improved hygiene.
Scarlet fever is among a list of many common childhood rashes, and it can be difficult to differentiate between these pathologies on clinical presentation. A few notable childhood dermatologic eruptions include erythema infectiosum (fifth disease), roseola (exanthema subitum or sixth disease), and measles. These cases can be distinguished clinically by the age of the patient, distribution, and quality of the symptoms. Laboratory testing may be used to confirm the diagnosis.
Erythema infectiosum is known as fifth disease or slapped-cheek rash because it commonly presents on the cheeks as a pink, maculopapular rash in a reticular pattern. The disease is caused by parvovirus B19 and is accompanied by low fever, malaise, headache, sore throat, and nausea, which precedes the erythematous rash. The facial rash appears first and is followed by patchy eruptions on the extremities. Appearance of the rash typically indicates the patient is no longer contagious, and patients are treated symptomatically with NSAIDs and antihistamines for associated pruritus.
Roseola infantum is commonly caused by human herpesvirus 6 and is usually found in children 3 years and younger. The defining symptom is a high fever, which is paired with a mild cough, runny nose, and diarrhea. A maculopapular rash appears after the fever subsides, starting centrally and spreading outward to the extremities. Although this rash is similar to measles, they can be differentiated by the order of onset. The rash caused by measles begins on the face and mouth (Koplik spots) and moves downward. Additionally, the patient appears generally healthy and the disease is self-limiting in roseola, while patients with measles will appear more ill and require further attention. Measles is caused by the measles virus of the genus Morbillivirus and is highly contagious. It is spread via respiratory route presenting with fever, cough, coryza, and conjunctivitis followed by the rash. Fortunately, the measles vaccine is in widespread use, so cases have declined over the years.
Our patient had a positive strep test. Influenza and coronavirus tests were negative. She was started on daily amoxicillin and the rash resolved within 2 days of taking the antibiotics.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Tampa, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Allmon A et al.. Am Fam Physician. 2015 Aug 1;92(3):211-6.
Moss WJ. Lancet. 2017 Dec 2;390(10111):2490-502.
Mullins TB and Krishnamurthy K. Roseola Infantum, in “StatPearls.” Treasure Islan, Fla.: StatPearls Publishing, 2022.
Pardo S and Perera TB. Scarlet Fever, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2022.
This condition typically presents in the setting of Streptococcus pyogenes pharyngitis, or strep throat, and is spread via mucosal transfer in close proximity such as classrooms and nurseries. The dermatologic symptoms are a result of the endotoxin produced by S. pyogenes, which is part of the group A Strep bacteria. Clinically, the presentation can be differentiated from an allergic eruption by its relation to acute pharyngitis, insidious onset, and lack of confluence of the lesions. Diagnosis is supported by a throat culture and rapid strep test, although a rapid test lacks reliability in older patients who are less commonly affected and likely to be carriers. First-line treatment is penicillin or amoxicillin, but first-generation cephalosporins, clindamycin, or erythromycin are sufficient if the patient is allergic to penicillins. Prognosis worsens as time between onset and treatment increases, but is overall excellent now with the introduction of antibiotics and improved hygiene.
Scarlet fever is among a list of many common childhood rashes, and it can be difficult to differentiate between these pathologies on clinical presentation. A few notable childhood dermatologic eruptions include erythema infectiosum (fifth disease), roseola (exanthema subitum or sixth disease), and measles. These cases can be distinguished clinically by the age of the patient, distribution, and quality of the symptoms. Laboratory testing may be used to confirm the diagnosis.
Erythema infectiosum is known as fifth disease or slapped-cheek rash because it commonly presents on the cheeks as a pink, maculopapular rash in a reticular pattern. The disease is caused by parvovirus B19 and is accompanied by low fever, malaise, headache, sore throat, and nausea, which precedes the erythematous rash. The facial rash appears first and is followed by patchy eruptions on the extremities. Appearance of the rash typically indicates the patient is no longer contagious, and patients are treated symptomatically with NSAIDs and antihistamines for associated pruritus.
Roseola infantum is commonly caused by human herpesvirus 6 and is usually found in children 3 years and younger. The defining symptom is a high fever, which is paired with a mild cough, runny nose, and diarrhea. A maculopapular rash appears after the fever subsides, starting centrally and spreading outward to the extremities. Although this rash is similar to measles, they can be differentiated by the order of onset. The rash caused by measles begins on the face and mouth (Koplik spots) and moves downward. Additionally, the patient appears generally healthy and the disease is self-limiting in roseola, while patients with measles will appear more ill and require further attention. Measles is caused by the measles virus of the genus Morbillivirus and is highly contagious. It is spread via respiratory route presenting with fever, cough, coryza, and conjunctivitis followed by the rash. Fortunately, the measles vaccine is in widespread use, so cases have declined over the years.
Our patient had a positive strep test. Influenza and coronavirus tests were negative. She was started on daily amoxicillin and the rash resolved within 2 days of taking the antibiotics.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Tampa, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Allmon A et al.. Am Fam Physician. 2015 Aug 1;92(3):211-6.
Moss WJ. Lancet. 2017 Dec 2;390(10111):2490-502.
Mullins TB and Krishnamurthy K. Roseola Infantum, in “StatPearls.” Treasure Islan, Fla.: StatPearls Publishing, 2022.
Pardo S and Perera TB. Scarlet Fever, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2022.
This condition typically presents in the setting of Streptococcus pyogenes pharyngitis, or strep throat, and is spread via mucosal transfer in close proximity such as classrooms and nurseries. The dermatologic symptoms are a result of the endotoxin produced by S. pyogenes, which is part of the group A Strep bacteria. Clinically, the presentation can be differentiated from an allergic eruption by its relation to acute pharyngitis, insidious onset, and lack of confluence of the lesions. Diagnosis is supported by a throat culture and rapid strep test, although a rapid test lacks reliability in older patients who are less commonly affected and likely to be carriers. First-line treatment is penicillin or amoxicillin, but first-generation cephalosporins, clindamycin, or erythromycin are sufficient if the patient is allergic to penicillins. Prognosis worsens as time between onset and treatment increases, but is overall excellent now with the introduction of antibiotics and improved hygiene.
Scarlet fever is among a list of many common childhood rashes, and it can be difficult to differentiate between these pathologies on clinical presentation. A few notable childhood dermatologic eruptions include erythema infectiosum (fifth disease), roseola (exanthema subitum or sixth disease), and measles. These cases can be distinguished clinically by the age of the patient, distribution, and quality of the symptoms. Laboratory testing may be used to confirm the diagnosis.
Erythema infectiosum is known as fifth disease or slapped-cheek rash because it commonly presents on the cheeks as a pink, maculopapular rash in a reticular pattern. The disease is caused by parvovirus B19 and is accompanied by low fever, malaise, headache, sore throat, and nausea, which precedes the erythematous rash. The facial rash appears first and is followed by patchy eruptions on the extremities. Appearance of the rash typically indicates the patient is no longer contagious, and patients are treated symptomatically with NSAIDs and antihistamines for associated pruritus.
Roseola infantum is commonly caused by human herpesvirus 6 and is usually found in children 3 years and younger. The defining symptom is a high fever, which is paired with a mild cough, runny nose, and diarrhea. A maculopapular rash appears after the fever subsides, starting centrally and spreading outward to the extremities. Although this rash is similar to measles, they can be differentiated by the order of onset. The rash caused by measles begins on the face and mouth (Koplik spots) and moves downward. Additionally, the patient appears generally healthy and the disease is self-limiting in roseola, while patients with measles will appear more ill and require further attention. Measles is caused by the measles virus of the genus Morbillivirus and is highly contagious. It is spread via respiratory route presenting with fever, cough, coryza, and conjunctivitis followed by the rash. Fortunately, the measles vaccine is in widespread use, so cases have declined over the years.
Our patient had a positive strep test. Influenza and coronavirus tests were negative. She was started on daily amoxicillin and the rash resolved within 2 days of taking the antibiotics.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Tampa, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Allmon A et al.. Am Fam Physician. 2015 Aug 1;92(3):211-6.
Moss WJ. Lancet. 2017 Dec 2;390(10111):2490-502.
Mullins TB and Krishnamurthy K. Roseola Infantum, in “StatPearls.” Treasure Islan, Fla.: StatPearls Publishing, 2022.
Pardo S and Perera TB. Scarlet Fever, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2022.
Review gives weight to supplements for hair loss
because of small sample sizes, heterogeneity of hair loss types in study subjects, or other limitations.
The review, published online in JAMA Dermatology, notes that “Twelve of the 20 nutritional interventions had high-quality studies suggesting objectively evaluated effectiveness.”
It is “ground breaking,” in part because of its breadth and depth, said Eva Simmons-O’Brien, MD, a dermatologist in Towson, Md., who often recommends supplements for her patients with hair loss. “It basically kind of vindicates what some of us have been doing for a number of years in terms of treating hair loss,” she told this news organization. “It should hopefully make it more commonplace for dermatologists to consider using nutritional supplements as an adjuvant to treating hair loss,” added Dr. Simmons-O’Brien.
The review “is very helpful,” agreed Lynne J. Goldberg, MD, professor of dermatology and pathology and laboratory medicine at Boston University. Dr. Goldberg noted that many patients are already taking supplements and want to know whether they are safe and effective. The review “points out what the problems are; it talks about what the individual ingredients are and what they do, what the problems are; and it concluded that some people may find these helpful. Which is exactly what I tell my patients,” said Dr. Goldberg, who is also director of the Hair Clinic at Boston Medical Center.
“For patients who are highly motivated and eager to try this, we’re hoping that this systematic review serves as a foundation to have a conversation,” study coauthor Arash Mostaghimi, MD, MPA, MPH, of the department of dermatology at Harvard Medical School, told this news organization. “When there’s medical uncertainty and the question is how much risk is one willing to take, the most important thing to do is to present the data and engage in shared decision-making with the patient,” noted Dr. Mostaghimi, who is also director of the inpatient dermatology consult service at Brigham and Women’s Hospital, Boston.
Surprising effectiveness
Going into the study, “we felt it would be likely that majority of nutritional supplements would either not be effective or not studied,” he said.
Dr. Mostaghimi and his coauthors conducted the study because so many patients take nutritional supplements to address hair loss, he said. An initial literature survey yielded more than 6,300 citations, but after screening and reviews, the authors included 30 articles for evaluation.
The review begins with a look at studies of saw palmetto (Serenoa repens), a botanical compound thought to inhibit the enzyme 5-alpha reductase (5AR), which converts testosterone to dihydroxytestosterone (DHT). DHT is a mediator of androgenic alopecia (AGA). The studies suggest that the compound might stabilize hair loss, “although its effect is likely less than that of finasteride,” write the authors. They also note that side effects associated with finasteride, such as sexual dysfunction, were also observed with saw palmetto “but to a lesser extent.”
For AGA, pumpkin seed oil may also be effective and a “potential alternative” to finasteride for AGA, and Forti5, a nutritional supplement that includes botanical 5AR inhibitors and other ingredients, had favorable effects in one study, the authors write. But neither has been compared to finasteride, and the Forti5 study lacked a control group.
The review also examines the micronutrients vitamin D, zinc, B vitamins, and antioxidants. Low levels of vitamin D have been associated with alopecia areata (AA), AGA, and telogen effluvium (TE) in some studies, and zinc deficiencies have been associated with TE, hair breakage, and thinning, according to the review. A single-arm vitamin D study showed improved results at 6 months for women with TE, but there was no control group and TE is self-resolving, the authors add. Studies in patients with normal zinc levels at baseline who had AA or hair loss showed significant hair regrowth and increased hair thickness and density, but the trials were a mishmash of controls and no controls and relied on self-perceived hair-loss data.
Larger more rigorous studies should be done to evaluate zinc’s effectiveness with AA, the authors comment.
Many patients take vitamin B7 (biotin) for hair loss. It has not been studied on its own but was an ingredient in some supplements in the review. Dr. Simmons-O’Brien said that biotin won’t result in new hair growth but that it can help strengthen the new hairs that grow as a result of other therapies. Both she and the study authors note that the Food and Drug Administration has warned against biotin supplementation because it can interfere with troponin and other test results.
The review also finds that immunomodulators –such as Chinese herbal extracts from paeony and glycyrrhizin – were effective in severe AA. Growth hormone modulators targeting deficiencies in insulin growth factor 1 or growth hormone are also promising. Studies of the modulators capsaicin and isoflavones – used topically – spurred hair growth, the authors write.
Products containing marine protein supplements, including Viviscal and Nourkrin, appeared effective in increasing hair counts in men and women, but the studies were funded by the manufacturer and were not well controlled. Side effects with Viviscal included bloating, according to the review.
The multi-ingredient supplements Nutrafol, Omni-Three, Apple Nutraceutical, and Lambdapil were also included in the review. Only Omni-Three showed no effectiveness, but studies of the other supplements had various limitations, including lack of controls and small sample sizes.
Complicated problem, multiple solutions
Given the many reasons for hair loss, multiple solutions are needed, the dermatologists note.
Dr. Mostaghimi said that he’s still a bit skeptical that supplements work as consistently as described or as well as described, given that he and his coauthors were unable to find any negative studies. In talking with patients who are taking supplements, he said that his first aim is to make sure they are safe. At least the supplements in the review have been studied for safety, he added.
He will encourage replacement of vitamin D or zinc or other vitamins or minerals if patients are deficient but said that he does not “actively encourage supplementation.”
Dr. Simmons-O’Brien said that, when evaluating patients with hair loss, she orders lab tests to determine whether the patient has anemia or a thyroid issue or deficiencies in vitamins or minerals or other nutritional deficiencies, asks about diet and styling practices, and takes a scalp biopsy. It is not uncommon to recommend supplementation on the basis of those findings, she added.
“As a hair-loss specialist, my job is to treat the patient at their level, in their framework, in their comfort zone,” said Dr. Goldberg. Some patients don’t want to take medications for hair loss, so she might recommend supplements in those cases but tells patients that they aren’t well studied.
She added that it can be hard to tell whether a supplement is working, particularly if it has multiple ingredients.
Dr. Mostaghimi reported consulting fees from Pfizer, Concert, Lilly, Hims and Hers, Equillium, AbbVie, Digital Diagnostics, and Bioniz and grants from Pfizer, all outside the submitted work. In addition, Dr. Mostaghimi disclosed that he is an associate editor of JAMA Dermatology but was not involved in any of the decisions regarding the review of the manuscript or its acceptance. No other disclosures were reported by the other study authors. Dr. Goldberg reported no disclosures. Dr. Simmons-O›Brien is a medical consultant for Isdin, but not for hair products.
A version of this article first appeared on Medscape.com.
because of small sample sizes, heterogeneity of hair loss types in study subjects, or other limitations.
The review, published online in JAMA Dermatology, notes that “Twelve of the 20 nutritional interventions had high-quality studies suggesting objectively evaluated effectiveness.”
It is “ground breaking,” in part because of its breadth and depth, said Eva Simmons-O’Brien, MD, a dermatologist in Towson, Md., who often recommends supplements for her patients with hair loss. “It basically kind of vindicates what some of us have been doing for a number of years in terms of treating hair loss,” she told this news organization. “It should hopefully make it more commonplace for dermatologists to consider using nutritional supplements as an adjuvant to treating hair loss,” added Dr. Simmons-O’Brien.
The review “is very helpful,” agreed Lynne J. Goldberg, MD, professor of dermatology and pathology and laboratory medicine at Boston University. Dr. Goldberg noted that many patients are already taking supplements and want to know whether they are safe and effective. The review “points out what the problems are; it talks about what the individual ingredients are and what they do, what the problems are; and it concluded that some people may find these helpful. Which is exactly what I tell my patients,” said Dr. Goldberg, who is also director of the Hair Clinic at Boston Medical Center.
“For patients who are highly motivated and eager to try this, we’re hoping that this systematic review serves as a foundation to have a conversation,” study coauthor Arash Mostaghimi, MD, MPA, MPH, of the department of dermatology at Harvard Medical School, told this news organization. “When there’s medical uncertainty and the question is how much risk is one willing to take, the most important thing to do is to present the data and engage in shared decision-making with the patient,” noted Dr. Mostaghimi, who is also director of the inpatient dermatology consult service at Brigham and Women’s Hospital, Boston.
Surprising effectiveness
Going into the study, “we felt it would be likely that majority of nutritional supplements would either not be effective or not studied,” he said.
Dr. Mostaghimi and his coauthors conducted the study because so many patients take nutritional supplements to address hair loss, he said. An initial literature survey yielded more than 6,300 citations, but after screening and reviews, the authors included 30 articles for evaluation.
The review begins with a look at studies of saw palmetto (Serenoa repens), a botanical compound thought to inhibit the enzyme 5-alpha reductase (5AR), which converts testosterone to dihydroxytestosterone (DHT). DHT is a mediator of androgenic alopecia (AGA). The studies suggest that the compound might stabilize hair loss, “although its effect is likely less than that of finasteride,” write the authors. They also note that side effects associated with finasteride, such as sexual dysfunction, were also observed with saw palmetto “but to a lesser extent.”
For AGA, pumpkin seed oil may also be effective and a “potential alternative” to finasteride for AGA, and Forti5, a nutritional supplement that includes botanical 5AR inhibitors and other ingredients, had favorable effects in one study, the authors write. But neither has been compared to finasteride, and the Forti5 study lacked a control group.
The review also examines the micronutrients vitamin D, zinc, B vitamins, and antioxidants. Low levels of vitamin D have been associated with alopecia areata (AA), AGA, and telogen effluvium (TE) in some studies, and zinc deficiencies have been associated with TE, hair breakage, and thinning, according to the review. A single-arm vitamin D study showed improved results at 6 months for women with TE, but there was no control group and TE is self-resolving, the authors add. Studies in patients with normal zinc levels at baseline who had AA or hair loss showed significant hair regrowth and increased hair thickness and density, but the trials were a mishmash of controls and no controls and relied on self-perceived hair-loss data.
Larger more rigorous studies should be done to evaluate zinc’s effectiveness with AA, the authors comment.
Many patients take vitamin B7 (biotin) for hair loss. It has not been studied on its own but was an ingredient in some supplements in the review. Dr. Simmons-O’Brien said that biotin won’t result in new hair growth but that it can help strengthen the new hairs that grow as a result of other therapies. Both she and the study authors note that the Food and Drug Administration has warned against biotin supplementation because it can interfere with troponin and other test results.
The review also finds that immunomodulators –such as Chinese herbal extracts from paeony and glycyrrhizin – were effective in severe AA. Growth hormone modulators targeting deficiencies in insulin growth factor 1 or growth hormone are also promising. Studies of the modulators capsaicin and isoflavones – used topically – spurred hair growth, the authors write.
Products containing marine protein supplements, including Viviscal and Nourkrin, appeared effective in increasing hair counts in men and women, but the studies were funded by the manufacturer and were not well controlled. Side effects with Viviscal included bloating, according to the review.
The multi-ingredient supplements Nutrafol, Omni-Three, Apple Nutraceutical, and Lambdapil were also included in the review. Only Omni-Three showed no effectiveness, but studies of the other supplements had various limitations, including lack of controls and small sample sizes.
Complicated problem, multiple solutions
Given the many reasons for hair loss, multiple solutions are needed, the dermatologists note.
Dr. Mostaghimi said that he’s still a bit skeptical that supplements work as consistently as described or as well as described, given that he and his coauthors were unable to find any negative studies. In talking with patients who are taking supplements, he said that his first aim is to make sure they are safe. At least the supplements in the review have been studied for safety, he added.
He will encourage replacement of vitamin D or zinc or other vitamins or minerals if patients are deficient but said that he does not “actively encourage supplementation.”
Dr. Simmons-O’Brien said that, when evaluating patients with hair loss, she orders lab tests to determine whether the patient has anemia or a thyroid issue or deficiencies in vitamins or minerals or other nutritional deficiencies, asks about diet and styling practices, and takes a scalp biopsy. It is not uncommon to recommend supplementation on the basis of those findings, she added.
“As a hair-loss specialist, my job is to treat the patient at their level, in their framework, in their comfort zone,” said Dr. Goldberg. Some patients don’t want to take medications for hair loss, so she might recommend supplements in those cases but tells patients that they aren’t well studied.
She added that it can be hard to tell whether a supplement is working, particularly if it has multiple ingredients.
Dr. Mostaghimi reported consulting fees from Pfizer, Concert, Lilly, Hims and Hers, Equillium, AbbVie, Digital Diagnostics, and Bioniz and grants from Pfizer, all outside the submitted work. In addition, Dr. Mostaghimi disclosed that he is an associate editor of JAMA Dermatology but was not involved in any of the decisions regarding the review of the manuscript or its acceptance. No other disclosures were reported by the other study authors. Dr. Goldberg reported no disclosures. Dr. Simmons-O›Brien is a medical consultant for Isdin, but not for hair products.
A version of this article first appeared on Medscape.com.
because of small sample sizes, heterogeneity of hair loss types in study subjects, or other limitations.
The review, published online in JAMA Dermatology, notes that “Twelve of the 20 nutritional interventions had high-quality studies suggesting objectively evaluated effectiveness.”
It is “ground breaking,” in part because of its breadth and depth, said Eva Simmons-O’Brien, MD, a dermatologist in Towson, Md., who often recommends supplements for her patients with hair loss. “It basically kind of vindicates what some of us have been doing for a number of years in terms of treating hair loss,” she told this news organization. “It should hopefully make it more commonplace for dermatologists to consider using nutritional supplements as an adjuvant to treating hair loss,” added Dr. Simmons-O’Brien.
The review “is very helpful,” agreed Lynne J. Goldberg, MD, professor of dermatology and pathology and laboratory medicine at Boston University. Dr. Goldberg noted that many patients are already taking supplements and want to know whether they are safe and effective. The review “points out what the problems are; it talks about what the individual ingredients are and what they do, what the problems are; and it concluded that some people may find these helpful. Which is exactly what I tell my patients,” said Dr. Goldberg, who is also director of the Hair Clinic at Boston Medical Center.
“For patients who are highly motivated and eager to try this, we’re hoping that this systematic review serves as a foundation to have a conversation,” study coauthor Arash Mostaghimi, MD, MPA, MPH, of the department of dermatology at Harvard Medical School, told this news organization. “When there’s medical uncertainty and the question is how much risk is one willing to take, the most important thing to do is to present the data and engage in shared decision-making with the patient,” noted Dr. Mostaghimi, who is also director of the inpatient dermatology consult service at Brigham and Women’s Hospital, Boston.
Surprising effectiveness
Going into the study, “we felt it would be likely that majority of nutritional supplements would either not be effective or not studied,” he said.
Dr. Mostaghimi and his coauthors conducted the study because so many patients take nutritional supplements to address hair loss, he said. An initial literature survey yielded more than 6,300 citations, but after screening and reviews, the authors included 30 articles for evaluation.
The review begins with a look at studies of saw palmetto (Serenoa repens), a botanical compound thought to inhibit the enzyme 5-alpha reductase (5AR), which converts testosterone to dihydroxytestosterone (DHT). DHT is a mediator of androgenic alopecia (AGA). The studies suggest that the compound might stabilize hair loss, “although its effect is likely less than that of finasteride,” write the authors. They also note that side effects associated with finasteride, such as sexual dysfunction, were also observed with saw palmetto “but to a lesser extent.”
For AGA, pumpkin seed oil may also be effective and a “potential alternative” to finasteride for AGA, and Forti5, a nutritional supplement that includes botanical 5AR inhibitors and other ingredients, had favorable effects in one study, the authors write. But neither has been compared to finasteride, and the Forti5 study lacked a control group.
The review also examines the micronutrients vitamin D, zinc, B vitamins, and antioxidants. Low levels of vitamin D have been associated with alopecia areata (AA), AGA, and telogen effluvium (TE) in some studies, and zinc deficiencies have been associated with TE, hair breakage, and thinning, according to the review. A single-arm vitamin D study showed improved results at 6 months for women with TE, but there was no control group and TE is self-resolving, the authors add. Studies in patients with normal zinc levels at baseline who had AA or hair loss showed significant hair regrowth and increased hair thickness and density, but the trials were a mishmash of controls and no controls and relied on self-perceived hair-loss data.
Larger more rigorous studies should be done to evaluate zinc’s effectiveness with AA, the authors comment.
Many patients take vitamin B7 (biotin) for hair loss. It has not been studied on its own but was an ingredient in some supplements in the review. Dr. Simmons-O’Brien said that biotin won’t result in new hair growth but that it can help strengthen the new hairs that grow as a result of other therapies. Both she and the study authors note that the Food and Drug Administration has warned against biotin supplementation because it can interfere with troponin and other test results.
The review also finds that immunomodulators –such as Chinese herbal extracts from paeony and glycyrrhizin – were effective in severe AA. Growth hormone modulators targeting deficiencies in insulin growth factor 1 or growth hormone are also promising. Studies of the modulators capsaicin and isoflavones – used topically – spurred hair growth, the authors write.
Products containing marine protein supplements, including Viviscal and Nourkrin, appeared effective in increasing hair counts in men and women, but the studies were funded by the manufacturer and were not well controlled. Side effects with Viviscal included bloating, according to the review.
The multi-ingredient supplements Nutrafol, Omni-Three, Apple Nutraceutical, and Lambdapil were also included in the review. Only Omni-Three showed no effectiveness, but studies of the other supplements had various limitations, including lack of controls and small sample sizes.
Complicated problem, multiple solutions
Given the many reasons for hair loss, multiple solutions are needed, the dermatologists note.
Dr. Mostaghimi said that he’s still a bit skeptical that supplements work as consistently as described or as well as described, given that he and his coauthors were unable to find any negative studies. In talking with patients who are taking supplements, he said that his first aim is to make sure they are safe. At least the supplements in the review have been studied for safety, he added.
He will encourage replacement of vitamin D or zinc or other vitamins or minerals if patients are deficient but said that he does not “actively encourage supplementation.”
Dr. Simmons-O’Brien said that, when evaluating patients with hair loss, she orders lab tests to determine whether the patient has anemia or a thyroid issue or deficiencies in vitamins or minerals or other nutritional deficiencies, asks about diet and styling practices, and takes a scalp biopsy. It is not uncommon to recommend supplementation on the basis of those findings, she added.
“As a hair-loss specialist, my job is to treat the patient at their level, in their framework, in their comfort zone,” said Dr. Goldberg. Some patients don’t want to take medications for hair loss, so she might recommend supplements in those cases but tells patients that they aren’t well studied.
She added that it can be hard to tell whether a supplement is working, particularly if it has multiple ingredients.
Dr. Mostaghimi reported consulting fees from Pfizer, Concert, Lilly, Hims and Hers, Equillium, AbbVie, Digital Diagnostics, and Bioniz and grants from Pfizer, all outside the submitted work. In addition, Dr. Mostaghimi disclosed that he is an associate editor of JAMA Dermatology but was not involved in any of the decisions regarding the review of the manuscript or its acceptance. No other disclosures were reported by the other study authors. Dr. Goldberg reported no disclosures. Dr. Simmons-O›Brien is a medical consultant for Isdin, but not for hair products.
A version of this article first appeared on Medscape.com.
Erythrasma
THE COMPARISON
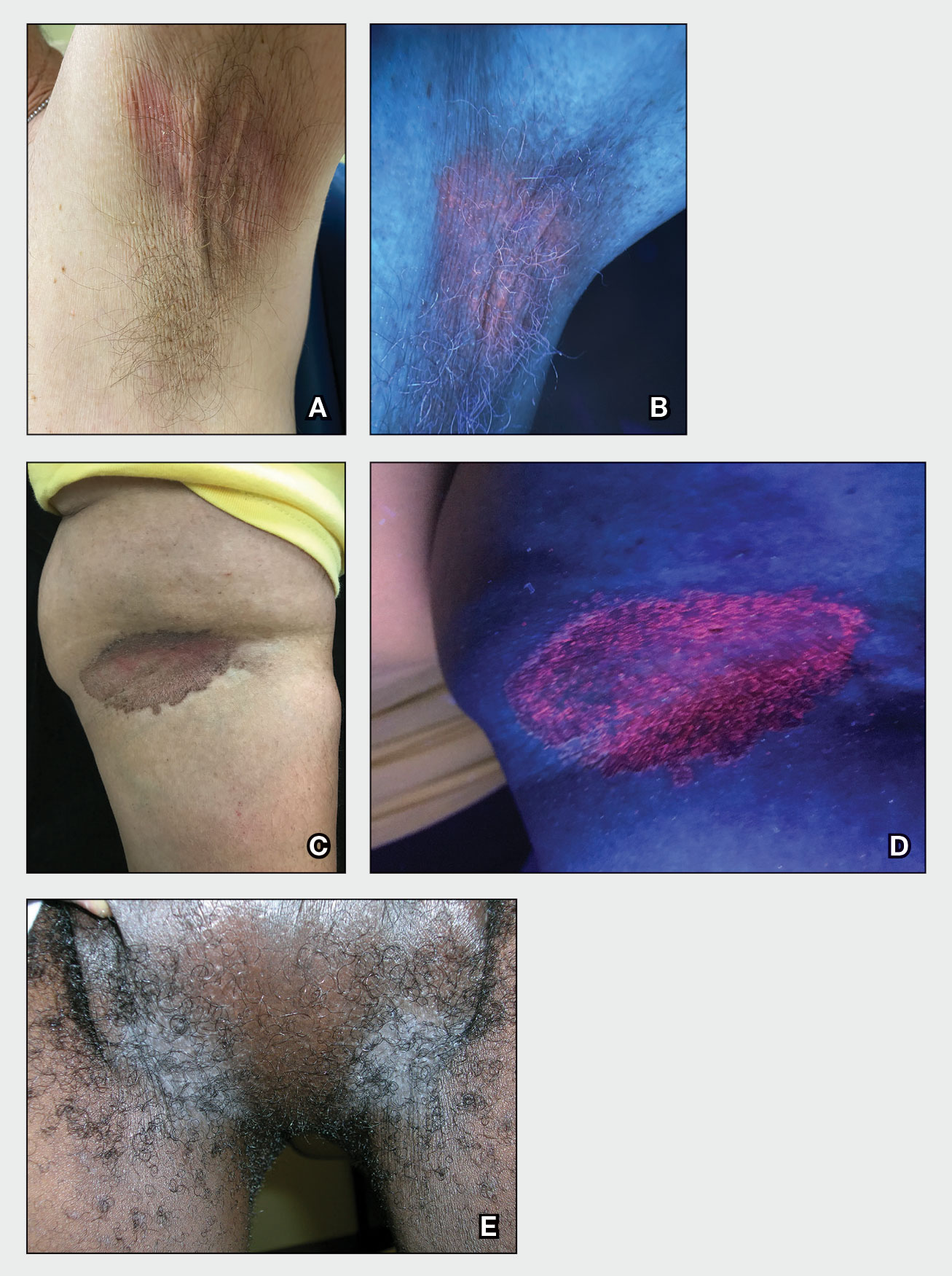
A and B Axilla of a 65-year-old White man with erythrasma showing a well-demarcated erythematous plaque with fine scale (A). Wood lamp examination of the area showed characteristic bright coral red fluorescence (B).
C and D A well-demarcated, red-brown plaque with fine scale in the antecubital fossa of an obese Hispanic woman (C). Wood lamp examination revealed bright coral red fluorescence (D).
E Hypopigmented patches in the groin with pruritus in a Black man. He also had erythrasma between the toes.
Erythrasma is a skin condition caused by acute or chronic infection of the outermost layer of the epidermis (stratum corneum) with Corynebacterium minutissimum. It has a predilection for intertriginous regions such as the axillae, groin, and interdigital spaces of the toes. It can be associated with pruritus or can be asymptomatic.
Epidemiology
Erythrasma typically affects adults, with greater prevalence among those residing in shared living facilities, such as dormitories or nursing homes, or in humid climates.1 It is a common disorder with an estimated prevalence of 17.6% of bacterial skin infections in elderly patients and 44% of diabetic interdigital toe space infections.2,3
Key clinical features
Erythrasma can manifest as red-brown hyperpigmented plaques with fine scale and little central clearing (Figures A and C) or as a hypopigmented patch (Figure E) with a sharply marginated, hyperpigmented border in patients with skin of color. In the interdigital toe spaces, the skin often is white and macerated. These findings may appear in patients of all skin tones.
Worth noting
• Corynebacterium minutissimum produces coproporphyrin III, which glows fluorescent red under Wood lamp examination (Figures B and D). A recent shower or bath may remove the fluorescent coproporphyrins and cause a false-negative result. The interdigital space between the fourth and fifth toes is a common location for C minutissimum; thus clinicians should consider examining these areas with a Wood lamp.
• Associated risk factors include obesity, immunosuppression, diabetes mellitus, and excessive sweating.1
• The differential diagnosis includes intertrigo, inverse psoriasis, confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome), acanthosis nigricans, seborrheic dermatitis, and tinea pedis when present in the interdigital toe spaces. Plaques occurring in circular patterns may be mistaken for tinea corporis or pityriasis rotunda.
• There is a high prevalence of erythrasma in patients with inverse psoriasis, and it may exacerbate psoriatic plaques.4
• Treatment options include application of topical clindamycin or erythromycin to the affected area.1 Some patients have responded to topical mupiricin.2 For larger areas, a 1-g dose of clarithromycin5 or a 14-day course of erythromycin may be appropriate.1 Avoid prescribing clarithromycin to patients with preexisting heart disease due to its increased risk for cardiac events or death; consider other agents.
Health disparity highlight
Obesity, most prevalent in non-Hispanic Black adults (49.9%) and Hispanic adults (45.6%) followed by non- Hispanic White adults (41.4%),6 may cause velvety dark plaques on the neck called acanthosis nigricans. However, acute or chronic erythrasma also may cause hyperpigmentation of the body folds. Although the pathology of erythrasma is due to bacterial infection of the superficial layer of the stratum corneum, acanthosis nigricans is due to fibroblast proliferation and stimulation of epidermal keratinocytes likely from increased growth factors and insulinlike growth factor.7 If erythrasma is mistaken for acanthosis nigricans, the patient may be counseled inappropriately that the hyperpigmentation is something not easily resolved and subsequently left with an active treatable condition that adversely affects their quality of life.
- Groves JB, Nassereddin A, Freeman AM. Erythrasma. In: StatPearls. StatPearls Publishing; August 11, 2021. Accessed November 17, 2022. https://www.ncbi.nlm.nih.gov/books/NBK513352/
- Forouzan P, Cohen PR. Erythrasma revisited: diagnosis, differential diagnoses, and comprehensive review of treatment [published online September 30, 2020]. Cureus. 2020;12:E10733. doi:10.7759/cureus.10733
- Polat M, I˙lhan MN. Dermatological complaints of the elderly attending a dermatology outpatient clinic in Turkey: a prospective study over a one-year period. Acta Dermatovenerol Croat. 2015;23:277-281.
- Janeczek M, Kozel Z, Bhasin R, et al. High prevalence of erythrasma in patients with inverse psoriasis: a cross-sectional study. J Clin Aesthet Dermatol. 2020;13:12-14.
- Khan MJ. Interdigital pedal erythrasma treated with one-time dose of oral clarithromycin 1 g: two case reports [published online February 6, 2020]. Clin Case Rep. 2020;8:672-674. doi:10.1002/ccr3.2712
- Stierman B, Afful J, Carroll M, et al. National Health and Nutrition Examination Survey 2017–March 2020 Prepandemic Data Files Development of Files and Prevalence Estimates for Selected Health Outcomes. National Health Statistics Reports. Published June 14, 2021. Accessed November 17, 2022. https://stacks.cdc.gov/view/cdc/106273
- Brady MF, Rawla P. Acanthosis nigricans. In: StatPearls [Internet]. StatPearls Publishing; 2022. Updated October 9, 2022. Accessed November 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK431057
THE COMPARISON
A and B Axilla of a 65-year-old White man with erythrasma showing a well-demarcated erythematous plaque with fine scale (A). Wood lamp examination of the area showed characteristic bright coral red fluorescence (B).
C and D A well-demarcated, red-brown plaque with fine scale in the antecubital fossa of an obese Hispanic woman (C). Wood lamp examination revealed bright coral red fluorescence (D).
E Hypopigmented patches in the groin with pruritus in a Black man. He also had erythrasma between the toes.
Erythrasma is a skin condition caused by acute or chronic infection of the outermost layer of the epidermis (stratum corneum) with Corynebacterium minutissimum. It has a predilection for intertriginous regions such as the axillae, groin, and interdigital spaces of the toes. It can be associated with pruritus or can be asymptomatic.

