User login
Surprising brain activity moments before death
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
All the participants in the study I am going to tell you about this week died. And three of them died twice. But their deaths provide us with a fascinating window into the complex electrochemistry of the dying brain. What we might be looking at, indeed, is the physiologic correlate of the near-death experience.
The concept of the near-death experience is culturally ubiquitous. And though the content seems to track along culture lines – Western Christians are more likely to report seeing guardian angels, while Hindus are more likely to report seeing messengers of the god of death – certain factors seem to transcend culture: an out-of-body experience; a feeling of peace; and, of course, the light at the end of the tunnel.
As a materialist, I won’t discuss the possibility that these commonalities reflect some metaphysical structure to the afterlife. More likely, it seems to me, is that the commonalities result from the fact that the experience is mediated by our brains, and our brains, when dying, may be more alike than different.
We are talking about this study, appearing in the Proceedings of the National Academy of Sciences, by Jimo Borjigin and her team.
Dr. Borjigin studies the neural correlates of consciousness, perhaps one of the biggest questions in all of science today. To wit,
The study in question follows four unconscious patients –comatose patients, really – as life-sustaining support was withdrawn, up until the moment of death. Three had suffered severe anoxic brain injury in the setting of prolonged cardiac arrest. Though the heart was restarted, the brain damage was severe. The fourth had a large brain hemorrhage. All four patients were thus comatose and, though not brain-dead, unresponsive – with the lowest possible Glasgow Coma Scale score. No response to outside stimuli.
The families had made the decision to withdraw life support – to remove the breathing tube – but agreed to enroll their loved one in the study.
The team applied EEG leads to the head, EKG leads to the chest, and other monitoring equipment to observe the physiologic changes that occurred as the comatose and unresponsive patient died.
As the heart rhythm evolved from this: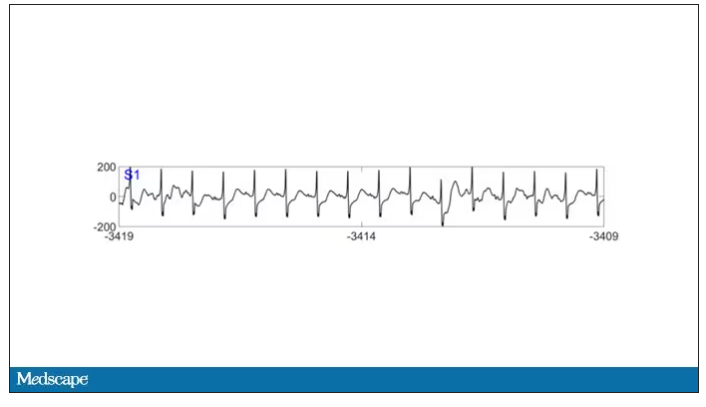
To this: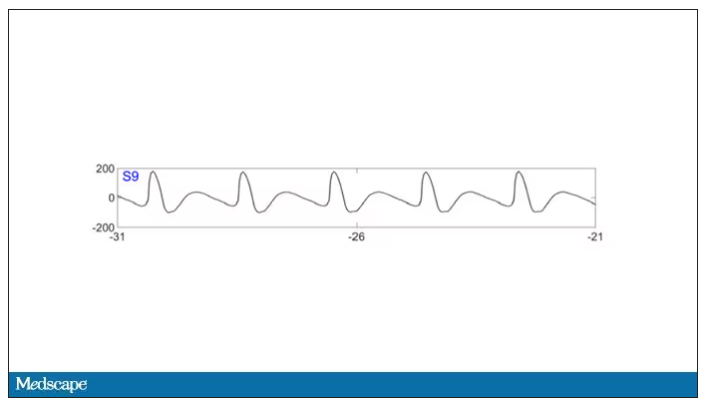
And eventually stopped.
But this is a study about the brain, not the heart.
Prior to the withdrawal of life support, the brain electrical signals looked like this: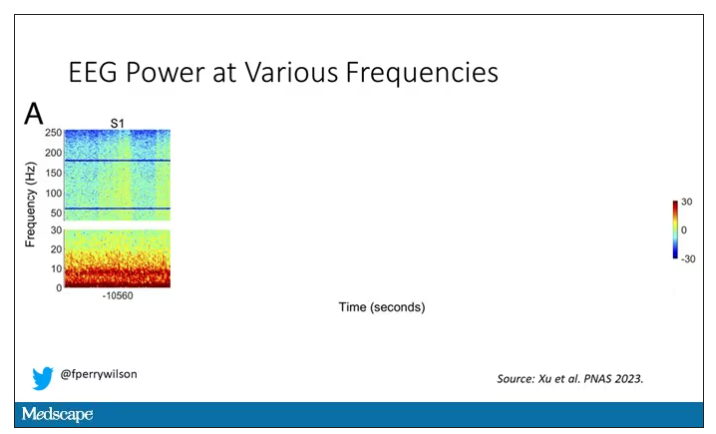
What you see is the EEG power at various frequencies, with red being higher. All the red was down at the low frequencies. Consciousness, at least as we understand it, is a higher-frequency phenomenon.
Right after the breathing tube was removed, the power didn’t change too much, but you can see some increased activity at the higher frequencies.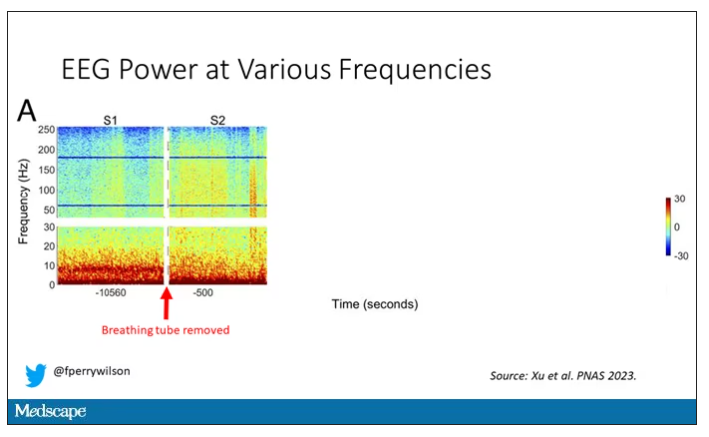
But in two of the four patients, something really surprising happened. Watch what happens as the brain gets closer and closer to death.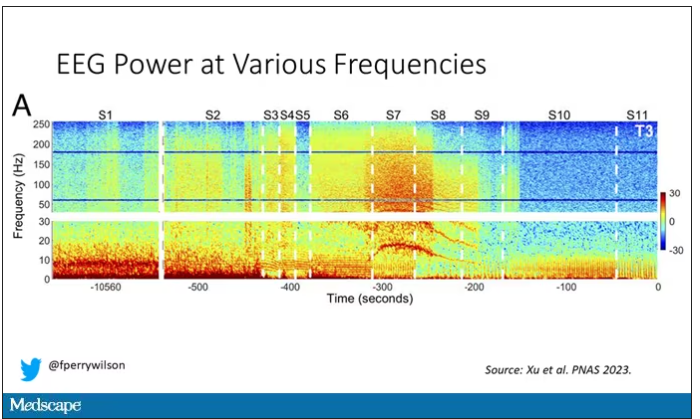
Here, about 300 seconds before death, there was a power surge at the high gamma frequencies.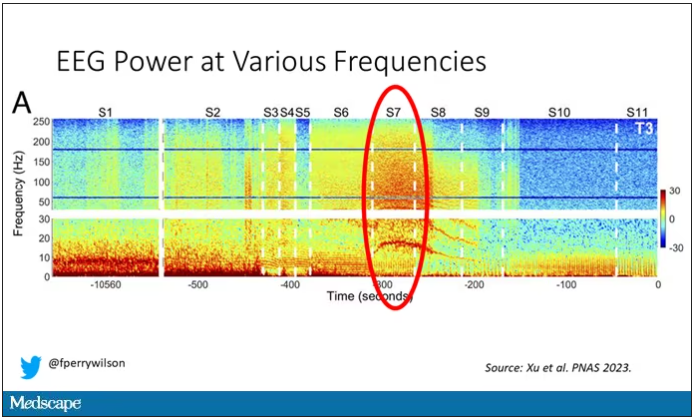
This spike in power occurred in the somatosensory cortex and the dorsolateral prefrontal cortex, areas that are associated with conscious experience. It seems that this patient, 5 minutes before death, was experiencing something.
But I know what you’re thinking. This is a brain that is not receiving oxygen. Cells are going to become disordered quickly and start firing randomly – a last gasp, so to speak, before the end. Meaningless noise.
But connectivity mapping tells a different story. The signals seem to have structure.
Those high-frequency power surges increased connectivity in the posterior cortical “hot zone,” an area of the brain many researchers feel is necessary for conscious perception. This figure is not a map of raw brain electrical output like the one I showed before, but of coherence between brain regions in the consciousness hot zone. Those red areas indicate cross-talk – not the disordered scream of dying neurons, but a last set of messages passing back and forth from the parietal and posterior temporal lobes.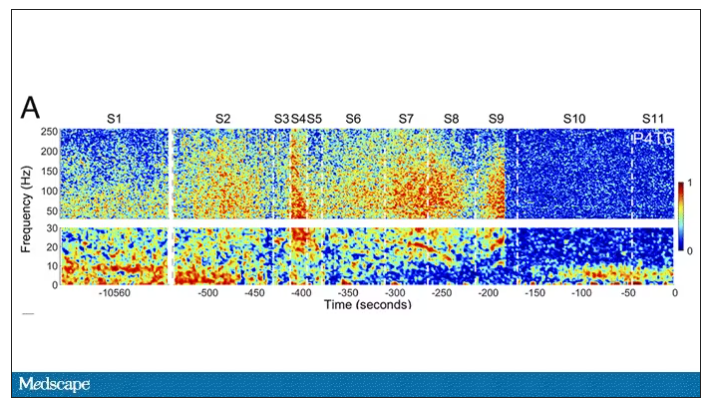
In fact, the electrical patterns of the brains in these patients looked very similar to the patterns seen in dreaming humans, as well as in patients with epilepsy who report sensations of out-of-body experiences.
It’s critical to realize two things here. First, these signals of consciousness were not present before life support was withdrawn. These comatose patients had minimal brain activity; there was no evidence that they were experiencing anything before the process of dying began. These brains are behaving fundamentally differently near death.
But second, we must realize that, although the brains of these individuals, in their last moments, appeared to be acting in a way that conscious brains act, we have no way of knowing if the patients were truly having a conscious experience. As I said, all the patients in the study died. Short of those metaphysics I alluded to earlier, we will have no way to ask them how they experienced their final moments.
Let’s be clear: This study doesn’t answer the question of what happens when we die. It says nothing about life after death or the existence or persistence of the soul. But what it does do is shed light on an incredibly difficult problem in neuroscience: the problem of consciousness. And as studies like this move forward, we may discover that the root of consciousness comes not from the breath of God or the energy of a living universe, but from very specific parts of the very complicated machine that is the brain, acting together to produce something transcendent. And to me, that is no less sublime.
Dr. Wilson is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator, Yale University, New Haven, Conn. His science communication work can be found in the Huffington Post, on NPR, and on Medscape. He tweets @fperrywilson and his new book, How Medicine Works and When It Doesn’t, is available now. Dr. Wilson has disclosed no relevant financial relationships.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
All the participants in the study I am going to tell you about this week died. And three of them died twice. But their deaths provide us with a fascinating window into the complex electrochemistry of the dying brain. What we might be looking at, indeed, is the physiologic correlate of the near-death experience.
The concept of the near-death experience is culturally ubiquitous. And though the content seems to track along culture lines – Western Christians are more likely to report seeing guardian angels, while Hindus are more likely to report seeing messengers of the god of death – certain factors seem to transcend culture: an out-of-body experience; a feeling of peace; and, of course, the light at the end of the tunnel.
As a materialist, I won’t discuss the possibility that these commonalities reflect some metaphysical structure to the afterlife. More likely, it seems to me, is that the commonalities result from the fact that the experience is mediated by our brains, and our brains, when dying, may be more alike than different.
We are talking about this study, appearing in the Proceedings of the National Academy of Sciences, by Jimo Borjigin and her team.
Dr. Borjigin studies the neural correlates of consciousness, perhaps one of the biggest questions in all of science today. To wit,
The study in question follows four unconscious patients –comatose patients, really – as life-sustaining support was withdrawn, up until the moment of death. Three had suffered severe anoxic brain injury in the setting of prolonged cardiac arrest. Though the heart was restarted, the brain damage was severe. The fourth had a large brain hemorrhage. All four patients were thus comatose and, though not brain-dead, unresponsive – with the lowest possible Glasgow Coma Scale score. No response to outside stimuli.
The families had made the decision to withdraw life support – to remove the breathing tube – but agreed to enroll their loved one in the study.
The team applied EEG leads to the head, EKG leads to the chest, and other monitoring equipment to observe the physiologic changes that occurred as the comatose and unresponsive patient died.
As the heart rhythm evolved from this:
To this:
And eventually stopped.
But this is a study about the brain, not the heart.
Prior to the withdrawal of life support, the brain electrical signals looked like this:
What you see is the EEG power at various frequencies, with red being higher. All the red was down at the low frequencies. Consciousness, at least as we understand it, is a higher-frequency phenomenon.
Right after the breathing tube was removed, the power didn’t change too much, but you can see some increased activity at the higher frequencies.
But in two of the four patients, something really surprising happened. Watch what happens as the brain gets closer and closer to death.
Here, about 300 seconds before death, there was a power surge at the high gamma frequencies.
This spike in power occurred in the somatosensory cortex and the dorsolateral prefrontal cortex, areas that are associated with conscious experience. It seems that this patient, 5 minutes before death, was experiencing something.
But I know what you’re thinking. This is a brain that is not receiving oxygen. Cells are going to become disordered quickly and start firing randomly – a last gasp, so to speak, before the end. Meaningless noise.
But connectivity mapping tells a different story. The signals seem to have structure.
Those high-frequency power surges increased connectivity in the posterior cortical “hot zone,” an area of the brain many researchers feel is necessary for conscious perception. This figure is not a map of raw brain electrical output like the one I showed before, but of coherence between brain regions in the consciousness hot zone. Those red areas indicate cross-talk – not the disordered scream of dying neurons, but a last set of messages passing back and forth from the parietal and posterior temporal lobes.
In fact, the electrical patterns of the brains in these patients looked very similar to the patterns seen in dreaming humans, as well as in patients with epilepsy who report sensations of out-of-body experiences.
It’s critical to realize two things here. First, these signals of consciousness were not present before life support was withdrawn. These comatose patients had minimal brain activity; there was no evidence that they were experiencing anything before the process of dying began. These brains are behaving fundamentally differently near death.
But second, we must realize that, although the brains of these individuals, in their last moments, appeared to be acting in a way that conscious brains act, we have no way of knowing if the patients were truly having a conscious experience. As I said, all the patients in the study died. Short of those metaphysics I alluded to earlier, we will have no way to ask them how they experienced their final moments.
Let’s be clear: This study doesn’t answer the question of what happens when we die. It says nothing about life after death or the existence or persistence of the soul. But what it does do is shed light on an incredibly difficult problem in neuroscience: the problem of consciousness. And as studies like this move forward, we may discover that the root of consciousness comes not from the breath of God or the energy of a living universe, but from very specific parts of the very complicated machine that is the brain, acting together to produce something transcendent. And to me, that is no less sublime.
Dr. Wilson is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator, Yale University, New Haven, Conn. His science communication work can be found in the Huffington Post, on NPR, and on Medscape. He tweets @fperrywilson and his new book, How Medicine Works and When It Doesn’t, is available now. Dr. Wilson has disclosed no relevant financial relationships.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
All the participants in the study I am going to tell you about this week died. And three of them died twice. But their deaths provide us with a fascinating window into the complex electrochemistry of the dying brain. What we might be looking at, indeed, is the physiologic correlate of the near-death experience.
The concept of the near-death experience is culturally ubiquitous. And though the content seems to track along culture lines – Western Christians are more likely to report seeing guardian angels, while Hindus are more likely to report seeing messengers of the god of death – certain factors seem to transcend culture: an out-of-body experience; a feeling of peace; and, of course, the light at the end of the tunnel.
As a materialist, I won’t discuss the possibility that these commonalities reflect some metaphysical structure to the afterlife. More likely, it seems to me, is that the commonalities result from the fact that the experience is mediated by our brains, and our brains, when dying, may be more alike than different.
We are talking about this study, appearing in the Proceedings of the National Academy of Sciences, by Jimo Borjigin and her team.
Dr. Borjigin studies the neural correlates of consciousness, perhaps one of the biggest questions in all of science today. To wit,
The study in question follows four unconscious patients –comatose patients, really – as life-sustaining support was withdrawn, up until the moment of death. Three had suffered severe anoxic brain injury in the setting of prolonged cardiac arrest. Though the heart was restarted, the brain damage was severe. The fourth had a large brain hemorrhage. All four patients were thus comatose and, though not brain-dead, unresponsive – with the lowest possible Glasgow Coma Scale score. No response to outside stimuli.
The families had made the decision to withdraw life support – to remove the breathing tube – but agreed to enroll their loved one in the study.
The team applied EEG leads to the head, EKG leads to the chest, and other monitoring equipment to observe the physiologic changes that occurred as the comatose and unresponsive patient died.
As the heart rhythm evolved from this:
To this:
And eventually stopped.
But this is a study about the brain, not the heart.
Prior to the withdrawal of life support, the brain electrical signals looked like this:
What you see is the EEG power at various frequencies, with red being higher. All the red was down at the low frequencies. Consciousness, at least as we understand it, is a higher-frequency phenomenon.
Right after the breathing tube was removed, the power didn’t change too much, but you can see some increased activity at the higher frequencies.
But in two of the four patients, something really surprising happened. Watch what happens as the brain gets closer and closer to death.
Here, about 300 seconds before death, there was a power surge at the high gamma frequencies.
This spike in power occurred in the somatosensory cortex and the dorsolateral prefrontal cortex, areas that are associated with conscious experience. It seems that this patient, 5 minutes before death, was experiencing something.
But I know what you’re thinking. This is a brain that is not receiving oxygen. Cells are going to become disordered quickly and start firing randomly – a last gasp, so to speak, before the end. Meaningless noise.
But connectivity mapping tells a different story. The signals seem to have structure.
Those high-frequency power surges increased connectivity in the posterior cortical “hot zone,” an area of the brain many researchers feel is necessary for conscious perception. This figure is not a map of raw brain electrical output like the one I showed before, but of coherence between brain regions in the consciousness hot zone. Those red areas indicate cross-talk – not the disordered scream of dying neurons, but a last set of messages passing back and forth from the parietal and posterior temporal lobes.
In fact, the electrical patterns of the brains in these patients looked very similar to the patterns seen in dreaming humans, as well as in patients with epilepsy who report sensations of out-of-body experiences.
It’s critical to realize two things here. First, these signals of consciousness were not present before life support was withdrawn. These comatose patients had minimal brain activity; there was no evidence that they were experiencing anything before the process of dying began. These brains are behaving fundamentally differently near death.
But second, we must realize that, although the brains of these individuals, in their last moments, appeared to be acting in a way that conscious brains act, we have no way of knowing if the patients were truly having a conscious experience. As I said, all the patients in the study died. Short of those metaphysics I alluded to earlier, we will have no way to ask them how they experienced their final moments.
