User login
Synthetic triglyceride shows potential in Huntington’s disease
, according to data presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Reporting results of TRIHEP3 and an extension study, Fanny Mochel, MD, PhD, of Sorbonne University in Paris and the Paris Brain Institute, said in an interview that her group is the only one investigating triheptanoin to target caudate atrophy in Huntington’s disease. The Food and Drug Administration last year approved triheptanoin for the treatment of long-chain fatty acid oxidation disorders.
“The main findings are two observations: that patients were clinically stable based on their gradation of total motor score (TMS) on UHDRS (Unified Huntington’s Disease Rating Scale) after 1 year,” Dr. Mochel said in an interview. “The other is that we observed a reduction of the caudate atrophy progression that we usually see over 1 year by about 50%.”
TRIHEP3 randomized 100 patients with early-stage Huntington’s disease to triheptanoin 1g/kg daily and placebo. It followed on previous research in which the group used 31-phosphorus brain MR spectroscopy to demonstrate triheptanoin restored a normal brain energetic profile in patients with Huntington’s disease. TRIHEP3 was a 6-month randomized controlled trial at two centers, followed by a 6-month open-label phase. After that, 42 patients opted to participate in the 1-year extension study.
TRIHEP3 found no difference in caudate boundary shift integral (cBSI) at 6 months – the primary endpoint. But in the extension study, TMS tended to stabilize in patients treated for 1 year (0.6 ± 5.1), compared with those treated for 6 months (2.5 ± 4.5, P = .072).
Using a placebo control group from an external study of patients with Huntington’s disease with what Dr. Mochel described as “identical clinical characteristics,” she said the research confirmed TMS clinical stability in treated patients at 1 year (2.6 ± 4.6 vs. 0.6 ± 5.1, P = .057) and found significantly lower caudate atrophy (–3% vs. –6.7%, compared with baseline, P < .001).
Dr. Mochel also noted that Diffusion Tensor Imaging and Fixed-based analyses (FBA) showed fewer alterations in fiber metrics at 24 months in patients treated from baseline. FBA also showed improved fiber trophicity at 24 months in both groups.
‘The first good news’
Dr. Mochel noted that the Huntington’s disease community had been shaken in the spring by the failure of three trials of gene-targeting therapies for Huntington’s disease. Roche halted a phase 3 study of its antisense oligonucleotide (ASO) tominersen, and Wave Life Sciences scuttled two ASO programs in phase 1/2 trials.
“Triheptanoin is not going to cure Huntington’s disease; it’s a disease with many components, but it does work on the energy aspects and that seems to stabilize patients over the time of observation,” Dr. Mochel said. “That’s the first good news.”
She also noted that side effects were mainly gastrointestinal in nature, and they typically resolved with dietary management.
As a target in Huntington disease, the caudate nucleus is highly desirable, and caudate atrophy has been shown to occur even before the onset of motor symptoms, said N. Ahmad Aziz, MD, PhD, a neurologist and epidemiologist at the German Center for Neurodegenerative Diseases at the University of Bonn (Germany). “In this light, the findings of the trial conducted by Dr. Mochel and colleagues, which suggest that triheptanoin intake may slow down the rate of caudate atrophy in patients with early-stage Huntington’s disease, are highly promising,” Dr. Aziz said in an interview.
However, he noted that the improvement in caudate atrophy was only a secondary endpoint in the extension study. “Nevertheless, given triheptanoin’s biologically plausible mechanism of action – i.e., provision of substrates to the Krebs cycle and at least partial restoration of the well-documented defective mitochondrial function in Huntington’s disease – combined with its apparently relatively mild side-effect profile and good tolerability, I think that the preliminary findings of this trial are very promising and justify a larger phase 3 trial,” Dr. Aziz said.
Dr. Mochel said that the findings are prompting the investigators to consider just that.
Dr. Mochel has received consulting fees from and conducted investigator‐sponsored studies supported by Ultragenyx Pharmaceuticals. Dr. Aziz has no relevant financial relationships to disclose.
, according to data presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Reporting results of TRIHEP3 and an extension study, Fanny Mochel, MD, PhD, of Sorbonne University in Paris and the Paris Brain Institute, said in an interview that her group is the only one investigating triheptanoin to target caudate atrophy in Huntington’s disease. The Food and Drug Administration last year approved triheptanoin for the treatment of long-chain fatty acid oxidation disorders.
“The main findings are two observations: that patients were clinically stable based on their gradation of total motor score (TMS) on UHDRS (Unified Huntington’s Disease Rating Scale) after 1 year,” Dr. Mochel said in an interview. “The other is that we observed a reduction of the caudate atrophy progression that we usually see over 1 year by about 50%.”
TRIHEP3 randomized 100 patients with early-stage Huntington’s disease to triheptanoin 1g/kg daily and placebo. It followed on previous research in which the group used 31-phosphorus brain MR spectroscopy to demonstrate triheptanoin restored a normal brain energetic profile in patients with Huntington’s disease. TRIHEP3 was a 6-month randomized controlled trial at two centers, followed by a 6-month open-label phase. After that, 42 patients opted to participate in the 1-year extension study.
TRIHEP3 found no difference in caudate boundary shift integral (cBSI) at 6 months – the primary endpoint. But in the extension study, TMS tended to stabilize in patients treated for 1 year (0.6 ± 5.1), compared with those treated for 6 months (2.5 ± 4.5, P = .072).
Using a placebo control group from an external study of patients with Huntington’s disease with what Dr. Mochel described as “identical clinical characteristics,” she said the research confirmed TMS clinical stability in treated patients at 1 year (2.6 ± 4.6 vs. 0.6 ± 5.1, P = .057) and found significantly lower caudate atrophy (–3% vs. –6.7%, compared with baseline, P < .001).
Dr. Mochel also noted that Diffusion Tensor Imaging and Fixed-based analyses (FBA) showed fewer alterations in fiber metrics at 24 months in patients treated from baseline. FBA also showed improved fiber trophicity at 24 months in both groups.
‘The first good news’
Dr. Mochel noted that the Huntington’s disease community had been shaken in the spring by the failure of three trials of gene-targeting therapies for Huntington’s disease. Roche halted a phase 3 study of its antisense oligonucleotide (ASO) tominersen, and Wave Life Sciences scuttled two ASO programs in phase 1/2 trials.
“Triheptanoin is not going to cure Huntington’s disease; it’s a disease with many components, but it does work on the energy aspects and that seems to stabilize patients over the time of observation,” Dr. Mochel said. “That’s the first good news.”
She also noted that side effects were mainly gastrointestinal in nature, and they typically resolved with dietary management.
As a target in Huntington disease, the caudate nucleus is highly desirable, and caudate atrophy has been shown to occur even before the onset of motor symptoms, said N. Ahmad Aziz, MD, PhD, a neurologist and epidemiologist at the German Center for Neurodegenerative Diseases at the University of Bonn (Germany). “In this light, the findings of the trial conducted by Dr. Mochel and colleagues, which suggest that triheptanoin intake may slow down the rate of caudate atrophy in patients with early-stage Huntington’s disease, are highly promising,” Dr. Aziz said in an interview.
However, he noted that the improvement in caudate atrophy was only a secondary endpoint in the extension study. “Nevertheless, given triheptanoin’s biologically plausible mechanism of action – i.e., provision of substrates to the Krebs cycle and at least partial restoration of the well-documented defective mitochondrial function in Huntington’s disease – combined with its apparently relatively mild side-effect profile and good tolerability, I think that the preliminary findings of this trial are very promising and justify a larger phase 3 trial,” Dr. Aziz said.
Dr. Mochel said that the findings are prompting the investigators to consider just that.
Dr. Mochel has received consulting fees from and conducted investigator‐sponsored studies supported by Ultragenyx Pharmaceuticals. Dr. Aziz has no relevant financial relationships to disclose.
, according to data presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Reporting results of TRIHEP3 and an extension study, Fanny Mochel, MD, PhD, of Sorbonne University in Paris and the Paris Brain Institute, said in an interview that her group is the only one investigating triheptanoin to target caudate atrophy in Huntington’s disease. The Food and Drug Administration last year approved triheptanoin for the treatment of long-chain fatty acid oxidation disorders.
“The main findings are two observations: that patients were clinically stable based on their gradation of total motor score (TMS) on UHDRS (Unified Huntington’s Disease Rating Scale) after 1 year,” Dr. Mochel said in an interview. “The other is that we observed a reduction of the caudate atrophy progression that we usually see over 1 year by about 50%.”
TRIHEP3 randomized 100 patients with early-stage Huntington’s disease to triheptanoin 1g/kg daily and placebo. It followed on previous research in which the group used 31-phosphorus brain MR spectroscopy to demonstrate triheptanoin restored a normal brain energetic profile in patients with Huntington’s disease. TRIHEP3 was a 6-month randomized controlled trial at two centers, followed by a 6-month open-label phase. After that, 42 patients opted to participate in the 1-year extension study.
TRIHEP3 found no difference in caudate boundary shift integral (cBSI) at 6 months – the primary endpoint. But in the extension study, TMS tended to stabilize in patients treated for 1 year (0.6 ± 5.1), compared with those treated for 6 months (2.5 ± 4.5, P = .072).
Using a placebo control group from an external study of patients with Huntington’s disease with what Dr. Mochel described as “identical clinical characteristics,” she said the research confirmed TMS clinical stability in treated patients at 1 year (2.6 ± 4.6 vs. 0.6 ± 5.1, P = .057) and found significantly lower caudate atrophy (–3% vs. –6.7%, compared with baseline, P < .001).
Dr. Mochel also noted that Diffusion Tensor Imaging and Fixed-based analyses (FBA) showed fewer alterations in fiber metrics at 24 months in patients treated from baseline. FBA also showed improved fiber trophicity at 24 months in both groups.
‘The first good news’
Dr. Mochel noted that the Huntington’s disease community had been shaken in the spring by the failure of three trials of gene-targeting therapies for Huntington’s disease. Roche halted a phase 3 study of its antisense oligonucleotide (ASO) tominersen, and Wave Life Sciences scuttled two ASO programs in phase 1/2 trials.
“Triheptanoin is not going to cure Huntington’s disease; it’s a disease with many components, but it does work on the energy aspects and that seems to stabilize patients over the time of observation,” Dr. Mochel said. “That’s the first good news.”
She also noted that side effects were mainly gastrointestinal in nature, and they typically resolved with dietary management.
As a target in Huntington disease, the caudate nucleus is highly desirable, and caudate atrophy has been shown to occur even before the onset of motor symptoms, said N. Ahmad Aziz, MD, PhD, a neurologist and epidemiologist at the German Center for Neurodegenerative Diseases at the University of Bonn (Germany). “In this light, the findings of the trial conducted by Dr. Mochel and colleagues, which suggest that triheptanoin intake may slow down the rate of caudate atrophy in patients with early-stage Huntington’s disease, are highly promising,” Dr. Aziz said in an interview.
However, he noted that the improvement in caudate atrophy was only a secondary endpoint in the extension study. “Nevertheless, given triheptanoin’s biologically plausible mechanism of action – i.e., provision of substrates to the Krebs cycle and at least partial restoration of the well-documented defective mitochondrial function in Huntington’s disease – combined with its apparently relatively mild side-effect profile and good tolerability, I think that the preliminary findings of this trial are very promising and justify a larger phase 3 trial,” Dr. Aziz said.
Dr. Mochel said that the findings are prompting the investigators to consider just that.
Dr. Mochel has received consulting fees from and conducted investigator‐sponsored studies supported by Ultragenyx Pharmaceuticals. Dr. Aziz has no relevant financial relationships to disclose.
FROM MDS VIRTUAL CONGRESS 2021
Mean leadership
The differences between the mean and median of leadership data
Let me apologize for misleading all of you; this is not an article about malignant physician leaders; instead, it goes over the numbers and trends uncovered by the 2020 State of Hospital Medicine report (SoHM).1 The hospital medicine leader ends up doing many tasks like planning, growth, collaboration, finance, recruiting, scheduling, onboarding, coaching, and most near and dear to our hearts, putting out the fires and conflict resolution.
Ratio of leadership FTE to physician hospitalists FTE
If my pun has already put you off, you can avoid reading the rest of the piece and go to the 2020 SoHM to look at pages 52 (Table 3.7c), 121 (Table 4.7c), and 166 (Table 5.7c). It has a newly added table (3.7c), and it is phenomenal; it is the ratio of leadership FTE to physician hospitalists FTE. As an avid user of SoHM, I always ended up doing a makeshift calculation to “guesstimate” this number. Now that we have it calculated for us and the ultimate revelation lies in its narrow range across all groups. We might differ in the region, employment type, academics, teaching, or size, but this range is relatively narrow.
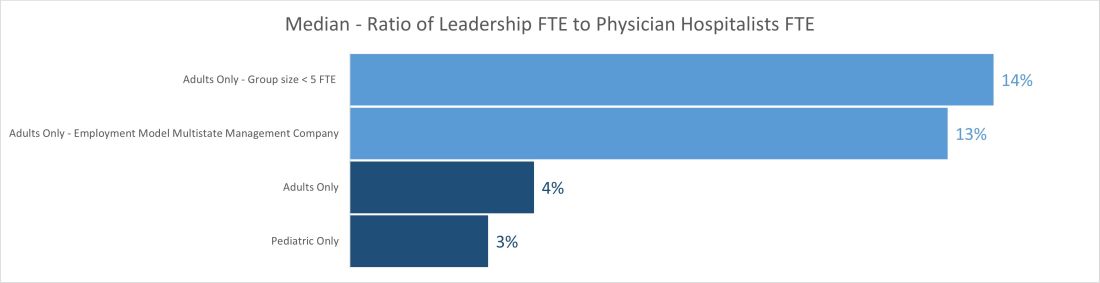
The median ratio of leadership FTE to total FTE lies between 2% and 5% in pediatric groups and between 3% and 6% for most adult groups. The only two outliers are on the adult side, with less than 5 FTE and multistate management companies. The higher median for the less than 5 FTE group size is understandable because of the small number of hospitalist FTEs that the leader’s time must be spread over. Even a small amount of dedicated leadership time will result in a high ratio of leader time to hospitalist clinical time if the group is very small. The multistate management company is probably a result of multiple layers of physician leadership (for example, regional medical directors) and travel-related time adjustments. Still, it raises the question of why the local leadership is not developed to decrease the leadership cost and better access.
Another helpful pattern is the decrease in standard deviation with the increase in group size. The hospital medicine leaders and CEOs of the hospital need to watch this number closely; any extremes on high or low side would be indicators for a deep dive in leadership structure and health.
Total number and total dedicated FTE for all physician leaders
Once we start seeing the differences between the mean and median of leadership data, we can see the median is relatively static while the mean has increased year after year and took a big jump in the 2020 SoHM. The chart below shows trends for the number of individuals in leadership positions (“Total No” and total FTEs allocated to leadership (“Total FTE”) over the last several surveys. The data is heavily skewed toward the right (positive); so, it makes sense to use the median in this case rather than mean. A few factors could explain the right skew of data.
- Large groups of 30 or more hospitalists are increasing, and so is their leadership need.
- There is more recognition of the need for dedicated leadership individuals and FTE.
- The leadership is getting less concentrated among just one or a few leaders.
- Outliers on the high side.
- Lower bounds of 0 or 0.1 FTE.
Highest-ranked leader dedicated FTE and premium compensation
Another pleasing trend is an increase in dedicated FTE for the highest-paid leader. Like any skill-set development, leadership requires the investment of deliberate practice, financial acumen, negotiation skills, and increased vulnerability. Time helps way more in developing these skill sets than money. SoHM trends show increase in dedicated FTE for the highest physician leader over the years and static premium compensation.