Epidemiology
Erythrasma typically affects adults, with greater prevalence among those residing in shared living facilities, such as dormitories or nursing homes, or in humid climates.1 It is a common disorder with an estimated prevalence of 17.6% of bacterial skin infections in elderly patients and 44% of diabetic interdigital toe space infections.2,3
Key clinical features
Erythrasma can manifest as red-brown hyperpigmented plaques with fine scale and little central clearing (Figures A and C) or as a hypopigmented patch (Figure E) with a sharply marginated, hyperpigmented border in patients with skin of color. In the interdigital toe spaces, the skin often is white and macerated. These findings may appear in patients of all skin tones.
Worth noting
• Corynebacterium minutissimum produces coproporphyrin III, which glows fluorescent red under Wood lamp examination (Figures B and D). A recent shower or bath may remove the fluorescent coproporphyrins and cause a false-negative result. The interdigital space between the fourth and fifth toes is a common location for C minutissimum; thus clinicians should consider examining these areas with a Wood lamp.
• Associated risk factors include obesity, immunosuppression, diabetes mellitus, and excessive sweating.1
• The differential diagnosis includes intertrigo, inverse psoriasis, confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome), acanthosis nigricans, seborrheic dermatitis, and tinea pedis when present in the interdigital toe spaces. Plaques occurring in circular patterns may be mistaken for tinea corporis or pityriasis rotunda.
• There is a high prevalence of erythrasma in patients with inverse psoriasis, and it may exacerbate psoriatic plaques.4
• Treatment options include application of topical clindamycin or erythromycin to the affected area.1 Some patients have responded to topical mupiricin.2 For larger areas, a 1-g dose of clarithromycin5 or a 14-day course of erythromycin may be appropriate.1 Avoid prescribing clarithromycin to patients with preexisting heart disease due to its increased risk for cardiac events or death; consider other agents.
Health disparity highlight
Obesity, most prevalent in non-Hispanic Black adults (49.9%) and Hispanic adults (45.6%) followed by non- Hispanic White adults (41.4%),6 may cause velvety dark plaques on the neck called acanthosis nigricans. However, acute or chronic erythrasma also may cause hyperpigmentation of the body folds. Although the pathology of erythrasma is due to bacterial infection of the superficial layer of the stratum corneum, acanthosis nigricans is due to fibroblast proliferation and stimulation of epidermal keratinocytes likely from increased growth factors and insulinlike growth factor.7 If erythrasma is mistaken for acanthosis nigricans, the patient may be counseled inappropriately that the hyperpigmentation is something not easily resolved and subsequently left with an active treatable condition that adversely affects their quality of life.
THE COMPARISON
A and B Axilla of a 65-year-old White man with erythrasma showing a well-demarcated erythematous plaque with fine scale (A). Wood lamp examination of the area showed characteristic bright coral red fluorescence (B).
C and D A well-demarcated, red-brown plaque with fine scale in the antecubital fossa of an obese Hispanic woman (C). Wood lamp examination revealed bright coral red fluorescence (D).
E Hypopigmented patches in the groin with pruritus in a Black man. He also had erythrasma between the toes.
Erythrasma is a skin condition caused by acute or chronic infection of the outermost layer of the epidermis (stratum corneum) with Corynebacterium minutissimum. It has a predilection for intertriginous regions such as the axillae, groin, and interdigital spaces of the toes. It can be associated with pruritus or can be asymptomatic.

Epidemiology
Erythrasma typically affects adults, with greater prevalence among those residing in shared living facilities, such as dormitories or nursing homes, or in humid climates.1 It is a common disorder with an estimated prevalence of 17.6% of bacterial skin infections in elderly patients and 44% of diabetic interdigital toe space infections.2,3
Key clinical features
Erythrasma can manifest as red-brown hyperpigmented plaques with fine scale and little central clearing (Figures A and C) or as a hypopigmented patch (Figure E) with a sharply marginated, hyperpigmented border in patients with skin of color. In the interdigital toe spaces, the skin often is white and macerated. These findings may appear in patients of all skin tones.
Worth noting
• Corynebacterium minutissimum produces coproporphyrin III, which glows fluorescent red under Wood lamp examination (Figures B and D). A recent shower or bath may remove the fluorescent coproporphyrins and cause a false-negative result. The interdigital space between the fourth and fifth toes is a common location for C minutissimum; thus clinicians should consider examining these areas with a Wood lamp.
• Associated risk factors include obesity, immunosuppression, diabetes mellitus, and excessive sweating.1
• The differential diagnosis includes intertrigo, inverse psoriasis, confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome), acanthosis nigricans, seborrheic dermatitis, and tinea pedis when present in the interdigital toe spaces. Plaques occurring in circular patterns may be mistaken for tinea corporis or pityriasis rotunda.
• There is a high prevalence of erythrasma in patients with inverse psoriasis, and it may exacerbate psoriatic plaques.4
• Treatment options include application of topical clindamycin or erythromycin to the affected area.1 Some patients have responded to topical mupiricin.2 For larger areas, a 1-g dose of clarithromycin5 or a 14-day course of erythromycin may be appropriate.1 Avoid prescribing clarithromycin to patients with preexisting heart disease due to its increased risk for cardiac events or death; consider other agents.
Health disparity highlight
Obesity, most prevalent in non-Hispanic Black adults (49.9%) and Hispanic adults (45.6%) followed by non- Hispanic White adults (41.4%),6 may cause velvety dark plaques on the neck called acanthosis nigricans. However, acute or chronic erythrasma also may cause hyperpigmentation of the body folds. Although the pathology of erythrasma is due to bacterial infection of the superficial layer of the stratum corneum, acanthosis nigricans is due to fibroblast proliferation and stimulation of epidermal keratinocytes likely from increased growth factors and insulinlike growth factor.7 If erythrasma is mistaken for acanthosis nigricans, the patient may be counseled inappropriately that the hyperpigmentation is something not easily resolved and subsequently left with an active treatable condition that adversely affects their quality of life.
- Groves JB, Nassereddin A, Freeman AM. Erythrasma. In: StatPearls. StatPearls Publishing; August 11, 2021. Accessed November 17, 2022. https://www.ncbi.nlm.nih.gov/books/NBK513352/
- Forouzan P, Cohen PR. Erythrasma revisited: diagnosis, differential diagnoses, and comprehensive review of treatment [published online September 30, 2020]. Cureus. 2020;12:E10733. doi:10.7759/cureus.10733
- Polat M, I˙lhan MN. Dermatological complaints of the elderly attending a dermatology outpatient clinic in Turkey: a prospective study over a one-year period. Acta Dermatovenerol Croat. 2015;23:277-281.
- Janeczek M, Kozel Z, Bhasin R, et al. High prevalence of erythrasma in patients with inverse psoriasis: a cross-sectional study. J Clin Aesthet Dermatol. 2020;13:12-14.
- Khan MJ. Interdigital pedal erythrasma treated with one-time dose of oral clarithromycin 1 g: two case reports [published online February 6, 2020]. Clin Case Rep. 2020;8:672-674. doi:10.1002/ccr3.2712
- Stierman B, Afful J, Carroll M, et al. National Health and Nutrition Examination Survey 2017–March 2020 Prepandemic Data Files Development of Files and Prevalence Estimates for Selected Health Outcomes. National Health Statistics Reports. Published June 14, 2021. Accessed November 17, 2022. https://stacks.cdc.gov/view/cdc/106273
- Brady MF, Rawla P. Acanthosis nigricans. In: StatPearls [Internet]. StatPearls Publishing; 2022. Updated October 9, 2022. Accessed November 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK431057
- Groves JB, Nassereddin A, Freeman AM. Erythrasma. In: StatPearls. StatPearls Publishing; August 11, 2021. Accessed November 17, 2022. https://www.ncbi.nlm.nih.gov/books/NBK513352/
- Forouzan P, Cohen PR. Erythrasma revisited: diagnosis, differential diagnoses, and comprehensive review of treatment [published online September 30, 2020]. Cureus. 2020;12:E10733. doi:10.7759/cureus.10733
- Polat M, I˙lhan MN. Dermatological complaints of the elderly attending a dermatology outpatient clinic in Turkey: a prospective study over a one-year period. Acta Dermatovenerol Croat. 2015;23:277-281.
- Janeczek M, Kozel Z, Bhasin R, et al. High prevalence of erythrasma in patients with inverse psoriasis: a cross-sectional study. J Clin Aesthet Dermatol. 2020;13:12-14.
- Khan MJ. Interdigital pedal erythrasma treated with one-time dose of oral clarithromycin 1 g: two case reports [published online February 6, 2020]. Clin Case Rep. 2020;8:672-674. doi:10.1002/ccr3.2712
- Stierman B, Afful J, Carroll M, et al. National Health and Nutrition Examination Survey 2017–March 2020 Prepandemic Data Files Development of Files and Prevalence Estimates for Selected Health Outcomes. National Health Statistics Reports. Published June 14, 2021. Accessed November 17, 2022. https://stacks.cdc.gov/view/cdc/106273
- Brady MF, Rawla P. Acanthosis nigricans. In: StatPearls [Internet]. StatPearls Publishing; 2022. Updated October 9, 2022. Accessed November 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK431057
Mobile Enlarging Scalp Nodule
The Diagnosis: Hybrid Schwannoma-Perineurioma
Hybrid nerve sheath tumors are rare entities that display features of more than one nerve sheath tumor such as neurofibromas, schwannomas, and perineuriomas.1 These tumors often are found in the dermis or subcutaneous tissue of the extremities and abdomen2; however, cases of hybrid peripheral nerve sheath tumors have been reported in many anatomical locations without a gender predilection.3 The most common type of hybrid nerve sheath tumor is a schwannoma-perineurioma.3,4 Histologically, they are well-circumscribed lesions composed of both spindled Schwann cells with plump nuclei and spindled perineural cells with more elongated thin nuclei.5 Although the Schwann cell component tends to predominate, the 2 cell populations interdigitate, making it challenging to definitively distinguish them by hematoxylin and eosin staining alone.4 However, immunohistochemical (IHC) staining can be used to help distinguish the 2 separate cell populations. Staining for S-100 and SRY-box transcription factor 10 (SOX-10) will be positive in the Schwann cell component, and staining for epithelial membrane antigen, Claudin-1, or glucose transporter-1 (Figure 1) will be positive in the perineural component. Other hybrid forms of benign nerve sheath tumors include neurofibroma-schwannoma and neurofibromaperineurioma.4 Neurofibroma-schwannomas usually have a schwannoma component containing Antoni A areas with palisading Verocay bodies. The neurofibroma cells typically have wavy elongated nuclei, fibroblasts, and mucinous myxoid material.3 Neurofibroma-perineurioma is the least common hybrid tumor. These hybrid tumors have a plexiform neurofibroma appearance with areas of perineural differentiation, which can be difficult to identify on routine histology and typically will require IHC staining to appreciate. The neurofibroma component will stain positive for S-100 and negative for markers of perineural differentiation, including epithelial membrane antigen, glucose transporter-1, and Claudin-1.3 Although schwannoma-perineuriomas are benign sporadic tumors not associated with neurofibromatosis, neurofibromaschwannomas are associated with neurofibromatosis types 1 and 2 (NF1 and NF2). Neurofibroma-perineurioma tumors usually are associated with only NF1.3,6
Schwannomas typically present in middle-aged patients as tumors located on flexor surfaces.7 Although perineural cells can be seen at the periphery of a schwannoma forming a capsule, they do not interdigitate between the Schwann cells. Schwannomas are composed almost entirely of well-differentiated Schwann cells.1,4,8 Schwannomas classically are well-circumscribed, encapsulated, biphasic lesions with alternating compact areas (Antoni A) and loosely arranged areas (Antoni B). The spindled cells occasionally may display nuclear palisading within the Antoni A areas, known as Verocay bodies (Figure 2). Antoni B areas are more disorganized and hypocellular with variable macrophage infiltrate.1,4,8 The Schwann cells predominantly will have bland cytologic features, but scattered areas of degenerative nuclear atypia (also known as ancient change) may be present.4 Multiple schwannomas are associated with NF2 gene mutations and loss of merlin protein.8 There are different subtypes of schwannomas, including cellular and plexiform schwannomas.4 Because schwannomas are benign nerve sheath lesions, treatment typically consists of excision with careful dissection around the involved nerve.9
Neurofibromas are the most common peripheral nerve sheath tumors of the skin with no notable anatomic prediction, though one study found them to be more prevalent in the upper extremities.10 They typically present as sporadic solitary lesions, but multiple lesions may appear as superficial pedunculated growths that present in those aged 20 to 30 years.11 Microscopically, neurofibromas typically are not well circumscribed and have an infiltrative growth pattern. Neurofibromas are composed of cytologically bland spindled Schwann cells with thin wavy nuclei in a variable myxoid stroma (Figure 3). In addition to Schwann cells, neurofibromas contain other cell components, including fibroblasts, mast cells, perineurial-like cells, and residual axons.4 Neurofibromas typically are located in the dermis but may extend into the subcutaneous tissue. Clinically, the overlying skin may show hyperpigmentation.8 Neurofibromas can be localized, diffuse, or plexiform, with the majority being localized. Diffuse neurofibromas clinically have a raised plaque appearance. Treatment is unnecessary because these lesions are benign.7
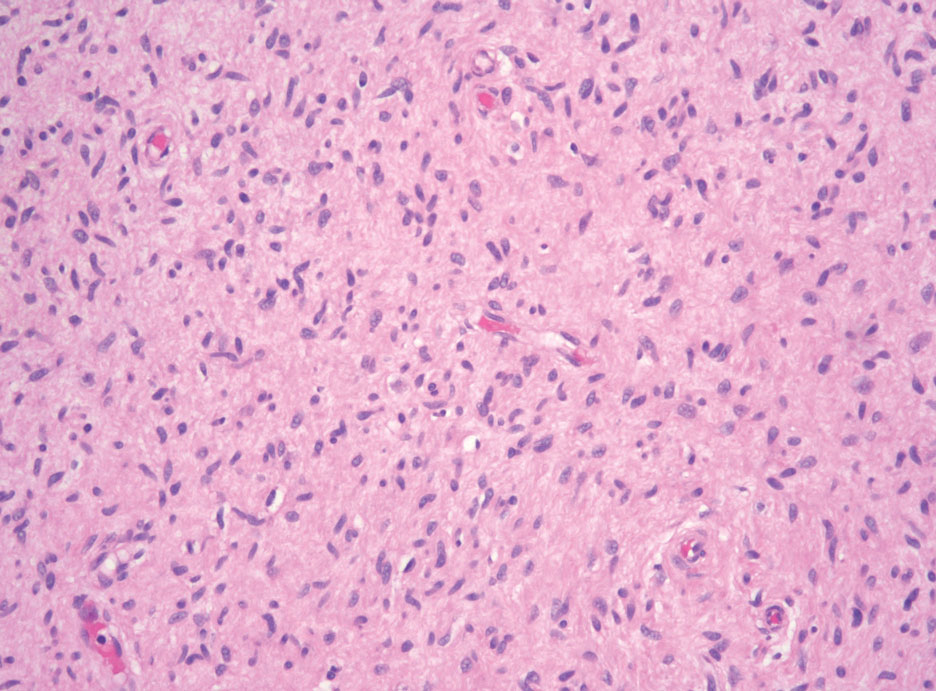
Desmoplastic melanoma (DM) is another diagnosis in the differential for this case. Patients with DM are older compared to non-DM melanoma patients, with a male predilection.12 Desmoplastic melanomas are more likely to be located on the head and neck. In approximately one-third of cases, no in situ component will be identified, leading to confusion of the dermal lesion as a neural lesion or an area of scar formation. Microscopically, DM presents as a variable cellular infiltrative tumor composed of spindle cells with varying degrees of nuclear atypia. The spindled melanocytes are within a collagenous (desmoplastic) stroma (Figure 4).13 Desmoplastic melanoma has been described with a low mitotic index, leading to misdiagnosis with benign spindle cell neoplasms.14 The spindle cells should be positive for S-100 and SOX-10 with IHC staining. Unlike other melanomas, human melanoma black 45 and Melan-A often are negative or only focally positive. Treatment of DM is similar to non-DM in that wide local excision usually is employed. A systematic review evaluating sentinel lymph node biopsy (SLNB) recommended consideration of SLNB in mixed DM but not for pure DM, as rates of positive SLNB were much lower in the latter.15
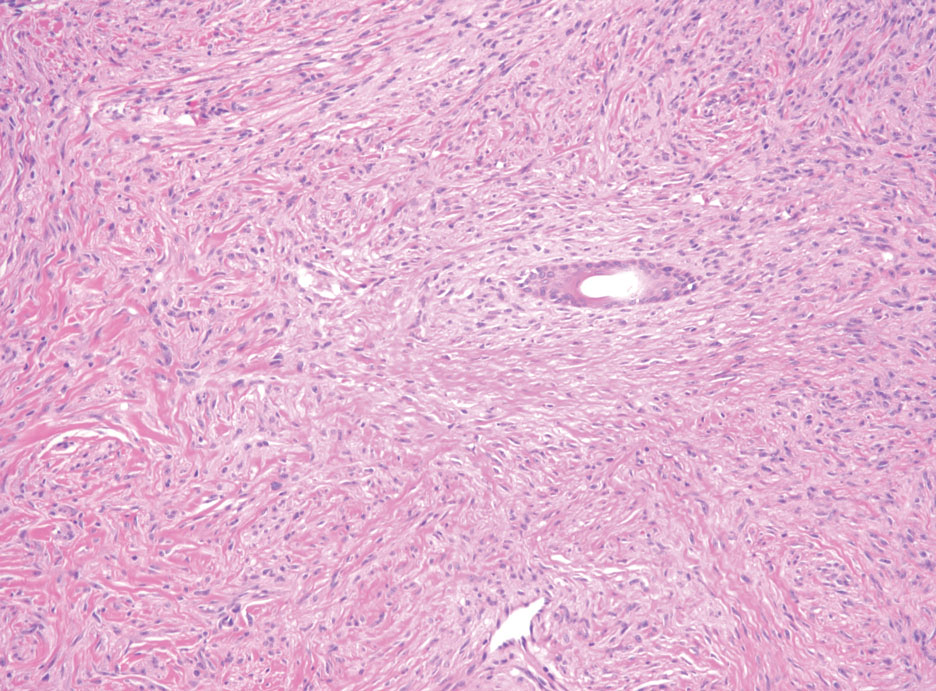
Patients with malignant peripheral nerve sheath tumor (MPNST) usually present with an enlarging mass, pain, or neurologic symptoms. Most cases of MPNST are located on the trunk or extremities.16 Plexiform neurofibromas, especially in adults with NF1, have the potential to transform into an MPNST.4 In fact, MPNST is the most common malignancy in patients with NF1.17 Pediatric cancer survivors also are predisposed to MPNST, with a 40-fold increase in incidence compared to the general population.18 Transformation from schwannoma to MPNST is rare but has been reported.8 Histologically, spindle cells easily can be appreciated with a fasciculated growth pattern (Figure 5). Mitotic activity and tumor necrosis may be present. Diagnosis of these tumors historically has been challenging, though recent research has identified inactivation of polycomb repressive complex 2 in 70% to 90% of MPNSTs. Because of polycomb repressive complex 2 inactivation, there is loss of stone H3K27 trimethylation that can be capitalized on for MPNST diagnosis.19 Negative IHC staining for H3K27 trimethylation has been found to be highly specific for MPNST. Negative staining for different cytokeratin and melanoma markers can be helpful in differentiating it from carcinomas and melanoma. The only curative treatment for MPNST is complete excision, leaving patients with recurrent, refractory, and metastatic cases to be encouraged for enrollment in clinical trials. The 5-year survival rates for patients with MPNST reported in the literature range from 20% to 50%.20
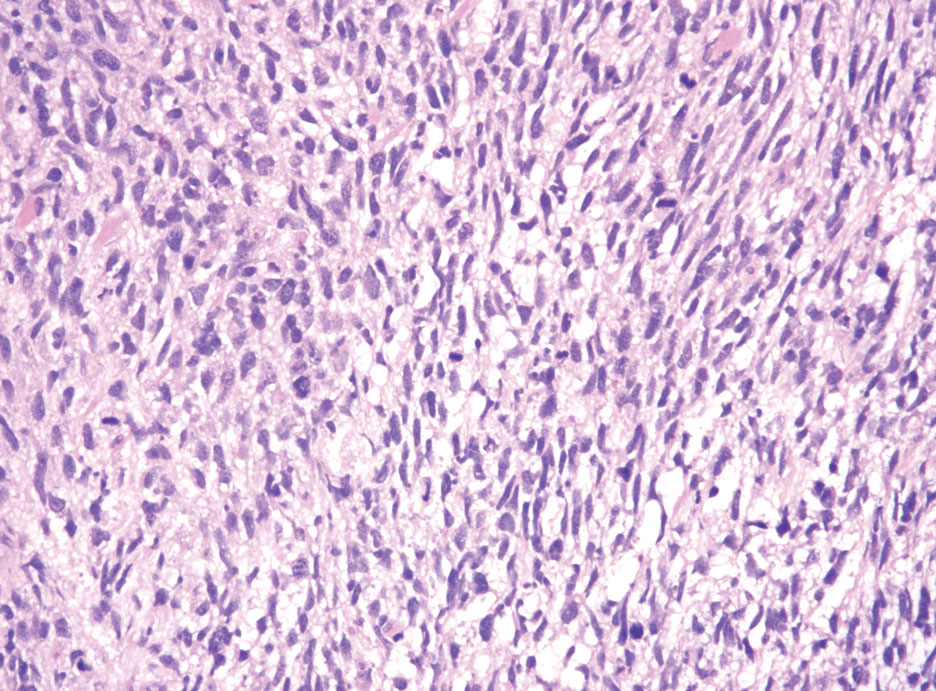
- Hornick JL, Bundock EA, Fletcher CD. Hybrid schwannoma /perineurioma: clinicopathologic analysis of 42 distinctive benign nerve sheath tumors. Am J Surg Pathol. 2009;33:1554-1561.
- Leung KCP, Chan E, Ng HYJ, et al. Novel case of hybrid perineuriomaneurofibroma of the orbit. Can J Ophthalmol. 2019;54:E283-E285.
- Ud Din N, Ahmad Z, Abdul-Ghafar J, et al. Hybrid peripheral nerve sheath tumors: report of five cases and detailed review of literature. BMC Cancer. 2017;17:349. doi:10.1186/s12885-017-3350-1
- Belakhoua SM, Rodriguez FJ. Diagnostic pathology of tumors of peripheral nerve. Neurosurgery. 2021;88:443-456.
- Michal M, Kazakov DV, Michal M. Hybrid peripheral nerve sheath tumors: a review. Cesk Patol. 2017;53:81-88.
- Harder A, Wesemann M, Hagel C, et al. Hybrid neurofibroma /schwannoma is overrepresented among schwannomatosis and neurofibromatosis patients. Am J Surg Pathol. 2012;36:702-709.
- Bhattacharyya AK, Perrin R, Guha A. Peripheral nerve tumors: management strategies and molecular insights. J Neurooncol. 2004;69:335-349.
- Pytel P, Anthony DC. Peripheral nerves and skeletal muscle. In: Kumar V, Abbas AK, Aster JC, eds. Robbins and Cotran Pathologic Basis of Disease. 10th ed. Elsevier/Saunders; 2015:1218-1239.
- Strike SA, Puhaindran ME. Nerve tumors of the upper extremity. Clin Plast Surg. 2019;46:347-350.
- Kim DH, Murovic JA, Tiel RL, et al. A series of 397 peripheral neural sheath tumors: 30-year experience at Louisiana State University Health Sciences Center. J Neurosurg. 2005;102:246-255.
- Pilavaki M, Chourmouzi D, Kiziridou A, et al. Imaging of peripheral nerve sheath tumors with pathologic correlation: pictorial review. Eur J Radiol. 2004;52:229-239.
- Murali R, Shaw HM, Lai K, et al. Prognostic factors in cutaneous desmoplastic melanoma: a study of 252 patients. Cancer. 2010; 116:4130-4138.
- Chen LL, Jaimes N, Barker CA, et al. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68:825-833.
- de Almeida LS, Requena L, Rutten A, et al. Desmoplastic malignant melanoma: a clinicopathologic analysis of 113 cases. Am J Dermatopathol. 2008;30:207-215.
- Dunne JA, Wormald JC, Steele J, et al. Is sentinel lymph node biopsy warranted for desmoplastic melanoma? a systematic review. J Plast Reconstr Aesthet Surg. 2017;70:274-280.
- Patel TD, Shaigany K, Fang CH, et al. Comparative analysis of head and neck and non-head and neck malignant peripheral nerve sheath tumors. Otolaryngol Head Neck Surg. 2016;154:113-120.
- Prudner BC, Ball T, Rathore R, et al. Diagnosis and management of malignant peripheral nerve sheath tumors: current practice and future perspectives. Neurooncol Adv. 2020;2(suppl 1):I40-I9.
- Bright CJ, Hawkins MM, Winter DL, et al. Risk of soft-tissue sarcoma among 69,460 five-year survivors of childhood cancer in Europe. J Natl Cancer Inst. 2018;110:649-660.
- Schaefer I-M, Fletcher CD, Hornick JL. Loss of H3K27 trimethylation distinguishes malignant peripheral nerve sheath tumors from histologic mimics. Mod Pathol. 2016;29:4-13.
- Kolberg M, Holand M, Agesen TH, et al. Survival meta-analyses for >1800 malignant peripheral nerve sheath tumor patients with and without neurofibromatosis type 1. Neuro Oncol. 2013;15:135-147.
The Diagnosis: Hybrid Schwannoma-Perineurioma
Hybrid nerve sheath tumors are rare entities that display features of more than one nerve sheath tumor such as neurofibromas, schwannomas, and perineuriomas.1 These tumors often are found in the dermis or subcutaneous tissue of the extremities and abdomen2; however, cases of hybrid peripheral nerve sheath tumors have been reported in many anatomical locations without a gender predilection.3 The most common type of hybrid nerve sheath tumor is a schwannoma-perineurioma.3,4 Histologically, they are well-circumscribed lesions composed of both spindled Schwann cells with plump nuclei and spindled perineural cells with more elongated thin nuclei.5 Although the Schwann cell component tends to predominate, the 2 cell populations interdigitate, making it challenging to definitively distinguish them by hematoxylin and eosin staining alone.4 However, immunohistochemical (IHC) staining can be used to help distinguish the 2 separate cell populations. Staining for S-100 and SRY-box transcription factor 10 (SOX-10) will be positive in the Schwann cell component, and staining for epithelial membrane antigen, Claudin-1, or glucose transporter-1 (Figure 1) will be positive in the perineural component. Other hybrid forms of benign nerve sheath tumors include neurofibroma-schwannoma and neurofibromaperineurioma.4 Neurofibroma-schwannomas usually have a schwannoma component containing Antoni A areas with palisading Verocay bodies. The neurofibroma cells typically have wavy elongated nuclei, fibroblasts, and mucinous myxoid material.3 Neurofibroma-perineurioma is the least common hybrid tumor. These hybrid tumors have a plexiform neurofibroma appearance with areas of perineural differentiation, which can be difficult to identify on routine histology and typically will require IHC staining to appreciate. The neurofibroma component will stain positive for S-100 and negative for markers of perineural differentiation, including epithelial membrane antigen, glucose transporter-1, and Claudin-1.3 Although schwannoma-perineuriomas are benign sporadic tumors not associated with neurofibromatosis, neurofibromaschwannomas are associated with neurofibromatosis types 1 and 2 (NF1 and NF2). Neurofibroma-perineurioma tumors usually are associated with only NF1.3,6