Let’s be clear: This study doesn’t answer the question of what happens when we die. It says nothing about life after death or the existence or persistence of the soul. But what it does do is shed light on an incredibly difficult problem in neuroscience: the problem of consciousness. And as studies like this move forward, we may discover that the root of consciousness comes not from the breath of God or the energy of a living universe, but from very specific parts of the very complicated machine that is the brain, acting together to produce something transcendent. And to me, that is no less sublime.
Dr. Wilson is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator, Yale University, New Haven, Conn. His science communication work can be found in the Huffington Post, on NPR, and on Medscape. He tweets @fperrywilson and his new book, How Medicine Works and When It Doesn’t, is available now. Dr. Wilson has disclosed no relevant financial relationships.
Malaria: Not just someone else’s problem
What is the most dangerous animal on Earth? Which one has killed more humans since we first began walking upright?
The mind leaps to the vicious and dangerous – great white sharks. lions. tigers. crocodiles. The fearsome predators of the planet But realistically, more people are killed and injured by large herbivores each year than predators. Just watch news updates from Yellowstone during their busy season.
Anyway, the correct answer is ... none of the above.
It’s the mosquito, and the many microbes it’s a vector for. Malaria, in particular. Even the once-devastating bubonic plague is no longer a major concern.
What do Presidents Washington, Kennedy, Eisenhower, Lincoln, Monroe, Grant, Garfield, Jackson, Teddy Roosevelt, and other historical VIPs like Oliver Cromwell, King Tut, and numerous kings, queens, and popes all have in common? They all had malaria. Cromwell, Tut, and many royal and religious figures died of it.
You can make a solid argument that malaria is the disease that’s affected the course of history more than any other (you could make a good case for the plague, too, but it’s less relevant today). The control of malaria is what allowed the Panama canal to happen.
I’m bringing this up because, mostly overlooked in the news recently as we argued about light beer endorsements, TV pundits, and the NFL draft, is the approval and gradual increase in use of a malaria vaccine.
This is a pretty big deal given the scope of the problem and the fact that the most effective prevention up until recently was a mosquito net.
We tend to see malaria as someone else’s problem, something that affects the tropics, but forget that as recently as the 1940s it was still common in the U.S. During the Civil War as many as 1 million soldiers were infected with it. Given the right conditions it could easily return here.
Which is why we should be more aware of these things. As COVID showed, infectious diseases are never some other country’s, or continent’s, problem. They affect all of us either directly or indirectly. In the interconnected economies of the world illnesses in one area can spread to others. Even if they don’t they can still have significant effects on supply chains, since so much of what we depend on comes from somewhere else.
COVID, by comparison, is small beer. Just think about smallpox, or the plague, or polio, as to what an unchecked disease can do to a society until medicine catches up with it.
There will always be new diseases. Microbes and humans have been in a state of hostilities for a few million years now, and likely always will be. But every victory along the way is a victory for everyone, regardless of who they are or where they live.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
What is the most dangerous animal on Earth? Which one has killed more humans since we first began walking upright?
The mind leaps to the vicious and dangerous – great white sharks. lions. tigers. crocodiles. The fearsome predators of the planet But realistically, more people are killed and injured by large herbivores each year than predators. Just watch news updates from Yellowstone during their busy season.
Anyway, the correct answer is ... none of the above.
It’s the mosquito, and the many microbes it’s a vector for. Malaria, in particular. Even the once-devastating bubonic plague is no longer a major concern.
What do Presidents Washington, Kennedy, Eisenhower, Lincoln, Monroe, Grant, Garfield, Jackson, Teddy Roosevelt, and other historical VIPs like Oliver Cromwell, King Tut, and numerous kings, queens, and popes all have in common? They all had malaria. Cromwell, Tut, and many royal and religious figures died of it.
You can make a solid argument that malaria is the disease that’s affected the course of history more than any other (you could make a good case for the plague, too, but it’s less relevant today). The control of malaria is what allowed the Panama canal to happen.
I’m bringing this up because, mostly overlooked in the news recently as we argued about light beer endorsements, TV pundits, and the NFL draft, is the approval and gradual increase in use of a malaria vaccine.
This is a pretty big deal given the scope of the problem and the fact that the most effective prevention up until recently was a mosquito net.
We tend to see malaria as someone else’s problem, something that affects the tropics, but forget that as recently as the 1940s it was still common in the U.S. During the Civil War as many as 1 million soldiers were infected with it. Given the right conditions it could easily return here.
Which is why we should be more aware of these things. As COVID showed, infectious diseases are never some other country’s, or continent’s, problem. They affect all of us either directly or indirectly. In the interconnected economies of the world illnesses in one area can spread to others. Even if they don’t they can still have significant effects on supply chains, since so much of what we depend on comes from somewhere else.
COVID, by comparison, is small beer. Just think about smallpox, or the plague, or polio, as to what an unchecked disease can do to a society until medicine catches up with it.
There will always be new diseases. Microbes and humans have been in a state of hostilities for a few million years now, and likely always will be. But every victory along the way is a victory for everyone, regardless of who they are or where they live.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
What is the most dangerous animal on Earth? Which one has killed more humans since we first began walking upright?
The mind leaps to the vicious and dangerous – great white sharks. lions. tigers. crocodiles. The fearsome predators of the planet But realistically, more people are killed and injured by large herbivores each year than predators. Just watch news updates from Yellowstone during their busy season.
Anyway, the correct answer is ... none of the above.
It’s the mosquito, and the many microbes it’s a vector for. Malaria, in particular. Even the once-devastating bubonic plague is no longer a major concern.
What do Presidents Washington, Kennedy, Eisenhower, Lincoln, Monroe, Grant, Garfield, Jackson, Teddy Roosevelt, and other historical VIPs like Oliver Cromwell, King Tut, and numerous kings, queens, and popes all have in common? They all had malaria. Cromwell, Tut, and many royal and religious figures died of it.
You can make a solid argument that malaria is the disease that’s affected the course of history more than any other (you could make a good case for the plague, too, but it’s less relevant today). The control of malaria is what allowed the Panama canal to happen.
I’m bringing this up because, mostly overlooked in the news recently as we argued about light beer endorsements, TV pundits, and the NFL draft, is the approval and gradual increase in use of a malaria vaccine.
This is a pretty big deal given the scope of the problem and the fact that the most effective prevention up until recently was a mosquito net.
We tend to see malaria as someone else’s problem, something that affects the tropics, but forget that as recently as the 1940s it was still common in the U.S. During the Civil War as many as 1 million soldiers were infected with it. Given the right conditions it could easily return here.
Which is why we should be more aware of these things. As COVID showed, infectious diseases are never some other country’s, or continent’s, problem. They affect all of us either directly or indirectly. In the interconnected economies of the world illnesses in one area can spread to others. Even if they don’t they can still have significant effects on supply chains, since so much of what we depend on comes from somewhere else.
COVID, by comparison, is small beer. Just think about smallpox, or the plague, or polio, as to what an unchecked disease can do to a society until medicine catches up with it.
There will always be new diseases. Microbes and humans have been in a state of hostilities for a few million years now, and likely always will be. But every victory along the way is a victory for everyone, regardless of who they are or where they live.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Getting a white-bagging exemption: A win for the patient, employer, and rheumatologist
Whether it’s filling out a prior authorization form or testifying before Congress, it is an action we perform that ultimately helps our patients achieve that care. We are familiar with many of the obstacles that block the path to the best care and interfere with our patient-doctor relationships. Much work has been done to pass legislation in the states to mitigate some of those obstacles, such as unreasonable step therapy regimens, nonmedical switching, and copay accumulators.
Unfortunately, that state legislation does not cover patients who work for companies that are self-insured. Self-insured employers, which account for about 60% of America’s workers, directly pay for the health benefits offered to employees instead of buying “fully funded” insurance plans. Most of those self-funded plans fall under “ERISA” protections and are regulated by the federal Department of Labor. ERISA stands for Employee Retirement Income Security Act. The law, which was enacted in 1974, also covers employee health plans. These plans must act as a fiduciary, meaning they must look after the well-being of the employees, including their finances and those of the plan itself.
The Coalition of State Rheumatology Organizations (CSRO) has learned of a number of issues involving patients who work for self-funded companies, regulated by ERISA. One such issue is that of mandated “white bagging.” White bagging has been discussed in “Rheum for Action” in the past. There is a long list of white-bagging problems, including dosing issues, lack of “chain of custody” with the medications, delays in treatment, mandatory up-front payments by the patient, and wastage of unused medication. However, there is another issue that is of concern not only to the employees (our patients) but to the employer as well.
Employers’ fiduciary responsibility
As mentioned earlier, the employers who self insure are responsible for the financial well-being of their employee and the plan itself. Therefore, if certain practices are mandated within the health plan that harm our patients or the plan financially, the company could be in violation of their fiduciary duty. Rheumatologists have said that buying and billing the drug to the medical side of the health plan in many cases costs much less than white bagging. Conceivably, that could result in breach of an employer’s fiduciary duty to their employee.
Evidence for violating fiduciary duty
CSRO recently received redacted receipts comparing costs between the two models of drug acquisition for a patient in an ERISA plan. White bagging for the patient occurred in 2021, and in 2022 an exemption was granted for the rheumatologist to buy and bill the administered medication. Unfortunately, the exemption to buy and bill in 2023 was denied and continues to be denied (as of this writing). A comparison of the receipts revealed the company was charged over $40,000 for the white-bagged medication in 2021, and the patient’s cost share for that year was $525. Under the traditional buy-and-bill acquisition model in 2022, the company was charged around $12,000 for the medication and the patient’s cost share was $30. There is a clear difference in cost to the employee and plan between the two acquisition models.
Is this major company unknowingly violating its fiduciary duty by mandating white bagging as per their contract with one of the three big pharmacy benefit managers (PBMs)? If so, how does something like this happen with a large national company that has ERISA attorneys looking over the contracts with the PBMs?
Why is white bagging mandated?
Often, white bagging is mandated because the cost of infusions in a hospital outpatient facility can be very high. Nationally, it has been shown that hospitals charge four to five times the cost they paid for the drug, and the 100 most expensive hospitals charge 10-18 times the cost of their drugs. With these up-charges, white bagging could easily be a lower cost for employee and company. But across-the-board mandating of white bagging ignores that physician office–based infusions may offer a much lower cost to employees and the employer.
Another reason large and small self-funded companies may unknowingly sign contracts that are often more profitable to the PBM than to the employer is that the employer pharmacy benefit consultants are paid handsomely by the big PBMs and have been known to “rig” the contract in favor of the PBM, according to Paul Holmes, an ERISA attorney with a focus in pharmacy health plan contracts. Clearly, the PBM profits more with white-bagged medicines billed through the pharmacy (PBM) side of insurance as opposed to buy-and-bill medications that are billed on the medical side of insurance. So mandated white bagging is often included in these contracts, ignoring the lower cost in an infusion suite at a physician’s office.
Suggestions for employers
Employers and employees should be able to obtain the costs of mandated, white-bagged drugs from their PBMs because the Consolidated Appropriations Act of 2021 (CAA) mandates that group health plans ensure access to cost data. The employer should also have access to their consultant’s compensation from the PBM as Section 202 in the CAA states that employer benefit consultants must “disclose actual and anticipated cash and non-cash compensation they expect to earn in connection with the sale, renewal, and extension of group health insurance.”
It would be wise for all self-insured companies to use this section to see how much their consultants are being influenced by the company that they are recommending. Additionally, the companies should consider hiring ERISA attorneys that understand not only the legalese of the contract with a PBM but also the pharmacy lingo, such as the difference between maximum allowable cost, average wholesale price, average sales price, and average manufacturer’s price.
Suggestion for the rheumatologist
This leads to a suggestion to rheumatologists trying to get an exemption from mandated white bagging. If a patient has already had white-bagged medication, have them obtain a receipt from the PBM for their charges to the plan for the medication. If the patient has not gone through the white bagging yet, the PBM should be able to tell the plan the cost of the white-bagged medication and the cost to the patient. Compare those costs with what would be charged through buy and bill, and if it is less, present that evidence to the employer and remind them of their fiduciary responsibility to their employees.
Granted, this process may take more effort than filling out a prior authorization, but getting the white-bag exemption will help the patient, the employer, and the rheumatologist in the long run. A win-win-win!
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s Vice President of Advocacy and Government Affairs and its immediate Past President, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Whether it’s filling out a prior authorization form or testifying before Congress, it is an action we perform that ultimately helps our patients achieve that care. We are familiar with many of the obstacles that block the path to the best care and interfere with our patient-doctor relationships. Much work has been done to pass legislation in the states to mitigate some of those obstacles, such as unreasonable step therapy regimens, nonmedical switching, and copay accumulators.
Unfortunately, that state legislation does not cover patients who work for companies that are self-insured. Self-insured employers, which account for about 60% of America’s workers, directly pay for the health benefits offered to employees instead of buying “fully funded” insurance plans. Most of those self-funded plans fall under “ERISA” protections and are regulated by the federal Department of Labor. ERISA stands for Employee Retirement Income Security Act. The law, which was enacted in 1974, also covers employee health plans. These plans must act as a fiduciary, meaning they must look after the well-being of the employees, including their finances and those of the plan itself.
The Coalition of State Rheumatology Organizations (CSRO) has learned of a number of issues involving patients who work for self-funded companies, regulated by ERISA. One such issue is that of mandated “white bagging.” White bagging has been discussed in “Rheum for Action” in the past. There is a long list of white-bagging problems, including dosing issues, lack of “chain of custody” with the medications, delays in treatment, mandatory up-front payments by the patient, and wastage of unused medication. However, there is another issue that is of concern not only to the employees (our patients) but to the employer as well.
Employers’ fiduciary responsibility
As mentioned earlier, the employers who self insure are responsible for the financial well-being of their employee and the plan itself. Therefore, if certain practices are mandated within the health plan that harm our patients or the plan financially, the company could be in violation of their fiduciary duty. Rheumatologists have said that buying and billing the drug to the medical side of the health plan in many cases costs much less than white bagging. Conceivably, that could result in breach of an employer’s fiduciary duty to their employee.
Evidence for violating fiduciary duty
CSRO recently received redacted receipts comparing costs between the two models of drug acquisition for a patient in an ERISA plan. White bagging for the patient occurred in 2021, and in 2022 an exemption was granted for the rheumatologist to buy and bill the administered medication. Unfortunately, the exemption to buy and bill in 2023 was denied and continues to be denied (as of this writing). A comparison of the receipts revealed the company was charged over $40,000 for the white-bagged medication in 2021, and the patient’s cost share for that year was $525. Under the traditional buy-and-bill acquisition model in 2022, the company was charged around $12,000 for the medication and the patient’s cost share was $30. There is a clear difference in cost to the employee and plan between the two acquisition models.
Is this major company unknowingly violating its fiduciary duty by mandating white bagging as per their contract with one of the three big pharmacy benefit managers (PBMs)? If so, how does something like this happen with a large national company that has ERISA attorneys looking over the contracts with the PBMs?
Why is white bagging mandated?
Often, white bagging is mandated because the cost of infusions in a hospital outpatient facility can be very high. Nationally, it has been shown that hospitals charge four to five times the cost they paid for the drug, and the 100 most expensive hospitals charge 10-18 times the cost of their drugs. With these up-charges, white bagging could easily be a lower cost for employee and company. But across-the-board mandating of white bagging ignores that physician office–based infusions may offer a much lower cost to employees and the employer.
Another reason large and small self-funded companies may unknowingly sign contracts that are often more profitable to the PBM than to the employer is that the employer pharmacy benefit consultants are paid handsomely by the big PBMs and have been known to “rig” the contract in favor of the PBM, according to Paul Holmes, an ERISA attorney with a focus in pharmacy health plan contracts. Clearly, the PBM profits more with white-bagged medicines billed through the pharmacy (PBM) side of insurance as opposed to buy-and-bill medications that are billed on the medical side of insurance. So mandated white bagging is often included in these contracts, ignoring the lower cost in an infusion suite at a physician’s office.
Suggestions for employers
Employers and employees should be able to obtain the costs of mandated, white-bagged drugs from their PBMs because the Consolidated Appropriations Act of 2021 (CAA) mandates that group health plans ensure access to cost data. The employer should also have access to their consultant’s compensation from the PBM as Section 202 in the CAA states that employer benefit consultants must “disclose actual and anticipated cash and non-cash compensation they expect to earn in connection with the sale, renewal, and extension of group health insurance.”
It would be wise for all self-insured companies to use this section to see how much their consultants are being influenced by the company that they are recommending. Additionally, the companies should consider hiring ERISA attorneys that understand not only the legalese of the contract with a PBM but also the pharmacy lingo, such as the difference between maximum allowable cost, average wholesale price, average sales price, and average manufacturer’s price.
Suggestion for the rheumatologist
This leads to a suggestion to rheumatologists trying to get an exemption from mandated white bagging. If a patient has already had white-bagged medication, have them obtain a receipt from the PBM for their charges to the plan for the medication. If the patient has not gone through the white bagging yet, the PBM should be able to tell the plan the cost of the white-bagged medication and the cost to the patient. Compare those costs with what would be charged through buy and bill, and if it is less, present that evidence to the employer and remind them of their fiduciary responsibility to their employees.
Granted, this process may take more effort than filling out a prior authorization, but getting the white-bag exemption will help the patient, the employer, and the rheumatologist in the long run. A win-win-win!
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s Vice President of Advocacy and Government Affairs and its immediate Past President, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Whether it’s filling out a prior authorization form or testifying before Congress, it is an action we perform that ultimately helps our patients achieve that care. We are familiar with many of the obstacles that block the path to the best care and interfere with our patient-doctor relationships. Much work has been done to pass legislation in the states to mitigate some of those obstacles, such as unreasonable step therapy regimens, nonmedical switching, and copay accumulators.
Unfortunately, that state legislation does not cover patients who work for companies that are self-insured. Self-insured employers, which account for about 60% of America’s workers, directly pay for the health benefits offered to employees instead of buying “fully funded” insurance plans. Most of those self-funded plans fall under “ERISA” protections and are regulated by the federal Department of Labor. ERISA stands for Employee Retirement Income Security Act. The law, which was enacted in 1974, also covers employee health plans. These plans must act as a fiduciary, meaning they must look after the well-being of the employees, including their finances and those of the plan itself.
The Coalition of State Rheumatology Organizations (CSRO) has learned of a number of issues involving patients who work for self-funded companies, regulated by ERISA. One such issue is that of mandated “white bagging.” White bagging has been discussed in “Rheum for Action” in the past. There is a long list of white-bagging problems, including dosing issues, lack of “chain of custody” with the medications, delays in treatment, mandatory up-front payments by the patient, and wastage of unused medication. However, there is another issue that is of concern not only to the employees (our patients) but to the employer as well.
Employers’ fiduciary responsibility
As mentioned earlier, the employers who self insure are responsible for the financial well-being of their employee and the plan itself. Therefore, if certain practices are mandated within the health plan that harm our patients or the plan financially, the company could be in violation of their fiduciary duty. Rheumatologists have said that buying and billing the drug to the medical side of the health plan in many cases costs much less than white bagging. Conceivably, that could result in breach of an employer’s fiduciary duty to their employee.
Evidence for violating fiduciary duty
CSRO recently received redacted receipts comparing costs between the two models of drug acquisition for a patient in an ERISA plan. White bagging for the patient occurred in 2021, and in 2022 an exemption was granted for the rheumatologist to buy and bill the administered medication. Unfortunately, the exemption to buy and bill in 2023 was denied and continues to be denied (as of this writing). A comparison of the receipts revealed the company was charged over $40,000 for the white-bagged medication in 2021, and the patient’s cost share for that year was $525. Under the traditional buy-and-bill acquisition model in 2022, the company was charged around $12,000 for the medication and the patient’s cost share was $30. There is a clear difference in cost to the employee and plan between the two acquisition models.