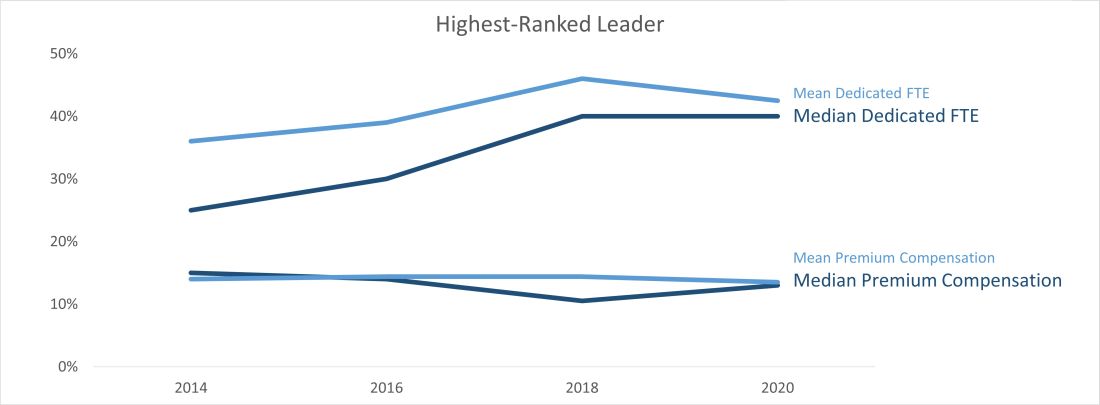
At last, we can say median leadership is always better than “mean” leadership in skewed data. Pun apart, every group needs leadership, and SoHM offers a nice window to the trends in leadership amongst many practice groups. It is a valuable resource for every group.
Dr. Chadha is chief of the division of hospital medicine at the University of Kentucky Healthcare, Lexington. He actively leads efforts of recruiting, practice analysis, and operation of the group. He is finishing his first tenure in the Practice Analysis Committee. He is often found spending a lot more than required time with spreadsheets and graphs.
Reference
1. 2020 State of Hospital Medicine. www.hospitalmedicine.org/practice-management/shms-state-of-hospital-medicine/
The differences between the mean and median of leadership data
The differences between the mean and median of leadership data
Let me apologize for misleading all of you; this is not an article about malignant physician leaders; instead, it goes over the numbers and trends uncovered by the 2020 State of Hospital Medicine report (SoHM).1 The hospital medicine leader ends up doing many tasks like planning, growth, collaboration, finance, recruiting, scheduling, onboarding, coaching, and most near and dear to our hearts, putting out the fires and conflict resolution.
Ratio of leadership FTE to physician hospitalists FTE
If my pun has already put you off, you can avoid reading the rest of the piece and go to the 2020 SoHM to look at pages 52 (Table 3.7c), 121 (Table 4.7c), and 166 (Table 5.7c). It has a newly added table (3.7c), and it is phenomenal; it is the ratio of leadership FTE to physician hospitalists FTE. As an avid user of SoHM, I always ended up doing a makeshift calculation to “guesstimate” this number. Now that we have it calculated for us and the ultimate revelation lies in its narrow range across all groups. We might differ in the region, employment type, academics, teaching, or size, but this range is relatively narrow.

The median ratio of leadership FTE to total FTE lies between 2% and 5% in pediatric groups and between 3% and 6% for most adult groups. The only two outliers are on the adult side, with less than 5 FTE and multistate management companies. The higher median for the less than 5 FTE group size is understandable because of the small number of hospitalist FTEs that the leader’s time must be spread over. Even a small amount of dedicated leadership time will result in a high ratio of leader time to hospitalist clinical time if the group is very small. The multistate management company is probably a result of multiple layers of physician leadership (for example, regional medical directors) and travel-related time adjustments. Still, it raises the question of why the local leadership is not developed to decrease the leadership cost and better access.
Another helpful pattern is the decrease in standard deviation with the increase in group size. The hospital medicine leaders and CEOs of the hospital need to watch this number closely; any extremes on high or low side would be indicators for a deep dive in leadership structure and health.
Total number and total dedicated FTE for all physician leaders
Once we start seeing the differences between the mean and median of leadership data, we can see the median is relatively static while the mean has increased year after year and took a big jump in the 2020 SoHM. The chart below shows trends for the number of individuals in leadership positions (“Total No” and total FTEs allocated to leadership (“Total FTE”) over the last several surveys. The data is heavily skewed toward the right (positive); so, it makes sense to use the median in this case rather than mean. A few factors could explain the right skew of data.
- Large groups of 30 or more hospitalists are increasing, and so is their leadership need.
- There is more recognition of the need for dedicated leadership individuals and FTE.
- The leadership is getting less concentrated among just one or a few leaders.
- Outliers on the high side.
- Lower bounds of 0 or 0.1 FTE.
Highest-ranked leader dedicated FTE and premium compensation
Another pleasing trend is an increase in dedicated FTE for the highest-paid leader. Like any skill-set development, leadership requires the investment of deliberate practice, financial acumen, negotiation skills, and increased vulnerability. Time helps way more in developing these skill sets than money. SoHM trends show increase in dedicated FTE for the highest physician leader over the years and static premium compensation.

At last, we can say median leadership is always better than “mean” leadership in skewed data. Pun apart, every group needs leadership, and SoHM offers a nice window to the trends in leadership amongst many practice groups. It is a valuable resource for every group.
Dr. Chadha is chief of the division of hospital medicine at the University of Kentucky Healthcare, Lexington. He actively leads efforts of recruiting, practice analysis, and operation of the group. He is finishing his first tenure in the Practice Analysis Committee. He is often found spending a lot more than required time with spreadsheets and graphs.
Reference
1. 2020 State of Hospital Medicine. www.hospitalmedicine.org/practice-management/shms-state-of-hospital-medicine/
Let me apologize for misleading all of you; this is not an article about malignant physician leaders; instead, it goes over the numbers and trends uncovered by the 2020 State of Hospital Medicine report (SoHM).1 The hospital medicine leader ends up doing many tasks like planning, growth, collaboration, finance, recruiting, scheduling, onboarding, coaching, and most near and dear to our hearts, putting out the fires and conflict resolution.
Ratio of leadership FTE to physician hospitalists FTE
If my pun has already put you off, you can avoid reading the rest of the piece and go to the 2020 SoHM to look at pages 52 (Table 3.7c), 121 (Table 4.7c), and 166 (Table 5.7c). It has a newly added table (3.7c), and it is phenomenal; it is the ratio of leadership FTE to physician hospitalists FTE. As an avid user of SoHM, I always ended up doing a makeshift calculation to “guesstimate” this number. Now that we have it calculated for us and the ultimate revelation lies in its narrow range across all groups. We might differ in the region, employment type, academics, teaching, or size, but this range is relatively narrow.

The median ratio of leadership FTE to total FTE lies between 2% and 5% in pediatric groups and between 3% and 6% for most adult groups. The only two outliers are on the adult side, with less than 5 FTE and multistate management companies. The higher median for the less than 5 FTE group size is understandable because of the small number of hospitalist FTEs that the leader’s time must be spread over. Even a small amount of dedicated leadership time will result in a high ratio of leader time to hospitalist clinical time if the group is very small. The multistate management company is probably a result of multiple layers of physician leadership (for example, regional medical directors) and travel-related time adjustments. Still, it raises the question of why the local leadership is not developed to decrease the leadership cost and better access.
Another helpful pattern is the decrease in standard deviation with the increase in group size. The hospital medicine leaders and CEOs of the hospital need to watch this number closely; any extremes on high or low side would be indicators for a deep dive in leadership structure and health.
Total number and total dedicated FTE for all physician leaders
Once we start seeing the differences between the mean and median of leadership data, we can see the median is relatively static while the mean has increased year after year and took a big jump in the 2020 SoHM. The chart below shows trends for the number of individuals in leadership positions (“Total No” and total FTEs allocated to leadership (“Total FTE”) over the last several surveys. The data is heavily skewed toward the right (positive); so, it makes sense to use the median in this case rather than mean. A few factors could explain the right skew of data.
- Large groups of 30 or more hospitalists are increasing, and so is their leadership need.
- There is more recognition of the need for dedicated leadership individuals and FTE.
- The leadership is getting less concentrated among just one or a few leaders.
- Outliers on the high side.
- Lower bounds of 0 or 0.1 FTE.
Highest-ranked leader dedicated FTE and premium compensation
Another pleasing trend is an increase in dedicated FTE for the highest-paid leader. Like any skill-set development, leadership requires the investment of deliberate practice, financial acumen, negotiation skills, and increased vulnerability. Time helps way more in developing these skill sets than money. SoHM trends show increase in dedicated FTE for the highest physician leader over the years and static premium compensation.

At last, we can say median leadership is always better than “mean” leadership in skewed data. Pun apart, every group needs leadership, and SoHM offers a nice window to the trends in leadership amongst many practice groups. It is a valuable resource for every group.
Dr. Chadha is chief of the division of hospital medicine at the University of Kentucky Healthcare, Lexington. He actively leads efforts of recruiting, practice analysis, and operation of the group. He is finishing his first tenure in the Practice Analysis Committee. He is often found spending a lot more than required time with spreadsheets and graphs.
Reference
1. 2020 State of Hospital Medicine. www.hospitalmedicine.org/practice-management/shms-state-of-hospital-medicine/
Pandemic restrictions ignite innovative pivot for psychiatry
As medical school faculty members – and our students – know well, the COVID-19 pandemic forced us to become creative and shift much of our curricula online. Many hospitals chose to limit medical student rotations because of safety concerns. Students fell victim to canceled psychiatry rotations and electives during the pandemic’s early days. Privacy issues, combined with stigma tied to mental illness, made this shift to virtual instruction particularly challenging. But as a field, we persevered! And, as we learned during our shift toward telemedicine, many of the changes we made in medical education are probably here to stay.
Our team at the New York Institute of Technology College of Osteopathic Medicine (NYITCOM) was able to implement a novel curriculum that allowed our students to learn psychiatry and maintain high-quality medical school education.
We developed an online course for third-year students’ rotation in psychiatry, with several modules that focused on a variety of psychiatric topics and disorders, including the basic classifications and categories of depression, anxiety, personality disorders, and psychotic disorders. There were also video encounters available showing actual patient encounters. On completion of the online module, a faculty session was held to discuss topics of concern/confusion to the students, areas of interest, and a variety of related topics, such as professionalism in psychiatry, essentials of the mental status exam, management of diverse populations, and COVID repercussions in psychiatry.
For fourth-year students, we developed a telemedicine psychiatry elective, which allowed the students to observe psychiatric evaluations, psychiatric medication review visits, and even follow-up psychotherapy sessions, with the school’s clinical psychologists. The new method was minimally invasive, and it was accepted by patients and welcomed by the students.
During a time when hospitals were limiting onsite student rotations and discouraging patient contact, medical students still needed to experience patient interactions. As the director of the school’s Center for Behavioral Health, I designed an additional program that allowed students to participate in observing patients who presented with psychiatric complaints and symptoms. It had to be confidential in nature, accessible, and safe.
I recalled my own training in a hospital setting, where students and residents were allowed to observe a patient being evaluated by an attending, through a one-way mirror. It was a method that was acceptable at the time in a hospital, but unfortunately, not in a private office setting. As such, students and residents experienced such an interaction in acute inpatient and/or outpatient clinics of a hospital. The experience was invaluable.
The concept was simple, yet very efficient. The clinicians in the Center for Behavioral Health were seeing all patients with psychiatric needs via a HIPAA-compliant telemedicine platform. Access was granted for students – with the patient’s consent – and they “entered the session” without being seen or heard. This presented little to no distraction to the patient, and the student was able to observe a range of clinical sessions.
The course also provided online supplemental modules, including essential psychiatric topics, psychopharmacology, and a psychotherapeutic module that discussed a myriad of therapeutic interventions. In addition, the student was supervised weekly by the course director, the psychopharmacologist, and the clinical psychologist. The course director provided daily wrap-up reviews as well.
Originally, this new approach was envisioned as a temporary solution for use during the pandemic. But it has become clear that this approach would be beneficial post pandemic as well. Most of the students who participated in the course were actually interested in pursuing psychiatry as their future specialty. It allowed them to observe a population of patients firsthand that they might encounter in private practice, as opposed to only hospital settings.
Being present in a session with a patient with psychiatric symptoms and diagnoses has always been a challenge. Many patients refuse to have another medical professional in the room because of the intimate details being discussed and their associated stigma. The patients’ inability to see or hear the student during the sessions allows them to ignore the students’ presence – or at least not be intimidated by it. This, therefore, allows the students access and affords them a unique and memorable educational experience.
The pandemic curtailed and altered medical students’ traditional exposure to patients, but we found innovative ways to redefine it. As difficult as COVID-19 has been for the health care community, we have been able to use the restrictions forced by the pandemic to identify innovative ways to improve the education of our medical students.
In addition to serving as director of the Center for Behavioral Health at NYITCOM in Old Westbury, N.Y., Dr. Jarkon is assistant professor in the department of family medicine. She has no disclosures.
As medical school faculty members – and our students – know well, the COVID-19 pandemic forced us to become creative and shift much of our curricula online. Many hospitals chose to limit medical student rotations because of safety concerns. Students fell victim to canceled psychiatry rotations and electives during the pandemic’s early days. Privacy issues, combined with stigma tied to mental illness, made this shift to virtual instruction particularly challenging. But as a field, we persevered! And, as we learned during our shift toward telemedicine, many of the changes we made in medical education are probably here to stay.
Our team at the New York Institute of Technology College of Osteopathic Medicine (NYITCOM) was able to implement a novel curriculum that allowed our students to learn psychiatry and maintain high-quality medical school education.
We developed an online course for third-year students’ rotation in psychiatry, with several modules that focused on a variety of psychiatric topics and disorders, including the basic classifications and categories of depression, anxiety, personality disorders, and psychotic disorders. There were also video encounters available showing actual patient encounters. On completion of the online module, a faculty session was held to discuss topics of concern/confusion to the students, areas of interest, and a variety of related topics, such as professionalism in psychiatry, essentials of the mental status exam, management of diverse populations, and COVID repercussions in psychiatry.
For fourth-year students, we developed a telemedicine psychiatry elective, which allowed the students to observe psychiatric evaluations, psychiatric medication review visits, and even follow-up psychotherapy sessions, with the school’s clinical psychologists. The new method was minimally invasive, and it was accepted by patients and welcomed by the students.
During a time when hospitals were limiting onsite student rotations and discouraging patient contact, medical students still needed to experience patient interactions. As the director of the school’s Center for Behavioral Health, I designed an additional program that allowed students to participate in observing patients who presented with psychiatric complaints and symptoms. It had to be confidential in nature, accessible, and safe.
I recalled my own training in a hospital setting, where students and residents were allowed to observe a patient being evaluated by an attending, through a one-way mirror. It was a method that was acceptable at the time in a hospital, but unfortunately, not in a private office setting. As such, students and residents experienced such an interaction in acute inpatient and/or outpatient clinics of a hospital. The experience was invaluable.
The concept was simple, yet very efficient. The clinicians in the Center for Behavioral Health were seeing all patients with psychiatric needs via a HIPAA-compliant telemedicine platform. Access was granted for students – with the patient’s consent – and they “entered the session” without being seen or heard. This presented little to no distraction to the patient, and the student was able to observe a range of clinical sessions.
The course also provided online supplemental modules, including essential psychiatric topics, psychopharmacology, and a psychotherapeutic module that discussed a myriad of therapeutic interventions. In addition, the student was supervised weekly by the course director, the psychopharmacologist, and the clinical psychologist. The course director provided daily wrap-up reviews as well.
Originally, this new approach was envisioned as a temporary solution for use during the pandemic. But it has become clear that this approach would be beneficial post pandemic as well. Most of the students who participated in the course were actually interested in pursuing psychiatry as their future specialty. It allowed them to observe a population of patients firsthand that they might encounter in private practice, as opposed to only hospital settings.
Being present in a session with a patient with psychiatric symptoms and diagnoses has always been a challenge. Many patients refuse to have another medical professional in the room because of the intimate details being discussed and their associated stigma. The patients’ inability to see or hear the student during the sessions allows them to ignore the students’ presence – or at least not be intimidated by it. This, therefore, allows the students access and affords them a unique and memorable educational experience.
The pandemic curtailed and altered medical students’ traditional exposure to patients, but we found innovative ways to redefine it. As difficult as COVID-19 has been for the health care community, we have been able to use the restrictions forced by the pandemic to identify innovative ways to improve the education of our medical students.
In addition to serving as director of the Center for Behavioral Health at NYITCOM in Old Westbury, N.Y., Dr. Jarkon is assistant professor in the department of family medicine. She has no disclosures.