Schwannomas typically present in middle-aged patients as tumors located on flexor surfaces.7 Although perineural cells can be seen at the periphery of a schwannoma forming a capsule, they do not interdigitate between the Schwann cells. Schwannomas are composed almost entirely of well-differentiated Schwann cells.1,4,8 Schwannomas classically are well-circumscribed, encapsulated, biphasic lesions with alternating compact areas (Antoni A) and loosely arranged areas (Antoni B). The spindled cells occasionally may display nuclear palisading within the Antoni A areas, known as Verocay bodies (Figure 2). Antoni B areas are more disorganized and hypocellular with variable macrophage infiltrate.1,4,8 The Schwann cells predominantly will have bland cytologic features, but scattered areas of degenerative nuclear atypia (also known as ancient change) may be present.4 Multiple schwannomas are associated with NF2 gene mutations and loss of merlin protein.8 There are different subtypes of schwannomas, including cellular and plexiform schwannomas.4 Because schwannomas are benign nerve sheath lesions, treatment typically consists of excision with careful dissection around the involved nerve.9

Neurofibromas are the most common peripheral nerve sheath tumors of the skin with no notable anatomic prediction, though one study found them to be more prevalent in the upper extremities.10 They typically present as sporadic solitary lesions, but multiple lesions may appear as superficial pedunculated growths that present in those aged 20 to 30 years.11 Microscopically, neurofibromas typically are not well circumscribed and have an infiltrative growth pattern. Neurofibromas are composed of cytologically bland spindled Schwann cells with thin wavy nuclei in a variable myxoid stroma (Figure 3). In addition to Schwann cells, neurofibromas contain other cell components, including fibroblasts, mast cells, perineurial-like cells, and residual axons.4 Neurofibromas typically are located in the dermis but may extend into the subcutaneous tissue. Clinically, the overlying skin may show hyperpigmentation.8 Neurofibromas can be localized, diffuse, or plexiform, with the majority being localized. Diffuse neurofibromas clinically have a raised plaque appearance. Treatment is unnecessary because these lesions are benign.7

Desmoplastic melanoma (DM) is another diagnosis in the differential for this case. Patients with DM are older compared to non-DM melanoma patients, with a male predilection.12 Desmoplastic melanomas are more likely to be located on the head and neck. In approximately one-third of cases, no in situ component will be identified, leading to confusion of the dermal lesion as a neural lesion or an area of scar formation. Microscopically, DM presents as a variable cellular infiltrative tumor composed of spindle cells with varying degrees of nuclear atypia. The spindled melanocytes are within a collagenous (desmoplastic) stroma (Figure 4).13 Desmoplastic melanoma has been described with a low mitotic index, leading to misdiagnosis with benign spindle cell neoplasms.14 The spindle cells should be positive for S-100 and SOX-10 with IHC staining. Unlike other melanomas, human melanoma black 45 and Melan-A often are negative or only focally positive. Treatment of DM is similar to non-DM in that wide local excision usually is employed. A systematic review evaluating sentinel lymph node biopsy (SLNB) recommended consideration of SLNB in mixed DM but not for pure DM, as rates of positive SLNB were much lower in the latter.15

Patients with malignant peripheral nerve sheath tumor (MPNST) usually present with an enlarging mass, pain, or neurologic symptoms. Most cases of MPNST are located on the trunk or extremities.16 Plexiform neurofibromas, especially in adults with NF1, have the potential to transform into an MPNST.4 In fact, MPNST is the most common malignancy in patients with NF1.17 Pediatric cancer survivors also are predisposed to MPNST, with a 40-fold increase in incidence compared to the general population.18 Transformation from schwannoma to MPNST is rare but has been reported.8 Histologically, spindle cells easily can be appreciated with a fasciculated growth pattern (Figure 5). Mitotic activity and tumor necrosis may be present. Diagnosis of these tumors historically has been challenging, though recent research has identified inactivation of polycomb repressive complex 2 in 70% to 90% of MPNSTs. Because of polycomb repressive complex 2 inactivation, there is loss of stone H3K27 trimethylation that can be capitalized on for MPNST diagnosis.19 Negative IHC staining for H3K27 trimethylation has been found to be highly specific for MPNST. Negative staining for different cytokeratin and melanoma markers can be helpful in differentiating it from carcinomas and melanoma. The only curative treatment for MPNST is complete excision, leaving patients with recurrent, refractory, and metastatic cases to be encouraged for enrollment in clinical trials. The 5-year survival rates for patients with MPNST reported in the literature range from 20% to 50%.20

The Diagnosis: Hybrid Schwannoma-Perineurioma
Hybrid nerve sheath tumors are rare entities that display features of more than one nerve sheath tumor such as neurofibromas, schwannomas, and perineuriomas.1 These tumors often are found in the dermis or subcutaneous tissue of the extremities and abdomen2; however, cases of hybrid peripheral nerve sheath tumors have been reported in many anatomical locations without a gender predilection.3 The most common type of hybrid nerve sheath tumor is a schwannoma-perineurioma.3,4 Histologically, they are well-circumscribed lesions composed of both spindled Schwann cells with plump nuclei and spindled perineural cells with more elongated thin nuclei.5 Although the Schwann cell component tends to predominate, the 2 cell populations interdigitate, making it challenging to definitively distinguish them by hematoxylin and eosin staining alone.4 However, immunohistochemical (IHC) staining can be used to help distinguish the 2 separate cell populations. Staining for S-100 and SRY-box transcription factor 10 (SOX-10) will be positive in the Schwann cell component, and staining for epithelial membrane antigen, Claudin-1, or glucose transporter-1 (Figure 1) will be positive in the perineural component. Other hybrid forms of benign nerve sheath tumors include neurofibroma-schwannoma and neurofibromaperineurioma.4 Neurofibroma-schwannomas usually have a schwannoma component containing Antoni A areas with palisading Verocay bodies. The neurofibroma cells typically have wavy elongated nuclei, fibroblasts, and mucinous myxoid material.3 Neurofibroma-perineurioma is the least common hybrid tumor. These hybrid tumors have a plexiform neurofibroma appearance with areas of perineural differentiation, which can be difficult to identify on routine histology and typically will require IHC staining to appreciate. The neurofibroma component will stain positive for S-100 and negative for markers of perineural differentiation, including epithelial membrane antigen, glucose transporter-1, and Claudin-1.3 Although schwannoma-perineuriomas are benign sporadic tumors not associated with neurofibromatosis, neurofibromaschwannomas are associated with neurofibromatosis types 1 and 2 (NF1 and NF2). Neurofibroma-perineurioma tumors usually are associated with only NF1.3,6

Schwannomas typically present in middle-aged patients as tumors located on flexor surfaces.7 Although perineural cells can be seen at the periphery of a schwannoma forming a capsule, they do not interdigitate between the Schwann cells. Schwannomas are composed almost entirely of well-differentiated Schwann cells.1,4,8 Schwannomas classically are well-circumscribed, encapsulated, biphasic lesions with alternating compact areas (Antoni A) and loosely arranged areas (Antoni B). The spindled cells occasionally may display nuclear palisading within the Antoni A areas, known as Verocay bodies (Figure 2). Antoni B areas are more disorganized and hypocellular with variable macrophage infiltrate.1,4,8 The Schwann cells predominantly will have bland cytologic features, but scattered areas of degenerative nuclear atypia (also known as ancient change) may be present.4 Multiple schwannomas are associated with NF2 gene mutations and loss of merlin protein.8 There are different subtypes of schwannomas, including cellular and plexiform schwannomas.4 Because schwannomas are benign nerve sheath lesions, treatment typically consists of excision with careful dissection around the involved nerve.9

Neurofibromas are the most common peripheral nerve sheath tumors of the skin with no notable anatomic prediction, though one study found them to be more prevalent in the upper extremities.10 They typically present as sporadic solitary lesions, but multiple lesions may appear as superficial pedunculated growths that present in those aged 20 to 30 years.11 Microscopically, neurofibromas typically are not well circumscribed and have an infiltrative growth pattern. Neurofibromas are composed of cytologically bland spindled Schwann cells with thin wavy nuclei in a variable myxoid stroma (Figure 3). In addition to Schwann cells, neurofibromas contain other cell components, including fibroblasts, mast cells, perineurial-like cells, and residual axons.4 Neurofibromas typically are located in the dermis but may extend into the subcutaneous tissue. Clinically, the overlying skin may show hyperpigmentation.8 Neurofibromas can be localized, diffuse, or plexiform, with the majority being localized. Diffuse neurofibromas clinically have a raised plaque appearance. Treatment is unnecessary because these lesions are benign.7

Desmoplastic melanoma (DM) is another diagnosis in the differential for this case. Patients with DM are older compared to non-DM melanoma patients, with a male predilection.12 Desmoplastic melanomas are more likely to be located on the head and neck. In approximately one-third of cases, no in situ component will be identified, leading to confusion of the dermal lesion as a neural lesion or an area of scar formation. Microscopically, DM presents as a variable cellular infiltrative tumor composed of spindle cells with varying degrees of nuclear atypia. The spindled melanocytes are within a collagenous (desmoplastic) stroma (Figure 4).13 Desmoplastic melanoma has been described with a low mitotic index, leading to misdiagnosis with benign spindle cell neoplasms.14 The spindle cells should be positive for S-100 and SOX-10 with IHC staining. Unlike other melanomas, human melanoma black 45 and Melan-A often are negative or only focally positive. Treatment of DM is similar to non-DM in that wide local excision usually is employed. A systematic review evaluating sentinel lymph node biopsy (SLNB) recommended consideration of SLNB in mixed DM but not for pure DM, as rates of positive SLNB were much lower in the latter.15

Patients with malignant peripheral nerve sheath tumor (MPNST) usually present with an enlarging mass, pain, or neurologic symptoms. Most cases of MPNST are located on the trunk or extremities.16 Plexiform neurofibromas, especially in adults with NF1, have the potential to transform into an MPNST.4 In fact, MPNST is the most common malignancy in patients with NF1.17 Pediatric cancer survivors also are predisposed to MPNST, with a 40-fold increase in incidence compared to the general population.18 Transformation from schwannoma to MPNST is rare but has been reported.8 Histologically, spindle cells easily can be appreciated with a fasciculated growth pattern (Figure 5). Mitotic activity and tumor necrosis may be present. Diagnosis of these tumors historically has been challenging, though recent research has identified inactivation of polycomb repressive complex 2 in 70% to 90% of MPNSTs. Because of polycomb repressive complex 2 inactivation, there is loss of stone H3K27 trimethylation that can be capitalized on for MPNST diagnosis.19 Negative IHC staining for H3K27 trimethylation has been found to be highly specific for MPNST. Negative staining for different cytokeratin and melanoma markers can be helpful in differentiating it from carcinomas and melanoma. The only curative treatment for MPNST is complete excision, leaving patients with recurrent, refractory, and metastatic cases to be encouraged for enrollment in clinical trials. The 5-year survival rates for patients with MPNST reported in the literature range from 20% to 50%.20