Is this major company unknowingly violating its fiduciary duty by mandating white bagging as per their contract with one of the three big pharmacy benefit managers (PBMs)? If so, how does something like this happen with a large national company that has ERISA attorneys looking over the contracts with the PBMs?
Why is white bagging mandated?
Often, white bagging is mandated because the cost of infusions in a hospital outpatient facility can be very high. Nationally, it has been shown that hospitals charge four to five times the cost they paid for the drug, and the 100 most expensive hospitals charge 10-18 times the cost of their drugs. With these up-charges, white bagging could easily be a lower cost for employee and company. But across-the-board mandating of white bagging ignores that physician office–based infusions may offer a much lower cost to employees and the employer.
Another reason large and small self-funded companies may unknowingly sign contracts that are often more profitable to the PBM than to the employer is that the employer pharmacy benefit consultants are paid handsomely by the big PBMs and have been known to “rig” the contract in favor of the PBM, according to Paul Holmes, an ERISA attorney with a focus in pharmacy health plan contracts. Clearly, the PBM profits more with white-bagged medicines billed through the pharmacy (PBM) side of insurance as opposed to buy-and-bill medications that are billed on the medical side of insurance. So mandated white bagging is often included in these contracts, ignoring the lower cost in an infusion suite at a physician’s office.
Suggestions for employers
Employers and employees should be able to obtain the costs of mandated, white-bagged drugs from their PBMs because the Consolidated Appropriations Act of 2021 (CAA) mandates that group health plans ensure access to cost data. The employer should also have access to their consultant’s compensation from the PBM as Section 202 in the CAA states that employer benefit consultants must “disclose actual and anticipated cash and non-cash compensation they expect to earn in connection with the sale, renewal, and extension of group health insurance.”
It would be wise for all self-insured companies to use this section to see how much their consultants are being influenced by the company that they are recommending. Additionally, the companies should consider hiring ERISA attorneys that understand not only the legalese of the contract with a PBM but also the pharmacy lingo, such as the difference between maximum allowable cost, average wholesale price, average sales price, and average manufacturer’s price.
Suggestion for the rheumatologist
This leads to a suggestion to rheumatologists trying to get an exemption from mandated white bagging. If a patient has already had white-bagged medication, have them obtain a receipt from the PBM for their charges to the plan for the medication. If the patient has not gone through the white bagging yet, the PBM should be able to tell the plan the cost of the white-bagged medication and the cost to the patient. Compare those costs with what would be charged through buy and bill, and if it is less, present that evidence to the employer and remind them of their fiduciary responsibility to their employees.
Granted, this process may take more effort than filling out a prior authorization, but getting the white-bag exemption will help the patient, the employer, and the rheumatologist in the long run. A win-win-win!
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s Vice President of Advocacy and Government Affairs and its immediate Past President, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Some decisions aren’t right or wrong; they’re just devastating
There is one situation, while not common, that is often among the most difficult for me: the person who must be told at diagnosis that they are already dying. I am still reminded of a patient I saw early in my career.
A woman in her 40s was admitted to the hospital complaining of severe shortness of breath. In retrospect, she had been sick for months. She had not sought help because she was young and thought it would pass – the results of a “bad bug” that she just couldn’t shake.
But in the past few weeks, the persistence of symptoms became associated with weight loss, profound fatigue, loss of appetite, and nausea.
By the time she was hospitalized she was emaciated, though she appeared pregnant – a sign of the fluid that had built up in her abdomen. Imaging showed that her abdomen was filled with disease (carcinomatosis) and her liver and lungs were nearly replaced with metastatic disease.
A biopsy revealed an aggressive cancer that had no identifying histologic marker: carcinoma, not otherwise specified, or cancer of unknown primary.
I still remember seeing her. She had a deer-in-headlights stare that held me as I approached. I introduced myself and sat down so we were eye to eye.
“Tell me what you know,” I said.
“I know I have cancer and they don’t know where it started. I know surgery is not an option and that’s why they’ve asked you to come. Whatever. I’m ready. I want to fight this because I know I can beat it,” she said.
I remember that she looked very sick; her thin face and arms contrasted with her large, distended abdomen. Her breathing was labored, her skin almost gray. For a moment I didn’t know what to say.
As doctors, we like to believe that our decisions are guided by data: the randomized trials and meta-analyses that set standards of care; phase 2 trials that establish evidence (or lack thereof) of activity; case-control studies that suggest the impacts of treatment; and at the very least, case studies that document that “N of 1” experience. We have expert panels and pathways that lay out what treatments we should be using to help ensure access to quality care in every clinic on every corner of every cancer center in the United States.
These data and pathways tell us objectively what we can expect from therapy, who is at most risk for toxicities, and profiles of patients for whom treatment is not likely to be of benefit. In an ideal world, this objectivity would help us help people decide on an approach. But life is not objective, and sometimes individualizing care is as important as data.
In this scenario, I knew only one thing: She was dying. She had an overwhelming tumor burden. But I still asked myself a question that many in, and outside of, oncology ask themselves: Could she be saved?
This question was made even more difficult because she was young. She had her whole life ahead of her. It seemed incongruous that she would be here now, facing the gravity of her situation.
Looking at her, I saw the person, not a data point in a trial or a statistic in a textbook. She was terrified. And she was not ready to die.
I sat down and reviewed what I knew about her cancer and what I did not know. I went through potential treatments we could try and the toxicities associated with each. I made clear that these treatments, based on how sick she was, could kill her.
“Whatever we do,” I said, “you do not have disease that I can cure.”
She cried then, realizing what a horrible situation she was in and that she would no longer go back to her normal life. Indeed, she seemed to grasp that she was probably facing the end of her life and that it could be short.
“My concern is,” I continued, “that treatment could do the exact opposite of what I hope it would do. It could kill you sooner than this cancer will.”
Instead of making a treatment plan, I decided that it would be best to come back another day, so I said my goodbyes and left. Still, I could not stop thinking about her and what I should suggest as her next steps.
I asked colleagues what they would suggest. Some recommended hospice care, others recommended treatment. Clearly, there was no one way to proceed.
One might wonder: Why is it so hard to do the right thing?
Ask any clinician and I think you will hear the same answer: Because we do not have the luxury of certainty.
Am I certain that this person will not benefit from intubation? Am I certain that she has only weeks to live? Am I sure that there are no treatments that will work?
The answer to these questions is no – I am not certain. It is that uncertainty that always makes me pause because it reminds me of my own humanity.
I stopped by the next day to see her surrounded by family. After some pleasantries I took the opportunity to reiterate much of our conversation from the other day. After some questions, I looked at her and asked if she wanted to talk more about her options. I was prepared to suggest treatment, anticipating that she would want it. Instead, she told me she didn’t want to proceed.
“I feel like I’m dying, and if what you have to give me isn’t going to cure me, then I’d prefer not to suffer while it happens. You said it’s up to me. I don’t want it.”
First, do no harm. It’s one of the tenets of medicine – to provide care that will benefit the people who have trusted us with their lives, whether that be longevity, relief of symptoms, or helping them achieve their last wishes. Throughout one’s life, goals might change but that edict remains the same.
But that can be difficult, especially in oncology and especially when one is not prepared for their own end of life. It can be hard for doctors to discuss the end of life; it’s easier to focus on the next treatment, instilling hope that there’s more that can be done. And there are people with end-stage cancer who insist on continuing treatment in the same circumstances, preferring to “die fighting” than to “give up.” Involving supportive and palliative care specialists early has helped in both situations, which is certainly a good thing.
We talked a while more and then arranged for our palliative care team to see her. I wish I could say I was at peace with her decision, but I wasn’t. The truth is, whatever she decided would probably have the same impact: I wouldn’t be able to stop thinking about it.
Dr. Dizon is professor of medicine, department of medicine, at Brown University and director of medical oncology at Rhode Island Hospital, both in Providence, R.I. He disclosed conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
There is one situation, while not common, that is often among the most difficult for me: the person who must be told at diagnosis that they are already dying. I am still reminded of a patient I saw early in my career.
A woman in her 40s was admitted to the hospital complaining of severe shortness of breath. In retrospect, she had been sick for months. She had not sought help because she was young and thought it would pass – the results of a “bad bug” that she just couldn’t shake.
But in the past few weeks, the persistence of symptoms became associated with weight loss, profound fatigue, loss of appetite, and nausea.
By the time she was hospitalized she was emaciated, though she appeared pregnant – a sign of the fluid that had built up in her abdomen. Imaging showed that her abdomen was filled with disease (carcinomatosis) and her liver and lungs were nearly replaced with metastatic disease.
A biopsy revealed an aggressive cancer that had no identifying histologic marker: carcinoma, not otherwise specified, or cancer of unknown primary.
I still remember seeing her. She had a deer-in-headlights stare that held me as I approached. I introduced myself and sat down so we were eye to eye.
“Tell me what you know,” I said.
“I know I have cancer and they don’t know where it started. I know surgery is not an option and that’s why they’ve asked you to come. Whatever. I’m ready. I want to fight this because I know I can beat it,” she said.
I remember that she looked very sick; her thin face and arms contrasted with her large, distended abdomen. Her breathing was labored, her skin almost gray. For a moment I didn’t know what to say.
As doctors, we like to believe that our decisions are guided by data: the randomized trials and meta-analyses that set standards of care; phase 2 trials that establish evidence (or lack thereof) of activity; case-control studies that suggest the impacts of treatment; and at the very least, case studies that document that “N of 1” experience. We have expert panels and pathways that lay out what treatments we should be using to help ensure access to quality care in every clinic on every corner of every cancer center in the United States.
These data and pathways tell us objectively what we can expect from therapy, who is at most risk for toxicities, and profiles of patients for whom treatment is not likely to be of benefit. In an ideal world, this objectivity would help us help people decide on an approach. But life is not objective, and sometimes individualizing care is as important as data.
In this scenario, I knew only one thing: She was dying. She had an overwhelming tumor burden. But I still asked myself a question that many in, and outside of, oncology ask themselves: Could she be saved?
This question was made even more difficult because she was young. She had her whole life ahead of her. It seemed incongruous that she would be here now, facing the gravity of her situation.
Looking at her, I saw the person, not a data point in a trial or a statistic in a textbook. She was terrified. And she was not ready to die.
I sat down and reviewed what I knew about her cancer and what I did not know. I went through potential treatments we could try and the toxicities associated with each. I made clear that these treatments, based on how sick she was, could kill her.
“Whatever we do,” I said, “you do not have disease that I can cure.”
She cried then, realizing what a horrible situation she was in and that she would no longer go back to her normal life. Indeed, she seemed to grasp that she was probably facing the end of her life and that it could be short.
“My concern is,” I continued, “that treatment could do the exact opposite of what I hope it would do. It could kill you sooner than this cancer will.”
Instead of making a treatment plan, I decided that it would be best to come back another day, so I said my goodbyes and left. Still, I could not stop thinking about her and what I should suggest as her next steps.
I asked colleagues what they would suggest. Some recommended hospice care, others recommended treatment. Clearly, there was no one way to proceed.
One might wonder: Why is it so hard to do the right thing?
Ask any clinician and I think you will hear the same answer: Because we do not have the luxury of certainty.
Am I certain that this person will not benefit from intubation? Am I certain that she has only weeks to live? Am I sure that there are no treatments that will work?
The answer to these questions is no – I am not certain. It is that uncertainty that always makes me pause because it reminds me of my own humanity.
I stopped by the next day to see her surrounded by family. After some pleasantries I took the opportunity to reiterate much of our conversation from the other day. After some questions, I looked at her and asked if she wanted to talk more about her options. I was prepared to suggest treatment, anticipating that she would want it. Instead, she told me she didn’t want to proceed.
“I feel like I’m dying, and if what you have to give me isn’t going to cure me, then I’d prefer not to suffer while it happens. You said it’s up to me. I don’t want it.”
First, do no harm. It’s one of the tenets of medicine – to provide care that will benefit the people who have trusted us with their lives, whether that be longevity, relief of symptoms, or helping them achieve their last wishes. Throughout one’s life, goals might change but that edict remains the same.
But that can be difficult, especially in oncology and especially when one is not prepared for their own end of life. It can be hard for doctors to discuss the end of life; it’s easier to focus on the next treatment, instilling hope that there’s more that can be done. And there are people with end-stage cancer who insist on continuing treatment in the same circumstances, preferring to “die fighting” than to “give up.” Involving supportive and palliative care specialists early has helped in both situations, which is certainly a good thing.
We talked a while more and then arranged for our palliative care team to see her. I wish I could say I was at peace with her decision, but I wasn’t. The truth is, whatever she decided would probably have the same impact: I wouldn’t be able to stop thinking about it.
Dr. Dizon is professor of medicine, department of medicine, at Brown University and director of medical oncology at Rhode Island Hospital, both in Providence, R.I. He disclosed conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
There is one situation, while not common, that is often among the most difficult for me: the person who must be told at diagnosis that they are already dying. I am still reminded of a patient I saw early in my career.
A woman in her 40s was admitted to the hospital complaining of severe shortness of breath. In retrospect, she had been sick for months. She had not sought help because she was young and thought it would pass – the results of a “bad bug” that she just couldn’t shake.
But in the past few weeks, the persistence of symptoms became associated with weight loss, profound fatigue, loss of appetite, and nausea.
By the time she was hospitalized she was emaciated, though she appeared pregnant – a sign of the fluid that had built up in her abdomen. Imaging showed that her abdomen was filled with disease (carcinomatosis) and her liver and lungs were nearly replaced with metastatic disease.
A biopsy revealed an aggressive cancer that had no identifying histologic marker: carcinoma, not otherwise specified, or cancer of unknown primary.
I still remember seeing her. She had a deer-in-headlights stare that held me as I approached. I introduced myself and sat down so we were eye to eye.
“Tell me what you know,” I said.
“I know I have cancer and they don’t know where it started. I know surgery is not an option and that’s why they’ve asked you to come. Whatever. I’m ready. I want to fight this because I know I can beat it,” she said.
I remember that she looked very sick; her thin face and arms contrasted with her large, distended abdomen. Her breathing was labored, her skin almost gray. For a moment I didn’t know what to say.
As doctors, we like to believe that our decisions are guided by data: the randomized trials and meta-analyses that set standards of care; phase 2 trials that establish evidence (or lack thereof) of activity; case-control studies that suggest the impacts of treatment; and at the very least, case studies that document that “N of 1” experience. We have expert panels and pathways that lay out what treatments we should be using to help ensure access to quality care in every clinic on every corner of every cancer center in the United States.
These data and pathways tell us objectively what we can expect from therapy, who is at most risk for toxicities, and profiles of patients for whom treatment is not likely to be of benefit. In an ideal world, this objectivity would help us help people decide on an approach. But life is not objective, and sometimes individualizing care is as important as data.
In this scenario, I knew only one thing: She was dying. She had an overwhelming tumor burden. But I still asked myself a question that many in, and outside of, oncology ask themselves: Could she be saved?
This question was made even more difficult because she was young. She had her whole life ahead of her. It seemed incongruous that she would be here now, facing the gravity of her situation.
Looking at her, I saw the person, not a data point in a trial or a statistic in a textbook. She was terrified. And she was not ready to die.
I sat down and reviewed what I knew about her cancer and what I did not know. I went through potential treatments we could try and the toxicities associated with each. I made clear that these treatments, based on how sick she was, could kill her.
“Whatever we do,” I said, “you do not have disease that I can cure.”
She cried then, realizing what a horrible situation she was in and that she would no longer go back to her normal life. Indeed, she seemed to grasp that she was probably facing the end of her life and that it could be short.
“My concern is,” I continued, “that treatment could do the exact opposite of what I hope it would do. It could kill you sooner than this cancer will.”
Instead of making a treatment plan, I decided that it would be best to come back another day, so I said my goodbyes and left. Still, I could not stop thinking about her and what I should suggest as her next steps.
I asked colleagues what they would suggest. Some recommended hospice care, others recommended treatment. Clearly, there was no one way to proceed.
One might wonder: Why is it so hard to do the right thing?
Ask any clinician and I think you will hear the same answer: Because we do not have the luxury of certainty.
Am I certain that this person will not benefit from intubation? Am I certain that she has only weeks to live? Am I sure that there are no treatments that will work?
The answer to these questions is no – I am not certain. It is that uncertainty that always makes me pause because it reminds me of my own humanity.
I stopped by the next day to see her surrounded by family. After some pleasantries I took the opportunity to reiterate much of our conversation from the other day. After some questions, I looked at her and asked if she wanted to talk more about her options. I was prepared to suggest treatment, anticipating that she would want it. Instead, she told me she didn’t want to proceed.
“I feel like I’m dying, and if what you have to give me isn’t going to cure me, then I’d prefer not to suffer while it happens. You said it’s up to me. I don’t want it.”
First, do no harm. It’s one of the tenets of medicine – to provide care that will benefit the people who have trusted us with their lives, whether that be longevity, relief of symptoms, or helping them achieve their last wishes. Throughout one’s life, goals might change but that edict remains the same.
But that can be difficult, especially in oncology and especially when one is not prepared for their own end of life. It can be hard for doctors to discuss the end of life; it’s easier to focus on the next treatment, instilling hope that there’s more that can be done. And there are people with end-stage cancer who insist on continuing treatment in the same circumstances, preferring to “die fighting” than to “give up.” Involving supportive and palliative care specialists early has helped in both situations, which is certainly a good thing.
We talked a while more and then arranged for our palliative care team to see her. I wish I could say I was at peace with her decision, but I wasn’t. The truth is, whatever she decided would probably have the same impact: I wouldn’t be able to stop thinking about it.
Dr. Dizon is professor of medicine, department of medicine, at Brown University and director of medical oncology at Rhode Island Hospital, both in Providence, R.I. He disclosed conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
Pancreas cysts – What’s the best approach?
Dear colleagues,
Pancreas cysts have become almost ubiquitous in this era of high-resolution cross-sectional imaging. They are a common GI consult with patients and providers worried about the potential risk of malignant transformation. Despite significant research over the past few decades, predicting the natural history of these cysts, especially the side-branch intraductal papillary mucinous neoplasms (IPMNs), remains difficult. There have been a variety of expert recommendations and guidelines, but heterogeneity exists in management especially regarding timing of endoscopic ultrasound, imaging surveillance, and cessation of surveillance. Some centers will present these cysts at multidisciplinary conferences, while others will follow general or local algorithms. In this issue of Perspectives, Dr. Lauren G. Khanna, assistant professor of medicine at NYU Langone Health, New York, and Dr. Santhi Vege, professor of medicine at the Mayo Clinic, Rochester, Minn., present updated and differing approaches to managing these cysts. Which side of the debate are you on? We welcome your thoughts, questions and input– share with us on Twitter @AGA_GIHN
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Conn., and chief of endoscopy at West Haven (Conn.) VA Medical Center. He is an associate editor for GI & Hepatology News.
Continuing pancreas cyst surveillance indefinitely is reasonable
BY LAUREN G. KHANNA, MD, MS
Pancreas cysts remain a clinical challenge. The true incidence of pancreas cysts is unknown, but from MRI and autopsy series, may be up to 50%. Patients presenting with a pancreas cyst often have significant anxiety about their risk of pancreas cancer. We as a medical community initially did too; but over the past few decades as we have gathered more data, we have become more comfortable observing many pancreas cysts. Yet our recommendations for how, how often, and for how long to evaluate pancreas cysts are still very much under debate; there are multiple guidelines with discordant recommendations. In this article, I will discuss my approach to patients with a pancreas cyst.