As medical school faculty members – and our students – know well, the COVID-19 pandemic forced us to become creative and shift much of our curricula online. Many hospitals chose to limit medical student rotations because of safety concerns. Students fell victim to canceled psychiatry rotations and electives during the pandemic’s early days. Privacy issues, combined with stigma tied to mental illness, made this shift to virtual instruction particularly challenging. But as a field, we persevered! And, as we learned during our shift toward telemedicine, many of the changes we made in medical education are probably here to stay.
Our team at the New York Institute of Technology College of Osteopathic Medicine (NYITCOM) was able to implement a novel curriculum that allowed our students to learn psychiatry and maintain high-quality medical school education.
We developed an online course for third-year students’ rotation in psychiatry, with several modules that focused on a variety of psychiatric topics and disorders, including the basic classifications and categories of depression, anxiety, personality disorders, and psychotic disorders. There were also video encounters available showing actual patient encounters. On completion of the online module, a faculty session was held to discuss topics of concern/confusion to the students, areas of interest, and a variety of related topics, such as professionalism in psychiatry, essentials of the mental status exam, management of diverse populations, and COVID repercussions in psychiatry.
For fourth-year students, we developed a telemedicine psychiatry elective, which allowed the students to observe psychiatric evaluations, psychiatric medication review visits, and even follow-up psychotherapy sessions, with the school’s clinical psychologists. The new method was minimally invasive, and it was accepted by patients and welcomed by the students.
During a time when hospitals were limiting onsite student rotations and discouraging patient contact, medical students still needed to experience patient interactions. As the director of the school’s Center for Behavioral Health, I designed an additional program that allowed students to participate in observing patients who presented with psychiatric complaints and symptoms. It had to be confidential in nature, accessible, and safe.
I recalled my own training in a hospital setting, where students and residents were allowed to observe a patient being evaluated by an attending, through a one-way mirror. It was a method that was acceptable at the time in a hospital, but unfortunately, not in a private office setting. As such, students and residents experienced such an interaction in acute inpatient and/or outpatient clinics of a hospital. The experience was invaluable.
The concept was simple, yet very efficient. The clinicians in the Center for Behavioral Health were seeing all patients with psychiatric needs via a HIPAA-compliant telemedicine platform. Access was granted for students – with the patient’s consent – and they “entered the session” without being seen or heard. This presented little to no distraction to the patient, and the student was able to observe a range of clinical sessions.
The course also provided online supplemental modules, including essential psychiatric topics, psychopharmacology, and a psychotherapeutic module that discussed a myriad of therapeutic interventions. In addition, the student was supervised weekly by the course director, the psychopharmacologist, and the clinical psychologist. The course director provided daily wrap-up reviews as well.
Originally, this new approach was envisioned as a temporary solution for use during the pandemic. But it has become clear that this approach would be beneficial post pandemic as well. Most of the students who participated in the course were actually interested in pursuing psychiatry as their future specialty. It allowed them to observe a population of patients firsthand that they might encounter in private practice, as opposed to only hospital settings.
Being present in a session with a patient with psychiatric symptoms and diagnoses has always been a challenge. Many patients refuse to have another medical professional in the room because of the intimate details being discussed and their associated stigma. The patients’ inability to see or hear the student during the sessions allows them to ignore the students’ presence – or at least not be intimidated by it. This, therefore, allows the students access and affords them a unique and memorable educational experience.
The pandemic curtailed and altered medical students’ traditional exposure to patients, but we found innovative ways to redefine it. As difficult as COVID-19 has been for the health care community, we have been able to use the restrictions forced by the pandemic to identify innovative ways to improve the education of our medical students.
In addition to serving as director of the Center for Behavioral Health at NYITCOM in Old Westbury, N.Y., Dr. Jarkon is assistant professor in the department of family medicine. She has no disclosures.
Cardiogenic shock teams again tied to lower mortality
A large multicenter study provides further evidence supporting the rationale for multidisciplinary teams for cardiogenic shock, one of the most lethal diseases in cardiovascular medicine.
The analysis of 24 critical care ICUs in the Critical Care Cardiology Trials Network showed that the presence of a shock team was independently associated with a 28% lower risk for CICU mortality (23% vs. 29%; odds ratio, 0.72; P = .016).
Patients treated by a shock team also had significantly shorter CICU stays and less need for mechanical ventilation or renal replacement therapy, as reported in the Journal of the American College of Cardiology.
“It’s observational, but the association that we’re seeing here, just because of our sample size, is the strongest that’s been published yet,” lead author Alexander Papolos, MD, MedStar Washington Hospital Center, said in an interview.
Although a causal relationship cannot be drawn, the authors suggest several factors that could explain the findings, including a shock team’s ability to rapidly diagnose and treat cardiogenic shock before multiorgan dysfunction occurs.
Centers with shock teams also used significantly more pulmonary artery catheters (60% vs. 49%; adjusted OR, 1.86; P < .001) and placed them earlier (0.3 vs. 0.66 days; P = .019).
Pulmonary artery catheter (PAC) use has declined after earlier trials like ESCAPE showed little or no benefit in other acutely ill patient groups, but positive results have been reported recently in cardiogenic shock, where a PAC is needed to determine the severity of the lesion and the phenotype, Dr. Papolos observed.
A 2018 study showed PAC use was tied to increased survival among patients with acute myocardial infarction cardiogenic shock (AMI-CS) supported with the Impella (Abiomed) device. Additionally, a 2021 study by the Cardiogenic Shock Working Group demonstrated a dose-dependent survival response based on the completeness of hemodynamic assessment by PAC prior to initiating mechanical circulatory support (MCS).
A third factor might be that a structured, team-based evaluation can facilitate timely and optimal MCS device selection, deployment, and management, suggested Dr. Papolos.
Centers with shock teams used more advanced types of MCS – defined as Impella, TandemHeart (LivaNova), extracorporeal membrane oxygenation, and temporary or durable surgical ventricular assist devices – than those without a shock team (53% vs. 43%; adjusted OR, 1.73; P = .005) and did so more often as the initial device (42% vs. 28%; P = .002).
Overall MCS use was lower at shock team centers (35% vs. 43%), driven by less frequent use of intra-aortic balloon pumps (58% vs. 72%).
“The standard, basic MCS has always been the balloon pump because it’s something that’s easy to put in at the cath lab or at the bedside,” Dr. Papolos said. “So, if you take away having all of the information and having the right people at the table to discuss what the best level of support is, then you’re going to end up with balloon pumps, and that’s what we saw here.”
The study involved 6,872 consecutive medical admissions at 24 level 1 CICU centers during an annual 2-month period from 2017 to 2019. Of these, 1,242 admissions were for cardiogenic shock and 546 (44%) were treated at one of 10 centers with a shock team.
Shock team centers had higher-acuity patients than centers without a shock team (Sequential Organ Failure Assessment score, 4 vs. 3) but a similar proportion of patients with AMI-CS (27% vs. 28%).
Among all admissions, CICU mortality was not significantly different between centers with and without a shock team.
For cardiogenic shock patients treated at centers with and without a shock team, the median CICU stay was 4.0 and 5.1 days, respectively, mechanical ventilation was used in 41% and 52%, respectively, and new renal replacement therapy in 11% and 19%, respectively (P < .001 for all).
Shock team centers used significantly more PACs for AMI-CS and non–AMI-CS admissions; advanced MCS therapy was also greater in the AMI-CS subgroup.
Lower CICU mortality at shock team centers persisted among patients with non-AMI-CS (adjusted OR, 0.67; P = .017) and AMI-CS (adjusted OR, 0.79; P = .344).
“This analysis supports that all AHA level 1 cardiac ICUs should strongly consider having a shock team,” Dr. Papolos said.
Evidence from single centers and the National Cardiogenic Shock Initiative has shown improved survival with a cardiogenic shock algorithm, but this is the first report specifically comparing no shock teams with shock teams, Perwaiz Meraj, MD, Northwell Health, Manhansett, N.Y., told this news organization.
“People may say that it’s just another paper that’s saying, ‘shock teams, shock teams, rah, rah, rah,’ but it’s important for all of us to really take a close look under the covers and see how are we best managing these patients, what teams are we putting together, and to create systems of care, where if you’re at a center that really doesn’t have the capabilities of doing this, then you should partner up with a center that does,” he said.
Notably, the 10 shock teams were present only in medium or large urban, academic medical centers with more than 500 beds. Although they followed individual protocols, survey results show service-line representation, structure, and operations were similar across centers.
They all had a centralized way to activate the shock team, the service was 24/7, and members came from areas such as critical care cardiology (100%), cardiac surgery (100%), interventional cardiology (90%), advanced heart failure (80%), and extracorporeal membrane oxygenation service (70%).
Limitations of the study include the possibility of residual confounding, the fact that the registry did not capture patients with cardiogenic shock managed outside the CICU or the time of onset of cardiogenic shock, and data were limited on inotropic strategies, sedation practices, and ventilator management, the authors wrote.
“Although many critics will continue to discuss the lack of randomized controlled trials in cardiogenic shock, this paper supports the process previously outlined of a multidisciplinary team-based approach improving survival,” Dr. Meraj and William W. O’Neill, MD, director of the Center for Structural Heart Disease and Henry Ford Health System, Detroit, and the force behind the National Cardiogenic Shock Initiative, wrote in an accompanying editorial.
They point out that the report doesn’t address the escalation of care based on invasive hemodynamics in the CICU and the protocols to prevent acute vascular/limb complications (ALI) that can arise from the use of MCS.
“Many procedural techniques and novel CICU models exist to mitigate the risk of ALI in CS patients with MCS,” they wrote. “Finally, escalation of care and support is vital to the continued success of any shock team and center.”
One coauthor has served as a consultant to Abbott. Another has served as a consultant to the Abiomed critical care advisory board. All other authors reported having no relevant financial relationships. Dr. Meraj has received research and grant funding from Abiomed, Medtronic, CSI, and Boston Scientific. Dr. O’Neill has received consulting/speaker honoraria from Abiomed, Boston Scientific, and Abbott.
A version of this article first appeared on Medscape.com.
A large multicenter study provides further evidence supporting the rationale for multidisciplinary teams for cardiogenic shock, one of the most lethal diseases in cardiovascular medicine.
The analysis of 24 critical care ICUs in the Critical Care Cardiology Trials Network showed that the presence of a shock team was independently associated with a 28% lower risk for CICU mortality (23% vs. 29%; odds ratio, 0.72; P = .016).
Patients treated by a shock team also had significantly shorter CICU stays and less need for mechanical ventilation or renal replacement therapy, as reported in the Journal of the American College of Cardiology.
“It’s observational, but the association that we’re seeing here, just because of our sample size, is the strongest that’s been published yet,” lead author Alexander Papolos, MD, MedStar Washington Hospital Center, said in an interview.
Although a causal relationship cannot be drawn, the authors suggest several factors that could explain the findings, including a shock team’s ability to rapidly diagnose and treat cardiogenic shock before multiorgan dysfunction occurs.
Centers with shock teams also used significantly more pulmonary artery catheters (60% vs. 49%; adjusted OR, 1.86; P < .001) and placed them earlier (0.3 vs. 0.66 days; P = .019).
Pulmonary artery catheter (PAC) use has declined after earlier trials like ESCAPE showed little or no benefit in other acutely ill patient groups, but positive results have been reported recently in cardiogenic shock, where a PAC is needed to determine the severity of the lesion and the phenotype, Dr. Papolos observed.
A 2018 study showed PAC use was tied to increased survival among patients with acute myocardial infarction cardiogenic shock (AMI-CS) supported with the Impella (Abiomed) device. Additionally, a 2021 study by the Cardiogenic Shock Working Group demonstrated a dose-dependent survival response based on the completeness of hemodynamic assessment by PAC prior to initiating mechanical circulatory support (MCS).
A third factor might be that a structured, team-based evaluation can facilitate timely and optimal MCS device selection, deployment, and management, suggested Dr. Papolos.
Centers with shock teams used more advanced types of MCS – defined as Impella, TandemHeart (LivaNova), extracorporeal membrane oxygenation, and temporary or durable surgical ventricular assist devices – than those without a shock team (53% vs. 43%; adjusted OR, 1.73; P = .005) and did so more often as the initial device (42% vs. 28%; P = .002).
Overall MCS use was lower at shock team centers (35% vs. 43%), driven by less frequent use of intra-aortic balloon pumps (58% vs. 72%).
“The standard, basic MCS has always been the balloon pump because it’s something that’s easy to put in at the cath lab or at the bedside,” Dr. Papolos said. “So, if you take away having all of the information and having the right people at the table to discuss what the best level of support is, then you’re going to end up with balloon pumps, and that’s what we saw here.”
The study involved 6,872 consecutive medical admissions at 24 level 1 CICU centers during an annual 2-month period from 2017 to 2019. Of these, 1,242 admissions were for cardiogenic shock and 546 (44%) were treated at one of 10 centers with a shock team.
Shock team centers had higher-acuity patients than centers without a shock team (Sequential Organ Failure Assessment score, 4 vs. 3) but a similar proportion of patients with AMI-CS (27% vs. 28%).
Among all admissions, CICU mortality was not significantly different between centers with and without a shock team.
For cardiogenic shock patients treated at centers with and without a shock team, the median CICU stay was 4.0 and 5.1 days, respectively, mechanical ventilation was used in 41% and 52%, respectively, and new renal replacement therapy in 11% and 19%, respectively (P < .001 for all).
Shock team centers used significantly more PACs for AMI-CS and non–AMI-CS admissions; advanced MCS therapy was also greater in the AMI-CS subgroup.
Lower CICU mortality at shock team centers persisted among patients with non-AMI-CS (adjusted OR, 0.67; P = .017) and AMI-CS (adjusted OR, 0.79; P = .344).
“This analysis supports that all AHA level 1 cardiac ICUs should strongly consider having a shock team,” Dr. Papolos said.
Evidence from single centers and the National Cardiogenic Shock Initiative has shown improved survival with a cardiogenic shock algorithm, but this is the first report specifically comparing no shock teams with shock teams, Perwaiz Meraj, MD, Northwell Health, Manhansett, N.Y., told this news organization.
“People may say that it’s just another paper that’s saying, ‘shock teams, shock teams, rah, rah, rah,’ but it’s important for all of us to really take a close look under the covers and see how are we best managing these patients, what teams are we putting together, and to create systems of care, where if you’re at a center that really doesn’t have the capabilities of doing this, then you should partner up with a center that does,” he said.
Notably, the 10 shock teams were present only in medium or large urban, academic medical centers with more than 500 beds. Although they followed individual protocols, survey results show service-line representation, structure, and operations were similar across centers.
They all had a centralized way to activate the shock team, the service was 24/7, and members came from areas such as critical care cardiology (100%), cardiac surgery (100%), interventional cardiology (90%), advanced heart failure (80%), and extracorporeal membrane oxygenation service (70%).
Limitations of the study include the possibility of residual confounding, the fact that the registry did not capture patients with cardiogenic shock managed outside the CICU or the time of onset of cardiogenic shock, and data were limited on inotropic strategies, sedation practices, and ventilator management, the authors wrote.
“Although many critics will continue to discuss the lack of randomized controlled trials in cardiogenic shock, this paper supports the process previously outlined of a multidisciplinary team-based approach improving survival,” Dr. Meraj and William W. O’Neill, MD, director of the Center for Structural Heart Disease and Henry Ford Health System, Detroit, and the force behind the National Cardiogenic Shock Initiative, wrote in an accompanying editorial.
They point out that the report doesn’t address the escalation of care based on invasive hemodynamics in the CICU and the protocols to prevent acute vascular/limb complications (ALI) that can arise from the use of MCS.
“Many procedural techniques and novel CICU models exist to mitigate the risk of ALI in CS patients with MCS,” they wrote. “Finally, escalation of care and support is vital to the continued success of any shock team and center.”
One coauthor has served as a consultant to Abbott. Another has served as a consultant to the Abiomed critical care advisory board. All other authors reported having no relevant financial relationships. Dr. Meraj has received research and grant funding from Abiomed, Medtronic, CSI, and Boston Scientific. Dr. O’Neill has received consulting/speaker honoraria from Abiomed, Boston Scientific, and Abbott.
A version of this article first appeared on Medscape.com.
A large multicenter study provides further evidence supporting the rationale for multidisciplinary teams for cardiogenic shock, one of the most lethal diseases in cardiovascular medicine.
The analysis of 24 critical care ICUs in the Critical Care Cardiology Trials Network showed that the presence of a shock team was independently associated with a 28% lower risk for CICU mortality (23% vs. 29%; odds ratio, 0.72; P = .016).