- Hornick JL, Bundock EA, Fletcher CD. Hybrid schwannoma /perineurioma: clinicopathologic analysis of 42 distinctive benign nerve sheath tumors. Am J Surg Pathol. 2009;33:1554-1561.
- Leung KCP, Chan E, Ng HYJ, et al. Novel case of hybrid perineuriomaneurofibroma of the orbit. Can J Ophthalmol. 2019;54:E283-E285.
- Ud Din N, Ahmad Z, Abdul-Ghafar J, et al. Hybrid peripheral nerve sheath tumors: report of five cases and detailed review of literature. BMC Cancer. 2017;17:349. doi:10.1186/s12885-017-3350-1
- Belakhoua SM, Rodriguez FJ. Diagnostic pathology of tumors of peripheral nerve. Neurosurgery. 2021;88:443-456.
- Michal M, Kazakov DV, Michal M. Hybrid peripheral nerve sheath tumors: a review. Cesk Patol. 2017;53:81-88.
- Harder A, Wesemann M, Hagel C, et al. Hybrid neurofibroma /schwannoma is overrepresented among schwannomatosis and neurofibromatosis patients. Am J Surg Pathol. 2012;36:702-709.
- Bhattacharyya AK, Perrin R, Guha A. Peripheral nerve tumors: management strategies and molecular insights. J Neurooncol. 2004;69:335-349.
- Pytel P, Anthony DC. Peripheral nerves and skeletal muscle. In: Kumar V, Abbas AK, Aster JC, eds. Robbins and Cotran Pathologic Basis of Disease. 10th ed. Elsevier/Saunders; 2015:1218-1239.
- Strike SA, Puhaindran ME. Nerve tumors of the upper extremity. Clin Plast Surg. 2019;46:347-350.
- Kim DH, Murovic JA, Tiel RL, et al. A series of 397 peripheral neural sheath tumors: 30-year experience at Louisiana State University Health Sciences Center. J Neurosurg. 2005;102:246-255.
- Pilavaki M, Chourmouzi D, Kiziridou A, et al. Imaging of peripheral nerve sheath tumors with pathologic correlation: pictorial review. Eur J Radiol. 2004;52:229-239.
- Murali R, Shaw HM, Lai K, et al. Prognostic factors in cutaneous desmoplastic melanoma: a study of 252 patients. Cancer. 2010; 116:4130-4138.
- Chen LL, Jaimes N, Barker CA, et al. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68:825-833.
- de Almeida LS, Requena L, Rutten A, et al. Desmoplastic malignant melanoma: a clinicopathologic analysis of 113 cases. Am J Dermatopathol. 2008;30:207-215.
- Dunne JA, Wormald JC, Steele J, et al. Is sentinel lymph node biopsy warranted for desmoplastic melanoma? a systematic review. J Plast Reconstr Aesthet Surg. 2017;70:274-280.
- Patel TD, Shaigany K, Fang CH, et al. Comparative analysis of head and neck and non-head and neck malignant peripheral nerve sheath tumors. Otolaryngol Head Neck Surg. 2016;154:113-120.
- Prudner BC, Ball T, Rathore R, et al. Diagnosis and management of malignant peripheral nerve sheath tumors: current practice and future perspectives. Neurooncol Adv. 2020;2(suppl 1):I40-I9.
- Bright CJ, Hawkins MM, Winter DL, et al. Risk of soft-tissue sarcoma among 69,460 five-year survivors of childhood cancer in Europe. J Natl Cancer Inst. 2018;110:649-660.
- Schaefer I-M, Fletcher CD, Hornick JL. Loss of H3K27 trimethylation distinguishes malignant peripheral nerve sheath tumors from histologic mimics. Mod Pathol. 2016;29:4-13.
- Kolberg M, Holand M, Agesen TH, et al. Survival meta-analyses for >1800 malignant peripheral nerve sheath tumor patients with and without neurofibromatosis type 1. Neuro Oncol. 2013;15:135-147.
- Hornick JL, Bundock EA, Fletcher CD. Hybrid schwannoma /perineurioma: clinicopathologic analysis of 42 distinctive benign nerve sheath tumors. Am J Surg Pathol. 2009;33:1554-1561.
- Leung KCP, Chan E, Ng HYJ, et al. Novel case of hybrid perineuriomaneurofibroma of the orbit. Can J Ophthalmol. 2019;54:E283-E285.
- Ud Din N, Ahmad Z, Abdul-Ghafar J, et al. Hybrid peripheral nerve sheath tumors: report of five cases and detailed review of literature. BMC Cancer. 2017;17:349. doi:10.1186/s12885-017-3350-1
- Belakhoua SM, Rodriguez FJ. Diagnostic pathology of tumors of peripheral nerve. Neurosurgery. 2021;88:443-456.
- Michal M, Kazakov DV, Michal M. Hybrid peripheral nerve sheath tumors: a review. Cesk Patol. 2017;53:81-88.
- Harder A, Wesemann M, Hagel C, et al. Hybrid neurofibroma /schwannoma is overrepresented among schwannomatosis and neurofibromatosis patients. Am J Surg Pathol. 2012;36:702-709.
- Bhattacharyya AK, Perrin R, Guha A. Peripheral nerve tumors: management strategies and molecular insights. J Neurooncol. 2004;69:335-349.
- Pytel P, Anthony DC. Peripheral nerves and skeletal muscle. In: Kumar V, Abbas AK, Aster JC, eds. Robbins and Cotran Pathologic Basis of Disease. 10th ed. Elsevier/Saunders; 2015:1218-1239.
- Strike SA, Puhaindran ME. Nerve tumors of the upper extremity. Clin Plast Surg. 2019;46:347-350.
- Kim DH, Murovic JA, Tiel RL, et al. A series of 397 peripheral neural sheath tumors: 30-year experience at Louisiana State University Health Sciences Center. J Neurosurg. 2005;102:246-255.
- Pilavaki M, Chourmouzi D, Kiziridou A, et al. Imaging of peripheral nerve sheath tumors with pathologic correlation: pictorial review. Eur J Radiol. 2004;52:229-239.
- Murali R, Shaw HM, Lai K, et al. Prognostic factors in cutaneous desmoplastic melanoma: a study of 252 patients. Cancer. 2010; 116:4130-4138.
- Chen LL, Jaimes N, Barker CA, et al. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68:825-833.
- de Almeida LS, Requena L, Rutten A, et al. Desmoplastic malignant melanoma: a clinicopathologic analysis of 113 cases. Am J Dermatopathol. 2008;30:207-215.
- Dunne JA, Wormald JC, Steele J, et al. Is sentinel lymph node biopsy warranted for desmoplastic melanoma? a systematic review. J Plast Reconstr Aesthet Surg. 2017;70:274-280.
- Patel TD, Shaigany K, Fang CH, et al. Comparative analysis of head and neck and non-head and neck malignant peripheral nerve sheath tumors. Otolaryngol Head Neck Surg. 2016;154:113-120.
- Prudner BC, Ball T, Rathore R, et al. Diagnosis and management of malignant peripheral nerve sheath tumors: current practice and future perspectives. Neurooncol Adv. 2020;2(suppl 1):I40-I9.
- Bright CJ, Hawkins MM, Winter DL, et al. Risk of soft-tissue sarcoma among 69,460 five-year survivors of childhood cancer in Europe. J Natl Cancer Inst. 2018;110:649-660.
- Schaefer I-M, Fletcher CD, Hornick JL. Loss of H3K27 trimethylation distinguishes malignant peripheral nerve sheath tumors from histologic mimics. Mod Pathol. 2016;29:4-13.
- Kolberg M, Holand M, Agesen TH, et al. Survival meta-analyses for >1800 malignant peripheral nerve sheath tumor patients with and without neurofibromatosis type 1. Neuro Oncol. 2013;15:135-147.
A 50-year-old man presented with a 2.5-cm, subcutaneous, freely mobile nodule on the occipital scalp that first appeared 35 years prior but recently had started enlarging. Histologically the lesion was well circumscribed. Immunohistochemical staining was positive for SRY-box transcription factor 10 in some of the spindle cells, and staining for epithelial membrane antigen was positive in a separate population of intermixed spindle cells.
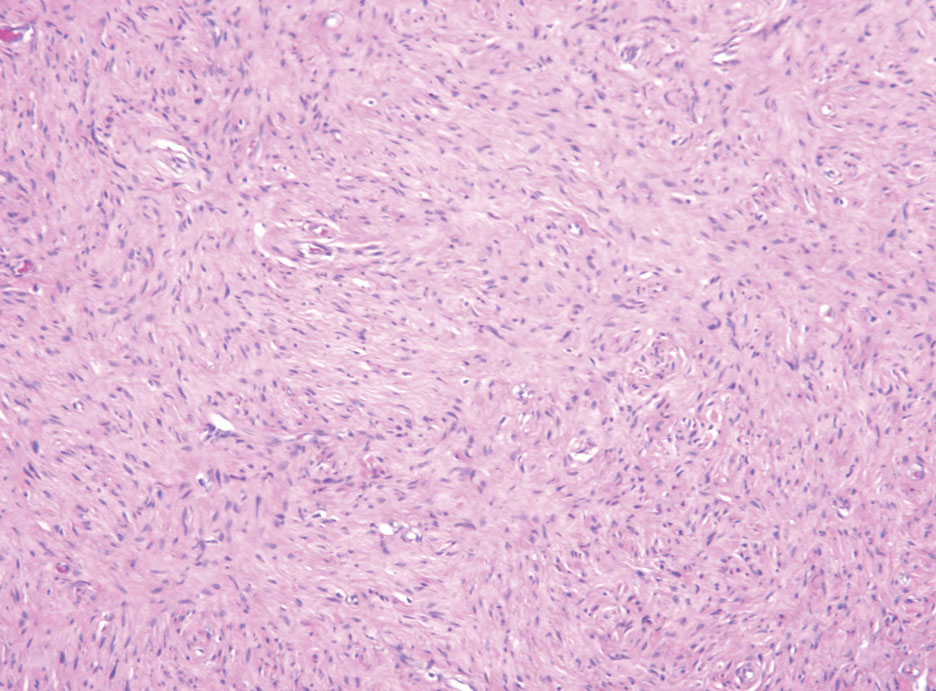
New melting hydrogel bandage could treat burn wounds faster, with less pain
Surgically debriding burn wounds can be tedious for doctors and excruciating for patients. To change that, bioengineers have created a new hydrogel formula that dissolves rapidly from wound sites, melting off in 6 minutes or less.
“The removal of dressings, with the current standard of care, is very hard and time-consuming. It becomes very painful for the patient. People are screaming, or they’re given a lot of opioids,” said senior author O. Berk Usta, PhD, of the Center for Engineering in Medicine and Surgery at Massachusetts General Hospital, Boston. “Those are the things we wanted to minimize: the pain and the time.”
Although beneficial for all patients, a short, painless bandage change would be a particular boon for younger patients. At the pediatric burns care center at Shriners Hospitals for Children (an MGH partner), researchers “observe a lot of children who go through therapy or treatment after burns,” said Dr. Usta. The team at MGH collaborated with scientists at Tufts University, Boston, with those patients in mind, setting out to create a new hydrogel that would transform burn wound care.
A better bandage
Hydrogels provide cooling relief to burn wounds and maintain a moist environment that can speed healing. There are currently hydrogel sheets and hydrogel-infused dressings, as well as gel that is applied directly to burn wounds before being covered with protective material. These dressings must be replaced frequently to prevent infections, but that can be unbearably painful and drawn out, as dressings often stick to wounds.
Mechanical debridement can be especially difficult for second-degree burn patients, whose wounds may still retain nerve endings. Debridement tends to also remove some healthy tissue and can damage newly formed tissue, slowing down healing.
“It can take up to 2, 3 hours, and it requires multiple people working on it,” said Dr. Usta.
The new hydrogel treatment can be applied directly to a wound and it forms a protective barrier around the site in 15 seconds. The hydrogel is then covered by a protective dressing until it needs to be changed.
“After you take off the protective covering, you add another solution, which dissolves the [hydrogel] dressing, so that it can be easily removed from the burn site,” Dr. Usta said.
The solution dissolves the hydrogel in 4-6 minutes.
Hybrid gels
Many hydrogels currently used for burn wounds feature physically cross-linked molecules. This makes them strong and capable of retaining moisture, but also difficult to dissolve. The researchers used a different approach.
“This is not physical cross-linking like the traditional approaches, but rather, softer covalent bonds between the different molecules. And that’s why, when you bring in another solution, the hydrogel dissolves away,” Dr. Usta said.
The new hydrogels rely on a supramolecular assembly: a network of synthetic polymers whose connections can be reversed more easily, meaning they can be dissolved quickly. Another standout feature of the new hydrogels is their hybrid composition, displaying characteristics of both liquids and solids. The polymers are knitted together into a mesh-like network that enables water retention, with the goal of maintaining the moist environment needed for wound healing.
The supramolecular assembly is also greener, Dr. Usta explained; traditional cross-linking approaches produce a lot of toxic by-products that could harm the environment.
And whereas traditional hydrogels can require a dozen chemistry steps to produce, the new hydrogels are ready after mixing two solutions, Dr. Usta explained. This makes them easy to prepare at bedside, ideal for treating large wounds in the ER or even on battlefields.
When tested in vitro, using skin cells, and in vivo, on mice, the new hydrogels were shown to be safe to use on wounds. Additional studies on mice, as well as large animals, will focus on safety and efficacy, and may be followed by human clinical trials, said Dr. Usta.
“The next phase of the project will be to look at whether these dressings will help wound healing by creating a moist environment,” said Dr. Usta.
The researchers are also exploring how to manufacture individual prewrapped hydrogels that could be applied in a clinical setting – or even in people’s homes. The consumer market is “another possibility,” said Dr. Usta, particularly among patients with “smaller, more superficial burns” or patients whose large burn wounds are still healing once they leave the hospital.
This research was supported by the National Institutes of Health, National Science Foundation, Massachusetts General Hospital Executive Committee on Research Interim Support Fund, and Shriners Hospitals.
A version of this article first appeared on Medscape.com.
Surgically debriding burn wounds can be tedious for doctors and excruciating for patients. To change that, bioengineers have created a new hydrogel formula that dissolves rapidly from wound sites, melting off in 6 minutes or less.
“The removal of dressings, with the current standard of care, is very hard and time-consuming. It becomes very painful for the patient. People are screaming, or they’re given a lot of opioids,” said senior author O. Berk Usta, PhD, of the Center for Engineering in Medicine and Surgery at Massachusetts General Hospital, Boston. “Those are the things we wanted to minimize: the pain and the time.”
Although beneficial for all patients, a short, painless bandage change would be a particular boon for younger patients. At the pediatric burns care center at Shriners Hospitals for Children (an MGH partner), researchers “observe a lot of children who go through therapy or treatment after burns,” said Dr. Usta. The team at MGH collaborated with scientists at Tufts University, Boston, with those patients in mind, setting out to create a new hydrogel that would transform burn wound care.
A better bandage
Hydrogels provide cooling relief to burn wounds and maintain a moist environment that can speed healing. There are currently hydrogel sheets and hydrogel-infused dressings, as well as gel that is applied directly to burn wounds before being covered with protective material. These dressings must be replaced frequently to prevent infections, but that can be unbearably painful and drawn out, as dressings often stick to wounds.
Mechanical debridement can be especially difficult for second-degree burn patients, whose wounds may still retain nerve endings. Debridement tends to also remove some healthy tissue and can damage newly formed tissue, slowing down healing.
“It can take up to 2, 3 hours, and it requires multiple people working on it,” said Dr. Usta.
The new hydrogel treatment can be applied directly to a wound and it forms a protective barrier around the site in 15 seconds. The hydrogel is then covered by a protective dressing until it needs to be changed.
“After you take off the protective covering, you add another solution, which dissolves the [hydrogel] dressing, so that it can be easily removed from the burn site,” Dr. Usta said.
The solution dissolves the hydrogel in 4-6 minutes.
Hybrid gels
Many hydrogels currently used for burn wounds feature physically cross-linked molecules. This makes them strong and capable of retaining moisture, but also difficult to dissolve. The researchers used a different approach.
“This is not physical cross-linking like the traditional approaches, but rather, softer covalent bonds between the different molecules. And that’s why, when you bring in another solution, the hydrogel dissolves away,” Dr. Usta said.
The new hydrogels rely on a supramolecular assembly: a network of synthetic polymers whose connections can be reversed more easily, meaning they can be dissolved quickly. Another standout feature of the new hydrogels is their hybrid composition, displaying characteristics of both liquids and solids. The polymers are knitted together into a mesh-like network that enables water retention, with the goal of maintaining the moist environment needed for wound healing.
The supramolecular assembly is also greener, Dr. Usta explained; traditional cross-linking approaches produce a lot of toxic by-products that could harm the environment.
And whereas traditional hydrogels can require a dozen chemistry steps to produce, the new hydrogels are ready after mixing two solutions, Dr. Usta explained. This makes them easy to prepare at bedside, ideal for treating large wounds in the ER or even on battlefields.
When tested in vitro, using skin cells, and in vivo, on mice, the new hydrogels were shown to be safe to use on wounds. Additional studies on mice, as well as large animals, will focus on safety and efficacy, and may be followed by human clinical trials, said Dr. Usta.
“The next phase of the project will be to look at whether these dressings will help wound healing by creating a moist environment,” said Dr. Usta.
The researchers are also exploring how to manufacture individual prewrapped hydrogels that could be applied in a clinical setting – or even in people’s homes. The consumer market is “another possibility,” said Dr. Usta, particularly among patients with “smaller, more superficial burns” or patients whose large burn wounds are still healing once they leave the hospital.
This research was supported by the National Institutes of Health, National Science Foundation, Massachusetts General Hospital Executive Committee on Research Interim Support Fund, and Shriners Hospitals.
A version of this article first appeared on Medscape.com.
Surgically debriding burn wounds can be tedious for doctors and excruciating for patients. To change that, bioengineers have created a new hydrogel formula that dissolves rapidly from wound sites, melting off in 6 minutes or less.
“The removal of dressings, with the current standard of care, is very hard and time-consuming. It becomes very painful for the patient. People are screaming, or they’re given a lot of opioids,” said senior author O. Berk Usta, PhD, of the Center for Engineering in Medicine and Surgery at Massachusetts General Hospital, Boston. “Those are the things we wanted to minimize: the pain and the time.”
Although beneficial for all patients, a short, painless bandage change would be a particular boon for younger patients. At the pediatric burns care center at Shriners Hospitals for Children (an MGH partner), researchers “observe a lot of children who go through therapy or treatment after burns,” said Dr. Usta. The team at MGH collaborated with scientists at Tufts University, Boston, with those patients in mind, setting out to create a new hydrogel that would transform burn wound care.
A better bandage
Hydrogels provide cooling relief to burn wounds and maintain a moist environment that can speed healing. There are currently hydrogel sheets and hydrogel-infused dressings, as well as gel that is applied directly to burn wounds before being covered with protective material. These dressings must be replaced frequently to prevent infections, but that can be unbearably painful and drawn out, as dressings often stick to wounds.
Mechanical debridement can be especially difficult for second-degree burn patients, whose wounds may still retain nerve endings. Debridement tends to also remove some healthy tissue and can damage newly formed tissue, slowing down healing.
“It can take up to 2, 3 hours, and it requires multiple people working on it,” said Dr. Usta.
The new hydrogel treatment can be applied directly to a wound and it forms a protective barrier around the site in 15 seconds. The hydrogel is then covered by a protective dressing until it needs to be changed.
“After you take off the protective covering, you add another solution, which dissolves the [hydrogel] dressing, so that it can be easily removed from the burn site,” Dr. Usta said.
The solution dissolves the hydrogel in 4-6 minutes.
Hybrid gels
Many hydrogels currently used for burn wounds feature physically cross-linked molecules. This makes them strong and capable of retaining moisture, but also difficult to dissolve. The researchers used a different approach.
“This is not physical cross-linking like the traditional approaches, but rather, softer covalent bonds between the different molecules. And that’s why, when you bring in another solution, the hydrogel dissolves away,” Dr. Usta said.
The new hydrogels rely on a supramolecular assembly: a network of synthetic polymers whose connections can be reversed more easily, meaning they can be dissolved quickly. Another standout feature of the new hydrogels is their hybrid composition, displaying characteristics of both liquids and solids. The polymers are knitted together into a mesh-like network that enables water retention, with the goal of maintaining the moist environment needed for wound healing.
The supramolecular assembly is also greener, Dr. Usta explained; traditional cross-linking approaches produce a lot of toxic by-products that could harm the environment.
And whereas traditional hydrogels can require a dozen chemistry steps to produce, the new hydrogels are ready after mixing two solutions, Dr. Usta explained. This makes them easy to prepare at bedside, ideal for treating large wounds in the ER or even on battlefields.
When tested in vitro, using skin cells, and in vivo, on mice, the new hydrogels were shown to be safe to use on wounds. Additional studies on mice, as well as large animals, will focus on safety and efficacy, and may be followed by human clinical trials, said Dr. Usta.
“The next phase of the project will be to look at whether these dressings will help wound healing by creating a moist environment,” said Dr. Usta.
The researchers are also exploring how to manufacture individual prewrapped hydrogels that could be applied in a clinical setting – or even in people’s homes. The consumer market is “another possibility,” said Dr. Usta, particularly among patients with “smaller, more superficial burns” or patients whose large burn wounds are still healing once they leave the hospital.
This research was supported by the National Institutes of Health, National Science Foundation, Massachusetts General Hospital Executive Committee on Research Interim Support Fund, and Shriners Hospitals.
A version of this article first appeared on Medscape.com.
FROM BIOACTIVE MATERIALS
Janus Kinase Inhibitors in the Treatment of Atopic Dermatitis: Military Considerations
The atopic dermatitis (AD) therapeutic landscape is changing considerably with the advent of Janus kinase (JAK) inhibitors. Several JAK inhibitors recently have been approved by the US Food and Drug Administration, building off years of foundational research aimed at elucidating the downstream effects of the JAK–signal transducer and activator of transcription (STAT) pathway and its role in AD pathogenesis. Agents within this promising new class of drugs have performed well vs placebo in phase 2 and 3 clinical trials. This article reviews relevant trial efficacy and safety data of several JAK inhibitors as well as the implications of the use of these medications in AD patients, with specific considerations unique to active-duty military personnel.
Background on JAK Inhibitors
The hematopoietin superfamily of cytokine receptors encompasses a broad group that includes receptors for immune (eg, IL-2, IL-4, IFN-γ), hematopoietic (eg, erythropoietin, thrombopoietin, granulocyte-macrophage colony-stimulating factor), and nonimmune (eg, prolactin, leptin, growth hormone) cytokines. These cytokines signal via the JAK-STAT pathway. The hematopoietin family of cytokine receptors lacks intrinsic enzymatic activity, and as a result, they rely on JAK enzymes to transmit their signals intracellularly after cytokine binding to the receptor.1 Janus, of Roman mythology, was the god of doorways and archways and was commonly depicted with 2 heads. Janus kinases were named for their 2 “faces,” the kinase domain with its adjacent regulatory kinaselike domains.2 The binding of a cytokine to its receptor triggers engagement of the receptor by JAKs, leading to phosphorylation of both the JAKs and the receptor. Subsequent recruitment and phosphorylation of STAT proteins occurs. Following STAT phosphorylation, the STAT proteins dissociate, dimerize, and translocate to the nucleus, where they enact changes in cell behavior through transcriptional effects.1
Humans possess only 4 JAKs. Janus kinase 1, JAK2, and tyrosine kinase 2 are widely expressed, whereas JAK3 expression is largely limited to immune cells. Thus, there is notable overlap in the use of the 4 JAKs among the relatively larger number of various cytokines that utilize them to propagate intracellular signaling.1 Janus kinase 1 is important for signaling of receptors activated by a variety of interleukins, as well as IFN-α, IFN-β, and IFN-γ. Janus kinase 2 is important for signaling for the hormonelike cytokines erythropoietin, thrombopoietin, growth hormone, granulocyte-macrophage colony-stimulating factor, IL-3, and IL-5. Janus kinase 3 is important for hematopoietic cell proliferation and function.1
JAK Inhibitors and Atopic Dermatitis
Topical treatments, including corticosteroids and calcineurin inhibitors, are considered the standard-of-care therapy for most patients with AD; however, their clinical benefit often is limited by their anatomic use restrictions and local adverse events, including skin atrophy, striae, and application-site reactions such as stinging and burning.3 As a result, long-term application of these drugs, particularly in sensitive areas, is not recommended owing to safety/tolerability issues.3 Systemic immunomodulatory medications are indicated for patients with AD who do not achieve adequate disease control with topical treatments and/or phototherapy or for patients with severely impaired quality of life.4
Janus kinase inhibitors have several key benefits over biologics: oral and topical bioavailability, predictable pharmacokinetics, nonimmunogenicity, and dosing flexibility.4 Janus kinase 1 is central to the cell signaling of many cytokines involved in the pathogenesis of AD that comprise the T-helper lymphocytes type 2 axis: IL-4, IL-13, and thymic stromal lymphopoietin. Janus kinase signaling also may mediate itch responses by acting directly on sensory nerve fibers. Consequently, the substantial reduction in pruritus seen in many studies of JAK inhibitors is thought to be in part due to the effects on sensory nerve fibers in the skin and the blockade of early itch signaling in response to IL-4, IL-13, and IL-31.5
Abrocitinib is a JAK1 inhibitor with a similar side effect profile to upadacitinib. Both agents were approved by the FDA for the treatment of refractory moderate to severe AD on January 14, 2022.6 These are second-generation (also referred to as selective) oral JAK inhibitors with much greater inhibitory potency for JAK1 than for JAK2, JAK3, or tyrosine kinase 2, thereby reducing the risk for hematopoietic effects associated with JAK2 inhibition. The approval of abrocitinib stemmed from the phase 3 clinical trial JAK1 Atopic Dermatitis Efficacy and Safety (JADE)-MONO-1 (N=387),7 its replicate trial JADE-MONO-2 (N=391),8 and the JADE COMPARE trial.9 The JADE-MONO trials were multicenter, double-blind, placebo-controlled studies that enrolled patients 12 years and older with moderate to severe AD.7,8 Treatment groups consisted of 100-mg and 200-mg doses and were evaluated with the placebo group for their ability to achieve an investigator global assessment (IGA) score of 0 or 1 and eczema area and severity index 75 (EASI-75) at 12 weeks.7,8 Sixty-three percent of patients in the 200-mg group, 40% in the 100-mg group, and 12% in the placebo group reached the EASI-75 end point, and the differences in these response rates were statistically significant vs placebo (100 mg: 27.9% [95% CI, 17.4-38.3], P<.0001; 200 mg: 51.0% [95% CI, 40.5-61.5], P<.0001). Notably, 44% of patients using the 200-mg dose achieved almost complete or complete resolution of AD (IGA responders, improvement of ≥2 and IGA score of 0 or 1 at 12 weeks).7 In JADE-MONO-2, EASI-75 also was achieved significantly more frequently in the treatment groups compared with the placebo group at 12 weeks (200 mg: 61.0%; 100 mg: 44.5%; placebo: 10.4%; P<.001 vs placebo).8 Adjunctive therapy with topical corticosteroids was prohibited in both studies. A dose-dependent decrease in platelets was seen in both trials, as in the phase 2 trial that preceded them.10
The primary end point of the JADE COMPARE trial was to evaluate the efficacy of abrocitinib as compared with placebo at 12 weeks in adult patients with moderate to severe AD and in the setting of concomitant topical corticosteroid therapy.9 One of several secondary end points of this study compared the ability of dupilumab vs abrocitinib and placebo treatment groups to achieve itch reduction at 2 weeks, defined as 4-point improvement or more from baseline in the score on the Peak Pruritus Numerical Rating Scale (NRS), a well‐defined, reliable, sensitive, and valid scale for evaluating worst itch intensity in adults with moderate to severe AD.9,11 The primary end point was the same as in the other phase 3 studies and was met in the JADE COMPARE trial by all treatment arms. An EASI-75 was seen in 70.3% of patients treated with 200 mg of abrocitinib, 58.7% in the 100-mg abrocitinib group, 58.1% in the dupilumab group, and 27.1% in the placebo group (P<.001 for both abrocitinib doses vs placebo). Only the 200-mg dose of abrocitinib demonstrated superior itch response at week 2 compared with dupilumab (22.1% response rate difference [95% CI, 13.5-30.7; P<.001]). Both abrocitinib groups failed to demonstrate significant differences compared with dupilumab with respect to other secondary end points to include IGA response and EASI-75 at week 16.9
The most frequently reported treatment-associated adverse events were nausea, nasopharyngitis, upper respiratory tract infection, and headache, and the percentages were similar among trial groups.9 Acne was more frequently reported in the abrocitinib groups compared with placebo and the dupilumab group, and conjunctivitis was more frequently reported in the dupilumab group. Herpesvirus cutaneous infections were rare in the abrocitinib groups, as were other serious infections. No deaths, major adverse cardiovascular events (MACEs), or venous thromboembolic events (VTEs) occurred during the trial. Dose-dependent increases in creatinine phosphokinase were seen in the abrocitinib groups, whereas dose-dependent decreases were seen in platelet counts, with no patient demonstrating a platelet count below 75,000/mm3 during the study.9 Low-density lipoprotein cholesterol levels and high-density lipoprotein cholesterol levels increased in a dose-dependent manner as well, but the ratios of low-density lipoprotein to high-density lipoprotein were unchanged.9 The results of a phase 3, 92-week extension study, JADE EXTEND, were recently published and demonstrated a role for abrocitinib as a treatment for patients with moderate to severe AD, regardless of prior dupilumab response status.12
Upadacitinib, another selective JAK1 inhibitor, was approved following data from 2 replicate double-blind, phase 3, randomized, controlled trials—Measure Up 1 and Measure Up 2.13 Results demonstrated that monotherapy with once-daily upadacitinib 15 mg or 30 mg is an effective and well-tolerated treatment option for patients with moderate to severe AD vs placebo. All coprimary end points at week 16 were achieved in the upadacitinib groups in both trials. Acne, upper respiratory tract infections, nasopharyngitis, headache, and increase in serum creatinine phosphokinase levels were the most frequently reported adverse events. Rates of herpes zoster infection in upadacitinib groups were low.13
In the subsequent phase 3 AD Up trial, researchers evaluated the safety and efficacy of combination therapy with topical corticosteroids in patients aged 12 to 75 years.14 Upadacitinib groups again achieved the identical coprimary end points that were present in the Measure Up trials13 as well as all key secondary end points.14 Additionally, significant differences in secondary end points, such as a 4-point improvement in the Worst Pruritus NRS vs placebo, were noticed in both upadacitinib treatment groups as early as 1 week into the study (P<.0001), with maintenance of the effect through to week 16 (P<.0001).14 AD Up was followed by the Heads Up trial, a 24-week, phase 3, multicenter, double-blind, randomized, controlled trial comparing safety and efficacy of upadacitinib with dupilumab among 692 adults with moderate to severe AD.15 At week 16, a higher percentage of patients in the upadacitinib group achieved EASI-75 vs the dupilumab group (71.0% vs 61.1%, respectively; P=.006). The difference noted at week 2 was even more impressive, with 43.7% of patients in the upadacitinib treatment group achieving EASI-75 compared with 17.4% in the dupilumab group (P<.001). No new safety-related events were registered compared with the already available data for both drugs.15
Ruxolitinib (RUX) is a topical JAK1 and JAK2 inhibitor that was FDA approved in September 2021 for the treatment of AD.16 In a phase 2 clinical trial of 307 adult patients with 3% to 20% body surface area (BSA) affected with AD, significant reductions in itch NRS scores were observed within 36 hours after the first application of RUX cream 1.5% twice daily (-1.8 vs -0.2, P<.0001).17 These decreases were noted within the first 2 weeks of treatment for all the RUX cream regimens and were sustained through to week 8, the end of the double-blind period. At 4 weeks, change in itch from baseline was significantly reduced in the RUX 1.5% twice-daily group compared with the triamcinolone ointment 0.1% group (−4 vs −2.5, P=.003). During the open-label treatment period from 8 to 12 weeks, all patients who switched to RUX cream 1.5% twice daily noted further reductions in itch, and those who continued it demonstrated additional improvement.17
The recent FDA approval was further backed by positive phase 3 trial data from the TRuE-AD1 and TRuE-AD2 studies.18 Patients in these trials were aged 12 years and older and had AD for 2 or more years with an IGA score of 2 or 3 and 3% to 20% affected BSA. Patients were randomized to twice-daily RUX cream 0.75%, RUX cream 1.5%, or vehicle cream, and the primary end point was an IGA score of 0 or 1 and an improvement of 2 or more points from baseline at week 8. Significantly more patients achieved IGA treatment success with RUX cream 0.75% (TRuE-AD1, 50.0%; TRuE-AD2, 39.0%) and RUX cream 1.5% (TRuE-AD1, 53.8%; TRuE-AD2, 51.3%) vs vehicle (TRuE-AD1, 15.1%; TRuE-AD2, 7.6%; P<.0001) at week 8. The RUX groups experienced dramatically reduced itch compared with vehicle, with a mean reduction of approximately 3 points on the NRS at 8 weeks. Additionally, statistically significant itch reductions vs vehicle were reported within 12 hours of first application of RUX cream 1.5% (P<.05). Application-site reactions including stinging and burning occurred in less than 1% of patients, and none were considered clinically significant. Mean plasma concentrations of RUX were monitored during the phase 2 and 3 AD studies and did not lead to any clinically meaningful changes in hematologic parameters. The low bioavailability following topical application of RUX cream (6% in the TRuE-AD studies) allows for a targeted delivery of the active drug to lesional skin while reducing the safety issues associated with oral administration of JAK inhibitors.18
Baricitinib is a predominantly JAK1 and JAK2 inhibitor that was the first JAK inhibitor to be approved for the treatment of moderate to severe AD in the European Union and Japan.19 Although the FDA’s decision on baricitinib has lagged behind market competitors, in 2 phase 3 clinical trials, BREEZE-AD1 and BREEZE-AD2, baricitinib demonstrated benefit over placebo on clinically important measures of disease severity. The primary end point—the proportion of patients achieving an IGA score of 0 or 1 with an improvement of 2 or more points from baseline at week 16—was met by both tested doses of baricitinib (2 mg and 4 mg) vs placebo in BREEZE-AD1 (2 mg, P≤.05; 4 mg, P≤.001) and BREEZE-AD2 (2 mg, P≤.05; 4 mg, P≤.001). In addition, baricitinib 4 mg consistently demonstrated significant benefit over placebo on other clinically important measures of disease severity at week 16 to include itch (BREEZE-AD1 and BREEZE-AD2, P≤.001), sleep disturbance (BREEZE-AD1, P≤.01; BREEZE-AD2, P≤.001), and skin pain (BREEZE-AD1, P≤.01; BREEZE-AD2, P≤.001). Nasopharyngitis, upper respiratory tract infections, creatine phosphokinase elevations, and headaches were the most frequently reported adverse events. During the 16-week treatment period in these trials, no deaths, MACEs, or VTEs occurred.19 Similar results were seen in a long-term extension study, BREEZE-AD3.20 The combination of baricitinib and topical corticosteroids were evaluated in 2 additional phase 3 trials, BREEZE-AD421 and BREEZE-AD7.22 Although only baricitinib 4 mg met the primary end point of EASI-75 at week 16 in both trials, both dosing regimens plus topical corticosteroids demonstrated notable reduction in multiple clinical and quality-of-life indices prior to week 2 when compared with placebo plus topical corticosteroids.22,23
AD in Military Service Members
Atopic dermatitis is a common condition in the general population, with a prevalence of 7.3% (95% CI, 5.9-8.8) in a recent study of American adults.24 Historically, the burden of AD that would be expected among active-duty military service members given the prevalence among the general population has not been observed, in part because of the disqualifying nature of AD for enlistment.25 The Department of Defense Instruction 6130.03, Volume 1, Medical Standards for Military Service: Appointment, Enlistment, or Induction stipulates that a history of AD or eczema after the twelfth birthday or history of residual or recurrent lesions in characteristic areas (ie, face, neck, antecubital or popliteal fossae, occasionally wrists and hands) is disqualifying.26 Specific military services possess additional standards that further define limits within the aforementioned Department of Defense instruction.25 Additionally, there are service-specific policies in place that mandate medical evaluation boards to determine fitness for continued service in the event the condition interferes with the member’s ability to perform their duties. Insection 4.2 of the U.S. Navy Aeromedical Reference and Waiver Guide, further restrictions for aviation personnel are delineated: “Depending on the location of lesions, there can be interference with the wearing of flight gear. The symptoms, particularly itching, can be distracting in flight. Patients with atopic dermatitis are more susceptible to contact dermatitis due to irritants found in a military environment.” Ultimately, the document stipulates that symptom severity and the requirement for therapy will determine the aeromedical disposition. It specifically states that “[p]atients controlled on topical therapy over small areas and patients who are asymptomatic on stable doses of loratadine (Claritin) OR fexofenadine (Allegra) may be considered for waiver,” and “intermittent use of topical steroids over a limited area is compatible with waiver.”27 It follows that limited use of topical JAK inhibitors, such as RUX, would be compatible with a waiver, given the favorable side effect profile and requirement for use in patients with 20% or lower affected BSA.16 This is just one example of duty-specific and service-specific medical standards that exist that could impact the use of both topical and oral JAK inhibitors.
Use of oral JAK inhibitors in active-duty service members is less ideal for multiple reasons. A large randomized safety clinical trial of patients with rheumatoid arthritis who received tofacitinib and methotrexate was required by the FDA to evaluate the risk of MACEs, malignancy, and infections associated with JAK inhibitor treatment. Data from this trial showed a dose-dependent increased risk for MACEs, all-cause mortality, and thrombosis at both doses of tofacitinib compared with tumor necrosis factor inhibitors and a non–dose-dependent increased risk for malignancy excluding nonmelanoma skin cancer.28 In contrast to the MACE and VTE data from patients with diseases other than AD treated with JAK inhibitors, there has been only 1 patient who developed a pulmonary embolism while being treated with baricitinib 4 mg.22,29 Downstream effects from the above study were label recommendations to reserve the medicines for patients who had an inadequate response or intolerance to 1 or more tumor necrosis factor blockers and to carefully consider risks vs benefits in patients, in particular current or prior smokers, those with other cardiovascular risk factors or a history of VTE, and those with a malignancy history other than already treated nonmelanoma skin cancer.28
There are consistent observations of laboratory abnormalities with JAK inhibitors, as discussed above, to include creatine phosphokinase elevation and cytopenias.30 Although existing data demonstrate that cytopenias are less of a concern in the AD population compared with the rheumatoid arthritis population, baseline and periodic laboratory monitoring are still recommended. In general, pretreatment laboratory assessment prior to initiating an oral JAK inhibitor should consist of a complete blood cell count with differential, complete metabolic panel, tuberculosis screening, chronic hepatitis panel, HIV screening, and a fasting lipid panel.2 The feasibility of obtaining these laboratory measurements in an operational setting or sea-going platform is limited, but many deployed locations and naval vessels possess the laboratory capability to perform a complete blood cell count and complete metabolic panel. Overall tolerability of oral JAK inhibitors in the treatment of AD appears favorable based on studies that were mostly 16 weeks in duration. Few recent longer-term studies have confirmed this side effect profile, but additional studies are needed.
Final Thoughts
Janus kinase inhibitors are a promising therapeutic class with multiple recently FDA-approved agents for the treatment of moderate to severe AD, with new agents on the horizon. Available efficacy data are promising and balanced by a favorable safety profile in clinical trials to date. The oral and topical bioavailability of JAK inhibitors makes them attractive alternatives to existing therapies. The rapidity of itch reduction and AD improvement demonstrated in multiple trials has the potential to decrease the length of limited-duty assignments, potentially returning treated service members to full-duty status more expeditiously. Other applications include use of these medications in scenarios where injectable medications are either unavailable or unsupported.
In the active-duty population, both the condition and/or the treatment may be duty limiting. Service members with AD who require more than topical treatment may require a medical evaluation board to determine if they are still fit to serve. The deployed environment routinely exacerbates AD and exposes service members to infections and environments where immunosuppression can create more risks than in the general population. Nonbiologic medications, which do not require refrigeration, are an exciting option for our patients with AD, including those actively serving or considering serving in the military. However, all factors in any patient’s life should be considered. Therefore, it is important for the nonmilitary dermatologist to work with local military physicians and the patient to determine the optimal treatment regimen to result in the best possible outcome.
- Damsky W, Peterson D, Ramseier J, et al. The emerging role of Janus kinase inhibitors in the treatment of autoimmune and inflammatory diseases. J Allergy Clin Immunol. 2021;147:814-826.
- Gadina M, Le MT, Schwartz DM, et al. Janus kinases to jakinibs: from basic insights to clinical practice. Rheumatology (Oxford). 2019;58(suppl 1):i4-i6.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2, management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Cartron AM, Nguyen TH, Roh YS, et al. Janus kinase inhibitors for atopic dermatitis: a promising treatment modality. Clin Exp Dermatol. 2021;46:820-824.
- Oetjen LK, Mack MR, Feng J, et al. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell. 2017;171:217-228.e13.
- U.S. FDA approves Pfizer’s CIBINQO® (abrocitinib) for adults with moderate-to-severe atopic dermatitis [press release]. January 14, 2022. Accessed November 18, 2022. https://www.pfizer.com/news/press-release/press-release-detail/us-fda-approves-pfizers-cibinqor-abrocitinib-adults
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266.
- Silverberg JI, Simpson EL, Thyssen JP, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:863-873.
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112.
- Gooderham MJ, Forman SB, Bissonnette R, et al. Efficacy and safety of oral Janus kinase 1 inhibitor abrocitinib for patients with atopic dermatitis: a phase 2 randomized clinical trial. JAMA Dermatol. 2019;155:1371-1379. Published correction appears in JAMA Dermatol. 2020;156:104.
- Yosipovitch G, Reaney M, Mastey V, et al. Peak Pruritus Numerical Rating Scale: psychometric validation and responder definition for assessing itch in moderate-to-severe atopic dermatitis. Br J Dermatol. 2019;181:761-769.
- Shi VY, Bhutani T, Fonacier L, et al. Phase 3 efficacy and safety of abrocitinib in adults with moderate-to-severe atopic dermatitis after switching from dupilumab (JADE EXTEND). J Am Acad Dermatol. 2022;87:351-358.
- Guttman-Yassky E, Teixeira HD, Simpson EL, et al. Once-daily upadacitinib versus placebo in adolescents and adults with moderate-to-severe atopic dermatitis (Measure Up 1 and Measure Up 2): results from two replicate double-blind, randomised controlled phase 3 trials. Lancet. 2021;397:2151-2168.
- Reich K, Teixeira HD, de Bruin-Weller M, et al. Safety and efficacy of upadacitinib in combination with topical corticosteroids in adolescents and adults with moderate-to-severe atopic dermatitis (AD Up): results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021;397:2169-2181.
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2021;157:1047-1055. Published correction appears in JAMA Dermatol. 2022;158:219.
- FDA approves Opzelura. Drugs.com. September 21, 2021. Accessed October 6, 2022. https://www.drugs.com/newdrugs/fda-approves-opzelura-ruxolitinib-cream-atopic-dermatitis-ad-5666.html
- Kim BS, Sun K, Papp K, et al. Effects of ruxolitinib cream on pruritus and quality of life in atopic dermatitis: results from a phase 2, randomized, doseranging, vehicle- and active-controlled study. J Am Acad Dermatol. 2020;82:1305-1313.
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies. J Am Acad Dermatol. 2021;85:863-872.
- Simpson EL, Lacour JP, Spelman L, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis and inadequate response to topical corticosteroids: results from two randomized monotherapy phase III trials. Br J Dermatol. 2020;183:242-255.
- Silverberg JI, Simpson EL, Wollenberg A, et al. Long-term efficacy of baricitinib in adults with moderate to severe atopic dermatitis who were treatment responders or partial responders: an extension study of 2 randomized clinical trials. JAMA Dermatol. 2021;157:691-699.
- Lilly and Incyte announce top-line results from phase 3 study (BREEZE-AD4) of oral selective JAK inhibitor baricitinib in combination with topical corticosteroids in patients with moderate to severe atopic dermatitis not controlled with cyclosporine. January 27, 2020. Accessed November 18, 2022. https://investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-announce-top-line-results-phase-3-study-breeze
- Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:1333-1343.
- Wollenberg A, Nakahara T, Maari C, et al. Impact of baricitinib in combination with topical steroids on atopic dermatitis symptoms, quality of life and functioning in adult patients with moderate-to-severe atopic dermatitis from the BREEZE-AD7 phase 3 randomized trial. J Eur Acad Dermatol Venereol. 2021;35:1543-1552.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590.
- Jeter J, Bowen C. Atopic dermatitis and implications for military service. Mil Med. 2019;184:E177-E182.
- Department of Defense. Medical standards for military service: appointment, enlistment, or induction. DoD Instruction 6130.03. Vol 1. May 6, 2022. Accessed November 18, 2022. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_v1p.PDF?ver=9NsVi30gsHBBsRhMLcyVVQ%3d%3d
- Dermatitis. In: U.S. Navy Aeromedical Reference and Waiver Guide. Navy Medicine Operational Training Command and Naval Aerospace Medical Institute. August 11, 2021. Accessed November 18, 2022. https://www.med.navy.mil/Portals/62/Documents/NMFSC/NMOTC/NAMI/ARWG/Waiver%20Guide/ARWG%20COMPLETE_210811.pdf?ver=_pLPzFrtl8E2swFESnN4rA%3D%3D
- FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. FDA Drug Safety Podcast. U.S. Food and Drug Administration. Updated January 14, 2022. Accessed November 18, 2022. https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Chang PH, Huang SF, Chang PS, et al. Safety considerations of systemic Janus kinase inhibitors in atopic dermatitis applications. J Dermatol. 2021;48:1631-1639.
- Wood H, Chandler A, Nezamololama N, et al. Safety of Janus kinase (JAK) inhibitors in the short-term treatment of atopic dermatitis. Int J Dermatol. 2022;61:746-754.
The atopic dermatitis (AD) therapeutic landscape is changing considerably with the advent of Janus kinase (JAK) inhibitors. Several JAK inhibitors recently have been approved by the US Food and Drug Administration, building off years of foundational research aimed at elucidating the downstream effects of the JAK–signal transducer and activator of transcription (STAT) pathway and its role in AD pathogenesis. Agents within this promising new class of drugs have performed well vs placebo in phase 2 and 3 clinical trials. This article reviews relevant trial efficacy and safety data of several JAK inhibitors as well as the implications of the use of these medications in AD patients, with specific considerations unique to active-duty military personnel.
Background on JAK Inhibitors
The hematopoietin superfamily of cytokine receptors encompasses a broad group that includes receptors for immune (eg, IL-2, IL-4, IFN-γ), hematopoietic (eg, erythropoietin, thrombopoietin, granulocyte-macrophage colony-stimulating factor), and nonimmune (eg, prolactin, leptin, growth hormone) cytokines. These cytokines signal via the JAK-STAT pathway. The hematopoietin family of cytokine receptors lacks intrinsic enzymatic activity, and as a result, they rely on JAK enzymes to transmit their signals intracellularly after cytokine binding to the receptor.1 Janus, of Roman mythology, was the god of doorways and archways and was commonly depicted with 2 heads. Janus kinases were named for their 2 “faces,” the kinase domain with its adjacent regulatory kinaselike domains.2 The binding of a cytokine to its receptor triggers engagement of the receptor by JAKs, leading to phosphorylation of both the JAKs and the receptor. Subsequent recruitment and phosphorylation of STAT proteins occurs. Following STAT phosphorylation, the STAT proteins dissociate, dimerize, and translocate to the nucleus, where they enact changes in cell behavior through transcriptional effects.1
Humans possess only 4 JAKs. Janus kinase 1, JAK2, and tyrosine kinase 2 are widely expressed, whereas JAK3 expression is largely limited to immune cells. Thus, there is notable overlap in the use of the 4 JAKs among the relatively larger number of various cytokines that utilize them to propagate intracellular signaling.1 Janus kinase 1 is important for signaling of receptors activated by a variety of interleukins, as well as IFN-α, IFN-β, and IFN-γ. Janus kinase 2 is important for signaling for the hormonelike cytokines erythropoietin, thrombopoietin, growth hormone, granulocyte-macrophage colony-stimulating factor, IL-3, and IL-5. Janus kinase 3 is important for hematopoietic cell proliferation and function.1
JAK Inhibitors and Atopic Dermatitis
Topical treatments, including corticosteroids and calcineurin inhibitors, are considered the standard-of-care therapy for most patients with AD; however, their clinical benefit often is limited by their anatomic use restrictions and local adverse events, including skin atrophy, striae, and application-site reactions such as stinging and burning.3 As a result, long-term application of these drugs, particularly in sensitive areas, is not recommended owing to safety/tolerability issues.3 Systemic immunomodulatory medications are indicated for patients with AD who do not achieve adequate disease control with topical treatments and/or phototherapy or for patients with severely impaired quality of life.4
Janus kinase inhibitors have several key benefits over biologics: oral and topical bioavailability, predictable pharmacokinetics, nonimmunogenicity, and dosing flexibility.4 Janus kinase 1 is central to the cell signaling of many cytokines involved in the pathogenesis of AD that comprise the T-helper lymphocytes type 2 axis: IL-4, IL-13, and thymic stromal lymphopoietin. Janus kinase signaling also may mediate itch responses by acting directly on sensory nerve fibers. Consequently, the substantial reduction in pruritus seen in many studies of JAK inhibitors is thought to be in part due to the effects on sensory nerve fibers in the skin and the blockade of early itch signaling in response to IL-4, IL-13, and IL-31.5
Abrocitinib is a JAK1 inhibitor with a similar side effect profile to upadacitinib. Both agents were approved by the FDA for the treatment of refractory moderate to severe AD on January 14, 2022.6 These are second-generation (also referred to as selective) oral JAK inhibitors with much greater inhibitory potency for JAK1 than for JAK2, JAK3, or tyrosine kinase 2, thereby reducing the risk for hematopoietic effects associated with JAK2 inhibition. The approval of abrocitinib stemmed from the phase 3 clinical trial JAK1 Atopic Dermatitis Efficacy and Safety (JADE)-MONO-1 (N=387),7 its replicate trial JADE-MONO-2 (N=391),8 and the JADE COMPARE trial.9 The JADE-MONO trials were multicenter, double-blind, placebo-controlled studies that enrolled patients 12 years and older with moderate to severe AD.7,8 Treatment groups consisted of 100-mg and 200-mg doses and were evaluated with the placebo group for their ability to achieve an investigator global assessment (IGA) score of 0 or 1 and eczema area and severity index 75 (EASI-75) at 12 weeks.7,8 Sixty-three percent of patients in the 200-mg group, 40% in the 100-mg group, and 12% in the placebo group reached the EASI-75 end point, and the differences in these response rates were statistically significant vs placebo (100 mg: 27.9% [95% CI, 17.4-38.3], P<.0001; 200 mg: 51.0% [95% CI, 40.5-61.5], P<.0001). Notably, 44% of patients using the 200-mg dose achieved almost complete or complete resolution of AD (IGA responders, improvement of ≥2 and IGA score of 0 or 1 at 12 weeks).7 In JADE-MONO-2, EASI-75 also was achieved significantly more frequently in the treatment groups compared with the placebo group at 12 weeks (200 mg: 61.0%; 100 mg: 44.5%; placebo: 10.4%; P<.001 vs placebo).8 Adjunctive therapy with topical corticosteroids was prohibited in both studies. A dose-dependent decrease in platelets was seen in both trials, as in the phase 2 trial that preceded them.10
The primary end point of the JADE COMPARE trial was to evaluate the efficacy of abrocitinib as compared with placebo at 12 weeks in adult patients with moderate to severe AD and in the setting of concomitant topical corticosteroid therapy.9 One of several secondary end points of this study compared the ability of dupilumab vs abrocitinib and placebo treatment groups to achieve itch reduction at 2 weeks, defined as 4-point improvement or more from baseline in the score on the Peak Pruritus Numerical Rating Scale (NRS), a well‐defined, reliable, sensitive, and valid scale for evaluating worst itch intensity in adults with moderate to severe AD.9,11 The primary end point was the same as in the other phase 3 studies and was met in the JADE COMPARE trial by all treatment arms. An EASI-75 was seen in 70.3% of patients treated with 200 mg of abrocitinib, 58.7% in the 100-mg abrocitinib group, 58.1% in the dupilumab group, and 27.1% in the placebo group (P<.001 for both abrocitinib doses vs placebo). Only the 200-mg dose of abrocitinib demonstrated superior itch response at week 2 compared with dupilumab (22.1% response rate difference [95% CI, 13.5-30.7; P<.001]). Both abrocitinib groups failed to demonstrate significant differences compared with dupilumab with respect to other secondary end points to include IGA response and EASI-75 at week 16.9
The most frequently reported treatment-associated adverse events were nausea, nasopharyngitis, upper respiratory tract infection, and headache, and the percentages were similar among trial groups.9 Acne was more frequently reported in the abrocitinib groups compared with placebo and the dupilumab group, and conjunctivitis was more frequently reported in the dupilumab group. Herpesvirus cutaneous infections were rare in the abrocitinib groups, as were other serious infections. No deaths, major adverse cardiovascular events (MACEs), or venous thromboembolic events (VTEs) occurred during the trial. Dose-dependent increases in creatinine phosphokinase were seen in the abrocitinib groups, whereas dose-dependent decreases were seen in platelet counts, with no patient demonstrating a platelet count below 75,000/mm3 during the study.9 Low-density lipoprotein cholesterol levels and high-density lipoprotein cholesterol levels increased in a dose-dependent manner as well, but the ratios of low-density lipoprotein to high-density lipoprotein were unchanged.9 The results of a phase 3, 92-week extension study, JADE EXTEND, were recently published and demonstrated a role for abrocitinib as a treatment for patients with moderate to severe AD, regardless of prior dupilumab response status.12