At the first evaluation, I review available imaging to see if there are characteristic features to determine the type of pancreas cyst: IPMN (including main duct, branch duct, or mixed type), serous cystic neoplasm (SCA), mucinous cystic neoplasm (MCN), solid pseudopapillary neoplasm (SPN), cystic neuroendocrine tumor (NET), or pseudocyst. I also review symptoms, including abdominal pain, weight loss, history of pancreatitis, and onset of diabetes, and check hemoglobin A1c and Ca19-9. I often recommend magnetic resonance cholangiopancreatography (MRCP) if it has not already been obtained and is feasible (that is, if a patient does not have severe claustrophobia or a medical device incompatible with MRI). If a patient is not a candidate for treatment should a pancreatic malignancy be identified, because of age, comorbidities, or preference, I recommend no further evaluation.
Where cyst type remains unclear despite MRCP, and for cysts over 2 cm, I recommend endoscopic ultrasound (EUS) for fluid sampling to assist in determining cyst type and to rule out any other high-risk features. In accordance with international guidelines, if a patient has any concerning imaging features, including main pancreatic duct dilation >5 mm, solid component or mural nodule, or thickened or enhancing duct walls, regardless of cyst size, I recommend EUS to assess for and biopsy any solid component and to sample cyst fluid to examine for dysplasia. Given the lower sensitivity of CT for high-risk features, if MRCP is not feasible, for cysts 1-2 cm, I recommend EUS for better evaluation.
If a cyst is determined to be a cystic NET; main duct or mixed-type IPMN; MCN; or SPN; or a branch duct IPMN with mural nodule, high-grade dysplasia, or adenocarcinoma, and the patient is a surgical candidate, I refer the patient for surgical evaluation. If a cyst is determined to be an SCA, the malignant potential is minimal, and patients do not require follow-up. Patients with a pseudocyst are managed according to their clinical scenario.
Many patients have a proven or suspected branch duct IPMN, an indeterminate cyst, or multiple cysts. Cyst management during surveillance is then determined by the size of the largest cyst and stability of the cyst(s). Of note, patients with an IPMN also have been shown to have an elevated risk of concurrent pancreas adenocarcinoma, which I believe is one of the strongest arguments for heightened surveillance of the entire pancreas in pancreas cyst patients. EUS in particular can identify small or subtle lesions that are not detected by cross-sectional imaging.
If a patient has no prior imaging, in accordance with international and European guidelines, I recommend the first surveillance MRCP at a 6-month interval for cysts <2 cm, which may offer the opportunity to identify rapidly progressing cysts. If a patient has previous imaging available demonstrating stability, I recommend surveillance on an annual basis for cysts <2 cm. For patients with a cyst >2 cm, as above, I recommend EUS, and if there are no concerning features on imaging or EUS, I then recommend annual surveillance.
While the patient is under surveillance, if there is more than minimal cyst growth, a change in cyst appearance, or development of any imaging high-risk feature, pancreatitis, new onset or worsening diabetes, or elevation of Ca19-9, I recommend EUS for further evaluation and consideration of surgery based on EUS findings. If an asymptomatic cyst <2 cm remains stable for 5 years, I offer patients the option to extend imaging to every 2 years, if they are comfortable. In my experience, though, many patients prefer to continue annual imaging. The American Gastroenterological Association guidelines promote stopping surveillance after 5 years of stability, however there are studies demonstrating development of malignancy in cysts that were initially stable over the first 5 years of surveillance. Therefore, I discuss with patients that it is reasonable to continue cyst surveillance indefinitely, until they would no longer be interested in pursuing treatment of any kind if a malignant lesion were to be identified.
There are two special groups of pancreas cyst patients who warrant specific attention. Patients who are at elevated risk of pancreas adenocarcinoma because of an associated genetic mutation or a family history of pancreatic cancer already may be undergoing annual pancreas cancer screening with either MRCP, EUS, or alternating MRCP and EUS. When these high-risk patients also have pancreas cysts, I utilize whichever strategy would image their pancreas most frequently and do not extend beyond 1-year intervals. Another special group is patients who have undergone partial pancreatectomy for IPMN. As discussed above, given the elevated risk of concurrent pancreas adenocarcinoma in IPMN patients, I recommend indefinite continued surveillance of the remaining pancreas parenchyma in these patients.
Given the prevalence of pancreas cysts, it certainly would be convenient if guidelines were straightforward enough for primary care physicians to manage pancreas cyst surveillance, as they do for breast cancer screening. However, the complexities of pancreas cysts necessitate the expertise of gastroenterologists and pancreas surgeons, and a multidisciplinary team approach is best where possible.
Dr. Khanna is chief, advanced endoscopy, Tisch Hospital; director, NYU Advanced Endoscopy Fellowship; assistant professor of medicine, NYU Langone Health. Email: [email protected]. There are no relevant conflicts to disclose.
References
Tanaka M et al. Pancreatology. 2017 Sep-Oct;17(5):738-75.
Sahora K et al. Eur J Surg Oncol. 2016 Feb;42(2):197-204.
Del Chiaro M et al. Gut. 2018 May;67(5):789-804
Vege SS et al. Gastroenterology. 2015 Apr;148(4):819-22
Petrone MC et al. Clin Transl Gastroenterol. 2018 Jun 13;9(6):158
Pancreas cysts: More is not necessarily better!
BY SANTHI SWAROOP VEGE, MD
Pancreas cysts (PC) are very common, incidental findings on cross-sectional imaging, performed for non–pancreas-related symptoms. The important issues in management of patients with PC in my practice are the prevalence, natural history, frequency of occurrence of high-grade dysplasia (HGD) and/or pancreatic cancer (PDAC), concerning clinical symptoms and imaging findings, indications for EUS and fine-needle aspiration cytology, ideal method and frequency of surveillance, indications for surgery (up front and during follow-up), follow-up after surgery, stopping surveillance, costs, and unintentional harms of management. Good population-based evidence regarding many of the issues described above does not exist, and all information is from selected clinic, radiology, EUS, and surgical cohorts (very important when trying to assess the publications). Cohort studies should start with all PC undergoing surveillance and assess various outcomes, rather than looking backward from EUS or surgical cohorts.
The 2015 American Gastroenterological Association guidelines on asymptomatic neoplastic pancreas cysts, which I coauthored, recommend, consistent with principles of High Value Care (minimal unintentional harms and cost effectiveness), that two of three high-risk features (mural nodule, cyst size greater than 3 cm, and dilated pancreatic duct) be present for EUS-guided fine-needle aspiration (EUS-FNA). By the same token, they advise surgery for those with two of three high-risk features and or concerning features on EUS and cytology. Finally, they suggest stopping surveillance at 5 years if there are no significant changes. Rigorous GRADE methodology along with systematic review of all relevant questions (rather than cohorts of 500 or fewer patients) formed the basis of the guidelines. Those meta-analyses showed that risk of PDAC in mural nodules, cyst size >3 cm, and dilated pancreatic duct, while elevated, still is very low in absolute terms. Less than 20% of resections for highly selected, high-risk cysts showed PDAC. The guidelines were met with a lot of resistance from several societies and physician groups. The recommendations for stopping surveillance after 5 years and no surveillance for absent or low-grade dysplasia after surgery are hotly contested, and these areas need larger, long-term studies.
The whole area of cyst fluid molecular markers that would suggest mucinous type (KRAS and GNAS mutations) and, more importantly, the presence or imminent development of PDAC (next-generation sequencing or NGS) is an exciting field. One sincerely hopes that there will be a breakthrough in this area to achieve the holy grail. Cost effectiveness studies demonstrate the futility of existing guidelines and favor a less intensive approach. Guidelines are only a general framework, and management of individual patients in the clinic is entirely at the discretion of the treating physician. One should make every attempt to detect advanced lesions in PC, but such effort should not subject a large majority of patients to unintentional harms by overtreatment and add further to the burgeoning health care costs in the country.
PC are extremely common (10% of all abdominal imaging), increase with age, are seen in as many as 40%-50% of MRI examinations for nonpancreatic indications, and most (>50%) are IPMNs. Most of the debate centers around the concerns of PDAC and/or HGD associated with mucinous cysts (MCN, IPMN, side-branch, main duct, or mixed).
The various guidelines by multiple societies differ in some aspects, such as in selection of patients based on clinical, laboratory, and imaging findings for up-front surgery or surveillance, the frequency of surveillance based on the size of the cyst and the presence of other concerning cyst features (usually with MRCP), the indications for EUS (both initial and subsequent), importance of the magnitude of growth (most IPMNs slowly grow over a period of time), indications for surgery during surveillance and postsurgery surveillance, and the decision to stop surveillance at some point in time. The literature is replete with small case series reporting a proportion of cancers detected and often ignoring the harms of surgery. Incidence of and mortality caused by PDAC are very low (about 1% for both) in a large national cohort of VA pancreatic cyst patients with long-term follow-up and other studies.
Marcov modeling suggests that none of the guidelines would lead to cost-effective care with low mortality because of overtreatment of low-risk lesions, and a specificity of 67% or more for PDAC/HGB is required. AGA guidelines came close to it but with low sensitivity. Monte Carlo modeling suggests that less intensive strategies, compared with more intensive, result in a similar number of deaths at a much lower cost. While molecular markers in PC fluid are reported to increase the specificity of PDAC/HGD to greater than 70%, it should be observed that such validation was done in a small percentage of patients who had both those markers and resection.
The costs of expensive procedures like EUS, MRI, and surgery, the 3% complication rate with EUS-FNA (primarily acute pancreatitis), and the 1% mortality and approximately 20%-30% morbidity with surgery (bleeding, infection, fistula) and postpancreatectomy diabetes of approximately 30% in the long run need special attention.
In conclusion, one could say pancreas cysts are extremely frequent, most of the neoplastic cysts are mucinous (IPMN and MCN) and slowly growing over time without an associated cancer, and the greatest need at this time is to identify the small proportion of such cysts with PDAC and/or HGD. Until such time, judicious selection of patients for surveillance and reasonable intervals of such surveillance with selective use of EUS will help identify patients requiring resection. In our enthusiasm to detect every possible pancreatic cancer, we should not ignore the unintentional outcomes of surgery to a large majority of patients who would never develop PDAC and the astronomical costs associated with such practice.
Dr. Vege is professor of medicine at the Mayo Clinic. He reported having no conflicts of interest regarding this article.
References
Vege SS et al. Gastroenterology. 2015;148:819-22.
Lobo JM et al. Surgery. 2020;168:601-9.
Lennon AM and Vege SS. Clin Gastroenterol Hepatol. 2022;20:1663-7.
Harris RP. Ann Intern Med. 2015;162:787-9.
Dear colleagues,
Pancreas cysts have become almost ubiquitous in this era of high-resolution cross-sectional imaging. They are a common GI consult with patients and providers worried about the potential risk of malignant transformation. Despite significant research over the past few decades, predicting the natural history of these cysts, especially the side-branch intraductal papillary mucinous neoplasms (IPMNs), remains difficult. There have been a variety of expert recommendations and guidelines, but heterogeneity exists in management especially regarding timing of endoscopic ultrasound, imaging surveillance, and cessation of surveillance. Some centers will present these cysts at multidisciplinary conferences, while others will follow general or local algorithms. In this issue of Perspectives, Dr. Lauren G. Khanna, assistant professor of medicine at NYU Langone Health, New York, and Dr. Santhi Vege, professor of medicine at the Mayo Clinic, Rochester, Minn., present updated and differing approaches to managing these cysts. Which side of the debate are you on? We welcome your thoughts, questions and input– share with us on Twitter @AGA_GIHN
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Conn., and chief of endoscopy at West Haven (Conn.) VA Medical Center. He is an associate editor for GI & Hepatology News.
Continuing pancreas cyst surveillance indefinitely is reasonable
BY LAUREN G. KHANNA, MD, MS
Pancreas cysts remain a clinical challenge. The true incidence of pancreas cysts is unknown, but from MRI and autopsy series, may be up to 50%. Patients presenting with a pancreas cyst often have significant anxiety about their risk of pancreas cancer. We as a medical community initially did too; but over the past few decades as we have gathered more data, we have become more comfortable observing many pancreas cysts. Yet our recommendations for how, how often, and for how long to evaluate pancreas cysts are still very much under debate; there are multiple guidelines with discordant recommendations. In this article, I will discuss my approach to patients with a pancreas cyst.
At the first evaluation, I review available imaging to see if there are characteristic features to determine the type of pancreas cyst: IPMN (including main duct, branch duct, or mixed type), serous cystic neoplasm (SCA), mucinous cystic neoplasm (MCN), solid pseudopapillary neoplasm (SPN), cystic neuroendocrine tumor (NET), or pseudocyst. I also review symptoms, including abdominal pain, weight loss, history of pancreatitis, and onset of diabetes, and check hemoglobin A1c and Ca19-9. I often recommend magnetic resonance cholangiopancreatography (MRCP) if it has not already been obtained and is feasible (that is, if a patient does not have severe claustrophobia or a medical device incompatible with MRI). If a patient is not a candidate for treatment should a pancreatic malignancy be identified, because of age, comorbidities, or preference, I recommend no further evaluation.
Where cyst type remains unclear despite MRCP, and for cysts over 2 cm, I recommend endoscopic ultrasound (EUS) for fluid sampling to assist in determining cyst type and to rule out any other high-risk features. In accordance with international guidelines, if a patient has any concerning imaging features, including main pancreatic duct dilation >5 mm, solid component or mural nodule, or thickened or enhancing duct walls, regardless of cyst size, I recommend EUS to assess for and biopsy any solid component and to sample cyst fluid to examine for dysplasia. Given the lower sensitivity of CT for high-risk features, if MRCP is not feasible, for cysts 1-2 cm, I recommend EUS for better evaluation.
If a cyst is determined to be a cystic NET; main duct or mixed-type IPMN; MCN; or SPN; or a branch duct IPMN with mural nodule, high-grade dysplasia, or adenocarcinoma, and the patient is a surgical candidate, I refer the patient for surgical evaluation. If a cyst is determined to be an SCA, the malignant potential is minimal, and patients do not require follow-up. Patients with a pseudocyst are managed according to their clinical scenario.
Many patients have a proven or suspected branch duct IPMN, an indeterminate cyst, or multiple cysts. Cyst management during surveillance is then determined by the size of the largest cyst and stability of the cyst(s). Of note, patients with an IPMN also have been shown to have an elevated risk of concurrent pancreas adenocarcinoma, which I believe is one of the strongest arguments for heightened surveillance of the entire pancreas in pancreas cyst patients. EUS in particular can identify small or subtle lesions that are not detected by cross-sectional imaging.
If a patient has no prior imaging, in accordance with international and European guidelines, I recommend the first surveillance MRCP at a 6-month interval for cysts <2 cm, which may offer the opportunity to identify rapidly progressing cysts. If a patient has previous imaging available demonstrating stability, I recommend surveillance on an annual basis for cysts <2 cm. For patients with a cyst >2 cm, as above, I recommend EUS, and if there are no concerning features on imaging or EUS, I then recommend annual surveillance.
While the patient is under surveillance, if there is more than minimal cyst growth, a change in cyst appearance, or development of any imaging high-risk feature, pancreatitis, new onset or worsening diabetes, or elevation of Ca19-9, I recommend EUS for further evaluation and consideration of surgery based on EUS findings. If an asymptomatic cyst <2 cm remains stable for 5 years, I offer patients the option to extend imaging to every 2 years, if they are comfortable. In my experience, though, many patients prefer to continue annual imaging. The American Gastroenterological Association guidelines promote stopping surveillance after 5 years of stability, however there are studies demonstrating development of malignancy in cysts that were initially stable over the first 5 years of surveillance. Therefore, I discuss with patients that it is reasonable to continue cyst surveillance indefinitely, until they would no longer be interested in pursuing treatment of any kind if a malignant lesion were to be identified.
There are two special groups of pancreas cyst patients who warrant specific attention. Patients who are at elevated risk of pancreas adenocarcinoma because of an associated genetic mutation or a family history of pancreatic cancer already may be undergoing annual pancreas cancer screening with either MRCP, EUS, or alternating MRCP and EUS. When these high-risk patients also have pancreas cysts, I utilize whichever strategy would image their pancreas most frequently and do not extend beyond 1-year intervals. Another special group is patients who have undergone partial pancreatectomy for IPMN. As discussed above, given the elevated risk of concurrent pancreas adenocarcinoma in IPMN patients, I recommend indefinite continued surveillance of the remaining pancreas parenchyma in these patients.
Given the prevalence of pancreas cysts, it certainly would be convenient if guidelines were straightforward enough for primary care physicians to manage pancreas cyst surveillance, as they do for breast cancer screening. However, the complexities of pancreas cysts necessitate the expertise of gastroenterologists and pancreas surgeons, and a multidisciplinary team approach is best where possible.
Dr. Khanna is chief, advanced endoscopy, Tisch Hospital; director, NYU Advanced Endoscopy Fellowship; assistant professor of medicine, NYU Langone Health. Email: [email protected]. There are no relevant conflicts to disclose.
References
Tanaka M et al. Pancreatology. 2017 Sep-Oct;17(5):738-75.
Sahora K et al. Eur J Surg Oncol. 2016 Feb;42(2):197-204.
Del Chiaro M et al. Gut. 2018 May;67(5):789-804
Vege SS et al. Gastroenterology. 2015 Apr;148(4):819-22
Petrone MC et al. Clin Transl Gastroenterol. 2018 Jun 13;9(6):158
Pancreas cysts: More is not necessarily better!
BY SANTHI SWAROOP VEGE, MD
Pancreas cysts (PC) are very common, incidental findings on cross-sectional imaging, performed for non–pancreas-related symptoms. The important issues in management of patients with PC in my practice are the prevalence, natural history, frequency of occurrence of high-grade dysplasia (HGD) and/or pancreatic cancer (PDAC), concerning clinical symptoms and imaging findings, indications for EUS and fine-needle aspiration cytology, ideal method and frequency of surveillance, indications for surgery (up front and during follow-up), follow-up after surgery, stopping surveillance, costs, and unintentional harms of management. Good population-based evidence regarding many of the issues described above does not exist, and all information is from selected clinic, radiology, EUS, and surgical cohorts (very important when trying to assess the publications). Cohort studies should start with all PC undergoing surveillance and assess various outcomes, rather than looking backward from EUS or surgical cohorts.
The 2015 American Gastroenterological Association guidelines on asymptomatic neoplastic pancreas cysts, which I coauthored, recommend, consistent with principles of High Value Care (minimal unintentional harms and cost effectiveness), that two of three high-risk features (mural nodule, cyst size greater than 3 cm, and dilated pancreatic duct) be present for EUS-guided fine-needle aspiration (EUS-FNA). By the same token, they advise surgery for those with two of three high-risk features and or concerning features on EUS and cytology. Finally, they suggest stopping surveillance at 5 years if there are no significant changes. Rigorous GRADE methodology along with systematic review of all relevant questions (rather than cohorts of 500 or fewer patients) formed the basis of the guidelines. Those meta-analyses showed that risk of PDAC in mural nodules, cyst size >3 cm, and dilated pancreatic duct, while elevated, still is very low in absolute terms. Less than 20% of resections for highly selected, high-risk cysts showed PDAC. The guidelines were met with a lot of resistance from several societies and physician groups. The recommendations for stopping surveillance after 5 years and no surveillance for absent or low-grade dysplasia after surgery are hotly contested, and these areas need larger, long-term studies.