Patients treated by a shock team also had significantly shorter CICU stays and less need for mechanical ventilation or renal replacement therapy, as reported in the Journal of the American College of Cardiology.
“It’s observational, but the association that we’re seeing here, just because of our sample size, is the strongest that’s been published yet,” lead author Alexander Papolos, MD, MedStar Washington Hospital Center, said in an interview.
Although a causal relationship cannot be drawn, the authors suggest several factors that could explain the findings, including a shock team’s ability to rapidly diagnose and treat cardiogenic shock before multiorgan dysfunction occurs.
Centers with shock teams also used significantly more pulmonary artery catheters (60% vs. 49%; adjusted OR, 1.86; P < .001) and placed them earlier (0.3 vs. 0.66 days; P = .019).
Pulmonary artery catheter (PAC) use has declined after earlier trials like ESCAPE showed little or no benefit in other acutely ill patient groups, but positive results have been reported recently in cardiogenic shock, where a PAC is needed to determine the severity of the lesion and the phenotype, Dr. Papolos observed.
A 2018 study showed PAC use was tied to increased survival among patients with acute myocardial infarction cardiogenic shock (AMI-CS) supported with the Impella (Abiomed) device. Additionally, a 2021 study by the Cardiogenic Shock Working Group demonstrated a dose-dependent survival response based on the completeness of hemodynamic assessment by PAC prior to initiating mechanical circulatory support (MCS).
A third factor might be that a structured, team-based evaluation can facilitate timely and optimal MCS device selection, deployment, and management, suggested Dr. Papolos.
Centers with shock teams used more advanced types of MCS – defined as Impella, TandemHeart (LivaNova), extracorporeal membrane oxygenation, and temporary or durable surgical ventricular assist devices – than those without a shock team (53% vs. 43%; adjusted OR, 1.73; P = .005) and did so more often as the initial device (42% vs. 28%; P = .002).
Overall MCS use was lower at shock team centers (35% vs. 43%), driven by less frequent use of intra-aortic balloon pumps (58% vs. 72%).
“The standard, basic MCS has always been the balloon pump because it’s something that’s easy to put in at the cath lab or at the bedside,” Dr. Papolos said. “So, if you take away having all of the information and having the right people at the table to discuss what the best level of support is, then you’re going to end up with balloon pumps, and that’s what we saw here.”
The study involved 6,872 consecutive medical admissions at 24 level 1 CICU centers during an annual 2-month period from 2017 to 2019. Of these, 1,242 admissions were for cardiogenic shock and 546 (44%) were treated at one of 10 centers with a shock team.
Shock team centers had higher-acuity patients than centers without a shock team (Sequential Organ Failure Assessment score, 4 vs. 3) but a similar proportion of patients with AMI-CS (27% vs. 28%).
Among all admissions, CICU mortality was not significantly different between centers with and without a shock team.
For cardiogenic shock patients treated at centers with and without a shock team, the median CICU stay was 4.0 and 5.1 days, respectively, mechanical ventilation was used in 41% and 52%, respectively, and new renal replacement therapy in 11% and 19%, respectively (P < .001 for all).
Shock team centers used significantly more PACs for AMI-CS and non–AMI-CS admissions; advanced MCS therapy was also greater in the AMI-CS subgroup.
Lower CICU mortality at shock team centers persisted among patients with non-AMI-CS (adjusted OR, 0.67; P = .017) and AMI-CS (adjusted OR, 0.79; P = .344).
“This analysis supports that all AHA level 1 cardiac ICUs should strongly consider having a shock team,” Dr. Papolos said.
Evidence from single centers and the National Cardiogenic Shock Initiative has shown improved survival with a cardiogenic shock algorithm, but this is the first report specifically comparing no shock teams with shock teams, Perwaiz Meraj, MD, Northwell Health, Manhansett, N.Y., told this news organization.
“People may say that it’s just another paper that’s saying, ‘shock teams, shock teams, rah, rah, rah,’ but it’s important for all of us to really take a close look under the covers and see how are we best managing these patients, what teams are we putting together, and to create systems of care, where if you’re at a center that really doesn’t have the capabilities of doing this, then you should partner up with a center that does,” he said.
Notably, the 10 shock teams were present only in medium or large urban, academic medical centers with more than 500 beds. Although they followed individual protocols, survey results show service-line representation, structure, and operations were similar across centers.
They all had a centralized way to activate the shock team, the service was 24/7, and members came from areas such as critical care cardiology (100%), cardiac surgery (100%), interventional cardiology (90%), advanced heart failure (80%), and extracorporeal membrane oxygenation service (70%).
Limitations of the study include the possibility of residual confounding, the fact that the registry did not capture patients with cardiogenic shock managed outside the CICU or the time of onset of cardiogenic shock, and data were limited on inotropic strategies, sedation practices, and ventilator management, the authors wrote.
“Although many critics will continue to discuss the lack of randomized controlled trials in cardiogenic shock, this paper supports the process previously outlined of a multidisciplinary team-based approach improving survival,” Dr. Meraj and William W. O’Neill, MD, director of the Center for Structural Heart Disease and Henry Ford Health System, Detroit, and the force behind the National Cardiogenic Shock Initiative, wrote in an accompanying editorial.
They point out that the report doesn’t address the escalation of care based on invasive hemodynamics in the CICU and the protocols to prevent acute vascular/limb complications (ALI) that can arise from the use of MCS.
“Many procedural techniques and novel CICU models exist to mitigate the risk of ALI in CS patients with MCS,” they wrote. “Finally, escalation of care and support is vital to the continued success of any shock team and center.”
One coauthor has served as a consultant to Abbott. Another has served as a consultant to the Abiomed critical care advisory board. All other authors reported having no relevant financial relationships. Dr. Meraj has received research and grant funding from Abiomed, Medtronic, CSI, and Boston Scientific. Dr. O’Neill has received consulting/speaker honoraria from Abiomed, Boston Scientific, and Abbott.
A version of this article first appeared on Medscape.com.
Consensus statement warns against acetaminophen use during pregnancy
Pregnant women should use paracetamol/acetaminophen only with a medical indication and at the lowest effective dose for the shortest possible time, according to an international consensus statement published online Sept. 23 in Nature Reviews Endocrinology.
With global rates of use high and risks considered negligible, the expert panel of 13 U.S. and European authors call for focused research into how this analgesic and febrifuge may impair fetal development and lead to adverse outcomes in children. They outline several precautionary measures to be taken in the meantime.
According to first author and epidemiologist Ann Z. Bauer, ScD, a postdoctoral research fellow at the University of Massachusetts in Lowell, and colleagues, this drug is used by an estimated 65% of pregnant women in the United States, and more than 50% worldwide. It is currently the active ingredient in more than 600 prescription and nonprescription medications, including Tylenol, which historically has been deemed safe in all trimesters of pregnancy.
But a growing body of experimental and epidemiological evidence suggests prenatal exposure to paracetamol (N-acetyl-p-aminophenol, or APAP) might alter fetal development and elevate the risks of neurodevelopmental, reproductive and urogenital disorders in both sexes. Exposure in utero has been linked, for example, to potential behavioral problems in children.
The new recommendations are based on a review of experimental animal and cell-based research as well as human epidemiological data published from January 1995 to October 2020. The authors include clinicians, epidemiologists, and scientists specializing in toxicology, endocrinology, reproductive medicine and neurodevelopment.
Recommendations
Although the new guidance does not differ markedly from current advice, the authors believe stronger communication and greater awareness of risks are needed. In addition to restricting use of this medication to low doses for short periods when medically necessary, expectant mothers should receive counseling before conception or early in pregnancy. If uncertain about its use, they should consult their physicians or pharmacists.
In other recommendations, the panel said:
- The 2015 FDA Drug Safety Communication recommendations should be updated based on evaluation of all available scientific evidence.
- The European Medicines Agency Pharmacovigilance Risk Assessment Committee should review the most recent epidemiologic and experimental research and issue an updated Drug Safety Communication.
- Obstetric and gynecological associations should update their guidance after reviewing all available research.
- The Acetaminophen Awareness Coalition (“Know Your Dose” Campaign) should add standardized warnings and specifically advise pregnant women to forgo APAP unless it’s medically indicated.
- All sales of APAP-containing medications should be accompanied by recommendations specifically for use in pregnancy. This information should include warning labels on packaging, and if possible, APAP should be sold only in pharmacies (as in France).
Mechanism of action
APAP is an endocrine disruptor (Neuroscientist. 2020 Sep 11. doi: 10.1177/1073858420952046). “Chemicals that disrupt the endocrine system are concerning because they can interfere with the activity of endogenous hormones that are essential for healthy neurological, urogenital, and reproductive development,” researchers wrote.
“The precise mechanism is not clear but its toxicity is thought to be due mainly to hormone disruption,” Dr. Bauer said in an interview.
Moreover, APAP readily crosses the placenta and blood–brain barrier, and changes in APAP metabolism during pregnancy might make women and their fetuses more vulnerable to its toxic effects. For instance, the molar dose fraction of APAP converted to the oxidative metabolite N-acetyl-p-benzoquinone imine increases during pregnancy. In addition to its hepatotoxicity, this poisonous byproduct is thought to be a genotoxin that increases DNA cleavage by acting on the enzyme topoisomerase II.
Asked for her perspective on the statement, Kjersti Aagaard, MD, PhD, a professor of obstetrics and gynecology at Baylor College of Medicine and Texas Children’s Hospital in Houston, called the expert panel’s statement thoughtful and comprehensive, but she urged caution in interpreting the role of acetaminophen.
The challenge in linking any commonly used medication to adverse effects and congenital defects, she said, is “teasing out an association from causation. Given the commonality of the use of acetaminophen with the relative rarity of the outcomes, it is clear that not all cases of exposure result in adverse outcomes.”
As for judicious use, she said, one would be to reduce a high fever, which can cause miscarriage, neural tube defects, and potential heart disease in adulthood. Acetaminophen is the drug of choice in this case since nonsteroidal anti-inflammatory drugs such as ibuprofen are not recommended owing to their known risks to the fetal heart.
Dr. Aagaard emphasized that while acetaminophen use is temporally associated with learning and behavioral problems, and urogenital disorders at birth in male infants such as like hypospadias, so is exposure to multiple environmental chemicals and pollutants, as well as climate change. “It would be a real mistake with real life implications if we associated any congenital disease or disorder with a commonly used medication with known benefits if the true causal link lies elsewhere.”
She said the precautionary statements fall into the time-honored therapeutic principle of first do no harm. “However, the call for research action must be undertaken earnestly and sincerely.”
According to Dr. Bauer, the statement’s essential take-home message is that “physicians should educate themselves and educate women about what we’re learning about the risks of acetaminophen in pregnancy.” Risk can be minimized by using the lowest effective dose for the shortest time and only when medically indicated. “Pregnant women should speak to their physicians about acetaminophen. It’s about empowerment and making smart decisions,” she said.
This study received no specific funding. Coauthor Dr. R.T. Mitchell is supported by a UK Research Institute fellowship.
Pregnant women should use paracetamol/acetaminophen only with a medical indication and at the lowest effective dose for the shortest possible time, according to an international consensus statement published online Sept. 23 in Nature Reviews Endocrinology.
With global rates of use high and risks considered negligible, the expert panel of 13 U.S. and European authors call for focused research into how this analgesic and febrifuge may impair fetal development and lead to adverse outcomes in children. They outline several precautionary measures to be taken in the meantime.
According to first author and epidemiologist Ann Z. Bauer, ScD, a postdoctoral research fellow at the University of Massachusetts in Lowell, and colleagues, this drug is used by an estimated 65% of pregnant women in the United States, and more than 50% worldwide. It is currently the active ingredient in more than 600 prescription and nonprescription medications, including Tylenol, which historically has been deemed safe in all trimesters of pregnancy.
But a growing body of experimental and epidemiological evidence suggests prenatal exposure to paracetamol (N-acetyl-p-aminophenol, or APAP) might alter fetal development and elevate the risks of neurodevelopmental, reproductive and urogenital disorders in both sexes. Exposure in utero has been linked, for example, to potential behavioral problems in children.
The new recommendations are based on a review of experimental animal and cell-based research as well as human epidemiological data published from January 1995 to October 2020. The authors include clinicians, epidemiologists, and scientists specializing in toxicology, endocrinology, reproductive medicine and neurodevelopment.
Recommendations
Although the new guidance does not differ markedly from current advice, the authors believe stronger communication and greater awareness of risks are needed. In addition to restricting use of this medication to low doses for short periods when medically necessary, expectant mothers should receive counseling before conception or early in pregnancy. If uncertain about its use, they should consult their physicians or pharmacists.
In other recommendations, the panel said:
- The 2015 FDA Drug Safety Communication recommendations should be updated based on evaluation of all available scientific evidence.
- The European Medicines Agency Pharmacovigilance Risk Assessment Committee should review the most recent epidemiologic and experimental research and issue an updated Drug Safety Communication.
- Obstetric and gynecological associations should update their guidance after reviewing all available research.
- The Acetaminophen Awareness Coalition (“Know Your Dose” Campaign) should add standardized warnings and specifically advise pregnant women to forgo APAP unless it’s medically indicated.
- All sales of APAP-containing medications should be accompanied by recommendations specifically for use in pregnancy. This information should include warning labels on packaging, and if possible, APAP should be sold only in pharmacies (as in France).
Mechanism of action
APAP is an endocrine disruptor (Neuroscientist. 2020 Sep 11. doi: 10.1177/1073858420952046). “Chemicals that disrupt the endocrine system are concerning because they can interfere with the activity of endogenous hormones that are essential for healthy neurological, urogenital, and reproductive development,” researchers wrote.
“The precise mechanism is not clear but its toxicity is thought to be due mainly to hormone disruption,” Dr. Bauer said in an interview.
Moreover, APAP readily crosses the placenta and blood–brain barrier, and changes in APAP metabolism during pregnancy might make women and their fetuses more vulnerable to its toxic effects. For instance, the molar dose fraction of APAP converted to the oxidative metabolite N-acetyl-p-benzoquinone imine increases during pregnancy. In addition to its hepatotoxicity, this poisonous byproduct is thought to be a genotoxin that increases DNA cleavage by acting on the enzyme topoisomerase II.
Asked for her perspective on the statement, Kjersti Aagaard, MD, PhD, a professor of obstetrics and gynecology at Baylor College of Medicine and Texas Children’s Hospital in Houston, called the expert panel’s statement thoughtful and comprehensive, but she urged caution in interpreting the role of acetaminophen.
The challenge in linking any commonly used medication to adverse effects and congenital defects, she said, is “teasing out an association from causation. Given the commonality of the use of acetaminophen with the relative rarity of the outcomes, it is clear that not all cases of exposure result in adverse outcomes.”
As for judicious use, she said, one would be to reduce a high fever, which can cause miscarriage, neural tube defects, and potential heart disease in adulthood. Acetaminophen is the drug of choice in this case since nonsteroidal anti-inflammatory drugs such as ibuprofen are not recommended owing to their known risks to the fetal heart.
Dr. Aagaard emphasized that while acetaminophen use is temporally associated with learning and behavioral problems, and urogenital disorders at birth in male infants such as like hypospadias, so is exposure to multiple environmental chemicals and pollutants, as well as climate change. “It would be a real mistake with real life implications if we associated any congenital disease or disorder with a commonly used medication with known benefits if the true causal link lies elsewhere.”
She said the precautionary statements fall into the time-honored therapeutic principle of first do no harm. “However, the call for research action must be undertaken earnestly and sincerely.”
According to Dr. Bauer, the statement’s essential take-home message is that “physicians should educate themselves and educate women about what we’re learning about the risks of acetaminophen in pregnancy.” Risk can be minimized by using the lowest effective dose for the shortest time and only when medically indicated. “Pregnant women should speak to their physicians about acetaminophen. It’s about empowerment and making smart decisions,” she said.
This study received no specific funding. Coauthor Dr. R.T. Mitchell is supported by a UK Research Institute fellowship.
Pregnant women should use paracetamol/acetaminophen only with a medical indication and at the lowest effective dose for the shortest possible time, according to an international consensus statement published online Sept. 23 in Nature Reviews Endocrinology.