Upadacitinib, another selective JAK1 inhibitor, was approved following data from 2 replicate double-blind, phase 3, randomized, controlled trials—Measure Up 1 and Measure Up 2.13 Results demonstrated that monotherapy with once-daily upadacitinib 15 mg or 30 mg is an effective and well-tolerated treatment option for patients with moderate to severe AD vs placebo. All coprimary end points at week 16 were achieved in the upadacitinib groups in both trials. Acne, upper respiratory tract infections, nasopharyngitis, headache, and increase in serum creatinine phosphokinase levels were the most frequently reported adverse events. Rates of herpes zoster infection in upadacitinib groups were low.13
In the subsequent phase 3 AD Up trial, researchers evaluated the safety and efficacy of combination therapy with topical corticosteroids in patients aged 12 to 75 years.14 Upadacitinib groups again achieved the identical coprimary end points that were present in the Measure Up trials13 as well as all key secondary end points.14 Additionally, significant differences in secondary end points, such as a 4-point improvement in the Worst Pruritus NRS vs placebo, were noticed in both upadacitinib treatment groups as early as 1 week into the study (P<.0001), with maintenance of the effect through to week 16 (P<.0001).14 AD Up was followed by the Heads Up trial, a 24-week, phase 3, multicenter, double-blind, randomized, controlled trial comparing safety and efficacy of upadacitinib with dupilumab among 692 adults with moderate to severe AD.15 At week 16, a higher percentage of patients in the upadacitinib group achieved EASI-75 vs the dupilumab group (71.0% vs 61.1%, respectively; P=.006). The difference noted at week 2 was even more impressive, with 43.7% of patients in the upadacitinib treatment group achieving EASI-75 compared with 17.4% in the dupilumab group (P<.001). No new safety-related events were registered compared with the already available data for both drugs.15
Ruxolitinib (RUX) is a topical JAK1 and JAK2 inhibitor that was FDA approved in September 2021 for the treatment of AD.16 In a phase 2 clinical trial of 307 adult patients with 3% to 20% body surface area (BSA) affected with AD, significant reductions in itch NRS scores were observed within 36 hours after the first application of RUX cream 1.5% twice daily (-1.8 vs -0.2, P<.0001).17 These decreases were noted within the first 2 weeks of treatment for all the RUX cream regimens and were sustained through to week 8, the end of the double-blind period. At 4 weeks, change in itch from baseline was significantly reduced in the RUX 1.5% twice-daily group compared with the triamcinolone ointment 0.1% group (−4 vs −2.5, P=.003). During the open-label treatment period from 8 to 12 weeks, all patients who switched to RUX cream 1.5% twice daily noted further reductions in itch, and those who continued it demonstrated additional improvement.17
The recent FDA approval was further backed by positive phase 3 trial data from the TRuE-AD1 and TRuE-AD2 studies.18 Patients in these trials were aged 12 years and older and had AD for 2 or more years with an IGA score of 2 or 3 and 3% to 20% affected BSA. Patients were randomized to twice-daily RUX cream 0.75%, RUX cream 1.5%, or vehicle cream, and the primary end point was an IGA score of 0 or 1 and an improvement of 2 or more points from baseline at week 8. Significantly more patients achieved IGA treatment success with RUX cream 0.75% (TRuE-AD1, 50.0%; TRuE-AD2, 39.0%) and RUX cream 1.5% (TRuE-AD1, 53.8%; TRuE-AD2, 51.3%) vs vehicle (TRuE-AD1, 15.1%; TRuE-AD2, 7.6%; P<.0001) at week 8. The RUX groups experienced dramatically reduced itch compared with vehicle, with a mean reduction of approximately 3 points on the NRS at 8 weeks. Additionally, statistically significant itch reductions vs vehicle were reported within 12 hours of first application of RUX cream 1.5% (P<.05). Application-site reactions including stinging and burning occurred in less than 1% of patients, and none were considered clinically significant. Mean plasma concentrations of RUX were monitored during the phase 2 and 3 AD studies and did not lead to any clinically meaningful changes in hematologic parameters. The low bioavailability following topical application of RUX cream (6% in the TRuE-AD studies) allows for a targeted delivery of the active drug to lesional skin while reducing the safety issues associated with oral administration of JAK inhibitors.18
Baricitinib is a predominantly JAK1 and JAK2 inhibitor that was the first JAK inhibitor to be approved for the treatment of moderate to severe AD in the European Union and Japan.19 Although the FDA’s decision on baricitinib has lagged behind market competitors, in 2 phase 3 clinical trials, BREEZE-AD1 and BREEZE-AD2, baricitinib demonstrated benefit over placebo on clinically important measures of disease severity. The primary end point—the proportion of patients achieving an IGA score of 0 or 1 with an improvement of 2 or more points from baseline at week 16—was met by both tested doses of baricitinib (2 mg and 4 mg) vs placebo in BREEZE-AD1 (2 mg, P≤.05; 4 mg, P≤.001) and BREEZE-AD2 (2 mg, P≤.05; 4 mg, P≤.001). In addition, baricitinib 4 mg consistently demonstrated significant benefit over placebo on other clinically important measures of disease severity at week 16 to include itch (BREEZE-AD1 and BREEZE-AD2, P≤.001), sleep disturbance (BREEZE-AD1, P≤.01; BREEZE-AD2, P≤.001), and skin pain (BREEZE-AD1, P≤.01; BREEZE-AD2, P≤.001). Nasopharyngitis, upper respiratory tract infections, creatine phosphokinase elevations, and headaches were the most frequently reported adverse events. During the 16-week treatment period in these trials, no deaths, MACEs, or VTEs occurred.19 Similar results were seen in a long-term extension study, BREEZE-AD3.20 The combination of baricitinib and topical corticosteroids were evaluated in 2 additional phase 3 trials, BREEZE-AD421 and BREEZE-AD7.22 Although only baricitinib 4 mg met the primary end point of EASI-75 at week 16 in both trials, both dosing regimens plus topical corticosteroids demonstrated notable reduction in multiple clinical and quality-of-life indices prior to week 2 when compared with placebo plus topical corticosteroids.22,23
AD in Military Service Members
Atopic dermatitis is a common condition in the general population, with a prevalence of 7.3% (95% CI, 5.9-8.8) in a recent study of American adults.24 Historically, the burden of AD that would be expected among active-duty military service members given the prevalence among the general population has not been observed, in part because of the disqualifying nature of AD for enlistment.25 The Department of Defense Instruction 6130.03, Volume 1, Medical Standards for Military Service: Appointment, Enlistment, or Induction stipulates that a history of AD or eczema after the twelfth birthday or history of residual or recurrent lesions in characteristic areas (ie, face, neck, antecubital or popliteal fossae, occasionally wrists and hands) is disqualifying.26 Specific military services possess additional standards that further define limits within the aforementioned Department of Defense instruction.25 Additionally, there are service-specific policies in place that mandate medical evaluation boards to determine fitness for continued service in the event the condition interferes with the member’s ability to perform their duties. Insection 4.2 of the U.S. Navy Aeromedical Reference and Waiver Guide, further restrictions for aviation personnel are delineated: “Depending on the location of lesions, there can be interference with the wearing of flight gear. The symptoms, particularly itching, can be distracting in flight. Patients with atopic dermatitis are more susceptible to contact dermatitis due to irritants found in a military environment.” Ultimately, the document stipulates that symptom severity and the requirement for therapy will determine the aeromedical disposition. It specifically states that “[p]atients controlled on topical therapy over small areas and patients who are asymptomatic on stable doses of loratadine (Claritin) OR fexofenadine (Allegra) may be considered for waiver,” and “intermittent use of topical steroids over a limited area is compatible with waiver.”27 It follows that limited use of topical JAK inhibitors, such as RUX, would be compatible with a waiver, given the favorable side effect profile and requirement for use in patients with 20% or lower affected BSA.16 This is just one example of duty-specific and service-specific medical standards that exist that could impact the use of both topical and oral JAK inhibitors.
Use of oral JAK inhibitors in active-duty service members is less ideal for multiple reasons. A large randomized safety clinical trial of patients with rheumatoid arthritis who received tofacitinib and methotrexate was required by the FDA to evaluate the risk of MACEs, malignancy, and infections associated with JAK inhibitor treatment. Data from this trial showed a dose-dependent increased risk for MACEs, all-cause mortality, and thrombosis at both doses of tofacitinib compared with tumor necrosis factor inhibitors and a non–dose-dependent increased risk for malignancy excluding nonmelanoma skin cancer.28 In contrast to the MACE and VTE data from patients with diseases other than AD treated with JAK inhibitors, there has been only 1 patient who developed a pulmonary embolism while being treated with baricitinib 4 mg.22,29 Downstream effects from the above study were label recommendations to reserve the medicines for patients who had an inadequate response or intolerance to 1 or more tumor necrosis factor blockers and to carefully consider risks vs benefits in patients, in particular current or prior smokers, those with other cardiovascular risk factors or a history of VTE, and those with a malignancy history other than already treated nonmelanoma skin cancer.28
There are consistent observations of laboratory abnormalities with JAK inhibitors, as discussed above, to include creatine phosphokinase elevation and cytopenias.30 Although existing data demonstrate that cytopenias are less of a concern in the AD population compared with the rheumatoid arthritis population, baseline and periodic laboratory monitoring are still recommended. In general, pretreatment laboratory assessment prior to initiating an oral JAK inhibitor should consist of a complete blood cell count with differential, complete metabolic panel, tuberculosis screening, chronic hepatitis panel, HIV screening, and a fasting lipid panel.2 The feasibility of obtaining these laboratory measurements in an operational setting or sea-going platform is limited, but many deployed locations and naval vessels possess the laboratory capability to perform a complete blood cell count and complete metabolic panel. Overall tolerability of oral JAK inhibitors in the treatment of AD appears favorable based on studies that were mostly 16 weeks in duration. Few recent longer-term studies have confirmed this side effect profile, but additional studies are needed.
Final Thoughts
Janus kinase inhibitors are a promising therapeutic class with multiple recently FDA-approved agents for the treatment of moderate to severe AD, with new agents on the horizon. Available efficacy data are promising and balanced by a favorable safety profile in clinical trials to date. The oral and topical bioavailability of JAK inhibitors makes them attractive alternatives to existing therapies. The rapidity of itch reduction and AD improvement demonstrated in multiple trials has the potential to decrease the length of limited-duty assignments, potentially returning treated service members to full-duty status more expeditiously. Other applications include use of these medications in scenarios where injectable medications are either unavailable or unsupported.
In the active-duty population, both the condition and/or the treatment may be duty limiting. Service members with AD who require more than topical treatment may require a medical evaluation board to determine if they are still fit to serve. The deployed environment routinely exacerbates AD and exposes service members to infections and environments where immunosuppression can create more risks than in the general population. Nonbiologic medications, which do not require refrigeration, are an exciting option for our patients with AD, including those actively serving or considering serving in the military. However, all factors in any patient’s life should be considered. Therefore, it is important for the nonmilitary dermatologist to work with local military physicians and the patient to determine the optimal treatment regimen to result in the best possible outcome.
The atopic dermatitis (AD) therapeutic landscape is changing considerably with the advent of Janus kinase (JAK) inhibitors. Several JAK inhibitors recently have been approved by the US Food and Drug Administration, building off years of foundational research aimed at elucidating the downstream effects of the JAK–signal transducer and activator of transcription (STAT) pathway and its role in AD pathogenesis. Agents within this promising new class of drugs have performed well vs placebo in phase 2 and 3 clinical trials. This article reviews relevant trial efficacy and safety data of several JAK inhibitors as well as the implications of the use of these medications in AD patients, with specific considerations unique to active-duty military personnel.
Background on JAK Inhibitors
The hematopoietin superfamily of cytokine receptors encompasses a broad group that includes receptors for immune (eg, IL-2, IL-4, IFN-γ), hematopoietic (eg, erythropoietin, thrombopoietin, granulocyte-macrophage colony-stimulating factor), and nonimmune (eg, prolactin, leptin, growth hormone) cytokines. These cytokines signal via the JAK-STAT pathway. The hematopoietin family of cytokine receptors lacks intrinsic enzymatic activity, and as a result, they rely on JAK enzymes to transmit their signals intracellularly after cytokine binding to the receptor.1 Janus, of Roman mythology, was the god of doorways and archways and was commonly depicted with 2 heads. Janus kinases were named for their 2 “faces,” the kinase domain with its adjacent regulatory kinaselike domains.2 The binding of a cytokine to its receptor triggers engagement of the receptor by JAKs, leading to phosphorylation of both the JAKs and the receptor. Subsequent recruitment and phosphorylation of STAT proteins occurs. Following STAT phosphorylation, the STAT proteins dissociate, dimerize, and translocate to the nucleus, where they enact changes in cell behavior through transcriptional effects.1
Humans possess only 4 JAKs. Janus kinase 1, JAK2, and tyrosine kinase 2 are widely expressed, whereas JAK3 expression is largely limited to immune cells. Thus, there is notable overlap in the use of the 4 JAKs among the relatively larger number of various cytokines that utilize them to propagate intracellular signaling.1 Janus kinase 1 is important for signaling of receptors activated by a variety of interleukins, as well as IFN-α, IFN-β, and IFN-γ. Janus kinase 2 is important for signaling for the hormonelike cytokines erythropoietin, thrombopoietin, growth hormone, granulocyte-macrophage colony-stimulating factor, IL-3, and IL-5. Janus kinase 3 is important for hematopoietic cell proliferation and function.1
JAK Inhibitors and Atopic Dermatitis
Topical treatments, including corticosteroids and calcineurin inhibitors, are considered the standard-of-care therapy for most patients with AD; however, their clinical benefit often is limited by their anatomic use restrictions and local adverse events, including skin atrophy, striae, and application-site reactions such as stinging and burning.3 As a result, long-term application of these drugs, particularly in sensitive areas, is not recommended owing to safety/tolerability issues.3 Systemic immunomodulatory medications are indicated for patients with AD who do not achieve adequate disease control with topical treatments and/or phototherapy or for patients with severely impaired quality of life.4
Janus kinase inhibitors have several key benefits over biologics: oral and topical bioavailability, predictable pharmacokinetics, nonimmunogenicity, and dosing flexibility.4 Janus kinase 1 is central to the cell signaling of many cytokines involved in the pathogenesis of AD that comprise the T-helper lymphocytes type 2 axis: IL-4, IL-13, and thymic stromal lymphopoietin. Janus kinase signaling also may mediate itch responses by acting directly on sensory nerve fibers. Consequently, the substantial reduction in pruritus seen in many studies of JAK inhibitors is thought to be in part due to the effects on sensory nerve fibers in the skin and the blockade of early itch signaling in response to IL-4, IL-13, and IL-31.5
Abrocitinib is a JAK1 inhibitor with a similar side effect profile to upadacitinib. Both agents were approved by the FDA for the treatment of refractory moderate to severe AD on January 14, 2022.6 These are second-generation (also referred to as selective) oral JAK inhibitors with much greater inhibitory potency for JAK1 than for JAK2, JAK3, or tyrosine kinase 2, thereby reducing the risk for hematopoietic effects associated with JAK2 inhibition. The approval of abrocitinib stemmed from the phase 3 clinical trial JAK1 Atopic Dermatitis Efficacy and Safety (JADE)-MONO-1 (N=387),7 its replicate trial JADE-MONO-2 (N=391),8 and the JADE COMPARE trial.9 The JADE-MONO trials were multicenter, double-blind, placebo-controlled studies that enrolled patients 12 years and older with moderate to severe AD.7,8 Treatment groups consisted of 100-mg and 200-mg doses and were evaluated with the placebo group for their ability to achieve an investigator global assessment (IGA) score of 0 or 1 and eczema area and severity index 75 (EASI-75) at 12 weeks.7,8 Sixty-three percent of patients in the 200-mg group, 40% in the 100-mg group, and 12% in the placebo group reached the EASI-75 end point, and the differences in these response rates were statistically significant vs placebo (100 mg: 27.9% [95% CI, 17.4-38.3], P<.0001; 200 mg: 51.0% [95% CI, 40.5-61.5], P<.0001). Notably, 44% of patients using the 200-mg dose achieved almost complete or complete resolution of AD (IGA responders, improvement of ≥2 and IGA score of 0 or 1 at 12 weeks).7 In JADE-MONO-2, EASI-75 also was achieved significantly more frequently in the treatment groups compared with the placebo group at 12 weeks (200 mg: 61.0%; 100 mg: 44.5%; placebo: 10.4%; P<.001 vs placebo).8 Adjunctive therapy with topical corticosteroids was prohibited in both studies. A dose-dependent decrease in platelets was seen in both trials, as in the phase 2 trial that preceded them.10
The primary end point of the JADE COMPARE trial was to evaluate the efficacy of abrocitinib as compared with placebo at 12 weeks in adult patients with moderate to severe AD and in the setting of concomitant topical corticosteroid therapy.9 One of several secondary end points of this study compared the ability of dupilumab vs abrocitinib and placebo treatment groups to achieve itch reduction at 2 weeks, defined as 4-point improvement or more from baseline in the score on the Peak Pruritus Numerical Rating Scale (NRS), a well‐defined, reliable, sensitive, and valid scale for evaluating worst itch intensity in adults with moderate to severe AD.9,11 The primary end point was the same as in the other phase 3 studies and was met in the JADE COMPARE trial by all treatment arms. An EASI-75 was seen in 70.3% of patients treated with 200 mg of abrocitinib, 58.7% in the 100-mg abrocitinib group, 58.1% in the dupilumab group, and 27.1% in the placebo group (P<.001 for both abrocitinib doses vs placebo). Only the 200-mg dose of abrocitinib demonstrated superior itch response at week 2 compared with dupilumab (22.1% response rate difference [95% CI, 13.5-30.7; P<.001]). Both abrocitinib groups failed to demonstrate significant differences compared with dupilumab with respect to other secondary end points to include IGA response and EASI-75 at week 16.9
The most frequently reported treatment-associated adverse events were nausea, nasopharyngitis, upper respiratory tract infection, and headache, and the percentages were similar among trial groups.9 Acne was more frequently reported in the abrocitinib groups compared with placebo and the dupilumab group, and conjunctivitis was more frequently reported in the dupilumab group. Herpesvirus cutaneous infections were rare in the abrocitinib groups, as were other serious infections. No deaths, major adverse cardiovascular events (MACEs), or venous thromboembolic events (VTEs) occurred during the trial. Dose-dependent increases in creatinine phosphokinase were seen in the abrocitinib groups, whereas dose-dependent decreases were seen in platelet counts, with no patient demonstrating a platelet count below 75,000/mm3 during the study.9 Low-density lipoprotein cholesterol levels and high-density lipoprotein cholesterol levels increased in a dose-dependent manner as well, but the ratios of low-density lipoprotein to high-density lipoprotein were unchanged.9 The results of a phase 3, 92-week extension study, JADE EXTEND, were recently published and demonstrated a role for abrocitinib as a treatment for patients with moderate to severe AD, regardless of prior dupilumab response status.12
Upadacitinib, another selective JAK1 inhibitor, was approved following data from 2 replicate double-blind, phase 3, randomized, controlled trials—Measure Up 1 and Measure Up 2.13 Results demonstrated that monotherapy with once-daily upadacitinib 15 mg or 30 mg is an effective and well-tolerated treatment option for patients with moderate to severe AD vs placebo. All coprimary end points at week 16 were achieved in the upadacitinib groups in both trials. Acne, upper respiratory tract infections, nasopharyngitis, headache, and increase in serum creatinine phosphokinase levels were the most frequently reported adverse events. Rates of herpes zoster infection in upadacitinib groups were low.13
In the subsequent phase 3 AD Up trial, researchers evaluated the safety and efficacy of combination therapy with topical corticosteroids in patients aged 12 to 75 years.14 Upadacitinib groups again achieved the identical coprimary end points that were present in the Measure Up trials13 as well as all key secondary end points.14 Additionally, significant differences in secondary end points, such as a 4-point improvement in the Worst Pruritus NRS vs placebo, were noticed in both upadacitinib treatment groups as early as 1 week into the study (P<.0001), with maintenance of the effect through to week 16 (P<.0001).14 AD Up was followed by the Heads Up trial, a 24-week, phase 3, multicenter, double-blind, randomized, controlled trial comparing safety and efficacy of upadacitinib with dupilumab among 692 adults with moderate to severe AD.15 At week 16, a higher percentage of patients in the upadacitinib group achieved EASI-75 vs the dupilumab group (71.0% vs 61.1%, respectively; P=.006). The difference noted at week 2 was even more impressive, with 43.7% of patients in the upadacitinib treatment group achieving EASI-75 compared with 17.4% in the dupilumab group (P<.001). No new safety-related events were registered compared with the already available data for both drugs.15
Ruxolitinib (RUX) is a topical JAK1 and JAK2 inhibitor that was FDA approved in September 2021 for the treatment of AD.16 In a phase 2 clinical trial of 307 adult patients with 3% to 20% body surface area (BSA) affected with AD, significant reductions in itch NRS scores were observed within 36 hours after the first application of RUX cream 1.5% twice daily (-1.8 vs -0.2, P<.0001).17 These decreases were noted within the first 2 weeks of treatment for all the RUX cream regimens and were sustained through to week 8, the end of the double-blind period. At 4 weeks, change in itch from baseline was significantly reduced in the RUX 1.5% twice-daily group compared with the triamcinolone ointment 0.1% group (−4 vs −2.5, P=.003). During the open-label treatment period from 8 to 12 weeks, all patients who switched to RUX cream 1.5% twice daily noted further reductions in itch, and those who continued it demonstrated additional improvement.17
The recent FDA approval was further backed by positive phase 3 trial data from the TRuE-AD1 and TRuE-AD2 studies.18 Patients in these trials were aged 12 years and older and had AD for 2 or more years with an IGA score of 2 or 3 and 3% to 20% affected BSA. Patients were randomized to twice-daily RUX cream 0.75%, RUX cream 1.5%, or vehicle cream, and the primary end point was an IGA score of 0 or 1 and an improvement of 2 or more points from baseline at week 8. Significantly more patients achieved IGA treatment success with RUX cream 0.75% (TRuE-AD1, 50.0%; TRuE-AD2, 39.0%) and RUX cream 1.5% (TRuE-AD1, 53.8%; TRuE-AD2, 51.3%) vs vehicle (TRuE-AD1, 15.1%; TRuE-AD2, 7.6%; P<.0001) at week 8. The RUX groups experienced dramatically reduced itch compared with vehicle, with a mean reduction of approximately 3 points on the NRS at 8 weeks. Additionally, statistically significant itch reductions vs vehicle were reported within 12 hours of first application of RUX cream 1.5% (P<.05). Application-site reactions including stinging and burning occurred in less than 1% of patients, and none were considered clinically significant. Mean plasma concentrations of RUX were monitored during the phase 2 and 3 AD studies and did not lead to any clinically meaningful changes in hematologic parameters. The low bioavailability following topical application of RUX cream (6% in the TRuE-AD studies) allows for a targeted delivery of the active drug to lesional skin while reducing the safety issues associated with oral administration of JAK inhibitors.18
Baricitinib is a predominantly JAK1 and JAK2 inhibitor that was the first JAK inhibitor to be approved for the treatment of moderate to severe AD in the European Union and Japan.19 Although the FDA’s decision on baricitinib has lagged behind market competitors, in 2 phase 3 clinical trials, BREEZE-AD1 and BREEZE-AD2, baricitinib demonstrated benefit over placebo on clinically important measures of disease severity. The primary end point—the proportion of patients achieving an IGA score of 0 or 1 with an improvement of 2 or more points from baseline at week 16—was met by both tested doses of baricitinib (2 mg and 4 mg) vs placebo in BREEZE-AD1 (2 mg, P≤.05; 4 mg, P≤.001) and BREEZE-AD2 (2 mg, P≤.05; 4 mg, P≤.001). In addition, baricitinib 4 mg consistently demonstrated significant benefit over placebo on other clinically important measures of disease severity at week 16 to include itch (BREEZE-AD1 and BREEZE-AD2, P≤.001), sleep disturbance (BREEZE-AD1, P≤.01; BREEZE-AD2, P≤.001), and skin pain (BREEZE-AD1, P≤.01; BREEZE-AD2, P≤.001). Nasopharyngitis, upper respiratory tract infections, creatine phosphokinase elevations, and headaches were the most frequently reported adverse events. During the 16-week treatment period in these trials, no deaths, MACEs, or VTEs occurred.19 Similar results were seen in a long-term extension study, BREEZE-AD3.20 The combination of baricitinib and topical corticosteroids were evaluated in 2 additional phase 3 trials, BREEZE-AD421 and BREEZE-AD7.22 Although only baricitinib 4 mg met the primary end point of EASI-75 at week 16 in both trials, both dosing regimens plus topical corticosteroids demonstrated notable reduction in multiple clinical and quality-of-life indices prior to week 2 when compared with placebo plus topical corticosteroids.22,23
AD in Military Service Members
Atopic dermatitis is a common condition in the general population, with a prevalence of 7.3% (95% CI, 5.9-8.8) in a recent study of American adults.24 Historically, the burden of AD that would be expected among active-duty military service members given the prevalence among the general population has not been observed, in part because of the disqualifying nature of AD for enlistment.25 The Department of Defense Instruction 6130.03, Volume 1, Medical Standards for Military Service: Appointment, Enlistment, or Induction stipulates that a history of AD or eczema after the twelfth birthday or history of residual or recurrent lesions in characteristic areas (ie, face, neck, antecubital or popliteal fossae, occasionally wrists and hands) is disqualifying.26 Specific military services possess additional standards that further define limits within the aforementioned Department of Defense instruction.25 Additionally, there are service-specific policies in place that mandate medical evaluation boards to determine fitness for continued service in the event the condition interferes with the member’s ability to perform their duties. Insection 4.2 of the U.S. Navy Aeromedical Reference and Waiver Guide, further restrictions for aviation personnel are delineated: “Depending on the location of lesions, there can be interference with the wearing of flight gear. The symptoms, particularly itching, can be distracting in flight. Patients with atopic dermatitis are more susceptible to contact dermatitis due to irritants found in a military environment.” Ultimately, the document stipulates that symptom severity and the requirement for therapy will determine the aeromedical disposition. It specifically states that “[p]atients controlled on topical therapy over small areas and patients who are asymptomatic on stable doses of loratadine (Claritin) OR fexofenadine (Allegra) may be considered for waiver,” and “intermittent use of topical steroids over a limited area is compatible with waiver.”27 It follows that limited use of topical JAK inhibitors, such as RUX, would be compatible with a waiver, given the favorable side effect profile and requirement for use in patients with 20% or lower affected BSA.16 This is just one example of duty-specific and service-specific medical standards that exist that could impact the use of both topical and oral JAK inhibitors.
Use of oral JAK inhibitors in active-duty service members is less ideal for multiple reasons. A large randomized safety clinical trial of patients with rheumatoid arthritis who received tofacitinib and methotrexate was required by the FDA to evaluate the risk of MACEs, malignancy, and infections associated with JAK inhibitor treatment. Data from this trial showed a dose-dependent increased risk for MACEs, all-cause mortality, and thrombosis at both doses of tofacitinib compared with tumor necrosis factor inhibitors and a non–dose-dependent increased risk for malignancy excluding nonmelanoma skin cancer.28 In contrast to the MACE and VTE data from patients with diseases other than AD treated with JAK inhibitors, there has been only 1 patient who developed a pulmonary embolism while being treated with baricitinib 4 mg.22,29 Downstream effects from the above study were label recommendations to reserve the medicines for patients who had an inadequate response or intolerance to 1 or more tumor necrosis factor blockers and to carefully consider risks vs benefits in patients, in particular current or prior smokers, those with other cardiovascular risk factors or a history of VTE, and those with a malignancy history other than already treated nonmelanoma skin cancer.28
There are consistent observations of laboratory abnormalities with JAK inhibitors, as discussed above, to include creatine phosphokinase elevation and cytopenias.30 Although existing data demonstrate that cytopenias are less of a concern in the AD population compared with the rheumatoid arthritis population, baseline and periodic laboratory monitoring are still recommended. In general, pretreatment laboratory assessment prior to initiating an oral JAK inhibitor should consist of a complete blood cell count with differential, complete metabolic panel, tuberculosis screening, chronic hepatitis panel, HIV screening, and a fasting lipid panel.2 The feasibility of obtaining these laboratory measurements in an operational setting or sea-going platform is limited, but many deployed locations and naval vessels possess the laboratory capability to perform a complete blood cell count and complete metabolic panel. Overall tolerability of oral JAK inhibitors in the treatment of AD appears favorable based on studies that were mostly 16 weeks in duration. Few recent longer-term studies have confirmed this side effect profile, but additional studies are needed.
Final Thoughts
Janus kinase inhibitors are a promising therapeutic class with multiple recently FDA-approved agents for the treatment of moderate to severe AD, with new agents on the horizon. Available efficacy data are promising and balanced by a favorable safety profile in clinical trials to date. The oral and topical bioavailability of JAK inhibitors makes them attractive alternatives to existing therapies. The rapidity of itch reduction and AD improvement demonstrated in multiple trials has the potential to decrease the length of limited-duty assignments, potentially returning treated service members to full-duty status more expeditiously. Other applications include use of these medications in scenarios where injectable medications are either unavailable or unsupported.
In the active-duty population, both the condition and/or the treatment may be duty limiting. Service members with AD who require more than topical treatment may require a medical evaluation board to determine if they are still fit to serve. The deployed environment routinely exacerbates AD and exposes service members to infections and environments where immunosuppression can create more risks than in the general population. Nonbiologic medications, which do not require refrigeration, are an exciting option for our patients with AD, including those actively serving or considering serving in the military. However, all factors in any patient’s life should be considered. Therefore, it is important for the nonmilitary dermatologist to work with local military physicians and the patient to determine the optimal treatment regimen to result in the best possible outcome.
- Damsky W, Peterson D, Ramseier J, et al. The emerging role of Janus kinase inhibitors in the treatment of autoimmune and inflammatory diseases. J Allergy Clin Immunol. 2021;147:814-826.
- Gadina M, Le MT, Schwartz DM, et al. Janus kinases to jakinibs: from basic insights to clinical practice. Rheumatology (Oxford). 2019;58(suppl 1):i4-i6.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2, management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Cartron AM, Nguyen TH, Roh YS, et al. Janus kinase inhibitors for atopic dermatitis: a promising treatment modality. Clin Exp Dermatol. 2021;46:820-824.
- Oetjen LK, Mack MR, Feng J, et al. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell. 2017;171:217-228.e13.
- U.S. FDA approves Pfizer’s CIBINQO® (abrocitinib) for adults with moderate-to-severe atopic dermatitis [press release]. January 14, 2022. Accessed November 18, 2022. https://www.pfizer.com/news/press-release/press-release-detail/us-fda-approves-pfizers-cibinqor-abrocitinib-adults
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266.
- Silverberg JI, Simpson EL, Thyssen JP, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:863-873.
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112.
- Gooderham MJ, Forman SB, Bissonnette R, et al. Efficacy and safety of oral Janus kinase 1 inhibitor abrocitinib for patients with atopic dermatitis: a phase 2 randomized clinical trial. JAMA Dermatol. 2019;155:1371-1379. Published correction appears in JAMA Dermatol. 2020;156:104.
- Yosipovitch G, Reaney M, Mastey V, et al. Peak Pruritus Numerical Rating Scale: psychometric validation and responder definition for assessing itch in moderate-to-severe atopic dermatitis. Br J Dermatol. 2019;181:761-769.
- Shi VY, Bhutani T, Fonacier L, et al. Phase 3 efficacy and safety of abrocitinib in adults with moderate-to-severe atopic dermatitis after switching from dupilumab (JADE EXTEND). J Am Acad Dermatol. 2022;87:351-358.
- Guttman-Yassky E, Teixeira HD, Simpson EL, et al. Once-daily upadacitinib versus placebo in adolescents and adults with moderate-to-severe atopic dermatitis (Measure Up 1 and Measure Up 2): results from two replicate double-blind, randomised controlled phase 3 trials. Lancet. 2021;397:2151-2168.
- Reich K, Teixeira HD, de Bruin-Weller M, et al. Safety and efficacy of upadacitinib in combination with topical corticosteroids in adolescents and adults with moderate-to-severe atopic dermatitis (AD Up): results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021;397:2169-2181.
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2021;157:1047-1055. Published correction appears in JAMA Dermatol. 2022;158:219.
- FDA approves Opzelura. Drugs.com. September 21, 2021. Accessed October 6, 2022. https://www.drugs.com/newdrugs/fda-approves-opzelura-ruxolitinib-cream-atopic-dermatitis-ad-5666.html
- Kim BS, Sun K, Papp K, et al. Effects of ruxolitinib cream on pruritus and quality of life in atopic dermatitis: results from a phase 2, randomized, doseranging, vehicle- and active-controlled study. J Am Acad Dermatol. 2020;82:1305-1313.
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies. J Am Acad Dermatol. 2021;85:863-872.
- Simpson EL, Lacour JP, Spelman L, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis and inadequate response to topical corticosteroids: results from two randomized monotherapy phase III trials. Br J Dermatol. 2020;183:242-255.
- Silverberg JI, Simpson EL, Wollenberg A, et al. Long-term efficacy of baricitinib in adults with moderate to severe atopic dermatitis who were treatment responders or partial responders: an extension study of 2 randomized clinical trials. JAMA Dermatol. 2021;157:691-699.
- Lilly and Incyte announce top-line results from phase 3 study (BREEZE-AD4) of oral selective JAK inhibitor baricitinib in combination with topical corticosteroids in patients with moderate to severe atopic dermatitis not controlled with cyclosporine. January 27, 2020. Accessed November 18, 2022. https://investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-announce-top-line-results-phase-3-study-breeze
- Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:1333-1343.
- Wollenberg A, Nakahara T, Maari C, et al. Impact of baricitinib in combination with topical steroids on atopic dermatitis symptoms, quality of life and functioning in adult patients with moderate-to-severe atopic dermatitis from the BREEZE-AD7 phase 3 randomized trial. J Eur Acad Dermatol Venereol. 2021;35:1543-1552.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590.
- Jeter J, Bowen C. Atopic dermatitis and implications for military service. Mil Med. 2019;184:E177-E182.
- Department of Defense. Medical standards for military service: appointment, enlistment, or induction. DoD Instruction 6130.03. Vol 1. May 6, 2022. Accessed November 18, 2022. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_v1p.PDF?ver=9NsVi30gsHBBsRhMLcyVVQ%3d%3d
- Dermatitis. In: U.S. Navy Aeromedical Reference and Waiver Guide. Navy Medicine Operational Training Command and Naval Aerospace Medical Institute. August 11, 2021. Accessed November 18, 2022. https://www.med.navy.mil/Portals/62/Documents/NMFSC/NMOTC/NAMI/ARWG/Waiver%20Guide/ARWG%20COMPLETE_210811.pdf?ver=_pLPzFrtl8E2swFESnN4rA%3D%3D
- FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. FDA Drug Safety Podcast. U.S. Food and Drug Administration. Updated January 14, 2022. Accessed November 18, 2022. https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Chang PH, Huang SF, Chang PS, et al. Safety considerations of systemic Janus kinase inhibitors in atopic dermatitis applications. J Dermatol. 2021;48:1631-1639.
- Wood H, Chandler A, Nezamololama N, et al. Safety of Janus kinase (JAK) inhibitors in the short-term treatment of atopic dermatitis. Int J Dermatol. 2022;61:746-754.
- Damsky W, Peterson D, Ramseier J, et al. The emerging role of Janus kinase inhibitors in the treatment of autoimmune and inflammatory diseases. J Allergy Clin Immunol. 2021;147:814-826.
- Gadina M, Le MT, Schwartz DM, et al. Janus kinases to jakinibs: from basic insights to clinical practice. Rheumatology (Oxford). 2019;58(suppl 1):i4-i6.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2, management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Cartron AM, Nguyen TH, Roh YS, et al. Janus kinase inhibitors for atopic dermatitis: a promising treatment modality. Clin Exp Dermatol. 2021;46:820-824.
- Oetjen LK, Mack MR, Feng J, et al. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell. 2017;171:217-228.e13.
- U.S. FDA approves Pfizer’s CIBINQO® (abrocitinib) for adults with moderate-to-severe atopic dermatitis [press release]. January 14, 2022. Accessed November 18, 2022. https://www.pfizer.com/news/press-release/press-release-detail/us-fda-approves-pfizers-cibinqor-abrocitinib-adults
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266.
- Silverberg JI, Simpson EL, Thyssen JP, et al. Efficacy and safety of abrocitinib in patients with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:863-873.
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112.
- Gooderham MJ, Forman SB, Bissonnette R, et al. Efficacy and safety of oral Janus kinase 1 inhibitor abrocitinib for patients with atopic dermatitis: a phase 2 randomized clinical trial. JAMA Dermatol. 2019;155:1371-1379. Published correction appears in JAMA Dermatol. 2020;156:104.
- Yosipovitch G, Reaney M, Mastey V, et al. Peak Pruritus Numerical Rating Scale: psychometric validation and responder definition for assessing itch in moderate-to-severe atopic dermatitis. Br J Dermatol. 2019;181:761-769.
- Shi VY, Bhutani T, Fonacier L, et al. Phase 3 efficacy and safety of abrocitinib in adults with moderate-to-severe atopic dermatitis after switching from dupilumab (JADE EXTEND). J Am Acad Dermatol. 2022;87:351-358.
- Guttman-Yassky E, Teixeira HD, Simpson EL, et al. Once-daily upadacitinib versus placebo in adolescents and adults with moderate-to-severe atopic dermatitis (Measure Up 1 and Measure Up 2): results from two replicate double-blind, randomised controlled phase 3 trials. Lancet. 2021;397:2151-2168.
- Reich K, Teixeira HD, de Bruin-Weller M, et al. Safety and efficacy of upadacitinib in combination with topical corticosteroids in adolescents and adults with moderate-to-severe atopic dermatitis (AD Up): results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021;397:2169-2181.
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2021;157:1047-1055. Published correction appears in JAMA Dermatol. 2022;158:219.
- FDA approves Opzelura. Drugs.com. September 21, 2021. Accessed October 6, 2022. https://www.drugs.com/newdrugs/fda-approves-opzelura-ruxolitinib-cream-atopic-dermatitis-ad-5666.html
- Kim BS, Sun K, Papp K, et al. Effects of ruxolitinib cream on pruritus and quality of life in atopic dermatitis: results from a phase 2, randomized, doseranging, vehicle- and active-controlled study. J Am Acad Dermatol. 2020;82:1305-1313.
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies. J Am Acad Dermatol. 2021;85:863-872.
- Simpson EL, Lacour JP, Spelman L, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis and inadequate response to topical corticosteroids: results from two randomized monotherapy phase III trials. Br J Dermatol. 2020;183:242-255.
- Silverberg JI, Simpson EL, Wollenberg A, et al. Long-term efficacy of baricitinib in adults with moderate to severe atopic dermatitis who were treatment responders or partial responders: an extension study of 2 randomized clinical trials. JAMA Dermatol. 2021;157:691-699.
- Lilly and Incyte announce top-line results from phase 3 study (BREEZE-AD4) of oral selective JAK inhibitor baricitinib in combination with topical corticosteroids in patients with moderate to severe atopic dermatitis not controlled with cyclosporine. January 27, 2020. Accessed November 18, 2022. https://investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-announce-top-line-results-phase-3-study-breeze
- Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2020;156:1333-1343.
- Wollenberg A, Nakahara T, Maari C, et al. Impact of baricitinib in combination with topical steroids on atopic dermatitis symptoms, quality of life and functioning in adult patients with moderate-to-severe atopic dermatitis from the BREEZE-AD7 phase 3 randomized trial. J Eur Acad Dermatol Venereol. 2021;35:1543-1552.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590.
- Jeter J, Bowen C. Atopic dermatitis and implications for military service. Mil Med. 2019;184:E177-E182.
- Department of Defense. Medical standards for military service: appointment, enlistment, or induction. DoD Instruction 6130.03. Vol 1. May 6, 2022. Accessed November 18, 2022. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_v1p.PDF?ver=9NsVi30gsHBBsRhMLcyVVQ%3d%3d
- Dermatitis. In: U.S. Navy Aeromedical Reference and Waiver Guide. Navy Medicine Operational Training Command and Naval Aerospace Medical Institute. August 11, 2021. Accessed November 18, 2022. https://www.med.navy.mil/Portals/62/Documents/NMFSC/NMOTC/NAMI/ARWG/Waiver%20Guide/ARWG%20COMPLETE_210811.pdf?ver=_pLPzFrtl8E2swFESnN4rA%3D%3D
- FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. FDA Drug Safety Podcast. U.S. Food and Drug Administration. Updated January 14, 2022. Accessed November 18, 2022. https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-requires-warnings-about-increased-risk-serious-heart-related-events-cancer-blood-clots-and-death
- Chang PH, Huang SF, Chang PS, et al. Safety considerations of systemic Janus kinase inhibitors in atopic dermatitis applications. J Dermatol. 2021;48:1631-1639.
- Wood H, Chandler A, Nezamololama N, et al. Safety of Janus kinase (JAK) inhibitors in the short-term treatment of atopic dermatitis. Int J Dermatol. 2022;61:746-754.
Practice Points
- Oral Janus kinase (JAK) inhibitors are novel therapies available for the treatment of atopic dermatitis (AD), with multiple recently approved agents within the class.
- Recommended laboratory monitoring during treatment with oral JAK inhibitors may limit the use of these medications in the active-duty military population or in those with special-duty assignments.
- The oral and topical bioavailability of these medications makes them a more feasible option for deploying service members or for those requiring flexible dosing.
- The rapid improvement in AD seen in multiple trials of oral JAK inhibitors suggests these agents could prove useful in management of acute AD flares, especially in military environments, where injectable agents are either unavailable or unsupported.
Advocacy Update: Ringing in 2023
New Year, New Codes: A Win-Win for Digital Pathology
In July 2022, the American Medical Association CPT (Current Procedural Terminology) Editorial Panel released 13 new digital pathology add-on Category III codes for 2023 that the College of American Pathologists successfully advocated for inclusion.1 These codes are for reporting additional clinical staff work and service requirements associated with digitizing glass microscope slides for primary diagnosis (Table). They go into effect on January 1, 2023.
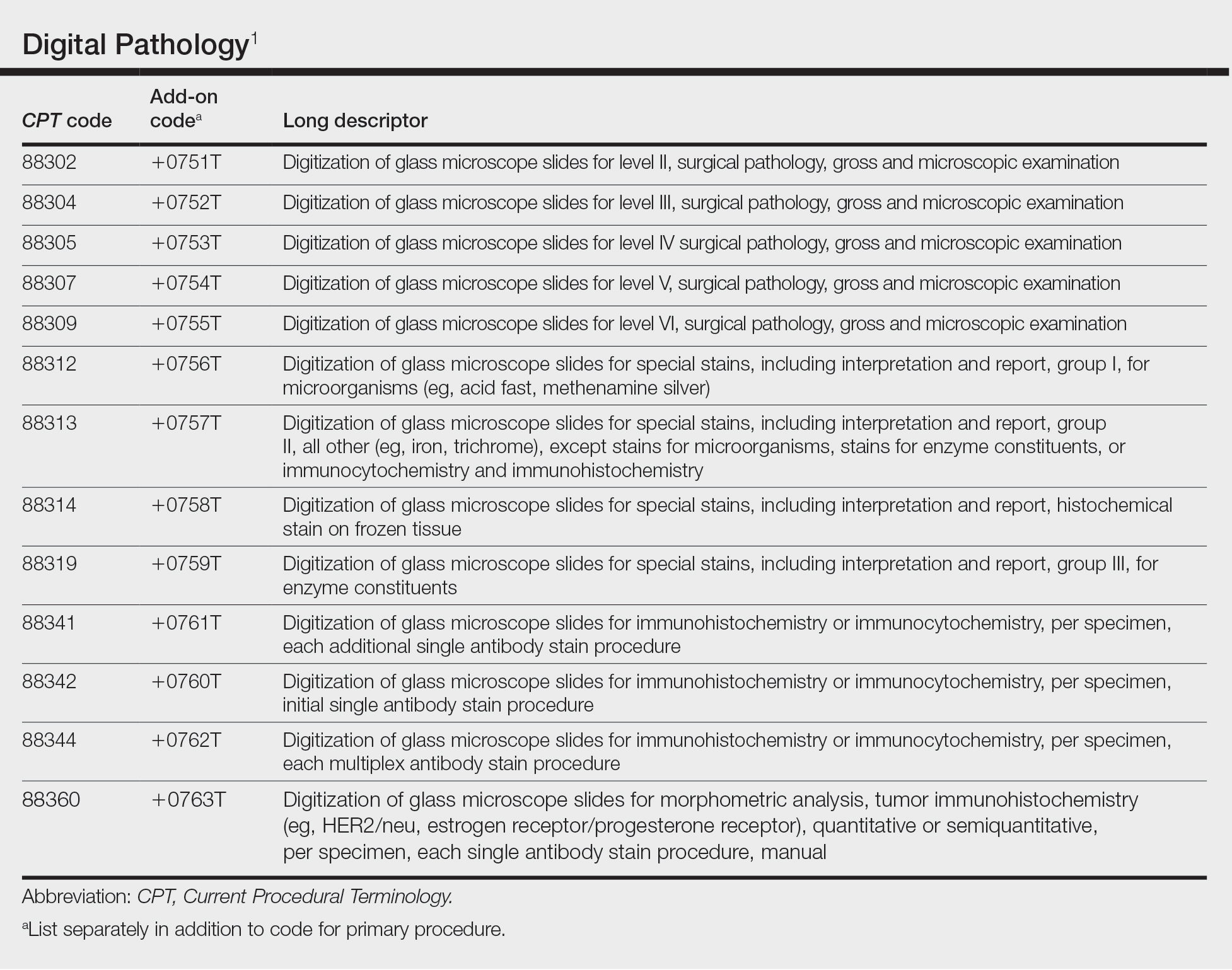
Although there is no additional compensation with the new Category III codes, dermatopathology laboratories will be able to report when they have made a diagnosis using digital pathology. The new CPT codes will provide payers with data they need to directly understand the utilization and increased value of digital pathology, which will bring dermatopathology laboratories one step closer to receiving additional reimbursement for digital interpretation.
The adoption of digital pathology has been accelerating in the United States but still lags behind many European countries where reimbursement for digital pathology has been established for many years. Many of the barriers to digital pathology have improved—cloud storage is more affordable, scanners have a higher throughput, digital pathology platforms have improved, and the US Food and Drug Administration has granted approvals for digital pathology. Digital pathology allows for more efficient workflow, which results in increased productivity and a reduction in turnaround times. It also allows for a wide spectrum of clinical applications and more innovation as well as research and educational applications.
The new Category III codes cannot be reported solely for archival purposes (eg, after the Category I service has already been performed and reported), solely for educational purposes (eg, when services are not used for individual patient reporting), solely for developing a database for training or validation of artificial intelligence algorithms, and solely for clinical conference presentations (eg, tumor board interdisciplinary conferences).
The new codes are a major victory for the adoption and future compensation for digital pathology.
New Year, New Cuts: Proposed 2023 Medicare Policy and Payment Changes for Dermatologists
The United States Spent $3.8 Trillion on Health Care in 2019: Where Did It Go?—In 2019, approximately $3.8 trillion was spent on health care in the United States (Figure 1). Physician services accounted for approximately 15% of total health care spending.2
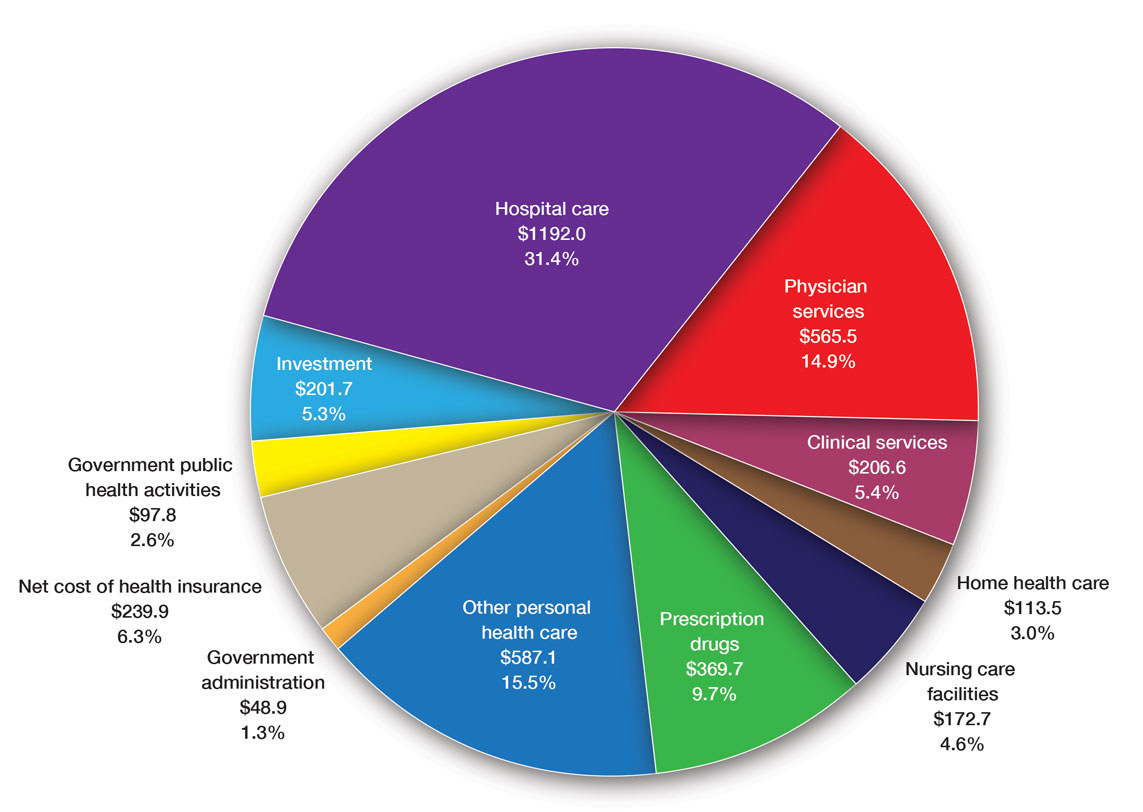
Medicare Payments for Physician Services—Medicare payments for physician services are determined by a relative value unit (RVU) multiplied by a conversion factor (CF). Relative value units were set up in 1992 by what is now the Centers for Medicare & Medicaid Services, and they calculated the time it took a physician to complete a task or RVU and multiplied it by $32.00 (CF).3
Thirty years later—in 2022—the CF is $34.61. If the CF had increased with inflation, it would be $59.00. If the Proposed Rule is adopted, the 2023 fee schedule payment formula would decrease by 4.4% (to $33.08) relative to that of the 2022 fee schedule ($34.61), which is a decrease of 8.2% since 2019 ($36.04). This decrease is due to expiration of the 3% increase to Medicare fee schedule payments for 2022 required by the Protecting Medicare and American Farmers from Sequester Cuts Act and the required budget neutrality adjustment required by changes in RVUs. Medicare physician payment has declined 22% from 2001 to 2022 (Figure 2).4,5
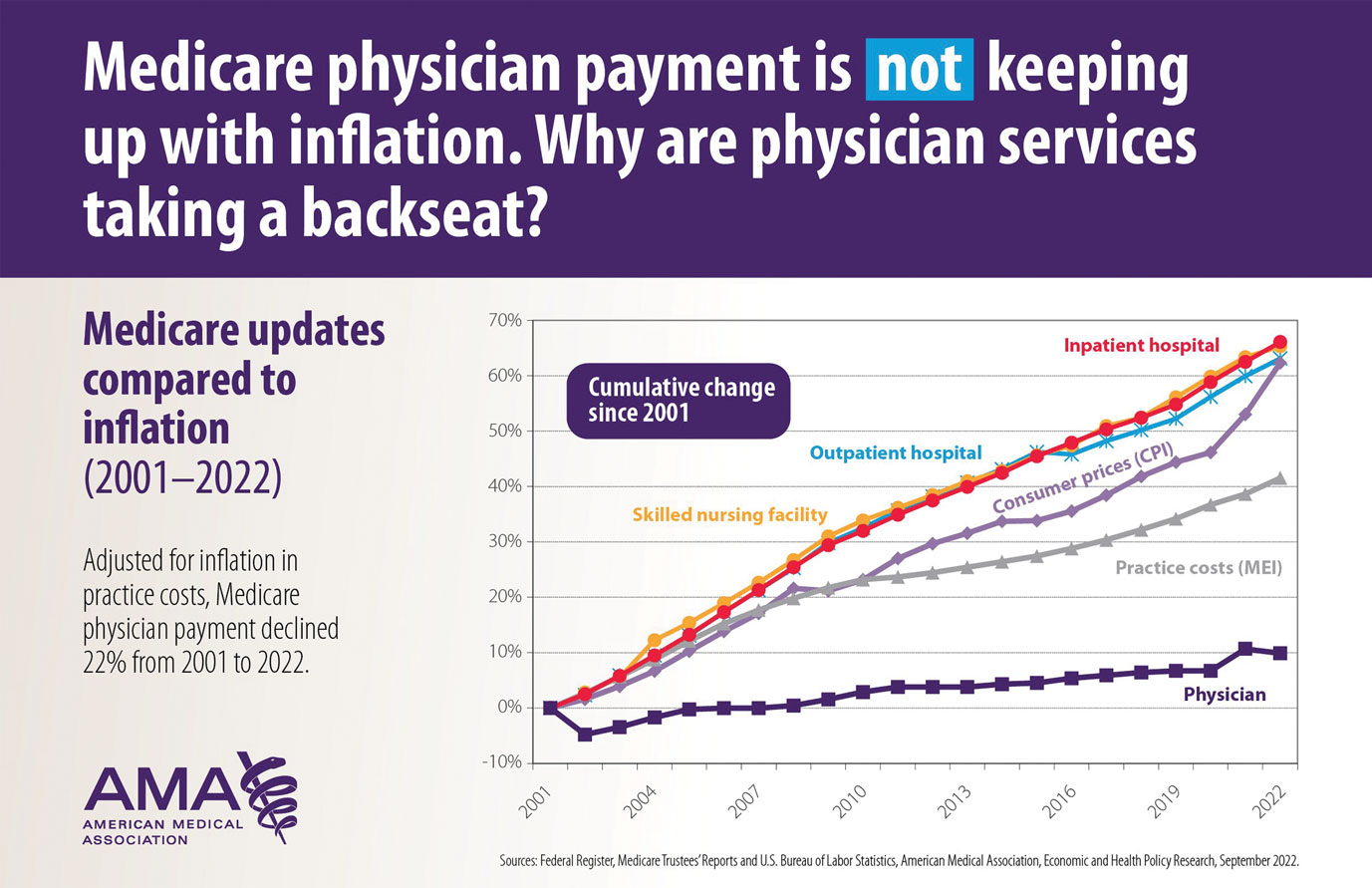
The adjustments to the CF typically are made based on 3 factors: (1) the Medicare Economic Index; (2) expenditure target “performance adjustment”; and (3) miscellaneous adjustments, including those for “budget neutrality” required by law.
Medicare Physician Payments Compared With Other Provider Types and Inflation—The proposed Medicare physician payment policy is unsustainable for outpatient dermatologists. Practice overhead has increased markedly since 1992. Other service providers, such as those in skilled nursing facilities and hospitals (Figure 3), have received favorable payment increases compared with practice cost inflation and the Consumer Price Index.3-6 Flat reimbursement affects all physicians who accept insurance, as even private insurers base their reimbursement on Medicare.
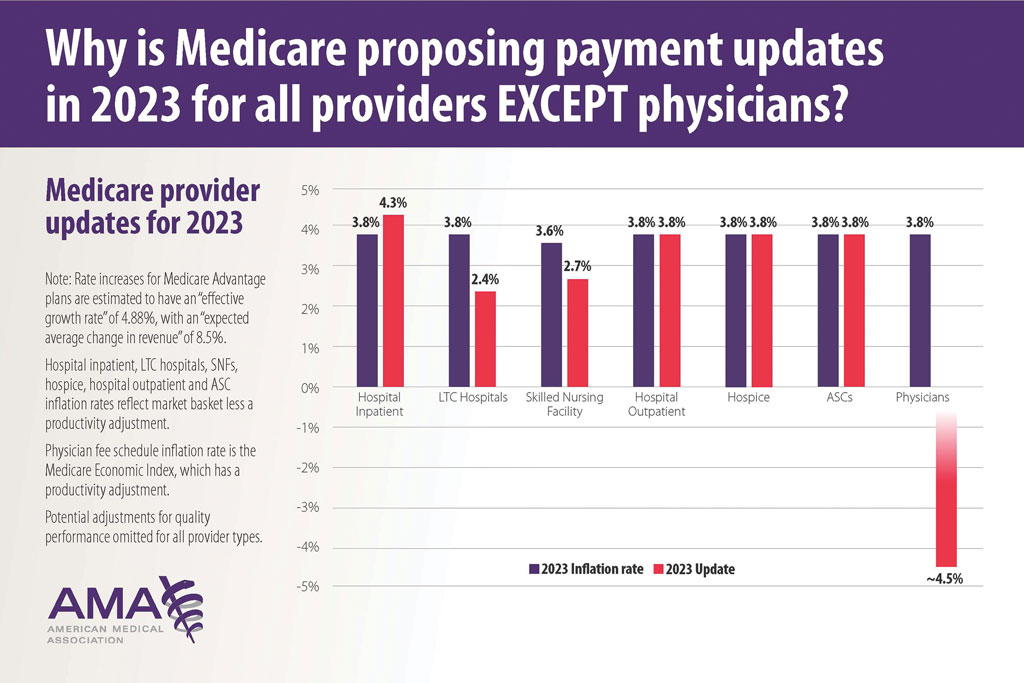
In addition, there are other issues resulting in decreased physician payments when evaluation and management services are reported with same-day procedures using modifier −25 as well as preserving or finding alternative strategies for 10- and 90-day global period payments for medical procedures. When Medicare cuts physician payments, dermatologists find it difficult to own and operate their own practices, resulting in health market consolidation, limited competition, increased health care costs, limited patient access to care, and decreased quality of health care.
Medicare Payment Reform—Medicare payment reform is necessary to stop annual payment cuts and create a stable predictable payment system that ensures patient access to quality, value-based care. Medicare physician payment reform needs to happen at a national level. The American Academy of Dermatology Association (AADA) is working with the House of Medicine and the medical specialty community to develop specific proposals, such as “Characteristics of a Rational Medicare Physician Payment System,” to reform Medicare’s payment system.7 Advocacy groups, including the AADA, have been working to mitigate the proposed 2023 cuts by engaging with Congress and urging them to act before these changes go into effect on January 1, 2023.
Make Advocacy Your New Year’s Resolution: AADA’s Top Advocacy Priorities
The AADA’s top priority is Medicare payment policies.3 In addition, the AADA is working on drug access and cost by cutting the bureaucratic red tape caused by prior authorization (PA) and step therapy policies. The AADA collaborates with manufacturers, the health care community, policymakers, private payers, pharmacists, pharmacy benefit managers, and patients to minimize and/or eliminate barriers that patients face in accessing needed medications. Specifically, the AADA advocates for legislation that limits obstacles associated with health insurance step therapy requirements, streamlines PA, and prohibits mid-year formulary changes.8
Step therapy requires that patients first try a medication specified by the insurance company; the therapy must fail before the patient is placed on the medication originally prescribed by the provider. Regarding PA, the AADA tries to ensure that determinations are standardized, requires the speed of determinations to be quantified and minimized, and ensures that PA and appeals policies do not unduly burden physicians or patients in accessing optimal drug therapy.8
Another advocacy priority is telehealth. The AADA is advocating for legislation on expansion of telehealth in underserved areas and modifications to state licensure requirements, liability issues, and reimbursement for store-and-forward technology. The AADA is involved in protecting scope of practice, truth in advertising, and access to specialty care, as well as monitoring legislation and regulation concerning the potential environmental impact of sunscreen ingredients, indoor tanning restrictions, and skin cancer prevention.8
Advocacy Matters and Makes a Difference—It is important to learn about and support advocacy priorities and efforts and join forces to protect your practice. The AADA advocacy priorities are to protect the value of dermatology services, mobilize dermatologists for political action, ensure dermatologists can participate in new payment models, and strengthen the profession.9 Physician advocacy is no longer an elective pursuit. We need to be involved and engaged through our medical societies to help patients, communities, and ourselves. All of us are in it together, and a collaborative collective voice can make a difference. Take action, join the AADA, and contact Congress today to stop Medicare payment cuts (https://takeaction.aad.org/).
- Kaplan KJ. AMA announces new add-on digital pathology codes—no reimbursement (yet). July 18, 2022. Accessed October 19, 2022. https://tissuepathology.com/2022/07/18/ama-announces-new-add-on-digital-pathology-codes-no-reimbursement-yet/
- Centers for Medicare & Medicaid Services. National Health Expenditure Data: NHE fact sheet. Published April 2020. Accessed November 21, 2022. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/NHE-Fact-Sheet
- Houghton V. Ask the expert (Dr. Mark Kaufmann): fighting for fair Medicare reimbursement. Dermatology World. October 2022. Accessed November 21, 2022. https://digitaleditions.walsworth.com/article/Advocacy+News/4355162/763056/article.html
- Federal Register, Medicare Trustees’ Reports and U.S. Bureau of Labor Statistics, AMA, Economic and Health Policy Research. September 2022. Accessed November 21, 2022. https://www.ama-assn.org/system/files/key-measures-medicare-economic-index-chart.pdf
- American Medical Association. Current Medicare payment system on unsustainable path: contact Congress. September 30, 2022. Accessed November 21, 2022. https://www.ama-assn.org/practice-management/medicare-medicaid/current-medicare-payment-system-unsustainable-path-contact
- U.S. Bureau of Labor Statistics, American Medical Association, Economic and Health Policy Research, February 2022. Accessed November 21, 2022. https://www.ama-assn.org/system/files/key-measures-medicare-economic-index-chart.pdf
- American Medical Association. Characteristics of a rational Medicare payment system. Accessed November 22, 2022. https://www.ama-assn.org/system/files/characteristics-rational-medicare-payment-principles-signatories.pdf
- Ensuring patient access to effective and affordable treatments remains a top priority for the AAD. Dermatology Practice Management. June 2020. Accessed November 21, 2022. https://dermatologypracticemanagement.com/issues/2020/june-2020-vol-1-no-1/11-supporting-access-to-treatment-exceptional-customer-experience-innovation-and-growth-a-conversation-with-sumner-madden
- Marteja L. Advocacy: when, where, and how for dermatologists. The Dermatologist. September 2021. Accessed November 21, 2022. https://www.hmpgloballearningnetwork.com/site/thederm/cover-story/advocacy-when-where-and-how-dermatologists
New Year, New Codes: A Win-Win for Digital Pathology
In July 2022, the American Medical Association CPT (Current Procedural Terminology) Editorial Panel released 13 new digital pathology add-on Category III codes for 2023 that the College of American Pathologists successfully advocated for inclusion.1 These codes are for reporting additional clinical staff work and service requirements associated with digitizing glass microscope slides for primary diagnosis (Table). They go into effect on January 1, 2023.