The whole area of cyst fluid molecular markers that would suggest mucinous type (KRAS and GNAS mutations) and, more importantly, the presence or imminent development of PDAC (next-generation sequencing or NGS) is an exciting field. One sincerely hopes that there will be a breakthrough in this area to achieve the holy grail. Cost effectiveness studies demonstrate the futility of existing guidelines and favor a less intensive approach. Guidelines are only a general framework, and management of individual patients in the clinic is entirely at the discretion of the treating physician. One should make every attempt to detect advanced lesions in PC, but such effort should not subject a large majority of patients to unintentional harms by overtreatment and add further to the burgeoning health care costs in the country.
PC are extremely common (10% of all abdominal imaging), increase with age, are seen in as many as 40%-50% of MRI examinations for nonpancreatic indications, and most (>50%) are IPMNs. Most of the debate centers around the concerns of PDAC and/or HGD associated with mucinous cysts (MCN, IPMN, side-branch, main duct, or mixed).
The various guidelines by multiple societies differ in some aspects, such as in selection of patients based on clinical, laboratory, and imaging findings for up-front surgery or surveillance, the frequency of surveillance based on the size of the cyst and the presence of other concerning cyst features (usually with MRCP), the indications for EUS (both initial and subsequent), importance of the magnitude of growth (most IPMNs slowly grow over a period of time), indications for surgery during surveillance and postsurgery surveillance, and the decision to stop surveillance at some point in time. The literature is replete with small case series reporting a proportion of cancers detected and often ignoring the harms of surgery. Incidence of and mortality caused by PDAC are very low (about 1% for both) in a large national cohort of VA pancreatic cyst patients with long-term follow-up and other studies.
Marcov modeling suggests that none of the guidelines would lead to cost-effective care with low mortality because of overtreatment of low-risk lesions, and a specificity of 67% or more for PDAC/HGB is required. AGA guidelines came close to it but with low sensitivity. Monte Carlo modeling suggests that less intensive strategies, compared with more intensive, result in a similar number of deaths at a much lower cost. While molecular markers in PC fluid are reported to increase the specificity of PDAC/HGD to greater than 70%, it should be observed that such validation was done in a small percentage of patients who had both those markers and resection.
The costs of expensive procedures like EUS, MRI, and surgery, the 3% complication rate with EUS-FNA (primarily acute pancreatitis), and the 1% mortality and approximately 20%-30% morbidity with surgery (bleeding, infection, fistula) and postpancreatectomy diabetes of approximately 30% in the long run need special attention.
In conclusion, one could say pancreas cysts are extremely frequent, most of the neoplastic cysts are mucinous (IPMN and MCN) and slowly growing over time without an associated cancer, and the greatest need at this time is to identify the small proportion of such cysts with PDAC and/or HGD. Until such time, judicious selection of patients for surveillance and reasonable intervals of such surveillance with selective use of EUS will help identify patients requiring resection. In our enthusiasm to detect every possible pancreatic cancer, we should not ignore the unintentional outcomes of surgery to a large majority of patients who would never develop PDAC and the astronomical costs associated with such practice.
Dr. Vege is professor of medicine at the Mayo Clinic. He reported having no conflicts of interest regarding this article.
References
Vege SS et al. Gastroenterology. 2015;148:819-22.
Lobo JM et al. Surgery. 2020;168:601-9.
Lennon AM and Vege SS. Clin Gastroenterol Hepatol. 2022;20:1663-7.
Harris RP. Ann Intern Med. 2015;162:787-9.
Dear colleagues,
Pancreas cysts have become almost ubiquitous in this era of high-resolution cross-sectional imaging. They are a common GI consult with patients and providers worried about the potential risk of malignant transformation. Despite significant research over the past few decades, predicting the natural history of these cysts, especially the side-branch intraductal papillary mucinous neoplasms (IPMNs), remains difficult. There have been a variety of expert recommendations and guidelines, but heterogeneity exists in management especially regarding timing of endoscopic ultrasound, imaging surveillance, and cessation of surveillance. Some centers will present these cysts at multidisciplinary conferences, while others will follow general or local algorithms. In this issue of Perspectives, Dr. Lauren G. Khanna, assistant professor of medicine at NYU Langone Health, New York, and Dr. Santhi Vege, professor of medicine at the Mayo Clinic, Rochester, Minn., present updated and differing approaches to managing these cysts. Which side of the debate are you on? We welcome your thoughts, questions and input– share with us on Twitter @AGA_GIHN
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Conn., and chief of endoscopy at West Haven (Conn.) VA Medical Center. He is an associate editor for GI & Hepatology News.
Continuing pancreas cyst surveillance indefinitely is reasonable
BY LAUREN G. KHANNA, MD, MS
Pancreas cysts remain a clinical challenge. The true incidence of pancreas cysts is unknown, but from MRI and autopsy series, may be up to 50%. Patients presenting with a pancreas cyst often have significant anxiety about their risk of pancreas cancer. We as a medical community initially did too; but over the past few decades as we have gathered more data, we have become more comfortable observing many pancreas cysts. Yet our recommendations for how, how often, and for how long to evaluate pancreas cysts are still very much under debate; there are multiple guidelines with discordant recommendations. In this article, I will discuss my approach to patients with a pancreas cyst.
At the first evaluation, I review available imaging to see if there are characteristic features to determine the type of pancreas cyst: IPMN (including main duct, branch duct, or mixed type), serous cystic neoplasm (SCA), mucinous cystic neoplasm (MCN), solid pseudopapillary neoplasm (SPN), cystic neuroendocrine tumor (NET), or pseudocyst. I also review symptoms, including abdominal pain, weight loss, history of pancreatitis, and onset of diabetes, and check hemoglobin A1c and Ca19-9. I often recommend magnetic resonance cholangiopancreatography (MRCP) if it has not already been obtained and is feasible (that is, if a patient does not have severe claustrophobia or a medical device incompatible with MRI). If a patient is not a candidate for treatment should a pancreatic malignancy be identified, because of age, comorbidities, or preference, I recommend no further evaluation.
Where cyst type remains unclear despite MRCP, and for cysts over 2 cm, I recommend endoscopic ultrasound (EUS) for fluid sampling to assist in determining cyst type and to rule out any other high-risk features. In accordance with international guidelines, if a patient has any concerning imaging features, including main pancreatic duct dilation >5 mm, solid component or mural nodule, or thickened or enhancing duct walls, regardless of cyst size, I recommend EUS to assess for and biopsy any solid component and to sample cyst fluid to examine for dysplasia. Given the lower sensitivity of CT for high-risk features, if MRCP is not feasible, for cysts 1-2 cm, I recommend EUS for better evaluation.
If a cyst is determined to be a cystic NET; main duct or mixed-type IPMN; MCN; or SPN; or a branch duct IPMN with mural nodule, high-grade dysplasia, or adenocarcinoma, and the patient is a surgical candidate, I refer the patient for surgical evaluation. If a cyst is determined to be an SCA, the malignant potential is minimal, and patients do not require follow-up. Patients with a pseudocyst are managed according to their clinical scenario.
Many patients have a proven or suspected branch duct IPMN, an indeterminate cyst, or multiple cysts. Cyst management during surveillance is then determined by the size of the largest cyst and stability of the cyst(s). Of note, patients with an IPMN also have been shown to have an elevated risk of concurrent pancreas adenocarcinoma, which I believe is one of the strongest arguments for heightened surveillance of the entire pancreas in pancreas cyst patients. EUS in particular can identify small or subtle lesions that are not detected by cross-sectional imaging.
If a patient has no prior imaging, in accordance with international and European guidelines, I recommend the first surveillance MRCP at a 6-month interval for cysts <2 cm, which may offer the opportunity to identify rapidly progressing cysts. If a patient has previous imaging available demonstrating stability, I recommend surveillance on an annual basis for cysts <2 cm. For patients with a cyst >2 cm, as above, I recommend EUS, and if there are no concerning features on imaging or EUS, I then recommend annual surveillance.
While the patient is under surveillance, if there is more than minimal cyst growth, a change in cyst appearance, or development of any imaging high-risk feature, pancreatitis, new onset or worsening diabetes, or elevation of Ca19-9, I recommend EUS for further evaluation and consideration of surgery based on EUS findings. If an asymptomatic cyst <2 cm remains stable for 5 years, I offer patients the option to extend imaging to every 2 years, if they are comfortable. In my experience, though, many patients prefer to continue annual imaging. The American Gastroenterological Association guidelines promote stopping surveillance after 5 years of stability, however there are studies demonstrating development of malignancy in cysts that were initially stable over the first 5 years of surveillance. Therefore, I discuss with patients that it is reasonable to continue cyst surveillance indefinitely, until they would no longer be interested in pursuing treatment of any kind if a malignant lesion were to be identified.
There are two special groups of pancreas cyst patients who warrant specific attention. Patients who are at elevated risk of pancreas adenocarcinoma because of an associated genetic mutation or a family history of pancreatic cancer already may be undergoing annual pancreas cancer screening with either MRCP, EUS, or alternating MRCP and EUS. When these high-risk patients also have pancreas cysts, I utilize whichever strategy would image their pancreas most frequently and do not extend beyond 1-year intervals. Another special group is patients who have undergone partial pancreatectomy for IPMN. As discussed above, given the elevated risk of concurrent pancreas adenocarcinoma in IPMN patients, I recommend indefinite continued surveillance of the remaining pancreas parenchyma in these patients.
Given the prevalence of pancreas cysts, it certainly would be convenient if guidelines were straightforward enough for primary care physicians to manage pancreas cyst surveillance, as they do for breast cancer screening. However, the complexities of pancreas cysts necessitate the expertise of gastroenterologists and pancreas surgeons, and a multidisciplinary team approach is best where possible.
Dr. Khanna is chief, advanced endoscopy, Tisch Hospital; director, NYU Advanced Endoscopy Fellowship; assistant professor of medicine, NYU Langone Health. Email: [email protected]. There are no relevant conflicts to disclose.
References
Tanaka M et al. Pancreatology. 2017 Sep-Oct;17(5):738-75.
Sahora K et al. Eur J Surg Oncol. 2016 Feb;42(2):197-204.
Del Chiaro M et al. Gut. 2018 May;67(5):789-804
Vege SS et al. Gastroenterology. 2015 Apr;148(4):819-22
Petrone MC et al. Clin Transl Gastroenterol. 2018 Jun 13;9(6):158
Pancreas cysts: More is not necessarily better!
BY SANTHI SWAROOP VEGE, MD
Pancreas cysts (PC) are very common, incidental findings on cross-sectional imaging, performed for non–pancreas-related symptoms. The important issues in management of patients with PC in my practice are the prevalence, natural history, frequency of occurrence of high-grade dysplasia (HGD) and/or pancreatic cancer (PDAC), concerning clinical symptoms and imaging findings, indications for EUS and fine-needle aspiration cytology, ideal method and frequency of surveillance, indications for surgery (up front and during follow-up), follow-up after surgery, stopping surveillance, costs, and unintentional harms of management. Good population-based evidence regarding many of the issues described above does not exist, and all information is from selected clinic, radiology, EUS, and surgical cohorts (very important when trying to assess the publications). Cohort studies should start with all PC undergoing surveillance and assess various outcomes, rather than looking backward from EUS or surgical cohorts.
The 2015 American Gastroenterological Association guidelines on asymptomatic neoplastic pancreas cysts, which I coauthored, recommend, consistent with principles of High Value Care (minimal unintentional harms and cost effectiveness), that two of three high-risk features (mural nodule, cyst size greater than 3 cm, and dilated pancreatic duct) be present for EUS-guided fine-needle aspiration (EUS-FNA). By the same token, they advise surgery for those with two of three high-risk features and or concerning features on EUS and cytology. Finally, they suggest stopping surveillance at 5 years if there are no significant changes. Rigorous GRADE methodology along with systematic review of all relevant questions (rather than cohorts of 500 or fewer patients) formed the basis of the guidelines. Those meta-analyses showed that risk of PDAC in mural nodules, cyst size >3 cm, and dilated pancreatic duct, while elevated, still is very low in absolute terms. Less than 20% of resections for highly selected, high-risk cysts showed PDAC. The guidelines were met with a lot of resistance from several societies and physician groups. The recommendations for stopping surveillance after 5 years and no surveillance for absent or low-grade dysplasia after surgery are hotly contested, and these areas need larger, long-term studies.
The whole area of cyst fluid molecular markers that would suggest mucinous type (KRAS and GNAS mutations) and, more importantly, the presence or imminent development of PDAC (next-generation sequencing or NGS) is an exciting field. One sincerely hopes that there will be a breakthrough in this area to achieve the holy grail. Cost effectiveness studies demonstrate the futility of existing guidelines and favor a less intensive approach. Guidelines are only a general framework, and management of individual patients in the clinic is entirely at the discretion of the treating physician. One should make every attempt to detect advanced lesions in PC, but such effort should not subject a large majority of patients to unintentional harms by overtreatment and add further to the burgeoning health care costs in the country.
PC are extremely common (10% of all abdominal imaging), increase with age, are seen in as many as 40%-50% of MRI examinations for nonpancreatic indications, and most (>50%) are IPMNs. Most of the debate centers around the concerns of PDAC and/or HGD associated with mucinous cysts (MCN, IPMN, side-branch, main duct, or mixed).
The various guidelines by multiple societies differ in some aspects, such as in selection of patients based on clinical, laboratory, and imaging findings for up-front surgery or surveillance, the frequency of surveillance based on the size of the cyst and the presence of other concerning cyst features (usually with MRCP), the indications for EUS (both initial and subsequent), importance of the magnitude of growth (most IPMNs slowly grow over a period of time), indications for surgery during surveillance and postsurgery surveillance, and the decision to stop surveillance at some point in time. The literature is replete with small case series reporting a proportion of cancers detected and often ignoring the harms of surgery. Incidence of and mortality caused by PDAC are very low (about 1% for both) in a large national cohort of VA pancreatic cyst patients with long-term follow-up and other studies.
Marcov modeling suggests that none of the guidelines would lead to cost-effective care with low mortality because of overtreatment of low-risk lesions, and a specificity of 67% or more for PDAC/HGB is required. AGA guidelines came close to it but with low sensitivity. Monte Carlo modeling suggests that less intensive strategies, compared with more intensive, result in a similar number of deaths at a much lower cost. While molecular markers in PC fluid are reported to increase the specificity of PDAC/HGD to greater than 70%, it should be observed that such validation was done in a small percentage of patients who had both those markers and resection.
The costs of expensive procedures like EUS, MRI, and surgery, the 3% complication rate with EUS-FNA (primarily acute pancreatitis), and the 1% mortality and approximately 20%-30% morbidity with surgery (bleeding, infection, fistula) and postpancreatectomy diabetes of approximately 30% in the long run need special attention.
In conclusion, one could say pancreas cysts are extremely frequent, most of the neoplastic cysts are mucinous (IPMN and MCN) and slowly growing over time without an associated cancer, and the greatest need at this time is to identify the small proportion of such cysts with PDAC and/or HGD. Until such time, judicious selection of patients for surveillance and reasonable intervals of such surveillance with selective use of EUS will help identify patients requiring resection. In our enthusiasm to detect every possible pancreatic cancer, we should not ignore the unintentional outcomes of surgery to a large majority of patients who would never develop PDAC and the astronomical costs associated with such practice.
Dr. Vege is professor of medicine at the Mayo Clinic. He reported having no conflicts of interest regarding this article.
References
Vege SS et al. Gastroenterology. 2015;148:819-22.
Lobo JM et al. Surgery. 2020;168:601-9.
Lennon AM and Vege SS. Clin Gastroenterol Hepatol. 2022;20:1663-7.
Harris RP. Ann Intern Med. 2015;162:787-9.
Understanding clinic-reported IVF success rates
The field of assisted reproductive technologies (ART) continues to evolve from its first successful birth in 1978 in England, and then in 1981 in the United States. Over the last 6 years, the total number of cycles in the U.S. has increased by 44% to nearly 370,000.
SART membership consists of more than 350 clinics throughout the United States, representing 80% of ART clinics. Over 95% of ART cycles in 2021 in the United States were performed in SART-member clinics.
SART is an invaluable resource for both patients and physicians. Their website includes a “Predict My Success” calculator that allows patients and physicians to enter individualized data to calculate the chance of having a baby over one or more complete cycles of IVF. To help us understand the pregnancy outcome data from ART – cycles per clinic along with national results – I posed the questions below to Amy Sparks, PhD, HCLD, director of the IVF and Andrology Laboratories and the Center for Advanced Reproductive Care at University of Iowa Hospitals and Clinics, Iowa City. Dr. Sparks is past president of SART and former chairperson of the SART Registry committee when the current Clinic Summary Report format was initially released.
Question: The Fertility Clinic Success Rate and Certification Act (FCSRCA) of 1992 mandated that all ART clinics report success rate data to the federal government, through the Centers for Disease Control and Prevention, in a standardized manner. As ART is the only field in medicine to be required to annually report their patient outcomes, that is, all initiated cycles and live births, why do you believe this law was enacted and is limited to reproductive medicine?
Answer: The FCSRCA of 1992 was enacted in response to the lack of open and reliable pregnancy success rate information for patients seeking infertility care using assisted reproductive technologies. Success rates of 25%-50% were being advertised by independent clinics when, nationally, fewer than 15% of ART procedures led to live births. The Federal Trade Commission said such claims were deceptive and filed charges against five clinics, saying they misrepresented their success in helping women become pregnant. The government won one case by court order and the other four cases were settled out of court.
This field of medicine was in the spotlight as the majority of patients lacked insurance coverage for their ART cycles, and there was a strong desire to protect consumers paying out of pocket for relatively low success. Recognizing that the FTC’s mission is to ensure truth in advertising and not regulate medical care, Congress passed the FCSRCA, mandating that all centers providing ART services report all initiated cycles and their outcomes. The CDC was appointed as the agency responsible for collecting cycle data and reporting outcomes. Centers not reporting their cycles are listed as nonreporting centers.
This act also established standards for accreditation of embryology laboratories including personnel and traditional clinical laboratory management requirements. These standards serve as the foundation for embryology laboratory accrediting agencies.
Q: Why have live-birth rates on SART appeared to be focused on “per IVF cycle” as opposed to the CDC reporting of live births “per embryo transfer?”
A: An ART cycle “start” is defined as the initiation of ovarian stimulation with medication that may or may not include administration of exogenous gonadotropins, followed by oocyte retrieval and embryo transfer. Not every patient beginning a cycle will undergo an oocyte retrieval and not all patients who undergo oocyte retrieval have an embryo transfer. The live-birth rates (LBR) for each of these steps of progression in the ART process are available in the SART and CDC reports.
In 2016, SART recognized that practices were foregoing fresh embryo transfer after oocyte retrieval, opting to cryopreserve all embryos to either accommodate genetic testing of the embryos prior to transfer or to avoid embryo transfer to an unfavorable uterine environment. In response to changes in practice and in an effort to deemphasize live birth per transfer, thereby alleviating a potential motivator or pressure for practitioners to transfer multiple embryos, SART moved to a report that displays the cumulative live-birth rate per cycle start for oocyte retrieval. The cumulative live-birth rate per cycle start for oocyte retrieval is the chance of live birth from transfers of embryos derived from the oocyte retrieval and performed within 1 year of the oocyte retrieval.