With global rates of use high and risks considered negligible, the expert panel of 13 U.S. and European authors call for focused research into how this analgesic and febrifuge may impair fetal development and lead to adverse outcomes in children. They outline several precautionary measures to be taken in the meantime.
According to first author and epidemiologist Ann Z. Bauer, ScD, a postdoctoral research fellow at the University of Massachusetts in Lowell, and colleagues, this drug is used by an estimated 65% of pregnant women in the United States, and more than 50% worldwide. It is currently the active ingredient in more than 600 prescription and nonprescription medications, including Tylenol, which historically has been deemed safe in all trimesters of pregnancy.
But a growing body of experimental and epidemiological evidence suggests prenatal exposure to paracetamol (N-acetyl-p-aminophenol, or APAP) might alter fetal development and elevate the risks of neurodevelopmental, reproductive and urogenital disorders in both sexes. Exposure in utero has been linked, for example, to potential behavioral problems in children.
The new recommendations are based on a review of experimental animal and cell-based research as well as human epidemiological data published from January 1995 to October 2020. The authors include clinicians, epidemiologists, and scientists specializing in toxicology, endocrinology, reproductive medicine and neurodevelopment.
Recommendations
Although the new guidance does not differ markedly from current advice, the authors believe stronger communication and greater awareness of risks are needed. In addition to restricting use of this medication to low doses for short periods when medically necessary, expectant mothers should receive counseling before conception or early in pregnancy. If uncertain about its use, they should consult their physicians or pharmacists.
In other recommendations, the panel said:
- The 2015 FDA Drug Safety Communication recommendations should be updated based on evaluation of all available scientific evidence.
- The European Medicines Agency Pharmacovigilance Risk Assessment Committee should review the most recent epidemiologic and experimental research and issue an updated Drug Safety Communication.
- Obstetric and gynecological associations should update their guidance after reviewing all available research.
- The Acetaminophen Awareness Coalition (“Know Your Dose” Campaign) should add standardized warnings and specifically advise pregnant women to forgo APAP unless it’s medically indicated.
- All sales of APAP-containing medications should be accompanied by recommendations specifically for use in pregnancy. This information should include warning labels on packaging, and if possible, APAP should be sold only in pharmacies (as in France).
Mechanism of action
APAP is an endocrine disruptor (Neuroscientist. 2020 Sep 11. doi: 10.1177/1073858420952046). “Chemicals that disrupt the endocrine system are concerning because they can interfere with the activity of endogenous hormones that are essential for healthy neurological, urogenital, and reproductive development,” researchers wrote.
“The precise mechanism is not clear but its toxicity is thought to be due mainly to hormone disruption,” Dr. Bauer said in an interview.
Moreover, APAP readily crosses the placenta and blood–brain barrier, and changes in APAP metabolism during pregnancy might make women and their fetuses more vulnerable to its toxic effects. For instance, the molar dose fraction of APAP converted to the oxidative metabolite N-acetyl-p-benzoquinone imine increases during pregnancy. In addition to its hepatotoxicity, this poisonous byproduct is thought to be a genotoxin that increases DNA cleavage by acting on the enzyme topoisomerase II.
Asked for her perspective on the statement, Kjersti Aagaard, MD, PhD, a professor of obstetrics and gynecology at Baylor College of Medicine and Texas Children’s Hospital in Houston, called the expert panel’s statement thoughtful and comprehensive, but she urged caution in interpreting the role of acetaminophen.
The challenge in linking any commonly used medication to adverse effects and congenital defects, she said, is “teasing out an association from causation. Given the commonality of the use of acetaminophen with the relative rarity of the outcomes, it is clear that not all cases of exposure result in adverse outcomes.”
As for judicious use, she said, one would be to reduce a high fever, which can cause miscarriage, neural tube defects, and potential heart disease in adulthood. Acetaminophen is the drug of choice in this case since nonsteroidal anti-inflammatory drugs such as ibuprofen are not recommended owing to their known risks to the fetal heart.
Dr. Aagaard emphasized that while acetaminophen use is temporally associated with learning and behavioral problems, and urogenital disorders at birth in male infants such as like hypospadias, so is exposure to multiple environmental chemicals and pollutants, as well as climate change. “It would be a real mistake with real life implications if we associated any congenital disease or disorder with a commonly used medication with known benefits if the true causal link lies elsewhere.”
She said the precautionary statements fall into the time-honored therapeutic principle of first do no harm. “However, the call for research action must be undertaken earnestly and sincerely.”
According to Dr. Bauer, the statement’s essential take-home message is that “physicians should educate themselves and educate women about what we’re learning about the risks of acetaminophen in pregnancy.” Risk can be minimized by using the lowest effective dose for the shortest time and only when medically indicated. “Pregnant women should speak to their physicians about acetaminophen. It’s about empowerment and making smart decisions,” she said.
This study received no specific funding. Coauthor Dr. R.T. Mitchell is supported by a UK Research Institute fellowship.
Datopotamab deruxtecan for advanced NSCLC encouraging so far
according to Edward B. Garon, MD, of the University of California, Los Angeles. Prior results from TROPION-PanTumor01, have demonstrated similarly encouraging activity and a manageable safety profile for Dato-DXd, Dr. Garon said in a 2021 European Society for Medical Oncology Congress virtual oral presentation on Sept. 19 (abstract LBA49).
Limited benefit from existing treatments
Once tyrosine kinase inhibitors and platinum chemotherapy have failed, patients with advanced/metastatic NSCLC with AGAs (e.g., EGFR or ALK mutations) derive limited benefit from existing treatments, Dr. Garon observed. Datopotamab deruxtecan is an antibody-drug conjugate composed of a humanized anti-TROP2 monoclonal antibody conjugated to a potent topoisomerase I inhibitor payload via a stable tetrapeptide-based cleavable linker. TROP2 is highly expressed in NSCLC, regardless of genomic mutation status and has been associated with poor prognosis. Patients in TROPION-PanTumor01 were not selected based on TROP2 expression or AGA status, Dr. Garon noted.
TROPION-PanTumor01 (NCT03401385), an ongoing multicenter, open-label, dose-expansion study evaluating datopotamab deruxtecan in solid tumors, including NSCLC in 210 patients, is assessing safety, pharmacokinetics, antitumor activity, and biomarkers. All included patients (n = 180; median age, 62 years; 56% female) had progressed after standard treatment or had measurable disease and had no standard treatment available. Stable/treated brain metastases were permitted.
Subgroup with AGAs
The current report includes outcomes from the subgroup of 34 patients with AGAs, who were treated with 4 (n = 8), 6 (n = 10), and 8 mg/kg (n = 16) of datopotamab deruxtecan. AGAs were EGFR in 29 patients, ALK in 3, and ROS1 and RET in 1 each. Most patients (82%) had received three or more prior regimens; 85% had prior TKI, and among EGFR mutation patients, 69% had received osimertinib. Prior systemic treatment consisted of immunotherapy in 41%, platinum-based chemotherapy in 91%, and tyrosine kinase inhibitor in 85%. The primary objectives were to establish the maximum tolerated dose, safety, and tolerability. Efficacy was a secondary outcome.
Treatment-emergent adverse events were reported in all patients, with grade 3 or higher events in 53%. Most common were grade 1-2 nausea, stomatitis, fatigue, and alopecia. Drug-attributed events in 88% were grade 3 or higher in 38%. Treatment-emergent adverse events led to discontinuation in 15%, dose interruption in 27% and dose reductions in 15%. One case of grade 5 interstitial lung disease, in the 8-mg group, was adjudicated as drug related. “The safety profile of Dato-DXd was manageable and consistent with that observed in the overall NSCLC population in TROPION-PanTumor01,” Dr. Garon said, “and were primarily nonhematologic.”
The objective response rate was 35%, all partial responses. The stable disease rate was 41%; the progressive disease rate was 6%. Median duration of response was 9.5 months (95% confidence interval, 3.3-NE). Dr. Garon noted that clinical activity was observed in EGFR (Ex 19del, L858R) including after osimertinib and across other AGAs.
Further evaluation ongoing
Further evaluation of datopotamab deruxtecan is ongoing in the TROPION-Lung05 study among NSCLC patients with AGAs after targeted therapy and platinum-based chemotherapy options have been exhausted. Eligible AGAs include EGFR (including exon 20 insertions), ALK, ROS1, RET, BRAF, NTRK and MET exon 14 skipping.
Session moderator David Gandara, MD, University of California Davis Health, questioned the rationale for targeting oncogene driven cancers with this particular drug: “Is this just because this is felt to be an unmet need, or is there higher expression or some other biologic rationale?”
Dr. Garon responded, “Why are we looking at these driver mutation–positive patients? I think it has less to do with mechanism and more to do with the differences in treatment between these driver mutation positive patients and the rest of the population. This is a group of patients which has TROP2, but TROP2 expression is seen really across non–small cell lung cancer. But, in fact, one of the reasons it has been postulated that TROP2 is not a good biomarker for this class of drugs to date, is that its expression is so ubiquitous in the disease.”
The study was funded by Daiichi Sankyo. Dr. Garon disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
according to Edward B. Garon, MD, of the University of California, Los Angeles. Prior results from TROPION-PanTumor01, have demonstrated similarly encouraging activity and a manageable safety profile for Dato-DXd, Dr. Garon said in a 2021 European Society for Medical Oncology Congress virtual oral presentation on Sept. 19 (abstract LBA49).
Limited benefit from existing treatments
Once tyrosine kinase inhibitors and platinum chemotherapy have failed, patients with advanced/metastatic NSCLC with AGAs (e.g., EGFR or ALK mutations) derive limited benefit from existing treatments, Dr. Garon observed. Datopotamab deruxtecan is an antibody-drug conjugate composed of a humanized anti-TROP2 monoclonal antibody conjugated to a potent topoisomerase I inhibitor payload via a stable tetrapeptide-based cleavable linker. TROP2 is highly expressed in NSCLC, regardless of genomic mutation status and has been associated with poor prognosis. Patients in TROPION-PanTumor01 were not selected based on TROP2 expression or AGA status, Dr. Garon noted.
TROPION-PanTumor01 (NCT03401385), an ongoing multicenter, open-label, dose-expansion study evaluating datopotamab deruxtecan in solid tumors, including NSCLC in 210 patients, is assessing safety, pharmacokinetics, antitumor activity, and biomarkers. All included patients (n = 180; median age, 62 years; 56% female) had progressed after standard treatment or had measurable disease and had no standard treatment available. Stable/treated brain metastases were permitted.
Subgroup with AGAs
The current report includes outcomes from the subgroup of 34 patients with AGAs, who were treated with 4 (n = 8), 6 (n = 10), and 8 mg/kg (n = 16) of datopotamab deruxtecan. AGAs were EGFR in 29 patients, ALK in 3, and ROS1 and RET in 1 each. Most patients (82%) had received three or more prior regimens; 85% had prior TKI, and among EGFR mutation patients, 69% had received osimertinib. Prior systemic treatment consisted of immunotherapy in 41%, platinum-based chemotherapy in 91%, and tyrosine kinase inhibitor in 85%. The primary objectives were to establish the maximum tolerated dose, safety, and tolerability. Efficacy was a secondary outcome.
Treatment-emergent adverse events were reported in all patients, with grade 3 or higher events in 53%. Most common were grade 1-2 nausea, stomatitis, fatigue, and alopecia. Drug-attributed events in 88% were grade 3 or higher in 38%. Treatment-emergent adverse events led to discontinuation in 15%, dose interruption in 27% and dose reductions in 15%. One case of grade 5 interstitial lung disease, in the 8-mg group, was adjudicated as drug related. “The safety profile of Dato-DXd was manageable and consistent with that observed in the overall NSCLC population in TROPION-PanTumor01,” Dr. Garon said, “and were primarily nonhematologic.”
The objective response rate was 35%, all partial responses. The stable disease rate was 41%; the progressive disease rate was 6%. Median duration of response was 9.5 months (95% confidence interval, 3.3-NE). Dr. Garon noted that clinical activity was observed in EGFR (Ex 19del, L858R) including after osimertinib and across other AGAs.
Further evaluation ongoing
Further evaluation of datopotamab deruxtecan is ongoing in the TROPION-Lung05 study among NSCLC patients with AGAs after targeted therapy and platinum-based chemotherapy options have been exhausted. Eligible AGAs include EGFR (including exon 20 insertions), ALK, ROS1, RET, BRAF, NTRK and MET exon 14 skipping.
Session moderator David Gandara, MD, University of California Davis Health, questioned the rationale for targeting oncogene driven cancers with this particular drug: “Is this just because this is felt to be an unmet need, or is there higher expression or some other biologic rationale?”
Dr. Garon responded, “Why are we looking at these driver mutation–positive patients? I think it has less to do with mechanism and more to do with the differences in treatment between these driver mutation positive patients and the rest of the population. This is a group of patients which has TROP2, but TROP2 expression is seen really across non–small cell lung cancer. But, in fact, one of the reasons it has been postulated that TROP2 is not a good biomarker for this class of drugs to date, is that its expression is so ubiquitous in the disease.”
The study was funded by Daiichi Sankyo. Dr. Garon disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
according to Edward B. Garon, MD, of the University of California, Los Angeles. Prior results from TROPION-PanTumor01, have demonstrated similarly encouraging activity and a manageable safety profile for Dato-DXd, Dr. Garon said in a 2021 European Society for Medical Oncology Congress virtual oral presentation on Sept. 19 (abstract LBA49).
Limited benefit from existing treatments
Once tyrosine kinase inhibitors and platinum chemotherapy have failed, patients with advanced/metastatic NSCLC with AGAs (e.g., EGFR or ALK mutations) derive limited benefit from existing treatments, Dr. Garon observed. Datopotamab deruxtecan is an antibody-drug conjugate composed of a humanized anti-TROP2 monoclonal antibody conjugated to a potent topoisomerase I inhibitor payload via a stable tetrapeptide-based cleavable linker. TROP2 is highly expressed in NSCLC, regardless of genomic mutation status and has been associated with poor prognosis. Patients in TROPION-PanTumor01 were not selected based on TROP2 expression or AGA status, Dr. Garon noted.
TROPION-PanTumor01 (NCT03401385), an ongoing multicenter, open-label, dose-expansion study evaluating datopotamab deruxtecan in solid tumors, including NSCLC in 210 patients, is assessing safety, pharmacokinetics, antitumor activity, and biomarkers. All included patients (n = 180; median age, 62 years; 56% female) had progressed after standard treatment or had measurable disease and had no standard treatment available. Stable/treated brain metastases were permitted.
Subgroup with AGAs
The current report includes outcomes from the subgroup of 34 patients with AGAs, who were treated with 4 (n = 8), 6 (n = 10), and 8 mg/kg (n = 16) of datopotamab deruxtecan. AGAs were EGFR in 29 patients, ALK in 3, and ROS1 and RET in 1 each. Most patients (82%) had received three or more prior regimens; 85% had prior TKI, and among EGFR mutation patients, 69% had received osimertinib. Prior systemic treatment consisted of immunotherapy in 41%, platinum-based chemotherapy in 91%, and tyrosine kinase inhibitor in 85%. The primary objectives were to establish the maximum tolerated dose, safety, and tolerability. Efficacy was a secondary outcome.
Treatment-emergent adverse events were reported in all patients, with grade 3 or higher events in 53%. Most common were grade 1-2 nausea, stomatitis, fatigue, and alopecia. Drug-attributed events in 88% were grade 3 or higher in 38%. Treatment-emergent adverse events led to discontinuation in 15%, dose interruption in 27% and dose reductions in 15%. One case of grade 5 interstitial lung disease, in the 8-mg group, was adjudicated as drug related. “The safety profile of Dato-DXd was manageable and consistent with that observed in the overall NSCLC population in TROPION-PanTumor01,” Dr. Garon said, “and were primarily nonhematologic.”
The objective response rate was 35%, all partial responses. The stable disease rate was 41%; the progressive disease rate was 6%. Median duration of response was 9.5 months (95% confidence interval, 3.3-NE). Dr. Garon noted that clinical activity was observed in EGFR (Ex 19del, L858R) including after osimertinib and across other AGAs.
Further evaluation ongoing
Further evaluation of datopotamab deruxtecan is ongoing in the TROPION-Lung05 study among NSCLC patients with AGAs after targeted therapy and platinum-based chemotherapy options have been exhausted. Eligible AGAs include EGFR (including exon 20 insertions), ALK, ROS1, RET, BRAF, NTRK and MET exon 14 skipping.