Although there is no additional compensation with the new Category III codes, dermatopathology laboratories will be able to report when they have made a diagnosis using digital pathology. The new CPT codes will provide payers with data they need to directly understand the utilization and increased value of digital pathology, which will bring dermatopathology laboratories one step closer to receiving additional reimbursement for digital interpretation.
The adoption of digital pathology has been accelerating in the United States but still lags behind many European countries where reimbursement for digital pathology has been established for many years. Many of the barriers to digital pathology have improved—cloud storage is more affordable, scanners have a higher throughput, digital pathology platforms have improved, and the US Food and Drug Administration has granted approvals for digital pathology. Digital pathology allows for more efficient workflow, which results in increased productivity and a reduction in turnaround times. It also allows for a wide spectrum of clinical applications and more innovation as well as research and educational applications.
The new Category III codes cannot be reported solely for archival purposes (eg, after the Category I service has already been performed and reported), solely for educational purposes (eg, when services are not used for individual patient reporting), solely for developing a database for training or validation of artificial intelligence algorithms, and solely for clinical conference presentations (eg, tumor board interdisciplinary conferences).
The new codes are a major victory for the adoption and future compensation for digital pathology.
New Year, New Cuts: Proposed 2023 Medicare Policy and Payment Changes for Dermatologists
The United States Spent $3.8 Trillion on Health Care in 2019: Where Did It Go?—In 2019, approximately $3.8 trillion was spent on health care in the United States (Figure 1). Physician services accounted for approximately 15% of total health care spending.2