This change in reporting further reduced the pressure to transfer multiple embryos and encouraged elective, single-embryo transfer. The outcome per transfer is no longer the report’s primary focus.
Q: The latest pregnancy outcomes statistics are from the year 2020 and are finalized by the CDC. Why does the SART website have this same year labeled “preliminary” outcomes?
A: Shortly after the 2016 SART report change, the CDC made similar changes to their report. The difference is that SART provides a “preliminary” report of outcomes within the year of the cycle start for oocyte retrieval. The cumulative outcome is not “finalized” until the following year as transfers may be performed as late as 12 months after the oocyte retrieval.
SART has opted to report both the “preliminary” or interim outcome and the “final” outcome a year later. The CDC has opted to limit their report to “final” outcomes. I’m happy to report that SART recently released the final report for 2021 cycles.
Q: Have national success rates in the United States continued to rise or have they plateaued?
A: It appears that success rates have plateaued; however, we find ourselves at another point where practice patterns and patients’ approach to using ART for family building have changed.
Recognizing the impact of maternal aging on reproductive potential, patients are opting to undergo multiple ART cycles to cryopreserve embryos for family building before they attempt to get pregnant. This family-building path reduces the value of measuring the LBR per cycle start as we may not know the outcome for many years. SART leaders are deliberating intently as to how to best represent this growing patient population in outcome reporting.
Q: Can you comment on the reduction of multiple gestations with the increasing use of single-embryo transfer?
A: The reduction in emphasis on live births per transfer, emphasis on singleton live-birth rates in both the SART and CDC reports, and American Society for Reproductive Medicine practice committee guidelines strongly supporting single embryo transfer have significantly reduced the rate of multiple gestations.
A decade ago, only a third of the transfers were single-embryo transfers and over 25% of live births resulted in a multiple birth. Today, the majority of embryo transfers are elective, single-embryo transfers, and the multiple birth rate has been reduced by nearly 80%. In 2020, 93% of live births from IVF were singletons.
Q: SART offers an online IVF calculator so both patients and physicians can plug in data for an approximate cumulative success rate for up to three IVF cycles. The calculator pools data from all U.S.-reporting IVF centers. Can you explain what an “IVF cycle” is and what patient information is required? Why do success rates increase over time?
A: Each “IVF cycle” is a cycle start for an oocyte retrieval and all transfers of embryos from that cycle within a year of the oocyte retrieval. If the first cycle and subsequent transfers do not lead to a live birth, patients still have a chance to achieve a live birth with a second or third cycle. The success rate increases over time as it reflects the chance of success for a population of patients, with some achieving a live birth after the first cycle and additional patients who achieve success following their third cycle.
Q: The SART IVF calculator can be used with no prior IVF cycles or following an unsuccessful cycle. Are there data to support an estimation of outcome following two or even more unsuccessful cycles?
A: The variables in the SART IVF calculator are based upon the cycle-specific data from patients seeking care at SART member clinics. The current predictor was built with data from cycles performed in 2015-2016. SART is adjusting the predictor and developing a calculator that will be routinely updated, accordingly.
Q: Only approximately 40% of states have some form of infertility coverage law in place; however the number of IVF cycles in the United States continues to increase on an annual basis. What do you think are the driving factors behind this?
A: Advocacy efforts to improve patients’ access to infertility care have included giving patients tools to encourage their employers to include infertility care in their health care benefits package. More recently, the “Great Resignation” has led to the “Great Recruitment” and employers are recognizing that the addition of infertility care to health care benefits is a powerful recruitment tool.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
The field of assisted reproductive technologies (ART) continues to evolve from its first successful birth in 1978 in England, and then in 1981 in the United States. Over the last 6 years, the total number of cycles in the U.S. has increased by 44% to nearly 370,000.
SART membership consists of more than 350 clinics throughout the United States, representing 80% of ART clinics. Over 95% of ART cycles in 2021 in the United States were performed in SART-member clinics.
SART is an invaluable resource for both patients and physicians. Their website includes a “Predict My Success” calculator that allows patients and physicians to enter individualized data to calculate the chance of having a baby over one or more complete cycles of IVF. To help us understand the pregnancy outcome data from ART – cycles per clinic along with national results – I posed the questions below to Amy Sparks, PhD, HCLD, director of the IVF and Andrology Laboratories and the Center for Advanced Reproductive Care at University of Iowa Hospitals and Clinics, Iowa City. Dr. Sparks is past president of SART and former chairperson of the SART Registry committee when the current Clinic Summary Report format was initially released.
Question: The Fertility Clinic Success Rate and Certification Act (FCSRCA) of 1992 mandated that all ART clinics report success rate data to the federal government, through the Centers for Disease Control and Prevention, in a standardized manner. As ART is the only field in medicine to be required to annually report their patient outcomes, that is, all initiated cycles and live births, why do you believe this law was enacted and is limited to reproductive medicine?
Answer: The FCSRCA of 1992 was enacted in response to the lack of open and reliable pregnancy success rate information for patients seeking infertility care using assisted reproductive technologies. Success rates of 25%-50% were being advertised by independent clinics when, nationally, fewer than 15% of ART procedures led to live births. The Federal Trade Commission said such claims were deceptive and filed charges against five clinics, saying they misrepresented their success in helping women become pregnant. The government won one case by court order and the other four cases were settled out of court.
This field of medicine was in the spotlight as the majority of patients lacked insurance coverage for their ART cycles, and there was a strong desire to protect consumers paying out of pocket for relatively low success. Recognizing that the FTC’s mission is to ensure truth in advertising and not regulate medical care, Congress passed the FCSRCA, mandating that all centers providing ART services report all initiated cycles and their outcomes. The CDC was appointed as the agency responsible for collecting cycle data and reporting outcomes. Centers not reporting their cycles are listed as nonreporting centers.
This act also established standards for accreditation of embryology laboratories including personnel and traditional clinical laboratory management requirements. These standards serve as the foundation for embryology laboratory accrediting agencies.
Q: Why have live-birth rates on SART appeared to be focused on “per IVF cycle” as opposed to the CDC reporting of live births “per embryo transfer?”
A: An ART cycle “start” is defined as the initiation of ovarian stimulation with medication that may or may not include administration of exogenous gonadotropins, followed by oocyte retrieval and embryo transfer. Not every patient beginning a cycle will undergo an oocyte retrieval and not all patients who undergo oocyte retrieval have an embryo transfer. The live-birth rates (LBR) for each of these steps of progression in the ART process are available in the SART and CDC reports.
In 2016, SART recognized that practices were foregoing fresh embryo transfer after oocyte retrieval, opting to cryopreserve all embryos to either accommodate genetic testing of the embryos prior to transfer or to avoid embryo transfer to an unfavorable uterine environment. In response to changes in practice and in an effort to deemphasize live birth per transfer, thereby alleviating a potential motivator or pressure for practitioners to transfer multiple embryos, SART moved to a report that displays the cumulative live-birth rate per cycle start for oocyte retrieval. The cumulative live-birth rate per cycle start for oocyte retrieval is the chance of live birth from transfers of embryos derived from the oocyte retrieval and performed within 1 year of the oocyte retrieval.
This change in reporting further reduced the pressure to transfer multiple embryos and encouraged elective, single-embryo transfer. The outcome per transfer is no longer the report’s primary focus.
Q: The latest pregnancy outcomes statistics are from the year 2020 and are finalized by the CDC. Why does the SART website have this same year labeled “preliminary” outcomes?
A: Shortly after the 2016 SART report change, the CDC made similar changes to their report. The difference is that SART provides a “preliminary” report of outcomes within the year of the cycle start for oocyte retrieval. The cumulative outcome is not “finalized” until the following year as transfers may be performed as late as 12 months after the oocyte retrieval.
SART has opted to report both the “preliminary” or interim outcome and the “final” outcome a year later. The CDC has opted to limit their report to “final” outcomes. I’m happy to report that SART recently released the final report for 2021 cycles.
Q: Have national success rates in the United States continued to rise or have they plateaued?
A: It appears that success rates have plateaued; however, we find ourselves at another point where practice patterns and patients’ approach to using ART for family building have changed.
Recognizing the impact of maternal aging on reproductive potential, patients are opting to undergo multiple ART cycles to cryopreserve embryos for family building before they attempt to get pregnant. This family-building path reduces the value of measuring the LBR per cycle start as we may not know the outcome for many years. SART leaders are deliberating intently as to how to best represent this growing patient population in outcome reporting.
Q: Can you comment on the reduction of multiple gestations with the increasing use of single-embryo transfer?
A: The reduction in emphasis on live births per transfer, emphasis on singleton live-birth rates in both the SART and CDC reports, and American Society for Reproductive Medicine practice committee guidelines strongly supporting single embryo transfer have significantly reduced the rate of multiple gestations.
A decade ago, only a third of the transfers were single-embryo transfers and over 25% of live births resulted in a multiple birth. Today, the majority of embryo transfers are elective, single-embryo transfers, and the multiple birth rate has been reduced by nearly 80%. In 2020, 93% of live births from IVF were singletons.
Q: SART offers an online IVF calculator so both patients and physicians can plug in data for an approximate cumulative success rate for up to three IVF cycles. The calculator pools data from all U.S.-reporting IVF centers. Can you explain what an “IVF cycle” is and what patient information is required? Why do success rates increase over time?
A: Each “IVF cycle” is a cycle start for an oocyte retrieval and all transfers of embryos from that cycle within a year of the oocyte retrieval. If the first cycle and subsequent transfers do not lead to a live birth, patients still have a chance to achieve a live birth with a second or third cycle. The success rate increases over time as it reflects the chance of success for a population of patients, with some achieving a live birth after the first cycle and additional patients who achieve success following their third cycle.
Q: The SART IVF calculator can be used with no prior IVF cycles or following an unsuccessful cycle. Are there data to support an estimation of outcome following two or even more unsuccessful cycles?
A: The variables in the SART IVF calculator are based upon the cycle-specific data from patients seeking care at SART member clinics. The current predictor was built with data from cycles performed in 2015-2016. SART is adjusting the predictor and developing a calculator that will be routinely updated, accordingly.
Q: Only approximately 40% of states have some form of infertility coverage law in place; however the number of IVF cycles in the United States continues to increase on an annual basis. What do you think are the driving factors behind this?
A: Advocacy efforts to improve patients’ access to infertility care have included giving patients tools to encourage their employers to include infertility care in their health care benefits package. More recently, the “Great Resignation” has led to the “Great Recruitment” and employers are recognizing that the addition of infertility care to health care benefits is a powerful recruitment tool.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
The field of assisted reproductive technologies (ART) continues to evolve from its first successful birth in 1978 in England, and then in 1981 in the United States. Over the last 6 years, the total number of cycles in the U.S. has increased by 44% to nearly 370,000.
SART membership consists of more than 350 clinics throughout the United States, representing 80% of ART clinics. Over 95% of ART cycles in 2021 in the United States were performed in SART-member clinics.
SART is an invaluable resource for both patients and physicians. Their website includes a “Predict My Success” calculator that allows patients and physicians to enter individualized data to calculate the chance of having a baby over one or more complete cycles of IVF. To help us understand the pregnancy outcome data from ART – cycles per clinic along with national results – I posed the questions below to Amy Sparks, PhD, HCLD, director of the IVF and Andrology Laboratories and the Center for Advanced Reproductive Care at University of Iowa Hospitals and Clinics, Iowa City. Dr. Sparks is past president of SART and former chairperson of the SART Registry committee when the current Clinic Summary Report format was initially released.
Question: The Fertility Clinic Success Rate and Certification Act (FCSRCA) of 1992 mandated that all ART clinics report success rate data to the federal government, through the Centers for Disease Control and Prevention, in a standardized manner. As ART is the only field in medicine to be required to annually report their patient outcomes, that is, all initiated cycles and live births, why do you believe this law was enacted and is limited to reproductive medicine?
Answer: The FCSRCA of 1992 was enacted in response to the lack of open and reliable pregnancy success rate information for patients seeking infertility care using assisted reproductive technologies. Success rates of 25%-50% were being advertised by independent clinics when, nationally, fewer than 15% of ART procedures led to live births. The Federal Trade Commission said such claims were deceptive and filed charges against five clinics, saying they misrepresented their success in helping women become pregnant. The government won one case by court order and the other four cases were settled out of court.
This field of medicine was in the spotlight as the majority of patients lacked insurance coverage for their ART cycles, and there was a strong desire to protect consumers paying out of pocket for relatively low success. Recognizing that the FTC’s mission is to ensure truth in advertising and not regulate medical care, Congress passed the FCSRCA, mandating that all centers providing ART services report all initiated cycles and their outcomes. The CDC was appointed as the agency responsible for collecting cycle data and reporting outcomes. Centers not reporting their cycles are listed as nonreporting centers.
This act also established standards for accreditation of embryology laboratories including personnel and traditional clinical laboratory management requirements. These standards serve as the foundation for embryology laboratory accrediting agencies.
Q: Why have live-birth rates on SART appeared to be focused on “per IVF cycle” as opposed to the CDC reporting of live births “per embryo transfer?”
A: An ART cycle “start” is defined as the initiation of ovarian stimulation with medication that may or may not include administration of exogenous gonadotropins, followed by oocyte retrieval and embryo transfer. Not every patient beginning a cycle will undergo an oocyte retrieval and not all patients who undergo oocyte retrieval have an embryo transfer. The live-birth rates (LBR) for each of these steps of progression in the ART process are available in the SART and CDC reports.
In 2016, SART recognized that practices were foregoing fresh embryo transfer after oocyte retrieval, opting to cryopreserve all embryos to either accommodate genetic testing of the embryos prior to transfer or to avoid embryo transfer to an unfavorable uterine environment. In response to changes in practice and in an effort to deemphasize live birth per transfer, thereby alleviating a potential motivator or pressure for practitioners to transfer multiple embryos, SART moved to a report that displays the cumulative live-birth rate per cycle start for oocyte retrieval. The cumulative live-birth rate per cycle start for oocyte retrieval is the chance of live birth from transfers of embryos derived from the oocyte retrieval and performed within 1 year of the oocyte retrieval.
This change in reporting further reduced the pressure to transfer multiple embryos and encouraged elective, single-embryo transfer. The outcome per transfer is no longer the report’s primary focus.
Q: The latest pregnancy outcomes statistics are from the year 2020 and are finalized by the CDC. Why does the SART website have this same year labeled “preliminary” outcomes?
A: Shortly after the 2016 SART report change, the CDC made similar changes to their report. The difference is that SART provides a “preliminary” report of outcomes within the year of the cycle start for oocyte retrieval. The cumulative outcome is not “finalized” until the following year as transfers may be performed as late as 12 months after the oocyte retrieval.
SART has opted to report both the “preliminary” or interim outcome and the “final” outcome a year later. The CDC has opted to limit their report to “final” outcomes. I’m happy to report that SART recently released the final report for 2021 cycles.
Q: Have national success rates in the United States continued to rise or have they plateaued?
A: It appears that success rates have plateaued; however, we find ourselves at another point where practice patterns and patients’ approach to using ART for family building have changed.
Recognizing the impact of maternal aging on reproductive potential, patients are opting to undergo multiple ART cycles to cryopreserve embryos for family building before they attempt to get pregnant. This family-building path reduces the value of measuring the LBR per cycle start as we may not know the outcome for many years. SART leaders are deliberating intently as to how to best represent this growing patient population in outcome reporting.
Q: Can you comment on the reduction of multiple gestations with the increasing use of single-embryo transfer?
A: The reduction in emphasis on live births per transfer, emphasis on singleton live-birth rates in both the SART and CDC reports, and American Society for Reproductive Medicine practice committee guidelines strongly supporting single embryo transfer have significantly reduced the rate of multiple gestations.
A decade ago, only a third of the transfers were single-embryo transfers and over 25% of live births resulted in a multiple birth. Today, the majority of embryo transfers are elective, single-embryo transfers, and the multiple birth rate has been reduced by nearly 80%. In 2020, 93% of live births from IVF were singletons.
Q: SART offers an online IVF calculator so both patients and physicians can plug in data for an approximate cumulative success rate for up to three IVF cycles. The calculator pools data from all U.S.-reporting IVF centers. Can you explain what an “IVF cycle” is and what patient information is required? Why do success rates increase over time?
A: Each “IVF cycle” is a cycle start for an oocyte retrieval and all transfers of embryos from that cycle within a year of the oocyte retrieval. If the first cycle and subsequent transfers do not lead to a live birth, patients still have a chance to achieve a live birth with a second or third cycle. The success rate increases over time as it reflects the chance of success for a population of patients, with some achieving a live birth after the first cycle and additional patients who achieve success following their third cycle.
Q: The SART IVF calculator can be used with no prior IVF cycles or following an unsuccessful cycle. Are there data to support an estimation of outcome following two or even more unsuccessful cycles?
A: The variables in the SART IVF calculator are based upon the cycle-specific data from patients seeking care at SART member clinics. The current predictor was built with data from cycles performed in 2015-2016. SART is adjusting the predictor and developing a calculator that will be routinely updated, accordingly.
Q: Only approximately 40% of states have some form of infertility coverage law in place; however the number of IVF cycles in the United States continues to increase on an annual basis. What do you think are the driving factors behind this?
A: Advocacy efforts to improve patients’ access to infertility care have included giving patients tools to encourage their employers to include infertility care in their health care benefits package. More recently, the “Great Resignation” has led to the “Great Recruitment” and employers are recognizing that the addition of infertility care to health care benefits is a powerful recruitment tool.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
A clash of expectations
A few weeks ago I asked what changes would have to occur to return urgent care to its former place under the umbrella of the primary care pediatrician. Several responses that I received and the recent story about screenings in this magazine (April 2023) have prompted me to ask the broader question of what is a pediatrician? More specifically, what is the role of a primary care pediatrician?
I think we can agree that a pediatrician is someone who has dedicated his or her training to learning about and then treating the diseases of children. There are pediatricians whose focus is on newborns. There are others who specialize by organ system or by the intensity of the disease (for example, hospitalists and ED physicians). In Great Britain, and to some extent Canada, “paediatricians” serve primarily as consultants to other health care providers. In this country, however, we tend to think of a pediatrician as a frontline primary care physician with general expertise in children. It is those providers (myself included) to whom I address my questions: “What is our role? What is our primary mission?” Are the expectations that we and others have for us realistic given the realities of 21st-century America? And, is our failure to meet some of those expectations contributing to our burnout?
Are we preventionists? I have always thought that one of the things that sets us apart from other specialties is our focus on prevention. We’ve done a pretty good job with infectious diseases thanks to vaccines and antibiotics. But, when I look at the children who grew to be obese adults under my care I have to say that I and my peers have done an abysmal job of prevention. And that is just one example.
Are we educators responsible for helping parents learn what we consider to be the best child-rearing practices? The Latin root of the word “doctor” means teacher. But, education done well is a very time-consuming process. How many of us have time in the office to really teach? Furthermore, some recent studies on managing vaccine deniers suggests that education doesn’t work with people who have long-held beliefs.
Are we data-entry clerks tasked with documenting our every professional step to validate our value to society and the correctness of our methods? It seems that there are some folks who believe we should be.
Are we screeners? TSA agents with white coats and stethoscopes responsible for screening the entire population for potential threats that weren’t obvious to our thoughtful history taking and careful physical examinations?
And finally, are we healers? If you haven’t already disabused yourself of that myth please take a moment to consider the number of cures you have orchestrated in the last 10 years.