Session moderator David Gandara, MD, University of California Davis Health, questioned the rationale for targeting oncogene driven cancers with this particular drug: “Is this just because this is felt to be an unmet need, or is there higher expression or some other biologic rationale?”
Dr. Garon responded, “Why are we looking at these driver mutation–positive patients? I think it has less to do with mechanism and more to do with the differences in treatment between these driver mutation positive patients and the rest of the population. This is a group of patients which has TROP2, but TROP2 expression is seen really across non–small cell lung cancer. But, in fact, one of the reasons it has been postulated that TROP2 is not a good biomarker for this class of drugs to date, is that its expression is so ubiquitous in the disease.”
The study was funded by Daiichi Sankyo. Dr. Garon disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
FROM ESMO 2021
TULIP trial shows extended survival in HER2+ metastatic breast cancer
, according to Cristina Saura Manich, MD, Hospital Universitario Valle de Hebrón, Barcelona. In TULIP, trastuzumab duocarmazine (SYD985, Byondis B.V., NL) was compared with physician’s choice of chemotherapy, Dr. Saura said at the virtual European Society for Medical Oncology Congress 2021 on Sept. 18 (abstract LBA15).
Trastuzumab duocarmazine, Dr. Manich noted, is a novel HER2-targeting antibody–drug conjugate based on trastuzumab and a cleavable linker-duocarmycin (vc-seco-DUBA) payload. Its three-way mechanism of action includes uptake of the antibody–drug conjugate by internalization and intracellular release of the payload, and two bystander effects: proteolytic cleavage and subsequent release of payload in the tumor microenvironment and diffusion of active payload to neighboring tumor cells.
Two or more prior therapies for metastatic breast cancer
TULIP investigators enrolled 437 patients from 83 sites in 11 countries with HER2-positive locally advanced or metastatic breast cancer who had received two or more therapies for metastatic disease (treatment for brain metastases allowed). They were randomized 2:1 to SYD985 (1.2 mg/kg IV every 21 days [n = 291]) or physician’s choice (PC) [n = 146] of one of three trastuzumab-containing combinations or lapatinib plus capecitabine. Treatment was continued until progression or unacceptable toxicity. The primary endpoint was centrally assessed PFS.
Longer progression-free survival with SYD985
Median age was 57 years, and the median number of prior metastatic breast cancer regimens was 4.7. Centrally reviewed progression-free survival was significantly longer in the SYD985 group at 7.0 months (5.4-7.2) versus 4.9 months (4.0-5.5) for PC (hazard ratio [HR], 0.64, 95% confidence interval [CI], 0.49-0.84, P = .002). Subgroup analysis, also centrally reviewed, revealed numerical advantage for SYD985 over physician choice across all categories (except for ECOG status 2). Analysis of progression-free survival by investigators showed a similar benefit for SYD985 (6.9 months versus 4.6 months, HR, 0.60, P < .001).
A first look at median overall survival showed a nonsignificant advantage for SYD985 (20.4 months versus 16.3 months (HR, 0.83, 95% CI, 0.62-1.09, P = .153). The overall response rate (partial or complete response) was similar between groups at 27.8% for SYD985 and 29.5% for PC, with reductions in target lesion measurement at 70.2% and 32.2% for SYD985 and physician choice, respectively. The clinical benefit rates were 38.5% for SYD985 and 32.2% for physician choice.
Ocular toxicity
Most patients had at least one treatment-related adverse event (96.5% SD985, 96.4% PC), and grade 3 or higher event rates were similar between groups (52.8% SYD985, 48.2% PC). The most frequently reported adverse events for SYD985 were ocular toxicity, with conjunctivitis reported in 38.2%, and keratitis in 38.2%, with fatigue at 33.3%; for physician’s choice these were diarrhea (35.8%), nausea (31.4%), and fatigue (29.9%). Interstitial lung disease pneumonitis was reported for 7.6% (5.2% grade 1-2) of patients treated with SYD985, including two grade 5 events. Eye toxicity led to discontinuations in 20.8% of SYD985 patients, dose modifications in 22.9%, with dose modifications for interstitial lung disease/pneumonitis in 5.2% of SYD985 patients. Six fatalities (2.1%) were reported in the SYD985 group, with four attributed to treatment. Assessment of health-related quality of life showed no significant difference between groups.
Dr. Manich outlined risk mitigation strategies. Patients with prior keratitis were excluded and patients were given prophylactic lubricating eye drops and regular eye exams by ophthalmologists. Treatment was discontinued if grade 3 or higher keratitis developed, and was delayed if grade 3 conjunctivitis developed until it reduced to grade 2. Also, patients with prior pneumonitis were excluded and CT lung scans were evaluated for lung changes. New or worsening respiratory symptoms triggered a full diagnostic workup. Treatment was discontinued for grade 2 or higher pneumonitis and delayed until resolution for grade 1 pneumonitis.
Another option
“It is encouraging to observe clinically meaningful and potentially practice changing PFS improvements in patients receiving treatment in the third line and beyond,” said Aditya Bardia, MD, of Massachusetts General Hospital and Harvard Medical School, Boston. “Several agents have been approved as treatments for HER2-positive metastatic breast cancer in recent years – including T-DXd, neratinib, tucatinib, and margetuximab – and [vic-]trastuzumab duocarmazine could eventually be another option.”
“At this time, there is only a minor 2-month difference in progression-free survival and a nonsignificant overall survival difference,” said Fatima Cardoso, MD, of Champalimaud Cancer Center, Lisbon, Portugal. “With the high incidence of ocular toxicity and four toxic deaths, we cannot recommend this drug for clinical practice, in my opinion.”
Dr. Manich concluded, “SYD985 can provide a new treatment option for patients with pretreated locally advanced or metastatic HER2-positive metastatic breast cancer.”
The study was funded by Byondis B.V. The authors disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
, according to Cristina Saura Manich, MD, Hospital Universitario Valle de Hebrón, Barcelona. In TULIP, trastuzumab duocarmazine (SYD985, Byondis B.V., NL) was compared with physician’s choice of chemotherapy, Dr. Saura said at the virtual European Society for Medical Oncology Congress 2021 on Sept. 18 (abstract LBA15).
Trastuzumab duocarmazine, Dr. Manich noted, is a novel HER2-targeting antibody–drug conjugate based on trastuzumab and a cleavable linker-duocarmycin (vc-seco-DUBA) payload. Its three-way mechanism of action includes uptake of the antibody–drug conjugate by internalization and intracellular release of the payload, and two bystander effects: proteolytic cleavage and subsequent release of payload in the tumor microenvironment and diffusion of active payload to neighboring tumor cells.
Two or more prior therapies for metastatic breast cancer
TULIP investigators enrolled 437 patients from 83 sites in 11 countries with HER2-positive locally advanced or metastatic breast cancer who had received two or more therapies for metastatic disease (treatment for brain metastases allowed). They were randomized 2:1 to SYD985 (1.2 mg/kg IV every 21 days [n = 291]) or physician’s choice (PC) [n = 146] of one of three trastuzumab-containing combinations or lapatinib plus capecitabine. Treatment was continued until progression or unacceptable toxicity. The primary endpoint was centrally assessed PFS.
Longer progression-free survival with SYD985
Median age was 57 years, and the median number of prior metastatic breast cancer regimens was 4.7. Centrally reviewed progression-free survival was significantly longer in the SYD985 group at 7.0 months (5.4-7.2) versus 4.9 months (4.0-5.5) for PC (hazard ratio [HR], 0.64, 95% confidence interval [CI], 0.49-0.84, P = .002). Subgroup analysis, also centrally reviewed, revealed numerical advantage for SYD985 over physician choice across all categories (except for ECOG status 2). Analysis of progression-free survival by investigators showed a similar benefit for SYD985 (6.9 months versus 4.6 months, HR, 0.60, P < .001).
A first look at median overall survival showed a nonsignificant advantage for SYD985 (20.4 months versus 16.3 months (HR, 0.83, 95% CI, 0.62-1.09, P = .153). The overall response rate (partial or complete response) was similar between groups at 27.8% for SYD985 and 29.5% for PC, with reductions in target lesion measurement at 70.2% and 32.2% for SYD985 and physician choice, respectively. The clinical benefit rates were 38.5% for SYD985 and 32.2% for physician choice.
Ocular toxicity
Most patients had at least one treatment-related adverse event (96.5% SD985, 96.4% PC), and grade 3 or higher event rates were similar between groups (52.8% SYD985, 48.2% PC). The most frequently reported adverse events for SYD985 were ocular toxicity, with conjunctivitis reported in 38.2%, and keratitis in 38.2%, with fatigue at 33.3%; for physician’s choice these were diarrhea (35.8%), nausea (31.4%), and fatigue (29.9%). Interstitial lung disease pneumonitis was reported for 7.6% (5.2% grade 1-2) of patients treated with SYD985, including two grade 5 events. Eye toxicity led to discontinuations in 20.8% of SYD985 patients, dose modifications in 22.9%, with dose modifications for interstitial lung disease/pneumonitis in 5.2% of SYD985 patients. Six fatalities (2.1%) were reported in the SYD985 group, with four attributed to treatment. Assessment of health-related quality of life showed no significant difference between groups.
Dr. Manich outlined risk mitigation strategies. Patients with prior keratitis were excluded and patients were given prophylactic lubricating eye drops and regular eye exams by ophthalmologists. Treatment was discontinued if grade 3 or higher keratitis developed, and was delayed if grade 3 conjunctivitis developed until it reduced to grade 2. Also, patients with prior pneumonitis were excluded and CT lung scans were evaluated for lung changes. New or worsening respiratory symptoms triggered a full diagnostic workup. Treatment was discontinued for grade 2 or higher pneumonitis and delayed until resolution for grade 1 pneumonitis.
Another option
“It is encouraging to observe clinically meaningful and potentially practice changing PFS improvements in patients receiving treatment in the third line and beyond,” said Aditya Bardia, MD, of Massachusetts General Hospital and Harvard Medical School, Boston. “Several agents have been approved as treatments for HER2-positive metastatic breast cancer in recent years – including T-DXd, neratinib, tucatinib, and margetuximab – and [vic-]trastuzumab duocarmazine could eventually be another option.”
“At this time, there is only a minor 2-month difference in progression-free survival and a nonsignificant overall survival difference,” said Fatima Cardoso, MD, of Champalimaud Cancer Center, Lisbon, Portugal. “With the high incidence of ocular toxicity and four toxic deaths, we cannot recommend this drug for clinical practice, in my opinion.”
Dr. Manich concluded, “SYD985 can provide a new treatment option for patients with pretreated locally advanced or metastatic HER2-positive metastatic breast cancer.”
The study was funded by Byondis B.V. The authors disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
, according to Cristina Saura Manich, MD, Hospital Universitario Valle de Hebrón, Barcelona. In TULIP, trastuzumab duocarmazine (SYD985, Byondis B.V., NL) was compared with physician’s choice of chemotherapy, Dr. Saura said at the virtual European Society for Medical Oncology Congress 2021 on Sept. 18 (abstract LBA15).
Trastuzumab duocarmazine, Dr. Manich noted, is a novel HER2-targeting antibody–drug conjugate based on trastuzumab and a cleavable linker-duocarmycin (vc-seco-DUBA) payload. Its three-way mechanism of action includes uptake of the antibody–drug conjugate by internalization and intracellular release of the payload, and two bystander effects: proteolytic cleavage and subsequent release of payload in the tumor microenvironment and diffusion of active payload to neighboring tumor cells.
Two or more prior therapies for metastatic breast cancer
TULIP investigators enrolled 437 patients from 83 sites in 11 countries with HER2-positive locally advanced or metastatic breast cancer who had received two or more therapies for metastatic disease (treatment for brain metastases allowed). They were randomized 2:1 to SYD985 (1.2 mg/kg IV every 21 days [n = 291]) or physician’s choice (PC) [n = 146] of one of three trastuzumab-containing combinations or lapatinib plus capecitabine. Treatment was continued until progression or unacceptable toxicity. The primary endpoint was centrally assessed PFS.
Longer progression-free survival with SYD985
Median age was 57 years, and the median number of prior metastatic breast cancer regimens was 4.7. Centrally reviewed progression-free survival was significantly longer in the SYD985 group at 7.0 months (5.4-7.2) versus 4.9 months (4.0-5.5) for PC (hazard ratio [HR], 0.64, 95% confidence interval [CI], 0.49-0.84, P = .002). Subgroup analysis, also centrally reviewed, revealed numerical advantage for SYD985 over physician choice across all categories (except for ECOG status 2). Analysis of progression-free survival by investigators showed a similar benefit for SYD985 (6.9 months versus 4.6 months, HR, 0.60, P < .001).
A first look at median overall survival showed a nonsignificant advantage for SYD985 (20.4 months versus 16.3 months (HR, 0.83, 95% CI, 0.62-1.09, P = .153). The overall response rate (partial or complete response) was similar between groups at 27.8% for SYD985 and 29.5% for PC, with reductions in target lesion measurement at 70.2% and 32.2% for SYD985 and physician choice, respectively. The clinical benefit rates were 38.5% for SYD985 and 32.2% for physician choice.
Ocular toxicity
Most patients had at least one treatment-related adverse event (96.5% SD985, 96.4% PC), and grade 3 or higher event rates were similar between groups (52.8% SYD985, 48.2% PC). The most frequently reported adverse events for SYD985 were ocular toxicity, with conjunctivitis reported in 38.2%, and keratitis in 38.2%, with fatigue at 33.3%; for physician’s choice these were diarrhea (35.8%), nausea (31.4%), and fatigue (29.9%). Interstitial lung disease pneumonitis was reported for 7.6% (5.2% grade 1-2) of patients treated with SYD985, including two grade 5 events. Eye toxicity led to discontinuations in 20.8% of SYD985 patients, dose modifications in 22.9%, with dose modifications for interstitial lung disease/pneumonitis in 5.2% of SYD985 patients. Six fatalities (2.1%) were reported in the SYD985 group, with four attributed to treatment. Assessment of health-related quality of life showed no significant difference between groups.
Dr. Manich outlined risk mitigation strategies. Patients with prior keratitis were excluded and patients were given prophylactic lubricating eye drops and regular eye exams by ophthalmologists. Treatment was discontinued if grade 3 or higher keratitis developed, and was delayed if grade 3 conjunctivitis developed until it reduced to grade 2. Also, patients with prior pneumonitis were excluded and CT lung scans were evaluated for lung changes. New or worsening respiratory symptoms triggered a full diagnostic workup. Treatment was discontinued for grade 2 or higher pneumonitis and delayed until resolution for grade 1 pneumonitis.
Another option
“It is encouraging to observe clinically meaningful and potentially practice changing PFS improvements in patients receiving treatment in the third line and beyond,” said Aditya Bardia, MD, of Massachusetts General Hospital and Harvard Medical School, Boston. “Several agents have been approved as treatments for HER2-positive metastatic breast cancer in recent years – including T-DXd, neratinib, tucatinib, and margetuximab – and [vic-]trastuzumab duocarmazine could eventually be another option.”
“At this time, there is only a minor 2-month difference in progression-free survival and a nonsignificant overall survival difference,” said Fatima Cardoso, MD, of Champalimaud Cancer Center, Lisbon, Portugal. “With the high incidence of ocular toxicity and four toxic deaths, we cannot recommend this drug for clinical practice, in my opinion.”
Dr. Manich concluded, “SYD985 can provide a new treatment option for patients with pretreated locally advanced or metastatic HER2-positive metastatic breast cancer.”
The study was funded by Byondis B.V. The authors disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
FROM ESMO 2021
ctDNA may be a better surrogate for survival than RECIST
according to Alexander Noor Shoushtari, MD, Memorial Sloan Kettering Cancer Center, New York.
Tebentafusp is the first therapy to demonstrate an overall survival (OS) benefit in uveal melanoma, Dr. Shoushtari noted in a 2021 European Society of Medical Oncology Congress virtual oral presentation Sept. 17 (abstract 17570). He noted further that, in prior research, OS was improved regardless of RECISTv1.1 best response, suggesting that better surrogate efficacy endpoints are needed.