Medicare Payments for Physician Services—Medicare payments for physician services are determined by a relative value unit (RVU) multiplied by a conversion factor (CF). Relative value units were set up in 1992 by what is now the Centers for Medicare & Medicaid Services, and they calculated the time it took a physician to complete a task or RVU and multiplied it by $32.00 (CF).3
Thirty years later—in 2022—the CF is $34.61. If the CF had increased with inflation, it would be $59.00. If the Proposed Rule is adopted, the 2023 fee schedule payment formula would decrease by 4.4% (to $33.08) relative to that of the 2022 fee schedule ($34.61), which is a decrease of 8.2% since 2019 ($36.04). This decrease is due to expiration of the 3% increase to Medicare fee schedule payments for 2022 required by the Protecting Medicare and American Farmers from Sequester Cuts Act and the required budget neutrality adjustment required by changes in RVUs. Medicare physician payment has declined 22% from 2001 to 2022 (Figure 2).4,5

The adjustments to the CF typically are made based on 3 factors: (1) the Medicare Economic Index; (2) expenditure target “performance adjustment”; and (3) miscellaneous adjustments, including those for “budget neutrality” required by law.
Medicare Physician Payments Compared With Other Provider Types and Inflation—The proposed Medicare physician payment policy is unsustainable for outpatient dermatologists. Practice overhead has increased markedly since 1992. Other service providers, such as those in skilled nursing facilities and hospitals (Figure 3), have received favorable payment increases compared with practice cost inflation and the Consumer Price Index.3-6 Flat reimbursement affects all physicians who accept insurance, as even private insurers base their reimbursement on Medicare.

In addition, there are other issues resulting in decreased physician payments when evaluation and management services are reported with same-day procedures using modifier −25 as well as preserving or finding alternative strategies for 10- and 90-day global period payments for medical procedures. When Medicare cuts physician payments, dermatologists find it difficult to own and operate their own practices, resulting in health market consolidation, limited competition, increased health care costs, limited patient access to care, and decreased quality of health care.
Medicare Payment Reform—Medicare payment reform is necessary to stop annual payment cuts and create a stable predictable payment system that ensures patient access to quality, value-based care. Medicare physician payment reform needs to happen at a national level. The American Academy of Dermatology Association (AADA) is working with the House of Medicine and the medical specialty community to develop specific proposals, such as “Characteristics of a Rational Medicare Physician Payment System,” to reform Medicare’s payment system.7 Advocacy groups, including the AADA, have been working to mitigate the proposed 2023 cuts by engaging with Congress and urging them to act before these changes go into effect on January 1, 2023.
Make Advocacy Your New Year’s Resolution: AADA’s Top Advocacy Priorities
The AADA’s top priority is Medicare payment policies.3 In addition, the AADA is working on drug access and cost by cutting the bureaucratic red tape caused by prior authorization (PA) and step therapy policies. The AADA collaborates with manufacturers, the health care community, policymakers, private payers, pharmacists, pharmacy benefit managers, and patients to minimize and/or eliminate barriers that patients face in accessing needed medications. Specifically, the AADA advocates for legislation that limits obstacles associated with health insurance step therapy requirements, streamlines PA, and prohibits mid-year formulary changes.8
Step therapy requires that patients first try a medication specified by the insurance company; the therapy must fail before the patient is placed on the medication originally prescribed by the provider. Regarding PA, the AADA tries to ensure that determinations are standardized, requires the speed of determinations to be quantified and minimized, and ensures that PA and appeals policies do not unduly burden physicians or patients in accessing optimal drug therapy.8
Another advocacy priority is telehealth. The AADA is advocating for legislation on expansion of telehealth in underserved areas and modifications to state licensure requirements, liability issues, and reimbursement for store-and-forward technology. The AADA is involved in protecting scope of practice, truth in advertising, and access to specialty care, as well as monitoring legislation and regulation concerning the potential environmental impact of sunscreen ingredients, indoor tanning restrictions, and skin cancer prevention.8
Advocacy Matters and Makes a Difference—It is important to learn about and support advocacy priorities and efforts and join forces to protect your practice. The AADA advocacy priorities are to protect the value of dermatology services, mobilize dermatologists for political action, ensure dermatologists can participate in new payment models, and strengthen the profession.9 Physician advocacy is no longer an elective pursuit. We need to be involved and engaged through our medical societies to help patients, communities, and ourselves. All of us are in it together, and a collaborative collective voice can make a difference. Take action, join the AADA, and contact Congress today to stop Medicare payment cuts (https://takeaction.aad.org/).
New Year, New Codes: A Win-Win for Digital Pathology
In July 2022, the American Medical Association CPT (Current Procedural Terminology) Editorial Panel released 13 new digital pathology add-on Category III codes for 2023 that the College of American Pathologists successfully advocated for inclusion.1 These codes are for reporting additional clinical staff work and service requirements associated with digitizing glass microscope slides for primary diagnosis (Table). They go into effect on January 1, 2023.