The answer is that we can and maybe should be all of those things but we and those who advise us and support us must have reasonable expectations of how difficult it can be to be all those things to all of our patients in the real world of primary care pediatrics. We aren’t social engineers who can level every inequality nor can we orchestrate changes in a society that leans toward enabling unhealthy lifestyles.
The American Academy of Pediatrics must shoulder some of the blame for this discrepancy between expectations and reality. In the Pediatric News article on screening, Susan Kressly, MD, the chair of the American Academy of Pediatrics’s Section on Administration and Practice shares some common-sense observations on how screening can be applied thoughtfully. However, this isn’t how it is usually portrayed in the top-down rollout as each advocacy group releases its next best screening recommendations.
Faced with this clash or expectations I have always chosen to think small. I live in a small town in a small state. I look at each patient and each family, one at a time, with its strengths and its vulnerabilities as a given. I try to educate and prevent as their needs and my time allows. I screen when something makes me feel uncomfortable. Long ago I retired my aspirations as a healer and instead have focussed on being a soother.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
A few weeks ago I asked what changes would have to occur to return urgent care to its former place under the umbrella of the primary care pediatrician. Several responses that I received and the recent story about screenings in this magazine (April 2023) have prompted me to ask the broader question of what is a pediatrician? More specifically, what is the role of a primary care pediatrician?
I think we can agree that a pediatrician is someone who has dedicated his or her training to learning about and then treating the diseases of children. There are pediatricians whose focus is on newborns. There are others who specialize by organ system or by the intensity of the disease (for example, hospitalists and ED physicians). In Great Britain, and to some extent Canada, “paediatricians” serve primarily as consultants to other health care providers. In this country, however, we tend to think of a pediatrician as a frontline primary care physician with general expertise in children. It is those providers (myself included) to whom I address my questions: “What is our role? What is our primary mission?” Are the expectations that we and others have for us realistic given the realities of 21st-century America? And, is our failure to meet some of those expectations contributing to our burnout?
Are we preventionists? I have always thought that one of the things that sets us apart from other specialties is our focus on prevention. We’ve done a pretty good job with infectious diseases thanks to vaccines and antibiotics. But, when I look at the children who grew to be obese adults under my care I have to say that I and my peers have done an abysmal job of prevention. And that is just one example.
Are we educators responsible for helping parents learn what we consider to be the best child-rearing practices? The Latin root of the word “doctor” means teacher. But, education done well is a very time-consuming process. How many of us have time in the office to really teach? Furthermore, some recent studies on managing vaccine deniers suggests that education doesn’t work with people who have long-held beliefs.
Are we data-entry clerks tasked with documenting our every professional step to validate our value to society and the correctness of our methods? It seems that there are some folks who believe we should be.
Are we screeners? TSA agents with white coats and stethoscopes responsible for screening the entire population for potential threats that weren’t obvious to our thoughtful history taking and careful physical examinations?
And finally, are we healers? If you haven’t already disabused yourself of that myth please take a moment to consider the number of cures you have orchestrated in the last 10 years.
The answer is that we can and maybe should be all of those things but we and those who advise us and support us must have reasonable expectations of how difficult it can be to be all those things to all of our patients in the real world of primary care pediatrics. We aren’t social engineers who can level every inequality nor can we orchestrate changes in a society that leans toward enabling unhealthy lifestyles.
The American Academy of Pediatrics must shoulder some of the blame for this discrepancy between expectations and reality. In the Pediatric News article on screening, Susan Kressly, MD, the chair of the American Academy of Pediatrics’s Section on Administration and Practice shares some common-sense observations on how screening can be applied thoughtfully. However, this isn’t how it is usually portrayed in the top-down rollout as each advocacy group releases its next best screening recommendations.
Faced with this clash or expectations I have always chosen to think small. I live in a small town in a small state. I look at each patient and each family, one at a time, with its strengths and its vulnerabilities as a given. I try to educate and prevent as their needs and my time allows. I screen when something makes me feel uncomfortable. Long ago I retired my aspirations as a healer and instead have focussed on being a soother.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
A few weeks ago I asked what changes would have to occur to return urgent care to its former place under the umbrella of the primary care pediatrician. Several responses that I received and the recent story about screenings in this magazine (April 2023) have prompted me to ask the broader question of what is a pediatrician? More specifically, what is the role of a primary care pediatrician?
I think we can agree that a pediatrician is someone who has dedicated his or her training to learning about and then treating the diseases of children. There are pediatricians whose focus is on newborns. There are others who specialize by organ system or by the intensity of the disease (for example, hospitalists and ED physicians). In Great Britain, and to some extent Canada, “paediatricians” serve primarily as consultants to other health care providers. In this country, however, we tend to think of a pediatrician as a frontline primary care physician with general expertise in children. It is those providers (myself included) to whom I address my questions: “What is our role? What is our primary mission?” Are the expectations that we and others have for us realistic given the realities of 21st-century America? And, is our failure to meet some of those expectations contributing to our burnout?
Are we preventionists? I have always thought that one of the things that sets us apart from other specialties is our focus on prevention. We’ve done a pretty good job with infectious diseases thanks to vaccines and antibiotics. But, when I look at the children who grew to be obese adults under my care I have to say that I and my peers have done an abysmal job of prevention. And that is just one example.
Are we educators responsible for helping parents learn what we consider to be the best child-rearing practices? The Latin root of the word “doctor” means teacher. But, education done well is a very time-consuming process. How many of us have time in the office to really teach? Furthermore, some recent studies on managing vaccine deniers suggests that education doesn’t work with people who have long-held beliefs.
Are we data-entry clerks tasked with documenting our every professional step to validate our value to society and the correctness of our methods? It seems that there are some folks who believe we should be.
Are we screeners? TSA agents with white coats and stethoscopes responsible for screening the entire population for potential threats that weren’t obvious to our thoughtful history taking and careful physical examinations?
And finally, are we healers? If you haven’t already disabused yourself of that myth please take a moment to consider the number of cures you have orchestrated in the last 10 years.
The answer is that we can and maybe should be all of those things but we and those who advise us and support us must have reasonable expectations of how difficult it can be to be all those things to all of our patients in the real world of primary care pediatrics. We aren’t social engineers who can level every inequality nor can we orchestrate changes in a society that leans toward enabling unhealthy lifestyles.
The American Academy of Pediatrics must shoulder some of the blame for this discrepancy between expectations and reality. In the Pediatric News article on screening, Susan Kressly, MD, the chair of the American Academy of Pediatrics’s Section on Administration and Practice shares some common-sense observations on how screening can be applied thoughtfully. However, this isn’t how it is usually portrayed in the top-down rollout as each advocacy group releases its next best screening recommendations.
Faced with this clash or expectations I have always chosen to think small. I live in a small town in a small state. I look at each patient and each family, one at a time, with its strengths and its vulnerabilities as a given. I try to educate and prevent as their needs and my time allows. I screen when something makes me feel uncomfortable. Long ago I retired my aspirations as a healer and instead have focussed on being a soother.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Drive, chip, and putt your way to osteoarthritis relief
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
The newest form of mommy shaming: The 'narcissistic mother'
Narcissists appear to be everywhere. A few minutes on the Internet shows the dangers of narcissistic romantic partners, friends, and employers. Identifying and limiting the reach of their manipulative and self-centered endeavors is cast as both urgent and necessary. The destructive powers of the narcissistic mother are viewed as especially in need of remedy, and any bookstore can reveal the risks they pose: “Will I Ever Be Good Enough? Healing the Daughters of Narcissistic Mothers;” “You’re Not Crazy – It’s Your Mother: Freedom for Daughters of Narcissistic Mothers;” “Healing for Daughters of Narcissistic Mothers: A Practical Guide on How to Recover from the Childhood Trauma of Toxic Relationship with Your Mother and How You Can Handle Her Abuse Now As An Adult” – to name just a few (there are more).
As a psychologist specializing in parental estrangement, I (Dr. Coleman) regularly see letters from adult children explaining their discovery-through-therapy that their mother is a narcissist. The proclamation often comes when the therapist has never met the mother. Typically, the discovery is presented as a justification for ending the relationship with the parent. While these mothers could rightly be accused of being anxious, over-involved, depressed, or hurt by the lack of gratitude or reciprocity, the vast majority are not narcissists.
Which begs the question, why are so many being labeled in this way? Are therapists only now discovering the power of narcissistic mothers? Have they always existed, casting their spells upon unwary children? Are those now-grown children only today able to disentangle themselves from the longstanding, pervasive, and harmful influence of these parents, with the help of therapy? Or is this the newest form of mommy shaming as it engages head-on with our Diagnostic and Statistical Manuals?
We believe it is the latter.
Blaming mothers has a long reach. Mothers have been blamed for causing schizophrenia, autism, homosexuality, and effeminacy in men. While we used to call people selfish and “controlling,” narcissism is a more consequential label as it confers diagnostic validity from the mental health profession. Worse, it suggests an individual beyond reach, where the only answer is distance, containment, or estrangement.
The rise of the narcissistic mother comes during a time when, for the past 4 decades, the average working mother spends more time with her children than stay-at-home moms did in the supposed halcyon days of the 1960s’ middle class, before “parenting” was a common term. A variety of economists and sociologists observed that an increase in parental effort became necessary to launch children into adulthood given the retreat of governmental and corporate support for parents that began in the 1980s.
“The financial and emotional burden on families has grown in ways that were almost unimaginable just a half-century ago,” writes the University of Pennsylvania sociologist Frank Furstenberg in “On a New Schedule: Transitions to Adulthood and Family Change.” In addition, a view of children as vulnerable and in need of intense parental investment gained momentum over the course of the 20th century and has continued unabated into the present. As a result, an environment of intense maternal preoccupation, worry, guilt, and involvement with children’s grades, safety, health, and emotional states – referred to as “helicopter” and “tiger” mothering – grew into the norm across the classes.
While prior generations of parents could, by today’s standards, be viewed as being insufficiently involved, today’s parents have become “over-involved” – aided by the ability of parents to be in constant contact with their adult children through technology. While this shift to a more hands-on, more conscientious parenting has been a boon to parent–adult child relationships in the main, the downside has meant, for some, too much of a good thing. From that perspective, pathologizing a mother’s involvement or her expressions of hurt for that child’s lack of availability provides a shield against the child’s feelings of guilt or obligation.
Diagnoses can serve a social purpose: They can allow individuals to use the authority of our profession to decide who to be close to and who to let go. They can provide insulation against feelings of obligation or guilt. They create a way to label behavior as dysfunctional that in other eras or cultures would be considered normal, even valued. To that extent, diagnoses don’t occur in a cultural void. They are inextricably tied to larger ideals, be they individualistic – as exists in the United States – or collectivist, as exists in many other parts of the world.
While we have decided what parents owe our children, it is unclear what parents might ask in return. To that end, mothers who want more interest, availability, or gratitude today are vulnerable to being cast as selfish, uncaring, needy, and controlling. They can now be viewed as failing in their task of selfless devotion. Their desires for closeness or repair can be regarded as incompatible with the quest for the adult child’s self-fulfillment and identity; her identification with her children too great a barrier to their individuation.
There may well be good reasons to estrange family members for their intolerable behaviors, especially ones who have threatened personal safety. Yet, while there are plenty of problematic parents, few meet the diagnostic criteria of narcissistic personality disorder. More important, such labels can discourage a discussion of boundaries that both the parents and the adult children might find acceptable – which sometimes means asking family members to tolerate behavior or individuals not to their liking.
Diagnoses carry enormous social weight and can facilitate estrangements or negativity to mothers that are far more workable than our patients’ characterization of them might lead them or us to believe. ; it devalues their years of love and dedication, however flawed; and it weakens the fabric of connection that could otherwise exist. Rather than provide a path toward compassion or understanding, “narcissistic mother” just becomes the latest form of mommy shaming.
Dr. Coleman is a clinical psychologist and author of “Rules of Estrangement: Why Adult Children Cut Ties and How to Heal the Conflict” (New York: Penguin Random House, 2021). Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is an assistant professor of psychiatry and behavioral sciences at Johns Hopkins University in Baltimore.
Narcissists appear to be everywhere. A few minutes on the Internet shows the dangers of narcissistic romantic partners, friends, and employers. Identifying and limiting the reach of their manipulative and self-centered endeavors is cast as both urgent and necessary. The destructive powers of the narcissistic mother are viewed as especially in need of remedy, and any bookstore can reveal the risks they pose: “Will I Ever Be Good Enough? Healing the Daughters of Narcissistic Mothers;” “You’re Not Crazy – It’s Your Mother: Freedom for Daughters of Narcissistic Mothers;” “Healing for Daughters of Narcissistic Mothers: A Practical Guide on How to Recover from the Childhood Trauma of Toxic Relationship with Your Mother and How You Can Handle Her Abuse Now As An Adult” – to name just a few (there are more).
As a psychologist specializing in parental estrangement, I (Dr. Coleman) regularly see letters from adult children explaining their discovery-through-therapy that their mother is a narcissist. The proclamation often comes when the therapist has never met the mother. Typically, the discovery is presented as a justification for ending the relationship with the parent. While these mothers could rightly be accused of being anxious, over-involved, depressed, or hurt by the lack of gratitude or reciprocity, the vast majority are not narcissists.
Which begs the question, why are so many being labeled in this way? Are therapists only now discovering the power of narcissistic mothers? Have they always existed, casting their spells upon unwary children? Are those now-grown children only today able to disentangle themselves from the longstanding, pervasive, and harmful influence of these parents, with the help of therapy? Or is this the newest form of mommy shaming as it engages head-on with our Diagnostic and Statistical Manuals?
We believe it is the latter.
Blaming mothers has a long reach. Mothers have been blamed for causing schizophrenia, autism, homosexuality, and effeminacy in men. While we used to call people selfish and “controlling,” narcissism is a more consequential label as it confers diagnostic validity from the mental health profession. Worse, it suggests an individual beyond reach, where the only answer is distance, containment, or estrangement.
The rise of the narcissistic mother comes during a time when, for the past 4 decades, the average working mother spends more time with her children than stay-at-home moms did in the supposed halcyon days of the 1960s’ middle class, before “parenting” was a common term. A variety of economists and sociologists observed that an increase in parental effort became necessary to launch children into adulthood given the retreat of governmental and corporate support for parents that began in the 1980s.
“The financial and emotional burden on families has grown in ways that were almost unimaginable just a half-century ago,” writes the University of Pennsylvania sociologist Frank Furstenberg in “On a New Schedule: Transitions to Adulthood and Family Change.” In addition, a view of children as vulnerable and in need of intense parental investment gained momentum over the course of the 20th century and has continued unabated into the present. As a result, an environment of intense maternal preoccupation, worry, guilt, and involvement with children’s grades, safety, health, and emotional states – referred to as “helicopter” and “tiger” mothering – grew into the norm across the classes.
While prior generations of parents could, by today’s standards, be viewed as being insufficiently involved, today’s parents have become “over-involved” – aided by the ability of parents to be in constant contact with their adult children through technology. While this shift to a more hands-on, more conscientious parenting has been a boon to parent–adult child relationships in the main, the downside has meant, for some, too much of a good thing. From that perspective, pathologizing a mother’s involvement or her expressions of hurt for that child’s lack of availability provides a shield against the child’s feelings of guilt or obligation.
Diagnoses can serve a social purpose: They can allow individuals to use the authority of our profession to decide who to be close to and who to let go. They can provide insulation against feelings of obligation or guilt. They create a way to label behavior as dysfunctional that in other eras or cultures would be considered normal, even valued. To that extent, diagnoses don’t occur in a cultural void. They are inextricably tied to larger ideals, be they individualistic – as exists in the United States – or collectivist, as exists in many other parts of the world.
While we have decided what parents owe our children, it is unclear what parents might ask in return. To that end, mothers who want more interest, availability, or gratitude today are vulnerable to being cast as selfish, uncaring, needy, and controlling. They can now be viewed as failing in their task of selfless devotion. Their desires for closeness or repair can be regarded as incompatible with the quest for the adult child’s self-fulfillment and identity; her identification with her children too great a barrier to their individuation.
There may well be good reasons to estrange family members for their intolerable behaviors, especially ones who have threatened personal safety. Yet, while there are plenty of problematic parents, few meet the diagnostic criteria of narcissistic personality disorder. More important, such labels can discourage a discussion of boundaries that both the parents and the adult children might find acceptable – which sometimes means asking family members to tolerate behavior or individuals not to their liking.
Diagnoses carry enormous social weight and can facilitate estrangements or negativity to mothers that are far more workable than our patients’ characterization of them might lead them or us to believe. ; it devalues their years of love and dedication, however flawed; and it weakens the fabric of connection that could otherwise exist. Rather than provide a path toward compassion or understanding, “narcissistic mother” just becomes the latest form of mommy shaming.
Dr. Coleman is a clinical psychologist and author of “Rules of Estrangement: Why Adult Children Cut Ties and How to Heal the Conflict” (New York: Penguin Random House, 2021). Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is an assistant professor of psychiatry and behavioral sciences at Johns Hopkins University in Baltimore.
Narcissists appear to be everywhere. A few minutes on the Internet shows the dangers of narcissistic romantic partners, friends, and employers. Identifying and limiting the reach of their manipulative and self-centered endeavors is cast as both urgent and necessary. The destructive powers of the narcissistic mother are viewed as especially in need of remedy, and any bookstore can reveal the risks they pose: “Will I Ever Be Good Enough? Healing the Daughters of Narcissistic Mothers;” “You’re Not Crazy – It’s Your Mother: Freedom for Daughters of Narcissistic Mothers;” “Healing for Daughters of Narcissistic Mothers: A Practical Guide on How to Recover from the Childhood Trauma of Toxic Relationship with Your Mother and How You Can Handle Her Abuse Now As An Adult” – to name just a few (there are more).
As a psychologist specializing in parental estrangement, I (Dr. Coleman) regularly see letters from adult children explaining their discovery-through-therapy that their mother is a narcissist. The proclamation often comes when the therapist has never met the mother. Typically, the discovery is presented as a justification for ending the relationship with the parent. While these mothers could rightly be accused of being anxious, over-involved, depressed, or hurt by the lack of gratitude or reciprocity, the vast majority are not narcissists.
Which begs the question, why are so many being labeled in this way? Are therapists only now discovering the power of narcissistic mothers? Have they always existed, casting their spells upon unwary children? Are those now-grown children only today able to disentangle themselves from the longstanding, pervasive, and harmful influence of these parents, with the help of therapy? Or is this the newest form of mommy shaming as it engages head-on with our Diagnostic and Statistical Manuals?
We believe it is the latter.
Blaming mothers has a long reach. Mothers have been blamed for causing schizophrenia, autism, homosexuality, and effeminacy in men. While we used to call people selfish and “controlling,” narcissism is a more consequential label as it confers diagnostic validity from the mental health profession. Worse, it suggests an individual beyond reach, where the only answer is distance, containment, or estrangement.
The rise of the narcissistic mother comes during a time when, for the past 4 decades, the average working mother spends more time with her children than stay-at-home moms did in the supposed halcyon days of the 1960s’ middle class, before “parenting” was a common term. A variety of economists and sociologists observed that an increase in parental effort became necessary to launch children into adulthood given the retreat of governmental and corporate support for parents that began in the 1980s.
“The financial and emotional burden on families has grown in ways that were almost unimaginable just a half-century ago,” writes the University of Pennsylvania sociologist Frank Furstenberg in “On a New Schedule: Transitions to Adulthood and Family Change.” In addition, a view of children as vulnerable and in need of intense parental investment gained momentum over the course of the 20th century and has continued unabated into the present. As a result, an environment of intense maternal preoccupation, worry, guilt, and involvement with children’s grades, safety, health, and emotional states – referred to as “helicopter” and “tiger” mothering – grew into the norm across the classes.