Uveal melanoma is a rare melanoma type with low mutational burden, but frequent liver metastases. Benefit from immune checkpoint inhibitors is poor, and there is no established standard of care once the disease becomes metastatic. “Immune checkpoint inhibitors are not as good for treating this type of melanoma as they are for treating cutaneous disease, and traditionally preferred treatment is within clinical trials,” Dr. Shoushtari said. In frontline trials, 1-year survival has been in the 50% range. Tebentafusp is an investigational, first-in-class bispecific soluble T-cell receptor (TCR) therapeutic. It is designed to target gp100 (a melanoma-associated antigen) through a high-affinity TCR-binding domain and an anti-CD3 T-cell–engaging domain, which redirects T cells to kill gp100 positive melanocytic expressing tumor cells.
Prior research has demonstrated a disconnect between RECIST response classification and tebentafusp OS benefit. In the IMCgp100-202 study among patients treated first-line for metastatic uveal melanoma with tebentafusp or investigator choice, intent-to-treat analysis showed a survival probability benefit for tebentafusp (hazard ratio, 0.51; 95% CI, 0.37-0.71), with a best response of progressive disease population HR of 0.43 (95% CI, 0.27-0.68). While the RECIST response rate was only 9.1%, the HR for progression-free survival was 0.73 (95% CI, 0.58-0.94). “That suggests that RECIST is not a fantastic way to predict who will benefit from this drug,” Dr. Shoushtari stated.
Similarly in the IMCgp100-102 study of tebentafusp monotherapy in second-line metastatic uveal melanoma (n = 127), the RECIST response rate was 5%. Duration of response was 8.7 months and median OS was 16.8 months. Historical second-line OS has been reported at 7.8 months. The 1- and 2-year survival (62%/37%) compared favorably with historical rates (37%/15%), as well. Dr. Shoushtari noted that 92% of patients had detectable ctDNA with mutations in known uveal melanoma oncogenes. He pointed out that baseline ctDNA levels significantly correlated with tumor burden. Also, 70% of evaluable patients had any ctDNA reduction, with 0.5-3.2 log reduction in 99.9%, a 0.5 log reduction in 68% and 3 log reduction (cleared) in 14% of patients. ctDNA reduction, Dr. Shoushtari said, was associated with greater mean tumor shrinkage and with less tumor growth. Importantly, there was a linear correlation between ctDNA reduction and better OS (R2, 0.88, P < .0001).
Among all evaluable patients, comparing those with less than 0.5 log ctDNA reduction with those with at least a 0.5 log reduction showed a hazard ratio of 0.56 (95% CI, 0.32-0.95; P = .03). Among those whose best response was progressive disease, 35% had at least a 0.5 log reduction in ctDNA with an OS hazard ratio of 0.44 (95% CI, 0.2-0.94; P = .027), compared with less than a 0.5 log reduction. Among those whose best response was stable disease, 28% had at least 1 log reduction with a hazard ratio of 0.48 (95% CI 0.16-1.43; P = .16) for OS, compared with those with less than 1 log reduction. Dr. Shoushtari pointed out that “14% of patients cleared ctDNA, including some (n = 12) with best RECIST responses of stable or progressive disease. All patients with ctDNA clearance were alive beyond 1 year; with a hazard ratio, compared to those who had not cleared ctDNA, of 0.14 (95% CI, 0.03-0.57).
Summing up, Dr. Shoushtari said that ctDNA was detectable in more than 90% of second-line tebentafusp-treated patients with metastatic uveal melanoma and correlated with tumor burden. About 70% had ctDNA reduction versus 39% with tumor shrinkage and 5% RECIST response. The linear correlation between the magnitude of ctDNA reduction and improved OS on tebentafusp, was uncoupled from best RECIST response. “For tebentafusp, ctDNA reduction may be a better surrogate of overall survival than RECIST response.”
The ESMO-appointed discussant for the study, Christian Rolfo, MD, PhD, MBA, Icahan School of Medicine at Mount Sinai, New York, examined the tebentafusp study author’s claim that the radiographic assessment of tumors may underestimate the effect of tebentafusp, compared with ctDNA. The strengths of the study include, he said, that it is a drug- and tumor-specific evaluation of the role of ctDNA as a surrogate of response. “Its strength is that it shows an important correlation between ctDNA levels and overall survival, and that response rate is evaluated better with ctDNA.” A question that remains open, Dr. Rolfo added, is whether RECIST criteria are still a good comparator for biologic response.
The study was funded by Immunocore Dr. Shoushtari disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
according to Alexander Noor Shoushtari, MD, Memorial Sloan Kettering Cancer Center, New York.
Tebentafusp is the first therapy to demonstrate an overall survival (OS) benefit in uveal melanoma, Dr. Shoushtari noted in a 2021 European Society of Medical Oncology Congress virtual oral presentation Sept. 17 (abstract 17570). He noted further that, in prior research, OS was improved regardless of RECISTv1.1 best response, suggesting that better surrogate efficacy endpoints are needed.
Uveal melanoma is a rare melanoma type with low mutational burden, but frequent liver metastases. Benefit from immune checkpoint inhibitors is poor, and there is no established standard of care once the disease becomes metastatic. “Immune checkpoint inhibitors are not as good for treating this type of melanoma as they are for treating cutaneous disease, and traditionally preferred treatment is within clinical trials,” Dr. Shoushtari said. In frontline trials, 1-year survival has been in the 50% range. Tebentafusp is an investigational, first-in-class bispecific soluble T-cell receptor (TCR) therapeutic. It is designed to target gp100 (a melanoma-associated antigen) through a high-affinity TCR-binding domain and an anti-CD3 T-cell–engaging domain, which redirects T cells to kill gp100 positive melanocytic expressing tumor cells.
Prior research has demonstrated a disconnect between RECIST response classification and tebentafusp OS benefit. In the IMCgp100-202 study among patients treated first-line for metastatic uveal melanoma with tebentafusp or investigator choice, intent-to-treat analysis showed a survival probability benefit for tebentafusp (hazard ratio, 0.51; 95% CI, 0.37-0.71), with a best response of progressive disease population HR of 0.43 (95% CI, 0.27-0.68). While the RECIST response rate was only 9.1%, the HR for progression-free survival was 0.73 (95% CI, 0.58-0.94). “That suggests that RECIST is not a fantastic way to predict who will benefit from this drug,” Dr. Shoushtari stated.
Similarly in the IMCgp100-102 study of tebentafusp monotherapy in second-line metastatic uveal melanoma (n = 127), the RECIST response rate was 5%. Duration of response was 8.7 months and median OS was 16.8 months. Historical second-line OS has been reported at 7.8 months. The 1- and 2-year survival (62%/37%) compared favorably with historical rates (37%/15%), as well. Dr. Shoushtari noted that 92% of patients had detectable ctDNA with mutations in known uveal melanoma oncogenes. He pointed out that baseline ctDNA levels significantly correlated with tumor burden. Also, 70% of evaluable patients had any ctDNA reduction, with 0.5-3.2 log reduction in 99.9%, a 0.5 log reduction in 68% and 3 log reduction (cleared) in 14% of patients. ctDNA reduction, Dr. Shoushtari said, was associated with greater mean tumor shrinkage and with less tumor growth. Importantly, there was a linear correlation between ctDNA reduction and better OS (R2, 0.88, P < .0001).
Among all evaluable patients, comparing those with less than 0.5 log ctDNA reduction with those with at least a 0.5 log reduction showed a hazard ratio of 0.56 (95% CI, 0.32-0.95; P = .03). Among those whose best response was progressive disease, 35% had at least a 0.5 log reduction in ctDNA with an OS hazard ratio of 0.44 (95% CI, 0.2-0.94; P = .027), compared with less than a 0.5 log reduction. Among those whose best response was stable disease, 28% had at least 1 log reduction with a hazard ratio of 0.48 (95% CI 0.16-1.43; P = .16) for OS, compared with those with less than 1 log reduction. Dr. Shoushtari pointed out that “14% of patients cleared ctDNA, including some (n = 12) with best RECIST responses of stable or progressive disease. All patients with ctDNA clearance were alive beyond 1 year; with a hazard ratio, compared to those who had not cleared ctDNA, of 0.14 (95% CI, 0.03-0.57).
Summing up, Dr. Shoushtari said that ctDNA was detectable in more than 90% of second-line tebentafusp-treated patients with metastatic uveal melanoma and correlated with tumor burden. About 70% had ctDNA reduction versus 39% with tumor shrinkage and 5% RECIST response. The linear correlation between the magnitude of ctDNA reduction and improved OS on tebentafusp, was uncoupled from best RECIST response. “For tebentafusp, ctDNA reduction may be a better surrogate of overall survival than RECIST response.”
The ESMO-appointed discussant for the study, Christian Rolfo, MD, PhD, MBA, Icahan School of Medicine at Mount Sinai, New York, examined the tebentafusp study author’s claim that the radiographic assessment of tumors may underestimate the effect of tebentafusp, compared with ctDNA. The strengths of the study include, he said, that it is a drug- and tumor-specific evaluation of the role of ctDNA as a surrogate of response. “Its strength is that it shows an important correlation between ctDNA levels and overall survival, and that response rate is evaluated better with ctDNA.” A question that remains open, Dr. Rolfo added, is whether RECIST criteria are still a good comparator for biologic response.
The study was funded by Immunocore Dr. Shoushtari disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
according to Alexander Noor Shoushtari, MD, Memorial Sloan Kettering Cancer Center, New York.
Tebentafusp is the first therapy to demonstrate an overall survival (OS) benefit in uveal melanoma, Dr. Shoushtari noted in a 2021 European Society of Medical Oncology Congress virtual oral presentation Sept. 17 (abstract 17570). He noted further that, in prior research, OS was improved regardless of RECISTv1.1 best response, suggesting that better surrogate efficacy endpoints are needed.
Uveal melanoma is a rare melanoma type with low mutational burden, but frequent liver metastases. Benefit from immune checkpoint inhibitors is poor, and there is no established standard of care once the disease becomes metastatic. “Immune checkpoint inhibitors are not as good for treating this type of melanoma as they are for treating cutaneous disease, and traditionally preferred treatment is within clinical trials,” Dr. Shoushtari said. In frontline trials, 1-year survival has been in the 50% range. Tebentafusp is an investigational, first-in-class bispecific soluble T-cell receptor (TCR) therapeutic. It is designed to target gp100 (a melanoma-associated antigen) through a high-affinity TCR-binding domain and an anti-CD3 T-cell–engaging domain, which redirects T cells to kill gp100 positive melanocytic expressing tumor cells.
Prior research has demonstrated a disconnect between RECIST response classification and tebentafusp OS benefit. In the IMCgp100-202 study among patients treated first-line for metastatic uveal melanoma with tebentafusp or investigator choice, intent-to-treat analysis showed a survival probability benefit for tebentafusp (hazard ratio, 0.51; 95% CI, 0.37-0.71), with a best response of progressive disease population HR of 0.43 (95% CI, 0.27-0.68). While the RECIST response rate was only 9.1%, the HR for progression-free survival was 0.73 (95% CI, 0.58-0.94). “That suggests that RECIST is not a fantastic way to predict who will benefit from this drug,” Dr. Shoushtari stated.
Similarly in the IMCgp100-102 study of tebentafusp monotherapy in second-line metastatic uveal melanoma (n = 127), the RECIST response rate was 5%. Duration of response was 8.7 months and median OS was 16.8 months. Historical second-line OS has been reported at 7.8 months. The 1- and 2-year survival (62%/37%) compared favorably with historical rates (37%/15%), as well. Dr. Shoushtari noted that 92% of patients had detectable ctDNA with mutations in known uveal melanoma oncogenes. He pointed out that baseline ctDNA levels significantly correlated with tumor burden. Also, 70% of evaluable patients had any ctDNA reduction, with 0.5-3.2 log reduction in 99.9%, a 0.5 log reduction in 68% and 3 log reduction (cleared) in 14% of patients. ctDNA reduction, Dr. Shoushtari said, was associated with greater mean tumor shrinkage and with less tumor growth. Importantly, there was a linear correlation between ctDNA reduction and better OS (R2, 0.88, P < .0001).
Among all evaluable patients, comparing those with less than 0.5 log ctDNA reduction with those with at least a 0.5 log reduction showed a hazard ratio of 0.56 (95% CI, 0.32-0.95; P = .03). Among those whose best response was progressive disease, 35% had at least a 0.5 log reduction in ctDNA with an OS hazard ratio of 0.44 (95% CI, 0.2-0.94; P = .027), compared with less than a 0.5 log reduction. Among those whose best response was stable disease, 28% had at least 1 log reduction with a hazard ratio of 0.48 (95% CI 0.16-1.43; P = .16) for OS, compared with those with less than 1 log reduction. Dr. Shoushtari pointed out that “14% of patients cleared ctDNA, including some (n = 12) with best RECIST responses of stable or progressive disease. All patients with ctDNA clearance were alive beyond 1 year; with a hazard ratio, compared to those who had not cleared ctDNA, of 0.14 (95% CI, 0.03-0.57).
Summing up, Dr. Shoushtari said that ctDNA was detectable in more than 90% of second-line tebentafusp-treated patients with metastatic uveal melanoma and correlated with tumor burden. About 70% had ctDNA reduction versus 39% with tumor shrinkage and 5% RECIST response. The linear correlation between the magnitude of ctDNA reduction and improved OS on tebentafusp, was uncoupled from best RECIST response. “For tebentafusp, ctDNA reduction may be a better surrogate of overall survival than RECIST response.”
The ESMO-appointed discussant for the study, Christian Rolfo, MD, PhD, MBA, Icahan School of Medicine at Mount Sinai, New York, examined the tebentafusp study author’s claim that the radiographic assessment of tumors may underestimate the effect of tebentafusp, compared with ctDNA. The strengths of the study include, he said, that it is a drug- and tumor-specific evaluation of the role of ctDNA as a surrogate of response. “Its strength is that it shows an important correlation between ctDNA levels and overall survival, and that response rate is evaluated better with ctDNA.” A question that remains open, Dr. Rolfo added, is whether RECIST criteria are still a good comparator for biologic response.
The study was funded by Immunocore Dr. Shoushtari disclosed numerous pharmaceutical-related financial interests.
This article was updated Sept. 24, 2021.
FROM ESMO 2021
Your bathroom towel rack has a dirty little secret
Bacteria get the rack ... the towel rack
Obviously, bathrooms have germs. Some people are cleaner about their bathrooms than others, but in general most people just try not to think about the microscopic critters crawling about.
Now you would probably think that the toilet is the dirtiest part of the bathroom because that’s where ... you know, most of the business takes place. Or maybe you’d guess the floor. Truth be told, though, the dirtiest part of the bathroom is where the towels are hung.
According to research conducted by electric heating company Rointe in the United Kingdom, bathroom radiators and towel racks/bars are the most germy and dirty parts of the bathroom.
Company investigators examined five bathrooms using swabs that changed color on contact with bacteria and found that 60% of towel racks and radiators were “really dirty,” compared with 50% of sink drains and just 10% of toilets.
Most people probably pay more attention to the sink, floors, and toilets while cleaning, the company suggested, and dampness is a factor in bacteria growth, so it’s no surprise that towels that stay wet on a rack are prime spots for dust, mildew, and mold.
The toilet may be busier, but you don’t put your face in it.
Anti-vaxxers would like to be called ‘purebloods’
COVID-19 anti-vaxxers are an interesting bunch, to be kind. And TikTok is a wacky place. So you can just imagine that anti-vaxxer TikTok is a very strange place. The citizens of anti-vax TikTok have decided that the real reason so many people dislike them is branding. They consider anti-vaccination to be a negative word (duh), so they now want to be referred to as “purebloods.”
Harry Potter doesn’t quite occupy the zeitgeist as it once did, so let’s give you a reminder: In the books, purebloods came from old wizarding families and claimed not to have any Muggle, or nonmagic, blood. While having pure wizard blood was no guarantee of being a villain, most of them were. In addition, it is made quite clear throughout the novels that having supposedly pure blood had no relevance on one’s wizarding ability. Pureblood was a meaningless title, and only the characters with small, cruel minds concerned themselves over it.
Perhaps the anti-vaxxers have decided that they want to be called the same thing. Maybe they just like the name. It does sound impressive and vaguely regal: Pureblood. Like something the nobles of medieval Europe might have used.
Critical-thinking skills may be in short supply here, or maybe the anti-vaxxers know exactly what they’re doing.