Although there is no additional compensation with the new Category III codes, dermatopathology laboratories will be able to report when they have made a diagnosis using digital pathology. The new CPT codes will provide payers with data they need to directly understand the utilization and increased value of digital pathology, which will bring dermatopathology laboratories one step closer to receiving additional reimbursement for digital interpretation.
The adoption of digital pathology has been accelerating in the United States but still lags behind many European countries where reimbursement for digital pathology has been established for many years. Many of the barriers to digital pathology have improved—cloud storage is more affordable, scanners have a higher throughput, digital pathology platforms have improved, and the US Food and Drug Administration has granted approvals for digital pathology. Digital pathology allows for more efficient workflow, which results in increased productivity and a reduction in turnaround times. It also allows for a wide spectrum of clinical applications and more innovation as well as research and educational applications.
The new Category III codes cannot be reported solely for archival purposes (eg, after the Category I service has already been performed and reported), solely for educational purposes (eg, when services are not used for individual patient reporting), solely for developing a database for training or validation of artificial intelligence algorithms, and solely for clinical conference presentations (eg, tumor board interdisciplinary conferences).
The new codes are a major victory for the adoption and future compensation for digital pathology.
New Year, New Cuts: Proposed 2023 Medicare Policy and Payment Changes for Dermatologists
The United States Spent $3.8 Trillion on Health Care in 2019: Where Did It Go?—In 2019, approximately $3.8 trillion was spent on health care in the United States (Figure 1). Physician services accounted for approximately 15% of total health care spending.2

Medicare Payments for Physician Services—Medicare payments for physician services are determined by a relative value unit (RVU) multiplied by a conversion factor (CF). Relative value units were set up in 1992 by what is now the Centers for Medicare & Medicaid Services, and they calculated the time it took a physician to complete a task or RVU and multiplied it by $32.00 (CF).3
Thirty years later—in 2022—the CF is $34.61. If the CF had increased with inflation, it would be $59.00. If the Proposed Rule is adopted, the 2023 fee schedule payment formula would decrease by 4.4% (to $33.08) relative to that of the 2022 fee schedule ($34.61), which is a decrease of 8.2% since 2019 ($36.04). This decrease is due to expiration of the 3% increase to Medicare fee schedule payments for 2022 required by the Protecting Medicare and American Farmers from Sequester Cuts Act and the required budget neutrality adjustment required by changes in RVUs. Medicare physician payment has declined 22% from 2001 to 2022 (Figure 2).4,5

The adjustments to the CF typically are made based on 3 factors: (1) the Medicare Economic Index; (2) expenditure target “performance adjustment”; and (3) miscellaneous adjustments, including those for “budget neutrality” required by law.
Medicare Physician Payments Compared With Other Provider Types and Inflation—The proposed Medicare physician payment policy is unsustainable for outpatient dermatologists. Practice overhead has increased markedly since 1992. Other service providers, such as those in skilled nursing facilities and hospitals (Figure 3), have received favorable payment increases compared with practice cost inflation and the Consumer Price Index.3-6 Flat reimbursement affects all physicians who accept insurance, as even private insurers base their reimbursement on Medicare.

In addition, there are other issues resulting in decreased physician payments when evaluation and management services are reported with same-day procedures using modifier −25 as well as preserving or finding alternative strategies for 10- and 90-day global period payments for medical procedures. When Medicare cuts physician payments, dermatologists find it difficult to own and operate their own practices, resulting in health market consolidation, limited competition, increased health care costs, limited patient access to care, and decreased quality of health care.
Medicare Payment Reform—Medicare payment reform is necessary to stop annual payment cuts and create a stable predictable payment system that ensures patient access to quality, value-based care. Medicare physician payment reform needs to happen at a national level. The American Academy of Dermatology Association (AADA) is working with the House of Medicine and the medical specialty community to develop specific proposals, such as “Characteristics of a Rational Medicare Physician Payment System,” to reform Medicare’s payment system.7 Advocacy groups, including the AADA, have been working to mitigate the proposed 2023 cuts by engaging with Congress and urging them to act before these changes go into effect on January 1, 2023.
Make Advocacy Your New Year’s Resolution: AADA’s Top Advocacy Priorities
The AADA’s top priority is Medicare payment policies.3 In addition, the AADA is working on drug access and cost by cutting the bureaucratic red tape caused by prior authorization (PA) and step therapy policies. The AADA collaborates with manufacturers, the health care community, policymakers, private payers, pharmacists, pharmacy benefit managers, and patients to minimize and/or eliminate barriers that patients face in accessing needed medications. Specifically, the AADA advocates for legislation that limits obstacles associated with health insurance step therapy requirements, streamlines PA, and prohibits mid-year formulary changes.8
Step therapy requires that patients first try a medication specified by the insurance company; the therapy must fail before the patient is placed on the medication originally prescribed by the provider. Regarding PA, the AADA tries to ensure that determinations are standardized, requires the speed of determinations to be quantified and minimized, and ensures that PA and appeals policies do not unduly burden physicians or patients in accessing optimal drug therapy.8
Another advocacy priority is telehealth. The AADA is advocating for legislation on expansion of telehealth in underserved areas and modifications to state licensure requirements, liability issues, and reimbursement for store-and-forward technology. The AADA is involved in protecting scope of practice, truth in advertising, and access to specialty care, as well as monitoring legislation and regulation concerning the potential environmental impact of sunscreen ingredients, indoor tanning restrictions, and skin cancer prevention.8
Advocacy Matters and Makes a Difference—It is important to learn about and support advocacy priorities and efforts and join forces to protect your practice. The AADA advocacy priorities are to protect the value of dermatology services, mobilize dermatologists for political action, ensure dermatologists can participate in new payment models, and strengthen the profession.9 Physician advocacy is no longer an elective pursuit. We need to be involved and engaged through our medical societies to help patients, communities, and ourselves. All of us are in it together, and a collaborative collective voice can make a difference. Take action, join the AADA, and contact Congress today to stop Medicare payment cuts (https://takeaction.aad.org/).
- Kaplan KJ. AMA announces new add-on digital pathology codes—no reimbursement (yet). July 18, 2022. Accessed October 19, 2022. https://tissuepathology.com/2022/07/18/ama-announces-new-add-on-digital-pathology-codes-no-reimbursement-yet/
- Centers for Medicare & Medicaid Services. National Health Expenditure Data: NHE fact sheet. Published April 2020. Accessed November 21, 2022. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/NHE-Fact-Sheet
- Houghton V. Ask the expert (Dr. Mark Kaufmann): fighting for fair Medicare reimbursement. Dermatology World. October 2022. Accessed November 21, 2022. https://digitaleditions.walsworth.com/article/Advocacy+News/4355162/763056/article.html
- Federal Register, Medicare Trustees’ Reports and U.S. Bureau of Labor Statistics, AMA, Economic and Health Policy Research. September 2022. Accessed November 21, 2022. https://www.ama-assn.org/system/files/key-measures-medicare-economic-index-chart.pdf
- American Medical Association. Current Medicare payment system on unsustainable path: contact Congress. September 30, 2022. Accessed November 21, 2022. https://www.ama-assn.org/practice-management/medicare-medicaid/current-medicare-payment-system-unsustainable-path-contact
- U.S. Bureau of Labor Statistics, American Medical Association, Economic and Health Policy Research, February 2022. Accessed November 21, 2022. https://www.ama-assn.org/system/files/key-measures-medicare-economic-index-chart.pdf
- American Medical Association. Characteristics of a rational Medicare payment system. Accessed November 22, 2022. https://www.ama-assn.org/system/files/characteristics-rational-medicare-payment-principles-signatories.pdf
- Ensuring patient access to effective and affordable treatments remains a top priority for the AAD. Dermatology Practice Management. June 2020. Accessed November 21, 2022. https://dermatologypracticemanagement.com/issues/2020/june-2020-vol-1-no-1/11-supporting-access-to-treatment-exceptional-customer-experience-innovation-and-growth-a-conversation-with-sumner-madden
- Marteja L. Advocacy: when, where, and how for dermatologists. The Dermatologist. September 2021. Accessed November 21, 2022. https://www.hmpgloballearningnetwork.com/site/thederm/cover-story/advocacy-when-where-and-how-dermatologists
- Kaplan KJ. AMA announces new add-on digital pathology codes—no reimbursement (yet). July 18, 2022. Accessed October 19, 2022. https://tissuepathology.com/2022/07/18/ama-announces-new-add-on-digital-pathology-codes-no-reimbursement-yet/
- Centers for Medicare & Medicaid Services. National Health Expenditure Data: NHE fact sheet. Published April 2020. Accessed November 21, 2022. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/NHE-Fact-Sheet
- Houghton V. Ask the expert (Dr. Mark Kaufmann): fighting for fair Medicare reimbursement. Dermatology World. October 2022. Accessed November 21, 2022. https://digitaleditions.walsworth.com/article/Advocacy+News/4355162/763056/article.html
- Federal Register, Medicare Trustees’ Reports and U.S. Bureau of Labor Statistics, AMA, Economic and Health Policy Research. September 2022. Accessed November 21, 2022. https://www.ama-assn.org/system/files/key-measures-medicare-economic-index-chart.pdf
- American Medical Association. Current Medicare payment system on unsustainable path: contact Congress. September 30, 2022. Accessed November 21, 2022. https://www.ama-assn.org/practice-management/medicare-medicaid/current-medicare-payment-system-unsustainable-path-contact
- U.S. Bureau of Labor Statistics, American Medical Association, Economic and Health Policy Research, February 2022. Accessed November 21, 2022. https://www.ama-assn.org/system/files/key-measures-medicare-economic-index-chart.pdf
- American Medical Association. Characteristics of a rational Medicare payment system. Accessed November 22, 2022. https://www.ama-assn.org/system/files/characteristics-rational-medicare-payment-principles-signatories.pdf
- Ensuring patient access to effective and affordable treatments remains a top priority for the AAD. Dermatology Practice Management. June 2020. Accessed November 21, 2022. https://dermatologypracticemanagement.com/issues/2020/june-2020-vol-1-no-1/11-supporting-access-to-treatment-exceptional-customer-experience-innovation-and-growth-a-conversation-with-sumner-madden
- Marteja L. Advocacy: when, where, and how for dermatologists. The Dermatologist. September 2021. Accessed November 21, 2022. https://www.hmpgloballearningnetwork.com/site/thederm/cover-story/advocacy-when-where-and-how-dermatologists
Practice Points
- New digital pathology codes proposed by the American Medical Association can be used starting January 1, 2023.
- A proposed 2023 fee schedule negatively impacting dermatology practices was published by the Centers for Medicare & Medicaid Services in July 2022.
- Advocacy involvement provides a collaborative collective voice for our specialty to help our patients improve their care.
Nurturing a Satisfying Career in Dermatology
The residents of our program asked me to serve as their commencement speaker in June. Since I was retiring from my position as department chair, this touching honor seemed a fitting capstone for my career. It gave me the opportunity to reflect on the enormity of the changes that have occurred between my graduation from residency in 1983 and the current time, which is marked by disruption from the digital revolution and the COVID-19 pandemic. Throughout this 40-year period, there were times of external global turmoil, economic instability, significant changes in the business of medicine, stressful changes in documentation of competency and certification, and the difficult transition to electronic medical records. Another epidemic—AIDS—changed surgical practices. During my residency, we did biopsies without wearing gloves or masks. Gloves were added to protect the person doing the procedure as well as to prevent spread of disease to other patients, not to reduce the infection rate for the patient undergoing the procedure. Of course, change in the last 40 years also occurred outside of work and included various familial stresses. The irritations of daily life easily mounted up to being overwhelming. However, I had gone to work every day for 40 years, seeking to do my best for my patients and my colleagues and the staff with whom I worked, sometimes feeling successful and sometimes feeling incompetent. Some days went smoothly, and some days were filled with challenges that I could not begin to imagine how I would solve. I have a habit of seeing problems rather than successes, which creates its own difficulties. I did, however, grab opportunities that continually improved my practice of medicine and allowed me to serve in several professional positions as well as in leadership positions of multiple professional societies. As I prepared the commencement address, I realized that the totality of my career was very satisfying.
The Merriam-Webster dictionary definition of satisfying is “producing pleasure or contentment by providing what is needed or wanted.”1 My use of the word means that my career over the long term has pleased me—maybe not some of the people I reported to, but rather me.
My approach to my career can be summarized in 3 words: purpose, serendipity, and curiosity.
The first element is purpose. Job satisfaction generally is associated with work being aligned with values, an appreciation that you are accomplishing the purpose with which you set out on your journey. It is not associated with every day being wonderful and problem free or every task being completed without setbacks or complications. The reality of working is not that every moment brings pure happiness or that every task fulfills a passion. How does a person ensure that the days add up to be satisfying? Start with values. Why did you decide to pursue medical school? Some may have chosen it for economic security, but there are many ways to achieve economic security. Maybe being a physician feeds into the family lore, but families generally have broad ranges of acceptable careers. Maybe it appealed scientifically, but a PhD in biology also fulfills that interest. Maybe it is that you noticed respect for physicians in the community when you were growing up, but that is changing and does not represent an internal value anyway. Consider your values carefully, write them down, and keep them at the forefront of the day. Go back to them consciously any time you have a rough day and understand why you are doing what you are doing. When you are 55 years old and going through your umpteenth change in reimbursement process, go back to the day you decided on medicine as a career. Focus on your values as the grounding for your purpose. Also note that purpose is different than goals. Some goals will be reached, and some will not. Goals change with external realities and/ or internal factors. Purpose and values remain the same if we have thoughtfully identified them.
The second element is serendipity. Serendipity often is thought of as luck, as karma, as being in the right place at the right time. It feels random, and at first glance it appears that purpose and serendipity are complete opposites and do not intersect. Serendipity is, however, not just luck. It is an ability to distinguish events and observations in meaningful ways. It is a close relative of creativity and benefits from sloppiness, playfulness, tinkering, and discussion. It cannot exist in a vacuum. History is replete with serendipitous discoveries. It is thought that James Watson and Francis Crick would never have been able to elucidate the nature of DNA without sharing offices with people with whom they argued daily. In fact, figuring out the DNA structure was not even the main focus of their laboratories. It was just a side angle that several people loved to think about. Appreciating serendipity by being truly open to opportunities that are out on the wings brings experiences that are deeply rewarding even if not planned. I had no idea at all, no plan, no goal of serving as president of the American Academy of Dermatology or as Department Chair, and yet these happened. These experiences have allowed me to work on my purpose as I have defined it. How can you harness serendipity in your own life? My philosophy may be somewhat simple, but I think if you show up every day doing the best job you can at the tasks on hand, doors will appear, at odd intervals and in odd directions. You must be open enough and in tune with your purpose to an extent that you can sense the direction in which to turn and what doorways through which to walk.
The third element is curiosity. One definition is that curiosity is the motivation to learn new information. Another definition is that curiosity is a special form of information seeking distinguished by the fact that it is internally motivated. We are all familiar with intellectual curiosity. For example, a patient has a basal cell carcinoma on the upper back. What does the literature say about the cure rates of various treatments for that particular tumor? In addition, we can be curious about other things as well. Is it a really small tumor? How was it found and why is the patient anxious? Why does it make me irritated that the patient is worried about such a small, easily treated tumor? Or is it a large neglected tumor? Why was it not treated before? Why does it make me sad that it is so large? Why does it annoy me that I have a difficult situation to manage? Being able to define an emotional reaction by being curious about its presence helps us manage destructive responses and promote more positive outcomes. This curiosity is related to emotional intelligence and is mindfully harnessed by effective leaders. Curiosity will get you through tough days when your office team is stressed and the tough years that are complicated by professional and personal challenges.
Curiosity also will help you identify your purpose and harness serendipity, and so we come full circle with our 3 elements: purpose, serendipity, and curiosity.
My wish for all of you is that when you are at the tail end of your career, you will look back and say, “This has been a great ride.” I am very grateful that I can acknowledge this for myself. I have been so fortunate to have found dermatology, where I can go to work every day making a difference for patients in a stimulating environment with good colleagues. One of my values is to try and make life better in some way for everyone around me, even if it is just a smile at the start of the workday. As I look back, this value has allowed me to meet interesting people, hear fascinating stories, make good friends, and have enduring relationships. I have held onto fellow travelers, and we have supported each other through tough times as well as celebrated together the good times.
Nurturing a satisfying career includes these essential fundamentals. First, accept the reality of constant change. Second, develop productive relationships with fellow travelers. And third and most importantly, go forth with purpose, serendipity, and curiosity.
- Merriam-Webster. Satisfying. Merriam-Webster.com Dictionary. Accessed November 18, 2022. https://www.merriam-webster.com/dictionary/satisfying
The residents of our program asked me to serve as their commencement speaker in June. Since I was retiring from my position as department chair, this touching honor seemed a fitting capstone for my career. It gave me the opportunity to reflect on the enormity of the changes that have occurred between my graduation from residency in 1983 and the current time, which is marked by disruption from the digital revolution and the COVID-19 pandemic. Throughout this 40-year period, there were times of external global turmoil, economic instability, significant changes in the business of medicine, stressful changes in documentation of competency and certification, and the difficult transition to electronic medical records. Another epidemic—AIDS—changed surgical practices. During my residency, we did biopsies without wearing gloves or masks. Gloves were added to protect the person doing the procedure as well as to prevent spread of disease to other patients, not to reduce the infection rate for the patient undergoing the procedure. Of course, change in the last 40 years also occurred outside of work and included various familial stresses. The irritations of daily life easily mounted up to being overwhelming. However, I had gone to work every day for 40 years, seeking to do my best for my patients and my colleagues and the staff with whom I worked, sometimes feeling successful and sometimes feeling incompetent. Some days went smoothly, and some days were filled with challenges that I could not begin to imagine how I would solve. I have a habit of seeing problems rather than successes, which creates its own difficulties. I did, however, grab opportunities that continually improved my practice of medicine and allowed me to serve in several professional positions as well as in leadership positions of multiple professional societies. As I prepared the commencement address, I realized that the totality of my career was very satisfying.
The Merriam-Webster dictionary definition of satisfying is “producing pleasure or contentment by providing what is needed or wanted.”1 My use of the word means that my career over the long term has pleased me—maybe not some of the people I reported to, but rather me.
My approach to my career can be summarized in 3 words: purpose, serendipity, and curiosity.
The first element is purpose. Job satisfaction generally is associated with work being aligned with values, an appreciation that you are accomplishing the purpose with which you set out on your journey. It is not associated with every day being wonderful and problem free or every task being completed without setbacks or complications. The reality of working is not that every moment brings pure happiness or that every task fulfills a passion. How does a person ensure that the days add up to be satisfying? Start with values. Why did you decide to pursue medical school? Some may have chosen it for economic security, but there are many ways to achieve economic security. Maybe being a physician feeds into the family lore, but families generally have broad ranges of acceptable careers. Maybe it appealed scientifically, but a PhD in biology also fulfills that interest. Maybe it is that you noticed respect for physicians in the community when you were growing up, but that is changing and does not represent an internal value anyway. Consider your values carefully, write them down, and keep them at the forefront of the day. Go back to them consciously any time you have a rough day and understand why you are doing what you are doing. When you are 55 years old and going through your umpteenth change in reimbursement process, go back to the day you decided on medicine as a career. Focus on your values as the grounding for your purpose. Also note that purpose is different than goals. Some goals will be reached, and some will not. Goals change with external realities and/ or internal factors. Purpose and values remain the same if we have thoughtfully identified them.
The second element is serendipity. Serendipity often is thought of as luck, as karma, as being in the right place at the right time. It feels random, and at first glance it appears that purpose and serendipity are complete opposites and do not intersect. Serendipity is, however, not just luck. It is an ability to distinguish events and observations in meaningful ways. It is a close relative of creativity and benefits from sloppiness, playfulness, tinkering, and discussion. It cannot exist in a vacuum. History is replete with serendipitous discoveries. It is thought that James Watson and Francis Crick would never have been able to elucidate the nature of DNA without sharing offices with people with whom they argued daily. In fact, figuring out the DNA structure was not even the main focus of their laboratories. It was just a side angle that several people loved to think about. Appreciating serendipity by being truly open to opportunities that are out on the wings brings experiences that are deeply rewarding even if not planned. I had no idea at all, no plan, no goal of serving as president of the American Academy of Dermatology or as Department Chair, and yet these happened. These experiences have allowed me to work on my purpose as I have defined it. How can you harness serendipity in your own life? My philosophy may be somewhat simple, but I think if you show up every day doing the best job you can at the tasks on hand, doors will appear, at odd intervals and in odd directions. You must be open enough and in tune with your purpose to an extent that you can sense the direction in which to turn and what doorways through which to walk.
The third element is curiosity. One definition is that curiosity is the motivation to learn new information. Another definition is that curiosity is a special form of information seeking distinguished by the fact that it is internally motivated. We are all familiar with intellectual curiosity. For example, a patient has a basal cell carcinoma on the upper back. What does the literature say about the cure rates of various treatments for that particular tumor? In addition, we can be curious about other things as well. Is it a really small tumor? How was it found and why is the patient anxious? Why does it make me irritated that the patient is worried about such a small, easily treated tumor? Or is it a large neglected tumor? Why was it not treated before? Why does it make me sad that it is so large? Why does it annoy me that I have a difficult situation to manage? Being able to define an emotional reaction by being curious about its presence helps us manage destructive responses and promote more positive outcomes. This curiosity is related to emotional intelligence and is mindfully harnessed by effective leaders. Curiosity will get you through tough days when your office team is stressed and the tough years that are complicated by professional and personal challenges.
Curiosity also will help you identify your purpose and harness serendipity, and so we come full circle with our 3 elements: purpose, serendipity, and curiosity.
My wish for all of you is that when you are at the tail end of your career, you will look back and say, “This has been a great ride.” I am very grateful that I can acknowledge this for myself. I have been so fortunate to have found dermatology, where I can go to work every day making a difference for patients in a stimulating environment with good colleagues. One of my values is to try and make life better in some way for everyone around me, even if it is just a smile at the start of the workday. As I look back, this value has allowed me to meet interesting people, hear fascinating stories, make good friends, and have enduring relationships. I have held onto fellow travelers, and we have supported each other through tough times as well as celebrated together the good times.
Nurturing a satisfying career includes these essential fundamentals. First, accept the reality of constant change. Second, develop productive relationships with fellow travelers. And third and most importantly, go forth with purpose, serendipity, and curiosity.
The residents of our program asked me to serve as their commencement speaker in June. Since I was retiring from my position as department chair, this touching honor seemed a fitting capstone for my career. It gave me the opportunity to reflect on the enormity of the changes that have occurred between my graduation from residency in 1983 and the current time, which is marked by disruption from the digital revolution and the COVID-19 pandemic. Throughout this 40-year period, there were times of external global turmoil, economic instability, significant changes in the business of medicine, stressful changes in documentation of competency and certification, and the difficult transition to electronic medical records. Another epidemic—AIDS—changed surgical practices. During my residency, we did biopsies without wearing gloves or masks. Gloves were added to protect the person doing the procedure as well as to prevent spread of disease to other patients, not to reduce the infection rate for the patient undergoing the procedure. Of course, change in the last 40 years also occurred outside of work and included various familial stresses. The irritations of daily life easily mounted up to being overwhelming. However, I had gone to work every day for 40 years, seeking to do my best for my patients and my colleagues and the staff with whom I worked, sometimes feeling successful and sometimes feeling incompetent. Some days went smoothly, and some days were filled with challenges that I could not begin to imagine how I would solve. I have a habit of seeing problems rather than successes, which creates its own difficulties. I did, however, grab opportunities that continually improved my practice of medicine and allowed me to serve in several professional positions as well as in leadership positions of multiple professional societies. As I prepared the commencement address, I realized that the totality of my career was very satisfying.
The Merriam-Webster dictionary definition of satisfying is “producing pleasure or contentment by providing what is needed or wanted.”1 My use of the word means that my career over the long term has pleased me—maybe not some of the people I reported to, but rather me.
My approach to my career can be summarized in 3 words: purpose, serendipity, and curiosity.
The first element is purpose. Job satisfaction generally is associated with work being aligned with values, an appreciation that you are accomplishing the purpose with which you set out on your journey. It is not associated with every day being wonderful and problem free or every task being completed without setbacks or complications. The reality of working is not that every moment brings pure happiness or that every task fulfills a passion. How does a person ensure that the days add up to be satisfying? Start with values. Why did you decide to pursue medical school? Some may have chosen it for economic security, but there are many ways to achieve economic security. Maybe being a physician feeds into the family lore, but families generally have broad ranges of acceptable careers. Maybe it appealed scientifically, but a PhD in biology also fulfills that interest. Maybe it is that you noticed respect for physicians in the community when you were growing up, but that is changing and does not represent an internal value anyway. Consider your values carefully, write them down, and keep them at the forefront of the day. Go back to them consciously any time you have a rough day and understand why you are doing what you are doing. When you are 55 years old and going through your umpteenth change in reimbursement process, go back to the day you decided on medicine as a career. Focus on your values as the grounding for your purpose. Also note that purpose is different than goals. Some goals will be reached, and some will not. Goals change with external realities and/ or internal factors. Purpose and values remain the same if we have thoughtfully identified them.
The second element is serendipity. Serendipity often is thought of as luck, as karma, as being in the right place at the right time. It feels random, and at first glance it appears that purpose and serendipity are complete opposites and do not intersect. Serendipity is, however, not just luck. It is an ability to distinguish events and observations in meaningful ways. It is a close relative of creativity and benefits from sloppiness, playfulness, tinkering, and discussion. It cannot exist in a vacuum. History is replete with serendipitous discoveries. It is thought that James Watson and Francis Crick would never have been able to elucidate the nature of DNA without sharing offices with people with whom they argued daily. In fact, figuring out the DNA structure was not even the main focus of their laboratories. It was just a side angle that several people loved to think about. Appreciating serendipity by being truly open to opportunities that are out on the wings brings experiences that are deeply rewarding even if not planned. I had no idea at all, no plan, no goal of serving as president of the American Academy of Dermatology or as Department Chair, and yet these happened. These experiences have allowed me to work on my purpose as I have defined it. How can you harness serendipity in your own life? My philosophy may be somewhat simple, but I think if you show up every day doing the best job you can at the tasks on hand, doors will appear, at odd intervals and in odd directions. You must be open enough and in tune with your purpose to an extent that you can sense the direction in which to turn and what doorways through which to walk.
The third element is curiosity. One definition is that curiosity is the motivation to learn new information. Another definition is that curiosity is a special form of information seeking distinguished by the fact that it is internally motivated. We are all familiar with intellectual curiosity. For example, a patient has a basal cell carcinoma on the upper back. What does the literature say about the cure rates of various treatments for that particular tumor? In addition, we can be curious about other things as well. Is it a really small tumor? How was it found and why is the patient anxious? Why does it make me irritated that the patient is worried about such a small, easily treated tumor? Or is it a large neglected tumor? Why was it not treated before? Why does it make me sad that it is so large? Why does it annoy me that I have a difficult situation to manage? Being able to define an emotional reaction by being curious about its presence helps us manage destructive responses and promote more positive outcomes. This curiosity is related to emotional intelligence and is mindfully harnessed by effective leaders. Curiosity will get you through tough days when your office team is stressed and the tough years that are complicated by professional and personal challenges.
Curiosity also will help you identify your purpose and harness serendipity, and so we come full circle with our 3 elements: purpose, serendipity, and curiosity.
My wish for all of you is that when you are at the tail end of your career, you will look back and say, “This has been a great ride.” I am very grateful that I can acknowledge this for myself. I have been so fortunate to have found dermatology, where I can go to work every day making a difference for patients in a stimulating environment with good colleagues. One of my values is to try and make life better in some way for everyone around me, even if it is just a smile at the start of the workday. As I look back, this value has allowed me to meet interesting people, hear fascinating stories, make good friends, and have enduring relationships. I have held onto fellow travelers, and we have supported each other through tough times as well as celebrated together the good times.
Nurturing a satisfying career includes these essential fundamentals. First, accept the reality of constant change. Second, develop productive relationships with fellow travelers. And third and most importantly, go forth with purpose, serendipity, and curiosity.
- Merriam-Webster. Satisfying. Merriam-Webster.com Dictionary. Accessed November 18, 2022. https://www.merriam-webster.com/dictionary/satisfying
- Merriam-Webster. Satisfying. Merriam-Webster.com Dictionary. Accessed November 18, 2022. https://www.merriam-webster.com/dictionary/satisfying