While prior generations of parents could, by today’s standards, be viewed as being insufficiently involved, today’s parents have become “over-involved” – aided by the ability of parents to be in constant contact with their adult children through technology. While this shift to a more hands-on, more conscientious parenting has been a boon to parent–adult child relationships in the main, the downside has meant, for some, too much of a good thing. From that perspective, pathologizing a mother’s involvement or her expressions of hurt for that child’s lack of availability provides a shield against the child’s feelings of guilt or obligation.
Diagnoses can serve a social purpose: They can allow individuals to use the authority of our profession to decide who to be close to and who to let go. They can provide insulation against feelings of obligation or guilt. They create a way to label behavior as dysfunctional that in other eras or cultures would be considered normal, even valued. To that extent, diagnoses don’t occur in a cultural void. They are inextricably tied to larger ideals, be they individualistic – as exists in the United States – or collectivist, as exists in many other parts of the world.
While we have decided what parents owe our children, it is unclear what parents might ask in return. To that end, mothers who want more interest, availability, or gratitude today are vulnerable to being cast as selfish, uncaring, needy, and controlling. They can now be viewed as failing in their task of selfless devotion. Their desires for closeness or repair can be regarded as incompatible with the quest for the adult child’s self-fulfillment and identity; her identification with her children too great a barrier to their individuation.
There may well be good reasons to estrange family members for their intolerable behaviors, especially ones who have threatened personal safety. Yet, while there are plenty of problematic parents, few meet the diagnostic criteria of narcissistic personality disorder. More important, such labels can discourage a discussion of boundaries that both the parents and the adult children might find acceptable – which sometimes means asking family members to tolerate behavior or individuals not to their liking.
Diagnoses carry enormous social weight and can facilitate estrangements or negativity to mothers that are far more workable than our patients’ characterization of them might lead them or us to believe. ; it devalues their years of love and dedication, however flawed; and it weakens the fabric of connection that could otherwise exist. Rather than provide a path toward compassion or understanding, “narcissistic mother” just becomes the latest form of mommy shaming.
Dr. Coleman is a clinical psychologist and author of “Rules of Estrangement: Why Adult Children Cut Ties and How to Heal the Conflict” (New York: Penguin Random House, 2021). Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is an assistant professor of psychiatry and behavioral sciences at Johns Hopkins University in Baltimore.
Child’s health improves by applying new obesity guidelines
At age 15 years, Maya was referred by her primary care provider to our pediatric obesity center. She weighed 151 kg and had a body mass index (BMI) over 48 kg/m2. One year earlier, she had been diagnosed with hypertension and prediabetes.
A review of her growth charts showed that she had been in the 95th percentile at age 8 years. Her weight had steadily risen, with an exponential increase of 55 lb between 2020 and 2022, during the COVID-19 pandemic. Her primary care provider monitored her from age 8 to 12 years, providing nutrition and physical activity counseling.
In February, the American Academy of Pediatrics released new clinical practice guidelines for managing childhood obesity. A better understanding of the pathophysiology has challenged the old-worn concept of lack of will power and personal responsibility as the cause of obesity, which has been the basis for weight-related bias and stigma. The updated guidelines have also been influenced by lifestyle intervention studies, the US Food and Drug Administration approval of new anti-obesity medications, and the 2013 designation of obesity as a disease by the American Medical Association.
We used these updated guidelines in our approach to treating Maya.
Starting with the assessment
In the new AAP guidelines, assessing the genetic, environmental, and social-determinant risks for obesity form the basis for evaluation and intervention. Following this approach, we conducted a complete medical evaluation of Maya, including a review of her symptoms and her family history along with a physical examination to assess for comorbidities and other cause of obesity (for example, genetic, hypothyroidism).
We also collected information regarding her diet and behaviors (for example, drinking sweet beverages, fruit and vegetable intake, parent feeding style, portion sizes, emotional eating, hyperphagia), physical activity behaviors (for example, physical education, organized sports), screen time, social drivers of health (for example, food insecurity, neighborhood, school environment), family and household factors (for example, family composition, support, number of caregivers, parenting style) and mental and physical health (autism, attention-deficit/hyperactivity disorder, history of being bullied, developmental and physical disabilities). Because Maya had a BMI of 48, she met the criterion for severe obesity, which is having a BMI at least 120% of the 95th percentile.
The guidelines use BMI as a criterion for screening for obesity because it is inexpensive and easy to obtain in the clinic setting. The Centers for Disease Control and Prevention growth chart uses BMI as well. Recently, there has been controversy about solely using BMI to define obesity, which is a point that the guidelines address by emphasizing evaluation of the whole child along with BMI to make a diagnosis of obesity.
The child’s age and the severity of their obesity drive the evaluation for comorbidities and treatment. In children aged 10 years or older, pediatricians and other primary care providers should evaluate for lipid abnormalities, abnormal glucose metabolism, and abnormal liver function in children and adolescents with obesity (BMI ≥ 95th percentile).
Maya presented with snoring, early-morning headaches, daytime sleepiness, and abdominal pain. A sleep study revealed an apnea-hypopnea index of 15, indicating obstructive sleep apnea, and she was placed on a continuous positive airway pressure machine.
Her laboratory studies showed elevated triglycerides of 169 mg/dL and abnormal ALT (123 IU/L). Potential causes of elevated liver function test results (such as abnormal ceruloplasmin levels or infectious or autoimmune hepatitis) were excluded, and a liver ultrasound with elastography indicated steatohepatitis. Maya was referred to gastroenterology for nonalcoholic fatty liver disease.
Maya experienced depressive symptoms, including difficulty with peer relationships and declining academic performance. Her Patient Health Questionnaire–9 score was 21, with a moderate impact on her daily functioning. Prior attempts at counseling had been sporadic and not helpful. She was diagnosed with intermittent moderate clinical depression, started on a selective serotonin reuptake inhibitor, and resumed counseling with a new therapist.
Considering treatment options
Based on shared decision-making, our team began a more intensive lifestyle behavior treatment as recommended in the updated guidelines. Maya chose to decrease sugar-sweetened beverages as her initial nutrition goal, a change that can lead to a reduction of liver function test results and triglycerides, even in the absence of weight loss.
As emphasized in the guidelines, we stressed the importance of managing obesity and comorbidities concurrently to the family. In addition to lifestyle behavior intervention, once her mental health stabilized, Maya and her mother opted for bariatric surgery. Sleeve gastrectomy was elected because she met the criteria.
If the child already has obesity, the guidelines discourage watchful waiting (that is, the expectation that the child will grow into their weight) as Maya’s primary care provider had done when she was younger. The staged treatment approach where progressively more intensive interventions are adopted (a hallmark of the 2007 guidelines) is no longer recommended. Rather, the primary care provider should offer treatment options guided by age, severity of obesity, and comorbidities.
Maya completed a bariatric preoperative program, extensive mental health evaluation, and tolerated the sleeve gastrectomy well with no complications. At her 6-month postoperative visit, she had lost 99 lb (45 kg) since the surgery, with an 18% decline in BMI. She is taking daily multivitamins as well as calcium and vitamin D. She continues to incorporate healthy eating into her life, with a focus on adequate protein intake and is exercising three to four times per week in the apartment complex gym. She reports better physical and mental health, her school performance has improved, and she still receives regular counseling.
Maya’s story outlines the benefits of early and intensive intervention as recommended by the new AAP guidelines. The shift from some of the earlier recommendations is partly driven by the persistence of childhood obesity into adulthood, especially for older children with serious psychosocial and physical comorbidities. Hopefully by implementing the new guidelines, the physician can provide empathetic, bias-free, and effective care that recognizes the needs and environment of the whole child.
Dr. Salhah is a pediatric endocrinology fellow at Nationwide Children’s Hospital, Columbus, Ohio. Dr. Eneli is director of the Center for Healthy Weight and Nutrition at Nationwide Children’s Hospital. Dr. Salhah reported no conflicts of interest. Dr. Eneli reported receiving research grants and income from the National Institutes of Health, the AAP, and the National Academy of Medicine.
A version of this article first appeared on Medscape.com.
At age 15 years, Maya was referred by her primary care provider to our pediatric obesity center. She weighed 151 kg and had a body mass index (BMI) over 48 kg/m2. One year earlier, she had been diagnosed with hypertension and prediabetes.
A review of her growth charts showed that she had been in the 95th percentile at age 8 years. Her weight had steadily risen, with an exponential increase of 55 lb between 2020 and 2022, during the COVID-19 pandemic. Her primary care provider monitored her from age 8 to 12 years, providing nutrition and physical activity counseling.
In February, the American Academy of Pediatrics released new clinical practice guidelines for managing childhood obesity. A better understanding of the pathophysiology has challenged the old-worn concept of lack of will power and personal responsibility as the cause of obesity, which has been the basis for weight-related bias and stigma. The updated guidelines have also been influenced by lifestyle intervention studies, the US Food and Drug Administration approval of new anti-obesity medications, and the 2013 designation of obesity as a disease by the American Medical Association.
We used these updated guidelines in our approach to treating Maya.
Starting with the assessment
In the new AAP guidelines, assessing the genetic, environmental, and social-determinant risks for obesity form the basis for evaluation and intervention. Following this approach, we conducted a complete medical evaluation of Maya, including a review of her symptoms and her family history along with a physical examination to assess for comorbidities and other cause of obesity (for example, genetic, hypothyroidism).
We also collected information regarding her diet and behaviors (for example, drinking sweet beverages, fruit and vegetable intake, parent feeding style, portion sizes, emotional eating, hyperphagia), physical activity behaviors (for example, physical education, organized sports), screen time, social drivers of health (for example, food insecurity, neighborhood, school environment), family and household factors (for example, family composition, support, number of caregivers, parenting style) and mental and physical health (autism, attention-deficit/hyperactivity disorder, history of being bullied, developmental and physical disabilities). Because Maya had a BMI of 48, she met the criterion for severe obesity, which is having a BMI at least 120% of the 95th percentile.
The guidelines use BMI as a criterion for screening for obesity because it is inexpensive and easy to obtain in the clinic setting. The Centers for Disease Control and Prevention growth chart uses BMI as well. Recently, there has been controversy about solely using BMI to define obesity, which is a point that the guidelines address by emphasizing evaluation of the whole child along with BMI to make a diagnosis of obesity.
The child’s age and the severity of their obesity drive the evaluation for comorbidities and treatment. In children aged 10 years or older, pediatricians and other primary care providers should evaluate for lipid abnormalities, abnormal glucose metabolism, and abnormal liver function in children and adolescents with obesity (BMI ≥ 95th percentile).
Maya presented with snoring, early-morning headaches, daytime sleepiness, and abdominal pain. A sleep study revealed an apnea-hypopnea index of 15, indicating obstructive sleep apnea, and she was placed on a continuous positive airway pressure machine.
Her laboratory studies showed elevated triglycerides of 169 mg/dL and abnormal ALT (123 IU/L). Potential causes of elevated liver function test results (such as abnormal ceruloplasmin levels or infectious or autoimmune hepatitis) were excluded, and a liver ultrasound with elastography indicated steatohepatitis. Maya was referred to gastroenterology for nonalcoholic fatty liver disease.
Maya experienced depressive symptoms, including difficulty with peer relationships and declining academic performance. Her Patient Health Questionnaire–9 score was 21, with a moderate impact on her daily functioning. Prior attempts at counseling had been sporadic and not helpful. She was diagnosed with intermittent moderate clinical depression, started on a selective serotonin reuptake inhibitor, and resumed counseling with a new therapist.
Considering treatment options
Based on shared decision-making, our team began a more intensive lifestyle behavior treatment as recommended in the updated guidelines. Maya chose to decrease sugar-sweetened beverages as her initial nutrition goal, a change that can lead to a reduction of liver function test results and triglycerides, even in the absence of weight loss.
As emphasized in the guidelines, we stressed the importance of managing obesity and comorbidities concurrently to the family. In addition to lifestyle behavior intervention, once her mental health stabilized, Maya and her mother opted for bariatric surgery. Sleeve gastrectomy was elected because she met the criteria.
If the child already has obesity, the guidelines discourage watchful waiting (that is, the expectation that the child will grow into their weight) as Maya’s primary care provider had done when she was younger. The staged treatment approach where progressively more intensive interventions are adopted (a hallmark of the 2007 guidelines) is no longer recommended. Rather, the primary care provider should offer treatment options guided by age, severity of obesity, and comorbidities.
Maya completed a bariatric preoperative program, extensive mental health evaluation, and tolerated the sleeve gastrectomy well with no complications. At her 6-month postoperative visit, she had lost 99 lb (45 kg) since the surgery, with an 18% decline in BMI. She is taking daily multivitamins as well as calcium and vitamin D. She continues to incorporate healthy eating into her life, with a focus on adequate protein intake and is exercising three to four times per week in the apartment complex gym. She reports better physical and mental health, her school performance has improved, and she still receives regular counseling.
Maya’s story outlines the benefits of early and intensive intervention as recommended by the new AAP guidelines. The shift from some of the earlier recommendations is partly driven by the persistence of childhood obesity into adulthood, especially for older children with serious psychosocial and physical comorbidities. Hopefully by implementing the new guidelines, the physician can provide empathetic, bias-free, and effective care that recognizes the needs and environment of the whole child.
Dr. Salhah is a pediatric endocrinology fellow at Nationwide Children’s Hospital, Columbus, Ohio. Dr. Eneli is director of the Center for Healthy Weight and Nutrition at Nationwide Children’s Hospital. Dr. Salhah reported no conflicts of interest. Dr. Eneli reported receiving research grants and income from the National Institutes of Health, the AAP, and the National Academy of Medicine.
A version of this article first appeared on Medscape.com.
At age 15 years, Maya was referred by her primary care provider to our pediatric obesity center. She weighed 151 kg and had a body mass index (BMI) over 48 kg/m2. One year earlier, she had been diagnosed with hypertension and prediabetes.
A review of her growth charts showed that she had been in the 95th percentile at age 8 years. Her weight had steadily risen, with an exponential increase of 55 lb between 2020 and 2022, during the COVID-19 pandemic. Her primary care provider monitored her from age 8 to 12 years, providing nutrition and physical activity counseling.
In February, the American Academy of Pediatrics released new clinical practice guidelines for managing childhood obesity. A better understanding of the pathophysiology has challenged the old-worn concept of lack of will power and personal responsibility as the cause of obesity, which has been the basis for weight-related bias and stigma. The updated guidelines have also been influenced by lifestyle intervention studies, the US Food and Drug Administration approval of new anti-obesity medications, and the 2013 designation of obesity as a disease by the American Medical Association.
We used these updated guidelines in our approach to treating Maya.
Starting with the assessment
In the new AAP guidelines, assessing the genetic, environmental, and social-determinant risks for obesity form the basis for evaluation and intervention. Following this approach, we conducted a complete medical evaluation of Maya, including a review of her symptoms and her family history along with a physical examination to assess for comorbidities and other cause of obesity (for example, genetic, hypothyroidism).
We also collected information regarding her diet and behaviors (for example, drinking sweet beverages, fruit and vegetable intake, parent feeding style, portion sizes, emotional eating, hyperphagia), physical activity behaviors (for example, physical education, organized sports), screen time, social drivers of health (for example, food insecurity, neighborhood, school environment), family and household factors (for example, family composition, support, number of caregivers, parenting style) and mental and physical health (autism, attention-deficit/hyperactivity disorder, history of being bullied, developmental and physical disabilities). Because Maya had a BMI of 48, she met the criterion for severe obesity, which is having a BMI at least 120% of the 95th percentile.
The guidelines use BMI as a criterion for screening for obesity because it is inexpensive and easy to obtain in the clinic setting. The Centers for Disease Control and Prevention growth chart uses BMI as well. Recently, there has been controversy about solely using BMI to define obesity, which is a point that the guidelines address by emphasizing evaluation of the whole child along with BMI to make a diagnosis of obesity.
The child’s age and the severity of their obesity drive the evaluation for comorbidities and treatment. In children aged 10 years or older, pediatricians and other primary care providers should evaluate for lipid abnormalities, abnormal glucose metabolism, and abnormal liver function in children and adolescents with obesity (BMI ≥ 95th percentile).
Maya presented with snoring, early-morning headaches, daytime sleepiness, and abdominal pain. A sleep study revealed an apnea-hypopnea index of 15, indicating obstructive sleep apnea, and she was placed on a continuous positive airway pressure machine.
Her laboratory studies showed elevated triglycerides of 169 mg/dL and abnormal ALT (123 IU/L). Potential causes of elevated liver function test results (such as abnormal ceruloplasmin levels or infectious or autoimmune hepatitis) were excluded, and a liver ultrasound with elastography indicated steatohepatitis. Maya was referred to gastroenterology for nonalcoholic fatty liver disease.
Maya experienced depressive symptoms, including difficulty with peer relationships and declining academic performance. Her Patient Health Questionnaire–9 score was 21, with a moderate impact on her daily functioning. Prior attempts at counseling had been sporadic and not helpful. She was diagnosed with intermittent moderate clinical depression, started on a selective serotonin reuptake inhibitor, and resumed counseling with a new therapist.
Considering treatment options
Based on shared decision-making, our team began a more intensive lifestyle behavior treatment as recommended in the updated guidelines. Maya chose to decrease sugar-sweetened beverages as her initial nutrition goal, a change that can lead to a reduction of liver function test results and triglycerides, even in the absence of weight loss.
As emphasized in the guidelines, we stressed the importance of managing obesity and comorbidities concurrently to the family. In addition to lifestyle behavior intervention, once her mental health stabilized, Maya and her mother opted for bariatric surgery. Sleeve gastrectomy was elected because she met the criteria.
If the child already has obesity, the guidelines discourage watchful waiting (that is, the expectation that the child will grow into their weight) as Maya’s primary care provider had done when she was younger. The staged treatment approach where progressively more intensive interventions are adopted (a hallmark of the 2007 guidelines) is no longer recommended. Rather, the primary care provider should offer treatment options guided by age, severity of obesity, and comorbidities.
Maya completed a bariatric preoperative program, extensive mental health evaluation, and tolerated the sleeve gastrectomy well with no complications. At her 6-month postoperative visit, she had lost 99 lb (45 kg) since the surgery, with an 18% decline in BMI. She is taking daily multivitamins as well as calcium and vitamin D. She continues to incorporate healthy eating into her life, with a focus on adequate protein intake and is exercising three to four times per week in the apartment complex gym. She reports better physical and mental health, her school performance has improved, and she still receives regular counseling.
Maya’s story outlines the benefits of early and intensive intervention as recommended by the new AAP guidelines. The shift from some of the earlier recommendations is partly driven by the persistence of childhood obesity into adulthood, especially for older children with serious psychosocial and physical comorbidities. Hopefully by implementing the new guidelines, the physician can provide empathetic, bias-free, and effective care that recognizes the needs and environment of the whole child.
Dr. Salhah is a pediatric endocrinology fellow at Nationwide Children’s Hospital, Columbus, Ohio. Dr. Eneli is director of the Center for Healthy Weight and Nutrition at Nationwide Children’s Hospital. Dr. Salhah reported no conflicts of interest. Dr. Eneli reported receiving research grants and income from the National Institutes of Health, the AAP, and the National Academy of Medicine.
A version of this article first appeared on Medscape.com.



