Hated broccoli? Blame your DNA
Were you that kid who would rather sit at the table for hours than eat your broccoli? Well, as much as your parents might have pushed you, new research suggests that it might be their fault you didn’t like it to begin with.
Investigators at Australia’s national science agency, CSIRO, recently reported that distaste for Brassica vegetables – broccoli, Brussels sprouts, cabbage, and cauliflower – can be traced to the oral microbiome.
These vegetables have a compound called S-methyl-L-cysteine sulfoxide that gives off sulfurous odors ... mmm, sulfurous ... when mixed with an enzyme in the plant, and that enzyme is also produced by bacteria in some people’s oral microbiomes. So why do adults tolerate these Brassica veggies more than children? It’s all about levels.
The researchers tested the idea by asking 98 child/parent pairs to rate the odors and by using gas chromatography-olfactometry-mass spectrometry to identify the odor-active compounds in both raw and steamed cauliflower and broccoli. The children whose saliva produced high levels of sulfur volatiles disliked Brassica vegetables the most, they reported, and the children with high levels of sulfur volatiles usually had parents who produced high levels.
Despite that connection, however, the distaste for raw Brassica seen in children wasn’t seen in adults.
Maybe it’s not that taste buds change as we age, maybe we just learn to tolerate the sulfurousness.
Bacteria get the rack ... the towel rack
Obviously, bathrooms have germs. Some people are cleaner about their bathrooms than others, but in general most people just try not to think about the microscopic critters crawling about.
Now you would probably think that the toilet is the dirtiest part of the bathroom because that’s where ... you know, most of the business takes place. Or maybe you’d guess the floor. Truth be told, though, the dirtiest part of the bathroom is where the towels are hung.
According to research conducted by electric heating company Rointe in the United Kingdom, bathroom radiators and towel racks/bars are the most germy and dirty parts of the bathroom.
Company investigators examined five bathrooms using swabs that changed color on contact with bacteria and found that 60% of towel racks and radiators were “really dirty,” compared with 50% of sink drains and just 10% of toilets.
Most people probably pay more attention to the sink, floors, and toilets while cleaning, the company suggested, and dampness is a factor in bacteria growth, so it’s no surprise that towels that stay wet on a rack are prime spots for dust, mildew, and mold.
The toilet may be busier, but you don’t put your face in it.
Anti-vaxxers would like to be called ‘purebloods’
COVID-19 anti-vaxxers are an interesting bunch, to be kind. And TikTok is a wacky place. So you can just imagine that anti-vaxxer TikTok is a very strange place. The citizens of anti-vax TikTok have decided that the real reason so many people dislike them is branding. They consider anti-vaccination to be a negative word (duh), so they now want to be referred to as “purebloods.”
Harry Potter doesn’t quite occupy the zeitgeist as it once did, so let’s give you a reminder: In the books, purebloods came from old wizarding families and claimed not to have any Muggle, or nonmagic, blood. While having pure wizard blood was no guarantee of being a villain, most of them were. In addition, it is made quite clear throughout the novels that having supposedly pure blood had no relevance on one’s wizarding ability. Pureblood was a meaningless title, and only the characters with small, cruel minds concerned themselves over it.
Perhaps the anti-vaxxers have decided that they want to be called the same thing. Maybe they just like the name. It does sound impressive and vaguely regal: Pureblood. Like something the nobles of medieval Europe might have used.
Critical-thinking skills may be in short supply here, or maybe the anti-vaxxers know exactly what they’re doing.
Hated broccoli? Blame your DNA
Were you that kid who would rather sit at the table for hours than eat your broccoli? Well, as much as your parents might have pushed you, new research suggests that it might be their fault you didn’t like it to begin with.
Investigators at Australia’s national science agency, CSIRO, recently reported that distaste for Brassica vegetables – broccoli, Brussels sprouts, cabbage, and cauliflower – can be traced to the oral microbiome.
These vegetables have a compound called S-methyl-L-cysteine sulfoxide that gives off sulfurous odors ... mmm, sulfurous ... when mixed with an enzyme in the plant, and that enzyme is also produced by bacteria in some people’s oral microbiomes. So why do adults tolerate these Brassica veggies more than children? It’s all about levels.
The researchers tested the idea by asking 98 child/parent pairs to rate the odors and by using gas chromatography-olfactometry-mass spectrometry to identify the odor-active compounds in both raw and steamed cauliflower and broccoli. The children whose saliva produced high levels of sulfur volatiles disliked Brassica vegetables the most, they reported, and the children with high levels of sulfur volatiles usually had parents who produced high levels.
Despite that connection, however, the distaste for raw Brassica seen in children wasn’t seen in adults.
Maybe it’s not that taste buds change as we age, maybe we just learn to tolerate the sulfurousness.
Bacteria get the rack ... the towel rack
Obviously, bathrooms have germs. Some people are cleaner about their bathrooms than others, but in general most people just try not to think about the microscopic critters crawling about.
Now you would probably think that the toilet is the dirtiest part of the bathroom because that’s where ... you know, most of the business takes place. Or maybe you’d guess the floor. Truth be told, though, the dirtiest part of the bathroom is where the towels are hung.
According to research conducted by electric heating company Rointe in the United Kingdom, bathroom radiators and towel racks/bars are the most germy and dirty parts of the bathroom.
Company investigators examined five bathrooms using swabs that changed color on contact with bacteria and found that 60% of towel racks and radiators were “really dirty,” compared with 50% of sink drains and just 10% of toilets.
Most people probably pay more attention to the sink, floors, and toilets while cleaning, the company suggested, and dampness is a factor in bacteria growth, so it’s no surprise that towels that stay wet on a rack are prime spots for dust, mildew, and mold.
The toilet may be busier, but you don’t put your face in it.
Anti-vaxxers would like to be called ‘purebloods’
COVID-19 anti-vaxxers are an interesting bunch, to be kind. And TikTok is a wacky place. So you can just imagine that anti-vaxxer TikTok is a very strange place. The citizens of anti-vax TikTok have decided that the real reason so many people dislike them is branding. They consider anti-vaccination to be a negative word (duh), so they now want to be referred to as “purebloods.”
Harry Potter doesn’t quite occupy the zeitgeist as it once did, so let’s give you a reminder: In the books, purebloods came from old wizarding families and claimed not to have any Muggle, or nonmagic, blood. While having pure wizard blood was no guarantee of being a villain, most of them were. In addition, it is made quite clear throughout the novels that having supposedly pure blood had no relevance on one’s wizarding ability. Pureblood was a meaningless title, and only the characters with small, cruel minds concerned themselves over it.
Perhaps the anti-vaxxers have decided that they want to be called the same thing. Maybe they just like the name. It does sound impressive and vaguely regal: Pureblood. Like something the nobles of medieval Europe might have used.
Critical-thinking skills may be in short supply here, or maybe the anti-vaxxers know exactly what they’re doing.
Hated broccoli? Blame your DNA
Were you that kid who would rather sit at the table for hours than eat your broccoli? Well, as much as your parents might have pushed you, new research suggests that it might be their fault you didn’t like it to begin with.
Investigators at Australia’s national science agency, CSIRO, recently reported that distaste for Brassica vegetables – broccoli, Brussels sprouts, cabbage, and cauliflower – can be traced to the oral microbiome.
These vegetables have a compound called S-methyl-L-cysteine sulfoxide that gives off sulfurous odors ... mmm, sulfurous ... when mixed with an enzyme in the plant, and that enzyme is also produced by bacteria in some people’s oral microbiomes. So why do adults tolerate these Brassica veggies more than children? It’s all about levels.
The researchers tested the idea by asking 98 child/parent pairs to rate the odors and by using gas chromatography-olfactometry-mass spectrometry to identify the odor-active compounds in both raw and steamed cauliflower and broccoli. The children whose saliva produced high levels of sulfur volatiles disliked Brassica vegetables the most, they reported, and the children with high levels of sulfur volatiles usually had parents who produced high levels.
Despite that connection, however, the distaste for raw Brassica seen in children wasn’t seen in adults.
Maybe it’s not that taste buds change as we age, maybe we just learn to tolerate the sulfurousness.
Methylation patterns correlate with prognosis in children with T-ALL
In childhood patients with T-cell acute lymphoblastic leukemia (T-ALL) the hypomethylation status of the arginine synthetase (ASNS) gene was significantly associated with poor therapeutic outcome, according to the results of a Japanese cohort study published online in Blood Advances.
Researchers Koshi Akahane, PHD, of the University of Yamanashi, Kofu, Japan, and colleagues conducted a comprehensive genetic analysis of diagnostic samples of 22 cell lines from childhood patients with T-ALL in Japan.
They also correlated known methylation status with outcomes in two large patient cohorts of Japanese children with T-ALL: the Tokyo Children’s Cancer Study Group (n = 57) and the Japan Association of Childhood Leukemia Study Group (n = 20).
Methylation results
For the 22 cell lines tested, sequencing technology revealed a stepwise allele-specific methylation of the ASNS gene. Mean ASNS gene expression level was significantly upregulated in 14 weakly methylated cell lines (P = .0001), but not significantly upregulated in 3 intermediately methylated cell lines (P = .25) or in 5 highly methylated cell lines (P = .063).
Among the 77 patient cohorts, 20 (26%) samples showed high methylation (> 66.7%), while 15 (19%) samples and 42 (55%) samples showed intermediate (33.3%-66.7%) and weak (< 33.3%) methylation status, respectively.
For the 75 patients where information was provided on outcomes, 25 patients (33%) showed induction failure or disease relapse. A highly methylated status of the ASNS gene was significantly more common in non-refractory/relapse cases (18/50 cases, 36%), while intermediately or weakly methylated status was more common in refractory/relapsed cases (23/25 cases, 92 %; P = .0001). In a log-rank test, the patients with weakly methylated status of the ASNS gene showed significantly shorter event-free survival and overall survival than the patients with an intermediate or highly methylated status (P = .00012 and P = .00016, respectively).
Asparaginase sensitivity
Asparaginase treatment is a key component of chemotherapy for patients with T-ALL, according to the researchers. Asparaginase depletes serum asparagine by deamination into aspartic acid. While normal hematopoietic cells can survive due to ASNS activity, leukemia cells are expected to undergo apoptosis due to silencing of the ASNS gene.
ASNS hypomethylation status was significantly associated with poor therapeutic outcome, and all cases with the poor prognostic SPI1 fusion exclusively showed ASNS hypomethylation status. These observations demonstrate that ASNS hypomethylation status is associated with asparaginase resistance, the researchers stated.
“ASNS methylation status may be a clinically useful biomarker to predict sensitivity to asparaginase therapy in T-ALL patients. Considering the severe complications of asparaginase therapy particularly in adolescents and adults, stratifying prospective asparaginase therapy according to ASNS methylation status may be beneficial for safer and more effective treatment of T-ALL patients,” the researchers concluded.
The authors reported that they had no conflicts of interest.
In childhood patients with T-cell acute lymphoblastic leukemia (T-ALL) the hypomethylation status of the arginine synthetase (ASNS) gene was significantly associated with poor therapeutic outcome, according to the results of a Japanese cohort study published online in Blood Advances.
Researchers Koshi Akahane, PHD, of the University of Yamanashi, Kofu, Japan, and colleagues conducted a comprehensive genetic analysis of diagnostic samples of 22 cell lines from childhood patients with T-ALL in Japan.
They also correlated known methylation status with outcomes in two large patient cohorts of Japanese children with T-ALL: the Tokyo Children’s Cancer Study Group (n = 57) and the Japan Association of Childhood Leukemia Study Group (n = 20).
Methylation results
For the 22 cell lines tested, sequencing technology revealed a stepwise allele-specific methylation of the ASNS gene. Mean ASNS gene expression level was significantly upregulated in 14 weakly methylated cell lines (P = .0001), but not significantly upregulated in 3 intermediately methylated cell lines (P = .25) or in 5 highly methylated cell lines (P = .063).
Among the 77 patient cohorts, 20 (26%) samples showed high methylation (> 66.7%), while 15 (19%) samples and 42 (55%) samples showed intermediate (33.3%-66.7%) and weak (< 33.3%) methylation status, respectively.
For the 75 patients where information was provided on outcomes, 25 patients (33%) showed induction failure or disease relapse. A highly methylated status of the ASNS gene was significantly more common in non-refractory/relapse cases (18/50 cases, 36%), while intermediately or weakly methylated status was more common in refractory/relapsed cases (23/25 cases, 92 %; P = .0001). In a log-rank test, the patients with weakly methylated status of the ASNS gene showed significantly shorter event-free survival and overall survival than the patients with an intermediate or highly methylated status (P = .00012 and P = .00016, respectively).
Asparaginase sensitivity
Asparaginase treatment is a key component of chemotherapy for patients with T-ALL, according to the researchers. Asparaginase depletes serum asparagine by deamination into aspartic acid. While normal hematopoietic cells can survive due to ASNS activity, leukemia cells are expected to undergo apoptosis due to silencing of the ASNS gene.
ASNS hypomethylation status was significantly associated with poor therapeutic outcome, and all cases with the poor prognostic SPI1 fusion exclusively showed ASNS hypomethylation status. These observations demonstrate that ASNS hypomethylation status is associated with asparaginase resistance, the researchers stated.
“ASNS methylation status may be a clinically useful biomarker to predict sensitivity to asparaginase therapy in T-ALL patients. Considering the severe complications of asparaginase therapy particularly in adolescents and adults, stratifying prospective asparaginase therapy according to ASNS methylation status may be beneficial for safer and more effective treatment of T-ALL patients,” the researchers concluded.
The authors reported that they had no conflicts of interest.
In childhood patients with T-cell acute lymphoblastic leukemia (T-ALL) the hypomethylation status of the arginine synthetase (ASNS) gene was significantly associated with poor therapeutic outcome, according to the results of a Japanese cohort study published online in Blood Advances.
Researchers Koshi Akahane, PHD, of the University of Yamanashi, Kofu, Japan, and colleagues conducted a comprehensive genetic analysis of diagnostic samples of 22 cell lines from childhood patients with T-ALL in Japan.
They also correlated known methylation status with outcomes in two large patient cohorts of Japanese children with T-ALL: the Tokyo Children’s Cancer Study Group (n = 57) and the Japan Association of Childhood Leukemia Study Group (n = 20).
Methylation results
For the 22 cell lines tested, sequencing technology revealed a stepwise allele-specific methylation of the ASNS gene. Mean ASNS gene expression level was significantly upregulated in 14 weakly methylated cell lines (P = .0001), but not significantly upregulated in 3 intermediately methylated cell lines (P = .25) or in 5 highly methylated cell lines (P = .063).
Among the 77 patient cohorts, 20 (26%) samples showed high methylation (> 66.7%), while 15 (19%) samples and 42 (55%) samples showed intermediate (33.3%-66.7%) and weak (< 33.3%) methylation status, respectively.
For the 75 patients where information was provided on outcomes, 25 patients (33%) showed induction failure or disease relapse. A highly methylated status of the ASNS gene was significantly more common in non-refractory/relapse cases (18/50 cases, 36%), while intermediately or weakly methylated status was more common in refractory/relapsed cases (23/25 cases, 92 %; P = .0001). In a log-rank test, the patients with weakly methylated status of the ASNS gene showed significantly shorter event-free survival and overall survival than the patients with an intermediate or highly methylated status (P = .00012 and P = .00016, respectively).
Asparaginase sensitivity
Asparaginase treatment is a key component of chemotherapy for patients with T-ALL, according to the researchers. Asparaginase depletes serum asparagine by deamination into aspartic acid. While normal hematopoietic cells can survive due to ASNS activity, leukemia cells are expected to undergo apoptosis due to silencing of the ASNS gene.
ASNS hypomethylation status was significantly associated with poor therapeutic outcome, and all cases with the poor prognostic SPI1 fusion exclusively showed ASNS hypomethylation status. These observations demonstrate that ASNS hypomethylation status is associated with asparaginase resistance, the researchers stated.
“ASNS methylation status may be a clinically useful biomarker to predict sensitivity to asparaginase therapy in T-ALL patients. Considering the severe complications of asparaginase therapy particularly in adolescents and adults, stratifying prospective asparaginase therapy according to ASNS methylation status may be beneficial for safer and more effective treatment of T-ALL patients,” the researchers concluded.
The authors reported that they had no conflicts of interest.
FROM BLOOD ADVANCES












